User login
News and Views that Matter to Pediatricians
The leading independent newspaper covering news and commentary in pediatrics.
Whooping Cough Likely on Pace for a 5-Year High
Like many diseases, whooping cough reached record low levels during the early days of the COVID pandemic.
More than 10,000 cases of whooping cough have been reported in the United States so far this year, and weekly reports say cases have more than tripled 2023 levels as of June, according to the Centers for Disease Control and Prevention (CDC). In 2023, there were 2815 cases reported during the entire year.
“The number of reported cases this year is close to what was seen at the same time in 2019, prior to the pandemic,” the CDC reported. There were 18,617 cases of whooping cough in 2019.
There were 259 cases reported nationwide for the week ending Aug. 3, with nearly half occurring in the mid-Atlantic region. Public health officials believe the resurgence of whooping cough is likely due to declining vaccination rates, mainly due to the missed vaccines during the height of the COVID pandemic. The diphtheria, tetanus, and pertussis vaccines (DTaP) have been given together since the 1940s, typically during infancy and again during early childhood. In 1941, there were more than 220,000 cases of whooping cough.
Whooping cough is caused by the bacteria Bordetella pertussis. The bacteria attach to tiny, hair-like extensions in the upper respiratory system called cilia, and toxins released by them damage the cilia and cause airways to swell. Early symptoms are similar to the common cold, but the condition eventually leads to coughing fits and a high-pitched “whoop” sound made when inhaling after a fit subsides. Coughing fits can be so severe that people can fracture a rib.
Vaccinated people may get a less severe illness, compared to unvaccinated people, the CDC says. Babies and children are particularly at risk for severe and even potentially deadly complications. About one in three babies under age 1 who get whooping cough will need to be hospitalized, and among those hospitalized babies, 1 in 100 die from complications.
A version of this article appeared on WebMD.com.
Like many diseases, whooping cough reached record low levels during the early days of the COVID pandemic.
More than 10,000 cases of whooping cough have been reported in the United States so far this year, and weekly reports say cases have more than tripled 2023 levels as of June, according to the Centers for Disease Control and Prevention (CDC). In 2023, there were 2815 cases reported during the entire year.
“The number of reported cases this year is close to what was seen at the same time in 2019, prior to the pandemic,” the CDC reported. There were 18,617 cases of whooping cough in 2019.
There were 259 cases reported nationwide for the week ending Aug. 3, with nearly half occurring in the mid-Atlantic region. Public health officials believe the resurgence of whooping cough is likely due to declining vaccination rates, mainly due to the missed vaccines during the height of the COVID pandemic. The diphtheria, tetanus, and pertussis vaccines (DTaP) have been given together since the 1940s, typically during infancy and again during early childhood. In 1941, there were more than 220,000 cases of whooping cough.
Whooping cough is caused by the bacteria Bordetella pertussis. The bacteria attach to tiny, hair-like extensions in the upper respiratory system called cilia, and toxins released by them damage the cilia and cause airways to swell. Early symptoms are similar to the common cold, but the condition eventually leads to coughing fits and a high-pitched “whoop” sound made when inhaling after a fit subsides. Coughing fits can be so severe that people can fracture a rib.
Vaccinated people may get a less severe illness, compared to unvaccinated people, the CDC says. Babies and children are particularly at risk for severe and even potentially deadly complications. About one in three babies under age 1 who get whooping cough will need to be hospitalized, and among those hospitalized babies, 1 in 100 die from complications.
A version of this article appeared on WebMD.com.
Like many diseases, whooping cough reached record low levels during the early days of the COVID pandemic.
More than 10,000 cases of whooping cough have been reported in the United States so far this year, and weekly reports say cases have more than tripled 2023 levels as of June, according to the Centers for Disease Control and Prevention (CDC). In 2023, there were 2815 cases reported during the entire year.
“The number of reported cases this year is close to what was seen at the same time in 2019, prior to the pandemic,” the CDC reported. There were 18,617 cases of whooping cough in 2019.
There were 259 cases reported nationwide for the week ending Aug. 3, with nearly half occurring in the mid-Atlantic region. Public health officials believe the resurgence of whooping cough is likely due to declining vaccination rates, mainly due to the missed vaccines during the height of the COVID pandemic. The diphtheria, tetanus, and pertussis vaccines (DTaP) have been given together since the 1940s, typically during infancy and again during early childhood. In 1941, there were more than 220,000 cases of whooping cough.
Whooping cough is caused by the bacteria Bordetella pertussis. The bacteria attach to tiny, hair-like extensions in the upper respiratory system called cilia, and toxins released by them damage the cilia and cause airways to swell. Early symptoms are similar to the common cold, but the condition eventually leads to coughing fits and a high-pitched “whoop” sound made when inhaling after a fit subsides. Coughing fits can be so severe that people can fracture a rib.
Vaccinated people may get a less severe illness, compared to unvaccinated people, the CDC says. Babies and children are particularly at risk for severe and even potentially deadly complications. About one in three babies under age 1 who get whooping cough will need to be hospitalized, and among those hospitalized babies, 1 in 100 die from complications.
A version of this article appeared on WebMD.com.
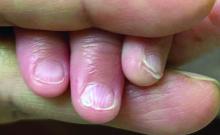
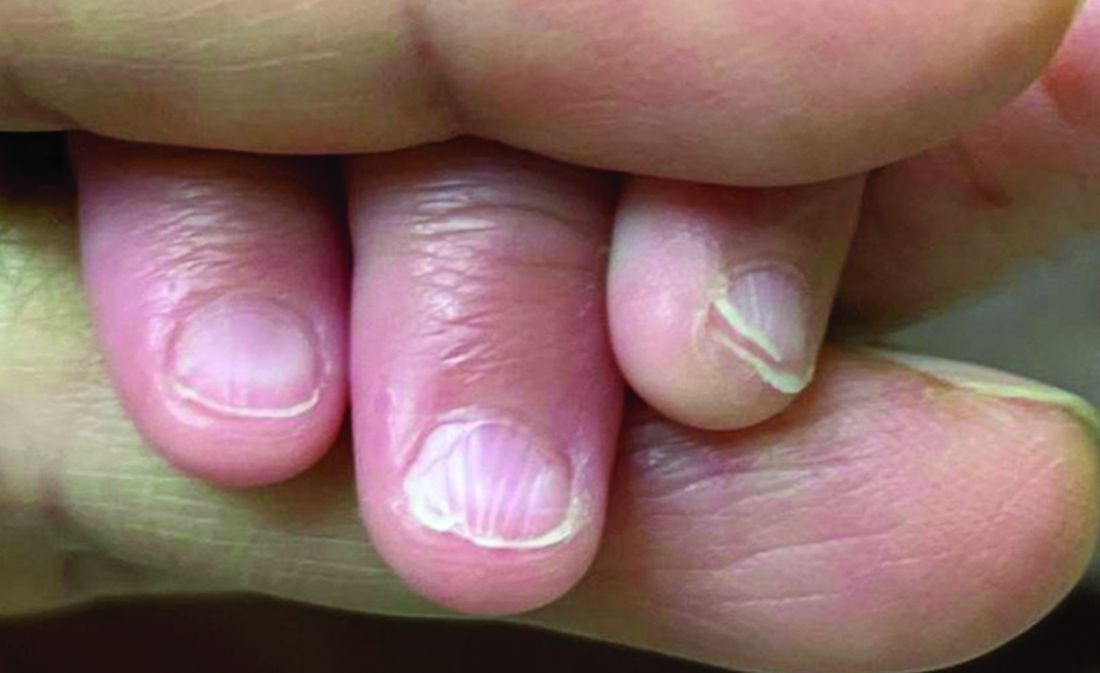
A 7-Month-Old Female Presented With Nail Changes
Given the clinical presentation and the absence of other systemic or dermatological findings, the diagnosis of chevron nails was made.
Discussion
The condition is characterized by transverse ridges on the nails that converge towards the center, forming a V or chevron shape. This condition was first described by Perry et al. and later by Shuster et al., who explained that the condition might result from axial growth of the nail with synchronous growth occurring from a chevron-shaped growing edge of the nail root. Alternatively, Shuster suggested that sequential growth, with localized variation in the nail production rate, could propagate a wave from the center of the nail to the edge.
The etiology of chevron nails is not well understood, but it is believed to result from temporary disruptions in the nail matrix, possibly related to minor illness or physiological stress during infancy.
In the case of our 7-month-old patient, the history of mild upper respiratory infections might have contributed to the development of chevron nails. However, the lack of other significant illness, skin involvement, or systemic findings supports the benign and self-limiting nature of this condition. Parents were reassured that chevron nails typically resolve on their own as the child grows and that no specific treatment is necessary.
Differential Diagnosis
The differential diagnosis of transverse nail changes in children includes other conditions such as trachyonychia, lichen planus, Darier disease, and pachyonychia congenita.
Trachyonychia, also known as “sandpaper nails,” trachyonychia is characterized by the roughening of the nail surface, giving it a dull and ridged appearance. The condition may affect all 20 nails and is often associated with underlying dermatological conditions such as lichen planus or alopecia areata. Unlike chevron nails, trachyonychia presents with more diffuse nail changes and does not typically feature the distinct V-shaped ridging seen in this patient.
Lichen planus is an inflammatory condition that can affect the skin, mucous membranes, and nails. Nail involvement in lichen planus can lead to longitudinal ridging, thinning, and sometimes even complete nail loss. The absence of other characteristic features of lichen planus, such as violaceous papules on the skin or white lacy patterns on mucous membranes (Wickham striae), makes this diagnosis less likely in our patient.
Darier disease, also known as keratosis follicularis, is a genetic disorder characterized by greasy, warty papules primarily on seborrheic areas of the skin, nail abnormalities, and sometimes mucosal involvement. Nail changes in Darier disease include longitudinal red and white streaks, V-shaped notching at the free edge of the nails, and subungual hyperkeratosis. These nail changes are more severe and distinct than the simple transverse ridging seen in chevron nails. The absence of other clinical signs of Darier disease, such as skin papules or characteristic nail notching, makes this diagnosis unlikely in our patient.
Pachyonychia congenita is a rare genetic disorder characterized by thickened nails (pachyonychia), painful plantar keratoderma, and sometimes oral leukokeratosis. The condition typically presents with significant nail thickening and other systemic findings, which were absent in our patient. The distinct pattern of V-shaped ridging observed in chevron nails does not align with the typical presentation of pachyonychia congenita.
Next Steps
No specific treatment is required for chevron nails. The condition is typically self-resolving, and the nails usually return to a normal appearance as the child continues to grow. Parents were advised to monitor the nails for any changes or new symptoms and were reassured about the benign nature of the findings. Follow-up was scheduled to ensure the resolution of the condition as the child develops.
Conclusion
Chevron nails are an important consideration in the differential diagnosis of transverse nail ridging in infants and young children. While the condition is benign and self-limiting, it is crucial to differentiate it from other nail dystrophies, such as trachyonychia, lichen planus, Darier disease, and pachyonychia congenita, which may require further investigation or intervention. Awareness of chevron nails can help prevent unnecessary worry and provide reassurance to parents and caregivers.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
Suggested Reading
Delano S, Belazarian L. Chevron nails: A normal variant in the pediatric population. Pediatr Dermatol. 2014 Jan-Feb;31(1):e24-5. doi: 10.1111/pde.12193.
John JM et al. Chevron nail — An under-recognised normal variant of nail development. Arch Dis Child. 2024 Jul 18;109(8):648. doi: 10.1136/archdischild-2024-326975.
Shuster S. The significance of chevron nails. Br J Dermatol. 1996;135:151–152. doi: 10.1046/j.1365-2133.1996.d01-961.x.
Starace M et al. Nail disorders in children. Skin Appendage Disord. 2018 Oct;4(4):217-229. doi: 10.1159/000486020.
Given the clinical presentation and the absence of other systemic or dermatological findings, the diagnosis of chevron nails was made.
Discussion
The condition is characterized by transverse ridges on the nails that converge towards the center, forming a V or chevron shape. This condition was first described by Perry et al. and later by Shuster et al., who explained that the condition might result from axial growth of the nail with synchronous growth occurring from a chevron-shaped growing edge of the nail root. Alternatively, Shuster suggested that sequential growth, with localized variation in the nail production rate, could propagate a wave from the center of the nail to the edge.
The etiology of chevron nails is not well understood, but it is believed to result from temporary disruptions in the nail matrix, possibly related to minor illness or physiological stress during infancy.
In the case of our 7-month-old patient, the history of mild upper respiratory infections might have contributed to the development of chevron nails. However, the lack of other significant illness, skin involvement, or systemic findings supports the benign and self-limiting nature of this condition. Parents were reassured that chevron nails typically resolve on their own as the child grows and that no specific treatment is necessary.
Differential Diagnosis
The differential diagnosis of transverse nail changes in children includes other conditions such as trachyonychia, lichen planus, Darier disease, and pachyonychia congenita.
Trachyonychia, also known as “sandpaper nails,” trachyonychia is characterized by the roughening of the nail surface, giving it a dull and ridged appearance. The condition may affect all 20 nails and is often associated with underlying dermatological conditions such as lichen planus or alopecia areata. Unlike chevron nails, trachyonychia presents with more diffuse nail changes and does not typically feature the distinct V-shaped ridging seen in this patient.
Lichen planus is an inflammatory condition that can affect the skin, mucous membranes, and nails. Nail involvement in lichen planus can lead to longitudinal ridging, thinning, and sometimes even complete nail loss. The absence of other characteristic features of lichen planus, such as violaceous papules on the skin or white lacy patterns on mucous membranes (Wickham striae), makes this diagnosis less likely in our patient.
Darier disease, also known as keratosis follicularis, is a genetic disorder characterized by greasy, warty papules primarily on seborrheic areas of the skin, nail abnormalities, and sometimes mucosal involvement. Nail changes in Darier disease include longitudinal red and white streaks, V-shaped notching at the free edge of the nails, and subungual hyperkeratosis. These nail changes are more severe and distinct than the simple transverse ridging seen in chevron nails. The absence of other clinical signs of Darier disease, such as skin papules or characteristic nail notching, makes this diagnosis unlikely in our patient.
Pachyonychia congenita is a rare genetic disorder characterized by thickened nails (pachyonychia), painful plantar keratoderma, and sometimes oral leukokeratosis. The condition typically presents with significant nail thickening and other systemic findings, which were absent in our patient. The distinct pattern of V-shaped ridging observed in chevron nails does not align with the typical presentation of pachyonychia congenita.
Next Steps
No specific treatment is required for chevron nails. The condition is typically self-resolving, and the nails usually return to a normal appearance as the child continues to grow. Parents were advised to monitor the nails for any changes or new symptoms and were reassured about the benign nature of the findings. Follow-up was scheduled to ensure the resolution of the condition as the child develops.
Conclusion
Chevron nails are an important consideration in the differential diagnosis of transverse nail ridging in infants and young children. While the condition is benign and self-limiting, it is crucial to differentiate it from other nail dystrophies, such as trachyonychia, lichen planus, Darier disease, and pachyonychia congenita, which may require further investigation or intervention. Awareness of chevron nails can help prevent unnecessary worry and provide reassurance to parents and caregivers.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
Suggested Reading
Delano S, Belazarian L. Chevron nails: A normal variant in the pediatric population. Pediatr Dermatol. 2014 Jan-Feb;31(1):e24-5. doi: 10.1111/pde.12193.
John JM et al. Chevron nail — An under-recognised normal variant of nail development. Arch Dis Child. 2024 Jul 18;109(8):648. doi: 10.1136/archdischild-2024-326975.
Shuster S. The significance of chevron nails. Br J Dermatol. 1996;135:151–152. doi: 10.1046/j.1365-2133.1996.d01-961.x.
Starace M et al. Nail disorders in children. Skin Appendage Disord. 2018 Oct;4(4):217-229. doi: 10.1159/000486020.
Given the clinical presentation and the absence of other systemic or dermatological findings, the diagnosis of chevron nails was made.
Discussion
The condition is characterized by transverse ridges on the nails that converge towards the center, forming a V or chevron shape. This condition was first described by Perry et al. and later by Shuster et al., who explained that the condition might result from axial growth of the nail with synchronous growth occurring from a chevron-shaped growing edge of the nail root. Alternatively, Shuster suggested that sequential growth, with localized variation in the nail production rate, could propagate a wave from the center of the nail to the edge.
The etiology of chevron nails is not well understood, but it is believed to result from temporary disruptions in the nail matrix, possibly related to minor illness or physiological stress during infancy.
In the case of our 7-month-old patient, the history of mild upper respiratory infections might have contributed to the development of chevron nails. However, the lack of other significant illness, skin involvement, or systemic findings supports the benign and self-limiting nature of this condition. Parents were reassured that chevron nails typically resolve on their own as the child grows and that no specific treatment is necessary.
Differential Diagnosis
The differential diagnosis of transverse nail changes in children includes other conditions such as trachyonychia, lichen planus, Darier disease, and pachyonychia congenita.
Trachyonychia, also known as “sandpaper nails,” trachyonychia is characterized by the roughening of the nail surface, giving it a dull and ridged appearance. The condition may affect all 20 nails and is often associated with underlying dermatological conditions such as lichen planus or alopecia areata. Unlike chevron nails, trachyonychia presents with more diffuse nail changes and does not typically feature the distinct V-shaped ridging seen in this patient.
Lichen planus is an inflammatory condition that can affect the skin, mucous membranes, and nails. Nail involvement in lichen planus can lead to longitudinal ridging, thinning, and sometimes even complete nail loss. The absence of other characteristic features of lichen planus, such as violaceous papules on the skin or white lacy patterns on mucous membranes (Wickham striae), makes this diagnosis less likely in our patient.
Darier disease, also known as keratosis follicularis, is a genetic disorder characterized by greasy, warty papules primarily on seborrheic areas of the skin, nail abnormalities, and sometimes mucosal involvement. Nail changes in Darier disease include longitudinal red and white streaks, V-shaped notching at the free edge of the nails, and subungual hyperkeratosis. These nail changes are more severe and distinct than the simple transverse ridging seen in chevron nails. The absence of other clinical signs of Darier disease, such as skin papules or characteristic nail notching, makes this diagnosis unlikely in our patient.
Pachyonychia congenita is a rare genetic disorder characterized by thickened nails (pachyonychia), painful plantar keratoderma, and sometimes oral leukokeratosis. The condition typically presents with significant nail thickening and other systemic findings, which were absent in our patient. The distinct pattern of V-shaped ridging observed in chevron nails does not align with the typical presentation of pachyonychia congenita.
