User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
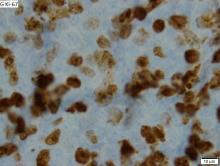
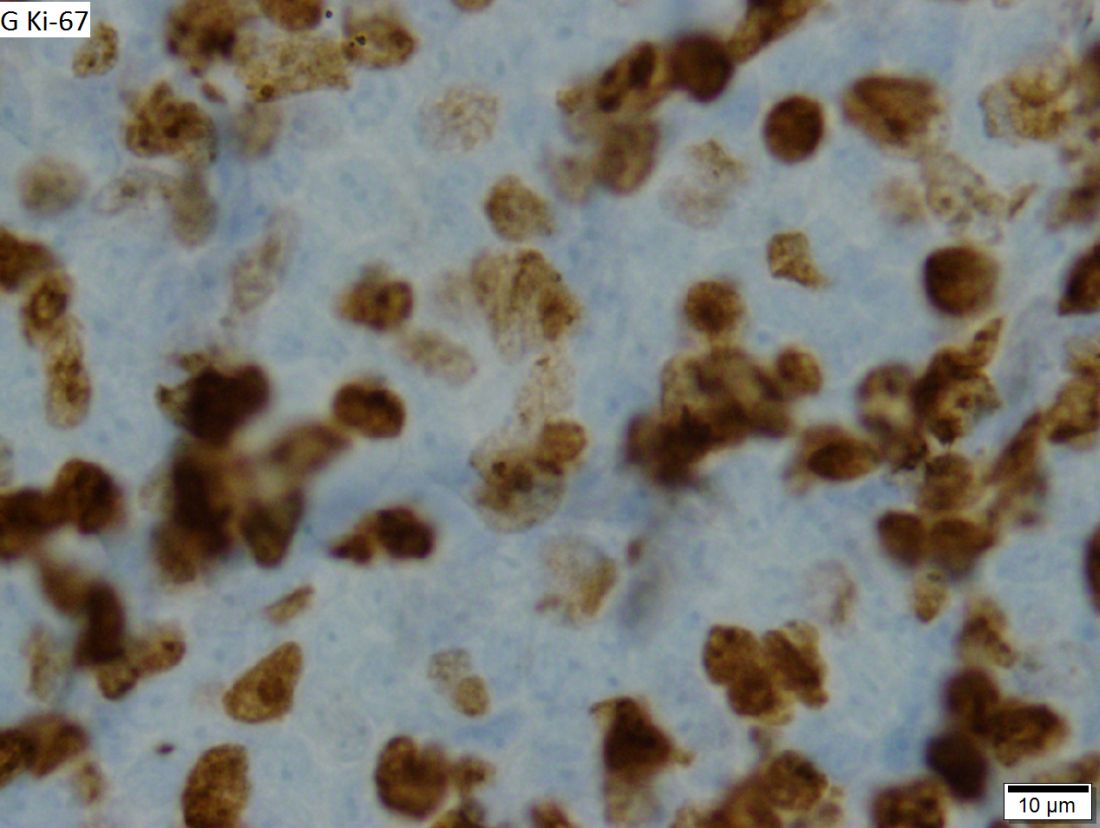
FDA approves pembrolizumab for relapsed/refractory PMBCL
The immune checkpoint inhibitor in adult and pediatric patients.
The Food and Drug Administration based the accelerated approval on results from 53 patients with relapsed or refractory primary mediastinal large B-cell lymphoma in the KEYNOTE-170 trial. In the phase 2 trial, patients received 200 mg of pembrolizumab intravenously for 3 weeks until unacceptable toxicity or documented disease progression occurred. This continued for up to 24 months in patients who did not display progression. The overall response rate to pembrolizumab was 45% (95% CI, 32-60), which included both complete (11%) and partial (34%) responses. The median duration of response was not met within the follow-up period (median, 9.7 months) and the median time to first objective response was 2.8 months.
The recommended dose for pembrolizumab in adults is 200 mg every 3 weeks. It is recommended that pediatric patients receive 2 mg/kg every 3 weeks, with a maximum dose of 200 mg.
The most common adverse reactions to pembrolizumab were musculoskeletal pain, upper respiratory tract infection, pyrexia, fatigue, cough, dyspnea, diarrhea, nausea, arrhythmia, and headache. In total, a quarter of patients with adverse reactions required systemic treatment with a corticosteroid and 26% of patients had serious adverse reactions.
Pembrolizumab was approved via the FDA’s accelerated approval process, which allows for earlier approval of drugs that treat serious medical conditions and fulfill an unmet medical need. The drug was approved based on tumor response rate and durability of response, the FDA noted.
The immune checkpoint inhibitor in adult and pediatric patients.
The Food and Drug Administration based the accelerated approval on results from 53 patients with relapsed or refractory primary mediastinal large B-cell lymphoma in the KEYNOTE-170 trial. In the phase 2 trial, patients received 200 mg of pembrolizumab intravenously for 3 weeks until unacceptable toxicity or documented disease progression occurred. This continued for up to 24 months in patients who did not display progression. The overall response rate to pembrolizumab was 45% (95% CI, 32-60), which included both complete (11%) and partial (34%) responses. The median duration of response was not met within the follow-up period (median, 9.7 months) and the median time to first objective response was 2.8 months.
The recommended dose for pembrolizumab in adults is 200 mg every 3 weeks. It is recommended that pediatric patients receive 2 mg/kg every 3 weeks, with a maximum dose of 200 mg.
The most common adverse reactions to pembrolizumab were musculoskeletal pain, upper respiratory tract infection, pyrexia, fatigue, cough, dyspnea, diarrhea, nausea, arrhythmia, and headache. In total, a quarter of patients with adverse reactions required systemic treatment with a corticosteroid and 26% of patients had serious adverse reactions.
Pembrolizumab was approved via the FDA’s accelerated approval process, which allows for earlier approval of drugs that treat serious medical conditions and fulfill an unmet medical need. The drug was approved based on tumor response rate and durability of response, the FDA noted.
The immune checkpoint inhibitor in adult and pediatric patients.
The Food and Drug Administration based the accelerated approval on results from 53 patients with relapsed or refractory primary mediastinal large B-cell lymphoma in the KEYNOTE-170 trial. In the phase 2 trial, patients received 200 mg of pembrolizumab intravenously for 3 weeks until unacceptable toxicity or documented disease progression occurred. This continued for up to 24 months in patients who did not display progression. The overall response rate to pembrolizumab was 45% (95% CI, 32-60), which included both complete (11%) and partial (34%) responses. The median duration of response was not met within the follow-up period (median, 9.7 months) and the median time to first objective response was 2.8 months.
The recommended dose for pembrolizumab in adults is 200 mg every 3 weeks. It is recommended that pediatric patients receive 2 mg/kg every 3 weeks, with a maximum dose of 200 mg.
The most common adverse reactions to pembrolizumab were musculoskeletal pain, upper respiratory tract infection, pyrexia, fatigue, cough, dyspnea, diarrhea, nausea, arrhythmia, and headache. In total, a quarter of patients with adverse reactions required systemic treatment with a corticosteroid and 26% of patients had serious adverse reactions.
Pembrolizumab was approved via the FDA’s accelerated approval process, which allows for earlier approval of drugs that treat serious medical conditions and fulfill an unmet medical need. The drug was approved based on tumor response rate and durability of response, the FDA noted.
FDA grants regular approval to venetoclax for CLL/SLL
Venetoclax (Venclexta) has received regular approval from the Food and Drug Administration for the treatment of patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL), with or without 17p deletion, who have received at least one prior therapy.
The approval was based results from the MURANO trial of 389 patients, which was a randomized, multicenter, open-label trial of venetoclax plus rituximab versus bendamustine plus rituximab.
Neutropenia, diarrhea, upper respiratory tract infection, fatigue, cough, and nausea were the most common adverse events seen in the venetoclax arm. Grade 3 or 4 neutropenia developed in 64% of those patients, and grade 4 in 31%. The most common infection in venetoclax patients was pneumonia, but overall, 21% of patients in that arm experienced some kind of infection.
Because of the rapid reduction in tumor size, tumor lysis syndrome is possible with venetoclax treatment, the FDA noted.
In 2016, the FDA granted accelerated approval to venetoclax for treatment of patients with CLL with 17d deletion who had received at least one prior line of therapy.
Venetoclax (Venclexta) has received regular approval from the Food and Drug Administration for the treatment of patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL), with or without 17p deletion, who have received at least one prior therapy.
The approval was based results from the MURANO trial of 389 patients, which was a randomized, multicenter, open-label trial of venetoclax plus rituximab versus bendamustine plus rituximab.
Neutropenia, diarrhea, upper respiratory tract infection, fatigue, cough, and nausea were the most common adverse events seen in the venetoclax arm. Grade 3 or 4 neutropenia developed in 64% of those patients, and grade 4 in 31%. The most common infection in venetoclax patients was pneumonia, but overall, 21% of patients in that arm experienced some kind of infection.
Because of the rapid reduction in tumor size, tumor lysis syndrome is possible with venetoclax treatment, the FDA noted.
In 2016, the FDA granted accelerated approval to venetoclax for treatment of patients with CLL with 17d deletion who had received at least one prior line of therapy.
Venetoclax (Venclexta) has received regular approval from the Food and Drug Administration for the treatment of patients with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL), with or without 17p deletion, who have received at least one prior therapy.
The approval was based results from the MURANO trial of 389 patients, which was a randomized, multicenter, open-label trial of venetoclax plus rituximab versus bendamustine plus rituximab.
Neutropenia, diarrhea, upper respiratory tract infection, fatigue, cough, and nausea were the most common adverse events seen in the venetoclax arm. Grade 3 or 4 neutropenia developed in 64% of those patients, and grade 4 in 31%. The most common infection in venetoclax patients was pneumonia, but overall, 21% of patients in that arm experienced some kind of infection.
Because of the rapid reduction in tumor size, tumor lysis syndrome is possible with venetoclax treatment, the FDA noted.
In 2016, the FDA granted accelerated approval to venetoclax for treatment of patients with CLL with 17d deletion who had received at least one prior line of therapy.
