User login
Increased sudden death risk in HIV linked to cardiac fibrosis
SEATTLE – A marked increase in the risk of sudden cardiac death among people with HIV correlates with a significantly higher burden of myocardial fibrosis, according to an autopsy study presented at the Conference on Retroviruses and Opportunistic Infections.
Fibrosis is a known trigger for fatal arrhythmias, so the take home is that fibrosis should be considered as a criteria for defibrillator implantation in HIV patients, said lead investigator Zian Tseng, MD, a cardiologist, cardiac electrophysiologist, and professor of medicine at the University of California, San Francisco.
The finding also speaks to a larger issue. The main criterion right now for implantation is an ejection fraction below 35%, but “there are a lot of people who die suddenly with normal ejection fractions,” and not just people with HIV, he said.
Many of those deaths might be prevented if fibrosis is added to implantation criteria. All that’s needed for assessment is a cardiac MRI, Dr. Tseng said.
The approach would be particularly fruitful for HIV patients, but cardiac fibrosis “isn’t just an” HIV problem, he said.
The conclusions have their roots in an investigation to determine the true incidence of sudden cardiac death (SCD) in the general public. SCD is commonly listed on death certificates, but it’s a presumed diagnosis, based on the best guesses of paramedics and clinicians. Autopsy is the only way to know for sure if a death was truly due to a sudden cardiac arrhythmia, or even related to the heart,
To clear the wheat from the chaff, Dr. Tseng and his colleagues performed autopsies on 525 out-of-hospital SCD cases among adults in San Francisco from 2011-2016; to qualify, the cases had to meet World Health Organization SCD criteria, meaning unexpected death within 1 hour of symptom onset, or, in unwitnessed cases, within 24 hours of when the person was last seen alive and well.
Cases were considered sudden arrhythmic death – and, therefore, true SCD – if no extracardiac causes of death or acute heart failure were found on autopsy. Overall, 40% of deaths attributed to SCD “were not sudden or unexpected, and nearly half of presumed SCDs were not arrhythmic.” The findings had “implications for ... mortality data, clinical trials, and cohort studies,” Dr. Tseng and his team concluded (Circulation. 2018 Jun 19;137[25]:2689-2700).
They next turned their attention to HIV. It’s known that the virus increases the risk of strokes, heart attacks, and heart failure; the researchers wanted to see if it did the same for SCD. The HIV results were presented at CROI.
Forty-seven presumed SCD cases with HIV met inclusion criteria during the study period. Based on the earlier findings and epidemiological data, people with HIV had more than an 80% higher risk of SCD and an almost 60% higher risk of confirmed arrhythmic death than did the general public. Similar to the general population, only about half of presumed SCD cases were confirmed on autopsy. About one-third of what turned out to be non-cardiac HIV deaths were due to occult overdose, versus 13.5% in the general population, which points to the increased need for drug screening and treatment in HIV.
Beyond that, though, the team found that the burden of myocardial fibrosis in HIV “was profound,” far surpassing what was found in SCD deaths in the general population. After adjustment for age, gender, and heart disease, “sudden cardiac deaths with HIV had 60% higher interstitial fibrosis by myocardial trichrome staining. Cardiac fibrosis, a known substrate for fatal arrhythmias in the general population, may underlie the mechanism by which HIV increases the risk” of sudden death in HIV, Dr. Tseng said.
It could be that the virus enters heart cells and sets off an inflammatory cardiomyopathy, or perhaps it’s related to chronic inflammation caused by the virus. Whatever the case, infection seems to have an “independent effect” on increasing fibrosis among people with HIV, he said.
Intriguingly, a large epidemiologic study in United States veterans, also presented at CROI, found a higher risk of SCD among HIV patients, but only if their infections were active over an extended period of time, as indicated by sustained high viral loads and low CD4 cell counts. Dr. Tseng was involved in that work, as well, but noted that the number of HIV SCD cases in the San Francisco study was too small to draw meaningful conclusions regarding the relationship between disease control and cardiac fibrosis.
Cardiac defibrillators can prevent arrhythmic death, so, at least for now, he said that the autopsy study findings mean that criteria for implantation should be broadened to include extensive cardiac fibrosis.
The work was funded by the National Institutes of Health. Dr. Tseng didn’t have any disclosures.
SOURCE: Tseng ZH et al. CROI 2019 abstract 32
SEATTLE – A marked increase in the risk of sudden cardiac death among people with HIV correlates with a significantly higher burden of myocardial fibrosis, according to an autopsy study presented at the Conference on Retroviruses and Opportunistic Infections.
Fibrosis is a known trigger for fatal arrhythmias, so the take home is that fibrosis should be considered as a criteria for defibrillator implantation in HIV patients, said lead investigator Zian Tseng, MD, a cardiologist, cardiac electrophysiologist, and professor of medicine at the University of California, San Francisco.
The finding also speaks to a larger issue. The main criterion right now for implantation is an ejection fraction below 35%, but “there are a lot of people who die suddenly with normal ejection fractions,” and not just people with HIV, he said.
Many of those deaths might be prevented if fibrosis is added to implantation criteria. All that’s needed for assessment is a cardiac MRI, Dr. Tseng said.
The approach would be particularly fruitful for HIV patients, but cardiac fibrosis “isn’t just an” HIV problem, he said.
The conclusions have their roots in an investigation to determine the true incidence of sudden cardiac death (SCD) in the general public. SCD is commonly listed on death certificates, but it’s a presumed diagnosis, based on the best guesses of paramedics and clinicians. Autopsy is the only way to know for sure if a death was truly due to a sudden cardiac arrhythmia, or even related to the heart,
To clear the wheat from the chaff, Dr. Tseng and his colleagues performed autopsies on 525 out-of-hospital SCD cases among adults in San Francisco from 2011-2016; to qualify, the cases had to meet World Health Organization SCD criteria, meaning unexpected death within 1 hour of symptom onset, or, in unwitnessed cases, within 24 hours of when the person was last seen alive and well.
Cases were considered sudden arrhythmic death – and, therefore, true SCD – if no extracardiac causes of death or acute heart failure were found on autopsy. Overall, 40% of deaths attributed to SCD “were not sudden or unexpected, and nearly half of presumed SCDs were not arrhythmic.” The findings had “implications for ... mortality data, clinical trials, and cohort studies,” Dr. Tseng and his team concluded (Circulation. 2018 Jun 19;137[25]:2689-2700).
They next turned their attention to HIV. It’s known that the virus increases the risk of strokes, heart attacks, and heart failure; the researchers wanted to see if it did the same for SCD. The HIV results were presented at CROI.
Forty-seven presumed SCD cases with HIV met inclusion criteria during the study period. Based on the earlier findings and epidemiological data, people with HIV had more than an 80% higher risk of SCD and an almost 60% higher risk of confirmed arrhythmic death than did the general public. Similar to the general population, only about half of presumed SCD cases were confirmed on autopsy. About one-third of what turned out to be non-cardiac HIV deaths were due to occult overdose, versus 13.5% in the general population, which points to the increased need for drug screening and treatment in HIV.
Beyond that, though, the team found that the burden of myocardial fibrosis in HIV “was profound,” far surpassing what was found in SCD deaths in the general population. After adjustment for age, gender, and heart disease, “sudden cardiac deaths with HIV had 60% higher interstitial fibrosis by myocardial trichrome staining. Cardiac fibrosis, a known substrate for fatal arrhythmias in the general population, may underlie the mechanism by which HIV increases the risk” of sudden death in HIV, Dr. Tseng said.
It could be that the virus enters heart cells and sets off an inflammatory cardiomyopathy, or perhaps it’s related to chronic inflammation caused by the virus. Whatever the case, infection seems to have an “independent effect” on increasing fibrosis among people with HIV, he said.
Intriguingly, a large epidemiologic study in United States veterans, also presented at CROI, found a higher risk of SCD among HIV patients, but only if their infections were active over an extended period of time, as indicated by sustained high viral loads and low CD4 cell counts. Dr. Tseng was involved in that work, as well, but noted that the number of HIV SCD cases in the San Francisco study was too small to draw meaningful conclusions regarding the relationship between disease control and cardiac fibrosis.
Cardiac defibrillators can prevent arrhythmic death, so, at least for now, he said that the autopsy study findings mean that criteria for implantation should be broadened to include extensive cardiac fibrosis.
The work was funded by the National Institutes of Health. Dr. Tseng didn’t have any disclosures.
SOURCE: Tseng ZH et al. CROI 2019 abstract 32
SEATTLE – A marked increase in the risk of sudden cardiac death among people with HIV correlates with a significantly higher burden of myocardial fibrosis, according to an autopsy study presented at the Conference on Retroviruses and Opportunistic Infections.
Fibrosis is a known trigger for fatal arrhythmias, so the take home is that fibrosis should be considered as a criteria for defibrillator implantation in HIV patients, said lead investigator Zian Tseng, MD, a cardiologist, cardiac electrophysiologist, and professor of medicine at the University of California, San Francisco.
The finding also speaks to a larger issue. The main criterion right now for implantation is an ejection fraction below 35%, but “there are a lot of people who die suddenly with normal ejection fractions,” and not just people with HIV, he said.
Many of those deaths might be prevented if fibrosis is added to implantation criteria. All that’s needed for assessment is a cardiac MRI, Dr. Tseng said.
The approach would be particularly fruitful for HIV patients, but cardiac fibrosis “isn’t just an” HIV problem, he said.
The conclusions have their roots in an investigation to determine the true incidence of sudden cardiac death (SCD) in the general public. SCD is commonly listed on death certificates, but it’s a presumed diagnosis, based on the best guesses of paramedics and clinicians. Autopsy is the only way to know for sure if a death was truly due to a sudden cardiac arrhythmia, or even related to the heart,
To clear the wheat from the chaff, Dr. Tseng and his colleagues performed autopsies on 525 out-of-hospital SCD cases among adults in San Francisco from 2011-2016; to qualify, the cases had to meet World Health Organization SCD criteria, meaning unexpected death within 1 hour of symptom onset, or, in unwitnessed cases, within 24 hours of when the person was last seen alive and well.
Cases were considered sudden arrhythmic death – and, therefore, true SCD – if no extracardiac causes of death or acute heart failure were found on autopsy. Overall, 40% of deaths attributed to SCD “were not sudden or unexpected, and nearly half of presumed SCDs were not arrhythmic.” The findings had “implications for ... mortality data, clinical trials, and cohort studies,” Dr. Tseng and his team concluded (Circulation. 2018 Jun 19;137[25]:2689-2700).
They next turned their attention to HIV. It’s known that the virus increases the risk of strokes, heart attacks, and heart failure; the researchers wanted to see if it did the same for SCD. The HIV results were presented at CROI.
Forty-seven presumed SCD cases with HIV met inclusion criteria during the study period. Based on the earlier findings and epidemiological data, people with HIV had more than an 80% higher risk of SCD and an almost 60% higher risk of confirmed arrhythmic death than did the general public. Similar to the general population, only about half of presumed SCD cases were confirmed on autopsy. About one-third of what turned out to be non-cardiac HIV deaths were due to occult overdose, versus 13.5% in the general population, which points to the increased need for drug screening and treatment in HIV.
Beyond that, though, the team found that the burden of myocardial fibrosis in HIV “was profound,” far surpassing what was found in SCD deaths in the general population. After adjustment for age, gender, and heart disease, “sudden cardiac deaths with HIV had 60% higher interstitial fibrosis by myocardial trichrome staining. Cardiac fibrosis, a known substrate for fatal arrhythmias in the general population, may underlie the mechanism by which HIV increases the risk” of sudden death in HIV, Dr. Tseng said.
It could be that the virus enters heart cells and sets off an inflammatory cardiomyopathy, or perhaps it’s related to chronic inflammation caused by the virus. Whatever the case, infection seems to have an “independent effect” on increasing fibrosis among people with HIV, he said.
Intriguingly, a large epidemiologic study in United States veterans, also presented at CROI, found a higher risk of SCD among HIV patients, but only if their infections were active over an extended period of time, as indicated by sustained high viral loads and low CD4 cell counts. Dr. Tseng was involved in that work, as well, but noted that the number of HIV SCD cases in the San Francisco study was too small to draw meaningful conclusions regarding the relationship between disease control and cardiac fibrosis.
Cardiac defibrillators can prevent arrhythmic death, so, at least for now, he said that the autopsy study findings mean that criteria for implantation should be broadened to include extensive cardiac fibrosis.
The work was funded by the National Institutes of Health. Dr. Tseng didn’t have any disclosures.
SOURCE: Tseng ZH et al. CROI 2019 abstract 32
REPORTING FROM CROI 2019
FDA examines changing donation policies for men who have sex with men
The
At a meeting of the FDA’s Blood Products Advisory Committee, the agency shared the content of the 5-item questionnaire and reviewed the proposed study design with committee members, who were asked to comment – but not vote – on the best path forward for MSM donation policies.
The FDA is “committed to ongoing evaluation of the MSM deferral policy” and remains open to adjusting the policy based on the best available scientific evidence, said Barbee Whitaker, PhD, a lead scientist in the agency’s Office of Emerging and Transfusion Transmitted Disease
After recruiting 2,000 men who have had sex with men at least once during the past 3 months, the study will aim to identify individuals who have very recently become HIV infected, in order to assess the discriminant function of the set of behavioral questions that are proposed in the questionnaire.
