User login
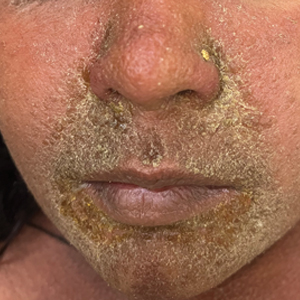
Children With Severe Atopic Dermatitis Catch Up on Growth With Dupilumab
AMSTERDAM — , revealed a post hoc trial analysis.
The research was presented at the European Academy of Dermatology and Venereology (EADV) 2024 Congress.
The trial included a “rigorously selected … well-characterized, well-studied” population of children aged 6-11 years, said presenter Alan D. Irvine, MD, DSc, professor of dermatology, Trinity College Dublin, Ireland.
It showed that “severe atopic dermatitis does cause restriction of growth, as well as a higher weight, and therefore obviously a higher BMI [body mass index].”
He continued, however, that children at the lower percentiles of height receiving prompt treatment with dupilumab (Dupixent) “were able to rapidly move through the centiles over the 16 weeks of the study, and that may be the window for catch-up growth … when children are growing rapidly.”
Anna Yasmine Kirkorian, MD, chief of dermatology, Children’s National Hospital, Washington, DC, who was not involved in the study, said that she was “surprised” at the degree of growth achieved over the study period, as height is not something that jumps up “overnight.”
“On the other hand, it fits with my experience with children who’ve had the brakes on all of their life due to inflammation, whether it be height, going to school, sleeping — everything is sort of put on pause by this terrible inflammatory process,” she said.
“When you take the brakes off, they get to be who they are going to be,” Kirkorian added. “So I was surprised by the speed of it, but not by the fact that height was acquired.”
Her belief is that in the pre-dupilumab era, severe atopic dermatitis was often insufficiently controlled, so children were “smaller than you would predict from parental height,” and the treatment is “allowing them to reach their genetic potential.”
Post Hoc Analysis
In his presentation, Irvine emphasized that it has been clearly demonstrated that adolescents with moderate and severe atopic dermatitis have a significantly higher likelihood of being below the 25th percentile of height on growth reference charts.
Such children are also at a higher risk of having low bone mineral density and low serum alkaline phosphatase (ALP) levels . While data presented at the EADV 2023 Congress showed that dupilumab significantly increased serum levels of bone ALP compared with placebo, the underlying mechanism remains unclear.
For the current analysis, Irvine and colleagues determined that the proportion of children aged 6-11 years with severe atopic dermatitis and lower stature reach a ≥ 5 centile improvement in height following 16 weeks of dupilumab treatment.
They examined data from the LIBERTY AD PEDS trial, in which patients aged 6-11 years with severe atopic dermatitis were randomized to 300 mg dupilumab every 4 weeks or placebo along with a mild or moderately potent topical corticosteroid. The study found that, overall, dupilumab was associated with significant improvements in signs, symptoms, and quality of life compared with placebo.
Height measures at baseline revealed that “more boys and more girls were below the 50th centile than you would predict for a healthy, normal control population,” Irvine said. “If we look at weight, we see the opposite,” he continued, “with a disproportionate number of boys and girls who are above the 50th centile for weight at baseline.”
Consequently, “we’re seeing these children who are shorter and heavier than the predicted healthy weight range and, as a result, obviously have higher BMI,” Irvine noted, with 67% girls and 62% boys found to have a higher BMI than normal for their age.
After 16 weeks of treatment with dupilumab, there was a much greater gain in height than that seen among those on placebo, with the most pronounced effect seen in children who had the lowest height at baseline. Indeed, among children in the lowest 25% height percentile at baseline, 30.6% on dupilumab vs 11.9% on placebo experienced an increase in height of 5 centiles or more(P < .05).
“This reflects what we see in clinical practice,” Irvine said. “Children often grow dramatically on treatment for atopic dermatitis.”
Among patients with a baseline height below the 30th percentile, 31.9% treated with dupilumab vs 11.1% treated with placebo gained at least 5 centiles in height. The figures for children below the 40th height percentile at baseline were 31.3% vs 15.5% (P < .05 for both).
Although there remained a marked difference in the proportion of children below the 50th height percentile at baseline gaining 5 centiles or more in height, at 29.0% with dupilumab versus 15.7% with placebo, it was no longer significant.
“So the effect of catch-up growth, or growth through the centiles, is most marked in those who are in the 40th centile or below,” Irvine said, indicating that the “more growth restricted kids have much more potential to catch up.”
‘Convincing’ Data
Overall, Kirkorian said in the interview, the data are “convincing” and support her view that severe atopic dermatitis is a “terrible chronic disease that we really underappreciate.” Atopic dermatitis, she added, “should get the respect that any severe chronic illness would have, whether that be arthritis, diabetes, or cardiac disease, because it is a systemic disorder that … profoundly affects quality of life, every minute of every day.”
However, “we don’t get all the referrals we should, until the child has suffered for years and years, and the family has suffered,” as there is a bias that it can be outgrown — although not everybody does — and it “doesn’t look as conspicuous as other chronic skin disorders,” such as psoriasis.
“Now with this study,” Kirkorian said, “it gives us a really compelling point to make to parents, to the community, and to insurers that not only are we affecting the quality of life from the itch standpoint [with dupilumab] but we may have long profound effects on growth and bone health.”
The research was sponsored by Sanofi and Regeneron Pharmaceuticals. Irvine declared relationships with AbbVie, Arena Pharmaceuticals, BenevolentAI, Chugai Pharmaceutical, Dermavant, Eli Lily, Genentech, LEO Pharma, Menlo Therapeutics, Novartis, Pfizer, Regeneron, Sanofi, UCB, DS Biopharma, and Inflazome. Kirkorian declared relationships with Dermavant, Verrica Pharmaceuticals, Pfizer, and Incyte.
A version of this article first appeared on Medscape.com.
AMSTERDAM — , revealed a post hoc trial analysis.
The research was presented at the European Academy of Dermatology and Venereology (EADV) 2024 Congress.
The trial included a “rigorously selected … well-characterized, well-studied” population of children aged 6-11 years, said presenter Alan D. Irvine, MD, DSc, professor of dermatology, Trinity College Dublin, Ireland.
It showed that “severe atopic dermatitis does cause restriction of growth, as well as a higher weight, and therefore obviously a higher BMI [body mass index].”
He continued, however, that children at the lower percentiles of height receiving prompt treatment with dupilumab (Dupixent) “were able to rapidly move through the centiles over the 16 weeks of the study, and that may be the window for catch-up growth … when children are growing rapidly.”
Anna Yasmine Kirkorian, MD, chief of dermatology, Children’s National Hospital, Washington, DC, who was not involved in the study, said that she was “surprised” at the degree of growth achieved over the study period, as height is not something that jumps up “overnight.”
“On the other hand, it fits with my experience with children who’ve had the brakes on all of their life due to inflammation, whether it be height, going to school, sleeping — everything is sort of put on pause by this terrible inflammatory process,” she said.
“When you take the brakes off, they get to be who they are going to be,” Kirkorian added. “So I was surprised by the speed of it, but not by the fact that height was acquired.”
Her belief is that in the pre-dupilumab era, severe atopic dermatitis was often insufficiently controlled, so children were “smaller than you would predict from parental height,” and the treatment is “allowing them to reach their genetic potential.”
Post Hoc Analysis
In his presentation, Irvine emphasized that it has been clearly demonstrated that adolescents with moderate and severe atopic dermatitis have a significantly higher likelihood of being below the 25th percentile of height on growth reference charts.
Such children are also at a higher risk of having low bone mineral density and low serum alkaline phosphatase (ALP) levels . While data presented at the EADV 2023 Congress showed that dupilumab significantly increased serum levels of bone ALP compared with placebo, the underlying mechanism remains unclear.
For the current analysis, Irvine and colleagues determined that the proportion of children aged 6-11 years with severe atopic dermatitis and lower stature reach a ≥ 5 centile improvement in height following 16 weeks of dupilumab treatment.
They examined data from the LIBERTY AD PEDS trial, in which patients aged 6-11 years with severe atopic dermatitis were randomized to 300 mg dupilumab every 4 weeks or placebo along with a mild or moderately potent topical corticosteroid. The study found that, overall, dupilumab was associated with significant improvements in signs, symptoms, and quality of life compared with placebo.
Height measures at baseline revealed that “more boys and more girls were below the 50th centile than you would predict for a healthy, normal control population,” Irvine said. “If we look at weight, we see the opposite,” he continued, “with a disproportionate number of boys and girls who are above the 50th centile for weight at baseline.”
Consequently, “we’re seeing these children who are shorter and heavier than the predicted healthy weight range and, as a result, obviously have higher BMI,” Irvine noted, with 67% girls and 62% boys found to have a higher BMI than normal for their age.
After 16 weeks of treatment with dupilumab, there was a much greater gain in height than that seen among those on placebo, with the most pronounced effect seen in children who had the lowest height at baseline. Indeed, among children in the lowest 25% height percentile at baseline, 30.6% on dupilumab vs 11.9% on placebo experienced an increase in height of 5 centiles or more(P < .05).
“This reflects what we see in clinical practice,” Irvine said. “Children often grow dramatically on treatment for atopic dermatitis.”
Among patients with a baseline height below the 30th percentile, 31.9% treated with dupilumab vs 11.1% treated with placebo gained at least 5 centiles in height. The figures for children below the 40th height percentile at baseline were 31.3% vs 15.5% (P < .05 for both).
Although there remained a marked difference in the proportion of children below the 50th height percentile at baseline gaining 5 centiles or more in height, at 29.0% with dupilumab versus 15.7% with placebo, it was no longer significant.
“So the effect of catch-up growth, or growth through the centiles, is most marked in those who are in the 40th centile or below,” Irvine said, indicating that the “more growth restricted kids have much more potential to catch up.”
‘Convincing’ Data
Overall, Kirkorian said in the interview, the data are “convincing” and support her view that severe atopic dermatitis is a “terrible chronic disease that we really underappreciate.” Atopic dermatitis, she added, “should get the respect that any severe chronic illness would have, whether that be arthritis, diabetes, or cardiac disease, because it is a systemic disorder that … profoundly affects quality of life, every minute of every day.”
However, “we don’t get all the referrals we should, until the child has suffered for years and years, and the family has suffered,” as there is a bias that it can be outgrown — although not everybody does — and it “doesn’t look as conspicuous as other chronic skin disorders,” such as psoriasis.
“Now with this study,” Kirkorian said, “it gives us a really compelling point to make to parents, to the community, and to insurers that not only are we affecting the quality of life from the itch standpoint [with dupilumab] but we may have long profound effects on growth and bone health.”
The research was sponsored by Sanofi and Regeneron Pharmaceuticals. Irvine declared relationships with AbbVie, Arena Pharmaceuticals, BenevolentAI, Chugai Pharmaceutical, Dermavant, Eli Lily, Genentech, LEO Pharma, Menlo Therapeutics, Novartis, Pfizer, Regeneron, Sanofi, UCB, DS Biopharma, and Inflazome. Kirkorian declared relationships with Dermavant, Verrica Pharmaceuticals, Pfizer, and Incyte.
A version of this article first appeared on Medscape.com.
AMSTERDAM — , revealed a post hoc trial analysis.
The research was presented at the European Academy of Dermatology and Venereology (EADV) 2024 Congress.
The trial included a “rigorously selected … well-characterized, well-studied” population of children aged 6-11 years, said presenter Alan D. Irvine, MD, DSc, professor of dermatology, Trinity College Dublin, Ireland.
It showed that “severe atopic dermatitis does cause restriction of growth, as well as a higher weight, and therefore obviously a higher BMI [body mass index].”
He continued, however, that children at the lower percentiles of height receiving prompt treatment with dupilumab (Dupixent) “were able to rapidly move through the centiles over the 16 weeks of the study, and that may be the window for catch-up growth … when children are growing rapidly.”
Anna Yasmine Kirkorian, MD, chief of dermatology, Children’s National Hospital, Washington, DC, who was not involved in the study, said that she was “surprised” at the degree of growth achieved over the study period, as height is not something that jumps up “overnight.”
“On the other hand, it fits with my experience with children who’ve had the brakes on all of their life due to inflammation, whether it be height, going to school, sleeping — everything is sort of put on pause by this terrible inflammatory process,” she said.
“When you take the brakes off, they get to be who they are going to be,” Kirkorian added. “So I was surprised by the speed of it, but not by the fact that height was acquired.”
Her belief is that in the pre-dupilumab era, severe atopic dermatitis was often insufficiently controlled, so children were “smaller than you would predict from parental height,” and the treatment is “allowing them to reach their genetic potential.”
Post Hoc Analysis
In his presentation, Irvine emphasized that it has been clearly demonstrated that adolescents with moderate and severe atopic dermatitis have a significantly higher likelihood of being below the 25th percentile of height on growth reference charts.
Such children are also at a higher risk of having low bone mineral density and low serum alkaline phosphatase (ALP) levels . While data presented at the EADV 2023 Congress showed that dupilumab significantly increased serum levels of bone ALP compared with placebo, the underlying mechanism remains unclear.
For the current analysis, Irvine and colleagues determined that the proportion of children aged 6-11 years with severe atopic dermatitis and lower stature reach a ≥ 5 centile improvement in height following 16 weeks of dupilumab treatment.
They examined data from the LIBERTY AD PEDS trial, in which patients aged 6-11 years with severe atopic dermatitis were randomized to 300 mg dupilumab every 4 weeks or placebo along with a mild or moderately potent topical corticosteroid. The study found that, overall, dupilumab was associated with significant improvements in signs, symptoms, and quality of life compared with placebo.
Height measures at baseline revealed that “more boys and more girls were below the 50th centile than you would predict for a healthy, normal control population,” Irvine said. “If we look at weight, we see the opposite,” he continued, “with a disproportionate number of boys and girls who are above the 50th centile for weight at baseline.”
Consequently, “we’re seeing these children who are shorter and heavier than the predicted healthy weight range and, as a result, obviously have higher BMI,” Irvine noted, with 67% girls and 62% boys found to have a higher BMI than normal for their age.
After 16 weeks of treatment with dupilumab, there was a much greater gain in height than that seen among those on placebo, with the most pronounced effect seen in children who had the lowest height at baseline. Indeed, among children in the lowest 25% height percentile at baseline, 30.6% on dupilumab vs 11.9% on placebo experienced an increase in height of 5 centiles or more(P < .05).
“This reflects what we see in clinical practice,” Irvine said. “Children often grow dramatically on treatment for atopic dermatitis.”
Among patients with a baseline height below the 30th percentile, 31.9% treated with dupilumab vs 11.1% treated with placebo gained at least 5 centiles in height. The figures for children below the 40th height percentile at baseline were 31.3% vs 15.5% (P < .05 for both).
Although there remained a marked difference in the proportion of children below the 50th height percentile at baseline gaining 5 centiles or more in height, at 29.0% with dupilumab versus 15.7% with placebo, it was no longer significant.
“So the effect of catch-up growth, or growth through the centiles, is most marked in those who are in the 40th centile or below,” Irvine said, indicating that the “more growth restricted kids have much more potential to catch up.”
‘Convincing’ Data
Overall, Kirkorian said in the interview, the data are “convincing” and support her view that severe atopic dermatitis is a “terrible chronic disease that we really underappreciate.” Atopic dermatitis, she added, “should get the respect that any severe chronic illness would have, whether that be arthritis, diabetes, or cardiac disease, because it is a systemic disorder that … profoundly affects quality of life, every minute of every day.”
However, “we don’t get all the referrals we should, until the child has suffered for years and years, and the family has suffered,” as there is a bias that it can be outgrown — although not everybody does — and it “doesn’t look as conspicuous as other chronic skin disorders,” such as psoriasis.
“Now with this study,” Kirkorian said, “it gives us a really compelling point to make to parents, to the community, and to insurers that not only are we affecting the quality of life from the itch standpoint [with dupilumab] but we may have long profound effects on growth and bone health.”
The research was sponsored by Sanofi and Regeneron Pharmaceuticals. Irvine declared relationships with AbbVie, Arena Pharmaceuticals, BenevolentAI, Chugai Pharmaceutical, Dermavant, Eli Lily, Genentech, LEO Pharma, Menlo Therapeutics, Novartis, Pfizer, Regeneron, Sanofi, UCB, DS Biopharma, and Inflazome. Kirkorian declared relationships with Dermavant, Verrica Pharmaceuticals, Pfizer, and Incyte.
A version of this article first appeared on Medscape.com.
FROM EADV 2024
Nemolizumab Benefits for Atopic Dermatitis Maintained in Long-Term Follow-Up Study
(AD), revealed an interim analysis of the ARCADIA open-label extension study.
The research was presented at the European Academy of Dermatology and Venereology (EADV) 2024 Congress.
The results showed nemolizumab was associated with “ongoing clinically meaningful improvements in itch, skin lesions, and sleep disturbance,” said study presenter Diamant Thaçi, MD, PhD, of the Comprehensive Center for Inflammation Medicine, University of Lü̈beck in Germany.
Moreover, “patient-reported outcomes, including quality of life ... continued to improve over 56 weeks of treatment.” In addition, Thaçi added, the “safety data support the long-term use of nemolizumab for the treatment of adolescent and adult patients with moderate to severe atopic dermatitis.”
He explained that interleukin (IL) 31 is a key neuroimmune cytokine in AD, triggering itch, skin barrier disruption, and exacerbation of inflammation via its receptor. Nemolizumab inhibits IL-31 receptor binding and was shown in the ARCADIA 1 and ARCADIA 2 trials to provide, along with background topical corticosteroids, clinically meaningful improvements in itch, skin lesions, and sleep for up to weeks 48 of follow-up in adolescents and adults with moderate to severe AD.
The current open-label long-term extension study involved patients who were enrolled in both ARCADIA 1 and 2 trials, as well as those from four phase 2 and 2b studies, a phase 3b study, and adolescents who had not been included in a trial but who met the criteria for the extension study. All patients, whether they started on placebo plus background topical corticosteroids in a prior study, were treated with nemolizumab 30 mg subcutaneously every 4 weeks along with topical corticosteroids.
The interim analysis included all efficacy and safety data up to the cutoff of September 30, 2022, on 723 patients who had completed 56 weeks of treatment among the 1751 patients initially enrolled in the extension study.
The results showed that, regardless of whether patients were nemolizumab naive at enrollment or had previously taken the drug, there were increases in the proportion of patients with an Investigator Global Assessment (IGA) score of 0/1 and an Eczema Area and Severity Index (EASI) score of at least 75 (EASI-75) over the 56 weeks of the study.
In those naive to nemolizumab, the increase in the proportion with an IGA score of 0/1 increased from 17.7% at baseline to 49.0% at 56 weeks, while the proportion with an EASI-75 increased from 24.0% to 78.7%.
The increase in the proportion of patients with an IGA score 0/1 among those who had previously received nemolizumab increased from 28.5% at baseline to 47.1% at 56 weeks. The proportion with an EASI-75 was 38.1% at baseline, rising to 73.0% at 56 weeks.
Increases in the proportion of patients with an EASI score of at least 50 and at least 90 were also seen with nemolizumab, as were increases in the proportion of patients with an improvement of at least four points on the SCORing Atopic Dermatitis Pruritus visual analogue scale and Sleep loss scores.
Similarly, the proportion of patients with a reduction in Dermatology Life Quality Index of at least four points increased over the study period.
Regarding safety, Thaçi said, there appeared to be fewer adverse events than had been previously reported with nemolizumab. “We don’t see any signs of conjunctivitis,” he continued, or significant risk of infection apart from for COVID-19, but he pointed out that the study was conducted during the pandemic, which was “a very difficult time.”
The most common treatment-related adverse events were, aside from COVID-19, nasopharyngitis in about 10%-11% of patients, upper respiratory tract infection in about 6% to almost 7%, and headache in about 5%.
Among the adverse events of special interest, newly diagnosed asthma or worsening of asthma occurred in 4.7%-4.8% of patients, while peripheral edema was seen in 0.8%-1.7%.
“Besides this, the study results are really looking very good,” he said, adding: “It means, in a long-term study, we can say today that nemolizumab has revealed the [same] safety profile that was shown in the ARCADIA 1 and 2 trials.”
Alan D. Irvine, MD, DSc, professor of dermatology, Trinity College Dublin in Ireland, who was not involved in the study, underlined that the current interim assessment does not represent the complete dataset and is based on observed cases rather than a more rigorous methodology, such as net reclassification improvement analysis.
“So it makes it a little harder to interpret when you don’t know how many people are dropping out and why they’re dropping out,” he told this news organization. “That said, those who remain on drug out to 56 weeks do experience ongoing improvement in disease control.”
Consequently, “the most reliable message you can take from this interim analysis of long-term data is that there were no new safety signals,” and nemolizumab looks “safe and well-tolerated.”
Where nemolizumab would fit into the treatment pathway for moderate to severe AD remains an open question, Irvine said, although he believes that IL-13 pathway inhibitors such as dupilumab, tralokinumab, and lebrikizumab “will remain the treatment of choice for the immediate future due to prescriber familiarity and good efficacy data.”
However, for patients who are unsuitable for IL-13 inhibitors and/or Janus kinase inhibitors such as abrocitinib and upadacitinib, nemolizumab “could be an interesting alternative.”
“That’s probably where it is going to start,” Irvine said, “and then obviously that will change over time and as the data mature and prescribers become more familiar with the drug in the real world.”
Nemolizumab (Nemluvio) is approved for treating prurigo nodularis (PN) in the United States and in Japan and is under Food and Drug Administration review for treating AD. It is also under review for PN and AD in Europe, Canada, the United Kingdom, and several other countries, according to Galderma. It is also approved for treating pruritus associated with AD in pediatric, adolescent, and adult patients in Japan.
The study was funded by Galderma. Thaçi declared relationships with AbbVie, Almirall, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Celltrion, Galderma, Janssen-Cilag, Kyowa Kirin, LEO Pharma, L’Oréal, Eli Lilly, Novartis, Pfizer, Regeneron, Sanofi, Target RWE, and UCB. Irvine declared relationships with AbbVie, Arena Pharmaceuticals, BenevolentAl, Chugai Pharmaceutical, Dermavant, Eli Lily, Genentech, LEO Pharma, Menlo Therapeutics, Novartis, Pfizer, Regeneron, Sanofi, UCB, DS Biopharma, and Inflazome.
A version of this article first appeared on Medscape.com.
(AD), revealed an interim analysis of the ARCADIA open-label extension study.
The research was presented at the European Academy of Dermatology and Venereology (EADV) 2024 Congress.
The results showed nemolizumab was associated with “ongoing clinically meaningful improvements in itch, skin lesions, and sleep disturbance,” said study presenter Diamant Thaçi, MD, PhD, of the Comprehensive Center for Inflammation Medicine, University of Lü̈beck in Germany.
