User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
More weight loss linked with more benefit in STEP-HFpEF
AMSTERDAM – , including symptoms and physical limitations, exercise capacity, and inflammation, new analyses from the trial show.
At the annual congress of the European Society of Cardiology where he presented these new findings, Mikhail N. Kosiborod, MD, also posited that weight loss produced by weekly subcutaneous injections of 2.4 mg semaglutide (Wegovy) for 52 weeks in the study does not fully explain the multiple mechanisms that may be involved in producing this intervention’s effects in the STEP-HFpEF trial.
His report earlier at the congress and in a simultaneously published report of the trial’s primary outcomes established a role for medically induced weight loss in managing patients with obesity-phenotype HFpEF in a total of 529 randomized individuals with HFpEF and obesity but without diabetes.
The new analyses showed that for one of the two primary endpoints – the change from baseline in patients’ assessment on the Kansas City Cardiomyopathy Questionnaire Clinical Summary Score (KCCQ), the placebo-adjusted average change was a 16.1-point improvement in the 51 people with a 5%-10% weight loss during the 1-year study, and a 21.6-point improvement in the 58 who had at least a 20% weight loss, a between-group average 5.5 point difference that represents a clinically meaningful incremental improvement in this validated metric of symptoms and functional limitations.
Similar weight-related differences in benefit also occurred for the secondary outcomes of changes from baseline in 6-minute walk distance and in levels of C-reactive protein (CRP), a measure of systemic inflammation.
In an adjusted regression model, every 10% drop from baseline body weight was significantly linked with a 6.4-point improvement in KCCQ score, a 14.4 meter improvement in 6-minute walk distance, and a 28% relative reduction from baseline in CRP, reported Dr. Kosiborod, a cardiologist and codirector of the Haverty Cardiometabolic Center of Excellence at Saint Luke’s Mid America Heart Institute in Kansas City, Mo.
These new, prespecified analyses also showed that people with obesity and HFpEF responded roughly the same to semaglutide treatment compared with placebo-treated controls regardless of their starting body mass index, including people with class 1 (30-34 kg/m2), class 2 (35-39 kg/m2), and class 3 (≥ 40 kg/m2) obesity.
Simultaneously with Dr. Kosiborod’s report at the congress, these findings appeared in a report posted online in Nature Medicine.
Not every benefit was fully mediated by weight loss
These analyses “do not tell us how much of the benefit was mediated by weight loss, but the data do say that the more weight a person lost, the more benefit they got,” Dr. Kosiborod explained in an interview. “That is not the same as saying that everything is mediated by weight. It doesn’t say that nothing beyond weight loss matters.”
He and his associates are planning a mediation analysis of data from STEP-HFpEF that will more directly address this issue.
“It’s likely that people who lost more weight with semaglutide also had greater benefits from other effects of semaglutide at the same time. Weight loss is a good surrogate marker” for the range of effects that a person receives from treatment with semaglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist, Dr. Kosiborod said.
“GLP-1 receptor agonists may have direct effects on atherosclerosis, as well as other effects that are uncoupled from weight loss,” such as proven anti-inflammatory effects, he added.
Another exploratory effect from semaglutide treatment in the study and reported by Dr. Kosiborod was a significant reduction in serum levels of N-terminal pro brain natriuretic peptide, an association never previously seen with weight loss in people with heart failure.
“The outcomes we’ve already seen in STEP-HFpEF were largely symptomatic, which are extraordinarily important, but there may be a completely different relationship between weight and clinical events,” said John E. Deanfield, PhD, a professor of cardiology at University College Hospital, London, who was not involved in the study.
Dr. Deanfield noted that important prognostic markers such as cholesterol levels and blood pressure reductions are usually not temporally related to weight loss. “The idea that [the benefits seen in STEP-HFpEF] are purely from weight loss is something we need to be careful about,” he said.
“My gut feeling is that at least 75% of the effect [in STEP-HFpEF} was due to weight loss,” said Naveed Sattar, PhD, professor of metabolic medicine at the University of Glasgow, who was not associated with the research.
STEP-HFpEF was funded by Novo Nordisk, the company that markets semaglutide (Wegovy). Dr. Kosiborod has been a consultant and adviser to, and has received honoraria from, Novo Nordisk. He has been a consultant to numerous other companies, received research grants from AstraZeneca, Boehringer Ingelheim, and Pfizer, honoraria from AstraZeneca, and is a stockholder in Artera Health and Saghmos Therapeutics. Dr. Deanfield has been a consultant to Novo Nordisk as well as to Aegerion, Amgen, Bayer, Boehringer Ingelheim, Merck, Novartis, Pfizer, Sanofi, and Takeda, and has received research funding from Aegerion, Colgate, MSD, Pfizer, and Roche. Dr. Sattar has been a consultant to Novo Nordisk as well as to Abbott, Amgen, AstraZeneca, Boehringer Ingelheim, Lilly, Novartis, Pfizer, and Roche Diagnostics.
A version of this article first appeared on Medscape.com.
AMSTERDAM – , including symptoms and physical limitations, exercise capacity, and inflammation, new analyses from the trial show.
At the annual congress of the European Society of Cardiology where he presented these new findings, Mikhail N. Kosiborod, MD, also posited that weight loss produced by weekly subcutaneous injections of 2.4 mg semaglutide (Wegovy) for 52 weeks in the study does not fully explain the multiple mechanisms that may be involved in producing this intervention’s effects in the STEP-HFpEF trial.
His report earlier at the congress and in a simultaneously published report of the trial’s primary outcomes established a role for medically induced weight loss in managing patients with obesity-phenotype HFpEF in a total of 529 randomized individuals with HFpEF and obesity but without diabetes.
The new analyses showed that for one of the two primary endpoints – the change from baseline in patients’ assessment on the Kansas City Cardiomyopathy Questionnaire Clinical Summary Score (KCCQ), the placebo-adjusted average change was a 16.1-point improvement in the 51 people with a 5%-10% weight loss during the 1-year study, and a 21.6-point improvement in the 58 who had at least a 20% weight loss, a between-group average 5.5 point difference that represents a clinically meaningful incremental improvement in this validated metric of symptoms and functional limitations.
Similar weight-related differences in benefit also occurred for the secondary outcomes of changes from baseline in 6-minute walk distance and in levels of C-reactive protein (CRP), a measure of systemic inflammation.
In an adjusted regression model, every 10% drop from baseline body weight was significantly linked with a 6.4-point improvement in KCCQ score, a 14.4 meter improvement in 6-minute walk distance, and a 28% relative reduction from baseline in CRP, reported Dr. Kosiborod, a cardiologist and codirector of the Haverty Cardiometabolic Center of Excellence at Saint Luke’s Mid America Heart Institute in Kansas City, Mo.
These new, prespecified analyses also showed that people with obesity and HFpEF responded roughly the same to semaglutide treatment compared with placebo-treated controls regardless of their starting body mass index, including people with class 1 (30-34 kg/m2), class 2 (35-39 kg/m2), and class 3 (≥ 40 kg/m2) obesity.
Simultaneously with Dr. Kosiborod’s report at the congress, these findings appeared in a report posted online in Nature Medicine.
Not every benefit was fully mediated by weight loss
These analyses “do not tell us how much of the benefit was mediated by weight loss, but the data do say that the more weight a person lost, the more benefit they got,” Dr. Kosiborod explained in an interview. “That is not the same as saying that everything is mediated by weight. It doesn’t say that nothing beyond weight loss matters.”
He and his associates are planning a mediation analysis of data from STEP-HFpEF that will more directly address this issue.
“It’s likely that people who lost more weight with semaglutide also had greater benefits from other effects of semaglutide at the same time. Weight loss is a good surrogate marker” for the range of effects that a person receives from treatment with semaglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist, Dr. Kosiborod said.
“GLP-1 receptor agonists may have direct effects on atherosclerosis, as well as other effects that are uncoupled from weight loss,” such as proven anti-inflammatory effects, he added.
Another exploratory effect from semaglutide treatment in the study and reported by Dr. Kosiborod was a significant reduction in serum levels of N-terminal pro brain natriuretic peptide, an association never previously seen with weight loss in people with heart failure.
“The outcomes we’ve already seen in STEP-HFpEF were largely symptomatic, which are extraordinarily important, but there may be a completely different relationship between weight and clinical events,” said John E. Deanfield, PhD, a professor of cardiology at University College Hospital, London, who was not involved in the study.
Dr. Deanfield noted that important prognostic markers such as cholesterol levels and blood pressure reductions are usually not temporally related to weight loss. “The idea that [the benefits seen in STEP-HFpEF] are purely from weight loss is something we need to be careful about,” he said.
“My gut feeling is that at least 75% of the effect [in STEP-HFpEF} was due to weight loss,” said Naveed Sattar, PhD, professor of metabolic medicine at the University of Glasgow, who was not associated with the research.
STEP-HFpEF was funded by Novo Nordisk, the company that markets semaglutide (Wegovy). Dr. Kosiborod has been a consultant and adviser to, and has received honoraria from, Novo Nordisk. He has been a consultant to numerous other companies, received research grants from AstraZeneca, Boehringer Ingelheim, and Pfizer, honoraria from AstraZeneca, and is a stockholder in Artera Health and Saghmos Therapeutics. Dr. Deanfield has been a consultant to Novo Nordisk as well as to Aegerion, Amgen, Bayer, Boehringer Ingelheim, Merck, Novartis, Pfizer, Sanofi, and Takeda, and has received research funding from Aegerion, Colgate, MSD, Pfizer, and Roche. Dr. Sattar has been a consultant to Novo Nordisk as well as to Abbott, Amgen, AstraZeneca, Boehringer Ingelheim, Lilly, Novartis, Pfizer, and Roche Diagnostics.
A version of this article first appeared on Medscape.com.
AMSTERDAM – , including symptoms and physical limitations, exercise capacity, and inflammation, new analyses from the trial show.
At the annual congress of the European Society of Cardiology where he presented these new findings, Mikhail N. Kosiborod, MD, also posited that weight loss produced by weekly subcutaneous injections of 2.4 mg semaglutide (Wegovy) for 52 weeks in the study does not fully explain the multiple mechanisms that may be involved in producing this intervention’s effects in the STEP-HFpEF trial.
His report earlier at the congress and in a simultaneously published report of the trial’s primary outcomes established a role for medically induced weight loss in managing patients with obesity-phenotype HFpEF in a total of 529 randomized individuals with HFpEF and obesity but without diabetes.
The new analyses showed that for one of the two primary endpoints – the change from baseline in patients’ assessment on the Kansas City Cardiomyopathy Questionnaire Clinical Summary Score (KCCQ), the placebo-adjusted average change was a 16.1-point improvement in the 51 people with a 5%-10% weight loss during the 1-year study, and a 21.6-point improvement in the 58 who had at least a 20% weight loss, a between-group average 5.5 point difference that represents a clinically meaningful incremental improvement in this validated metric of symptoms and functional limitations.
Similar weight-related differences in benefit also occurred for the secondary outcomes of changes from baseline in 6-minute walk distance and in levels of C-reactive protein (CRP), a measure of systemic inflammation.
In an adjusted regression model, every 10% drop from baseline body weight was significantly linked with a 6.4-point improvement in KCCQ score, a 14.4 meter improvement in 6-minute walk distance, and a 28% relative reduction from baseline in CRP, reported Dr. Kosiborod, a cardiologist and codirector of the Haverty Cardiometabolic Center of Excellence at Saint Luke’s Mid America Heart Institute in Kansas City, Mo.
These new, prespecified analyses also showed that people with obesity and HFpEF responded roughly the same to semaglutide treatment compared with placebo-treated controls regardless of their starting body mass index, including people with class 1 (30-34 kg/m2), class 2 (35-39 kg/m2), and class 3 (≥ 40 kg/m2) obesity.
Simultaneously with Dr. Kosiborod’s report at the congress, these findings appeared in a report posted online in Nature Medicine.
Not every benefit was fully mediated by weight loss
These analyses “do not tell us how much of the benefit was mediated by weight loss, but the data do say that the more weight a person lost, the more benefit they got,” Dr. Kosiborod explained in an interview. “That is not the same as saying that everything is mediated by weight. It doesn’t say that nothing beyond weight loss matters.”
He and his associates are planning a mediation analysis of data from STEP-HFpEF that will more directly address this issue.
“It’s likely that people who lost more weight with semaglutide also had greater benefits from other effects of semaglutide at the same time. Weight loss is a good surrogate marker” for the range of effects that a person receives from treatment with semaglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist, Dr. Kosiborod said.
“GLP-1 receptor agonists may have direct effects on atherosclerosis, as well as other effects that are uncoupled from weight loss,” such as proven anti-inflammatory effects, he added.
Another exploratory effect from semaglutide treatment in the study and reported by Dr. Kosiborod was a significant reduction in serum levels of N-terminal pro brain natriuretic peptide, an association never previously seen with weight loss in people with heart failure.
“The outcomes we’ve already seen in STEP-HFpEF were largely symptomatic, which are extraordinarily important, but there may be a completely different relationship between weight and clinical events,” said John E. Deanfield, PhD, a professor of cardiology at University College Hospital, London, who was not involved in the study.
Dr. Deanfield noted that important prognostic markers such as cholesterol levels and blood pressure reductions are usually not temporally related to weight loss. “The idea that [the benefits seen in STEP-HFpEF] are purely from weight loss is something we need to be careful about,” he said.
“My gut feeling is that at least 75% of the effect [in STEP-HFpEF} was due to weight loss,” said Naveed Sattar, PhD, professor of metabolic medicine at the University of Glasgow, who was not associated with the research.
STEP-HFpEF was funded by Novo Nordisk, the company that markets semaglutide (Wegovy). Dr. Kosiborod has been a consultant and adviser to, and has received honoraria from, Novo Nordisk. He has been a consultant to numerous other companies, received research grants from AstraZeneca, Boehringer Ingelheim, and Pfizer, honoraria from AstraZeneca, and is a stockholder in Artera Health and Saghmos Therapeutics. Dr. Deanfield has been a consultant to Novo Nordisk as well as to Aegerion, Amgen, Bayer, Boehringer Ingelheim, Merck, Novartis, Pfizer, Sanofi, and Takeda, and has received research funding from Aegerion, Colgate, MSD, Pfizer, and Roche. Dr. Sattar has been a consultant to Novo Nordisk as well as to Abbott, Amgen, AstraZeneca, Boehringer Ingelheim, Lilly, Novartis, Pfizer, and Roche Diagnostics.
A version of this article first appeared on Medscape.com.
AT THE ESC CONGRESS 2023
Both too much and not enough sleep raises T2D risk
TOPLINE:
suggests an analysis of a Dutch study.
METHODOLOGY:
- Data on 5,561 participants aged 40–75 years from The Maastricht Study who completed the baseline survey between November 2010 and January 2018 and had full data available were included.
- Sleep duration was assessed as the in-bed time in minutes, using a median of 7 nights’ data from an activPAL3 (PAL Technologies) accelerometer, which is worn on the thigh.
- Glucose metabolism was determined via an oral glucose tolerance test and categorized as prediabetes or type 2 diabetes in line with World Health Organization diagnostic criteria.
- The association between sleep duration and type 2 diabetes was assessed on multivariate logistic regression analysis, taking into account a range of potential confounding factors.
TAKEAWAY:
- The mean age of the participants was 60.1 years, and there was an even split between men and women. In all, 832 had prediabetes and 1,341 type 2 diabetes, and the mean sleep duration was 8.3 hours.
- The results indicated there was a U-shaped relationship between sleep duration and type 2 diabetes, so that both long and short sleep durations increased the risk.
