User login
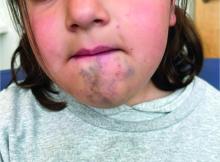
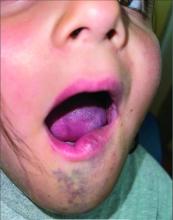
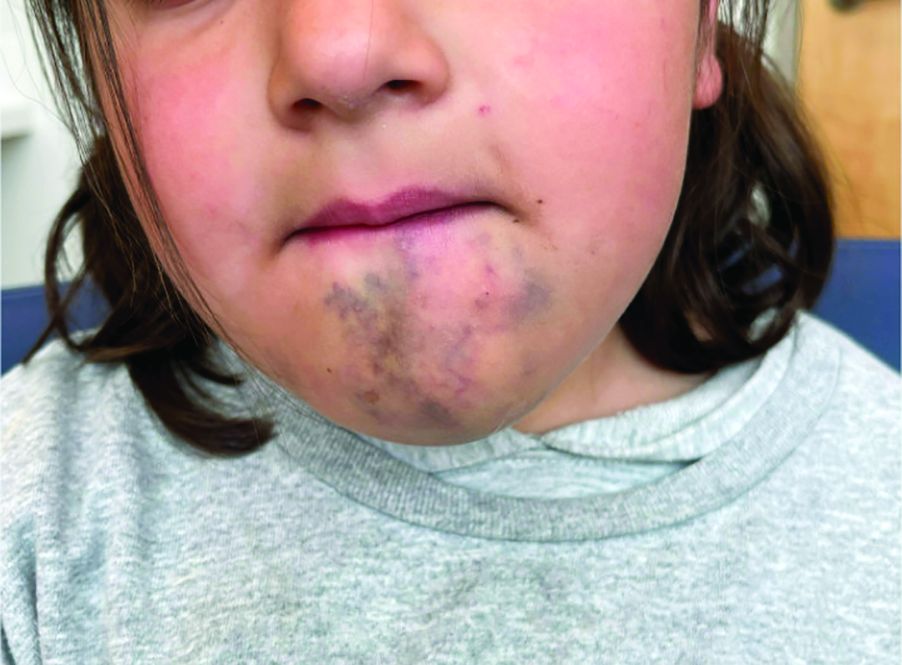
A 6-Year-Old Female Presents With a Bruise-Like Lesion on the Lip, Tongue, and Chin Area Present Since Birth
Diagnosis: Venous Malformation
Although present at birth, they are not always clinically evident early in life. They also tend to grow with the child without spontaneous regression, causing potential cosmetic concerns or complications from impingement on surrounding tissue.
Venous malformations appear with a bluish color appearing beneath the skin and can vary significantly in size and severity. Venous malformations are compressible and characterized by low to stagnant blood flow, which can spontaneously thrombose. Clinically, this may cause pain, swelling, skin changes, tissue and limb overgrowth, or functional impairment depending on location and size.
Venous malformations result from disorganized angiogenesis secondary to sporadic mutations in somatic cells. The most common implicated gene is TEK, a receptor tyrosine kinase. PIK3CA has also been involved. Both genes are involved in the PI3K/AKT/mTOR pathway, which regulates cell growth, proliferation, and angiogenesis. In venous endothelial cells, abnormal angiogenesis and vessel maturation may lead to venous malformation formation. Dysplastic vessels frequently separate from normal veins but may be contiguous with the deep venous system.
Diagnosis involves clinical history and physical examination. Imaging with ultrasound and magnetic resonance imaging (MRI) may be utilized. While ultrasound may be preferred for superficial venous malformations, MRI or MRI with MR angiography (MRA) is the preferred method for venous malformation assessment. Genetic testing may be appropriate for complex malformations, as classification of lesions by underlying mutation may allow targeted therapy.
This patient’s past MRI and MRA findings were consistent with a venous malformation.
Treatment
Venous malformations rarely regress spontaneously. Treatment is required if venous malformations are symptomatic, which may include pain, swelling, deformity, thrombosis, or interference with daily activities of living. Treatment plans require consideration of patient goals of care. The main categories of therapy are embolization/sclerotherapy, surgical resection, and molecular targeted therapy.
Sclerotherapy is a well-tolerated and efficacious first-line therapy. It can be used as either nonsurgical curative therapy or preoperative adjunct therapy to minimize blood loss before surgical resection. While surgical resection may cause scarring, multimodal approaches with sclerotherapy or laser therapy can decrease complications. Molecular therapies aim to reduce vascular proliferation and symptoms. Referral to hematology/oncology for evaluation and consideration of chemotherapeutic agents may be required. Sirolimus has been shown in mice models to inhibit an endothelial cell tyrosine kinase receptor that plays a role in venous malformation growth. Multiple studies have proved its efficacy in managing complicated vascular anomalies, including venous malformations. Alpelisib is an inhibitor of PI3KCA, which is part of the pathway that contributes to venous malformation formation. Dactolisib, a dual inhibitor of the PI3KA and mTOR pathways, and rebastinib, a TEK inhibitor, are being investigated.
Differential Diagnoses
The differential diagnosis includes dermal melanocytosis, nevus of Ota, hemangioma of infancy, and ashy dermatosis. In addition, venous malformations can be part of more complex vascular malformations.
Dermal melanocytosis, also known as Mongolian spots, are blue-gray patches of discoloration on the skin that appear at birth or shortly after. They result from the arrest of dermal melanocytes in the dermis during fetal life and tissue modeling. They are commonly observed in those of Asian or African descent with darker skin types. Most often, they are located in the lumbar or sacral-gluteal region. Unlike venous malformations, they are benign and do not involve vascular abnormalities. They typically fade over time.
