User login
VIDEO: Regionalized STEMI care slashes in-hospital mortality
ANAHEIM, CALIF. – An American Heart Association program aimed at streamlining care of patients with ST-elevation MI resulted in a dramatic near-halving of in-hospital mortality, compared with STEMI patients treated in hospitals not participating in the project, James G. Jollis, MD, reported at the American Heart Association scientific sessions.
He presented the results of the STEMI ACCELERATOR 2 study, which involved 12 participating metropolitan regions across the United States, 132 percutaneous coronary intervention–capable hospitals, and 946 emergency medical services agencies. The ACCELERATOR 2 program entailed regional implementation of a structured STEMI care plan in which EMS personnel were trained to obtain prehospital ECGs and to activate cardiac catheterization labs prior to hospital arrival, bypassing the emergency department when appropriate.
Key elements of the project, which was part of the AHA’s Mission: Lifeline program, included having participating hospitals measure their performance of key processes and send that information as well as patient outcome data to the National Cardiovascular Data Registry’s ACTION–Get With The Guidelines registry. The hospitals in turn received quarterly feedback reports containing blinded hospital comparisons.
Dr. Jollis and his coinvestigators worked to obtain buy-in from local stakeholders, organize regional leadership, and help in drafting a central regional STEMI plan featuring prespecified treatment protocols.
The STEMI ACCELERATOR 2 study was carried out in 2015-2017, during which 10,730 patients with STEMI were transported directly to participating hospitals with PCI capability.
The primary study outcome was the change from the first to the final quarter of the study in the proportion of EMS-transported patients with a time from first medical contact to treatment in the cath lab of 90 minutes or less. This improved significantly, from 67% at baseline to 74% in the final quarter. Nine of the 12 participating regions reduced their time from first medical contact to treatment in the cath lab, and eight reached the national of goal of having 75% of STEMI patients treated within 90 minutes.
The other key time-to-care measures improved, too: At baseline, only 38% of patients had a time from first medical contact to cath lab activation of 20 minutes or less; by the final quarter, this figure had climbed to 56%. That’s an important metric, as evidenced by the study finding that in-hospital mortality occurred in 4.5% of patients with a time from first medical contact to cath lab activation of more than 20 minutes, compared with 2.2% in those with a time of 20 minutes or less.
Also, the proportion of patients who spent 20 minutes or less in the emergency department improved from 33% to 43%.
In-hospital mortality improved from 4.4% in the baseline quarter to 2.3% in the final quarter. No similar improvement in in-hospital mortality occurred in a comparison group of 22,651 STEMI patients treated at hospitals not involved in ACCELERATOR 2.
A significant reduction in the rate of in-hospital congestive heart failure occurred in the ACCELERATOR 2 centers, from 7.4% at baseline to 5.0%. In contrast, stroke, cardiogenic shock, and major bleeding rates were unchanged over time.
The ACCELERATOR 2 model of emergency cardiovascular care is designed to be highly generalizable, according to Dr. Jollis.
“This study supports the implementation of regionally coordinated systems across the United States to abort heart attacks, save lives, and enable heart attack victims to return to their families and productive lives,” he said.
The ACCELERATOR 2 operations manual – essentially a blueprint for organizing a regional STEMI system of care – is available gratis.
Dr. Allen, a cardiologist at the University of Colorado, Denver, said the ACCELERATOR 2 model has been successful because it is consistent with a fundamental principle of implementation science as described by Carolyn Clancy, MD, Executive in Charge at the Veterans Health Affairs Administration, who has said it’s a matter of making the right thing to do the easy thing to do.
Gregg C. Fonarow, MD, founder of the Get With the Guidelines program, predicted that the success of this program will lead to a ramping up of efforts to regionalize and coordinate STEMI care across the country. “I hope and anticipate that the AHA will take and run with the ACCELERATOR 2 model and adopt this into Mission: Lifeline, hoping to make this the standard approach to further improving care and outcomes in these patients,” said Dr. Fonarow, professor and cochief of cardiology at the University of California, Los Angeles, in a video interview.
Simultaneous with his presentation at the AHA conference, the results of STEMI ACCELERATOR 2 were published online in Circulation (2017 Nov 14; doi: 0.1161/CIRCULATIONAHA.117.032446).
The trial was sponsored by research and educational grants from AstraZeneca and The Medicines Company. Dr. Jollis reported having no financial conflicts of interest.
ANAHEIM, CALIF. – An American Heart Association program aimed at streamlining care of patients with ST-elevation MI resulted in a dramatic near-halving of in-hospital mortality, compared with STEMI patients treated in hospitals not participating in the project, James G. Jollis, MD, reported at the American Heart Association scientific sessions.
He presented the results of the STEMI ACCELERATOR 2 study, which involved 12 participating metropolitan regions across the United States, 132 percutaneous coronary intervention–capable hospitals, and 946 emergency medical services agencies. The ACCELERATOR 2 program entailed regional implementation of a structured STEMI care plan in which EMS personnel were trained to obtain prehospital ECGs and to activate cardiac catheterization labs prior to hospital arrival, bypassing the emergency department when appropriate.
Key elements of the project, which was part of the AHA’s Mission: Lifeline program, included having participating hospitals measure their performance of key processes and send that information as well as patient outcome data to the National Cardiovascular Data Registry’s ACTION–Get With The Guidelines registry. The hospitals in turn received quarterly feedback reports containing blinded hospital comparisons.
Dr. Jollis and his coinvestigators worked to obtain buy-in from local stakeholders, organize regional leadership, and help in drafting a central regional STEMI plan featuring prespecified treatment protocols.
The STEMI ACCELERATOR 2 study was carried out in 2015-2017, during which 10,730 patients with STEMI were transported directly to participating hospitals with PCI capability.
The primary study outcome was the change from the first to the final quarter of the study in the proportion of EMS-transported patients with a time from first medical contact to treatment in the cath lab of 90 minutes or less. This improved significantly, from 67% at baseline to 74% in the final quarter. Nine of the 12 participating regions reduced their time from first medical contact to treatment in the cath lab, and eight reached the national of goal of having 75% of STEMI patients treated within 90 minutes.
The other key time-to-care measures improved, too: At baseline, only 38% of patients had a time from first medical contact to cath lab activation of 20 minutes or less; by the final quarter, this figure had climbed to 56%. That’s an important metric, as evidenced by the study finding that in-hospital mortality occurred in 4.5% of patients with a time from first medical contact to cath lab activation of more than 20 minutes, compared with 2.2% in those with a time of 20 minutes or less.
Also, the proportion of patients who spent 20 minutes or less in the emergency department improved from 33% to 43%.
In-hospital mortality improved from 4.4% in the baseline quarter to 2.3% in the final quarter. No similar improvement in in-hospital mortality occurred in a comparison group of 22,651 STEMI patients treated at hospitals not involved in ACCELERATOR 2.
A significant reduction in the rate of in-hospital congestive heart failure occurred in the ACCELERATOR 2 centers, from 7.4% at baseline to 5.0%. In contrast, stroke, cardiogenic shock, and major bleeding rates were unchanged over time.
The ACCELERATOR 2 model of emergency cardiovascular care is designed to be highly generalizable, according to Dr. Jollis.
“This study supports the implementation of regionally coordinated systems across the United States to abort heart attacks, save lives, and enable heart attack victims to return to their families and productive lives,” he said.
The ACCELERATOR 2 operations manual – essentially a blueprint for organizing a regional STEMI system of care – is available gratis.
Dr. Allen, a cardiologist at the University of Colorado, Denver, said the ACCELERATOR 2 model has been successful because it is consistent with a fundamental principle of implementation science as described by Carolyn Clancy, MD, Executive in Charge at the Veterans Health Affairs Administration, who has said it’s a matter of making the right thing to do the easy thing to do.
Gregg C. Fonarow, MD, founder of the Get With the Guidelines program, predicted that the success of this program will lead to a ramping up of efforts to regionalize and coordinate STEMI care across the country. “I hope and anticipate that the AHA will take and run with the ACCELERATOR 2 model and adopt this into Mission: Lifeline, hoping to make this the standard approach to further improving care and outcomes in these patients,” said Dr. Fonarow, professor and cochief of cardiology at the University of California, Los Angeles, in a video interview.
Simultaneous with his presentation at the AHA conference, the results of STEMI ACCELERATOR 2 were published online in Circulation (2017 Nov 14; doi: 0.1161/CIRCULATIONAHA.117.032446).
The trial was sponsored by research and educational grants from AstraZeneca and The Medicines Company. Dr. Jollis reported having no financial conflicts of interest.
ANAHEIM, CALIF. – An American Heart Association program aimed at streamlining care of patients with ST-elevation MI resulted in a dramatic near-halving of in-hospital mortality, compared with STEMI patients treated in hospitals not participating in the project, James G. Jollis, MD, reported at the American Heart Association scientific sessions.
He presented the results of the STEMI ACCELERATOR 2 study, which involved 12 participating metropolitan regions across the United States, 132 percutaneous coronary intervention–capable hospitals, and 946 emergency medical services agencies. The ACCELERATOR 2 program entailed regional implementation of a structured STEMI care plan in which EMS personnel were trained to obtain prehospital ECGs and to activate cardiac catheterization labs prior to hospital arrival, bypassing the emergency department when appropriate.
Key elements of the project, which was part of the AHA’s Mission: Lifeline program, included having participating hospitals measure their performance of key processes and send that information as well as patient outcome data to the National Cardiovascular Data Registry’s ACTION–Get With The Guidelines registry. The hospitals in turn received quarterly feedback reports containing blinded hospital comparisons.
Dr. Jollis and his coinvestigators worked to obtain buy-in from local stakeholders, organize regional leadership, and help in drafting a central regional STEMI plan featuring prespecified treatment protocols.
The STEMI ACCELERATOR 2 study was carried out in 2015-2017, during which 10,730 patients with STEMI were transported directly to participating hospitals with PCI capability.
The primary study outcome was the change from the first to the final quarter of the study in the proportion of EMS-transported patients with a time from first medical contact to treatment in the cath lab of 90 minutes or less. This improved significantly, from 67% at baseline to 74% in the final quarter. Nine of the 12 participating regions reduced their time from first medical contact to treatment in the cath lab, and eight reached the national of goal of having 75% of STEMI patients treated within 90 minutes.
The other key time-to-care measures improved, too: At baseline, only 38% of patients had a time from first medical contact to cath lab activation of 20 minutes or less; by the final quarter, this figure had climbed to 56%. That’s an important metric, as evidenced by the study finding that in-hospital mortality occurred in 4.5% of patients with a time from first medical contact to cath lab activation of more than 20 minutes, compared with 2.2% in those with a time of 20 minutes or less.
Also, the proportion of patients who spent 20 minutes or less in the emergency department improved from 33% to 43%.
In-hospital mortality improved from 4.4% in the baseline quarter to 2.3% in the final quarter. No similar improvement in in-hospital mortality occurred in a comparison group of 22,651 STEMI patients treated at hospitals not involved in ACCELERATOR 2.
A significant reduction in the rate of in-hospital congestive heart failure occurred in the ACCELERATOR 2 centers, from 7.4% at baseline to 5.0%. In contrast, stroke, cardiogenic shock, and major bleeding rates were unchanged over time.
The ACCELERATOR 2 model of emergency cardiovascular care is designed to be highly generalizable, according to Dr. Jollis.
“This study supports the implementation of regionally coordinated systems across the United States to abort heart attacks, save lives, and enable heart attack victims to return to their families and productive lives,” he said.
The ACCELERATOR 2 operations manual – essentially a blueprint for organizing a regional STEMI system of care – is available gratis.
Dr. Allen, a cardiologist at the University of Colorado, Denver, said the ACCELERATOR 2 model has been successful because it is consistent with a fundamental principle of implementation science as described by Carolyn Clancy, MD, Executive in Charge at the Veterans Health Affairs Administration, who has said it’s a matter of making the right thing to do the easy thing to do.
Gregg C. Fonarow, MD, founder of the Get With the Guidelines program, predicted that the success of this program will lead to a ramping up of efforts to regionalize and coordinate STEMI care across the country. “I hope and anticipate that the AHA will take and run with the ACCELERATOR 2 model and adopt this into Mission: Lifeline, hoping to make this the standard approach to further improving care and outcomes in these patients,” said Dr. Fonarow, professor and cochief of cardiology at the University of California, Los Angeles, in a video interview.
Simultaneous with his presentation at the AHA conference, the results of STEMI ACCELERATOR 2 were published online in Circulation (2017 Nov 14; doi: 0.1161/CIRCULATIONAHA.117.032446).
The trial was sponsored by research and educational grants from AstraZeneca and The Medicines Company. Dr. Jollis reported having no financial conflicts of interest.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point:
Major finding: The in-hospital mortality rate of STEMI patients dropped from 4.4% in the baseline quarter to 2.3% in the final quarter of a study that examined the impact of introducing regionalized STEMI care.
Data source: This was a prospective study of an intervention that involved implementation of regionalized STEMI care in a dozen U.S. metropolitan areas.
Disclosures: The study was sponsored by research and educational grants from AstraZeneca and The Medicines Company. The presenter reported having no financial conflicts of interest.
Bicarb, acetylcysteine during angiography don’t protect kidneys
Periprocedural administration of intravenous sodium bicarbonate did not improve outcomes compared with standard sodium chloride in patients with impaired kidney function undergoing angiography, according to results of a randomized study of 5,177 patients.
In addition, there was no benefit for oral acetylcysteine administration over placebo for mitigating those same postangiography risks, Steven D. Weisbord, MD, said at the American Heart Association scientific sessions in Anaheim, Calif.
Hypothetically, both sodium bicarbonate and acetylcysteine could help prevent acute kidney injury associated with contrast material used during angiography, said Dr. Weisbord of the University of Pittsburgh.
However, multiple studies of the two agents have yielded “inconsistent results … consequently, equipoise exists regarding these interventions, despite their widespread use in clinical practice,” Dr. Weisbord said.
To provide more definitive evidence, Dr. Weisbord and his colleagues conducted PRESERVE, a multicenter, randomized, controlled trial comprising 5,177 patients scheduled for angiography who were at high risk of renal complications. Using a 2-by-2 factorial design, patients were randomized to receive intravenous 1.26% sodium bicarbonate or intravenous 0.9% sodium chloride, and to 5 days of oral acetylcysteine or oral placebo.
They found no significant differences between arms in the study’s composite primary endpoint of death, need for dialysis, or persistent increase in serum creatinine by 50% or more.
That composite endpoint occurred in 4.4% of patients receiving sodium bicarbonate, and similarly in 4.7% of patients receiving sodium chloride.
Likewise, the endpoint occurred in 4.6% of patients in the acetylcysteine group and 4.5% of the placebo group, Dr. Weisbord reported.
The investigators had planned to enroll 7,680 patients, but the sponsor of the trial stopped the study after enrollment of 5,177 based on the results showing no significant benefit of either treatment, he noted.
There are a few reasons why results of PRESERVE might show a lack of benefit for these agents, in contrast to some previous studies suggesting both the treatments might reduce risk of contrast-associated renal complications in high-risk patients.
Notably, “most of these interventions have been underpowered,” Dr. Weisbord noted.
Also, most previous trials used a primary endpoint of increase in blood creatinine level within days of the angiography. By contrast, the primary endpoint of the current study was a composite of serious adverse events “that are recognized sequelae of acute kidney injury,” he added.
Although subsequent investigations could shed new light on the controversy, the findings of PRESERVE support the “strong likelihood that these interventions are not clinically effective” in preventing acute kidney injury or longer-term adverse outcomes after angiography, he concluded.
The PRESERVE results were published simultaneously with Dr. Weisbord’s presentation (N Engl J Med. 2017 Nov 12. doi: 10.1056/NEJMoa1710933).
The study was supported by the U.S. Department of Veterans Affairs Office of Research and Development and the National Health and Medical Research Council of Australia. Dr. Weisbord reported receiving personal fees from Durect outside the submitted work.
Periprocedural administration of intravenous sodium bicarbonate did not improve outcomes compared with standard sodium chloride in patients with impaired kidney function undergoing angiography, according to results of a randomized study of 5,177 patients.
In addition, there was no benefit for oral acetylcysteine administration over placebo for mitigating those same postangiography risks, Steven D. Weisbord, MD, said at the American Heart Association scientific sessions in Anaheim, Calif.
Hypothetically, both sodium bicarbonate and acetylcysteine could help prevent acute kidney injury associated with contrast material used during angiography, said Dr. Weisbord of the University of Pittsburgh.
However, multiple studies of the two agents have yielded “inconsistent results … consequently, equipoise exists regarding these interventions, despite their widespread use in clinical practice,” Dr. Weisbord said.
To provide more definitive evidence, Dr. Weisbord and his colleagues conducted PRESERVE, a multicenter, randomized, controlled trial comprising 5,177 patients scheduled for angiography who were at high risk of renal complications. Using a 2-by-2 factorial design, patients were randomized to receive intravenous 1.26% sodium bicarbonate or intravenous 0.9% sodium chloride, and to 5 days of oral acetylcysteine or oral placebo.