Next Steps
No specific treatment is required for chevron nails. The condition is typically self-resolving, and the nails usually return to a normal appearance as the child continues to grow. Parents were advised to monitor the nails for any changes or new symptoms and were reassured about the benign nature of the findings. Follow-up was scheduled to ensure the resolution of the condition as the child develops.
Conclusion
Chevron nails are an important consideration in the differential diagnosis of transverse nail ridging in infants and young children. While the condition is benign and self-limiting, it is crucial to differentiate it from other nail dystrophies, such as trachyonychia, lichen planus, Darier disease, and pachyonychia congenita, which may require further investigation or intervention. Awareness of chevron nails can help prevent unnecessary worry and provide reassurance to parents and caregivers.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
Suggested Reading
Delano S, Belazarian L. Chevron nails: A normal variant in the pediatric population. Pediatr Dermatol. 2014 Jan-Feb;31(1):e24-5. doi: 10.1111/pde.12193.
John JM et al. Chevron nail — An under-recognised normal variant of nail development. Arch Dis Child. 2024 Jul 18;109(8):648. doi: 10.1136/archdischild-2024-326975.
Shuster S. The significance of chevron nails. Br J Dermatol. 1996;135:151–152. doi: 10.1046/j.1365-2133.1996.d01-961.x.
Starace M et al. Nail disorders in children. Skin Appendage Disord. 2018 Oct;4(4):217-229. doi: 10.1159/000486020.
There was no family history of similar nail findings and no relatives had a history of chronic skin conditions or congenital nail disorders.
On physical examination, several of the child’s fingernails exhibited distinct longitudinal ridges, with a characteristic pattern where the ridges converged at the center of the nail, forming a V-shape. There were no other concerning dermatologic findings, such as rashes, plaques, or erosions, and the skin and hair appeared otherwise normal. The rest of the physical exam was unremarkable.
FTC Interim Report on Pharmacy Middlemen Is First Step of Many Needed in Addressing Drug Costs, Access
Rising consolidation among pharmacy benefit managers (PBMs) allows the companies to profit at the expense of patients and independent pharmacists. That’s the conclusion of a recent Federal Trade Commission (FTC) report on interim findings from the agency’s ongoing investigation of PBMs.
Lawmakers are increasingly scrutinizing the industry amid growing concern among physicians and consumers about how PBMs exploit their market dominance. The top six PBMs managed 94% of US drug claims in 2023, with the majority handled by the industry’s three giants: CVS Caremark, Cigna’s Express Scripts, and United Healthcare’s OptumRx.
PBMs manage prescription drug benefits for health insurers, Medicare Part D drug plans, and large employers. They act as middlemen between health insurers and pharmacies, developing formularies of covered drugs and promising savings from the discounts and rebates they negotiate with drugmakers.
The FTC’s interim report found that the giant PBMs often exercise significant control over what drugs are available and at what price and which pharmacies patients can use to access their prescribed medications. Consumers suffer as a result, the report concluded.
Madelaine A. Feldman, MD, vice president for advocacy and government affairs for the Coalition of State Rheumatology Organizations, shared her perspective on the FTC report in an email Q&A with this news organization. She is affiliated with The Rheumatology Group, based in Metairie, Louisiana.
Dr. Feldman has long tracked the PBM industry and appeared as a witness before influential government panels, including the House Energy and Commerce Committee. She has highlighted for lawmakers the challenges physicians face in helping patients get needed medicines.
For example, she shared cases of PBMs steering patients toward the more expensive of three widely used rheumatoid arthritis medicines that have a similar mechanism of action, the Janus kinase (JAK) inhibitors, Dr. Feldman said.
One of the drugs cost roughly half of the other two — about $30,000 per year vs $65,000-$70,000. Yet only the two expensive drugs were included in the PBM formulary. As a result, the cheapest drug holds only a sliver of market share; the remainder is dominated by the two expensive products, she told the House Oversight and Accountability Committee in 2021.
This Q&A has been edited for length and clarity.
What would you want federal and state policymakers to do in response to the FTC’s report?
I think Congress needs to clearly delineate the differences between anticompetitive pharmacy issues, drug pricing issues, and their effect on formulary construction issues.
Lawmakers should demand more transparency and consider legislation that would remove perverse incentives that prompt PBMs to choose higher priced drugs for their formularies.
That may require other regulatory or legislative actions to ensure lower prices (not higher kickbacks) are incentivized. Ultimately, in order to gain true competition within the health insurance business, these oligopolies of multiple businesses need to be broken up. Anything less seems to be nibbling around the edges and allows the Big Three to continue their “whack-a mole” in circumventing piecemeal regulatory and legislative policies.
You’ve followed PBM practices closely for many years. Was there anything in this interim FTC report that surprised you?
Though not surprised, I am glad that it was released because it had been a year in investigation and there were many requests for some type of substantive report.
Two things that are missing that I feel are paramount are investigating how the three big PBMs are causing physical harm to patients as a result of the profit component in formulary construction and the profound financial impact of hidden PBM profit centers in self-insured employer health plans.
What we have seen over the years is the result of the perverse incentives for the PBMs to prefer the most profitable medications on their formularies.
They use utilization management tools such as step therapy, nonmedical switching, and exclusions to maintain their formularies’ profitability. These tools have been shown to delay and deny the proper care of patients, resulting in not just monetary but physical harm as well.
I would think the physical harm done to patients in manipulating the formularies should be addressed in this report as well and, in fact, may be the most important aspect of consumer protection of this issue.
In terms of the FTC’s mission to not “unduly burden” legitimate business, I would like to see the sector of self-insured employers addressed.
The report details how PBMs steer prescriptions to their affiliated pharmacies. The FTC says that can push smaller pharmacies out of the market, ultimately leading to higher costs and lower quality services for people. What’s your perspective?
Having more community pharmacies is better than having less. We are seeing more “pharmacy deserts” in rural areas as a result of many community pharmacies having to close.
The FTC voted 4-1 to allow staff to issue the interim report, with Commissioner Melissa Holyoak voting no. And some FTC commissioners seem divided on the usefulness of the report. Why?
Commissioner Holyoak states the “the Report leaves us without a better understanding of the competition concerns surrounding PBMs or how consumers are impacted by PBM practices.”
I do agree with her that the harm to patients’ medical status was not even addressed as far as I could tell in this report. There are multiple news articles and reports on the harms inflicted upon patients by the UM tools that drive the construction of ever changing formularies, all based on contracting with manufacturers that result in the highest profit for the PBM.
Holyoak also states, “Among other critical conclusions, the Report does not address the seemingly contradictory conclusions in the 2005 Report that PBMs, including vertically owned PBMs, generated cost savings for consumers.”
That may be true, but in 2005, the rise of PBMs was just beginning and the huge vertical and horizontal integration had yet to begin. Also, 2005 was still in the beginning of the biologic drug deluge, which did create competition to get on the formulary. Since then, PBMs have done nothing to control the rise in prices but instead, apparently have used the competition to get higher price concessions from manufacturers based on a percentage of the list price to line their pockets.
Commissioner Ferguson agreed with releasing the report but he had many issues with this report including the lack of PBM response.
I do agree with him that the FTC should have used some type of “force” to get the information they needed from the PBMs. The Big Three are known for obfuscation and delaying providing information to legislative and regulatory agencies.
A version of this article appeared on Medscape.com.
Rising consolidation among pharmacy benefit managers (PBMs) allows the companies to profit at the expense of patients and independent pharmacists. That’s the conclusion of a recent Federal Trade Commission (FTC) report on interim findings from the agency’s ongoing investigation of PBMs.
Lawmakers are increasingly scrutinizing the industry amid growing concern among physicians and consumers about how PBMs exploit their market dominance. The top six PBMs managed 94% of US drug claims in 2023, with the majority handled by the industry’s three giants: CVS Caremark, Cigna’s Express Scripts, and United Healthcare’s OptumRx.
PBMs manage prescription drug benefits for health insurers, Medicare Part D drug plans, and large employers. They act as middlemen between health insurers and pharmacies, developing formularies of covered drugs and promising savings from the discounts and rebates they negotiate with drugmakers.
The FTC’s interim report found that the giant PBMs often exercise significant control over what drugs are available and at what price and which pharmacies patients can use to access their prescribed medications. Consumers suffer as a result, the report concluded.
Madelaine A. Feldman, MD, vice president for advocacy and government affairs for the Coalition of State Rheumatology Organizations, shared her perspective on the FTC report in an email Q&A with this news organization. She is affiliated with The Rheumatology Group, based in Metairie, Louisiana.
Dr. Feldman has long tracked the PBM industry and appeared as a witness before influential government panels, including the House Energy and Commerce Committee. She has highlighted for lawmakers the challenges physicians face in helping patients get needed medicines.
For example, she shared cases of PBMs steering patients toward the more expensive of three widely used rheumatoid arthritis medicines that have a similar mechanism of action, the Janus kinase (JAK) inhibitors, Dr. Feldman said.
One of the drugs cost roughly half of the other two — about $30,000 per year vs $65,000-$70,000. Yet only the two expensive drugs were included in the PBM formulary. As a result, the cheapest drug holds only a sliver of market share; the remainder is dominated by the two expensive products, she told the House Oversight and Accountability Committee in 2021.
This Q&A has been edited for length and clarity.
What would you want federal and state policymakers to do in response to the FTC’s report?
I think Congress needs to clearly delineate the differences between anticompetitive pharmacy issues, drug pricing issues, and their effect on formulary construction issues.
Lawmakers should demand more transparency and consider legislation that would remove perverse incentives that prompt PBMs to choose higher priced drugs for their formularies.
That may require other regulatory or legislative actions to ensure lower prices (not higher kickbacks) are incentivized. Ultimately, in order to gain true competition within the health insurance business, these oligopolies of multiple businesses need to be broken up. Anything less seems to be nibbling around the edges and allows the Big Three to continue their “whack-a mole” in circumventing piecemeal regulatory and legislative policies.
You’ve followed PBM practices closely for many years. Was there anything in this interim FTC report that surprised you?
Though not surprised, I am glad that it was released because it had been a year in investigation and there were many requests for some type of substantive report.
Two things that are missing that I feel are paramount are investigating how the three big PBMs are causing physical harm to patients as a result of the profit component in formulary construction and the profound financial impact of hidden PBM profit centers in self-insured employer health plans.
What we have seen over the years is the result of the perverse incentives for the PBMs to prefer the most profitable medications on their formularies.
They use utilization management tools such as step therapy, nonmedical switching, and exclusions to maintain their formularies’ profitability. These tools have been shown to delay and deny the proper care of patients, resulting in not just monetary but physical harm as well.
I would think the physical harm done to patients in manipulating the formularies should be addressed in this report as well and, in fact, may be the most important aspect of consumer protection of this issue.
In terms of the FTC’s mission to not “unduly burden” legitimate business, I would like to see the sector of self-insured employers addressed.
The report details how PBMs steer prescriptions to their affiliated pharmacies. The FTC says that can push smaller pharmacies out of the market, ultimately leading to higher costs and lower quality services for people. What’s your perspective?
Having more community pharmacies is better than having less. We are seeing more “pharmacy deserts” in rural areas as a result of many community pharmacies having to close.
The FTC voted 4-1 to allow staff to issue the interim report, with Commissioner Melissa Holyoak voting no. And some FTC commissioners seem divided on the usefulness of the report. Why?
Commissioner Holyoak states the “the Report leaves us without a better understanding of the competition concerns surrounding PBMs or how consumers are impacted by PBM practices.”
I do agree with her that the harm to patients’ medical status was not even addressed as far as I could tell in this report. There are multiple news articles and reports on the harms inflicted upon patients by the UM tools that drive the construction of ever changing formularies, all based on contracting with manufacturers that result in the highest profit for the PBM.
Holyoak also states, “Among other critical conclusions, the Report does not address the seemingly contradictory conclusions in the 2005 Report that PBMs, including vertically owned PBMs, generated cost savings for consumers.”
That may be true, but in 2005, the rise of PBMs was just beginning and the huge vertical and horizontal integration had yet to begin. Also, 2005 was still in the beginning of the biologic drug deluge, which did create competition to get on the formulary. Since then, PBMs have done nothing to control the rise in prices but instead, apparently have used the competition to get higher price concessions from manufacturers based on a percentage of the list price to line their pockets.
Commissioner Ferguson agreed with releasing the report but he had many issues with this report including the lack of PBM response.
I do agree with him that the FTC should have used some type of “force” to get the information they needed from the PBMs. The Big Three are known for obfuscation and delaying providing information to legislative and regulatory agencies.
A version of this article appeared on Medscape.com.
Rising consolidation among pharmacy benefit managers (PBMs) allows the companies to profit at the expense of patients and independent pharmacists. That’s the conclusion of a recent Federal Trade Commission (FTC) report on interim findings from the agency’s ongoing investigation of PBMs.
Lawmakers are increasingly scrutinizing the industry amid growing concern among physicians and consumers about how PBMs exploit their market dominance. The top six PBMs managed 94% of US drug claims in 2023, with the majority handled by the industry’s three giants: CVS Caremark, Cigna’s Express Scripts, and United Healthcare’s OptumRx.
PBMs manage prescription drug benefits for health insurers, Medicare Part D drug plans, and large employers. They act as middlemen between health insurers and pharmacies, developing formularies of covered drugs and promising savings from the discounts and rebates they negotiate with drugmakers.
The FTC’s interim report found that the giant PBMs often exercise significant control over what drugs are available and at what price and which pharmacies patients can use to access their prescribed medications. Consumers suffer as a result, the report concluded.
Madelaine A. Feldman, MD, vice president for advocacy and government affairs for the Coalition of State Rheumatology Organizations, shared her perspective on the FTC report in an email Q&A with this news organization. She is affiliated with The Rheumatology Group, based in Metairie, Louisiana.
Dr. Feldman has long tracked the PBM industry and appeared as a witness before influential government panels, including the House Energy and Commerce Committee. She has highlighted for lawmakers the challenges physicians face in helping patients get needed medicines.
For example, she shared cases of PBMs steering patients toward the more expensive of three widely used rheumatoid arthritis medicines that have a similar mechanism of action, the Janus kinase (JAK) inhibitors, Dr. Feldman said.
One of the drugs cost roughly half of the other two — about $30,000 per year vs $65,000-$70,000. Yet only the two expensive drugs were included in the PBM formulary. As a result, the cheapest drug holds only a sliver of market share; the remainder is dominated by the two expensive products, she told the House Oversight and Accountability Committee in 2021.
This Q&A has been edited for length and clarity.
What would you want federal and state policymakers to do in response to the FTC’s report?
I think Congress needs to clearly delineate the differences between anticompetitive pharmacy issues, drug pricing issues, and their effect on formulary construction issues.
Lawmakers should demand more transparency and consider legislation that would remove perverse incentives that prompt PBMs to choose higher priced drugs for their formularies.
That may require other regulatory or legislative actions to ensure lower prices (not higher kickbacks) are incentivized. Ultimately, in order to gain true competition within the health insurance business, these oligopolies of multiple businesses need to be broken up. Anything less seems to be nibbling around the edges and allows the Big Three to continue their “whack-a mole” in circumventing piecemeal regulatory and legislative policies.
You’ve followed PBM practices closely for many years. Was there anything in this interim FTC report that surprised you?
Though not surprised, I am glad that it was released because it had been a year in investigation and there were many requests for some type of substantive report.
Two things that are missing that I feel are paramount are investigating how the three big PBMs are causing physical harm to patients as a result of the profit component in formulary construction and the profound financial impact of hidden PBM profit centers in self-insured employer health plans.
What we have seen over the years is the result of the perverse incentives for the PBMs to prefer the most profitable medications on their formularies.
They use utilization management tools such as step therapy, nonmedical switching, and exclusions to maintain their formularies’ profitability. These tools have been shown to delay and deny the proper care of patients, resulting in not just monetary but physical harm as well.
I would think the physical harm done to patients in manipulating the formularies should be addressed in this report as well and, in fact, may be the most important aspect of consumer protection of this issue.
In terms of the FTC’s mission to not “unduly burden” legitimate business, I would like to see the sector of self-insured employers addressed.
The report details how PBMs steer prescriptions to their affiliated pharmacies. The FTC says that can push smaller pharmacies out of the market, ultimately leading to higher costs and lower quality services for people. What’s your perspective?
Having more community pharmacies is better than having less. We are seeing more “pharmacy deserts” in rural areas as a result of many community pharmacies having to close.
The FTC voted 4-1 to allow staff to issue the interim report, with Commissioner Melissa Holyoak voting no. And some FTC commissioners seem divided on the usefulness of the report. Why?
Commissioner Holyoak states the “the Report leaves us without a better understanding of the competition concerns surrounding PBMs or how consumers are impacted by PBM practices.”
I do agree with her that the harm to patients’ medical status was not even addressed as far as I could tell in this report. There are multiple news articles and reports on the harms inflicted upon patients by the UM tools that drive the construction of ever changing formularies, all based on contracting with manufacturers that result in the highest profit for the PBM.
Holyoak also states, “Among other critical conclusions, the Report does not address the seemingly contradictory conclusions in the 2005 Report that PBMs, including vertically owned PBMs, generated cost savings for consumers.”
That may be true, but in 2005, the rise of PBMs was just beginning and the huge vertical and horizontal integration had yet to begin. Also, 2005 was still in the beginning of the biologic drug deluge, which did create competition to get on the formulary. Since then, PBMs have done nothing to control the rise in prices but instead, apparently have used the competition to get higher price concessions from manufacturers based on a percentage of the list price to line their pockets.
Commissioner Ferguson agreed with releasing the report but he had many issues with this report including the lack of PBM response.
I do agree with him that the FTC should have used some type of “force” to get the information they needed from the PBMs. The Big Three are known for obfuscation and delaying providing information to legislative and regulatory agencies.
A version of this article appeared on Medscape.com.
Family Not Flourishing? The Hidden Issue of Food Insecurity and How We Can Help
As pediatric providers we are dedicated to helping children have optimal health. And what could be more basic to health than having enough food? Yet, even in one of the richest countries on the planet, as much as 25% of US families are reported to have “food insecurity.”
What does this mean? The US Department of Agriculture (USDA), the agency tracking and addressing food issues, defines food security as “marginal” when there are one or two indications, typically anxiety over food sufficiency or shortage of food even with little or no alteration of diet or intake. “Low” includes reduced quality, variety, or desirability of the diet but little or no reduced intake. When eating patterns are disrupted and intake is reduced, this is considered “very low food security.” “Hunger” refers to an individual’s physiological state when prolonged, involuntary lack of food results in discomfort, illness, weakness, or pain beyond the usual uneasy sensation. Pediatric researchers include in the definition lack of access to enough food for an active and healthy life. I will use the common term “food insecurity” here.