The crux of the problem currently, noted Dr. Whitaker, is identifying those individuals who are very recently infected with HIV. Nucleic acid testing has tightened the window of undetectability considerably, but the current 12-month deferral window after men have had sexual contact with other men is designed to ensure safety of the blood supply.
Social justice concerns have been raised about the blanket deferral, said Dr. Whitaker; the behavioral questions in the pilot study will ask about the number of different sexual partners men have had within the past 1, 3, and 12 months and ask about the type of sexual contact (oral sex, or anal penetrative or receptive intercourse). The questionnaire also asks about sex with a partner known to be HIV positive, condom use, and use of pre-exposure prophylaxis (PrEP).
The FDA will ask for proposals to conduct the study with an eye to having sites in such cities as Washington, Atlanta, and Miami, which have high incidences of HIV, to improve chances of early detection.
The behavioral questionnaire is not seen as an immediate replacement for the 12-month deferral policy, the FDA made clear in its briefing documents and in discussion with the committee. Instead, its utility will be in the information gleaned from the pilot study and a follow-on that may include several hundred thousand individuals. These data should provide “population-based evidence upon which to base regulatory decisions to ensure blood safety,” she said.
Donation policies outside the United States
Whether a change in blood donation deferral policies for MSM would be a shortened window or a move toward a behavioral questionnaire is currently not known. Globally, a variety of practices are used for blood screening, said Mindy Goldman, MD, medical director of Canadian Blood Services, who reviewed international perspectives on blood donation for MSM.
“There’s no general consensus on donation deferrals internationally,” she said. Factors influencing policy can include epidemiology, risk analysis, modeling, and history of response to threats in the past.
However, “there’s basically a couple of main approaches” to handling deferrals for MSM, Dr. Goldman said. One is time-based deferral – the strategy used in the United States, as well as Canada, the United Kingdom, Japan, and Australia.
Japan and the U.K. have recently moved to 3-month deferral periods, a figure arrived at by doubling the window period for nucleic acid testing for HIV, roughly, Dr. Goldman said. Early data from the U.K. experience has not shown an increase in HIV rates among donors, or an increase in NAT-only positive donors, she said. An application to move from a 12-month to a 3-month deferral period is pending in Canada.
A strong advantage of time-based deferral as a risk management strategy, Dr. Goldman said, is standardization. “For us, standardization is close to godliness.”
However, she added, “another major limitation is that you’re still deferring all sexually active MSM, including those who are in a stable monogamous relationship from donating. From a justice perspective for the lowest risk population of MSM – they are still being deferred using this type of approach.”
Some nations, such as Spain and Italy, use individual risk assessment via physician-led interviews. These approaches are often not standardized. “There’s no national uniform questionnaire, so there’s less standardization, and more variability between blood centers,” Dr. Goldman said. “So you wind up trying to compare apples with oranges.”
This means the results are harder to evaluate on a national level. However, there appears to be higher residual risk, with HIV rates among first-time donors approaching those of the general population, Dr. Goldman said.
Another strategy, used in France, is a test-retest model, where blood from first-time MSM that initially tests negative for HIV is held until the individual returns for re-testing or an additional donation, with a second negative test. This approach increases operational complexity and cost, noted Dr. Goldman, and because of the short shelf life of platelets, it’s not practical for this blood component.
In general questioning and discussion after this and other background presentations, the committee could agree on one point: this isn’t an easy question.
“I’m increasingly struck by how difficult this problem is,” said committee member Roger Lewis, MD, PhD, professor at the University of California, Los Angeles, and chair of the department of emergency medicine at Harbor-UCLA Medical Center. Regarding just the problem of completing the pilot study, Dr. Lewis commented, “It sounds like it’s going to be impossible to get the data that directly answers the questions.”
Peter Marx, MD, PhD, who directs the FDA’s Center for Biologics Evaluation and Research (CBER), which oversees blood products safety, joined the discussion to acknowledge the difficulty, but underscore the social importance of a careful examination of the current MSM donation policy.
“We understand the issues here…. With all due respect to our European colleagues, there’s not enough data. That’s the point of this study; we also know that the U.S. has a very different epidemiology of HIV than the U.K. and a lot of other places,” Dr. Marx said. “The pilot study is a way to get some data where we might be able to get away from a time-based deferral. The LGBT community finds any time-based deferral discriminatory.”
Pathogen reduction technology
The committee heard a proposal for a completely different strategy during its afternoon session: pathogen reduction technology (PRT) holds promise to achieve virtual elimination of HIV and other pathogens from donated blood products.
The FDA is reviewing a variance request from the nonprofit blood donation organization Bloodworks Northwest organization to use PRT for apheresis platelet donations from MSM who would otherwise be deferred because of sexual activity within the 12-month deferral window.
James AuBuchon, MD, president of Bloodworks Northwest, explained that his organization takes in about 225,000 donations annually. The variance sought would use the FDA-approved INTERCEPT device to achieve pathogen reduction for donations that meet all requirements except the MSM deferral, and that would still undergo all relevant transfusion transmitted infection testing.
The INTERCEPT device uses amotosalen, which intercalates with DNA and RNA, inactivating it after exposure to ultraviolet A light. Amotosalen is then removed from the blood product before administration. The pathogen reduction activity doesn’t interfere with platelets or plasma, and is active against a wide range of viruses, bacteria, and fungal pathogens, explained Dr. AuBuchon, who is also a professor of hematology at the University of Washington, Seattle.
Dr. AuBuchon walked the committee through procedures designed to flag donors for PRT platelet apheresis, and to ensure these donations receive the intended PRT treatment. Platelets were chosen for this variance request, he explained, because demand outstrips supply. “We are all spending additional time and resources in recruiting a new framework and demographic, and it is exceedingly difficult to keep enough donors coming through the door,” he said. “Our platelet utilization climbs continually – it’s up 15% in the last 4 years.”
Committee members circled around the idea that all risk can’t be eliminated, even with the highly effective PRT technology. But the risk is exceedingly low, said committee chair Richard Kaufman, MD, medical director of the adult transfusion service at Brigham and Women’s Hospital, Boston. “It’s not possible to get rid of the window. We can kind of hammer down the risk by shrinking down the window by using incredibly sensitive tests. But that risk continues to exist. Pathogen reduction can take care of that residual risk…. So what’s left is really quite a low risk,” Dr. Kaufman said.
Susan Stramer, PhD, vice president of scientific affairs for the American Red Cross, concurred, noting that pathogen reduction techniques are already in use for many other blood products, particularly within the plasma industry.
Wrapping up, Dr. Kaufman asked individual committee members to summarize their position on the variance request, though the FDA had not placed a voting question before the committee. Consensus in the room was that this real-world examination of PRT could point to a path to expanding the donor pool while maintaining patient safety – a concern all agreed was paramount.
The FDA usually follows the recommendations of its committees.
The
At a meeting of the FDA’s Blood Products Advisory Committee, the agency shared the content of the 5-item questionnaire and reviewed the proposed study design with committee members, who were asked to comment – but not vote – on the best path forward for MSM donation policies.
The FDA is “committed to ongoing evaluation of the MSM deferral policy” and remains open to adjusting the policy based on the best available scientific evidence, said Barbee Whitaker, PhD, a lead scientist in the agency’s Office of Emerging and Transfusion Transmitted Disease
After recruiting 2,000 men who have had sex with men at least once during the past 3 months, the study will aim to identify individuals who have very recently become HIV infected, in order to assess the discriminant function of the set of behavioral questions that are proposed in the questionnaire.
The crux of the problem currently, noted Dr. Whitaker, is identifying those individuals who are very recently infected with HIV. Nucleic acid testing has tightened the window of undetectability considerably, but the current 12-month deferral window after men have had sexual contact with other men is designed to ensure safety of the blood supply.
Social justice concerns have been raised about the blanket deferral, said Dr. Whitaker; the behavioral questions in the pilot study will ask about the number of different sexual partners men have had within the past 1, 3, and 12 months and ask about the type of sexual contact (oral sex, or anal penetrative or receptive intercourse). The questionnaire also asks about sex with a partner known to be HIV positive, condom use, and use of pre-exposure prophylaxis (PrEP).
The FDA will ask for proposals to conduct the study with an eye to having sites in such cities as Washington, Atlanta, and Miami, which have high incidences of HIV, to improve chances of early detection.
The behavioral questionnaire is not seen as an immediate replacement for the 12-month deferral policy, the FDA made clear in its briefing documents and in discussion with the committee. Instead, its utility will be in the information gleaned from the pilot study and a follow-on that may include several hundred thousand individuals. These data should provide “population-based evidence upon which to base regulatory decisions to ensure blood safety,” she said.
Donation policies outside the United States
Whether a change in blood donation deferral policies for MSM would be a shortened window or a move toward a behavioral questionnaire is currently not known. Globally, a variety of practices are used for blood screening, said Mindy Goldman, MD, medical director of Canadian Blood Services, who reviewed international perspectives on blood donation for MSM.
“There’s no general consensus on donation deferrals internationally,” she said. Factors influencing policy can include epidemiology, risk analysis, modeling, and history of response to threats in the past.
However, “there’s basically a couple of main approaches” to handling deferrals for MSM, Dr. Goldman said. One is time-based deferral – the strategy used in the United States, as well as Canada, the United Kingdom, Japan, and Australia.
Japan and the U.K. have recently moved to 3-month deferral periods, a figure arrived at by doubling the window period for nucleic acid testing for HIV, roughly, Dr. Goldman said. Early data from the U.K. experience has not shown an increase in HIV rates among donors, or an increase in NAT-only positive donors, she said. An application to move from a 12-month to a 3-month deferral period is pending in Canada.
A strong advantage of time-based deferral as a risk management strategy, Dr. Goldman said, is standardization. “For us, standardization is close to godliness.”
However, she added, “another major limitation is that you’re still deferring all sexually active MSM, including those who are in a stable monogamous relationship from donating. From a justice perspective for the lowest risk population of MSM – they are still being deferred using this type of approach.”
Some nations, such as Spain and Italy, use individual risk assessment via physician-led interviews. These approaches are often not standardized. “There’s no national uniform questionnaire, so there’s less standardization, and more variability between blood centers,” Dr. Goldman said. “So you wind up trying to compare apples with oranges.”
This means the results are harder to evaluate on a national level. However, there appears to be higher residual risk, with HIV rates among first-time donors approaching those of the general population, Dr. Goldman said.
Another strategy, used in France, is a test-retest model, where blood from first-time MSM that initially tests negative for HIV is held until the individual returns for re-testing or an additional donation, with a second negative test. This approach increases operational complexity and cost, noted Dr. Goldman, and because of the short shelf life of platelets, it’s not practical for this blood component.
In general questioning and discussion after this and other background presentations, the committee could agree on one point: this isn’t an easy question.
“I’m increasingly struck by how difficult this problem is,” said committee member Roger Lewis, MD, PhD, professor at the University of California, Los Angeles, and chair of the department of emergency medicine at Harbor-UCLA Medical Center. Regarding just the problem of completing the pilot study, Dr. Lewis commented, “It sounds like it’s going to be impossible to get the data that directly answers the questions.”
Peter Marx, MD, PhD, who directs the FDA’s Center for Biologics Evaluation and Research (CBER), which oversees blood products safety, joined the discussion to acknowledge the difficulty, but underscore the social importance of a careful examination of the current MSM donation policy.
“We understand the issues here…. With all due respect to our European colleagues, there’s not enough data. That’s the point of this study; we also know that the U.S. has a very different epidemiology of HIV than the U.K. and a lot of other places,” Dr. Marx said. “The pilot study is a way to get some data where we might be able to get away from a time-based deferral. The LGBT community finds any time-based deferral discriminatory.”
Pathogen reduction technology
The committee heard a proposal for a completely different strategy during its afternoon session: pathogen reduction technology (PRT) holds promise to achieve virtual elimination of HIV and other pathogens from donated blood products.
The FDA is reviewing a variance request from the nonprofit blood donation organization Bloodworks Northwest organization to use PRT for apheresis platelet donations from MSM who would otherwise be deferred because of sexual activity within the 12-month deferral window.
James AuBuchon, MD, president of Bloodworks Northwest, explained that his organization takes in about 225,000 donations annually. The variance sought would use the FDA-approved INTERCEPT device to achieve pathogen reduction for donations that meet all requirements except the MSM deferral, and that would still undergo all relevant transfusion transmitted infection testing.
The INTERCEPT device uses amotosalen, which intercalates with DNA and RNA, inactivating it after exposure to ultraviolet A light. Amotosalen is then removed from the blood product before administration. The pathogen reduction activity doesn’t interfere with platelets or plasma, and is active against a wide range of viruses, bacteria, and fungal pathogens, explained Dr. AuBuchon, who is also a professor of hematology at the University of Washington, Seattle.
Dr. AuBuchon walked the committee through procedures designed to flag donors for PRT platelet apheresis, and to ensure these donations receive the intended PRT treatment. Platelets were chosen for this variance request, he explained, because demand outstrips supply. “We are all spending additional time and resources in recruiting a new framework and demographic, and it is exceedingly difficult to keep enough donors coming through the door,” he said. “Our platelet utilization climbs continually – it’s up 15% in the last 4 years.”
Committee members circled around the idea that all risk can’t be eliminated, even with the highly effective PRT technology. But the risk is exceedingly low, said committee chair Richard Kaufman, MD, medical director of the adult transfusion service at Brigham and Women’s Hospital, Boston. “It’s not possible to get rid of the window. We can kind of hammer down the risk by shrinking down the window by using incredibly sensitive tests. But that risk continues to exist. Pathogen reduction can take care of that residual risk…. So what’s left is really quite a low risk,” Dr. Kaufman said.
