User login
Lipids and Dementia: A Complex and Evolving Story
The relationship between lipid levels and the development of dementia is an evolving but confusing landscape.
“This is an incredibly complex area, and there really isn’t a clear consensus on this subject because different lipid classes reflect different things,” according to Betsy Mills, PhD, assistant director of aging and Alzheimer’s prevention at the Alzheimer’s Drug Discovery Foundation.
Some studies suggest that excessive lipid levels may increase the risk of developing dementia and Alzheimer’s disease (AD). Others imply that elevated low-density lipoprotein (LDL) cholesterol or even triglycerides may offer some protection against subsequent dementia whereas higher levels of high-density lipoprotein (HDL) cholesterol, hitherto thought to be protective, may have a deleterious effect.
“It depends on what lipids you’re measuring, what you’re using to measure those lipids, what age the person is, and multiple other factors,” Dr. Mills told this news organization.
Teasing out the variables and potential mechanisms for the association between lipids and dementia risk necessitates understanding the role that lipids play in the healthy brain, the negative impact of brain lipid dysregulation, and the interplay between cholesterol in the central nervous system (CNS) and the cholesterol in the rest of the body.
Beyond Amyloid
The role of lipids in AD risk has historically been “overlooked,” says Scott Hansen, PhD, associate professor, Department of Molecular Medicine, Herbert Wertheim UF Scripps Institute for Biomedical Innovation and Technology, Florida.
“The common narrative is that amyloid is the culprit in AD and certainly that’s the case in familial AD,” he told this news organization. “It’s been assumed that because amyloid deposits are also found in the brains of people with late-onset AD — which is the vast majority of cases — amyloid is the cause, but that’s not clear at all.”
The “limited clinical success” of aducanumab, its “extremely small efficacy” — despite its obvious success in eradicating the amyloid plaques — suggests there’s “much more to the story than amyloid.”
He and a growing community of scientists recognize the role of inflammation and lipids. “The major finding of my lab is that cholesterol actually drives the synthesis of amyloid via inflammation. In other words, amyloid is downstream of cholesterol. Cholesterol drives the inflammation, and the inflammation drives amyloid,” he said.
‘Lipid Invasion Model’
Because the brain is an incredibly lipid-rich organ, Dr. Mills said that “any dysregulation in lipid homeostasis will impact the brain because cholesterol is needed for the myelin sheaths, cell membranes, and other functions.”
A healthy brain relies upon healthy lipid regulation, and “since the first description of AD over 100 years ago, the disease has been associated with altered lipids in the brain,” Dr. Hansen noted.
He cited the “ lipid invasion model” as a way of understanding brain lipid dysregulation. This hypothesis posits that AD is driven by external lipids that enter the brain as a result of damage to the blood-brain barrier (BBB).
“Cholesterol in the brain and cholesterol in the periphery — meaning, in the rest of the body, outside the brain — are separate,” Dr. Hansen explained. “The brain produces its own cholesterol and keeps tight control of it.”
Under normal circumstances, cholesterol from the diet doesn’t enter the brain. “Each pool of cholesterol — in the brain and in the periphery — has its own distinct regulatory mechanisms, target cells, and transport mechanisms.”
When the BBB has been compromised, it becomes permeable, allowing LDL cholesterol to enter the brain, said Dr. Hansen. Then the brain’s own lipoproteins transport the invading cholesterol, allowing it to be taken up by neurons. In turn, this causes neuronal amyloid levels to rise, ultimately leading to the creation of amyloid-b plaques. It also plays a role in tau phosphorylation. Both are key features of AD pathology.
Elevated levels of cholesterol and other lipids have been found in amyloid plaques, Dr. Hansen noted. Moreover, studies of brains of patients with AD have pointed to BBB damage.
And the risk factors for AD overlap with the risk factors for damage to the BBB (such as, aging, brain trauma, hypertension, stress, sleep deprivation, smoking, excess alcohol, obesity, diabetes, and APOE4 genotype), according to the lipid invasion model paper cited by Dr. Hansen.
‘Chicken and Egg’
“There is a strong link between the brain and the heart, and we know that cardiovascular risk factors have an overlap with dementia risk factors — especially vascular dementia,” said Dr. Mills.
She explained that an atherogenic lipid profile results in narrowing of the arteries, with less blood reaching the brain. “This can lead to stress in the brain, which drives inflammation and pathology.”
But cholesterol itself plays an important role in inflammation, Dr. Hansen said. In the periphery, it is “part of an integral response to tissue damage and infection.”
In the brain, once cholesterol is synthesized by the astrocytes, it is transported to neurons via the apolipoprotein E (APOE) protein, which plays a role in brain cholesterol homeostasis, Dr. Mills explained. Those with the ε4 allele of APOE (APOE4) tend to have faultier transport and storage of lipids in the brain, relative to the other APOE variants.
It’s known that individuals with APOE4 are particularly vulnerable to late-onset AD, Dr. Hansen observed. By contrast, APOE2 has a more protective effect. “Most people have APOE3, which is ‘in between,’ ” he said.
When there is neuronal uptake of “invading cholesterol,” not only is amyloid produced but also neuroinflammatory cytokines, further driving inflammation. A vicious cycle ensues: Cholesterol induces cytokine release; and cytokine release, in turn, induces cholesterol synthesis — which “suggests an autocatalytic function of cholesterol in the escalation of inflammation,” Dr. Hansen suggested. He noted that permeability of the BBB also allows inflammatory cytokines from elsewhere in the body to invade the brain, further driving inflammation.
Dr. Mills elaborated: “We know that generally, in dementia, there appear to be some changes in cholesterol metabolism in the brain, but it’s a chicken-and-egg question. We know that as the disease progresses, neurons are dying and getting remodeled. Do these changes have to do with the degenerative process, or are the changes in the cholesterol metabolism actually driving the degenerative disease process? It’s probably a combination, but it’s unclear at this point.”
Lipids in Plasma vs CSF
Dr. Mills explained that HDL particles in the brain differ from those in the periphery. “In the CNS, you have ‘HDL-like particles,’ which are similar in size and composition [to HDL in the periphery] but aren’t the same particles.” The brain itself generates HDL-like lipoproteins, which are produced by astrocytes and other glial cells and found in cerebrospinal fluid (CSF).
Dyslipidemia in the periphery can be a marker for cardiovascular pathology. In the brain, “it can be an indication that there is active damage going on, depending on which compartment you’re looking at.”
She noted that plasma lipid levels and brain CSF lipid levels are “very different.” Research suggests that HDL in the CSF exhibits similar heterogeneity to plasma HDL, but these CSF lipoproteins present at 100-fold lower concentrations, compared to plasma HDL and have unique combinations of protein subpopulations. Lipidomics analysis studies show that these compartments “get very different readings, in terms of the predominant lipid disease state, and they are regulated differently from the way lipids in the periphery are regulated.”
In the brain, the cholesterol “needs to get shuttled from glial cells to neurons,” so defects in the transport process can disrupt overall brain homeostasis, said Dr. Mills. But since the brain system is separate from the peripheral system, measuring plasma lipids is more likely to point to cardiovascular risks, while changes reflected in CSF lipids are “more indicative of alteration in lipid homeostasis in the brain.”
HDL and Triglycerides: A Complicated Story
Dr. Mills noted that HDL in the periphery is “very complicated,” and the idea that HDL, as a measure on its own, is “necessarily ‘good’ isn’t particularly informative.” Rather, HDL is “extremely heterogeneous, very diverse, has different lipid compositions, different classes, and different modifications.” For example, like oxidized LDL, oxidized HDL is also “bad,” preventing the HDL from having protective functions.
Similarly, the apolipoproteins associated with HDL can affect the function of the HDL. “Our understanding of the HDL-like particles in the CNS is limited, but we do understand the APOE4 link,” Dr. Mills said. “It seems that the HDL-like particles containing APOE2 or APOE3 are larger and are more effective at transferring the lipids and cholesterol linked to them relative to APOE4-containing particles.”
Because HDL is more complex than simply being “good,” measuring HDL doesn’t “give you the full story,” said Dr. Mills. She speculates that this may be why there are studies suggesting that high levels of HDL might not have protective benefits and might even be detrimental. This makes it difficult to look at population studies, where the different subclasses of HDL are not necessarily captured in depth.
Dr. Mills pointed to another confounding factor, which is that much of the risk for the development of AD appears to be related to the interaction of HDL, LDL, and triglycerides. “When you look at each of these individually, you get a lot of heterogeneity, and it’s unclear what’s driving what,” she said.
An advantage of observational studies is that they give information about which of these markers are associated with trends and disease risks in specific groups vs others.
“For example, higher levels of triglycerides are associated with cardiovascular risk more in women, relative to men,” she said. And the triglyceride-to-HDL ratio seems “particularly robust” as a measure of cardiovascular health and risk.
The interpretation of associations with triglycerides can be “tricky” and “confusing” because results differ so much between studies, she said. “There are differences between middle age and older age, which have to do with age-related changes in metabolism and lipid metabolism and not necessarily that the markers are indicating something different,” she said.
Some research has suggested that triglycerides may have a protective effect against dementia, noted Uma Naidoo, MD, director of nutritional and lifestyle psychiatry, Massachusetts General Hospital, and director of nutritional psychiatry at MGH Academy.
This may be because the brain “runs mostly on energy from burning triglycerides,” suggested Dr. Naidoo, author of the books Calm Your Mind With Food and This Is Your Brain on Food.
In addition, having higher levels of triglycerides may be linked with having overall healthier behaviors, Dr. Naidoo told this news organization.
Dr. Mills said that in middle-aged individuals, high levels of LDL-C and triglycerides are “often indicative of more atherogenic particles and risk to cardiovascular health, which is a generally negative trajectory. But in older individuals, things become more complicated because there are differences in terms of clearance of some of these particles, tissue clearance and distribution, and nutrient status. So for older individuals, it seems that fluctuations in either direction—either too high or too low—tend to be more informative that some overall dysregulation is going on the system.”
She emphasized that, in this “emerging area, looking at only one or two studies is confusing. But if you look at the spectrum of studies, you can see a pattern, which is that the regulation gets ‘off,’ as people age.”
The Potential Role of Statins
Dr. Mills speculated that there may be “neuroprotective benefits for some of the statins which appear to be related to cardiovascular benefits. But at this point, we don’t have any clear data whether statins actually directly impact brain cholesterol, since it’s a separate pool.”
They could help “by increasing blood flow and reducing narrowing of the arteries, but any direct impact on the brain is still under investigation.”
Dr. Hansen pointed to research suggesting statins taken at midlife appear to be cardioprotective and may be protective of brain health as well, whereas statins initiated in older age do not appear to have these benefits.
He speculated that one reason statins seem less helpful when initiated later in life is that the BBB has already been damaged by systemic inflammation in the periphery, and the neuroinflammatory process resulting in neuronal destruction is already underway. “I think statins aren’t going to fix that problem, so although lowering cholesterol can be helpful in some respects, it might be too late to affect cognition because the nerves have already died and won’t grow back.”
Can Dietary Approaches Help?
Dr. Naidoo said that when looking at neurologic and psychiatric disease, “it’s important to think about the ‘long game’ — how can we improve our blood and cardiovascular health earlier in life to help potentiate healthy aging?”
From a nutritional psychiatry standpoint, Dr. Naidoo focuses on nourishing the gut microbiome and decreasing inflammation. “A healthy and balanced microbiome supports cognition, while the composition of gut bacteria is actually drastically different in patients with neurological diseases, such as AD.”
She recommends a nutrient-dense, anti-inflammatory diet including probiotic-rich foods (such as kimchi, sauerkraut, plain yogurt, and miso). Moreover, “the quality and structure of our fatty acids may be relevant as well: Increasing our intake of polyunsaturated fatty acids and avoiding processed fats like trans fats and hydrogenated oils may benefit our overall brain health.”
Dr. Naidoo recommends extra-virgin olive oil as a source of healthy fat. Its consumption is linked to lower incidence of AD by way of encouraging autophagy, which she calls “our own process of “cellular cleanup.’”
Dr. Naidoo believes that clinicians’ guidance to patients should “focus on healthy nutrition and other lifestyle practices, such as exercise, outdoor time, good sleep, and stress reduction.”
Dr. Mills notes the importance of omega-3 fatty acids, such as docosahexaenoic acid (DHA) , for brain health. “DHA is a major lipid component of neuronal membranes,” she said. “Because of inefficiencies in metabolism with APOE4, people tend to metabolize more of the lipids on the membranes themselves, so they have higher lipid membrane turnover and a greater need to supplement. Supplementing particularly through diet, with foods such as fatty fish rich in omega-3, can help boost the levels to help keep neuronal membranes intact.”
What This Means for the Clinician
“At this point, we see all of these associations between lipids and dementia, but we haven’t worked out exactly what it means on the individual level for an individual patient,” said Dr. Mills. Certainly, the picture is complex, and the understanding is growing and shifting. “The clinical applications remain unclear.”
One potential clinical take-home is that clinicians might consider tracking lipid levels over time. “If you follow a patient and see an increase or decrease [in lipid levels], that can be informative.” Looking at ratios of lipids might be more useful than looking only at a change in a single measure. “If you see trends in a variety of measures that track with one another, it might be more of a sign that something is potentially wrong.”
Whether the patient should first try a lifestyle intervention or might need medication is a “personalized clinical decision, depending on the individual, their risk factors, and how their levels are going,” said Dr. Mills.
Dr. Mills, Dr. Hansen, and Dr. Naidoo declared no relevant financial relationships.
A version of this article appeared on Medscape.com.
The relationship between lipid levels and the development of dementia is an evolving but confusing landscape.
“This is an incredibly complex area, and there really isn’t a clear consensus on this subject because different lipid classes reflect different things,” according to Betsy Mills, PhD, assistant director of aging and Alzheimer’s prevention at the Alzheimer’s Drug Discovery Foundation.
Some studies suggest that excessive lipid levels may increase the risk of developing dementia and Alzheimer’s disease (AD). Others imply that elevated low-density lipoprotein (LDL) cholesterol or even triglycerides may offer some protection against subsequent dementia whereas higher levels of high-density lipoprotein (HDL) cholesterol, hitherto thought to be protective, may have a deleterious effect.
“It depends on what lipids you’re measuring, what you’re using to measure those lipids, what age the person is, and multiple other factors,” Dr. Mills told this news organization.
Teasing out the variables and potential mechanisms for the association between lipids and dementia risk necessitates understanding the role that lipids play in the healthy brain, the negative impact of brain lipid dysregulation, and the interplay between cholesterol in the central nervous system (CNS) and the cholesterol in the rest of the body.
Beyond Amyloid
The role of lipids in AD risk has historically been “overlooked,” says Scott Hansen, PhD, associate professor, Department of Molecular Medicine, Herbert Wertheim UF Scripps Institute for Biomedical Innovation and Technology, Florida.
“The common narrative is that amyloid is the culprit in AD and certainly that’s the case in familial AD,” he told this news organization. “It’s been assumed that because amyloid deposits are also found in the brains of people with late-onset AD — which is the vast majority of cases — amyloid is the cause, but that’s not clear at all.”
The “limited clinical success” of aducanumab, its “extremely small efficacy” — despite its obvious success in eradicating the amyloid plaques — suggests there’s “much more to the story than amyloid.”
