User login
Acute respiratory distress syndrome: Implications of recent studies
Continued progress in understanding the pathophysiology of acute respiratory distress syndrome (ARDS) is translating into changes in the way we diagnose and manage it. Over the past 20 years, low tidal volume,1 positive end-expiratory pressure (PEEP),2 and fluid restriction3 have become the standard of care. A multidisciplinary approach, including targeted use of sedatives, early mobilization, and protocols for weaning from the ventilator, has also brought about substantial changes in ARDS management and its outcomes.4–6
In this article, we review the most relevant articles about ARDS in the last 5 years. We include the new definition of ARDS and studies of ventilatory and nonventilatory therapies that have implications in managing patients with ARDS.
A STANDARDIZED APPROACH
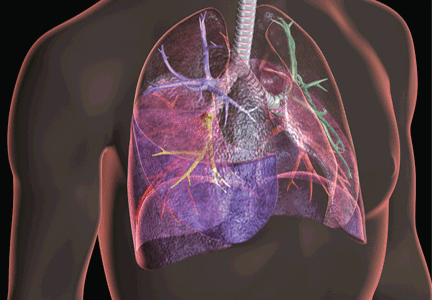
ARDS is characterized by damage to the alveolar architecture, severe hypoxemia, and bilateral parenchymal opacities.
The working definition of ARDS developed in 1994 by the American-European Consensus Conference (AECC) was the basis for enrollment in most of the landmark trials and observational studies over the past 20 years.7,8 However, it was limited in its reliability and validity.
An updated definition
In 2011, the ARDS Definition Task Force, using a novel consensus process, updated the ARDS definition,9 focusing on its feasibility, reliability, and validity in predicting response to therapies and outcomes in ARDS. This new “Berlin” definition is not substantially different from the old, but defines the criteria more specifically:
- Bilateral opacities, unexplained by nodules, atelectasis, or effusion, on chest radiography or computed tomography
- New or worsening respiratory symptoms, or a clinical insult associated with ARDS within 7 days of diagnosis
- Objective assessment of cardiac function (eg, with echocardiography) to exclude cardiogenic pulmonary edema
- Hypoxemia, with a partial pressure of arterial oxygen divided by the percentage of inspired oxygen (PaO2/FiO2 ratio) of 300 mm Hg or less despite noninvasive or invasive mechanical ventilation with PEEP or continuous positive airway pressure (CPAP) of at least 5 cm H2O.
In addition, the new definition classifies the severity of disease on the basis of the degree of hypoxemia, ie, the PaO2/FiO2 ratio:
- Mild: PaO2/FiO2 ratio > 200 and ≤ 300 mm Hg
- Moderate: PaO2/FiO2 ratio > 100 and ≤ 200 mm Hg
- Severe: PaO2/FiO2 ratio ≤ 100 mm Hg.
The term “acute lung injury” has been eliminated, as has the previous criterion of a pulmonary artery wedge pressure of 18 mm Hg or less.
The panel also evaluated four ancillary variables for predicting outcomes in severe ARDS:
- Compliance of the respiratory system less than or equal to 40 mL/cm H2O
- Radiographic severity (involvement of three or four quadrants on chest radiography)
- PEEP of 10 cm H2O or greater
- Corrected expired volume 10 L/min or greater.
The task force evaluated the reliability and validity of this definition in a meta-analysis of 4,400 patients previously enrolled in randomized controlled trials or observational studies.
Findings. The Berlin definition predicted the risk of death better than the AECC definition. The mortality rate increased with the severity of ARDS, from 27% with mild disease to 32% with moderate disease to 45% with severe disease. The four ancillary variables did not contribute to the predictive validity of severe ARDS for mortality and were removed from the definition.
Thille et al10 retrospectively reviewed autopsy findings from 712 patients and found that the new definition identified a homogeneous group who had severe ARDS.10
Conclusions. The new definition may overcome some of the limitations of the old one, but it needs to be validated in clinical practice, especially its ability to predict death.
VENTILATORY SUPPORT
Prompt recognition, lung-protective ventilation, and a conservative fluid strategy remain the cornerstones of ARDS management. However, other strategies are being tested.
Prone-position ventilation in severe ARDS: The right therapy in a specific population
Prone-position ventilation was first described almost 30 years ago, but it has been used inconsistently in clinical practice.
Physiologic and observational studies indicated that prone positioning might improve survival in patients with ARDS, but several randomized trials failed to demonstrate any positive effect on outcomes.11,12 Some trials also reported a higher rate of complications with this intervention.13 However, meta-analyses suggested that prone-position ventilation might have a beneficial effect in patients with severe ARDS (defined as a PaO2/FiO2 ratio ≤ 100 mm Hg).14
In view of these findings, investigators conducted a trial of prone-position ventilation exclusively in patients with severe ARDS.
The PROSEVA study
The Proning Severe ARDS Patients (PROSEVA) study was a randomized controlled trial designed to determine whether prone-position ventilation, applied early, would improve outcomes in patients with severe ARDS.15
In PROSEVA, 466 patients with severe ARDS (defined as a PaO2/FiO2 ratio < 150 mm Hg, FiO2 ≥ 60%, and PEEP ≥ 5 cm H2O) underwent either at least 16 hours of prone positioning or were left in the supine position after 12 to 24 hours of initial conventional mechanical ventilation. The patients were recruited from centers in France and Spain where prone-position ventilation had been used in daily practice for more than 5 years.
The primary outcome studied was the rate of death at 28 days. The secondary end points were the death rate at day 90, rates of successful extubation, the length of stay in the intensive care unit, and complications.
Findings. At study entry, the patients in the supine group were sicker, more of them required a vasopressor, and fewer of them were receiving neuromuscular blocking agents than those in the prone group. These baseline differences may have influenced the outcomes; the unadjusted 28-day mortality rate was 16.0% in the prone group compared with 32.8% in the supine group (P < .001). However, the hazard ratio for death with prone positioning was 0.39 (95% confidence interval [CI] 0.25–0.63) even after adjusting for severity and the use of vasopressors and neuromuscular blocking agents. Prone-position ventilation was not associated with a higher incidence of complications, and the rate of successful extubation was higher.
Conclusions. In patients with severe ARDS, early use of prolonged prone positioning significantly decreased the 28-day and 90-day mortality rates. This trial has made prone positioning one of the strategies in managing patients with early severe ARDS. To minimize complications such as pressure ulcers and line or tube dislodgement, personnel caring for these patients must follow a protocol and undergo specific training.
These results were corroborated by a meta-analysis by Beitler et al16 that found a significant decrease in mortality rate with prone-position ventilation even in older studies when lung-protective ventilation strategies were separated from high-tidal-volume ventilation.
High-frequency oscillatory ventilation: No benefit in two trials
Observational data and experimental studies suggested that high-frequency oscillatory ventilation (HFOV) is superior to conventional mechanical ventilation in ARDS patients.17,18 However, outdated and cumbersome equipment, lack of protocols, and a lack of high-quality evidence led to limited and inconsistent use of HFOV, mainly as a rescue therapy in ARDS.19
Over the last few years, HFOV has been gaining acceptance, especially earlier in the course of ARDS.20 After preliminary clinical trials reported promising results, two trials conducted in Canada and the United Kingdom compared HFOV vs conventional mechanical ventilation in patients with ARDS.
The OSCAR study
The Oscillation in ARDS (OSCAR) study21 was a “pragmatic” trial22 (ie, it had minimal exclusion criteria) of the safety and effectiveness of HFOV as a primary ventilatory strategy for ARDS. It included 795 patients randomized to receive conventional ventilation (n = 397) or HFOV (n = 398). Research centers followed detailed algorithms for HFOV management and adopted their usual practice for conventional ventilation. Medical care was given according to the clinician’s judgment.
The primary outcome studied was survival at 30 days. The secondary outcomes were all-cause mortality in the intensive care unit and the hospital, duration of mechanical ventilation, and use of antimicrobial, sedative, vasoactive, and neuromuscular-blocking drugs.
Findings. The patient baseline characteristics were similar in both groups.
There was no significant difference in intensive care unit mortality rates, hospital mortality rates, or mortality rates at 30 days (41.7% in the HFOV group vs 41.1% in the conventional ventilation group; P = .85, 95% CI 6.1–7.5) even after adjustments for center or severity of illness.
The duration of mechanical ventilation was similar in both groups (14.9 ± 13.3 days in the HFOV group vs 14.1 ± 13.4 days in the conventional ventilation group, P = .41). However, sedatives and neuromuscular-blocking drugs were used more often and longer in the HFOV group than in the conventional ventilation group. There was no difference in the use of vasoactive or antimicrobial medications.
Conclusions. This multicenter randomized control trial did not demonstrate any benefit from using HFOV for routine management of ARDS. Its pragmatic design made it less likely to reach a firm conclusion,22 but it at least made a case against routinely using HFOV in patients with ARDS.
The OSCILLATE study
The Oscillation for Acute Respiratory Distress Syndrome Treated Early (OSCILLATE) study23 assessed the safety and efficacy of HFOV as a treatment for early-onset moderate-to-severe ARDS.
The inclusion criteria were similar to those in the OSCAR trial except that pulmonary symptoms had to be present less than 2 weeks and ARDS assessment was done under standard ventilator settings. As this was an efficacy trial, it had more exclusion criteria than the OSCAR trial. A total of 548 patients were randomized to receive conventional ventilation (n = 273) or HFOV (n = 275). The baseline characteristics were similar between groups.
Conventional ventilation was given according to a protocol used in an earlier trial2 and included recruitment maneuvers. HFOV was given in centers that had experience in this treatment, and there were protocols for ventilation management, hemodynamic optimization, and weaning. All other care was left to the clinician’s choice.
The primary outcome studied was in-hospital mortality. The investigators also evaluated whether there were interactions between the treatment and baseline severity of lung injury and center experience with HFOV.
Findings. The trial was stopped after an interim analysis found that HFOV might be harmful, although the statistical threshold for stopping was not reached. The in-hospital mortality rate was 47% in the HFOV group and 35% in the control group (relative risk of death with HFOV 1.33, 95% CI 1.09–1.64, P = .005). HFOV was worse than conventional ventilation regardless of the severity of disease or center experience. The HFOV group had higher mean airway pressures but similar FiO2 compared with the conventional ventilation group.
The HFOV group received significantly more vasopressors, sedatives, and neuromuscular blockers. This group’s fluid balance was higher as well, but not significantly so. Refractory hypoxemia (defined as PaO2 < 60 mm Hg for 1 hour with an FiO2 of 1.0 and neuromuscular blockade) was more frequent in the conventional ventilation group, but the number of deaths in the subgroup with refractory hypoxemia was similar with either treatment.
Conclusions. This multicenter randomized controlled trial demonstrated that HFOV was harmful when used routinely to manage ARDS. The trial’s protocol was based on the results of a pilot study carried out by the same investigators, which provided the best evidence available regarding the safety of HFOV at that time.
The results of the OSCAR and OSCILLATE trials have quelled enthusiasm for early, routine use of HFOV in ARDS. Although there are concerns that the protocol (ie, the way HFOV was implemented) rather than HFOV itself may have led to worse outcomes, there is no signal to support its routine use. We need further studies to define if it remains a viable rescue therapy.
Extracorporeal membrane oxygenation: Is it a viable option in severe ARDS?
Extracorporeal membrane oxygenation (ECMO) uses cardiopulmonary bypass technology to provide gas exchange. In patients with severe hypoxemia, ECMO can ensure adequate oxygenation and ventilation while ensuring the optimization of lung-protective ventilation. But ECMO was never as successful in adults with ARDS as it was in children and neonates.24
The first two trials of ECMO in ARDS24,25 reported equal or worse survival rates compared with conventional ventilation, and the overall mortality rate in these studies was staggeringly high. However, these studies were carried out before the era of lung-protective ventilation and at a time when ECMO technology was relatively primitive.
With new technology such as venovenous circuits and smaller cannulas, ECMO has gained more acceptance. It was used in patients with severe or refractory hypoxemia associated with ARDS during the H1N1 pandemic.26,27
The CESAR trial
The Conventional Ventilatory Support Versus Extracorporeal Membrane Oxygenation for Severe Adult Respiratory Failure (CESAR) trial28 assessed the safety, clinical efficacy, and cost-effectiveness of ECMO in managing severe ARDS. It compared best standard practice vs a protocol that included ECMO. The trial was conducted from 2001 to 2006.
Patients with severe ARDS, as defined by a Murray score29 greater than 3 or uncompensated hypercapnea, were prospectively randomized and recruited from an ECMO center and 148 tertiary intensive care units and referral hospitals in England. This was a pragmatic trial, with minimal exclusion criteria (essentially, mechanical ventilation with high pressures and high FiO2 for more than 7 days, intracranial bleeding, or contraindication to heparinization).
A total of 180 patients were randomized in a one-to-one ratio to receive ECMO or conventional management. The ventilator management in the conventional treatment group was not done according to a protocol but in general was low-volume and low-pressure. All patients randomized to ECMO were transferred to the ECMO center and treated according to a standardized ventilation protocol. After 12 hours, if predefined goals were not reached, venovenous ECMO was started. Patients assigned to conventional management could not cross over to ECMO.
The primary outcomes were death or severe disability at 6 months after randomization, and cost-effectiveness. The secondary outcomes were hospital resource use (eg, rescue techniques, length of stay, duration of ECMO) and health status after 6 months.
Findings. The groups were similar at baseline. Sixty-eight (75%) of the 90 patients randomized to receive ECMO actually received it. Of the 22 patients who did not receive ECMO, 16 (18% of the 90) improved on conventional therapy, 5 (6%) died during or before transfer, and 1 could not receive heparin.
Two patients had severe complications in the ECMO group: one had an arterial puncture, and one had an oxygen delivery failure during transport. In each case, these events contributed to the death of the patient.
More patients in the ECMO group received lung-protective ventilation, 84 (93%) vs 63 (70%).
The primary outcome, ie, death or severe disability at 6 months, occurred in 33 (37%) of the 90 patients in the ECMO group and in 46 (53%) of the patients in the conventional management group (relative risk 0.69, 95% CI 0.05–0.97, P = .03). More patients in the ECMO group survived, but the difference was not statistically significant (relative risk of death 0.73, 95% CI 0.52–1.03, P = .07). The most common cause of death in the ECMO group was multiorgan failure (42%), whereas in the conventional management group, the most common cause of death was respiratory failure (60%).
Length of stay in the hospital and in the critical care unit and health care costs were double for patients in the ECMO group. There was no difference in quality-of-life markers at 6 months in the survivors.
Conclusions. This pragmatic trial demonstrated that a protocol that includes ECMO could improve survival rates in ARDS.
Of note, the ECMO group got care in regional centers that used protocols. Therefore, in interpreting the results of this trial, we have to consider that being in a center with protocol-specified care for ARDS could drive some of the difference in mortality rates.
Regardless, this trial demonstrated that ECMO is feasible and led to better outcomes than expected. The findings were encouraging, and spurred the use of ECMO in severe ARDS during the 2009 H1N1 pandemic. Two propensity-matched studies and a number of case series reported a survival benefit associated with the use of ECMO in patients with severe ARDS.27,30
A recent meta-analysis also reported that ECMO might lower the mortality rate in ARDS; however, the patients in the H1N1 pandemic were younger and usually had isolated respiratory failure.31
The success of ECMO has opened new possibilities in the management of ARDS. As the technology improves and our experience increases, ECMO will likely gain more acceptance as a treatment for severe ARDS.
Airway pressure release ventilation
The use of airway pressure release ventilation and other ventilator modalities in ARDS is not supported by current evidence, though results of clinical trials may influence our practice in the future.
PHARMACOTHERAPY IN ARDS
The pathogenesis of ARDS includes damage to the alveolar-capillary membrane, with leakage of protein-rich edema fluid into alveoli. This damage is propagated by a complex inflammatory response including but not limited to neutrophil activation, free-radical formation, dysregulation of the coagulation system, and extensive release of inflammatory mediators.32,33 As a consequence, there are multiple potential targets for pharmacologic therapy in ARDS.
A variety of drugs, including corticosteroids, anti-inflammatory agents, immune-modulating agents, pulmonary vasodilators, antioxidants, and surfactants, have been studied in patients with ARDS.34 But effective pharmacotherapy for ARDS remains extremely limited.
Neuromuscular blockade in early severe ARDS
Mechanical ventilation can result in injurious stretching of the lung parenchyma, either from alveolar overdistention (volutrauma) or from continual recruitment and derecruitment of unstable lung units during the ventilator cycle (atelectrauma).35 Ventilator-induced lung injury can be exacerbated by asynchronous breathing.
In theory, neuromuscular blockers could minimize patient-ventilator asynchrony and provide much better control of tidal volume and pressure in patients with ARDS. This may result in less volutrauma and atelectrauma associated with asynchronous breathing. Data also suggest that cisatracurium (Nimbex), a neuromuscular blocking agent, may have a direct effect on the amount of inflammation in lungs with ARDS.36
The ACURASYS study
The ARDS et Curarisation Systématique (ACURASYS) study37 was a randomized trial in 340 patients undergoing mechanical ventilation for severe ARDS to evaluate the impact of neuromuscular blockade within the first 48 hours in this population.
The primary outcome was the mortality rate before hospital discharge or within 90 days of study entry. Secondary outcomes included the 28-day mortality rate, the rate of intensive care unit-acquired paresis, and the number of ventilator-free days. To be included, patients had to have been mechanically ventilated for less than 48 hours and to meet the AECC criteria for severe ARDS, with a PaO2/FiO2 ratio less than 150 mm Hg.
The intervention group received a continuous infusion of cisatracurium for 48 hours, while the control patients received placebo. Muscle strength was evaluated by clinical scoring of strength in different muscle groups.
Findings. The study groups were similar at baseline.
The crude 90-day mortality rate was lower in the cisatracurium group (31.6% vs 40.7%, P = .08). Regression analysis showed an improved 90-day survival rate with the use of this neuromuscular blocker after adjustment for severity of illness and the severity of ARDS (based on degree of hypoxemia and plateau pressures) (hazard ratio for death at 90 days 0.68; 95% CI 0.48–0.98; P = .04). The rate of paresis acquired in the intensive care unit did not differ significantly between the two groups.
Conclusion. In patients with severe ARDS, giving a neuromuscular blocking agent early improved the survival rate and increased the time off the ventilator without increasing muscle weakness.
These data are in line with similar findings from two other studies published by the same group.38,39 A meta-analysis of 432 patients showed that the use of neuromuscular blockade in early severe ARDS is associated with a statistically significant effect on early mortality (relative risk 0.66, 95% CI 0.50–0.87).40 The pooled analysis of these trials did not show any statistically significant critical-illness polyneuropathy.
These results need to be interpreted carefully, as we have inadequate data to see if they generalize to different intensive care units, and the evaluation and categorization of critical-illness polyneuropathy remains to be defined.
Cisatracurium is a promising treatment for moderate to severe ARDS and merits investigation in a large confirmatory randomized controlled trial.
Other pharmacologic agents
A number of other drugs have been studied in ARDS patients, including both inhaled and intravenous beta agonists,41,42 statins,43 and nutritional supplements.44 But as with other drugs previously studied in ARDS such as corticosteroids, N-acetylcysteine, and surfactant,34 these agents showed no effect on outcomes. In fact, a recent trial of intravenous salbutamol in ARDS patients was stopped after an interim analysis because of a higher incidence of arrhythmias and lactic acidosis with this agent.42
These findings reaffirm that pharmacologic therapy needs to be carefully considered, and potential harms associated with these therapies need to be addressed before they are introduced in the care of critically ill patients.
- Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. Acute Respiratory Distress Syndrome Network. N Engl J Med 2000; 342:1301–1308.
- Meade MO, Cook DJ, Guyatt GH, et al; Lung Open Ventilation Study Investigators. Ventilation strategy using low tidal volumes, recruitment maneuvers, and high positive end-expiratory pressure for acute lung injury and acute respiratory distress syndrome: a randomized controlled trial. JAMA 2008; 299:637–645.
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network; Wiedemann HP, Wheeler AP, Bernard GR, et al. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med 2006; 354:2564–2575.
- Girard TD, Kress JP, Fuchs BD, et al. Efficacy and safety of a paired sedation and ventilator weaning protocol for mechanically ventilated patients in intensive care (Awakening and Breathing Controlled trial): a randomised controlled trial. Lancet 2008; 371:126–134.
- Schweickert WD, Pohlman MC, Pohlman AS, et al. Early physical and occupational therapy in mechanically ventilated, critically ill patients: a randomised controlled trial. Lancet 2009; 373:1874–1882.
- Ely EW, Baker AM, Dunagan DP, et al. Effect on the duration of mechanical ventilation of identifying patients capable of breathing spontaneously. N Engl J Med 1996; 335:1864–1869.
- Bernard GR, Artigas A, Brigham KL, et al. The American-European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med 1994; 149:818–824.
- Ferguson ND, Fan E, Camporota L, et al. The Berlin definition of ARDS: an expanded rationale, justification, and supplementary material. Intensive Care Med 2012; 38:1573–1582.
- ARDS Definition Task Force; Ranieri VM, Rubenfeld GD, Thompson BT, et al. Acute respiratory distress syndrome: the Berlin definition. JAMA 2012; 307:2526–2533.
- Thille AW, Esteban A, Fernández-Segoviano P, et al. Comparison of the Berlin definition for acute respiratory distress syndrome with autopsy. Am J Respir Crit Care Med 2013; 187:761–767.
- Gattinoni L, Tognoni G, Pesenti A, et al; Prone-Supine Study Group. Effect of prone positioning on the survival of patients with acute respiratory failure. N Engl J Med 2001; 345:568–573.
- Taccone P, Pesenti A, Latini R, et al; Prone-Supine II Study Group. Prone positioning in patients with moderate and severe acute respiratory distress syndrome: a randomized controlled trial. JAMA 2009; 302:1977–1984.
- Mancebo J, Fernández R, Blanch L, et al. A multicenter trial of prolonged prone ventilation in severe acute respiratory distress syndrome. Am J Respir Crit Care Med 2006; 173:1233–1239.
- Sud S, Friedrich JO, Taccone P, et al. Prone ventilation reduces mortality in patients with acute respiratory failure and severe hypoxemia: systematic review and meta-analysis. Intensive Care Med 2010; 36:585–599.
- Guérin C, Reignier J, Richard JC, et al; PROSEVA Study Group. Prone positioning in severe acute respiratory distress syndrome. N Engl J Med 2013; 368:2159–2168.
- Beitler JR, Shaefi S, Montesi SB, et al. Prone positioning reduces mortality from acute respiratory distress syndrome in the low tidal volume era: a meta-analysis. Intensive Care Med 2014; 40:332–341.
- Chan KP, Stewart TE, Mehta S. High-frequency oscillatory ventilation for adult patients with ARDS. Chest 2007; 131:1907–1916.
- Fessler HE, Hager DN, Brower RG. Feasibility of very high-frequency ventilation in adults with acute respiratory distress syndrome. Crit Care Med 2008; 36:1043–1048.
- Mehta S, Granton J, MacDonald RJ, et al. High-frequency oscillatory ventilation in adults: the Toronto experience. Chest 2004; 126:518–527.
- Ferguson ND, Chiche JD, Kacmarek RM, et al. Combining high-frequency oscillatory ventilation and recruitment maneuvers in adults with early acute respiratory distress syndrome: the Treatment with Oscillation and an Open Lung Strategy (TOOLS) Trial pilot study. Crit Care Med 2005; 33:479–486.
- Young D, Lamb SE, Shah S, et al; OSCAR Study Group. High-frequency oscillation for acute respiratory distress syndrome. N Engl J Med 2013; 368:806–813.
- Thorpe KE, Zwarenstein M, Oxman AD, et al. A pragmatic-explanatory continuum indicator summary (PRECIS): a tool to help trial designers. J Clin Epidemiol 2009; 62:464–475.
- Ferguson ND, Cook DJ, Guyatt GH, et al; OSCILLATE Trial Investigators; Canadian Critical Care Trials Group. High-frequency oscillation in early acute respiratory distress syndrome. N Engl J Med 2013; 368:795–805.
- Morris AH, Wallace CJ, Menlove RL, et al. Randomized clinical trial of pressure-controlled inverse ratio ventilation and extracorporeal CO2 removal for adult respiratory distress syndrome. Am J Respir Crit Care Med 1994; 149:295–305.
- Zapol WM, Snider MT, Hill JD, et al. Extracorporeal membrane oxygenation in severe acute respiratory failure. A randomized prospective study. JAMA 1979; 242:2193–2196.
- Australia and New Zealand Extracorporeal Membrane Oxygenation (ANZ ECMO) Influenza Investigators; Davies A, Jones D, Bailey M, et al. Extracorporeal Membrane Oxygenation for 2009 Influenza A(H1N1) Acute Respiratory Distress Syndrome. JAMA 2009; 302:1888–1895.
- Pham T, Combes A, Rozé H, et al; REVA Research Network. Extracorporeal membrane oxygenation for pandemic influenza A(H1N1)-induced acute respiratory distress syndrome: a cohort study and propensity-matched analysis. Am J Respir Crit Care Med 2013; 187:276–285.
- Peek GJ, Mugford M, Tiruvoipati R, et al; CESAR trial collaboration. Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): a multicentre randomised controlled trial. Lancet 2009; 374:1351–1363.
- Murray JF, Matthay MA, Luce JM, Flick MR. An expanded definition of the adult respiratory distress syndrome. Am Rev Respir Dis 1988; 138:720–723.
- Noah MA, Peek GJ, Finney SJ, et al. Referral to an extracorporeal membrane oxygenation center and mortality among patients with severe 2009 influenza A(H1N1). JAMA 2011; 306:1659–1668.
- Zampieri FG, Mendes PV, Ranzani OT, et al. Extracorporeal membrane oxygenation for severe respiratory failure in adult patients: a systematic review and meta-analysis of current evidence. J Crit Care 2013; 28:998–1005.
- Raghavendran K, Pryhuber GS, Chess PR, Davidson BA, Knight PR, Notter RH. Pharmacotherapy of acute lung injury and acute respiratory distress syndrome. Curr Med Chem 2008; 15:1911–1924.
- Adhikari N, Burns KE, Meade MO. Pharmacologic treatments for acute respiratory distress syndrome and acute lung injury: systematic review and meta-analysis. Treat Respir Med 2004; 3:307–328.
- Adhikari N, Burns KE, Meade MO. Pharmacologic therapies for adults with acute lung injury and acute respiratory distress syndrome. Cochrane Database Syst Rev 2004; 4:CD004477.
- Terragni PP, Rosboch GL, Lisi A, Viale AG, Ranieri VM. How respiratory system mechanics may help in minimising ventilator-induced lung injury in ARDS patients. Eur Respir J Suppl 2003; 42:15s–21s.
- Forel JM, Roch A, Papazian L. Paralytics in critical care: not always the bad guy. Curr Opin Crit Care 2009; 15:59–66.
- Papazian L, Forel JM, Gacouin A, et al; ACURASYS Study Investigators. Neuromuscular blockers in early acute respiratory distress syndrome. N Engl J Med 2010; 363:1107–1116.
- Gainnier M, Roch A, Forel JM, et al. Effect of neuromuscular blocking agents on gas exchange in patients presenting with acute respiratory distress syndrome. Crit Care Med 2004; 32:113–19.
- Forel JM, Roch A, Marin V, et al. Neuromuscular blocking agents decrease inflammatory response in patients presenting with acute respiratory distress syndrome. Crit Care Med 2006; 34:2749–2757.
- Alhazzani W, Alshahrani M, Jaeschke R, et al. Neuromuscular blocking agents in acute respiratory distress syndrome: a systematic review and meta-analysis of randomized controlled trials. Crit Care 2013; 17:R43.
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network; Matthay MA, Brower RG, Carson S, et al. Randomized, placebo-controlled clinical trial of an aerosolized beta-2-agonist for treatment of acute lung injury. Am J Respir Crit Care Med 2011; 184:561–568.
- Gao Smith F, Perkins GD, Gates S, et al; BALTI-2 study investigators. Effect of intravenous beta-2 agonist treatment on clinical outcomes in acute respiratory distress syndrome (BALTI-2): a multicentre, randomised controlled trial. Lancet 2012; 379:229–235.
- Craig TR, Duffy MJ, Shyamsundar M, et al. A randomized clinical trial of hydroxymethylglutaryl-coenzyme a reductase inhibition for acute lung injury (The HARP Study). Am J Respir Crit Care Med 2011; 183:620–626.
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network; Rice TW, Wheeler AP, Thompson BT, et al. Initial trophic vs full enteral feeding in patients with acute lung injury: the EDEN randomized trial. JAMA 2012; 307:795–803.
Continued progress in understanding the pathophysiology of acute respiratory distress syndrome (ARDS) is translating into changes in the way we diagnose and manage it. Over the past 20 years, low tidal volume,1 positive end-expiratory pressure (PEEP),2 and fluid restriction3 have become the standard of care. A multidisciplinary approach, including targeted use of sedatives, early mobilization, and protocols for weaning from the ventilator, has also brought about substantial changes in ARDS management and its outcomes.4–6
In this article, we review the most relevant articles about ARDS in the last 5 years. We include the new definition of ARDS and studies of ventilatory and nonventilatory therapies that have implications in managing patients with ARDS.
A STANDARDIZED APPROACH
ARDS is characterized by damage to the alveolar architecture, severe hypoxemia, and bilateral parenchymal opacities.
The working definition of ARDS developed in 1994 by the American-European Consensus Conference (AECC) was the basis for enrollment in most of the landmark trials and observational studies over the past 20 years.7,8 However, it was limited in its reliability and validity.
An updated definition
In 2011, the ARDS Definition Task Force, using a novel consensus process, updated the ARDS definition,9 focusing on its feasibility, reliability, and validity in predicting response to therapies and outcomes in ARDS. This new “Berlin” definition is not substantially different from the old, but defines the criteria more specifically:
- Bilateral opacities, unexplained by nodules, atelectasis, or effusion, on chest radiography or computed tomography
- New or worsening respiratory symptoms, or a clinical insult associated with ARDS within 7 days of diagnosis
- Objective assessment of cardiac function (eg, with echocardiography) to exclude cardiogenic pulmonary edema
- Hypoxemia, with a partial pressure of arterial oxygen divided by the percentage of inspired oxygen (PaO2/FiO2 ratio) of 300 mm Hg or less despite noninvasive or invasive mechanical ventilation with PEEP or continuous positive airway pressure (CPAP) of at least 5 cm H2O.
In addition, the new definition classifies the severity of disease on the basis of the degree of hypoxemia, ie, the PaO2/FiO2 ratio:
- Mild: PaO2/FiO2 ratio > 200 and ≤ 300 mm Hg
- Moderate: PaO2/FiO2 ratio > 100 and ≤ 200 mm Hg
- Severe: PaO2/FiO2 ratio ≤ 100 mm Hg.
The term “acute lung injury” has been eliminated, as has the previous criterion of a pulmonary artery wedge pressure of 18 mm Hg or less.
The panel also evaluated four ancillary variables for predicting outcomes in severe ARDS:
- Compliance of the respiratory system less than or equal to 40 mL/cm H2O
- Radiographic severity (involvement of three or four quadrants on chest radiography)
- PEEP of 10 cm H2O or greater
- Corrected expired volume 10 L/min or greater.
