User login
Flu activity at ‘high’ level in South Carolina
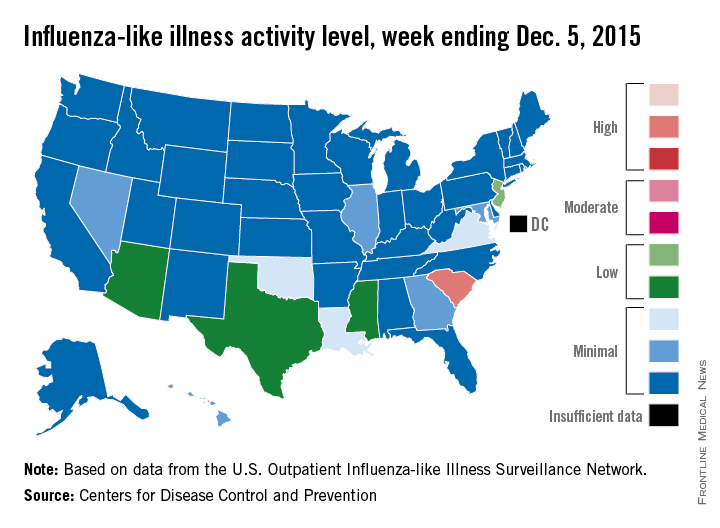
South Carolina officially became the first state to reach a “high” level of influenza activity during the 2015-2016 flu season, the Centers for Disease Control and Prevention reported Dec. 11.
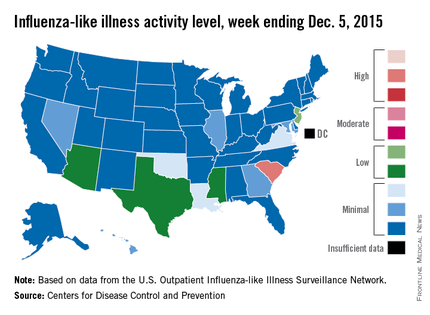
Activity of influenza-like illness (ILI) in the state was at level 9 on a scale of 1-10 for the week ending Dec. 5, 2015. For the country as a whole, however, activity was down a bit, with 13 states at level 2 or higher, compared with 20 states the week before. Among those with reduced ILI activity was Oklahoma, which went from level 6 for the week ending Nov. 28 to level 3 for the week ending Dec. 5, according to the CDC report.
For the week, 1.8% of patient visits reported to the U.S. Outpatient Influenza-like Illness Surveillance Network were for ILI, which is below the national baseline of 2.1%. This measure peaked at almost 6% during last year’s flu season, with that high coming at the end of Dec. 2014. The pattern was similar for the 2013-2014 season, with a peak of about 4.5% during the same week at the end of December, the report showed.
There was one influenza-related pediatric death for the week ending Dec. 5, bringing the total to three that have been reported for the season, the CDC said.
South Carolina officially became the first state to reach a “high” level of influenza activity during the 2015-2016 flu season, the Centers for Disease Control and Prevention reported Dec. 11.
Activity of influenza-like illness (ILI) in the state was at level 9 on a scale of 1-10 for the week ending Dec. 5, 2015. For the country as a whole, however, activity was down a bit, with 13 states at level 2 or higher, compared with 20 states the week before. Among those with reduced ILI activity was Oklahoma, which went from level 6 for the week ending Nov. 28 to level 3 for the week ending Dec. 5, according to the CDC report.

For the week, 1.8% of patient visits reported to the U.S. Outpatient Influenza-like Illness Surveillance Network were for ILI, which is below the national baseline of 2.1%. This measure peaked at almost 6% during last year’s flu season, with that high coming at the end of Dec. 2014. The pattern was similar for the 2013-2014 season, with a peak of about 4.5% during the same week at the end of December, the report showed.
There was one influenza-related pediatric death for the week ending Dec. 5, bringing the total to three that have been reported for the season, the CDC said.
South Carolina officially became the first state to reach a “high” level of influenza activity during the 2015-2016 flu season, the Centers for Disease Control and Prevention reported Dec. 11.
Activity of influenza-like illness (ILI) in the state was at level 9 on a scale of 1-10 for the week ending Dec. 5, 2015. For the country as a whole, however, activity was down a bit, with 13 states at level 2 or higher, compared with 20 states the week before. Among those with reduced ILI activity was Oklahoma, which went from level 6 for the week ending Nov. 28 to level 3 for the week ending Dec. 5, according to the CDC report.

For the week, 1.8% of patient visits reported to the U.S. Outpatient Influenza-like Illness Surveillance Network were for ILI, which is below the national baseline of 2.1%. This measure peaked at almost 6% during last year’s flu season, with that high coming at the end of Dec. 2014. The pattern was similar for the 2013-2014 season, with a peak of about 4.5% during the same week at the end of December, the report showed.
There was one influenza-related pediatric death for the week ending Dec. 5, bringing the total to three that have been reported for the season, the CDC said.
‘Moderate’ flu activity seen in two U.S. states
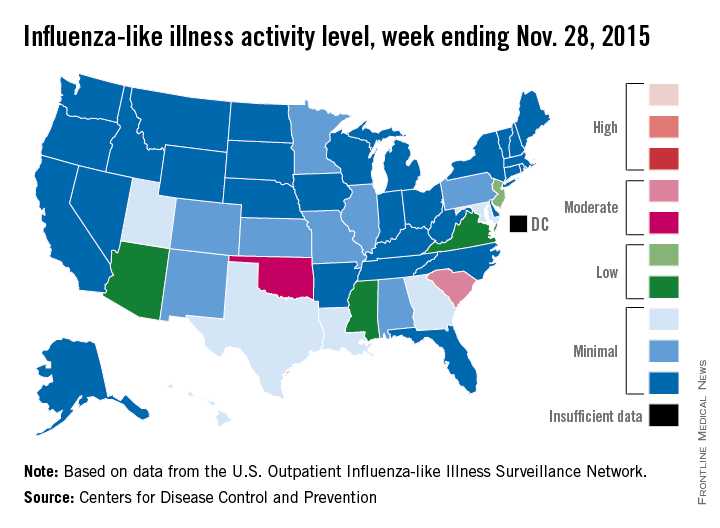
Two U.S. states experienced “moderate” activity of influenza-like illness for the week ending Nov. 28, 2015 – week 7 of the 2015-2016 flu season – the Centers for Disease Control and Prevention reported Dec. 4.
South Carolina had the highest (level 7) activity for the week, with Oklahoma joined by Puerto Rico at level 6. New Jersey was at the highest level (level 5) of “low” activity, while Arizona, Mississippi, and Virginia were a notch lower (level 4) but still in the “low” zone. All told, 20 states had flu activity of level 2 or higher, according to the CDC.
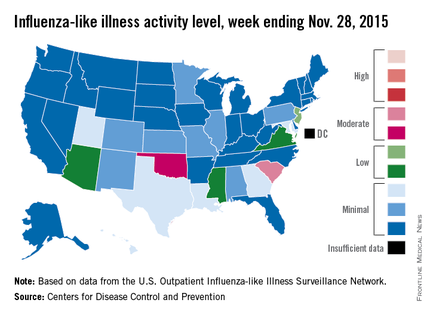
There were no influenza-associated pediatric deaths reported during the week, with two such deaths reported for the 2015-2016 season so far. For week 7 nationwide, 1.9% of patient visits reported through the U.S. Outpatient Influenza-like Illness Surveillance Network were the result of influenza-like illness – defined as a temperature of 100° F or greater and cough and/or sore throat – which is below the national baseline of 2.1%, the CDC said.
During week 7, 1.5% of the 11,288 specimens tested were positive for influenza, with 60% positive for influenza A and 40% positive for influenza B. For the season overall, 1.2% of the 102,675 specimens tested have been positive, with a 61/39 split for influenza A and B, the CDC noted.
Two U.S. states experienced “moderate” activity of influenza-like illness for the week ending Nov. 28, 2015 – week 7 of the 2015-2016 flu season – the Centers for Disease Control and Prevention reported Dec. 4.
South Carolina had the highest (level 7) activity for the week, with Oklahoma joined by Puerto Rico at level 6. New Jersey was at the highest level (level 5) of “low” activity, while Arizona, Mississippi, and Virginia were a notch lower (level 4) but still in the “low” zone. All told, 20 states had flu activity of level 2 or higher, according to the CDC.

There were no influenza-associated pediatric deaths reported during the week, with two such deaths reported for the 2015-2016 season so far. For week 7 nationwide, 1.9% of patient visits reported through the U.S. Outpatient Influenza-like Illness Surveillance Network were the result of influenza-like illness – defined as a temperature of 100° F or greater and cough and/or sore throat – which is below the national baseline of 2.1%, the CDC said.
