User login
STS, new president apologize for predecessor’s speech amid Twitter backlash
The Society of Thoracic Surgeons (STS) and its newly installed president have posted an apology for a speech delivered by its outgoing president that appeared, in part, to disparage affirmative action as a means to promote diversity, equity, and inclusion in the field.
The speech, entitled “Three Score & More,” presented Jan. 22 at the STS 58th annual meeting in San Diego by John H. Calhoon, MD, University of Texas Health Science Center at San Antonio, unleashed a cascade of tweets, some circumspect but many expressing outrage and dismay.
Many of the tweets were from individuals who acknowledged not hearing the speech but who had seen at least one accompanying slide which, by then, had been widely circulated on the platform. It contained phrases such as “Affirmative Action is not equal opportunity” and “Defining people by color, gender, religion only tends to ingrain bias and discrimination,” all under the heading of “Virtuous Ideals.”
Reactions on Twitter included comments such as “This is bad beyond description” and a description of the slide’s content as “the blueprint & thought process for those actively maintaining Whiteness & the Patriarchy in medicine.”
Following an early onslaught of such tweets, the STS and new president Thomas E. MacGillivray, MD, MedStar Health, Washington, issued a statement disowning at least the controversial parts of Dr. Calhoon’s presentation, stating they were “inconsistent with STS’s core values of diversity, equity, and inclusion.”
The post continues, “The STS apologizes for these remarks. We know these comments were hurtful and we regret the pain they have caused to so many valued colleagues.” It then states, “Diversity, equity, and inclusion are central principles of our Society, and what we strive for in our profession and our practice. STS is committed to learning from this experience and taking action to reinforce our commitment to these values.”
“I believe that either the slide and/or my remarks were misinterpreted by some. I don’t want to hurt anybody. I’m profoundly sorry and apologize,” Dr. Calhoon said in an interview.
“I’m proud of my own group’s record on diversity and using equity and inclusion to get there,” he said. “We’re committed to it. We’ve had a wonderfully diverse group. I tried to highlight that in my remarks.”
About the Twitter response to the slide in question, Dr. Calhoon said, “I have no idea how they were thinking.” He added, “I can only comment that I’m really proud of our record and, for that matter, the STS’s record on diversity, equity, and inclusion.”
A version of this article first appeared on Medscape.com.
The Society of Thoracic Surgeons (STS) and its newly installed president have posted an apology for a speech delivered by its outgoing president that appeared, in part, to disparage affirmative action as a means to promote diversity, equity, and inclusion in the field.
The speech, entitled “Three Score & More,” presented Jan. 22 at the STS 58th annual meeting in San Diego by John H. Calhoon, MD, University of Texas Health Science Center at San Antonio, unleashed a cascade of tweets, some circumspect but many expressing outrage and dismay.
Many of the tweets were from individuals who acknowledged not hearing the speech but who had seen at least one accompanying slide which, by then, had been widely circulated on the platform. It contained phrases such as “Affirmative Action is not equal opportunity” and “Defining people by color, gender, religion only tends to ingrain bias and discrimination,” all under the heading of “Virtuous Ideals.”
Reactions on Twitter included comments such as “This is bad beyond description” and a description of the slide’s content as “the blueprint & thought process for those actively maintaining Whiteness & the Patriarchy in medicine.”
Following an early onslaught of such tweets, the STS and new president Thomas E. MacGillivray, MD, MedStar Health, Washington, issued a statement disowning at least the controversial parts of Dr. Calhoon’s presentation, stating they were “inconsistent with STS’s core values of diversity, equity, and inclusion.”
The post continues, “The STS apologizes for these remarks. We know these comments were hurtful and we regret the pain they have caused to so many valued colleagues.” It then states, “Diversity, equity, and inclusion are central principles of our Society, and what we strive for in our profession and our practice. STS is committed to learning from this experience and taking action to reinforce our commitment to these values.”
“I believe that either the slide and/or my remarks were misinterpreted by some. I don’t want to hurt anybody. I’m profoundly sorry and apologize,” Dr. Calhoon said in an interview.
“I’m proud of my own group’s record on diversity and using equity and inclusion to get there,” he said. “We’re committed to it. We’ve had a wonderfully diverse group. I tried to highlight that in my remarks.”
About the Twitter response to the slide in question, Dr. Calhoon said, “I have no idea how they were thinking.” He added, “I can only comment that I’m really proud of our record and, for that matter, the STS’s record on diversity, equity, and inclusion.”
A version of this article first appeared on Medscape.com.
The Society of Thoracic Surgeons (STS) and its newly installed president have posted an apology for a speech delivered by its outgoing president that appeared, in part, to disparage affirmative action as a means to promote diversity, equity, and inclusion in the field.
The speech, entitled “Three Score & More,” presented Jan. 22 at the STS 58th annual meeting in San Diego by John H. Calhoon, MD, University of Texas Health Science Center at San Antonio, unleashed a cascade of tweets, some circumspect but many expressing outrage and dismay.
Many of the tweets were from individuals who acknowledged not hearing the speech but who had seen at least one accompanying slide which, by then, had been widely circulated on the platform. It contained phrases such as “Affirmative Action is not equal opportunity” and “Defining people by color, gender, religion only tends to ingrain bias and discrimination,” all under the heading of “Virtuous Ideals.”
Reactions on Twitter included comments such as “This is bad beyond description” and a description of the slide’s content as “the blueprint & thought process for those actively maintaining Whiteness & the Patriarchy in medicine.”
Following an early onslaught of such tweets, the STS and new president Thomas E. MacGillivray, MD, MedStar Health, Washington, issued a statement disowning at least the controversial parts of Dr. Calhoon’s presentation, stating they were “inconsistent with STS’s core values of diversity, equity, and inclusion.”
The post continues, “The STS apologizes for these remarks. We know these comments were hurtful and we regret the pain they have caused to so many valued colleagues.” It then states, “Diversity, equity, and inclusion are central principles of our Society, and what we strive for in our profession and our practice. STS is committed to learning from this experience and taking action to reinforce our commitment to these values.”
“I believe that either the slide and/or my remarks were misinterpreted by some. I don’t want to hurt anybody. I’m profoundly sorry and apologize,” Dr. Calhoon said in an interview.
“I’m proud of my own group’s record on diversity and using equity and inclusion to get there,” he said. “We’re committed to it. We’ve had a wonderfully diverse group. I tried to highlight that in my remarks.”
About the Twitter response to the slide in question, Dr. Calhoon said, “I have no idea how they were thinking.” He added, “I can only comment that I’m really proud of our record and, for that matter, the STS’s record on diversity, equity, and inclusion.”
A version of this article first appeared on Medscape.com.
Damar Hamlin’s cardiac arrest: Key lessons
This discussion was recorded on Jan. 9, 2023. This transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome. I’m Dr. Robert D. Glatter, medical adviser for Medscape Emergency Medicine. Today, we have Dr. Paul E. Pepe, an emergency medicine physician based in Florida and a highly recognized expert in emergency medical services (EMS), critical care, sports and event medicine, and resuscitation. Also joining us is Dr. Michael S. (“Mick”) Malloy, an emergency medicine physician based in Ireland, also an expert in prehospital care, resuscitation, and sports and event medicine. Welcome, gentlemen.
Dr. Pepe: Thanks for having us here.
Dr. Glatter: the Buffalo Bills safety who went down suffering a cardiac arrest in front of millions of people. Although we don’t know the exact cause of the events that transpired, the goal of our discussion is to guide our audience through a systematic approach to evaluation and management of an athlete suffering blunt force chest and neck trauma, and then suffering a cardiac arrest. We do know, obviously, that Damar was successfully resuscitated, thanks to the medical staff and trainers.
Almost 50 years ago, Chuck Hughes, a Detroit Lions receiver, went down and died with just a minute to go in the game and, unfortunately, didn’t survive.
Paul, can you tell me your impressions after viewing the replay of the events that evening? What were the most likely causes of this syncopal event and the subsequent cardiac arrest?
Dr. Pepe: We don’t know anything specifically. It’s being kept private about what the events were. It’s a little bit complicated in a sense that he basically had an extended resuscitation in the hospital. My experience has been that most people that have ventricular fibrillation, from whatever cause, will most likely be waking up on the field if you get to them. I’ve had personal experience with that.
More importantly than when it starts, when someone goes down on the field, both Dr. Malloy and I take a broader view. We don’t get tunnel vision and think, “Oh, it was a traumatic event,” or “It was cardiac event,” and we just have our minds open. There are many things that could make you stop breathing on the field. It could be a neck or a severe head injury, and then any kind of other internal injury that occurs.
When I saw in the video that Damar Hamlin stood up, that made it a less likely to be a spinal injury. He seemed to be physically functioning, and then he suddenly collapsed. That went along with something that looks like a ventricular fibrillation or ventricular tachycardia type of event and made me think right away that it was commotio cordis. I’m not a Latin scholar, but commotio is like commotion. A literal translation might be an agitation of the heart. I was thinking that he probably got hit somewhere in the middle of the chest at the right moment where the heart is resetting in that repolarization phase, like an R-on-T phenomenon, and then caused this sudden ventricular dysrhythmia.
Most people associate it to that because we have a couple of dozen cases a year of people getting hockey pucks or a baseball hitting their chest, which is very common with adolescents. On the other hand, you can’t get it from a blunt injury like this, and it was too early for it to be, say, a direct cardiac contusion, unless there was a direct injury there. It just happened so quickly.
In Europe, they’ve had a large amount of experience with this same kind of problem before, even just from a direct shoulder hit, for example. Mick Malloy is the dean of the faculty of sports and exercise medicine at the Royal College of Surgeons in Ireland and has vast experience, and now he is the person overseeing the procedures for this. Mick, have you had those kinds of experiences as well?
Dr. Molloy: Yes. It’s something that has occurred over recent decades and has been more recognized. I note that in professional sports, it’s a very different thing because you’ve got such huge teams and teams trained to respond very quickly. And that’s the most important thing in this scenario – having a team that is well functioning as a high-class emergency response team ready to get out on to that field very quickly after the person collapses, getting the automated external defibrillator (AED) on, and then recognizing whether there needs to be a shock given or not. The machine will tell you all that.
In our scenario, we run courses called CARES (Care of the Athlete Resuscitation and Emergencies in Sport) to make sure that our team physicians and team physiotherapists and trainers are all speaking as one when an emergency arises.
I don’t worry so much about the professional sport. It’s more with the amateur sports and the kids sports that I get a bit more concerned because there isn’t the same level of medical care there. Having everybody trained in basic life support would be very important to reduce unnecessary deaths from these types of conditions.
As Paul mentioned, there is a very specific cardiac cause in some of these circumstances, where you get hit just at the wrong time and that hit occurs at a particular electrical point in time. It causes this ventricular fibrillation, and the only real treatment there is the defibrillator as quickly as possible.
Dr. Glatter: What you’re saying ultimately is an important part about rapid defibrillation, and at first, cardiopulmonary resuscitation (CPR). People are concerned about whether they should begin CPR. We’re talking about out-of-hospital cardiac arrest that is outside of a football stadium, for example. Some people are obsessed with taking a person’s pulse, and that’s been a point of contention. If someone is unconscious and not breathing, we should start CPR. Wouldn›t you agree? They will wake up quickly if you begin chest compressions if they’re not necessary.
Dr. Pepe: I tell people, just do it. You’re right, people will wake up and feel it if they don’t need it.
Getting back to Mick’s point of having things ready to go, for example, 8 years ago, we had a professional player on the bench who suddenly collapsed right there in front of the entire audience. We immediately did CPR, and we got the AED on. We shocked him and he was ready, willing, and able to get back on the bench again. It turns out he had underlying coronary artery disease, but we got him back right away.
I did an initial study where we placed an AED in a public place at the Chicago O’Hare Airport to see if the public would use these. Most cardiac arrests occur at home, of course, but in public places, that was a good place to try it. We had almost 10 cases the first year. What was fascinating was that we had almost no survivors over the previous decade, even though there were paramedics at the airport. When we put these out there, we had nine people go down that first year, and six people who had never operated an AED or seen one before knew to get one and use it. Every one of those people survived neurologically intact, and almost every person was waking up before traditional responders got there. That’s how effective this is, but you need to know where the AED is.
Dr. Glatter: How to turn it on, where it is, and how to operate it.
Dr. Pepe: That was the point: These rescuers saved lives in the first year, and it was tremendous. Two points I make about it are that one, you need to know where it is, and two, just go turn it on. It gives you the instructions to follow through; just be in the Nike mode, because it basically won’t hurt a person. It’s rare that there’s ever been any complication of that. The machine algorithms are so good.
Dr. Glatter: Mick, I want to turn to you about the European experience. Specifically in Denmark, we know that there’s a large public health initiative to have AEDs accessible. There have been studies showing that when the public is engaged, especially with studies looking at an app when access is available, survivability doubled in the past 10 years from having access to AEDs. What’s your experience in Ireland in terms of public access to defibrillators?
Dr. Molloy: We’ve got two different streams here. There was a big push to have more AEDs at all sports venues. That was great, but some of the sporting clubs put them inside the locked door. I said that there’s no point to that because nobody can access it. You need to have an external building and you need to leave it open. If somebody needs to use it, they need to know how to get it, open it, and get away, and not get in through a locked door to get access to a defibrillator. We have AEDs now in most stadiums and even in small rural areas, where you might have only 200 people turn up for a game.
From another public access side, if you dial in – in our scenario, it’s 112, not 911 –we have Community First Responder groups. In the rural areas, you have local people who’ve been trained in basic life support and community first response who have AEDs. They’ll have periods of the day where they come home from work as a teacher, a nurse, a policeman, or a fireman, and they turn on an app on their phone and say, “I’m available for the next 5 hours.” If there’s a cardiac arrest rung in within 5 miles of their community, they will drive directly there with the AED that they have. We’ve had numerous saves from that in the country because it could take 40 minutes to get an EMS vehicle there, and obviously, time is crucial in these scenarios. Our dispatchers will talk people through CPR, and then the community responders arrive with the AED. It has been a fantastic initiative.
Dr. Pepe: In many places, people have apps on their phones where they’re locked into the system, and it will go off and tell them there is something nearby and even GPS them into it, and it’s been fantastic.
The two points I want to make to responding to what we just heard Dean Malloy say is one, we always have a designated spot to have these in various places. If I’m at City Hall, we always have them near the red elevators on every floor and down at security. In all the public high schools, we always have one right below the clock where everybody can see it. We set it up in a very standardized form that anybody and everybody will know where it is at the time an event happens.
The other point he made about having the response teams is fantastic. I live in a large high rise and there are two complexes with many people here, and many are older, so there’s going to be a higher risk for having an event. In fact, we’ve just had one recently. The concept we developed here was a community emergency response team, where we sometimes have doctors, nurses, and paramedics who live here be on call and be responsible, or you could try to find an AED. More importantly, we made sure everybody here knew where they were and where to get them. We’ve got most of the people trained, and we’re doing more training in what actions to take during these periods of time when such events happen.
Dr. Glatter: Yes, it’s critical. I wanted to point out that we’ve looked at the use of drones, especially here in the United States. There have been some pilot studies looking at their utility in the setting of out-of-hospital cardiac arrest. I want to get both of your thoughts on this and the feasibility of this.
Dr. Molloy: In a rural area, it’s a fantastic idea. You’re going to get something there as the crow flies very quickly. You probably have to look at exactly in, say, a rural area like Ireland of 32,000 square kilometers, how many you›ll have to put, what kind of distances they can realistically cover, and make sure the batteries are charged. Certainly, that’s a very good initiative because with the AEDs, you can’t do anything wrong. You can’t give a shock unless a shock needs to be given. The machine directs you what to do, so somebody who has had no training can pick one of these out of the box and start to work with it quickly and confidently that they can’t do anything wrong.
It’s a great idea. It would be a little expensive potentially at the moment in getting the drones and having that volume of drones around. In the U.S., you have completely different air traffic than we have, and in cities, you have more helicopters flying around. We certainly wouldn’t have that in our cities because that could cause a challenge if you’ve got drones flying around as well. It’s about making it safe that nothing else can go wrong from a drone in somebody else’s flight path.
Dr. Pepe: In my experience, the earlier the intervention, the better the results. There is a limit here in terms of the drones if they just can’t get there soon enough. Having said that, we are so fortunate in the city of Seattle to have most citizens knowing CPR, and we’d get that person resuscitated because they were doing such a good job with the CPR up front.
That’s why you’re going to see the Buffalo Bills player survive neurologically intact – because he did get immediate treatment right then and there. In the future, we may even have some better devices that will actually even restore normal blood flow right then and there while you’re still in cardiac arrest. There are limitations in every case. But on the other hand, it’s exciting and it paid off in this case recently.
Dr. Molloy: Just a point of interest coming from this small little country over here. The first portable defibrillator was developed in Belfast, Ireland, in the back of a cardiac response car. Despite us being a tiny little country, we do have some advances ahead of the United States.
Dr. Pepe: That was a breakthrough. Dr. Frank Pantridge and John Geddes did this great work and that caught the imagination of everybody here. At first, they were just going out to give people oxygen and sedate them for their chest pain. It turned out that their defibrillators are what made the difference as they went out there. Absolutely, I have to acknowledge the folks in Ireland for giving us this. Many of the EMS systems got started because of the article they published in The Lancet back in 1967.
Dr. Glatter: I wanted to briefly talk about screening of the athletes at the high school/college level, but also at the professional level. Obviously, there are issues, including the risk for false-positives in terms of low incidence, but there are also false negatives, as the case with Christian Eriksen, who had a cardiac arrest in 2021 and who has been through extensive testing. We can debate the validity of such testing, but I wanted to get both of your takes on the utility of screening in such a population.
Dr. Molloy: That’s a very emotive subject. False-positives are difficult because you’re now saying to somebody that they can’t compete in your sport at a decent level. The difficult part is telling somebody that this is the end of their career.
The false-negative is a little bit more difficult. I don’t know Christian Eriksen and I’m not involved in his team in any way, but that is a one-point examination, and you’re dependent on the scale of the process interpreting the ECG, which is again only a couple of seconds and that particular arrhythmia may not have shown up on that.
Also, athletes, by nature of what they’re doing, are operating at 99% of efficiency on a frequent basis. They are at the peak of their physiologic fitness, and it does make them a little bit more prone to picking up viral illnesses from time to time. They may get a small viral myopericarditis, which causes a new arrhythmia that nobody knew about. They had the screening 2 or 3 years ago, and they now developed a new problem because of what they do, which just may not show up.
I was actually surprised that the gentleman came through it very well, which is fantastic. He wasn’t allowed to play football in the country where he was employed, and he has now moved to another country and is playing football with a defibrillator inserted. I don’t know what the rules are in American football where you can play with implantable defibrillators. I’m not so sure it’s a great idea to do that.
Dr. Pepe: One thing that we should bring up is that there are athletes with underlying cardiomyopathies or hypertrophies and things like that, but that was unlikely in this case. It’s possible, but it’s unlikely, because it would have manifested itself before. In terms of screening, I’ve met some very smart medical doctors who have run those tests, and they have been very encouraged even at the high school levels to have screenings done, whether it’s electrocardiography, echocardiography, and so on. I have to reiterate what Dr Malloy just said in that it may have its downsides as well. If you can pick up real obvious cases, I think that may be of value.
Dr. Glatter: I want to conclude and get some pearls and takeaways from each of you regarding the events that transpired and what our audience can really hold onto.
Dr. Molloy: Look at Formula One in the past 50 years. In Formula One, in the beginning it was a 2-minute job to change a tire. Now, they have this down where they’re measuring in fractions of a second and criticizing each other if one guy is 2.6 seconds and the other guy is 2.9 seconds. For me, that’s phenomenal. It takes me 25 minutes to change a tire.
We’ve looked at that from a resuscitation perspective, and we now do pit crew resuscitation before our events. We’ve planned our team and know who’s going to be occupying what role. After the events at the UEFA championships, we had a new rule brought in by UEFA where they handed me a new document saying, “This is what we would like you to do for resuscitation.” It was a three-man triangle, and I said, “No, we’re not going to do that here.” And they said, “Why, you have to; it’s our rule.”
I said, “No, our rule in Ireland is we have a six-person triangle. We’re not downing our standards because of what you have internationally. You’re covering games in some very low-resource environments, I know that. We have a particular standard here that we’re sticking to. We have a six-person group. We know what we’re all doing; we come very quickly to those downed players and get involved and we’ve had good outcomes, so we’re not going to change the standards.”
That’s the thing: You need to practice these things. The players don’t go out on the weekend and do a move for the very first time without practicing it hundreds of times. We need to look at it the same way as the medical team who are looking after that group of players and the crowd because we also look after the crowd.
A particular challenge in some of our stadiums is that the upper decks are so steep, and it’s very hard to get a patient onto a trolley and do CPR as you’re bringing them down to a zone to get them flat. We’ve had to come up with some innovative techniques to try and do that and accommodate that using some of the mechanical CPR devices. That’s the result you’ll only get from having practiced these events and trying to extricate patients. We want to check response times, so you have to practice your response team activity very frequently.
Dr. Pepe: There are two points made by Mick that I want to react to. One, the pit crew approach is critical in so many ways. We do the same thing in what we call the medical first attack, where we knew who the A, B, and C person would be. When we took it out to the NBA trainers, I recommended for them to have a similar approach so that if an event does happen right in the middle of prime time, they are coordinated.
The second point is that we do mass-gathering medicine. It’s not just the sportspeople on the field or the entertainers that we’re looking after; it is the people in the stands. We will see a cardiac arrest once a month. If you think about it, you might see a cardiac arrest occur in any community on a regular basis. Now you’ve got 100,000 people in one stadium, and something is bound to go wrong over those 3 or 4 hours where they are there and may have a critical emergency. Preparation for all of that is really important as well.
The final point is that on a day-to-day basis, most cardiac arrests do occur in the home. Granted, 80% of them are nonshockable cases, but the people who are more apt to survive are going to be the ones who have an electrical event. In fact, when we looked at our data years ago, we found that, of the cases of people with ventricular fibrillation that we resuscitated, half didn’t even have heart damage. Their enzymes were normal. It was a pure electrical event, and they were more resuscitable. They may have an underlying problem, but we can fix that once we get them back.