The task force evaluated the reliability and validity of this definition in a meta-analysis of 4,400 patients previously enrolled in randomized controlled trials or observational studies.
Findings. The Berlin definition predicted the risk of death better than the AECC definition. The mortality rate increased with the severity of ARDS, from 27% with mild disease to 32% with moderate disease to 45% with severe disease. The four ancillary variables did not contribute to the predictive validity of severe ARDS for mortality and were removed from the definition.
Thille et al10 retrospectively reviewed autopsy findings from 712 patients and found that the new definition identified a homogeneous group who had severe ARDS.10
Conclusions. The new definition may overcome some of the limitations of the old one, but it needs to be validated in clinical practice, especially its ability to predict death.
VENTILATORY SUPPORT
Prompt recognition, lung-protective ventilation, and a conservative fluid strategy remain the cornerstones of ARDS management. However, other strategies are being tested.
Prone-position ventilation in severe ARDS: The right therapy in a specific population
Prone-position ventilation was first described almost 30 years ago, but it has been used inconsistently in clinical practice.
Physiologic and observational studies indicated that prone positioning might improve survival in patients with ARDS, but several randomized trials failed to demonstrate any positive effect on outcomes.11,12 Some trials also reported a higher rate of complications with this intervention.13 However, meta-analyses suggested that prone-position ventilation might have a beneficial effect in patients with severe ARDS (defined as a PaO2/FiO2 ratio ≤ 100 mm Hg).14
In view of these findings, investigators conducted a trial of prone-position ventilation exclusively in patients with severe ARDS.
The PROSEVA study
The Proning Severe ARDS Patients (PROSEVA) study was a randomized controlled trial designed to determine whether prone-position ventilation, applied early, would improve outcomes in patients with severe ARDS.15
In PROSEVA, 466 patients with severe ARDS (defined as a PaO2/FiO2 ratio < 150 mm Hg, FiO2 ≥ 60%, and PEEP ≥ 5 cm H2O) underwent either at least 16 hours of prone positioning or were left in the supine position after 12 to 24 hours of initial conventional mechanical ventilation. The patients were recruited from centers in France and Spain where prone-position ventilation had been used in daily practice for more than 5 years.
The primary outcome studied was the rate of death at 28 days. The secondary end points were the death rate at day 90, rates of successful extubation, the length of stay in the intensive care unit, and complications.
Findings. At study entry, the patients in the supine group were sicker, more of them required a vasopressor, and fewer of them were receiving neuromuscular blocking agents than those in the prone group. These baseline differences may have influenced the outcomes; the unadjusted 28-day mortality rate was 16.0% in the prone group compared with 32.8% in the supine group (P < .001). However, the hazard ratio for death with prone positioning was 0.39 (95% confidence interval [CI] 0.25–0.63) even after adjusting for severity and the use of vasopressors and neuromuscular blocking agents. Prone-position ventilation was not associated with a higher incidence of complications, and the rate of successful extubation was higher.
Conclusions. In patients with severe ARDS, early use of prolonged prone positioning significantly decreased the 28-day and 90-day mortality rates. This trial has made prone positioning one of the strategies in managing patients with early severe ARDS. To minimize complications such as pressure ulcers and line or tube dislodgement, personnel caring for these patients must follow a protocol and undergo specific training.
These results were corroborated by a meta-analysis by Beitler et al16 that found a significant decrease in mortality rate with prone-position ventilation even in older studies when lung-protective ventilation strategies were separated from high-tidal-volume ventilation.
High-frequency oscillatory ventilation: No benefit in two trials
Observational data and experimental studies suggested that high-frequency oscillatory ventilation (HFOV) is superior to conventional mechanical ventilation in ARDS patients.17,18 However, outdated and cumbersome equipment, lack of protocols, and a lack of high-quality evidence led to limited and inconsistent use of HFOV, mainly as a rescue therapy in ARDS.19
Over the last few years, HFOV has been gaining acceptance, especially earlier in the course of ARDS.20 After preliminary clinical trials reported promising results, two trials conducted in Canada and the United Kingdom compared HFOV vs conventional mechanical ventilation in patients with ARDS.
The OSCAR study
The Oscillation in ARDS (OSCAR) study21 was a “pragmatic” trial22 (ie, it had minimal exclusion criteria) of the safety and effectiveness of HFOV as a primary ventilatory strategy for ARDS. It included 795 patients randomized to receive conventional ventilation (n = 397) or HFOV (n = 398). Research centers followed detailed algorithms for HFOV management and adopted their usual practice for conventional ventilation. Medical care was given according to the clinician’s judgment.
The primary outcome studied was survival at 30 days. The secondary outcomes were all-cause mortality in the intensive care unit and the hospital, duration of mechanical ventilation, and use of antimicrobial, sedative, vasoactive, and neuromuscular-blocking drugs.
Findings. The patient baseline characteristics were similar in both groups.
There was no significant difference in intensive care unit mortality rates, hospital mortality rates, or mortality rates at 30 days (41.7% in the HFOV group vs 41.1% in the conventional ventilation group; P = .85, 95% CI 6.1–7.5) even after adjustments for center or severity of illness.
The duration of mechanical ventilation was similar in both groups (14.9 ± 13.3 days in the HFOV group vs 14.1 ± 13.4 days in the conventional ventilation group, P = .41). However, sedatives and neuromuscular-blocking drugs were used more often and longer in the HFOV group than in the conventional ventilation group. There was no difference in the use of vasoactive or antimicrobial medications.
Conclusions. This multicenter randomized control trial did not demonstrate any benefit from using HFOV for routine management of ARDS. Its pragmatic design made it less likely to reach a firm conclusion,22 but it at least made a case against routinely using HFOV in patients with ARDS.
The OSCILLATE study
The Oscillation for Acute Respiratory Distress Syndrome Treated Early (OSCILLATE) study23 assessed the safety and efficacy of HFOV as a treatment for early-onset moderate-to-severe ARDS.
The inclusion criteria were similar to those in the OSCAR trial except that pulmonary symptoms had to be present less than 2 weeks and ARDS assessment was done under standard ventilator settings. As this was an efficacy trial, it had more exclusion criteria than the OSCAR trial. A total of 548 patients were randomized to receive conventional ventilation (n = 273) or HFOV (n = 275). The baseline characteristics were similar between groups.
Conventional ventilation was given according to a protocol used in an earlier trial2 and included recruitment maneuvers. HFOV was given in centers that had experience in this treatment, and there were protocols for ventilation management, hemodynamic optimization, and weaning. All other care was left to the clinician’s choice.
The primary outcome studied was in-hospital mortality. The investigators also evaluated whether there were interactions between the treatment and baseline severity of lung injury and center experience with HFOV.
Findings. The trial was stopped after an interim analysis found that HFOV might be harmful, although the statistical threshold for stopping was not reached. The in-hospital mortality rate was 47% in the HFOV group and 35% in the control group (relative risk of death with HFOV 1.33, 95% CI 1.09–1.64, P = .005). HFOV was worse than conventional ventilation regardless of the severity of disease or center experience. The HFOV group had higher mean airway pressures but similar FiO2 compared with the conventional ventilation group.
The HFOV group received significantly more vasopressors, sedatives, and neuromuscular blockers. This group’s fluid balance was higher as well, but not significantly so. Refractory hypoxemia (defined as PaO2 < 60 mm Hg for 1 hour with an FiO2 of 1.0 and neuromuscular blockade) was more frequent in the conventional ventilation group, but the number of deaths in the subgroup with refractory hypoxemia was similar with either treatment.
Conclusions. This multicenter randomized controlled trial demonstrated that HFOV was harmful when used routinely to manage ARDS. The trial’s protocol was based on the results of a pilot study carried out by the same investigators, which provided the best evidence available regarding the safety of HFOV at that time.
The results of the OSCAR and OSCILLATE trials have quelled enthusiasm for early, routine use of HFOV in ARDS. Although there are concerns that the protocol (ie, the way HFOV was implemented) rather than HFOV itself may have led to worse outcomes, there is no signal to support its routine use. We need further studies to define if it remains a viable rescue therapy.
Extracorporeal membrane oxygenation: Is it a viable option in severe ARDS?
Extracorporeal membrane oxygenation (ECMO) uses cardiopulmonary bypass technology to provide gas exchange. In patients with severe hypoxemia, ECMO can ensure adequate oxygenation and ventilation while ensuring the optimization of lung-protective ventilation. But ECMO was never as successful in adults with ARDS as it was in children and neonates.24
The first two trials of ECMO in ARDS24,25 reported equal or worse survival rates compared with conventional ventilation, and the overall mortality rate in these studies was staggeringly high. However, these studies were carried out before the era of lung-protective ventilation and at a time when ECMO technology was relatively primitive.
With new technology such as venovenous circuits and smaller cannulas, ECMO has gained more acceptance. It was used in patients with severe or refractory hypoxemia associated with ARDS during the H1N1 pandemic.26,27
The CESAR trial
The Conventional Ventilatory Support Versus Extracorporeal Membrane Oxygenation for Severe Adult Respiratory Failure (CESAR) trial28 assessed the safety, clinical efficacy, and cost-effectiveness of ECMO in managing severe ARDS. It compared best standard practice vs a protocol that included ECMO. The trial was conducted from 2001 to 2006.
Patients with severe ARDS, as defined by a Murray score29 greater than 3 or uncompensated hypercapnea, were prospectively randomized and recruited from an ECMO center and 148 tertiary intensive care units and referral hospitals in England. This was a pragmatic trial, with minimal exclusion criteria (essentially, mechanical ventilation with high pressures and high FiO2 for more than 7 days, intracranial bleeding, or contraindication to heparinization).
A total of 180 patients were randomized in a one-to-one ratio to receive ECMO or conventional management. The ventilator management in the conventional treatment group was not done according to a protocol but in general was low-volume and low-pressure. All patients randomized to ECMO were transferred to the ECMO center and treated according to a standardized ventilation protocol. After 12 hours, if predefined goals were not reached, venovenous ECMO was started. Patients assigned to conventional management could not cross over to ECMO.
The primary outcomes were death or severe disability at 6 months after randomization, and cost-effectiveness. The secondary outcomes were hospital resource use (eg, rescue techniques, length of stay, duration of ECMO) and health status after 6 months.
Findings. The groups were similar at baseline. Sixty-eight (75%) of the 90 patients randomized to receive ECMO actually received it. Of the 22 patients who did not receive ECMO, 16 (18% of the 90) improved on conventional therapy, 5 (6%) died during or before transfer, and 1 could not receive heparin.
Two patients had severe complications in the ECMO group: one had an arterial puncture, and one had an oxygen delivery failure during transport. In each case, these events contributed to the death of the patient.
More patients in the ECMO group received lung-protective ventilation, 84 (93%) vs 63 (70%).
The primary outcome, ie, death or severe disability at 6 months, occurred in 33 (37%) of the 90 patients in the ECMO group and in 46 (53%) of the patients in the conventional management group (relative risk 0.69, 95% CI 0.05–0.97, P = .03). More patients in the ECMO group survived, but the difference was not statistically significant (relative risk of death 0.73, 95% CI 0.52–1.03, P = .07). The most common cause of death in the ECMO group was multiorgan failure (42%), whereas in the conventional management group, the most common cause of death was respiratory failure (60%).
Length of stay in the hospital and in the critical care unit and health care costs were double for patients in the ECMO group. There was no difference in quality-of-life markers at 6 months in the survivors.
Conclusions. This pragmatic trial demonstrated that a protocol that includes ECMO could improve survival rates in ARDS.
Of note, the ECMO group got care in regional centers that used protocols. Therefore, in interpreting the results of this trial, we have to consider that being in a center with protocol-specified care for ARDS could drive some of the difference in mortality rates.
Regardless, this trial demonstrated that ECMO is feasible and led to better outcomes than expected. The findings were encouraging, and spurred the use of ECMO in severe ARDS during the 2009 H1N1 pandemic. Two propensity-matched studies and a number of case series reported a survival benefit associated with the use of ECMO in patients with severe ARDS.27,30
A recent meta-analysis also reported that ECMO might lower the mortality rate in ARDS; however, the patients in the H1N1 pandemic were younger and usually had isolated respiratory failure.31
The success of ECMO has opened new possibilities in the management of ARDS. As the technology improves and our experience increases, ECMO will likely gain more acceptance as a treatment for severe ARDS.
Airway pressure release ventilation
The use of airway pressure release ventilation and other ventilator modalities in ARDS is not supported by current evidence, though results of clinical trials may influence our practice in the future.
PHARMACOTHERAPY IN ARDS
The pathogenesis of ARDS includes damage to the alveolar-capillary membrane, with leakage of protein-rich edema fluid into alveoli. This damage is propagated by a complex inflammatory response including but not limited to neutrophil activation, free-radical formation, dysregulation of the coagulation system, and extensive release of inflammatory mediators.32,33 As a consequence, there are multiple potential targets for pharmacologic therapy in ARDS.
A variety of drugs, including corticosteroids, anti-inflammatory agents, immune-modulating agents, pulmonary vasodilators, antioxidants, and surfactants, have been studied in patients with ARDS.34 But effective pharmacotherapy for ARDS remains extremely limited.
Neuromuscular blockade in early severe ARDS
Mechanical ventilation can result in injurious stretching of the lung parenchyma, either from alveolar overdistention (volutrauma) or from continual recruitment and derecruitment of unstable lung units during the ventilator cycle (atelectrauma).35 Ventilator-induced lung injury can be exacerbated by asynchronous breathing.
In theory, neuromuscular blockers could minimize patient-ventilator asynchrony and provide much better control of tidal volume and pressure in patients with ARDS. This may result in less volutrauma and atelectrauma associated with asynchronous breathing. Data also suggest that cisatracurium (Nimbex), a neuromuscular blocking agent, may have a direct effect on the amount of inflammation in lungs with ARDS.36
The ACURASYS study
The ARDS et Curarisation Systématique (ACURASYS) study37 was a randomized trial in 340 patients undergoing mechanical ventilation for severe ARDS to evaluate the impact of neuromuscular blockade within the first 48 hours in this population.
The primary outcome was the mortality rate before hospital discharge or within 90 days of study entry. Secondary outcomes included the 28-day mortality rate, the rate of intensive care unit-acquired paresis, and the number of ventilator-free days. To be included, patients had to have been mechanically ventilated for less than 48 hours and to meet the AECC criteria for severe ARDS, with a PaO2/FiO2 ratio less than 150 mm Hg.
The intervention group received a continuous infusion of cisatracurium for 48 hours, while the control patients received placebo. Muscle strength was evaluated by clinical scoring of strength in different muscle groups.
Findings. The study groups were similar at baseline.
The crude 90-day mortality rate was lower in the cisatracurium group (31.6% vs 40.7%, P = .08). Regression analysis showed an improved 90-day survival rate with the use of this neuromuscular blocker after adjustment for severity of illness and the severity of ARDS (based on degree of hypoxemia and plateau pressures) (hazard ratio for death at 90 days 0.68; 95% CI 0.48–0.98; P = .04). The rate of paresis acquired in the intensive care unit did not differ significantly between the two groups.
Conclusion. In patients with severe ARDS, giving a neuromuscular blocking agent early improved the survival rate and increased the time off the ventilator without increasing muscle weakness.
These data are in line with similar findings from two other studies published by the same group.38,39 A meta-analysis of 432 patients showed that the use of neuromuscular blockade in early severe ARDS is associated with a statistically significant effect on early mortality (relative risk 0.66, 95% CI 0.50–0.87).40 The pooled analysis of these trials did not show any statistically significant critical-illness polyneuropathy.
These results need to be interpreted carefully, as we have inadequate data to see if they generalize to different intensive care units, and the evaluation and categorization of critical-illness polyneuropathy remains to be defined.
Cisatracurium is a promising treatment for moderate to severe ARDS and merits investigation in a large confirmatory randomized controlled trial.
Other pharmacologic agents
A number of other drugs have been studied in ARDS patients, including both inhaled and intravenous beta agonists,41,42 statins,43 and nutritional supplements.44 But as with other drugs previously studied in ARDS such as corticosteroids, N-acetylcysteine, and surfactant,34 these agents showed no effect on outcomes. In fact, a recent trial of intravenous salbutamol in ARDS patients was stopped after an interim analysis because of a higher incidence of arrhythmias and lactic acidosis with this agent.42
These findings reaffirm that pharmacologic therapy needs to be carefully considered, and potential harms associated with these therapies need to be addressed before they are introduced in the care of critically ill patients.
Continued progress in understanding the pathophysiology of acute respiratory distress syndrome (ARDS) is translating into changes in the way we diagnose and manage it. Over the past 20 years, low tidal volume,1 positive end-expiratory pressure (PEEP),2 and fluid restriction3 have become the standard of care. A multidisciplinary approach, including targeted use of sedatives, early mobilization, and protocols for weaning from the ventilator, has also brought about substantial changes in ARDS management and its outcomes.4–6
In this article, we review the most relevant articles about ARDS in the last 5 years. We include the new definition of ARDS and studies of ventilatory and nonventilatory therapies that have implications in managing patients with ARDS.
A STANDARDIZED APPROACH
ARDS is characterized by damage to the alveolar architecture, severe hypoxemia, and bilateral parenchymal opacities.
The working definition of ARDS developed in 1994 by the American-European Consensus Conference (AECC) was the basis for enrollment in most of the landmark trials and observational studies over the past 20 years.7,8 However, it was limited in its reliability and validity.
An updated definition
In 2011, the ARDS Definition Task Force, using a novel consensus process, updated the ARDS definition,9 focusing on its feasibility, reliability, and validity in predicting response to therapies and outcomes in ARDS. This new “Berlin” definition is not substantially different from the old, but defines the criteria more specifically:
- Bilateral opacities, unexplained by nodules, atelectasis, or effusion, on chest radiography or computed tomography
- New or worsening respiratory symptoms, or a clinical insult associated with ARDS within 7 days of diagnosis
- Objective assessment of cardiac function (eg, with echocardiography) to exclude cardiogenic pulmonary edema
- Hypoxemia, with a partial pressure of arterial oxygen divided by the percentage of inspired oxygen (PaO2/FiO2 ratio) of 300 mm Hg or less despite noninvasive or invasive mechanical ventilation with PEEP or continuous positive airway pressure (CPAP) of at least 5 cm H2O.
In addition, the new definition classifies the severity of disease on the basis of the degree of hypoxemia, ie, the PaO2/FiO2 ratio:
- Mild: PaO2/FiO2 ratio > 200 and ≤ 300 mm Hg
- Moderate: PaO2/FiO2 ratio > 100 and ≤ 200 mm Hg
- Severe: PaO2/FiO2 ratio ≤ 100 mm Hg.
The term “acute lung injury” has been eliminated, as has the previous criterion of a pulmonary artery wedge pressure of 18 mm Hg or less.
The panel also evaluated four ancillary variables for predicting outcomes in severe ARDS:
- Compliance of the respiratory system less than or equal to 40 mL/cm H2O
- Radiographic severity (involvement of three or four quadrants on chest radiography)
- PEEP of 10 cm H2O or greater
- Corrected expired volume 10 L/min or greater.
The task force evaluated the reliability and validity of this definition in a meta-analysis of 4,400 patients previously enrolled in randomized controlled trials or observational studies.
Findings. The Berlin definition predicted the risk of death better than the AECC definition. The mortality rate increased with the severity of ARDS, from 27% with mild disease to 32% with moderate disease to 45% with severe disease. The four ancillary variables did not contribute to the predictive validity of severe ARDS for mortality and were removed from the definition.
Thille et al10 retrospectively reviewed autopsy findings from 712 patients and found that the new definition identified a homogeneous group who had severe ARDS.10
Conclusions. The new definition may overcome some of the limitations of the old one, but it needs to be validated in clinical practice, especially its ability to predict death.
VENTILATORY SUPPORT
Prompt recognition, lung-protective ventilation, and a conservative fluid strategy remain the cornerstones of ARDS management. However, other strategies are being tested.
Prone-position ventilation in severe ARDS: The right therapy in a specific population
Prone-position ventilation was first described almost 30 years ago, but it has been used inconsistently in clinical practice.
Physiologic and observational studies indicated that prone positioning might improve survival in patients with ARDS, but several randomized trials failed to demonstrate any positive effect on outcomes.11,12 Some trials also reported a higher rate of complications with this intervention.13 However, meta-analyses suggested that prone-position ventilation might have a beneficial effect in patients with severe ARDS (defined as a PaO2/FiO2 ratio ≤ 100 mm Hg).14
In view of these findings, investigators conducted a trial of prone-position ventilation exclusively in patients with severe ARDS.
The PROSEVA study
The Proning Severe ARDS Patients (PROSEVA) study was a randomized controlled trial designed to determine whether prone-position ventilation, applied early, would improve outcomes in patients with severe ARDS.15
In PROSEVA, 466 patients with severe ARDS (defined as a PaO2/FiO2 ratio < 150 mm Hg, FiO2 ≥ 60%, and PEEP ≥ 5 cm H2O) underwent either at least 16 hours of prone positioning or were left in the supine position after 12 to 24 hours of initial conventional mechanical ventilation. The patients were recruited from centers in France and Spain where prone-position ventilation had been used in daily practice for more than 5 years.
The primary outcome studied was the rate of death at 28 days. The secondary end points were the death rate at day 90, rates of successful extubation, the length of stay in the intensive care unit, and complications.
Findings. At study entry, the patients in the supine group were sicker, more of them required a vasopressor, and fewer of them were receiving neuromuscular blocking agents than those in the prone group. These baseline differences may have influenced the outcomes; the unadjusted 28-day mortality rate was 16.0% in the prone group compared with 32.8% in the supine group (P < .001). However, the hazard ratio for death with prone positioning was 0.39 (95% confidence interval [CI] 0.25–0.63) even after adjusting for severity and the use of vasopressors and neuromuscular blocking agents. Prone-position ventilation was not associated with a higher incidence of complications, and the rate of successful extubation was higher.
Conclusions. In patients with severe ARDS, early use of prolonged prone positioning significantly decreased the 28-day and 90-day mortality rates. This trial has made prone positioning one of the strategies in managing patients with early severe ARDS. To minimize complications such as pressure ulcers and line or tube dislodgement, personnel caring for these patients must follow a protocol and undergo specific training.
These results were corroborated by a meta-analysis by Beitler et al16 that found a significant decrease in mortality rate with prone-position ventilation even in older studies when lung-protective ventilation strategies were separated from high-tidal-volume ventilation.
High-frequency oscillatory ventilation: No benefit in two trials
Observational data and experimental studies suggested that high-frequency oscillatory ventilation (HFOV) is superior to conventional mechanical ventilation in ARDS patients.17,18 However, outdated and cumbersome equipment, lack of protocols, and a lack of high-quality evidence led to limited and inconsistent use of HFOV, mainly as a rescue therapy in ARDS.19
Over the last few years, HFOV has been gaining acceptance, especially earlier in the course of ARDS.20 After preliminary clinical trials reported promising results, two trials conducted in Canada and the United Kingdom compared HFOV vs conventional mechanical ventilation in patients with ARDS.
The OSCAR study
The Oscillation in ARDS (OSCAR) study21 was a “pragmatic” trial22 (ie, it had minimal exclusion criteria) of the safety and effectiveness of HFOV as a primary ventilatory strategy for ARDS. It included 795 patients randomized to receive conventional ventilation (n = 397) or HFOV (n = 398). Research centers followed detailed algorithms for HFOV management and adopted their usual practice for conventional ventilation. Medical care was given according to the clinician’s judgment.
The primary outcome studied was survival at 30 days. The secondary outcomes were all-cause mortality in the intensive care unit and the hospital, duration of mechanical ventilation, and use of antimicrobial, sedative, vasoactive, and neuromuscular-blocking drugs.
Findings. The patient baseline characteristics were similar in both groups.
There was no significant difference in intensive care unit mortality rates, hospital mortality rates, or mortality rates at 30 days (41.7% in the HFOV group vs 41.1% in the conventional ventilation group; P = .85, 95% CI 6.1–7.5) even after adjustments for center or severity of illness.
The duration of mechanical ventilation was similar in both groups (14.9 ± 13.3 days in the HFOV group vs 14.1 ± 13.4 days in the conventional ventilation group, P = .41). However, sedatives and neuromuscular-blocking drugs were used more often and longer in the HFOV group than in the conventional ventilation group. There was no difference in the use of vasoactive or antimicrobial medications.
Conclusions. This multicenter randomized control trial did not demonstrate any benefit from using HFOV for routine management of ARDS. Its pragmatic design made it less likely to reach a firm conclusion,22 but it at least made a case against routinely using HFOV in patients with ARDS.
The OSCILLATE study
The Oscillation for Acute Respiratory Distress Syndrome Treated Early (OSCILLATE) study23 assessed the safety and efficacy of HFOV as a treatment for early-onset moderate-to-severe ARDS.
The inclusion criteria were similar to those in the OSCAR trial except that pulmonary symptoms had to be present less than 2 weeks and ARDS assessment was done under standard ventilator settings. As this was an efficacy trial, it had more exclusion criteria than the OSCAR trial. A total of 548 patients were randomized to receive conventional ventilation (n = 273) or HFOV (n = 275). The baseline characteristics were similar between groups.
Conventional ventilation was given according to a protocol used in an earlier trial2 and included recruitment maneuvers. HFOV was given in centers that had experience in this treatment, and there were protocols for ventilation management, hemodynamic optimization, and weaning. All other care was left to the clinician’s choice.
The primary outcome studied was in-hospital mortality. The investigators also evaluated whether there were interactions between the treatment and baseline severity of lung injury and center experience with HFOV.
Findings. The trial was stopped after an interim analysis found that HFOV might be harmful, although the statistical threshold for stopping was not reached. The in-hospital mortality rate was 47% in the HFOV group and 35% in the control group (relative risk of death with HFOV 1.33, 95% CI 1.09–1.64, P = .005). HFOV was worse than conventional ventilation regardless of the severity of disease or center experience. The HFOV group had higher mean airway pressures but similar FiO2 compared with the conventional ventilation group.
The HFOV group received significantly more vasopressors, sedatives, and neuromuscular blockers. This group’s fluid balance was higher as well, but not significantly so. Refractory hypoxemia (defined as PaO2 < 60 mm Hg for 1 hour with an FiO2 of 1.0 and neuromuscular blockade) was more frequent in the conventional ventilation group, but the number of deaths in the subgroup with refractory hypoxemia was similar with either treatment.
Conclusions. This multicenter randomized controlled trial demonstrated that HFOV was harmful when used routinely to manage ARDS. The trial’s protocol was based on the results of a pilot study carried out by the same investigators, which provided the best evidence available regarding the safety of HFOV at that time.
The results of the OSCAR and OSCILLATE trials have quelled enthusiasm for early, routine use of HFOV in ARDS. Although there are concerns that the protocol (ie, the way HFOV was implemented) rather than HFOV itself may have led to worse outcomes, there is no signal to support its routine use. We need further studies to define if it remains a viable rescue therapy.
Extracorporeal membrane oxygenation: Is it a viable option in severe ARDS?
Extracorporeal membrane oxygenation (ECMO) uses cardiopulmonary bypass technology to provide gas exchange. In patients with severe hypoxemia, ECMO can ensure adequate oxygenation and ventilation while ensuring the optimization of lung-protective ventilation. But ECMO was never as successful in adults with ARDS as it was in children and neonates.24
The first two trials of ECMO in ARDS24,25 reported equal or worse survival rates compared with conventional ventilation, and the overall mortality rate in these studies was staggeringly high. However, these studies were carried out before the era of lung-protective ventilation and at a time when ECMO technology was relatively primitive.
With new technology such as venovenous circuits and smaller cannulas, ECMO has gained more acceptance. It was used in patients with severe or refractory hypoxemia associated with ARDS during the H1N1 pandemic.26,27
The CESAR trial
The Conventional Ventilatory Support Versus Extracorporeal Membrane Oxygenation for Severe Adult Respiratory Failure (CESAR) trial28 assessed the safety, clinical efficacy, and cost-effectiveness of ECMO in managing severe ARDS. It compared best standard practice vs a protocol that included ECMO. The trial was conducted from 2001 to 2006.
Patients with severe ARDS, as defined by a Murray score29 greater than 3 or uncompensated hypercapnea, were prospectively randomized and recruited from an ECMO center and 148 tertiary intensive care units and referral hospitals in England. This was a pragmatic trial, with minimal exclusion criteria (essentially, mechanical ventilation with high pressures and high FiO2 for more than 7 days, intracranial bleeding, or contraindication to heparinization).
A total of 180 patients were randomized in a one-to-one ratio to receive ECMO or conventional management. The ventilator management in the conventional treatment group was not done according to a protocol but in general was low-volume and low-pressure. All patients randomized to ECMO were transferred to the ECMO center and treated according to a standardized ventilation protocol. After 12 hours, if predefined goals were not reached, venovenous ECMO was started. Patients assigned to conventional management could not cross over to ECMO.
The primary outcomes were death or severe disability at 6 months after randomization, and cost-effectiveness. The secondary outcomes were hospital resource use (eg, rescue techniques, length of stay, duration of ECMO) and health status after 6 months.
Findings. The groups were similar at baseline. Sixty-eight (75%) of the 90 patients randomized to receive ECMO actually received it. Of the 22 patients who did not receive ECMO, 16 (18% of the 90) improved on conventional therapy, 5 (6%) died during or before transfer, and 1 could not receive heparin.
Two patients had severe complications in the ECMO group: one had an arterial puncture, and one had an oxygen delivery failure during transport. In each case, these events contributed to the death of the patient.
More patients in the ECMO group received lung-protective ventilation, 84 (93%) vs 63 (70%).
The primary outcome, ie, death or severe disability at 6 months, occurred in 33 (37%) of the 90 patients in the ECMO group and in 46 (53%) of the patients in the conventional management group (relative risk 0.69, 95% CI 0.05–0.97, P = .03). More patients in the ECMO group survived, but the difference was not statistically significant (relative risk of death 0.73, 95% CI 0.52–1.03, P = .07). The most common cause of death in the ECMO group was multiorgan failure (42%), whereas in the conventional management group, the most common cause of death was respiratory failure (60%).
Length of stay in the hospital and in the critical care unit and health care costs were double for patients in the ECMO group. There was no difference in quality-of-life markers at 6 months in the survivors.
Conclusions. This pragmatic trial demonstrated that a protocol that includes ECMO could improve survival rates in ARDS.
Of note, the ECMO group got care in regional centers that used protocols. Therefore, in interpreting the results of this trial, we have to consider that being in a center with protocol-specified care for ARDS could drive some of the difference in mortality rates.
Regardless, this trial demonstrated that ECMO is feasible and led to better outcomes than expected. The findings were encouraging, and spurred the use of ECMO in severe ARDS during the 2009 H1N1 pandemic. Two propensity-matched studies and a number of case series reported a survival benefit associated with the use of ECMO in patients with severe ARDS.27,30
A recent meta-analysis also reported that ECMO might lower the mortality rate in ARDS; however, the patients in the H1N1 pandemic were younger and usually had isolated respiratory failure.31
The success of ECMO has opened new possibilities in the management of ARDS. As the technology improves and our experience increases, ECMO will likely gain more acceptance as a treatment for severe ARDS.
Airway pressure release ventilation
The use of airway pressure release ventilation and other ventilator modalities in ARDS is not supported by current evidence, though results of clinical trials may influence our practice in the future.
