User login
ChatGPT Enhances Readability of Cancer Information for Patients
TOPLINE:
The artificial intelligence (AI) chatbot ChatGPT can significantly improve the readability of online cancer-related patient information while maintaining the content’s quality, a recent study found.
METHODOLOGY:
- Patients with cancer often search for cancer information online after their diagnosis, with most seeking information from their oncologists’ websites. However, the online materials often exceed the average reading level of the US population, limiting accessibility and comprehension.
- Researchers asked ChatGPT 4.0 to rewrite content about breast, colon, lung, prostate, and pancreas cancer, aiming for a sixth-grade readability level. The content came from a random sample of documents from 34 patient-facing websites associated with National Comprehensive Cancer Network (NCCN) member institutions.
- Readability, accuracy, similarity, and quality of the rewritten content were assessed using several established metrics and tools, including an F1 score, which assesses the precision and recall of a machine-learning model; a cosine similarity score, which measures similarities and is often used to detect plagiarism; and the DISCERN instrument, which helps assess the quality of the AI-rewritten information.
- The primary outcome was the mean readability score for the original and AI-generated content.
TAKEAWAY:
- The original content had an average readability level equivalent to a university freshman (grade 13). Following the AI revision, the readability level improved to a high school freshman level (grade 9).
- The rewritten content had high accuracy, with an overall F1 score of 0.87 (a good score is 0.8-0.9).
- The rewritten content had a high cosine similarity score of 0.915 (scores range from 0 to 1, with 0 indicating no similarity and 1 indicating complete similarity). Researchers attributed the improved readability to the use of simpler words and shorter sentences.
- Quality assessment using the DISCERN instrument showed that the AI-rewritten content maintained a “good” quality rating, similar to that of the original content.
IN PRACTICE:
Society has become increasingly dependent on online educational materials, and considering that more than half of Americans may not be literate beyond an eighth-grade level, our AI intervention offers a potential low-cost solution to narrow the gap between patient health literacy and content received from the nation’s leading cancer centers, the authors wrote.
SOURCE:
The study, with first author Andres A. Abreu, MD, with UT Southwestern Medical Center, Dallas, Texas, was published online in the Journal of the National Comprehensive Cancer Network.
LIMITATIONS:
The study was limited to English-language content from NCCN member websites, so the findings may not be generalizable to other sources or languages. Readability alone cannot guarantee comprehension. Factors such as material design and audiovisual aids were not evaluated.
DISCLOSURES:
The study did not report a funding source. The authors reported several disclosures but none related to the study. Herbert J. Zeh disclosed serving as a scientific advisor for Surgical Safety Technologies; Dr. Polanco disclosed serving as a consultant for Iota Biosciences and Palisade Bio and as a proctor for Intuitive Surgical.
A version of this article first appeared on Medscape.com.
TOPLINE:
The artificial intelligence (AI) chatbot ChatGPT can significantly improve the readability of online cancer-related patient information while maintaining the content’s quality, a recent study found.
METHODOLOGY:
- Patients with cancer often search for cancer information online after their diagnosis, with most seeking information from their oncologists’ websites. However, the online materials often exceed the average reading level of the US population, limiting accessibility and comprehension.
- Researchers asked ChatGPT 4.0 to rewrite content about breast, colon, lung, prostate, and pancreas cancer, aiming for a sixth-grade readability level. The content came from a random sample of documents from 34 patient-facing websites associated with National Comprehensive Cancer Network (NCCN) member institutions.
- Readability, accuracy, similarity, and quality of the rewritten content were assessed using several established metrics and tools, including an F1 score, which assesses the precision and recall of a machine-learning model; a cosine similarity score, which measures similarities and is often used to detect plagiarism; and the DISCERN instrument, which helps assess the quality of the AI-rewritten information.
- The primary outcome was the mean readability score for the original and AI-generated content.
TAKEAWAY:
- The original content had an average readability level equivalent to a university freshman (grade 13). Following the AI revision, the readability level improved to a high school freshman level (grade 9).
- The rewritten content had high accuracy, with an overall F1 score of 0.87 (a good score is 0.8-0.9).
- The rewritten content had a high cosine similarity score of 0.915 (scores range from 0 to 1, with 0 indicating no similarity and 1 indicating complete similarity). Researchers attributed the improved readability to the use of simpler words and shorter sentences.
- Quality assessment using the DISCERN instrument showed that the AI-rewritten content maintained a “good” quality rating, similar to that of the original content.
IN PRACTICE:
Society has become increasingly dependent on online educational materials, and considering that more than half of Americans may not be literate beyond an eighth-grade level, our AI intervention offers a potential low-cost solution to narrow the gap between patient health literacy and content received from the nation’s leading cancer centers, the authors wrote.
SOURCE:
The study, with first author Andres A. Abreu, MD, with UT Southwestern Medical Center, Dallas, Texas, was published online in the Journal of the National Comprehensive Cancer Network.
LIMITATIONS:
The study was limited to English-language content from NCCN member websites, so the findings may not be generalizable to other sources or languages. Readability alone cannot guarantee comprehension. Factors such as material design and audiovisual aids were not evaluated.
DISCLOSURES:
The study did not report a funding source. The authors reported several disclosures but none related to the study. Herbert J. Zeh disclosed serving as a scientific advisor for Surgical Safety Technologies; Dr. Polanco disclosed serving as a consultant for Iota Biosciences and Palisade Bio and as a proctor for Intuitive Surgical.
A version of this article first appeared on Medscape.com.
TOPLINE:
The artificial intelligence (AI) chatbot ChatGPT can significantly improve the readability of online cancer-related patient information while maintaining the content’s quality, a recent study found.
METHODOLOGY:
- Patients with cancer often search for cancer information online after their diagnosis, with most seeking information from their oncologists’ websites. However, the online materials often exceed the average reading level of the US population, limiting accessibility and comprehension.
- Researchers asked ChatGPT 4.0 to rewrite content about breast, colon, lung, prostate, and pancreas cancer, aiming for a sixth-grade readability level. The content came from a random sample of documents from 34 patient-facing websites associated with National Comprehensive Cancer Network (NCCN) member institutions.
- Readability, accuracy, similarity, and quality of the rewritten content were assessed using several established metrics and tools, including an F1 score, which assesses the precision and recall of a machine-learning model; a cosine similarity score, which measures similarities and is often used to detect plagiarism; and the DISCERN instrument, which helps assess the quality of the AI-rewritten information.
- The primary outcome was the mean readability score for the original and AI-generated content.
TAKEAWAY:
- The original content had an average readability level equivalent to a university freshman (grade 13). Following the AI revision, the readability level improved to a high school freshman level (grade 9).
- The rewritten content had high accuracy, with an overall F1 score of 0.87 (a good score is 0.8-0.9).
- The rewritten content had a high cosine similarity score of 0.915 (scores range from 0 to 1, with 0 indicating no similarity and 1 indicating complete similarity). Researchers attributed the improved readability to the use of simpler words and shorter sentences.
- Quality assessment using the DISCERN instrument showed that the AI-rewritten content maintained a “good” quality rating, similar to that of the original content.
IN PRACTICE:
Society has become increasingly dependent on online educational materials, and considering that more than half of Americans may not be literate beyond an eighth-grade level, our AI intervention offers a potential low-cost solution to narrow the gap between patient health literacy and content received from the nation’s leading cancer centers, the authors wrote.
SOURCE:
The study, with first author Andres A. Abreu, MD, with UT Southwestern Medical Center, Dallas, Texas, was published online in the Journal of the National Comprehensive Cancer Network.
LIMITATIONS:
The study was limited to English-language content from NCCN member websites, so the findings may not be generalizable to other sources or languages. Readability alone cannot guarantee comprehension. Factors such as material design and audiovisual aids were not evaluated.
DISCLOSURES:
The study did not report a funding source. The authors reported several disclosures but none related to the study. Herbert J. Zeh disclosed serving as a scientific advisor for Surgical Safety Technologies; Dr. Polanco disclosed serving as a consultant for Iota Biosciences and Palisade Bio and as a proctor for Intuitive Surgical.
A version of this article first appeared on Medscape.com.
FDA Expands Durvalumab Label to Endometrial Cancer
Originally approved in 2017, the programmed death ligand 1 inhibitor caries previously approved indications for non–small cell lung cancer, biliary tract cancer, and hepatocellular carcinoma.