He and a growing community of scientists recognize the role of inflammation and lipids. “The major finding of my lab is that cholesterol actually drives the synthesis of amyloid via inflammation. In other words, amyloid is downstream of cholesterol. Cholesterol drives the inflammation, and the inflammation drives amyloid,” he said.
‘Lipid Invasion Model’
Because the brain is an incredibly lipid-rich organ, Dr. Mills said that “any dysregulation in lipid homeostasis will impact the brain because cholesterol is needed for the myelin sheaths, cell membranes, and other functions.”
A healthy brain relies upon healthy lipid regulation, and “since the first description of AD over 100 years ago, the disease has been associated with altered lipids in the brain,” Dr. Hansen noted.
He cited the “ lipid invasion model” as a way of understanding brain lipid dysregulation. This hypothesis posits that AD is driven by external lipids that enter the brain as a result of damage to the blood-brain barrier (BBB).
“Cholesterol in the brain and cholesterol in the periphery — meaning, in the rest of the body, outside the brain — are separate,” Dr. Hansen explained. “The brain produces its own cholesterol and keeps tight control of it.”
Under normal circumstances, cholesterol from the diet doesn’t enter the brain. “Each pool of cholesterol — in the brain and in the periphery — has its own distinct regulatory mechanisms, target cells, and transport mechanisms.”
When the BBB has been compromised, it becomes permeable, allowing LDL cholesterol to enter the brain, said Dr. Hansen. Then the brain’s own lipoproteins transport the invading cholesterol, allowing it to be taken up by neurons. In turn, this causes neuronal amyloid levels to rise, ultimately leading to the creation of amyloid-b plaques. It also plays a role in tau phosphorylation. Both are key features of AD pathology.
Elevated levels of cholesterol and other lipids have been found in amyloid plaques, Dr. Hansen noted. Moreover, studies of brains of patients with AD have pointed to BBB damage.
And the risk factors for AD overlap with the risk factors for damage to the BBB (such as, aging, brain trauma, hypertension, stress, sleep deprivation, smoking, excess alcohol, obesity, diabetes, and APOE4 genotype), according to the lipid invasion model paper cited by Dr. Hansen.
‘Chicken and Egg’
“There is a strong link between the brain and the heart, and we know that cardiovascular risk factors have an overlap with dementia risk factors — especially vascular dementia,” said Dr. Mills.
She explained that an atherogenic lipid profile results in narrowing of the arteries, with less blood reaching the brain. “This can lead to stress in the brain, which drives inflammation and pathology.”
But cholesterol itself plays an important role in inflammation, Dr. Hansen said. In the periphery, it is “part of an integral response to tissue damage and infection.”
In the brain, once cholesterol is synthesized by the astrocytes, it is transported to neurons via the apolipoprotein E (APOE) protein, which plays a role in brain cholesterol homeostasis, Dr. Mills explained. Those with the ε4 allele of APOE (APOE4) tend to have faultier transport and storage of lipids in the brain, relative to the other APOE variants.
It’s known that individuals with APOE4 are particularly vulnerable to late-onset AD, Dr. Hansen observed. By contrast, APOE2 has a more protective effect. “Most people have APOE3, which is ‘in between,’ ” he said.
When there is neuronal uptake of “invading cholesterol,” not only is amyloid produced but also neuroinflammatory cytokines, further driving inflammation. A vicious cycle ensues: Cholesterol induces cytokine release; and cytokine release, in turn, induces cholesterol synthesis — which “suggests an autocatalytic function of cholesterol in the escalation of inflammation,” Dr. Hansen suggested. He noted that permeability of the BBB also allows inflammatory cytokines from elsewhere in the body to invade the brain, further driving inflammation.
Dr. Mills elaborated: “We know that generally, in dementia, there appear to be some changes in cholesterol metabolism in the brain, but it’s a chicken-and-egg question. We know that as the disease progresses, neurons are dying and getting remodeled. Do these changes have to do with the degenerative process, or are the changes in the cholesterol metabolism actually driving the degenerative disease process? It’s probably a combination, but it’s unclear at this point.”
Lipids in Plasma vs CSF
Dr. Mills explained that HDL particles in the brain differ from those in the periphery. “In the CNS, you have ‘HDL-like particles,’ which are similar in size and composition [to HDL in the periphery] but aren’t the same particles.” The brain itself generates HDL-like lipoproteins, which are produced by astrocytes and other glial cells and found in cerebrospinal fluid (CSF).
Dyslipidemia in the periphery can be a marker for cardiovascular pathology. In the brain, “it can be an indication that there is active damage going on, depending on which compartment you’re looking at.”
She noted that plasma lipid levels and brain CSF lipid levels are “very different.” Research suggests that HDL in the CSF exhibits similar heterogeneity to plasma HDL, but these CSF lipoproteins present at 100-fold lower concentrations, compared to plasma HDL and have unique combinations of protein subpopulations. Lipidomics analysis studies show that these compartments “get very different readings, in terms of the predominant lipid disease state, and they are regulated differently from the way lipids in the periphery are regulated.”
In the brain, the cholesterol “needs to get shuttled from glial cells to neurons,” so defects in the transport process can disrupt overall brain homeostasis, said Dr. Mills. But since the brain system is separate from the peripheral system, measuring plasma lipids is more likely to point to cardiovascular risks, while changes reflected in CSF lipids are “more indicative of alteration in lipid homeostasis in the brain.”
HDL and Triglycerides: A Complicated Story
Dr. Mills noted that HDL in the periphery is “very complicated,” and the idea that HDL, as a measure on its own, is “necessarily ‘good’ isn’t particularly informative.” Rather, HDL is “extremely heterogeneous, very diverse, has different lipid compositions, different classes, and different modifications.” For example, like oxidized LDL, oxidized HDL is also “bad,” preventing the HDL from having protective functions.
Similarly, the apolipoproteins associated with HDL can affect the function of the HDL. “Our understanding of the HDL-like particles in the CNS is limited, but we do understand the APOE4 link,” Dr. Mills said. “It seems that the HDL-like particles containing APOE2 or APOE3 are larger and are more effective at transferring the lipids and cholesterol linked to them relative to APOE4-containing particles.”
Because HDL is more complex than simply being “good,” measuring HDL doesn’t “give you the full story,” said Dr. Mills. She speculates that this may be why there are studies suggesting that high levels of HDL might not have protective benefits and might even be detrimental. This makes it difficult to look at population studies, where the different subclasses of HDL are not necessarily captured in depth.
Dr. Mills pointed to another confounding factor, which is that much of the risk for the development of AD appears to be related to the interaction of HDL, LDL, and triglycerides. “When you look at each of these individually, you get a lot of heterogeneity, and it’s unclear what’s driving what,” she said.
An advantage of observational studies is that they give information about which of these markers are associated with trends and disease risks in specific groups vs others.
“For example, higher levels of triglycerides are associated with cardiovascular risk more in women, relative to men,” she said. And the triglyceride-to-HDL ratio seems “particularly robust” as a measure of cardiovascular health and risk.
The interpretation of associations with triglycerides can be “tricky” and “confusing” because results differ so much between studies, she said. “There are differences between middle age and older age, which have to do with age-related changes in metabolism and lipid metabolism and not necessarily that the markers are indicating something different,” she said.
Some research has suggested that triglycerides may have a protective effect against dementia, noted Uma Naidoo, MD, director of nutritional and lifestyle psychiatry, Massachusetts General Hospital, and director of nutritional psychiatry at MGH Academy.
This may be because the brain “runs mostly on energy from burning triglycerides,” suggested Dr. Naidoo, author of the books Calm Your Mind With Food and This Is Your Brain on Food.
In addition, having higher levels of triglycerides may be linked with having overall healthier behaviors, Dr. Naidoo told this news organization.
Dr. Mills said that in middle-aged individuals, high levels of LDL-C and triglycerides are “often indicative of more atherogenic particles and risk to cardiovascular health, which is a generally negative trajectory. But in older individuals, things become more complicated because there are differences in terms of clearance of some of these particles, tissue clearance and distribution, and nutrient status. So for older individuals, it seems that fluctuations in either direction—either too high or too low—tend to be more informative that some overall dysregulation is going on the system.”
She emphasized that, in this “emerging area, looking at only one or two studies is confusing. But if you look at the spectrum of studies, you can see a pattern, which is that the regulation gets ‘off,’ as people age.”
The Potential Role of Statins
Dr. Mills speculated that there may be “neuroprotective benefits for some of the statins which appear to be related to cardiovascular benefits. But at this point, we don’t have any clear data whether statins actually directly impact brain cholesterol, since it’s a separate pool.”
They could help “by increasing blood flow and reducing narrowing of the arteries, but any direct impact on the brain is still under investigation.”
Dr. Hansen pointed to research suggesting statins taken at midlife appear to be cardioprotective and may be protective of brain health as well, whereas statins initiated in older age do not appear to have these benefits.
He speculated that one reason statins seem less helpful when initiated later in life is that the BBB has already been damaged by systemic inflammation in the periphery, and the neuroinflammatory process resulting in neuronal destruction is already underway. “I think statins aren’t going to fix that problem, so although lowering cholesterol can be helpful in some respects, it might be too late to affect cognition because the nerves have already died and won’t grow back.”
Can Dietary Approaches Help?
Dr. Naidoo said that when looking at neurologic and psychiatric disease, “it’s important to think about the ‘long game’ — how can we improve our blood and cardiovascular health earlier in life to help potentiate healthy aging?”
From a nutritional psychiatry standpoint, Dr. Naidoo focuses on nourishing the gut microbiome and decreasing inflammation. “A healthy and balanced microbiome supports cognition, while the composition of gut bacteria is actually drastically different in patients with neurological diseases, such as AD.”
She recommends a nutrient-dense, anti-inflammatory diet including probiotic-rich foods (such as kimchi, sauerkraut, plain yogurt, and miso). Moreover, “the quality and structure of our fatty acids may be relevant as well: Increasing our intake of polyunsaturated fatty acids and avoiding processed fats like trans fats and hydrogenated oils may benefit our overall brain health.”
Dr. Naidoo recommends extra-virgin olive oil as a source of healthy fat. Its consumption is linked to lower incidence of AD by way of encouraging autophagy, which she calls “our own process of “cellular cleanup.’”
Dr. Naidoo believes that clinicians’ guidance to patients should “focus on healthy nutrition and other lifestyle practices, such as exercise, outdoor time, good sleep, and stress reduction.”
Dr. Mills notes the importance of omega-3 fatty acids, such as docosahexaenoic acid (DHA) , for brain health. “DHA is a major lipid component of neuronal membranes,” she said. “Because of inefficiencies in metabolism with APOE4, people tend to metabolize more of the lipids on the membranes themselves, so they have higher lipid membrane turnover and a greater need to supplement. Supplementing particularly through diet, with foods such as fatty fish rich in omega-3, can help boost the levels to help keep neuronal membranes intact.”
What This Means for the Clinician
“At this point, we see all of these associations between lipids and dementia, but we haven’t worked out exactly what it means on the individual level for an individual patient,” said Dr. Mills. Certainly, the picture is complex, and the understanding is growing and shifting. “The clinical applications remain unclear.”
One potential clinical take-home is that clinicians might consider tracking lipid levels over time. “If you follow a patient and see an increase or decrease [in lipid levels], that can be informative.” Looking at ratios of lipids might be more useful than looking only at a change in a single measure. “If you see trends in a variety of measures that track with one another, it might be more of a sign that something is potentially wrong.”
Whether the patient should first try a lifestyle intervention or might need medication is a “personalized clinical decision, depending on the individual, their risk factors, and how their levels are going,” said Dr. Mills.
Dr. Mills, Dr. Hansen, and Dr. Naidoo declared no relevant financial relationships.
A version of this article appeared on Medscape.com.
The relationship between lipid levels and the development of dementia is an evolving but confusing landscape.
“This is an incredibly complex area, and there really isn’t a clear consensus on this subject because different lipid classes reflect different things,” according to Betsy Mills, PhD, assistant director of aging and Alzheimer’s prevention at the Alzheimer’s Drug Discovery Foundation.
Some studies suggest that excessive lipid levels may increase the risk of developing dementia and Alzheimer’s disease (AD). Others imply that elevated low-density lipoprotein (LDL) cholesterol or even triglycerides may offer some protection against subsequent dementia whereas higher levels of high-density lipoprotein (HDL) cholesterol, hitherto thought to be protective, may have a deleterious effect.
“It depends on what lipids you’re measuring, what you’re using to measure those lipids, what age the person is, and multiple other factors,” Dr. Mills told this news organization.
Teasing out the variables and potential mechanisms for the association between lipids and dementia risk necessitates understanding the role that lipids play in the healthy brain, the negative impact of brain lipid dysregulation, and the interplay between cholesterol in the central nervous system (CNS) and the cholesterol in the rest of the body.
Beyond Amyloid
The role of lipids in AD risk has historically been “overlooked,” says Scott Hansen, PhD, associate professor, Department of Molecular Medicine, Herbert Wertheim UF Scripps Institute for Biomedical Innovation and Technology, Florida.
“The common narrative is that amyloid is the culprit in AD and certainly that’s the case in familial AD,” he told this news organization. “It’s been assumed that because amyloid deposits are also found in the brains of people with late-onset AD — which is the vast majority of cases — amyloid is the cause, but that’s not clear at all.”
The “limited clinical success” of aducanumab, its “extremely small efficacy” — despite its obvious success in eradicating the amyloid plaques — suggests there’s “much more to the story than amyloid.”
He and a growing community of scientists recognize the role of inflammation and lipids. “The major finding of my lab is that cholesterol actually drives the synthesis of amyloid via inflammation. In other words, amyloid is downstream of cholesterol. Cholesterol drives the inflammation, and the inflammation drives amyloid,” he said.
‘Lipid Invasion Model’
Because the brain is an incredibly lipid-rich organ, Dr. Mills said that “any dysregulation in lipid homeostasis will impact the brain because cholesterol is needed for the myelin sheaths, cell membranes, and other functions.”
A healthy brain relies upon healthy lipid regulation, and “since the first description of AD over 100 years ago, the disease has been associated with altered lipids in the brain,” Dr. Hansen noted.
He cited the “ lipid invasion model” as a way of understanding brain lipid dysregulation. This hypothesis posits that AD is driven by external lipids that enter the brain as a result of damage to the blood-brain barrier (BBB).
“Cholesterol in the brain and cholesterol in the periphery — meaning, in the rest of the body, outside the brain — are separate,” Dr. Hansen explained. “The brain produces its own cholesterol and keeps tight control of it.”
Under normal circumstances, cholesterol from the diet doesn’t enter the brain. “Each pool of cholesterol — in the brain and in the periphery — has its own distinct regulatory mechanisms, target cells, and transport mechanisms.”
When the BBB has been compromised, it becomes permeable, allowing LDL cholesterol to enter the brain, said Dr. Hansen. Then the brain’s own lipoproteins transport the invading cholesterol, allowing it to be taken up by neurons. In turn, this causes neuronal amyloid levels to rise, ultimately leading to the creation of amyloid-b plaques. It also plays a role in tau phosphorylation. Both are key features of AD pathology.
Elevated levels of cholesterol and other lipids have been found in amyloid plaques, Dr. Hansen noted. Moreover, studies of brains of patients with AD have pointed to BBB damage.
