User login
Sotagliflozin’s HFpEF benefit confirmed by new analyses
It’s now official: The investigational sodium-glucose cotransporter (SGLT) 1/2 inhibitor sotagliflozin is the first agent clearly shown in a prespecified analysis of randomized trials to improve clinical outcomes in patients with heart failure with reduced ejection fraction (HFpEF).
Researchers who ran the SCORED and SOLOIST-WHF pivotal trials for sotagliflozin first made that claim in November 2020 when reporting top-line results from a prespecified meta-analysis of the two trials during the American Heart Association annual scientific sessions. A follow-up report during the annual scientific sessions of the American College of Cardiology fleshed out the evidence and firmed up their landmark conclusion.
The meta-analysis (Abstract 410-08) included 4,500 patients with type 2 diabetes and diagnosed heart failure at entry; its primary endpoint, which was the same in both trials, was the combined incidence of cardiovascular death and the total number of either hospitalization for heart failure or urgent outpatient visits for heart failure.
Compared with placebo, treatment with sotagliflozin for a median of about 15 months dropped this composite endpoint by a relative 33% among the 1,931 who began the study with a left ventricular ejection fraction (LVEF) of at least 50% (HFpEF), by a relative 22% in the 1,758 patients who entered with an LVEF of less than 40% (patients with heart failure with reduced ejection fraction), and by a relative 43% among the 811 patients who began with an LVEF of 40%-49% (patients with heart failure with mid-range ejection fraction). The relative risk reductions were significant for all three subgroups, Deepak L. Bhatt, MD, reported at the meeting.
Equally effective ‘across the full range of LVEFs.’
Perhaps as notable and unprecedented was the further finding that the clinical benefits seen with treatment of patients with type 2 diabetes with sotagliflozin was consistent regardless of the ejection fraction they had at entry. Enrolled patients with baseline LVEFs in the range of 25% received a relative benefit from sotagliflozin treatment that was statistically no different from the benefit seen in patients who entered with an LVEF in the neighborhood of 45%, 65%, or at any other level across the LVEF spectrum, a finding that Dr. Bhatt called “remarkable” during a press briefing. “The results show the benefit of sotagliflozin across the full range of LVEFs.”
“We are very excited in the heart failure world by the SGLT2 inhibitors; we’ve been impressed by their reduction in heart failure hospitalizations, but we wonder about the patients with HFpEF, where we haven’t had a blockbuster drug to give,” said Ileana L. Piña, MD, a heart failure specialist and medical officer with the Food and Drug Administration.
The new findings “look like they could pose a regulatory indication [for sotagliflozin] for patients with type 2 diabetes and heart failure across the entire spectrum of heart failure,” said Christopher M. O’Connor, MD, a heart failure specialist and president of the Inova Heart & Vascular institute in Falls Church, Va., and designated discussant for Dr. Bhatt’s report.
SCORED randomized 10,584 patients with type 2 diabetes and chronic kidney disease to treatment with sotagliflozin or placebo on top of guideline-directed medical therapy. During a median 16 months of treatment, the combined primary endpoint occurred at a rate of 5.6 events/100 patient years on sotagliflozin and 7.5 events/100 patient years in the controls, a significant 26% relative reduction with sotagliflozin (N Engl J Med. 2021 Jan 14;384[2]:129-39). Nearly a third of the enrolled patients had heart failure, with representation across the range of LVEF.
SOLOIST-WHF randomized 1,222 patients with type 2 diabetes who were recently hospitalized for worsening heart failure. During a median 9 months of follow-up, the primary endpoint occurred at a rate of 51 events/100 patient years in the sotagliflozin-treated patients and a rate of 76 events/100 patient years in the controls, a significant 33% relative reduction with sotagliflozin (N Engl J Med. 2021 Jan 14;384[2]:117-28). Both trials stopped prematurely because of sponsorship issues.
In addition to the 4,500 patients with heart failure at entry in both trials, SCORED included a total of more than 6,700 without diagnosed heart failure at baseline, and in this subgroup treatment with sotagliflozin cut the incidence of the primary endpoint by a significant 27% compared with control patients.
A significant on-treatment reduction in CV death
Other new, notable findings from the meta-analysis included the observation that while treatment with sotagliflozin failed to produce a significant reduction in cardiovascular death, compared with placebo, in the intent-to-treat analysis of all patients and of those with heart failure at baseline (it produced nonsignificant point-estimate reductions of 11% compared with placebo for all patients, and of 23% for patients who began the study with heart failure), it did result in a significant 23% relative risk reduction when the researchers focused on patients while they remained adherent to their sotagliflozin regimen (the on-treatment analysis). This 23% relative reduction appeared among all enrolled patients, as well as in the subgroup that started with diagnosed heart failure.
“Given the totality of data from the SGLT2 inhibitors, I think this is a real finding,” Dr. Bhatt said.
Additional analyses also showed that, among women, treatment with sotagliflozin was linked with significant relative reductions in the primary endpoint of roughly 30% compared with placebo among all patients, and also among those with heart failure at baseline. “HFpEF is a problem particularly in older women, and we showed that the benefit was consistent in men and women,” Dr. Bhatt said.
He acknowledged that results are expected soon from two pivotal trials that are examining two different SGLT2 inhibitors, dapagliflozin and empagliflozin, in patients with HFpEF. “I think there will be a class effect for both SGLT2 inhibitors and sotagliflozin for reducing heart failure events in patients with HFpEF, and I predict that the dapagliflozin and empagliflozin trials will have positive results,” Dr. Bhatt said.
Sotagliflozin differs from the SGLT2 inhibitors by also inhibiting SGLT1, an enzyme found in the gastrointestinal system that, when inhibited, results in increased glucose excretion from the gut and a cut in bloodstream levels of postprandial glucose levels. The Food and Drug Administration accepted data from SCORED and SOLOIST-WHF as part of the evidence the agency is now considering for granting a new drug approval to sotagliflozin.
SCORED and SOLOIST-WHF were initially sponsored by Sanofi, and later by Lexicon Pharmaceuticals. Dr. Bhatt’s institution, Brigham and Women’s Hospital, has received funding from Sanofi and Lexicon Pharmaceuticals. He has been a consultant to and received honoraria from K2P, Level Ex, and MJH Life Sciences; he has been an adviser to Cardax, Cereno Scientific, Myokardia, Novo Nordisk, Phase Bio, and PLx Pharma; and he has received research funding from numerous companies. Dr. Piña has no relevant disclosures. Dr. O’Connor has been a consultant to Arena, Bayer, Bristol-Myers Squibb, Merck, and Windtree, and he has an ownership interest in Biscardia.
It’s now official: The investigational sodium-glucose cotransporter (SGLT) 1/2 inhibitor sotagliflozin is the first agent clearly shown in a prespecified analysis of randomized trials to improve clinical outcomes in patients with heart failure with reduced ejection fraction (HFpEF).
Researchers who ran the SCORED and SOLOIST-WHF pivotal trials for sotagliflozin first made that claim in November 2020 when reporting top-line results from a prespecified meta-analysis of the two trials during the American Heart Association annual scientific sessions. A follow-up report during the annual scientific sessions of the American College of Cardiology fleshed out the evidence and firmed up their landmark conclusion.
The meta-analysis (Abstract 410-08) included 4,500 patients with type 2 diabetes and diagnosed heart failure at entry; its primary endpoint, which was the same in both trials, was the combined incidence of cardiovascular death and the total number of either hospitalization for heart failure or urgent outpatient visits for heart failure.
Compared with placebo, treatment with sotagliflozin for a median of about 15 months dropped this composite endpoint by a relative 33% among the 1,931 who began the study with a left ventricular ejection fraction (LVEF) of at least 50% (HFpEF), by a relative 22% in the 1,758 patients who entered with an LVEF of less than 40% (patients with heart failure with reduced ejection fraction), and by a relative 43% among the 811 patients who began with an LVEF of 40%-49% (patients with heart failure with mid-range ejection fraction). The relative risk reductions were significant for all three subgroups, Deepak L. Bhatt, MD, reported at the meeting.
Equally effective ‘across the full range of LVEFs.’
Perhaps as notable and unprecedented was the further finding that the clinical benefits seen with treatment of patients with type 2 diabetes with sotagliflozin was consistent regardless of the ejection fraction they had at entry. Enrolled patients with baseline LVEFs in the range of 25% received a relative benefit from sotagliflozin treatment that was statistically no different from the benefit seen in patients who entered with an LVEF in the neighborhood of 45%, 65%, or at any other level across the LVEF spectrum, a finding that Dr. Bhatt called “remarkable” during a press briefing. “The results show the benefit of sotagliflozin across the full range of LVEFs.”
“We are very excited in the heart failure world by the SGLT2 inhibitors; we’ve been impressed by their reduction in heart failure hospitalizations, but we wonder about the patients with HFpEF, where we haven’t had a blockbuster drug to give,” said Ileana L. Piña, MD, a heart failure specialist and medical officer with the Food and Drug Administration.
The new findings “look like they could pose a regulatory indication [for sotagliflozin] for patients with type 2 diabetes and heart failure across the entire spectrum of heart failure,” said Christopher M. O’Connor, MD, a heart failure specialist and president of the Inova Heart & Vascular institute in Falls Church, Va., and designated discussant for Dr. Bhatt’s report.
SCORED randomized 10,584 patients with type 2 diabetes and chronic kidney disease to treatment with sotagliflozin or placebo on top of guideline-directed medical therapy. During a median 16 months of treatment, the combined primary endpoint occurred at a rate of 5.6 events/100 patient years on sotagliflozin and 7.5 events/100 patient years in the controls, a significant 26% relative reduction with sotagliflozin (N Engl J Med. 2021 Jan 14;384[2]:129-39). Nearly a third of the enrolled patients had heart failure, with representation across the range of LVEF.
SOLOIST-WHF randomized 1,222 patients with type 2 diabetes who were recently hospitalized for worsening heart failure. During a median 9 months of follow-up, the primary endpoint occurred at a rate of 51 events/100 patient years in the sotagliflozin-treated patients and a rate of 76 events/100 patient years in the controls, a significant 33% relative reduction with sotagliflozin (N Engl J Med. 2021 Jan 14;384[2]:117-28). Both trials stopped prematurely because of sponsorship issues.
In addition to the 4,500 patients with heart failure at entry in both trials, SCORED included a total of more than 6,700 without diagnosed heart failure at baseline, and in this subgroup treatment with sotagliflozin cut the incidence of the primary endpoint by a significant 27% compared with control patients.
A significant on-treatment reduction in CV death
Other new, notable findings from the meta-analysis included the observation that while treatment with sotagliflozin failed to produce a significant reduction in cardiovascular death, compared with placebo, in the intent-to-treat analysis of all patients and of those with heart failure at baseline (it produced nonsignificant point-estimate reductions of 11% compared with placebo for all patients, and of 23% for patients who began the study with heart failure), it did result in a significant 23% relative risk reduction when the researchers focused on patients while they remained adherent to their sotagliflozin regimen (the on-treatment analysis). This 23% relative reduction appeared among all enrolled patients, as well as in the subgroup that started with diagnosed heart failure.
“Given the totality of data from the SGLT2 inhibitors, I think this is a real finding,” Dr. Bhatt said.
Additional analyses also showed that, among women, treatment with sotagliflozin was linked with significant relative reductions in the primary endpoint of roughly 30% compared with placebo among all patients, and also among those with heart failure at baseline. “HFpEF is a problem particularly in older women, and we showed that the benefit was consistent in men and women,” Dr. Bhatt said.
He acknowledged that results are expected soon from two pivotal trials that are examining two different SGLT2 inhibitors, dapagliflozin and empagliflozin, in patients with HFpEF. “I think there will be a class effect for both SGLT2 inhibitors and sotagliflozin for reducing heart failure events in patients with HFpEF, and I predict that the dapagliflozin and empagliflozin trials will have positive results,” Dr. Bhatt said.
Sotagliflozin differs from the SGLT2 inhibitors by also inhibiting SGLT1, an enzyme found in the gastrointestinal system that, when inhibited, results in increased glucose excretion from the gut and a cut in bloodstream levels of postprandial glucose levels. The Food and Drug Administration accepted data from SCORED and SOLOIST-WHF as part of the evidence the agency is now considering for granting a new drug approval to sotagliflozin.
SCORED and SOLOIST-WHF were initially sponsored by Sanofi, and later by Lexicon Pharmaceuticals. Dr. Bhatt’s institution, Brigham and Women’s Hospital, has received funding from Sanofi and Lexicon Pharmaceuticals. He has been a consultant to and received honoraria from K2P, Level Ex, and MJH Life Sciences; he has been an adviser to Cardax, Cereno Scientific, Myokardia, Novo Nordisk, Phase Bio, and PLx Pharma; and he has received research funding from numerous companies. Dr. Piña has no relevant disclosures. Dr. O’Connor has been a consultant to Arena, Bayer, Bristol-Myers Squibb, Merck, and Windtree, and he has an ownership interest in Biscardia.
It’s now official: The investigational sodium-glucose cotransporter (SGLT) 1/2 inhibitor sotagliflozin is the first agent clearly shown in a prespecified analysis of randomized trials to improve clinical outcomes in patients with heart failure with reduced ejection fraction (HFpEF).
Researchers who ran the SCORED and SOLOIST-WHF pivotal trials for sotagliflozin first made that claim in November 2020 when reporting top-line results from a prespecified meta-analysis of the two trials during the American Heart Association annual scientific sessions. A follow-up report during the annual scientific sessions of the American College of Cardiology fleshed out the evidence and firmed up their landmark conclusion.
The meta-analysis (Abstract 410-08) included 4,500 patients with type 2 diabetes and diagnosed heart failure at entry; its primary endpoint, which was the same in both trials, was the combined incidence of cardiovascular death and the total number of either hospitalization for heart failure or urgent outpatient visits for heart failure.
Compared with placebo, treatment with sotagliflozin for a median of about 15 months dropped this composite endpoint by a relative 33% among the 1,931 who began the study with a left ventricular ejection fraction (LVEF) of at least 50% (HFpEF), by a relative 22% in the 1,758 patients who entered with an LVEF of less than 40% (patients with heart failure with reduced ejection fraction), and by a relative 43% among the 811 patients who began with an LVEF of 40%-49% (patients with heart failure with mid-range ejection fraction). The relative risk reductions were significant for all three subgroups, Deepak L. Bhatt, MD, reported at the meeting.
Equally effective ‘across the full range of LVEFs.’
Perhaps as notable and unprecedented was the further finding that the clinical benefits seen with treatment of patients with type 2 diabetes with sotagliflozin was consistent regardless of the ejection fraction they had at entry. Enrolled patients with baseline LVEFs in the range of 25% received a relative benefit from sotagliflozin treatment that was statistically no different from the benefit seen in patients who entered with an LVEF in the neighborhood of 45%, 65%, or at any other level across the LVEF spectrum, a finding that Dr. Bhatt called “remarkable” during a press briefing. “The results show the benefit of sotagliflozin across the full range of LVEFs.”
“We are very excited in the heart failure world by the SGLT2 inhibitors; we’ve been impressed by their reduction in heart failure hospitalizations, but we wonder about the patients with HFpEF, where we haven’t had a blockbuster drug to give,” said Ileana L. Piña, MD, a heart failure specialist and medical officer with the Food and Drug Administration.
The new findings “look like they could pose a regulatory indication [for sotagliflozin] for patients with type 2 diabetes and heart failure across the entire spectrum of heart failure,” said Christopher M. O’Connor, MD, a heart failure specialist and president of the Inova Heart & Vascular institute in Falls Church, Va., and designated discussant for Dr. Bhatt’s report.
SCORED randomized 10,584 patients with type 2 diabetes and chronic kidney disease to treatment with sotagliflozin or placebo on top of guideline-directed medical therapy. During a median 16 months of treatment, the combined primary endpoint occurred at a rate of 5.6 events/100 patient years on sotagliflozin and 7.5 events/100 patient years in the controls, a significant 26% relative reduction with sotagliflozin (N Engl J Med. 2021 Jan 14;384[2]:129-39). Nearly a third of the enrolled patients had heart failure, with representation across the range of LVEF.
SOLOIST-WHF randomized 1,222 patients with type 2 diabetes who were recently hospitalized for worsening heart failure. During a median 9 months of follow-up, the primary endpoint occurred at a rate of 51 events/100 patient years in the sotagliflozin-treated patients and a rate of 76 events/100 patient years in the controls, a significant 33% relative reduction with sotagliflozin (N Engl J Med. 2021 Jan 14;384[2]:117-28). Both trials stopped prematurely because of sponsorship issues.
In addition to the 4,500 patients with heart failure at entry in both trials, SCORED included a total of more than 6,700 without diagnosed heart failure at baseline, and in this subgroup treatment with sotagliflozin cut the incidence of the primary endpoint by a significant 27% compared with control patients.
A significant on-treatment reduction in CV death
Other new, notable findings from the meta-analysis included the observation that while treatment with sotagliflozin failed to produce a significant reduction in cardiovascular death, compared with placebo, in the intent-to-treat analysis of all patients and of those with heart failure at baseline (it produced nonsignificant point-estimate reductions of 11% compared with placebo for all patients, and of 23% for patients who began the study with heart failure), it did result in a significant 23% relative risk reduction when the researchers focused on patients while they remained adherent to their sotagliflozin regimen (the on-treatment analysis). This 23% relative reduction appeared among all enrolled patients, as well as in the subgroup that started with diagnosed heart failure.
“Given the totality of data from the SGLT2 inhibitors, I think this is a real finding,” Dr. Bhatt said.
Additional analyses also showed that, among women, treatment with sotagliflozin was linked with significant relative reductions in the primary endpoint of roughly 30% compared with placebo among all patients, and also among those with heart failure at baseline. “HFpEF is a problem particularly in older women, and we showed that the benefit was consistent in men and women,” Dr. Bhatt said.
He acknowledged that results are expected soon from two pivotal trials that are examining two different SGLT2 inhibitors, dapagliflozin and empagliflozin, in patients with HFpEF. “I think there will be a class effect for both SGLT2 inhibitors and sotagliflozin for reducing heart failure events in patients with HFpEF, and I predict that the dapagliflozin and empagliflozin trials will have positive results,” Dr. Bhatt said.
Sotagliflozin differs from the SGLT2 inhibitors by also inhibiting SGLT1, an enzyme found in the gastrointestinal system that, when inhibited, results in increased glucose excretion from the gut and a cut in bloodstream levels of postprandial glucose levels. The Food and Drug Administration accepted data from SCORED and SOLOIST-WHF as part of the evidence the agency is now considering for granting a new drug approval to sotagliflozin.
SCORED and SOLOIST-WHF were initially sponsored by Sanofi, and later by Lexicon Pharmaceuticals. Dr. Bhatt’s institution, Brigham and Women’s Hospital, has received funding from Sanofi and Lexicon Pharmaceuticals. He has been a consultant to and received honoraria from K2P, Level Ex, and MJH Life Sciences; he has been an adviser to Cardax, Cereno Scientific, Myokardia, Novo Nordisk, Phase Bio, and PLx Pharma; and he has received research funding from numerous companies. Dr. Piña has no relevant disclosures. Dr. O’Connor has been a consultant to Arena, Bayer, Bristol-Myers Squibb, Merck, and Windtree, and he has an ownership interest in Biscardia.
FROM ACC 2021
Underlying heart rhythm, not ICD shocks, drives mortality
Combined data from five implantable cardioverter-defibrillator (ICD) trials suggest that it is the underlying arrhythmic disorder, rather than the ICD therapy itself, that affects mortality in these patients.
Analysis of the MADIT II, MADIT-RISK, MADIT-CRT, MADIT-RIT, and RAID trials showed that the major determinant of mortality in patients receiving a primary prevention ICD was the arrhythmic substrate that leads to occurrence of fast ventricular tachycardia (VT), defined as ≥ 200 bpm, or ventricular fibrillation (VF), not adverse effects of the ICD shock therapy itself.
Patients experiencing an episode of VT had more than a twofold increased risk for death during a follow-up of 2½ years; however, ICD therapies for VT less than 200 bpm and inappropriate ICD shocks were not associated with a higher risk for death.
The findings were published online in the Journal of the American College of Cardiology.
“We know that patients receiving an ICD shock have increased mortality during subsequent follow-up,” first author Mehmet K. Aktas, MD, MBA, University of Rochester (N.Y.), said in an interview.
“There are conflicting data on the impact of ICD shocks on subsequent mortality, and in this study, we aimed to determine whether shocks per se increase subsequent mortality risk or whether the arrhythmic substrate that leads to ICD therapy results in subsequent risk of death,” Dr. Aktas said.
He and his team evaluated the association of ICD therapy with subsequent mortality according to the type of ICD therapy (model I), type of arrhythmia for which ICD therapy was delivered (model II), combined assessment of all arrhythmia and therapy types during follow-up (model III), and incremental risk associated with repeated ICD shocks (model IV).
The study cohort included 5,516 patients. Of these, 1,001 patients (18%) received appropriate ICD therapy and 561 (10%) received inappropriate ICD therapy during an average of 2.4 years.
Patients receiving an appropriate ICD therapy were more likely to be male and to have prior atrial arrhythmia and nonsustained VT compared with those without ICD therapy.
Patients receiving an inappropriate shock were more likely to be younger, to be African American, and to be less likely to have prior nonsustained VT, compared with those without ICD therapy.
Most patients (90%) were receiving beta-blockers and angiotensin-converting enzyme inhibitors or angiotensin receptor blockers regardless of device therapy during follow-up, and 10% of patients were treated with amiodarone.
In model I, at 3 years, the cumulative probability of death following an appropriate ICD shock was 38% compared with no appropriate ICD shock (P < .001). Inappropriate shock alone was not associated with mortality risk.
In model II, which looked at the type of arrhythmia for which ICD therapy was delivered, the cumulative death rate at 3 years following the first occurrence of ICD therapy for VT ≥ 200 beats/min or VF was 27%, compared with 10% in patients not experiencing VT ≥ 200 beats/min or VF (P < .001).
In model III, the highest risk for death was observed following shocks delivered after a failed antitachycardia pacing (ATP) for fast VT (hazard ratio [HR], 3.05), followed by ICD shock for VF (HR, 2.86), ICD shock for fast VT without a prior ATP (HR, 2.83), and ICD shock for slower VT (< 200 beats/min) without a prior ATP (HR, 2.39).
In contrast, other types of appropriate and inappropriate shock or ATP therapies were not associated with a significant risk increase.
In model IV, which assessed the association of shock therapy counts with the risk for death, two or more ICD appropriate shocks were not associated with increased risk after the first appropriate ICD shock.
“Our findings shed light on the mechanisms associated with increased mortality risk in primary prevention ICD recipients,” Dr. Aktas said.
“Studies that evaluate interventions focused on treating and stabilizing the myocardial substrate, which promotes ventricular tachyarrhythmias, such as catheter ablation, are needed to improve survival in heart failure patients,” he added.
Thoughtful study design
In an accompanying editorial, Rajat Deo, MD, and Naga Venkata K. Pothineni, MD, both from the University of Pennsylvania, Philadelphia, praised the researchers for their “thoughtful study design.”
“The take-home message that is most relevant to our clinical practice is clear: Sustained ventricular arrhythmias are a prognostic marker of death and heart failure hospitalization,” they wrote.
The editorialists also commented on the higher rate of inappropriate ICD therapies in African Americans.
“It is concerning to observe that Black patients had a markedly higher rate of inappropriate ICD therapies compared with White patients – and this was in the setting of some of the most respectable, established, and well-funded clinical trials,” they wrote.
Reasons for disparities in outcomes include access to appropriate and affordable medical therapies, access to specialty clinics and caregivers, remote ICD monitoring, and compliance issues.
“Future work will need to understand how the social determinants of health including race affect the treatment and outcomes of our primary prevention ICD population,” they wrote.
Identifying and characterizing the arrhythmic substrate will become a key component of sudden cardiac death risk stratification, the editorialists predicted.
“Concurrently, we must continue to partner with industry colleagues and work with our professional societies to ensure health equity across our patient population,” they concluded.
Dr. Aktas has received research grants from Boston Scientific and Medtronic. Dr. Deo and his coeditorialists report no relevant financial relationships. The MADIT trials were funded by an unrestricted research grant from Boston Scientific to the University of Rochester Medical Center. The RAID trial was funded by the National Institutes of Health.
A version of this article first appeared on Medscape.com.
Combined data from five implantable cardioverter-defibrillator (ICD) trials suggest that it is the underlying arrhythmic disorder, rather than the ICD therapy itself, that affects mortality in these patients.
Analysis of the MADIT II, MADIT-RISK, MADIT-CRT, MADIT-RIT, and RAID trials showed that the major determinant of mortality in patients receiving a primary prevention ICD was the arrhythmic substrate that leads to occurrence of fast ventricular tachycardia (VT), defined as ≥ 200 bpm, or ventricular fibrillation (VF), not adverse effects of the ICD shock therapy itself.