Approval of the new indication was based on the phase 3 DUO-E trial, which included 95 women with newly diagnosed advanced or recurrent dMMR endometrial cancer. Patients were randomized to durvalumab 1120 mg or placebo with carboplatin plus paclitaxel every 3 weeks for a maximum of six cycles followed by durvalumab 1500 mg every 4 weeks until disease progression.
Median progression-free survival (PFS) was 7 months in the placebo arm but not reached in the durvalumab group. Overall survival outcomes were immature at the PFS analysis.
A quarter or more of durvalumab patients experienced peripheral neuropathy, musculoskeletal pain, nausea, alopecia, fatigue, abdominal pain, constipation, rash, diarrhea, vomiting, and cough.
The recommended treatment regimen for dMMR endometrial cancer in women who weigh ≥ 30 kg is 1120 mg with carboplatin plus paclitaxel every 3 weeks for six cycles, followed by single-agent durvalumab 1500 mg every 4 weeks.
The price of 2.4 mL of durvalumab at a concentration of 50 mg/mL is $1027, according to drugs.com.
A version of this article appeared on Medscape.com.
Originally approved in 2017, the programmed death ligand 1 inhibitor caries previously approved indications for non–small cell lung cancer, biliary tract cancer, and hepatocellular carcinoma.
Approval of the new indication was based on the phase 3 DUO-E trial, which included 95 women with newly diagnosed advanced or recurrent dMMR endometrial cancer. Patients were randomized to durvalumab 1120 mg or placebo with carboplatin plus paclitaxel every 3 weeks for a maximum of six cycles followed by durvalumab 1500 mg every 4 weeks until disease progression.
Median progression-free survival (PFS) was 7 months in the placebo arm but not reached in the durvalumab group. Overall survival outcomes were immature at the PFS analysis.
A quarter or more of durvalumab patients experienced peripheral neuropathy, musculoskeletal pain, nausea, alopecia, fatigue, abdominal pain, constipation, rash, diarrhea, vomiting, and cough.
The recommended treatment regimen for dMMR endometrial cancer in women who weigh ≥ 30 kg is 1120 mg with carboplatin plus paclitaxel every 3 weeks for six cycles, followed by single-agent durvalumab 1500 mg every 4 weeks.
The price of 2.4 mL of durvalumab at a concentration of 50 mg/mL is $1027, according to drugs.com.
A version of this article appeared on Medscape.com.
Originally approved in 2017, the programmed death ligand 1 inhibitor caries previously approved indications for non–small cell lung cancer, biliary tract cancer, and hepatocellular carcinoma.
Approval of the new indication was based on the phase 3 DUO-E trial, which included 95 women with newly diagnosed advanced or recurrent dMMR endometrial cancer. Patients were randomized to durvalumab 1120 mg or placebo with carboplatin plus paclitaxel every 3 weeks for a maximum of six cycles followed by durvalumab 1500 mg every 4 weeks until disease progression.
Median progression-free survival (PFS) was 7 months in the placebo arm but not reached in the durvalumab group. Overall survival outcomes were immature at the PFS analysis.
A quarter or more of durvalumab patients experienced peripheral neuropathy, musculoskeletal pain, nausea, alopecia, fatigue, abdominal pain, constipation, rash, diarrhea, vomiting, and cough.
The recommended treatment regimen for dMMR endometrial cancer in women who weigh ≥ 30 kg is 1120 mg with carboplatin plus paclitaxel every 3 weeks for six cycles, followed by single-agent durvalumab 1500 mg every 4 weeks.
The price of 2.4 mL of durvalumab at a concentration of 50 mg/mL is $1027, according to drugs.com.
A version of this article appeared on Medscape.com.
Surgeons Most Likely to Behave Unprofessionally: Study
Most doctors mind their manners. But surgeons are the most likely to be reported for unprofessional behavior, while physicians practicing in pediatric settings are the least likely, according to a recent study of more than 35,000 physicians.
The research, published on June 6 in JAMA Network Open, found that fewer than 10% of physicians were reported by their coworkers for at least one instance of unprofessional behavior, and only 1% showed a pattern of such reports.
Data were gathered from the Center for Patient and Professional Advocacy’s (CPPA’s) Coworker Observation Reporting System (CORS) program, a national collaborative in which 193 participating hospitals and practice sites file safety-event reports involving medical workers’ unprofessional behaviors. An algorithm that weights CORS reports based on recency and severity was used to analyze the data. The study was spearheaded by William O. Cooper, MD, MPH, director of the CPPA at Vanderbilt University Medical Center, Nashville, Tennessee.
The retrospective cohort study included deidentified data on credentialed physicians, not including residents or fellows, who practiced at a CORS site between 2018 and 2022.
Why Surgeons?
The authors speculated that the reason surgeons were reported for unprofessional behavior more often than their colleagues in nonsurgical specialties was because surgery is a more stressful environment than other specialties and requires more teamwork, resulting in more interactions during high-stakes events.
Daniel Katz, MD, professor and vice chair of education for the Department of Anesthesiology, Perioperative and Pain Medicine at the Icahn School of Medicine at Mount Sinai, New York City, added that part of the problem is that surgeons are expected to perform at very high levels all the time.
“When things that are outside the control of the surgeon don’t go well,” Dr. Katz said, “that can lead to increased frustration and negative emotions, which will then bring out these kinds of behaviors.”
Types of Unprofessional Behaviors
The most common out-of-bounds behaviors reported involved disrespectful communication or lack of professional responsibility. In one example, a physician called a coworker a “bossy cow” when the coworker reminded the physician of the need to do a timeout before beginning a bronchoscopy.
In another case involving professional responsibility, a coworker asked a physician if the team should wait for a disoriented patient’s spouse to arrive. The doctor’s response: “We’ll be here all night if we do that. If you won’t sign as a witness, I’ll get someone else who will.”
The least common reports involved unprofessionalism related to medical care or professional integrity. One cited a physician removing a Foley catheter without wearing gloves and having visible urine on his hands and not washing them before touching other things in the room. In a reported lapse of professional integrity, a physician billed at level five after spending only 4 minutes with a patient.
Impact of Unprofessional Behavior
Unprofessional behavior among physicians is more than just unpleasant. It can threaten the functioning of teams and increase patient complications. In addition, individuals who model unprofessional behaviors are associated with increased malpractice claims, the study’s authors wrote.
Dr. Katz agreed that unprofessional behavior is damaging to both patients and the profession as a whole.
However, this doesn’t happen because some doctors are bad, he said. Physicians today are working in a pressure cooker. The current healthcare environment, with its increased administrative burdens, lack of staffing, and other problems, has increased the overall level of stress and led to burnout among healthcare personnel.
“You have to fix the system to create a working environment that doesn’t cause somebody to explode,” Dr. Katz said.
The goal of the CORS program and this study, Dr. Cooper said, is to help physicians better weather these stresses.
Study Limitations
The authors noted some weaknesses in the study. Some unprofessional behavior may go unreported because of fear of retaliation or for other reasons victims or witnesses did not feel safe to report their colleagues. Also, reports were not evaluated to ensure the truth of the accusations. The records reviewed did not include the gender of the physician, though the researchers pointed out that previous studies have shown that women are less likely than men to receive CORS reports.
A version of this article appeared on Medscape.com.
Most doctors mind their manners. But surgeons are the most likely to be reported for unprofessional behavior, while physicians practicing in pediatric settings are the least likely, according to a recent study of more than 35,000 physicians.
The research, published on June 6 in JAMA Network Open, found that fewer than 10% of physicians were reported by their coworkers for at least one instance of unprofessional behavior, and only 1% showed a pattern of such reports.
Data were gathered from the Center for Patient and Professional Advocacy’s (CPPA’s) Coworker Observation Reporting System (CORS) program, a national collaborative in which 193 participating hospitals and practice sites file safety-event reports involving medical workers’ unprofessional behaviors. An algorithm that weights CORS reports based on recency and severity was used to analyze the data. The study was spearheaded by William O. Cooper, MD, MPH, director of the CPPA at Vanderbilt University Medical Center, Nashville, Tennessee.
The retrospective cohort study included deidentified data on credentialed physicians, not including residents or fellows, who practiced at a CORS site between 2018 and 2022.