Children under 3 years old in homes with food insecurity have been found to be sick more often, recover more slowly from illness, and be hospitalized more frequently. Deficiencies in nutrition vary by age, with children under 6 having low vegetable intake and low iron, ages 6-11 excess sugary food intake and lower bone density in boys, and adolescents, although harder to measure, had low iron.
Physical and Mental Effects of Food Insecurity
Associated with food insecurity in the home are more developmental delays in children 4-36 months old. Beyond that, children of all ages have lower cognitive indicators, dysregulated behavior, and emotional distress than those in homes with secure food access. These are persisting deficits: kindergarten children with food insecurity have lower math and reading abilities over at least 4 years.
Mental health is also affected by food insecurity. Reviews of multiple studies of children from preschool through adolescence show more child hyperactivity, emotional dysregulation, anxiety, depression, and stress beyond those attributable to their mother’s depression and anxiety. Food insecurity in the early years is associated with mental health issues even into adolescence. School aged children and youth are well aware of the family’s struggle with food access, even when their parents do not realize this. In addition to the anxiety and depression, they may feel shame or be socially ostracized. They may eat less, or choose low-quality foods to cope. Adolescents experiencing food insecurity report greater dysthymia and suicidal ideation. It is unknown whether these mental health difficulties are due to the stress, shame, or decreased intake of macronutrients important to emotional regulation or all of these. One implication is that pediatric providers should also screen for food insecurity as well as other social drivers of health (SDOH) when addressing developmental, behavioral, or mental health issues, not just at well visits.
While we worry about effects for the child, impact of food insecurity on caregivers is significant for parenting as well as adult well-being beginning prenatally. First trimester food insecurity is associated with increased maternal stress at 2 months postpartum and lower bonding scores at 6 months, although this is moderated by social support. The stress of food insecurity and other SDoH present are associated with parental depression, anxiety, and toxic stress, making optimal parenting difficult. Caregivers experiencing insecure food access worry most about their children and may reduce their own eating and food quality to spare the child. More than 30% of families indicated that they had to choose between paying for food and paying for medicine or medical care, jeopardizing their health, making this an important point of discussion for us as well.
Quality Versus Quantity
The total amount of food is not the only factor in adequate child nutrition. Healthy foods usually cost more and also may not be conveniently available. There are so called “food deserts,” areas with few/no full-service grocery stores, and also “food swamps” where unhealthy foods (eg fast food) are more available than healthy options. Life stress, higher in low-income populations, increases the impulse to consume sweet or high-fat “comfort foods” (we all know this!) due to the rush of calories and quick satiety. Children may be influenced in their food choices by media messages about non-nutritious foods. All of these may explain the association of food insecurity with obesity in both children and adults. It also sets them up for lifetime health problems of diabetes, hyperlipidemia, and cardiovascular conditions, especially in racial and ethnic minority groups and the poor.
The Larger Picture
Obvious to us all, low income is the main reason for inadequate access to enough or good quality food. Over 60% of families with food insecurity had incomes below the poverty threshold in 2013. Households without children are half as likely to be food insecure. But as 30% of food-insecure households have incomes above the eligibility cut offs for food programs — typically 130% of poverty for Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) or 185% for Supplemental Nutrition Assistance Program (SNAP) — it is clear the problem is not related solely to poverty. Even small changes in income or expenses, such as a car breaking down, or heating or medical bills, can quickly result in inability to afford food, especially in areas of high food costs. This is particularly true for immigrant, large, and single-parent families and those with less education. Federal food programs do not cover all food needs for every family.
But we can’t tell if a child lives in a family with food insecurity by whether the child is thin, dropping growth percentiles, or receiving Medicaid insurance. Parents, and even youth, may be reluctant to tell us that they do not have enough to eat out of pride, fear, of prejudice, being reported to a contentious ex, being detected as an illegal immigrant, or even reported for neglect and having their child removed. Because of the suffering and impacts of food insecurity on child well-being, the American Academy of Pediatrics (AAP) published a Policy Statement in 2015, reaffirmed in 2021, recommending screening for food insecurity at all well visits and a toolkit to help. The USDA 18-item Household Food Security Scale (HFSS) has been the gold-standard screen, but affirmative answers to either of the 2-item Hunger Vital Sign (HVS) questionnaire identifies food insecurity with a sensitivity of 97% and a specificity of 83% compared with the HFSS. The questions ask how often the following were true in the past year: 1) “We worried whether our food would run out before we got money to buy more” and 2) “The food we bought just didn’t last and we didn’t have money to buy more.” This brief screen is now recommended and practical.
Screening for Food Insecurity
All set to manage food insecurity in your practice, then? Not exactly. Screening is only useful if it results in access to food. A study in a majority low-income clinic found that parents reported food insecurity 7% of the time when the clinician asked the HVS questions versus 45% when they self-reported on paper. Parent focus groups revealed reasons for the discrepant underreporting to the clinician: shame, concerns about stigma, and fear of the child being taken away. They felt more comfortable reporting about their child than about their own family situation. When asked what the clinician did that helped them disclose food insecurity, the caregivers cited strong interpersonal skills, open body language, and empathy. They also requested being given resources for other social issues, not just food insecurity. Clinic staff found paper screening inconvenient and recommended using tablet devices (such as with CHADIS that also scores and provides interviewing help, education, and local resource listings). Clinicians found the need for a follow-up conversation time consuming. Clinic staff thought screening could be facilitated by clinician’s initiating conversations, taking care about children present, and normalizing the screen as applying to all. Caregivers wanted know the use and privacy of the information. This same clinic referred the caregivers to a Benefits Data Trust with a goal of enrolling them in food programs. Of the food insecure, 55% were referred but only one third could be reached by phone with three attempts by the benefits group. Subsequent enrollment of those reached had barriers of verification requirements, wait times, and perceived mistreatment. The program concluded that this difficult two-step process of screening and referral would be improved by an integrated system of screening and enrollment in public benefits. Provision of information about free local food resources is also important, as 84% of those already receiving SNAP benefits remained food insecure.
Offering Assistance
To assist families where food insecurity is found, we need to understand the options of services both for referrals and advocacy. The AAP toolkit is designed to help. For pregnant and postpartum women and children 0-5 years in families with income less than 130% of the poverty level, the WIC program provides electronic cards to purchase approved categories of healthy food from participating vendors. For families with incomes less than 185% of the poverty level, the SNAP program, formerly called food stamps, provides benefits. There are other programs including free and reduced cost National School Breakfast and/or Lunch programs (best when open to all), Child and Adult Care Food Program for institutions (which may include medical offices), and Summer Food Service Programs providing lunch at community sites. Since not all food-insecure families are eligible for the above services, it is important that we are ready to provide information about local food banks, pantries, and low- or no-cost produce programs (see Healthy Food Bank Hub, Feeding America, 2-1-1, or FindHelp.org).
As pediatric providers we have a special opportunity and responsibility to expand our capabilities for sensitively addressing and advocating for help for food insecurity to improve the outlook for the families under our care.
Dr. Howard is assistant professor of pediatrics at The Johns Hopkins University School of Medicine, Baltimore, and creator of CHADIS. She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at pdnews@mdedge.com.
As pediatric providers we are dedicated to helping children have optimal health. And what could be more basic to health than having enough food? Yet, even in one of the richest countries on the planet, as much as 25% of US families are reported to have “food insecurity.”
What does this mean? The US Department of Agriculture (USDA), the agency tracking and addressing food issues, defines food security as “marginal” when there are one or two indications, typically anxiety over food sufficiency or shortage of food even with little or no alteration of diet or intake. “Low” includes reduced quality, variety, or desirability of the diet but little or no reduced intake. When eating patterns are disrupted and intake is reduced, this is considered “very low food security.” “Hunger” refers to an individual’s physiological state when prolonged, involuntary lack of food results in discomfort, illness, weakness, or pain beyond the usual uneasy sensation. Pediatric researchers include in the definition lack of access to enough food for an active and healthy life. I will use the common term “food insecurity” here.
Children under 3 years old in homes with food insecurity have been found to be sick more often, recover more slowly from illness, and be hospitalized more frequently. Deficiencies in nutrition vary by age, with children under 6 having low vegetable intake and low iron, ages 6-11 excess sugary food intake and lower bone density in boys, and adolescents, although harder to measure, had low iron.
Physical and Mental Effects of Food Insecurity
Associated with food insecurity in the home are more developmental delays in children 4-36 months old. Beyond that, children of all ages have lower cognitive indicators, dysregulated behavior, and emotional distress than those in homes with secure food access. These are persisting deficits: kindergarten children with food insecurity have lower math and reading abilities over at least 4 years.
Mental health is also affected by food insecurity. Reviews of multiple studies of children from preschool through adolescence show more child hyperactivity, emotional dysregulation, anxiety, depression, and stress beyond those attributable to their mother’s depression and anxiety. Food insecurity in the early years is associated with mental health issues even into adolescence. School aged children and youth are well aware of the family’s struggle with food access, even when their parents do not realize this. In addition to the anxiety and depression, they may feel shame or be socially ostracized. They may eat less, or choose low-quality foods to cope. Adolescents experiencing food insecurity report greater dysthymia and suicidal ideation. It is unknown whether these mental health difficulties are due to the stress, shame, or decreased intake of macronutrients important to emotional regulation or all of these. One implication is that pediatric providers should also screen for food insecurity as well as other social drivers of health (SDOH) when addressing developmental, behavioral, or mental health issues, not just at well visits.
While we worry about effects for the child, impact of food insecurity on caregivers is significant for parenting as well as adult well-being beginning prenatally. First trimester food insecurity is associated with increased maternal stress at 2 months postpartum and lower bonding scores at 6 months, although this is moderated by social support. The stress of food insecurity and other SDoH present are associated with parental depression, anxiety, and toxic stress, making optimal parenting difficult. Caregivers experiencing insecure food access worry most about their children and may reduce their own eating and food quality to spare the child. More than 30% of families indicated that they had to choose between paying for food and paying for medicine or medical care, jeopardizing their health, making this an important point of discussion for us as well.
Quality Versus Quantity
The total amount of food is not the only factor in adequate child nutrition. Healthy foods usually cost more and also may not be conveniently available. There are so called “food deserts,” areas with few/no full-service grocery stores, and also “food swamps” where unhealthy foods (eg fast food) are more available than healthy options. Life stress, higher in low-income populations, increases the impulse to consume sweet or high-fat “comfort foods” (we all know this!) due to the rush of calories and quick satiety. Children may be influenced in their food choices by media messages about non-nutritious foods. All of these may explain the association of food insecurity with obesity in both children and adults. It also sets them up for lifetime health problems of diabetes, hyperlipidemia, and cardiovascular conditions, especially in racial and ethnic minority groups and the poor.
The Larger Picture
Obvious to us all, low income is the main reason for inadequate access to enough or good quality food. Over 60% of families with food insecurity had incomes below the poverty threshold in 2013. Households without children are half as likely to be food insecure. But as 30% of food-insecure households have incomes above the eligibility cut offs for food programs — typically 130% of poverty for Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) or 185% for Supplemental Nutrition Assistance Program (SNAP) — it is clear the problem is not related solely to poverty. Even small changes in income or expenses, such as a car breaking down, or heating or medical bills, can quickly result in inability to afford food, especially in areas of high food costs. This is particularly true for immigrant, large, and single-parent families and those with less education. Federal food programs do not cover all food needs for every family.
But we can’t tell if a child lives in a family with food insecurity by whether the child is thin, dropping growth percentiles, or receiving Medicaid insurance. Parents, and even youth, may be reluctant to tell us that they do not have enough to eat out of pride, fear, of prejudice, being reported to a contentious ex, being detected as an illegal immigrant, or even reported for neglect and having their child removed. Because of the suffering and impacts of food insecurity on child well-being, the American Academy of Pediatrics (AAP) published a Policy Statement in 2015, reaffirmed in 2021, recommending screening for food insecurity at all well visits and a toolkit to help. The USDA 18-item Household Food Security Scale (HFSS) has been the gold-standard screen, but affirmative answers to either of the 2-item Hunger Vital Sign (HVS) questionnaire identifies food insecurity with a sensitivity of 97% and a specificity of 83% compared with the HFSS. The questions ask how often the following were true in the past year: 1) “We worried whether our food would run out before we got money to buy more” and 2) “The food we bought just didn’t last and we didn’t have money to buy more.” This brief screen is now recommended and practical.
Screening for Food Insecurity
All set to manage food insecurity in your practice, then? Not exactly. Screening is only useful if it results in access to food. A study in a majority low-income clinic found that parents reported food insecurity 7% of the time when the clinician asked the HVS questions versus 45% when they self-reported on paper. Parent focus groups revealed reasons for the discrepant underreporting to the clinician: shame, concerns about stigma, and fear of the child being taken away. They felt more comfortable reporting about their child than about their own family situation. When asked what the clinician did that helped them disclose food insecurity, the caregivers cited strong interpersonal skills, open body language, and empathy. They also requested being given resources for other social issues, not just food insecurity. Clinic staff found paper screening inconvenient and recommended using tablet devices (such as with CHADIS that also scores and provides interviewing help, education, and local resource listings). Clinicians found the need for a follow-up conversation time consuming. Clinic staff thought screening could be facilitated by clinician’s initiating conversations, taking care about children present, and normalizing the screen as applying to all. Caregivers wanted know the use and privacy of the information. This same clinic referred the caregivers to a Benefits Data Trust with a goal of enrolling them in food programs. Of the food insecure, 55% were referred but only one third could be reached by phone with three attempts by the benefits group. Subsequent enrollment of those reached had barriers of verification requirements, wait times, and perceived mistreatment. The program concluded that this difficult two-step process of screening and referral would be improved by an integrated system of screening and enrollment in public benefits. Provision of information about free local food resources is also important, as 84% of those already receiving SNAP benefits remained food insecure.
Offering Assistance
To assist families where food insecurity is found, we need to understand the options of services both for referrals and advocacy. The AAP toolkit is designed to help. For pregnant and postpartum women and children 0-5 years in families with income less than 130% of the poverty level, the WIC program provides electronic cards to purchase approved categories of healthy food from participating vendors. For families with incomes less than 185% of the poverty level, the SNAP program, formerly called food stamps, provides benefits. There are other programs including free and reduced cost National School Breakfast and/or Lunch programs (best when open to all), Child and Adult Care Food Program for institutions (which may include medical offices), and Summer Food Service Programs providing lunch at community sites. Since not all food-insecure families are eligible for the above services, it is important that we are ready to provide information about local food banks, pantries, and low- or no-cost produce programs (see Healthy Food Bank Hub, Feeding America, 2-1-1, or FindHelp.org).
As pediatric providers we have a special opportunity and responsibility to expand our capabilities for sensitively addressing and advocating for help for food insecurity to improve the outlook for the families under our care.
Dr. Howard is assistant professor of pediatrics at The Johns Hopkins University School of Medicine, Baltimore, and creator of CHADIS. She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at pdnews@mdedge.com.
As pediatric providers we are dedicated to helping children have optimal health. And what could be more basic to health than having enough food? Yet, even in one of the richest countries on the planet, as much as 25% of US families are reported to have “food insecurity.”
What does this mean? The US Department of Agriculture (USDA), the agency tracking and addressing food issues, defines food security as “marginal” when there are one or two indications, typically anxiety over food sufficiency or shortage of food even with little or no alteration of diet or intake. “Low” includes reduced quality, variety, or desirability of the diet but little or no reduced intake. When eating patterns are disrupted and intake is reduced, this is considered “very low food security.” “Hunger” refers to an individual’s physiological state when prolonged, involuntary lack of food results in discomfort, illness, weakness, or pain beyond the usual uneasy sensation. Pediatric researchers include in the definition lack of access to enough food for an active and healthy life. I will use the common term “food insecurity” here.
Children under 3 years old in homes with food insecurity have been found to be sick more often, recover more slowly from illness, and be hospitalized more frequently. Deficiencies in nutrition vary by age, with children under 6 having low vegetable intake and low iron, ages 6-11 excess sugary food intake and lower bone density in boys, and adolescents, although harder to measure, had low iron.
Physical and Mental Effects of Food Insecurity
Associated with food insecurity in the home are more developmental delays in children 4-36 months old. Beyond that, children of all ages have lower cognitive indicators, dysregulated behavior, and emotional distress than those in homes with secure food access. These are persisting deficits: kindergarten children with food insecurity have lower math and reading abilities over at least 4 years.
Mental health is also affected by food insecurity. Reviews of multiple studies of children from preschool through adolescence show more child hyperactivity, emotional dysregulation, anxiety, depression, and stress beyond those attributable to their mother’s depression and anxiety. Food insecurity in the early years is associated with mental health issues even into adolescence. School aged children and youth are well aware of the family’s struggle with food access, even when their parents do not realize this. In addition to the anxiety and depression, they may feel shame or be socially ostracized. They may eat less, or choose low-quality foods to cope. Adolescents experiencing food insecurity report greater dysthymia and suicidal ideation. It is unknown whether these mental health difficulties are due to the stress, shame, or decreased intake of macronutrients important to emotional regulation or all of these. One implication is that pediatric providers should also screen for food insecurity as well as other social drivers of health (SDOH) when addressing developmental, behavioral, or mental health issues, not just at well visits.
While we worry about effects for the child, impact of food insecurity on caregivers is significant for parenting as well as adult well-being beginning prenatally. First trimester food insecurity is associated with increased maternal stress at 2 months postpartum and lower bonding scores at 6 months, although this is moderated by social support. The stress of food insecurity and other SDoH present are associated with parental depression, anxiety, and toxic stress, making optimal parenting difficult. Caregivers experiencing insecure food access worry most about their children and may reduce their own eating and food quality to spare the child. More than 30% of families indicated that they had to choose between paying for food and paying for medicine or medical care, jeopardizing their health, making this an important point of discussion for us as well.
Quality Versus Quantity
The total amount of food is not the only factor in adequate child nutrition. Healthy foods usually cost more and also may not be conveniently available. There are so called “food deserts,” areas with few/no full-service grocery stores, and also “food swamps” where unhealthy foods (eg fast food) are more available than healthy options. Life stress, higher in low-income populations, increases the impulse to consume sweet or high-fat “comfort foods” (we all know this!) due to the rush of calories and quick satiety. Children may be influenced in their food choices by media messages about non-nutritious foods. All of these may explain the association of food insecurity with obesity in both children and adults. It also sets them up for lifetime health problems of diabetes, hyperlipidemia, and cardiovascular conditions, especially in racial and ethnic minority groups and the poor.