During week 7, 1.5% of the 11,288 specimens tested were positive for influenza, with 60% positive for influenza A and 40% positive for influenza B. For the season overall, 1.2% of the 102,675 specimens tested have been positive, with a 61/39 split for influenza A and B, the CDC noted.
Two U.S. states experienced “moderate” activity of influenza-like illness for the week ending Nov. 28, 2015 – week 7 of the 2015-2016 flu season – the Centers for Disease Control and Prevention reported Dec. 4.
South Carolina had the highest (level 7) activity for the week, with Oklahoma joined by Puerto Rico at level 6. New Jersey was at the highest level (level 5) of “low” activity, while Arizona, Mississippi, and Virginia were a notch lower (level 4) but still in the “low” zone. All told, 20 states had flu activity of level 2 or higher, according to the CDC.

There were no influenza-associated pediatric deaths reported during the week, with two such deaths reported for the 2015-2016 season so far. For week 7 nationwide, 1.9% of patient visits reported through the U.S. Outpatient Influenza-like Illness Surveillance Network were the result of influenza-like illness – defined as a temperature of 100° F or greater and cough and/or sore throat – which is below the national baseline of 2.1%, the CDC said.
During week 7, 1.5% of the 11,288 specimens tested were positive for influenza, with 60% positive for influenza A and 40% positive for influenza B. For the season overall, 1.2% of the 102,675 specimens tested have been positive, with a 61/39 split for influenza A and B, the CDC noted.
Study: Exposure history critical to design of universal flu vaccine
In a study with implications for the development of new influenza vaccine strategies, researchers discovered that – among patients who received the 2009 H1N1 influenza vaccine – individuals with low levels of H1N1-specific antibodies prior to vaccination produced a more broadly protective immune response against the influenza virus than patients with high levels of H1N1-specific antibodies prior to vaccination.
A research team led by Patrick C. Wilson, Ph.D., of the Knapp Center for Lupus and Immunology Research at the University of Chicago, studied the B cell response in patients who received the pandemic 2009 H1N1 vaccine 2 years in a row and had varied histories of influenza exposure. All patients were 18 years or older, healthy, and had not received the yearly influenza vaccine prior to participating in the study. The researchers compared the patients’ “vaccine-induced plasmablast response upon first vaccination with the pandemic H1N1 strain in 2009-2010” with the patients’ plasmablast response upon revaccination with this same strain in 2010-2011 or 2011-2012. Each of the 21 study participants provided the researchers with at least four H1N1-specific plasmablasts.
The researchers “analyzed the immunoglobulin regions, strain specificity, and functional properties of the antibodies produced by this plasmablast population at the single-cell level across multiple years,” which allowed them to directly evaluate the effect of immune memory on the specificity of the current response to the virus.
Among the study’s findings was that “only individuals with low preexisting serological levels of pandemic H1N1 specific antibodies generated a broadly neutralizing plasmablast response directed toward the [hemagglutinin] stalk,” which is part of the hemagglutinin protein located on the surface of the influenza virus.
“[W]e demonstrate that the immune subdominance of the [hemagglutinin] stalk is a function of both the poor accessibility to the broadly protective epitopes and the inherent polyreactivity of the antibodies that can bind. We conclude that immunological memory profoundly shapes the viral epitopes targeted upon exposure with divergent influenza strains and determines the likelihood of generating a broadly protective response,” said Dr. Wilson and his coauthors. The authors reported no conflicts of interest.
Read the full study in Science Translational Medicine (doi: 10.1126/scitranslmed.aad0522).
In a study with implications for the development of new influenza vaccine strategies, researchers discovered that – among patients who received the 2009 H1N1 influenza vaccine – individuals with low levels of H1N1-specific antibodies prior to vaccination produced a more broadly protective immune response against the influenza virus than patients with high levels of H1N1-specific antibodies prior to vaccination.
A research team led by Patrick C. Wilson, Ph.D., of the Knapp Center for Lupus and Immunology Research at the University of Chicago, studied the B cell response in patients who received the pandemic 2009 H1N1 vaccine 2 years in a row and had varied histories of influenza exposure. All patients were 18 years or older, healthy, and had not received the yearly influenza vaccine prior to participating in the study. The researchers compared the patients’ “vaccine-induced plasmablast response upon first vaccination with the pandemic H1N1 strain in 2009-2010” with the patients’ plasmablast response upon revaccination with this same strain in 2010-2011 or 2011-2012. Each of the 21 study participants provided the researchers with at least four H1N1-specific plasmablasts.
The researchers “analyzed the immunoglobulin regions, strain specificity, and functional properties of the antibodies produced by this plasmablast population at the single-cell level across multiple years,” which allowed them to directly evaluate the effect of immune memory on the specificity of the current response to the virus.
Among the study’s findings was that “only individuals with low preexisting serological levels of pandemic H1N1 specific antibodies generated a broadly neutralizing plasmablast response directed toward the [hemagglutinin] stalk,” which is part of the hemagglutinin protein located on the surface of the influenza virus.
“[W]e demonstrate that the immune subdominance of the [hemagglutinin] stalk is a function of both the poor accessibility to the broadly protective epitopes and the inherent polyreactivity of the antibodies that can bind. We conclude that immunological memory profoundly shapes the viral epitopes targeted upon exposure with divergent influenza strains and determines the likelihood of generating a broadly protective response,” said Dr. Wilson and his coauthors. The authors reported no conflicts of interest.
Read the full study in Science Translational Medicine (doi: 10.1126/scitranslmed.aad0522).
In a study with implications for the development of new influenza vaccine strategies, researchers discovered that – among patients who received the 2009 H1N1 influenza vaccine – individuals with low levels of H1N1-specific antibodies prior to vaccination produced a more broadly protective immune response against the influenza virus than patients with high levels of H1N1-specific antibodies prior to vaccination.
A research team led by Patrick C. Wilson, Ph.D., of the Knapp Center for Lupus and Immunology Research at the University of Chicago, studied the B cell response in patients who received the pandemic 2009 H1N1 vaccine 2 years in a row and had varied histories of influenza exposure. All patients were 18 years or older, healthy, and had not received the yearly influenza vaccine prior to participating in the study. The researchers compared the patients’ “vaccine-induced plasmablast response upon first vaccination with the pandemic H1N1 strain in 2009-2010” with the patients’ plasmablast response upon revaccination with this same strain in 2010-2011 or 2011-2012. Each of the 21 study participants provided the researchers with at least four H1N1-specific plasmablasts.
The researchers “analyzed the immunoglobulin regions, strain specificity, and functional properties of the antibodies produced by this plasmablast population at the single-cell level across multiple years,” which allowed them to directly evaluate the effect of immune memory on the specificity of the current response to the virus.
Among the study’s findings was that “only individuals with low preexisting serological levels of pandemic H1N1 specific antibodies generated a broadly neutralizing plasmablast response directed toward the [hemagglutinin] stalk,” which is part of the hemagglutinin protein located on the surface of the influenza virus.
“[W]e demonstrate that the immune subdominance of the [hemagglutinin] stalk is a function of both the poor accessibility to the broadly protective epitopes and the inherent polyreactivity of the antibodies that can bind. We conclude that immunological memory profoundly shapes the viral epitopes targeted upon exposure with divergent influenza strains and determines the likelihood of generating a broadly protective response,” said Dr. Wilson and his coauthors. The authors reported no conflicts of interest.
Read the full study in Science Translational Medicine (doi: 10.1126/scitranslmed.aad0522).
FROM SCIENCE TRANSLATIONAL MEDICINE
U.S. flu activity increases slightly
Activity of influenza-like illness (ILI) “increased slightly in the United States” during week 5 of the 2015-2016 influenza season, the Centers for Disease Control and Prevention reported Nov. 20.
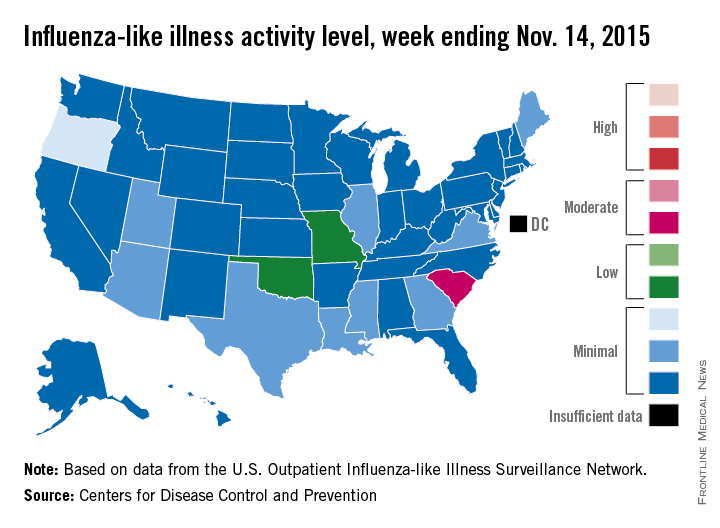
Thirteen states were above level-1 activity as of Nov. 14, 2015, compared with seven the week before. South Carolina jumped all the way up to “moderate” activity (level 6) and Missouri and Oklahoma moved into the low-activity category (level 4). Oregon remained at a still-minimal level 3, while Arizona, Georgia, Illinois, Louisiana, Maine, Mississippi, Texas, Utah, and Virginia are at level 2, according to the CDC.