Moreover, “patient-reported outcomes, including quality of life ... continued to improve over 56 weeks of treatment.” In addition, Thaçi added, the “safety data support the long-term use of nemolizumab for the treatment of adolescent and adult patients with moderate to severe atopic dermatitis.”
He explained that interleukin (IL) 31 is a key neuroimmune cytokine in AD, triggering itch, skin barrier disruption, and exacerbation of inflammation via its receptor. Nemolizumab inhibits IL-31 receptor binding and was shown in the ARCADIA 1 and ARCADIA 2 trials to provide, along with background topical corticosteroids, clinically meaningful improvements in itch, skin lesions, and sleep for up to weeks 48 of follow-up in adolescents and adults with moderate to severe AD.
The current open-label long-term extension study involved patients who were enrolled in both ARCADIA 1 and 2 trials, as well as those from four phase 2 and 2b studies, a phase 3b study, and adolescents who had not been included in a trial but who met the criteria for the extension study. All patients, whether they started on placebo plus background topical corticosteroids in a prior study, were treated with nemolizumab 30 mg subcutaneously every 4 weeks along with topical corticosteroids.
The interim analysis included all efficacy and safety data up to the cutoff of September 30, 2022, on 723 patients who had completed 56 weeks of treatment among the 1751 patients initially enrolled in the extension study.
The results showed that, regardless of whether patients were nemolizumab naive at enrollment or had previously taken the drug, there were increases in the proportion of patients with an Investigator Global Assessment (IGA) score of 0/1 and an Eczema Area and Severity Index (EASI) score of at least 75 (EASI-75) over the 56 weeks of the study.
In those naive to nemolizumab, the increase in the proportion with an IGA score of 0/1 increased from 17.7% at baseline to 49.0% at 56 weeks, while the proportion with an EASI-75 increased from 24.0% to 78.7%.
The increase in the proportion of patients with an IGA score 0/1 among those who had previously received nemolizumab increased from 28.5% at baseline to 47.1% at 56 weeks. The proportion with an EASI-75 was 38.1% at baseline, rising to 73.0% at 56 weeks.
Increases in the proportion of patients with an EASI score of at least 50 and at least 90 were also seen with nemolizumab, as were increases in the proportion of patients with an improvement of at least four points on the SCORing Atopic Dermatitis Pruritus visual analogue scale and Sleep loss scores.
Similarly, the proportion of patients with a reduction in Dermatology Life Quality Index of at least four points increased over the study period.
Regarding safety, Thaçi said, there appeared to be fewer adverse events than had been previously reported with nemolizumab. “We don’t see any signs of conjunctivitis,” he continued, or significant risk of infection apart from for COVID-19, but he pointed out that the study was conducted during the pandemic, which was “a very difficult time.”
The most common treatment-related adverse events were, aside from COVID-19, nasopharyngitis in about 10%-11% of patients, upper respiratory tract infection in about 6% to almost 7%, and headache in about 5%.
Among the adverse events of special interest, newly diagnosed asthma or worsening of asthma occurred in 4.7%-4.8% of patients, while peripheral edema was seen in 0.8%-1.7%.
“Besides this, the study results are really looking very good,” he said, adding: “It means, in a long-term study, we can say today that nemolizumab has revealed the [same] safety profile that was shown in the ARCADIA 1 and 2 trials.”
Alan D. Irvine, MD, DSc, professor of dermatology, Trinity College Dublin in Ireland, who was not involved in the study, underlined that the current interim assessment does not represent the complete dataset and is based on observed cases rather than a more rigorous methodology, such as net reclassification improvement analysis.
“So it makes it a little harder to interpret when you don’t know how many people are dropping out and why they’re dropping out,” he told this news organization. “That said, those who remain on drug out to 56 weeks do experience ongoing improvement in disease control.”
Consequently, “the most reliable message you can take from this interim analysis of long-term data is that there were no new safety signals,” and nemolizumab looks “safe and well-tolerated.”
Where nemolizumab would fit into the treatment pathway for moderate to severe AD remains an open question, Irvine said, although he believes that IL-13 pathway inhibitors such as dupilumab, tralokinumab, and lebrikizumab “will remain the treatment of choice for the immediate future due to prescriber familiarity and good efficacy data.”
However, for patients who are unsuitable for IL-13 inhibitors and/or Janus kinase inhibitors such as abrocitinib and upadacitinib, nemolizumab “could be an interesting alternative.”
“That’s probably where it is going to start,” Irvine said, “and then obviously that will change over time and as the data mature and prescribers become more familiar with the drug in the real world.”
Nemolizumab (Nemluvio) is approved for treating prurigo nodularis (PN) in the United States and in Japan and is under Food and Drug Administration review for treating AD. It is also under review for PN and AD in Europe, Canada, the United Kingdom, and several other countries, according to Galderma. It is also approved for treating pruritus associated with AD in pediatric, adolescent, and adult patients in Japan.
The study was funded by Galderma. Thaçi declared relationships with AbbVie, Almirall, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Celltrion, Galderma, Janssen-Cilag, Kyowa Kirin, LEO Pharma, L’Oréal, Eli Lilly, Novartis, Pfizer, Regeneron, Sanofi, Target RWE, and UCB. Irvine declared relationships with AbbVie, Arena Pharmaceuticals, BenevolentAl, Chugai Pharmaceutical, Dermavant, Eli Lily, Genentech, LEO Pharma, Menlo Therapeutics, Novartis, Pfizer, Regeneron, Sanofi, UCB, DS Biopharma, and Inflazome.
A version of this article first appeared on Medscape.com.
(AD), revealed an interim analysis of the ARCADIA open-label extension study.
The research was presented at the European Academy of Dermatology and Venereology (EADV) 2024 Congress.
The results showed nemolizumab was associated with “ongoing clinically meaningful improvements in itch, skin lesions, and sleep disturbance,” said study presenter Diamant Thaçi, MD, PhD, of the Comprehensive Center for Inflammation Medicine, University of Lü̈beck in Germany.
Moreover, “patient-reported outcomes, including quality of life ... continued to improve over 56 weeks of treatment.” In addition, Thaçi added, the “safety data support the long-term use of nemolizumab for the treatment of adolescent and adult patients with moderate to severe atopic dermatitis.”
He explained that interleukin (IL) 31 is a key neuroimmune cytokine in AD, triggering itch, skin barrier disruption, and exacerbation of inflammation via its receptor. Nemolizumab inhibits IL-31 receptor binding and was shown in the ARCADIA 1 and ARCADIA 2 trials to provide, along with background topical corticosteroids, clinically meaningful improvements in itch, skin lesions, and sleep for up to weeks 48 of follow-up in adolescents and adults with moderate to severe AD.
The current open-label long-term extension study involved patients who were enrolled in both ARCADIA 1 and 2 trials, as well as those from four phase 2 and 2b studies, a phase 3b study, and adolescents who had not been included in a trial but who met the criteria for the extension study. All patients, whether they started on placebo plus background topical corticosteroids in a prior study, were treated with nemolizumab 30 mg subcutaneously every 4 weeks along with topical corticosteroids.
The interim analysis included all efficacy and safety data up to the cutoff of September 30, 2022, on 723 patients who had completed 56 weeks of treatment among the 1751 patients initially enrolled in the extension study.
The results showed that, regardless of whether patients were nemolizumab naive at enrollment or had previously taken the drug, there were increases in the proportion of patients with an Investigator Global Assessment (IGA) score of 0/1 and an Eczema Area and Severity Index (EASI) score of at least 75 (EASI-75) over the 56 weeks of the study.
In those naive to nemolizumab, the increase in the proportion with an IGA score of 0/1 increased from 17.7% at baseline to 49.0% at 56 weeks, while the proportion with an EASI-75 increased from 24.0% to 78.7%.
The increase in the proportion of patients with an IGA score 0/1 among those who had previously received nemolizumab increased from 28.5% at baseline to 47.1% at 56 weeks. The proportion with an EASI-75 was 38.1% at baseline, rising to 73.0% at 56 weeks.
Increases in the proportion of patients with an EASI score of at least 50 and at least 90 were also seen with nemolizumab, as were increases in the proportion of patients with an improvement of at least four points on the SCORing Atopic Dermatitis Pruritus visual analogue scale and Sleep loss scores.
Similarly, the proportion of patients with a reduction in Dermatology Life Quality Index of at least four points increased over the study period.
Regarding safety, Thaçi said, there appeared to be fewer adverse events than had been previously reported with nemolizumab. “We don’t see any signs of conjunctivitis,” he continued, or significant risk of infection apart from for COVID-19, but he pointed out that the study was conducted during the pandemic, which was “a very difficult time.”
The most common treatment-related adverse events were, aside from COVID-19, nasopharyngitis in about 10%-11% of patients, upper respiratory tract infection in about 6% to almost 7%, and headache in about 5%.
Among the adverse events of special interest, newly diagnosed asthma or worsening of asthma occurred in 4.7%-4.8% of patients, while peripheral edema was seen in 0.8%-1.7%.
“Besides this, the study results are really looking very good,” he said, adding: “It means, in a long-term study, we can say today that nemolizumab has revealed the [same] safety profile that was shown in the ARCADIA 1 and 2 trials.”
Alan D. Irvine, MD, DSc, professor of dermatology, Trinity College Dublin in Ireland, who was not involved in the study, underlined that the current interim assessment does not represent the complete dataset and is based on observed cases rather than a more rigorous methodology, such as net reclassification improvement analysis.
“So it makes it a little harder to interpret when you don’t know how many people are dropping out and why they’re dropping out,” he told this news organization. “That said, those who remain on drug out to 56 weeks do experience ongoing improvement in disease control.”
Consequently, “the most reliable message you can take from this interim analysis of long-term data is that there were no new safety signals,” and nemolizumab looks “safe and well-tolerated.”
Where nemolizumab would fit into the treatment pathway for moderate to severe AD remains an open question, Irvine said, although he believes that IL-13 pathway inhibitors such as dupilumab, tralokinumab, and lebrikizumab “will remain the treatment of choice for the immediate future due to prescriber familiarity and good efficacy data.”
However, for patients who are unsuitable for IL-13 inhibitors and/or Janus kinase inhibitors such as abrocitinib and upadacitinib, nemolizumab “could be an interesting alternative.”
“That’s probably where it is going to start,” Irvine said, “and then obviously that will change over time and as the data mature and prescribers become more familiar with the drug in the real world.”
Nemolizumab (Nemluvio) is approved for treating prurigo nodularis (PN) in the United States and in Japan and is under Food and Drug Administration review for treating AD. It is also under review for PN and AD in Europe, Canada, the United Kingdom, and several other countries, according to Galderma. It is also approved for treating pruritus associated with AD in pediatric, adolescent, and adult patients in Japan.
The study was funded by Galderma. Thaçi declared relationships with AbbVie, Almirall, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Celltrion, Galderma, Janssen-Cilag, Kyowa Kirin, LEO Pharma, L’Oréal, Eli Lilly, Novartis, Pfizer, Regeneron, Sanofi, Target RWE, and UCB. Irvine declared relationships with AbbVie, Arena Pharmaceuticals, BenevolentAl, Chugai Pharmaceutical, Dermavant, Eli Lily, Genentech, LEO Pharma, Menlo Therapeutics, Novartis, Pfizer, Regeneron, Sanofi, UCB, DS Biopharma, and Inflazome.
A version of this article first appeared on Medscape.com.
FROM EADV 2024
Study Finds No Increased MACE Risk for JAK Inhibitors in Patients With Atopic Dermatitis
, suggested the results of a large, US-based, retrospective cohort study.
This holds true even in individuals aged 50 years or older, whose age puts them at increased cardiovascular (CV) risk, said Amina El Ayadi, PhD, of the University of Texas Medical Branch at Galveston. He presented the findings at the recent European Academy of Dermatology and Venereology (EADV) 2024 Congress.
Specifically, the analysis looked at treatment with the oral JAK1 inhibitors upadacitinib (Rinvoq) and abrocitinib (Cibinqo), both approved for treating AD in the United States, and found that the relative risk for MACE, such as acute myocardial infarction, cardiac arrest, stroke, or acute deep vein thrombosis, was ≤ 1.0 compared with those not treated with a JAKi.
Similarly, the relative risk for other CV safety endpoints, such as having an abnormal ECG or pericardial effusion, was also around 1.0. There was a slight increase in the relative risk for arrhythmias, peripheral edema, angina pectoris, or heart failure, but no value went > 1.6 and CIs spanned 1.0, indicating the results lack statistical significance.
Reassurance for Dermatologists?
“This suggests that oral administration of these drugs to the patient with atopic dermatitis does not increase the risk of major adverse cardiac events, and dermatologists, based on our data, can safely consider JAK inhibitors for treating moderate to severe dermatitis, even in patients with high risk for these diseases,” El Ayadi said during a late-breaking news session at the meeting.
Yolanda Gilaberte Calzada, MD, PhD, head of the Dermatology Department at Miguel Servet University Hospital in Zaragoza, Spain, who was one of the chairs for the session, said that this was “very good news for us.”
Gilaberte Calzada, president of the Spanish Academy of Dermatology and Venereology, asked if there were any data on the duration of treatment with the two JAKis included in the analysis. El Ayadi said that this was something that would be looked at in future data analyses.
Gilaberte Calzada also observed that because the CIs were wide, with more time, “we will have more defined data.”
Analyses Overview
For the two analyses — one in the overall population of patients with AD and the other in those aged 50 years or older — electronic medical record (EMR) data from the TriNetX Research Network were used. This is a global, federated health research network that contains EMRs for more than 275 million patients from over 120 healthcare organizations, El Ayadi explained.
To perform the analyses, the research team queried the TriNetX database to find all patients diagnosed with AD via the International Classification of Diseases, Tenth Revision code L20. They then determined if patients had been treated with JAKi or not, and specifically, with upadacitinib or abrocitinib. Those who had not received any JAKi treatment were the control population.
For the first analysis, no age-specific filter was applied. The investigators identified 1674 people with AD who had been treated with the JAKis and around 1.2 million who had not. Propensity score matching, based on age at diagnosis, biologic sex, and CV comorbidities, was performed to give a total of 1674 patients who had and 1674 who had not been treated with these medications.
In the second analysis, only those aged 50 years or older were considered; 875 patients who had received JAKi treatment were identified and around 250,000 who had not. Propensity score matching based on the same variables gave two groups of 875 people who had or had not taken a JAKi.
Queried over the age cutoff used, El Ayadi noted, “We did an analysis looking at patients 65 and older. However, we came up with lower patient numbers. … We do have this data, and we did not see any significant risk.”
The study was independently supported. El Ayadi and Gilaberte Calzada reported no conflicts of interest in relation to the presented findings.
A version of this article first appeared on Medscape.com.
, suggested the results of a large, US-based, retrospective cohort study.
This holds true even in individuals aged 50 years or older, whose age puts them at increased cardiovascular (CV) risk, said Amina El Ayadi, PhD, of the University of Texas Medical Branch at Galveston. He presented the findings at the recent European Academy of Dermatology and Venereology (EADV) 2024 Congress.
Specifically, the analysis looked at treatment with the oral JAK1 inhibitors upadacitinib (Rinvoq) and abrocitinib (Cibinqo), both approved for treating AD in the United States, and found that the relative risk for MACE, such as acute myocardial infarction, cardiac arrest, stroke, or acute deep vein thrombosis, was ≤ 1.0 compared with those not treated with a JAKi.
Similarly, the relative risk for other CV safety endpoints, such as having an abnormal ECG or pericardial effusion, was also around 1.0. There was a slight increase in the relative risk for arrhythmias, peripheral edema, angina pectoris, or heart failure, but no value went > 1.6 and CIs spanned 1.0, indicating the results lack statistical significance.
Reassurance for Dermatologists?
“This suggests that oral administration of these drugs to the patient with atopic dermatitis does not increase the risk of major adverse cardiac events, and dermatologists, based on our data, can safely consider JAK inhibitors for treating moderate to severe dermatitis, even in patients with high risk for these diseases,” El Ayadi said during a late-breaking news session at the meeting.
Yolanda Gilaberte Calzada, MD, PhD, head of the Dermatology Department at Miguel Servet University Hospital in Zaragoza, Spain, who was one of the chairs for the session, said that this was “very good news for us.”
Gilaberte Calzada, president of the Spanish Academy of Dermatology and Venereology, asked if there were any data on the duration of treatment with the two JAKis included in the analysis. El Ayadi said that this was something that would be looked at in future data analyses.
Gilaberte Calzada also observed that because the CIs were wide, with more time, “we will have more defined data.”
Analyses Overview
For the two analyses — one in the overall population of patients with AD and the other in those aged 50 years or older — electronic medical record (EMR) data from the TriNetX Research Network were used. This is a global, federated health research network that contains EMRs for more than 275 million patients from over 120 healthcare organizations, El Ayadi explained.
To perform the analyses, the research team queried the TriNetX database to find all patients diagnosed with AD via the International Classification of Diseases, Tenth Revision code L20. They then determined if patients had been treated with JAKi or not, and specifically, with upadacitinib or abrocitinib. Those who had not received any JAKi treatment were the control population.
For the first analysis, no age-specific filter was applied. The investigators identified 1674 people with AD who had been treated with the JAKis and around 1.2 million who had not. Propensity score matching, based on age at diagnosis, biologic sex, and CV comorbidities, was performed to give a total of 1674 patients who had and 1674 who had not been treated with these medications.
In the second analysis, only those aged 50 years or older were considered; 875 patients who had received JAKi treatment were identified and around 250,000 who had not. Propensity score matching based on the same variables gave two groups of 875 people who had or had not taken a JAKi.
Queried over the age cutoff used, El Ayadi noted, “We did an analysis looking at patients 65 and older. However, we came up with lower patient numbers. … We do have this data, and we did not see any significant risk.”
The study was independently supported. El Ayadi and Gilaberte Calzada reported no conflicts of interest in relation to the presented findings.
A version of this article first appeared on Medscape.com.
, suggested the results of a large, US-based, retrospective cohort study.
This holds true even in individuals aged 50 years or older, whose age puts them at increased cardiovascular (CV) risk, said Amina El Ayadi, PhD, of the University of Texas Medical Branch at Galveston. He presented the findings at the recent European Academy of Dermatology and Venereology (EADV) 2024 Congress.
Specifically, the analysis looked at treatment with the oral JAK1 inhibitors upadacitinib (Rinvoq) and abrocitinib (Cibinqo), both approved for treating AD in the United States, and found that the relative risk for MACE, such as acute myocardial infarction, cardiac arrest, stroke, or acute deep vein thrombosis, was ≤ 1.0 compared with those not treated with a JAKi.
Similarly, the relative risk for other CV safety endpoints, such as having an abnormal ECG or pericardial effusion, was also around 1.0. There was a slight increase in the relative risk for arrhythmias, peripheral edema, angina pectoris, or heart failure, but no value went > 1.6 and CIs spanned 1.0, indicating the results lack statistical significance.
Reassurance for Dermatologists?
“This suggests that oral administration of these drugs to the patient with atopic dermatitis does not increase the risk of major adverse cardiac events, and dermatologists, based on our data, can safely consider JAK inhibitors for treating moderate to severe dermatitis, even in patients with high risk for these diseases,” El Ayadi said during a late-breaking news session at the meeting.
Yolanda Gilaberte Calzada, MD, PhD, head of the Dermatology Department at Miguel Servet University Hospital in Zaragoza, Spain, who was one of the chairs for the session, said that this was “very good news for us.”
Gilaberte Calzada, president of the Spanish Academy of Dermatology and Venereology, asked if there were any data on the duration of treatment with the two JAKis included in the analysis. El Ayadi said that this was something that would be looked at in future data analyses.
Gilaberte Calzada also observed that because the CIs were wide, with more time, “we will have more defined data.”
Analyses Overview
For the two analyses — one in the overall population of patients with AD and the other in those aged 50 years or older — electronic medical record (EMR) data from the TriNetX Research Network were used. This is a global, federated health research network that contains EMRs for more than 275 million patients from over 120 healthcare organizations, El Ayadi explained.
To perform the analyses, the research team queried the TriNetX database to find all patients diagnosed with AD via the International Classification of Diseases, Tenth Revision code L20. They then determined if patients had been treated with JAKi or not, and specifically, with upadacitinib or abrocitinib. Those who had not received any JAKi treatment were the control population.
For the first analysis, no age-specific filter was applied. The investigators identified 1674 people with AD who had been treated with the JAKis and around 1.2 million who had not. Propensity score matching, based on age at diagnosis, biologic sex, and CV comorbidities, was performed to give a total of 1674 patients who had and 1674 who had not been treated with these medications.
In the second analysis, only those aged 50 years or older were considered; 875 patients who had received JAKi treatment were identified and around 250,000 who had not. Propensity score matching based on the same variables gave two groups of 875 people who had or had not taken a JAKi.
Queried over the age cutoff used, El Ayadi noted, “We did an analysis looking at patients 65 and older. However, we came up with lower patient numbers. … We do have this data, and we did not see any significant risk.”
The study was independently supported. El Ayadi and Gilaberte Calzada reported no conflicts of interest in relation to the presented findings.
A version of this article first appeared on Medscape.com.
FROM EADV 2024
Topical JAK Inhibitor Effective for Hand Eczema, Two Studies Suggest
suggested the results of two separate studies presented during the late-breaking sessions at the European Academy of Dermatology and Venereology (EADV) 2024 Congress.
In the 24-week, phase 3 DELTA FORCE trial, topical delgocitinib was compared head to head with oral alitretinoin for managing chronic hand eczema (CHE). Results showed that greater improvements from baseline to week 12 in Hand Eczema Severity Index (HECSI) scores could be achieved with delgocitinib cream than with alitretinoin capsules.
And in another analysis, which involved patients with the atopic subtype of CHE only, the application of topical delgocitinib was found to be as good as treatment with subcutaneous dupilumab (Dupixent) at improving both HECSI scores and the Investigator Global Assessment for CHE response (IGA-CHE).
Potentially a ‘Highly Impactful’ Therapy
“Chronic hand eczema is a common yet burdensome skin condition that poses a considerable challenge for dermatologists. Diversity in morphologic presentation and clinical etiology has been a key limitation for the development of a safe, targeted, one-size-fits-all therapeutic approach,” Raj Chovatiya, MD, PhD, clinical associate professor at Chicago Medical School, Rosalind Franklin University of Medicine and Science, and the founder and director of the Center for Medical Dermatology and Immunology Research in Chicago, Illinois, said in an interview.
“These data show that delgocitinib cream is poised to be a novel and highly impactful topical therapy for the treatment of CHE,” said Chovatiya.