Everybody needs to know how to do bystander CPR, and second, we must make sure we have AEDs strategically placed, as I alluded to before. We also go out to other parts of the community and give them advice. All those things must be put in place, but more importantly, just get the training and make the training simple. It’s really a “just do it” philosophy, but make it simple.
For example, when I teach a course, I can do it in 15 minutes, and people retain it because I keep reiterating things like, “Okay, there’s one thing you need to know about choking: Pop the cork.” You give them a physiologic image of what’s happening. Everybody says, “I remember you saying to just do it, pop the cork.”
With AEDs, know where it is – that’s why we should have it in standardized places. Go get it, turn it on, and then follow the instructions. Also, the most important thing is making sure you’re doing quality compressions; and there are videos that can help you with that, as well as classes that you can take that will get you through it.
Dr. Glatter: Absolutely. The public still has the misconception that you need to do mouth-to-mouth resuscitation. The message has not permeated through society that you don’t need to do mouth-to-mouth. Hands-only CPR is the gold standard now.
Dr. Pepe: If people have a reversible cause like ventricular fibrillation, often they’re already gasping, which is better than a delivered breath, by the way. Most important, then, are the compressions to make sure you have oxygen going up to the brain, because you’re still theoretically loaded with oxygen in your bloodstream if you had a sudden cardiac arrest from a ventricular fibrillation.
Your points are well taken, and we found that we had better outcomes when we just gave instructions to do compressions only, and that became the standard. Mick, you’ve had some experiences with that as well.
Dr. Molloy: If we’re going to have a long-term benefit from all this, we have to start doing this in elementary school and teaching kids basic life support and some basic health messaging.
I remember trying to get this across to a teacher one day and the teacher saying, “But why would we teach young kids to resuscitate each other?” I said, “I think you forget that the only 60-year-old person in the room is you. You train them, and we train them. They’re the ones who are going to respond and keep you alive. That’s the way you should be looking at this.” That completely changed the mindset of whether we should be doing this for the kids or not.
Dr. Pepe: In fact, what we find is that that’s exactly who gets saved. I had case after case where the kids at the school had learned CPR and saved the teachers or the administrator at the high school or elementary school. It’s a fantastic point that you bring up, Dr. Malloy.
Dr. Glatter: One other brief thing we can interject here is that the team was excellent on field in that they evaluated Damar Hamlin in a primary survey sense of ABCs (i.e., airway, breathing, and circulation) for things like a tension pneumothorax. In the sense in which he was hit, there are reversible causes. Making sure he didn’t have a tension pneumothorax that caused the arrest, in my mind, was critical.
Dr. Pepe: We do the same thing on a day-to-day basis with a car wreck, because it could be that the person had ventricular fibrillation and then had the wreck. It’s not always trauma. That’s a fantastic point that you’re making. That’s exactly what I think happened, and that’s what we do.
Dr. Glatter: Well, thank you, gentlemen. This was an informative and helpful discussion for our audience. I appreciate your time and expertise.
Dr. Glatter, is an attending physician at Lenox Hill Hospital in New York City and assistant professor of emergency medicine at Zucker School of Medicine at Hofstra/Northwell in Hempstead, New York. He is an editorial adviser and hosts the Hot Topics in EM series on Medscape. He is also a medical contributor for Forbes.
Dr. Pepe is a professor of internal medicine, surgery, pediatrics, public health, and emergency medicine at University of Texas Health Science Center in Houston. He’s also a global coordinator of the U.S. Metropolitan Municipalities EMS Medical Directors (“Eagles”) Coalition.
Dr. Molloy works clinically as a consultant in emergency medicine in Wexford General Hospital, part of the Ireland East Hospital Group (IEHG). Internationally, he is a member of the Disaster Medicine Section of the European Society of Emergency Medicine (EUSEM) and has been appointed by the Irish Medical Organization (IMO) as one of two Irish delegates to serve on the European Board and Section of Emergency Medicine of the European Union of Medical Specialists (UEMS), having served for a number of years on its predecessor, the Multidisciplinary Joint Committee on Emergency Medicine.
A version of this article first appeared on Medscape.com.
This discussion was recorded on Jan. 9, 2023. This transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome. I’m Dr. Robert D. Glatter, medical adviser for Medscape Emergency Medicine. Today, we have Dr. Paul E. Pepe, an emergency medicine physician based in Florida and a highly recognized expert in emergency medical services (EMS), critical care, sports and event medicine, and resuscitation. Also joining us is Dr. Michael S. (“Mick”) Malloy, an emergency medicine physician based in Ireland, also an expert in prehospital care, resuscitation, and sports and event medicine. Welcome, gentlemen.
Dr. Pepe: Thanks for having us here.
Dr. Glatter: the Buffalo Bills safety who went down suffering a cardiac arrest in front of millions of people. Although we don’t know the exact cause of the events that transpired, the goal of our discussion is to guide our audience through a systematic approach to evaluation and management of an athlete suffering blunt force chest and neck trauma, and then suffering a cardiac arrest. We do know, obviously, that Damar was successfully resuscitated, thanks to the medical staff and trainers.
Almost 50 years ago, Chuck Hughes, a Detroit Lions receiver, went down and died with just a minute to go in the game and, unfortunately, didn’t survive.
Paul, can you tell me your impressions after viewing the replay of the events that evening? What were the most likely causes of this syncopal event and the subsequent cardiac arrest?
Dr. Pepe: We don’t know anything specifically. It’s being kept private about what the events were. It’s a little bit complicated in a sense that he basically had an extended resuscitation in the hospital. My experience has been that most people that have ventricular fibrillation, from whatever cause, will most likely be waking up on the field if you get to them. I’ve had personal experience with that.
More importantly than when it starts, when someone goes down on the field, both Dr. Malloy and I take a broader view. We don’t get tunnel vision and think, “Oh, it was a traumatic event,” or “It was cardiac event,” and we just have our minds open. There are many things that could make you stop breathing on the field. It could be a neck or a severe head injury, and then any kind of other internal injury that occurs.
When I saw in the video that Damar Hamlin stood up, that made it a less likely to be a spinal injury. He seemed to be physically functioning, and then he suddenly collapsed. That went along with something that looks like a ventricular fibrillation or ventricular tachycardia type of event and made me think right away that it was commotio cordis. I’m not a Latin scholar, but commotio is like commotion. A literal translation might be an agitation of the heart. I was thinking that he probably got hit somewhere in the middle of the chest at the right moment where the heart is resetting in that repolarization phase, like an R-on-T phenomenon, and then caused this sudden ventricular dysrhythmia.
Most people associate it to that because we have a couple of dozen cases a year of people getting hockey pucks or a baseball hitting their chest, which is very common with adolescents. On the other hand, you can’t get it from a blunt injury like this, and it was too early for it to be, say, a direct cardiac contusion, unless there was a direct injury there. It just happened so quickly.
In Europe, they’ve had a large amount of experience with this same kind of problem before, even just from a direct shoulder hit, for example. Mick Malloy is the dean of the faculty of sports and exercise medicine at the Royal College of Surgeons in Ireland and has vast experience, and now he is the person overseeing the procedures for this. Mick, have you had those kinds of experiences as well?
Dr. Molloy: Yes. It’s something that has occurred over recent decades and has been more recognized. I note that in professional sports, it’s a very different thing because you’ve got such huge teams and teams trained to respond very quickly. And that’s the most important thing in this scenario – having a team that is well functioning as a high-class emergency response team ready to get out on to that field very quickly after the person collapses, getting the automated external defibrillator (AED) on, and then recognizing whether there needs to be a shock given or not. The machine will tell you all that.
In our scenario, we run courses called CARES (Care of the Athlete Resuscitation and Emergencies in Sport) to make sure that our team physicians and team physiotherapists and trainers are all speaking as one when an emergency arises.
I don’t worry so much about the professional sport. It’s more with the amateur sports and the kids sports that I get a bit more concerned because there isn’t the same level of medical care there. Having everybody trained in basic life support would be very important to reduce unnecessary deaths from these types of conditions.
As Paul mentioned, there is a very specific cardiac cause in some of these circumstances, where you get hit just at the wrong time and that hit occurs at a particular electrical point in time. It causes this ventricular fibrillation, and the only real treatment there is the defibrillator as quickly as possible.
Dr. Glatter: What you’re saying ultimately is an important part about rapid defibrillation, and at first, cardiopulmonary resuscitation (CPR). People are concerned about whether they should begin CPR. We’re talking about out-of-hospital cardiac arrest that is outside of a football stadium, for example. Some people are obsessed with taking a person’s pulse, and that’s been a point of contention. If someone is unconscious and not breathing, we should start CPR. Wouldn›t you agree? They will wake up quickly if you begin chest compressions if they’re not necessary.
Dr. Pepe: I tell people, just do it. You’re right, people will wake up and feel it if they don’t need it.
Getting back to Mick’s point of having things ready to go, for example, 8 years ago, we had a professional player on the bench who suddenly collapsed right there in front of the entire audience. We immediately did CPR, and we got the AED on. We shocked him and he was ready, willing, and able to get back on the bench again. It turns out he had underlying coronary artery disease, but we got him back right away.
I did an initial study where we placed an AED in a public place at the Chicago O’Hare Airport to see if the public would use these. Most cardiac arrests occur at home, of course, but in public places, that was a good place to try it. We had almost 10 cases the first year. What was fascinating was that we had almost no survivors over the previous decade, even though there were paramedics at the airport. When we put these out there, we had nine people go down that first year, and six people who had never operated an AED or seen one before knew to get one and use it. Every one of those people survived neurologically intact, and almost every person was waking up before traditional responders got there. That’s how effective this is, but you need to know where the AED is.
Dr. Glatter: How to turn it on, where it is, and how to operate it.
Dr. Pepe: That was the point: These rescuers saved lives in the first year, and it was tremendous. Two points I make about it are that one, you need to know where it is, and two, just go turn it on. It gives you the instructions to follow through; just be in the Nike mode, because it basically won’t hurt a person. It’s rare that there’s ever been any complication of that. The machine algorithms are so good.
Dr. Glatter: Mick, I want to turn to you about the European experience. Specifically in Denmark, we know that there’s a large public health initiative to have AEDs accessible. There have been studies showing that when the public is engaged, especially with studies looking at an app when access is available, survivability doubled in the past 10 years from having access to AEDs. What’s your experience in Ireland in terms of public access to defibrillators?
Dr. Molloy: We’ve got two different streams here. There was a big push to have more AEDs at all sports venues. That was great, but some of the sporting clubs put them inside the locked door. I said that there’s no point to that because nobody can access it. You need to have an external building and you need to leave it open. If somebody needs to use it, they need to know how to get it, open it, and get away, and not get in through a locked door to get access to a defibrillator. We have AEDs now in most stadiums and even in small rural areas, where you might have only 200 people turn up for a game.
From another public access side, if you dial in – in our scenario, it’s 112, not 911 –we have Community First Responder groups. In the rural areas, you have local people who’ve been trained in basic life support and community first response who have AEDs. They’ll have periods of the day where they come home from work as a teacher, a nurse, a policeman, or a fireman, and they turn on an app on their phone and say, “I’m available for the next 5 hours.” If there’s a cardiac arrest rung in within 5 miles of their community, they will drive directly there with the AED that they have. We’ve had numerous saves from that in the country because it could take 40 minutes to get an EMS vehicle there, and obviously, time is crucial in these scenarios. Our dispatchers will talk people through CPR, and then the community responders arrive with the AED. It has been a fantastic initiative.
Dr. Pepe: In many places, people have apps on their phones where they’re locked into the system, and it will go off and tell them there is something nearby and even GPS them into it, and it’s been fantastic.
The two points I want to make to responding to what we just heard Dean Malloy say is one, we always have a designated spot to have these in various places. If I’m at City Hall, we always have them near the red elevators on every floor and down at security. In all the public high schools, we always have one right below the clock where everybody can see it. We set it up in a very standardized form that anybody and everybody will know where it is at the time an event happens.
The other point he made about having the response teams is fantastic. I live in a large high rise and there are two complexes with many people here, and many are older, so there’s going to be a higher risk for having an event. In fact, we’ve just had one recently. The concept we developed here was a community emergency response team, where we sometimes have doctors, nurses, and paramedics who live here be on call and be responsible, or you could try to find an AED. More importantly, we made sure everybody here knew where they were and where to get them. We’ve got most of the people trained, and we’re doing more training in what actions to take during these periods of time when such events happen.
Dr. Glatter: Yes, it’s critical. I wanted to point out that we’ve looked at the use of drones, especially here in the United States. There have been some pilot studies looking at their utility in the setting of out-of-hospital cardiac arrest. I want to get both of your thoughts on this and the feasibility of this.
Dr. Molloy: In a rural area, it’s a fantastic idea. You’re going to get something there as the crow flies very quickly. You probably have to look at exactly in, say, a rural area like Ireland of 32,000 square kilometers, how many you›ll have to put, what kind of distances they can realistically cover, and make sure the batteries are charged. Certainly, that’s a very good initiative because with the AEDs, you can’t do anything wrong. You can’t give a shock unless a shock needs to be given. The machine directs you what to do, so somebody who has had no training can pick one of these out of the box and start to work with it quickly and confidently that they can’t do anything wrong.
It’s a great idea. It would be a little expensive potentially at the moment in getting the drones and having that volume of drones around. In the U.S., you have completely different air traffic than we have, and in cities, you have more helicopters flying around. We certainly wouldn’t have that in our cities because that could cause a challenge if you’ve got drones flying around as well. It’s about making it safe that nothing else can go wrong from a drone in somebody else’s flight path.
Dr. Pepe: In my experience, the earlier the intervention, the better the results. There is a limit here in terms of the drones if they just can’t get there soon enough. Having said that, we are so fortunate in the city of Seattle to have most citizens knowing CPR, and we’d get that person resuscitated because they were doing such a good job with the CPR up front.
That’s why you’re going to see the Buffalo Bills player survive neurologically intact – because he did get immediate treatment right then and there. In the future, we may even have some better devices that will actually even restore normal blood flow right then and there while you’re still in cardiac arrest. There are limitations in every case. But on the other hand, it’s exciting and it paid off in this case recently.
Dr. Molloy: Just a point of interest coming from this small little country over here. The first portable defibrillator was developed in Belfast, Ireland, in the back of a cardiac response car. Despite us being a tiny little country, we do have some advances ahead of the United States.
Dr. Pepe: That was a breakthrough. Dr. Frank Pantridge and John Geddes did this great work and that caught the imagination of everybody here. At first, they were just going out to give people oxygen and sedate them for their chest pain. It turned out that their defibrillators are what made the difference as they went out there. Absolutely, I have to acknowledge the folks in Ireland for giving us this. Many of the EMS systems got started because of the article they published in The Lancet back in 1967.
Dr. Glatter: I wanted to briefly talk about screening of the athletes at the high school/college level, but also at the professional level. Obviously, there are issues, including the risk for false-positives in terms of low incidence, but there are also false negatives, as the case with Christian Eriksen, who had a cardiac arrest in 2021 and who has been through extensive testing. We can debate the validity of such testing, but I wanted to get both of your takes on the utility of screening in such a population.
Dr. Molloy: That’s a very emotive subject. False-positives are difficult because you’re now saying to somebody that they can’t compete in your sport at a decent level. The difficult part is telling somebody that this is the end of their career.
The false-negative is a little bit more difficult. I don’t know Christian Eriksen and I’m not involved in his team in any way, but that is a one-point examination, and you’re dependent on the scale of the process interpreting the ECG, which is again only a couple of seconds and that particular arrhythmia may not have shown up on that.
Also, athletes, by nature of what they’re doing, are operating at 99% of efficiency on a frequent basis. They are at the peak of their physiologic fitness, and it does make them a little bit more prone to picking up viral illnesses from time to time. They may get a small viral myopericarditis, which causes a new arrhythmia that nobody knew about. They had the screening 2 or 3 years ago, and they now developed a new problem because of what they do, which just may not show up.
I was actually surprised that the gentleman came through it very well, which is fantastic. He wasn’t allowed to play football in the country where he was employed, and he has now moved to another country and is playing football with a defibrillator inserted. I don’t know what the rules are in American football where you can play with implantable defibrillators. I’m not so sure it’s a great idea to do that.
Dr. Pepe: One thing that we should bring up is that there are athletes with underlying cardiomyopathies or hypertrophies and things like that, but that was unlikely in this case. It’s possible, but it’s unlikely, because it would have manifested itself before. In terms of screening, I’ve met some very smart medical doctors who have run those tests, and they have been very encouraged even at the high school levels to have screenings done, whether it’s electrocardiography, echocardiography, and so on. I have to reiterate what Dr Malloy just said in that it may have its downsides as well. If you can pick up real obvious cases, I think that may be of value.
Dr. Glatter: I want to conclude and get some pearls and takeaways from each of you regarding the events that transpired and what our audience can really hold onto.
Dr. Molloy: Look at Formula One in the past 50 years. In Formula One, in the beginning it was a 2-minute job to change a tire. Now, they have this down where they’re measuring in fractions of a second and criticizing each other if one guy is 2.6 seconds and the other guy is 2.9 seconds. For me, that’s phenomenal. It takes me 25 minutes to change a tire.
We’ve looked at that from a resuscitation perspective, and we now do pit crew resuscitation before our events. We’ve planned our team and know who’s going to be occupying what role. After the events at the UEFA championships, we had a new rule brought in by UEFA where they handed me a new document saying, “This is what we would like you to do for resuscitation.” It was a three-man triangle, and I said, “No, we’re not going to do that here.” And they said, “Why, you have to; it’s our rule.”
I said, “No, our rule in Ireland is we have a six-person triangle. We’re not downing our standards because of what you have internationally. You’re covering games in some very low-resource environments, I know that. We have a particular standard here that we’re sticking to. We have a six-person group. We know what we’re all doing; we come very quickly to those downed players and get involved and we’ve had good outcomes, so we’re not going to change the standards.”
That’s the thing: You need to practice these things. The players don’t go out on the weekend and do a move for the very first time without practicing it hundreds of times. We need to look at it the same way as the medical team who are looking after that group of players and the crowd because we also look after the crowd.
A particular challenge in some of our stadiums is that the upper decks are so steep, and it’s very hard to get a patient onto a trolley and do CPR as you’re bringing them down to a zone to get them flat. We’ve had to come up with some innovative techniques to try and do that and accommodate that using some of the mechanical CPR devices. That’s the result you’ll only get from having practiced these events and trying to extricate patients. We want to check response times, so you have to practice your response team activity very frequently.
Dr. Pepe: There are two points made by Mick that I want to react to. One, the pit crew approach is critical in so many ways. We do the same thing in what we call the medical first attack, where we knew who the A, B, and C person would be. When we took it out to the NBA trainers, I recommended for them to have a similar approach so that if an event does happen right in the middle of prime time, they are coordinated.
The second point is that we do mass-gathering medicine. It’s not just the sportspeople on the field or the entertainers that we’re looking after; it is the people in the stands. We will see a cardiac arrest once a month. If you think about it, you might see a cardiac arrest occur in any community on a regular basis. Now you’ve got 100,000 people in one stadium, and something is bound to go wrong over those 3 or 4 hours where they are there and may have a critical emergency. Preparation for all of that is really important as well.
The final point is that on a day-to-day basis, most cardiac arrests do occur in the home. Granted, 80% of them are nonshockable cases, but the people who are more apt to survive are going to be the ones who have an electrical event. In fact, when we looked at our data years ago, we found that, of the cases of people with ventricular fibrillation that we resuscitated, half didn’t even have heart damage. Their enzymes were normal. It was a pure electrical event, and they were more resuscitable. They may have an underlying problem, but we can fix that once we get them back.
Everybody needs to know how to do bystander CPR, and second, we must make sure we have AEDs strategically placed, as I alluded to before. We also go out to other parts of the community and give them advice. All those things must be put in place, but more importantly, just get the training and make the training simple. It’s really a “just do it” philosophy, but make it simple.
For example, when I teach a course, I can do it in 15 minutes, and people retain it because I keep reiterating things like, “Okay, there’s one thing you need to know about choking: Pop the cork.” You give them a physiologic image of what’s happening. Everybody says, “I remember you saying to just do it, pop the cork.”
With AEDs, know where it is – that’s why we should have it in standardized places. Go get it, turn it on, and then follow the instructions. Also, the most important thing is making sure you’re doing quality compressions; and there are videos that can help you with that, as well as classes that you can take that will get you through it.
Dr. Glatter: Absolutely. The public still has the misconception that you need to do mouth-to-mouth resuscitation. The message has not permeated through society that you don’t need to do mouth-to-mouth. Hands-only CPR is the gold standard now.
Dr. Pepe: If people have a reversible cause like ventricular fibrillation, often they’re already gasping, which is better than a delivered breath, by the way. Most important, then, are the compressions to make sure you have oxygen going up to the brain, because you’re still theoretically loaded with oxygen in your bloodstream if you had a sudden cardiac arrest from a ventricular fibrillation.
Your points are well taken, and we found that we had better outcomes when we just gave instructions to do compressions only, and that became the standard. Mick, you’ve had some experiences with that as well.
Dr. Molloy: If we’re going to have a long-term benefit from all this, we have to start doing this in elementary school and teaching kids basic life support and some basic health messaging.
I remember trying to get this across to a teacher one day and the teacher saying, “But why would we teach young kids to resuscitate each other?” I said, “I think you forget that the only 60-year-old person in the room is you. You train them, and we train them. They’re the ones who are going to respond and keep you alive. That’s the way you should be looking at this.” That completely changed the mindset of whether we should be doing this for the kids or not.
Dr. Pepe: In fact, what we find is that that’s exactly who gets saved. I had case after case where the kids at the school had learned CPR and saved the teachers or the administrator at the high school or elementary school. It’s a fantastic point that you bring up, Dr. Malloy.
Dr. Glatter: One other brief thing we can interject here is that the team was excellent on field in that they evaluated Damar Hamlin in a primary survey sense of ABCs (i.e., airway, breathing, and circulation) for things like a tension pneumothorax. In the sense in which he was hit, there are reversible causes. Making sure he didn’t have a tension pneumothorax that caused the arrest, in my mind, was critical.