Patients experiencing an episode of VT had more than a twofold increased risk for death during a follow-up of 2½ years; however, ICD therapies for VT less than 200 bpm and inappropriate ICD shocks were not associated with a higher risk for death.
The findings were published online in the Journal of the American College of Cardiology.
“We know that patients receiving an ICD shock have increased mortality during subsequent follow-up,” first author Mehmet K. Aktas, MD, MBA, University of Rochester (N.Y.), said in an interview.
“There are conflicting data on the impact of ICD shocks on subsequent mortality, and in this study, we aimed to determine whether shocks per se increase subsequent mortality risk or whether the arrhythmic substrate that leads to ICD therapy results in subsequent risk of death,” Dr. Aktas said.
He and his team evaluated the association of ICD therapy with subsequent mortality according to the type of ICD therapy (model I), type of arrhythmia for which ICD therapy was delivered (model II), combined assessment of all arrhythmia and therapy types during follow-up (model III), and incremental risk associated with repeated ICD shocks (model IV).
The study cohort included 5,516 patients. Of these, 1,001 patients (18%) received appropriate ICD therapy and 561 (10%) received inappropriate ICD therapy during an average of 2.4 years.
Patients receiving an appropriate ICD therapy were more likely to be male and to have prior atrial arrhythmia and nonsustained VT compared with those without ICD therapy.
Patients receiving an inappropriate shock were more likely to be younger, to be African American, and to be less likely to have prior nonsustained VT, compared with those without ICD therapy.
Most patients (90%) were receiving beta-blockers and angiotensin-converting enzyme inhibitors or angiotensin receptor blockers regardless of device therapy during follow-up, and 10% of patients were treated with amiodarone.
In model I, at 3 years, the cumulative probability of death following an appropriate ICD shock was 38% compared with no appropriate ICD shock (P < .001). Inappropriate shock alone was not associated with mortality risk.
In model II, which looked at the type of arrhythmia for which ICD therapy was delivered, the cumulative death rate at 3 years following the first occurrence of ICD therapy for VT ≥ 200 beats/min or VF was 27%, compared with 10% in patients not experiencing VT ≥ 200 beats/min or VF (P < .001).
In model III, the highest risk for death was observed following shocks delivered after a failed antitachycardia pacing (ATP) for fast VT (hazard ratio [HR], 3.05), followed by ICD shock for VF (HR, 2.86), ICD shock for fast VT without a prior ATP (HR, 2.83), and ICD shock for slower VT (< 200 beats/min) without a prior ATP (HR, 2.39).
In contrast, other types of appropriate and inappropriate shock or ATP therapies were not associated with a significant risk increase.
In model IV, which assessed the association of shock therapy counts with the risk for death, two or more ICD appropriate shocks were not associated with increased risk after the first appropriate ICD shock.
“Our findings shed light on the mechanisms associated with increased mortality risk in primary prevention ICD recipients,” Dr. Aktas said.
“Studies that evaluate interventions focused on treating and stabilizing the myocardial substrate, which promotes ventricular tachyarrhythmias, such as catheter ablation, are needed to improve survival in heart failure patients,” he added.
Thoughtful study design
In an accompanying editorial, Rajat Deo, MD, and Naga Venkata K. Pothineni, MD, both from the University of Pennsylvania, Philadelphia, praised the researchers for their “thoughtful study design.”
“The take-home message that is most relevant to our clinical practice is clear: Sustained ventricular arrhythmias are a prognostic marker of death and heart failure hospitalization,” they wrote.
The editorialists also commented on the higher rate of inappropriate ICD therapies in African Americans.
“It is concerning to observe that Black patients had a markedly higher rate of inappropriate ICD therapies compared with White patients – and this was in the setting of some of the most respectable, established, and well-funded clinical trials,” they wrote.
Reasons for disparities in outcomes include access to appropriate and affordable medical therapies, access to specialty clinics and caregivers, remote ICD monitoring, and compliance issues.
“Future work will need to understand how the social determinants of health including race affect the treatment and outcomes of our primary prevention ICD population,” they wrote.
Identifying and characterizing the arrhythmic substrate will become a key component of sudden cardiac death risk stratification, the editorialists predicted.
“Concurrently, we must continue to partner with industry colleagues and work with our professional societies to ensure health equity across our patient population,” they concluded.
Dr. Aktas has received research grants from Boston Scientific and Medtronic. Dr. Deo and his coeditorialists report no relevant financial relationships. The MADIT trials were funded by an unrestricted research grant from Boston Scientific to the University of Rochester Medical Center. The RAID trial was funded by the National Institutes of Health.
A version of this article first appeared on Medscape.com.
Combined data from five implantable cardioverter-defibrillator (ICD) trials suggest that it is the underlying arrhythmic disorder, rather than the ICD therapy itself, that affects mortality in these patients.
Analysis of the MADIT II, MADIT-RISK, MADIT-CRT, MADIT-RIT, and RAID trials showed that the major determinant of mortality in patients receiving a primary prevention ICD was the arrhythmic substrate that leads to occurrence of fast ventricular tachycardia (VT), defined as ≥ 200 bpm, or ventricular fibrillation (VF), not adverse effects of the ICD shock therapy itself.
Patients experiencing an episode of VT had more than a twofold increased risk for death during a follow-up of 2½ years; however, ICD therapies for VT less than 200 bpm and inappropriate ICD shocks were not associated with a higher risk for death.
The findings were published online in the Journal of the American College of Cardiology.
“We know that patients receiving an ICD shock have increased mortality during subsequent follow-up,” first author Mehmet K. Aktas, MD, MBA, University of Rochester (N.Y.), said in an interview.
“There are conflicting data on the impact of ICD shocks on subsequent mortality, and in this study, we aimed to determine whether shocks per se increase subsequent mortality risk or whether the arrhythmic substrate that leads to ICD therapy results in subsequent risk of death,” Dr. Aktas said.
He and his team evaluated the association of ICD therapy with subsequent mortality according to the type of ICD therapy (model I), type of arrhythmia for which ICD therapy was delivered (model II), combined assessment of all arrhythmia and therapy types during follow-up (model III), and incremental risk associated with repeated ICD shocks (model IV).
The study cohort included 5,516 patients. Of these, 1,001 patients (18%) received appropriate ICD therapy and 561 (10%) received inappropriate ICD therapy during an average of 2.4 years.
Patients receiving an appropriate ICD therapy were more likely to be male and to have prior atrial arrhythmia and nonsustained VT compared with those without ICD therapy.
Patients receiving an inappropriate shock were more likely to be younger, to be African American, and to be less likely to have prior nonsustained VT, compared with those without ICD therapy.
Most patients (90%) were receiving beta-blockers and angiotensin-converting enzyme inhibitors or angiotensin receptor blockers regardless of device therapy during follow-up, and 10% of patients were treated with amiodarone.
In model I, at 3 years, the cumulative probability of death following an appropriate ICD shock was 38% compared with no appropriate ICD shock (P < .001). Inappropriate shock alone was not associated with mortality risk.
In model II, which looked at the type of arrhythmia for which ICD therapy was delivered, the cumulative death rate at 3 years following the first occurrence of ICD therapy for VT ≥ 200 beats/min or VF was 27%, compared with 10% in patients not experiencing VT ≥ 200 beats/min or VF (P < .001).
In model III, the highest risk for death was observed following shocks delivered after a failed antitachycardia pacing (ATP) for fast VT (hazard ratio [HR], 3.05), followed by ICD shock for VF (HR, 2.86), ICD shock for fast VT without a prior ATP (HR, 2.83), and ICD shock for slower VT (< 200 beats/min) without a prior ATP (HR, 2.39).
In contrast, other types of appropriate and inappropriate shock or ATP therapies were not associated with a significant risk increase.
In model IV, which assessed the association of shock therapy counts with the risk for death, two or more ICD appropriate shocks were not associated with increased risk after the first appropriate ICD shock.
“Our findings shed light on the mechanisms associated with increased mortality risk in primary prevention ICD recipients,” Dr. Aktas said.
“Studies that evaluate interventions focused on treating and stabilizing the myocardial substrate, which promotes ventricular tachyarrhythmias, such as catheter ablation, are needed to improve survival in heart failure patients,” he added.
Thoughtful study design
In an accompanying editorial, Rajat Deo, MD, and Naga Venkata K. Pothineni, MD, both from the University of Pennsylvania, Philadelphia, praised the researchers for their “thoughtful study design.”
“The take-home message that is most relevant to our clinical practice is clear: Sustained ventricular arrhythmias are a prognostic marker of death and heart failure hospitalization,” they wrote.
The editorialists also commented on the higher rate of inappropriate ICD therapies in African Americans.
“It is concerning to observe that Black patients had a markedly higher rate of inappropriate ICD therapies compared with White patients – and this was in the setting of some of the most respectable, established, and well-funded clinical trials,” they wrote.
Reasons for disparities in outcomes include access to appropriate and affordable medical therapies, access to specialty clinics and caregivers, remote ICD monitoring, and compliance issues.
“Future work will need to understand how the social determinants of health including race affect the treatment and outcomes of our primary prevention ICD population,” they wrote.
Identifying and characterizing the arrhythmic substrate will become a key component of sudden cardiac death risk stratification, the editorialists predicted.
“Concurrently, we must continue to partner with industry colleagues and work with our professional societies to ensure health equity across our patient population,” they concluded.
Dr. Aktas has received research grants from Boston Scientific and Medtronic. Dr. Deo and his coeditorialists report no relevant financial relationships. The MADIT trials were funded by an unrestricted research grant from Boston Scientific to the University of Rochester Medical Center. The RAID trial was funded by the National Institutes of Health.
A version of this article first appeared on Medscape.com.
FIDELIO-DKD: Finerenone cuts new-onset AFib in patients with type 2 diabetes and CKD
Finerenone treatment of patients with type 2 diabetes and diabetic kidney disease was linked to a significant drop in the incidence of new-onset atrial fibrillation as a prespecified, exploratory endpoint of the FIDELIO-DKD pivotal trial that randomized more than 5,700 patients.
Treatment with finerenone linked with a 29% relative reduction compared with placebo in incident cases of atrial fibrillation (AFib), Gerasimos Filippatos, MD, reported at the annual scientific sessions of the American College of Cardiology.
The absolute reduction was modest, a 1.3% reduction from the 4.5% incidence rate on placebo to a 3.2% rate on finerenone during a median 2.6 years of follow-up. Concurrently with the report, the results appeared online (J Am Coll Cardiol. 2021 May 17. doi: 10.1016/j.jacc.2021.04.079).
The analyses Dr. Filippatos presented also showed that whether or not patients had a history of AFib, there was no impact on either the primary benefit from finerenone treatment seen in FIDELIO-DKD, which was a significant 18% relative risk reduction compared with placebo in the combined rate of kidney failure, a 40% or greater decline from baseline in estimated glomerular filtration rate, or renal death.
Likewise, prior AFib status had no effect on the study’s key secondary endpoint, a significant 14% relative risk reduction in the combined rate of cardiovascular death, nonfatal MI, nonfatal stroke, or hospitalization for heart failure.
The primary results from FIDELIO-DKD (Efficacy and Safety of Finerenone in Subjects With Type 2 Diabetes Mellitus and Diabetic Kidney Disease) appeared in a 2020 report (N Engl J Med. 2020 Dec 3;383[23];2219-29).
‘Side benefits can be very helpful’
“It’s important to know of finerenone’s benefits beyond the primary outcome of a trial because side benefits can be very helpful,” said Anne B. Curtis, MD, an electrophysiologist and professor and chair of medicine at the University of Buffalo (N.Y.) School of Medicine and Biomedical Sciences. “It’s not a huge benefit, but this could be an added benefit for selected patients,” she said during a press briefing. “Background studies had shown favorable remodeling of the heart [by finerenone] that could affect AFib.”
Possible mitigating effects by finerenone on inflammation and fibrosis might also mediate the drug’s apparent effect on AFib, said Dr. Filippatos, professor of cardiology and director of the Heart Failure and Cardio-Oncology Clinic at Attikon University Hospital and the University of Athens.
He noted that additional data addressing a possible AFib effect of finerenone will emerge soon from the FIGARO-DKD trial, which enrolled patients similar to those in FIDELIO-DKD but with more moderate stages of kidney disease, and from the FINEARTS-HF trial, which is examining the effect of finerenone in patients with heart failure with an ejection fraction of at least 40%.
“Heart failure and AFib go together tightly. It’s worth studying this specifically, so we can see whether there is an impact of finerenone on patients with heart failure who may not necessarily have kidney disease or diabetes,” Dr. Curtis said.
Hypothesis-generating findings
The new findings reported by Dr. Filippatos “should be considered hypothesis generating. Until we have more information, upstream therapies, including mineralocorticoid receptor antagonists [MRAs, the umbrella drug class that includes finerenone], should be used in appropriate patient populations based on defined benefits with the hope they will also reduce the development of AFib and atrial flutter over time,” Gerald V. Naccarelli, MD, and coauthors wrote in an editorial that accompanied the report (J Am Coll Cardiol. 2021 May 17. doi: 10.1016/j.jacc.2021.04.080).
The FIDELIO-DKD trial randomized 5,734 patients at 913 sites in 48 countries, including 461 patients with a history of AFib. The observed link of finerenone treatment with a reduced incidence of AFib appeared consistent regardless of patients’ age, sex, race, their kidney characteristics at baseline, baseline levels of systolic blood pressure, serum potassium, body mass index, A1c, or use of glucose-lowering medications.
Finerenone belongs to a new class of MRAs that have a nonsteroidal structure, in contrast with the MRAs spironolactone and eplerenone. This means that finerenone does not produce steroidal-associated adverse effects linked with certain other MRAs, such as gynecomastia, and may also differ in other actions.
FIDELIO-DKD was sponsored by Bayer, the company developing finerenone. Dr. Filippatos has received lecture fees from or participated in the direction of trials on behalf of Bayer, as well as for Amgen, Boehringer Ingelheim, Medtronic, Novartis, Servier, and Vifor. Dr. Curtis is an adviser to and receives honoraria from St. Jude Medical, and receives honoraria from Medtronic. Dr. Naccarelli has been a consultant to Acesion, ARCA, GlaxoSmithKline, Janssen, Milestone, Omeicos, and Sanofi. His coauthors had no disclosures.
Finerenone treatment of patients with type 2 diabetes and diabetic kidney disease was linked to a significant drop in the incidence of new-onset atrial fibrillation as a prespecified, exploratory endpoint of the FIDELIO-DKD pivotal trial that randomized more than 5,700 patients.
Treatment with finerenone linked with a 29% relative reduction compared with placebo in incident cases of atrial fibrillation (AFib), Gerasimos Filippatos, MD, reported at the annual scientific sessions of the American College of Cardiology.
The absolute reduction was modest, a 1.3% reduction from the 4.5% incidence rate on placebo to a 3.2% rate on finerenone during a median 2.6 years of follow-up. Concurrently with the report, the results appeared online (J Am Coll Cardiol. 2021 May 17. doi: 10.1016/j.jacc.2021.04.079).
The analyses Dr. Filippatos presented also showed that whether or not patients had a history of AFib, there was no impact on either the primary benefit from finerenone treatment seen in FIDELIO-DKD, which was a significant 18% relative risk reduction compared with placebo in the combined rate of kidney failure, a 40% or greater decline from baseline in estimated glomerular filtration rate, or renal death.
Likewise, prior AFib status had no effect on the study’s key secondary endpoint, a significant 14% relative risk reduction in the combined rate of cardiovascular death, nonfatal MI, nonfatal stroke, or hospitalization for heart failure.
The primary results from FIDELIO-DKD (Efficacy and Safety of Finerenone in Subjects With Type 2 Diabetes Mellitus and Diabetic Kidney Disease) appeared in a 2020 report (N Engl J Med. 2020 Dec 3;383[23];2219-29).
‘Side benefits can be very helpful’
“It’s important to know of finerenone’s benefits beyond the primary outcome of a trial because side benefits can be very helpful,” said Anne B. Curtis, MD, an electrophysiologist and professor and chair of medicine at the University of Buffalo (N.Y.) School of Medicine and Biomedical Sciences. “It’s not a huge benefit, but this could be an added benefit for selected patients,” she said during a press briefing. “Background studies had shown favorable remodeling of the heart [by finerenone] that could affect AFib.”
Possible mitigating effects by finerenone on inflammation and fibrosis might also mediate the drug’s apparent effect on AFib, said Dr. Filippatos, professor of cardiology and director of the Heart Failure and Cardio-Oncology Clinic at Attikon University Hospital and the University of Athens.
He noted that additional data addressing a possible AFib effect of finerenone will emerge soon from the FIGARO-DKD trial, which enrolled patients similar to those in FIDELIO-DKD but with more moderate stages of kidney disease, and from the FINEARTS-HF trial, which is examining the effect of finerenone in patients with heart failure with an ejection fraction of at least 40%.
“Heart failure and AFib go together tightly. It’s worth studying this specifically, so we can see whether there is an impact of finerenone on patients with heart failure who may not necessarily have kidney disease or diabetes,” Dr. Curtis said.
Hypothesis-generating findings
The new findings reported by Dr. Filippatos “should be considered hypothesis generating. Until we have more information, upstream therapies, including mineralocorticoid receptor antagonists [MRAs, the umbrella drug class that includes finerenone], should be used in appropriate patient populations based on defined benefits with the hope they will also reduce the development of AFib and atrial flutter over time,” Gerald V. Naccarelli, MD, and coauthors wrote in an editorial that accompanied the report (J Am Coll Cardiol. 2021 May 17. doi: 10.1016/j.jacc.2021.04.080).
The FIDELIO-DKD trial randomized 5,734 patients at 913 sites in 48 countries, including 461 patients with a history of AFib. The observed link of finerenone treatment with a reduced incidence of AFib appeared consistent regardless of patients’ age, sex, race, their kidney characteristics at baseline, baseline levels of systolic blood pressure, serum potassium, body mass index, A1c, or use of glucose-lowering medications.
Finerenone belongs to a new class of MRAs that have a nonsteroidal structure, in contrast with the MRAs spironolactone and eplerenone. This means that finerenone does not produce steroidal-associated adverse effects linked with certain other MRAs, such as gynecomastia, and may also differ in other actions.
FIDELIO-DKD was sponsored by Bayer, the company developing finerenone. Dr. Filippatos has received lecture fees from or participated in the direction of trials on behalf of Bayer, as well as for Amgen, Boehringer Ingelheim, Medtronic, Novartis, Servier, and Vifor. Dr. Curtis is an adviser to and receives honoraria from St. Jude Medical, and receives honoraria from Medtronic. Dr. Naccarelli has been a consultant to Acesion, ARCA, GlaxoSmithKline, Janssen, Milestone, Omeicos, and Sanofi. His coauthors had no disclosures.
Finerenone treatment of patients with type 2 diabetes and diabetic kidney disease was linked to a significant drop in the incidence of new-onset atrial fibrillation as a prespecified, exploratory endpoint of the FIDELIO-DKD pivotal trial that randomized more than 5,700 patients.
Treatment with finerenone linked with a 29% relative reduction compared with placebo in incident cases of atrial fibrillation (AFib), Gerasimos Filippatos, MD, reported at the annual scientific sessions of the American College of Cardiology.
The absolute reduction was modest, a 1.3% reduction from the 4.5% incidence rate on placebo to a 3.2% rate on finerenone during a median 2.6 years of follow-up. Concurrently with the report, the results appeared online (J Am Coll Cardiol. 2021 May 17. doi: 10.1016/j.jacc.2021.04.079).
The analyses Dr. Filippatos presented also showed that whether or not patients had a history of AFib, there was no impact on either the primary benefit from finerenone treatment seen in FIDELIO-DKD, which was a significant 18% relative risk reduction compared with placebo in the combined rate of kidney failure, a 40% or greater decline from baseline in estimated glomerular filtration rate, or renal death.
Likewise, prior AFib status had no effect on the study’s key secondary endpoint, a significant 14% relative risk reduction in the combined rate of cardiovascular death, nonfatal MI, nonfatal stroke, or hospitalization for heart failure.
The primary results from FIDELIO-DKD (Efficacy and Safety of Finerenone in Subjects With Type 2 Diabetes Mellitus and Diabetic Kidney Disease) appeared in a 2020 report (N Engl J Med. 2020 Dec 3;383[23];2219-29).
‘Side benefits can be very helpful’
“It’s important to know of finerenone’s benefits beyond the primary outcome of a trial because side benefits can be very helpful,” said Anne B. Curtis, MD, an electrophysiologist and professor and chair of medicine at the University of Buffalo (N.Y.) School of Medicine and Biomedical Sciences. “It’s not a huge benefit, but this could be an added benefit for selected patients,” she said during a press briefing. “Background studies had shown favorable remodeling of the heart [by finerenone] that could affect AFib.”
Possible mitigating effects by finerenone on inflammation and fibrosis might also mediate the drug’s apparent effect on AFib, said Dr. Filippatos, professor of cardiology and director of the Heart Failure and Cardio-Oncology Clinic at Attikon University Hospital and the University of Athens.
He noted that additional data addressing a possible AFib effect of finerenone will emerge soon from the FIGARO-DKD trial, which enrolled patients similar to those in FIDELIO-DKD but with more moderate stages of kidney disease, and from the FINEARTS-HF trial, which is examining the effect of finerenone in patients with heart failure with an ejection fraction of at least 40%.
“Heart failure and AFib go together tightly. It’s worth studying this specifically, so we can see whether there is an impact of finerenone on patients with heart failure who may not necessarily have kidney disease or diabetes,” Dr. Curtis said.
Hypothesis-generating findings
The new findings reported by Dr. Filippatos “should be considered hypothesis generating. Until we have more information, upstream therapies, including mineralocorticoid receptor antagonists [MRAs, the umbrella drug class that includes finerenone], should be used in appropriate patient populations based on defined benefits with the hope they will also reduce the development of AFib and atrial flutter over time,” Gerald V. Naccarelli, MD, and coauthors wrote in an editorial that accompanied the report (J Am Coll Cardiol. 2021 May 17. doi: 10.1016/j.jacc.2021.04.080).
The FIDELIO-DKD trial randomized 5,734 patients at 913 sites in 48 countries, including 461 patients with a history of AFib. The observed link of finerenone treatment with a reduced incidence of AFib appeared consistent regardless of patients’ age, sex, race, their kidney characteristics at baseline, baseline levels of systolic blood pressure, serum potassium, body mass index, A1c, or use of glucose-lowering medications.
Finerenone belongs to a new class of MRAs that have a nonsteroidal structure, in contrast with the MRAs spironolactone and eplerenone. This means that finerenone does not produce steroidal-associated adverse effects linked with certain other MRAs, such as gynecomastia, and may also differ in other actions.
FIDELIO-DKD was sponsored by Bayer, the company developing finerenone. Dr. Filippatos has received lecture fees from or participated in the direction of trials on behalf of Bayer, as well as for Amgen, Boehringer Ingelheim, Medtronic, Novartis, Servier, and Vifor. Dr. Curtis is an adviser to and receives honoraria from St. Jude Medical, and receives honoraria from Medtronic. Dr. Naccarelli has been a consultant to Acesion, ARCA, GlaxoSmithKline, Janssen, Milestone, Omeicos, and Sanofi. His coauthors had no disclosures.
FROM ACC 2021
Dapagliflozin misses as treatment for COVID-19 but leaves intriguing signal for benefit
In patients hospitalized with COVID-19 infection, the sodium-glucose transporter 2 inhibitor dapagliflozin showed a trend for benefit relative to placebo on multiple outcomes, including the primary outcome of time to organ failure or death, according to results from the randomized DARE-19 trial.
Because of the failure to reach statistical significance, these results have no immediate relevance, but the trends support interest in further testing SGLT2 inhibitors in acute diseases posing a high risk for organ failure, according to Mikhail Kosiborod, MD.
In a trial that did not meet its primary endpoint, Dr. Kosiborod acknowledged that positive interpretations are speculative, but he does believe that there is one immediate take-home message.
“Our results do not support discontinuation of SGLT2 inhibitors in the setting of COVID-19 as long as patients are monitored,” said Dr. Kosiborod, director of cardiometabolic research at Saint Luke’s Mid-America Heart Institute, Kansas City, Mo.
At many institutions, it has been common to discontinue SGLT2 inhibitors in patients admitted with COVID-19. One reason was the concern that drugs in this class could exacerbate organ damage, particularly if they were to induced ketoacidosis. However, only 2 (0.003%) of 613 patients treated with dapagliflozin developed ketoacidosis, and the signal for organ protection overall, although not significant, was consistent.
“Numerically, fewer patients treated with dapagliflozin experienced organ failure and death, and this was consistent across systems, including the kidney,” Dr. Kosiborod said in presenting the study at the annual scientific sessions of the American College of Cardiology.
Overall, the study suggests that, in the context of COVID-19, dapagliflozin did not show harm and might have potential benefit, he added.