Nevus of Ota is a benign congenital condition that presents with blue-gray or brown patches of pigmentation on the skin around the eyes, cheeks, and forehead. They are dermal melanocytes with a speckled instead of uniform appearance. Nevus of Ota primarily affects individuals of Asian descent and typically presents in the trigeminal nerve distribution region. Treatment can be done to minimize deformity, generally with pigmented laser surgery.
Hemangiomas of infancy are common benign tumors of infancy caused by endothelial cell proliferation. They are characterized by rapid growth followed by spontaneous involution within the first year of life and for several years. Hemangiomas can be superficial, deep, or mixed with features of both superficial and deep. Superficial hemangiomas present as raised, lobulated, and bright red while deep hemangiomas present as a bluish-hued nodule, plaque, or tumor. They are diagnosed clinically but skin biopsies and imaging can confirm the suspected diagnosis. While hemangiomas may self-resolve, complicated hemangiomas can be treated with topical timolol, oral propranolol, topical and intralesional corticosteroids, pulsed-dye laser, and surgical resection.
Ashy dermatosis is a term for asymptomatic, gray-blue or ashy patches distributed symmetrically on the trunk, head, neck, and upper extremities. It primarily affects individuals with darker skin types (Fitzpatrick III-V), and is more common in patients with Hispanic, Asian, or African backgrounds. The direct cause of ashy dermatosis is unknown but it is thought to be linked to drug ingestion, genetics, infection, and immune-mediated mechanisms. The general treatment includes topical corticosteroids, clofazimine, topical calcineurin inhibitors, oral dapsone, phototherapy, topical retinoids, or isotretinoin to reduce inflammation and pigmentation.
Danny Lee and Samuel Le serve as research fellows and Jolina Bui as research associate in the Pediatric Dermatology Division of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Eichenfield is Distinguished Professor of Dermatology and Pediatrics and Vice-Chair of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. The authors have no relevant financial disclosures.
Suggested Reading
Agarwal P, Patel BC. Nevus of Ota and Ito. [Updated 2023 Jul 10]. In: StatPearls [Internet]. StatPearls Publishing; 2024.
Dompmartin A et al. The VASCERN-VASCA Working Group Diagnostic and Management Pathways for Venous Malformations. J Vasc Anom (Phila). 2023 Mar 23;4(2):e064.
Dompmartin A et al. Venous malformation: Update on aetiopathogenesis, diagnosis and management. Phlebology. 2010 Oct;25(5):224-235.
Gupta D, Thappa DM. Mongolian spots. Indian J Dermatol Venereol Leprol. 2013 Jul-Aug;79(4):469-478.
Krowchuk DP et al. Clinical Practice Guideline for the Management of Infantile Hemangiomas. Pediatrics. 2019 Jan;143(1):e20183475.
Nguyen K, Khachemoune A. Ashy dermatosis: A review. Dermatol Online J. 2019 May 15;25(5):13030/qt44f462s8.
Patel ND, Chong AT et al. Venous Malformations. Semin Intervent Radiol. 2022 Dec 20;39(5):498-507.
Diagnosis: Venous Malformation
Although present at birth, they are not always clinically evident early in life. They also tend to grow with the child without spontaneous regression, causing potential cosmetic concerns or complications from impingement on surrounding tissue.
Venous malformations appear with a bluish color appearing beneath the skin and can vary significantly in size and severity. Venous malformations are compressible and characterized by low to stagnant blood flow, which can spontaneously thrombose. Clinically, this may cause pain, swelling, skin changes, tissue and limb overgrowth, or functional impairment depending on location and size.
Venous malformations result from disorganized angiogenesis secondary to sporadic mutations in somatic cells. The most common implicated gene is TEK, a receptor tyrosine kinase. PIK3CA has also been involved. Both genes are involved in the PI3K/AKT/mTOR pathway, which regulates cell growth, proliferation, and angiogenesis. In venous endothelial cells, abnormal angiogenesis and vessel maturation may lead to venous malformation formation. Dysplastic vessels frequently separate from normal veins but may be contiguous with the deep venous system.
Diagnosis involves clinical history and physical examination. Imaging with ultrasound and magnetic resonance imaging (MRI) may be utilized. While ultrasound may be preferred for superficial venous malformations, MRI or MRI with MR angiography (MRA) is the preferred method for venous malformation assessment. Genetic testing may be appropriate for complex malformations, as classification of lesions by underlying mutation may allow targeted therapy.
This patient’s past MRI and MRA findings were consistent with a venous malformation.
Treatment
Venous malformations rarely regress spontaneously. Treatment is required if venous malformations are symptomatic, which may include pain, swelling, deformity, thrombosis, or interference with daily activities of living. Treatment plans require consideration of patient goals of care. The main categories of therapy are embolization/sclerotherapy, surgical resection, and molecular targeted therapy.
Sclerotherapy is a well-tolerated and efficacious first-line therapy. It can be used as either nonsurgical curative therapy or preoperative adjunct therapy to minimize blood loss before surgical resection. While surgical resection may cause scarring, multimodal approaches with sclerotherapy or laser therapy can decrease complications. Molecular therapies aim to reduce vascular proliferation and symptoms. Referral to hematology/oncology for evaluation and consideration of chemotherapeutic agents may be required. Sirolimus has been shown in mice models to inhibit an endothelial cell tyrosine kinase receptor that plays a role in venous malformation growth. Multiple studies have proved its efficacy in managing complicated vascular anomalies, including venous malformations. Alpelisib is an inhibitor of PI3KCA, which is part of the pathway that contributes to venous malformation formation. Dactolisib, a dual inhibitor of the PI3KA and mTOR pathways, and rebastinib, a TEK inhibitor, are being investigated.