They found no significant differences between arms in the study’s composite primary endpoint of death, need for dialysis, or persistent increase in serum creatinine by 50% or more.
That composite endpoint occurred in 4.4% of patients receiving sodium bicarbonate, and similarly in 4.7% of patients receiving sodium chloride.
Likewise, the endpoint occurred in 4.6% of patients in the acetylcysteine group and 4.5% of the placebo group, Dr. Weisbord reported.
The investigators had planned to enroll 7,680 patients, but the sponsor of the trial stopped the study after enrollment of 5,177 based on the results showing no significant benefit of either treatment, he noted.
There are a few reasons why results of PRESERVE might show a lack of benefit for these agents, in contrast to some previous studies suggesting both the treatments might reduce risk of contrast-associated renal complications in high-risk patients.
Notably, “most of these interventions have been underpowered,” Dr. Weisbord noted.
Also, most previous trials used a primary endpoint of increase in blood creatinine level within days of the angiography. By contrast, the primary endpoint of the current study was a composite of serious adverse events “that are recognized sequelae of acute kidney injury,” he added.
Although subsequent investigations could shed new light on the controversy, the findings of PRESERVE support the “strong likelihood that these interventions are not clinically effective” in preventing acute kidney injury or longer-term adverse outcomes after angiography, he concluded.
The PRESERVE results were published simultaneously with Dr. Weisbord’s presentation (N Engl J Med. 2017 Nov 12. doi: 10.1056/NEJMoa1710933).
The study was supported by the U.S. Department of Veterans Affairs Office of Research and Development and the National Health and Medical Research Council of Australia. Dr. Weisbord reported receiving personal fees from Durect outside the submitted work.
Periprocedural administration of intravenous sodium bicarbonate did not improve outcomes compared with standard sodium chloride in patients with impaired kidney function undergoing angiography, according to results of a randomized study of 5,177 patients.
In addition, there was no benefit for oral acetylcysteine administration over placebo for mitigating those same postangiography risks, Steven D. Weisbord, MD, said at the American Heart Association scientific sessions in Anaheim, Calif.
Hypothetically, both sodium bicarbonate and acetylcysteine could help prevent acute kidney injury associated with contrast material used during angiography, said Dr. Weisbord of the University of Pittsburgh.
However, multiple studies of the two agents have yielded “inconsistent results … consequently, equipoise exists regarding these interventions, despite their widespread use in clinical practice,” Dr. Weisbord said.
To provide more definitive evidence, Dr. Weisbord and his colleagues conducted PRESERVE, a multicenter, randomized, controlled trial comprising 5,177 patients scheduled for angiography who were at high risk of renal complications. Using a 2-by-2 factorial design, patients were randomized to receive intravenous 1.26% sodium bicarbonate or intravenous 0.9% sodium chloride, and to 5 days of oral acetylcysteine or oral placebo.
They found no significant differences between arms in the study’s composite primary endpoint of death, need for dialysis, or persistent increase in serum creatinine by 50% or more.
That composite endpoint occurred in 4.4% of patients receiving sodium bicarbonate, and similarly in 4.7% of patients receiving sodium chloride.
Likewise, the endpoint occurred in 4.6% of patients in the acetylcysteine group and 4.5% of the placebo group, Dr. Weisbord reported.
The investigators had planned to enroll 7,680 patients, but the sponsor of the trial stopped the study after enrollment of 5,177 based on the results showing no significant benefit of either treatment, he noted.
There are a few reasons why results of PRESERVE might show a lack of benefit for these agents, in contrast to some previous studies suggesting both the treatments might reduce risk of contrast-associated renal complications in high-risk patients.
Notably, “most of these interventions have been underpowered,” Dr. Weisbord noted.
Also, most previous trials used a primary endpoint of increase in blood creatinine level within days of the angiography. By contrast, the primary endpoint of the current study was a composite of serious adverse events “that are recognized sequelae of acute kidney injury,” he added.
Although subsequent investigations could shed new light on the controversy, the findings of PRESERVE support the “strong likelihood that these interventions are not clinically effective” in preventing acute kidney injury or longer-term adverse outcomes after angiography, he concluded.
The PRESERVE results were published simultaneously with Dr. Weisbord’s presentation (N Engl J Med. 2017 Nov 12. doi: 10.1056/NEJMoa1710933).
The study was supported by the U.S. Department of Veterans Affairs Office of Research and Development and the National Health and Medical Research Council of Australia. Dr. Weisbord reported receiving personal fees from Durect outside the submitted work.
FROM THE AHA SCIENTIFIC SESSIONS
Key clinical point:
Major finding: The composite primary endpoint of death, need for dialysis, or persistent increase in serum creatinine was similar regardless of which treatments the patients received.
Data source: PRESERVE, a randomized study using a 2-by-2 factorial design to evaluate intravenous sodium bicarbonate versus sodium chloride and acetylcysteine versus placebo in 5,177 patients at high risk of renal complications.
Disclosures: PRESERVE was supported by the U.S. Department of Veterans Affairs Office of Research and Development and the National Health and Medical Research Council of Australia. Dr. Weisbord reported receiving personal fees from Durect outside the submitted work.
VIDEO: U.S. hypertension guidelines reset threshold to 130/80 mm Hg
ANAHEIM, CALIF. – Thirty million Americans became hypertensive overnight on Nov. 13 with the introduction of new high blood pressure guidelines from the American College of Cardiology and American Heart Association.
That happened by resetting the definition of adult hypertension from the long-standing threshold of 140/90 mm Hg to a blood pressure at or above 130/80 mm Hg, a change that jumps the U.S. adult prevalence of hypertension from roughly 32% to 46%. Nearly half of all U.S. adults now have hypertension, bringing the total national hypertensive population to a staggering 103 million.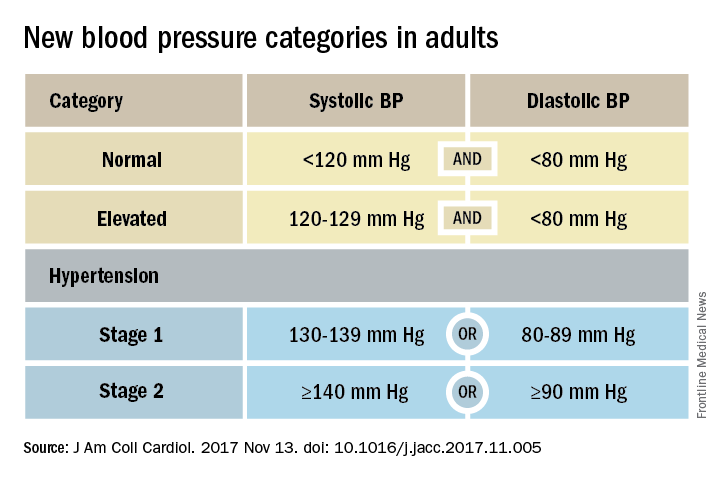
Goal is to transform care
But the new guidelines (J Am Coll Cardiol. 2017 Nov 13. doi: 10.1016/j.jacc.2017.11.005) for preventing, detecting, evaluating, and managing adult hypertension do lots more than just shake up the epidemiology of high blood pressure. With 106 total recommendations, the guidelines seek to transform every aspect of blood pressure in American medical practice, starting with how it’s measured and stretching to redefine applications of medical systems to try to ensure that every person with a blood pressure that truly falls outside the redefined limits gets a comprehensive package of interventions.
Many of these are “seismic changes,” said Lawrence J. Appel, MD. He particularly cited as seismic the new classification of stage 1 hypertension as a pressure at or above 130/80 mm Hg, the emphasis on using some form of out-of-office blood pressure measurement to confirm a diagnosis, the use of risk assessment when deciding whether to treat certain patients with drugs, and the same blood pressure goal of less than 130/80 mm Hg for all hypertensives, regardless of age, as long as they remain ambulatory and community dwelling.
One goal for all adults
“The systolic blood pressure goal for older people has gone from 140 mm Hg to 150 mm Hg and now to 130 mm Hg in the space of 2-3 years,” commented Dr. Appel, professor of epidemiology at Johns Hopkins University in Baltimore and not involved in the guideline-writing process.
In fact, the guidelines simplified the treatment goal all around, to less than 130/80 mm Hg for patients with diabetes, those with chronic kidney disease, and the elderly; that goal remains the same for all adults.
“It will be clearer and easier now that everyone should be less than 130/80 mm Hg. You won’t need to remember a second target,” said Sandra J. Taler, MD, a nephrologist and professor of medicine at the Mayo Clinic in Rochester, Minn., and a member of the guidelines task force.
“Some people may be upset that we changed the rules on them. They had normal blood pressure yesterday, and today it’s high. But it’s a good awakening, especially for using lifestyle interventions,” Dr. Taler said in an interview.
Preferred intervention: Lifestyle, not drugs
Lifestyle optimization is repeatedly cited as the cornerstone of intervention for everyone, including those with elevated blood pressure with a systolic pressure of 120-129 mm Hg, and as the only endorsed intervention for patients with hypertension of 130-139 mm Hg but below a 10% risk for a cardiovascular disease event during the next 10 years on the American College of Cardiology’s online risk calculator. The guidelines list six lifestyle goals: weight loss, following a DASH diet, reducing sodium, enhancing potassium, 90-150 min/wk of physical activity, and moderate alcohol intake.
Team-based care essential
The guidelines also put unprecedented emphasis on using a team-based management approach, which means having nurses, nurse practitioners, pharmacists, dietitians, and other clinicians, allowing for more frequent and focused care. Dr. Whelton and others cited in particular the VA Health System and Kaiser-Permanente as operating team-based and system-driven blood pressure management programs that have resulted in control rates for more than 90% of hypertensive patients. The team-based approach is also a key in the Target:BP program that the American Heart Association and American Medical Association founded. Target:BP will be instrumental in promoting implementation of the new guidelines, Dr. Carey said. Another systems recommendation is that every patient with hypertension should have a “clear, detailed, and current evidence-based plan of care.”
“Using nurse practitioners, physician assistants, and pharmacists has been shown to improve blood pressure levels,” and health systems that use this approach have had “great success,” commented Donald M. Lloyd-Jones, MD, professor and chairman of preventive medicine at Northwestern University in Chicago and not part of the guidelines task force. Some systems have used this approach to achieve high levels of blood pressure control. Now that financial penalties and incentives from payers also exist to push for higher levels of blood pressure control, the alignment of financial and health incentives should result in big changes, Dr. Lloyd-Jones predicted in a video interview.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
ANAHEIM, CALIF. – Thirty million Americans became hypertensive overnight on Nov. 13 with the introduction of new high blood pressure guidelines from the American College of Cardiology and American Heart Association.
That happened by resetting the definition of adult hypertension from the long-standing threshold of 140/90 mm Hg to a blood pressure at or above 130/80 mm Hg, a change that jumps the U.S. adult prevalence of hypertension from roughly 32% to 46%. Nearly half of all U.S. adults now have hypertension, bringing the total national hypertensive population to a staggering 103 million.
Goal is to transform care
But the new guidelines (J Am Coll Cardiol. 2017 Nov 13. doi: 10.1016/j.jacc.2017.11.005) for preventing, detecting, evaluating, and managing adult hypertension do lots more than just shake up the epidemiology of high blood pressure. With 106 total recommendations, the guidelines seek to transform every aspect of blood pressure in American medical practice, starting with how it’s measured and stretching to redefine applications of medical systems to try to ensure that every person with a blood pressure that truly falls outside the redefined limits gets a comprehensive package of interventions.
Many of these are “seismic changes,” said Lawrence J. Appel, MD. He particularly cited as seismic the new classification of stage 1 hypertension as a pressure at or above 130/80 mm Hg, the emphasis on using some form of out-of-office blood pressure measurement to confirm a diagnosis, the use of risk assessment when deciding whether to treat certain patients with drugs, and the same blood pressure goal of less than 130/80 mm Hg for all hypertensives, regardless of age, as long as they remain ambulatory and community dwelling.
One goal for all adults
“The systolic blood pressure goal for older people has gone from 140 mm Hg to 150 mm Hg and now to 130 mm Hg in the space of 2-3 years,” commented Dr. Appel, professor of epidemiology at Johns Hopkins University in Baltimore and not involved in the guideline-writing process.
In fact, the guidelines simplified the treatment goal all around, to less than 130/80 mm Hg for patients with diabetes, those with chronic kidney disease, and the elderly; that goal remains the same for all adults.
“It will be clearer and easier now that everyone should be less than 130/80 mm Hg. You won’t need to remember a second target,” said Sandra J. Taler, MD, a nephrologist and professor of medicine at the Mayo Clinic in Rochester, Minn., and a member of the guidelines task force.
“Some people may be upset that we changed the rules on them. They had normal blood pressure yesterday, and today it’s high. But it’s a good awakening, especially for using lifestyle interventions,” Dr. Taler said in an interview.
Preferred intervention: Lifestyle, not drugs
Lifestyle optimization is repeatedly cited as the cornerstone of intervention for everyone, including those with elevated blood pressure with a systolic pressure of 120-129 mm Hg, and as the only endorsed intervention for patients with hypertension of 130-139 mm Hg but below a 10% risk for a cardiovascular disease event during the next 10 years on the American College of Cardiology’s online risk calculator. The guidelines list six lifestyle goals: weight loss, following a DASH diet, reducing sodium, enhancing potassium, 90-150 min/wk of physical activity, and moderate alcohol intake.
Team-based care essential
The guidelines also put unprecedented emphasis on using a team-based management approach, which means having nurses, nurse practitioners, pharmacists, dietitians, and other clinicians, allowing for more frequent and focused care. Dr. Whelton and others cited in particular the VA Health System and Kaiser-Permanente as operating team-based and system-driven blood pressure management programs that have resulted in control rates for more than 90% of hypertensive patients. The team-based approach is also a key in the Target:BP program that the American Heart Association and American Medical Association founded. Target:BP will be instrumental in promoting implementation of the new guidelines, Dr. Carey said. Another systems recommendation is that every patient with hypertension should have a “clear, detailed, and current evidence-based plan of care.”
“Using nurse practitioners, physician assistants, and pharmacists has been shown to improve blood pressure levels,” and health systems that use this approach have had “great success,” commented Donald M. Lloyd-Jones, MD, professor and chairman of preventive medicine at Northwestern University in Chicago and not part of the guidelines task force. Some systems have used this approach to achieve high levels of blood pressure control. Now that financial penalties and incentives from payers also exist to push for higher levels of blood pressure control, the alignment of financial and health incentives should result in big changes, Dr. Lloyd-Jones predicted in a video interview.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
ANAHEIM, CALIF. – Thirty million Americans became hypertensive overnight on Nov. 13 with the introduction of new high blood pressure guidelines from the American College of Cardiology and American Heart Association.
That happened by resetting the definition of adult hypertension from the long-standing threshold of 140/90 mm Hg to a blood pressure at or above 130/80 mm Hg, a change that jumps the U.S. adult prevalence of hypertension from roughly 32% to 46%. Nearly half of all U.S. adults now have hypertension, bringing the total national hypertensive population to a staggering 103 million.
Goal is to transform care
But the new guidelines (J Am Coll Cardiol. 2017 Nov 13. doi: 10.1016/j.jacc.2017.11.005) for preventing, detecting, evaluating, and managing adult hypertension do lots more than just shake up the epidemiology of high blood pressure. With 106 total recommendations, the guidelines seek to transform every aspect of blood pressure in American medical practice, starting with how it’s measured and stretching to redefine applications of medical systems to try to ensure that every person with a blood pressure that truly falls outside the redefined limits gets a comprehensive package of interventions.
Many of these are “seismic changes,” said Lawrence J. Appel, MD. He particularly cited as seismic the new classification of stage 1 hypertension as a pressure at or above 130/80 mm Hg, the emphasis on using some form of out-of-office blood pressure measurement to confirm a diagnosis, the use of risk assessment when deciding whether to treat certain patients with drugs, and the same blood pressure goal of less than 130/80 mm Hg for all hypertensives, regardless of age, as long as they remain ambulatory and community dwelling.
One goal for all adults
“The systolic blood pressure goal for older people has gone from 140 mm Hg to 150 mm Hg and now to 130 mm Hg in the space of 2-3 years,” commented Dr. Appel, professor of epidemiology at Johns Hopkins University in Baltimore and not involved in the guideline-writing process.
In fact, the guidelines simplified the treatment goal all around, to less than 130/80 mm Hg for patients with diabetes, those with chronic kidney disease, and the elderly; that goal remains the same for all adults.
“It will be clearer and easier now that everyone should be less than 130/80 mm Hg. You won’t need to remember a second target,” said Sandra J. Taler, MD, a nephrologist and professor of medicine at the Mayo Clinic in Rochester, Minn., and a member of the guidelines task force.
“Some people may be upset that we changed the rules on them. They had normal blood pressure yesterday, and today it’s high. But it’s a good awakening, especially for using lifestyle interventions,” Dr. Taler said in an interview.