Why Surgeons?
The authors speculated that the reason surgeons were reported for unprofessional behavior more often than their colleagues in nonsurgical specialties was because surgery is a more stressful environment than other specialties and requires more teamwork, resulting in more interactions during high-stakes events.
Daniel Katz, MD, professor and vice chair of education for the Department of Anesthesiology, Perioperative and Pain Medicine at the Icahn School of Medicine at Mount Sinai, New York City, added that part of the problem is that surgeons are expected to perform at very high levels all the time.
“When things that are outside the control of the surgeon don’t go well,” Dr. Katz said, “that can lead to increased frustration and negative emotions, which will then bring out these kinds of behaviors.”
Types of Unprofessional Behaviors
The most common out-of-bounds behaviors reported involved disrespectful communication or lack of professional responsibility. In one example, a physician called a coworker a “bossy cow” when the coworker reminded the physician of the need to do a timeout before beginning a bronchoscopy.
In another case involving professional responsibility, a coworker asked a physician if the team should wait for a disoriented patient’s spouse to arrive. The doctor’s response: “We’ll be here all night if we do that. If you won’t sign as a witness, I’ll get someone else who will.”
The least common reports involved unprofessionalism related to medical care or professional integrity. One cited a physician removing a Foley catheter without wearing gloves and having visible urine on his hands and not washing them before touching other things in the room. In a reported lapse of professional integrity, a physician billed at level five after spending only 4 minutes with a patient.
Impact of Unprofessional Behavior
Unprofessional behavior among physicians is more than just unpleasant. It can threaten the functioning of teams and increase patient complications. In addition, individuals who model unprofessional behaviors are associated with increased malpractice claims, the study’s authors wrote.
Dr. Katz agreed that unprofessional behavior is damaging to both patients and the profession as a whole.
However, this doesn’t happen because some doctors are bad, he said. Physicians today are working in a pressure cooker. The current healthcare environment, with its increased administrative burdens, lack of staffing, and other problems, has increased the overall level of stress and led to burnout among healthcare personnel.
“You have to fix the system to create a working environment that doesn’t cause somebody to explode,” Dr. Katz said.
The goal of the CORS program and this study, Dr. Cooper said, is to help physicians better weather these stresses.
Study Limitations
The authors noted some weaknesses in the study. Some unprofessional behavior may go unreported because of fear of retaliation or for other reasons victims or witnesses did not feel safe to report their colleagues. Also, reports were not evaluated to ensure the truth of the accusations. The records reviewed did not include the gender of the physician, though the researchers pointed out that previous studies have shown that women are less likely than men to receive CORS reports.
A version of this article appeared on Medscape.com.
Most doctors mind their manners. But surgeons are the most likely to be reported for unprofessional behavior, while physicians practicing in pediatric settings are the least likely, according to a recent study of more than 35,000 physicians.
The research, published on June 6 in JAMA Network Open, found that fewer than 10% of physicians were reported by their coworkers for at least one instance of unprofessional behavior, and only 1% showed a pattern of such reports.
Data were gathered from the Center for Patient and Professional Advocacy’s (CPPA’s) Coworker Observation Reporting System (CORS) program, a national collaborative in which 193 participating hospitals and practice sites file safety-event reports involving medical workers’ unprofessional behaviors. An algorithm that weights CORS reports based on recency and severity was used to analyze the data. The study was spearheaded by William O. Cooper, MD, MPH, director of the CPPA at Vanderbilt University Medical Center, Nashville, Tennessee.
The retrospective cohort study included deidentified data on credentialed physicians, not including residents or fellows, who practiced at a CORS site between 2018 and 2022.
Why Surgeons?
The authors speculated that the reason surgeons were reported for unprofessional behavior more often than their colleagues in nonsurgical specialties was because surgery is a more stressful environment than other specialties and requires more teamwork, resulting in more interactions during high-stakes events.
Daniel Katz, MD, professor and vice chair of education for the Department of Anesthesiology, Perioperative and Pain Medicine at the Icahn School of Medicine at Mount Sinai, New York City, added that part of the problem is that surgeons are expected to perform at very high levels all the time.
“When things that are outside the control of the surgeon don’t go well,” Dr. Katz said, “that can lead to increased frustration and negative emotions, which will then bring out these kinds of behaviors.”
Types of Unprofessional Behaviors
The most common out-of-bounds behaviors reported involved disrespectful communication or lack of professional responsibility. In one example, a physician called a coworker a “bossy cow” when the coworker reminded the physician of the need to do a timeout before beginning a bronchoscopy.
In another case involving professional responsibility, a coworker asked a physician if the team should wait for a disoriented patient’s spouse to arrive. The doctor’s response: “We’ll be here all night if we do that. If you won’t sign as a witness, I’ll get someone else who will.”
The least common reports involved unprofessionalism related to medical care or professional integrity. One cited a physician removing a Foley catheter without wearing gloves and having visible urine on his hands and not washing them before touching other things in the room. In a reported lapse of professional integrity, a physician billed at level five after spending only 4 minutes with a patient.
Impact of Unprofessional Behavior
Unprofessional behavior among physicians is more than just unpleasant. It can threaten the functioning of teams and increase patient complications. In addition, individuals who model unprofessional behaviors are associated with increased malpractice claims, the study’s authors wrote.
Dr. Katz agreed that unprofessional behavior is damaging to both patients and the profession as a whole.
However, this doesn’t happen because some doctors are bad, he said. Physicians today are working in a pressure cooker. The current healthcare environment, with its increased administrative burdens, lack of staffing, and other problems, has increased the overall level of stress and led to burnout among healthcare personnel.
“You have to fix the system to create a working environment that doesn’t cause somebody to explode,” Dr. Katz said.
The goal of the CORS program and this study, Dr. Cooper said, is to help physicians better weather these stresses.
Study Limitations
The authors noted some weaknesses in the study. Some unprofessional behavior may go unreported because of fear of retaliation or for other reasons victims or witnesses did not feel safe to report their colleagues. Also, reports were not evaluated to ensure the truth of the accusations. The records reviewed did not include the gender of the physician, though the researchers pointed out that previous studies have shown that women are less likely than men to receive CORS reports.
A version of this article appeared on Medscape.com.
DEA Training Mandate: 8 Hours of My Life I’d Like Back
It’s time to renew two of my three narcotic prescribing licenses. For the first time in my career, I’ve waffled on whether the financial outlay to the US Drug Enforcement Agency (DEA) is worth it.
At $888 each, I’ve considered letting two licenses lapse because I only work part-time in Montana. But several friends advised me to keep a “spare” in case I transfer to a new location.
I thought about just paying the fees until I could do a little more research, but there is no mechanism for a refund unless I die within the first year of the 3-year cycle, provide incorrect credit card digits, or accidentally duplicate payments.
The renewal fee is just part of the issue.
Mandatory 8-Hour Training
I also received an alert about the requirement for more “narcotics prescribing education” thanks to the Medication Access and Training Expansion Act (MATE).
The requirement seems counterintuitive because opioid prescribing has decreased for the 10th consecutive year, according to the AMA Overdose Epidemic Report. The continuing rise in overdose deaths is largely due to illegitimate manufacturing of synthetic opioids.
I’ve written zero outpatient narcotics prescriptions in the past 6 years, and I’ve written very few in my 33 years of practice. My use is limited to intravenous morphine for flash pulmonary edema or refractory angina, but unless you graduated from a training program within 5 years of the June 2023 mandate or are boarded in addiction medicine, there is no way to escape the 8-hour education requirement.
The problem is that these courses are never just 8 hours in duration. After signing up for one such CME course that cost $150, I was still dying of boredom and at risk for DVT 4 days later. That’s how long it took to sit through.
Instead of the 30 seconds it should have taken to review the simple instructions to deliver Narcan, there were scores of screens followed by juvenile quizlets and cartoons. All but about 2 hours out of the 4 days is now relegated to that category of “hours of my life that I can never get back.” Additionally, none of that mandatory “education” will change my prescribing habits one whit.
And beware the penalty. 
Of course, I would always be truthful when asked to check the box on the DEA renewal application attesting to my having completed the required education. On the outside chance that you plan to check the yes box without completing the relevant courses, those found guilty of such false claims could be fined up to $250,000 and subject to “not more than four years in prison,” or both. Yikes!