Susan Stramer, PhD, vice president of scientific affairs for the American Red Cross, concurred, noting that pathogen reduction techniques are already in use for many other blood products, particularly within the plasma industry.
Wrapping up, Dr. Kaufman asked individual committee members to summarize their position on the variance request, though the FDA had not placed a voting question before the committee. Consensus in the room was that this real-world examination of PRT could point to a path to expanding the donor pool while maintaining patient safety – a concern all agreed was paramount.
The FDA usually follows the recommendations of its committees.
The
At a meeting of the FDA’s Blood Products Advisory Committee, the agency shared the content of the 5-item questionnaire and reviewed the proposed study design with committee members, who were asked to comment – but not vote – on the best path forward for MSM donation policies.
The FDA is “committed to ongoing evaluation of the MSM deferral policy” and remains open to adjusting the policy based on the best available scientific evidence, said Barbee Whitaker, PhD, a lead scientist in the agency’s Office of Emerging and Transfusion Transmitted Disease
After recruiting 2,000 men who have had sex with men at least once during the past 3 months, the study will aim to identify individuals who have very recently become HIV infected, in order to assess the discriminant function of the set of behavioral questions that are proposed in the questionnaire.
The crux of the problem currently, noted Dr. Whitaker, is identifying those individuals who are very recently infected with HIV. Nucleic acid testing has tightened the window of undetectability considerably, but the current 12-month deferral window after men have had sexual contact with other men is designed to ensure safety of the blood supply.
Social justice concerns have been raised about the blanket deferral, said Dr. Whitaker; the behavioral questions in the pilot study will ask about the number of different sexual partners men have had within the past 1, 3, and 12 months and ask about the type of sexual contact (oral sex, or anal penetrative or receptive intercourse). The questionnaire also asks about sex with a partner known to be HIV positive, condom use, and use of pre-exposure prophylaxis (PrEP).
The FDA will ask for proposals to conduct the study with an eye to having sites in such cities as Washington, Atlanta, and Miami, which have high incidences of HIV, to improve chances of early detection.
The behavioral questionnaire is not seen as an immediate replacement for the 12-month deferral policy, the FDA made clear in its briefing documents and in discussion with the committee. Instead, its utility will be in the information gleaned from the pilot study and a follow-on that may include several hundred thousand individuals. These data should provide “population-based evidence upon which to base regulatory decisions to ensure blood safety,” she said.
Donation policies outside the United States
Whether a change in blood donation deferral policies for MSM would be a shortened window or a move toward a behavioral questionnaire is currently not known. Globally, a variety of practices are used for blood screening, said Mindy Goldman, MD, medical director of Canadian Blood Services, who reviewed international perspectives on blood donation for MSM.
“There’s no general consensus on donation deferrals internationally,” she said. Factors influencing policy can include epidemiology, risk analysis, modeling, and history of response to threats in the past.
However, “there’s basically a couple of main approaches” to handling deferrals for MSM, Dr. Goldman said. One is time-based deferral – the strategy used in the United States, as well as Canada, the United Kingdom, Japan, and Australia.
Japan and the U.K. have recently moved to 3-month deferral periods, a figure arrived at by doubling the window period for nucleic acid testing for HIV, roughly, Dr. Goldman said. Early data from the U.K. experience has not shown an increase in HIV rates among donors, or an increase in NAT-only positive donors, she said. An application to move from a 12-month to a 3-month deferral period is pending in Canada.
A strong advantage of time-based deferral as a risk management strategy, Dr. Goldman said, is standardization. “For us, standardization is close to godliness.”
However, she added, “another major limitation is that you’re still deferring all sexually active MSM, including those who are in a stable monogamous relationship from donating. From a justice perspective for the lowest risk population of MSM – they are still being deferred using this type of approach.”
Some nations, such as Spain and Italy, use individual risk assessment via physician-led interviews. These approaches are often not standardized. “There’s no national uniform questionnaire, so there’s less standardization, and more variability between blood centers,” Dr. Goldman said. “So you wind up trying to compare apples with oranges.”
This means the results are harder to evaluate on a national level. However, there appears to be higher residual risk, with HIV rates among first-time donors approaching those of the general population, Dr. Goldman said.
Another strategy, used in France, is a test-retest model, where blood from first-time MSM that initially tests negative for HIV is held until the individual returns for re-testing or an additional donation, with a second negative test. This approach increases operational complexity and cost, noted Dr. Goldman, and because of the short shelf life of platelets, it’s not practical for this blood component.
In general questioning and discussion after this and other background presentations, the committee could agree on one point: this isn’t an easy question.
“I’m increasingly struck by how difficult this problem is,” said committee member Roger Lewis, MD, PhD, professor at the University of California, Los Angeles, and chair of the department of emergency medicine at Harbor-UCLA Medical Center. Regarding just the problem of completing the pilot study, Dr. Lewis commented, “It sounds like it’s going to be impossible to get the data that directly answers the questions.”
Peter Marx, MD, PhD, who directs the FDA’s Center for Biologics Evaluation and Research (CBER), which oversees blood products safety, joined the discussion to acknowledge the difficulty, but underscore the social importance of a careful examination of the current MSM donation policy.
“We understand the issues here…. With all due respect to our European colleagues, there’s not enough data. That’s the point of this study; we also know that the U.S. has a very different epidemiology of HIV than the U.K. and a lot of other places,” Dr. Marx said. “The pilot study is a way to get some data where we might be able to get away from a time-based deferral. The LGBT community finds any time-based deferral discriminatory.”
Pathogen reduction technology
The committee heard a proposal for a completely different strategy during its afternoon session: pathogen reduction technology (PRT) holds promise to achieve virtual elimination of HIV and other pathogens from donated blood products.
The FDA is reviewing a variance request from the nonprofit blood donation organization Bloodworks Northwest organization to use PRT for apheresis platelet donations from MSM who would otherwise be deferred because of sexual activity within the 12-month deferral window.
James AuBuchon, MD, president of Bloodworks Northwest, explained that his organization takes in about 225,000 donations annually. The variance sought would use the FDA-approved INTERCEPT device to achieve pathogen reduction for donations that meet all requirements except the MSM deferral, and that would still undergo all relevant transfusion transmitted infection testing.
The INTERCEPT device uses amotosalen, which intercalates with DNA and RNA, inactivating it after exposure to ultraviolet A light. Amotosalen is then removed from the blood product before administration. The pathogen reduction activity doesn’t interfere with platelets or plasma, and is active against a wide range of viruses, bacteria, and fungal pathogens, explained Dr. AuBuchon, who is also a professor of hematology at the University of Washington, Seattle.
Dr. AuBuchon walked the committee through procedures designed to flag donors for PRT platelet apheresis, and to ensure these donations receive the intended PRT treatment. Platelets were chosen for this variance request, he explained, because demand outstrips supply. “We are all spending additional time and resources in recruiting a new framework and demographic, and it is exceedingly difficult to keep enough donors coming through the door,” he said. “Our platelet utilization climbs continually – it’s up 15% in the last 4 years.”
Committee members circled around the idea that all risk can’t be eliminated, even with the highly effective PRT technology. But the risk is exceedingly low, said committee chair Richard Kaufman, MD, medical director of the adult transfusion service at Brigham and Women’s Hospital, Boston. “It’s not possible to get rid of the window. We can kind of hammer down the risk by shrinking down the window by using incredibly sensitive tests. But that risk continues to exist. Pathogen reduction can take care of that residual risk…. So what’s left is really quite a low risk,” Dr. Kaufman said.
Susan Stramer, PhD, vice president of scientific affairs for the American Red Cross, concurred, noting that pathogen reduction techniques are already in use for many other blood products, particularly within the plasma industry.
Wrapping up, Dr. Kaufman asked individual committee members to summarize their position on the variance request, though the FDA had not placed a voting question before the committee. Consensus in the room was that this real-world examination of PRT could point to a path to expanding the donor pool while maintaining patient safety – a concern all agreed was paramount.
The FDA usually follows the recommendations of its committees.
FROM AN FDA ADVISORY COMMITTEE MEETING
CDC exhorts more testing and treatment of HIV
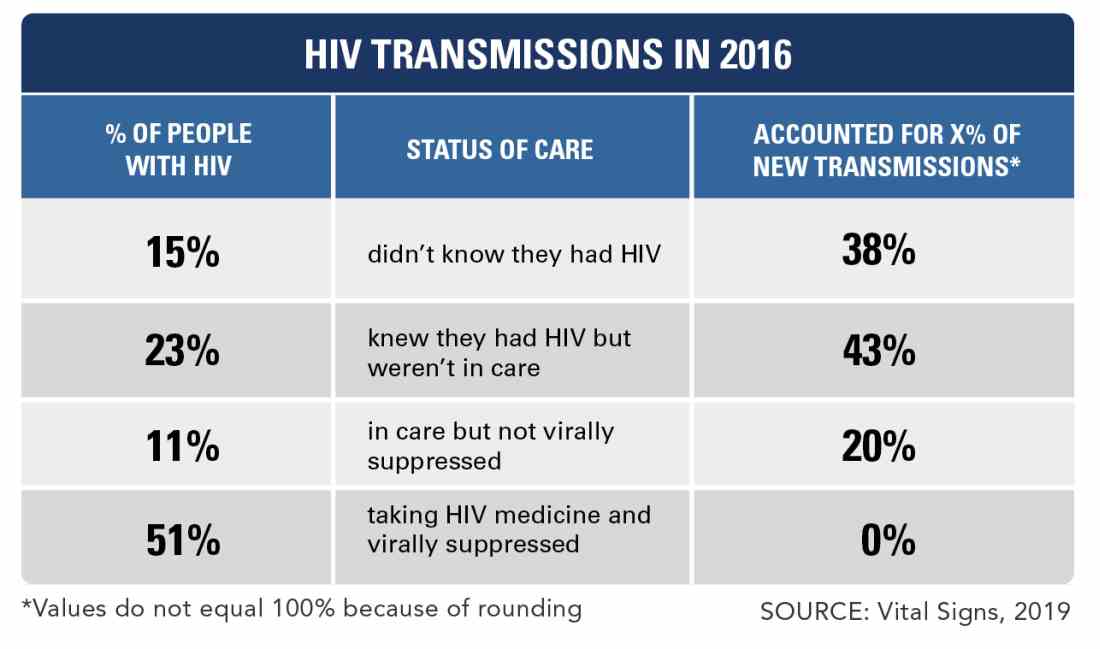
Approximately 80% of HIV infections in the United States in 2016 were spread by almost 40% of infected individuals who did not know their status or were not receiving care, according to data from the Centers for Disease Control and Prevention.
However, leadership at the Department of Health & Human Services has developed a “bold but completely achievable” plan to reduce HIV infections within the next decade, Vice Adm. Jerome M. Adams, MD, the U.S. surgeon general, said in a teleconference to announce the results of a new Vital Signs report on the impact of undiagnosed and untreated HIV.
In the early release Vital Signs from Morbidity and Mortality Weekly Report, Li Zihao, PhD, and his colleagues at the CDC used a model to estimate rates of HIV transmission in 2016 based on data from the National HIV Behavioral Surveillance on needle-sharing behavior and sexual behaviors. The overall transmission rate was 3.5/100,000 person-years. Of these transmissions, 73.0% were from men who have sex with men, 9.7% from intravenous drug users, and 12% from heterosexuals.
The percentages of transmissions for those who were acutely ill with HIV but unaware, not acutely ill but unaware, aware of HIV infection but not treated, receiving care but not virally suppressed, and receiving care and virally suppressed were 4.0%, 33.6%, 42.6%, 19.8%, and 0%, respectively, the researchers said.
The study “emphasizes the impact that HIV resources could have,” by showing the importance of identifying infected individuals early and using the tools now available to treat them before they can transmit the disease, Jonathan Mermin, MD, director of the CDC’s National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention, said during the conference.
“Today’s treatment regimens are simpler than those prescribed in the past, sometimes requiring only single-tablet formulations, with fewer side effects; most persons with HIV infection can achieve viral suppression within 6 months of initiating treatment,” the researchers wrote.
In the wake of the findings and at the start of the CDC’s 2019 National HIV Prevention Conference, the CDC proposed a federal initiative, “Ending the HIV Epidemic: A Plan for America.”
The goal is to reduce the incidence of new HIV infections by at least 90% over the next decade, starting with a focus on parts of the country with the highest disease burden, according to the CDC.
“Today’s Vital Signs report illustrates how a goal that once seemed impossible is now within our reach.” Robert R. Redfield, MD, director of the Centers for Disease Control and Prevention, said during the conference. “If we increase access to testing and treatment for people with HIV, we could prevent a lion’s share of infections,” he said.
The plan involves working to identify individuals at risk, treating those who test positive as quickly as possible, and keeping them in care. Updated information on the CDC website provides more details for clinicians on how to have conversations about HIV with patients, the latest information about antiretroviral therapy, and details about prevention for partners including post- and pre-exposure prophylaxis (PEP and PreP), and condoms.
Eugene McCray, MD, director of the CDC’s Division of HIV/AIDS Prevention, emphasized that the CDC recommends HIV testing for all individuals aged 13-64 years at least once in their lives. He added that everyone who tests positive should seek medical care, that everyone with HIV deserves support to combat stigma, and that those at risk should be empowered to take advantage of proven effective prevention strategies.