Differential Diagnoses
The differential diagnosis includes dermal melanocytosis, nevus of Ota, hemangioma of infancy, and ashy dermatosis. In addition, venous malformations can be part of more complex vascular malformations.
Dermal melanocytosis, also known as Mongolian spots, are blue-gray patches of discoloration on the skin that appear at birth or shortly after. They result from the arrest of dermal melanocytes in the dermis during fetal life and tissue modeling. They are commonly observed in those of Asian or African descent with darker skin types. Most often, they are located in the lumbar or sacral-gluteal region. Unlike venous malformations, they are benign and do not involve vascular abnormalities. They typically fade over time.
Nevus of Ota is a benign congenital condition that presents with blue-gray or brown patches of pigmentation on the skin around the eyes, cheeks, and forehead. They are dermal melanocytes with a speckled instead of uniform appearance. Nevus of Ota primarily affects individuals of Asian descent and typically presents in the trigeminal nerve distribution region. Treatment can be done to minimize deformity, generally with pigmented laser surgery.
Hemangiomas of infancy are common benign tumors of infancy caused by endothelial cell proliferation. They are characterized by rapid growth followed by spontaneous involution within the first year of life and for several years. Hemangiomas can be superficial, deep, or mixed with features of both superficial and deep. Superficial hemangiomas present as raised, lobulated, and bright red while deep hemangiomas present as a bluish-hued nodule, plaque, or tumor. They are diagnosed clinically but skin biopsies and imaging can confirm the suspected diagnosis. While hemangiomas may self-resolve, complicated hemangiomas can be treated with topical timolol, oral propranolol, topical and intralesional corticosteroids, pulsed-dye laser, and surgical resection.
Ashy dermatosis is a term for asymptomatic, gray-blue or ashy patches distributed symmetrically on the trunk, head, neck, and upper extremities. It primarily affects individuals with darker skin types (Fitzpatrick III-V), and is more common in patients with Hispanic, Asian, or African backgrounds. The direct cause of ashy dermatosis is unknown but it is thought to be linked to drug ingestion, genetics, infection, and immune-mediated mechanisms. The general treatment includes topical corticosteroids, clofazimine, topical calcineurin inhibitors, oral dapsone, phototherapy, topical retinoids, or isotretinoin to reduce inflammation and pigmentation.
Danny Lee and Samuel Le serve as research fellows and Jolina Bui as research associate in the Pediatric Dermatology Division of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Eichenfield is Distinguished Professor of Dermatology and Pediatrics and Vice-Chair of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. The authors have no relevant financial disclosures.
Suggested Reading
Agarwal P, Patel BC. Nevus of Ota and Ito. [Updated 2023 Jul 10]. In: StatPearls [Internet]. StatPearls Publishing; 2024.
Dompmartin A et al. The VASCERN-VASCA Working Group Diagnostic and Management Pathways for Venous Malformations. J Vasc Anom (Phila). 2023 Mar 23;4(2):e064.
Dompmartin A et al. Venous malformation: Update on aetiopathogenesis, diagnosis and management. Phlebology. 2010 Oct;25(5):224-235.
Gupta D, Thappa DM. Mongolian spots. Indian J Dermatol Venereol Leprol. 2013 Jul-Aug;79(4):469-478.
Krowchuk DP et al. Clinical Practice Guideline for the Management of Infantile Hemangiomas. Pediatrics. 2019 Jan;143(1):e20183475.
Nguyen K, Khachemoune A. Ashy dermatosis: A review. Dermatol Online J. 2019 May 15;25(5):13030/qt44f462s8.
Patel ND, Chong AT et al. Venous Malformations. Semin Intervent Radiol. 2022 Dec 20;39(5):498-507.
Diagnosis: Venous Malformation
Although present at birth, they are not always clinically evident early in life. They also tend to grow with the child without spontaneous regression, causing potential cosmetic concerns or complications from impingement on surrounding tissue.
Venous malformations appear with a bluish color appearing beneath the skin and can vary significantly in size and severity. Venous malformations are compressible and characterized by low to stagnant blood flow, which can spontaneously thrombose. Clinically, this may cause pain, swelling, skin changes, tissue and limb overgrowth, or functional impairment depending on location and size.
Venous malformations result from disorganized angiogenesis secondary to sporadic mutations in somatic cells. The most common implicated gene is TEK, a receptor tyrosine kinase. PIK3CA has also been involved. Both genes are involved in the PI3K/AKT/mTOR pathway, which regulates cell growth, proliferation, and angiogenesis. In venous endothelial cells, abnormal angiogenesis and vessel maturation may lead to venous malformation formation. Dysplastic vessels frequently separate from normal veins but may be contiguous with the deep venous system.
Diagnosis involves clinical history and physical examination. Imaging with ultrasound and magnetic resonance imaging (MRI) may be utilized. While ultrasound may be preferred for superficial venous malformations, MRI or MRI with MR angiography (MRA) is the preferred method for venous malformation assessment. Genetic testing may be appropriate for complex malformations, as classification of lesions by underlying mutation may allow targeted therapy.
This patient’s past MRI and MRA findings were consistent with a venous malformation.
Treatment
Venous malformations rarely regress spontaneously. Treatment is required if venous malformations are symptomatic, which may include pain, swelling, deformity, thrombosis, or interference with daily activities of living. Treatment plans require consideration of patient goals of care. The main categories of therapy are embolization/sclerotherapy, surgical resection, and molecular targeted therapy.