The Larger Picture
Obvious to us all, low income is the main reason for inadequate access to enough or good quality food. Over 60% of families with food insecurity had incomes below the poverty threshold in 2013. Households without children are half as likely to be food insecure. But as 30% of food-insecure households have incomes above the eligibility cut offs for food programs — typically 130% of poverty for Special Supplemental Nutrition Program for Women, Infants, and Children (WIC) or 185% for Supplemental Nutrition Assistance Program (SNAP) — it is clear the problem is not related solely to poverty. Even small changes in income or expenses, such as a car breaking down, or heating or medical bills, can quickly result in inability to afford food, especially in areas of high food costs. This is particularly true for immigrant, large, and single-parent families and those with less education. Federal food programs do not cover all food needs for every family.
But we can’t tell if a child lives in a family with food insecurity by whether the child is thin, dropping growth percentiles, or receiving Medicaid insurance. Parents, and even youth, may be reluctant to tell us that they do not have enough to eat out of pride, fear, of prejudice, being reported to a contentious ex, being detected as an illegal immigrant, or even reported for neglect and having their child removed. Because of the suffering and impacts of food insecurity on child well-being, the American Academy of Pediatrics (AAP) published a Policy Statement in 2015, reaffirmed in 2021, recommending screening for food insecurity at all well visits and a toolkit to help. The USDA 18-item Household Food Security Scale (HFSS) has been the gold-standard screen, but affirmative answers to either of the 2-item Hunger Vital Sign (HVS) questionnaire identifies food insecurity with a sensitivity of 97% and a specificity of 83% compared with the HFSS. The questions ask how often the following were true in the past year: 1) “We worried whether our food would run out before we got money to buy more” and 2) “The food we bought just didn’t last and we didn’t have money to buy more.” This brief screen is now recommended and practical.
Screening for Food Insecurity
All set to manage food insecurity in your practice, then? Not exactly. Screening is only useful if it results in access to food. A study in a majority low-income clinic found that parents reported food insecurity 7% of the time when the clinician asked the HVS questions versus 45% when they self-reported on paper. Parent focus groups revealed reasons for the discrepant underreporting to the clinician: shame, concerns about stigma, and fear of the child being taken away. They felt more comfortable reporting about their child than about their own family situation. When asked what the clinician did that helped them disclose food insecurity, the caregivers cited strong interpersonal skills, open body language, and empathy. They also requested being given resources for other social issues, not just food insecurity. Clinic staff found paper screening inconvenient and recommended using tablet devices (such as with CHADIS that also scores and provides interviewing help, education, and local resource listings). Clinicians found the need for a follow-up conversation time consuming. Clinic staff thought screening could be facilitated by clinician’s initiating conversations, taking care about children present, and normalizing the screen as applying to all. Caregivers wanted know the use and privacy of the information. This same clinic referred the caregivers to a Benefits Data Trust with a goal of enrolling them in food programs. Of the food insecure, 55% were referred but only one third could be reached by phone with three attempts by the benefits group. Subsequent enrollment of those reached had barriers of verification requirements, wait times, and perceived mistreatment. The program concluded that this difficult two-step process of screening and referral would be improved by an integrated system of screening and enrollment in public benefits. Provision of information about free local food resources is also important, as 84% of those already receiving SNAP benefits remained food insecure.
Offering Assistance
To assist families where food insecurity is found, we need to understand the options of services both for referrals and advocacy. The AAP toolkit is designed to help. For pregnant and postpartum women and children 0-5 years in families with income less than 130% of the poverty level, the WIC program provides electronic cards to purchase approved categories of healthy food from participating vendors. For families with incomes less than 185% of the poverty level, the SNAP program, formerly called food stamps, provides benefits. There are other programs including free and reduced cost National School Breakfast and/or Lunch programs (best when open to all), Child and Adult Care Food Program for institutions (which may include medical offices), and Summer Food Service Programs providing lunch at community sites. Since not all food-insecure families are eligible for the above services, it is important that we are ready to provide information about local food banks, pantries, and low- or no-cost produce programs (see Healthy Food Bank Hub, Feeding America, 2-1-1, or FindHelp.org).
As pediatric providers we have a special opportunity and responsibility to expand our capabilities for sensitively addressing and advocating for help for food insecurity to improve the outlook for the families under our care.
Dr. Howard is assistant professor of pediatrics at The Johns Hopkins University School of Medicine, Baltimore, and creator of CHADIS. She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at pdnews@mdedge.com.
What You Need to Know About Oropouche Virus Disease
The European Centre for Disease Prevention and Control (ECDC) has issued a warning to travelers in areas in South and Central America and the Caribbean affected by a current outbreak of Oropouche virus (OROV) disease. The ECDC said that there had been more than 8000 cases reported in these areas since January, with 19 imported cases reported in Europe for the first time in June and July. Of these, 12 were in Spain, five were in Italy, and two were in Germany.
The ECDC’s Threat Assessment Brief of Aug. 9 said that one of those affected had traveled to Brazil and the other 18 to Cuba; however, outbreaks have also been reported this year in Bolivia, Colombia, and Peru. Though the overall risk for infection to European travelers to OROV-epidemic countries was assessed as moderate, it was higher in the more affected municipalities of the northern states of Brazil and/or the Amazon region, and/or if personal protection measures are not taken.
An editorial published Aug. 8 in The Lancet Infectious Diseases described OROV as a “mysterious threat,” which there is limited knowledge about despite some half a million cases recorded since it was first detected in Trinidad and Tobago in 1955.
OROV is transmitted primarily through bites from infected midges (Culicoides paraensis). However, some mosquitoes species can also spread the virus, which causes symptoms very similar to other arbovirus diseases from the same regions, such as dengue, chikungunya, and Zika virus infection.
Most cases are mild, but meningitis and encephalitis can occur as well as possible fetal death and deformities after infection in pregnancy. Last month, the first fatal cases were reported in two young Brazilian women who, concerningly, had no comorbidities.
This news organization asked Jan Felix Drexler, MD, of the Institute of Virology at Charité – Universitätsmedizin in Berlin, Germany, who has studied the emergence of Oropouche fever in Latin America, what clinicians should know about OROV disease.
What are the main symptoms of OROV disease for which clinicians should be alert?
The main symptoms are not different from other arboviral infections, ie, fever, maybe joint and muscle pain, maybe rash. The problem is that we do not know how often severe disease may occur because we do not know whether the severe cases that have been postulated, including death in apparently healthy people and congenital infection, are due to increased testing; an altered virus; or an altered, more intense circulation (so that many more infections simply lead to rare severe cases appearing). Be alert and ask for testing in your patients.
What is the differential diagnosis if a recent traveler to affected regions presents with symptoms? Are there any clues to suggest whether the disease is Oropouche as opposed to Zika, etc.?
The main message is: Do not assume a particular infection based on clinical symptoms. If your patient is returning from or living in an endemic area, consider OROV disease in the differential diagnosis.
What personal protective measures should clinicians advise travelers in affected areas to take? Do these differ from normal mosquito precautions?
Repellents are extremely important as usual. However, there are differences. Mosquito nets’ hole sizes need to be smaller than those used against the vectors of malaria or dengue; in other words, they need to have a higher mesh. The problem is that nets with high mesh are complicated in very hot and humid conditions because they also limit ventilation. Travelers should discuss with local suppliers about the best trade-off.
The risk for midge bites is likely highest at dawn and dusk in still and humid conditions. So on the one hand, one could recommend avoiding those areas and being outside during those times of the day. On the other hand, specific recommendations cannot be made robustly because we cannot exclude other invertebrate vectors at current knowledge. Some studies have implicated that mosquitoes may also transmit the virus. If that holds true, then we are back to reducing any bite.
Should pregnant women be advised to avoid travel to affected regions?
Not immediately, but caution must be taken. We simply do not have sufficient data to gauge the risk for potential congenital infection. Much more epidemiologic data and controlled infection experiments will be required to make evidence-based recommendations.
All the cases reported in Europe so far were imported from Cuba and Brazil. Is there any risk for local transmission, eg, via midges/mosquitoes that might hitch a ride on an aircraft, as in cases of airport malaria?
Not immediately, but it cannot be excluded. We know very little about the infection intensity in the vectors. Controlled infection experiments, including robustness of vectors against commonly used insecticides in airplanes, need to be done.
What is the risk for an animal reservoir emerging in Europe?
We do not know, but there is also no reason for ringing the alarm bells. Controlled infection experiments and surveillance will be required.
Is treatment purely supportive or are there any specific agents worth trying in case of severe symptoms/neurologic involvement?
No specific treatment can be recommended as is. However, severe dengue illustrates the relevance of supportive treatment, which is hugely effective in reducing mortality.
The Lancet paper states: “Several laboratory tests have been developed but robust commercial tests are hardly available.” How likely is it that laboratories in Europe will have the capability to test for the Oropouche organism?
European laboratory networks have already taken action, and testing is now available at least in the major and reference laboratories. If a clinician asks for OROV testing, they will probably get a robust answer in a reasonable timespan. Of course, that can be improved once we have more cases and more laboratories will be equipped for testing.
Is there anything else you think clinicians should be aware of?
The most important is to think beyond the textbooks we know from medical school. Things change rapidly in a connected world under altered climate conditions.
Dr. Drexler has no conflicts of interest to declare.
A version of this article first appeared on Medscape.com.
The European Centre for Disease Prevention and Control (ECDC) has issued a warning to travelers in areas in South and Central America and the Caribbean affected by a current outbreak of Oropouche virus (OROV) disease. The ECDC said that there had been more than 8000 cases reported in these areas since January, with 19 imported cases reported in Europe for the first time in June and July. Of these, 12 were in Spain, five were in Italy, and two were in Germany.
The ECDC’s Threat Assessment Brief of Aug. 9 said that one of those affected had traveled to Brazil and the other 18 to Cuba; however, outbreaks have also been reported this year in Bolivia, Colombia, and Peru. Though the overall risk for infection to European travelers to OROV-epidemic countries was assessed as moderate, it was higher in the more affected municipalities of the northern states of Brazil and/or the Amazon region, and/or if personal protection measures are not taken.
An editorial published Aug. 8 in The Lancet Infectious Diseases described OROV as a “mysterious threat,” which there is limited knowledge about despite some half a million cases recorded since it was first detected in Trinidad and Tobago in 1955.
OROV is transmitted primarily through bites from infected midges (Culicoides paraensis). However, some mosquitoes species can also spread the virus, which causes symptoms very similar to other arbovirus diseases from the same regions, such as dengue, chikungunya, and Zika virus infection.
Most cases are mild, but meningitis and encephalitis can occur as well as possible fetal death and deformities after infection in pregnancy. Last month, the first fatal cases were reported in two young Brazilian women who, concerningly, had no comorbidities.
This news organization asked Jan Felix Drexler, MD, of the Institute of Virology at Charité – Universitätsmedizin in Berlin, Germany, who has studied the emergence of Oropouche fever in Latin America, what clinicians should know about OROV disease.
What are the main symptoms of OROV disease for which clinicians should be alert?
The main symptoms are not different from other arboviral infections, ie, fever, maybe joint and muscle pain, maybe rash. The problem is that we do not know how often severe disease may occur because we do not know whether the severe cases that have been postulated, including death in apparently healthy people and congenital infection, are due to increased testing; an altered virus; or an altered, more intense circulation (so that many more infections simply lead to rare severe cases appearing). Be alert and ask for testing in your patients.
What is the differential diagnosis if a recent traveler to affected regions presents with symptoms? Are there any clues to suggest whether the disease is Oropouche as opposed to Zika, etc.?
The main message is: Do not assume a particular infection based on clinical symptoms. If your patient is returning from or living in an endemic area, consider OROV disease in the differential diagnosis.
What personal protective measures should clinicians advise travelers in affected areas to take? Do these differ from normal mosquito precautions?
Repellents are extremely important as usual. However, there are differences. Mosquito nets’ hole sizes need to be smaller than those used against the vectors of malaria or dengue; in other words, they need to have a higher mesh. The problem is that nets with high mesh are complicated in very hot and humid conditions because they also limit ventilation. Travelers should discuss with local suppliers about the best trade-off.
The risk for midge bites is likely highest at dawn and dusk in still and humid conditions. So on the one hand, one could recommend avoiding those areas and being outside during those times of the day. On the other hand, specific recommendations cannot be made robustly because we cannot exclude other invertebrate vectors at current knowledge. Some studies have implicated that mosquitoes may also transmit the virus. If that holds true, then we are back to reducing any bite.
Should pregnant women be advised to avoid travel to affected regions?
Not immediately, but caution must be taken. We simply do not have sufficient data to gauge the risk for potential congenital infection. Much more epidemiologic data and controlled infection experiments will be required to make evidence-based recommendations.
All the cases reported in Europe so far were imported from Cuba and Brazil. Is there any risk for local transmission, eg, via midges/mosquitoes that might hitch a ride on an aircraft, as in cases of airport malaria?
Not immediately, but it cannot be excluded. We know very little about the infection intensity in the vectors. Controlled infection experiments, including robustness of vectors against commonly used insecticides in airplanes, need to be done.
What is the risk for an animal reservoir emerging in Europe?
We do not know, but there is also no reason for ringing the alarm bells. Controlled infection experiments and surveillance will be required.
Is treatment purely supportive or are there any specific agents worth trying in case of severe symptoms/neurologic involvement?
No specific treatment can be recommended as is. However, severe dengue illustrates the relevance of supportive treatment, which is hugely effective in reducing mortality.
The Lancet paper states: “Several laboratory tests have been developed but robust commercial tests are hardly available.” How likely is it that laboratories in Europe will have the capability to test for the Oropouche organism?
European laboratory networks have already taken action, and testing is now available at least in the major and reference laboratories. If a clinician asks for OROV testing, they will probably get a robust answer in a reasonable timespan. Of course, that can be improved once we have more cases and more laboratories will be equipped for testing.
Is there anything else you think clinicians should be aware of?
The most important is to think beyond the textbooks we know from medical school. Things change rapidly in a connected world under altered climate conditions.
Dr. Drexler has no conflicts of interest to declare.
A version of this article first appeared on Medscape.com.
The European Centre for Disease Prevention and Control (ECDC) has issued a warning to travelers in areas in South and Central America and the Caribbean affected by a current outbreak of Oropouche virus (OROV) disease. The ECDC said that there had been more than 8000 cases reported in these areas since January, with 19 imported cases reported in Europe for the first time in June and July. Of these, 12 were in Spain, five were in Italy, and two were in Germany.
The ECDC’s Threat Assessment Brief of Aug. 9 said that one of those affected had traveled to Brazil and the other 18 to Cuba; however, outbreaks have also been reported this year in Bolivia, Colombia, and Peru. Though the overall risk for infection to European travelers to OROV-epidemic countries was assessed as moderate, it was higher in the more affected municipalities of the northern states of Brazil and/or the Amazon region, and/or if personal protection measures are not taken.
An editorial published Aug. 8 in The Lancet Infectious Diseases described OROV as a “mysterious threat,” which there is limited knowledge about despite some half a million cases recorded since it was first detected in Trinidad and Tobago in 1955.
OROV is transmitted primarily through bites from infected midges (Culicoides paraensis). However, some mosquitoes species can also spread the virus, which causes symptoms very similar to other arbovirus diseases from the same regions, such as dengue, chikungunya, and Zika virus infection.
Most cases are mild, but meningitis and encephalitis can occur as well as possible fetal death and deformities after infection in pregnancy. Last month, the first fatal cases were reported in two young Brazilian women who, concerningly, had no comorbidities.
This news organization asked Jan Felix Drexler, MD, of the Institute of Virology at Charité – Universitätsmedizin in Berlin, Germany, who has studied the emergence of Oropouche fever in Latin America, what clinicians should know about OROV disease.
What are the main symptoms of OROV disease for which clinicians should be alert?
The main symptoms are not different from other arboviral infections, ie, fever, maybe joint and muscle pain, maybe rash. The problem is that we do not know how often severe disease may occur because we do not know whether the severe cases that have been postulated, including death in apparently healthy people and congenital infection, are due to increased testing; an altered virus; or an altered, more intense circulation (so that many more infections simply lead to rare severe cases appearing). Be alert and ask for testing in your patients.
What is the differential diagnosis if a recent traveler to affected regions presents with symptoms? Are there any clues to suggest whether the disease is Oropouche as opposed to Zika, etc.?
The main message is: Do not assume a particular infection based on clinical symptoms. If your patient is returning from or living in an endemic area, consider OROV disease in the differential diagnosis.
What personal protective measures should clinicians advise travelers in affected areas to take? Do these differ from normal mosquito precautions?
Repellents are extremely important as usual. However, there are differences. Mosquito nets’ hole sizes need to be smaller than those used against the vectors of malaria or dengue; in other words, they need to have a higher mesh. The problem is that nets with high mesh are complicated in very hot and humid conditions because they also limit ventilation. Travelers should discuss with local suppliers about the best trade-off.
The risk for midge bites is likely highest at dawn and dusk in still and humid conditions. So on the one hand, one could recommend avoiding those areas and being outside during those times of the day. On the other hand, specific recommendations cannot be made robustly because we cannot exclude other invertebrate vectors at current knowledge. Some studies have implicated that mosquitoes may also transmit the virus. If that holds true, then we are back to reducing any bite.
Should pregnant women be advised to avoid travel to affected regions?
Not immediately, but caution must be taken. We simply do not have sufficient data to gauge the risk for potential congenital infection. Much more epidemiologic data and controlled infection experiments will be required to make evidence-based recommendations.
All the cases reported in Europe so far were imported from Cuba and Brazil. Is there any risk for local transmission, eg, via midges/mosquitoes that might hitch a ride on an aircraft, as in cases of airport malaria?
Not immediately, but it cannot be excluded. We know very little about the infection intensity in the vectors. Controlled infection experiments, including robustness of vectors against commonly used insecticides in airplanes, need to be done.
What is the risk for an animal reservoir emerging in Europe?
We do not know, but there is also no reason for ringing the alarm bells. Controlled infection experiments and surveillance will be required.
Is treatment purely supportive or are there any specific agents worth trying in case of severe symptoms/neurologic involvement?
No specific treatment can be recommended as is. However, severe dengue illustrates the relevance of supportive treatment, which is hugely effective in reducing mortality.
The Lancet paper states: “Several laboratory tests have been developed but robust commercial tests are hardly available.” How likely is it that laboratories in Europe will have the capability to test for the Oropouche organism?
European laboratory networks have already taken action, and testing is now available at least in the major and reference laboratories. If a clinician asks for OROV testing, they will probably get a robust answer in a reasonable timespan. Of course, that can be improved once we have more cases and more laboratories will be equipped for testing.
