User login
Medical Communities Go Virtual
Throughout history, physicians have formed communities to aid in the dissemination of knowledge, skills, and professional norms. From local physician groups to international societies and conferences, this drive to connect with members of our profession across the globe is timeless. We do so to learn from each other and continue to move the field of medicine forward.
Yet, these communities are being strained by necessary physical distancing required during the COVID-19 pandemic. Many physicians accustomed to a sense of community are now finding themselves surprisingly isolated and alone. Into this distanced landscape, however, new digital groups—specifically social media (SoMe), online learning communities, and virtual conferences—have emerged. We are all active members in virtual communities; all of the authors are team members of The Clinical Problem Solvers podcast and one author of this paper, A.P., has previously served as the medical education lead for the Human Diagnosis Project. Both entities are described later in this article. Here, we provide an overview of these virtual communities and discuss how they have the potential to more equitably and effectively disseminate medical knowledge and education both during and after the COVID-19 pandemic (Table).
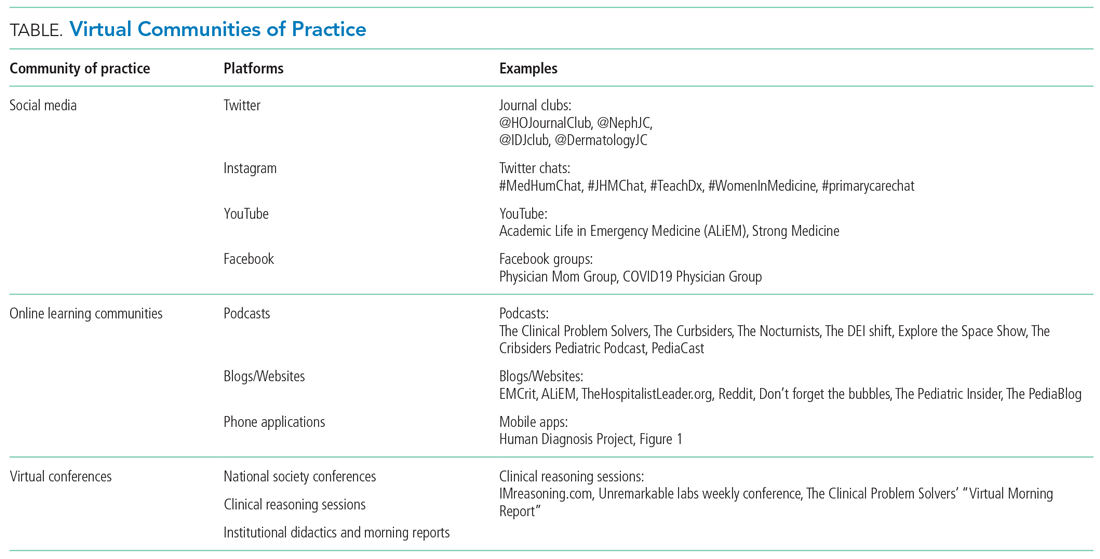
SOCIAL MEDIA
Even prior to the COVID-19 pandemic, SoMe—especially Twitter—had become a virtual gathering place where digital colleagues exchange Twitter handles like business cards.1,2 They celebrate each other’s achievements and provide support during difficult times.
Importantly, the format of Twitter tends toward a flattened hierarchy. It is this egalitarian nature that has served SoMe well in its position as a modern learning community. Users from across the experience spectrum engage with and create novel educational content. This often occurs in the form of Tweetorials, or short lessons conveyed over a series of linked tweets. These have gained immense popularity on the platform and are becoming increasingly recognized forms of scholarship.3 Further, case-based lessons have become ubiquitous and are valuable opportunities for users to learn from other members of their digital communities. During the current pandemic, SoMe has become extremely important in the early dissemination and critique of the slew of research on the COVID-19 crisis.4
Beyond its role as an educational platform, SoMe functions as a virtual gathering place for members of the medical community to discuss topics relevant to the field. Subspecialists and researchers have gathered in digital journal clubs (eg, #NephJC, #IDJClub, #BloodandBone) and a number of journals have hosted live Twitter chats covering topics like controversies in clinical practice or professional development (eg, #JHMChat). More recently, social issues affecting the medical field, such as gender equity and the growing antiracism movement, have led to robust discussion on this medium.
Beyond Twitter, many medical professionals gather and exchange ideas on other platforms. Virtual networking and educational groups have arisen using Slack and Facebook.5-7 Trainees and faculty members alike consume and produce content on YouTube, which often serve to teach technical skills.8 Given widespread use of SoMe, we anticipate that the range of platforms utilized by medical professionals will continue to expand in the future.
ONLINE LEARNING COMMUNITIES
There have long existed multiple print and online forums dedicated to the development of clinical skills. These include clinical challenges in medical journals, interactive online cases, and more formal diagnostic education curricula at academic centers.9-11 With the COVID-19 pandemic, it has become more difficult to ensure that trainees have an in-person learning community to discuss and receive feedback. This has led to a wider adoption of application-based clinical exercises, educational podcasts, and curricular innovations to support these virtual efforts.
The Human Diagnosis Project (Human Dx) is a smart-phone application that provides a platform for individuals to submit clinical cases that can be rapidly peer-reviewed and disseminated to the larger user pool. Human Dx is notable for fostering a strong sense of community amongst its users.12,13 Case consumers and case creators are able to engage in further discussion after solving a case, and opportunities for feedback and growth are ample.
Medical education podcasts have taken on greater importance during the pandemic.14,15 Many educators have begun referring their learners towards certain podcasts as in-person learning communities have been put on hold. Medical professionals may appreciate the up-to-date and candid conversations held on many podcasts, which can provide both educationally useful and emotionally sympathetic connections to their distanced peers. Similarly, while academic clinicians previously benefitted from invited grand rounds speakers, they may now find that such expert discussants are most easily accessible through their appearances on podcasts.
As institutions suspended clerkships during the pandemic, many created virtual communities for trainees to engage in diagnostic reasoning and education. They built novel curricula that meld asynchronous learning with online community-based learning.14 Gamified learning tools and quizzes have also been incorporated into these hybrid curricula to help ensure participation of learners within their virtual communities.16,17
VIRTUAL CONFERENCES
Perhaps the most notable advance in digital communities catalyzed by the COVID-19 pandemic has been the increasing reliance on and comfort with video-based software. While many of our clinical, administrative, and social activities have migrated toward these virtual environments, they have also been used for a variety of activities related to education and professional development.
As institutions struggled to adapt to physical distancing, many medical schools and residency programs have moved their regular meetings and conferences to virtual platforms. Similar free and open-access conferences have also emerged, including the “Virtual Morning Report” (VMR) series from The Clinical Problem Solvers podcast, wherein a few individuals are invited to discuss a case on the video conference, with the remainder of the audience contributing via the chat feature.
Beyond the growing popularity of video conferencing for education, these virtual sessions have become their own community. On The Clinical Problem Solvers VMR, many participants, ranging from preclinical students to seasoned attendings, show up on a daily basis and interact with each other as close friends, as do members of more insular institutional sessions (eg, residency run reports). In these strangely isolating times, many of us have experienced comfort in seeing the faces of our friends and colleagues joining us to listen and discuss cases.
Separately, many professional societies have struggled with how to replace their large yearly in-person conferences, which would pose substantial infectious risks were they to be held in person. While many of those scheduled to occur during the early days of the pandemic were canceled or held limited online sessions, the trend towards virtual conference platforms seems to be accelerating. Organizers of the 2020 Conference on Retroviruses and Opportunistic Infections (March 8-11, 2020) decided to convert from an in-person to entirely virtual conference 48 hours before it started. With the benefit of more forewarning, other conferences are planning and exploring best practices to promote networking and advancement of research goals at future academic meetings.18,19
BENEFITS OF VIRTUAL COMMUNITIES
The growing importance of these new digital communities could be viewed as a necessary evolution in the way that we gather and learn from each other. Traditional physician communities were inherently restricted by location, specialty, and hierarchy, thereby limiting the dissemination of knowledge and changes to professional norms. These restrictions could conceivably insulate and promote elite institutions in a fashion that perpetuates the inequalities within global medical systems. Unrestricted and open-access virtual communities, in contrast, have the potential to remove historical barriers and connect first-class mentors with trainees they would never have met otherwise.
Beyond promoting a more equitable distribution of knowledge and resources, these virtual communities are well suited to harness the benefits of group learning. The concept of communities of practice (CoP) refers to groupings of individuals involved in a personal or professional endeavor, with the community facilitating advancement of their own knowledge and skill set. Members of the CoP learn from each other, with more established members passing down essential knowledge and cultural norms. The three main components of CoP are maintaining a social network, a mutual enterprise (eg, a common goal), and a shared repertoire (eg, experiences, languages, etc).
Designing virtual learning spaces with these aspects in mind may allow these communities to function as CoPs. Some strategies include use of chat functions in videoconferences (to promote further dialogue) and development of dedicated sessions for specific subgroups or aims (eg, professional mentorship). The anticipated benefits of integrating virtual CoPs into medical education are notable, as a number of studies have already suggested that they are effective for disseminating knowledge, enhancing social learning, and aiding with professional development.7,20-23 These virtual CoPs continue to evolve, however, and further research is warranted to clarify how best to utilize them in medical education and professional societies.
CONCLUSION
Amidst the tragic loss of lives and financial calamity, the COVID-19 pandemic has also spurred innovation and change in the way health professionals learn and communicate. Going forward, the medical establishment should capitalize on these recent innovations and work to further build, recognize, and foster such digital gathering spaces in order to more equitably and effectively disseminate knowledge and educational resources.
Despite physical distancing, health professionals have grown closer during these past few months. Innovations spurred by the pandemic have made us stronger and more united. Our experience with social media, online learning communities, and virtual conferences suggests the opportunity to grow and evolve from this experience. As Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said in March 2020, “...life is not going to be how it used to be [after the pandemic]…” Let’s hope he’s right.
ACKNOWLEDGMENTS
We thank Reza Manesh, MD, Rabih Geha, MD, and Jack Penner, MD, for their careful review of the manuscript.