And the risk factors for AD overlap with the risk factors for damage to the BBB (such as, aging, brain trauma, hypertension, stress, sleep deprivation, smoking, excess alcohol, obesity, diabetes, and APOE4 genotype), according to the lipid invasion model paper cited by Dr. Hansen.
‘Chicken and Egg’
“There is a strong link between the brain and the heart, and we know that cardiovascular risk factors have an overlap with dementia risk factors — especially vascular dementia,” said Dr. Mills.
She explained that an atherogenic lipid profile results in narrowing of the arteries, with less blood reaching the brain. “This can lead to stress in the brain, which drives inflammation and pathology.”
But cholesterol itself plays an important role in inflammation, Dr. Hansen said. In the periphery, it is “part of an integral response to tissue damage and infection.”
In the brain, once cholesterol is synthesized by the astrocytes, it is transported to neurons via the apolipoprotein E (APOE) protein, which plays a role in brain cholesterol homeostasis, Dr. Mills explained. Those with the ε4 allele of APOE (APOE4) tend to have faultier transport and storage of lipids in the brain, relative to the other APOE variants.
It’s known that individuals with APOE4 are particularly vulnerable to late-onset AD, Dr. Hansen observed. By contrast, APOE2 has a more protective effect. “Most people have APOE3, which is ‘in between,’ ” he said.
When there is neuronal uptake of “invading cholesterol,” not only is amyloid produced but also neuroinflammatory cytokines, further driving inflammation. A vicious cycle ensues: Cholesterol induces cytokine release; and cytokine release, in turn, induces cholesterol synthesis — which “suggests an autocatalytic function of cholesterol in the escalation of inflammation,” Dr. Hansen suggested. He noted that permeability of the BBB also allows inflammatory cytokines from elsewhere in the body to invade the brain, further driving inflammation.
Dr. Mills elaborated: “We know that generally, in dementia, there appear to be some changes in cholesterol metabolism in the brain, but it’s a chicken-and-egg question. We know that as the disease progresses, neurons are dying and getting remodeled. Do these changes have to do with the degenerative process, or are the changes in the cholesterol metabolism actually driving the degenerative disease process? It’s probably a combination, but it’s unclear at this point.”
Lipids in Plasma vs CSF
Dr. Mills explained that HDL particles in the brain differ from those in the periphery. “In the CNS, you have ‘HDL-like particles,’ which are similar in size and composition [to HDL in the periphery] but aren’t the same particles.” The brain itself generates HDL-like lipoproteins, which are produced by astrocytes and other glial cells and found in cerebrospinal fluid (CSF).
Dyslipidemia in the periphery can be a marker for cardiovascular pathology. In the brain, “it can be an indication that there is active damage going on, depending on which compartment you’re looking at.”
She noted that plasma lipid levels and brain CSF lipid levels are “very different.” Research suggests that HDL in the CSF exhibits similar heterogeneity to plasma HDL, but these CSF lipoproteins present at 100-fold lower concentrations, compared to plasma HDL and have unique combinations of protein subpopulations. Lipidomics analysis studies show that these compartments “get very different readings, in terms of the predominant lipid disease state, and they are regulated differently from the way lipids in the periphery are regulated.”
In the brain, the cholesterol “needs to get shuttled from glial cells to neurons,” so defects in the transport process can disrupt overall brain homeostasis, said Dr. Mills. But since the brain system is separate from the peripheral system, measuring plasma lipids is more likely to point to cardiovascular risks, while changes reflected in CSF lipids are “more indicative of alteration in lipid homeostasis in the brain.”
HDL and Triglycerides: A Complicated Story
Dr. Mills noted that HDL in the periphery is “very complicated,” and the idea that HDL, as a measure on its own, is “necessarily ‘good’ isn’t particularly informative.” Rather, HDL is “extremely heterogeneous, very diverse, has different lipid compositions, different classes, and different modifications.” For example, like oxidized LDL, oxidized HDL is also “bad,” preventing the HDL from having protective functions.
Similarly, the apolipoproteins associated with HDL can affect the function of the HDL. “Our understanding of the HDL-like particles in the CNS is limited, but we do understand the APOE4 link,” Dr. Mills said. “It seems that the HDL-like particles containing APOE2 or APOE3 are larger and are more effective at transferring the lipids and cholesterol linked to them relative to APOE4-containing particles.”
Because HDL is more complex than simply being “good,” measuring HDL doesn’t “give you the full story,” said Dr. Mills. She speculates that this may be why there are studies suggesting that high levels of HDL might not have protective benefits and might even be detrimental. This makes it difficult to look at population studies, where the different subclasses of HDL are not necessarily captured in depth.
Dr. Mills pointed to another confounding factor, which is that much of the risk for the development of AD appears to be related to the interaction of HDL, LDL, and triglycerides. “When you look at each of these individually, you get a lot of heterogeneity, and it’s unclear what’s driving what,” she said.
An advantage of observational studies is that they give information about which of these markers are associated with trends and disease risks in specific groups vs others.
“For example, higher levels of triglycerides are associated with cardiovascular risk more in women, relative to men,” she said. And the triglyceride-to-HDL ratio seems “particularly robust” as a measure of cardiovascular health and risk.
The interpretation of associations with triglycerides can be “tricky” and “confusing” because results differ so much between studies, she said. “There are differences between middle age and older age, which have to do with age-related changes in metabolism and lipid metabolism and not necessarily that the markers are indicating something different,” she said.
Some research has suggested that triglycerides may have a protective effect against dementia, noted Uma Naidoo, MD, director of nutritional and lifestyle psychiatry, Massachusetts General Hospital, and director of nutritional psychiatry at MGH Academy.
This may be because the brain “runs mostly on energy from burning triglycerides,” suggested Dr. Naidoo, author of the books Calm Your Mind With Food and This Is Your Brain on Food.
In addition, having higher levels of triglycerides may be linked with having overall healthier behaviors, Dr. Naidoo told this news organization.
Dr. Mills said that in middle-aged individuals, high levels of LDL-C and triglycerides are “often indicative of more atherogenic particles and risk to cardiovascular health, which is a generally negative trajectory. But in older individuals, things become more complicated because there are differences in terms of clearance of some of these particles, tissue clearance and distribution, and nutrient status. So for older individuals, it seems that fluctuations in either direction—either too high or too low—tend to be more informative that some overall dysregulation is going on the system.”
She emphasized that, in this “emerging area, looking at only one or two studies is confusing. But if you look at the spectrum of studies, you can see a pattern, which is that the regulation gets ‘off,’ as people age.”
The Potential Role of Statins
Dr. Mills speculated that there may be “neuroprotective benefits for some of the statins which appear to be related to cardiovascular benefits. But at this point, we don’t have any clear data whether statins actually directly impact brain cholesterol, since it’s a separate pool.”
They could help “by increasing blood flow and reducing narrowing of the arteries, but any direct impact on the brain is still under investigation.”
Dr. Hansen pointed to research suggesting statins taken at midlife appear to be cardioprotective and may be protective of brain health as well, whereas statins initiated in older age do not appear to have these benefits.
He speculated that one reason statins seem less helpful when initiated later in life is that the BBB has already been damaged by systemic inflammation in the periphery, and the neuroinflammatory process resulting in neuronal destruction is already underway. “I think statins aren’t going to fix that problem, so although lowering cholesterol can be helpful in some respects, it might be too late to affect cognition because the nerves have already died and won’t grow back.”
Can Dietary Approaches Help?
Dr. Naidoo said that when looking at neurologic and psychiatric disease, “it’s important to think about the ‘long game’ — how can we improve our blood and cardiovascular health earlier in life to help potentiate healthy aging?”
From a nutritional psychiatry standpoint, Dr. Naidoo focuses on nourishing the gut microbiome and decreasing inflammation. “A healthy and balanced microbiome supports cognition, while the composition of gut bacteria is actually drastically different in patients with neurological diseases, such as AD.”
She recommends a nutrient-dense, anti-inflammatory diet including probiotic-rich foods (such as kimchi, sauerkraut, plain yogurt, and miso). Moreover, “the quality and structure of our fatty acids may be relevant as well: Increasing our intake of polyunsaturated fatty acids and avoiding processed fats like trans fats and hydrogenated oils may benefit our overall brain health.”
Dr. Naidoo recommends extra-virgin olive oil as a source of healthy fat. Its consumption is linked to lower incidence of AD by way of encouraging autophagy, which she calls “our own process of “cellular cleanup.’”
Dr. Naidoo believes that clinicians’ guidance to patients should “focus on healthy nutrition and other lifestyle practices, such as exercise, outdoor time, good sleep, and stress reduction.”
Dr. Mills notes the importance of omega-3 fatty acids, such as docosahexaenoic acid (DHA) , for brain health. “DHA is a major lipid component of neuronal membranes,” she said. “Because of inefficiencies in metabolism with APOE4, people tend to metabolize more of the lipids on the membranes themselves, so they have higher lipid membrane turnover and a greater need to supplement. Supplementing particularly through diet, with foods such as fatty fish rich in omega-3, can help boost the levels to help keep neuronal membranes intact.”
What This Means for the Clinician
“At this point, we see all of these associations between lipids and dementia, but we haven’t worked out exactly what it means on the individual level for an individual patient,” said Dr. Mills. Certainly, the picture is complex, and the understanding is growing and shifting. “The clinical applications remain unclear.”
One potential clinical take-home is that clinicians might consider tracking lipid levels over time. “If you follow a patient and see an increase or decrease [in lipid levels], that can be informative.” Looking at ratios of lipids might be more useful than looking only at a change in a single measure. “If you see trends in a variety of measures that track with one another, it might be more of a sign that something is potentially wrong.”
Whether the patient should first try a lifestyle intervention or might need medication is a “personalized clinical decision, depending on the individual, their risk factors, and how their levels are going,” said Dr. Mills.
Dr. Mills, Dr. Hansen, and Dr. Naidoo declared no relevant financial relationships.
A version of this article appeared on Medscape.com.
Loss of appetite and mood fluctuations
Given the patient's results on the genetic panel and MRI, as well as the noted cognitive decline and increased aggression, this patient is suspected of having limbic-predominant age-related TDP-43 encephalopathy (LATE) secondary to AD and is referred to the neurologist on her multidisciplinary care team for further consultation and testing.
AD is one of the most common forms of dementia. More than 6 million people in the United States have clinical AD or mild cognitive impairment because of AD. LATE is a new classification of dementia, identified in 2019, that mimics AD but is a unique disease entity driven by the misfolding of the protein TDP-43, which regulates gene expression in the brain. Misfolded TDP-43 protein is common among older adults aged ≥ 85 years, and about a quarter of this population has enough misfolded TDP-43 protein to affect their memory and cognition.
Diagnosing AD currently relies on a clinical approach. A complete physical examination, with a detailed neurologic examination and a mental status examination, is used to evaluate disease stage. Initial mental status testing evaluates attention and concentration, recent and remote memory, language, praxis, executive function, and visuospatial function. Because LATE is a newly discovered form of dementia, there are no set guidelines on diagnosing LATE and no robust biomarker for TDP-43. What is known about LATE has been gleaned mostly from retrospective clinicopathologic studies.
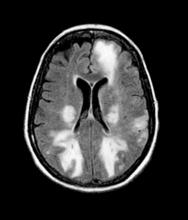
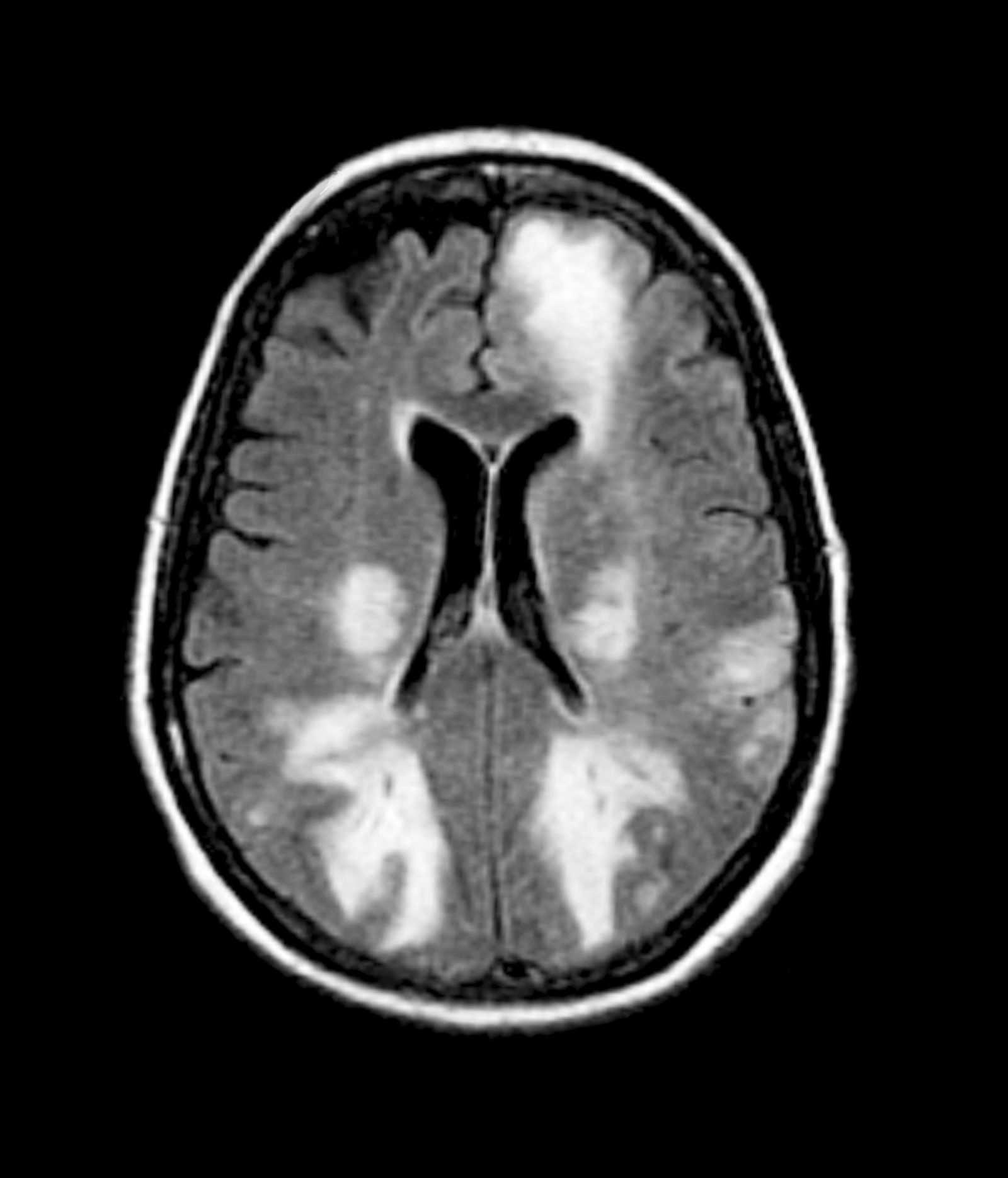
The LATE consensus working group reports that the clinical course of disease, as studied by autopsy-proven LATE neuropathologic change (LATE-NC), is described as an "amnestic cognitive syndrome that can evolve to incorporate multiple cognitive domains and ultimately to impair activities of daily living." Researchers are currently analyzing different clinical assessments and neuroimaging with MRI to characterize LATE. A group of international researchers recently published a set of clinical criteria for limbic-predominant amnestic neurodegenerative syndrome (LANS), which is associated with LATE-NC. Their criteria include "core, standard and advanced features that are measurable in vivo, including older age at evaluation, mild clinical syndrome, disproportionate hippocampal atrophy, impaired semantic memory, limbic hypometabolism, absence of neocortical degenerative patterns and low likelihood of neocortical tau, with degrees of certainty (highest, high, moderate, low)." Other neuroimaging studies of autopsy-confirmed LATE-NC have shown that atrophy is mostly focused in the medial temporal lobe with marked reduced hippocampal volume.