PHARMACOTHERAPY IN ARDS
The pathogenesis of ARDS includes damage to the alveolar-capillary membrane, with leakage of protein-rich edema fluid into alveoli. This damage is propagated by a complex inflammatory response including but not limited to neutrophil activation, free-radical formation, dysregulation of the coagulation system, and extensive release of inflammatory mediators.32,33 As a consequence, there are multiple potential targets for pharmacologic therapy in ARDS.
A variety of drugs, including corticosteroids, anti-inflammatory agents, immune-modulating agents, pulmonary vasodilators, antioxidants, and surfactants, have been studied in patients with ARDS.34 But effective pharmacotherapy for ARDS remains extremely limited.
Neuromuscular blockade in early severe ARDS
Mechanical ventilation can result in injurious stretching of the lung parenchyma, either from alveolar overdistention (volutrauma) or from continual recruitment and derecruitment of unstable lung units during the ventilator cycle (atelectrauma).35 Ventilator-induced lung injury can be exacerbated by asynchronous breathing.
In theory, neuromuscular blockers could minimize patient-ventilator asynchrony and provide much better control of tidal volume and pressure in patients with ARDS. This may result in less volutrauma and atelectrauma associated with asynchronous breathing. Data also suggest that cisatracurium (Nimbex), a neuromuscular blocking agent, may have a direct effect on the amount of inflammation in lungs with ARDS.36
The ACURASYS study
The ARDS et Curarisation Systématique (ACURASYS) study37 was a randomized trial in 340 patients undergoing mechanical ventilation for severe ARDS to evaluate the impact of neuromuscular blockade within the first 48 hours in this population.
The primary outcome was the mortality rate before hospital discharge or within 90 days of study entry. Secondary outcomes included the 28-day mortality rate, the rate of intensive care unit-acquired paresis, and the number of ventilator-free days. To be included, patients had to have been mechanically ventilated for less than 48 hours and to meet the AECC criteria for severe ARDS, with a PaO2/FiO2 ratio less than 150 mm Hg.
The intervention group received a continuous infusion of cisatracurium for 48 hours, while the control patients received placebo. Muscle strength was evaluated by clinical scoring of strength in different muscle groups.
Findings. The study groups were similar at baseline.
The crude 90-day mortality rate was lower in the cisatracurium group (31.6% vs 40.7%, P = .08). Regression analysis showed an improved 90-day survival rate with the use of this neuromuscular blocker after adjustment for severity of illness and the severity of ARDS (based on degree of hypoxemia and plateau pressures) (hazard ratio for death at 90 days 0.68; 95% CI 0.48–0.98; P = .04). The rate of paresis acquired in the intensive care unit did not differ significantly between the two groups.
Conclusion. In patients with severe ARDS, giving a neuromuscular blocking agent early improved the survival rate and increased the time off the ventilator without increasing muscle weakness.
These data are in line with similar findings from two other studies published by the same group.38,39 A meta-analysis of 432 patients showed that the use of neuromuscular blockade in early severe ARDS is associated with a statistically significant effect on early mortality (relative risk 0.66, 95% CI 0.50–0.87).40 The pooled analysis of these trials did not show any statistically significant critical-illness polyneuropathy.
These results need to be interpreted carefully, as we have inadequate data to see if they generalize to different intensive care units, and the evaluation and categorization of critical-illness polyneuropathy remains to be defined.
Cisatracurium is a promising treatment for moderate to severe ARDS and merits investigation in a large confirmatory randomized controlled trial.
Other pharmacologic agents
A number of other drugs have been studied in ARDS patients, including both inhaled and intravenous beta agonists,41,42 statins,43 and nutritional supplements.44 But as with other drugs previously studied in ARDS such as corticosteroids, N-acetylcysteine, and surfactant,34 these agents showed no effect on outcomes. In fact, a recent trial of intravenous salbutamol in ARDS patients was stopped after an interim analysis because of a higher incidence of arrhythmias and lactic acidosis with this agent.42
These findings reaffirm that pharmacologic therapy needs to be carefully considered, and potential harms associated with these therapies need to be addressed before they are introduced in the care of critically ill patients.
- Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. Acute Respiratory Distress Syndrome Network. N Engl J Med 2000; 342:1301–1308.
- Meade MO, Cook DJ, Guyatt GH, et al; Lung Open Ventilation Study Investigators. Ventilation strategy using low tidal volumes, recruitment maneuvers, and high positive end-expiratory pressure for acute lung injury and acute respiratory distress syndrome: a randomized controlled trial. JAMA 2008; 299:637–645.
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network; Wiedemann HP, Wheeler AP, Bernard GR, et al. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med 2006; 354:2564–2575.
- Girard TD, Kress JP, Fuchs BD, et al. Efficacy and safety of a paired sedation and ventilator weaning protocol for mechanically ventilated patients in intensive care (Awakening and Breathing Controlled trial): a randomised controlled trial. Lancet 2008; 371:126–134.
- Schweickert WD, Pohlman MC, Pohlman AS, et al. Early physical and occupational therapy in mechanically ventilated, critically ill patients: a randomised controlled trial. Lancet 2009; 373:1874–1882.
- Ely EW, Baker AM, Dunagan DP, et al. Effect on the duration of mechanical ventilation of identifying patients capable of breathing spontaneously. N Engl J Med 1996; 335:1864–1869.
- Bernard GR, Artigas A, Brigham KL, et al. The American-European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med 1994; 149:818–824.
- Ferguson ND, Fan E, Camporota L, et al. The Berlin definition of ARDS: an expanded rationale, justification, and supplementary material. Intensive Care Med 2012; 38:1573–1582.
- ARDS Definition Task Force; Ranieri VM, Rubenfeld GD, Thompson BT, et al. Acute respiratory distress syndrome: the Berlin definition. JAMA 2012; 307:2526–2533.
- Thille AW, Esteban A, Fernández-Segoviano P, et al. Comparison of the Berlin definition for acute respiratory distress syndrome with autopsy. Am J Respir Crit Care Med 2013; 187:761–767.
- Gattinoni L, Tognoni G, Pesenti A, et al; Prone-Supine Study Group. Effect of prone positioning on the survival of patients with acute respiratory failure. N Engl J Med 2001; 345:568–573.
- Taccone P, Pesenti A, Latini R, et al; Prone-Supine II Study Group. Prone positioning in patients with moderate and severe acute respiratory distress syndrome: a randomized controlled trial. JAMA 2009; 302:1977–1984.
- Mancebo J, Fernández R, Blanch L, et al. A multicenter trial of prolonged prone ventilation in severe acute respiratory distress syndrome. Am J Respir Crit Care Med 2006; 173:1233–1239.
- Sud S, Friedrich JO, Taccone P, et al. Prone ventilation reduces mortality in patients with acute respiratory failure and severe hypoxemia: systematic review and meta-analysis. Intensive Care Med 2010; 36:585–599.
- Guérin C, Reignier J, Richard JC, et al; PROSEVA Study Group. Prone positioning in severe acute respiratory distress syndrome. N Engl J Med 2013; 368:2159–2168.
- Beitler JR, Shaefi S, Montesi SB, et al. Prone positioning reduces mortality from acute respiratory distress syndrome in the low tidal volume era: a meta-analysis. Intensive Care Med 2014; 40:332–341.
- Chan KP, Stewart TE, Mehta S. High-frequency oscillatory ventilation for adult patients with ARDS. Chest 2007; 131:1907–1916.
- Fessler HE, Hager DN, Brower RG. Feasibility of very high-frequency ventilation in adults with acute respiratory distress syndrome. Crit Care Med 2008; 36:1043–1048.
- Mehta S, Granton J, MacDonald RJ, et al. High-frequency oscillatory ventilation in adults: the Toronto experience. Chest 2004; 126:518–527.
- Ferguson ND, Chiche JD, Kacmarek RM, et al. Combining high-frequency oscillatory ventilation and recruitment maneuvers in adults with early acute respiratory distress syndrome: the Treatment with Oscillation and an Open Lung Strategy (TOOLS) Trial pilot study. Crit Care Med 2005; 33:479–486.
- Young D, Lamb SE, Shah S, et al; OSCAR Study Group. High-frequency oscillation for acute respiratory distress syndrome. N Engl J Med 2013; 368:806–813.
- Thorpe KE, Zwarenstein M, Oxman AD, et al. A pragmatic-explanatory continuum indicator summary (PRECIS): a tool to help trial designers. J Clin Epidemiol 2009; 62:464–475.
- Ferguson ND, Cook DJ, Guyatt GH, et al; OSCILLATE Trial Investigators; Canadian Critical Care Trials Group. High-frequency oscillation in early acute respiratory distress syndrome. N Engl J Med 2013; 368:795–805.
- Morris AH, Wallace CJ, Menlove RL, et al. Randomized clinical trial of pressure-controlled inverse ratio ventilation and extracorporeal CO2 removal for adult respiratory distress syndrome. Am J Respir Crit Care Med 1994; 149:295–305.
- Zapol WM, Snider MT, Hill JD, et al. Extracorporeal membrane oxygenation in severe acute respiratory failure. A randomized prospective study. JAMA 1979; 242:2193–2196.
- Australia and New Zealand Extracorporeal Membrane Oxygenation (ANZ ECMO) Influenza Investigators; Davies A, Jones D, Bailey M, et al. Extracorporeal Membrane Oxygenation for 2009 Influenza A(H1N1) Acute Respiratory Distress Syndrome. JAMA 2009; 302:1888–1895.
- Pham T, Combes A, Rozé H, et al; REVA Research Network. Extracorporeal membrane oxygenation for pandemic influenza A(H1N1)-induced acute respiratory distress syndrome: a cohort study and propensity-matched analysis. Am J Respir Crit Care Med 2013; 187:276–285.
- Peek GJ, Mugford M, Tiruvoipati R, et al; CESAR trial collaboration. Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): a multicentre randomised controlled trial. Lancet 2009; 374:1351–1363.
- Murray JF, Matthay MA, Luce JM, Flick MR. An expanded definition of the adult respiratory distress syndrome. Am Rev Respir Dis 1988; 138:720–723.
- Noah MA, Peek GJ, Finney SJ, et al. Referral to an extracorporeal membrane oxygenation center and mortality among patients with severe 2009 influenza A(H1N1). JAMA 2011; 306:1659–1668.
- Zampieri FG, Mendes PV, Ranzani OT, et al. Extracorporeal membrane oxygenation for severe respiratory failure in adult patients: a systematic review and meta-analysis of current evidence. J Crit Care 2013; 28:998–1005.
- Raghavendran K, Pryhuber GS, Chess PR, Davidson BA, Knight PR, Notter RH. Pharmacotherapy of acute lung injury and acute respiratory distress syndrome. Curr Med Chem 2008; 15:1911–1924.
- Adhikari N, Burns KE, Meade MO. Pharmacologic treatments for acute respiratory distress syndrome and acute lung injury: systematic review and meta-analysis. Treat Respir Med 2004; 3:307–328.
- Adhikari N, Burns KE, Meade MO. Pharmacologic therapies for adults with acute lung injury and acute respiratory distress syndrome. Cochrane Database Syst Rev 2004; 4:CD004477.
- Terragni PP, Rosboch GL, Lisi A, Viale AG, Ranieri VM. How respiratory system mechanics may help in minimising ventilator-induced lung injury in ARDS patients. Eur Respir J Suppl 2003; 42:15s–21s.
- Forel JM, Roch A, Papazian L. Paralytics in critical care: not always the bad guy. Curr Opin Crit Care 2009; 15:59–66.
- Papazian L, Forel JM, Gacouin A, et al; ACURASYS Study Investigators. Neuromuscular blockers in early acute respiratory distress syndrome. N Engl J Med 2010; 363:1107–1116.
- Gainnier M, Roch A, Forel JM, et al. Effect of neuromuscular blocking agents on gas exchange in patients presenting with acute respiratory distress syndrome. Crit Care Med 2004; 32:113–19.
- Forel JM, Roch A, Marin V, et al. Neuromuscular blocking agents decrease inflammatory response in patients presenting with acute respiratory distress syndrome. Crit Care Med 2006; 34:2749–2757.
- Alhazzani W, Alshahrani M, Jaeschke R, et al. Neuromuscular blocking agents in acute respiratory distress syndrome: a systematic review and meta-analysis of randomized controlled trials. Crit Care 2013; 17:R43.
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network; Matthay MA, Brower RG, Carson S, et al. Randomized, placebo-controlled clinical trial of an aerosolized beta-2-agonist for treatment of acute lung injury. Am J Respir Crit Care Med 2011; 184:561–568.
- Gao Smith F, Perkins GD, Gates S, et al; BALTI-2 study investigators. Effect of intravenous beta-2 agonist treatment on clinical outcomes in acute respiratory distress syndrome (BALTI-2): a multicentre, randomised controlled trial. Lancet 2012; 379:229–235.
- Craig TR, Duffy MJ, Shyamsundar M, et al. A randomized clinical trial of hydroxymethylglutaryl-coenzyme a reductase inhibition for acute lung injury (The HARP Study). Am J Respir Crit Care Med 2011; 183:620–626.
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network; Rice TW, Wheeler AP, Thompson BT, et al. Initial trophic vs full enteral feeding in patients with acute lung injury: the EDEN randomized trial. JAMA 2012; 307:795–803.
- Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. Acute Respiratory Distress Syndrome Network. N Engl J Med 2000; 342:1301–1308.
- Meade MO, Cook DJ, Guyatt GH, et al; Lung Open Ventilation Study Investigators. Ventilation strategy using low tidal volumes, recruitment maneuvers, and high positive end-expiratory pressure for acute lung injury and acute respiratory distress syndrome: a randomized controlled trial. JAMA 2008; 299:637–645.
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network; Wiedemann HP, Wheeler AP, Bernard GR, et al. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med 2006; 354:2564–2575.
- Girard TD, Kress JP, Fuchs BD, et al. Efficacy and safety of a paired sedation and ventilator weaning protocol for mechanically ventilated patients in intensive care (Awakening and Breathing Controlled trial): a randomised controlled trial. Lancet 2008; 371:126–134.
- Schweickert WD, Pohlman MC, Pohlman AS, et al. Early physical and occupational therapy in mechanically ventilated, critically ill patients: a randomised controlled trial. Lancet 2009; 373:1874–1882.
- Ely EW, Baker AM, Dunagan DP, et al. Effect on the duration of mechanical ventilation of identifying patients capable of breathing spontaneously. N Engl J Med 1996; 335:1864–1869.
- Bernard GR, Artigas A, Brigham KL, et al. The American-European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med 1994; 149:818–824.
- Ferguson ND, Fan E, Camporota L, et al. The Berlin definition of ARDS: an expanded rationale, justification, and supplementary material. Intensive Care Med 2012; 38:1573–1582.
- ARDS Definition Task Force; Ranieri VM, Rubenfeld GD, Thompson BT, et al. Acute respiratory distress syndrome: the Berlin definition. JAMA 2012; 307:2526–2533.
- Thille AW, Esteban A, Fernández-Segoviano P, et al. Comparison of the Berlin definition for acute respiratory distress syndrome with autopsy. Am J Respir Crit Care Med 2013; 187:761–767.
- Gattinoni L, Tognoni G, Pesenti A, et al; Prone-Supine Study Group. Effect of prone positioning on the survival of patients with acute respiratory failure. N Engl J Med 2001; 345:568–573.
- Taccone P, Pesenti A, Latini R, et al; Prone-Supine II Study Group. Prone positioning in patients with moderate and severe acute respiratory distress syndrome: a randomized controlled trial. JAMA 2009; 302:1977–1984.
- Mancebo J, Fernández R, Blanch L, et al. A multicenter trial of prolonged prone ventilation in severe acute respiratory distress syndrome. Am J Respir Crit Care Med 2006; 173:1233–1239.
- Sud S, Friedrich JO, Taccone P, et al. Prone ventilation reduces mortality in patients with acute respiratory failure and severe hypoxemia: systematic review and meta-analysis. Intensive Care Med 2010; 36:585–599.
- Guérin C, Reignier J, Richard JC, et al; PROSEVA Study Group. Prone positioning in severe acute respiratory distress syndrome. N Engl J Med 2013; 368:2159–2168.
- Beitler JR, Shaefi S, Montesi SB, et al. Prone positioning reduces mortality from acute respiratory distress syndrome in the low tidal volume era: a meta-analysis. Intensive Care Med 2014; 40:332–341.
- Chan KP, Stewart TE, Mehta S. High-frequency oscillatory ventilation for adult patients with ARDS. Chest 2007; 131:1907–1916.
- Fessler HE, Hager DN, Brower RG. Feasibility of very high-frequency ventilation in adults with acute respiratory distress syndrome. Crit Care Med 2008; 36:1043–1048.
- Mehta S, Granton J, MacDonald RJ, et al. High-frequency oscillatory ventilation in adults: the Toronto experience. Chest 2004; 126:518–527.
- Ferguson ND, Chiche JD, Kacmarek RM, et al. Combining high-frequency oscillatory ventilation and recruitment maneuvers in adults with early acute respiratory distress syndrome: the Treatment with Oscillation and an Open Lung Strategy (TOOLS) Trial pilot study. Crit Care Med 2005; 33:479–486.
- Young D, Lamb SE, Shah S, et al; OSCAR Study Group. High-frequency oscillation for acute respiratory distress syndrome. N Engl J Med 2013; 368:806–813.
- Thorpe KE, Zwarenstein M, Oxman AD, et al. A pragmatic-explanatory continuum indicator summary (PRECIS): a tool to help trial designers. J Clin Epidemiol 2009; 62:464–475.
- Ferguson ND, Cook DJ, Guyatt GH, et al; OSCILLATE Trial Investigators; Canadian Critical Care Trials Group. High-frequency oscillation in early acute respiratory distress syndrome. N Engl J Med 2013; 368:795–805.
- Morris AH, Wallace CJ, Menlove RL, et al. Randomized clinical trial of pressure-controlled inverse ratio ventilation and extracorporeal CO2 removal for adult respiratory distress syndrome. Am J Respir Crit Care Med 1994; 149:295–305.
- Zapol WM, Snider MT, Hill JD, et al. Extracorporeal membrane oxygenation in severe acute respiratory failure. A randomized prospective study. JAMA 1979; 242:2193–2196.
- Australia and New Zealand Extracorporeal Membrane Oxygenation (ANZ ECMO) Influenza Investigators; Davies A, Jones D, Bailey M, et al. Extracorporeal Membrane Oxygenation for 2009 Influenza A(H1N1) Acute Respiratory Distress Syndrome. JAMA 2009; 302:1888–1895.
- Pham T, Combes A, Rozé H, et al; REVA Research Network. Extracorporeal membrane oxygenation for pandemic influenza A(H1N1)-induced acute respiratory distress syndrome: a cohort study and propensity-matched analysis. Am J Respir Crit Care Med 2013; 187:276–285.
- Peek GJ, Mugford M, Tiruvoipati R, et al; CESAR trial collaboration. Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): a multicentre randomised controlled trial. Lancet 2009; 374:1351–1363.
- Murray JF, Matthay MA, Luce JM, Flick MR. An expanded definition of the adult respiratory distress syndrome. Am Rev Respir Dis 1988; 138:720–723.
- Noah MA, Peek GJ, Finney SJ, et al. Referral to an extracorporeal membrane oxygenation center and mortality among patients with severe 2009 influenza A(H1N1). JAMA 2011; 306:1659–1668.
- Zampieri FG, Mendes PV, Ranzani OT, et al. Extracorporeal membrane oxygenation for severe respiratory failure in adult patients: a systematic review and meta-analysis of current evidence. J Crit Care 2013; 28:998–1005.
- Raghavendran K, Pryhuber GS, Chess PR, Davidson BA, Knight PR, Notter RH. Pharmacotherapy of acute lung injury and acute respiratory distress syndrome. Curr Med Chem 2008; 15:1911–1924.
- Adhikari N, Burns KE, Meade MO. Pharmacologic treatments for acute respiratory distress syndrome and acute lung injury: systematic review and meta-analysis. Treat Respir Med 2004; 3:307–328.
- Adhikari N, Burns KE, Meade MO. Pharmacologic therapies for adults with acute lung injury and acute respiratory distress syndrome. Cochrane Database Syst Rev 2004; 4:CD004477.
- Terragni PP, Rosboch GL, Lisi A, Viale AG, Ranieri VM. How respiratory system mechanics may help in minimising ventilator-induced lung injury in ARDS patients. Eur Respir J Suppl 2003; 42:15s–21s.
- Forel JM, Roch A, Papazian L. Paralytics in critical care: not always the bad guy. Curr Opin Crit Care 2009; 15:59–66.
- Papazian L, Forel JM, Gacouin A, et al; ACURASYS Study Investigators. Neuromuscular blockers in early acute respiratory distress syndrome. N Engl J Med 2010; 363:1107–1116.
- Gainnier M, Roch A, Forel JM, et al. Effect of neuromuscular blocking agents on gas exchange in patients presenting with acute respiratory distress syndrome. Crit Care Med 2004; 32:113–19.
- Forel JM, Roch A, Marin V, et al. Neuromuscular blocking agents decrease inflammatory response in patients presenting with acute respiratory distress syndrome. Crit Care Med 2006; 34:2749–2757.
- Alhazzani W, Alshahrani M, Jaeschke R, et al. Neuromuscular blocking agents in acute respiratory distress syndrome: a systematic review and meta-analysis of randomized controlled trials. Crit Care 2013; 17:R43.
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network; Matthay MA, Brower RG, Carson S, et al. Randomized, placebo-controlled clinical trial of an aerosolized beta-2-agonist for treatment of acute lung injury. Am J Respir Crit Care Med 2011; 184:561–568.
- Gao Smith F, Perkins GD, Gates S, et al; BALTI-2 study investigators. Effect of intravenous beta-2 agonist treatment on clinical outcomes in acute respiratory distress syndrome (BALTI-2): a multicentre, randomised controlled trial. Lancet 2012; 379:229–235.
- Craig TR, Duffy MJ, Shyamsundar M, et al. A randomized clinical trial of hydroxymethylglutaryl-coenzyme a reductase inhibition for acute lung injury (The HARP Study). Am J Respir Crit Care Med 2011; 183:620–626.
- National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network; Rice TW, Wheeler AP, Thompson BT, et al. Initial trophic vs full enteral feeding in patients with acute lung injury: the EDEN randomized trial. JAMA 2012; 307:795–803.
KEY POINTS
- The new definition of ARDS categorizes it as mild, moderate, or severe on the basis of oxygenation, specifically, the PaO2/FiO2 ratio.
- Neuromuscular blockade and prone positioning, used early in moderate or severe cases of ARDS, have shown some promise in trials, but questions remain about their application in critically ill patients.
- Based on two large trials, HFOV is no longer recommended as a primary therapy for ARDS, but it may still be considered as a rescue therapy in patients with refractory hypoxemia.
- In light of observational studies and randomized trials, ECMO should be considered an option in cases of refractory hypoxemia.
PICC Placement and Related Complications
Peripherally inserted central venous catheters (PICCs) are used for a variety of indications, including administration of long‐term intravenous (IV) antibiotics, home IV medications, chemotherapy, and parenteral nutrition.[1, 2, 3] Additionally, PICCs have also been recognized as an alternative to large‐bore central venous catheters such as subclavian or internal jugular central venous catheters. PICCs have been associated with fewer bloodstream infections in patients with cancer than tunneled catheters.[4] Compared to central venous catheters, they demonstrate reduced complication rates,[5] decreased cost,[6] and increased safety for longer durations of use.[1, 2, 3, 7, 8, 9]
Despite the numerous benefits of PICCs, Prandoni et al. estimate an all‐cause complication rate of 12% to 17% with the use of PICCs.[10] Associated complications include infection,[11] pain, bleeding, and mechanical dysfunction, all of which contribute to patient discomfort and additional healthcare costs.[12] Bloodstream infections, for example, had previously been thought to occur at a substantially lower rate in PICCs than central venous catheters.[13] However, a recent systematic review suggests the rate of PICC‐associated bloodstream infections in the inpatient setting is actually comparable to that of central venous catheters.[14] Perhaps the most serious PICC‐associated complication is catheter‐related venous thrombosis. A recent systematic review and meta‐analysis found evidence to suggest the rate of catheter‐related venous thrombosis was highest in patients with cancer or critical illness15; additionally, rates of thrombosis associated with PICCs were higher than those associated with subclavian or internal jugular central venous catheters.[15, 16] Fletcher et al. showed an 8.1% incidence of symptomatic PICC‐related upper extremity deep vein thrombosis (DVT) in the neurosurgical intensive care unit, with 15% of patients subsequently developing a pulmonary embolism.[17] A recent prospective, randomized controlled trial by Itkin et al. similarly demonstrated symptomatic DVT rates of approximately 4%.[18] However, in this study, when PICCs were routinely screened for thrombosis (with or without associated symptoms), approximately 72% demonstrated thrombosis,[18] suggesting that many PICC‐associated thromboses may be clinically undetected. This may have far‐reaching clinical significance, as pulmonary embolism complicates upper extremity DVT in 9% of cases and can result in a mortality rate as high as 25%.[10, 19]
Some strategies to reduce the rate of catheter‐related complications include identification of characteristics that put patients at risk. Many potential risk factors have been investigated, including catheter size,[12, 20, 21, 22, 23, 24] choice of vein,[24] location of catheter tip,[25] and history of malignancy or prior DVT.[12] However, to date, no definitive consensus has been reached. Special attention has been paid to the investigation of underlying risk factors and treatment for catheter‐related DVT, given its significant morbidity and mortality. Results have been equivocal, though, and in some instances, complicated by a diagnosis of underlying malignancy.[26, 27, 28]
As PICCs become more widely utilized, assessments of factors that place patients at greater risk of PICC‐related complications are needed.[21] The purpose of this study was to establish the incidence of complications associated with PICCs placed in the inpatient setting and examine risk factors predisposing patients to these complications.
MATERIALS AND METHODS
Study Design
A case control analysis of adult inpatients who underwent PICC placement between January 2009 and January 2010 was conducted at Scott & White Healthcare (now Baylor Scott & White Healthcare) to determine the incidence and risk factors for PICC‐associated complications.
Study Site
The study took place at Scott & White Memorial Hospital in Temple, Texas, a 636‐bed multispecialty teaching hospital and level 1 trauma center. It is part of a healthcare system that includes 12 hospitals and more than 60 regional clinics, all of which share an electronic medical record to enable full integration.
Human Subjects Approval
This study received approval from the institutional review board at Scott & White Healthcare.
PICC Placement Technique
Inpatient PICC placement was performed by the PICC consult service. The consult service was comprised of 3 separate provider teams: (1) internal medicine, including select hospitalists and internal medicine residents; (2) radiology, including interventional radiologists and radiology residents; and (3) nursing, including registered nurses with advanced training in PICC placement. Following placement of a consult, the PICC consult service assessed the patient, obtained consent, and subsequently placed the catheter. Members of the PICC consult service followed a system‐wide protocol wherein target veins were identified by ultrasound prior to attempting catheter placement, and actual placement of the PICC was ultrasound guided. Images obtained during the procedure were permanently documented in the medical record. At the time of this study, no formal protocol existed wherein target veins were mapped for caliber. Operators relied on their professional judgment to determine if vein caliber appeared sufficient to accommodate catheter placement.
All PICCs were placed using industry standard sterile precautions. A universally accepted modified Seldinger technique was used to obtain venous access.[29] A guidewire was then positioned in the desired vessel to facilitate proper venous placement of the catheter. During the course of the study period, catheters used were either single‐ (4 Fr) or double lumen (5 Fr).
Catheters were placed at the bedside by hospitalists or registered nurse teams; the location of the catheter tip at the cavoatrial junction was confirmed by chest radiography. Catheter insertions by radiologists were performed in the interventional radiology suite, and confirmation of location of the catheter tip was obtained with fluoroscopy.
PICC Maintenance
Following placement, nurses managed the PICC site according to nursing policy. Per policy, the site was assessed each shift. Documentation of assessment was recorded in nursing notes. Routine dressing changes were performed every 7 days, and as needed, to maintain a sterile site. Date and time of dressing changes were documented in nursing notes and on the PICC dressing. Catheter hubs and injection ports were disinfected with an antiseptic preparation for 15 seconds and allowed to air dry for 30 seconds prior to accessing the catheter. Catheters were flushed with 10 mL of normal saline before and after use. Any abnormality noted during PICC assessment was relayed to the primary provider. If the catheter did not flush readily or demonstrate appropriate blood return, nursing staff obtained an order for alteplase to be administered in an effort to salvage the line. PICCs were discontinued at the discretion of the healthcare provider.
Participants
Records of all patients 18 years of age and older who underwent PICC placement between January 2009 and January 2010 were reviewed (N=1444) for study inclusion. There were no exclusion criteria.
Data Collection
Patients who experienced complications were identified by electronic medical record review. One‐to‐one matching was performed for age and gender‐matched controls randomly selected from inpatients who underwent PICC placement during the same time period without complications. A total of 170 cases with PICC‐related complications were identified. One hundred seventy exact age‐ and gender‐matched controls, who based upon documentation available in the electronic medical record did not experience complications, were then randomly selected. Prior to data collection, the research team reviewed and discussed the data collection form and agreed upon a standardized protocol for data collection. Data collection was completed by authors J.M. and J.H. on the standardized data collection form. Although a formal analysis of inter‐rater agreement was not performed, J.M. and J.H. discussed any items where questions arose and arrived at a consensus decision regarding completion of the data point.
End points of the chart review were completion of medical therapy for which the PICC was indicated (eg, IV antibiotics or total parenteral nutrition [TPN]) or documentation of a complication that led to 1 of the following: discontinuation of the PICC or adjustment of either catheter placement or medical therapy. All complications were identified via International Classification of Diseases, 9th Revision codes and systematic chart review.
Complications resulting in discontinuation of the PICC, adjustment of catheter placement, or change in medical therapy were identified by review of nursing or physician documentation, and were categorized as follows: mechanical complications (defined as loss of the ability of the catheter to flush or draw properly, inadvertent catheter dislodgement, or retained portion of the catheter following catheter removal), catheter‐associated bloodstream infection (development of a positive blood culture attributable to the central catheter with no other clearly identifiable source of bacteremia present), cellulitis (defined as cellulitis in the extremity where the catheter was placed), bleeding from the site of catheter, fever (for which no other cause could be identified), and catheter‐associated thrombosis (identified by Doppler ultrasonography in patients exhibiting symptoms such as pain, swelling, redness, or warmth in the extremity in which the PICC was placed).[30]
Demographic data were collected, including insurance status, age, ethnicity, and gender. Clinical data included body mass index (BMI), presence of malnutrition (defined by a serum albumin of less than 3 g/dL),[31] previous or active cancer, previous DVT, use of anticoagulants (eg, warfarin, heparin, or low‐molecular‐weight heparin) or antiplatelet agent (eg, aspirin or clopidogrel) at the time of placement, and indication for PICC placement. A patient's history of previous or active cancer and previous DVT were identified by clinical documentation. Indications for PICC placement included: treating infectious processes (ie, infusion of antimicrobials), providing TPN, chemotherapy administration, and IV access. Catheter‐specific data were also collected and included venous access obtained (cephalic, basilic, brachial), catheter size (single lumen [4 Fr] or double lumen [5 Fr]), type of complication, and time to complication. The procedure note accompanying PICC placement was reviewed for data regarding time of day inserted (with after hours defined as documentation of placement occurring after 5 pm), and procedure operator to identify type of team (internal medicine, radiology, nursing) responsible for placement.