Larry Houck, a former DEA investigator, explained that “[t]here are lot of people who are coming up for renewal and log on but still don’t know this is a requirement.” Neither ignorance nor complacency is an acceptable defense.
Changes Needed
The only good thing that came of those 4 long days of opioid education was a motivation to drive change in our current licensing and educational experience. Why not use this opportunity to reform the DEA-physician/prescriber relationship?
The educational requirements should be curtailed for those of us who do not provide outpatient narcotic prescriptions even if we use inpatient opioids. Meds with low abuse potential should be rescheduled to minimize who gets caught in the broad net of the education requirement.
We should reduce overregulation of the legitimate prescribers by lowering, instead of increasing, licensing fees. We should change to a single license number that covers every state. In this digital age, there is no legitimate excuse to prevent this from happening.
After all, the settlements from opioid manufacturers and distributors will in time total $50 billion. It seems that at least some of the responsibilities of the DEA could shift to states, cities, and towns.
My friend Siamak Karimian, MD, who provides locum services in multiple states, pays for seven active DEA licenses every 3 years. He pointed out the hypocrisy in the current regulatory system: “It’s funny that you can have only one DEA or state license and work for the government in all other states or territories with no limits, including the VA, Indian healthcare systems, or prison systems.”
All other prescribers require a separate DEA number for every state. Ultimately, you’d think tracking prescriptions for a single DEA number should be far simpler than tracking someone with seven.
Competent physicians not guilty of criminal overprescribing seem to be the last to be considered in nearly every healthcare endeavor these days. It would be refreshing if they would reduce our fees and prevent this waste of our time.
And while we are at it, perhaps a more fitting punishment is due for Richard Sackler and all the Purdue Pharma–affiliated family members. The Sacklers will pay out $6 billion in exchange for immunity against civil litigation. That doesn’t seem like much when they are worth $11 billion.
Perhaps they should be made to take an 8-hour course on opioid prescribing, annually and in perpetuity. Let’s see them complete a few quizlets and sit through screens of instruction on how to administer Naloxone. Of course, that would be a mild punishment for those who manufactured a drug that killed hundreds of thousands. But it would be a start.
Dr. Walton-Shirley, a clinical cardiologist in Nashville, Tennessee, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
It’s time to renew two of my three narcotic prescribing licenses. For the first time in my career, I’ve waffled on whether the financial outlay to the US Drug Enforcement Agency (DEA) is worth it.
At $888 each, I’ve considered letting two licenses lapse because I only work part-time in Montana. But several friends advised me to keep a “spare” in case I transfer to a new location.
I thought about just paying the fees until I could do a little more research, but there is no mechanism for a refund unless I die within the first year of the 3-year cycle, provide incorrect credit card digits, or accidentally duplicate payments.
The renewal fee is just part of the issue.
Mandatory 8-Hour Training
I also received an alert about the requirement for more “narcotics prescribing education” thanks to the Medication Access and Training Expansion Act (MATE).
The requirement seems counterintuitive because opioid prescribing has decreased for the 10th consecutive year, according to the AMA Overdose Epidemic Report. The continuing rise in overdose deaths is largely due to illegitimate manufacturing of synthetic opioids.
I’ve written zero outpatient narcotics prescriptions in the past 6 years, and I’ve written very few in my 33 years of practice. My use is limited to intravenous morphine for flash pulmonary edema or refractory angina, but unless you graduated from a training program within 5 years of the June 2023 mandate or are boarded in addiction medicine, there is no way to escape the 8-hour education requirement.
The problem is that these courses are never just 8 hours in duration. After signing up for one such CME course that cost $150, I was still dying of boredom and at risk for DVT 4 days later. That’s how long it took to sit through.
Instead of the 30 seconds it should have taken to review the simple instructions to deliver Narcan, there were scores of screens followed by juvenile quizlets and cartoons. All but about 2 hours out of the 4 days is now relegated to that category of “hours of my life that I can never get back.” Additionally, none of that mandatory “education” will change my prescribing habits one whit.
And beware the penalty. 
Of course, I would always be truthful when asked to check the box on the DEA renewal application attesting to my having completed the required education. On the outside chance that you plan to check the yes box without completing the relevant courses, those found guilty of such false claims could be fined up to $250,000 and subject to “not more than four years in prison,” or both. Yikes!
Larry Houck, a former DEA investigator, explained that “[t]here are lot of people who are coming up for renewal and log on but still don’t know this is a requirement.” Neither ignorance nor complacency is an acceptable defense.
Changes Needed
The only good thing that came of those 4 long days of opioid education was a motivation to drive change in our current licensing and educational experience. Why not use this opportunity to reform the DEA-physician/prescriber relationship?
The educational requirements should be curtailed for those of us who do not provide outpatient narcotic prescriptions even if we use inpatient opioids. Meds with low abuse potential should be rescheduled to minimize who gets caught in the broad net of the education requirement.
We should reduce overregulation of the legitimate prescribers by lowering, instead of increasing, licensing fees. We should change to a single license number that covers every state. In this digital age, there is no legitimate excuse to prevent this from happening.
After all, the settlements from opioid manufacturers and distributors will in time total $50 billion. It seems that at least some of the responsibilities of the DEA could shift to states, cities, and towns.
My friend Siamak Karimian, MD, who provides locum services in multiple states, pays for seven active DEA licenses every 3 years. He pointed out the hypocrisy in the current regulatory system: “It’s funny that you can have only one DEA or state license and work for the government in all other states or territories with no limits, including the VA, Indian healthcare systems, or prison systems.”
All other prescribers require a separate DEA number for every state. Ultimately, you’d think tracking prescriptions for a single DEA number should be far simpler than tracking someone with seven.
Competent physicians not guilty of criminal overprescribing seem to be the last to be considered in nearly every healthcare endeavor these days. It would be refreshing if they would reduce our fees and prevent this waste of our time.
And while we are at it, perhaps a more fitting punishment is due for Richard Sackler and all the Purdue Pharma–affiliated family members. The Sacklers will pay out $6 billion in exchange for immunity against civil litigation. That doesn’t seem like much when they are worth $11 billion.
Perhaps they should be made to take an 8-hour course on opioid prescribing, annually and in perpetuity. Let’s see them complete a few quizlets and sit through screens of instruction on how to administer Naloxone. Of course, that would be a mild punishment for those who manufactured a drug that killed hundreds of thousands. But it would be a start.
Dr. Walton-Shirley, a clinical cardiologist in Nashville, Tennessee, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
It’s time to renew two of my three narcotic prescribing licenses. For the first time in my career, I’ve waffled on whether the financial outlay to the US Drug Enforcement Agency (DEA) is worth it.
At $888 each, I’ve considered letting two licenses lapse because I only work part-time in Montana. But several friends advised me to keep a “spare” in case I transfer to a new location.
I thought about just paying the fees until I could do a little more research, but there is no mechanism for a refund unless I die within the first year of the 3-year cycle, provide incorrect credit card digits, or accidentally duplicate payments.
The renewal fee is just part of the issue.
Mandatory 8-Hour Training
I also received an alert about the requirement for more “narcotics prescribing education” thanks to the Medication Access and Training Expansion Act (MATE).
The requirement seems counterintuitive because opioid prescribing has decreased for the 10th consecutive year, according to the AMA Overdose Epidemic Report. The continuing rise in overdose deaths is largely due to illegitimate manufacturing of synthetic opioids.
I’ve written zero outpatient narcotics prescriptions in the past 6 years, and I’ve written very few in my 33 years of practice. My use is limited to intravenous morphine for flash pulmonary edema or refractory angina, but unless you graduated from a training program within 5 years of the June 2023 mandate or are boarded in addiction medicine, there is no way to escape the 8-hour education requirement.
The problem is that these courses are never just 8 hours in duration. After signing up for one such CME course that cost $150, I was still dying of boredom and at risk for DVT 4 days later. That’s how long it took to sit through.
Instead of the 30 seconds it should have taken to review the simple instructions to deliver Narcan, there were scores of screens followed by juvenile quizlets and cartoons. All but about 2 hours out of the 4 days is now relegated to that category of “hours of my life that I can never get back.” Additionally, none of that mandatory “education” will change my prescribing habits one whit.