- In the fully adjusted model, a sleep duration of 5 hours was associated with an odds ratio for type 2 diabetes versus 8 hours sleep of 2.9. For a sleep duration of 12 hours, the odds ratio was 1.8.
- The association between sleep duration and diabetes was not significant.
IN PRACTICE:
The results “support the idea that sleep duration could be a relevant risk factor for type 2 diabetes independent of lifestyle risk factors, including diet, physical activity, smoking behavior, and alcohol consumption,” wrote the authors.
“These findings underpin the importance of promoting healthy sleep habits to avoid sleep deprivation,” they added.
STUDY DETAILS:
The research was led by Jeroen D. Albers, MSc, department of social medicine, Maastricht (the Netherlands) University, and published in Sleep Health. It is an analysis of The Maastricht Study.
LIMITATIONS:
The study is limited by its cross-sectional nature, particularly because there are “plausible causal paths between sleep duration and type 2 in both directions,” the authors note. The accelerometer used in the study also cannot reliably distinguish between waking and sleeping time in bed, with the potential for misclassification. Daytime naps were also not included, and long-term changes sleep patterns were not measured. In addition, it was not possible to control for some potential confounding factors.
DISCLOSURES:
The Maastricht Study was supported by the European Regional Development Fund via OP-Zuid, the Province of Limburg, the Dutch Ministry of Economic Affairs, Stichting De Weijerhorst, the Pearl String Initiative Diabetes, the School for Cardiovascular Diseases, the School for Public Health and Primary Care, the School for Nutrition and Translational Research in Metabolism, Stichting Annadal, Health Foundation Limburg, and unrestricted grants from Janssen-Cilag, Novo Nordisk, and Sanofi Aventis Netherlands. One author declares a relationship with Novo Nordisk outside the submitted work. No other relevant financial relationships were declared.
A version of this article first appeared on Medscape.com.
TOPLINE:
suggests an analysis of a Dutch study.
METHODOLOGY:
- Data on 5,561 participants aged 40–75 years from The Maastricht Study who completed the baseline survey between November 2010 and January 2018 and had full data available were included.
- Sleep duration was assessed as the in-bed time in minutes, using a median of 7 nights’ data from an activPAL3 (PAL Technologies) accelerometer, which is worn on the thigh.
- Glucose metabolism was determined via an oral glucose tolerance test and categorized as prediabetes or type 2 diabetes in line with World Health Organization diagnostic criteria.
- The association between sleep duration and type 2 diabetes was assessed on multivariate logistic regression analysis, taking into account a range of potential confounding factors.
TAKEAWAY:
- The mean age of the participants was 60.1 years, and there was an even split between men and women. In all, 832 had prediabetes and 1,341 type 2 diabetes, and the mean sleep duration was 8.3 hours.
- The results indicated there was a U-shaped relationship between sleep duration and type 2 diabetes, so that both long and short sleep durations increased the risk.
- In the fully adjusted model, a sleep duration of 5 hours was associated with an odds ratio for type 2 diabetes versus 8 hours sleep of 2.9. For a sleep duration of 12 hours, the odds ratio was 1.8.
- The association between sleep duration and diabetes was not significant.
IN PRACTICE:
The results “support the idea that sleep duration could be a relevant risk factor for type 2 diabetes independent of lifestyle risk factors, including diet, physical activity, smoking behavior, and alcohol consumption,” wrote the authors.
“These findings underpin the importance of promoting healthy sleep habits to avoid sleep deprivation,” they added.
STUDY DETAILS:
The research was led by Jeroen D. Albers, MSc, department of social medicine, Maastricht (the Netherlands) University, and published in Sleep Health. It is an analysis of The Maastricht Study.
LIMITATIONS:
The study is limited by its cross-sectional nature, particularly because there are “plausible causal paths between sleep duration and type 2 in both directions,” the authors note. The accelerometer used in the study also cannot reliably distinguish between waking and sleeping time in bed, with the potential for misclassification. Daytime naps were also not included, and long-term changes sleep patterns were not measured. In addition, it was not possible to control for some potential confounding factors.
DISCLOSURES:
The Maastricht Study was supported by the European Regional Development Fund via OP-Zuid, the Province of Limburg, the Dutch Ministry of Economic Affairs, Stichting De Weijerhorst, the Pearl String Initiative Diabetes, the School for Cardiovascular Diseases, the School for Public Health and Primary Care, the School for Nutrition and Translational Research in Metabolism, Stichting Annadal, Health Foundation Limburg, and unrestricted grants from Janssen-Cilag, Novo Nordisk, and Sanofi Aventis Netherlands. One author declares a relationship with Novo Nordisk outside the submitted work. No other relevant financial relationships were declared.
A version of this article first appeared on Medscape.com.
TOPLINE:
suggests an analysis of a Dutch study.
METHODOLOGY:
- Data on 5,561 participants aged 40–75 years from The Maastricht Study who completed the baseline survey between November 2010 and January 2018 and had full data available were included.
- Sleep duration was assessed as the in-bed time in minutes, using a median of 7 nights’ data from an activPAL3 (PAL Technologies) accelerometer, which is worn on the thigh.
- Glucose metabolism was determined via an oral glucose tolerance test and categorized as prediabetes or type 2 diabetes in line with World Health Organization diagnostic criteria.
- The association between sleep duration and type 2 diabetes was assessed on multivariate logistic regression analysis, taking into account a range of potential confounding factors.
TAKEAWAY:
- The mean age of the participants was 60.1 years, and there was an even split between men and women. In all, 832 had prediabetes and 1,341 type 2 diabetes, and the mean sleep duration was 8.3 hours.
- The results indicated there was a U-shaped relationship between sleep duration and type 2 diabetes, so that both long and short sleep durations increased the risk.
- In the fully adjusted model, a sleep duration of 5 hours was associated with an odds ratio for type 2 diabetes versus 8 hours sleep of 2.9. For a sleep duration of 12 hours, the odds ratio was 1.8.
- The association between sleep duration and diabetes was not significant.
IN PRACTICE:
The results “support the idea that sleep duration could be a relevant risk factor for type 2 diabetes independent of lifestyle risk factors, including diet, physical activity, smoking behavior, and alcohol consumption,” wrote the authors.
“These findings underpin the importance of promoting healthy sleep habits to avoid sleep deprivation,” they added.
STUDY DETAILS:
The research was led by Jeroen D. Albers, MSc, department of social medicine, Maastricht (the Netherlands) University, and published in Sleep Health. It is an analysis of The Maastricht Study.
LIMITATIONS:
The study is limited by its cross-sectional nature, particularly because there are “plausible causal paths between sleep duration and type 2 in both directions,” the authors note. The accelerometer used in the study also cannot reliably distinguish between waking and sleeping time in bed, with the potential for misclassification. Daytime naps were also not included, and long-term changes sleep patterns were not measured. In addition, it was not possible to control for some potential confounding factors.
DISCLOSURES:
The Maastricht Study was supported by the European Regional Development Fund via OP-Zuid, the Province of Limburg, the Dutch Ministry of Economic Affairs, Stichting De Weijerhorst, the Pearl String Initiative Diabetes, the School for Cardiovascular Diseases, the School for Public Health and Primary Care, the School for Nutrition and Translational Research in Metabolism, Stichting Annadal, Health Foundation Limburg, and unrestricted grants from Janssen-Cilag, Novo Nordisk, and Sanofi Aventis Netherlands. One author declares a relationship with Novo Nordisk outside the submitted work. No other relevant financial relationships were declared.
A version of this article first appeared on Medscape.com.
FROM SLEEP HEALTH
Severe COVID may cause long-term cellular changes: Study
The small study, published in Cell and funded by the National Institutes of Health, details how immune cells were analyzed through blood samples collected from 38 patients recovering from severe COVID and other critical illnesses, and from 19 healthy people. Researchers from Weill Cornell Medicine, New York, and The Jackson Laboratory for Genomic Medicine, Farmington, Conn., found through isolating hematopoietic stem cells that people recovering from severe bouts of COVID had changes to their DNA that were passed down to offspring cells.
The research team, led by Steven Josefowicz, PhD, of Weill Cornell’s pathology department, and Duygu Ucar, PhD, associate professor at The Jackson Laboratory for Genomic Medicine, discovered that this chain reaction of stem cell changes caused a boost in the production of monocytes. The authors found that, due to the innate cellular changes from a severe case of COVID, patients in recovery ended up producing a larger amount of inflammatory cytokines, rather than monocytes – distinct from samples collected from healthy patients and those recovering from other critical illnesses.
These changes to patients’ epigenetic landscapes were observed even a year after the initial COVID-19 infection. While the small participant pool meant that the research team could not establish a direct line between these innate changes and any ensuing health outcomes, the research provides us with clues as to why patients continue to struggle with inflammation and long COVID symptoms well after they recover.
While the authors reiterate the study’s limitations and hesitate to make any clear-cut associations between the results and long-term health outcomes, Wolfgang Leitner, PhD, from the NIH’s National Institute of Allergy and Infectious Diseases, predicts that long COVID can, at least in part, be explained by the changes in innate immune responses.
“Ideally, the authors would have had cells from each patient before they got infected, as a comparator, to see what the epigenetic landscape was before COVID changed it,” said Dr. Leitner. “Clear links between the severity of COVID and genetics were discovered already early in the pandemic and this paper should prompt follow-up studies that link mutations in immune genes with the epigenetic changes described here.”
Dr. Leitner said he had some initial predictions about the long-term impact of COVID-19, but he had not anticipated some of what the study’s findings now show.
“Unlike in the case of, for example, influenza, where the lungs go into ‘repair mode’ after the infection has been resolved – which leaves people susceptible to secondary infections for up to several months – this study shows that after severe COVID, the immune system remains in ‘emergency mode’ and in a heightened state of inflammation,” said Dr. Leitner.
“That further aggravates the problem the initial strong inflammation causes: even higher risk of autoimmune disease, but also, cancer.”
Commenting on the findings, Eric Topol, MD, editor-in-chief of Medscape Medical News, said the study presents “evidence that a key line of immune cells are essentially irrevocably, epigenetically altered and activated.
“You do not want to have this [COVID],” he added.
The study also highlights the researchers’ novel approach to isolating hematopoietic stem cells, found largely in bone marrow. This type of research has been limited in the past because of how costly and invasive it can be to analyze cells in bone marrow. But, by isolating and enriching hematopoietic stem cells, the team can decipher the full cellular diversity of the cells’ bone marrow counterparts.
“This revelation opened the doors to study, at single-cell resolution, how stem cells are affected upon infection and vaccination with a simple blood draw,” representatives from the Jackson lab said in a press release.
A version of this article appeared on Medscape.com.
The small study, published in Cell and funded by the National Institutes of Health, details how immune cells were analyzed through blood samples collected from 38 patients recovering from severe COVID and other critical illnesses, and from 19 healthy people. Researchers from Weill Cornell Medicine, New York, and The Jackson Laboratory for Genomic Medicine, Farmington, Conn., found through isolating hematopoietic stem cells that people recovering from severe bouts of COVID had changes to their DNA that were passed down to offspring cells.
The research team, led by Steven Josefowicz, PhD, of Weill Cornell’s pathology department, and Duygu Ucar, PhD, associate professor at The Jackson Laboratory for Genomic Medicine, discovered that this chain reaction of stem cell changes caused a boost in the production of monocytes. The authors found that, due to the innate cellular changes from a severe case of COVID, patients in recovery ended up producing a larger amount of inflammatory cytokines, rather than monocytes – distinct from samples collected from healthy patients and those recovering from other critical illnesses.
These changes to patients’ epigenetic landscapes were observed even a year after the initial COVID-19 infection. While the small participant pool meant that the research team could not establish a direct line between these innate changes and any ensuing health outcomes, the research provides us with clues as to why patients continue to struggle with inflammation and long COVID symptoms well after they recover.
While the authors reiterate the study’s limitations and hesitate to make any clear-cut associations between the results and long-term health outcomes, Wolfgang Leitner, PhD, from the NIH’s National Institute of Allergy and Infectious Diseases, predicts that long COVID can, at least in part, be explained by the changes in innate immune responses.
“Ideally, the authors would have had cells from each patient before they got infected, as a comparator, to see what the epigenetic landscape was before COVID changed it,” said Dr. Leitner. “Clear links between the severity of COVID and genetics were discovered already early in the pandemic and this paper should prompt follow-up studies that link mutations in immune genes with the epigenetic changes described here.”
Dr. Leitner said he had some initial predictions about the long-term impact of COVID-19, but he had not anticipated some of what the study’s findings now show.
“Unlike in the case of, for example, influenza, where the lungs go into ‘repair mode’ after the infection has been resolved – which leaves people susceptible to secondary infections for up to several months – this study shows that after severe COVID, the immune system remains in ‘emergency mode’ and in a heightened state of inflammation,” said Dr. Leitner.
“That further aggravates the problem the initial strong inflammation causes: even higher risk of autoimmune disease, but also, cancer.”
Commenting on the findings, Eric Topol, MD, editor-in-chief of Medscape Medical News, said the study presents “evidence that a key line of immune cells are essentially irrevocably, epigenetically altered and activated.
“You do not want to have this [COVID],” he added.
The study also highlights the researchers’ novel approach to isolating hematopoietic stem cells, found largely in bone marrow. This type of research has been limited in the past because of how costly and invasive it can be to analyze cells in bone marrow. But, by isolating and enriching hematopoietic stem cells, the team can decipher the full cellular diversity of the cells’ bone marrow counterparts.
“This revelation opened the doors to study, at single-cell resolution, how stem cells are affected upon infection and vaccination with a simple blood draw,” representatives from the Jackson lab said in a press release.
A version of this article appeared on Medscape.com.
The small study, published in Cell and funded by the National Institutes of Health, details how immune cells were analyzed through blood samples collected from 38 patients recovering from severe COVID and other critical illnesses, and from 19 healthy people. Researchers from Weill Cornell Medicine, New York, and The Jackson Laboratory for Genomic Medicine, Farmington, Conn., found through isolating hematopoietic stem cells that people recovering from severe bouts of COVID had changes to their DNA that were passed down to offspring cells.
The research team, led by Steven Josefowicz, PhD, of Weill Cornell’s pathology department, and Duygu Ucar, PhD, associate professor at The Jackson Laboratory for Genomic Medicine, discovered that this chain reaction of stem cell changes caused a boost in the production of monocytes. The authors found that, due to the innate cellular changes from a severe case of COVID, patients in recovery ended up producing a larger amount of inflammatory cytokines, rather than monocytes – distinct from samples collected from healthy patients and those recovering from other critical illnesses.
These changes to patients’ epigenetic landscapes were observed even a year after the initial COVID-19 infection. While the small participant pool meant that the research team could not establish a direct line between these innate changes and any ensuing health outcomes, the research provides us with clues as to why patients continue to struggle with inflammation and long COVID symptoms well after they recover.
While the authors reiterate the study’s limitations and hesitate to make any clear-cut associations between the results and long-term health outcomes, Wolfgang Leitner, PhD, from the NIH’s National Institute of Allergy and Infectious Diseases, predicts that long COVID can, at least in part, be explained by the changes in innate immune responses.
“Ideally, the authors would have had cells from each patient before they got infected, as a comparator, to see what the epigenetic landscape was before COVID changed it,” said Dr. Leitner. “Clear links between the severity of COVID and genetics were discovered already early in the pandemic and this paper should prompt follow-up studies that link mutations in immune genes with the epigenetic changes described here.”
Dr. Leitner said he had some initial predictions about the long-term impact of COVID-19, but he had not anticipated some of what the study’s findings now show.