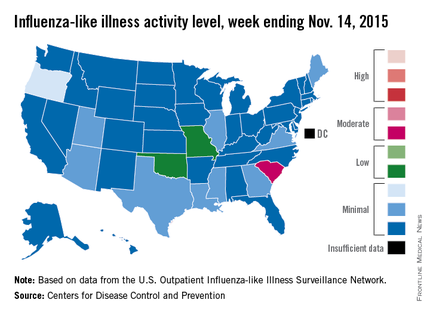
The first influenza-associated pediatric death was reported this week, although it actually occurred during week 4 (the week ending Nov. 7), the CDC said. There has been an average of 143 flu-associated pediatric deaths over the last three flu seasons.
ILI is defined as fever (temperature of 100° F or greater) and cough and/or sore throat. Activity level within a state is the proportion of outpatient visits to health care providers for influenza-like illness.
That proportion for the United States overall was 1.6%, which is up from last week’s 1.4% but still below the national baseline of 2.1%, the CDC said.
Activity of influenza-like illness (ILI) “increased slightly in the United States” during week 5 of the 2015-2016 influenza season, the Centers for Disease Control and Prevention reported Nov. 20.
Thirteen states were above level-1 activity as of Nov. 14, 2015, compared with seven the week before. South Carolina jumped all the way up to “moderate” activity (level 6) and Missouri and Oklahoma moved into the low-activity category (level 4). Oregon remained at a still-minimal level 3, while Arizona, Georgia, Illinois, Louisiana, Maine, Mississippi, Texas, Utah, and Virginia are at level 2, according to the CDC.

The first influenza-associated pediatric death was reported this week, although it actually occurred during week 4 (the week ending Nov. 7), the CDC said. There has been an average of 143 flu-associated pediatric deaths over the last three flu seasons.
ILI is defined as fever (temperature of 100° F or greater) and cough and/or sore throat. Activity level within a state is the proportion of outpatient visits to health care providers for influenza-like illness.
That proportion for the United States overall was 1.6%, which is up from last week’s 1.4% but still below the national baseline of 2.1%, the CDC said.
Activity of influenza-like illness (ILI) “increased slightly in the United States” during week 5 of the 2015-2016 influenza season, the Centers for Disease Control and Prevention reported Nov. 20.
Thirteen states were above level-1 activity as of Nov. 14, 2015, compared with seven the week before. South Carolina jumped all the way up to “moderate” activity (level 6) and Missouri and Oklahoma moved into the low-activity category (level 4). Oregon remained at a still-minimal level 3, while Arizona, Georgia, Illinois, Louisiana, Maine, Mississippi, Texas, Utah, and Virginia are at level 2, according to the CDC.

The first influenza-associated pediatric death was reported this week, although it actually occurred during week 4 (the week ending Nov. 7), the CDC said. There has been an average of 143 flu-associated pediatric deaths over the last three flu seasons.
ILI is defined as fever (temperature of 100° F or greater) and cough and/or sore throat. Activity level within a state is the proportion of outpatient visits to health care providers for influenza-like illness.
That proportion for the United States overall was 1.6%, which is up from last week’s 1.4% but still below the national baseline of 2.1%, the CDC said.
Prime-boost flu vaccination strategy effective in children
A prime-boost flu vaccination strategy proved effective in children and adolescents in an open-label phase III trial conducted by the vaccine manufacturer and reported online in the Pediatric Infectious Disease Journal.
This proof-of-concept result suggests that at the outbreak of the next flu pandemic, immediate priming with two doses of a subtype-matched flu vaccine, followed by boosting once a strain-matched vaccine becomes available, will offer the pediatric population better protection than simply waiting for adequate quantities of the strain-matched vaccine to be developed and marketed.
It is impossible to predict which specific viral strain will cause the next flu pandemic, but H5N1 is considered a likely candidate virus, “to the extent that vaccine manufacturers and regulatory authorities are actively developing H5N1 vaccines” for all age groups, said Dr. Patricia Izurieta of GlaxoSmithKline Vaccines, Belgium, and her associates.
Waiting for a pandemic to begin and then producing strain-specific vaccines could take as long as 6 months, and even then supplies probably would be limited. Experts have theorized that preemptive “priming” with already stockpiled H5N1 vaccines immediately after a pandemic begins may provide broad cross-protection that could mitigate the intensity of the subsequent pandemic, potentially reducing morbidity, mortality, and viral transmission.
To test this theory, Dr. Izurieta and her associates mimicked a potential flu pandemic by priming children and adolescents with two doses of an H5N1-AS03 vaccine, giving a booster dose of a specific H5N1 strain at 6 months, and assessing antibody response to all the vaccinations through 12 months. (Assessing actual vaccine efficacy was impossible because it would be unethical to determine this by exposing children to a flu virus.)
A total of 520 participants at a single medical center in the Philippines who were aged 3-18 years (mean age, 9 years) were assigned to two intervention groups and two control groups. Approximately half of these children and adolescents received the intervention – two priming doses of A/Indonesia/05/2005/H5N1-AS03B vaccine – while the control subjects received a single dose of hepatitis A vaccine. At day 182, half of the primed and half of the unprimed participants then received a booster dose of A/turkey/Turkey/01/2005-H5N1-As03B, while the remainder received the hepatitis A vaccine.
Compared with no priming, priming afforded superior seroconversion and putative seroprotection against the second, specific strain of H5N1. This robust protective effect was seen within 10 days after vaccination, occurred across all ages, and persisted through 12 months of follow-up, the investigators said (Ped Infect Dis J. 2015 Nov 6. [doi: 10.1097/INF.0000000000000968]).
Adverse events within 7 days of the priming vaccinations developed in 68%-76% of the intervention group, compared with only 37%-64% of the control group. Adverse events within 7 days of the booster vaccinations developed in 69% of the intervention group, compared with only 40% of the control group. The most common adverse events were upper respiratory tract infection (20% vs 14%), nasopharyngitis (4% vs 3%), and rhinitis (3% in both study groups). The most serious adverse events affected two children and were classified as grade 3.
The study results suggest that a prime-boost strategy can induce broad and long-lasting immunity and could be effectively employed in this age group in the prepandemic setting, Dr. Izurieta and her associates reported.
This trial was funded by GlaxoSmithKline, which also was involved in the design and conduct of the study, the data analysis, and the writing and publishing of the report. Dr. Izurieta and two of her associates are employed by GSK.
A prime-boost flu vaccination strategy proved effective in children and adolescents in an open-label phase III trial conducted by the vaccine manufacturer and reported online in the Pediatric Infectious Disease Journal.
This proof-of-concept result suggests that at the outbreak of the next flu pandemic, immediate priming with two doses of a subtype-matched flu vaccine, followed by boosting once a strain-matched vaccine becomes available, will offer the pediatric population better protection than simply waiting for adequate quantities of the strain-matched vaccine to be developed and marketed.
It is impossible to predict which specific viral strain will cause the next flu pandemic, but H5N1 is considered a likely candidate virus, “to the extent that vaccine manufacturers and regulatory authorities are actively developing H5N1 vaccines” for all age groups, said Dr. Patricia Izurieta of GlaxoSmithKline Vaccines, Belgium, and her associates.
Waiting for a pandemic to begin and then producing strain-specific vaccines could take as long as 6 months, and even then supplies probably would be limited. Experts have theorized that preemptive “priming” with already stockpiled H5N1 vaccines immediately after a pandemic begins may provide broad cross-protection that could mitigate the intensity of the subsequent pandemic, potentially reducing morbidity, mortality, and viral transmission.
To test this theory, Dr. Izurieta and her associates mimicked a potential flu pandemic by priming children and adolescents with two doses of an H5N1-AS03 vaccine, giving a booster dose of a specific H5N1 strain at 6 months, and assessing antibody response to all the vaccinations through 12 months. (Assessing actual vaccine efficacy was impossible because it would be unethical to determine this by exposing children to a flu virus.)
