User login
GLP-1 Receptor Agonists in Endoscopy
Dear colleagues,
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) are revolutionizing the field of obesity management and are now common medication in patients presenting for endoscopy. With their effect on gastric emptying, the American Society of Anesthesiologists has recommended cessation of such agents prior to endoscopy. However, is this necessary in patients who have been on a clear liquid diet in preparation for a colonoscopy or who are undergoing moderate sedation? Additionally, there are risks to holding GLP-1 RAs, especially for those taking them for glycemic control.
In this issue of Perspectives, Dr. Thomas Hickey and Dr. Ryan Pouliot discuss the nuances of pre-procedure cessation from an anesthesiologist’s perspective. Dr. Jana Al Hashash provides a gastroenterologist’s view, also highlighting the current paucity of evidence guiding management strategies. We hope these pieces will help your discussions in managing GLP-1 RAs prior to endoscopy in your own practice. We welcome your thoughts on this issue on X @AGA_GIHN.
Gyanprakash A. Ketwaroo, MD, MSc, is associate professor of medicine, Yale University, New Haven, Connecticut, and chief of endoscopy at West Haven (Connecticut) VA Medical Center. He is an associate editor for GI & Hepatology News.
GLP-1 Receptor Agonists in Endoscopy
BY THOMAS R. HICKEY, MD; RYAN C. POULIOT, MD
In response to the recent dramatic increase in GLP-1 receptor agonist (GLP-1RA) prescribing and at the urging of its membership, the American Society of Anesthesiologists issued guidance on the preoperative management of these medications. The big takeaways were recommendations that patients on daily dosing should hold their dose on the day of a procedure, and that patients on weekly dosing should hold their dose a week prior.
The ASA guidance recognizes the sparse available evidence base and makes its recommendations in the spirit of patient safety, presuming that a more conservative approach will mitigate risk of rare but potentially devastating pulmonary aspiration, until prospective evidence informs the ideal approach. Until that approach is defined, whether more or less conservative, it is expected that anesthesiologists will adhere to their professional society’s recommendations.
Meanwhile, the American Gastroenterological Association Institute Rapid Clinical Practice Update (CPU) makes little distinction in the management of the endoscopy patient on GLP-1RA. A key refrain throughout the CPU is that there is no actionable data to justify the harms that may come to patients from stopping these medications (e.g., withdrawal of benefit to glycemic control and cardiovascular health) and in delaying or canceling procedures, which could lead to further stress on an overburdened workforce and add complexity to periprocedural processes.
Anesthesiologists should rightly consider themselves leaders in patient safety. As such, when a serious safety concern emerges they should be compelled to caution despite the possibility of other harms, until their concerns are mitigated by robust clinical evidence. Thankfully these questions are quite amenable to research, and prospective trials are already reporting compelling data that residual gastric contents, clearly a risk factor for aspiration, are increased in GLP-1RA groups compared to controls. This is evident even while following recommended fasting times and abstinences from these medications, and adjusting for confounders (e.g., age, diabetes, body mass index).1,2 It logically follows that large studies are likely to find an increased aspiration risk in GLP-1RA populations. Indeed, this increased risk has already been identified in a large retrospective study of endoscopy patients.3 These findings support the ASA’s caution. Additional data indicate that standard fasting guidelines in this patient population may be inadequate.4
The ASA guidance does not differentiate between patients undergoing surgery in the operating room and procedures in the endoscopy suite. Part of our task is to provide perspective on whether GLP-1RA management deserves different treatment for endoscopy patients. We can only speculate pending further data. For example, a prolonged fasting period including a full day of clears, with or without a bowel prep, intuitively protects against pulmonary aspiration. However, this is unlikely to mitigate an anesthesiologist’s concern that administration of propofol, frequently to a state of general anesthesia with an unsecured airway and resulting in a patient devoid of airway protection reflexes, is an inherently higher risk scenario for aspiration compared to surgery in the operating room with a secured airway. We also expect prospective trials will confirm retrospective findings that both propofol and procedures including upper endoscopy confer a higher risk for aspiration compared with conscious sedation and colonoscopy.3
We suggest a reasonable approach based on society guidance and existing evidence, pending additional data. Endoscopists and anesthesiologists should continue this important conversation with a specific focus on risks and benefits in order to decrease conflict and achieve consensus. If anesthesia care is desired, the patient instructions should be updated to reflect ASA guidance. Special attention should be paid to the “gray area,” for example those who did not hold the GLP-1 agonist as recommended.
This category of patients can be considered on a case-by-case basis by the anesthesiologist, proceduralist, and patient, with a range of options including: proceeding with endoscopist-directed sedation, proceeding with anesthesiology-administered conscious sedation, rescheduling the procedure, and proceeding with general anesthesia with rapid-sequence intubation. In addition to patient factors (e.g., GI symptoms, urgency of procedure), this consideration would vary based on local resources (e.g., presence or absence of anesthesia support staff, emergency airway equipment, nursing staff to comfort recovering patients after general endotracheal anesthesia), and aspiration risk inherent to the procedure (e.g., upper and or combination upper and lower endoscopy vs colonoscopy alone). Proficiency and availability of point-of-care ultrasound are rapidly increasing; adoption of a pre-procedure gastric ultrasound to assess for solids, thick liquids, or large volume of clear liquids may provide a less nuanced, more objective means to address this question.
While the question of periprocedural management of these medications has generated intense interest among anesthesiologists and endoscopists alike, it is worth noting the net positive health effects these drugs are likely to have on our patients, including improved glycemic control, significant weight loss, and decreased cardiovascular risk. We are eager to see whether these benefits translate into an overall improvement in periprocedural outcomes, including in our endoscopy patients.
Dr. Hickey is assistant professor of anesthesiology at the Yale University School of Medicine, New Haven, Connecticut, and the VA Connecticut Healthcare System. Dr. Pouliot is assistant professor of anesthesiology at the Geisel School of Medicine at Dartmouth, Hanover, New Hampshire, and Dartmouth-Hitchcock Medical Center, Lebanon, New Hampshire.
References
1. Sherwin M et al. Influence of semaglutide use on the presence of residual gastric solids on gastric ultrasound: A prospective observational study in volunteers without obesity recently started on semaglutide. Can J Anaesth. 2023 Aug. doi:10.1007/s12630-023-02549-5.
2. Wu F et al. Association of glucagon-like peptide receptor 1 agonist therapy with the presence of gastric contents in fasting patients undergoing endoscopy under anesthesia care: A historical cohort study. Can J Anaesth. 2024 Mar 14. doi:10.1007/s12630-024-02719-z.
3. Yeo YH et al. Increased risk of aspiration pneumonia associated with endoscopic procedures among patients with glucagon-like peptide 1 receptor agonist use. Gastroenterology. 2024 Mar 27. doi:10.1053/j.gastro.2024.03.015.
4. Sen S et al. Glucagon-like peptide-1 receptor agonist use and residual gastric content before anesthesia. JAMA Surg. 2024 Mar 6. doi:10.1001/jamasurg.2024.0111.
The Impact of GLP-1 Receptor Agonists On Endoscopy
BY JANA G. AL HASHASH, MD, MSc, AGAF
Glucagon-like peptide 1 receptor agonists (GLP-1 RAs) have been approved for the treatment of type 2 diabetes mellitus since 2005. They have become more widely used over the last couple of years for weight loss in individuals who suffer from adiposity-based chronic disease.
The remarkable positive effects that GLP-1 RAs have had on weight loss as well as other medical conditions such as heart disease, hypertension, metabolic dysfunction–associated steatotic liver disease, among many others, have gained these drugs more traction. Even in situations when insurance companies deny coverage of GLP-1 RAs, many patients have been resorting to other routes to obtain these medications, commonly by purchasing them from online compounding pharmacies.
As such, more and more of our patients who present to endoscopy suites across the country are on one of the available GLP-1 RAs. This has necessitated endoscopists and anesthesiologists to become more familiar with the impact of GLP-1 RAs on patients undergoing endoscopic procedures.
Similar to narcotics, GLP-1 RAs affect gastrointestinal motility and delay gastric emptying. Common side effects of patients receiving GLP-1 RAs include nausea, vomiting, and increased satiety. Patients on GLP-1 RAs for weight loss may also have other contributing risk factors for gastroparesis such as diabetes mellitus which may further delay gastric emptying.
For endoscopists, our goals are to achieve the highest quality examination in the safest way possible. As such, being on a GLP-1 RAs could compromise both goals; but to date, the exact impact of these drugs on exam quality and patient safety is yet to be determined.
Studies have shown that patients on GLP-1 RAs have increased gastric residue on upper endoscopy compared with patients not on GLP-1 RAs. The effect of this increased residue on aspiration risk and clinically meaningful patient outcomes is being investigated, and the available published data are conflicting. Additionally, other published cases have shown that GLP-1 RAs are associated with increased solid gastric residue but not liquids, and that symptoms of dyspepsia and abdominal bloating are associated with an increased probability of residual gastric content.
Given the valid concern for increased gastric content residue, anesthesia specialists became more strict about which GLP-1 RA users they would agree to sedate, which ones they would intubate, and which procedures they would cancel. As one would imagine, cancellation and intubation rates have been increasing, and these have affected the schedules of patients, their families, and physicians.
The concern with GLP-1 RAs does not only apply to upper endoscopies, but also impacts colonoscopies. In addition to the concerns of aspiration and pneumonia, studies have shown that the use of GLP-1 RAs may be associated with a lower quality of bowel preparation and higher need for repeat colonoscopy. A study, which I believe is critical, showed that patients on GLP-1 RAs who were scheduled for upper endoscopy and colonoscopy were found to have less gastric residue and less risk of complications when compared with patients who were only having an upper endoscopy. This study sets the stage for a modified prep for patients on GLP-1 RAs prior to their procedures, since patients who received a modified/extended liquid diet on the day prior to their procedure (those preparing for a colonoscopy), had a protective effect against retained gastric content.
Clearly, there is a knowledge gap and a need for guidance. In our recently published AGA Rapid CPU, we advised an individualized approach to managing patients on GLP-1 RAs in the pre-endoscopic setting. Factors to consider are the indication for the GLP-1 RAs, the dose being used, duration of use, and indication and urgency of the procedure, as well as the presence of symptoms in the preoperative area (i.e., do patients have any nausea, vomiting, dyspepsia, etc.). Also an important factor is the facility in which the endoscopy will be taking place, as certain centers have the capacity to act fast and prevent complications or address them in a timely manner while other centers may not be prepared.
We proposed that a modified liquid diet be considered in patients prior to their endoscopies by advising patients to adhere to a clear liquid diet the day before the procedure, as this may help decrease gastric residue and be the safest and best approach for patients on GLP-1 RAs. Of course, it is important to note that more prospective studies are needed to inform clinical practice, and until then, we will have to individualize our approach and continue to put patient safety first.
Dr. Al Hashash is a gastroenterologist and associate professor of medicine at Mayo Clinic, Jacksonville, Florida.
Dear colleagues,
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) are revolutionizing the field of obesity management and are now common medication in patients presenting for endoscopy. With their effect on gastric emptying, the American Society of Anesthesiologists has recommended cessation of such agents prior to endoscopy. However, is this necessary in patients who have been on a clear liquid diet in preparation for a colonoscopy or who are undergoing moderate sedation? Additionally, there are risks to holding GLP-1 RAs, especially for those taking them for glycemic control.
In this issue of Perspectives, Dr. Thomas Hickey and Dr. Ryan Pouliot discuss the nuances of pre-procedure cessation from an anesthesiologist’s perspective. Dr. Jana Al Hashash provides a gastroenterologist’s view, also highlighting the current paucity of evidence guiding management strategies. We hope these pieces will help your discussions in managing GLP-1 RAs prior to endoscopy in your own practice. We welcome your thoughts on this issue on X @AGA_GIHN.
Gyanprakash A. Ketwaroo, MD, MSc, is associate professor of medicine, Yale University, New Haven, Connecticut, and chief of endoscopy at West Haven (Connecticut) VA Medical Center. He is an associate editor for GI & Hepatology News.
GLP-1 Receptor Agonists in Endoscopy
BY THOMAS R. HICKEY, MD; RYAN C. POULIOT, MD
In response to the recent dramatic increase in GLP-1 receptor agonist (GLP-1RA) prescribing and at the urging of its membership, the American Society of Anesthesiologists issued guidance on the preoperative management of these medications. The big takeaways were recommendations that patients on daily dosing should hold their dose on the day of a procedure, and that patients on weekly dosing should hold their dose a week prior.
The ASA guidance recognizes the sparse available evidence base and makes its recommendations in the spirit of patient safety, presuming that a more conservative approach will mitigate risk of rare but potentially devastating pulmonary aspiration, until prospective evidence informs the ideal approach. Until that approach is defined, whether more or less conservative, it is expected that anesthesiologists will adhere to their professional society’s recommendations.
Meanwhile, the American Gastroenterological Association Institute Rapid Clinical Practice Update (CPU) makes little distinction in the management of the endoscopy patient on GLP-1RA. A key refrain throughout the CPU is that there is no actionable data to justify the harms that may come to patients from stopping these medications (e.g., withdrawal of benefit to glycemic control and cardiovascular health) and in delaying or canceling procedures, which could lead to further stress on an overburdened workforce and add complexity to periprocedural processes.
Anesthesiologists should rightly consider themselves leaders in patient safety. As such, when a serious safety concern emerges they should be compelled to caution despite the possibility of other harms, until their concerns are mitigated by robust clinical evidence. Thankfully these questions are quite amenable to research, and prospective trials are already reporting compelling data that residual gastric contents, clearly a risk factor for aspiration, are increased in GLP-1RA groups compared to controls. This is evident even while following recommended fasting times and abstinences from these medications, and adjusting for confounders (e.g., age, diabetes, body mass index).1,2 It logically follows that large studies are likely to find an increased aspiration risk in GLP-1RA populations. Indeed, this increased risk has already been identified in a large retrospective study of endoscopy patients.3 These findings support the ASA’s caution. Additional data indicate that standard fasting guidelines in this patient population may be inadequate.4
The ASA guidance does not differentiate between patients undergoing surgery in the operating room and procedures in the endoscopy suite. Part of our task is to provide perspective on whether GLP-1RA management deserves different treatment for endoscopy patients. We can only speculate pending further data. For example, a prolonged fasting period including a full day of clears, with or without a bowel prep, intuitively protects against pulmonary aspiration. However, this is unlikely to mitigate an anesthesiologist’s concern that administration of propofol, frequently to a state of general anesthesia with an unsecured airway and resulting in a patient devoid of airway protection reflexes, is an inherently higher risk scenario for aspiration compared to surgery in the operating room with a secured airway. We also expect prospective trials will confirm retrospective findings that both propofol and procedures including upper endoscopy confer a higher risk for aspiration compared with conscious sedation and colonoscopy.3
We suggest a reasonable approach based on society guidance and existing evidence, pending additional data. Endoscopists and anesthesiologists should continue this important conversation with a specific focus on risks and benefits in order to decrease conflict and achieve consensus. If anesthesia care is desired, the patient instructions should be updated to reflect ASA guidance. Special attention should be paid to the “gray area,” for example those who did not hold the GLP-1 agonist as recommended.
This category of patients can be considered on a case-by-case basis by the anesthesiologist, proceduralist, and patient, with a range of options including: proceeding with endoscopist-directed sedation, proceeding with anesthesiology-administered conscious sedation, rescheduling the procedure, and proceeding with general anesthesia with rapid-sequence intubation. In addition to patient factors (e.g., GI symptoms, urgency of procedure), this consideration would vary based on local resources (e.g., presence or absence of anesthesia support staff, emergency airway equipment, nursing staff to comfort recovering patients after general endotracheal anesthesia), and aspiration risk inherent to the procedure (e.g., upper and or combination upper and lower endoscopy vs colonoscopy alone). Proficiency and availability of point-of-care ultrasound are rapidly increasing; adoption of a pre-procedure gastric ultrasound to assess for solids, thick liquids, or large volume of clear liquids may provide a less nuanced, more objective means to address this question.
While the question of periprocedural management of these medications has generated intense interest among anesthesiologists and endoscopists alike, it is worth noting the net positive health effects these drugs are likely to have on our patients, including improved glycemic control, significant weight loss, and decreased cardiovascular risk. We are eager to see whether these benefits translate into an overall improvement in periprocedural outcomes, including in our endoscopy patients.
Dr. Hickey is assistant professor of anesthesiology at the Yale University School of Medicine, New Haven, Connecticut, and the VA Connecticut Healthcare System. Dr. Pouliot is assistant professor of anesthesiology at the Geisel School of Medicine at Dartmouth, Hanover, New Hampshire, and Dartmouth-Hitchcock Medical Center, Lebanon, New Hampshire.
References
1. Sherwin M et al. Influence of semaglutide use on the presence of residual gastric solids on gastric ultrasound: A prospective observational study in volunteers without obesity recently started on semaglutide. Can J Anaesth. 2023 Aug. doi:10.1007/s12630-023-02549-5.
2. Wu F et al. Association of glucagon-like peptide receptor 1 agonist therapy with the presence of gastric contents in fasting patients undergoing endoscopy under anesthesia care: A historical cohort study. Can J Anaesth. 2024 Mar 14. doi:10.1007/s12630-024-02719-z.
3. Yeo YH et al. Increased risk of aspiration pneumonia associated with endoscopic procedures among patients with glucagon-like peptide 1 receptor agonist use. Gastroenterology. 2024 Mar 27. doi:10.1053/j.gastro.2024.03.015.
4. Sen S et al. Glucagon-like peptide-1 receptor agonist use and residual gastric content before anesthesia. JAMA Surg. 2024 Mar 6. doi:10.1001/jamasurg.2024.0111.
The Impact of GLP-1 Receptor Agonists On Endoscopy
BY JANA G. AL HASHASH, MD, MSc, AGAF
Glucagon-like peptide 1 receptor agonists (GLP-1 RAs) have been approved for the treatment of type 2 diabetes mellitus since 2005. They have become more widely used over the last couple of years for weight loss in individuals who suffer from adiposity-based chronic disease.
The remarkable positive effects that GLP-1 RAs have had on weight loss as well as other medical conditions such as heart disease, hypertension, metabolic dysfunction–associated steatotic liver disease, among many others, have gained these drugs more traction. Even in situations when insurance companies deny coverage of GLP-1 RAs, many patients have been resorting to other routes to obtain these medications, commonly by purchasing them from online compounding pharmacies.
As such, more and more of our patients who present to endoscopy suites across the country are on one of the available GLP-1 RAs. This has necessitated endoscopists and anesthesiologists to become more familiar with the impact of GLP-1 RAs on patients undergoing endoscopic procedures.
Similar to narcotics, GLP-1 RAs affect gastrointestinal motility and delay gastric emptying. Common side effects of patients receiving GLP-1 RAs include nausea, vomiting, and increased satiety. Patients on GLP-1 RAs for weight loss may also have other contributing risk factors for gastroparesis such as diabetes mellitus which may further delay gastric emptying.
For endoscopists, our goals are to achieve the highest quality examination in the safest way possible. As such, being on a GLP-1 RAs could compromise both goals; but to date, the exact impact of these drugs on exam quality and patient safety is yet to be determined.
Studies have shown that patients on GLP-1 RAs have increased gastric residue on upper endoscopy compared with patients not on GLP-1 RAs. The effect of this increased residue on aspiration risk and clinically meaningful patient outcomes is being investigated, and the available published data are conflicting. Additionally, other published cases have shown that GLP-1 RAs are associated with increased solid gastric residue but not liquids, and that symptoms of dyspepsia and abdominal bloating are associated with an increased probability of residual gastric content.
Given the valid concern for increased gastric content residue, anesthesia specialists became more strict about which GLP-1 RA users they would agree to sedate, which ones they would intubate, and which procedures they would cancel. As one would imagine, cancellation and intubation rates have been increasing, and these have affected the schedules of patients, their families, and physicians.
The concern with GLP-1 RAs does not only apply to upper endoscopies, but also impacts colonoscopies. In addition to the concerns of aspiration and pneumonia, studies have shown that the use of GLP-1 RAs may be associated with a lower quality of bowel preparation and higher need for repeat colonoscopy. A study, which I believe is critical, showed that patients on GLP-1 RAs who were scheduled for upper endoscopy and colonoscopy were found to have less gastric residue and less risk of complications when compared with patients who were only having an upper endoscopy. This study sets the stage for a modified prep for patients on GLP-1 RAs prior to their procedures, since patients who received a modified/extended liquid diet on the day prior to their procedure (those preparing for a colonoscopy), had a protective effect against retained gastric content.
Clearly, there is a knowledge gap and a need for guidance. In our recently published AGA Rapid CPU, we advised an individualized approach to managing patients on GLP-1 RAs in the pre-endoscopic setting. Factors to consider are the indication for the GLP-1 RAs, the dose being used, duration of use, and indication and urgency of the procedure, as well as the presence of symptoms in the preoperative area (i.e., do patients have any nausea, vomiting, dyspepsia, etc.). Also an important factor is the facility in which the endoscopy will be taking place, as certain centers have the capacity to act fast and prevent complications or address them in a timely manner while other centers may not be prepared.
We proposed that a modified liquid diet be considered in patients prior to their endoscopies by advising patients to adhere to a clear liquid diet the day before the procedure, as this may help decrease gastric residue and be the safest and best approach for patients on GLP-1 RAs. Of course, it is important to note that more prospective studies are needed to inform clinical practice, and until then, we will have to individualize our approach and continue to put patient safety first.
Dr. Al Hashash is a gastroenterologist and associate professor of medicine at Mayo Clinic, Jacksonville, Florida.
Dear colleagues,
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) are revolutionizing the field of obesity management and are now common medication in patients presenting for endoscopy. With their effect on gastric emptying, the American Society of Anesthesiologists has recommended cessation of such agents prior to endoscopy. However, is this necessary in patients who have been on a clear liquid diet in preparation for a colonoscopy or who are undergoing moderate sedation? Additionally, there are risks to holding GLP-1 RAs, especially for those taking them for glycemic control.
In this issue of Perspectives, Dr. Thomas Hickey and Dr. Ryan Pouliot discuss the nuances of pre-procedure cessation from an anesthesiologist’s perspective. Dr. Jana Al Hashash provides a gastroenterologist’s view, also highlighting the current paucity of evidence guiding management strategies. We hope these pieces will help your discussions in managing GLP-1 RAs prior to endoscopy in your own practice. We welcome your thoughts on this issue on X @AGA_GIHN.
Gyanprakash A. Ketwaroo, MD, MSc, is associate professor of medicine, Yale University, New Haven, Connecticut, and chief of endoscopy at West Haven (Connecticut) VA Medical Center. He is an associate editor for GI & Hepatology News.
GLP-1 Receptor Agonists in Endoscopy
BY THOMAS R. HICKEY, MD; RYAN C. POULIOT, MD
In response to the recent dramatic increase in GLP-1 receptor agonist (GLP-1RA) prescribing and at the urging of its membership, the American Society of Anesthesiologists issued guidance on the preoperative management of these medications. The big takeaways were recommendations that patients on daily dosing should hold their dose on the day of a procedure, and that patients on weekly dosing should hold their dose a week prior.
The ASA guidance recognizes the sparse available evidence base and makes its recommendations in the spirit of patient safety, presuming that a more conservative approach will mitigate risk of rare but potentially devastating pulmonary aspiration, until prospective evidence informs the ideal approach. Until that approach is defined, whether more or less conservative, it is expected that anesthesiologists will adhere to their professional society’s recommendations.
Meanwhile, the American Gastroenterological Association Institute Rapid Clinical Practice Update (CPU) makes little distinction in the management of the endoscopy patient on GLP-1RA. A key refrain throughout the CPU is that there is no actionable data to justify the harms that may come to patients from stopping these medications (e.g., withdrawal of benefit to glycemic control and cardiovascular health) and in delaying or canceling procedures, which could lead to further stress on an overburdened workforce and add complexity to periprocedural processes.