DELTA FORCE showed that the efficacy and safety of delgocitinib cream was “superior to alitretinoin, the only approved oral option for CHE,” he said. And the other study, a comparative analysis, showed that delgocitinib’s efficacy was “comparable to the biologic dupilumab specifically for the treatment of atopic hand eczema,” said Chovatiya, one of the authors of that study. He was not an author of the DELTA FORCE study.
DELTA FORCE
While it remains an investigational drug in the United States, where it is under Food and Drug Administration review for CHE, delgocitinib cream (Anzupgo) was recently approved by the European Commission for use in adults with moderate to severe hand eczema who do not respond to or who are unable to use topical corticosteroids. Approval was based on data from two phase 3 studies , DELTA 1 and DELTA 2, which compared delgocitinib cream against a cream vehicle, as well as an open-label, long-term extension study, DELTA 3.
In the DELTA FORCE study, 513 adults with severe CHE (IGA-CHE score of 4) were recruited at 102 clinical centers in Europe and North America and randomly allocated to topical treatment with delgocitinib cream, 20 mg/g applied twice daily, or alitretinoin capsules, 30 mg once daily. Treatment with delgocitinib was for 16 weeks, and treatment with alitretinoin was for 12 weeks. The latter’s dose could be reduced to 10 mg in the event of intolerability, and both treatments could be reintroduced if necessary, with a final follow-up at 24 weeks.
Study investigator Ana Maria Giménez-Arnau, MD, PhD, of the Hospital del Mar Research Institute, Pompeu Fabra University, Barcelona, Spain, who presented the findings, noted that alitretinoin (Toctino) is an oral systemic retinoid approved in a few European countries, Canada, Israel, and South Korea for the treatment of severe CHE.
The mean age of the participants was 45 years, almost two thirds were women, and the majority (93%) were White; 90% of patients had been recruited in Europe. The median duration of CHE was 4 years.
At baseline, the median HECSI score was recorded as 79.5 in the delgocitinib arm and 80.0 in the alitretinoin arm. At 12 weeks, the least squares mean change in HECSI score from baseline was –67.6 in the delgocitinib arm and –51.5 in the alitretinoin arm, giving a significant difference of –16.1 between the two groups (P < .001).
Giménez-Arnau reported that delgocitinib also outperformed alitretinoin for all other endpoints assessed, including the following: ≥ 90% improvement in HECSI (HECSI-90), IGA-CHE treatment success (defined as a score of 0/1 indicating clear/almost clear skin), changes in Hand Eczema Symptom Diary (HESD) itch and HESD pain scores, area under the curve for HECSI-90, change in Dermatology Life Quality Index score — which were all assessed at 12 weeks — and change in HECSI from baseline to week 24.
There was “significant improvement in the reduction of the HECSI from the first week” of treatment, Giménez-Arnau said at the meeting. Notably, that the effect increased to 12 weeks and then was sustained. A similar pattern was seen for IGA-CHE treatment success and for HESD pain. This is important as “chronic hand eczema is really painful,” she said.
As for safety, 49.4% of patients in the delgocitinib arm vs 76.1% of patients in the alitretinoin arm experienced any type of adverse event (AE). Serious AEs occurred in 2% and 4.9% of patients in each group, respectively, with fewer AEs leading to trial drug discontinuation observed in the delgocitinib arm (1.2% vs 10.1%). The proportion of AEs “probably or possibly” related to the trial drug was 9.5% in the delgocitinib group vs 54.3% in the alitretinoin group.
Comparison With Dupilumab in Another Trial
Delgocitinib is no longer just an investigational medication, April W. Armstrong, MD, MPH, professor and chief of dermatology, University of California, Los Angeles, said during a separate late-breaking presentation at the EADV 2024 meeting. “I think it’s big news because now we have an important topical option for our patients with chronic hand eczema.”
Armstrong presented a matched-adjusted indirect comparison (MAIC) of delgocitinib vs dupilumab for the treatment of moderate to severe atopic hand eczema, which she described as “the next best thing” to a head-to-head trial.
MAICs are where patient level data from one or more clinical trials evaluating drug “A” are compared with aggregate data from one or more clinical trials evaluating drug “B.” In this case, individual patient data from the DELTA 1 and DELTA 2 trials of delgocitinib were compared with published aggregate data from the phase 3 LIBERTY-AD-HAFT trial of dupilumab.
A total of 201 patients with atopic hand eczema in the DELTA 1 and DELTA 2 trials were matched to 133 patients in the LIBERTY-AD-HAFT trial. Of these, 128 had been treated with delgocitinib cream, 73 with a cream vehicle, 67 with subcutaneous dupilumab, and 66 with a subcutaneous placebo.
“We’re trying to compare as much as possible apples to apples here in terms of the etiology of hand eczema,” Armstrong said. She noted that after matching and weighting based on age, sex, race, and baseline HECSI score, baseline characteristics in the two groups of patients were similar. The mean age was about 35.8 years in the two active treatment arms and 33.4 years in the two placebo arms, and mean baseline HESCI scores were about 79-80.
The endpoints compared were ≥ 75% improvement in HECSI; HECSI-90, HECSI percentage improvement, and IGA-CHE in the DELTA 1 and DELTA 2 trials; or a Hand and Foot IGA score of 0/1.
“The key message to take away from this is that there were no statistically significant differences between topical delgocitinib twice daily vs subcutaneous injection of dupilumab by week 16 in the treatment of patients with atopic hand dermatitis,” Armstrong reported. Odds ratios varied between 1.1 and 1.3 for the various endpoints.
Menno de Rie, MD, PhD, professor of dermatology and immunology at Amsterdam University Medical Center in the Netherlands, who cochaired the session, said that “I appreciate very much that you took the effort to compare these totally different compounds and showed us the methodology that you did. It’s really very impressive.”
Topical, Systemic, or Both?
Armstrong was questioned on how to manage someone with atopic hand dermatitis who developed lesions elsewhere on the body.
“I would take a really individualized approach to this patient,” she responded. If the eczema has been limited to the hands and has been there for a while, then perhaps delgocitinib would be her choice, but if they developed lesions elsewhere on the body, then a systemic treatment such as dupilumab may be preferable.
“The good thing is that this study shows that you can offer the patient either of those options and really engage the patient in a shared decision-making process.”
And with regards to whether the two might possibly be used together, Armstrong acknowledged insurance coverage restrictions could be a limiting factor in the United States, but elsewhere — and from a scientific point of view — this could make sense.
“If we have a patient, for example, who has moderate to severe atopic dermatitis involving the body, but also very severe hand eczema as well, one may possibly consider a combination of a systemic medication that’s helpful for the extensive area of involvement on the body ... and now you have a topical therapy, delgocitinib, where you can use it locally, have very deep efficacy locally, to kind of help augment that disease phenotype in that patient population.”
The studies were funded by Leo Pharma. Chovatiya, Giménez-Arnau, and Armstrong acknowledged ties to LEO Pharma, among other pharmaceutical companies.
A version of this article first appeared on Medscape.com.
suggested the results of two separate studies presented during the late-breaking sessions at the European Academy of Dermatology and Venereology (EADV) 2024 Congress.
In the 24-week, phase 3 DELTA FORCE trial, topical delgocitinib was compared head to head with oral alitretinoin for managing chronic hand eczema (CHE). Results showed that greater improvements from baseline to week 12 in Hand Eczema Severity Index (HECSI) scores could be achieved with delgocitinib cream than with alitretinoin capsules.
And in another analysis, which involved patients with the atopic subtype of CHE only, the application of topical delgocitinib was found to be as good as treatment with subcutaneous dupilumab (Dupixent) at improving both HECSI scores and the Investigator Global Assessment for CHE response (IGA-CHE).
Potentially a ‘Highly Impactful’ Therapy
“Chronic hand eczema is a common yet burdensome skin condition that poses a considerable challenge for dermatologists. Diversity in morphologic presentation and clinical etiology has been a key limitation for the development of a safe, targeted, one-size-fits-all therapeutic approach,” Raj Chovatiya, MD, PhD, clinical associate professor at Chicago Medical School, Rosalind Franklin University of Medicine and Science, and the founder and director of the Center for Medical Dermatology and Immunology Research in Chicago, Illinois, said in an interview.
“These data show that delgocitinib cream is poised to be a novel and highly impactful topical therapy for the treatment of CHE,” said Chovatiya.
DELTA FORCE showed that the efficacy and safety of delgocitinib cream was “superior to alitretinoin, the only approved oral option for CHE,” he said. And the other study, a comparative analysis, showed that delgocitinib’s efficacy was “comparable to the biologic dupilumab specifically for the treatment of atopic hand eczema,” said Chovatiya, one of the authors of that study. He was not an author of the DELTA FORCE study.
DELTA FORCE
While it remains an investigational drug in the United States, where it is under Food and Drug Administration review for CHE, delgocitinib cream (Anzupgo) was recently approved by the European Commission for use in adults with moderate to severe hand eczema who do not respond to or who are unable to use topical corticosteroids. Approval was based on data from two phase 3 studies , DELTA 1 and DELTA 2, which compared delgocitinib cream against a cream vehicle, as well as an open-label, long-term extension study, DELTA 3.
In the DELTA FORCE study, 513 adults with severe CHE (IGA-CHE score of 4) were recruited at 102 clinical centers in Europe and North America and randomly allocated to topical treatment with delgocitinib cream, 20 mg/g applied twice daily, or alitretinoin capsules, 30 mg once daily. Treatment with delgocitinib was for 16 weeks, and treatment with alitretinoin was for 12 weeks. The latter’s dose could be reduced to 10 mg in the event of intolerability, and both treatments could be reintroduced if necessary, with a final follow-up at 24 weeks.
Study investigator Ana Maria Giménez-Arnau, MD, PhD, of the Hospital del Mar Research Institute, Pompeu Fabra University, Barcelona, Spain, who presented the findings, noted that alitretinoin (Toctino) is an oral systemic retinoid approved in a few European countries, Canada, Israel, and South Korea for the treatment of severe CHE.
The mean age of the participants was 45 years, almost two thirds were women, and the majority (93%) were White; 90% of patients had been recruited in Europe. The median duration of CHE was 4 years.
At baseline, the median HECSI score was recorded as 79.5 in the delgocitinib arm and 80.0 in the alitretinoin arm. At 12 weeks, the least squares mean change in HECSI score from baseline was –67.6 in the delgocitinib arm and –51.5 in the alitretinoin arm, giving a significant difference of –16.1 between the two groups (P < .001).
Giménez-Arnau reported that delgocitinib also outperformed alitretinoin for all other endpoints assessed, including the following: ≥ 90% improvement in HECSI (HECSI-90), IGA-CHE treatment success (defined as a score of 0/1 indicating clear/almost clear skin), changes in Hand Eczema Symptom Diary (HESD) itch and HESD pain scores, area under the curve for HECSI-90, change in Dermatology Life Quality Index score — which were all assessed at 12 weeks — and change in HECSI from baseline to week 24.
There was “significant improvement in the reduction of the HECSI from the first week” of treatment, Giménez-Arnau said at the meeting. Notably, that the effect increased to 12 weeks and then was sustained. A similar pattern was seen for IGA-CHE treatment success and for HESD pain. This is important as “chronic hand eczema is really painful,” she said.
As for safety, 49.4% of patients in the delgocitinib arm vs 76.1% of patients in the alitretinoin arm experienced any type of adverse event (AE). Serious AEs occurred in 2% and 4.9% of patients in each group, respectively, with fewer AEs leading to trial drug discontinuation observed in the delgocitinib arm (1.2% vs 10.1%). The proportion of AEs “probably or possibly” related to the trial drug was 9.5% in the delgocitinib group vs 54.3% in the alitretinoin group.
Comparison With Dupilumab in Another Trial
Delgocitinib is no longer just an investigational medication, April W. Armstrong, MD, MPH, professor and chief of dermatology, University of California, Los Angeles, said during a separate late-breaking presentation at the EADV 2024 meeting. “I think it’s big news because now we have an important topical option for our patients with chronic hand eczema.”
Armstrong presented a matched-adjusted indirect comparison (MAIC) of delgocitinib vs dupilumab for the treatment of moderate to severe atopic hand eczema, which she described as “the next best thing” to a head-to-head trial.
MAICs are where patient level data from one or more clinical trials evaluating drug “A” are compared with aggregate data from one or more clinical trials evaluating drug “B.” In this case, individual patient data from the DELTA 1 and DELTA 2 trials of delgocitinib were compared with published aggregate data from the phase 3 LIBERTY-AD-HAFT trial of dupilumab.
A total of 201 patients with atopic hand eczema in the DELTA 1 and DELTA 2 trials were matched to 133 patients in the LIBERTY-AD-HAFT trial. Of these, 128 had been treated with delgocitinib cream, 73 with a cream vehicle, 67 with subcutaneous dupilumab, and 66 with a subcutaneous placebo.
“We’re trying to compare as much as possible apples to apples here in terms of the etiology of hand eczema,” Armstrong said. She noted that after matching and weighting based on age, sex, race, and baseline HECSI score, baseline characteristics in the two groups of patients were similar. The mean age was about 35.8 years in the two active treatment arms and 33.4 years in the two placebo arms, and mean baseline HESCI scores were about 79-80.
The endpoints compared were ≥ 75% improvement in HECSI; HECSI-90, HECSI percentage improvement, and IGA-CHE in the DELTA 1 and DELTA 2 trials; or a Hand and Foot IGA score of 0/1.
“The key message to take away from this is that there were no statistically significant differences between topical delgocitinib twice daily vs subcutaneous injection of dupilumab by week 16 in the treatment of patients with atopic hand dermatitis,” Armstrong reported. Odds ratios varied between 1.1 and 1.3 for the various endpoints.
Menno de Rie, MD, PhD, professor of dermatology and immunology at Amsterdam University Medical Center in the Netherlands, who cochaired the session, said that “I appreciate very much that you took the effort to compare these totally different compounds and showed us the methodology that you did. It’s really very impressive.”
Topical, Systemic, or Both?
Armstrong was questioned on how to manage someone with atopic hand dermatitis who developed lesions elsewhere on the body.
“I would take a really individualized approach to this patient,” she responded. If the eczema has been limited to the hands and has been there for a while, then perhaps delgocitinib would be her choice, but if they developed lesions elsewhere on the body, then a systemic treatment such as dupilumab may be preferable.
“The good thing is that this study shows that you can offer the patient either of those options and really engage the patient in a shared decision-making process.”
And with regards to whether the two might possibly be used together, Armstrong acknowledged insurance coverage restrictions could be a limiting factor in the United States, but elsewhere — and from a scientific point of view — this could make sense.
“If we have a patient, for example, who has moderate to severe atopic dermatitis involving the body, but also very severe hand eczema as well, one may possibly consider a combination of a systemic medication that’s helpful for the extensive area of involvement on the body ... and now you have a topical therapy, delgocitinib, where you can use it locally, have very deep efficacy locally, to kind of help augment that disease phenotype in that patient population.”
The studies were funded by Leo Pharma. Chovatiya, Giménez-Arnau, and Armstrong acknowledged ties to LEO Pharma, among other pharmaceutical companies.
A version of this article first appeared on Medscape.com.
suggested the results of two separate studies presented during the late-breaking sessions at the European Academy of Dermatology and Venereology (EADV) 2024 Congress.
In the 24-week, phase 3 DELTA FORCE trial, topical delgocitinib was compared head to head with oral alitretinoin for managing chronic hand eczema (CHE). Results showed that greater improvements from baseline to week 12 in Hand Eczema Severity Index (HECSI) scores could be achieved with delgocitinib cream than with alitretinoin capsules.
And in another analysis, which involved patients with the atopic subtype of CHE only, the application of topical delgocitinib was found to be as good as treatment with subcutaneous dupilumab (Dupixent) at improving both HECSI scores and the Investigator Global Assessment for CHE response (IGA-CHE).
Potentially a ‘Highly Impactful’ Therapy
“Chronic hand eczema is a common yet burdensome skin condition that poses a considerable challenge for dermatologists. Diversity in morphologic presentation and clinical etiology has been a key limitation for the development of a safe, targeted, one-size-fits-all therapeutic approach,” Raj Chovatiya, MD, PhD, clinical associate professor at Chicago Medical School, Rosalind Franklin University of Medicine and Science, and the founder and director of the Center for Medical Dermatology and Immunology Research in Chicago, Illinois, said in an interview.
“These data show that delgocitinib cream is poised to be a novel and highly impactful topical therapy for the treatment of CHE,” said Chovatiya.
DELTA FORCE showed that the efficacy and safety of delgocitinib cream was “superior to alitretinoin, the only approved oral option for CHE,” he said. And the other study, a comparative analysis, showed that delgocitinib’s efficacy was “comparable to the biologic dupilumab specifically for the treatment of atopic hand eczema,” said Chovatiya, one of the authors of that study. He was not an author of the DELTA FORCE study.
DELTA FORCE
While it remains an investigational drug in the United States, where it is under Food and Drug Administration review for CHE, delgocitinib cream (Anzupgo) was recently approved by the European Commission for use in adults with moderate to severe hand eczema who do not respond to or who are unable to use topical corticosteroids. Approval was based on data from two phase 3 studies , DELTA 1 and DELTA 2, which compared delgocitinib cream against a cream vehicle, as well as an open-label, long-term extension study, DELTA 3.
In the DELTA FORCE study, 513 adults with severe CHE (IGA-CHE score of 4) were recruited at 102 clinical centers in Europe and North America and randomly allocated to topical treatment with delgocitinib cream, 20 mg/g applied twice daily, or alitretinoin capsules, 30 mg once daily. Treatment with delgocitinib was for 16 weeks, and treatment with alitretinoin was for 12 weeks. The latter’s dose could be reduced to 10 mg in the event of intolerability, and both treatments could be reintroduced if necessary, with a final follow-up at 24 weeks.
Study investigator Ana Maria Giménez-Arnau, MD, PhD, of the Hospital del Mar Research Institute, Pompeu Fabra University, Barcelona, Spain, who presented the findings, noted that alitretinoin (Toctino) is an oral systemic retinoid approved in a few European countries, Canada, Israel, and South Korea for the treatment of severe CHE.
The mean age of the participants was 45 years, almost two thirds were women, and the majority (93%) were White; 90% of patients had been recruited in Europe. The median duration of CHE was 4 years.
At baseline, the median HECSI score was recorded as 79.5 in the delgocitinib arm and 80.0 in the alitretinoin arm. At 12 weeks, the least squares mean change in HECSI score from baseline was –67.6 in the delgocitinib arm and –51.5 in the alitretinoin arm, giving a significant difference of –16.1 between the two groups (P < .001).
Giménez-Arnau reported that delgocitinib also outperformed alitretinoin for all other endpoints assessed, including the following: ≥ 90% improvement in HECSI (HECSI-90), IGA-CHE treatment success (defined as a score of 0/1 indicating clear/almost clear skin), changes in Hand Eczema Symptom Diary (HESD) itch and HESD pain scores, area under the curve for HECSI-90, change in Dermatology Life Quality Index score — which were all assessed at 12 weeks — and change in HECSI from baseline to week 24.
There was “significant improvement in the reduction of the HECSI from the first week” of treatment, Giménez-Arnau said at the meeting. Notably, that the effect increased to 12 weeks and then was sustained. A similar pattern was seen for IGA-CHE treatment success and for HESD pain. This is important as “chronic hand eczema is really painful,” she said.
As for safety, 49.4% of patients in the delgocitinib arm vs 76.1% of patients in the alitretinoin arm experienced any type of adverse event (AE). Serious AEs occurred in 2% and 4.9% of patients in each group, respectively, with fewer AEs leading to trial drug discontinuation observed in the delgocitinib arm (1.2% vs 10.1%). The proportion of AEs “probably or possibly” related to the trial drug was 9.5% in the delgocitinib group vs 54.3% in the alitretinoin group.
Comparison With Dupilumab in Another Trial
Delgocitinib is no longer just an investigational medication, April W. Armstrong, MD, MPH, professor and chief of dermatology, University of California, Los Angeles, said during a separate late-breaking presentation at the EADV 2024 meeting. “I think it’s big news because now we have an important topical option for our patients with chronic hand eczema.”
Armstrong presented a matched-adjusted indirect comparison (MAIC) of delgocitinib vs dupilumab for the treatment of moderate to severe atopic hand eczema, which she described as “the next best thing” to a head-to-head trial.
MAICs are where patient level data from one or more clinical trials evaluating drug “A” are compared with aggregate data from one or more clinical trials evaluating drug “B.” In this case, individual patient data from the DELTA 1 and DELTA 2 trials of delgocitinib were compared with published aggregate data from the phase 3 LIBERTY-AD-HAFT trial of dupilumab.
A total of 201 patients with atopic hand eczema in the DELTA 1 and DELTA 2 trials were matched to 133 patients in the LIBERTY-AD-HAFT trial. Of these, 128 had been treated with delgocitinib cream, 73 with a cream vehicle, 67 with subcutaneous dupilumab, and 66 with a subcutaneous placebo.
“We’re trying to compare as much as possible apples to apples here in terms of the etiology of hand eczema,” Armstrong said. She noted that after matching and weighting based on age, sex, race, and baseline HECSI score, baseline characteristics in the two groups of patients were similar. The mean age was about 35.8 years in the two active treatment arms and 33.4 years in the two placebo arms, and mean baseline HESCI scores were about 79-80.
The endpoints compared were ≥ 75% improvement in HECSI; HECSI-90, HECSI percentage improvement, and IGA-CHE in the DELTA 1 and DELTA 2 trials; or a Hand and Foot IGA score of 0/1.
“The key message to take away from this is that there were no statistically significant differences between topical delgocitinib twice daily vs subcutaneous injection of dupilumab by week 16 in the treatment of patients with atopic hand dermatitis,” Armstrong reported. Odds ratios varied between 1.1 and 1.3 for the various endpoints.
Menno de Rie, MD, PhD, professor of dermatology and immunology at Amsterdam University Medical Center in the Netherlands, who cochaired the session, said that “I appreciate very much that you took the effort to compare these totally different compounds and showed us the methodology that you did. It’s really very impressive.”
Topical, Systemic, or Both?
Armstrong was questioned on how to manage someone with atopic hand dermatitis who developed lesions elsewhere on the body.
“I would take a really individualized approach to this patient,” she responded. If the eczema has been limited to the hands and has been there for a while, then perhaps delgocitinib would be her choice, but if they developed lesions elsewhere on the body, then a systemic treatment such as dupilumab may be preferable.
“The good thing is that this study shows that you can offer the patient either of those options and really engage the patient in a shared decision-making process.”
And with regards to whether the two might possibly be used together, Armstrong acknowledged insurance coverage restrictions could be a limiting factor in the United States, but elsewhere — and from a scientific point of view — this could make sense.