Preferred intervention: Lifestyle, not drugs
Lifestyle optimization is repeatedly cited as the cornerstone of intervention for everyone, including those with elevated blood pressure with a systolic pressure of 120-129 mm Hg, and as the only endorsed intervention for patients with hypertension of 130-139 mm Hg but below a 10% risk for a cardiovascular disease event during the next 10 years on the American College of Cardiology’s online risk calculator. The guidelines list six lifestyle goals: weight loss, following a DASH diet, reducing sodium, enhancing potassium, 90-150 min/wk of physical activity, and moderate alcohol intake.
Team-based care essential
The guidelines also put unprecedented emphasis on using a team-based management approach, which means having nurses, nurse practitioners, pharmacists, dietitians, and other clinicians, allowing for more frequent and focused care. Dr. Whelton and others cited in particular the VA Health System and Kaiser-Permanente as operating team-based and system-driven blood pressure management programs that have resulted in control rates for more than 90% of hypertensive patients. The team-based approach is also a key in the Target:BP program that the American Heart Association and American Medical Association founded. Target:BP will be instrumental in promoting implementation of the new guidelines, Dr. Carey said. Another systems recommendation is that every patient with hypertension should have a “clear, detailed, and current evidence-based plan of care.”
“Using nurse practitioners, physician assistants, and pharmacists has been shown to improve blood pressure levels,” and health systems that use this approach have had “great success,” commented Donald M. Lloyd-Jones, MD, professor and chairman of preventive medicine at Northwestern University in Chicago and not part of the guidelines task force. Some systems have used this approach to achieve high levels of blood pressure control. Now that financial penalties and incentives from payers also exist to push for higher levels of blood pressure control, the alignment of financial and health incentives should result in big changes, Dr. Lloyd-Jones predicted in a video interview.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
EXPERT ANALYSIS FROM THE AHA SCIENTIFIC SESSIONS
Targeting PCSK9 inhibitors to reap most benefit
ANAHEIM, CALIF. – Patients with symptomatic peripheral artery disease or a high-risk history of MI got the biggest bang for the buck from aggressive LDL cholesterol lowering with evolocumab in two new prespecified subgroup analyses from the landmark FOURIER trial presented at the American Heart Association scientific sessions.
“At the end of the day, not all of our patients with ASCVD [atherosclerotic cardiovascular disease] can have these expensive medications. These subgroup analyses will help clinicians to target use of PCSK9 inhibitors to the patients who will benefit the most,” Lynne T. Braun, PhD, commented in her role as discussant of the two secondary analyses, presented back to back in a late-breaking science session. Dr. Braun is a professor in the department of internal medicine at Rush University, Chicago.
The FOURIER trial included 27,564 high-risk patients with prior MI, stroke, and/or symptomatic peripheral arterial disease (PAD) who had an LDL cholesterol level of 70 mg/dL or more on high- or moderate-intensity statin therapy. They were randomized in double-blind fashion to add-on subcutaneous evolocumab (Repatha) at either 140 mg every 2 weeks or 420 mg/month or to placebo, for a median of 2.5 years of follow-up. The evolocumab group experienced a 59% reduction in LDL cholesterol, compared with the controls on background statin therapy plus placebo, down to a mean LDL cholesterol level of just 30 mg/dL.
As previously reported, the risk of the primary composite endpoint – comprising cardiovascular death, MI, stroke, unstable angina, or coronary revascularization – was reduced by 15% in the evolocumab group at 3 years. The secondary endpoint of cardiovascular death, MI, or stroke was reduced by 20%, from 9.9% to 7.9% (N Engl J Med. 2017;376:1713-22).
Evolocumab tamed PAD
At the AHA scientific sessions, Marc P. Bonaca, MD, presented a secondary analysis restricted to the 3,642 FOURIER participants with symptomatic PAD. The goal was to answer two unresolved questions: Does LDL cholesterol lowering beyond what’s achievable with a statin further reduce PAD patients’ cardiovascular risk? And does it reduce their risk of major adverse leg events (MALE), defined as a composite of acute limb ischemia, major amputation, and urgent revascularization?
The rate of the composite endpoint comprising cardiovascular death, MI, or stroke was 13% over 3 years in PAD patients randomized to placebo, which was 81% greater than the 7.6% rate in placebo-treated participants with a baseline history of stroke or MI but no PAD, in an analysis adjusted for demographics, cardiovascular risk factors, kidney function, body mass index, and prior revascularization.
The event rate was even higher in patients with PAD plus a history of MI or stroke, at 14.9%. Evolocumab reduced that risk by 27%, compared with placebo in patients with PAD, for an absolute risk reduction of 3.5% and a number-needed-to-treat (NNT) of 29 for 2.5 years.
The benefit of evolocumab was even more pronounced in the subgroup of 1,505 patients with baseline PAD but no prior MI or stroke: a 43% relative risk reduction, from 10.3% to 5.5%, for an absolute risk reduction of 4.8% and a NNT of 21.
A linear relationship was seen between the MALE rate during follow-up and LDL cholesterol level after 1 month of therapy, down to an LDL cholesterol level of less than 10 mg/dL. The clinically relevant composite endpoint of MACE (major adverse cardiovascular events – a composite of cardiovascular death, MI, and stroke) or MALE in patients with baseline PAD but no history of MI or stroke occurred in 12.8% of controls and 6.5% of the evolocumab group. This translated to a 48% relative risk reduction, a 6.3% absolute risk reduction, and a NNT of 16. The event curves in the evolocumab and control arms separated quite early, within the first 90 days of treatment.
The take home message: “LDL reduction to very low levels should be considered in patients with PAD, regardless of their history of MI or stroke, to reduce the risk of MACE [major adverse cardiovascular event] and MALE,” declared Dr. Bonaca of Brigham and Women’s Hospital and Harvard Medical School, both in Boston.
Spotting the patients with a history of MI who’re at highest risk
Marc S. Sabatine, MD, presented the subanalysis involving the 22,351 FOURIER patients with a prior MI. He and his coinvestigators identified three high-risk features within this group: an MI within the past 2 years, a history of two or more MIs, and residual multivessel CAD. Each of these three features was individually associated with a 34%-90% increased risk of MACE during follow-up. All told, 63% of FOURIER participants with prior MI had one or more of the high-risk features.
The use of evolocumab in patients with at least one of the three high-risk features was associated with a 22% relative risk reduction and an absolute 2.5% risk reduction, compared with placebo. The event curves diverged at about 6 months, and the gap between them steadily widened during follow-up. Extrapolating from this pattern, it’s likely that evolocumab would achieve an absolute 5% risk reduction in MACE, compared with placebo over 5 years, with an NNT of 20, according to Dr. Sabatine, professor of medicine at Harvard Medical School and chairman of the Thrombolysis in Myocardial Infarction (TIMI) Study Group.
Lingering questions
Dr. Braun was particularly impressed that the absolute risk reduction in MACE was even larger in patients with baseline PAD but no history of stroke or MI than in PAD patients with such a history. She added that, while she recognizes the value of selecting objectively assessable hard clinical MACE as the primary endpoint in FOURIER, her own patients care even more about other outcomes.
“What my patients with PAD care most about is whether profound LDL lowering translates to less claudication, improved quality of life, and greater physical activity tolerance. These were prespecified secondary outcomes in FOURIER, and I look forward to future reports addressing those issues,” she said.
Another unanswered question involves the mechanism by which intensive LDL cholesterol lowering results in fewer MACE and MALE events in high-risk subgroups. The possibilities include the plaque regression that was documented in the GLAGOV trial, an anti-inflammatory plaque-stabilizing effect being exerted through PCSK9 inhibition, or perhaps the PCSK9 inhibitors’ ability to moderately lower lipoprotein(a) cholesterol levels.
Simultaneous with Dr. Bonaca’s presentation at the AHA, the FOURIER PAD analysis was published online in Circulation (2017 Nov 13; doi: 10.1161/CIRCULATIONAHA.117.032235).
The FOURIER trial was sponsored by Amgen. Dr. Bonaca and Dr. Sabatine reported receiving research grants from and serving as consultants to Amgen and other companies.
ANAHEIM, CALIF. – Patients with symptomatic peripheral artery disease or a high-risk history of MI got the biggest bang for the buck from aggressive LDL cholesterol lowering with evolocumab in two new prespecified subgroup analyses from the landmark FOURIER trial presented at the American Heart Association scientific sessions.
“At the end of the day, not all of our patients with ASCVD [atherosclerotic cardiovascular disease] can have these expensive medications. These subgroup analyses will help clinicians to target use of PCSK9 inhibitors to the patients who will benefit the most,” Lynne T. Braun, PhD, commented in her role as discussant of the two secondary analyses, presented back to back in a late-breaking science session. Dr. Braun is a professor in the department of internal medicine at Rush University, Chicago.
The FOURIER trial included 27,564 high-risk patients with prior MI, stroke, and/or symptomatic peripheral arterial disease (PAD) who had an LDL cholesterol level of 70 mg/dL or more on high- or moderate-intensity statin therapy. They were randomized in double-blind fashion to add-on subcutaneous evolocumab (Repatha) at either 140 mg every 2 weeks or 420 mg/month or to placebo, for a median of 2.5 years of follow-up. The evolocumab group experienced a 59% reduction in LDL cholesterol, compared with the controls on background statin therapy plus placebo, down to a mean LDL cholesterol level of just 30 mg/dL.
As previously reported, the risk of the primary composite endpoint – comprising cardiovascular death, MI, stroke, unstable angina, or coronary revascularization – was reduced by 15% in the evolocumab group at 3 years. The secondary endpoint of cardiovascular death, MI, or stroke was reduced by 20%, from 9.9% to 7.9% (N Engl J Med. 2017;376:1713-22).
Evolocumab tamed PAD
At the AHA scientific sessions, Marc P. Bonaca, MD, presented a secondary analysis restricted to the 3,642 FOURIER participants with symptomatic PAD. The goal was to answer two unresolved questions: Does LDL cholesterol lowering beyond what’s achievable with a statin further reduce PAD patients’ cardiovascular risk? And does it reduce their risk of major adverse leg events (MALE), defined as a composite of acute limb ischemia, major amputation, and urgent revascularization?
The rate of the composite endpoint comprising cardiovascular death, MI, or stroke was 13% over 3 years in PAD patients randomized to placebo, which was 81% greater than the 7.6% rate in placebo-treated participants with a baseline history of stroke or MI but no PAD, in an analysis adjusted for demographics, cardiovascular risk factors, kidney function, body mass index, and prior revascularization.
The event rate was even higher in patients with PAD plus a history of MI or stroke, at 14.9%. Evolocumab reduced that risk by 27%, compared with placebo in patients with PAD, for an absolute risk reduction of 3.5% and a number-needed-to-treat (NNT) of 29 for 2.5 years.
The benefit of evolocumab was even more pronounced in the subgroup of 1,505 patients with baseline PAD but no prior MI or stroke: a 43% relative risk reduction, from 10.3% to 5.5%, for an absolute risk reduction of 4.8% and a NNT of 21.
A linear relationship was seen between the MALE rate during follow-up and LDL cholesterol level after 1 month of therapy, down to an LDL cholesterol level of less than 10 mg/dL. The clinically relevant composite endpoint of MACE (major adverse cardiovascular events – a composite of cardiovascular death, MI, and stroke) or MALE in patients with baseline PAD but no history of MI or stroke occurred in 12.8% of controls and 6.5% of the evolocumab group. This translated to a 48% relative risk reduction, a 6.3% absolute risk reduction, and a NNT of 16. The event curves in the evolocumab and control arms separated quite early, within the first 90 days of treatment.
The take home message: “LDL reduction to very low levels should be considered in patients with PAD, regardless of their history of MI or stroke, to reduce the risk of MACE [major adverse cardiovascular event] and MALE,” declared Dr. Bonaca of Brigham and Women’s Hospital and Harvard Medical School, both in Boston.
Spotting the patients with a history of MI who’re at highest risk
Marc S. Sabatine, MD, presented the subanalysis involving the 22,351 FOURIER patients with a prior MI. He and his coinvestigators identified three high-risk features within this group: an MI within the past 2 years, a history of two or more MIs, and residual multivessel CAD. Each of these three features was individually associated with a 34%-90% increased risk of MACE during follow-up. All told, 63% of FOURIER participants with prior MI had one or more of the high-risk features.
The use of evolocumab in patients with at least one of the three high-risk features was associated with a 22% relative risk reduction and an absolute 2.5% risk reduction, compared with placebo. The event curves diverged at about 6 months, and the gap between them steadily widened during follow-up. Extrapolating from this pattern, it’s likely that evolocumab would achieve an absolute 5% risk reduction in MACE, compared with placebo over 5 years, with an NNT of 20, according to Dr. Sabatine, professor of medicine at Harvard Medical School and chairman of the Thrombolysis in Myocardial Infarction (TIMI) Study Group.
Lingering questions
Dr. Braun was particularly impressed that the absolute risk reduction in MACE was even larger in patients with baseline PAD but no history of stroke or MI than in PAD patients with such a history. She added that, while she recognizes the value of selecting objectively assessable hard clinical MACE as the primary endpoint in FOURIER, her own patients care even more about other outcomes.
“What my patients with PAD care most about is whether profound LDL lowering translates to less claudication, improved quality of life, and greater physical activity tolerance. These were prespecified secondary outcomes in FOURIER, and I look forward to future reports addressing those issues,” she said.
Another unanswered question involves the mechanism by which intensive LDL cholesterol lowering results in fewer MACE and MALE events in high-risk subgroups. The possibilities include the plaque regression that was documented in the GLAGOV trial, an anti-inflammatory plaque-stabilizing effect being exerted through PCSK9 inhibition, or perhaps the PCSK9 inhibitors’ ability to moderately lower lipoprotein(a) cholesterol levels.
Simultaneous with Dr. Bonaca’s presentation at the AHA, the FOURIER PAD analysis was published online in Circulation (2017 Nov 13; doi: 10.1161/CIRCULATIONAHA.117.032235).
The FOURIER trial was sponsored by Amgen. Dr. Bonaca and Dr. Sabatine reported receiving research grants from and serving as consultants to Amgen and other companies.
ANAHEIM, CALIF. – Patients with symptomatic peripheral artery disease or a high-risk history of MI got the biggest bang for the buck from aggressive LDL cholesterol lowering with evolocumab in two new prespecified subgroup analyses from the landmark FOURIER trial presented at the American Heart Association scientific sessions.
“At the end of the day, not all of our patients with ASCVD [atherosclerotic cardiovascular disease] can have these expensive medications. These subgroup analyses will help clinicians to target use of PCSK9 inhibitors to the patients who will benefit the most,” Lynne T. Braun, PhD, commented in her role as discussant of the two secondary analyses, presented back to back in a late-breaking science session. Dr. Braun is a professor in the department of internal medicine at Rush University, Chicago.
The FOURIER trial included 27,564 high-risk patients with prior MI, stroke, and/or symptomatic peripheral arterial disease (PAD) who had an LDL cholesterol level of 70 mg/dL or more on high- or moderate-intensity statin therapy. They were randomized in double-blind fashion to add-on subcutaneous evolocumab (Repatha) at either 140 mg every 2 weeks or 420 mg/month or to placebo, for a median of 2.5 years of follow-up. The evolocumab group experienced a 59% reduction in LDL cholesterol, compared with the controls on background statin therapy plus placebo, down to a mean LDL cholesterol level of just 30 mg/dL.
As previously reported, the risk of the primary composite endpoint – comprising cardiovascular death, MI, stroke, unstable angina, or coronary revascularization – was reduced by 15% in the evolocumab group at 3 years. The secondary endpoint of cardiovascular death, MI, or stroke was reduced by 20%, from 9.9% to 7.9% (N Engl J Med. 2017;376:1713-22).
Evolocumab tamed PAD
At the AHA scientific sessions, Marc P. Bonaca, MD, presented a secondary analysis restricted to the 3,642 FOURIER participants with symptomatic PAD. The goal was to answer two unresolved questions: Does LDL cholesterol lowering beyond what’s achievable with a statin further reduce PAD patients’ cardiovascular risk? And does it reduce their risk of major adverse leg events (MALE), defined as a composite of acute limb ischemia, major amputation, and urgent revascularization?
The rate of the composite endpoint comprising cardiovascular death, MI, or stroke was 13% over 3 years in PAD patients randomized to placebo, which was 81% greater than the 7.6% rate in placebo-treated participants with a baseline history of stroke or MI but no PAD, in an analysis adjusted for demographics, cardiovascular risk factors, kidney function, body mass index, and prior revascularization.
The event rate was even higher in patients with PAD plus a history of MI or stroke, at 14.9%. Evolocumab reduced that risk by 27%, compared with placebo in patients with PAD, for an absolute risk reduction of 3.5% and a number-needed-to-treat (NNT) of 29 for 2.5 years.