A total of 520 participants at a single medical center in the Philippines who were aged 3-18 years (mean age, 9 years) were assigned to two intervention groups and two control groups. Approximately half of these children and adolescents received the intervention – two priming doses of A/Indonesia/05/2005/H5N1-AS03B vaccine – while the control subjects received a single dose of hepatitis A vaccine. At day 182, half of the primed and half of the unprimed participants then received a booster dose of A/turkey/Turkey/01/2005-H5N1-As03B, while the remainder received the hepatitis A vaccine.
Compared with no priming, priming afforded superior seroconversion and putative seroprotection against the second, specific strain of H5N1. This robust protective effect was seen within 10 days after vaccination, occurred across all ages, and persisted through 12 months of follow-up, the investigators said (Ped Infect Dis J. 2015 Nov 6. [doi: 10.1097/INF.0000000000000968]).
Adverse events within 7 days of the priming vaccinations developed in 68%-76% of the intervention group, compared with only 37%-64% of the control group. Adverse events within 7 days of the booster vaccinations developed in 69% of the intervention group, compared with only 40% of the control group. The most common adverse events were upper respiratory tract infection (20% vs 14%), nasopharyngitis (4% vs 3%), and rhinitis (3% in both study groups). The most serious adverse events affected two children and were classified as grade 3.
The study results suggest that a prime-boost strategy can induce broad and long-lasting immunity and could be effectively employed in this age group in the prepandemic setting, Dr. Izurieta and her associates reported.
This trial was funded by GlaxoSmithKline, which also was involved in the design and conduct of the study, the data analysis, and the writing and publishing of the report. Dr. Izurieta and two of her associates are employed by GSK.
A prime-boost flu vaccination strategy proved effective in children and adolescents in an open-label phase III trial conducted by the vaccine manufacturer and reported online in the Pediatric Infectious Disease Journal.
This proof-of-concept result suggests that at the outbreak of the next flu pandemic, immediate priming with two doses of a subtype-matched flu vaccine, followed by boosting once a strain-matched vaccine becomes available, will offer the pediatric population better protection than simply waiting for adequate quantities of the strain-matched vaccine to be developed and marketed.
It is impossible to predict which specific viral strain will cause the next flu pandemic, but H5N1 is considered a likely candidate virus, “to the extent that vaccine manufacturers and regulatory authorities are actively developing H5N1 vaccines” for all age groups, said Dr. Patricia Izurieta of GlaxoSmithKline Vaccines, Belgium, and her associates.
Waiting for a pandemic to begin and then producing strain-specific vaccines could take as long as 6 months, and even then supplies probably would be limited. Experts have theorized that preemptive “priming” with already stockpiled H5N1 vaccines immediately after a pandemic begins may provide broad cross-protection that could mitigate the intensity of the subsequent pandemic, potentially reducing morbidity, mortality, and viral transmission.
To test this theory, Dr. Izurieta and her associates mimicked a potential flu pandemic by priming children and adolescents with two doses of an H5N1-AS03 vaccine, giving a booster dose of a specific H5N1 strain at 6 months, and assessing antibody response to all the vaccinations through 12 months. (Assessing actual vaccine efficacy was impossible because it would be unethical to determine this by exposing children to a flu virus.)
A total of 520 participants at a single medical center in the Philippines who were aged 3-18 years (mean age, 9 years) were assigned to two intervention groups and two control groups. Approximately half of these children and adolescents received the intervention – two priming doses of A/Indonesia/05/2005/H5N1-AS03B vaccine – while the control subjects received a single dose of hepatitis A vaccine. At day 182, half of the primed and half of the unprimed participants then received a booster dose of A/turkey/Turkey/01/2005-H5N1-As03B, while the remainder received the hepatitis A vaccine.
Compared with no priming, priming afforded superior seroconversion and putative seroprotection against the second, specific strain of H5N1. This robust protective effect was seen within 10 days after vaccination, occurred across all ages, and persisted through 12 months of follow-up, the investigators said (Ped Infect Dis J. 2015 Nov 6. [doi: 10.1097/INF.0000000000000968]).
Adverse events within 7 days of the priming vaccinations developed in 68%-76% of the intervention group, compared with only 37%-64% of the control group. Adverse events within 7 days of the booster vaccinations developed in 69% of the intervention group, compared with only 40% of the control group. The most common adverse events were upper respiratory tract infection (20% vs 14%), nasopharyngitis (4% vs 3%), and rhinitis (3% in both study groups). The most serious adverse events affected two children and were classified as grade 3.
The study results suggest that a prime-boost strategy can induce broad and long-lasting immunity and could be effectively employed in this age group in the prepandemic setting, Dr. Izurieta and her associates reported.
This trial was funded by GlaxoSmithKline, which also was involved in the design and conduct of the study, the data analysis, and the writing and publishing of the report. Dr. Izurieta and two of her associates are employed by GSK.
FROM THE PEDIATRIC INFECTIOUS DISEASE JOURNAL
Key clinical point: A prime-boost flu vaccination strategy was found effective in children and adolescents.
Major finding: Compared with no priming, priming afforded superior seroconversion and putative seroprotection against the second, specific strain of H5N1.
Data source: An industry-sponsored, single-center, open-label phase III trial involving 520 participants aged 3-18 years followed for 1 year.
Disclosures: This trial was funded by GlaxoSmithKline, which also was involved in the design and conduct of the study, the data analysis, and the writing and publishing of the report. Dr. Izurieta and two of her associates are employed by GSK.
U.S. influenza activity minimal so far
Four weeks into the 2015-2016 flu season, activity levels of influenza-like illness (ILI) are minimal in all 50 states, the Centers for Disease Control and Prevention reported Nov. 13.
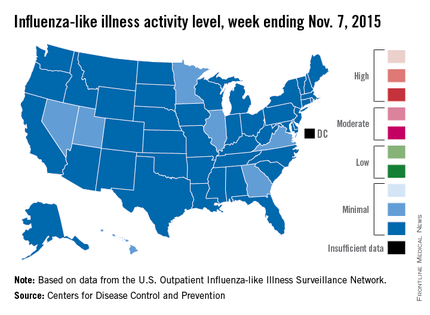
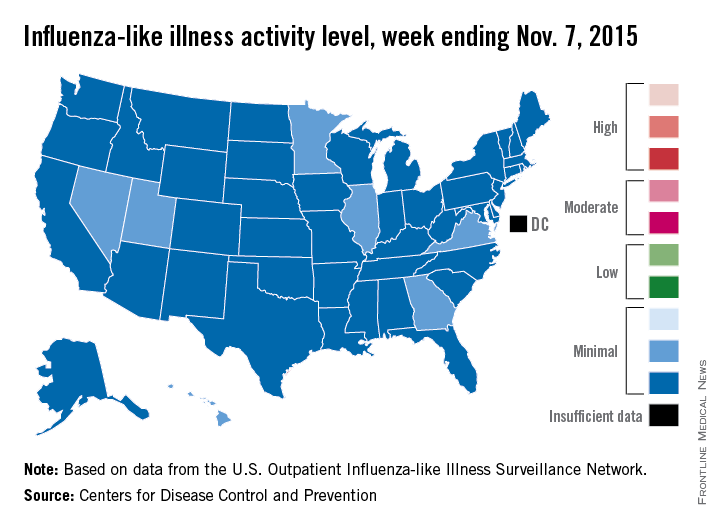
Seven states – Georgia, Hawaii, Illinois, Minnesota, Nevada, Utah, and Virginia – had level 2 activity for the week ending Nov. 7, 2015, which, although still minimal, was higher than the level 1 activity in the other 43 states. There was a moderate level of influenza-like illness activity (level 7) in Puerto Rico, which was up from level 6 the week before, and there were insufficient data to determine flu activity in the District of Columbia, according to the CDC.
ILI is defined as fever (temperature of 100° F or greater) and cough and/or sore throat. Activity level within a state is the proportion of outpatient visits to health care providers for influenza-like illness.
For the country overall, the proportion of outpatient visits for ILI was 1.4%, which is below the national baseline of 2.1% for week 4 of the flu season, the CDC noted.
Four weeks into the 2015-2016 flu season, activity levels of influenza-like illness (ILI) are minimal in all 50 states, the Centers for Disease Control and Prevention reported Nov. 13.

Seven states – Georgia, Hawaii, Illinois, Minnesota, Nevada, Utah, and Virginia – had level 2 activity for the week ending Nov. 7, 2015, which, although still minimal, was higher than the level 1 activity in the other 43 states. There was a moderate level of influenza-like illness activity (level 7) in Puerto Rico, which was up from level 6 the week before, and there were insufficient data to determine flu activity in the District of Columbia, according to the CDC.
ILI is defined as fever (temperature of 100° F or greater) and cough and/or sore throat. Activity level within a state is the proportion of outpatient visits to health care providers for influenza-like illness.