“If we have a patient, for example, who has moderate to severe atopic dermatitis involving the body, but also very severe hand eczema as well, one may possibly consider a combination of a systemic medication that’s helpful for the extensive area of involvement on the body ... and now you have a topical therapy, delgocitinib, where you can use it locally, have very deep efficacy locally, to kind of help augment that disease phenotype in that patient population.”
The studies were funded by Leo Pharma. Chovatiya, Giménez-Arnau, and Armstrong acknowledged ties to LEO Pharma, among other pharmaceutical companies.
A version of this article first appeared on Medscape.com.
FROM EADV 2024
Sea Buckthorn
A member of the Elaeagnaceae family, Hippophae rhamnoides, better known as sea buckthorn, is a high-altitude wild shrub endemic to Europe and Asia with edible fruits and a lengthy record of use in traditional Chinese medicine.1-6 Used as a health supplement and consumed in the diet throughout the world,5 sea buckthorn berries, seeds, and leaves have been used in traditional medicine to treat burns/injuries, edema, hypertension, inflammation, skin grafts, ulcers, and wounds.4,7
This hardy plant is associated with a wide range of biologic activities, including anti-atherogenic, anti-atopic dermatitis, antibacterial, anticancer, antifungal, anti-inflammatory, antimicrobial, antioxidant, anti-psoriasis, anti-sebum, anti-stress, anti-tumor, cytoprotective, hepatoprotective, immunomodulatory, neuroprotective, radioprotective, and tissue regenerative functions.4,5,8-11
Key Constituents
Functional constituents identified in sea buckthorn include alkaloids, carotenoids, flavonoids, lignans, organic acids, phenolic acids, proanthocyanidins, polyunsaturated acids (including omega-3, -6, -7, and -9), steroids, tannins, terpenoids, and volatile oils, as well as nutritional compounds such as minerals, proteins, and vitamins.4,5,11 Sea buckthorn pericarp oil contains copious amounts of saturated palmitic acid (29%-36%) and omega-7 unsaturated palmitoleic acid (36%-48%), which fosters cutaneous and mucosal epithelialization, as well as linoleic (10%-12%) and oleic (4%-6%) acids.12,6 Significant amounts of carotenoids as well as alpha‐linolenic fatty acid (38%), linoleic (36%), oleic (13%), and palmitic (7%) acids are present in sea buckthorn seed oil.6
Polysaccharides
In an expansive review on the pharmacological activities of sea buckthorn polysaccharides, Teng and colleagues reported in April 2024 that 20 diverse polysaccharides have been culled from sea buckthorn and exhibited various healthy activities, including antioxidant, anti-fatigue, anti-inflammatory, anti-obesity, anti-tumor, hepatoprotective, hypoglycemic, and immunoregulation, and regulation of intestinal flora activities.1
Proanthocyanidins and Anti-Aging
In 2023, Liu and colleagues investigated the anti–skin aging impact of sea buckthorn proanthocyanidins in D-galactose-induced aging in mice given the known free radical scavenging activity of these compounds. They found the proanthocyanidins mitigated D-galactose-induced aging and can augment the total antioxidant capacity of the body. Sea buckthorn proanthocyanidins can further attenuate the effects of skin aging by regulating the TGF-beta1/Smads pathway and MMPs/TIMP system, thus amplifying collagen I and tropoelastin content.13
A year earlier, many of the same investigators assessed the possible protective activity of sea buckthorn proanthocyanidins against cutaneous aging engendered by oxidative stress from hydrogen peroxide. The compounds amplified superoxide dismutase and glutathione antioxidant functions. The extracts also fostered collagen I production in aging human skin fibroblasts via the TGF-beta1/Smads pathway and hindered collagen I degradation by regulating the MMPs/TIMPs system, which maintained extracellular matrix integrity. Senescent cell migration was also promoted with 100 mcg/mL of sea buckthorn proanthocyanidins. The researchers concluded that this sets the stage for investigating how sea buckthorn proanthocyanidins can be incorporated in cosmetic formulations.14 In a separate study, Liu and colleagues demonstrated that sea buckthorn proanthocyanidins can attenuate oxidative damage and protect mitochondrial function.9
Acne and Barrier Functions
The extracts of H rhamnoides and Cassia fistula in a combined formulation were found to be effective in lowering skin sebum content in humans with grade I and grade II acne vulgaris in a 2014 single-blind, randomized, placebo-controlled, split-face study with two groups of 25 patients each (aged 18-37 years).15 Khan and colleagues have also reported that a sea buckthorn oil-in-water emulsion improved barrier function in human skin as tested by a tewameter and corneometer (noninvasive probes) in 13 healthy males with a mean age of 27 ± 4.8 years.16
Anti-Aging, Antioxidant, Antibacterial, Skin-Whitening Activity
Zaman and colleagues reported in 2011 that results from an in vivo study of the effects of a sea buckthorn fruit extract topical cream on stratum corneum water content and transepidermal water loss indicated that the formulation enhanced cell surface integrin expression thus facilitating collagen contraction.17
In 2012, Khan and colleagues reported amelioration in skin elasticity, thus achieving an anti-aging result, from the use of a water-in-oil–based hydroalcoholic cream loaded with fruit extract of H rhamnoides, as measured with a Cutometer.18 The previous year, some of the same researchers reported that the antioxidants and flavonoids found in a topical sea buckthorn formulation could decrease cutaneous melanin and erythema levels.
More recently, Gęgotek and colleagues found that sea buckthorn seed oil prevented redox balance and lipid metabolism disturbances in skin fibroblasts and keratinocytes caused by UVA or UVB. They suggested that such findings point to the potential of this natural agent to confer anti-inflammatory properties and photoprotection to the skin.19
In 2020, Ivanišová and colleagues investigated the antioxidant and antimicrobial activities of H rhamnoides 100% oil, 100% juice, dry berries, and tea (dry berries, leaves, and twigs). They found that all of the studied sea buckthorn products displayed high antioxidant activity (identified through DPPH radical scavenging and molybdenum reducing antioxidant power tests). Sea buckthorn juice contained the highest total content of polyphenols, flavonoids, and carotenoids. All of the tested products also exhibited substantial antibacterial activity against the tested microbes.20
Burns and Wound Healing
In a preclinical study of the effects of sea buckthorn leaf extracts on wound healing in albino rats using an excision-punch wound model in 2005, Gupta and colleagues found that twice daily topical application of the aqueous leaf extract fostered wound healing. This was indicated by higher hydroxyproline and protein levels, a diminished wound area, and lower lipid peroxide levels. The investigators suggested that sea buckthorn may facilitate wound healing at least in part because of elevated antioxidant activity in the granulation tissue.3
A year later, Wang and colleagues reported on observations of using H rhamnoides oil, a traditional Chinese herbal medicine derived from sea buckthorn fruit, as a burn treatment. In the study, 151 burn patients received an H rhamnoides oil dressing (changed every other day until wound healing) that was covered with a disinfecting dressing. The dressing reduced swelling and effusion, and alleviated pain, with patients receiving the sea buckthorn dressing experiencing greater apparent exudation reduction, pain reduction, and more rapid epithelial cell growth and wound healing than controls (treated only with Vaseline gauze). The difference between the two groups was statistically significant.21
Conclusion
Sea buckthorn has been used for hundreds if not thousands of years in traditional medical applications, including for dermatologic purposes. Emerging data appear to support the use of this dynamic plant for consideration in dermatologic applications. As is often the case, much more work is necessary in the form of randomized controlled trials to determine the effectiveness of sea buckthorn formulations as well as the most appropriate avenues of research or uses for dermatologic application of this traditionally used botanical agent.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur in Miami. She founded the division of cosmetic dermatology at the University of Miami in 1997. The third edition of her bestselling textbook, “Cosmetic Dermatology,” was published in 2022. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Johnson & Johnson, and Burt’s Bees. She is the CEO of Skin Type Solutions, a SaaS company used to generate skin care routines in office and as a e-commerce solution. Write to her at dermnews@mdedge.com.
References
1. Teng H et al. J Ethnopharmacol. 2024 Apr 24;324:117809. doi: 10.1016/j.jep.2024.117809.
2. Wang Z et al. Int J Biol Macromol. 2024 Apr;263(Pt 1):130206. doi: 10.1016/j.ijbiomac.2024.130206.
3. Gupta A et al. Int J Low Extrem Wounds. 2005 Jun;4(2):88-92. doi: 10.1177/1534734605277401.
4. Pundir S et al. J Ethnopharmacol. 2021 Feb 10;266:113434. doi: 10.1016/j.jep.2020.113434.
5. Ma QG et al. J Agric Food Chem. 2023 Mar 29;71(12):4769-4788. doi: 10.1021/acs.jafc.2c06916.
6. Poljšak N et al. Phytother Res. 2020 Feb;34(2):254-269. doi: 10.1002/ptr.6524. doi: 10.1002/ptr.6524.
7. Upadhyay NK et al. Evid Based Complement Alternat Med. 2011;2011:659705. doi: 10.1093/ecam/nep189.
8. Suryakumar G, Gupta A. J Ethnopharmacol. 2011 Nov 18;138(2):268-78. doi: 10.1016/j.jep.2011.09.024.
9. Liu K et al. Front Pharmacol. 2022 Jul 8;13:914146. doi: 10.3389/fphar.2022.914146.
10. Akhtar N et al. J Pharm Bioallied Sci. 2010 Jan;2(1):13-7. doi: 10.4103/0975-7406.62698.
11. Ren R et al. RSC Adv. 2020 Dec 17;10(73):44654-44671. doi: 10.1039/d0ra06488b.
12. Ito H et al. Burns. 2014 May;40(3):511-9. doi: 10.1016/j.burns.2013.08.011.
13. Liu X et al. Food Sci Nutr. 2023 Dec 7;12(2):1082-1094. doi: 10.1002/fsn3.3823.
14. Liu X at al. Antioxidants (Basel). 2022 Sep 25;11(10):1900. doi: 10.3390/antiox11101900.
15. Khan BA, Akhtar N. Postepy Dermatol Alergol. 2014 Aug;31(4):229-234. doi: 10.5114/pdia.2014.40934.
16. Khan BA, Akhtar N. Pak J Pharm Sci. 2014 Nov;27(6):1919-22.
17. Khan AB et al. African J Pharm Pharmacol. 2011 Aug;5(8):1092-5.
18. Khan BA, Akhtar N, Braga VA. Trop J Pharm Res. 2012;11(6):955-62.
19. Gęgotek A et al. Antioxidants (Basel). 2018 Aug 23;7(9):110. doi: 10.3390/antiox7090110.
20. Ivanišová E et al. Acta Sci Pol Technol Aliment. 2020 Apr-Jun;19(2):195-205. doi: 10.17306/J.AFS.0809.
21. Wang ZY, Luo XL, He CP. Nan Fang Yi Ke Da Xue Xue Bao. 2006 Jan;26(1):124-5.
A member of the Elaeagnaceae family, Hippophae rhamnoides, better known as sea buckthorn, is a high-altitude wild shrub endemic to Europe and Asia with edible fruits and a lengthy record of use in traditional Chinese medicine.1-6 Used as a health supplement and consumed in the diet throughout the world,5 sea buckthorn berries, seeds, and leaves have been used in traditional medicine to treat burns/injuries, edema, hypertension, inflammation, skin grafts, ulcers, and wounds.4,7
This hardy plant is associated with a wide range of biologic activities, including anti-atherogenic, anti-atopic dermatitis, antibacterial, anticancer, antifungal, anti-inflammatory, antimicrobial, antioxidant, anti-psoriasis, anti-sebum, anti-stress, anti-tumor, cytoprotective, hepatoprotective, immunomodulatory, neuroprotective, radioprotective, and tissue regenerative functions.4,5,8-11
Key Constituents
Functional constituents identified in sea buckthorn include alkaloids, carotenoids, flavonoids, lignans, organic acids, phenolic acids, proanthocyanidins, polyunsaturated acids (including omega-3, -6, -7, and -9), steroids, tannins, terpenoids, and volatile oils, as well as nutritional compounds such as minerals, proteins, and vitamins.4,5,11 Sea buckthorn pericarp oil contains copious amounts of saturated palmitic acid (29%-36%) and omega-7 unsaturated palmitoleic acid (36%-48%), which fosters cutaneous and mucosal epithelialization, as well as linoleic (10%-12%) and oleic (4%-6%) acids.12,6 Significant amounts of carotenoids as well as alpha‐linolenic fatty acid (38%), linoleic (36%), oleic (13%), and palmitic (7%) acids are present in sea buckthorn seed oil.6
Polysaccharides
In an expansive review on the pharmacological activities of sea buckthorn polysaccharides, Teng and colleagues reported in April 2024 that 20 diverse polysaccharides have been culled from sea buckthorn and exhibited various healthy activities, including antioxidant, anti-fatigue, anti-inflammatory, anti-obesity, anti-tumor, hepatoprotective, hypoglycemic, and immunoregulation, and regulation of intestinal flora activities.1
Proanthocyanidins and Anti-Aging
In 2023, Liu and colleagues investigated the anti–skin aging impact of sea buckthorn proanthocyanidins in D-galactose-induced aging in mice given the known free radical scavenging activity of these compounds. They found the proanthocyanidins mitigated D-galactose-induced aging and can augment the total antioxidant capacity of the body. Sea buckthorn proanthocyanidins can further attenuate the effects of skin aging by regulating the TGF-beta1/Smads pathway and MMPs/TIMP system, thus amplifying collagen I and tropoelastin content.13
A year earlier, many of the same investigators assessed the possible protective activity of sea buckthorn proanthocyanidins against cutaneous aging engendered by oxidative stress from hydrogen peroxide. The compounds amplified superoxide dismutase and glutathione antioxidant functions. The extracts also fostered collagen I production in aging human skin fibroblasts via the TGF-beta1/Smads pathway and hindered collagen I degradation by regulating the MMPs/TIMPs system, which maintained extracellular matrix integrity. Senescent cell migration was also promoted with 100 mcg/mL of sea buckthorn proanthocyanidins. The researchers concluded that this sets the stage for investigating how sea buckthorn proanthocyanidins can be incorporated in cosmetic formulations.14 In a separate study, Liu and colleagues demonstrated that sea buckthorn proanthocyanidins can attenuate oxidative damage and protect mitochondrial function.9
Acne and Barrier Functions
The extracts of H rhamnoides and Cassia fistula in a combined formulation were found to be effective in lowering skin sebum content in humans with grade I and grade II acne vulgaris in a 2014 single-blind, randomized, placebo-controlled, split-face study with two groups of 25 patients each (aged 18-37 years).15 Khan and colleagues have also reported that a sea buckthorn oil-in-water emulsion improved barrier function in human skin as tested by a tewameter and corneometer (noninvasive probes) in 13 healthy males with a mean age of 27 ± 4.8 years.16
Anti-Aging, Antioxidant, Antibacterial, Skin-Whitening Activity
Zaman and colleagues reported in 2011 that results from an in vivo study of the effects of a sea buckthorn fruit extract topical cream on stratum corneum water content and transepidermal water loss indicated that the formulation enhanced cell surface integrin expression thus facilitating collagen contraction.17
In 2012, Khan and colleagues reported amelioration in skin elasticity, thus achieving an anti-aging result, from the use of a water-in-oil–based hydroalcoholic cream loaded with fruit extract of H rhamnoides, as measured with a Cutometer.18 The previous year, some of the same researchers reported that the antioxidants and flavonoids found in a topical sea buckthorn formulation could decrease cutaneous melanin and erythema levels.
More recently, Gęgotek and colleagues found that sea buckthorn seed oil prevented redox balance and lipid metabolism disturbances in skin fibroblasts and keratinocytes caused by UVA or UVB. They suggested that such findings point to the potential of this natural agent to confer anti-inflammatory properties and photoprotection to the skin.19
In 2020, Ivanišová and colleagues investigated the antioxidant and antimicrobial activities of H rhamnoides 100% oil, 100% juice, dry berries, and tea (dry berries, leaves, and twigs). They found that all of the studied sea buckthorn products displayed high antioxidant activity (identified through DPPH radical scavenging and molybdenum reducing antioxidant power tests). Sea buckthorn juice contained the highest total content of polyphenols, flavonoids, and carotenoids. All of the tested products also exhibited substantial antibacterial activity against the tested microbes.20
Burns and Wound Healing
In a preclinical study of the effects of sea buckthorn leaf extracts on wound healing in albino rats using an excision-punch wound model in 2005, Gupta and colleagues found that twice daily topical application of the aqueous leaf extract fostered wound healing. This was indicated by higher hydroxyproline and protein levels, a diminished wound area, and lower lipid peroxide levels. The investigators suggested that sea buckthorn may facilitate wound healing at least in part because of elevated antioxidant activity in the granulation tissue.3
A year later, Wang and colleagues reported on observations of using H rhamnoides oil, a traditional Chinese herbal medicine derived from sea buckthorn fruit, as a burn treatment. In the study, 151 burn patients received an H rhamnoides oil dressing (changed every other day until wound healing) that was covered with a disinfecting dressing. The dressing reduced swelling and effusion, and alleviated pain, with patients receiving the sea buckthorn dressing experiencing greater apparent exudation reduction, pain reduction, and more rapid epithelial cell growth and wound healing than controls (treated only with Vaseline gauze). The difference between the two groups was statistically significant.21
Conclusion
Sea buckthorn has been used for hundreds if not thousands of years in traditional medical applications, including for dermatologic purposes. Emerging data appear to support the use of this dynamic plant for consideration in dermatologic applications. As is often the case, much more work is necessary in the form of randomized controlled trials to determine the effectiveness of sea buckthorn formulations as well as the most appropriate avenues of research or uses for dermatologic application of this traditionally used botanical agent.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur in Miami. She founded the division of cosmetic dermatology at the University of Miami in 1997. The third edition of her bestselling textbook, “Cosmetic Dermatology,” was published in 2022. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Johnson & Johnson, and Burt’s Bees. She is the CEO of Skin Type Solutions, a SaaS company used to generate skin care routines in office and as a e-commerce solution. Write to her at dermnews@mdedge.com.
References
1. Teng H et al. J Ethnopharmacol. 2024 Apr 24;324:117809. doi: 10.1016/j.jep.2024.117809.
2. Wang Z et al. Int J Biol Macromol. 2024 Apr;263(Pt 1):130206. doi: 10.1016/j.ijbiomac.2024.130206.
3. Gupta A et al. Int J Low Extrem Wounds. 2005 Jun;4(2):88-92. doi: 10.1177/1534734605277401.
4. Pundir S et al. J Ethnopharmacol. 2021 Feb 10;266:113434. doi: 10.1016/j.jep.2020.113434.
5. Ma QG et al. J Agric Food Chem. 2023 Mar 29;71(12):4769-4788. doi: 10.1021/acs.jafc.2c06916.
6. Poljšak N et al. Phytother Res. 2020 Feb;34(2):254-269. doi: 10.1002/ptr.6524. doi: 10.1002/ptr.6524.
7. Upadhyay NK et al. Evid Based Complement Alternat Med. 2011;2011:659705. doi: 10.1093/ecam/nep189.
8. Suryakumar G, Gupta A. J Ethnopharmacol. 2011 Nov 18;138(2):268-78. doi: 10.1016/j.jep.2011.09.024.
9. Liu K et al. Front Pharmacol. 2022 Jul 8;13:914146. doi: 10.3389/fphar.2022.914146.
10. Akhtar N et al. J Pharm Bioallied Sci. 2010 Jan;2(1):13-7. doi: 10.4103/0975-7406.62698.
11. Ren R et al. RSC Adv. 2020 Dec 17;10(73):44654-44671. doi: 10.1039/d0ra06488b.
12. Ito H et al. Burns. 2014 May;40(3):511-9. doi: 10.1016/j.burns.2013.08.011.
13. Liu X et al. Food Sci Nutr. 2023 Dec 7;12(2):1082-1094. doi: 10.1002/fsn3.3823.
14. Liu X at al. Antioxidants (Basel). 2022 Sep 25;11(10):1900. doi: 10.3390/antiox11101900.
15. Khan BA, Akhtar N. Postepy Dermatol Alergol. 2014 Aug;31(4):229-234. doi: 10.5114/pdia.2014.40934.
16. Khan BA, Akhtar N. Pak J Pharm Sci. 2014 Nov;27(6):1919-22.
17. Khan AB et al. African J Pharm Pharmacol. 2011 Aug;5(8):1092-5.
18. Khan BA, Akhtar N, Braga VA. Trop J Pharm Res. 2012;11(6):955-62.
19. Gęgotek A et al. Antioxidants (Basel). 2018 Aug 23;7(9):110. doi: 10.3390/antiox7090110.
20. Ivanišová E et al. Acta Sci Pol Technol Aliment. 2020 Apr-Jun;19(2):195-205. doi: 10.17306/J.AFS.0809.
21. Wang ZY, Luo XL, He CP. Nan Fang Yi Ke Da Xue Xue Bao. 2006 Jan;26(1):124-5.
A member of the Elaeagnaceae family, Hippophae rhamnoides, better known as sea buckthorn, is a high-altitude wild shrub endemic to Europe and Asia with edible fruits and a lengthy record of use in traditional Chinese medicine.1-6 Used as a health supplement and consumed in the diet throughout the world,5 sea buckthorn berries, seeds, and leaves have been used in traditional medicine to treat burns/injuries, edema, hypertension, inflammation, skin grafts, ulcers, and wounds.4,7
This hardy plant is associated with a wide range of biologic activities, including anti-atherogenic, anti-atopic dermatitis, antibacterial, anticancer, antifungal, anti-inflammatory, antimicrobial, antioxidant, anti-psoriasis, anti-sebum, anti-stress, anti-tumor, cytoprotective, hepatoprotective, immunomodulatory, neuroprotective, radioprotective, and tissue regenerative functions.4,5,8-11
Key Constituents
Functional constituents identified in sea buckthorn include alkaloids, carotenoids, flavonoids, lignans, organic acids, phenolic acids, proanthocyanidins, polyunsaturated acids (including omega-3, -6, -7, and -9), steroids, tannins, terpenoids, and volatile oils, as well as nutritional compounds such as minerals, proteins, and vitamins.4,5,11 Sea buckthorn pericarp oil contains copious amounts of saturated palmitic acid (29%-36%) and omega-7 unsaturated palmitoleic acid (36%-48%), which fosters cutaneous and mucosal epithelialization, as well as linoleic (10%-12%) and oleic (4%-6%) acids.12,6 Significant amounts of carotenoids as well as alpha‐linolenic fatty acid (38%), linoleic (36%), oleic (13%), and palmitic (7%) acids are present in sea buckthorn seed oil.6
Polysaccharides
In an expansive review on the pharmacological activities of sea buckthorn polysaccharides, Teng and colleagues reported in April 2024 that 20 diverse polysaccharides have been culled from sea buckthorn and exhibited various healthy activities, including antioxidant, anti-fatigue, anti-inflammatory, anti-obesity, anti-tumor, hepatoprotective, hypoglycemic, and immunoregulation, and regulation of intestinal flora activities.1
Proanthocyanidins and Anti-Aging
In 2023, Liu and colleagues investigated the anti–skin aging impact of sea buckthorn proanthocyanidins in D-galactose-induced aging in mice given the known free radical scavenging activity of these compounds. They found the proanthocyanidins mitigated D-galactose-induced aging and can augment the total antioxidant capacity of the body. Sea buckthorn proanthocyanidins can further attenuate the effects of skin aging by regulating the TGF-beta1/Smads pathway and MMPs/TIMP system, thus amplifying collagen I and tropoelastin content.13
A year earlier, many of the same investigators assessed the possible protective activity of sea buckthorn proanthocyanidins against cutaneous aging engendered by oxidative stress from hydrogen peroxide. The compounds amplified superoxide dismutase and glutathione antioxidant functions. The extracts also fostered collagen I production in aging human skin fibroblasts via the TGF-beta1/Smads pathway and hindered collagen I degradation by regulating the MMPs/TIMPs system, which maintained extracellular matrix integrity. Senescent cell migration was also promoted with 100 mcg/mL of sea buckthorn proanthocyanidins. The researchers concluded that this sets the stage for investigating how sea buckthorn proanthocyanidins can be incorporated in cosmetic formulations.14 In a separate study, Liu and colleagues demonstrated that sea buckthorn proanthocyanidins can attenuate oxidative damage and protect mitochondrial function.9
Acne and Barrier Functions
The extracts of H rhamnoides and Cassia fistula in a combined formulation were found to be effective in lowering skin sebum content in humans with grade I and grade II acne vulgaris in a 2014 single-blind, randomized, placebo-controlled, split-face study with two groups of 25 patients each (aged 18-37 years).15 Khan and colleagues have also reported that a sea buckthorn oil-in-water emulsion improved barrier function in human skin as tested by a tewameter and corneometer (noninvasive probes) in 13 healthy males with a mean age of 27 ± 4.8 years.16
Anti-Aging, Antioxidant, Antibacterial, Skin-Whitening Activity
Zaman and colleagues reported in 2011 that results from an in vivo study of the effects of a sea buckthorn fruit extract topical cream on stratum corneum water content and transepidermal water loss indicated that the formulation enhanced cell surface integrin expression thus facilitating collagen contraction.17
In 2012, Khan and colleagues reported amelioration in skin elasticity, thus achieving an anti-aging result, from the use of a water-in-oil–based hydroalcoholic cream loaded with fruit extract of H rhamnoides, as measured with a Cutometer.18 The previous year, some of the same researchers reported that the antioxidants and flavonoids found in a topical sea buckthorn formulation could decrease cutaneous melanin and erythema levels.