1. Markham MJ, Gentile D, Graham DL. Social media for networking, professional development, and patient engagement. Am Soc Clin Oncol Educ Book. 2017;37:782-787. https://doi.org/10.1200/EDBK_180077
2. Melvin L, Chan T. Using Twitter in clinical education and practice. J Grad Med Educ. 2014;6(3):581-582. https://doi.org/10.4300/JGME-D-14-00342.1
3. Breu AC. Why is a cow? Curiosity, Tweetorials, and the return to why. N Engl J Med. 2019;381(12):1097-1098. https://doi.org/10.1056/NEJMp1906790
4. Chan AKM, Nickson CP, Rudolph JW, Lee A, Joynt GM. Social media for rapid knowledge dissemination: early experience from the COVID-19 pandemic. Anaesthesia. 2020:10.1111/anae.15057. https://doi.org/10.1111/anae.15057
5. Pander T, Pinilla S, Dimitriadis K, Fischer MR. The use of Facebook in medical education--a literature review. GMS Z Med Ausbild. 2014;31(3):Doc33. https://doi.org/10.3205/zma000925
6. Cree-Green M, Carreau AM, Davis SM, et al. Peer mentoring for professional and personal growth in academic medicine. J Investig Med. 2020;68(6):1128-1134. https://doi.org/10.1136/jim-2020-001391
7. Yarris LM, Chan TM, Gottlieb M, Juve AM. Finding your people in the digital age: virtual communities of practice to promote education scholarship. J Grad Med Educ. 2019;11(1):1-5. https://doi.org/10.4300/JGME-D-18-01093.1
8. Sterling M, Leung P, Wright D, Bishop TF. The use of social media in graduate medical education: a systematic review. Acad Med. 2017;92(7):1043-1056. https://doi.org/10.1097/ACM.0000000000001617
9. Manesh R, Dhaliwal G. Digital tools to enhance clinical reasoning. Med Clin North Am. 2018;102(3):559-565. https://doi.org/10.1016/j.mcna.2017.12.015
10. Subramanian A, Connor DM, Berger G, et al. A curriculum for diagnostic reasoning: JGIM’s exercises in clinical reasoning. J Gen Intern Med. 2019;34(3):344-345. https://doi.org/10.1007/s11606-018-4689-y
11. Olson APJ, Singhal G, Dhaliwal G. Diagnosis education - an emerging field. Diagnosis (Berl). 2019;6(2):75-77. https://doi.org/10.1515/dx-2019-0029
12. Chatterjee S, Desai S, Manesh R, Sun J, Nundy S, Wright SM. Assessment of a simulated case-based measurement of physician diagnostic performance. JAMA Netw Open. 2019;2(1):e187006. https://doi.org/10.1001/jamanetworkopen.2018.7006
13. Russell SW, Desai SV, O’Rourke P, et al. The genealogy of teaching clinical reasoning and diagnostic skill: the GEL Study. Diagnosis (Berl). 2020;7(3):197-203. https://doi.org/10.1515/dx-2019-0107
14. Geha R, Dhaliwal G. Pilot virtual clerkship curriculum during the COVID-19 pandemic: podcasts, peers, and problem-solving. Med Educ. 2020;54(9):855-856. https://doi.org/10.1111/medu.14246
15. AlGaeed M, Grewal M, Richardson PK, Leon Guerrero CR. COVID-19: Neurology residents’ perspective. J Clin Neurosci. 2020;78:452-453. https://doi.org/10.1016/j.jocn.2020.05.032
16. Moro C, Stromberga Z. Enhancing variety through gamified, interactive learning experiences. Med Educ. 2020. Online ahead of print. https://doi.org/10.1111/medu.14251
17. Morawo A, Sun C, Lowden M. Enhancing engagement during live virtual learning using interactive quizzes. Med Educ. 2020. Online ahead of print. https://doi.org/10.1111/medu.14253
18. Rubinger L, Gazendam A, Ekhtiari S, et al. Maximizing virtual meetings and conferences: a review of best practices. Int Orthop. 2020;44(8):1461-1466. https://doi.org/10.1007/s00264-020-04615-9
19. Woolston C. Learning to love virtual conferences in the coronavirus era. Nature. 2020;582(7810):135-136. https://doi.org/10.1038/d41586-020-01489-0
20. Cruess RL, Cruess SR, Steinert Y. Medicine as a community of practice: implications for medical education. Acad Med. 2018;93(2):185-191. https://doi.org/10.1097/ACM.0000000000001826
21. McLoughlin C, Patel KD, O’Callaghan T, Reeves S. The use of virtual communities of practice to improve interprofessional collaboration and education: findings from an integrated review. J Interprof Care. 2018;32(2):136-142. https://doi.org/10.1080/13561820.2017.1377692
22. Barnett S, Jones SC, Caton T, Iverson D, Bennett S, Robinson L. Implementing a virtual community of practice for family physician training: a mixed-methods case study. J Med Internet Res. 2014;16(3):e83. https://doi.org/10.2196/jmir.3083
23. Healy MG, Traeger LN, Axelsson CGS, et al. NEJM Knowledge+ Question of the Week: a novel virtual learning community effectively utilizing an online discussion forum. Med Teach. 2019;41(11):1270-1276. https://doi.org/10.1080/0142159X.2019.1635685
Throughout history, physicians have formed communities to aid in the dissemination of knowledge, skills, and professional norms. From local physician groups to international societies and conferences, this drive to connect with members of our profession across the globe is timeless. We do so to learn from each other and continue to move the field of medicine forward.
Yet, these communities are being strained by necessary physical distancing required during the COVID-19 pandemic. Many physicians accustomed to a sense of community are now finding themselves surprisingly isolated and alone. Into this distanced landscape, however, new digital groups—specifically social media (SoMe), online learning communities, and virtual conferences—have emerged. We are all active members in virtual communities; all of the authors are team members of The Clinical Problem Solvers podcast and one author of this paper, A.P., has previously served as the medical education lead for the Human Diagnosis Project. Both entities are described later in this article. Here, we provide an overview of these virtual communities and discuss how they have the potential to more equitably and effectively disseminate medical knowledge and education both during and after the COVID-19 pandemic (Table).

SOCIAL MEDIA
Even prior to the COVID-19 pandemic, SoMe—especially Twitter—had become a virtual gathering place where digital colleagues exchange Twitter handles like business cards.1,2 They celebrate each other’s achievements and provide support during difficult times.
Importantly, the format of Twitter tends toward a flattened hierarchy. It is this egalitarian nature that has served SoMe well in its position as a modern learning community. Users from across the experience spectrum engage with and create novel educational content. This often occurs in the form of Tweetorials, or short lessons conveyed over a series of linked tweets. These have gained immense popularity on the platform and are becoming increasingly recognized forms of scholarship.3 Further, case-based lessons have become ubiquitous and are valuable opportunities for users to learn from other members of their digital communities. During the current pandemic, SoMe has become extremely important in the early dissemination and critique of the slew of research on the COVID-19 crisis.4
Beyond its role as an educational platform, SoMe functions as a virtual gathering place for members of the medical community to discuss topics relevant to the field. Subspecialists and researchers have gathered in digital journal clubs (eg, #NephJC, #IDJClub, #BloodandBone) and a number of journals have hosted live Twitter chats covering topics like controversies in clinical practice or professional development (eg, #JHMChat). More recently, social issues affecting the medical field, such as gender equity and the growing antiracism movement, have led to robust discussion on this medium.
Beyond Twitter, many medical professionals gather and exchange ideas on other platforms. Virtual networking and educational groups have arisen using Slack and Facebook.5-7 Trainees and faculty members alike consume and produce content on YouTube, which often serve to teach technical skills.8 Given widespread use of SoMe, we anticipate that the range of platforms utilized by medical professionals will continue to expand in the future.
ONLINE LEARNING COMMUNITIES
There have long existed multiple print and online forums dedicated to the development of clinical skills. These include clinical challenges in medical journals, interactive online cases, and more formal diagnostic education curricula at academic centers.9-11 With the COVID-19 pandemic, it has become more difficult to ensure that trainees have an in-person learning community to discuss and receive feedback. This has led to a wider adoption of application-based clinical exercises, educational podcasts, and curricular innovations to support these virtual efforts.
The Human Diagnosis Project (Human Dx) is a smart-phone application that provides a platform for individuals to submit clinical cases that can be rapidly peer-reviewed and disseminated to the larger user pool. Human Dx is notable for fostering a strong sense of community amongst its users.12,13 Case consumers and case creators are able to engage in further discussion after solving a case, and opportunities for feedback and growth are ample.
Medical education podcasts have taken on greater importance during the pandemic.14,15 Many educators have begun referring their learners towards certain podcasts as in-person learning communities have been put on hold. Medical professionals may appreciate the up-to-date and candid conversations held on many podcasts, which can provide both educationally useful and emotionally sympathetic connections to their distanced peers. Similarly, while academic clinicians previously benefitted from invited grand rounds speakers, they may now find that such expert discussants are most easily accessible through their appearances on podcasts.
As institutions suspended clerkships during the pandemic, many created virtual communities for trainees to engage in diagnostic reasoning and education. They built novel curricula that meld asynchronous learning with online community-based learning.14 Gamified learning tools and quizzes have also been incorporated into these hybrid curricula to help ensure participation of learners within their virtual communities.16,17
VIRTUAL CONFERENCES
Perhaps the most notable advance in digital communities catalyzed by the COVID-19 pandemic has been the increasing reliance on and comfort with video-based software. While many of our clinical, administrative, and social activities have migrated toward these virtual environments, they have also been used for a variety of activities related to education and professional development.
As institutions struggled to adapt to physical distancing, many medical schools and residency programs have moved their regular meetings and conferences to virtual platforms. Similar free and open-access conferences have also emerged, including the “Virtual Morning Report” (VMR) series from The Clinical Problem Solvers podcast, wherein a few individuals are invited to discuss a case on the video conference, with the remainder of the audience contributing via the chat feature.
Beyond the growing popularity of video conferencing for education, these virtual sessions have become their own community. On The Clinical Problem Solvers VMR, many participants, ranging from preclinical students to seasoned attendings, show up on a daily basis and interact with each other as close friends, as do members of more insular institutional sessions (eg, residency run reports). In these strangely isolating times, many of us have experienced comfort in seeing the faces of our friends and colleagues joining us to listen and discuss cases.
Separately, many professional societies have struggled with how to replace their large yearly in-person conferences, which would pose substantial infectious risks were they to be held in person. While many of those scheduled to occur during the early days of the pandemic were canceled or held limited online sessions, the trend towards virtual conference platforms seems to be accelerating. Organizers of the 2020 Conference on Retroviruses and Opportunistic Infections (March 8-11, 2020) decided to convert from an in-person to entirely virtual conference 48 hours before it started. With the benefit of more forewarning, other conferences are planning and exploring best practices to promote networking and advancement of research goals at future academic meetings.18,19
BENEFITS OF VIRTUAL COMMUNITIES
The growing importance of these new digital communities could be viewed as a necessary evolution in the way that we gather and learn from each other. Traditional physician communities were inherently restricted by location, specialty, and hierarchy, thereby limiting the dissemination of knowledge and changes to professional norms. These restrictions could conceivably insulate and promote elite institutions in a fashion that perpetuates the inequalities within global medical systems. Unrestricted and open-access virtual communities, in contrast, have the potential to remove historical barriers and connect first-class mentors with trainees they would never have met otherwise.
Beyond promoting a more equitable distribution of knowledge and resources, these virtual communities are well suited to harness the benefits of group learning. The concept of communities of practice (CoP) refers to groupings of individuals involved in a personal or professional endeavor, with the community facilitating advancement of their own knowledge and skill set. Members of the CoP learn from each other, with more established members passing down essential knowledge and cultural norms. The three main components of CoP are maintaining a social network, a mutual enterprise (eg, a common goal), and a shared repertoire (eg, experiences, languages, etc).
Designing virtual learning spaces with these aspects in mind may allow these communities to function as CoPs. Some strategies include use of chat functions in videoconferences (to promote further dialogue) and development of dedicated sessions for specific subgroups or aims (eg, professional mentorship). The anticipated benefits of integrating virtual CoPs into medical education are notable, as a number of studies have already suggested that they are effective for disseminating knowledge, enhancing social learning, and aiding with professional development.7,20-23 These virtual CoPs continue to evolve, however, and further research is warranted to clarify how best to utilize them in medical education and professional societies.
CONCLUSION
Amidst the tragic loss of lives and financial calamity, the COVID-19 pandemic has also spurred innovation and change in the way health professionals learn and communicate. Going forward, the medical establishment should capitalize on these recent innovations and work to further build, recognize, and foster such digital gathering spaces in order to more equitably and effectively disseminate knowledge and educational resources.
Despite physical distancing, health professionals have grown closer during these past few months. Innovations spurred by the pandemic have made us stronger and more united. Our experience with social media, online learning communities, and virtual conferences suggests the opportunity to grow and evolve from this experience. As Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said in March 2020, “...life is not going to be how it used to be [after the pandemic]…” Let’s hope he’s right.
ACKNOWLEDGMENTS
We thank Reza Manesh, MD, Rabih Geha, MD, and Jack Penner, MD, for their careful review of the manuscript.
Throughout history, physicians have formed communities to aid in the dissemination of knowledge, skills, and professional norms. From local physician groups to international societies and conferences, this drive to connect with members of our profession across the globe is timeless. We do so to learn from each other and continue to move the field of medicine forward.
Yet, these communities are being strained by necessary physical distancing required during the COVID-19 pandemic. Many physicians accustomed to a sense of community are now finding themselves surprisingly isolated and alone. Into this distanced landscape, however, new digital groups—specifically social media (SoMe), online learning communities, and virtual conferences—have emerged. We are all active members in virtual communities; all of the authors are team members of The Clinical Problem Solvers podcast and one author of this paper, A.P., has previously served as the medical education lead for the Human Diagnosis Project. Both entities are described later in this article. Here, we provide an overview of these virtual communities and discuss how they have the potential to more equitably and effectively disseminate medical knowledge and education both during and after the COVID-19 pandemic (Table).

SOCIAL MEDIA
Even prior to the COVID-19 pandemic, SoMe—especially Twitter—had become a virtual gathering place where digital colleagues exchange Twitter handles like business cards.1,2 They celebrate each other’s achievements and provide support during difficult times.