Dr. Pepe: We do the same thing on a day-to-day basis with a car wreck, because it could be that the person had ventricular fibrillation and then had the wreck. It’s not always trauma. That’s a fantastic point that you’re making. That’s exactly what I think happened, and that’s what we do.
Dr. Glatter: Well, thank you, gentlemen. This was an informative and helpful discussion for our audience. I appreciate your time and expertise.
Dr. Glatter, is an attending physician at Lenox Hill Hospital in New York City and assistant professor of emergency medicine at Zucker School of Medicine at Hofstra/Northwell in Hempstead, New York. He is an editorial adviser and hosts the Hot Topics in EM series on Medscape. He is also a medical contributor for Forbes.
Dr. Pepe is a professor of internal medicine, surgery, pediatrics, public health, and emergency medicine at University of Texas Health Science Center in Houston. He’s also a global coordinator of the U.S. Metropolitan Municipalities EMS Medical Directors (“Eagles”) Coalition.
Dr. Molloy works clinically as a consultant in emergency medicine in Wexford General Hospital, part of the Ireland East Hospital Group (IEHG). Internationally, he is a member of the Disaster Medicine Section of the European Society of Emergency Medicine (EUSEM) and has been appointed by the Irish Medical Organization (IMO) as one of two Irish delegates to serve on the European Board and Section of Emergency Medicine of the European Union of Medical Specialists (UEMS), having served for a number of years on its predecessor, the Multidisciplinary Joint Committee on Emergency Medicine.
A version of this article first appeared on Medscape.com.
This discussion was recorded on Jan. 9, 2023. This transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome. I’m Dr. Robert D. Glatter, medical adviser for Medscape Emergency Medicine. Today, we have Dr. Paul E. Pepe, an emergency medicine physician based in Florida and a highly recognized expert in emergency medical services (EMS), critical care, sports and event medicine, and resuscitation. Also joining us is Dr. Michael S. (“Mick”) Malloy, an emergency medicine physician based in Ireland, also an expert in prehospital care, resuscitation, and sports and event medicine. Welcome, gentlemen.
Dr. Pepe: Thanks for having us here.
Dr. Glatter: the Buffalo Bills safety who went down suffering a cardiac arrest in front of millions of people. Although we don’t know the exact cause of the events that transpired, the goal of our discussion is to guide our audience through a systematic approach to evaluation and management of an athlete suffering blunt force chest and neck trauma, and then suffering a cardiac arrest. We do know, obviously, that Damar was successfully resuscitated, thanks to the medical staff and trainers.
Almost 50 years ago, Chuck Hughes, a Detroit Lions receiver, went down and died with just a minute to go in the game and, unfortunately, didn’t survive.
Paul, can you tell me your impressions after viewing the replay of the events that evening? What were the most likely causes of this syncopal event and the subsequent cardiac arrest?
Dr. Pepe: We don’t know anything specifically. It’s being kept private about what the events were. It’s a little bit complicated in a sense that he basically had an extended resuscitation in the hospital. My experience has been that most people that have ventricular fibrillation, from whatever cause, will most likely be waking up on the field if you get to them. I’ve had personal experience with that.
More importantly than when it starts, when someone goes down on the field, both Dr. Malloy and I take a broader view. We don’t get tunnel vision and think, “Oh, it was a traumatic event,” or “It was cardiac event,” and we just have our minds open. There are many things that could make you stop breathing on the field. It could be a neck or a severe head injury, and then any kind of other internal injury that occurs.
When I saw in the video that Damar Hamlin stood up, that made it a less likely to be a spinal injury. He seemed to be physically functioning, and then he suddenly collapsed. That went along with something that looks like a ventricular fibrillation or ventricular tachycardia type of event and made me think right away that it was commotio cordis. I’m not a Latin scholar, but commotio is like commotion. A literal translation might be an agitation of the heart. I was thinking that he probably got hit somewhere in the middle of the chest at the right moment where the heart is resetting in that repolarization phase, like an R-on-T phenomenon, and then caused this sudden ventricular dysrhythmia.
Most people associate it to that because we have a couple of dozen cases a year of people getting hockey pucks or a baseball hitting their chest, which is very common with adolescents. On the other hand, you can’t get it from a blunt injury like this, and it was too early for it to be, say, a direct cardiac contusion, unless there was a direct injury there. It just happened so quickly.
In Europe, they’ve had a large amount of experience with this same kind of problem before, even just from a direct shoulder hit, for example. Mick Malloy is the dean of the faculty of sports and exercise medicine at the Royal College of Surgeons in Ireland and has vast experience, and now he is the person overseeing the procedures for this. Mick, have you had those kinds of experiences as well?
Dr. Molloy: Yes. It’s something that has occurred over recent decades and has been more recognized. I note that in professional sports, it’s a very different thing because you’ve got such huge teams and teams trained to respond very quickly. And that’s the most important thing in this scenario – having a team that is well functioning as a high-class emergency response team ready to get out on to that field very quickly after the person collapses, getting the automated external defibrillator (AED) on, and then recognizing whether there needs to be a shock given or not. The machine will tell you all that.
In our scenario, we run courses called CARES (Care of the Athlete Resuscitation and Emergencies in Sport) to make sure that our team physicians and team physiotherapists and trainers are all speaking as one when an emergency arises.
I don’t worry so much about the professional sport. It’s more with the amateur sports and the kids sports that I get a bit more concerned because there isn’t the same level of medical care there. Having everybody trained in basic life support would be very important to reduce unnecessary deaths from these types of conditions.
As Paul mentioned, there is a very specific cardiac cause in some of these circumstances, where you get hit just at the wrong time and that hit occurs at a particular electrical point in time. It causes this ventricular fibrillation, and the only real treatment there is the defibrillator as quickly as possible.
Dr. Glatter: What you’re saying ultimately is an important part about rapid defibrillation, and at first, cardiopulmonary resuscitation (CPR). People are concerned about whether they should begin CPR. We’re talking about out-of-hospital cardiac arrest that is outside of a football stadium, for example. Some people are obsessed with taking a person’s pulse, and that’s been a point of contention. If someone is unconscious and not breathing, we should start CPR. Wouldn›t you agree? They will wake up quickly if you begin chest compressions if they’re not necessary.
Dr. Pepe: I tell people, just do it. You’re right, people will wake up and feel it if they don’t need it.
Getting back to Mick’s point of having things ready to go, for example, 8 years ago, we had a professional player on the bench who suddenly collapsed right there in front of the entire audience. We immediately did CPR, and we got the AED on. We shocked him and he was ready, willing, and able to get back on the bench again. It turns out he had underlying coronary artery disease, but we got him back right away.
I did an initial study where we placed an AED in a public place at the Chicago O’Hare Airport to see if the public would use these. Most cardiac arrests occur at home, of course, but in public places, that was a good place to try it. We had almost 10 cases the first year. What was fascinating was that we had almost no survivors over the previous decade, even though there were paramedics at the airport. When we put these out there, we had nine people go down that first year, and six people who had never operated an AED or seen one before knew to get one and use it. Every one of those people survived neurologically intact, and almost every person was waking up before traditional responders got there. That’s how effective this is, but you need to know where the AED is.
Dr. Glatter: How to turn it on, where it is, and how to operate it.
Dr. Pepe: That was the point: These rescuers saved lives in the first year, and it was tremendous. Two points I make about it are that one, you need to know where it is, and two, just go turn it on. It gives you the instructions to follow through; just be in the Nike mode, because it basically won’t hurt a person. It’s rare that there’s ever been any complication of that. The machine algorithms are so good.
Dr. Glatter: Mick, I want to turn to you about the European experience. Specifically in Denmark, we know that there’s a large public health initiative to have AEDs accessible. There have been studies showing that when the public is engaged, especially with studies looking at an app when access is available, survivability doubled in the past 10 years from having access to AEDs. What’s your experience in Ireland in terms of public access to defibrillators?
Dr. Molloy: We’ve got two different streams here. There was a big push to have more AEDs at all sports venues. That was great, but some of the sporting clubs put them inside the locked door. I said that there’s no point to that because nobody can access it. You need to have an external building and you need to leave it open. If somebody needs to use it, they need to know how to get it, open it, and get away, and not get in through a locked door to get access to a defibrillator. We have AEDs now in most stadiums and even in small rural areas, where you might have only 200 people turn up for a game.
From another public access side, if you dial in – in our scenario, it’s 112, not 911 –we have Community First Responder groups. In the rural areas, you have local people who’ve been trained in basic life support and community first response who have AEDs. They’ll have periods of the day where they come home from work as a teacher, a nurse, a policeman, or a fireman, and they turn on an app on their phone and say, “I’m available for the next 5 hours.” If there’s a cardiac arrest rung in within 5 miles of their community, they will drive directly there with the AED that they have. We’ve had numerous saves from that in the country because it could take 40 minutes to get an EMS vehicle there, and obviously, time is crucial in these scenarios. Our dispatchers will talk people through CPR, and then the community responders arrive with the AED. It has been a fantastic initiative.
Dr. Pepe: In many places, people have apps on their phones where they’re locked into the system, and it will go off and tell them there is something nearby and even GPS them into it, and it’s been fantastic.
The two points I want to make to responding to what we just heard Dean Malloy say is one, we always have a designated spot to have these in various places. If I’m at City Hall, we always have them near the red elevators on every floor and down at security. In all the public high schools, we always have one right below the clock where everybody can see it. We set it up in a very standardized form that anybody and everybody will know where it is at the time an event happens.
The other point he made about having the response teams is fantastic. I live in a large high rise and there are two complexes with many people here, and many are older, so there’s going to be a higher risk for having an event. In fact, we’ve just had one recently. The concept we developed here was a community emergency response team, where we sometimes have doctors, nurses, and paramedics who live here be on call and be responsible, or you could try to find an AED. More importantly, we made sure everybody here knew where they were and where to get them. We’ve got most of the people trained, and we’re doing more training in what actions to take during these periods of time when such events happen.
Dr. Glatter: Yes, it’s critical. I wanted to point out that we’ve looked at the use of drones, especially here in the United States. There have been some pilot studies looking at their utility in the setting of out-of-hospital cardiac arrest. I want to get both of your thoughts on this and the feasibility of this.
Dr. Molloy: In a rural area, it’s a fantastic idea. You’re going to get something there as the crow flies very quickly. You probably have to look at exactly in, say, a rural area like Ireland of 32,000 square kilometers, how many you›ll have to put, what kind of distances they can realistically cover, and make sure the batteries are charged. Certainly, that’s a very good initiative because with the AEDs, you can’t do anything wrong. You can’t give a shock unless a shock needs to be given. The machine directs you what to do, so somebody who has had no training can pick one of these out of the box and start to work with it quickly and confidently that they can’t do anything wrong.
It’s a great idea. It would be a little expensive potentially at the moment in getting the drones and having that volume of drones around. In the U.S., you have completely different air traffic than we have, and in cities, you have more helicopters flying around. We certainly wouldn’t have that in our cities because that could cause a challenge if you’ve got drones flying around as well. It’s about making it safe that nothing else can go wrong from a drone in somebody else’s flight path.
Dr. Pepe: In my experience, the earlier the intervention, the better the results. There is a limit here in terms of the drones if they just can’t get there soon enough. Having said that, we are so fortunate in the city of Seattle to have most citizens knowing CPR, and we’d get that person resuscitated because they were doing such a good job with the CPR up front.
That’s why you’re going to see the Buffalo Bills player survive neurologically intact – because he did get immediate treatment right then and there. In the future, we may even have some better devices that will actually even restore normal blood flow right then and there while you’re still in cardiac arrest. There are limitations in every case. But on the other hand, it’s exciting and it paid off in this case recently.
Dr. Molloy: Just a point of interest coming from this small little country over here. The first portable defibrillator was developed in Belfast, Ireland, in the back of a cardiac response car. Despite us being a tiny little country, we do have some advances ahead of the United States.
Dr. Pepe: That was a breakthrough. Dr. Frank Pantridge and John Geddes did this great work and that caught the imagination of everybody here. At first, they were just going out to give people oxygen and sedate them for their chest pain. It turned out that their defibrillators are what made the difference as they went out there. Absolutely, I have to acknowledge the folks in Ireland for giving us this. Many of the EMS systems got started because of the article they published in The Lancet back in 1967.
Dr. Glatter: I wanted to briefly talk about screening of the athletes at the high school/college level, but also at the professional level. Obviously, there are issues, including the risk for false-positives in terms of low incidence, but there are also false negatives, as the case with Christian Eriksen, who had a cardiac arrest in 2021 and who has been through extensive testing. We can debate the validity of such testing, but I wanted to get both of your takes on the utility of screening in such a population.
Dr. Molloy: That’s a very emotive subject. False-positives are difficult because you’re now saying to somebody that they can’t compete in your sport at a decent level. The difficult part is telling somebody that this is the end of their career.
The false-negative is a little bit more difficult. I don’t know Christian Eriksen and I’m not involved in his team in any way, but that is a one-point examination, and you’re dependent on the scale of the process interpreting the ECG, which is again only a couple of seconds and that particular arrhythmia may not have shown up on that.
Also, athletes, by nature of what they’re doing, are operating at 99% of efficiency on a frequent basis. They are at the peak of their physiologic fitness, and it does make them a little bit more prone to picking up viral illnesses from time to time. They may get a small viral myopericarditis, which causes a new arrhythmia that nobody knew about. They had the screening 2 or 3 years ago, and they now developed a new problem because of what they do, which just may not show up.
I was actually surprised that the gentleman came through it very well, which is fantastic. He wasn’t allowed to play football in the country where he was employed, and he has now moved to another country and is playing football with a defibrillator inserted. I don’t know what the rules are in American football where you can play with implantable defibrillators. I’m not so sure it’s a great idea to do that.
Dr. Pepe: One thing that we should bring up is that there are athletes with underlying cardiomyopathies or hypertrophies and things like that, but that was unlikely in this case. It’s possible, but it’s unlikely, because it would have manifested itself before. In terms of screening, I’ve met some very smart medical doctors who have run those tests, and they have been very encouraged even at the high school levels to have screenings done, whether it’s electrocardiography, echocardiography, and so on. I have to reiterate what Dr Malloy just said in that it may have its downsides as well. If you can pick up real obvious cases, I think that may be of value.
Dr. Glatter: I want to conclude and get some pearls and takeaways from each of you regarding the events that transpired and what our audience can really hold onto.
Dr. Molloy: Look at Formula One in the past 50 years. In Formula One, in the beginning it was a 2-minute job to change a tire. Now, they have this down where they’re measuring in fractions of a second and criticizing each other if one guy is 2.6 seconds and the other guy is 2.9 seconds. For me, that’s phenomenal. It takes me 25 minutes to change a tire.
We’ve looked at that from a resuscitation perspective, and we now do pit crew resuscitation before our events. We’ve planned our team and know who’s going to be occupying what role. After the events at the UEFA championships, we had a new rule brought in by UEFA where they handed me a new document saying, “This is what we would like you to do for resuscitation.” It was a three-man triangle, and I said, “No, we’re not going to do that here.” And they said, “Why, you have to; it’s our rule.”
I said, “No, our rule in Ireland is we have a six-person triangle. We’re not downing our standards because of what you have internationally. You’re covering games in some very low-resource environments, I know that. We have a particular standard here that we’re sticking to. We have a six-person group. We know what we’re all doing; we come very quickly to those downed players and get involved and we’ve had good outcomes, so we’re not going to change the standards.”
That’s the thing: You need to practice these things. The players don’t go out on the weekend and do a move for the very first time without practicing it hundreds of times. We need to look at it the same way as the medical team who are looking after that group of players and the crowd because we also look after the crowd.
A particular challenge in some of our stadiums is that the upper decks are so steep, and it’s very hard to get a patient onto a trolley and do CPR as you’re bringing them down to a zone to get them flat. We’ve had to come up with some innovative techniques to try and do that and accommodate that using some of the mechanical CPR devices. That’s the result you’ll only get from having practiced these events and trying to extricate patients. We want to check response times, so you have to practice your response team activity very frequently.
Dr. Pepe: There are two points made by Mick that I want to react to. One, the pit crew approach is critical in so many ways. We do the same thing in what we call the medical first attack, where we knew who the A, B, and C person would be. When we took it out to the NBA trainers, I recommended for them to have a similar approach so that if an event does happen right in the middle of prime time, they are coordinated.
The second point is that we do mass-gathering medicine. It’s not just the sportspeople on the field or the entertainers that we’re looking after; it is the people in the stands. We will see a cardiac arrest once a month. If you think about it, you might see a cardiac arrest occur in any community on a regular basis. Now you’ve got 100,000 people in one stadium, and something is bound to go wrong over those 3 or 4 hours where they are there and may have a critical emergency. Preparation for all of that is really important as well.
The final point is that on a day-to-day basis, most cardiac arrests do occur in the home. Granted, 80% of them are nonshockable cases, but the people who are more apt to survive are going to be the ones who have an electrical event. In fact, when we looked at our data years ago, we found that, of the cases of people with ventricular fibrillation that we resuscitated, half didn’t even have heart damage. Their enzymes were normal. It was a pure electrical event, and they were more resuscitable. They may have an underlying problem, but we can fix that once we get them back.
Everybody needs to know how to do bystander CPR, and second, we must make sure we have AEDs strategically placed, as I alluded to before. We also go out to other parts of the community and give them advice. All those things must be put in place, but more importantly, just get the training and make the training simple. It’s really a “just do it” philosophy, but make it simple.
For example, when I teach a course, I can do it in 15 minutes, and people retain it because I keep reiterating things like, “Okay, there’s one thing you need to know about choking: Pop the cork.” You give them a physiologic image of what’s happening. Everybody says, “I remember you saying to just do it, pop the cork.”
With AEDs, know where it is – that’s why we should have it in standardized places. Go get it, turn it on, and then follow the instructions. Also, the most important thing is making sure you’re doing quality compressions; and there are videos that can help you with that, as well as classes that you can take that will get you through it.
Dr. Glatter: Absolutely. The public still has the misconception that you need to do mouth-to-mouth resuscitation. The message has not permeated through society that you don’t need to do mouth-to-mouth. Hands-only CPR is the gold standard now.
Dr. Pepe: If people have a reversible cause like ventricular fibrillation, often they’re already gasping, which is better than a delivered breath, by the way. Most important, then, are the compressions to make sure you have oxygen going up to the brain, because you’re still theoretically loaded with oxygen in your bloodstream if you had a sudden cardiac arrest from a ventricular fibrillation.
Your points are well taken, and we found that we had better outcomes when we just gave instructions to do compressions only, and that became the standard. Mick, you’ve had some experiences with that as well.
Dr. Molloy: If we’re going to have a long-term benefit from all this, we have to start doing this in elementary school and teaching kids basic life support and some basic health messaging.
I remember trying to get this across to a teacher one day and the teacher saying, “But why would we teach young kids to resuscitate each other?” I said, “I think you forget that the only 60-year-old person in the room is you. You train them, and we train them. They’re the ones who are going to respond and keep you alive. That’s the way you should be looking at this.” That completely changed the mindset of whether we should be doing this for the kids or not.
Dr. Pepe: In fact, what we find is that that’s exactly who gets saved. I had case after case where the kids at the school had learned CPR and saved the teachers or the administrator at the high school or elementary school. It’s a fantastic point that you bring up, Dr. Malloy.
Dr. Glatter: One other brief thing we can interject here is that the team was excellent on field in that they evaluated Damar Hamlin in a primary survey sense of ABCs (i.e., airway, breathing, and circulation) for things like a tension pneumothorax. In the sense in which he was hit, there are reversible causes. Making sure he didn’t have a tension pneumothorax that caused the arrest, in my mind, was critical.
Dr. Pepe: We do the same thing on a day-to-day basis with a car wreck, because it could be that the person had ventricular fibrillation and then had the wreck. It’s not always trauma. That’s a fantastic point that you’re making. That’s exactly what I think happened, and that’s what we do.
Dr. Glatter: Well, thank you, gentlemen. This was an informative and helpful discussion for our audience. I appreciate your time and expertise.
Dr. Glatter, is an attending physician at Lenox Hill Hospital in New York City and assistant professor of emergency medicine at Zucker School of Medicine at Hofstra/Northwell in Hempstead, New York. He is an editorial adviser and hosts the Hot Topics in EM series on Medscape. He is also a medical contributor for Forbes.
Dr. Pepe is a professor of internal medicine, surgery, pediatrics, public health, and emergency medicine at University of Texas Health Science Center in Houston. He’s also a global coordinator of the U.S. Metropolitan Municipalities EMS Medical Directors (“Eagles”) Coalition.
Dr. Molloy works clinically as a consultant in emergency medicine in Wexford General Hospital, part of the Ireland East Hospital Group (IEHG). Internationally, he is a member of the Disaster Medicine Section of the European Society of Emergency Medicine (EUSEM) and has been appointed by the Irish Medical Organization (IMO) as one of two Irish delegates to serve on the European Board and Section of Emergency Medicine of the European Union of Medical Specialists (UEMS), having served for a number of years on its predecessor, the Multidisciplinary Joint Committee on Emergency Medicine.
A version of this article first appeared on Medscape.com.
Simulation-based training effective for transesophageal echo
Simulation-based teaching of transesophageal echocardiography (TEE) improved cardiology fellows’ knowledge, skills, and comfort with the procedure, compared with traditional training, a new study shows.
“TEE learning may be hampered by the lack of availability of teachers and equipment and by the need for esophageal intubation, which is semi-invasive,” Augustin Coisne, MD, PhD, of the Cardiovascular Research Foundation in New York, said in an interview. “In this setting, simulation emerges as a key educational tool, but we were lacking evidence supporting simulation-based educational programs.”
Fellows in the simulation group achieved higher theoretical test scores and practical test scores after the training than did those in the traditional group.
Furthermore, Dr. Coisne said, “the results of the subgroup analyses were surprising and unexpected. The effect of the simulation-based training was greater among fellows at the beginning of fellowship – i.e., 2 years or less of training – in both theoretical and practical tests and in women [versus men] for the theoretical test.”