The group reports that LATE and AD probably share pathophysiologic mechanisms. One of the universally accepted hallmarks of AD is the formation of beta-amyloid plaques, which are dense, mostly insoluble deposits of beta-amyloid protein that develop around neurons in the hippocampus and other regions in the cerebral cortex used for decision-making. These plaques disrupt brain function and lead to brain atrophy. The LATE group also reports that this same pathology has been noted with LATE: "Many subjects with LATE-NC have comorbid brain pathologies, often including amyloid-beta plaques and tauopathy." That said, genetic studies have helped identify five genes with risk alleles for LATE (GRN, TMEM106B, ABCC9, KCNMB2, and APOE), suggesting disease-specific underlying mechanisms compared to AD.
Patient and caregiver education and guidance is vital with a dementia diagnosis. If LATE and/or AD are suspected, physicians should encourage the involvement of family and friends who agree to become more involved in the patient's care as the disease progresses. These individuals need to understand the patient's wishes around care, especially for the future when the patient is no longer able to make decisions. The patient may also consider establishing medical advance directives and durable power of attorney for medical and financial decision-making. Caregivers supporting the patient are encouraged to help balance the physical needs of the patient while maintaining respect for them as a competent adult to the extent allowed by the progression of their disease.
Because LATE is a new classification of dementia, there are no known effective treatments. One ongoing study is testing the use of autologous bone marrow–derived stem cells to help improve cognitive impairment among patients with LATE, AD, and other dementias. Current AD treatments are focused on symptomatic therapies that modulate neurotransmitters — either acetylcholine or glutamate. The standard medical treatment includes cholinesterase inhibitors and a partial N-methyl-D-aspartate antagonist. Two amyloid-directed antibodies (aducanumab, lecanemab) are currently available in the United States for individuals with AD exhibiting mild cognitive impairment or mild dementia. A third agent currently in clinical trials (donanemab) has shown significantly slowed clinical progression after 1.5 years among clinical trial participants with early symptomatic AD and amyloid and tau pathology.
Shaheen E. Lakhan, MD, PhD, MS, MEd, Chief of Pain Management, Carilion Clinic and Virginia Tech Carilion School of Medicine, Roanoke, Virginia.
Disclosure: Shaheen E. Lakhan, MD, PhD, MS, MEd, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
Given the patient's results on the genetic panel and MRI, as well as the noted cognitive decline and increased aggression, this patient is suspected of having limbic-predominant age-related TDP-43 encephalopathy (LATE) secondary to AD and is referred to the neurologist on her multidisciplinary care team for further consultation and testing.
AD is one of the most common forms of dementia. More than 6 million people in the United States have clinical AD or mild cognitive impairment because of AD. LATE is a new classification of dementia, identified in 2019, that mimics AD but is a unique disease entity driven by the misfolding of the protein TDP-43, which regulates gene expression in the brain. Misfolded TDP-43 protein is common among older adults aged ≥ 85 years, and about a quarter of this population has enough misfolded TDP-43 protein to affect their memory and cognition.
Diagnosing AD currently relies on a clinical approach. A complete physical examination, with a detailed neurologic examination and a mental status examination, is used to evaluate disease stage. Initial mental status testing evaluates attention and concentration, recent and remote memory, language, praxis, executive function, and visuospatial function. Because LATE is a newly discovered form of dementia, there are no set guidelines on diagnosing LATE and no robust biomarker for TDP-43. What is known about LATE has been gleaned mostly from retrospective clinicopathologic studies.
The LATE consensus working group reports that the clinical course of disease, as studied by autopsy-proven LATE neuropathologic change (LATE-NC), is described as an "amnestic cognitive syndrome that can evolve to incorporate multiple cognitive domains and ultimately to impair activities of daily living." Researchers are currently analyzing different clinical assessments and neuroimaging with MRI to characterize LATE. A group of international researchers recently published a set of clinical criteria for limbic-predominant amnestic neurodegenerative syndrome (LANS), which is associated with LATE-NC. Their criteria include "core, standard and advanced features that are measurable in vivo, including older age at evaluation, mild clinical syndrome, disproportionate hippocampal atrophy, impaired semantic memory, limbic hypometabolism, absence of neocortical degenerative patterns and low likelihood of neocortical tau, with degrees of certainty (highest, high, moderate, low)." Other neuroimaging studies of autopsy-confirmed LATE-NC have shown that atrophy is mostly focused in the medial temporal lobe with marked reduced hippocampal volume.
The group reports that LATE and AD probably share pathophysiologic mechanisms. One of the universally accepted hallmarks of AD is the formation of beta-amyloid plaques, which are dense, mostly insoluble deposits of beta-amyloid protein that develop around neurons in the hippocampus and other regions in the cerebral cortex used for decision-making. These plaques disrupt brain function and lead to brain atrophy. The LATE group also reports that this same pathology has been noted with LATE: "Many subjects with LATE-NC have comorbid brain pathologies, often including amyloid-beta plaques and tauopathy." That said, genetic studies have helped identify five genes with risk alleles for LATE (GRN, TMEM106B, ABCC9, KCNMB2, and APOE), suggesting disease-specific underlying mechanisms compared to AD.
Patient and caregiver education and guidance is vital with a dementia diagnosis. If LATE and/or AD are suspected, physicians should encourage the involvement of family and friends who agree to become more involved in the patient's care as the disease progresses. These individuals need to understand the patient's wishes around care, especially for the future when the patient is no longer able to make decisions. The patient may also consider establishing medical advance directives and durable power of attorney for medical and financial decision-making. Caregivers supporting the patient are encouraged to help balance the physical needs of the patient while maintaining respect for them as a competent adult to the extent allowed by the progression of their disease.
Because LATE is a new classification of dementia, there are no known effective treatments. One ongoing study is testing the use of autologous bone marrow–derived stem cells to help improve cognitive impairment among patients with LATE, AD, and other dementias. Current AD treatments are focused on symptomatic therapies that modulate neurotransmitters — either acetylcholine or glutamate. The standard medical treatment includes cholinesterase inhibitors and a partial N-methyl-D-aspartate antagonist. Two amyloid-directed antibodies (aducanumab, lecanemab) are currently available in the United States for individuals with AD exhibiting mild cognitive impairment or mild dementia. A third agent currently in clinical trials (donanemab) has shown significantly slowed clinical progression after 1.5 years among clinical trial participants with early symptomatic AD and amyloid and tau pathology.
Shaheen E. Lakhan, MD, PhD, MS, MEd, Chief of Pain Management, Carilion Clinic and Virginia Tech Carilion School of Medicine, Roanoke, Virginia.
Disclosure: Shaheen E. Lakhan, MD, PhD, MS, MEd, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
Given the patient's results on the genetic panel and MRI, as well as the noted cognitive decline and increased aggression, this patient is suspected of having limbic-predominant age-related TDP-43 encephalopathy (LATE) secondary to AD and is referred to the neurologist on her multidisciplinary care team for further consultation and testing.
AD is one of the most common forms of dementia. More than 6 million people in the United States have clinical AD or mild cognitive impairment because of AD. LATE is a new classification of dementia, identified in 2019, that mimics AD but is a unique disease entity driven by the misfolding of the protein TDP-43, which regulates gene expression in the brain. Misfolded TDP-43 protein is common among older adults aged ≥ 85 years, and about a quarter of this population has enough misfolded TDP-43 protein to affect their memory and cognition.
Diagnosing AD currently relies on a clinical approach. A complete physical examination, with a detailed neurologic examination and a mental status examination, is used to evaluate disease stage. Initial mental status testing evaluates attention and concentration, recent and remote memory, language, praxis, executive function, and visuospatial function. Because LATE is a newly discovered form of dementia, there are no set guidelines on diagnosing LATE and no robust biomarker for TDP-43. What is known about LATE has been gleaned mostly from retrospective clinicopathologic studies.
The LATE consensus working group reports that the clinical course of disease, as studied by autopsy-proven LATE neuropathologic change (LATE-NC), is described as an "amnestic cognitive syndrome that can evolve to incorporate multiple cognitive domains and ultimately to impair activities of daily living." Researchers are currently analyzing different clinical assessments and neuroimaging with MRI to characterize LATE. A group of international researchers recently published a set of clinical criteria for limbic-predominant amnestic neurodegenerative syndrome (LANS), which is associated with LATE-NC. Their criteria include "core, standard and advanced features that are measurable in vivo, including older age at evaluation, mild clinical syndrome, disproportionate hippocampal atrophy, impaired semantic memory, limbic hypometabolism, absence of neocortical degenerative patterns and low likelihood of neocortical tau, with degrees of certainty (highest, high, moderate, low)." Other neuroimaging studies of autopsy-confirmed LATE-NC have shown that atrophy is mostly focused in the medial temporal lobe with marked reduced hippocampal volume.
The group reports that LATE and AD probably share pathophysiologic mechanisms. One of the universally accepted hallmarks of AD is the formation of beta-amyloid plaques, which are dense, mostly insoluble deposits of beta-amyloid protein that develop around neurons in the hippocampus and other regions in the cerebral cortex used for decision-making. These plaques disrupt brain function and lead to brain atrophy. The LATE group also reports that this same pathology has been noted with LATE: "Many subjects with LATE-NC have comorbid brain pathologies, often including amyloid-beta plaques and tauopathy." That said, genetic studies have helped identify five genes with risk alleles for LATE (GRN, TMEM106B, ABCC9, KCNMB2, and APOE), suggesting disease-specific underlying mechanisms compared to AD.
Patient and caregiver education and guidance is vital with a dementia diagnosis. If LATE and/or AD are suspected, physicians should encourage the involvement of family and friends who agree to become more involved in the patient's care as the disease progresses. These individuals need to understand the patient's wishes around care, especially for the future when the patient is no longer able to make decisions. The patient may also consider establishing medical advance directives and durable power of attorney for medical and financial decision-making. Caregivers supporting the patient are encouraged to help balance the physical needs of the patient while maintaining respect for them as a competent adult to the extent allowed by the progression of their disease.
Because LATE is a new classification of dementia, there are no known effective treatments. One ongoing study is testing the use of autologous bone marrow–derived stem cells to help improve cognitive impairment among patients with LATE, AD, and other dementias. Current AD treatments are focused on symptomatic therapies that modulate neurotransmitters — either acetylcholine or glutamate. The standard medical treatment includes cholinesterase inhibitors and a partial N-methyl-D-aspartate antagonist. Two amyloid-directed antibodies (aducanumab, lecanemab) are currently available in the United States for individuals with AD exhibiting mild cognitive impairment or mild dementia. A third agent currently in clinical trials (donanemab) has shown significantly slowed clinical progression after 1.5 years among clinical trial participants with early symptomatic AD and amyloid and tau pathology.
Shaheen E. Lakhan, MD, PhD, MS, MEd, Chief of Pain Management, Carilion Clinic and Virginia Tech Carilion School of Medicine, Roanoke, Virginia.
Disclosure: Shaheen E. Lakhan, MD, PhD, MS, MEd, has disclosed no relevant financial relationships.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
An 85-year-old woman presents to her geriatrician with her daughter, who is her primary caregiver. Seven years ago, the patient was diagnosed with mild Alzheimer's disease (AD). Her symptoms at diagnosis were irritability, forgetfulness, and panic attacks. Cognitive, behavioral, and functional assessments showed levels of decline; neurologic examination revealed mild hyposmia. The patient has been living with her daughter ever since her AD diagnosis.
At today's visit, the daughter reports that her mother has been experiencing loss of appetite and wide mood fluctuations with moments of unusual agitation. In addition, she tells the geriatrician that her mother has had trouble maintaining her balance and seems to have lost her sense of time. The patient has difficulty remembering what month and day it is, and how long it's been since her brother came to visit — which has been every Sunday like clockwork since the patient moved in with her daughter. The daughter also notes that her mother loses track of the story line when she is watching movie and TV shows lately.
The physician orders a brain MRI and genetic panel. MRI reveals atrophy in the frontal cortex as well as the medial temporal lobe, with hippocampal sclerosis. The genetic panel shows APOE and TMEM106 mutations.
Modifiable Risk Factors for Young-Onset Dementia Flagged
TOPLINE:
In addition to better known risk factors such as diabetes, stroke, heart disease, and depression, findings of a large study suggested vitamin D deficiency, elevated C-reactive protein (CRP) levels, and social isolation increase the risk for young-onset dementia (YOD).
METHODOLOGY:
- The study included 356,052 participants younger than 65 years (mean baseline age, 54.6 years) without dementia from the UK Biobank, an ongoing prospective cohort study.
- Participants underwent a comprehensive baseline assessment, provided biological samples, completed touch screen questionnaires, and underwent a physical examination.
- Researchers identified incident all-cause YOD cases from hospital inpatient registers or death register linkage.
- The researchers detected 39 potential risk factors and grouped them into domains of sociodemographic, genetic, lifestyle, environmental, vitamin D and CRP levels, cardiometabolic, psychiatric, and other factors.
- Researchers analyzed incidence rates of YOD for 5-year age bands starting at age 40 years and separately for men and women.
TAKEAWAY:
- During a mean follow-up of 8.12 years, there were 485 incident YOD cases (incidence rate of 16.8 per 100,000 person-years; 95% CI 15.4-18.3).
- The final analysis identified 15 risk factors associated with significantly higher incidence of YOD, including traditional factors like stroke (hazard ratio [HR], 2.07), heart disease (HR, 1.61), diabetes (HR, 1.65), and depression (HR, 3.25) but also less-recognized risk factors like vitamin D deficiency (< 10 ng/mL; HR, 1.59), high CRP levels (> 1 mg/dL; HR, 1.54), and social isolation (infrequent visits to friends or family; HR, 1.53), with lower socioeconomic status (HR, 1.82), having two apolipoprotein E epsilon-4 alleles (HR, 1.87), orthostatic hypotension, which the authors said may be an early sign of Parkinson dementia or Lewy body dementia (HR, 4.20), and hearing impairment (HR, 1.56) also increasing risk.
- Interestingly, some alcohol use seemed to be protective (moderate or heavy alcohol use had a lower association with YOD than alcohol abstinence, possibly due to the “healthy drinker effect” where people who drink are healthier than abstainers who may have illnesses preventing them from drinking, said the authors), as was higher education level and higher than normative handgrip strength (less strength is a proxy for physical frailty).
- Men with diabetes had higher YOD risk than those without diabetes, while there was no association with diabetes in women; on the other hand, women with high CRP levels had greater YOD risk than those with low levels, while there was no association with CRP in men.