Sclerotherapy is a well-tolerated and efficacious first-line therapy. It can be used as either nonsurgical curative therapy or preoperative adjunct therapy to minimize blood loss before surgical resection. While surgical resection may cause scarring, multimodal approaches with sclerotherapy or laser therapy can decrease complications. Molecular therapies aim to reduce vascular proliferation and symptoms. Referral to hematology/oncology for evaluation and consideration of chemotherapeutic agents may be required. Sirolimus has been shown in mice models to inhibit an endothelial cell tyrosine kinase receptor that plays a role in venous malformation growth. Multiple studies have proved its efficacy in managing complicated vascular anomalies, including venous malformations. Alpelisib is an inhibitor of PI3KCA, which is part of the pathway that contributes to venous malformation formation. Dactolisib, a dual inhibitor of the PI3KA and mTOR pathways, and rebastinib, a TEK inhibitor, are being investigated.
Differential Diagnoses
The differential diagnosis includes dermal melanocytosis, nevus of Ota, hemangioma of infancy, and ashy dermatosis. In addition, venous malformations can be part of more complex vascular malformations.
Dermal melanocytosis, also known as Mongolian spots, are blue-gray patches of discoloration on the skin that appear at birth or shortly after. They result from the arrest of dermal melanocytes in the dermis during fetal life and tissue modeling. They are commonly observed in those of Asian or African descent with darker skin types. Most often, they are located in the lumbar or sacral-gluteal region. Unlike venous malformations, they are benign and do not involve vascular abnormalities. They typically fade over time.
Nevus of Ota is a benign congenital condition that presents with blue-gray or brown patches of pigmentation on the skin around the eyes, cheeks, and forehead. They are dermal melanocytes with a speckled instead of uniform appearance. Nevus of Ota primarily affects individuals of Asian descent and typically presents in the trigeminal nerve distribution region. Treatment can be done to minimize deformity, generally with pigmented laser surgery.
Hemangiomas of infancy are common benign tumors of infancy caused by endothelial cell proliferation. They are characterized by rapid growth followed by spontaneous involution within the first year of life and for several years. Hemangiomas can be superficial, deep, or mixed with features of both superficial and deep. Superficial hemangiomas present as raised, lobulated, and bright red while deep hemangiomas present as a bluish-hued nodule, plaque, or tumor. They are diagnosed clinically but skin biopsies and imaging can confirm the suspected diagnosis. While hemangiomas may self-resolve, complicated hemangiomas can be treated with topical timolol, oral propranolol, topical and intralesional corticosteroids, pulsed-dye laser, and surgical resection.
Ashy dermatosis is a term for asymptomatic, gray-blue or ashy patches distributed symmetrically on the trunk, head, neck, and upper extremities. It primarily affects individuals with darker skin types (Fitzpatrick III-V), and is more common in patients with Hispanic, Asian, or African backgrounds. The direct cause of ashy dermatosis is unknown but it is thought to be linked to drug ingestion, genetics, infection, and immune-mediated mechanisms. The general treatment includes topical corticosteroids, clofazimine, topical calcineurin inhibitors, oral dapsone, phototherapy, topical retinoids, or isotretinoin to reduce inflammation and pigmentation.
Danny Lee and Samuel Le serve as research fellows and Jolina Bui as research associate in the Pediatric Dermatology Division of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Eichenfield is Distinguished Professor of Dermatology and Pediatrics and Vice-Chair of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. The authors have no relevant financial disclosures.
Suggested Reading
Agarwal P, Patel BC. Nevus of Ota and Ito. [Updated 2023 Jul 10]. In: StatPearls [Internet]. StatPearls Publishing; 2024.
Dompmartin A et al. The VASCERN-VASCA Working Group Diagnostic and Management Pathways for Venous Malformations. J Vasc Anom (Phila). 2023 Mar 23;4(2):e064.
Dompmartin A et al. Venous malformation: Update on aetiopathogenesis, diagnosis and management. Phlebology. 2010 Oct;25(5):224-235.
Gupta D, Thappa DM. Mongolian spots. Indian J Dermatol Venereol Leprol. 2013 Jul-Aug;79(4):469-478.
Krowchuk DP et al. Clinical Practice Guideline for the Management of Infantile Hemangiomas. Pediatrics. 2019 Jan;143(1):e20183475.
Nguyen K, Khachemoune A. Ashy dermatosis: A review. Dermatol Online J. 2019 May 15;25(5):13030/qt44f462s8.
Patel ND, Chong AT et al. Venous Malformations. Semin Intervent Radiol. 2022 Dec 20;39(5):498-507.
A 6-year-old girl presents with a bruise-like lesion on the lip, tongue, and chin area present since birth. The family states that her tongue has been increasing in size and is painful. On physical exam, she presents with left lower mucosal lip fullness and an overlying violaceous hue extending into the oral mucosa and onto the left tongue. The left portion of the dorsal tongue displays an increased thickness and bluish discoloration and there is a pink, smooth papule on the left anterolateral tongue.