More recently, Gęgotek and colleagues found that sea buckthorn seed oil prevented redox balance and lipid metabolism disturbances in skin fibroblasts and keratinocytes caused by UVA or UVB. They suggested that such findings point to the potential of this natural agent to confer anti-inflammatory properties and photoprotection to the skin.19
In 2020, Ivanišová and colleagues investigated the antioxidant and antimicrobial activities of H rhamnoides 100% oil, 100% juice, dry berries, and tea (dry berries, leaves, and twigs). They found that all of the studied sea buckthorn products displayed high antioxidant activity (identified through DPPH radical scavenging and molybdenum reducing antioxidant power tests). Sea buckthorn juice contained the highest total content of polyphenols, flavonoids, and carotenoids. All of the tested products also exhibited substantial antibacterial activity against the tested microbes.20
Burns and Wound Healing
In a preclinical study of the effects of sea buckthorn leaf extracts on wound healing in albino rats using an excision-punch wound model in 2005, Gupta and colleagues found that twice daily topical application of the aqueous leaf extract fostered wound healing. This was indicated by higher hydroxyproline and protein levels, a diminished wound area, and lower lipid peroxide levels. The investigators suggested that sea buckthorn may facilitate wound healing at least in part because of elevated antioxidant activity in the granulation tissue.3
A year later, Wang and colleagues reported on observations of using H rhamnoides oil, a traditional Chinese herbal medicine derived from sea buckthorn fruit, as a burn treatment. In the study, 151 burn patients received an H rhamnoides oil dressing (changed every other day until wound healing) that was covered with a disinfecting dressing. The dressing reduced swelling and effusion, and alleviated pain, with patients receiving the sea buckthorn dressing experiencing greater apparent exudation reduction, pain reduction, and more rapid epithelial cell growth and wound healing than controls (treated only with Vaseline gauze). The difference between the two groups was statistically significant.21
Conclusion
Sea buckthorn has been used for hundreds if not thousands of years in traditional medical applications, including for dermatologic purposes. Emerging data appear to support the use of this dynamic plant for consideration in dermatologic applications. As is often the case, much more work is necessary in the form of randomized controlled trials to determine the effectiveness of sea buckthorn formulations as well as the most appropriate avenues of research or uses for dermatologic application of this traditionally used botanical agent.
Dr. Baumann is a private practice dermatologist, researcher, author, and entrepreneur in Miami. She founded the division of cosmetic dermatology at the University of Miami in 1997. The third edition of her bestselling textbook, “Cosmetic Dermatology,” was published in 2022. Dr. Baumann has received funding for advisory boards and/or clinical research trials from Allergan, Galderma, Johnson & Johnson, and Burt’s Bees. She is the CEO of Skin Type Solutions, a SaaS company used to generate skin care routines in office and as a e-commerce solution. Write to her at dermnews@mdedge.com.
References
1. Teng H et al. J Ethnopharmacol. 2024 Apr 24;324:117809. doi: 10.1016/j.jep.2024.117809.
2. Wang Z et al. Int J Biol Macromol. 2024 Apr;263(Pt 1):130206. doi: 10.1016/j.ijbiomac.2024.130206.
3. Gupta A et al. Int J Low Extrem Wounds. 2005 Jun;4(2):88-92. doi: 10.1177/1534734605277401.
4. Pundir S et al. J Ethnopharmacol. 2021 Feb 10;266:113434. doi: 10.1016/j.jep.2020.113434.
5. Ma QG et al. J Agric Food Chem. 2023 Mar 29;71(12):4769-4788. doi: 10.1021/acs.jafc.2c06916.
6. Poljšak N et al. Phytother Res. 2020 Feb;34(2):254-269. doi: 10.1002/ptr.6524. doi: 10.1002/ptr.6524.
7. Upadhyay NK et al. Evid Based Complement Alternat Med. 2011;2011:659705. doi: 10.1093/ecam/nep189.
8. Suryakumar G, Gupta A. J Ethnopharmacol. 2011 Nov 18;138(2):268-78. doi: 10.1016/j.jep.2011.09.024.
9. Liu K et al. Front Pharmacol. 2022 Jul 8;13:914146. doi: 10.3389/fphar.2022.914146.
10. Akhtar N et al. J Pharm Bioallied Sci. 2010 Jan;2(1):13-7. doi: 10.4103/0975-7406.62698.
11. Ren R et al. RSC Adv. 2020 Dec 17;10(73):44654-44671. doi: 10.1039/d0ra06488b.
12. Ito H et al. Burns. 2014 May;40(3):511-9. doi: 10.1016/j.burns.2013.08.011.
13. Liu X et al. Food Sci Nutr. 2023 Dec 7;12(2):1082-1094. doi: 10.1002/fsn3.3823.
14. Liu X at al. Antioxidants (Basel). 2022 Sep 25;11(10):1900. doi: 10.3390/antiox11101900.
15. Khan BA, Akhtar N. Postepy Dermatol Alergol. 2014 Aug;31(4):229-234. doi: 10.5114/pdia.2014.40934.
16. Khan BA, Akhtar N. Pak J Pharm Sci. 2014 Nov;27(6):1919-22.
17. Khan AB et al. African J Pharm Pharmacol. 2011 Aug;5(8):1092-5.
18. Khan BA, Akhtar N, Braga VA. Trop J Pharm Res. 2012;11(6):955-62.
19. Gęgotek A et al. Antioxidants (Basel). 2018 Aug 23;7(9):110. doi: 10.3390/antiox7090110.
20. Ivanišová E et al. Acta Sci Pol Technol Aliment. 2020 Apr-Jun;19(2):195-205. doi: 10.17306/J.AFS.0809.
21. Wang ZY, Luo XL, He CP. Nan Fang Yi Ke Da Xue Xue Bao. 2006 Jan;26(1):124-5.
Gardasil 9 at 10 Years: Vaccine Protects Against Multiple Cancers
Vaccination against human papilloma virus (HPV), a group of more than 200 viruses infecting at least 50% of sexually active people over their lifetimes, has proved more than 90% effective for preventing several diseases caused by high-risk HPV types.
Gardasil 4: 2006
It started in 2006 with the approval of Human Papillomavirus Quadrivalent, types 6, 11, 16, and 18 (Gardasil 4). Merck’s vaccine began to lower rates of cervical cancer, a major global killer of women.
“It’s fair to say the vaccine has been an American and a global public health success story in reducing rates of cervical cancer,” Paula M. Cuccaro, PhD, assistant professor of health promotion and behavioral sciences at University of Texas School of Public Health, Houston, said in an interview.
How does a common virus trigger such a lethal gynecologic malignancy? “It knocks out two important cancer suppressor genes in cells,” explained Christina Annunziata,MD, PhD, a medical oncologist and senior vice president of extramural discovery science for the American Cancer Society. HPV oncoproteins are encoded by the E6 and E7 genes. As in other DNA tumor viruses, the E6 and E7 proteins functionally inactivate the tumor suppressor proteins p53 and pRB, respectively.
US Prevalence
Despite screening and vaccination, cervical cancer is still very much around. This year, 13,820 new cases of invasive cervical cancer will be diagnosed in the United States, and approximately 4360 women will die of it, according to the American Cancer Society. Even before the advent of Gardasil 4, incidence rates had already decreased by more than half from the mid-1970s to the mid-2000s, thanks largely to Pap smear screening programs for treatable premalignant lesions. “The US rate had dropped to about 20 per 100,000 women even before Gardasil 4,” said Annunziata. “After the introduction of the first vaccine, it decreased to 7 per 100,000, a decrease of about 30%, but it remains plateaued now at about the same level.”
Although the past decade has seen rates generally stabilize, there have been some changes in different age groups. In women ages 30-44, rates increased 1.7% each year from 2012 to 2019, while rates declined 11% each year for women ages 20-24— probably reflecting the impact of the first wave of prevention from Gardasil 4.
In one 2021 population-based study of US cancer registry data from 1999 to 2017, rates of both cervical squamous cell carcinoma and adenocarcinoma dropped. The largest declines occurred in females 15-20 years old, the age group most likely to be vaccinated against HPV but not typically screened, suggesting a vaccine-related effect.
Gardasil 9: 2014
With the 2014 approval of the vaccine’s second iteration, Gardasil 9, which replaced Gardasil 4 and targeted 9 HPV strains, immunization has taken broader aim. The strains covered by Gardasil 9 protect against oropharyngeal and other head and neck cancers — as well as penile, anal, vulvar, and vaginal malignancies and premalignancies, and genital warts in both sexes ages 9-45.
It may be years, however, before the impact of the newer polyvalent formulation is felt. “While the first vaccine has been successful against the prevalent strains of HPV linked to cervical cancer, it’s a little early to call it for the newer vaccine since oropharyngeal cancers tend to develop later in older men,” Cuccaro said. “But the types of HPV linked to mouth and throat cancers and covered by the newer vaccines are much less prevalent in those who are vaccinated. The strains not covered in the vaccine you see are equally present in the vaccinated and non-vaccinated.”
Angela L. Myers, MD, MPH, division director of infectious diseases and medical director of the Center for Wellbeing at Children’s Mercy in Kansas City, Missouri, added, “Unlike for cervical cancer, there are no screening programs for oropharyngeal lesions, so you have to wait to see rates until actual cancer develops.”
A 2023 review reported that HPV vaccination reduced levels of oropharyngeal HPV positivity in men, strengthening the case for pangender immunization.
And in a recent phase 3 doubled-blind trial, GARDASIL 9 reduced the incidence of anogenital persistent infection caused by nine types of HPV compared with a placebo.
Increasing Uptake
The current public health aim is to have 80% of young people in the targeted age group vaccinated with two doses. Today, uptake among those 9-26 years old stands at about 78% of girls and 75% of boys for the first dose, said Annunziata. “But it’s only about 61% for the two doses in the current series, and we want to improve that.”
Some parents may still harbor fears that immunizing teens and tweens — both the American Academy of Pediatrics and the American Cancer Society recommend immunization at age 9 — will open the door to precocious sexual activity.
“But overall, uptake in tweens and young teens has increased because the messaging has changed,” said Myers, with the rationale now focusing on cancer prevention not sexual-infection prophylaxis. “This is similar to the hepatitis B vaccine, which used to be given to young adults and is now given to newborns to prevent cancer.”
Cuccaro added that a proactive presentation by healthcare professionals has a significant effect on vaccine uptake and increases the odds of vaccination ninefold. “Providers should take a presumptive approach and avoid just offering the vaccine as an option. It should be included with regular childhood vaccinations,” she said. “And the advantage of starting early at age 9 is that you can spread the doses out across other regular childhood vaccinations, whereas if you start at age 11, you need to add the HPV vaccine to three other vaccines that are given at that time.”
After age 15, three doses are necessary. “Providers should stress to parents that it’s most effective when given before young people become sexually active and exposed to HPV,” Cuccaro said. And Myers stressed that despite the vaccine’s effectiveness, routine screening for cervical premalignancies is still important.
Despite increasing coverage, vaccination rates have some distance to go before the public health target of at least 80% uptake of the series in the targeted age group, Cuccaro cautioned.
On the global stage, barriers to immunization remain, but the World Health Organization has endorsed a campaign to eradicate cervical cancer through HPV vaccination. It has predicted that the 21st century may be the last to experience HPV-associated cancers, currently responsible for more than 300,000 annual deaths worldwide.
A Brief History of HPV Vaccines
- 1951. Cervical cancer patient Henrietta Lacks’ rapidly dividing cervical cells are collected by George Otto Gey at Johns Hopkins Hospital. They create the first immortal cell line (HeLa) used to study cancers and vaccines worldwide.
- 1976. Harald zur Hausen suggests that genital wart-associated HPV, not herpes simplex, is the probable cause of cervical cancer.
- 1983. HPV is confirmed as a cause of cancer.
- 1991. The first HPV vaccine is developed.
- 2002. Proof of principle and protective efficacy for the monovalent HPV 16 are shown.
- 2006. Merck’s Gardasil 4 (HPV 4) is FDA approved in girls ages 9-26 for protection against strains 6, 11, 16, and 18 — the cause of more than 70% of cervical cancer cases.
- 2009. Approval of Gardasil 4 is expanded to boys ages 9-26 for the prevention of genital warts.
- 2009. The FDA approves GlaxoSmithKline’s Cervarix (HPV 16 and 18) for girls and young women. The vaccine was withdrawn from the US market in 2016 following the success of Gardasil 9 but is used abroad for HPV cancer prevention.
- 2014. The 9-valent recombinant vaccine Gardasil 9 is FDA approved for protection against several low-risk, wart-causing HPV strains as well as the high-risk cancer strains targeted by HPV 4.
- 2018. The FDA expands approval to include females and males 27-45 years old.
- 2020. The FDA extends approval of Gardasil 9 to include prevention not only of cervical cancer but also, vaginal, vulvar, anal, oropharyngeal, and other head and neck cancers.
Annunziata, Cuccaro, and Myers had no competing interests to declare.
A version of this article appeared on Medscape.com.
Vaccination against human papilloma virus (HPV), a group of more than 200 viruses infecting at least 50% of sexually active people over their lifetimes, has proved more than 90% effective for preventing several diseases caused by high-risk HPV types.
Gardasil 4: 2006
It started in 2006 with the approval of Human Papillomavirus Quadrivalent, types 6, 11, 16, and 18 (Gardasil 4). Merck’s vaccine began to lower rates of cervical cancer, a major global killer of women.
“It’s fair to say the vaccine has been an American and a global public health success story in reducing rates of cervical cancer,” Paula M. Cuccaro, PhD, assistant professor of health promotion and behavioral sciences at University of Texas School of Public Health, Houston, said in an interview.
How does a common virus trigger such a lethal gynecologic malignancy? “It knocks out two important cancer suppressor genes in cells,” explained Christina Annunziata,MD, PhD, a medical oncologist and senior vice president of extramural discovery science for the American Cancer Society. HPV oncoproteins are encoded by the E6 and E7 genes. As in other DNA tumor viruses, the E6 and E7 proteins functionally inactivate the tumor suppressor proteins p53 and pRB, respectively.
US Prevalence
Despite screening and vaccination, cervical cancer is still very much around. This year, 13,820 new cases of invasive cervical cancer will be diagnosed in the United States, and approximately 4360 women will die of it, according to the American Cancer Society. Even before the advent of Gardasil 4, incidence rates had already decreased by more than half from the mid-1970s to the mid-2000s, thanks largely to Pap smear screening programs for treatable premalignant lesions. “The US rate had dropped to about 20 per 100,000 women even before Gardasil 4,” said Annunziata. “After the introduction of the first vaccine, it decreased to 7 per 100,000, a decrease of about 30%, but it remains plateaued now at about the same level.”
Although the past decade has seen rates generally stabilize, there have been some changes in different age groups. In women ages 30-44, rates increased 1.7% each year from 2012 to 2019, while rates declined 11% each year for women ages 20-24— probably reflecting the impact of the first wave of prevention from Gardasil 4.
In one 2021 population-based study of US cancer registry data from 1999 to 2017, rates of both cervical squamous cell carcinoma and adenocarcinoma dropped. The largest declines occurred in females 15-20 years old, the age group most likely to be vaccinated against HPV but not typically screened, suggesting a vaccine-related effect.
Gardasil 9: 2014
With the 2014 approval of the vaccine’s second iteration, Gardasil 9, which replaced Gardasil 4 and targeted 9 HPV strains, immunization has taken broader aim. The strains covered by Gardasil 9 protect against oropharyngeal and other head and neck cancers — as well as penile, anal, vulvar, and vaginal malignancies and premalignancies, and genital warts in both sexes ages 9-45.
It may be years, however, before the impact of the newer polyvalent formulation is felt. “While the first vaccine has been successful against the prevalent strains of HPV linked to cervical cancer, it’s a little early to call it for the newer vaccine since oropharyngeal cancers tend to develop later in older men,” Cuccaro said. “But the types of HPV linked to mouth and throat cancers and covered by the newer vaccines are much less prevalent in those who are vaccinated. The strains not covered in the vaccine you see are equally present in the vaccinated and non-vaccinated.”
Angela L. Myers, MD, MPH, division director of infectious diseases and medical director of the Center for Wellbeing at Children’s Mercy in Kansas City, Missouri, added, “Unlike for cervical cancer, there are no screening programs for oropharyngeal lesions, so you have to wait to see rates until actual cancer develops.”
A 2023 review reported that HPV vaccination reduced levels of oropharyngeal HPV positivity in men, strengthening the case for pangender immunization.
And in a recent phase 3 doubled-blind trial, GARDASIL 9 reduced the incidence of anogenital persistent infection caused by nine types of HPV compared with a placebo.
Increasing Uptake
The current public health aim is to have 80% of young people in the targeted age group vaccinated with two doses. Today, uptake among those 9-26 years old stands at about 78% of girls and 75% of boys for the first dose, said Annunziata. “But it’s only about 61% for the two doses in the current series, and we want to improve that.”
Some parents may still harbor fears that immunizing teens and tweens — both the American Academy of Pediatrics and the American Cancer Society recommend immunization at age 9 — will open the door to precocious sexual activity.
“But overall, uptake in tweens and young teens has increased because the messaging has changed,” said Myers, with the rationale now focusing on cancer prevention not sexual-infection prophylaxis. “This is similar to the hepatitis B vaccine, which used to be given to young adults and is now given to newborns to prevent cancer.”
Cuccaro added that a proactive presentation by healthcare professionals has a significant effect on vaccine uptake and increases the odds of vaccination ninefold. “Providers should take a presumptive approach and avoid just offering the vaccine as an option. It should be included with regular childhood vaccinations,” she said. “And the advantage of starting early at age 9 is that you can spread the doses out across other regular childhood vaccinations, whereas if you start at age 11, you need to add the HPV vaccine to three other vaccines that are given at that time.”
After age 15, three doses are necessary. “Providers should stress to parents that it’s most effective when given before young people become sexually active and exposed to HPV,” Cuccaro said. And Myers stressed that despite the vaccine’s effectiveness, routine screening for cervical premalignancies is still important.
Despite increasing coverage, vaccination rates have some distance to go before the public health target of at least 80% uptake of the series in the targeted age group, Cuccaro cautioned.
On the global stage, barriers to immunization remain, but the World Health Organization has endorsed a campaign to eradicate cervical cancer through HPV vaccination. It has predicted that the 21st century may be the last to experience HPV-associated cancers, currently responsible for more than 300,000 annual deaths worldwide.
A Brief History of HPV Vaccines
- 1951. Cervical cancer patient Henrietta Lacks’ rapidly dividing cervical cells are collected by George Otto Gey at Johns Hopkins Hospital. They create the first immortal cell line (HeLa) used to study cancers and vaccines worldwide.
- 1976. Harald zur Hausen suggests that genital wart-associated HPV, not herpes simplex, is the probable cause of cervical cancer.
- 1983. HPV is confirmed as a cause of cancer.
- 1991. The first HPV vaccine is developed.
- 2002. Proof of principle and protective efficacy for the monovalent HPV 16 are shown.
- 2006. Merck’s Gardasil 4 (HPV 4) is FDA approved in girls ages 9-26 for protection against strains 6, 11, 16, and 18 — the cause of more than 70% of cervical cancer cases.
- 2009. Approval of Gardasil 4 is expanded to boys ages 9-26 for the prevention of genital warts.
- 2009. The FDA approves GlaxoSmithKline’s Cervarix (HPV 16 and 18) for girls and young women. The vaccine was withdrawn from the US market in 2016 following the success of Gardasil 9 but is used abroad for HPV cancer prevention.
- 2014. The 9-valent recombinant vaccine Gardasil 9 is FDA approved for protection against several low-risk, wart-causing HPV strains as well as the high-risk cancer strains targeted by HPV 4.
- 2018. The FDA expands approval to include females and males 27-45 years old.