IN PRACTICE:
“While further exploration of these risk factors is necessary to identify potential underlying mechanisms, addressing these modifiable factors may prove effective in mitigating the risk of developing YOD and can be readily integrated in current dementia prevention initiatives,” the investigators wrote.
SOURCE:
The study was led by Stevie Hendriks, PhD, Department of Psychiatry and Neuropsychology, Maastricht University, Maastricht, the Netherlands. It was published online in JAMA Neurology.
LIMITATIONS:
The study was observational and so can’t infer causality. Several factors were based on self-reported data, which might be a source of response bias. Factors not considered in the study, for example, family history of dementia and drug (other than alcohol) use disorder, may have confounded associations. Some factors including orthostatic hypotension had few exposed cases, leading to decreased power to detect associations. Hospital and death records may not have captured all YOD cases. The UK Biobank is overrepresented by healthy and White participants, so results may not be generalizable to other racial and ethnic groups. The analyses only focused on all-cause dementia.
DISCLOSURES:
The study was supported by Alzheimer Netherlands. Hendriks has no relevant conflicts of interest; see paper for disclosures of other authors.
A version of this article appeared on Medscape.com.
TOPLINE:
In addition to better known risk factors such as diabetes, stroke, heart disease, and depression, findings of a large study suggested vitamin D deficiency, elevated C-reactive protein (CRP) levels, and social isolation increase the risk for young-onset dementia (YOD).
METHODOLOGY:
- The study included 356,052 participants younger than 65 years (mean baseline age, 54.6 years) without dementia from the UK Biobank, an ongoing prospective cohort study.
- Participants underwent a comprehensive baseline assessment, provided biological samples, completed touch screen questionnaires, and underwent a physical examination.
- Researchers identified incident all-cause YOD cases from hospital inpatient registers or death register linkage.
- The researchers detected 39 potential risk factors and grouped them into domains of sociodemographic, genetic, lifestyle, environmental, vitamin D and CRP levels, cardiometabolic, psychiatric, and other factors.
- Researchers analyzed incidence rates of YOD for 5-year age bands starting at age 40 years and separately for men and women.
TAKEAWAY:
- During a mean follow-up of 8.12 years, there were 485 incident YOD cases (incidence rate of 16.8 per 100,000 person-years; 95% CI 15.4-18.3).
- The final analysis identified 15 risk factors associated with significantly higher incidence of YOD, including traditional factors like stroke (hazard ratio [HR], 2.07), heart disease (HR, 1.61), diabetes (HR, 1.65), and depression (HR, 3.25) but also less-recognized risk factors like vitamin D deficiency (< 10 ng/mL; HR, 1.59), high CRP levels (> 1 mg/dL; HR, 1.54), and social isolation (infrequent visits to friends or family; HR, 1.53), with lower socioeconomic status (HR, 1.82), having two apolipoprotein E epsilon-4 alleles (HR, 1.87), orthostatic hypotension, which the authors said may be an early sign of Parkinson dementia or Lewy body dementia (HR, 4.20), and hearing impairment (HR, 1.56) also increasing risk.
- Interestingly, some alcohol use seemed to be protective (moderate or heavy alcohol use had a lower association with YOD than alcohol abstinence, possibly due to the “healthy drinker effect” where people who drink are healthier than abstainers who may have illnesses preventing them from drinking, said the authors), as was higher education level and higher than normative handgrip strength (less strength is a proxy for physical frailty).
- Men with diabetes had higher YOD risk than those without diabetes, while there was no association with diabetes in women; on the other hand, women with high CRP levels had greater YOD risk than those with low levels, while there was no association with CRP in men.
IN PRACTICE:
“While further exploration of these risk factors is necessary to identify potential underlying mechanisms, addressing these modifiable factors may prove effective in mitigating the risk of developing YOD and can be readily integrated in current dementia prevention initiatives,” the investigators wrote.
SOURCE:
The study was led by Stevie Hendriks, PhD, Department of Psychiatry and Neuropsychology, Maastricht University, Maastricht, the Netherlands. It was published online in JAMA Neurology.
LIMITATIONS:
The study was observational and so can’t infer causality. Several factors were based on self-reported data, which might be a source of response bias. Factors not considered in the study, for example, family history of dementia and drug (other than alcohol) use disorder, may have confounded associations. Some factors including orthostatic hypotension had few exposed cases, leading to decreased power to detect associations. Hospital and death records may not have captured all YOD cases. The UK Biobank is overrepresented by healthy and White participants, so results may not be generalizable to other racial and ethnic groups. The analyses only focused on all-cause dementia.
DISCLOSURES:
The study was supported by Alzheimer Netherlands. Hendriks has no relevant conflicts of interest; see paper for disclosures of other authors.
A version of this article appeared on Medscape.com.
TOPLINE:
In addition to better known risk factors such as diabetes, stroke, heart disease, and depression, findings of a large study suggested vitamin D deficiency, elevated C-reactive protein (CRP) levels, and social isolation increase the risk for young-onset dementia (YOD).
METHODOLOGY:
- The study included 356,052 participants younger than 65 years (mean baseline age, 54.6 years) without dementia from the UK Biobank, an ongoing prospective cohort study.
- Participants underwent a comprehensive baseline assessment, provided biological samples, completed touch screen questionnaires, and underwent a physical examination.
- Researchers identified incident all-cause YOD cases from hospital inpatient registers or death register linkage.
- The researchers detected 39 potential risk factors and grouped them into domains of sociodemographic, genetic, lifestyle, environmental, vitamin D and CRP levels, cardiometabolic, psychiatric, and other factors.
- Researchers analyzed incidence rates of YOD for 5-year age bands starting at age 40 years and separately for men and women.
TAKEAWAY:
- During a mean follow-up of 8.12 years, there were 485 incident YOD cases (incidence rate of 16.8 per 100,000 person-years; 95% CI 15.4-18.3).
- The final analysis identified 15 risk factors associated with significantly higher incidence of YOD, including traditional factors like stroke (hazard ratio [HR], 2.07), heart disease (HR, 1.61), diabetes (HR, 1.65), and depression (HR, 3.25) but also less-recognized risk factors like vitamin D deficiency (< 10 ng/mL; HR, 1.59), high CRP levels (> 1 mg/dL; HR, 1.54), and social isolation (infrequent visits to friends or family; HR, 1.53), with lower socioeconomic status (HR, 1.82), having two apolipoprotein E epsilon-4 alleles (HR, 1.87), orthostatic hypotension, which the authors said may be an early sign of Parkinson dementia or Lewy body dementia (HR, 4.20), and hearing impairment (HR, 1.56) also increasing risk.
- Interestingly, some alcohol use seemed to be protective (moderate or heavy alcohol use had a lower association with YOD than alcohol abstinence, possibly due to the “healthy drinker effect” where people who drink are healthier than abstainers who may have illnesses preventing them from drinking, said the authors), as was higher education level and higher than normative handgrip strength (less strength is a proxy for physical frailty).
- Men with diabetes had higher YOD risk than those without diabetes, while there was no association with diabetes in women; on the other hand, women with high CRP levels had greater YOD risk than those with low levels, while there was no association with CRP in men.
IN PRACTICE:
“While further exploration of these risk factors is necessary to identify potential underlying mechanisms, addressing these modifiable factors may prove effective in mitigating the risk of developing YOD and can be readily integrated in current dementia prevention initiatives,” the investigators wrote.
SOURCE:
The study was led by Stevie Hendriks, PhD, Department of Psychiatry and Neuropsychology, Maastricht University, Maastricht, the Netherlands. It was published online in JAMA Neurology.
LIMITATIONS:
The study was observational and so can’t infer causality. Several factors were based on self-reported data, which might be a source of response bias. Factors not considered in the study, for example, family history of dementia and drug (other than alcohol) use disorder, may have confounded associations. Some factors including orthostatic hypotension had few exposed cases, leading to decreased power to detect associations. Hospital and death records may not have captured all YOD cases. The UK Biobank is overrepresented by healthy and White participants, so results may not be generalizable to other racial and ethnic groups. The analyses only focused on all-cause dementia.
DISCLOSURES:
The study was supported by Alzheimer Netherlands. Hendriks has no relevant conflicts of interest; see paper for disclosures of other authors.
A version of this article appeared on Medscape.com.
Poor Oral Health Tied to Worse Brain Health
In a large observational study of middle-aged adults without stroke or dementia, poor oral health was strongly associated with multiple neuroimaging markers of white matter injury.
“Because the neuroimaging markers evaluated in this study precede and are established risk factors of stroke and dementia, our results suggest that oral health, an easily modifiable process, may be a promising target for very early interventions focused on improving brain health,” wrote the authors, led by Cyprien A. Rivier, MD, MS, with the Department of Neurology, Yale University School of Medicine, New Haven, Connecticut.
The study was published online on December 20, 2023, in Neurology.
Research data came from 40,175 adults (mean age, 55 years; 53% women) with no history of stroke or dementia who enrolled in the UK Biobank from 2006 to 2010 and had brain MRI between 2014 and 2016.
Altogether, 5470 (14%) participants had poor oral health, defined as the presence of dentures or loose teeth. Those with poor (vs optimal) oral health were older, more likely to be male, and had higher prevalence of hypertension, hypercholesterolemia, diabetes, overweight/obesity, and current or past smoking history.
In a multivariable model, poor oral health was associated with a 9% increase in white matter hyperintensity (WMH) volume (P < .001), a well-established marker of clinically silent cerebrovascular disease.
Poor oral health was also associated with a 10% change in aggregate fractional anisotropy (FA) score (P < .001) and a 5% change in aggregate mean diffusivity (MD) score (P < .001), two diffusion tensor imaging metrics that accurately represent white matter disintegrity.
Genetic analyses using Mendelian randomization confirmed these associations. Individuals who were genetically prone to poor oral health had a 30% increase in WMH volume (P < .001), 43% change in aggregate FA score (P < .001), and 10% change in aggregate MD score (P < .01), the researchers reported.
These findings, they noted, add to prior epidemiologic evidence for an association between poor oral health and a higher risk for clinical outcomes related to brain health, including cognitive decline.
‘Huge Dividends’
The authors of an accompanying editorial praised the authors for looking at the consequences of poor oral health in a “new and powerful way by using as their outcome MRI-defined white matter injury, which is associated with, but antedates by many years, cognitive decline and stroke.”
“The fact that these imaging changes are seen in asymptomatic persons offers the hope that if the association is causal, interventions to improve oral health could pay huge dividends in subsequent brain health,” wrote Steven J. Kittner, MD, MPH, and Breana L. Taylor, MD, with the Department of Neurology, University of Maryland School of Medicine in Baltimore.
“The mechanisms mediating the relationship between the oral health genetic risk score and white matter injury are likely to be complex, but the authors have taken an important step forward in addressing a hypothesis of immense public health importance,” they added.
Data from the World Health Organization suggested that oral diseases, which are largely preventable, affect nearly 3.5 billion people globally, with three out of four people affected in middle-income countries.
Funding for the study was provided in part by grants from the National Institutes of Health, the American Heart Association, and the Neurocritical Care Society Research Fellowship. The authors and editorialists disclosed no relevant conflicts of interest.
Megan Brooks has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
In a large observational study of middle-aged adults without stroke or dementia, poor oral health was strongly associated with multiple neuroimaging markers of white matter injury.
“Because the neuroimaging markers evaluated in this study precede and are established risk factors of stroke and dementia, our results suggest that oral health, an easily modifiable process, may be a promising target for very early interventions focused on improving brain health,” wrote the authors, led by Cyprien A. Rivier, MD, MS, with the Department of Neurology, Yale University School of Medicine, New Haven, Connecticut.
The study was published online on December 20, 2023, in Neurology.
Research data came from 40,175 adults (mean age, 55 years; 53% women) with no history of stroke or dementia who enrolled in the UK Biobank from 2006 to 2010 and had brain MRI between 2014 and 2016.
Altogether, 5470 (14%) participants had poor oral health, defined as the presence of dentures or loose teeth. Those with poor (vs optimal) oral health were older, more likely to be male, and had higher prevalence of hypertension, hypercholesterolemia, diabetes, overweight/obesity, and current or past smoking history.
In a multivariable model, poor oral health was associated with a 9% increase in white matter hyperintensity (WMH) volume (P < .001), a well-established marker of clinically silent cerebrovascular disease.
Poor oral health was also associated with a 10% change in aggregate fractional anisotropy (FA) score (P < .001) and a 5% change in aggregate mean diffusivity (MD) score (P < .001), two diffusion tensor imaging metrics that accurately represent white matter disintegrity.
Genetic analyses using Mendelian randomization confirmed these associations. Individuals who were genetically prone to poor oral health had a 30% increase in WMH volume (P < .001), 43% change in aggregate FA score (P < .001), and 10% change in aggregate MD score (P < .01), the researchers reported.
These findings, they noted, add to prior epidemiologic evidence for an association between poor oral health and a higher risk for clinical outcomes related to brain health, including cognitive decline.
‘Huge Dividends’
The authors of an accompanying editorial praised the authors for looking at the consequences of poor oral health in a “new and powerful way by using as their outcome MRI-defined white matter injury, which is associated with, but antedates by many years, cognitive decline and stroke.”
“The fact that these imaging changes are seen in asymptomatic persons offers the hope that if the association is causal, interventions to improve oral health could pay huge dividends in subsequent brain health,” wrote Steven J. Kittner, MD, MPH, and Breana L. Taylor, MD, with the Department of Neurology, University of Maryland School of Medicine in Baltimore.
“The mechanisms mediating the relationship between the oral health genetic risk score and white matter injury are likely to be complex, but the authors have taken an important step forward in addressing a hypothesis of immense public health importance,” they added.
Data from the World Health Organization suggested that oral diseases, which are largely preventable, affect nearly 3.5 billion people globally, with three out of four people affected in middle-income countries.
Funding for the study was provided in part by grants from the National Institutes of Health, the American Heart Association, and the Neurocritical Care Society Research Fellowship. The authors and editorialists disclosed no relevant conflicts of interest.
Megan Brooks has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
In a large observational study of middle-aged adults without stroke or dementia, poor oral health was strongly associated with multiple neuroimaging markers of white matter injury.
“Because the neuroimaging markers evaluated in this study precede and are established risk factors of stroke and dementia, our results suggest that oral health, an easily modifiable process, may be a promising target for very early interventions focused on improving brain health,” wrote the authors, led by Cyprien A. Rivier, MD, MS, with the Department of Neurology, Yale University School of Medicine, New Haven, Connecticut.
The study was published online on December 20, 2023, in Neurology.
Research data came from 40,175 adults (mean age, 55 years; 53% women) with no history of stroke or dementia who enrolled in the UK Biobank from 2006 to 2010 and had brain MRI between 2014 and 2016.
Altogether, 5470 (14%) participants had poor oral health, defined as the presence of dentures or loose teeth. Those with poor (vs optimal) oral health were older, more likely to be male, and had higher prevalence of hypertension, hypercholesterolemia, diabetes, overweight/obesity, and current or past smoking history.
In a multivariable model, poor oral health was associated with a 9% increase in white matter hyperintensity (WMH) volume (P < .001), a well-established marker of clinically silent cerebrovascular disease.