Venetoclax plus ibrutinib yields encouraging MRD results in first-line CLL
CHICAGO – The combination of ibrutinib plus venetoclax yielded a high rate of undetectable minimal residual disease (MRD) when used as first-line treatment for chronic lymphocytic leukemia (CLL), according to preliminary results of the CAPTIVATE trial.
Of the first 30 patients in the trial, 23 (77%) had undetectable blood MRD after just six cycles of combined treatment, said investigator William G. Wierda, MD, PhD, of the University of Texas MD Anderson Cancer Center, Houston.
“These early results show a highly active and safe treatment with 12 cycles of combined treatment with ibrutinib and venetoclax,” Dr. Wierda said in a presentation of the CAPTIVATE results at the annual meeting of the American Society of Clinical Oncology.
Those MRD results are “at least as good as we can achieve with chemoimmunotherapy,” Bruce D. Cheson, MD, head of hematology at Georgetown University, Washington, said during a discussion of the CAPTIVATE study results.
Dr. Cheson referenced MRD results from a 2016 analysis of the CLL8 and CLL10 trials, which included patients treated with fludarabine, cyclophosphamide, and rituximab (FCR) and bendamustine plus rituximab (BR). In that analysis, 33.6% of patients achieved MRD-negative complete response and 29.1% achieved MRD-negative partial response.
In CAPTIVATE, by contrast, all of the complete remissions were MRD negative, as were a majority of the partial responders, Dr. Cheson noted.
Venetoclax and ibrutinib have “clinically complimentary activity” that provided a rationale for combining the two, Dr. Wierda said at ASCO. Ibrutinib is a BTK inhibitor that has a high rate of response and durable disease control, though continuous treatment is indicated, he said, because most patients achieve partial remissions as best response and continue to have residual disease in blood or bone marrow. Venetoclax, he added, is a BCL-2 inhibitor that produces durable partial remissions, though “residual disease is typically present in the form of persistently enlarged lymph nodes,” he said. “Venetoclax is highly effective at clearing disease from blood and bone marrow.”
The phase 2 CAPTIVATE trial includes a total of 164 patients younger than 70 years of age who receive a 3-cycle ibrutinib lead-in, followed by ibrutinib plus venetoclax for 12 cycles. At that point, patients are randomized according to MRD status. Patients with confirmed undetectable MRD are randomized to further treatment with ibrutinib or placebo, and those with undetectable MRD not confirmed are randomized to ibrutinib versus ibrutinib plus venetoclax.
In addition to early efficacy data, Dr. Wierda also reported some safety data. Compared with the single-agent ibrutinib lead-in period, combined ibrutinib plus venetoclax treatment had more gastrointestinal-associated events and neutropenia. Almost half of patients (45%) have had a treatment-related grade 3-4 adverse event, though just 18 (11%) have had treatment-related adverse events classified as serious, and there have been no adverse event-related deaths on study.
The high activity of ibrutinib plus venetoclax in CAPTIVATE supports further study of the combination, Dr. Wierda said. A randomized, open-label phase 3 trial of ibrutinib plus venetoclax versus chlorambucil plus obinutuzumab as first-line treatment for CLL is currently recruiting.
The study was sponsored by Pharmacyclics, an AbbVie company. Dr. Wierda reported consulting and research funding from Pharmacyclics, AbbVie, and several other companies.
SOURCE: Wierda WG et al. ASCO 2018, Abstract 7502.
CHICAGO – The combination of ibrutinib plus venetoclax yielded a high rate of undetectable minimal residual disease (MRD) when used as first-line treatment for chronic lymphocytic leukemia (CLL), according to preliminary results of the CAPTIVATE trial.
Of the first 30 patients in the trial, 23 (77%) had undetectable blood MRD after just six cycles of combined treatment, said investigator William G. Wierda, MD, PhD, of the University of Texas MD Anderson Cancer Center, Houston.
“These early results show a highly active and safe treatment with 12 cycles of combined treatment with ibrutinib and venetoclax,” Dr. Wierda said in a presentation of the CAPTIVATE results at the annual meeting of the American Society of Clinical Oncology.
Those MRD results are “at least as good as we can achieve with chemoimmunotherapy,” Bruce D. Cheson, MD, head of hematology at Georgetown University, Washington, said during a discussion of the CAPTIVATE study results.
Dr. Cheson referenced MRD results from a 2016 analysis of the CLL8 and CLL10 trials, which included patients treated with fludarabine, cyclophosphamide, and rituximab (FCR) and bendamustine plus rituximab (BR). In that analysis, 33.6% of patients achieved MRD-negative complete response and 29.1% achieved MRD-negative partial response.
In CAPTIVATE, by contrast, all of the complete remissions were MRD negative, as were a majority of the partial responders, Dr. Cheson noted.
Venetoclax and ibrutinib have “clinically complimentary activity” that provided a rationale for combining the two, Dr. Wierda said at ASCO. Ibrutinib is a BTK inhibitor that has a high rate of response and durable disease control, though continuous treatment is indicated, he said, because most patients achieve partial remissions as best response and continue to have residual disease in blood or bone marrow. Venetoclax, he added, is a BCL-2 inhibitor that produces durable partial remissions, though “residual disease is typically present in the form of persistently enlarged lymph nodes,” he said. “Venetoclax is highly effective at clearing disease from blood and bone marrow.”
The phase 2 CAPTIVATE trial includes a total of 164 patients younger than 70 years of age who receive a 3-cycle ibrutinib lead-in, followed by ibrutinib plus venetoclax for 12 cycles. At that point, patients are randomized according to MRD status. Patients with confirmed undetectable MRD are randomized to further treatment with ibrutinib or placebo, and those with undetectable MRD not confirmed are randomized to ibrutinib versus ibrutinib plus venetoclax.
In addition to early efficacy data, Dr. Wierda also reported some safety data. Compared with the single-agent ibrutinib lead-in period, combined ibrutinib plus venetoclax treatment had more gastrointestinal-associated events and neutropenia. Almost half of patients (45%) have had a treatment-related grade 3-4 adverse event, though just 18 (11%) have had treatment-related adverse events classified as serious, and there have been no adverse event-related deaths on study.
The high activity of ibrutinib plus venetoclax in CAPTIVATE supports further study of the combination, Dr. Wierda said. A randomized, open-label phase 3 trial of ibrutinib plus venetoclax versus chlorambucil plus obinutuzumab as first-line treatment for CLL is currently recruiting.
The study was sponsored by Pharmacyclics, an AbbVie company. Dr. Wierda reported consulting and research funding from Pharmacyclics, AbbVie, and several other companies.
SOURCE: Wierda WG et al. ASCO 2018, Abstract 7502.
CHICAGO – The combination of ibrutinib plus venetoclax yielded a high rate of undetectable minimal residual disease (MRD) when used as first-line treatment for chronic lymphocytic leukemia (CLL), according to preliminary results of the CAPTIVATE trial.
Of the first 30 patients in the trial, 23 (77%) had undetectable blood MRD after just six cycles of combined treatment, said investigator William G. Wierda, MD, PhD, of the University of Texas MD Anderson Cancer Center, Houston.
“These early results show a highly active and safe treatment with 12 cycles of combined treatment with ibrutinib and venetoclax,” Dr. Wierda said in a presentation of the CAPTIVATE results at the annual meeting of the American Society of Clinical Oncology.
Those MRD results are “at least as good as we can achieve with chemoimmunotherapy,” Bruce D. Cheson, MD, head of hematology at Georgetown University, Washington, said during a discussion of the CAPTIVATE study results.
Dr. Cheson referenced MRD results from a 2016 analysis of the CLL8 and CLL10 trials, which included patients treated with fludarabine, cyclophosphamide, and rituximab (FCR) and bendamustine plus rituximab (BR). In that analysis, 33.6% of patients achieved MRD-negative complete response and 29.1% achieved MRD-negative partial response.
In CAPTIVATE, by contrast, all of the complete remissions were MRD negative, as were a majority of the partial responders, Dr. Cheson noted.
Venetoclax and ibrutinib have “clinically complimentary activity” that provided a rationale for combining the two, Dr. Wierda said at ASCO. Ibrutinib is a BTK inhibitor that has a high rate of response and durable disease control, though continuous treatment is indicated, he said, because most patients achieve partial remissions as best response and continue to have residual disease in blood or bone marrow. Venetoclax, he added, is a BCL-2 inhibitor that produces durable partial remissions, though “residual disease is typically present in the form of persistently enlarged lymph nodes,” he said. “Venetoclax is highly effective at clearing disease from blood and bone marrow.”
The phase 2 CAPTIVATE trial includes a total of 164 patients younger than 70 years of age who receive a 3-cycle ibrutinib lead-in, followed by ibrutinib plus venetoclax for 12 cycles. At that point, patients are randomized according to MRD status. Patients with confirmed undetectable MRD are randomized to further treatment with ibrutinib or placebo, and those with undetectable MRD not confirmed are randomized to ibrutinib versus ibrutinib plus venetoclax.
In addition to early efficacy data, Dr. Wierda also reported some safety data. Compared with the single-agent ibrutinib lead-in period, combined ibrutinib plus venetoclax treatment had more gastrointestinal-associated events and neutropenia. Almost half of patients (45%) have had a treatment-related grade 3-4 adverse event, though just 18 (11%) have had treatment-related adverse events classified as serious, and there have been no adverse event-related deaths on study.
The high activity of ibrutinib plus venetoclax in CAPTIVATE supports further study of the combination, Dr. Wierda said. A randomized, open-label phase 3 trial of ibrutinib plus venetoclax versus chlorambucil plus obinutuzumab as first-line treatment for CLL is currently recruiting.
The study was sponsored by Pharmacyclics, an AbbVie company. Dr. Wierda reported consulting and research funding from Pharmacyclics, AbbVie, and several other companies.
SOURCE: Wierda WG et al. ASCO 2018, Abstract 7502.
REPORTING FROM ASCO 2018
Key clinical point:
Major finding: Of 14 patients, 12 (86%) who completed 12 cycles of treatment had undetectable bone marrow MRD.
Study details: Early results of the phase 2 CAPTIVATE trial including 164 patients younger than 70 years of age with previously untreated CLL.
Disclosures: The study was sponsored by Pharmacyclics, an Abbvie company. Dr. Wierda reported consulting and research funding from Pharmacyclics, AbbVie, and several other companies.
Source: Wierda WG et al. ASCO 2018, Abstract 7502.