Their results, from the randomized SIMULATOR study, were published online in JAMA Cardiology.
More ready, more confident
The researchers randomly assigned 324 cardiology fellows (mean age, 26.4 years; about 30% women) inexperienced in TEE from 42 French university centers to TEE training with or without simulation support. Both groups participated in traditional didactic training using e-learning with an online course that is compulsory for all cardiology fellows in France.
The simulation group also participated in two 2-hour teaching sessions using a TEE simulator.
Each fellow completed a theoretical and a practical test prior to training to assess their baseline TEE level and again 3 months after the end of the training program. A TEE simulator (U/S Mentor Simulator; 3D Systems Simbionix) was used for all tests, and 24 certified echocardiography teachers served as both trainers and raters.
The theoretical tests included 20 online video-based questions to evaluate recognition of standard TEE views, normal anatomy, and some pathological cases. Fellows had 90 seconds to choose the best answer for each question from five multiple-choice options.
For the practical tests, fellows had 3 minutes to familiarize themselves with the handling of the simulator, without specific training and before the probe introduction.
They were asked to show 10 basic views on the simulator and had a maximum of 1 minute for each view.
The coprimary outcomes were the scores in the final theoretical and practical tests. TEE duration and the fellows’ self-assessment of their proficiency were also evaluated.
At baseline, the theoretical and practical test scores were similar between the groups (33.0 for the simulator group vs. 32.5 for the traditional group, and 44.2 vs. 46.1, respectively).
After training, the fellows in the simulation group had higher theoretical and practical test scores than those in the traditional group (47.2% vs. 38.3% and 74.5% vs. 59.0%, respectively).
Score changes were consistently higher when the pretraining scores were lower, an association that was stronger in the simulation group.
Dr. Coisne noted that subgroup analyses showed that the effectiveness of the simulation training was greater when performed at the beginning of the fellowship. On the theoretical test, the point increase was 11.9 for the simulation group versus 4.25 points for the traditional training group; for the practical test, the increases were 24.0 points versus 10.1 points.
After training, it took significantly less time for the simulation group to complete a TEE than it did the traditional group (8.3 vs. 9.4 minutes).
Furthermore, simulation group fellows reported that they felt more ready (mean score, 3.0 vs. 1.7) and more confident (mean score, 3.3 vs. 2.4) about performing a TEE alone after training.
“The simulation approach is definitively scalable to every institution,” Dr. Coisne said. “However, a medico-economic analysis should be interesting because the cost of the simulator and its maintenance might be a limitation to spread simulation-based teaching. The possibility for smaller hospitals to pool their financial input to share a TEE simulator could be considered to increase its cost-effectiveness.”
Real-world outcomes required
Commenting on the study, S. Justin Szawlewicz, MD, chair of cardiovascular medicine at Deborah Heart and Lung Center in Brown Mills, N.J., pointed out that the authors indicated that the number of TEEs performed by the trainees was not collected.
“This would be useful information to determine if those who received simulator training sought out and performed more TEEs, and also to determine if cardiology trainees in France perform a similar number of TEEs as cardiology trainees in the United States.”
In addition, he said, “the 4 hours of simulator training in TEE is extra education and experience that the standard trainees didn’t get. Would 4 extra hours of standard training didactics also improve trainees’ scores?”
Noting that the fellows’ ability to perform TEE in real patients was not assessed, Dr. Szawlewicz said, “a study could be designed that evaluated TEE images from real patients to see if trainees receiving simulator training performed better, more comprehensive and efficient TEEs than standard training.”
Nevertheless, he concluded, “Four hours of simulator training appears to improve TEE knowledge and skills. This is something we would consider at our institution.”
Like Dr. Szawlewicz, Michael Spooner, MD, MBA, of Mercy One North Iowa Heart Center in Mason City, and Kathryn Bertlacher, MD, of the University of Pittsburgh Medical Center, noted in a related editorial, “data are not provided about change in the learner’s behavior or performance on an actual TEE after the course, nor are there data about clinical outcomes such as patient safety or completeness of subsequent TEEs.
“This limitation, which is a limitation of most of the existing TEE simulation literature, is a high bar to cross,” they concluded. “Reaching this bar will require studies such as this to provide foundational understanding.”
Twin-Medical provided the TEE simulators. No relevant conflicts of interest were disclosed.
A version of this article first appeared on Medscape.com.
Simulation-based teaching of transesophageal echocardiography (TEE) improved cardiology fellows’ knowledge, skills, and comfort with the procedure, compared with traditional training, a new study shows.
“TEE learning may be hampered by the lack of availability of teachers and equipment and by the need for esophageal intubation, which is semi-invasive,” Augustin Coisne, MD, PhD, of the Cardiovascular Research Foundation in New York, said in an interview. “In this setting, simulation emerges as a key educational tool, but we were lacking evidence supporting simulation-based educational programs.”
Fellows in the simulation group achieved higher theoretical test scores and practical test scores after the training than did those in the traditional group.
Furthermore, Dr. Coisne said, “the results of the subgroup analyses were surprising and unexpected. The effect of the simulation-based training was greater among fellows at the beginning of fellowship – i.e., 2 years or less of training – in both theoretical and practical tests and in women [versus men] for the theoretical test.”
Their results, from the randomized SIMULATOR study, were published online in JAMA Cardiology.
More ready, more confident
The researchers randomly assigned 324 cardiology fellows (mean age, 26.4 years; about 30% women) inexperienced in TEE from 42 French university centers to TEE training with or without simulation support. Both groups participated in traditional didactic training using e-learning with an online course that is compulsory for all cardiology fellows in France.
The simulation group also participated in two 2-hour teaching sessions using a TEE simulator.
Each fellow completed a theoretical and a practical test prior to training to assess their baseline TEE level and again 3 months after the end of the training program. A TEE simulator (U/S Mentor Simulator; 3D Systems Simbionix) was used for all tests, and 24 certified echocardiography teachers served as both trainers and raters.
The theoretical tests included 20 online video-based questions to evaluate recognition of standard TEE views, normal anatomy, and some pathological cases. Fellows had 90 seconds to choose the best answer for each question from five multiple-choice options.
For the practical tests, fellows had 3 minutes to familiarize themselves with the handling of the simulator, without specific training and before the probe introduction.
They were asked to show 10 basic views on the simulator and had a maximum of 1 minute for each view.
The coprimary outcomes were the scores in the final theoretical and practical tests. TEE duration and the fellows’ self-assessment of their proficiency were also evaluated.
At baseline, the theoretical and practical test scores were similar between the groups (33.0 for the simulator group vs. 32.5 for the traditional group, and 44.2 vs. 46.1, respectively).
After training, the fellows in the simulation group had higher theoretical and practical test scores than those in the traditional group (47.2% vs. 38.3% and 74.5% vs. 59.0%, respectively).
Score changes were consistently higher when the pretraining scores were lower, an association that was stronger in the simulation group.
Dr. Coisne noted that subgroup analyses showed that the effectiveness of the simulation training was greater when performed at the beginning of the fellowship. On the theoretical test, the point increase was 11.9 for the simulation group versus 4.25 points for the traditional training group; for the practical test, the increases were 24.0 points versus 10.1 points.
After training, it took significantly less time for the simulation group to complete a TEE than it did the traditional group (8.3 vs. 9.4 minutes).
Furthermore, simulation group fellows reported that they felt more ready (mean score, 3.0 vs. 1.7) and more confident (mean score, 3.3 vs. 2.4) about performing a TEE alone after training.
“The simulation approach is definitively scalable to every institution,” Dr. Coisne said. “However, a medico-economic analysis should be interesting because the cost of the simulator and its maintenance might be a limitation to spread simulation-based teaching. The possibility for smaller hospitals to pool their financial input to share a TEE simulator could be considered to increase its cost-effectiveness.”
Real-world outcomes required
Commenting on the study, S. Justin Szawlewicz, MD, chair of cardiovascular medicine at Deborah Heart and Lung Center in Brown Mills, N.J., pointed out that the authors indicated that the number of TEEs performed by the trainees was not collected.
“This would be useful information to determine if those who received simulator training sought out and performed more TEEs, and also to determine if cardiology trainees in France perform a similar number of TEEs as cardiology trainees in the United States.”
In addition, he said, “the 4 hours of simulator training in TEE is extra education and experience that the standard trainees didn’t get. Would 4 extra hours of standard training didactics also improve trainees’ scores?”
Noting that the fellows’ ability to perform TEE in real patients was not assessed, Dr. Szawlewicz said, “a study could be designed that evaluated TEE images from real patients to see if trainees receiving simulator training performed better, more comprehensive and efficient TEEs than standard training.”
Nevertheless, he concluded, “Four hours of simulator training appears to improve TEE knowledge and skills. This is something we would consider at our institution.”
Like Dr. Szawlewicz, Michael Spooner, MD, MBA, of Mercy One North Iowa Heart Center in Mason City, and Kathryn Bertlacher, MD, of the University of Pittsburgh Medical Center, noted in a related editorial, “data are not provided about change in the learner’s behavior or performance on an actual TEE after the course, nor are there data about clinical outcomes such as patient safety or completeness of subsequent TEEs.
“This limitation, which is a limitation of most of the existing TEE simulation literature, is a high bar to cross,” they concluded. “Reaching this bar will require studies such as this to provide foundational understanding.”
Twin-Medical provided the TEE simulators. No relevant conflicts of interest were disclosed.
A version of this article first appeared on Medscape.com.
Simulation-based teaching of transesophageal echocardiography (TEE) improved cardiology fellows’ knowledge, skills, and comfort with the procedure, compared with traditional training, a new study shows.
“TEE learning may be hampered by the lack of availability of teachers and equipment and by the need for esophageal intubation, which is semi-invasive,” Augustin Coisne, MD, PhD, of the Cardiovascular Research Foundation in New York, said in an interview. “In this setting, simulation emerges as a key educational tool, but we were lacking evidence supporting simulation-based educational programs.”
Fellows in the simulation group achieved higher theoretical test scores and practical test scores after the training than did those in the traditional group.
Furthermore, Dr. Coisne said, “the results of the subgroup analyses were surprising and unexpected. The effect of the simulation-based training was greater among fellows at the beginning of fellowship – i.e., 2 years or less of training – in both theoretical and practical tests and in women [versus men] for the theoretical test.”
Their results, from the randomized SIMULATOR study, were published online in JAMA Cardiology.
More ready, more confident
The researchers randomly assigned 324 cardiology fellows (mean age, 26.4 years; about 30% women) inexperienced in TEE from 42 French university centers to TEE training with or without simulation support. Both groups participated in traditional didactic training using e-learning with an online course that is compulsory for all cardiology fellows in France.
The simulation group also participated in two 2-hour teaching sessions using a TEE simulator.
Each fellow completed a theoretical and a practical test prior to training to assess their baseline TEE level and again 3 months after the end of the training program. A TEE simulator (U/S Mentor Simulator; 3D Systems Simbionix) was used for all tests, and 24 certified echocardiography teachers served as both trainers and raters.
The theoretical tests included 20 online video-based questions to evaluate recognition of standard TEE views, normal anatomy, and some pathological cases. Fellows had 90 seconds to choose the best answer for each question from five multiple-choice options.
For the practical tests, fellows had 3 minutes to familiarize themselves with the handling of the simulator, without specific training and before the probe introduction.
They were asked to show 10 basic views on the simulator and had a maximum of 1 minute for each view.
The coprimary outcomes were the scores in the final theoretical and practical tests. TEE duration and the fellows’ self-assessment of their proficiency were also evaluated.
At baseline, the theoretical and practical test scores were similar between the groups (33.0 for the simulator group vs. 32.5 for the traditional group, and 44.2 vs. 46.1, respectively).
After training, the fellows in the simulation group had higher theoretical and practical test scores than those in the traditional group (47.2% vs. 38.3% and 74.5% vs. 59.0%, respectively).
Score changes were consistently higher when the pretraining scores were lower, an association that was stronger in the simulation group.
Dr. Coisne noted that subgroup analyses showed that the effectiveness of the simulation training was greater when performed at the beginning of the fellowship. On the theoretical test, the point increase was 11.9 for the simulation group versus 4.25 points for the traditional training group; for the practical test, the increases were 24.0 points versus 10.1 points.
After training, it took significantly less time for the simulation group to complete a TEE than it did the traditional group (8.3 vs. 9.4 minutes).
Furthermore, simulation group fellows reported that they felt more ready (mean score, 3.0 vs. 1.7) and more confident (mean score, 3.3 vs. 2.4) about performing a TEE alone after training.
“The simulation approach is definitively scalable to every institution,” Dr. Coisne said. “However, a medico-economic analysis should be interesting because the cost of the simulator and its maintenance might be a limitation to spread simulation-based teaching. The possibility for smaller hospitals to pool their financial input to share a TEE simulator could be considered to increase its cost-effectiveness.”
Real-world outcomes required
Commenting on the study, S. Justin Szawlewicz, MD, chair of cardiovascular medicine at Deborah Heart and Lung Center in Brown Mills, N.J., pointed out that the authors indicated that the number of TEEs performed by the trainees was not collected.
“This would be useful information to determine if those who received simulator training sought out and performed more TEEs, and also to determine if cardiology trainees in France perform a similar number of TEEs as cardiology trainees in the United States.”
In addition, he said, “the 4 hours of simulator training in TEE is extra education and experience that the standard trainees didn’t get. Would 4 extra hours of standard training didactics also improve trainees’ scores?”
Noting that the fellows’ ability to perform TEE in real patients was not assessed, Dr. Szawlewicz said, “a study could be designed that evaluated TEE images from real patients to see if trainees receiving simulator training performed better, more comprehensive and efficient TEEs than standard training.”
Nevertheless, he concluded, “Four hours of simulator training appears to improve TEE knowledge and skills. This is something we would consider at our institution.”
Like Dr. Szawlewicz, Michael Spooner, MD, MBA, of Mercy One North Iowa Heart Center in Mason City, and Kathryn Bertlacher, MD, of the University of Pittsburgh Medical Center, noted in a related editorial, “data are not provided about change in the learner’s behavior or performance on an actual TEE after the course, nor are there data about clinical outcomes such as patient safety or completeness of subsequent TEEs.
“This limitation, which is a limitation of most of the existing TEE simulation literature, is a high bar to cross,” they concluded. “Reaching this bar will require studies such as this to provide foundational understanding.”
Twin-Medical provided the TEE simulators. No relevant conflicts of interest were disclosed.
A version of this article first appeared on Medscape.com.
FROM JAMA CARDIOLOGY
By the numbers: Cardiology slow to add women, IMGs join more quickly
Despite Mark Twain’s assertion that “there are three kinds of lies: lies, damned lies, and statistics,” we’re going to dive into 20 years’ worth of data and, hopefully, come up with a few statistics that shed some light on the specialty’s workforce since Cardiology News published its first issue in February 2003.
We start with a major issue over these last 20 years: The participation of women in the specialty.
Back in July of 2002, just a few months before the first issue of Cardiology News was published, W. Bruce Fye, MD, then-president of the American College of Cardiology, wrote, “We need to do more to attract female medical graduates to our specialty because they represent almost one-half of the new doctors trained in this country. Cardiology needs to take full advantage of this large talent pool”
Data from the American Medical Association confirm that assertion: Of the nearly 20,000 postgraduate cardiologists in practice that year, only 7.8% were women. And that was at a time when more than 42% of medical school graduates were women, Dr. Fye noted, while also pointing out that “only 10% of cardiology trainees are female, and just 6% of ACC fellows are women.”
The gap between men and women has closed somewhat in the last 20 years, but the specialty continues to lag behind the profession as a whole. Women represented 16.7% of cardiologists in 2022, versus 37% of physicians overall, AMA data show. In 2019, for the first time, the majority of U.S. medical school students (50.5%) were women, according to the Association of American Medical Colleges.
A look at residency numbers from the Accreditation Council for Graduate Medical Education shows that continued slow improvement in the number of women can be expected, as 25.5% of cardiovascular disease residents were women during the 2021-2022 academic year. Only 2 of the 19 other internal medicine subspecialties were lower, and they happened to be interventional cardiology (20.1%) and clinical cardiac electrophysiology (14.5%).
When men are added to the mix, cardiovascular disease had a total of 3,320 active residents training in 268 programs in 2021-2022, making it the largest of the IM subspecialties in both respects. The resident total is up 57% since 2003, when it came in at 2,117, while programs have increased 55% from the 173 that were operating 2 decades ago. During the year in the middle (2011-2012), there were 2,521 residents in 187 programs, so a larger share of the growth has occurred in the last 10 years, the ACGME data indicate.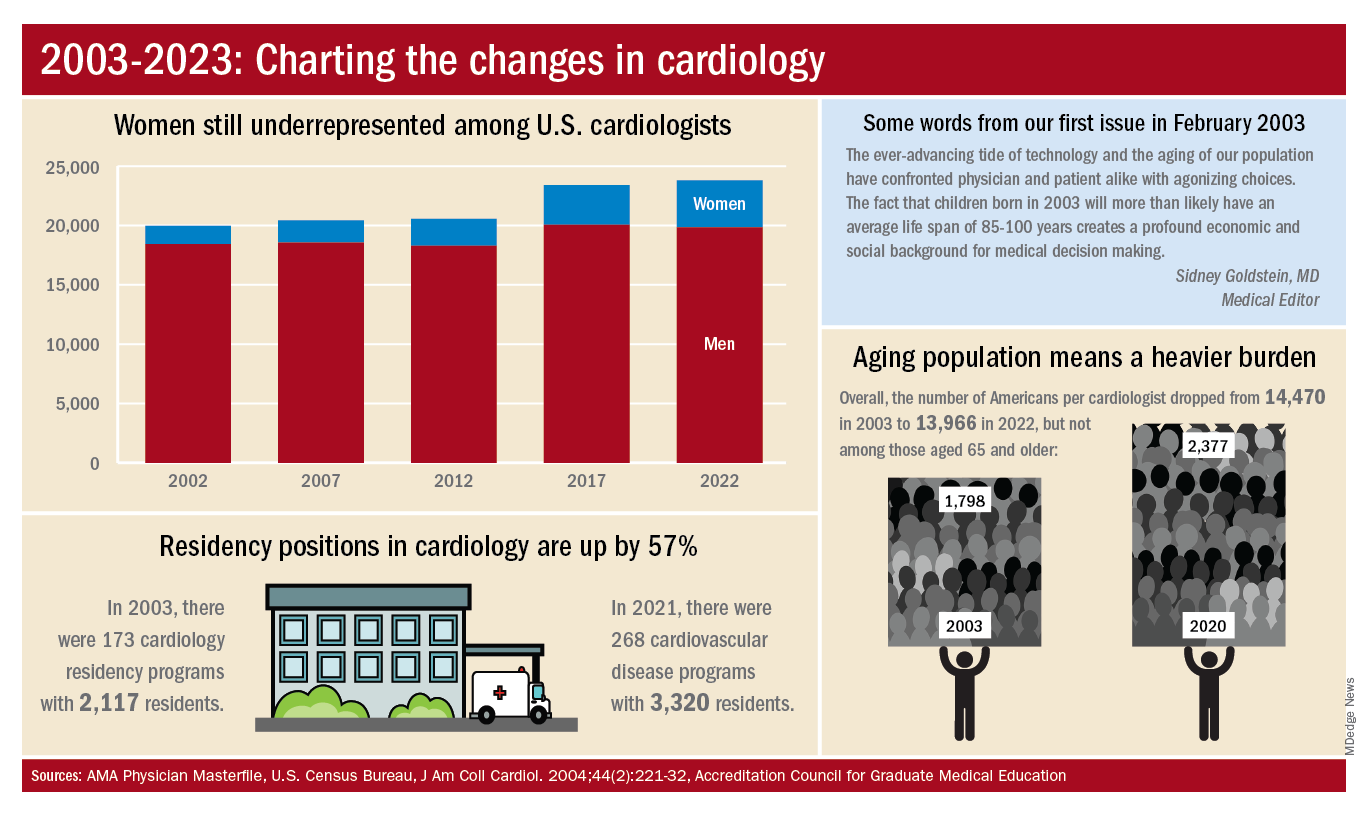
The shortage of cardiologists that Dr. Fye and others wrote about 20 years ago has not gone away. A 2018 report from health consulting firm PYA noted the increase in obesity and the low number of medical school graduates choosing the specialty. “Older and fewer physicians specializing in cardiology, coupled with the aging of baby boomers and gravitation toward practice in urban areas, will continue to exacerbate shortages in physician services in the specialty of cardiology, especially in rural areas, over the next decade,” PYA principal Lyle Oelrich wrote.
A little math appears to back up the claims of a cardiologist shortage. Based on census figures for the U.S. population in 2003, there were 14,470 Americans for each of the cardiologists reported by the AMA. That figure dropped to 13,966 by 2022, which seems like an improvement, but it comes with a caveat. The number of Americans aged 65 years and older increased from 1,798 to 2,377 per cardiologist as of 2020, the latest year for which population data were available by age.
One source of growth in the cardiology workforce has been perhaps its most significant minority: international medical graduates. Even by 2004, IMGs represented a much larger segment of all cardiologists (30.0%) than did women (9.3%), based on AMA data. To put it another way, there were more IMGs specializing in cardiovascular disease (6,615) in 2004 than there were women (3,963) in 2022.
The latest data on cardiology training programs – overall numbers were not available – put IMGs at 39.2% for the 2019-2020 academic year. The 2022 fellowship match provides a slightly smaller proportion of IMGs (37.4%) filling cardiovascular disease positions, according to the National Resident Matching Program.
Despite Mark Twain’s assertion that “there are three kinds of lies: lies, damned lies, and statistics,” we’re going to dive into 20 years’ worth of data and, hopefully, come up with a few statistics that shed some light on the specialty’s workforce since Cardiology News published its first issue in February 2003.
We start with a major issue over these last 20 years: The participation of women in the specialty.