Is there anything else you think clinicians should be aware of?
The most important is to think beyond the textbooks we know from medical school. Things change rapidly in a connected world under altered climate conditions.
Dr. Drexler has no conflicts of interest to declare.
A version of this article first appeared on Medscape.com.
What Would ‘Project 2025’ Mean for Health and Healthcare?
The Heritage Foundation sponsored and developed Project 2025 for the explicit, stated purpose of building a conservative victory through policy, personnel, and training with a 180-day game plan after a sympathetic new President of the United States takes office. To date, Project 2025 has not been formally endorsed by any presidential campaign.
Chapter 14 of the “Mandate for Leadership” is an exhaustive proposed overhaul of the Department of Health and Human Services (HHS), one of the major existing arms of the executive branch of the US government.
The mandate’s sweeping recommendations, if implemented, would impact the lives of all Americans and all healthcare workers, as outlined in the following excerpts.
Healthcare-Related Excerpts From Project 2025
- “From the moment of conception, every human being possesses inherent dignity and worth, and our humanity does not depend on our age, stage of development, race, or abilities. The Secretary must ensure that all HHS programs and activities are rooted in a deep respect for innocent human life from day one until natural death: Abortion and euthanasia are not health care.”
- “Unfortunately, family policies and programs under President Biden’s HHS are fraught with agenda items focusing on ‘LGBTQ+ equity,’ subsidizing single motherhood, disincentivizing work, and penalizing marriage. These policies should be repealed and replaced by policies that support the formation of stable, married, nuclear families.”
- “The next Administration should guard against the regulatory capture of our public health agencies by pharmaceutical companies, insurers, hospital conglomerates, and related economic interests that these agencies are meant to regulate. We must erect robust firewalls to mitigate these obvious financial conflicts of interest.”
- “All National Institutes of Health, Centers for Disease Control and Prevention, and Food and Drug Administration regulators should be entirely free from private biopharmaceutical funding. In this realm, ‘public–private partnerships’ is a euphemism for agency capture, a thin veneer for corporatism. Funding for agencies and individual government researchers must come directly from the government with robust congressional oversight.”
- “The CDC [Centers for Disease Control and Prevention] operates several programs related to vaccine safety including the Vaccine Adverse Event Reporting System (VAERS); Vaccine Safety Datalink (VSD); and Clinical Immunization Safety Assessment (CISA) Project. Those functions and their associated funding should be transferred to the FDA [Food and Drug Administration], which is responsible for post-market surveillance and evaluation of all other drugs and biological products.”
- “Because liberal states have now become sanctuaries for abortion tourism, HHS should use every available tool, including the cutting of funds, to ensure that every state reports exactly how many abortions take place within its borders, at what gestational age of the child, for what reason, the mother’s state of residence, and by what method. It should also ensure that statistics are separated by category: spontaneous miscarriage; treatments that incidentally result in the death of a child (such as chemotherapy); stillbirths; and induced abortion. In addition, CDC should require monitoring and reporting for complications due to abortion and every instance of children being born alive after an abortion.”
- “The CDC should immediately end its collection of data on gender identity, which legitimizes the unscientific notion that men can become women (and vice versa) and encourages the phenomenon of ever-multiplying subjective identities.”
- “A test developed by a lab in accordance with the protocols developed by another lab (non-commercial sharing) currently constitutes a ‘new’ laboratory-developed test because the lab in which it will be used is different from the initial developing lab. To encourage interlaboratory collaboration and discourage duplicative test creation (and associated regulatory and logistical burdens), the FDA should introduce mechanisms through which laboratory-developed tests can easily be shared with other laboratories without the current regulatory burdens.”
- “[FDA should] Reverse its approval of chemical abortion drugs because the politicized approval process was illegal from the start. The FDA failed to abide by its legal obligations to protect the health, safety, and welfare of girls and women.”
- “[FDA should] Stop promoting or approving mail-order abortions in violation of long-standing federal laws that prohibit the mailing and interstate carriage of abortion drugs.”
- “[HHS should] Promptly restore the ethics advisory committee to oversee abortion-derived fetal tissue research, and Congress should prohibit such research altogether.”
- “[HHS should] End intramural research projects using tissue from aborted children within the NIH, which should end its human embryonic stem cell registry.”
- “Under Francis Collins, NIH became so focused on the #MeToo movement that it refused to sponsor scientific conferences unless there were a certain number of women panelists, which violates federal civil rights law against sex discrimination. This quota practice should be ended, and the NIH Office of Equity, Diversity, and Inclusion, which pushes such unlawful actions, should be abolished.”
- “Make Medicare Advantage [MA] the default enrollment option.”
- “[Legislation reforming legacy (non-MA) Medicare should] Repeal harmful health policies enacted under the Obama and Biden Administrations such as the Medicare Shared Savings Program and Inflation Reduction Act.”
- “…the next Administration should] Add work requirements and match Medicaid benefits to beneficiary needs. Because Medicaid serves a broad and diverse group of individuals, it should be flexible enough to accommodate different designs for different groups.”
- “The No Surprises Act should scrap the dispute resolution process in favor of a truth-in-advertising approach that will protect consumers and free doctors, insurers, and arbiters from confused and conflicting standards for resolving disputes that the disputing parties can best resolve themselves.”
- “Prohibit abortion travel funding. Providing funding for abortions increases the number of abortions and violates the conscience and religious freedom rights of Americans who object to subsidizing the taking of life.”
- “Prohibit Planned Parenthood from receiving Medicaid funds. During the 2020–2021 reporting period, Planned Parenthood performed more than 383,000 abortions.”
- “Protect faith-based grant recipients from religious liberty violations and maintain a biblically based, social science–reinforced definition of marriage and family. Social science reports that assess the objective outcomes for children raised in homes aside from a heterosexual, intact marriage are clear.”
- “Allocate funding to strategy programs promoting father involvement or terminate parental rights quickly.”
- “Eliminate the Head Start program.”
- “Support palliative care. Physician-assisted suicide (PAS) is legal in 10 states and the District of Columbia. Legalizing PAS is a grave mistake that endangers the weak and vulnerable, corrupts the practice of medicine and the doctor–patient relationship, compromises the family and intergenerational commitments, and betrays human dignity and equality before the law.”
- “Eliminate men’s preventive services from the women’s preventive services mandate. In December 2021, HRSA [Health Resources and Services Administration] updated its women’s preventive services guidelines to include male condoms.”
- “Prioritize funding for home-based childcare, not universal day care.”
- “ The Office of the Secretary should eliminate the HHS Reproductive Healthcare Access Task Force and install a pro-life task force to ensure that all of the department’s divisions seek to use their authority to promote the life and health of women and their unborn children.”
- “The ASH [Assistant Secretary for Health] and SG [Surgeon General] positions should be combined into one four-star position with the rank, responsibilities, and authority of the ASH retained but with the title of Surgeon General.”
- “OCR [Office for Civil Rights] should withdraw its Health Insurance Portability and Accountability Act (HIPAA) guidance on abortion.”
Dr. Lundberg is Editor in Chief, Cancer Commons, and has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The Heritage Foundation sponsored and developed Project 2025 for the explicit, stated purpose of building a conservative victory through policy, personnel, and training with a 180-day game plan after a sympathetic new President of the United States takes office. To date, Project 2025 has not been formally endorsed by any presidential campaign.
Chapter 14 of the “Mandate for Leadership” is an exhaustive proposed overhaul of the Department of Health and Human Services (HHS), one of the major existing arms of the executive branch of the US government.
The mandate’s sweeping recommendations, if implemented, would impact the lives of all Americans and all healthcare workers, as outlined in the following excerpts.
Healthcare-Related Excerpts From Project 2025
- “From the moment of conception, every human being possesses inherent dignity and worth, and our humanity does not depend on our age, stage of development, race, or abilities. The Secretary must ensure that all HHS programs and activities are rooted in a deep respect for innocent human life from day one until natural death: Abortion and euthanasia are not health care.”
- “Unfortunately, family policies and programs under President Biden’s HHS are fraught with agenda items focusing on ‘LGBTQ+ equity,’ subsidizing single motherhood, disincentivizing work, and penalizing marriage. These policies should be repealed and replaced by policies that support the formation of stable, married, nuclear families.”
- “The next Administration should guard against the regulatory capture of our public health agencies by pharmaceutical companies, insurers, hospital conglomerates, and related economic interests that these agencies are meant to regulate. We must erect robust firewalls to mitigate these obvious financial conflicts of interest.”
- “All National Institutes of Health, Centers for Disease Control and Prevention, and Food and Drug Administration regulators should be entirely free from private biopharmaceutical funding. In this realm, ‘public–private partnerships’ is a euphemism for agency capture, a thin veneer for corporatism. Funding for agencies and individual government researchers must come directly from the government with robust congressional oversight.”
- “The CDC [Centers for Disease Control and Prevention] operates several programs related to vaccine safety including the Vaccine Adverse Event Reporting System (VAERS); Vaccine Safety Datalink (VSD); and Clinical Immunization Safety Assessment (CISA) Project. Those functions and their associated funding should be transferred to the FDA [Food and Drug Administration], which is responsible for post-market surveillance and evaluation of all other drugs and biological products.”
- “Because liberal states have now become sanctuaries for abortion tourism, HHS should use every available tool, including the cutting of funds, to ensure that every state reports exactly how many abortions take place within its borders, at what gestational age of the child, for what reason, the mother’s state of residence, and by what method. It should also ensure that statistics are separated by category: spontaneous miscarriage; treatments that incidentally result in the death of a child (such as chemotherapy); stillbirths; and induced abortion. In addition, CDC should require monitoring and reporting for complications due to abortion and every instance of children being born alive after an abortion.”
- “The CDC should immediately end its collection of data on gender identity, which legitimizes the unscientific notion that men can become women (and vice versa) and encourages the phenomenon of ever-multiplying subjective identities.”
- “A test developed by a lab in accordance with the protocols developed by another lab (non-commercial sharing) currently constitutes a ‘new’ laboratory-developed test because the lab in which it will be used is different from the initial developing lab. To encourage interlaboratory collaboration and discourage duplicative test creation (and associated regulatory and logistical burdens), the FDA should introduce mechanisms through which laboratory-developed tests can easily be shared with other laboratories without the current regulatory burdens.”
- “[FDA should] Reverse its approval of chemical abortion drugs because the politicized approval process was illegal from the start. The FDA failed to abide by its legal obligations to protect the health, safety, and welfare of girls and women.”
- “[FDA should] Stop promoting or approving mail-order abortions in violation of long-standing federal laws that prohibit the mailing and interstate carriage of abortion drugs.”
- “[HHS should] Promptly restore the ethics advisory committee to oversee abortion-derived fetal tissue research, and Congress should prohibit such research altogether.”
- “[HHS should] End intramural research projects using tissue from aborted children within the NIH, which should end its human embryonic stem cell registry.”
- “Under Francis Collins, NIH became so focused on the #MeToo movement that it refused to sponsor scientific conferences unless there were a certain number of women panelists, which violates federal civil rights law against sex discrimination. This quota practice should be ended, and the NIH Office of Equity, Diversity, and Inclusion, which pushes such unlawful actions, should be abolished.”
- “Make Medicare Advantage [MA] the default enrollment option.”
- “[Legislation reforming legacy (non-MA) Medicare should] Repeal harmful health policies enacted under the Obama and Biden Administrations such as the Medicare Shared Savings Program and Inflation Reduction Act.”
- “…the next Administration should] Add work requirements and match Medicaid benefits to beneficiary needs. Because Medicaid serves a broad and diverse group of individuals, it should be flexible enough to accommodate different designs for different groups.”
- “The No Surprises Act should scrap the dispute resolution process in favor of a truth-in-advertising approach that will protect consumers and free doctors, insurers, and arbiters from confused and conflicting standards for resolving disputes that the disputing parties can best resolve themselves.”
- “Prohibit abortion travel funding. Providing funding for abortions increases the number of abortions and violates the conscience and religious freedom rights of Americans who object to subsidizing the taking of life.”
- “Prohibit Planned Parenthood from receiving Medicaid funds. During the 2020–2021 reporting period, Planned Parenthood performed more than 383,000 abortions.”
- “Protect faith-based grant recipients from religious liberty violations and maintain a biblically based, social science–reinforced definition of marriage and family. Social science reports that assess the objective outcomes for children raised in homes aside from a heterosexual, intact marriage are clear.”
- “Allocate funding to strategy programs promoting father involvement or terminate parental rights quickly.”
- “Eliminate the Head Start program.”
- “Support palliative care. Physician-assisted suicide (PAS) is legal in 10 states and the District of Columbia. Legalizing PAS is a grave mistake that endangers the weak and vulnerable, corrupts the practice of medicine and the doctor–patient relationship, compromises the family and intergenerational commitments, and betrays human dignity and equality before the law.”
- “Eliminate men’s preventive services from the women’s preventive services mandate. In December 2021, HRSA [Health Resources and Services Administration] updated its women’s preventive services guidelines to include male condoms.”
- “Prioritize funding for home-based childcare, not universal day care.”
- “ The Office of the Secretary should eliminate the HHS Reproductive Healthcare Access Task Force and install a pro-life task force to ensure that all of the department’s divisions seek to use their authority to promote the life and health of women and their unborn children.”
- “The ASH [Assistant Secretary for Health] and SG [Surgeon General] positions should be combined into one four-star position with the rank, responsibilities, and authority of the ASH retained but with the title of Surgeon General.”
- “OCR [Office for Civil Rights] should withdraw its Health Insurance Portability and Accountability Act (HIPAA) guidance on abortion.”
Dr. Lundberg is Editor in Chief, Cancer Commons, and has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The Heritage Foundation sponsored and developed Project 2025 for the explicit, stated purpose of building a conservative victory through policy, personnel, and training with a 180-day game plan after a sympathetic new President of the United States takes office. To date, Project 2025 has not been formally endorsed by any presidential campaign.
Chapter 14 of the “Mandate for Leadership” is an exhaustive proposed overhaul of the Department of Health and Human Services (HHS), one of the major existing arms of the executive branch of the US government.
The mandate’s sweeping recommendations, if implemented, would impact the lives of all Americans and all healthcare workers, as outlined in the following excerpts.
Healthcare-Related Excerpts From Project 2025
- “From the moment of conception, every human being possesses inherent dignity and worth, and our humanity does not depend on our age, stage of development, race, or abilities. The Secretary must ensure that all HHS programs and activities are rooted in a deep respect for innocent human life from day one until natural death: Abortion and euthanasia are not health care.”
- “Unfortunately, family policies and programs under President Biden’s HHS are fraught with agenda items focusing on ‘LGBTQ+ equity,’ subsidizing single motherhood, disincentivizing work, and penalizing marriage. These policies should be repealed and replaced by policies that support the formation of stable, married, nuclear families.”
- “The next Administration should guard against the regulatory capture of our public health agencies by pharmaceutical companies, insurers, hospital conglomerates, and related economic interests that these agencies are meant to regulate. We must erect robust firewalls to mitigate these obvious financial conflicts of interest.”
- “All National Institutes of Health, Centers for Disease Control and Prevention, and Food and Drug Administration regulators should be entirely free from private biopharmaceutical funding. In this realm, ‘public–private partnerships’ is a euphemism for agency capture, a thin veneer for corporatism. Funding for agencies and individual government researchers must come directly from the government with robust congressional oversight.”
- “The CDC [Centers for Disease Control and Prevention] operates several programs related to vaccine safety including the Vaccine Adverse Event Reporting System (VAERS); Vaccine Safety Datalink (VSD); and Clinical Immunization Safety Assessment (CISA) Project. Those functions and their associated funding should be transferred to the FDA [Food and Drug Administration], which is responsible for post-market surveillance and evaluation of all other drugs and biological products.”
- “Because liberal states have now become sanctuaries for abortion tourism, HHS should use every available tool, including the cutting of funds, to ensure that every state reports exactly how many abortions take place within its borders, at what gestational age of the child, for what reason, the mother’s state of residence, and by what method. It should also ensure that statistics are separated by category: spontaneous miscarriage; treatments that incidentally result in the death of a child (such as chemotherapy); stillbirths; and induced abortion. In addition, CDC should require monitoring and reporting for complications due to abortion and every instance of children being born alive after an abortion.”
- “The CDC should immediately end its collection of data on gender identity, which legitimizes the unscientific notion that men can become women (and vice versa) and encourages the phenomenon of ever-multiplying subjective identities.”
- “A test developed by a lab in accordance with the protocols developed by another lab (non-commercial sharing) currently constitutes a ‘new’ laboratory-developed test because the lab in which it will be used is different from the initial developing lab. To encourage interlaboratory collaboration and discourage duplicative test creation (and associated regulatory and logistical burdens), the FDA should introduce mechanisms through which laboratory-developed tests can easily be shared with other laboratories without the current regulatory burdens.”
- “[FDA should] Reverse its approval of chemical abortion drugs because the politicized approval process was illegal from the start. The FDA failed to abide by its legal obligations to protect the health, safety, and welfare of girls and women.”
- “[FDA should] Stop promoting or approving mail-order abortions in violation of long-standing federal laws that prohibit the mailing and interstate carriage of abortion drugs.”
- “[HHS should] Promptly restore the ethics advisory committee to oversee abortion-derived fetal tissue research, and Congress should prohibit such research altogether.”
- “[HHS should] End intramural research projects using tissue from aborted children within the NIH, which should end its human embryonic stem cell registry.”
- “Under Francis Collins, NIH became so focused on the #MeToo movement that it refused to sponsor scientific conferences unless there were a certain number of women panelists, which violates federal civil rights law against sex discrimination. This quota practice should be ended, and the NIH Office of Equity, Diversity, and Inclusion, which pushes such unlawful actions, should be abolished.”
- “Make Medicare Advantage [MA] the default enrollment option.”
- “[Legislation reforming legacy (non-MA) Medicare should] Repeal harmful health policies enacted under the Obama and Biden Administrations such as the Medicare Shared Savings Program and Inflation Reduction Act.”
- “…the next Administration should] Add work requirements and match Medicaid benefits to beneficiary needs. Because Medicaid serves a broad and diverse group of individuals, it should be flexible enough to accommodate different designs for different groups.”
- “The No Surprises Act should scrap the dispute resolution process in favor of a truth-in-advertising approach that will protect consumers and free doctors, insurers, and arbiters from confused and conflicting standards for resolving disputes that the disputing parties can best resolve themselves.”
- “Prohibit abortion travel funding. Providing funding for abortions increases the number of abortions and violates the conscience and religious freedom rights of Americans who object to subsidizing the taking of life.”