Chemo-free regimen appears viable in previously untreated FL
CHICAGO – Lenalidomide plus rituximab (R2) had comparable efficacy versus standard chemoimmunotherapy in patients with previously untreated follicular lymphoma, according to results from a phase 3 trial.
RELEVANCE is the first randomized, phase 3 trial to examine a chemotherapy-free regimen in this setting.
Response and progression-free survival (PFS) results were similar for patients who received R2 followed by rituximab maintenance and patients assigned to chemotherapy plus rituximab and rituximab maintenance, in study results presented at the annual meeting of the American Society of Clinical Oncology.
“These results show that lenalidomide plus rituximab, which is a novel immunomodulatory approach, is a potential first-line option for patients with follicular lymphoma that require treatment,” said investigator Nathan H. Fowler, MD, of the University of Texas MD Anderson Cancer Center, Houston.
But since the study was designed as a superiority trial, rather than a noninferiority trial, and it failed to meet its primary endpoint of superior complete remission (CR) or CR unconfirmed (CRu) at 120 weeks, said Bruce D. Cheson, MD, head of hematology at Georgetown University, Washington.
R2 had a similar PFS overall and in all major patient subgroups, similar overall survival, less nonhematologic toxicity aside from rash, less neutropenia, and fewer infections despite increased use of growth factors in the chemoimmunotherapy arm, Dr. Cheson said in a presentation commenting on the results. “Therefore, I agree with Dr. Fowler’s conclusion that R2 can be considered as an option for the front-line therapy of patients with follicular lymphoma,” Dr. Cheson said.
The RELEVANCE study included 1,030 patients (median age, 59 years) with previously untreated, advanced follicular lymphoma requiring treatment. They were randomized 1:1 to either lenalidomide plus rituximab followed by rituximab maintenance, or R-chemotherapy followed by rituximab maintenance.
For patients randomly assigned to R-chemotherapy, physicians could choose among three standard regimens: rituximab plus bendamustine (R-B), rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), or rituximab plus cyclophosphamide, vincristine, and prednisone (R-CVP).
There was no statistical difference between treatment approaches in CR/CRu at 120 weeks, which was 48% in the R2 arm and 53% in the R-chemotherapy arm (P = 0.13). Best CR/CRu also was not statistically different between arms (59% and 67%, respectively), as was best overall response rate (84% and 89%). The 3-year duration of response was 77% in the R2 arm and 74% for R-chemotherapy.
With 37.9 months median follow-up, progression-free survival was “nearly identical” between the two groups, Dr. Fowler said, at 77% for R2 and 78% for R-chemotherapy (P = 0.48). The 3-year overall survival was 94% in both the R2 and R-chemotherapy arms, though survival data are still immature, Dr. Fowler noted.
Grade 3/4 neutropenia was more common in the R-chemotherapy arm, resulting in higher rates of febrile neutropenia, according to Dr. Fowler, who also noted that rash and cutaneous reactions were more common with R2. About 70% of patients in each arm were able to tolerate treatment, and reasons for discontinuation were “fairly similar” between arms, Dr. Fowler added.
Second primary malignancies occurred in 7% of patients in the R2 arm and 10% of the R-chemotherapy arm.
The study was sponsored was Celgene and the Lymphoma Academic Research Organisation. Dr. Fowler reported disclosures related to Abbvie, Celgene, Janssen, Merck, and Roche.
SOURCE: Fowler NH et al. ASCO 2018, Abstract 7500.
CHICAGO – Lenalidomide plus rituximab (R2) had comparable efficacy versus standard chemoimmunotherapy in patients with previously untreated follicular lymphoma, according to results from a phase 3 trial.
RELEVANCE is the first randomized, phase 3 trial to examine a chemotherapy-free regimen in this setting.
Response and progression-free survival (PFS) results were similar for patients who received R2 followed by rituximab maintenance and patients assigned to chemotherapy plus rituximab and rituximab maintenance, in study results presented at the annual meeting of the American Society of Clinical Oncology.
“These results show that lenalidomide plus rituximab, which is a novel immunomodulatory approach, is a potential first-line option for patients with follicular lymphoma that require treatment,” said investigator Nathan H. Fowler, MD, of the University of Texas MD Anderson Cancer Center, Houston.
But since the study was designed as a superiority trial, rather than a noninferiority trial, and it failed to meet its primary endpoint of superior complete remission (CR) or CR unconfirmed (CRu) at 120 weeks, said Bruce D. Cheson, MD, head of hematology at Georgetown University, Washington.
R2 had a similar PFS overall and in all major patient subgroups, similar overall survival, less nonhematologic toxicity aside from rash, less neutropenia, and fewer infections despite increased use of growth factors in the chemoimmunotherapy arm, Dr. Cheson said in a presentation commenting on the results. “Therefore, I agree with Dr. Fowler’s conclusion that R2 can be considered as an option for the front-line therapy of patients with follicular lymphoma,” Dr. Cheson said.
The RELEVANCE study included 1,030 patients (median age, 59 years) with previously untreated, advanced follicular lymphoma requiring treatment. They were randomized 1:1 to either lenalidomide plus rituximab followed by rituximab maintenance, or R-chemotherapy followed by rituximab maintenance.
For patients randomly assigned to R-chemotherapy, physicians could choose among three standard regimens: rituximab plus bendamustine (R-B), rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), or rituximab plus cyclophosphamide, vincristine, and prednisone (R-CVP).
There was no statistical difference between treatment approaches in CR/CRu at 120 weeks, which was 48% in the R2 arm and 53% in the R-chemotherapy arm (P = 0.13). Best CR/CRu also was not statistically different between arms (59% and 67%, respectively), as was best overall response rate (84% and 89%). The 3-year duration of response was 77% in the R2 arm and 74% for R-chemotherapy.
With 37.9 months median follow-up, progression-free survival was “nearly identical” between the two groups, Dr. Fowler said, at 77% for R2 and 78% for R-chemotherapy (P = 0.48). The 3-year overall survival was 94% in both the R2 and R-chemotherapy arms, though survival data are still immature, Dr. Fowler noted.
Grade 3/4 neutropenia was more common in the R-chemotherapy arm, resulting in higher rates of febrile neutropenia, according to Dr. Fowler, who also noted that rash and cutaneous reactions were more common with R2. About 70% of patients in each arm were able to tolerate treatment, and reasons for discontinuation were “fairly similar” between arms, Dr. Fowler added.
Second primary malignancies occurred in 7% of patients in the R2 arm and 10% of the R-chemotherapy arm.
The study was sponsored was Celgene and the Lymphoma Academic Research Organisation. Dr. Fowler reported disclosures related to Abbvie, Celgene, Janssen, Merck, and Roche.
SOURCE: Fowler NH et al. ASCO 2018, Abstract 7500.
CHICAGO – Lenalidomide plus rituximab (R2) had comparable efficacy versus standard chemoimmunotherapy in patients with previously untreated follicular lymphoma, according to results from a phase 3 trial.
RELEVANCE is the first randomized, phase 3 trial to examine a chemotherapy-free regimen in this setting.
Response and progression-free survival (PFS) results were similar for patients who received R2 followed by rituximab maintenance and patients assigned to chemotherapy plus rituximab and rituximab maintenance, in study results presented at the annual meeting of the American Society of Clinical Oncology.
“These results show that lenalidomide plus rituximab, which is a novel immunomodulatory approach, is a potential first-line option for patients with follicular lymphoma that require treatment,” said investigator Nathan H. Fowler, MD, of the University of Texas MD Anderson Cancer Center, Houston.
But since the study was designed as a superiority trial, rather than a noninferiority trial, and it failed to meet its primary endpoint of superior complete remission (CR) or CR unconfirmed (CRu) at 120 weeks, said Bruce D. Cheson, MD, head of hematology at Georgetown University, Washington.
R2 had a similar PFS overall and in all major patient subgroups, similar overall survival, less nonhematologic toxicity aside from rash, less neutropenia, and fewer infections despite increased use of growth factors in the chemoimmunotherapy arm, Dr. Cheson said in a presentation commenting on the results. “Therefore, I agree with Dr. Fowler’s conclusion that R2 can be considered as an option for the front-line therapy of patients with follicular lymphoma,” Dr. Cheson said.
The RELEVANCE study included 1,030 patients (median age, 59 years) with previously untreated, advanced follicular lymphoma requiring treatment. They were randomized 1:1 to either lenalidomide plus rituximab followed by rituximab maintenance, or R-chemotherapy followed by rituximab maintenance.
For patients randomly assigned to R-chemotherapy, physicians could choose among three standard regimens: rituximab plus bendamustine (R-B), rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), or rituximab plus cyclophosphamide, vincristine, and prednisone (R-CVP).
There was no statistical difference between treatment approaches in CR/CRu at 120 weeks, which was 48% in the R2 arm and 53% in the R-chemotherapy arm (P = 0.13). Best CR/CRu also was not statistically different between arms (59% and 67%, respectively), as was best overall response rate (84% and 89%). The 3-year duration of response was 77% in the R2 arm and 74% for R-chemotherapy.
With 37.9 months median follow-up, progression-free survival was “nearly identical” between the two groups, Dr. Fowler said, at 77% for R2 and 78% for R-chemotherapy (P = 0.48). The 3-year overall survival was 94% in both the R2 and R-chemotherapy arms, though survival data are still immature, Dr. Fowler noted.
Grade 3/4 neutropenia was more common in the R-chemotherapy arm, resulting in higher rates of febrile neutropenia, according to Dr. Fowler, who also noted that rash and cutaneous reactions were more common with R2. About 70% of patients in each arm were able to tolerate treatment, and reasons for discontinuation were “fairly similar” between arms, Dr. Fowler added.
Second primary malignancies occurred in 7% of patients in the R2 arm and 10% of the R-chemotherapy arm.
The study was sponsored was Celgene and the Lymphoma Academic Research Organisation. Dr. Fowler reported disclosures related to Abbvie, Celgene, Janssen, Merck, and Roche.
SOURCE: Fowler NH et al. ASCO 2018, Abstract 7500.
REPORTING FROM ASCO 2018
Key clinical point: in patients with previously untreated follicular lymphoma.
Major finding: With 37.9 months’ median follow-up, progression-free survival was “nearly identical” between the two groups, at 77% for R2 and 78% for rituximab chemotherapy (P = 0.48).
