User login
Bronchiolitis and Discharge Criteria
Although bronchiolitis is the leading cause of hospitalization for US infants,[1] there is a lack of basic prospective data about the expected inpatient clinical course and ongoing uncertainty about when a hospitalized child is ready for discharge to home.[2] This lack of data about children's readiness for discharge may result in variable hospital length‐of‐stay (LOS).[3, 4, 5]
One specific source of variability in discharge readiness and LOS variability may be the lack of consensus about safe threshold oxygen saturation values for discharge in children hospitalized with bronchiolitis.[6, 7] In 2006, the Scottish Intercollegiate Guidelines Network recommended a discharge room air oxygen (RAO2) saturation threshold of 95%.[8] The same year, the American Academy of Pediatrics (AAP) bronchiolitis clinical practice guideline stated that oxygen is not needed for children with RAO2 saturations 90% who are feeding well and have minimal respiratory distress.[9] There is a need for prospective studies to help clinicians make evidenced‐based discharge decisions for this common condition.
We performed a prospective, multicenter, multiyear study[10, 11, 12] to examine the typical inpatient clinical course of and to develop hospital discharge guidelines for children age <2 years hospitalized with bronchiolitis. We hypothesized that children would not worsen clinically and would be safe to discharge home once their respiratory status improved and they were able to remain hydrated.
METHODS
Study Design and Population
We conducted a prospective, multicenter cohort study for 3 consecutive years during the 2007 to 2010 winter seasons, as part of the Multicenter Airway Research Collaboration (MARC), a program of the Emergency Medicine Network (
All patients were treated at the discretion of the treating physician. Inclusion criteria were an attending physician's diagnosis of bronchiolitis, age <2 years, and the ability of the parent/guardian to give informed consent. The exclusion criteria were previous enrollment and transfer to a participating hospital >48 hours after the original admission time. Therefore, children with comorbid conditions were included in this study. All consent and data forms were translated into Spanish. The institutional review board at each of the 16 participating hospitals approved the study.
Of the 2207 enrolled children, we excluded 109 (5%) children with a hospital LOS <1 day due to inadequate time to capture the required data for the present analysis. Among the 2098 remaining children, 1916 (91%) had daily inpatient data on all factors used to define clinical improvement and clinical worsening. Thus, the analytic cohort was comprised of 1916 children hospitalized for bronchiolitis.
Data Collection
Investigators conducted detailed structured interviews. Chart reviews were conducted to obtain preadmission and daily hospital clinical data including respiratory rates, daily respiratory rate trends, degree of retractions, oxygen saturation, daily oxygen saturation trends, medical management, and disposition. These data were manually reviewed, and site investigators were queried about missing data and discrepancies. A follow‐up telephone interview was conducted with families 1 week after discharge to examine relapse events at both 24 hours and 7 days.
We used the question: How long ago did the following symptoms [eg, difficulty breathing] begin [for the] current illness? to estimate the onset of the current illness. Pulse was categorized as low, normal, or high based on age‐related heart rate values.[13] Presence of apnea was recorded daily by site investigators.[14]
Nasopharyngeal Aspirate Collection and Virology Testing
As described previously, site teams used a standardized protocol to collect nasopharyngeal aspirates,[11] which were tested for respiratory syncytial virus (RSV) types A and B; rhinovirus (RV); parainfluenza virus types 1, 2, and 3; influenza virus types A and B; 2009 novel H1N1; human metapneumovirus; coronaviruses NL‐63, HKU1, OC43, and 229E; enterovirus, and adenovirus using polymerase chain reaction.[11, 15, 16, 17]
Defining Clinical Improvement and Worsening
Clinical improvement criteria were based on the 2006 AAP guidelines.[9] For respiratory rate and oxygen saturation, clinicians estimated average daily respiratory rate and oxygen saturation based on the recorded readings from the previous 24 hours. This estimation reflects the process clinicians use when rounding on their hospitalized patients, and thus may be more similar to standard clinical practice than a calculated mean. The respiratory rate criteria are adjusted for age.[18, 19] For daily estimated average oxygen saturation we used the AAP criteria of RAO2 saturation of 90%. Considering that oxygen saturation is the main determinant of LOS,[20] healthy infants age <6 months may have transient oxygen saturations of around 80%,[21] and that errors in estimation may occur, we included a lowest RAO2 of 88% in our improvement criteria. By combining the dichotomized estimated oxygen saturation (90% or not) with the lower limit of 88%, there was little room for erroneous conclusions. A child was considered clinically improved on the earliest date he/she met all of the following criteria: (1) none or mild retractions and improved or stable retractions compared with the previous inpatient day; (2) daily estimated average respiratory rate (RR) <60 breaths per minute for age <6 months, <55 breaths/minute for age 6 to 11 months, and <45 breaths/minute for age 12 months with a decreasing or stable trend over the course of the current day; (3) daily estimated average RAO2 saturation 90%, lowest RAO2 saturation 88%[21]; and (4) not receiving intravenous (IV) fluids or for children receiving IV fluids a clinician report of the child maintaining oral hydration. Children who reached the clinical improvement criteria were considered clinically worse if they required intensive care or had the inverse of 1 of the improvement criteria: moderate/severe retractions that were worse compared with the previous inpatient day, daily average RR 60 with an increasing trend over the current day, need for oxygen, or need for IV fluids.
Statistical Analyses
All analyses were performed using Stata 12.0 (StataCorp, College Station, TX). Data are presented as proportions with 95% confidence intervals (95% CIs), means with standard deviations, and medians with interquartile ranges (IQR). To examine potential factors associated with clinical worsening after reaching clinical improvement, we used 2, Fisher exact, Student t test, and Kruskall‐Wallis tests, as appropriate.
Adjusted analyses used generalized linear mixed models with a logit link to identify independent risk factors for worsening after reaching clinical improvement. Fixed effects for patient‐level factors and a random site effect were used. Factors were tested for inclusion in the multivariable model if they were found to be associated with worsening in unadjusted analyses (P<0.20) or were considered clinically important. Results are reported as odds ratios with 95% CIs.
We performed several sensitivity analyses to evaluate these improvement criteria: (1) we excluded the lowest RAO2 saturation requirement of 88%, (2) we examined a 94% daily estimated average RAO2 saturation threshold,[22] (3) we examined a 95% daily estimated average RAO2 saturation threshold,[8] and (4) we examined children age <12 months with no history of wheeze.
RESULTS
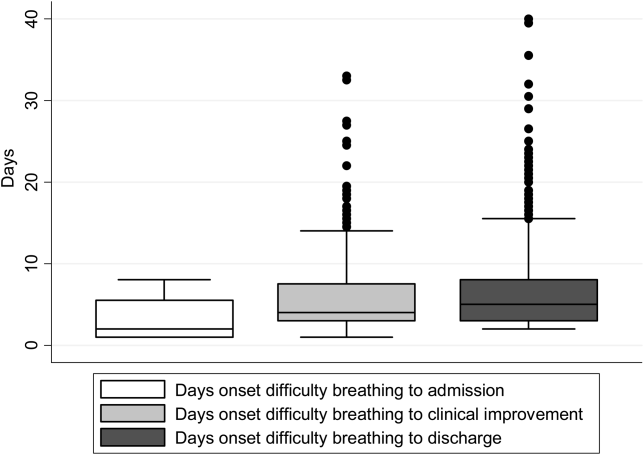
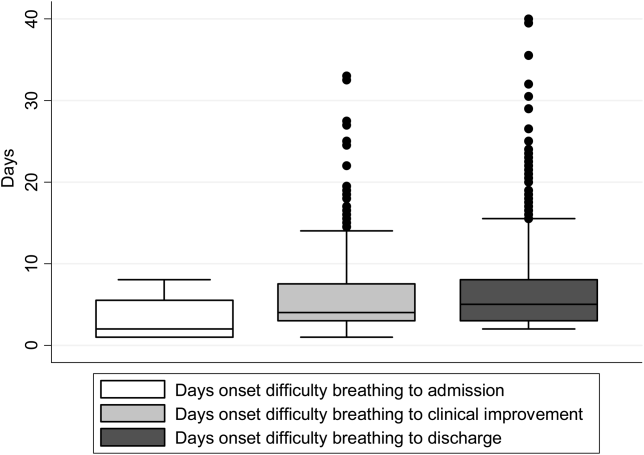
There were 1916 children hospitalized with bronchiolitis with data on all factors used to define clinical improvement and clinical worsening. The median number of days from the beginning of difficulty breathing until admission was 2 days (IQR, 15.5 days; range, 18 days) and from the beginning of difficulty breathing until clinical improvement was 4 days (IQR, 37.5 days; range, 133 days) (Figure 1). The variance for days to admission was significantly less than the variance for days to clinical improvement (P<0.001).
In this observational study, clinicians discharged 214 (11%) of the 1916 children before meeting the definition of clinical improvement. Thus, 1702 (89%; 95% CI: 87%‐90%) children reached the clinical improvement criteria, had a LOS >1 day, and had data on all factors (Figure 2).
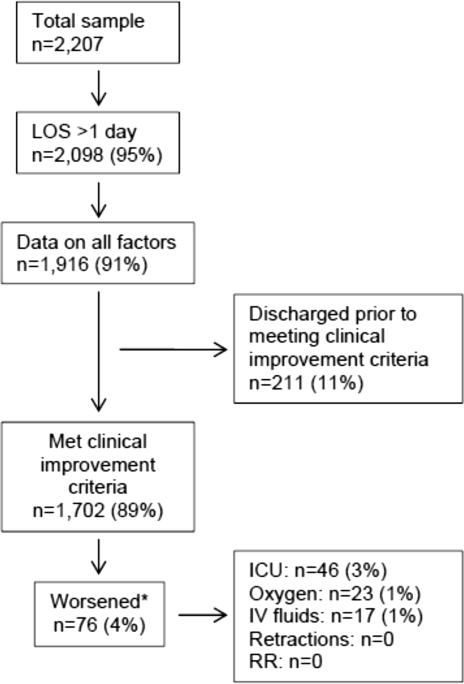
Of the 1702 children who met the clinical improvement criteria, there were 76 children (4%; 95% CI: 3%5%) who worsened (Figure 2). The worsening occurred within a median of 1 day (IQR, 13 days) of clinical improvement. Forty‐six (3%) of the children required transfer to the ICU (1 required intubation, 1 required continuous positive airway pressure, and 4 had apnea), 23 (1%) required oxygen, and 17 (1%) required IV fluids. Eight percent of children met multiple criteria for worsening. A comparison between children who did and did not worsen is shown in Table 1. In general, children who worsened after improvement were younger and born earlier. These children also presented in more severe respiratory distress, had moderate or severe retractions, oxygen saturation <85% at hospitalization, inadequate oral intake, and apnea documented during the hospitalization. Neither viral etiology nor site of care influenced whether the children worsened after improving. However, stratified analysis of children based on initial location of admission (ie, ICU or ward) showed that among the children admitted to the ICU from the emergency department (ED), 89% met the improvement criteria and 19% clinically worsened. In contrast, among children admitted to the ward from the ED, 89% met the improvement criteria, and only 2% clinically worsened. Stratified multivariable models based on the initial location of admission from the ED (ie, ICU or ward) were not possible due to small sample sizes after stratification. None of these children had relapse events requiring rehospitalization within either 24 hours or 7 days of discharge.
| Did Not Worsen, n=1,626 | Worsened, n=76 | P Value | |
|---|---|---|---|
| |||
| Demographic characteristics | |||
| Age <2 months, % | 29 | 57 | <0.001 |
| Month of birth, % | 0.02 | ||
| OctoberMarch | 61 | 75 | |
| AprilSeptember | 39 | 25 | |
| Sex, % | 0.51 | ||
| Male | 59 | 55 | |
| Female | 41 | 45 | |
| Race, % | 0.050 | ||
| White | 63 | 58 | |
| Black | 23 | 34 | |
| Other or missing | 14 | 8 | |
| Hispanic ethnicity, % | 37 | 22 | 0.01 |
| Insurance, % | 0.87 | ||
| Nonprivate | 68 | 67 | |
| Private | 32 | 33 | |
| Medical history | |||
| Gestational age <37 weeks, % | 23 | 39 | 0.002 |
| Birth weight, % | 0.52 | ||
| <5 lbs | 13 | 12 | |
| 5 lbs | 34 | 41 | |
| 7 lbs | 53 | 47 | |
| Mother's age, median (IQR) | 27 (2333) | 27 (2233) | 0.54 |
| Is or was breastfed, % | 61 | 51 | 0.10 |
| Smoked during pregnancy, % | 15 | 20 | 0.22 |
| Exposure to smoke, % | 13 | 20 | 0.11 |
| Family history of asthma, % | 0.89 | ||
| Neither parent | 68 | 64 | |
| Either mother or father | 27 | 30 | |
| Both parents | 4 | 4 | |
| Do not know/missing | 2 | 1 | |
| History of wheezing, % | 23 | 17 | 0.24 |
| History of eczema, % | 16 | 7 | 0.04 |
| History of intubation, % | 9 | 12 | 0.50 |
| Major, relevant, comorbid medical disorder, % | 20 | 24 | 0.46 |
| Current illness | |||
| When difficulty breathing began, preadmission, % | 0.63 | ||
| 1 day | 70 | 75 | |
| <1 day | 28 | 23 | |
| No difficulty preadmission | 2 | 3 | |
| Weight, lbs, median (IQR) | 12.3 (8.817.4) | 9.0 (6.613.2) | 0.001 |
| Temperature, F, median (IQR) | 99.5 (98.6100.6) | 99.4 (98.1100.4) | 0.06 |
| Pulse, beats per minute by age | 0.82 | ||
| Low | 0.3 | 0 | |
| Normal | 48 | 46 | |
| High | 51 | 54 | |
| Respiratory rate, breaths per minute, median (IQR) | 48 (4060) | 48 (3864) | 0.28 |
| Retractions, % | 0.001 | ||
| None | 22 | 25 | |
| Mild | 43 | 24 | |
| Moderate | 26 | 33 | |
| Severe | 4 | 12 | |
| Missing | 5 | 7 | |
| Oxygen saturation by pulse oximetry or ABG, % | 0.001 | ||
| <85 | 4 | 12 | |
| 8587.9 | 3 | 4 | |
| 8889.9 | 5 | 0 | |
| 9093.9 | 18 | 11 | |
| 94 | 72 | 73 | |
| Oral intake, % | <0.001 | ||
| Adequate | 45 | 22 | |
| Inadequate | 42 | 63 | |
| Missing | 13 | 14 | |
| Presence of apnea, % | 7 | 24 | <0.001 |
| RSV‐A, % | 44 | 41 | 0.54 |
| RSV‐B, % | 30 | 25 | 0.36 |
| HRV, % | 24 | 24 | 0.88 |
| Chest x‐ray results during ED/preadmission visit | |||
| Atelectasis | 12 | 13 | 0.77 |
| Infiltrate | 13 | 11 | 0.50 |
| Hyperinflated | 18 | 21 | 0.47 |
| Peribronchial cuffing/thickening | 23 | 17 | 0.32 |
| Normal | 14 | 16 | 0.75 |
| White blood count, median (IQR) | 11.2 (8.714.4) | 11.9 (9.214.4) | 0.60 |
| Platelet count, median (IQR) | 395 (317490) | 430 (299537) | 0.56 |
| Sodium, median (IQR) | 138 (136140) | 137 (135138) | 0.19 |
| Hospital length of stay, median (IQR) | 2 (14) | 4.5 (28) | <0.001 |
| One‐week follow‐up | |||
| Relapse within 24 hours of hospital discharge requiring hospital admission, % | 0.5 | 0 | 0.56 |
| Relapse within 7 days of hospital discharge requiring hospital admission, % | 1 | 0 | 0.35 |
On multivariable analysis (Table 2), independent risk factors for worsening after reaching the clinical improvement criteria were young age, preterm birth, and presenting to care with more severe bronchiolitis represented by severe retractions, inadequate oral intake, or apnea. To further evaluate the improvement criteria in the current analysis, multiple sensitivity analyses were conducted. The frequency of clinical worsening after reaching the improvement criteria was stable when we examined different RA02 criteria in sensitivity analyses: (1) excluding RA02 as a criterion for improvement: 90% met improvement criteria and 4% experienced clinical worsening, (2) changing the average RA02 threshold for clinical improvement to 94%: 62% met improvement criteria and 6% experienced clinical worsening, and (3) changing the average RA02 threshold for clinical improvement to 95%: 47% met improvement criteria and 5% experienced clinical worsening. Furthermore, stratifying by age <2 months and restricting to more stringent definitions of bronchiolitis (ie, age <1 year or age <1 year+no history of wheezing) also did not materially change the results (see Supporting Figure 1 in the online version of this article).
| Odds Ratio | 95% CI | P Value | |
|---|---|---|---|
| |||
| Age <2 months | 3.51 | 2.07‐5.94 | <0.001 |
| Gestational age <37 weeks | 1.94 | 1.13‐3.32 | 0.02 |
| Retractions | |||
| None | 1.30 | 0.80‐3.23 | 0.19 |
| Mild | 1.0 | Reference | |
| Moderate | 1.91 | 0.99‐3.71 | 0.06 |
| Severe | 5.55 | 2.1214.50 | <0.001 |
| Missing | 1.70 | 0.53‐5.42 | 0.37 |
| Oral intake | |||
| Adequate | 1.00 | Reference | |
| Inadequate | 2.54 | 1.39‐4.62 | 0.002 |
| Unknown/missing | 1.88 | 0.79‐4.44 | 0.15 |
| Presence of apnea | 2.87 | 1.45‐5.68 | 0.003 |
We compared the 214 children who were discharged prior to reaching clinical improvement with the 1702 children who reached the clinical improvement criteria. The 214 children were less likely to be age <2 months (22% vs 30%; P=0.02). These 2 groups (214 vs 1702) were similar with respect to severe retractions (2% vs 4%; P=0.13), median respiratory rate (48 vs 48; P=0.42), oxygen saturation <90% (15% vs 11%; P=0.07), inadequate oral intake (50% vs 43%; P=0.13), and rates of relapse events requiring rehospitalization within both 24 hours (0.6% vs 0.6%; P=0.88) and 7 days (1% vs 1%; P=0.90) of discharge.
DISCUSSION
In this large, multicenter, multiyear study of children hospitalized with bronchiolitis, we found that children present to a hospital in a relatively narrow time frame, but their time to recovery in the hospital is highly variable. Nonetheless, 96% of children continued to improve once they had: (1) improving or stable retractions rated as none/mild, (2) a decreasing or stable RR by age, (3) estimated average RAO2 saturation 90% and lowest RAO2 saturation of 88%, and (4) were hydrated. The 4% of children who worsened after clinically improving were more likely to be age <2 months, born <37 weeks, and present with more severe distress (ie, severe retractions, inadequate oral intake, or apnea). Based on the low risk of worsening after clinical improvement, especially among children admitted to the regular ward (2%), we believe these 4 clinical criteria could be used as discharge criteria for this common pediatric illness with a predominantly monophasic clinical course.
Variability in hospital LOS for children with bronchiolitis exists in the United States[3] and internationally.[4, 5] Cheung and colleagues analyzed administrative data from over 75,000 children admitted for bronchiolitis in England between April 2007 and March 2010 and found sixfold variation in LOS between sites. They concluded that this LOS variability was due in part to providers' clinical decision making.[5] Srivastava and colleagues[23] addressed variable clinician decision making in bronchiolitis and 10 other common pediatric conditions by embedding discharge criteria developed by expert consensus into admission order sets. They found that for children with bronchiolitis, the embedded discharge criteria reduced the median LOS from 1.91 to 1.87 days. In contrast to the single‐center data presented by White and colleagues,[24] the prospective, multicenter MARC‐30 data provide a clear understanding of the normal clinical course for children hospitalized with bronchiolitis, determine if children clinically worsen after clinical improvement, and provide data about discharge criteria for children hospitalized with bronchiolitis. Although there is a lack of rigorous published data, the lower tract symptoms of bronchiolitis (eg, cough, retractions) are said to peak on days 5 to 7 of illness and then gradually resolve.[25] In the present study, we found that the time from the onset of difficulty breathing until hospital admission is less variable than the time from the onset of difficulty breathing until either clinical improvement or discharge. Although 75% of children have clinically improved within 7.5 days of difficulty breathing based on the IQR results, the remaining 25% may have a more prolonged recovery in the hospital of up to 3 weeks. Interestingly, prolonged recovery times from bronchiolitis have also been noted in children presenting to the ED[26] and in an outpatient population.[27] It is unclear why 20% to 25% of children at different levels of severity of illness have prolonged recovery from bronchiolitis, but this group of children requires further investigation.
Given the variability of recovery times, clinicians may have difficulty knowing when a child is ready for hospital discharge. One of the main stumbling blocks for discharge readiness in children with bronchiolitis is the interpretation of the oxygen saturation value.[6, 8, 9, 20, 28] However, it should be considered that interpreting the oxygen saturation in a child who is clinically improving in the hospital setting is different than interpreting the oxygen saturation of a child in the ED or the clinic whose clinical course is less certain.[22] In the hospital setting, using the oxygen saturation value in in the AAP guideline,[9] 4% of children clinically worsened after they met the improvement criteria, a clinical pattern observed previously with supplemental oxygen.[28] This unpredictability may explain some of the variation in providers' clinical decision making.[5] The children who worsened, and therefore deserve more cautious discharge planning, were young (<2 months), premature (<37 weeks gestational age), and presented in more severe distress. Those children admitted to the ICU from the ED worsened more commonly than children admitted to the ward (19% vs 2%). Interestingly, the viral etiology of the child's bronchiolitis did not influence whether a child worsened after reaching the improvement criteria. Therefore, although children with RV bronchiolitis have a shorter hospital LOS than children with RSV bronchiolitis,[11] the pattern of recovery did not differ by viral etiology.
In addition to unsafe discharges, clinicians may be concerned about the possibility of readmissions. Although somewhat controversial, hospital readmission is being used as a quality of care metric.[29, 30, 31] One response to minimize readmissions would be for clinicians to observe children for longer than clinically indicated.[32] However, shorter LOS is not necessarily associated with increased readmission rates.[33] Given that the geometric mean of hospital charges per child with bronchiolitis increased from $6380 in 2000 to $8530 in 2009,[34] the potential for safely reducing hospital LOS by using the discharge criteria proposed in the current study instead of other criteria[8] may net substantial cost savings. Furthermore, reducing LOS would decrease the time children expose others to these respiratory viruses and possibly reduce medical errors.[35]
Our study has some potential limitations. Because the study participants were all hospitalized, these data do not inform admission or discharge decisions from either the ED or the clinic; but other data address those clinical scenarios.[22] Also, the 16 sites that participated in this study were large, urban teaching hospitals. Consequently, these results are not necessarily generalizable to smaller community hospitals. Although numerous data points were required to enter the analytic cohort, only 9% of the sample was excluded for missing data. There were 214 children who did not meet our improvement criteria by the time of discharge. Although the inability to include these children in the analysis may be seen as a limitation, this practice variability underscores the need for more data about discharging hospitalized children with bronchiolitis. Last, site teams reviewed medical records daily. More frequent recording of the clinical course would have yielded more granular data, but the current methodology replicates how data are generally presented during patient care rounds, when decisions about suitability for discharge are often considered.
CONCLUSION
We documented in this large multicenter study that most children hospitalized with bronchiolitis had a wide range of time to recovery, but the vast majority continued to improve once they reached the identified clinical criteria that predict a safe discharge to home. The children who worsened after clinical improvement were more likely to be younger, premature infants presenting in more severe distress. Although additional prospective validation of these hospital discharge criteria is warranted, these data may help clinicians make more evidence‐based discharge decisions for a common pediatric illness with high practice variation, both in the United States[3] and in other countries.[4, 5]
Acknowledgements
Collaborators in the MARC‐30 Study: Besh Barcega, MD, Loma Linda University Children's Hospital, Loma Linda, CA; John Cheng, MD, Children's Healthcare of Atlanta at Egleston, Atlanta, GA; Dorothy Damore, MD, New York Presbyterian Hospital‐Cornell, New York, NY; Carlos Delgado, MD, Children's Healthcare of Atlanta at Egleston, Atlanta, GA; Haitham Haddad, MD, Rainbow Babies & Children's Hospital, Cleveland, OH; Paul Hain, MD, Monroe Carell Jr. Children's Hospital at Vanderbilt, Nashville, TN; Frank LoVecchio, DO, Maricopa Medical Center, Phoenix, AZ; Charles Macias, MD MPH, Texas Children's Hospital, Houston, TX; Jonathan Mansbach, MD, MPH, Boston Children's Hospital, Boston, MA; Eugene Mowad, MD, Akron Children's Hospital, Akron, OH; Brian Pate, MD, Children's Mercy Hospital, Kansas City, MO; Mark Riederer, MD, Monroe Carell Jr. Children's Hospital at Vanderbilt, Nashville, TN; M. Jason Sanders, MD, Children's Memorial Hermann Hospital, Houston, TX; Alan R. Schroeder, MD, Santa Clara Valley Medical Center, San Jose, CA; Nikhil Shah, MD, New York Presbyterian Hospital‐Cornell, New York, NY; Michelle Stevenson, MD, MS, Kosair Children's Hospital, Louisville, KY; Erin Stucky Fisher, MD, Rady Children's Hospital, San Diego, CA; Stephen Teach, MD, MPH, Children's National Medical Center, Washington, DC; Lisa Zaoutis, MD, Children's Hospital of Philadelphia, Philadelphia, PA.
Disclosures: This study was supported by grants U01 AI‐67693 and K23 AI‐77801 from the National Institutes of Health (Bethesda, MD). The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Allergy and Infectious Diseases or the National Institutes of Health. Drs. Mansbach and Piedra have provided consultation to Regeneron Pharmaceuticals. Otherwise, no authors report any potential conflicts of interest, including relevant financial interests, activities, relationships, and affiliations.
- , , , , . Infectious disease hospitalizations among infants in the United States. Pediatrics. 2008;121(2):244–252.
- . “A hospital is no place to be sick” Samuel Goldwyn (1882–1974). Arch Dis Child. 2009;94(8):565–566.
- , , , , , Variation in inpatient diagnostic testing and management of bronchiolitis. Pediatrics. 2005;115(4):878–884.
- , , , International variation in the management of infants hospitalized with respiratory syncytial virus. International RSV Study Group. Eur J Pediatr. 1998;157(3):215–220.
- , , , , . Population variation in admission rates and duration of inpatient stay for bronchiolitis in England. Arch Dis Child. 2013;98(1):57–59.
- , , , . Impact of pulse oximetry and oxygen therapy on length of stay in bronchiolitis hospitalizations. Arch Pediatr Adolesc Med. 2004;158(6):527–530.
- , , . Pulse oximetry in pediatric practice. Pediatrics. 2011;128(4):740–752.
- Scottish Intercollegiate Guidelines Network. Bronchiolitis in children (SIGN 91). In: NHS Quality Improvement Scotland. Edinburgh, Scotland: Scottish Intercollegiate Guidelines Network; 2006.
- , , , et al. Diagnosis and management of bronchiolitis. Pediatrics. 2006;118(4):1774–1793.
- , , , et al. Prospective multicenter study of children with bronchiolitis requiring mechanical ventilation. Pediatrics. 2012;130(3):e492–e500.
- , , , et al. Prospective multicenter study of viral etiology and hospital length of stay in children with severe bronchiolitis. Arch Pediatr Adolesc Med. 2012;166(8):700–706.
- , , , et al. Apnea in children hospitalized with bronchiolitis. Pediatrics. 2013;132(5):e1194–e1201.
- . Evaluation of the cardiovascular system: history and physical evaluation. In: Kliegman RM, Stanton BF, St. Geme JW III, Schor NF, Behrman RF, eds. Nelson Textbook of Pediatrics. Philadelphia, PA: Elsevier Saunders; 2011:1529–1536.
- , , , et al. Apnea in children hospitalized with bronchiolitis. Pediatrics. 2013;132(5):e1194–e1201.
- , , , et al. Respiratory viral infections in patients with chronic, obstructive pulmonary disease. J Infect. 2005;50(4):322–330.
- , , , . Evaluation of real‐time PCR for diagnosis of Bordetella pertussis infection. BMC Infect Dis. 2006;6:62.
- , , , , . Evaluation of three real‐time PCR assays for detection of Mycoplasma pneumoniae in an outbreak investigation. J Clin Microbiol. 2008;46(9):3116–3118.
- , , , et al. Normal ranges of heart rate and respiratory rate in children from birth to 18 years of age: a systematic review of observational studies. Lancet. 2011;377(9770):1011–1018.