The benefit of evolocumab was even more pronounced in the subgroup of 1,505 patients with baseline PAD but no prior MI or stroke: a 43% relative risk reduction, from 10.3% to 5.5%, for an absolute risk reduction of 4.8% and a NNT of 21.
A linear relationship was seen between the MALE rate during follow-up and LDL cholesterol level after 1 month of therapy, down to an LDL cholesterol level of less than 10 mg/dL. The clinically relevant composite endpoint of MACE (major adverse cardiovascular events – a composite of cardiovascular death, MI, and stroke) or MALE in patients with baseline PAD but no history of MI or stroke occurred in 12.8% of controls and 6.5% of the evolocumab group. This translated to a 48% relative risk reduction, a 6.3% absolute risk reduction, and a NNT of 16. The event curves in the evolocumab and control arms separated quite early, within the first 90 days of treatment.
The take home message: “LDL reduction to very low levels should be considered in patients with PAD, regardless of their history of MI or stroke, to reduce the risk of MACE [major adverse cardiovascular event] and MALE,” declared Dr. Bonaca of Brigham and Women’s Hospital and Harvard Medical School, both in Boston.
Spotting the patients with a history of MI who’re at highest risk
Marc S. Sabatine, MD, presented the subanalysis involving the 22,351 FOURIER patients with a prior MI. He and his coinvestigators identified three high-risk features within this group: an MI within the past 2 years, a history of two or more MIs, and residual multivessel CAD. Each of these three features was individually associated with a 34%-90% increased risk of MACE during follow-up. All told, 63% of FOURIER participants with prior MI had one or more of the high-risk features.
The use of evolocumab in patients with at least one of the three high-risk features was associated with a 22% relative risk reduction and an absolute 2.5% risk reduction, compared with placebo. The event curves diverged at about 6 months, and the gap between them steadily widened during follow-up. Extrapolating from this pattern, it’s likely that evolocumab would achieve an absolute 5% risk reduction in MACE, compared with placebo over 5 years, with an NNT of 20, according to Dr. Sabatine, professor of medicine at Harvard Medical School and chairman of the Thrombolysis in Myocardial Infarction (TIMI) Study Group.
Lingering questions
Dr. Braun was particularly impressed that the absolute risk reduction in MACE was even larger in patients with baseline PAD but no history of stroke or MI than in PAD patients with such a history. She added that, while she recognizes the value of selecting objectively assessable hard clinical MACE as the primary endpoint in FOURIER, her own patients care even more about other outcomes.
“What my patients with PAD care most about is whether profound LDL lowering translates to less claudication, improved quality of life, and greater physical activity tolerance. These were prespecified secondary outcomes in FOURIER, and I look forward to future reports addressing those issues,” she said.
Another unanswered question involves the mechanism by which intensive LDL cholesterol lowering results in fewer MACE and MALE events in high-risk subgroups. The possibilities include the plaque regression that was documented in the GLAGOV trial, an anti-inflammatory plaque-stabilizing effect being exerted through PCSK9 inhibition, or perhaps the PCSK9 inhibitors’ ability to moderately lower lipoprotein(a) cholesterol levels.
Simultaneous with Dr. Bonaca’s presentation at the AHA, the FOURIER PAD analysis was published online in Circulation (2017 Nov 13; doi: 10.1161/CIRCULATIONAHA.117.032235).
The FOURIER trial was sponsored by Amgen. Dr. Bonaca and Dr. Sabatine reported receiving research grants from and serving as consultants to Amgen and other companies.
EXPERT ANALYSIS FROM THE AHA SCIENTIFIC SESSIONS
Direct oral anticoagulants okay during AF device placement
ANAHEIM, CALIF. – Whether direct oral anticoagulants are continued or interrupted for device placement in atrial fibrillation patients, the risk of device pocket hematoma or stroke is very low, based on results of the BRUISE CONTROL–2 trial in more than 600 subjects.
Either strategy is reasonable depending on the clinical scenario, coprincipal investigator David Birnie, MD, said in presenting the results at the American Heart Association scientific sessions.
When atrial fibrillation (AF) patients on direct oral anticoagulants (DOACs) present for device surgery, there’s concern that keeping them on the drugs will increase the bleeding risk, but that taking them off will increase the stroke risk. “We sought to resolve this dilemma,” said Dr. Birnie, an electrophysiologist and director of the arrhythmia service at the University of Ottawa Heart Institute.
The subjects were on dabigatran, rivaroxaban, or apixaban, about a third in each group; 328 were randomized to continue their daily dosing, including on the day of surgery. The other 334 were randomized to interrupted treatment. For rivaroxaban and apixaban, that meant taking their last dose 2 days before surgery. Dabigatran patients discontinued the drug 1-2 days beforehand, depending on glomerular filtration rate. Patients resumed treatment about 24 hours after surgery. CHA2DS2-VASc scores were a mean of 3.9 in both arms, and at least 2 in all participants.
The rate of clinically significant hematoma – the primary outcome in the study, defined as a hematoma requiring prolonged hospitalization, interrupted postoperative anticoagulation, or reoperation to evacuate – was identical in both arms, 2.1% (seven patients each). There were two ischemic strokes, one in each arm. There was one delayed cardiac tamponade in the continuation arm and one pericardial effusion in the interrupted arm. The three deaths in the trial were not related to device placement.
So, what to do depends on the clinical scenario, Dr. Birnie said in an interview. If someone needs urgent placement and there’s no time to wait for DOAC washout, “it’s quite reasonable to go ahead.” Also, “if somebody is at extremely high risk for stroke, then it’s very reasonable to continue the drug.”
On the other hand, “if someone has a much lower stroke risk, then the risk-benefit ratio is probably in the opposite direction, so temporarily discontinuing the drug is the right thing to do,” he said.
Dr. Birnie cautioned that although continued DOAC may reduce the risk of thromboembolism, “this study was not designed with power to answer this.”
“We are already putting these findings into practice” in Ottawa, he said. “Our protocol” – as in many places – “ was always to stop anticoagulation for 2 or 3 days, but now, for very high-risk patients – high-risk AF, unstable temporary pacing, that type of thing – we are very comfortable continuing it,” he said. The study follows up a previous randomized trial by Dr. Birnie and his colleagues that pitted continued warfarin against heparin bridging for AF device placement. There were far fewer device pocket hematomas with uninterrupted warfarin (N Engl J Med. 2013 May 30;368[22]:2084-93).
The team wanted to repeat the study using DOACs, since their use has grown substantially, with the majority of AF patients now on them.
The arms in BRUISE CONTROL–2 (Strategy of Continued Versus Interrupted Novel Oral Anticoagulant at Time of Device Surgery in Patients With Moderate to High Risk of Arterial Thromboembolic Events) were well matched, with a mean age of about 74 years; men made up more than 70% of the subjects in both arms. About 17% of the participants were on chronic aspirin therapy and about 4% were on clopidogrel, in each arm. The uninterrupted DOAC group went about 14 hours between their last preop and first postop DOAC dose. The interrupted group went about 72 hours.
BRUISE CONTROL–2 was funded by the Heart and Stroke Foundation of Canada, Boehringer Ingelheim, Bayer, Pfizer, and Bristol-Myers Squibb, among others. Dr. Birnie had no relevant financial disclosures.
ANAHEIM, CALIF. – Whether direct oral anticoagulants are continued or interrupted for device placement in atrial fibrillation patients, the risk of device pocket hematoma or stroke is very low, based on results of the BRUISE CONTROL–2 trial in more than 600 subjects.
Either strategy is reasonable depending on the clinical scenario, coprincipal investigator David Birnie, MD, said in presenting the results at the American Heart Association scientific sessions.
When atrial fibrillation (AF) patients on direct oral anticoagulants (DOACs) present for device surgery, there’s concern that keeping them on the drugs will increase the bleeding risk, but that taking them off will increase the stroke risk. “We sought to resolve this dilemma,” said Dr. Birnie, an electrophysiologist and director of the arrhythmia service at the University of Ottawa Heart Institute.
The subjects were on dabigatran, rivaroxaban, or apixaban, about a third in each group; 328 were randomized to continue their daily dosing, including on the day of surgery. The other 334 were randomized to interrupted treatment. For rivaroxaban and apixaban, that meant taking their last dose 2 days before surgery. Dabigatran patients discontinued the drug 1-2 days beforehand, depending on glomerular filtration rate. Patients resumed treatment about 24 hours after surgery. CHA2DS2-VASc scores were a mean of 3.9 in both arms, and at least 2 in all participants.
The rate of clinically significant hematoma – the primary outcome in the study, defined as a hematoma requiring prolonged hospitalization, interrupted postoperative anticoagulation, or reoperation to evacuate – was identical in both arms, 2.1% (seven patients each). There were two ischemic strokes, one in each arm. There was one delayed cardiac tamponade in the continuation arm and one pericardial effusion in the interrupted arm. The three deaths in the trial were not related to device placement.
So, what to do depends on the clinical scenario, Dr. Birnie said in an interview. If someone needs urgent placement and there’s no time to wait for DOAC washout, “it’s quite reasonable to go ahead.” Also, “if somebody is at extremely high risk for stroke, then it’s very reasonable to continue the drug.”
On the other hand, “if someone has a much lower stroke risk, then the risk-benefit ratio is probably in the opposite direction, so temporarily discontinuing the drug is the right thing to do,” he said.
Dr. Birnie cautioned that although continued DOAC may reduce the risk of thromboembolism, “this study was not designed with power to answer this.”
“We are already putting these findings into practice” in Ottawa, he said. “Our protocol” – as in many places – “ was always to stop anticoagulation for 2 or 3 days, but now, for very high-risk patients – high-risk AF, unstable temporary pacing, that type of thing – we are very comfortable continuing it,” he said. The study follows up a previous randomized trial by Dr. Birnie and his colleagues that pitted continued warfarin against heparin bridging for AF device placement. There were far fewer device pocket hematomas with uninterrupted warfarin (N Engl J Med. 2013 May 30;368[22]:2084-93).
The team wanted to repeat the study using DOACs, since their use has grown substantially, with the majority of AF patients now on them.
The arms in BRUISE CONTROL–2 (Strategy of Continued Versus Interrupted Novel Oral Anticoagulant at Time of Device Surgery in Patients With Moderate to High Risk of Arterial Thromboembolic Events) were well matched, with a mean age of about 74 years; men made up more than 70% of the subjects in both arms. About 17% of the participants were on chronic aspirin therapy and about 4% were on clopidogrel, in each arm. The uninterrupted DOAC group went about 14 hours between their last preop and first postop DOAC dose. The interrupted group went about 72 hours.
BRUISE CONTROL–2 was funded by the Heart and Stroke Foundation of Canada, Boehringer Ingelheim, Bayer, Pfizer, and Bristol-Myers Squibb, among others. Dr. Birnie had no relevant financial disclosures.
ANAHEIM, CALIF. – Whether direct oral anticoagulants are continued or interrupted for device placement in atrial fibrillation patients, the risk of device pocket hematoma or stroke is very low, based on results of the BRUISE CONTROL–2 trial in more than 600 subjects.
Either strategy is reasonable depending on the clinical scenario, coprincipal investigator David Birnie, MD, said in presenting the results at the American Heart Association scientific sessions.
When atrial fibrillation (AF) patients on direct oral anticoagulants (DOACs) present for device surgery, there’s concern that keeping them on the drugs will increase the bleeding risk, but that taking them off will increase the stroke risk. “We sought to resolve this dilemma,” said Dr. Birnie, an electrophysiologist and director of the arrhythmia service at the University of Ottawa Heart Institute.
The subjects were on dabigatran, rivaroxaban, or apixaban, about a third in each group; 328 were randomized to continue their daily dosing, including on the day of surgery. The other 334 were randomized to interrupted treatment. For rivaroxaban and apixaban, that meant taking their last dose 2 days before surgery. Dabigatran patients discontinued the drug 1-2 days beforehand, depending on glomerular filtration rate. Patients resumed treatment about 24 hours after surgery. CHA2DS2-VASc scores were a mean of 3.9 in both arms, and at least 2 in all participants.
The rate of clinically significant hematoma – the primary outcome in the study, defined as a hematoma requiring prolonged hospitalization, interrupted postoperative anticoagulation, or reoperation to evacuate – was identical in both arms, 2.1% (seven patients each). There were two ischemic strokes, one in each arm. There was one delayed cardiac tamponade in the continuation arm and one pericardial effusion in the interrupted arm. The three deaths in the trial were not related to device placement.
So, what to do depends on the clinical scenario, Dr. Birnie said in an interview. If someone needs urgent placement and there’s no time to wait for DOAC washout, “it’s quite reasonable to go ahead.” Also, “if somebody is at extremely high risk for stroke, then it’s very reasonable to continue the drug.”
On the other hand, “if someone has a much lower stroke risk, then the risk-benefit ratio is probably in the opposite direction, so temporarily discontinuing the drug is the right thing to do,” he said.
Dr. Birnie cautioned that although continued DOAC may reduce the risk of thromboembolism, “this study was not designed with power to answer this.”
“We are already putting these findings into practice” in Ottawa, he said. “Our protocol” – as in many places – “ was always to stop anticoagulation for 2 or 3 days, but now, for very high-risk patients – high-risk AF, unstable temporary pacing, that type of thing – we are very comfortable continuing it,” he said. The study follows up a previous randomized trial by Dr. Birnie and his colleagues that pitted continued warfarin against heparin bridging for AF device placement. There were far fewer device pocket hematomas with uninterrupted warfarin (N Engl J Med. 2013 May 30;368[22]:2084-93).
The team wanted to repeat the study using DOACs, since their use has grown substantially, with the majority of AF patients now on them.
The arms in BRUISE CONTROL–2 (Strategy of Continued Versus Interrupted Novel Oral Anticoagulant at Time of Device Surgery in Patients With Moderate to High Risk of Arterial Thromboembolic Events) were well matched, with a mean age of about 74 years; men made up more than 70% of the subjects in both arms. About 17% of the participants were on chronic aspirin therapy and about 4% were on clopidogrel, in each arm. The uninterrupted DOAC group went about 14 hours between their last preop and first postop DOAC dose. The interrupted group went about 72 hours.
BRUISE CONTROL–2 was funded by the Heart and Stroke Foundation of Canada, Boehringer Ingelheim, Bayer, Pfizer, and Bristol-Myers Squibb, among others. Dr. Birnie had no relevant financial disclosures.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point:
Major finding: The rate of clinically significant hematoma was identical in both arms, at 2.1% (seven patients each).
Data source: BRUISE CONTROL-2, a randomized trial with more than 600 subjects.
Disclosures: The work was funded by the Heart and Stroke Foundation of Canada, Boehringer Ingelheim, Bayer, Pfizer, and Bristol-Myers Squibb, among others. The presenter had no relevant financial disclosures.
DAPT produces better CABG outcomes than aspirin alone
ANAHEIM, CALIF. – Treatment with dual-antiplatelet therapy following coronary artery bypass grafting with a saphenous vein maintained vein-graft patency better than aspirin alone in a randomized, multicenter trial with 500 patients.
After 1 year of dual-antiplatelet therapy (DAPT) with ticagrelor (Brilinta) and aspirin, 89% of saphenous-vein grafts remained patent, compared with a 77% patency rate in saphenous-vein grafts in patients treated with aspirin alone, a statistically significant difference for the study’s primary endpoint, Qiang Zhao, MD, said at the American Hart Association scientific sessions. The data, collected at six Chinese centers, also showed a nominal decrease in the combined rate of cardiovascular death, MI, and stroke: 2% with DAPT and 5% with aspirin alone. It further showed an increase in major or bypass-related bleeds: 2% with DAPT and none with aspirin alone, reported Dr. Zhao, professor and director of cardiac surgery at Ruijin Hospital in Shanghai, China.
“If this result were repeated in a larger study it would be important,” John H. Alexander, MD, professor of medicine at Duke University in Durham, N.C., commented in a video interview.
The Compare the Efficacy of Different Antiplatelet Therapy Strategy After Coronary Artery Bypass Graft Surgery (DACAB) trial randomized patients who underwent coronary artery bypass grafting (CABG). They averaged about 64 years of age, and received an average of nearly four grafts each including an average of nearly three saphenous vein grafts. The study assigned patients to one of three treatment arms starting within 24 hours after surgery: 168 received ticagrelor 90 mg twice daily plus aspirin 100 mg once daily, 166 got ticagrelor alone, and 166 received aspirin alone. Treatment continued for 1 year.
“Some surgeons and physicians currently prescribe DAPT to CABG patients, but there is not much evidence of its benefit. The DACAB trial is useful, but you need to show that it does not just improve patency but that patients also have better outcomes. The excess of major bleeds is a big deal. It gives one pause about adopting DAPT as standard treatment,” Dr. Gardner said.