Study details: RELEVANCE, a phase 3, randomized clinical trial including 1,030 patients with previously untreated, advanced follicular lymphoma requiring treatment.
Disclosures: The study was sponsored was Celgene and the Lymphoma Academic Research Organisation. Dr. Fowler reported disclosures related to AbbVie, Celgene, Janssen, Merck, and Roche.
Source: Fowler NH et al. ASCO 2018, Abstract 7500.
Novel antibody shifts ‘eat me/don’t eat me’ balance in refractory NHL
CHICAGO – A first-in-class antibody targeting the macrophage checkpoint CD47 is a promising novel immunotherapy in non-Hodgkin lymphoma, according to Ranjana H. Advani, MD, of Stanford (Calif.) Cancer Institute.
Treatment with Hu5F9-G4 (5F9), an antibody designed to overcome the “don’t eat me” signal associated with CD47, produced “encouraging” antitumor activity in a phase 1b study of 22 patients, Dr. Advani said in an oral abstract presentation at the annual meeting of the American Society of Clinical Oncology.
“5F9 was well tolerated in combination with rituximab, with no maximum tolerated dose achieved,” said Dr. Advani, noting that there were complete remissions in 43% of the refractory follicular lymphoma patients and 33% of refractory diffuse large B-cell lymphoma patients in the phase 1b/2 study.
The antibody has an on-target anemia effect that occurs upon administration, but that was mitigated considerably by a priming and maintenance dosing approach, she added.
The study has demonstrated “excellent” response rates in a highly refractory patient population, said Caron A. Jacobson, MD, of Dana-Farber Cancer Institute and Harvard Medical School in Boston. “Targeting CD47 ... really helps to shift the balance from ‘don’t eat me’ to ‘eat me,’ ” Dr. Jacobson said at the meeting.
“Importantly, we saw very little toxicity in the study, with very few grade 4 adverse events and no immune-related adverse events,” she added.
Most adverse events were grade 1 or 2, with the most common being the expected on-target anemia associated with 5F9. Using an initial priming dose of 5F9 results in a “temporary and mild decline” in hemoglobin due to clearance of aged red blood cells, Dr. Advani said.
The objective response rate in the study was 50%, with efficacy observed in rituximab-refractory patients, Dr. Advani said. With a median follow-up of greater than 6 months, just 1 of 11 responders had progressed. The median duration of response was not reached, with the longest complete remission lasting more than 14 months.
5F9 is able to selectively eliminate cancer cells through blockade of CD47, while rituximab enhances 5F9’s activity via antibody-dependent cellular phagocytosis, according to Dr. Advani.
“CD47 blockade takes the foot off the brakes, while rituximab puts the foot on the accelerator, leading to maximal tumor phagocytosis,” she said.
The Food and Drug Administration recently granted 5F9 a fast track designation for both diffuse large B-cell lymphoma and follicular lymphoma. Phase 2 investigations of 5F9 in these lymphomas are ongoing, Dr. Advani said.
The trial is sponsored by Forty Seven. Dr. Advani reported research funding from Forty Seven, which is developing 5F9, as well as disclosures related to AstraZeneca, Bayer, Bristol-Myers Squibb, Cell Medica, Genentech/Roche, Gilead Sciences, Pharmacyclics, and Seattle Genetics, among others.
SOURCE: Advani RH et al. ASCO 2018, abstract 7504.
CHICAGO – A first-in-class antibody targeting the macrophage checkpoint CD47 is a promising novel immunotherapy in non-Hodgkin lymphoma, according to Ranjana H. Advani, MD, of Stanford (Calif.) Cancer Institute.
Treatment with Hu5F9-G4 (5F9), an antibody designed to overcome the “don’t eat me” signal associated with CD47, produced “encouraging” antitumor activity in a phase 1b study of 22 patients, Dr. Advani said in an oral abstract presentation at the annual meeting of the American Society of Clinical Oncology.
“5F9 was well tolerated in combination with rituximab, with no maximum tolerated dose achieved,” said Dr. Advani, noting that there were complete remissions in 43% of the refractory follicular lymphoma patients and 33% of refractory diffuse large B-cell lymphoma patients in the phase 1b/2 study.
The antibody has an on-target anemia effect that occurs upon administration, but that was mitigated considerably by a priming and maintenance dosing approach, she added.
The study has demonstrated “excellent” response rates in a highly refractory patient population, said Caron A. Jacobson, MD, of Dana-Farber Cancer Institute and Harvard Medical School in Boston. “Targeting CD47 ... really helps to shift the balance from ‘don’t eat me’ to ‘eat me,’ ” Dr. Jacobson said at the meeting.
“Importantly, we saw very little toxicity in the study, with very few grade 4 adverse events and no immune-related adverse events,” she added.
Most adverse events were grade 1 or 2, with the most common being the expected on-target anemia associated with 5F9. Using an initial priming dose of 5F9 results in a “temporary and mild decline” in hemoglobin due to clearance of aged red blood cells, Dr. Advani said.
The objective response rate in the study was 50%, with efficacy observed in rituximab-refractory patients, Dr. Advani said. With a median follow-up of greater than 6 months, just 1 of 11 responders had progressed. The median duration of response was not reached, with the longest complete remission lasting more than 14 months.
5F9 is able to selectively eliminate cancer cells through blockade of CD47, while rituximab enhances 5F9’s activity via antibody-dependent cellular phagocytosis, according to Dr. Advani.
“CD47 blockade takes the foot off the brakes, while rituximab puts the foot on the accelerator, leading to maximal tumor phagocytosis,” she said.
The Food and Drug Administration recently granted 5F9 a fast track designation for both diffuse large B-cell lymphoma and follicular lymphoma. Phase 2 investigations of 5F9 in these lymphomas are ongoing, Dr. Advani said.
The trial is sponsored by Forty Seven. Dr. Advani reported research funding from Forty Seven, which is developing 5F9, as well as disclosures related to AstraZeneca, Bayer, Bristol-Myers Squibb, Cell Medica, Genentech/Roche, Gilead Sciences, Pharmacyclics, and Seattle Genetics, among others.
SOURCE: Advani RH et al. ASCO 2018, abstract 7504.
CHICAGO – A first-in-class antibody targeting the macrophage checkpoint CD47 is a promising novel immunotherapy in non-Hodgkin lymphoma, according to Ranjana H. Advani, MD, of Stanford (Calif.) Cancer Institute.
Treatment with Hu5F9-G4 (5F9), an antibody designed to overcome the “don’t eat me” signal associated with CD47, produced “encouraging” antitumor activity in a phase 1b study of 22 patients, Dr. Advani said in an oral abstract presentation at the annual meeting of the American Society of Clinical Oncology.
“5F9 was well tolerated in combination with rituximab, with no maximum tolerated dose achieved,” said Dr. Advani, noting that there were complete remissions in 43% of the refractory follicular lymphoma patients and 33% of refractory diffuse large B-cell lymphoma patients in the phase 1b/2 study.
The antibody has an on-target anemia effect that occurs upon administration, but that was mitigated considerably by a priming and maintenance dosing approach, she added.
The study has demonstrated “excellent” response rates in a highly refractory patient population, said Caron A. Jacobson, MD, of Dana-Farber Cancer Institute and Harvard Medical School in Boston. “Targeting CD47 ... really helps to shift the balance from ‘don’t eat me’ to ‘eat me,’ ” Dr. Jacobson said at the meeting.
“Importantly, we saw very little toxicity in the study, with very few grade 4 adverse events and no immune-related adverse events,” she added.
Most adverse events were grade 1 or 2, with the most common being the expected on-target anemia associated with 5F9. Using an initial priming dose of 5F9 results in a “temporary and mild decline” in hemoglobin due to clearance of aged red blood cells, Dr. Advani said.
The objective response rate in the study was 50%, with efficacy observed in rituximab-refractory patients, Dr. Advani said. With a median follow-up of greater than 6 months, just 1 of 11 responders had progressed. The median duration of response was not reached, with the longest complete remission lasting more than 14 months.
5F9 is able to selectively eliminate cancer cells through blockade of CD47, while rituximab enhances 5F9’s activity via antibody-dependent cellular phagocytosis, according to Dr. Advani.
“CD47 blockade takes the foot off the brakes, while rituximab puts the foot on the accelerator, leading to maximal tumor phagocytosis,” she said.
The Food and Drug Administration recently granted 5F9 a fast track designation for both diffuse large B-cell lymphoma and follicular lymphoma. Phase 2 investigations of 5F9 in these lymphomas are ongoing, Dr. Advani said.
The trial is sponsored by Forty Seven. Dr. Advani reported research funding from Forty Seven, which is developing 5F9, as well as disclosures related to AstraZeneca, Bayer, Bristol-Myers Squibb, Cell Medica, Genentech/Roche, Gilead Sciences, Pharmacyclics, and Seattle Genetics, among others.
SOURCE: Advani RH et al. ASCO 2018, abstract 7504.
REPORTING FROM ASCO 2018
Key clinical point:
Major finding: Complete responses were seen in 43% of follicular lymphoma (FL) patients and 33% of diffuse large B-cell lymphoma (DLBCL) patients.
Study details: Initial reported results from a phase 1b/2 study of 7 patients with FL and 15 patients with DLBCL.
Disclosures: Forty Seven sponsored the trial. Dr. Advani reported research funding from Forty Seven, which is developing 5F9, as well as disclosures related to AstraZeneca, Bayer, Bristol-Myers Squibb, Cell Medica, Genentech/Roche, Gilead Sciences, Pharmacyclics, and Seattle Genetics, among others.
Source: Advani RH et al. ASCO 2018, abstract 7504.
Ibrutinib plus rituximab amped PFS in Waldenström’s
, results of a randomized phase 3 trial show.
The results make ibrutinib plus rituximab “a new standard of care” for the disease, said investigator Meletios A. Dimopoulos, MD, with the Department of Clinical Therapeutics, National and Kapodistrian University of Athens, Greece.
“This is a combination with remarkable activity as far as progression-free survival is concerned, and well tolerated,” Dr. Dimopoulos said in a presentation of the data here at the 2018 annual meeting of the American Society of Clinical Oncology (ASCO).