For the country overall, the proportion of outpatient visits for ILI was 1.4%, which is below the national baseline of 2.1% for week 4 of the flu season, the CDC noted.
Four weeks into the 2015-2016 flu season, activity levels of influenza-like illness (ILI) are minimal in all 50 states, the Centers for Disease Control and Prevention reported Nov. 13.

Seven states – Georgia, Hawaii, Illinois, Minnesota, Nevada, Utah, and Virginia – had level 2 activity for the week ending Nov. 7, 2015, which, although still minimal, was higher than the level 1 activity in the other 43 states. There was a moderate level of influenza-like illness activity (level 7) in Puerto Rico, which was up from level 6 the week before, and there were insufficient data to determine flu activity in the District of Columbia, according to the CDC.
ILI is defined as fever (temperature of 100° F or greater) and cough and/or sore throat. Activity level within a state is the proportion of outpatient visits to health care providers for influenza-like illness.
For the country overall, the proportion of outpatient visits for ILI was 1.4%, which is below the national baseline of 2.1% for week 4 of the flu season, the CDC noted.
CHEST: Oral solithromycin shows pneumonia pivotal-trial efficacy
MONTREAL – A new, next-generation macrolide, solithromycin, showed safety and efficacy as a once-daily oral agent that was noninferior to the comparator oral antibiotic, the fluoroquinolone moxifloxacin, in a phase III trial.
Macrolide resistance among strains of Streptococcus pneumoniae that cause many U.S. cases of severe community-acquired pneumonia has become common, complicating treatment of this common infection with a macrolide, Dr. Carlos M. Barrera explained at the annual meeting of the American College of Chest Physicians.
The SOLITAIRE-ORAL (Efficacy and Safety Study of Oral Solithromycin [CEM-101] Compared to Oral Moxifloxacin in Treatment of Patients With Community-Acquired Bacterial Pneumonia) trial enrolled 860 patients with moderate to moderately severe community-acquired pneumonia.
About half of the patients enrolled in the trial underwent microbiologic assessment of their infecting pathogen, and about 40% of cases in each treatment arm had infections caused by S. pneumoniae. In this subgroup, the 5-day regimen of solithromycin tested in the study succeeded in clearing the infection in 89% of patients, comparable to the 83% success rate achieved with a 7-day course of moxifloxacin (Avelox), said Dr. Barrera, a pulmonologist who practices in Miami.
The study’s primary endpoint for Food and Drug Administration approval of solithromycin was early clinical response, defined as an improvement in at least two listed symptoms at 72 hours after onset of treatment. That endpoint occurred in 78% of patients enrolled in each of the two arms of the study.
The data make solithromycin look like a promising way to once again have a macrolide available for empiric oral treatment of more severe community-acquired pneumonia, pending full peer review of the data, commented Dr. Muthiah P. Muthiah, a pulmonologist at the University of Tennessee Health Science Center in Memphis.
“A couple of decades ago, you could comfortably treat a patient with severe community-acquired pneumonia with a macrolide, but you can’t do that anymore,” Dr. Muthiah said in an interview.
If the newly reported data on oral solithromycin hold up under further review, it would mean that solithromycin was as effective as a potent quinolone, which remains an effective monotherapy for community-acquired pneumonia in patients who do not require treatment in an intensive care unit, Dr. Muthiah noted.
A companion study, SOLITAIRE-IV, is a phase III pivotal trial assessing the safety and efficacy of solithromycin when begun intravenously for treating community-acquired pneumonia, followed by a switch to oral dosing, in comparison with intravenous followed by oral treatment with moxifloxacin.
Once those data are fully collected and analyzed, the company will submit the information from both trials to the FDA, said Dr. David Oldach, chief medical officer for Cempra.
Results from the intravenous trial, reported in a preliminary way by Cempra Oct. 16 in a press release, showed that the solithromycin treatment regimen tested in SOLITAIRE-IV met its noninferiority targets, compared with moxifloxacin. The safety results, however, showed that solithromycin produced a higher number of patients with a liver-enzyme elevation, compared with patients treated with moxifloxacin.
In SOLITAIRE-IV, Cempra reported that grade 3 increase in levels of alanine transaminase (ALT) occurred in 8% of patients on solithromycin and in 3% of patients on moxifloxacin. Grade 4 increases in ALT occurred in less than 1% of patients in both treatment arms.
In the current, orally administered trial, grade 3 ALT increases occurred in 5% of patients treated with solithromycin and in 2% of patients treated with moxifloxacin, Dr. Barrera reported. Grade 4 ALT increases occurred in 0.5% of patients treated with solithromycin and in 1.2% of those treated with moxifloxacin. No patients in either arm developed an elevation of both ALT and bilirubin, and the ALT increases seen were reversible and asymptomatic, Dr. Barrera said.
By other assessments, the safety profiles of solithromycin and moxifloxacin were similar: 7% of patients on solithromycin and 6% on moxifloxacin had a serious adverse event, and 4% of patients in each study arm discontinued treatment because of an adverse event.
SOLITAIRE-ORAL was sponsored by Cempra, the company developing solithromycin. Dr. Barrera has received research funding from Cempra. Dr. Muthiah had no disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
MONTREAL – A new, next-generation macrolide, solithromycin, showed safety and efficacy as a once-daily oral agent that was noninferior to the comparator oral antibiotic, the fluoroquinolone moxifloxacin, in a phase III trial.
Macrolide resistance among strains of Streptococcus pneumoniae that cause many U.S. cases of severe community-acquired pneumonia has become common, complicating treatment of this common infection with a macrolide, Dr. Carlos M. Barrera explained at the annual meeting of the American College of Chest Physicians.
The SOLITAIRE-ORAL (Efficacy and Safety Study of Oral Solithromycin [CEM-101] Compared to Oral Moxifloxacin in Treatment of Patients With Community-Acquired Bacterial Pneumonia) trial enrolled 860 patients with moderate to moderately severe community-acquired pneumonia.
About half of the patients enrolled in the trial underwent microbiologic assessment of their infecting pathogen, and about 40% of cases in each treatment arm had infections caused by S. pneumoniae. In this subgroup, the 5-day regimen of solithromycin tested in the study succeeded in clearing the infection in 89% of patients, comparable to the 83% success rate achieved with a 7-day course of moxifloxacin (Avelox), said Dr. Barrera, a pulmonologist who practices in Miami.
The study’s primary endpoint for Food and Drug Administration approval of solithromycin was early clinical response, defined as an improvement in at least two listed symptoms at 72 hours after onset of treatment. That endpoint occurred in 78% of patients enrolled in each of the two arms of the study.
The data make solithromycin look like a promising way to once again have a macrolide available for empiric oral treatment of more severe community-acquired pneumonia, pending full peer review of the data, commented Dr. Muthiah P. Muthiah, a pulmonologist at the University of Tennessee Health Science Center in Memphis.
“A couple of decades ago, you could comfortably treat a patient with severe community-acquired pneumonia with a macrolide, but you can’t do that anymore,” Dr. Muthiah said in an interview.
If the newly reported data on oral solithromycin hold up under further review, it would mean that solithromycin was as effective as a potent quinolone, which remains an effective monotherapy for community-acquired pneumonia in patients who do not require treatment in an intensive care unit, Dr. Muthiah noted.
A companion study, SOLITAIRE-IV, is a phase III pivotal trial assessing the safety and efficacy of solithromycin when begun intravenously for treating community-acquired pneumonia, followed by a switch to oral dosing, in comparison with intravenous followed by oral treatment with moxifloxacin.
Once those data are fully collected and analyzed, the company will submit the information from both trials to the FDA, said Dr. David Oldach, chief medical officer for Cempra.
Results from the intravenous trial, reported in a preliminary way by Cempra Oct. 16 in a press release, showed that the solithromycin treatment regimen tested in SOLITAIRE-IV met its noninferiority targets, compared with moxifloxacin. The safety results, however, showed that solithromycin produced a higher number of patients with a liver-enzyme elevation, compared with patients treated with moxifloxacin.
In SOLITAIRE-IV, Cempra reported that grade 3 increase in levels of alanine transaminase (ALT) occurred in 8% of patients on solithromycin and in 3% of patients on moxifloxacin. Grade 4 increases in ALT occurred in less than 1% of patients in both treatment arms.
In the current, orally administered trial, grade 3 ALT increases occurred in 5% of patients treated with solithromycin and in 2% of patients treated with moxifloxacin, Dr. Barrera reported. Grade 4 ALT increases occurred in 0.5% of patients treated with solithromycin and in 1.2% of those treated with moxifloxacin. No patients in either arm developed an elevation of both ALT and bilirubin, and the ALT increases seen were reversible and asymptomatic, Dr. Barrera said.