DACAB received no commercial funding. Dr. Zhao has been a speaker on behalf of and has received research funding from AstraZeneca, the company that markets ticagrelor (Brilinta). He has also been a speaker for Johnson & Johnson and Medtronic and has received research funding from Bayer, Novartis, and Sanofi. Dr. Gardner had no disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
Results from the DACAB trial showed that using aspirin and ticagrelor improved vein-graft patency, compared with using aspirin alone. It was a compelling result, but for the intermediate, imaging-based outcome of graft patency at 1 year after surgery. This finding is conclusive evidence that dual-antiplatelet therapy has some benefit.
But the findings from this trial, modestly sized with 500 patients, failed to prove that the clinical benefit from dual-antiplatelet therapy was worth the adverse effect of an increase in the rate of major and bypass-related bleeding. The study was underpowered to prove that dual-antiplatelet therapy had a clear beneficial impact on clinical outcomes such as cardiovascular death, MI, and stroke, although this combined rate went in the right direction with dual therapy, compared with aspirin alone. We need to see proof of a benefit for these clinical outcomes to justify using a treatment that causes an increase in major bleeds.
The DACAB results alone are not enough to justify a change in practice. It would be an important finding if the results could be replicated in a larger study. And if dual-antiplatelet therapy was proven to have a net clinical benefit for patients, we would still want to target it to patients with a higher ischemic risk and, in general, avoid using it in patients with a high bleeding risk.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
John H. Alexander, MD , is a cardiologist and professor of medicine at Duke University in Durham, N.C. He has been a consultant to and has received research funding from several companies, including AstraZeneca, the company that markets ticagrelor (Brilinta). He made these comments as designated discussant for the DACAB study and in a video interview .
Results from the DACAB trial showed that using aspirin and ticagrelor improved vein-graft patency, compared with using aspirin alone. It was a compelling result, but for the intermediate, imaging-based outcome of graft patency at 1 year after surgery. This finding is conclusive evidence that dual-antiplatelet therapy has some benefit.
But the findings from this trial, modestly sized with 500 patients, failed to prove that the clinical benefit from dual-antiplatelet therapy was worth the adverse effect of an increase in the rate of major and bypass-related bleeding. The study was underpowered to prove that dual-antiplatelet therapy had a clear beneficial impact on clinical outcomes such as cardiovascular death, MI, and stroke, although this combined rate went in the right direction with dual therapy, compared with aspirin alone. We need to see proof of a benefit for these clinical outcomes to justify using a treatment that causes an increase in major bleeds.
The DACAB results alone are not enough to justify a change in practice. It would be an important finding if the results could be replicated in a larger study. And if dual-antiplatelet therapy was proven to have a net clinical benefit for patients, we would still want to target it to patients with a higher ischemic risk and, in general, avoid using it in patients with a high bleeding risk.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
John H. Alexander, MD , is a cardiologist and professor of medicine at Duke University in Durham, N.C. He has been a consultant to and has received research funding from several companies, including AstraZeneca, the company that markets ticagrelor (Brilinta). He made these comments as designated discussant for the DACAB study and in a video interview .
Results from the DACAB trial showed that using aspirin and ticagrelor improved vein-graft patency, compared with using aspirin alone. It was a compelling result, but for the intermediate, imaging-based outcome of graft patency at 1 year after surgery. This finding is conclusive evidence that dual-antiplatelet therapy has some benefit.
But the findings from this trial, modestly sized with 500 patients, failed to prove that the clinical benefit from dual-antiplatelet therapy was worth the adverse effect of an increase in the rate of major and bypass-related bleeding. The study was underpowered to prove that dual-antiplatelet therapy had a clear beneficial impact on clinical outcomes such as cardiovascular death, MI, and stroke, although this combined rate went in the right direction with dual therapy, compared with aspirin alone. We need to see proof of a benefit for these clinical outcomes to justify using a treatment that causes an increase in major bleeds.
The DACAB results alone are not enough to justify a change in practice. It would be an important finding if the results could be replicated in a larger study. And if dual-antiplatelet therapy was proven to have a net clinical benefit for patients, we would still want to target it to patients with a higher ischemic risk and, in general, avoid using it in patients with a high bleeding risk.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
John H. Alexander, MD , is a cardiologist and professor of medicine at Duke University in Durham, N.C. He has been a consultant to and has received research funding from several companies, including AstraZeneca, the company that markets ticagrelor (Brilinta). He made these comments as designated discussant for the DACAB study and in a video interview .
ANAHEIM, CALIF. – Treatment with dual-antiplatelet therapy following coronary artery bypass grafting with a saphenous vein maintained vein-graft patency better than aspirin alone in a randomized, multicenter trial with 500 patients.
After 1 year of dual-antiplatelet therapy (DAPT) with ticagrelor (Brilinta) and aspirin, 89% of saphenous-vein grafts remained patent, compared with a 77% patency rate in saphenous-vein grafts in patients treated with aspirin alone, a statistically significant difference for the study’s primary endpoint, Qiang Zhao, MD, said at the American Hart Association scientific sessions. The data, collected at six Chinese centers, also showed a nominal decrease in the combined rate of cardiovascular death, MI, and stroke: 2% with DAPT and 5% with aspirin alone. It further showed an increase in major or bypass-related bleeds: 2% with DAPT and none with aspirin alone, reported Dr. Zhao, professor and director of cardiac surgery at Ruijin Hospital in Shanghai, China.
“If this result were repeated in a larger study it would be important,” John H. Alexander, MD, professor of medicine at Duke University in Durham, N.C., commented in a video interview.
The Compare the Efficacy of Different Antiplatelet Therapy Strategy After Coronary Artery Bypass Graft Surgery (DACAB) trial randomized patients who underwent coronary artery bypass grafting (CABG). They averaged about 64 years of age, and received an average of nearly four grafts each including an average of nearly three saphenous vein grafts. The study assigned patients to one of three treatment arms starting within 24 hours after surgery: 168 received ticagrelor 90 mg twice daily plus aspirin 100 mg once daily, 166 got ticagrelor alone, and 166 received aspirin alone. Treatment continued for 1 year.
“Some surgeons and physicians currently prescribe DAPT to CABG patients, but there is not much evidence of its benefit. The DACAB trial is useful, but you need to show that it does not just improve patency but that patients also have better outcomes. The excess of major bleeds is a big deal. It gives one pause about adopting DAPT as standard treatment,” Dr. Gardner said.
DACAB received no commercial funding. Dr. Zhao has been a speaker on behalf of and has received research funding from AstraZeneca, the company that markets ticagrelor (Brilinta). He has also been a speaker for Johnson & Johnson and Medtronic and has received research funding from Bayer, Novartis, and Sanofi. Dr. Gardner had no disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
ANAHEIM, CALIF. – Treatment with dual-antiplatelet therapy following coronary artery bypass grafting with a saphenous vein maintained vein-graft patency better than aspirin alone in a randomized, multicenter trial with 500 patients.
After 1 year of dual-antiplatelet therapy (DAPT) with ticagrelor (Brilinta) and aspirin, 89% of saphenous-vein grafts remained patent, compared with a 77% patency rate in saphenous-vein grafts in patients treated with aspirin alone, a statistically significant difference for the study’s primary endpoint, Qiang Zhao, MD, said at the American Hart Association scientific sessions. The data, collected at six Chinese centers, also showed a nominal decrease in the combined rate of cardiovascular death, MI, and stroke: 2% with DAPT and 5% with aspirin alone. It further showed an increase in major or bypass-related bleeds: 2% with DAPT and none with aspirin alone, reported Dr. Zhao, professor and director of cardiac surgery at Ruijin Hospital in Shanghai, China.
“If this result were repeated in a larger study it would be important,” John H. Alexander, MD, professor of medicine at Duke University in Durham, N.C., commented in a video interview.
The Compare the Efficacy of Different Antiplatelet Therapy Strategy After Coronary Artery Bypass Graft Surgery (DACAB) trial randomized patients who underwent coronary artery bypass grafting (CABG). They averaged about 64 years of age, and received an average of nearly four grafts each including an average of nearly three saphenous vein grafts. The study assigned patients to one of three treatment arms starting within 24 hours after surgery: 168 received ticagrelor 90 mg twice daily plus aspirin 100 mg once daily, 166 got ticagrelor alone, and 166 received aspirin alone. Treatment continued for 1 year.
“Some surgeons and physicians currently prescribe DAPT to CABG patients, but there is not much evidence of its benefit. The DACAB trial is useful, but you need to show that it does not just improve patency but that patients also have better outcomes. The excess of major bleeds is a big deal. It gives one pause about adopting DAPT as standard treatment,” Dr. Gardner said.
DACAB received no commercial funding. Dr. Zhao has been a speaker on behalf of and has received research funding from AstraZeneca, the company that markets ticagrelor (Brilinta). He has also been a speaker for Johnson & Johnson and Medtronic and has received research funding from Bayer, Novartis, and Sanofi. Dr. Gardner had no disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point:
Major finding: The 1-year saphenous-vein graft patency rate was 89% with DAPT treatment and 77% with aspirin alone. Data source: DACAB, a multicenter, randomized trial with 500 Chinese patients.
Disclosures: DACAB received no commercial funding. Dr. Zhao has been a speaker on behalf of and has received research funding from AstraZeneca, the company that markets ticagrelor (Brilinta). He has also been a speaker for Johnson & Johnson and Medtronic and has received research funding from Bayer, Novartis, and Sanofi.
Delay predicting outcome in comatose cardiac arrest
ANAHEIM, CALIF. – Withdrawal of life-sustaining systemic therapies in comatose patients after out-of-hospital cardiac arrest as advised in current guidelines often occurs too early, resulting in the death of many patients who could potentially survive with good outcome, according to the results of NORCAST, the Norwegian Cardiorespiratory Arrest Study.
“The take-home message is to be patient and wait. Three days may be too early to make decisions on the patient,” Kjetil Sunde, MD, said in presenting the study findings at the Resuscitation Science Symposium held during the American Heart Association scientific sessions.
The European Resuscitation Council and European Society of Intensive Care Medicine have jointly recommended a prognostic algorithm in which a multimodal assessment is made on patients who are still comatose on day 3 after cardiac arrest. But this advice is based on expert opinion and has never been validated. This was the impetus for the prospective NORCAST study.
Current practice in the management of out-of-hospital cardiac arrest patients who are comatose upon hospital admission is to induce therapeutic hypothermia, with targeted temperature management to 33° C for 24 hours under deep sedation. The study hypothesis was that this strategy delays the time to awakening and that, as a consequence, the recommended prognostic tests that are usually done on day 3 after withdrawal of sedation are rendered insufficiently reliable. Thus, decisions to withdraw life-supporting therapies at that point will reduce the survival potential of this population, Dr. Sunde explained.
NORCAST was a prospective observational study that included 259 patients admitted to Oslo University Hospital in a comatose state after out-of-hospital cardiac arrest. In this unselected group, 81% had a cardiac cause for their arrest; the remainder had hypoxic arrest. All patients underwent therapeutic hypothermia, then a period of nonhypothermia followed by sedation withdrawal.
All of the widely used multimodal prognostic tests were ordered, including serial measurement of serum neuron-specific enolase; neurophysiologic testing using EEG and sensory-evoked potential readings obtained both during hypothermia and again at least 3 days after sedation withdrawal; a standardized clinical neurologic exam including assessment of brainstem reflexes and a Glasgow Coma Scale rating 3 days after sedation withdrawal; and a transcranial Doppler study and cerebral MRI on day 5-7. However, the treatment team was blinded to the results of these tests and was encouraged to delay withdrawal of life-supporting therapies as long as possible.
Key findings
Out of 259 patients who were comatose upon admission, 54% were alive at 6 months – and 91% of them had a CPC of 1 or 2.
The final tally at 6 months: 44% of patients were CPC 1, 5.5% were CPC 2, 4% were CPC 3, meaning severely disabled, and 46.5% were CPC 5, which is brain dead.
Withdrawal of life-supporting therapies occurred in 73 patients, or 28%, and 71% of those patients died, few of them in the early days.
Among patients with a CPC score of 1 or 2 at 6 months, only 20% were awake on day 1-3 following admission. Fifty-seven percent awoke on day 4-7, but importantly, 23% of patients with a good outcome at 6 months were not yet awake on day 8.
Three days after withdrawal of sedation, 49% of patients were rated as having a Glasgow Coma Scale score of 3-8, while 51% were Glasgow Coma Scale 9-15. Moreover, at that time 26% of patients with a good outcome as defined by a CPC of 1 or 2 at 6 months were still in a coma.
“So a lot of patients were still affected by their disease or by sedation at that point. That’s an important finding,” Dr. Sunde said.
Some prognostic tests were highly unreliable
A standout in poor performance was the widely utilized standard of a time to return of spontaneous circulation greater than 25 minutes as a predictor of poor cerebral outcome. In fact, it had a 34% false-positive rate.
“I think it’s really useless to use that. I would rather have return to spontaneous circulation after 40 minutes of good-quality CPR than not have it with 25 minutes of lesser-quality CPR,” he commented.
Similarly, a Glasgow Coma Scale score of 9 or less or a Glasgow Coma Scale-Motor score of 1-3 upon assessment 3 days after sedation withdrawal had false-positive rates of 30% and 34%, respectively.
During hypothermia, EEG abnormalities had a high false-positive rate, and sensory-evoked potential findings were difficult to interpret.
Predictors showing utility
Several clinical factors predicted poor cerebral outcome with low false-positive rates: Unwitnessed cardiac arrest had a false-positive rate of only 4%; initial presentation in asystole or with pulseless electrical activity had a false-positive rate of 6%; and no bystander CPR had a false-positive rate of 13%.
Abnormal sensory-evoked potential or EEG findings 3 days after sedation withdrawal had low false-positive rates as prognosticators of poor cerebral outcome. An EEG showing burst suppression or epileptiform activity had a “pretty good” false-positive rate of only 7%, Dr. Sunde noted. Bilaterally absent N20 sensory-evoked potential findings, while uncommon, had a false-positive rate of zero. A serum neuron-specific enolase level greater than 80 mcg/mL had a 3% false-positive rate, in sharp contrast to the previously recommended cutoff of more than 33 mcg/mL, which had an unacceptable 38% false-positive rate.
“We should avoid using single predictors in decision making and be patient, especially if we have a witnessed ventricular fibrillation with bystander CPR, independent of time to return of spontaneous circulation,” he concluded.
Dr. Sunde and his coinvestigators plan to present numerous further follow-up studies from NORCAST, including the results of comprehensive cognitive function testing 6-9 months after cardiac arrest in all survivors, coupled with interviews with their close relatives, as well as cognitive function and quality-of-life measurements 3-6 years after cardiac arrest along with interviews with relatives.
Several audience members rose to declare that they’ve been waiting for data such as this for a long time. Session chair Karl B. Kern, MD, professor of medicine at the University of Arizona, Tucson, and codirector of the University of Arizona Sarver Heart Center, commented, “We’ve been talking about whether 3 days is too early for a number of years, and clearly from your data it is. It was twice as long before most of them woke up.”
Dr. Sunde reported having no financial conflicts of interest regarding the NORCAST study, which was sponsored by Oslo University Hospital.
ANAHEIM, CALIF. – Withdrawal of life-sustaining systemic therapies in comatose patients after out-of-hospital cardiac arrest as advised in current guidelines often occurs too early, resulting in the death of many patients who could potentially survive with good outcome, according to the results of NORCAST, the Norwegian Cardiorespiratory Arrest Study.
“The take-home message is to be patient and wait. Three days may be too early to make decisions on the patient,” Kjetil Sunde, MD, said in presenting the study findings at the Resuscitation Science Symposium held during the American Heart Association scientific sessions.
The European Resuscitation Council and European Society of Intensive Care Medicine have jointly recommended a prognostic algorithm in which a multimodal assessment is made on patients who are still comatose on day 3 after cardiac arrest. But this advice is based on expert opinion and has never been validated. This was the impetus for the prospective NORCAST study.
Current practice in the management of out-of-hospital cardiac arrest patients who are comatose upon hospital admission is to induce therapeutic hypothermia, with targeted temperature management to 33° C for 24 hours under deep sedation. The study hypothesis was that this strategy delays the time to awakening and that, as a consequence, the recommended prognostic tests that are usually done on day 3 after withdrawal of sedation are rendered insufficiently reliable. Thus, decisions to withdraw life-supporting therapies at that point will reduce the survival potential of this population, Dr. Sunde explained.
NORCAST was a prospective observational study that included 259 patients admitted to Oslo University Hospital in a comatose state after out-of-hospital cardiac arrest. In this unselected group, 81% had a cardiac cause for their arrest; the remainder had hypoxic arrest. All patients underwent therapeutic hypothermia, then a period of nonhypothermia followed by sedation withdrawal.
All of the widely used multimodal prognostic tests were ordered, including serial measurement of serum neuron-specific enolase; neurophysiologic testing using EEG and sensory-evoked potential readings obtained both during hypothermia and again at least 3 days after sedation withdrawal; a standardized clinical neurologic exam including assessment of brainstem reflexes and a Glasgow Coma Scale rating 3 days after sedation withdrawal; and a transcranial Doppler study and cerebral MRI on day 5-7. However, the treatment team was blinded to the results of these tests and was encouraged to delay withdrawal of life-supporting therapies as long as possible.