“Unlike in the case of, for example, influenza, where the lungs go into ‘repair mode’ after the infection has been resolved – which leaves people susceptible to secondary infections for up to several months – this study shows that after severe COVID, the immune system remains in ‘emergency mode’ and in a heightened state of inflammation,” said Dr. Leitner.
“That further aggravates the problem the initial strong inflammation causes: even higher risk of autoimmune disease, but also, cancer.”
Commenting on the findings, Eric Topol, MD, editor-in-chief of Medscape Medical News, said the study presents “evidence that a key line of immune cells are essentially irrevocably, epigenetically altered and activated.
“You do not want to have this [COVID],” he added.
The study also highlights the researchers’ novel approach to isolating hematopoietic stem cells, found largely in bone marrow. This type of research has been limited in the past because of how costly and invasive it can be to analyze cells in bone marrow. But, by isolating and enriching hematopoietic stem cells, the team can decipher the full cellular diversity of the cells’ bone marrow counterparts.
“This revelation opened the doors to study, at single-cell resolution, how stem cells are affected upon infection and vaccination with a simple blood draw,” representatives from the Jackson lab said in a press release.
A version of this article appeared on Medscape.com.
FROM CELL
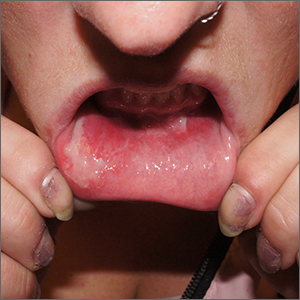
Inner lip erosions
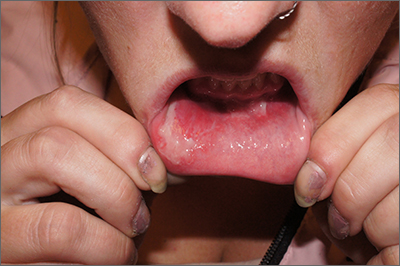
The patient was having a flare of pemphigus vulgaris (PV), a rare and sometimes life-threatening acquired autoimmune blistering disease that affects the skin and/or mucosa. Ashkenazi Jewish patients and patients from Mediterranean and Middle Eastern countries are more likely to be affected.
In PV, acquired autoantibodies target the desmosomes that connect epithelial cells together, weakening the intercellular adhesion. It can affect skin, mucosa, or both. Patients present with fragile bullae or ulcers. The connections between the cells are often so damaged that rubbing on the skin creates a new blister called “Nikolsky sign.” In the mouth, bullae erode rapidly. Look for disease affecting the ocular conjunctiva or sclera, as well. PV can also occasionally affect the nasopharynx and esophagus, usually manifesting as hemoptysis, dysphagia, and nosebleeds with ulcer seen on endoscopy or otolaryngoscopy.
Although PV is often severe (and can warrant hospitalization when significant body surface area is involved), some patients may have few active lesions and can be managed safely as outpatients.
The diagnosis requires 2 biopsies and serum for indirect immunofluorescence. One biopsy (either by punch or shave to the upper dermis) is taken from the edge of a bulla or ulcer. Another biopsy (by punch or shave) is taken from nearby normal-looking skin or mucosa for testing the direct immunofluorescence pattern. In the mucosa, a punch biopsy may be left open or closed with absorbable sutures. A serum sample is taken for indirect immunofluorescence to differentiate pemphigus vulgaris from other forms of pemphigus.1
PV is treated by suppressing the immune system. Focal disease may be treated with super-potent topical steroids, including clobetasol 0.05% ointment. Even in the mouth, topical clobetasol 0.05% may be used off-label twice daily until control is achieved. When topical treatment is used in the mouth, advise patients to apply the clobetasol ointment to a piece of gauze and place the gauze (ointment side down) over affected areas for 20 to 30 minutes twice daily.
Patients with widespread or severe disease should be hospitalized. In severe cases, supportive wound care is provided, and treatment is aimed at immunosuppression. Systemic options include high-dose prednisone 0.5 to 1 mg/kg daily until clear, a steroid-sparing immunosuppressant such as mycophenolate mofetil up to 1000 mg bid, or rituximab in 1 of several regimens.
Three years prior to this patient’s visit, she had been successfully treated for PV with a course of rituximab. To treat the current flare, she was started on prednisone 60 mg/d. In addition, the plan was for her to complete 2 infusions of 1000 mg rituximab 2 weeks apart.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, Maine.
1. Didona, D, Schmidt, MF, Maglie, R, et al. Pemphigus and pemphigoids: clinical presentation, diagnosis and therapy. J Dtsch Dermatol Ges. 2023;1-20. doi: 10.1111/ddg.15174

The patient was having a flare of pemphigus vulgaris (PV), a rare and sometimes life-threatening acquired autoimmune blistering disease that affects the skin and/or mucosa. Ashkenazi Jewish patients and patients from Mediterranean and Middle Eastern countries are more likely to be affected.
In PV, acquired autoantibodies target the desmosomes that connect epithelial cells together, weakening the intercellular adhesion. It can affect skin, mucosa, or both. Patients present with fragile bullae or ulcers. The connections between the cells are often so damaged that rubbing on the skin creates a new blister called “Nikolsky sign.” In the mouth, bullae erode rapidly. Look for disease affecting the ocular conjunctiva or sclera, as well. PV can also occasionally affect the nasopharynx and esophagus, usually manifesting as hemoptysis, dysphagia, and nosebleeds with ulcer seen on endoscopy or otolaryngoscopy.
Although PV is often severe (and can warrant hospitalization when significant body surface area is involved), some patients may have few active lesions and can be managed safely as outpatients.
The diagnosis requires 2 biopsies and serum for indirect immunofluorescence. One biopsy (either by punch or shave to the upper dermis) is taken from the edge of a bulla or ulcer. Another biopsy (by punch or shave) is taken from nearby normal-looking skin or mucosa for testing the direct immunofluorescence pattern. In the mucosa, a punch biopsy may be left open or closed with absorbable sutures. A serum sample is taken for indirect immunofluorescence to differentiate pemphigus vulgaris from other forms of pemphigus.1
PV is treated by suppressing the immune system. Focal disease may be treated with super-potent topical steroids, including clobetasol 0.05% ointment. Even in the mouth, topical clobetasol 0.05% may be used off-label twice daily until control is achieved. When topical treatment is used in the mouth, advise patients to apply the clobetasol ointment to a piece of gauze and place the gauze (ointment side down) over affected areas for 20 to 30 minutes twice daily.
Patients with widespread or severe disease should be hospitalized. In severe cases, supportive wound care is provided, and treatment is aimed at immunosuppression. Systemic options include high-dose prednisone 0.5 to 1 mg/kg daily until clear, a steroid-sparing immunosuppressant such as mycophenolate mofetil up to 1000 mg bid, or rituximab in 1 of several regimens.
Three years prior to this patient’s visit, she had been successfully treated for PV with a course of rituximab. To treat the current flare, she was started on prednisone 60 mg/d. In addition, the plan was for her to complete 2 infusions of 1000 mg rituximab 2 weeks apart.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, Maine.

The patient was having a flare of pemphigus vulgaris (PV), a rare and sometimes life-threatening acquired autoimmune blistering disease that affects the skin and/or mucosa. Ashkenazi Jewish patients and patients from Mediterranean and Middle Eastern countries are more likely to be affected.
In PV, acquired autoantibodies target the desmosomes that connect epithelial cells together, weakening the intercellular adhesion. It can affect skin, mucosa, or both. Patients present with fragile bullae or ulcers. The connections between the cells are often so damaged that rubbing on the skin creates a new blister called “Nikolsky sign.” In the mouth, bullae erode rapidly. Look for disease affecting the ocular conjunctiva or sclera, as well. PV can also occasionally affect the nasopharynx and esophagus, usually manifesting as hemoptysis, dysphagia, and nosebleeds with ulcer seen on endoscopy or otolaryngoscopy.
Although PV is often severe (and can warrant hospitalization when significant body surface area is involved), some patients may have few active lesions and can be managed safely as outpatients.
The diagnosis requires 2 biopsies and serum for indirect immunofluorescence. One biopsy (either by punch or shave to the upper dermis) is taken from the edge of a bulla or ulcer. Another biopsy (by punch or shave) is taken from nearby normal-looking skin or mucosa for testing the direct immunofluorescence pattern. In the mucosa, a punch biopsy may be left open or closed with absorbable sutures. A serum sample is taken for indirect immunofluorescence to differentiate pemphigus vulgaris from other forms of pemphigus.1
PV is treated by suppressing the immune system. Focal disease may be treated with super-potent topical steroids, including clobetasol 0.05% ointment. Even in the mouth, topical clobetasol 0.05% may be used off-label twice daily until control is achieved. When topical treatment is used in the mouth, advise patients to apply the clobetasol ointment to a piece of gauze and place the gauze (ointment side down) over affected areas for 20 to 30 minutes twice daily.
Patients with widespread or severe disease should be hospitalized. In severe cases, supportive wound care is provided, and treatment is aimed at immunosuppression. Systemic options include high-dose prednisone 0.5 to 1 mg/kg daily until clear, a steroid-sparing immunosuppressant such as mycophenolate mofetil up to 1000 mg bid, or rituximab in 1 of several regimens.
Three years prior to this patient’s visit, she had been successfully treated for PV with a course of rituximab. To treat the current flare, she was started on prednisone 60 mg/d. In addition, the plan was for her to complete 2 infusions of 1000 mg rituximab 2 weeks apart.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, Maine.
1. Didona, D, Schmidt, MF, Maglie, R, et al. Pemphigus and pemphigoids: clinical presentation, diagnosis and therapy. J Dtsch Dermatol Ges. 2023;1-20. doi: 10.1111/ddg.15174
1. Didona, D, Schmidt, MF, Maglie, R, et al. Pemphigus and pemphigoids: clinical presentation, diagnosis and therapy. J Dtsch Dermatol Ges. 2023;1-20. doi: 10.1111/ddg.15174
Use of mental health services soared during pandemic
By the end of August 2022, overall use of mental health services was almost 40% higher than before the COVID-19 pandemic, while spending increased by 54%, according to a new study by researchers at the RAND Corporation.
During the early phase of the pandemic, from mid-March to mid-December 2020, before the vaccine was available, in-person visits decreased by 40%, while telehealth visits increased by 1,000%, reported Jonathan H. Cantor, PhD, and colleagues at RAND, and at Castlight Health, a benefit coordination provider, in a paper published online in JAMA Health Forum.
Between December 2020 and August 2022, telehealth visits stayed stable, but in-person visits creeped back up, eventually reaching 80% of prepandemic levels. However, “total utilization was higher than before the pandemic,” Dr. Cantor, a policy researcher at RAND, told this news organization.
“It could be that it’s easier for individuals to receive care via telehealth, but it could also just be that there’s a greater demand or need since the pandemic,” said Dr. Cantor. “We’ll just need more research to actually unpack what’s going on,” he said.
Initial per capita spending increased by about a third and was up overall by more than half. But it’s not clear how much of that is due to utilization or to price of services, said Dr. Cantor. Spending for telehealth services remained stable in the post-vaccine period, while spending on in-person visits returned to prepandemic levels.
Dr. Cantor and his colleagues were not able to determine whether utilization was by new or existing patients, but he said that would be good data to have. “It would be really important to know whether or not folks are initiating care because telehealth is making it easier,” he said.
The authors analyzed about 1.5 million claims for anxiety disorders, major depressive disorder, bipolar disorder, schizophrenia, and posttraumatic stress disorder, out of claims submitted by 7 million commercially insured adults whose self-insured employers used the Castlight benefit.
Dr. Cantor noted that this is just a small subset of the U.S. population. He said he’d like to have data from Medicare and Medicaid to fully assess the impact of the COVID-19 pandemic on mental health and of telehealth visits.
“This is a still-burgeoning field,” he said about telehealth. “We’re still trying to get a handle on how things are operating, given that there’s been so much change so rapidly.”
Meanwhile, 152 major employers responding to a large national survey this summer said that they’ve been grappling with how COVID-19 has affected workers. The employers include 72 Fortune 100 companies and provide health coverage for more than 60 million workers, retirees, and their families.
Seventy-seven percent said they are currently seeing an increase in depression, anxiety, and substance use disorders as a result of the pandemic, according to the Business Group on Health’s survey. That’s up from 44% in 2022.
Going forward, employers will focus on increasing access to mental health services, the survey reported.
“Our survey found that in 2024 and for the near future, employers will be acutely focused on addressing employees’ mental health needs while ensuring access and lowering cost barriers,” Ellen Kelsay, president and CEO of Business Group on Health, said in a statement.
The study was supported by grants from the National Institute of Mental Health and the National Institute on Aging. Coauthor Dena Bravata, MD, a Castlight employee, reported receiving personal fees from Castlight Health during the conduct of the study. Coauthor Christopher M. Whaley, a RAND employee, reported receiving personal fees from Castlight Health outside the submitted work.
A version of this article appeared on Medscape.com.
By the end of August 2022, overall use of mental health services was almost 40% higher than before the COVID-19 pandemic, while spending increased by 54%, according to a new study by researchers at the RAND Corporation.
During the early phase of the pandemic, from mid-March to mid-December 2020, before the vaccine was available, in-person visits decreased by 40%, while telehealth visits increased by 1,000%, reported Jonathan H. Cantor, PhD, and colleagues at RAND, and at Castlight Health, a benefit coordination provider, in a paper published online in JAMA Health Forum.
Between December 2020 and August 2022, telehealth visits stayed stable, but in-person visits creeped back up, eventually reaching 80% of prepandemic levels. However, “total utilization was higher than before the pandemic,” Dr. Cantor, a policy researcher at RAND, told this news organization.
“It could be that it’s easier for individuals to receive care via telehealth, but it could also just be that there’s a greater demand or need since the pandemic,” said Dr. Cantor. “We’ll just need more research to actually unpack what’s going on,” he said.
Initial per capita spending increased by about a third and was up overall by more than half. But it’s not clear how much of that is due to utilization or to price of services, said Dr. Cantor. Spending for telehealth services remained stable in the post-vaccine period, while spending on in-person visits returned to prepandemic levels.
Dr. Cantor and his colleagues were not able to determine whether utilization was by new or existing patients, but he said that would be good data to have. “It would be really important to know whether or not folks are initiating care because telehealth is making it easier,” he said.
The authors analyzed about 1.5 million claims for anxiety disorders, major depressive disorder, bipolar disorder, schizophrenia, and posttraumatic stress disorder, out of claims submitted by 7 million commercially insured adults whose self-insured employers used the Castlight benefit.
Dr. Cantor noted that this is just a small subset of the U.S. population. He said he’d like to have data from Medicare and Medicaid to fully assess the impact of the COVID-19 pandemic on mental health and of telehealth visits.
“This is a still-burgeoning field,” he said about telehealth. “We’re still trying to get a handle on how things are operating, given that there’s been so much change so rapidly.”
Meanwhile, 152 major employers responding to a large national survey this summer said that they’ve been grappling with how COVID-19 has affected workers. The employers include 72 Fortune 100 companies and provide health coverage for more than 60 million workers, retirees, and their families.
Seventy-seven percent said they are currently seeing an increase in depression, anxiety, and substance use disorders as a result of the pandemic, according to the Business Group on Health’s survey. That’s up from 44% in 2022.
Going forward, employers will focus on increasing access to mental health services, the survey reported.
“Our survey found that in 2024 and for the near future, employers will be acutely focused on addressing employees’ mental health needs while ensuring access and lowering cost barriers,” Ellen Kelsay, president and CEO of Business Group on Health, said in a statement.