Data Analysis
Demographic characteristics and potential risk factors for patients in both the case and control groups of the study were summarized using descriptive statistics: mean ( standard deviation [SD]) for continuous variables and frequency (percent) for categorical variables. Univariate and multivariable conditional logistic regression analyses of variables that were potential risk factors of PICC‐related complications were utilized. A stepwise selection method was used for multivariable conditional logistic regression models. Alpha=0.2 was used for the significance to enter the model, and =0.05 was used for significance level to remain in the model. Attribution of PICC‐related complications was evaluated in terms of odds ratios (OR) and 95% confidence interval (CI). A P value of <0.05 indicated statistical significance. No prospective power analysis was performed. However, for a retrospective power analysis for 1:1 matching with 170 cases and 170 matched controls, assuming 20% of controls were affected and an of 0.05, one would achieve 80% power to detect an odds ratio of 2. SAS 9.2 (SAS Institute Inc., Cary, NC) was used for data analysis.
RESULTS
In 2009, 1444 PICCs were placed, and 170 cases in which patients experienced complications associated with PICC placement were identified, resulting in a complication rate of 11.77% (95% CI: 10.11%‐13.44%). The most common complications experienced by our patient population included catheter‐associated thrombosis (3%, n = 46), mechanical complications (4%, n=67), inadvertent catheter dislodgement (2%, n=36), mechanical dysfunction (2%, n=30), retained portion of the catheter following catheter removal (<1%, n=1), catheter‐associated bloodstream infections (2%, n=24), and cellulitis at the catheter insertion site (1%, n=15). Other documented complications included unexplained fever and bleeding (Table 1).
| Complication | N (%) |
|---|---|
| |
| Thrombosis | 46 (3) |
| Infection | 24 (2) |
| Cellulitis | 15 (1) |
| Mechanical complications* | 67 (4) |
| Unexplained fever | 15 (1) |
| Bleeding | 3 (0) |
| No complication | 1,274 (88) |
The mean age of the total cohort (N=340), comprised of case (N=170) and control (N=170) groups, was 58 years (SD 17), and 55% (n=94) were females. There were no significant differences in complications between groups based on ethnicity (P=0.66). In the case group, 46% (n=78) of PICCs were placed by the radiology team, 41% (n=69) were placed by the internal medicine team, and 14% (n=23) were placed by nursing. In the control group, 44% (n=74) of PICCs were placed by radiology, 36% (n=62) by internal medicine, and 20% (n=34) by nursing. Based on univariate conditional analysis, provider team was not significantly associated with complications (P=0.29).
Predictors of All‐Cause Complications
Based upon univariate conditional logistic regression analyses of complications related to PICC placement (N=340), the following variables demonstrated a statistically significant increased risk for complications: malnutrition (OR: 1.88 [95% CI: 1.023.44], P=0.04) and after‐hours placement (OR: 8.67 [95% CI: 2.62‐28.63], P=0.0004) (Table 2). Anticoagulation was associated with a decreased risk of complications (OR: 0.27 [95% CI: 0.16‐0.45], P=0.04). Based upon multivariable logistic regression analysis, after‐hours placement (OR: 9.52 [95% CI: 2.68‐33.78], P=0.0005) and BMI >30 (OR: 1.98 [95% CI: 1.09‐3.61], P=0.02) were significantly associated with an increased risk of PICC‐associated complications. Conversely, anticoagulation/antiplatelet use was associated with a decreased risk of complications (OR: 0.24 [95% CI: 0.14‐0.43], P<0.0001).
| Variable | Case, N (%) | Control, N (%) | Univariate | Multivariable | ||
|---|---|---|---|---|---|---|
| OR (95% CI) | P Value | AOR (95% CI) | P Value | |||
| ||||||
| Age, y, meanSD | 5817 | 5817 | ||||
| BMI, meanSD | 29.29.5 | 27.97.9 | 1.02 (0.991.05) | 0.12 | ||
| 30 | 108 (64) | 116 (68%) | 1.00 | 1.00 | ||
| >30 | 62 (36) | 54 (32%) | 1.29 (0.792.11) | 0.32 | 1.98 (1.093.61) | 0.02 |
| Length of stay, d, meanSD | 1822 | 1416 | 1.01 (1.001.03) | 0.06 | ||
| Length of stay group, d | 0.11a | |||||
| <7 | 41 (24) | 52 (31) | 1.00 | |||
| 729 | 101 (59) | 103 (61) | 1.19 (0.721.98) | 0.49 | ||
| 30 | 28 (16) | 15 (9) | 2.21 (1.074.58) | 0.03 | ||
| Gender | ||||||
| Female | 94 (55) | 94 (55) | ||||
| Male | 76 (45) | 76 (45) | ||||
| Ethnicity | 0.66a | |||||
| Caucasian | 131 (77) | 125 (74) | 1.00 | |||
| African American | 26 (15) | 28 (16) | 0.88 (0.481.60) | 0.67 | ||
| Hispanic/Asian | 13 (8) | 17 (10) | 0.70 (0.311.58) | 0.38 | ||
| Provider team | 0.29a | |||||
| Radiology | 78 (46) | 74 (44) | 1.00 | |||
| Internal medicine | 69 (41) | 62 (36) | 1.05 (0.681.64) | 0.82 | ||
| Nursing | 23 (14) | 34 (20) | 0.65 (0.351.19) | 0.16 | ||
| Insuranceb | 0.22a | |||||
| Private insurance | 46 (27) | 42 (25) | 1.00 | |||
| Uninsured | 17 (10) | 24 (14) | 0.73 (0.351.55) | 0.41 | ||
| Medicare | 57 (34) | 62 (37) | 0.73 (0.381.40) | 0.34 | ||
| Medicaid | 39 (23) | 25 (15) | 1.51 (0.743.06) | 0.26 | ||
| Tricare/Veterans Administration | 11 (6) | 16 (9) | 0.59 (0.241.45) | 0.25 | ||
| History of DVT | 27 (16) | 26 (15) | 1.05 (0.581.91) | 0.88 | ||
| Malnutritionb | 149 (88) | 134 (79) | 1.88 (1.023.44) | 0.04 | ||
| Cancer | 25 (15) | 36 (21) | 0.58 (0.311.09) | 0.09 | ||
| Fluoroscopy | 129 (76) | 139 (82) | 0.71 (0.421.19) | 0.19 | ||
| Anticoagulation use | 50 (29) | 100 (59) | 0.27 (0.160.45) | <0.0001 | 0.24 (0.140.43) | <0.0001 |
| Multilumenc | 99 (58) | 111 (66) | 0.70 (0.441.11) | 0.13 | ||
| Veinb | 0.39a | |||||
| Basilic | 98 (58) | 86 (51) | 1.00 | |||
| Cephalic | 11 (6) | 8 (5) | 1.37 (0.483.89) | 0.55 | ||
| Brachial | 61 (36) | 74 (44) | 0.70 (0.451.09) | 0.12 | ||
| Internal mammary | 0 (0) | 1 (1) | <0.001 (<0.001>999) | 0.99 | ||
| Time of dayb | ||||||
| Morning/afternoon | 144 (85) | 166 (98) | 1.00 | 1.00 | ||
| After hours | 26 (15) | 3 (2) | 8.67 (2.6228.63) | 0.0004 | 9.52 (2.6833.78) | 0.0005 |
| Indication for PICC | 0.02a | |||||
| Infection | 88 (52) | 71 (42) | 1.00 | |||
| Pneumonia | 21 (12) | 14 (8) | 1.07 (0.502.29) | 0.87 | ||
| Chemotherapy | 5 (3) | 2 (1) | 1.84 (0.349.93) | 0.48 | ||
| IV access | 36 (21) | 66 (39) | 0.44 (0.250.75) | 0.003 | ||
| Total parenteral nutrition | 20 (12) | 17 (10) | 0.96 (0.442.14) | 0.93 | ||
Predictors of Nonmechanical Complications
To study risk factors related to nonmechanical complications, a secondary analysis (N=206) was performed in which all patients who experienced mechanical complications (N=67) and matched controls (N=67) were excluded. Based upon multivariable logistic regression analysis, after‐hours placement (OR: 6.93 [95% CI: 1.35‐35.56], P=0.02) and malnutrition (OR: 2.83 [95% CI: 1.037.81], P=0.04) were significantly associated with increased risk of nonmechanical complications. The use of anticoagulation/antiplatelet agents was associated with decreased risk of nonmechanical complications (OR: 0.17 [95% CI: 0.07‐0.40], P<0.0001). Variables not significantly associated with nonmechanical complications included BMI>30, previous history of DVT, history of cancer, catheter size, and venous access choice (Table 3).
| Variable | Case, N (%) | Control, N (%) | Univariate | Multivariable | ||
|---|---|---|---|---|---|---|
| OR (95% CI) | P Value | AOR (95% CI) | P Value | |||
| ||||||
| Age, y, meanSD | 5816 | 5816 | ||||
| BMI, meanSD | 29.79.8 | 28.57.9 | 1.03 (0.991.07) | 0.22 | ||
| 30 | 64 (62) | 68 (66) | 1.00 | |||
| >30 | 39 (38) | 35 (34) | 1.27 (0.642.49) | 0.49 | ||
| Length of stay, d, meanSD | 2026 | 1418 | 1.02 (1.001.03) | 0.08 | ||
| Length of stay group, d | 0.03a | |||||
| <7 | 22 (21) | 28 (27) | 1.00 | |||
| 729 | 60 (58) | 68 (66) | 0.95 (0.491.82) | 0.87 | ||
| 30 | 21 (20) | 7 (7) | 3.24 (1.238.54) | 0.02 | ||
| Gender | ||||||
| Female | 63 (61) | 63 (61) | ||||
| Male | 40 (39) | 40 (39) | ||||
| Ethnicity | 0.95a | |||||
| Caucasian | 75 (73) | 75 (73) | 1.00 | |||
| African American | 19 (18) | 18 (17) | 1.06 (0.512.21) | 0.87 | ||
| Hispanic/Asian | 9 (9) | 10 (10) | 0.88 (0.322.44) | 0.81 | ||
| Provider team | 0.81a | |||||
| Radiology | 43 (42) | 44 (43) | 1.00 | |||
| Internal medicine | 45 (44) | 41 (40) | 1.11 (0.621.96) | 0.73 | ||
| Nursing | 15 (15) | 18 (17) | 0.86 (0.391.90) | 0.71 | ||
| Insuranceb | 0.22a | |||||
| Private insurance | 29 (28) | 27 (26) | 1.00 | |||
| Uninsured | 13 (13) | 12 (12) | 1.18 (0.433.26) | 0.74 | ||
| Medicare | 32 (31) | 40 (39) | 0.52 (0.211.29) | 0.16 | ||
| Medicaid | 21 (20) | 12 (12) | 1.81 (0.694.74) | 0.23 | ||
| Tricare/Veterans Administration | 8 (8) | 11 (11) | 0.58 (0.191.79) | 0.34 | ||
| History of DVT | 15 (15) | 15 (15) | 1.00 (0.462.16) | 1.00 | ||
| Malnutritionb | 93 (90) | 79 (77) | 2.86 (1.216.76) | 0.02 | 2.83 (1.037.81) | 0.04 |
| Cancer | 17 (17) | 22 (21) | 0.67 (0.301.48) | 0.32 | ||
| Fluoroscopy | 78 (76) | 85 (83) | 0.65 (0.321.31) | 0.23 | ||
| Anticoagulation use | 29 (28) | 60 (58) | 0.21 (0.100.44) | <0.0001 | 0.17 (0.070.40) | <0.0001 |
| Multilumenc | 64 (62) | 67 (66) | 0.83 (0.461.51) | 0.55 | ||
| Veinb | 0.32a | |||||
| Basilic | 54 (52) | 49 (48) | 1.00 | |||
| Cephalic | 8 (8) | 3 (3) | 2.45 (0.649.32) | 0.19 | ||
| Brachial | 41 (40) | 49 (48) | 0.72 (0.421.24) | 0.24 | ||
| Internal mammary | 0 (0) | 1 (1) | <0.001 (<0.001>999) | 0.99 | ||
| Time of dayb | ||||||
| Morning/afternoon | 87 (84) | 100 (98) | 1.00 | 1.00 | ||
| After hours | 16 (16) | 2 (2) | 8.00 (1.8434.79) | 0.006 | 6.93 (1.3535.56) | 0.02 |
| Indication for PICC | 0.13 | |||||
| Infectiond | 52 (50) | 45 (44) | 1.00 | |||
| Pneumonia | 14 (14) | 7 (7) | 1.46 (0.514.18) | 0.48 | ||
| Chemotherapy | 5 (5) | 0 (0) | >999 (<0.001>999) | 0.99 | ||
| IV access | 22 (21) | 43 (42) | 0.48 (0.240.96) | 0.04 | ||
| Total parenteral nutrition | 10 (10) | 8 (8) | 1.08 (0.323.62) | 0.90 | ||
Predictors of Thrombotic Complications
Of 1444 patients who underwent PICC placement, 3% (n=46) were subsequently diagnosed with a catheter‐associated thrombosis, representing 27% of all observed complications. In an attempt to better identify factors predisposing patients to thrombotic complications, an additional subgroup analysis (N=92) was performed on those patients who experienced catheter‐associated thrombosis (N=46) and matched controls (N=46). Variables examined in the analysis included BMI, length of stay (LOS), history of DVT, history of cancer, utilization of anticoagulation/antiplatelet agents, malnutrition, and catheter size.
Based on conditional univariate analyses, the following variables were significantly associated with increased risk of catheter‐associated thrombosis: LOS (as a continuous variable) (OR: 1.04 [95% CI: 1.001.09], P=0.05), malnutrition (OR: 4 [95% CI: 1.1314.18], P=0.03), and after‐hours placement (OR: 8.00 [95% CI: 1.0063.96], P=0.05) (Table 4). Use of anticoagulation/antiplatelet agents (OR: 0.29 [95% CI: 0.11‐0.80], P=0.02) was associated with decreased risk of thrombosis. History of previous DVT and history of cancer were nonsignificant. In the multivariable logistic regression model, malnutrition (OR: 10.16 [95% CI: 1.76‐58.71], P=0.01) remained associated with increased risk of catheter‐associated thrombosis, whereas use of anticoagulation/antiplatelet agents (OR: 0.11 [95% CI: 0.02‐0.51], P=0.005) was associated with decreased risk of catheter‐associated thrombosis (Table 4).
| Variable | Case, N (%) | Control, N (%) | Univariate | Multivariable | ||
|---|---|---|---|---|---|---|
| OR (95% CI) | P Value | AOR (95% CI) | P Value | |||
| ||||||
| Age, y, meanSD | 5818 | 5818 | ||||
| BMI, meanSD | 27.77.1 | 27.77.8 | 1.00 (0.931.08) | 0.98 | ||
| 30 | 34 (74) | 33 (72) | ||||
| >30 | 12 (26) | 13 (28) | 0.83 (0.252.73) | 0.76 | ||
| Length of stay, d, meanSD | 1712 | 119 | 1.04 (1.001.09) | 0.05 | ||
| Length of stay group, d | 0.15 | |||||
| <7 | 8 (17) | 14 (30) | 1.00 | |||
| 729 | 29 (63) | 30 (65) | 1.13 (0.413.07) | 0.82 | ||
| 30 | 9 (20) | 2 (4) | 4.65 (0.9822.13) | 0.05 | ||
| Gender | ||||||
| Female | 26 (57) | 26 (57) | ||||
| Male | 20 (43) | 20 (43) | ||||
| Ethnicity | 0.44a | |||||
| Caucasian | 31 (67) | 36 (78) | 1.00 | |||
| African American | 11 (24) | 6 (13) | 2.02 (0.695.93) | 0.20 | ||
| Hispanic/Asian | 4 (9) | 4 (9) | 1.12 (0.225.68) | 0.89 | ||
| Provider team | 0.26a | |||||
| Radiology | 23 (50) | 19 (41) | 1.00 | |||
| Internal medicine | 20 (43) | 18 (39) | 1.00 (0.432.31) | 1.00 | ||
| Nursing | 3(7) | 9 (20) | 0.33 (0.091.27) | 0.11 | ||
| Insuranceb | 0.38a | |||||
| Private insurance | 13 (28) | 11 (24) | 1.00 | |||
| Uninsured | 8 (17) | 4 (9) | 2.01 (0.3810.58) | 0.41 | ||
| Medicare | 14 (30) | 21 (47) | 0.39 (0.101.47) | 0.16 | ||
| Medicaid | 8 (17) | 7 (16) | 1.23 (0.285.36) | 0.78 | ||
| Tricare/Veterans Administration | 3 (7) | 2 (4) | 1.01 (0.128.27) | 1.00 | ||
| History of DVT | 7 (15) | 8 (17) | 0.88 (0.322.41) | 0.80 | ||
| Malnutritionb | 43 (93) | 33 (73) | 4.00 (1.1314.18) | 0.03 | 10.16 (1.7658.71) | 0.01 |
| Cancer | 10 (22) | 13 (28) | 0.67 (0.241.87) | 0.44 | ||
| Fluoroscopy | 33 (72) | 39 (85) | 0.46 (0.161.31) | 0.14 | ||
| Anticoagulation use | 16 (35) | 28 (61) | 0.29 (0.110.80) | 0.02 | 0.11 (0.020.51) | 0.005 |
| Multilumenc | 22 (48) | 28 (62) | 0.53 (0.231.26) | 0.15 | ||
| Veinb | 0.93a | |||||
| Basilic | 24 (52) | 21 (47) | 1.00 | |||
| Cephalic | 1 (2) | 1 (2) | 0.86 (0.0514.39) | 0.92 | ||
| Brachial | 21 (46) | 22 (49) | 0.75 (0.311.79) | 0.51 | ||
| Internal mammary | 0 (0) | 1 (2) | <0.001 (<0.001>999) | 0.99 | ||
| Time of dayb | ||||||
| Morning/afternoon | 38 (83) | 44 (98) | 1.00 | |||
| After hours | 8 (17) | 1 (2) | 8.00 (1.0063.96) | 0.05 | ||
| Indication for PICC | 0.80a | |||||
| Infectiond | 20 (43) | 17 (37) | 1.00 | |||
| Pneumonia | 5 (11) | 6 (13) | 0.60 (0.142.56) | 0.49 | ||
| Chemotherapy | 3 (7) | 0 (0) | >999 (<0.001>999) | 0.99 | ||
| IV access | 14 (30) | 20 (43) | 0.58 (0.231.44) | 0.24 | ||
| Total parenteral nutrition | 4 (9) | 3 (7) | 1.22 (0.197.70) | 0.83 | ||
DISCUSSION
The goal of this study was to identify factors related to PICC placement that place the general population of patients at risk. The type and rate of complications associated with PICCs in this study were similar to those previously reported in the literature including catheter‐related infection and thrombosis.[10, 32] Two unique risk factors, not well recognized previously,[10, 27, 28, 33] were observed in this study: malnutrition and after‐hours placement. Malnutrition, defined as serum albumin <3 g/dL was associated with an increase in PICC‐related complications (such as catheter‐associated bloodstream infections and cellulitis) and catheter‐related thrombosis. Malnutrition itself has long been associated with a decreased resistance to infection[34]; in addition, low serum albumin may also be a marker of the presence of other severe comorbidities, which may contribute to increased risk of thrombosis. It has been noted in previous studies that critical illness increases risk of thrombosis.[15] Despite an exhaustive search of the literature, we have been unable to find additional studies examining the extent to which malnutrition may impact PICC‐associated complications.
After‐hours placement was also associated with increased nonmechanical complications, as well as catheter‐related thrombosis. In an effort to improve both patient and consulting provider satisfaction and provide more expedient service, PICCs were often placed after hours (between 5 pm and 8 am) by both interventional radiology (n=14) and internal medicine (n=15) teams.
LOS has been associated with PICC placement complications in other studies.[12] In both primary and secondary analyses, hospital stays >30 days were associated with a higher risk of complications than hospitalizations <7 days. In light of the clinical significance of catheter‐related thrombosis, a subgroup analysis of patients with an LOS >30 days was conducted. The conditional univariate regression analysis showed an increased risk with greater LOS, malnutrition, and after‐hours placement. Use of anticoagulant or antiplatelet agents were associated with decreased risk of thrombosis (Table 4). The association between LOS and PICC‐related thrombosis is consistent with findings from Evans et al. involving 1728 patients in a similar center.[12] In these circumstances, increased LOS may be a surrogate marker for increased severity of illness, in that those patients who are more ill require lengthier hospitalizations. In a systematic review and meta‐analysis, Chopra et al. observed that increased severity of illness correlated with higher rates of catheter‐associated thrombosis, which is supportive of these findings.[15]
In the multivariate logistic regression analysis, BMI >30 was associated with a statistically significant increased risk for PICC‐associated complications after adjusting for anticoagulation and time of placement (Table 2). In the secondary analysis, where patients with mechanical complications were removed, BMI >30 was no longer associated with an increased risk for PICC‐associated complications (Table 3). This suggests that patients with a BMI >30 had an increased risk of mechanical complications, but were not necessarily at increased risk of developing other complications, such as catheter‐related thrombosis, infection, or bleeding. This finding is congruent with studies by Evans et al.,[12] who found no association between BMI and catheter‐associated thrombosis. Our association between BMI and complications is unique; to date, there are few additional studies that examine the extent to which BMI impacts the rate and type of complications associated with PICCs. At this time, the mechanism of the association between mechanical complications (such as inadvertent catheter removal or mechanical malfunction) and BMI is uncertain and warrants further investigation.
Use of Anticoagulant Agents
Anticoagulant (ie, any agent used for DVT prophylaxis or therapeutic anticoagulation) or antiplatelet agent use at the time of PICC placement and during the patient's hospitalization was associated with a decreased risk of thrombosis in our analysis. However, it should be noted that no specific anticoagulant agent was studied, and that antiplatelet agents were included in this analysis, unlike that of Evans et al.[12] Although current literature in oncologic populations, as well as the evidence‐based clinical practice guidelines, recommend against routine use of venous thromboprophylaxis in patients with central venous catheters,[33, 35, 36, 37] we believe this deserves further study, particularly in light of conflicting data in this area.[38, 39] Evans et al.[12] noted that although use of anticoagulants initially appeared to be associated with greater incidence of upper extremity venous thrombosis, when previous diagnosis of DVT was removed from the analysis the association was no longer significant.
In our analyses, no associations between catheter size, choice of venous access, history of previous deep venous thrombosis, or history of malignancy and risk for complications were found. Our findings differed from previous studies, where a relationship between increasing catheter bore size and site of access have been associated with increased PICC‐related thrombosis or other complications.[12, 20, 40, 41] There were also no significant differences in risk for complications between provider teams (eg, internal medicine, radiology, nursing) for PICCs placed during the morning or afternoon, which is consistent with findings by Funk et al.[1] Yet, after‐hours placement of PICCs was associated with greater complications than daytime placement. Although the exploration of factors associated with after‐hours placement was beyond the scope of this study, the findings from this study caused the authors, primarily comprised of members of the internal medicine inpatient medicine division, to reexamine the division's protocol on PICC placement. A consensus decision was made to discontinue after‐hours placement of PICCs by internal medicine teams in an effort to promote patient safety until further data could be collected. As a result, internal medicine teams no longer place PICCs after regular working hours at our institution.
Limitations
Limitations include the categorization of antiplatelet and anticoagulant agents together. We did not distinguish between high‐ and low‐dose aspirin, nor did we distinguish between therapeutic dosing of heparin and low‐molecular‐weight heparin versus DVT prophylaxis dosing. Additionally, for patients who were on warfarin or heparin drip, we did not evaluate for therapeutic range of international normalized ratio or partial thromboplastin time, as this was beyond the present scope of this study. In addition, malnutrition defined by albumin alone may have been somewhat narrow, as conditions aside from malnutrition can impact albumin levels. In future evaluations, this relationship may be clarified by including other determinants of clinical malnutrition including BMI <18 or the measurement of prealbumin. For determination of after‐hours placement of PICCs, we relied upon time of procedure dictation, assuming that all dictations immediately followed catheter placement. If there was a lapse in time between catheter placement and dictation, the category may have been recorded in error. Another limitation of after‐hours categorization was that we were unable to determine whether the PICC was placed on a weekend or holiday.
CONCLUSIONS AND FUTURE DIRECTIONS
Our results suggest that more stringent screening of patients undergoing PICC placement may reduce the risk of complications, with special attention to characteristics such as BMI >30, increased LOS, and protein‐calorie malnutrition (albumin <3). Furthermore, placement of PICC lines in emergent or after‐hours settings should be carefully considered and weighed against relative risks of central venous catheter placement. Further examination of the role anticoagulant and antiplatelet agents may have in the prevention of catheter‐related thrombosis should be undertaken. We hope that the identification of these risk factors will decrease the rate of complications and ultimately enhance patient safety and satisfaction.
Acknowledgments
The authors sincerely thank Glen Cryer, Publications Manager, Baylor Scott & White Health, for his assistance with this article.
Disclosures: Nothing to report.
- , , . Two‐year trends of peripherally inserted central catheter‐line complications at a tertiary‐care hospital: role of nursing expertise. Infect Control Hosp Epidemiol. 2001;22:377–379.
- , , . Peripherally inserted central catheters in the intensive care unit. J Intensive Care Med. 1996;11:49–54.
- , , , . Peripherally inserted central catheters in an acute‐care hospital. Arch Intern Med. 1994;154:1833–1837.
- , , , et al. Catheter‐associated bloodstream infection incidence and risk factors in adults with cancer: a prospective cohort study. J Hosp Infect. 2011;78:26–30.
- , . Peripherally inserted central catheters: a report of 2506 catheter days. JPEN J Parenter Enteral Nutr. 1995;19:133–136.
- , , , . Peripherally inserted central catheters in general medicine. Mayo Clin Proc. 1997;72:225–233.
- , , . Bedside placement of peripherally inserted central catheters: a cost‐effectiveness analysis. Radiology. 1998;206:423–428.
- , , , . Frequent asymptomatic pulmonary embolism in patients with deep venous thrombosis. JAMA. 1994;271:223–225.
- , . Experience with PICC at a university medical center. J Intraven Nurs. 1997;20:141–147.
- , , , et al. Upper‐extremity deep vein thrombosis. Risk factors, diagnosis, and complications. Arch Intern Med. 1997;157:57–62.
- , , , . Previous PICC placement may be associated with catheter‐related infections in hemodialysis patients. Cardiovasc Intervent Radiol. 2011;34:120–123.
- , , , et al. Risk of symptomatic DVT associated with peripherally inserted central catheters. Chest. 2010;138:803–810.
- . Be picky about PICCs. ACP Hospitalist, American College of Physicians website. Available at: http://www.acphospitalist.org/archives/2013/09/coverstory.htm. Accessed January 4, 2014.
- , , , , . The risk of bloodstream infection associated with peripherally inserted central catheters compared with central venous catheters in adults: a systematic review and meta‐analysis. Infect Control Hosp Epidemiol. 2013;34:908–918.
- , , , et al. Risk of venous thromboembolism associated with peripherally inserted central catheters: a systematic review and meta‐analysis. Lancet. 2013;382:311–325.
- , , , et al. Prevention of VTE in nonsurgical patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence‐based practice guidelines. Chest. 2012;141(2 suppl):e195S–e226S.
- , , . The clinical significance of peripherally inserted central venous catheter‐related deep vein thrombosis. Neurocrit Care. 2011;15:454–460.
- , , , , , . Peripherally inserted central catheter thrombosis‐reverse tapered versus nontapered catheters: a randomized controlled study. J Vasc Interv Radiol. 2014;25:85–91.
- , , , et al. Upper extremity venous thrombosis diagnosed by duplex scanning. Am J Surg. 1990;160:202–206.
- , , , et al. Reduction of peripherally inserted central catheter‐associated DVT. Chest. 2013;143:627–633.
- . Preventing DVT in peripherally inserted central catheters. Chest. 2013;143:589–590.
- , , , et al. Incidence of upper limb venous thrombosis associated with peripherally inserted central catheters (PICC). Br J Radiol. 2005;78:596–600.
- , , , , , . Computer identification of symptomatic deep venous thrombosis associated with peripherally inserted central catheters. AMIA Annu Symp Proc. 2007:226–230.
- , . Venous thrombosis related to peripherally inserted central catheters. J Vasc Interv Radiol. 2000;11:837–884.
- , , , , . To clot or not to clot? That is the question in central venous catheters. Clin Radiol. 2004;59:349–355.
- , , , et al. The Incidence of PICC line–associated thrombosis with and without the use of prophylactic anticoagulants. JPEN J Parenter Enteral Nutr. 2008;32:443–447.
- , , , , . Efficacy of multifaceted interventions in reducing complications of peripherally inserted central catheter in adult oncology patients. Support Care Cancer. 2010;18:1293–1298.
- , , , , . Peripherally inserted central venous catheter‐associated thrombosis: retrospective analysis of clinical risk factors in adult patients. South Med J. 2006;99:1073–1077.
- . The Seldinger method for PICC insertion. J Intraven Nurs. 1989;12:238–243.
- , , , et al. Venous thrombosis associated with the placement of peripherally inserted central catheters. J Vasc Interv Radiol. 2000;11:1309–1314.
- . Protein‐energy undernutrition. Merk Manual of Diagnosis and Therapy. 18th ed. Available at: http://www.merckmanuals.com/professional/nutritional_disorders/undernutrition/protein‐energy_undernutrition.html. Accessed May 26, 2013.
- , , , , . Risk of venous thromboembolism in hospitalized patients with peripherally inserted central catheters. J Hosp Med. 2009;4:417–422.
- , , , . Risk factors for upper extremity venous thrombosis associated with peripherally inserted central venous catheters. J Vasc Access. 2012;13:231–238.
- . Historical concepts of interactions, synergism and antagonism between nutrition and infection. J Nutr. 2003;133:316S–321S.
- , , , et al. 2008 SOR guidelines for the prevention and treatment of thrombosis associated with central venous catheters in patients with cancer: report from the working group. Ann Oncol. 2009;20:1459–1471.
- , , , , . Thromboprophylaxis in cancer patients with central venous catheters. A systematic review and meta‐analysis. Thromb Haemost. 2008;99:38–43.
- , , , . Thromboprophylaxis for catheter‐related thrombosis in patients with cancer: a systematic review of the randomized, controlled trials. J Thromb Haemost. 2007;5:2552–2554.
- , , , et al. Prevention of central venous catheter associated thrombosis using minidose warfarin in patients with haematological malignancies. Br J Haematol. 1998;10:483–486.
- , , , et al. Very low doses of warfarin can prevent thrombosis in central venous catheters. A randomized prospective trial. Ann Intern Med. 1990;112:423–428.
- , , , , , . The peripherally inserted central catheter (PICC): a prospective study of its natural history after cubital fossa insertion. Anesth Intensive Care. 2002;30:21–24.
- . Thrombotic complications in intravenous access. J Intraven Nurs. 1998;21:96–100.