And beware the penalty. 
Of course, I would always be truthful when asked to check the box on the DEA renewal application attesting to my having completed the required education. On the outside chance that you plan to check the yes box without completing the relevant courses, those found guilty of such false claims could be fined up to $250,000 and subject to “not more than four years in prison,” or both. Yikes!
Larry Houck, a former DEA investigator, explained that “[t]here are lot of people who are coming up for renewal and log on but still don’t know this is a requirement.” Neither ignorance nor complacency is an acceptable defense.
Changes Needed
The only good thing that came of those 4 long days of opioid education was a motivation to drive change in our current licensing and educational experience. Why not use this opportunity to reform the DEA-physician/prescriber relationship?
The educational requirements should be curtailed for those of us who do not provide outpatient narcotic prescriptions even if we use inpatient opioids. Meds with low abuse potential should be rescheduled to minimize who gets caught in the broad net of the education requirement.
We should reduce overregulation of the legitimate prescribers by lowering, instead of increasing, licensing fees. We should change to a single license number that covers every state. In this digital age, there is no legitimate excuse to prevent this from happening.
After all, the settlements from opioid manufacturers and distributors will in time total $50 billion. It seems that at least some of the responsibilities of the DEA could shift to states, cities, and towns.
My friend Siamak Karimian, MD, who provides locum services in multiple states, pays for seven active DEA licenses every 3 years. He pointed out the hypocrisy in the current regulatory system: “It’s funny that you can have only one DEA or state license and work for the government in all other states or territories with no limits, including the VA, Indian healthcare systems, or prison systems.”
All other prescribers require a separate DEA number for every state. Ultimately, you’d think tracking prescriptions for a single DEA number should be far simpler than tracking someone with seven.
Competent physicians not guilty of criminal overprescribing seem to be the last to be considered in nearly every healthcare endeavor these days. It would be refreshing if they would reduce our fees and prevent this waste of our time.
And while we are at it, perhaps a more fitting punishment is due for Richard Sackler and all the Purdue Pharma–affiliated family members. The Sacklers will pay out $6 billion in exchange for immunity against civil litigation. That doesn’t seem like much when they are worth $11 billion.
Perhaps they should be made to take an 8-hour course on opioid prescribing, annually and in perpetuity. Let’s see them complete a few quizlets and sit through screens of instruction on how to administer Naloxone. Of course, that would be a mild punishment for those who manufactured a drug that killed hundreds of thousands. But it would be a start.
Dr. Walton-Shirley, a clinical cardiologist in Nashville, Tennessee, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
AMA Wrestles With AI But Acts on Prior Authorization, Other Concerns
The largest US physician organization wrestled with the professional risks and rewards of artificial intelligence (AI) at its annual meeting, delaying action even as it adopted new policies on prior authorization and other concerns for clinicians and patients.
Physicians and medical students at the annual meeting of the American Medical Association (AMA) House of Delegates in Chicago intensely debated a report and two key resolutions on AI but could not reach consensus, pushing off decision-making until a future meeting in November.
One resolution would establish “augmented intelligence” as the preferred term for AI, reflecting the desired role of these tools in supporting — not making — physicians’ decisions. The other resolution focused on insurers’ use of AI in determining medical necessity.
(See specific policies adopted at the meeting, held June 8-12, below.)
A comprehensive AMA trustees’ report on AI considered additional issues including requirements for disclosing AI use, liability for harms due to flawed application of AI, data privacy, and cybersecurity.
The AMA intends to “continue to methodically assess these issues and make informed recommendations in proposing new policy,” said Bobby Mukkamala, MD, an otolaryngologist from Flint, Michigan, who became the AMA’s new president-elect.
AMA members at the meeting largely applauded the aim of these AI proposals, but some objected to parts of the trustees’ report.
They raised questions about what, exactly, constitutes an AI-powered service and whether all AI tools need the kind of guardrails the AMA may seek. There also were concerns about calls to make AI use more transparent.
While transparency might be an admirable goal, it might prove too hard to achieve given that AI-powered tools and products are already woven into medical practice in ways that physicians may not know or understand, said Christopher Libby, MD, MPH, a clinical informaticist and emergency physician at Cedars Sinai Medical Center in Los Angeles.
“It’s hard for the practicing clinician to know how every piece of technology works in order to describe it to the patient,” Dr. Libby said at the meeting. “How many people here can identify when algorithms are used in their EHR today?”
He suggested asking for more transparency from the companies that make and sell AI-powered software and tools to insurers and healthcare systems.
Steven H. Kroft, MD, the editor of the American Journal of Clinical Pathology, raised concerns about the unintended harm that unchecked use of AI may pose to scientific research.
He asked the AMA to address “a significant omission in an otherwise comprehensive report” — the need to protect the integrity of study results that can direct patient care.
“While sham science is not a new issue, large language models make it far easier for authors to generate fake papers and far harder for editors, reviewers, and publishers to identify them,” Dr. Kroft said. “This is a rapidly growing phenomenon that is threatening the integrity of the literature. These papers become embedded in the evidence bases that drive clinical decision-making.”
AMA has been working with specialty societies and outside AI experts to refine an effective set of recommendations. The new policies, once finalized, are intended to build on steps AMA already has taken, including last year releasing principles for AI development, deployment, and use.
Congress Mulling
The AMA delegates are far from alone in facing AI policy challenges.
Leaders in Congress also are examining AI guardrails, with influential panels such as the Senate Finance and House Energy and Commerce committees holding hearings.
A key congressional AI effort to watch is the expected implementation of a bipartisan Senate “road map,” which Senate Majority Leader Chuck Schumer (D-NY) and colleagues released in May, said Miranda A. Franco, a senior policy advisor at the law firm Holland & Knight.
The product of many months of deliberation, this Senate road map identifies priorities for future legislation, including:
- Creating appropriate guardrails and safety measures to protect patients.
- Making healthcare and biomedical data available for machine learning and data science research while carefully addressing privacy issues.
- Providing transparency for clinicians and the public about the use of AI in medical products and clinical support services, including the data used to train models.
- Examining the Centers for Medicare & Medicaid Services’ reimbursement mechanisms as well as guardrails to ensure accountability, appropriate use, and broad application of AI across all populations.
Congress likely will address issues of AI in healthcare in piecemeal fashion, taking on different aspects of these challenges at different times, Ms. Franco said. The Senate road map gives the key committees directions on where to proceed in their efforts to develop new laws.
“I think this is all going to be slow and rolling, not big and sweeping,” Ms. Franco told this news organization. “I don’t think we’re going to see an encompassing AI bill.”
AMA Policies Adopted on Other Issues
At the June meeting, AMA delegates adopted the following policies aiming to:
- Increase oversight and accountability of health insurers’ use of prior authorization controls on patient access to care.
- Encourage policy changes allowing physicians to receive loan forgiveness when they practice in an Indian Health Service, Tribal, or Urban Indian Health Program, similar to physicians practicing in a Veterans Administration facility.
- Advocate for federal policy that limits a patient’s out-of-pocket cost to be the same or less than the amount that a patient with traditional Medicare plus a Medigap plan would pay.
- Oppose state or national legislation that could criminalize in vitro fertilization.
- Limit what the AMA calls the “expensive” cost for Medicare Advantage enrollees who need physician-administered drugs or biologics.
- Help physicians address the handling of de-identified patient data in a rapidly changing digital health ecosystem.
- Support efforts to decriminalize the possession of non-prescribed buprenorphine for personal use by individuals who lack access to a physician for the treatment of opioid use disorder.
- Expand access to hearing, vision, and dental care. The new AMA policy advocates working with state medical associations to support coverage of hearing exams, hearing aids, cochlear implants, and vision exams and aids. The revised AMA policy also supports working with the American Dental Association and other national organizations to improve access to dental care for people enrolled in Medicare, Medicaid, and CHIP programs.
- Increase enrollment of more women and sexual and gender minority populations in clinical trials.
A version of this article first appeared on Medscape.com.
The largest US physician organization wrestled with the professional risks and rewards of artificial intelligence (AI) at its annual meeting, delaying action even as it adopted new policies on prior authorization and other concerns for clinicians and patients.