Anesthesiologists should rightly consider themselves leaders in patient safety. As such, when a serious safety concern emerges they should be compelled to caution despite the possibility of other harms, until their concerns are mitigated by robust clinical evidence. Thankfully these questions are quite amenable to research, and prospective trials are already reporting compelling data that residual gastric contents, clearly a risk factor for aspiration, are increased in GLP-1RA groups compared to controls. This is evident even while following recommended fasting times and abstinences from these medications, and adjusting for confounders (e.g., age, diabetes, body mass index).1,2 It logically follows that large studies are likely to find an increased aspiration risk in GLP-1RA populations. Indeed, this increased risk has already been identified in a large retrospective study of endoscopy patients.3 These findings support the ASA’s caution. Additional data indicate that standard fasting guidelines in this patient population may be inadequate.4
The ASA guidance does not differentiate between patients undergoing surgery in the operating room and procedures in the endoscopy suite. Part of our task is to provide perspective on whether GLP-1RA management deserves different treatment for endoscopy patients. We can only speculate pending further data. For example, a prolonged fasting period including a full day of clears, with or without a bowel prep, intuitively protects against pulmonary aspiration. However, this is unlikely to mitigate an anesthesiologist’s concern that administration of propofol, frequently to a state of general anesthesia with an unsecured airway and resulting in a patient devoid of airway protection reflexes, is an inherently higher risk scenario for aspiration compared to surgery in the operating room with a secured airway. We also expect prospective trials will confirm retrospective findings that both propofol and procedures including upper endoscopy confer a higher risk for aspiration compared with conscious sedation and colonoscopy.3
We suggest a reasonable approach based on society guidance and existing evidence, pending additional data. Endoscopists and anesthesiologists should continue this important conversation with a specific focus on risks and benefits in order to decrease conflict and achieve consensus. If anesthesia care is desired, the patient instructions should be updated to reflect ASA guidance. Special attention should be paid to the “gray area,” for example those who did not hold the GLP-1 agonist as recommended.
This category of patients can be considered on a case-by-case basis by the anesthesiologist, proceduralist, and patient, with a range of options including: proceeding with endoscopist-directed sedation, proceeding with anesthesiology-administered conscious sedation, rescheduling the procedure, and proceeding with general anesthesia with rapid-sequence intubation. In addition to patient factors (e.g., GI symptoms, urgency of procedure), this consideration would vary based on local resources (e.g., presence or absence of anesthesia support staff, emergency airway equipment, nursing staff to comfort recovering patients after general endotracheal anesthesia), and aspiration risk inherent to the procedure (e.g., upper and or combination upper and lower endoscopy vs colonoscopy alone). Proficiency and availability of point-of-care ultrasound are rapidly increasing; adoption of a pre-procedure gastric ultrasound to assess for solids, thick liquids, or large volume of clear liquids may provide a less nuanced, more objective means to address this question.
While the question of periprocedural management of these medications has generated intense interest among anesthesiologists and endoscopists alike, it is worth noting the net positive health effects these drugs are likely to have on our patients, including improved glycemic control, significant weight loss, and decreased cardiovascular risk. We are eager to see whether these benefits translate into an overall improvement in periprocedural outcomes, including in our endoscopy patients.
Dr. Hickey is assistant professor of anesthesiology at the Yale University School of Medicine, New Haven, Connecticut, and the VA Connecticut Healthcare System. Dr. Pouliot is assistant professor of anesthesiology at the Geisel School of Medicine at Dartmouth, Hanover, New Hampshire, and Dartmouth-Hitchcock Medical Center, Lebanon, New Hampshire.
References
1. Sherwin M et al. Influence of semaglutide use on the presence of residual gastric solids on gastric ultrasound: A prospective observational study in volunteers without obesity recently started on semaglutide. Can J Anaesth. 2023 Aug. doi:10.1007/s12630-023-02549-5.
2. Wu F et al. Association of glucagon-like peptide receptor 1 agonist therapy with the presence of gastric contents in fasting patients undergoing endoscopy under anesthesia care: A historical cohort study. Can J Anaesth. 2024 Mar 14. doi:10.1007/s12630-024-02719-z.
3. Yeo YH et al. Increased risk of aspiration pneumonia associated with endoscopic procedures among patients with glucagon-like peptide 1 receptor agonist use. Gastroenterology. 2024 Mar 27. doi:10.1053/j.gastro.2024.03.015.
4. Sen S et al. Glucagon-like peptide-1 receptor agonist use and residual gastric content before anesthesia. JAMA Surg. 2024 Mar 6. doi:10.1001/jamasurg.2024.0111.
The Impact of GLP-1 Receptor Agonists On Endoscopy
BY JANA G. AL HASHASH, MD, MSc, AGAF
Glucagon-like peptide 1 receptor agonists (GLP-1 RAs) have been approved for the treatment of type 2 diabetes mellitus since 2005. They have become more widely used over the last couple of years for weight loss in individuals who suffer from adiposity-based chronic disease.
The remarkable positive effects that GLP-1 RAs have had on weight loss as well as other medical conditions such as heart disease, hypertension, metabolic dysfunction–associated steatotic liver disease, among many others, have gained these drugs more traction. Even in situations when insurance companies deny coverage of GLP-1 RAs, many patients have been resorting to other routes to obtain these medications, commonly by purchasing them from online compounding pharmacies.
As such, more and more of our patients who present to endoscopy suites across the country are on one of the available GLP-1 RAs. This has necessitated endoscopists and anesthesiologists to become more familiar with the impact of GLP-1 RAs on patients undergoing endoscopic procedures.
Similar to narcotics, GLP-1 RAs affect gastrointestinal motility and delay gastric emptying. Common side effects of patients receiving GLP-1 RAs include nausea, vomiting, and increased satiety. Patients on GLP-1 RAs for weight loss may also have other contributing risk factors for gastroparesis such as diabetes mellitus which may further delay gastric emptying.
For endoscopists, our goals are to achieve the highest quality examination in the safest way possible. As such, being on a GLP-1 RAs could compromise both goals; but to date, the exact impact of these drugs on exam quality and patient safety is yet to be determined.
Studies have shown that patients on GLP-1 RAs have increased gastric residue on upper endoscopy compared with patients not on GLP-1 RAs. The effect of this increased residue on aspiration risk and clinically meaningful patient outcomes is being investigated, and the available published data are conflicting. Additionally, other published cases have shown that GLP-1 RAs are associated with increased solid gastric residue but not liquids, and that symptoms of dyspepsia and abdominal bloating are associated with an increased probability of residual gastric content.
Given the valid concern for increased gastric content residue, anesthesia specialists became more strict about which GLP-1 RA users they would agree to sedate, which ones they would intubate, and which procedures they would cancel. As one would imagine, cancellation and intubation rates have been increasing, and these have affected the schedules of patients, their families, and physicians.
The concern with GLP-1 RAs does not only apply to upper endoscopies, but also impacts colonoscopies. In addition to the concerns of aspiration and pneumonia, studies have shown that the use of GLP-1 RAs may be associated with a lower quality of bowel preparation and higher need for repeat colonoscopy. A study, which I believe is critical, showed that patients on GLP-1 RAs who were scheduled for upper endoscopy and colonoscopy were found to have less gastric residue and less risk of complications when compared with patients who were only having an upper endoscopy. This study sets the stage for a modified prep for patients on GLP-1 RAs prior to their procedures, since patients who received a modified/extended liquid diet on the day prior to their procedure (those preparing for a colonoscopy), had a protective effect against retained gastric content.
Clearly, there is a knowledge gap and a need for guidance. In our recently published AGA Rapid CPU, we advised an individualized approach to managing patients on GLP-1 RAs in the pre-endoscopic setting. Factors to consider are the indication for the GLP-1 RAs, the dose being used, duration of use, and indication and urgency of the procedure, as well as the presence of symptoms in the preoperative area (i.e., do patients have any nausea, vomiting, dyspepsia, etc.). Also an important factor is the facility in which the endoscopy will be taking place, as certain centers have the capacity to act fast and prevent complications or address them in a timely manner while other centers may not be prepared.
We proposed that a modified liquid diet be considered in patients prior to their endoscopies by advising patients to adhere to a clear liquid diet the day before the procedure, as this may help decrease gastric residue and be the safest and best approach for patients on GLP-1 RAs. Of course, it is important to note that more prospective studies are needed to inform clinical practice, and until then, we will have to individualize our approach and continue to put patient safety first.
Dr. Al Hashash is a gastroenterologist and associate professor of medicine at Mayo Clinic, Jacksonville, Florida.
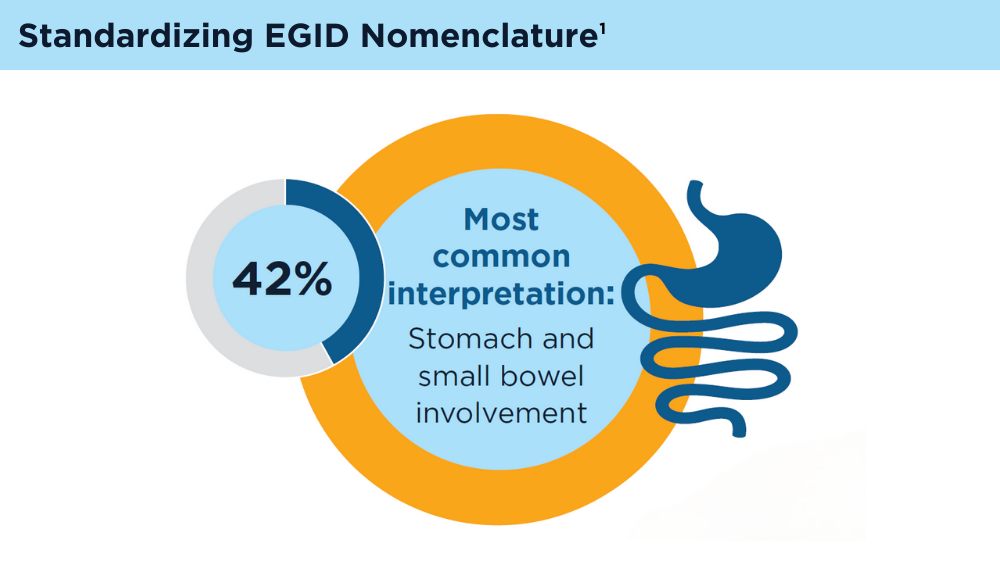
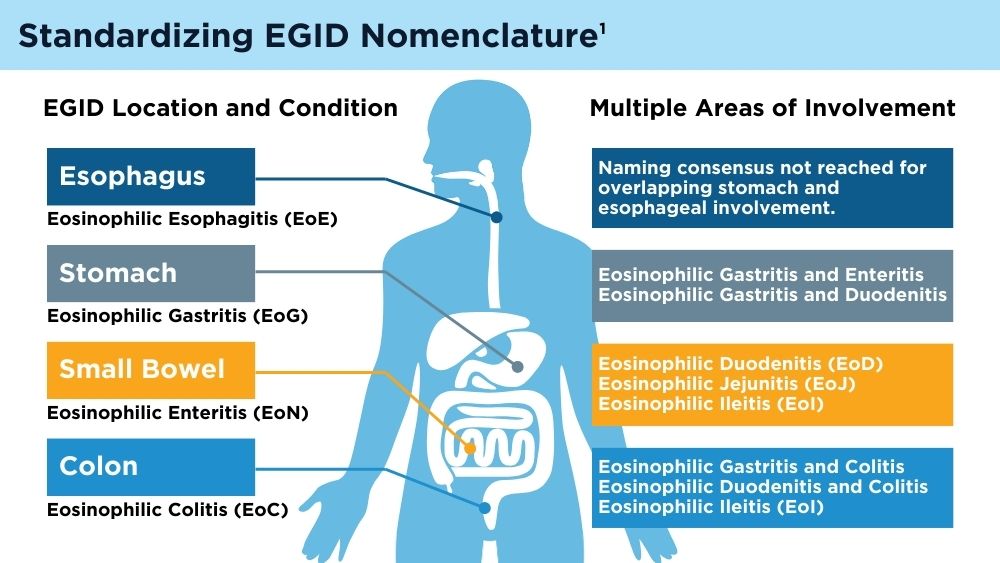
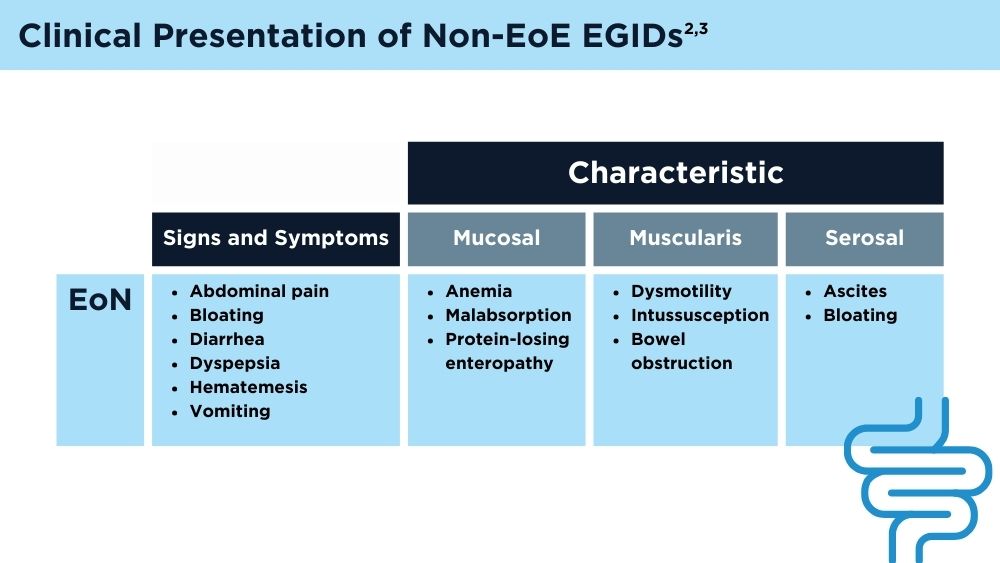
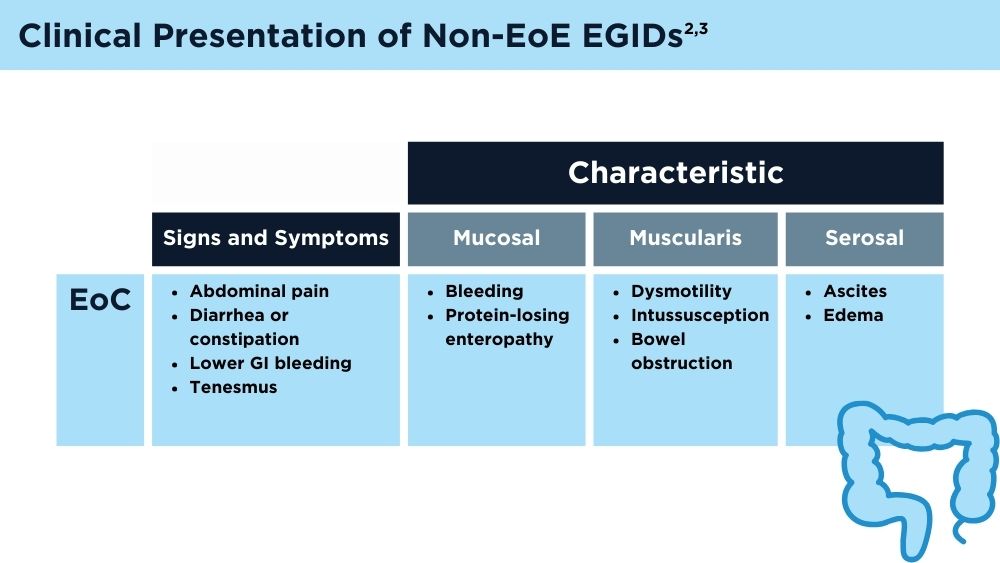
Eosinophilic Gastrointestinal Diseases: Beyond EoE
- Dellon ES, Gonsalves N, Abonia JP, et al. International consensus recommendations for eosinophilic gastrointestinal disease nomenclature. Clin Gastroenterol Hepatol. 2022;20(11):2474-2484.e3. doi:10.1016/j.cgh.2022.02.017
- Naramore S, Gupta SK. Nonesophageal eosinophilic gastrointestinal disorders: clinical care and future directions. J Pediatr Gastroenterol Nutr. 2018;67(3):318-321. doi:10.1097/MPG.0000000000002040
- Kinoshita Y, Sanuki T. Review of non-eosinophilic esophagitis-eosinophilic gastrointestinal disease (non-EoE-EGID) and a case series of twenty-eight affected patients. Biomolecules. 2023;13(9):1417. doi:10.3390/biom13091417
- Gonsalves N, Doerfler B, Zalewski A, et al. Prospective study of an amino acid-based elemental diet in an eosinophilic gastritis and gastroenteritis nutrition trial. J Allergy Clin Immunol. 2023;152(3):676-688. doi:10.1016/j.jaci.2023.05.024
- Oshima T. Biologic therapies targeting eosinophilic gastrointestinal diseases. Intern Med. 2023;62(23):3429-3430. doi:10.2169/internalmedicine.1911-23
- Pineton de Chambrun G, Gonzalez F, Canva JY, et al. Natural history of eosinophilic gastroenteritis. Clin Gastroenterol Hepatol. 2011;9(11):950-956.e1. doi:10.1016/j.cgh.2011.07.017
- Hirano I, Collins MH, King E, et al; CEGIR Investigators. Prospective endoscopic activity assessment for eosinophilic gastritis in a multi-site cohort. Am J Gastroenterol. 2022;117(3):413-423. doi:10.14309/ajg.0000000000001625
- Pesek RD, Reed CC, Muir AB, et al; Consortium of Eosinophilic Gastrointestinal Disease Researchers (CEGIR). Increasing rates of diagnosis, substantial co-occurrence, and variable treatment patterns of eosinophilic gastritis, gastroenteritis, and colitis based on 10-year data across a multicenter consortium. Am J Gastroenterol. 2019;114(6):984-994. doi:10.14309/ajg.0000000000000228
- Dellon ES, Gonsalves N, Abonia JP, et al. International consensus recommendations for eosinophilic gastrointestinal disease nomenclature. Clin Gastroenterol Hepatol. 2022;20(11):2474-2484.e3. doi:10.1016/j.cgh.2022.02.017
- Naramore S, Gupta SK. Nonesophageal eosinophilic gastrointestinal disorders: clinical care and future directions. J Pediatr Gastroenterol Nutr. 2018;67(3):318-321. doi:10.1097/MPG.0000000000002040
- Kinoshita Y, Sanuki T. Review of non-eosinophilic esophagitis-eosinophilic gastrointestinal disease (non-EoE-EGID) and a case series of twenty-eight affected patients. Biomolecules. 2023;13(9):1417. doi:10.3390/biom13091417
- Gonsalves N, Doerfler B, Zalewski A, et al. Prospective study of an amino acid-based elemental diet in an eosinophilic gastritis and gastroenteritis nutrition trial. J Allergy Clin Immunol. 2023;152(3):676-688. doi:10.1016/j.jaci.2023.05.024
- Oshima T. Biologic therapies targeting eosinophilic gastrointestinal diseases. Intern Med. 2023;62(23):3429-3430. doi:10.2169/internalmedicine.1911-23
- Pineton de Chambrun G, Gonzalez F, Canva JY, et al. Natural history of eosinophilic gastroenteritis. Clin Gastroenterol Hepatol. 2011;9(11):950-956.e1. doi:10.1016/j.cgh.2011.07.017
- Hirano I, Collins MH, King E, et al; CEGIR Investigators. Prospective endoscopic activity assessment for eosinophilic gastritis in a multi-site cohort. Am J Gastroenterol. 2022;117(3):413-423. doi:10.14309/ajg.0000000000001625
- Pesek RD, Reed CC, Muir AB, et al; Consortium of Eosinophilic Gastrointestinal Disease Researchers (CEGIR). Increasing rates of diagnosis, substantial co-occurrence, and variable treatment patterns of eosinophilic gastritis, gastroenteritis, and colitis based on 10-year data across a multicenter consortium. Am J Gastroenterol. 2019;114(6):984-994. doi:10.14309/ajg.0000000000000228
- Dellon ES, Gonsalves N, Abonia JP, et al. International consensus recommendations for eosinophilic gastrointestinal disease nomenclature. Clin Gastroenterol Hepatol. 2022;20(11):2474-2484.e3. doi:10.1016/j.cgh.2022.02.017
- Naramore S, Gupta SK. Nonesophageal eosinophilic gastrointestinal disorders: clinical care and future directions. J Pediatr Gastroenterol Nutr. 2018;67(3):318-321. doi:10.1097/MPG.0000000000002040
- Kinoshita Y, Sanuki T. Review of non-eosinophilic esophagitis-eosinophilic gastrointestinal disease (non-EoE-EGID) and a case series of twenty-eight affected patients. Biomolecules. 2023;13(9):1417. doi:10.3390/biom13091417
- Gonsalves N, Doerfler B, Zalewski A, et al. Prospective study of an amino acid-based elemental diet in an eosinophilic gastritis and gastroenteritis nutrition trial. J Allergy Clin Immunol. 2023;152(3):676-688. doi:10.1016/j.jaci.2023.05.024
- Oshima T. Biologic therapies targeting eosinophilic gastrointestinal diseases. Intern Med. 2023;62(23):3429-3430. doi:10.2169/internalmedicine.1911-23
- Pineton de Chambrun G, Gonzalez F, Canva JY, et al. Natural history of eosinophilic gastroenteritis. Clin Gastroenterol Hepatol. 2011;9(11):950-956.e1. doi:10.1016/j.cgh.2011.07.017
- Hirano I, Collins MH, King E, et al; CEGIR Investigators. Prospective endoscopic activity assessment for eosinophilic gastritis in a multi-site cohort. Am J Gastroenterol. 2022;117(3):413-423. doi:10.14309/ajg.0000000000001625
- Pesek RD, Reed CC, Muir AB, et al; Consortium of Eosinophilic Gastrointestinal Disease Researchers (CEGIR). Increasing rates of diagnosis, substantial co-occurrence, and variable treatment patterns of eosinophilic gastritis, gastroenteritis, and colitis based on 10-year data across a multicenter consortium. Am J Gastroenterol. 2019;114(6):984-994. doi:10.14309/ajg.0000000000000228
Healthy Sleep Linked to Lower Odds for Digestive Diseases
TOPLINE:
Healthier sleep is associated with lower odds of developing a wide range of gastrointestinal conditions, regardless of genetic susceptibility, new research revealed.
METHODOLOGY:
- Due to the widespread prevalence of sleep issues and a growing burden of digestive diseases globally, researchers investigated the association between sleep quality and digestive disorders in a prospective cohort study of 410,586 people in the UK Biobank.
- Five individual sleep behaviors were assessed: sleep duration, insomnia, snoring, daytime sleepiness, and chronotype.
- A healthy sleep was defined as a morning chronotype, 7-8 hours of sleep duration, no self-reported snoring, never or rare insomnia, and a low frequency of daytime sleepiness, for a score of 5/5.
- The study investigators tracked the development of 16 digestive diseases over a mean period of 13.2 years.
- As well as looking at healthy sleep scores, researchers considered genetic susceptibility to gastrointestinal conditions.
TAKEAWAY:
- Of the 16 digestive diseases looked at, the reduction of risk was highest for irritable bowel syndrome at 50% (HR, 0.50; 95% CI, 0.45-0.57).
- A healthy sleep score was also associated with 37% reduced odds for metabolic dysfunction–associated steatotic liver disease (formerly known as nonalcoholic fatty liver disease; HR, 0.63; 95% CI, 0.55-0.71), 35% lower chance for peptic ulcer (HR, 0.65; 95% CI, 0.058-0.74), 34% reduced chance for dyspepsia (HR, 0.66; 95% CI, 0.58-0.75), and a 25% lower risk for diverticulosis (HR, 0.75; 95% CI, 0.71-0.80).
- High genetic risk and poor sleep scores were also associated with increased odds (53% to > 200%) of developing digestive diseases.
- However, healthy sleep reduced the risk for digestive diseases regardless of genetic susceptibility.