By other assessments, the safety profiles of solithromycin and moxifloxacin were similar: 7% of patients on solithromycin and 6% on moxifloxacin had a serious adverse event, and 4% of patients in each study arm discontinued treatment because of an adverse event.
SOLITAIRE-ORAL was sponsored by Cempra, the company developing solithromycin. Dr. Barrera has received research funding from Cempra. Dr. Muthiah had no disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
MONTREAL – A new, next-generation macrolide, solithromycin, showed safety and efficacy as a once-daily oral agent that was noninferior to the comparator oral antibiotic, the fluoroquinolone moxifloxacin, in a phase III trial.
Macrolide resistance among strains of Streptococcus pneumoniae that cause many U.S. cases of severe community-acquired pneumonia has become common, complicating treatment of this common infection with a macrolide, Dr. Carlos M. Barrera explained at the annual meeting of the American College of Chest Physicians.
The SOLITAIRE-ORAL (Efficacy and Safety Study of Oral Solithromycin [CEM-101] Compared to Oral Moxifloxacin in Treatment of Patients With Community-Acquired Bacterial Pneumonia) trial enrolled 860 patients with moderate to moderately severe community-acquired pneumonia.
About half of the patients enrolled in the trial underwent microbiologic assessment of their infecting pathogen, and about 40% of cases in each treatment arm had infections caused by S. pneumoniae. In this subgroup, the 5-day regimen of solithromycin tested in the study succeeded in clearing the infection in 89% of patients, comparable to the 83% success rate achieved with a 7-day course of moxifloxacin (Avelox), said Dr. Barrera, a pulmonologist who practices in Miami.
The study’s primary endpoint for Food and Drug Administration approval of solithromycin was early clinical response, defined as an improvement in at least two listed symptoms at 72 hours after onset of treatment. That endpoint occurred in 78% of patients enrolled in each of the two arms of the study.
The data make solithromycin look like a promising way to once again have a macrolide available for empiric oral treatment of more severe community-acquired pneumonia, pending full peer review of the data, commented Dr. Muthiah P. Muthiah, a pulmonologist at the University of Tennessee Health Science Center in Memphis.
“A couple of decades ago, you could comfortably treat a patient with severe community-acquired pneumonia with a macrolide, but you can’t do that anymore,” Dr. Muthiah said in an interview.
If the newly reported data on oral solithromycin hold up under further review, it would mean that solithromycin was as effective as a potent quinolone, which remains an effective monotherapy for community-acquired pneumonia in patients who do not require treatment in an intensive care unit, Dr. Muthiah noted.
A companion study, SOLITAIRE-IV, is a phase III pivotal trial assessing the safety and efficacy of solithromycin when begun intravenously for treating community-acquired pneumonia, followed by a switch to oral dosing, in comparison with intravenous followed by oral treatment with moxifloxacin.
Once those data are fully collected and analyzed, the company will submit the information from both trials to the FDA, said Dr. David Oldach, chief medical officer for Cempra.
Results from the intravenous trial, reported in a preliminary way by Cempra Oct. 16 in a press release, showed that the solithromycin treatment regimen tested in SOLITAIRE-IV met its noninferiority targets, compared with moxifloxacin. The safety results, however, showed that solithromycin produced a higher number of patients with a liver-enzyme elevation, compared with patients treated with moxifloxacin.
In SOLITAIRE-IV, Cempra reported that grade 3 increase in levels of alanine transaminase (ALT) occurred in 8% of patients on solithromycin and in 3% of patients on moxifloxacin. Grade 4 increases in ALT occurred in less than 1% of patients in both treatment arms.
In the current, orally administered trial, grade 3 ALT increases occurred in 5% of patients treated with solithromycin and in 2% of patients treated with moxifloxacin, Dr. Barrera reported. Grade 4 ALT increases occurred in 0.5% of patients treated with solithromycin and in 1.2% of those treated with moxifloxacin. No patients in either arm developed an elevation of both ALT and bilirubin, and the ALT increases seen were reversible and asymptomatic, Dr. Barrera said.
By other assessments, the safety profiles of solithromycin and moxifloxacin were similar: 7% of patients on solithromycin and 6% on moxifloxacin had a serious adverse event, and 4% of patients in each study arm discontinued treatment because of an adverse event.
SOLITAIRE-ORAL was sponsored by Cempra, the company developing solithromycin. Dr. Barrera has received research funding from Cempra. Dr. Muthiah had no disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @mitchelzoler
AT CHEST 2015
Key clinical point: A next-generation, orally administered macrolide, solithromycin, showed efficacy that was noninferior to moxifloxacin against moderate to moderately severe community-acquired pneumonia in a phase III pivotal trial.
Major finding: Both solithromycin and moxifloxacin produced a 78% early clinical response rate, the study’s primary endpoint for Food and Drug Administration approval.
Data source: SOLITAIRE-ORAL, a multicenter, international phase III trial involving 860 patients with community-acquired pneumonia.
Disclosures: SOLITAIRE-ORAL was sponsored by Cempra, the company developing solithromycin. Dr. Barrera has received research funding from Cempra. Dr. Muthiah had no disclosures.
No flu vaccine for patients with egg allergy?
A 35-year-old woman with asthma presents for a follow-up visit in October. You recommend that she receive the influenza vaccine. She tells you that she cannot take the influenza vaccine because she is allergic to eggs.
What do you recommend?
A. Give her the influenza vaccine.
B. Give her an oseltamivir prescription, and have her start it if any flu-like symptoms appear.
C. Give her the nasal influenza vaccine.
D. Give her the cell-based influenza vaccine.
The clinic I work in asks all patients if they have allergy to eggs before giving the influenza vaccine. If the patient replies yes, then the vaccine is not given and the physician is consulted.
For many years, allergy to egg was considered a contraindication to receiving the influenza vaccine. This contraindication was based on the fear that administering a vaccine that was grown in eggs and could contain egg protein might cause anaphylaxis in patients with immunoglobulin E antibodies against egg proteins.
Fortunately, there is a good evidence base that shows that administering influenza vaccine to patients with egg allergy is safe.
This is extremely important information, because it is estimated that there are about 200,000-300,000 hospitalizations annually because of influenza. For the 2012-2013 influenza season, the CDC estimated that the flu vaccine prevented 6.6 million cases of influenza, 3.2 million doctor visits, and 79,000 hospitalizations. There were 170 pediatric deaths from the flu during the 2012-2013 influenza season (MMWR Morb Mortal Wkly Rep. 2013 Dec 13;62[49]:997-1000). The need for widespread vaccination is great, and decreasing the number of people unable to receive the vaccine is an important goal.
There are many studies in children and adults that show that those with egg allergy can be safely vaccinated with influenza vaccine. Dr. John M. James and colleagues reported a study of mostly children with egg allergy confirmed with skin testing (average age of the study group was 3 years) receiving influenza vaccine (J Pediatr. 1998 Nov;133[5]:624-8). A total of 83 patients with egg allergy received the vaccine (including 27 patients with a history of anaphylaxis or severe reactions after egg ingestion). No patients suffered severe reactions with the vaccine, with only four patients having mild, self-limited symptoms.
In another study, Dr. Anne Des Roches and colleagues performed a prospective, cohort study recruiting and vaccinating egg-allergic patients with trivalent inactivated influenza vaccine between 2010 and 2012 (J Allergy Clin Immunol. 2012 Nov;130[5]:1213-1216.e1). In the second year of the study, the focus was on recruiting patients with a history of anaphylaxis or severe cardiopulmonary symptoms upon egg ingestion. In addition, a retrospective study of all egg-allergic patients who had received an influenza vaccine between 2007 and 2010 was included.
A total of 457 doses of vaccine were administered to 367 patients with egg allergy, of whom 132 had a history of severe allergy. No patients developed anaphylaxis, and 13 patients developed mild allergiclike symptoms in the 24 hours after vaccination.
In an authoritative review on the subject of influenza vaccination in egg-allergic patients, Dr. John Kelso reported on 28 studies with a total of 4,315 patients with egg allergy, including 656 with history of anaphylaxis with egg ingestion (Expert Rev Vaccines. 2014 Aug;13[8]:1049-57). None of these patients developed a serious reaction when they received influenza vaccine.
Dr. Des Roches and colleagues reported on a prospective, cohort study in which 68 children with previous egg allergy received intranasal live attenuated influenza vaccine (J Allergy Clin Immunol Pract. 2015 Jan-Feb;3[1]:138-9). No patients had anaphylaxis or a severe allergic reaction. There were more adverse reactions in the patients with egg (7 patients) than in the control group (1 patient), but these were mild and nonspecific (abdominal pain, nasal congestion, headache, and cough).