Poor oral health was also associated with a 10% change in aggregate fractional anisotropy (FA) score (P < .001) and a 5% change in aggregate mean diffusivity (MD) score (P < .001), two diffusion tensor imaging metrics that accurately represent white matter disintegrity.
Genetic analyses using Mendelian randomization confirmed these associations. Individuals who were genetically prone to poor oral health had a 30% increase in WMH volume (P < .001), 43% change in aggregate FA score (P < .001), and 10% change in aggregate MD score (P < .01), the researchers reported.
These findings, they noted, add to prior epidemiologic evidence for an association between poor oral health and a higher risk for clinical outcomes related to brain health, including cognitive decline.
‘Huge Dividends’
The authors of an accompanying editorial praised the authors for looking at the consequences of poor oral health in a “new and powerful way by using as their outcome MRI-defined white matter injury, which is associated with, but antedates by many years, cognitive decline and stroke.”
“The fact that these imaging changes are seen in asymptomatic persons offers the hope that if the association is causal, interventions to improve oral health could pay huge dividends in subsequent brain health,” wrote Steven J. Kittner, MD, MPH, and Breana L. Taylor, MD, with the Department of Neurology, University of Maryland School of Medicine in Baltimore.
“The mechanisms mediating the relationship between the oral health genetic risk score and white matter injury are likely to be complex, but the authors have taken an important step forward in addressing a hypothesis of immense public health importance,” they added.
Data from the World Health Organization suggested that oral diseases, which are largely preventable, affect nearly 3.5 billion people globally, with three out of four people affected in middle-income countries.
Funding for the study was provided in part by grants from the National Institutes of Health, the American Heart Association, and the Neurocritical Care Society Research Fellowship. The authors and editorialists disclosed no relevant conflicts of interest.
Megan Brooks has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Epilepsy Linked to Earlier, More Rapid, Cognitive Decline
ORLANDO — People with epilepsy are more likely to decline cognitively compared with those without epilepsy, new research suggests.
Results of the large, longitudinal study show that seizures predicted earlier conversion time from normal cognition to mild cognitive impairment (MCI) but were not associated with conversion from MCI to dementia.
“Modifiable cardiovascular risk factors such as hypertension and diabetes need to be treated more aggressively because they can impact cognition, but epilepsy is another risk factor that needs to be treated in a timely fashion because it appears to be also associated with cognitive impairment,” said study investigator Ifrah Zawar MD, assistant professor, Department of Neurology, University of Virginia in Charlottesville.
The study (abstract #2.172) was presented on December 2 at the American Epilepsy Society annual meeting.
An Understudied Issue
Comorbid seizures occur in up to 64% of those with dementia, and patients with dementia and epilepsy have a more aggressive disease course, faster cognitive decline, and more severe neuronal loss, Dr. Zawar told Medscape Medical News.
But the impact of seizures on the conversion of cognitively healthy to MCI and from MCI to dementia, after accounting for cardiovascular risk factors, has not been well studied.
Researchers analyzed longitudinal data of 13,726 patients, mean age about 70 years, who were cognitively healthy or had mild cognitive impairment (MCI). Participants were recruited from 39 Alzheimer’s Disease (AD) centers in the United States from 2005 to 2021.
Investigators categorized participants into three groups: active (having had seizures in the past year and/or requiring active treatment; N = 118), resolved (not on any treatment for the past year and not having seizures; N = 226), and no seizures (never having had seizures; N = 13,382).
The primary outcome was conversion from cognitively healthy to MCI/dementia and from MCI to dementia in those with and without active epilepsy and resolved epilepsy.
Factors associated with conversion from cognitively healthy to MCI among those with current or active epilepsy included older age (P <.001 for ages 60-80 years and P =.002 for age 80 years or older vs younger than 60 years), male sex (P <.001), lower education (P <.001), hypertension (P <.001), and diabetes (P <.001).
The hazard ratio (HR) for earlier conversion from healthy to worse cognition among those with active epilepsy was 1.76 (95% CI, 1.38-2.24; P <.001), even after accounting for risk factors.
Kaplan-Meier curves showed that the median time to convert from healthy cognition to MCI among people with active epilepsy was about 5 years compared with about 9 years for those with resolved epilepsy and 10.5 years for those without epilepsy.
The story was similar for faster conversion from MCI to dementia. Compared with having no epilepsy, the HR for faster conversion for active epilepsy was 1.44 (95% CI, 1.20-1.73; P <.001).
In addition, the median time to conversion from MCI to dementia was about 3 years for those with active epilepsy compared with about 5 years for those with resolved epilepsy and about 5 years for those without epilepsy.
“It’s important for physicians to understand that uncontrolled epilepsy or active epilepsy is going to impact patients’ cognition adversely, which in itself is associated with increased comorbidity and mortality,” said Dr. Zawar.
The mechanism driving the acceleration to worse cognition in people with epilepsy is “complicated and involves a multitude of factors,” she said.
The researchers did not specifically investigate how use of antiseizure medications correlated with cognitive outcomes, but Dr. Zawar believes that “epilepsy in itself impacts cognition.”
The researchers also didn’t have EEG data for study participants who were recruited from Alzheimer’s disease centers where EEGs aren’t routinely carried out, so such data for many patients may not necessarily exist, said Dr. Zawar.
Important Research
Commenting for this news organization, Bruce Hermann, PhD, professor emeritus, Department of Neurology, University of Wisconsin School of Medicine and Public Health, said that the study is important because of the, “tremendous interest and concern about aging with epilepsy.”
“We want to know how people with chronic epilepsy age cognitively and what’s the cognitive course of those who have late onset epilepsy, particularly those with unknown etiology,” he added.
Dr. Hermann noted that much of the research in this area has been relatively small and single-center investigations.
“These larger-scale investigations from outside the epilepsy community are so important because they have data on large numbers of subjects, they have cognitive data, and follow-ups over long periods of time, and they’re providing some really novel information,” Dr. Hermann said.
He added that terms used in the dementia world such as MCI and frank dementia are somewhat foreign to epileptologists. In addition, interventions to delay, treat, or prevent cognitive decline such as exercise, diet, social activity, and mental stimulation that are regularly discussed by dementia experts are underrepresented in the epilepsy world.
“The things they talk about in memory clinics in the aging world almost routinely have not penetrated to the epilepsy clinics for aging individuals and for the epilepsy community in general.”
The study used the Montreal Cognitive Assessment to identify cognitive decline. “It would be nice to see how these people look with traditional neuropsychological tests,” said Dr. Hermann.
He added that information on the impact of epilepsy on different MCI phenotypes, for example, pure memory impairment subtype; pure nonmemory subtype; and multiple domain subtype, would also be useful.
The study was supported by the AES and the Alzheimer’s Association.
Dr. Zawar and Dr. Hermann report no relevant disclosures.
A version of this article appeared on Medscape.com.
ORLANDO — People with epilepsy are more likely to decline cognitively compared with those without epilepsy, new research suggests.
Results of the large, longitudinal study show that seizures predicted earlier conversion time from normal cognition to mild cognitive impairment (MCI) but were not associated with conversion from MCI to dementia.
“Modifiable cardiovascular risk factors such as hypertension and diabetes need to be treated more aggressively because they can impact cognition, but epilepsy is another risk factor that needs to be treated in a timely fashion because it appears to be also associated with cognitive impairment,” said study investigator Ifrah Zawar MD, assistant professor, Department of Neurology, University of Virginia in Charlottesville.
The study (abstract #2.172) was presented on December 2 at the American Epilepsy Society annual meeting.
An Understudied Issue
Comorbid seizures occur in up to 64% of those with dementia, and patients with dementia and epilepsy have a more aggressive disease course, faster cognitive decline, and more severe neuronal loss, Dr. Zawar told Medscape Medical News.
But the impact of seizures on the conversion of cognitively healthy to MCI and from MCI to dementia, after accounting for cardiovascular risk factors, has not been well studied.
Researchers analyzed longitudinal data of 13,726 patients, mean age about 70 years, who were cognitively healthy or had mild cognitive impairment (MCI). Participants were recruited from 39 Alzheimer’s Disease (AD) centers in the United States from 2005 to 2021.
Investigators categorized participants into three groups: active (having had seizures in the past year and/or requiring active treatment; N = 118), resolved (not on any treatment for the past year and not having seizures; N = 226), and no seizures (never having had seizures; N = 13,382).
The primary outcome was conversion from cognitively healthy to MCI/dementia and from MCI to dementia in those with and without active epilepsy and resolved epilepsy.
Factors associated with conversion from cognitively healthy to MCI among those with current or active epilepsy included older age (P <.001 for ages 60-80 years and P =.002 for age 80 years or older vs younger than 60 years), male sex (P <.001), lower education (P <.001), hypertension (P <.001), and diabetes (P <.001).
The hazard ratio (HR) for earlier conversion from healthy to worse cognition among those with active epilepsy was 1.76 (95% CI, 1.38-2.24; P <.001), even after accounting for risk factors.
Kaplan-Meier curves showed that the median time to convert from healthy cognition to MCI among people with active epilepsy was about 5 years compared with about 9 years for those with resolved epilepsy and 10.5 years for those without epilepsy.
The story was similar for faster conversion from MCI to dementia. Compared with having no epilepsy, the HR for faster conversion for active epilepsy was 1.44 (95% CI, 1.20-1.73; P <.001).
In addition, the median time to conversion from MCI to dementia was about 3 years for those with active epilepsy compared with about 5 years for those with resolved epilepsy and about 5 years for those without epilepsy.
“It’s important for physicians to understand that uncontrolled epilepsy or active epilepsy is going to impact patients’ cognition adversely, which in itself is associated with increased comorbidity and mortality,” said Dr. Zawar.
The mechanism driving the acceleration to worse cognition in people with epilepsy is “complicated and involves a multitude of factors,” she said.
The researchers did not specifically investigate how use of antiseizure medications correlated with cognitive outcomes, but Dr. Zawar believes that “epilepsy in itself impacts cognition.”
The researchers also didn’t have EEG data for study participants who were recruited from Alzheimer’s disease centers where EEGs aren’t routinely carried out, so such data for many patients may not necessarily exist, said Dr. Zawar.
Important Research
Commenting for this news organization, Bruce Hermann, PhD, professor emeritus, Department of Neurology, University of Wisconsin School of Medicine and Public Health, said that the study is important because of the, “tremendous interest and concern about aging with epilepsy.”
“We want to know how people with chronic epilepsy age cognitively and what’s the cognitive course of those who have late onset epilepsy, particularly those with unknown etiology,” he added.
Dr. Hermann noted that much of the research in this area has been relatively small and single-center investigations.
“These larger-scale investigations from outside the epilepsy community are so important because they have data on large numbers of subjects, they have cognitive data, and follow-ups over long periods of time, and they’re providing some really novel information,” Dr. Hermann said.
He added that terms used in the dementia world such as MCI and frank dementia are somewhat foreign to epileptologists. In addition, interventions to delay, treat, or prevent cognitive decline such as exercise, diet, social activity, and mental stimulation that are regularly discussed by dementia experts are underrepresented in the epilepsy world.
“The things they talk about in memory clinics in the aging world almost routinely have not penetrated to the epilepsy clinics for aging individuals and for the epilepsy community in general.”
The study used the Montreal Cognitive Assessment to identify cognitive decline. “It would be nice to see how these people look with traditional neuropsychological tests,” said Dr. Hermann.
He added that information on the impact of epilepsy on different MCI phenotypes, for example, pure memory impairment subtype; pure nonmemory subtype; and multiple domain subtype, would also be useful.
The study was supported by the AES and the Alzheimer’s Association.
Dr. Zawar and Dr. Hermann report no relevant disclosures.
A version of this article appeared on Medscape.com.
ORLANDO — People with epilepsy are more likely to decline cognitively compared with those without epilepsy, new research suggests.
Results of the large, longitudinal study show that seizures predicted earlier conversion time from normal cognition to mild cognitive impairment (MCI) but were not associated with conversion from MCI to dementia.
“Modifiable cardiovascular risk factors such as hypertension and diabetes need to be treated more aggressively because they can impact cognition, but epilepsy is another risk factor that needs to be treated in a timely fashion because it appears to be also associated with cognitive impairment,” said study investigator Ifrah Zawar MD, assistant professor, Department of Neurology, University of Virginia in Charlottesville.
The study (abstract #2.172) was presented on December 2 at the American Epilepsy Society annual meeting.
An Understudied Issue
Comorbid seizures occur in up to 64% of those with dementia, and patients with dementia and epilepsy have a more aggressive disease course, faster cognitive decline, and more severe neuronal loss, Dr. Zawar told Medscape Medical News.
But the impact of seizures on the conversion of cognitively healthy to MCI and from MCI to dementia, after accounting for cardiovascular risk factors, has not been well studied.
Researchers analyzed longitudinal data of 13,726 patients, mean age about 70 years, who were cognitively healthy or had mild cognitive impairment (MCI). Participants were recruited from 39 Alzheimer’s Disease (AD) centers in the United States from 2005 to 2021.
Investigators categorized participants into three groups: active (having had seizures in the past year and/or requiring active treatment; N = 118), resolved (not on any treatment for the past year and not having seizures; N = 226), and no seizures (never having had seizures; N = 13,382).
The primary outcome was conversion from cognitively healthy to MCI/dementia and from MCI to dementia in those with and without active epilepsy and resolved epilepsy.
Factors associated with conversion from cognitively healthy to MCI among those with current or active epilepsy included older age (P <.001 for ages 60-80 years and P =.002 for age 80 years or older vs younger than 60 years), male sex (P <.001), lower education (P <.001), hypertension (P <.001), and diabetes (P <.001).
The hazard ratio (HR) for earlier conversion from healthy to worse cognition among those with active epilepsy was 1.76 (95% CI, 1.38-2.24; P <.001), even after accounting for risk factors.
Kaplan-Meier curves showed that the median time to convert from healthy cognition to MCI among people with active epilepsy was about 5 years compared with about 9 years for those with resolved epilepsy and 10.5 years for those without epilepsy.
The story was similar for faster conversion from MCI to dementia. Compared with having no epilepsy, the HR for faster conversion for active epilepsy was 1.44 (95% CI, 1.20-1.73; P <.001).
In addition, the median time to conversion from MCI to dementia was about 3 years for those with active epilepsy compared with about 5 years for those with resolved epilepsy and about 5 years for those without epilepsy.
“It’s important for physicians to understand that uncontrolled epilepsy or active epilepsy is going to impact patients’ cognition adversely, which in itself is associated with increased comorbidity and mortality,” said Dr. Zawar.
The mechanism driving the acceleration to worse cognition in people with epilepsy is “complicated and involves a multitude of factors,” she said.
The researchers did not specifically investigate how use of antiseizure medications correlated with cognitive outcomes, but Dr. Zawar believes that “epilepsy in itself impacts cognition.”
The researchers also didn’t have EEG data for study participants who were recruited from Alzheimer’s disease centers where EEGs aren’t routinely carried out, so such data for many patients may not necessarily exist, said Dr. Zawar.
Important Research
Commenting for this news organization, Bruce Hermann, PhD, professor emeritus, Department of Neurology, University of Wisconsin School of Medicine and Public Health, said that the study is important because of the, “tremendous interest and concern about aging with epilepsy.”
“We want to know how people with chronic epilepsy age cognitively and what’s the cognitive course of those who have late onset epilepsy, particularly those with unknown etiology,” he added.