- , , , , , . Development of heart and respiratory rate percentile curves for hospitalized children. Pediatrics. 2013;131(4):e1150–e1157.
- , . Effect of oxygen supplementation on length of stay for infants hospitalized with acute viral bronchiolitis. Pediatrics. 2008;121(3):470–475.
- , , , et al. Longitudinal assessment of hemoglobin oxygen saturation in healthy infants during the first 6 months of age. Collaborative Home Infant Monitoring Evaluation (CHIME) Study Group. J Pediatr. 1999;135(5):580–586.
- , , , et al. Prospective multicenter study of bronchiolitis: predicting safe discharges from the emergency department. Pediatrics. 2008;121(4):680–688.
- , , , et al. Delays in discharge in a tertiary care pediatric hospital. J Hosp Med. 2009;4(8):481–485.
- , , , et al. Using quality improvement to optimise paediatric discharge efficiency. BMJ Qual Saf. 2014;23(5):428–436.
- , . Bronchiolitis in infants and children: treatment; outcome; and prevention. In: Torchia M, ed. UpToDate. Alphen aan den Rijn, the Netherlands; Wolters Kluwer Health; 2013.
- , . Duration of illness in infants with bronchiolitis evaluated in the emergency department. Pediatrics. 2010;126(2):285–290.
- , , . Duration of illness in ambulatory children diagnosed with bronchiolitis. Arch Pediatr Adolesc Med. 2000;154(10):997–1000.
- , , , et al. A clinical pathway for bronchiolitis is effective in reducing readmission rates. J Pediatr. 2005;147(5):622–626.
- , , , et al. Measuring hospital quality using pediatric readmission and revisit rates. Pediatrics. 2013;132(3):429–436.
- , , , et al. Pediatric readmission prevalence and variability across hospitals. JAMA. 2013;309(4):372–380.
- , , , , , . Preventability of early readmissions at a children's hospital. Pediatrics. 2013;131(1):e171–e181.
- , . Hospital readmission: quality indicator or statistical inevitability? Pediatrics. 2013;132(3):569–570.
- , , , et al. Children's hospitals with shorter lengths of stay do not have higher readmission rates. J Pediatr. 2013;163(4):1034–1038.e1.
- , , , , . Trends in bronchiolitis hospitalizations in the United States, 2000–2009. Pediatrics. 2013;132(1):28–36.
- , , , . Preventable adverse events in infants hospitalized with bronchiolitis. Pediatrics. 2005;116(3):603–608.
Although bronchiolitis is the leading cause of hospitalization for US infants,[1] there is a lack of basic prospective data about the expected inpatient clinical course and ongoing uncertainty about when a hospitalized child is ready for discharge to home.[2] This lack of data about children's readiness for discharge may result in variable hospital length‐of‐stay (LOS).[3, 4, 5]
One specific source of variability in discharge readiness and LOS variability may be the lack of consensus about safe threshold oxygen saturation values for discharge in children hospitalized with bronchiolitis.[6, 7] In 2006, the Scottish Intercollegiate Guidelines Network recommended a discharge room air oxygen (RAO2) saturation threshold of 95%.[8] The same year, the American Academy of Pediatrics (AAP) bronchiolitis clinical practice guideline stated that oxygen is not needed for children with RAO2 saturations 90% who are feeding well and have minimal respiratory distress.[9] There is a need for prospective studies to help clinicians make evidenced‐based discharge decisions for this common condition.
We performed a prospective, multicenter, multiyear study[10, 11, 12] to examine the typical inpatient clinical course of and to develop hospital discharge guidelines for children age <2 years hospitalized with bronchiolitis. We hypothesized that children would not worsen clinically and would be safe to discharge home once their respiratory status improved and they were able to remain hydrated.
METHODS
Study Design and Population
We conducted a prospective, multicenter cohort study for 3 consecutive years during the 2007 to 2010 winter seasons, as part of the Multicenter Airway Research Collaboration (MARC), a program of the Emergency Medicine Network (
All patients were treated at the discretion of the treating physician. Inclusion criteria were an attending physician's diagnosis of bronchiolitis, age <2 years, and the ability of the parent/guardian to give informed consent. The exclusion criteria were previous enrollment and transfer to a participating hospital >48 hours after the original admission time. Therefore, children with comorbid conditions were included in this study. All consent and data forms were translated into Spanish. The institutional review board at each of the 16 participating hospitals approved the study.
Of the 2207 enrolled children, we excluded 109 (5%) children with a hospital LOS <1 day due to inadequate time to capture the required data for the present analysis. Among the 2098 remaining children, 1916 (91%) had daily inpatient data on all factors used to define clinical improvement and clinical worsening. Thus, the analytic cohort was comprised of 1916 children hospitalized for bronchiolitis.
Data Collection
Investigators conducted detailed structured interviews. Chart reviews were conducted to obtain preadmission and daily hospital clinical data including respiratory rates, daily respiratory rate trends, degree of retractions, oxygen saturation, daily oxygen saturation trends, medical management, and disposition. These data were manually reviewed, and site investigators were queried about missing data and discrepancies. A follow‐up telephone interview was conducted with families 1 week after discharge to examine relapse events at both 24 hours and 7 days.
We used the question: How long ago did the following symptoms [eg, difficulty breathing] begin [for the] current illness? to estimate the onset of the current illness. Pulse was categorized as low, normal, or high based on age‐related heart rate values.[13] Presence of apnea was recorded daily by site investigators.[14]
Nasopharyngeal Aspirate Collection and Virology Testing
As described previously, site teams used a standardized protocol to collect nasopharyngeal aspirates,[11] which were tested for respiratory syncytial virus (RSV) types A and B; rhinovirus (RV); parainfluenza virus types 1, 2, and 3; influenza virus types A and B; 2009 novel H1N1; human metapneumovirus; coronaviruses NL‐63, HKU1, OC43, and 229E; enterovirus, and adenovirus using polymerase chain reaction.[11, 15, 16, 17]
Defining Clinical Improvement and Worsening
Clinical improvement criteria were based on the 2006 AAP guidelines.[9] For respiratory rate and oxygen saturation, clinicians estimated average daily respiratory rate and oxygen saturation based on the recorded readings from the previous 24 hours. This estimation reflects the process clinicians use when rounding on their hospitalized patients, and thus may be more similar to standard clinical practice than a calculated mean. The respiratory rate criteria are adjusted for age.[18, 19] For daily estimated average oxygen saturation we used the AAP criteria of RAO2 saturation of 90%. Considering that oxygen saturation is the main determinant of LOS,[20] healthy infants age <6 months may have transient oxygen saturations of around 80%,[21] and that errors in estimation may occur, we included a lowest RAO2 of 88% in our improvement criteria. By combining the dichotomized estimated oxygen saturation (90% or not) with the lower limit of 88%, there was little room for erroneous conclusions. A child was considered clinically improved on the earliest date he/she met all of the following criteria: (1) none or mild retractions and improved or stable retractions compared with the previous inpatient day; (2) daily estimated average respiratory rate (RR) <60 breaths per minute for age <6 months, <55 breaths/minute for age 6 to 11 months, and <45 breaths/minute for age 12 months with a decreasing or stable trend over the course of the current day; (3) daily estimated average RAO2 saturation 90%, lowest RAO2 saturation 88%[21]; and (4) not receiving intravenous (IV) fluids or for children receiving IV fluids a clinician report of the child maintaining oral hydration. Children who reached the clinical improvement criteria were considered clinically worse if they required intensive care or had the inverse of 1 of the improvement criteria: moderate/severe retractions that were worse compared with the previous inpatient day, daily average RR 60 with an increasing trend over the current day, need for oxygen, or need for IV fluids.
Statistical Analyses
All analyses were performed using Stata 12.0 (StataCorp, College Station, TX). Data are presented as proportions with 95% confidence intervals (95% CIs), means with standard deviations, and medians with interquartile ranges (IQR). To examine potential factors associated with clinical worsening after reaching clinical improvement, we used 2, Fisher exact, Student t test, and Kruskall‐Wallis tests, as appropriate.
Adjusted analyses used generalized linear mixed models with a logit link to identify independent risk factors for worsening after reaching clinical improvement. Fixed effects for patient‐level factors and a random site effect were used. Factors were tested for inclusion in the multivariable model if they were found to be associated with worsening in unadjusted analyses (P<0.20) or were considered clinically important. Results are reported as odds ratios with 95% CIs.
We performed several sensitivity analyses to evaluate these improvement criteria: (1) we excluded the lowest RAO2 saturation requirement of 88%, (2) we examined a 94% daily estimated average RAO2 saturation threshold,[22] (3) we examined a 95% daily estimated average RAO2 saturation threshold,[8] and (4) we examined children age <12 months with no history of wheeze.
RESULTS
There were 1916 children hospitalized with bronchiolitis with data on all factors used to define clinical improvement and clinical worsening. The median number of days from the beginning of difficulty breathing until admission was 2 days (IQR, 15.5 days; range, 18 days) and from the beginning of difficulty breathing until clinical improvement was 4 days (IQR, 37.5 days; range, 133 days) (Figure 1). The variance for days to admission was significantly less than the variance for days to clinical improvement (P<0.001).

In this observational study, clinicians discharged 214 (11%) of the 1916 children before meeting the definition of clinical improvement. Thus, 1702 (89%; 95% CI: 87%‐90%) children reached the clinical improvement criteria, had a LOS >1 day, and had data on all factors (Figure 2).

Of the 1702 children who met the clinical improvement criteria, there were 76 children (4%; 95% CI: 3%5%) who worsened (Figure 2). The worsening occurred within a median of 1 day (IQR, 13 days) of clinical improvement. Forty‐six (3%) of the children required transfer to the ICU (1 required intubation, 1 required continuous positive airway pressure, and 4 had apnea), 23 (1%) required oxygen, and 17 (1%) required IV fluids. Eight percent of children met multiple criteria for worsening. A comparison between children who did and did not worsen is shown in Table 1. In general, children who worsened after improvement were younger and born earlier. These children also presented in more severe respiratory distress, had moderate or severe retractions, oxygen saturation <85% at hospitalization, inadequate oral intake, and apnea documented during the hospitalization. Neither viral etiology nor site of care influenced whether the children worsened after improving. However, stratified analysis of children based on initial location of admission (ie, ICU or ward) showed that among the children admitted to the ICU from the emergency department (ED), 89% met the improvement criteria and 19% clinically worsened. In contrast, among children admitted to the ward from the ED, 89% met the improvement criteria, and only 2% clinically worsened. Stratified multivariable models based on the initial location of admission from the ED (ie, ICU or ward) were not possible due to small sample sizes after stratification. None of these children had relapse events requiring rehospitalization within either 24 hours or 7 days of discharge.
| Did Not Worsen, n=1,626 | Worsened, n=76 | P Value | |
|---|---|---|---|
| |||
| Demographic characteristics | |||
| Age <2 months, % | 29 | 57 | <0.001 |
| Month of birth, % | 0.02 | ||
| OctoberMarch | 61 | 75 | |
| AprilSeptember | 39 | 25 | |
| Sex, % | 0.51 | ||
| Male | 59 | 55 | |
| Female | 41 | 45 | |
| Race, % | 0.050 | ||
| White | 63 | 58 | |
| Black | 23 | 34 | |
| Other or missing | 14 | 8 | |
| Hispanic ethnicity, % | 37 | 22 | 0.01 |
| Insurance, % | 0.87 | ||
| Nonprivate | 68 | 67 | |
| Private | 32 | 33 | |
| Medical history | |||
| Gestational age <37 weeks, % | 23 | 39 | 0.002 |
| Birth weight, % | 0.52 | ||
| <5 lbs | 13 | 12 | |
| 5 lbs | 34 | 41 | |
| 7 lbs | 53 | 47 | |
| Mother's age, median (IQR) | 27 (2333) | 27 (2233) | 0.54 |
| Is or was breastfed, % | 61 | 51 | 0.10 |
| Smoked during pregnancy, % | 15 | 20 | 0.22 |
| Exposure to smoke, % | 13 | 20 | 0.11 |
| Family history of asthma, % | 0.89 | ||
| Neither parent | 68 | 64 | |
| Either mother or father | 27 | 30 | |
| Both parents | 4 | 4 | |
| Do not know/missing | 2 | 1 | |
| History of wheezing, % | 23 | 17 | 0.24 |
| History of eczema, % | 16 | 7 | 0.04 |
| History of intubation, % | 9 | 12 | 0.50 |
| Major, relevant, comorbid medical disorder, % | 20 | 24 | 0.46 |
| Current illness | |||
| When difficulty breathing began, preadmission, % | 0.63 | ||
| 1 day | 70 | 75 | |
| <1 day | 28 | 23 | |
| No difficulty preadmission | 2 | 3 | |
| Weight, lbs, median (IQR) | 12.3 (8.817.4) | 9.0 (6.613.2) | 0.001 |
| Temperature, F, median (IQR) | 99.5 (98.6100.6) | 99.4 (98.1100.4) | 0.06 |
| Pulse, beats per minute by age | 0.82 | ||
| Low | 0.3 | 0 | |
| Normal | 48 | 46 | |
| High | 51 | 54 | |
| Respiratory rate, breaths per minute, median (IQR) | 48 (4060) | 48 (3864) | 0.28 |
| Retractions, % | 0.001 | ||
| None | 22 | 25 | |
| Mild | 43 | 24 | |
| Moderate | 26 | 33 | |
| Severe | 4 | 12 | |
| Missing | 5 | 7 | |
| Oxygen saturation by pulse oximetry or ABG, % | 0.001 | ||
| <85 | 4 | 12 | |
| 8587.9 | 3 | 4 | |
| 8889.9 | 5 | 0 | |
| 9093.9 | 18 | 11 | |
| 94 | 72 | 73 | |
| Oral intake, % | <0.001 | ||
| Adequate | 45 | 22 | |
| Inadequate | 42 | 63 | |
| Missing | 13 | 14 | |
| Presence of apnea, % | 7 | 24 | <0.001 |
| RSV‐A, % | 44 | 41 | 0.54 |
| RSV‐B, % | 30 | 25 | 0.36 |
| HRV, % | 24 | 24 | 0.88 |
| Chest x‐ray results during ED/preadmission visit | |||
| Atelectasis | 12 | 13 | 0.77 |
| Infiltrate | 13 | 11 | 0.50 |
| Hyperinflated | 18 | 21 | 0.47 |
| Peribronchial cuffing/thickening | 23 | 17 | 0.32 |
| Normal | 14 | 16 | 0.75 |
| White blood count, median (IQR) | 11.2 (8.714.4) | 11.9 (9.214.4) | 0.60 |
| Platelet count, median (IQR) | 395 (317490) | 430 (299537) | 0.56 |
| Sodium, median (IQR) | 138 (136140) | 137 (135138) | 0.19 |
| Hospital length of stay, median (IQR) | 2 (14) | 4.5 (28) | <0.001 |
| One‐week follow‐up | |||
| Relapse within 24 hours of hospital discharge requiring hospital admission, % | 0.5 | 0 | 0.56 |
| Relapse within 7 days of hospital discharge requiring hospital admission, % | 1 | 0 | 0.35 |
On multivariable analysis (Table 2), independent risk factors for worsening after reaching the clinical improvement criteria were young age, preterm birth, and presenting to care with more severe bronchiolitis represented by severe retractions, inadequate oral intake, or apnea. To further evaluate the improvement criteria in the current analysis, multiple sensitivity analyses were conducted. The frequency of clinical worsening after reaching the improvement criteria was stable when we examined different RA02 criteria in sensitivity analyses: (1) excluding RA02 as a criterion for improvement: 90% met improvement criteria and 4% experienced clinical worsening, (2) changing the average RA02 threshold for clinical improvement to 94%: 62% met improvement criteria and 6% experienced clinical worsening, and (3) changing the average RA02 threshold for clinical improvement to 95%: 47% met improvement criteria and 5% experienced clinical worsening. Furthermore, stratifying by age <2 months and restricting to more stringent definitions of bronchiolitis (ie, age <1 year or age <1 year+no history of wheezing) also did not materially change the results (see Supporting Figure 1 in the online version of this article).
| Odds Ratio | 95% CI | P Value | |
|---|---|---|---|
| |||
| Age <2 months | 3.51 | 2.07‐5.94 | <0.001 |
| Gestational age <37 weeks | 1.94 | 1.13‐3.32 | 0.02 |
| Retractions | |||
| None | 1.30 | 0.80‐3.23 | 0.19 |
| Mild | 1.0 | Reference | |
| Moderate | 1.91 | 0.99‐3.71 | 0.06 |
| Severe | 5.55 | 2.1214.50 | <0.001 |
| Missing | 1.70 | 0.53‐5.42 | 0.37 |
| Oral intake | |||
| Adequate | 1.00 | Reference | |
| Inadequate | 2.54 | 1.39‐4.62 | 0.002 |
| Unknown/missing | 1.88 | 0.79‐4.44 | 0.15 |
| Presence of apnea | 2.87 | 1.45‐5.68 | 0.003 |
We compared the 214 children who were discharged prior to reaching clinical improvement with the 1702 children who reached the clinical improvement criteria. The 214 children were less likely to be age <2 months (22% vs 30%; P=0.02). These 2 groups (214 vs 1702) were similar with respect to severe retractions (2% vs 4%; P=0.13), median respiratory rate (48 vs 48; P=0.42), oxygen saturation <90% (15% vs 11%; P=0.07), inadequate oral intake (50% vs 43%; P=0.13), and rates of relapse events requiring rehospitalization within both 24 hours (0.6% vs 0.6%; P=0.88) and 7 days (1% vs 1%; P=0.90) of discharge.
DISCUSSION
In this large, multicenter, multiyear study of children hospitalized with bronchiolitis, we found that children present to a hospital in a relatively narrow time frame, but their time to recovery in the hospital is highly variable. Nonetheless, 96% of children continued to improve once they had: (1) improving or stable retractions rated as none/mild, (2) a decreasing or stable RR by age, (3) estimated average RAO2 saturation 90% and lowest RAO2 saturation of 88%, and (4) were hydrated. The 4% of children who worsened after clinically improving were more likely to be age <2 months, born <37 weeks, and present with more severe distress (ie, severe retractions, inadequate oral intake, or apnea). Based on the low risk of worsening after clinical improvement, especially among children admitted to the regular ward (2%), we believe these 4 clinical criteria could be used as discharge criteria for this common pediatric illness with a predominantly monophasic clinical course.
Variability in hospital LOS for children with bronchiolitis exists in the United States[3] and internationally.[4, 5] Cheung and colleagues analyzed administrative data from over 75,000 children admitted for bronchiolitis in England between April 2007 and March 2010 and found sixfold variation in LOS between sites. They concluded that this LOS variability was due in part to providers' clinical decision making.[5] Srivastava and colleagues[23] addressed variable clinician decision making in bronchiolitis and 10 other common pediatric conditions by embedding discharge criteria developed by expert consensus into admission order sets. They found that for children with bronchiolitis, the embedded discharge criteria reduced the median LOS from 1.91 to 1.87 days. In contrast to the single‐center data presented by White and colleagues,[24] the prospective, multicenter MARC‐30 data provide a clear understanding of the normal clinical course for children hospitalized with bronchiolitis, determine if children clinically worsen after clinical improvement, and provide data about discharge criteria for children hospitalized with bronchiolitis. Although there is a lack of rigorous published data, the lower tract symptoms of bronchiolitis (eg, cough, retractions) are said to peak on days 5 to 7 of illness and then gradually resolve.[25] In the present study, we found that the time from the onset of difficulty breathing until hospital admission is less variable than the time from the onset of difficulty breathing until either clinical improvement or discharge. Although 75% of children have clinically improved within 7.5 days of difficulty breathing based on the IQR results, the remaining 25% may have a more prolonged recovery in the hospital of up to 3 weeks. Interestingly, prolonged recovery times from bronchiolitis have also been noted in children presenting to the ED[26] and in an outpatient population.[27] It is unclear why 20% to 25% of children at different levels of severity of illness have prolonged recovery from bronchiolitis, but this group of children requires further investigation.
Given the variability of recovery times, clinicians may have difficulty knowing when a child is ready for hospital discharge. One of the main stumbling blocks for discharge readiness in children with bronchiolitis is the interpretation of the oxygen saturation value.[6, 8, 9, 20, 28] However, it should be considered that interpreting the oxygen saturation in a child who is clinically improving in the hospital setting is different than interpreting the oxygen saturation of a child in the ED or the clinic whose clinical course is less certain.[22] In the hospital setting, using the oxygen saturation value in in the AAP guideline,[9] 4% of children clinically worsened after they met the improvement criteria, a clinical pattern observed previously with supplemental oxygen.[28] This unpredictability may explain some of the variation in providers' clinical decision making.[5] The children who worsened, and therefore deserve more cautious discharge planning, were young (<2 months), premature (<37 weeks gestational age), and presented in more severe distress. Those children admitted to the ICU from the ED worsened more commonly than children admitted to the ward (19% vs 2%). Interestingly, the viral etiology of the child's bronchiolitis did not influence whether a child worsened after reaching the improvement criteria. Therefore, although children with RV bronchiolitis have a shorter hospital LOS than children with RSV bronchiolitis,[11] the pattern of recovery did not differ by viral etiology.
In addition to unsafe discharges, clinicians may be concerned about the possibility of readmissions. Although somewhat controversial, hospital readmission is being used as a quality of care metric.[29, 30, 31] One response to minimize readmissions would be for clinicians to observe children for longer than clinically indicated.[32] However, shorter LOS is not necessarily associated with increased readmission rates.[33] Given that the geometric mean of hospital charges per child with bronchiolitis increased from $6380 in 2000 to $8530 in 2009,[34] the potential for safely reducing hospital LOS by using the discharge criteria proposed in the current study instead of other criteria[8] may net substantial cost savings. Furthermore, reducing LOS would decrease the time children expose others to these respiratory viruses and possibly reduce medical errors.[35]
Our study has some potential limitations. Because the study participants were all hospitalized, these data do not inform admission or discharge decisions from either the ED or the clinic; but other data address those clinical scenarios.[22] Also, the 16 sites that participated in this study were large, urban teaching hospitals. Consequently, these results are not necessarily generalizable to smaller community hospitals. Although numerous data points were required to enter the analytic cohort, only 9% of the sample was excluded for missing data. There were 214 children who did not meet our improvement criteria by the time of discharge. Although the inability to include these children in the analysis may be seen as a limitation, this practice variability underscores the need for more data about discharging hospitalized children with bronchiolitis. Last, site teams reviewed medical records daily. More frequent recording of the clinical course would have yielded more granular data, but the current methodology replicates how data are generally presented during patient care rounds, when decisions about suitability for discharge are often considered.
CONCLUSION
We documented in this large multicenter study that most children hospitalized with bronchiolitis had a wide range of time to recovery, but the vast majority continued to improve once they reached the identified clinical criteria that predict a safe discharge to home. The children who worsened after clinical improvement were more likely to be younger, premature infants presenting in more severe distress. Although additional prospective validation of these hospital discharge criteria is warranted, these data may help clinicians make more evidence‐based discharge decisions for a common pediatric illness with high practice variation, both in the United States[3] and in other countries.[4, 5]
Acknowledgements
Collaborators in the MARC‐30 Study: Besh Barcega, MD, Loma Linda University Children's Hospital, Loma Linda, CA; John Cheng, MD, Children's Healthcare of Atlanta at Egleston, Atlanta, GA; Dorothy Damore, MD, New York Presbyterian Hospital‐Cornell, New York, NY; Carlos Delgado, MD, Children's Healthcare of Atlanta at Egleston, Atlanta, GA; Haitham Haddad, MD, Rainbow Babies & Children's Hospital, Cleveland, OH; Paul Hain, MD, Monroe Carell Jr. Children's Hospital at Vanderbilt, Nashville, TN; Frank LoVecchio, DO, Maricopa Medical Center, Phoenix, AZ; Charles Macias, MD MPH, Texas Children's Hospital, Houston, TX; Jonathan Mansbach, MD, MPH, Boston Children's Hospital, Boston, MA; Eugene Mowad, MD, Akron Children's Hospital, Akron, OH; Brian Pate, MD, Children's Mercy Hospital, Kansas City, MO; Mark Riederer, MD, Monroe Carell Jr. Children's Hospital at Vanderbilt, Nashville, TN; M. Jason Sanders, MD, Children's Memorial Hermann Hospital, Houston, TX; Alan R. Schroeder, MD, Santa Clara Valley Medical Center, San Jose, CA; Nikhil Shah, MD, New York Presbyterian Hospital‐Cornell, New York, NY; Michelle Stevenson, MD, MS, Kosair Children's Hospital, Louisville, KY; Erin Stucky Fisher, MD, Rady Children's Hospital, San Diego, CA; Stephen Teach, MD, MPH, Children's National Medical Center, Washington, DC; Lisa Zaoutis, MD, Children's Hospital of Philadelphia, Philadelphia, PA.
Disclosures: This study was supported by grants U01 AI‐67693 and K23 AI‐77801 from the National Institutes of Health (Bethesda, MD). The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Allergy and Infectious Diseases or the National Institutes of Health. Drs. Mansbach and Piedra have provided consultation to Regeneron Pharmaceuticals. Otherwise, no authors report any potential conflicts of interest, including relevant financial interests, activities, relationships, and affiliations.
Although bronchiolitis is the leading cause of hospitalization for US infants,[1] there is a lack of basic prospective data about the expected inpatient clinical course and ongoing uncertainty about when a hospitalized child is ready for discharge to home.[2] This lack of data about children's readiness for discharge may result in variable hospital length‐of‐stay (LOS).[3, 4, 5]
One specific source of variability in discharge readiness and LOS variability may be the lack of consensus about safe threshold oxygen saturation values for discharge in children hospitalized with bronchiolitis.[6, 7] In 2006, the Scottish Intercollegiate Guidelines Network recommended a discharge room air oxygen (RAO2) saturation threshold of 95%.[8] The same year, the American Academy of Pediatrics (AAP) bronchiolitis clinical practice guideline stated that oxygen is not needed for children with RAO2 saturations 90% who are feeding well and have minimal respiratory distress.[9] There is a need for prospective studies to help clinicians make evidenced‐based discharge decisions for this common condition.
We performed a prospective, multicenter, multiyear study[10, 11, 12] to examine the typical inpatient clinical course of and to develop hospital discharge guidelines for children age <2 years hospitalized with bronchiolitis. We hypothesized that children would not worsen clinically and would be safe to discharge home once their respiratory status improved and they were able to remain hydrated.
METHODS
Study Design and Population
We conducted a prospective, multicenter cohort study for 3 consecutive years during the 2007 to 2010 winter seasons, as part of the Multicenter Airway Research Collaboration (MARC), a program of the Emergency Medicine Network (
All patients were treated at the discretion of the treating physician. Inclusion criteria were an attending physician's diagnosis of bronchiolitis, age <2 years, and the ability of the parent/guardian to give informed consent. The exclusion criteria were previous enrollment and transfer to a participating hospital >48 hours after the original admission time. Therefore, children with comorbid conditions were included in this study. All consent and data forms were translated into Spanish. The institutional review board at each of the 16 participating hospitals approved the study.
Of the 2207 enrolled children, we excluded 109 (5%) children with a hospital LOS <1 day due to inadequate time to capture the required data for the present analysis. Among the 2098 remaining children, 1916 (91%) had daily inpatient data on all factors used to define clinical improvement and clinical worsening. Thus, the analytic cohort was comprised of 1916 children hospitalized for bronchiolitis.
Data Collection
Investigators conducted detailed structured interviews. Chart reviews were conducted to obtain preadmission and daily hospital clinical data including respiratory rates, daily respiratory rate trends, degree of retractions, oxygen saturation, daily oxygen saturation trends, medical management, and disposition. These data were manually reviewed, and site investigators were queried about missing data and discrepancies. A follow‐up telephone interview was conducted with families 1 week after discharge to examine relapse events at both 24 hours and 7 days.