- “Prohibit Planned Parenthood from receiving Medicaid funds. During the 2020–2021 reporting period, Planned Parenthood performed more than 383,000 abortions.”
- “Protect faith-based grant recipients from religious liberty violations and maintain a biblically based, social science–reinforced definition of marriage and family. Social science reports that assess the objective outcomes for children raised in homes aside from a heterosexual, intact marriage are clear.”
- “Allocate funding to strategy programs promoting father involvement or terminate parental rights quickly.”
- “Eliminate the Head Start program.”
- “Support palliative care. Physician-assisted suicide (PAS) is legal in 10 states and the District of Columbia. Legalizing PAS is a grave mistake that endangers the weak and vulnerable, corrupts the practice of medicine and the doctor–patient relationship, compromises the family and intergenerational commitments, and betrays human dignity and equality before the law.”
- “Eliminate men’s preventive services from the women’s preventive services mandate. In December 2021, HRSA [Health Resources and Services Administration] updated its women’s preventive services guidelines to include male condoms.”
- “Prioritize funding for home-based childcare, not universal day care.”
- “ The Office of the Secretary should eliminate the HHS Reproductive Healthcare Access Task Force and install a pro-life task force to ensure that all of the department’s divisions seek to use their authority to promote the life and health of women and their unborn children.”
- “The ASH [Assistant Secretary for Health] and SG [Surgeon General] positions should be combined into one four-star position with the rank, responsibilities, and authority of the ASH retained but with the title of Surgeon General.”
- “OCR [Office for Civil Rights] should withdraw its Health Insurance Portability and Accountability Act (HIPAA) guidance on abortion.”
Dr. Lundberg is Editor in Chief, Cancer Commons, and has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New Biological Pathway May Explain BPA Exposure, Autism Link
BPA is a potent endocrine disruptor found in polycarbonate plastics and epoxy resins and has been banned by the Food and Drug Administration for use in baby bottles, sippy cups, and infant formula packaging.
“Exposure to BPA has already been shown in some studies to be associated with subsequent autism in offspring,” lead researcher Anne-Louise Ponsonby, PhD, The Florey Institute, Heidelberg, Australia, said in a statement.
“Our work is important because it demonstrates one of the biological mechanisms potentially involved. BPA can disrupt hormone-controlled male fetal brain development in several ways, including silencing a key enzyme, aromatase, that controls neurohormones and is especially important in fetal male brain development. This appears to be part of the autism puzzle,” she said.
Brain aromatase, encoded by CYP19A1, converts neural androgens to neural estrogens and has been implicated in ASD. Postmortem analyses of men with ASD also show markedly reduced aromatase activity.
The findings were published online in Nature Communications.
New Biological Mechanism
For the study, the researchers analyzed data from the Barwon Infant Study in 1067 infants in Australia. At age 7-11 years, 43 children had a confirmed ASD diagnosis, and 249 infants with Child Behavior Checklist (CBCL) data at age 2 years had an autism spectrum problem score above the median.
The researchers developed a CYP19A1 genetic score for aromatase activity based on five single nucleotide polymorphisms associated with lower estrogen levels. Among 595 children with prenatal BPA and CBCL, those with three or more variants were classified as “low aromatase activity” and the remaining were classified as “high.”
In regression analyses, boys with low aromatase activity and high prenatal BPA exposure (top quartile > 2.18 µg/L) were 3.5 times more likely to have autism symptoms at age 2 years (odds ratio [OR], 3.56; 95% CI, 1.13-11.22).
The odds of a confirmed ASD diagnosis were six times higher at age 9 years only in men with low aromatase activity (OR, 6.24; 95% CI, 1.02-38.26).
The researchers also found that higher BPA levels predicted higher methylation in cord blood across the CYP19A1 brain promoter PI.f region (P = .009).
To replicate the findings, data were used from the Columbia Centre for Children’s Health Study–Mothers and Newborns cohort in the United States. Once again, the BPA level was associated with hypermethylation of the aromatase brain promoter PI.f (P = .0089).
In both cohorts, there was evidence that the effect of increased BPA on brain-derived neurotrophic factor hypermethylation was mediated partly through higher aromatase gene methylation (P = .001).
To validate the findings, the researchers examined human neuroblastoma SH-SY5Y cell lines and found aromatase protein levels were more than halved in the presence of BPA 50 µg/L (P = .01).
Additionally, mouse studies showed that male mice exposed to BPA 50 µg/L mid-gestation and male aromatase knockout mice — but not female mice — had social behavior deficits, such as interacting with a strange mouse, as well as structural and functional brain changes.
“We found that BPA suppresses the aromatase enzyme and is associated with anatomical, neurologic, and behavioral changes in the male mice that may be consistent with autism spectrum disorder,” Wah Chin Boon, PhD, co–lead researcher and research fellow, also with The Florey Institute, said in a statement.
“This is the first time a biological pathway has been identified that might help explain the connection between autism and BPA,” she said.
“In this study, not only were the levels of BPA higher than most people would be exposed to, but in at least one of the experiments the mice were injected with BPA directly, whereas humans would be exposed via food and drink,” observed Oliver Jones, PhD, MSc, professor of chemistry, RMIT University, Melbourne, Australia. “If you ingest the food, it undergoes metabolism before it gets to the bloodstream, which reduces the effective dose.”
Dr. Jones said further studies with larger numbers of participants measuring BPA throughout pregnancy and other chemicals the mother and child were exposed to are needed to be sure of any such link. “Just because there is a possible mechanism in place does not automatically mean that it is activated,” he said.
Dr. Ponsonby pointed out that BPA and other endocrine-disrupting chemicals are “almost impossible for individuals to avoid” and can enter the body through plastic food and drink packaging, home renovation fumes, and sources such as cosmetics.
Fatty Acid Helpful?
Building on earlier observations that 10-hydroxy-2-decenoic acid (10HDA) may have estrogenic modulating activities, the researchers conducted additional studies suggesting that 10HDA may be effective as a competitive ligand that could counteract the effects of BPA on estrogen signaling within cells.
Further, among 3-week-old mice pups prenatally exposed to BPA, daily injections of 10HDA for 3 weeks showed striking and significant improvements in social interaction. Stopping 10HDA resulted in a deficit in social interaction that was again ameliorated by subsequent 10HDA treatment.
“10-hydroxy-2-decenoic acid shows early indications of potential in activating opposing biological pathways to improve autism-like characteristics when administered to animals that have been prenatally exposed to BPA,” Dr. Boon said. “It warrants further studies to see whether this potential treatment could be realized in humans.”
Reached for comment, Dr. Jones said “the human studies are not strong at all,” in large part because BPA levels were tested only once at 36 weeks in the BIS cohort.
“I would argue that if BPA is in the urine, it has been excreted and is no longer in the bloodstream, thus not able to affect the child,” he said. “I’d also argue that a single measurement at 36 weeks cannot give you any idea of the mother’s exposure to BPA over the rest of the pregnancy or what the child was exposed to after birth.”
The study was funded by the Minderoo Foundation, the National Health and Medical Research Council of Australia, the Australian Research Council, and numerous other sponsors. Dr. Boon is a coinventor on “Methods of treating neurodevelopmental diseases and disorders” and is a board member of Meizon Innovation Holdings. Dr. Ponsonby is a scientific adviser to Meizon Innovation Holdings. The remaining authors declared no competing interests.
A version of this article first appeared on Medscape.com.
BPA is a potent endocrine disruptor found in polycarbonate plastics and epoxy resins and has been banned by the Food and Drug Administration for use in baby bottles, sippy cups, and infant formula packaging.
“Exposure to BPA has already been shown in some studies to be associated with subsequent autism in offspring,” lead researcher Anne-Louise Ponsonby, PhD, The Florey Institute, Heidelberg, Australia, said in a statement.
“Our work is important because it demonstrates one of the biological mechanisms potentially involved. BPA can disrupt hormone-controlled male fetal brain development in several ways, including silencing a key enzyme, aromatase, that controls neurohormones and is especially important in fetal male brain development. This appears to be part of the autism puzzle,” she said.
Brain aromatase, encoded by CYP19A1, converts neural androgens to neural estrogens and has been implicated in ASD. Postmortem analyses of men with ASD also show markedly reduced aromatase activity.
The findings were published online in Nature Communications.
New Biological Mechanism
For the study, the researchers analyzed data from the Barwon Infant Study in 1067 infants in Australia. At age 7-11 years, 43 children had a confirmed ASD diagnosis, and 249 infants with Child Behavior Checklist (CBCL) data at age 2 years had an autism spectrum problem score above the median.
The researchers developed a CYP19A1 genetic score for aromatase activity based on five single nucleotide polymorphisms associated with lower estrogen levels. Among 595 children with prenatal BPA and CBCL, those with three or more variants were classified as “low aromatase activity” and the remaining were classified as “high.”
In regression analyses, boys with low aromatase activity and high prenatal BPA exposure (top quartile > 2.18 µg/L) were 3.5 times more likely to have autism symptoms at age 2 years (odds ratio [OR], 3.56; 95% CI, 1.13-11.22).
The odds of a confirmed ASD diagnosis were six times higher at age 9 years only in men with low aromatase activity (OR, 6.24; 95% CI, 1.02-38.26).
The researchers also found that higher BPA levels predicted higher methylation in cord blood across the CYP19A1 brain promoter PI.f region (P = .009).
To replicate the findings, data were used from the Columbia Centre for Children’s Health Study–Mothers and Newborns cohort in the United States. Once again, the BPA level was associated with hypermethylation of the aromatase brain promoter PI.f (P = .0089).
In both cohorts, there was evidence that the effect of increased BPA on brain-derived neurotrophic factor hypermethylation was mediated partly through higher aromatase gene methylation (P = .001).
To validate the findings, the researchers examined human neuroblastoma SH-SY5Y cell lines and found aromatase protein levels were more than halved in the presence of BPA 50 µg/L (P = .01).
Additionally, mouse studies showed that male mice exposed to BPA 50 µg/L mid-gestation and male aromatase knockout mice — but not female mice — had social behavior deficits, such as interacting with a strange mouse, as well as structural and functional brain changes.
“We found that BPA suppresses the aromatase enzyme and is associated with anatomical, neurologic, and behavioral changes in the male mice that may be consistent with autism spectrum disorder,” Wah Chin Boon, PhD, co–lead researcher and research fellow, also with The Florey Institute, said in a statement.
“This is the first time a biological pathway has been identified that might help explain the connection between autism and BPA,” she said.
“In this study, not only were the levels of BPA higher than most people would be exposed to, but in at least one of the experiments the mice were injected with BPA directly, whereas humans would be exposed via food and drink,” observed Oliver Jones, PhD, MSc, professor of chemistry, RMIT University, Melbourne, Australia. “If you ingest the food, it undergoes metabolism before it gets to the bloodstream, which reduces the effective dose.”
Dr. Jones said further studies with larger numbers of participants measuring BPA throughout pregnancy and other chemicals the mother and child were exposed to are needed to be sure of any such link. “Just because there is a possible mechanism in place does not automatically mean that it is activated,” he said.
Dr. Ponsonby pointed out that BPA and other endocrine-disrupting chemicals are “almost impossible for individuals to avoid” and can enter the body through plastic food and drink packaging, home renovation fumes, and sources such as cosmetics.
Fatty Acid Helpful?
Building on earlier observations that 10-hydroxy-2-decenoic acid (10HDA) may have estrogenic modulating activities, the researchers conducted additional studies suggesting that 10HDA may be effective as a competitive ligand that could counteract the effects of BPA on estrogen signaling within cells.
Further, among 3-week-old mice pups prenatally exposed to BPA, daily injections of 10HDA for 3 weeks showed striking and significant improvements in social interaction. Stopping 10HDA resulted in a deficit in social interaction that was again ameliorated by subsequent 10HDA treatment.
“10-hydroxy-2-decenoic acid shows early indications of potential in activating opposing biological pathways to improve autism-like characteristics when administered to animals that have been prenatally exposed to BPA,” Dr. Boon said. “It warrants further studies to see whether this potential treatment could be realized in humans.”
Reached for comment, Dr. Jones said “the human studies are not strong at all,” in large part because BPA levels were tested only once at 36 weeks in the BIS cohort.
“I would argue that if BPA is in the urine, it has been excreted and is no longer in the bloodstream, thus not able to affect the child,” he said. “I’d also argue that a single measurement at 36 weeks cannot give you any idea of the mother’s exposure to BPA over the rest of the pregnancy or what the child was exposed to after birth.”
The study was funded by the Minderoo Foundation, the National Health and Medical Research Council of Australia, the Australian Research Council, and numerous other sponsors. Dr. Boon is a coinventor on “Methods of treating neurodevelopmental diseases and disorders” and is a board member of Meizon Innovation Holdings. Dr. Ponsonby is a scientific adviser to Meizon Innovation Holdings. The remaining authors declared no competing interests.
A version of this article first appeared on Medscape.com.
BPA is a potent endocrine disruptor found in polycarbonate plastics and epoxy resins and has been banned by the Food and Drug Administration for use in baby bottles, sippy cups, and infant formula packaging.
“Exposure to BPA has already been shown in some studies to be associated with subsequent autism in offspring,” lead researcher Anne-Louise Ponsonby, PhD, The Florey Institute, Heidelberg, Australia, said in a statement.
“Our work is important because it demonstrates one of the biological mechanisms potentially involved. BPA can disrupt hormone-controlled male fetal brain development in several ways, including silencing a key enzyme, aromatase, that controls neurohormones and is especially important in fetal male brain development. This appears to be part of the autism puzzle,” she said.
Brain aromatase, encoded by CYP19A1, converts neural androgens to neural estrogens and has been implicated in ASD. Postmortem analyses of men with ASD also show markedly reduced aromatase activity.
The findings were published online in Nature Communications.
New Biological Mechanism
For the study, the researchers analyzed data from the Barwon Infant Study in 1067 infants in Australia. At age 7-11 years, 43 children had a confirmed ASD diagnosis, and 249 infants with Child Behavior Checklist (CBCL) data at age 2 years had an autism spectrum problem score above the median.
The researchers developed a CYP19A1 genetic score for aromatase activity based on five single nucleotide polymorphisms associated with lower estrogen levels. Among 595 children with prenatal BPA and CBCL, those with three or more variants were classified as “low aromatase activity” and the remaining were classified as “high.”
In regression analyses, boys with low aromatase activity and high prenatal BPA exposure (top quartile > 2.18 µg/L) were 3.5 times more likely to have autism symptoms at age 2 years (odds ratio [OR], 3.56; 95% CI, 1.13-11.22).
The odds of a confirmed ASD diagnosis were six times higher at age 9 years only in men with low aromatase activity (OR, 6.24; 95% CI, 1.02-38.26).
The researchers also found that higher BPA levels predicted higher methylation in cord blood across the CYP19A1 brain promoter PI.f region (P = .009).
To replicate the findings, data were used from the Columbia Centre for Children’s Health Study–Mothers and Newborns cohort in the United States. Once again, the BPA level was associated with hypermethylation of the aromatase brain promoter PI.f (P = .0089).
In both cohorts, there was evidence that the effect of increased BPA on brain-derived neurotrophic factor hypermethylation was mediated partly through higher aromatase gene methylation (P = .001).
To validate the findings, the researchers examined human neuroblastoma SH-SY5Y cell lines and found aromatase protein levels were more than halved in the presence of BPA 50 µg/L (P = .01).
Additionally, mouse studies showed that male mice exposed to BPA 50 µg/L mid-gestation and male aromatase knockout mice — but not female mice — had social behavior deficits, such as interacting with a strange mouse, as well as structural and functional brain changes.
“We found that BPA suppresses the aromatase enzyme and is associated with anatomical, neurologic, and behavioral changes in the male mice that may be consistent with autism spectrum disorder,” Wah Chin Boon, PhD, co–lead researcher and research fellow, also with The Florey Institute, said in a statement.
“This is the first time a biological pathway has been identified that might help explain the connection between autism and BPA,” she said.
“In this study, not only were the levels of BPA higher than most people would be exposed to, but in at least one of the experiments the mice were injected with BPA directly, whereas humans would be exposed via food and drink,” observed Oliver Jones, PhD, MSc, professor of chemistry, RMIT University, Melbourne, Australia. “If you ingest the food, it undergoes metabolism before it gets to the bloodstream, which reduces the effective dose.”
Dr. Jones said further studies with larger numbers of participants measuring BPA throughout pregnancy and other chemicals the mother and child were exposed to are needed to be sure of any such link. “Just because there is a possible mechanism in place does not automatically mean that it is activated,” he said.
Dr. Ponsonby pointed out that BPA and other endocrine-disrupting chemicals are “almost impossible for individuals to avoid” and can enter the body through plastic food and drink packaging, home renovation fumes, and sources such as cosmetics.
Fatty Acid Helpful?
Building on earlier observations that 10-hydroxy-2-decenoic acid (10HDA) may have estrogenic modulating activities, the researchers conducted additional studies suggesting that 10HDA may be effective as a competitive ligand that could counteract the effects of BPA on estrogen signaling within cells.
Further, among 3-week-old mice pups prenatally exposed to BPA, daily injections of 10HDA for 3 weeks showed striking and significant improvements in social interaction. Stopping 10HDA resulted in a deficit in social interaction that was again ameliorated by subsequent 10HDA treatment.
“10-hydroxy-2-decenoic acid shows early indications of potential in activating opposing biological pathways to improve autism-like characteristics when administered to animals that have been prenatally exposed to BPA,” Dr. Boon said. “It warrants further studies to see whether this potential treatment could be realized in humans.”
Reached for comment, Dr. Jones said “the human studies are not strong at all,” in large part because BPA levels were tested only once at 36 weeks in the BIS cohort.
“I would argue that if BPA is in the urine, it has been excreted and is no longer in the bloodstream, thus not able to affect the child,” he said. “I’d also argue that a single measurement at 36 weeks cannot give you any idea of the mother’s exposure to BPA over the rest of the pregnancy or what the child was exposed to after birth.”
The study was funded by the Minderoo Foundation, the National Health and Medical Research Council of Australia, the Australian Research Council, and numerous other sponsors. Dr. Boon is a coinventor on “Methods of treating neurodevelopmental diseases and disorders” and is a board member of Meizon Innovation Holdings. Dr. Ponsonby is a scientific adviser to Meizon Innovation Holdings. The remaining authors declared no competing interests.
A version of this article first appeared on Medscape.com.
FROM NATURE COMMUNICATIONS
How Common Is Pediatric Emergency Mistriage?