Key findings
Out of 259 patients who were comatose upon admission, 54% were alive at 6 months – and 91% of them had a CPC of 1 or 2.
The final tally at 6 months: 44% of patients were CPC 1, 5.5% were CPC 2, 4% were CPC 3, meaning severely disabled, and 46.5% were CPC 5, which is brain dead.
Withdrawal of life-supporting therapies occurred in 73 patients, or 28%, and 71% of those patients died, few of them in the early days.
Among patients with a CPC score of 1 or 2 at 6 months, only 20% were awake on day 1-3 following admission. Fifty-seven percent awoke on day 4-7, but importantly, 23% of patients with a good outcome at 6 months were not yet awake on day 8.
Three days after withdrawal of sedation, 49% of patients were rated as having a Glasgow Coma Scale score of 3-8, while 51% were Glasgow Coma Scale 9-15. Moreover, at that time 26% of patients with a good outcome as defined by a CPC of 1 or 2 at 6 months were still in a coma.
“So a lot of patients were still affected by their disease or by sedation at that point. That’s an important finding,” Dr. Sunde said.
Some prognostic tests were highly unreliable
A standout in poor performance was the widely utilized standard of a time to return of spontaneous circulation greater than 25 minutes as a predictor of poor cerebral outcome. In fact, it had a 34% false-positive rate.
“I think it’s really useless to use that. I would rather have return to spontaneous circulation after 40 minutes of good-quality CPR than not have it with 25 minutes of lesser-quality CPR,” he commented.
Similarly, a Glasgow Coma Scale score of 9 or less or a Glasgow Coma Scale-Motor score of 1-3 upon assessment 3 days after sedation withdrawal had false-positive rates of 30% and 34%, respectively.
During hypothermia, EEG abnormalities had a high false-positive rate, and sensory-evoked potential findings were difficult to interpret.
Predictors showing utility
Several clinical factors predicted poor cerebral outcome with low false-positive rates: Unwitnessed cardiac arrest had a false-positive rate of only 4%; initial presentation in asystole or with pulseless electrical activity had a false-positive rate of 6%; and no bystander CPR had a false-positive rate of 13%.
Abnormal sensory-evoked potential or EEG findings 3 days after sedation withdrawal had low false-positive rates as prognosticators of poor cerebral outcome. An EEG showing burst suppression or epileptiform activity had a “pretty good” false-positive rate of only 7%, Dr. Sunde noted. Bilaterally absent N20 sensory-evoked potential findings, while uncommon, had a false-positive rate of zero. A serum neuron-specific enolase level greater than 80 mcg/mL had a 3% false-positive rate, in sharp contrast to the previously recommended cutoff of more than 33 mcg/mL, which had an unacceptable 38% false-positive rate.
“We should avoid using single predictors in decision making and be patient, especially if we have a witnessed ventricular fibrillation with bystander CPR, independent of time to return of spontaneous circulation,” he concluded.
Dr. Sunde and his coinvestigators plan to present numerous further follow-up studies from NORCAST, including the results of comprehensive cognitive function testing 6-9 months after cardiac arrest in all survivors, coupled with interviews with their close relatives, as well as cognitive function and quality-of-life measurements 3-6 years after cardiac arrest along with interviews with relatives.
Several audience members rose to declare that they’ve been waiting for data such as this for a long time. Session chair Karl B. Kern, MD, professor of medicine at the University of Arizona, Tucson, and codirector of the University of Arizona Sarver Heart Center, commented, “We’ve been talking about whether 3 days is too early for a number of years, and clearly from your data it is. It was twice as long before most of them woke up.”
Dr. Sunde reported having no financial conflicts of interest regarding the NORCAST study, which was sponsored by Oslo University Hospital.
ANAHEIM, CALIF. – Withdrawal of life-sustaining systemic therapies in comatose patients after out-of-hospital cardiac arrest as advised in current guidelines often occurs too early, resulting in the death of many patients who could potentially survive with good outcome, according to the results of NORCAST, the Norwegian Cardiorespiratory Arrest Study.
“The take-home message is to be patient and wait. Three days may be too early to make decisions on the patient,” Kjetil Sunde, MD, said in presenting the study findings at the Resuscitation Science Symposium held during the American Heart Association scientific sessions.
The European Resuscitation Council and European Society of Intensive Care Medicine have jointly recommended a prognostic algorithm in which a multimodal assessment is made on patients who are still comatose on day 3 after cardiac arrest. But this advice is based on expert opinion and has never been validated. This was the impetus for the prospective NORCAST study.
Current practice in the management of out-of-hospital cardiac arrest patients who are comatose upon hospital admission is to induce therapeutic hypothermia, with targeted temperature management to 33° C for 24 hours under deep sedation. The study hypothesis was that this strategy delays the time to awakening and that, as a consequence, the recommended prognostic tests that are usually done on day 3 after withdrawal of sedation are rendered insufficiently reliable. Thus, decisions to withdraw life-supporting therapies at that point will reduce the survival potential of this population, Dr. Sunde explained.
NORCAST was a prospective observational study that included 259 patients admitted to Oslo University Hospital in a comatose state after out-of-hospital cardiac arrest. In this unselected group, 81% had a cardiac cause for their arrest; the remainder had hypoxic arrest. All patients underwent therapeutic hypothermia, then a period of nonhypothermia followed by sedation withdrawal.
All of the widely used multimodal prognostic tests were ordered, including serial measurement of serum neuron-specific enolase; neurophysiologic testing using EEG and sensory-evoked potential readings obtained both during hypothermia and again at least 3 days after sedation withdrawal; a standardized clinical neurologic exam including assessment of brainstem reflexes and a Glasgow Coma Scale rating 3 days after sedation withdrawal; and a transcranial Doppler study and cerebral MRI on day 5-7. However, the treatment team was blinded to the results of these tests and was encouraged to delay withdrawal of life-supporting therapies as long as possible.
Key findings
Out of 259 patients who were comatose upon admission, 54% were alive at 6 months – and 91% of them had a CPC of 1 or 2.
The final tally at 6 months: 44% of patients were CPC 1, 5.5% were CPC 2, 4% were CPC 3, meaning severely disabled, and 46.5% were CPC 5, which is brain dead.
Withdrawal of life-supporting therapies occurred in 73 patients, or 28%, and 71% of those patients died, few of them in the early days.
Among patients with a CPC score of 1 or 2 at 6 months, only 20% were awake on day 1-3 following admission. Fifty-seven percent awoke on day 4-7, but importantly, 23% of patients with a good outcome at 6 months were not yet awake on day 8.
Three days after withdrawal of sedation, 49% of patients were rated as having a Glasgow Coma Scale score of 3-8, while 51% were Glasgow Coma Scale 9-15. Moreover, at that time 26% of patients with a good outcome as defined by a CPC of 1 or 2 at 6 months were still in a coma.
“So a lot of patients were still affected by their disease or by sedation at that point. That’s an important finding,” Dr. Sunde said.
Some prognostic tests were highly unreliable
A standout in poor performance was the widely utilized standard of a time to return of spontaneous circulation greater than 25 minutes as a predictor of poor cerebral outcome. In fact, it had a 34% false-positive rate.
“I think it’s really useless to use that. I would rather have return to spontaneous circulation after 40 minutes of good-quality CPR than not have it with 25 minutes of lesser-quality CPR,” he commented.
Similarly, a Glasgow Coma Scale score of 9 or less or a Glasgow Coma Scale-Motor score of 1-3 upon assessment 3 days after sedation withdrawal had false-positive rates of 30% and 34%, respectively.
During hypothermia, EEG abnormalities had a high false-positive rate, and sensory-evoked potential findings were difficult to interpret.
Predictors showing utility
Several clinical factors predicted poor cerebral outcome with low false-positive rates: Unwitnessed cardiac arrest had a false-positive rate of only 4%; initial presentation in asystole or with pulseless electrical activity had a false-positive rate of 6%; and no bystander CPR had a false-positive rate of 13%.
Abnormal sensory-evoked potential or EEG findings 3 days after sedation withdrawal had low false-positive rates as prognosticators of poor cerebral outcome. An EEG showing burst suppression or epileptiform activity had a “pretty good” false-positive rate of only 7%, Dr. Sunde noted. Bilaterally absent N20 sensory-evoked potential findings, while uncommon, had a false-positive rate of zero. A serum neuron-specific enolase level greater than 80 mcg/mL had a 3% false-positive rate, in sharp contrast to the previously recommended cutoff of more than 33 mcg/mL, which had an unacceptable 38% false-positive rate.
“We should avoid using single predictors in decision making and be patient, especially if we have a witnessed ventricular fibrillation with bystander CPR, independent of time to return of spontaneous circulation,” he concluded.
Dr. Sunde and his coinvestigators plan to present numerous further follow-up studies from NORCAST, including the results of comprehensive cognitive function testing 6-9 months after cardiac arrest in all survivors, coupled with interviews with their close relatives, as well as cognitive function and quality-of-life measurements 3-6 years after cardiac arrest along with interviews with relatives.
Several audience members rose to declare that they’ve been waiting for data such as this for a long time. Session chair Karl B. Kern, MD, professor of medicine at the University of Arizona, Tucson, and codirector of the University of Arizona Sarver Heart Center, commented, “We’ve been talking about whether 3 days is too early for a number of years, and clearly from your data it is. It was twice as long before most of them woke up.”
Dr. Sunde reported having no financial conflicts of interest regarding the NORCAST study, which was sponsored by Oslo University Hospital.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point:
Major finding: Patients with good cerebral function 6 months after admission in a comatose state stemming from out-of-hospital cardiac arrest awoke a mean of 6.2 days post cardiac arrest, far later than most prognostic assessments take place.
Data source: NORCAST, a prospective observational study, included 259 patients who were comatose upon hospital admission after out-of-hospital cardiac arrest.
Disclosures: NORCAST was sponsored by Oslo University Hospital. The presenter reported having no financial conflicts.
Low-sodium DASH benefits increase with higher blood pressures
ANAHEIM, CALIF. – The low-sodium DASH diet lowered systolic BP a mean of 20.8 mm Hg among patients with a baseline systolic pressure of 150-159 mm Hg, and did so in just 4 weeks, according to a new analysis of the DASH-Sodium trial.
The original 2001 study found that combining low sodium and the DASH [Dietary Approaches to Stop Hypertension] diet lowered blood pressure more than either alone, but results were not broken out by hypertension severity (N Engl J Med. 2001 Jan 4;344[1]:3-10).
It found that there were “progressively greater reductions at higher levels of baseline systolic BP (SBP). Among participants with baseline SBP at or above 150 mm Hg, “mean SBP reduction was striking,” said Dr. Juraschek, of Harvard Medical School/Beth Israel Deaconess Medical Center, Boston.
The original trial randomized 208 subjects to the DASH diet and 204 to a control diet similar to what most Americans eat. While on their diets, the subjects cycled through three sodium levels for 4 weeks each: 1.5 g/d, 2.4 g/d, and 3.3 g/d. Although deemed high sodium in the study, 3.3 g/d is typical of the American diet.
The new study analyzed outcomes according to four baseline SBP categories: 120-129, 130-139, 140-149, and 150-159 mm Hg.
Among subjects on the control diet, reducing sodium from high to low intake reduced SBP 3.20, 8.56, 8.99, and 7.04 mm Hg across the four baseline SBP categories (P = .004). Among patients consuming high sodium, the DASH diet, compared with the control diet, reduced SBP 4.5, 4.3, 4.7, and 10.6 mm Hg, but the trend was not statistically significant.
The low-sodium DASH diet, versus the high-sodium control diet, reduced SBP 5.3, 7.5, 9.7, and 20.8 mm Hg in subjects with baseline SBP at or above 150 mmHg (P < .001).
“The DASH diet with low sodium, compared with the control diet with high sodium, lowered SBP by nearly 10 mm Hg among those with a baseline SBP of 140-149 mm Hg and [greater than] 20 mm Hg among those with a baseline systolic BP [at or above] 150 mm Hg. SBP levels between 140 and 159 mm Hg represent the majority of patients with hypertension. Thus, our findings suggest that most adults with uncontrolled BP can experience substantial reductions in SBP from dietary changes alone,” the investigator said.
“To place our results in context, compared to placebo, angiotensin-converting enzyme inhibitors reduce SBP by 12 mm Hg, beta-blockers reduce SBP by 13 mm Hg, and calcium-channel blockers reduce SBP by 16 mm Hg,” he said.
“For many patients, it’s hard to take that step to be on a chronic medication. A lot of them want to talk about diet, but” find it hard to believe that something as simple as changing what you eat could beat drugs. “It’s important for both patients and physicians to realize that if you take this seriously, you can have significant reductions in your blood pressure. We should take it seriously as the first step. That’s the key take away,” Dr. Juraschek said in an interview.
None of the participants were on blood pressure medications; 57% were women, and 57% were black. The mean age was 48 years, and mean baseline BP was 135/86 mm Hg. The DASH diet includes whole grains, poultry, fish, and nuts, with reductions in red meat, sweets, and sugary drinks.
The results were published, online simultaneously with Dr. Juraschek’s presentation (J Am Coll Cardiol. 2017 Nov 12;doi: 10.1016/j.jacc.2017.10.011).
The original study was funded by the National Institutes of Health. Dr. Juraschek had no relevant disclosures.
ANAHEIM, CALIF. – The low-sodium DASH diet lowered systolic BP a mean of 20.8 mm Hg among patients with a baseline systolic pressure of 150-159 mm Hg, and did so in just 4 weeks, according to a new analysis of the DASH-Sodium trial.
The original 2001 study found that combining low sodium and the DASH [Dietary Approaches to Stop Hypertension] diet lowered blood pressure more than either alone, but results were not broken out by hypertension severity (N Engl J Med. 2001 Jan 4;344[1]:3-10).
It found that there were “progressively greater reductions at higher levels of baseline systolic BP (SBP). Among participants with baseline SBP at or above 150 mm Hg, “mean SBP reduction was striking,” said Dr. Juraschek, of Harvard Medical School/Beth Israel Deaconess Medical Center, Boston.
The original trial randomized 208 subjects to the DASH diet and 204 to a control diet similar to what most Americans eat. While on their diets, the subjects cycled through three sodium levels for 4 weeks each: 1.5 g/d, 2.4 g/d, and 3.3 g/d. Although deemed high sodium in the study, 3.3 g/d is typical of the American diet.
The new study analyzed outcomes according to four baseline SBP categories: 120-129, 130-139, 140-149, and 150-159 mm Hg.
Among subjects on the control diet, reducing sodium from high to low intake reduced SBP 3.20, 8.56, 8.99, and 7.04 mm Hg across the four baseline SBP categories (P = .004). Among patients consuming high sodium, the DASH diet, compared with the control diet, reduced SBP 4.5, 4.3, 4.7, and 10.6 mm Hg, but the trend was not statistically significant.
The low-sodium DASH diet, versus the high-sodium control diet, reduced SBP 5.3, 7.5, 9.7, and 20.8 mm Hg in subjects with baseline SBP at or above 150 mmHg (P < .001).
“The DASH diet with low sodium, compared with the control diet with high sodium, lowered SBP by nearly 10 mm Hg among those with a baseline SBP of 140-149 mm Hg and [greater than] 20 mm Hg among those with a baseline systolic BP [at or above] 150 mm Hg. SBP levels between 140 and 159 mm Hg represent the majority of patients with hypertension. Thus, our findings suggest that most adults with uncontrolled BP can experience substantial reductions in SBP from dietary changes alone,” the investigator said.
“To place our results in context, compared to placebo, angiotensin-converting enzyme inhibitors reduce SBP by 12 mm Hg, beta-blockers reduce SBP by 13 mm Hg, and calcium-channel blockers reduce SBP by 16 mm Hg,” he said.
“For many patients, it’s hard to take that step to be on a chronic medication. A lot of them want to talk about diet, but” find it hard to believe that something as simple as changing what you eat could beat drugs. “It’s important for both patients and physicians to realize that if you take this seriously, you can have significant reductions in your blood pressure. We should take it seriously as the first step. That’s the key take away,” Dr. Juraschek said in an interview.
None of the participants were on blood pressure medications; 57% were women, and 57% were black. The mean age was 48 years, and mean baseline BP was 135/86 mm Hg. The DASH diet includes whole grains, poultry, fish, and nuts, with reductions in red meat, sweets, and sugary drinks.
The results were published, online simultaneously with Dr. Juraschek’s presentation (J Am Coll Cardiol. 2017 Nov 12;doi: 10.1016/j.jacc.2017.10.011).
The original study was funded by the National Institutes of Health. Dr. Juraschek had no relevant disclosures.
ANAHEIM, CALIF. – The low-sodium DASH diet lowered systolic BP a mean of 20.8 mm Hg among patients with a baseline systolic pressure of 150-159 mm Hg, and did so in just 4 weeks, according to a new analysis of the DASH-Sodium trial.