The study was supported by grants from the National Institute of Mental Health and the National Institute on Aging. Coauthor Dena Bravata, MD, a Castlight employee, reported receiving personal fees from Castlight Health during the conduct of the study. Coauthor Christopher M. Whaley, a RAND employee, reported receiving personal fees from Castlight Health outside the submitted work.
A version of this article appeared on Medscape.com.
By the end of August 2022, overall use of mental health services was almost 40% higher than before the COVID-19 pandemic, while spending increased by 54%, according to a new study by researchers at the RAND Corporation.
During the early phase of the pandemic, from mid-March to mid-December 2020, before the vaccine was available, in-person visits decreased by 40%, while telehealth visits increased by 1,000%, reported Jonathan H. Cantor, PhD, and colleagues at RAND, and at Castlight Health, a benefit coordination provider, in a paper published online in JAMA Health Forum.
Between December 2020 and August 2022, telehealth visits stayed stable, but in-person visits creeped back up, eventually reaching 80% of prepandemic levels. However, “total utilization was higher than before the pandemic,” Dr. Cantor, a policy researcher at RAND, told this news organization.
“It could be that it’s easier for individuals to receive care via telehealth, but it could also just be that there’s a greater demand or need since the pandemic,” said Dr. Cantor. “We’ll just need more research to actually unpack what’s going on,” he said.
Initial per capita spending increased by about a third and was up overall by more than half. But it’s not clear how much of that is due to utilization or to price of services, said Dr. Cantor. Spending for telehealth services remained stable in the post-vaccine period, while spending on in-person visits returned to prepandemic levels.
Dr. Cantor and his colleagues were not able to determine whether utilization was by new or existing patients, but he said that would be good data to have. “It would be really important to know whether or not folks are initiating care because telehealth is making it easier,” he said.
The authors analyzed about 1.5 million claims for anxiety disorders, major depressive disorder, bipolar disorder, schizophrenia, and posttraumatic stress disorder, out of claims submitted by 7 million commercially insured adults whose self-insured employers used the Castlight benefit.
Dr. Cantor noted that this is just a small subset of the U.S. population. He said he’d like to have data from Medicare and Medicaid to fully assess the impact of the COVID-19 pandemic on mental health and of telehealth visits.
“This is a still-burgeoning field,” he said about telehealth. “We’re still trying to get a handle on how things are operating, given that there’s been so much change so rapidly.”
Meanwhile, 152 major employers responding to a large national survey this summer said that they’ve been grappling with how COVID-19 has affected workers. The employers include 72 Fortune 100 companies and provide health coverage for more than 60 million workers, retirees, and their families.
Seventy-seven percent said they are currently seeing an increase in depression, anxiety, and substance use disorders as a result of the pandemic, according to the Business Group on Health’s survey. That’s up from 44% in 2022.
Going forward, employers will focus on increasing access to mental health services, the survey reported.
“Our survey found that in 2024 and for the near future, employers will be acutely focused on addressing employees’ mental health needs while ensuring access and lowering cost barriers,” Ellen Kelsay, president and CEO of Business Group on Health, said in a statement.
The study was supported by grants from the National Institute of Mental Health and the National Institute on Aging. Coauthor Dena Bravata, MD, a Castlight employee, reported receiving personal fees from Castlight Health during the conduct of the study. Coauthor Christopher M. Whaley, a RAND employee, reported receiving personal fees from Castlight Health outside the submitted work.
A version of this article appeared on Medscape.com.
Anticoagulation no benefit in presumed AFib detected by cardiac devices
AMSTERDAM – Among patients with atrial high-rate episodes detected by implantable devices, anticoagulation with edoxaban did not significantly reduce the incidence of a composite outcome of cardiovascular death, stroke, or systemic embolism in comparison with placebo but was associated with a higher bleeding risk in the NOAH-AFNET 6 trial.
“ They do not need to be anticoagulated. That is a relief,” the lead investigator of the trial, Paulus Kirchhof, MD, University Heart and Vascular Center Hamburg (Germany), said in an interview.
Dr. Kirchhof pointed out that this result was unexpected. “Many of us thought that because atrial high-rate episodes look like AF[ib] when they occur, then they are an indication for anticoagulation. But based on these results from the first-ever randomized trial on this population, there is no need for anticoagulation in these patients.”
Dr. Kirchhof presented the NOAH-AFNET 6 trial at the annual congress of the European Society of Cardiology. The study was also simultaneously published in the New England Journal of Medicine.
The trial recruited patients with implanted devices that enable continuous monitoring of atrial rhythm, such as pacemakers and defibrillators. “Because we can record the rhythm day and night with these devices, they pick up small abnormalities. About 20% of these patients experience these occasional atrial high-rate episodes – short episodes that look like AF[ib], but they are rare and brief,” Dr. Kirchhof noted.
He explained that whether the occurrence of these atrial high-rate episodes in patients without AFib, as documented on a conventional electrocardiogram, justifies the initiation of anticoagulants has been unclear. “But this trial tells us that these episodes are different to AF[ib] that is diagnosed on ECG,” he added.
Another finding in the trial was that among these patients, there was an unexpectedly low rate of stroke, despite the patients’ having a CHADSVASC score of 4.
“Based on the result of this trial, these occasional atrial high-rate episodes do not appear to be associated with stroke. It appears quite benign,” Dr. Kirchhof said.
Implications for wearable technology?
He said the results may also have implications for wearable devices that pick up abnormal heart rhythm, such as smartwatches.
“We don’t know exactly what these wearable technologies are picking up, but most likely it is these atrial high-rate episodes. But we need more research on the value of these wearable technologies; we need randomized trials in this particular patient population before we consider anticoagulation in these patients,” Dr. Kirchhof stated.
The NOAH-AFNET 6 study was an event-driven, double-blind, double-dummy, randomized trial involving 2,536 patients aged 65 years or older who had atrial high-rate episodes that lasted for at least 6 minutes and who had at least one additional risk factor for stroke.
Patients were randomly assigned in a 1:1 ratio to receive edoxaban or placebo. The primary efficacy outcome was a composite of cardiovascular death, stroke, or systemic embolism, evaluated in a time-to-event analysis. The safety outcome was a composite of death from any cause or major bleeding.
The mean age of the patients was 78 years, 37.4% were women, and the median duration of atrial high-rate episodes was 2.8 hours. The trial was terminated early, at a median follow-up of 21 months, on the basis of safety concerns and the results of an independent, informal assessment of futility for the efficacy of edoxaban; at termination, the planned enrollment had been completed.
Results showed that a primary efficacy outcome event occurred in 83 patients (3.2% per patient-year) in the edoxaban group and in 101 patients (4.0% per patient-year) in the placebo group (hazard ratio, 0.81; 95% confidence interval, 0.60-1.08; P = .15). The incidence of stroke was approximately 1% per patient-year in both groups.
A safety outcome event occurred in 149 patients (5.9% per patient-year) in the edoxaban group and in 114 patients (4.5% per patient-year) in the placebo group (HR, 1.31; 95% CI, 1.02-1.67; P = .03).
ECG-diagnosed AFib developed in 462 of 2,536 patients (18.2% total, 8.7% per patient-year).
In the NEJM article, the authors wrote that the findings of this trial – the low incidence of stroke that was not further reduced by treatment with edoxaban – may make it appropriate to withhold anticoagulant therapy for patients with atrial high-rate episodes.
The main difference between the population studied in this trial and patients with AFib, as documented on an ECG, appears to be the paucity and brevity of atrial arrhythmias in patients with atrial high-rate episodes (termed low arrhythmia burden). Published reports show that a low arrhythmia burden contributes to a low incidence of stroke among patients with AFib, the study authors wrote.
They added that the low rate of stroke in this trial suggests that in addition to clinical risk prediction formulas for stroke, methods to improve the estimation of stroke risk among patients with infrequent atrial arrhythmias detected by long-term monitoring are needed to guide decision-making on the use of anticoagulation.
Commenting on the NOAH-AFNET 6 results, the comoderator of the ESC HOTLINE session at which they were presented, Barbara Casadei, MD, John Radcliffe Hospital, Oxford, England, said: “Finally we know what to with these patients. Before we just had a variety of opinions with no evidence. I think that the trial really highlights that patients who come to the doctor with symptoms of AF[ib] or who have ECG-documented AF[ib] have a much higher risk of cardioembolic stroke than patients in whom this presumed AF[ib] is picked up incidentally from implanted devices.”
She added: “The stroke rates are very low in this trial, so anticoagulation was never going to work. But this is an important finding. We know that anticoagulants are not a free lunch. There is a significant bleeding risk. These results suggest that unless a patient has clinical AF[ib] that shows up on an ECG then we need to more cautious in prescribing anticoagulation.”
Also commenting on the study, immediate past president of the American College of Cardiology Ed Fry, MD, Ascension Indiana St. Vincent Heart Center, Indianapolis, said the management of patients with implanted cardiac devices or personal wearable technology that has picked up an abnormal rhythm suggestive of AFib was a big question in clinical practice.
“These episodes could be AF[ib], which comes with an increased stroke risk, but it could also be something else like atrial tachycardia or supraventricular tachycardia, which do not confer an increased stroke risk,” he explained.
“This study shows that without a firm diagnosis of AF[ib] on an ECG or some sort of continuous AF[ib] monitoring device, we are going to be anticoagulating people who don’t need it. They were exposed to the risk of bleeding without getting the benefit of a reduction in stroke risk,” Dr. Fry noted.
“The important outcome from this trial is that it gives comfort in we can be more confident in withholding anticoagulation until we get a firm diagnosis of AF[ib]. If we have a high index of suspicion that this could be AF[ib], then we can arrange for a further testing,” he added.
Second trial reporting soon
A trial similar to NOAH-AFNET 6 is currently underway – the ARTESIA trial, which is expected to be reported later in 2023.
“We are in close contact with the leadership of that trial, and we hope to do some meta-analysis,” Dr. Kirchhof said. “But I think today we’ve gone from no evidence to one outcome-based trial which shows there is no reason to use anticoagulation in these patients with atrial high-rate episodes. I think this is reason to change practice now, but yes, of course we need to look at the data in totality once the second trial has reported.”
But the lead investigator of the ARTESIA trial, Stuart Connolly, MD, McMaster University, Hamilton, Ont., does not believe the NOAH-AFNET 6 trial should change practice at this time.
“This trial fails to adequately address the critical issue that drives clinical decision-making in these patients because it is underpowered for the most important endpoint of stroke,” he said in an interview.
“The key question is whether anticoagulation reduces stroke in these patients,” he added. “To answer that, a clinical trial needs to have a lot of strokes, and this trial had very few. The trial was stopped early and had way too few strokes to properly answer this key question.”
The NOAH-AFNET 6 trial was an investigator-initiated trial funded by the German Center for Cardiovascular Research and Daiichi Sankyo Europe. Dr. Kirchhof reported research support from several drug and device companies active in AFib. He is also listed as an inventor on two patents held by the University of Hamburg on AFib therapy and AFib markers.
A version of this article first appeared on Medscape.com.
AMSTERDAM – Among patients with atrial high-rate episodes detected by implantable devices, anticoagulation with edoxaban did not significantly reduce the incidence of a composite outcome of cardiovascular death, stroke, or systemic embolism in comparison with placebo but was associated with a higher bleeding risk in the NOAH-AFNET 6 trial.
“ They do not need to be anticoagulated. That is a relief,” the lead investigator of the trial, Paulus Kirchhof, MD, University Heart and Vascular Center Hamburg (Germany), said in an interview.
Dr. Kirchhof pointed out that this result was unexpected. “Many of us thought that because atrial high-rate episodes look like AF[ib] when they occur, then they are an indication for anticoagulation. But based on these results from the first-ever randomized trial on this population, there is no need for anticoagulation in these patients.”
Dr. Kirchhof presented the NOAH-AFNET 6 trial at the annual congress of the European Society of Cardiology. The study was also simultaneously published in the New England Journal of Medicine.
The trial recruited patients with implanted devices that enable continuous monitoring of atrial rhythm, such as pacemakers and defibrillators. “Because we can record the rhythm day and night with these devices, they pick up small abnormalities. About 20% of these patients experience these occasional atrial high-rate episodes – short episodes that look like AF[ib], but they are rare and brief,” Dr. Kirchhof noted.
He explained that whether the occurrence of these atrial high-rate episodes in patients without AFib, as documented on a conventional electrocardiogram, justifies the initiation of anticoagulants has been unclear. “But this trial tells us that these episodes are different to AF[ib] that is diagnosed on ECG,” he added.
Another finding in the trial was that among these patients, there was an unexpectedly low rate of stroke, despite the patients’ having a CHADSVASC score of 4.
“Based on the result of this trial, these occasional atrial high-rate episodes do not appear to be associated with stroke. It appears quite benign,” Dr. Kirchhof said.
Implications for wearable technology?
He said the results may also have implications for wearable devices that pick up abnormal heart rhythm, such as smartwatches.
“We don’t know exactly what these wearable technologies are picking up, but most likely it is these atrial high-rate episodes. But we need more research on the value of these wearable technologies; we need randomized trials in this particular patient population before we consider anticoagulation in these patients,” Dr. Kirchhof stated.
The NOAH-AFNET 6 study was an event-driven, double-blind, double-dummy, randomized trial involving 2,536 patients aged 65 years or older who had atrial high-rate episodes that lasted for at least 6 minutes and who had at least one additional risk factor for stroke.
Patients were randomly assigned in a 1:1 ratio to receive edoxaban or placebo. The primary efficacy outcome was a composite of cardiovascular death, stroke, or systemic embolism, evaluated in a time-to-event analysis. The safety outcome was a composite of death from any cause or major bleeding.
The mean age of the patients was 78 years, 37.4% were women, and the median duration of atrial high-rate episodes was 2.8 hours. The trial was terminated early, at a median follow-up of 21 months, on the basis of safety concerns and the results of an independent, informal assessment of futility for the efficacy of edoxaban; at termination, the planned enrollment had been completed.
Results showed that a primary efficacy outcome event occurred in 83 patients (3.2% per patient-year) in the edoxaban group and in 101 patients (4.0% per patient-year) in the placebo group (hazard ratio, 0.81; 95% confidence interval, 0.60-1.08; P = .15). The incidence of stroke was approximately 1% per patient-year in both groups.
A safety outcome event occurred in 149 patients (5.9% per patient-year) in the edoxaban group and in 114 patients (4.5% per patient-year) in the placebo group (HR, 1.31; 95% CI, 1.02-1.67; P = .03).
ECG-diagnosed AFib developed in 462 of 2,536 patients (18.2% total, 8.7% per patient-year).
In the NEJM article, the authors wrote that the findings of this trial – the low incidence of stroke that was not further reduced by treatment with edoxaban – may make it appropriate to withhold anticoagulant therapy for patients with atrial high-rate episodes.
The main difference between the population studied in this trial and patients with AFib, as documented on an ECG, appears to be the paucity and brevity of atrial arrhythmias in patients with atrial high-rate episodes (termed low arrhythmia burden). Published reports show that a low arrhythmia burden contributes to a low incidence of stroke among patients with AFib, the study authors wrote.
They added that the low rate of stroke in this trial suggests that in addition to clinical risk prediction formulas for stroke, methods to improve the estimation of stroke risk among patients with infrequent atrial arrhythmias detected by long-term monitoring are needed to guide decision-making on the use of anticoagulation.
Commenting on the NOAH-AFNET 6 results, the comoderator of the ESC HOTLINE session at which they were presented, Barbara Casadei, MD, John Radcliffe Hospital, Oxford, England, said: “Finally we know what to with these patients. Before we just had a variety of opinions with no evidence. I think that the trial really highlights that patients who come to the doctor with symptoms of AF[ib] or who have ECG-documented AF[ib] have a much higher risk of cardioembolic stroke than patients in whom this presumed AF[ib] is picked up incidentally from implanted devices.”