The 2012 adverse reactions to vaccines practice parameter update recommended that patients with egg allergy should receive influenza vaccinations (trivalent influenza vaccine), because the risks of vaccinating are outweighed by the risks of not vaccinating (J Allergy Clin Immunol. 2012 Jul;130[1]:25-43).
A subsequent recommendation takes this a step further, recommending that all patients with egg allergy of any severity should receive inactivated influenza vaccine annually, using any age-approved brand (Ann Allergy Asthma Immunol. 2013 Oct;111[4]:301-2). In addition, there are no special waiting periods after vaccination of egg allergic patients beyond what is standard practice for any vaccine.
I think that we have plenty of evidence now to immunize all patients who report egg allergy, and to do so in the primary care setting.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at dpaauw@uw.edu.
A 35-year-old woman with asthma presents for a follow-up visit in October. You recommend that she receive the influenza vaccine. She tells you that she cannot take the influenza vaccine because she is allergic to eggs.
What do you recommend?
A. Give her the influenza vaccine.
B. Give her an oseltamivir prescription, and have her start it if any flu-like symptoms appear.
C. Give her the nasal influenza vaccine.
D. Give her the cell-based influenza vaccine.
The clinic I work in asks all patients if they have allergy to eggs before giving the influenza vaccine. If the patient replies yes, then the vaccine is not given and the physician is consulted.
For many years, allergy to egg was considered a contraindication to receiving the influenza vaccine. This contraindication was based on the fear that administering a vaccine that was grown in eggs and could contain egg protein might cause anaphylaxis in patients with immunoglobulin E antibodies against egg proteins.
Fortunately, there is a good evidence base that shows that administering influenza vaccine to patients with egg allergy is safe.
This is extremely important information, because it is estimated that there are about 200,000-300,000 hospitalizations annually because of influenza. For the 2012-2013 influenza season, the CDC estimated that the flu vaccine prevented 6.6 million cases of influenza, 3.2 million doctor visits, and 79,000 hospitalizations. There were 170 pediatric deaths from the flu during the 2012-2013 influenza season (MMWR Morb Mortal Wkly Rep. 2013 Dec 13;62[49]:997-1000). The need for widespread vaccination is great, and decreasing the number of people unable to receive the vaccine is an important goal.
There are many studies in children and adults that show that those with egg allergy can be safely vaccinated with influenza vaccine. Dr. John M. James and colleagues reported a study of mostly children with egg allergy confirmed with skin testing (average age of the study group was 3 years) receiving influenza vaccine (J Pediatr. 1998 Nov;133[5]:624-8). A total of 83 patients with egg allergy received the vaccine (including 27 patients with a history of anaphylaxis or severe reactions after egg ingestion). No patients suffered severe reactions with the vaccine, with only four patients having mild, self-limited symptoms.
In another study, Dr. Anne Des Roches and colleagues performed a prospective, cohort study recruiting and vaccinating egg-allergic patients with trivalent inactivated influenza vaccine between 2010 and 2012 (J Allergy Clin Immunol. 2012 Nov;130[5]:1213-1216.e1). In the second year of the study, the focus was on recruiting patients with a history of anaphylaxis or severe cardiopulmonary symptoms upon egg ingestion. In addition, a retrospective study of all egg-allergic patients who had received an influenza vaccine between 2007 and 2010 was included.
A total of 457 doses of vaccine were administered to 367 patients with egg allergy, of whom 132 had a history of severe allergy. No patients developed anaphylaxis, and 13 patients developed mild allergiclike symptoms in the 24 hours after vaccination.
In an authoritative review on the subject of influenza vaccination in egg-allergic patients, Dr. John Kelso reported on 28 studies with a total of 4,315 patients with egg allergy, including 656 with history of anaphylaxis with egg ingestion (Expert Rev Vaccines. 2014 Aug;13[8]:1049-57). None of these patients developed a serious reaction when they received influenza vaccine.
Dr. Des Roches and colleagues reported on a prospective, cohort study in which 68 children with previous egg allergy received intranasal live attenuated influenza vaccine (J Allergy Clin Immunol Pract. 2015 Jan-Feb;3[1]:138-9). No patients had anaphylaxis or a severe allergic reaction. There were more adverse reactions in the patients with egg (7 patients) than in the control group (1 patient), but these were mild and nonspecific (abdominal pain, nasal congestion, headache, and cough).
The 2012 adverse reactions to vaccines practice parameter update recommended that patients with egg allergy should receive influenza vaccinations (trivalent influenza vaccine), because the risks of vaccinating are outweighed by the risks of not vaccinating (J Allergy Clin Immunol. 2012 Jul;130[1]:25-43).
A subsequent recommendation takes this a step further, recommending that all patients with egg allergy of any severity should receive inactivated influenza vaccine annually, using any age-approved brand (Ann Allergy Asthma Immunol. 2013 Oct;111[4]:301-2). In addition, there are no special waiting periods after vaccination of egg allergic patients beyond what is standard practice for any vaccine.
I think that we have plenty of evidence now to immunize all patients who report egg allergy, and to do so in the primary care setting.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at dpaauw@uw.edu.
A 35-year-old woman with asthma presents for a follow-up visit in October. You recommend that she receive the influenza vaccine. She tells you that she cannot take the influenza vaccine because she is allergic to eggs.
What do you recommend?
A. Give her the influenza vaccine.
B. Give her an oseltamivir prescription, and have her start it if any flu-like symptoms appear.
C. Give her the nasal influenza vaccine.
D. Give her the cell-based influenza vaccine.
The clinic I work in asks all patients if they have allergy to eggs before giving the influenza vaccine. If the patient replies yes, then the vaccine is not given and the physician is consulted.
For many years, allergy to egg was considered a contraindication to receiving the influenza vaccine. This contraindication was based on the fear that administering a vaccine that was grown in eggs and could contain egg protein might cause anaphylaxis in patients with immunoglobulin E antibodies against egg proteins.
Fortunately, there is a good evidence base that shows that administering influenza vaccine to patients with egg allergy is safe.
This is extremely important information, because it is estimated that there are about 200,000-300,000 hospitalizations annually because of influenza. For the 2012-2013 influenza season, the CDC estimated that the flu vaccine prevented 6.6 million cases of influenza, 3.2 million doctor visits, and 79,000 hospitalizations. There were 170 pediatric deaths from the flu during the 2012-2013 influenza season (MMWR Morb Mortal Wkly Rep. 2013 Dec 13;62[49]:997-1000). The need for widespread vaccination is great, and decreasing the number of people unable to receive the vaccine is an important goal.
There are many studies in children and adults that show that those with egg allergy can be safely vaccinated with influenza vaccine. Dr. John M. James and colleagues reported a study of mostly children with egg allergy confirmed with skin testing (average age of the study group was 3 years) receiving influenza vaccine (J Pediatr. 1998 Nov;133[5]:624-8). A total of 83 patients with egg allergy received the vaccine (including 27 patients with a history of anaphylaxis or severe reactions after egg ingestion). No patients suffered severe reactions with the vaccine, with only four patients having mild, self-limited symptoms.
In another study, Dr. Anne Des Roches and colleagues performed a prospective, cohort study recruiting and vaccinating egg-allergic patients with trivalent inactivated influenza vaccine between 2010 and 2012 (J Allergy Clin Immunol. 2012 Nov;130[5]:1213-1216.e1). In the second year of the study, the focus was on recruiting patients with a history of anaphylaxis or severe cardiopulmonary symptoms upon egg ingestion. In addition, a retrospective study of all egg-allergic patients who had received an influenza vaccine between 2007 and 2010 was included.
A total of 457 doses of vaccine were administered to 367 patients with egg allergy, of whom 132 had a history of severe allergy. No patients developed anaphylaxis, and 13 patients developed mild allergiclike symptoms in the 24 hours after vaccination.
In an authoritative review on the subject of influenza vaccination in egg-allergic patients, Dr. John Kelso reported on 28 studies with a total of 4,315 patients with egg allergy, including 656 with history of anaphylaxis with egg ingestion (Expert Rev Vaccines. 2014 Aug;13[8]:1049-57). None of these patients developed a serious reaction when they received influenza vaccine.
Dr. Des Roches and colleagues reported on a prospective, cohort study in which 68 children with previous egg allergy received intranasal live attenuated influenza vaccine (J Allergy Clin Immunol Pract. 2015 Jan-Feb;3[1]:138-9). No patients had anaphylaxis or a severe allergic reaction. There were more adverse reactions in the patients with egg (7 patients) than in the control group (1 patient), but these were mild and nonspecific (abdominal pain, nasal congestion, headache, and cough).
The 2012 adverse reactions to vaccines practice parameter update recommended that patients with egg allergy should receive influenza vaccinations (trivalent influenza vaccine), because the risks of vaccinating are outweighed by the risks of not vaccinating (J Allergy Clin Immunol. 2012 Jul;130[1]:25-43).