We used the question: How long ago did the following symptoms [eg, difficulty breathing] begin [for the] current illness? to estimate the onset of the current illness. Pulse was categorized as low, normal, or high based on age‐related heart rate values.[13] Presence of apnea was recorded daily by site investigators.[14]
Nasopharyngeal Aspirate Collection and Virology Testing
As described previously, site teams used a standardized protocol to collect nasopharyngeal aspirates,[11] which were tested for respiratory syncytial virus (RSV) types A and B; rhinovirus (RV); parainfluenza virus types 1, 2, and 3; influenza virus types A and B; 2009 novel H1N1; human metapneumovirus; coronaviruses NL‐63, HKU1, OC43, and 229E; enterovirus, and adenovirus using polymerase chain reaction.[11, 15, 16, 17]
Defining Clinical Improvement and Worsening
Clinical improvement criteria were based on the 2006 AAP guidelines.[9] For respiratory rate and oxygen saturation, clinicians estimated average daily respiratory rate and oxygen saturation based on the recorded readings from the previous 24 hours. This estimation reflects the process clinicians use when rounding on their hospitalized patients, and thus may be more similar to standard clinical practice than a calculated mean. The respiratory rate criteria are adjusted for age.[18, 19] For daily estimated average oxygen saturation we used the AAP criteria of RAO2 saturation of 90%. Considering that oxygen saturation is the main determinant of LOS,[20] healthy infants age <6 months may have transient oxygen saturations of around 80%,[21] and that errors in estimation may occur, we included a lowest RAO2 of 88% in our improvement criteria. By combining the dichotomized estimated oxygen saturation (90% or not) with the lower limit of 88%, there was little room for erroneous conclusions. A child was considered clinically improved on the earliest date he/she met all of the following criteria: (1) none or mild retractions and improved or stable retractions compared with the previous inpatient day; (2) daily estimated average respiratory rate (RR) <60 breaths per minute for age <6 months, <55 breaths/minute for age 6 to 11 months, and <45 breaths/minute for age 12 months with a decreasing or stable trend over the course of the current day; (3) daily estimated average RAO2 saturation 90%, lowest RAO2 saturation 88%[21]; and (4) not receiving intravenous (IV) fluids or for children receiving IV fluids a clinician report of the child maintaining oral hydration. Children who reached the clinical improvement criteria were considered clinically worse if they required intensive care or had the inverse of 1 of the improvement criteria: moderate/severe retractions that were worse compared with the previous inpatient day, daily average RR 60 with an increasing trend over the current day, need for oxygen, or need for IV fluids.
Statistical Analyses
All analyses were performed using Stata 12.0 (StataCorp, College Station, TX). Data are presented as proportions with 95% confidence intervals (95% CIs), means with standard deviations, and medians with interquartile ranges (IQR). To examine potential factors associated with clinical worsening after reaching clinical improvement, we used 2, Fisher exact, Student t test, and Kruskall‐Wallis tests, as appropriate.
Adjusted analyses used generalized linear mixed models with a logit link to identify independent risk factors for worsening after reaching clinical improvement. Fixed effects for patient‐level factors and a random site effect were used. Factors were tested for inclusion in the multivariable model if they were found to be associated with worsening in unadjusted analyses (P<0.20) or were considered clinically important. Results are reported as odds ratios with 95% CIs.
We performed several sensitivity analyses to evaluate these improvement criteria: (1) we excluded the lowest RAO2 saturation requirement of 88%, (2) we examined a 94% daily estimated average RAO2 saturation threshold,[22] (3) we examined a 95% daily estimated average RAO2 saturation threshold,[8] and (4) we examined children age <12 months with no history of wheeze.
RESULTS
There were 1916 children hospitalized with bronchiolitis with data on all factors used to define clinical improvement and clinical worsening. The median number of days from the beginning of difficulty breathing until admission was 2 days (IQR, 15.5 days; range, 18 days) and from the beginning of difficulty breathing until clinical improvement was 4 days (IQR, 37.5 days; range, 133 days) (Figure 1). The variance for days to admission was significantly less than the variance for days to clinical improvement (P<0.001).

In this observational study, clinicians discharged 214 (11%) of the 1916 children before meeting the definition of clinical improvement. Thus, 1702 (89%; 95% CI: 87%‐90%) children reached the clinical improvement criteria, had a LOS >1 day, and had data on all factors (Figure 2).

Of the 1702 children who met the clinical improvement criteria, there were 76 children (4%; 95% CI: 3%5%) who worsened (Figure 2). The worsening occurred within a median of 1 day (IQR, 13 days) of clinical improvement. Forty‐six (3%) of the children required transfer to the ICU (1 required intubation, 1 required continuous positive airway pressure, and 4 had apnea), 23 (1%) required oxygen, and 17 (1%) required IV fluids. Eight percent of children met multiple criteria for worsening. A comparison between children who did and did not worsen is shown in Table 1. In general, children who worsened after improvement were younger and born earlier. These children also presented in more severe respiratory distress, had moderate or severe retractions, oxygen saturation <85% at hospitalization, inadequate oral intake, and apnea documented during the hospitalization. Neither viral etiology nor site of care influenced whether the children worsened after improving. However, stratified analysis of children based on initial location of admission (ie, ICU or ward) showed that among the children admitted to the ICU from the emergency department (ED), 89% met the improvement criteria and 19% clinically worsened. In contrast, among children admitted to the ward from the ED, 89% met the improvement criteria, and only 2% clinically worsened. Stratified multivariable models based on the initial location of admission from the ED (ie, ICU or ward) were not possible due to small sample sizes after stratification. None of these children had relapse events requiring rehospitalization within either 24 hours or 7 days of discharge.
| Did Not Worsen, n=1,626 | Worsened, n=76 | P Value | |
|---|---|---|---|
| |||
| Demographic characteristics | |||
| Age <2 months, % | 29 | 57 | <0.001 |
| Month of birth, % | 0.02 | ||
| OctoberMarch | 61 | 75 | |
| AprilSeptember | 39 | 25 | |
| Sex, % | 0.51 | ||
| Male | 59 | 55 | |
| Female | 41 | 45 | |
| Race, % | 0.050 | ||
| White | 63 | 58 | |
| Black | 23 | 34 | |
| Other or missing | 14 | 8 | |
| Hispanic ethnicity, % | 37 | 22 | 0.01 |
| Insurance, % | 0.87 | ||
| Nonprivate | 68 | 67 | |
| Private | 32 | 33 | |
| Medical history | |||
| Gestational age <37 weeks, % | 23 | 39 | 0.002 |
| Birth weight, % | 0.52 | ||
| <5 lbs | 13 | 12 | |
| 5 lbs | 34 | 41 | |
| 7 lbs | 53 | 47 | |
| Mother's age, median (IQR) | 27 (2333) | 27 (2233) | 0.54 |
| Is or was breastfed, % | 61 | 51 | 0.10 |
| Smoked during pregnancy, % | 15 | 20 | 0.22 |
| Exposure to smoke, % | 13 | 20 | 0.11 |
| Family history of asthma, % | 0.89 | ||
| Neither parent | 68 | 64 | |
| Either mother or father | 27 | 30 | |
| Both parents | 4 | 4 | |
| Do not know/missing | 2 | 1 | |
| History of wheezing, % | 23 | 17 | 0.24 |
| History of eczema, % | 16 | 7 | 0.04 |
| History of intubation, % | 9 | 12 | 0.50 |
| Major, relevant, comorbid medical disorder, % | 20 | 24 | 0.46 |
| Current illness | |||
| When difficulty breathing began, preadmission, % | 0.63 | ||
| 1 day | 70 | 75 | |
| <1 day | 28 | 23 | |
| No difficulty preadmission | 2 | 3 | |
| Weight, lbs, median (IQR) | 12.3 (8.817.4) | 9.0 (6.613.2) | 0.001 |
| Temperature, F, median (IQR) | 99.5 (98.6100.6) | 99.4 (98.1100.4) | 0.06 |
| Pulse, beats per minute by age | 0.82 | ||
| Low | 0.3 | 0 | |
| Normal | 48 | 46 | |
| High | 51 | 54 | |
| Respiratory rate, breaths per minute, median (IQR) | 48 (4060) | 48 (3864) | 0.28 |
| Retractions, % | 0.001 | ||
| None | 22 | 25 | |
| Mild | 43 | 24 | |
| Moderate | 26 | 33 | |
| Severe | 4 | 12 | |
| Missing | 5 | 7 | |
| Oxygen saturation by pulse oximetry or ABG, % | 0.001 | ||
| <85 | 4 | 12 | |
| 8587.9 | 3 | 4 | |
| 8889.9 | 5 | 0 | |
| 9093.9 | 18 | 11 | |
| 94 | 72 | 73 | |
| Oral intake, % | <0.001 | ||
| Adequate | 45 | 22 | |
| Inadequate | 42 | 63 | |
| Missing | 13 | 14 | |
| Presence of apnea, % | 7 | 24 | <0.001 |
| RSV‐A, % | 44 | 41 | 0.54 |
| RSV‐B, % | 30 | 25 | 0.36 |
| HRV, % | 24 | 24 | 0.88 |
| Chest x‐ray results during ED/preadmission visit | |||
| Atelectasis | 12 | 13 | 0.77 |
| Infiltrate | 13 | 11 | 0.50 |
| Hyperinflated | 18 | 21 | 0.47 |
| Peribronchial cuffing/thickening | 23 | 17 | 0.32 |
| Normal | 14 | 16 | 0.75 |
| White blood count, median (IQR) | 11.2 (8.714.4) | 11.9 (9.214.4) | 0.60 |
| Platelet count, median (IQR) | 395 (317490) | 430 (299537) | 0.56 |
| Sodium, median (IQR) | 138 (136140) | 137 (135138) | 0.19 |
| Hospital length of stay, median (IQR) | 2 (14) | 4.5 (28) | <0.001 |
| One‐week follow‐up | |||
| Relapse within 24 hours of hospital discharge requiring hospital admission, % | 0.5 | 0 | 0.56 |
| Relapse within 7 days of hospital discharge requiring hospital admission, % | 1 | 0 | 0.35 |
On multivariable analysis (Table 2), independent risk factors for worsening after reaching the clinical improvement criteria were young age, preterm birth, and presenting to care with more severe bronchiolitis represented by severe retractions, inadequate oral intake, or apnea. To further evaluate the improvement criteria in the current analysis, multiple sensitivity analyses were conducted. The frequency of clinical worsening after reaching the improvement criteria was stable when we examined different RA02 criteria in sensitivity analyses: (1) excluding RA02 as a criterion for improvement: 90% met improvement criteria and 4% experienced clinical worsening, (2) changing the average RA02 threshold for clinical improvement to 94%: 62% met improvement criteria and 6% experienced clinical worsening, and (3) changing the average RA02 threshold for clinical improvement to 95%: 47% met improvement criteria and 5% experienced clinical worsening. Furthermore, stratifying by age <2 months and restricting to more stringent definitions of bronchiolitis (ie, age <1 year or age <1 year+no history of wheezing) also did not materially change the results (see Supporting Figure 1 in the online version of this article).
| Odds Ratio | 95% CI | P Value | |
|---|---|---|---|
| |||
| Age <2 months | 3.51 | 2.07‐5.94 | <0.001 |
| Gestational age <37 weeks | 1.94 | 1.13‐3.32 | 0.02 |
| Retractions | |||
| None | 1.30 | 0.80‐3.23 | 0.19 |
| Mild | 1.0 | Reference | |
| Moderate | 1.91 | 0.99‐3.71 | 0.06 |
| Severe | 5.55 | 2.1214.50 | <0.001 |
| Missing | 1.70 | 0.53‐5.42 | 0.37 |
| Oral intake | |||
| Adequate | 1.00 | Reference | |
| Inadequate | 2.54 | 1.39‐4.62 | 0.002 |
| Unknown/missing | 1.88 | 0.79‐4.44 | 0.15 |
| Presence of apnea | 2.87 | 1.45‐5.68 | 0.003 |
We compared the 214 children who were discharged prior to reaching clinical improvement with the 1702 children who reached the clinical improvement criteria. The 214 children were less likely to be age <2 months (22% vs 30%; P=0.02). These 2 groups (214 vs 1702) were similar with respect to severe retractions (2% vs 4%; P=0.13), median respiratory rate (48 vs 48; P=0.42), oxygen saturation <90% (15% vs 11%; P=0.07), inadequate oral intake (50% vs 43%; P=0.13), and rates of relapse events requiring rehospitalization within both 24 hours (0.6% vs 0.6%; P=0.88) and 7 days (1% vs 1%; P=0.90) of discharge.
DISCUSSION
In this large, multicenter, multiyear study of children hospitalized with bronchiolitis, we found that children present to a hospital in a relatively narrow time frame, but their time to recovery in the hospital is highly variable. Nonetheless, 96% of children continued to improve once they had: (1) improving or stable retractions rated as none/mild, (2) a decreasing or stable RR by age, (3) estimated average RAO2 saturation 90% and lowest RAO2 saturation of 88%, and (4) were hydrated. The 4% of children who worsened after clinically improving were more likely to be age <2 months, born <37 weeks, and present with more severe distress (ie, severe retractions, inadequate oral intake, or apnea). Based on the low risk of worsening after clinical improvement, especially among children admitted to the regular ward (2%), we believe these 4 clinical criteria could be used as discharge criteria for this common pediatric illness with a predominantly monophasic clinical course.
Variability in hospital LOS for children with bronchiolitis exists in the United States[3] and internationally.[4, 5] Cheung and colleagues analyzed administrative data from over 75,000 children admitted for bronchiolitis in England between April 2007 and March 2010 and found sixfold variation in LOS between sites. They concluded that this LOS variability was due in part to providers' clinical decision making.[5] Srivastava and colleagues[23] addressed variable clinician decision making in bronchiolitis and 10 other common pediatric conditions by embedding discharge criteria developed by expert consensus into admission order sets. They found that for children with bronchiolitis, the embedded discharge criteria reduced the median LOS from 1.91 to 1.87 days. In contrast to the single‐center data presented by White and colleagues,[24] the prospective, multicenter MARC‐30 data provide a clear understanding of the normal clinical course for children hospitalized with bronchiolitis, determine if children clinically worsen after clinical improvement, and provide data about discharge criteria for children hospitalized with bronchiolitis. Although there is a lack of rigorous published data, the lower tract symptoms of bronchiolitis (eg, cough, retractions) are said to peak on days 5 to 7 of illness and then gradually resolve.[25] In the present study, we found that the time from the onset of difficulty breathing until hospital admission is less variable than the time from the onset of difficulty breathing until either clinical improvement or discharge. Although 75% of children have clinically improved within 7.5 days of difficulty breathing based on the IQR results, the remaining 25% may have a more prolonged recovery in the hospital of up to 3 weeks. Interestingly, prolonged recovery times from bronchiolitis have also been noted in children presenting to the ED[26] and in an outpatient population.[27] It is unclear why 20% to 25% of children at different levels of severity of illness have prolonged recovery from bronchiolitis, but this group of children requires further investigation.
Given the variability of recovery times, clinicians may have difficulty knowing when a child is ready for hospital discharge. One of the main stumbling blocks for discharge readiness in children with bronchiolitis is the interpretation of the oxygen saturation value.[6, 8, 9, 20, 28] However, it should be considered that interpreting the oxygen saturation in a child who is clinically improving in the hospital setting is different than interpreting the oxygen saturation of a child in the ED or the clinic whose clinical course is less certain.[22] In the hospital setting, using the oxygen saturation value in in the AAP guideline,[9] 4% of children clinically worsened after they met the improvement criteria, a clinical pattern observed previously with supplemental oxygen.[28] This unpredictability may explain some of the variation in providers' clinical decision making.[5] The children who worsened, and therefore deserve more cautious discharge planning, were young (<2 months), premature (<37 weeks gestational age), and presented in more severe distress. Those children admitted to the ICU from the ED worsened more commonly than children admitted to the ward (19% vs 2%). Interestingly, the viral etiology of the child's bronchiolitis did not influence whether a child worsened after reaching the improvement criteria. Therefore, although children with RV bronchiolitis have a shorter hospital LOS than children with RSV bronchiolitis,[11] the pattern of recovery did not differ by viral etiology.
In addition to unsafe discharges, clinicians may be concerned about the possibility of readmissions. Although somewhat controversial, hospital readmission is being used as a quality of care metric.[29, 30, 31] One response to minimize readmissions would be for clinicians to observe children for longer than clinically indicated.[32] However, shorter LOS is not necessarily associated with increased readmission rates.[33] Given that the geometric mean of hospital charges per child with bronchiolitis increased from $6380 in 2000 to $8530 in 2009,[34] the potential for safely reducing hospital LOS by using the discharge criteria proposed in the current study instead of other criteria[8] may net substantial cost savings. Furthermore, reducing LOS would decrease the time children expose others to these respiratory viruses and possibly reduce medical errors.[35]
Our study has some potential limitations. Because the study participants were all hospitalized, these data do not inform admission or discharge decisions from either the ED or the clinic; but other data address those clinical scenarios.[22] Also, the 16 sites that participated in this study were large, urban teaching hospitals. Consequently, these results are not necessarily generalizable to smaller community hospitals. Although numerous data points were required to enter the analytic cohort, only 9% of the sample was excluded for missing data. There were 214 children who did not meet our improvement criteria by the time of discharge. Although the inability to include these children in the analysis may be seen as a limitation, this practice variability underscores the need for more data about discharging hospitalized children with bronchiolitis. Last, site teams reviewed medical records daily. More frequent recording of the clinical course would have yielded more granular data, but the current methodology replicates how data are generally presented during patient care rounds, when decisions about suitability for discharge are often considered.
CONCLUSION
We documented in this large multicenter study that most children hospitalized with bronchiolitis had a wide range of time to recovery, but the vast majority continued to improve once they reached the identified clinical criteria that predict a safe discharge to home. The children who worsened after clinical improvement were more likely to be younger, premature infants presenting in more severe distress. Although additional prospective validation of these hospital discharge criteria is warranted, these data may help clinicians make more evidence‐based discharge decisions for a common pediatric illness with high practice variation, both in the United States[3] and in other countries.[4, 5]
Acknowledgements
Collaborators in the MARC‐30 Study: Besh Barcega, MD, Loma Linda University Children's Hospital, Loma Linda, CA; John Cheng, MD, Children's Healthcare of Atlanta at Egleston, Atlanta, GA; Dorothy Damore, MD, New York Presbyterian Hospital‐Cornell, New York, NY; Carlos Delgado, MD, Children's Healthcare of Atlanta at Egleston, Atlanta, GA; Haitham Haddad, MD, Rainbow Babies & Children's Hospital, Cleveland, OH; Paul Hain, MD, Monroe Carell Jr. Children's Hospital at Vanderbilt, Nashville, TN; Frank LoVecchio, DO, Maricopa Medical Center, Phoenix, AZ; Charles Macias, MD MPH, Texas Children's Hospital, Houston, TX; Jonathan Mansbach, MD, MPH, Boston Children's Hospital, Boston, MA; Eugene Mowad, MD, Akron Children's Hospital, Akron, OH; Brian Pate, MD, Children's Mercy Hospital, Kansas City, MO; Mark Riederer, MD, Monroe Carell Jr. Children's Hospital at Vanderbilt, Nashville, TN; M. Jason Sanders, MD, Children's Memorial Hermann Hospital, Houston, TX; Alan R. Schroeder, MD, Santa Clara Valley Medical Center, San Jose, CA; Nikhil Shah, MD, New York Presbyterian Hospital‐Cornell, New York, NY; Michelle Stevenson, MD, MS, Kosair Children's Hospital, Louisville, KY; Erin Stucky Fisher, MD, Rady Children's Hospital, San Diego, CA; Stephen Teach, MD, MPH, Children's National Medical Center, Washington, DC; Lisa Zaoutis, MD, Children's Hospital of Philadelphia, Philadelphia, PA.
Disclosures: This study was supported by grants U01 AI‐67693 and K23 AI‐77801 from the National Institutes of Health (Bethesda, MD). The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Allergy and Infectious Diseases or the National Institutes of Health. Drs. Mansbach and Piedra have provided consultation to Regeneron Pharmaceuticals. Otherwise, no authors report any potential conflicts of interest, including relevant financial interests, activities, relationships, and affiliations.
- , , , , . Infectious disease hospitalizations among infants in the United States. Pediatrics. 2008;121(2):244–252.
- . “A hospital is no place to be sick” Samuel Goldwyn (1882–1974). Arch Dis Child. 2009;94(8):565–566.
- , , , , , Variation in inpatient diagnostic testing and management of bronchiolitis. Pediatrics. 2005;115(4):878–884.
- , , , International variation in the management of infants hospitalized with respiratory syncytial virus. International RSV Study Group. Eur J Pediatr. 1998;157(3):215–220.
- , , , , . Population variation in admission rates and duration of inpatient stay for bronchiolitis in England. Arch Dis Child. 2013;98(1):57–59.
- , , , . Impact of pulse oximetry and oxygen therapy on length of stay in bronchiolitis hospitalizations. Arch Pediatr Adolesc Med. 2004;158(6):527–530.
- , , . Pulse oximetry in pediatric practice. Pediatrics. 2011;128(4):740–752.
- Scottish Intercollegiate Guidelines Network. Bronchiolitis in children (SIGN 91). In: NHS Quality Improvement Scotland. Edinburgh, Scotland: Scottish Intercollegiate Guidelines Network; 2006.
- , , , et al. Diagnosis and management of bronchiolitis. Pediatrics. 2006;118(4):1774–1793.
- , , , et al. Prospective multicenter study of children with bronchiolitis requiring mechanical ventilation. Pediatrics. 2012;130(3):e492–e500.
- , , , et al. Prospective multicenter study of viral etiology and hospital length of stay in children with severe bronchiolitis. Arch Pediatr Adolesc Med. 2012;166(8):700–706.
- , , , et al. Apnea in children hospitalized with bronchiolitis. Pediatrics. 2013;132(5):e1194–e1201.
- . Evaluation of the cardiovascular system: history and physical evaluation. In: Kliegman RM, Stanton BF, St. Geme JW III, Schor NF, Behrman RF, eds. Nelson Textbook of Pediatrics. Philadelphia, PA: Elsevier Saunders; 2011:1529–1536.
- , , , et al. Apnea in children hospitalized with bronchiolitis. Pediatrics. 2013;132(5):e1194–e1201.
- , , , et al. Respiratory viral infections in patients with chronic, obstructive pulmonary disease. J Infect. 2005;50(4):322–330.
- , , , . Evaluation of real‐time PCR for diagnosis of Bordetella pertussis infection. BMC Infect Dis. 2006;6:62.
- , , , , . Evaluation of three real‐time PCR assays for detection of Mycoplasma pneumoniae in an outbreak investigation. J Clin Microbiol. 2008;46(9):3116–3118.
- , , , et al. Normal ranges of heart rate and respiratory rate in children from birth to 18 years of age: a systematic review of observational studies. Lancet. 2011;377(9770):1011–1018.
- , , , , , . Development of heart and respiratory rate percentile curves for hospitalized children. Pediatrics. 2013;131(4):e1150–e1157.
- , . Effect of oxygen supplementation on length of stay for infants hospitalized with acute viral bronchiolitis. Pediatrics. 2008;121(3):470–475.
- , , , et al. Longitudinal assessment of hemoglobin oxygen saturation in healthy infants during the first 6 months of age. Collaborative Home Infant Monitoring Evaluation (CHIME) Study Group. J Pediatr. 1999;135(5):580–586.
- , , , et al. Prospective multicenter study of bronchiolitis: predicting safe discharges from the emergency department. Pediatrics. 2008;121(4):680–688.
- , , , et al. Delays in discharge in a tertiary care pediatric hospital. J Hosp Med. 2009;4(8):481–485.
- , , , et al. Using quality improvement to optimise paediatric discharge efficiency. BMJ Qual Saf. 2014;23(5):428–436.
- , . Bronchiolitis in infants and children: treatment; outcome; and prevention. In: Torchia M, ed. UpToDate. Alphen aan den Rijn, the Netherlands; Wolters Kluwer Health; 2013.
- , . Duration of illness in infants with bronchiolitis evaluated in the emergency department. Pediatrics. 2010;126(2):285–290.
- , , . Duration of illness in ambulatory children diagnosed with bronchiolitis. Arch Pediatr Adolesc Med. 2000;154(10):997–1000.
- , , , et al. A clinical pathway for bronchiolitis is effective in reducing readmission rates. J Pediatr. 2005;147(5):622–626.
- , , , et al. Measuring hospital quality using pediatric readmission and revisit rates. Pediatrics. 2013;132(3):429–436.
- , , , et al. Pediatric readmission prevalence and variability across hospitals. JAMA. 2013;309(4):372–380.
- , , , , , . Preventability of early readmissions at a children's hospital. Pediatrics. 2013;131(1):e171–e181.
- , . Hospital readmission: quality indicator or statistical inevitability? Pediatrics. 2013;132(3):569–570.
- , , , et al. Children's hospitals with shorter lengths of stay do not have higher readmission rates. J Pediatr. 2013;163(4):1034–1038.e1.
- , , , , . Trends in bronchiolitis hospitalizations in the United States, 2000–2009. Pediatrics. 2013;132(1):28–36.
- , , , . Preventable adverse events in infants hospitalized with bronchiolitis. Pediatrics. 2005;116(3):603–608.
- , , , , . Infectious disease hospitalizations among infants in the United States. Pediatrics. 2008;121(2):244–252.
- . “A hospital is no place to be sick” Samuel Goldwyn (1882–1974). Arch Dis Child. 2009;94(8):565–566.
- , , , , , Variation in inpatient diagnostic testing and management of bronchiolitis. Pediatrics. 2005;115(4):878–884.
- , , , International variation in the management of infants hospitalized with respiratory syncytial virus. International RSV Study Group. Eur J Pediatr. 1998;157(3):215–220.
- , , , , . Population variation in admission rates and duration of inpatient stay for bronchiolitis in England. Arch Dis Child. 2013;98(1):57–59.
- , , , . Impact of pulse oximetry and oxygen therapy on length of stay in bronchiolitis hospitalizations. Arch Pediatr Adolesc Med. 2004;158(6):527–530.
- , , . Pulse oximetry in pediatric practice. Pediatrics. 2011;128(4):740–752.
- Scottish Intercollegiate Guidelines Network. Bronchiolitis in children (SIGN 91). In: NHS Quality Improvement Scotland. Edinburgh, Scotland: Scottish Intercollegiate Guidelines Network; 2006.
- , , , et al. Diagnosis and management of bronchiolitis. Pediatrics. 2006;118(4):1774–1793.
- , , , et al. Prospective multicenter study of children with bronchiolitis requiring mechanical ventilation. Pediatrics. 2012;130(3):e492–e500.
- , , , et al. Prospective multicenter study of viral etiology and hospital length of stay in children with severe bronchiolitis. Arch Pediatr Adolesc Med. 2012;166(8):700–706.
- , , , et al. Apnea in children hospitalized with bronchiolitis. Pediatrics. 2013;132(5):e1194–e1201.
- . Evaluation of the cardiovascular system: history and physical evaluation. In: Kliegman RM, Stanton BF, St. Geme JW III, Schor NF, Behrman RF, eds. Nelson Textbook of Pediatrics. Philadelphia, PA: Elsevier Saunders; 2011:1529–1536.
- , , , et al. Apnea in children hospitalized with bronchiolitis. Pediatrics. 2013;132(5):e1194–e1201.
- , , , et al. Respiratory viral infections in patients with chronic, obstructive pulmonary disease. J Infect. 2005;50(4):322–330.
- , , , . Evaluation of real‐time PCR for diagnosis of Bordetella pertussis infection. BMC Infect Dis. 2006;6:62.
- , , , , . Evaluation of three real‐time PCR assays for detection of Mycoplasma pneumoniae in an outbreak investigation. J Clin Microbiol. 2008;46(9):3116–3118.
- , , , et al. Normal ranges of heart rate and respiratory rate in children from birth to 18 years of age: a systematic review of observational studies. Lancet. 2011;377(9770):1011–1018.
- , , , , , . Development of heart and respiratory rate percentile curves for hospitalized children. Pediatrics. 2013;131(4):e1150–e1157.
- , . Effect of oxygen supplementation on length of stay for infants hospitalized with acute viral bronchiolitis. Pediatrics. 2008;121(3):470–475.
- , , , et al. Longitudinal assessment of hemoglobin oxygen saturation in healthy infants during the first 6 months of age. Collaborative Home Infant Monitoring Evaluation (CHIME) Study Group. J Pediatr. 1999;135(5):580–586.
- , , , et al. Prospective multicenter study of bronchiolitis: predicting safe discharges from the emergency department. Pediatrics. 2008;121(4):680–688.
- , , , et al. Delays in discharge in a tertiary care pediatric hospital. J Hosp Med. 2009;4(8):481–485.
- , , , et al. Using quality improvement to optimise paediatric discharge efficiency. BMJ Qual Saf. 2014;23(5):428–436.
- , . Bronchiolitis in infants and children: treatment; outcome; and prevention. In: Torchia M, ed. UpToDate. Alphen aan den Rijn, the Netherlands; Wolters Kluwer Health; 2013.
- , . Duration of illness in infants with bronchiolitis evaluated in the emergency department. Pediatrics. 2010;126(2):285–290.
- , , . Duration of illness in ambulatory children diagnosed with bronchiolitis. Arch Pediatr Adolesc Med. 2000;154(10):997–1000.