Importantly, the format of Twitter tends toward a flattened hierarchy. It is this egalitarian nature that has served SoMe well in its position as a modern learning community. Users from across the experience spectrum engage with and create novel educational content. This often occurs in the form of Tweetorials, or short lessons conveyed over a series of linked tweets. These have gained immense popularity on the platform and are becoming increasingly recognized forms of scholarship.3 Further, case-based lessons have become ubiquitous and are valuable opportunities for users to learn from other members of their digital communities. During the current pandemic, SoMe has become extremely important in the early dissemination and critique of the slew of research on the COVID-19 crisis.4
Beyond its role as an educational platform, SoMe functions as a virtual gathering place for members of the medical community to discuss topics relevant to the field. Subspecialists and researchers have gathered in digital journal clubs (eg, #NephJC, #IDJClub, #BloodandBone) and a number of journals have hosted live Twitter chats covering topics like controversies in clinical practice or professional development (eg, #JHMChat). More recently, social issues affecting the medical field, such as gender equity and the growing antiracism movement, have led to robust discussion on this medium.
Beyond Twitter, many medical professionals gather and exchange ideas on other platforms. Virtual networking and educational groups have arisen using Slack and Facebook.5-7 Trainees and faculty members alike consume and produce content on YouTube, which often serve to teach technical skills.8 Given widespread use of SoMe, we anticipate that the range of platforms utilized by medical professionals will continue to expand in the future.
ONLINE LEARNING COMMUNITIES
There have long existed multiple print and online forums dedicated to the development of clinical skills. These include clinical challenges in medical journals, interactive online cases, and more formal diagnostic education curricula at academic centers.9-11 With the COVID-19 pandemic, it has become more difficult to ensure that trainees have an in-person learning community to discuss and receive feedback. This has led to a wider adoption of application-based clinical exercises, educational podcasts, and curricular innovations to support these virtual efforts.
The Human Diagnosis Project (Human Dx) is a smart-phone application that provides a platform for individuals to submit clinical cases that can be rapidly peer-reviewed and disseminated to the larger user pool. Human Dx is notable for fostering a strong sense of community amongst its users.12,13 Case consumers and case creators are able to engage in further discussion after solving a case, and opportunities for feedback and growth are ample.
Medical education podcasts have taken on greater importance during the pandemic.14,15 Many educators have begun referring their learners towards certain podcasts as in-person learning communities have been put on hold. Medical professionals may appreciate the up-to-date and candid conversations held on many podcasts, which can provide both educationally useful and emotionally sympathetic connections to their distanced peers. Similarly, while academic clinicians previously benefitted from invited grand rounds speakers, they may now find that such expert discussants are most easily accessible through their appearances on podcasts.
As institutions suspended clerkships during the pandemic, many created virtual communities for trainees to engage in diagnostic reasoning and education. They built novel curricula that meld asynchronous learning with online community-based learning.14 Gamified learning tools and quizzes have also been incorporated into these hybrid curricula to help ensure participation of learners within their virtual communities.16,17
VIRTUAL CONFERENCES
Perhaps the most notable advance in digital communities catalyzed by the COVID-19 pandemic has been the increasing reliance on and comfort with video-based software. While many of our clinical, administrative, and social activities have migrated toward these virtual environments, they have also been used for a variety of activities related to education and professional development.
As institutions struggled to adapt to physical distancing, many medical schools and residency programs have moved their regular meetings and conferences to virtual platforms. Similar free and open-access conferences have also emerged, including the “Virtual Morning Report” (VMR) series from The Clinical Problem Solvers podcast, wherein a few individuals are invited to discuss a case on the video conference, with the remainder of the audience contributing via the chat feature.
Beyond the growing popularity of video conferencing for education, these virtual sessions have become their own community. On The Clinical Problem Solvers VMR, many participants, ranging from preclinical students to seasoned attendings, show up on a daily basis and interact with each other as close friends, as do members of more insular institutional sessions (eg, residency run reports). In these strangely isolating times, many of us have experienced comfort in seeing the faces of our friends and colleagues joining us to listen and discuss cases.
Separately, many professional societies have struggled with how to replace their large yearly in-person conferences, which would pose substantial infectious risks were they to be held in person. While many of those scheduled to occur during the early days of the pandemic were canceled or held limited online sessions, the trend towards virtual conference platforms seems to be accelerating. Organizers of the 2020 Conference on Retroviruses and Opportunistic Infections (March 8-11, 2020) decided to convert from an in-person to entirely virtual conference 48 hours before it started. With the benefit of more forewarning, other conferences are planning and exploring best practices to promote networking and advancement of research goals at future academic meetings.18,19
BENEFITS OF VIRTUAL COMMUNITIES
The growing importance of these new digital communities could be viewed as a necessary evolution in the way that we gather and learn from each other. Traditional physician communities were inherently restricted by location, specialty, and hierarchy, thereby limiting the dissemination of knowledge and changes to professional norms. These restrictions could conceivably insulate and promote elite institutions in a fashion that perpetuates the inequalities within global medical systems. Unrestricted and open-access virtual communities, in contrast, have the potential to remove historical barriers and connect first-class mentors with trainees they would never have met otherwise.
Beyond promoting a more equitable distribution of knowledge and resources, these virtual communities are well suited to harness the benefits of group learning. The concept of communities of practice (CoP) refers to groupings of individuals involved in a personal or professional endeavor, with the community facilitating advancement of their own knowledge and skill set. Members of the CoP learn from each other, with more established members passing down essential knowledge and cultural norms. The three main components of CoP are maintaining a social network, a mutual enterprise (eg, a common goal), and a shared repertoire (eg, experiences, languages, etc).
Designing virtual learning spaces with these aspects in mind may allow these communities to function as CoPs. Some strategies include use of chat functions in videoconferences (to promote further dialogue) and development of dedicated sessions for specific subgroups or aims (eg, professional mentorship). The anticipated benefits of integrating virtual CoPs into medical education are notable, as a number of studies have already suggested that they are effective for disseminating knowledge, enhancing social learning, and aiding with professional development.7,20-23 These virtual CoPs continue to evolve, however, and further research is warranted to clarify how best to utilize them in medical education and professional societies.
CONCLUSION
Amidst the tragic loss of lives and financial calamity, the COVID-19 pandemic has also spurred innovation and change in the way health professionals learn and communicate. Going forward, the medical establishment should capitalize on these recent innovations and work to further build, recognize, and foster such digital gathering spaces in order to more equitably and effectively disseminate knowledge and educational resources.
Despite physical distancing, health professionals have grown closer during these past few months. Innovations spurred by the pandemic have made us stronger and more united. Our experience with social media, online learning communities, and virtual conferences suggests the opportunity to grow and evolve from this experience. As Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said in March 2020, “...life is not going to be how it used to be [after the pandemic]…” Let’s hope he’s right.
ACKNOWLEDGMENTS
We thank Reza Manesh, MD, Rabih Geha, MD, and Jack Penner, MD, for their careful review of the manuscript.
1. Markham MJ, Gentile D, Graham DL. Social media for networking, professional development, and patient engagement. Am Soc Clin Oncol Educ Book. 2017;37:782-787. https://doi.org/10.1200/EDBK_180077
2. Melvin L, Chan T. Using Twitter in clinical education and practice. J Grad Med Educ. 2014;6(3):581-582. https://doi.org/10.4300/JGME-D-14-00342.1
3. Breu AC. Why is a cow? Curiosity, Tweetorials, and the return to why. N Engl J Med. 2019;381(12):1097-1098. https://doi.org/10.1056/NEJMp1906790
4. Chan AKM, Nickson CP, Rudolph JW, Lee A, Joynt GM. Social media for rapid knowledge dissemination: early experience from the COVID-19 pandemic. Anaesthesia. 2020:10.1111/anae.15057. https://doi.org/10.1111/anae.15057
5. Pander T, Pinilla S, Dimitriadis K, Fischer MR. The use of Facebook in medical education--a literature review. GMS Z Med Ausbild. 2014;31(3):Doc33. https://doi.org/10.3205/zma000925
6. Cree-Green M, Carreau AM, Davis SM, et al. Peer mentoring for professional and personal growth in academic medicine. J Investig Med. 2020;68(6):1128-1134. https://doi.org/10.1136/jim-2020-001391
7. Yarris LM, Chan TM, Gottlieb M, Juve AM. Finding your people in the digital age: virtual communities of practice to promote education scholarship. J Grad Med Educ. 2019;11(1):1-5. https://doi.org/10.4300/JGME-D-18-01093.1
8. Sterling M, Leung P, Wright D, Bishop TF. The use of social media in graduate medical education: a systematic review. Acad Med. 2017;92(7):1043-1056. https://doi.org/10.1097/ACM.0000000000001617
9. Manesh R, Dhaliwal G. Digital tools to enhance clinical reasoning. Med Clin North Am. 2018;102(3):559-565. https://doi.org/10.1016/j.mcna.2017.12.015
10. Subramanian A, Connor DM, Berger G, et al. A curriculum for diagnostic reasoning: JGIM’s exercises in clinical reasoning. J Gen Intern Med. 2019;34(3):344-345. https://doi.org/10.1007/s11606-018-4689-y
11. Olson APJ, Singhal G, Dhaliwal G. Diagnosis education - an emerging field. Diagnosis (Berl). 2019;6(2):75-77. https://doi.org/10.1515/dx-2019-0029
12. Chatterjee S, Desai S, Manesh R, Sun J, Nundy S, Wright SM. Assessment of a simulated case-based measurement of physician diagnostic performance. JAMA Netw Open. 2019;2(1):e187006. https://doi.org/10.1001/jamanetworkopen.2018.7006
13. Russell SW, Desai SV, O’Rourke P, et al. The genealogy of teaching clinical reasoning and diagnostic skill: the GEL Study. Diagnosis (Berl). 2020;7(3):197-203. https://doi.org/10.1515/dx-2019-0107
14. Geha R, Dhaliwal G. Pilot virtual clerkship curriculum during the COVID-19 pandemic: podcasts, peers, and problem-solving. Med Educ. 2020;54(9):855-856. https://doi.org/10.1111/medu.14246
15. AlGaeed M, Grewal M, Richardson PK, Leon Guerrero CR. COVID-19: Neurology residents’ perspective. J Clin Neurosci. 2020;78:452-453. https://doi.org/10.1016/j.jocn.2020.05.032
16. Moro C, Stromberga Z. Enhancing variety through gamified, interactive learning experiences. Med Educ. 2020. Online ahead of print. https://doi.org/10.1111/medu.14251
17. Morawo A, Sun C, Lowden M. Enhancing engagement during live virtual learning using interactive quizzes. Med Educ. 2020. Online ahead of print. https://doi.org/10.1111/medu.14253
18. Rubinger L, Gazendam A, Ekhtiari S, et al. Maximizing virtual meetings and conferences: a review of best practices. Int Orthop. 2020;44(8):1461-1466. https://doi.org/10.1007/s00264-020-04615-9
19. Woolston C. Learning to love virtual conferences in the coronavirus era. Nature. 2020;582(7810):135-136. https://doi.org/10.1038/d41586-020-01489-0
20. Cruess RL, Cruess SR, Steinert Y. Medicine as a community of practice: implications for medical education. Acad Med. 2018;93(2):185-191. https://doi.org/10.1097/ACM.0000000000001826