, according to a multicenter retrospective study published in JAMA Pediatrics. Researchers also identified gender, age, race, ethnicity, and comorbidity disparities in those who were undertriaged.
The researchers found that only 34.1% of visits were correctly triaged while 58.5% were overtriaged and 7.4% were undertriaged. The findings were based on analysis of more than 1 million pediatric emergency visits over a 5-year period that used the Emergency Severity Index (ESI) version 4 for triage.
“The ESI had poor sensitivity in identifying a critically ill pediatric patient, and undertriage occurred in 1 in 14 children,” wrote Dana R. Sax, MD, a senior emergency physician at The Permanente Medical Group in northern California, and her colleagues.

“More than 90% of pediatric visits were assigned a mid to low triage acuity category, and actual resource use and care intensity frequently did not align with ESI predictions,” the authors wrote. “Our findings highlight an opportunity to improve triage for pediatric patients to mitigate critical undertriage, optimize resource decisions, standardize processes across time and setting, and promote more equitable care.”
The authors added that the study findings are currently being used by the Permanente system “to develop standardized triage education across centers to improve early identification of high-risk patients.”
Disparities in Emergency Care
The results underscore the need for more work to address disparities in emergency care, wrote Warren D. Frankenberger, PhD, RN, a nurse scientist at Children’s Hospital of Philadelphia, and two colleagues in an accompanying editorial.
“Decisions in triage can have significant downstream effects on subsequent care during the ED visit,” they wrote in their editorial. “Given that the triage process in most instances is fully executed by nurses, nurse researchers are in a key position to evaluate these and other covariates to influence further improvements in triage.” They suggested that use of clinical decision support tools and artificial intelligence (AI) may improve the triage process, albeit with the caveat that AI often relies on models with pre-existing historical bias that may perpetuate structural inequalities.
Study Methodology
The researchers analyzed 1,016,816 pediatric visits at 21 emergency departments in Kaiser Permanente Northern California between January 2016 and December 2020. The patients were an average 7 years old, and 47% were female. The researchers excluded visits that lacked ESI data or had incomplete ED time variables as well as those with patients who left against medical advice, were not seen, or were transferred from another ED.
The study relied on novel definitions of ESI undertriage and overtriage developed through a modified Delphi process by a team of four emergency physicians, one pediatric emergency physician, two emergency nurses, and one pediatric ICU physician. The definition involved comparing ESI levels to the clinical outcomes and resource use.
Resources included laboratory analysis, electrocardiography, radiography, CT, MRI, diagnostic ultrasonography (not point of care), angiography, IV fluids, and IV, intramuscular, or nebulized medications. Resources did not include “oral medications, tetanus immunizations, point-of-care testing, history and physical examination, saline or heparin lock, prescription refills, simple wound care, crutches, splints, and slings.”
Level 1 events were those requiring time-sensitive, critical intervention, including high-risk sepsis. Level 2 events included most level 1 events that occurred after the first hour (except operating room admission or hospital transfer) as well as respiratory therapy, toxicology consult, lumbar puncture, suicidality as chief concern, at least 2 doses of albuterol or continuous albuterol nebulization, a skeletal survey x-ray order, and medical social work consult with an ED length of stay of at least 2 hours. Level 3 events included IV mediation order, any CT order, OR admission or hospital transfer after one hour, or any pediatric hospitalist consult.
Analyzing the ED Visits
Overtriaged cases were ESI level 1 or 2 cases in which fewer than 2 resources were used; level 3 cases where fewer than 2 resources were used and no level 1 or 2 events occurred; and level 4 cases where no resources were used.
Undertriaged cases were defined as the following:
- ESI level 5 cases where any resource was used and any level 1, 2, or 3 events occurred.
- Level 4 cases where more than 1 resource was used and any level 1, 2, or 3 events occurred.
- Level 3 cases where any level 1 event occurred, more than one level 2 event occurred, or any level 2 event occurred and more than one additional ED resource type was used.
- Level 2 cases where any level 1 event occurred.
About half the visits (51%) were assigned ESI 3, which was the category with the highest proportion of mistriage. After adjusting for study facility and triage vital signs, the researchers found that children age 6 and older were more likely to be undertriaged than those younger than 6, particularly those age 15 and older (relative risk [RR], 1.36).
Undertriage was also modestly more likely with male patients (female patients’ RR, 0.93), patients with comorbidities (RR, 1.11-1.2), patients who arrived by ambulance (RR, 1.04), and patients who were Asian (RR, 1.10), Black (RR, 1.05), or Hispanic (RR, 1.04). Undertriage became gradually less likely with each additional year in the study period, with an RR of 0.89 in 2019 and 2020.
Among the study’s limitations were use of ESI version 4, instead of the currently used 5, and the omission of common procedures from the outcome definition that “may systematically bias the analysis toward overtriage,” the editorial noted. The authors also did not include pain as a variable in the analysis, which can often indicate patient acuity.
Further, this study was unable to include covariates identified in other research that may influence clinical decision-making, such as “the presenting illness or injury, children with complex medical needs, and language proficiency,” Dr. Frankenberger and colleagues wrote. “Furthermore, environmental stressors, such as ED volume and crowding, can influence how a nurse prioritizes care and may increase bias in decision-making and/or increase practice variability.”
The study was funded by the Kaiser Permanente Northern California (KPNC) Community Health program. One author had consulting payments from CSL Behring and Abbott Point-of-Care, and six of the authors have received grant funding from the KPNC Community Health program. The editorial authors reported no conflicts of interest.
, according to a multicenter retrospective study published in JAMA Pediatrics. Researchers also identified gender, age, race, ethnicity, and comorbidity disparities in those who were undertriaged.
The researchers found that only 34.1% of visits were correctly triaged while 58.5% were overtriaged and 7.4% were undertriaged. The findings were based on analysis of more than 1 million pediatric emergency visits over a 5-year period that used the Emergency Severity Index (ESI) version 4 for triage.
“The ESI had poor sensitivity in identifying a critically ill pediatric patient, and undertriage occurred in 1 in 14 children,” wrote Dana R. Sax, MD, a senior emergency physician at The Permanente Medical Group in northern California, and her colleagues.

“More than 90% of pediatric visits were assigned a mid to low triage acuity category, and actual resource use and care intensity frequently did not align with ESI predictions,” the authors wrote. “Our findings highlight an opportunity to improve triage for pediatric patients to mitigate critical undertriage, optimize resource decisions, standardize processes across time and setting, and promote more equitable care.”
The authors added that the study findings are currently being used by the Permanente system “to develop standardized triage education across centers to improve early identification of high-risk patients.”
Disparities in Emergency Care
The results underscore the need for more work to address disparities in emergency care, wrote Warren D. Frankenberger, PhD, RN, a nurse scientist at Children’s Hospital of Philadelphia, and two colleagues in an accompanying editorial.
“Decisions in triage can have significant downstream effects on subsequent care during the ED visit,” they wrote in their editorial. “Given that the triage process in most instances is fully executed by nurses, nurse researchers are in a key position to evaluate these and other covariates to influence further improvements in triage.” They suggested that use of clinical decision support tools and artificial intelligence (AI) may improve the triage process, albeit with the caveat that AI often relies on models with pre-existing historical bias that may perpetuate structural inequalities.
Study Methodology
The researchers analyzed 1,016,816 pediatric visits at 21 emergency departments in Kaiser Permanente Northern California between January 2016 and December 2020. The patients were an average 7 years old, and 47% were female. The researchers excluded visits that lacked ESI data or had incomplete ED time variables as well as those with patients who left against medical advice, were not seen, or were transferred from another ED.
The study relied on novel definitions of ESI undertriage and overtriage developed through a modified Delphi process by a team of four emergency physicians, one pediatric emergency physician, two emergency nurses, and one pediatric ICU physician. The definition involved comparing ESI levels to the clinical outcomes and resource use.
Resources included laboratory analysis, electrocardiography, radiography, CT, MRI, diagnostic ultrasonography (not point of care), angiography, IV fluids, and IV, intramuscular, or nebulized medications. Resources did not include “oral medications, tetanus immunizations, point-of-care testing, history and physical examination, saline or heparin lock, prescription refills, simple wound care, crutches, splints, and slings.”
Level 1 events were those requiring time-sensitive, critical intervention, including high-risk sepsis. Level 2 events included most level 1 events that occurred after the first hour (except operating room admission or hospital transfer) as well as respiratory therapy, toxicology consult, lumbar puncture, suicidality as chief concern, at least 2 doses of albuterol or continuous albuterol nebulization, a skeletal survey x-ray order, and medical social work consult with an ED length of stay of at least 2 hours. Level 3 events included IV mediation order, any CT order, OR admission or hospital transfer after one hour, or any pediatric hospitalist consult.
Analyzing the ED Visits
Overtriaged cases were ESI level 1 or 2 cases in which fewer than 2 resources were used; level 3 cases where fewer than 2 resources were used and no level 1 or 2 events occurred; and level 4 cases where no resources were used.
Undertriaged cases were defined as the following:
- ESI level 5 cases where any resource was used and any level 1, 2, or 3 events occurred.
- Level 4 cases where more than 1 resource was used and any level 1, 2, or 3 events occurred.
- Level 3 cases where any level 1 event occurred, more than one level 2 event occurred, or any level 2 event occurred and more than one additional ED resource type was used.
- Level 2 cases where any level 1 event occurred.
About half the visits (51%) were assigned ESI 3, which was the category with the highest proportion of mistriage. After adjusting for study facility and triage vital signs, the researchers found that children age 6 and older were more likely to be undertriaged than those younger than 6, particularly those age 15 and older (relative risk [RR], 1.36).
Undertriage was also modestly more likely with male patients (female patients’ RR, 0.93), patients with comorbidities (RR, 1.11-1.2), patients who arrived by ambulance (RR, 1.04), and patients who were Asian (RR, 1.10), Black (RR, 1.05), or Hispanic (RR, 1.04). Undertriage became gradually less likely with each additional year in the study period, with an RR of 0.89 in 2019 and 2020.
Among the study’s limitations were use of ESI version 4, instead of the currently used 5, and the omission of common procedures from the outcome definition that “may systematically bias the analysis toward overtriage,” the editorial noted. The authors also did not include pain as a variable in the analysis, which can often indicate patient acuity.
Further, this study was unable to include covariates identified in other research that may influence clinical decision-making, such as “the presenting illness or injury, children with complex medical needs, and language proficiency,” Dr. Frankenberger and colleagues wrote. “Furthermore, environmental stressors, such as ED volume and crowding, can influence how a nurse prioritizes care and may increase bias in decision-making and/or increase practice variability.”
The study was funded by the Kaiser Permanente Northern California (KPNC) Community Health program. One author had consulting payments from CSL Behring and Abbott Point-of-Care, and six of the authors have received grant funding from the KPNC Community Health program. The editorial authors reported no conflicts of interest.
, according to a multicenter retrospective study published in JAMA Pediatrics. Researchers also identified gender, age, race, ethnicity, and comorbidity disparities in those who were undertriaged.
The researchers found that only 34.1% of visits were correctly triaged while 58.5% were overtriaged and 7.4% were undertriaged. The findings were based on analysis of more than 1 million pediatric emergency visits over a 5-year period that used the Emergency Severity Index (ESI) version 4 for triage.
“The ESI had poor sensitivity in identifying a critically ill pediatric patient, and undertriage occurred in 1 in 14 children,” wrote Dana R. Sax, MD, a senior emergency physician at The Permanente Medical Group in northern California, and her colleagues.

“More than 90% of pediatric visits were assigned a mid to low triage acuity category, and actual resource use and care intensity frequently did not align with ESI predictions,” the authors wrote. “Our findings highlight an opportunity to improve triage for pediatric patients to mitigate critical undertriage, optimize resource decisions, standardize processes across time and setting, and promote more equitable care.”
The authors added that the study findings are currently being used by the Permanente system “to develop standardized triage education across centers to improve early identification of high-risk patients.”
Disparities in Emergency Care
The results underscore the need for more work to address disparities in emergency care, wrote Warren D. Frankenberger, PhD, RN, a nurse scientist at Children’s Hospital of Philadelphia, and two colleagues in an accompanying editorial.
“Decisions in triage can have significant downstream effects on subsequent care during the ED visit,” they wrote in their editorial. “Given that the triage process in most instances is fully executed by nurses, nurse researchers are in a key position to evaluate these and other covariates to influence further improvements in triage.” They suggested that use of clinical decision support tools and artificial intelligence (AI) may improve the triage process, albeit with the caveat that AI often relies on models with pre-existing historical bias that may perpetuate structural inequalities.
Study Methodology
The researchers analyzed 1,016,816 pediatric visits at 21 emergency departments in Kaiser Permanente Northern California between January 2016 and December 2020. The patients were an average 7 years old, and 47% were female. The researchers excluded visits that lacked ESI data or had incomplete ED time variables as well as those with patients who left against medical advice, were not seen, or were transferred from another ED.
The study relied on novel definitions of ESI undertriage and overtriage developed through a modified Delphi process by a team of four emergency physicians, one pediatric emergency physician, two emergency nurses, and one pediatric ICU physician. The definition involved comparing ESI levels to the clinical outcomes and resource use.
Resources included laboratory analysis, electrocardiography, radiography, CT, MRI, diagnostic ultrasonography (not point of care), angiography, IV fluids, and IV, intramuscular, or nebulized medications. Resources did not include “oral medications, tetanus immunizations, point-of-care testing, history and physical examination, saline or heparin lock, prescription refills, simple wound care, crutches, splints, and slings.”
Level 1 events were those requiring time-sensitive, critical intervention, including high-risk sepsis. Level 2 events included most level 1 events that occurred after the first hour (except operating room admission or hospital transfer) as well as respiratory therapy, toxicology consult, lumbar puncture, suicidality as chief concern, at least 2 doses of albuterol or continuous albuterol nebulization, a skeletal survey x-ray order, and medical social work consult with an ED length of stay of at least 2 hours. Level 3 events included IV mediation order, any CT order, OR admission or hospital transfer after one hour, or any pediatric hospitalist consult.
Analyzing the ED Visits
Overtriaged cases were ESI level 1 or 2 cases in which fewer than 2 resources were used; level 3 cases where fewer than 2 resources were used and no level 1 or 2 events occurred; and level 4 cases where no resources were used.
Undertriaged cases were defined as the following:
- ESI level 5 cases where any resource was used and any level 1, 2, or 3 events occurred.
- Level 4 cases where more than 1 resource was used and any level 1, 2, or 3 events occurred.
- Level 3 cases where any level 1 event occurred, more than one level 2 event occurred, or any level 2 event occurred and more than one additional ED resource type was used.
- Level 2 cases where any level 1 event occurred.
About half the visits (51%) were assigned ESI 3, which was the category with the highest proportion of mistriage. After adjusting for study facility and triage vital signs, the researchers found that children age 6 and older were more likely to be undertriaged than those younger than 6, particularly those age 15 and older (relative risk [RR], 1.36).
Undertriage was also modestly more likely with male patients (female patients’ RR, 0.93), patients with comorbidities (RR, 1.11-1.2), patients who arrived by ambulance (RR, 1.04), and patients who were Asian (RR, 1.10), Black (RR, 1.05), or Hispanic (RR, 1.04). Undertriage became gradually less likely with each additional year in the study period, with an RR of 0.89 in 2019 and 2020.
Among the study’s limitations were use of ESI version 4, instead of the currently used 5, and the omission of common procedures from the outcome definition that “may systematically bias the analysis toward overtriage,” the editorial noted. The authors also did not include pain as a variable in the analysis, which can often indicate patient acuity.
Further, this study was unable to include covariates identified in other research that may influence clinical decision-making, such as “the presenting illness or injury, children with complex medical needs, and language proficiency,” Dr. Frankenberger and colleagues wrote. “Furthermore, environmental stressors, such as ED volume and crowding, can influence how a nurse prioritizes care and may increase bias in decision-making and/or increase practice variability.”
The study was funded by the Kaiser Permanente Northern California (KPNC) Community Health program. One author had consulting payments from CSL Behring and Abbott Point-of-Care, and six of the authors have received grant funding from the KPNC Community Health program. The editorial authors reported no conflicts of interest.
FROM JAMA PEDIATRICS
E-Bikes: The Good ... and the Ugly
Bicycles have been woven into my life since I first straddled a hand-me-down with a fan belt drive when I was 3. At age 12 my friend Ricky and I took a 250 mile–plus 2-night adventure on our 3-speed “English” style bikes. We still marvel that our parents let us do it when neither cell phones nor GPS existed.
I have always bike commuted to work, including the years when that involved a perilous navigation into Boston from the suburbs. In our mid-50s my wife and I biked from Washington state back here to Maine with another couple unsupported. We continue to do at least one self-guided cycle tour out of the country each year.
Not surprisingly, I keep a close eye on what’s happening in the bicycle market. For decades the trends have shifted back and forth between sleek road models and beefier off-roaders. There have been boom years here and there for the dealers and manufacturers, but nothing like what the bike industry is experiencing now with the arrival of e-bikes on the market. Driven primarily by electrification, micromobility ridership (which includes conventional bikes and scooters) has grown more than 50-fold over the last 10 years. Projections suggest the market’s value will be $300 billion by 2030.
It doesn’t take an MBA with a major in marketing to understand the broad appeal of electrification. Most adults have ridden a bicycle as children, but several decades of gap years has left many of them with a level of fitness that makes pedaling against the wind or up any incline difficult and unappealing. An e-bike can put even the least fitness conscious back in the saddle and open the options for outdoor recreation they haven’t dreamed of since childhood.
In large part the people flocking to e-bikes are retiree’s who thought they were “over the hill.” They are having so much fun they don’t care if the Lycra-clad “serious” cyclists notice the battery bulge in the frame on their e-bikes. Another group of e-bike adopters are motivated by the “greenness” of a fossil-fuel–free electric powered transportation which, with minimal compromise, can be used as they would a car around town and for longer commutes than they would have considered on a purely pedal-powered bicycle.
Unfortunately, there is a growing group of younger e-bike riders who are motivated and uninhibited by the potential that the power boost of a small electric motor can provide. And here is where the ugliness begins to intrude on what was otherwise a beautiful and expanding landscape. However, it is the young who are, not surprisingly, drawn to the speed, and with any vehicle – motorized or conventional – as speed increases so does the frequency and seriousness of accidents.
The term e-bike covers a broad range of vehicles, from those designated class 1, which require pedaling and are limited to 20 miles per hour, to class 3, which may have a throttle and unmodified can hit 28 mph. Class 2 bikes have a throttle that will allow the rider to reach 20 mph without pedaling. Modifying any class of e-bike can substantially increase its speed, but this is more common in classes 2 and 3. As an example, some very fast micromobiles are considered unclassified e-bikes and avoid being labeled motorcycles simply because they have pedals.