- , , , et al. A clinical pathway for bronchiolitis is effective in reducing readmission rates. J Pediatr. 2005;147(5):622–626.
- , , , et al. Measuring hospital quality using pediatric readmission and revisit rates. Pediatrics. 2013;132(3):429–436.
- , , , et al. Pediatric readmission prevalence and variability across hospitals. JAMA. 2013;309(4):372–380.
- , , , , , . Preventability of early readmissions at a children's hospital. Pediatrics. 2013;131(1):e171–e181.
- , . Hospital readmission: quality indicator or statistical inevitability? Pediatrics. 2013;132(3):569–570.
- , , , et al. Children's hospitals with shorter lengths of stay do not have higher readmission rates. J Pediatr. 2013;163(4):1034–1038.e1.
- , , , , . Trends in bronchiolitis hospitalizations in the United States, 2000–2009. Pediatrics. 2013;132(1):28–36.
- , , , . Preventable adverse events in infants hospitalized with bronchiolitis. Pediatrics. 2005;116(3):603–608.
© 2015 Society of Hospital Medicine
Race and Ethnicity in Bronchiolitis
Bronchiolitis is the leading cause of hospitalization for infants in the United States, costs more than $500 million annually, and has seen a 30% increase ($1.34 billion to $1.73 billion) in related hospital charges from 2000 to 2009.13 Almost all children <2 years old are infected with respiratory syncytial virus, the most common cause of bronchiolitis, with 40% developing clinically recognizable bronchiolitis and 2% becoming hospitalized with severe bronchiolitis.[4, 5] Current American Academy of Pediatrics (AAP) guidelines state that routine use of bronchodilators, corticosteroids, and chest x‐rays is not recommended, and supportive care is strongly encouraged.[6] However, a lack of consensus among clinicians persists regarding bronchiolitis management.[7, 8, 9, 10] Although minority children and those with a lower socioeconomic status (SES) in the United States are more likely to present with bronchiolitis to the emergency department (ED) and be subsequently admitted when compared to the general population,[11, 12, 13] to our knowledge, no study has yet examined if race/ethnicity is independently associated with differences in the presentation and management of severe bronchiolitis (ie, bronchiolitis causing hospitalization). Although a prior bronchiolitis‐related study reported that Hispanic children had a longer ED length of stay (LOS) than non‐Hispanic white (NHW) and non‐Hispanic black (NHB) children,[14] other studies concluded that race/ethnicity were not predictors of intensive care unit (ICU) admission or unscheduled healthcare visits post‐ED discharge.[15, 16]
Determining if race/ethnicity is independently associated with certain bronchiolitis management tendencies has implications from both a health disparities standpoint (ie, unequal care based on race/ethnicity) and from a clinical perspective (ie, the potential of certain practices, such as clinical pathways, to increase the likelihood of equitable treatment). To address this knowledge gap, we examined prospective data from a multicenter study designed to evaluate multiple factors related to bronchiolitis hospitalization.
METHODS
Study Design
We conducted a multicenter prospective cohort for 3 consecutive years (20072010) as part of the Multicenter Airway Research Collaboration (MARC), a division of the Emergency Medicine Network (EMNet) (
All patients were treated at the discretion of their physician. Inclusion criteria were hospital admission with physician diagnosis of bronchiolitis, age <2 years, and ability of the child's guardian (eg, parent) to give informed consent. Patients were enrolled within 18 hours of admission. Physician diagnosis of bronchiolitis followed the AAP definition of a child with an acute respiratory illness with some combination of rhinitis, cough, tachypnea, wheezing, crackles, and/or retractions.[6] The exclusion criteria were previous enrollment and if a patient was transferred to a participating site hospital>48 hours after the initial admission. All consent and data forms were translated into Spanish. The institutional review board at each participating site approved the study.
Data Collection
Site investigators used a standardized protocol to enroll 2207 patients admitted with bronchiolitis. Investigators conducted a structured interview that assessed patients' demographic characteristics, medical and environmental history, duration of symptoms, and details of the acute illness. Race/ethnicity was assigned by report of the child's guardian to standard US Census groups. For the purpose of this analysis, mutually exclusive race/ethnicity categories were determined: NHW, NHB, or Hispanic. Non‐Hispanic patients who identified as being both white and black were categorized as NHB. Patients were excluded from analysis if neither white or black race nor Hispanic ethnicity were reported (eg, if only Asian race was reported) because of small numbers (n=67), as were patients missing all race/ethnicity data (n=10). This resulted in a total of 2130 (97%) patients in our analytical dataset. SES was assessed with 2 variables: insurance status (public, private, none) and family income, estimated by matching patients' home ZIP codes and year of enrollment to ZIP code‐based median household annual incomes obtained from Esri Business Analyst Desktop (Esri, Redlands, CA).[17]
ED and daily clinical data, including laboratory tests (eg, complete blood count, basic metabolic panel, urine analysis, blood culture), respiratory rates, oxygen saturation, medical management, and disposition were obtained by medical chart review. Additionally, in an attempt to evaluate bronchiolitis severity at presentation, a modified respiratory distress severity score (RDSS) was calculated based on 4 assessments made during the preadmission visit (ie, ED or office visit before hospital admission): respiratory rate by age, presence of wheezing (yes or no), air entry (normal, mild difficulty, or moderate/severe), and retractions (none, mild, or moderate/severe).[18] Each component was assigned a score of 0, 1, or 2, with the exception of wheeze, which was assigned either a 0 (no wheeze) or a 2 (wheeze), and then summed for a possible total score of 0 to 8.
Last, a follow‐up telephone interview was conducted 1 week after hospital discharge for each enrolled patient. Interviews assessed acute relapse, recent symptoms, and provided additional end points for longitudinal analysis of specific symptoms. All data were manually reviewed at the EMNet Coordinating Center, and site investigators were queried about missing data and discrepancies identified.
Outcome Measures
The major outcomes of this analysis were: albuterol and corticosteroid (inhaled or systemic) use during preadmission visit and hospitalization, chest x‐rays performed at preadmission visit and hospitalization, need for intensive respiratory support (ie, receiving continuous positive airway pressure [CPAP], intubation, or ICU admission), hospital LOS 3 days, discharge on inhaled corticosteroids, and relapse of bronchiolitis requiring medical attention and a change of medication within 1 week of discharge.
Statistical Analysis
Stata 11.2 (StataCorp, College Station, TX) was used for all analyses. We examined unadjusted differences between racial/ethnic groups and clinical presentation, patient management, and outcomes using 2, Fisher exact, or Kruskal‐Wallis test, as appropriate, with results reported as proportions with 95% confidence interval (CI) or median with interquartile range (IQR). Imputed values, calculated with the Stata impute command, were used to calculate the RDSS when 1 of the 4 components was missing; patients missing more than 1 component were not assigned an RDSS value. Multivariable logistic regression was conducted to evaluate the adjusted association between race/ethnicity and the outcomes listed above. Besides race/ethnicity, all multivariable models included the demographic variables of age, sex, insurance, and median household income. Other factors were considered for inclusion if they were associated with the outcome in unadjusted analyses (P<0.20) or deemed clinically relevant. All models were adjusted for the possibility of clustering by site. Results are reported for the race/ethnicity factor as odds ratios with 95% CI.
RESULTS
Of the 2130 subjects included in this analysis, 818 (38%) were NHW, 511 (24%) were NHB, and 801 (38%) were Hispanic. The median age for children was 4.0 months (IQR, 1.88.5 months), and 60% were male. Most children were publicly insured (65%), 31% had private insurance, and approximately 4% had no insurance. The median household income defined by patient ZIP code was $51,810 (IQR, $39,916$66,272), and nearly all children (97%) had a primary care provider (PCP). Approximately 21% of all children had relevant comorbidities and 17% of children were enrolled from the ICU. Overall, the median LOS was 2 days (IQR, 14 days).
The unadjusted associations between race/ethnicity and other demographic and historical characteristics are shown in Table 1. NHB and Hispanic children were more likely to have public insurance and less likely to have relevant major comorbidities when compared to NHW children. With regard to care received the week before hospitalization, NHW children were more likely to have visited their PCP, taken corticosteroids and/or antibiotics, and were least likely to have visited an ED when compared to NHB and Hispanic children.
| White, Non‐Hispanic, n=818, % | Black, Non‐Hispanic, n=511, % | Hispanic, n=801, % | P | |
|---|---|---|---|---|
| ||||
| Demographic characteristics | ||||
| Age, months, median (IQR) | 3.2 (1.57.4) | 5.0 (1.99.2) | 4.4 (2.09.1) | <0.001 |
| Female | 39.7 | 40.9 | 40.4 | 0.91 |
| Insurance | <0.001 | |||
| Private | 56.7 | 17.0 | 13.1 | |
| Medicaid | 32.8 | 70.9 | 77.5 | |
| Other public | 6.8 | 7.6 | 4.2 | |
| None | 3.7 | 4.6 | 5.3 | |
| Median household income by ZIP code, US$, median (IQR) | $60,406 ($48,086$75,077) | $44,191 ($32,922$55,640) | $50,394 ($39,242$62,148) | <0.001 |
| Has primary care provider | 98.5 | 97.3 | 95.4 | 0.001 |
| History | ||||
| Gestational age at birth | 0.002 | |||
| <32 weeks | 4.5 | 9.2 | 6.6 | |
| 3235 weeks | 5.7 | 8.8 | 6.0 | |
| 3537 weeks | 13.3 | 10.0 | 9.7 | |
| 37 weeks | 76.0 | 71.4 | 76.9 | |
| Missing | 0.4 | 0.6 | 0.7 | |
| Weight when born | <0.001 | |||
| <3 pounds | 3.3 | 6.9 | 5.8 | |
| 34.9 pounds | 6.8 | 11.9 | 6.2 | |
| 56.9 pounds | 33.4 | 39.7 | 33.2 | |
| >7 pounds | 55.8 | 40.3 | 53.6 | |
| Missing | 0.7 | 1.4 | 1.4 | |
| Kept in an ICU, premature nursery, or any type of special‐care facility when born | 24.5 | 29.7 | 24.9 | 0.07 |
| Breast fed | 62.3 | 49.1 | 65.5 | <0.001 |
| Attends daycare | 20.7 | 25.2 | 13.3 | <0.001 |
| Number of other children (<18 years old) living in home | <0.001 | |||
| 1 | 24.2 | 25.6 | 17.0 | |
| 2 | 42.7 | 29.2 | 28.3 | |
| 3 | 33.1 | 45.2 | 54.7 | |
| Neither parent has asthma | 66.1 | 56.6 | 77.7 | <0.001 |
| Maternal smoking during pregnancy | 21.8 | 21.3 | 6.0 | <0.001 |
| Secondhand smoke exposure | 12.9 | 20.2 | 8.7 | <0.001 |
| History of wheezing | 21.1 | 25.9 | 21.8 | 0.12 |
| Ever intubated | 9.5 | 13.2 | 9.2 | 0.05 |
| Major relevant comorbidities | 23.6 | 21.1 | 18.2 | 0.03 |
| Received palivizumab (respiratory syncytial virus vaccine) | 8.7 | 12.7 | 8.9 | 0.04 |
| Received influenza vaccine this year | 20.2 | 24.7 | 21.2 | 0.15 |
| In past 12 months, admitted overnight to hospital for bronchiolitis/wheezing/reactive airway disease | 45.0 | 57.9 | 55.9 | 0.06 |
| In past 12 months, admitted overnight to hospital for pneumonia | 16.1 | 14.9 | 25.0 | 0.049 |
| Current illness (before index visit) | ||||
| Any primary care provider or clinic visits during past week | 75.0 | 44.1 | 58.3 | <0.001 |
| Any ED visits during past week | 29.1 | 30.3 | 34.6 | 0.049 |
| Over the past week used inhaled bronchodilator | 40.6 | 36.2 | 37.0 | 0.18 |
| Over the past week used inhaled/nebulized corticosteroids | 8.7 | 8.1 | 7.7 | 0.76 |
| Over the past week taken any steroid liquids or pills or shots for bronchiolitis | 12.8 | 11.7 | 8.3 | 0.012 |
| Over the past week taken antibiotics | 21.9 | 17.0 | 17.9 | 0.045 |
| Onset of difficulty breathing before admission | 0.03 | |||
| None | 2.0 | 2.2 | 2.4 | |
| <24 hours | 28.8 | 27.2 | 25.1 | |
| 13 days | 41.1 | 41.6 | 45.9 | |
| 47 days | 22.1 | 19.1 | 21.2 | |
| >7 days | 6.0 | 9.9 | 5.3 | |
| Over the past 24 hours, the level of discomfort or distress felt by the child because of symptoms | <0.001 | |||
| Mild | 15.5 | 21.3 | 18.5 | |
| Moderate | 47.8 | 39.3 | 37.2 | |
| Severe | 36.1 | 37.6 | 42.6 | |
The unadjusted associations between race/ethnicity and clinical characteristics at preadmission visit and hospital admission are shown in Table 2. RDSS values were calculated for 2130 children; 1,752 (82%) RDSS values contained all 4 components. Of those requiring imputed values, 234 (11%) were missing 1 component, and 139 (7%) were missing more than 1 component. Per RDSS scores, NHB children presented with a more severe case of bronchiolitis when compared to NHW and Hispanic children. During admission, minority children were more likely to receive nebulized albuterol and less likely to visit the ICU. NHB children received the least inpatient laboratory testing and were least likely to receive chest x‐rays during hospital admission among all groups.
| White, Non‐Hispanic, n=818, % | Black, Non‐Hispanic, n=511, % | Hispanic, n=801, % | P | |
|---|---|---|---|---|
| ||||
| Preadmission clinical findings and treatments | ||||
| Reason brought to the hospital | ||||
| Fever | 29.7 | 30.2 | 40.7 | <0.001 |
| Fussy | 32.6 | 31.6 | 28.4 | 0.18 |
| Ear Infection | 6.0 | 4.3 | 4.4 | 0.23 |
| Not drinking well | 35.0 | 27.5 | 27.2 | 0.001 |
| Cough | 54.1 | 55.5 | 61.3 | 0.009 |
| Other reasons | 29.0 | 27.6 | 23.5 | 0.04 |
| Apnea | 8.5 | 6.2 | 6.1 | 0.11 |
| Respiratory rate (breaths/minute) | 0.001 | |||
| <40 | 23.8 | 18.2 | 28.1 | |
| 4049 | 30.8 | 30.7 | 29.3 | |
| 5059 | 17.6 | 15.5 | 16.0 | |
| 60 | 27.8 | 35.6 | 26.6 | |
| Presence of cough | 83.0 | 88.0 | 87.0 | 0.045 |
| Presence of wheezing | 63.0 | 69.0 | 63.0 | 0.42 |
| Fever (temperature 100.4F) | 22.7 | 28.5 | 35.4 | <0.001 |
| Retractions | 0.002 | |||
| None | 22.9 | 19.0 | 23.0 | |
| Mild | 39.4 | 41.7 | 44.4 | |
| Moderate/severe | 28.4 | 31.1 | 28.1 | |
| Missing | 9.4 | 8.2 | 4.5 | |
| Air entry on auscultation | 0.01 | |||
| Normal | 39.6 | 31.7 | 33.6 | |
| Mild difficulty | 31.4 | 33.5 | 36.3 | |
| Moderate difficulty | 11.0 | 14.3 | 14.1 | |
| Severe difficulty | 2.0 | 2.3 | 2.6 | |
| Missing | 16.0 | 18.2 | 13.4 | |
| Oxygen saturation on room air <90 | 12.2 | 9.8 | 11.8 | 0.37 |
| Given nebulized albuterol | 53.0 | 65.0 | 63.0 | <0.001 |
| Given nebulized epinephrine | 15.4 | 20.4 | 18.4 | 0.06 |
| Given steroids, inhaled or systemic | 16.0 | 20.5 | 19.4 | 0.08 |
| Given antibiotics | 25.5 | 22.6 | 27.8 | 0.12 |
| Oral Intake | <0.001 | |||
| Adequate | 41.3 | 50.7 | 40.8 | |
| Inadequate | 43.5 | 32.5 | 47.7 | |
| Missing | 15.2 | 16.8 | 11.5 | |
| IV placed | 56.9 | 51.5 | 61.9 | 0.001 |
| Any laboratory tests | 86.3 | 91.3 | 88.0 | 0.02 |
| Chest x‐ray | 59.0 | 64.0 | 65.0 | 0.03 |
| RDSS, tertiles | <0.001 | |||
| 1 (3.00) | 36.0 | 24.0 | 34.0 | |
| 2 (3.0115.00) | 30.0 | 33.0 | 34.0 | |
| 3 (>5) | 25.0 | 36.0 | 27.0 | |
| Not calculated | 9.0 | 6.0 | 4.0 | |
| Virology results | ||||
| Respiratory syncytial virus | 75.9 | 67.5 | 71.0 | 0.003 |
| Human rhinovirus | 23.8 | 30.1 | 25.0 | 0.03 |
| Human metapneumovirus | 6.1 | 6.8 | 8.4 | 0.20 |
| Inpatient clinical findings and treatments | ||||
| Length of stay 3 days | 46.5 | 39.3 | 45.4 | 0.03 |
| Ever in observation unit | 8.6 | 7.8 | 4.0 | 0.001 |
| Ever in regular ward | 89.4 | 93.0 | 94.5 | 0.001 |
| Ever in step‐down unit | 5.0 | 3.2 | 7.8 | 0.002 |
| Ever in ICU | 20.3 | 15.0 | 15.9 | 0.02 |
| Required CPAP or intubation | 7.7 | 4.6 | 8.8 | 0.02 |
| Given nebulized albuterol | 37.6 | 48.0 | 46.7 | <0.001 |
| Given nebulized epinephrine | 10.7 | 14.9 | 13.0 | 0.07 |
| Given steroids, inhaled or systemic | 21.3 | 27.3 | 23.5 | 0.047 |
| Given antibiotics | 38.9 | 34.3 | 38.6 | 0.19 |
| Received IV fluids | 53.1 | 45.6 | 57.1 | <0.001 |
| Any laboratory tests | 52.2 | 41.6 | 51.7 | <0.001 |
| Chest x‐ray | 27.1 | 18.6 | 22.9 | 0.002 |
Discharge treatment and outcomes at 1‐week follow‐up are shown in Table 3. A total of 1771 patients (83%) were reached by telephone. No statistically significant differences between racial/ethnic groups were found regarding hospital discharge on corticosteroids and likelihood of bronchiolitis‐related relapse.
| White, Non‐Hispanic, n=818, % | Black, Non‐Hispanic, n=511, % | Hispanic, n=801, % | P | |
|---|---|---|---|---|
| Discharged on inhaled corticosteroids | 9.5 | 11.1 | 13.3 | 0.08 |
| Discharged on oral corticosteroids | 9.8 | 12.4 | 8.5 | 0.11 |
| Child's condition at 1‐week follow‐up compared to on discharge | 0.001 | |||
| Much worse/worse | 1.8 | 0.7 | 0.4 | |
| About the same | 3.4 | 6.4 | 2.5 | |
| Better | 38.2 | 39.1 | 34.2 | |
| All better | 56.6 | 53.7 | 62.9 | |
| Child's cough at 1‐week follow‐up compared to on discharge | 0.10 | |||
| Much worse/worse | 2.1 | 1.2 | 1.0 | |
| About the same | 5.0 | 8.4 | 5.2 | |
| Better | 29.4 | 31.2 | 28.6 | |
| All better | 63.5 | 59.2 | 65.2 | |
| Bronchiolitis relapse | 10.7 | 11.9 | 10.3 | 0.81 |
Given the large potential for confounding regarding our initial findings, we examined multivariable‐adjusted associations of race/ethnicity and bronchiolitis management (Table 4). Receiving albuterol during the preadmission visit and chest x‐rays during hospitalization remained significantly associated with race/ethnicity in adjusted analyses, as NHB children were most likely to receive albuterol during the preadmission visit but least likely to receive chest x‐rays during hospitalization. Several outcomes with statistically significant differences found during unadjusted analyses (eg, chest x‐rays at preadmission visit, albuterol during hospitalization, CPAP/emntubation use, ICU admission, and LOS) were not independently associated with race/ethnicity in multivariable models. By contrast, adjusted analyses revealed Hispanic children as significantly more likely to be discharged on inhaled corticosteroids when compared to NHW and NHB children; this association had borderline statistical significance (P=0.08) in the unadjusted analysis. Last, we observed no significant racial/ethnic differences with respect to corticosteroids given at preadmission visit or hospitalization as well as no differences regarding bronchiolitis‐related relapse in either unadjusted or adjusted analyses.
| White, Non‐Hispanic | Black, Non‐Hispanic | Hispanic | ||||
|---|---|---|---|---|---|---|
| OR | 95%CI | OR | 95%CI | OR | 95%CI | |
| ||||||
| Preadmission visit | ||||||
| Chest x‐ray* | 1.00 | (Reference) | 1.06 | (0.831.36) | 1.09 | (0.741.60) |
| Albuterol use | 1.00 | (Reference) | 1.58 | (1.202.07) | 1.42 | (0.892.26) |
| Steroid use, inhaled or systemic | 1.00 | (Reference) | 1.05 | (0.721.54) | 1.11 | (0.751.65) |
| During hospitalization | ||||||
| Chest x‐ray‖ | 1.00 | (Reference) | 0.66 | (0.490.90) | 0.95 | (0.601.50) |
| Albuterol use | 1.00 | (Reference) | 1.21 | (0.821.79) | 1.23 | (0.632.38) |
| Steroid use, inhaled or systemic# | 1.00 | (Reference) | 1.13 | (0.721.80) | 1.19 | (0.791.79) |
| ICU care** | 1.00 | (Reference) | 0.74 | (0.421.29) | 0.87 | (0.631.21) |
| Required CPAP/emntubation | 1.00 | (Reference) | 0.72 | (0.361.41) | 1.84 | (0.933.64) |
| Length of stay >3 days | 1.00 | (Reference) | 0.77 | (0.581.03) | 1.05 | (0.761.47) |
| Discharge | ||||||
| Discharged on inhaled steroids | 1.00 | (Reference) | 1.31 | (0.862.00) | 1.92 | (1.193.10) |
| Bronchiolitis relapse‖‖ | 1.00 | (Reference) | 1.08 | (0.621.87) | 0.96 | (0.551.65) |
DISCUSSION
It is unclear if management and treatment differences found in children with severe bronchiolitis are associated with race/ethnicity. We sought to determine if such differences exist by analyzing data from a prospective multicenter cohort study. Differences in management and treatment are discussed in the context of AAP guidelines, as they are widely used in clinical practice.
The RDSS was used to help assess severity of illness across race/ethnicity. NHB children had the highest RDSS score (ie, most severe bronchiolitis presentation) compared to NHW and Hispanic children. The reason for this difference in severity is unclear, but a potential explanation may be that minority communities lack access to care and as a result delay care and treatment for respiratory disease until care seems absolutely necessary.[19] Indeed, in our sample, minority children were less likely to visit their PCP and take corticosteroids the week before hospitalization when compared with NHW children. Our finding runs counter to a similar study by Boudreaux et al. that found no association between race/ethnicity and the clinical presentation of children with acute asthma during the preadmission setting.[20] The more severe bronchiolitis presentation among NHB children may have suggested that these children would require a longer hospital LOS (3 days). However, our multivariable analysis found no difference in LOS across racial/ethnic groups. This LOS finding is intriguing given previous studies suggesting that minorities, of diverse ages and with diverse diagnoses, were more likely to have a shorter LOS (as well as less likely to be admitted to the ICU with a similar diagnosis) when compared to nonminorities.[21, 22] Additionally, because our study sampled 16 sites, variation in clinical judgment and pediatric ICU protocol may have also played a role.[23]
Our findings also shed light on how differences in bronchiolitis management relate to AAP guidelines. According to the AAP, corticosteroid medications should not be used routinely in the management of bronchiolitis. Despite this recommendation, previous reports indicate that up to 60% of infants with severe bronchiolitis receive corticosteroid therapy.[24, 25] Our finding that Hispanic children with severe bronchiolitis were most likely to be discharged on inhaled corticosteroids is potentially concerning, as it exposes a subset of children to treatment that is not recommended. On the other hand, given the increased risk of future asthma in Hispanic communities, a higher use of inhaled corticosteroids might be seen as appropriate. Either way, our findings are inconsistent with related studies concluding that racial minority pediatric patients with asthma were less likely to receive inhaled corticosteroids.[26, 27, 28, 29] Similarly, NHB children were most likely to receive albuterol during the preadmission visit on multivariable analysis. Although a trial dose of albuterol may be common practice in treating severe bronchiolitis, AAP recommendations do not support its routine application. Increased albuterol during preadmission may have been related to an elevated bronchiolitis severity at presentation among NHB children (as indicated by the RDSS). Potential reasons for these 2 differences in treatment remain unclear. They may represent medical management efforts by discharging physicians to prescribe: (1) corticosteroids to racial/ethnic communities with a higher risk of childhood asthma; (2) albuterol to children presenting with a more severe case of bronchiolitis. These possibilities merit further study.
The AAP also recommends diagnosis of bronchiolitis on the basis of history and physical examination; laboratory and radiologic studies should not be routinely used for diagnostic purposes. Although it is possible for chest radiograph abnormalities to be consistent with bronchiolitis, there is little evidence that an abnormal finding is associated with disease severity.[30] The clinical value of diagnostic testing in children with bronchiolitis is not well supported by evidence, and limiting exposure to radiation should be a priority.[31, 32] Our analysis found that NHW and Hispanic children were more likely to receive chest x‐rays while hospitalized when compared with NHB children. Unnecessary and increased radiation exposure in children is potentially harmful and warrants intervention to minimize risk.
Establishing systematic clinical pathways in bronchiolitis management may address the practice variation found nationwide and across race/ethnicity in this study. Although clinical guidelines provide general recommendations, clinical pathways are defined treatment protocols aiming to standardize and optimize patient outcomes and clinical efficiency. The incorporation of clinical pathways into healthcare systems has increased recently as a result of their favorable association with medical complications, healthcare costs, and LOS.[33] With respect to bronchiolitis, implementation of clinical pathways has proven to reduce use of inappropriate therapies, decrease risk of bronchiolitis‐related hospital readmission, and help with discharge planning.[30, 34, 35]
Notwithstanding the differences found in this study, management of children with bronchiolitis was, in many respects, comparable across racial/ethnic groups. For example, our multivariable analysis found no significant differences across racial/ethnic groups with respect to chest x‐rays and corticosteroid use during the preadmission visit, administration of albuterol or corticosteroids during hospitalization, use of CPAP/emntubation, ICU admission, hospital LOS, or likelihood of a bronchiolitis‐related relapse. The general lack of race/ethnic differences is consistent with similar research on inpatient management of acute asthma.[36]
This study has potential limitations. The hospitals participating in the study are predominantly urban, academically affiliated hospitals. This may result in findings that are less generalizable to rural and community hospitals. Second, the race/ethnicity classification used does not take into consideration the diversity and complexity of defining race/ethnicity in the United States. Third, bronchiolitis is defined as a clinical diagnosis that can encapsulate multiple lower respiratory infection diagnoses. As a result, there may have been variability in clinical and institutional practice. An additional limitation was utilizing RDSS to assess bronchiolitis severity. Although there is currently no validated, universally accepted score to assess bronchiolitis severity, several scores are available in the literature with varying performance. Last, the ZIP code‐based median household incomes used to assess SES are higher than federal data in similar geographic locations, potentially resulting in findings that are less generalizable.
CONCLUSION
This multicenter prospective cohort study found several differences in bronchiolitis presentation and management among children stratified by race/ethnicity in 16 geographically dispersed sites after controlling for multiple factors including SES. Our analysis showed that, when compared to NHW children, NHB children were more likely to be given albuterol during the preadmission visit and less likely to receive chest x‐rays as inpatients; Hispanic children were more likely to be discharged on inhaled corticosteroids. These differences are concerning for 2 reasons: (1) based on current evidence, race/ethnicity should not affect care in children with severe bronchiolitis; and (2) the observed differences in diagnostic testing and treatment are not recommended by the evidence‐based AAP guidelines. It is also important to note that these differences do not demonstrate that a specific race/ethnicity received better or worse clinical care. The goal of this analysis was not to determine the effectiveness of certain management tendencies in children with severe bronchiolitis, but rather to examine differences in the presentation and management of children from different racial/ethnic groups. The causes for the observed findings require further study. In the meantime, we suggest increasing the number of hospitals that incorporate clinical care pathways for severe bronchiolitis to control variation in practice and limit the impact that race/ethnicity may have in the provision of services.