IN PRACTICE:
“Our findings underscore the potential holistic impact of different sleep behaviors in mitigating the risk of digestive diseases in clinical practice,” wrote Shiyi Yu, MD, of Guangdong Provincial People’s Hospital, Guangzhou, Guangdong, China, and colleagues.
Poor sleep can also change our gut microbiome, Dr. Yu told this news organization. If you don’t sleep well, the repair of the gut lining cannot be finished during the night.
SOURCE:
The study was presented at the Digestive Disease Week® (DDW), 2024, annual meeting.
DISCLOSURES:
Dr. Yu had no relevant financial disclosures.
A version of this article appeared on Medscape.com.
TOPLINE:
Healthier sleep is associated with lower odds of developing a wide range of gastrointestinal conditions, regardless of genetic susceptibility, new research revealed.
METHODOLOGY:
- Due to the widespread prevalence of sleep issues and a growing burden of digestive diseases globally, researchers investigated the association between sleep quality and digestive disorders in a prospective cohort study of 410,586 people in the UK Biobank.
- Five individual sleep behaviors were assessed: sleep duration, insomnia, snoring, daytime sleepiness, and chronotype.
- A healthy sleep was defined as a morning chronotype, 7-8 hours of sleep duration, no self-reported snoring, never or rare insomnia, and a low frequency of daytime sleepiness, for a score of 5/5.
- The study investigators tracked the development of 16 digestive diseases over a mean period of 13.2 years.
- As well as looking at healthy sleep scores, researchers considered genetic susceptibility to gastrointestinal conditions.
TAKEAWAY:
- Of the 16 digestive diseases looked at, the reduction of risk was highest for irritable bowel syndrome at 50% (HR, 0.50; 95% CI, 0.45-0.57).
- A healthy sleep score was also associated with 37% reduced odds for metabolic dysfunction–associated steatotic liver disease (formerly known as nonalcoholic fatty liver disease; HR, 0.63; 95% CI, 0.55-0.71), 35% lower chance for peptic ulcer (HR, 0.65; 95% CI, 0.058-0.74), 34% reduced chance for dyspepsia (HR, 0.66; 95% CI, 0.58-0.75), and a 25% lower risk for diverticulosis (HR, 0.75; 95% CI, 0.71-0.80).
- High genetic risk and poor sleep scores were also associated with increased odds (53% to > 200%) of developing digestive diseases.
- However, healthy sleep reduced the risk for digestive diseases regardless of genetic susceptibility.
IN PRACTICE:
“Our findings underscore the potential holistic impact of different sleep behaviors in mitigating the risk of digestive diseases in clinical practice,” wrote Shiyi Yu, MD, of Guangdong Provincial People’s Hospital, Guangzhou, Guangdong, China, and colleagues.
Poor sleep can also change our gut microbiome, Dr. Yu told this news organization. If you don’t sleep well, the repair of the gut lining cannot be finished during the night.
SOURCE:
The study was presented at the Digestive Disease Week® (DDW), 2024, annual meeting.
DISCLOSURES:
Dr. Yu had no relevant financial disclosures.
A version of this article appeared on Medscape.com.
TOPLINE:
Healthier sleep is associated with lower odds of developing a wide range of gastrointestinal conditions, regardless of genetic susceptibility, new research revealed.
METHODOLOGY:
- Due to the widespread prevalence of sleep issues and a growing burden of digestive diseases globally, researchers investigated the association between sleep quality and digestive disorders in a prospective cohort study of 410,586 people in the UK Biobank.
- Five individual sleep behaviors were assessed: sleep duration, insomnia, snoring, daytime sleepiness, and chronotype.
- A healthy sleep was defined as a morning chronotype, 7-8 hours of sleep duration, no self-reported snoring, never or rare insomnia, and a low frequency of daytime sleepiness, for a score of 5/5.
- The study investigators tracked the development of 16 digestive diseases over a mean period of 13.2 years.
- As well as looking at healthy sleep scores, researchers considered genetic susceptibility to gastrointestinal conditions.
TAKEAWAY:
- Of the 16 digestive diseases looked at, the reduction of risk was highest for irritable bowel syndrome at 50% (HR, 0.50; 95% CI, 0.45-0.57).
- A healthy sleep score was also associated with 37% reduced odds for metabolic dysfunction–associated steatotic liver disease (formerly known as nonalcoholic fatty liver disease; HR, 0.63; 95% CI, 0.55-0.71), 35% lower chance for peptic ulcer (HR, 0.65; 95% CI, 0.058-0.74), 34% reduced chance for dyspepsia (HR, 0.66; 95% CI, 0.58-0.75), and a 25% lower risk for diverticulosis (HR, 0.75; 95% CI, 0.71-0.80).
- High genetic risk and poor sleep scores were also associated with increased odds (53% to > 200%) of developing digestive diseases.
- However, healthy sleep reduced the risk for digestive diseases regardless of genetic susceptibility.
IN PRACTICE:
“Our findings underscore the potential holistic impact of different sleep behaviors in mitigating the risk of digestive diseases in clinical practice,” wrote Shiyi Yu, MD, of Guangdong Provincial People’s Hospital, Guangzhou, Guangdong, China, and colleagues.
Poor sleep can also change our gut microbiome, Dr. Yu told this news organization. If you don’t sleep well, the repair of the gut lining cannot be finished during the night.
SOURCE:
The study was presented at the Digestive Disease Week® (DDW), 2024, annual meeting.
DISCLOSURES:
Dr. Yu had no relevant financial disclosures.
A version of this article appeared on Medscape.com.
Endoscopic Procedure Targets ‘Hunger Hormone’ for Weight Loss
WASHINGTON — .
“Patients reported a decrease in hunger, appetite, and cravings and an increase in control over [their] eating,” said senior study investigator Christopher McGowan, MD, AGAF, a gastroenterologist in private practice and medical director of True You Weight Loss in Cary, North Carolina.
“They generally described that their relationship with food had changed,” Dr. McGowan said at a May 8 press briefing during which his research (Abstract 516) was previewed for Digestive Disease Week® (DDW).
Researchers targeted the gastric fundus because its mucosal lining contains 80%-90% of the cells that produce ghrelin. When a person diets and/or loses weight, ghrelin levels increase, making the person hungrier and preventing sustained weight loss, Dr. McGowan said.
Previously, the only proven way to reduce ghrelin was to surgically remove or bypass the fundus. Weight-loss medications like Wegovy, Zepbound, and Ozempic target a different hormonal pathway, glucagon-like peptide 1 (GLP-1).
“What we’ve learned from the GLP-1 medications is the profound impact of reducing hunger,” Dr. McGowan said. “That’s what patients describe quite often — that it really changes their life and their quality of life. That’s really, really important.”
Major Findings
In the trial, 10 women (mean age, 38 years; mean body mass index, 40.2) underwent endoscopic fundic mucosal ablation via hybrid argon plasma coagulation in an ambulatory setting under general anesthesia from November 1, 2022, to April 14, 2023. The procedure took less than an hour on average, and the technique gave them easy access to the fundus, Dr. McGowan said.
Compared with baseline, there were multiple beneficial outcomes at 6 months:
- 45% less circulating ghrelin in the blood.
- 53% drop in ghrelin-producing cells in the fundus.
- 42% reduction in stomach capacity.
- 43% decrease in hunger, appetite, and cravings.
- 7.7% body weight loss.
Over the 6 months of the study, mean ghrelin concentrations dropped from 461.6 pg/mL at baseline to 254.8 pg/mL (P = .006).
It is fascinating that the hormone ghrelin decreased just by ablating, said Loren Laine, MD, AGAF, professor of medicine (digestive diseases) at Yale School of Medicine and chair of DDW 2024. “They used the same device that we use to treat bleeding ulcers or lesions in the stomach and applied it broadly over the whole upper part of the stomach.”
In a standard nutrient drink test, the maximum tolerated volume among participants dropped from a mean 27.3 oz at baseline to 15.8 oz at 6-month follow-up (P = .004).
Participants also completed three questionnaires. From baseline to 6 months, their DAILY EATS mean hunger score decreased from 6.2 to 4 (P = .002), mean Eating Drivers Index score dropped from 7 to 4 (P < .001), and WEL-SF score improved from 47.7 to 62.4 (P = .001).
Repeat endoscopy at 6 months showed that the gastric fundus contracted and healed. An unexpected and beneficial finding was fibrotic tissue, which made the fundus less able to expand, Dr. McGowan said. A smaller fundus “is critical for feeling full.”
No serious adverse events were reported. Participants described gas pressure, mild nausea, and cramping, all of which lasted 1-3 days, he said.
“The key here is preserving safety. This is why we use the technique of injecting a fluid cushion prior to ablating, so we’re not entering any deeper layers of the stomach,” Dr. McGowan said. “Importantly, there are no nerve receptors within the lining of the stomach, so there’s no pain from this procedure.”
Another Anti-Obesity Tool?
“We’re all familiar with the epidemic that is obesity affecting nearly one in two adults, and the profound impact that it has on patients’ health, their quality of life, as well as the healthcare system,” Dr. McGowan said. “It’s clear that we need every tool possible to address this because we know that obesity is not a matter of willpower. It’s a disease.”
Gastric fundus ablation “may represent, and frankly should represent, a treatment option for the greater than 100 million US adults with obesity,” he added.
Not every patient wants to or can access GLP-1 medications, Dr. McGowan said. Also, “there’s a difference between taking a medication long-term, requiring an injection every week, vs a single intervention in time that carries forward.”
Ablation could also help people transition after they stop GLP-1 medications to help them maintain their weight loss, he said.
Weight loss is the endpoint you care about the most, said Dr. Laine, who co-moderated the press briefing.
Though the weight loss of 7.7% was not a large percentage, it was only 10 patients. We will have to see whether the total body weight loss is different when they do the procedure in more patients or if they can combine different mechanisms, Dr. Laine said.
It remains unclear whether gastric fundus ablation would be a stand-alone procedure or used in combination with another endoscopic weight-management intervention, bariatric surgery, or medication.
The endoscopic sleeve, which is a stomach-reducing procedure, is very effective, but it doesn’t diminish hunger, Dr. McGowan said. Combining it with ablation may be “a best-of-both-worlds scenario.”
Dr. Laine added that another open question is whether the gastric fundal accommodation will be associated with any side effects such as dyspepsia.
Dr. McGowan reported consulting for Boston Scientific and Apollo Endosurgery. Dr. Laine reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
WASHINGTON — .
“Patients reported a decrease in hunger, appetite, and cravings and an increase in control over [their] eating,” said senior study investigator Christopher McGowan, MD, AGAF, a gastroenterologist in private practice and medical director of True You Weight Loss in Cary, North Carolina.
“They generally described that their relationship with food had changed,” Dr. McGowan said at a May 8 press briefing during which his research (Abstract 516) was previewed for Digestive Disease Week® (DDW).
Researchers targeted the gastric fundus because its mucosal lining contains 80%-90% of the cells that produce ghrelin. When a person diets and/or loses weight, ghrelin levels increase, making the person hungrier and preventing sustained weight loss, Dr. McGowan said.
Previously, the only proven way to reduce ghrelin was to surgically remove or bypass the fundus. Weight-loss medications like Wegovy, Zepbound, and Ozempic target a different hormonal pathway, glucagon-like peptide 1 (GLP-1).
“What we’ve learned from the GLP-1 medications is the profound impact of reducing hunger,” Dr. McGowan said. “That’s what patients describe quite often — that it really changes their life and their quality of life. That’s really, really important.”
Major Findings
In the trial, 10 women (mean age, 38 years; mean body mass index, 40.2) underwent endoscopic fundic mucosal ablation via hybrid argon plasma coagulation in an ambulatory setting under general anesthesia from November 1, 2022, to April 14, 2023. The procedure took less than an hour on average, and the technique gave them easy access to the fundus, Dr. McGowan said.
Compared with baseline, there were multiple beneficial outcomes at 6 months:
- 45% less circulating ghrelin in the blood.
- 53% drop in ghrelin-producing cells in the fundus.
- 42% reduction in stomach capacity.
- 43% decrease in hunger, appetite, and cravings.
- 7.7% body weight loss.
Over the 6 months of the study, mean ghrelin concentrations dropped from 461.6 pg/mL at baseline to 254.8 pg/mL (P = .006).
It is fascinating that the hormone ghrelin decreased just by ablating, said Loren Laine, MD, AGAF, professor of medicine (digestive diseases) at Yale School of Medicine and chair of DDW 2024. “They used the same device that we use to treat bleeding ulcers or lesions in the stomach and applied it broadly over the whole upper part of the stomach.”
In a standard nutrient drink test, the maximum tolerated volume among participants dropped from a mean 27.3 oz at baseline to 15.8 oz at 6-month follow-up (P = .004).
Participants also completed three questionnaires. From baseline to 6 months, their DAILY EATS mean hunger score decreased from 6.2 to 4 (P = .002), mean Eating Drivers Index score dropped from 7 to 4 (P < .001), and WEL-SF score improved from 47.7 to 62.4 (P = .001).
Repeat endoscopy at 6 months showed that the gastric fundus contracted and healed. An unexpected and beneficial finding was fibrotic tissue, which made the fundus less able to expand, Dr. McGowan said. A smaller fundus “is critical for feeling full.”
No serious adverse events were reported. Participants described gas pressure, mild nausea, and cramping, all of which lasted 1-3 days, he said.
“The key here is preserving safety. This is why we use the technique of injecting a fluid cushion prior to ablating, so we’re not entering any deeper layers of the stomach,” Dr. McGowan said. “Importantly, there are no nerve receptors within the lining of the stomach, so there’s no pain from this procedure.”
Another Anti-Obesity Tool?
“We’re all familiar with the epidemic that is obesity affecting nearly one in two adults, and the profound impact that it has on patients’ health, their quality of life, as well as the healthcare system,” Dr. McGowan said. “It’s clear that we need every tool possible to address this because we know that obesity is not a matter of willpower. It’s a disease.”
Gastric fundus ablation “may represent, and frankly should represent, a treatment option for the greater than 100 million US adults with obesity,” he added.
Not every patient wants to or can access GLP-1 medications, Dr. McGowan said. Also, “there’s a difference between taking a medication long-term, requiring an injection every week, vs a single intervention in time that carries forward.”
Ablation could also help people transition after they stop GLP-1 medications to help them maintain their weight loss, he said.
Weight loss is the endpoint you care about the most, said Dr. Laine, who co-moderated the press briefing.
Though the weight loss of 7.7% was not a large percentage, it was only 10 patients. We will have to see whether the total body weight loss is different when they do the procedure in more patients or if they can combine different mechanisms, Dr. Laine said.
It remains unclear whether gastric fundus ablation would be a stand-alone procedure or used in combination with another endoscopic weight-management intervention, bariatric surgery, or medication.
The endoscopic sleeve, which is a stomach-reducing procedure, is very effective, but it doesn’t diminish hunger, Dr. McGowan said. Combining it with ablation may be “a best-of-both-worlds scenario.”
Dr. Laine added that another open question is whether the gastric fundal accommodation will be associated with any side effects such as dyspepsia.
Dr. McGowan reported consulting for Boston Scientific and Apollo Endosurgery. Dr. Laine reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
WASHINGTON — .
“Patients reported a decrease in hunger, appetite, and cravings and an increase in control over [their] eating,” said senior study investigator Christopher McGowan, MD, AGAF, a gastroenterologist in private practice and medical director of True You Weight Loss in Cary, North Carolina.
“They generally described that their relationship with food had changed,” Dr. McGowan said at a May 8 press briefing during which his research (Abstract 516) was previewed for Digestive Disease Week® (DDW).
Researchers targeted the gastric fundus because its mucosal lining contains 80%-90% of the cells that produce ghrelin. When a person diets and/or loses weight, ghrelin levels increase, making the person hungrier and preventing sustained weight loss, Dr. McGowan said.
Previously, the only proven way to reduce ghrelin was to surgically remove or bypass the fundus. Weight-loss medications like Wegovy, Zepbound, and Ozempic target a different hormonal pathway, glucagon-like peptide 1 (GLP-1).
“What we’ve learned from the GLP-1 medications is the profound impact of reducing hunger,” Dr. McGowan said. “That’s what patients describe quite often — that it really changes their life and their quality of life. That’s really, really important.”
Major Findings
In the trial, 10 women (mean age, 38 years; mean body mass index, 40.2) underwent endoscopic fundic mucosal ablation via hybrid argon plasma coagulation in an ambulatory setting under general anesthesia from November 1, 2022, to April 14, 2023. The procedure took less than an hour on average, and the technique gave them easy access to the fundus, Dr. McGowan said.
Compared with baseline, there were multiple beneficial outcomes at 6 months:
- 45% less circulating ghrelin in the blood.
- 53% drop in ghrelin-producing cells in the fundus.
- 42% reduction in stomach capacity.
- 43% decrease in hunger, appetite, and cravings.
- 7.7% body weight loss.
Over the 6 months of the study, mean ghrelin concentrations dropped from 461.6 pg/mL at baseline to 254.8 pg/mL (P = .006).
It is fascinating that the hormone ghrelin decreased just by ablating, said Loren Laine, MD, AGAF, professor of medicine (digestive diseases) at Yale School of Medicine and chair of DDW 2024. “They used the same device that we use to treat bleeding ulcers or lesions in the stomach and applied it broadly over the whole upper part of the stomach.”
In a standard nutrient drink test, the maximum tolerated volume among participants dropped from a mean 27.3 oz at baseline to 15.8 oz at 6-month follow-up (P = .004).
Participants also completed three questionnaires. From baseline to 6 months, their DAILY EATS mean hunger score decreased from 6.2 to 4 (P = .002), mean Eating Drivers Index score dropped from 7 to 4 (P < .001), and WEL-SF score improved from 47.7 to 62.4 (P = .001).
Repeat endoscopy at 6 months showed that the gastric fundus contracted and healed. An unexpected and beneficial finding was fibrotic tissue, which made the fundus less able to expand, Dr. McGowan said. A smaller fundus “is critical for feeling full.”
No serious adverse events were reported. Participants described gas pressure, mild nausea, and cramping, all of which lasted 1-3 days, he said.
“The key here is preserving safety. This is why we use the technique of injecting a fluid cushion prior to ablating, so we’re not entering any deeper layers of the stomach,” Dr. McGowan said. “Importantly, there are no nerve receptors within the lining of the stomach, so there’s no pain from this procedure.”
Another Anti-Obesity Tool?
“We’re all familiar with the epidemic that is obesity affecting nearly one in two adults, and the profound impact that it has on patients’ health, their quality of life, as well as the healthcare system,” Dr. McGowan said. “It’s clear that we need every tool possible to address this because we know that obesity is not a matter of willpower. It’s a disease.”
Gastric fundus ablation “may represent, and frankly should represent, a treatment option for the greater than 100 million US adults with obesity,” he added.
Not every patient wants to or can access GLP-1 medications, Dr. McGowan said. Also, “there’s a difference between taking a medication long-term, requiring an injection every week, vs a single intervention in time that carries forward.”
Ablation could also help people transition after they stop GLP-1 medications to help them maintain their weight loss, he said.
Weight loss is the endpoint you care about the most, said Dr. Laine, who co-moderated the press briefing.
Though the weight loss of 7.7% was not a large percentage, it was only 10 patients. We will have to see whether the total body weight loss is different when they do the procedure in more patients or if they can combine different mechanisms, Dr. Laine said.
It remains unclear whether gastric fundus ablation would be a stand-alone procedure or used in combination with another endoscopic weight-management intervention, bariatric surgery, or medication.
The endoscopic sleeve, which is a stomach-reducing procedure, is very effective, but it doesn’t diminish hunger, Dr. McGowan said. Combining it with ablation may be “a best-of-both-worlds scenario.”
Dr. Laine added that another open question is whether the gastric fundal accommodation will be associated with any side effects such as dyspepsia.
Dr. McGowan reported consulting for Boston Scientific and Apollo Endosurgery. Dr. Laine reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM DDW 2024
AGA Clinical Guideline Stresses Patient Preferences in Barrett’s Treatment
Published in Gastroenterology , the clinical practice guideline makes five main recommendations — one strong and four conditional — based on very low to moderate evidence. It also stresses that providers should practice shared decision making according to patient preferences and risk perception.
For the most part, the new guideline is not a significant departure from the way expert endoscopists are currently practicing EET for BE and related neoplasia, gastroenterologist Joel H. Rubenstein, MD, MSc, AGAF, of the Barrett’s Esophagus Program in the Division of Gastroenterology at University of Michigan Medical School at Ann Arbor, said in an interview. One of three first authors of the guideline, Dr. Rubenstein added, “There is, however, considerable variability in how endoscopists practice, and we hope this guidance will serve as a useful resource to refer to for best practices.”
Added gastroenterologist Tarek Sawas, MD, MPH, assistant professor of internal medicine at UT Southwestern Medical Center in Dallas, “We hope the update will provide some clarity for practice and for implementation, while allowing gastroenterologists the freedom to decide what is best for patients based on lesion characteristics.”
Dr. Sawas added that one of the differences in the new guideline relates to the approach to low-grade dysplasia. While earlier guidance favored treatment over surveillance, patient preferences should now be factored into management. “Some patients are risk-averse and prefer to wait and watch, while others place more value on treatment and just want to get on with it,” he said.
When this guideline was circulated for public comment, “the areas prompting the most feedback was on our current suggestions against the routine use of EET in non-dysplastic BE and for the use of either endoscopic mucosal resection [EMR] or endoscopic submucosal dissection [ESD] for resection — with the expectation that the vast majority may be managed with EMR,” Dr. Rubenstein said.
“We felt that ESD would work best for larger lesions,” explained Dr. Sawas. “There aren’t a lot data in this area, just some observational studies, but we should have more data for comparison in the next few years.”
The incidence of esophageal adenocarcinoma continues to rise and an update was deemed in order since the AGA’s last formal guidance on this subject using the systematic GRADE (Grading of Recommendations Assessment, Development, and Evaluation) methodology was issued in 2011. “In the following time span, there’s been a lot of research, particularly with regard to management of low-grade dysplasia and endoscopic resection techniques,” Dr. Rubenstein said.
Key Recommendations
The 14 guideline panelists made the following suggestions for treatment and implementation based on different levels of certainty of evidence (CoE):
1. If high-grade dysplasia (HGD) is present, EET is recommended over surveillance, with subsequent surveillance performed at 3, 6, and 12 months, and annually thereafter. (Strong recommendation, moderate CoE).
Surveillance endoscopies should obtain targeted tissue samples of visible lesions and random biopsies of the cardia and distal 2 cm of the tubular esophagus.
2. In patients with low-grade dysplasia, EET is also preferred to surveillance. But for those placing a higher value on the certain harms and a lower value on the uncertain benefits of EET for reducing mortality, surveillance endoscopy is a reasonable option. (Conditional recommendation, low CoE).
Following EET, clinicians should perform surveillance at years 1 and 3 after complete eradication of intestinal metaplasia, then revert to the surveillance intervals used in non-dysplastic BE.
3. For non-dysplastic BE, the AGA advises against the routine use of EET. (Conditional recommendation, low CoE).
4. Patients undergoing EET should have resection of visible lesions followed by ablation of the remaining BE segment rather than resection of the entire segment.
In patients with only a small area of BE beyond the visible lesion, endoscopic resection is acceptable and may be preferred over repeated ablation. Radiofrequency ablation is the preferred ablative modality. (Conditional recommendation, very low CoE).
5. For treating visible neoplastic lesions the AGA suggests either EMR or ESD based on lesion characteristics. (Conditional recommendation, very low CoE).
Patients with suspected T1 esophageal adenocarcinoma (EAC) should be considered for EET. Endoscopic resection is recommended over endoscopic ultrasound for distinguishing EAC from HGD and for staging depth of invasion.
The vast majority of neoplastic lesions may be managed with EMR rather than ESD. Patients who have bulky lesions, or lesions highly suspicious of at least T1b invasion and are deemed candidates for endoscopic resection might benefit from ESD over EMR. Those with previously failed EMR might benefit from ESD.
As to the generally low quality of the supporting evidence, Dr. Rubenstein said, “Unfortunately, very few decisions we make in medicine are supported by high certainty of evidence, but we still have to make a decision.” He pointed out that the guideline highlights areas for future research that could help strengthen or change the guideline’s recommendations.