Dr. Hermann noted that much of the research in this area has been relatively small and single-center investigations.
“These larger-scale investigations from outside the epilepsy community are so important because they have data on large numbers of subjects, they have cognitive data, and follow-ups over long periods of time, and they’re providing some really novel information,” Dr. Hermann said.
He added that terms used in the dementia world such as MCI and frank dementia are somewhat foreign to epileptologists. In addition, interventions to delay, treat, or prevent cognitive decline such as exercise, diet, social activity, and mental stimulation that are regularly discussed by dementia experts are underrepresented in the epilepsy world.
“The things they talk about in memory clinics in the aging world almost routinely have not penetrated to the epilepsy clinics for aging individuals and for the epilepsy community in general.”
The study used the Montreal Cognitive Assessment to identify cognitive decline. “It would be nice to see how these people look with traditional neuropsychological tests,” said Dr. Hermann.
He added that information on the impact of epilepsy on different MCI phenotypes, for example, pure memory impairment subtype; pure nonmemory subtype; and multiple domain subtype, would also be useful.
The study was supported by the AES and the Alzheimer’s Association.
Dr. Zawar and Dr. Hermann report no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM AES 2023
Regular Physical Activity Linked to Larger Brain Volume
TOPLINE:
, new data suggest.
METHODOLOGY:
- The potential neuroprotective effects of regular physical activity on brain structure are unclear despite reported links between physical activity and reduced dementia risk.
- To investigate, researchers analyzed MRI brain scans from 10,125 healthy adults (mean age, 53 years; 52% male) who self-reported their level of physical activity.
- Moderate to vigorous physical activities, defined as those increasing respiration and pulse rate for at least 10 continuous minutes, was modeled with brain volumes, adjusting for covariates.
- The threshold for defining physically active (vs nonactive) adults was intentionally set at 2.5 days per week, a level far lower than current guidelines.
TAKEAWAY:
- Three quarters of the cohort reported engaging in moderate to vigorous physical activity approximately 4 days per week.
- Physically active adults tended to be younger, with a higher proportion of White individuals, and with lower rates of hypertension and type 2 diabetes.
- After adjusting for multiple factors, increased days of moderate to vigorous activity correlated with larger normalized brain volume in multiple regions including total gray matter; white matter; hippocampus; and frontal, parietal, and occipital lobes.
IN PRACTICE:
“We found that even moderate levels of physical activity, such as taking fewer than 4,000 steps a day, can have a positive effect on brain health. This is much less than the often-suggested 10,000 steps, making it a more achievable goal for many people,” co-author David Merrill, MD, with Pacific Brain Health Center, Santa Monica, California, said in a statement.
SOURCE:
The study, with first author Cyrus A. Raji, MD, PhD, Washington University School of Medicine, St. Louis, was published online in the Journal of Alzheimer’s Disease.
LIMITATIONS:
Participants self-reported physical activity in the past 2 weeks, which does not reflect a lifetime of activity levels. The correlation identified between physical activity and brain volumes may not be solely attributable to physical activity alone.
DISCLOSURES:
The study received funding from several health centers and foundations. Dr. Raji consults for Brainreader ApS, Neurevolution LLC, Apollo Health, Voxelwise Imaging Technology, and Pacific Neuroscience Foundation and is an editorial board member of the Journal of Alzheimer’s Disease but was not involved in the peer-review process.
A version of this article appeared on Medscape.com.
TOPLINE:
, new data suggest.
METHODOLOGY:
- The potential neuroprotective effects of regular physical activity on brain structure are unclear despite reported links between physical activity and reduced dementia risk.
- To investigate, researchers analyzed MRI brain scans from 10,125 healthy adults (mean age, 53 years; 52% male) who self-reported their level of physical activity.
- Moderate to vigorous physical activities, defined as those increasing respiration and pulse rate for at least 10 continuous minutes, was modeled with brain volumes, adjusting for covariates.
- The threshold for defining physically active (vs nonactive) adults was intentionally set at 2.5 days per week, a level far lower than current guidelines.
TAKEAWAY:
- Three quarters of the cohort reported engaging in moderate to vigorous physical activity approximately 4 days per week.
- Physically active adults tended to be younger, with a higher proportion of White individuals, and with lower rates of hypertension and type 2 diabetes.
- After adjusting for multiple factors, increased days of moderate to vigorous activity correlated with larger normalized brain volume in multiple regions including total gray matter; white matter; hippocampus; and frontal, parietal, and occipital lobes.
IN PRACTICE:
“We found that even moderate levels of physical activity, such as taking fewer than 4,000 steps a day, can have a positive effect on brain health. This is much less than the often-suggested 10,000 steps, making it a more achievable goal for many people,” co-author David Merrill, MD, with Pacific Brain Health Center, Santa Monica, California, said in a statement.
SOURCE:
The study, with first author Cyrus A. Raji, MD, PhD, Washington University School of Medicine, St. Louis, was published online in the Journal of Alzheimer’s Disease.
LIMITATIONS:
Participants self-reported physical activity in the past 2 weeks, which does not reflect a lifetime of activity levels. The correlation identified between physical activity and brain volumes may not be solely attributable to physical activity alone.
DISCLOSURES:
The study received funding from several health centers and foundations. Dr. Raji consults for Brainreader ApS, Neurevolution LLC, Apollo Health, Voxelwise Imaging Technology, and Pacific Neuroscience Foundation and is an editorial board member of the Journal of Alzheimer’s Disease but was not involved in the peer-review process.
A version of this article appeared on Medscape.com.
TOPLINE:
, new data suggest.
METHODOLOGY:
- The potential neuroprotective effects of regular physical activity on brain structure are unclear despite reported links between physical activity and reduced dementia risk.
- To investigate, researchers analyzed MRI brain scans from 10,125 healthy adults (mean age, 53 years; 52% male) who self-reported their level of physical activity.
- Moderate to vigorous physical activities, defined as those increasing respiration and pulse rate for at least 10 continuous minutes, was modeled with brain volumes, adjusting for covariates.
- The threshold for defining physically active (vs nonactive) adults was intentionally set at 2.5 days per week, a level far lower than current guidelines.
TAKEAWAY:
- Three quarters of the cohort reported engaging in moderate to vigorous physical activity approximately 4 days per week.
- Physically active adults tended to be younger, with a higher proportion of White individuals, and with lower rates of hypertension and type 2 diabetes.
- After adjusting for multiple factors, increased days of moderate to vigorous activity correlated with larger normalized brain volume in multiple regions including total gray matter; white matter; hippocampus; and frontal, parietal, and occipital lobes.
IN PRACTICE:
“We found that even moderate levels of physical activity, such as taking fewer than 4,000 steps a day, can have a positive effect on brain health. This is much less than the often-suggested 10,000 steps, making it a more achievable goal for many people,” co-author David Merrill, MD, with Pacific Brain Health Center, Santa Monica, California, said in a statement.
SOURCE:
The study, with first author Cyrus A. Raji, MD, PhD, Washington University School of Medicine, St. Louis, was published online in the Journal of Alzheimer’s Disease.
LIMITATIONS:
Participants self-reported physical activity in the past 2 weeks, which does not reflect a lifetime of activity levels. The correlation identified between physical activity and brain volumes may not be solely attributable to physical activity alone.
DISCLOSURES:
The study received funding from several health centers and foundations. Dr. Raji consults for Brainreader ApS, Neurevolution LLC, Apollo Health, Voxelwise Imaging Technology, and Pacific Neuroscience Foundation and is an editorial board member of the Journal of Alzheimer’s Disease but was not involved in the peer-review process.
A version of this article appeared on Medscape.com.
H pylori Infection Linked to Increased Alzheimer’s Risk
TOPLINE:
results of a large and lengthy population-based study suggest.
METHODOLOGY:
- Researchers identified all cases with a first-time diagnosis of AD and matched each AD case to up to 40 AD-free control cases on the basis of age, sex, cohort entry date, and duration of follow-up.
- The exposure of interest was CAHPI, defined based on an algorithm using clinical guidelines and recommendations on the management of H pylori (HP) infection, with researchers focusing on infected individuals presenting with symptoms or developing serious complications from the infection.
- Researchers performed several sensitivity analyses, which included repeating the primary analysis using alternate lag periods, restricting the cohort to participants with AD (not vascular, alcoholic, and unspecified dementia), and using salmonellosis, an infection not previously associated with AD, as a negative control exposure.
TAKEAWAY:
- Compared with no exposure to CAHPI, exposure to CAHPI was associated with a moderately increased risk for AD (odds ratio [OR], 1.11; 95% CI, 1.01-1.21), with no major effect modification by demographics or socioeconomic status.
- The increased risk peaked 7.3-10.8 years after CAHPI onset (OR, 1.24; 95% CI, 1.05-1.47) before decreasing.
- Sensitivity analyses yielded findings that were overall consistent with those of the primary analysis.
- The analysis with salmonellosis as a negative control exposure showed no association with the risk for AD (OR, 1.03; 95% CI, 0.82-1.29).
IN PRACTICE:
“These results support the notion of HP infection as a potential modifiable risk factor of AD” and “pave the way for future randomized controlled trials that would assess the impact and cost-effectiveness of population-based targeted interventions such as individualized HP eradication programs, on the development of AD,” the authors write.
SOURCE:
The study was conducted by Antonios Douros, Department of Medicine, and Department of Epidemiology, Biostatistics, and Occupational Health, McGill University, Montreal, Quebec, Canada, and colleagues. It was published online in Alzheimer’s & Dementia.
LIMITATIONS:
Given the observational nature of the study, residual confounding is possible. Because the exposure definition was on the basis of CAHPI recorded by general practitioners, exposure misclassification due to symptomatic patients not seeking primary care is possible, as is outcome misclassification. The authors can’t rule out the possibility of an association between asymptomatic H pylori infection and AD risk.
DISCLOSURES:
The study received funding from the Canadian Institutes of Health Research. Douros has no relevant conflicts of interest; see paper for disclosures of other authors.
Pauline Anderson has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
results of a large and lengthy population-based study suggest.
METHODOLOGY:
- Researchers identified all cases with a first-time diagnosis of AD and matched each AD case to up to 40 AD-free control cases on the basis of age, sex, cohort entry date, and duration of follow-up.
- The exposure of interest was CAHPI, defined based on an algorithm using clinical guidelines and recommendations on the management of H pylori (HP) infection, with researchers focusing on infected individuals presenting with symptoms or developing serious complications from the infection.
- Researchers performed several sensitivity analyses, which included repeating the primary analysis using alternate lag periods, restricting the cohort to participants with AD (not vascular, alcoholic, and unspecified dementia), and using salmonellosis, an infection not previously associated with AD, as a negative control exposure.
TAKEAWAY:
- Compared with no exposure to CAHPI, exposure to CAHPI was associated with a moderately increased risk for AD (odds ratio [OR], 1.11; 95% CI, 1.01-1.21), with no major effect modification by demographics or socioeconomic status.
- The increased risk peaked 7.3-10.8 years after CAHPI onset (OR, 1.24; 95% CI, 1.05-1.47) before decreasing.
- Sensitivity analyses yielded findings that were overall consistent with those of the primary analysis.
- The analysis with salmonellosis as a negative control exposure showed no association with the risk for AD (OR, 1.03; 95% CI, 0.82-1.29).
IN PRACTICE:
“These results support the notion of HP infection as a potential modifiable risk factor of AD” and “pave the way for future randomized controlled trials that would assess the impact and cost-effectiveness of population-based targeted interventions such as individualized HP eradication programs, on the development of AD,” the authors write.
SOURCE:
The study was conducted by Antonios Douros, Department of Medicine, and Department of Epidemiology, Biostatistics, and Occupational Health, McGill University, Montreal, Quebec, Canada, and colleagues. It was published online in Alzheimer’s & Dementia.
LIMITATIONS:
Given the observational nature of the study, residual confounding is possible. Because the exposure definition was on the basis of CAHPI recorded by general practitioners, exposure misclassification due to symptomatic patients not seeking primary care is possible, as is outcome misclassification. The authors can’t rule out the possibility of an association between asymptomatic H pylori infection and AD risk.
DISCLOSURES:
The study received funding from the Canadian Institutes of Health Research. Douros has no relevant conflicts of interest; see paper for disclosures of other authors.
Pauline Anderson has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
results of a large and lengthy population-based study suggest.
METHODOLOGY:
- Researchers identified all cases with a first-time diagnosis of AD and matched each AD case to up to 40 AD-free control cases on the basis of age, sex, cohort entry date, and duration of follow-up.
- The exposure of interest was CAHPI, defined based on an algorithm using clinical guidelines and recommendations on the management of H pylori (HP) infection, with researchers focusing on infected individuals presenting with symptoms or developing serious complications from the infection.
- Researchers performed several sensitivity analyses, which included repeating the primary analysis using alternate lag periods, restricting the cohort to participants with AD (not vascular, alcoholic, and unspecified dementia), and using salmonellosis, an infection not previously associated with AD, as a negative control exposure.
TAKEAWAY:
- Compared with no exposure to CAHPI, exposure to CAHPI was associated with a moderately increased risk for AD (odds ratio [OR], 1.11; 95% CI, 1.01-1.21), with no major effect modification by demographics or socioeconomic status.
- The increased risk peaked 7.3-10.8 years after CAHPI onset (OR, 1.24; 95% CI, 1.05-1.47) before decreasing.
- Sensitivity analyses yielded findings that were overall consistent with those of the primary analysis.
- The analysis with salmonellosis as a negative control exposure showed no association with the risk for AD (OR, 1.03; 95% CI, 0.82-1.29).
IN PRACTICE:
“These results support the notion of HP infection as a potential modifiable risk factor of AD” and “pave the way for future randomized controlled trials that would assess the impact and cost-effectiveness of population-based targeted interventions such as individualized HP eradication programs, on the development of AD,” the authors write.
SOURCE:
The study was conducted by Antonios Douros, Department of Medicine, and Department of Epidemiology, Biostatistics, and Occupational Health, McGill University, Montreal, Quebec, Canada, and colleagues. It was published online in Alzheimer’s & Dementia.
LIMITATIONS:
Given the observational nature of the study, residual confounding is possible. Because the exposure definition was on the basis of CAHPI recorded by general practitioners, exposure misclassification due to symptomatic patients not seeking primary care is possible, as is outcome misclassification. The authors can’t rule out the possibility of an association between asymptomatic H pylori infection and AD risk.
DISCLOSURES:
The study received funding from the Canadian Institutes of Health Research. Douros has no relevant conflicts of interest; see paper for disclosures of other authors.
Pauline Anderson has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Hearing Aids and Dementia Risk Study Retracted
The study was published April 13 in The Lancet Public Health and reported at that time. It was retracted by the journal on December 12.
According to the retraction notice, the journal editors in late November were informed by the authors of the paper that an error was introduced in the output format setting of their SAS codes, which led to data for people with hearing loss using hearing aids and those with hearing loss without using hearing aids being switched.
This led to errors in their analysis, “which render their findings and conclusions false and misleading,” the retraction notice states.
These errors were identified by the researchers following an exchange with scientists seeking to reproduce the authors’ findings.In a statement, The Lancet Group said it “takes issues relating to research integrity extremely seriously” and follows best-practice guidance from the Committee on Publication Ethics (COPE) and the International Committee of Medical Journal Editors (ICMJE).