Physicians and medical students at the annual meeting of the American Medical Association (AMA) House of Delegates in Chicago intensely debated a report and two key resolutions on AI but could not reach consensus, pushing off decision-making until a future meeting in November.
One resolution would establish “augmented intelligence” as the preferred term for AI, reflecting the desired role of these tools in supporting — not making — physicians’ decisions. The other resolution focused on insurers’ use of AI in determining medical necessity.
(See specific policies adopted at the meeting, held June 8-12, below.)
A comprehensive AMA trustees’ report on AI considered additional issues including requirements for disclosing AI use, liability for harms due to flawed application of AI, data privacy, and cybersecurity.
The AMA intends to “continue to methodically assess these issues and make informed recommendations in proposing new policy,” said Bobby Mukkamala, MD, an otolaryngologist from Flint, Michigan, who became the AMA’s new president-elect.
AMA members at the meeting largely applauded the aim of these AI proposals, but some objected to parts of the trustees’ report.
They raised questions about what, exactly, constitutes an AI-powered service and whether all AI tools need the kind of guardrails the AMA may seek. There also were concerns about calls to make AI use more transparent.
While transparency might be an admirable goal, it might prove too hard to achieve given that AI-powered tools and products are already woven into medical practice in ways that physicians may not know or understand, said Christopher Libby, MD, MPH, a clinical informaticist and emergency physician at Cedars Sinai Medical Center in Los Angeles.
“It’s hard for the practicing clinician to know how every piece of technology works in order to describe it to the patient,” Dr. Libby said at the meeting. “How many people here can identify when algorithms are used in their EHR today?”
He suggested asking for more transparency from the companies that make and sell AI-powered software and tools to insurers and healthcare systems.
Steven H. Kroft, MD, the editor of the American Journal of Clinical Pathology, raised concerns about the unintended harm that unchecked use of AI may pose to scientific research.
He asked the AMA to address “a significant omission in an otherwise comprehensive report” — the need to protect the integrity of study results that can direct patient care.
“While sham science is not a new issue, large language models make it far easier for authors to generate fake papers and far harder for editors, reviewers, and publishers to identify them,” Dr. Kroft said. “This is a rapidly growing phenomenon that is threatening the integrity of the literature. These papers become embedded in the evidence bases that drive clinical decision-making.”
AMA has been working with specialty societies and outside AI experts to refine an effective set of recommendations. The new policies, once finalized, are intended to build on steps AMA already has taken, including last year releasing principles for AI development, deployment, and use.
Congress Mulling
The AMA delegates are far from alone in facing AI policy challenges.
Leaders in Congress also are examining AI guardrails, with influential panels such as the Senate Finance and House Energy and Commerce committees holding hearings.
A key congressional AI effort to watch is the expected implementation of a bipartisan Senate “road map,” which Senate Majority Leader Chuck Schumer (D-NY) and colleagues released in May, said Miranda A. Franco, a senior policy advisor at the law firm Holland & Knight.
The product of many months of deliberation, this Senate road map identifies priorities for future legislation, including:
- Creating appropriate guardrails and safety measures to protect patients.
- Making healthcare and biomedical data available for machine learning and data science research while carefully addressing privacy issues.
- Providing transparency for clinicians and the public about the use of AI in medical products and clinical support services, including the data used to train models.
- Examining the Centers for Medicare & Medicaid Services’ reimbursement mechanisms as well as guardrails to ensure accountability, appropriate use, and broad application of AI across all populations.
Congress likely will address issues of AI in healthcare in piecemeal fashion, taking on different aspects of these challenges at different times, Ms. Franco said. The Senate road map gives the key committees directions on where to proceed in their efforts to develop new laws.
“I think this is all going to be slow and rolling, not big and sweeping,” Ms. Franco told this news organization. “I don’t think we’re going to see an encompassing AI bill.”
AMA Policies Adopted on Other Issues
At the June meeting, AMA delegates adopted the following policies aiming to:
- Increase oversight and accountability of health insurers’ use of prior authorization controls on patient access to care.
- Encourage policy changes allowing physicians to receive loan forgiveness when they practice in an Indian Health Service, Tribal, or Urban Indian Health Program, similar to physicians practicing in a Veterans Administration facility.
- Advocate for federal policy that limits a patient’s out-of-pocket cost to be the same or less than the amount that a patient with traditional Medicare plus a Medigap plan would pay.
- Oppose state or national legislation that could criminalize in vitro fertilization.
- Limit what the AMA calls the “expensive” cost for Medicare Advantage enrollees who need physician-administered drugs or biologics.
- Help physicians address the handling of de-identified patient data in a rapidly changing digital health ecosystem.
- Support efforts to decriminalize the possession of non-prescribed buprenorphine for personal use by individuals who lack access to a physician for the treatment of opioid use disorder.
- Expand access to hearing, vision, and dental care. The new AMA policy advocates working with state medical associations to support coverage of hearing exams, hearing aids, cochlear implants, and vision exams and aids. The revised AMA policy also supports working with the American Dental Association and other national organizations to improve access to dental care for people enrolled in Medicare, Medicaid, and CHIP programs.
- Increase enrollment of more women and sexual and gender minority populations in clinical trials.
A version of this article first appeared on Medscape.com.
The largest US physician organization wrestled with the professional risks and rewards of artificial intelligence (AI) at its annual meeting, delaying action even as it adopted new policies on prior authorization and other concerns for clinicians and patients.
Physicians and medical students at the annual meeting of the American Medical Association (AMA) House of Delegates in Chicago intensely debated a report and two key resolutions on AI but could not reach consensus, pushing off decision-making until a future meeting in November.
One resolution would establish “augmented intelligence” as the preferred term for AI, reflecting the desired role of these tools in supporting — not making — physicians’ decisions. The other resolution focused on insurers’ use of AI in determining medical necessity.
(See specific policies adopted at the meeting, held June 8-12, below.)
A comprehensive AMA trustees’ report on AI considered additional issues including requirements for disclosing AI use, liability for harms due to flawed application of AI, data privacy, and cybersecurity.
The AMA intends to “continue to methodically assess these issues and make informed recommendations in proposing new policy,” said Bobby Mukkamala, MD, an otolaryngologist from Flint, Michigan, who became the AMA’s new president-elect.
AMA members at the meeting largely applauded the aim of these AI proposals, but some objected to parts of the trustees’ report.
They raised questions about what, exactly, constitutes an AI-powered service and whether all AI tools need the kind of guardrails the AMA may seek. There also were concerns about calls to make AI use more transparent.
While transparency might be an admirable goal, it might prove too hard to achieve given that AI-powered tools and products are already woven into medical practice in ways that physicians may not know or understand, said Christopher Libby, MD, MPH, a clinical informaticist and emergency physician at Cedars Sinai Medical Center in Los Angeles.
“It’s hard for the practicing clinician to know how every piece of technology works in order to describe it to the patient,” Dr. Libby said at the meeting. “How many people here can identify when algorithms are used in their EHR today?”
He suggested asking for more transparency from the companies that make and sell AI-powered software and tools to insurers and healthcare systems.
Steven H. Kroft, MD, the editor of the American Journal of Clinical Pathology, raised concerns about the unintended harm that unchecked use of AI may pose to scientific research.
He asked the AMA to address “a significant omission in an otherwise comprehensive report” — the need to protect the integrity of study results that can direct patient care.
“While sham science is not a new issue, large language models make it far easier for authors to generate fake papers and far harder for editors, reviewers, and publishers to identify them,” Dr. Kroft said. “This is a rapidly growing phenomenon that is threatening the integrity of the literature. These papers become embedded in the evidence bases that drive clinical decision-making.”
AMA has been working with specialty societies and outside AI experts to refine an effective set of recommendations. The new policies, once finalized, are intended to build on steps AMA already has taken, including last year releasing principles for AI development, deployment, and use.
Congress Mulling
The AMA delegates are far from alone in facing AI policy challenges.
Leaders in Congress also are examining AI guardrails, with influential panels such as the Senate Finance and House Energy and Commerce committees holding hearings.
A key congressional AI effort to watch is the expected implementation of a bipartisan Senate “road map,” which Senate Majority Leader Chuck Schumer (D-NY) and colleagues released in May, said Miranda A. Franco, a senior policy advisor at the law firm Holland & Knight.
The product of many months of deliberation, this Senate road map identifies priorities for future legislation, including:
- Creating appropriate guardrails and safety measures to protect patients.