Considering benefits and harms, the panelists concluded that overall CoE across critical desirable outcomes of disease progression to EAC was moderate. Patient-important outcomes informing the harms were strictures, major bleeding perforation, and serious adverse events.
Lifestyle
The guidance also urges providers to counsel BE patients on tobacco cessation and weight loss if needed, and notes the specter of cancer may incentivize patients to make lifestyle changes.
The most common causes of death in EET patients are cardiovascular disease and other cancers, for which tobacco use and obesity are also major risk factors, and tobacco is associated with strictures, the panelists wrote. “The prospect of progression to cancer in patients with dysplastic BE often holds greater valence than prior counseling attempts, and patients may re-commit to such efforts following consultation for EET.”
Going Forward
Areas for future attention include:
- Identifying populations with non-dysplastic BE whose risk warrants EET
- Balancing risk and benefit of EET in low-grade dysplasia
- Randomized controlled trials comparing EMR and ESD in higher-risk lesions
- Optimal management of post-EET pain
- Stricture prevention and control
- Managing resistant/recurrent disease beyond reflux control
- Optimal surveillance and biopsy strategies following EETThis guideline was supported by the National Institutes of Health, the Department of Defense, the Veterans Administration Health Services and Research Division, and the Katy O. and Paul M. Rady Endowed Chair in Esophageal Cancer Research at the University of Colorado.
Dr. Sawas had no competing interests to disclose. Dr. Rubenstein reported research funding from Lucid Diagnostics.
Several other panelists reported research funding or consultation fees from various pharmaceutical and biotechnology companies.
Published in Gastroenterology , the clinical practice guideline makes five main recommendations — one strong and four conditional — based on very low to moderate evidence. It also stresses that providers should practice shared decision making according to patient preferences and risk perception.
For the most part, the new guideline is not a significant departure from the way expert endoscopists are currently practicing EET for BE and related neoplasia, gastroenterologist Joel H. Rubenstein, MD, MSc, AGAF, of the Barrett’s Esophagus Program in the Division of Gastroenterology at University of Michigan Medical School at Ann Arbor, said in an interview. One of three first authors of the guideline, Dr. Rubenstein added, “There is, however, considerable variability in how endoscopists practice, and we hope this guidance will serve as a useful resource to refer to for best practices.”
Added gastroenterologist Tarek Sawas, MD, MPH, assistant professor of internal medicine at UT Southwestern Medical Center in Dallas, “We hope the update will provide some clarity for practice and for implementation, while allowing gastroenterologists the freedom to decide what is best for patients based on lesion characteristics.”
Dr. Sawas added that one of the differences in the new guideline relates to the approach to low-grade dysplasia. While earlier guidance favored treatment over surveillance, patient preferences should now be factored into management. “Some patients are risk-averse and prefer to wait and watch, while others place more value on treatment and just want to get on with it,” he said.
When this guideline was circulated for public comment, “the areas prompting the most feedback was on our current suggestions against the routine use of EET in non-dysplastic BE and for the use of either endoscopic mucosal resection [EMR] or endoscopic submucosal dissection [ESD] for resection — with the expectation that the vast majority may be managed with EMR,” Dr. Rubenstein said.
“We felt that ESD would work best for larger lesions,” explained Dr. Sawas. “There aren’t a lot data in this area, just some observational studies, but we should have more data for comparison in the next few years.”
The incidence of esophageal adenocarcinoma continues to rise and an update was deemed in order since the AGA’s last formal guidance on this subject using the systematic GRADE (Grading of Recommendations Assessment, Development, and Evaluation) methodology was issued in 2011. “In the following time span, there’s been a lot of research, particularly with regard to management of low-grade dysplasia and endoscopic resection techniques,” Dr. Rubenstein said.
Key Recommendations
The 14 guideline panelists made the following suggestions for treatment and implementation based on different levels of certainty of evidence (CoE):
1. If high-grade dysplasia (HGD) is present, EET is recommended over surveillance, with subsequent surveillance performed at 3, 6, and 12 months, and annually thereafter. (Strong recommendation, moderate CoE).
Surveillance endoscopies should obtain targeted tissue samples of visible lesions and random biopsies of the cardia and distal 2 cm of the tubular esophagus.
2. In patients with low-grade dysplasia, EET is also preferred to surveillance. But for those placing a higher value on the certain harms and a lower value on the uncertain benefits of EET for reducing mortality, surveillance endoscopy is a reasonable option. (Conditional recommendation, low CoE).
Following EET, clinicians should perform surveillance at years 1 and 3 after complete eradication of intestinal metaplasia, then revert to the surveillance intervals used in non-dysplastic BE.
3. For non-dysplastic BE, the AGA advises against the routine use of EET. (Conditional recommendation, low CoE).
4. Patients undergoing EET should have resection of visible lesions followed by ablation of the remaining BE segment rather than resection of the entire segment.
In patients with only a small area of BE beyond the visible lesion, endoscopic resection is acceptable and may be preferred over repeated ablation. Radiofrequency ablation is the preferred ablative modality. (Conditional recommendation, very low CoE).
5. For treating visible neoplastic lesions the AGA suggests either EMR or ESD based on lesion characteristics. (Conditional recommendation, very low CoE).
Patients with suspected T1 esophageal adenocarcinoma (EAC) should be considered for EET. Endoscopic resection is recommended over endoscopic ultrasound for distinguishing EAC from HGD and for staging depth of invasion.
The vast majority of neoplastic lesions may be managed with EMR rather than ESD. Patients who have bulky lesions, or lesions highly suspicious of at least T1b invasion and are deemed candidates for endoscopic resection might benefit from ESD over EMR. Those with previously failed EMR might benefit from ESD.
As to the generally low quality of the supporting evidence, Dr. Rubenstein said, “Unfortunately, very few decisions we make in medicine are supported by high certainty of evidence, but we still have to make a decision.” He pointed out that the guideline highlights areas for future research that could help strengthen or change the guideline’s recommendations.
Considering benefits and harms, the panelists concluded that overall CoE across critical desirable outcomes of disease progression to EAC was moderate. Patient-important outcomes informing the harms were strictures, major bleeding perforation, and serious adverse events.
Lifestyle
The guidance also urges providers to counsel BE patients on tobacco cessation and weight loss if needed, and notes the specter of cancer may incentivize patients to make lifestyle changes.
The most common causes of death in EET patients are cardiovascular disease and other cancers, for which tobacco use and obesity are also major risk factors, and tobacco is associated with strictures, the panelists wrote. “The prospect of progression to cancer in patients with dysplastic BE often holds greater valence than prior counseling attempts, and patients may re-commit to such efforts following consultation for EET.”
Going Forward
Areas for future attention include:
- Identifying populations with non-dysplastic BE whose risk warrants EET
- Balancing risk and benefit of EET in low-grade dysplasia
- Randomized controlled trials comparing EMR and ESD in higher-risk lesions
- Optimal management of post-EET pain
- Stricture prevention and control
- Managing resistant/recurrent disease beyond reflux control
- Optimal surveillance and biopsy strategies following EETThis guideline was supported by the National Institutes of Health, the Department of Defense, the Veterans Administration Health Services and Research Division, and the Katy O. and Paul M. Rady Endowed Chair in Esophageal Cancer Research at the University of Colorado.
Dr. Sawas had no competing interests to disclose. Dr. Rubenstein reported research funding from Lucid Diagnostics.
Several other panelists reported research funding or consultation fees from various pharmaceutical and biotechnology companies.
Published in Gastroenterology , the clinical practice guideline makes five main recommendations — one strong and four conditional — based on very low to moderate evidence. It also stresses that providers should practice shared decision making according to patient preferences and risk perception.
For the most part, the new guideline is not a significant departure from the way expert endoscopists are currently practicing EET for BE and related neoplasia, gastroenterologist Joel H. Rubenstein, MD, MSc, AGAF, of the Barrett’s Esophagus Program in the Division of Gastroenterology at University of Michigan Medical School at Ann Arbor, said in an interview. One of three first authors of the guideline, Dr. Rubenstein added, “There is, however, considerable variability in how endoscopists practice, and we hope this guidance will serve as a useful resource to refer to for best practices.”
Added gastroenterologist Tarek Sawas, MD, MPH, assistant professor of internal medicine at UT Southwestern Medical Center in Dallas, “We hope the update will provide some clarity for practice and for implementation, while allowing gastroenterologists the freedom to decide what is best for patients based on lesion characteristics.”
Dr. Sawas added that one of the differences in the new guideline relates to the approach to low-grade dysplasia. While earlier guidance favored treatment over surveillance, patient preferences should now be factored into management. “Some patients are risk-averse and prefer to wait and watch, while others place more value on treatment and just want to get on with it,” he said.
When this guideline was circulated for public comment, “the areas prompting the most feedback was on our current suggestions against the routine use of EET in non-dysplastic BE and for the use of either endoscopic mucosal resection [EMR] or endoscopic submucosal dissection [ESD] for resection — with the expectation that the vast majority may be managed with EMR,” Dr. Rubenstein said.
“We felt that ESD would work best for larger lesions,” explained Dr. Sawas. “There aren’t a lot data in this area, just some observational studies, but we should have more data for comparison in the next few years.”
The incidence of esophageal adenocarcinoma continues to rise and an update was deemed in order since the AGA’s last formal guidance on this subject using the systematic GRADE (Grading of Recommendations Assessment, Development, and Evaluation) methodology was issued in 2011. “In the following time span, there’s been a lot of research, particularly with regard to management of low-grade dysplasia and endoscopic resection techniques,” Dr. Rubenstein said.
Key Recommendations
The 14 guideline panelists made the following suggestions for treatment and implementation based on different levels of certainty of evidence (CoE):
1. If high-grade dysplasia (HGD) is present, EET is recommended over surveillance, with subsequent surveillance performed at 3, 6, and 12 months, and annually thereafter. (Strong recommendation, moderate CoE).
Surveillance endoscopies should obtain targeted tissue samples of visible lesions and random biopsies of the cardia and distal 2 cm of the tubular esophagus.
2. In patients with low-grade dysplasia, EET is also preferred to surveillance. But for those placing a higher value on the certain harms and a lower value on the uncertain benefits of EET for reducing mortality, surveillance endoscopy is a reasonable option. (Conditional recommendation, low CoE).
Following EET, clinicians should perform surveillance at years 1 and 3 after complete eradication of intestinal metaplasia, then revert to the surveillance intervals used in non-dysplastic BE.
3. For non-dysplastic BE, the AGA advises against the routine use of EET. (Conditional recommendation, low CoE).
4. Patients undergoing EET should have resection of visible lesions followed by ablation of the remaining BE segment rather than resection of the entire segment.
In patients with only a small area of BE beyond the visible lesion, endoscopic resection is acceptable and may be preferred over repeated ablation. Radiofrequency ablation is the preferred ablative modality. (Conditional recommendation, very low CoE).
5. For treating visible neoplastic lesions the AGA suggests either EMR or ESD based on lesion characteristics. (Conditional recommendation, very low CoE).
Patients with suspected T1 esophageal adenocarcinoma (EAC) should be considered for EET. Endoscopic resection is recommended over endoscopic ultrasound for distinguishing EAC from HGD and for staging depth of invasion.
The vast majority of neoplastic lesions may be managed with EMR rather than ESD. Patients who have bulky lesions, or lesions highly suspicious of at least T1b invasion and are deemed candidates for endoscopic resection might benefit from ESD over EMR. Those with previously failed EMR might benefit from ESD.
As to the generally low quality of the supporting evidence, Dr. Rubenstein said, “Unfortunately, very few decisions we make in medicine are supported by high certainty of evidence, but we still have to make a decision.” He pointed out that the guideline highlights areas for future research that could help strengthen or change the guideline’s recommendations.
Considering benefits and harms, the panelists concluded that overall CoE across critical desirable outcomes of disease progression to EAC was moderate. Patient-important outcomes informing the harms were strictures, major bleeding perforation, and serious adverse events.
Lifestyle
The guidance also urges providers to counsel BE patients on tobacco cessation and weight loss if needed, and notes the specter of cancer may incentivize patients to make lifestyle changes.
The most common causes of death in EET patients are cardiovascular disease and other cancers, for which tobacco use and obesity are also major risk factors, and tobacco is associated with strictures, the panelists wrote. “The prospect of progression to cancer in patients with dysplastic BE often holds greater valence than prior counseling attempts, and patients may re-commit to such efforts following consultation for EET.”
Going Forward
Areas for future attention include:
- Identifying populations with non-dysplastic BE whose risk warrants EET
- Balancing risk and benefit of EET in low-grade dysplasia
- Randomized controlled trials comparing EMR and ESD in higher-risk lesions
- Optimal management of post-EET pain
- Stricture prevention and control
- Managing resistant/recurrent disease beyond reflux control
- Optimal surveillance and biopsy strategies following EETThis guideline was supported by the National Institutes of Health, the Department of Defense, the Veterans Administration Health Services and Research Division, and the Katy O. and Paul M. Rady Endowed Chair in Esophageal Cancer Research at the University of Colorado.
Dr. Sawas had no competing interests to disclose. Dr. Rubenstein reported research funding from Lucid Diagnostics.
Several other panelists reported research funding or consultation fees from various pharmaceutical and biotechnology companies.
FROM GASTROENTEROLOGY
Endoscopic Management of Barrett’s Esophagus
Introduction
Barrett’s esophagus (BE) is characterized by the replacement of squamous epithelium by columnar metaplasia of the distal esophagus (>1 cm length). It is a precancerous condition, with 3%-5% of patients with BE developing esophageal adenocarcinoma (EAC) in their lifetime. EAC is one of the cancers with high morbidity and mortality (5-year survival < 20%), and its incidence has been on the rise. Studies examining the natural history of BE have demonstrated that the progression happens through a metaplasia-dysplasia-neoplasia sequence. Therefore, early detection of BE and timely management to prevent progression to EAC is crucial.
Grades of Dysplasia
The current gold standard for the diagnosis of BE neoplasia includes a high-quality endoscopic evaluation and biopsies. Biopsies should be obtained from any visible lesions (nodules, ulcers) followed by a random 4-quadrant fashion (Seattle protocol) interval of the entire length of the BE segment. It is essential to pay attention to the results of the biopsy that have been obtained since it will not only determine the surveillance interval but is crucial in planning any necessary endoscopic therapy. The possible results of the biopsy and its implications are:
- No intestinal metaplasia (IM): This would rule out Barrett’s esophagus and no further surveillance would be necessary. A recent population-based study of over 1 million patients showed a 55% and 61% reduced risk of upper gastrointestinal (UGI) cancer and deaths respectively after a negative endoscopy.1
- Intestinal metaplasia with no dysplasia (non-dysplastic BE): Biopsies confirm presence of intestinal metaplasia in the biopsies without any evidence of dysplasia. While the rate of progression to EAC is low (0.07%-0.25%), it is not absent and thus surveillance would be indicated. Current guidelines suggest repeating an endoscopy with biopsy in 5 years if the length of BE is < 3 cm or 3 years if length of BE ≥ 3 cm.2
- Indeterminate for dysplasia (BE-IND): Biopsies confirm IM but are not able to definitively rule out dysplasia. This can be seen in about 4%-8% of the biopsies obtained. The progression rates to EAC are reported to be comparable or lower to low-grade dysplasia (LGD), so the current recommendation is to intensify acid reduction therapy and repeat endoscopy in 6 months. If repeat endoscopy downgrades to non-dysplastic, then can follow surveillance according to NDBE protocol; otherwise recommend continuing surveillance every 12 months.
- Low-grade dysplasia (BE-LGD): Biopsies confirm IM but also show tightly packed overlapping basal nuclei with hyperchromasia and irregular contours, basal stratification of nuclei, and diminished goblet and columnar cell mucus. There is significant inter-observer variability reported,3 and thus the slides must be reviewed by a second pathologist with experience in BE to confirm the findings. Once confirmed, based on risk factors such as presence of multifocal LGD, persistence of LGD, presence of visible lesions, etc., the patient can be offered Barrett’s endoscopic therapy (BET) or undergo continued surveillance. The decision of pursuing one or the other would be dependent on patient preference and shared decision-making between the patient and the provider.
- High-grade dysplasia (BE-HGD): Biopsies confirm IM with cells showing greater degree of cytologic and architectural alterations of dysplasia than LGD but without overt neoplastic features. Over 40% of the patients would progress to EAC and thus the current recommendations would be to recommend BET in these patients.4
- Esophageal adenocarcinoma (EAC): Biopsies demonstrate neoplasia. If the neoplastic changes are limited to the mucosa (T1a) on endoscopic ultrasound or cross-sectional imaging, then BET is suggested. If there is involvement of submucosa, then depending on the depth of invasion, absence of high-risk features (poor differentiation, lymphovascular invasion), BET can be considered as an alternative to esophagectomy.
Lesion Detection on Endoscopy
Data from large population-based studies with at least 3 years of follow-up reported that 58%-66% of EAC detected during endoscopy were diagnosed within 1 year of an index Barrett’s esophagus screening endoscopy, or post-endoscopy Barrett’s neoplasia, and were considered likely to have been missed during index endoscopy.5 This underscores the importance of careful and systematic endoscopic examination during an upper endoscopy.
Studies have also demonstrated that longer examination time was associated with significantly higher detection of HGD/EAC.6,7 Careful examination of the tubular esophagus and gastroesophageal junction (GEJ) should be performed in forward and retroflexed views looking for any subtle areas of nodularity, loop distortion, variability in vascular patterns, mucosal changes concerning for dysplasia or neoplasia. Use of high-definition white light endoscopy (HD-WLE) and virtual chromoendoscopy techniques such as narrow banding imaging (NBI) or blue laser imaging (BLI) are currently recommended in the guidelines.2 Spray chromoendoscopy using acetic acid can also be utilized. Another exciting development is the use of artificial intelligence (AI) in detecting and diagnosing BE associated lesions and neoplasia.
Barrett’s Endoscopic Therapy (BET)
Patients with visible lesions, dysplasia, or early EAC are candidates for BET (Table 1). 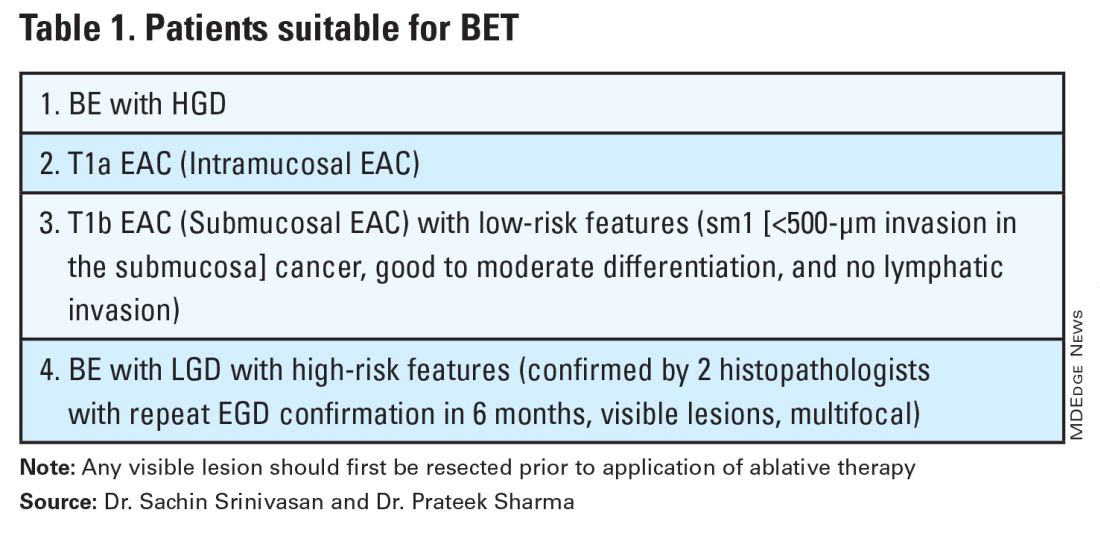
BET involves resective and ablative modalities. The resective modalities include endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD) and are the modalities of choice for nodular or raised lesions.
EMR involves endoscopic resection of abnormal mucosa using either lift-assisted technique or multi-band ligation (Figure 1). 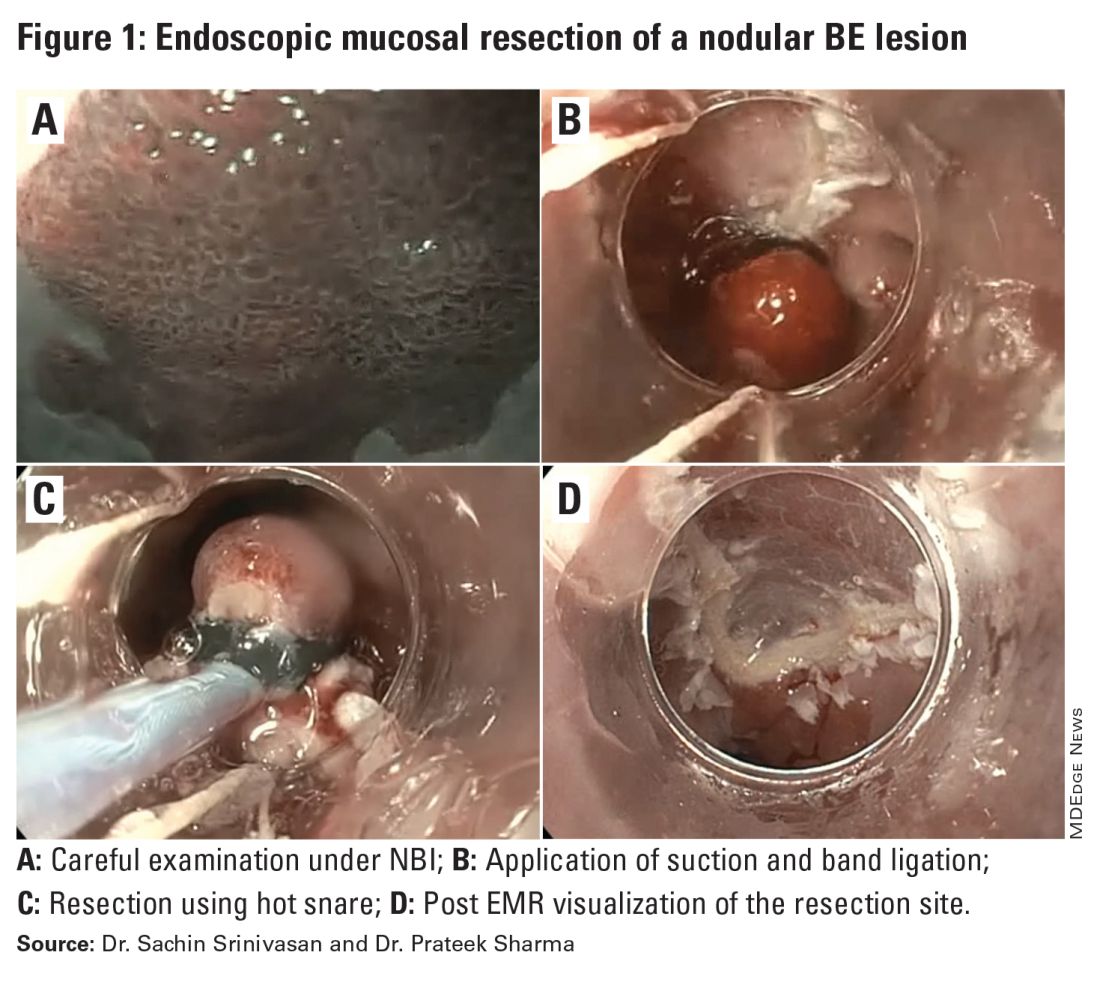
ESD, on the other hand, involves submucosal dissection and perimeter resection of the lesion, thus providing the advantage of an en-bloc resection. In a recent randomized controlled trial (RCT) of 40 patients undergoing ESD vs EMR for HGD/EAC, ESD was better for curative resection (R0) (58%) compared with EMR (12%); however, the remission rates at 3 months were comparable with two perforations reported in the ESD group while there were no complications in the EMR group.8
There is an apparent learning curve when it comes to these advanced techniques, and with more experience, we are seeing comparable results for both these modalities. However, given the complexity and time required for the procedure, current practices typically involve preserving ESD for lesions > 2 cm, those having a likelihood of cancer in the superficial submucosa, or those that EMR cannot remove due to underlying fibrosis or post-EMR recurrence.