“Retractions are a rare but important part of the publishing process, and we are grateful to the scientists who prompted the re-examination of the data,” the statement reads.
Despite the retraction, other studies have suggested a link between hearing and dementia.
One study of US Medicare beneficiaries found a 61% higher dementia prevalence in those with moderate to severe hearing loss compared to those with normal hearing.
In this research, even mild hearing loss was associated with increased dementia risk, although it was not statistically significant, and use of hearing aids was tied to a 32% decrease in dementia prevalence.
In addition, a large meta-analysis showed that hearing aids significantly reduce the risk for cognitive decline and dementia and even improve short-term cognitive function in individuals with hearing loss.
A version of this article appeared on Medscape.com.
The study was published April 13 in The Lancet Public Health and reported at that time. It was retracted by the journal on December 12.
According to the retraction notice, the journal editors in late November were informed by the authors of the paper that an error was introduced in the output format setting of their SAS codes, which led to data for people with hearing loss using hearing aids and those with hearing loss without using hearing aids being switched.
This led to errors in their analysis, “which render their findings and conclusions false and misleading,” the retraction notice states.
These errors were identified by the researchers following an exchange with scientists seeking to reproduce the authors’ findings.In a statement, The Lancet Group said it “takes issues relating to research integrity extremely seriously” and follows best-practice guidance from the Committee on Publication Ethics (COPE) and the International Committee of Medical Journal Editors (ICMJE).
“Retractions are a rare but important part of the publishing process, and we are grateful to the scientists who prompted the re-examination of the data,” the statement reads.
Despite the retraction, other studies have suggested a link between hearing and dementia.
One study of US Medicare beneficiaries found a 61% higher dementia prevalence in those with moderate to severe hearing loss compared to those with normal hearing.
In this research, even mild hearing loss was associated with increased dementia risk, although it was not statistically significant, and use of hearing aids was tied to a 32% decrease in dementia prevalence.
In addition, a large meta-analysis showed that hearing aids significantly reduce the risk for cognitive decline and dementia and even improve short-term cognitive function in individuals with hearing loss.
A version of this article appeared on Medscape.com.
The study was published April 13 in The Lancet Public Health and reported at that time. It was retracted by the journal on December 12.
According to the retraction notice, the journal editors in late November were informed by the authors of the paper that an error was introduced in the output format setting of their SAS codes, which led to data for people with hearing loss using hearing aids and those with hearing loss without using hearing aids being switched.
This led to errors in their analysis, “which render their findings and conclusions false and misleading,” the retraction notice states.
These errors were identified by the researchers following an exchange with scientists seeking to reproduce the authors’ findings.In a statement, The Lancet Group said it “takes issues relating to research integrity extremely seriously” and follows best-practice guidance from the Committee on Publication Ethics (COPE) and the International Committee of Medical Journal Editors (ICMJE).
“Retractions are a rare but important part of the publishing process, and we are grateful to the scientists who prompted the re-examination of the data,” the statement reads.
Despite the retraction, other studies have suggested a link between hearing and dementia.
One study of US Medicare beneficiaries found a 61% higher dementia prevalence in those with moderate to severe hearing loss compared to those with normal hearing.
In this research, even mild hearing loss was associated with increased dementia risk, although it was not statistically significant, and use of hearing aids was tied to a 32% decrease in dementia prevalence.
In addition, a large meta-analysis showed that hearing aids significantly reduce the risk for cognitive decline and dementia and even improve short-term cognitive function in individuals with hearing loss.
A version of this article appeared on Medscape.com.
FROM THE LANCET PUBLIC HEALTH
Light therapy a beacon of hope for Alzheimer’s?
TOPLINE:
Light therapy leads to significant improvement in several sleep measures and helps alleviate depression and agitation in patients with Alzheimer’s disease (AD), a meta-analysis of 15 high-quality trials shows.
METHODOLOGY:
- This meta-analysis included 15 randomized controlled trials involving 598 patients with mild to moderate AD.
- The included trials were written in English, published between 2005 and 2022, and performed in seven countries. A fixed-effects model was used for data analysis.
TAKEAWAY:
- Light therapy significantly improved sleep efficiency (mean difference [MD], −2.42; P < .00001), increased interdaily stability (MD, −0.04; P < .00001), and reduced intradaily variability (MD, −0.04; P < .00001), indicating better sleep quality.
- Light therapy reduced agitation (MD, −3.97; P < .00001), depression (MD, −2.55; P < .00001), and caregiver burden (MD, −3.57; P < .00001).
- Light therapy also had a significant advantage over usual care in reducing the severity of psychobehavioral symptoms as assessed by the Neuropsychiatric Inventory (MD, −3.07; P < .00001).
- Light therapy had no statistically significant effect on improving cognitive function as measured by the Mini-Mental State Examination.
IN PRACTICE:
“These findings, combined with its low side-effects, suggest the role of light therapy as a promising treatment for AD. Although light therapy has fewer side effects than pharmacological treatment, adverse behavioral outcomes in patients due to bright light exposure should be considered,” the authors wrote.
SOURCE:
The study by Lili Zang and colleagues from Weifang Medical University School of Nursing, Shandong Province, China, was published online on December 6, 2023, in PLOS One.
LIMITATIONS:
The types and degrees of dementia in the included studies were inconsistent, potentially affecting the outcome indicators. Some articles did not clearly describe their randomization and allocation concealment methods, indicating possible bias in these studies.
DISCLOSURES:
The study was supported by the Natural Science Foundation of Shandong Province, China. The authors declared no competing interests.
Megan Brooks has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
Light therapy leads to significant improvement in several sleep measures and helps alleviate depression and agitation in patients with Alzheimer’s disease (AD), a meta-analysis of 15 high-quality trials shows.
METHODOLOGY:
- This meta-analysis included 15 randomized controlled trials involving 598 patients with mild to moderate AD.
- The included trials were written in English, published between 2005 and 2022, and performed in seven countries. A fixed-effects model was used for data analysis.
TAKEAWAY:
- Light therapy significantly improved sleep efficiency (mean difference [MD], −2.42; P < .00001), increased interdaily stability (MD, −0.04; P < .00001), and reduced intradaily variability (MD, −0.04; P < .00001), indicating better sleep quality.
- Light therapy reduced agitation (MD, −3.97; P < .00001), depression (MD, −2.55; P < .00001), and caregiver burden (MD, −3.57; P < .00001).
- Light therapy also had a significant advantage over usual care in reducing the severity of psychobehavioral symptoms as assessed by the Neuropsychiatric Inventory (MD, −3.07; P < .00001).
- Light therapy had no statistically significant effect on improving cognitive function as measured by the Mini-Mental State Examination.
IN PRACTICE:
“These findings, combined with its low side-effects, suggest the role of light therapy as a promising treatment for AD. Although light therapy has fewer side effects than pharmacological treatment, adverse behavioral outcomes in patients due to bright light exposure should be considered,” the authors wrote.
SOURCE:
The study by Lili Zang and colleagues from Weifang Medical University School of Nursing, Shandong Province, China, was published online on December 6, 2023, in PLOS One.
LIMITATIONS:
The types and degrees of dementia in the included studies were inconsistent, potentially affecting the outcome indicators. Some articles did not clearly describe their randomization and allocation concealment methods, indicating possible bias in these studies.
DISCLOSURES:
The study was supported by the Natural Science Foundation of Shandong Province, China. The authors declared no competing interests.
Megan Brooks has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
Light therapy leads to significant improvement in several sleep measures and helps alleviate depression and agitation in patients with Alzheimer’s disease (AD), a meta-analysis of 15 high-quality trials shows.
METHODOLOGY:
- This meta-analysis included 15 randomized controlled trials involving 598 patients with mild to moderate AD.
- The included trials were written in English, published between 2005 and 2022, and performed in seven countries. A fixed-effects model was used for data analysis.
TAKEAWAY:
- Light therapy significantly improved sleep efficiency (mean difference [MD], −2.42; P < .00001), increased interdaily stability (MD, −0.04; P < .00001), and reduced intradaily variability (MD, −0.04; P < .00001), indicating better sleep quality.
- Light therapy reduced agitation (MD, −3.97; P < .00001), depression (MD, −2.55; P < .00001), and caregiver burden (MD, −3.57; P < .00001).
- Light therapy also had a significant advantage over usual care in reducing the severity of psychobehavioral symptoms as assessed by the Neuropsychiatric Inventory (MD, −3.07; P < .00001).
- Light therapy had no statistically significant effect on improving cognitive function as measured by the Mini-Mental State Examination.
IN PRACTICE:
“These findings, combined with its low side-effects, suggest the role of light therapy as a promising treatment for AD. Although light therapy has fewer side effects than pharmacological treatment, adverse behavioral outcomes in patients due to bright light exposure should be considered,” the authors wrote.
SOURCE:
The study by Lili Zang and colleagues from Weifang Medical University School of Nursing, Shandong Province, China, was published online on December 6, 2023, in PLOS One.
LIMITATIONS:
The types and degrees of dementia in the included studies were inconsistent, potentially affecting the outcome indicators. Some articles did not clearly describe their randomization and allocation concealment methods, indicating possible bias in these studies.
DISCLOSURES:
The study was supported by the Natural Science Foundation of Shandong Province, China. The authors declared no competing interests.
Megan Brooks has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Specific personality traits may influence dementia risk
TOPLINE:
, whereas those who score highly for neuroticism and have a negative outlook may be at increased risk, new research suggests.
METHODOLOGY:
- Researchers examined the link between the “big five” personality traits (conscientiousness, extraversion, openness to experience, neuroticism, and agreeableness) and subjective well-being (positive and negative affect and life satisfaction) and clinical symptoms of dementia (cognitive test performance) and neuropathology at autopsy.
- Data for the meta-analysis came from eight longitudinal studies with 44,531 adults (aged 49-81 years at baseline; 26%-61% women) followed for up to 21 years, during which 1703 incident cases of dementia occurred.
- Bayesian multilevel models tested whether personality traits and subjective well-being differentially predicted neuropsychological and neuropathologic characteristics of dementia.
TAKEAWAY:
- High neuroticism, negative affect, and low conscientiousness were risk factors for dementia, whereas conscientiousness, extraversion, and positive affect were protective.
- Across all analyses, there was directional consistency in estimates across samples, which is noteworthy given between-study differences in sociodemographic and design characteristics.
- No consistent associations were found between psychological factors and neuropathology.
- However, individuals higher in conscientiousness who did not receive a clinical diagnosis tended to have a lower Braak stage at autopsy, suggesting the possibility that conscientiousness is related to cognitive resilience.
IN PRACTICE:
“These results replicate and extend evidence that personality traits may assist in early identification and dementia-care planning strategies, as well as risk stratification for dementia diagnosis. Moreover, our findings provide further support for recommendations to incorporate psychological trait measures into clinical screening or diagnosis criteria,” the authors write. SOURCE:
The study, with first author Emorie Beck, PhD, Department of Psychology, University of California, Davis, was published online on November 29, 2023, in Alzheimer’s & Dementia.
LIMITATIONS:
Access to autopsy data was limited. The findings may not generalize across racial groups. The analysis did not examine dynamic associations between changing personality and cognition and neuropathology over time.
DISCLOSURES:
The study was supported by grants from the National Institute on Aging. The authors have declared no conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
, whereas those who score highly for neuroticism and have a negative outlook may be at increased risk, new research suggests.
METHODOLOGY:
- Researchers examined the link between the “big five” personality traits (conscientiousness, extraversion, openness to experience, neuroticism, and agreeableness) and subjective well-being (positive and negative affect and life satisfaction) and clinical symptoms of dementia (cognitive test performance) and neuropathology at autopsy.
- Data for the meta-analysis came from eight longitudinal studies with 44,531 adults (aged 49-81 years at baseline; 26%-61% women) followed for up to 21 years, during which 1703 incident cases of dementia occurred.
- Bayesian multilevel models tested whether personality traits and subjective well-being differentially predicted neuropsychological and neuropathologic characteristics of dementia.
TAKEAWAY:
- High neuroticism, negative affect, and low conscientiousness were risk factors for dementia, whereas conscientiousness, extraversion, and positive affect were protective.
- Across all analyses, there was directional consistency in estimates across samples, which is noteworthy given between-study differences in sociodemographic and design characteristics.
- No consistent associations were found between psychological factors and neuropathology.
- However, individuals higher in conscientiousness who did not receive a clinical diagnosis tended to have a lower Braak stage at autopsy, suggesting the possibility that conscientiousness is related to cognitive resilience.
IN PRACTICE:
“These results replicate and extend evidence that personality traits may assist in early identification and dementia-care planning strategies, as well as risk stratification for dementia diagnosis. Moreover, our findings provide further support for recommendations to incorporate psychological trait measures into clinical screening or diagnosis criteria,” the authors write. SOURCE:
The study, with first author Emorie Beck, PhD, Department of Psychology, University of California, Davis, was published online on November 29, 2023, in Alzheimer’s & Dementia.
LIMITATIONS:
Access to autopsy data was limited. The findings may not generalize across racial groups. The analysis did not examine dynamic associations between changing personality and cognition and neuropathology over time.
DISCLOSURES:
The study was supported by grants from the National Institute on Aging. The authors have declared no conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
, whereas those who score highly for neuroticism and have a negative outlook may be at increased risk, new research suggests.
METHODOLOGY:
- Researchers examined the link between the “big five” personality traits (conscientiousness, extraversion, openness to experience, neuroticism, and agreeableness) and subjective well-being (positive and negative affect and life satisfaction) and clinical symptoms of dementia (cognitive test performance) and neuropathology at autopsy.
- Data for the meta-analysis came from eight longitudinal studies with 44,531 adults (aged 49-81 years at baseline; 26%-61% women) followed for up to 21 years, during which 1703 incident cases of dementia occurred.
- Bayesian multilevel models tested whether personality traits and subjective well-being differentially predicted neuropsychological and neuropathologic characteristics of dementia.
TAKEAWAY:
- High neuroticism, negative affect, and low conscientiousness were risk factors for dementia, whereas conscientiousness, extraversion, and positive affect were protective.
- Across all analyses, there was directional consistency in estimates across samples, which is noteworthy given between-study differences in sociodemographic and design characteristics.
- No consistent associations were found between psychological factors and neuropathology.
- However, individuals higher in conscientiousness who did not receive a clinical diagnosis tended to have a lower Braak stage at autopsy, suggesting the possibility that conscientiousness is related to cognitive resilience.
IN PRACTICE:
“These results replicate and extend evidence that personality traits may assist in early identification and dementia-care planning strategies, as well as risk stratification for dementia diagnosis. Moreover, our findings provide further support for recommendations to incorporate psychological trait measures into clinical screening or diagnosis criteria,” the authors write. SOURCE:
The study, with first author Emorie Beck, PhD, Department of Psychology, University of California, Davis, was published online on November 29, 2023, in Alzheimer’s & Dementia.
LIMITATIONS:
Access to autopsy data was limited. The findings may not generalize across racial groups. The analysis did not examine dynamic associations between changing personality and cognition and neuropathology over time.
DISCLOSURES:
The study was supported by grants from the National Institute on Aging. The authors have declared no conflicts of interest.
A version of this article first appeared on Medscape.com.