The original 2001 study found that combining low sodium and the DASH [Dietary Approaches to Stop Hypertension] diet lowered blood pressure more than either alone, but results were not broken out by hypertension severity (N Engl J Med. 2001 Jan 4;344[1]:3-10).
It found that there were “progressively greater reductions at higher levels of baseline systolic BP (SBP). Among participants with baseline SBP at or above 150 mm Hg, “mean SBP reduction was striking,” said Dr. Juraschek, of Harvard Medical School/Beth Israel Deaconess Medical Center, Boston.
The original trial randomized 208 subjects to the DASH diet and 204 to a control diet similar to what most Americans eat. While on their diets, the subjects cycled through three sodium levels for 4 weeks each: 1.5 g/d, 2.4 g/d, and 3.3 g/d. Although deemed high sodium in the study, 3.3 g/d is typical of the American diet.
The new study analyzed outcomes according to four baseline SBP categories: 120-129, 130-139, 140-149, and 150-159 mm Hg.
Among subjects on the control diet, reducing sodium from high to low intake reduced SBP 3.20, 8.56, 8.99, and 7.04 mm Hg across the four baseline SBP categories (P = .004). Among patients consuming high sodium, the DASH diet, compared with the control diet, reduced SBP 4.5, 4.3, 4.7, and 10.6 mm Hg, but the trend was not statistically significant.
The low-sodium DASH diet, versus the high-sodium control diet, reduced SBP 5.3, 7.5, 9.7, and 20.8 mm Hg in subjects with baseline SBP at or above 150 mmHg (P < .001).
“The DASH diet with low sodium, compared with the control diet with high sodium, lowered SBP by nearly 10 mm Hg among those with a baseline SBP of 140-149 mm Hg and [greater than] 20 mm Hg among those with a baseline systolic BP [at or above] 150 mm Hg. SBP levels between 140 and 159 mm Hg represent the majority of patients with hypertension. Thus, our findings suggest that most adults with uncontrolled BP can experience substantial reductions in SBP from dietary changes alone,” the investigator said.
“To place our results in context, compared to placebo, angiotensin-converting enzyme inhibitors reduce SBP by 12 mm Hg, beta-blockers reduce SBP by 13 mm Hg, and calcium-channel blockers reduce SBP by 16 mm Hg,” he said.
“For many patients, it’s hard to take that step to be on a chronic medication. A lot of them want to talk about diet, but” find it hard to believe that something as simple as changing what you eat could beat drugs. “It’s important for both patients and physicians to realize that if you take this seriously, you can have significant reductions in your blood pressure. We should take it seriously as the first step. That’s the key take away,” Dr. Juraschek said in an interview.
None of the participants were on blood pressure medications; 57% were women, and 57% were black. The mean age was 48 years, and mean baseline BP was 135/86 mm Hg. The DASH diet includes whole grains, poultry, fish, and nuts, with reductions in red meat, sweets, and sugary drinks.
The results were published, online simultaneously with Dr. Juraschek’s presentation (J Am Coll Cardiol. 2017 Nov 12;doi: 10.1016/j.jacc.2017.10.011).
The original study was funded by the National Institutes of Health. Dr. Juraschek had no relevant disclosures.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point:
Major finding: The low-sodium DASH diet lowered systolic BP a mean of 20.8 mm Hg among patients with a baseline systolic pressure of 150-159 mm Hg, and did so in just 4 weeks.
Data source: New analysis of the landmark DASH-Sodium trial.
Disclosures: The original study was funded by the National Institutes of Health. The lead investigator in the new analysis didn’t have any relevant disclosures.
PCI outcomes not better at top-ranked hospitals
Outcomes after percutaneous coronary intervention (PCI) are not superior when performed in U.S. hospitals ranked as “best” in a prominent national rating system as compared with nonranked hospitals, according to results of a recent retrospective analysis.
Rates of in-hospital mortality, acute kidney injury, and bleeding were similar for hospitals in the 2015 U.S. News & World Report’s “Best Hospitals” rankings and nonranked hospitals, Devraj Sukul, MD, reported at the American Heart Association Scientific Sessions.
“These findings should reassure patients that safe and appropriate PCI is being performed across the country,” said Dr. Sukul of the Division of Cardiovascular Medicine, University of Michigan, Ann Arbor.
The findings, published simultaneously (JACC Cardiovasc Interv. 2017 Nov 12. doi: 10.1016/j.jcin.2017.10.042) were based on a retrospective analysis of PCIs documented in the National Cardiovascular Data Registry CathPCI Registry.
Dr. Sukul and his colleagues limited their analysis to hospitals that both participated in that registry and performed at least 400 PCIs during July 2014–June 2015. That narrowed it down to 654 hospitals, including 44 out of the 50 hospitals ranked by U.S. News & World Report in 2015.
A total of 509,153 PCIs were performed over the 1-year study period, including 55,550 (10.9%) performed at the top-ranked hospitals.
After adjusting for patient risk, there was no difference in post-PCI in-hospital mortality between top-ranked and nonranked hospitals investigators reported (adjusted odds ratio, 0.96; P = .64).
There were also no differences in acute kidney injury (adjusted OR, 1.10; P = .1) or bleeding (adjusted OR, 1.15; P = .052) for top-ranked vs. nonranked hospitals, according to investigators.
In addition, top-ranked hospitals had a “slightly lower proportion” of appropriate PCI, Dr. Sukul reported.
Though rates of appropriate PCI were relatively high in both groups, odds of appropriate PCI were nevertheless significantly higher at nonranked hospitals (89.2% for ranked and 92.8% for nonranked hospitals; P less than .001).
Appropriate PCIs – those based on evidence-based indications – have been increasingly emphasized over the past decade.
Although some recent reports suggest hospital-level appropriateness may not necessarily correlate with clinical outcomes, Dr. Sukul remarked, “we believe that PCI appropriateness is an important indicator of quality, serving as a measure of physician decision-making when faced with treating the vast array of coronary artery disease presentations.”
Dr. Sukul is supported by a National Institutes of Health postdoctoral research training grant.
It should be welcome news to the public that outcomes of PCI conducted at top-ranked hospitals were not superior to those of procedures performed at nonranked hospitals.
This study addresses what is often the foremost question of a patient and their family in their hometown: Is my local hospital doing a good job? To the extent measured by the variables in this study, it is reassuring that the answer appears to be “Yes.”
It is hard to argue that health care should be immune from rankings in an era where consumers have access to ratings for just about every product and service available.
However, the public may be confused regarding the multiple national hospital ranking systems that are available today, particularly since these rating systems do not consistently identify hospitals as top performers.
Each rating system uses different data sources, has its own rating methodology, defines different measures of performance, and has a different focus. Many have argued that transparency will improve health care but, for the public, this is getting to the point of “too much information.”
Gregory J. Dehmer, MD, of the Department of Medicine (Cardiology Division) Texas A&M University, and Baylor Scott & White Health, Temple, made the comments above in an accompanying editorial (JACC Cardiovasc Interv. 2017 Nov 1. doi: 10.1016/j.jcin.2017.11.001). He reported no financial relationships relevant to the topic.
It should be welcome news to the public that outcomes of PCI conducted at top-ranked hospitals were not superior to those of procedures performed at nonranked hospitals.
This study addresses what is often the foremost question of a patient and their family in their hometown: Is my local hospital doing a good job? To the extent measured by the variables in this study, it is reassuring that the answer appears to be “Yes.”
It is hard to argue that health care should be immune from rankings in an era where consumers have access to ratings for just about every product and service available.
However, the public may be confused regarding the multiple national hospital ranking systems that are available today, particularly since these rating systems do not consistently identify hospitals as top performers.
Each rating system uses different data sources, has its own rating methodology, defines different measures of performance, and has a different focus. Many have argued that transparency will improve health care but, for the public, this is getting to the point of “too much information.”
Gregory J. Dehmer, MD, of the Department of Medicine (Cardiology Division) Texas A&M University, and Baylor Scott & White Health, Temple, made the comments above in an accompanying editorial (JACC Cardiovasc Interv. 2017 Nov 1. doi: 10.1016/j.jcin.2017.11.001). He reported no financial relationships relevant to the topic.
It should be welcome news to the public that outcomes of PCI conducted at top-ranked hospitals were not superior to those of procedures performed at nonranked hospitals.
This study addresses what is often the foremost question of a patient and their family in their hometown: Is my local hospital doing a good job? To the extent measured by the variables in this study, it is reassuring that the answer appears to be “Yes.”
It is hard to argue that health care should be immune from rankings in an era where consumers have access to ratings for just about every product and service available.
However, the public may be confused regarding the multiple national hospital ranking systems that are available today, particularly since these rating systems do not consistently identify hospitals as top performers.
Each rating system uses different data sources, has its own rating methodology, defines different measures of performance, and has a different focus. Many have argued that transparency will improve health care but, for the public, this is getting to the point of “too much information.”
Gregory J. Dehmer, MD, of the Department of Medicine (Cardiology Division) Texas A&M University, and Baylor Scott & White Health, Temple, made the comments above in an accompanying editorial (JACC Cardiovasc Interv. 2017 Nov 1. doi: 10.1016/j.jcin.2017.11.001). He reported no financial relationships relevant to the topic.
Outcomes after percutaneous coronary intervention (PCI) are not superior when performed in U.S. hospitals ranked as “best” in a prominent national rating system as compared with nonranked hospitals, according to results of a recent retrospective analysis.
Rates of in-hospital mortality, acute kidney injury, and bleeding were similar for hospitals in the 2015 U.S. News & World Report’s “Best Hospitals” rankings and nonranked hospitals, Devraj Sukul, MD, reported at the American Heart Association Scientific Sessions.
“These findings should reassure patients that safe and appropriate PCI is being performed across the country,” said Dr. Sukul of the Division of Cardiovascular Medicine, University of Michigan, Ann Arbor.
The findings, published simultaneously (JACC Cardiovasc Interv. 2017 Nov 12. doi: 10.1016/j.jcin.2017.10.042) were based on a retrospective analysis of PCIs documented in the National Cardiovascular Data Registry CathPCI Registry.
Dr. Sukul and his colleagues limited their analysis to hospitals that both participated in that registry and performed at least 400 PCIs during July 2014–June 2015. That narrowed it down to 654 hospitals, including 44 out of the 50 hospitals ranked by U.S. News & World Report in 2015.
A total of 509,153 PCIs were performed over the 1-year study period, including 55,550 (10.9%) performed at the top-ranked hospitals.
After adjusting for patient risk, there was no difference in post-PCI in-hospital mortality between top-ranked and nonranked hospitals investigators reported (adjusted odds ratio, 0.96; P = .64).
There were also no differences in acute kidney injury (adjusted OR, 1.10; P = .1) or bleeding (adjusted OR, 1.15; P = .052) for top-ranked vs. nonranked hospitals, according to investigators.
In addition, top-ranked hospitals had a “slightly lower proportion” of appropriate PCI, Dr. Sukul reported.
Though rates of appropriate PCI were relatively high in both groups, odds of appropriate PCI were nevertheless significantly higher at nonranked hospitals (89.2% for ranked and 92.8% for nonranked hospitals; P less than .001).
Appropriate PCIs – those based on evidence-based indications – have been increasingly emphasized over the past decade.
Although some recent reports suggest hospital-level appropriateness may not necessarily correlate with clinical outcomes, Dr. Sukul remarked, “we believe that PCI appropriateness is an important indicator of quality, serving as a measure of physician decision-making when faced with treating the vast array of coronary artery disease presentations.”
Dr. Sukul is supported by a National Institutes of Health postdoctoral research training grant.
Outcomes after percutaneous coronary intervention (PCI) are not superior when performed in U.S. hospitals ranked as “best” in a prominent national rating system as compared with nonranked hospitals, according to results of a recent retrospective analysis.
Rates of in-hospital mortality, acute kidney injury, and bleeding were similar for hospitals in the 2015 U.S. News & World Report’s “Best Hospitals” rankings and nonranked hospitals, Devraj Sukul, MD, reported at the American Heart Association Scientific Sessions.
“These findings should reassure patients that safe and appropriate PCI is being performed across the country,” said Dr. Sukul of the Division of Cardiovascular Medicine, University of Michigan, Ann Arbor.
The findings, published simultaneously (JACC Cardiovasc Interv. 2017 Nov 12. doi: 10.1016/j.jcin.2017.10.042) were based on a retrospective analysis of PCIs documented in the National Cardiovascular Data Registry CathPCI Registry.
Dr. Sukul and his colleagues limited their analysis to hospitals that both participated in that registry and performed at least 400 PCIs during July 2014–June 2015. That narrowed it down to 654 hospitals, including 44 out of the 50 hospitals ranked by U.S. News & World Report in 2015.
A total of 509,153 PCIs were performed over the 1-year study period, including 55,550 (10.9%) performed at the top-ranked hospitals.
After adjusting for patient risk, there was no difference in post-PCI in-hospital mortality between top-ranked and nonranked hospitals investigators reported (adjusted odds ratio, 0.96; P = .64).
There were also no differences in acute kidney injury (adjusted OR, 1.10; P = .1) or bleeding (adjusted OR, 1.15; P = .052) for top-ranked vs. nonranked hospitals, according to investigators.
In addition, top-ranked hospitals had a “slightly lower proportion” of appropriate PCI, Dr. Sukul reported.
Though rates of appropriate PCI were relatively high in both groups, odds of appropriate PCI were nevertheless significantly higher at nonranked hospitals (89.2% for ranked and 92.8% for nonranked hospitals; P less than .001).
Appropriate PCIs – those based on evidence-based indications – have been increasingly emphasized over the past decade.
Although some recent reports suggest hospital-level appropriateness may not necessarily correlate with clinical outcomes, Dr. Sukul remarked, “we believe that PCI appropriateness is an important indicator of quality, serving as a measure of physician decision-making when faced with treating the vast array of coronary artery disease presentations.”
Dr. Sukul is supported by a National Institutes of Health postdoctoral research training grant.
FROM THE AHA SCIENTIFIC SESSIONS
Key clinical point: Percutaneous coronary intervention (PCI) performed at the 50 “Best Hospitals” in U.S. News & World Report rankings was not associated with better outcomes, compared with PCI at other hospitals.
Major finding: There was no significant difference between ranked and nonranked hospitals for PCI-associated in-hospital mortality (adjusted OR, 0.96; 95% CI, 0.83-1.12; P = 0.64), acute kidney injury, or bleeding.
Data source: A retrospective analysis of 509,153 PCIs included in the National Cardiovascular Data Registry CathPCI Registry.
Disclosures: First author Dr. Devraj Sukul is supported by a National Institutes of Health postdoctoral research training grant. Coauthors reported disclosures including AstraZeneca, Regado Biosciences, and Pfizer, among others.
ORBITA: PCI no better than meds for stable angina
DENVER – The first-ever blinded, sham-controlled randomized trial of percutaneous coronary intervention for stable angina failed to show a significant improvement in exercise time for PCI, compared with placebo PCI, Rasha Al-Lamee, MD, reported at the Transcatheter Cardiovascular Therapeutics annual educational meeting.
The blockbuster results of the ORBITA trial, published online in the Lancet simultaneously with Dr. Al-Lamee’s presentation in Denver, quickly went viral, with a story splashed across the front page of the New York Times under the headline “‘Unbelievable’: Heart Stents Fail to Ease Chest Pain.” Interventional cardiology thought leaders at TCT said the newspaper piece, and a Lancet editorial commentary entitled “Last nail in the coffin for PCI in stable angina?” that accompanied publication of ORBITA, failed to convey the study’s major limitations, drawbacks that Dr. Al-Lamee readily acknowledged.
What ORBITA did
ORBITA (Objective Randomized Blinded Investigation with Optimal Medical Therapy of Angioplasty in Stable Angina) included 200 patients referred to five U.K. cardiac catheterization labs for diagnostic angiography. Participants had to have stable angina, single-vessel disease, and at least one 70% or greater stenosis; in fact, their stenotic severity averaged 84.4% by quantitative coronary angiography.
The patients received 6 weeks of intensive medical therapy during which they were uptitrated to an average of three antianginal medications. They then underwent either real or sham PCI followed by 6 weeks of recovery, during which both the patients and care team remained blinded. Then the same assessments done before randomization were repeated, including exercise treadmill testing, the Seattle Angina Questionnaire, and dobutamine stress echocardiography, explained Dr. Al-Lamee of Imperial College London.
The primary outcome was achievement of at least a 30-second greater improvement in total exercise time following PCI, compared with sham PCI, an effect size chosen based on placebo-controlled studies of antianginal drugs. The PCI group improved by a mean of 28.4 seconds, the controls by 11.8 seconds, and the resultant 16.6-second difference made for a negative result (Lancet. 2017 Nov 2;doi: 10.1016/S0140-6736[17]32714-9).