- 2020. The FDA extends approval of Gardasil 9 to include prevention not only of cervical cancer but also, vaginal, vulvar, anal, oropharyngeal, and other head and neck cancers.
Annunziata, Cuccaro, and Myers had no competing interests to declare.
A version of this article appeared on Medscape.com.
Vaccination against human papilloma virus (HPV), a group of more than 200 viruses infecting at least 50% of sexually active people over their lifetimes, has proved more than 90% effective for preventing several diseases caused by high-risk HPV types.
Gardasil 4: 2006
It started in 2006 with the approval of Human Papillomavirus Quadrivalent, types 6, 11, 16, and 18 (Gardasil 4). Merck’s vaccine began to lower rates of cervical cancer, a major global killer of women.
“It’s fair to say the vaccine has been an American and a global public health success story in reducing rates of cervical cancer,” Paula M. Cuccaro, PhD, assistant professor of health promotion and behavioral sciences at University of Texas School of Public Health, Houston, said in an interview.
How does a common virus trigger such a lethal gynecologic malignancy? “It knocks out two important cancer suppressor genes in cells,” explained Christina Annunziata,MD, PhD, a medical oncologist and senior vice president of extramural discovery science for the American Cancer Society. HPV oncoproteins are encoded by the E6 and E7 genes. As in other DNA tumor viruses, the E6 and E7 proteins functionally inactivate the tumor suppressor proteins p53 and pRB, respectively.
US Prevalence
Despite screening and vaccination, cervical cancer is still very much around. This year, 13,820 new cases of invasive cervical cancer will be diagnosed in the United States, and approximately 4360 women will die of it, according to the American Cancer Society. Even before the advent of Gardasil 4, incidence rates had already decreased by more than half from the mid-1970s to the mid-2000s, thanks largely to Pap smear screening programs for treatable premalignant lesions. “The US rate had dropped to about 20 per 100,000 women even before Gardasil 4,” said Annunziata. “After the introduction of the first vaccine, it decreased to 7 per 100,000, a decrease of about 30%, but it remains plateaued now at about the same level.”
Although the past decade has seen rates generally stabilize, there have been some changes in different age groups. In women ages 30-44, rates increased 1.7% each year from 2012 to 2019, while rates declined 11% each year for women ages 20-24— probably reflecting the impact of the first wave of prevention from Gardasil 4.
In one 2021 population-based study of US cancer registry data from 1999 to 2017, rates of both cervical squamous cell carcinoma and adenocarcinoma dropped. The largest declines occurred in females 15-20 years old, the age group most likely to be vaccinated against HPV but not typically screened, suggesting a vaccine-related effect.
Gardasil 9: 2014
With the 2014 approval of the vaccine’s second iteration, Gardasil 9, which replaced Gardasil 4 and targeted 9 HPV strains, immunization has taken broader aim. The strains covered by Gardasil 9 protect against oropharyngeal and other head and neck cancers — as well as penile, anal, vulvar, and vaginal malignancies and premalignancies, and genital warts in both sexes ages 9-45.
It may be years, however, before the impact of the newer polyvalent formulation is felt. “While the first vaccine has been successful against the prevalent strains of HPV linked to cervical cancer, it’s a little early to call it for the newer vaccine since oropharyngeal cancers tend to develop later in older men,” Cuccaro said. “But the types of HPV linked to mouth and throat cancers and covered by the newer vaccines are much less prevalent in those who are vaccinated. The strains not covered in the vaccine you see are equally present in the vaccinated and non-vaccinated.”
Angela L. Myers, MD, MPH, division director of infectious diseases and medical director of the Center for Wellbeing at Children’s Mercy in Kansas City, Missouri, added, “Unlike for cervical cancer, there are no screening programs for oropharyngeal lesions, so you have to wait to see rates until actual cancer develops.”
A 2023 review reported that HPV vaccination reduced levels of oropharyngeal HPV positivity in men, strengthening the case for pangender immunization.
And in a recent phase 3 doubled-blind trial, GARDASIL 9 reduced the incidence of anogenital persistent infection caused by nine types of HPV compared with a placebo.
Increasing Uptake
The current public health aim is to have 80% of young people in the targeted age group vaccinated with two doses. Today, uptake among those 9-26 years old stands at about 78% of girls and 75% of boys for the first dose, said Annunziata. “But it’s only about 61% for the two doses in the current series, and we want to improve that.”
Some parents may still harbor fears that immunizing teens and tweens — both the American Academy of Pediatrics and the American Cancer Society recommend immunization at age 9 — will open the door to precocious sexual activity.
“But overall, uptake in tweens and young teens has increased because the messaging has changed,” said Myers, with the rationale now focusing on cancer prevention not sexual-infection prophylaxis. “This is similar to the hepatitis B vaccine, which used to be given to young adults and is now given to newborns to prevent cancer.”
Cuccaro added that a proactive presentation by healthcare professionals has a significant effect on vaccine uptake and increases the odds of vaccination ninefold. “Providers should take a presumptive approach and avoid just offering the vaccine as an option. It should be included with regular childhood vaccinations,” she said. “And the advantage of starting early at age 9 is that you can spread the doses out across other regular childhood vaccinations, whereas if you start at age 11, you need to add the HPV vaccine to three other vaccines that are given at that time.”
After age 15, three doses are necessary. “Providers should stress to parents that it’s most effective when given before young people become sexually active and exposed to HPV,” Cuccaro said. And Myers stressed that despite the vaccine’s effectiveness, routine screening for cervical premalignancies is still important.
Despite increasing coverage, vaccination rates have some distance to go before the public health target of at least 80% uptake of the series in the targeted age group, Cuccaro cautioned.
On the global stage, barriers to immunization remain, but the World Health Organization has endorsed a campaign to eradicate cervical cancer through HPV vaccination. It has predicted that the 21st century may be the last to experience HPV-associated cancers, currently responsible for more than 300,000 annual deaths worldwide.
A Brief History of HPV Vaccines
- 1951. Cervical cancer patient Henrietta Lacks’ rapidly dividing cervical cells are collected by George Otto Gey at Johns Hopkins Hospital. They create the first immortal cell line (HeLa) used to study cancers and vaccines worldwide.
- 1976. Harald zur Hausen suggests that genital wart-associated HPV, not herpes simplex, is the probable cause of cervical cancer.
- 1983. HPV is confirmed as a cause of cancer.
- 1991. The first HPV vaccine is developed.
- 2002. Proof of principle and protective efficacy for the monovalent HPV 16 are shown.
- 2006. Merck’s Gardasil 4 (HPV 4) is FDA approved in girls ages 9-26 for protection against strains 6, 11, 16, and 18 — the cause of more than 70% of cervical cancer cases.
- 2009. Approval of Gardasil 4 is expanded to boys ages 9-26 for the prevention of genital warts.
- 2009. The FDA approves GlaxoSmithKline’s Cervarix (HPV 16 and 18) for girls and young women. The vaccine was withdrawn from the US market in 2016 following the success of Gardasil 9 but is used abroad for HPV cancer prevention.
- 2014. The 9-valent recombinant vaccine Gardasil 9 is FDA approved for protection against several low-risk, wart-causing HPV strains as well as the high-risk cancer strains targeted by HPV 4.
- 2018. The FDA expands approval to include females and males 27-45 years old.
- 2020. The FDA extends approval of Gardasil 9 to include prevention not only of cervical cancer but also, vaginal, vulvar, anal, oropharyngeal, and other head and neck cancers.
Annunziata, Cuccaro, and Myers had no competing interests to declare.
A version of this article appeared on Medscape.com.
Parent Perceptions Drive Diet Changes for Children With Atopic Dermatitis
based on survey data from nearly 300 parents.
Although atopic dermatitis can be associated with an increased risk for food allergies, major allergy organizations do not currently recommend elimination diets as a treatment for atopic dermatitis, said Nadia Makkoukdji, MD, a pediatrician at Jackson Memorial Hospital, Miami, in a presentation at the American College of Allergy, Asthma, and Immunology (ACAAI) Annual Scientific Meeting.
“A fear of drastic dietary changes often prevents families from seeking the care their children need,” Makkoukdji said in an interview. In the clinical setting, Makkoukdji noted that she has seen many patients who have started food elimination diets on their own or as recommended by other doctors, and that these diets can lead to dangers such as the development of immunoglobulin E–mediated food allergies on reintroduction of eliminated foods and malnutrition. They can also produce “emotional stress in children and anxiety or depression, while also adding stress to parents and the entire family.”
Makkoukdji conducted the study to explore parents’ perceptions of these diets in management of their children’s atopic dermatitis, she said.
In the study, Makkoukdji and colleagues sought to understand parents’ perceptions of the role of diet in atopic dermatitis in their children. The researchers reviewed surveys from 298 parents of children with atopic dermatitis who were seen at a single academic center. Parents completed the surveys in the emergency department or in an allergy, dermatology, and general pediatrics clinic.
Overall, 42% of parents identified food triggers for their child’s atopic dermatitis. The most commonly identified triggers were milk (32%), tree nuts/seeds/peanuts (16%), and eggs (11%).
Of the parents who reported food triggers, 23% removed the suspected trigger food from the child’s diet completely, 20% removed suspected trigger foods from their own diets while breastfeeding, and 19% changed their infant’s formula.
In the wake of the elimination diets, 38% of the parents reported no improvement in their child’s atopic dermatitis, 35% reported a 25% improvement, and 9% reported complete resolution. The majority (79%) reintroduced eliminated foods and reported no recurrence of atopic dermatitis symptoms.
The researchers were surprised by how many parents changed their child’s diet in the belief that certain foods exacerbated their child’s atopic dermatitis, “although this perception aligns with the common concern that food allergens can trigger or worsen atopic dermatitis flares,” Makkoukdji said.
The current study highlights the need for more awareness of the limited impact of dietary modifications on atopic dermatitis in the absence of confirmed food allergies, Makkoukdji said. “Our study shows that food elimination diets are still commonly being used by parents in the local Miami population.”
The findings were limited by several factors, including the use of data from a single center and the focus only on pediatric patients, but the primary goal was to assess parental perceptions of AD flares in relation to dietary choices, said Makkoukdji. “Future studies that include larger and more diverse populations would be valuable for the field.”
Dietary Modifications Don’t Live Up to Hype
“Food continues to be one of the most discussed aspects of atopic dermatitis,” Peter Lio, MD, clinical assistant professor of dermatology and pediatrics at Northwestern University Feinberg School of Medicine, Chicago, Illinois, said in an interview.
“Almost all of my patients and families ask about dietary modifications, even though almost all of them have experimented with it to some degree,” said Lio. In his experience, diet plays a small role, if any, in the day-to-day management of atopic dermatitis.
This lack of effect of dietary changes is often frustrating to patients because of the persistent “common wisdom” that points to diet as a root cause of atopic dermatitis, Lio said. “Many practitioners continue to recommend excluding foods such as gluten or dairy from the diet, but generally these are only of modest help,” and although patients wish that dietary changes would fix the problem, most are left wondering why these changes didn’t help them.
The current study findings “reflect my own experience after nearly 20 years of being deeply immersed in the world of atopic dermatitis,” Lio said. Although the takeaway message does not argue against eating healthy foods, some foods do seem to make AD worse in some patients and may have nonallergic pro-inflammatory effects.
“In those cases, it is reasonable to limit or avoid those foods. However, it is extremely difficult to tell what food or foods are driving flare-ups when things are out of control, so dietary modification is generally not the best place to start,” he said.
True food allergies are much more common in patients with atopic dermatitis compared with individuals without atopic dermatitis, but the current study is not addressing these types of allergies, Lio emphasized. “If someone has true allergy to peanuts, for example, they should not be eating them; we also know that they are not ‘cheating’ because these patients would not merely have an eczema flare; they would have urticaria, angioedema, or anaphylaxis. There is tremendous confusion around this point and lots of confusion around allergy testing and its limitations.”
In addition, patients with atopic dermatitis are more likely than those without atopic dermatitis to have abnormalities in the gut microbiome and gut barrier, Lio said.
Abnormalities in the gut microbiome are different from the concept of allergy and may fall into the more complex category of barrier and microbiome disruptors, he said. Therefore, “the food category may not be nearly as important as the specific preparation of the food along with the additives (such as preservatives and emulsifiers) that may actually be driving the problem.”
Although in the past many clinicians advised patients to try cutting out certain foods to see whether atopic dermatitis symptoms improved, this strategy is not without risk, said Lio. “There have been incredible advancements in understanding the role of the gut in tolerization to foods.” Recent research has shown that by eating foods regularly, particularly those such as peanuts that seem to have more allergic potential, the body becomes tolerant, and this prevents the development of true food allergies.
As for additional research, many questions remain about the effects of types of foods, processing methods, and timing of introduction of foods on atopic dermatitis, Lio noted.
“Atopic dermatitis is a systemic condition with the immune system, with the skin/gut/respiratory barriers and microbiome involved; I think we now have a broader view of how big and complex the landscape really is,” he said.
The study received no outside funding. The researchers had no financial conflicts to disclose. Lio had no disclosures relevant to elimination diets but disclosed serving on the speakers bureau for AbbVie, Arcutis Biotherapeutics, Eli Lilly, Galderma, Hyphens Pharma, Incyte, La Roche–Posay/L’Oréal, Pfizer, Pierre Fabre Dermatologie, Regeneron/Sanofi Genzyme, and Verrica Pharmaceuticals; serving on consulting/advisory boards; or having stock options for many pharmaceutical companies. Lio also disclosed a patent pending for a Theraplex product with royalties paid and is a board member and Scientific Advisory Committee member emeritus of the National Eczema Association.
A version of this article first appeared on Medscape.com.
based on survey data from nearly 300 parents.
Although atopic dermatitis can be associated with an increased risk for food allergies, major allergy organizations do not currently recommend elimination diets as a treatment for atopic dermatitis, said Nadia Makkoukdji, MD, a pediatrician at Jackson Memorial Hospital, Miami, in a presentation at the American College of Allergy, Asthma, and Immunology (ACAAI) Annual Scientific Meeting.
“A fear of drastic dietary changes often prevents families from seeking the care their children need,” Makkoukdji said in an interview. In the clinical setting, Makkoukdji noted that she has seen many patients who have started food elimination diets on their own or as recommended by other doctors, and that these diets can lead to dangers such as the development of immunoglobulin E–mediated food allergies on reintroduction of eliminated foods and malnutrition. They can also produce “emotional stress in children and anxiety or depression, while also adding stress to parents and the entire family.”
Makkoukdji conducted the study to explore parents’ perceptions of these diets in management of their children’s atopic dermatitis, she said.
In the study, Makkoukdji and colleagues sought to understand parents’ perceptions of the role of diet in atopic dermatitis in their children. The researchers reviewed surveys from 298 parents of children with atopic dermatitis who were seen at a single academic center. Parents completed the surveys in the emergency department or in an allergy, dermatology, and general pediatrics clinic.
Overall, 42% of parents identified food triggers for their child’s atopic dermatitis. The most commonly identified triggers were milk (32%), tree nuts/seeds/peanuts (16%), and eggs (11%).
Of the parents who reported food triggers, 23% removed the suspected trigger food from the child’s diet completely, 20% removed suspected trigger foods from their own diets while breastfeeding, and 19% changed their infant’s formula.
In the wake of the elimination diets, 38% of the parents reported no improvement in their child’s atopic dermatitis, 35% reported a 25% improvement, and 9% reported complete resolution. The majority (79%) reintroduced eliminated foods and reported no recurrence of atopic dermatitis symptoms.
The researchers were surprised by how many parents changed their child’s diet in the belief that certain foods exacerbated their child’s atopic dermatitis, “although this perception aligns with the common concern that food allergens can trigger or worsen atopic dermatitis flares,” Makkoukdji said.
The current study highlights the need for more awareness of the limited impact of dietary modifications on atopic dermatitis in the absence of confirmed food allergies, Makkoukdji said. “Our study shows that food elimination diets are still commonly being used by parents in the local Miami population.”
The findings were limited by several factors, including the use of data from a single center and the focus only on pediatric patients, but the primary goal was to assess parental perceptions of AD flares in relation to dietary choices, said Makkoukdji. “Future studies that include larger and more diverse populations would be valuable for the field.”
Dietary Modifications Don’t Live Up to Hype
“Food continues to be one of the most discussed aspects of atopic dermatitis,” Peter Lio, MD, clinical assistant professor of dermatology and pediatrics at Northwestern University Feinberg School of Medicine, Chicago, Illinois, said in an interview.
“Almost all of my patients and families ask about dietary modifications, even though almost all of them have experimented with it to some degree,” said Lio. In his experience, diet plays a small role, if any, in the day-to-day management of atopic dermatitis.
This lack of effect of dietary changes is often frustrating to patients because of the persistent “common wisdom” that points to diet as a root cause of atopic dermatitis, Lio said. “Many practitioners continue to recommend excluding foods such as gluten or dairy from the diet, but generally these are only of modest help,” and although patients wish that dietary changes would fix the problem, most are left wondering why these changes didn’t help them.
The current study findings “reflect my own experience after nearly 20 years of being deeply immersed in the world of atopic dermatitis,” Lio said. Although the takeaway message does not argue against eating healthy foods, some foods do seem to make AD worse in some patients and may have nonallergic pro-inflammatory effects.
“In those cases, it is reasonable to limit or avoid those foods. However, it is extremely difficult to tell what food or foods are driving flare-ups when things are out of control, so dietary modification is generally not the best place to start,” he said.
True food allergies are much more common in patients with atopic dermatitis compared with individuals without atopic dermatitis, but the current study is not addressing these types of allergies, Lio emphasized. “If someone has true allergy to peanuts, for example, they should not be eating them; we also know that they are not ‘cheating’ because these patients would not merely have an eczema flare; they would have urticaria, angioedema, or anaphylaxis. There is tremendous confusion around this point and lots of confusion around allergy testing and its limitations.”
In addition, patients with atopic dermatitis are more likely than those without atopic dermatitis to have abnormalities in the gut microbiome and gut barrier, Lio said.
Abnormalities in the gut microbiome are different from the concept of allergy and may fall into the more complex category of barrier and microbiome disruptors, he said. Therefore, “the food category may not be nearly as important as the specific preparation of the food along with the additives (such as preservatives and emulsifiers) that may actually be driving the problem.”
Although in the past many clinicians advised patients to try cutting out certain foods to see whether atopic dermatitis symptoms improved, this strategy is not without risk, said Lio. “There have been incredible advancements in understanding the role of the gut in tolerization to foods.” Recent research has shown that by eating foods regularly, particularly those such as peanuts that seem to have more allergic potential, the body becomes tolerant, and this prevents the development of true food allergies.
As for additional research, many questions remain about the effects of types of foods, processing methods, and timing of introduction of foods on atopic dermatitis, Lio noted.
“Atopic dermatitis is a systemic condition with the immune system, with the skin/gut/respiratory barriers and microbiome involved; I think we now have a broader view of how big and complex the landscape really is,” he said.
The study received no outside funding. The researchers had no financial conflicts to disclose. Lio had no disclosures relevant to elimination diets but disclosed serving on the speakers bureau for AbbVie, Arcutis Biotherapeutics, Eli Lilly, Galderma, Hyphens Pharma, Incyte, La Roche–Posay/L’Oréal, Pfizer, Pierre Fabre Dermatologie, Regeneron/Sanofi Genzyme, and Verrica Pharmaceuticals; serving on consulting/advisory boards; or having stock options for many pharmaceutical companies. Lio also disclosed a patent pending for a Theraplex product with royalties paid and is a board member and Scientific Advisory Committee member emeritus of the National Eczema Association.
A version of this article first appeared on Medscape.com.
based on survey data from nearly 300 parents.
Although atopic dermatitis can be associated with an increased risk for food allergies, major allergy organizations do not currently recommend elimination diets as a treatment for atopic dermatitis, said Nadia Makkoukdji, MD, a pediatrician at Jackson Memorial Hospital, Miami, in a presentation at the American College of Allergy, Asthma, and Immunology (ACAAI) Annual Scientific Meeting.
“A fear of drastic dietary changes often prevents families from seeking the care their children need,” Makkoukdji said in an interview. In the clinical setting, Makkoukdji noted that she has seen many patients who have started food elimination diets on their own or as recommended by other doctors, and that these diets can lead to dangers such as the development of immunoglobulin E–mediated food allergies on reintroduction of eliminated foods and malnutrition. They can also produce “emotional stress in children and anxiety or depression, while also adding stress to parents and the entire family.”
Makkoukdji conducted the study to explore parents’ perceptions of these diets in management of their children’s atopic dermatitis, she said.
In the study, Makkoukdji and colleagues sought to understand parents’ perceptions of the role of diet in atopic dermatitis in their children. The researchers reviewed surveys from 298 parents of children with atopic dermatitis who were seen at a single academic center. Parents completed the surveys in the emergency department or in an allergy, dermatology, and general pediatrics clinic.
Overall, 42% of parents identified food triggers for their child’s atopic dermatitis. The most commonly identified triggers were milk (32%), tree nuts/seeds/peanuts (16%), and eggs (11%).
Of the parents who reported food triggers, 23% removed the suspected trigger food from the child’s diet completely, 20% removed suspected trigger foods from their own diets while breastfeeding, and 19% changed their infant’s formula.
In the wake of the elimination diets, 38% of the parents reported no improvement in their child’s atopic dermatitis, 35% reported a 25% improvement, and 9% reported complete resolution. The majority (79%) reintroduced eliminated foods and reported no recurrence of atopic dermatitis symptoms.
The researchers were surprised by how many parents changed their child’s diet in the belief that certain foods exacerbated their child’s atopic dermatitis, “although this perception aligns with the common concern that food allergens can trigger or worsen atopic dermatitis flares,” Makkoukdji said.
The current study highlights the need for more awareness of the limited impact of dietary modifications on atopic dermatitis in the absence of confirmed food allergies, Makkoukdji said. “Our study shows that food elimination diets are still commonly being used by parents in the local Miami population.”
The findings were limited by several factors, including the use of data from a single center and the focus only on pediatric patients, but the primary goal was to assess parental perceptions of AD flares in relation to dietary choices, said Makkoukdji. “Future studies that include larger and more diverse populations would be valuable for the field.”
Dietary Modifications Don’t Live Up to Hype
“Food continues to be one of the most discussed aspects of atopic dermatitis,” Peter Lio, MD, clinical assistant professor of dermatology and pediatrics at Northwestern University Feinberg School of Medicine, Chicago, Illinois, said in an interview.
“Almost all of my patients and families ask about dietary modifications, even though almost all of them have experimented with it to some degree,” said Lio. In his experience, diet plays a small role, if any, in the day-to-day management of atopic dermatitis.