Acknowledgements
The authors thank the MARC‐30 investigators for their ongoing dedication to bronchiolitis research.
Disclosures: This study was supported by the grant U01 AI‐67693 (Camargo) from the National Institutes of Health (Bethesda, MD). The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Allergy and Infectious Diseases or the National Institutes of Health. The authors have no financial relationships relevant to this article or conflicts of interest to disclose. Mr. Santiago conceptualized the analysis, interpreted the data, drafted the initial manuscript, and approved the final manuscript as submitted. Dr. Mansbach conceptualized and designed the initial study, coordinated data collection at 1 of the sites, critically reviewed the manuscript, and approved the final manuscript as submitted. Dr. Chou was responsible for analysis and interpretation of data, critically reviewed the manuscript, and approved the final manuscript as submitted. Dr. Delgado coordinated data collection at 1 of the sites, critically reviewed the manuscript, and approved the final manuscript as submitted. Dr. Piedra conceptualized and designed the initial study, coordinated virology testing, critically reviewed the manuscript, and approved the final manuscript as submitted. Ms. Sullivan coordinated data collection at all sites, critically reviewed the manuscript, and approved the final manuscript as submitted. Ms. Espinola was responsible for data management, analysis and interpretation of data, drafting of the initial manuscript, and approved the final manuscript as submitted. Dr. Camargo conceptualized and designed the initial study, assisted with data analysis and interpretation of data, critically reviewed the manuscript, and approved the final manuscript as submitted.
APPENDIX
| Principal Investigators at the 16 Participating Sites in MARC‐30 | |
| Besh Barcega, MD | Loma Linda University Children's Hospital, Loma Linda, CA |
| John Cheng, MD and Carlos Delgado, MD | Children's Healthcare of Atlanta at Egleston, Atlanta, GA |
| Dorothy Damore, MD and Nikhil Shah, MD | New York Presbyterian Hospital, New York, NY |
| Haitham Haddad, MD | Rainbow Babies & Children's Hospital, Cleveland, OH |
| Paul Hain, MD and Mark Riederer, MD | Monroe Carell Jr. Children's Hospital at Vanderbilt, Nashville, TN |
| Frank LoVecchio, DO | Maricopa Medical Center, Phoenix, AZ |
| Charles Macias, MD, MPH | Texas Children's Hospital, Houston, TX |
| Jonathan Mansbach, MD, MPH | Boston Children's Hospital, Boston, MA |
| Eugene Mowad, MD | Akron Children's Hospital, Akron, OH |
| Brian Pate, MD | Children's Mercy Hospital & Clinics, Kansas City, MO |
| M. Jason Sanders, MD | Children's Memorial Hermann Hospital, Houston, TX |
| Alan Schroeder, MD | Santa Clara Valley Medical Center, San Jose, CA |
| Michelle Stevenson, MD, MS | Kosair Children's Hospital, Louisville, KY |
| Erin Stucky Fisher, MD | Rady Children's Hospital, San Diego, CA |
| Stephen Teach, MD, MPH | Children's National Medical Center, Washington, DC |
| Lisa Zaoutis, MD | Children's Hospital of Philadelphia, Philadelphia, PA |
- , , . Direct medical costs of bronchiolitis‐related hospitalizations in the United States. Pediatrics. 2006;118(6):2418–2423.
- , , , , . Trends in bronchiolitis hospitalizations in the United States, 2000–2009. Pediatrics. 2013;132(1):28–36.
- , , , , . Infectious disease hospitalizations among infants in the United States. Pediatrics. 2008;121(2):244–252.
- , . Respiratory syncytial virus. Curr Probl Pediatr. 1993;23(2):50–79.
- , , , , . Rates of hospitalization for respiratory syncytial virus infection among children in Medicaid. J Pediatr. 2000;137(6):865–870.
- American Academy of Pediatrics Subcommittee on Diagnosis and Management of Bronchiolitis. Diagnosis and management of bronchiolitis. Pediatrics. 2006;118(4):1774–1793.
- , , . US outpatient office visits for bronchiolitis, 1993–2004. Ambul Pediatr. 2007;7(4):304–307.
- , , , et al. Study of interobserver reliability in clinical assessment of RSV lower respiratory illness: a pediatric investigators collaborative network for infections in Canada (PICNIC) study. Pediatr Pulmonol. 1996;22(1):23–27.
- , , , , . Effect of practice variation on resource utilization in infants hospitalized for viral lower respiratory illness. Pediatrics. 2001;108(4):851–855.
- , , . Bronchiolitis in US emergency departments 1992 to 2000: epidemiology and practice variation. Pediatr Emerg Care. 2005;21(4):242–247.
- , . Recent trends in severe respiratory syncytial virus (RSV) among US infants, 1997 to 2000. J Pediatr. 2003; 143(5 suppl):S127–S132.
- , , , , . Risk of respiratory syncytial virus infection for infants from low‐income families in relationship to age, sex, ethnic group, and maternal antibody level. J Pediatr. 1981;98(5):708–715.
- , , . Socioeconomic environmental factors and hospitalization for acute bronchiolitis during infancy. Acta Paediatr. 2002;91(3):335–338.
- , , , , . Factors associated with longer emergency department length of stay for children with bronchiolitis: a prospective multicenter study. Pediatr Emerg Care. 2009;25(10):636–641.
- , , , , . Prospective multicenter bronchiolitis study: predicting intensive care unit admissions. Acad Emerg Med. 2008;15(10):887–894.
- , , , , . Prospective multicenter study of bronchiolitis: predictors of an unscheduled visit after discharge from the emergency department. Acad Emerg Med. 2010;17(4):376–382.
- Esri. Demographic, consumer, and business data. Available at: http://www.esri.com/data/esri_data/demographic‐overview/demographic. Accessed July 25, 2013.
- , , . A randomized trial of home oxygen therapy from the emergency department for acute bronchiolitis. Pediatrics. 2006;117(3):633–640.
- , , , , , . Adherence to therapy and access to care: the relationship to excess asthma morbidity in African‐American children. Pediatr Asthma Aller. 1994;8(3):179–184.
- , , , . Race/ethnicity and asthma among children presenting to the emergency department: differences in disease severity and management. Pediatrics. 2003;111(5 pt 1):e615–e621.
- , , , . Relationship between patient race and the intensity of hospital services. Med Care. 1987;25(7):592–603.
- , , , , . African‐American and white patients admitted to the intensive care unit: is there a difference in therapy and outcome? Crit Care Med. 1995;23(4):626–636.
- , , . Acute severe asthma: differences in therapies and outcomes among pediatric intensive care units. Crit Care Med. 2002;30(3):581–585.
- , , , . International variation in the management of infants hospitalized with respiratory syncytial virus. International RSV Study Group. Eur J Pediatr. 1998;157(3):215–220.
- , , , , . Effect of practice variation on resource utilization in infants for viral lower respiratory illness. Pediatrics. 2001;108:851–855.
- , , , , , . Impact of site of care, race, and Hispanic ethnicity on medication use for childhood asthma. Pediatrics. 2002;109(1):E1.
- , , , et al. Racial/ethnic variation in asthma status and management practices among children in managed Medicaid. Pediatrics. 2002;109(5):857–865.
- , , , . Treatment adherence among low‐income children with asthma. J Pediatr Psychol. 1998;23(6):345–349.
- American Academy of Pediatrics Steering Committee on Quality Improvement and Management. Classifying recommendations for clinical practice guidelines. Pediatrics. 2004;114(3):874–877.
- , , . Randomised controlled trial of clinical outcome after chest radiograph in ambulatory acute lower‐respiratory infection in children. Lancet. 1998;351(9100):404–408.
- . Cancer risks following diagnostic and therapeutic radiation exposure in children. Pediatr Radiol. 2006;36(suppl 2):121–125.
- , , , et al. Clinical pathways: effects on professional practice, patient outcomes, length of stay and hospital costs. Cochrane Database Syst Rev. 2010;(3):CD006632.
- , , . An evidence‐based clinical pathway for bronchiolitis safely reduces antibiotic overuse. Am J Med Qual. 2002;17(5):195–199.
- , , , et al. A clinical pathway for bronchiolitis is effective in reducing readmission rates. J Pediatr. 2005;147(5):622–626.
- , , , et al. Decreasing unnecessary utilization in acute bronchiolitis care: results from the value in inpatient pediatrics network. J Hosp Med. 2013;8(1):25–30.
- , , . Race/Ethnicity differences in the inpatient management of acute asthma in the United States. Chest. 2009;135(6):1527–1534.
Bronchiolitis is the leading cause of hospitalization for infants in the United States, costs more than $500 million annually, and has seen a 30% increase ($1.34 billion to $1.73 billion) in related hospital charges from 2000 to 2009.13 Almost all children <2 years old are infected with respiratory syncytial virus, the most common cause of bronchiolitis, with 40% developing clinically recognizable bronchiolitis and 2% becoming hospitalized with severe bronchiolitis.[4, 5] Current American Academy of Pediatrics (AAP) guidelines state that routine use of bronchodilators, corticosteroids, and chest x‐rays is not recommended, and supportive care is strongly encouraged.[6] However, a lack of consensus among clinicians persists regarding bronchiolitis management.[7, 8, 9, 10] Although minority children and those with a lower socioeconomic status (SES) in the United States are more likely to present with bronchiolitis to the emergency department (ED) and be subsequently admitted when compared to the general population,[11, 12, 13] to our knowledge, no study has yet examined if race/ethnicity is independently associated with differences in the presentation and management of severe bronchiolitis (ie, bronchiolitis causing hospitalization). Although a prior bronchiolitis‐related study reported that Hispanic children had a longer ED length of stay (LOS) than non‐Hispanic white (NHW) and non‐Hispanic black (NHB) children,[14] other studies concluded that race/ethnicity were not predictors of intensive care unit (ICU) admission or unscheduled healthcare visits post‐ED discharge.[15, 16]
Determining if race/ethnicity is independently associated with certain bronchiolitis management tendencies has implications from both a health disparities standpoint (ie, unequal care based on race/ethnicity) and from a clinical perspective (ie, the potential of certain practices, such as clinical pathways, to increase the likelihood of equitable treatment). To address this knowledge gap, we examined prospective data from a multicenter study designed to evaluate multiple factors related to bronchiolitis hospitalization.
METHODS
Study Design
We conducted a multicenter prospective cohort for 3 consecutive years (20072010) as part of the Multicenter Airway Research Collaboration (MARC), a division of the Emergency Medicine Network (EMNet) (
All patients were treated at the discretion of their physician. Inclusion criteria were hospital admission with physician diagnosis of bronchiolitis, age <2 years, and ability of the child's guardian (eg, parent) to give informed consent. Patients were enrolled within 18 hours of admission. Physician diagnosis of bronchiolitis followed the AAP definition of a child with an acute respiratory illness with some combination of rhinitis, cough, tachypnea, wheezing, crackles, and/or retractions.[6] The exclusion criteria were previous enrollment and if a patient was transferred to a participating site hospital>48 hours after the initial admission. All consent and data forms were translated into Spanish. The institutional review board at each participating site approved the study.
Data Collection
Site investigators used a standardized protocol to enroll 2207 patients admitted with bronchiolitis. Investigators conducted a structured interview that assessed patients' demographic characteristics, medical and environmental history, duration of symptoms, and details of the acute illness. Race/ethnicity was assigned by report of the child's guardian to standard US Census groups. For the purpose of this analysis, mutually exclusive race/ethnicity categories were determined: NHW, NHB, or Hispanic. Non‐Hispanic patients who identified as being both white and black were categorized as NHB. Patients were excluded from analysis if neither white or black race nor Hispanic ethnicity were reported (eg, if only Asian race was reported) because of small numbers (n=67), as were patients missing all race/ethnicity data (n=10). This resulted in a total of 2130 (97%) patients in our analytical dataset. SES was assessed with 2 variables: insurance status (public, private, none) and family income, estimated by matching patients' home ZIP codes and year of enrollment to ZIP code‐based median household annual incomes obtained from Esri Business Analyst Desktop (Esri, Redlands, CA).[17]
ED and daily clinical data, including laboratory tests (eg, complete blood count, basic metabolic panel, urine analysis, blood culture), respiratory rates, oxygen saturation, medical management, and disposition were obtained by medical chart review. Additionally, in an attempt to evaluate bronchiolitis severity at presentation, a modified respiratory distress severity score (RDSS) was calculated based on 4 assessments made during the preadmission visit (ie, ED or office visit before hospital admission): respiratory rate by age, presence of wheezing (yes or no), air entry (normal, mild difficulty, or moderate/severe), and retractions (none, mild, or moderate/severe).[18] Each component was assigned a score of 0, 1, or 2, with the exception of wheeze, which was assigned either a 0 (no wheeze) or a 2 (wheeze), and then summed for a possible total score of 0 to 8.
Last, a follow‐up telephone interview was conducted 1 week after hospital discharge for each enrolled patient. Interviews assessed acute relapse, recent symptoms, and provided additional end points for longitudinal analysis of specific symptoms. All data were manually reviewed at the EMNet Coordinating Center, and site investigators were queried about missing data and discrepancies identified.
Outcome Measures
The major outcomes of this analysis were: albuterol and corticosteroid (inhaled or systemic) use during preadmission visit and hospitalization, chest x‐rays performed at preadmission visit and hospitalization, need for intensive respiratory support (ie, receiving continuous positive airway pressure [CPAP], intubation, or ICU admission), hospital LOS 3 days, discharge on inhaled corticosteroids, and relapse of bronchiolitis requiring medical attention and a change of medication within 1 week of discharge.
Statistical Analysis
Stata 11.2 (StataCorp, College Station, TX) was used for all analyses. We examined unadjusted differences between racial/ethnic groups and clinical presentation, patient management, and outcomes using 2, Fisher exact, or Kruskal‐Wallis test, as appropriate, with results reported as proportions with 95% confidence interval (CI) or median with interquartile range (IQR). Imputed values, calculated with the Stata impute command, were used to calculate the RDSS when 1 of the 4 components was missing; patients missing more than 1 component were not assigned an RDSS value. Multivariable logistic regression was conducted to evaluate the adjusted association between race/ethnicity and the outcomes listed above. Besides race/ethnicity, all multivariable models included the demographic variables of age, sex, insurance, and median household income. Other factors were considered for inclusion if they were associated with the outcome in unadjusted analyses (P<0.20) or deemed clinically relevant. All models were adjusted for the possibility of clustering by site. Results are reported for the race/ethnicity factor as odds ratios with 95% CI.
RESULTS
Of the 2130 subjects included in this analysis, 818 (38%) were NHW, 511 (24%) were NHB, and 801 (38%) were Hispanic. The median age for children was 4.0 months (IQR, 1.88.5 months), and 60% were male. Most children were publicly insured (65%), 31% had private insurance, and approximately 4% had no insurance. The median household income defined by patient ZIP code was $51,810 (IQR, $39,916$66,272), and nearly all children (97%) had a primary care provider (PCP). Approximately 21% of all children had relevant comorbidities and 17% of children were enrolled from the ICU. Overall, the median LOS was 2 days (IQR, 14 days).
The unadjusted associations between race/ethnicity and other demographic and historical characteristics are shown in Table 1. NHB and Hispanic children were more likely to have public insurance and less likely to have relevant major comorbidities when compared to NHW children. With regard to care received the week before hospitalization, NHW children were more likely to have visited their PCP, taken corticosteroids and/or antibiotics, and were least likely to have visited an ED when compared to NHB and Hispanic children.
| White, Non‐Hispanic, n=818, % | Black, Non‐Hispanic, n=511, % | Hispanic, n=801, % | P | |
|---|---|---|---|---|
| ||||
| Demographic characteristics | ||||
| Age, months, median (IQR) | 3.2 (1.57.4) | 5.0 (1.99.2) | 4.4 (2.09.1) | <0.001 |
| Female | 39.7 | 40.9 | 40.4 | 0.91 |
| Insurance | <0.001 | |||
| Private | 56.7 | 17.0 | 13.1 | |
| Medicaid | 32.8 | 70.9 | 77.5 | |
| Other public | 6.8 | 7.6 | 4.2 | |
| None | 3.7 | 4.6 | 5.3 | |
| Median household income by ZIP code, US$, median (IQR) | $60,406 ($48,086$75,077) | $44,191 ($32,922$55,640) | $50,394 ($39,242$62,148) | <0.001 |
| Has primary care provider | 98.5 | 97.3 | 95.4 | 0.001 |
| History | ||||
| Gestational age at birth | 0.002 | |||
| <32 weeks | 4.5 | 9.2 | 6.6 | |
| 3235 weeks | 5.7 | 8.8 | 6.0 | |
| 3537 weeks | 13.3 | 10.0 | 9.7 | |
| 37 weeks | 76.0 | 71.4 | 76.9 | |
| Missing | 0.4 | 0.6 | 0.7 | |
| Weight when born | <0.001 | |||
| <3 pounds | 3.3 | 6.9 | 5.8 | |
| 34.9 pounds | 6.8 | 11.9 | 6.2 | |
| 56.9 pounds | 33.4 | 39.7 | 33.2 | |
| >7 pounds | 55.8 | 40.3 | 53.6 | |
| Missing | 0.7 | 1.4 | 1.4 | |
| Kept in an ICU, premature nursery, or any type of special‐care facility when born | 24.5 | 29.7 | 24.9 | 0.07 |
| Breast fed | 62.3 | 49.1 | 65.5 | <0.001 |
| Attends daycare | 20.7 | 25.2 | 13.3 | <0.001 |
| Number of other children (<18 years old) living in home | <0.001 | |||
| 1 | 24.2 | 25.6 | 17.0 | |
| 2 | 42.7 | 29.2 | 28.3 | |
| 3 | 33.1 | 45.2 | 54.7 | |
| Neither parent has asthma | 66.1 | 56.6 | 77.7 | <0.001 |
| Maternal smoking during pregnancy | 21.8 | 21.3 | 6.0 | <0.001 |
| Secondhand smoke exposure | 12.9 | 20.2 | 8.7 | <0.001 |
| History of wheezing | 21.1 | 25.9 | 21.8 | 0.12 |
| Ever intubated | 9.5 | 13.2 | 9.2 | 0.05 |
| Major relevant comorbidities | 23.6 | 21.1 | 18.2 | 0.03 |
| Received palivizumab (respiratory syncytial virus vaccine) | 8.7 | 12.7 | 8.9 | 0.04 |
| Received influenza vaccine this year | 20.2 | 24.7 | 21.2 | 0.15 |
| In past 12 months, admitted overnight to hospital for bronchiolitis/wheezing/reactive airway disease | 45.0 | 57.9 | 55.9 | 0.06 |
| In past 12 months, admitted overnight to hospital for pneumonia | 16.1 | 14.9 | 25.0 | 0.049 |
| Current illness (before index visit) | ||||
| Any primary care provider or clinic visits during past week | 75.0 | 44.1 | 58.3 | <0.001 |
| Any ED visits during past week | 29.1 | 30.3 | 34.6 | 0.049 |
| Over the past week used inhaled bronchodilator | 40.6 | 36.2 | 37.0 | 0.18 |
| Over the past week used inhaled/nebulized corticosteroids | 8.7 | 8.1 | 7.7 | 0.76 |
| Over the past week taken any steroid liquids or pills or shots for bronchiolitis | 12.8 | 11.7 | 8.3 | 0.012 |
| Over the past week taken antibiotics | 21.9 | 17.0 | 17.9 | 0.045 |
| Onset of difficulty breathing before admission | 0.03 | |||
| None | 2.0 | 2.2 | 2.4 | |
| <24 hours | 28.8 | 27.2 | 25.1 | |
| 13 days | 41.1 | 41.6 | 45.9 | |
| 47 days | 22.1 | 19.1 | 21.2 | |
| >7 days | 6.0 | 9.9 | 5.3 | |
| Over the past 24 hours, the level of discomfort or distress felt by the child because of symptoms | <0.001 | |||
| Mild | 15.5 | 21.3 | 18.5 | |
| Moderate | 47.8 | 39.3 | 37.2 | |
| Severe | 36.1 | 37.6 | 42.6 | |
The unadjusted associations between race/ethnicity and clinical characteristics at preadmission visit and hospital admission are shown in Table 2. RDSS values were calculated for 2130 children; 1,752 (82%) RDSS values contained all 4 components. Of those requiring imputed values, 234 (11%) were missing 1 component, and 139 (7%) were missing more than 1 component. Per RDSS scores, NHB children presented with a more severe case of bronchiolitis when compared to NHW and Hispanic children. During admission, minority children were more likely to receive nebulized albuterol and less likely to visit the ICU. NHB children received the least inpatient laboratory testing and were least likely to receive chest x‐rays during hospital admission among all groups.
| White, Non‐Hispanic, n=818, % | Black, Non‐Hispanic, n=511, % | Hispanic, n=801, % | P | |
|---|---|---|---|---|
| ||||
| Preadmission clinical findings and treatments | ||||
| Reason brought to the hospital | ||||
| Fever | 29.7 | 30.2 | 40.7 | <0.001 |
| Fussy | 32.6 | 31.6 | 28.4 | 0.18 |
| Ear Infection | 6.0 | 4.3 | 4.4 | 0.23 |
| Not drinking well | 35.0 | 27.5 | 27.2 | 0.001 |
| Cough | 54.1 | 55.5 | 61.3 | 0.009 |
| Other reasons | 29.0 | 27.6 | 23.5 | 0.04 |
| Apnea | 8.5 | 6.2 | 6.1 | 0.11 |
| Respiratory rate (breaths/minute) | 0.001 | |||
| <40 | 23.8 | 18.2 | 28.1 | |
| 4049 | 30.8 | 30.7 | 29.3 | |
| 5059 | 17.6 | 15.5 | 16.0 | |
| 60 | 27.8 | 35.6 | 26.6 | |
| Presence of cough | 83.0 | 88.0 | 87.0 | 0.045 |
| Presence of wheezing | 63.0 | 69.0 | 63.0 | 0.42 |
| Fever (temperature 100.4F) | 22.7 | 28.5 | 35.4 | <0.001 |
| Retractions | 0.002 | |||
| None | 22.9 | 19.0 | 23.0 | |
| Mild | 39.4 | 41.7 | 44.4 | |
| Moderate/severe | 28.4 | 31.1 | 28.1 | |
| Missing | 9.4 | 8.2 | 4.5 | |
| Air entry on auscultation | 0.01 | |||
| Normal | 39.6 | 31.7 | 33.6 | |
| Mild difficulty | 31.4 | 33.5 | 36.3 | |
| Moderate difficulty | 11.0 | 14.3 | 14.1 | |
| Severe difficulty | 2.0 | 2.3 | 2.6 | |
| Missing | 16.0 | 18.2 | 13.4 | |
| Oxygen saturation on room air <90 | 12.2 | 9.8 | 11.8 | 0.37 |
| Given nebulized albuterol | 53.0 | 65.0 | 63.0 | <0.001 |
| Given nebulized epinephrine | 15.4 | 20.4 | 18.4 | 0.06 |
| Given steroids, inhaled or systemic | 16.0 | 20.5 | 19.4 | 0.08 |
| Given antibiotics | 25.5 | 22.6 | 27.8 | 0.12 |
| Oral Intake | <0.001 | |||
| Adequate | 41.3 | 50.7 | 40.8 | |
| Inadequate | 43.5 | 32.5 | 47.7 | |
| Missing | 15.2 | 16.8 | 11.5 | |
| IV placed | 56.9 | 51.5 | 61.9 | 0.001 |
| Any laboratory tests | 86.3 | 91.3 | 88.0 | 0.02 |
| Chest x‐ray | 59.0 | 64.0 | 65.0 | 0.03 |
| RDSS, tertiles | <0.001 | |||
| 1 (3.00) | 36.0 | 24.0 | 34.0 | |
| 2 (3.0115.00) | 30.0 | 33.0 | 34.0 | |
| 3 (>5) | 25.0 | 36.0 | 27.0 | |
| Not calculated | 9.0 | 6.0 | 4.0 | |
| Virology results | ||||
| Respiratory syncytial virus | 75.9 | 67.5 | 71.0 | 0.003 |
| Human rhinovirus | 23.8 | 30.1 | 25.0 | 0.03 |
| Human metapneumovirus | 6.1 | 6.8 | 8.4 | 0.20 |
| Inpatient clinical findings and treatments | ||||
| Length of stay 3 days | 46.5 | 39.3 | 45.4 | 0.03 |
| Ever in observation unit | 8.6 | 7.8 | 4.0 | 0.001 |
| Ever in regular ward | 89.4 | 93.0 | 94.5 | 0.001 |
| Ever in step‐down unit | 5.0 | 3.2 | 7.8 | 0.002 |
| Ever in ICU | 20.3 | 15.0 | 15.9 | 0.02 |
| Required CPAP or intubation | 7.7 | 4.6 | 8.8 | 0.02 |
| Given nebulized albuterol | 37.6 | 48.0 | 46.7 | <0.001 |
| Given nebulized epinephrine | 10.7 | 14.9 | 13.0 | 0.07 |
| Given steroids, inhaled or systemic | 21.3 | 27.3 | 23.5 | 0.047 |
| Given antibiotics | 38.9 | 34.3 | 38.6 | 0.19 |
| Received IV fluids | 53.1 | 45.6 | 57.1 | <0.001 |
| Any laboratory tests | 52.2 | 41.6 | 51.7 | <0.001 |
| Chest x‐ray | 27.1 | 18.6 | 22.9 | 0.002 |
Discharge treatment and outcomes at 1‐week follow‐up are shown in Table 3. A total of 1771 patients (83%) were reached by telephone. No statistically significant differences between racial/ethnic groups were found regarding hospital discharge on corticosteroids and likelihood of bronchiolitis‐related relapse.
| White, Non‐Hispanic, n=818, % | Black, Non‐Hispanic, n=511, % | Hispanic, n=801, % | P | |
|---|---|---|---|---|
| Discharged on inhaled corticosteroids | 9.5 | 11.1 | 13.3 | 0.08 |
| Discharged on oral corticosteroids | 9.8 | 12.4 | 8.5 | 0.11 |
| Child's condition at 1‐week follow‐up compared to on discharge | 0.001 | |||
| Much worse/worse | 1.8 | 0.7 | 0.4 | |
| About the same | 3.4 | 6.4 | 2.5 | |
| Better | 38.2 | 39.1 | 34.2 | |
| All better | 56.6 | 53.7 | 62.9 | |
| Child's cough at 1‐week follow‐up compared to on discharge | 0.10 | |||
| Much worse/worse | 2.1 | 1.2 | 1.0 | |
| About the same | 5.0 | 8.4 | 5.2 | |
| Better | 29.4 | 31.2 | 28.6 | |
| All better | 63.5 | 59.2 | 65.2 | |
| Bronchiolitis relapse | 10.7 | 11.9 | 10.3 | 0.81 |
Given the large potential for confounding regarding our initial findings, we examined multivariable‐adjusted associations of race/ethnicity and bronchiolitis management (Table 4). Receiving albuterol during the preadmission visit and chest x‐rays during hospitalization remained significantly associated with race/ethnicity in adjusted analyses, as NHB children were most likely to receive albuterol during the preadmission visit but least likely to receive chest x‐rays during hospitalization. Several outcomes with statistically significant differences found during unadjusted analyses (eg, chest x‐rays at preadmission visit, albuterol during hospitalization, CPAP/emntubation use, ICU admission, and LOS) were not independently associated with race/ethnicity in multivariable models. By contrast, adjusted analyses revealed Hispanic children as significantly more likely to be discharged on inhaled corticosteroids when compared to NHW and NHB children; this association had borderline statistical significance (P=0.08) in the unadjusted analysis. Last, we observed no significant racial/ethnic differences with respect to corticosteroids given at preadmission visit or hospitalization as well as no differences regarding bronchiolitis‐related relapse in either unadjusted or adjusted analyses.
| White, Non‐Hispanic | Black, Non‐Hispanic | Hispanic | ||||
|---|---|---|---|---|---|---|
| OR | 95%CI | OR | 95%CI | OR | 95%CI | |
| ||||||
| Preadmission visit | ||||||
| Chest x‐ray* | 1.00 | (Reference) | 1.06 | (0.831.36) | 1.09 | (0.741.60) |
| Albuterol use | 1.00 | (Reference) | 1.58 | (1.202.07) | 1.42 | (0.892.26) |
| Steroid use, inhaled or systemic | 1.00 | (Reference) | 1.05 | (0.721.54) | 1.11 | (0.751.65) |
| During hospitalization | ||||||
| Chest x‐ray‖ | 1.00 | (Reference) | 0.66 | (0.490.90) | 0.95 | (0.601.50) |
| Albuterol use | 1.00 | (Reference) | 1.21 | (0.821.79) | 1.23 | (0.632.38) |
| Steroid use, inhaled or systemic# | 1.00 | (Reference) | 1.13 | (0.721.80) | 1.19 | (0.791.79) |
| ICU care** | 1.00 | (Reference) | 0.74 | (0.421.29) | 0.87 | (0.631.21) |
| Required CPAP/emntubation | 1.00 | (Reference) | 0.72 | (0.361.41) | 1.84 | (0.933.64) |
| Length of stay >3 days | 1.00 | (Reference) | 0.77 | (0.581.03) | 1.05 | (0.761.47) |
| Discharge | ||||||
| Discharged on inhaled steroids | 1.00 | (Reference) | 1.31 | (0.862.00) | 1.92 | (1.193.10) |
| Bronchiolitis relapse‖‖ | 1.00 | (Reference) | 1.08 | (0.621.87) | 0.96 | (0.551.65) |
DISCUSSION
It is unclear if management and treatment differences found in children with severe bronchiolitis are associated with race/ethnicity. We sought to determine if such differences exist by analyzing data from a prospective multicenter cohort study. Differences in management and treatment are discussed in the context of AAP guidelines, as they are widely used in clinical practice.