DARE-19 was rapidly conceived, designed, and implemented during the early stages of the COVID-19 pandemic. Based on prior evidence that SGLT2 inhibitors “favorably affect a number of pathophysiologic pathways disrupted during acute illness” and that drugs in this class have provided organ protection in the context of heart failure, chronic kidney disease, and other cardiometabolic conditions, the study was designed to test the hypothesis that this mechanism might improve outcomes in patients hospitalized with COVID-19, Dr. Kosiborod said.
The entry criteria included confirmed or suspected COVID-19 with an onset of 4 days of fewer and one additional risk factor, such as atherosclerotic cardiovascular disease, hypertension, or type 2 diabetes. Patients with significant renal impairment or a history of diabetic ketoacidosis were excluded.
On top of standard treatments for COVID-19, patients were randomized to 10 mg dapagliflozin or placebo once daily. There were two primary endpoints. That of prevention was time to criteria for respiratory, cardiovascular, or renal organ failure or death. The second primary outcome, for recovery, was a hierarchical composite for four endpoints: death, organ failure, status at 30 days if hospitalized, and time to discharge if this occurred before day 30.
Of the 1,250 patients randomized at 95 sites in seven countries, 617 in the dapagliflozin group and 620 patients in the placebo group completed the study. Baseline characteristics, which included a mean of age of 62 years; types of comorbidities; and types of treatments were similar.
Results for two primary endpoints
The curves for the primary outcome of prevention had already separated by day 3 and continued to widen over the 30 days in which outcomes were compared. At the end of 30 days, 11.2% of the dapagliflozin group and 13.8% of the placebo group had an event. By hazard ratio, dapagliflozin was linked to 20% nonsignificant relative protection from events (hazard ratio, 0.80; 95% confidence interval, 0.58-1.10).
The trend (P = .168) for the primary endpoint for prevention was reflected in the individual components. For dapagliflozin related to placebo, there were generally similar or greater reductions in new or worsening organ failure (HR, 0.80), cardiac decompensation (HR, 0.81), respiratory decompensation (HR, 0.85), and kidney decompensation (HR, 0.65). None were statistically significant, but the confidence intervals were tight with the upper end never exceeding 1.20.
Moreover, the relative risk reduction for all-cause mortality moved in the same direction (HR, 0.77; 95% CI, 0.52-1.16).
In the hierarchical composite endpoint of recovery, there was no significant difference in the time to discharge, but again many recovery metrics numerically favored dapagliflozin with an overall difference producing a statistical trend (P = .14) similar to organ failure events and death.
In safety analyses, dapagliflozin consistently outperformed placebo across a broad array of safety measure, including any severe adverse event (65% vs. 82%), any adverse event with an outcome of death (32% vs. 48%), discontinuation caused by an adverse event (44% vs. 55%), and acute kidney injury (21% vs. 34%).
Data could fuel related studies
According to Ana Barac, MD, PhD, director of the cardio-oncology program in the Medstar Heart and Vascular Institute, Washington, these data are “thought provoking.” Although this was a negative trial, she said that it generates an “exciting hypothesis” about the potential of SGLT2 inhibitors to provide organ protection. She called for studies to pursue this path of research.
More immediately, Dr. Barac agreed that these data argue against stopping SGLT2 inhibitors in patients admitted to a hospital for COVID-19 infection.
“These data show that these drugs are not going to lead to harm, but they might lead to benefit,” she said.
For James Januzzi, MD, a cardiologist at Massachusetts General Hospital and professor of medicine at Harvard Medical School, both in Boston, DARE-19 was perhaps most impressive because of its rigorous design and execution in the midst of a pandemic.
Over the past year, “the medical literature was flooded with grossly underpowered, poorly designed, single-center studies” yielding results that have been hard to interpret, Dr. Januzzi said. Despite the fact that this study failed to confirm its hypothesis, he said the investigators deserve praise for the quality of the work.
Dr. Januzzi also believes the study is not without clinically relevant findings, particularly the fact that dapagliflozin was associated with a lower rate of adverse events than placebo. This, at least, provides reassurance about the safety of this drug in the setting of COVID-19 infection.
Dr. Kosiborod reported financial relationships with more than 10 pharmaceutical companies, including AstraZeneca, which provided funding for DARE-19. Dr. Barac reported financial relationships with Bristol-Myers Squibb and CTI BioPharma. Dr. Januzzi reported financial relationships with Boehringer Ingelheim, GE Healthcare, Johnson & Johnson, Merck, Novartis, Pfizer, and Roche.
In patients hospitalized with COVID-19 infection, the sodium-glucose transporter 2 inhibitor dapagliflozin showed a trend for benefit relative to placebo on multiple outcomes, including the primary outcome of time to organ failure or death, according to results from the randomized DARE-19 trial.
Because of the failure to reach statistical significance, these results have no immediate relevance, but the trends support interest in further testing SGLT2 inhibitors in acute diseases posing a high risk for organ failure, according to Mikhail Kosiborod, MD.
In a trial that did not meet its primary endpoint, Dr. Kosiborod acknowledged that positive interpretations are speculative, but he does believe that there is one immediate take-home message.
“Our results do not support discontinuation of SGLT2 inhibitors in the setting of COVID-19 as long as patients are monitored,” said Dr. Kosiborod, director of cardiometabolic research at Saint Luke’s Mid-America Heart Institute, Kansas City, Mo.
At many institutions, it has been common to discontinue SGLT2 inhibitors in patients admitted with COVID-19. One reason was the concern that drugs in this class could exacerbate organ damage, particularly if they were to induced ketoacidosis. However, only 2 (0.003%) of 613 patients treated with dapagliflozin developed ketoacidosis, and the signal for organ protection overall, although not significant, was consistent.
“Numerically, fewer patients treated with dapagliflozin experienced organ failure and death, and this was consistent across systems, including the kidney,” Dr. Kosiborod said in presenting the study at the annual scientific sessions of the American College of Cardiology.
Overall, the study suggests that, in the context of COVID-19, dapagliflozin did not show harm and might have potential benefit, he added.
DARE-19 was rapidly conceived, designed, and implemented during the early stages of the COVID-19 pandemic. Based on prior evidence that SGLT2 inhibitors “favorably affect a number of pathophysiologic pathways disrupted during acute illness” and that drugs in this class have provided organ protection in the context of heart failure, chronic kidney disease, and other cardiometabolic conditions, the study was designed to test the hypothesis that this mechanism might improve outcomes in patients hospitalized with COVID-19, Dr. Kosiborod said.
The entry criteria included confirmed or suspected COVID-19 with an onset of 4 days of fewer and one additional risk factor, such as atherosclerotic cardiovascular disease, hypertension, or type 2 diabetes. Patients with significant renal impairment or a history of diabetic ketoacidosis were excluded.
On top of standard treatments for COVID-19, patients were randomized to 10 mg dapagliflozin or placebo once daily. There were two primary endpoints. That of prevention was time to criteria for respiratory, cardiovascular, or renal organ failure or death. The second primary outcome, for recovery, was a hierarchical composite for four endpoints: death, organ failure, status at 30 days if hospitalized, and time to discharge if this occurred before day 30.
Of the 1,250 patients randomized at 95 sites in seven countries, 617 in the dapagliflozin group and 620 patients in the placebo group completed the study. Baseline characteristics, which included a mean of age of 62 years; types of comorbidities; and types of treatments were similar.
Results for two primary endpoints
The curves for the primary outcome of prevention had already separated by day 3 and continued to widen over the 30 days in which outcomes were compared. At the end of 30 days, 11.2% of the dapagliflozin group and 13.8% of the placebo group had an event. By hazard ratio, dapagliflozin was linked to 20% nonsignificant relative protection from events (hazard ratio, 0.80; 95% confidence interval, 0.58-1.10).
The trend (P = .168) for the primary endpoint for prevention was reflected in the individual components. For dapagliflozin related to placebo, there were generally similar or greater reductions in new or worsening organ failure (HR, 0.80), cardiac decompensation (HR, 0.81), respiratory decompensation (HR, 0.85), and kidney decompensation (HR, 0.65). None were statistically significant, but the confidence intervals were tight with the upper end never exceeding 1.20.
Moreover, the relative risk reduction for all-cause mortality moved in the same direction (HR, 0.77; 95% CI, 0.52-1.16).
In the hierarchical composite endpoint of recovery, there was no significant difference in the time to discharge, but again many recovery metrics numerically favored dapagliflozin with an overall difference producing a statistical trend (P = .14) similar to organ failure events and death.
In safety analyses, dapagliflozin consistently outperformed placebo across a broad array of safety measure, including any severe adverse event (65% vs. 82%), any adverse event with an outcome of death (32% vs. 48%), discontinuation caused by an adverse event (44% vs. 55%), and acute kidney injury (21% vs. 34%).
Data could fuel related studies
According to Ana Barac, MD, PhD, director of the cardio-oncology program in the Medstar Heart and Vascular Institute, Washington, these data are “thought provoking.” Although this was a negative trial, she said that it generates an “exciting hypothesis” about the potential of SGLT2 inhibitors to provide organ protection. She called for studies to pursue this path of research.
More immediately, Dr. Barac agreed that these data argue against stopping SGLT2 inhibitors in patients admitted to a hospital for COVID-19 infection.
“These data show that these drugs are not going to lead to harm, but they might lead to benefit,” she said.
For James Januzzi, MD, a cardiologist at Massachusetts General Hospital and professor of medicine at Harvard Medical School, both in Boston, DARE-19 was perhaps most impressive because of its rigorous design and execution in the midst of a pandemic.
Over the past year, “the medical literature was flooded with grossly underpowered, poorly designed, single-center studies” yielding results that have been hard to interpret, Dr. Januzzi said. Despite the fact that this study failed to confirm its hypothesis, he said the investigators deserve praise for the quality of the work.
Dr. Januzzi also believes the study is not without clinically relevant findings, particularly the fact that dapagliflozin was associated with a lower rate of adverse events than placebo. This, at least, provides reassurance about the safety of this drug in the setting of COVID-19 infection.
Dr. Kosiborod reported financial relationships with more than 10 pharmaceutical companies, including AstraZeneca, which provided funding for DARE-19. Dr. Barac reported financial relationships with Bristol-Myers Squibb and CTI BioPharma. Dr. Januzzi reported financial relationships with Boehringer Ingelheim, GE Healthcare, Johnson & Johnson, Merck, Novartis, Pfizer, and Roche.
In patients hospitalized with COVID-19 infection, the sodium-glucose transporter 2 inhibitor dapagliflozin showed a trend for benefit relative to placebo on multiple outcomes, including the primary outcome of time to organ failure or death, according to results from the randomized DARE-19 trial.
Because of the failure to reach statistical significance, these results have no immediate relevance, but the trends support interest in further testing SGLT2 inhibitors in acute diseases posing a high risk for organ failure, according to Mikhail Kosiborod, MD.
In a trial that did not meet its primary endpoint, Dr. Kosiborod acknowledged that positive interpretations are speculative, but he does believe that there is one immediate take-home message.
“Our results do not support discontinuation of SGLT2 inhibitors in the setting of COVID-19 as long as patients are monitored,” said Dr. Kosiborod, director of cardiometabolic research at Saint Luke’s Mid-America Heart Institute, Kansas City, Mo.
At many institutions, it has been common to discontinue SGLT2 inhibitors in patients admitted with COVID-19. One reason was the concern that drugs in this class could exacerbate organ damage, particularly if they were to induced ketoacidosis. However, only 2 (0.003%) of 613 patients treated with dapagliflozin developed ketoacidosis, and the signal for organ protection overall, although not significant, was consistent.
“Numerically, fewer patients treated with dapagliflozin experienced organ failure and death, and this was consistent across systems, including the kidney,” Dr. Kosiborod said in presenting the study at the annual scientific sessions of the American College of Cardiology.
Overall, the study suggests that, in the context of COVID-19, dapagliflozin did not show harm and might have potential benefit, he added.
DARE-19 was rapidly conceived, designed, and implemented during the early stages of the COVID-19 pandemic. Based on prior evidence that SGLT2 inhibitors “favorably affect a number of pathophysiologic pathways disrupted during acute illness” and that drugs in this class have provided organ protection in the context of heart failure, chronic kidney disease, and other cardiometabolic conditions, the study was designed to test the hypothesis that this mechanism might improve outcomes in patients hospitalized with COVID-19, Dr. Kosiborod said.
The entry criteria included confirmed or suspected COVID-19 with an onset of 4 days of fewer and one additional risk factor, such as atherosclerotic cardiovascular disease, hypertension, or type 2 diabetes. Patients with significant renal impairment or a history of diabetic ketoacidosis were excluded.
On top of standard treatments for COVID-19, patients were randomized to 10 mg dapagliflozin or placebo once daily. There were two primary endpoints. That of prevention was time to criteria for respiratory, cardiovascular, or renal organ failure or death. The second primary outcome, for recovery, was a hierarchical composite for four endpoints: death, organ failure, status at 30 days if hospitalized, and time to discharge if this occurred before day 30.
Of the 1,250 patients randomized at 95 sites in seven countries, 617 in the dapagliflozin group and 620 patients in the placebo group completed the study. Baseline characteristics, which included a mean of age of 62 years; types of comorbidities; and types of treatments were similar.
Results for two primary endpoints
The curves for the primary outcome of prevention had already separated by day 3 and continued to widen over the 30 days in which outcomes were compared. At the end of 30 days, 11.2% of the dapagliflozin group and 13.8% of the placebo group had an event. By hazard ratio, dapagliflozin was linked to 20% nonsignificant relative protection from events (hazard ratio, 0.80; 95% confidence interval, 0.58-1.10).
The trend (P = .168) for the primary endpoint for prevention was reflected in the individual components. For dapagliflozin related to placebo, there were generally similar or greater reductions in new or worsening organ failure (HR, 0.80), cardiac decompensation (HR, 0.81), respiratory decompensation (HR, 0.85), and kidney decompensation (HR, 0.65). None were statistically significant, but the confidence intervals were tight with the upper end never exceeding 1.20.
Moreover, the relative risk reduction for all-cause mortality moved in the same direction (HR, 0.77; 95% CI, 0.52-1.16).
In the hierarchical composite endpoint of recovery, there was no significant difference in the time to discharge, but again many recovery metrics numerically favored dapagliflozin with an overall difference producing a statistical trend (P = .14) similar to organ failure events and death.
In safety analyses, dapagliflozin consistently outperformed placebo across a broad array of safety measure, including any severe adverse event (65% vs. 82%), any adverse event with an outcome of death (32% vs. 48%), discontinuation caused by an adverse event (44% vs. 55%), and acute kidney injury (21% vs. 34%).
Data could fuel related studies
According to Ana Barac, MD, PhD, director of the cardio-oncology program in the Medstar Heart and Vascular Institute, Washington, these data are “thought provoking.” Although this was a negative trial, she said that it generates an “exciting hypothesis” about the potential of SGLT2 inhibitors to provide organ protection. She called for studies to pursue this path of research.
More immediately, Dr. Barac agreed that these data argue against stopping SGLT2 inhibitors in patients admitted to a hospital for COVID-19 infection.
“These data show that these drugs are not going to lead to harm, but they might lead to benefit,” she said.
For James Januzzi, MD, a cardiologist at Massachusetts General Hospital and professor of medicine at Harvard Medical School, both in Boston, DARE-19 was perhaps most impressive because of its rigorous design and execution in the midst of a pandemic.
Over the past year, “the medical literature was flooded with grossly underpowered, poorly designed, single-center studies” yielding results that have been hard to interpret, Dr. Januzzi said. Despite the fact that this study failed to confirm its hypothesis, he said the investigators deserve praise for the quality of the work.
Dr. Januzzi also believes the study is not without clinically relevant findings, particularly the fact that dapagliflozin was associated with a lower rate of adverse events than placebo. This, at least, provides reassurance about the safety of this drug in the setting of COVID-19 infection.
Dr. Kosiborod reported financial relationships with more than 10 pharmaceutical companies, including AstraZeneca, which provided funding for DARE-19. Dr. Barac reported financial relationships with Bristol-Myers Squibb and CTI BioPharma. Dr. Januzzi reported financial relationships with Boehringer Ingelheim, GE Healthcare, Johnson & Johnson, Merck, Novartis, Pfizer, and Roche.
FROM ACC 2021
Novel rehab program fights frailty, boosts capacity in advanced HF
A novel physical rehabilitation program for patients with advanced heart failure that aimed to improve their ability to exercise before focusing on endurance was successful in a randomized trial in ways that seem to have eluded some earlier exercise-training studies in the setting of HF.
The often-frail patients following the training regimen, initiated before discharge from hospitalization for acute decompensation, worked on capabilities such as mobility, balance, and strength deemed necessary if exercises meant to build exercise capacity were to succeed.
A huge percentage stayed with the 12-week program, which featured personalized, one-on-one training from a physical therapist. The patients benefited, with improvements in balance, walking ability, and strength, which were followed by significant gains in 6-minute walk distance (6MWD) and measures of physical functioning, frailty, and quality of life. The patients then continued elements of the program at home out to 6 months.
At that time, death and rehospitalizations did not differ between those assigned to the regimen and similar patients who had not participated in the program, although the trial wasn’t powered for clinical events.
The rehab strategy seemed to work across a wide range of patient subgroups. In particular, there was evidence that the benefits were more pronounced in patients with HF and preserved ejection fraction (HFpEF) than in those with HF and reduced ejection fraction (HFrEF), observed Dalane W. Kitzman, MD, Wake Forest University, Winston-Salem, N.C.
Dr. Kitzman presented results from the REHAB-HF (Rehabilitation Therapy in Older Acute Heart Failure Patients) trial at the annual scientific sessions of the American College of Cardiology and is lead author on its same-day publication in the New England Journal of Medicine.
An earlier pilot program unexpectedly showed that such patients recently hospitalized with HF “have significant impairments in mobility and balance,” he explained. If so, “it would be hazardous to subject them to traditional endurance training, such as walking-based treadmill or even bicycle.”
The unusual program, said Dr. Kitzman, looks to those issues before engaging the patients in endurance exercise by addressing mobility, balance, and basic strength – enough to repeatedly stand up from a sitting position, for example. “If you’re not able to stand with confidence, then you’re not able to walk on a treadmill.”
This model of exercise rehab “is used in geriatrics research, and enables them to safely increase endurance. It’s well known from geriatric studies that if you go directly to endurance in these, frail, older patients, you have little improvement and often have injuries and falls,” he added.
Guidance from telemedicine?
The functional outcomes examined in REHAB-HF “are the ones that matter to patients the most,” observed Eileen M. Handberg, PhD, of Shands Hospital at the University of Florida, Gainesville, at a presentation on the trial for the media.
“This is about being able to get out of a chair without assistance, not falling, walking farther, and feeling better as opposed to the more traditional outcome measure that has been used in cardiac rehab trials, which has been the exercise treadmill test – which most patients don’t have the capacity to do very well anyway,” said Dr. Handberg, who is not a part of REHAB-HF.
“This opens up rehab, potentially, to the more sick, who also need a better quality of life,” she said.
However, many patients invited to participate in the trial could not because they lived too far from the program, Dr. Handberg observed. “It would be nice to see if the lessons from COVID-19 might apply to this population” by making participation possible remotely, “perhaps using family members as rehab assistance,” she said.
“I was really very impressed that you had 83% adherence to a home exercise 6 months down the road, which far eclipses what we had in HF-ACTION,” said Vera Bittner, MD, University of Alabama at Birmingham, as the invited discussant following Dr. Kitzman’s formal presentation of the trial. “And it certainly eclipses what we see in the typical cardiac rehab program.”
Both Dr. Bittner and Dr. Kitzman participated in HF-ACTION, a randomized exercise-training trial for patients with chronic, stable HFrEF who were all-around less sick than those in REHAB-HF.
Four functional domains
Historically, HF exercise or rehab trials have excluded patients hospitalized with acute decompensation, and third-party reimbursement often has not covered such programs because of a lack of supporting evidence and a supposed potential for harm, Dr. Kitzman said.
Entry to REHAB-HF required the patients to be fit enough to walk 4 meters, with or without a walker or other assistant device, and to have been in the hospital for at least 24 hours with a primary diagnosis of acute decompensated HF.
The intervention relied on exercises aimed at improving the four functional domains of strength, balance, mobility, and – when those three were sufficiently developed – endurance, Dr. Kitzman and associates wrote in their published report.
“The intervention was initiated in the hospital when feasible and was subsequently transitioned to an outpatient facility as soon as possible after discharge,” they wrote. Afterward, “a key goal of the intervention during the first 3 months [the outpatient phase] was to prepare the patient to transition to the independent maintenance phase (months 4-6).”
The study’s control patients “received frequent calls from study staff to try to approximate the increased attention received by the intervention group,” Dr. Kitzman said in an interview. “They were allowed to receive all usual care as ordered by their treating physicians. This included, if ordered, standard physical therapy or cardiac rehabilitation” in 43% of the control cohort. Of the trial’s 349 patients, those assigned to the intervention scored significantly higher on the three-component Short Physical Performance Battery (SPPB) at 12 weeks than those assigned to a usual care approach that included, for some, more conventional cardiac rehabilitation (8.3 vs. 6.9; P < .001).
The SPPB, validated in trials as a proxy for clinical outcomes includes tests of balance while standing, gait speed during a 4-minute walk, and strength. The latter is the test that measures time needed to rise from a chair five times.
They also showed consistent gains in other measures of physical functioning and quality of life by 12 weeks months.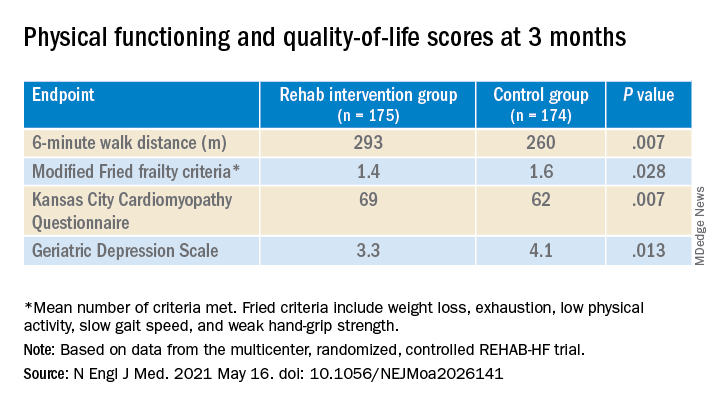
The observed SPPB treatment effect is “impressive” and “compares very favorably with previously reported estimates,” observed an accompanying editorial from Stefan D. Anker, MD, PhD, of the German Center for Cardiovascular Research and Charité Universitätsmedizin, Berlin, and Andrew J.S. Coats, DM, of the University of Warwick, Coventry, England.
“Similarly, the between-group differences seen in 6-minute walk distance (34 m) and gait speed (0.12 m/s) are clinically meaningful and sizable.”
They propose that some of the substantial quality-of-life benefit in the intervention group “may be due to better physical performance, and that part may be due to improvements in psychosocial factors and mood. It appears that exercise also resulted in patients becoming happier, or at least less depressed, as evidenced by the positive results on the Geriatric Depression Scale.”
Similar results across most subgroups
In subgroup analyses, the intervention was successful against the standard-care approach in both men and women at all ages and regardless of ejection fraction; symptom status; and whether the patient had diabetes, ischemic heart disease, or atrial fibrillation, or was obese.
Clinical outcomes were not significantly different at 6 months. The rate of death from any cause was 13% for the intervention group and 10% for the control group. There were 194 and 213 hospitalizations from any cause, respectively.
Not included in the trial’s current publication but soon to be published, Dr. Kitzman said when interviewed, is a comparison of outcomes in patients with HFpEF and HFrEF. “We found at baseline that those with HFpEF had worse impairment in physical function, quality of life, and frailty. After the intervention, there appeared to be consistently larger improvements in all outcomes, including SPPB, 6-minute walk, qualify of life, and frailty, in HFpEF versus HFrEF.”
The signals of potential benefit in HFpEF extended to clinical endpoints, he said. In contrast to similar rates of all-cause rehospitalization in HFrEF, “in patients with HFpEF, rehospitalizations were 17% lower in the intervention group, compared to the control group.” Still, he noted, the interaction P value wasn’t significant.
However, Dr. Kitzman added, mortality in the intervention group, compared with the control group, was reduced by 35% among patients with HFpEF, “but was 250% higher in HFrEF,” with a significant interaction P value.
He was careful to note that, as a phase 2 trial, REHAB-HF was underpowered for clinical events, “and even the results in the HFpEF group should not be seen as adequate evidence to change clinical care.” They were from an exploratory analysis that included relatively few events.
“Because definitive demonstration of improvement in clinical events is critical for altering clinical care guidelines and for third-party payer reimbursement decisions, we believe that a subsequent phase 3 trial is needed and are currently planning toward that,” Dr. Kitzman said.