This lack of effect of dietary changes is often frustrating to patients because of the persistent “common wisdom” that points to diet as a root cause of atopic dermatitis, Lio said. “Many practitioners continue to recommend excluding foods such as gluten or dairy from the diet, but generally these are only of modest help,” and although patients wish that dietary changes would fix the problem, most are left wondering why these changes didn’t help them.
The current study findings “reflect my own experience after nearly 20 years of being deeply immersed in the world of atopic dermatitis,” Lio said. Although the takeaway message does not argue against eating healthy foods, some foods do seem to make AD worse in some patients and may have nonallergic pro-inflammatory effects.
“In those cases, it is reasonable to limit or avoid those foods. However, it is extremely difficult to tell what food or foods are driving flare-ups when things are out of control, so dietary modification is generally not the best place to start,” he said.
True food allergies are much more common in patients with atopic dermatitis compared with individuals without atopic dermatitis, but the current study is not addressing these types of allergies, Lio emphasized. “If someone has true allergy to peanuts, for example, they should not be eating them; we also know that they are not ‘cheating’ because these patients would not merely have an eczema flare; they would have urticaria, angioedema, or anaphylaxis. There is tremendous confusion around this point and lots of confusion around allergy testing and its limitations.”
In addition, patients with atopic dermatitis are more likely than those without atopic dermatitis to have abnormalities in the gut microbiome and gut barrier, Lio said.
Abnormalities in the gut microbiome are different from the concept of allergy and may fall into the more complex category of barrier and microbiome disruptors, he said. Therefore, “the food category may not be nearly as important as the specific preparation of the food along with the additives (such as preservatives and emulsifiers) that may actually be driving the problem.”
Although in the past many clinicians advised patients to try cutting out certain foods to see whether atopic dermatitis symptoms improved, this strategy is not without risk, said Lio. “There have been incredible advancements in understanding the role of the gut in tolerization to foods.” Recent research has shown that by eating foods regularly, particularly those such as peanuts that seem to have more allergic potential, the body becomes tolerant, and this prevents the development of true food allergies.
As for additional research, many questions remain about the effects of types of foods, processing methods, and timing of introduction of foods on atopic dermatitis, Lio noted.
“Atopic dermatitis is a systemic condition with the immune system, with the skin/gut/respiratory barriers and microbiome involved; I think we now have a broader view of how big and complex the landscape really is,” he said.
The study received no outside funding. The researchers had no financial conflicts to disclose. Lio had no disclosures relevant to elimination diets but disclosed serving on the speakers bureau for AbbVie, Arcutis Biotherapeutics, Eli Lilly, Galderma, Hyphens Pharma, Incyte, La Roche–Posay/L’Oréal, Pfizer, Pierre Fabre Dermatologie, Regeneron/Sanofi Genzyme, and Verrica Pharmaceuticals; serving on consulting/advisory boards; or having stock options for many pharmaceutical companies. Lio also disclosed a patent pending for a Theraplex product with royalties paid and is a board member and Scientific Advisory Committee member emeritus of the National Eczema Association.
A version of this article first appeared on Medscape.com.
FROM ACAAI 2024
Atopic Dermatitis: Upadacitinib Effectiveness Maintained Through 76 weeks Among Adolescents
TOPLINE:
across three phase 3 trials.
METHODOLOGY:
- Researchers conducted three double-blind, placebo-controlled phase 3 randomized clinical trials (Measure Up 1, Measure Up 2, and AD Up) involving 542 adolescents aged 12-17 years with moderate to severe AD.
- Participants were randomized to receive the oral Janus kinase inhibitor upadacitinib (15 mg or 30 mg once daily) or placebo, with or without topical corticosteroids, for 16 weeks, followed by rerandomization of patients in the placebo group to upadacitinib for up to 76 weeks.
- Study endpoints were at least a 75%, 90%, or 100% reduction in the Eczema Area and Severity Index (EASI-75, EASI-90, and EASI-100, respectively), Validated Investigator Global Assessment for AD (vIGA-AD) score of 0 or 1, and a ≥ 4-point improvement in the Worst Pruritus Numerical Rating Scale (WP-NRS).
- Adverse events were monitored, including serious infections, herpes zoster, and creatine kinase elevation.
TAKEAWAY:
- Among those who continued treatment on upadacitinib, 15 mg and 30 mg, EASI-75 response rates were maintained or improved through week 76 in all three studies. Patients who switched from placebo to upadacitinib also experienced improvements in EASI-75 through week 76.
- The proportion of patients who achieved EASI-90 and EASI-100 responses increased, and in general, were maintained from week 16 through week 76 in all three studies; the proportion was numerically higher among patients on 30 mg for all three studies.
- The proportion of adolescents achieving vIGA-AD score of 0 or 1 and WP-NRS improvement of ≥ 4 points was sustained or improved through 76 weeks.
- Serious infections were reported in five patients or fewer in each treatment group for all three studies. All opportunistic infections were eczema herpeticum; most cases were not serious, or were mild or moderate, and in general, did not require stopping treatment.
IN PRACTICE:
“These results through 76 weeks demonstrated that upadacitinib, with a favorable benefit-risk profile, was an effective long-term treatment option for adolescents with moderate to severe AD,” the authors wrote.
SOURCE:
The study was led by Amy S. Paller, MD, professor and chair of dermatology, Northwestern University, Chicago, and was published online on October 23 in JAMA Dermatology.
LIMITATIONS:
The study limitations included a small sample size, and the findings did not extend to patients under 12 years or those weighing < 40 kg.
DISCLOSURES:
This study was supported by AbbVie. Paller received grants and personal fees from pharmaceutical companies including AbbVie during the conduct of the study. Several authors reported financial ties with various sources, including AbbVie.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
across three phase 3 trials.
METHODOLOGY:
- Researchers conducted three double-blind, placebo-controlled phase 3 randomized clinical trials (Measure Up 1, Measure Up 2, and AD Up) involving 542 adolescents aged 12-17 years with moderate to severe AD.
- Participants were randomized to receive the oral Janus kinase inhibitor upadacitinib (15 mg or 30 mg once daily) or placebo, with or without topical corticosteroids, for 16 weeks, followed by rerandomization of patients in the placebo group to upadacitinib for up to 76 weeks.
- Study endpoints were at least a 75%, 90%, or 100% reduction in the Eczema Area and Severity Index (EASI-75, EASI-90, and EASI-100, respectively), Validated Investigator Global Assessment for AD (vIGA-AD) score of 0 or 1, and a ≥ 4-point improvement in the Worst Pruritus Numerical Rating Scale (WP-NRS).
- Adverse events were monitored, including serious infections, herpes zoster, and creatine kinase elevation.
TAKEAWAY:
- Among those who continued treatment on upadacitinib, 15 mg and 30 mg, EASI-75 response rates were maintained or improved through week 76 in all three studies. Patients who switched from placebo to upadacitinib also experienced improvements in EASI-75 through week 76.
- The proportion of patients who achieved EASI-90 and EASI-100 responses increased, and in general, were maintained from week 16 through week 76 in all three studies; the proportion was numerically higher among patients on 30 mg for all three studies.
- The proportion of adolescents achieving vIGA-AD score of 0 or 1 and WP-NRS improvement of ≥ 4 points was sustained or improved through 76 weeks.
- Serious infections were reported in five patients or fewer in each treatment group for all three studies. All opportunistic infections were eczema herpeticum; most cases were not serious, or were mild or moderate, and in general, did not require stopping treatment.
IN PRACTICE:
“These results through 76 weeks demonstrated that upadacitinib, with a favorable benefit-risk profile, was an effective long-term treatment option for adolescents with moderate to severe AD,” the authors wrote.
SOURCE:
The study was led by Amy S. Paller, MD, professor and chair of dermatology, Northwestern University, Chicago, and was published online on October 23 in JAMA Dermatology.
LIMITATIONS:
The study limitations included a small sample size, and the findings did not extend to patients under 12 years or those weighing < 40 kg.
DISCLOSURES:
This study was supported by AbbVie. Paller received grants and personal fees from pharmaceutical companies including AbbVie during the conduct of the study. Several authors reported financial ties with various sources, including AbbVie.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
across three phase 3 trials.
METHODOLOGY:
- Researchers conducted three double-blind, placebo-controlled phase 3 randomized clinical trials (Measure Up 1, Measure Up 2, and AD Up) involving 542 adolescents aged 12-17 years with moderate to severe AD.
- Participants were randomized to receive the oral Janus kinase inhibitor upadacitinib (15 mg or 30 mg once daily) or placebo, with or without topical corticosteroids, for 16 weeks, followed by rerandomization of patients in the placebo group to upadacitinib for up to 76 weeks.
- Study endpoints were at least a 75%, 90%, or 100% reduction in the Eczema Area and Severity Index (EASI-75, EASI-90, and EASI-100, respectively), Validated Investigator Global Assessment for AD (vIGA-AD) score of 0 or 1, and a ≥ 4-point improvement in the Worst Pruritus Numerical Rating Scale (WP-NRS).
- Adverse events were monitored, including serious infections, herpes zoster, and creatine kinase elevation.
TAKEAWAY:
- Among those who continued treatment on upadacitinib, 15 mg and 30 mg, EASI-75 response rates were maintained or improved through week 76 in all three studies. Patients who switched from placebo to upadacitinib also experienced improvements in EASI-75 through week 76.
- The proportion of patients who achieved EASI-90 and EASI-100 responses increased, and in general, were maintained from week 16 through week 76 in all three studies; the proportion was numerically higher among patients on 30 mg for all three studies.
- The proportion of adolescents achieving vIGA-AD score of 0 or 1 and WP-NRS improvement of ≥ 4 points was sustained or improved through 76 weeks.
- Serious infections were reported in five patients or fewer in each treatment group for all three studies. All opportunistic infections were eczema herpeticum; most cases were not serious, or were mild or moderate, and in general, did not require stopping treatment.
IN PRACTICE:
“These results through 76 weeks demonstrated that upadacitinib, with a favorable benefit-risk profile, was an effective long-term treatment option for adolescents with moderate to severe AD,” the authors wrote.
SOURCE:
The study was led by Amy S. Paller, MD, professor and chair of dermatology, Northwestern University, Chicago, and was published online on October 23 in JAMA Dermatology.
LIMITATIONS:
The study limitations included a small sample size, and the findings did not extend to patients under 12 years or those weighing < 40 kg.
DISCLOSURES:
This study was supported by AbbVie. Paller received grants and personal fees from pharmaceutical companies including AbbVie during the conduct of the study. Several authors reported financial ties with various sources, including AbbVie.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Phenytoin-Induced DRESS Syndrome: Clinical and Laboratory Characteristics
To the Editor:
Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome—a severe cutaneous adverse drug reaction—is characterized by a cutaneous rash and systemic upset in the form of various internal organ and hematologic disturbances. This delayed and idiosyncratic syndrome went by several names, including anticonvulsant hypersensitivity syndrome, before Bocquet et al1 proposed the term DRESS syndrome.
Phenytoin, a hydantoin derivative used in neurology, was implicated in 41% of cases of DRESS syndrome in a study of 100 patients conducted in southern India.2,3 While DRESS syndrome is a newer name, the clinical picture of DRESS secondary to phenytoin use remains similar in that it manifests with a morbilliform rash and systemic upset. We sought to describe the clinical and laboratory characteristics of phenytoin-induced DRESS syndrome in this case series.
The analysis included 23 patients with DRESS syndrome secondary to phenytoin use who presented to a tertiary care institution in North India between July 2021 and December 2022, satisfied the European Registry of Severe Cutaneous Adverse Reaction (RegiSCAR) criteria,4 and achieved a DRESS diagnostic score of more than 1. The mean age of the patients was 44 years (range, 14–74 years). There was a slight female predominance with a male to female ratio of 0.9:1. More than half of the patients (52.2% [12/23]) presented directly to the dermatology outpatient department; the remaining patients were referred from other departments (47.8% [11/23]). Patients primarily were receiving phenytoin for neurologic indications. Specific reasons included antiseizure prophylaxis following a traffic accident (34.8% [8/23]); epilepsy (26.1% [6/23]); and neoplastic (17.4% [4/23]), vascular (17.4% [4/23]), and infectious (4.3% [1/23]) causes. The mean latency period from drug intake to symptom onset was 29 days (range, 6–62 days), and the mean illness duration was 9 days (range, 1–45 days).
The majority of patients experienced pruritus (91.3% [21/23]) and fever (74.0% [17/23]), and all initially had a rash. Maculopapular morphology was seen in all patients. Erythema multiforme–like (17.4% [4/23]), erythrodermic (17.4% [4/23]), and vesicular (13.0% [3/23]) rashes also were documented (Figure 1). The trunk (100% [23/23]) and extremities (95.7% [22/23]) were involved most often, followed by the palms and soles (56.5% [13/23]). The mean total body surface area affected was 73.65%. Only 7 patients (30.4%) had mucosal involvement; nonhemorrhagic cheilitis was the most common manifestation.
Facial edema, a hallmark feature of DRESS syndrome, was noted in 69.6% (16/23) of patients (Figure 2). Lymphadenopathy was present in 43.5% (10/23) of patients; of those cases, the inguinal (40.0%; n=4) and cervical (30%; n=3) nodes most commonly were involved. Although DRESS syndrome can affect internal organs, this was an issue for only 2 (8.7%) patients who experienced mild hepatomegaly.
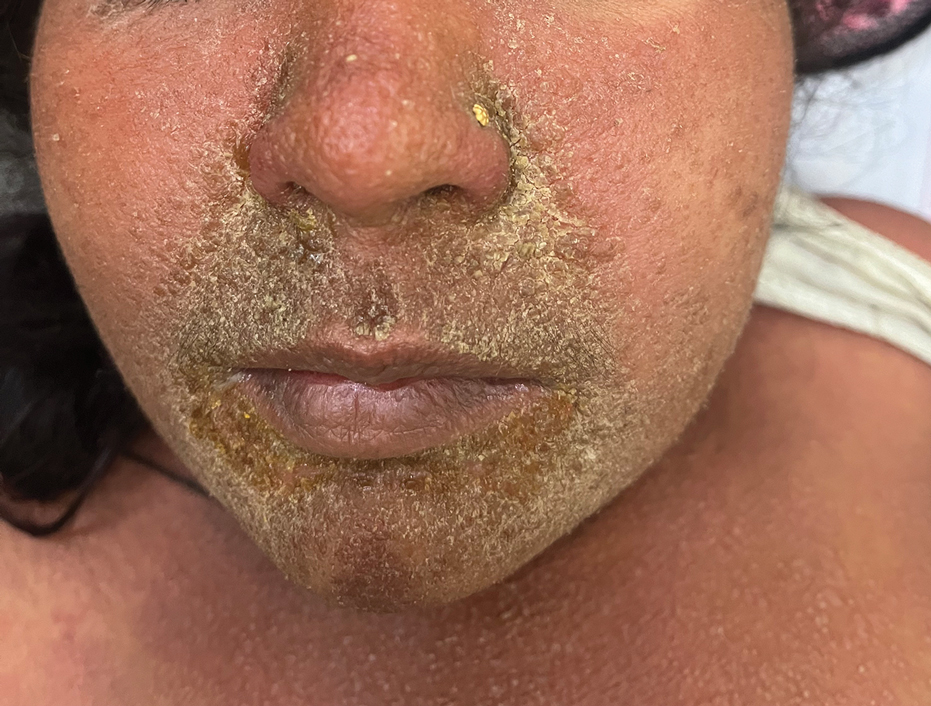
Laboratory investigations revealed a mean differential eosinophil percentage of 10.3% (reference range, 1%–4%), while the mean absolute eosinophil count was 1.0634×109/L (reference range, 0.02–0.5×109/L). Other hematologic findings included the mean percentages of neutrophils (60%; reference range, 50%–60%), lymphocytes (19.95%; reference range, 20%–50%), and monocytes (8.70%; reference range, 2%–8%).
Liver function tests revealed transaminitis5 as the most common finding, with mean aspartate aminotransferase levels of 109 U/L (reference range, 8–33 U/L), mean alanine aminotransferase of 97.9 U/L (reference range, 7–56 U/L), and mean alkaline phosphatase levels of 211.35 U/L (reference range, 44–147 U/L). Half of the patients had notable (>2 times the upper limit of normal) transaminitis.
Renal blood workup revealed slightly elevated blood urea nitrogen levels with a mean value of 28.4 mg/dL (reference range, 6–24 mg/dL), and mean serum creatinine was 0.78 mg/dL (reference range for men, 0.7–1.3 mg/dL; for women, 0.6–1.1 mg/dL).
All patients were treated with oral steroids (prednisolone 1 mg/kg/d) before tapering slowly over the following 6 to 8 weeks. The culprit drug (phenytoin) was stopped on the day of presentation. Resolution of rash and itching was seen in all patients by 3 weeks after presentation without any relapse by follow-up at 6 weeks from presentation to the hospital.
Our case series seeks to discuss the clinical and laboratory features of phenytoin-induced DRESS syndrome. Our patients had more erythrodermic and erythema multiforme–like morphologies, less mucosal involvement, more hepatic involvement, and earlier resolution.
- Bocquet H, Bagot M, Roujeau JC. Drug-induced pseudolymphoma and drug hypersensitivity syndrome (drug rash with eosinophilia and systemic symptoms: DRESS). Semin Cutan Med Surg. 1996;15:250-257. doi:10.1016/s1085-5629(96)80038-1
- Patocka J, Wu Q, Nepovimova E, et al. Phenytoin—an anti-seizure drug: overview of its chemistry, pharmacology and toxicology. Food Chem Toxicol. 2020;142:111393. doi:10.1016/j.fct.2020.111393
- Sasidharanpillai S, Chathoth AT, Khader A, et al. Predictors of disease severity in drug reaction with eosinophilia and systemic symptoms. Indian J Dermatol Venereol Leprol. 2019;85:266-275. doi:10.4103/ijdvl.IJDVL_482_17
- Kardaun SH, Sekula P, Valeyrie-Allanore L, et al. Drug reaction with eosinophilia and systemic symptoms (DRESS): an original multisystem adverse drug reaction. Results from the prospective RegiSCAR study. Brit J Dermatol. 2013;169:1071-1080.
- Morán-Mariños C, Alva-Diaz C, De la Cruz Ramirez W, et al. Drug reaction with eosinophilia and systemic symptoms (DRESS) induced by phenytoin re-exposure: case report and systematic review. Acta Clin Belg. 2022;77:177-185. doi:10.1080/17843286.2020.1767459
To the Editor:
Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome—a severe cutaneous adverse drug reaction—is characterized by a cutaneous rash and systemic upset in the form of various internal organ and hematologic disturbances. This delayed and idiosyncratic syndrome went by several names, including anticonvulsant hypersensitivity syndrome, before Bocquet et al1 proposed the term DRESS syndrome.
Phenytoin, a hydantoin derivative used in neurology, was implicated in 41% of cases of DRESS syndrome in a study of 100 patients conducted in southern India.2,3 While DRESS syndrome is a newer name, the clinical picture of DRESS secondary to phenytoin use remains similar in that it manifests with a morbilliform rash and systemic upset. We sought to describe the clinical and laboratory characteristics of phenytoin-induced DRESS syndrome in this case series.
The analysis included 23 patients with DRESS syndrome secondary to phenytoin use who presented to a tertiary care institution in North India between July 2021 and December 2022, satisfied the European Registry of Severe Cutaneous Adverse Reaction (RegiSCAR) criteria,4 and achieved a DRESS diagnostic score of more than 1. The mean age of the patients was 44 years (range, 14–74 years). There was a slight female predominance with a male to female ratio of 0.9:1. More than half of the patients (52.2% [12/23]) presented directly to the dermatology outpatient department; the remaining patients were referred from other departments (47.8% [11/23]). Patients primarily were receiving phenytoin for neurologic indications. Specific reasons included antiseizure prophylaxis following a traffic accident (34.8% [8/23]); epilepsy (26.1% [6/23]); and neoplastic (17.4% [4/23]), vascular (17.4% [4/23]), and infectious (4.3% [1/23]) causes. The mean latency period from drug intake to symptom onset was 29 days (range, 6–62 days), and the mean illness duration was 9 days (range, 1–45 days).
The majority of patients experienced pruritus (91.3% [21/23]) and fever (74.0% [17/23]), and all initially had a rash. Maculopapular morphology was seen in all patients. Erythema multiforme–like (17.4% [4/23]), erythrodermic (17.4% [4/23]), and vesicular (13.0% [3/23]) rashes also were documented (Figure 1). The trunk (100% [23/23]) and extremities (95.7% [22/23]) were involved most often, followed by the palms and soles (56.5% [13/23]). The mean total body surface area affected was 73.65%. Only 7 patients (30.4%) had mucosal involvement; nonhemorrhagic cheilitis was the most common manifestation.

Facial edema, a hallmark feature of DRESS syndrome, was noted in 69.6% (16/23) of patients (Figure 2). Lymphadenopathy was present in 43.5% (10/23) of patients; of those cases, the inguinal (40.0%; n=4) and cervical (30%; n=3) nodes most commonly were involved. Although DRESS syndrome can affect internal organs, this was an issue for only 2 (8.7%) patients who experienced mild hepatomegaly.

Laboratory investigations revealed a mean differential eosinophil percentage of 10.3% (reference range, 1%–4%), while the mean absolute eosinophil count was 1.0634×109/L (reference range, 0.02–0.5×109/L). Other hematologic findings included the mean percentages of neutrophils (60%; reference range, 50%–60%), lymphocytes (19.95%; reference range, 20%–50%), and monocytes (8.70%; reference range, 2%–8%).
Liver function tests revealed transaminitis5 as the most common finding, with mean aspartate aminotransferase levels of 109 U/L (reference range, 8–33 U/L), mean alanine aminotransferase of 97.9 U/L (reference range, 7–56 U/L), and mean alkaline phosphatase levels of 211.35 U/L (reference range, 44–147 U/L). Half of the patients had notable (>2 times the upper limit of normal) transaminitis.
Renal blood workup revealed slightly elevated blood urea nitrogen levels with a mean value of 28.4 mg/dL (reference range, 6–24 mg/dL), and mean serum creatinine was 0.78 mg/dL (reference range for men, 0.7–1.3 mg/dL; for women, 0.6–1.1 mg/dL).
All patients were treated with oral steroids (prednisolone 1 mg/kg/d) before tapering slowly over the following 6 to 8 weeks. The culprit drug (phenytoin) was stopped on the day of presentation. Resolution of rash and itching was seen in all patients by 3 weeks after presentation without any relapse by follow-up at 6 weeks from presentation to the hospital.
Our case series seeks to discuss the clinical and laboratory features of phenytoin-induced DRESS syndrome. Our patients had more erythrodermic and erythema multiforme–like morphologies, less mucosal involvement, more hepatic involvement, and earlier resolution.