Clinicians can access the updated CDC page on caring for HIV patients here.
SOURCE: Li Z et al. MMWR Morb Mortal Wkly Rep. 2019 March 18. doi: org/10.15585/mmwr.mm6811e1.
Approximately 80% of HIV infections in the United States in 2016 were spread by almost 40% of infected individuals who did not know their status or were not receiving care, according to data from the Centers for Disease Control and Prevention.

However, leadership at the Department of Health & Human Services has developed a “bold but completely achievable” plan to reduce HIV infections within the next decade, Vice Adm. Jerome M. Adams, MD, the U.S. surgeon general, said in a teleconference to announce the results of a new Vital Signs report on the impact of undiagnosed and untreated HIV.
In the early release Vital Signs from Morbidity and Mortality Weekly Report, Li Zihao, PhD, and his colleagues at the CDC used a model to estimate rates of HIV transmission in 2016 based on data from the National HIV Behavioral Surveillance on needle-sharing behavior and sexual behaviors. The overall transmission rate was 3.5/100,000 person-years. Of these transmissions, 73.0% were from men who have sex with men, 9.7% from intravenous drug users, and 12% from heterosexuals.
The percentages of transmissions for those who were acutely ill with HIV but unaware, not acutely ill but unaware, aware of HIV infection but not treated, receiving care but not virally suppressed, and receiving care and virally suppressed were 4.0%, 33.6%, 42.6%, 19.8%, and 0%, respectively, the researchers said.
The study “emphasizes the impact that HIV resources could have,” by showing the importance of identifying infected individuals early and using the tools now available to treat them before they can transmit the disease, Jonathan Mermin, MD, director of the CDC’s National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention, said during the conference.
“Today’s treatment regimens are simpler than those prescribed in the past, sometimes requiring only single-tablet formulations, with fewer side effects; most persons with HIV infection can achieve viral suppression within 6 months of initiating treatment,” the researchers wrote.
In the wake of the findings and at the start of the CDC’s 2019 National HIV Prevention Conference, the CDC proposed a federal initiative, “Ending the HIV Epidemic: A Plan for America.”
The goal is to reduce the incidence of new HIV infections by at least 90% over the next decade, starting with a focus on parts of the country with the highest disease burden, according to the CDC.
“Today’s Vital Signs report illustrates how a goal that once seemed impossible is now within our reach.” Robert R. Redfield, MD, director of the Centers for Disease Control and Prevention, said during the conference. “If we increase access to testing and treatment for people with HIV, we could prevent a lion’s share of infections,” he said.
The plan involves working to identify individuals at risk, treating those who test positive as quickly as possible, and keeping them in care. Updated information on the CDC website provides more details for clinicians on how to have conversations about HIV with patients, the latest information about antiretroviral therapy, and details about prevention for partners including post- and pre-exposure prophylaxis (PEP and PreP), and condoms.
Eugene McCray, MD, director of the CDC’s Division of HIV/AIDS Prevention, emphasized that the CDC recommends HIV testing for all individuals aged 13-64 years at least once in their lives. He added that everyone who tests positive should seek medical care, that everyone with HIV deserves support to combat stigma, and that those at risk should be empowered to take advantage of proven effective prevention strategies.
Clinicians can access the updated CDC page on caring for HIV patients here.
SOURCE: Li Z et al. MMWR Morb Mortal Wkly Rep. 2019 March 18. doi: org/10.15585/mmwr.mm6811e1.
Approximately 80% of HIV infections in the United States in 2016 were spread by almost 40% of infected individuals who did not know their status or were not receiving care, according to data from the Centers for Disease Control and Prevention.

However, leadership at the Department of Health & Human Services has developed a “bold but completely achievable” plan to reduce HIV infections within the next decade, Vice Adm. Jerome M. Adams, MD, the U.S. surgeon general, said in a teleconference to announce the results of a new Vital Signs report on the impact of undiagnosed and untreated HIV.
In the early release Vital Signs from Morbidity and Mortality Weekly Report, Li Zihao, PhD, and his colleagues at the CDC used a model to estimate rates of HIV transmission in 2016 based on data from the National HIV Behavioral Surveillance on needle-sharing behavior and sexual behaviors. The overall transmission rate was 3.5/100,000 person-years. Of these transmissions, 73.0% were from men who have sex with men, 9.7% from intravenous drug users, and 12% from heterosexuals.
The percentages of transmissions for those who were acutely ill with HIV but unaware, not acutely ill but unaware, aware of HIV infection but not treated, receiving care but not virally suppressed, and receiving care and virally suppressed were 4.0%, 33.6%, 42.6%, 19.8%, and 0%, respectively, the researchers said.
The study “emphasizes the impact that HIV resources could have,” by showing the importance of identifying infected individuals early and using the tools now available to treat them before they can transmit the disease, Jonathan Mermin, MD, director of the CDC’s National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention, said during the conference.
“Today’s treatment regimens are simpler than those prescribed in the past, sometimes requiring only single-tablet formulations, with fewer side effects; most persons with HIV infection can achieve viral suppression within 6 months of initiating treatment,” the researchers wrote.
In the wake of the findings and at the start of the CDC’s 2019 National HIV Prevention Conference, the CDC proposed a federal initiative, “Ending the HIV Epidemic: A Plan for America.”
The goal is to reduce the incidence of new HIV infections by at least 90% over the next decade, starting with a focus on parts of the country with the highest disease burden, according to the CDC.
“Today’s Vital Signs report illustrates how a goal that once seemed impossible is now within our reach.” Robert R. Redfield, MD, director of the Centers for Disease Control and Prevention, said during the conference. “If we increase access to testing and treatment for people with HIV, we could prevent a lion’s share of infections,” he said.
The plan involves working to identify individuals at risk, treating those who test positive as quickly as possible, and keeping them in care. Updated information on the CDC website provides more details for clinicians on how to have conversations about HIV with patients, the latest information about antiretroviral therapy, and details about prevention for partners including post- and pre-exposure prophylaxis (PEP and PreP), and condoms.
Eugene McCray, MD, director of the CDC’s Division of HIV/AIDS Prevention, emphasized that the CDC recommends HIV testing for all individuals aged 13-64 years at least once in their lives. He added that everyone who tests positive should seek medical care, that everyone with HIV deserves support to combat stigma, and that those at risk should be empowered to take advantage of proven effective prevention strategies.
Clinicians can access the updated CDC page on caring for HIV patients here.
SOURCE: Li Z et al. MMWR Morb Mortal Wkly Rep. 2019 March 18. doi: org/10.15585/mmwr.mm6811e1.
FROM THE CDC
New study a reprieve for isoniazid during HIV pregnancy?
SEATTLE – At the 2018 Conference on Retroviruses and Opportunistic Infections, investigators reported that the rate of adverse pregnancy outcomes was 23% among HIV-positive women who took isoniazid to prevent tuberculosis during pregnancy, but just 17% among women who waited until after delivery.
It was an international, randomized trial that included 156 women, and outcomes included in utero demise and low birth weight. The investigators concluded that the World Health Organization needed to reconsider its recommendation for isoniazid preventative treatment (IPT) during HIV pregnancy in areas where tuberculosis (TB) is common.
The news was in the exact opposite direction at this year’s conference: An observational study of 151 pregnant HIV-positive women in Soweto, South Africa, found no evidence of poor maternal or infant outcomes with isoniazid prophylaxis, according to investigators led by Nicole Salazar-Austin, MD, a pediatric instructor at Johns Hopkins University, Baltimore.
The team was alarmed by the 2018 findings, so it decided to take a closer look with their own data. They compared outcomes in 69 HIV-positive women (46%) who reported initiating IPT during pregnancy with 82 who did not from January 2011 through January 2014. There was higher use of combination antiretroviral therapy in the IPT group, and viral loads below 400 copies/mL were more common.
The proportion of neonates born prematurely was lower in the IPT group than in the group not using it, but this did not reach significance (10% vs. 22%; P = 0.06). There was no difference in fetal demise (1% in both groups); low birth weight (9% vs. 12%; P = 0.51); or congenital anomalies (1% vs. 2%, P = 1.0).
A composite of the four outcomes actually showed fewer events among infants exposed to IPT (16% vs. 28%; P = 0.08). The outcomes held when stratified by viral load suppression and when other causes of poor infant outcomes – advanced HIV disease, advanced maternal age, and low weight gain during pregnancy – were taken into account.
As an observational study based on self-report, the new work was much less rigorous than the 2018 randomized trial, Dr. Salazar-Austin noted. Also, the work was part of a larger project that was not designed specifically to study pregnancy outcomes. IPT was initiated by public antenatal and HIV clinics, not by the study team.
Still, “we hope that these results provide some reassurance that IPT can be used in the second or third trimester among pregnant women with HIV,” Dr. Salazar-Austin said.
So, how to mesh the two studies? Like everything in medicine, it comes down to a risk/benefit analysis.
“For pregnant women living in a household with an active TB case, maybe the benefit outweighs the risk, especially if they are living with HIV. Whether all women living with HIV in a high [TB] burden [area] should receive isoniazid is still somewhat in question. I think we need to weigh” IPT during HIV pregnancy on an “individual basis moving forward,” she said.
Women in the observational trial were a median of about 30 years old. The median CD4 T-cell count at enrollment was about 370 cells/mm3.
The work was funded by the National Institutes of Health. Dr. Salazar-Austin didn’t have any disclosures.
SOURCE: Salazar-Austin NM et al. CROI 2019, Abstract 77.
SEATTLE – At the 2018 Conference on Retroviruses and Opportunistic Infections, investigators reported that the rate of adverse pregnancy outcomes was 23% among HIV-positive women who took isoniazid to prevent tuberculosis during pregnancy, but just 17% among women who waited until after delivery.
It was an international, randomized trial that included 156 women, and outcomes included in utero demise and low birth weight. The investigators concluded that the World Health Organization needed to reconsider its recommendation for isoniazid preventative treatment (IPT) during HIV pregnancy in areas where tuberculosis (TB) is common.
The news was in the exact opposite direction at this year’s conference: An observational study of 151 pregnant HIV-positive women in Soweto, South Africa, found no evidence of poor maternal or infant outcomes with isoniazid prophylaxis, according to investigators led by Nicole Salazar-Austin, MD, a pediatric instructor at Johns Hopkins University, Baltimore.
The team was alarmed by the 2018 findings, so it decided to take a closer look with their own data. They compared outcomes in 69 HIV-positive women (46%) who reported initiating IPT during pregnancy with 82 who did not from January 2011 through January 2014. There was higher use of combination antiretroviral therapy in the IPT group, and viral loads below 400 copies/mL were more common.
The proportion of neonates born prematurely was lower in the IPT group than in the group not using it, but this did not reach significance (10% vs. 22%; P = 0.06). There was no difference in fetal demise (1% in both groups); low birth weight (9% vs. 12%; P = 0.51); or congenital anomalies (1% vs. 2%, P = 1.0).
A composite of the four outcomes actually showed fewer events among infants exposed to IPT (16% vs. 28%; P = 0.08). The outcomes held when stratified by viral load suppression and when other causes of poor infant outcomes – advanced HIV disease, advanced maternal age, and low weight gain during pregnancy – were taken into account.
As an observational study based on self-report, the new work was much less rigorous than the 2018 randomized trial, Dr. Salazar-Austin noted. Also, the work was part of a larger project that was not designed specifically to study pregnancy outcomes. IPT was initiated by public antenatal and HIV clinics, not by the study team.
Still, “we hope that these results provide some reassurance that IPT can be used in the second or third trimester among pregnant women with HIV,” Dr. Salazar-Austin said.
So, how to mesh the two studies? Like everything in medicine, it comes down to a risk/benefit analysis.
“For pregnant women living in a household with an active TB case, maybe the benefit outweighs the risk, especially if they are living with HIV. Whether all women living with HIV in a high [TB] burden [area] should receive isoniazid is still somewhat in question. I think we need to weigh” IPT during HIV pregnancy on an “individual basis moving forward,” she said.
Women in the observational trial were a median of about 30 years old. The median CD4 T-cell count at enrollment was about 370 cells/mm3.
The work was funded by the National Institutes of Health. Dr. Salazar-Austin didn’t have any disclosures.
SOURCE: Salazar-Austin NM et al. CROI 2019, Abstract 77.
SEATTLE – At the 2018 Conference on Retroviruses and Opportunistic Infections, investigators reported that the rate of adverse pregnancy outcomes was 23% among HIV-positive women who took isoniazid to prevent tuberculosis during pregnancy, but just 17% among women who waited until after delivery.
It was an international, randomized trial that included 156 women, and outcomes included in utero demise and low birth weight. The investigators concluded that the World Health Organization needed to reconsider its recommendation for isoniazid preventative treatment (IPT) during HIV pregnancy in areas where tuberculosis (TB) is common.
The news was in the exact opposite direction at this year’s conference: An observational study of 151 pregnant HIV-positive women in Soweto, South Africa, found no evidence of poor maternal or infant outcomes with isoniazid prophylaxis, according to investigators led by Nicole Salazar-Austin, MD, a pediatric instructor at Johns Hopkins University, Baltimore.
The team was alarmed by the 2018 findings, so it decided to take a closer look with their own data. They compared outcomes in 69 HIV-positive women (46%) who reported initiating IPT during pregnancy with 82 who did not from January 2011 through January 2014. There was higher use of combination antiretroviral therapy in the IPT group, and viral loads below 400 copies/mL were more common.