She added: “The stroke rates are very low in this trial, so anticoagulation was never going to work. But this is an important finding. We know that anticoagulants are not a free lunch. There is a significant bleeding risk. These results suggest that unless a patient has clinical AF[ib] that shows up on an ECG then we need to more cautious in prescribing anticoagulation.”
Also commenting on the study, immediate past president of the American College of Cardiology Ed Fry, MD, Ascension Indiana St. Vincent Heart Center, Indianapolis, said the management of patients with implanted cardiac devices or personal wearable technology that has picked up an abnormal rhythm suggestive of AFib was a big question in clinical practice.
“These episodes could be AF[ib], which comes with an increased stroke risk, but it could also be something else like atrial tachycardia or supraventricular tachycardia, which do not confer an increased stroke risk,” he explained.
“This study shows that without a firm diagnosis of AF[ib] on an ECG or some sort of continuous AF[ib] monitoring device, we are going to be anticoagulating people who don’t need it. They were exposed to the risk of bleeding without getting the benefit of a reduction in stroke risk,” Dr. Fry noted.
“The important outcome from this trial is that it gives comfort in we can be more confident in withholding anticoagulation until we get a firm diagnosis of AF[ib]. If we have a high index of suspicion that this could be AF[ib], then we can arrange for a further testing,” he added.
Second trial reporting soon
A trial similar to NOAH-AFNET 6 is currently underway – the ARTESIA trial, which is expected to be reported later in 2023.
“We are in close contact with the leadership of that trial, and we hope to do some meta-analysis,” Dr. Kirchhof said. “But I think today we’ve gone from no evidence to one outcome-based trial which shows there is no reason to use anticoagulation in these patients with atrial high-rate episodes. I think this is reason to change practice now, but yes, of course we need to look at the data in totality once the second trial has reported.”
But the lead investigator of the ARTESIA trial, Stuart Connolly, MD, McMaster University, Hamilton, Ont., does not believe the NOAH-AFNET 6 trial should change practice at this time.
“This trial fails to adequately address the critical issue that drives clinical decision-making in these patients because it is underpowered for the most important endpoint of stroke,” he said in an interview.
“The key question is whether anticoagulation reduces stroke in these patients,” he added. “To answer that, a clinical trial needs to have a lot of strokes, and this trial had very few. The trial was stopped early and had way too few strokes to properly answer this key question.”
The NOAH-AFNET 6 trial was an investigator-initiated trial funded by the German Center for Cardiovascular Research and Daiichi Sankyo Europe. Dr. Kirchhof reported research support from several drug and device companies active in AFib. He is also listed as an inventor on two patents held by the University of Hamburg on AFib therapy and AFib markers.
A version of this article first appeared on Medscape.com.
AMSTERDAM – Among patients with atrial high-rate episodes detected by implantable devices, anticoagulation with edoxaban did not significantly reduce the incidence of a composite outcome of cardiovascular death, stroke, or systemic embolism in comparison with placebo but was associated with a higher bleeding risk in the NOAH-AFNET 6 trial.
“ They do not need to be anticoagulated. That is a relief,” the lead investigator of the trial, Paulus Kirchhof, MD, University Heart and Vascular Center Hamburg (Germany), said in an interview.
Dr. Kirchhof pointed out that this result was unexpected. “Many of us thought that because atrial high-rate episodes look like AF[ib] when they occur, then they are an indication for anticoagulation. But based on these results from the first-ever randomized trial on this population, there is no need for anticoagulation in these patients.”
Dr. Kirchhof presented the NOAH-AFNET 6 trial at the annual congress of the European Society of Cardiology. The study was also simultaneously published in the New England Journal of Medicine.
The trial recruited patients with implanted devices that enable continuous monitoring of atrial rhythm, such as pacemakers and defibrillators. “Because we can record the rhythm day and night with these devices, they pick up small abnormalities. About 20% of these patients experience these occasional atrial high-rate episodes – short episodes that look like AF[ib], but they are rare and brief,” Dr. Kirchhof noted.
He explained that whether the occurrence of these atrial high-rate episodes in patients without AFib, as documented on a conventional electrocardiogram, justifies the initiation of anticoagulants has been unclear. “But this trial tells us that these episodes are different to AF[ib] that is diagnosed on ECG,” he added.
Another finding in the trial was that among these patients, there was an unexpectedly low rate of stroke, despite the patients’ having a CHADSVASC score of 4.
“Based on the result of this trial, these occasional atrial high-rate episodes do not appear to be associated with stroke. It appears quite benign,” Dr. Kirchhof said.
Implications for wearable technology?
He said the results may also have implications for wearable devices that pick up abnormal heart rhythm, such as smartwatches.
“We don’t know exactly what these wearable technologies are picking up, but most likely it is these atrial high-rate episodes. But we need more research on the value of these wearable technologies; we need randomized trials in this particular patient population before we consider anticoagulation in these patients,” Dr. Kirchhof stated.
The NOAH-AFNET 6 study was an event-driven, double-blind, double-dummy, randomized trial involving 2,536 patients aged 65 years or older who had atrial high-rate episodes that lasted for at least 6 minutes and who had at least one additional risk factor for stroke.
Patients were randomly assigned in a 1:1 ratio to receive edoxaban or placebo. The primary efficacy outcome was a composite of cardiovascular death, stroke, or systemic embolism, evaluated in a time-to-event analysis. The safety outcome was a composite of death from any cause or major bleeding.
The mean age of the patients was 78 years, 37.4% were women, and the median duration of atrial high-rate episodes was 2.8 hours. The trial was terminated early, at a median follow-up of 21 months, on the basis of safety concerns and the results of an independent, informal assessment of futility for the efficacy of edoxaban; at termination, the planned enrollment had been completed.
Results showed that a primary efficacy outcome event occurred in 83 patients (3.2% per patient-year) in the edoxaban group and in 101 patients (4.0% per patient-year) in the placebo group (hazard ratio, 0.81; 95% confidence interval, 0.60-1.08; P = .15). The incidence of stroke was approximately 1% per patient-year in both groups.
A safety outcome event occurred in 149 patients (5.9% per patient-year) in the edoxaban group and in 114 patients (4.5% per patient-year) in the placebo group (HR, 1.31; 95% CI, 1.02-1.67; P = .03).
ECG-diagnosed AFib developed in 462 of 2,536 patients (18.2% total, 8.7% per patient-year).
In the NEJM article, the authors wrote that the findings of this trial – the low incidence of stroke that was not further reduced by treatment with edoxaban – may make it appropriate to withhold anticoagulant therapy for patients with atrial high-rate episodes.
The main difference between the population studied in this trial and patients with AFib, as documented on an ECG, appears to be the paucity and brevity of atrial arrhythmias in patients with atrial high-rate episodes (termed low arrhythmia burden). Published reports show that a low arrhythmia burden contributes to a low incidence of stroke among patients with AFib, the study authors wrote.
They added that the low rate of stroke in this trial suggests that in addition to clinical risk prediction formulas for stroke, methods to improve the estimation of stroke risk among patients with infrequent atrial arrhythmias detected by long-term monitoring are needed to guide decision-making on the use of anticoagulation.
Commenting on the NOAH-AFNET 6 results, the comoderator of the ESC HOTLINE session at which they were presented, Barbara Casadei, MD, John Radcliffe Hospital, Oxford, England, said: “Finally we know what to with these patients. Before we just had a variety of opinions with no evidence. I think that the trial really highlights that patients who come to the doctor with symptoms of AF[ib] or who have ECG-documented AF[ib] have a much higher risk of cardioembolic stroke than patients in whom this presumed AF[ib] is picked up incidentally from implanted devices.”
She added: “The stroke rates are very low in this trial, so anticoagulation was never going to work. But this is an important finding. We know that anticoagulants are not a free lunch. There is a significant bleeding risk. These results suggest that unless a patient has clinical AF[ib] that shows up on an ECG then we need to more cautious in prescribing anticoagulation.”
Also commenting on the study, immediate past president of the American College of Cardiology Ed Fry, MD, Ascension Indiana St. Vincent Heart Center, Indianapolis, said the management of patients with implanted cardiac devices or personal wearable technology that has picked up an abnormal rhythm suggestive of AFib was a big question in clinical practice.
“These episodes could be AF[ib], which comes with an increased stroke risk, but it could also be something else like atrial tachycardia or supraventricular tachycardia, which do not confer an increased stroke risk,” he explained.
“This study shows that without a firm diagnosis of AF[ib] on an ECG or some sort of continuous AF[ib] monitoring device, we are going to be anticoagulating people who don’t need it. They were exposed to the risk of bleeding without getting the benefit of a reduction in stroke risk,” Dr. Fry noted.
“The important outcome from this trial is that it gives comfort in we can be more confident in withholding anticoagulation until we get a firm diagnosis of AF[ib]. If we have a high index of suspicion that this could be AF[ib], then we can arrange for a further testing,” he added.
Second trial reporting soon
A trial similar to NOAH-AFNET 6 is currently underway – the ARTESIA trial, which is expected to be reported later in 2023.
“We are in close contact with the leadership of that trial, and we hope to do some meta-analysis,” Dr. Kirchhof said. “But I think today we’ve gone from no evidence to one outcome-based trial which shows there is no reason to use anticoagulation in these patients with atrial high-rate episodes. I think this is reason to change practice now, but yes, of course we need to look at the data in totality once the second trial has reported.”
But the lead investigator of the ARTESIA trial, Stuart Connolly, MD, McMaster University, Hamilton, Ont., does not believe the NOAH-AFNET 6 trial should change practice at this time.
“This trial fails to adequately address the critical issue that drives clinical decision-making in these patients because it is underpowered for the most important endpoint of stroke,” he said in an interview.
“The key question is whether anticoagulation reduces stroke in these patients,” he added. “To answer that, a clinical trial needs to have a lot of strokes, and this trial had very few. The trial was stopped early and had way too few strokes to properly answer this key question.”
The NOAH-AFNET 6 trial was an investigator-initiated trial funded by the German Center for Cardiovascular Research and Daiichi Sankyo Europe. Dr. Kirchhof reported research support from several drug and device companies active in AFib. He is also listed as an inventor on two patents held by the University of Hamburg on AFib therapy and AFib markers.
A version of this article first appeared on Medscape.com.
AT THE ESC CONGRESS 2023
ESC backs SGLT2 inhibitor plus GLP-1 in diabetes with high CVD risk
AMSTERDAM – The era of guidelines that recommended treatment with either a sodium-glucose cotransporter-2 (SGLT-2) inhibitor or a glucagonlike peptide-1 (GLP-1) receptor agonist in people with type 2 diabetes mellitus and established cardiovascular disease (CVD) ended with new recommendations from the European Society of Cardiology that call for starting both classes simultaneously.
said Darren K. McGuire, MD, at the annual congress of the European Society of Cardiology.
The society’s new guidelines for managing CVD in patients with diabetes, released on Aug. 25 and presented in several sessions at the Congress, also break with the past by calling for starting treatment with both an SGLT-2 inhibitor and a GLP-1 receptor agonist without regard to a person’s existing level of glucose control, including their current and target hemoglobin A1c levels, and regardless of background therapy, added Dr. McGuire, a cardiologist and professor at the UT Southwestern Medical Center in Dallas and a member of the ESC panel that wrote the new guidelines.
Instead, the new guidance calls for starting both drug classes promptly in people diagnosed with type 2 diabetes and established atherosclerotic CVD.
Both the previous ESC guidelines from 2019 as well as the current Standards of Care for 2023 document from the American Diabetes Association call for using one class or the other, but they hedge on combined treatment as discretionary.
Different mechanisms mean additive benefits
“With increasing numbers of patients with type 2 diabetes in trials for SGLT-2 inhibitors or GLP-1 receptor agonists who were also on the other drug class, we’ve done large, stratified analyses that suggest no treatment-effect modification” when people received agents from both drug classes, Dr. McGuire explained in an interview. “While we don’t understand the mechanisms of action of these drugs for CVD, we’ve become very confident that they use different mechanisms” that appear to have at least partially additive effects.
“Their benefits for CVD risk reduction are completely independent of their glucose effects. They are cardiology drugs,” Dr. McGuire added.
The new ESC guidelines highlight two other clinical settings where people with type 2 diabetes should receive an SGLT-2 inhibitor regardless of their existing level of glucose control and any other medical treatment: people with heart failure and people with chronic kidney disease (CKD) based on a depressed estimated glomerular filtration rate and an elevated urine albumin-to-creatinine ratio.
Nephropathy was considered by the ESC’s guideline panel to confer risk that is similar to that of established atherosclerotic CVD, Dr. McGuire said.
The guidelines also, for the first time for ESC recommendations, made treatment with finerenone (Kerendia, Bayer) a class 1 level A recommendation for people with type 2 diabetes and CKD.
SCORE2-Diabetes risk estimator
Another major change in the new ESC guideline revision is introduction of a CVD risk calculator intended to estimate the risk among people with type 2 diabetes but without established CVD, heart failure, or CKD.
Called the SCORE2-Diabetes risk estimator, it calculates a person’s 10-year risk for CVD and includes adjustment based on the European region where a person lives; it also tallies different risk levels for women and for men.
The researchers who developed the SCORE2-Diabetes calculator used data from nearly 230,000 people to devise the tool and then validated it with data from an additional 217,000 Europeans with type 2 diabetes.
Key features of the calculator include its use of routinely collected clinical values, such as age, sex, systolic blood pressure, smoking status, serum cholesterol levels, age at diabetes diagnosis, hemoglobin A1c level, and estimated glomerular filtration rate.
“For the first time we have a clear score to categorize risk” in people with type 2 diabetes and identify who needs more aggressive treatment to prevent CVD development,” said Emanuele Di Angelantonio, MD, PhD, deputy director of the cardiovascular epidemiology unit at the University of Cambridge (England).
The guidelines say that people who have a low (< 5%) or moderate (5%-9%) 10-year risk for CVD are possible candidates for metformin treatment. Those with high (10%-19%) or very high (≥ 20%) risk are possible candidates for treatment with metformin and/or an SGLT-2 inhibitor and/or a GLP-1 receptor agonist, said Dr. Di Angelantonio during his talk at the congress on the new risk score.
“The risk score is a good addition” because it estimates future CVD risk better and more systematically than usual practice, which generally relies on no systematic tool, said Naveed Sattar, PhD, professor of metabolic medicine at the University of Glasgow (Scotland) and also a member of the guideline-writing panel.
The new risk score “is a reasonable way” to identify people without CVD but at elevated risk who might benefit from treatment with a relatively expensive drug, such as an SGLT-2 inhibitor, Dr. Sattar said in an interview. “It doesn’t rely on any fancy biomarkers or imaging, and it takes about 30 seconds to calculate. It’s not perfect, but it gets the job done,” and it will increase the number of people with type 2 diabetes who will receive an SGLT-2 inhibitor, he predicted.
Dr. McGuire has been a consultant to Altimmune, Applied Therapeutics, AstraZeneca, Bayer, Boehringer-Ingelheim, Intercept, Lexion, Lilly, Merck, New Amsterdam, and Pfizer. Dr. Di Angelantonio had no disclosures. Dr. Sattar has been a consultant to Abbott, Amgen, AstraZeneca, Boehringer Ingelheim, Lilly, Novartis, Novo Nordisk, Pfizer, and Roche Diagnostics.
A version of this article appeared on Medscape.com.