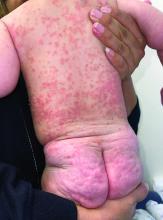
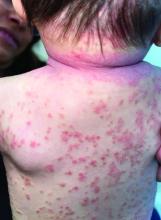
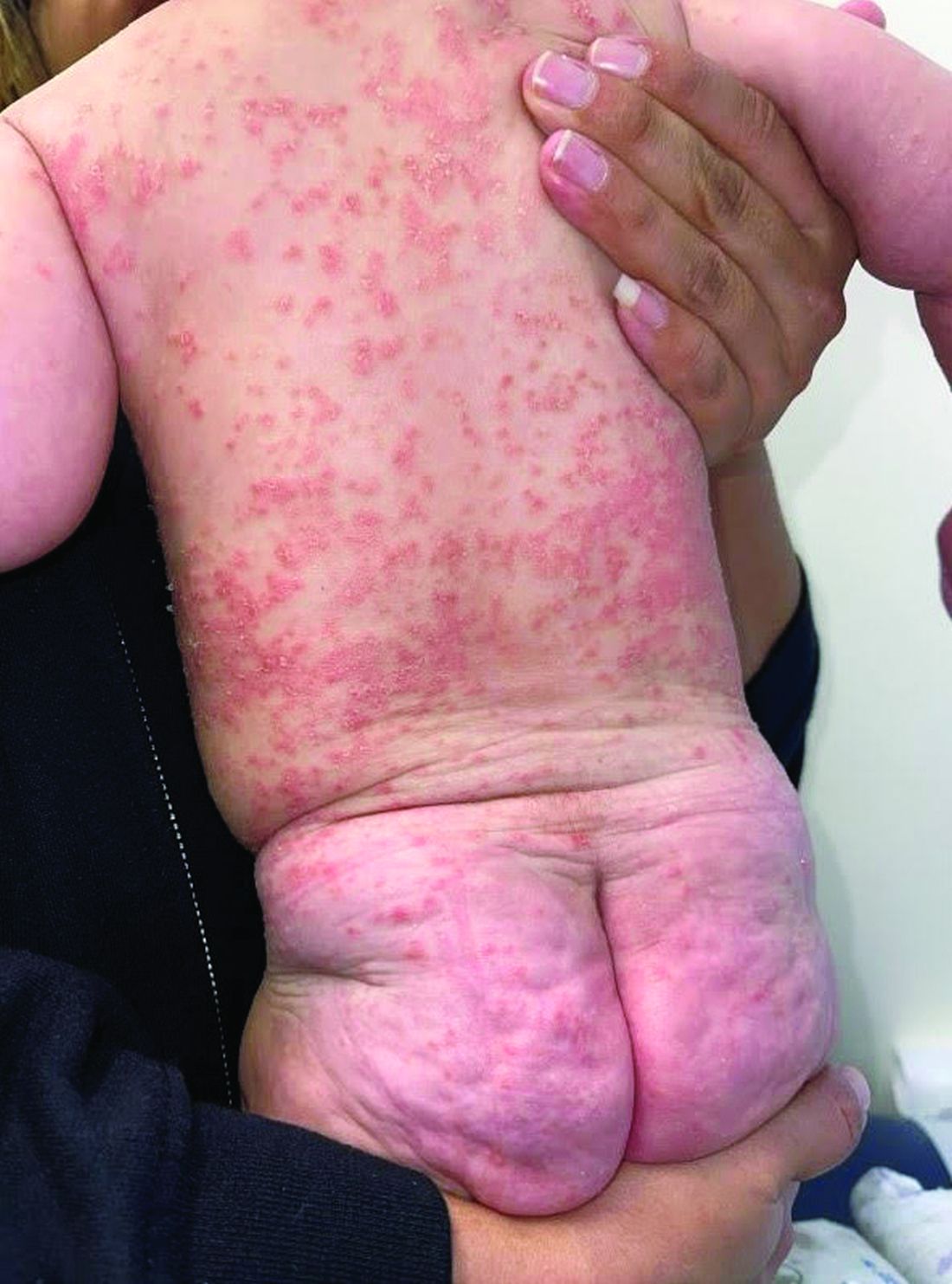
A 4-month-old male was referred for a 3-week history of an itchy generalized rash that started on the neck
Diagnosis: Infection-induced psoriasis (guttate-type, induced by streptococcal intertrigo)
Psoriasis is a chronic inflammatory disorder characterized by well-defined, scaly, erythematous plaques. Guttate psoriasis is a distinct variant of psoriasis that is more common in children and adolescents. Guttate psoriasis usually presents with multiple, scattered, small, drop-like (“guttate”), scaly, erythematous papules and plaques.
The pathophysiology of psoriasis involves an interplay between genetic and environmental factors. Guttate psoriasis is a chronic T-cell–mediated inflammatory disease in which there is an altered balance between T-helper-1 (TH1) and TH2 cells, transcription factor genes, and their products. HLA B-13, B-17, and Cw6 are human leukocyte antigen alleles implicated in genetic susceptibility. It is hypothesized that streptococcal infection precipitates guttate psoriasis by streptococcal superantigen–driven activation of cutaneous lymphocyte-associated antigen (CLA)–positive lymphocytes. It has been shown that streptococcal exotoxins and streptococcal M proteins act as superantigens.
Diagnosis is often made clinically based on characteristic physical findings and a possible preceding history of streptococcal infection. In patients with streptococcal infection, culture from an appropriate site and measurement of serum antistreptococcal antibody titers (for example, anti-DNase, antihyaluronidase and antistreptolysin-O) can help. A skin biopsy is usually not necessary but may be considered.
This patient presented with intertrigo of the neck and axillae at the time of presentation with the papulosquamous rash. Culture of the intertrigo yielded 4+ Group A beta streptococcus.
Treatment
Although there is currently no cure for guttate psoriasis, various treatment options can relieve symptoms and clear skin lesions, and infection-triggered lesions may remit, usually within several months. However, guttate psoriasis may persist and progress to chronic plaque psoriasis. Many treatment options are based mainly on clinical trials targeted for plaque psoriasis treatment.
For mild psoriasis, topical corticosteroids are first-line treatment. Other topical steroids include vitamin D analogs (calcipotriene), topical retinoids (tazarotene), topical calcineurin inhibitors (tacrolimus and pimecrolimus), and newer non-steroidal anti-inflammatory agents (roflumilast or tapinarof), neither approved yet in this young age group. In more severe cases, phototherapy with UVB light, traditional systemic immunosuppressive agents (methotrexate, cyclosporine) or targeted biologic therapies may be considered.