The proportion of neonates born prematurely was lower in the IPT group than in the group not using it, but this did not reach significance (10% vs. 22%; P = 0.06). There was no difference in fetal demise (1% in both groups); low birth weight (9% vs. 12%; P = 0.51); or congenital anomalies (1% vs. 2%, P = 1.0).
A composite of the four outcomes actually showed fewer events among infants exposed to IPT (16% vs. 28%; P = 0.08). The outcomes held when stratified by viral load suppression and when other causes of poor infant outcomes – advanced HIV disease, advanced maternal age, and low weight gain during pregnancy – were taken into account.
As an observational study based on self-report, the new work was much less rigorous than the 2018 randomized trial, Dr. Salazar-Austin noted. Also, the work was part of a larger project that was not designed specifically to study pregnancy outcomes. IPT was initiated by public antenatal and HIV clinics, not by the study team.
Still, “we hope that these results provide some reassurance that IPT can be used in the second or third trimester among pregnant women with HIV,” Dr. Salazar-Austin said.
So, how to mesh the two studies? Like everything in medicine, it comes down to a risk/benefit analysis.
“For pregnant women living in a household with an active TB case, maybe the benefit outweighs the risk, especially if they are living with HIV. Whether all women living with HIV in a high [TB] burden [area] should receive isoniazid is still somewhat in question. I think we need to weigh” IPT during HIV pregnancy on an “individual basis moving forward,” she said.
Women in the observational trial were a median of about 30 years old. The median CD4 T-cell count at enrollment was about 370 cells/mm3.
The work was funded by the National Institutes of Health. Dr. Salazar-Austin didn’t have any disclosures.
SOURCE: Salazar-Austin NM et al. CROI 2019, Abstract 77.
REPORTING FROM CROI 2019
Repeat VTE risk heightened in HIV patients
SEATTLE – HIV infection is associated with increased risk of recurrent venous thromboembolism, especially within 1 year of the initial episode. The finding, presented during a poster session at the Conference on Retroviruses & Opportunistic Infections, follows up on an earlier study that found that first-time VTE risk also is higher among HIV-positive individuals than in the general population.
The conclusion about first-time VTE risk, published earlier this year in Lancet HIV, came from a comparison between the ATHENA (AIDS Therapy Evaluation in the Netherlands) cohort and European population-level of studies of VTE. It found a crude incidence of 2.33 VTE events per 1,000 person-years In HIV patients, with heightened odds when CD4 cell counts were below 200 cells/mcL (adjusted hazard ratio, 3.40).
The new work represents a follow-up and compared results from ATHENA (153 patients with HIV and first VTE) and the Dutch MEGA cohort (4,005 patients without HIV, with first VTE), which includes the general population. Overall, 26% of patients in the ATHENA cohort experienced a second VTE event, compared with 16% of the general population. At 1 year after anticoagulation withdrawal, HIV-positive individuals were at 67% increased risk (HR, 1.67). At 6-years after withdrawal, the relationship was not statistically significant (HR, 1.22).
Researchers also found that CD4 cell-count recovery was associated with lowered risk, with every 100 cell-count increase between initial VTE diagnosis and anticoagulant withdrawal linked to a 20% reduction in risk (HR, 0.80).
“The clinical question is: If it’s true you have an increased risk of recurrence, should you be continuing anticoagulant therapy longer in people with HIV? This poster doesn’t answer that question and you probably need a randomized, controlled trial to look at that,” Peter Reiss, MD, professor of medicine at Amsterdam University Medical Center, said in an interview during the conference.
In the absence of a clear answer, it’s sensible for clinicians to be aware of the potential increased risk, much as clinicians have internalized the increased risk of atherosclerotic vascular disease in HIV patients. “I think the publication [in Lancet HIV] as well as this poster suggest that on the venous side of things there may also be an accentuated risk,” said Dr. Reiss.
Heidi Crane, MD, a professor of medicine at the University of Washington, Seattle, presented a poster examining the underlying factors that may predispose HIV patients to first-time VTE events. Her team performed an adjudicated review of VTE cases among HIV patients at six institutions and found that the risk factors appeared to be distinct from those seen in the general population.
The traditional long plane ride was less common in this population, while factors such as injected drug use and pneumonia were more common. The VTE events occurred at a median age of 49 years; 30% of the patients had a detectable viral load. “We’re seeing a little more (VTE) than you might expect, and in a younger population than you might have guessed,” said Dr. Crane in an interview.
The most frequent predisposing risk factors were recent hospitalization (40%), infection (40%), or immobilization/bed rest (24%) within the past 90 days, and injectable drug use (22%). “It’s not just the traditional risk factors. Some HIV-specific risk factors are driving this,” said Dr. Crane.
She also aims to learn more about the specifics of risk factors, such as catheter-associated thromboses. The team is working to increase the sample size in order to parse out the relationships with specific outcomes.
In the meantime, the data further characterize the health challenges facing people living with HIV. “This is another example demonstrating that comorbid conditions among patients with HIV that are often considered age related occur at much younger ages in our population,” said Dr. Crane.
SOURCE: Rokx C et al. CROI 2019, Abstract 636; and Tenforde MW et al. CROI 2019, Abstract 637.
.
SEATTLE – HIV infection is associated with increased risk of recurrent venous thromboembolism, especially within 1 year of the initial episode. The finding, presented during a poster session at the Conference on Retroviruses & Opportunistic Infections, follows up on an earlier study that found that first-time VTE risk also is higher among HIV-positive individuals than in the general population.
The conclusion about first-time VTE risk, published earlier this year in Lancet HIV, came from a comparison between the ATHENA (AIDS Therapy Evaluation in the Netherlands) cohort and European population-level of studies of VTE. It found a crude incidence of 2.33 VTE events per 1,000 person-years In HIV patients, with heightened odds when CD4 cell counts were below 200 cells/mcL (adjusted hazard ratio, 3.40).
The new work represents a follow-up and compared results from ATHENA (153 patients with HIV and first VTE) and the Dutch MEGA cohort (4,005 patients without HIV, with first VTE), which includes the general population. Overall, 26% of patients in the ATHENA cohort experienced a second VTE event, compared with 16% of the general population. At 1 year after anticoagulation withdrawal, HIV-positive individuals were at 67% increased risk (HR, 1.67). At 6-years after withdrawal, the relationship was not statistically significant (HR, 1.22).
Researchers also found that CD4 cell-count recovery was associated with lowered risk, with every 100 cell-count increase between initial VTE diagnosis and anticoagulant withdrawal linked to a 20% reduction in risk (HR, 0.80).
“The clinical question is: If it’s true you have an increased risk of recurrence, should you be continuing anticoagulant therapy longer in people with HIV? This poster doesn’t answer that question and you probably need a randomized, controlled trial to look at that,” Peter Reiss, MD, professor of medicine at Amsterdam University Medical Center, said in an interview during the conference.
In the absence of a clear answer, it’s sensible for clinicians to be aware of the potential increased risk, much as clinicians have internalized the increased risk of atherosclerotic vascular disease in HIV patients. “I think the publication [in Lancet HIV] as well as this poster suggest that on the venous side of things there may also be an accentuated risk,” said Dr. Reiss.
Heidi Crane, MD, a professor of medicine at the University of Washington, Seattle, presented a poster examining the underlying factors that may predispose HIV patients to first-time VTE events. Her team performed an adjudicated review of VTE cases among HIV patients at six institutions and found that the risk factors appeared to be distinct from those seen in the general population.
The traditional long plane ride was less common in this population, while factors such as injected drug use and pneumonia were more common. The VTE events occurred at a median age of 49 years; 30% of the patients had a detectable viral load. “We’re seeing a little more (VTE) than you might expect, and in a younger population than you might have guessed,” said Dr. Crane in an interview.
The most frequent predisposing risk factors were recent hospitalization (40%), infection (40%), or immobilization/bed rest (24%) within the past 90 days, and injectable drug use (22%). “It’s not just the traditional risk factors. Some HIV-specific risk factors are driving this,” said Dr. Crane.
She also aims to learn more about the specifics of risk factors, such as catheter-associated thromboses. The team is working to increase the sample size in order to parse out the relationships with specific outcomes.
In the meantime, the data further characterize the health challenges facing people living with HIV. “This is another example demonstrating that comorbid conditions among patients with HIV that are often considered age related occur at much younger ages in our population,” said Dr. Crane.
SOURCE: Rokx C et al. CROI 2019, Abstract 636; and Tenforde MW et al. CROI 2019, Abstract 637.
.
SEATTLE – HIV infection is associated with increased risk of recurrent venous thromboembolism, especially within 1 year of the initial episode. The finding, presented during a poster session at the Conference on Retroviruses & Opportunistic Infections, follows up on an earlier study that found that first-time VTE risk also is higher among HIV-positive individuals than in the general population.
The conclusion about first-time VTE risk, published earlier this year in Lancet HIV, came from a comparison between the ATHENA (AIDS Therapy Evaluation in the Netherlands) cohort and European population-level of studies of VTE. It found a crude incidence of 2.33 VTE events per 1,000 person-years In HIV patients, with heightened odds when CD4 cell counts were below 200 cells/mcL (adjusted hazard ratio, 3.40).
The new work represents a follow-up and compared results from ATHENA (153 patients with HIV and first VTE) and the Dutch MEGA cohort (4,005 patients without HIV, with first VTE), which includes the general population. Overall, 26% of patients in the ATHENA cohort experienced a second VTE event, compared with 16% of the general population. At 1 year after anticoagulation withdrawal, HIV-positive individuals were at 67% increased risk (HR, 1.67). At 6-years after withdrawal, the relationship was not statistically significant (HR, 1.22).
Researchers also found that CD4 cell-count recovery was associated with lowered risk, with every 100 cell-count increase between initial VTE diagnosis and anticoagulant withdrawal linked to a 20% reduction in risk (HR, 0.80).
“The clinical question is: If it’s true you have an increased risk of recurrence, should you be continuing anticoagulant therapy longer in people with HIV? This poster doesn’t answer that question and you probably need a randomized, controlled trial to look at that,” Peter Reiss, MD, professor of medicine at Amsterdam University Medical Center, said in an interview during the conference.
In the absence of a clear answer, it’s sensible for clinicians to be aware of the potential increased risk, much as clinicians have internalized the increased risk of atherosclerotic vascular disease in HIV patients. “I think the publication [in Lancet HIV] as well as this poster suggest that on the venous side of things there may also be an accentuated risk,” said Dr. Reiss.
Heidi Crane, MD, a professor of medicine at the University of Washington, Seattle, presented a poster examining the underlying factors that may predispose HIV patients to first-time VTE events. Her team performed an adjudicated review of VTE cases among HIV patients at six institutions and found that the risk factors appeared to be distinct from those seen in the general population.
The traditional long plane ride was less common in this population, while factors such as injected drug use and pneumonia were more common. The VTE events occurred at a median age of 49 years; 30% of the patients had a detectable viral load. “We’re seeing a little more (VTE) than you might expect, and in a younger population than you might have guessed,” said Dr. Crane in an interview.
The most frequent predisposing risk factors were recent hospitalization (40%), infection (40%), or immobilization/bed rest (24%) within the past 90 days, and injectable drug use (22%). “It’s not just the traditional risk factors. Some HIV-specific risk factors are driving this,” said Dr. Crane.
She also aims to learn more about the specifics of risk factors, such as catheter-associated thromboses. The team is working to increase the sample size in order to parse out the relationships with specific outcomes.
In the meantime, the data further characterize the health challenges facing people living with HIV. “This is another example demonstrating that comorbid conditions among patients with HIV that are often considered age related occur at much younger ages in our population,” said Dr. Crane.
SOURCE: Rokx C et al. CROI 2019, Abstract 636; and Tenforde MW et al. CROI 2019, Abstract 637.
.
REPORTING FROM CROI 2019
Very early ART may benefit infants with HIV
SEATTLE – Antiretroviral therapy (ART) in the earliest weeks of life is associated with reduced time to suppression, according to a new study. Each week that treatment was delayed, patients had a 35% reduction in odds of achieving earlier viral load (VL) suppression.
Previous research had already shown that patients treated in the first year of life have better outcomes, including shortened time to viral suppression, and a lower reservoir size. Those studies looked at median age of ART start by month, and a review of the literature revealed that no research had been done on the first month.
The research was presented at the Conference on Retroviruses & Opportunistic Infections by Sara Domínguez Rodríguez, biostatistician and data manager in the pediatric infectious disease service at Hospital 12 Octubre, Madrid.
The researchers retrospectively analyzed 44 patients who were treated in the first 28 days of life, who had uninterrupted ART for at least 2 years, and who had VL measured at least twice during follow-up. Among these, 25 patients received ART in the first week, 19 in weeks 2-4. Patients treated prophylactically with AZT + 3TC (lamivudine) + NVP (nevirapine) any time in the first 15 days of life were considered treated at day 1.
Five of the patients were from the United Kingdom, 23 from Spain, 3 from Italy, and 13 from Thailand. Fifty-seven percent were girls; 35% were preterm. Patients treated in the first week had a higher log10 HIV viral load at ART initiation (P = .02). There was no significant difference between the two groups with respect to CD4 count at ART initiation.
The time to suppression was not significantly different between the two groups, nor was the percentage of patients suppressed at various time points. Patients treated in the first week had reached suppression more often at 3 months and 6 months, but neither result reached statistical significance.