AMSTERDAM – The era of guidelines that recommended treatment with either a sodium-glucose cotransporter-2 (SGLT-2) inhibitor or a glucagonlike peptide-1 (GLP-1) receptor agonist in people with type 2 diabetes mellitus and established cardiovascular disease (CVD) ended with new recommendations from the European Society of Cardiology that call for starting both classes simultaneously.
said Darren K. McGuire, MD, at the annual congress of the European Society of Cardiology.
The society’s new guidelines for managing CVD in patients with diabetes, released on Aug. 25 and presented in several sessions at the Congress, also break with the past by calling for starting treatment with both an SGLT-2 inhibitor and a GLP-1 receptor agonist without regard to a person’s existing level of glucose control, including their current and target hemoglobin A1c levels, and regardless of background therapy, added Dr. McGuire, a cardiologist and professor at the UT Southwestern Medical Center in Dallas and a member of the ESC panel that wrote the new guidelines.
Instead, the new guidance calls for starting both drug classes promptly in people diagnosed with type 2 diabetes and established atherosclerotic CVD.
Both the previous ESC guidelines from 2019 as well as the current Standards of Care for 2023 document from the American Diabetes Association call for using one class or the other, but they hedge on combined treatment as discretionary.
Different mechanisms mean additive benefits
“With increasing numbers of patients with type 2 diabetes in trials for SGLT-2 inhibitors or GLP-1 receptor agonists who were also on the other drug class, we’ve done large, stratified analyses that suggest no treatment-effect modification” when people received agents from both drug classes, Dr. McGuire explained in an interview. “While we don’t understand the mechanisms of action of these drugs for CVD, we’ve become very confident that they use different mechanisms” that appear to have at least partially additive effects.
“Their benefits for CVD risk reduction are completely independent of their glucose effects. They are cardiology drugs,” Dr. McGuire added.
The new ESC guidelines highlight two other clinical settings where people with type 2 diabetes should receive an SGLT-2 inhibitor regardless of their existing level of glucose control and any other medical treatment: people with heart failure and people with chronic kidney disease (CKD) based on a depressed estimated glomerular filtration rate and an elevated urine albumin-to-creatinine ratio.
Nephropathy was considered by the ESC’s guideline panel to confer risk that is similar to that of established atherosclerotic CVD, Dr. McGuire said.
The guidelines also, for the first time for ESC recommendations, made treatment with finerenone (Kerendia, Bayer) a class 1 level A recommendation for people with type 2 diabetes and CKD.
SCORE2-Diabetes risk estimator
Another major change in the new ESC guideline revision is introduction of a CVD risk calculator intended to estimate the risk among people with type 2 diabetes but without established CVD, heart failure, or CKD.
Called the SCORE2-Diabetes risk estimator, it calculates a person’s 10-year risk for CVD and includes adjustment based on the European region where a person lives; it also tallies different risk levels for women and for men.
The researchers who developed the SCORE2-Diabetes calculator used data from nearly 230,000 people to devise the tool and then validated it with data from an additional 217,000 Europeans with type 2 diabetes.
Key features of the calculator include its use of routinely collected clinical values, such as age, sex, systolic blood pressure, smoking status, serum cholesterol levels, age at diabetes diagnosis, hemoglobin A1c level, and estimated glomerular filtration rate.
“For the first time we have a clear score to categorize risk” in people with type 2 diabetes and identify who needs more aggressive treatment to prevent CVD development,” said Emanuele Di Angelantonio, MD, PhD, deputy director of the cardiovascular epidemiology unit at the University of Cambridge (England).
The guidelines say that people who have a low (< 5%) or moderate (5%-9%) 10-year risk for CVD are possible candidates for metformin treatment. Those with high (10%-19%) or very high (≥ 20%) risk are possible candidates for treatment with metformin and/or an SGLT-2 inhibitor and/or a GLP-1 receptor agonist, said Dr. Di Angelantonio during his talk at the congress on the new risk score.
“The risk score is a good addition” because it estimates future CVD risk better and more systematically than usual practice, which generally relies on no systematic tool, said Naveed Sattar, PhD, professor of metabolic medicine at the University of Glasgow (Scotland) and also a member of the guideline-writing panel.
The new risk score “is a reasonable way” to identify people without CVD but at elevated risk who might benefit from treatment with a relatively expensive drug, such as an SGLT-2 inhibitor, Dr. Sattar said in an interview. “It doesn’t rely on any fancy biomarkers or imaging, and it takes about 30 seconds to calculate. It’s not perfect, but it gets the job done,” and it will increase the number of people with type 2 diabetes who will receive an SGLT-2 inhibitor, he predicted.
Dr. McGuire has been a consultant to Altimmune, Applied Therapeutics, AstraZeneca, Bayer, Boehringer-Ingelheim, Intercept, Lexion, Lilly, Merck, New Amsterdam, and Pfizer. Dr. Di Angelantonio had no disclosures. Dr. Sattar has been a consultant to Abbott, Amgen, AstraZeneca, Boehringer Ingelheim, Lilly, Novartis, Novo Nordisk, Pfizer, and Roche Diagnostics.
A version of this article appeared on Medscape.com.
AMSTERDAM – The era of guidelines that recommended treatment with either a sodium-glucose cotransporter-2 (SGLT-2) inhibitor or a glucagonlike peptide-1 (GLP-1) receptor agonist in people with type 2 diabetes mellitus and established cardiovascular disease (CVD) ended with new recommendations from the European Society of Cardiology that call for starting both classes simultaneously.
said Darren K. McGuire, MD, at the annual congress of the European Society of Cardiology.
The society’s new guidelines for managing CVD in patients with diabetes, released on Aug. 25 and presented in several sessions at the Congress, also break with the past by calling for starting treatment with both an SGLT-2 inhibitor and a GLP-1 receptor agonist without regard to a person’s existing level of glucose control, including their current and target hemoglobin A1c levels, and regardless of background therapy, added Dr. McGuire, a cardiologist and professor at the UT Southwestern Medical Center in Dallas and a member of the ESC panel that wrote the new guidelines.
Instead, the new guidance calls for starting both drug classes promptly in people diagnosed with type 2 diabetes and established atherosclerotic CVD.
Both the previous ESC guidelines from 2019 as well as the current Standards of Care for 2023 document from the American Diabetes Association call for using one class or the other, but they hedge on combined treatment as discretionary.
Different mechanisms mean additive benefits
“With increasing numbers of patients with type 2 diabetes in trials for SGLT-2 inhibitors or GLP-1 receptor agonists who were also on the other drug class, we’ve done large, stratified analyses that suggest no treatment-effect modification” when people received agents from both drug classes, Dr. McGuire explained in an interview. “While we don’t understand the mechanisms of action of these drugs for CVD, we’ve become very confident that they use different mechanisms” that appear to have at least partially additive effects.
“Their benefits for CVD risk reduction are completely independent of their glucose effects. They are cardiology drugs,” Dr. McGuire added.
The new ESC guidelines highlight two other clinical settings where people with type 2 diabetes should receive an SGLT-2 inhibitor regardless of their existing level of glucose control and any other medical treatment: people with heart failure and people with chronic kidney disease (CKD) based on a depressed estimated glomerular filtration rate and an elevated urine albumin-to-creatinine ratio.
Nephropathy was considered by the ESC’s guideline panel to confer risk that is similar to that of established atherosclerotic CVD, Dr. McGuire said.
The guidelines also, for the first time for ESC recommendations, made treatment with finerenone (Kerendia, Bayer) a class 1 level A recommendation for people with type 2 diabetes and CKD.
SCORE2-Diabetes risk estimator
Another major change in the new ESC guideline revision is introduction of a CVD risk calculator intended to estimate the risk among people with type 2 diabetes but without established CVD, heart failure, or CKD.
Called the SCORE2-Diabetes risk estimator, it calculates a person’s 10-year risk for CVD and includes adjustment based on the European region where a person lives; it also tallies different risk levels for women and for men.
The researchers who developed the SCORE2-Diabetes calculator used data from nearly 230,000 people to devise the tool and then validated it with data from an additional 217,000 Europeans with type 2 diabetes.
Key features of the calculator include its use of routinely collected clinical values, such as age, sex, systolic blood pressure, smoking status, serum cholesterol levels, age at diabetes diagnosis, hemoglobin A1c level, and estimated glomerular filtration rate.
“For the first time we have a clear score to categorize risk” in people with type 2 diabetes and identify who needs more aggressive treatment to prevent CVD development,” said Emanuele Di Angelantonio, MD, PhD, deputy director of the cardiovascular epidemiology unit at the University of Cambridge (England).
The guidelines say that people who have a low (< 5%) or moderate (5%-9%) 10-year risk for CVD are possible candidates for metformin treatment. Those with high (10%-19%) or very high (≥ 20%) risk are possible candidates for treatment with metformin and/or an SGLT-2 inhibitor and/or a GLP-1 receptor agonist, said Dr. Di Angelantonio during his talk at the congress on the new risk score.
“The risk score is a good addition” because it estimates future CVD risk better and more systematically than usual practice, which generally relies on no systematic tool, said Naveed Sattar, PhD, professor of metabolic medicine at the University of Glasgow (Scotland) and also a member of the guideline-writing panel.
The new risk score “is a reasonable way” to identify people without CVD but at elevated risk who might benefit from treatment with a relatively expensive drug, such as an SGLT-2 inhibitor, Dr. Sattar said in an interview. “It doesn’t rely on any fancy biomarkers or imaging, and it takes about 30 seconds to calculate. It’s not perfect, but it gets the job done,” and it will increase the number of people with type 2 diabetes who will receive an SGLT-2 inhibitor, he predicted.
Dr. McGuire has been a consultant to Altimmune, Applied Therapeutics, AstraZeneca, Bayer, Boehringer-Ingelheim, Intercept, Lexion, Lilly, Merck, New Amsterdam, and Pfizer. Dr. Di Angelantonio had no disclosures. Dr. Sattar has been a consultant to Abbott, Amgen, AstraZeneca, Boehringer Ingelheim, Lilly, Novartis, Novo Nordisk, Pfizer, and Roche Diagnostics.
A version of this article appeared on Medscape.com.
AT ESC CONGRESS 2023
Number of people with long COVID could be vastly underestimated
It’s been estimated that up to one-third of people who survive acute SARS-CoV-2 infection will suffer a post-viral syndrome with lingering neurologic and other symptoms – now known as long COVID or neurological postacute sequelae of SARS-CoV-2 infection (Neuro-PASC).
However, Researchers found a significant proportion of patients in their small study who had never tested positive for COVID-19 but who were having symptoms of long COVID nevertheless showed evidence of immune responses consistent with previous exposure.
“We estimate that millions of people got COVID in the U.S. during the first year of the pandemic and then developed long COVID, yet they did not get a positive COVID diagnosis because of testing limitations,” Igor J. Koralnik, MD, of Northwestern Medicine Comprehensive COVID-19 Center in Chicago, said in an interview.
He noted that many post-COVID-19 clinics in the United States don’t accept people with long COVID symptoms who do not have a positive test result.
Patients with long COVID symptoms but without laboratory evidence of prior infection, “who have often been rejected and stigmatized, should feel vindicated by the results of our study,” Dr. Koralnik said.
“We think that those patients deserve the same clinical care as those with a positive test, as well as inclusion in research studies. This is what we are doing at Northwestern Medicine’s Comprehensive COVID[-19] Center,” Dr. Koralnik added.
The study was published online in the journal Neurology: Neuroimmunology & Neuroinflammation.
Delayed care
The researchers measured SARS-CoV-2-specific humoral and cell-mediated immune responses against nucleocapsid protein and spike proteins, which indicate a prior COVID-19 infection, in 29 patients with post-viral syndrome after suspected COVID-19, including neurologic symptoms such as cognitive impairment, headache, and fatigue, but who did not have a confirmed positive COVID-19 test.
They did the same in 32 age- and sex-matched COVID long haulers with confirmed Neuro-PASC and 18 healthy controls with none of the symptoms of long COVID and no known exposure to SARS-CoV-2 or positive test result.
They found that 12 of the 29 patients (41%) with post-viral syndrome (but no positive COVID-19 test) had detectable humoral and cellular immune responses consistent with prior exposure to SARS-CoV-2. Three-quarters harbored antinucleocapsid and 50% harbored antispike responses.
“Our data suggest that at least 4 million people with post-viral syndrome similar to long COVID may indeed have detectable immune responses to support a COVID diagnosis,” Dr. Koralnik said in a news release.
The 12 patients with post-viral syndrome but without a confirmed COVID-19 test had neurologic symptoms similar to those of patients with confirmed Neuro-PASC.
However, lack of a confirmed COVID-19 diagnosis likely contributed to the 5-month delay in the median time from symptom onset to clinic visit, the researchers said. They were evaluated at a median of 10.7 months vs. 5.4 months for Neuro-PASC patients.
Dr. Koralnik said in an interview that the “most important take-home message” of the study is that patients with post-viral syndrome often present with clinical manifestations similar to those of confirmed patients with Neuro-PASC, suggesting that a positive result by commercially available SARS-CoV-2 diagnostic test should not be a prerequisite for accessing care.
Patients with post-viral syndrome may benefit from the same clinical care as confirmed patients with Neuro-PASC, and the absence of a positive SARS-CoV-2 test should not preclude or delay treatment, he added.
A version of this article first appeared on Medscape.com .
This article was updated 8/28/23.
It’s been estimated that up to one-third of people who survive acute SARS-CoV-2 infection will suffer a post-viral syndrome with lingering neurologic and other symptoms – now known as long COVID or neurological postacute sequelae of SARS-CoV-2 infection (Neuro-PASC).
However, Researchers found a significant proportion of patients in their small study who had never tested positive for COVID-19 but who were having symptoms of long COVID nevertheless showed evidence of immune responses consistent with previous exposure.
“We estimate that millions of people got COVID in the U.S. during the first year of the pandemic and then developed long COVID, yet they did not get a positive COVID diagnosis because of testing limitations,” Igor J. Koralnik, MD, of Northwestern Medicine Comprehensive COVID-19 Center in Chicago, said in an interview.
He noted that many post-COVID-19 clinics in the United States don’t accept people with long COVID symptoms who do not have a positive test result.
Patients with long COVID symptoms but without laboratory evidence of prior infection, “who have often been rejected and stigmatized, should feel vindicated by the results of our study,” Dr. Koralnik said.
“We think that those patients deserve the same clinical care as those with a positive test, as well as inclusion in research studies. This is what we are doing at Northwestern Medicine’s Comprehensive COVID[-19] Center,” Dr. Koralnik added.
The study was published online in the journal Neurology: Neuroimmunology & Neuroinflammation.
Delayed care
The researchers measured SARS-CoV-2-specific humoral and cell-mediated immune responses against nucleocapsid protein and spike proteins, which indicate a prior COVID-19 infection, in 29 patients with post-viral syndrome after suspected COVID-19, including neurologic symptoms such as cognitive impairment, headache, and fatigue, but who did not have a confirmed positive COVID-19 test.
They did the same in 32 age- and sex-matched COVID long haulers with confirmed Neuro-PASC and 18 healthy controls with none of the symptoms of long COVID and no known exposure to SARS-CoV-2 or positive test result.
They found that 12 of the 29 patients (41%) with post-viral syndrome (but no positive COVID-19 test) had detectable humoral and cellular immune responses consistent with prior exposure to SARS-CoV-2. Three-quarters harbored antinucleocapsid and 50% harbored antispike responses.
“Our data suggest that at least 4 million people with post-viral syndrome similar to long COVID may indeed have detectable immune responses to support a COVID diagnosis,” Dr. Koralnik said in a news release.
The 12 patients with post-viral syndrome but without a confirmed COVID-19 test had neurologic symptoms similar to those of patients with confirmed Neuro-PASC.