21. McLoughlin C, Patel KD, O’Callaghan T, Reeves S. The use of virtual communities of practice to improve interprofessional collaboration and education: findings from an integrated review. J Interprof Care. 2018;32(2):136-142. https://doi.org/10.1080/13561820.2017.1377692
22. Barnett S, Jones SC, Caton T, Iverson D, Bennett S, Robinson L. Implementing a virtual community of practice for family physician training: a mixed-methods case study. J Med Internet Res. 2014;16(3):e83. https://doi.org/10.2196/jmir.3083
23. Healy MG, Traeger LN, Axelsson CGS, et al. NEJM Knowledge+ Question of the Week: a novel virtual learning community effectively utilizing an online discussion forum. Med Teach. 2019;41(11):1270-1276. https://doi.org/10.1080/0142159X.2019.1635685
1. Markham MJ, Gentile D, Graham DL. Social media for networking, professional development, and patient engagement. Am Soc Clin Oncol Educ Book. 2017;37:782-787. https://doi.org/10.1200/EDBK_180077
2. Melvin L, Chan T. Using Twitter in clinical education and practice. J Grad Med Educ. 2014;6(3):581-582. https://doi.org/10.4300/JGME-D-14-00342.1
3. Breu AC. Why is a cow? Curiosity, Tweetorials, and the return to why. N Engl J Med. 2019;381(12):1097-1098. https://doi.org/10.1056/NEJMp1906790
4. Chan AKM, Nickson CP, Rudolph JW, Lee A, Joynt GM. Social media for rapid knowledge dissemination: early experience from the COVID-19 pandemic. Anaesthesia. 2020:10.1111/anae.15057. https://doi.org/10.1111/anae.15057
5. Pander T, Pinilla S, Dimitriadis K, Fischer MR. The use of Facebook in medical education--a literature review. GMS Z Med Ausbild. 2014;31(3):Doc33. https://doi.org/10.3205/zma000925
6. Cree-Green M, Carreau AM, Davis SM, et al. Peer mentoring for professional and personal growth in academic medicine. J Investig Med. 2020;68(6):1128-1134. https://doi.org/10.1136/jim-2020-001391
7. Yarris LM, Chan TM, Gottlieb M, Juve AM. Finding your people in the digital age: virtual communities of practice to promote education scholarship. J Grad Med Educ. 2019;11(1):1-5. https://doi.org/10.4300/JGME-D-18-01093.1
8. Sterling M, Leung P, Wright D, Bishop TF. The use of social media in graduate medical education: a systematic review. Acad Med. 2017;92(7):1043-1056. https://doi.org/10.1097/ACM.0000000000001617
9. Manesh R, Dhaliwal G. Digital tools to enhance clinical reasoning. Med Clin North Am. 2018;102(3):559-565. https://doi.org/10.1016/j.mcna.2017.12.015
10. Subramanian A, Connor DM, Berger G, et al. A curriculum for diagnostic reasoning: JGIM’s exercises in clinical reasoning. J Gen Intern Med. 2019;34(3):344-345. https://doi.org/10.1007/s11606-018-4689-y
11. Olson APJ, Singhal G, Dhaliwal G. Diagnosis education - an emerging field. Diagnosis (Berl). 2019;6(2):75-77. https://doi.org/10.1515/dx-2019-0029
12. Chatterjee S, Desai S, Manesh R, Sun J, Nundy S, Wright SM. Assessment of a simulated case-based measurement of physician diagnostic performance. JAMA Netw Open. 2019;2(1):e187006. https://doi.org/10.1001/jamanetworkopen.2018.7006
13. Russell SW, Desai SV, O’Rourke P, et al. The genealogy of teaching clinical reasoning and diagnostic skill: the GEL Study. Diagnosis (Berl). 2020;7(3):197-203. https://doi.org/10.1515/dx-2019-0107
14. Geha R, Dhaliwal G. Pilot virtual clerkship curriculum during the COVID-19 pandemic: podcasts, peers, and problem-solving. Med Educ. 2020;54(9):855-856. https://doi.org/10.1111/medu.14246
15. AlGaeed M, Grewal M, Richardson PK, Leon Guerrero CR. COVID-19: Neurology residents’ perspective. J Clin Neurosci. 2020;78:452-453. https://doi.org/10.1016/j.jocn.2020.05.032
16. Moro C, Stromberga Z. Enhancing variety through gamified, interactive learning experiences. Med Educ. 2020. Online ahead of print. https://doi.org/10.1111/medu.14251
17. Morawo A, Sun C, Lowden M. Enhancing engagement during live virtual learning using interactive quizzes. Med Educ. 2020. Online ahead of print. https://doi.org/10.1111/medu.14253
18. Rubinger L, Gazendam A, Ekhtiari S, et al. Maximizing virtual meetings and conferences: a review of best practices. Int Orthop. 2020;44(8):1461-1466. https://doi.org/10.1007/s00264-020-04615-9
19. Woolston C. Learning to love virtual conferences in the coronavirus era. Nature. 2020;582(7810):135-136. https://doi.org/10.1038/d41586-020-01489-0
20. Cruess RL, Cruess SR, Steinert Y. Medicine as a community of practice: implications for medical education. Acad Med. 2018;93(2):185-191. https://doi.org/10.1097/ACM.0000000000001826
21. McLoughlin C, Patel KD, O’Callaghan T, Reeves S. The use of virtual communities of practice to improve interprofessional collaboration and education: findings from an integrated review. J Interprof Care. 2018;32(2):136-142. https://doi.org/10.1080/13561820.2017.1377692
22. Barnett S, Jones SC, Caton T, Iverson D, Bennett S, Robinson L. Implementing a virtual community of practice for family physician training: a mixed-methods case study. J Med Internet Res. 2014;16(3):e83. https://doi.org/10.2196/jmir.3083
23. Healy MG, Traeger LN, Axelsson CGS, et al. NEJM Knowledge+ Question of the Week: a novel virtual learning community effectively utilizing an online discussion forum. Med Teach. 2019;41(11):1270-1276. https://doi.org/10.1080/0142159X.2019.1635685
© 2020 Society of Hospital Medicine
Left Out in the Cold
A previously healthy 4-year-old boy presented to his pediatrician for nasal congestion, left ear pain, and intermittent fevers, which he’d been experiencing for 2 days. His exam was consistent with acute otitis media. Cefdinir was prescribed given a rash allergy to amoxicillin. His fever, congestion, and otalgia improved the next day.
Three days later he developed abdominal pain, fever, and labored breathing; his mother brought him to the emergency department (ED). His temperature was 38.0 °C, heart rate 141 beats per minute, blood pressure 117/71 mm Hg, respiratory rate 22 breaths per minute; he had oxygen saturation of 96% on ambient air. Despite mild accessory muscle use, he appeared comfortable and interactive. His left tympanic membrane was bulging without erythema. His neck was supple and mucous membranes moist. He had neither cervical lymphadenopathy nor conjunctival pallor. The cardiopulmonary exam was normal except for tachycardia. His abdomen was soft and not distended without organomegaly or tenderness.
Upper respiratory tract symptoms are commonly encountered in pediatrics and most often result from self-limited viral processes. Evaluation of a child with upper respiratory tract symptoms aims to identify serious causes like meningitis, as well as assessing the need for antimicrobial therapy. Supportive management is often appropriate in otitis media. His new, more concerning symptoms portend either a progression of the original process causing his upper respiratory tract symptoms or a separate etiology. It is key to determine which signs and symptoms are associated with the primary process and which are compensatory or secondary. If he were to be more ill appearing, for example, it is possible that his respiratory distress may be related to an underlying systemic illness rather than a primary lung process. Respiratory distress, abdominal pain, and fever could be a result of sepsis from an intrabdominal process such as ruptured appendicitis, intussusception, or malrotation with volvulus. Other causes of sepsis, such as meningitis or severe mastoiditis, both rare complications of otitis media, should be considered, although he does not appear severely ill. Acute myelogenous leukemia or other malignancies and illnesses associated with immunodeficiency can present with sepsis and chloromas in the middle ear that can be misconstrued as otitis media.
A chest radiograph demonstrated left lower lobe patchy opacities concerning for pneumonia. Rapid respiratory syncytial virus and influenza antigen test results were negative. Laboratory testing for general bloodwork was not obtained. He was administered a single dose of intramuscular ceftriaxone, prescribed a 5-day course of azithromycin, and discharged home. The child’s breathing gradually improved, but he continued to have subjective fevers. Two days later, he developed dark red urine. His mother brought him back to the outpatient clinic.
At the time of the ED visit, a diagnosis of community-acquired pneumonia was plausible given fever, mildly increased work of breathing, and an opacification on chest radiography. Most community-acquired pneumonia is caused by viruses; common bacterial causes for his age include Streptococcus pneumoniae and Moraxella catarrhalis. The first-line treatment for uncomplicated community-acquired pneumonia in children is amoxicillin, but this was appropriately avoided given his allergy.
The persistent fevers are surprising. The improvement in breathing corresponds to the treatment (and resolution) of community-acquired pneumonia. However, the development of dark urine does not. Red urine—in the absence of ingested pigments (such as those found in beets)—usually results from hematuria, hemoglobinuria, or myoglobinuria. Gross hematuria can originate from the kidneys to the urethral meatus. Abdominal masses, kidney trauma, or underlying kidney disease may all present with gross hematuria (or microscopic hematuria, seen only on urinalysis). The urine should be examined for the presence of heme, protein, and for evidence of infection; microscopy should be performed to examine for cellular casts and dysmorphic red cells. Tests of renal function, a comprehensive metabolic panel, evaluation of hematologic indexes, and assessments of inflammatory markers should be performed.
The child lived with his parents and had no siblings. He experienced no physical trauma, and there was no family history of kidney disease or hematuria. His father had a persistent cough and fever for 1 month, but recovered around the time the patient began to experience his initial symptoms. This was the patient’s third diagnosis of pneumonia. He had not traveled and was up to date with immunizations. He attended day care.
The fact that this is not the first episode of “pneumonia” raises important possibilities. The most likely one is that the child has had multiple viral infections; however, he could have an underlying primary immunodeficiency (PI) that predisposes him to recurrent infections. More severe PIs often present with recurrent sepsis, bacteremia, and failure to thrive, none of which were present in this case. Less severe PIs (such as selective IgA deficiency) could be possible. Another possibility is that these recurrent episodes of pneumonia are a relapsing and remitting noninfectious process, such as an antineutrophil cytoplasmic antibodies–associated vasculitis or anti–glomerular basement membrane disease. The patient’s father’s recent prolonged respiratory symptoms may be suggestive of pertussis or a “walking pneumonia” potentially caused by Mycoplasma or another atypical bacterium.
His temperature was 36.9 °C, heart rate 107 beats per minute, blood pressure was 106/67 mm Hg, and respiratory rate was 24 breaths per minute with oxygen saturation of 100% on ambient air. He was well appearing. His mucous membranes were moist, and oropharynx was clear. He had scleral icterus. The cardiopulmonary exam was normal. He had no significant lymphadenopathy, hepatosplenomegaly, or rashes.
The finding of jaundice is an important diagnostic pivot point, especially when combined with hematuria. The next step is determining if the jaundice is resulting from unconjugated or conjugated hyperbilirubinemia; the former most often stems from hemolysis or impairment in conjugation, while the latter results from intrahepatic or extrahepatic biliary defects. Tests for hepatobiliary injury including evaluations of alanine and aspartate aminotransferases and alkaline phosphatase, as well as for hepatic function such as tests of coagulation, should be performed.
The patient was referred to the ED and admitted for further evaluation. A complete blood count revealed a white blood cell (WBC) count of 10,700/µL (61% polymorphonuclear neutrophils, 30% lymphocytes, 5% monocytes, 3% eosinophils, 1% basophils), hemoglobin count was 10.3 g/dL (reticulocyte 2% with absolute reticulocyte count 58,400/μL), and platelet count was 265,000/µL. Components of the basic metabolic panel were within reference ranges except for a mildly elevated blood urea nitrogen level of 14 mg/dL with normal creatinine level of 0.3 mg/dL. Total protein was 6.7 g/dL (reference range, 6.4-8.3) and albumin 3.9 g/dL (reference range, 3.4-4.8). Alkaline phosphatase level was 188 U/L (reference range, 44-147), aspartate aminotransferase level 76 U/L (reference range, 0-40), and alanine aminotransferase level 12 U/L (reference range, 7-40). Total bilirubin level was 2.4 mg/dL (reference range, less than 1.5) with direct bilirubin level of 0.4 mg/dL. His C-reactive protein level was 1.5 mg/mL (reference range, 0-0.75). Creatinine kinase (CK) level was 2,550 U/L (reference range, 2-198). International Normalized Ratio (INR) was 1.0. Urinalysis was notable for 2+ proteinuria, large hemoglobin pigment, and 6 red blood cells per high power field (reference range, 0-4).
His blood urea nitrogen is elevated, reflecting either prerenal azotemia or increased absorption of nitrogenous products. Unconjugated hyperbilirubinemia may result from impaired hepatic bilirubin uptake (such as in heart failure or portosystemic shunts), impaired bilirubin conjugation (resulting from genetic conditions or drugs), or excess bilirubin production (such as in hemolysis); his anemia and lack of other evidence of hepatic dysfunction point to hemolysis as the etiology. The reticulocyte production index is approximately 1%, which suggests that an increase in erythrocyte generation is present but inadequate. This, however, does not mean that an erythrocyte production abnormality is present since reticulocytosis can be delayed in many cases of acute hemolytic anemia. It is also possible that the same hemolytic process is affecting mature and immature erythrocytes. A peripheral blood smear should be reviewed for evidence of intravascular hemolysis and testing for autoimmune hemolysis should be performed. Notably, his white blood cell and platelet counts are preserved, which makes a bone marrow–involved malignancy or infiltrative process less likely. The alkaline phosphatase elevation may result from either intrahepatic or extrahepatic biliopathy; bone damage is also possible. The elevation of aspartate aminotransferase, CK, and potassium, along with marked urinary heme pigment, may indicate muscle damage; the most common myositis in children is benign acute childhood myositis resulting from viral infection. However, the moderate level of CK elevation seen in this case is nonspecific and can result from many different etiologies. A metabolic myopathy, such as carnitine palmitoyltransferase II deficiency, can be made worse by metabolic stress and result in rhabdomyolysis; the presentations of inborn errors of metabolism are varied and a planned-out, stepwise approach in evaluation is fundamental.
Lactic acid dehydrogenase (LDH) level was 1,457 U/L (reference range, 140-280), and haptoglobin level was less than 6 mg/dL (reference range, 30-200). Peripheral blood smear demonstrated occasional atypical, reactive-appearing lymphocytes with red cell clumping and agglutination, as well as rare target, burr, and fragmented red cells. Test results for urine myoglobin were negative. Results of urine culture were negative. No blood culture was collected.
The elevated LDH, decreased haptoglobin, and findings on the peripheral blood smear confirm hemolysis. The clumping of erythrocytes can be artifactual in the preparation of peripheral smears, but when considered in the context of hemolysis, may be clinically important. Clumping of erythrocytes on the peripheral smear indicates the binding of a protein to antigens on the erythrocyte membrane; when this occurs below body temperature, this is consistent with the presence of a “cold agglutinin,” usually an IgM antibody directed at erythrocyte surface antigens that causes agglutination and destruction, especially in cooler areas of the body. This is a well-known complication of Mycoplasma pneumoniae infections as well as Epstein-Barr virus (EBV) infections; it may also occur with lymphoid malignancies or autoimmune disease.
Direct Coombs IgG test findings were negative, direct Coombs C3 test was positive, and direct Coombs polyspecific test was positive. M pneumoniae IgG antibody level was 1.4 mg/dL (reference ranges: <0.9, negative; 0.91-1.09, equivocal; >1.1, positive); M pneumoniae IgM level was 529 U/mL (reference range: <770, negative). EBV capsid IgM and IgG levels were undetectable. EBV nuclear antigen IgG level was also undetectable. EBV viral load was fewer than 10 copies/mL. Antinuclear antibodies (ANA) level was negative. General IgE and IgM levels were normal, at 11 and 81 mg/dL, respectively. Repeat complete blood count showed WBC of 7,800/µL, hemoglobin of 8.7 g/dL, and platelet count of 341,000/µL. The patient’s hemoglobin remained stable during hospitalization.
This directed testing is helpful in further classifying the patient’s hemolytic anemia. Autoimmune hemolytic anemias are classified into warm antibody–mediated, cold antibody–mediated, and mixed-type forms; drug-induced and alloimmune hemolytic anemias also occur. In addition, both systemic lupus erythematosus and antiphospholipid antibody syndrome can have hemolytic anemia with variable Coombs testing results; neither fit well in this case. The absence of red blood cell–directed IgG antibodies substantially decreases the likelihood of warm antibody–mediated hemolytic anemia. In cold antibody–mediated hemolytic anemia, antibodies bind to the erythrocyte membrane and then adhere to complement C3, which leads to both intravascular and extravascular hemolysis. Important types of cold antibody–mediated hemolytic anemia in children are primary and secondary cold agglutinin disease, along with paroxysmal cold hemoglobinuria. The Donath-Landsteiner test can be helpful in differentiating these conditions. Antibodies to Mycoplasma may be delayed in response to acute infection, and a child who is reinfected may only produce IgG antibodies. Given the patient’s clinical stability and previous health, the most likely diagnosis is Mycoplasma-induced cold antibody–mediated hemolytic anemia. It may be helpful to check convalescent titers to Mycoplasma in 2 to 4 weeks.
Donath-Landsteiner (D-L) antibody test results were positive. Medication-derived hemolytic anemia testing was conducted, but the presence of positive D-L antibody makes the test results inconclusive. This ultimately led to a diagnosis of paroxysmal cold hemoglobinuria (PCH), presumably triggered by a viral syndrome. Convalescent titers to Mycoplasma were not checked given clinical improvement. Because the patient’s hemoglobin was stable during hospitalization, he was not treated with steroids. His parents were counseled on avoiding cold temperatures for several days. Within 1 month, his hemoglobin had recovered without further evidence of hemolysis.
DISCUSSION
Hemolytic anemia refers to the accelerated destruction of red blood cells and can be further classified as acquired or hereditary.1 Hereditary conditions causing hemolytic anemia include enzymopathies (eg, glucose-6-phosphate dehydrogenase deficiency), hemoglobinopathies (eg, sickle cell disease), and membrane abnormalities (eg, hereditary spherocytosis). Acquired pathologies include microangiopathic hemolytic anemia (MAHA), anemias directly caused by certain infections such as malaria, and immune-mediated (Coombs-positive) hemolytic anemias.
MAHA can sometimes be life-threatening and is therefore important to identify quickly. In the right clinical context, such processes may be rapidly recognized by the presence of schistocytes on blood smear in addition to an elevated serum LDH level. Schistocytes suggest mechanical destruction of erythrocytes in the vasculature, the hallmark of MAHA. Important MAHAs include thrombocytopenic purpura, hemolytic-uremic syndrome, and disseminated intravascular coagulation. Though this patient did have a mildly elevated LDH, MAHA was less likely because there were no schistocytes on the blood smear.
Autoimmune hemolytic anemias (AIHAs) are another important subset of acquired hemolytic anemias. AIHAs occur when there is antibody-mediated destruction of erythrocytes. The direct Coombs test evaluates for antibody- or complement-coated erythrocytes. After administration of anti-IgG and anti-C3 serum, the test evaluates for agglutination of the red cells caused by attached antibodies or complement. Coombs-positive AIHA can also be categorized by the temperature of agglutination. “Warm” hemolysis often involves IgG autoantibodies (ie, warm agglutinins), while “cold” antibodies, usually IgM autoantibodies, bind at colder temperatures (0-4 °C) and activate complements, including C3. In this patient, the Coombs C3 was positive while the Coombs IgG was negative, which is more suggestive of a cold complement–mediated pathway.
Cold AIHA can be further categorized into primary cold agglutinin disease, secondary cold agglutinin disease, and PCH. Primary cold agglutinin disease is an autoimmune disorder that mostly occurs in adults. Secondary cold AIHA can often be triggered by bacterial infection (commonly M pneumoniae) or viruses including EBV, measles, and mumps.2 Medications, including penicillin and cephalosporins, can also be implicated. Secondary cold AIHA is also linked with autoimmune diseases, such as systemic lupus erythematosus and lymphoproliferative disorders. PCH can be identified with the unique presence of a specific autoantibody (ie, D-L autoantibody) that agglutinates at cold temperatures but dissociates on subsequent rewarming.3 Complement remains affixed and activates hemolysis.
The D-L antibody responsible for PCH is an IgG antibody to the P-antigen present on the erythrocyte surface. Since the Coombs test is conducted at normal temperature, it will be positive for the affixed complement but not for IgG. The underlying mechanism for PCH was proposed by Julius Donath, MD, and Karl Landsteiner, MD, in 1904 and is considered to be the first description of autoimmune disease being precipitated by antibodies.4 The D-L antibody test itself is uncommonly performed and somewhat difficult to interpret, particularly in adults, and may lead to false-negative results.5
PCH is an acquired, cold AIHA more common to children6,7 and may account for up to 33% of pediatric AIHA cases.8 Typical presentation is after an upper respiratory tract illness; however, the trigger is often not identified. Implicated triggers include a number of viruses.9 Clinical presentation includes findings of intravascular hemolysis similar to those in our patient. The pathogenic IgG autoantibody is polyclonal and is likely formed because of immune stimulation, which is consistent with the predominance of nonmalignant triggers of this disease process.10 Hemolysis and associated symptoms are often exacerbated with cold exposure; both typically resolve within 2 weeks. In recurrent cases, which are a minority, immunosuppression may be considered.10
PCH remains an often-understated cause of hemolytic anemia particularly in children. Lacking obvious pathognomonic clinical symptoms, it may be overlooked for other forms of AIHA or MAHA. However, with a structured approach to evaluation, as with this patient who had hematuria and jaundice, early diagnosis can prevent an unnecessarily extensive workup and can provide reassurance to patient and parents. By understanding the basic categories of hemolytic anemia, the relevant blood testing available, and interpretation of Coombs test results, clinicians can ensure that PCH is a diagnosis that is not left out in the cold.
KEY TEACHING POINTS
- Examination for schistocytes on a blood smear can help identify life-threatening causes of hemolytic anemia.
- Characterization of cold AIHA includes defining the underlying etiology as primary cold agglutinin disease, secondary cold agglutinin disease, or PCH.
- PCH is a cold AIHA that is an underrecognized cause of hemolytic anemia in children. The diagnosis of PCH is made by testing for the presence of the D-L antibody.
1. Dhaliwal G, Cornett PA, Tierney LM Jr. Hemolytic anemia. Am Fam Physician. 2004;69(11):2599-2606.
2. Djaldetti M. Paroxysmal cold hemoglobinuria. CRC Crit Rev Clin Lab Sci. 1978;9(1):49-83. https://doi.org/10.3109/10408367809150915
3. Levine P, Celano MJ, Falkowski F. The specificity of the antibody in paroxysmal cold hemoglobinuria (P.C.H.). Transfusion. 1963;3(4):278-280. https://doi.org/10.1111/j.1537-2995.1963.tb04643.x
4. Donath J, Landsteiner K. Uber Paroxysmale Hamoglobinurie. Munch Med Wochenschr. 1904;51:1590-1593
5. Zeller MP, Arnold DM, Al Habsi K, et al. Paroxysmal cold hemoglobinuria: a difficult diagnosis in adult patients. Transfusion. 2017;57(1):137-143. https://doi.org/10.1111/trf.13888
6. Göttsche B, Salama A, Mueller-Eckhardt C. Donath-Landsteiner autoimmune hemolytic anemia in children. a study of 22 cases. Vox Sang. 1990;58(4):281-286. https://doi.org/10.1111/j.1423-0410.1990.tb05000.x
7. Sokol RJ, Booker DJ, Stamps R. Erythropoiesis: paroxysmal cold haemoglobinuria: a clinico-pathological study of patients with a positive Donath-Landsteiner test. Hematology. 1999;4(2):137-164. https://doi.org/10.1080/10245332.1999.11746439
8. Petz LD. Cold antibody autoimmune hemolytic anemias. Blood Rev. 2008;22(1):1-15. https://doi.org/10.1016/j.blre.2007.08.002
9. Leibrandt R, Angelino K, Vizel-Schwartz M, Shapira I. Paroxysmal cold hemoglobinuria in an adult with respiratory syncytial virus. Case Rep Hematol. 2018;2018:1-3. https://doi.org/10.1155/2018/7586719
10. Gertz MA. Management of cold haemolytic syndrome. Br J Haematol. 2007;138(4):422-429. https://doi.org/10.1111/j.1365-2141.2007.06664.x
A previously healthy 4-year-old boy presented to his pediatrician for nasal congestion, left ear pain, and intermittent fevers, which he’d been experiencing for 2 days. His exam was consistent with acute otitis media. Cefdinir was prescribed given a rash allergy to amoxicillin. His fever, congestion, and otalgia improved the next day.
Three days later he developed abdominal pain, fever, and labored breathing; his mother brought him to the emergency department (ED). His temperature was 38.0 °C, heart rate 141 beats per minute, blood pressure 117/71 mm Hg, respiratory rate 22 breaths per minute; he had oxygen saturation of 96% on ambient air. Despite mild accessory muscle use, he appeared comfortable and interactive. His left tympanic membrane was bulging without erythema. His neck was supple and mucous membranes moist. He had neither cervical lymphadenopathy nor conjunctival pallor. The cardiopulmonary exam was normal except for tachycardia. His abdomen was soft and not distended without organomegaly or tenderness.
Upper respiratory tract symptoms are commonly encountered in pediatrics and most often result from self-limited viral processes. Evaluation of a child with upper respiratory tract symptoms aims to identify serious causes like meningitis, as well as assessing the need for antimicrobial therapy. Supportive management is often appropriate in otitis media. His new, more concerning symptoms portend either a progression of the original process causing his upper respiratory tract symptoms or a separate etiology. It is key to determine which signs and symptoms are associated with the primary process and which are compensatory or secondary. If he were to be more ill appearing, for example, it is possible that his respiratory distress may be related to an underlying systemic illness rather than a primary lung process. Respiratory distress, abdominal pain, and fever could be a result of sepsis from an intrabdominal process such as ruptured appendicitis, intussusception, or malrotation with volvulus. Other causes of sepsis, such as meningitis or severe mastoiditis, both rare complications of otitis media, should be considered, although he does not appear severely ill. Acute myelogenous leukemia or other malignancies and illnesses associated with immunodeficiency can present with sepsis and chloromas in the middle ear that can be misconstrued as otitis media.
A chest radiograph demonstrated left lower lobe patchy opacities concerning for pneumonia. Rapid respiratory syncytial virus and influenza antigen test results were negative. Laboratory testing for general bloodwork was not obtained. He was administered a single dose of intramuscular ceftriaxone, prescribed a 5-day course of azithromycin, and discharged home. The child’s breathing gradually improved, but he continued to have subjective fevers. Two days later, he developed dark red urine. His mother brought him back to the outpatient clinic.
At the time of the ED visit, a diagnosis of community-acquired pneumonia was plausible given fever, mildly increased work of breathing, and an opacification on chest radiography. Most community-acquired pneumonia is caused by viruses; common bacterial causes for his age include Streptococcus pneumoniae and Moraxella catarrhalis. The first-line treatment for uncomplicated community-acquired pneumonia in children is amoxicillin, but this was appropriately avoided given his allergy.
The persistent fevers are surprising. The improvement in breathing corresponds to the treatment (and resolution) of community-acquired pneumonia. However, the development of dark urine does not. Red urine—in the absence of ingested pigments (such as those found in beets)—usually results from hematuria, hemoglobinuria, or myoglobinuria. Gross hematuria can originate from the kidneys to the urethral meatus. Abdominal masses, kidney trauma, or underlying kidney disease may all present with gross hematuria (or microscopic hematuria, seen only on urinalysis). The urine should be examined for the presence of heme, protein, and for evidence of infection; microscopy should be performed to examine for cellular casts and dysmorphic red cells. Tests of renal function, a comprehensive metabolic panel, evaluation of hematologic indexes, and assessments of inflammatory markers should be performed.
The child lived with his parents and had no siblings. He experienced no physical trauma, and there was no family history of kidney disease or hematuria. His father had a persistent cough and fever for 1 month, but recovered around the time the patient began to experience his initial symptoms. This was the patient’s third diagnosis of pneumonia. He had not traveled and was up to date with immunizations. He attended day care.
The fact that this is not the first episode of “pneumonia” raises important possibilities. The most likely one is that the child has had multiple viral infections; however, he could have an underlying primary immunodeficiency (PI) that predisposes him to recurrent infections. More severe PIs often present with recurrent sepsis, bacteremia, and failure to thrive, none of which were present in this case. Less severe PIs (such as selective IgA deficiency) could be possible. Another possibility is that these recurrent episodes of pneumonia are a relapsing and remitting noninfectious process, such as an antineutrophil cytoplasmic antibodies–associated vasculitis or anti–glomerular basement membrane disease. The patient’s father’s recent prolonged respiratory symptoms may be suggestive of pertussis or a “walking pneumonia” potentially caused by Mycoplasma or another atypical bacterium.
His temperature was 36.9 °C, heart rate 107 beats per minute, blood pressure was 106/67 mm Hg, and respiratory rate was 24 breaths per minute with oxygen saturation of 100% on ambient air. He was well appearing. His mucous membranes were moist, and oropharynx was clear. He had scleral icterus. The cardiopulmonary exam was normal. He had no significant lymphadenopathy, hepatosplenomegaly, or rashes.
The finding of jaundice is an important diagnostic pivot point, especially when combined with hematuria. The next step is determining if the jaundice is resulting from unconjugated or conjugated hyperbilirubinemia; the former most often stems from hemolysis or impairment in conjugation, while the latter results from intrahepatic or extrahepatic biliary defects. Tests for hepatobiliary injury including evaluations of alanine and aspartate aminotransferases and alkaline phosphatase, as well as for hepatic function such as tests of coagulation, should be performed.
The patient was referred to the ED and admitted for further evaluation. A complete blood count revealed a white blood cell (WBC) count of 10,700/µL (61% polymorphonuclear neutrophils, 30% lymphocytes, 5% monocytes, 3% eosinophils, 1% basophils), hemoglobin count was 10.3 g/dL (reticulocyte 2% with absolute reticulocyte count 58,400/μL), and platelet count was 265,000/µL. Components of the basic metabolic panel were within reference ranges except for a mildly elevated blood urea nitrogen level of 14 mg/dL with normal creatinine level of 0.3 mg/dL. Total protein was 6.7 g/dL (reference range, 6.4-8.3) and albumin 3.9 g/dL (reference range, 3.4-4.8). Alkaline phosphatase level was 188 U/L (reference range, 44-147), aspartate aminotransferase level 76 U/L (reference range, 0-40), and alanine aminotransferase level 12 U/L (reference range, 7-40). Total bilirubin level was 2.4 mg/dL (reference range, less than 1.5) with direct bilirubin level of 0.4 mg/dL. His C-reactive protein level was 1.5 mg/mL (reference range, 0-0.75). Creatinine kinase (CK) level was 2,550 U/L (reference range, 2-198). International Normalized Ratio (INR) was 1.0. Urinalysis was notable for 2+ proteinuria, large hemoglobin pigment, and 6 red blood cells per high power field (reference range, 0-4).
His blood urea nitrogen is elevated, reflecting either prerenal azotemia or increased absorption of nitrogenous products. Unconjugated hyperbilirubinemia may result from impaired hepatic bilirubin uptake (such as in heart failure or portosystemic shunts), impaired bilirubin conjugation (resulting from genetic conditions or drugs), or excess bilirubin production (such as in hemolysis); his anemia and lack of other evidence of hepatic dysfunction point to hemolysis as the etiology. The reticulocyte production index is approximately 1%, which suggests that an increase in erythrocyte generation is present but inadequate. This, however, does not mean that an erythrocyte production abnormality is present since reticulocytosis can be delayed in many cases of acute hemolytic anemia. It is also possible that the same hemolytic process is affecting mature and immature erythrocytes. A peripheral blood smear should be reviewed for evidence of intravascular hemolysis and testing for autoimmune hemolysis should be performed. Notably, his white blood cell and platelet counts are preserved, which makes a bone marrow–involved malignancy or infiltrative process less likely. The alkaline phosphatase elevation may result from either intrahepatic or extrahepatic biliopathy; bone damage is also possible. The elevation of aspartate aminotransferase, CK, and potassium, along with marked urinary heme pigment, may indicate muscle damage; the most common myositis in children is benign acute childhood myositis resulting from viral infection. However, the moderate level of CK elevation seen in this case is nonspecific and can result from many different etiologies. A metabolic myopathy, such as carnitine palmitoyltransferase II deficiency, can be made worse by metabolic stress and result in rhabdomyolysis; the presentations of inborn errors of metabolism are varied and a planned-out, stepwise approach in evaluation is fundamental.
Lactic acid dehydrogenase (LDH) level was 1,457 U/L (reference range, 140-280), and haptoglobin level was less than 6 mg/dL (reference range, 30-200). Peripheral blood smear demonstrated occasional atypical, reactive-appearing lymphocytes with red cell clumping and agglutination, as well as rare target, burr, and fragmented red cells. Test results for urine myoglobin were negative. Results of urine culture were negative. No blood culture was collected.
The elevated LDH, decreased haptoglobin, and findings on the peripheral blood smear confirm hemolysis. The clumping of erythrocytes can be artifactual in the preparation of peripheral smears, but when considered in the context of hemolysis, may be clinically important. Clumping of erythrocytes on the peripheral smear indicates the binding of a protein to antigens on the erythrocyte membrane; when this occurs below body temperature, this is consistent with the presence of a “cold agglutinin,” usually an IgM antibody directed at erythrocyte surface antigens that causes agglutination and destruction, especially in cooler areas of the body. This is a well-known complication of Mycoplasma pneumoniae infections as well as Epstein-Barr virus (EBV) infections; it may also occur with lymphoid malignancies or autoimmune disease.
Direct Coombs IgG test findings were negative, direct Coombs C3 test was positive, and direct Coombs polyspecific test was positive. M pneumoniae IgG antibody level was 1.4 mg/dL (reference ranges: <0.9, negative; 0.91-1.09, equivocal; >1.1, positive); M pneumoniae IgM level was 529 U/mL (reference range: <770, negative). EBV capsid IgM and IgG levels were undetectable. EBV nuclear antigen IgG level was also undetectable. EBV viral load was fewer than 10 copies/mL. Antinuclear antibodies (ANA) level was negative. General IgE and IgM levels were normal, at 11 and 81 mg/dL, respectively. Repeat complete blood count showed WBC of 7,800/µL, hemoglobin of 8.7 g/dL, and platelet count of 341,000/µL. The patient’s hemoglobin remained stable during hospitalization.
This directed testing is helpful in further classifying the patient’s hemolytic anemia. Autoimmune hemolytic anemias are classified into warm antibody–mediated, cold antibody–mediated, and mixed-type forms; drug-induced and alloimmune hemolytic anemias also occur. In addition, both systemic lupus erythematosus and antiphospholipid antibody syndrome can have hemolytic anemia with variable Coombs testing results; neither fit well in this case. The absence of red blood cell–directed IgG antibodies substantially decreases the likelihood of warm antibody–mediated hemolytic anemia. In cold antibody–mediated hemolytic anemia, antibodies bind to the erythrocyte membrane and then adhere to complement C3, which leads to both intravascular and extravascular hemolysis. Important types of cold antibody–mediated hemolytic anemia in children are primary and secondary cold agglutinin disease, along with paroxysmal cold hemoglobinuria. The Donath-Landsteiner test can be helpful in differentiating these conditions. Antibodies to Mycoplasma may be delayed in response to acute infection, and a child who is reinfected may only produce IgG antibodies. Given the patient’s clinical stability and previous health, the most likely diagnosis is Mycoplasma-induced cold antibody–mediated hemolytic anemia. It may be helpful to check convalescent titers to Mycoplasma in 2 to 4 weeks.
Donath-Landsteiner (D-L) antibody test results were positive. Medication-derived hemolytic anemia testing was conducted, but the presence of positive D-L antibody makes the test results inconclusive. This ultimately led to a diagnosis of paroxysmal cold hemoglobinuria (PCH), presumably triggered by a viral syndrome. Convalescent titers to Mycoplasma were not checked given clinical improvement. Because the patient’s hemoglobin was stable during hospitalization, he was not treated with steroids. His parents were counseled on avoiding cold temperatures for several days. Within 1 month, his hemoglobin had recovered without further evidence of hemolysis.
DISCUSSION
Hemolytic anemia refers to the accelerated destruction of red blood cells and can be further classified as acquired or hereditary.1 Hereditary conditions causing hemolytic anemia include enzymopathies (eg, glucose-6-phosphate dehydrogenase deficiency), hemoglobinopathies (eg, sickle cell disease), and membrane abnormalities (eg, hereditary spherocytosis). Acquired pathologies include microangiopathic hemolytic anemia (MAHA), anemias directly caused by certain infections such as malaria, and immune-mediated (Coombs-positive) hemolytic anemias.
MAHA can sometimes be life-threatening and is therefore important to identify quickly. In the right clinical context, such processes may be rapidly recognized by the presence of schistocytes on blood smear in addition to an elevated serum LDH level. Schistocytes suggest mechanical destruction of erythrocytes in the vasculature, the hallmark of MAHA. Important MAHAs include thrombocytopenic purpura, hemolytic-uremic syndrome, and disseminated intravascular coagulation. Though this patient did have a mildly elevated LDH, MAHA was less likely because there were no schistocytes on the blood smear.
Autoimmune hemolytic anemias (AIHAs) are another important subset of acquired hemolytic anemias. AIHAs occur when there is antibody-mediated destruction of erythrocytes. The direct Coombs test evaluates for antibody- or complement-coated erythrocytes. After administration of anti-IgG and anti-C3 serum, the test evaluates for agglutination of the red cells caused by attached antibodies or complement. Coombs-positive AIHA can also be categorized by the temperature of agglutination. “Warm” hemolysis often involves IgG autoantibodies (ie, warm agglutinins), while “cold” antibodies, usually IgM autoantibodies, bind at colder temperatures (0-4 °C) and activate complements, including C3. In this patient, the Coombs C3 was positive while the Coombs IgG was negative, which is more suggestive of a cold complement–mediated pathway.
Cold AIHA can be further categorized into primary cold agglutinin disease, secondary cold agglutinin disease, and PCH. Primary cold agglutinin disease is an autoimmune disorder that mostly occurs in adults. Secondary cold AIHA can often be triggered by bacterial infection (commonly M pneumoniae) or viruses including EBV, measles, and mumps.2 Medications, including penicillin and cephalosporins, can also be implicated. Secondary cold AIHA is also linked with autoimmune diseases, such as systemic lupus erythematosus and lymphoproliferative disorders. PCH can be identified with the unique presence of a specific autoantibody (ie, D-L autoantibody) that agglutinates at cold temperatures but dissociates on subsequent rewarming.3 Complement remains affixed and activates hemolysis.
The D-L antibody responsible for PCH is an IgG antibody to the P-antigen present on the erythrocyte surface. Since the Coombs test is conducted at normal temperature, it will be positive for the affixed complement but not for IgG. The underlying mechanism for PCH was proposed by Julius Donath, MD, and Karl Landsteiner, MD, in 1904 and is considered to be the first description of autoimmune disease being precipitated by antibodies.4 The D-L antibody test itself is uncommonly performed and somewhat difficult to interpret, particularly in adults, and may lead to false-negative results.5
PCH is an acquired, cold AIHA more common to children6,7 and may account for up to 33% of pediatric AIHA cases.8 Typical presentation is after an upper respiratory tract illness; however, the trigger is often not identified. Implicated triggers include a number of viruses.9 Clinical presentation includes findings of intravascular hemolysis similar to those in our patient. The pathogenic IgG autoantibody is polyclonal and is likely formed because of immune stimulation, which is consistent with the predominance of nonmalignant triggers of this disease process.10 Hemolysis and associated symptoms are often exacerbated with cold exposure; both typically resolve within 2 weeks. In recurrent cases, which are a minority, immunosuppression may be considered.10
PCH remains an often-understated cause of hemolytic anemia particularly in children. Lacking obvious pathognomonic clinical symptoms, it may be overlooked for other forms of AIHA or MAHA. However, with a structured approach to evaluation, as with this patient who had hematuria and jaundice, early diagnosis can prevent an unnecessarily extensive workup and can provide reassurance to patient and parents. By understanding the basic categories of hemolytic anemia, the relevant blood testing available, and interpretation of Coombs test results, clinicians can ensure that PCH is a diagnosis that is not left out in the cold.
KEY TEACHING POINTS
- Examination for schistocytes on a blood smear can help identify life-threatening causes of hemolytic anemia.
- Characterization of cold AIHA includes defining the underlying etiology as primary cold agglutinin disease, secondary cold agglutinin disease, or PCH.
- PCH is a cold AIHA that is an underrecognized cause of hemolytic anemia in children. The diagnosis of PCH is made by testing for the presence of the D-L antibody.
A previously healthy 4-year-old boy presented to his pediatrician for nasal congestion, left ear pain, and intermittent fevers, which he’d been experiencing for 2 days. His exam was consistent with acute otitis media. Cefdinir was prescribed given a rash allergy to amoxicillin. His fever, congestion, and otalgia improved the next day.
Three days later he developed abdominal pain, fever, and labored breathing; his mother brought him to the emergency department (ED). His temperature was 38.0 °C, heart rate 141 beats per minute, blood pressure 117/71 mm Hg, respiratory rate 22 breaths per minute; he had oxygen saturation of 96% on ambient air. Despite mild accessory muscle use, he appeared comfortable and interactive. His left tympanic membrane was bulging without erythema. His neck was supple and mucous membranes moist. He had neither cervical lymphadenopathy nor conjunctival pallor. The cardiopulmonary exam was normal except for tachycardia. His abdomen was soft and not distended without organomegaly or tenderness.
Upper respiratory tract symptoms are commonly encountered in pediatrics and most often result from self-limited viral processes. Evaluation of a child with upper respiratory tract symptoms aims to identify serious causes like meningitis, as well as assessing the need for antimicrobial therapy. Supportive management is often appropriate in otitis media. His new, more concerning symptoms portend either a progression of the original process causing his upper respiratory tract symptoms or a separate etiology. It is key to determine which signs and symptoms are associated with the primary process and which are compensatory or secondary. If he were to be more ill appearing, for example, it is possible that his respiratory distress may be related to an underlying systemic illness rather than a primary lung process. Respiratory distress, abdominal pain, and fever could be a result of sepsis from an intrabdominal process such as ruptured appendicitis, intussusception, or malrotation with volvulus. Other causes of sepsis, such as meningitis or severe mastoiditis, both rare complications of otitis media, should be considered, although he does not appear severely ill. Acute myelogenous leukemia or other malignancies and illnesses associated with immunodeficiency can present with sepsis and chloromas in the middle ear that can be misconstrued as otitis media.
A chest radiograph demonstrated left lower lobe patchy opacities concerning for pneumonia. Rapid respiratory syncytial virus and influenza antigen test results were negative. Laboratory testing for general bloodwork was not obtained. He was administered a single dose of intramuscular ceftriaxone, prescribed a 5-day course of azithromycin, and discharged home. The child’s breathing gradually improved, but he continued to have subjective fevers. Two days later, he developed dark red urine. His mother brought him back to the outpatient clinic.
At the time of the ED visit, a diagnosis of community-acquired pneumonia was plausible given fever, mildly increased work of breathing, and an opacification on chest radiography. Most community-acquired pneumonia is caused by viruses; common bacterial causes for his age include Streptococcus pneumoniae and Moraxella catarrhalis. The first-line treatment for uncomplicated community-acquired pneumonia in children is amoxicillin, but this was appropriately avoided given his allergy.
The persistent fevers are surprising. The improvement in breathing corresponds to the treatment (and resolution) of community-acquired pneumonia. However, the development of dark urine does not. Red urine—in the absence of ingested pigments (such as those found in beets)—usually results from hematuria, hemoglobinuria, or myoglobinuria. Gross hematuria can originate from the kidneys to the urethral meatus. Abdominal masses, kidney trauma, or underlying kidney disease may all present with gross hematuria (or microscopic hematuria, seen only on urinalysis). The urine should be examined for the presence of heme, protein, and for evidence of infection; microscopy should be performed to examine for cellular casts and dysmorphic red cells. Tests of renal function, a comprehensive metabolic panel, evaluation of hematologic indexes, and assessments of inflammatory markers should be performed.
The child lived with his parents and had no siblings. He experienced no physical trauma, and there was no family history of kidney disease or hematuria. His father had a persistent cough and fever for 1 month, but recovered around the time the patient began to experience his initial symptoms. This was the patient’s third diagnosis of pneumonia. He had not traveled and was up to date with immunizations. He attended day care.
The fact that this is not the first episode of “pneumonia” raises important possibilities. The most likely one is that the child has had multiple viral infections; however, he could have an underlying primary immunodeficiency (PI) that predisposes him to recurrent infections. More severe PIs often present with recurrent sepsis, bacteremia, and failure to thrive, none of which were present in this case. Less severe PIs (such as selective IgA deficiency) could be possible. Another possibility is that these recurrent episodes of pneumonia are a relapsing and remitting noninfectious process, such as an antineutrophil cytoplasmic antibodies–associated vasculitis or anti–glomerular basement membrane disease. The patient’s father’s recent prolonged respiratory symptoms may be suggestive of pertussis or a “walking pneumonia” potentially caused by Mycoplasma or another atypical bacterium.
His temperature was 36.9 °C, heart rate 107 beats per minute, blood pressure was 106/67 mm Hg, and respiratory rate was 24 breaths per minute with oxygen saturation of 100% on ambient air. He was well appearing. His mucous membranes were moist, and oropharynx was clear. He had scleral icterus. The cardiopulmonary exam was normal. He had no significant lymphadenopathy, hepatosplenomegaly, or rashes.
The finding of jaundice is an important diagnostic pivot point, especially when combined with hematuria. The next step is determining if the jaundice is resulting from unconjugated or conjugated hyperbilirubinemia; the former most often stems from hemolysis or impairment in conjugation, while the latter results from intrahepatic or extrahepatic biliary defects. Tests for hepatobiliary injury including evaluations of alanine and aspartate aminotransferases and alkaline phosphatase, as well as for hepatic function such as tests of coagulation, should be performed.
The patient was referred to the ED and admitted for further evaluation. A complete blood count revealed a white blood cell (WBC) count of 10,700/µL (61% polymorphonuclear neutrophils, 30% lymphocytes, 5% monocytes, 3% eosinophils, 1% basophils), hemoglobin count was 10.3 g/dL (reticulocyte 2% with absolute reticulocyte count 58,400/μL), and platelet count was 265,000/µL. Components of the basic metabolic panel were within reference ranges except for a mildly elevated blood urea nitrogen level of 14 mg/dL with normal creatinine level of 0.3 mg/dL. Total protein was 6.7 g/dL (reference range, 6.4-8.3) and albumin 3.9 g/dL (reference range, 3.4-4.8). Alkaline phosphatase level was 188 U/L (reference range, 44-147), aspartate aminotransferase level 76 U/L (reference range, 0-40), and alanine aminotransferase level 12 U/L (reference range, 7-40). Total bilirubin level was 2.4 mg/dL (reference range, less than 1.5) with direct bilirubin level of 0.4 mg/dL. His C-reactive protein level was 1.5 mg/mL (reference range, 0-0.75). Creatinine kinase (CK) level was 2,550 U/L (reference range, 2-198). International Normalized Ratio (INR) was 1.0. Urinalysis was notable for 2+ proteinuria, large hemoglobin pigment, and 6 red blood cells per high power field (reference range, 0-4).
His blood urea nitrogen is elevated, reflecting either prerenal azotemia or increased absorption of nitrogenous products. Unconjugated hyperbilirubinemia may result from impaired hepatic bilirubin uptake (such as in heart failure or portosystemic shunts), impaired bilirubin conjugation (resulting from genetic conditions or drugs), or excess bilirubin production (such as in hemolysis); his anemia and lack of other evidence of hepatic dysfunction point to hemolysis as the etiology. The reticulocyte production index is approximately 1%, which suggests that an increase in erythrocyte generation is present but inadequate. This, however, does not mean that an erythrocyte production abnormality is present since reticulocytosis can be delayed in many cases of acute hemolytic anemia. It is also possible that the same hemolytic process is affecting mature and immature erythrocytes. A peripheral blood smear should be reviewed for evidence of intravascular hemolysis and testing for autoimmune hemolysis should be performed. Notably, his white blood cell and platelet counts are preserved, which makes a bone marrow–involved malignancy or infiltrative process less likely. The alkaline phosphatase elevation may result from either intrahepatic or extrahepatic biliopathy; bone damage is also possible. The elevation of aspartate aminotransferase, CK, and potassium, along with marked urinary heme pigment, may indicate muscle damage; the most common myositis in children is benign acute childhood myositis resulting from viral infection. However, the moderate level of CK elevation seen in this case is nonspecific and can result from many different etiologies. A metabolic myopathy, such as carnitine palmitoyltransferase II deficiency, can be made worse by metabolic stress and result in rhabdomyolysis; the presentations of inborn errors of metabolism are varied and a planned-out, stepwise approach in evaluation is fundamental.
Lactic acid dehydrogenase (LDH) level was 1,457 U/L (reference range, 140-280), and haptoglobin level was less than 6 mg/dL (reference range, 30-200). Peripheral blood smear demonstrated occasional atypical, reactive-appearing lymphocytes with red cell clumping and agglutination, as well as rare target, burr, and fragmented red cells. Test results for urine myoglobin were negative. Results of urine culture were negative. No blood culture was collected.
The elevated LDH, decreased haptoglobin, and findings on the peripheral blood smear confirm hemolysis. The clumping of erythrocytes can be artifactual in the preparation of peripheral smears, but when considered in the context of hemolysis, may be clinically important. Clumping of erythrocytes on the peripheral smear indicates the binding of a protein to antigens on the erythrocyte membrane; when this occurs below body temperature, this is consistent with the presence of a “cold agglutinin,” usually an IgM antibody directed at erythrocyte surface antigens that causes agglutination and destruction, especially in cooler areas of the body. This is a well-known complication of Mycoplasma pneumoniae infections as well as Epstein-Barr virus (EBV) infections; it may also occur with lymphoid malignancies or autoimmune disease.
Direct Coombs IgG test findings were negative, direct Coombs C3 test was positive, and direct Coombs polyspecific test was positive. M pneumoniae IgG antibody level was 1.4 mg/dL (reference ranges: <0.9, negative; 0.91-1.09, equivocal; >1.1, positive); M pneumoniae IgM level was 529 U/mL (reference range: <770, negative). EBV capsid IgM and IgG levels were undetectable. EBV nuclear antigen IgG level was also undetectable. EBV viral load was fewer than 10 copies/mL. Antinuclear antibodies (ANA) level was negative. General IgE and IgM levels were normal, at 11 and 81 mg/dL, respectively. Repeat complete blood count showed WBC of 7,800/µL, hemoglobin of 8.7 g/dL, and platelet count of 341,000/µL. The patient’s hemoglobin remained stable during hospitalization.
This directed testing is helpful in further classifying the patient’s hemolytic anemia. Autoimmune hemolytic anemias are classified into warm antibody–mediated, cold antibody–mediated, and mixed-type forms; drug-induced and alloimmune hemolytic anemias also occur. In addition, both systemic lupus erythematosus and antiphospholipid antibody syndrome can have hemolytic anemia with variable Coombs testing results; neither fit well in this case. The absence of red blood cell–directed IgG antibodies substantially decreases the likelihood of warm antibody–mediated hemolytic anemia. In cold antibody–mediated hemolytic anemia, antibodies bind to the erythrocyte membrane and then adhere to complement C3, which leads to both intravascular and extravascular hemolysis. Important types of cold antibody–mediated hemolytic anemia in children are primary and secondary cold agglutinin disease, along with paroxysmal cold hemoglobinuria. The Donath-Landsteiner test can be helpful in differentiating these conditions. Antibodies to Mycoplasma may be delayed in response to acute infection, and a child who is reinfected may only produce IgG antibodies. Given the patient’s clinical stability and previous health, the most likely diagnosis is Mycoplasma-induced cold antibody–mediated hemolytic anemia. It may be helpful to check convalescent titers to Mycoplasma in 2 to 4 weeks.
Donath-Landsteiner (D-L) antibody test results were positive. Medication-derived hemolytic anemia testing was conducted, but the presence of positive D-L antibody makes the test results inconclusive. This ultimately led to a diagnosis of paroxysmal cold hemoglobinuria (PCH), presumably triggered by a viral syndrome. Convalescent titers to Mycoplasma were not checked given clinical improvement. Because the patient’s hemoglobin was stable during hospitalization, he was not treated with steroids. His parents were counseled on avoiding cold temperatures for several days. Within 1 month, his hemoglobin had recovered without further evidence of hemolysis.
DISCUSSION
Hemolytic anemia refers to the accelerated destruction of red blood cells and can be further classified as acquired or hereditary.1 Hereditary conditions causing hemolytic anemia include enzymopathies (eg, glucose-6-phosphate dehydrogenase deficiency), hemoglobinopathies (eg, sickle cell disease), and membrane abnormalities (eg, hereditary spherocytosis). Acquired pathologies include microangiopathic hemolytic anemia (MAHA), anemias directly caused by certain infections such as malaria, and immune-mediated (Coombs-positive) hemolytic anemias.
MAHA can sometimes be life-threatening and is therefore important to identify quickly. In the right clinical context, such processes may be rapidly recognized by the presence of schistocytes on blood smear in addition to an elevated serum LDH level. Schistocytes suggest mechanical destruction of erythrocytes in the vasculature, the hallmark of MAHA. Important MAHAs include thrombocytopenic purpura, hemolytic-uremic syndrome, and disseminated intravascular coagulation. Though this patient did have a mildly elevated LDH, MAHA was less likely because there were no schistocytes on the blood smear.
Autoimmune hemolytic anemias (AIHAs) are another important subset of acquired hemolytic anemias. AIHAs occur when there is antibody-mediated destruction of erythrocytes. The direct Coombs test evaluates for antibody- or complement-coated erythrocytes. After administration of anti-IgG and anti-C3 serum, the test evaluates for agglutination of the red cells caused by attached antibodies or complement. Coombs-positive AIHA can also be categorized by the temperature of agglutination. “Warm” hemolysis often involves IgG autoantibodies (ie, warm agglutinins), while “cold” antibodies, usually IgM autoantibodies, bind at colder temperatures (0-4 °C) and activate complements, including C3. In this patient, the Coombs C3 was positive while the Coombs IgG was negative, which is more suggestive of a cold complement–mediated pathway.
Cold AIHA can be further categorized into primary cold agglutinin disease, secondary cold agglutinin disease, and PCH. Primary cold agglutinin disease is an autoimmune disorder that mostly occurs in adults. Secondary cold AIHA can often be triggered by bacterial infection (commonly M pneumoniae) or viruses including EBV, measles, and mumps.2 Medications, including penicillin and cephalosporins, can also be implicated. Secondary cold AIHA is also linked with autoimmune diseases, such as systemic lupus erythematosus and lymphoproliferative disorders. PCH can be identified with the unique presence of a specific autoantibody (ie, D-L autoantibody) that agglutinates at cold temperatures but dissociates on subsequent rewarming.3 Complement remains affixed and activates hemolysis.
The D-L antibody responsible for PCH is an IgG antibody to the P-antigen present on the erythrocyte surface. Since the Coombs test is conducted at normal temperature, it will be positive for the affixed complement but not for IgG. The underlying mechanism for PCH was proposed by Julius Donath, MD, and Karl Landsteiner, MD, in 1904 and is considered to be the first description of autoimmune disease being precipitated by antibodies.4 The D-L antibody test itself is uncommonly performed and somewhat difficult to interpret, particularly in adults, and may lead to false-negative results.5
PCH is an acquired, cold AIHA more common to children6,7 and may account for up to 33% of pediatric AIHA cases.8 Typical presentation is after an upper respiratory tract illness; however, the trigger is often not identified. Implicated triggers include a number of viruses.9 Clinical presentation includes findings of intravascular hemolysis similar to those in our patient. The pathogenic IgG autoantibody is polyclonal and is likely formed because of immune stimulation, which is consistent with the predominance of nonmalignant triggers of this disease process.10 Hemolysis and associated symptoms are often exacerbated with cold exposure; both typically resolve within 2 weeks. In recurrent cases, which are a minority, immunosuppression may be considered.10
PCH remains an often-understated cause of hemolytic anemia particularly in children. Lacking obvious pathognomonic clinical symptoms, it may be overlooked for other forms of AIHA or MAHA. However, with a structured approach to evaluation, as with this patient who had hematuria and jaundice, early diagnosis can prevent an unnecessarily extensive workup and can provide reassurance to patient and parents. By understanding the basic categories of hemolytic anemia, the relevant blood testing available, and interpretation of Coombs test results, clinicians can ensure that PCH is a diagnosis that is not left out in the cold.
KEY TEACHING POINTS
- Examination for schistocytes on a blood smear can help identify life-threatening causes of hemolytic anemia.
- Characterization of cold AIHA includes defining the underlying etiology as primary cold agglutinin disease, secondary cold agglutinin disease, or PCH.
- PCH is a cold AIHA that is an underrecognized cause of hemolytic anemia in children. The diagnosis of PCH is made by testing for the presence of the D-L antibody.
1. Dhaliwal G, Cornett PA, Tierney LM Jr. Hemolytic anemia. Am Fam Physician. 2004;69(11):2599-2606.
2. Djaldetti M. Paroxysmal cold hemoglobinuria. CRC Crit Rev Clin Lab Sci. 1978;9(1):49-83. https://doi.org/10.3109/10408367809150915
3. Levine P, Celano MJ, Falkowski F. The specificity of the antibody in paroxysmal cold hemoglobinuria (P.C.H.). Transfusion. 1963;3(4):278-280. https://doi.org/10.1111/j.1537-2995.1963.tb04643.x
4. Donath J, Landsteiner K. Uber Paroxysmale Hamoglobinurie. Munch Med Wochenschr. 1904;51:1590-1593
5. Zeller MP, Arnold DM, Al Habsi K, et al. Paroxysmal cold hemoglobinuria: a difficult diagnosis in adult patients. Transfusion. 2017;57(1):137-143. https://doi.org/10.1111/trf.13888
6. Göttsche B, Salama A, Mueller-Eckhardt C. Donath-Landsteiner autoimmune hemolytic anemia in children. a study of 22 cases. Vox Sang. 1990;58(4):281-286. https://doi.org/10.1111/j.1423-0410.1990.tb05000.x
7. Sokol RJ, Booker DJ, Stamps R. Erythropoiesis: paroxysmal cold haemoglobinuria: a clinico-pathological study of patients with a positive Donath-Landsteiner test. Hematology. 1999;4(2):137-164. https://doi.org/10.1080/10245332.1999.11746439
8. Petz LD. Cold antibody autoimmune hemolytic anemias. Blood Rev. 2008;22(1):1-15. https://doi.org/10.1016/j.blre.2007.08.002
9. Leibrandt R, Angelino K, Vizel-Schwartz M, Shapira I. Paroxysmal cold hemoglobinuria in an adult with respiratory syncytial virus. Case Rep Hematol. 2018;2018:1-3. https://doi.org/10.1155/2018/7586719
10. Gertz MA. Management of cold haemolytic syndrome. Br J Haematol. 2007;138(4):422-429. https://doi.org/10.1111/j.1365-2141.2007.06664.x
1. Dhaliwal G, Cornett PA, Tierney LM Jr. Hemolytic anemia. Am Fam Physician. 2004;69(11):2599-2606.
2. Djaldetti M. Paroxysmal cold hemoglobinuria. CRC Crit Rev Clin Lab Sci. 1978;9(1):49-83. https://doi.org/10.3109/10408367809150915
3. Levine P, Celano MJ, Falkowski F. The specificity of the antibody in paroxysmal cold hemoglobinuria (P.C.H.). Transfusion. 1963;3(4):278-280. https://doi.org/10.1111/j.1537-2995.1963.tb04643.x
4. Donath J, Landsteiner K. Uber Paroxysmale Hamoglobinurie. Munch Med Wochenschr. 1904;51:1590-1593
5. Zeller MP, Arnold DM, Al Habsi K, et al. Paroxysmal cold hemoglobinuria: a difficult diagnosis in adult patients. Transfusion. 2017;57(1):137-143. https://doi.org/10.1111/trf.13888
6. Göttsche B, Salama A, Mueller-Eckhardt C. Donath-Landsteiner autoimmune hemolytic anemia in children. a study of 22 cases. Vox Sang. 1990;58(4):281-286. https://doi.org/10.1111/j.1423-0410.1990.tb05000.x
7. Sokol RJ, Booker DJ, Stamps R. Erythropoiesis: paroxysmal cold haemoglobinuria: a clinico-pathological study of patients with a positive Donath-Landsteiner test. Hematology. 1999;4(2):137-164. https://doi.org/10.1080/10245332.1999.11746439
8. Petz LD. Cold antibody autoimmune hemolytic anemias. Blood Rev. 2008;22(1):1-15. https://doi.org/10.1016/j.blre.2007.08.002
9. Leibrandt R, Angelino K, Vizel-Schwartz M, Shapira I. Paroxysmal cold hemoglobinuria in an adult with respiratory syncytial virus. Case Rep Hematol. 2018;2018:1-3. https://doi.org/10.1155/2018/7586719
10. Gertz MA. Management of cold haemolytic syndrome. Br J Haematol. 2007;138(4):422-429. https://doi.org/10.1111/j.1365-2141.2007.06664.x
© 2020 Society of Hospital Medicine