Differential Diagnosis
The differential diagnosis may include generalized intertrigo, pityriasis rubra pilaris, tinea corporis, atopic dermatitis, and staphylococcal scalded skin syndrome. Guttate psoriasis can be distinguished by history and physical exam. Further studies such as potassium hydroxide (KOH) scrapings may be helpful in ruling out the other disorders.
Intertrigo is an inflammatory condition of the flexural surfaces irritated by warm temperatures, friction, moisture, and poor ventilation that is commonly associated with Candida infection and/or streptococcal infection. Candidal intertrigo can present with erythematous patches or plaques in an intertriginous area that may develop erosions, macerations, fissures, crust, and weeping. Satellite papules and pustules are pathognomonic for Candida species. Streptococcal intertrigo usually presents with bright red color and may be painful or pruritic. Perianal streptococcal infection is reported as a trigger of guttate psoriasis in pediatric patients.
Pityriasis rubra pilaris is a rare inflammatory papulosquamous disorder with an unknown etiology. Red-orange papules and plaques, hyperkeratotic follicular papules, and palmoplantar hyperkeratosis are primary features. Diagnosis is based on clinical and histopathology. Pityriasis rubra pilaris is self-limited and asymptomatic in many cases. Treatment may not be required, but combination therapy with topical agents includes emollients, keratolytic agents (for example, urea, salicylic acid, alpha-hydroxy acids), topical corticosteroids, tazarotene, and topical calcineurin inhibitors. Systemic agents include oral retinoids and methotrexate.
Atopic dermatitis is a chronic inflammatory skin disease that involves genetic and environmental factors, leading to abnormalities in the epidermis and the immune system presenting with its typical morphology and distribution. The morphology of eczematous lesions is distinct from papulosquamous lesions of psoriasis.
Staphylococcal scalded skin syndrome is a toxin-mediated skin disorder which presents with denuded, peeling skin due to epidermolytic exotoxin producing Staphylococcus species. Fever, erythematous rash, malaise, skin pain, and irritability presents initially. Progressive desquamation with accentuation in folds is typical, with progression usually within 1-2 days. Systemic antibiotics covering Staphylococcus should be administered early. Emollients and nonadherent dressings should be applied to affected areas to promote healing. Supportive care includes dehydration management, temperature regulation, and nutrition. Skin desquamation usually occurs within 5 days with resolution within 2 weeks.
This infant displayed streptococcal intertrigo which triggered an early presentation of guttate psoriasis. The patient was managed with completion of a course of oral cephalexin, midstrength topical corticosteroids to the truncal lesions, and mild topical corticosteroids to the face and diaper area with good clinical response.
Danny Lee and Samuel Le serve as research fellows in the Pediatric Dermatology Division of the Department of Dermatology at the University of California San Diego and Rady Children’s Hospital, San Diego. Dr. Eichenfield is Distinguished Professor of Dermatology and Pediatrics and Vice-Chair of the Department of Dermatology at the University of California San Diego and Rady Children’s Hospital, San Diego. The authors have no relevant financial disclosures.
Suggested Reading
Leung AK et al. Childhood guttate psoriasis: An updated review. Drugs Context. 2023 Oct 23:12:2023-8-2. doi: 10.7573/dic.2023-8-2.
Galili E et al. New-onset guttate psoriasis: A long-term follow-up study. Dermatology. 2023;239(2):188-194. doi: 10.1159/000527737.
Duffin KC et al. Advances and controversies in our understanding of guttate and plaque psoriasis. J Rheumatol. 2023 Nov;50(Suppl 2):4-7. doi: 10.3899/jrheum.2023-0500.
Saleh D, Tanner LS. Guttate Psoriasis. [Updated 2023 Jul 31]. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2023 Jan-. Available from: www.ncbi.nlm.nih.gov/books/NBK482498/
Dupire G et al. Antistreptococcal interventions for guttate and chronic plaque psoriasis. Cochrane Database Syst Rev. 2019 Mar 5;3(3):CD011571. doi: 10.1002/14651858.CD011571.pub2.
Diagnosis: Infection-induced psoriasis (guttate-type, induced by streptococcal intertrigo)
Psoriasis is a chronic inflammatory disorder characterized by well-defined, scaly, erythematous plaques. Guttate psoriasis is a distinct variant of psoriasis that is more common in children and adolescents. Guttate psoriasis usually presents with multiple, scattered, small, drop-like (“guttate”), scaly, erythematous papules and plaques.
The pathophysiology of psoriasis involves an interplay between genetic and environmental factors. Guttate psoriasis is a chronic T-cell–mediated inflammatory disease in which there is an altered balance between T-helper-1 (TH1) and TH2 cells, transcription factor genes, and their products. HLA B-13, B-17, and Cw6 are human leukocyte antigen alleles implicated in genetic susceptibility. It is hypothesized that streptococcal infection precipitates guttate psoriasis by streptococcal superantigen–driven activation of cutaneous lymphocyte-associated antigen (CLA)–positive lymphocytes. It has been shown that streptococcal exotoxins and streptococcal M proteins act as superantigens.
Diagnosis is often made clinically based on characteristic physical findings and a possible preceding history of streptococcal infection. In patients with streptococcal infection, culture from an appropriate site and measurement of serum antistreptococcal antibody titers (for example, anti-DNase, antihyaluronidase and antistreptolysin-O) can help. A skin biopsy is usually not necessary but may be considered.
This patient presented with intertrigo of the neck and axillae at the time of presentation with the papulosquamous rash. Culture of the intertrigo yielded 4+ Group A beta streptococcus.