To the Editor:
Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome—a severe cutaneous adverse drug reaction—is characterized by a cutaneous rash and systemic upset in the form of various internal organ and hematologic disturbances. This delayed and idiosyncratic syndrome went by several names, including anticonvulsant hypersensitivity syndrome, before Bocquet et al1 proposed the term DRESS syndrome.
Phenytoin, a hydantoin derivative used in neurology, was implicated in 41% of cases of DRESS syndrome in a study of 100 patients conducted in southern India.2,3 While DRESS syndrome is a newer name, the clinical picture of DRESS secondary to phenytoin use remains similar in that it manifests with a morbilliform rash and systemic upset. We sought to describe the clinical and laboratory characteristics of phenytoin-induced DRESS syndrome in this case series.
The analysis included 23 patients with DRESS syndrome secondary to phenytoin use who presented to a tertiary care institution in North India between July 2021 and December 2022, satisfied the European Registry of Severe Cutaneous Adverse Reaction (RegiSCAR) criteria,4 and achieved a DRESS diagnostic score of more than 1. The mean age of the patients was 44 years (range, 14–74 years). There was a slight female predominance with a male to female ratio of 0.9:1. More than half of the patients (52.2% [12/23]) presented directly to the dermatology outpatient department; the remaining patients were referred from other departments (47.8% [11/23]). Patients primarily were receiving phenytoin for neurologic indications. Specific reasons included antiseizure prophylaxis following a traffic accident (34.8% [8/23]); epilepsy (26.1% [6/23]); and neoplastic (17.4% [4/23]), vascular (17.4% [4/23]), and infectious (4.3% [1/23]) causes. The mean latency period from drug intake to symptom onset was 29 days (range, 6–62 days), and the mean illness duration was 9 days (range, 1–45 days).
The majority of patients experienced pruritus (91.3% [21/23]) and fever (74.0% [17/23]), and all initially had a rash. Maculopapular morphology was seen in all patients. Erythema multiforme–like (17.4% [4/23]), erythrodermic (17.4% [4/23]), and vesicular (13.0% [3/23]) rashes also were documented (Figure 1). The trunk (100% [23/23]) and extremities (95.7% [22/23]) were involved most often, followed by the palms and soles (56.5% [13/23]). The mean total body surface area affected was 73.65%. Only 7 patients (30.4%) had mucosal involvement; nonhemorrhagic cheilitis was the most common manifestation.

Facial edema, a hallmark feature of DRESS syndrome, was noted in 69.6% (16/23) of patients (Figure 2). Lymphadenopathy was present in 43.5% (10/23) of patients; of those cases, the inguinal (40.0%; n=4) and cervical (30%; n=3) nodes most commonly were involved. Although DRESS syndrome can affect internal organs, this was an issue for only 2 (8.7%) patients who experienced mild hepatomegaly.

Laboratory investigations revealed a mean differential eosinophil percentage of 10.3% (reference range, 1%–4%), while the mean absolute eosinophil count was 1.0634×109/L (reference range, 0.02–0.5×109/L). Other hematologic findings included the mean percentages of neutrophils (60%; reference range, 50%–60%), lymphocytes (19.95%; reference range, 20%–50%), and monocytes (8.70%; reference range, 2%–8%).
Liver function tests revealed transaminitis5 as the most common finding, with mean aspartate aminotransferase levels of 109 U/L (reference range, 8–33 U/L), mean alanine aminotransferase of 97.9 U/L (reference range, 7–56 U/L), and mean alkaline phosphatase levels of 211.35 U/L (reference range, 44–147 U/L). Half of the patients had notable (>2 times the upper limit of normal) transaminitis.
Renal blood workup revealed slightly elevated blood urea nitrogen levels with a mean value of 28.4 mg/dL (reference range, 6–24 mg/dL), and mean serum creatinine was 0.78 mg/dL (reference range for men, 0.7–1.3 mg/dL; for women, 0.6–1.1 mg/dL).
All patients were treated with oral steroids (prednisolone 1 mg/kg/d) before tapering slowly over the following 6 to 8 weeks. The culprit drug (phenytoin) was stopped on the day of presentation. Resolution of rash and itching was seen in all patients by 3 weeks after presentation without any relapse by follow-up at 6 weeks from presentation to the hospital.
Our case series seeks to discuss the clinical and laboratory features of phenytoin-induced DRESS syndrome. Our patients had more erythrodermic and erythema multiforme–like morphologies, less mucosal involvement, more hepatic involvement, and earlier resolution.
- Bocquet H, Bagot M, Roujeau JC. Drug-induced pseudolymphoma and drug hypersensitivity syndrome (drug rash with eosinophilia and systemic symptoms: DRESS). Semin Cutan Med Surg. 1996;15:250-257. doi:10.1016/s1085-5629(96)80038-1
- Patocka J, Wu Q, Nepovimova E, et al. Phenytoin—an anti-seizure drug: overview of its chemistry, pharmacology and toxicology. Food Chem Toxicol. 2020;142:111393. doi:10.1016/j.fct.2020.111393
- Sasidharanpillai S, Chathoth AT, Khader A, et al. Predictors of disease severity in drug reaction with eosinophilia and systemic symptoms. Indian J Dermatol Venereol Leprol. 2019;85:266-275. doi:10.4103/ijdvl.IJDVL_482_17
- Kardaun SH, Sekula P, Valeyrie-Allanore L, et al. Drug reaction with eosinophilia and systemic symptoms (DRESS): an original multisystem adverse drug reaction. Results from the prospective RegiSCAR study. Brit J Dermatol. 2013;169:1071-1080.
- Morán-Mariños C, Alva-Diaz C, De la Cruz Ramirez W, et al. Drug reaction with eosinophilia and systemic symptoms (DRESS) induced by phenytoin re-exposure: case report and systematic review. Acta Clin Belg. 2022;77:177-185. doi:10.1080/17843286.2020.1767459
- Bocquet H, Bagot M, Roujeau JC. Drug-induced pseudolymphoma and drug hypersensitivity syndrome (drug rash with eosinophilia and systemic symptoms: DRESS). Semin Cutan Med Surg. 1996;15:250-257. doi:10.1016/s1085-5629(96)80038-1
- Patocka J, Wu Q, Nepovimova E, et al. Phenytoin—an anti-seizure drug: overview of its chemistry, pharmacology and toxicology. Food Chem Toxicol. 2020;142:111393. doi:10.1016/j.fct.2020.111393
- Sasidharanpillai S, Chathoth AT, Khader A, et al. Predictors of disease severity in drug reaction with eosinophilia and systemic symptoms. Indian J Dermatol Venereol Leprol. 2019;85:266-275. doi:10.4103/ijdvl.IJDVL_482_17
- Kardaun SH, Sekula P, Valeyrie-Allanore L, et al. Drug reaction with eosinophilia and systemic symptoms (DRESS): an original multisystem adverse drug reaction. Results from the prospective RegiSCAR study. Brit J Dermatol. 2013;169:1071-1080.
- Morán-Mariños C, Alva-Diaz C, De la Cruz Ramirez W, et al. Drug reaction with eosinophilia and systemic symptoms (DRESS) induced by phenytoin re-exposure: case report and systematic review. Acta Clin Belg. 2022;77:177-185. doi:10.1080/17843286.2020.1767459
Practice Points
- Phenytoin has been implicated in drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome, and common symptoms include rash, pruritus, and fever.
- Transaminitis may occur in patients with DRESS syndrome secondary to phenytoin use.
Over 3 Years, Atopic Dermatitis Well-Controlled with Lebrikizumab
AMSTERDAM — among those followed up for an additional 2 years, according to the latest data from an extension study.
At the end of the maintenance phase of the pivotal trials at 12 months, 84% of the patients enrolled into the extension had clear or almost clear skin, as per the Investigator Global Assessment (IGA). This overall figure as well as the proportion with even better responses have persisted unchanged, reported Diamant Thaçi, MD, PhD, professor and head of the Comprehensive Center for Inflammatory Medicine, University of Lübeck in Germany.
Responses at 3 Years Maintained
“This is really quite remarkable,” Dr. Thaçi said. “Roughly all the patients maintained their response.” These results became even more remarkable when patients were assessed for their use of adjunctive therapy to control flares.
“Over the whole follow-up, 90% had no need for topical corticosteroids or any other rescue therapy,” Dr. Thaçi reported, providing data from the ADjoin lebrikizumab extension study during a late-breaking news session at the annual meeting of the European Academy of Dermatology and Venereology.
The patients in ADjoin were enrolled from the pivotal phase 3 ADvocate 1 and 2 trials completed almost 2 years ago and published together in March 2023. Lebrikizumab was approved in the United States in September 2024 for moderate to severe AD in patients aged ≥ 12 years, following previous approvals in Europe in 2023 and in Japan in January 2024.
In these two identical trials with a total of 564 patients, the primary endpoint was an IGA of 0 or 1, signifying clear or almost clear skin. At nearly 40%, the proportion of patients reaching this outcome at 16 weeks was about threefold greater (P < .001) on lebrikizumab than on placebo. The benefit was similar on secondary endpoints, such as 75% improvement in the Eczema Area and Severity Index (EASI75) score.
At the end of the double-blind, placebo-controlled 16-week phase of the ADvocate 1 and 2 trials, which enrolled adults and adolescents aged ≥ 12 years, responders were enrolled into a maintenance phase in which they were rerandomized to 250 mg lebrikizumab every 2 weeks (Q2W) or every 4 weeks (Q4W). The latter is the approved maintenance dose.
At the end of the maintenance phase, which lasted another 32 weeks (total exposure of 52 weeks for those initially randomized to lebrikizumab), patients were invited into the ADjoin extension. The only exclusions from the extension were serious adverse events related to lebrikizumab and noncompliance.
Response Curves Appear as Straight Lines
Over the next 2 years of ADjoin, response curves appeared as straight lines not only for the overall response but when patients were stratified for different levels of response at the extension study entry. Specifically, 81.5% and 83.3% had an IGA score of 0 or 1 in the Q2W and Q4W arms at completion of the ADvocate 16-week double-blind phase. At 3 years, the rates were 84.0% and 82.9%, respectively.
For the subgroup who entered ADjoin with an EASI75 or an EASI90 response, the persistence of this level of response over 2 years was similar, although there was some gain observed among those who entered the trial with an EASI75 response.
“Not only did these patients maintain their response, but the response on average slowly improved, so that there were more patients with an EASI90 response at the 3-year timepoint,” Dr. Thaçi said.
Of the 181 patients in the ADjoin extension, 82 patients were maintained on Q2W dosing and 99 were maintained on Q4W lebrikizumab. Their mean age was about 35 years, more than half were women, and nearly 40% had severe AD at the time they enrolled in the ADvocate trials. There was essentially no difference in response rates among those in the Q2W and Q4W arms over time in ADjoin.
Side Effect Profile Essentially Unchanged
The side effect and tolerability profiles, which were favorable in the original 16-week placebo-controlled study, have remained unchanged over the subsequent maintenance phase and through the additional 2 years of the ADjoin extension.
“There continued to be reports of conjunctivitis, which is very specific for anti–IL-13 therapies,” Dr. Thaçi said. However, he said that the incidence did not increase over time, and because it was easy to treat, “most patients do not discontinue lebrikizumab for this reason.” Moreover, he said the impression was that “the number of patients experiencing adverse effects has been decreasing over time.”
Calling these long-term results “very exciting,” Dr. Thaçi called lebrikizumab “a very valuable option for long-term AD care.”
Asked for his perspective on the results, Jonathan I. Silverberg, MD, PhD, Director of Clinical Research, Department of Dermatology, George Washington University, Washington, DC, said that it is important to study long-term efficacy, and these results are positive. Without direct comparisons to other biologics available for AD, nothing can be implied about the relative efficacy of monoclonal antibodies approved for AD.
“These data are important both from an efficacy and safety perspective” for those advising patients who need chronic AD treatment, said Dr. Silverberg, who was the principal investigator of the ADvocate trials.
Earlier this year, 5-year follow-up data were published for dupilumab. Of 326 patients who remained on therapy this long, 220 (67%) maintained an IGA of 0 or 1 at the end of the study. There were no unexpected adverse events, which were generally stable or declined throughout the study.
Dr. Thaçi has financial relationships with AbbVie, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Celltrion, Galderma, Leo Pharma, L’Oreal, Janssen-Cilag, New Bridge, Novartis, Pfizer, Regeneron, Roche, Sanofi, Sun Pharma, UCB, and Vichy. Dr. Silverberg reported financial relationships with more than 40 pharmaceutical companies including those that make drugs for AD.
A version of this article appeared on Medscape.com.
AMSTERDAM — among those followed up for an additional 2 years, according to the latest data from an extension study.
At the end of the maintenance phase of the pivotal trials at 12 months, 84% of the patients enrolled into the extension had clear or almost clear skin, as per the Investigator Global Assessment (IGA). This overall figure as well as the proportion with even better responses have persisted unchanged, reported Diamant Thaçi, MD, PhD, professor and head of the Comprehensive Center for Inflammatory Medicine, University of Lübeck in Germany.
Responses at 3 Years Maintained
“This is really quite remarkable,” Dr. Thaçi said. “Roughly all the patients maintained their response.” These results became even more remarkable when patients were assessed for their use of adjunctive therapy to control flares.
“Over the whole follow-up, 90% had no need for topical corticosteroids or any other rescue therapy,” Dr. Thaçi reported, providing data from the ADjoin lebrikizumab extension study during a late-breaking news session at the annual meeting of the European Academy of Dermatology and Venereology.
The patients in ADjoin were enrolled from the pivotal phase 3 ADvocate 1 and 2 trials completed almost 2 years ago and published together in March 2023. Lebrikizumab was approved in the United States in September 2024 for moderate to severe AD in patients aged ≥ 12 years, following previous approvals in Europe in 2023 and in Japan in January 2024.
In these two identical trials with a total of 564 patients, the primary endpoint was an IGA of 0 or 1, signifying clear or almost clear skin. At nearly 40%, the proportion of patients reaching this outcome at 16 weeks was about threefold greater (P < .001) on lebrikizumab than on placebo. The benefit was similar on secondary endpoints, such as 75% improvement in the Eczema Area and Severity Index (EASI75) score.
At the end of the double-blind, placebo-controlled 16-week phase of the ADvocate 1 and 2 trials, which enrolled adults and adolescents aged ≥ 12 years, responders were enrolled into a maintenance phase in which they were rerandomized to 250 mg lebrikizumab every 2 weeks (Q2W) or every 4 weeks (Q4W). The latter is the approved maintenance dose.
At the end of the maintenance phase, which lasted another 32 weeks (total exposure of 52 weeks for those initially randomized to lebrikizumab), patients were invited into the ADjoin extension. The only exclusions from the extension were serious adverse events related to lebrikizumab and noncompliance.
Response Curves Appear as Straight Lines
Over the next 2 years of ADjoin, response curves appeared as straight lines not only for the overall response but when patients were stratified for different levels of response at the extension study entry. Specifically, 81.5% and 83.3% had an IGA score of 0 or 1 in the Q2W and Q4W arms at completion of the ADvocate 16-week double-blind phase. At 3 years, the rates were 84.0% and 82.9%, respectively.
For the subgroup who entered ADjoin with an EASI75 or an EASI90 response, the persistence of this level of response over 2 years was similar, although there was some gain observed among those who entered the trial with an EASI75 response.
“Not only did these patients maintain their response, but the response on average slowly improved, so that there were more patients with an EASI90 response at the 3-year timepoint,” Dr. Thaçi said.
Of the 181 patients in the ADjoin extension, 82 patients were maintained on Q2W dosing and 99 were maintained on Q4W lebrikizumab. Their mean age was about 35 years, more than half were women, and nearly 40% had severe AD at the time they enrolled in the ADvocate trials. There was essentially no difference in response rates among those in the Q2W and Q4W arms over time in ADjoin.
Side Effect Profile Essentially Unchanged
The side effect and tolerability profiles, which were favorable in the original 16-week placebo-controlled study, have remained unchanged over the subsequent maintenance phase and through the additional 2 years of the ADjoin extension.
“There continued to be reports of conjunctivitis, which is very specific for anti–IL-13 therapies,” Dr. Thaçi said. However, he said that the incidence did not increase over time, and because it was easy to treat, “most patients do not discontinue lebrikizumab for this reason.” Moreover, he said the impression was that “the number of patients experiencing adverse effects has been decreasing over time.”
Calling these long-term results “very exciting,” Dr. Thaçi called lebrikizumab “a very valuable option for long-term AD care.”
Asked for his perspective on the results, Jonathan I. Silverberg, MD, PhD, Director of Clinical Research, Department of Dermatology, George Washington University, Washington, DC, said that it is important to study long-term efficacy, and these results are positive. Without direct comparisons to other biologics available for AD, nothing can be implied about the relative efficacy of monoclonal antibodies approved for AD.
“These data are important both from an efficacy and safety perspective” for those advising patients who need chronic AD treatment, said Dr. Silverberg, who was the principal investigator of the ADvocate trials.
Earlier this year, 5-year follow-up data were published for dupilumab. Of 326 patients who remained on therapy this long, 220 (67%) maintained an IGA of 0 or 1 at the end of the study. There were no unexpected adverse events, which were generally stable or declined throughout the study.
Dr. Thaçi has financial relationships with AbbVie, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Celltrion, Galderma, Leo Pharma, L’Oreal, Janssen-Cilag, New Bridge, Novartis, Pfizer, Regeneron, Roche, Sanofi, Sun Pharma, UCB, and Vichy. Dr. Silverberg reported financial relationships with more than 40 pharmaceutical companies including those that make drugs for AD.
A version of this article appeared on Medscape.com.
AMSTERDAM — among those followed up for an additional 2 years, according to the latest data from an extension study.
At the end of the maintenance phase of the pivotal trials at 12 months, 84% of the patients enrolled into the extension had clear or almost clear skin, as per the Investigator Global Assessment (IGA). This overall figure as well as the proportion with even better responses have persisted unchanged, reported Diamant Thaçi, MD, PhD, professor and head of the Comprehensive Center for Inflammatory Medicine, University of Lübeck in Germany.
Responses at 3 Years Maintained
“This is really quite remarkable,” Dr. Thaçi said. “Roughly all the patients maintained their response.” These results became even more remarkable when patients were assessed for their use of adjunctive therapy to control flares.
“Over the whole follow-up, 90% had no need for topical corticosteroids or any other rescue therapy,” Dr. Thaçi reported, providing data from the ADjoin lebrikizumab extension study during a late-breaking news session at the annual meeting of the European Academy of Dermatology and Venereology.
The patients in ADjoin were enrolled from the pivotal phase 3 ADvocate 1 and 2 trials completed almost 2 years ago and published together in March 2023. Lebrikizumab was approved in the United States in September 2024 for moderate to severe AD in patients aged ≥ 12 years, following previous approvals in Europe in 2023 and in Japan in January 2024.
In these two identical trials with a total of 564 patients, the primary endpoint was an IGA of 0 or 1, signifying clear or almost clear skin. At nearly 40%, the proportion of patients reaching this outcome at 16 weeks was about threefold greater (P < .001) on lebrikizumab than on placebo. The benefit was similar on secondary endpoints, such as 75% improvement in the Eczema Area and Severity Index (EASI75) score.
At the end of the double-blind, placebo-controlled 16-week phase of the ADvocate 1 and 2 trials, which enrolled adults and adolescents aged ≥ 12 years, responders were enrolled into a maintenance phase in which they were rerandomized to 250 mg lebrikizumab every 2 weeks (Q2W) or every 4 weeks (Q4W). The latter is the approved maintenance dose.
At the end of the maintenance phase, which lasted another 32 weeks (total exposure of 52 weeks for those initially randomized to lebrikizumab), patients were invited into the ADjoin extension. The only exclusions from the extension were serious adverse events related to lebrikizumab and noncompliance.
Response Curves Appear as Straight Lines
Over the next 2 years of ADjoin, response curves appeared as straight lines not only for the overall response but when patients were stratified for different levels of response at the extension study entry. Specifically, 81.5% and 83.3% had an IGA score of 0 or 1 in the Q2W and Q4W arms at completion of the ADvocate 16-week double-blind phase. At 3 years, the rates were 84.0% and 82.9%, respectively.
For the subgroup who entered ADjoin with an EASI75 or an EASI90 response, the persistence of this level of response over 2 years was similar, although there was some gain observed among those who entered the trial with an EASI75 response.
“Not only did these patients maintain their response, but the response on average slowly improved, so that there were more patients with an EASI90 response at the 3-year timepoint,” Dr. Thaçi said.
Of the 181 patients in the ADjoin extension, 82 patients were maintained on Q2W dosing and 99 were maintained on Q4W lebrikizumab. Their mean age was about 35 years, more than half were women, and nearly 40% had severe AD at the time they enrolled in the ADvocate trials. There was essentially no difference in response rates among those in the Q2W and Q4W arms over time in ADjoin.
Side Effect Profile Essentially Unchanged
The side effect and tolerability profiles, which were favorable in the original 16-week placebo-controlled study, have remained unchanged over the subsequent maintenance phase and through the additional 2 years of the ADjoin extension.
“There continued to be reports of conjunctivitis, which is very specific for anti–IL-13 therapies,” Dr. Thaçi said. However, he said that the incidence did not increase over time, and because it was easy to treat, “most patients do not discontinue lebrikizumab for this reason.” Moreover, he said the impression was that “the number of patients experiencing adverse effects has been decreasing over time.”
Calling these long-term results “very exciting,” Dr. Thaçi called lebrikizumab “a very valuable option for long-term AD care.”
Asked for his perspective on the results, Jonathan I. Silverberg, MD, PhD, Director of Clinical Research, Department of Dermatology, George Washington University, Washington, DC, said that it is important to study long-term efficacy, and these results are positive. Without direct comparisons to other biologics available for AD, nothing can be implied about the relative efficacy of monoclonal antibodies approved for AD.
“These data are important both from an efficacy and safety perspective” for those advising patients who need chronic AD treatment, said Dr. Silverberg, who was the principal investigator of the ADvocate trials.
Earlier this year, 5-year follow-up data were published for dupilumab. Of 326 patients who remained on therapy this long, 220 (67%) maintained an IGA of 0 or 1 at the end of the study. There were no unexpected adverse events, which were generally stable or declined throughout the study.
Dr. Thaçi has financial relationships with AbbVie, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Celltrion, Galderma, Leo Pharma, L’Oreal, Janssen-Cilag, New Bridge, Novartis, Pfizer, Regeneron, Roche, Sanofi, Sun Pharma, UCB, and Vichy. Dr. Silverberg reported financial relationships with more than 40 pharmaceutical companies including those that make drugs for AD.
A version of this article appeared on Medscape.com.
FROM EADV 2024