The study was supported by research grants from the National Institutes of Health, the Kermit Glenn Phillips II Chair in Cardiovascular Medicine, and the Oristano Family Fund at Wake Forest. Dr. Kitzman disclosed receiving consulting fees or honoraria from AbbVie, AstraZeneca, Bayer Healthcare, Boehringer Ingelheim, CinRx, Corviamedical, GlaxoSmithKline, and Merck; and having an unspecified relationship with Gilead. Dr. Handberg disclosed receiving grants from Aastom Biosciences, Abbott Laboratories, Amgen, Amorcyte, AstraZeneca, Biocardia, Boehringer Ingelheim, Capricor, Cytori Therapeutics, Department of Defense, Direct Flow Medical, Everyfit, Gilead, Ionis, Medtronic, Merck, Mesoblast, Relypsa, and Sanofi-Aventis. Dr. Bittner discloses receiving consulting fees or honoraria from Pfizer and Sanofi; receiving research grants from Amgen and The Medicines Company; and having unspecified relationships with AstraZeneca, DalCor, Esperion, and Sanofi-Aventis. Dr. Anker reported receiving grants and personal fees from Abbott Vascular and Vifor; personal fees from Bayer, Boehringer Ingelheim, Novartis, Servier, Cardiac Dimensions, Thermo Fisher Scientific, AstraZeneca, Occlutech, Actimed, and Respicardia. Dr. Coats disclosed receiving personal fees from AstraZeneca, Bayer, Boehringer Ingelheim, Menarini, Novartis, Nutricia, Servier, Vifor, Abbott, Actimed, Arena, Cardiac Dimensions, Corvia, CVRx, Enopace, ESN Cleer, Faraday, WL Gore, Impulse Dynamics, and Respicardia.
A version of this article first appeared on Medscape.com.
A novel physical rehabilitation program for patients with advanced heart failure that aimed to improve their ability to exercise before focusing on endurance was successful in a randomized trial in ways that seem to have eluded some earlier exercise-training studies in the setting of HF.
The often-frail patients following the training regimen, initiated before discharge from hospitalization for acute decompensation, worked on capabilities such as mobility, balance, and strength deemed necessary if exercises meant to build exercise capacity were to succeed.
A huge percentage stayed with the 12-week program, which featured personalized, one-on-one training from a physical therapist. The patients benefited, with improvements in balance, walking ability, and strength, which were followed by significant gains in 6-minute walk distance (6MWD) and measures of physical functioning, frailty, and quality of life. The patients then continued elements of the program at home out to 6 months.
At that time, death and rehospitalizations did not differ between those assigned to the regimen and similar patients who had not participated in the program, although the trial wasn’t powered for clinical events.
The rehab strategy seemed to work across a wide range of patient subgroups. In particular, there was evidence that the benefits were more pronounced in patients with HF and preserved ejection fraction (HFpEF) than in those with HF and reduced ejection fraction (HFrEF), observed Dalane W. Kitzman, MD, Wake Forest University, Winston-Salem, N.C.
Dr. Kitzman presented results from the REHAB-HF (Rehabilitation Therapy in Older Acute Heart Failure Patients) trial at the annual scientific sessions of the American College of Cardiology and is lead author on its same-day publication in the New England Journal of Medicine.
An earlier pilot program unexpectedly showed that such patients recently hospitalized with HF “have significant impairments in mobility and balance,” he explained. If so, “it would be hazardous to subject them to traditional endurance training, such as walking-based treadmill or even bicycle.”
The unusual program, said Dr. Kitzman, looks to those issues before engaging the patients in endurance exercise by addressing mobility, balance, and basic strength – enough to repeatedly stand up from a sitting position, for example. “If you’re not able to stand with confidence, then you’re not able to walk on a treadmill.”
This model of exercise rehab “is used in geriatrics research, and enables them to safely increase endurance. It’s well known from geriatric studies that if you go directly to endurance in these, frail, older patients, you have little improvement and often have injuries and falls,” he added.
Guidance from telemedicine?
The functional outcomes examined in REHAB-HF “are the ones that matter to patients the most,” observed Eileen M. Handberg, PhD, of Shands Hospital at the University of Florida, Gainesville, at a presentation on the trial for the media.
“This is about being able to get out of a chair without assistance, not falling, walking farther, and feeling better as opposed to the more traditional outcome measure that has been used in cardiac rehab trials, which has been the exercise treadmill test – which most patients don’t have the capacity to do very well anyway,” said Dr. Handberg, who is not a part of REHAB-HF.
“This opens up rehab, potentially, to the more sick, who also need a better quality of life,” she said.
However, many patients invited to participate in the trial could not because they lived too far from the program, Dr. Handberg observed. “It would be nice to see if the lessons from COVID-19 might apply to this population” by making participation possible remotely, “perhaps using family members as rehab assistance,” she said.
“I was really very impressed that you had 83% adherence to a home exercise 6 months down the road, which far eclipses what we had in HF-ACTION,” said Vera Bittner, MD, University of Alabama at Birmingham, as the invited discussant following Dr. Kitzman’s formal presentation of the trial. “And it certainly eclipses what we see in the typical cardiac rehab program.”
Both Dr. Bittner and Dr. Kitzman participated in HF-ACTION, a randomized exercise-training trial for patients with chronic, stable HFrEF who were all-around less sick than those in REHAB-HF.
Four functional domains
Historically, HF exercise or rehab trials have excluded patients hospitalized with acute decompensation, and third-party reimbursement often has not covered such programs because of a lack of supporting evidence and a supposed potential for harm, Dr. Kitzman said.
Entry to REHAB-HF required the patients to be fit enough to walk 4 meters, with or without a walker or other assistant device, and to have been in the hospital for at least 24 hours with a primary diagnosis of acute decompensated HF.
The intervention relied on exercises aimed at improving the four functional domains of strength, balance, mobility, and – when those three were sufficiently developed – endurance, Dr. Kitzman and associates wrote in their published report.
“The intervention was initiated in the hospital when feasible and was subsequently transitioned to an outpatient facility as soon as possible after discharge,” they wrote. Afterward, “a key goal of the intervention during the first 3 months [the outpatient phase] was to prepare the patient to transition to the independent maintenance phase (months 4-6).”
The study’s control patients “received frequent calls from study staff to try to approximate the increased attention received by the intervention group,” Dr. Kitzman said in an interview. “They were allowed to receive all usual care as ordered by their treating physicians. This included, if ordered, standard physical therapy or cardiac rehabilitation” in 43% of the control cohort. Of the trial’s 349 patients, those assigned to the intervention scored significantly higher on the three-component Short Physical Performance Battery (SPPB) at 12 weeks than those assigned to a usual care approach that included, for some, more conventional cardiac rehabilitation (8.3 vs. 6.9; P < .001).
The SPPB, validated in trials as a proxy for clinical outcomes includes tests of balance while standing, gait speed during a 4-minute walk, and strength. The latter is the test that measures time needed to rise from a chair five times.
They also showed consistent gains in other measures of physical functioning and quality of life by 12 weeks months.
The observed SPPB treatment effect is “impressive” and “compares very favorably with previously reported estimates,” observed an accompanying editorial from Stefan D. Anker, MD, PhD, of the German Center for Cardiovascular Research and Charité Universitätsmedizin, Berlin, and Andrew J.S. Coats, DM, of the University of Warwick, Coventry, England.
“Similarly, the between-group differences seen in 6-minute walk distance (34 m) and gait speed (0.12 m/s) are clinically meaningful and sizable.”
They propose that some of the substantial quality-of-life benefit in the intervention group “may be due to better physical performance, and that part may be due to improvements in psychosocial factors and mood. It appears that exercise also resulted in patients becoming happier, or at least less depressed, as evidenced by the positive results on the Geriatric Depression Scale.”
Similar results across most subgroups
In subgroup analyses, the intervention was successful against the standard-care approach in both men and women at all ages and regardless of ejection fraction; symptom status; and whether the patient had diabetes, ischemic heart disease, or atrial fibrillation, or was obese.
Clinical outcomes were not significantly different at 6 months. The rate of death from any cause was 13% for the intervention group and 10% for the control group. There were 194 and 213 hospitalizations from any cause, respectively.
Not included in the trial’s current publication but soon to be published, Dr. Kitzman said when interviewed, is a comparison of outcomes in patients with HFpEF and HFrEF. “We found at baseline that those with HFpEF had worse impairment in physical function, quality of life, and frailty. After the intervention, there appeared to be consistently larger improvements in all outcomes, including SPPB, 6-minute walk, qualify of life, and frailty, in HFpEF versus HFrEF.”
The signals of potential benefit in HFpEF extended to clinical endpoints, he said. In contrast to similar rates of all-cause rehospitalization in HFrEF, “in patients with HFpEF, rehospitalizations were 17% lower in the intervention group, compared to the control group.” Still, he noted, the interaction P value wasn’t significant.
However, Dr. Kitzman added, mortality in the intervention group, compared with the control group, was reduced by 35% among patients with HFpEF, “but was 250% higher in HFrEF,” with a significant interaction P value.
He was careful to note that, as a phase 2 trial, REHAB-HF was underpowered for clinical events, “and even the results in the HFpEF group should not be seen as adequate evidence to change clinical care.” They were from an exploratory analysis that included relatively few events.
“Because definitive demonstration of improvement in clinical events is critical for altering clinical care guidelines and for third-party payer reimbursement decisions, we believe that a subsequent phase 3 trial is needed and are currently planning toward that,” Dr. Kitzman said.
The study was supported by research grants from the National Institutes of Health, the Kermit Glenn Phillips II Chair in Cardiovascular Medicine, and the Oristano Family Fund at Wake Forest. Dr. Kitzman disclosed receiving consulting fees or honoraria from AbbVie, AstraZeneca, Bayer Healthcare, Boehringer Ingelheim, CinRx, Corviamedical, GlaxoSmithKline, and Merck; and having an unspecified relationship with Gilead. Dr. Handberg disclosed receiving grants from Aastom Biosciences, Abbott Laboratories, Amgen, Amorcyte, AstraZeneca, Biocardia, Boehringer Ingelheim, Capricor, Cytori Therapeutics, Department of Defense, Direct Flow Medical, Everyfit, Gilead, Ionis, Medtronic, Merck, Mesoblast, Relypsa, and Sanofi-Aventis. Dr. Bittner discloses receiving consulting fees or honoraria from Pfizer and Sanofi; receiving research grants from Amgen and The Medicines Company; and having unspecified relationships with AstraZeneca, DalCor, Esperion, and Sanofi-Aventis. Dr. Anker reported receiving grants and personal fees from Abbott Vascular and Vifor; personal fees from Bayer, Boehringer Ingelheim, Novartis, Servier, Cardiac Dimensions, Thermo Fisher Scientific, AstraZeneca, Occlutech, Actimed, and Respicardia. Dr. Coats disclosed receiving personal fees from AstraZeneca, Bayer, Boehringer Ingelheim, Menarini, Novartis, Nutricia, Servier, Vifor, Abbott, Actimed, Arena, Cardiac Dimensions, Corvia, CVRx, Enopace, ESN Cleer, Faraday, WL Gore, Impulse Dynamics, and Respicardia.
A version of this article first appeared on Medscape.com.
A novel physical rehabilitation program for patients with advanced heart failure that aimed to improve their ability to exercise before focusing on endurance was successful in a randomized trial in ways that seem to have eluded some earlier exercise-training studies in the setting of HF.
The often-frail patients following the training regimen, initiated before discharge from hospitalization for acute decompensation, worked on capabilities such as mobility, balance, and strength deemed necessary if exercises meant to build exercise capacity were to succeed.
A huge percentage stayed with the 12-week program, which featured personalized, one-on-one training from a physical therapist. The patients benefited, with improvements in balance, walking ability, and strength, which were followed by significant gains in 6-minute walk distance (6MWD) and measures of physical functioning, frailty, and quality of life. The patients then continued elements of the program at home out to 6 months.
At that time, death and rehospitalizations did not differ between those assigned to the regimen and similar patients who had not participated in the program, although the trial wasn’t powered for clinical events.
The rehab strategy seemed to work across a wide range of patient subgroups. In particular, there was evidence that the benefits were more pronounced in patients with HF and preserved ejection fraction (HFpEF) than in those with HF and reduced ejection fraction (HFrEF), observed Dalane W. Kitzman, MD, Wake Forest University, Winston-Salem, N.C.
Dr. Kitzman presented results from the REHAB-HF (Rehabilitation Therapy in Older Acute Heart Failure Patients) trial at the annual scientific sessions of the American College of Cardiology and is lead author on its same-day publication in the New England Journal of Medicine.
An earlier pilot program unexpectedly showed that such patients recently hospitalized with HF “have significant impairments in mobility and balance,” he explained. If so, “it would be hazardous to subject them to traditional endurance training, such as walking-based treadmill or even bicycle.”
The unusual program, said Dr. Kitzman, looks to those issues before engaging the patients in endurance exercise by addressing mobility, balance, and basic strength – enough to repeatedly stand up from a sitting position, for example. “If you’re not able to stand with confidence, then you’re not able to walk on a treadmill.”
This model of exercise rehab “is used in geriatrics research, and enables them to safely increase endurance. It’s well known from geriatric studies that if you go directly to endurance in these, frail, older patients, you have little improvement and often have injuries and falls,” he added.
Guidance from telemedicine?
The functional outcomes examined in REHAB-HF “are the ones that matter to patients the most,” observed Eileen M. Handberg, PhD, of Shands Hospital at the University of Florida, Gainesville, at a presentation on the trial for the media.
“This is about being able to get out of a chair without assistance, not falling, walking farther, and feeling better as opposed to the more traditional outcome measure that has been used in cardiac rehab trials, which has been the exercise treadmill test – which most patients don’t have the capacity to do very well anyway,” said Dr. Handberg, who is not a part of REHAB-HF.
“This opens up rehab, potentially, to the more sick, who also need a better quality of life,” she said.
However, many patients invited to participate in the trial could not because they lived too far from the program, Dr. Handberg observed. “It would be nice to see if the lessons from COVID-19 might apply to this population” by making participation possible remotely, “perhaps using family members as rehab assistance,” she said.
“I was really very impressed that you had 83% adherence to a home exercise 6 months down the road, which far eclipses what we had in HF-ACTION,” said Vera Bittner, MD, University of Alabama at Birmingham, as the invited discussant following Dr. Kitzman’s formal presentation of the trial. “And it certainly eclipses what we see in the typical cardiac rehab program.”
Both Dr. Bittner and Dr. Kitzman participated in HF-ACTION, a randomized exercise-training trial for patients with chronic, stable HFrEF who were all-around less sick than those in REHAB-HF.
Four functional domains
Historically, HF exercise or rehab trials have excluded patients hospitalized with acute decompensation, and third-party reimbursement often has not covered such programs because of a lack of supporting evidence and a supposed potential for harm, Dr. Kitzman said.
Entry to REHAB-HF required the patients to be fit enough to walk 4 meters, with or without a walker or other assistant device, and to have been in the hospital for at least 24 hours with a primary diagnosis of acute decompensated HF.
The intervention relied on exercises aimed at improving the four functional domains of strength, balance, mobility, and – when those three were sufficiently developed – endurance, Dr. Kitzman and associates wrote in their published report.
“The intervention was initiated in the hospital when feasible and was subsequently transitioned to an outpatient facility as soon as possible after discharge,” they wrote. Afterward, “a key goal of the intervention during the first 3 months [the outpatient phase] was to prepare the patient to transition to the independent maintenance phase (months 4-6).”
The study’s control patients “received frequent calls from study staff to try to approximate the increased attention received by the intervention group,” Dr. Kitzman said in an interview. “They were allowed to receive all usual care as ordered by their treating physicians. This included, if ordered, standard physical therapy or cardiac rehabilitation” in 43% of the control cohort. Of the trial’s 349 patients, those assigned to the intervention scored significantly higher on the three-component Short Physical Performance Battery (SPPB) at 12 weeks than those assigned to a usual care approach that included, for some, more conventional cardiac rehabilitation (8.3 vs. 6.9; P < .001).
The SPPB, validated in trials as a proxy for clinical outcomes includes tests of balance while standing, gait speed during a 4-minute walk, and strength. The latter is the test that measures time needed to rise from a chair five times.
They also showed consistent gains in other measures of physical functioning and quality of life by 12 weeks months.
The observed SPPB treatment effect is “impressive” and “compares very favorably with previously reported estimates,” observed an accompanying editorial from Stefan D. Anker, MD, PhD, of the German Center for Cardiovascular Research and Charité Universitätsmedizin, Berlin, and Andrew J.S. Coats, DM, of the University of Warwick, Coventry, England.
“Similarly, the between-group differences seen in 6-minute walk distance (34 m) and gait speed (0.12 m/s) are clinically meaningful and sizable.”
They propose that some of the substantial quality-of-life benefit in the intervention group “may be due to better physical performance, and that part may be due to improvements in psychosocial factors and mood. It appears that exercise also resulted in patients becoming happier, or at least less depressed, as evidenced by the positive results on the Geriatric Depression Scale.”
Similar results across most subgroups
In subgroup analyses, the intervention was successful against the standard-care approach in both men and women at all ages and regardless of ejection fraction; symptom status; and whether the patient had diabetes, ischemic heart disease, or atrial fibrillation, or was obese.
Clinical outcomes were not significantly different at 6 months. The rate of death from any cause was 13% for the intervention group and 10% for the control group. There were 194 and 213 hospitalizations from any cause, respectively.
Not included in the trial’s current publication but soon to be published, Dr. Kitzman said when interviewed, is a comparison of outcomes in patients with HFpEF and HFrEF. “We found at baseline that those with HFpEF had worse impairment in physical function, quality of life, and frailty. After the intervention, there appeared to be consistently larger improvements in all outcomes, including SPPB, 6-minute walk, qualify of life, and frailty, in HFpEF versus HFrEF.”
The signals of potential benefit in HFpEF extended to clinical endpoints, he said. In contrast to similar rates of all-cause rehospitalization in HFrEF, “in patients with HFpEF, rehospitalizations were 17% lower in the intervention group, compared to the control group.” Still, he noted, the interaction P value wasn’t significant.
However, Dr. Kitzman added, mortality in the intervention group, compared with the control group, was reduced by 35% among patients with HFpEF, “but was 250% higher in HFrEF,” with a significant interaction P value.
He was careful to note that, as a phase 2 trial, REHAB-HF was underpowered for clinical events, “and even the results in the HFpEF group should not be seen as adequate evidence to change clinical care.” They were from an exploratory analysis that included relatively few events.
“Because definitive demonstration of improvement in clinical events is critical for altering clinical care guidelines and for third-party payer reimbursement decisions, we believe that a subsequent phase 3 trial is needed and are currently planning toward that,” Dr. Kitzman said.
The study was supported by research grants from the National Institutes of Health, the Kermit Glenn Phillips II Chair in Cardiovascular Medicine, and the Oristano Family Fund at Wake Forest. Dr. Kitzman disclosed receiving consulting fees or honoraria from AbbVie, AstraZeneca, Bayer Healthcare, Boehringer Ingelheim, CinRx, Corviamedical, GlaxoSmithKline, and Merck; and having an unspecified relationship with Gilead. Dr. Handberg disclosed receiving grants from Aastom Biosciences, Abbott Laboratories, Amgen, Amorcyte, AstraZeneca, Biocardia, Boehringer Ingelheim, Capricor, Cytori Therapeutics, Department of Defense, Direct Flow Medical, Everyfit, Gilead, Ionis, Medtronic, Merck, Mesoblast, Relypsa, and Sanofi-Aventis. Dr. Bittner discloses receiving consulting fees or honoraria from Pfizer and Sanofi; receiving research grants from Amgen and The Medicines Company; and having unspecified relationships with AstraZeneca, DalCor, Esperion, and Sanofi-Aventis. Dr. Anker reported receiving grants and personal fees from Abbott Vascular and Vifor; personal fees from Bayer, Boehringer Ingelheim, Novartis, Servier, Cardiac Dimensions, Thermo Fisher Scientific, AstraZeneca, Occlutech, Actimed, and Respicardia. Dr. Coats disclosed receiving personal fees from AstraZeneca, Bayer, Boehringer Ingelheim, Menarini, Novartis, Nutricia, Servier, Vifor, Abbott, Actimed, Arena, Cardiac Dimensions, Corvia, CVRx, Enopace, ESN Cleer, Faraday, WL Gore, Impulse Dynamics, and Respicardia.
A version of this article first appeared on Medscape.com.
Nasal spray resurrected after showing clinical benefits for PSVT
Significant improvement in the control of symptoms related to paroxysmal supraventricular tachycardia (PSVT) is resurrecting etripamil as a self-administered nasal spray a year after it failed to meet the primary endpoint in a phase 3 trial, according to a new analysis from this same study presented at the annual scientific sessions of the American College of Cardiology.
In the phase 3 NODE-301 trial, presented at the 2020 Heart Rhythm Society annual meeting, etripamil did not show an advantage over placebo at 5 hours for achieving sinus rhythm. Nevertheless, a new presentation of the secondary outcomes suggests substantial clinical benefit.
These advantages include significant reductions in PSVT symptoms, a trend for fewer emergency room visits, and a degree of patient satisfaction that appears meaningful, according to Bruce S. Stambler, MD, an electrophysiologist affiliated with Piedmont Heart Institute, Atlanta.
The data, despite the phase 3 trial results, “support continued development of etripamil nasal spray acute treatment of PSVT,” Dr. Stambler said.
Etripamil is an L-type calcium channel blocker. When administered by nasal spray, it reaches peak effects within about 10 minutes. But the action is short, with a decline in antiarrhythmia effects beginning about 30 minutes after the peak effect.
In the NODE-301 trial, which employed a 2:1 randomization ratio, 138 patients self-administered 70 mg of etripamil or placebo immediately upon experiencing a suspected episode of PSVT.
Up until 45 minutes, the proportion of episodes that converted to sinus rhythm was about 66% greater (hazard ratio, 1.66; P = .02) on etripamil than placebo, but the advantage was then lost. By predefined primary endpoint of 5 hours, when 100% of placebo patients but not all etripamil patients had converted, there was a slight but nonstatistical advantage for placebo (HR 1.08; P = .1212).
However, because of the rapid onset and then the rapid offset of this agent, the 5-hour time point for comparing effects might not have been the optimal duration to compare effects, according to Dr. Stambler.
On the basis of safety of etripamil, which was not associated with any significant adverse events in NODE-301, and the early clinical effect, the investigators have looked again at the data.
For relief of patient-reported symptoms and patient-reported satisfaction, which were secondary endpoints of the study, the data support a clinical role, according to this new analysis.
Specifically, there were large differences on a 7-point scale for all of the measured symptoms of PSVT in favor of etripamil, including rapid pulse (P = .002), palpitations (P = .0001), dizziness (P = 0.01), shortness of breath (P = 0.008), and anxiety (P = 0.006). A numerical advantage for chest pain did not reach significance.
“In general, patients reported scores of 4 to 5 on this scale, which corresponds to ‘not satisfied’ to ‘satisfied,’ while the placebo-treated patients reported scores of 2 to 3, which corresponds to ‘dissatisfied’ or ‘very dissatisfied,’ ” Dr. Stambler reported.
The favorable patient experience is also reflected in the Treatment Satisfaction with Questionnaire for Medication (TSQM-9), which was another NODE-301 endpoint. Evaluated when patients were still blinded to their assigned therapy, the advantage of etripamil over placebo for both global satisfaction (P = .007) and treatment effectiveness (P = .002) were also highly statistically significant.
The subjective experience of patients appeared to be reflected in objective measures. When the two groups were compared for interventions in an emergency room, the need was reduced by about half (12.1% vs. 24.5%; P = .051) among those treated with etripamil. Although this just missed the conventional measure of statistical significance, it was close. Similarly, patients randomized to etripamil required numerically fewer rescue medications (14.0% vs. 26.5%; P = .059).
Adenosine was the most common of the rescue medications, according to Dr. Stambler. He said there was no difference between the groups in use of rescue oral therapies.
When comparing etripamil and placebo in the subgroup that did visit an emergency room for PSVT, there was a delay in ER visits among those randomized to etripamil (116 vs. 79 minutes; P < 0.05), suggesting that this agent reduced the sense of urgency when PSVT symptoms develop, according to Dr. Stambler.
On average, the patients who enrolled in this trial had a PSVT history of about 1.5 years. In the year prior to enrollment, the mean number of ER visits was about nine.
In the trial design, patients were required to take a test dose of etripamil under observation by a physician before being sent home with their assigned therapy, but Dr. Stambler does not believe that the requirement, if the drug is approved, will be in the label.
Unexpectedly, many patients had symptom relief even without converting to sinus rhythm, Dr. Stambler acknowledged. He speculated that the reduction in heart rate associated with etripamil might have provided a relief of symptoms sufficient to relieve anxiety, producing the relative advantage for patient satisfaction.
Jodie L. Hurwitz, MD, director of the electrophysiology lab at Medical City Hospital, Dallas, indicated that there is a need for new options for PSVT. An expert panelist during the session where these data were presented, she was particularly interested in rapid symptom relief.
“It would be great to have a therapy that could be self-administered at home. Patients would like it, too,” she said.