The ablative modalities include radiofrequency ablation (RFA), cryotherapy, and hybrid argon plasma coagulation (hybrid APC). These modalities are used for flat lesions, and as therapy following endoscopic resection of nodular lesions to treat residual flat segment of BE. RFA, one of the earliest introduced endoscopic modalities, involves applying directed and controlled heat energy to ablate lesions. Current devices allow circumferential or focal application of RFA. It is a safe and effective modality with good complete eradication of IM (CE-IM) (71%-93%) and complete eradication of dysplasia (CE-D) (91%-100%) rates. These results have been sustained even at 2 years, with the most recent long-term data from a registry study showing a relapse rate of 6% for dysplasia and 19% for IM after 8 years, suggesting durability of this treatment.9
Cryotherapy involves the application of liquid nitrogen or rapidly expanding CO2 to the abnormal mucosa, leading to the rapid freezing and thawing that leads to the death of the cells. Cryogen can be applied as a spray or using a balloon with the spray nozzle in the center. This modality can be used to treat focal lesions and/or larger segments. While it has not been systematically compared with RFA, rates of CE-IM up to 81% and CE-D up to 97% are reported. Hybrid APC involves the use of submucosal saline injection to provide a protective cushion before APC is applied. It has CE-IM rate of 69% and CE-D rate of 67%-86%.10 In a recent RCT of 101 patients randomized to RFA or hybrid APC, CE-IM rates were similar (RFA:74.2% vs hAPC: 82.9%).11
Recently, another technique called radiofrequency vapor ablation (RFVA) is being evaluated, which involves ablating BE segment using vapor at 100° C generated with an RF electrode. A proof-of-concept study of 15 patients showed median squamous conversion of 55% (IQR 33-74) and 98% (IQR 56-99) for 1- and 3-second applications, respectively, with no reported adverse events.12
Barrett’s Refractory to Endoscopic Therapy
Failure of BET is defined as persistent columnar lined epithelium (intestinal metaplasia) with inadequate response, after adequate attempts at endoscopic ablation therapy (after resection) with at least four ablation sessions.13 If encountered, special attention must be given to check compliance with proton pump inhibitors (PPIs), previous incomplete resection, and presence of large hiatal hernia. If CE-IM is not achieved after multiple sessions, change of ablative modality is typically considered. In addition, careful examination for visible lesions should be performed and even if a small one is noted, this should be first resected prior to application of any ablative therapy.
Currently there are no guideline recommendations regarding the preference of one endoscopic modality over another or consideration of potential endoscopic or surgical fundoplication. These modalities primarily rely on technologies available at an institution and the preference of a provider based on their training and experience. Most studies indicate 1-3 sessions (~ 3 months apart) of ablative treatment before achieving CE-IM.
Success and Adverse Events of BET
In a recent real-world study of over 27,000 patients with dysplastic BE, 5295 underwent BET. Analysis showed that patients with HGD/EAC who had BET had a significantly lower 3-year mortality (HGD: RR, 0.59; 95%CI, 0.49-0.71; EAC: RR, 0.53; 95% CI, 0.44-0.65) compared with those who did not undergo BET. Esophageal strictures were the most common adverse event and were noted in 6.5%, followed by chest pain (1.8%), upper GI bleeding (0.47%), and esophageal perforation (0.2%).14
In general, adverse events can be divided into immediate and delayed adverse events. Immediate adverse events typically involve bleeding and perforation that can occur during or shortly after the procedure. These are reported at higher rates with resective modalities compared with ablative therapies. Standard endoscopic techniques involving coagulation grasper or clips can be used to achieve hemostasis. Endoscopic suturing devices offer the ability to contain any perforation. The need for surgical intervention is small and limited to adverse events not detected during the procedure.
Delayed adverse events such as stricture and stenosis are higher for resective modalities (up to 30%), especially when involving more significant than 75% of the esophageal circumference. Post-procedural pain/dysphagia is most common after ablative therapies. Dysphagia reported after any endoscopic therapy should be promptly evaluated, and sequential dilation until the goal esophageal lumen is achieved should be performed every 2-4 weeks.
Recurrences and Surveillance After BET
What is established is that recurrences can occur and may be subtle, therefore detailed endoscopic surveillance is required. In a prospective study, recurrence rates of 15%-16% for IM and 3%-5% for any dysplasia were reported, with the majority being in the first 2 years after achieving CE-IM.15 A systematic review of 21 studies looking at the location of recurrences suggested that the majority (56%) occur in the distal esophagus. Of those that occur in the esophagus, about 80% of them were in the distal 2 cm of the esophagus and only 50% of the recurrences were visible recurrences, thus reiterating the importance of meticulous examination and systematic biopsies.16
On the contrary, a recent single-center study of 217 patients who had achieved CE-IM with 5.5 years of follow-up demonstrated a 26% and 8% recurrence of IM and dysplasia, respectively. One hundred percent of the recurrence in the esophagus was reported as visible.17 Therefore, follow-up endoscopy surveillance protocol after CE-IM should still involve meticulous examination, biopsy of visible lesions, and systematic biopsies for non-visible lesions from the original BE segment, similar to those patients who have not needed BET.
Current guidelines based on expert consensus and evidence recommend surveillance after CE-IM based on original most advanced histology:2
1. LGD: 1 year, 3 years, and every 2 years after that.
2. HGD/EAC: 3 months, 6 months, 12 months, and annually after that.
There is no clear guideline on when to stop surveillance since the longest available follow-up is around 10 years, and recurrences are still detected. A potential surveillance endpoint may be based on age and comorbidities, especially those that would preclude a patient from being a candidate for BET.
When Should a Patient Be Referred?
BE patients with visible lesions and/or dysplastic changes in the biopsy who would require BET should be considered for referral to high-volume centers. Studies have shown higher success for CE-IM and lower rates of adverse events and recurrences in these patients managed at expert centers. The presence of a multidisciplinary team involving pathologists, surgeons, and oncologists is critical and offers a timely opportunity in case of need for a high-risk patient.
Conclusion
BE is a precursor to EAC, with rising incidence and poor 5-year survival. Endoscopic diagnosis is the gold standard and requires a high-quality examination and biopsies. Based on histopathology, a systematic surveillance and BET plan should be performed to achieve CE-IM in patients with dysplasia. Once CE-IM is achieved, regular surveillance should be performed with careful attention to recurrences and complications from the BET modalities.
Dr. Srinivasan and Dr. Sharma are based at the University of Kansas Medical Center, Kansas City, Kansas, and the Kansas City Veterans Affairs Medical Center, Kansas City, Missouri. Dr. Srinivasan has no relevant disclosures. Dr. Sharma disclosed research grants from ERBE, Ironwood Pharmaceuticals, Olympus, and Medtronic. He has served as a consultant for Takeda, Samsung Bioepis, Olympus, and Lumendi, and reports other funding from Medtronic, Fujifilm Medical Systems USA, and Salix.
References
1. Holmberg D, et al. Incidence and mortality in upper gastrointestinal cancer after negative endoscopy for gastroesophageal reflux disease. Gastroenterology. 2022;162(2):431-438.e4.
2. Shaheen NJ, et al. Diagnosis and management of Barrett’s esophagus: An updated ACG guideline. Am J Gastroenterol. 2022 Apr;117(4):559-587.
3. Pech O, et al. Inter-observer variability in the diagnosis of low-grade dysplasia in pathologists: A comparison between experienced and inexperienced pathologists. Gastrointest Endosc. 2006 Apr;63(5):AB130.
4. Krishnamoorthi R, et al. Factors associated with progression of Barrett’s esophagus: A systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2018 Jul;16(7):1046-1055.e8.
5. Visrodia K, et al. Magnitude of missed esophageal adenocarcinoma after Barrett’s esophagus diagnosis: A systematic review and meta-analysis. Gastroenterology. 2016 Mar;150(3):599-607.e7; quiz e14-5.
6. Perisetti A, Sharma P. Tips for improving the identification of neoplastic visible lesions in Barrett’s esophagus. Gastrointest Endosc. 2023 Feb;97(2):248-250.
7. Gupta N, et al. Longer inspection time is associated with increased detection of high-grade dysplasia and esophageal adenocarcinoma in Barrett’s esophagus. Gastrointest Endosc. 2012 Sep;76(3):531-538.
8. Terheggen G, et al. A randomised trial of endoscopic submucosal dissection versus endoscopic mucosal resection for early Barrett’s neoplasia. Gut. 2017 May;66(5):783-793.
9. Wolfson P, et al. Endoscopic eradication therapy for Barrett’s esophagus-related neoplasia: A final 10-year report from the UK National HALO Radiofrequency Ablation Registry. Gastrointest Endosc. 2022 Aug;96(2):223-233.
10. Rösch T, et al. 1151 Multicenter feasibility study of combined injection and argon plasma coagulation (hybrid-APC) in the ablation therapy of neoplastic Barrett esophagus. Gastrointest Endosc. 2017;85(5):AB154.
11. Knabe M, et al. Radiofrequency ablation versus hybrid argon plasma coagulation in Barrett’s esophagus: A prospective randomised trial. Surg Endosc. 2023;37(10):7803-7811.
12. Van Munster SN, et al. Radiofrequency vapor ablation for Barrett’s esophagus: Feasibility, safety, and proof of concept in a stepwise study with in vitro, animal, and the first in-human application. Endoscopy. 2021 Nov;53(11):1162-1168.
13. Emura F, et al. Rio de Janeiro global consensus on landmarks, definitions, and classifications in Barrett’s esophagus: World Endoscopy Organization Delphi study. Gastroenterology. 2022 Jul;163(1):84-96.e2.
14. Singh RR, et al. Real-world evidence of safety and effectiveness of Barrett’s endoscopic therapy. Gastrointest Endosc. 2023 Aug;98(2):155-161.e1.
15. Wani S, et al. Recurrence Is rare following complete eradication of intestinal metaplasia in patients with Barrett’s esophagus and peaks at 18 months. Clin Gastroenterol Hepatol. 2020 Oct;18(11):2609-2617.e2.
16. Duvvuri A, et al. Mo1273 Location and pattern of recurrences in patients with Barrett’s esophagus after endoscopic therapy: A systematic review and critical analysis of the published literature. Gastrointest Endosc. 2020;91(6):AB410-1.
17. He T, et al. Location and appearance of dysplastic Barrett’s esophagus recurrence after endoscopic eradication therapy: No additional yield from random biopsy sampling neosquamous mucosa. Gastrointest Endosc. 2023 Nov;98(5):722-732.
Introduction
Barrett’s esophagus (BE) is characterized by the replacement of squamous epithelium by columnar metaplasia of the distal esophagus (>1 cm length). It is a precancerous condition, with 3%-5% of patients with BE developing esophageal adenocarcinoma (EAC) in their lifetime. EAC is one of the cancers with high morbidity and mortality (5-year survival < 20%), and its incidence has been on the rise. Studies examining the natural history of BE have demonstrated that the progression happens through a metaplasia-dysplasia-neoplasia sequence. Therefore, early detection of BE and timely management to prevent progression to EAC is crucial.
Grades of Dysplasia
The current gold standard for the diagnosis of BE neoplasia includes a high-quality endoscopic evaluation and biopsies. Biopsies should be obtained from any visible lesions (nodules, ulcers) followed by a random 4-quadrant fashion (Seattle protocol) interval of the entire length of the BE segment. It is essential to pay attention to the results of the biopsy that have been obtained since it will not only determine the surveillance interval but is crucial in planning any necessary endoscopic therapy. The possible results of the biopsy and its implications are:
- No intestinal metaplasia (IM): This would rule out Barrett’s esophagus and no further surveillance would be necessary. A recent population-based study of over 1 million patients showed a 55% and 61% reduced risk of upper gastrointestinal (UGI) cancer and deaths respectively after a negative endoscopy.1
- Intestinal metaplasia with no dysplasia (non-dysplastic BE): Biopsies confirm presence of intestinal metaplasia in the biopsies without any evidence of dysplasia. While the rate of progression to EAC is low (0.07%-0.25%), it is not absent and thus surveillance would be indicated. Current guidelines suggest repeating an endoscopy with biopsy in 5 years if the length of BE is < 3 cm or 3 years if length of BE ≥ 3 cm.2
- Indeterminate for dysplasia (BE-IND): Biopsies confirm IM but are not able to definitively rule out dysplasia. This can be seen in about 4%-8% of the biopsies obtained. The progression rates to EAC are reported to be comparable or lower to low-grade dysplasia (LGD), so the current recommendation is to intensify acid reduction therapy and repeat endoscopy in 6 months. If repeat endoscopy downgrades to non-dysplastic, then can follow surveillance according to NDBE protocol; otherwise recommend continuing surveillance every 12 months.
- Low-grade dysplasia (BE-LGD): Biopsies confirm IM but also show tightly packed overlapping basal nuclei with hyperchromasia and irregular contours, basal stratification of nuclei, and diminished goblet and columnar cell mucus. There is significant inter-observer variability reported,3 and thus the slides must be reviewed by a second pathologist with experience in BE to confirm the findings. Once confirmed, based on risk factors such as presence of multifocal LGD, persistence of LGD, presence of visible lesions, etc., the patient can be offered Barrett’s endoscopic therapy (BET) or undergo continued surveillance. The decision of pursuing one or the other would be dependent on patient preference and shared decision-making between the patient and the provider.
- High-grade dysplasia (BE-HGD): Biopsies confirm IM with cells showing greater degree of cytologic and architectural alterations of dysplasia than LGD but without overt neoplastic features. Over 40% of the patients would progress to EAC and thus the current recommendations would be to recommend BET in these patients.4
- Esophageal adenocarcinoma (EAC): Biopsies demonstrate neoplasia. If the neoplastic changes are limited to the mucosa (T1a) on endoscopic ultrasound or cross-sectional imaging, then BET is suggested. If there is involvement of submucosa, then depending on the depth of invasion, absence of high-risk features (poor differentiation, lymphovascular invasion), BET can be considered as an alternative to esophagectomy.
Lesion Detection on Endoscopy
Data from large population-based studies with at least 3 years of follow-up reported that 58%-66% of EAC detected during endoscopy were diagnosed within 1 year of an index Barrett’s esophagus screening endoscopy, or post-endoscopy Barrett’s neoplasia, and were considered likely to have been missed during index endoscopy.5 This underscores the importance of careful and systematic endoscopic examination during an upper endoscopy.
Studies have also demonstrated that longer examination time was associated with significantly higher detection of HGD/EAC.6,7 Careful examination of the tubular esophagus and gastroesophageal junction (GEJ) should be performed in forward and retroflexed views looking for any subtle areas of nodularity, loop distortion, variability in vascular patterns, mucosal changes concerning for dysplasia or neoplasia. Use of high-definition white light endoscopy (HD-WLE) and virtual chromoendoscopy techniques such as narrow banding imaging (NBI) or blue laser imaging (BLI) are currently recommended in the guidelines.2 Spray chromoendoscopy using acetic acid can also be utilized. Another exciting development is the use of artificial intelligence (AI) in detecting and diagnosing BE associated lesions and neoplasia.
Barrett’s Endoscopic Therapy (BET)
Patients with visible lesions, dysplasia, or early EAC are candidates for BET (Table 1). 
BET involves resective and ablative modalities. The resective modalities include endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD) and are the modalities of choice for nodular or raised lesions.
EMR involves endoscopic resection of abnormal mucosa using either lift-assisted technique or multi-band ligation (Figure 1). 
ESD, on the other hand, involves submucosal dissection and perimeter resection of the lesion, thus providing the advantage of an en-bloc resection. In a recent randomized controlled trial (RCT) of 40 patients undergoing ESD vs EMR for HGD/EAC, ESD was better for curative resection (R0) (58%) compared with EMR (12%); however, the remission rates at 3 months were comparable with two perforations reported in the ESD group while there were no complications in the EMR group.8
There is an apparent learning curve when it comes to these advanced techniques, and with more experience, we are seeing comparable results for both these modalities. However, given the complexity and time required for the procedure, current practices typically involve preserving ESD for lesions > 2 cm, those having a likelihood of cancer in the superficial submucosa, or those that EMR cannot remove due to underlying fibrosis or post-EMR recurrence.
The ablative modalities include radiofrequency ablation (RFA), cryotherapy, and hybrid argon plasma coagulation (hybrid APC). These modalities are used for flat lesions, and as therapy following endoscopic resection of nodular lesions to treat residual flat segment of BE. RFA, one of the earliest introduced endoscopic modalities, involves applying directed and controlled heat energy to ablate lesions. Current devices allow circumferential or focal application of RFA. It is a safe and effective modality with good complete eradication of IM (CE-IM) (71%-93%) and complete eradication of dysplasia (CE-D) (91%-100%) rates. These results have been sustained even at 2 years, with the most recent long-term data from a registry study showing a relapse rate of 6% for dysplasia and 19% for IM after 8 years, suggesting durability of this treatment.9
Cryotherapy involves the application of liquid nitrogen or rapidly expanding CO2 to the abnormal mucosa, leading to the rapid freezing and thawing that leads to the death of the cells. Cryogen can be applied as a spray or using a balloon with the spray nozzle in the center. This modality can be used to treat focal lesions and/or larger segments. While it has not been systematically compared with RFA, rates of CE-IM up to 81% and CE-D up to 97% are reported. Hybrid APC involves the use of submucosal saline injection to provide a protective cushion before APC is applied. It has CE-IM rate of 69% and CE-D rate of 67%-86%.10 In a recent RCT of 101 patients randomized to RFA or hybrid APC, CE-IM rates were similar (RFA:74.2% vs hAPC: 82.9%).11
Recently, another technique called radiofrequency vapor ablation (RFVA) is being evaluated, which involves ablating BE segment using vapor at 100° C generated with an RF electrode. A proof-of-concept study of 15 patients showed median squamous conversion of 55% (IQR 33-74) and 98% (IQR 56-99) for 1- and 3-second applications, respectively, with no reported adverse events.12
Barrett’s Refractory to Endoscopic Therapy
Failure of BET is defined as persistent columnar lined epithelium (intestinal metaplasia) with inadequate response, after adequate attempts at endoscopic ablation therapy (after resection) with at least four ablation sessions.13 If encountered, special attention must be given to check compliance with proton pump inhibitors (PPIs), previous incomplete resection, and presence of large hiatal hernia. If CE-IM is not achieved after multiple sessions, change of ablative modality is typically considered. In addition, careful examination for visible lesions should be performed and even if a small one is noted, this should be first resected prior to application of any ablative therapy.
Currently there are no guideline recommendations regarding the preference of one endoscopic modality over another or consideration of potential endoscopic or surgical fundoplication. These modalities primarily rely on technologies available at an institution and the preference of a provider based on their training and experience. Most studies indicate 1-3 sessions (~ 3 months apart) of ablative treatment before achieving CE-IM.
Success and Adverse Events of BET
In a recent real-world study of over 27,000 patients with dysplastic BE, 5295 underwent BET. Analysis showed that patients with HGD/EAC who had BET had a significantly lower 3-year mortality (HGD: RR, 0.59; 95%CI, 0.49-0.71; EAC: RR, 0.53; 95% CI, 0.44-0.65) compared with those who did not undergo BET. Esophageal strictures were the most common adverse event and were noted in 6.5%, followed by chest pain (1.8%), upper GI bleeding (0.47%), and esophageal perforation (0.2%).14
In general, adverse events can be divided into immediate and delayed adverse events. Immediate adverse events typically involve bleeding and perforation that can occur during or shortly after the procedure. These are reported at higher rates with resective modalities compared with ablative therapies. Standard endoscopic techniques involving coagulation grasper or clips can be used to achieve hemostasis. Endoscopic suturing devices offer the ability to contain any perforation. The need for surgical intervention is small and limited to adverse events not detected during the procedure.
Delayed adverse events such as stricture and stenosis are higher for resective modalities (up to 30%), especially when involving more significant than 75% of the esophageal circumference. Post-procedural pain/dysphagia is most common after ablative therapies. Dysphagia reported after any endoscopic therapy should be promptly evaluated, and sequential dilation until the goal esophageal lumen is achieved should be performed every 2-4 weeks.
Recurrences and Surveillance After BET
What is established is that recurrences can occur and may be subtle, therefore detailed endoscopic surveillance is required. In a prospective study, recurrence rates of 15%-16% for IM and 3%-5% for any dysplasia were reported, with the majority being in the first 2 years after achieving CE-IM.15 A systematic review of 21 studies looking at the location of recurrences suggested that the majority (56%) occur in the distal esophagus. Of those that occur in the esophagus, about 80% of them were in the distal 2 cm of the esophagus and only 50% of the recurrences were visible recurrences, thus reiterating the importance of meticulous examination and systematic biopsies.16
On the contrary, a recent single-center study of 217 patients who had achieved CE-IM with 5.5 years of follow-up demonstrated a 26% and 8% recurrence of IM and dysplasia, respectively. One hundred percent of the recurrence in the esophagus was reported as visible.17 Therefore, follow-up endoscopy surveillance protocol after CE-IM should still involve meticulous examination, biopsy of visible lesions, and systematic biopsies for non-visible lesions from the original BE segment, similar to those patients who have not needed BET.
Current guidelines based on expert consensus and evidence recommend surveillance after CE-IM based on original most advanced histology:2
1. LGD: 1 year, 3 years, and every 2 years after that.
2. HGD/EAC: 3 months, 6 months, 12 months, and annually after that.
There is no clear guideline on when to stop surveillance since the longest available follow-up is around 10 years, and recurrences are still detected. A potential surveillance endpoint may be based on age and comorbidities, especially those that would preclude a patient from being a candidate for BET.
When Should a Patient Be Referred?
BE patients with visible lesions and/or dysplastic changes in the biopsy who would require BET should be considered for referral to high-volume centers. Studies have shown higher success for CE-IM and lower rates of adverse events and recurrences in these patients managed at expert centers. The presence of a multidisciplinary team involving pathologists, surgeons, and oncologists is critical and offers a timely opportunity in case of need for a high-risk patient.
Conclusion
BE is a precursor to EAC, with rising incidence and poor 5-year survival. Endoscopic diagnosis is the gold standard and requires a high-quality examination and biopsies. Based on histopathology, a systematic surveillance and BET plan should be performed to achieve CE-IM in patients with dysplasia. Once CE-IM is achieved, regular surveillance should be performed with careful attention to recurrences and complications from the BET modalities.
Dr. Srinivasan and Dr. Sharma are based at the University of Kansas Medical Center, Kansas City, Kansas, and the Kansas City Veterans Affairs Medical Center, Kansas City, Missouri. Dr. Srinivasan has no relevant disclosures. Dr. Sharma disclosed research grants from ERBE, Ironwood Pharmaceuticals, Olympus, and Medtronic. He has served as a consultant for Takeda, Samsung Bioepis, Olympus, and Lumendi, and reports other funding from Medtronic, Fujifilm Medical Systems USA, and Salix.
References
1. Holmberg D, et al. Incidence and mortality in upper gastrointestinal cancer after negative endoscopy for gastroesophageal reflux disease. Gastroenterology. 2022;162(2):431-438.e4.
2. Shaheen NJ, et al. Diagnosis and management of Barrett’s esophagus: An updated ACG guideline. Am J Gastroenterol. 2022 Apr;117(4):559-587.
3. Pech O, et al. Inter-observer variability in the diagnosis of low-grade dysplasia in pathologists: A comparison between experienced and inexperienced pathologists. Gastrointest Endosc. 2006 Apr;63(5):AB130.
4. Krishnamoorthi R, et al. Factors associated with progression of Barrett’s esophagus: A systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2018 Jul;16(7):1046-1055.e8.
5. Visrodia K, et al. Magnitude of missed esophageal adenocarcinoma after Barrett’s esophagus diagnosis: A systematic review and meta-analysis. Gastroenterology. 2016 Mar;150(3):599-607.e7; quiz e14-5.