One has to give some credit to the e-bike industry for eventually adopting this classification system. But, we must give the rest of us, including parents and public safety officials, a failing grade for doing a poor job of translating these scores into enforceable regulations to protect both riders and pedestrians from serious injury.
On the governmental side only a little more than half of US states have used the three category classification to craft their regulations. Many jurisdictions have failed to differentiate between streets, sidewalks, and trails. Regulations vary from state to state, and many states leave it up to local communities. From my experience chairing our town’s Bicycle and Pedestrian Advisory Committee, I can tell you that even “progressive” communities are struggling to decide who can ride what where. The result has been that people of all ages, but mostly adolescents, are traveling on busy streets and sidewalks at speeds that put themselves and pedestrians at risk.
On the parental side of the problem are families that have either allowed or enabled their children to ride class 2 and 3 e-bikes without proper safety equipment or consideration for the safety of the rest of the community. Currently, this is not much of a problem here in Maine thanks to the weather and the high price of e-bikes. However, I frequently visit an affluent community in the San Francisco Bay Area, where it is not uncommon to see middle school children speeding along well in excess of 20 mph.
Unfortunately this is another example, like television and cell phone, in which our society has been unable to keep up with technology by molding the behavior of our children and/or creating enforceable rules that allow us to reap the benefits of new discoveries while minimizing the collateral damage that can accompany them.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
Bicycles have been woven into my life since I first straddled a hand-me-down with a fan belt drive when I was 3. At age 12 my friend Ricky and I took a 250 mile–plus 2-night adventure on our 3-speed “English” style bikes. We still marvel that our parents let us do it when neither cell phones nor GPS existed.
I have always bike commuted to work, including the years when that involved a perilous navigation into Boston from the suburbs. In our mid-50s my wife and I biked from Washington state back here to Maine with another couple unsupported. We continue to do at least one self-guided cycle tour out of the country each year.
Not surprisingly, I keep a close eye on what’s happening in the bicycle market. For decades the trends have shifted back and forth between sleek road models and beefier off-roaders. There have been boom years here and there for the dealers and manufacturers, but nothing like what the bike industry is experiencing now with the arrival of e-bikes on the market. Driven primarily by electrification, micromobility ridership (which includes conventional bikes and scooters) has grown more than 50-fold over the last 10 years. Projections suggest the market’s value will be $300 billion by 2030.
It doesn’t take an MBA with a major in marketing to understand the broad appeal of electrification. Most adults have ridden a bicycle as children, but several decades of gap years has left many of them with a level of fitness that makes pedaling against the wind or up any incline difficult and unappealing. An e-bike can put even the least fitness conscious back in the saddle and open the options for outdoor recreation they haven’t dreamed of since childhood.
In large part the people flocking to e-bikes are retiree’s who thought they were “over the hill.” They are having so much fun they don’t care if the Lycra-clad “serious” cyclists notice the battery bulge in the frame on their e-bikes. Another group of e-bike adopters are motivated by the “greenness” of a fossil-fuel–free electric powered transportation which, with minimal compromise, can be used as they would a car around town and for longer commutes than they would have considered on a purely pedal-powered bicycle.
Unfortunately, there is a growing group of younger e-bike riders who are motivated and uninhibited by the potential that the power boost of a small electric motor can provide. And here is where the ugliness begins to intrude on what was otherwise a beautiful and expanding landscape. However, it is the young who are, not surprisingly, drawn to the speed, and with any vehicle – motorized or conventional – as speed increases so does the frequency and seriousness of accidents.
The term e-bike covers a broad range of vehicles, from those designated class 1, which require pedaling and are limited to 20 miles per hour, to class 3, which may have a throttle and unmodified can hit 28 mph. Class 2 bikes have a throttle that will allow the rider to reach 20 mph without pedaling. Modifying any class of e-bike can substantially increase its speed, but this is more common in classes 2 and 3. As an example, some very fast micromobiles are considered unclassified e-bikes and avoid being labeled motorcycles simply because they have pedals.
One has to give some credit to the e-bike industry for eventually adopting this classification system. But, we must give the rest of us, including parents and public safety officials, a failing grade for doing a poor job of translating these scores into enforceable regulations to protect both riders and pedestrians from serious injury.
On the governmental side only a little more than half of US states have used the three category classification to craft their regulations. Many jurisdictions have failed to differentiate between streets, sidewalks, and trails. Regulations vary from state to state, and many states leave it up to local communities. From my experience chairing our town’s Bicycle and Pedestrian Advisory Committee, I can tell you that even “progressive” communities are struggling to decide who can ride what where. The result has been that people of all ages, but mostly adolescents, are traveling on busy streets and sidewalks at speeds that put themselves and pedestrians at risk.
On the parental side of the problem are families that have either allowed or enabled their children to ride class 2 and 3 e-bikes without proper safety equipment or consideration for the safety of the rest of the community. Currently, this is not much of a problem here in Maine thanks to the weather and the high price of e-bikes. However, I frequently visit an affluent community in the San Francisco Bay Area, where it is not uncommon to see middle school children speeding along well in excess of 20 mph.
Unfortunately this is another example, like television and cell phone, in which our society has been unable to keep up with technology by molding the behavior of our children and/or creating enforceable rules that allow us to reap the benefits of new discoveries while minimizing the collateral damage that can accompany them.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
Bicycles have been woven into my life since I first straddled a hand-me-down with a fan belt drive when I was 3. At age 12 my friend Ricky and I took a 250 mile–plus 2-night adventure on our 3-speed “English” style bikes. We still marvel that our parents let us do it when neither cell phones nor GPS existed.
I have always bike commuted to work, including the years when that involved a perilous navigation into Boston from the suburbs. In our mid-50s my wife and I biked from Washington state back here to Maine with another couple unsupported. We continue to do at least one self-guided cycle tour out of the country each year.
Not surprisingly, I keep a close eye on what’s happening in the bicycle market. For decades the trends have shifted back and forth between sleek road models and beefier off-roaders. There have been boom years here and there for the dealers and manufacturers, but nothing like what the bike industry is experiencing now with the arrival of e-bikes on the market. Driven primarily by electrification, micromobility ridership (which includes conventional bikes and scooters) has grown more than 50-fold over the last 10 years. Projections suggest the market’s value will be $300 billion by 2030.
It doesn’t take an MBA with a major in marketing to understand the broad appeal of electrification. Most adults have ridden a bicycle as children, but several decades of gap years has left many of them with a level of fitness that makes pedaling against the wind or up any incline difficult and unappealing. An e-bike can put even the least fitness conscious back in the saddle and open the options for outdoor recreation they haven’t dreamed of since childhood.
In large part the people flocking to e-bikes are retiree’s who thought they were “over the hill.” They are having so much fun they don’t care if the Lycra-clad “serious” cyclists notice the battery bulge in the frame on their e-bikes. Another group of e-bike adopters are motivated by the “greenness” of a fossil-fuel–free electric powered transportation which, with minimal compromise, can be used as they would a car around town and for longer commutes than they would have considered on a purely pedal-powered bicycle.
Unfortunately, there is a growing group of younger e-bike riders who are motivated and uninhibited by the potential that the power boost of a small electric motor can provide. And here is where the ugliness begins to intrude on what was otherwise a beautiful and expanding landscape. However, it is the young who are, not surprisingly, drawn to the speed, and with any vehicle – motorized or conventional – as speed increases so does the frequency and seriousness of accidents.
The term e-bike covers a broad range of vehicles, from those designated class 1, which require pedaling and are limited to 20 miles per hour, to class 3, which may have a throttle and unmodified can hit 28 mph. Class 2 bikes have a throttle that will allow the rider to reach 20 mph without pedaling. Modifying any class of e-bike can substantially increase its speed, but this is more common in classes 2 and 3. As an example, some very fast micromobiles are considered unclassified e-bikes and avoid being labeled motorcycles simply because they have pedals.
One has to give some credit to the e-bike industry for eventually adopting this classification system. But, we must give the rest of us, including parents and public safety officials, a failing grade for doing a poor job of translating these scores into enforceable regulations to protect both riders and pedestrians from serious injury.
On the governmental side only a little more than half of US states have used the three category classification to craft their regulations. Many jurisdictions have failed to differentiate between streets, sidewalks, and trails. Regulations vary from state to state, and many states leave it up to local communities. From my experience chairing our town’s Bicycle and Pedestrian Advisory Committee, I can tell you that even “progressive” communities are struggling to decide who can ride what where. The result has been that people of all ages, but mostly adolescents, are traveling on busy streets and sidewalks at speeds that put themselves and pedestrians at risk.
On the parental side of the problem are families that have either allowed or enabled their children to ride class 2 and 3 e-bikes without proper safety equipment or consideration for the safety of the rest of the community. Currently, this is not much of a problem here in Maine thanks to the weather and the high price of e-bikes. However, I frequently visit an affluent community in the San Francisco Bay Area, where it is not uncommon to see middle school children speeding along well in excess of 20 mph.
Unfortunately this is another example, like television and cell phone, in which our society has been unable to keep up with technology by molding the behavior of our children and/or creating enforceable rules that allow us to reap the benefits of new discoveries while minimizing the collateral damage that can accompany them.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
Doctor I-Don’t-Know
Many, many years ago there was a Thanksgiving when as I was just beginning to earn a reputation in my wife’s family. There were no place cards on the table and the usual hovering and jockeying seats was well underway. From behind me I heard one of my young nieces pipe up: “I want to sit next to Doctor I-don’t-know.”
After a few words of negotiation we were all settled in our places and ready to enjoy our meal. It took only a few seconds of introspection for me to grasp how I had received that moniker, which some physicians might consider disrespectful.
I was the only physician within several generations of that family and, as such, my in-laws thought it only appropriate to ask me medical questions. They courteously seemed to avoid personal questions about their own health and were particularly careful not to roll up their sleeves or unbutton their shirts to show me a lesion or a recently acquired surgical scar. No, my wife’s family members were curious. They wanted answers to deeper questions, the hard science so to speak. “How does aspirin work?” was a typical and painful example. Maybe pharmacologists today have better answers but 40 years ago I’m not so sure; I certainly didn’t know back then and would reply, “I don’t know.” Probably for the third or fourth time that day.
Usually I genuinely didn’t know the answer. However, sometimes my answer was going to be so different from the beliefs and biases of my inquisitor that, in the interest of expediency, “I don’t know” seemed the most appropriate response.
If you were reading Letters from Maine 25 years ago, that scenario might sound familiar. I have chosen to pull it out of the archives as a jumping-off point for a consideration of the unfortunate example some of us set when the COVID pandemic threw a tsunami of unknowns at us. Too many physician-“experts” were afraid to say, “I don’t know.” Instead, and maybe because, they themselves were afraid that the patients couldn’t handle the truth that none of us in the profession knew the correct answers. When so many initial pronouncements proved incorrect, it was too late to undo the damage that had been done to the community’s trust in the rest of us.
It turns out that my in-laws were not the only folks who thought of me as Doctor I-don’t-know. One of the perks of remaining in the same community after one retires is that encounters with former patients and their parents happen frequently. On more than one occasion a parent has thanked me for admitting my ignorance. Some have even claimed that my candid approach was what they remembered most fondly. And, that quality increased their trust when I finally provided an answer.
There is an art to delivering “I don’t know.” Thirty years ago I would excuse myself and tell the family I was going to my office to pull a book off the shelf or call a previous mentor. Now one only needs to ask Dr. Google. No need to leave the room. If appropriate, the provider can swing the computer screen so that the patient can share in the search for the answer.
That strategy only works when the provider merely needs to update or expand his/her knowledge. However, there are those difficult situations when no one could know the answer given the current parameters of the patient’s situation. More lab work might be needed. It may be too early in the trajectory of the patient’s illness for the illnesses signs and symptoms to declare themselves.
In these situations “I don’t know” must be followed by a “but.” It is what comes after that “but” and how it is delivered that can convert the provider’s admission of ignorance into a demonstration of his or her character. Is he/she a caring person trying to understand the patient’s concerns? Willing to enter into a cooperative relationship as together they search for the cause and hopefully for a cure for the patient’s currently mysterious illness?
I recently read about a physician who is encouraging medical educators to incorporate more discussions of “humility” and its role in patient care into the medical school and postgraduate training curricula. He feels, as do I, that
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
Many, many years ago there was a Thanksgiving when as I was just beginning to earn a reputation in my wife’s family. There were no place cards on the table and the usual hovering and jockeying seats was well underway. From behind me I heard one of my young nieces pipe up: “I want to sit next to Doctor I-don’t-know.”
After a few words of negotiation we were all settled in our places and ready to enjoy our meal. It took only a few seconds of introspection for me to grasp how I had received that moniker, which some physicians might consider disrespectful.
I was the only physician within several generations of that family and, as such, my in-laws thought it only appropriate to ask me medical questions. They courteously seemed to avoid personal questions about their own health and were particularly careful not to roll up their sleeves or unbutton their shirts to show me a lesion or a recently acquired surgical scar. No, my wife’s family members were curious. They wanted answers to deeper questions, the hard science so to speak. “How does aspirin work?” was a typical and painful example. Maybe pharmacologists today have better answers but 40 years ago I’m not so sure; I certainly didn’t know back then and would reply, “I don’t know.” Probably for the third or fourth time that day.
Usually I genuinely didn’t know the answer. However, sometimes my answer was going to be so different from the beliefs and biases of my inquisitor that, in the interest of expediency, “I don’t know” seemed the most appropriate response.
If you were reading Letters from Maine 25 years ago, that scenario might sound familiar. I have chosen to pull it out of the archives as a jumping-off point for a consideration of the unfortunate example some of us set when the COVID pandemic threw a tsunami of unknowns at us. Too many physician-“experts” were afraid to say, “I don’t know.” Instead, and maybe because, they themselves were afraid that the patients couldn’t handle the truth that none of us in the profession knew the correct answers. When so many initial pronouncements proved incorrect, it was too late to undo the damage that had been done to the community’s trust in the rest of us.
It turns out that my in-laws were not the only folks who thought of me as Doctor I-don’t-know. One of the perks of remaining in the same community after one retires is that encounters with former patients and their parents happen frequently. On more than one occasion a parent has thanked me for admitting my ignorance. Some have even claimed that my candid approach was what they remembered most fondly. And, that quality increased their trust when I finally provided an answer.
There is an art to delivering “I don’t know.” Thirty years ago I would excuse myself and tell the family I was going to my office to pull a book off the shelf or call a previous mentor. Now one only needs to ask Dr. Google. No need to leave the room. If appropriate, the provider can swing the computer screen so that the patient can share in the search for the answer.
That strategy only works when the provider merely needs to update or expand his/her knowledge. However, there are those difficult situations when no one could know the answer given the current parameters of the patient’s situation. More lab work might be needed. It may be too early in the trajectory of the patient’s illness for the illnesses signs and symptoms to declare themselves.
In these situations “I don’t know” must be followed by a “but.” It is what comes after that “but” and how it is delivered that can convert the provider’s admission of ignorance into a demonstration of his or her character. Is he/she a caring person trying to understand the patient’s concerns? Willing to enter into a cooperative relationship as together they search for the cause and hopefully for a cure for the patient’s currently mysterious illness?
I recently read about a physician who is encouraging medical educators to incorporate more discussions of “humility” and its role in patient care into the medical school and postgraduate training curricula. He feels, as do I, that
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
Many, many years ago there was a Thanksgiving when as I was just beginning to earn a reputation in my wife’s family. There were no place cards on the table and the usual hovering and jockeying seats was well underway. From behind me I heard one of my young nieces pipe up: “I want to sit next to Doctor I-don’t-know.”
After a few words of negotiation we were all settled in our places and ready to enjoy our meal. It took only a few seconds of introspection for me to grasp how I had received that moniker, which some physicians might consider disrespectful.
I was the only physician within several generations of that family and, as such, my in-laws thought it only appropriate to ask me medical questions. They courteously seemed to avoid personal questions about their own health and were particularly careful not to roll up their sleeves or unbutton their shirts to show me a lesion or a recently acquired surgical scar. No, my wife’s family members were curious. They wanted answers to deeper questions, the hard science so to speak. “How does aspirin work?” was a typical and painful example. Maybe pharmacologists today have better answers but 40 years ago I’m not so sure; I certainly didn’t know back then and would reply, “I don’t know.” Probably for the third or fourth time that day.
Usually I genuinely didn’t know the answer. However, sometimes my answer was going to be so different from the beliefs and biases of my inquisitor that, in the interest of expediency, “I don’t know” seemed the most appropriate response.
If you were reading Letters from Maine 25 years ago, that scenario might sound familiar. I have chosen to pull it out of the archives as a jumping-off point for a consideration of the unfortunate example some of us set when the COVID pandemic threw a tsunami of unknowns at us. Too many physician-“experts” were afraid to say, “I don’t know.” Instead, and maybe because, they themselves were afraid that the patients couldn’t handle the truth that none of us in the profession knew the correct answers. When so many initial pronouncements proved incorrect, it was too late to undo the damage that had been done to the community’s trust in the rest of us.
It turns out that my in-laws were not the only folks who thought of me as Doctor I-don’t-know. One of the perks of remaining in the same community after one retires is that encounters with former patients and their parents happen frequently. On more than one occasion a parent has thanked me for admitting my ignorance. Some have even claimed that my candid approach was what they remembered most fondly. And, that quality increased their trust when I finally provided an answer.
There is an art to delivering “I don’t know.” Thirty years ago I would excuse myself and tell the family I was going to my office to pull a book off the shelf or call a previous mentor. Now one only needs to ask Dr. Google. No need to leave the room. If appropriate, the provider can swing the computer screen so that the patient can share in the search for the answer.
That strategy only works when the provider merely needs to update or expand his/her knowledge. However, there are those difficult situations when no one could know the answer given the current parameters of the patient’s situation. More lab work might be needed. It may be too early in the trajectory of the patient’s illness for the illnesses signs and symptoms to declare themselves.
In these situations “I don’t know” must be followed by a “but.” It is what comes after that “but” and how it is delivered that can convert the provider’s admission of ignorance into a demonstration of his or her character. Is he/she a caring person trying to understand the patient’s concerns? Willing to enter into a cooperative relationship as together they search for the cause and hopefully for a cure for the patient’s currently mysterious illness?
I recently read about a physician who is encouraging medical educators to incorporate more discussions of “humility” and its role in patient care into the medical school and postgraduate training curricula. He feels, as do I, that
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.