Mary N. Walsh, MD, a heart failure specialist affiliated with Indiana University, Indianapolis, sees a potential role of a self-administered therapy like etripamil in conjunction with wearable devices. She noted that the proportion of patients using these devices to monitor arrhythmias is increasing, providing a role for an easily transportable therapy that could be used quickly when symptoms develop.
However, after the negative phase 3 trial, more data must now be collected to satisfy the regulatory authorities that this agent is safe and effective. Dr. Stambler said that the developer is now committed to pursue these studies.
Dr. Stambler has a financial relationship with Milestone Pharmaceuticals, which is developing etripamil nasal spray and was the sponsor of this trial. Dr. Walsh and Dr. Hurwitz have no potential relevant conflicts of interest.
Significant improvement in the control of symptoms related to paroxysmal supraventricular tachycardia (PSVT) is resurrecting etripamil as a self-administered nasal spray a year after it failed to meet the primary endpoint in a phase 3 trial, according to a new analysis from this same study presented at the annual scientific sessions of the American College of Cardiology.
In the phase 3 NODE-301 trial, presented at the 2020 Heart Rhythm Society annual meeting, etripamil did not show an advantage over placebo at 5 hours for achieving sinus rhythm. Nevertheless, a new presentation of the secondary outcomes suggests substantial clinical benefit.
These advantages include significant reductions in PSVT symptoms, a trend for fewer emergency room visits, and a degree of patient satisfaction that appears meaningful, according to Bruce S. Stambler, MD, an electrophysiologist affiliated with Piedmont Heart Institute, Atlanta.
The data, despite the phase 3 trial results, “support continued development of etripamil nasal spray acute treatment of PSVT,” Dr. Stambler said.
Etripamil is an L-type calcium channel blocker. When administered by nasal spray, it reaches peak effects within about 10 minutes. But the action is short, with a decline in antiarrhythmia effects beginning about 30 minutes after the peak effect.
In the NODE-301 trial, which employed a 2:1 randomization ratio, 138 patients self-administered 70 mg of etripamil or placebo immediately upon experiencing a suspected episode of PSVT.
Up until 45 minutes, the proportion of episodes that converted to sinus rhythm was about 66% greater (hazard ratio, 1.66; P = .02) on etripamil than placebo, but the advantage was then lost. By predefined primary endpoint of 5 hours, when 100% of placebo patients but not all etripamil patients had converted, there was a slight but nonstatistical advantage for placebo (HR 1.08; P = .1212).
However, because of the rapid onset and then the rapid offset of this agent, the 5-hour time point for comparing effects might not have been the optimal duration to compare effects, according to Dr. Stambler.
On the basis of safety of etripamil, which was not associated with any significant adverse events in NODE-301, and the early clinical effect, the investigators have looked again at the data.
For relief of patient-reported symptoms and patient-reported satisfaction, which were secondary endpoints of the study, the data support a clinical role, according to this new analysis.
Specifically, there were large differences on a 7-point scale for all of the measured symptoms of PSVT in favor of etripamil, including rapid pulse (P = .002), palpitations (P = .0001), dizziness (P = 0.01), shortness of breath (P = 0.008), and anxiety (P = 0.006). A numerical advantage for chest pain did not reach significance.
“In general, patients reported scores of 4 to 5 on this scale, which corresponds to ‘not satisfied’ to ‘satisfied,’ while the placebo-treated patients reported scores of 2 to 3, which corresponds to ‘dissatisfied’ or ‘very dissatisfied,’ ” Dr. Stambler reported.
The favorable patient experience is also reflected in the Treatment Satisfaction with Questionnaire for Medication (TSQM-9), which was another NODE-301 endpoint. Evaluated when patients were still blinded to their assigned therapy, the advantage of etripamil over placebo for both global satisfaction (P = .007) and treatment effectiveness (P = .002) were also highly statistically significant.
The subjective experience of patients appeared to be reflected in objective measures. When the two groups were compared for interventions in an emergency room, the need was reduced by about half (12.1% vs. 24.5%; P = .051) among those treated with etripamil. Although this just missed the conventional measure of statistical significance, it was close. Similarly, patients randomized to etripamil required numerically fewer rescue medications (14.0% vs. 26.5%; P = .059).
Adenosine was the most common of the rescue medications, according to Dr. Stambler. He said there was no difference between the groups in use of rescue oral therapies.
When comparing etripamil and placebo in the subgroup that did visit an emergency room for PSVT, there was a delay in ER visits among those randomized to etripamil (116 vs. 79 minutes; P < 0.05), suggesting that this agent reduced the sense of urgency when PSVT symptoms develop, according to Dr. Stambler.
On average, the patients who enrolled in this trial had a PSVT history of about 1.5 years. In the year prior to enrollment, the mean number of ER visits was about nine.
In the trial design, patients were required to take a test dose of etripamil under observation by a physician before being sent home with their assigned therapy, but Dr. Stambler does not believe that the requirement, if the drug is approved, will be in the label.
Unexpectedly, many patients had symptom relief even without converting to sinus rhythm, Dr. Stambler acknowledged. He speculated that the reduction in heart rate associated with etripamil might have provided a relief of symptoms sufficient to relieve anxiety, producing the relative advantage for patient satisfaction.
Jodie L. Hurwitz, MD, director of the electrophysiology lab at Medical City Hospital, Dallas, indicated that there is a need for new options for PSVT. An expert panelist during the session where these data were presented, she was particularly interested in rapid symptom relief.
“It would be great to have a therapy that could be self-administered at home. Patients would like it, too,” she said.
Mary N. Walsh, MD, a heart failure specialist affiliated with Indiana University, Indianapolis, sees a potential role of a self-administered therapy like etripamil in conjunction with wearable devices. She noted that the proportion of patients using these devices to monitor arrhythmias is increasing, providing a role for an easily transportable therapy that could be used quickly when symptoms develop.
However, after the negative phase 3 trial, more data must now be collected to satisfy the regulatory authorities that this agent is safe and effective. Dr. Stambler said that the developer is now committed to pursue these studies.
Dr. Stambler has a financial relationship with Milestone Pharmaceuticals, which is developing etripamil nasal spray and was the sponsor of this trial. Dr. Walsh and Dr. Hurwitz have no potential relevant conflicts of interest.
Significant improvement in the control of symptoms related to paroxysmal supraventricular tachycardia (PSVT) is resurrecting etripamil as a self-administered nasal spray a year after it failed to meet the primary endpoint in a phase 3 trial, according to a new analysis from this same study presented at the annual scientific sessions of the American College of Cardiology.
In the phase 3 NODE-301 trial, presented at the 2020 Heart Rhythm Society annual meeting, etripamil did not show an advantage over placebo at 5 hours for achieving sinus rhythm. Nevertheless, a new presentation of the secondary outcomes suggests substantial clinical benefit.
These advantages include significant reductions in PSVT symptoms, a trend for fewer emergency room visits, and a degree of patient satisfaction that appears meaningful, according to Bruce S. Stambler, MD, an electrophysiologist affiliated with Piedmont Heart Institute, Atlanta.
The data, despite the phase 3 trial results, “support continued development of etripamil nasal spray acute treatment of PSVT,” Dr. Stambler said.
Etripamil is an L-type calcium channel blocker. When administered by nasal spray, it reaches peak effects within about 10 minutes. But the action is short, with a decline in antiarrhythmia effects beginning about 30 minutes after the peak effect.
In the NODE-301 trial, which employed a 2:1 randomization ratio, 138 patients self-administered 70 mg of etripamil or placebo immediately upon experiencing a suspected episode of PSVT.
Up until 45 minutes, the proportion of episodes that converted to sinus rhythm was about 66% greater (hazard ratio, 1.66; P = .02) on etripamil than placebo, but the advantage was then lost. By predefined primary endpoint of 5 hours, when 100% of placebo patients but not all etripamil patients had converted, there was a slight but nonstatistical advantage for placebo (HR 1.08; P = .1212).
However, because of the rapid onset and then the rapid offset of this agent, the 5-hour time point for comparing effects might not have been the optimal duration to compare effects, according to Dr. Stambler.
On the basis of safety of etripamil, which was not associated with any significant adverse events in NODE-301, and the early clinical effect, the investigators have looked again at the data.
For relief of patient-reported symptoms and patient-reported satisfaction, which were secondary endpoints of the study, the data support a clinical role, according to this new analysis.
Specifically, there were large differences on a 7-point scale for all of the measured symptoms of PSVT in favor of etripamil, including rapid pulse (P = .002), palpitations (P = .0001), dizziness (P = 0.01), shortness of breath (P = 0.008), and anxiety (P = 0.006). A numerical advantage for chest pain did not reach significance.
“In general, patients reported scores of 4 to 5 on this scale, which corresponds to ‘not satisfied’ to ‘satisfied,’ while the placebo-treated patients reported scores of 2 to 3, which corresponds to ‘dissatisfied’ or ‘very dissatisfied,’ ” Dr. Stambler reported.
The favorable patient experience is also reflected in the Treatment Satisfaction with Questionnaire for Medication (TSQM-9), which was another NODE-301 endpoint. Evaluated when patients were still blinded to their assigned therapy, the advantage of etripamil over placebo for both global satisfaction (P = .007) and treatment effectiveness (P = .002) were also highly statistically significant.
The subjective experience of patients appeared to be reflected in objective measures. When the two groups were compared for interventions in an emergency room, the need was reduced by about half (12.1% vs. 24.5%; P = .051) among those treated with etripamil. Although this just missed the conventional measure of statistical significance, it was close. Similarly, patients randomized to etripamil required numerically fewer rescue medications (14.0% vs. 26.5%; P = .059).
Adenosine was the most common of the rescue medications, according to Dr. Stambler. He said there was no difference between the groups in use of rescue oral therapies.
When comparing etripamil and placebo in the subgroup that did visit an emergency room for PSVT, there was a delay in ER visits among those randomized to etripamil (116 vs. 79 minutes; P < 0.05), suggesting that this agent reduced the sense of urgency when PSVT symptoms develop, according to Dr. Stambler.
On average, the patients who enrolled in this trial had a PSVT history of about 1.5 years. In the year prior to enrollment, the mean number of ER visits was about nine.
In the trial design, patients were required to take a test dose of etripamil under observation by a physician before being sent home with their assigned therapy, but Dr. Stambler does not believe that the requirement, if the drug is approved, will be in the label.
Unexpectedly, many patients had symptom relief even without converting to sinus rhythm, Dr. Stambler acknowledged. He speculated that the reduction in heart rate associated with etripamil might have provided a relief of symptoms sufficient to relieve anxiety, producing the relative advantage for patient satisfaction.
Jodie L. Hurwitz, MD, director of the electrophysiology lab at Medical City Hospital, Dallas, indicated that there is a need for new options for PSVT. An expert panelist during the session where these data were presented, she was particularly interested in rapid symptom relief.
“It would be great to have a therapy that could be self-administered at home. Patients would like it, too,” she said.
Mary N. Walsh, MD, a heart failure specialist affiliated with Indiana University, Indianapolis, sees a potential role of a self-administered therapy like etripamil in conjunction with wearable devices. She noted that the proportion of patients using these devices to monitor arrhythmias is increasing, providing a role for an easily transportable therapy that could be used quickly when symptoms develop.
However, after the negative phase 3 trial, more data must now be collected to satisfy the regulatory authorities that this agent is safe and effective. Dr. Stambler said that the developer is now committed to pursue these studies.
Dr. Stambler has a financial relationship with Milestone Pharmaceuticals, which is developing etripamil nasal spray and was the sponsor of this trial. Dr. Walsh and Dr. Hurwitz have no potential relevant conflicts of interest.
FROM ACC 2021
LAAOS III: Surgical LAA closure cuts AFib stroke risk by one third
Left atrial appendage occlusion performed at the time of other heart surgery reduces the risk for stroke by about one-third in high-risk patients with atrial fibrillation (AFib), according to results of the Left Atrial Appendage Occlusion Study III (LAAOS III).
At 3.8 years’ follow-up, the primary endpoint of ischemic stroke or systemic embolism occurred in 4.8% of patients randomly assigned to left atrial appendage occlusion (LAAO) and 7.0% of those with no occlusion. This translated into a 33% relative risk reduction (hazard ratio, 0.67; 95% confidence interval, 0.53-0.85; P = .001).
In a landmark analysis, the effect was present early on but was more pronounced after the first 30 days, reducing the relative risk by 42% (HR, 0.58; 95% CI, 0.42-0.80), the researchers report.
The reduction in ongoing stroke risk was on top of oral anticoagulation (OAC) and consistent across all subgroups, Richard Whitlock, MD, PhD, professor of surgery, McMaster University, Hamilton, Ont., reported in a late-breaking trial session at the annual scientific sessions of the American College of Cardiology.
The procedure was safe and added, on average, just 6 minutes to cardiopulmonary bypass time, according to the results, simultaneously published in the New England Journal of Medicine.
“Any patient who comes to the operating room who fits the profile of a LAAOS III patient – so has atrial fibrillation and an elevated stroke risk based on their CHA2DS2-VASc score – the appendage should come off,” he said in an interview.
Commenting during the formal discussion, panelist Michael J. Mack, MD, of Baylor Health Care System in Houston, said, “This is potentially a game-changing, practice-changing study” but asked if there are any patients who shouldn’t undergo LAAO, such as those with heart failure (HF).
Dr. Whitlock said about 10%-15% of patients coming for heart surgery have a history of AFib and “as surgeons, you do need to individualize therapy. If you have a very frail patient, have concerns about tissue quality, you really need to think about how you would occlude the left atrial appendage or if you would occlude.”
Reassuringly, he noted, the data show no increase in HF hospitalizations and a beneficial effect on stroke among patients with HF and those with low ejection fractions, below 50%.
Observational data on surgical occlusion have been inconsistent, and current guidelines offer a weak recommendation in patients with AFib who have a contraindication to long-term anticoagulation. This is the first study to definitively prove that ischemic stroke is reduced by managing the left atrial appendage, he said in an interview.
“The previous percutaneous trials failed to demonstrate that; they demonstrated noninferiority but it was driven primarily by the avoidance of hemorrhagic events or strokes through taking patients off oral anticoagulation,” he said.
The results should translate into a class I guideline recommendation, he added. “This opens up a new paradigm of treatment for atrial fibrillation and stroke prevention in that it is really the first study that has looked at the additive effects of managing the left atrial appendage in addition to oral anticoagulation, and it’s protective on top of oral anticoagulation. That is a paradigm shift.”
In an accompanying editorial, Richard L. Page, MD, University of Vermont in Burlington, said the trial provides no insight on the possible benefit of surgical occlusion in patients unable to receive anticoagulation or with a lower CHA2DS2-VASc score, but he agreed a class I recommendation is likely for the population studied.
“I hope and anticipate that the results of this paper will strengthen the guideline indications for surgical left atrial appendage occlusion and will increase the number of cardiac surgeons who routinely perform this add-on procedure,” he said. “While many already perform this procedure, cardiac surgeons should now feel more comfortable that surgical left atrial appendage occlusion is indicated and supported by high-quality randomized data.”
Unfortunately, LAAOS III does not answer the question of whether patients can come off anticoagulation, but it does show surgical occlusion provides added protection from strokes, which can be huge with atrial fibrillation, Dr. Whitlock said.
“I spoke with a patient today who is an active 66-year-old individual on a [direct oral anticoagulant], and his stroke risk has been further reduced by 30%-40%, so he was ecstatic to hear the results,” Dr. Whitlock said. “I think it’s peace of mind.”
Global, nonindustry effort
LAAOS III investigators at 105 centers in 27 countries enrolled 4,811 patients undergoing cardiac surgery (mean age, 71 years; 68% male) who had a CHA2DS2-VASc score of at least 2.
In all, 4,770 were randomly assigned to no LAAO or occlusion via the preferred technique of amputation with suture closure of the stump as well as stapler occlusion, or epicardial device closure with the AtriClip (AtriCure) or TigerPaw (Maquet Medical). The treating team, researchers, and patients were blinded to assignment.
Patients were followed every 6 months with a validated stroke questionnaire. The trial was stopped early by the data safety monitoring board after the second interim analysis.
The mean CHA2DS2-VASc score was 4.2, one-third of patients had permanent AFib, 9% had a history of stroke, and more than two-thirds underwent a valve procedure, which makes LAAOS III unique, as many previous trials excluded valvular AFib, Dr. Whitlock pointed out.
Operative outcomes in the LAAO and no-LAAO groups were as follows:
- Bypass time: mean, 119 minutes vs. 113 minutes.
- Cross-clamp time: mean, 86 minutes vs. 82 minutes.
- Chest tube output: median, 520 mL vs. 500 mL.
- Reoperation for bleeding: both, 4.0%.
- Prolonged hospitalization due to HF: 5 vs. 14 events.
- 30-day mortality: 3.7% vs 4.0%.
The primary safety outcome of HF hospitalization at 3.8 years occurred in 7.7% of patients with LAAO and 6.8% without occlusion (HR, 1.13; 95% CI, 0.92-1.40), despite concerns that taking off the appendage could worsen HF risk by impairing renal clearance of salt and water.
“There’s observational data on either side of the fence, so it was an important endpoint that people were concerned about,” Dr. Whitlock told this news organization. “We had a data collection firm dedicated to admission for heart failure to really tease that out and, in the end, we saw no adverse effect.”
Although rates of ischemic stroke at 3.8 years were lower with LAAO than without (4.2% vs. 6.6%; HR, 0.62; 95% CI, 0.48-0.80), there was no difference in systemic embolism (0.3% for both) or death (22.6% vs. 22.5%).
In LAAOS III, fewer than 2% of the deaths were attributed to stroke, which is consistent with large stroke registries, Dr. Whitlock said. “Stroke is not what causes people with atrial fibrillation to die; it’s actually the progression on to heart failure.”
The positive effect on stroke was consistent across all subgroups, including sex, age, rheumatic heart disease, type of OAC at baseline, CHA2DS2-VASc score (≤4 vs. >4), type of surgery, history of heart failure or hypertension, and prior stroke/transient ischemic attack/systemic embolism.
Panelist Anne B. Curtis, MD, State University of New York at Buffalo, expressed surprise that about half of patients at baseline were not receiving anticoagulation and questioned whether event rates varied among those who did and didn’t stay on OAC.
Dr. Whitlock noted that OAC is often underused in AFib and that analyses showed the effects were consistent whether patients were on or off anticoagulants.
The study was sponsored by the Population Health Research Institute, McMaster University. Dr. Whitlock reported no relevant disclosures. Dr. Curtis reported consultant fees/honoraria from Abbott, Janssen, Medtronic, Milestone Pharmaceuticals, and Sanofi Aventis, and data safety monitoring board participation for Medtronic.
A version of this article first appeared on Medscape.com.
Left atrial appendage occlusion performed at the time of other heart surgery reduces the risk for stroke by about one-third in high-risk patients with atrial fibrillation (AFib), according to results of the Left Atrial Appendage Occlusion Study III (LAAOS III).
At 3.8 years’ follow-up, the primary endpoint of ischemic stroke or systemic embolism occurred in 4.8% of patients randomly assigned to left atrial appendage occlusion (LAAO) and 7.0% of those with no occlusion. This translated into a 33% relative risk reduction (hazard ratio, 0.67; 95% confidence interval, 0.53-0.85; P = .001).
In a landmark analysis, the effect was present early on but was more pronounced after the first 30 days, reducing the relative risk by 42% (HR, 0.58; 95% CI, 0.42-0.80), the researchers report.
The reduction in ongoing stroke risk was on top of oral anticoagulation (OAC) and consistent across all subgroups, Richard Whitlock, MD, PhD, professor of surgery, McMaster University, Hamilton, Ont., reported in a late-breaking trial session at the annual scientific sessions of the American College of Cardiology.
The procedure was safe and added, on average, just 6 minutes to cardiopulmonary bypass time, according to the results, simultaneously published in the New England Journal of Medicine.
“Any patient who comes to the operating room who fits the profile of a LAAOS III patient – so has atrial fibrillation and an elevated stroke risk based on their CHA2DS2-VASc score – the appendage should come off,” he said in an interview.
Commenting during the formal discussion, panelist Michael J. Mack, MD, of Baylor Health Care System in Houston, said, “This is potentially a game-changing, practice-changing study” but asked if there are any patients who shouldn’t undergo LAAO, such as those with heart failure (HF).
Dr. Whitlock said about 10%-15% of patients coming for heart surgery have a history of AFib and “as surgeons, you do need to individualize therapy. If you have a very frail patient, have concerns about tissue quality, you really need to think about how you would occlude the left atrial appendage or if you would occlude.”
Reassuringly, he noted, the data show no increase in HF hospitalizations and a beneficial effect on stroke among patients with HF and those with low ejection fractions, below 50%.
Observational data on surgical occlusion have been inconsistent, and current guidelines offer a weak recommendation in patients with AFib who have a contraindication to long-term anticoagulation. This is the first study to definitively prove that ischemic stroke is reduced by managing the left atrial appendage, he said in an interview.
“The previous percutaneous trials failed to demonstrate that; they demonstrated noninferiority but it was driven primarily by the avoidance of hemorrhagic events or strokes through taking patients off oral anticoagulation,” he said.
The results should translate into a class I guideline recommendation, he added. “This opens up a new paradigm of treatment for atrial fibrillation and stroke prevention in that it is really the first study that has looked at the additive effects of managing the left atrial appendage in addition to oral anticoagulation, and it’s protective on top of oral anticoagulation. That is a paradigm shift.”
In an accompanying editorial, Richard L. Page, MD, University of Vermont in Burlington, said the trial provides no insight on the possible benefit of surgical occlusion in patients unable to receive anticoagulation or with a lower CHA2DS2-VASc score, but he agreed a class I recommendation is likely for the population studied.
“I hope and anticipate that the results of this paper will strengthen the guideline indications for surgical left atrial appendage occlusion and will increase the number of cardiac surgeons who routinely perform this add-on procedure,” he said. “While many already perform this procedure, cardiac surgeons should now feel more comfortable that surgical left atrial appendage occlusion is indicated and supported by high-quality randomized data.”
Unfortunately, LAAOS III does not answer the question of whether patients can come off anticoagulation, but it does show surgical occlusion provides added protection from strokes, which can be huge with atrial fibrillation, Dr. Whitlock said.
“I spoke with a patient today who is an active 66-year-old individual on a [direct oral anticoagulant], and his stroke risk has been further reduced by 30%-40%, so he was ecstatic to hear the results,” Dr. Whitlock said. “I think it’s peace of mind.”
Global, nonindustry effort
LAAOS III investigators at 105 centers in 27 countries enrolled 4,811 patients undergoing cardiac surgery (mean age, 71 years; 68% male) who had a CHA2DS2-VASc score of at least 2.
In all, 4,770 were randomly assigned to no LAAO or occlusion via the preferred technique of amputation with suture closure of the stump as well as stapler occlusion, or epicardial device closure with the AtriClip (AtriCure) or TigerPaw (Maquet Medical). The treating team, researchers, and patients were blinded to assignment.
Patients were followed every 6 months with a validated stroke questionnaire. The trial was stopped early by the data safety monitoring board after the second interim analysis.
The mean CHA2DS2-VASc score was 4.2, one-third of patients had permanent AFib, 9% had a history of stroke, and more than two-thirds underwent a valve procedure, which makes LAAOS III unique, as many previous trials excluded valvular AFib, Dr. Whitlock pointed out.
Operative outcomes in the LAAO and no-LAAO groups were as follows:
- Bypass time: mean, 119 minutes vs. 113 minutes.
- Cross-clamp time: mean, 86 minutes vs. 82 minutes.
- Chest tube output: median, 520 mL vs. 500 mL.
- Reoperation for bleeding: both, 4.0%.
- Prolonged hospitalization due to HF: 5 vs. 14 events.
- 30-day mortality: 3.7% vs 4.0%.
The primary safety outcome of HF hospitalization at 3.8 years occurred in 7.7% of patients with LAAO and 6.8% without occlusion (HR, 1.13; 95% CI, 0.92-1.40), despite concerns that taking off the appendage could worsen HF risk by impairing renal clearance of salt and water.
“There’s observational data on either side of the fence, so it was an important endpoint that people were concerned about,” Dr. Whitlock told this news organization. “We had a data collection firm dedicated to admission for heart failure to really tease that out and, in the end, we saw no adverse effect.”
Although rates of ischemic stroke at 3.8 years were lower with LAAO than without (4.2% vs. 6.6%; HR, 0.62; 95% CI, 0.48-0.80), there was no difference in systemic embolism (0.3% for both) or death (22.6% vs. 22.5%).
In LAAOS III, fewer than 2% of the deaths were attributed to stroke, which is consistent with large stroke registries, Dr. Whitlock said. “Stroke is not what causes people with atrial fibrillation to die; it’s actually the progression on to heart failure.”
The positive effect on stroke was consistent across all subgroups, including sex, age, rheumatic heart disease, type of OAC at baseline, CHA2DS2-VASc score (≤4 vs. >4), type of surgery, history of heart failure or hypertension, and prior stroke/transient ischemic attack/systemic embolism.