The small sample size of the study produced a challenge, and that led the team to consider suppression time and age as continuous variables. That revealed a curve that favored treatment in the first week of life: Each week of delay reduced the probability of achieving early viral suppression by 35% (hazard ratio, 0.65; 95% confidence interval, 0.46-0.92). “This means that if you delay the age at ART in terms of weeks, the probability of achieving suppression (over) time decreases, and this effect is particularly seen in the first year of follow-up,” Dr. Domínguez Rodríguez said in an interview.
Some might have concerns that treating children too early could have adverse effects. This study, though small, showed promising results. “You might think if you treat very early, maybe the child will not tolerate the medicine or keep on with the treatment, and we saw no difference. So that supports treating early,” senior author Pablo Rojo Conejo, PhD, an infectious disease specialist at Hospital 12 de Octubre, said in an interview.
Others in attendance found the results encouraging. “It’s very good news to see that early starting impacts the life of these children,” Filipe de Barros Perini, MD, head of care and treatment of the HIV program at the Brazilian Ministry of Health, said in an interview.
SOURCE: Domínguez Rodríguez S et al. CROI 2019, Abstract 44.
SEATTLE – Antiretroviral therapy (ART) in the earliest weeks of life is associated with reduced time to suppression, according to a new study. Each week that treatment was delayed, patients had a 35% reduction in odds of achieving earlier viral load (VL) suppression.
Previous research had already shown that patients treated in the first year of life have better outcomes, including shortened time to viral suppression, and a lower reservoir size. Those studies looked at median age of ART start by month, and a review of the literature revealed that no research had been done on the first month.
The research was presented at the Conference on Retroviruses & Opportunistic Infections by Sara Domínguez Rodríguez, biostatistician and data manager in the pediatric infectious disease service at Hospital 12 Octubre, Madrid.
The researchers retrospectively analyzed 44 patients who were treated in the first 28 days of life, who had uninterrupted ART for at least 2 years, and who had VL measured at least twice during follow-up. Among these, 25 patients received ART in the first week, 19 in weeks 2-4. Patients treated prophylactically with AZT + 3TC (lamivudine) + NVP (nevirapine) any time in the first 15 days of life were considered treated at day 1.
Five of the patients were from the United Kingdom, 23 from Spain, 3 from Italy, and 13 from Thailand. Fifty-seven percent were girls; 35% were preterm. Patients treated in the first week had a higher log10 HIV viral load at ART initiation (P = .02). There was no significant difference between the two groups with respect to CD4 count at ART initiation.
The time to suppression was not significantly different between the two groups, nor was the percentage of patients suppressed at various time points. Patients treated in the first week had reached suppression more often at 3 months and 6 months, but neither result reached statistical significance.
The small sample size of the study produced a challenge, and that led the team to consider suppression time and age as continuous variables. That revealed a curve that favored treatment in the first week of life: Each week of delay reduced the probability of achieving early viral suppression by 35% (hazard ratio, 0.65; 95% confidence interval, 0.46-0.92). “This means that if you delay the age at ART in terms of weeks, the probability of achieving suppression (over) time decreases, and this effect is particularly seen in the first year of follow-up,” Dr. Domínguez Rodríguez said in an interview.
Some might have concerns that treating children too early could have adverse effects. This study, though small, showed promising results. “You might think if you treat very early, maybe the child will not tolerate the medicine or keep on with the treatment, and we saw no difference. So that supports treating early,” senior author Pablo Rojo Conejo, PhD, an infectious disease specialist at Hospital 12 de Octubre, said in an interview.
Others in attendance found the results encouraging. “It’s very good news to see that early starting impacts the life of these children,” Filipe de Barros Perini, MD, head of care and treatment of the HIV program at the Brazilian Ministry of Health, said in an interview.
SOURCE: Domínguez Rodríguez S et al. CROI 2019, Abstract 44.
SEATTLE – Antiretroviral therapy (ART) in the earliest weeks of life is associated with reduced time to suppression, according to a new study. Each week that treatment was delayed, patients had a 35% reduction in odds of achieving earlier viral load (VL) suppression.
Previous research had already shown that patients treated in the first year of life have better outcomes, including shortened time to viral suppression, and a lower reservoir size. Those studies looked at median age of ART start by month, and a review of the literature revealed that no research had been done on the first month.
The research was presented at the Conference on Retroviruses & Opportunistic Infections by Sara Domínguez Rodríguez, biostatistician and data manager in the pediatric infectious disease service at Hospital 12 Octubre, Madrid.
The researchers retrospectively analyzed 44 patients who were treated in the first 28 days of life, who had uninterrupted ART for at least 2 years, and who had VL measured at least twice during follow-up. Among these, 25 patients received ART in the first week, 19 in weeks 2-4. Patients treated prophylactically with AZT + 3TC (lamivudine) + NVP (nevirapine) any time in the first 15 days of life were considered treated at day 1.
Five of the patients were from the United Kingdom, 23 from Spain, 3 from Italy, and 13 from Thailand. Fifty-seven percent were girls; 35% were preterm. Patients treated in the first week had a higher log10 HIV viral load at ART initiation (P = .02). There was no significant difference between the two groups with respect to CD4 count at ART initiation.
The time to suppression was not significantly different between the two groups, nor was the percentage of patients suppressed at various time points. Patients treated in the first week had reached suppression more often at 3 months and 6 months, but neither result reached statistical significance.
The small sample size of the study produced a challenge, and that led the team to consider suppression time and age as continuous variables. That revealed a curve that favored treatment in the first week of life: Each week of delay reduced the probability of achieving early viral suppression by 35% (hazard ratio, 0.65; 95% confidence interval, 0.46-0.92). “This means that if you delay the age at ART in terms of weeks, the probability of achieving suppression (over) time decreases, and this effect is particularly seen in the first year of follow-up,” Dr. Domínguez Rodríguez said in an interview.
Some might have concerns that treating children too early could have adverse effects. This study, though small, showed promising results. “You might think if you treat very early, maybe the child will not tolerate the medicine or keep on with the treatment, and we saw no difference. So that supports treating early,” senior author Pablo Rojo Conejo, PhD, an infectious disease specialist at Hospital 12 de Octubre, said in an interview.
Others in attendance found the results encouraging. “It’s very good news to see that early starting impacts the life of these children,” Filipe de Barros Perini, MD, head of care and treatment of the HIV program at the Brazilian Ministry of Health, said in an interview.
SOURCE: Domínguez Rodríguez S et al. CROI 2019, Abstract 44.
REPORTING FROM CROI 2019
For now, HIV cure is worse than infection
SEATTLE – The most important thing to know about the apparent HIV cure widely reported in the press recently is that the treatment was worse than the infection, according to John Mellors, MD, chief of the division of infectious diseases at the University of Pittsburgh.
Dr. Mellors moderated a presentation at the Conference on Retroviruses and Opportunistic Infections about what the cure involved.
An HIV-positive man with advanced Hodgkin’s lymphoma had an allogeneic bone marrow transplant in 2016 after failing first-line chemotherapy and multiple salvage regimens. His donor was homozygous for a gene mutation that prevents HIV from entering new cells. The transplant took; the man’s hematopoietic system was replaced by one with the anti-HIV mutation; and there’s been no trace of active virus in his system since. He’s been off of antiretrovirals for a year and a half. The cancer hasn’t returned.
He’s been dubbed the “London patient.” He joins the “Berlin patient” as the second person who appears to have been freed of infection following a stem cell transplant with the anti-HIV mutation. The Berlin patient recently identified himself as Timothy Ray Brown; he was in the audience at CROI and was applauded for coming forward and sharing his story.
Mr. Brown received a transplant for acute myeloid leukemia and has been off antiretrovirals now for about a decade with no evidence of viral rebound.
Although it didn’t get much attention at CROI, there was a poster of a similar approach seeming to work in a third patient, also with leukemia. It’s been tried – but failed – in two others: one patient died of their lymphoma and the transplant failed in the other, said lead investigator on the London patient case, Ravindra Gupta, MD, a professor in the division of infection & immunity at University College London.
Dr. Mellors pointed out that “the two people who have been cured had lethal malignancies that were unresponsive to conventional therapy and had the extreme measure of an allogeneic bone marrow transplant. Allogeneic bone marrow transplant is not a walk in the park. It has a mortality of 10%-25%, which is completely unacceptable” when “patients feel great on one pill a day” for HIV remission and pretty much have a normal life span.
The transplant reactivated both cytomegalovirus and Epstein Barr virus in the London patient, and he developed graft-versus-host colitis. He survived all three complications.
The take-home message is that “these cases are inspirational. The work ahead is to find out how to deliver the same results with less-extreme measures,” Dr. Mellors said.
The donors for both the London and Berlin patients were homozygous for a delta 32 deletion in the receptor most commonly used by HIV-1 to enter host cells, CCR5. Cells that carry the mutation don’t express the receptor, preventing infection. The mutation prevalence is about 1% among Europeans.
Of the two failed cases, the person who died of lymphoma had a strain of HIV-1 that used a different receptor – CXCR4 – so it’s doubtful the transplant would have worked even if he had survived his cancer.
The London patient wasn’t what’s called an “elite controller,” one of those rare people who suppress HIV without antiretrovirals. His viral loads bounced right back when we was taken off them prior to transplant.
He’s not interested in an active sex life at the moment, Dr. Gupta said, but he might be soon, so Dr. Gupta plans a discussion with him in the near future. Although the London patient could well be immune to HIV that uses the CCR5 receptor, he might not be immune to CXCR4 virus, and he might still be able to produce infectious CCR5 particles. Time will tell.
There was no industry funding for the work. Dr. Gupta is a consultant for ViiV Healthcare and a speaker for Gilead.
SOURCE: Gupta RK et al. CROI 2019, Abstract 29.
SEATTLE – The most important thing to know about the apparent HIV cure widely reported in the press recently is that the treatment was worse than the infection, according to John Mellors, MD, chief of the division of infectious diseases at the University of Pittsburgh.
Dr. Mellors moderated a presentation at the Conference on Retroviruses and Opportunistic Infections about what the cure involved.
An HIV-positive man with advanced Hodgkin’s lymphoma had an allogeneic bone marrow transplant in 2016 after failing first-line chemotherapy and multiple salvage regimens. His donor was homozygous for a gene mutation that prevents HIV from entering new cells. The transplant took; the man’s hematopoietic system was replaced by one with the anti-HIV mutation; and there’s been no trace of active virus in his system since. He’s been off of antiretrovirals for a year and a half. The cancer hasn’t returned.
He’s been dubbed the “London patient.” He joins the “Berlin patient” as the second person who appears to have been freed of infection following a stem cell transplant with the anti-HIV mutation. The Berlin patient recently identified himself as Timothy Ray Brown; he was in the audience at CROI and was applauded for coming forward and sharing his story.
Mr. Brown received a transplant for acute myeloid leukemia and has been off antiretrovirals now for about a decade with no evidence of viral rebound.
Although it didn’t get much attention at CROI, there was a poster of a similar approach seeming to work in a third patient, also with leukemia. It’s been tried – but failed – in two others: one patient died of their lymphoma and the transplant failed in the other, said lead investigator on the London patient case, Ravindra Gupta, MD, a professor in the division of infection & immunity at University College London.
Dr. Mellors pointed out that “the two people who have been cured had lethal malignancies that were unresponsive to conventional therapy and had the extreme measure of an allogeneic bone marrow transplant. Allogeneic bone marrow transplant is not a walk in the park. It has a mortality of 10%-25%, which is completely unacceptable” when “patients feel great on one pill a day” for HIV remission and pretty much have a normal life span.
The transplant reactivated both cytomegalovirus and Epstein Barr virus in the London patient, and he developed graft-versus-host colitis. He survived all three complications.
The take-home message is that “these cases are inspirational. The work ahead is to find out how to deliver the same results with less-extreme measures,” Dr. Mellors said.
The donors for both the London and Berlin patients were homozygous for a delta 32 deletion in the receptor most commonly used by HIV-1 to enter host cells, CCR5. Cells that carry the mutation don’t express the receptor, preventing infection. The mutation prevalence is about 1% among Europeans.
Of the two failed cases, the person who died of lymphoma had a strain of HIV-1 that used a different receptor – CXCR4 – so it’s doubtful the transplant would have worked even if he had survived his cancer.
The London patient wasn’t what’s called an “elite controller,” one of those rare people who suppress HIV without antiretrovirals. His viral loads bounced right back when we was taken off them prior to transplant.
He’s not interested in an active sex life at the moment, Dr. Gupta said, but he might be soon, so Dr. Gupta plans a discussion with him in the near future. Although the London patient could well be immune to HIV that uses the CCR5 receptor, he might not be immune to CXCR4 virus, and he might still be able to produce infectious CCR5 particles. Time will tell.
There was no industry funding for the work. Dr. Gupta is a consultant for ViiV Healthcare and a speaker for Gilead.
SOURCE: Gupta RK et al. CROI 2019, Abstract 29.
SEATTLE – The most important thing to know about the apparent HIV cure widely reported in the press recently is that the treatment was worse than the infection, according to John Mellors, MD, chief of the division of infectious diseases at the University of Pittsburgh.
Dr. Mellors moderated a presentation at the Conference on Retroviruses and Opportunistic Infections about what the cure involved.
An HIV-positive man with advanced Hodgkin’s lymphoma had an allogeneic bone marrow transplant in 2016 after failing first-line chemotherapy and multiple salvage regimens. His donor was homozygous for a gene mutation that prevents HIV from entering new cells. The transplant took; the man’s hematopoietic system was replaced by one with the anti-HIV mutation; and there’s been no trace of active virus in his system since. He’s been off of antiretrovirals for a year and a half. The cancer hasn’t returned.