The RDSS was used to help assess severity of illness across race/ethnicity. NHB children had the highest RDSS score (ie, most severe bronchiolitis presentation) compared to NHW and Hispanic children. The reason for this difference in severity is unclear, but a potential explanation may be that minority communities lack access to care and as a result delay care and treatment for respiratory disease until care seems absolutely necessary.[19] Indeed, in our sample, minority children were less likely to visit their PCP and take corticosteroids the week before hospitalization when compared with NHW children. Our finding runs counter to a similar study by Boudreaux et al. that found no association between race/ethnicity and the clinical presentation of children with acute asthma during the preadmission setting.[20] The more severe bronchiolitis presentation among NHB children may have suggested that these children would require a longer hospital LOS (3 days). However, our multivariable analysis found no difference in LOS across racial/ethnic groups. This LOS finding is intriguing given previous studies suggesting that minorities, of diverse ages and with diverse diagnoses, were more likely to have a shorter LOS (as well as less likely to be admitted to the ICU with a similar diagnosis) when compared to nonminorities.[21, 22] Additionally, because our study sampled 16 sites, variation in clinical judgment and pediatric ICU protocol may have also played a role.[23]
Our findings also shed light on how differences in bronchiolitis management relate to AAP guidelines. According to the AAP, corticosteroid medications should not be used routinely in the management of bronchiolitis. Despite this recommendation, previous reports indicate that up to 60% of infants with severe bronchiolitis receive corticosteroid therapy.[24, 25] Our finding that Hispanic children with severe bronchiolitis were most likely to be discharged on inhaled corticosteroids is potentially concerning, as it exposes a subset of children to treatment that is not recommended. On the other hand, given the increased risk of future asthma in Hispanic communities, a higher use of inhaled corticosteroids might be seen as appropriate. Either way, our findings are inconsistent with related studies concluding that racial minority pediatric patients with asthma were less likely to receive inhaled corticosteroids.[26, 27, 28, 29] Similarly, NHB children were most likely to receive albuterol during the preadmission visit on multivariable analysis. Although a trial dose of albuterol may be common practice in treating severe bronchiolitis, AAP recommendations do not support its routine application. Increased albuterol during preadmission may have been related to an elevated bronchiolitis severity at presentation among NHB children (as indicated by the RDSS). Potential reasons for these 2 differences in treatment remain unclear. They may represent medical management efforts by discharging physicians to prescribe: (1) corticosteroids to racial/ethnic communities with a higher risk of childhood asthma; (2) albuterol to children presenting with a more severe case of bronchiolitis. These possibilities merit further study.
The AAP also recommends diagnosis of bronchiolitis on the basis of history and physical examination; laboratory and radiologic studies should not be routinely used for diagnostic purposes. Although it is possible for chest radiograph abnormalities to be consistent with bronchiolitis, there is little evidence that an abnormal finding is associated with disease severity.[30] The clinical value of diagnostic testing in children with bronchiolitis is not well supported by evidence, and limiting exposure to radiation should be a priority.[31, 32] Our analysis found that NHW and Hispanic children were more likely to receive chest x‐rays while hospitalized when compared with NHB children. Unnecessary and increased radiation exposure in children is potentially harmful and warrants intervention to minimize risk.
Establishing systematic clinical pathways in bronchiolitis management may address the practice variation found nationwide and across race/ethnicity in this study. Although clinical guidelines provide general recommendations, clinical pathways are defined treatment protocols aiming to standardize and optimize patient outcomes and clinical efficiency. The incorporation of clinical pathways into healthcare systems has increased recently as a result of their favorable association with medical complications, healthcare costs, and LOS.[33] With respect to bronchiolitis, implementation of clinical pathways has proven to reduce use of inappropriate therapies, decrease risk of bronchiolitis‐related hospital readmission, and help with discharge planning.[30, 34, 35]
Notwithstanding the differences found in this study, management of children with bronchiolitis was, in many respects, comparable across racial/ethnic groups. For example, our multivariable analysis found no significant differences across racial/ethnic groups with respect to chest x‐rays and corticosteroid use during the preadmission visit, administration of albuterol or corticosteroids during hospitalization, use of CPAP/emntubation, ICU admission, hospital LOS, or likelihood of a bronchiolitis‐related relapse. The general lack of race/ethnic differences is consistent with similar research on inpatient management of acute asthma.[36]
This study has potential limitations. The hospitals participating in the study are predominantly urban, academically affiliated hospitals. This may result in findings that are less generalizable to rural and community hospitals. Second, the race/ethnicity classification used does not take into consideration the diversity and complexity of defining race/ethnicity in the United States. Third, bronchiolitis is defined as a clinical diagnosis that can encapsulate multiple lower respiratory infection diagnoses. As a result, there may have been variability in clinical and institutional practice. An additional limitation was utilizing RDSS to assess bronchiolitis severity. Although there is currently no validated, universally accepted score to assess bronchiolitis severity, several scores are available in the literature with varying performance. Last, the ZIP code‐based median household incomes used to assess SES are higher than federal data in similar geographic locations, potentially resulting in findings that are less generalizable.
CONCLUSION
This multicenter prospective cohort study found several differences in bronchiolitis presentation and management among children stratified by race/ethnicity in 16 geographically dispersed sites after controlling for multiple factors including SES. Our analysis showed that, when compared to NHW children, NHB children were more likely to be given albuterol during the preadmission visit and less likely to receive chest x‐rays as inpatients; Hispanic children were more likely to be discharged on inhaled corticosteroids. These differences are concerning for 2 reasons: (1) based on current evidence, race/ethnicity should not affect care in children with severe bronchiolitis; and (2) the observed differences in diagnostic testing and treatment are not recommended by the evidence‐based AAP guidelines. It is also important to note that these differences do not demonstrate that a specific race/ethnicity received better or worse clinical care. The goal of this analysis was not to determine the effectiveness of certain management tendencies in children with severe bronchiolitis, but rather to examine differences in the presentation and management of children from different racial/ethnic groups. The causes for the observed findings require further study. In the meantime, we suggest increasing the number of hospitals that incorporate clinical care pathways for severe bronchiolitis to control variation in practice and limit the impact that race/ethnicity may have in the provision of services.
Acknowledgements
The authors thank the MARC‐30 investigators for their ongoing dedication to bronchiolitis research.
Disclosures: This study was supported by the grant U01 AI‐67693 (Camargo) from the National Institutes of Health (Bethesda, MD). The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Allergy and Infectious Diseases or the National Institutes of Health. The authors have no financial relationships relevant to this article or conflicts of interest to disclose. Mr. Santiago conceptualized the analysis, interpreted the data, drafted the initial manuscript, and approved the final manuscript as submitted. Dr. Mansbach conceptualized and designed the initial study, coordinated data collection at 1 of the sites, critically reviewed the manuscript, and approved the final manuscript as submitted. Dr. Chou was responsible for analysis and interpretation of data, critically reviewed the manuscript, and approved the final manuscript as submitted. Dr. Delgado coordinated data collection at 1 of the sites, critically reviewed the manuscript, and approved the final manuscript as submitted. Dr. Piedra conceptualized and designed the initial study, coordinated virology testing, critically reviewed the manuscript, and approved the final manuscript as submitted. Ms. Sullivan coordinated data collection at all sites, critically reviewed the manuscript, and approved the final manuscript as submitted. Ms. Espinola was responsible for data management, analysis and interpretation of data, drafting of the initial manuscript, and approved the final manuscript as submitted. Dr. Camargo conceptualized and designed the initial study, assisted with data analysis and interpretation of data, critically reviewed the manuscript, and approved the final manuscript as submitted.
APPENDIX
| Principal Investigators at the 16 Participating Sites in MARC‐30 | |
| Besh Barcega, MD | Loma Linda University Children's Hospital, Loma Linda, CA |
| John Cheng, MD and Carlos Delgado, MD | Children's Healthcare of Atlanta at Egleston, Atlanta, GA |
| Dorothy Damore, MD and Nikhil Shah, MD | New York Presbyterian Hospital, New York, NY |
| Haitham Haddad, MD | Rainbow Babies & Children's Hospital, Cleveland, OH |
| Paul Hain, MD and Mark Riederer, MD | Monroe Carell Jr. Children's Hospital at Vanderbilt, Nashville, TN |
| Frank LoVecchio, DO | Maricopa Medical Center, Phoenix, AZ |
| Charles Macias, MD, MPH | Texas Children's Hospital, Houston, TX |
| Jonathan Mansbach, MD, MPH | Boston Children's Hospital, Boston, MA |
| Eugene Mowad, MD | Akron Children's Hospital, Akron, OH |
| Brian Pate, MD | Children's Mercy Hospital & Clinics, Kansas City, MO |
| M. Jason Sanders, MD | Children's Memorial Hermann Hospital, Houston, TX |
| Alan Schroeder, MD | Santa Clara Valley Medical Center, San Jose, CA |
| Michelle Stevenson, MD, MS | Kosair Children's Hospital, Louisville, KY |
| Erin Stucky Fisher, MD | Rady Children's Hospital, San Diego, CA |
| Stephen Teach, MD, MPH | Children's National Medical Center, Washington, DC |
| Lisa Zaoutis, MD | Children's Hospital of Philadelphia, Philadelphia, PA |
Bronchiolitis is the leading cause of hospitalization for infants in the United States, costs more than $500 million annually, and has seen a 30% increase ($1.34 billion to $1.73 billion) in related hospital charges from 2000 to 2009.13 Almost all children <2 years old are infected with respiratory syncytial virus, the most common cause of bronchiolitis, with 40% developing clinically recognizable bronchiolitis and 2% becoming hospitalized with severe bronchiolitis.[4, 5] Current American Academy of Pediatrics (AAP) guidelines state that routine use of bronchodilators, corticosteroids, and chest x‐rays is not recommended, and supportive care is strongly encouraged.[6] However, a lack of consensus among clinicians persists regarding bronchiolitis management.[7, 8, 9, 10] Although minority children and those with a lower socioeconomic status (SES) in the United States are more likely to present with bronchiolitis to the emergency department (ED) and be subsequently admitted when compared to the general population,[11, 12, 13] to our knowledge, no study has yet examined if race/ethnicity is independently associated with differences in the presentation and management of severe bronchiolitis (ie, bronchiolitis causing hospitalization). Although a prior bronchiolitis‐related study reported that Hispanic children had a longer ED length of stay (LOS) than non‐Hispanic white (NHW) and non‐Hispanic black (NHB) children,[14] other studies concluded that race/ethnicity were not predictors of intensive care unit (ICU) admission or unscheduled healthcare visits post‐ED discharge.[15, 16]
Determining if race/ethnicity is independently associated with certain bronchiolitis management tendencies has implications from both a health disparities standpoint (ie, unequal care based on race/ethnicity) and from a clinical perspective (ie, the potential of certain practices, such as clinical pathways, to increase the likelihood of equitable treatment). To address this knowledge gap, we examined prospective data from a multicenter study designed to evaluate multiple factors related to bronchiolitis hospitalization.
METHODS
Study Design
We conducted a multicenter prospective cohort for 3 consecutive years (20072010) as part of the Multicenter Airway Research Collaboration (MARC), a division of the Emergency Medicine Network (EMNet) (
All patients were treated at the discretion of their physician. Inclusion criteria were hospital admission with physician diagnosis of bronchiolitis, age <2 years, and ability of the child's guardian (eg, parent) to give informed consent. Patients were enrolled within 18 hours of admission. Physician diagnosis of bronchiolitis followed the AAP definition of a child with an acute respiratory illness with some combination of rhinitis, cough, tachypnea, wheezing, crackles, and/or retractions.[6] The exclusion criteria were previous enrollment and if a patient was transferred to a participating site hospital>48 hours after the initial admission. All consent and data forms were translated into Spanish. The institutional review board at each participating site approved the study.
Data Collection
Site investigators used a standardized protocol to enroll 2207 patients admitted with bronchiolitis. Investigators conducted a structured interview that assessed patients' demographic characteristics, medical and environmental history, duration of symptoms, and details of the acute illness. Race/ethnicity was assigned by report of the child's guardian to standard US Census groups. For the purpose of this analysis, mutually exclusive race/ethnicity categories were determined: NHW, NHB, or Hispanic. Non‐Hispanic patients who identified as being both white and black were categorized as NHB. Patients were excluded from analysis if neither white or black race nor Hispanic ethnicity were reported (eg, if only Asian race was reported) because of small numbers (n=67), as were patients missing all race/ethnicity data (n=10). This resulted in a total of 2130 (97%) patients in our analytical dataset. SES was assessed with 2 variables: insurance status (public, private, none) and family income, estimated by matching patients' home ZIP codes and year of enrollment to ZIP code‐based median household annual incomes obtained from Esri Business Analyst Desktop (Esri, Redlands, CA).[17]
ED and daily clinical data, including laboratory tests (eg, complete blood count, basic metabolic panel, urine analysis, blood culture), respiratory rates, oxygen saturation, medical management, and disposition were obtained by medical chart review. Additionally, in an attempt to evaluate bronchiolitis severity at presentation, a modified respiratory distress severity score (RDSS) was calculated based on 4 assessments made during the preadmission visit (ie, ED or office visit before hospital admission): respiratory rate by age, presence of wheezing (yes or no), air entry (normal, mild difficulty, or moderate/severe), and retractions (none, mild, or moderate/severe).[18] Each component was assigned a score of 0, 1, or 2, with the exception of wheeze, which was assigned either a 0 (no wheeze) or a 2 (wheeze), and then summed for a possible total score of 0 to 8.
Last, a follow‐up telephone interview was conducted 1 week after hospital discharge for each enrolled patient. Interviews assessed acute relapse, recent symptoms, and provided additional end points for longitudinal analysis of specific symptoms. All data were manually reviewed at the EMNet Coordinating Center, and site investigators were queried about missing data and discrepancies identified.
Outcome Measures
The major outcomes of this analysis were: albuterol and corticosteroid (inhaled or systemic) use during preadmission visit and hospitalization, chest x‐rays performed at preadmission visit and hospitalization, need for intensive respiratory support (ie, receiving continuous positive airway pressure [CPAP], intubation, or ICU admission), hospital LOS 3 days, discharge on inhaled corticosteroids, and relapse of bronchiolitis requiring medical attention and a change of medication within 1 week of discharge.
Statistical Analysis
Stata 11.2 (StataCorp, College Station, TX) was used for all analyses. We examined unadjusted differences between racial/ethnic groups and clinical presentation, patient management, and outcomes using 2, Fisher exact, or Kruskal‐Wallis test, as appropriate, with results reported as proportions with 95% confidence interval (CI) or median with interquartile range (IQR). Imputed values, calculated with the Stata impute command, were used to calculate the RDSS when 1 of the 4 components was missing; patients missing more than 1 component were not assigned an RDSS value. Multivariable logistic regression was conducted to evaluate the adjusted association between race/ethnicity and the outcomes listed above. Besides race/ethnicity, all multivariable models included the demographic variables of age, sex, insurance, and median household income. Other factors were considered for inclusion if they were associated with the outcome in unadjusted analyses (P<0.20) or deemed clinically relevant. All models were adjusted for the possibility of clustering by site. Results are reported for the race/ethnicity factor as odds ratios with 95% CI.
RESULTS
Of the 2130 subjects included in this analysis, 818 (38%) were NHW, 511 (24%) were NHB, and 801 (38%) were Hispanic. The median age for children was 4.0 months (IQR, 1.88.5 months), and 60% were male. Most children were publicly insured (65%), 31% had private insurance, and approximately 4% had no insurance. The median household income defined by patient ZIP code was $51,810 (IQR, $39,916$66,272), and nearly all children (97%) had a primary care provider (PCP). Approximately 21% of all children had relevant comorbidities and 17% of children were enrolled from the ICU. Overall, the median LOS was 2 days (IQR, 14 days).
The unadjusted associations between race/ethnicity and other demographic and historical characteristics are shown in Table 1. NHB and Hispanic children were more likely to have public insurance and less likely to have relevant major comorbidities when compared to NHW children. With regard to care received the week before hospitalization, NHW children were more likely to have visited their PCP, taken corticosteroids and/or antibiotics, and were least likely to have visited an ED when compared to NHB and Hispanic children.
| White, Non‐Hispanic, n=818, % | Black, Non‐Hispanic, n=511, % | Hispanic, n=801, % | P | |
|---|---|---|---|---|
| ||||
| Demographic characteristics | ||||
| Age, months, median (IQR) | 3.2 (1.57.4) | 5.0 (1.99.2) | 4.4 (2.09.1) | <0.001 |
| Female | 39.7 | 40.9 | 40.4 | 0.91 |
| Insurance | <0.001 | |||
| Private | 56.7 | 17.0 | 13.1 | |
| Medicaid | 32.8 | 70.9 | 77.5 | |
| Other public | 6.8 | 7.6 | 4.2 | |
| None | 3.7 | 4.6 | 5.3 | |
| Median household income by ZIP code, US$, median (IQR) | $60,406 ($48,086$75,077) | $44,191 ($32,922$55,640) | $50,394 ($39,242$62,148) | <0.001 |
| Has primary care provider | 98.5 | 97.3 | 95.4 | 0.001 |
| History | ||||
| Gestational age at birth | 0.002 | |||
| <32 weeks | 4.5 | 9.2 | 6.6 | |
| 3235 weeks | 5.7 | 8.8 | 6.0 | |
| 3537 weeks | 13.3 | 10.0 | 9.7 | |
| 37 weeks | 76.0 | 71.4 | 76.9 | |
| Missing | 0.4 | 0.6 | 0.7 | |
| Weight when born | <0.001 | |||
| <3 pounds | 3.3 | 6.9 | 5.8 | |
| 34.9 pounds | 6.8 | 11.9 | 6.2 | |
| 56.9 pounds | 33.4 | 39.7 | 33.2 | |
| >7 pounds | 55.8 | 40.3 | 53.6 | |
| Missing | 0.7 | 1.4 | 1.4 | |
| Kept in an ICU, premature nursery, or any type of special‐care facility when born | 24.5 | 29.7 | 24.9 | 0.07 |
| Breast fed | 62.3 | 49.1 | 65.5 | <0.001 |
| Attends daycare | 20.7 | 25.2 | 13.3 | <0.001 |
| Number of other children (<18 years old) living in home | <0.001 | |||
| 1 | 24.2 | 25.6 | 17.0 | |
| 2 | 42.7 | 29.2 | 28.3 | |
| 3 | 33.1 | 45.2 | 54.7 | |
| Neither parent has asthma | 66.1 | 56.6 | 77.7 | <0.001 |
| Maternal smoking during pregnancy | 21.8 | 21.3 | 6.0 | <0.001 |
| Secondhand smoke exposure | 12.9 | 20.2 | 8.7 | <0.001 |
| History of wheezing | 21.1 | 25.9 | 21.8 | 0.12 |
| Ever intubated | 9.5 | 13.2 | 9.2 | 0.05 |
| Major relevant comorbidities | 23.6 | 21.1 | 18.2 | 0.03 |
| Received palivizumab (respiratory syncytial virus vaccine) | 8.7 | 12.7 | 8.9 | 0.04 |
| Received influenza vaccine this year | 20.2 | 24.7 | 21.2 | 0.15 |
| In past 12 months, admitted overnight to hospital for bronchiolitis/wheezing/reactive airway disease | 45.0 | 57.9 | 55.9 | 0.06 |
| In past 12 months, admitted overnight to hospital for pneumonia | 16.1 | 14.9 | 25.0 | 0.049 |
| Current illness (before index visit) | ||||
| Any primary care provider or clinic visits during past week | 75.0 | 44.1 | 58.3 | <0.001 |
| Any ED visits during past week | 29.1 | 30.3 | 34.6 | 0.049 |
| Over the past week used inhaled bronchodilator | 40.6 | 36.2 | 37.0 | 0.18 |
| Over the past week used inhaled/nebulized corticosteroids | 8.7 | 8.1 | 7.7 | 0.76 |
| Over the past week taken any steroid liquids or pills or shots for bronchiolitis | 12.8 | 11.7 | 8.3 | 0.012 |
| Over the past week taken antibiotics | 21.9 | 17.0 | 17.9 | 0.045 |
| Onset of difficulty breathing before admission | 0.03 | |||
| None | 2.0 | 2.2 | 2.4 | |
| <24 hours | 28.8 | 27.2 | 25.1 | |
| 13 days | 41.1 | 41.6 | 45.9 | |
| 47 days | 22.1 | 19.1 | 21.2 | |
| >7 days | 6.0 | 9.9 | 5.3 | |
| Over the past 24 hours, the level of discomfort or distress felt by the child because of symptoms | <0.001 | |||
| Mild | 15.5 | 21.3 | 18.5 | |
| Moderate | 47.8 | 39.3 | 37.2 | |
| Severe | 36.1 | 37.6 | 42.6 | |
The unadjusted associations between race/ethnicity and clinical characteristics at preadmission visit and hospital admission are shown in Table 2. RDSS values were calculated for 2130 children; 1,752 (82%) RDSS values contained all 4 components. Of those requiring imputed values, 234 (11%) were missing 1 component, and 139 (7%) were missing more than 1 component. Per RDSS scores, NHB children presented with a more severe case of bronchiolitis when compared to NHW and Hispanic children. During admission, minority children were more likely to receive nebulized albuterol and less likely to visit the ICU. NHB children received the least inpatient laboratory testing and were least likely to receive chest x‐rays during hospital admission among all groups.
| White, Non‐Hispanic, n=818, % | Black, Non‐Hispanic, n=511, % | Hispanic, n=801, % | P | |
|---|---|---|---|---|
| ||||
| Preadmission clinical findings and treatments | ||||
| Reason brought to the hospital | ||||
| Fever | 29.7 | 30.2 | 40.7 | <0.001 |
| Fussy | 32.6 | 31.6 | 28.4 | 0.18 |
| Ear Infection | 6.0 | 4.3 | 4.4 | 0.23 |
| Not drinking well | 35.0 | 27.5 | 27.2 | 0.001 |
| Cough | 54.1 | 55.5 | 61.3 | 0.009 |
| Other reasons | 29.0 | 27.6 | 23.5 | 0.04 |
| Apnea | 8.5 | 6.2 | 6.1 | 0.11 |
| Respiratory rate (breaths/minute) | 0.001 | |||
| <40 | 23.8 | 18.2 | 28.1 | |
| 4049 | 30.8 | 30.7 | 29.3 | |
| 5059 | 17.6 | 15.5 | 16.0 | |
| 60 | 27.8 | 35.6 | 26.6 | |
| Presence of cough | 83.0 | 88.0 | 87.0 | 0.045 |
| Presence of wheezing | 63.0 | 69.0 | 63.0 | 0.42 |
| Fever (temperature 100.4F) | 22.7 | 28.5 | 35.4 | <0.001 |
| Retractions | 0.002 | |||
| None | 22.9 | 19.0 | 23.0 | |
| Mild | 39.4 | 41.7 | 44.4 | |
| Moderate/severe | 28.4 | 31.1 | 28.1 | |
| Missing | 9.4 | 8.2 | 4.5 | |
| Air entry on auscultation | 0.01 | |||
| Normal | 39.6 | 31.7 | 33.6 | |
| Mild difficulty | 31.4 | 33.5 | 36.3 | |
| Moderate difficulty | 11.0 | 14.3 | 14.1 | |
| Severe difficulty | 2.0 | 2.3 | 2.6 | |
| Missing | 16.0 | 18.2 | 13.4 | |
| Oxygen saturation on room air <90 | 12.2 | 9.8 | 11.8 | 0.37 |
| Given nebulized albuterol | 53.0 | 65.0 | 63.0 | <0.001 |
| Given nebulized epinephrine | 15.4 | 20.4 | 18.4 | 0.06 |
| Given steroids, inhaled or systemic | 16.0 | 20.5 | 19.4 | 0.08 |
| Given antibiotics | 25.5 | 22.6 | 27.8 | 0.12 |
| Oral Intake | <0.001 | |||
| Adequate | 41.3 | 50.7 | 40.8 | |
| Inadequate | 43.5 | 32.5 | 47.7 | |
| Missing | 15.2 | 16.8 | 11.5 | |
| IV placed | 56.9 | 51.5 | 61.9 | 0.001 |
| Any laboratory tests | 86.3 | 91.3 | 88.0 | 0.02 |
| Chest x‐ray | 59.0 | 64.0 | 65.0 | 0.03 |
| RDSS, tertiles | <0.001 | |||
| 1 (3.00) | 36.0 | 24.0 | 34.0 | |
| 2 (3.0115.00) | 30.0 | 33.0 | 34.0 | |
| 3 (>5) | 25.0 | 36.0 | 27.0 | |
| Not calculated | 9.0 | 6.0 | 4.0 | |
| Virology results | ||||
| Respiratory syncytial virus | 75.9 | 67.5 | 71.0 | 0.003 |
| Human rhinovirus | 23.8 | 30.1 | 25.0 | 0.03 |
| Human metapneumovirus | 6.1 | 6.8 | 8.4 | 0.20 |
| Inpatient clinical findings and treatments | ||||
| Length of stay 3 days | 46.5 | 39.3 | 45.4 | 0.03 |
| Ever in observation unit | 8.6 | 7.8 | 4.0 | 0.001 |
| Ever in regular ward | 89.4 | 93.0 | 94.5 | 0.001 |
| Ever in step‐down unit | 5.0 | 3.2 | 7.8 | 0.002 |
| Ever in ICU | 20.3 | 15.0 | 15.9 | 0.02 |
| Required CPAP or intubation | 7.7 | 4.6 | 8.8 | 0.02 |
| Given nebulized albuterol | 37.6 | 48.0 | 46.7 | <0.001 |
| Given nebulized epinephrine | 10.7 | 14.9 | 13.0 | 0.07 |
| Given steroids, inhaled or systemic | 21.3 | 27.3 | 23.5 | 0.047 |
| Given antibiotics | 38.9 | 34.3 | 38.6 | 0.19 |
| Received IV fluids | 53.1 | 45.6 | 57.1 | <0.001 |
| Any laboratory tests | 52.2 | 41.6 | 51.7 | <0.001 |
| Chest x‐ray | 27.1 | 18.6 | 22.9 | 0.002 |
Discharge treatment and outcomes at 1‐week follow‐up are shown in Table 3. A total of 1771 patients (83%) were reached by telephone. No statistically significant differences between racial/ethnic groups were found regarding hospital discharge on corticosteroids and likelihood of bronchiolitis‐related relapse.
| White, Non‐Hispanic, n=818, % | Black, Non‐Hispanic, n=511, % | Hispanic, n=801, % | P | |
|---|---|---|---|---|
| Discharged on inhaled corticosteroids | 9.5 | 11.1 | 13.3 | 0.08 |
| Discharged on oral corticosteroids | 9.8 | 12.4 | 8.5 | 0.11 |
| Child's condition at 1‐week follow‐up compared to on discharge | 0.001 | |||
| Much worse/worse | 1.8 | 0.7 | 0.4 | |
| About the same | 3.4 | 6.4 | 2.5 | |
| Better | 38.2 | 39.1 | 34.2 | |
| All better | 56.6 | 53.7 | 62.9 | |
| Child's cough at 1‐week follow‐up compared to on discharge | 0.10 | |||
| Much worse/worse | 2.1 | 1.2 | 1.0 | |
| About the same | 5.0 | 8.4 | 5.2 | |
| Better | 29.4 | 31.2 | 28.6 | |
| All better | 63.5 | 59.2 | 65.2 | |
| Bronchiolitis relapse | 10.7 | 11.9 | 10.3 | 0.81 |
Given the large potential for confounding regarding our initial findings, we examined multivariable‐adjusted associations of race/ethnicity and bronchiolitis management (Table 4). Receiving albuterol during the preadmission visit and chest x‐rays during hospitalization remained significantly associated with race/ethnicity in adjusted analyses, as NHB children were most likely to receive albuterol during the preadmission visit but least likely to receive chest x‐rays during hospitalization. Several outcomes with statistically significant differences found during unadjusted analyses (eg, chest x‐rays at preadmission visit, albuterol during hospitalization, CPAP/emntubation use, ICU admission, and LOS) were not independently associated with race/ethnicity in multivariable models. By contrast, adjusted analyses revealed Hispanic children as significantly more likely to be discharged on inhaled corticosteroids when compared to NHW and NHB children; this association had borderline statistical significance (P=0.08) in the unadjusted analysis. Last, we observed no significant racial/ethnic differences with respect to corticosteroids given at preadmission visit or hospitalization as well as no differences regarding bronchiolitis‐related relapse in either unadjusted or adjusted analyses.
| White, Non‐Hispanic | Black, Non‐Hispanic | Hispanic | ||||
|---|---|---|---|---|---|---|
| OR | 95%CI | OR | 95%CI | OR | 95%CI | |
| ||||||
| Preadmission visit | ||||||
| Chest x‐ray* | 1.00 | (Reference) | 1.06 | (0.831.36) | 1.09 | (0.741.60) |
| Albuterol use | 1.00 | (Reference) | 1.58 | (1.202.07) | 1.42 | (0.892.26) |
| Steroid use, inhaled or systemic | 1.00 | (Reference) | 1.05 | (0.721.54) | 1.11 | (0.751.65) |
| During hospitalization | ||||||
| Chest x‐ray‖ | 1.00 | (Reference) | 0.66 | (0.490.90) | 0.95 | (0.601.50) |
| Albuterol use | 1.00 | (Reference) | 1.21 | (0.821.79) | 1.23 | (0.632.38) |
| Steroid use, inhaled or systemic# | 1.00 | (Reference) | 1.13 | (0.721.80) | 1.19 | (0.791.79) |
| ICU care** | 1.00 | (Reference) | 0.74 | (0.421.29) | 0.87 | (0.631.21) |
| Required CPAP/emntubation | 1.00 | (Reference) | 0.72 | (0.361.41) | 1.84 | (0.933.64) |
| Length of stay >3 days | 1.00 | (Reference) | 0.77 | (0.581.03) | 1.05 | (0.761.47) |
| Discharge | ||||||
| Discharged on inhaled steroids | 1.00 | (Reference) | 1.31 | (0.862.00) | 1.92 | (1.193.10) |
| Bronchiolitis relapse‖‖ | 1.00 | (Reference) | 1.08 | (0.621.87) | 0.96 | (0.551.65) |
DISCUSSION
It is unclear if management and treatment differences found in children with severe bronchiolitis are associated with race/ethnicity. We sought to determine if such differences exist by analyzing data from a prospective multicenter cohort study. Differences in management and treatment are discussed in the context of AAP guidelines, as they are widely used in clinical practice.
The RDSS was used to help assess severity of illness across race/ethnicity. NHB children had the highest RDSS score (ie, most severe bronchiolitis presentation) compared to NHW and Hispanic children. The reason for this difference in severity is unclear, but a potential explanation may be that minority communities lack access to care and as a result delay care and treatment for respiratory disease until care seems absolutely necessary.[19] Indeed, in our sample, minority children were less likely to visit their PCP and take corticosteroids the week before hospitalization when compared with NHW children. Our finding runs counter to a similar study by Boudreaux et al. that found no association between race/ethnicity and the clinical presentation of children with acute asthma during the preadmission setting.[20] The more severe bronchiolitis presentation among NHB children may have suggested that these children would require a longer hospital LOS (3 days). However, our multivariable analysis found no difference in LOS across racial/ethnic groups. This LOS finding is intriguing given previous studies suggesting that minorities, of diverse ages and with diverse diagnoses, were more likely to have a shorter LOS (as well as less likely to be admitted to the ICU with a similar diagnosis) when compared to nonminorities.[21, 22] Additionally, because our study sampled 16 sites, variation in clinical judgment and pediatric ICU protocol may have also played a role.[23]
Our findings also shed light on how differences in bronchiolitis management relate to AAP guidelines. According to the AAP, corticosteroid medications should not be used routinely in the management of bronchiolitis. Despite this recommendation, previous reports indicate that up to 60% of infants with severe bronchiolitis receive corticosteroid therapy.[24, 25] Our finding that Hispanic children with severe bronchiolitis were most likely to be discharged on inhaled corticosteroids is potentially concerning, as it exposes a subset of children to treatment that is not recommended. On the other hand, given the increased risk of future asthma in Hispanic communities, a higher use of inhaled corticosteroids might be seen as appropriate. Either way, our findings are inconsistent with related studies concluding that racial minority pediatric patients with asthma were less likely to receive inhaled corticosteroids.[26, 27, 28, 29] Similarly, NHB children were most likely to receive albuterol during the preadmission visit on multivariable analysis. Although a trial dose of albuterol may be common practice in treating severe bronchiolitis, AAP recommendations do not support its routine application. Increased albuterol during preadmission may have been related to an elevated bronchiolitis severity at presentation among NHB children (as indicated by the RDSS). Potential reasons for these 2 differences in treatment remain unclear. They may represent medical management efforts by discharging physicians to prescribe: (1) corticosteroids to racial/ethnic communities with a higher risk of childhood asthma; (2) albuterol to children presenting with a more severe case of bronchiolitis. These possibilities merit further study.
The AAP also recommends diagnosis of bronchiolitis on the basis of history and physical examination; laboratory and radiologic studies should not be routinely used for diagnostic purposes. Although it is possible for chest radiograph abnormalities to be consistent with bronchiolitis, there is little evidence that an abnormal finding is associated with disease severity.[30] The clinical value of diagnostic testing in children with bronchiolitis is not well supported by evidence, and limiting exposure to radiation should be a priority.[31, 32] Our analysis found that NHW and Hispanic children were more likely to receive chest x‐rays while hospitalized when compared with NHB children. Unnecessary and increased radiation exposure in children is potentially harmful and warrants intervention to minimize risk.
Establishing systematic clinical pathways in bronchiolitis management may address the practice variation found nationwide and across race/ethnicity in this study. Although clinical guidelines provide general recommendations, clinical pathways are defined treatment protocols aiming to standardize and optimize patient outcomes and clinical efficiency. The incorporation of clinical pathways into healthcare systems has increased recently as a result of their favorable association with medical complications, healthcare costs, and LOS.[33] With respect to bronchiolitis, implementation of clinical pathways has proven to reduce use of inappropriate therapies, decrease risk of bronchiolitis‐related hospital readmission, and help with discharge planning.[30, 34, 35]
Notwithstanding the differences found in this study, management of children with bronchiolitis was, in many respects, comparable across racial/ethnic groups. For example, our multivariable analysis found no significant differences across racial/ethnic groups with respect to chest x‐rays and corticosteroid use during the preadmission visit, administration of albuterol or corticosteroids during hospitalization, use of CPAP/emntubation, ICU admission, hospital LOS, or likelihood of a bronchiolitis‐related relapse. The general lack of race/ethnic differences is consistent with similar research on inpatient management of acute asthma.[36]
This study has potential limitations. The hospitals participating in the study are predominantly urban, academically affiliated hospitals. This may result in findings that are less generalizable to rural and community hospitals. Second, the race/ethnicity classification used does not take into consideration the diversity and complexity of defining race/ethnicity in the United States. Third, bronchiolitis is defined as a clinical diagnosis that can encapsulate multiple lower respiratory infection diagnoses. As a result, there may have been variability in clinical and institutional practice. An additional limitation was utilizing RDSS to assess bronchiolitis severity. Although there is currently no validated, universally accepted score to assess bronchiolitis severity, several scores are available in the literature with varying performance. Last, the ZIP code‐based median household incomes used to assess SES are higher than federal data in similar geographic locations, potentially resulting in findings that are less generalizable.
CONCLUSION
This multicenter prospective cohort study found several differences in bronchiolitis presentation and management among children stratified by race/ethnicity in 16 geographically dispersed sites after controlling for multiple factors including SES. Our analysis showed that, when compared to NHW children, NHB children were more likely to be given albuterol during the preadmission visit and less likely to receive chest x‐rays as inpatients; Hispanic children were more likely to be discharged on inhaled corticosteroids. These differences are concerning for 2 reasons: (1) based on current evidence, race/ethnicity should not affect care in children with severe bronchiolitis; and (2) the observed differences in diagnostic testing and treatment are not recommended by the evidence‐based AAP guidelines. It is also important to note that these differences do not demonstrate that a specific race/ethnicity received better or worse clinical care. The goal of this analysis was not to determine the effectiveness of certain management tendencies in children with severe bronchiolitis, but rather to examine differences in the presentation and management of children from different racial/ethnic groups. The causes for the observed findings require further study. In the meantime, we suggest increasing the number of hospitals that incorporate clinical care pathways for severe bronchiolitis to control variation in practice and limit the impact that race/ethnicity may have in the provision of services.
Acknowledgements
The authors thank the MARC‐30 investigators for their ongoing dedication to bronchiolitis research.
Disclosures: This study was supported by the grant U01 AI‐67693 (Camargo) from the National Institutes of Health (Bethesda, MD). The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Allergy and Infectious Diseases or the National Institutes of Health. The authors have no financial relationships relevant to this article or conflicts of interest to disclose. Mr. Santiago conceptualized the analysis, interpreted the data, drafted the initial manuscript, and approved the final manuscript as submitted. Dr. Mansbach conceptualized and designed the initial study, coordinated data collection at 1 of the sites, critically reviewed the manuscript, and approved the final manuscript as submitted. Dr. Chou was responsible for analysis and interpretation of data, critically reviewed the manuscript, and approved the final manuscript as submitted. Dr. Delgado coordinated data collection at 1 of the sites, critically reviewed the manuscript, and approved the final manuscript as submitted. Dr. Piedra conceptualized and designed the initial study, coordinated virology testing, critically reviewed the manuscript, and approved the final manuscript as submitted. Ms. Sullivan coordinated data collection at all sites, critically reviewed the manuscript, and approved the final manuscript as submitted. Ms. Espinola was responsible for data management, analysis and interpretation of data, drafting of the initial manuscript, and approved the final manuscript as submitted. Dr. Camargo conceptualized and designed the initial study, assisted with data analysis and interpretation of data, critically reviewed the manuscript, and approved the final manuscript as submitted.
APPENDIX
| Principal Investigators at the 16 Participating Sites in MARC‐30 | |
| Besh Barcega, MD | Loma Linda University Children's Hospital, Loma Linda, CA |
| John Cheng, MD and Carlos Delgado, MD | Children's Healthcare of Atlanta at Egleston, Atlanta, GA |
| Dorothy Damore, MD and Nikhil Shah, MD | New York Presbyterian Hospital, New York, NY |
| Haitham Haddad, MD | Rainbow Babies & Children's Hospital, Cleveland, OH |
| Paul Hain, MD and Mark Riederer, MD | Monroe Carell Jr. Children's Hospital at Vanderbilt, Nashville, TN |
| Frank LoVecchio, DO | Maricopa Medical Center, Phoenix, AZ |
| Charles Macias, MD, MPH | Texas Children's Hospital, Houston, TX |
| Jonathan Mansbach, MD, MPH | Boston Children's Hospital, Boston, MA |
| Eugene Mowad, MD | Akron Children's Hospital, Akron, OH |
| Brian Pate, MD | Children's Mercy Hospital & Clinics, Kansas City, MO |
| M. Jason Sanders, MD | Children's Memorial Hermann Hospital, Houston, TX |
| Alan Schroeder, MD | Santa Clara Valley Medical Center, San Jose, CA |
| Michelle Stevenson, MD, MS | Kosair Children's Hospital, Louisville, KY |
| Erin Stucky Fisher, MD | Rady Children's Hospital, San Diego, CA |
| Stephen Teach, MD, MPH | Children's National Medical Center, Washington, DC |
| Lisa Zaoutis, MD | Children's Hospital of Philadelphia, Philadelphia, PA |
- , , . Direct medical costs of bronchiolitis‐related hospitalizations in the United States. Pediatrics. 2006;118(6):2418–2423.
- , , , , . Trends in bronchiolitis hospitalizations in the United States, 2000–2009. Pediatrics. 2013;132(1):28–36.
- , , , , . Infectious disease hospitalizations among infants in the United States. Pediatrics. 2008;121(2):244–252.
- , . Respiratory syncytial virus. Curr Probl Pediatr. 1993;23(2):50–79.
- , , , , . Rates of hospitalization for respiratory syncytial virus infection among children in Medicaid. J Pediatr. 2000;137(6):865–870.
- American Academy of Pediatrics Subcommittee on Diagnosis and Management of Bronchiolitis. Diagnosis and management of bronchiolitis. Pediatrics. 2006;118(4):1774–1793.
- , , . US outpatient office visits for bronchiolitis, 1993–2004. Ambul Pediatr. 2007;7(4):304–307.
- , , , et al. Study of interobserver reliability in clinical assessment of RSV lower respiratory illness: a pediatric investigators collaborative network for infections in Canada (PICNIC) study. Pediatr Pulmonol. 1996;22(1):23–27.
- , , , , . Effect of practice variation on resource utilization in infants hospitalized for viral lower respiratory illness. Pediatrics. 2001;108(4):851–855.
- , , . Bronchiolitis in US emergency departments 1992 to 2000: epidemiology and practice variation. Pediatr Emerg Care. 2005;21(4):242–247.
- , . Recent trends in severe respiratory syncytial virus (RSV) among US infants, 1997 to 2000. J Pediatr. 2003; 143(5 suppl):S127–S132.
- , , , , . Risk of respiratory syncytial virus infection for infants from low‐income families in relationship to age, sex, ethnic group, and maternal antibody level. J Pediatr. 1981;98(5):708–715.
- , , . Socioeconomic environmental factors and hospitalization for acute bronchiolitis during infancy. Acta Paediatr. 2002;91(3):335–338.
- , , , , . Factors associated with longer emergency department length of stay for children with bronchiolitis: a prospective multicenter study. Pediatr Emerg Care. 2009;25(10):636–641.
- , , , , . Prospective multicenter bronchiolitis study: predicting intensive care unit admissions. Acad Emerg Med. 2008;15(10):887–894.
- , , , , . Prospective multicenter study of bronchiolitis: predictors of an unscheduled visit after discharge from the emergency department. Acad Emerg Med. 2010;17(4):376–382.
- Esri. Demographic, consumer, and business data. Available at: http://www.esri.com/data/esri_data/demographic‐overview/demographic. Accessed July 25, 2013.
- , , . A randomized trial of home oxygen therapy from the emergency department for acute bronchiolitis. Pediatrics. 2006;117(3):633–640.
- , , , , , . Adherence to therapy and access to care: the relationship to excess asthma morbidity in African‐American children. Pediatr Asthma Aller. 1994;8(3):179–184.
- , , , . Race/ethnicity and asthma among children presenting to the emergency department: differences in disease severity and management. Pediatrics. 2003;111(5 pt 1):e615–e621.
- , , , . Relationship between patient race and the intensity of hospital services. Med Care. 1987;25(7):592–603.
- , , , , . African‐American and white patients admitted to the intensive care unit: is there a difference in therapy and outcome? Crit Care Med. 1995;23(4):626–636.
- , , . Acute severe asthma: differences in therapies and outcomes among pediatric intensive care units. Crit Care Med. 2002;30(3):581–585.
- , , , . International variation in the management of infants hospitalized with respiratory syncytial virus. International RSV Study Group. Eur J Pediatr. 1998;157(3):215–220.
- , , , , . Effect of practice variation on resource utilization in infants for viral lower respiratory illness. Pediatrics. 2001;108:851–855.
- , , , , , . Impact of site of care, race, and Hispanic ethnicity on medication use for childhood asthma. Pediatrics. 2002;109(1):E1.
- , , , et al. Racial/ethnic variation in asthma status and management practices among children in managed Medicaid. Pediatrics. 2002;109(5):857–865.
- , , , . Treatment adherence among low‐income children with asthma. J Pediatr Psychol. 1998;23(6):345–349.
- American Academy of Pediatrics Steering Committee on Quality Improvement and Management. Classifying recommendations for clinical practice guidelines. Pediatrics. 2004;114(3):874–877.
- , , . Randomised controlled trial of clinical outcome after chest radiograph in ambulatory acute lower‐respiratory infection in children. Lancet. 1998;351(9100):404–408.
- . Cancer risks following diagnostic and therapeutic radiation exposure in children. Pediatr Radiol. 2006;36(suppl 2):121–125.
- , , , et al. Clinical pathways: effects on professional practice, patient outcomes, length of stay and hospital costs. Cochrane Database Syst Rev. 2010;(3):CD006632.
- , , . An evidence‐based clinical pathway for bronchiolitis safely reduces antibiotic overuse. Am J Med Qual. 2002;17(5):195–199.
- , , , et al. A clinical pathway for bronchiolitis is effective in reducing readmission rates. J Pediatr. 2005;147(5):622–626.
- , , , et al. Decreasing unnecessary utilization in acute bronchiolitis care: results from the value in inpatient pediatrics network. J Hosp Med. 2013;8(1):25–30.
- , , . Race/Ethnicity differences in the inpatient management of acute asthma in the United States. Chest. 2009;135(6):1527–1534.
- , , . Direct medical costs of bronchiolitis‐related hospitalizations in the United States. Pediatrics. 2006;118(6):2418–2423.
- , , , , . Trends in bronchiolitis hospitalizations in the United States, 2000–2009. Pediatrics. 2013;132(1):28–36.
- , , , , . Infectious disease hospitalizations among infants in the United States. Pediatrics. 2008;121(2):244–252.
- , . Respiratory syncytial virus. Curr Probl Pediatr. 1993;23(2):50–79.
- , , , , . Rates of hospitalization for respiratory syncytial virus infection among children in Medicaid. J Pediatr. 2000;137(6):865–870.
- American Academy of Pediatrics Subcommittee on Diagnosis and Management of Bronchiolitis. Diagnosis and management of bronchiolitis. Pediatrics. 2006;118(4):1774–1793.
- , , . US outpatient office visits for bronchiolitis, 1993–2004. Ambul Pediatr. 2007;7(4):304–307.
- , , , et al. Study of interobserver reliability in clinical assessment of RSV lower respiratory illness: a pediatric investigators collaborative network for infections in Canada (PICNIC) study. Pediatr Pulmonol. 1996;22(1):23–27.
- , , , , . Effect of practice variation on resource utilization in infants hospitalized for viral lower respiratory illness. Pediatrics. 2001;108(4):851–855.
- , , . Bronchiolitis in US emergency departments 1992 to 2000: epidemiology and practice variation. Pediatr Emerg Care. 2005;21(4):242–247.
- , . Recent trends in severe respiratory syncytial virus (RSV) among US infants, 1997 to 2000. J Pediatr. 2003; 143(5 suppl):S127–S132.
- , , , , . Risk of respiratory syncytial virus infection for infants from low‐income families in relationship to age, sex, ethnic group, and maternal antibody level. J Pediatr. 1981;98(5):708–715.
- , , . Socioeconomic environmental factors and hospitalization for acute bronchiolitis during infancy. Acta Paediatr. 2002;91(3):335–338.
- , , , , . Factors associated with longer emergency department length of stay for children with bronchiolitis: a prospective multicenter study. Pediatr Emerg Care. 2009;25(10):636–641.
- , , , , . Prospective multicenter bronchiolitis study: predicting intensive care unit admissions. Acad Emerg Med. 2008;15(10):887–894.
- , , , , . Prospective multicenter study of bronchiolitis: predictors of an unscheduled visit after discharge from the emergency department. Acad Emerg Med. 2010;17(4):376–382.
- Esri. Demographic, consumer, and business data. Available at: http://www.esri.com/data/esri_data/demographic‐overview/demographic. Accessed July 25, 2013.
- , , . A randomized trial of home oxygen therapy from the emergency department for acute bronchiolitis. Pediatrics. 2006;117(3):633–640.
- , , , , , . Adherence to therapy and access to care: the relationship to excess asthma morbidity in African‐American children. Pediatr Asthma Aller. 1994;8(3):179–184.
- , , , . Race/ethnicity and asthma among children presenting to the emergency department: differences in disease severity and management. Pediatrics. 2003;111(5 pt 1):e615–e621.
- , , , . Relationship between patient race and the intensity of hospital services. Med Care. 1987;25(7):592–603.
- , , , , . African‐American and white patients admitted to the intensive care unit: is there a difference in therapy and outcome? Crit Care Med. 1995;23(4):626–636.
- , , . Acute severe asthma: differences in therapies and outcomes among pediatric intensive care units. Crit Care Med. 2002;30(3):581–585.
- , , , . International variation in the management of infants hospitalized with respiratory syncytial virus. International RSV Study Group. Eur J Pediatr. 1998;157(3):215–220.
- , , , , . Effect of practice variation on resource utilization in infants for viral lower respiratory illness. Pediatrics. 2001;108:851–855.
- , , , , , . Impact of site of care, race, and Hispanic ethnicity on medication use for childhood asthma. Pediatrics. 2002;109(1):E1.
- , , , et al. Racial/ethnic variation in asthma status and management practices among children in managed Medicaid. Pediatrics. 2002;109(5):857–865.
- , , , . Treatment adherence among low‐income children with asthma. J Pediatr Psychol. 1998;23(6):345–349.
- American Academy of Pediatrics Steering Committee on Quality Improvement and Management. Classifying recommendations for clinical practice guidelines. Pediatrics. 2004;114(3):874–877.
- , , . Randomised controlled trial of clinical outcome after chest radiograph in ambulatory acute lower‐respiratory infection in children. Lancet. 1998;351(9100):404–408.
- . Cancer risks following diagnostic and therapeutic radiation exposure in children. Pediatr Radiol. 2006;36(suppl 2):121–125.
- , , , et al. Clinical pathways: effects on professional practice, patient outcomes, length of stay and hospital costs. Cochrane Database Syst Rev. 2010;(3):CD006632.
- , , . An evidence‐based clinical pathway for bronchiolitis safely reduces antibiotic overuse. Am J Med Qual. 2002;17(5):195–199.
- , , , et al. A clinical pathway for bronchiolitis is effective in reducing readmission rates. J Pediatr. 2005;147(5):622–626.
- , , , et al. Decreasing unnecessary utilization in acute bronchiolitis care: results from the value in inpatient pediatrics network. J Hosp Med. 2013;8(1):25–30.
- , , . Race/Ethnicity differences in the inpatient management of acute asthma in the United States. Chest. 2009;135(6):1527–1534.
© 2014 Society of Hospital Medicine
Aging and Inpatient Demand
The number of older people in the United States is expected to increase, due to the aging of the post‐World War II baby boomers.[1] For example, those aged 65 years are expected to number 88.5 million in 2050, more than double the number in 2010 of 40.2 million. This demographic shift has raised concerns about future hospital capacity, but the scope of the problem has not been quantified.[2]
A recent analysis calculated the number and length of emergency department visits expected to occur based on the aging of the US population.[3] One finding was that hospital admissions would increase 23% faster than population growth. However, this considered only hospitalizations originating in the emergency department and did not consider all‐source hospitalizations. We obtained data on all‐source hospitalizations and applied them to the US Census Bureau's demographic projections for the future through 2050. This provides a base‐case estimate for how inpatient demand would change if all other influences remained equal. The goal was to isolate the effect of population age makeup on inpatient requirements while holding other influences constant.
METHODS
We used the method of actuarial life table adjustment as described previously.[3] To calculate age‐specific hospitalization rates, we estimated age‐specific hospitalization frequencies (counts) in the United States for 2011 from the Nationwide Inpatient Sample (NIS).[4] This is a stratified probability sample of US community hospitals, defined as all nonfederal, short term, general, and other specialty hospitals, excluding hospital units of institutions. Veterans hospitals and other federal facilities, short‐term rehabilitation hospitals, long‐term non‐acute care hospitals, psychiatric hospitals, and alcoholism/chemical dependency treatment facilities were excluded from NIS 2011. Of hospitals in the sample, 21% are government (nonfederal) owned.
We converted age‐specific hospitalization frequencies derived from this sample into rates by dividing each stratum‐specific admission count by the 2011 population count in each age stratum from the US Census Bureau.[5] The Census Bureau provides detailed predictions of the US population through 2050. Births, deaths, and net international migration are projected for each birth cohort. Using 2011 as the origin, we applied baseline age‐specific hospitalization rates stratum‐wise to the general population expected by the Census Bureau in future years. This gave us stratum‐specific hospitalization frequencies for each future year. We summed these to arrive at the aggregate anticipated hospitalization frequency in each year. For our main outcome measure, we calculated the ratio of change in hospitalization frequency to change in population, comparing each future year to the 2011 baseline. We also calculated aggregate inpatient days, using the same data sources and methods. Our institutional review board exempted this study from review. We used Stata 13.0 (StataCorp, College Station, TX), and Microsoft Excel (Microsoft, Redmond, WA) for all analyses.
RESULTS
Baseline data are displayed in Figure 1. In 2011, there were 0.23 hospitalizations per US resident aged 0 to 4 years, and 0.01 per resident aged 5 to 9 years. From this age forward, hospitalization rates increased steadily with advancing age, reaching 0.63 per resident aged 90 to 94 years. Length of stay also was generally associated with age, though there was a peak among older children.
Projections through 2050 are shown in Table 1 and Figure 2. Table 1 displays the population projections of the US Census Bureau, which expects the US population to increase by 41% between now and 2050. Also shown in the table are our projections, which indicate that, all other things being equal, the annual number of inpatient admissions in the US will increase by 67%. The ratio of 67% to 41% is 1.18, meaning that the frequency of inpatient admissions will grow 18% more than population growth due to the aging of the population. The aggregate number of inpatient days will increase 22% more than population growth. Overall, inpatient capacity must expand by 72% to keep pace.
| Year | Population | Hospital Admissions | Aggregate Inpatient Days | Population: Ratio of Future Year to 2011 | Admissions: Ratio of Future Year to 2011 | Ratio of Admission Increase to Population Increase | Aggregate Inpatient Days: Ratio of Future Year to 2011 | Ratio of Increase in Inpatient Days to Population Increase |
|---|---|---|---|---|---|---|---|---|
| ||||||||
| 2011 | 311,591,917 | 38,560,751* | 177,501,515 | 1 | 1 | 1 | 1 | 1 |
| 2015 | 325,539,790 | 41,093,154 | 189,520,706 | 1.04 | 1.07 | 1.02 | 1.07 | 1.02 |
| 2020 | 341,386,665 | 44,196,669 | 205,205,962 | 1.10 | 1.15 | 1.05 | 1.16 | 1.06 |
| 2025 | 357,451,620 | 47,655,492 | 222,911,204 | 1.15 | 1.24 | 1.08 | 1.26 | 1.09 |
| 2030 | 373,503,674 | 51,365,441 | 241,852,384 | 1.20 | 1.33 | 1.11 | 1.36 | 1.14 |
| 2035 | 389,531,156 | 55,091,242 | 260,603,998 | 1.25 | 1.43 | 1.14 | 1.47 | 1.17 |
| 2040 | 405,655,295 | 58,524,016 | 277,530,732 | 1.30 | 1.52 | 1.17 | 1.56 | 1.20 |
| 2045 | 422,058,629 | 61,525,903 | 292,014,192 | 1.35 | 1.60 | 1.18 | 1.65 | 1.21 |
| 2050 | 439,010,253 | 64,249,181 | 304,945,179 | 1.41 | 1.67 | 1.18 | 1.72 | 1.22 |
DISCUSSION
Although US hospital capacity has fallen over the past 3 decades,[6, 7] our analysis suggests that demand for inpatient beds will increase 22% faster than population growth by 2050. The total projected demand increase is 72%, including that attributable to population growth and that attributable to population aging.
These are ceteris paribus projections, which reveal the changes in inpatient demand that would result if 2 conditions held: (1) the US Census Bureau's expectations for population makeup proved correct, and (2) age‐specific hospitalization rates and lengths of stay did not change. In reality, age‐specific hospitalization rates and lengths of stay could change. Examples of change drivers include epidemics, technology, and financial incentives provided by third‐party payers.[7] For example, if an epidemic of a new disease were to occur, age‐specific hospitalization rates could increase across all age groups. Our projections depict what would happen in the absence of any such change. This is useful because we do not know if changes in age‐specific hospitalization rates will occur, and whether there will be increases or decreases. Therefore, our projections should not be viewed as literal predictions, but rather as pieces of the puzzle, necessary but not sufficient elements of an understanding of what the future may hold for inpatient demand.