Back in July of 2002, just a few months before the first issue of Cardiology News was published, W. Bruce Fye, MD, then-president of the American College of Cardiology, wrote, “We need to do more to attract female medical graduates to our specialty because they represent almost one-half of the new doctors trained in this country. Cardiology needs to take full advantage of this large talent pool”
Data from the American Medical Association confirm that assertion: Of the nearly 20,000 postgraduate cardiologists in practice that year, only 7.8% were women. And that was at a time when more than 42% of medical school graduates were women, Dr. Fye noted, while also pointing out that “only 10% of cardiology trainees are female, and just 6% of ACC fellows are women.”
The gap between men and women has closed somewhat in the last 20 years, but the specialty continues to lag behind the profession as a whole. Women represented 16.7% of cardiologists in 2022, versus 37% of physicians overall, AMA data show. In 2019, for the first time, the majority of U.S. medical school students (50.5%) were women, according to the Association of American Medical Colleges.
A look at residency numbers from the Accreditation Council for Graduate Medical Education shows that continued slow improvement in the number of women can be expected, as 25.5% of cardiovascular disease residents were women during the 2021-2022 academic year. Only 2 of the 19 other internal medicine subspecialties were lower, and they happened to be interventional cardiology (20.1%) and clinical cardiac electrophysiology (14.5%).
When men are added to the mix, cardiovascular disease had a total of 3,320 active residents training in 268 programs in 2021-2022, making it the largest of the IM subspecialties in both respects. The resident total is up 57% since 2003, when it came in at 2,117, while programs have increased 55% from the 173 that were operating 2 decades ago. During the year in the middle (2011-2012), there were 2,521 residents in 187 programs, so a larger share of the growth has occurred in the last 10 years, the ACGME data indicate.
The shortage of cardiologists that Dr. Fye and others wrote about 20 years ago has not gone away. A 2018 report from health consulting firm PYA noted the increase in obesity and the low number of medical school graduates choosing the specialty. “Older and fewer physicians specializing in cardiology, coupled with the aging of baby boomers and gravitation toward practice in urban areas, will continue to exacerbate shortages in physician services in the specialty of cardiology, especially in rural areas, over the next decade,” PYA principal Lyle Oelrich wrote.
A little math appears to back up the claims of a cardiologist shortage. Based on census figures for the U.S. population in 2003, there were 14,470 Americans for each of the cardiologists reported by the AMA. That figure dropped to 13,966 by 2022, which seems like an improvement, but it comes with a caveat. The number of Americans aged 65 years and older increased from 1,798 to 2,377 per cardiologist as of 2020, the latest year for which population data were available by age.
One source of growth in the cardiology workforce has been perhaps its most significant minority: international medical graduates. Even by 2004, IMGs represented a much larger segment of all cardiologists (30.0%) than did women (9.3%), based on AMA data. To put it another way, there were more IMGs specializing in cardiovascular disease (6,615) in 2004 than there were women (3,963) in 2022.
The latest data on cardiology training programs – overall numbers were not available – put IMGs at 39.2% for the 2019-2020 academic year. The 2022 fellowship match provides a slightly smaller proportion of IMGs (37.4%) filling cardiovascular disease positions, according to the National Resident Matching Program.
Despite Mark Twain’s assertion that “there are three kinds of lies: lies, damned lies, and statistics,” we’re going to dive into 20 years’ worth of data and, hopefully, come up with a few statistics that shed some light on the specialty’s workforce since Cardiology News published its first issue in February 2003.
We start with a major issue over these last 20 years: The participation of women in the specialty.
Back in July of 2002, just a few months before the first issue of Cardiology News was published, W. Bruce Fye, MD, then-president of the American College of Cardiology, wrote, “We need to do more to attract female medical graduates to our specialty because they represent almost one-half of the new doctors trained in this country. Cardiology needs to take full advantage of this large talent pool”
Data from the American Medical Association confirm that assertion: Of the nearly 20,000 postgraduate cardiologists in practice that year, only 7.8% were women. And that was at a time when more than 42% of medical school graduates were women, Dr. Fye noted, while also pointing out that “only 10% of cardiology trainees are female, and just 6% of ACC fellows are women.”
The gap between men and women has closed somewhat in the last 20 years, but the specialty continues to lag behind the profession as a whole. Women represented 16.7% of cardiologists in 2022, versus 37% of physicians overall, AMA data show. In 2019, for the first time, the majority of U.S. medical school students (50.5%) were women, according to the Association of American Medical Colleges.
A look at residency numbers from the Accreditation Council for Graduate Medical Education shows that continued slow improvement in the number of women can be expected, as 25.5% of cardiovascular disease residents were women during the 2021-2022 academic year. Only 2 of the 19 other internal medicine subspecialties were lower, and they happened to be interventional cardiology (20.1%) and clinical cardiac electrophysiology (14.5%).
When men are added to the mix, cardiovascular disease had a total of 3,320 active residents training in 268 programs in 2021-2022, making it the largest of the IM subspecialties in both respects. The resident total is up 57% since 2003, when it came in at 2,117, while programs have increased 55% from the 173 that were operating 2 decades ago. During the year in the middle (2011-2012), there were 2,521 residents in 187 programs, so a larger share of the growth has occurred in the last 10 years, the ACGME data indicate.
The shortage of cardiologists that Dr. Fye and others wrote about 20 years ago has not gone away. A 2018 report from health consulting firm PYA noted the increase in obesity and the low number of medical school graduates choosing the specialty. “Older and fewer physicians specializing in cardiology, coupled with the aging of baby boomers and gravitation toward practice in urban areas, will continue to exacerbate shortages in physician services in the specialty of cardiology, especially in rural areas, over the next decade,” PYA principal Lyle Oelrich wrote.
A little math appears to back up the claims of a cardiologist shortage. Based on census figures for the U.S. population in 2003, there were 14,470 Americans for each of the cardiologists reported by the AMA. That figure dropped to 13,966 by 2022, which seems like an improvement, but it comes with a caveat. The number of Americans aged 65 years and older increased from 1,798 to 2,377 per cardiologist as of 2020, the latest year for which population data were available by age.
One source of growth in the cardiology workforce has been perhaps its most significant minority: international medical graduates. Even by 2004, IMGs represented a much larger segment of all cardiologists (30.0%) than did women (9.3%), based on AMA data. To put it another way, there were more IMGs specializing in cardiovascular disease (6,615) in 2004 than there were women (3,963) in 2022.
The latest data on cardiology training programs – overall numbers were not available – put IMGs at 39.2% for the 2019-2020 academic year. The 2022 fellowship match provides a slightly smaller proportion of IMGs (37.4%) filling cardiovascular disease positions, according to the National Resident Matching Program.
After PCI, 1-month beats 12-month DAPT in high-risk patients
Replacing dual-antiplatelet therapy (DAPT) with clopidogrel alone 1 month after percutaneous intervention (PCI) offers a lower risk of bleeding with comparable protection against cardiovascular events, according to two subgroup analyses of the Japanese STOPDAPT-2 and STOPDAPT-2 ACS trials.
The objective of these two analyses was to evaluate whether there was a benefit-to-risk ratio advantage for those who entered the study with high bleeding risk or who had undergone a complex PCI. Overall, bleeding risk was reduced without a major increase in cardiovascular events regardless of subgroup, according to results published by a multicenter group of Japanese investigators.
In this substudy, like the previously published studies from which the data were drawn, the primary endpoint was a composite of cardiovascular death, myocardial infarction, definite stent thrombosis, stroke, and Thrombolysis In Myocardial Infarction bleeding (major or minor).
The proportion of patients in the 1-month and 12-month DAPT groups reaching this composite endpoint at 1 year was not significantly different among patients stratified by baseline bleeding risk or by PCI complexity, according to a multicenter group of authors led by Takeshi Kimura, MD, department of cardiovascular medicine, Kyoto University.
Shortened DAPT is focus of multiple trials
The new analysis, published in JACC Asia, is a follow-up to the 2019 STOPDAPT-2 trial, published in JAMA, and the 2022 STOPDAPT-2 ACS trial, published in JAMA Cardiology. The first tested 1- versus 12-month DAPT in PCI patients receiving a drug-eluting stent. The second study compared the same strategies in patients undergoing PCI to treat an acute coronary syndrome (ACS).
Both studies were conducted in Japan. DAPT consisted of the P2Y12 receptor inhibitor clopidogrel plus aspirin. The experimental arm received this regimen for 1 month followed by clopidogrel monotherapy. The control arm remained on DAPT for 12 months.
The study is potentially important because it addresses the challenge of finding “the sweet spot of antiplatelet therapy in East Asian patients,” according to the coauthors of an accompanying editorial in the same issue of JACC Asia.
Previous data suggest East Asians have a higher risk of bleeding but lower anti-ischemic benefits from DAPT therapy, explained the coauthors, Antonio Greco, MD and Davide Capodanno, MD, PhD, both from the University of Catania (Italy). They praised the effort to explore this question.
In the STOPDAPT-2 trial, the shortened DAPT regimen was associated with a significantly lower rate of a composite endpoint of cardiovascular and bleeding events than standard DAPT, meeting criteria for superiority as well as noninferiority. In the STOPDAPT-2 ACS trial, shortened DAPT failed to achieve noninferiority to standard DAPT because of an increase in cardiovascular events despite a reduction in bleeding events.
Neither of these studies specifically compared shortened to standard DAPT in patients with high bleeding risk or in patients who underwent complex PCI, which are among the most common patient groups in which to consider a modified DAPT regimen. To do this, two new substudies were performed with the combined data from 5,997 patients in the two STOPDAPT-2 trials.
Two candidate groups for shortened DAPT evaluated
In the first substudy, the 1,893 patients who met criteria for high bleeding risk were compared with the 4,104 who did not. In those with a high risk of bleeding, the proportion reaching a primary endpoint at 1 year was lower, but not significantly different, for those on 1-month versus standard DAPT (5.01% vs. 5.14%). This was also true in those without an elevated bleeding risk (1.90% vs. 2.02%).
In the second substudy, 999 patients who had a complex PCI, defined by such characteristics as implantation of at least three stents or chronic total occlusion in the target lesions, were compared with the 4,998 who did not. Again, the primary endpoint was lower in both those who had a complex PCI (3.15% vs. 4.07%) and those who did not (2.78% vs. 2.82%).
Not surprisingly, patients with a high bleeding risk benefited from a substantially lower risk of bleeding events on the 1-month DAPT regimen (0.66% vs. 2.27%). The cost was a higher risk of cardiovascular events (4.35% vs. 3.52%), but this difference did not reach significance. Those without an elevated bleeding risk also had a lower risk of bleeding events (0.43% vs. 0.85%) but a higher risk of cardiovascular events (1.56% vs. 1.22%). Again, differences were nonsignificant. In the substudy evaluating DAPT duration in relation to complex PCI, the rate of cardiovascular events at 1 year in those treated with short versus 12-month DAPT was nearly identical (2.53% vs. 2.52%). In the non–complex PCI patients, event rates were nonsignificantly greater on the shortened DAPT regimen (2.38% vs. 1.86%), but the bleeding rate was lower on shortened DAPT whether PCI had been complex (0.63% vs. 1.75%) or not (0.48% vs. 1.22%).
In the absence of any major signal that complex PCI benefited from longer duration DAPT, “complex PCI might not be an appropriate determinant for DAPT durations,” according to Dr. Kimura and coinvestigators.
Study data might not be generalizable
Dr. Greco and Dr. Capodanno pointed out that there are differences between patients and PCI practices in Japan relative to other areas of the world, limiting the generalizability of these findings even if the question is relevant.
“This is an approach that might be suggested for patients at high bleeding risk who have the characteristics of the patients enrolled in the STOPDAPT-2 trials,” Dr. Capodanno said in an interview. In his own PCI practice treating ACS patients, “I would not feel safe enough with clopidogrel monotherapy after only 1 month.”
He considers the ACS population to have a particularly “delicate bleeding-ischemia trade-off,” which is why he thinks this question is relevant and needs to be explored further in additional populations. However, he might design trials differently in his own practice setting. For example, he would at the very least be interested in testing a more potent P2Y12 inhibitor such as ticagrelor when considering a single antiplatelet agent after a limited course of DAPT.
One message from this study is that “bleeding risk trumps PCI complexity,” according to Deepak L. Bhatt, MD, who recently assumed the position of director of Mount Sinai Heart in New York. He liked the approach the investigators took to address a complex and relevant clinical issue, but he also expressed reservations about the clinical applicability of this subgroup analysis.
“We really need more data before uniformly shortening DAPT duration in all patients,” Dr. Bhatt said in an interview. He considers this a hot clinical issue that is likely to generate more trials. He hopes these will provide more definitive evidence of when and how DAPT duration can be reduced. Overall, he anticipates progress toward tailoring therapy in specific populations in order to achieve the best risk-to-benefit balance.
Dr. Kimura has financial relationships with Boston Scientific, Daiichi Sankyo, Sanofi, Terumo, and Abbott Medical Japan, which provided funding for the STOPDAPT-2 and STOPDAPT-2 ACS trials. Dr. Capodanno reported financial relationships with Amgen, Arena, Chiesi, Daiichi Sakyo, Sanofi Aventis, and Terumo. Dr. Bhatt reported financial relationships with more than 20 pharmaceutical companies, including Abbott Medical.
Replacing dual-antiplatelet therapy (DAPT) with clopidogrel alone 1 month after percutaneous intervention (PCI) offers a lower risk of bleeding with comparable protection against cardiovascular events, according to two subgroup analyses of the Japanese STOPDAPT-2 and STOPDAPT-2 ACS trials.
The objective of these two analyses was to evaluate whether there was a benefit-to-risk ratio advantage for those who entered the study with high bleeding risk or who had undergone a complex PCI. Overall, bleeding risk was reduced without a major increase in cardiovascular events regardless of subgroup, according to results published by a multicenter group of Japanese investigators.
In this substudy, like the previously published studies from which the data were drawn, the primary endpoint was a composite of cardiovascular death, myocardial infarction, definite stent thrombosis, stroke, and Thrombolysis In Myocardial Infarction bleeding (major or minor).
The proportion of patients in the 1-month and 12-month DAPT groups reaching this composite endpoint at 1 year was not significantly different among patients stratified by baseline bleeding risk or by PCI complexity, according to a multicenter group of authors led by Takeshi Kimura, MD, department of cardiovascular medicine, Kyoto University.
Shortened DAPT is focus of multiple trials
The new analysis, published in JACC Asia, is a follow-up to the 2019 STOPDAPT-2 trial, published in JAMA, and the 2022 STOPDAPT-2 ACS trial, published in JAMA Cardiology. The first tested 1- versus 12-month DAPT in PCI patients receiving a drug-eluting stent. The second study compared the same strategies in patients undergoing PCI to treat an acute coronary syndrome (ACS).
Both studies were conducted in Japan. DAPT consisted of the P2Y12 receptor inhibitor clopidogrel plus aspirin. The experimental arm received this regimen for 1 month followed by clopidogrel monotherapy. The control arm remained on DAPT for 12 months.
The study is potentially important because it addresses the challenge of finding “the sweet spot of antiplatelet therapy in East Asian patients,” according to the coauthors of an accompanying editorial in the same issue of JACC Asia.
Previous data suggest East Asians have a higher risk of bleeding but lower anti-ischemic benefits from DAPT therapy, explained the coauthors, Antonio Greco, MD and Davide Capodanno, MD, PhD, both from the University of Catania (Italy). They praised the effort to explore this question.
In the STOPDAPT-2 trial, the shortened DAPT regimen was associated with a significantly lower rate of a composite endpoint of cardiovascular and bleeding events than standard DAPT, meeting criteria for superiority as well as noninferiority. In the STOPDAPT-2 ACS trial, shortened DAPT failed to achieve noninferiority to standard DAPT because of an increase in cardiovascular events despite a reduction in bleeding events.
Neither of these studies specifically compared shortened to standard DAPT in patients with high bleeding risk or in patients who underwent complex PCI, which are among the most common patient groups in which to consider a modified DAPT regimen. To do this, two new substudies were performed with the combined data from 5,997 patients in the two STOPDAPT-2 trials.
Two candidate groups for shortened DAPT evaluated
In the first substudy, the 1,893 patients who met criteria for high bleeding risk were compared with the 4,104 who did not. In those with a high risk of bleeding, the proportion reaching a primary endpoint at 1 year was lower, but not significantly different, for those on 1-month versus standard DAPT (5.01% vs. 5.14%). This was also true in those without an elevated bleeding risk (1.90% vs. 2.02%).
In the second substudy, 999 patients who had a complex PCI, defined by such characteristics as implantation of at least three stents or chronic total occlusion in the target lesions, were compared with the 4,998 who did not. Again, the primary endpoint was lower in both those who had a complex PCI (3.15% vs. 4.07%) and those who did not (2.78% vs. 2.82%).
Not surprisingly, patients with a high bleeding risk benefited from a substantially lower risk of bleeding events on the 1-month DAPT regimen (0.66% vs. 2.27%). The cost was a higher risk of cardiovascular events (4.35% vs. 3.52%), but this difference did not reach significance. Those without an elevated bleeding risk also had a lower risk of bleeding events (0.43% vs. 0.85%) but a higher risk of cardiovascular events (1.56% vs. 1.22%). Again, differences were nonsignificant. In the substudy evaluating DAPT duration in relation to complex PCI, the rate of cardiovascular events at 1 year in those treated with short versus 12-month DAPT was nearly identical (2.53% vs. 2.52%). In the non–complex PCI patients, event rates were nonsignificantly greater on the shortened DAPT regimen (2.38% vs. 1.86%), but the bleeding rate was lower on shortened DAPT whether PCI had been complex (0.63% vs. 1.75%) or not (0.48% vs. 1.22%).
In the absence of any major signal that complex PCI benefited from longer duration DAPT, “complex PCI might not be an appropriate determinant for DAPT durations,” according to Dr. Kimura and coinvestigators.
Study data might not be generalizable
Dr. Greco and Dr. Capodanno pointed out that there are differences between patients and PCI practices in Japan relative to other areas of the world, limiting the generalizability of these findings even if the question is relevant.
“This is an approach that might be suggested for patients at high bleeding risk who have the characteristics of the patients enrolled in the STOPDAPT-2 trials,” Dr. Capodanno said in an interview. In his own PCI practice treating ACS patients, “I would not feel safe enough with clopidogrel monotherapy after only 1 month.”
He considers the ACS population to have a particularly “delicate bleeding-ischemia trade-off,” which is why he thinks this question is relevant and needs to be explored further in additional populations. However, he might design trials differently in his own practice setting. For example, he would at the very least be interested in testing a more potent P2Y12 inhibitor such as ticagrelor when considering a single antiplatelet agent after a limited course of DAPT.
One message from this study is that “bleeding risk trumps PCI complexity,” according to Deepak L. Bhatt, MD, who recently assumed the position of director of Mount Sinai Heart in New York. He liked the approach the investigators took to address a complex and relevant clinical issue, but he also expressed reservations about the clinical applicability of this subgroup analysis.
“We really need more data before uniformly shortening DAPT duration in all patients,” Dr. Bhatt said in an interview. He considers this a hot clinical issue that is likely to generate more trials. He hopes these will provide more definitive evidence of when and how DAPT duration can be reduced. Overall, he anticipates progress toward tailoring therapy in specific populations in order to achieve the best risk-to-benefit balance.
Dr. Kimura has financial relationships with Boston Scientific, Daiichi Sankyo, Sanofi, Terumo, and Abbott Medical Japan, which provided funding for the STOPDAPT-2 and STOPDAPT-2 ACS trials. Dr. Capodanno reported financial relationships with Amgen, Arena, Chiesi, Daiichi Sakyo, Sanofi Aventis, and Terumo. Dr. Bhatt reported financial relationships with more than 20 pharmaceutical companies, including Abbott Medical.
Replacing dual-antiplatelet therapy (DAPT) with clopidogrel alone 1 month after percutaneous intervention (PCI) offers a lower risk of bleeding with comparable protection against cardiovascular events, according to two subgroup analyses of the Japanese STOPDAPT-2 and STOPDAPT-2 ACS trials.
The objective of these two analyses was to evaluate whether there was a benefit-to-risk ratio advantage for those who entered the study with high bleeding risk or who had undergone a complex PCI. Overall, bleeding risk was reduced without a major increase in cardiovascular events regardless of subgroup, according to results published by a multicenter group of Japanese investigators.
In this substudy, like the previously published studies from which the data were drawn, the primary endpoint was a composite of cardiovascular death, myocardial infarction, definite stent thrombosis, stroke, and Thrombolysis In Myocardial Infarction bleeding (major or minor).
The proportion of patients in the 1-month and 12-month DAPT groups reaching this composite endpoint at 1 year was not significantly different among patients stratified by baseline bleeding risk or by PCI complexity, according to a multicenter group of authors led by Takeshi Kimura, MD, department of cardiovascular medicine, Kyoto University.
Shortened DAPT is focus of multiple trials
The new analysis, published in JACC Asia, is a follow-up to the 2019 STOPDAPT-2 trial, published in JAMA, and the 2022 STOPDAPT-2 ACS trial, published in JAMA Cardiology. The first tested 1- versus 12-month DAPT in PCI patients receiving a drug-eluting stent. The second study compared the same strategies in patients undergoing PCI to treat an acute coronary syndrome (ACS).
Both studies were conducted in Japan. DAPT consisted of the P2Y12 receptor inhibitor clopidogrel plus aspirin. The experimental arm received this regimen for 1 month followed by clopidogrel monotherapy. The control arm remained on DAPT for 12 months.
The study is potentially important because it addresses the challenge of finding “the sweet spot of antiplatelet therapy in East Asian patients,” according to the coauthors of an accompanying editorial in the same issue of JACC Asia.
Previous data suggest East Asians have a higher risk of bleeding but lower anti-ischemic benefits from DAPT therapy, explained the coauthors, Antonio Greco, MD and Davide Capodanno, MD, PhD, both from the University of Catania (Italy). They praised the effort to explore this question.
In the STOPDAPT-2 trial, the shortened DAPT regimen was associated with a significantly lower rate of a composite endpoint of cardiovascular and bleeding events than standard DAPT, meeting criteria for superiority as well as noninferiority. In the STOPDAPT-2 ACS trial, shortened DAPT failed to achieve noninferiority to standard DAPT because of an increase in cardiovascular events despite a reduction in bleeding events.
Neither of these studies specifically compared shortened to standard DAPT in patients with high bleeding risk or in patients who underwent complex PCI, which are among the most common patient groups in which to consider a modified DAPT regimen. To do this, two new substudies were performed with the combined data from 5,997 patients in the two STOPDAPT-2 trials.
Two candidate groups for shortened DAPT evaluated
In the first substudy, the 1,893 patients who met criteria for high bleeding risk were compared with the 4,104 who did not. In those with a high risk of bleeding, the proportion reaching a primary endpoint at 1 year was lower, but not significantly different, for those on 1-month versus standard DAPT (5.01% vs. 5.14%). This was also true in those without an elevated bleeding risk (1.90% vs. 2.02%).
In the second substudy, 999 patients who had a complex PCI, defined by such characteristics as implantation of at least three stents or chronic total occlusion in the target lesions, were compared with the 4,998 who did not. Again, the primary endpoint was lower in both those who had a complex PCI (3.15% vs. 4.07%) and those who did not (2.78% vs. 2.82%).
Not surprisingly, patients with a high bleeding risk benefited from a substantially lower risk of bleeding events on the 1-month DAPT regimen (0.66% vs. 2.27%). The cost was a higher risk of cardiovascular events (4.35% vs. 3.52%), but this difference did not reach significance. Those without an elevated bleeding risk also had a lower risk of bleeding events (0.43% vs. 0.85%) but a higher risk of cardiovascular events (1.56% vs. 1.22%). Again, differences were nonsignificant. In the substudy evaluating DAPT duration in relation to complex PCI, the rate of cardiovascular events at 1 year in those treated with short versus 12-month DAPT was nearly identical (2.53% vs. 2.52%). In the non–complex PCI patients, event rates were nonsignificantly greater on the shortened DAPT regimen (2.38% vs. 1.86%), but the bleeding rate was lower on shortened DAPT whether PCI had been complex (0.63% vs. 1.75%) or not (0.48% vs. 1.22%).
In the absence of any major signal that complex PCI benefited from longer duration DAPT, “complex PCI might not be an appropriate determinant for DAPT durations,” according to Dr. Kimura and coinvestigators.
Study data might not be generalizable
Dr. Greco and Dr. Capodanno pointed out that there are differences between patients and PCI practices in Japan relative to other areas of the world, limiting the generalizability of these findings even if the question is relevant.
“This is an approach that might be suggested for patients at high bleeding risk who have the characteristics of the patients enrolled in the STOPDAPT-2 trials,” Dr. Capodanno said in an interview. In his own PCI practice treating ACS patients, “I would not feel safe enough with clopidogrel monotherapy after only 1 month.”
He considers the ACS population to have a particularly “delicate bleeding-ischemia trade-off,” which is why he thinks this question is relevant and needs to be explored further in additional populations. However, he might design trials differently in his own practice setting. For example, he would at the very least be interested in testing a more potent P2Y12 inhibitor such as ticagrelor when considering a single antiplatelet agent after a limited course of DAPT.
One message from this study is that “bleeding risk trumps PCI complexity,” according to Deepak L. Bhatt, MD, who recently assumed the position of director of Mount Sinai Heart in New York. He liked the approach the investigators took to address a complex and relevant clinical issue, but he also expressed reservations about the clinical applicability of this subgroup analysis.
“We really need more data before uniformly shortening DAPT duration in all patients,” Dr. Bhatt said in an interview. He considers this a hot clinical issue that is likely to generate more trials. He hopes these will provide more definitive evidence of when and how DAPT duration can be reduced. Overall, he anticipates progress toward tailoring therapy in specific populations in order to achieve the best risk-to-benefit balance.
Dr. Kimura has financial relationships with Boston Scientific, Daiichi Sankyo, Sanofi, Terumo, and Abbott Medical Japan, which provided funding for the STOPDAPT-2 and STOPDAPT-2 ACS trials. Dr. Capodanno reported financial relationships with Amgen, Arena, Chiesi, Daiichi Sakyo, Sanofi Aventis, and Terumo. Dr. Bhatt reported financial relationships with more than 20 pharmaceutical companies, including Abbott Medical.
FROM JACC ASIA
Arkansas cardiologist pays $900K to settle false claims allegations
in violation of the False Claims Act.
As part of the settlement, Dr. Tauth will enter into an integrity agreement with the U.S. Department of Health & Human Services, according to a news release from Henry Leventis, U.S. attorney for the Middle District of Tennessee.
“Health care fraud is a top priority of this office. We will aggressively pursue all those who are involved in fraud against government programs,” Mr. Leventis said.
Dr. Tauth formerly treated patients at National Park Medical Center (NPMC) in Hot Springs. The alleged false claims were submitted from September 2013 through August 2019.
The settlement with Dr. Tauth, aged 60, follows a November 2019 voluntary disclosure of the alleged false claims by Brentwood, Tenn.–based Lifepoint Health, which acquired NPMC and Hot Springs Cardiology Associates in November 2018.
NPMC and Hot Springs Cardiology entered into a settlement in October 2020 for the alleged violations and agreed to pay roughly $14.6 million, which includes over $9 million in restitution, according to the news release.
NPMC CEO Scott Smith said NPMC is “committed to maintaining high standards of integrity, legal compliance, and quality care for our patients. We regularly monitor our processes, procedures, and reporting and actively self-report concerns to regulators to ensure we are upholding these standards across our organization.”
“We are proud that our hospital took the appropriate steps to promptly self-report and finalize a settlement with the government for a swift resolution more than 2 years ago,” Mr. Smith said.
Dr. Tauth, however, maintains that the allegations made by NPMC are false.
“I am pleased to have reached a settlement agreement with the Department of Justice regarding allegations brought to them by my former employer, National Park Medical Center,” he said in a statement.
“The settlement agreement specifically states that it is not ‘an admission of liability’ by me, and I remain steadfast in my position that the allegations made by my former employer are false and without merit,” Dr. Tauth added.
He further stated that he has “chosen to enter into the settlement agreement because the legal process initiated by National Park’s allegations has been emotionally and financially damaging to me and my family in the extreme, and a settlement puts an end to the delays, uncertainties, inconveniences, and expenses of protracted litigation. Settlement is in the best interests of my family, my patients, and my medical practice.”
Dr. Tauth said he is “extremely grateful for the support I have received from my patients, medical staff, colleagues, friends, and family during this difficult time, and I look forward to providing high-quality cardiac care in the greater Hot Springs community for many years to come.”
A version of this article first appeared on Medscape.com.
in violation of the False Claims Act.
As part of the settlement, Dr. Tauth will enter into an integrity agreement with the U.S. Department of Health & Human Services, according to a news release from Henry Leventis, U.S. attorney for the Middle District of Tennessee.
“Health care fraud is a top priority of this office. We will aggressively pursue all those who are involved in fraud against government programs,” Mr. Leventis said.
Dr. Tauth formerly treated patients at National Park Medical Center (NPMC) in Hot Springs. The alleged false claims were submitted from September 2013 through August 2019.
The settlement with Dr. Tauth, aged 60, follows a November 2019 voluntary disclosure of the alleged false claims by Brentwood, Tenn.–based Lifepoint Health, which acquired NPMC and Hot Springs Cardiology Associates in November 2018.
NPMC and Hot Springs Cardiology entered into a settlement in October 2020 for the alleged violations and agreed to pay roughly $14.6 million, which includes over $9 million in restitution, according to the news release.
NPMC CEO Scott Smith said NPMC is “committed to maintaining high standards of integrity, legal compliance, and quality care for our patients. We regularly monitor our processes, procedures, and reporting and actively self-report concerns to regulators to ensure we are upholding these standards across our organization.”
“We are proud that our hospital took the appropriate steps to promptly self-report and finalize a settlement with the government for a swift resolution more than 2 years ago,” Mr. Smith said.
Dr. Tauth, however, maintains that the allegations made by NPMC are false.
“I am pleased to have reached a settlement agreement with the Department of Justice regarding allegations brought to them by my former employer, National Park Medical Center,” he said in a statement.
“The settlement agreement specifically states that it is not ‘an admission of liability’ by me, and I remain steadfast in my position that the allegations made by my former employer are false and without merit,” Dr. Tauth added.
He further stated that he has “chosen to enter into the settlement agreement because the legal process initiated by National Park’s allegations has been emotionally and financially damaging to me and my family in the extreme, and a settlement puts an end to the delays, uncertainties, inconveniences, and expenses of protracted litigation. Settlement is in the best interests of my family, my patients, and my medical practice.”
Dr. Tauth said he is “extremely grateful for the support I have received from my patients, medical staff, colleagues, friends, and family during this difficult time, and I look forward to providing high-quality cardiac care in the greater Hot Springs community for many years to come.”
A version of this article first appeared on Medscape.com.
in violation of the False Claims Act.
As part of the settlement, Dr. Tauth will enter into an integrity agreement with the U.S. Department of Health & Human Services, according to a news release from Henry Leventis, U.S. attorney for the Middle District of Tennessee.
“Health care fraud is a top priority of this office. We will aggressively pursue all those who are involved in fraud against government programs,” Mr. Leventis said.
Dr. Tauth formerly treated patients at National Park Medical Center (NPMC) in Hot Springs. The alleged false claims were submitted from September 2013 through August 2019.
The settlement with Dr. Tauth, aged 60, follows a November 2019 voluntary disclosure of the alleged false claims by Brentwood, Tenn.–based Lifepoint Health, which acquired NPMC and Hot Springs Cardiology Associates in November 2018.
NPMC and Hot Springs Cardiology entered into a settlement in October 2020 for the alleged violations and agreed to pay roughly $14.6 million, which includes over $9 million in restitution, according to the news release.
NPMC CEO Scott Smith said NPMC is “committed to maintaining high standards of integrity, legal compliance, and quality care for our patients. We regularly monitor our processes, procedures, and reporting and actively self-report concerns to regulators to ensure we are upholding these standards across our organization.”
“We are proud that our hospital took the appropriate steps to promptly self-report and finalize a settlement with the government for a swift resolution more than 2 years ago,” Mr. Smith said.
Dr. Tauth, however, maintains that the allegations made by NPMC are false.
“I am pleased to have reached a settlement agreement with the Department of Justice regarding allegations brought to them by my former employer, National Park Medical Center,” he said in a statement.
“The settlement agreement specifically states that it is not ‘an admission of liability’ by me, and I remain steadfast in my position that the allegations made by my former employer are false and without merit,” Dr. Tauth added.
He further stated that he has “chosen to enter into the settlement agreement because the legal process initiated by National Park’s allegations has been emotionally and financially damaging to me and my family in the extreme, and a settlement puts an end to the delays, uncertainties, inconveniences, and expenses of protracted litigation. Settlement is in the best interests of my family, my patients, and my medical practice.”
Dr. Tauth said he is “extremely grateful for the support I have received from my patients, medical staff, colleagues, friends, and family during this difficult time, and I look forward to providing high-quality cardiac care in the greater Hot Springs community for many years to come.”
A version of this article first appeared on Medscape.com.
Five thoughts on the Damar Hamlin collapse
The obvious first statement is that it’s neither wise nor appropriate to speculate on the specifics of Damar Hamlin’s cardiac event during a football game on Jan. 2 (including the possibility of commotio cordis) or his ongoing care. The public nature of his collapse induces intense curiosity but people with illness deserve privacy. Privacy in health care is in short supply. I disagree strongly with those who say his doctors ought to be giving public updates. That’s up to the family.
But there are important general concepts to consider about this incident. These include ...
Cardiac arrest can happen to anyone
People with structural heart disease or other chronic illnesses have a higher risk of arrhythmia, but the notion that athletes are immune from cardiac arrest is wrong. This sentence almost seems too obvious to write, but to this day, I hear clinicians express surprise that an athletic person has heart disease.
Survival turns on rapid and effective intervention
In the old days of electrophysiology, we used to test implantable cardioverter-defibrillators during an implant procedure by inducing ventricular fibrillation (VF) and watching the device convert it. Thankfully, trials have shown that this is no longer necessary for most implants.
When you induce VF In the EP lab, you learn quickly that a) it causes loss of consciousness in a matter of seconds, b) rapid defibrillation restores consciousness, often without the patients knowing or remembering they passed out, and c) the failure of the shock to terminate VF results in deterioration in a matter of 1-2 minutes. Even 1 minute in VF feels so long.
Need is an appropriate word in VF treatment
Clinicians often use the verb need. As in, this patient needs this pill or this procedure. It’s rarely appropriate.
But in the case of treating VF, patients truly need rapid defibrillation. Survival of out-of-hospital cardiac arrest is low because there just aren’t enough automated external defibrillators (AEDs) or people trained to use them. A study of patients who had out-of-hospital cardiac arrest in Denmark found that 30-day survival almost doubled (28.8% vs. 16.4%), when the nearest AED was accessible.
Bystanders must act
The public messages are simple: If a person loses consciousness in front of you, and is not breathing normally, assume it is a cardiac arrest, call 911 to get professional help, and start hands-only chest compressions. Don’t spend time checking for a pulse or trying to wake the person. If this is not a cardiac arrest, they will soon tell you to stop compressing their chest. Seconds matter.
Chest compressions are important but what is really needed is defibrillation. A crucial step in CPR is to send someone to get an AED and get the pads attached. If this is a shockable rhythm, deliver the shock. Hamlin’s collapse emphasizes the importance of the AED; without it, his survival to the hospital would have been unlikely.
Widespread preparticipation screening of young athletes remains a bad idea
Whenever cardiac arrest occurs in an athlete, in such a public way, people think about prevention. Surely it is better to prevent such an event than react to it, goes the thinking. The argument against this idea has four prongs:
The incidence of cardiac disease in a young athlete is extremely low, which sets up a situation where most “positive” tests are false positive. A false positive screening ECG or echocardiogram can create harm in multiple ways. One is the risk from downstream procedures, but worse is the inappropriate disqualification from sport. Healthwise, few harms could be greater than creating long-term fear of exercise in someone.
There is also the problem of false-negative screening tests. An ECG may be normal in the setting of hypertrophic cardiomyopathy. And a normal echocardiogram does not exclude arrhythmogenic right ventricular cardiomyopathy or other genetic causes of cardiac arrest. In a 2018 study from a major sports cardiology center in London, 6 of the 8 sudden cardiac deaths in their series were in athletes who had no detectable abnormalities on screening.
Even when disease is found, it’s not clear that prohibiting participation in sports prevents sudden death. Many previous class III recommendations against participation in sport now carry class II – may be considered – designations.
Finally, screening for any disease loses value as treatments improve. Public education regarding rapid intervention with CPR and AED use is the best treatment option. A great example is the case of Christian Erikson, a Danish soccer player who suffered cardiac arrest during a match at the European Championships in 2021 and was rapidly defibrillated on the field. Therapy was so effective that he was conscious and able to wave to fans on his way out of the stadium. He has now returned to elite competition.
Proponents of screening might oppose my take by saying that National Football League players are intensely screened. But this is different from widespread screening of high school and college athletes. It might sound harsh to say, but professional teams have dualities of interests in the health of their athletes given the million-dollar contracts.
What’s more, professional teams can afford to hire expert cardiologists to perform the testing. This would likely reduce the rate of false-positive findings, compared with screening in the community setting. I often have young people referred to me because of asymptomatic bradycardia found during athletic screening – an obviously normal finding.
Conclusions
As long as there are sports, there will be athletes who suffer cardiac arrest.
We can both hope for Hamlin’s full recovery and learn lessons to help reduce the rate of death from out-of-hospital cardiac arrest. This mostly involves education on how to help fellow humans and a public health commitment to access to AEDs.
John Mandrola, MD, practices cardiac electrophysiology in Louisville, Ky. and is a writer and podcaster for Medscape. He has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
The obvious first statement is that it’s neither wise nor appropriate to speculate on the specifics of Damar Hamlin’s cardiac event during a football game on Jan. 2 (including the possibility of commotio cordis) or his ongoing care. The public nature of his collapse induces intense curiosity but people with illness deserve privacy. Privacy in health care is in short supply. I disagree strongly with those who say his doctors ought to be giving public updates. That’s up to the family.
But there are important general concepts to consider about this incident. These include ...
Cardiac arrest can happen to anyone
People with structural heart disease or other chronic illnesses have a higher risk of arrhythmia, but the notion that athletes are immune from cardiac arrest is wrong. This sentence almost seems too obvious to write, but to this day, I hear clinicians express surprise that an athletic person has heart disease.
Survival turns on rapid and effective intervention
In the old days of electrophysiology, we used to test implantable cardioverter-defibrillators during an implant procedure by inducing ventricular fibrillation (VF) and watching the device convert it. Thankfully, trials have shown that this is no longer necessary for most implants.
When you induce VF In the EP lab, you learn quickly that a) it causes loss of consciousness in a matter of seconds, b) rapid defibrillation restores consciousness, often without the patients knowing or remembering they passed out, and c) the failure of the shock to terminate VF results in deterioration in a matter of 1-2 minutes. Even 1 minute in VF feels so long.
Need is an appropriate word in VF treatment
Clinicians often use the verb need. As in, this patient needs this pill or this procedure. It’s rarely appropriate.
But in the case of treating VF, patients truly need rapid defibrillation. Survival of out-of-hospital cardiac arrest is low because there just aren’t enough automated external defibrillators (AEDs) or people trained to use them. A study of patients who had out-of-hospital cardiac arrest in Denmark found that 30-day survival almost doubled (28.8% vs. 16.4%), when the nearest AED was accessible.
Bystanders must act
The public messages are simple: If a person loses consciousness in front of you, and is not breathing normally, assume it is a cardiac arrest, call 911 to get professional help, and start hands-only chest compressions. Don’t spend time checking for a pulse or trying to wake the person. If this is not a cardiac arrest, they will soon tell you to stop compressing their chest. Seconds matter.
Chest compressions are important but what is really needed is defibrillation. A crucial step in CPR is to send someone to get an AED and get the pads attached. If this is a shockable rhythm, deliver the shock. Hamlin’s collapse emphasizes the importance of the AED; without it, his survival to the hospital would have been unlikely.
Widespread preparticipation screening of young athletes remains a bad idea
Whenever cardiac arrest occurs in an athlete, in such a public way, people think about prevention. Surely it is better to prevent such an event than react to it, goes the thinking. The argument against this idea has four prongs:
The incidence of cardiac disease in a young athlete is extremely low, which sets up a situation where most “positive” tests are false positive. A false positive screening ECG or echocardiogram can create harm in multiple ways. One is the risk from downstream procedures, but worse is the inappropriate disqualification from sport. Healthwise, few harms could be greater than creating long-term fear of exercise in someone.
There is also the problem of false-negative screening tests. An ECG may be normal in the setting of hypertrophic cardiomyopathy. And a normal echocardiogram does not exclude arrhythmogenic right ventricular cardiomyopathy or other genetic causes of cardiac arrest. In a 2018 study from a major sports cardiology center in London, 6 of the 8 sudden cardiac deaths in their series were in athletes who had no detectable abnormalities on screening.
Even when disease is found, it’s not clear that prohibiting participation in sports prevents sudden death. Many previous class III recommendations against participation in sport now carry class II – may be considered – designations.
Finally, screening for any disease loses value as treatments improve. Public education regarding rapid intervention with CPR and AED use is the best treatment option. A great example is the case of Christian Erikson, a Danish soccer player who suffered cardiac arrest during a match at the European Championships in 2021 and was rapidly defibrillated on the field. Therapy was so effective that he was conscious and able to wave to fans on his way out of the stadium. He has now returned to elite competition.
Proponents of screening might oppose my take by saying that National Football League players are intensely screened. But this is different from widespread screening of high school and college athletes. It might sound harsh to say, but professional teams have dualities of interests in the health of their athletes given the million-dollar contracts.
What’s more, professional teams can afford to hire expert cardiologists to perform the testing. This would likely reduce the rate of false-positive findings, compared with screening in the community setting. I often have young people referred to me because of asymptomatic bradycardia found during athletic screening – an obviously normal finding.
Conclusions
As long as there are sports, there will be athletes who suffer cardiac arrest.
We can both hope for Hamlin’s full recovery and learn lessons to help reduce the rate of death from out-of-hospital cardiac arrest. This mostly involves education on how to help fellow humans and a public health commitment to access to AEDs.
John Mandrola, MD, practices cardiac electrophysiology in Louisville, Ky. and is a writer and podcaster for Medscape. He has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
The obvious first statement is that it’s neither wise nor appropriate to speculate on the specifics of Damar Hamlin’s cardiac event during a football game on Jan. 2 (including the possibility of commotio cordis) or his ongoing care. The public nature of his collapse induces intense curiosity but people with illness deserve privacy. Privacy in health care is in short supply. I disagree strongly with those who say his doctors ought to be giving public updates. That’s up to the family.
But there are important general concepts to consider about this incident. These include ...
Cardiac arrest can happen to anyone
People with structural heart disease or other chronic illnesses have a higher risk of arrhythmia, but the notion that athletes are immune from cardiac arrest is wrong. This sentence almost seems too obvious to write, but to this day, I hear clinicians express surprise that an athletic person has heart disease.
Survival turns on rapid and effective intervention
In the old days of electrophysiology, we used to test implantable cardioverter-defibrillators during an implant procedure by inducing ventricular fibrillation (VF) and watching the device convert it. Thankfully, trials have shown that this is no longer necessary for most implants.
When you induce VF In the EP lab, you learn quickly that a) it causes loss of consciousness in a matter of seconds, b) rapid defibrillation restores consciousness, often without the patients knowing or remembering they passed out, and c) the failure of the shock to terminate VF results in deterioration in a matter of 1-2 minutes. Even 1 minute in VF feels so long.
Need is an appropriate word in VF treatment
Clinicians often use the verb need. As in, this patient needs this pill or this procedure. It’s rarely appropriate.
But in the case of treating VF, patients truly need rapid defibrillation. Survival of out-of-hospital cardiac arrest is low because there just aren’t enough automated external defibrillators (AEDs) or people trained to use them. A study of patients who had out-of-hospital cardiac arrest in Denmark found that 30-day survival almost doubled (28.8% vs. 16.4%), when the nearest AED was accessible.
Bystanders must act
The public messages are simple: If a person loses consciousness in front of you, and is not breathing normally, assume it is a cardiac arrest, call 911 to get professional help, and start hands-only chest compressions. Don’t spend time checking for a pulse or trying to wake the person. If this is not a cardiac arrest, they will soon tell you to stop compressing their chest. Seconds matter.
Chest compressions are important but what is really needed is defibrillation. A crucial step in CPR is to send someone to get an AED and get the pads attached. If this is a shockable rhythm, deliver the shock. Hamlin’s collapse emphasizes the importance of the AED; without it, his survival to the hospital would have been unlikely.
Widespread preparticipation screening of young athletes remains a bad idea
Whenever cardiac arrest occurs in an athlete, in such a public way, people think about prevention. Surely it is better to prevent such an event than react to it, goes the thinking. The argument against this idea has four prongs:
The incidence of cardiac disease in a young athlete is extremely low, which sets up a situation where most “positive” tests are false positive. A false positive screening ECG or echocardiogram can create harm in multiple ways. One is the risk from downstream procedures, but worse is the inappropriate disqualification from sport. Healthwise, few harms could be greater than creating long-term fear of exercise in someone.
There is also the problem of false-negative screening tests. An ECG may be normal in the setting of hypertrophic cardiomyopathy. And a normal echocardiogram does not exclude arrhythmogenic right ventricular cardiomyopathy or other genetic causes of cardiac arrest. In a 2018 study from a major sports cardiology center in London, 6 of the 8 sudden cardiac deaths in their series were in athletes who had no detectable abnormalities on screening.
Even when disease is found, it’s not clear that prohibiting participation in sports prevents sudden death. Many previous class III recommendations against participation in sport now carry class II – may be considered – designations.
Finally, screening for any disease loses value as treatments improve. Public education regarding rapid intervention with CPR and AED use is the best treatment option. A great example is the case of Christian Erikson, a Danish soccer player who suffered cardiac arrest during a match at the European Championships in 2021 and was rapidly defibrillated on the field. Therapy was so effective that he was conscious and able to wave to fans on his way out of the stadium. He has now returned to elite competition.
Proponents of screening might oppose my take by saying that National Football League players are intensely screened. But this is different from widespread screening of high school and college athletes. It might sound harsh to say, but professional teams have dualities of interests in the health of their athletes given the million-dollar contracts.
What’s more, professional teams can afford to hire expert cardiologists to perform the testing. This would likely reduce the rate of false-positive findings, compared with screening in the community setting. I often have young people referred to me because of asymptomatic bradycardia found during athletic screening – an obviously normal finding.
Conclusions
As long as there are sports, there will be athletes who suffer cardiac arrest.
We can both hope for Hamlin’s full recovery and learn lessons to help reduce the rate of death from out-of-hospital cardiac arrest. This mostly involves education on how to help fellow humans and a public health commitment to access to AEDs.
John Mandrola, MD, practices cardiac electrophysiology in Louisville, Ky. and is a writer and podcaster for Medscape. He has disclosed no relevant financial relationships. A version of this article first appeared on Medscape.com.
STEMI times to treatment usually miss established goals
Therapy initiated within national treatment-time goals set a decade ago for patients with ST-segment elevation myocardial infarction (STEMI) remains associated with improved survival in recent years. But for many such patients, time from first symptoms to initiation of reperfusion therapy still fails to meet those goals, suggests a cross-sectional registry analysis.
For example, patients initially transported to centers with percutaneous coronary intervention (PCI) capability had a median treatment time of 148 minutes, in the analysis spanning the second quarter (Q2) of 2018 to the third quarter (Q3) of 2021. But the goal for centers called for treatment initiation within 90 minutes for at least 75% of such STEMI patients.
Moreover, overall STEMI treatment times and in-hospital mortality rose in tandem significantly from Q2 2018 through the first quarter (Q1) of 2021, which included the first year of the COVID-19 pandemic. Median time to treatment went from 86 minutes to 91 minutes during that period. Meanwhile, in-hospital mortality went from 5.6% to 8.7%, report the study authors led by James G. Jollis, MD, Duke University, Durham, N.C.
Their report, based on 114,871 STEMI patients at 601 US hospitals contributing to the Get With The Guidelines – Coronary Artery Disease registry, was published online in JAMA.
Of those patients, 25,085 had been transferred from non-PCI hospitals, 32,483 were walk-ins, and 57,303 arrived via emergency medical services (EMS). Their median times from symptom onset to PCI were 240, 195, and 148 minutes, respectively.
In-hospital mortality was significantly reduced in an adjusted analysis for patients treated within target times, compared with those whose treatment missed the time goals, regardless of whether they were transported by EMS, walked into a hospital with on-site PCI, or were transferred from a non-PCI center (Table 1).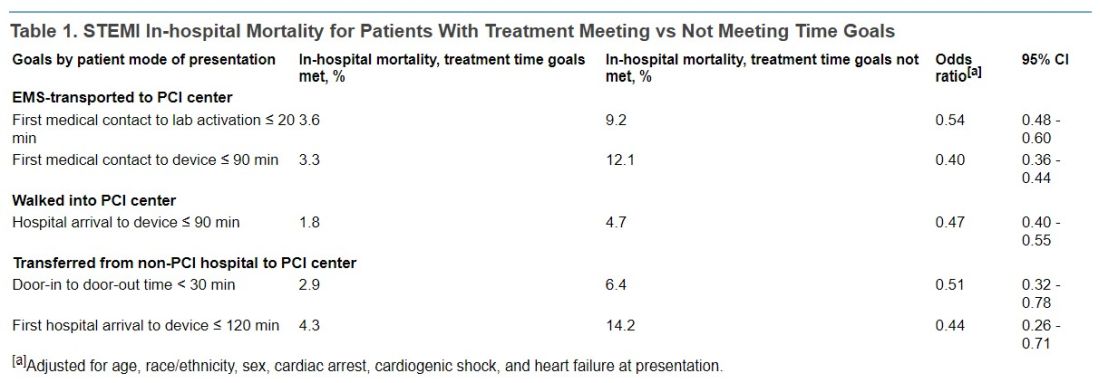
Regardless of mode of patient presentation, treatment time goals were not met most of the time, the group reports. Patients who required interhospital transfer experienced the longest system delays; only 17% were treated within 120 minutes.
Among patients who received primary PCI, 20% had a registry-defined hospital-specified reason for delay, including cardiac arrest and/or need for intubation in 6.8%, “difficulty crossing the culprit lesion” in 3.8%, and “other reasons” in 5.8%, the group reports.
“In 2020, a new reason for delay was added to the registry, ‘need for additional personal protective equipment for suspected/confirmed infectious disease.’ This reason was most commonly used in the second quarter of 2020 (6%) and then declined over time to 1% in the final 2 quarters,” they write.
“Thus, active SARS-CoV-2 infection appeared to have a smaller direct role in longer treatment times or worse outcomes.” Rather, they continue, “the pandemic potentially had a significant indirect role as hospitals were overwhelmed with patients, EMS and hospitals were challenged in maintaining paramedic and nurse staffing and intensive care bed availability, and patients experienced delayed care due to barriers to access or perceived fear of becoming entangled in an overwhelmed medical system.”
Still an important quality metric
STEMI treatment times remain an important quality metric to which hospitals should continue to pay attention because shorter times improve patient care, Deepak Bhatt, MD, MPH, told this news organization.
“Having said that, as with all metrics, one needs to be thoughtful and realize that a difference of a couple of minutes is probably not a crucial thing,” said Dr. Bhatt, Brigham and Women’s Hospital and Harvard Medical School, Boston, who was not involved with the current study.
Interhospital transfers indeed involve longer delays, he observed, suggesting that regional integrated health systems should develop methods for optimizing STEMI care – even, for example, if they involve bypassing non-PCI centers or stopping patients briefly for stabilization followed by rapid transport to a PCI-capable facility.
“That, of course, requires cooperation among hospitals. Sometimes that requires hospitals putting aside economic considerations and just focusing on doing the right thing for that individual patient,” Dr. Bhatt said.
Transfer delays are common for patients presenting with STEMI at hospitals without PCI capability, he noted. “Having clear protocols in place that expedite that type of transfer, I think, could go a long way in reducing the time to treatment in patients that are presenting to the hospital without cath labs. That’s an important message that these data provide.”
The onset of COVID-19 led to widespread delays in STEMI time to treatment early in the pandemic. There were concerns about exposing cath lab personnel to SARS-CoV-2 and potential adverse consequences of sick personnel being unable to provide patient care in the subsequent weeks and months, Dr. Bhatt observed.
However, “All of that seems to have quieted down, and I don’t think COVID is impacting time to treatment right now.”
‘Suboptimal compliance’ with standards
The current findings of “suboptimal compliance with national targets underscore why reassessing quality metrics, in light of changing practice patterns and other secular trends, is critical,” write Andrew S. Oseran, MD, MBA, and Robert W. Yeh, MD, both of Harvard Medical School, in an accompanying editorial.
“While the importance of coordinated and expeditious care for this high-risk patient population is undeniable, the specific actions that hospitals can – or should – take to further improve overall STEMI outcomes are less clear,” they say.
“As physicians contemplate the optimal path forward in managing the care of STEMI patients, they must recognize the clinical and operational nuance that exists in caring for this diverse population and acknowledge the trade-offs associated with uniform quality metrics,” write the editorialists.
“Global reductions in time to treatment for STEMI patients has been one of health care’s great success stories. As we move forward, it may be time to consider whether efforts to achieve additional improvement in target treatment times will result in substantive benefits, or whether we have reached the point of diminishing returns.”
A version of this article first appeared on Medscape.com.
Therapy initiated within national treatment-time goals set a decade ago for patients with ST-segment elevation myocardial infarction (STEMI) remains associated with improved survival in recent years. But for many such patients, time from first symptoms to initiation of reperfusion therapy still fails to meet those goals, suggests a cross-sectional registry analysis.
For example, patients initially transported to centers with percutaneous coronary intervention (PCI) capability had a median treatment time of 148 minutes, in the analysis spanning the second quarter (Q2) of 2018 to the third quarter (Q3) of 2021. But the goal for centers called for treatment initiation within 90 minutes for at least 75% of such STEMI patients.
Moreover, overall STEMI treatment times and in-hospital mortality rose in tandem significantly from Q2 2018 through the first quarter (Q1) of 2021, which included the first year of the COVID-19 pandemic. Median time to treatment went from 86 minutes to 91 minutes during that period. Meanwhile, in-hospital mortality went from 5.6% to 8.7%, report the study authors led by James G. Jollis, MD, Duke University, Durham, N.C.
Their report, based on 114,871 STEMI patients at 601 US hospitals contributing to the Get With The Guidelines – Coronary Artery Disease registry, was published online in JAMA.
Of those patients, 25,085 had been transferred from non-PCI hospitals, 32,483 were walk-ins, and 57,303 arrived via emergency medical services (EMS). Their median times from symptom onset to PCI were 240, 195, and 148 minutes, respectively.
In-hospital mortality was significantly reduced in an adjusted analysis for patients treated within target times, compared with those whose treatment missed the time goals, regardless of whether they were transported by EMS, walked into a hospital with on-site PCI, or were transferred from a non-PCI center (Table 1).
Regardless of mode of patient presentation, treatment time goals were not met most of the time, the group reports. Patients who required interhospital transfer experienced the longest system delays; only 17% were treated within 120 minutes.
Among patients who received primary PCI, 20% had a registry-defined hospital-specified reason for delay, including cardiac arrest and/or need for intubation in 6.8%, “difficulty crossing the culprit lesion” in 3.8%, and “other reasons” in 5.8%, the group reports.
“In 2020, a new reason for delay was added to the registry, ‘need for additional personal protective equipment for suspected/confirmed infectious disease.’ This reason was most commonly used in the second quarter of 2020 (6%) and then declined over time to 1% in the final 2 quarters,” they write.
“Thus, active SARS-CoV-2 infection appeared to have a smaller direct role in longer treatment times or worse outcomes.” Rather, they continue, “the pandemic potentially had a significant indirect role as hospitals were overwhelmed with patients, EMS and hospitals were challenged in maintaining paramedic and nurse staffing and intensive care bed availability, and patients experienced delayed care due to barriers to access or perceived fear of becoming entangled in an overwhelmed medical system.”
Still an important quality metric
STEMI treatment times remain an important quality metric to which hospitals should continue to pay attention because shorter times improve patient care, Deepak Bhatt, MD, MPH, told this news organization.
“Having said that, as with all metrics, one needs to be thoughtful and realize that a difference of a couple of minutes is probably not a crucial thing,” said Dr. Bhatt, Brigham and Women’s Hospital and Harvard Medical School, Boston, who was not involved with the current study.
Interhospital transfers indeed involve longer delays, he observed, suggesting that regional integrated health systems should develop methods for optimizing STEMI care – even, for example, if they involve bypassing non-PCI centers or stopping patients briefly for stabilization followed by rapid transport to a PCI-capable facility.
“That, of course, requires cooperation among hospitals. Sometimes that requires hospitals putting aside economic considerations and just focusing on doing the right thing for that individual patient,” Dr. Bhatt said.
Transfer delays are common for patients presenting with STEMI at hospitals without PCI capability, he noted. “Having clear protocols in place that expedite that type of transfer, I think, could go a long way in reducing the time to treatment in patients that are presenting to the hospital without cath labs. That’s an important message that these data provide.”
The onset of COVID-19 led to widespread delays in STEMI time to treatment early in the pandemic. There were concerns about exposing cath lab personnel to SARS-CoV-2 and potential adverse consequences of sick personnel being unable to provide patient care in the subsequent weeks and months, Dr. Bhatt observed.
However, “All of that seems to have quieted down, and I don’t think COVID is impacting time to treatment right now.”
‘Suboptimal compliance’ with standards
The current findings of “suboptimal compliance with national targets underscore why reassessing quality metrics, in light of changing practice patterns and other secular trends, is critical,” write Andrew S. Oseran, MD, MBA, and Robert W. Yeh, MD, both of Harvard Medical School, in an accompanying editorial.
“While the importance of coordinated and expeditious care for this high-risk patient population is undeniable, the specific actions that hospitals can – or should – take to further improve overall STEMI outcomes are less clear,” they say.
“As physicians contemplate the optimal path forward in managing the care of STEMI patients, they must recognize the clinical and operational nuance that exists in caring for this diverse population and acknowledge the trade-offs associated with uniform quality metrics,” write the editorialists.
“Global reductions in time to treatment for STEMI patients has been one of health care’s great success stories. As we move forward, it may be time to consider whether efforts to achieve additional improvement in target treatment times will result in substantive benefits, or whether we have reached the point of diminishing returns.”
A version of this article first appeared on Medscape.com.
Therapy initiated within national treatment-time goals set a decade ago for patients with ST-segment elevation myocardial infarction (STEMI) remains associated with improved survival in recent years. But for many such patients, time from first symptoms to initiation of reperfusion therapy still fails to meet those goals, suggests a cross-sectional registry analysis.
For example, patients initially transported to centers with percutaneous coronary intervention (PCI) capability had a median treatment time of 148 minutes, in the analysis spanning the second quarter (Q2) of 2018 to the third quarter (Q3) of 2021. But the goal for centers called for treatment initiation within 90 minutes for at least 75% of such STEMI patients.
Moreover, overall STEMI treatment times and in-hospital mortality rose in tandem significantly from Q2 2018 through the first quarter (Q1) of 2021, which included the first year of the COVID-19 pandemic. Median time to treatment went from 86 minutes to 91 minutes during that period. Meanwhile, in-hospital mortality went from 5.6% to 8.7%, report the study authors led by James G. Jollis, MD, Duke University, Durham, N.C.
Their report, based on 114,871 STEMI patients at 601 US hospitals contributing to the Get With The Guidelines – Coronary Artery Disease registry, was published online in JAMA.
Of those patients, 25,085 had been transferred from non-PCI hospitals, 32,483 were walk-ins, and 57,303 arrived via emergency medical services (EMS). Their median times from symptom onset to PCI were 240, 195, and 148 minutes, respectively.
In-hospital mortality was significantly reduced in an adjusted analysis for patients treated within target times, compared with those whose treatment missed the time goals, regardless of whether they were transported by EMS, walked into a hospital with on-site PCI, or were transferred from a non-PCI center (Table 1).
Regardless of mode of patient presentation, treatment time goals were not met most of the time, the group reports. Patients who required interhospital transfer experienced the longest system delays; only 17% were treated within 120 minutes.
Among patients who received primary PCI, 20% had a registry-defined hospital-specified reason for delay, including cardiac arrest and/or need for intubation in 6.8%, “difficulty crossing the culprit lesion” in 3.8%, and “other reasons” in 5.8%, the group reports.
“In 2020, a new reason for delay was added to the registry, ‘need for additional personal protective equipment for suspected/confirmed infectious disease.’ This reason was most commonly used in the second quarter of 2020 (6%) and then declined over time to 1% in the final 2 quarters,” they write.
“Thus, active SARS-CoV-2 infection appeared to have a smaller direct role in longer treatment times or worse outcomes.” Rather, they continue, “the pandemic potentially had a significant indirect role as hospitals were overwhelmed with patients, EMS and hospitals were challenged in maintaining paramedic and nurse staffing and intensive care bed availability, and patients experienced delayed care due to barriers to access or perceived fear of becoming entangled in an overwhelmed medical system.”
Still an important quality metric
STEMI treatment times remain an important quality metric to which hospitals should continue to pay attention because shorter times improve patient care, Deepak Bhatt, MD, MPH, told this news organization.
“Having said that, as with all metrics, one needs to be thoughtful and realize that a difference of a couple of minutes is probably not a crucial thing,” said Dr. Bhatt, Brigham and Women’s Hospital and Harvard Medical School, Boston, who was not involved with the current study.
Interhospital transfers indeed involve longer delays, he observed, suggesting that regional integrated health systems should develop methods for optimizing STEMI care – even, for example, if they involve bypassing non-PCI centers or stopping patients briefly for stabilization followed by rapid transport to a PCI-capable facility.
“That, of course, requires cooperation among hospitals. Sometimes that requires hospitals putting aside economic considerations and just focusing on doing the right thing for that individual patient,” Dr. Bhatt said.
Transfer delays are common for patients presenting with STEMI at hospitals without PCI capability, he noted. “Having clear protocols in place that expedite that type of transfer, I think, could go a long way in reducing the time to treatment in patients that are presenting to the hospital without cath labs. That’s an important message that these data provide.”
The onset of COVID-19 led to widespread delays in STEMI time to treatment early in the pandemic. There were concerns about exposing cath lab personnel to SARS-CoV-2 and potential adverse consequences of sick personnel being unable to provide patient care in the subsequent weeks and months, Dr. Bhatt observed.
However, “All of that seems to have quieted down, and I don’t think COVID is impacting time to treatment right now.”
‘Suboptimal compliance’ with standards
The current findings of “suboptimal compliance with national targets underscore why reassessing quality metrics, in light of changing practice patterns and other secular trends, is critical,” write Andrew S. Oseran, MD, MBA, and Robert W. Yeh, MD, both of Harvard Medical School, in an accompanying editorial.
“While the importance of coordinated and expeditious care for this high-risk patient population is undeniable, the specific actions that hospitals can – or should – take to further improve overall STEMI outcomes are less clear,” they say.
“As physicians contemplate the optimal path forward in managing the care of STEMI patients, they must recognize the clinical and operational nuance that exists in caring for this diverse population and acknowledge the trade-offs associated with uniform quality metrics,” write the editorialists.
“Global reductions in time to treatment for STEMI patients has been one of health care’s great success stories. As we move forward, it may be time to consider whether efforts to achieve additional improvement in target treatment times will result in substantive benefits, or whether we have reached the point of diminishing returns.”
A version of this article first appeared on Medscape.com.
FROM JAMA
Top cardiology societies call for revamp of clinical trials
Leading cardiology societies have issued a “call for action” on a global scale to reinvent randomized clinical trials fit for the 21st century.
“Randomized trials are an essential tool for reliably assessing the effects of treatments, but they have become too costly and too burdensome,” first author Louise Bowman, University of Oxford, England, told this news organization. “We urgently need to modernize our approach to clinical trials in order to continue to improve patient care.”
The joint opinion is from the European Society of Cardiology, the American Heart Association, the American College of Cardiology, and the World Heart Federation. It was simultaneously published online in the European Heart Journal, Circulation, Journal of the American College of Cardiology, and Global Heart.
The authors note that the availability of large-scale “real-world” data is increasingly being touted as a way to bypass the challenges of conducting randomized trials. Yet, observational analyses of real-world data “are not a suitable alternative to randomization,” Prof. Bowman said.
Cardiology has historically led the way in transforming clinical practice with groundbreaking “mega-trials,” such as the International Study of Infarct Survival (ISIS), Gruppo Italiano per lo Studio della Streptochinasi nell’Infarto (GISSI), and Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries (GUSTO).
But over the past 25 years, there has been a huge increase in the rules and related bureaucracy governing clinical trials, which hinders the ability to conduct trials swiftly and affordably, the authors point out.
The COVID-19 pandemic has shown that important clinical trials can be performed quickly and efficiently in busy hospitals, they note.
“The RECOVERY trial in COVID-19 has been an excellent example of this, with results that are estimated to have saved around 1 million lives worldwide within just 1 year,” Prof. Bowman told this news organization.
A Good Clinical Trials Collaborative made up of key stakeholders recently developed new guidelines designed to promote better, more efficient randomized controlled trials.
“If widely adopted and used alongside valuable 21st century electronic health records, we could transform the clinical trials landscape and do many more high-quality trials very cost-effectively,” Prof. Bowman said.
“Widespread adoption and implementation of the revised guidelines will require collaboration with a wide range of national and international organizations, including patient, professional, academic, and industry groups, funders and government organizations, and ethics, health policy, and regulatory bodies,” Prof. Bowman acknowledged.
“This is work that the Good Clinical Trials Collaborative is leading. It is hoped that this endorsement by the joint cardiovascular societies will increase awareness and provide valuable support to his important work,” she added.
No commercial funding was received. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Leading cardiology societies have issued a “call for action” on a global scale to reinvent randomized clinical trials fit for the 21st century.
“Randomized trials are an essential tool for reliably assessing the effects of treatments, but they have become too costly and too burdensome,” first author Louise Bowman, University of Oxford, England, told this news organization. “We urgently need to modernize our approach to clinical trials in order to continue to improve patient care.”
The joint opinion is from the European Society of Cardiology, the American Heart Association, the American College of Cardiology, and the World Heart Federation. It was simultaneously published online in the European Heart Journal, Circulation, Journal of the American College of Cardiology, and Global Heart.
The authors note that the availability of large-scale “real-world” data is increasingly being touted as a way to bypass the challenges of conducting randomized trials. Yet, observational analyses of real-world data “are not a suitable alternative to randomization,” Prof. Bowman said.
Cardiology has historically led the way in transforming clinical practice with groundbreaking “mega-trials,” such as the International Study of Infarct Survival (ISIS), Gruppo Italiano per lo Studio della Streptochinasi nell’Infarto (GISSI), and Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries (GUSTO).
But over the past 25 years, there has been a huge increase in the rules and related bureaucracy governing clinical trials, which hinders the ability to conduct trials swiftly and affordably, the authors point out.
The COVID-19 pandemic has shown that important clinical trials can be performed quickly and efficiently in busy hospitals, they note.
“The RECOVERY trial in COVID-19 has been an excellent example of this, with results that are estimated to have saved around 1 million lives worldwide within just 1 year,” Prof. Bowman told this news organization.
A Good Clinical Trials Collaborative made up of key stakeholders recently developed new guidelines designed to promote better, more efficient randomized controlled trials.
“If widely adopted and used alongside valuable 21st century electronic health records, we could transform the clinical trials landscape and do many more high-quality trials very cost-effectively,” Prof. Bowman said.
“Widespread adoption and implementation of the revised guidelines will require collaboration with a wide range of national and international organizations, including patient, professional, academic, and industry groups, funders and government organizations, and ethics, health policy, and regulatory bodies,” Prof. Bowman acknowledged.
“This is work that the Good Clinical Trials Collaborative is leading. It is hoped that this endorsement by the joint cardiovascular societies will increase awareness and provide valuable support to his important work,” she added.
No commercial funding was received. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Leading cardiology societies have issued a “call for action” on a global scale to reinvent randomized clinical trials fit for the 21st century.
“Randomized trials are an essential tool for reliably assessing the effects of treatments, but they have become too costly and too burdensome,” first author Louise Bowman, University of Oxford, England, told this news organization. “We urgently need to modernize our approach to clinical trials in order to continue to improve patient care.”
The joint opinion is from the European Society of Cardiology, the American Heart Association, the American College of Cardiology, and the World Heart Federation. It was simultaneously published online in the European Heart Journal, Circulation, Journal of the American College of Cardiology, and Global Heart.
The authors note that the availability of large-scale “real-world” data is increasingly being touted as a way to bypass the challenges of conducting randomized trials. Yet, observational analyses of real-world data “are not a suitable alternative to randomization,” Prof. Bowman said.
Cardiology has historically led the way in transforming clinical practice with groundbreaking “mega-trials,” such as the International Study of Infarct Survival (ISIS), Gruppo Italiano per lo Studio della Streptochinasi nell’Infarto (GISSI), and Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries (GUSTO).
But over the past 25 years, there has been a huge increase in the rules and related bureaucracy governing clinical trials, which hinders the ability to conduct trials swiftly and affordably, the authors point out.
The COVID-19 pandemic has shown that important clinical trials can be performed quickly and efficiently in busy hospitals, they note.
“The RECOVERY trial in COVID-19 has been an excellent example of this, with results that are estimated to have saved around 1 million lives worldwide within just 1 year,” Prof. Bowman told this news organization.
A Good Clinical Trials Collaborative made up of key stakeholders recently developed new guidelines designed to promote better, more efficient randomized controlled trials.
“If widely adopted and used alongside valuable 21st century electronic health records, we could transform the clinical trials landscape and do many more high-quality trials very cost-effectively,” Prof. Bowman said.
“Widespread adoption and implementation of the revised guidelines will require collaboration with a wide range of national and international organizations, including patient, professional, academic, and industry groups, funders and government organizations, and ethics, health policy, and regulatory bodies,” Prof. Bowman acknowledged.
“This is work that the Good Clinical Trials Collaborative is leading. It is hoped that this endorsement by the joint cardiovascular societies will increase awareness and provide valuable support to his important work,” she added.
No commercial funding was received. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Survival varies widely for cardiac arrests in U.S. cath labs
The chance of surviving a cardiac arrest varies widely across hospitals in the United States, even when the arrest occurs in the highly controlled setting of a cardiac catheterization lab, a new study indicates.
Among 4,787 patients who arrested in the cath lab at 231 hospitals in the Get With The Guidelines (GWTG) Resuscitation registry, only about one-third survived to discharge. The median risk-adjusted survival rate (RASR) for all hospitals was 36%.
When stratified by RASR tertiles, however, median survival rates were 20%, 36%, and 52% for hospitals in the lowest, middle, and highest tertiles.
The odds of survival differed by 71% in similar patients presenting at two randomly selected hospitals (median odds ratio, 1.71; 95% confidence interval, 1.52-1.87).
“The good news is that cardiac arrests in the cath lab are relatively infrequent, but the bad news is that they still occur and the outcomes are, in general, pretty dismal,” senior author Deepak L. Bhatt, MD, MPH, said in an interview. “So anything that we can do as hospitals [and] health care systems to improve the care of these patients could go a long way.”
He noted that data are sparse on cardiac arrests in the cath lab but that recent studies examining in-hospital arrests in the same registry report lower hospital-wide survival rates, between 17% and 24%.
Nevertheless, the current study included only those hospitals motivated and with the resources to participate in the American Heart Association’s voluntary GWTG Resuscitation registry between January 2003 and December 2017.
“It probably does provide the best case scenario of what’s going on and, if we included every hospital in the United States or the world, probably the outcomes would be substantially worse,” said Dr. Bhatt, who was recently named director of Mount Sinai Heart and the first Dr. Valentin Fuster Professor of Cardiovascular Medicine, New York.
The results were published in JACC Cardiovascular Interventions.
Hospital and patient factors
Possible explanations for the wide disparity in survival are the small number of cardiac arrests in the cath lab, the increasing complexity of cases, and the fact that patients are often very sick and may experience a problem during a procedure, or both, Dr. Bhatt suggested. Cath labs also vary in how they handle resuscitative efforts and access to advanced mechanical support devices, such as extracorporeal membrane oxygenation (ECMO).
“It’s not available in every cath lab and, even in hospitals that have it, they may not have a given ECMO circuit available at the exact time the patient’s having a cardiac arrest,” he said. “That’s one example of something that can make, in my opinion, a big difference in whether a patient lives or dies if they’re having a cardiac arrest but may not always be easily deployed.”
When the investigators looked specifically at hospital-level factors, only yearly volume of cardiac arrests in the cath lab was significantly associated with risk-adjusted survival (P < .01), whereas hospital size, rural or urban setting, teaching status, and geographic location were not.
In multivariate adjusted analyses, factors associated with survival to discharge included age (OR, 0.78), Black race (OR, 0.68), respiratory insufficiency (OR, 0.75), and initial cardiac arrest rhythm (OR, 3.32).
The median hospital RASR was 27% higher for ventricular tachycardia or ventricular fibrillation arrests than for arrests with a nonshockable rhythm of asystole and pulseless electrical activity (55% vs. 28%).
Notably, hospitals in the lowest tertile of risk-adjusted survival rates had a higher prevalence of non-White patients, renal and respiratory comorbidity, and arrest with nonshockable rhythm.
“We want to make sure as we’re contemplating whether to resuscitate a patient or how aggressively to resuscitate, that we aren’t letting any of our own biases, whether they have to do with race or potentially sex and gender, interfere with more objective assessments of whether the patient can in fact be saved or not,” Dr. Bhatt said.
Reached for comment, Srihari S. Naidu, MD, who chaired the writing group for the Society for Cardiovascular Angiography and Interventions’ (SCAI) consensus statement on cardiogenic shock and co-authored its document on best practices in the cardiac cath lab, said the findings show that survival in the cath lab is higher than that seen in-hospital. “Still, there’s a lot of room for improvement,” he said.
He was particularly struck by the variability in survival. “Underprivileged individuals, so those who are non-White populations and have respiratory and renal problems, they seem to have a worse survival and that makes sense – patients with comorbidities – but it feeds into the issue of, ‘Are we treating our population similarly in terms of their baseline race and ethnicity as a gap in care?’ ”
Better survival at hospitals with high volumes likely reflects more experience in handling these events, a rapid response and personnel to help with resuscitation, and overall better critical care and cath lab environment, said Dr. Naidu, director of the cardiac cath lab at Westchester Medical Center and professor of medicine at New York Medical College, both in Valhalla, N.Y.
“So that leads into two things,” he said. “One is that probably we should be working on having all high-risk patients go to centers of excellence. So, for example, [for] patients in shock, patients with STEMI, regionalization of care to the high-volume cath labs that are experienced in cardiac arrest and critical care management may be a way to go.”
“Second, if experience counts, can that experience be simulated through drills and simulations in the cath lab?” Dr. Naidu said. “Should all cath labs have drills where we have a cardiac arrest patient, and how would we respond to that? Who’s going to do the compressions? Where’s the mechanical support device? What are the things we need to have a seamless cardiac arrest protocol for arrests during the cath lab?”
Dr. Bhatt and colleagues acknowledge that despite adjustment for many key variables, the study lacked procedural details that may affect survival and information related to resuscitation efforts.
“We really do need to focus more research efforts, potentially more in the way of quality-improvement efforts, to try and help patients get these sorts of patients who are in dire straits to the cath lab but hopefully also through the hospital discharge and back home,” Dr. Bhatt said.
In an editorial accompanying the study, Matthew L. Tomey, MD, Icahn School of Medicine at Mount Sinai, New York, writes that the “findings and limitations of this study together sound a call to action.”
He also signaled the need for more research and for registries and reporting instruments to capture variables particular to in-laboratory cardiac arrest and resuscitation in the cardiac cath lab. “A necessary first step is the development of consensus data elements for supplemental reporting in cases of ILCA,” such as indication for cath lab presentation, timing of arrest relative to procedure, and cause of arrest.
Dr. Bhatt reported numerous relationships with industry. Dr. Naidu and Dr. Tomey report having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The chance of surviving a cardiac arrest varies widely across hospitals in the United States, even when the arrest occurs in the highly controlled setting of a cardiac catheterization lab, a new study indicates.
Among 4,787 patients who arrested in the cath lab at 231 hospitals in the Get With The Guidelines (GWTG) Resuscitation registry, only about one-third survived to discharge. The median risk-adjusted survival rate (RASR) for all hospitals was 36%.
When stratified by RASR tertiles, however, median survival rates were 20%, 36%, and 52% for hospitals in the lowest, middle, and highest tertiles.
The odds of survival differed by 71% in similar patients presenting at two randomly selected hospitals (median odds ratio, 1.71; 95% confidence interval, 1.52-1.87).
“The good news is that cardiac arrests in the cath lab are relatively infrequent, but the bad news is that they still occur and the outcomes are, in general, pretty dismal,” senior author Deepak L. Bhatt, MD, MPH, said in an interview. “So anything that we can do as hospitals [and] health care systems to improve the care of these patients could go a long way.”
He noted that data are sparse on cardiac arrests in the cath lab but that recent studies examining in-hospital arrests in the same registry report lower hospital-wide survival rates, between 17% and 24%.
Nevertheless, the current study included only those hospitals motivated and with the resources to participate in the American Heart Association’s voluntary GWTG Resuscitation registry between January 2003 and December 2017.
“It probably does provide the best case scenario of what’s going on and, if we included every hospital in the United States or the world, probably the outcomes would be substantially worse,” said Dr. Bhatt, who was recently named director of Mount Sinai Heart and the first Dr. Valentin Fuster Professor of Cardiovascular Medicine, New York.
The results were published in JACC Cardiovascular Interventions.
Hospital and patient factors
Possible explanations for the wide disparity in survival are the small number of cardiac arrests in the cath lab, the increasing complexity of cases, and the fact that patients are often very sick and may experience a problem during a procedure, or both, Dr. Bhatt suggested. Cath labs also vary in how they handle resuscitative efforts and access to advanced mechanical support devices, such as extracorporeal membrane oxygenation (ECMO).
“It’s not available in every cath lab and, even in hospitals that have it, they may not have a given ECMO circuit available at the exact time the patient’s having a cardiac arrest,” he said. “That’s one example of something that can make, in my opinion, a big difference in whether a patient lives or dies if they’re having a cardiac arrest but may not always be easily deployed.”
When the investigators looked specifically at hospital-level factors, only yearly volume of cardiac arrests in the cath lab was significantly associated with risk-adjusted survival (P < .01), whereas hospital size, rural or urban setting, teaching status, and geographic location were not.
In multivariate adjusted analyses, factors associated with survival to discharge included age (OR, 0.78), Black race (OR, 0.68), respiratory insufficiency (OR, 0.75), and initial cardiac arrest rhythm (OR, 3.32).
The median hospital RASR was 27% higher for ventricular tachycardia or ventricular fibrillation arrests than for arrests with a nonshockable rhythm of asystole and pulseless electrical activity (55% vs. 28%).
Notably, hospitals in the lowest tertile of risk-adjusted survival rates had a higher prevalence of non-White patients, renal and respiratory comorbidity, and arrest with nonshockable rhythm.
“We want to make sure as we’re contemplating whether to resuscitate a patient or how aggressively to resuscitate, that we aren’t letting any of our own biases, whether they have to do with race or potentially sex and gender, interfere with more objective assessments of whether the patient can in fact be saved or not,” Dr. Bhatt said.
Reached for comment, Srihari S. Naidu, MD, who chaired the writing group for the Society for Cardiovascular Angiography and Interventions’ (SCAI) consensus statement on cardiogenic shock and co-authored its document on best practices in the cardiac cath lab, said the findings show that survival in the cath lab is higher than that seen in-hospital. “Still, there’s a lot of room for improvement,” he said.
He was particularly struck by the variability in survival. “Underprivileged individuals, so those who are non-White populations and have respiratory and renal problems, they seem to have a worse survival and that makes sense – patients with comorbidities – but it feeds into the issue of, ‘Are we treating our population similarly in terms of their baseline race and ethnicity as a gap in care?’ ”
Better survival at hospitals with high volumes likely reflects more experience in handling these events, a rapid response and personnel to help with resuscitation, and overall better critical care and cath lab environment, said Dr. Naidu, director of the cardiac cath lab at Westchester Medical Center and professor of medicine at New York Medical College, both in Valhalla, N.Y.
“So that leads into two things,” he said. “One is that probably we should be working on having all high-risk patients go to centers of excellence. So, for example, [for] patients in shock, patients with STEMI, regionalization of care to the high-volume cath labs that are experienced in cardiac arrest and critical care management may be a way to go.”
“Second, if experience counts, can that experience be simulated through drills and simulations in the cath lab?” Dr. Naidu said. “Should all cath labs have drills where we have a cardiac arrest patient, and how would we respond to that? Who’s going to do the compressions? Where’s the mechanical support device? What are the things we need to have a seamless cardiac arrest protocol for arrests during the cath lab?”
Dr. Bhatt and colleagues acknowledge that despite adjustment for many key variables, the study lacked procedural details that may affect survival and information related to resuscitation efforts.
“We really do need to focus more research efforts, potentially more in the way of quality-improvement efforts, to try and help patients get these sorts of patients who are in dire straits to the cath lab but hopefully also through the hospital discharge and back home,” Dr. Bhatt said.
In an editorial accompanying the study, Matthew L. Tomey, MD, Icahn School of Medicine at Mount Sinai, New York, writes that the “findings and limitations of this study together sound a call to action.”
He also signaled the need for more research and for registries and reporting instruments to capture variables particular to in-laboratory cardiac arrest and resuscitation in the cardiac cath lab. “A necessary first step is the development of consensus data elements for supplemental reporting in cases of ILCA,” such as indication for cath lab presentation, timing of arrest relative to procedure, and cause of arrest.
Dr. Bhatt reported numerous relationships with industry. Dr. Naidu and Dr. Tomey report having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The chance of surviving a cardiac arrest varies widely across hospitals in the United States, even when the arrest occurs in the highly controlled setting of a cardiac catheterization lab, a new study indicates.
Among 4,787 patients who arrested in the cath lab at 231 hospitals in the Get With The Guidelines (GWTG) Resuscitation registry, only about one-third survived to discharge. The median risk-adjusted survival rate (RASR) for all hospitals was 36%.
When stratified by RASR tertiles, however, median survival rates were 20%, 36%, and 52% for hospitals in the lowest, middle, and highest tertiles.
The odds of survival differed by 71% in similar patients presenting at two randomly selected hospitals (median odds ratio, 1.71; 95% confidence interval, 1.52-1.87).
“The good news is that cardiac arrests in the cath lab are relatively infrequent, but the bad news is that they still occur and the outcomes are, in general, pretty dismal,” senior author Deepak L. Bhatt, MD, MPH, said in an interview. “So anything that we can do as hospitals [and] health care systems to improve the care of these patients could go a long way.”
He noted that data are sparse on cardiac arrests in the cath lab but that recent studies examining in-hospital arrests in the same registry report lower hospital-wide survival rates, between 17% and 24%.
Nevertheless, the current study included only those hospitals motivated and with the resources to participate in the American Heart Association’s voluntary GWTG Resuscitation registry between January 2003 and December 2017.
“It probably does provide the best case scenario of what’s going on and, if we included every hospital in the United States or the world, probably the outcomes would be substantially worse,” said Dr. Bhatt, who was recently named director of Mount Sinai Heart and the first Dr. Valentin Fuster Professor of Cardiovascular Medicine, New York.
The results were published in JACC Cardiovascular Interventions.
Hospital and patient factors
Possible explanations for the wide disparity in survival are the small number of cardiac arrests in the cath lab, the increasing complexity of cases, and the fact that patients are often very sick and may experience a problem during a procedure, or both, Dr. Bhatt suggested. Cath labs also vary in how they handle resuscitative efforts and access to advanced mechanical support devices, such as extracorporeal membrane oxygenation (ECMO).
“It’s not available in every cath lab and, even in hospitals that have it, they may not have a given ECMO circuit available at the exact time the patient’s having a cardiac arrest,” he said. “That’s one example of something that can make, in my opinion, a big difference in whether a patient lives or dies if they’re having a cardiac arrest but may not always be easily deployed.”
When the investigators looked specifically at hospital-level factors, only yearly volume of cardiac arrests in the cath lab was significantly associated with risk-adjusted survival (P < .01), whereas hospital size, rural or urban setting, teaching status, and geographic location were not.
In multivariate adjusted analyses, factors associated with survival to discharge included age (OR, 0.78), Black race (OR, 0.68), respiratory insufficiency (OR, 0.75), and initial cardiac arrest rhythm (OR, 3.32).
The median hospital RASR was 27% higher for ventricular tachycardia or ventricular fibrillation arrests than for arrests with a nonshockable rhythm of asystole and pulseless electrical activity (55% vs. 28%).
Notably, hospitals in the lowest tertile of risk-adjusted survival rates had a higher prevalence of non-White patients, renal and respiratory comorbidity, and arrest with nonshockable rhythm.
“We want to make sure as we’re contemplating whether to resuscitate a patient or how aggressively to resuscitate, that we aren’t letting any of our own biases, whether they have to do with race or potentially sex and gender, interfere with more objective assessments of whether the patient can in fact be saved or not,” Dr. Bhatt said.
Reached for comment, Srihari S. Naidu, MD, who chaired the writing group for the Society for Cardiovascular Angiography and Interventions’ (SCAI) consensus statement on cardiogenic shock and co-authored its document on best practices in the cardiac cath lab, said the findings show that survival in the cath lab is higher than that seen in-hospital. “Still, there’s a lot of room for improvement,” he said.
He was particularly struck by the variability in survival. “Underprivileged individuals, so those who are non-White populations and have respiratory and renal problems, they seem to have a worse survival and that makes sense – patients with comorbidities – but it feeds into the issue of, ‘Are we treating our population similarly in terms of their baseline race and ethnicity as a gap in care?’ ”
Better survival at hospitals with high volumes likely reflects more experience in handling these events, a rapid response and personnel to help with resuscitation, and overall better critical care and cath lab environment, said Dr. Naidu, director of the cardiac cath lab at Westchester Medical Center and professor of medicine at New York Medical College, both in Valhalla, N.Y.
“So that leads into two things,” he said. “One is that probably we should be working on having all high-risk patients go to centers of excellence. So, for example, [for] patients in shock, patients with STEMI, regionalization of care to the high-volume cath labs that are experienced in cardiac arrest and critical care management may be a way to go.”
“Second, if experience counts, can that experience be simulated through drills and simulations in the cath lab?” Dr. Naidu said. “Should all cath labs have drills where we have a cardiac arrest patient, and how would we respond to that? Who’s going to do the compressions? Where’s the mechanical support device? What are the things we need to have a seamless cardiac arrest protocol for arrests during the cath lab?”
Dr. Bhatt and colleagues acknowledge that despite adjustment for many key variables, the study lacked procedural details that may affect survival and information related to resuscitation efforts.
“We really do need to focus more research efforts, potentially more in the way of quality-improvement efforts, to try and help patients get these sorts of patients who are in dire straits to the cath lab but hopefully also through the hospital discharge and back home,” Dr. Bhatt said.
In an editorial accompanying the study, Matthew L. Tomey, MD, Icahn School of Medicine at Mount Sinai, New York, writes that the “findings and limitations of this study together sound a call to action.”
He also signaled the need for more research and for registries and reporting instruments to capture variables particular to in-laboratory cardiac arrest and resuscitation in the cardiac cath lab. “A necessary first step is the development of consensus data elements for supplemental reporting in cases of ILCA,” such as indication for cath lab presentation, timing of arrest relative to procedure, and cause of arrest.
Dr. Bhatt reported numerous relationships with industry. Dr. Naidu and Dr. Tomey report having no relevant financial relationships.
A version of this article first appeared on Medscape.com.