Treatment
Although there is currently no cure for guttate psoriasis, various treatment options can relieve symptoms and clear skin lesions, and infection-triggered lesions may remit, usually within several months. However, guttate psoriasis may persist and progress to chronic plaque psoriasis. Many treatment options are based mainly on clinical trials targeted for plaque psoriasis treatment.
For mild psoriasis, topical corticosteroids are first-line treatment. Other topical steroids include vitamin D analogs (calcipotriene), topical retinoids (tazarotene), topical calcineurin inhibitors (tacrolimus and pimecrolimus), and newer non-steroidal anti-inflammatory agents (roflumilast or tapinarof), neither approved yet in this young age group. In more severe cases, phototherapy with UVB light, traditional systemic immunosuppressive agents (methotrexate, cyclosporine) or targeted biologic therapies may be considered.
Differential Diagnosis
The differential diagnosis may include generalized intertrigo, pityriasis rubra pilaris, tinea corporis, atopic dermatitis, and staphylococcal scalded skin syndrome. Guttate psoriasis can be distinguished by history and physical exam. Further studies such as potassium hydroxide (KOH) scrapings may be helpful in ruling out the other disorders.
Intertrigo is an inflammatory condition of the flexural surfaces irritated by warm temperatures, friction, moisture, and poor ventilation that is commonly associated with Candida infection and/or streptococcal infection. Candidal intertrigo can present with erythematous patches or plaques in an intertriginous area that may develop erosions, macerations, fissures, crust, and weeping. Satellite papules and pustules are pathognomonic for Candida species. Streptococcal intertrigo usually presents with bright red color and may be painful or pruritic. Perianal streptococcal infection is reported as a trigger of guttate psoriasis in pediatric patients.
Pityriasis rubra pilaris is a rare inflammatory papulosquamous disorder with an unknown etiology. Red-orange papules and plaques, hyperkeratotic follicular papules, and palmoplantar hyperkeratosis are primary features. Diagnosis is based on clinical and histopathology. Pityriasis rubra pilaris is self-limited and asymptomatic in many cases. Treatment may not be required, but combination therapy with topical agents includes emollients, keratolytic agents (for example, urea, salicylic acid, alpha-hydroxy acids), topical corticosteroids, tazarotene, and topical calcineurin inhibitors. Systemic agents include oral retinoids and methotrexate.
Atopic dermatitis is a chronic inflammatory skin disease that involves genetic and environmental factors, leading to abnormalities in the epidermis and the immune system presenting with its typical morphology and distribution. The morphology of eczematous lesions is distinct from papulosquamous lesions of psoriasis.
Staphylococcal scalded skin syndrome is a toxin-mediated skin disorder which presents with denuded, peeling skin due to epidermolytic exotoxin producing Staphylococcus species. Fever, erythematous rash, malaise, skin pain, and irritability presents initially. Progressive desquamation with accentuation in folds is typical, with progression usually within 1-2 days. Systemic antibiotics covering Staphylococcus should be administered early. Emollients and nonadherent dressings should be applied to affected areas to promote healing. Supportive care includes dehydration management, temperature regulation, and nutrition. Skin desquamation usually occurs within 5 days with resolution within 2 weeks.
This infant displayed streptococcal intertrigo which triggered an early presentation of guttate psoriasis. The patient was managed with completion of a course of oral cephalexin, midstrength topical corticosteroids to the truncal lesions, and mild topical corticosteroids to the face and diaper area with good clinical response.
Danny Lee and Samuel Le serve as research fellows in the Pediatric Dermatology Division of the Department of Dermatology at the University of California San Diego and Rady Children’s Hospital, San Diego. Dr. Eichenfield is Distinguished Professor of Dermatology and Pediatrics and Vice-Chair of the Department of Dermatology at the University of California San Diego and Rady Children’s Hospital, San Diego. The authors have no relevant financial disclosures.
Suggested Reading
Leung AK et al. Childhood guttate psoriasis: An updated review. Drugs Context. 2023 Oct 23:12:2023-8-2. doi: 10.7573/dic.2023-8-2.
Galili E et al. New-onset guttate psoriasis: A long-term follow-up study. Dermatology. 2023;239(2):188-194. doi: 10.1159/000527737.
Duffin KC et al. Advances and controversies in our understanding of guttate and plaque psoriasis. J Rheumatol. 2023 Nov;50(Suppl 2):4-7. doi: 10.3899/jrheum.2023-0500.
Saleh D, Tanner LS. Guttate Psoriasis. [Updated 2023 Jul 31]. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2023 Jan-. Available from: www.ncbi.nlm.nih.gov/books/NBK482498/
Dupire G et al. Antistreptococcal interventions for guttate and chronic plaque psoriasis. Cochrane Database Syst Rev. 2019 Mar 5;3(3):CD011571. doi: 10.1002/14651858.CD011571.pub2.
Diagnosis: Infection-induced psoriasis (guttate-type, induced by streptococcal intertrigo)
Psoriasis is a chronic inflammatory disorder characterized by well-defined, scaly, erythematous plaques. Guttate psoriasis is a distinct variant of psoriasis that is more common in children and adolescents. Guttate psoriasis usually presents with multiple, scattered, small, drop-like (“guttate”), scaly, erythematous papules and plaques.
The pathophysiology of psoriasis involves an interplay between genetic and environmental factors. Guttate psoriasis is a chronic T-cell–mediated inflammatory disease in which there is an altered balance between T-helper-1 (TH1) and TH2 cells, transcription factor genes, and their products. HLA B-13, B-17, and Cw6 are human leukocyte antigen alleles implicated in genetic susceptibility. It is hypothesized that streptococcal infection precipitates guttate psoriasis by streptococcal superantigen–driven activation of cutaneous lymphocyte-associated antigen (CLA)–positive lymphocytes. It has been shown that streptococcal exotoxins and streptococcal M proteins act as superantigens.