PCI did, however, result in significant improvement in the secondary endpoint of ischemia reduction as assessed by blinded evaluation of dobutamine stress echocardiography results. The PCI group’s mean peak stress wall motion index score improved from 1.11 prerandomization to 1.03 – that is, normal – at follow-up 6 weeks post procedure while remaining unchanged in the sham PCI group, Dr. Al-Lamee noted at the meeting, sponsored by the Cardiovascular Research Foundation.
What the results mean
Dr. Al-Lamee said the ORBITA results should enable cardiologists to sit with patients similar to those in the trial and have a more informed, patient-centered discussion in which intensive medical management can be offered as an initial first-line option with an understanding that it will likely improve their symptoms to the same degree as angioplasty.
“There will be those patients who would rather avoid having to take high doses of antianginal medications with the side effects they involve, who may well prefer to have an upfront procedure with a small risk in order to reduce their pill count, and who also would rather have improved blood flow to the heart, which may have prognostic implications,” Dr. Al-Lamee said.
Carl L. Tommaso, MD, part of the panel of discussants at the late-breaking clinical trials session in which Dr. Al-Lamee presented the ORBITA findings, applauded the investigators for their ingenious study design, which included elaborate blinding techniques involving music played through headphones throughout the procedure, heavy sedation, separate angioplasty and clinical care teams, the same postprocedural instructions and discharge letter, and dual-antiplatelet therapy in both study arms.
“This is a great study. I don’t think any of us could get this study past an institutional review board in the United States,” commented Dr. Tommaso, director of the cardiac catheterization laboratory at Skokie (Ill.) Hospital.
He added, however, that he wouldn’t have performed PCI on the basis of angiographic findings alone in stable angina patients with a 9-minute treadmill exercise time.
Where OPTIMA fell short
Angiography vs. functional testing
“Twenty-nine percent of patients, we’d all agree, should not have had angioplasty because they had no ischemia,” said Dr. Stone, professor of medicine at Columbia University, New York, and director of the TCT conference.
All subjects in ORBITA did indeed undergo measurement of both FFR and instant Wave-Free Ratio (iFR) while on the table immediately before and after their real or sham PCI. The mean stenosis severity was 0.69 by FFR and 0.76 by iFR, readings indicative of significantly impaired flow. However, the operators were blinded as to those results. The rationale for withholding that information was that, even though it has been shown to be clinically useful, studies show that 80% of angioplasties are done based upon angiography alone, and the ORBITA investigators wanted the study to reflect routine clinical practice, Dr. Al-Lamee explained.
“I think one of the many lessons coming out of this trial is to see the discrepancy between the angiogram and functional testing. We cannot guide our therapy solely by the angiogram. We have to get physiologic data and consider that together with symptoms in the patient’s clinical context,” said panelist Allen Jeremias, MD, director of interventional cardiology research at St. Francis Hospital in Rosyln, N.Y.
Commentary goes too far
The “last-nail-in-the-coffin” Lancet commentary (2017 Nov 2. doi: 10.1016/S0140-6736[17]32757-5) penned by David L. Brown, MD, of Washington University in St. Louis and Rita F. Redberg, MD, of the University of California, San Francisco, emphatically declared that the ORBITA results mean all cardiology guidelines should be revised to downgrade the recommendation for PCI in patients with angina despite medical therapy. Dr. Al-Lamee was one of many at TCT 2017 who took strong exception to that.
“This is the first trial of its kind. I think it would be very easy to take the results of this trial and overextrapolate. To downgrade the guideline recommendations based on this study would be an incredibly large overreach,” she said.
Ajay J. Kirtane, MD, who chaired a press conference in which Dr. Al-Lamee presented the ORBITA results, had a further criticism of the editorial.
“Some of the risks of PCI as described in the editorial are just factually inaccurate. An MI rate of 15%, an acute kidney injury rate of 13% – those are simply factually incorrect,” said Dr. Kirtane, director of the cardiac catheterization laboratories at New York-Presbyterian/Columbia University Medical Center.
The ORBITA trial was sponsored by Imperial College London and funded by grants from the National Institute of Health Research Imperial Biomedical Research Center and charity organizations. Dr. Al-Lamee reported serving as a paid consultant to Philips Volcano, which supplied the coronary pressure wires for physiologic testing.
DENVER – The first-ever blinded, sham-controlled randomized trial of percutaneous coronary intervention for stable angina failed to show a significant improvement in exercise time for PCI, compared with placebo PCI, Rasha Al-Lamee, MD, reported at the Transcatheter Cardiovascular Therapeutics annual educational meeting.
The blockbuster results of the ORBITA trial, published online in the Lancet simultaneously with Dr. Al-Lamee’s presentation in Denver, quickly went viral, with a story splashed across the front page of the New York Times under the headline “‘Unbelievable’: Heart Stents Fail to Ease Chest Pain.” Interventional cardiology thought leaders at TCT said the newspaper piece, and a Lancet editorial commentary entitled “Last nail in the coffin for PCI in stable angina?” that accompanied publication of ORBITA, failed to convey the study’s major limitations, drawbacks that Dr. Al-Lamee readily acknowledged.
What ORBITA did
ORBITA (Objective Randomized Blinded Investigation with Optimal Medical Therapy of Angioplasty in Stable Angina) included 200 patients referred to five U.K. cardiac catheterization labs for diagnostic angiography. Participants had to have stable angina, single-vessel disease, and at least one 70% or greater stenosis; in fact, their stenotic severity averaged 84.4% by quantitative coronary angiography.
The patients received 6 weeks of intensive medical therapy during which they were uptitrated to an average of three antianginal medications. They then underwent either real or sham PCI followed by 6 weeks of recovery, during which both the patients and care team remained blinded. Then the same assessments done before randomization were repeated, including exercise treadmill testing, the Seattle Angina Questionnaire, and dobutamine stress echocardiography, explained Dr. Al-Lamee of Imperial College London.
The primary outcome was achievement of at least a 30-second greater improvement in total exercise time following PCI, compared with sham PCI, an effect size chosen based on placebo-controlled studies of antianginal drugs. The PCI group improved by a mean of 28.4 seconds, the controls by 11.8 seconds, and the resultant 16.6-second difference made for a negative result (Lancet. 2017 Nov 2;doi: 10.1016/S0140-6736[17]32714-9).
PCI did, however, result in significant improvement in the secondary endpoint of ischemia reduction as assessed by blinded evaluation of dobutamine stress echocardiography results. The PCI group’s mean peak stress wall motion index score improved from 1.11 prerandomization to 1.03 – that is, normal – at follow-up 6 weeks post procedure while remaining unchanged in the sham PCI group, Dr. Al-Lamee noted at the meeting, sponsored by the Cardiovascular Research Foundation.
What the results mean
Dr. Al-Lamee said the ORBITA results should enable cardiologists to sit with patients similar to those in the trial and have a more informed, patient-centered discussion in which intensive medical management can be offered as an initial first-line option with an understanding that it will likely improve their symptoms to the same degree as angioplasty.
“There will be those patients who would rather avoid having to take high doses of antianginal medications with the side effects they involve, who may well prefer to have an upfront procedure with a small risk in order to reduce their pill count, and who also would rather have improved blood flow to the heart, which may have prognostic implications,” Dr. Al-Lamee said.
Carl L. Tommaso, MD, part of the panel of discussants at the late-breaking clinical trials session in which Dr. Al-Lamee presented the ORBITA findings, applauded the investigators for their ingenious study design, which included elaborate blinding techniques involving music played through headphones throughout the procedure, heavy sedation, separate angioplasty and clinical care teams, the same postprocedural instructions and discharge letter, and dual-antiplatelet therapy in both study arms.
“This is a great study. I don’t think any of us could get this study past an institutional review board in the United States,” commented Dr. Tommaso, director of the cardiac catheterization laboratory at Skokie (Ill.) Hospital.
He added, however, that he wouldn’t have performed PCI on the basis of angiographic findings alone in stable angina patients with a 9-minute treadmill exercise time.
Where OPTIMA fell short
Angiography vs. functional testing
“Twenty-nine percent of patients, we’d all agree, should not have had angioplasty because they had no ischemia,” said Dr. Stone, professor of medicine at Columbia University, New York, and director of the TCT conference.
All subjects in ORBITA did indeed undergo measurement of both FFR and instant Wave-Free Ratio (iFR) while on the table immediately before and after their real or sham PCI. The mean stenosis severity was 0.69 by FFR and 0.76 by iFR, readings indicative of significantly impaired flow. However, the operators were blinded as to those results. The rationale for withholding that information was that, even though it has been shown to be clinically useful, studies show that 80% of angioplasties are done based upon angiography alone, and the ORBITA investigators wanted the study to reflect routine clinical practice, Dr. Al-Lamee explained.
“I think one of the many lessons coming out of this trial is to see the discrepancy between the angiogram and functional testing. We cannot guide our therapy solely by the angiogram. We have to get physiologic data and consider that together with symptoms in the patient’s clinical context,” said panelist Allen Jeremias, MD, director of interventional cardiology research at St. Francis Hospital in Rosyln, N.Y.
Commentary goes too far
The “last-nail-in-the-coffin” Lancet commentary (2017 Nov 2. doi: 10.1016/S0140-6736[17]32757-5) penned by David L. Brown, MD, of Washington University in St. Louis and Rita F. Redberg, MD, of the University of California, San Francisco, emphatically declared that the ORBITA results mean all cardiology guidelines should be revised to downgrade the recommendation for PCI in patients with angina despite medical therapy. Dr. Al-Lamee was one of many at TCT 2017 who took strong exception to that.
“This is the first trial of its kind. I think it would be very easy to take the results of this trial and overextrapolate. To downgrade the guideline recommendations based on this study would be an incredibly large overreach,” she said.
Ajay J. Kirtane, MD, who chaired a press conference in which Dr. Al-Lamee presented the ORBITA results, had a further criticism of the editorial.
“Some of the risks of PCI as described in the editorial are just factually inaccurate. An MI rate of 15%, an acute kidney injury rate of 13% – those are simply factually incorrect,” said Dr. Kirtane, director of the cardiac catheterization laboratories at New York-Presbyterian/Columbia University Medical Center.
The ORBITA trial was sponsored by Imperial College London and funded by grants from the National Institute of Health Research Imperial Biomedical Research Center and charity organizations. Dr. Al-Lamee reported serving as a paid consultant to Philips Volcano, which supplied the coronary pressure wires for physiologic testing.
DENVER – The first-ever blinded, sham-controlled randomized trial of percutaneous coronary intervention for stable angina failed to show a significant improvement in exercise time for PCI, compared with placebo PCI, Rasha Al-Lamee, MD, reported at the Transcatheter Cardiovascular Therapeutics annual educational meeting.
The blockbuster results of the ORBITA trial, published online in the Lancet simultaneously with Dr. Al-Lamee’s presentation in Denver, quickly went viral, with a story splashed across the front page of the New York Times under the headline “‘Unbelievable’: Heart Stents Fail to Ease Chest Pain.” Interventional cardiology thought leaders at TCT said the newspaper piece, and a Lancet editorial commentary entitled “Last nail in the coffin for PCI in stable angina?” that accompanied publication of ORBITA, failed to convey the study’s major limitations, drawbacks that Dr. Al-Lamee readily acknowledged.
What ORBITA did
ORBITA (Objective Randomized Blinded Investigation with Optimal Medical Therapy of Angioplasty in Stable Angina) included 200 patients referred to five U.K. cardiac catheterization labs for diagnostic angiography. Participants had to have stable angina, single-vessel disease, and at least one 70% or greater stenosis; in fact, their stenotic severity averaged 84.4% by quantitative coronary angiography.
The patients received 6 weeks of intensive medical therapy during which they were uptitrated to an average of three antianginal medications. They then underwent either real or sham PCI followed by 6 weeks of recovery, during which both the patients and care team remained blinded. Then the same assessments done before randomization were repeated, including exercise treadmill testing, the Seattle Angina Questionnaire, and dobutamine stress echocardiography, explained Dr. Al-Lamee of Imperial College London.
The primary outcome was achievement of at least a 30-second greater improvement in total exercise time following PCI, compared with sham PCI, an effect size chosen based on placebo-controlled studies of antianginal drugs. The PCI group improved by a mean of 28.4 seconds, the controls by 11.8 seconds, and the resultant 16.6-second difference made for a negative result (Lancet. 2017 Nov 2;doi: 10.1016/S0140-6736[17]32714-9).
PCI did, however, result in significant improvement in the secondary endpoint of ischemia reduction as assessed by blinded evaluation of dobutamine stress echocardiography results. The PCI group’s mean peak stress wall motion index score improved from 1.11 prerandomization to 1.03 – that is, normal – at follow-up 6 weeks post procedure while remaining unchanged in the sham PCI group, Dr. Al-Lamee noted at the meeting, sponsored by the Cardiovascular Research Foundation.
What the results mean
Dr. Al-Lamee said the ORBITA results should enable cardiologists to sit with patients similar to those in the trial and have a more informed, patient-centered discussion in which intensive medical management can be offered as an initial first-line option with an understanding that it will likely improve their symptoms to the same degree as angioplasty.
“There will be those patients who would rather avoid having to take high doses of antianginal medications with the side effects they involve, who may well prefer to have an upfront procedure with a small risk in order to reduce their pill count, and who also would rather have improved blood flow to the heart, which may have prognostic implications,” Dr. Al-Lamee said.
Carl L. Tommaso, MD, part of the panel of discussants at the late-breaking clinical trials session in which Dr. Al-Lamee presented the ORBITA findings, applauded the investigators for their ingenious study design, which included elaborate blinding techniques involving music played through headphones throughout the procedure, heavy sedation, separate angioplasty and clinical care teams, the same postprocedural instructions and discharge letter, and dual-antiplatelet therapy in both study arms.
“This is a great study. I don’t think any of us could get this study past an institutional review board in the United States,” commented Dr. Tommaso, director of the cardiac catheterization laboratory at Skokie (Ill.) Hospital.
He added, however, that he wouldn’t have performed PCI on the basis of angiographic findings alone in stable angina patients with a 9-minute treadmill exercise time.
Where OPTIMA fell short
Angiography vs. functional testing
“Twenty-nine percent of patients, we’d all agree, should not have had angioplasty because they had no ischemia,” said Dr. Stone, professor of medicine at Columbia University, New York, and director of the TCT conference.
All subjects in ORBITA did indeed undergo measurement of both FFR and instant Wave-Free Ratio (iFR) while on the table immediately before and after their real or sham PCI. The mean stenosis severity was 0.69 by FFR and 0.76 by iFR, readings indicative of significantly impaired flow. However, the operators were blinded as to those results. The rationale for withholding that information was that, even though it has been shown to be clinically useful, studies show that 80% of angioplasties are done based upon angiography alone, and the ORBITA investigators wanted the study to reflect routine clinical practice, Dr. Al-Lamee explained.
“I think one of the many lessons coming out of this trial is to see the discrepancy between the angiogram and functional testing. We cannot guide our therapy solely by the angiogram. We have to get physiologic data and consider that together with symptoms in the patient’s clinical context,” said panelist Allen Jeremias, MD, director of interventional cardiology research at St. Francis Hospital in Rosyln, N.Y.
Commentary goes too far
The “last-nail-in-the-coffin” Lancet commentary (2017 Nov 2. doi: 10.1016/S0140-6736[17]32757-5) penned by David L. Brown, MD, of Washington University in St. Louis and Rita F. Redberg, MD, of the University of California, San Francisco, emphatically declared that the ORBITA results mean all cardiology guidelines should be revised to downgrade the recommendation for PCI in patients with angina despite medical therapy. Dr. Al-Lamee was one of many at TCT 2017 who took strong exception to that.
“This is the first trial of its kind. I think it would be very easy to take the results of this trial and overextrapolate. To downgrade the guideline recommendations based on this study would be an incredibly large overreach,” she said.
Ajay J. Kirtane, MD, who chaired a press conference in which Dr. Al-Lamee presented the ORBITA results, had a further criticism of the editorial.
“Some of the risks of PCI as described in the editorial are just factually inaccurate. An MI rate of 15%, an acute kidney injury rate of 13% – those are simply factually incorrect,” said Dr. Kirtane, director of the cardiac catheterization laboratories at New York-Presbyterian/Columbia University Medical Center.
The ORBITA trial was sponsored by Imperial College London and funded by grants from the National Institute of Health Research Imperial Biomedical Research Center and charity organizations. Dr. Al-Lamee reported serving as a paid consultant to Philips Volcano, which supplied the coronary pressure wires for physiologic testing.
AT TCT 2017
Key clinical point:
Major finding: PCI on top of intensive antianginal medications was not significantly more effective at improving exercise tolerance than sham PCI.
Data source: ORBITA, a randomized, multicenter, blinded, sham-controlled study of 200 patients with mild angina and single-vessel CAD.
Disclosures: ORBITA was sponsored by Imperial College London and funded by grants from the National Institute of Health Research Imperial Biomedical Research Center and charity organizations. The presenter reported serving as a paid consultant to Philips Volcano, which supplied the coronary pressure wires for physiologic testing.




