Ibrutinib, a BTK inhibitor, was approved as a single agent in 2015 for the treatment of Waldenström’s macroglobulinemia.* Clinical practice guidelines from the National Comprehensive Cancer Network (NCCN) list ibrutinib as an “other” recommended regimen, noting that study data show a “lower overall and absence of major responses” reported for MYD88 wild-type patients.Rituximab alone and in combination with other agents has clinical activity in Waldenström’s. Moreover, preclinical studies have shown that rituximab and ibrutinib have synergistic activity, Dr. Dimopoulos said, presenting data from the iNNOVATE Study Group and the European Consortium for Waldenström’s Macroglobulinemia that was published concurrently in the New England Journal of Medicine.
They initiated a study of 150 symptomatic patients randomized to either ibrutinib plus rituximab or placebo plus rituximab, with a primary end point of progression-free survival.
They also assessed mutational status of MYD88 and CXCR4 in bone marrow samples, given that previous data suggest outcomes of ibrutinib treatment for this disease depend on MYD88 and CXCR4 mutational status, they said in the report.
Progression-free survival at 30 months was 82% for ibrutinib plus rituximab, compared to 28% for placebo plus rituximab (hazard ratio for progression or death, 0.20; P less than 0.001). Further, ibrutinib and rituximab had a benefit that was independent of the MYD88 or CXCR4 genotype, the investigators reported.
“Response rates with ibrutinib–rituximab were similar across different CXCR4 genotypes, but were slightly lower among patients who did not have the activating MYD88 L265P mutation, which triggers the growth of malignant cells through BTK and hematopoietic-cell kinase, both of which are targeted by ibrutinib,” they wrote.
Atrial fibrillation of grade 3 or higher occurred in 12% of the ibrutinib-rituximab and in 1% of placebo-rituximab groups. In the ibrutinib-rituximab group, the majority of these cases occurred in patients older than 75 years of age, Dr. Dimopoulos reported at ASCO.
Grade 3 or greater hypertension occurred in 13% and 4% of the ibrutinib-rituximab and placebo-rituximab groups respectively, while respiratory tract infections occurred in 4% and 0%. Conversely, the ibrutinib-rituximab arm had a lower rate of grade 3 or greater infusion reactions (1% vs. 16%), he said.
Based on this report, ibrutinib plus rituximab is “appealing, primarily in MYD88-mutated patients, which is the vast majority of Waldenström’s patients,” said Craig Hofmeister, MD, MPH, who commented on the study in a ASCO presentation discussing the results of this trial.
Dr. Hofmeister, of Winship Cancer Institute, Emory University, Atlanta, noted the numerically lower response rate in patients without the MYD88 mutation “makes me wonder whether I would want to have ibrutinib if I was MYD88 wild type.”
Atrial fibrillation and infections on the ibrutinib-rituximab regimen may require close monitoring and should be managed appropriately, he added. “Atrial fibrillation seems to be a consistent theme with ibrutinib, certainly in patients who are older and have probably more heart disease.”
The study (NCT02165397) was funded by Pharmacyclics and Janssen Research and Development. Dr. Dimopoulos reported personal fees from Amgen, Celgene, Janssen, and Takeda, outside of the submitted work. Co-authors reported disclosures related to Pharmacyclics, Bristol-Myers Squibb, Gilead, Roche, and AbbVie, among others. Dr. Hofmeister had no relevant financial disclosures.
SOURCE: Dimopoulos MA, et al. N Engl J Med. 2018 Jun 1. ASCO Abstract 8003.
*Correction, 8/27/2018: An earlier version of this story misstated the date of ibrutinib's approval as a single agent in the treatment of Waldenström’s .
, results of a randomized phase 3 trial show.
The results make ibrutinib plus rituximab “a new standard of care” for the disease, said investigator Meletios A. Dimopoulos, MD, with the Department of Clinical Therapeutics, National and Kapodistrian University of Athens, Greece.
“This is a combination with remarkable activity as far as progression-free survival is concerned, and well tolerated,” Dr. Dimopoulos said in a presentation of the data here at the 2018 annual meeting of the American Society of Clinical Oncology (ASCO).
Ibrutinib, a BTK inhibitor, was approved as a single agent in 2015 for the treatment of Waldenström’s macroglobulinemia.* Clinical practice guidelines from the National Comprehensive Cancer Network (NCCN) list ibrutinib as an “other” recommended regimen, noting that study data show a “lower overall and absence of major responses” reported for MYD88 wild-type patients.Rituximab alone and in combination with other agents has clinical activity in Waldenström’s. Moreover, preclinical studies have shown that rituximab and ibrutinib have synergistic activity, Dr. Dimopoulos said, presenting data from the iNNOVATE Study Group and the European Consortium for Waldenström’s Macroglobulinemia that was published concurrently in the New England Journal of Medicine.
They initiated a study of 150 symptomatic patients randomized to either ibrutinib plus rituximab or placebo plus rituximab, with a primary end point of progression-free survival.
They also assessed mutational status of MYD88 and CXCR4 in bone marrow samples, given that previous data suggest outcomes of ibrutinib treatment for this disease depend on MYD88 and CXCR4 mutational status, they said in the report.
Progression-free survival at 30 months was 82% for ibrutinib plus rituximab, compared to 28% for placebo plus rituximab (hazard ratio for progression or death, 0.20; P less than 0.001). Further, ibrutinib and rituximab had a benefit that was independent of the MYD88 or CXCR4 genotype, the investigators reported.
“Response rates with ibrutinib–rituximab were similar across different CXCR4 genotypes, but were slightly lower among patients who did not have the activating MYD88 L265P mutation, which triggers the growth of malignant cells through BTK and hematopoietic-cell kinase, both of which are targeted by ibrutinib,” they wrote.
Atrial fibrillation of grade 3 or higher occurred in 12% of the ibrutinib-rituximab and in 1% of placebo-rituximab groups. In the ibrutinib-rituximab group, the majority of these cases occurred in patients older than 75 years of age, Dr. Dimopoulos reported at ASCO.
Grade 3 or greater hypertension occurred in 13% and 4% of the ibrutinib-rituximab and placebo-rituximab groups respectively, while respiratory tract infections occurred in 4% and 0%. Conversely, the ibrutinib-rituximab arm had a lower rate of grade 3 or greater infusion reactions (1% vs. 16%), he said.
Based on this report, ibrutinib plus rituximab is “appealing, primarily in MYD88-mutated patients, which is the vast majority of Waldenström’s patients,” said Craig Hofmeister, MD, MPH, who commented on the study in a ASCO presentation discussing the results of this trial.
Dr. Hofmeister, of Winship Cancer Institute, Emory University, Atlanta, noted the numerically lower response rate in patients without the MYD88 mutation “makes me wonder whether I would want to have ibrutinib if I was MYD88 wild type.”
Atrial fibrillation and infections on the ibrutinib-rituximab regimen may require close monitoring and should be managed appropriately, he added. “Atrial fibrillation seems to be a consistent theme with ibrutinib, certainly in patients who are older and have probably more heart disease.”
The study (NCT02165397) was funded by Pharmacyclics and Janssen Research and Development. Dr. Dimopoulos reported personal fees from Amgen, Celgene, Janssen, and Takeda, outside of the submitted work. Co-authors reported disclosures related to Pharmacyclics, Bristol-Myers Squibb, Gilead, Roche, and AbbVie, among others. Dr. Hofmeister had no relevant financial disclosures.
SOURCE: Dimopoulos MA, et al. N Engl J Med. 2018 Jun 1. ASCO Abstract 8003.
*Correction, 8/27/2018: An earlier version of this story misstated the date of ibrutinib's approval as a single agent in the treatment of Waldenström’s .
, results of a randomized phase 3 trial show.
The results make ibrutinib plus rituximab “a new standard of care” for the disease, said investigator Meletios A. Dimopoulos, MD, with the Department of Clinical Therapeutics, National and Kapodistrian University of Athens, Greece.
“This is a combination with remarkable activity as far as progression-free survival is concerned, and well tolerated,” Dr. Dimopoulos said in a presentation of the data here at the 2018 annual meeting of the American Society of Clinical Oncology (ASCO).
Ibrutinib, a BTK inhibitor, was approved as a single agent in 2015 for the treatment of Waldenström’s macroglobulinemia.* Clinical practice guidelines from the National Comprehensive Cancer Network (NCCN) list ibrutinib as an “other” recommended regimen, noting that study data show a “lower overall and absence of major responses” reported for MYD88 wild-type patients.Rituximab alone and in combination with other agents has clinical activity in Waldenström’s. Moreover, preclinical studies have shown that rituximab and ibrutinib have synergistic activity, Dr. Dimopoulos said, presenting data from the iNNOVATE Study Group and the European Consortium for Waldenström’s Macroglobulinemia that was published concurrently in the New England Journal of Medicine.
They initiated a study of 150 symptomatic patients randomized to either ibrutinib plus rituximab or placebo plus rituximab, with a primary end point of progression-free survival.
They also assessed mutational status of MYD88 and CXCR4 in bone marrow samples, given that previous data suggest outcomes of ibrutinib treatment for this disease depend on MYD88 and CXCR4 mutational status, they said in the report.
Progression-free survival at 30 months was 82% for ibrutinib plus rituximab, compared to 28% for placebo plus rituximab (hazard ratio for progression or death, 0.20; P less than 0.001). Further, ibrutinib and rituximab had a benefit that was independent of the MYD88 or CXCR4 genotype, the investigators reported.
“Response rates with ibrutinib–rituximab were similar across different CXCR4 genotypes, but were slightly lower among patients who did not have the activating MYD88 L265P mutation, which triggers the growth of malignant cells through BTK and hematopoietic-cell kinase, both of which are targeted by ibrutinib,” they wrote.
Atrial fibrillation of grade 3 or higher occurred in 12% of the ibrutinib-rituximab and in 1% of placebo-rituximab groups. In the ibrutinib-rituximab group, the majority of these cases occurred in patients older than 75 years of age, Dr. Dimopoulos reported at ASCO.
Grade 3 or greater hypertension occurred in 13% and 4% of the ibrutinib-rituximab and placebo-rituximab groups respectively, while respiratory tract infections occurred in 4% and 0%. Conversely, the ibrutinib-rituximab arm had a lower rate of grade 3 or greater infusion reactions (1% vs. 16%), he said.
Based on this report, ibrutinib plus rituximab is “appealing, primarily in MYD88-mutated patients, which is the vast majority of Waldenström’s patients,” said Craig Hofmeister, MD, MPH, who commented on the study in a ASCO presentation discussing the results of this trial.
Dr. Hofmeister, of Winship Cancer Institute, Emory University, Atlanta, noted the numerically lower response rate in patients without the MYD88 mutation “makes me wonder whether I would want to have ibrutinib if I was MYD88 wild type.”
Atrial fibrillation and infections on the ibrutinib-rituximab regimen may require close monitoring and should be managed appropriately, he added. “Atrial fibrillation seems to be a consistent theme with ibrutinib, certainly in patients who are older and have probably more heart disease.”
The study (NCT02165397) was funded by Pharmacyclics and Janssen Research and Development. Dr. Dimopoulos reported personal fees from Amgen, Celgene, Janssen, and Takeda, outside of the submitted work. Co-authors reported disclosures related to Pharmacyclics, Bristol-Myers Squibb, Gilead, Roche, and AbbVie, among others. Dr. Hofmeister had no relevant financial disclosures.
SOURCE: Dimopoulos MA, et al. N Engl J Med. 2018 Jun 1. ASCO Abstract 8003.
*Correction, 8/27/2018: An earlier version of this story misstated the date of ibrutinib's approval as a single agent in the treatment of Waldenström’s .
REPORTING FROM ASCO 2018
Key clinical point: Adding ibrutinib to rituximab treatment improved progression-free survival in patients with Waldenström’s macroglobulinemia.
Major finding: The rate of 30-month progression-free survival was 82% for the combination of ibrutinib plus rituximab, as compared with 28% for placebo plus rituximab (hazard ratio, 0.20; P less than 0.001).
Study details: A phase 3 trial including 150 symptomatic patients with Waldenström’s macroglobulinemia who had received no previous treatment or had disease recurrence.
Disclosures: The study (NCT02165397) was funded by Pharmacyclics and Janssen Research and Development. Dr. Dimopoulos reported personal fees from Amgen, Celgene, Janssen, and Takeda, outside of the submitted work. Co-authors reported disclosures related to Pharmacyclics, Bristol-Myers Squibb, Gilead, Roche, and AbbVie, among others. Dr. Hofmeister had no relevant financial disclosures.
Source: Dimopoulos MA, et al. N Engl J Med. 2018 Jun 1. ASCO Abstract 8003.
Overcoming TP53 mutation proves difficult in MCL
, new findings suggest.
“TP53 mutated MCL remains a major challenge, and our results underline the importance of molecular profiling, including TP53 status, in future trials exploring novel agents,” wrote Christian Winther Eskelund, MD, of Rigshospitalet in Copenhagen, and his colleagues. The findings were published in Haematologica.
The researchers noted that the results will need validation in a larger cohort of patients.
They performed an analysis of 50 MCL patients who enrolled in the Nordic MCL4 trial between 2009 and 2013. Patients were either over age 65 years or were younger but unfit for autologous stem cell transplantation. Despite the addition of lenalidomide to the chemoimmunotherapy regimen, patients with TP53 mutations had worse overall and progression-free survival and were significantly quicker to experience relapse.
After a median follow up of 47 months, median overall survival was 25 months for patients with TP53 mutations, compared with 69 months for those without (P less than .0001). Similarly, median progression-free survival was 10 months in patients with the mutation, compared with 42 months in patients without it (P = .001). Time to relapse was a median of 10 months in these mutated patients, compared with 58 months for unmutated MCL patients (P less than .0001).
TP53 mutations were identified in six patients (14%), one of whom withdrew consent at day 28 of the study. Of the remaining patients with mutations, all of them either progressed or relapsed during the study and none were alive at the most recent follow-up. During the study, patients received an induction phase of six cycles of lenalidomide plus bendamustine-rituximab (weeks 1-24), followed by a maintenance phase of lenalidomide (weeks 25-56).
SOURCE: Eskelund CW et al. Haematologica. 2018 May 24. doi: 10.3324/haematol.2018.194399.
, new findings suggest.
“TP53 mutated MCL remains a major challenge, and our results underline the importance of molecular profiling, including TP53 status, in future trials exploring novel agents,” wrote Christian Winther Eskelund, MD, of Rigshospitalet in Copenhagen, and his colleagues. The findings were published in Haematologica.
The researchers noted that the results will need validation in a larger cohort of patients.
They performed an analysis of 50 MCL patients who enrolled in the Nordic MCL4 trial between 2009 and 2013. Patients were either over age 65 years or were younger but unfit for autologous stem cell transplantation. Despite the addition of lenalidomide to the chemoimmunotherapy regimen, patients with TP53 mutations had worse overall and progression-free survival and were significantly quicker to experience relapse.
After a median follow up of 47 months, median overall survival was 25 months for patients with TP53 mutations, compared with 69 months for those without (P less than .0001). Similarly, median progression-free survival was 10 months in patients with the mutation, compared with 42 months in patients without it (P = .001). Time to relapse was a median of 10 months in these mutated patients, compared with 58 months for unmutated MCL patients (P less than .0001).
TP53 mutations were identified in six patients (14%), one of whom withdrew consent at day 28 of the study. Of the remaining patients with mutations, all of them either progressed or relapsed during the study and none were alive at the most recent follow-up. During the study, patients received an induction phase of six cycles of lenalidomide plus bendamustine-rituximab (weeks 1-24), followed by a maintenance phase of lenalidomide (weeks 25-56).
SOURCE: Eskelund CW et al. Haematologica. 2018 May 24. doi: 10.3324/haematol.2018.194399.
, new findings suggest.
“TP53 mutated MCL remains a major challenge, and our results underline the importance of molecular profiling, including TP53 status, in future trials exploring novel agents,” wrote Christian Winther Eskelund, MD, of Rigshospitalet in Copenhagen, and his colleagues. The findings were published in Haematologica.
The researchers noted that the results will need validation in a larger cohort of patients.
They performed an analysis of 50 MCL patients who enrolled in the Nordic MCL4 trial between 2009 and 2013. Patients were either over age 65 years or were younger but unfit for autologous stem cell transplantation. Despite the addition of lenalidomide to the chemoimmunotherapy regimen, patients with TP53 mutations had worse overall and progression-free survival and were significantly quicker to experience relapse.
After a median follow up of 47 months, median overall survival was 25 months for patients with TP53 mutations, compared with 69 months for those without (P less than .0001). Similarly, median progression-free survival was 10 months in patients with the mutation, compared with 42 months in patients without it (P = .001). Time to relapse was a median of 10 months in these mutated patients, compared with 58 months for unmutated MCL patients (P less than .0001).
TP53 mutations were identified in six patients (14%), one of whom withdrew consent at day 28 of the study. Of the remaining patients with mutations, all of them either progressed or relapsed during the study and none were alive at the most recent follow-up. During the study, patients received an induction phase of six cycles of lenalidomide plus bendamustine-rituximab (weeks 1-24), followed by a maintenance phase of lenalidomide (weeks 25-56).
SOURCE: Eskelund CW et al. Haematologica. 2018 May 24. doi: 10.3324/haematol.2018.194399.
FROM HAEMATOLOGICA
Older CLL and NHL patients are more vulnerable to toxicities
Older patients with chronic lymphocytic leukemia (CLL) and non-Hodgkin lymphoma (NHL) have an increased odds of experiencing treatment-related toxicities, even when treated with novel agents, according to findings reported in the Journal of Geriatric Oncology.
An analysis of 1,199 patients showed that CLL patients aged 65 years and older had significantly higher odds than younger patients of developing a grade three or four hematologic toxicity (adjusted odds ratio, 1.70; P = .009; 95% confidence interval, 1.57-1.84) or nonhematologic toxicity (OR, 1.47; P = .022; 95% CI, 1.39-1.55).
Investigators analyzed data obtained from the Alliance for Clinical Trials in Oncology to compare the incidence of toxicities between age groups. Of the 1,199 patients included in the analysis, 409 were 65 years of age or older, and 790 were younger than 65 years. Among these patients, 438 received only therapy with novel agents including biologic combinations, monoclonal antibodies, cell cycle inhibitors, chemoimmunotherapy, and immunomodulators, and 761 received novel agents in addition to chemotherapy.
Overall, 68% of CLL patients and 35% of NHL patients had at least one grade three or four hematologic toxicity, compared with 48% and 54% for nonhematologic toxicities, respectively.
Older CLL patients with at least one grade three or four toxicity in the first 3 months had similar overall survival (OS) and progression-free survival (PFS) as those without a toxicity. In contrast, older NHL patients with at least one grade three or four hematologic toxicity in the first 3 months had worse OS (HR, 3.14; P = .006; 95% CI, 2.25-4.39) and PFS (HR, 3.06; P = .011; 95% CI, 2.10-4.45) than patients without these toxicities. Nonhematologic toxicities were not significantly associated with survival outcomes for patients with NHL.
“The observed associations between hematologic toxicity and OS/PFS among older patients with NHL require further investigation,” the researchers wrote. “These findings could represent a direct effect of toxicity due to decreased physiologic reserve, decreased drug clearance, or an increased sensitivity of tissue to novel agents.”
The study was supported by a National Institutes of Health grant. Researchers reported relationships with Bartlett, KITE, Pfizer, Seattle Genetics, Roche-Genentech, Celgene, Pharmacyclics, and Gilead.
SOURCE: Tallarico M et al. J Geriatr Oncol. 2018 Apr 16. pii: S1879-4068(18)30131-0.
Older patients with chronic lymphocytic leukemia (CLL) and non-Hodgkin lymphoma (NHL) have an increased odds of experiencing treatment-related toxicities, even when treated with novel agents, according to findings reported in the Journal of Geriatric Oncology.
An analysis of 1,199 patients showed that CLL patients aged 65 years and older had significantly higher odds than younger patients of developing a grade three or four hematologic toxicity (adjusted odds ratio, 1.70; P = .009; 95% confidence interval, 1.57-1.84) or nonhematologic toxicity (OR, 1.47; P = .022; 95% CI, 1.39-1.55).
Investigators analyzed data obtained from the Alliance for Clinical Trials in Oncology to compare the incidence of toxicities between age groups. Of the 1,199 patients included in the analysis, 409 were 65 years of age or older, and 790 were younger than 65 years. Among these patients, 438 received only therapy with novel agents including biologic combinations, monoclonal antibodies, cell cycle inhibitors, chemoimmunotherapy, and immunomodulators, and 761 received novel agents in addition to chemotherapy.
Overall, 68% of CLL patients and 35% of NHL patients had at least one grade three or four hematologic toxicity, compared with 48% and 54% for nonhematologic toxicities, respectively.
Older CLL patients with at least one grade three or four toxicity in the first 3 months had similar overall survival (OS) and progression-free survival (PFS) as those without a toxicity. In contrast, older NHL patients with at least one grade three or four hematologic toxicity in the first 3 months had worse OS (HR, 3.14; P = .006; 95% CI, 2.25-4.39) and PFS (HR, 3.06; P = .011; 95% CI, 2.10-4.45) than patients without these toxicities. Nonhematologic toxicities were not significantly associated with survival outcomes for patients with NHL.
“The observed associations between hematologic toxicity and OS/PFS among older patients with NHL require further investigation,” the researchers wrote. “These findings could represent a direct effect of toxicity due to decreased physiologic reserve, decreased drug clearance, or an increased sensitivity of tissue to novel agents.”
The study was supported by a National Institutes of Health grant. Researchers reported relationships with Bartlett, KITE, Pfizer, Seattle Genetics, Roche-Genentech, Celgene, Pharmacyclics, and Gilead.
SOURCE: Tallarico M et al. J Geriatr Oncol. 2018 Apr 16. pii: S1879-4068(18)30131-0.
Older patients with chronic lymphocytic leukemia (CLL) and non-Hodgkin lymphoma (NHL) have an increased odds of experiencing treatment-related toxicities, even when treated with novel agents, according to findings reported in the Journal of Geriatric Oncology.
An analysis of 1,199 patients showed that CLL patients aged 65 years and older had significantly higher odds than younger patients of developing a grade three or four hematologic toxicity (adjusted odds ratio, 1.70; P = .009; 95% confidence interval, 1.57-1.84) or nonhematologic toxicity (OR, 1.47; P = .022; 95% CI, 1.39-1.55).
Investigators analyzed data obtained from the Alliance for Clinical Trials in Oncology to compare the incidence of toxicities between age groups. Of the 1,199 patients included in the analysis, 409 were 65 years of age or older, and 790 were younger than 65 years. Among these patients, 438 received only therapy with novel agents including biologic combinations, monoclonal antibodies, cell cycle inhibitors, chemoimmunotherapy, and immunomodulators, and 761 received novel agents in addition to chemotherapy.
Overall, 68% of CLL patients and 35% of NHL patients had at least one grade three or four hematologic toxicity, compared with 48% and 54% for nonhematologic toxicities, respectively.
Older CLL patients with at least one grade three or four toxicity in the first 3 months had similar overall survival (OS) and progression-free survival (PFS) as those without a toxicity. In contrast, older NHL patients with at least one grade three or four hematologic toxicity in the first 3 months had worse OS (HR, 3.14; P = .006; 95% CI, 2.25-4.39) and PFS (HR, 3.06; P = .011; 95% CI, 2.10-4.45) than patients without these toxicities. Nonhematologic toxicities were not significantly associated with survival outcomes for patients with NHL.
“The observed associations between hematologic toxicity and OS/PFS among older patients with NHL require further investigation,” the researchers wrote. “These findings could represent a direct effect of toxicity due to decreased physiologic reserve, decreased drug clearance, or an increased sensitivity of tissue to novel agents.”
The study was supported by a National Institutes of Health grant. Researchers reported relationships with Bartlett, KITE, Pfizer, Seattle Genetics, Roche-Genentech, Celgene, Pharmacyclics, and Gilead.
SOURCE: Tallarico M et al. J Geriatr Oncol. 2018 Apr 16. pii: S1879-4068(18)30131-0.
FROM THE JOURNAL OF GERIATRIC ONCOLOGY
Key clinical point:
Major finding: Older CLL patients had significantly higher odds of developing a grade three or four hematologic toxicity (adjusted odds ratio, 1.70; P = .009; 95% confidence interval, 1.57-1.84) or nonhematologic toxicity (OR, 1.47; P = .022; 95% CI 1.39-1.55).
Study details: An analysis of data from 1,199 CLL and NHL patients in the Alliance for Clinical Trials in Oncology.
Disclosures: The study was supported by a National Institutes of Health grant. Researchers reported relationships with Bartlett, KITE, Pfizer, Seattle Genetics, Roche-Genentech, Celgene, Pharmacyclics, and Gilead.
Source: Tallarico M et al. J Geriatr Oncol. 2018 Apr 16. pii: S1879-4068(18)30131-0.
Physical inactivity linked to lymphoma risk
A lifetime of physical inactivity could significantly increase the risk of developing both Hodgkin and non-Hodgkin lymphoma, according to a case-control study.
Researchers examined self-reported lifetime physical activity in 87 patients with Hodgkin lymphoma and 236 patients with non-Hodgkin lymphoma, as well as 1,300 cancer-free controls.
Researchers also found a nearly threefold higher risk of Hodgkin lymphoma among overweight and obese individuals who were physically inactive, compared with those who were active (odds ratio, 2.79; P = .01). Similarly, physically inactive individuals who had never smoked had a greater than threefold increase in risk, compared with never-smokers who were active (OR, 3.30; P less than .001). But despite these significant associations for smoking and weight, the small samples sizes meant they were not significant in multivariable-adjusted models, the researchers noted. For non-Hodgkin lymphoma, the associations between obesity/overweight and smoking status were also not statistically significant in multivariable-adjusted models.
Previous studies looking at the role of physical activity in Hodgkin and non-Hodgkin lymphoma had yielded mixed and inconclusive results. Since then, researchers have begun to specifically consider the role of physical inactivity, rather than physical activity, as the exposure of interest.
“An additional advantage of identifying inactivity as the exposure of interest is that a body of literature suggests that those who are at the lower end of the physical activity continuum are less likely to overreport physical activity than those who engage in greater levels of physical activity,” the researchers wrote.
They acknowledged that relying on self-reported levels of physical activity was a limitation of their study. However, they also pointed out that previous research suggested that simplified physical activity questionnaires that took a dichotomous approach to activity/inactivity were effective at identifying the most physically inactivity individuals in a population.
“Continued evidence for adverse associations between physical inactivity and cancer endpoints substantiate a powerful public health message that any amount of regular activity appears to associate with decreased cancer risk,” they wrote.
One researcher was supported by the New York State Department of Health. No conflicts of interest were reported.
SOURCE: Etter JL et al. Leuk Res. 2018 Mar 27;69:7-11.
A lifetime of physical inactivity could significantly increase the risk of developing both Hodgkin and non-Hodgkin lymphoma, according to a case-control study.
Researchers examined self-reported lifetime physical activity in 87 patients with Hodgkin lymphoma and 236 patients with non-Hodgkin lymphoma, as well as 1,300 cancer-free controls.
Researchers also found a nearly threefold higher risk of Hodgkin lymphoma among overweight and obese individuals who were physically inactive, compared with those who were active (odds ratio, 2.79; P = .01). Similarly, physically inactive individuals who had never smoked had a greater than threefold increase in risk, compared with never-smokers who were active (OR, 3.30; P less than .001). But despite these significant associations for smoking and weight, the small samples sizes meant they were not significant in multivariable-adjusted models, the researchers noted. For non-Hodgkin lymphoma, the associations between obesity/overweight and smoking status were also not statistically significant in multivariable-adjusted models.
Previous studies looking at the role of physical activity in Hodgkin and non-Hodgkin lymphoma had yielded mixed and inconclusive results. Since then, researchers have begun to specifically consider the role of physical inactivity, rather than physical activity, as the exposure of interest.
“An additional advantage of identifying inactivity as the exposure of interest is that a body of literature suggests that those who are at the lower end of the physical activity continuum are less likely to overreport physical activity than those who engage in greater levels of physical activity,” the researchers wrote.
They acknowledged that relying on self-reported levels of physical activity was a limitation of their study. However, they also pointed out that previous research suggested that simplified physical activity questionnaires that took a dichotomous approach to activity/inactivity were effective at identifying the most physically inactivity individuals in a population.
“Continued evidence for adverse associations between physical inactivity and cancer endpoints substantiate a powerful public health message that any amount of regular activity appears to associate with decreased cancer risk,” they wrote.
One researcher was supported by the New York State Department of Health. No conflicts of interest were reported.
SOURCE: Etter JL et al. Leuk Res. 2018 Mar 27;69:7-11.
A lifetime of physical inactivity could significantly increase the risk of developing both Hodgkin and non-Hodgkin lymphoma, according to a case-control study.
Researchers examined self-reported lifetime physical activity in 87 patients with Hodgkin lymphoma and 236 patients with non-Hodgkin lymphoma, as well as 1,300 cancer-free controls.
Researchers also found a nearly threefold higher risk of Hodgkin lymphoma among overweight and obese individuals who were physically inactive, compared with those who were active (odds ratio, 2.79; P = .01). Similarly, physically inactive individuals who had never smoked had a greater than threefold increase in risk, compared with never-smokers who were active (OR, 3.30; P less than .001). But despite these significant associations for smoking and weight, the small samples sizes meant they were not significant in multivariable-adjusted models, the researchers noted. For non-Hodgkin lymphoma, the associations between obesity/overweight and smoking status were also not statistically significant in multivariable-adjusted models.
Previous studies looking at the role of physical activity in Hodgkin and non-Hodgkin lymphoma had yielded mixed and inconclusive results. Since then, researchers have begun to specifically consider the role of physical inactivity, rather than physical activity, as the exposure of interest.
“An additional advantage of identifying inactivity as the exposure of interest is that a body of literature suggests that those who are at the lower end of the physical activity continuum are less likely to overreport physical activity than those who engage in greater levels of physical activity,” the researchers wrote.
They acknowledged that relying on self-reported levels of physical activity was a limitation of their study. However, they also pointed out that previous research suggested that simplified physical activity questionnaires that took a dichotomous approach to activity/inactivity were effective at identifying the most physically inactivity individuals in a population.
“Continued evidence for adverse associations between physical inactivity and cancer endpoints substantiate a powerful public health message that any amount of regular activity appears to associate with decreased cancer risk,” they wrote.
One researcher was supported by the New York State Department of Health. No conflicts of interest were reported.
SOURCE: Etter JL et al. Leuk Res. 2018 Mar 27;69:7-11.
FROM LEUKEMIA RESEARCH
Key clinical point:
Major finding: A lifetime of physical inactivity is associated with a 90% increased risk of Hodgkin lymphoma and a 35% increased risk of non-Hodgkin lymphoma.Study details: A case-control study in 323 patients with Hodgkin or non-Hodgkin lymphoma and 1,300 cancer-free controls.
Disclosures: One researcher was supported by the New York State Department of Health. No conflicts of interest were declared.
Source: Etter JL et al. Leuk Res. 2018 Mar 27;69:7-11.
Pediatric cancers are on the rise
PITTSBURGH – The incidence of many pediatric cancers are on the rise, and the increase is occurring in nearly all demographic groups studied, according to the latest data from the U.S. Centers for Disease Control and Prevention.
Pediatric cancers that increased significantly in incidence from 2001 through 2014, compared with previous time periods, include thyroid carcinoma, hepatic tumors, lymphomas, renal tumors, and brain tumors. Other cancer types remained unchanged, except malignant melanoma, which saw a significant decline in incidence over the same period, reported David A. Siegel, MD, of the Epidemic Intelligence Service at the CDC in Atlanta.
Recent studies of trends in pediatric cancer have either used data from before 2010 or covered less than a third of the U.S. population, the investigators noted.
To get a more accurate estimate of current trends, the investigators relied on the United States Cancer Statistics, which combines data from the Surveillance, Epidemiology, and End Results (SEER) program and the National Program of Cancer Registries. Together, the combined databases cover 100% of the U.S. population.
Dr. Siegel and his colleagues looked at cancer incidence rates and trends among individuals younger than 20 years of age from across 48 states from 2001 to 2014 – Mississippi, Nevada, and the District of Columbia were not included.
They used a joinpoint regression method to calculate average annual percent change (AAPC) in rates, then stratified rates and trends by sex, age, and race/ethnicity; location; economic status; and cancer type.
During the 14-year period of the study, there were a total of 196,200 incident cases of pediatric cancer, for an overall cancer incidence rate of 173 per million. The pediatric cancer with the highest incident rate was leukemia of any type (45.6 per million), brain tumors (30.8), and lymphomas (26.0).
Incidence rates were highest among males, patients from infancy through age 4, non-Hispanic whites, children who live in the Northeast region, those who live in the wealthiest counties, and those who live in urban/metropolitan counties. The overall pediatric cancer incidence rate increased, with an AAPC of 0.7 (95% confidence interval, 0.5-0.8).
“Rates increased in each stratum of sex, age, and race/ethnicity (except non-Hispanic American Indian/Alaska Native), region, economic status, and rural/urban classification,” the investigators wrote.
Cancers with significantly increased AAPC included thyroid carcinomas (AAPC, 4.8), hepatic tumors (2.5), lymphomas (1.7), renal tumors (0.6), and brain tumors (all types, 0.4).
There were no significant changes in the incidence of either germ cell cancer, retinoblastoma, leukemia, neuroblastoma, soft-tissue sarcomas, or bone tumors.
The only significant decrease over the study period was in the incidence of melanoma in children (–2.6).
“Possible causes of increasing rates might include changes in diagnostic, coding, and reporting standards, increased detection, population-based changes (such as increasing obesity), and environmental exposures,” they wrote.
Public health campaigns about the dangers of UV exposure and promoting the use of sunscreens may account for the decline in the incidence of malignant melanoma, they suggested.
The study was supported by the CDC. Dr. Siegel and coauthors are CDC employees. They reported having no conflicts of interest.
SOURCE: Siegel DA et al. ASPHO 2018, Abstract 605.
PITTSBURGH – The incidence of many pediatric cancers are on the rise, and the increase is occurring in nearly all demographic groups studied, according to the latest data from the U.S. Centers for Disease Control and Prevention.
Pediatric cancers that increased significantly in incidence from 2001 through 2014, compared with previous time periods, include thyroid carcinoma, hepatic tumors, lymphomas, renal tumors, and brain tumors. Other cancer types remained unchanged, except malignant melanoma, which saw a significant decline in incidence over the same period, reported David A. Siegel, MD, of the Epidemic Intelligence Service at the CDC in Atlanta.
Recent studies of trends in pediatric cancer have either used data from before 2010 or covered less than a third of the U.S. population, the investigators noted.
To get a more accurate estimate of current trends, the investigators relied on the United States Cancer Statistics, which combines data from the Surveillance, Epidemiology, and End Results (SEER) program and the National Program of Cancer Registries. Together, the combined databases cover 100% of the U.S. population.
Dr. Siegel and his colleagues looked at cancer incidence rates and trends among individuals younger than 20 years of age from across 48 states from 2001 to 2014 – Mississippi, Nevada, and the District of Columbia were not included.
They used a joinpoint regression method to calculate average annual percent change (AAPC) in rates, then stratified rates and trends by sex, age, and race/ethnicity; location; economic status; and cancer type.
During the 14-year period of the study, there were a total of 196,200 incident cases of pediatric cancer, for an overall cancer incidence rate of 173 per million. The pediatric cancer with the highest incident rate was leukemia of any type (45.6 per million), brain tumors (30.8), and lymphomas (26.0).
Incidence rates were highest among males, patients from infancy through age 4, non-Hispanic whites, children who live in the Northeast region, those who live in the wealthiest counties, and those who live in urban/metropolitan counties. The overall pediatric cancer incidence rate increased, with an AAPC of 0.7 (95% confidence interval, 0.5-0.8).
“Rates increased in each stratum of sex, age, and race/ethnicity (except non-Hispanic American Indian/Alaska Native), region, economic status, and rural/urban classification,” the investigators wrote.
Cancers with significantly increased AAPC included thyroid carcinomas (AAPC, 4.8), hepatic tumors (2.5), lymphomas (1.7), renal tumors (0.6), and brain tumors (all types, 0.4).
There were no significant changes in the incidence of either germ cell cancer, retinoblastoma, leukemia, neuroblastoma, soft-tissue sarcomas, or bone tumors.
The only significant decrease over the study period was in the incidence of melanoma in children (–2.6).
“Possible causes of increasing rates might include changes in diagnostic, coding, and reporting standards, increased detection, population-based changes (such as increasing obesity), and environmental exposures,” they wrote.
Public health campaigns about the dangers of UV exposure and promoting the use of sunscreens may account for the decline in the incidence of malignant melanoma, they suggested.
The study was supported by the CDC. Dr. Siegel and coauthors are CDC employees. They reported having no conflicts of interest.
SOURCE: Siegel DA et al. ASPHO 2018, Abstract 605.
PITTSBURGH – The incidence of many pediatric cancers are on the rise, and the increase is occurring in nearly all demographic groups studied, according to the latest data from the U.S. Centers for Disease Control and Prevention.
Pediatric cancers that increased significantly in incidence from 2001 through 2014, compared with previous time periods, include thyroid carcinoma, hepatic tumors, lymphomas, renal tumors, and brain tumors. Other cancer types remained unchanged, except malignant melanoma, which saw a significant decline in incidence over the same period, reported David A. Siegel, MD, of the Epidemic Intelligence Service at the CDC in Atlanta.
Recent studies of trends in pediatric cancer have either used data from before 2010 or covered less than a third of the U.S. population, the investigators noted.
To get a more accurate estimate of current trends, the investigators relied on the United States Cancer Statistics, which combines data from the Surveillance, Epidemiology, and End Results (SEER) program and the National Program of Cancer Registries. Together, the combined databases cover 100% of the U.S. population.
Dr. Siegel and his colleagues looked at cancer incidence rates and trends among individuals younger than 20 years of age from across 48 states from 2001 to 2014 – Mississippi, Nevada, and the District of Columbia were not included.
They used a joinpoint regression method to calculate average annual percent change (AAPC) in rates, then stratified rates and trends by sex, age, and race/ethnicity; location; economic status; and cancer type.
During the 14-year period of the study, there were a total of 196,200 incident cases of pediatric cancer, for an overall cancer incidence rate of 173 per million. The pediatric cancer with the highest incident rate was leukemia of any type (45.6 per million), brain tumors (30.8), and lymphomas (26.0).
Incidence rates were highest among males, patients from infancy through age 4, non-Hispanic whites, children who live in the Northeast region, those who live in the wealthiest counties, and those who live in urban/metropolitan counties. The overall pediatric cancer incidence rate increased, with an AAPC of 0.7 (95% confidence interval, 0.5-0.8).
“Rates increased in each stratum of sex, age, and race/ethnicity (except non-Hispanic American Indian/Alaska Native), region, economic status, and rural/urban classification,” the investigators wrote.
Cancers with significantly increased AAPC included thyroid carcinomas (AAPC, 4.8), hepatic tumors (2.5), lymphomas (1.7), renal tumors (0.6), and brain tumors (all types, 0.4).
There were no significant changes in the incidence of either germ cell cancer, retinoblastoma, leukemia, neuroblastoma, soft-tissue sarcomas, or bone tumors.
The only significant decrease over the study period was in the incidence of melanoma in children (–2.6).
“Possible causes of increasing rates might include changes in diagnostic, coding, and reporting standards, increased detection, population-based changes (such as increasing obesity), and environmental exposures,” they wrote.
Public health campaigns about the dangers of UV exposure and promoting the use of sunscreens may account for the decline in the incidence of malignant melanoma, they suggested.
The study was supported by the CDC. Dr. Siegel and coauthors are CDC employees. They reported having no conflicts of interest.
SOURCE: Siegel DA et al. ASPHO 2018, Abstract 605.
REPORTING FROM ASPHO 2018
Key clinical point: Major finding: From 2001 to 2014, there were 196,200 incident cases of pediatric cancer for an overall cancer incidence rate of 173 per 1 million.
Study details: A review of data from the United States Cancer Statistics for children under age 20.
Disclosures: The CDC supported the study. Dr. Siegel and his coauthors are CDC employees. They reported having no conflicts of interest.
Source: Siegel DA et al. ASPHO 2018, Abstract 605.