6. Perisetti A, Sharma P. Tips for improving the identification of neoplastic visible lesions in Barrett’s esophagus. Gastrointest Endosc. 2023 Feb;97(2):248-250.
7. Gupta N, et al. Longer inspection time is associated with increased detection of high-grade dysplasia and esophageal adenocarcinoma in Barrett’s esophagus. Gastrointest Endosc. 2012 Sep;76(3):531-538.
8. Terheggen G, et al. A randomised trial of endoscopic submucosal dissection versus endoscopic mucosal resection for early Barrett’s neoplasia. Gut. 2017 May;66(5):783-793.
9. Wolfson P, et al. Endoscopic eradication therapy for Barrett’s esophagus-related neoplasia: A final 10-year report from the UK National HALO Radiofrequency Ablation Registry. Gastrointest Endosc. 2022 Aug;96(2):223-233.
10. Rösch T, et al. 1151 Multicenter feasibility study of combined injection and argon plasma coagulation (hybrid-APC) in the ablation therapy of neoplastic Barrett esophagus. Gastrointest Endosc. 2017;85(5):AB154.
11. Knabe M, et al. Radiofrequency ablation versus hybrid argon plasma coagulation in Barrett’s esophagus: A prospective randomised trial. Surg Endosc. 2023;37(10):7803-7811.
12. Van Munster SN, et al. Radiofrequency vapor ablation for Barrett’s esophagus: Feasibility, safety, and proof of concept in a stepwise study with in vitro, animal, and the first in-human application. Endoscopy. 2021 Nov;53(11):1162-1168.
13. Emura F, et al. Rio de Janeiro global consensus on landmarks, definitions, and classifications in Barrett’s esophagus: World Endoscopy Organization Delphi study. Gastroenterology. 2022 Jul;163(1):84-96.e2.
14. Singh RR, et al. Real-world evidence of safety and effectiveness of Barrett’s endoscopic therapy. Gastrointest Endosc. 2023 Aug;98(2):155-161.e1.
15. Wani S, et al. Recurrence Is rare following complete eradication of intestinal metaplasia in patients with Barrett’s esophagus and peaks at 18 months. Clin Gastroenterol Hepatol. 2020 Oct;18(11):2609-2617.e2.
16. Duvvuri A, et al. Mo1273 Location and pattern of recurrences in patients with Barrett’s esophagus after endoscopic therapy: A systematic review and critical analysis of the published literature. Gastrointest Endosc. 2020;91(6):AB410-1.
17. He T, et al. Location and appearance of dysplastic Barrett’s esophagus recurrence after endoscopic eradication therapy: No additional yield from random biopsy sampling neosquamous mucosa. Gastrointest Endosc. 2023 Nov;98(5):722-732.
Introduction
Barrett’s esophagus (BE) is characterized by the replacement of squamous epithelium by columnar metaplasia of the distal esophagus (>1 cm length). It is a precancerous condition, with 3%-5% of patients with BE developing esophageal adenocarcinoma (EAC) in their lifetime. EAC is one of the cancers with high morbidity and mortality (5-year survival < 20%), and its incidence has been on the rise. Studies examining the natural history of BE have demonstrated that the progression happens through a metaplasia-dysplasia-neoplasia sequence. Therefore, early detection of BE and timely management to prevent progression to EAC is crucial.
Grades of Dysplasia
The current gold standard for the diagnosis of BE neoplasia includes a high-quality endoscopic evaluation and biopsies. Biopsies should be obtained from any visible lesions (nodules, ulcers) followed by a random 4-quadrant fashion (Seattle protocol) interval of the entire length of the BE segment. It is essential to pay attention to the results of the biopsy that have been obtained since it will not only determine the surveillance interval but is crucial in planning any necessary endoscopic therapy. The possible results of the biopsy and its implications are:
- No intestinal metaplasia (IM): This would rule out Barrett’s esophagus and no further surveillance would be necessary. A recent population-based study of over 1 million patients showed a 55% and 61% reduced risk of upper gastrointestinal (UGI) cancer and deaths respectively after a negative endoscopy.1
- Intestinal metaplasia with no dysplasia (non-dysplastic BE): Biopsies confirm presence of intestinal metaplasia in the biopsies without any evidence of dysplasia. While the rate of progression to EAC is low (0.07%-0.25%), it is not absent and thus surveillance would be indicated. Current guidelines suggest repeating an endoscopy with biopsy in 5 years if the length of BE is < 3 cm or 3 years if length of BE ≥ 3 cm.2
- Indeterminate for dysplasia (BE-IND): Biopsies confirm IM but are not able to definitively rule out dysplasia. This can be seen in about 4%-8% of the biopsies obtained. The progression rates to EAC are reported to be comparable or lower to low-grade dysplasia (LGD), so the current recommendation is to intensify acid reduction therapy and repeat endoscopy in 6 months. If repeat endoscopy downgrades to non-dysplastic, then can follow surveillance according to NDBE protocol; otherwise recommend continuing surveillance every 12 months.
- Low-grade dysplasia (BE-LGD): Biopsies confirm IM but also show tightly packed overlapping basal nuclei with hyperchromasia and irregular contours, basal stratification of nuclei, and diminished goblet and columnar cell mucus. There is significant inter-observer variability reported,3 and thus the slides must be reviewed by a second pathologist with experience in BE to confirm the findings. Once confirmed, based on risk factors such as presence of multifocal LGD, persistence of LGD, presence of visible lesions, etc., the patient can be offered Barrett’s endoscopic therapy (BET) or undergo continued surveillance. The decision of pursuing one or the other would be dependent on patient preference and shared decision-making between the patient and the provider.
- High-grade dysplasia (BE-HGD): Biopsies confirm IM with cells showing greater degree of cytologic and architectural alterations of dysplasia than LGD but without overt neoplastic features. Over 40% of the patients would progress to EAC and thus the current recommendations would be to recommend BET in these patients.4
- Esophageal adenocarcinoma (EAC): Biopsies demonstrate neoplasia. If the neoplastic changes are limited to the mucosa (T1a) on endoscopic ultrasound or cross-sectional imaging, then BET is suggested. If there is involvement of submucosa, then depending on the depth of invasion, absence of high-risk features (poor differentiation, lymphovascular invasion), BET can be considered as an alternative to esophagectomy.
Lesion Detection on Endoscopy
Data from large population-based studies with at least 3 years of follow-up reported that 58%-66% of EAC detected during endoscopy were diagnosed within 1 year of an index Barrett’s esophagus screening endoscopy, or post-endoscopy Barrett’s neoplasia, and were considered likely to have been missed during index endoscopy.5 This underscores the importance of careful and systematic endoscopic examination during an upper endoscopy.
Studies have also demonstrated that longer examination time was associated with significantly higher detection of HGD/EAC.6,7 Careful examination of the tubular esophagus and gastroesophageal junction (GEJ) should be performed in forward and retroflexed views looking for any subtle areas of nodularity, loop distortion, variability in vascular patterns, mucosal changes concerning for dysplasia or neoplasia. Use of high-definition white light endoscopy (HD-WLE) and virtual chromoendoscopy techniques such as narrow banding imaging (NBI) or blue laser imaging (BLI) are currently recommended in the guidelines.2 Spray chromoendoscopy using acetic acid can also be utilized. Another exciting development is the use of artificial intelligence (AI) in detecting and diagnosing BE associated lesions and neoplasia.
Barrett’s Endoscopic Therapy (BET)
Patients with visible lesions, dysplasia, or early EAC are candidates for BET (Table 1). 
BET involves resective and ablative modalities. The resective modalities include endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD) and are the modalities of choice for nodular or raised lesions.
EMR involves endoscopic resection of abnormal mucosa using either lift-assisted technique or multi-band ligation (Figure 1). 
ESD, on the other hand, involves submucosal dissection and perimeter resection of the lesion, thus providing the advantage of an en-bloc resection. In a recent randomized controlled trial (RCT) of 40 patients undergoing ESD vs EMR for HGD/EAC, ESD was better for curative resection (R0) (58%) compared with EMR (12%); however, the remission rates at 3 months were comparable with two perforations reported in the ESD group while there were no complications in the EMR group.8
There is an apparent learning curve when it comes to these advanced techniques, and with more experience, we are seeing comparable results for both these modalities. However, given the complexity and time required for the procedure, current practices typically involve preserving ESD for lesions > 2 cm, those having a likelihood of cancer in the superficial submucosa, or those that EMR cannot remove due to underlying fibrosis or post-EMR recurrence.
The ablative modalities include radiofrequency ablation (RFA), cryotherapy, and hybrid argon plasma coagulation (hybrid APC). These modalities are used for flat lesions, and as therapy following endoscopic resection of nodular lesions to treat residual flat segment of BE. RFA, one of the earliest introduced endoscopic modalities, involves applying directed and controlled heat energy to ablate lesions. Current devices allow circumferential or focal application of RFA. It is a safe and effective modality with good complete eradication of IM (CE-IM) (71%-93%) and complete eradication of dysplasia (CE-D) (91%-100%) rates. These results have been sustained even at 2 years, with the most recent long-term data from a registry study showing a relapse rate of 6% for dysplasia and 19% for IM after 8 years, suggesting durability of this treatment.9
Cryotherapy involves the application of liquid nitrogen or rapidly expanding CO2 to the abnormal mucosa, leading to the rapid freezing and thawing that leads to the death of the cells. Cryogen can be applied as a spray or using a balloon with the spray nozzle in the center. This modality can be used to treat focal lesions and/or larger segments. While it has not been systematically compared with RFA, rates of CE-IM up to 81% and CE-D up to 97% are reported. Hybrid APC involves the use of submucosal saline injection to provide a protective cushion before APC is applied. It has CE-IM rate of 69% and CE-D rate of 67%-86%.10 In a recent RCT of 101 patients randomized to RFA or hybrid APC, CE-IM rates were similar (RFA:74.2% vs hAPC: 82.9%).11
Recently, another technique called radiofrequency vapor ablation (RFVA) is being evaluated, which involves ablating BE segment using vapor at 100° C generated with an RF electrode. A proof-of-concept study of 15 patients showed median squamous conversion of 55% (IQR 33-74) and 98% (IQR 56-99) for 1- and 3-second applications, respectively, with no reported adverse events.12
Barrett’s Refractory to Endoscopic Therapy
Failure of BET is defined as persistent columnar lined epithelium (intestinal metaplasia) with inadequate response, after adequate attempts at endoscopic ablation therapy (after resection) with at least four ablation sessions.13 If encountered, special attention must be given to check compliance with proton pump inhibitors (PPIs), previous incomplete resection, and presence of large hiatal hernia. If CE-IM is not achieved after multiple sessions, change of ablative modality is typically considered. In addition, careful examination for visible lesions should be performed and even if a small one is noted, this should be first resected prior to application of any ablative therapy.
Currently there are no guideline recommendations regarding the preference of one endoscopic modality over another or consideration of potential endoscopic or surgical fundoplication. These modalities primarily rely on technologies available at an institution and the preference of a provider based on their training and experience. Most studies indicate 1-3 sessions (~ 3 months apart) of ablative treatment before achieving CE-IM.
Success and Adverse Events of BET
In a recent real-world study of over 27,000 patients with dysplastic BE, 5295 underwent BET. Analysis showed that patients with HGD/EAC who had BET had a significantly lower 3-year mortality (HGD: RR, 0.59; 95%CI, 0.49-0.71; EAC: RR, 0.53; 95% CI, 0.44-0.65) compared with those who did not undergo BET. Esophageal strictures were the most common adverse event and were noted in 6.5%, followed by chest pain (1.8%), upper GI bleeding (0.47%), and esophageal perforation (0.2%).14
In general, adverse events can be divided into immediate and delayed adverse events. Immediate adverse events typically involve bleeding and perforation that can occur during or shortly after the procedure. These are reported at higher rates with resective modalities compared with ablative therapies. Standard endoscopic techniques involving coagulation grasper or clips can be used to achieve hemostasis. Endoscopic suturing devices offer the ability to contain any perforation. The need for surgical intervention is small and limited to adverse events not detected during the procedure.
Delayed adverse events such as stricture and stenosis are higher for resective modalities (up to 30%), especially when involving more significant than 75% of the esophageal circumference. Post-procedural pain/dysphagia is most common after ablative therapies. Dysphagia reported after any endoscopic therapy should be promptly evaluated, and sequential dilation until the goal esophageal lumen is achieved should be performed every 2-4 weeks.
Recurrences and Surveillance After BET
What is established is that recurrences can occur and may be subtle, therefore detailed endoscopic surveillance is required. In a prospective study, recurrence rates of 15%-16% for IM and 3%-5% for any dysplasia were reported, with the majority being in the first 2 years after achieving CE-IM.15 A systematic review of 21 studies looking at the location of recurrences suggested that the majority (56%) occur in the distal esophagus. Of those that occur in the esophagus, about 80% of them were in the distal 2 cm of the esophagus and only 50% of the recurrences were visible recurrences, thus reiterating the importance of meticulous examination and systematic biopsies.16
On the contrary, a recent single-center study of 217 patients who had achieved CE-IM with 5.5 years of follow-up demonstrated a 26% and 8% recurrence of IM and dysplasia, respectively. One hundred percent of the recurrence in the esophagus was reported as visible.17 Therefore, follow-up endoscopy surveillance protocol after CE-IM should still involve meticulous examination, biopsy of visible lesions, and systematic biopsies for non-visible lesions from the original BE segment, similar to those patients who have not needed BET.
Current guidelines based on expert consensus and evidence recommend surveillance after CE-IM based on original most advanced histology:2
1. LGD: 1 year, 3 years, and every 2 years after that.
2. HGD/EAC: 3 months, 6 months, 12 months, and annually after that.
There is no clear guideline on when to stop surveillance since the longest available follow-up is around 10 years, and recurrences are still detected. A potential surveillance endpoint may be based on age and comorbidities, especially those that would preclude a patient from being a candidate for BET.
When Should a Patient Be Referred?
BE patients with visible lesions and/or dysplastic changes in the biopsy who would require BET should be considered for referral to high-volume centers. Studies have shown higher success for CE-IM and lower rates of adverse events and recurrences in these patients managed at expert centers. The presence of a multidisciplinary team involving pathologists, surgeons, and oncologists is critical and offers a timely opportunity in case of need for a high-risk patient.
Conclusion
BE is a precursor to EAC, with rising incidence and poor 5-year survival. Endoscopic diagnosis is the gold standard and requires a high-quality examination and biopsies. Based on histopathology, a systematic surveillance and BET plan should be performed to achieve CE-IM in patients with dysplasia. Once CE-IM is achieved, regular surveillance should be performed with careful attention to recurrences and complications from the BET modalities.
Dr. Srinivasan and Dr. Sharma are based at the University of Kansas Medical Center, Kansas City, Kansas, and the Kansas City Veterans Affairs Medical Center, Kansas City, Missouri. Dr. Srinivasan has no relevant disclosures. Dr. Sharma disclosed research grants from ERBE, Ironwood Pharmaceuticals, Olympus, and Medtronic. He has served as a consultant for Takeda, Samsung Bioepis, Olympus, and Lumendi, and reports other funding from Medtronic, Fujifilm Medical Systems USA, and Salix.
References
1. Holmberg D, et al. Incidence and mortality in upper gastrointestinal cancer after negative endoscopy for gastroesophageal reflux disease. Gastroenterology. 2022;162(2):431-438.e4.
2. Shaheen NJ, et al. Diagnosis and management of Barrett’s esophagus: An updated ACG guideline. Am J Gastroenterol. 2022 Apr;117(4):559-587.
3. Pech O, et al. Inter-observer variability in the diagnosis of low-grade dysplasia in pathologists: A comparison between experienced and inexperienced pathologists. Gastrointest Endosc. 2006 Apr;63(5):AB130.
4. Krishnamoorthi R, et al. Factors associated with progression of Barrett’s esophagus: A systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2018 Jul;16(7):1046-1055.e8.
5. Visrodia K, et al. Magnitude of missed esophageal adenocarcinoma after Barrett’s esophagus diagnosis: A systematic review and meta-analysis. Gastroenterology. 2016 Mar;150(3):599-607.e7; quiz e14-5.
6. Perisetti A, Sharma P. Tips for improving the identification of neoplastic visible lesions in Barrett’s esophagus. Gastrointest Endosc. 2023 Feb;97(2):248-250.
7. Gupta N, et al. Longer inspection time is associated with increased detection of high-grade dysplasia and esophageal adenocarcinoma in Barrett’s esophagus. Gastrointest Endosc. 2012 Sep;76(3):531-538.
8. Terheggen G, et al. A randomised trial of endoscopic submucosal dissection versus endoscopic mucosal resection for early Barrett’s neoplasia. Gut. 2017 May;66(5):783-793.
9. Wolfson P, et al. Endoscopic eradication therapy for Barrett’s esophagus-related neoplasia: A final 10-year report from the UK National HALO Radiofrequency Ablation Registry. Gastrointest Endosc. 2022 Aug;96(2):223-233.
10. Rösch T, et al. 1151 Multicenter feasibility study of combined injection and argon plasma coagulation (hybrid-APC) in the ablation therapy of neoplastic Barrett esophagus. Gastrointest Endosc. 2017;85(5):AB154.
11. Knabe M, et al. Radiofrequency ablation versus hybrid argon plasma coagulation in Barrett’s esophagus: A prospective randomised trial. Surg Endosc. 2023;37(10):7803-7811.
12. Van Munster SN, et al. Radiofrequency vapor ablation for Barrett’s esophagus: Feasibility, safety, and proof of concept in a stepwise study with in vitro, animal, and the first in-human application. Endoscopy. 2021 Nov;53(11):1162-1168.
13. Emura F, et al. Rio de Janeiro global consensus on landmarks, definitions, and classifications in Barrett’s esophagus: World Endoscopy Organization Delphi study. Gastroenterology. 2022 Jul;163(1):84-96.e2.
14. Singh RR, et al. Real-world evidence of safety and effectiveness of Barrett’s endoscopic therapy. Gastrointest Endosc. 2023 Aug;98(2):155-161.e1.
15. Wani S, et al. Recurrence Is rare following complete eradication of intestinal metaplasia in patients with Barrett’s esophagus and peaks at 18 months. Clin Gastroenterol Hepatol. 2020 Oct;18(11):2609-2617.e2.
16. Duvvuri A, et al. Mo1273 Location and pattern of recurrences in patients with Barrett’s esophagus after endoscopic therapy: A systematic review and critical analysis of the published literature. Gastrointest Endosc. 2020;91(6):AB410-1.
17. He T, et al. Location and appearance of dysplastic Barrett’s esophagus recurrence after endoscopic eradication therapy: No additional yield from random biopsy sampling neosquamous mucosa. Gastrointest Endosc. 2023 Nov;98(5):722-732.
May 2024 – ICYMI
Gastroenterology
January 2024
Hirano I, et al; ASCENT WORKING GROUP. Ascending to New Heights for Novel Therapeutics for Eosinophilic Esophagitis. Gastroenterology. 2024 Jan;166(1):1-10. doi: 10.1053/j.gastro.2023.09.004. Epub 2023 Sep 9. PMID: 37690772; PMCID: PMC10872872.
Åkerström JH, et al. Antireflux Surgery Versus Antireflux Medication and Risk of Esophageal Adenocarcinoma in Patients With Barrett’s Esophagus. Gastroenterology. 2024 Jan;166(1):132-138.e3. doi: 10.1053/j.gastro.2023.08.050. Epub 2023 Sep 9. PMID: 37690771.
Barnes EL, et al; AGA Clinical Guidelines Committee. AGA Clinical Practice Guideline on the Management of Pouchitis and Inflammatory Pouch Disorders. Gastroenterology. 2024 Jan;166(1):59-85. doi: 10.1053/j.gastro.2023.10.015. PMID: 38128971.
February 2024
Yoo HW, et al. Helicobacter pylori Treatment and Gastric Cancer Risk After Endoscopic Resection of Dysplasia: A Nationwide Cohort Study. Gastroenterology. 2024 Feb;166(2):313-322.e3. doi: 10.1053/j.gastro.2023.10.013. Epub 2023 Oct 18. PMID: 37863270.
Yang J, et al. High Soluble Fiber Promotes Colorectal Tumorigenesis Through Modulating Gut Microbiota and Metabolites in Mice. Gastroenterology. 2024 Feb;166(2):323-337.e7. doi: 10.1053/j.gastro.2023.10.012. Epub 2023 Oct 18. PMID: 37858797.
Young E, et al. Texture and Color Enhancement Imaging Improves Colonic Adenoma Detection: A Multicenter Randomized Controlled Trial. Gastroenterology. 2024 Feb;166(2):338-340.e3. doi: 10.1053/j.gastro.2023.10.008. Epub 2023 Oct 14. PMID: 37839498.
Clinical Gastroenterology and Hepatology
January 2024
Overbeek KA, et al; Dutch Familial Pancreatic Cancer Surveillance Study work group. Intraductal Papillary Mucinous Neoplasms in High-Risk Individuals: Incidence, Growth Rate, and Malignancy Risk. Clin Gastroenterol Hepatol. 2024 Jan;22(1):62-71.e7. doi: 10.1016/j.cgh.2023.03.035. Epub 2023 Apr 7. PMID: 37031711.
Reddy CA, et al. Achalasia is Strongly Associated With Eosinophilic Esophagitis and Other Allergic Disorders. Clin Gastroenterol Hepatol. 2024 Jan;22(1):34-41.e2. doi: 10.1016/j.cgh.2023.06.013. Epub 2023 Jun 28. PMID: 37391057; PMCID: PMC10753026.
Thiruvengadam NR, et al. The Clinical Impact and Cost-Effectiveness of Surveillance of Incidentally Detected Gastric Intestinal Metaplasia: A Microsimulation Analysis. Clin Gastroenterol Hepatol. 2024 Jan;22(1):51-61. doi: 10.1016/j.cgh.2023.05.028. Epub 2023 Jun 9. Erratum in: Clin Gastroenterol Hepatol. 2024 Jan 19;: PMID: 37302442.
February 2024
Goodoory VC, et al. Systematic Review and Meta-analysis: Efficacy of Mesalamine in Irritable Bowel Syndrome. Clin Gastroenterol Hepatol. 2024 Feb;22(2):243-251.e5. doi: 10.1016/j.cgh.2023.02.014. Epub 2023 Feb 27. PMID: 36858143.
Brenner DM, et al. Development and Current State of Digital Therapeutics for Irritable Bowel Syndrome. Clin Gastroenterol Hepatol. 2024 Feb;22(2):222-234. doi: 10.1016/j.cgh.2023.09.013. Epub 2023 Sep 22. PMID: 37743035.
Techniques and Innovations in Gastrointestinal Endoscopy
January 2024
Ramirez PR, et al. Gaps and Improvement Opportunities in Post-Colonoscopy Communication. Tech Innov Gastrointest Endosc. 2024 Jan;26(1):90-92. doi: 10.1016/j.tige.2023.10.001. Epub 2023 Oct 22.
Gonzaga ER, et al. Gastric Peroral Endoscopic Myotomy (G-POEM) for the Management of Gastroparesis. Tech Innov Gastrointest Endosc. 2024 Jan; 26(1): 46-55. doi: 10.1016/j.tige.2023.09.002. Epub 2023 Oct 13.
Wang D, et al. Sphincterotomy vs Sham Procedure for Pain Relief in Sphincter of Oddi Dysfunction: Systematic Review and Meta-analysis. Tech Innov Gastrointest Endosc. 2024 Jan;26(1): 30-37. doi: 10.1016/j.tige.2023.10.003. Epub 2023 Nov 8.
Gastro Hep Advances
January 2024
Adeniran E, et al. Intense and Sustained Alcohol Consumption Associated With Acute Pancreatitis Warrants Early Intervention. Gastro Hep Advances. 2024 Jan;3(1):61-63. doi: 10.1016/j.gastha.2023.08.017. Epub 2023 Sep 2.