Diagnosis is often made clinically based on characteristic physical findings and a possible preceding history of streptococcal infection. In patients with streptococcal infection, culture from an appropriate site and measurement of serum antistreptococcal antibody titers (for example, anti-DNase, antihyaluronidase and antistreptolysin-O) can help. A skin biopsy is usually not necessary but may be considered.
This patient presented with intertrigo of the neck and axillae at the time of presentation with the papulosquamous rash. Culture of the intertrigo yielded 4+ Group A beta streptococcus.
Treatment
Although there is currently no cure for guttate psoriasis, various treatment options can relieve symptoms and clear skin lesions, and infection-triggered lesions may remit, usually within several months. However, guttate psoriasis may persist and progress to chronic plaque psoriasis. Many treatment options are based mainly on clinical trials targeted for plaque psoriasis treatment.
For mild psoriasis, topical corticosteroids are first-line treatment. Other topical steroids include vitamin D analogs (calcipotriene), topical retinoids (tazarotene), topical calcineurin inhibitors (tacrolimus and pimecrolimus), and newer non-steroidal anti-inflammatory agents (roflumilast or tapinarof), neither approved yet in this young age group. In more severe cases, phototherapy with UVB light, traditional systemic immunosuppressive agents (methotrexate, cyclosporine) or targeted biologic therapies may be considered.
Differential Diagnosis
The differential diagnosis may include generalized intertrigo, pityriasis rubra pilaris, tinea corporis, atopic dermatitis, and staphylococcal scalded skin syndrome. Guttate psoriasis can be distinguished by history and physical exam. Further studies such as potassium hydroxide (KOH) scrapings may be helpful in ruling out the other disorders.
Intertrigo is an inflammatory condition of the flexural surfaces irritated by warm temperatures, friction, moisture, and poor ventilation that is commonly associated with Candida infection and/or streptococcal infection. Candidal intertrigo can present with erythematous patches or plaques in an intertriginous area that may develop erosions, macerations, fissures, crust, and weeping. Satellite papules and pustules are pathognomonic for Candida species. Streptococcal intertrigo usually presents with bright red color and may be painful or pruritic. Perianal streptococcal infection is reported as a trigger of guttate psoriasis in pediatric patients.
Pityriasis rubra pilaris is a rare inflammatory papulosquamous disorder with an unknown etiology. Red-orange papules and plaques, hyperkeratotic follicular papules, and palmoplantar hyperkeratosis are primary features. Diagnosis is based on clinical and histopathology. Pityriasis rubra pilaris is self-limited and asymptomatic in many cases. Treatment may not be required, but combination therapy with topical agents includes emollients, keratolytic agents (for example, urea, salicylic acid, alpha-hydroxy acids), topical corticosteroids, tazarotene, and topical calcineurin inhibitors. Systemic agents include oral retinoids and methotrexate.
Atopic dermatitis is a chronic inflammatory skin disease that involves genetic and environmental factors, leading to abnormalities in the epidermis and the immune system presenting with its typical morphology and distribution. The morphology of eczematous lesions is distinct from papulosquamous lesions of psoriasis.
Staphylococcal scalded skin syndrome is a toxin-mediated skin disorder which presents with denuded, peeling skin due to epidermolytic exotoxin producing Staphylococcus species. Fever, erythematous rash, malaise, skin pain, and irritability presents initially. Progressive desquamation with accentuation in folds is typical, with progression usually within 1-2 days. Systemic antibiotics covering Staphylococcus should be administered early. Emollients and nonadherent dressings should be applied to affected areas to promote healing. Supportive care includes dehydration management, temperature regulation, and nutrition. Skin desquamation usually occurs within 5 days with resolution within 2 weeks.
This infant displayed streptococcal intertrigo which triggered an early presentation of guttate psoriasis. The patient was managed with completion of a course of oral cephalexin, midstrength topical corticosteroids to the truncal lesions, and mild topical corticosteroids to the face and diaper area with good clinical response.
Danny Lee and Samuel Le serve as research fellows in the Pediatric Dermatology Division of the Department of Dermatology at the University of California San Diego and Rady Children’s Hospital, San Diego. Dr. Eichenfield is Distinguished Professor of Dermatology and Pediatrics and Vice-Chair of the Department of Dermatology at the University of California San Diego and Rady Children’s Hospital, San Diego. The authors have no relevant financial disclosures.
Suggested Reading
Leung AK et al. Childhood guttate psoriasis: An updated review. Drugs Context. 2023 Oct 23:12:2023-8-2. doi: 10.7573/dic.2023-8-2.
Galili E et al. New-onset guttate psoriasis: A long-term follow-up study. Dermatology. 2023;239(2):188-194. doi: 10.1159/000527737.
Duffin KC et al. Advances and controversies in our understanding of guttate and plaque psoriasis. J Rheumatol. 2023 Nov;50(Suppl 2):4-7. doi: 10.3899/jrheum.2023-0500.
Saleh D, Tanner LS. Guttate Psoriasis. [Updated 2023 Jul 31]. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2023 Jan-. Available from: www.ncbi.nlm.nih.gov/books/NBK482498/
Dupire G et al. Antistreptococcal interventions for guttate and chronic plaque psoriasis. Cochrane Database Syst Rev. 2019 Mar 5;3(3):CD011571. doi: 10.1002/14651858.CD011571.pub2.
On physical exam, there was an erythematous patch with overlying areas of macerations on the neck and axilla. The trunk, extremities, and diaper area had multiple psoriasiform erythematous thin plaques with overlying scales.