He’s been dubbed the “London patient.” He joins the “Berlin patient” as the second person who appears to have been freed of infection following a stem cell transplant with the anti-HIV mutation. The Berlin patient recently identified himself as Timothy Ray Brown; he was in the audience at CROI and was applauded for coming forward and sharing his story.
Mr. Brown received a transplant for acute myeloid leukemia and has been off antiretrovirals now for about a decade with no evidence of viral rebound.
Although it didn’t get much attention at CROI, there was a poster of a similar approach seeming to work in a third patient, also with leukemia. It’s been tried – but failed – in two others: one patient died of their lymphoma and the transplant failed in the other, said lead investigator on the London patient case, Ravindra Gupta, MD, a professor in the division of infection & immunity at University College London.
Dr. Mellors pointed out that “the two people who have been cured had lethal malignancies that were unresponsive to conventional therapy and had the extreme measure of an allogeneic bone marrow transplant. Allogeneic bone marrow transplant is not a walk in the park. It has a mortality of 10%-25%, which is completely unacceptable” when “patients feel great on one pill a day” for HIV remission and pretty much have a normal life span.
The transplant reactivated both cytomegalovirus and Epstein Barr virus in the London patient, and he developed graft-versus-host colitis. He survived all three complications.
The take-home message is that “these cases are inspirational. The work ahead is to find out how to deliver the same results with less-extreme measures,” Dr. Mellors said.
The donors for both the London and Berlin patients were homozygous for a delta 32 deletion in the receptor most commonly used by HIV-1 to enter host cells, CCR5. Cells that carry the mutation don’t express the receptor, preventing infection. The mutation prevalence is about 1% among Europeans.
Of the two failed cases, the person who died of lymphoma had a strain of HIV-1 that used a different receptor – CXCR4 – so it’s doubtful the transplant would have worked even if he had survived his cancer.
The London patient wasn’t what’s called an “elite controller,” one of those rare people who suppress HIV without antiretrovirals. His viral loads bounced right back when we was taken off them prior to transplant.
He’s not interested in an active sex life at the moment, Dr. Gupta said, but he might be soon, so Dr. Gupta plans a discussion with him in the near future. Although the London patient could well be immune to HIV that uses the CCR5 receptor, he might not be immune to CXCR4 virus, and he might still be able to produce infectious CCR5 particles. Time will tell.
There was no industry funding for the work. Dr. Gupta is a consultant for ViiV Healthcare and a speaker for Gilead.
SOURCE: Gupta RK et al. CROI 2019, Abstract 29.
REPORTING FROM CROI 2019
HCC with no cirrhosis is more common in HIV patients
SEATTLE – Hepatocellular carcinoma (HCC) is on the rise in HIV-positive individuals, its incidence having quadrupled since 1996, and HIV-positive individuals have about a 300% increase risk of HCC, compared with the general population. However, more than 40% of patients with HIV who develop HCC have a Fibrosis-4 score (FIB-4) suggesting a lack of cirrhosis, according to a new retrospective analysis. By contrast, only about 13% of typical HCC patients have no cirrhosis.
The study also revealed some of the risk factors associated with HCC in this population, including longer duration of HIV viremia and lower CD4 cell counts, as well as markers of metabolic syndrome. “There was some signal that perhaps other markers of metabolic syndrome, obesity and diabetes, were more prevalent in those [who developed HCC] without advanced fibrosis or cirrhosis, suggesting that there may be other underlying etiologies of liver disease that we should be wary of when evaluating somebody for their risk of HCC,” Jessie Torgersen, MD, said in an interview.
Dr. Torgersen is an instructor of medicine at the University of Pennsylvania, Philadelphia. She presented the study at the Conference on Retroviruses & Opportunistic Infections.
The results of the study are tantalizing, but not yet practice changing. “I don’t think we have enough information from this study to recommend a dramatic overhaul of the current HCC screening guidelines, but with the anticipated elimination of hepatitis C, I think the emergence of [metabolic factors and their] contributions to our HIV-positive population’s risk of HCC needs to be better understood. Hopefully this will serve as a first step in further understanding those risks,” Dr. Torgersen said.
She also hopes to get a better handle on the biological mechanisms that might drive HCC in the absence of cirrhosis. “While the mechanisms are unclear as to why HCC would develop in HIV-positive patients without cirrhosis, there are a lot of biologically plausible mechanisms that seem to make [sense],” said Dr. Torgersen. The team hopes to get a better understanding of those mechanisms in order to information evaluation and screening for HCC.
The researchers analyzed data from the Veterans Affairs Cancer Registry as well as EMRs for HIV-positive veterans across the United States. The study included 2,497 participants with a FIB-4 score greater than 3.25, and 29,836 with an FIB-4 score less than or equal to 3.25. At baseline, subjects with FIB-4 greater than 3.25 were more likely to have an alcohol-related diagnosis (47% vs. 29%), be positive for hepatitis C virus RNA (59% vs. 30%), be positive for the hepatitis B surface antigen (10% versus 5%), have HIV RNA greater than or equal to 500 copies/mL (63% vs. 56%), and to have a CD4+ cell count less than 200 cells/m3 (39% vs. 26%).
A total of 278 subjects were diagnosed with HCC; 43% had an FIB-4 less than or equal to 3.25. Among those 43%, more patients had a body mass index of 30 or higher (16% vs. 12%), had diabetes (31% vs. 25%), and tested positive for the hepatitis B surface antigen (26% vs. 17%).
Among subjects with FIB-4 less than or equal to 3.25, factors associated with greater HCC risk included higher HIV RNA level (hazard ratio, 1.24 per 1.0 log10 copies/mL), CD4+ cell count less than 200 cells/m3 (HR, 1.78), hepatitis C virus infection (HR, 6.32), and positive hepatitis B surface antigen (HR, 4.93).
Among subjects with FIB-4 greater than 3.25, increased HCC risk was associated with HCV infection (HR, 6.18) and positive hepatitis B surface antigen (HR, 2.12).
The study was funded by the National Institutes of Health. Dr. Torgersen reported no financial disclosures.
SOURCE: Torgersen J et al. CROI 2019, Abstract 90.
SEATTLE – Hepatocellular carcinoma (HCC) is on the rise in HIV-positive individuals, its incidence having quadrupled since 1996, and HIV-positive individuals have about a 300% increase risk of HCC, compared with the general population. However, more than 40% of patients with HIV who develop HCC have a Fibrosis-4 score (FIB-4) suggesting a lack of cirrhosis, according to a new retrospective analysis. By contrast, only about 13% of typical HCC patients have no cirrhosis.
The study also revealed some of the risk factors associated with HCC in this population, including longer duration of HIV viremia and lower CD4 cell counts, as well as markers of metabolic syndrome. “There was some signal that perhaps other markers of metabolic syndrome, obesity and diabetes, were more prevalent in those [who developed HCC] without advanced fibrosis or cirrhosis, suggesting that there may be other underlying etiologies of liver disease that we should be wary of when evaluating somebody for their risk of HCC,” Jessie Torgersen, MD, said in an interview.
Dr. Torgersen is an instructor of medicine at the University of Pennsylvania, Philadelphia. She presented the study at the Conference on Retroviruses & Opportunistic Infections.
The results of the study are tantalizing, but not yet practice changing. “I don’t think we have enough information from this study to recommend a dramatic overhaul of the current HCC screening guidelines, but with the anticipated elimination of hepatitis C, I think the emergence of [metabolic factors and their] contributions to our HIV-positive population’s risk of HCC needs to be better understood. Hopefully this will serve as a first step in further understanding those risks,” Dr. Torgersen said.
She also hopes to get a better handle on the biological mechanisms that might drive HCC in the absence of cirrhosis. “While the mechanisms are unclear as to why HCC would develop in HIV-positive patients without cirrhosis, there are a lot of biologically plausible mechanisms that seem to make [sense],” said Dr. Torgersen. The team hopes to get a better understanding of those mechanisms in order to information evaluation and screening for HCC.
The researchers analyzed data from the Veterans Affairs Cancer Registry as well as EMRs for HIV-positive veterans across the United States. The study included 2,497 participants with a FIB-4 score greater than 3.25, and 29,836 with an FIB-4 score less than or equal to 3.25. At baseline, subjects with FIB-4 greater than 3.25 were more likely to have an alcohol-related diagnosis (47% vs. 29%), be positive for hepatitis C virus RNA (59% vs. 30%), be positive for the hepatitis B surface antigen (10% versus 5%), have HIV RNA greater than or equal to 500 copies/mL (63% vs. 56%), and to have a CD4+ cell count less than 200 cells/m3 (39% vs. 26%).
A total of 278 subjects were diagnosed with HCC; 43% had an FIB-4 less than or equal to 3.25. Among those 43%, more patients had a body mass index of 30 or higher (16% vs. 12%), had diabetes (31% vs. 25%), and tested positive for the hepatitis B surface antigen (26% vs. 17%).
Among subjects with FIB-4 less than or equal to 3.25, factors associated with greater HCC risk included higher HIV RNA level (hazard ratio, 1.24 per 1.0 log10 copies/mL), CD4+ cell count less than 200 cells/m3 (HR, 1.78), hepatitis C virus infection (HR, 6.32), and positive hepatitis B surface antigen (HR, 4.93).
Among subjects with FIB-4 greater than 3.25, increased HCC risk was associated with HCV infection (HR, 6.18) and positive hepatitis B surface antigen (HR, 2.12).
The study was funded by the National Institutes of Health. Dr. Torgersen reported no financial disclosures.
SOURCE: Torgersen J et al. CROI 2019, Abstract 90.
SEATTLE – Hepatocellular carcinoma (HCC) is on the rise in HIV-positive individuals, its incidence having quadrupled since 1996, and HIV-positive individuals have about a 300% increase risk of HCC, compared with the general population. However, more than 40% of patients with HIV who develop HCC have a Fibrosis-4 score (FIB-4) suggesting a lack of cirrhosis, according to a new retrospective analysis. By contrast, only about 13% of typical HCC patients have no cirrhosis.
The study also revealed some of the risk factors associated with HCC in this population, including longer duration of HIV viremia and lower CD4 cell counts, as well as markers of metabolic syndrome. “There was some signal that perhaps other markers of metabolic syndrome, obesity and diabetes, were more prevalent in those [who developed HCC] without advanced fibrosis or cirrhosis, suggesting that there may be other underlying etiologies of liver disease that we should be wary of when evaluating somebody for their risk of HCC,” Jessie Torgersen, MD, said in an interview.
Dr. Torgersen is an instructor of medicine at the University of Pennsylvania, Philadelphia. She presented the study at the Conference on Retroviruses & Opportunistic Infections.
The results of the study are tantalizing, but not yet practice changing. “I don’t think we have enough information from this study to recommend a dramatic overhaul of the current HCC screening guidelines, but with the anticipated elimination of hepatitis C, I think the emergence of [metabolic factors and their] contributions to our HIV-positive population’s risk of HCC needs to be better understood. Hopefully this will serve as a first step in further understanding those risks,” Dr. Torgersen said.
She also hopes to get a better handle on the biological mechanisms that might drive HCC in the absence of cirrhosis. “While the mechanisms are unclear as to why HCC would develop in HIV-positive patients without cirrhosis, there are a lot of biologically plausible mechanisms that seem to make [sense],” said Dr. Torgersen. The team hopes to get a better understanding of those mechanisms in order to information evaluation and screening for HCC.
The researchers analyzed data from the Veterans Affairs Cancer Registry as well as EMRs for HIV-positive veterans across the United States. The study included 2,497 participants with a FIB-4 score greater than 3.25, and 29,836 with an FIB-4 score less than or equal to 3.25. At baseline, subjects with FIB-4 greater than 3.25 were more likely to have an alcohol-related diagnosis (47% vs. 29%), be positive for hepatitis C virus RNA (59% vs. 30%), be positive for the hepatitis B surface antigen (10% versus 5%), have HIV RNA greater than or equal to 500 copies/mL (63% vs. 56%), and to have a CD4+ cell count less than 200 cells/m3 (39% vs. 26%).
A total of 278 subjects were diagnosed with HCC; 43% had an FIB-4 less than or equal to 3.25. Among those 43%, more patients had a body mass index of 30 or higher (16% vs. 12%), had diabetes (31% vs. 25%), and tested positive for the hepatitis B surface antigen (26% vs. 17%).
Among subjects with FIB-4 less than or equal to 3.25, factors associated with greater HCC risk included higher HIV RNA level (hazard ratio, 1.24 per 1.0 log10 copies/mL), CD4+ cell count less than 200 cells/m3 (HR, 1.78), hepatitis C virus infection (HR, 6.32), and positive hepatitis B surface antigen (HR, 4.93).
Among subjects with FIB-4 greater than 3.25, increased HCC risk was associated with HCV infection (HR, 6.18) and positive hepatitis B surface antigen (HR, 2.12).
The study was funded by the National Institutes of Health. Dr. Torgersen reported no financial disclosures.
SOURCE: Torgersen J et al. CROI 2019, Abstract 90.
REPORTING FROM CROI 2019
ICYMI: Noninferior tuberculosis prevention in HIV has shorter duration
The primary endpoint – first case of tuberculosis or death from tuberculosis or unknown cause among patients with HIV – was reported in 2% of both arms in the open-label, phase 3, noninferiority trial BRIEF TB (NCT01404312).