Alkhouri N, et al. A Novel Prescription Digital Therapeutic Option for the Treatment of Metabolic Dysfunction-Associated Steatotic Liver Disease. Gastro Hep Advances. 2024 Jan;3(1): 9-16. doi: 10.1016/j.gastha.2023.08.019. Epub 2023 Oct 1.
Gastroenterology
January 2024
Hirano I, et al; ASCENT WORKING GROUP. Ascending to New Heights for Novel Therapeutics for Eosinophilic Esophagitis. Gastroenterology. 2024 Jan;166(1):1-10. doi: 10.1053/j.gastro.2023.09.004. Epub 2023 Sep 9. PMID: 37690772; PMCID: PMC10872872.
Åkerström JH, et al. Antireflux Surgery Versus Antireflux Medication and Risk of Esophageal Adenocarcinoma in Patients With Barrett’s Esophagus. Gastroenterology. 2024 Jan;166(1):132-138.e3. doi: 10.1053/j.gastro.2023.08.050. Epub 2023 Sep 9. PMID: 37690771.
Barnes EL, et al; AGA Clinical Guidelines Committee. AGA Clinical Practice Guideline on the Management of Pouchitis and Inflammatory Pouch Disorders. Gastroenterology. 2024 Jan;166(1):59-85. doi: 10.1053/j.gastro.2023.10.015. PMID: 38128971.
February 2024
Yoo HW, et al. Helicobacter pylori Treatment and Gastric Cancer Risk After Endoscopic Resection of Dysplasia: A Nationwide Cohort Study. Gastroenterology. 2024 Feb;166(2):313-322.e3. doi: 10.1053/j.gastro.2023.10.013. Epub 2023 Oct 18. PMID: 37863270.
Yang J, et al. High Soluble Fiber Promotes Colorectal Tumorigenesis Through Modulating Gut Microbiota and Metabolites in Mice. Gastroenterology. 2024 Feb;166(2):323-337.e7. doi: 10.1053/j.gastro.2023.10.012. Epub 2023 Oct 18. PMID: 37858797.
Young E, et al. Texture and Color Enhancement Imaging Improves Colonic Adenoma Detection: A Multicenter Randomized Controlled Trial. Gastroenterology. 2024 Feb;166(2):338-340.e3. doi: 10.1053/j.gastro.2023.10.008. Epub 2023 Oct 14. PMID: 37839498.
Clinical Gastroenterology and Hepatology
January 2024
Overbeek KA, et al; Dutch Familial Pancreatic Cancer Surveillance Study work group. Intraductal Papillary Mucinous Neoplasms in High-Risk Individuals: Incidence, Growth Rate, and Malignancy Risk. Clin Gastroenterol Hepatol. 2024 Jan;22(1):62-71.e7. doi: 10.1016/j.cgh.2023.03.035. Epub 2023 Apr 7. PMID: 37031711.
Reddy CA, et al. Achalasia is Strongly Associated With Eosinophilic Esophagitis and Other Allergic Disorders. Clin Gastroenterol Hepatol. 2024 Jan;22(1):34-41.e2. doi: 10.1016/j.cgh.2023.06.013. Epub 2023 Jun 28. PMID: 37391057; PMCID: PMC10753026.
Thiruvengadam NR, et al. The Clinical Impact and Cost-Effectiveness of Surveillance of Incidentally Detected Gastric Intestinal Metaplasia: A Microsimulation Analysis. Clin Gastroenterol Hepatol. 2024 Jan;22(1):51-61. doi: 10.1016/j.cgh.2023.05.028. Epub 2023 Jun 9. Erratum in: Clin Gastroenterol Hepatol. 2024 Jan 19;: PMID: 37302442.
February 2024
Goodoory VC, et al. Systematic Review and Meta-analysis: Efficacy of Mesalamine in Irritable Bowel Syndrome. Clin Gastroenterol Hepatol. 2024 Feb;22(2):243-251.e5. doi: 10.1016/j.cgh.2023.02.014. Epub 2023 Feb 27. PMID: 36858143.
Brenner DM, et al. Development and Current State of Digital Therapeutics for Irritable Bowel Syndrome. Clin Gastroenterol Hepatol. 2024 Feb;22(2):222-234. doi: 10.1016/j.cgh.2023.09.013. Epub 2023 Sep 22. PMID: 37743035.
Techniques and Innovations in Gastrointestinal Endoscopy
January 2024
Ramirez PR, et al. Gaps and Improvement Opportunities in Post-Colonoscopy Communication. Tech Innov Gastrointest Endosc. 2024 Jan;26(1):90-92. doi: 10.1016/j.tige.2023.10.001. Epub 2023 Oct 22.
Gonzaga ER, et al. Gastric Peroral Endoscopic Myotomy (G-POEM) for the Management of Gastroparesis. Tech Innov Gastrointest Endosc. 2024 Jan; 26(1): 46-55. doi: 10.1016/j.tige.2023.09.002. Epub 2023 Oct 13.
Wang D, et al. Sphincterotomy vs Sham Procedure for Pain Relief in Sphincter of Oddi Dysfunction: Systematic Review and Meta-analysis. Tech Innov Gastrointest Endosc. 2024 Jan;26(1): 30-37. doi: 10.1016/j.tige.2023.10.003. Epub 2023 Nov 8.
Gastro Hep Advances
January 2024
Adeniran E, et al. Intense and Sustained Alcohol Consumption Associated With Acute Pancreatitis Warrants Early Intervention. Gastro Hep Advances. 2024 Jan;3(1):61-63. doi: 10.1016/j.gastha.2023.08.017. Epub 2023 Sep 2.
Alkhouri N, et al. A Novel Prescription Digital Therapeutic Option for the Treatment of Metabolic Dysfunction-Associated Steatotic Liver Disease. Gastro Hep Advances. 2024 Jan;3(1): 9-16. doi: 10.1016/j.gastha.2023.08.019. Epub 2023 Oct 1.
Gastroenterology
January 2024
Hirano I, et al; ASCENT WORKING GROUP. Ascending to New Heights for Novel Therapeutics for Eosinophilic Esophagitis. Gastroenterology. 2024 Jan;166(1):1-10. doi: 10.1053/j.gastro.2023.09.004. Epub 2023 Sep 9. PMID: 37690772; PMCID: PMC10872872.
Åkerström JH, et al. Antireflux Surgery Versus Antireflux Medication and Risk of Esophageal Adenocarcinoma in Patients With Barrett’s Esophagus. Gastroenterology. 2024 Jan;166(1):132-138.e3. doi: 10.1053/j.gastro.2023.08.050. Epub 2023 Sep 9. PMID: 37690771.
Barnes EL, et al; AGA Clinical Guidelines Committee. AGA Clinical Practice Guideline on the Management of Pouchitis and Inflammatory Pouch Disorders. Gastroenterology. 2024 Jan;166(1):59-85. doi: 10.1053/j.gastro.2023.10.015. PMID: 38128971.
February 2024
Yoo HW, et al. Helicobacter pylori Treatment and Gastric Cancer Risk After Endoscopic Resection of Dysplasia: A Nationwide Cohort Study. Gastroenterology. 2024 Feb;166(2):313-322.e3. doi: 10.1053/j.gastro.2023.10.013. Epub 2023 Oct 18. PMID: 37863270.
Yang J, et al. High Soluble Fiber Promotes Colorectal Tumorigenesis Through Modulating Gut Microbiota and Metabolites in Mice. Gastroenterology. 2024 Feb;166(2):323-337.e7. doi: 10.1053/j.gastro.2023.10.012. Epub 2023 Oct 18. PMID: 37858797.
Young E, et al. Texture and Color Enhancement Imaging Improves Colonic Adenoma Detection: A Multicenter Randomized Controlled Trial. Gastroenterology. 2024 Feb;166(2):338-340.e3. doi: 10.1053/j.gastro.2023.10.008. Epub 2023 Oct 14. PMID: 37839498.
Clinical Gastroenterology and Hepatology
January 2024
Overbeek KA, et al; Dutch Familial Pancreatic Cancer Surveillance Study work group. Intraductal Papillary Mucinous Neoplasms in High-Risk Individuals: Incidence, Growth Rate, and Malignancy Risk. Clin Gastroenterol Hepatol. 2024 Jan;22(1):62-71.e7. doi: 10.1016/j.cgh.2023.03.035. Epub 2023 Apr 7. PMID: 37031711.
Reddy CA, et al. Achalasia is Strongly Associated With Eosinophilic Esophagitis and Other Allergic Disorders. Clin Gastroenterol Hepatol. 2024 Jan;22(1):34-41.e2. doi: 10.1016/j.cgh.2023.06.013. Epub 2023 Jun 28. PMID: 37391057; PMCID: PMC10753026.
Thiruvengadam NR, et al. The Clinical Impact and Cost-Effectiveness of Surveillance of Incidentally Detected Gastric Intestinal Metaplasia: A Microsimulation Analysis. Clin Gastroenterol Hepatol. 2024 Jan;22(1):51-61. doi: 10.1016/j.cgh.2023.05.028. Epub 2023 Jun 9. Erratum in: Clin Gastroenterol Hepatol. 2024 Jan 19;: PMID: 37302442.
February 2024
Goodoory VC, et al. Systematic Review and Meta-analysis: Efficacy of Mesalamine in Irritable Bowel Syndrome. Clin Gastroenterol Hepatol. 2024 Feb;22(2):243-251.e5. doi: 10.1016/j.cgh.2023.02.014. Epub 2023 Feb 27. PMID: 36858143.
Brenner DM, et al. Development and Current State of Digital Therapeutics for Irritable Bowel Syndrome. Clin Gastroenterol Hepatol. 2024 Feb;22(2):222-234. doi: 10.1016/j.cgh.2023.09.013. Epub 2023 Sep 22. PMID: 37743035.
Techniques and Innovations in Gastrointestinal Endoscopy
January 2024
Ramirez PR, et al. Gaps and Improvement Opportunities in Post-Colonoscopy Communication. Tech Innov Gastrointest Endosc. 2024 Jan;26(1):90-92. doi: 10.1016/j.tige.2023.10.001. Epub 2023 Oct 22.
Gonzaga ER, et al. Gastric Peroral Endoscopic Myotomy (G-POEM) for the Management of Gastroparesis. Tech Innov Gastrointest Endosc. 2024 Jan; 26(1): 46-55. doi: 10.1016/j.tige.2023.09.002. Epub 2023 Oct 13.
Wang D, et al. Sphincterotomy vs Sham Procedure for Pain Relief in Sphincter of Oddi Dysfunction: Systematic Review and Meta-analysis. Tech Innov Gastrointest Endosc. 2024 Jan;26(1): 30-37. doi: 10.1016/j.tige.2023.10.003. Epub 2023 Nov 8.
Gastro Hep Advances
January 2024
Adeniran E, et al. Intense and Sustained Alcohol Consumption Associated With Acute Pancreatitis Warrants Early Intervention. Gastro Hep Advances. 2024 Jan;3(1):61-63. doi: 10.1016/j.gastha.2023.08.017. Epub 2023 Sep 2.
Alkhouri N, et al. A Novel Prescription Digital Therapeutic Option for the Treatment of Metabolic Dysfunction-Associated Steatotic Liver Disease. Gastro Hep Advances. 2024 Jan;3(1): 9-16. doi: 10.1016/j.gastha.2023.08.019. Epub 2023 Oct 1.
GLP-1s May Increase Post-Endoscopy Aspiration Pneumonia Risk
, according to a new large population-based study.
In June 2023, the American Society of Anesthesiologists (ASA) recommended holding GLP-1 RAs before an endoscopic or surgical procedure to reduce the risk for complications associated with anesthesia and delayed stomach emptying.
In response, the American Gastroenterological Association (AGA) published a rapid clinical practice update in November 2023 that found insufficient evidence to support patients stopping the medications before endoscopic procedures.
“It is known that GLP-1 RAs significantly reduce the motility of the stomach and small bowel. As more and more patients are being started on GLP-1 RAs at higher doses and longer half-life, the question became whether the current recommended fasting durations are enough to reasonably assume the stomach is empty prior to procedures that require sedation,” said senior author Ali Rezaie, MD, medical director of the GI Motility Program at Cedars-Sinai Medical Center in Los Angeles.
“We wanted to see if these medications in fact increased the chance of aspiration before the ASA suggestion went into effect,” he said. “However, this is not an easy task, as aspiration is a rare event and a large sample size is needed to confidently answer that question. That is why we evaluated nearly 1 million cases.”
The study was published online in Gastroenterology.
Analyzing GLP-1 RA Use
Dr. Rezaie and colleagues conducted a population-based, retrospective cohort study of the TriNetX dataset, which includes 114 million deidentified individual health records from 80 healthcare organizations. The research team analyzed nearly 1 million records for adult patients between ages 21 and 70 who underwent upper and lower endoscopies between January 2018 and December 2020.
The researchers defined GLP-1 RA users as those who had the medication for more than 6 months and two or more refills within 6 months before the procedure. They adjusted for 59 factors that could affect gut motility or aspiration risks, such as obesity, numerous chronic diseases, and dozens of medications. The primary outcome was aspiration pneumonia within a month after the procedure.
Among 963,184 patients who underwent endoscopy, 46,935 (4.9%) were considered GLP-1 RA users. Among those, 20,099 GLP-1 RA users met the inclusion criteria and had their results compared with non-GLP-1 RA users.
After propensity score matching for the 59 potential confounders, GLP-1 RA use had a higher incidence rate of aspiration pneumonia (0.83% vs 0.63%) and was associated with a significantly higher risk for aspiration pneumonia, with a hazard ratio (HR) of 1.33.
An even higher risk was seen among patients with propofol-assisted endoscopies (HR, 1.49) but not among those without propofol (HR, 1.31).
In a subgroup analysis based on endoscopy type, an elevated risk was observed among patients who underwent upper endoscopy (HR, 1.82) and combined upper and lower endoscopy (HR, 2.26) but not lower endoscopy (HR, 0.56).
“The results were not necessarily surprising given the mechanism of action of GLP-1 RAs. However, for the first time, this was shown with a clinically relevant outcome, such as aspiration pneumonia,” Dr. Rezaie said. “Aspiration during sedation can have devastating consequences, and the 0.2% difference in risk of aspiration can have a significant effect on healthcare as well.”
More than 20 million endoscopies are performed across the United States annually. Based on the assumption that about 3% of those patients are taking GLP-1 RAs, about 1200 aspiration cases per year can be prevented by raising awareness, he said.
Considering Next Steps
The varying risk profiles observed with separate sedation and endoscopy types point to a need for more tailored guidance in managing GLP-1 RA use before a procedure, the study authors wrote.
Although holding the medications before endoscopy may disrupt diabetes management, the potential increased risk for aspiration could justify a change in practice, particularly for upper endoscopy and propofol-associated procedures, they added.
At the same time, additional studies are needed to understand the optimal drug withholding windows before endoscopies and other procedures, they concluded.
“We will need more data on what is the optimal duration of holding GLP-1 RAs,” Dr. Rezaie said. “But given our data and current ASA guidance, stopping these medications prior to elective procedures is the safe thing to do.”
For now, AGA guidance remains the same as offered in the November 2023 update, suggesting an individual approach for each patient on a GLP-1 RA rather than a “blanket statement” on how to manage all patients taking these medications.
“Overall, I believe that this study is important, but we require more high-level data to inform clinical decision-making regarding patients using GLP-1 receptor agonists prior to gastrointestinal endoscopy,” said Andrew Y. Wang, MD, AGAF, chief of gastroenterology and hepatology and director of interventional endoscopy at the University of Virginia in Charlottesville.
Dr. Wang, who wasn’t involved with this study, coauthored the AGA rapid clinical practice update. He and colleagues advised continuing with a procedure as planned for patients on GLP-1 RAs who followed standard preprocedure fasting instructions and didn’t have nausea, vomiting, dyspepsia, or abdominal distention.
Among patients with symptoms that suggest retained gastric contents, rapid sequence intubation may be considered, though it may not be possible in ambulatory or office-based endoscopy settings, Dr. Wang and colleagues wrote. As another option in lieu of stopping GLP-1 RAs, patients can be placed on a liquid diet for 1 day before the procedure.
“While this study found a signal suggesting that patients using GLP-1 RAs had an increased risk of aspiration pneumonia within 1 month following upper endoscopy or combined upper and lower endoscopy, it does not inform us if having patients stop GLP-1 RAs before endoscopic procedures — especially for a single dose — will mitigate this potential risk,” Dr. Wang said.
“It was also interesting that these investigators found that patients taking GLP-1 RAs who underwent lower endoscopy alone were not at increased risk for aspiration pneumonia,” Dr. Wang noted.
The authors didn’t report a funding source and disclosed no potential conflicts. Dr. Wang reported no relevant disclosures.
A version of this article appeared on Medscape.com.
, according to a new large population-based study.
In June 2023, the American Society of Anesthesiologists (ASA) recommended holding GLP-1 RAs before an endoscopic or surgical procedure to reduce the risk for complications associated with anesthesia and delayed stomach emptying.
In response, the American Gastroenterological Association (AGA) published a rapid clinical practice update in November 2023 that found insufficient evidence to support patients stopping the medications before endoscopic procedures.
“It is known that GLP-1 RAs significantly reduce the motility of the stomach and small bowel. As more and more patients are being started on GLP-1 RAs at higher doses and longer half-life, the question became whether the current recommended fasting durations are enough to reasonably assume the stomach is empty prior to procedures that require sedation,” said senior author Ali Rezaie, MD, medical director of the GI Motility Program at Cedars-Sinai Medical Center in Los Angeles.
“We wanted to see if these medications in fact increased the chance of aspiration before the ASA suggestion went into effect,” he said. “However, this is not an easy task, as aspiration is a rare event and a large sample size is needed to confidently answer that question. That is why we evaluated nearly 1 million cases.”
The study was published online in Gastroenterology.
Analyzing GLP-1 RA Use
Dr. Rezaie and colleagues conducted a population-based, retrospective cohort study of the TriNetX dataset, which includes 114 million deidentified individual health records from 80 healthcare organizations. The research team analyzed nearly 1 million records for adult patients between ages 21 and 70 who underwent upper and lower endoscopies between January 2018 and December 2020.
The researchers defined GLP-1 RA users as those who had the medication for more than 6 months and two or more refills within 6 months before the procedure. They adjusted for 59 factors that could affect gut motility or aspiration risks, such as obesity, numerous chronic diseases, and dozens of medications. The primary outcome was aspiration pneumonia within a month after the procedure.
Among 963,184 patients who underwent endoscopy, 46,935 (4.9%) were considered GLP-1 RA users. Among those, 20,099 GLP-1 RA users met the inclusion criteria and had their results compared with non-GLP-1 RA users.
After propensity score matching for the 59 potential confounders, GLP-1 RA use had a higher incidence rate of aspiration pneumonia (0.83% vs 0.63%) and was associated with a significantly higher risk for aspiration pneumonia, with a hazard ratio (HR) of 1.33.
An even higher risk was seen among patients with propofol-assisted endoscopies (HR, 1.49) but not among those without propofol (HR, 1.31).
In a subgroup analysis based on endoscopy type, an elevated risk was observed among patients who underwent upper endoscopy (HR, 1.82) and combined upper and lower endoscopy (HR, 2.26) but not lower endoscopy (HR, 0.56).
“The results were not necessarily surprising given the mechanism of action of GLP-1 RAs. However, for the first time, this was shown with a clinically relevant outcome, such as aspiration pneumonia,” Dr. Rezaie said. “Aspiration during sedation can have devastating consequences, and the 0.2% difference in risk of aspiration can have a significant effect on healthcare as well.”
More than 20 million endoscopies are performed across the United States annually. Based on the assumption that about 3% of those patients are taking GLP-1 RAs, about 1200 aspiration cases per year can be prevented by raising awareness, he said.
Considering Next Steps
The varying risk profiles observed with separate sedation and endoscopy types point to a need for more tailored guidance in managing GLP-1 RA use before a procedure, the study authors wrote.
Although holding the medications before endoscopy may disrupt diabetes management, the potential increased risk for aspiration could justify a change in practice, particularly for upper endoscopy and propofol-associated procedures, they added.
At the same time, additional studies are needed to understand the optimal drug withholding windows before endoscopies and other procedures, they concluded.
“We will need more data on what is the optimal duration of holding GLP-1 RAs,” Dr. Rezaie said. “But given our data and current ASA guidance, stopping these medications prior to elective procedures is the safe thing to do.”
For now, AGA guidance remains the same as offered in the November 2023 update, suggesting an individual approach for each patient on a GLP-1 RA rather than a “blanket statement” on how to manage all patients taking these medications.
“Overall, I believe that this study is important, but we require more high-level data to inform clinical decision-making regarding patients using GLP-1 receptor agonists prior to gastrointestinal endoscopy,” said Andrew Y. Wang, MD, AGAF, chief of gastroenterology and hepatology and director of interventional endoscopy at the University of Virginia in Charlottesville.
Dr. Wang, who wasn’t involved with this study, coauthored the AGA rapid clinical practice update. He and colleagues advised continuing with a procedure as planned for patients on GLP-1 RAs who followed standard preprocedure fasting instructions and didn’t have nausea, vomiting, dyspepsia, or abdominal distention.
Among patients with symptoms that suggest retained gastric contents, rapid sequence intubation may be considered, though it may not be possible in ambulatory or office-based endoscopy settings, Dr. Wang and colleagues wrote. As another option in lieu of stopping GLP-1 RAs, patients can be placed on a liquid diet for 1 day before the procedure.
“While this study found a signal suggesting that patients using GLP-1 RAs had an increased risk of aspiration pneumonia within 1 month following upper endoscopy or combined upper and lower endoscopy, it does not inform us if having patients stop GLP-1 RAs before endoscopic procedures — especially for a single dose — will mitigate this potential risk,” Dr. Wang said.
“It was also interesting that these investigators found that patients taking GLP-1 RAs who underwent lower endoscopy alone were not at increased risk for aspiration pneumonia,” Dr. Wang noted.
The authors didn’t report a funding source and disclosed no potential conflicts. Dr. Wang reported no relevant disclosures.
A version of this article appeared on Medscape.com.
, according to a new large population-based study.
In June 2023, the American Society of Anesthesiologists (ASA) recommended holding GLP-1 RAs before an endoscopic or surgical procedure to reduce the risk for complications associated with anesthesia and delayed stomach emptying.
In response, the American Gastroenterological Association (AGA) published a rapid clinical practice update in November 2023 that found insufficient evidence to support patients stopping the medications before endoscopic procedures.
“It is known that GLP-1 RAs significantly reduce the motility of the stomach and small bowel. As more and more patients are being started on GLP-1 RAs at higher doses and longer half-life, the question became whether the current recommended fasting durations are enough to reasonably assume the stomach is empty prior to procedures that require sedation,” said senior author Ali Rezaie, MD, medical director of the GI Motility Program at Cedars-Sinai Medical Center in Los Angeles.
“We wanted to see if these medications in fact increased the chance of aspiration before the ASA suggestion went into effect,” he said. “However, this is not an easy task, as aspiration is a rare event and a large sample size is needed to confidently answer that question. That is why we evaluated nearly 1 million cases.”
The study was published online in Gastroenterology.
Analyzing GLP-1 RA Use
Dr. Rezaie and colleagues conducted a population-based, retrospective cohort study of the TriNetX dataset, which includes 114 million deidentified individual health records from 80 healthcare organizations. The research team analyzed nearly 1 million records for adult patients between ages 21 and 70 who underwent upper and lower endoscopies between January 2018 and December 2020.
The researchers defined GLP-1 RA users as those who had the medication for more than 6 months and two or more refills within 6 months before the procedure. They adjusted for 59 factors that could affect gut motility or aspiration risks, such as obesity, numerous chronic diseases, and dozens of medications. The primary outcome was aspiration pneumonia within a month after the procedure.
Among 963,184 patients who underwent endoscopy, 46,935 (4.9%) were considered GLP-1 RA users. Among those, 20,099 GLP-1 RA users met the inclusion criteria and had their results compared with non-GLP-1 RA users.
After propensity score matching for the 59 potential confounders, GLP-1 RA use had a higher incidence rate of aspiration pneumonia (0.83% vs 0.63%) and was associated with a significantly higher risk for aspiration pneumonia, with a hazard ratio (HR) of 1.33.