Peripherally inserted central venous catheters (PICCs) are used for a variety of indications, including administration of long‐term intravenous (IV) antibiotics, home IV medications, chemotherapy, and parenteral nutrition.[1, 2, 3] Additionally, PICCs have also been recognized as an alternative to large‐bore central venous catheters such as subclavian or internal jugular central venous catheters. PICCs have been associated with fewer bloodstream infections in patients with cancer than tunneled catheters.[4] Compared to central venous catheters, they demonstrate reduced complication rates,[5] decreased cost,[6] and increased safety for longer durations of use.[1, 2, 3, 7, 8, 9]
Despite the numerous benefits of PICCs, Prandoni et al. estimate an all‐cause complication rate of 12% to 17% with the use of PICCs.[10] Associated complications include infection,[11] pain, bleeding, and mechanical dysfunction, all of which contribute to patient discomfort and additional healthcare costs.[12] Bloodstream infections, for example, had previously been thought to occur at a substantially lower rate in PICCs than central venous catheters.[13] However, a recent systematic review suggests the rate of PICC‐associated bloodstream infections in the inpatient setting is actually comparable to that of central venous catheters.[14] Perhaps the most serious PICC‐associated complication is catheter‐related venous thrombosis. A recent systematic review and meta‐analysis found evidence to suggest the rate of catheter‐related venous thrombosis was highest in patients with cancer or critical illness15; additionally, rates of thrombosis associated with PICCs were higher than those associated with subclavian or internal jugular central venous catheters.[15, 16] Fletcher et al. showed an 8.1% incidence of symptomatic PICC‐related upper extremity deep vein thrombosis (DVT) in the neurosurgical intensive care unit, with 15% of patients subsequently developing a pulmonary embolism.[17] A recent prospective, randomized controlled trial by Itkin et al. similarly demonstrated symptomatic DVT rates of approximately 4%.[18] However, in this study, when PICCs were routinely screened for thrombosis (with or without associated symptoms), approximately 72% demonstrated thrombosis,[18] suggesting that many PICC‐associated thromboses may be clinically undetected. This may have far‐reaching clinical significance, as pulmonary embolism complicates upper extremity DVT in 9% of cases and can result in a mortality rate as high as 25%.[10, 19]
Some strategies to reduce the rate of catheter‐related complications include identification of characteristics that put patients at risk. Many potential risk factors have been investigated, including catheter size,[12, 20, 21, 22, 23, 24] choice of vein,[24] location of catheter tip,[25] and history of malignancy or prior DVT.[12] However, to date, no definitive consensus has been reached. Special attention has been paid to the investigation of underlying risk factors and treatment for catheter‐related DVT, given its significant morbidity and mortality. Results have been equivocal, though, and in some instances, complicated by a diagnosis of underlying malignancy.[26, 27, 28]
As PICCs become more widely utilized, assessments of factors that place patients at greater risk of PICC‐related complications are needed.[21] The purpose of this study was to establish the incidence of complications associated with PICCs placed in the inpatient setting and examine risk factors predisposing patients to these complications.
MATERIALS AND METHODS
Study Design
A case control analysis of adult inpatients who underwent PICC placement between January 2009 and January 2010 was conducted at Scott & White Healthcare (now Baylor Scott & White Healthcare) to determine the incidence and risk factors for PICC‐associated complications.
Study Site
The study took place at Scott & White Memorial Hospital in Temple, Texas, a 636‐bed multispecialty teaching hospital and level 1 trauma center. It is part of a healthcare system that includes 12 hospitals and more than 60 regional clinics, all of which share an electronic medical record to enable full integration.
Human Subjects Approval
This study received approval from the institutional review board at Scott & White Healthcare.
PICC Placement Technique
Inpatient PICC placement was performed by the PICC consult service. The consult service was comprised of 3 separate provider teams: (1) internal medicine, including select hospitalists and internal medicine residents; (2) radiology, including interventional radiologists and radiology residents; and (3) nursing, including registered nurses with advanced training in PICC placement. Following placement of a consult, the PICC consult service assessed the patient, obtained consent, and subsequently placed the catheter. Members of the PICC consult service followed a system‐wide protocol wherein target veins were identified by ultrasound prior to attempting catheter placement, and actual placement of the PICC was ultrasound guided. Images obtained during the procedure were permanently documented in the medical record. At the time of this study, no formal protocol existed wherein target veins were mapped for caliber. Operators relied on their professional judgment to determine if vein caliber appeared sufficient to accommodate catheter placement.
All PICCs were placed using industry standard sterile precautions. A universally accepted modified Seldinger technique was used to obtain venous access.[29] A guidewire was then positioned in the desired vessel to facilitate proper venous placement of the catheter. During the course of the study period, catheters used were either single‐ (4 Fr) or double lumen (5 Fr).
Catheters were placed at the bedside by hospitalists or registered nurse teams; the location of the catheter tip at the cavoatrial junction was confirmed by chest radiography. Catheter insertions by radiologists were performed in the interventional radiology suite, and confirmation of location of the catheter tip was obtained with fluoroscopy.
PICC Maintenance
Following placement, nurses managed the PICC site according to nursing policy. Per policy, the site was assessed each shift. Documentation of assessment was recorded in nursing notes. Routine dressing changes were performed every 7 days, and as needed, to maintain a sterile site. Date and time of dressing changes were documented in nursing notes and on the PICC dressing. Catheter hubs and injection ports were disinfected with an antiseptic preparation for 15 seconds and allowed to air dry for 30 seconds prior to accessing the catheter. Catheters were flushed with 10 mL of normal saline before and after use. Any abnormality noted during PICC assessment was relayed to the primary provider. If the catheter did not flush readily or demonstrate appropriate blood return, nursing staff obtained an order for alteplase to be administered in an effort to salvage the line. PICCs were discontinued at the discretion of the healthcare provider.
Participants
Records of all patients 18 years of age and older who underwent PICC placement between January 2009 and January 2010 were reviewed (N=1444) for study inclusion. There were no exclusion criteria.
Data Collection
Patients who experienced complications were identified by electronic medical record review. One‐to‐one matching was performed for age and gender‐matched controls randomly selected from inpatients who underwent PICC placement during the same time period without complications. A total of 170 cases with PICC‐related complications were identified. One hundred seventy exact age‐ and gender‐matched controls, who based upon documentation available in the electronic medical record did not experience complications, were then randomly selected. Prior to data collection, the research team reviewed and discussed the data collection form and agreed upon a standardized protocol for data collection. Data collection was completed by authors J.M. and J.H. on the standardized data collection form. Although a formal analysis of inter‐rater agreement was not performed, J.M. and J.H. discussed any items where questions arose and arrived at a consensus decision regarding completion of the data point.
End points of the chart review were completion of medical therapy for which the PICC was indicated (eg, IV antibiotics or total parenteral nutrition [TPN]) or documentation of a complication that led to 1 of the following: discontinuation of the PICC or adjustment of either catheter placement or medical therapy. All complications were identified via International Classification of Diseases, 9th Revision codes and systematic chart review.
Complications resulting in discontinuation of the PICC, adjustment of catheter placement, or change in medical therapy were identified by review of nursing or physician documentation, and were categorized as follows: mechanical complications (defined as loss of the ability of the catheter to flush or draw properly, inadvertent catheter dislodgement, or retained portion of the catheter following catheter removal), catheter‐associated bloodstream infection (development of a positive blood culture attributable to the central catheter with no other clearly identifiable source of bacteremia present), cellulitis (defined as cellulitis in the extremity where the catheter was placed), bleeding from the site of catheter, fever (for which no other cause could be identified), and catheter‐associated thrombosis (identified by Doppler ultrasonography in patients exhibiting symptoms such as pain, swelling, redness, or warmth in the extremity in which the PICC was placed).[30]
Demographic data were collected, including insurance status, age, ethnicity, and gender. Clinical data included body mass index (BMI), presence of malnutrition (defined by a serum albumin of less than 3 g/dL),[31] previous or active cancer, previous DVT, use of anticoagulants (eg, warfarin, heparin, or low‐molecular‐weight heparin) or antiplatelet agent (eg, aspirin or clopidogrel) at the time of placement, and indication for PICC placement. A patient's history of previous or active cancer and previous DVT were identified by clinical documentation. Indications for PICC placement included: treating infectious processes (ie, infusion of antimicrobials), providing TPN, chemotherapy administration, and IV access. Catheter‐specific data were also collected and included venous access obtained (cephalic, basilic, brachial), catheter size (single lumen [4 Fr] or double lumen [5 Fr]), type of complication, and time to complication. The procedure note accompanying PICC placement was reviewed for data regarding time of day inserted (with after hours defined as documentation of placement occurring after 5 pm), and procedure operator to identify type of team (internal medicine, radiology, nursing) responsible for placement.
Data Analysis
Demographic characteristics and potential risk factors for patients in both the case and control groups of the study were summarized using descriptive statistics: mean ( standard deviation [SD]) for continuous variables and frequency (percent) for categorical variables. Univariate and multivariable conditional logistic regression analyses of variables that were potential risk factors of PICC‐related complications were utilized. A stepwise selection method was used for multivariable conditional logistic regression models. Alpha=0.2 was used for the significance to enter the model, and =0.05 was used for significance level to remain in the model. Attribution of PICC‐related complications was evaluated in terms of odds ratios (OR) and 95% confidence interval (CI). A P value of <0.05 indicated statistical significance. No prospective power analysis was performed. However, for a retrospective power analysis for 1:1 matching with 170 cases and 170 matched controls, assuming 20% of controls were affected and an of 0.05, one would achieve 80% power to detect an odds ratio of 2. SAS 9.2 (SAS Institute Inc., Cary, NC) was used for data analysis.
RESULTS
In 2009, 1444 PICCs were placed, and 170 cases in which patients experienced complications associated with PICC placement were identified, resulting in a complication rate of 11.77% (95% CI: 10.11%‐13.44%). The most common complications experienced by our patient population included catheter‐associated thrombosis (3%, n = 46), mechanical complications (4%, n=67), inadvertent catheter dislodgement (2%, n=36), mechanical dysfunction (2%, n=30), retained portion of the catheter following catheter removal (<1%, n=1), catheter‐associated bloodstream infections (2%, n=24), and cellulitis at the catheter insertion site (1%, n=15). Other documented complications included unexplained fever and bleeding (Table 1).
| Complication | N (%) |
|---|---|
| |
| Thrombosis | 46 (3) |
| Infection | 24 (2) |
| Cellulitis | 15 (1) |
| Mechanical complications* | 67 (4) |
| Unexplained fever | 15 (1) |
| Bleeding | 3 (0) |
| No complication | 1,274 (88) |
The mean age of the total cohort (N=340), comprised of case (N=170) and control (N=170) groups, was 58 years (SD 17), and 55% (n=94) were females. There were no significant differences in complications between groups based on ethnicity (P=0.66). In the case group, 46% (n=78) of PICCs were placed by the radiology team, 41% (n=69) were placed by the internal medicine team, and 14% (n=23) were placed by nursing. In the control group, 44% (n=74) of PICCs were placed by radiology, 36% (n=62) by internal medicine, and 20% (n=34) by nursing. Based on univariate conditional analysis, provider team was not significantly associated with complications (P=0.29).
Predictors of All‐Cause Complications
Based upon univariate conditional logistic regression analyses of complications related to PICC placement (N=340), the following variables demonstrated a statistically significant increased risk for complications: malnutrition (OR: 1.88 [95% CI: 1.023.44], P=0.04) and after‐hours placement (OR: 8.67 [95% CI: 2.62‐28.63], P=0.0004) (Table 2). Anticoagulation was associated with a decreased risk of complications (OR: 0.27 [95% CI: 0.16‐0.45], P=0.04). Based upon multivariable logistic regression analysis, after‐hours placement (OR: 9.52 [95% CI: 2.68‐33.78], P=0.0005) and BMI >30 (OR: 1.98 [95% CI: 1.09‐3.61], P=0.02) were significantly associated with an increased risk of PICC‐associated complications. Conversely, anticoagulation/antiplatelet use was associated with a decreased risk of complications (OR: 0.24 [95% CI: 0.14‐0.43], P<0.0001).
| Variable | Case, N (%) | Control, N (%) | Univariate | Multivariable | ||
|---|---|---|---|---|---|---|
| OR (95% CI) | P Value | AOR (95% CI) | P Value | |||
| ||||||
| Age, y, meanSD | 5817 | 5817 | ||||
| BMI, meanSD | 29.29.5 | 27.97.9 | 1.02 (0.991.05) | 0.12 | ||
| 30 | 108 (64) | 116 (68%) | 1.00 | 1.00 | ||
| >30 | 62 (36) | 54 (32%) | 1.29 (0.792.11) | 0.32 | 1.98 (1.093.61) | 0.02 |
| Length of stay, d, meanSD | 1822 | 1416 | 1.01 (1.001.03) | 0.06 | ||
| Length of stay group, d | 0.11a | |||||
| <7 | 41 (24) | 52 (31) | 1.00 | |||
| 729 | 101 (59) | 103 (61) | 1.19 (0.721.98) | 0.49 | ||
| 30 | 28 (16) | 15 (9) | 2.21 (1.074.58) | 0.03 | ||
| Gender | ||||||
| Female | 94 (55) | 94 (55) | ||||
| Male | 76 (45) | 76 (45) | ||||
| Ethnicity | 0.66a | |||||
| Caucasian | 131 (77) | 125 (74) | 1.00 | |||
| African American | 26 (15) | 28 (16) | 0.88 (0.481.60) | 0.67 | ||
| Hispanic/Asian | 13 (8) | 17 (10) | 0.70 (0.311.58) | 0.38 | ||
| Provider team | 0.29a | |||||
| Radiology | 78 (46) | 74 (44) | 1.00 | |||
| Internal medicine | 69 (41) | 62 (36) | 1.05 (0.681.64) | 0.82 | ||
| Nursing | 23 (14) | 34 (20) | 0.65 (0.351.19) | 0.16 | ||
| Insuranceb | 0.22a | |||||
| Private insurance | 46 (27) | 42 (25) | 1.00 | |||
| Uninsured | 17 (10) | 24 (14) | 0.73 (0.351.55) | 0.41 | ||
| Medicare | 57 (34) | 62 (37) | 0.73 (0.381.40) | 0.34 | ||
| Medicaid | 39 (23) | 25 (15) | 1.51 (0.743.06) | 0.26 | ||
| Tricare/Veterans Administration | 11 (6) | 16 (9) | 0.59 (0.241.45) | 0.25 | ||
| History of DVT | 27 (16) | 26 (15) | 1.05 (0.581.91) | 0.88 | ||
| Malnutritionb | 149 (88) | 134 (79) | 1.88 (1.023.44) | 0.04 | ||
| Cancer | 25 (15) | 36 (21) | 0.58 (0.311.09) | 0.09 | ||
| Fluoroscopy | 129 (76) | 139 (82) | 0.71 (0.421.19) | 0.19 | ||
| Anticoagulation use | 50 (29) | 100 (59) | 0.27 (0.160.45) | <0.0001 | 0.24 (0.140.43) | <0.0001 |
| Multilumenc | 99 (58) | 111 (66) | 0.70 (0.441.11) | 0.13 | ||
| Veinb | 0.39a | |||||
| Basilic | 98 (58) | 86 (51) | 1.00 | |||
| Cephalic | 11 (6) | 8 (5) | 1.37 (0.483.89) | 0.55 | ||
| Brachial | 61 (36) | 74 (44) | 0.70 (0.451.09) | 0.12 | ||
| Internal mammary | 0 (0) | 1 (1) | <0.001 (<0.001>999) | 0.99 | ||
| Time of dayb | ||||||
| Morning/afternoon | 144 (85) | 166 (98) | 1.00 | 1.00 | ||
| After hours | 26 (15) | 3 (2) | 8.67 (2.6228.63) | 0.0004 | 9.52 (2.6833.78) | 0.0005 |
| Indication for PICC | 0.02a | |||||
| Infection | 88 (52) | 71 (42) | 1.00 | |||
| Pneumonia | 21 (12) | 14 (8) | 1.07 (0.502.29) | 0.87 | ||
| Chemotherapy | 5 (3) | 2 (1) | 1.84 (0.349.93) | 0.48 | ||
| IV access | 36 (21) | 66 (39) | 0.44 (0.250.75) | 0.003 | ||
| Total parenteral nutrition | 20 (12) | 17 (10) | 0.96 (0.442.14) | 0.93 | ||
Predictors of Nonmechanical Complications
To study risk factors related to nonmechanical complications, a secondary analysis (N=206) was performed in which all patients who experienced mechanical complications (N=67) and matched controls (N=67) were excluded. Based upon multivariable logistic regression analysis, after‐hours placement (OR: 6.93 [95% CI: 1.35‐35.56], P=0.02) and malnutrition (OR: 2.83 [95% CI: 1.037.81], P=0.04) were significantly associated with increased risk of nonmechanical complications. The use of anticoagulation/antiplatelet agents was associated with decreased risk of nonmechanical complications (OR: 0.17 [95% CI: 0.07‐0.40], P<0.0001). Variables not significantly associated with nonmechanical complications included BMI>30, previous history of DVT, history of cancer, catheter size, and venous access choice (Table 3).
| Variable | Case, N (%) | Control, N (%) | Univariate | Multivariable | ||
|---|---|---|---|---|---|---|
| OR (95% CI) | P Value | AOR (95% CI) | P Value | |||
| ||||||
| Age, y, meanSD | 5816 | 5816 | ||||
| BMI, meanSD | 29.79.8 | 28.57.9 | 1.03 (0.991.07) | 0.22 | ||
| 30 | 64 (62) | 68 (66) | 1.00 | |||
| >30 | 39 (38) | 35 (34) | 1.27 (0.642.49) | 0.49 | ||
| Length of stay, d, meanSD | 2026 | 1418 | 1.02 (1.001.03) | 0.08 | ||
| Length of stay group, d | 0.03a | |||||
| <7 | 22 (21) | 28 (27) | 1.00 | |||
| 729 | 60 (58) | 68 (66) | 0.95 (0.491.82) | 0.87 | ||
| 30 | 21 (20) | 7 (7) | 3.24 (1.238.54) | 0.02 | ||
| Gender | ||||||
| Female | 63 (61) | 63 (61) | ||||
| Male | 40 (39) | 40 (39) | ||||
| Ethnicity | 0.95a | |||||
| Caucasian | 75 (73) | 75 (73) | 1.00 | |||
| African American | 19 (18) | 18 (17) | 1.06 (0.512.21) | 0.87 | ||
| Hispanic/Asian | 9 (9) | 10 (10) | 0.88 (0.322.44) | 0.81 | ||
| Provider team | 0.81a | |||||
| Radiology | 43 (42) | 44 (43) | 1.00 | |||
| Internal medicine | 45 (44) | 41 (40) | 1.11 (0.621.96) | 0.73 | ||
| Nursing | 15 (15) | 18 (17) | 0.86 (0.391.90) | 0.71 | ||
| Insuranceb | 0.22a | |||||
| Private insurance | 29 (28) | 27 (26) | 1.00 | |||
| Uninsured | 13 (13) | 12 (12) | 1.18 (0.433.26) | 0.74 | ||
| Medicare | 32 (31) | 40 (39) | 0.52 (0.211.29) | 0.16 | ||
| Medicaid | 21 (20) | 12 (12) | 1.81 (0.694.74) | 0.23 | ||
| Tricare/Veterans Administration | 8 (8) | 11 (11) | 0.58 (0.191.79) | 0.34 | ||
| History of DVT | 15 (15) | 15 (15) | 1.00 (0.462.16) | 1.00 | ||
| Malnutritionb | 93 (90) | 79 (77) | 2.86 (1.216.76) | 0.02 | 2.83 (1.037.81) | 0.04 |
| Cancer | 17 (17) | 22 (21) | 0.67 (0.301.48) | 0.32 | ||
| Fluoroscopy | 78 (76) | 85 (83) | 0.65 (0.321.31) | 0.23 | ||
| Anticoagulation use | 29 (28) | 60 (58) | 0.21 (0.100.44) | <0.0001 | 0.17 (0.070.40) | <0.0001 |
| Multilumenc | 64 (62) | 67 (66) | 0.83 (0.461.51) | 0.55 | ||
| Veinb | 0.32a | |||||
| Basilic | 54 (52) | 49 (48) | 1.00 | |||
| Cephalic | 8 (8) | 3 (3) | 2.45 (0.649.32) | 0.19 | ||
| Brachial | 41 (40) | 49 (48) | 0.72 (0.421.24) | 0.24 | ||
| Internal mammary | 0 (0) | 1 (1) | <0.001 (<0.001>999) | 0.99 | ||
| Time of dayb | ||||||
| Morning/afternoon | 87 (84) | 100 (98) | 1.00 | 1.00 | ||
| After hours | 16 (16) | 2 (2) | 8.00 (1.8434.79) | 0.006 | 6.93 (1.3535.56) | 0.02 |
| Indication for PICC | 0.13 | |||||
| Infectiond | 52 (50) | 45 (44) | 1.00 | |||
| Pneumonia | 14 (14) | 7 (7) | 1.46 (0.514.18) | 0.48 | ||
| Chemotherapy | 5 (5) | 0 (0) | >999 (<0.001>999) | 0.99 | ||
| IV access | 22 (21) | 43 (42) | 0.48 (0.240.96) | 0.04 | ||
| Total parenteral nutrition | 10 (10) | 8 (8) | 1.08 (0.323.62) | 0.90 | ||
Predictors of Thrombotic Complications
Of 1444 patients who underwent PICC placement, 3% (n=46) were subsequently diagnosed with a catheter‐associated thrombosis, representing 27% of all observed complications. In an attempt to better identify factors predisposing patients to thrombotic complications, an additional subgroup analysis (N=92) was performed on those patients who experienced catheter‐associated thrombosis (N=46) and matched controls (N=46). Variables examined in the analysis included BMI, length of stay (LOS), history of DVT, history of cancer, utilization of anticoagulation/antiplatelet agents, malnutrition, and catheter size.
Based on conditional univariate analyses, the following variables were significantly associated with increased risk of catheter‐associated thrombosis: LOS (as a continuous variable) (OR: 1.04 [95% CI: 1.001.09], P=0.05), malnutrition (OR: 4 [95% CI: 1.1314.18], P=0.03), and after‐hours placement (OR: 8.00 [95% CI: 1.0063.96], P=0.05) (Table 4). Use of anticoagulation/antiplatelet agents (OR: 0.29 [95% CI: 0.11‐0.80], P=0.02) was associated with decreased risk of thrombosis. History of previous DVT and history of cancer were nonsignificant. In the multivariable logistic regression model, malnutrition (OR: 10.16 [95% CI: 1.76‐58.71], P=0.01) remained associated with increased risk of catheter‐associated thrombosis, whereas use of anticoagulation/antiplatelet agents (OR: 0.11 [95% CI: 0.02‐0.51], P=0.005) was associated with decreased risk of catheter‐associated thrombosis (Table 4).
| Variable | Case, N (%) | Control, N (%) | Univariate | Multivariable | ||
|---|---|---|---|---|---|---|
| OR (95% CI) | P Value | AOR (95% CI) | P Value | |||
| ||||||
| Age, y, meanSD | 5818 | 5818 | ||||
| BMI, meanSD | 27.77.1 | 27.77.8 | 1.00 (0.931.08) | 0.98 | ||
| 30 | 34 (74) | 33 (72) | ||||
| >30 | 12 (26) | 13 (28) | 0.83 (0.252.73) | 0.76 | ||
| Length of stay, d, meanSD | 1712 | 119 | 1.04 (1.001.09) | 0.05 | ||
| Length of stay group, d | 0.15 | |||||
| <7 | 8 (17) | 14 (30) | 1.00 | |||
| 729 | 29 (63) | 30 (65) | 1.13 (0.413.07) | 0.82 | ||
| 30 | 9 (20) | 2 (4) | 4.65 (0.9822.13) | 0.05 | ||
| Gender | ||||||
| Female | 26 (57) | 26 (57) | ||||
| Male | 20 (43) | 20 (43) | ||||
| Ethnicity | 0.44a | |||||
| Caucasian | 31 (67) | 36 (78) | 1.00 | |||
| African American | 11 (24) | 6 (13) | 2.02 (0.695.93) | 0.20 | ||
| Hispanic/Asian | 4 (9) | 4 (9) | 1.12 (0.225.68) | 0.89 | ||
| Provider team | 0.26a | |||||
| Radiology | 23 (50) | 19 (41) | 1.00 | |||
| Internal medicine | 20 (43) | 18 (39) | 1.00 (0.432.31) | 1.00 | ||
| Nursing | 3(7) | 9 (20) | 0.33 (0.091.27) | 0.11 | ||
| Insuranceb | 0.38a | |||||
| Private insurance | 13 (28) | 11 (24) | 1.00 | |||
| Uninsured | 8 (17) | 4 (9) | 2.01 (0.3810.58) | 0.41 | ||
| Medicare | 14 (30) | 21 (47) | 0.39 (0.101.47) | 0.16 | ||
| Medicaid | 8 (17) | 7 (16) | 1.23 (0.285.36) | 0.78 | ||
| Tricare/Veterans Administration | 3 (7) | 2 (4) | 1.01 (0.128.27) | 1.00 | ||
| History of DVT | 7 (15) | 8 (17) | 0.88 (0.322.41) | 0.80 | ||
| Malnutritionb | 43 (93) | 33 (73) | 4.00 (1.1314.18) | 0.03 | 10.16 (1.7658.71) | 0.01 |
| Cancer | 10 (22) | 13 (28) | 0.67 (0.241.87) | 0.44 | ||
| Fluoroscopy | 33 (72) | 39 (85) | 0.46 (0.161.31) | 0.14 | ||
| Anticoagulation use | 16 (35) | 28 (61) | 0.29 (0.110.80) | 0.02 | 0.11 (0.020.51) | 0.005 |
| Multilumenc | 22 (48) | 28 (62) | 0.53 (0.231.26) | 0.15 | ||
| Veinb | 0.93a | |||||
| Basilic | 24 (52) | 21 (47) | 1.00 | |||
| Cephalic | 1 (2) | 1 (2) | 0.86 (0.0514.39) | 0.92 | ||
| Brachial | 21 (46) | 22 (49) | 0.75 (0.311.79) | 0.51 | ||
| Internal mammary | 0 (0) | 1 (2) | <0.001 (<0.001>999) | 0.99 | ||
| Time of dayb | ||||||
| Morning/afternoon | 38 (83) | 44 (98) | 1.00 | |||
| After hours | 8 (17) | 1 (2) | 8.00 (1.0063.96) | 0.05 | ||
| Indication for PICC | 0.80a | |||||
| Infectiond | 20 (43) | 17 (37) | 1.00 | |||
| Pneumonia | 5 (11) | 6 (13) | 0.60 (0.142.56) | 0.49 | ||
| Chemotherapy | 3 (7) | 0 (0) | >999 (<0.001>999) | 0.99 | ||
| IV access | 14 (30) | 20 (43) | 0.58 (0.231.44) | 0.24 | ||
| Total parenteral nutrition | 4 (9) | 3 (7) | 1.22 (0.197.70) | 0.83 | ||
DISCUSSION
The goal of this study was to identify factors related to PICC placement that place the general population of patients at risk. The type and rate of complications associated with PICCs in this study were similar to those previously reported in the literature including catheter‐related infection and thrombosis.[10, 32] Two unique risk factors, not well recognized previously,[10, 27, 28, 33] were observed in this study: malnutrition and after‐hours placement. Malnutrition, defined as serum albumin <3 g/dL was associated with an increase in PICC‐related complications (such as catheter‐associated bloodstream infections and cellulitis) and catheter‐related thrombosis. Malnutrition itself has long been associated with a decreased resistance to infection[34]; in addition, low serum albumin may also be a marker of the presence of other severe comorbidities, which may contribute to increased risk of thrombosis. It has been noted in previous studies that critical illness increases risk of thrombosis.[15] Despite an exhaustive search of the literature, we have been unable to find additional studies examining the extent to which malnutrition may impact PICC‐associated complications.
After‐hours placement was also associated with increased nonmechanical complications, as well as catheter‐related thrombosis. In an effort to improve both patient and consulting provider satisfaction and provide more expedient service, PICCs were often placed after hours (between 5 pm and 8 am) by both interventional radiology (n=14) and internal medicine (n=15) teams.
LOS has been associated with PICC placement complications in other studies.[12] In both primary and secondary analyses, hospital stays >30 days were associated with a higher risk of complications than hospitalizations <7 days. In light of the clinical significance of catheter‐related thrombosis, a subgroup analysis of patients with an LOS >30 days was conducted. The conditional univariate regression analysis showed an increased risk with greater LOS, malnutrition, and after‐hours placement. Use of anticoagulant or antiplatelet agents were associated with decreased risk of thrombosis (Table 4). The association between LOS and PICC‐related thrombosis is consistent with findings from Evans et al. involving 1728 patients in a similar center.[12] In these circumstances, increased LOS may be a surrogate marker for increased severity of illness, in that those patients who are more ill require lengthier hospitalizations. In a systematic review and meta‐analysis, Chopra et al. observed that increased severity of illness correlated with higher rates of catheter‐associated thrombosis, which is supportive of these findings.[15]
In the multivariate logistic regression analysis, BMI >30 was associated with a statistically significant increased risk for PICC‐associated complications after adjusting for anticoagulation and time of placement (Table 2). In the secondary analysis, where patients with mechanical complications were removed, BMI >30 was no longer associated with an increased risk for PICC‐associated complications (Table 3). This suggests that patients with a BMI >30 had an increased risk of mechanical complications, but were not necessarily at increased risk of developing other complications, such as catheter‐related thrombosis, infection, or bleeding. This finding is congruent with studies by Evans et al.,[12] who found no association between BMI and catheter‐associated thrombosis. Our association between BMI and complications is unique; to date, there are few additional studies that examine the extent to which BMI impacts the rate and type of complications associated with PICCs. At this time, the mechanism of the association between mechanical complications (such as inadvertent catheter removal or mechanical malfunction) and BMI is uncertain and warrants further investigation.
Use of Anticoagulant Agents
Anticoagulant (ie, any agent used for DVT prophylaxis or therapeutic anticoagulation) or antiplatelet agent use at the time of PICC placement and during the patient's hospitalization was associated with a decreased risk of thrombosis in our analysis. However, it should be noted that no specific anticoagulant agent was studied, and that antiplatelet agents were included in this analysis, unlike that of Evans et al.[12] Although current literature in oncologic populations, as well as the evidence‐based clinical practice guidelines, recommend against routine use of venous thromboprophylaxis in patients with central venous catheters,[33, 35, 36, 37] we believe this deserves further study, particularly in light of conflicting data in this area.[38, 39] Evans et al.[12] noted that although use of anticoagulants initially appeared to be associated with greater incidence of upper extremity venous thrombosis, when previous diagnosis of DVT was removed from the analysis the association was no longer significant.
In our analyses, no associations between catheter size, choice of venous access, history of previous deep venous thrombosis, or history of malignancy and risk for complications were found. Our findings differed from previous studies, where a relationship between increasing catheter bore size and site of access have been associated with increased PICC‐related thrombosis or other complications.[12, 20, 40, 41] There were also no significant differences in risk for complications between provider teams (eg, internal medicine, radiology, nursing) for PICCs placed during the morning or afternoon, which is consistent with findings by Funk et al.[1] Yet, after‐hours placement of PICCs was associated with greater complications than daytime placement. Although the exploration of factors associated with after‐hours placement was beyond the scope of this study, the findings from this study caused the authors, primarily comprised of members of the internal medicine inpatient medicine division, to reexamine the division's protocol on PICC placement. A consensus decision was made to discontinue after‐hours placement of PICCs by internal medicine teams in an effort to promote patient safety until further data could be collected. As a result, internal medicine teams no longer place PICCs after regular working hours at our institution.
Limitations
Limitations include the categorization of antiplatelet and anticoagulant agents together. We did not distinguish between high‐ and low‐dose aspirin, nor did we distinguish between therapeutic dosing of heparin and low‐molecular‐weight heparin versus DVT prophylaxis dosing. Additionally, for patients who were on warfarin or heparin drip, we did not evaluate for therapeutic range of international normalized ratio or partial thromboplastin time, as this was beyond the present scope of this study. In addition, malnutrition defined by albumin alone may have been somewhat narrow, as conditions aside from malnutrition can impact albumin levels. In future evaluations, this relationship may be clarified by including other determinants of clinical malnutrition including BMI <18 or the measurement of prealbumin. For determination of after‐hours placement of PICCs, we relied upon time of procedure dictation, assuming that all dictations immediately followed catheter placement. If there was a lapse in time between catheter placement and dictation, the category may have been recorded in error. Another limitation of after‐hours categorization was that we were unable to determine whether the PICC was placed on a weekend or holiday.
CONCLUSIONS AND FUTURE DIRECTIONS
Our results suggest that more stringent screening of patients undergoing PICC placement may reduce the risk of complications, with special attention to characteristics such as BMI >30, increased LOS, and protein‐calorie malnutrition (albumin <3). Furthermore, placement of PICC lines in emergent or after‐hours settings should be carefully considered and weighed against relative risks of central venous catheter placement. Further examination of the role anticoagulant and antiplatelet agents may have in the prevention of catheter‐related thrombosis should be undertaken. We hope that the identification of these risk factors will decrease the rate of complications and ultimately enhance patient safety and satisfaction.
Acknowledgments
The authors sincerely thank Glen Cryer, Publications Manager, Baylor Scott & White Health, for his assistance with this article.
Disclosures: Nothing to report.
Peripherally inserted central venous catheters (PICCs) are used for a variety of indications, including administration of long‐term intravenous (IV) antibiotics, home IV medications, chemotherapy, and parenteral nutrition.[1, 2, 3] Additionally, PICCs have also been recognized as an alternative to large‐bore central venous catheters such as subclavian or internal jugular central venous catheters. PICCs have been associated with fewer bloodstream infections in patients with cancer than tunneled catheters.[4] Compared to central venous catheters, they demonstrate reduced complication rates,[5] decreased cost,[6] and increased safety for longer durations of use.[1, 2, 3, 7, 8, 9]
Despite the numerous benefits of PICCs, Prandoni et al. estimate an all‐cause complication rate of 12% to 17% with the use of PICCs.[10] Associated complications include infection,[11] pain, bleeding, and mechanical dysfunction, all of which contribute to patient discomfort and additional healthcare costs.[12] Bloodstream infections, for example, had previously been thought to occur at a substantially lower rate in PICCs than central venous catheters.[13] However, a recent systematic review suggests the rate of PICC‐associated bloodstream infections in the inpatient setting is actually comparable to that of central venous catheters.[14] Perhaps the most serious PICC‐associated complication is catheter‐related venous thrombosis. A recent systematic review and meta‐analysis found evidence to suggest the rate of catheter‐related venous thrombosis was highest in patients with cancer or critical illness15; additionally, rates of thrombosis associated with PICCs were higher than those associated with subclavian or internal jugular central venous catheters.[15, 16] Fletcher et al. showed an 8.1% incidence of symptomatic PICC‐related upper extremity deep vein thrombosis (DVT) in the neurosurgical intensive care unit, with 15% of patients subsequently developing a pulmonary embolism.[17] A recent prospective, randomized controlled trial by Itkin et al. similarly demonstrated symptomatic DVT rates of approximately 4%.[18] However, in this study, when PICCs were routinely screened for thrombosis (with or without associated symptoms), approximately 72% demonstrated thrombosis,[18] suggesting that many PICC‐associated thromboses may be clinically undetected. This may have far‐reaching clinical significance, as pulmonary embolism complicates upper extremity DVT in 9% of cases and can result in a mortality rate as high as 25%.[10, 19]
Some strategies to reduce the rate of catheter‐related complications include identification of characteristics that put patients at risk. Many potential risk factors have been investigated, including catheter size,[12, 20, 21, 22, 23, 24] choice of vein,[24] location of catheter tip,[25] and history of malignancy or prior DVT.[12] However, to date, no definitive consensus has been reached. Special attention has been paid to the investigation of underlying risk factors and treatment for catheter‐related DVT, given its significant morbidity and mortality. Results have been equivocal, though, and in some instances, complicated by a diagnosis of underlying malignancy.[26, 27, 28]
As PICCs become more widely utilized, assessments of factors that place patients at greater risk of PICC‐related complications are needed.[21] The purpose of this study was to establish the incidence of complications associated with PICCs placed in the inpatient setting and examine risk factors predisposing patients to these complications.
MATERIALS AND METHODS
Study Design
A case control analysis of adult inpatients who underwent PICC placement between January 2009 and January 2010 was conducted at Scott & White Healthcare (now Baylor Scott & White Healthcare) to determine the incidence and risk factors for PICC‐associated complications.
Study Site
The study took place at Scott & White Memorial Hospital in Temple, Texas, a 636‐bed multispecialty teaching hospital and level 1 trauma center. It is part of a healthcare system that includes 12 hospitals and more than 60 regional clinics, all of which share an electronic medical record to enable full integration.
Human Subjects Approval
This study received approval from the institutional review board at Scott & White Healthcare.
PICC Placement Technique
Inpatient PICC placement was performed by the PICC consult service. The consult service was comprised of 3 separate provider teams: (1) internal medicine, including select hospitalists and internal medicine residents; (2) radiology, including interventional radiologists and radiology residents; and (3) nursing, including registered nurses with advanced training in PICC placement. Following placement of a consult, the PICC consult service assessed the patient, obtained consent, and subsequently placed the catheter. Members of the PICC consult service followed a system‐wide protocol wherein target veins were identified by ultrasound prior to attempting catheter placement, and actual placement of the PICC was ultrasound guided. Images obtained during the procedure were permanently documented in the medical record. At the time of this study, no formal protocol existed wherein target veins were mapped for caliber. Operators relied on their professional judgment to determine if vein caliber appeared sufficient to accommodate catheter placement.
All PICCs were placed using industry standard sterile precautions. A universally accepted modified Seldinger technique was used to obtain venous access.[29] A guidewire was then positioned in the desired vessel to facilitate proper venous placement of the catheter. During the course of the study period, catheters used were either single‐ (4 Fr) or double lumen (5 Fr).
Catheters were placed at the bedside by hospitalists or registered nurse teams; the location of the catheter tip at the cavoatrial junction was confirmed by chest radiography. Catheter insertions by radiologists were performed in the interventional radiology suite, and confirmation of location of the catheter tip was obtained with fluoroscopy.
PICC Maintenance
Following placement, nurses managed the PICC site according to nursing policy. Per policy, the site was assessed each shift. Documentation of assessment was recorded in nursing notes. Routine dressing changes were performed every 7 days, and as needed, to maintain a sterile site. Date and time of dressing changes were documented in nursing notes and on the PICC dressing. Catheter hubs and injection ports were disinfected with an antiseptic preparation for 15 seconds and allowed to air dry for 30 seconds prior to accessing the catheter. Catheters were flushed with 10 mL of normal saline before and after use. Any abnormality noted during PICC assessment was relayed to the primary provider. If the catheter did not flush readily or demonstrate appropriate blood return, nursing staff obtained an order for alteplase to be administered in an effort to salvage the line. PICCs were discontinued at the discretion of the healthcare provider.
Participants
Records of all patients 18 years of age and older who underwent PICC placement between January 2009 and January 2010 were reviewed (N=1444) for study inclusion. There were no exclusion criteria.
Data Collection
Patients who experienced complications were identified by electronic medical record review. One‐to‐one matching was performed for age and gender‐matched controls randomly selected from inpatients who underwent PICC placement during the same time period without complications. A total of 170 cases with PICC‐related complications were identified. One hundred seventy exact age‐ and gender‐matched controls, who based upon documentation available in the electronic medical record did not experience complications, were then randomly selected. Prior to data collection, the research team reviewed and discussed the data collection form and agreed upon a standardized protocol for data collection. Data collection was completed by authors J.M. and J.H. on the standardized data collection form. Although a formal analysis of inter‐rater agreement was not performed, J.M. and J.H. discussed any items where questions arose and arrived at a consensus decision regarding completion of the data point.
End points of the chart review were completion of medical therapy for which the PICC was indicated (eg, IV antibiotics or total parenteral nutrition [TPN]) or documentation of a complication that led to 1 of the following: discontinuation of the PICC or adjustment of either catheter placement or medical therapy. All complications were identified via International Classification of Diseases, 9th Revision codes and systematic chart review.
Complications resulting in discontinuation of the PICC, adjustment of catheter placement, or change in medical therapy were identified by review of nursing or physician documentation, and were categorized as follows: mechanical complications (defined as loss of the ability of the catheter to flush or draw properly, inadvertent catheter dislodgement, or retained portion of the catheter following catheter removal), catheter‐associated bloodstream infection (development of a positive blood culture attributable to the central catheter with no other clearly identifiable source of bacteremia present), cellulitis (defined as cellulitis in the extremity where the catheter was placed), bleeding from the site of catheter, fever (for which no other cause could be identified), and catheter‐associated thrombosis (identified by Doppler ultrasonography in patients exhibiting symptoms such as pain, swelling, redness, or warmth in the extremity in which the PICC was placed).[30]
Demographic data were collected, including insurance status, age, ethnicity, and gender. Clinical data included body mass index (BMI), presence of malnutrition (defined by a serum albumin of less than 3 g/dL),[31] previous or active cancer, previous DVT, use of anticoagulants (eg, warfarin, heparin, or low‐molecular‐weight heparin) or antiplatelet agent (eg, aspirin or clopidogrel) at the time of placement, and indication for PICC placement. A patient's history of previous or active cancer and previous DVT were identified by clinical documentation. Indications for PICC placement included: treating infectious processes (ie, infusion of antimicrobials), providing TPN, chemotherapy administration, and IV access. Catheter‐specific data were also collected and included venous access obtained (cephalic, basilic, brachial), catheter size (single lumen [4 Fr] or double lumen [5 Fr]), type of complication, and time to complication. The procedure note accompanying PICC placement was reviewed for data regarding time of day inserted (with after hours defined as documentation of placement occurring after 5 pm), and procedure operator to identify type of team (internal medicine, radiology, nursing) responsible for placement.
Data Analysis
Demographic characteristics and potential risk factors for patients in both the case and control groups of the study were summarized using descriptive statistics: mean ( standard deviation [SD]) for continuous variables and frequency (percent) for categorical variables. Univariate and multivariable conditional logistic regression analyses of variables that were potential risk factors of PICC‐related complications were utilized. A stepwise selection method was used for multivariable conditional logistic regression models. Alpha=0.2 was used for the significance to enter the model, and =0.05 was used for significance level to remain in the model. Attribution of PICC‐related complications was evaluated in terms of odds ratios (OR) and 95% confidence interval (CI). A P value of <0.05 indicated statistical significance. No prospective power analysis was performed. However, for a retrospective power analysis for 1:1 matching with 170 cases and 170 matched controls, assuming 20% of controls were affected and an of 0.05, one would achieve 80% power to detect an odds ratio of 2. SAS 9.2 (SAS Institute Inc., Cary, NC) was used for data analysis.
RESULTS
In 2009, 1444 PICCs were placed, and 170 cases in which patients experienced complications associated with PICC placement were identified, resulting in a complication rate of 11.77% (95% CI: 10.11%‐13.44%). The most common complications experienced by our patient population included catheter‐associated thrombosis (3%, n = 46), mechanical complications (4%, n=67), inadvertent catheter dislodgement (2%, n=36), mechanical dysfunction (2%, n=30), retained portion of the catheter following catheter removal (<1%, n=1), catheter‐associated bloodstream infections (2%, n=24), and cellulitis at the catheter insertion site (1%, n=15). Other documented complications included unexplained fever and bleeding (Table 1).
| Complication | N (%) |
|---|---|
| |
| Thrombosis | 46 (3) |
| Infection | 24 (2) |
| Cellulitis | 15 (1) |
| Mechanical complications* | 67 (4) |
| Unexplained fever | 15 (1) |
| Bleeding | 3 (0) |
| No complication | 1,274 (88) |
The mean age of the total cohort (N=340), comprised of case (N=170) and control (N=170) groups, was 58 years (SD 17), and 55% (n=94) were females. There were no significant differences in complications between groups based on ethnicity (P=0.66). In the case group, 46% (n=78) of PICCs were placed by the radiology team, 41% (n=69) were placed by the internal medicine team, and 14% (n=23) were placed by nursing. In the control group, 44% (n=74) of PICCs were placed by radiology, 36% (n=62) by internal medicine, and 20% (n=34) by nursing. Based on univariate conditional analysis, provider team was not significantly associated with complications (P=0.29).
Predictors of All‐Cause Complications
Based upon univariate conditional logistic regression analyses of complications related to PICC placement (N=340), the following variables demonstrated a statistically significant increased risk for complications: malnutrition (OR: 1.88 [95% CI: 1.023.44], P=0.04) and after‐hours placement (OR: 8.67 [95% CI: 2.62‐28.63], P=0.0004) (Table 2). Anticoagulation was associated with a decreased risk of complications (OR: 0.27 [95% CI: 0.16‐0.45], P=0.04). Based upon multivariable logistic regression analysis, after‐hours placement (OR: 9.52 [95% CI: 2.68‐33.78], P=0.0005) and BMI >30 (OR: 1.98 [95% CI: 1.09‐3.61], P=0.02) were significantly associated with an increased risk of PICC‐associated complications. Conversely, anticoagulation/antiplatelet use was associated with a decreased risk of complications (OR: 0.24 [95% CI: 0.14‐0.43], P<0.0001).
| Variable | Case, N (%) | Control, N (%) | Univariate | Multivariable | ||
|---|---|---|---|---|---|---|
| OR (95% CI) | P Value | AOR (95% CI) | P Value | |||
| ||||||
| Age, y, meanSD | 5817 | 5817 | ||||
| BMI, meanSD | 29.29.5 | 27.97.9 | 1.02 (0.991.05) | 0.12 | ||
| 30 | 108 (64) | 116 (68%) | 1.00 | 1.00 | ||
| >30 | 62 (36) | 54 (32%) | 1.29 (0.792.11) | 0.32 | 1.98 (1.093.61) | 0.02 |
| Length of stay, d, meanSD | 1822 | 1416 | 1.01 (1.001.03) | 0.06 | ||
| Length of stay group, d | 0.11a | |||||
| <7 | 41 (24) | 52 (31) | 1.00 | |||
| 729 | 101 (59) | 103 (61) | 1.19 (0.721.98) | 0.49 | ||
| 30 | 28 (16) | 15 (9) | 2.21 (1.074.58) | 0.03 | ||
| Gender | ||||||
| Female | 94 (55) | 94 (55) | ||||
| Male | 76 (45) | 76 (45) | ||||
| Ethnicity | 0.66a | |||||
| Caucasian | 131 (77) | 125 (74) | 1.00 | |||
| African American | 26 (15) | 28 (16) | 0.88 (0.481.60) | 0.67 | ||
| Hispanic/Asian | 13 (8) | 17 (10) | 0.70 (0.311.58) | 0.38 | ||
| Provider team | 0.29a | |||||
| Radiology | 78 (46) | 74 (44) | 1.00 | |||
| Internal medicine | 69 (41) | 62 (36) | 1.05 (0.681.64) | 0.82 | ||
| Nursing | 23 (14) | 34 (20) | 0.65 (0.351.19) | 0.16 | ||
| Insuranceb | 0.22a | |||||
| Private insurance | 46 (27) | 42 (25) | 1.00 | |||
| Uninsured | 17 (10) | 24 (14) | 0.73 (0.351.55) | 0.41 | ||
| Medicare | 57 (34) | 62 (37) | 0.73 (0.381.40) | 0.34 | ||
| Medicaid | 39 (23) | 25 (15) | 1.51 (0.743.06) | 0.26 | ||
| Tricare/Veterans Administration | 11 (6) | 16 (9) | 0.59 (0.241.45) | 0.25 | ||
| History of DVT | 27 (16) | 26 (15) | 1.05 (0.581.91) | 0.88 | ||
| Malnutritionb | 149 (88) | 134 (79) | 1.88 (1.023.44) | 0.04 | ||
| Cancer | 25 (15) | 36 (21) | 0.58 (0.311.09) | 0.09 | ||
| Fluoroscopy | 129 (76) | 139 (82) | 0.71 (0.421.19) | 0.19 | ||
| Anticoagulation use | 50 (29) | 100 (59) | 0.27 (0.160.45) | <0.0001 | 0.24 (0.140.43) | <0.0001 |
| Multilumenc | 99 (58) | 111 (66) | 0.70 (0.441.11) | 0.13 | ||
| Veinb | 0.39a | |||||
| Basilic | 98 (58) | 86 (51) | 1.00 | |||
| Cephalic | 11 (6) | 8 (5) | 1.37 (0.483.89) | 0.55 | ||
| Brachial | 61 (36) | 74 (44) | 0.70 (0.451.09) | 0.12 | ||
| Internal mammary | 0 (0) | 1 (1) | <0.001 (<0.001>999) | 0.99 | ||
| Time of dayb | ||||||
| Morning/afternoon | 144 (85) | 166 (98) | 1.00 | 1.00 | ||
| After hours | 26 (15) | 3 (2) | 8.67 (2.6228.63) | 0.0004 | 9.52 (2.6833.78) | 0.0005 |
| Indication for PICC | 0.02a | |||||
| Infection | 88 (52) | 71 (42) | 1.00 | |||
| Pneumonia | 21 (12) | 14 (8) | 1.07 (0.502.29) | 0.87 | ||
| Chemotherapy | 5 (3) | 2 (1) | 1.84 (0.349.93) | 0.48 | ||
| IV access | 36 (21) | 66 (39) | 0.44 (0.250.75) | 0.003 | ||
| Total parenteral nutrition | 20 (12) | 17 (10) | 0.96 (0.442.14) | 0.93 | ||
Predictors of Nonmechanical Complications
To study risk factors related to nonmechanical complications, a secondary analysis (N=206) was performed in which all patients who experienced mechanical complications (N=67) and matched controls (N=67) were excluded. Based upon multivariable logistic regression analysis, after‐hours placement (OR: 6.93 [95% CI: 1.35‐35.56], P=0.02) and malnutrition (OR: 2.83 [95% CI: 1.037.81], P=0.04) were significantly associated with increased risk of nonmechanical complications. The use of anticoagulation/antiplatelet agents was associated with decreased risk of nonmechanical complications (OR: 0.17 [95% CI: 0.07‐0.40], P<0.0001). Variables not significantly associated with nonmechanical complications included BMI>30, previous history of DVT, history of cancer, catheter size, and venous access choice (Table 3).
| Variable | Case, N (%) | Control, N (%) | Univariate | Multivariable | ||
|---|---|---|---|---|---|---|
| OR (95% CI) | P Value | AOR (95% CI) | P Value | |||
| ||||||
| Age, y, meanSD | 5816 | 5816 | ||||
| BMI, meanSD | 29.79.8 | 28.57.9 | 1.03 (0.991.07) | 0.22 | ||
| 30 | 64 (62) | 68 (66) | 1.00 | |||
| >30 | 39 (38) | 35 (34) | 1.27 (0.642.49) | 0.49 | ||
| Length of stay, d, meanSD | 2026 | 1418 | 1.02 (1.001.03) | 0.08 | ||
| Length of stay group, d | 0.03a | |||||
| <7 | 22 (21) | 28 (27) | 1.00 | |||
| 729 | 60 (58) | 68 (66) | 0.95 (0.491.82) | 0.87 | ||
| 30 | 21 (20) | 7 (7) | 3.24 (1.238.54) | 0.02 | ||
| Gender | ||||||
| Female | 63 (61) | 63 (61) | ||||
| Male | 40 (39) | 40 (39) | ||||
| Ethnicity | 0.95a | |||||
| Caucasian | 75 (73) | 75 (73) | 1.00 | |||
| African American | 19 (18) | 18 (17) | 1.06 (0.512.21) | 0.87 | ||
| Hispanic/Asian | 9 (9) | 10 (10) | 0.88 (0.322.44) | 0.81 | ||
| Provider team | 0.81a | |||||
| Radiology | 43 (42) | 44 (43) | 1.00 | |||
| Internal medicine | 45 (44) | 41 (40) | 1.11 (0.621.96) | 0.73 | ||
| Nursing | 15 (15) | 18 (17) | 0.86 (0.391.90) | 0.71 | ||
| Insuranceb | 0.22a | |||||
| Private insurance | 29 (28) | 27 (26) | 1.00 | |||
| Uninsured | 13 (13) | 12 (12) | 1.18 (0.433.26) | 0.74 | ||
| Medicare | 32 (31) | 40 (39) | 0.52 (0.211.29) | 0.16 | ||
| Medicaid | 21 (20) | 12 (12) | 1.81 (0.694.74) | 0.23 | ||
| Tricare/Veterans Administration | 8 (8) | 11 (11) | 0.58 (0.191.79) | 0.34 | ||
| History of DVT | 15 (15) | 15 (15) | 1.00 (0.462.16) | 1.00 | ||
| Malnutritionb | 93 (90) | 79 (77) | 2.86 (1.216.76) | 0.02 | 2.83 (1.037.81) | 0.04 |
| Cancer | 17 (17) | 22 (21) | 0.67 (0.301.48) | 0.32 | ||
| Fluoroscopy | 78 (76) | 85 (83) | 0.65 (0.321.31) | 0.23 | ||
| Anticoagulation use | 29 (28) | 60 (58) | 0.21 (0.100.44) | <0.0001 | 0.17 (0.070.40) | <0.0001 |
| Multilumenc | 64 (62) | 67 (66) | 0.83 (0.461.51) | 0.55 | ||
| Veinb | 0.32a | |||||
| Basilic | 54 (52) | 49 (48) | 1.00 | |||
| Cephalic | 8 (8) | 3 (3) | 2.45 (0.649.32) | 0.19 | ||
| Brachial | 41 (40) | 49 (48) | 0.72 (0.421.24) | 0.24 | ||
| Internal mammary | 0 (0) | 1 (1) | <0.001 (<0.001>999) | 0.99 | ||
| Time of dayb | ||||||
| Morning/afternoon | 87 (84) | 100 (98) | 1.00 | 1.00 | ||
| After hours | 16 (16) | 2 (2) | 8.00 (1.8434.79) | 0.006 | 6.93 (1.3535.56) | 0.02 |
| Indication for PICC | 0.13 | |||||
| Infectiond | 52 (50) | 45 (44) | 1.00 | |||
| Pneumonia | 14 (14) | 7 (7) | 1.46 (0.514.18) | 0.48 | ||
| Chemotherapy | 5 (5) | 0 (0) | >999 (<0.001>999) | 0.99 | ||
| IV access | 22 (21) | 43 (42) | 0.48 (0.240.96) | 0.04 | ||
| Total parenteral nutrition | 10 (10) | 8 (8) | 1.08 (0.323.62) | 0.90 | ||
Predictors of Thrombotic Complications
Of 1444 patients who underwent PICC placement, 3% (n=46) were subsequently diagnosed with a catheter‐associated thrombosis, representing 27% of all observed complications. In an attempt to better identify factors predisposing patients to thrombotic complications, an additional subgroup analysis (N=92) was performed on those patients who experienced catheter‐associated thrombosis (N=46) and matched controls (N=46). Variables examined in the analysis included BMI, length of stay (LOS), history of DVT, history of cancer, utilization of anticoagulation/antiplatelet agents, malnutrition, and catheter size.
Based on conditional univariate analyses, the following variables were significantly associated with increased risk of catheter‐associated thrombosis: LOS (as a continuous variable) (OR: 1.04 [95% CI: 1.001.09], P=0.05), malnutrition (OR: 4 [95% CI: 1.1314.18], P=0.03), and after‐hours placement (OR: 8.00 [95% CI: 1.0063.96], P=0.05) (Table 4). Use of anticoagulation/antiplatelet agents (OR: 0.29 [95% CI: 0.11‐0.80], P=0.02) was associated with decreased risk of thrombosis. History of previous DVT and history of cancer were nonsignificant. In the multivariable logistic regression model, malnutrition (OR: 10.16 [95% CI: 1.76‐58.71], P=0.01) remained associated with increased risk of catheter‐associated thrombosis, whereas use of anticoagulation/antiplatelet agents (OR: 0.11 [95% CI: 0.02‐0.51], P=0.005) was associated with decreased risk of catheter‐associated thrombosis (Table 4).
| Variable | Case, N (%) | Control, N (%) | Univariate | Multivariable | ||
|---|---|---|---|---|---|---|
| OR (95% CI) | P Value | AOR (95% CI) | P Value | |||
| ||||||
| Age, y, meanSD | 5818 | 5818 | ||||
| BMI, meanSD | 27.77.1 | 27.77.8 | 1.00 (0.931.08) | 0.98 | ||
| 30 | 34 (74) | 33 (72) | ||||
| >30 | 12 (26) | 13 (28) | 0.83 (0.252.73) | 0.76 | ||
| Length of stay, d, meanSD | 1712 | 119 | 1.04 (1.001.09) | 0.05 | ||
| Length of stay group, d | 0.15 | |||||
| <7 | 8 (17) | 14 (30) | 1.00 | |||
| 729 | 29 (63) | 30 (65) | 1.13 (0.413.07) | 0.82 | ||
| 30 | 9 (20) | 2 (4) | 4.65 (0.9822.13) | 0.05 | ||
| Gender | ||||||
| Female | 26 (57) | 26 (57) | ||||
| Male | 20 (43) | 20 (43) | ||||
| Ethnicity | 0.44a | |||||
| Caucasian | 31 (67) | 36 (78) | 1.00 | |||
| African American | 11 (24) | 6 (13) | 2.02 (0.695.93) | 0.20 | ||
| Hispanic/Asian | 4 (9) | 4 (9) | 1.12 (0.225.68) | 0.89 | ||
| Provider team | 0.26a | |||||
| Radiology | 23 (50) | 19 (41) | 1.00 | |||
| Internal medicine | 20 (43) | 18 (39) | 1.00 (0.432.31) | 1.00 | ||
| Nursing | 3(7) | 9 (20) | 0.33 (0.091.27) | 0.11 | ||
| Insuranceb | 0.38a | |||||
| Private insurance | 13 (28) | 11 (24) | 1.00 | |||
| Uninsured | 8 (17) | 4 (9) | 2.01 (0.3810.58) | 0.41 | ||
| Medicare | 14 (30) | 21 (47) | 0.39 (0.101.47) | 0.16 | ||
| Medicaid | 8 (17) | 7 (16) | 1.23 (0.285.36) | 0.78 | ||
| Tricare/Veterans Administration | 3 (7) | 2 (4) | 1.01 (0.128.27) | 1.00 | ||
| History of DVT | 7 (15) | 8 (17) | 0.88 (0.322.41) | 0.80 | ||
| Malnutritionb | 43 (93) | 33 (73) | 4.00 (1.1314.18) | 0.03 | 10.16 (1.7658.71) | 0.01 |
| Cancer | 10 (22) | 13 (28) | 0.67 (0.241.87) | 0.44 | ||
| Fluoroscopy | 33 (72) | 39 (85) | 0.46 (0.161.31) | 0.14 | ||
| Anticoagulation use | 16 (35) | 28 (61) | 0.29 (0.110.80) | 0.02 | 0.11 (0.020.51) | 0.005 |
| Multilumenc | 22 (48) | 28 (62) | 0.53 (0.231.26) | 0.15 | ||
| Veinb | 0.93a | |||||
| Basilic | 24 (52) | 21 (47) | 1.00 | |||
| Cephalic | 1 (2) | 1 (2) | 0.86 (0.0514.39) | 0.92 | ||
| Brachial | 21 (46) | 22 (49) | 0.75 (0.311.79) | 0.51 | ||
| Internal mammary | 0 (0) | 1 (2) | <0.001 (<0.001>999) | 0.99 | ||
| Time of dayb | ||||||
| Morning/afternoon | 38 (83) | 44 (98) | 1.00 | |||
| After hours | 8 (17) | 1 (2) | 8.00 (1.0063.96) | 0.05 | ||
| Indication for PICC | 0.80a | |||||
| Infectiond | 20 (43) | 17 (37) | 1.00 | |||
| Pneumonia | 5 (11) | 6 (13) | 0.60 (0.142.56) | 0.49 | ||
| Chemotherapy | 3 (7) | 0 (0) | >999 (<0.001>999) | 0.99 | ||
| IV access | 14 (30) | 20 (43) | 0.58 (0.231.44) | 0.24 | ||
| Total parenteral nutrition | 4 (9) | 3 (7) | 1.22 (0.197.70) | 0.83 | ||
DISCUSSION
The goal of this study was to identify factors related to PICC placement that place the general population of patients at risk. The type and rate of complications associated with PICCs in this study were similar to those previously reported in the literature including catheter‐related infection and thrombosis.[10, 32] Two unique risk factors, not well recognized previously,[10, 27, 28, 33] were observed in this study: malnutrition and after‐hours placement. Malnutrition, defined as serum albumin <3 g/dL was associated with an increase in PICC‐related complications (such as catheter‐associated bloodstream infections and cellulitis) and catheter‐related thrombosis. Malnutrition itself has long been associated with a decreased resistance to infection[34]; in addition, low serum albumin may also be a marker of the presence of other severe comorbidities, which may contribute to increased risk of thrombosis. It has been noted in previous studies that critical illness increases risk of thrombosis.[15] Despite an exhaustive search of the literature, we have been unable to find additional studies examining the extent to which malnutrition may impact PICC‐associated complications.
After‐hours placement was also associated with increased nonmechanical complications, as well as catheter‐related thrombosis. In an effort to improve both patient and consulting provider satisfaction and provide more expedient service, PICCs were often placed after hours (between 5 pm and 8 am) by both interventional radiology (n=14) and internal medicine (n=15) teams.
LOS has been associated with PICC placement complications in other studies.[12] In both primary and secondary analyses, hospital stays >30 days were associated with a higher risk of complications than hospitalizations <7 days. In light of the clinical significance of catheter‐related thrombosis, a subgroup analysis of patients with an LOS >30 days was conducted. The conditional univariate regression analysis showed an increased risk with greater LOS, malnutrition, and after‐hours placement. Use of anticoagulant or antiplatelet agents were associated with decreased risk of thrombosis (Table 4). The association between LOS and PICC‐related thrombosis is consistent with findings from Evans et al. involving 1728 patients in a similar center.[12] In these circumstances, increased LOS may be a surrogate marker for increased severity of illness, in that those patients who are more ill require lengthier hospitalizations. In a systematic review and meta‐analysis, Chopra et al. observed that increased severity of illness correlated with higher rates of catheter‐associated thrombosis, which is supportive of these findings.[15]
In the multivariate logistic regression analysis, BMI >30 was associated with a statistically significant increased risk for PICC‐associated complications after adjusting for anticoagulation and time of placement (Table 2). In the secondary analysis, where patients with mechanical complications were removed, BMI >30 was no longer associated with an increased risk for PICC‐associated complications (Table 3). This suggests that patients with a BMI >30 had an increased risk of mechanical complications, but were not necessarily at increased risk of developing other complications, such as catheter‐related thrombosis, infection, or bleeding. This finding is congruent with studies by Evans et al.,[12] who found no association between BMI and catheter‐associated thrombosis. Our association between BMI and complications is unique; to date, there are few additional studies that examine the extent to which BMI impacts the rate and type of complications associated with PICCs. At this time, the mechanism of the association between mechanical complications (such as inadvertent catheter removal or mechanical malfunction) and BMI is uncertain and warrants further investigation.
Use of Anticoagulant Agents
Anticoagulant (ie, any agent used for DVT prophylaxis or therapeutic anticoagulation) or antiplatelet agent use at the time of PICC placement and during the patient's hospitalization was associated with a decreased risk of thrombosis in our analysis. However, it should be noted that no specific anticoagulant agent was studied, and that antiplatelet agents were included in this analysis, unlike that of Evans et al.[12] Although current literature in oncologic populations, as well as the evidence‐based clinical practice guidelines, recommend against routine use of venous thromboprophylaxis in patients with central venous catheters,[33, 35, 36, 37] we believe this deserves further study, particularly in light of conflicting data in this area.[38, 39] Evans et al.[12] noted that although use of anticoagulants initially appeared to be associated with greater incidence of upper extremity venous thrombosis, when previous diagnosis of DVT was removed from the analysis the association was no longer significant.
In our analyses, no associations between catheter size, choice of venous access, history of previous deep venous thrombosis, or history of malignancy and risk for complications were found. Our findings differed from previous studies, where a relationship between increasing catheter bore size and site of access have been associated with increased PICC‐related thrombosis or other complications.[12, 20, 40, 41] There were also no significant differences in risk for complications between provider teams (eg, internal medicine, radiology, nursing) for PICCs placed during the morning or afternoon, which is consistent with findings by Funk et al.[1] Yet, after‐hours placement of PICCs was associated with greater complications than daytime placement. Although the exploration of factors associated with after‐hours placement was beyond the scope of this study, the findings from this study caused the authors, primarily comprised of members of the internal medicine inpatient medicine division, to reexamine the division's protocol on PICC placement. A consensus decision was made to discontinue after‐hours placement of PICCs by internal medicine teams in an effort to promote patient safety until further data could be collected. As a result, internal medicine teams no longer place PICCs after regular working hours at our institution.
Limitations
Limitations include the categorization of antiplatelet and anticoagulant agents together. We did not distinguish between high‐ and low‐dose aspirin, nor did we distinguish between therapeutic dosing of heparin and low‐molecular‐weight heparin versus DVT prophylaxis dosing. Additionally, for patients who were on warfarin or heparin drip, we did not evaluate for therapeutic range of international normalized ratio or partial thromboplastin time, as this was beyond the present scope of this study. In addition, malnutrition defined by albumin alone may have been somewhat narrow, as conditions aside from malnutrition can impact albumin levels. In future evaluations, this relationship may be clarified by including other determinants of clinical malnutrition including BMI <18 or the measurement of prealbumin. For determination of after‐hours placement of PICCs, we relied upon time of procedure dictation, assuming that all dictations immediately followed catheter placement. If there was a lapse in time between catheter placement and dictation, the category may have been recorded in error. Another limitation of after‐hours categorization was that we were unable to determine whether the PICC was placed on a weekend or holiday.
CONCLUSIONS AND FUTURE DIRECTIONS
Our results suggest that more stringent screening of patients undergoing PICC placement may reduce the risk of complications, with special attention to characteristics such as BMI >30, increased LOS, and protein‐calorie malnutrition (albumin <3). Furthermore, placement of PICC lines in emergent or after‐hours settings should be carefully considered and weighed against relative risks of central venous catheter placement. Further examination of the role anticoagulant and antiplatelet agents may have in the prevention of catheter‐related thrombosis should be undertaken. We hope that the identification of these risk factors will decrease the rate of complications and ultimately enhance patient safety and satisfaction.
Acknowledgments
The authors sincerely thank Glen Cryer, Publications Manager, Baylor Scott & White Health, for his assistance with this article.
Disclosures: Nothing to report.
- , , . Two‐year trends of peripherally inserted central catheter‐line complications at a tertiary‐care hospital: role of nursing expertise. Infect Control Hosp Epidemiol. 2001;22:377–379.
- , , . Peripherally inserted central catheters in the intensive care unit. J Intensive Care Med. 1996;11:49–54.
- , , , . Peripherally inserted central catheters in an acute‐care hospital. Arch Intern Med. 1994;154:1833–1837.
- , , , et al. Catheter‐associated bloodstream infection incidence and risk factors in adults with cancer: a prospective cohort study. J Hosp Infect. 2011;78:26–30.
- , . Peripherally inserted central catheters: a report of 2506 catheter days. JPEN J Parenter Enteral Nutr. 1995;19:133–136.
- , , , . Peripherally inserted central catheters in general medicine. Mayo Clin Proc. 1997;72:225–233.
- , , . Bedside placement of peripherally inserted central catheters: a cost‐effectiveness analysis. Radiology. 1998;206:423–428.
- , , , . Frequent asymptomatic pulmonary embolism in patients with deep venous thrombosis. JAMA. 1994;271:223–225.
- , . Experience with PICC at a university medical center. J Intraven Nurs. 1997;20:141–147.
- , , , et al. Upper‐extremity deep vein thrombosis. Risk factors, diagnosis, and complications. Arch Intern Med. 1997;157:57–62.
- , , , . Previous PICC placement may be associated with catheter‐related infections in hemodialysis patients. Cardiovasc Intervent Radiol. 2011;34:120–123.
- , , , et al. Risk of symptomatic DVT associated with peripherally inserted central catheters. Chest. 2010;138:803–810.
- . Be picky about PICCs. ACP Hospitalist, American College of Physicians website. Available at: http://www.acphospitalist.org/archives/2013/09/coverstory.htm. Accessed January 4, 2014.
- , , , , . The risk of bloodstream infection associated with peripherally inserted central catheters compared with central venous catheters in adults: a systematic review and meta‐analysis. Infect Control Hosp Epidemiol. 2013;34:908–918.
- , , , et al. Risk of venous thromboembolism associated with peripherally inserted central catheters: a systematic review and meta‐analysis. Lancet. 2013;382:311–325.
- , , , et al. Prevention of VTE in nonsurgical patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence‐based practice guidelines. Chest. 2012;141(2 suppl):e195S–e226S.
- , , . The clinical significance of peripherally inserted central venous catheter‐related deep vein thrombosis. Neurocrit Care. 2011;15:454–460.
- , , , , , . Peripherally inserted central catheter thrombosis‐reverse tapered versus nontapered catheters: a randomized controlled study. J Vasc Interv Radiol. 2014;25:85–91.
- , , , et al. Upper extremity venous thrombosis diagnosed by duplex scanning. Am J Surg. 1990;160:202–206.
- , , , et al. Reduction of peripherally inserted central catheter‐associated DVT. Chest. 2013;143:627–633.
- . Preventing DVT in peripherally inserted central catheters. Chest. 2013;143:589–590.
- , , , et al. Incidence of upper limb venous thrombosis associated with peripherally inserted central catheters (PICC). Br J Radiol. 2005;78:596–600.
- , , , , , . Computer identification of symptomatic deep venous thrombosis associated with peripherally inserted central catheters. AMIA Annu Symp Proc. 2007:226–230.
- , . Venous thrombosis related to peripherally inserted central catheters. J Vasc Interv Radiol. 2000;11:837–884.
- , , , , . To clot or not to clot? That is the question in central venous catheters. Clin Radiol. 2004;59:349–355.
- , , , et al. The Incidence of PICC line–associated thrombosis with and without the use of prophylactic anticoagulants. JPEN J Parenter Enteral Nutr. 2008;32:443–447.
- , , , , . Efficacy of multifaceted interventions in reducing complications of peripherally inserted central catheter in adult oncology patients. Support Care Cancer. 2010;18:1293–1298.
- , , , , . Peripherally inserted central venous catheter‐associated thrombosis: retrospective analysis of clinical risk factors in adult patients. South Med J. 2006;99:1073–1077.
- . The Seldinger method for PICC insertion. J Intraven Nurs. 1989;12:238–243.
- , , , et al. Venous thrombosis associated with the placement of peripherally inserted central catheters. J Vasc Interv Radiol. 2000;11:1309–1314.
- . Protein‐energy undernutrition. Merk Manual of Diagnosis and Therapy. 18th ed. Available at: http://www.merckmanuals.com/professional/nutritional_disorders/undernutrition/protein‐energy_undernutrition.html. Accessed May 26, 2013.
- , , , , . Risk of venous thromboembolism in hospitalized patients with peripherally inserted central catheters. J Hosp Med. 2009;4:417–422.
- , , , . Risk factors for upper extremity venous thrombosis associated with peripherally inserted central venous catheters. J Vasc Access. 2012;13:231–238.
- . Historical concepts of interactions, synergism and antagonism between nutrition and infection. J Nutr. 2003;133:316S–321S.
- , , , et al. 2008 SOR guidelines for the prevention and treatment of thrombosis associated with central venous catheters in patients with cancer: report from the working group. Ann Oncol. 2009;20:1459–1471.
- , , , , . Thromboprophylaxis in cancer patients with central venous catheters. A systematic review and meta‐analysis. Thromb Haemost. 2008;99:38–43.
- , , , . Thromboprophylaxis for catheter‐related thrombosis in patients with cancer: a systematic review of the randomized, controlled trials. J Thromb Haemost. 2007;5:2552–2554.
- , , , et al. Prevention of central venous catheter associated thrombosis using minidose warfarin in patients with haematological malignancies. Br J Haematol. 1998;10:483–486.
- , , , et al. Very low doses of warfarin can prevent thrombosis in central venous catheters. A randomized prospective trial. Ann Intern Med. 1990;112:423–428.
- , , , , , . The peripherally inserted central catheter (PICC): a prospective study of its natural history after cubital fossa insertion. Anesth Intensive Care. 2002;30:21–24.
- . Thrombotic complications in intravenous access. J Intraven Nurs. 1998;21:96–100.
- , , . Two‐year trends of peripherally inserted central catheter‐line complications at a tertiary‐care hospital: role of nursing expertise. Infect Control Hosp Epidemiol. 2001;22:377–379.
- , , . Peripherally inserted central catheters in the intensive care unit. J Intensive Care Med. 1996;11:49–54.
- , , , . Peripherally inserted central catheters in an acute‐care hospital. Arch Intern Med. 1994;154:1833–1837.
- , , , et al. Catheter‐associated bloodstream infection incidence and risk factors in adults with cancer: a prospective cohort study. J Hosp Infect. 2011;78:26–30.
- , . Peripherally inserted central catheters: a report of 2506 catheter days. JPEN J Parenter Enteral Nutr. 1995;19:133–136.
- , , , . Peripherally inserted central catheters in general medicine. Mayo Clin Proc. 1997;72:225–233.
- , , . Bedside placement of peripherally inserted central catheters: a cost‐effectiveness analysis. Radiology. 1998;206:423–428.
- , , , . Frequent asymptomatic pulmonary embolism in patients with deep venous thrombosis. JAMA. 1994;271:223–225.
- , . Experience with PICC at a university medical center. J Intraven Nurs. 1997;20:141–147.
- , , , et al. Upper‐extremity deep vein thrombosis. Risk factors, diagnosis, and complications. Arch Intern Med. 1997;157:57–62.
- , , , . Previous PICC placement may be associated with catheter‐related infections in hemodialysis patients. Cardiovasc Intervent Radiol. 2011;34:120–123.
- , , , et al. Risk of symptomatic DVT associated with peripherally inserted central catheters. Chest. 2010;138:803–810.
- . Be picky about PICCs. ACP Hospitalist, American College of Physicians website. Available at: http://www.acphospitalist.org/archives/2013/09/coverstory.htm. Accessed January 4, 2014.
- , , , , . The risk of bloodstream infection associated with peripherally inserted central catheters compared with central venous catheters in adults: a systematic review and meta‐analysis. Infect Control Hosp Epidemiol. 2013;34:908–918.
- , , , et al. Risk of venous thromboembolism associated with peripherally inserted central catheters: a systematic review and meta‐analysis. Lancet. 2013;382:311–325.
- , , , et al. Prevention of VTE in nonsurgical patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence‐based practice guidelines. Chest. 2012;141(2 suppl):e195S–e226S.
- , , . The clinical significance of peripherally inserted central venous catheter‐related deep vein thrombosis. Neurocrit Care. 2011;15:454–460.
- , , , , , . Peripherally inserted central catheter thrombosis‐reverse tapered versus nontapered catheters: a randomized controlled study. J Vasc Interv Radiol. 2014;25:85–91.
- , , , et al. Upper extremity venous thrombosis diagnosed by duplex scanning. Am J Surg. 1990;160:202–206.
- , , , et al. Reduction of peripherally inserted central catheter‐associated DVT. Chest. 2013;143:627–633.
- . Preventing DVT in peripherally inserted central catheters. Chest. 2013;143:589–590.
- , , , et al. Incidence of upper limb venous thrombosis associated with peripherally inserted central catheters (PICC). Br J Radiol. 2005;78:596–600.
- , , , , , . Computer identification of symptomatic deep venous thrombosis associated with peripherally inserted central catheters. AMIA Annu Symp Proc. 2007:226–230.
- , . Venous thrombosis related to peripherally inserted central catheters. J Vasc Interv Radiol. 2000;11:837–884.
- , , , , . To clot or not to clot? That is the question in central venous catheters. Clin Radiol. 2004;59:349–355.
- , , , et al. The Incidence of PICC line–associated thrombosis with and without the use of prophylactic anticoagulants. JPEN J Parenter Enteral Nutr. 2008;32:443–447.
- , , , , . Efficacy of multifaceted interventions in reducing complications of peripherally inserted central catheter in adult oncology patients. Support Care Cancer. 2010;18:1293–1298.
- , , , , . Peripherally inserted central venous catheter‐associated thrombosis: retrospective analysis of clinical risk factors in adult patients. South Med J. 2006;99:1073–1077.
- . The Seldinger method for PICC insertion. J Intraven Nurs. 1989;12:238–243.
- , , , et al. Venous thrombosis associated with the placement of peripherally inserted central catheters. J Vasc Interv Radiol. 2000;11:1309–1314.
- . Protein‐energy undernutrition. Merk Manual of Diagnosis and Therapy. 18th ed. Available at: http://www.merckmanuals.com/professional/nutritional_disorders/undernutrition/protein‐energy_undernutrition.html. Accessed May 26, 2013.
- , , , , . Risk of venous thromboembolism in hospitalized patients with peripherally inserted central catheters. J Hosp Med. 2009;4:417–422.
- , , , . Risk factors for upper extremity venous thrombosis associated with peripherally inserted central venous catheters. J Vasc Access. 2012;13:231–238.
- . Historical concepts of interactions, synergism and antagonism between nutrition and infection. J Nutr. 2003;133:316S–321S.
- , , , et al. 2008 SOR guidelines for the prevention and treatment of thrombosis associated with central venous catheters in patients with cancer: report from the working group. Ann Oncol. 2009;20:1459–1471.
- , , , , . Thromboprophylaxis in cancer patients with central venous catheters. A systematic review and meta‐analysis. Thromb Haemost. 2008;99:38–43.
- , , , . Thromboprophylaxis for catheter‐related thrombosis in patients with cancer: a systematic review of the randomized, controlled trials. J Thromb Haemost. 2007;5:2552–2554.
- , , , et al. Prevention of central venous catheter associated thrombosis using minidose warfarin in patients with haematological malignancies. Br J Haematol. 1998;10:483–486.
- , , , et al. Very low doses of warfarin can prevent thrombosis in central venous catheters. A randomized prospective trial. Ann Intern Med. 1990;112:423–428.
- , , , , , . The peripherally inserted central catheter (PICC): a prospective study of its natural history after cubital fossa insertion. Anesth Intensive Care. 2002;30:21–24.
- . Thrombotic complications in intravenous access. J Intraven Nurs. 1998;21:96–100.
© 2014 Society of Hospital Medicine
How to interpret surveys in medical research: A practical approach
Surveys are common in medical research. Although survey research may be subject to inherent self-report bias, surveys have a great impact on policies and practices in medicine, often forming the basis for recommendations or new guidelines.1,2 To interpret and use survey research results, clinicians should be familiar with key elements involved in the creation and validation of surveys.
The purpose of this article is to provide readers with a basic framework for evaluating surveys to allow them to be more informed as consumers of survey research.
IMPORTANT TOOLS IN MEDICAL RESEARCH
Surveys are important tools for answering questions on topics that are difficult to assess using other methods.3 They allow us to gather data systematically from subjects by asking questions, in order to make inferences about a larger population.3,4 Clinicians use surveys to explore the opinions, beliefs, and perceptions of a group, or to investigate physician practice patterns and adherence to clinical guidelines. They may also use surveys to better understand why patients are not engaging in recommended behavioral or lifestyle changes.
Survey methods include interviews (in person, by phone) and questionnaires (paper-and-pencil, e-mailed, online).4
A well-constructed, validated survey can provide powerful data that may influence clinical practice, guide future research development, or drive the development and provision of needed programs and services. Surveys have the potential to transform the ways in which we think about and practice medicine.
READER BEWARE
While survey research in health care appears to have grown exponentially, the quality of reported survey research has not necessarily increased over time.
For consumers of survey research, the adage “reader beware” is apt. Although a considerable number of studies have examined the effects of survey methodology on the validity, reliability, and generalizability of the results,4 medical journals differ in their requirements for reporting survey methods.
In an analysis of 117 articles, Bennett et al3 found that more than 80% did not fully describe the survey development process or pretesting methods. They also found limited guidance and lack of consensus about the best way to report survey research. Of 95 surveys requiring scoring, 66% did not report scoring practices.
Duffett et al5 noted that of 127 critical care medicine surveys, only 36% had been pretested or pilot-tested, and half of all surveys reviewed did not include participant demographics or included only minimal information.
Because journal reporting practices differ, physicians may be unaware of the steps involved in survey construction and validation. Knowledge of these steps is helpful not only in constructing surveys but also in assessing published articles that used survey research.
LIMITATIONS OF SURVEY RESEARCH
Indirect measures of attitudes and behaviors
Surveys that rely on participants’ self-reports of behaviors, attitudes, beliefs, or actions are indirect measures and are susceptible to self-report and social-desirability biases. Participants may overestimate their own expertise or knowledge in self-report surveys. They may wish to reduce embarrassment6 or answer in ways that would make them “look better,”7 resulting in social-desirability bias. These issues need to be mentioned in the limitations section in papers reporting survey research.
Questions and response choices
The data derived from surveys are only as good as the questions that are asked.8 Stone9 noted that questions should be intelligible, unambiguous, and unbiased. If respondents do not comprehend questions as researchers intended, if questionnaire response choices are inadequate, or if questions trigger unintended emotional responses,10–14 researchers may unwittingly introduce error, which will affect the validity of results. Even seemingly objective questions, such as those related to clinical algorithm use, practice patterns, or equipment available to hospital staff, may be interpreted differently by different respondents.
In their eagerness to launch a survey, clinician researchers may not realize that it must be carefully constructed. A focus on question development and validation is critical, as the questions determine the quality of the data derived from the survey.8 Even the position of the question or answer in the survey can affect how participants respond,15 as they may be guided to a response choice by preceding questions.16
WHAT DO YOU NEED TO KNOW ABOUT ASSESSING SURVEY RESEARCH?
What follows are questions and a basic framework that can be used to evaluate published survey research. Recommendations are based on the work of survey scientists,4,7,10,14,15,17,18 survey researchers in medicine and the social sciences, and national standards for test and questionnaire construction and validation (Table 1).4,19,20
Who created the survey? How did they do it?
How the survey was created should be sufficiently described to allow readers to judge the adequacy of instrument development.3–5 It is generally recommended that feedback from multiple sources be solicited during survey creation. Both questionnaire-design experts and subject-matter experts are considered critical in the process.4
What question was the survey designed to answer?
Is the objective of the study articulated in the paper? 3,20 To judge survey research, readers need to know if the survey appears to adequately address the research question or questions and the objectives of the study in terms of methods used.4
Was evidence on validity gathered?
Instrument pretesting and field testing are considered best practices by the American Association for Public Opinion Research, a professional organization for US survey scientists.4
Pretesting can include cognitive interviewing, the use of questionnaire appraisal tools, and hybrid methods, all of which are aimed at addressing validity issues.21 Pretesting with a group of participants similar to the target population allows for assessment of item ambiguity, instrument ease of use, adequacy of response categories (response choices), and time to completion.4,12
Cognitive interviewing is designed to explore respondents’ comprehension of questions, response processes, and decision processes governing how they answer questions.4,7,10,11 In cognitive interviewing, respondents are generally interviewed one on one. Techniques vary, but typically include “think alouds” (in which a respondent is asked to verbalize thoughts while responding to questions) and “verbal probing” (in which the respondent answers a question, then is asked follow-up questions as the interviewer probes for information related to the response choice or question itself).7 These techniques can provide evidence that researchers are actually measuring what they set out to measure and not an unrelated construct.4,19
Field testing of a survey under realistic conditions can help to uncover problems in administration, such as issues in standardization of key procedures, and to ensure that the survey was administered as the researchers intended.21,22 Field testing is vital before phone or in-person interviews to ensure standardization of any critical procedures. Pilot testing in a sample similar to the intended population allows for further refinement, with deletion of problem items, before the survey is launched.15
Because even “objective” questions can be somewhat subjective, all research surveys should go through some type of pretesting.4,21 Based on the results of pretesting and field testing, surveys should then be revised before launch.4,21 If an article on a self-report survey makes no mention of survey validation steps, readers may well question the validity of the results.
Are the survey questions and response choices understandable?
Is the meaning of each question unambiguous? Is the reading level appropriate for the sample population (a critical consideration in patient surveys)? Do any of the items actually ask two different questions?13 An example would be: “Was the representative courteous and prompt?” as it is possible to be courteous, but not prompt, and vice versa. If so, respondents may be confused or frustrated in attempting to answer it. If a rating scale is used throughout the questionnaire, are the anchors appropriate? For example, a question may be written in such a way that respondents want to answer “yes/no” or “agree/disagree,” but the scale used may include response options such as “poor,” “marginal,” “good,” and “excellent.” Items with Likert-response formats are commonly used in self-report surveys and allow participants to respond to a statement by choosing from a range of responses (eg, strongly disagree to strongly agree), often spaced horizontally under a line.
It is recommended that surveys also include options for answers beyond the response choices provided,20 such as comment boxes or fill-in-the-blank items. Surveys with a closed-response format may constrain the quality of data collected because investigators may not foresee all possible answers. Surveys need to be available for review either within the article itself, in an appendix, or as supplementary material that is available elsewhere.
Does the sample appear to be appropriate?
Articles that report the results of surveys should describe the target population, the sample design, and, in a demographic table, respondents and nonrespondents. To judge appropriateness, several questions can be asked regarding sampling:
Target population. Is the population of interest (ie, the target population) described, including regional demographics, if applicable? The relationship between the sample and the target population is important, as a nonrepresentative sample may result in misleading conclusions about the population of interest.
Sampling frame. Who had an opportunity to participate in the survey? At its simplest, the sampling frame establishes who (or what, in the case of institutions) should be included within the sample. This is typically a list of elements (Groves et al4) that acts to “frame” or define the sample to be selected. Where the target population may be all academic internal medicine physicians in the United States, the sampling frame may be all male and female US physicians who are members of particular internal medicine professional organizations, identified by their directory email addresses.
Sample design. How was the sample actually selected?4 For example, did investigators use a convenience sample of colleagues at other institutions or use a stratified random sample, ensuring adequate representation of respondents with certain characteristics?
Description of respondents. How is the sample of respondents described? Are demographic features reported, including statistics on regional or national representativeness?5 Does the sample of survey respondents appear to be representative of the researcher’s population of interest (ie, the target population)?3,23 If not, is this adequately described in the limitations section? Although outcomes will not be available on nonrespondents, demographic and baseline data often are available and should be reported. Are there systematic differences between respondents and nonrespondents?
Was the response rate adequate?
Was the response rate adequate, given the number of participants initially recruited? If the response rate was not adequate, did the researchers discuss this limitation?
Maximum response rate, defined as the total number of surveys returned divided by the total number of surveys sent,18 may be difficult to calculate with electronic or Web-based survey platforms. When the maximum response rate cannot be calculated, this issue needs to be addressed in the article’s limitations section.
The number of surveys has increased across fields over the past few decades, but survey response rates in general have decreased.17,21,24,25 In fields outside of clinical medicine, response rates in the 40% range are common.17 In the 1990s, the mean response rate for surveys published in medical journals (mailed surveys) was approximately 60%.26 A 2001 review of physician questionnaire studies found a similar average response rate (61%), with a 52% response rate for large-sample surveys.27 In 2002, Field et al28 examined the impact of incentives in physician survey studies and found response rates ranging from 8.5% to 80%.
Importantly, electronically delivered surveys (e-mail, Web-based) often have lower response rates than mailed surveys.24,29 Nominal financial incentives have been associated with enhanced response rates.28
A relatively low response rate does not necessarily mean you cannot trust the data. Survey scientists note that the representativeness of the sample may be more critical than response rate alone.17 Studies with small sample sizes may be more representative—and findings more valid—than those with large samples, if large samples are nonrepresentative when considering the target population.17
Do the conclusions go beyond the data?
Are the inferences overreaching, in view of the survey design? In studies with low response rates and nonrepresentative samples, researchers must be careful in interpreting the results. If the results cannot be generalized beyond the research sample, is this clear from the limitations, discussion, and conclusion sections?
In this review, we have summarized the findings of three published surveys1,2,30 and commented on how they appear to meet—or don’t quite meet—recommendations for survey development, validation, and use. The papers chosen were deemed strong examples in particular categories, such as description of survey authorship,1 instrument validation,30 sampling methodology,2 and response rate.1
It should be noted that even when surveys are conducted with the utmost rigor, survey reporting may leave out critical details. Survey methodology may not be adequately described for a variety of reasons, including researchers’ training in survey design and methodology; a lack of universally accepted journal-reporting guidelines3; and even journals’ space limitations. At times, journals may excise descriptions of survey development and validation, deeming these sections superfluous. Limitations sections can be critical to interpreting the results of survey research and evaluating the scope of conclusions.
- Jha AK, DesRoches CM, Campbell EG, et al. Use of electronic health records in US hospitals. N Engl J Med 2009; 360:1628–1638.
- Angus DC, Shorr AF, White A, Dremsizov TT, Schmitz RJ, Kelley MA; Committee on Manpower for Pulmonary and Critical Care Societies (COMPACCS). Critical care delivery in the United States: distribution of services and compliance with Leapfrog recommendations. Crit Care Med 2006; 34:1016–1024.
- Bennett C, Khangura S, Brehaut JC, et al. Reporting guidelines for survey research: an analysis of published guidance and reporting practices. PLoS Med 2010; 8:e1001069.
- Groves RM, Fowler FJ, Couper MP, Lepkowski JM, Singer E, Tourangeau R. Survey Methodology. 2nd ed. Hoboken, NJ: John Wiley and Sons, Inc; 2009.
- Duffett M, Burns KE, Adhikari NK, et al. Quality of reporting of surveys in critical care journals: a methodologic review. Crit Care Med 2012; 40:441–449.
- Mattell MS, Jacoby J. Is there an optimal number of alternatives for Likert-scale items? Effects of testing time and scale properties. J Appl Psychol 1972; 56:506–509.
- Willis GB. Cognitive Interviewing. A “How To” Guide. Research Triangle Institute. Presented at the meeting of the American Statistical Association; 1999. http://fog.its.uiowa.edu/~c07b209/interview.pdf. Accessed June 3, 2013.
- Schwarz N. Self-reports. How the questions shape the answers. Amer Psychol 1999; 54:93–105.
- Stone DH. Design a questionnaire. BMJ 1993; 307:1264–1266.
- Willis GB, Royston P, Bercini D. The use of verbal report methods in the development and testing of survey questionnaires. Appl Cogn Psychol 1991; 5:251–267.
- Desimone LM, LeFloch KC. Are we asking the right questions? Using cognitive interviews to improve surveys in education research. Educ Eval Policy Anal 2004; 26:1–22.
- Presser S, Couper MP, Lessler JT, et al. Methods for testing and evaluating survey questions. Public Opin Q 2004; 68:109–130.
- Rogers G. Accreditation Board for Engineering and Technology (ABET), Inc. Sample Protocol for Pilot Testing Survey Items. www.abet.org/WorkArea/DownloadAsset.aspx?id=1299. Accessed January 22, 2013.
- Schwarz N, Oyserman D. Asking questions about behavior: cognition, communication, and questionnaire construction. Am J Eval 2001; 22:127–160.
- Bradburn N, Sudman S, Wansink B. Asking Questions. The Definitive Guide to Questionnaire Design—For Market Research, Political Polls, and Social and Health Questionnaires. San Francisco, CA: Jossey-Bass; 2004.
- Stone AA, Broderick JE, Schwartz JE, Schwarz N. Context effects in survey ratings of health, symptoms, and satisfaction. Med Care 2008; 46:662–667.
- Cook C, Heath F, Thompson RL. A meta-analysis of response rates in Web or internet-based surveys. Educ Psychol Meas 2000; 60:821–836.
- Kaplowitz MD, Hadlock TD, Levine R. A comparison of Web and mail survey response rates. Public Opin Q 2004; 68:94–101.
- American Educational Research Association. Standards for Educational and Psychological Testing/American Educational Research Association, American Psychological Association, National Council on Measurement in Education. Washington, DC: American Educational Research Association; 1999.
- Burns KE, Duffett M, Kho ME, et al; ACCADEMY Group. A guide for the design and conduct of self-administered surveys of clinicians. CMAJ 2008; 179:245–252.
- American Association for Public Opinion Research (AAPOR). http://www.aapor.org/Home.htm. Accessed June 3, 2013.
- National Center for Education Statistics. Planning and Design of Surveys. http://nces.ed.gov/statprog/2002/std2_1.asp. Accessed January 22, 2013.
- Bordens KS, Abbott BB. Research Design and Methods. A Process Approach. 6th ed. New York, NY: McGraw-Hill; 2004.
- Sheehan K. Email survey response rates: a review. JCMC 2001. http://jcmc.indiana.edu/vol6/issue2/sheehan.html. Accessed January 22, 2013.
- Baruch Y, Holtom BC. Survey response rate levels and trends in organizational research. Hum Relat 2008; 61:1139–1160.
- Asch DA, Jedrziewski MK, Christakis NA. Response rates to mail surveys published in medical journals. J Clin Epidemiol 1997; 50:1129–1136.
- Cummings SM, Savitz LA, Konrad TR. Reported response rates to mailed physician questionnaires. Health Services Res 2001; 35:1347–1355.
- Field TS, Cadoret CA, Brown ML, et al. Surveying physicians. Do components of the “Total Design Approach” to optimizing survey response rates apply to physicians? Med Care 2002; 40:596–606.
- Converse PD, Wolfe EW, Huang X, Oswald FL. Response rates for mixed-mode surveys using mail and e-mail/Web. Am J Eval 2008; 29:99–107.
- Hirshberg E, Lacroix J, Sward K, Willson D, Morris AH. Blood glucose control in critically ill adults and children: a survey on stated practice. Chest 2008; 133:1328–1335.
Surveys are common in medical research. Although survey research may be subject to inherent self-report bias, surveys have a great impact on policies and practices in medicine, often forming the basis for recommendations or new guidelines.1,2 To interpret and use survey research results, clinicians should be familiar with key elements involved in the creation and validation of surveys.
The purpose of this article is to provide readers with a basic framework for evaluating surveys to allow them to be more informed as consumers of survey research.
IMPORTANT TOOLS IN MEDICAL RESEARCH
Surveys are important tools for answering questions on topics that are difficult to assess using other methods.3 They allow us to gather data systematically from subjects by asking questions, in order to make inferences about a larger population.3,4 Clinicians use surveys to explore the opinions, beliefs, and perceptions of a group, or to investigate physician practice patterns and adherence to clinical guidelines. They may also use surveys to better understand why patients are not engaging in recommended behavioral or lifestyle changes.
Survey methods include interviews (in person, by phone) and questionnaires (paper-and-pencil, e-mailed, online).4
A well-constructed, validated survey can provide powerful data that may influence clinical practice, guide future research development, or drive the development and provision of needed programs and services. Surveys have the potential to transform the ways in which we think about and practice medicine.
READER BEWARE
While survey research in health care appears to have grown exponentially, the quality of reported survey research has not necessarily increased over time.
For consumers of survey research, the adage “reader beware” is apt. Although a considerable number of studies have examined the effects of survey methodology on the validity, reliability, and generalizability of the results,4 medical journals differ in their requirements for reporting survey methods.
In an analysis of 117 articles, Bennett et al3 found that more than 80% did not fully describe the survey development process or pretesting methods. They also found limited guidance and lack of consensus about the best way to report survey research. Of 95 surveys requiring scoring, 66% did not report scoring practices.
Duffett et al5 noted that of 127 critical care medicine surveys, only 36% had been pretested or pilot-tested, and half of all surveys reviewed did not include participant demographics or included only minimal information.
Because journal reporting practices differ, physicians may be unaware of the steps involved in survey construction and validation. Knowledge of these steps is helpful not only in constructing surveys but also in assessing published articles that used survey research.
LIMITATIONS OF SURVEY RESEARCH
Indirect measures of attitudes and behaviors
Surveys that rely on participants’ self-reports of behaviors, attitudes, beliefs, or actions are indirect measures and are susceptible to self-report and social-desirability biases. Participants may overestimate their own expertise or knowledge in self-report surveys. They may wish to reduce embarrassment6 or answer in ways that would make them “look better,”7 resulting in social-desirability bias. These issues need to be mentioned in the limitations section in papers reporting survey research.
Questions and response choices
The data derived from surveys are only as good as the questions that are asked.8 Stone9 noted that questions should be intelligible, unambiguous, and unbiased. If respondents do not comprehend questions as researchers intended, if questionnaire response choices are inadequate, or if questions trigger unintended emotional responses,10–14 researchers may unwittingly introduce error, which will affect the validity of results. Even seemingly objective questions, such as those related to clinical algorithm use, practice patterns, or equipment available to hospital staff, may be interpreted differently by different respondents.
In their eagerness to launch a survey, clinician researchers may not realize that it must be carefully constructed. A focus on question development and validation is critical, as the questions determine the quality of the data derived from the survey.8 Even the position of the question or answer in the survey can affect how participants respond,15 as they may be guided to a response choice by preceding questions.16
WHAT DO YOU NEED TO KNOW ABOUT ASSESSING SURVEY RESEARCH?
What follows are questions and a basic framework that can be used to evaluate published survey research. Recommendations are based on the work of survey scientists,4,7,10,14,15,17,18 survey researchers in medicine and the social sciences, and national standards for test and questionnaire construction and validation (Table 1).4,19,20
Who created the survey? How did they do it?
How the survey was created should be sufficiently described to allow readers to judge the adequacy of instrument development.3–5 It is generally recommended that feedback from multiple sources be solicited during survey creation. Both questionnaire-design experts and subject-matter experts are considered critical in the process.4
What question was the survey designed to answer?
Is the objective of the study articulated in the paper? 3,20 To judge survey research, readers need to know if the survey appears to adequately address the research question or questions and the objectives of the study in terms of methods used.4
Was evidence on validity gathered?
Instrument pretesting and field testing are considered best practices by the American Association for Public Opinion Research, a professional organization for US survey scientists.4
Pretesting can include cognitive interviewing, the use of questionnaire appraisal tools, and hybrid methods, all of which are aimed at addressing validity issues.21 Pretesting with a group of participants similar to the target population allows for assessment of item ambiguity, instrument ease of use, adequacy of response categories (response choices), and time to completion.4,12
Cognitive interviewing is designed to explore respondents’ comprehension of questions, response processes, and decision processes governing how they answer questions.4,7,10,11 In cognitive interviewing, respondents are generally interviewed one on one. Techniques vary, but typically include “think alouds” (in which a respondent is asked to verbalize thoughts while responding to questions) and “verbal probing” (in which the respondent answers a question, then is asked follow-up questions as the interviewer probes for information related to the response choice or question itself).7 These techniques can provide evidence that researchers are actually measuring what they set out to measure and not an unrelated construct.4,19
Field testing of a survey under realistic conditions can help to uncover problems in administration, such as issues in standardization of key procedures, and to ensure that the survey was administered as the researchers intended.21,22 Field testing is vital before phone or in-person interviews to ensure standardization of any critical procedures. Pilot testing in a sample similar to the intended population allows for further refinement, with deletion of problem items, before the survey is launched.15
Because even “objective” questions can be somewhat subjective, all research surveys should go through some type of pretesting.4,21 Based on the results of pretesting and field testing, surveys should then be revised before launch.4,21 If an article on a self-report survey makes no mention of survey validation steps, readers may well question the validity of the results.
Are the survey questions and response choices understandable?
Is the meaning of each question unambiguous? Is the reading level appropriate for the sample population (a critical consideration in patient surveys)? Do any of the items actually ask two different questions?13 An example would be: “Was the representative courteous and prompt?” as it is possible to be courteous, but not prompt, and vice versa. If so, respondents may be confused or frustrated in attempting to answer it. If a rating scale is used throughout the questionnaire, are the anchors appropriate? For example, a question may be written in such a way that respondents want to answer “yes/no” or “agree/disagree,” but the scale used may include response options such as “poor,” “marginal,” “good,” and “excellent.” Items with Likert-response formats are commonly used in self-report surveys and allow participants to respond to a statement by choosing from a range of responses (eg, strongly disagree to strongly agree), often spaced horizontally under a line.
It is recommended that surveys also include options for answers beyond the response choices provided,20 such as comment boxes or fill-in-the-blank items. Surveys with a closed-response format may constrain the quality of data collected because investigators may not foresee all possible answers. Surveys need to be available for review either within the article itself, in an appendix, or as supplementary material that is available elsewhere.
Does the sample appear to be appropriate?
Articles that report the results of surveys should describe the target population, the sample design, and, in a demographic table, respondents and nonrespondents. To judge appropriateness, several questions can be asked regarding sampling:
Target population. Is the population of interest (ie, the target population) described, including regional demographics, if applicable? The relationship between the sample and the target population is important, as a nonrepresentative sample may result in misleading conclusions about the population of interest.
Sampling frame. Who had an opportunity to participate in the survey? At its simplest, the sampling frame establishes who (or what, in the case of institutions) should be included within the sample. This is typically a list of elements (Groves et al4) that acts to “frame” or define the sample to be selected. Where the target population may be all academic internal medicine physicians in the United States, the sampling frame may be all male and female US physicians who are members of particular internal medicine professional organizations, identified by their directory email addresses.
Sample design. How was the sample actually selected?4 For example, did investigators use a convenience sample of colleagues at other institutions or use a stratified random sample, ensuring adequate representation of respondents with certain characteristics?
Description of respondents. How is the sample of respondents described? Are demographic features reported, including statistics on regional or national representativeness?5 Does the sample of survey respondents appear to be representative of the researcher’s population of interest (ie, the target population)?3,23 If not, is this adequately described in the limitations section? Although outcomes will not be available on nonrespondents, demographic and baseline data often are available and should be reported. Are there systematic differences between respondents and nonrespondents?
Was the response rate adequate?
Was the response rate adequate, given the number of participants initially recruited? If the response rate was not adequate, did the researchers discuss this limitation?
Maximum response rate, defined as the total number of surveys returned divided by the total number of surveys sent,18 may be difficult to calculate with electronic or Web-based survey platforms. When the maximum response rate cannot be calculated, this issue needs to be addressed in the article’s limitations section.
The number of surveys has increased across fields over the past few decades, but survey response rates in general have decreased.17,21,24,25 In fields outside of clinical medicine, response rates in the 40% range are common.17 In the 1990s, the mean response rate for surveys published in medical journals (mailed surveys) was approximately 60%.26 A 2001 review of physician questionnaire studies found a similar average response rate (61%), with a 52% response rate for large-sample surveys.27 In 2002, Field et al28 examined the impact of incentives in physician survey studies and found response rates ranging from 8.5% to 80%.
Importantly, electronically delivered surveys (e-mail, Web-based) often have lower response rates than mailed surveys.24,29 Nominal financial incentives have been associated with enhanced response rates.28
A relatively low response rate does not necessarily mean you cannot trust the data. Survey scientists note that the representativeness of the sample may be more critical than response rate alone.17 Studies with small sample sizes may be more representative—and findings more valid—than those with large samples, if large samples are nonrepresentative when considering the target population.17
Do the conclusions go beyond the data?
Are the inferences overreaching, in view of the survey design? In studies with low response rates and nonrepresentative samples, researchers must be careful in interpreting the results. If the results cannot be generalized beyond the research sample, is this clear from the limitations, discussion, and conclusion sections?
In this review, we have summarized the findings of three published surveys1,2,30 and commented on how they appear to meet—or don’t quite meet—recommendations for survey development, validation, and use. The papers chosen were deemed strong examples in particular categories, such as description of survey authorship,1 instrument validation,30 sampling methodology,2 and response rate.1
It should be noted that even when surveys are conducted with the utmost rigor, survey reporting may leave out critical details. Survey methodology may not be adequately described for a variety of reasons, including researchers’ training in survey design and methodology; a lack of universally accepted journal-reporting guidelines3; and even journals’ space limitations. At times, journals may excise descriptions of survey development and validation, deeming these sections superfluous. Limitations sections can be critical to interpreting the results of survey research and evaluating the scope of conclusions.
Surveys are common in medical research. Although survey research may be subject to inherent self-report bias, surveys have a great impact on policies and practices in medicine, often forming the basis for recommendations or new guidelines.1,2 To interpret and use survey research results, clinicians should be familiar with key elements involved in the creation and validation of surveys.
The purpose of this article is to provide readers with a basic framework for evaluating surveys to allow them to be more informed as consumers of survey research.
IMPORTANT TOOLS IN MEDICAL RESEARCH
Surveys are important tools for answering questions on topics that are difficult to assess using other methods.3 They allow us to gather data systematically from subjects by asking questions, in order to make inferences about a larger population.3,4 Clinicians use surveys to explore the opinions, beliefs, and perceptions of a group, or to investigate physician practice patterns and adherence to clinical guidelines. They may also use surveys to better understand why patients are not engaging in recommended behavioral or lifestyle changes.
Survey methods include interviews (in person, by phone) and questionnaires (paper-and-pencil, e-mailed, online).4
A well-constructed, validated survey can provide powerful data that may influence clinical practice, guide future research development, or drive the development and provision of needed programs and services. Surveys have the potential to transform the ways in which we think about and practice medicine.
READER BEWARE
While survey research in health care appears to have grown exponentially, the quality of reported survey research has not necessarily increased over time.
For consumers of survey research, the adage “reader beware” is apt. Although a considerable number of studies have examined the effects of survey methodology on the validity, reliability, and generalizability of the results,4 medical journals differ in their requirements for reporting survey methods.
In an analysis of 117 articles, Bennett et al3 found that more than 80% did not fully describe the survey development process or pretesting methods. They also found limited guidance and lack of consensus about the best way to report survey research. Of 95 surveys requiring scoring, 66% did not report scoring practices.
Duffett et al5 noted that of 127 critical care medicine surveys, only 36% had been pretested or pilot-tested, and half of all surveys reviewed did not include participant demographics or included only minimal information.
Because journal reporting practices differ, physicians may be unaware of the steps involved in survey construction and validation. Knowledge of these steps is helpful not only in constructing surveys but also in assessing published articles that used survey research.
LIMITATIONS OF SURVEY RESEARCH
Indirect measures of attitudes and behaviors
Surveys that rely on participants’ self-reports of behaviors, attitudes, beliefs, or actions are indirect measures and are susceptible to self-report and social-desirability biases. Participants may overestimate their own expertise or knowledge in self-report surveys. They may wish to reduce embarrassment6 or answer in ways that would make them “look better,”7 resulting in social-desirability bias. These issues need to be mentioned in the limitations section in papers reporting survey research.
Questions and response choices
The data derived from surveys are only as good as the questions that are asked.8 Stone9 noted that questions should be intelligible, unambiguous, and unbiased. If respondents do not comprehend questions as researchers intended, if questionnaire response choices are inadequate, or if questions trigger unintended emotional responses,10–14 researchers may unwittingly introduce error, which will affect the validity of results. Even seemingly objective questions, such as those related to clinical algorithm use, practice patterns, or equipment available to hospital staff, may be interpreted differently by different respondents.
In their eagerness to launch a survey, clinician researchers may not realize that it must be carefully constructed. A focus on question development and validation is critical, as the questions determine the quality of the data derived from the survey.8 Even the position of the question or answer in the survey can affect how participants respond,15 as they may be guided to a response choice by preceding questions.16
WHAT DO YOU NEED TO KNOW ABOUT ASSESSING SURVEY RESEARCH?
What follows are questions and a basic framework that can be used to evaluate published survey research. Recommendations are based on the work of survey scientists,4,7,10,14,15,17,18 survey researchers in medicine and the social sciences, and national standards for test and questionnaire construction and validation (Table 1).4,19,20
Who created the survey? How did they do it?
How the survey was created should be sufficiently described to allow readers to judge the adequacy of instrument development.3–5 It is generally recommended that feedback from multiple sources be solicited during survey creation. Both questionnaire-design experts and subject-matter experts are considered critical in the process.4
What question was the survey designed to answer?
Is the objective of the study articulated in the paper? 3,20 To judge survey research, readers need to know if the survey appears to adequately address the research question or questions and the objectives of the study in terms of methods used.4
Was evidence on validity gathered?
Instrument pretesting and field testing are considered best practices by the American Association for Public Opinion Research, a professional organization for US survey scientists.4
Pretesting can include cognitive interviewing, the use of questionnaire appraisal tools, and hybrid methods, all of which are aimed at addressing validity issues.21 Pretesting with a group of participants similar to the target population allows for assessment of item ambiguity, instrument ease of use, adequacy of response categories (response choices), and time to completion.4,12
Cognitive interviewing is designed to explore respondents’ comprehension of questions, response processes, and decision processes governing how they answer questions.4,7,10,11 In cognitive interviewing, respondents are generally interviewed one on one. Techniques vary, but typically include “think alouds” (in which a respondent is asked to verbalize thoughts while responding to questions) and “verbal probing” (in which the respondent answers a question, then is asked follow-up questions as the interviewer probes for information related to the response choice or question itself).7 These techniques can provide evidence that researchers are actually measuring what they set out to measure and not an unrelated construct.4,19
Field testing of a survey under realistic conditions can help to uncover problems in administration, such as issues in standardization of key procedures, and to ensure that the survey was administered as the researchers intended.21,22 Field testing is vital before phone or in-person interviews to ensure standardization of any critical procedures. Pilot testing in a sample similar to the intended population allows for further refinement, with deletion of problem items, before the survey is launched.15
Because even “objective” questions can be somewhat subjective, all research surveys should go through some type of pretesting.4,21 Based on the results of pretesting and field testing, surveys should then be revised before launch.4,21 If an article on a self-report survey makes no mention of survey validation steps, readers may well question the validity of the results.
Are the survey questions and response choices understandable?
Is the meaning of each question unambiguous? Is the reading level appropriate for the sample population (a critical consideration in patient surveys)? Do any of the items actually ask two different questions?13 An example would be: “Was the representative courteous and prompt?” as it is possible to be courteous, but not prompt, and vice versa. If so, respondents may be confused or frustrated in attempting to answer it. If a rating scale is used throughout the questionnaire, are the anchors appropriate? For example, a question may be written in such a way that respondents want to answer “yes/no” or “agree/disagree,” but the scale used may include response options such as “poor,” “marginal,” “good,” and “excellent.” Items with Likert-response formats are commonly used in self-report surveys and allow participants to respond to a statement by choosing from a range of responses (eg, strongly disagree to strongly agree), often spaced horizontally under a line.
It is recommended that surveys also include options for answers beyond the response choices provided,20 such as comment boxes or fill-in-the-blank items. Surveys with a closed-response format may constrain the quality of data collected because investigators may not foresee all possible answers. Surveys need to be available for review either within the article itself, in an appendix, or as supplementary material that is available elsewhere.
Does the sample appear to be appropriate?
Articles that report the results of surveys should describe the target population, the sample design, and, in a demographic table, respondents and nonrespondents. To judge appropriateness, several questions can be asked regarding sampling:
Target population. Is the population of interest (ie, the target population) described, including regional demographics, if applicable? The relationship between the sample and the target population is important, as a nonrepresentative sample may result in misleading conclusions about the population of interest.
Sampling frame. Who had an opportunity to participate in the survey? At its simplest, the sampling frame establishes who (or what, in the case of institutions) should be included within the sample. This is typically a list of elements (Groves et al4) that acts to “frame” or define the sample to be selected. Where the target population may be all academic internal medicine physicians in the United States, the sampling frame may be all male and female US physicians who are members of particular internal medicine professional organizations, identified by their directory email addresses.
Sample design. How was the sample actually selected?4 For example, did investigators use a convenience sample of colleagues at other institutions or use a stratified random sample, ensuring adequate representation of respondents with certain characteristics?
Description of respondents. How is the sample of respondents described? Are demographic features reported, including statistics on regional or national representativeness?5 Does the sample of survey respondents appear to be representative of the researcher’s population of interest (ie, the target population)?3,23 If not, is this adequately described in the limitations section? Although outcomes will not be available on nonrespondents, demographic and baseline data often are available and should be reported. Are there systematic differences between respondents and nonrespondents?
Was the response rate adequate?
Was the response rate adequate, given the number of participants initially recruited? If the response rate was not adequate, did the researchers discuss this limitation?
Maximum response rate, defined as the total number of surveys returned divided by the total number of surveys sent,18 may be difficult to calculate with electronic or Web-based survey platforms. When the maximum response rate cannot be calculated, this issue needs to be addressed in the article’s limitations section.
The number of surveys has increased across fields over the past few decades, but survey response rates in general have decreased.17,21,24,25 In fields outside of clinical medicine, response rates in the 40% range are common.17 In the 1990s, the mean response rate for surveys published in medical journals (mailed surveys) was approximately 60%.26 A 2001 review of physician questionnaire studies found a similar average response rate (61%), with a 52% response rate for large-sample surveys.27 In 2002, Field et al28 examined the impact of incentives in physician survey studies and found response rates ranging from 8.5% to 80%.
Importantly, electronically delivered surveys (e-mail, Web-based) often have lower response rates than mailed surveys.24,29 Nominal financial incentives have been associated with enhanced response rates.28
A relatively low response rate does not necessarily mean you cannot trust the data. Survey scientists note that the representativeness of the sample may be more critical than response rate alone.17 Studies with small sample sizes may be more representative—and findings more valid—than those with large samples, if large samples are nonrepresentative when considering the target population.17
Do the conclusions go beyond the data?
Are the inferences overreaching, in view of the survey design? In studies with low response rates and nonrepresentative samples, researchers must be careful in interpreting the results. If the results cannot be generalized beyond the research sample, is this clear from the limitations, discussion, and conclusion sections?
In this review, we have summarized the findings of three published surveys1,2,30 and commented on how they appear to meet—or don’t quite meet—recommendations for survey development, validation, and use. The papers chosen were deemed strong examples in particular categories, such as description of survey authorship,1 instrument validation,30 sampling methodology,2 and response rate.1
It should be noted that even when surveys are conducted with the utmost rigor, survey reporting may leave out critical details. Survey methodology may not be adequately described for a variety of reasons, including researchers’ training in survey design and methodology; a lack of universally accepted journal-reporting guidelines3; and even journals’ space limitations. At times, journals may excise descriptions of survey development and validation, deeming these sections superfluous. Limitations sections can be critical to interpreting the results of survey research and evaluating the scope of conclusions.
- Jha AK, DesRoches CM, Campbell EG, et al. Use of electronic health records in US hospitals. N Engl J Med 2009; 360:1628–1638.
- Angus DC, Shorr AF, White A, Dremsizov TT, Schmitz RJ, Kelley MA; Committee on Manpower for Pulmonary and Critical Care Societies (COMPACCS). Critical care delivery in the United States: distribution of services and compliance with Leapfrog recommendations. Crit Care Med 2006; 34:1016–1024.
- Bennett C, Khangura S, Brehaut JC, et al. Reporting guidelines for survey research: an analysis of published guidance and reporting practices. PLoS Med 2010; 8:e1001069.
- Groves RM, Fowler FJ, Couper MP, Lepkowski JM, Singer E, Tourangeau R. Survey Methodology. 2nd ed. Hoboken, NJ: John Wiley and Sons, Inc; 2009.
- Duffett M, Burns KE, Adhikari NK, et al. Quality of reporting of surveys in critical care journals: a methodologic review. Crit Care Med 2012; 40:441–449.
- Mattell MS, Jacoby J. Is there an optimal number of alternatives for Likert-scale items? Effects of testing time and scale properties. J Appl Psychol 1972; 56:506–509.
- Willis GB. Cognitive Interviewing. A “How To” Guide. Research Triangle Institute. Presented at the meeting of the American Statistical Association; 1999. http://fog.its.uiowa.edu/~c07b209/interview.pdf. Accessed June 3, 2013.
- Schwarz N. Self-reports. How the questions shape the answers. Amer Psychol 1999; 54:93–105.
- Stone DH. Design a questionnaire. BMJ 1993; 307:1264–1266.
- Willis GB, Royston P, Bercini D. The use of verbal report methods in the development and testing of survey questionnaires. Appl Cogn Psychol 1991; 5:251–267.
- Desimone LM, LeFloch KC. Are we asking the right questions? Using cognitive interviews to improve surveys in education research. Educ Eval Policy Anal 2004; 26:1–22.
- Presser S, Couper MP, Lessler JT, et al. Methods for testing and evaluating survey questions. Public Opin Q 2004; 68:109–130.
- Rogers G. Accreditation Board for Engineering and Technology (ABET), Inc. Sample Protocol for Pilot Testing Survey Items. www.abet.org/WorkArea/DownloadAsset.aspx?id=1299. Accessed January 22, 2013.
- Schwarz N, Oyserman D. Asking questions about behavior: cognition, communication, and questionnaire construction. Am J Eval 2001; 22:127–160.
- Bradburn N, Sudman S, Wansink B. Asking Questions. The Definitive Guide to Questionnaire Design—For Market Research, Political Polls, and Social and Health Questionnaires. San Francisco, CA: Jossey-Bass; 2004.
- Stone AA, Broderick JE, Schwartz JE, Schwarz N. Context effects in survey ratings of health, symptoms, and satisfaction. Med Care 2008; 46:662–667.
- Cook C, Heath F, Thompson RL. A meta-analysis of response rates in Web or internet-based surveys. Educ Psychol Meas 2000; 60:821–836.
- Kaplowitz MD, Hadlock TD, Levine R. A comparison of Web and mail survey response rates. Public Opin Q 2004; 68:94–101.
- American Educational Research Association. Standards for Educational and Psychological Testing/American Educational Research Association, American Psychological Association, National Council on Measurement in Education. Washington, DC: American Educational Research Association; 1999.
- Burns KE, Duffett M, Kho ME, et al; ACCADEMY Group. A guide for the design and conduct of self-administered surveys of clinicians. CMAJ 2008; 179:245–252.
- American Association for Public Opinion Research (AAPOR). http://www.aapor.org/Home.htm. Accessed June 3, 2013.
- National Center for Education Statistics. Planning and Design of Surveys. http://nces.ed.gov/statprog/2002/std2_1.asp. Accessed January 22, 2013.
- Bordens KS, Abbott BB. Research Design and Methods. A Process Approach. 6th ed. New York, NY: McGraw-Hill; 2004.
- Sheehan K. Email survey response rates: a review. JCMC 2001. http://jcmc.indiana.edu/vol6/issue2/sheehan.html. Accessed January 22, 2013.
- Baruch Y, Holtom BC. Survey response rate levels and trends in organizational research. Hum Relat 2008; 61:1139–1160.
- Asch DA, Jedrziewski MK, Christakis NA. Response rates to mail surveys published in medical journals. J Clin Epidemiol 1997; 50:1129–1136.
- Cummings SM, Savitz LA, Konrad TR. Reported response rates to mailed physician questionnaires. Health Services Res 2001; 35:1347–1355.
- Field TS, Cadoret CA, Brown ML, et al. Surveying physicians. Do components of the “Total Design Approach” to optimizing survey response rates apply to physicians? Med Care 2002; 40:596–606.
- Converse PD, Wolfe EW, Huang X, Oswald FL. Response rates for mixed-mode surveys using mail and e-mail/Web. Am J Eval 2008; 29:99–107.
- Hirshberg E, Lacroix J, Sward K, Willson D, Morris AH. Blood glucose control in critically ill adults and children: a survey on stated practice. Chest 2008; 133:1328–1335.
- Jha AK, DesRoches CM, Campbell EG, et al. Use of electronic health records in US hospitals. N Engl J Med 2009; 360:1628–1638.
- Angus DC, Shorr AF, White A, Dremsizov TT, Schmitz RJ, Kelley MA; Committee on Manpower for Pulmonary and Critical Care Societies (COMPACCS). Critical care delivery in the United States: distribution of services and compliance with Leapfrog recommendations. Crit Care Med 2006; 34:1016–1024.
- Bennett C, Khangura S, Brehaut JC, et al. Reporting guidelines for survey research: an analysis of published guidance and reporting practices. PLoS Med 2010; 8:e1001069.
- Groves RM, Fowler FJ, Couper MP, Lepkowski JM, Singer E, Tourangeau R. Survey Methodology. 2nd ed. Hoboken, NJ: John Wiley and Sons, Inc; 2009.
- Duffett M, Burns KE, Adhikari NK, et al. Quality of reporting of surveys in critical care journals: a methodologic review. Crit Care Med 2012; 40:441–449.
- Mattell MS, Jacoby J. Is there an optimal number of alternatives for Likert-scale items? Effects of testing time and scale properties. J Appl Psychol 1972; 56:506–509.
- Willis GB. Cognitive Interviewing. A “How To” Guide. Research Triangle Institute. Presented at the meeting of the American Statistical Association; 1999. http://fog.its.uiowa.edu/~c07b209/interview.pdf. Accessed June 3, 2013.
- Schwarz N. Self-reports. How the questions shape the answers. Amer Psychol 1999; 54:93–105.
- Stone DH. Design a questionnaire. BMJ 1993; 307:1264–1266.
- Willis GB, Royston P, Bercini D. The use of verbal report methods in the development and testing of survey questionnaires. Appl Cogn Psychol 1991; 5:251–267.
- Desimone LM, LeFloch KC. Are we asking the right questions? Using cognitive interviews to improve surveys in education research. Educ Eval Policy Anal 2004; 26:1–22.
- Presser S, Couper MP, Lessler JT, et al. Methods for testing and evaluating survey questions. Public Opin Q 2004; 68:109–130.
- Rogers G. Accreditation Board for Engineering and Technology (ABET), Inc. Sample Protocol for Pilot Testing Survey Items. www.abet.org/WorkArea/DownloadAsset.aspx?id=1299. Accessed January 22, 2013.
- Schwarz N, Oyserman D. Asking questions about behavior: cognition, communication, and questionnaire construction. Am J Eval 2001; 22:127–160.
- Bradburn N, Sudman S, Wansink B. Asking Questions. The Definitive Guide to Questionnaire Design—For Market Research, Political Polls, and Social and Health Questionnaires. San Francisco, CA: Jossey-Bass; 2004.
- Stone AA, Broderick JE, Schwartz JE, Schwarz N. Context effects in survey ratings of health, symptoms, and satisfaction. Med Care 2008; 46:662–667.
- Cook C, Heath F, Thompson RL. A meta-analysis of response rates in Web or internet-based surveys. Educ Psychol Meas 2000; 60:821–836.
- Kaplowitz MD, Hadlock TD, Levine R. A comparison of Web and mail survey response rates. Public Opin Q 2004; 68:94–101.
- American Educational Research Association. Standards for Educational and Psychological Testing/American Educational Research Association, American Psychological Association, National Council on Measurement in Education. Washington, DC: American Educational Research Association; 1999.
- Burns KE, Duffett M, Kho ME, et al; ACCADEMY Group. A guide for the design and conduct of self-administered surveys of clinicians. CMAJ 2008; 179:245–252.
- American Association for Public Opinion Research (AAPOR). http://www.aapor.org/Home.htm. Accessed June 3, 2013.
- National Center for Education Statistics. Planning and Design of Surveys. http://nces.ed.gov/statprog/2002/std2_1.asp. Accessed January 22, 2013.
- Bordens KS, Abbott BB. Research Design and Methods. A Process Approach. 6th ed. New York, NY: McGraw-Hill; 2004.
- Sheehan K. Email survey response rates: a review. JCMC 2001. http://jcmc.indiana.edu/vol6/issue2/sheehan.html. Accessed January 22, 2013.
- Baruch Y, Holtom BC. Survey response rate levels and trends in organizational research. Hum Relat 2008; 61:1139–1160.
- Asch DA, Jedrziewski MK, Christakis NA. Response rates to mail surveys published in medical journals. J Clin Epidemiol 1997; 50:1129–1136.
- Cummings SM, Savitz LA, Konrad TR. Reported response rates to mailed physician questionnaires. Health Services Res 2001; 35:1347–1355.
- Field TS, Cadoret CA, Brown ML, et al. Surveying physicians. Do components of the “Total Design Approach” to optimizing survey response rates apply to physicians? Med Care 2002; 40:596–606.
- Converse PD, Wolfe EW, Huang X, Oswald FL. Response rates for mixed-mode surveys using mail and e-mail/Web. Am J Eval 2008; 29:99–107.
- Hirshberg E, Lacroix J, Sward K, Willson D, Morris AH. Blood glucose control in critically ill adults and children: a survey on stated practice. Chest 2008; 133:1328–1335.
KEY POINTS
- Most survey reports do not adequately describe their methods.
- Surveys that rely on participants’ self-reports of behaviors, attitudes, beliefs, or actions are indirect measures and are susceptible to self-report and social-desirability biases.
- Informed readers need to consider a survey’s authorship, objective, validation, items, response choices, sampling representativeness, response rate, generalizability, and scope of the conclusions.