A subsequent recommendation takes this a step further, recommending that all patients with egg allergy of any severity should receive inactivated influenza vaccine annually, using any age-approved brand (Ann Allergy Asthma Immunol. 2013 Oct;111[4]:301-2). In addition, there are no special waiting periods after vaccination of egg allergic patients beyond what is standard practice for any vaccine.
I think that we have plenty of evidence now to immunize all patients who report egg allergy, and to do so in the primary care setting.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at dpaauw@uw.edu.
VIDEO: Flu shot lowered hospitalization risk for influenza pneumonia
Getting a flu shot may be a highly effective method to prevent hospitalization for influenza-associated pneumonia.
That’s according to researchers who found that patients hospitalized with influenza-associated pneumonia were more likely to not have been vaccinated than patients whose pneumonia was due to other causes.
In video interviews, Dr. Kathryn M. Edwards and Dr. Carlos G. Grijalva of Vanderbilt University, Nashville, discussed their study of patients admitted through the emergency department for pneumonia and the benefits of flu vaccination in preventing hospitalization for influenza pneumonia.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Getting a flu shot may be a highly effective method to prevent hospitalization for influenza-associated pneumonia.
That’s according to researchers who found that patients hospitalized with influenza-associated pneumonia were more likely to not have been vaccinated than patients whose pneumonia was due to other causes.
In video interviews, Dr. Kathryn M. Edwards and Dr. Carlos G. Grijalva of Vanderbilt University, Nashville, discussed their study of patients admitted through the emergency department for pneumonia and the benefits of flu vaccination in preventing hospitalization for influenza pneumonia.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Getting a flu shot may be a highly effective method to prevent hospitalization for influenza-associated pneumonia.
That’s according to researchers who found that patients hospitalized with influenza-associated pneumonia were more likely to not have been vaccinated than patients whose pneumonia was due to other causes.
In video interviews, Dr. Kathryn M. Edwards and Dr. Carlos G. Grijalva of Vanderbilt University, Nashville, discussed their study of patients admitted through the emergency department for pneumonia and the benefits of flu vaccination in preventing hospitalization for influenza pneumonia.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
FROM JAMA
Peramivir effective against most flu viruses circulating globally
SAN DIEGO – The neuraminidase inhibitor peramivir inhibited about 99% of seasonal influenza A and B viruses circulating globally during the 2013-2014 and 2014-2015 influenza seasons, a large analysis demonstrated.
“The frequency of H1N1pdm09 viruses carrying neuraminidase (NA) H275Y remained low during both seasons; this mutation confers resistance to oseltamivir and peramivir,” said Margaret Okomo-Adhiambo, Ph.D., at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy. In addition, “a small proportion of viruses contained other neuraminidase changes that affect binding of peramivir to viral enzymes and may decrease virus susceptibility. These changes need to be closely monitored.”
Approved by the FDA in December of 2014, peramivir (Rapivab) is the only antiviral agent for influenza treatment to come to market in nearly 20 years. Approved for intravenous administration as a single dose, it is indicated for adults with acute uncomplicated influenza who may have trouble taking orally administered or inhaled neuraminidase (NA) inhibitors. Other NA inhibitors approved by the FDA for influenza infection include oseltamivir, which is orally administered, and zanamivir, which is inhaled.
For the current analysis, Dr. Okomo-Adhiambo of the influenza division at the Centers for Disease Control and Prevention, Atlanta, and her associates tested influenza virus susceptibility to peramivir during the 2013-2014 and 2014-2015 influenza seasons as part of the World Health Organization Global Influenza Surveillance and Response System. A total of 8,426 viruses were tested, 75% of which were circulating in the United States.
Dr. Okomo-Adhiambo reported that during the 2013-2014 and 2014-2015 influenza seasons, about 99% of influenza type A and B viruses were inhibited by peramivir, except for a few viruses belonging to subtype A(H1N1)pdm09 (1.5%), subtype A(H3N2) (0.2%), and type B (0.4%). In addition, NA activity of type A viruses was five to six times more sensitive to inhibition by peramivir, compared with type B NA.
She concluded her presentation by noting that studies “are needed to establish molecular markers of clinically relevant resistance to peramivir.”
The researchers reported having no financial disclosures.
SAN DIEGO – The neuraminidase inhibitor peramivir inhibited about 99% of seasonal influenza A and B viruses circulating globally during the 2013-2014 and 2014-2015 influenza seasons, a large analysis demonstrated.
“The frequency of H1N1pdm09 viruses carrying neuraminidase (NA) H275Y remained low during both seasons; this mutation confers resistance to oseltamivir and peramivir,” said Margaret Okomo-Adhiambo, Ph.D., at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy. In addition, “a small proportion of viruses contained other neuraminidase changes that affect binding of peramivir to viral enzymes and may decrease virus susceptibility. These changes need to be closely monitored.”
Approved by the FDA in December of 2014, peramivir (Rapivab) is the only antiviral agent for influenza treatment to come to market in nearly 20 years. Approved for intravenous administration as a single dose, it is indicated for adults with acute uncomplicated influenza who may have trouble taking orally administered or inhaled neuraminidase (NA) inhibitors. Other NA inhibitors approved by the FDA for influenza infection include oseltamivir, which is orally administered, and zanamivir, which is inhaled.
For the current analysis, Dr. Okomo-Adhiambo of the influenza division at the Centers for Disease Control and Prevention, Atlanta, and her associates tested influenza virus susceptibility to peramivir during the 2013-2014 and 2014-2015 influenza seasons as part of the World Health Organization Global Influenza Surveillance and Response System. A total of 8,426 viruses were tested, 75% of which were circulating in the United States.
Dr. Okomo-Adhiambo reported that during the 2013-2014 and 2014-2015 influenza seasons, about 99% of influenza type A and B viruses were inhibited by peramivir, except for a few viruses belonging to subtype A(H1N1)pdm09 (1.5%), subtype A(H3N2) (0.2%), and type B (0.4%). In addition, NA activity of type A viruses was five to six times more sensitive to inhibition by peramivir, compared with type B NA.
She concluded her presentation by noting that studies “are needed to establish molecular markers of clinically relevant resistance to peramivir.”
The researchers reported having no financial disclosures.
SAN DIEGO – The neuraminidase inhibitor peramivir inhibited about 99% of seasonal influenza A and B viruses circulating globally during the 2013-2014 and 2014-2015 influenza seasons, a large analysis demonstrated.
“The frequency of H1N1pdm09 viruses carrying neuraminidase (NA) H275Y remained low during both seasons; this mutation confers resistance to oseltamivir and peramivir,” said Margaret Okomo-Adhiambo, Ph.D., at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy. In addition, “a small proportion of viruses contained other neuraminidase changes that affect binding of peramivir to viral enzymes and may decrease virus susceptibility. These changes need to be closely monitored.”
Approved by the FDA in December of 2014, peramivir (Rapivab) is the only antiviral agent for influenza treatment to come to market in nearly 20 years. Approved for intravenous administration as a single dose, it is indicated for adults with acute uncomplicated influenza who may have trouble taking orally administered or inhaled neuraminidase (NA) inhibitors. Other NA inhibitors approved by the FDA for influenza infection include oseltamivir, which is orally administered, and zanamivir, which is inhaled.
For the current analysis, Dr. Okomo-Adhiambo of the influenza division at the Centers for Disease Control and Prevention, Atlanta, and her associates tested influenza virus susceptibility to peramivir during the 2013-2014 and 2014-2015 influenza seasons as part of the World Health Organization Global Influenza Surveillance and Response System. A total of 8,426 viruses were tested, 75% of which were circulating in the United States.
Dr. Okomo-Adhiambo reported that during the 2013-2014 and 2014-2015 influenza seasons, about 99% of influenza type A and B viruses were inhibited by peramivir, except for a few viruses belonging to subtype A(H1N1)pdm09 (1.5%), subtype A(H3N2) (0.2%), and type B (0.4%). In addition, NA activity of type A viruses was five to six times more sensitive to inhibition by peramivir, compared with type B NA.
She concluded her presentation by noting that studies “are needed to establish molecular markers of clinically relevant resistance to peramivir.”
The researchers reported having no financial disclosures.
AT ICAAC 2015
Key clinical point: Peramivir is potently effective against seasonal influenza viruses circulating globally.
Major finding: During the 2013-2014 and 2014-2015 influenza seasons, about 99% of influenza type A and B viruses were inhibited by peramivir.
Data source: An analysis of 8,426 influenza viruses that were tested during the 2013-2014 and 2014-2015 influenza seasons as part of the World Health Organization Global Influenza Surveillance and Response System.
Disclosures: The researchers reporting having no financial disclosures.