An even higher risk was seen among patients with propofol-assisted endoscopies (HR, 1.49) but not among those without propofol (HR, 1.31).
In a subgroup analysis based on endoscopy type, an elevated risk was observed among patients who underwent upper endoscopy (HR, 1.82) and combined upper and lower endoscopy (HR, 2.26) but not lower endoscopy (HR, 0.56).
“The results were not necessarily surprising given the mechanism of action of GLP-1 RAs. However, for the first time, this was shown with a clinically relevant outcome, such as aspiration pneumonia,” Dr. Rezaie said. “Aspiration during sedation can have devastating consequences, and the 0.2% difference in risk of aspiration can have a significant effect on healthcare as well.”
More than 20 million endoscopies are performed across the United States annually. Based on the assumption that about 3% of those patients are taking GLP-1 RAs, about 1200 aspiration cases per year can be prevented by raising awareness, he said.
Considering Next Steps
The varying risk profiles observed with separate sedation and endoscopy types point to a need for more tailored guidance in managing GLP-1 RA use before a procedure, the study authors wrote.
Although holding the medications before endoscopy may disrupt diabetes management, the potential increased risk for aspiration could justify a change in practice, particularly for upper endoscopy and propofol-associated procedures, they added.
At the same time, additional studies are needed to understand the optimal drug withholding windows before endoscopies and other procedures, they concluded.
“We will need more data on what is the optimal duration of holding GLP-1 RAs,” Dr. Rezaie said. “But given our data and current ASA guidance, stopping these medications prior to elective procedures is the safe thing to do.”
For now, AGA guidance remains the same as offered in the November 2023 update, suggesting an individual approach for each patient on a GLP-1 RA rather than a “blanket statement” on how to manage all patients taking these medications.
“Overall, I believe that this study is important, but we require more high-level data to inform clinical decision-making regarding patients using GLP-1 receptor agonists prior to gastrointestinal endoscopy,” said Andrew Y. Wang, MD, AGAF, chief of gastroenterology and hepatology and director of interventional endoscopy at the University of Virginia in Charlottesville.
Dr. Wang, who wasn’t involved with this study, coauthored the AGA rapid clinical practice update. He and colleagues advised continuing with a procedure as planned for patients on GLP-1 RAs who followed standard preprocedure fasting instructions and didn’t have nausea, vomiting, dyspepsia, or abdominal distention.
Among patients with symptoms that suggest retained gastric contents, rapid sequence intubation may be considered, though it may not be possible in ambulatory or office-based endoscopy settings, Dr. Wang and colleagues wrote. As another option in lieu of stopping GLP-1 RAs, patients can be placed on a liquid diet for 1 day before the procedure.
“While this study found a signal suggesting that patients using GLP-1 RAs had an increased risk of aspiration pneumonia within 1 month following upper endoscopy or combined upper and lower endoscopy, it does not inform us if having patients stop GLP-1 RAs before endoscopic procedures — especially for a single dose — will mitigate this potential risk,” Dr. Wang said.
“It was also interesting that these investigators found that patients taking GLP-1 RAs who underwent lower endoscopy alone were not at increased risk for aspiration pneumonia,” Dr. Wang noted.
The authors didn’t report a funding source and disclosed no potential conflicts. Dr. Wang reported no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM GASTROENTEROLOGY
AGA Defines Diagnostic, Treatment Approach to Cannabinoid Hyperemesis Syndrome
.
CHS, which is triggered by chronic cannabis usage and manifests with GI and autonomic symptoms, is on the rise in the United States, yet underdiagnosis remains a challenge and clinical data are scarce, reported lead update panelist Alberto Rubio Tapia, MD, of Cleveland Clinic, Cleveland, Ohio, and colleagues.
“Although cannabis use has been reported for many decades, some of its unique adverse effects of nausea, vomiting, and abdominal pain, termed CHS, were noted relatively recently,” the panelists wrote in Gastroenterology. “The objective of this article was to help practitioners define the appropriate approach to the diagnosis and management of CHS.”
According to the update, the typical CHS patient is male with a years-long history of daily or near-daily cannabis use. Paradoxically, while cannabis use drives this condition, some patients with CHS report that cannabis use relieves their symptoms.The update describes CHS as a subtype of cyclical vomiting syndrome (CVS), and offers diagnostic criteria for CHS, reproduced below verbatim:
- Clinical features: stereotypical episodic vomiting resembling CVS in terms of onset, with frequency 3 or more times annually;
- Cannabis use patterns: duration of cannabis use more than 1 year before symptom onset; frequency more than 4 times per week, on average;
- Cannabis cessation: resolution of symptoms after a period of abstinence from cannabis use for at least 6 months, or at least equal to the total duration of 3 typical vomiting cycles in that patient.
As CHS is a subtype of CVS, the update also provides an outline and management guide for this broader condition, which is characterized by four phases: inter-episodic, prodromal, emetic, and recovery.
During the inter-episodic phase, patients will have minimal or no symptoms, although almost one third will describe dyspepsia or nausea. Prophylactic medications in this period include tricyclics, mitochondrial supplements like CoQ10 and vitamin B12, NK1 antagonists, and anticonvulsants.
The prodromal phase is characterized by abdominal pain and nausea with a duration of 30-90 minutes. During this time patients may have autonomic symptoms like sweating and feeling hot or cold. Psychological symptoms may include feelings of panic and being “out of control.” Abortive medications are appropriate during this period, according to the update, like triptans and antiemetics.
Next comes the emetic phase, in which patients exhibit “relentless vomiting,” retching, abdominal pain, neurological symptoms and extreme thirst. Because an empty stomach may provide relief, inducing emesis may be considered, along with rest in a quiet dark room and supportive care.
Finally, the vomiting subsides during the recovery phase, when it is possible to restart oral intake and resume normal activities.
While this framework may be useful when managing patients with CHS, intervention should be centered around cannabis cessation, according to the update.
“For long-term management, counseling to achieve marijuana cessation and tricyclic antidepressants, such as amitriptyline, are the mainstay of therapy,” Dr. Rubio Tapia and colleagues wrote.
Advising patients to stop cannabis “cold turkey” is not recommended, they added, as this may bring on withdrawal symptoms, and it tends to be ineffective in this population, which has a high recidivism rate.
“Co-management with a psychologist or psychiatrist may be helpful for patients who have a lack of response to standard therapies or extensive psychiatric comorbidity,” the panelists wrote. “Anxiety and depression are very common associated conditions.”
Dr. Rubio Tapia and colleagues concluded with a call for more research.
“Further understanding of CHS pathophysiology and evidence-based therapies are urgently needed,” they wrote.
This update was commissioned and approved by the AGA. The update panelists disclosed relationships with Evoke Pharma, RedHill Biopharma, Takeda, and others.
.
CHS, which is triggered by chronic cannabis usage and manifests with GI and autonomic symptoms, is on the rise in the United States, yet underdiagnosis remains a challenge and clinical data are scarce, reported lead update panelist Alberto Rubio Tapia, MD, of Cleveland Clinic, Cleveland, Ohio, and colleagues.
“Although cannabis use has been reported for many decades, some of its unique adverse effects of nausea, vomiting, and abdominal pain, termed CHS, were noted relatively recently,” the panelists wrote in Gastroenterology. “The objective of this article was to help practitioners define the appropriate approach to the diagnosis and management of CHS.”
According to the update, the typical CHS patient is male with a years-long history of daily or near-daily cannabis use. Paradoxically, while cannabis use drives this condition, some patients with CHS report that cannabis use relieves their symptoms.The update describes CHS as a subtype of cyclical vomiting syndrome (CVS), and offers diagnostic criteria for CHS, reproduced below verbatim:
- Clinical features: stereotypical episodic vomiting resembling CVS in terms of onset, with frequency 3 or more times annually;
- Cannabis use patterns: duration of cannabis use more than 1 year before symptom onset; frequency more than 4 times per week, on average;
- Cannabis cessation: resolution of symptoms after a period of abstinence from cannabis use for at least 6 months, or at least equal to the total duration of 3 typical vomiting cycles in that patient.
As CHS is a subtype of CVS, the update also provides an outline and management guide for this broader condition, which is characterized by four phases: inter-episodic, prodromal, emetic, and recovery.
During the inter-episodic phase, patients will have minimal or no symptoms, although almost one third will describe dyspepsia or nausea. Prophylactic medications in this period include tricyclics, mitochondrial supplements like CoQ10 and vitamin B12, NK1 antagonists, and anticonvulsants.
The prodromal phase is characterized by abdominal pain and nausea with a duration of 30-90 minutes. During this time patients may have autonomic symptoms like sweating and feeling hot or cold. Psychological symptoms may include feelings of panic and being “out of control.” Abortive medications are appropriate during this period, according to the update, like triptans and antiemetics.
Next comes the emetic phase, in which patients exhibit “relentless vomiting,” retching, abdominal pain, neurological symptoms and extreme thirst. Because an empty stomach may provide relief, inducing emesis may be considered, along with rest in a quiet dark room and supportive care.
Finally, the vomiting subsides during the recovery phase, when it is possible to restart oral intake and resume normal activities.
While this framework may be useful when managing patients with CHS, intervention should be centered around cannabis cessation, according to the update.
“For long-term management, counseling to achieve marijuana cessation and tricyclic antidepressants, such as amitriptyline, are the mainstay of therapy,” Dr. Rubio Tapia and colleagues wrote.
Advising patients to stop cannabis “cold turkey” is not recommended, they added, as this may bring on withdrawal symptoms, and it tends to be ineffective in this population, which has a high recidivism rate.
“Co-management with a psychologist or psychiatrist may be helpful for patients who have a lack of response to standard therapies or extensive psychiatric comorbidity,” the panelists wrote. “Anxiety and depression are very common associated conditions.”
Dr. Rubio Tapia and colleagues concluded with a call for more research.
“Further understanding of CHS pathophysiology and evidence-based therapies are urgently needed,” they wrote.
This update was commissioned and approved by the AGA. The update panelists disclosed relationships with Evoke Pharma, RedHill Biopharma, Takeda, and others.
.
CHS, which is triggered by chronic cannabis usage and manifests with GI and autonomic symptoms, is on the rise in the United States, yet underdiagnosis remains a challenge and clinical data are scarce, reported lead update panelist Alberto Rubio Tapia, MD, of Cleveland Clinic, Cleveland, Ohio, and colleagues.
“Although cannabis use has been reported for many decades, some of its unique adverse effects of nausea, vomiting, and abdominal pain, termed CHS, were noted relatively recently,” the panelists wrote in Gastroenterology. “The objective of this article was to help practitioners define the appropriate approach to the diagnosis and management of CHS.”
According to the update, the typical CHS patient is male with a years-long history of daily or near-daily cannabis use. Paradoxically, while cannabis use drives this condition, some patients with CHS report that cannabis use relieves their symptoms.The update describes CHS as a subtype of cyclical vomiting syndrome (CVS), and offers diagnostic criteria for CHS, reproduced below verbatim:
- Clinical features: stereotypical episodic vomiting resembling CVS in terms of onset, with frequency 3 or more times annually;
- Cannabis use patterns: duration of cannabis use more than 1 year before symptom onset; frequency more than 4 times per week, on average;
- Cannabis cessation: resolution of symptoms after a period of abstinence from cannabis use for at least 6 months, or at least equal to the total duration of 3 typical vomiting cycles in that patient.
As CHS is a subtype of CVS, the update also provides an outline and management guide for this broader condition, which is characterized by four phases: inter-episodic, prodromal, emetic, and recovery.
During the inter-episodic phase, patients will have minimal or no symptoms, although almost one third will describe dyspepsia or nausea. Prophylactic medications in this period include tricyclics, mitochondrial supplements like CoQ10 and vitamin B12, NK1 antagonists, and anticonvulsants.
The prodromal phase is characterized by abdominal pain and nausea with a duration of 30-90 minutes. During this time patients may have autonomic symptoms like sweating and feeling hot or cold. Psychological symptoms may include feelings of panic and being “out of control.” Abortive medications are appropriate during this period, according to the update, like triptans and antiemetics.
Next comes the emetic phase, in which patients exhibit “relentless vomiting,” retching, abdominal pain, neurological symptoms and extreme thirst. Because an empty stomach may provide relief, inducing emesis may be considered, along with rest in a quiet dark room and supportive care.
Finally, the vomiting subsides during the recovery phase, when it is possible to restart oral intake and resume normal activities.
While this framework may be useful when managing patients with CHS, intervention should be centered around cannabis cessation, according to the update.
“For long-term management, counseling to achieve marijuana cessation and tricyclic antidepressants, such as amitriptyline, are the mainstay of therapy,” Dr. Rubio Tapia and colleagues wrote.
Advising patients to stop cannabis “cold turkey” is not recommended, they added, as this may bring on withdrawal symptoms, and it tends to be ineffective in this population, which has a high recidivism rate.
“Co-management with a psychologist or psychiatrist may be helpful for patients who have a lack of response to standard therapies or extensive psychiatric comorbidity,” the panelists wrote. “Anxiety and depression are very common associated conditions.”
Dr. Rubio Tapia and colleagues concluded with a call for more research.
“Further understanding of CHS pathophysiology and evidence-based therapies are urgently needed,” they wrote.
This update was commissioned and approved by the AGA. The update panelists disclosed relationships with Evoke Pharma, RedHill Biopharma, Takeda, and others.
FROM GASTROENTEROLOGY
AGA Clinical Practice Update Describes High-Quality Upper Endoscopy
The update, authored by Satish Nagula, MD, of Icahn School of Medicine at Mount Sinai, New York, NY, and colleagues, includes nine pieces of best practice advice that address procedure optimization, evaluation of suspected premalignancy, and postprocedure follow-up evaluation.
“Defining what constitutes a high-quality esophagogastroduodenoscopy (EGD) poses somewhat of a challenge because the spectrum of indications and the breadth of benign and (pre)malignant disease pathology in the upper GI tract is very broad,” the update panelists wrote in Clinical Gastroenterology and Hepatology. “Standardizing the measures defining a high-quality upper endoscopic examination is one of the first steps for assessing quality.”
Preprocedure Recommendations
Dr. Nagula and colleagues first emphasized that EGD should be performed for an appropriate indication, citing a recent meta-analysis that found 21.7% of upper endoscopy procedures were performed for an inappropriate indication. Of note, diagnostic yields were 42% higher in procedures performed for an appropriate indication.
After ensuring an appropriate indication, the update also encourages clinicians to inform patients of the various benefits, risks, and alternatives of the procedure prior to providing consent.
Intraprocedure Recommendations
During the procedure, endoscopists should take several steps to ensure optimal visualization of tissues, according to the update.
First, a high-definition (HD) white-light endoscopy system should be employed.
“Although HD imaging is a standard feature of newer-generation endoscopes, legacy standard-definition scopes remain in use,” Dr. Nagula and colleagues noted. “Moreover, to provide true HD image resolution, each component of the system (eg, the endoscope video chip, the processor, the monitor, and transmission cables) must be HD compatible.”
This HD-compatible system should be coupled with image-enhancing technology to further improve lesion detection. In Barrett’s esophagus, the panelists noted, image enhancement can improve lesion detection as much as 20%.
They predicted that AI-assisted software may boost detection rates even higher: “Computer-aided detection and computer-aided diagnosis systems for upper endoscopy are still in the early phases of development but do show similar promise for improving the detection and characterization of upper GI tract neoplasia.”
Beyond selection of best available technologies, the update encourages more fundamental strategies to improve visualization, including mucosal cleansing and insufflation, with sufficient time spent inspecting the foregut mucosa via anterograde and retroflexed views.
Where appropriate, standardized biopsy protocols should be followed to evaluate and manage foregut conditions.
Postprocedure Recommendations
After the procedure, endoscopists should offer patients management recommendations based on the endoscopic findings and, if necessary, notify them that more recommendations may be forthcoming based on histopathology results, according to the update.
Similarly, endoscopists should follow established surveillance intervals for future procedures, with modifications made as needed, based on histopathology findings.
Document, Document, Document
Throughout the update, Dr. Nagula and colleagues repeatedly emphasize the importance of documentation, from preprocedural discussions with patients through planned surveillance schedules.
However, the recommendations are clear about “weighing the practical implications” of “onerous” documentation, particularly photodocumentation requirements. For instance, the authors note that “there are some scenarios in which more rigorous photodocumentation standards during upper endoscopy should be considered, such as patients with risk factors for neoplasia,” but at the very least “photodocumentation of any suspicious abnormalities, ideally with annotations, is strongly advised.”
Moving Toward Quality Standardization for Upper Endoscopy
“These best practice advice statements are intended to improve measurable clinical, patient-reported, and economic healthcare outcomes and are not meant to put an additional burden on endoscopists,” the panelists wrote. “Ideally, future research will set threshold indicators of adherence to these best practices that optimally are associated with these aforementioned objective outcomes.”
This update was commissioned and approved by AGA. The update panelists disclosed relationships with Covidien LP, Fujifilm USA, Mahana Therapeutics, and others.
The update, authored by Satish Nagula, MD, of Icahn School of Medicine at Mount Sinai, New York, NY, and colleagues, includes nine pieces of best practice advice that address procedure optimization, evaluation of suspected premalignancy, and postprocedure follow-up evaluation.
“Defining what constitutes a high-quality esophagogastroduodenoscopy (EGD) poses somewhat of a challenge because the spectrum of indications and the breadth of benign and (pre)malignant disease pathology in the upper GI tract is very broad,” the update panelists wrote in Clinical Gastroenterology and Hepatology. “Standardizing the measures defining a high-quality upper endoscopic examination is one of the first steps for assessing quality.”
Preprocedure Recommendations
Dr. Nagula and colleagues first emphasized that EGD should be performed for an appropriate indication, citing a recent meta-analysis that found 21.7% of upper endoscopy procedures were performed for an inappropriate indication. Of note, diagnostic yields were 42% higher in procedures performed for an appropriate indication.
After ensuring an appropriate indication, the update also encourages clinicians to inform patients of the various benefits, risks, and alternatives of the procedure prior to providing consent.
Intraprocedure Recommendations
During the procedure, endoscopists should take several steps to ensure optimal visualization of tissues, according to the update.
First, a high-definition (HD) white-light endoscopy system should be employed.
“Although HD imaging is a standard feature of newer-generation endoscopes, legacy standard-definition scopes remain in use,” Dr. Nagula and colleagues noted. “Moreover, to provide true HD image resolution, each component of the system (eg, the endoscope video chip, the processor, the monitor, and transmission cables) must be HD compatible.”
This HD-compatible system should be coupled with image-enhancing technology to further improve lesion detection. In Barrett’s esophagus, the panelists noted, image enhancement can improve lesion detection as much as 20%.
They predicted that AI-assisted software may boost detection rates even higher: “Computer-aided detection and computer-aided diagnosis systems for upper endoscopy are still in the early phases of development but do show similar promise for improving the detection and characterization of upper GI tract neoplasia.”
Beyond selection of best available technologies, the update encourages more fundamental strategies to improve visualization, including mucosal cleansing and insufflation, with sufficient time spent inspecting the foregut mucosa via anterograde and retroflexed views.
Where appropriate, standardized biopsy protocols should be followed to evaluate and manage foregut conditions.
Postprocedure Recommendations
After the procedure, endoscopists should offer patients management recommendations based on the endoscopic findings and, if necessary, notify them that more recommendations may be forthcoming based on histopathology results, according to the update.
Similarly, endoscopists should follow established surveillance intervals for future procedures, with modifications made as needed, based on histopathology findings.
Document, Document, Document
Throughout the update, Dr. Nagula and colleagues repeatedly emphasize the importance of documentation, from preprocedural discussions with patients through planned surveillance schedules.
However, the recommendations are clear about “weighing the practical implications” of “onerous” documentation, particularly photodocumentation requirements. For instance, the authors note that “there are some scenarios in which more rigorous photodocumentation standards during upper endoscopy should be considered, such as patients with risk factors for neoplasia,” but at the very least “photodocumentation of any suspicious abnormalities, ideally with annotations, is strongly advised.”
Moving Toward Quality Standardization for Upper Endoscopy
“These best practice advice statements are intended to improve measurable clinical, patient-reported, and economic healthcare outcomes and are not meant to put an additional burden on endoscopists,” the panelists wrote. “Ideally, future research will set threshold indicators of adherence to these best practices that optimally are associated with these aforementioned objective outcomes.”
This update was commissioned and approved by AGA. The update panelists disclosed relationships with Covidien LP, Fujifilm USA, Mahana Therapeutics, and others.
The update, authored by Satish Nagula, MD, of Icahn School of Medicine at Mount Sinai, New York, NY, and colleagues, includes nine pieces of best practice advice that address procedure optimization, evaluation of suspected premalignancy, and postprocedure follow-up evaluation.
“Defining what constitutes a high-quality esophagogastroduodenoscopy (EGD) poses somewhat of a challenge because the spectrum of indications and the breadth of benign and (pre)malignant disease pathology in the upper GI tract is very broad,” the update panelists wrote in Clinical Gastroenterology and Hepatology. “Standardizing the measures defining a high-quality upper endoscopic examination is one of the first steps for assessing quality.”
Preprocedure Recommendations
Dr. Nagula and colleagues first emphasized that EGD should be performed for an appropriate indication, citing a recent meta-analysis that found 21.7% of upper endoscopy procedures were performed for an inappropriate indication. Of note, diagnostic yields were 42% higher in procedures performed for an appropriate indication.
After ensuring an appropriate indication, the update also encourages clinicians to inform patients of the various benefits, risks, and alternatives of the procedure prior to providing consent.
Intraprocedure Recommendations
During the procedure, endoscopists should take several steps to ensure optimal visualization of tissues, according to the update.
First, a high-definition (HD) white-light endoscopy system should be employed.
“Although HD imaging is a standard feature of newer-generation endoscopes, legacy standard-definition scopes remain in use,” Dr. Nagula and colleagues noted. “Moreover, to provide true HD image resolution, each component of the system (eg, the endoscope video chip, the processor, the monitor, and transmission cables) must be HD compatible.”
This HD-compatible system should be coupled with image-enhancing technology to further improve lesion detection. In Barrett’s esophagus, the panelists noted, image enhancement can improve lesion detection as much as 20%.
They predicted that AI-assisted software may boost detection rates even higher: “Computer-aided detection and computer-aided diagnosis systems for upper endoscopy are still in the early phases of development but do show similar promise for improving the detection and characterization of upper GI tract neoplasia.”
Beyond selection of best available technologies, the update encourages more fundamental strategies to improve visualization, including mucosal cleansing and insufflation, with sufficient time spent inspecting the foregut mucosa via anterograde and retroflexed views.
Where appropriate, standardized biopsy protocols should be followed to evaluate and manage foregut conditions.
Postprocedure Recommendations
After the procedure, endoscopists should offer patients management recommendations based on the endoscopic findings and, if necessary, notify them that more recommendations may be forthcoming based on histopathology results, according to the update.
Similarly, endoscopists should follow established surveillance intervals for future procedures, with modifications made as needed, based on histopathology findings.
Document, Document, Document
Throughout the update, Dr. Nagula and colleagues repeatedly emphasize the importance of documentation, from preprocedural discussions with patients through planned surveillance schedules.
However, the recommendations are clear about “weighing the practical implications” of “onerous” documentation, particularly photodocumentation requirements. For instance, the authors note that “there are some scenarios in which more rigorous photodocumentation standards during upper endoscopy should be considered, such as patients with risk factors for neoplasia,” but at the very least “photodocumentation of any suspicious abnormalities, ideally with annotations, is strongly advised.”
Moving Toward Quality Standardization for Upper Endoscopy
“These best practice advice statements are intended to improve measurable clinical, patient-reported, and economic healthcare outcomes and are not meant to put an additional burden on endoscopists,” the panelists wrote. “Ideally, future research will set threshold indicators of adherence to these best practices that optimally are associated with these aforementioned objective outcomes.”
This update was commissioned and approved by AGA. The update panelists disclosed relationships with Covidien LP, Fujifilm USA, Mahana Therapeutics, and others.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY