User login
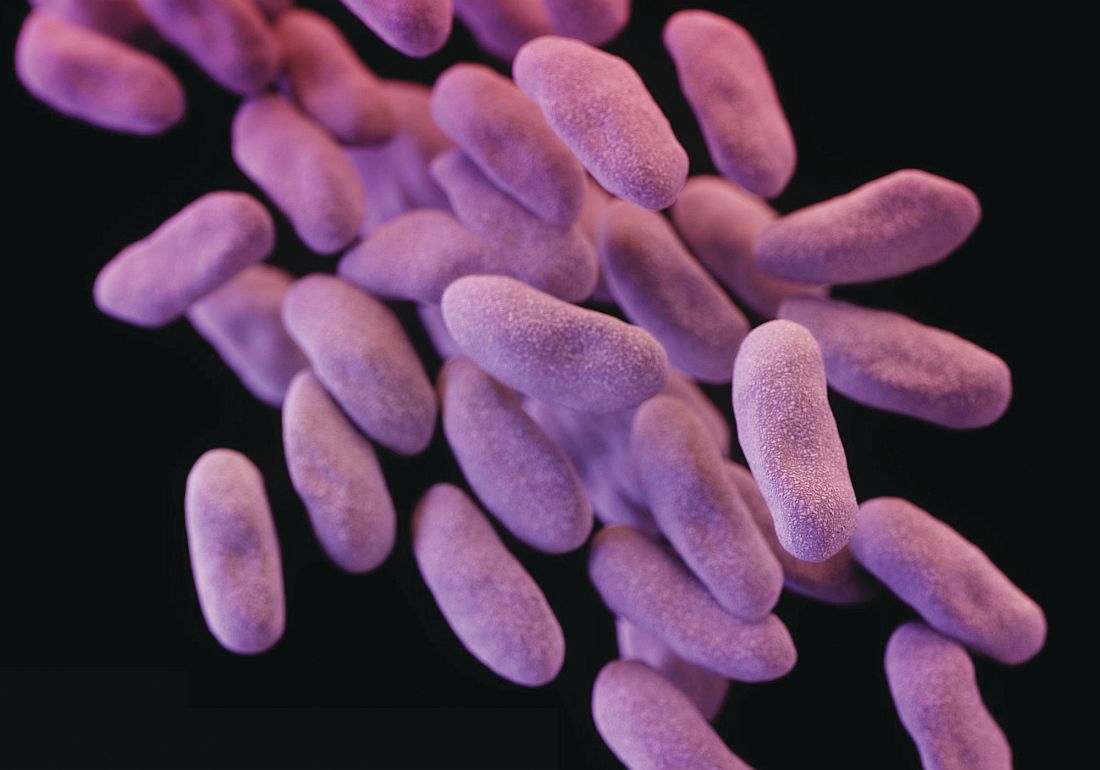
Sodium fusidate noninferior to linezolid for acute skin infections
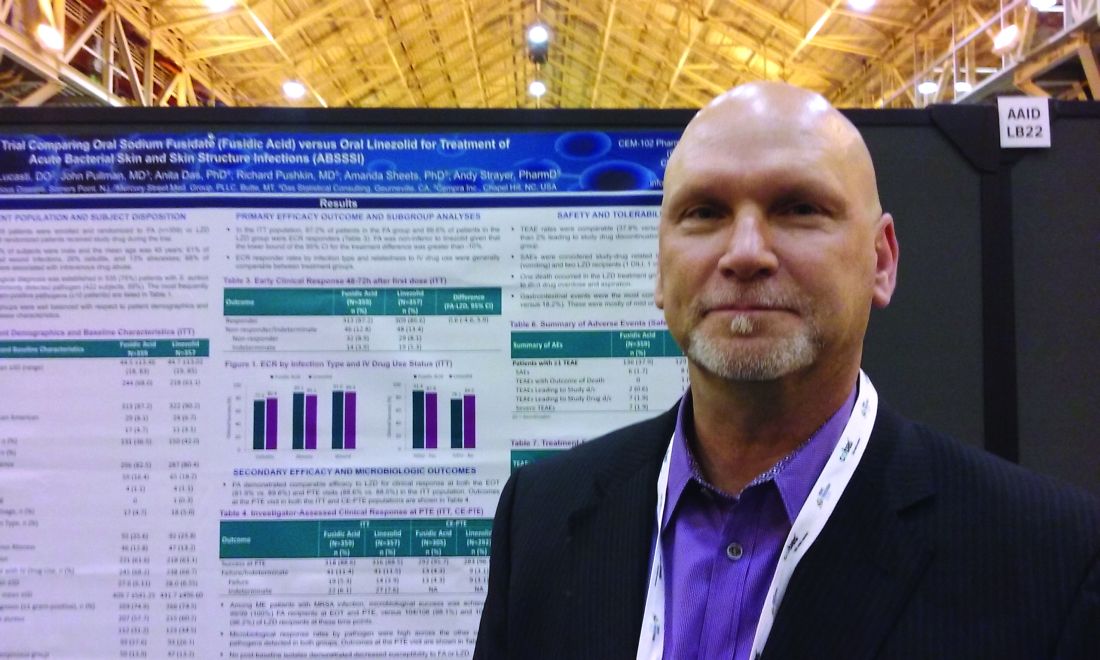
NEW ORLEANS – An oral antibiotic in development in the United States, fusidic acid (oral formulation, sodium fusidate) was noninferior to linezolid based on early clinical response in a randomized, double-blind, multicenter trial of 716 people with acute bacterial skin and skin structure infections (ABSSSI), including cellulitis, wound infection, and major cutaneous abscesses.
Early clinical response was defined as a 20% or greater reduction from baseline in the surface area of redness, edema, or induration at 48-72 hours after starting treatment with the study drugs. In an intent-to-treat analysis, 87.2% of patients randomized to fusidic acid and 86.6% of the linezolid group met this primary endpoint of the phase 3 study.
“Fusidic acid showed similar efficacy and comparable safety” that persisted through treatment, said Andy Strayer, PharmD, vice president of clinical programs at Cempra Pharmaceuticals, which is developing sodium fusidate as an oral agent to treat ABSSSI patients in the United States. Leo Pharmaceuticals has marketed sodium fusidate outside the United States in various formulations for decades.
Fusidic acid has potent activity against gram-positive aerobic organisms, including methicillin-resistant Staphylococcus aureus (MRSA). “Strikingly, fusidic acid showed 100% success in patients with MRSA in the microbiologically evaluable population at the end of treatment and posttherapy evaluation time points,” Dr. Strayer said at the annual meeting of the American Society for Microbiology. “Fusidic acid may offer an important oral therapy alternative for MRSA infection.”
“Fusidic acid, a drug long used in other parts of the world, has been demonstrated in this first phase 3 trial, to be a potential new option for the treatment of MRSA skin and skin structure infections in the U.S.,” said Carrie Cardenas, MD, lead study author and a principal investigator at eStudySite, San Diego, and an internist in private practice in La Mesa, California.
There was a microbiological diagnosis established in 75% of patients. S. aureus was the most commonly detected pathogen (422 patients; 59%), and the study included 235 patients diagnosed with MRSA infection.
About two-thirds, 65%, of participants were men. Mean age was 45 years. Infections were classified as wounds in 61%, cellulitis in 26%, and abscess in 13%. Notably, 68% of the recruited participants had ABSSSI associated with intravenous drug use, a “sometimes overlooked consequence of the ongoing epidemic of IV drug use in the U.S.,” Dr. Strayer said.
In terms of safety, treatment-emergent adverse event rates were comparable between the two groups (37.9% with fusidic acid versus 36.1% with linezolid). Gastrointestinal events were the most common adverse events, 22.8% versus 18.2%, respectively.
“Considering complicated skin infections are one of the most rapidly growing reasons for hospitalizations and emergency department visits each year, we anticipate that fusidic acid, if approved, may help clinicians decrease the length of inpatient stay or avoid hospitalization altogether,” Dr. Strayer said.
Cempra sponsored the study. Dr. Strayer is a Cempra employee and shareholder. Dr. Carrie Cardenas is a principal investigator at eStudySite, San Diego, and performs research for Cempra, Paratek, Debiopharm, Motif, Durata, MicuRx, Bristol-Myers Squibb, and Bayer.
NEW ORLEANS – An oral antibiotic in development in the United States, fusidic acid (oral formulation, sodium fusidate) was noninferior to linezolid based on early clinical response in a randomized, double-blind, multicenter trial of 716 people with acute bacterial skin and skin structure infections (ABSSSI), including cellulitis, wound infection, and major cutaneous abscesses.
Early clinical response was defined as a 20% or greater reduction from baseline in the surface area of redness, edema, or induration at 48-72 hours after starting treatment with the study drugs. In an intent-to-treat analysis, 87.2% of patients randomized to fusidic acid and 86.6% of the linezolid group met this primary endpoint of the phase 3 study.
“Fusidic acid showed similar efficacy and comparable safety” that persisted through treatment, said Andy Strayer, PharmD, vice president of clinical programs at Cempra Pharmaceuticals, which is developing sodium fusidate as an oral agent to treat ABSSSI patients in the United States. Leo Pharmaceuticals has marketed sodium fusidate outside the United States in various formulations for decades.
Fusidic acid has potent activity against gram-positive aerobic organisms, including methicillin-resistant Staphylococcus aureus (MRSA). “Strikingly, fusidic acid showed 100% success in patients with MRSA in the microbiologically evaluable population at the end of treatment and posttherapy evaluation time points,” Dr. Strayer said at the annual meeting of the American Society for Microbiology. “Fusidic acid may offer an important oral therapy alternative for MRSA infection.”
“Fusidic acid, a drug long used in other parts of the world, has been demonstrated in this first phase 3 trial, to be a potential new option for the treatment of MRSA skin and skin structure infections in the U.S.,” said Carrie Cardenas, MD, lead study author and a principal investigator at eStudySite, San Diego, and an internist in private practice in La Mesa, California.
There was a microbiological diagnosis established in 75% of patients. S. aureus was the most commonly detected pathogen (422 patients; 59%), and the study included 235 patients diagnosed with MRSA infection.
About two-thirds, 65%, of participants were men. Mean age was 45 years. Infections were classified as wounds in 61%, cellulitis in 26%, and abscess in 13%. Notably, 68% of the recruited participants had ABSSSI associated with intravenous drug use, a “sometimes overlooked consequence of the ongoing epidemic of IV drug use in the U.S.,” Dr. Strayer said.
In terms of safety, treatment-emergent adverse event rates were comparable between the two groups (37.9% with fusidic acid versus 36.1% with linezolid). Gastrointestinal events were the most common adverse events, 22.8% versus 18.2%, respectively.
“Considering complicated skin infections are one of the most rapidly growing reasons for hospitalizations and emergency department visits each year, we anticipate that fusidic acid, if approved, may help clinicians decrease the length of inpatient stay or avoid hospitalization altogether,” Dr. Strayer said.
Cempra sponsored the study. Dr. Strayer is a Cempra employee and shareholder. Dr. Carrie Cardenas is a principal investigator at eStudySite, San Diego, and performs research for Cempra, Paratek, Debiopharm, Motif, Durata, MicuRx, Bristol-Myers Squibb, and Bayer.
NEW ORLEANS – An oral antibiotic in development in the United States, fusidic acid (oral formulation, sodium fusidate) was noninferior to linezolid based on early clinical response in a randomized, double-blind, multicenter trial of 716 people with acute bacterial skin and skin structure infections (ABSSSI), including cellulitis, wound infection, and major cutaneous abscesses.
Early clinical response was defined as a 20% or greater reduction from baseline in the surface area of redness, edema, or induration at 48-72 hours after starting treatment with the study drugs. In an intent-to-treat analysis, 87.2% of patients randomized to fusidic acid and 86.6% of the linezolid group met this primary endpoint of the phase 3 study.
“Fusidic acid showed similar efficacy and comparable safety” that persisted through treatment, said Andy Strayer, PharmD, vice president of clinical programs at Cempra Pharmaceuticals, which is developing sodium fusidate as an oral agent to treat ABSSSI patients in the United States. Leo Pharmaceuticals has marketed sodium fusidate outside the United States in various formulations for decades.
Fusidic acid has potent activity against gram-positive aerobic organisms, including methicillin-resistant Staphylococcus aureus (MRSA). “Strikingly, fusidic acid showed 100% success in patients with MRSA in the microbiologically evaluable population at the end of treatment and posttherapy evaluation time points,” Dr. Strayer said at the annual meeting of the American Society for Microbiology. “Fusidic acid may offer an important oral therapy alternative for MRSA infection.”
“Fusidic acid, a drug long used in other parts of the world, has been demonstrated in this first phase 3 trial, to be a potential new option for the treatment of MRSA skin and skin structure infections in the U.S.,” said Carrie Cardenas, MD, lead study author and a principal investigator at eStudySite, San Diego, and an internist in private practice in La Mesa, California.
There was a microbiological diagnosis established in 75% of patients. S. aureus was the most commonly detected pathogen (422 patients; 59%), and the study included 235 patients diagnosed with MRSA infection.
About two-thirds, 65%, of participants were men. Mean age was 45 years. Infections were classified as wounds in 61%, cellulitis in 26%, and abscess in 13%. Notably, 68% of the recruited participants had ABSSSI associated with intravenous drug use, a “sometimes overlooked consequence of the ongoing epidemic of IV drug use in the U.S.,” Dr. Strayer said.
In terms of safety, treatment-emergent adverse event rates were comparable between the two groups (37.9% with fusidic acid versus 36.1% with linezolid). Gastrointestinal events were the most common adverse events, 22.8% versus 18.2%, respectively.
“Considering complicated skin infections are one of the most rapidly growing reasons for hospitalizations and emergency department visits each year, we anticipate that fusidic acid, if approved, may help clinicians decrease the length of inpatient stay or avoid hospitalization altogether,” Dr. Strayer said.
Cempra sponsored the study. Dr. Strayer is a Cempra employee and shareholder. Dr. Carrie Cardenas is a principal investigator at eStudySite, San Diego, and performs research for Cempra, Paratek, Debiopharm, Motif, Durata, MicuRx, Bristol-Myers Squibb, and Bayer.
AT ASM MICROBE 2017
Key clinical point: Sodium fusidate, active as fusidic acid, showed noninferiority to linezolid for early clinical response in ABSSI patients.
Major finding: 87.2% of patients given sodium fusidate and 86.6% of those receiving linezolid achieved an early clinical response.
Data source: Randomized, controlled, double-blind, phase 3 study with 716 participants.
Disclosures: Cempra sponsored the study. Dr. Carrier Cardenas is a researcher for Cempra, Paratek, Debiopharm, Motif, Durata, MicuRx, Bristol-Myers Squibb, and Bayer. Dr. Strayer is a Cempra employee and shareholder.
Ventricular assist devices linked to sepsis
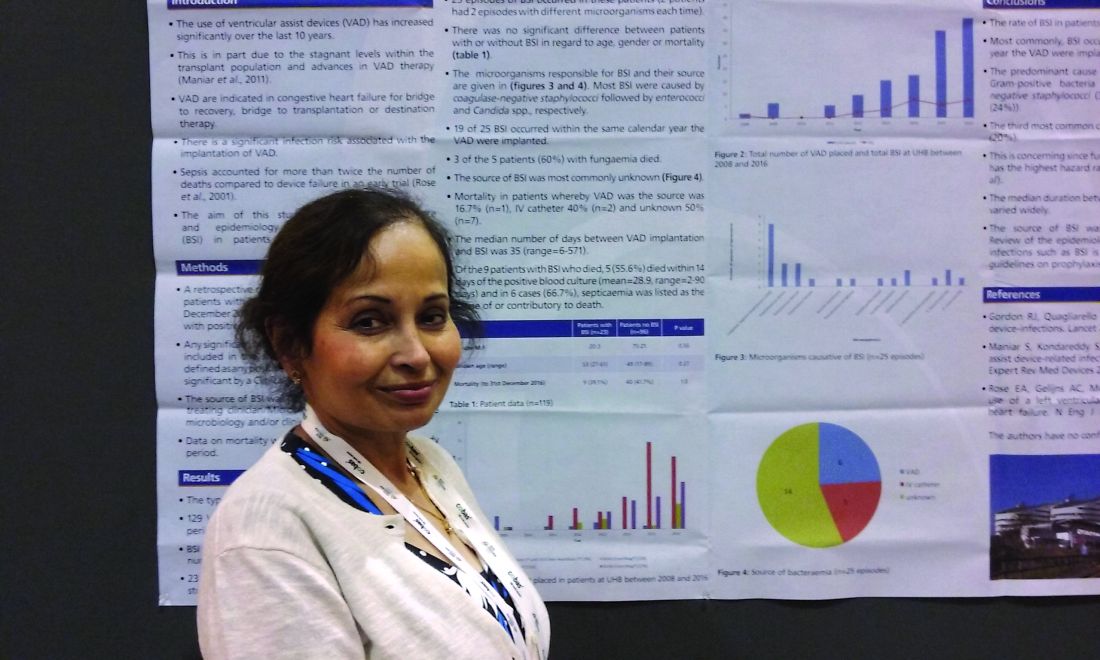
NEW ORLEANS – Back in 2008, there was only one case.
Since then, however, the number of patients with ventricular assist devices who developed sepsis while being treated in the cardiac unit at Queen Elizabeth Hospital in Birmingham, England, appeared to be noticeably growing. So, investigators launched a study to confirm their suspicions and to learn more about the underlying causes.
“Bloodstream infection is a serious infection, so I thought, ‘Let’s see what’s happening,’ ” explained Ira Das, MD, a consultant microbiologist at Queen Elizabeth Hospital.
Coagulase-negative staphylococci were the most common cause, present in 32% of the 25 cases. Sepsis was caused by Enterococcus faecium in 12%, Candida parapsilosis in 8%, and Staphylococcus aureus in 2%. Another 4% were either Enterococcus faecalis, Serratia marcescens, Pseudomonas aeruginosa, C. guilliermondii, or C. orthopsilosis. The remaining 16% of bloodstream infections were polymicrobial.
Less certain was the source of these infections.
“In the majority of cases, we didn’t know where it was coming from,” Dr. Das said at the annual meeting of the American Society for Microbiology. In 6 of the 25 cases, VAD was confirmed to be the focus of infection, either through imaging or because a failing component of the explanted device was examined later. An intravascular catheter was the source in another 5 patients, and in 14 cases, the source remained a mystery.
“Some of these infections just might have been hard to see,” Dr. Das said. “If the infection is inside the device, it’s not always easy to visualize.”
The study supports earlier findings from a review article that points to a significant infection risk associated with the implantation of VADs (Expert Rev Med Devices. 2011 Sep;8[5]:627-34). That article’s authors noted, “Despite recent improvements in outcomes, device-related infections remain a significant complication of LVAD [left ventricular assist device] therapy.”
In a previous study of people with end-stage heart failure, other investigators noted that, “despite the substantial survival benefit, the morbidity and mortality associated with the use of the left ventricular assist device were considerable. In particular, infection and mechanical failure of the device were major factors in the 2-year survival rate of only 23%” (N Engl J Med. 2001 Nov 15;345[20]:1435-43).
Similarly, in the current study, mortality was higher among those with sepsis and a VAD. Mortality was 39% – including eight patients who died with a VAD in situ and one following cardiac transplantation. However, Dr. Das cautioned, “It’s a small number, and there are other factors that could have contributed. They all go on anticoagulants so they have bleeding tendencies, and many of the patients are in the ICU with multiorgan failure.”
Infection prevention remains paramount to minimize mortality and other adverse events associated with a patient’s having a VAD. “We have to make sure that infection control procedures and our treatments are up to the optimal standard,” Dr. Das said. “It’s not easy to remove the device.”
Of the 129 VADs implanted, 68 were long-term LVADs, 11 were short-term LVADs, 15 were right ventricular devices, and 35 were biventricular devices.
The study is ongoing. The data presented at the meeting were collected up until December 2016.
“Since then, I’ve seen two more cases, and – very interestingly – one was Haemophilus influenzae,” Dr. Das said. “The patient was on the device, he was at home, and he came in with bacteremia.” Again, the source of infection proved elusive. “With H. influenzae, you would think it was coming from his chest, but the chest x-ray was normal.”
The second case, a patient with a coagulase-negative staphylococci bloodstream infection, was scheduled for a PET scan at the time of Dr. Das’ presentation to try to identify the source of infection.
Dr. Das had no relevant disclosures.
Modern technology saves our patients' lives, but there is always another side to the coin. Reports that LVAD devices are associated with a high incidence of bloodstream infections is important for future clinical practice. The fact that the causes and risk factors for these infections are unknown make this phenomena one of high interest.
Modern technology saves our patients' lives, but there is always another side to the coin. Reports that LVAD devices are associated with a high incidence of bloodstream infections is important for future clinical practice. The fact that the causes and risk factors for these infections are unknown make this phenomena one of high interest.
Modern technology saves our patients' lives, but there is always another side to the coin. Reports that LVAD devices are associated with a high incidence of bloodstream infections is important for future clinical practice. The fact that the causes and risk factors for these infections are unknown make this phenomena one of high interest.
NEW ORLEANS – Back in 2008, there was only one case.
Since then, however, the number of patients with ventricular assist devices who developed sepsis while being treated in the cardiac unit at Queen Elizabeth Hospital in Birmingham, England, appeared to be noticeably growing. So, investigators launched a study to confirm their suspicions and to learn more about the underlying causes.
“Bloodstream infection is a serious infection, so I thought, ‘Let’s see what’s happening,’ ” explained Ira Das, MD, a consultant microbiologist at Queen Elizabeth Hospital.
Coagulase-negative staphylococci were the most common cause, present in 32% of the 25 cases. Sepsis was caused by Enterococcus faecium in 12%, Candida parapsilosis in 8%, and Staphylococcus aureus in 2%. Another 4% were either Enterococcus faecalis, Serratia marcescens, Pseudomonas aeruginosa, C. guilliermondii, or C. orthopsilosis. The remaining 16% of bloodstream infections were polymicrobial.
Less certain was the source of these infections.
“In the majority of cases, we didn’t know where it was coming from,” Dr. Das said at the annual meeting of the American Society for Microbiology. In 6 of the 25 cases, VAD was confirmed to be the focus of infection, either through imaging or because a failing component of the explanted device was examined later. An intravascular catheter was the source in another 5 patients, and in 14 cases, the source remained a mystery.
“Some of these infections just might have been hard to see,” Dr. Das said. “If the infection is inside the device, it’s not always easy to visualize.”
The study supports earlier findings from a review article that points to a significant infection risk associated with the implantation of VADs (Expert Rev Med Devices. 2011 Sep;8[5]:627-34). That article’s authors noted, “Despite recent improvements in outcomes, device-related infections remain a significant complication of LVAD [left ventricular assist device] therapy.”
In a previous study of people with end-stage heart failure, other investigators noted that, “despite the substantial survival benefit, the morbidity and mortality associated with the use of the left ventricular assist device were considerable. In particular, infection and mechanical failure of the device were major factors in the 2-year survival rate of only 23%” (N Engl J Med. 2001 Nov 15;345[20]:1435-43).
Similarly, in the current study, mortality was higher among those with sepsis and a VAD. Mortality was 39% – including eight patients who died with a VAD in situ and one following cardiac transplantation. However, Dr. Das cautioned, “It’s a small number, and there are other factors that could have contributed. They all go on anticoagulants so they have bleeding tendencies, and many of the patients are in the ICU with multiorgan failure.”
Infection prevention remains paramount to minimize mortality and other adverse events associated with a patient’s having a VAD. “We have to make sure that infection control procedures and our treatments are up to the optimal standard,” Dr. Das said. “It’s not easy to remove the device.”
Of the 129 VADs implanted, 68 were long-term LVADs, 11 were short-term LVADs, 15 were right ventricular devices, and 35 were biventricular devices.
The study is ongoing. The data presented at the meeting were collected up until December 2016.
“Since then, I’ve seen two more cases, and – very interestingly – one was Haemophilus influenzae,” Dr. Das said. “The patient was on the device, he was at home, and he came in with bacteremia.” Again, the source of infection proved elusive. “With H. influenzae, you would think it was coming from his chest, but the chest x-ray was normal.”
The second case, a patient with a coagulase-negative staphylococci bloodstream infection, was scheduled for a PET scan at the time of Dr. Das’ presentation to try to identify the source of infection.
Dr. Das had no relevant disclosures.
NEW ORLEANS – Back in 2008, there was only one case.
Since then, however, the number of patients with ventricular assist devices who developed sepsis while being treated in the cardiac unit at Queen Elizabeth Hospital in Birmingham, England, appeared to be noticeably growing. So, investigators launched a study to confirm their suspicions and to learn more about the underlying causes.
“Bloodstream infection is a serious infection, so I thought, ‘Let’s see what’s happening,’ ” explained Ira Das, MD, a consultant microbiologist at Queen Elizabeth Hospital.
Coagulase-negative staphylococci were the most common cause, present in 32% of the 25 cases. Sepsis was caused by Enterococcus faecium in 12%, Candida parapsilosis in 8%, and Staphylococcus aureus in 2%. Another 4% were either Enterococcus faecalis, Serratia marcescens, Pseudomonas aeruginosa, C. guilliermondii, or C. orthopsilosis. The remaining 16% of bloodstream infections were polymicrobial.
Less certain was the source of these infections.
“In the majority of cases, we didn’t know where it was coming from,” Dr. Das said at the annual meeting of the American Society for Microbiology. In 6 of the 25 cases, VAD was confirmed to be the focus of infection, either through imaging or because a failing component of the explanted device was examined later. An intravascular catheter was the source in another 5 patients, and in 14 cases, the source remained a mystery.
“Some of these infections just might have been hard to see,” Dr. Das said. “If the infection is inside the device, it’s not always easy to visualize.”
The study supports earlier findings from a review article that points to a significant infection risk associated with the implantation of VADs (Expert Rev Med Devices. 2011 Sep;8[5]:627-34). That article’s authors noted, “Despite recent improvements in outcomes, device-related infections remain a significant complication of LVAD [left ventricular assist device] therapy.”
In a previous study of people with end-stage heart failure, other investigators noted that, “despite the substantial survival benefit, the morbidity and mortality associated with the use of the left ventricular assist device were considerable. In particular, infection and mechanical failure of the device were major factors in the 2-year survival rate of only 23%” (N Engl J Med. 2001 Nov 15;345[20]:1435-43).
Similarly, in the current study, mortality was higher among those with sepsis and a VAD. Mortality was 39% – including eight patients who died with a VAD in situ and one following cardiac transplantation. However, Dr. Das cautioned, “It’s a small number, and there are other factors that could have contributed. They all go on anticoagulants so they have bleeding tendencies, and many of the patients are in the ICU with multiorgan failure.”
Infection prevention remains paramount to minimize mortality and other adverse events associated with a patient’s having a VAD. “We have to make sure that infection control procedures and our treatments are up to the optimal standard,” Dr. Das said. “It’s not easy to remove the device.”
Of the 129 VADs implanted, 68 were long-term LVADs, 11 were short-term LVADs, 15 were right ventricular devices, and 35 were biventricular devices.
The study is ongoing. The data presented at the meeting were collected up until December 2016.
“Since then, I’ve seen two more cases, and – very interestingly – one was Haemophilus influenzae,” Dr. Das said. “The patient was on the device, he was at home, and he came in with bacteremia.” Again, the source of infection proved elusive. “With H. influenzae, you would think it was coming from his chest, but the chest x-ray was normal.”
The second case, a patient with a coagulase-negative staphylococci bloodstream infection, was scheduled for a PET scan at the time of Dr. Das’ presentation to try to identify the source of infection.
Dr. Das had no relevant disclosures.
AT ASM MICROBE 2017
Key clinical point: There may be a significant rate of bloodstream infections among people with a ventricular assist device.
Major finding: A total of 20% of the 118 people with a VAD had a bloodstream infection.
Data source: A retrospective study of 129 ventricular assist devices placed in 118 people between 2008 and 2016.
Disclosures: Dr. Das had no relevant disclosures.
Expanded urine culture identified more pathogens
NEW ORLEANS – With the trade-off of an extra 24 hours for results, an enhanced protocol to culture clinically relevant urinary pathogens detected significantly more unique pathogens associated with urinary tract infection, compared with standard cultures, in a study of 150 women.
“What we were able to see is that for about 90% of the samples that were called negative by standard [approach], we were able to detect bacteria through our protocol,” said Travis K. Price, a PhD candidate in the department of microbiology and immunology at Loyola University, Chicago.
Typically, when a urine sample is cultured for a UTI at Loyola University Medical Center, the standard protocol is for the lab to test 1 mcL of urine using agar plates incubated aerobically for 24 hours, Mr. Price said. “When we’re testing the urinary microbiome, we expand on that protocol. We use 100 times more urine, different plates, different environmental conditions, and we hold them for 48 hours instead of 24.”
The investigators prospectively recruited 150 women coming in to the urogynecology clinic – half who felt they had a UTI that day, half who did not. “We wanted to understand if using our enhanced protocol was beneficial and essentially leading to better patient outcomes,” Mr. Price said at the annual meeting of the American Society for Microbiology.
“Among the women who felt they had a UTI, standard culture only picked up 50% of the pathogens we were picking up with our protocol,” Mr. Price said. “And when we looked closer, we realized most of that was Escherichia coli.” Excluding samples positive for E. coli, standard culture detected only 12% of UTI pathogens, he added, compared with 77% detected with the expanded quantitative protocol.
The expanded protocol detected significantly more unique pathogen species, 95, compared with 11 with standard cultures. In addition, of all the uropathogens detected by the new protocol, the standard protocol missed 67%, or 122 of the total 182.
In terms of clinical practicality, Mr. Price and his colleagues looked at “different conditions, multiple volumes of urine, different plates, 24 versus 48 hours – and at the end tried to figure out what is the least amount of work you can do to get the most information.” They then developed a streamlined protocol that involves 100 mcL of urine, a CNA agar plate that selects for gram-positive organisms, a MacConkey agar using 5% CO2, and 48 hours of incubation. “It’s easy to implement,” he added. “The only issue is the longer incubation time could lead to delayed treatment, potentially.”
The streamlined protocol detected more uropathogens – 152 of the 182, for an 84% detection rate – compared with standard cultures, which detected 60 of the 182, or 33%.
The streamlined protocol markedly improved uropathogen detection, the authors wrote. “These findings support the necessity for an immediate change in urine culture procedures.”
Another aim of the study was to evaluate the optimal threshold for UTI colony counts. Traditionally, the cutoff is set at 105 colonies or greater for diagnosis of a UTI, Mr. Price said. “We found there were always higher pathogen colony counts in people who thought they had a UTI. But there wasn’t one threshold that would have caught all of these.”
Next, the investigators looked for a correlation between the colony count cutoff and clinical outcomes. “For people who had a colony count greater than 105 – typically, it was a gram-negative organism – most people were treated with an antibiotic, and a week later most people, 62%, reported feeling better,” Mr. Price said. “But people who didn’t have a pathogen greater than 105, some were not treated, and when we called them a week later, most reported they were not feeling better. ... This suggests this threshold is not actually appropriate.”
Going forward, the investigators just started a clinical trial using the enhanced culture to confirm whether or not their protocol leads to better outcomes for women with UTIs.
Mr. Price did not have any relevant disclosures.
NEW ORLEANS – With the trade-off of an extra 24 hours for results, an enhanced protocol to culture clinically relevant urinary pathogens detected significantly more unique pathogens associated with urinary tract infection, compared with standard cultures, in a study of 150 women.
“What we were able to see is that for about 90% of the samples that were called negative by standard [approach], we were able to detect bacteria through our protocol,” said Travis K. Price, a PhD candidate in the department of microbiology and immunology at Loyola University, Chicago.
Typically, when a urine sample is cultured for a UTI at Loyola University Medical Center, the standard protocol is for the lab to test 1 mcL of urine using agar plates incubated aerobically for 24 hours, Mr. Price said. “When we’re testing the urinary microbiome, we expand on that protocol. We use 100 times more urine, different plates, different environmental conditions, and we hold them for 48 hours instead of 24.”
The investigators prospectively recruited 150 women coming in to the urogynecology clinic – half who felt they had a UTI that day, half who did not. “We wanted to understand if using our enhanced protocol was beneficial and essentially leading to better patient outcomes,” Mr. Price said at the annual meeting of the American Society for Microbiology.
“Among the women who felt they had a UTI, standard culture only picked up 50% of the pathogens we were picking up with our protocol,” Mr. Price said. “And when we looked closer, we realized most of that was Escherichia coli.” Excluding samples positive for E. coli, standard culture detected only 12% of UTI pathogens, he added, compared with 77% detected with the expanded quantitative protocol.
The expanded protocol detected significantly more unique pathogen species, 95, compared with 11 with standard cultures. In addition, of all the uropathogens detected by the new protocol, the standard protocol missed 67%, or 122 of the total 182.
In terms of clinical practicality, Mr. Price and his colleagues looked at “different conditions, multiple volumes of urine, different plates, 24 versus 48 hours – and at the end tried to figure out what is the least amount of work you can do to get the most information.” They then developed a streamlined protocol that involves 100 mcL of urine, a CNA agar plate that selects for gram-positive organisms, a MacConkey agar using 5% CO2, and 48 hours of incubation. “It’s easy to implement,” he added. “The only issue is the longer incubation time could lead to delayed treatment, potentially.”
The streamlined protocol detected more uropathogens – 152 of the 182, for an 84% detection rate – compared with standard cultures, which detected 60 of the 182, or 33%.
The streamlined protocol markedly improved uropathogen detection, the authors wrote. “These findings support the necessity for an immediate change in urine culture procedures.”
Another aim of the study was to evaluate the optimal threshold for UTI colony counts. Traditionally, the cutoff is set at 105 colonies or greater for diagnosis of a UTI, Mr. Price said. “We found there were always higher pathogen colony counts in people who thought they had a UTI. But there wasn’t one threshold that would have caught all of these.”
Next, the investigators looked for a correlation between the colony count cutoff and clinical outcomes. “For people who had a colony count greater than 105 – typically, it was a gram-negative organism – most people were treated with an antibiotic, and a week later most people, 62%, reported feeling better,” Mr. Price said. “But people who didn’t have a pathogen greater than 105, some were not treated, and when we called them a week later, most reported they were not feeling better. ... This suggests this threshold is not actually appropriate.”
Going forward, the investigators just started a clinical trial using the enhanced culture to confirm whether or not their protocol leads to better outcomes for women with UTIs.
Mr. Price did not have any relevant disclosures.
NEW ORLEANS – With the trade-off of an extra 24 hours for results, an enhanced protocol to culture clinically relevant urinary pathogens detected significantly more unique pathogens associated with urinary tract infection, compared with standard cultures, in a study of 150 women.
“What we were able to see is that for about 90% of the samples that were called negative by standard [approach], we were able to detect bacteria through our protocol,” said Travis K. Price, a PhD candidate in the department of microbiology and immunology at Loyola University, Chicago.
Typically, when a urine sample is cultured for a UTI at Loyola University Medical Center, the standard protocol is for the lab to test 1 mcL of urine using agar plates incubated aerobically for 24 hours, Mr. Price said. “When we’re testing the urinary microbiome, we expand on that protocol. We use 100 times more urine, different plates, different environmental conditions, and we hold them for 48 hours instead of 24.”
The investigators prospectively recruited 150 women coming in to the urogynecology clinic – half who felt they had a UTI that day, half who did not. “We wanted to understand if using our enhanced protocol was beneficial and essentially leading to better patient outcomes,” Mr. Price said at the annual meeting of the American Society for Microbiology.
“Among the women who felt they had a UTI, standard culture only picked up 50% of the pathogens we were picking up with our protocol,” Mr. Price said. “And when we looked closer, we realized most of that was Escherichia coli.” Excluding samples positive for E. coli, standard culture detected only 12% of UTI pathogens, he added, compared with 77% detected with the expanded quantitative protocol.
The expanded protocol detected significantly more unique pathogen species, 95, compared with 11 with standard cultures. In addition, of all the uropathogens detected by the new protocol, the standard protocol missed 67%, or 122 of the total 182.
In terms of clinical practicality, Mr. Price and his colleagues looked at “different conditions, multiple volumes of urine, different plates, 24 versus 48 hours – and at the end tried to figure out what is the least amount of work you can do to get the most information.” They then developed a streamlined protocol that involves 100 mcL of urine, a CNA agar plate that selects for gram-positive organisms, a MacConkey agar using 5% CO2, and 48 hours of incubation. “It’s easy to implement,” he added. “The only issue is the longer incubation time could lead to delayed treatment, potentially.”
The streamlined protocol detected more uropathogens – 152 of the 182, for an 84% detection rate – compared with standard cultures, which detected 60 of the 182, or 33%.
The streamlined protocol markedly improved uropathogen detection, the authors wrote. “These findings support the necessity for an immediate change in urine culture procedures.”
Another aim of the study was to evaluate the optimal threshold for UTI colony counts. Traditionally, the cutoff is set at 105 colonies or greater for diagnosis of a UTI, Mr. Price said. “We found there were always higher pathogen colony counts in people who thought they had a UTI. But there wasn’t one threshold that would have caught all of these.”
Next, the investigators looked for a correlation between the colony count cutoff and clinical outcomes. “For people who had a colony count greater than 105 – typically, it was a gram-negative organism – most people were treated with an antibiotic, and a week later most people, 62%, reported feeling better,” Mr. Price said. “But people who didn’t have a pathogen greater than 105, some were not treated, and when we called them a week later, most reported they were not feeling better. ... This suggests this threshold is not actually appropriate.”
Going forward, the investigators just started a clinical trial using the enhanced culture to confirm whether or not their protocol leads to better outcomes for women with UTIs.
Mr. Price did not have any relevant disclosures.
AT ASM MICROBE 2017
Key clinical point:
Major finding: Standard cultures missed 67% (122 of 182) of the uropathogens identified with the expanded culture protocol.
Data source: A prospective study of 150 women comparing UTI pathogen detection between standard and expanded culture analysis.
Disclosures: Mr. Price did not have any relevant disclosures.
Adding cefepime to vancomycin improved MRSA bacteremia outcomes
NEW ORLEANS – Compared with vancomycin monotherapy, vancomycin combined with cefepime improved some outcomes for patients with methicillin-resistant Staphylococcus aureus (MRSA) bloodstream infections, a retrospective study of 109 patients revealed.
A lower likelihood of microbiological failure and fewer bloodstream infections persisting 7 days or more were the notable differences between treatment groups.
All patients had at least 72 hours of vancomycin therapy to treat MRSA bacteremia confirmed by blood culture. During 2008-2015, 38 adults received vancomycin monotherapy and 71 received vancomycin plus 24 hours or more of cefepime.
Compared with monotherapy, the combination treatment was associated with a nonsignificant reduction in the primary composite treatment failure outcome of 30-day all-cause mortality, in bacteremia duration of 7 days or more, and in 60-day bloodstream-infection recurrence: 55% for monotherapy versus 42% for combination therapy (P = .195). The difference was primarily associated with decreased duration of sepsis and fewer MRSA bloodstream infections persisting 7 days or more in the combination cohort.
Rates of bacteremia duration of 7 days or more were 42% in monotherapy patients and 20% in combination patients (P = .013). Differences in 60-day bloodstream-infection recurrence were nonsignificant, 8% versus 4%, respectively (P = .42).
Thirty-day mortality, however, was lower among monotherapy patients than combination patients – 13% vs. 25% – although the difference was nonsignificant (P = .21).
“From what I see here … it seems like they will have a lower duration of bacteremia, which is always great,” Ms. Atwan said. “You want to decrease length of stay in the hospital,” which will cut down on costs and on patients’ risks of getting more infections.
Although the primary outcome was a composite endpoint, “when we looked at them separately, we found the patients in the combination group had more mortality,” Ms. Atwan said at the annual meeting of the American Society for Microbiology. “That surprised me initially. But those patients are sicker and more likely to get dual coverage.”
The investigators confirmed the association between the severity of MRSA bacteremia and combination therapy by looking at Acute Physiology and Chronic Health Evaluation (APACHE II) scores. The median APACHE score was 23 in the combination group, compared with 13.5 in the monotherapy group (P = 0003). Higher APACHE scores were associated with greater odds of meeting the composite failure endpoint (adjusted odds ratio, 1.08) and of developing endocarditis (aOR, 3.6) in multivariate analyses.
More patients in the combination group had pneumonia as the primary source of infection than did patients in the monotherapy group: 54% vs. 29% (P = .016). Further, more of them had skin or soft tissue infections as the primary infection source: 29% vs. 13% (P = .036).
Although the exact mechanism remains unknown, synergy between the two agents could be caused by an increase in penicillin-binding proteins, Ms. Atwan said.
The study is still ongoing; Ms. Atwan hopes additional patients and data will lead to statistically significant differences between the outcomes of combination therapy and vancomycin monotherapy.
“I want to say that combination therapy is something you will always want to go to when you have a sicker patient, but I can’t really tell you that combination therapy is going to cause better outcomes for your patient,” she cautioned. “Hopefully, I can by the end of the study.”
In the meantime, “it looks like vancomycin and beta-lactams could be beneficial for MRSA bacteremia,” she added.
The researchers noted that although vancomycin monotherapy is a mainstay of treatment for MRSA bloodstream infections, emergence of reduced susceptibility and treatment failures warrants other therapeutic strategies.
Ms. Atwan had no relevant financial disclosures.
NEW ORLEANS – Compared with vancomycin monotherapy, vancomycin combined with cefepime improved some outcomes for patients with methicillin-resistant Staphylococcus aureus (MRSA) bloodstream infections, a retrospective study of 109 patients revealed.
A lower likelihood of microbiological failure and fewer bloodstream infections persisting 7 days or more were the notable differences between treatment groups.
All patients had at least 72 hours of vancomycin therapy to treat MRSA bacteremia confirmed by blood culture. During 2008-2015, 38 adults received vancomycin monotherapy and 71 received vancomycin plus 24 hours or more of cefepime.
Compared with monotherapy, the combination treatment was associated with a nonsignificant reduction in the primary composite treatment failure outcome of 30-day all-cause mortality, in bacteremia duration of 7 days or more, and in 60-day bloodstream-infection recurrence: 55% for monotherapy versus 42% for combination therapy (P = .195). The difference was primarily associated with decreased duration of sepsis and fewer MRSA bloodstream infections persisting 7 days or more in the combination cohort.
Rates of bacteremia duration of 7 days or more were 42% in monotherapy patients and 20% in combination patients (P = .013). Differences in 60-day bloodstream-infection recurrence were nonsignificant, 8% versus 4%, respectively (P = .42).
Thirty-day mortality, however, was lower among monotherapy patients than combination patients – 13% vs. 25% – although the difference was nonsignificant (P = .21).
“From what I see here … it seems like they will have a lower duration of bacteremia, which is always great,” Ms. Atwan said. “You want to decrease length of stay in the hospital,” which will cut down on costs and on patients’ risks of getting more infections.
Although the primary outcome was a composite endpoint, “when we looked at them separately, we found the patients in the combination group had more mortality,” Ms. Atwan said at the annual meeting of the American Society for Microbiology. “That surprised me initially. But those patients are sicker and more likely to get dual coverage.”
The investigators confirmed the association between the severity of MRSA bacteremia and combination therapy by looking at Acute Physiology and Chronic Health Evaluation (APACHE II) scores. The median APACHE score was 23 in the combination group, compared with 13.5 in the monotherapy group (P = 0003). Higher APACHE scores were associated with greater odds of meeting the composite failure endpoint (adjusted odds ratio, 1.08) and of developing endocarditis (aOR, 3.6) in multivariate analyses.
More patients in the combination group had pneumonia as the primary source of infection than did patients in the monotherapy group: 54% vs. 29% (P = .016). Further, more of them had skin or soft tissue infections as the primary infection source: 29% vs. 13% (P = .036).
Although the exact mechanism remains unknown, synergy between the two agents could be caused by an increase in penicillin-binding proteins, Ms. Atwan said.
The study is still ongoing; Ms. Atwan hopes additional patients and data will lead to statistically significant differences between the outcomes of combination therapy and vancomycin monotherapy.
“I want to say that combination therapy is something you will always want to go to when you have a sicker patient, but I can’t really tell you that combination therapy is going to cause better outcomes for your patient,” she cautioned. “Hopefully, I can by the end of the study.”
In the meantime, “it looks like vancomycin and beta-lactams could be beneficial for MRSA bacteremia,” she added.
The researchers noted that although vancomycin monotherapy is a mainstay of treatment for MRSA bloodstream infections, emergence of reduced susceptibility and treatment failures warrants other therapeutic strategies.
Ms. Atwan had no relevant financial disclosures.
NEW ORLEANS – Compared with vancomycin monotherapy, vancomycin combined with cefepime improved some outcomes for patients with methicillin-resistant Staphylococcus aureus (MRSA) bloodstream infections, a retrospective study of 109 patients revealed.
A lower likelihood of microbiological failure and fewer bloodstream infections persisting 7 days or more were the notable differences between treatment groups.
All patients had at least 72 hours of vancomycin therapy to treat MRSA bacteremia confirmed by blood culture. During 2008-2015, 38 adults received vancomycin monotherapy and 71 received vancomycin plus 24 hours or more of cefepime.
Compared with monotherapy, the combination treatment was associated with a nonsignificant reduction in the primary composite treatment failure outcome of 30-day all-cause mortality, in bacteremia duration of 7 days or more, and in 60-day bloodstream-infection recurrence: 55% for monotherapy versus 42% for combination therapy (P = .195). The difference was primarily associated with decreased duration of sepsis and fewer MRSA bloodstream infections persisting 7 days or more in the combination cohort.
Rates of bacteremia duration of 7 days or more were 42% in monotherapy patients and 20% in combination patients (P = .013). Differences in 60-day bloodstream-infection recurrence were nonsignificant, 8% versus 4%, respectively (P = .42).
Thirty-day mortality, however, was lower among monotherapy patients than combination patients – 13% vs. 25% – although the difference was nonsignificant (P = .21).
“From what I see here … it seems like they will have a lower duration of bacteremia, which is always great,” Ms. Atwan said. “You want to decrease length of stay in the hospital,” which will cut down on costs and on patients’ risks of getting more infections.
Although the primary outcome was a composite endpoint, “when we looked at them separately, we found the patients in the combination group had more mortality,” Ms. Atwan said at the annual meeting of the American Society for Microbiology. “That surprised me initially. But those patients are sicker and more likely to get dual coverage.”
The investigators confirmed the association between the severity of MRSA bacteremia and combination therapy by looking at Acute Physiology and Chronic Health Evaluation (APACHE II) scores. The median APACHE score was 23 in the combination group, compared with 13.5 in the monotherapy group (P = 0003). Higher APACHE scores were associated with greater odds of meeting the composite failure endpoint (adjusted odds ratio, 1.08) and of developing endocarditis (aOR, 3.6) in multivariate analyses.
More patients in the combination group had pneumonia as the primary source of infection than did patients in the monotherapy group: 54% vs. 29% (P = .016). Further, more of them had skin or soft tissue infections as the primary infection source: 29% vs. 13% (P = .036).
Although the exact mechanism remains unknown, synergy between the two agents could be caused by an increase in penicillin-binding proteins, Ms. Atwan said.
The study is still ongoing; Ms. Atwan hopes additional patients and data will lead to statistically significant differences between the outcomes of combination therapy and vancomycin monotherapy.
“I want to say that combination therapy is something you will always want to go to when you have a sicker patient, but I can’t really tell you that combination therapy is going to cause better outcomes for your patient,” she cautioned. “Hopefully, I can by the end of the study.”
In the meantime, “it looks like vancomycin and beta-lactams could be beneficial for MRSA bacteremia,” she added.
The researchers noted that although vancomycin monotherapy is a mainstay of treatment for MRSA bloodstream infections, emergence of reduced susceptibility and treatment failures warrants other therapeutic strategies.
Ms. Atwan had no relevant financial disclosures.
AT ASM MICROBE 2017
Key clinical point:
Major finding: Median duration of MRSA bacteremia was 4 days with combination therapy, versus 6 days with vancomycin alone.
Data source: A retrospective, single-center comparison of 109 patients treated with either vancomycin plus cefepime or vancomycin alone.
Disclosures: Safana M. Atwan had no relevant financial disclosures.
Vancomycin research reveals reasons for readmissions and prolonged stays
NEW ORLEANS – Approximately 20% of patients treated with vancomycin for an acute bacterial skin and skin structure infection remained in the hospital 8 days or longer, and about 7% experienced a readmission within 30 days, a retrospective study of 507 patients in the Geisinger Health System database showed.
“We found, for those who had a readmission, the major drivers were those who are your ‘health care frequent flyers’ – those who were admitted in the past 6 months,” said Thomas Lodise, PharmD, PhD, professor of pharmacy practice at Albany (N.Y.) College of Pharmacy and Health Sciences. “So, patients with a previous hospitalization are more likely to be treated again for all-cause admission within 30 days of discharge.” In addition, people with a lower-extremity abscess, particularly older patients with diabetes, and those with a traumatic wound were also more likely to return within 30 days.
Identifying the population at highest readmission risk could become more important soon. “Even though hospitals are not penalized for skin infection readmissions, there is some talk of adding that to the Medicare penalty,” Dr. Lodise said.
“It’s under review now by Medicare,” said Kenneth LaPensee, PhD, a consultant for Paratek Pharmaceuticals, King of Prussia, Pa., a firm developing an antibiotic to treat patients with an acute bacterial skin and skin structure infection (ABSSSI).
Dr. Lodise, Dr. LaPensee, and their colleagues studied adults hospitalized for an ABSSSI primary diagnosis based on ICD-9 codes and treated with at least 2 days of vancomycin. Participants were seen within the integrated Geisinger Health System between 2010 and 2015.
A total 6.9% of patients were readmitted within 30 days or had repeat emergency department visits. That group included more people with a body mass index of 36 kg/m2 or greater – 8.4%, compared with 6.2% of those with a BMI of less than 36. Other factors associated with readmission included smoking or a history of smoking (9.8% vs. 2.7% for nonsmokers) and a history of ABSSSI not requiring hospitalization vs. no prior history of ABSSSI, 22.2% vs. 6.6%, respectively.
In addition, those with a prior hospital admission not related to skin and skin structure infection were at higher risk, 8.8%, compared with 6.3% for those with no prior hospital admission. A prior hospital outpatient visit was likewise associated with a higher risk for readmission, 12.0%, compared with 5.4% without such a history.
Greater disease severity did not appear to correlate with a higher risk of 30-day readmission in the study. The researchers compared the groups by both Eron classification and the Charlson Comorbidity Index disease severity measures.
“People with more comorbidities had higher readmission rates, but it wasn’t statistically significant,” Dr. Lodise said. Also, “we saw some signaling – like with older age and some differences with race – [that] nonwhites were more likely to get readmitted. A total 6.7% of readmissions were among people older than 65 years, compared with 7.2% of younger people.” Advanced age was a factor in the bivariate analysis, but not in the logistic regression, Dr. LaPensee said.
“We’re going to repeat this in a larger data set. We’re planning for 10,000 patients,” Dr. Lodise said. “In our next cohort, we’ll be able to delineate more patient covariates.” An unanswered question is whether treatment with an agent other than vancomycin could improve readmission rates.
Using the same Geisinger database, the investigators also examined prolonged length of stay for patients with ABSSSIs treated with vancomycin. Almost one-fifth, 19.9%, met their definition of a prolonged stay of 8 days or longer.
The average length of stay was 7 days. “We were a bit surprised. We anticipated it being closer to 4 or 5 days,” Dr. Lodise said. “Then we wanted to find out who are these patients with these prolonged lengths of stay?
“The interesting thing was, things you think would be predictive, like increased age or high BMI [body mass index], were not,” Dr. Lodise said. “But what we did find is being elderly with diabetes and having a lower-extremity infection or a lower-extremity infection with an abscess – this was the group at greatest risk for a prolonged length of stay.” Those findings make sense, he added, because those patients tend to be slow responders, and because intravenous vancomycin has no oral, step-down formulation.
Unlike the readmission risk factors, Eron disease severity “was … very predictive of a prolonged length of stay,” Dr. Lodise said.
“These are really correlated – those patients with longer length of stay are more likely to get readmitted,” Dr. Lodise said. “In these more-difficult-to-treat patient populations, they really need more attention [to figure out] what is going on with them, why they keep coming back to the hospital, or why are they staying in so long.”
Dr. Lodise is a consultant for Paratek Pharmaceuticals, the study sponsor. Dr. LaPensee is a Paratek employee.
NEW ORLEANS – Approximately 20% of patients treated with vancomycin for an acute bacterial skin and skin structure infection remained in the hospital 8 days or longer, and about 7% experienced a readmission within 30 days, a retrospective study of 507 patients in the Geisinger Health System database showed.
“We found, for those who had a readmission, the major drivers were those who are your ‘health care frequent flyers’ – those who were admitted in the past 6 months,” said Thomas Lodise, PharmD, PhD, professor of pharmacy practice at Albany (N.Y.) College of Pharmacy and Health Sciences. “So, patients with a previous hospitalization are more likely to be treated again for all-cause admission within 30 days of discharge.” In addition, people with a lower-extremity abscess, particularly older patients with diabetes, and those with a traumatic wound were also more likely to return within 30 days.
Identifying the population at highest readmission risk could become more important soon. “Even though hospitals are not penalized for skin infection readmissions, there is some talk of adding that to the Medicare penalty,” Dr. Lodise said.
“It’s under review now by Medicare,” said Kenneth LaPensee, PhD, a consultant for Paratek Pharmaceuticals, King of Prussia, Pa., a firm developing an antibiotic to treat patients with an acute bacterial skin and skin structure infection (ABSSSI).
Dr. Lodise, Dr. LaPensee, and their colleagues studied adults hospitalized for an ABSSSI primary diagnosis based on ICD-9 codes and treated with at least 2 days of vancomycin. Participants were seen within the integrated Geisinger Health System between 2010 and 2015.
A total 6.9% of patients were readmitted within 30 days or had repeat emergency department visits. That group included more people with a body mass index of 36 kg/m2 or greater – 8.4%, compared with 6.2% of those with a BMI of less than 36. Other factors associated with readmission included smoking or a history of smoking (9.8% vs. 2.7% for nonsmokers) and a history of ABSSSI not requiring hospitalization vs. no prior history of ABSSSI, 22.2% vs. 6.6%, respectively.
In addition, those with a prior hospital admission not related to skin and skin structure infection were at higher risk, 8.8%, compared with 6.3% for those with no prior hospital admission. A prior hospital outpatient visit was likewise associated with a higher risk for readmission, 12.0%, compared with 5.4% without such a history.
Greater disease severity did not appear to correlate with a higher risk of 30-day readmission in the study. The researchers compared the groups by both Eron classification and the Charlson Comorbidity Index disease severity measures.
“People with more comorbidities had higher readmission rates, but it wasn’t statistically significant,” Dr. Lodise said. Also, “we saw some signaling – like with older age and some differences with race – [that] nonwhites were more likely to get readmitted. A total 6.7% of readmissions were among people older than 65 years, compared with 7.2% of younger people.” Advanced age was a factor in the bivariate analysis, but not in the logistic regression, Dr. LaPensee said.
“We’re going to repeat this in a larger data set. We’re planning for 10,000 patients,” Dr. Lodise said. “In our next cohort, we’ll be able to delineate more patient covariates.” An unanswered question is whether treatment with an agent other than vancomycin could improve readmission rates.
Using the same Geisinger database, the investigators also examined prolonged length of stay for patients with ABSSSIs treated with vancomycin. Almost one-fifth, 19.9%, met their definition of a prolonged stay of 8 days or longer.
The average length of stay was 7 days. “We were a bit surprised. We anticipated it being closer to 4 or 5 days,” Dr. Lodise said. “Then we wanted to find out who are these patients with these prolonged lengths of stay?
“The interesting thing was, things you think would be predictive, like increased age or high BMI [body mass index], were not,” Dr. Lodise said. “But what we did find is being elderly with diabetes and having a lower-extremity infection or a lower-extremity infection with an abscess – this was the group at greatest risk for a prolonged length of stay.” Those findings make sense, he added, because those patients tend to be slow responders, and because intravenous vancomycin has no oral, step-down formulation.
Unlike the readmission risk factors, Eron disease severity “was … very predictive of a prolonged length of stay,” Dr. Lodise said.
“These are really correlated – those patients with longer length of stay are more likely to get readmitted,” Dr. Lodise said. “In these more-difficult-to-treat patient populations, they really need more attention [to figure out] what is going on with them, why they keep coming back to the hospital, or why are they staying in so long.”
Dr. Lodise is a consultant for Paratek Pharmaceuticals, the study sponsor. Dr. LaPensee is a Paratek employee.
NEW ORLEANS – Approximately 20% of patients treated with vancomycin for an acute bacterial skin and skin structure infection remained in the hospital 8 days or longer, and about 7% experienced a readmission within 30 days, a retrospective study of 507 patients in the Geisinger Health System database showed.
“We found, for those who had a readmission, the major drivers were those who are your ‘health care frequent flyers’ – those who were admitted in the past 6 months,” said Thomas Lodise, PharmD, PhD, professor of pharmacy practice at Albany (N.Y.) College of Pharmacy and Health Sciences. “So, patients with a previous hospitalization are more likely to be treated again for all-cause admission within 30 days of discharge.” In addition, people with a lower-extremity abscess, particularly older patients with diabetes, and those with a traumatic wound were also more likely to return within 30 days.
Identifying the population at highest readmission risk could become more important soon. “Even though hospitals are not penalized for skin infection readmissions, there is some talk of adding that to the Medicare penalty,” Dr. Lodise said.
“It’s under review now by Medicare,” said Kenneth LaPensee, PhD, a consultant for Paratek Pharmaceuticals, King of Prussia, Pa., a firm developing an antibiotic to treat patients with an acute bacterial skin and skin structure infection (ABSSSI).
Dr. Lodise, Dr. LaPensee, and their colleagues studied adults hospitalized for an ABSSSI primary diagnosis based on ICD-9 codes and treated with at least 2 days of vancomycin. Participants were seen within the integrated Geisinger Health System between 2010 and 2015.
A total 6.9% of patients were readmitted within 30 days or had repeat emergency department visits. That group included more people with a body mass index of 36 kg/m2 or greater – 8.4%, compared with 6.2% of those with a BMI of less than 36. Other factors associated with readmission included smoking or a history of smoking (9.8% vs. 2.7% for nonsmokers) and a history of ABSSSI not requiring hospitalization vs. no prior history of ABSSSI, 22.2% vs. 6.6%, respectively.
In addition, those with a prior hospital admission not related to skin and skin structure infection were at higher risk, 8.8%, compared with 6.3% for those with no prior hospital admission. A prior hospital outpatient visit was likewise associated with a higher risk for readmission, 12.0%, compared with 5.4% without such a history.
Greater disease severity did not appear to correlate with a higher risk of 30-day readmission in the study. The researchers compared the groups by both Eron classification and the Charlson Comorbidity Index disease severity measures.
“People with more comorbidities had higher readmission rates, but it wasn’t statistically significant,” Dr. Lodise said. Also, “we saw some signaling – like with older age and some differences with race – [that] nonwhites were more likely to get readmitted. A total 6.7% of readmissions were among people older than 65 years, compared with 7.2% of younger people.” Advanced age was a factor in the bivariate analysis, but not in the logistic regression, Dr. LaPensee said.
“We’re going to repeat this in a larger data set. We’re planning for 10,000 patients,” Dr. Lodise said. “In our next cohort, we’ll be able to delineate more patient covariates.” An unanswered question is whether treatment with an agent other than vancomycin could improve readmission rates.
Using the same Geisinger database, the investigators also examined prolonged length of stay for patients with ABSSSIs treated with vancomycin. Almost one-fifth, 19.9%, met their definition of a prolonged stay of 8 days or longer.
The average length of stay was 7 days. “We were a bit surprised. We anticipated it being closer to 4 or 5 days,” Dr. Lodise said. “Then we wanted to find out who are these patients with these prolonged lengths of stay?
“The interesting thing was, things you think would be predictive, like increased age or high BMI [body mass index], were not,” Dr. Lodise said. “But what we did find is being elderly with diabetes and having a lower-extremity infection or a lower-extremity infection with an abscess – this was the group at greatest risk for a prolonged length of stay.” Those findings make sense, he added, because those patients tend to be slow responders, and because intravenous vancomycin has no oral, step-down formulation.
Unlike the readmission risk factors, Eron disease severity “was … very predictive of a prolonged length of stay,” Dr. Lodise said.
“These are really correlated – those patients with longer length of stay are more likely to get readmitted,” Dr. Lodise said. “In these more-difficult-to-treat patient populations, they really need more attention [to figure out] what is going on with them, why they keep coming back to the hospital, or why are they staying in so long.”
Dr. Lodise is a consultant for Paratek Pharmaceuticals, the study sponsor. Dr. LaPensee is a Paratek employee.
AT ASM MICROBE 2017
Key clinical point: Older patients with diabetes and lower-extremity abscesses are at particularly high risk for readmissions and prolonged length of hospital stay.
Major finding: Approximately 20% of patients treated with vancomycin for an acute bacterial skin and skin structure infection remained in the hospital 8 days or longer.
Data source: A review of 507 Geisinger Health System patients with acute bacterial skin and skin structure infections treated with at least 2 days of vancomycin.
Disclosures: Dr. Lodise is a consultant for Paratek Pharmaceuticals, the study sponsor. Dr. LaPensee is a Paratek employee.
Hospital isolates C. difficile carriers and rates drop
NEW ORLEANS – A Montreal hospital grappling with high Clostridium difficile infections rates launched an intervention in October 2013 to screen patients at admission and detect asymptomatic carriers, and investigators found 4.8% of 7,599 people admitted through the ED over 15 months were carriers of C. difficile.
To protect Jewish General Hospital physicians, staff and other patients from potential transmission, these patients were placed in isolation. However, because they were fairly numerous – 1 in 20 admissions – and because infectious disease (ID) experts feared a substantial backlash, these patients were put in less restrictive isolation. They were permitted to share rooms as long as the dividing curtains remained drawn, for example. In addition, clinicians could skip wearing traditional isolation hats and gowns.
The ID team at the hospital considered the intervention a success. “It is estimated we prevented 64 cases over 15 months,” Dr. Longtin said during a packed session at the annual meeting of the American Society for Microbiology.
The hospital’s C. difficile rate dropped from 6.9 per 10,000 patient-days before the screening and isolation protocol to 3.0 per 10,000 during the intervention. The difference was statically significant (P less than .001).
“Compared to other hospitals in the province, we used to be in the middle of the pack [for C. difficile infection rates], and now we are the lowest,” Dr. Longtin said.
Asymptomatic carriers were detected using rectal sampling with sterile swab and polymerase chain reaction analysis. Testing was performed 7 days a week and analyzed once daily, with results generated within 24 hours and documented in the patient chart. Only patients admitted through the ED were screened, which prompted some questions from colleagues, Dr. Longtin said. However, he defends this approach because the 30% or so of patients admitted from the ED tend to spend more days on the ward. The risk of becoming colonized increases steadily with duration of hospitalization. This occurs despite isolating patients with C. difficile infection. Initial results of the study were published in JAMA Internal Medicine (2016 Jun 1;176[6]:796-804).
Risk to health care workers
C. difficile carriers are contagious, but not as much as people with C. difficile infection, Dr. Longtin said. In one study, the microorganism was present on the skin of 61% of symptomatic carriers versus 78% of those infected (Clin Infect Dis. 2007 Oct 15;45[8]:992-8). In addition, C. difficile present on patient skin can be transferred to health care worker hands, even up to 6 weeks after resolution of associated diarrhea (Infect Control Hosp Epidemiol. 2016 Apr;37[4]:475-7).
Prior to the intervention, C. difficile prevention at Jewish General involved guidelines that “have not really changed in the last 20 years,” Dr. Longtin said. Contact precautions around infected patients, hand hygiene, environmental cleaning, and antibiotic stewardship were the main strategies.
“Despite all these measures, we were not completely blocking dissemination of C difficile in our hospital,” Dr. Longtin said. He added that soap and water are better than alcohol for C. difficile, “but honestly not very good. Even the best hand hygiene technique is poorly effective to remove C. difficile. On the other hand – get it? – gloves are very effective. We felt we had to combine hand washing with gloves.”
Hand hygiene compliance increased from 37% to 50% during the intervention, and Dr. Longtin expected further improvements over time.
Risk to other patients
“Transmission of C. difficile cannot only be explained by infected patients in a hospital, so likely carriers also play a role,” Dr. Longtin said.
Another set of investigators found that hospital patients exposed to a carrier of C. difficile had nearly twice the risk of acquiring the infection (odds ratio, 1.79) (Gastroenterology. 2017 Apr;152[5]:1031-41.e2).
“For every patient with C. difficile infection, it’s estimated there are 5-7 C. difficile carriers, so they are numerous as well,” he said.
The bigger picture
During the study period, the C. difficile infection trends did not significantly change on the city level, further supporting the effectiveness of the carrier screen-and-isolate strategy.
There was slight increase in antibiotic use during the intervention period, Dr. Longtin said. “The only type of antibiotics that really decreased were vancomycin and metronidazole... which suggests in turn there were fewer cases of C. difficile infection.”
Long-term follow-up is ongoing, Dr. Longtin said. “We have more than 3 years of intervention. In the past year, our rate was 2.2 per 10,000 patient-days.”
Unanswered questions include the generalizability of the results “because we’re a very pro–infection control hospital,” he said. In addition, a formal cost-benefit analysis of this strategy would be worthwhile in the future.
Dr. Longtin is a consultant for AMG Medical and receives research support from Merck and BD Medical.
NEW ORLEANS – A Montreal hospital grappling with high Clostridium difficile infections rates launched an intervention in October 2013 to screen patients at admission and detect asymptomatic carriers, and investigators found 4.8% of 7,599 people admitted through the ED over 15 months were carriers of C. difficile.
To protect Jewish General Hospital physicians, staff and other patients from potential transmission, these patients were placed in isolation. However, because they were fairly numerous – 1 in 20 admissions – and because infectious disease (ID) experts feared a substantial backlash, these patients were put in less restrictive isolation. They were permitted to share rooms as long as the dividing curtains remained drawn, for example. In addition, clinicians could skip wearing traditional isolation hats and gowns.
The ID team at the hospital considered the intervention a success. “It is estimated we prevented 64 cases over 15 months,” Dr. Longtin said during a packed session at the annual meeting of the American Society for Microbiology.
The hospital’s C. difficile rate dropped from 6.9 per 10,000 patient-days before the screening and isolation protocol to 3.0 per 10,000 during the intervention. The difference was statically significant (P less than .001).
“Compared to other hospitals in the province, we used to be in the middle of the pack [for C. difficile infection rates], and now we are the lowest,” Dr. Longtin said.
Asymptomatic carriers were detected using rectal sampling with sterile swab and polymerase chain reaction analysis. Testing was performed 7 days a week and analyzed once daily, with results generated within 24 hours and documented in the patient chart. Only patients admitted through the ED were screened, which prompted some questions from colleagues, Dr. Longtin said. However, he defends this approach because the 30% or so of patients admitted from the ED tend to spend more days on the ward. The risk of becoming colonized increases steadily with duration of hospitalization. This occurs despite isolating patients with C. difficile infection. Initial results of the study were published in JAMA Internal Medicine (2016 Jun 1;176[6]:796-804).
Risk to health care workers
C. difficile carriers are contagious, but not as much as people with C. difficile infection, Dr. Longtin said. In one study, the microorganism was present on the skin of 61% of symptomatic carriers versus 78% of those infected (Clin Infect Dis. 2007 Oct 15;45[8]:992-8). In addition, C. difficile present on patient skin can be transferred to health care worker hands, even up to 6 weeks after resolution of associated diarrhea (Infect Control Hosp Epidemiol. 2016 Apr;37[4]:475-7).
Prior to the intervention, C. difficile prevention at Jewish General involved guidelines that “have not really changed in the last 20 years,” Dr. Longtin said. Contact precautions around infected patients, hand hygiene, environmental cleaning, and antibiotic stewardship were the main strategies.
“Despite all these measures, we were not completely blocking dissemination of C difficile in our hospital,” Dr. Longtin said. He added that soap and water are better than alcohol for C. difficile, “but honestly not very good. Even the best hand hygiene technique is poorly effective to remove C. difficile. On the other hand – get it? – gloves are very effective. We felt we had to combine hand washing with gloves.”
Hand hygiene compliance increased from 37% to 50% during the intervention, and Dr. Longtin expected further improvements over time.
Risk to other patients
“Transmission of C. difficile cannot only be explained by infected patients in a hospital, so likely carriers also play a role,” Dr. Longtin said.
Another set of investigators found that hospital patients exposed to a carrier of C. difficile had nearly twice the risk of acquiring the infection (odds ratio, 1.79) (Gastroenterology. 2017 Apr;152[5]:1031-41.e2).
“For every patient with C. difficile infection, it’s estimated there are 5-7 C. difficile carriers, so they are numerous as well,” he said.
The bigger picture
During the study period, the C. difficile infection trends did not significantly change on the city level, further supporting the effectiveness of the carrier screen-and-isolate strategy.
There was slight increase in antibiotic use during the intervention period, Dr. Longtin said. “The only type of antibiotics that really decreased were vancomycin and metronidazole... which suggests in turn there were fewer cases of C. difficile infection.”
Long-term follow-up is ongoing, Dr. Longtin said. “We have more than 3 years of intervention. In the past year, our rate was 2.2 per 10,000 patient-days.”
Unanswered questions include the generalizability of the results “because we’re a very pro–infection control hospital,” he said. In addition, a formal cost-benefit analysis of this strategy would be worthwhile in the future.
Dr. Longtin is a consultant for AMG Medical and receives research support from Merck and BD Medical.
NEW ORLEANS – A Montreal hospital grappling with high Clostridium difficile infections rates launched an intervention in October 2013 to screen patients at admission and detect asymptomatic carriers, and investigators found 4.8% of 7,599 people admitted through the ED over 15 months were carriers of C. difficile.
To protect Jewish General Hospital physicians, staff and other patients from potential transmission, these patients were placed in isolation. However, because they were fairly numerous – 1 in 20 admissions – and because infectious disease (ID) experts feared a substantial backlash, these patients were put in less restrictive isolation. They were permitted to share rooms as long as the dividing curtains remained drawn, for example. In addition, clinicians could skip wearing traditional isolation hats and gowns.
The ID team at the hospital considered the intervention a success. “It is estimated we prevented 64 cases over 15 months,” Dr. Longtin said during a packed session at the annual meeting of the American Society for Microbiology.
The hospital’s C. difficile rate dropped from 6.9 per 10,000 patient-days before the screening and isolation protocol to 3.0 per 10,000 during the intervention. The difference was statically significant (P less than .001).
“Compared to other hospitals in the province, we used to be in the middle of the pack [for C. difficile infection rates], and now we are the lowest,” Dr. Longtin said.
Asymptomatic carriers were detected using rectal sampling with sterile swab and polymerase chain reaction analysis. Testing was performed 7 days a week and analyzed once daily, with results generated within 24 hours and documented in the patient chart. Only patients admitted through the ED were screened, which prompted some questions from colleagues, Dr. Longtin said. However, he defends this approach because the 30% or so of patients admitted from the ED tend to spend more days on the ward. The risk of becoming colonized increases steadily with duration of hospitalization. This occurs despite isolating patients with C. difficile infection. Initial results of the study were published in JAMA Internal Medicine (2016 Jun 1;176[6]:796-804).
Risk to health care workers
C. difficile carriers are contagious, but not as much as people with C. difficile infection, Dr. Longtin said. In one study, the microorganism was present on the skin of 61% of symptomatic carriers versus 78% of those infected (Clin Infect Dis. 2007 Oct 15;45[8]:992-8). In addition, C. difficile present on patient skin can be transferred to health care worker hands, even up to 6 weeks after resolution of associated diarrhea (Infect Control Hosp Epidemiol. 2016 Apr;37[4]:475-7).
Prior to the intervention, C. difficile prevention at Jewish General involved guidelines that “have not really changed in the last 20 years,” Dr. Longtin said. Contact precautions around infected patients, hand hygiene, environmental cleaning, and antibiotic stewardship were the main strategies.
“Despite all these measures, we were not completely blocking dissemination of C difficile in our hospital,” Dr. Longtin said. He added that soap and water are better than alcohol for C. difficile, “but honestly not very good. Even the best hand hygiene technique is poorly effective to remove C. difficile. On the other hand – get it? – gloves are very effective. We felt we had to combine hand washing with gloves.”
Hand hygiene compliance increased from 37% to 50% during the intervention, and Dr. Longtin expected further improvements over time.
Risk to other patients
“Transmission of C. difficile cannot only be explained by infected patients in a hospital, so likely carriers also play a role,” Dr. Longtin said.
Another set of investigators found that hospital patients exposed to a carrier of C. difficile had nearly twice the risk of acquiring the infection (odds ratio, 1.79) (Gastroenterology. 2017 Apr;152[5]:1031-41.e2).
“For every patient with C. difficile infection, it’s estimated there are 5-7 C. difficile carriers, so they are numerous as well,” he said.
The bigger picture
During the study period, the C. difficile infection trends did not significantly change on the city level, further supporting the effectiveness of the carrier screen-and-isolate strategy.
There was slight increase in antibiotic use during the intervention period, Dr. Longtin said. “The only type of antibiotics that really decreased were vancomycin and metronidazole... which suggests in turn there were fewer cases of C. difficile infection.”
Long-term follow-up is ongoing, Dr. Longtin said. “We have more than 3 years of intervention. In the past year, our rate was 2.2 per 10,000 patient-days.”
Unanswered questions include the generalizability of the results “because we’re a very pro–infection control hospital,” he said. In addition, a formal cost-benefit analysis of this strategy would be worthwhile in the future.
Dr. Longtin is a consultant for AMG Medical and receives research support from Merck and BD Medical.
AT ASM MICROBE 2017
Key clinical point: Identification and isolation of asymptomatic carriers of Clostridium difficile decreased a hospital’s infection rates over time.
Major finding: (P less than .001).
Data source: A study of 7,599 people screened at admission through the ED at an acute care hospital.
Disclosures: Dr. Longtin is a consultant for AMG Medical and receives research support from Merck and BD Medical.
C. difficile travels on the soles of our shoes
NEW ORLEANS – For an explanation to the spread of Clostridium difficile, one needs to look to our soles.
Based on ribosomal analysis, C. difficile often is transmitted from the hospital into the community and back into the hospital on the soles of shoes, researchers have concluded, based on findings from thousands of samples from patients, hospital environments, and shoes.
Dr. Alam of the University of Houston College of Pharmacy and his colleagues collected thousands of samples from the state of Texas to see how C. difficile strains in the community are related to clinical strains in the hospital.
The researchers collected 3,109 stool samples from people hospitalized with C. difficile, another 1,697 swabs taken from environmental surfaces in hospitals across the state, plus another 400 samples taken from the soles of shoes of clinicians and non–health care workers.
C. difficile was found in 44% of clinical stool samples, 13% of high-touch hospital environment surfaces, and 26% of community shoe sole samples. Among these positive C. difficile samples, toxigenic strains were detected in 93% of patient samples, 66% of hospital environment swabs and 64% of shoe samples. Importantly, the most predominant toxigenic strains appeared in all three sample types.
“When we collected some hospital environmental samples, we saw the isolate ribotypes perfectly matched the patient samples,” Dr. Alam said.
Further, “we saw the exact same ribotypes on our shoe bottoms, from these community, nonclinical sources,” Dr. Alam said. “Apparently, it seems, these dangerous pathogens are everywhere.”
In fact, “we may have brought many different strains from all over the world here to this meeting,” he added. “When we are taking antibiotics, we are susceptible to these different strains.”
Hospitals are cleaned daily, “but how many of us care about the shoes” on those who walk through the hospital, he asked. “We are loading the hospital with Clostridium difficile, and the hospital environment is also loaded with Clostridium difficile so we are bringing it into the community. We are spreading it everywhere.”
“Maybe we are blaming the doctors, nurses, and other staff, but [we are] not thinking about our shoes,” Dr. Alam added.
NEW ORLEANS – For an explanation to the spread of Clostridium difficile, one needs to look to our soles.
Based on ribosomal analysis, C. difficile often is transmitted from the hospital into the community and back into the hospital on the soles of shoes, researchers have concluded, based on findings from thousands of samples from patients, hospital environments, and shoes.
Dr. Alam of the University of Houston College of Pharmacy and his colleagues collected thousands of samples from the state of Texas to see how C. difficile strains in the community are related to clinical strains in the hospital.
The researchers collected 3,109 stool samples from people hospitalized with C. difficile, another 1,697 swabs taken from environmental surfaces in hospitals across the state, plus another 400 samples taken from the soles of shoes of clinicians and non–health care workers.
C. difficile was found in 44% of clinical stool samples, 13% of high-touch hospital environment surfaces, and 26% of community shoe sole samples. Among these positive C. difficile samples, toxigenic strains were detected in 93% of patient samples, 66% of hospital environment swabs and 64% of shoe samples. Importantly, the most predominant toxigenic strains appeared in all three sample types.
“When we collected some hospital environmental samples, we saw the isolate ribotypes perfectly matched the patient samples,” Dr. Alam said.
Further, “we saw the exact same ribotypes on our shoe bottoms, from these community, nonclinical sources,” Dr. Alam said. “Apparently, it seems, these dangerous pathogens are everywhere.”
In fact, “we may have brought many different strains from all over the world here to this meeting,” he added. “When we are taking antibiotics, we are susceptible to these different strains.”
Hospitals are cleaned daily, “but how many of us care about the shoes” on those who walk through the hospital, he asked. “We are loading the hospital with Clostridium difficile, and the hospital environment is also loaded with Clostridium difficile so we are bringing it into the community. We are spreading it everywhere.”
“Maybe we are blaming the doctors, nurses, and other staff, but [we are] not thinking about our shoes,” Dr. Alam added.
NEW ORLEANS – For an explanation to the spread of Clostridium difficile, one needs to look to our soles.
Based on ribosomal analysis, C. difficile often is transmitted from the hospital into the community and back into the hospital on the soles of shoes, researchers have concluded, based on findings from thousands of samples from patients, hospital environments, and shoes.
Dr. Alam of the University of Houston College of Pharmacy and his colleagues collected thousands of samples from the state of Texas to see how C. difficile strains in the community are related to clinical strains in the hospital.
The researchers collected 3,109 stool samples from people hospitalized with C. difficile, another 1,697 swabs taken from environmental surfaces in hospitals across the state, plus another 400 samples taken from the soles of shoes of clinicians and non–health care workers.
C. difficile was found in 44% of clinical stool samples, 13% of high-touch hospital environment surfaces, and 26% of community shoe sole samples. Among these positive C. difficile samples, toxigenic strains were detected in 93% of patient samples, 66% of hospital environment swabs and 64% of shoe samples. Importantly, the most predominant toxigenic strains appeared in all three sample types.
“When we collected some hospital environmental samples, we saw the isolate ribotypes perfectly matched the patient samples,” Dr. Alam said.
Further, “we saw the exact same ribotypes on our shoe bottoms, from these community, nonclinical sources,” Dr. Alam said. “Apparently, it seems, these dangerous pathogens are everywhere.”
In fact, “we may have brought many different strains from all over the world here to this meeting,” he added. “When we are taking antibiotics, we are susceptible to these different strains.”
Hospitals are cleaned daily, “but how many of us care about the shoes” on those who walk through the hospital, he asked. “We are loading the hospital with Clostridium difficile, and the hospital environment is also loaded with Clostridium difficile so we are bringing it into the community. We are spreading it everywhere.”
“Maybe we are blaming the doctors, nurses, and other staff, but [we are] not thinking about our shoes,” Dr. Alam added.
AT ASM MICROBE 2017
Key clinical point: Clostridium difficile transfer between the hospital and community and back could be driven in part by contaminated shoes.
Major finding: Toxigenic strains of C. difficile were found in 93% of patient samples, 66% of hospital environment swabs, and 64% of shoe samples.
Data source: Study of 3,109 stool samples from infected hospitalized patients, 1,697 hospital environmental surface swabs, and 400 samples from the soles of shoes.
Disclosures: Dr. Alam reported having no financial disclosures.
Ceftaroline shortens duration of MRSA bacteremia
NEW ORLEANS – Ceftaroline fosamil reduced the median duration of methicillin-resistant Staphylococcus aureus (MRSA) bacteremia by 2 days in Veterans Administration patients, a retrospective study showed.
Investigators identified 219 patients with MRSA within the Veterans Affairs (VA) medical system nationwide from 2011 to 2015. All patients received at least 48 hours of ceftaroline fosamil (Teflaro) therapy to treat MRSA bacteremia. “We know it has good activity against MRSA in vitro. We use it in bacteremia, but we don’t have a lot of clinical data to support or refute its use,” said Nicholas S. Britt, PharmD, a PGY2 infectious diseases resident at Barnes-Jewish Hospital in St. Louis.
“Ceftaroline was primarily used as second-line or salvage therapy … which is basically what we expected, based on how it’s used in clinical practice,” Dr. Britt said.
Treatment failures
A total of 88 of the 219 (40%) patients experienced treatment failure. This rate “seems kind of high, but, if you look at some of the other MRSA agents for bacteremia (vancomycin, for example), it usually has a treatment failure rate around 60%,” Dr. Britt said. “The outcomes were not as poor as I would expect with [patients] using it for second- and third-line therapy.”
Hospital-acquired infection (odds ratio, 2.11; P = .013), ICU admission (OR, 3.95; P less than .001) and infective endocarditis (OR, 4.77; P = .002) were significantly associated with treatment failure in a univariate analysis. “Admissions to the ICU and endocarditis were the big ones, factors you would associate with failure for most antibiotics,” Dr. Britt said. In a multivariate analysis, only ICU admission remained significantly associated with treatment failure (adjusted OR, 2.24; P = .028).
The investigators also looked at treatment failure with ceftaroline monotherapy, compared with its use in combination. There is in vitro data showing synergy when you add ceftaroline to daptomycin, vancomycin, or some of these other agents,” Dr. Britt said. However, he added, “We didn’t find any significant difference in outcomes when you added another agent.” Treatment failure with monotherapy was 35%, versus 46%, with combination treatment (P = .107).
“This could be because the sicker patients are the ones getting combination therapy.”
No observed differences by dosing
Dr. Britt and his colleagues also looked for any differences by dosing interval, “which hasn’t been evaluated extensively.”
The Food and Drug Administration labeled it for use every 12 hours, but treatment of MRSA bacteremia is an off-label use, Dr. Britt explained. Dosing every 8 hours instead improves the achievement of pharmacokinetic and pharmacodynamic parameters in in vitro studies. “Clinically, we’re almost always using it q8. They’re sick patients, so you don’t want to under-dose them. And ceftaroline is pretty well tolerated overall.”
“But, we didn’t really see any difference between the q8 and the q12” in terms of treatment failure. The rates were 36% and 42%, respectively, and not significantly different (P = .440). “Granted, patients who are sicker are probably going to get treated more aggressively,” Dr. Britt added.
The current research only focused on outcomes associated with ceftaroline. Going forward, Dr. Britt said, “We’re hoping to use this data to compare ceftaroline to other agents as well, probably as second-line therapy, since that’s how it’s used most often.”
Dr. Britt had no relevant financial disclosures.
NEW ORLEANS – Ceftaroline fosamil reduced the median duration of methicillin-resistant Staphylococcus aureus (MRSA) bacteremia by 2 days in Veterans Administration patients, a retrospective study showed.
Investigators identified 219 patients with MRSA within the Veterans Affairs (VA) medical system nationwide from 2011 to 2015. All patients received at least 48 hours of ceftaroline fosamil (Teflaro) therapy to treat MRSA bacteremia. “We know it has good activity against MRSA in vitro. We use it in bacteremia, but we don’t have a lot of clinical data to support or refute its use,” said Nicholas S. Britt, PharmD, a PGY2 infectious diseases resident at Barnes-Jewish Hospital in St. Louis.
“Ceftaroline was primarily used as second-line or salvage therapy … which is basically what we expected, based on how it’s used in clinical practice,” Dr. Britt said.
Treatment failures
A total of 88 of the 219 (40%) patients experienced treatment failure. This rate “seems kind of high, but, if you look at some of the other MRSA agents for bacteremia (vancomycin, for example), it usually has a treatment failure rate around 60%,” Dr. Britt said. “The outcomes were not as poor as I would expect with [patients] using it for second- and third-line therapy.”
Hospital-acquired infection (odds ratio, 2.11; P = .013), ICU admission (OR, 3.95; P less than .001) and infective endocarditis (OR, 4.77; P = .002) were significantly associated with treatment failure in a univariate analysis. “Admissions to the ICU and endocarditis were the big ones, factors you would associate with failure for most antibiotics,” Dr. Britt said. In a multivariate analysis, only ICU admission remained significantly associated with treatment failure (adjusted OR, 2.24; P = .028).
The investigators also looked at treatment failure with ceftaroline monotherapy, compared with its use in combination. There is in vitro data showing synergy when you add ceftaroline to daptomycin, vancomycin, or some of these other agents,” Dr. Britt said. However, he added, “We didn’t find any significant difference in outcomes when you added another agent.” Treatment failure with monotherapy was 35%, versus 46%, with combination treatment (P = .107).
“This could be because the sicker patients are the ones getting combination therapy.”
No observed differences by dosing
Dr. Britt and his colleagues also looked for any differences by dosing interval, “which hasn’t been evaluated extensively.”
The Food and Drug Administration labeled it for use every 12 hours, but treatment of MRSA bacteremia is an off-label use, Dr. Britt explained. Dosing every 8 hours instead improves the achievement of pharmacokinetic and pharmacodynamic parameters in in vitro studies. “Clinically, we’re almost always using it q8. They’re sick patients, so you don’t want to under-dose them. And ceftaroline is pretty well tolerated overall.”
“But, we didn’t really see any difference between the q8 and the q12” in terms of treatment failure. The rates were 36% and 42%, respectively, and not significantly different (P = .440). “Granted, patients who are sicker are probably going to get treated more aggressively,” Dr. Britt added.
The current research only focused on outcomes associated with ceftaroline. Going forward, Dr. Britt said, “We’re hoping to use this data to compare ceftaroline to other agents as well, probably as second-line therapy, since that’s how it’s used most often.”
Dr. Britt had no relevant financial disclosures.
NEW ORLEANS – Ceftaroline fosamil reduced the median duration of methicillin-resistant Staphylococcus aureus (MRSA) bacteremia by 2 days in Veterans Administration patients, a retrospective study showed.
Investigators identified 219 patients with MRSA within the Veterans Affairs (VA) medical system nationwide from 2011 to 2015. All patients received at least 48 hours of ceftaroline fosamil (Teflaro) therapy to treat MRSA bacteremia. “We know it has good activity against MRSA in vitro. We use it in bacteremia, but we don’t have a lot of clinical data to support or refute its use,” said Nicholas S. Britt, PharmD, a PGY2 infectious diseases resident at Barnes-Jewish Hospital in St. Louis.
“Ceftaroline was primarily used as second-line or salvage therapy … which is basically what we expected, based on how it’s used in clinical practice,” Dr. Britt said.
Treatment failures
A total of 88 of the 219 (40%) patients experienced treatment failure. This rate “seems kind of high, but, if you look at some of the other MRSA agents for bacteremia (vancomycin, for example), it usually has a treatment failure rate around 60%,” Dr. Britt said. “The outcomes were not as poor as I would expect with [patients] using it for second- and third-line therapy.”
Hospital-acquired infection (odds ratio, 2.11; P = .013), ICU admission (OR, 3.95; P less than .001) and infective endocarditis (OR, 4.77; P = .002) were significantly associated with treatment failure in a univariate analysis. “Admissions to the ICU and endocarditis were the big ones, factors you would associate with failure for most antibiotics,” Dr. Britt said. In a multivariate analysis, only ICU admission remained significantly associated with treatment failure (adjusted OR, 2.24; P = .028).
The investigators also looked at treatment failure with ceftaroline monotherapy, compared with its use in combination. There is in vitro data showing synergy when you add ceftaroline to daptomycin, vancomycin, or some of these other agents,” Dr. Britt said. However, he added, “We didn’t find any significant difference in outcomes when you added another agent.” Treatment failure with monotherapy was 35%, versus 46%, with combination treatment (P = .107).
“This could be because the sicker patients are the ones getting combination therapy.”
No observed differences by dosing
Dr. Britt and his colleagues also looked for any differences by dosing interval, “which hasn’t been evaluated extensively.”
The Food and Drug Administration labeled it for use every 12 hours, but treatment of MRSA bacteremia is an off-label use, Dr. Britt explained. Dosing every 8 hours instead improves the achievement of pharmacokinetic and pharmacodynamic parameters in in vitro studies. “Clinically, we’re almost always using it q8. They’re sick patients, so you don’t want to under-dose them. And ceftaroline is pretty well tolerated overall.”
“But, we didn’t really see any difference between the q8 and the q12” in terms of treatment failure. The rates were 36% and 42%, respectively, and not significantly different (P = .440). “Granted, patients who are sicker are probably going to get treated more aggressively,” Dr. Britt added.
The current research only focused on outcomes associated with ceftaroline. Going forward, Dr. Britt said, “We’re hoping to use this data to compare ceftaroline to other agents as well, probably as second-line therapy, since that’s how it’s used most often.”
Dr. Britt had no relevant financial disclosures.
AT ASM MICROBE 2017
Key clinical point:
Major finding: Median duration of MRSA bacteremia dropped from 2.79 days before to 1.18 days after initiation of ceftaroline (P less than .001).
Data source: A retrospective study of 219 hospitalized VA patients initiating ceftaroline for MRSA bacteremia.
Disclosures: Dr. Britt had no relevant financial disclosures.
Carbapenem-resistant sepsis risk factors vary significantly
NEW ORLEANS – Investigators discovered significant differences in risk factors when comparing 603 people hospitalized with carbapenem-resistant Gram-negative sepsis with either Enterobacteriaceae-caused or non-Enterobacteriaceae–caused infection.
“We know some of the virulence factors and the resistance mechanisms can differ between those two groups. We really wanted to see if that would influence outcomes,” said Nicholas S. Britt, PharmD, a PGY2 infectious disease resident at Barnes-Jewish Hospital in St. Louis. Mortality rates, however, did not differ significantly.
“Patients who had Enterobacteriaceae infections were more likely to have urinary tract infections, to be older patients, and to have higher APACHE (Acute Physiologic Assessment and Chronic Health Evaluation) II scores,” Dr. Britt said at the annual meeting of the American Society for Microbiology. These differences all were statistically significant, compared with those of the CRNE group (P less than .05).
In contrast, the non-Enterobacteriaceae patients tended to have more respiratory infections and more frequent central venous catheter use. This group also was more likely to have a history of carbapenem use and more frequent antimicrobial exposures overall and to present after solid organ transplantation. “The cystic fibrosis patients were more likely to get non-Enterobacteriaceae infections as well,” Dr. Britt added. These differences also were statistically significant (all P less than .05).
“I think the biggest takeaway from this study, honestly, is the number of patients infected with CRE, versus CRNE,” Dr. Britt said. “We know CRE are a serious public health threat, one of the biggest threats out there, but, if you look at the burden on carbapenem-resistant disease, it’s primarily the non-Enterobacteriaceae.”
In fact, more than three-quarters of the patient studied (78%) had CRNE infections, and Pseudomonas was a major driver, he added. “Carbapenem resistance in this group of patients is something we should be focusing on – not only the CRE – because we’re seeing more of the non-CRE clinically.”
Patient age, presence of bloodstream infection, and use of mechanical ventilation, vasopressors, and immunosuppression was associated with hospital mortality in the study. After adjusting for potential confounders, however, CRNE infection was not associated with increased hospital mortality, compared with CRE cases (adjusted odds ratio, 0.97; P = .917).
“Our mortality rate was 16%, which is comparable to [that of] other studies,” Dr. Britt said. “There doesn’t seem to be any difference in this outcome between the two groups.” Mortality was 16.4% in the CRE cohort, versus 16.5% in the CRNE cohort (P = 0.965).
NEW ORLEANS – Investigators discovered significant differences in risk factors when comparing 603 people hospitalized with carbapenem-resistant Gram-negative sepsis with either Enterobacteriaceae-caused or non-Enterobacteriaceae–caused infection.
“We know some of the virulence factors and the resistance mechanisms can differ between those two groups. We really wanted to see if that would influence outcomes,” said Nicholas S. Britt, PharmD, a PGY2 infectious disease resident at Barnes-Jewish Hospital in St. Louis. Mortality rates, however, did not differ significantly.
“Patients who had Enterobacteriaceae infections were more likely to have urinary tract infections, to be older patients, and to have higher APACHE (Acute Physiologic Assessment and Chronic Health Evaluation) II scores,” Dr. Britt said at the annual meeting of the American Society for Microbiology. These differences all were statistically significant, compared with those of the CRNE group (P less than .05).
In contrast, the non-Enterobacteriaceae patients tended to have more respiratory infections and more frequent central venous catheter use. This group also was more likely to have a history of carbapenem use and more frequent antimicrobial exposures overall and to present after solid organ transplantation. “The cystic fibrosis patients were more likely to get non-Enterobacteriaceae infections as well,” Dr. Britt added. These differences also were statistically significant (all P less than .05).
“I think the biggest takeaway from this study, honestly, is the number of patients infected with CRE, versus CRNE,” Dr. Britt said. “We know CRE are a serious public health threat, one of the biggest threats out there, but, if you look at the burden on carbapenem-resistant disease, it’s primarily the non-Enterobacteriaceae.”
In fact, more than three-quarters of the patient studied (78%) had CRNE infections, and Pseudomonas was a major driver, he added. “Carbapenem resistance in this group of patients is something we should be focusing on – not only the CRE – because we’re seeing more of the non-CRE clinically.”
Patient age, presence of bloodstream infection, and use of mechanical ventilation, vasopressors, and immunosuppression was associated with hospital mortality in the study. After adjusting for potential confounders, however, CRNE infection was not associated with increased hospital mortality, compared with CRE cases (adjusted odds ratio, 0.97; P = .917).
“Our mortality rate was 16%, which is comparable to [that of] other studies,” Dr. Britt said. “There doesn’t seem to be any difference in this outcome between the two groups.” Mortality was 16.4% in the CRE cohort, versus 16.5% in the CRNE cohort (P = 0.965).
NEW ORLEANS – Investigators discovered significant differences in risk factors when comparing 603 people hospitalized with carbapenem-resistant Gram-negative sepsis with either Enterobacteriaceae-caused or non-Enterobacteriaceae–caused infection.
“We know some of the virulence factors and the resistance mechanisms can differ between those two groups. We really wanted to see if that would influence outcomes,” said Nicholas S. Britt, PharmD, a PGY2 infectious disease resident at Barnes-Jewish Hospital in St. Louis. Mortality rates, however, did not differ significantly.
“Patients who had Enterobacteriaceae infections were more likely to have urinary tract infections, to be older patients, and to have higher APACHE (Acute Physiologic Assessment and Chronic Health Evaluation) II scores,” Dr. Britt said at the annual meeting of the American Society for Microbiology. These differences all were statistically significant, compared with those of the CRNE group (P less than .05).
In contrast, the non-Enterobacteriaceae patients tended to have more respiratory infections and more frequent central venous catheter use. This group also was more likely to have a history of carbapenem use and more frequent antimicrobial exposures overall and to present after solid organ transplantation. “The cystic fibrosis patients were more likely to get non-Enterobacteriaceae infections as well,” Dr. Britt added. These differences also were statistically significant (all P less than .05).
“I think the biggest takeaway from this study, honestly, is the number of patients infected with CRE, versus CRNE,” Dr. Britt said. “We know CRE are a serious public health threat, one of the biggest threats out there, but, if you look at the burden on carbapenem-resistant disease, it’s primarily the non-Enterobacteriaceae.”
In fact, more than three-quarters of the patient studied (78%) had CRNE infections, and Pseudomonas was a major driver, he added. “Carbapenem resistance in this group of patients is something we should be focusing on – not only the CRE – because we’re seeing more of the non-CRE clinically.”