In the New England Journal of Medicine (2019 Mar 14;380[11]:1001-11), 3,000 patients with HIV were randomized to receive either 1 month of rifapentine/isoniazid or 9 months of isoniazid monotherapy for prevention of tuberculosis. Although safety was also similar between arms, the completion rate was significantly higher in the combination treatment arm, compared with the monotherapy arm (97% vs. 90%; P less than .001).
We covered this story at the annual Conference on Retroviruses & Opportunistic Infections. See our coverage at the link below.
The primary endpoint – first case of tuberculosis or death from tuberculosis or unknown cause among patients with HIV – was reported in 2% of both arms in the open-label, phase 3, noninferiority trial BRIEF TB (NCT01404312).
In the New England Journal of Medicine (2019 Mar 14;380[11]:1001-11), 3,000 patients with HIV were randomized to receive either 1 month of rifapentine/isoniazid or 9 months of isoniazid monotherapy for prevention of tuberculosis. Although safety was also similar between arms, the completion rate was significantly higher in the combination treatment arm, compared with the monotherapy arm (97% vs. 90%; P less than .001).
We covered this story at the annual Conference on Retroviruses & Opportunistic Infections. See our coverage at the link below.
The primary endpoint – first case of tuberculosis or death from tuberculosis or unknown cause among patients with HIV – was reported in 2% of both arms in the open-label, phase 3, noninferiority trial BRIEF TB (NCT01404312).
In the New England Journal of Medicine (2019 Mar 14;380[11]:1001-11), 3,000 patients with HIV were randomized to receive either 1 month of rifapentine/isoniazid or 9 months of isoniazid monotherapy for prevention of tuberculosis. Although safety was also similar between arms, the completion rate was significantly higher in the combination treatment arm, compared with the monotherapy arm (97% vs. 90%; P less than .001).
We covered this story at the annual Conference on Retroviruses & Opportunistic Infections. See our coverage at the link below.
FROM NEW ENGLAND JOURNAL OF MEDICINE
Descovy noninferior to Truvada for PrEP
SEATTLE – Descovy [emtricitabine/tenofovir alafenamide (F/TAF]) was noninferior to Truvada [emtricitabine/tenofovir disoproxil fumarate (F/TDF)] for preexposure HIV prophylaxis in a blinded, randomized trial involving more than 5,000 men at high risk for the infection.
Both nucleotide reverse transcriptase inhibitors are made by Gilead, and the company funded the trial.
F/TDF (Truvada) has been a blockbuster for the company, both for HIV treatment and, since 2012, for preexposure prophylaxis (PrEP); it’s the only medication to carry the indication. However, F/TDF is set to go off patent in 2021, so the company has turned its development efforts to a successor, F/TAF (Descovy), a prodrug of tenofovir that is already approved for HIV treatment.
The new study builds a case for F/TAF for PrEP, but whether the results are strong enough to persuade people to opt for it over a much less expensive generic version of F/TDF remains to be seen.
The trial randomized 2,694 men who have sex with men to F/TAF, and 2,693 to F/TDF for up to 96 weeks. Entrance criteria included at least two episodes of unprotected anal sex in the previous 12 weeks, or a diagnosis of rectal gonorrhea, chlamydia, or syphilis in the previous 6 months.
More than half the men contracted at least one sexually transmitted infection during the trial, “which indicated to us that these were the right patients to be enrolled in the study,” lead investigator Charles Hare, MD, an infectious disease specialist at the University of California, San Francisco, said at the Conference on Retroviruses and Opportunistic Infections.
There were 15 new HIV infections in the F/TDF group (0.34 per 100 person-years), versus 7 in the F/TAF group (0.16 per 100 person-years). Almost all of the new infections were due to poor adherence – as proven by blood levels and dry blood spot testing – and most of the rest were in men who probably entered the trial with newly acquired HIV.
When those subjects were excluded, there were just two new onset HIV cases in subjects adherent to dosing, one in each arm. Infection rates were far lower than would have been expected had the subjects not been on PrEP.
One of Gilead’s main selling points for F/TAF over F/TDF is that the newer drug has better bone and renal safety, and there were slight biomarker differences in the trial that supported the assertion.
For instance, spine bone mineral density decreased 3% or greater from baseline in 10 F/TAF patients, but 27 men on F/TDF (P less than .001). Results were similar with hip bone density.
On the renal front, estimated glomerular filtration rates fell a median of 2.3 mL/min per 1.73 m2 in the F/TDF arm, but rose 1.8 mL/min per 1.73 m2 in men on F/TAF (P less than .001). Proximal tubular protein to creatinine ratios were largely unchanged from baseline with F/TAF, but slightly higher in the F/TDF group, at 48 weeks.
There were no statistically significant differences on actual safety outcomes – as opposed to biomarkers – between the two drugs or discontinuations due to side effects, which were rare and most often due to gastrointestinal issues. F/TAF patients gained about 1.1 kg in the trial, while weight held steady in the F/TDF arm. The study team plans to analyze lipid profile differences between the groups, since concern has been raised about F/TAF’s effect on them.
In a press conference at the conference, there was quite a bit of discussion about whether the results would justify using F/TAF for PrEP when less expensive generic versions of F/TDF become available.
“That’s a great question,” Dr. Hare said. “Both drugs actually performed quite well,” and both “do pretty well in terms of safety in this population.”
It’s not known at this point if the biomarker differences will prove to be clinically relevant. Hip fractures, kidney failure, and other problems are so rare in young, relatively healthy PrEP users that a trial to demonstrate clinical relevance would have to be huge, with years-long follow-up, Dr. Hare noted.
The average age of the men in this study was 36 years. Most were white, and about 60% lived in the United States. Other participants were from Canada or Europe.
The work was funded by Gilead; five investigators, including the senior investigator, were employees. Dr. Hare is an investigator for the company.
SOURCE: Hare CB et al. CROI 2019, Abstract 104 LB.
SEATTLE – Descovy [emtricitabine/tenofovir alafenamide (F/TAF]) was noninferior to Truvada [emtricitabine/tenofovir disoproxil fumarate (F/TDF)] for preexposure HIV prophylaxis in a blinded, randomized trial involving more than 5,000 men at high risk for the infection.
Both nucleotide reverse transcriptase inhibitors are made by Gilead, and the company funded the trial.
F/TDF (Truvada) has been a blockbuster for the company, both for HIV treatment and, since 2012, for preexposure prophylaxis (PrEP); it’s the only medication to carry the indication. However, F/TDF is set to go off patent in 2021, so the company has turned its development efforts to a successor, F/TAF (Descovy), a prodrug of tenofovir that is already approved for HIV treatment.
The new study builds a case for F/TAF for PrEP, but whether the results are strong enough to persuade people to opt for it over a much less expensive generic version of F/TDF remains to be seen.
The trial randomized 2,694 men who have sex with men to F/TAF, and 2,693 to F/TDF for up to 96 weeks. Entrance criteria included at least two episodes of unprotected anal sex in the previous 12 weeks, or a diagnosis of rectal gonorrhea, chlamydia, or syphilis in the previous 6 months.
More than half the men contracted at least one sexually transmitted infection during the trial, “which indicated to us that these were the right patients to be enrolled in the study,” lead investigator Charles Hare, MD, an infectious disease specialist at the University of California, San Francisco, said at the Conference on Retroviruses and Opportunistic Infections.
There were 15 new HIV infections in the F/TDF group (0.34 per 100 person-years), versus 7 in the F/TAF group (0.16 per 100 person-years). Almost all of the new infections were due to poor adherence – as proven by blood levels and dry blood spot testing – and most of the rest were in men who probably entered the trial with newly acquired HIV.
When those subjects were excluded, there were just two new onset HIV cases in subjects adherent to dosing, one in each arm. Infection rates were far lower than would have been expected had the subjects not been on PrEP.
One of Gilead’s main selling points for F/TAF over F/TDF is that the newer drug has better bone and renal safety, and there were slight biomarker differences in the trial that supported the assertion.
For instance, spine bone mineral density decreased 3% or greater from baseline in 10 F/TAF patients, but 27 men on F/TDF (P less than .001). Results were similar with hip bone density.
On the renal front, estimated glomerular filtration rates fell a median of 2.3 mL/min per 1.73 m2 in the F/TDF arm, but rose 1.8 mL/min per 1.73 m2 in men on F/TAF (P less than .001). Proximal tubular protein to creatinine ratios were largely unchanged from baseline with F/TAF, but slightly higher in the F/TDF group, at 48 weeks.
There were no statistically significant differences on actual safety outcomes – as opposed to biomarkers – between the two drugs or discontinuations due to side effects, which were rare and most often due to gastrointestinal issues. F/TAF patients gained about 1.1 kg in the trial, while weight held steady in the F/TDF arm. The study team plans to analyze lipid profile differences between the groups, since concern has been raised about F/TAF’s effect on them.
In a press conference at the conference, there was quite a bit of discussion about whether the results would justify using F/TAF for PrEP when less expensive generic versions of F/TDF become available.
“That’s a great question,” Dr. Hare said. “Both drugs actually performed quite well,” and both “do pretty well in terms of safety in this population.”
It’s not known at this point if the biomarker differences will prove to be clinically relevant. Hip fractures, kidney failure, and other problems are so rare in young, relatively healthy PrEP users that a trial to demonstrate clinical relevance would have to be huge, with years-long follow-up, Dr. Hare noted.
The average age of the men in this study was 36 years. Most were white, and about 60% lived in the United States. Other participants were from Canada or Europe.
The work was funded by Gilead; five investigators, including the senior investigator, were employees. Dr. Hare is an investigator for the company.
SOURCE: Hare CB et al. CROI 2019, Abstract 104 LB.
SEATTLE – Descovy [emtricitabine/tenofovir alafenamide (F/TAF]) was noninferior to Truvada [emtricitabine/tenofovir disoproxil fumarate (F/TDF)] for preexposure HIV prophylaxis in a blinded, randomized trial involving more than 5,000 men at high risk for the infection.
Both nucleotide reverse transcriptase inhibitors are made by Gilead, and the company funded the trial.
F/TDF (Truvada) has been a blockbuster for the company, both for HIV treatment and, since 2012, for preexposure prophylaxis (PrEP); it’s the only medication to carry the indication. However, F/TDF is set to go off patent in 2021, so the company has turned its development efforts to a successor, F/TAF (Descovy), a prodrug of tenofovir that is already approved for HIV treatment.
The new study builds a case for F/TAF for PrEP, but whether the results are strong enough to persuade people to opt for it over a much less expensive generic version of F/TDF remains to be seen.
The trial randomized 2,694 men who have sex with men to F/TAF, and 2,693 to F/TDF for up to 96 weeks. Entrance criteria included at least two episodes of unprotected anal sex in the previous 12 weeks, or a diagnosis of rectal gonorrhea, chlamydia, or syphilis in the previous 6 months.
More than half the men contracted at least one sexually transmitted infection during the trial, “which indicated to us that these were the right patients to be enrolled in the study,” lead investigator Charles Hare, MD, an infectious disease specialist at the University of California, San Francisco, said at the Conference on Retroviruses and Opportunistic Infections.
There were 15 new HIV infections in the F/TDF group (0.34 per 100 person-years), versus 7 in the F/TAF group (0.16 per 100 person-years). Almost all of the new infections were due to poor adherence – as proven by blood levels and dry blood spot testing – and most of the rest were in men who probably entered the trial with newly acquired HIV.
When those subjects were excluded, there were just two new onset HIV cases in subjects adherent to dosing, one in each arm. Infection rates were far lower than would have been expected had the subjects not been on PrEP.
One of Gilead’s main selling points for F/TAF over F/TDF is that the newer drug has better bone and renal safety, and there were slight biomarker differences in the trial that supported the assertion.
For instance, spine bone mineral density decreased 3% or greater from baseline in 10 F/TAF patients, but 27 men on F/TDF (P less than .001). Results were similar with hip bone density.
On the renal front, estimated glomerular filtration rates fell a median of 2.3 mL/min per 1.73 m2 in the F/TDF arm, but rose 1.8 mL/min per 1.73 m2 in men on F/TAF (P less than .001). Proximal tubular protein to creatinine ratios were largely unchanged from baseline with F/TAF, but slightly higher in the F/TDF group, at 48 weeks.
There were no statistically significant differences on actual safety outcomes – as opposed to biomarkers – between the two drugs or discontinuations due to side effects, which were rare and most often due to gastrointestinal issues. F/TAF patients gained about 1.1 kg in the trial, while weight held steady in the F/TDF arm. The study team plans to analyze lipid profile differences between the groups, since concern has been raised about F/TAF’s effect on them.
In a press conference at the conference, there was quite a bit of discussion about whether the results would justify using F/TAF for PrEP when less expensive generic versions of F/TDF become available.
“That’s a great question,” Dr. Hare said. “Both drugs actually performed quite well,” and both “do pretty well in terms of safety in this population.”
It’s not known at this point if the biomarker differences will prove to be clinically relevant. Hip fractures, kidney failure, and other problems are so rare in young, relatively healthy PrEP users that a trial to demonstrate clinical relevance would have to be huge, with years-long follow-up, Dr. Hare noted.
The average age of the men in this study was 36 years. Most were white, and about 60% lived in the United States. Other participants were from Canada or Europe.
The work was funded by Gilead; five investigators, including the senior investigator, were employees. Dr. Hare is an investigator for the company.
SOURCE: Hare CB et al. CROI 2019, Abstract 104 LB.
REPORTING FROM CROI 2019