Clinicians, academics, and government agencies have an interest in understanding inpatient supply and demand on national and local levels. However, their ability to influence supply is limited by the fact that of all registered hospitals in the United States, only 22% are government owned.[1] As a result, decisions about hospital construction and closure are generally left to the free market.[6] Nonetheless, we bear responsibility for monitoring supply and demand, and government regulation of hospitals and reimbursement for inpatient care mean that the public is not entirely without influence. Thirty‐two percent of US residents have government‐issued health insurance.[8]
In the early 20th century, very little healthcare took place in the inpatient setting. However, by the 1970s, inpatient care accounted for a large part of healthcare, due largely to changes in technology and reimbursement. This trend reversed in the 1980s and 1990s, and hospitals closed.[7] In 1975, there were 5875 hospitals in the United States, and in 2000 there were 4915.[6] The number of staffed beds decreased from 942,000 to 826,000.[6] In parallel, likely due to changes in technology (ie, the nature of healthcare), total inpatient days in community hospitals decreased from 223 million in 1991 to 187 million in 2011.[9] On the other hand, increasing access to insurance under the Affordable Care Act could increase utilization, as seen when a 30% increase in hospital utilization occurred when people were enrolled in Oregon's Medicaid program.[10] Also, hospital utilization may increase if Medicare patients require more services.[11]
Actuarial life table analysis has been used to make forecasts related to healthcare supply and demand, though we are not aware of prior applications to the question of hospitalization. A prior study used actuarial life table adjustment to forecast demand for emergency department services.[3] These methods have also been used to forecast the influence of longevity upon healthcare expenditures[12, 13, 14] and to predict demand for specialty services.[15, 16] Of note, rather than reporting ratios of demand growth to population growth, another option would have been to derive a compound growth rate. We are not aware of a precedent for such methods in the prior published applications of actuarial life table analysis and felt that such inductive methods would complicate the interpretation of our results.
The main limitation of our investigation is its scope. We used actuarial life table adjustment to isolate the effect of population aging upon demand for inpatient hospitalizations. This method does not yield a comprehensive prediction of inpatient demand, but rather provides a robust estimate under the assumption that all other things remain equal. Another obvious limitation is that our analysis has a nationwide scope, and was not designed to account for variation from one locale to the next. However, these methods can be used by local health authorities.
CONCLUSIONS
The US Census Bureau expects the US population to increase by 41% over the next 4 decades, and the number of US residents aged 65 years to more than double. Our results indicate that, all other things being equal, this will cause the number of hospital admissions to increase 18% faster than population growth, and the aggregate number of inpatient days to increase 22% faster than population growth. Including both population growth and population aging, the total projected increase required for inpatient capacity is 72%. This is a base‐case, ceteris paribus analysis, and understanding how demand for inpatient services may change will require multiple perspectives. Increasing access to insurance, changing poverty rates, and changes in healthcare delivery and technology are other important factors. The present analysis provides a focused estimate of the influence upon demand for inpatient services due to expected changes in our population's age distribution.
- American Hospital Association. Fast facts on US hospitals, 2011. Available at: http://www.aha.org/research/rc/stat‐studies/fast‐facts.shtml. Accessed August 7, 2013.
- American Hospital Association. Cracks in the foundation: averting a crisis in America's hospitals. AHA 2002. Available at: http://www.aha.org/content/00–10/cracksreprint08‐02.pdf. Accessed August 4, 2013.
- , , , , . Population aging and emergency departments: visits will not increase, lengths‐of‐stay and hospitalizations will. Health Aff (Millwood). 2013;32(7):1306–1312.
- HCUP Nationwide Inpatient Sample (NIS). Healthcare Cost and Utilization Project (HCUP). 2011. Agency for Healthcare Research and Quality, Rockville, MD. Available at: http://www.hcup‐us.ahrq.gov/nisoverview.jsp. Accessed July 25, 2013.
- Bureau of the Census. Population Projections by Age, Sex, Race, and Hispanic Origin: July 1, 2000–2050. Washington, DC: The Bureau; 2008.
- , , , . Does U.S. hospital capacity need to be expanded? Health Aff (Millwood). 2003;22(6):40–54.
- . Decline in hospital utilization and cost inflation under managed care in California. JAMA. 1996;276(13):1060–1064.
- , , . Income, poverty, and health insurance coverage in the United States, 2011. US Census Bureau. Available at: http://www.census.gov/prod/2012pubs/p60–243.pdf. Published September 2012. Accessed August 7, 2013.
- American Hospital Association. Trendwatch. Table 3.1: trends in inpatient utilization in community hospitals, 1991–2011. Available at: http://www.aha.org/research/reports/tw/chartbook/2013/table3‐1.pdf. Accessed November 9, 2013.
- , , , et al. The Oregon health insurance experiment: evidence from the first year. Q J Econ. 2012;127(3):1057–1106.
- American Hospital Association. Trendwatch. Are Medicare patients getting sicker? Available at: http://www.aha.org/research/reports/tw/12dec‐tw‐ptacuity.pdf. Accessed November 9, 2013.
- , , . Longevity and Medicare expenditures. N Engl J Med. 1995;332(15):999–1003.
- , . The aging of America. Impact on health care costs. JAMA. 1990;263(17):2335–2340.
- , . The effect of longevity on spending for acute and long‐term care. N Engl J Med. 2000;342(19):1409–1415.
- , , , . Demographics and cardiology, 1950–2050. J Am Coll Cardiol. 2000;35(4):1067–1081.
- , , , . Population‐based analysis of inpatient vascular procedures and predicting future workload and implications for training. J Vasc Surg. 2012;55(5):1394–1399; discussion 1399–1400.
The number of older people in the United States is expected to increase, due to the aging of the post‐World War II baby boomers.[1] For example, those aged 65 years are expected to number 88.5 million in 2050, more than double the number in 2010 of 40.2 million. This demographic shift has raised concerns about future hospital capacity, but the scope of the problem has not been quantified.[2]
A recent analysis calculated the number and length of emergency department visits expected to occur based on the aging of the US population.[3] One finding was that hospital admissions would increase 23% faster than population growth. However, this considered only hospitalizations originating in the emergency department and did not consider all‐source hospitalizations. We obtained data on all‐source hospitalizations and applied them to the US Census Bureau's demographic projections for the future through 2050. This provides a base‐case estimate for how inpatient demand would change if all other influences remained equal. The goal was to isolate the effect of population age makeup on inpatient requirements while holding other influences constant.
METHODS
We used the method of actuarial life table adjustment as described previously.[3] To calculate age‐specific hospitalization rates, we estimated age‐specific hospitalization frequencies (counts) in the United States for 2011 from the Nationwide Inpatient Sample (NIS).[4] This is a stratified probability sample of US community hospitals, defined as all nonfederal, short term, general, and other specialty hospitals, excluding hospital units of institutions. Veterans hospitals and other federal facilities, short‐term rehabilitation hospitals, long‐term non‐acute care hospitals, psychiatric hospitals, and alcoholism/chemical dependency treatment facilities were excluded from NIS 2011. Of hospitals in the sample, 21% are government (nonfederal) owned.
We converted age‐specific hospitalization frequencies derived from this sample into rates by dividing each stratum‐specific admission count by the 2011 population count in each age stratum from the US Census Bureau.[5] The Census Bureau provides detailed predictions of the US population through 2050. Births, deaths, and net international migration are projected for each birth cohort. Using 2011 as the origin, we applied baseline age‐specific hospitalization rates stratum‐wise to the general population expected by the Census Bureau in future years. This gave us stratum‐specific hospitalization frequencies for each future year. We summed these to arrive at the aggregate anticipated hospitalization frequency in each year. For our main outcome measure, we calculated the ratio of change in hospitalization frequency to change in population, comparing each future year to the 2011 baseline. We also calculated aggregate inpatient days, using the same data sources and methods. Our institutional review board exempted this study from review. We used Stata 13.0 (StataCorp, College Station, TX), and Microsoft Excel (Microsoft, Redmond, WA) for all analyses.
RESULTS
Baseline data are displayed in Figure 1. In 2011, there were 0.23 hospitalizations per US resident aged 0 to 4 years, and 0.01 per resident aged 5 to 9 years. From this age forward, hospitalization rates increased steadily with advancing age, reaching 0.63 per resident aged 90 to 94 years. Length of stay also was generally associated with age, though there was a peak among older children.

Projections through 2050 are shown in Table 1 and Figure 2. Table 1 displays the population projections of the US Census Bureau, which expects the US population to increase by 41% between now and 2050. Also shown in the table are our projections, which indicate that, all other things being equal, the annual number of inpatient admissions in the US will increase by 67%. The ratio of 67% to 41% is 1.18, meaning that the frequency of inpatient admissions will grow 18% more than population growth due to the aging of the population. The aggregate number of inpatient days will increase 22% more than population growth. Overall, inpatient capacity must expand by 72% to keep pace.

| Year | Population | Hospital Admissions | Aggregate Inpatient Days | Population: Ratio of Future Year to 2011 | Admissions: Ratio of Future Year to 2011 | Ratio of Admission Increase to Population Increase | Aggregate Inpatient Days: Ratio of Future Year to 2011 | Ratio of Increase in Inpatient Days to Population Increase |
|---|---|---|---|---|---|---|---|---|
| ||||||||
| 2011 | 311,591,917 | 38,560,751* | 177,501,515 | 1 | 1 | 1 | 1 | 1 |
| 2015 | 325,539,790 | 41,093,154 | 189,520,706 | 1.04 | 1.07 | 1.02 | 1.07 | 1.02 |
| 2020 | 341,386,665 | 44,196,669 | 205,205,962 | 1.10 | 1.15 | 1.05 | 1.16 | 1.06 |
| 2025 | 357,451,620 | 47,655,492 | 222,911,204 | 1.15 | 1.24 | 1.08 | 1.26 | 1.09 |
| 2030 | 373,503,674 | 51,365,441 | 241,852,384 | 1.20 | 1.33 | 1.11 | 1.36 | 1.14 |
| 2035 | 389,531,156 | 55,091,242 | 260,603,998 | 1.25 | 1.43 | 1.14 | 1.47 | 1.17 |
| 2040 | 405,655,295 | 58,524,016 | 277,530,732 | 1.30 | 1.52 | 1.17 | 1.56 | 1.20 |
| 2045 | 422,058,629 | 61,525,903 | 292,014,192 | 1.35 | 1.60 | 1.18 | 1.65 | 1.21 |
| 2050 | 439,010,253 | 64,249,181 | 304,945,179 | 1.41 | 1.67 | 1.18 | 1.72 | 1.22 |
DISCUSSION
Although US hospital capacity has fallen over the past 3 decades,[6, 7] our analysis suggests that demand for inpatient beds will increase 22% faster than population growth by 2050. The total projected demand increase is 72%, including that attributable to population growth and that attributable to population aging.
These are ceteris paribus projections, which reveal the changes in inpatient demand that would result if 2 conditions held: (1) the US Census Bureau's expectations for population makeup proved correct, and (2) age‐specific hospitalization rates and lengths of stay did not change. In reality, age‐specific hospitalization rates and lengths of stay could change. Examples of change drivers include epidemics, technology, and financial incentives provided by third‐party payers.[7] For example, if an epidemic of a new disease were to occur, age‐specific hospitalization rates could increase across all age groups. Our projections depict what would happen in the absence of any such change. This is useful because we do not know if changes in age‐specific hospitalization rates will occur, and whether there will be increases or decreases. Therefore, our projections should not be viewed as literal predictions, but rather as pieces of the puzzle, necessary but not sufficient elements of an understanding of what the future may hold for inpatient demand.
Clinicians, academics, and government agencies have an interest in understanding inpatient supply and demand on national and local levels. However, their ability to influence supply is limited by the fact that of all registered hospitals in the United States, only 22% are government owned.[1] As a result, decisions about hospital construction and closure are generally left to the free market.[6] Nonetheless, we bear responsibility for monitoring supply and demand, and government regulation of hospitals and reimbursement for inpatient care mean that the public is not entirely without influence. Thirty‐two percent of US residents have government‐issued health insurance.[8]
In the early 20th century, very little healthcare took place in the inpatient setting. However, by the 1970s, inpatient care accounted for a large part of healthcare, due largely to changes in technology and reimbursement. This trend reversed in the 1980s and 1990s, and hospitals closed.[7] In 1975, there were 5875 hospitals in the United States, and in 2000 there were 4915.[6] The number of staffed beds decreased from 942,000 to 826,000.[6] In parallel, likely due to changes in technology (ie, the nature of healthcare), total inpatient days in community hospitals decreased from 223 million in 1991 to 187 million in 2011.[9] On the other hand, increasing access to insurance under the Affordable Care Act could increase utilization, as seen when a 30% increase in hospital utilization occurred when people were enrolled in Oregon's Medicaid program.[10] Also, hospital utilization may increase if Medicare patients require more services.[11]
Actuarial life table analysis has been used to make forecasts related to healthcare supply and demand, though we are not aware of prior applications to the question of hospitalization. A prior study used actuarial life table adjustment to forecast demand for emergency department services.[3] These methods have also been used to forecast the influence of longevity upon healthcare expenditures[12, 13, 14] and to predict demand for specialty services.[15, 16] Of note, rather than reporting ratios of demand growth to population growth, another option would have been to derive a compound growth rate. We are not aware of a precedent for such methods in the prior published applications of actuarial life table analysis and felt that such inductive methods would complicate the interpretation of our results.
The main limitation of our investigation is its scope. We used actuarial life table adjustment to isolate the effect of population aging upon demand for inpatient hospitalizations. This method does not yield a comprehensive prediction of inpatient demand, but rather provides a robust estimate under the assumption that all other things remain equal. Another obvious limitation is that our analysis has a nationwide scope, and was not designed to account for variation from one locale to the next. However, these methods can be used by local health authorities.
CONCLUSIONS
The US Census Bureau expects the US population to increase by 41% over the next 4 decades, and the number of US residents aged 65 years to more than double. Our results indicate that, all other things being equal, this will cause the number of hospital admissions to increase 18% faster than population growth, and the aggregate number of inpatient days to increase 22% faster than population growth. Including both population growth and population aging, the total projected increase required for inpatient capacity is 72%. This is a base‐case, ceteris paribus analysis, and understanding how demand for inpatient services may change will require multiple perspectives. Increasing access to insurance, changing poverty rates, and changes in healthcare delivery and technology are other important factors. The present analysis provides a focused estimate of the influence upon demand for inpatient services due to expected changes in our population's age distribution.
The number of older people in the United States is expected to increase, due to the aging of the post‐World War II baby boomers.[1] For example, those aged 65 years are expected to number 88.5 million in 2050, more than double the number in 2010 of 40.2 million. This demographic shift has raised concerns about future hospital capacity, but the scope of the problem has not been quantified.[2]
A recent analysis calculated the number and length of emergency department visits expected to occur based on the aging of the US population.[3] One finding was that hospital admissions would increase 23% faster than population growth. However, this considered only hospitalizations originating in the emergency department and did not consider all‐source hospitalizations. We obtained data on all‐source hospitalizations and applied them to the US Census Bureau's demographic projections for the future through 2050. This provides a base‐case estimate for how inpatient demand would change if all other influences remained equal. The goal was to isolate the effect of population age makeup on inpatient requirements while holding other influences constant.
METHODS
We used the method of actuarial life table adjustment as described previously.[3] To calculate age‐specific hospitalization rates, we estimated age‐specific hospitalization frequencies (counts) in the United States for 2011 from the Nationwide Inpatient Sample (NIS).[4] This is a stratified probability sample of US community hospitals, defined as all nonfederal, short term, general, and other specialty hospitals, excluding hospital units of institutions. Veterans hospitals and other federal facilities, short‐term rehabilitation hospitals, long‐term non‐acute care hospitals, psychiatric hospitals, and alcoholism/chemical dependency treatment facilities were excluded from NIS 2011. Of hospitals in the sample, 21% are government (nonfederal) owned.
We converted age‐specific hospitalization frequencies derived from this sample into rates by dividing each stratum‐specific admission count by the 2011 population count in each age stratum from the US Census Bureau.[5] The Census Bureau provides detailed predictions of the US population through 2050. Births, deaths, and net international migration are projected for each birth cohort. Using 2011 as the origin, we applied baseline age‐specific hospitalization rates stratum‐wise to the general population expected by the Census Bureau in future years. This gave us stratum‐specific hospitalization frequencies for each future year. We summed these to arrive at the aggregate anticipated hospitalization frequency in each year. For our main outcome measure, we calculated the ratio of change in hospitalization frequency to change in population, comparing each future year to the 2011 baseline. We also calculated aggregate inpatient days, using the same data sources and methods. Our institutional review board exempted this study from review. We used Stata 13.0 (StataCorp, College Station, TX), and Microsoft Excel (Microsoft, Redmond, WA) for all analyses.
RESULTS
Baseline data are displayed in Figure 1. In 2011, there were 0.23 hospitalizations per US resident aged 0 to 4 years, and 0.01 per resident aged 5 to 9 years. From this age forward, hospitalization rates increased steadily with advancing age, reaching 0.63 per resident aged 90 to 94 years. Length of stay also was generally associated with age, though there was a peak among older children.

Projections through 2050 are shown in Table 1 and Figure 2. Table 1 displays the population projections of the US Census Bureau, which expects the US population to increase by 41% between now and 2050. Also shown in the table are our projections, which indicate that, all other things being equal, the annual number of inpatient admissions in the US will increase by 67%. The ratio of 67% to 41% is 1.18, meaning that the frequency of inpatient admissions will grow 18% more than population growth due to the aging of the population. The aggregate number of inpatient days will increase 22% more than population growth. Overall, inpatient capacity must expand by 72% to keep pace.

| Year | Population | Hospital Admissions | Aggregate Inpatient Days | Population: Ratio of Future Year to 2011 | Admissions: Ratio of Future Year to 2011 | Ratio of Admission Increase to Population Increase | Aggregate Inpatient Days: Ratio of Future Year to 2011 | Ratio of Increase in Inpatient Days to Population Increase |
|---|---|---|---|---|---|---|---|---|
| ||||||||
| 2011 | 311,591,917 | 38,560,751* | 177,501,515 | 1 | 1 | 1 | 1 | 1 |
| 2015 | 325,539,790 | 41,093,154 | 189,520,706 | 1.04 | 1.07 | 1.02 | 1.07 | 1.02 |
| 2020 | 341,386,665 | 44,196,669 | 205,205,962 | 1.10 | 1.15 | 1.05 | 1.16 | 1.06 |
| 2025 | 357,451,620 | 47,655,492 | 222,911,204 | 1.15 | 1.24 | 1.08 | 1.26 | 1.09 |
| 2030 | 373,503,674 | 51,365,441 | 241,852,384 | 1.20 | 1.33 | 1.11 | 1.36 | 1.14 |
| 2035 | 389,531,156 | 55,091,242 | 260,603,998 | 1.25 | 1.43 | 1.14 | 1.47 | 1.17 |
| 2040 | 405,655,295 | 58,524,016 | 277,530,732 | 1.30 | 1.52 | 1.17 | 1.56 | 1.20 |
| 2045 | 422,058,629 | 61,525,903 | 292,014,192 | 1.35 | 1.60 | 1.18 | 1.65 | 1.21 |
| 2050 | 439,010,253 | 64,249,181 | 304,945,179 | 1.41 | 1.67 | 1.18 | 1.72 | 1.22 |
DISCUSSION
Although US hospital capacity has fallen over the past 3 decades,[6, 7] our analysis suggests that demand for inpatient beds will increase 22% faster than population growth by 2050. The total projected demand increase is 72%, including that attributable to population growth and that attributable to population aging.
These are ceteris paribus projections, which reveal the changes in inpatient demand that would result if 2 conditions held: (1) the US Census Bureau's expectations for population makeup proved correct, and (2) age‐specific hospitalization rates and lengths of stay did not change. In reality, age‐specific hospitalization rates and lengths of stay could change. Examples of change drivers include epidemics, technology, and financial incentives provided by third‐party payers.[7] For example, if an epidemic of a new disease were to occur, age‐specific hospitalization rates could increase across all age groups. Our projections depict what would happen in the absence of any such change. This is useful because we do not know if changes in age‐specific hospitalization rates will occur, and whether there will be increases or decreases. Therefore, our projections should not be viewed as literal predictions, but rather as pieces of the puzzle, necessary but not sufficient elements of an understanding of what the future may hold for inpatient demand.
Clinicians, academics, and government agencies have an interest in understanding inpatient supply and demand on national and local levels. However, their ability to influence supply is limited by the fact that of all registered hospitals in the United States, only 22% are government owned.[1] As a result, decisions about hospital construction and closure are generally left to the free market.[6] Nonetheless, we bear responsibility for monitoring supply and demand, and government regulation of hospitals and reimbursement for inpatient care mean that the public is not entirely without influence. Thirty‐two percent of US residents have government‐issued health insurance.[8]
In the early 20th century, very little healthcare took place in the inpatient setting. However, by the 1970s, inpatient care accounted for a large part of healthcare, due largely to changes in technology and reimbursement. This trend reversed in the 1980s and 1990s, and hospitals closed.[7] In 1975, there were 5875 hospitals in the United States, and in 2000 there were 4915.[6] The number of staffed beds decreased from 942,000 to 826,000.[6] In parallel, likely due to changes in technology (ie, the nature of healthcare), total inpatient days in community hospitals decreased from 223 million in 1991 to 187 million in 2011.[9] On the other hand, increasing access to insurance under the Affordable Care Act could increase utilization, as seen when a 30% increase in hospital utilization occurred when people were enrolled in Oregon's Medicaid program.[10] Also, hospital utilization may increase if Medicare patients require more services.[11]
Actuarial life table analysis has been used to make forecasts related to healthcare supply and demand, though we are not aware of prior applications to the question of hospitalization. A prior study used actuarial life table adjustment to forecast demand for emergency department services.[3] These methods have also been used to forecast the influence of longevity upon healthcare expenditures[12, 13, 14] and to predict demand for specialty services.[15, 16] Of note, rather than reporting ratios of demand growth to population growth, another option would have been to derive a compound growth rate. We are not aware of a precedent for such methods in the prior published applications of actuarial life table analysis and felt that such inductive methods would complicate the interpretation of our results.
The main limitation of our investigation is its scope. We used actuarial life table adjustment to isolate the effect of population aging upon demand for inpatient hospitalizations. This method does not yield a comprehensive prediction of inpatient demand, but rather provides a robust estimate under the assumption that all other things remain equal. Another obvious limitation is that our analysis has a nationwide scope, and was not designed to account for variation from one locale to the next. However, these methods can be used by local health authorities.
CONCLUSIONS
The US Census Bureau expects the US population to increase by 41% over the next 4 decades, and the number of US residents aged 65 years to more than double. Our results indicate that, all other things being equal, this will cause the number of hospital admissions to increase 18% faster than population growth, and the aggregate number of inpatient days to increase 22% faster than population growth. Including both population growth and population aging, the total projected increase required for inpatient capacity is 72%. This is a base‐case, ceteris paribus analysis, and understanding how demand for inpatient services may change will require multiple perspectives. Increasing access to insurance, changing poverty rates, and changes in healthcare delivery and technology are other important factors. The present analysis provides a focused estimate of the influence upon demand for inpatient services due to expected changes in our population's age distribution.
- American Hospital Association. Fast facts on US hospitals, 2011. Available at: http://www.aha.org/research/rc/stat‐studies/fast‐facts.shtml. Accessed August 7, 2013.
- American Hospital Association. Cracks in the foundation: averting a crisis in America's hospitals. AHA 2002. Available at: http://www.aha.org/content/00–10/cracksreprint08‐02.pdf. Accessed August 4, 2013.
- , , , , . Population aging and emergency departments: visits will not increase, lengths‐of‐stay and hospitalizations will. Health Aff (Millwood). 2013;32(7):1306–1312.
- HCUP Nationwide Inpatient Sample (NIS). Healthcare Cost and Utilization Project (HCUP). 2011. Agency for Healthcare Research and Quality, Rockville, MD. Available at: http://www.hcup‐us.ahrq.gov/nisoverview.jsp. Accessed July 25, 2013.
- Bureau of the Census. Population Projections by Age, Sex, Race, and Hispanic Origin: July 1, 2000–2050. Washington, DC: The Bureau; 2008.
- , , , . Does U.S. hospital capacity need to be expanded? Health Aff (Millwood). 2003;22(6):40–54.
- . Decline in hospital utilization and cost inflation under managed care in California. JAMA. 1996;276(13):1060–1064.
- , , . Income, poverty, and health insurance coverage in the United States, 2011. US Census Bureau. Available at: http://www.census.gov/prod/2012pubs/p60–243.pdf. Published September 2012. Accessed August 7, 2013.
- American Hospital Association. Trendwatch. Table 3.1: trends in inpatient utilization in community hospitals, 1991–2011. Available at: http://www.aha.org/research/reports/tw/chartbook/2013/table3‐1.pdf. Accessed November 9, 2013.
- , , , et al. The Oregon health insurance experiment: evidence from the first year. Q J Econ. 2012;127(3):1057–1106.
- American Hospital Association. Trendwatch. Are Medicare patients getting sicker? Available at: http://www.aha.org/research/reports/tw/12dec‐tw‐ptacuity.pdf. Accessed November 9, 2013.
- , , . Longevity and Medicare expenditures. N Engl J Med. 1995;332(15):999–1003.
- , . The aging of America. Impact on health care costs. JAMA. 1990;263(17):2335–2340.
- , . The effect of longevity on spending for acute and long‐term care. N Engl J Med. 2000;342(19):1409–1415.
- , , , . Demographics and cardiology, 1950–2050. J Am Coll Cardiol. 2000;35(4):1067–1081.
- , , , . Population‐based analysis of inpatient vascular procedures and predicting future workload and implications for training. J Vasc Surg. 2012;55(5):1394–1399; discussion 1399–1400.
- American Hospital Association. Fast facts on US hospitals, 2011. Available at: http://www.aha.org/research/rc/stat‐studies/fast‐facts.shtml. Accessed August 7, 2013.
- American Hospital Association. Cracks in the foundation: averting a crisis in America's hospitals. AHA 2002. Available at: http://www.aha.org/content/00–10/cracksreprint08‐02.pdf. Accessed August 4, 2013.
- , , , , . Population aging and emergency departments: visits will not increase, lengths‐of‐stay and hospitalizations will. Health Aff (Millwood). 2013;32(7):1306–1312.
- HCUP Nationwide Inpatient Sample (NIS). Healthcare Cost and Utilization Project (HCUP). 2011. Agency for Healthcare Research and Quality, Rockville, MD. Available at: http://www.hcup‐us.ahrq.gov/nisoverview.jsp. Accessed July 25, 2013.
- Bureau of the Census. Population Projections by Age, Sex, Race, and Hispanic Origin: July 1, 2000–2050. Washington, DC: The Bureau; 2008.
- , , , . Does U.S. hospital capacity need to be expanded? Health Aff (Millwood). 2003;22(6):40–54.
- . Decline in hospital utilization and cost inflation under managed care in California. JAMA. 1996;276(13):1060–1064.
- , , . Income, poverty, and health insurance coverage in the United States, 2011. US Census Bureau. Available at: http://www.census.gov/prod/2012pubs/p60–243.pdf. Published September 2012. Accessed August 7, 2013.
- American Hospital Association. Trendwatch. Table 3.1: trends in inpatient utilization in community hospitals, 1991–2011. Available at: http://www.aha.org/research/reports/tw/chartbook/2013/table3‐1.pdf. Accessed November 9, 2013.
- , , , et al. The Oregon health insurance experiment: evidence from the first year. Q J Econ. 2012;127(3):1057–1106.
- American Hospital Association. Trendwatch. Are Medicare patients getting sicker? Available at: http://www.aha.org/research/reports/tw/12dec‐tw‐ptacuity.pdf. Accessed November 9, 2013.
- , , . Longevity and Medicare expenditures. N Engl J Med. 1995;332(15):999–1003.
- , . The aging of America. Impact on health care costs. JAMA. 1990;263(17):2335–2340.
- , . The effect of longevity on spending for acute and long‐term care. N Engl J Med. 2000;342(19):1409–1415.
- , , , . Demographics and cardiology, 1950–2050. J Am Coll Cardiol. 2000;35(4):1067–1081.
- , , , . Population‐based analysis of inpatient vascular procedures and predicting future workload and implications for training. J Vasc Surg. 2012;55(5):1394–1399; discussion 1399–1400.