However, lack of a confirmed COVID-19 diagnosis likely contributed to the 5-month delay in the median time from symptom onset to clinic visit, the researchers said. They were evaluated at a median of 10.7 months vs. 5.4 months for Neuro-PASC patients.
Dr. Koralnik said in an interview that the “most important take-home message” of the study is that patients with post-viral syndrome often present with clinical manifestations similar to those of confirmed patients with Neuro-PASC, suggesting that a positive result by commercially available SARS-CoV-2 diagnostic test should not be a prerequisite for accessing care.
Patients with post-viral syndrome may benefit from the same clinical care as confirmed patients with Neuro-PASC, and the absence of a positive SARS-CoV-2 test should not preclude or delay treatment, he added.
A version of this article first appeared on Medscape.com .
This article was updated 8/28/23.
It’s been estimated that up to one-third of people who survive acute SARS-CoV-2 infection will suffer a post-viral syndrome with lingering neurologic and other symptoms – now known as long COVID or neurological postacute sequelae of SARS-CoV-2 infection (Neuro-PASC).
However, Researchers found a significant proportion of patients in their small study who had never tested positive for COVID-19 but who were having symptoms of long COVID nevertheless showed evidence of immune responses consistent with previous exposure.
“We estimate that millions of people got COVID in the U.S. during the first year of the pandemic and then developed long COVID, yet they did not get a positive COVID diagnosis because of testing limitations,” Igor J. Koralnik, MD, of Northwestern Medicine Comprehensive COVID-19 Center in Chicago, said in an interview.
He noted that many post-COVID-19 clinics in the United States don’t accept people with long COVID symptoms who do not have a positive test result.
Patients with long COVID symptoms but without laboratory evidence of prior infection, “who have often been rejected and stigmatized, should feel vindicated by the results of our study,” Dr. Koralnik said.
“We think that those patients deserve the same clinical care as those with a positive test, as well as inclusion in research studies. This is what we are doing at Northwestern Medicine’s Comprehensive COVID[-19] Center,” Dr. Koralnik added.
The study was published online in the journal Neurology: Neuroimmunology & Neuroinflammation.
Delayed care
The researchers measured SARS-CoV-2-specific humoral and cell-mediated immune responses against nucleocapsid protein and spike proteins, which indicate a prior COVID-19 infection, in 29 patients with post-viral syndrome after suspected COVID-19, including neurologic symptoms such as cognitive impairment, headache, and fatigue, but who did not have a confirmed positive COVID-19 test.
They did the same in 32 age- and sex-matched COVID long haulers with confirmed Neuro-PASC and 18 healthy controls with none of the symptoms of long COVID and no known exposure to SARS-CoV-2 or positive test result.
They found that 12 of the 29 patients (41%) with post-viral syndrome (but no positive COVID-19 test) had detectable humoral and cellular immune responses consistent with prior exposure to SARS-CoV-2. Three-quarters harbored antinucleocapsid and 50% harbored antispike responses.
“Our data suggest that at least 4 million people with post-viral syndrome similar to long COVID may indeed have detectable immune responses to support a COVID diagnosis,” Dr. Koralnik said in a news release.
The 12 patients with post-viral syndrome but without a confirmed COVID-19 test had neurologic symptoms similar to those of patients with confirmed Neuro-PASC.
However, lack of a confirmed COVID-19 diagnosis likely contributed to the 5-month delay in the median time from symptom onset to clinic visit, the researchers said. They were evaluated at a median of 10.7 months vs. 5.4 months for Neuro-PASC patients.
Dr. Koralnik said in an interview that the “most important take-home message” of the study is that patients with post-viral syndrome often present with clinical manifestations similar to those of confirmed patients with Neuro-PASC, suggesting that a positive result by commercially available SARS-CoV-2 diagnostic test should not be a prerequisite for accessing care.
Patients with post-viral syndrome may benefit from the same clinical care as confirmed patients with Neuro-PASC, and the absence of a positive SARS-CoV-2 test should not preclude or delay treatment, he added.
A version of this article first appeared on Medscape.com .
This article was updated 8/28/23.
FROM NEUROLOGY, NEUROIMMUNOLOGY & NEUROINFLAMMATION
Liraglutide fixes learning limit tied to insulin resistance
A single injection of the GLP-1 receptor agonist liraglutide led to short-term normalization of associative learning in people with obesity and insulin resistance, a finding that suggests say the authors of a recent report in Nature Metabolism.
“We demonstrated that dopamine-driven associative learning about external sensory cues crucially depends on metabolic signaling,” said Marc Tittgemeyer, PhD, professor at the Max Planck Institute for Metabolism Research in Cologne, Germany, and senior author of the study. Study participants with impaired insulin sensitivity “exhibited a reduced amplitude of behavioral updating that was normalized” by a single subcutaneous injection of 0.6 mg of liraglutide (the starting daily dose for liraglutide for weight loss, available as Saxenda, Novo Nordisk) given the evening before testing.
The findings, from 30 adults with normal insulin sensitivity and normal weight and 24 adults with impaired insulin sensitivity and obesity, suggest that metabolic signals, particularly ones that promote energy restoration in a setting of energy deprivation caused by insulin or a glucagon-like peptide-1 (GLP-1) receptor agonist, “profoundly influence neuronal processing,” said Dr. Tittgemeyer. The findings suggest that impaired metabolic signaling such as occurs with insulin resistance in people with obesity can cause deficiencies in associative learning.
‘Liraglutide can normalize learning of associations’
“We show that in people with obesity, disrupted circuit mechanisms lead to impaired learning about sensory associations,” Dr. Tittgemeyer said in an interview. “The information provided by sensory systems that the brain must interpret to select a behavioral response are ‘off tune’ ” in these individuals.
“This is rather consequential for understanding food-intake behaviors. Modern obesity treatments, such as liraglutide, can normalize learning of associations and thereby render people susceptible again for sensory signals and make them more prone to react to subliminal interactions, such as weight-normalizing diets and conscious eating,” he added.
The normalization in associative learning that one dose of liraglutide produced in people with obesity “fits with studies showing that these drugs restore a normal feeling of satiety, causing people to eat less and therefore lose weight,” he explained.
Dr. Tittgemeyer noted that this effect is likely shared by other agents in the GLP-1 receptor agonist class, such as semaglutide (Ozempic, Wegovy, Novo Nordisk) but is likely not an effect when agents agonize receptors to other nutrient-stimulated hormones such as glucagon and the glucose-dependent insulinotropic polypeptide.
The findings “show that liraglutide restores associative learning in participants with greater insulin resistance,” a “highly relevant” discovery, commented Nils B. Kroemer, PhD, head of the section of medical psychology at the University of Bonn, Germany, who was not involved with this research, in a written statement.
The study run by Dr. Tittgemeyer and his associates included 54 healthy adult volunteers whom they assessed for insulin sensitivity with their homeostasis model assessment of insulin resistance. The researchers divided the cohort into groups; one group included 24 people with impaired insulin sensitivity, and one included 30 with normal insulin sensitivity. The average body mass index (BMI) of the normal sensitivity group was about 24 kg/m2; in the insulin-resistant subgroup, BMI averaged about 33 kg/m2.
The associative learning task tested the ability of participants to learn associations between auditory cues (a high or low tone) and a subsequent visual outcome (a picture of a face or a house). During each associative learning session, participants also underwent functional MRI of the brain.
Liraglutide treatment leveled learning
The results showed that the learning rate was significantly lower in the subgroup with impaired insulin sensitivity, compared with those with normal insulin sensitivity following treatment with a placebo injection. This indicates a decreased adaptation of learning to predictability variations in individuals with impaired insulin sensitivity.
In contrast, treatment with a single dose of liraglutide significantly enhanced the learning rate in the group with impaired insulin sensitivity but significantly reduced the learning rate in the group with normal insulin sensitivity. Liraglutide’s effect was twice as large in the group with impaired insulin sensitivity than in the group with normal insulin sensitivity, and these opposing effects of liraglutide resulted in a convergence of the two groups’ adaptive learning rates so that there wasn’t any significant between-group difference following liraglutide treatment.
After analyzing the functional MRI data along with the learning results, the researchers concluded that liraglutide normalized learning in individuals with impaired insulin sensitivity by enhancing adaptive prediction error encoding in the brain’s ventral striatum and mesocortical projection sites.
This apparent ability of GLP-1 analogues to correct this learning deficit in people with impaired insulin sensitivity and obesity has implications regarding potential benefit for people with other pathologies characterized by impaired dopaminergic function and associated with metabolic impairments, such as psychosis, Parkinson’s disease, and depression, the researchers say.
“The fascinating thing about GLP-1 receptor agonists is that they have an additional mechanism that relates to anti-inflammatory effects, especially for alleviating cell stress,” said Dr. Tittgemeyer. “Many ongoing clinical trials are assessing their effects in neuropsychiatric diseases,” he noted.
The study received no commercial funding. Dr. Tittgemyer and most of his coauthors had no disclosures. One coauthor had several disclosures, which are detailed in the report. Dr. Kroemer had no disclosures.
A version of this article first appeared on Medscape.com.
A single injection of the GLP-1 receptor agonist liraglutide led to short-term normalization of associative learning in people with obesity and insulin resistance, a finding that suggests say the authors of a recent report in Nature Metabolism.
“We demonstrated that dopamine-driven associative learning about external sensory cues crucially depends on metabolic signaling,” said Marc Tittgemeyer, PhD, professor at the Max Planck Institute for Metabolism Research in Cologne, Germany, and senior author of the study. Study participants with impaired insulin sensitivity “exhibited a reduced amplitude of behavioral updating that was normalized” by a single subcutaneous injection of 0.6 mg of liraglutide (the starting daily dose for liraglutide for weight loss, available as Saxenda, Novo Nordisk) given the evening before testing.
The findings, from 30 adults with normal insulin sensitivity and normal weight and 24 adults with impaired insulin sensitivity and obesity, suggest that metabolic signals, particularly ones that promote energy restoration in a setting of energy deprivation caused by insulin or a glucagon-like peptide-1 (GLP-1) receptor agonist, “profoundly influence neuronal processing,” said Dr. Tittgemeyer. The findings suggest that impaired metabolic signaling such as occurs with insulin resistance in people with obesity can cause deficiencies in associative learning.
‘Liraglutide can normalize learning of associations’
“We show that in people with obesity, disrupted circuit mechanisms lead to impaired learning about sensory associations,” Dr. Tittgemeyer said in an interview. “The information provided by sensory systems that the brain must interpret to select a behavioral response are ‘off tune’ ” in these individuals.
“This is rather consequential for understanding food-intake behaviors. Modern obesity treatments, such as liraglutide, can normalize learning of associations and thereby render people susceptible again for sensory signals and make them more prone to react to subliminal interactions, such as weight-normalizing diets and conscious eating,” he added.
The normalization in associative learning that one dose of liraglutide produced in people with obesity “fits with studies showing that these drugs restore a normal feeling of satiety, causing people to eat less and therefore lose weight,” he explained.
Dr. Tittgemeyer noted that this effect is likely shared by other agents in the GLP-1 receptor agonist class, such as semaglutide (Ozempic, Wegovy, Novo Nordisk) but is likely not an effect when agents agonize receptors to other nutrient-stimulated hormones such as glucagon and the glucose-dependent insulinotropic polypeptide.
The findings “show that liraglutide restores associative learning in participants with greater insulin resistance,” a “highly relevant” discovery, commented Nils B. Kroemer, PhD, head of the section of medical psychology at the University of Bonn, Germany, who was not involved with this research, in a written statement.
The study run by Dr. Tittgemeyer and his associates included 54 healthy adult volunteers whom they assessed for insulin sensitivity with their homeostasis model assessment of insulin resistance. The researchers divided the cohort into groups; one group included 24 people with impaired insulin sensitivity, and one included 30 with normal insulin sensitivity. The average body mass index (BMI) of the normal sensitivity group was about 24 kg/m2; in the insulin-resistant subgroup, BMI averaged about 33 kg/m2.
The associative learning task tested the ability of participants to learn associations between auditory cues (a high or low tone) and a subsequent visual outcome (a picture of a face or a house). During each associative learning session, participants also underwent functional MRI of the brain.
Liraglutide treatment leveled learning
The results showed that the learning rate was significantly lower in the subgroup with impaired insulin sensitivity, compared with those with normal insulin sensitivity following treatment with a placebo injection. This indicates a decreased adaptation of learning to predictability variations in individuals with impaired insulin sensitivity.
In contrast, treatment with a single dose of liraglutide significantly enhanced the learning rate in the group with impaired insulin sensitivity but significantly reduced the learning rate in the group with normal insulin sensitivity. Liraglutide’s effect was twice as large in the group with impaired insulin sensitivity than in the group with normal insulin sensitivity, and these opposing effects of liraglutide resulted in a convergence of the two groups’ adaptive learning rates so that there wasn’t any significant between-group difference following liraglutide treatment.
After analyzing the functional MRI data along with the learning results, the researchers concluded that liraglutide normalized learning in individuals with impaired insulin sensitivity by enhancing adaptive prediction error encoding in the brain’s ventral striatum and mesocortical projection sites.
This apparent ability of GLP-1 analogues to correct this learning deficit in people with impaired insulin sensitivity and obesity has implications regarding potential benefit for people with other pathologies characterized by impaired dopaminergic function and associated with metabolic impairments, such as psychosis, Parkinson’s disease, and depression, the researchers say.
“The fascinating thing about GLP-1 receptor agonists is that they have an additional mechanism that relates to anti-inflammatory effects, especially for alleviating cell stress,” said Dr. Tittgemeyer. “Many ongoing clinical trials are assessing their effects in neuropsychiatric diseases,” he noted.
The study received no commercial funding. Dr. Tittgemyer and most of his coauthors had no disclosures. One coauthor had several disclosures, which are detailed in the report. Dr. Kroemer had no disclosures.
A version of this article first appeared on Medscape.com.
A single injection of the GLP-1 receptor agonist liraglutide led to short-term normalization of associative learning in people with obesity and insulin resistance, a finding that suggests say the authors of a recent report in Nature Metabolism.
“We demonstrated that dopamine-driven associative learning about external sensory cues crucially depends on metabolic signaling,” said Marc Tittgemeyer, PhD, professor at the Max Planck Institute for Metabolism Research in Cologne, Germany, and senior author of the study. Study participants with impaired insulin sensitivity “exhibited a reduced amplitude of behavioral updating that was normalized” by a single subcutaneous injection of 0.6 mg of liraglutide (the starting daily dose for liraglutide for weight loss, available as Saxenda, Novo Nordisk) given the evening before testing.
The findings, from 30 adults with normal insulin sensitivity and normal weight and 24 adults with impaired insulin sensitivity and obesity, suggest that metabolic signals, particularly ones that promote energy restoration in a setting of energy deprivation caused by insulin or a glucagon-like peptide-1 (GLP-1) receptor agonist, “profoundly influence neuronal processing,” said Dr. Tittgemeyer. The findings suggest that impaired metabolic signaling such as occurs with insulin resistance in people with obesity can cause deficiencies in associative learning.
‘Liraglutide can normalize learning of associations’
“We show that in people with obesity, disrupted circuit mechanisms lead to impaired learning about sensory associations,” Dr. Tittgemeyer said in an interview. “The information provided by sensory systems that the brain must interpret to select a behavioral response are ‘off tune’ ” in these individuals.
“This is rather consequential for understanding food-intake behaviors. Modern obesity treatments, such as liraglutide, can normalize learning of associations and thereby render people susceptible again for sensory signals and make them more prone to react to subliminal interactions, such as weight-normalizing diets and conscious eating,” he added.
The normalization in associative learning that one dose of liraglutide produced in people with obesity “fits with studies showing that these drugs restore a normal feeling of satiety, causing people to eat less and therefore lose weight,” he explained.
Dr. Tittgemeyer noted that this effect is likely shared by other agents in the GLP-1 receptor agonist class, such as semaglutide (Ozempic, Wegovy, Novo Nordisk) but is likely not an effect when agents agonize receptors to other nutrient-stimulated hormones such as glucagon and the glucose-dependent insulinotropic polypeptide.
The findings “show that liraglutide restores associative learning in participants with greater insulin resistance,” a “highly relevant” discovery, commented Nils B. Kroemer, PhD, head of the section of medical psychology at the University of Bonn, Germany, who was not involved with this research, in a written statement.
The study run by Dr. Tittgemeyer and his associates included 54 healthy adult volunteers whom they assessed for insulin sensitivity with their homeostasis model assessment of insulin resistance. The researchers divided the cohort into groups; one group included 24 people with impaired insulin sensitivity, and one included 30 with normal insulin sensitivity. The average body mass index (BMI) of the normal sensitivity group was about 24 kg/m2; in the insulin-resistant subgroup, BMI averaged about 33 kg/m2.
The associative learning task tested the ability of participants to learn associations between auditory cues (a high or low tone) and a subsequent visual outcome (a picture of a face or a house). During each associative learning session, participants also underwent functional MRI of the brain.
Liraglutide treatment leveled learning
The results showed that the learning rate was significantly lower in the subgroup with impaired insulin sensitivity, compared with those with normal insulin sensitivity following treatment with a placebo injection. This indicates a decreased adaptation of learning to predictability variations in individuals with impaired insulin sensitivity.
In contrast, treatment with a single dose of liraglutide significantly enhanced the learning rate in the group with impaired insulin sensitivity but significantly reduced the learning rate in the group with normal insulin sensitivity. Liraglutide’s effect was twice as large in the group with impaired insulin sensitivity than in the group with normal insulin sensitivity, and these opposing effects of liraglutide resulted in a convergence of the two groups’ adaptive learning rates so that there wasn’t any significant between-group difference following liraglutide treatment.
After analyzing the functional MRI data along with the learning results, the researchers concluded that liraglutide normalized learning in individuals with impaired insulin sensitivity by enhancing adaptive prediction error encoding in the brain’s ventral striatum and mesocortical projection sites.
This apparent ability of GLP-1 analogues to correct this learning deficit in people with impaired insulin sensitivity and obesity has implications regarding potential benefit for people with other pathologies characterized by impaired dopaminergic function and associated with metabolic impairments, such as psychosis, Parkinson’s disease, and depression, the researchers say.
“The fascinating thing about GLP-1 receptor agonists is that they have an additional mechanism that relates to anti-inflammatory effects, especially for alleviating cell stress,” said Dr. Tittgemeyer. “Many ongoing clinical trials are assessing their effects in neuropsychiatric diseases,” he noted.
The study received no commercial funding. Dr. Tittgemyer and most of his coauthors had no disclosures. One coauthor had several disclosures, which are detailed in the report. Dr. Kroemer had no disclosures.
A version of this article first appeared on Medscape.com.
FROM NATURE METABOLISM
Apple cider vinegar, fenugreek best herbal remedies for T2D: Review
The review included 44 randomized clinical trials with more than 3,000 participants using six herbal remedies: apple cider vinegar, cinnamon, curcumin, fenugreek seeds, ginger, and saffron.
Apple cider vinegar, fenugreek seeds, curcumin (turmeric), and cinnamon resulted in statistically significant reductions in fasting blood glucose, compared with the control groups in the clinical trials. Out of all the remedies, the authors found apple cider vinegar to be the most effective for lowering fasting blood glucose levels.
The review also found that apple cider vinegar and fenugreek seeds had a statistically significant effect on reducing A1c, compared with the control groups. The authors found the herbal remedies made no difference to insulin level or homeostatic model assessment for insulin resistance (HOMA-IR).
The results are published online in Diabetes & Metabolic Syndrome: Clinical Research & Reviews. The authors said they hoped the review would help medical professionals and people with type 2 diabetes understand the effectiveness of different herbal remedies and consider incorporating these remedies into standard care.
“Some people use curcumin, some use ginger, some use apple cider [vinegar], but it’s not clear which is better,” said Shiv Mudgal, PhD, corresponding author of the paper and an associate professor in nursing at the All India Institute of Medical Sciences in Deoghar, India.
“We thought it would be nice to get some idea about how they work and how they compete with each other,” said Subodh Kumar, MD, the first author and an associate professor in pharmacology at the All India Institute of Medical Sciences in Deoghar, India.
They wanted to understand how the herbal remedies worked by including insulin level and HOMA-IR as measurable outcomes but found nothing conclusive. Instead, they speculated that the effect of apple cider vinegar and fenugreek seeds on blood glucose and A1c could be related to delayed gastric emptying, among other mechanisms.
However, the results should be interpreted with caution, said Dr. Kumar.
Apple cider vinegar had three clinical trials to back the finding, and fenugreek seeds had four studies supporting the results – fewer than the other included remedies. The authors also identified risks of bias from the randomization process and the allocation concealment process in several of the included trials.
Most of the studies included only short follow-up periods, meaning that the long-term effects of using these herbal remedies to help manage type 2 diabetes remain unclear.
The six herbal remedies included in the study were chosen out of dozens of popular complementary medicines for the strength and number of clinical trials backing their use.
The limited number included in the review is a drawback, according to Merlin Willcox, DPhil, a clinical lecturer in general practice at the University of Southampton, England, who was not involved in the research.
“It means they’ve left out stuff that’s potentially effective,” Dr. Willcox said in an interview.
Dr. Willcox, who has coauthored a review of herbal remedies for glycemic control in type 2 diabetes, said he was surprised that apple cider vinegar came out on top in this analysis.
His review concluded that aloe vera leaf gel, psyllium fiber, and fenugreek seeds appeared to be the most effective at reducing A1c, compared with the control groups of the included trials, out of 18 plant-based remedies.
There were no adverse effects associated with the herbal remedies, according to Dr. Mudgal. However, the evidence for the herbal remedies included in their review also lacked substantial follow-up periods assessing their long-term effects.
“You need to look at the evidence for each individual remedy; it’s not just about what plant it is, but it’s about what preparation, what dose. All of that comes into play,” Dr. Willcox said.
Up to 3.6 million people use herbal remedies to manage type 2 diabetes in the United States, according to a 2014 study cited by the review authors. The number is much higher elsewhere: As many as two-thirds of patients with diabetes in India and Saudi Arabia incorporate herbal remedies to help manage symptoms, whereas about half of patients with diabetes in the United Kingdom use herbal medicines.
Experts warn of the risks associated with using herbal medicines to complement traditional therapies.
“I caution my patients about dietary supplements and herbals because of the lack of high-quality data demonstrating efficacy and safety,” Katherine H. Saunders, MD, DABOM, cofounder of Intellihealth and clinical assistant professor of medicine at Weill Cornell Medicine, New York, said in an interview.
For Dr. Willcox, the risks relate to where patients get their information from. Many patients with type 2 diabetes are too scared to talk to their doctor about herbal medicines.
“They think their doctor is going to be negative or dismissive,” Dr. Willcox said. “So patients are getting their information from family and friends or from the Internet, which is not necessarily the most reliable, evidence-based source of information.”
The authors have reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
The review included 44 randomized clinical trials with more than 3,000 participants using six herbal remedies: apple cider vinegar, cinnamon, curcumin, fenugreek seeds, ginger, and saffron.
Apple cider vinegar, fenugreek seeds, curcumin (turmeric), and cinnamon resulted in statistically significant reductions in fasting blood glucose, compared with the control groups in the clinical trials. Out of all the remedies, the authors found apple cider vinegar to be the most effective for lowering fasting blood glucose levels.
The review also found that apple cider vinegar and fenugreek seeds had a statistically significant effect on reducing A1c, compared with the control groups. The authors found the herbal remedies made no difference to insulin level or homeostatic model assessment for insulin resistance (HOMA-IR).
The results are published online in Diabetes & Metabolic Syndrome: Clinical Research & Reviews. The authors said they hoped the review would help medical professionals and people with type 2 diabetes understand the effectiveness of different herbal remedies and consider incorporating these remedies into standard care.
“Some people use curcumin, some use ginger, some use apple cider [vinegar], but it’s not clear which is better,” said Shiv Mudgal, PhD, corresponding author of the paper and an associate professor in nursing at the All India Institute of Medical Sciences in Deoghar, India.
“We thought it would be nice to get some idea about how they work and how they compete with each other,” said Subodh Kumar, MD, the first author and an associate professor in pharmacology at the All India Institute of Medical Sciences in Deoghar, India.
They wanted to understand how the herbal remedies worked by including insulin level and HOMA-IR as measurable outcomes but found nothing conclusive. Instead, they speculated that the effect of apple cider vinegar and fenugreek seeds on blood glucose and A1c could be related to delayed gastric emptying, among other mechanisms.
However, the results should be interpreted with caution, said Dr. Kumar.
Apple cider vinegar had three clinical trials to back the finding, and fenugreek seeds had four studies supporting the results – fewer than the other included remedies. The authors also identified risks of bias from the randomization process and the allocation concealment process in several of the included trials.
Most of the studies included only short follow-up periods, meaning that the long-term effects of using these herbal remedies to help manage type 2 diabetes remain unclear.
The six herbal remedies included in the study were chosen out of dozens of popular complementary medicines for the strength and number of clinical trials backing their use.
The limited number included in the review is a drawback, according to Merlin Willcox, DPhil, a clinical lecturer in general practice at the University of Southampton, England, who was not involved in the research.
“It means they’ve left out stuff that’s potentially effective,” Dr. Willcox said in an interview.
Dr. Willcox, who has coauthored a review of herbal remedies for glycemic control in type 2 diabetes, said he was surprised that apple cider vinegar came out on top in this analysis.
His review concluded that aloe vera leaf gel, psyllium fiber, and fenugreek seeds appeared to be the most effective at reducing A1c, compared with the control groups of the included trials, out of 18 plant-based remedies.
There were no adverse effects associated with the herbal remedies, according to Dr. Mudgal. However, the evidence for the herbal remedies included in their review also lacked substantial follow-up periods assessing their long-term effects.
“You need to look at the evidence for each individual remedy; it’s not just about what plant it is, but it’s about what preparation, what dose. All of that comes into play,” Dr. Willcox said.
Up to 3.6 million people use herbal remedies to manage type 2 diabetes in the United States, according to a 2014 study cited by the review authors. The number is much higher elsewhere: As many as two-thirds of patients with diabetes in India and Saudi Arabia incorporate herbal remedies to help manage symptoms, whereas about half of patients with diabetes in the United Kingdom use herbal medicines.
Experts warn of the risks associated with using herbal medicines to complement traditional therapies.
“I caution my patients about dietary supplements and herbals because of the lack of high-quality data demonstrating efficacy and safety,” Katherine H. Saunders, MD, DABOM, cofounder of Intellihealth and clinical assistant professor of medicine at Weill Cornell Medicine, New York, said in an interview.
For Dr. Willcox, the risks relate to where patients get their information from. Many patients with type 2 diabetes are too scared to talk to their doctor about herbal medicines.
“They think their doctor is going to be negative or dismissive,” Dr. Willcox said. “So patients are getting their information from family and friends or from the Internet, which is not necessarily the most reliable, evidence-based source of information.”
The authors have reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
The review included 44 randomized clinical trials with more than 3,000 participants using six herbal remedies: apple cider vinegar, cinnamon, curcumin, fenugreek seeds, ginger, and saffron.
Apple cider vinegar, fenugreek seeds, curcumin (turmeric), and cinnamon resulted in statistically significant reductions in fasting blood glucose, compared with the control groups in the clinical trials. Out of all the remedies, the authors found apple cider vinegar to be the most effective for lowering fasting blood glucose levels.
The review also found that apple cider vinegar and fenugreek seeds had a statistically significant effect on reducing A1c, compared with the control groups. The authors found the herbal remedies made no difference to insulin level or homeostatic model assessment for insulin resistance (HOMA-IR).
The results are published online in Diabetes & Metabolic Syndrome: Clinical Research & Reviews. The authors said they hoped the review would help medical professionals and people with type 2 diabetes understand the effectiveness of different herbal remedies and consider incorporating these remedies into standard care.
“Some people use curcumin, some use ginger, some use apple cider [vinegar], but it’s not clear which is better,” said Shiv Mudgal, PhD, corresponding author of the paper and an associate professor in nursing at the All India Institute of Medical Sciences in Deoghar, India.
“We thought it would be nice to get some idea about how they work and how they compete with each other,” said Subodh Kumar, MD, the first author and an associate professor in pharmacology at the All India Institute of Medical Sciences in Deoghar, India.
They wanted to understand how the herbal remedies worked by including insulin level and HOMA-IR as measurable outcomes but found nothing conclusive. Instead, they speculated that the effect of apple cider vinegar and fenugreek seeds on blood glucose and A1c could be related to delayed gastric emptying, among other mechanisms.
However, the results should be interpreted with caution, said Dr. Kumar.
Apple cider vinegar had three clinical trials to back the finding, and fenugreek seeds had four studies supporting the results – fewer than the other included remedies. The authors also identified risks of bias from the randomization process and the allocation concealment process in several of the included trials.
Most of the studies included only short follow-up periods, meaning that the long-term effects of using these herbal remedies to help manage type 2 diabetes remain unclear.
The six herbal remedies included in the study were chosen out of dozens of popular complementary medicines for the strength and number of clinical trials backing their use.
The limited number included in the review is a drawback, according to Merlin Willcox, DPhil, a clinical lecturer in general practice at the University of Southampton, England, who was not involved in the research.
“It means they’ve left out stuff that’s potentially effective,” Dr. Willcox said in an interview.
Dr. Willcox, who has coauthored a review of herbal remedies for glycemic control in type 2 diabetes, said he was surprised that apple cider vinegar came out on top in this analysis.
His review concluded that aloe vera leaf gel, psyllium fiber, and fenugreek seeds appeared to be the most effective at reducing A1c, compared with the control groups of the included trials, out of 18 plant-based remedies.
There were no adverse effects associated with the herbal remedies, according to Dr. Mudgal. However, the evidence for the herbal remedies included in their review also lacked substantial follow-up periods assessing their long-term effects.
“You need to look at the evidence for each individual remedy; it’s not just about what plant it is, but it’s about what preparation, what dose. All of that comes into play,” Dr. Willcox said.
Up to 3.6 million people use herbal remedies to manage type 2 diabetes in the United States, according to a 2014 study cited by the review authors. The number is much higher elsewhere: As many as two-thirds of patients with diabetes in India and Saudi Arabia incorporate herbal remedies to help manage symptoms, whereas about half of patients with diabetes in the United Kingdom use herbal medicines.
Experts warn of the risks associated with using herbal medicines to complement traditional therapies.
“I caution my patients about dietary supplements and herbals because of the lack of high-quality data demonstrating efficacy and safety,” Katherine H. Saunders, MD, DABOM, cofounder of Intellihealth and clinical assistant professor of medicine at Weill Cornell Medicine, New York, said in an interview.
For Dr. Willcox, the risks relate to where patients get their information from. Many patients with type 2 diabetes are too scared to talk to their doctor about herbal medicines.
“They think their doctor is going to be negative or dismissive,” Dr. Willcox said. “So patients are getting their information from family and friends or from the Internet, which is not necessarily the most reliable, evidence-based source of information.”
The authors have reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
FROM DIABETES & METABOLIC SYNDROME: CLINICAL RESEARCH & REVIEWS