Patient age, presence of bloodstream infection, and use of mechanical ventilation, vasopressors, and immunosuppression was associated with hospital mortality in the study. After adjusting for potential confounders, however, CRNE infection was not associated with increased hospital mortality, compared with CRE cases (adjusted odds ratio, 0.97; P = .917).
“Our mortality rate was 16%, which is comparable to [that of] other studies,” Dr. Britt said. “There doesn’t seem to be any difference in this outcome between the two groups.” Mortality was 16.4% in the CRE cohort, versus 16.5% in the CRNE cohort (P = 0.965).
AT ASM MICROBE 2017
Key clinical point:
Major finding: Hospital mortality was 16% overall and did not differ significantly between Enterobacteriaceae and non-Enterobacteriaceae infections (16.5% vs. 16.4%; P = 0.965).
Data source: A retrospective cohort study of 603 patients with Gram-negative sepsis at a tertiary academic medical center from 2012 to 2016.
Disclosures: Dr. Britt did not have any relevant financial disclosures.
Hospital antibiograms may not apply to emergency department patients
NEW ORLEANS – Patients seen in the emergency department present with a wider range of risk for resistant infections than do hospitalized patients overall, and as a result, hospital-wide antibiograms that dictate empiric therapy may not translate well to the ED setting, a study showed.
Instead, investigators suggested that clinicians choose empiric antimicrobial therapy in the emergency department based on factors associated with differences in susceptibility.
The study researchers compared common antibiotics to treat Escherichia coli between adults treated at their institution overall versus the emergency department, and by sex, patient age, and whether people came to the ED from home versus a long-term care setting. They found some significant differences that could guide empiric treatment in the ED setting.
“Our ED pharmacist observed a lot of broad-spectrum antibiotic prescribing for UTIs,” lead investigator Sarah Jorgensen, PharmD, said at the annual meeting of the American Society for Microbiology. “Also, we’ve had a culture follow-up program in place for the last 5 years, and they had to intervene on a lot of postdischarge antibiotic mismatches.”
E. coli was the most common urinary pathogen detected in this study of 500 randomly selected ED patients with ICD-9/ICD-10 diagnostic codes for urinary tract infection. Investigators found E. coli in 64% of the 226 culture-positive patients presenting to the ED at Huntington Hospital in Pasadena, Calif., between July 2015 and June 2016.
“What was surprising for us, because our enrollment was based on ICD codes for a UTI, is that only about 50% had a positive urine culture,” said Dr. Jorgensen, a pharmacy resident at the University of Southern California in Los Angeles. “Urinalysis was positive in about 99% of the population.”
Dr. Jorgensen and her colleagues found overall low susceptibilities of 71% for ciprofloxacin, 66% for trimethoprim/sulfamethoxazole, and 67% for cefazolin susceptibilities in the ED. They also found that 8% of isolates were positive for extended-spectrum beta-lactamase isolates, as were 1% of E. coli isolates.
The 67% cefazolin susceptibility was significantly lower in the ED, compared with the 86% susceptibility in the institutional antibiogram (P less than .001).
The investigators found E. coli susceptibility to ciprofloxacin was lower in men, at 55%, compared with 74% among women, but the difference was not statistically significant (P = .14). A similar pattern emerged with cefazolin – 55% susceptibility among men and 69% among women (P = .26). In contrast, trimethoprim/sulfamethoxazole susceptibility trended higher in men, at 73%, vs. 64% among women in the ED (P = .63).
When they divided patients by age 50 years and younger versus those older than 50, the investigators found ampicillin susceptibilities were lower in the younger group, at 30%, compared with 51% among those in the older cohort (P = .03). Similarly, gentamicin susceptibilities were 80% in the younger group, compared with 92% in the older group (P = .04).
Ciprofloxacin susceptibility was significantly lower among people coming to the ED from a long-term care facility than among those coming from home – 35% vs. 77% (P less than .001). Differences in ciprofloxacin susceptibility between admitted and discharged patients was less striking – 63% vs. 78% (P = .04).
Nitrofurantoin was the only oral agent with susceptibility greater than 80% in all patient groups, with susceptibility ranging from 88% to 100%.
Because it typically takes 2-3 days to get the susceptibility results back at Huntington Hospital, many patients are discharged on empiric therapy, noted Mira Zurayk, PharmD, a resident at Huntington Hospital. That can present multiple challenges, particularly with homeless patients who are difficult to find and provide follow-up for, Dr. Jorgensen added.
Based partly on the study findings, the investigators developed a clinical algorithm specifically to address UTI antimicrobial prescriptions in the ED. The algorithm incorporates different recommendations for different groups of patients because of their different resistance trends.
“I think that is a good way to tailor empiric therapy when you don’t have culture results up front,” Dr. Jorgensen said. “We just implemented the algorithm, and I’m now analyzing the outcomes.”
Having more data on outcomes will help the clinicians target lowering the rate of “drug-bug mismatches,” as well as UTI-related revisits to the ED. In addition, the work could help expand the antibiotic stewardship program in the hospital to the ED for the first time, Dr. Jorgensen said.
Dr. Jorgensen and Dr. Zurayk had no relevant financial disclosures. One of the study coauthors, Annie Wong-Beringer, PharmD, receives grant funding from Merck.
NEW ORLEANS – Patients seen in the emergency department present with a wider range of risk for resistant infections than do hospitalized patients overall, and as a result, hospital-wide antibiograms that dictate empiric therapy may not translate well to the ED setting, a study showed.
Instead, investigators suggested that clinicians choose empiric antimicrobial therapy in the emergency department based on factors associated with differences in susceptibility.
The study researchers compared common antibiotics to treat Escherichia coli between adults treated at their institution overall versus the emergency department, and by sex, patient age, and whether people came to the ED from home versus a long-term care setting. They found some significant differences that could guide empiric treatment in the ED setting.
“Our ED pharmacist observed a lot of broad-spectrum antibiotic prescribing for UTIs,” lead investigator Sarah Jorgensen, PharmD, said at the annual meeting of the American Society for Microbiology. “Also, we’ve had a culture follow-up program in place for the last 5 years, and they had to intervene on a lot of postdischarge antibiotic mismatches.”
E. coli was the most common urinary pathogen detected in this study of 500 randomly selected ED patients with ICD-9/ICD-10 diagnostic codes for urinary tract infection. Investigators found E. coli in 64% of the 226 culture-positive patients presenting to the ED at Huntington Hospital in Pasadena, Calif., between July 2015 and June 2016.
“What was surprising for us, because our enrollment was based on ICD codes for a UTI, is that only about 50% had a positive urine culture,” said Dr. Jorgensen, a pharmacy resident at the University of Southern California in Los Angeles. “Urinalysis was positive in about 99% of the population.”
Dr. Jorgensen and her colleagues found overall low susceptibilities of 71% for ciprofloxacin, 66% for trimethoprim/sulfamethoxazole, and 67% for cefazolin susceptibilities in the ED. They also found that 8% of isolates were positive for extended-spectrum beta-lactamase isolates, as were 1% of E. coli isolates.
The 67% cefazolin susceptibility was significantly lower in the ED, compared with the 86% susceptibility in the institutional antibiogram (P less than .001).
The investigators found E. coli susceptibility to ciprofloxacin was lower in men, at 55%, compared with 74% among women, but the difference was not statistically significant (P = .14). A similar pattern emerged with cefazolin – 55% susceptibility among men and 69% among women (P = .26). In contrast, trimethoprim/sulfamethoxazole susceptibility trended higher in men, at 73%, vs. 64% among women in the ED (P = .63).
When they divided patients by age 50 years and younger versus those older than 50, the investigators found ampicillin susceptibilities were lower in the younger group, at 30%, compared with 51% among those in the older cohort (P = .03). Similarly, gentamicin susceptibilities were 80% in the younger group, compared with 92% in the older group (P = .04).
Ciprofloxacin susceptibility was significantly lower among people coming to the ED from a long-term care facility than among those coming from home – 35% vs. 77% (P less than .001). Differences in ciprofloxacin susceptibility between admitted and discharged patients was less striking – 63% vs. 78% (P = .04).
Nitrofurantoin was the only oral agent with susceptibility greater than 80% in all patient groups, with susceptibility ranging from 88% to 100%.
Because it typically takes 2-3 days to get the susceptibility results back at Huntington Hospital, many patients are discharged on empiric therapy, noted Mira Zurayk, PharmD, a resident at Huntington Hospital. That can present multiple challenges, particularly with homeless patients who are difficult to find and provide follow-up for, Dr. Jorgensen added.
Based partly on the study findings, the investigators developed a clinical algorithm specifically to address UTI antimicrobial prescriptions in the ED. The algorithm incorporates different recommendations for different groups of patients because of their different resistance trends.
“I think that is a good way to tailor empiric therapy when you don’t have culture results up front,” Dr. Jorgensen said. “We just implemented the algorithm, and I’m now analyzing the outcomes.”
Having more data on outcomes will help the clinicians target lowering the rate of “drug-bug mismatches,” as well as UTI-related revisits to the ED. In addition, the work could help expand the antibiotic stewardship program in the hospital to the ED for the first time, Dr. Jorgensen said.
Dr. Jorgensen and Dr. Zurayk had no relevant financial disclosures. One of the study coauthors, Annie Wong-Beringer, PharmD, receives grant funding from Merck.
NEW ORLEANS – Patients seen in the emergency department present with a wider range of risk for resistant infections than do hospitalized patients overall, and as a result, hospital-wide antibiograms that dictate empiric therapy may not translate well to the ED setting, a study showed.
Instead, investigators suggested that clinicians choose empiric antimicrobial therapy in the emergency department based on factors associated with differences in susceptibility.
The study researchers compared common antibiotics to treat Escherichia coli between adults treated at their institution overall versus the emergency department, and by sex, patient age, and whether people came to the ED from home versus a long-term care setting. They found some significant differences that could guide empiric treatment in the ED setting.
“Our ED pharmacist observed a lot of broad-spectrum antibiotic prescribing for UTIs,” lead investigator Sarah Jorgensen, PharmD, said at the annual meeting of the American Society for Microbiology. “Also, we’ve had a culture follow-up program in place for the last 5 years, and they had to intervene on a lot of postdischarge antibiotic mismatches.”
E. coli was the most common urinary pathogen detected in this study of 500 randomly selected ED patients with ICD-9/ICD-10 diagnostic codes for urinary tract infection. Investigators found E. coli in 64% of the 226 culture-positive patients presenting to the ED at Huntington Hospital in Pasadena, Calif., between July 2015 and June 2016.
“What was surprising for us, because our enrollment was based on ICD codes for a UTI, is that only about 50% had a positive urine culture,” said Dr. Jorgensen, a pharmacy resident at the University of Southern California in Los Angeles. “Urinalysis was positive in about 99% of the population.”
Dr. Jorgensen and her colleagues found overall low susceptibilities of 71% for ciprofloxacin, 66% for trimethoprim/sulfamethoxazole, and 67% for cefazolin susceptibilities in the ED. They also found that 8% of isolates were positive for extended-spectrum beta-lactamase isolates, as were 1% of E. coli isolates.
The 67% cefazolin susceptibility was significantly lower in the ED, compared with the 86% susceptibility in the institutional antibiogram (P less than .001).
The investigators found E. coli susceptibility to ciprofloxacin was lower in men, at 55%, compared with 74% among women, but the difference was not statistically significant (P = .14). A similar pattern emerged with cefazolin – 55% susceptibility among men and 69% among women (P = .26). In contrast, trimethoprim/sulfamethoxazole susceptibility trended higher in men, at 73%, vs. 64% among women in the ED (P = .63).
When they divided patients by age 50 years and younger versus those older than 50, the investigators found ampicillin susceptibilities were lower in the younger group, at 30%, compared with 51% among those in the older cohort (P = .03). Similarly, gentamicin susceptibilities were 80% in the younger group, compared with 92% in the older group (P = .04).
Ciprofloxacin susceptibility was significantly lower among people coming to the ED from a long-term care facility than among those coming from home – 35% vs. 77% (P less than .001). Differences in ciprofloxacin susceptibility between admitted and discharged patients was less striking – 63% vs. 78% (P = .04).
Nitrofurantoin was the only oral agent with susceptibility greater than 80% in all patient groups, with susceptibility ranging from 88% to 100%.
Because it typically takes 2-3 days to get the susceptibility results back at Huntington Hospital, many patients are discharged on empiric therapy, noted Mira Zurayk, PharmD, a resident at Huntington Hospital. That can present multiple challenges, particularly with homeless patients who are difficult to find and provide follow-up for, Dr. Jorgensen added.
Based partly on the study findings, the investigators developed a clinical algorithm specifically to address UTI antimicrobial prescriptions in the ED. The algorithm incorporates different recommendations for different groups of patients because of their different resistance trends.
“I think that is a good way to tailor empiric therapy when you don’t have culture results up front,” Dr. Jorgensen said. “We just implemented the algorithm, and I’m now analyzing the outcomes.”
Having more data on outcomes will help the clinicians target lowering the rate of “drug-bug mismatches,” as well as UTI-related revisits to the ED. In addition, the work could help expand the antibiotic stewardship program in the hospital to the ED for the first time, Dr. Jorgensen said.
Dr. Jorgensen and Dr. Zurayk had no relevant financial disclosures. One of the study coauthors, Annie Wong-Beringer, PharmD, receives grant funding from Merck.
AT ASM MICROBE 2017
Key clinical point: Escherichia coli susceptibility to common antibiotics in the emergency department varies by age, sex, and admitted versus discharge status.
Major finding: The only oral antimicrobial with greater than 80% susceptibility across all groups was nitrofurantoin.
Data source: A study of 500 randomly selected adults who presented to the ED with a urinary tract infection, based on ICD-9 or ICD-10 codes.
Disclosures: Dr. Jorgensen and Dr. Zurayk had no relevant financial disclosures. One study coauthor, Annie Wong-Beringer, PharmD, receives grant funding from Merck.