Panelist Anne B. Curtis, MD, State University of New York at Buffalo, expressed surprise that about half of patients at baseline were not receiving anticoagulation and questioned whether event rates varied among those who did and didn’t stay on OAC.
Dr. Whitlock noted that OAC is often underused in AFib and that analyses showed the effects were consistent whether patients were on or off anticoagulants.
The study was sponsored by the Population Health Research Institute, McMaster University. Dr. Whitlock reported no relevant disclosures. Dr. Curtis reported consultant fees/honoraria from Abbott, Janssen, Medtronic, Milestone Pharmaceuticals, and Sanofi Aventis, and data safety monitoring board participation for Medtronic.
A version of this article first appeared on Medscape.com.
Left atrial appendage occlusion performed at the time of other heart surgery reduces the risk for stroke by about one-third in high-risk patients with atrial fibrillation (AFib), according to results of the Left Atrial Appendage Occlusion Study III (LAAOS III).
At 3.8 years’ follow-up, the primary endpoint of ischemic stroke or systemic embolism occurred in 4.8% of patients randomly assigned to left atrial appendage occlusion (LAAO) and 7.0% of those with no occlusion. This translated into a 33% relative risk reduction (hazard ratio, 0.67; 95% confidence interval, 0.53-0.85; P = .001).
In a landmark analysis, the effect was present early on but was more pronounced after the first 30 days, reducing the relative risk by 42% (HR, 0.58; 95% CI, 0.42-0.80), the researchers report.
The reduction in ongoing stroke risk was on top of oral anticoagulation (OAC) and consistent across all subgroups, Richard Whitlock, MD, PhD, professor of surgery, McMaster University, Hamilton, Ont., reported in a late-breaking trial session at the annual scientific sessions of the American College of Cardiology.
The procedure was safe and added, on average, just 6 minutes to cardiopulmonary bypass time, according to the results, simultaneously published in the New England Journal of Medicine.
“Any patient who comes to the operating room who fits the profile of a LAAOS III patient – so has atrial fibrillation and an elevated stroke risk based on their CHA2DS2-VASc score – the appendage should come off,” he said in an interview.
Commenting during the formal discussion, panelist Michael J. Mack, MD, of Baylor Health Care System in Houston, said, “This is potentially a game-changing, practice-changing study” but asked if there are any patients who shouldn’t undergo LAAO, such as those with heart failure (HF).
Dr. Whitlock said about 10%-15% of patients coming for heart surgery have a history of AFib and “as surgeons, you do need to individualize therapy. If you have a very frail patient, have concerns about tissue quality, you really need to think about how you would occlude the left atrial appendage or if you would occlude.”
Reassuringly, he noted, the data show no increase in HF hospitalizations and a beneficial effect on stroke among patients with HF and those with low ejection fractions, below 50%.
Observational data on surgical occlusion have been inconsistent, and current guidelines offer a weak recommendation in patients with AFib who have a contraindication to long-term anticoagulation. This is the first study to definitively prove that ischemic stroke is reduced by managing the left atrial appendage, he said in an interview.
“The previous percutaneous trials failed to demonstrate that; they demonstrated noninferiority but it was driven primarily by the avoidance of hemorrhagic events or strokes through taking patients off oral anticoagulation,” he said.
The results should translate into a class I guideline recommendation, he added. “This opens up a new paradigm of treatment for atrial fibrillation and stroke prevention in that it is really the first study that has looked at the additive effects of managing the left atrial appendage in addition to oral anticoagulation, and it’s protective on top of oral anticoagulation. That is a paradigm shift.”
In an accompanying editorial, Richard L. Page, MD, University of Vermont in Burlington, said the trial provides no insight on the possible benefit of surgical occlusion in patients unable to receive anticoagulation or with a lower CHA2DS2-VASc score, but he agreed a class I recommendation is likely for the population studied.
“I hope and anticipate that the results of this paper will strengthen the guideline indications for surgical left atrial appendage occlusion and will increase the number of cardiac surgeons who routinely perform this add-on procedure,” he said. “While many already perform this procedure, cardiac surgeons should now feel more comfortable that surgical left atrial appendage occlusion is indicated and supported by high-quality randomized data.”
Unfortunately, LAAOS III does not answer the question of whether patients can come off anticoagulation, but it does show surgical occlusion provides added protection from strokes, which can be huge with atrial fibrillation, Dr. Whitlock said.
“I spoke with a patient today who is an active 66-year-old individual on a [direct oral anticoagulant], and his stroke risk has been further reduced by 30%-40%, so he was ecstatic to hear the results,” Dr. Whitlock said. “I think it’s peace of mind.”
Global, nonindustry effort
LAAOS III investigators at 105 centers in 27 countries enrolled 4,811 patients undergoing cardiac surgery (mean age, 71 years; 68% male) who had a CHA2DS2-VASc score of at least 2.
In all, 4,770 were randomly assigned to no LAAO or occlusion via the preferred technique of amputation with suture closure of the stump as well as stapler occlusion, or epicardial device closure with the AtriClip (AtriCure) or TigerPaw (Maquet Medical). The treating team, researchers, and patients were blinded to assignment.
Patients were followed every 6 months with a validated stroke questionnaire. The trial was stopped early by the data safety monitoring board after the second interim analysis.
The mean CHA2DS2-VASc score was 4.2, one-third of patients had permanent AFib, 9% had a history of stroke, and more than two-thirds underwent a valve procedure, which makes LAAOS III unique, as many previous trials excluded valvular AFib, Dr. Whitlock pointed out.
Operative outcomes in the LAAO and no-LAAO groups were as follows:
- Bypass time: mean, 119 minutes vs. 113 minutes.
- Cross-clamp time: mean, 86 minutes vs. 82 minutes.
- Chest tube output: median, 520 mL vs. 500 mL.
- Reoperation for bleeding: both, 4.0%.
- Prolonged hospitalization due to HF: 5 vs. 14 events.
- 30-day mortality: 3.7% vs 4.0%.
The primary safety outcome of HF hospitalization at 3.8 years occurred in 7.7% of patients with LAAO and 6.8% without occlusion (HR, 1.13; 95% CI, 0.92-1.40), despite concerns that taking off the appendage could worsen HF risk by impairing renal clearance of salt and water.
“There’s observational data on either side of the fence, so it was an important endpoint that people were concerned about,” Dr. Whitlock told this news organization. “We had a data collection firm dedicated to admission for heart failure to really tease that out and, in the end, we saw no adverse effect.”
Although rates of ischemic stroke at 3.8 years were lower with LAAO than without (4.2% vs. 6.6%; HR, 0.62; 95% CI, 0.48-0.80), there was no difference in systemic embolism (0.3% for both) or death (22.6% vs. 22.5%).
In LAAOS III, fewer than 2% of the deaths were attributed to stroke, which is consistent with large stroke registries, Dr. Whitlock said. “Stroke is not what causes people with atrial fibrillation to die; it’s actually the progression on to heart failure.”
The positive effect on stroke was consistent across all subgroups, including sex, age, rheumatic heart disease, type of OAC at baseline, CHA2DS2-VASc score (≤4 vs. >4), type of surgery, history of heart failure or hypertension, and prior stroke/transient ischemic attack/systemic embolism.
Panelist Anne B. Curtis, MD, State University of New York at Buffalo, expressed surprise that about half of patients at baseline were not receiving anticoagulation and questioned whether event rates varied among those who did and didn’t stay on OAC.
Dr. Whitlock noted that OAC is often underused in AFib and that analyses showed the effects were consistent whether patients were on or off anticoagulants.
The study was sponsored by the Population Health Research Institute, McMaster University. Dr. Whitlock reported no relevant disclosures. Dr. Curtis reported consultant fees/honoraria from Abbott, Janssen, Medtronic, Milestone Pharmaceuticals, and Sanofi Aventis, and data safety monitoring board participation for Medtronic.
A version of this article first appeared on Medscape.com.
FROM ACC 2021
PARADISE-MI: Sacubitril/valsartan can’t beat ramipril in patients with acute MI
Treatment with sacubitril/valsartan, a pillar of therapy for patients with chronic heart failure with below-normal ejection fraction, came suggestively close to showing efficacy for preventing cardiovascular death or heart failure events in patients who have just had an MI but have no history of heart failure in a controlled trial with more than 5,600 patients.
Although sacubitril/valsartan (Entresto) fell short of producing a significant benefit, it did show good safety that was similar to the study’s comparator treatment, ramipril, an agent from the angiotensin-converting enzyme inhibitor class that is a mainstay of treatment in these patients.
“To say that, with no run-in, sacubitril/valsartan is as well tolerated and as safe as one of the best-studied ACE inhibitors – ramipril – in acutely ill MI patients, is a big statement,” said Marc A. Pfeffer, MD, at the annual scientific sessions of the American College of Cardiology. This high level of safety without gradual uptitration of sacubitril/valsartan (Entresto) “should lower barriers” to broader use of the dual-drug formulation for its approved indication in patients with chronic heart failure, especially patients with a left ventricular ejection fraction that is below normal. In addition, results from the PARADISE-MI trial suggested that “patients seemed to benefit before they develop heart failure. We couldn’t prove that, but we should build on this, and make it easier for patients to use this treatment,” Dr. Pfeffer said during a press briefing following his talk at the sessions.
Preventing heart failures to come
Treatment with sacubitril/valsartan in acute MI patients within a few days of their event “is perhaps addressing prevention of the heart failure that’s to come,” commented Lynne W. Stevenson, MD, designated discussant for the report and professor of medicine at Vanderbilt University Medical Center in Nashville. “Patients who are destined to develop heart failure are beginning their treatment early. The subgroup analyses suggest that it’s the sicker patients who benefited the most,” she said.
But Dr. Pfeffer stressed that “I don’t think this is a subgroup discussion. I would like to pursue this, but that’s up to the sponsor,” Novartis, the company that markets sacubitril/valsartan.
‘Exceedingly reassuring’ safety
The safety data that Dr. Pfeffer reported “are exceedingly reassuring. We didn’t see a signal of harm, and in some of the exploratory endpoints there was some evidence of benefit, so we need to encourage you to continue,” commented Mary N. Walsh, MD, medical director of the heart failure and cardiac transplantation program at Ascension St. Vincent Heart Center of Indiana in Indianapolis.
The PARADISE-MI (Prospective ARNI vs. ACE Inhibitor Trial to Determine Superiority in Reducing Heart Failure Events After MI) trial enrolled 5,669 patients with no history of heart failure within an average of 4 days following an acute MI at 495 sites in 41 countries during 2016-2020, with 8% of enrolled patients from the United States. Patients averaged 64 years of age, about three-quarters were men, about 43% had a history of diabetes, and only 1% were Black; Dr. Pfeffer noted that this is because most patients came from countries with low Black populations. The enrollment criteria required a left ventricular ejection fraction no greater than 40%, and among the enrolled patients this averaged about 37%.
A 10% nonsignificant relative risk reduction for the primary endpoint
The study’s primary endpoint was the combined first-event rate of cardiovascular death, hospitalization for heart failure, or an outpatient visit for heart failure. During a median follow-up of 23 months, this occurred at a rate of 7.4/100 patient years in the ramipril arm and 6.7/100 patient years in the sacubitril/valsartan arm, a 10% relative risk reduction with sacubitril/valsartan that was not significant, which meant all other efficacy analyses were exploratory, Dr. Pfeffer stressed.
Several secondary efficacy analyses showed significant benefits from sacubitril/valsartan, compared with ramipril, including the total number of events that comprised the primary endpoint, with a 21% relative risk reduction associated with sacubitril/valsartan, as well as investigator-reported events. The primary-endpoint benefit from sacubitril/valsartan was also significant in two subgroup analyses: patients aged 65 years or older (roughly half the study cohort), who had a 24% relative risk reduction on sacubitril/valsartan, compared with ramipril, and the 88% of patients who received treatment with percutaneous coronary intervention for their acute MI, who had a 19% relative risk reduction on sacubitril/valsartan, compared with patients who received ramipril.
The study’s safety data showed nearly identical rates in the two treatment arms for total adverse events, serious adverse events, adverse events that led to stopping the study drug, as well as in laboratory measures. The biggest between-treatment differences were a modest excess of hypotension on sacubitril valsartan, 28%, compared with 22% on ramipril, and a modest excess rate of cough on ramipril, 13%, compared with 9% on sacubitril/valsartan.
The added insight the results provide about sacubitril/valsartan comes at a time when U.S. patients continue to struggle to get health insurance coverage for an agent that has been approved for U.S. use in treating heart failure since 2015.
“Our patients do not have access to this important treatment,” declared Dr. Walsh during the press briefing. “The prior authorization process is unbelievable, and some patients have no access unless they pay the full cost on their own. This is an important, real-world problem that we face with this drug.”
PARADISE-MI was sponsored by Novartis, the company that markets sacubitril/valsartan (Entresto). Dr. Pfeffer has received research funding from and is a consultant to Novartis. He is also a consultant to AstraZeneca, Boehringer Ingelheim, Corvidia, DalCor, Eli Lilly, GlaxoSmithKline, Novo Nordisk, Peerbridge, and Sanofi, and he holds equity in DalCor and Peerbridge. Dr. Stevenson has received honoraria from LivaNova and has received research support from Abbott. Dr. Walsh had no disclosures.
Treatment with sacubitril/valsartan, a pillar of therapy for patients with chronic heart failure with below-normal ejection fraction, came suggestively close to showing efficacy for preventing cardiovascular death or heart failure events in patients who have just had an MI but have no history of heart failure in a controlled trial with more than 5,600 patients.
Although sacubitril/valsartan (Entresto) fell short of producing a significant benefit, it did show good safety that was similar to the study’s comparator treatment, ramipril, an agent from the angiotensin-converting enzyme inhibitor class that is a mainstay of treatment in these patients.
“To say that, with no run-in, sacubitril/valsartan is as well tolerated and as safe as one of the best-studied ACE inhibitors – ramipril – in acutely ill MI patients, is a big statement,” said Marc A. Pfeffer, MD, at the annual scientific sessions of the American College of Cardiology. This high level of safety without gradual uptitration of sacubitril/valsartan (Entresto) “should lower barriers” to broader use of the dual-drug formulation for its approved indication in patients with chronic heart failure, especially patients with a left ventricular ejection fraction that is below normal. In addition, results from the PARADISE-MI trial suggested that “patients seemed to benefit before they develop heart failure. We couldn’t prove that, but we should build on this, and make it easier for patients to use this treatment,” Dr. Pfeffer said during a press briefing following his talk at the sessions.
Preventing heart failures to come
Treatment with sacubitril/valsartan in acute MI patients within a few days of their event “is perhaps addressing prevention of the heart failure that’s to come,” commented Lynne W. Stevenson, MD, designated discussant for the report and professor of medicine at Vanderbilt University Medical Center in Nashville. “Patients who are destined to develop heart failure are beginning their treatment early. The subgroup analyses suggest that it’s the sicker patients who benefited the most,” she said.
But Dr. Pfeffer stressed that “I don’t think this is a subgroup discussion. I would like to pursue this, but that’s up to the sponsor,” Novartis, the company that markets sacubitril/valsartan.
‘Exceedingly reassuring’ safety
The safety data that Dr. Pfeffer reported “are exceedingly reassuring. We didn’t see a signal of harm, and in some of the exploratory endpoints there was some evidence of benefit, so we need to encourage you to continue,” commented Mary N. Walsh, MD, medical director of the heart failure and cardiac transplantation program at Ascension St. Vincent Heart Center of Indiana in Indianapolis.
The PARADISE-MI (Prospective ARNI vs. ACE Inhibitor Trial to Determine Superiority in Reducing Heart Failure Events After MI) trial enrolled 5,669 patients with no history of heart failure within an average of 4 days following an acute MI at 495 sites in 41 countries during 2016-2020, with 8% of enrolled patients from the United States. Patients averaged 64 years of age, about three-quarters were men, about 43% had a history of diabetes, and only 1% were Black; Dr. Pfeffer noted that this is because most patients came from countries with low Black populations. The enrollment criteria required a left ventricular ejection fraction no greater than 40%, and among the enrolled patients this averaged about 37%.
A 10% nonsignificant relative risk reduction for the primary endpoint
The study’s primary endpoint was the combined first-event rate of cardiovascular death, hospitalization for heart failure, or an outpatient visit for heart failure. During a median follow-up of 23 months, this occurred at a rate of 7.4/100 patient years in the ramipril arm and 6.7/100 patient years in the sacubitril/valsartan arm, a 10% relative risk reduction with sacubitril/valsartan that was not significant, which meant all other efficacy analyses were exploratory, Dr. Pfeffer stressed.
Several secondary efficacy analyses showed significant benefits from sacubitril/valsartan, compared with ramipril, including the total number of events that comprised the primary endpoint, with a 21% relative risk reduction associated with sacubitril/valsartan, as well as investigator-reported events. The primary-endpoint benefit from sacubitril/valsartan was also significant in two subgroup analyses: patients aged 65 years or older (roughly half the study cohort), who had a 24% relative risk reduction on sacubitril/valsartan, compared with ramipril, and the 88% of patients who received treatment with percutaneous coronary intervention for their acute MI, who had a 19% relative risk reduction on sacubitril/valsartan, compared with patients who received ramipril.
The study’s safety data showed nearly identical rates in the two treatment arms for total adverse events, serious adverse events, adverse events that led to stopping the study drug, as well as in laboratory measures. The biggest between-treatment differences were a modest excess of hypotension on sacubitril valsartan, 28%, compared with 22% on ramipril, and a modest excess rate of cough on ramipril, 13%, compared with 9% on sacubitril/valsartan.
The added insight the results provide about sacubitril/valsartan comes at a time when U.S. patients continue to struggle to get health insurance coverage for an agent that has been approved for U.S. use in treating heart failure since 2015.
“Our patients do not have access to this important treatment,” declared Dr. Walsh during the press briefing. “The prior authorization process is unbelievable, and some patients have no access unless they pay the full cost on their own. This is an important, real-world problem that we face with this drug.”
PARADISE-MI was sponsored by Novartis, the company that markets sacubitril/valsartan (Entresto). Dr. Pfeffer has received research funding from and is a consultant to Novartis. He is also a consultant to AstraZeneca, Boehringer Ingelheim, Corvidia, DalCor, Eli Lilly, GlaxoSmithKline, Novo Nordisk, Peerbridge, and Sanofi, and he holds equity in DalCor and Peerbridge. Dr. Stevenson has received honoraria from LivaNova and has received research support from Abbott. Dr. Walsh had no disclosures.
Treatment with sacubitril/valsartan, a pillar of therapy for patients with chronic heart failure with below-normal ejection fraction, came suggestively close to showing efficacy for preventing cardiovascular death or heart failure events in patients who have just had an MI but have no history of heart failure in a controlled trial with more than 5,600 patients.
Although sacubitril/valsartan (Entresto) fell short of producing a significant benefit, it did show good safety that was similar to the study’s comparator treatment, ramipril, an agent from the angiotensin-converting enzyme inhibitor class that is a mainstay of treatment in these patients.
“To say that, with no run-in, sacubitril/valsartan is as well tolerated and as safe as one of the best-studied ACE inhibitors – ramipril – in acutely ill MI patients, is a big statement,” said Marc A. Pfeffer, MD, at the annual scientific sessions of the American College of Cardiology. This high level of safety without gradual uptitration of sacubitril/valsartan (Entresto) “should lower barriers” to broader use of the dual-drug formulation for its approved indication in patients with chronic heart failure, especially patients with a left ventricular ejection fraction that is below normal. In addition, results from the PARADISE-MI trial suggested that “patients seemed to benefit before they develop heart failure. We couldn’t prove that, but we should build on this, and make it easier for patients to use this treatment,” Dr. Pfeffer said during a press briefing following his talk at the sessions.
Preventing heart failures to come
Treatment with sacubitril/valsartan in acute MI patients within a few days of their event “is perhaps addressing prevention of the heart failure that’s to come,” commented Lynne W. Stevenson, MD, designated discussant for the report and professor of medicine at Vanderbilt University Medical Center in Nashville. “Patients who are destined to develop heart failure are beginning their treatment early. The subgroup analyses suggest that it’s the sicker patients who benefited the most,” she said.
But Dr. Pfeffer stressed that “I don’t think this is a subgroup discussion. I would like to pursue this, but that’s up to the sponsor,” Novartis, the company that markets sacubitril/valsartan.
‘Exceedingly reassuring’ safety
The safety data that Dr. Pfeffer reported “are exceedingly reassuring. We didn’t see a signal of harm, and in some of the exploratory endpoints there was some evidence of benefit, so we need to encourage you to continue,” commented Mary N. Walsh, MD, medical director of the heart failure and cardiac transplantation program at Ascension St. Vincent Heart Center of Indiana in Indianapolis.
The PARADISE-MI (Prospective ARNI vs. ACE Inhibitor Trial to Determine Superiority in Reducing Heart Failure Events After MI) trial enrolled 5,669 patients with no history of heart failure within an average of 4 days following an acute MI at 495 sites in 41 countries during 2016-2020, with 8% of enrolled patients from the United States. Patients averaged 64 years of age, about three-quarters were men, about 43% had a history of diabetes, and only 1% were Black; Dr. Pfeffer noted that this is because most patients came from countries with low Black populations. The enrollment criteria required a left ventricular ejection fraction no greater than 40%, and among the enrolled patients this averaged about 37%.
A 10% nonsignificant relative risk reduction for the primary endpoint
The study’s primary endpoint was the combined first-event rate of cardiovascular death, hospitalization for heart failure, or an outpatient visit for heart failure. During a median follow-up of 23 months, this occurred at a rate of 7.4/100 patient years in the ramipril arm and 6.7/100 patient years in the sacubitril/valsartan arm, a 10% relative risk reduction with sacubitril/valsartan that was not significant, which meant all other efficacy analyses were exploratory, Dr. Pfeffer stressed.
Several secondary efficacy analyses showed significant benefits from sacubitril/valsartan, compared with ramipril, including the total number of events that comprised the primary endpoint, with a 21% relative risk reduction associated with sacubitril/valsartan, as well as investigator-reported events. The primary-endpoint benefit from sacubitril/valsartan was also significant in two subgroup analyses: patients aged 65 years or older (roughly half the study cohort), who had a 24% relative risk reduction on sacubitril/valsartan, compared with ramipril, and the 88% of patients who received treatment with percutaneous coronary intervention for their acute MI, who had a 19% relative risk reduction on sacubitril/valsartan, compared with patients who received ramipril.
The study’s safety data showed nearly identical rates in the two treatment arms for total adverse events, serious adverse events, adverse events that led to stopping the study drug, as well as in laboratory measures. The biggest between-treatment differences were a modest excess of hypotension on sacubitril valsartan, 28%, compared with 22% on ramipril, and a modest excess rate of cough on ramipril, 13%, compared with 9% on sacubitril/valsartan.
The added insight the results provide about sacubitril/valsartan comes at a time when U.S. patients continue to struggle to get health insurance coverage for an agent that has been approved for U.S. use in treating heart failure since 2015.
“Our patients do not have access to this important treatment,” declared Dr. Walsh during the press briefing. “The prior authorization process is unbelievable, and some patients have no access unless they pay the full cost on their own. This is an important, real-world problem that we face with this drug.”
PARADISE-MI was sponsored by Novartis, the company that markets sacubitril/valsartan (Entresto). Dr. Pfeffer has received research funding from and is a consultant to Novartis. He is also a consultant to AstraZeneca, Boehringer Ingelheim, Corvidia, DalCor, Eli Lilly, GlaxoSmithKline, Novo Nordisk, Peerbridge, and Sanofi, and he holds equity in DalCor and Peerbridge. Dr. Stevenson has received honoraria from LivaNova and has received research support from Abbott. Dr. Walsh had no disclosures.
FROM ACC 2021
BERENICE: Further evidence of heart safety of dual HER2 blockade
Dual HER2 blockade with pertuzumab (Perjeta) and trastuzumab (Herceptin) on top of anthracycline-based neoadjuvant chemotherapy for early-stage breast cancer was associated with a low rate of clinically relevant cardiac events in the final follow-up of the BERENICE study.
After more than 5 years, 1.0%-1.5% of patients who had locally advanced, inflammatory, or early-stage breast cancer developed heart failure, and around 12%-13% showed any significant changes in left ventricular ejection fraction (LVEF).
Importantly, “there were no new safety concerns that arose during long-term follow-up,” study investigator Chau Dang, MD, said in presenting the findings at the European Society for Medical Oncology: Breast Cancer virtual meeting.
Dr. Dang, a medical oncologist at Memorial Sloan Kettering Cancer Centre in New York, reported that the most common cause of death was disease progression.
BERENICE was designed as a cardiac safety study and so not powered to look at long-term efficacy, which Dr. Dang was clear in reporting. Nevertheless event-free survival (EFS), invasive disease-free survival (IDFS), and overall survival (OS) rates at 5 years were all high, at least a respective 89.2%, 91%, and 93.8%, she said. “The medians have not been reached,” she observed.
“These data support the use of dual HER2 blockade with pertuzumab-trastuzumab–based regimens, including in combination with dose-dense, anthracycline-based chemotherapy, across the neoadjuvant and adjuvant treatment settings for the complete treatment of patients with HER2-positive early-stage breast cancer,” Dr. Dang said.
Evandro de Azambuja, MD, PhD, the invited discussant for the trial agreed that the regimens tested appeared “safe from a cardiac standpoint.” However, “you cannot forget that today we are using much less anthracyclines in our patient population.”
Patients in trials are also very different from those treated in clinical practice, often being younger and much fitter, he said. Therefore, it may be important to look at the baseline cardiac medications and comorbidities, Dr. de Azambuja, a medical oncologist at the Institut Jules Bordet in Brussels, Belgium, suggested.
That said, the BERENICE findings sit well with other trials that have been conducted, Dr. de Azambuja pointed out.
“If we look at other trials that have also tested dual HER2 blockade with anthracycline or nonanthracycline regimens, all of them reassure that dual blockade is not more cardiotoxic than single blockade,” he said. This includes trials such as TRYPHAENA, APHINITY, KRISTINE, NeoSphere and PEONY.
The 3-year IDFS rate of 91% in BERENICE also compares well to that seen in APHINITY (94%), Dr. de Azambuja said.
BERENICE study design
BERENICE was a multicenter, open-label, nonrandomized and noncomparative phase 2 trial that recruited 400 patients across 75 centers in 12 countries.
Eligibility criteria were that participants had to have been centrally confirmed HER2-positive locally advanced, inflammatory or early breast cancer, with the latter defined as tumors bigger than 2 cm or greater than 5 mm in size, and be node-positive. Patients also had to have a starting LVEF of 55% or higher.
Patients were allocated to one of two neoadjuvant chemotherapy regimens depending on the choice of their physician. One group received a regimen of dose-dense doxorubicin and cyclophosphamide (ddAC) given every 2 weeks for four cycles and then paclitaxel every week for 12 cycles. The other group received 5-fluorouracil, epirubicin, and cyclophosphamide (FEC) every 3 weeks for four cycles and then docetaxel every 3 weeks for four cycles.
Pertuzumab and trastuzumab were started at the same time as the taxanes in both groups and given every 3 weeks for four cycles. Patients then underwent surgery and continued pertuzumab/trastuzumab treatment alone for a further 13 cycles.
The co-primary endpoints were the incidence of New York Heart Association class III or IV heart failure and incidence of symptomatic and asymptomatic LVEF decline of 10% or more.
The primary analysis of the trial was published in 2018 and, at that time, it was reported that three patients in the ddAC cohort and none in the FEC cohort experienced heart failure. LVEF decline was observed in a respective 6.5% and 2% of patients.
Discussion points
Dr. de Azambuja noted that the contribution of the chemotherapy to the efficacy cannot be assessed because of the nonrandomized trial design. That should not matter, pointed out Sybille Loibl, MD, PhD, during discussion.
“I think it compares nicely to other trials that looked at dose-dense chemotherapy,” said Dr. Loibl, who is an associate professor at the University of Frankfurt in Germany. “It seems that, in the light of what we consider today probably one of the best anti-HER2 treatments, the chemotherapy is less relevant, and that’s why a dose-dense regimen doesn’t add so much on a standard anthracycline taxane-containing regimen.”
Dr. de Azambuja also commented on the assessment of cardiotoxicity and the use of reduced LVEF as a measure: LVEF decline is a late effect of cardiotoxicity, he observed, and he suggested a different approach in future trials.
“If you use Global Longitudinal Strain, this could be an optimal parameter to detect early subclinical LVEF dysfunction and you should consider it for the next trials looking for cardiac safety. Also, cardiac biomarkers. This was not implemented in this trial, and I strongly recommend this should be for the next trial.”
The BERENICE trial was funded by F. Hoffmann-La Roche. Dr. Dang disclosed receiving consultancy fees from F. Hoffmann-La Roche, Genentech, Daiichi Sankyo, Lilly, and Puma Biotechnology. Dr. de Azambuja was not involved in the study but disclosed receiving honoraria, travel grants, research grants from Roche and Genentech as well as from other companies. Dr. Loibl was one of the cochairs of the session and, among disclosures regarding many other companies, has been an invited speaker for Roche and received reimbursement via her institution for a writing engagement.
Dual HER2 blockade with pertuzumab (Perjeta) and trastuzumab (Herceptin) on top of anthracycline-based neoadjuvant chemotherapy for early-stage breast cancer was associated with a low rate of clinically relevant cardiac events in the final follow-up of the BERENICE study.
After more than 5 years, 1.0%-1.5% of patients who had locally advanced, inflammatory, or early-stage breast cancer developed heart failure, and around 12%-13% showed any significant changes in left ventricular ejection fraction (LVEF).
Importantly, “there were no new safety concerns that arose during long-term follow-up,” study investigator Chau Dang, MD, said in presenting the findings at the European Society for Medical Oncology: Breast Cancer virtual meeting.
Dr. Dang, a medical oncologist at Memorial Sloan Kettering Cancer Centre in New York, reported that the most common cause of death was disease progression.
BERENICE was designed as a cardiac safety study and so not powered to look at long-term efficacy, which Dr. Dang was clear in reporting. Nevertheless event-free survival (EFS), invasive disease-free survival (IDFS), and overall survival (OS) rates at 5 years were all high, at least a respective 89.2%, 91%, and 93.8%, she said. “The medians have not been reached,” she observed.
“These data support the use of dual HER2 blockade with pertuzumab-trastuzumab–based regimens, including in combination with dose-dense, anthracycline-based chemotherapy, across the neoadjuvant and adjuvant treatment settings for the complete treatment of patients with HER2-positive early-stage breast cancer,” Dr. Dang said.
Evandro de Azambuja, MD, PhD, the invited discussant for the trial agreed that the regimens tested appeared “safe from a cardiac standpoint.” However, “you cannot forget that today we are using much less anthracyclines in our patient population.”
Patients in trials are also very different from those treated in clinical practice, often being younger and much fitter, he said. Therefore, it may be important to look at the baseline cardiac medications and comorbidities, Dr. de Azambuja, a medical oncologist at the Institut Jules Bordet in Brussels, Belgium, suggested.
That said, the BERENICE findings sit well with other trials that have been conducted, Dr. de Azambuja pointed out.
“If we look at other trials that have also tested dual HER2 blockade with anthracycline or nonanthracycline regimens, all of them reassure that dual blockade is not more cardiotoxic than single blockade,” he said. This includes trials such as TRYPHAENA, APHINITY, KRISTINE, NeoSphere and PEONY.
The 3-year IDFS rate of 91% in BERENICE also compares well to that seen in APHINITY (94%), Dr. de Azambuja said.
BERENICE study design
BERENICE was a multicenter, open-label, nonrandomized and noncomparative phase 2 trial that recruited 400 patients across 75 centers in 12 countries.
Eligibility criteria were that participants had to have been centrally confirmed HER2-positive locally advanced, inflammatory or early breast cancer, with the latter defined as tumors bigger than 2 cm or greater than 5 mm in size, and be node-positive. Patients also had to have a starting LVEF of 55% or higher.
Patients were allocated to one of two neoadjuvant chemotherapy regimens depending on the choice of their physician. One group received a regimen of dose-dense doxorubicin and cyclophosphamide (ddAC) given every 2 weeks for four cycles and then paclitaxel every week for 12 cycles. The other group received 5-fluorouracil, epirubicin, and cyclophosphamide (FEC) every 3 weeks for four cycles and then docetaxel every 3 weeks for four cycles.
Pertuzumab and trastuzumab were started at the same time as the taxanes in both groups and given every 3 weeks for four cycles. Patients then underwent surgery and continued pertuzumab/trastuzumab treatment alone for a further 13 cycles.
The co-primary endpoints were the incidence of New York Heart Association class III or IV heart failure and incidence of symptomatic and asymptomatic LVEF decline of 10% or more.
The primary analysis of the trial was published in 2018 and, at that time, it was reported that three patients in the ddAC cohort and none in the FEC cohort experienced heart failure. LVEF decline was observed in a respective 6.5% and 2% of patients.
Discussion points
Dr. de Azambuja noted that the contribution of the chemotherapy to the efficacy cannot be assessed because of the nonrandomized trial design. That should not matter, pointed out Sybille Loibl, MD, PhD, during discussion.
“I think it compares nicely to other trials that looked at dose-dense chemotherapy,” said Dr. Loibl, who is an associate professor at the University of Frankfurt in Germany. “It seems that, in the light of what we consider today probably one of the best anti-HER2 treatments, the chemotherapy is less relevant, and that’s why a dose-dense regimen doesn’t add so much on a standard anthracycline taxane-containing regimen.”
Dr. de Azambuja also commented on the assessment of cardiotoxicity and the use of reduced LVEF as a measure: LVEF decline is a late effect of cardiotoxicity, he observed, and he suggested a different approach in future trials.
“If you use Global Longitudinal Strain, this could be an optimal parameter to detect early subclinical LVEF dysfunction and you should consider it for the next trials looking for cardiac safety. Also, cardiac biomarkers. This was not implemented in this trial, and I strongly recommend this should be for the next trial.”
The BERENICE trial was funded by F. Hoffmann-La Roche. Dr. Dang disclosed receiving consultancy fees from F. Hoffmann-La Roche, Genentech, Daiichi Sankyo, Lilly, and Puma Biotechnology. Dr. de Azambuja was not involved in the study but disclosed receiving honoraria, travel grants, research grants from Roche and Genentech as well as from other companies. Dr. Loibl was one of the cochairs of the session and, among disclosures regarding many other companies, has been an invited speaker for Roche and received reimbursement via her institution for a writing engagement.
Dual HER2 blockade with pertuzumab (Perjeta) and trastuzumab (Herceptin) on top of anthracycline-based neoadjuvant chemotherapy for early-stage breast cancer was associated with a low rate of clinically relevant cardiac events in the final follow-up of the BERENICE study.
After more than 5 years, 1.0%-1.5% of patients who had locally advanced, inflammatory, or early-stage breast cancer developed heart failure, and around 12%-13% showed any significant changes in left ventricular ejection fraction (LVEF).
Importantly, “there were no new safety concerns that arose during long-term follow-up,” study investigator Chau Dang, MD, said in presenting the findings at the European Society for Medical Oncology: Breast Cancer virtual meeting.
Dr. Dang, a medical oncologist at Memorial Sloan Kettering Cancer Centre in New York, reported that the most common cause of death was disease progression.
BERENICE was designed as a cardiac safety study and so not powered to look at long-term efficacy, which Dr. Dang was clear in reporting. Nevertheless event-free survival (EFS), invasive disease-free survival (IDFS), and overall survival (OS) rates at 5 years were all high, at least a respective 89.2%, 91%, and 93.8%, she said. “The medians have not been reached,” she observed.
“These data support the use of dual HER2 blockade with pertuzumab-trastuzumab–based regimens, including in combination with dose-dense, anthracycline-based chemotherapy, across the neoadjuvant and adjuvant treatment settings for the complete treatment of patients with HER2-positive early-stage breast cancer,” Dr. Dang said.
Evandro de Azambuja, MD, PhD, the invited discussant for the trial agreed that the regimens tested appeared “safe from a cardiac standpoint.” However, “you cannot forget that today we are using much less anthracyclines in our patient population.”
Patients in trials are also very different from those treated in clinical practice, often being younger and much fitter, he said. Therefore, it may be important to look at the baseline cardiac medications and comorbidities, Dr. de Azambuja, a medical oncologist at the Institut Jules Bordet in Brussels, Belgium, suggested.
That said, the BERENICE findings sit well with other trials that have been conducted, Dr. de Azambuja pointed out.
“If we look at other trials that have also tested dual HER2 blockade with anthracycline or nonanthracycline regimens, all of them reassure that dual blockade is not more cardiotoxic than single blockade,” he said. This includes trials such as TRYPHAENA, APHINITY, KRISTINE, NeoSphere and PEONY.
The 3-year IDFS rate of 91% in BERENICE also compares well to that seen in APHINITY (94%), Dr. de Azambuja said.
BERENICE study design
BERENICE was a multicenter, open-label, nonrandomized and noncomparative phase 2 trial that recruited 400 patients across 75 centers in 12 countries.
Eligibility criteria were that participants had to have been centrally confirmed HER2-positive locally advanced, inflammatory or early breast cancer, with the latter defined as tumors bigger than 2 cm or greater than 5 mm in size, and be node-positive. Patients also had to have a starting LVEF of 55% or higher.
Patients were allocated to one of two neoadjuvant chemotherapy regimens depending on the choice of their physician. One group received a regimen of dose-dense doxorubicin and cyclophosphamide (ddAC) given every 2 weeks for four cycles and then paclitaxel every week for 12 cycles. The other group received 5-fluorouracil, epirubicin, and cyclophosphamide (FEC) every 3 weeks for four cycles and then docetaxel every 3 weeks for four cycles.
Pertuzumab and trastuzumab were started at the same time as the taxanes in both groups and given every 3 weeks for four cycles. Patients then underwent surgery and continued pertuzumab/trastuzumab treatment alone for a further 13 cycles.
The co-primary endpoints were the incidence of New York Heart Association class III or IV heart failure and incidence of symptomatic and asymptomatic LVEF decline of 10% or more.
The primary analysis of the trial was published in 2018 and, at that time, it was reported that three patients in the ddAC cohort and none in the FEC cohort experienced heart failure. LVEF decline was observed in a respective 6.5% and 2% of patients.
Discussion points
Dr. de Azambuja noted that the contribution of the chemotherapy to the efficacy cannot be assessed because of the nonrandomized trial design. That should not matter, pointed out Sybille Loibl, MD, PhD, during discussion.
“I think it compares nicely to other trials that looked at dose-dense chemotherapy,” said Dr. Loibl, who is an associate professor at the University of Frankfurt in Germany. “It seems that, in the light of what we consider today probably one of the best anti-HER2 treatments, the chemotherapy is less relevant, and that’s why a dose-dense regimen doesn’t add so much on a standard anthracycline taxane-containing regimen.”
Dr. de Azambuja also commented on the assessment of cardiotoxicity and the use of reduced LVEF as a measure: LVEF decline is a late effect of cardiotoxicity, he observed, and he suggested a different approach in future trials.
“If you use Global Longitudinal Strain, this could be an optimal parameter to detect early subclinical LVEF dysfunction and you should consider it for the next trials looking for cardiac safety. Also, cardiac biomarkers. This was not implemented in this trial, and I strongly recommend this should be for the next trial.”
The BERENICE trial was funded by F. Hoffmann-La Roche. Dr. Dang disclosed receiving consultancy fees from F. Hoffmann-La Roche, Genentech, Daiichi Sankyo, Lilly, and Puma Biotechnology. Dr. de Azambuja was not involved in the study but disclosed receiving honoraria, travel grants, research grants from Roche and Genentech as well as from other companies. Dr. Loibl was one of the cochairs of the session and, among disclosures regarding many other companies, has been an invited speaker for Roche and received reimbursement via her institution for a writing engagement.
FROM ESMO BREAST CANCER 2021
ACC 21 looks to repeat success despite pandemic headwinds
The American College of Cardiology pulled off an impressive all-virtual meeting in March 2020, less than 3 weeks after canceling its in-person event and just 2 weeks after COVID-19 was declared a national emergency.
Optimistic plans for the annual scientific sessions of the American College of Cardiology (ACC 2021) to be a March hybrid affair in Atlanta pivoted not once, but twice, as the pandemic evolved, with the date pushed back 2 full months, to May 15-17, and the format revised to fully virtual.
“While this meeting is being delivered virtually, I think you’ll see there have been benefits in the time to plan and also the lessons that ACC has learned in virtual education over the past year. This has come together to really create a robust educational and scientific agenda,” ACC 2021 chair Pamela B. Morris, MD, said in a press conference focused on the upcoming meeting.
Over the 3 days, there will be more than 200 education sessions, 10 guideline-specific sessions, and 11 learning pathways that include core areas, but also special topics, such as COVID-19 and the emerging cardio-obstetrics subspecialty.
The meeting will be delivered through a new virtual education program built to optimize real-time interaction between faculty members and attendees, she said. A dedicated portal on the platform will allow attendees to interact virtually, for example, with presenters of the nearly 3,000 ePosters and 420 moderated posters.
For those suffering from Zoom fatigue, the increasingly popular Heart2Heart stage talks have also been converted to podcasts, which cover topics like gender equity in cardiology, the evolving role of advanced practice professionals, and “one of my favorites: art as a tool for healing,” said Dr. Morris, from the Medical University of South Carolina, Charleston. “Those sessions are really not to be missed.”
Reconnecting is an underlying theme of the meeting but the great divider will not be ignored. COVID-19 will be the focus of two 90-minute Intensive Sessions on Saturday, May 15, the first kicking off at 10:30 a.m. ET, with the Bishop Keynote lecture on bringing health equity to the frontline of cardiovascular care, followed by lessons learned during the pandemic, how to conduct clinical trials, and vaccine development.
The second session, set for 12:15 p.m., continues the “silver linings” theme, with case presentations on advances in telehealth, myocardial involvement, and thrombosis in COVID. For those wanting more, 18 abstracts are on tap in a 2-hour Spotlight on Special Topics session beginning at 2:30 p.m.
Asked about the pandemic’s effect on bringing science to fruition this past year, Dr. Morris said there’s no question it’s slowed some of the progress the cardiology community had made but, like clinical practice, “we’ve also surmounted many of those obstacles.”
“I think research has rebounded,” she said. “Just in terms of the number of abstracts and the quality of abstracts that were submitted this year, I don’t think there’s any question that we are right on par with previous years.”
Indeed, 5,258 abstracts from 76 countries were submitted, with more than 3,400 chosen for oral and poster presentation, including 25 late-breaking clinical trials to be presented in five sessions.
The late-breaking presentations and discussions will be prerecorded but speakers and panelists have been invited to be present during the streaming to answer live any questions that may arise in the chat box, ACC 2021 vice chair Douglas Drachman, MD, Massachusetts General Hospital, Boston, said in an interview.
Late-breaking clinical trials
The Joint ACC/JACC Late-Breaking Clinical Trials I (Saturday, May 15, 9:00 a.m.–-10:00 a.m.) kicks off with PARADISE-MI, the first head-to-head comparison of an angiotensin receptor neprilysin inhibitor (ARNI) and an ACE inhibitor in patients with reduced ejection fractions (EFs) after MI but no history of heart failure (HF), studying 200 mg sacubitril/valsartan (Entresto) versus 5 mg of ramipril, both twice daily, in 5,669 patients.
Sacubitril/valsartan was initially approved for HF with reduced EF and added a new indication to treat some HF patients with preserved EF. Novartis, however, recently told investors that although numerical trends consistently favored the ARNI over the ACE inhibitor ramipril, the phase 3 study failed to meet the primary endpoint for efficacy superiority of reducing the risk for cardiovascular (CV) death and HF events after an acute MI.
Second up is ADAPTABLE, which looks to close a surprising evidence gap over whether 81 mg or 325 mg daily is the optimal dose of the ubiquitously prescribed aspirin for secondary prevention in high-risk patients with established atherosclerotic CV disease.
The open-label, randomized study will look at efficacy and major bleeding over roughly 4 years in 15,000 patients within PCORnet, the National Patient-centered Clinical Research Network, a partnership of clinical research, health plan research, and patient-powered networks created to streamline patient-reported outcomes research.
“This study will not only give important clinical information for us, practically speaking, whether we should prescribe lower- or higher-dose aspirin, but it may also serve as a template for future pragmatic clinical trial design in the real world,” Dr. Drachman said during the press conference.
Up next is the 4,812-patient Canadian LAAOS III, the largest trial to examine the efficacy of left atrial appendage occlusion for stroke prevention in patients with atrial fibrillation (AFib) already undergoing cardiac surgery. The primary outcome is the first occurrence of stroke or systemic arterial embolism over an average follow-up of 4 years.
Percutaneous closure of the left atrial appendage (LAA) has been shown to reduce stroke in AFib patients at high-risk of bleeding on systemic anticoagulation. But these devices can be expensive and studies haven’t included patients who also have valvular heart disease, a group that actually comprises more than half of patients undergoing cardiac surgery who also have AFib, he noted.
At the same time, surgical LAA closure studies have been small and have had very mixed results. “There isn’t a large-scale rigorous assessment out there for these patients undergoing surgery, so I think this is going to be fascinating to see,” Dr. Drachman said.
The session closes with ATLANTIS, which looks to shed some light on the role of anticoagulation therapy in patients after transcatheter aortic valve replacement (TAVR or TAVI). POPular TAVI, presented at ACC 2020, showed aspirin alone was the preferred antithrombotic therapy over aspirin plus clopidogrel (Plavix) in patients not on oral anticoagulants, but the optimal anticoagulation regimen remains unsettled.
The French open-label, 1,510-patient ATLANTIS trial examined whether the novel oral anticoagulant apixaban (Eliquis) is superior in preventing CV events after TAVR, compared with antiplatelet therapy in patients without an indication for anticoagulation and compared with vitamin K antagonists in those receiving anticoagulants.
An ATLANTIS 4D CT substudy of valve thrombosis is also slated for Saturday’s Featured Clinical Research 1 session at 12:15 p.m. to 1:45 p.m..
Sunday LBCTs
Dr. Drachman highlighted a series of other late-breaking studies, including the global DARE-19 trial testing the diabetes and HF drug dapagliflozin (Farxiga) given with local standard-of-care therapy for 30 days in hospitalized COVID-19 patients with CV, metabolic, or renal risk factors.
Although sodium-glucose cotransporter-2 inhibitors have been white-hot of late, top-line results reported last month show dapagliflozin failed to achieve statistical significance for the primary endpoints of reducing organ dysfunction and all-cause mortality and for improving recovery. Details will be presented in the Joint ACC/JAMA Late-Breaking Clinical Trials II (Sunday, May 16, 8:00 a.m.-9:30 a.m.).
Two trials, FLOWER-MI and RADIANCE-HTN TRIO, were singled out in the Joint ACC/New England Journal of Medicine Late-Breaking Clinical Trials III (Sunday, May 16, 10:45 a.m.-12:00 p.m.). FLOWER-MI examines whether fractional flow reserve (FFR) is better than angiography to guide complete multivessel revascularization in ST-elevation MI patients with at least 50% stenosis in at least one nonculprit lesion requiring percutaneous coronary intervention (PCI). Recent studies have shown the superiority of FFR-guided PCI for nonculprit lesions, compared with culprit lesion treatment-only, but this is the first time FFR- and angiography-guided PCI have been compared in STEMI patients.
RADIANCE-HTN TRIO already tipped its hand, with top-line results reported in late 2020 showing that the trial met its primary efficacy endpoint of greater reduction in daytime blood pressure over 2 months with the Paradise endovascular ultrasound renal denervation system, compared with a sham procedure, in 136 patients with resistant hypertension, importantly, after being given a single pill containing a calcium channel blocker, angiotensin II receptor blocker, and diuretic.
Renal denervation for hypertension has been making something of a comeback, with the 2018 RADIANCE-HTN SOLO reporting better ambulatory blood pressure control with the Paradise system than with a sham procedure in the absence of antihypertensive agents. The device has been granted breakthrough device designation from the Food and Drug Administration for the treatment of hypertensive patients who are unable to sufficiently respond to or are intolerant of antihypertensive therapy.
Monday LBCTs
In the Late-Breaking Clinical Trials IV session (Monday, May 17, 8 a.m.–9:30 a.m.), Drachman called out a secondary analysis from GALATIC-HF looking at the impact of EF on the therapeutic effect of omecamtiv mecarbil. In last year’s primary analysis, the selective cardiac myosin activator produced a modest but significant reduction in HF events or CV death in 8,232 patients with HF and an EF of 35% or less.
Rounding out the list is the Canadian CAPITAL CHILL study of moderate versus mild therapeutic hypothermia in out-of-hospital cardiac arrest, to be presented in the final Late-Breaking Clinical Trials V session (Monday, May 17, 10:45 a.m.–12:00 p.m.).
The double-blind trial sought to determine whether neurologic outcomes at 6 months are improved by targeting a core temperature of 31 ˚C versus 34 ˚C after the return of spontaneous circulation in comatose survivors of out-of-hospital cardiac arrest.
“For me, I think this could really change practice and has personal relevance from experience with cardiac arrest survivors that I’ve known and care for very deeply,” Dr. Drachman said in an interview. “I think that there’s a lot of opportunity here as well.”
Asked what other trials have the potential to change practice, Dr. Drachman said FLOWER-MI holds particular interest because it looks at how to manage patients with STEMI with multiple lesions at the point of care.
“We’ve gained a lot of clarity from several other prior clinical trials, but this will help to answer the question in a slightly different way of saying: can you eyeball it, can you look at the angiogram and say whether or not that other, nonculprit lesion ought to be treated in the same hospitalization or should you really be using a pressure wire,” he said. “For me as an interventionalist, this is really important because when you finish up doing an intervention on a patient it might be the middle of the night and the patient may be more or less stable, but you’ve already exposed them to the risk of a procedure, should you then move on and do another aspect of the procedure to interrogate with a pressure wire a remaining narrowing? I think that’s very important; that’ll help me make decisions on a day-to-day basis.”
Dr. Drachman also cited RADIANCE-HTN TRIO because it employs an endovascular technique to control blood pressure in patients with hypertension, specifically those resistant to multiple drugs.
During the press conference, Dr. Morris, a preventive cardiologist, put her money on the ADAPTABLE study of aspirin dosing, reiterating that the unique trial design could inform future research, and on Sunday’s 8:45 a.m. late-breaking post hoc analysis from the STRENGTH trial that looks to pick up where the controversy over omega-3 fatty acid preparations left off at last year’s American Heart Association meeting.
A lack of benefit on CV event rates reported with Epanova, a high-dose combination of eicosapentaenoic acid (EPA) and docosahexaenoic acid, led to a contentious debate over how to reconcile STRENGTH with the findings from REDUCE-IT, which showed a 25% relative risk reduction in major CV events with the EPA product icosapent ethyl (Vascepa).
STRENGTH investigator Steven Nissen, MD, Cleveland Clinic, and REDUCE-IT investigator and session panelist Deepak Bhatt, MD, Brigham and Women’s Hospital, Boston, will share the virtual stage at ACC 2021, but Dr. Morris said the “good news” is both researchers know one another very well and “will really be focusing on no political issues, just the omega-3 fatty levels in the bloodstream and what does that mean in either trial.
“This is not designed to be a debate, point counterpoint,” she added.
For that, as all cardiologists and journalists know, there will be the wild and woolly #CardioTwitter sphere.
A version of this article first appeared on Medscape.com.
The American College of Cardiology pulled off an impressive all-virtual meeting in March 2020, less than 3 weeks after canceling its in-person event and just 2 weeks after COVID-19 was declared a national emergency.
Optimistic plans for the annual scientific sessions of the American College of Cardiology (ACC 2021) to be a March hybrid affair in Atlanta pivoted not once, but twice, as the pandemic evolved, with the date pushed back 2 full months, to May 15-17, and the format revised to fully virtual.
“While this meeting is being delivered virtually, I think you’ll see there have been benefits in the time to plan and also the lessons that ACC has learned in virtual education over the past year. This has come together to really create a robust educational and scientific agenda,” ACC 2021 chair Pamela B. Morris, MD, said in a press conference focused on the upcoming meeting.
Over the 3 days, there will be more than 200 education sessions, 10 guideline-specific sessions, and 11 learning pathways that include core areas, but also special topics, such as COVID-19 and the emerging cardio-obstetrics subspecialty.
The meeting will be delivered through a new virtual education program built to optimize real-time interaction between faculty members and attendees, she said. A dedicated portal on the platform will allow attendees to interact virtually, for example, with presenters of the nearly 3,000 ePosters and 420 moderated posters.
For those suffering from Zoom fatigue, the increasingly popular Heart2Heart stage talks have also been converted to podcasts, which cover topics like gender equity in cardiology, the evolving role of advanced practice professionals, and “one of my favorites: art as a tool for healing,” said Dr. Morris, from the Medical University of South Carolina, Charleston. “Those sessions are really not to be missed.”
Reconnecting is an underlying theme of the meeting but the great divider will not be ignored. COVID-19 will be the focus of two 90-minute Intensive Sessions on Saturday, May 15, the first kicking off at 10:30 a.m. ET, with the Bishop Keynote lecture on bringing health equity to the frontline of cardiovascular care, followed by lessons learned during the pandemic, how to conduct clinical trials, and vaccine development.
The second session, set for 12:15 p.m., continues the “silver linings” theme, with case presentations on advances in telehealth, myocardial involvement, and thrombosis in COVID. For those wanting more, 18 abstracts are on tap in a 2-hour Spotlight on Special Topics session beginning at 2:30 p.m.
Asked about the pandemic’s effect on bringing science to fruition this past year, Dr. Morris said there’s no question it’s slowed some of the progress the cardiology community had made but, like clinical practice, “we’ve also surmounted many of those obstacles.”
“I think research has rebounded,” she said. “Just in terms of the number of abstracts and the quality of abstracts that were submitted this year, I don’t think there’s any question that we are right on par with previous years.”
Indeed, 5,258 abstracts from 76 countries were submitted, with more than 3,400 chosen for oral and poster presentation, including 25 late-breaking clinical trials to be presented in five sessions.
The late-breaking presentations and discussions will be prerecorded but speakers and panelists have been invited to be present during the streaming to answer live any questions that may arise in the chat box, ACC 2021 vice chair Douglas Drachman, MD, Massachusetts General Hospital, Boston, said in an interview.
Late-breaking clinical trials
The Joint ACC/JACC Late-Breaking Clinical Trials I (Saturday, May 15, 9:00 a.m.–-10:00 a.m.) kicks off with PARADISE-MI, the first head-to-head comparison of an angiotensin receptor neprilysin inhibitor (ARNI) and an ACE inhibitor in patients with reduced ejection fractions (EFs) after MI but no history of heart failure (HF), studying 200 mg sacubitril/valsartan (Entresto) versus 5 mg of ramipril, both twice daily, in 5,669 patients.
Sacubitril/valsartan was initially approved for HF with reduced EF and added a new indication to treat some HF patients with preserved EF. Novartis, however, recently told investors that although numerical trends consistently favored the ARNI over the ACE inhibitor ramipril, the phase 3 study failed to meet the primary endpoint for efficacy superiority of reducing the risk for cardiovascular (CV) death and HF events after an acute MI.
Second up is ADAPTABLE, which looks to close a surprising evidence gap over whether 81 mg or 325 mg daily is the optimal dose of the ubiquitously prescribed aspirin for secondary prevention in high-risk patients with established atherosclerotic CV disease.
The open-label, randomized study will look at efficacy and major bleeding over roughly 4 years in 15,000 patients within PCORnet, the National Patient-centered Clinical Research Network, a partnership of clinical research, health plan research, and patient-powered networks created to streamline patient-reported outcomes research.
“This study will not only give important clinical information for us, practically speaking, whether we should prescribe lower- or higher-dose aspirin, but it may also serve as a template for future pragmatic clinical trial design in the real world,” Dr. Drachman said during the press conference.
Up next is the 4,812-patient Canadian LAAOS III, the largest trial to examine the efficacy of left atrial appendage occlusion for stroke prevention in patients with atrial fibrillation (AFib) already undergoing cardiac surgery. The primary outcome is the first occurrence of stroke or systemic arterial embolism over an average follow-up of 4 years.
Percutaneous closure of the left atrial appendage (LAA) has been shown to reduce stroke in AFib patients at high-risk of bleeding on systemic anticoagulation. But these devices can be expensive and studies haven’t included patients who also have valvular heart disease, a group that actually comprises more than half of patients undergoing cardiac surgery who also have AFib, he noted.
At the same time, surgical LAA closure studies have been small and have had very mixed results. “There isn’t a large-scale rigorous assessment out there for these patients undergoing surgery, so I think this is going to be fascinating to see,” Dr. Drachman said.
The session closes with ATLANTIS, which looks to shed some light on the role of anticoagulation therapy in patients after transcatheter aortic valve replacement (TAVR or TAVI). POPular TAVI, presented at ACC 2020, showed aspirin alone was the preferred antithrombotic therapy over aspirin plus clopidogrel (Plavix) in patients not on oral anticoagulants, but the optimal anticoagulation regimen remains unsettled.
The French open-label, 1,510-patient ATLANTIS trial examined whether the novel oral anticoagulant apixaban (Eliquis) is superior in preventing CV events after TAVR, compared with antiplatelet therapy in patients without an indication for anticoagulation and compared with vitamin K antagonists in those receiving anticoagulants.
An ATLANTIS 4D CT substudy of valve thrombosis is also slated for Saturday’s Featured Clinical Research 1 session at 12:15 p.m. to 1:45 p.m..
Sunday LBCTs
Dr. Drachman highlighted a series of other late-breaking studies, including the global DARE-19 trial testing the diabetes and HF drug dapagliflozin (Farxiga) given with local standard-of-care therapy for 30 days in hospitalized COVID-19 patients with CV, metabolic, or renal risk factors.
Although sodium-glucose cotransporter-2 inhibitors have been white-hot of late, top-line results reported last month show dapagliflozin failed to achieve statistical significance for the primary endpoints of reducing organ dysfunction and all-cause mortality and for improving recovery. Details will be presented in the Joint ACC/JAMA Late-Breaking Clinical Trials II (Sunday, May 16, 8:00 a.m.-9:30 a.m.).
Two trials, FLOWER-MI and RADIANCE-HTN TRIO, were singled out in the Joint ACC/New England Journal of Medicine Late-Breaking Clinical Trials III (Sunday, May 16, 10:45 a.m.-12:00 p.m.). FLOWER-MI examines whether fractional flow reserve (FFR) is better than angiography to guide complete multivessel revascularization in ST-elevation MI patients with at least 50% stenosis in at least one nonculprit lesion requiring percutaneous coronary intervention (PCI). Recent studies have shown the superiority of FFR-guided PCI for nonculprit lesions, compared with culprit lesion treatment-only, but this is the first time FFR- and angiography-guided PCI have been compared in STEMI patients.
RADIANCE-HTN TRIO already tipped its hand, with top-line results reported in late 2020 showing that the trial met its primary efficacy endpoint of greater reduction in daytime blood pressure over 2 months with the Paradise endovascular ultrasound renal denervation system, compared with a sham procedure, in 136 patients with resistant hypertension, importantly, after being given a single pill containing a calcium channel blocker, angiotensin II receptor blocker, and diuretic.
Renal denervation for hypertension has been making something of a comeback, with the 2018 RADIANCE-HTN SOLO reporting better ambulatory blood pressure control with the Paradise system than with a sham procedure in the absence of antihypertensive agents. The device has been granted breakthrough device designation from the Food and Drug Administration for the treatment of hypertensive patients who are unable to sufficiently respond to or are intolerant of antihypertensive therapy.
Monday LBCTs
In the Late-Breaking Clinical Trials IV session (Monday, May 17, 8 a.m.–9:30 a.m.), Drachman called out a secondary analysis from GALATIC-HF looking at the impact of EF on the therapeutic effect of omecamtiv mecarbil. In last year’s primary analysis, the selective cardiac myosin activator produced a modest but significant reduction in HF events or CV death in 8,232 patients with HF and an EF of 35% or less.
Rounding out the list is the Canadian CAPITAL CHILL study of moderate versus mild therapeutic hypothermia in out-of-hospital cardiac arrest, to be presented in the final Late-Breaking Clinical Trials V session (Monday, May 17, 10:45 a.m.–12:00 p.m.).
The double-blind trial sought to determine whether neurologic outcomes at 6 months are improved by targeting a core temperature of 31 ˚C versus 34 ˚C after the return of spontaneous circulation in comatose survivors of out-of-hospital cardiac arrest.
“For me, I think this could really change practice and has personal relevance from experience with cardiac arrest survivors that I’ve known and care for very deeply,” Dr. Drachman said in an interview. “I think that there’s a lot of opportunity here as well.”
Asked what other trials have the potential to change practice, Dr. Drachman said FLOWER-MI holds particular interest because it looks at how to manage patients with STEMI with multiple lesions at the point of care.
“We’ve gained a lot of clarity from several other prior clinical trials, but this will help to answer the question in a slightly different way of saying: can you eyeball it, can you look at the angiogram and say whether or not that other, nonculprit lesion ought to be treated in the same hospitalization or should you really be using a pressure wire,” he said. “For me as an interventionalist, this is really important because when you finish up doing an intervention on a patient it might be the middle of the night and the patient may be more or less stable, but you’ve already exposed them to the risk of a procedure, should you then move on and do another aspect of the procedure to interrogate with a pressure wire a remaining narrowing? I think that’s very important; that’ll help me make decisions on a day-to-day basis.”
Dr. Drachman also cited RADIANCE-HTN TRIO because it employs an endovascular technique to control blood pressure in patients with hypertension, specifically those resistant to multiple drugs.
During the press conference, Dr. Morris, a preventive cardiologist, put her money on the ADAPTABLE study of aspirin dosing, reiterating that the unique trial design could inform future research, and on Sunday’s 8:45 a.m. late-breaking post hoc analysis from the STRENGTH trial that looks to pick up where the controversy over omega-3 fatty acid preparations left off at last year’s American Heart Association meeting.
A lack of benefit on CV event rates reported with Epanova, a high-dose combination of eicosapentaenoic acid (EPA) and docosahexaenoic acid, led to a contentious debate over how to reconcile STRENGTH with the findings from REDUCE-IT, which showed a 25% relative risk reduction in major CV events with the EPA product icosapent ethyl (Vascepa).
STRENGTH investigator Steven Nissen, MD, Cleveland Clinic, and REDUCE-IT investigator and session panelist Deepak Bhatt, MD, Brigham and Women’s Hospital, Boston, will share the virtual stage at ACC 2021, but Dr. Morris said the “good news” is both researchers know one another very well and “will really be focusing on no political issues, just the omega-3 fatty levels in the bloodstream and what does that mean in either trial.
“This is not designed to be a debate, point counterpoint,” she added.
For that, as all cardiologists and journalists know, there will be the wild and woolly #CardioTwitter sphere.
A version of this article first appeared on Medscape.com.
The American College of Cardiology pulled off an impressive all-virtual meeting in March 2020, less than 3 weeks after canceling its in-person event and just 2 weeks after COVID-19 was declared a national emergency.
Optimistic plans for the annual scientific sessions of the American College of Cardiology (ACC 2021) to be a March hybrid affair in Atlanta pivoted not once, but twice, as the pandemic evolved, with the date pushed back 2 full months, to May 15-17, and the format revised to fully virtual.
“While this meeting is being delivered virtually, I think you’ll see there have been benefits in the time to plan and also the lessons that ACC has learned in virtual education over the past year. This has come together to really create a robust educational and scientific agenda,” ACC 2021 chair Pamela B. Morris, MD, said in a press conference focused on the upcoming meeting.
Over the 3 days, there will be more than 200 education sessions, 10 guideline-specific sessions, and 11 learning pathways that include core areas, but also special topics, such as COVID-19 and the emerging cardio-obstetrics subspecialty.
The meeting will be delivered through a new virtual education program built to optimize real-time interaction between faculty members and attendees, she said. A dedicated portal on the platform will allow attendees to interact virtually, for example, with presenters of the nearly 3,000 ePosters and 420 moderated posters.
For those suffering from Zoom fatigue, the increasingly popular Heart2Heart stage talks have also been converted to podcasts, which cover topics like gender equity in cardiology, the evolving role of advanced practice professionals, and “one of my favorites: art as a tool for healing,” said Dr. Morris, from the Medical University of South Carolina, Charleston. “Those sessions are really not to be missed.”
Reconnecting is an underlying theme of the meeting but the great divider will not be ignored. COVID-19 will be the focus of two 90-minute Intensive Sessions on Saturday, May 15, the first kicking off at 10:30 a.m. ET, with the Bishop Keynote lecture on bringing health equity to the frontline of cardiovascular care, followed by lessons learned during the pandemic, how to conduct clinical trials, and vaccine development.
The second session, set for 12:15 p.m., continues the “silver linings” theme, with case presentations on advances in telehealth, myocardial involvement, and thrombosis in COVID. For those wanting more, 18 abstracts are on tap in a 2-hour Spotlight on Special Topics session beginning at 2:30 p.m.
Asked about the pandemic’s effect on bringing science to fruition this past year, Dr. Morris said there’s no question it’s slowed some of the progress the cardiology community had made but, like clinical practice, “we’ve also surmounted many of those obstacles.”
“I think research has rebounded,” she said. “Just in terms of the number of abstracts and the quality of abstracts that were submitted this year, I don’t think there’s any question that we are right on par with previous years.”
Indeed, 5,258 abstracts from 76 countries were submitted, with more than 3,400 chosen for oral and poster presentation, including 25 late-breaking clinical trials to be presented in five sessions.
The late-breaking presentations and discussions will be prerecorded but speakers and panelists have been invited to be present during the streaming to answer live any questions that may arise in the chat box, ACC 2021 vice chair Douglas Drachman, MD, Massachusetts General Hospital, Boston, said in an interview.
Late-breaking clinical trials
The Joint ACC/JACC Late-Breaking Clinical Trials I (Saturday, May 15, 9:00 a.m.–-10:00 a.m.) kicks off with PARADISE-MI, the first head-to-head comparison of an angiotensin receptor neprilysin inhibitor (ARNI) and an ACE inhibitor in patients with reduced ejection fractions (EFs) after MI but no history of heart failure (HF), studying 200 mg sacubitril/valsartan (Entresto) versus 5 mg of ramipril, both twice daily, in 5,669 patients.
Sacubitril/valsartan was initially approved for HF with reduced EF and added a new indication to treat some HF patients with preserved EF. Novartis, however, recently told investors that although numerical trends consistently favored the ARNI over the ACE inhibitor ramipril, the phase 3 study failed to meet the primary endpoint for efficacy superiority of reducing the risk for cardiovascular (CV) death and HF events after an acute MI.
Second up is ADAPTABLE, which looks to close a surprising evidence gap over whether 81 mg or 325 mg daily is the optimal dose of the ubiquitously prescribed aspirin for secondary prevention in high-risk patients with established atherosclerotic CV disease.
The open-label, randomized study will look at efficacy and major bleeding over roughly 4 years in 15,000 patients within PCORnet, the National Patient-centered Clinical Research Network, a partnership of clinical research, health plan research, and patient-powered networks created to streamline patient-reported outcomes research.
“This study will not only give important clinical information for us, practically speaking, whether we should prescribe lower- or higher-dose aspirin, but it may also serve as a template for future pragmatic clinical trial design in the real world,” Dr. Drachman said during the press conference.
Up next is the 4,812-patient Canadian LAAOS III, the largest trial to examine the efficacy of left atrial appendage occlusion for stroke prevention in patients with atrial fibrillation (AFib) already undergoing cardiac surgery. The primary outcome is the first occurrence of stroke or systemic arterial embolism over an average follow-up of 4 years.
Percutaneous closure of the left atrial appendage (LAA) has been shown to reduce stroke in AFib patients at high-risk of bleeding on systemic anticoagulation. But these devices can be expensive and studies haven’t included patients who also have valvular heart disease, a group that actually comprises more than half of patients undergoing cardiac surgery who also have AFib, he noted.
At the same time, surgical LAA closure studies have been small and have had very mixed results. “There isn’t a large-scale rigorous assessment out there for these patients undergoing surgery, so I think this is going to be fascinating to see,” Dr. Drachman said.
The session closes with ATLANTIS, which looks to shed some light on the role of anticoagulation therapy in patients after transcatheter aortic valve replacement (TAVR or TAVI). POPular TAVI, presented at ACC 2020, showed aspirin alone was the preferred antithrombotic therapy over aspirin plus clopidogrel (Plavix) in patients not on oral anticoagulants, but the optimal anticoagulation regimen remains unsettled.
The French open-label, 1,510-patient ATLANTIS trial examined whether the novel oral anticoagulant apixaban (Eliquis) is superior in preventing CV events after TAVR, compared with antiplatelet therapy in patients without an indication for anticoagulation and compared with vitamin K antagonists in those receiving anticoagulants.
An ATLANTIS 4D CT substudy of valve thrombosis is also slated for Saturday’s Featured Clinical Research 1 session at 12:15 p.m. to 1:45 p.m..
Sunday LBCTs
Dr. Drachman highlighted a series of other late-breaking studies, including the global DARE-19 trial testing the diabetes and HF drug dapagliflozin (Farxiga) given with local standard-of-care therapy for 30 days in hospitalized COVID-19 patients with CV, metabolic, or renal risk factors.
Although sodium-glucose cotransporter-2 inhibitors have been white-hot of late, top-line results reported last month show dapagliflozin failed to achieve statistical significance for the primary endpoints of reducing organ dysfunction and all-cause mortality and for improving recovery. Details will be presented in the Joint ACC/JAMA Late-Breaking Clinical Trials II (Sunday, May 16, 8:00 a.m.-9:30 a.m.).
Two trials, FLOWER-MI and RADIANCE-HTN TRIO, were singled out in the Joint ACC/New England Journal of Medicine Late-Breaking Clinical Trials III (Sunday, May 16, 10:45 a.m.-12:00 p.m.). FLOWER-MI examines whether fractional flow reserve (FFR) is better than angiography to guide complete multivessel revascularization in ST-elevation MI patients with at least 50% stenosis in at least one nonculprit lesion requiring percutaneous coronary intervention (PCI). Recent studies have shown the superiority of FFR-guided PCI for nonculprit lesions, compared with culprit lesion treatment-only, but this is the first time FFR- and angiography-guided PCI have been compared in STEMI patients.
RADIANCE-HTN TRIO already tipped its hand, with top-line results reported in late 2020 showing that the trial met its primary efficacy endpoint of greater reduction in daytime blood pressure over 2 months with the Paradise endovascular ultrasound renal denervation system, compared with a sham procedure, in 136 patients with resistant hypertension, importantly, after being given a single pill containing a calcium channel blocker, angiotensin II receptor blocker, and diuretic.
Renal denervation for hypertension has been making something of a comeback, with the 2018 RADIANCE-HTN SOLO reporting better ambulatory blood pressure control with the Paradise system than with a sham procedure in the absence of antihypertensive agents. The device has been granted breakthrough device designation from the Food and Drug Administration for the treatment of hypertensive patients who are unable to sufficiently respond to or are intolerant of antihypertensive therapy.
Monday LBCTs
In the Late-Breaking Clinical Trials IV session (Monday, May 17, 8 a.m.–9:30 a.m.), Drachman called out a secondary analysis from GALATIC-HF looking at the impact of EF on the therapeutic effect of omecamtiv mecarbil. In last year’s primary analysis, the selective cardiac myosin activator produced a modest but significant reduction in HF events or CV death in 8,232 patients with HF and an EF of 35% or less.
Rounding out the list is the Canadian CAPITAL CHILL study of moderate versus mild therapeutic hypothermia in out-of-hospital cardiac arrest, to be presented in the final Late-Breaking Clinical Trials V session (Monday, May 17, 10:45 a.m.–12:00 p.m.).
The double-blind trial sought to determine whether neurologic outcomes at 6 months are improved by targeting a core temperature of 31 ˚C versus 34 ˚C after the return of spontaneous circulation in comatose survivors of out-of-hospital cardiac arrest.
“For me, I think this could really change practice and has personal relevance from experience with cardiac arrest survivors that I’ve known and care for very deeply,” Dr. Drachman said in an interview. “I think that there’s a lot of opportunity here as well.”
Asked what other trials have the potential to change practice, Dr. Drachman said FLOWER-MI holds particular interest because it looks at how to manage patients with STEMI with multiple lesions at the point of care.
“We’ve gained a lot of clarity from several other prior clinical trials, but this will help to answer the question in a slightly different way of saying: can you eyeball it, can you look at the angiogram and say whether or not that other, nonculprit lesion ought to be treated in the same hospitalization or should you really be using a pressure wire,” he said. “For me as an interventionalist, this is really important because when you finish up doing an intervention on a patient it might be the middle of the night and the patient may be more or less stable, but you’ve already exposed them to the risk of a procedure, should you then move on and do another aspect of the procedure to interrogate with a pressure wire a remaining narrowing? I think that’s very important; that’ll help me make decisions on a day-to-day basis.”
Dr. Drachman also cited RADIANCE-HTN TRIO because it employs an endovascular technique to control blood pressure in patients with hypertension, specifically those resistant to multiple drugs.
During the press conference, Dr. Morris, a preventive cardiologist, put her money on the ADAPTABLE study of aspirin dosing, reiterating that the unique trial design could inform future research, and on Sunday’s 8:45 a.m. late-breaking post hoc analysis from the STRENGTH trial that looks to pick up where the controversy over omega-3 fatty acid preparations left off at last year’s American Heart Association meeting.
A lack of benefit on CV event rates reported with Epanova, a high-dose combination of eicosapentaenoic acid (EPA) and docosahexaenoic acid, led to a contentious debate over how to reconcile STRENGTH with the findings from REDUCE-IT, which showed a 25% relative risk reduction in major CV events with the EPA product icosapent ethyl (Vascepa).
STRENGTH investigator Steven Nissen, MD, Cleveland Clinic, and REDUCE-IT investigator and session panelist Deepak Bhatt, MD, Brigham and Women’s Hospital, Boston, will share the virtual stage at ACC 2021, but Dr. Morris said the “good news” is both researchers know one another very well and “will really be focusing on no political issues, just the omega-3 fatty levels in the bloodstream and what does that mean in either trial.
“This is not designed to be a debate, point counterpoint,” she added.
For that, as all cardiologists and journalists know, there will be the wild and woolly #CardioTwitter sphere.
A version of this article first appeared on Medscape.com.