- Making healthcare and biomedical data available for machine learning and data science research while carefully addressing privacy issues.
- Providing transparency for clinicians and the public about the use of AI in medical products and clinical support services, including the data used to train models.
- Examining the Centers for Medicare & Medicaid Services’ reimbursement mechanisms as well as guardrails to ensure accountability, appropriate use, and broad application of AI across all populations.
Congress likely will address issues of AI in healthcare in piecemeal fashion, taking on different aspects of these challenges at different times, Ms. Franco said. The Senate road map gives the key committees directions on where to proceed in their efforts to develop new laws.
“I think this is all going to be slow and rolling, not big and sweeping,” Ms. Franco told this news organization. “I don’t think we’re going to see an encompassing AI bill.”
AMA Policies Adopted on Other Issues
At the June meeting, AMA delegates adopted the following policies aiming to:
- Increase oversight and accountability of health insurers’ use of prior authorization controls on patient access to care.
- Encourage policy changes allowing physicians to receive loan forgiveness when they practice in an Indian Health Service, Tribal, or Urban Indian Health Program, similar to physicians practicing in a Veterans Administration facility.
- Advocate for federal policy that limits a patient’s out-of-pocket cost to be the same or less than the amount that a patient with traditional Medicare plus a Medigap plan would pay.
- Oppose state or national legislation that could criminalize in vitro fertilization.
- Limit what the AMA calls the “expensive” cost for Medicare Advantage enrollees who need physician-administered drugs or biologics.
- Help physicians address the handling of de-identified patient data in a rapidly changing digital health ecosystem.
- Support efforts to decriminalize the possession of non-prescribed buprenorphine for personal use by individuals who lack access to a physician for the treatment of opioid use disorder.
- Expand access to hearing, vision, and dental care. The new AMA policy advocates working with state medical associations to support coverage of hearing exams, hearing aids, cochlear implants, and vision exams and aids. The revised AMA policy also supports working with the American Dental Association and other national organizations to improve access to dental care for people enrolled in Medicare, Medicaid, and CHIP programs.
- Increase enrollment of more women and sexual and gender minority populations in clinical trials.
A version of this article first appeared on Medscape.com.
Listen to earn your patients’ trust
Recently, I had an interesting conversation while getting my hair cut. It gave me a great deal of insight into some of the problems we have right now with how medical information is shared and some of the disconnect our patients may feel.
The young woman who was cutting my hair asked me what I did for an occupation. I said that I was a physician. She said, “Can I please ask you an important question?” She asked me what my thoughts were about the COVID vaccine. She prefaced it with “I am so confused on whether I should get the vaccine. I have seen a number of TikTok videos that talk about nano particles in the COVID vaccine that can be very dangerous.”
I discussed with her how the COVID vaccine actually works and shared with her the remarkable success of the vaccine. I asked her what side effects she was worried about from the vaccine and what her fears were. She said that she had heard that a lot of people had died from the vaccine. I told her that severe reactions from the vaccine were very uncommon.
She then made a very telling comment: “I wish I could talk to a doctor about my concerns. I have been going to the same health center for the last 5 years and every time I go I see a different person.” She added, “I rarely have more than 5-10 minutes with the person that I am seeing and I rarely get the opportunity to ask questions.”
She thanked me for the information and said that she would be getting the COVID vaccine in the future. She said it is so hard to know where to get information now and the very different things that she heard confused her. She told me that she thought her generation got most of its information from short sound bites or TikTok and Instagram videos.
Why did she trust me? I still think that the medical profession is respected. We are all pressured to do more with less time. Conversations where we can listen and then respond go a long way. We can always listen and learn what information people need and will appreciate. I was also struck by how alone this person felt in our health care system. She did not have a relationship with any one person whom she could trust and reach out to with questions. Relationships with our patients go a long way to establishing trust.
Pearl
It takes time to listen to and answer our patients’ questions. We need to do that to fight the waves of misinformation our patients face.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
Recently, I had an interesting conversation while getting my hair cut. It gave me a great deal of insight into some of the problems we have right now with how medical information is shared and some of the disconnect our patients may feel.
The young woman who was cutting my hair asked me what I did for an occupation. I said that I was a physician. She said, “Can I please ask you an important question?” She asked me what my thoughts were about the COVID vaccine. She prefaced it with “I am so confused on whether I should get the vaccine. I have seen a number of TikTok videos that talk about nano particles in the COVID vaccine that can be very dangerous.”
I discussed with her how the COVID vaccine actually works and shared with her the remarkable success of the vaccine. I asked her what side effects she was worried about from the vaccine and what her fears were. She said that she had heard that a lot of people had died from the vaccine. I told her that severe reactions from the vaccine were very uncommon.
She then made a very telling comment: “I wish I could talk to a doctor about my concerns. I have been going to the same health center for the last 5 years and every time I go I see a different person.” She added, “I rarely have more than 5-10 minutes with the person that I am seeing and I rarely get the opportunity to ask questions.”
She thanked me for the information and said that she would be getting the COVID vaccine in the future. She said it is so hard to know where to get information now and the very different things that she heard confused her. She told me that she thought her generation got most of its information from short sound bites or TikTok and Instagram videos.
Why did she trust me? I still think that the medical profession is respected. We are all pressured to do more with less time. Conversations where we can listen and then respond go a long way. We can always listen and learn what information people need and will appreciate. I was also struck by how alone this person felt in our health care system. She did not have a relationship with any one person whom she could trust and reach out to with questions. Relationships with our patients go a long way to establishing trust.
Pearl
It takes time to listen to and answer our patients’ questions. We need to do that to fight the waves of misinformation our patients face.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
Recently, I had an interesting conversation while getting my hair cut. It gave me a great deal of insight into some of the problems we have right now with how medical information is shared and some of the disconnect our patients may feel.
The young woman who was cutting my hair asked me what I did for an occupation. I said that I was a physician. She said, “Can I please ask you an important question?” She asked me what my thoughts were about the COVID vaccine. She prefaced it with “I am so confused on whether I should get the vaccine. I have seen a number of TikTok videos that talk about nano particles in the COVID vaccine that can be very dangerous.”
I discussed with her how the COVID vaccine actually works and shared with her the remarkable success of the vaccine. I asked her what side effects she was worried about from the vaccine and what her fears were. She said that she had heard that a lot of people had died from the vaccine. I told her that severe reactions from the vaccine were very uncommon.
She then made a very telling comment: “I wish I could talk to a doctor about my concerns. I have been going to the same health center for the last 5 years and every time I go I see a different person.” She added, “I rarely have more than 5-10 minutes with the person that I am seeing and I rarely get the opportunity to ask questions.”
She thanked me for the information and said that she would be getting the COVID vaccine in the future. She said it is so hard to know where to get information now and the very different things that she heard confused her. She told me that she thought her generation got most of its information from short sound bites or TikTok and Instagram videos.
Why did she trust me? I still think that the medical profession is respected. We are all pressured to do more with less time. Conversations where we can listen and then respond go a long way. We can always listen and learn what information people need and will appreciate. I was also struck by how alone this person felt in our health care system. She did not have a relationship with any one person whom she could trust and reach out to with questions. Relationships with our patients go a long way to establishing trust.
Pearl
It takes time to listen to and answer our patients’ questions. We need to do that to fight the waves of misinformation our patients face.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
Knowing My Limits
The records came in by fax. A patient who’d recently moved here and needed to connect with a local neurologist.
When I had time, I flipped through the records. He needed ongoing treatment for a rare neurological disease that I’d heard of, but wasn’t otherwise familiar with. It didn’t even exist in the textbooks or conferences when I was in residency. I’d never seen a case of it, just read about it here and there in journals.
I looked it up, reviewed current treatment options, monitoring, and other knowledge about it, then stared at the notes for a minute. Finally, after thinking it over, I attached a sticky note for my secretary that, if the person called, to redirect them to one of the local subspecialty neurology centers.
I have nothing against this patient, but realistically he would be better served seeing someone with time to keep up on advancements in esoteric disorders, not a general neurologist like myself.
Isn’t that why we have subspecialty centers?
Some of it is also me. There was a time in my career when keeping up on newly discovered disorders and their treatments was, well, cool. But after 25 years in practice, that changes.
It’s important to be at least somewhat aware of new developments (such as in this case) as you may encounter them, and need to know when it’s something you can handle and when to send it elsewhere.
Driving home that afternoon I thought, “I’m an old dog. I don’t want to learn new tricks.” Maybe that’s all it is. There are other neurologists my age and older who thrive on the challenge of learning about and treating new and rare disorders that were unknown when they started out. There’s nothing wrong with that.
But I’ve never pretended to be an academic or sub-sub-specialist. My patients depend on me to stay up to date on the large number of commonly seen neurological disorders, and I do my best to do that.
It ain’t easy being an old dog.
Dr. Block has a solo neurology practice in Scottsdale, Arizona.
The records came in by fax. A patient who’d recently moved here and needed to connect with a local neurologist.
When I had time, I flipped through the records. He needed ongoing treatment for a rare neurological disease that I’d heard of, but wasn’t otherwise familiar with. It didn’t even exist in the textbooks or conferences when I was in residency. I’d never seen a case of it, just read about it here and there in journals.
I looked it up, reviewed current treatment options, monitoring, and other knowledge about it, then stared at the notes for a minute. Finally, after thinking it over, I attached a sticky note for my secretary that, if the person called, to redirect them to one of the local subspecialty neurology centers.
I have nothing against this patient, but realistically he would be better served seeing someone with time to keep up on advancements in esoteric disorders, not a general neurologist like myself.
Isn’t that why we have subspecialty centers?
Some of it is also me. There was a time in my career when keeping up on newly discovered disorders and their treatments was, well, cool. But after 25 years in practice, that changes.
It’s important to be at least somewhat aware of new developments (such as in this case) as you may encounter them, and need to know when it’s something you can handle and when to send it elsewhere.
Driving home that afternoon I thought, “I’m an old dog. I don’t want to learn new tricks.” Maybe that’s all it is. There are other neurologists my age and older who thrive on the challenge of learning about and treating new and rare disorders that were unknown when they started out. There’s nothing wrong with that.
But I’ve never pretended to be an academic or sub-sub-specialist. My patients depend on me to stay up to date on the large number of commonly seen neurological disorders, and I do my best to do that.
It ain’t easy being an old dog.
Dr. Block has a solo neurology practice in Scottsdale, Arizona.
The records came in by fax. A patient who’d recently moved here and needed to connect with a local neurologist.
When I had time, I flipped through the records. He needed ongoing treatment for a rare neurological disease that I’d heard of, but wasn’t otherwise familiar with. It didn’t even exist in the textbooks or conferences when I was in residency. I’d never seen a case of it, just read about it here and there in journals.
I looked it up, reviewed current treatment options, monitoring, and other knowledge about it, then stared at the notes for a minute. Finally, after thinking it over, I attached a sticky note for my secretary that, if the person called, to redirect them to one of the local subspecialty neurology centers.
I have nothing against this patient, but realistically he would be better served seeing someone with time to keep up on advancements in esoteric disorders, not a general neurologist like myself.
Isn’t that why we have subspecialty centers?
Some of it is also me. There was a time in my career when keeping up on newly discovered disorders and their treatments was, well, cool. But after 25 years in practice, that changes.
It’s important to be at least somewhat aware of new developments (such as in this case) as you may encounter them, and need to know when it’s something you can handle and when to send it elsewhere.
Driving home that afternoon I thought, “I’m an old dog. I don’t want to learn new tricks.” Maybe that’s all it is. There are other neurologists my age and older who thrive on the challenge of learning about and treating new and rare disorders that were unknown when they started out. There’s nothing wrong with that.
But I’ve never pretended to be an academic or sub-sub-specialist. My patients depend on me to stay up to date on the large number of commonly seen neurological disorders, and I do my best to do that.
It ain’t easy being an old dog.
Dr. Block has a solo neurology practice in Scottsdale, Arizona.
Vitamin D Test Inaccuracies Persist Despite Gains in Field: CDC
Some vitamin D tests may give misleading results despite progress made in recent years to improve the quality of these assays, according to the US Centers for Disease Control and Prevention (CDC).
Otoe Sugahara manager of the CDC Vitamin D Standardization-Certification Program (VDSCP), presented an update of her group’s work at ENDO 2024, the Endocrine Society’s annual meeting in Boston.
“Though most vitamin D tests in our program have improved, there still remain some sample-specific inaccuracies. The CDC is working with program participants to address these situations,” Ms. Sugahara said in a statement released by the Endocrine Society.
For example, some assays measure other compounds besides 25-hydroxyvitamin D, which can falsely elevate results of some blood samples, Ms. Sugahara reported. Thus, some tests may be misclassified, with results seen as sufficient from samples that should have indicated a vitamin D deficiency.
“While most vitamin D tests are effective, it is important for healthcare providers to be aware of the potential inconsistencies associated with vitamin D tests to avoid misclassification of the patients,” Ms. Sugahara and coauthors said in an abstract provided by the Endocrine Society.
Ms. Sugahara’s report provided a snapshot of the state of longstanding efforts to improve the quality of a widely performed service in US healthcare: testing vitamin D levels.
These include an international collaboration that gave rise in 2010 to a vitamin D standardization program, from which the CDC’s VDSCP certification emerged. Among the leaders of these efforts was Christopher Sempos, PhD, then with the Office of Dietary Supplements at the National Institutes of Health.
Many clinicians may not be aware of the concerns about the accuracy of vitamin D tests that led to the drive for standardization, Dr. Sempos, now retired, said in an interview. And, in his view, it’s something that busy practitioners should not have to consider.
“They have literally thousands of diseases they have to be able to recognize and diagnose,” Dr. Sempos said. “They should be able to count on the laboratory system to give them accurate and precise data.”
‘Nudging’ Toward Better Results
The CDC’s certification program gives labs and companies detailed information about the analytical accuracy and precision of their vitamin D tests.
This feedback has paid off with improved results, Andy Hoofnagle, MD, PhD, professor of laboratory medicine and pathology at the University of Washington in Seattle, told this news organization. It helps by “nudging manufacturers in the right direction,” he said.
“Some manufacturers reformulated, others recalibrated, which is a lot of effort on their part, so that when the patient get a number, it actually means the right thing,” said Dr. Hoofnagle, who is also chair of the Accuracy-Based Programs Committee of the College of American Pathologists.
“There are still many immunoassays on the market that aren’t giving the correct results, unfortunately, but the standardization certification program has really pushed the field in the right direction,” he said.
US scientists use two main types of technologies to measure vitamin D in the blood, Ms. Sugahara said. One is mass spectrometry, which separately measures 25-hydroxyvitamin D2 and D3 and sums the values. The other type, immunoassay, measures both compounds at the same time and reports one result for total 25-hydroxyvitamin D.
At the ENDO 2024 meeting, Ms. Sugahara reported generally positive trends seen in the VDSCP. For example, the program looks at specific tests’ bias, or the deviation of test results from the true value, as determined with the CDC’s reference method for vitamin D.
Average calibration bias was less than 1% for all assays in the VDSCP in 2022, Ms. Sugahara said. The average calibration bias for immunoassays was 0.86%, and for assays using mass spectrometry, it was 0.55%, Ms. Sugahara reported.
These are improved results compared with 2019 data, in which mass spectrometry–based assays had a mean bias of 1.9% and immunoassays had a mean bias of 2.4%, the CDC told this news organization in an email exchange.
The CDC said the VDSCP supports laboratories and researchers from around the world, including ones based in the US, China, Australia, Japan, and Korea.
Call for Research
Vitamin D tests are widely administered despite questions about their benefit for people who do not appear likely to be deficient of it.
The Endocrine Society’s newly released practice guideline recommends against routine testing of blood vitamin D levels in the general population.
Laboratory testing has increased over the years owing to studies reporting associations between blood vitamin D [25(OH)D] levels and a variety of common disorders, including musculoskeletal, metabolic, cardiovascular, malignant, autoimmune, and infectious diseases, wrote Marie B. Demay, MD, of Harvard Medical School in Boston, and coauthors in the new guideline. It was published on June 3 in The Journal of Clinical Endocrinology & Metabolism.
‘”Although a causal link between serum 25(OH)D concentrations and many disorders has not been clearly established, these associations have led to widespread supplementation with vitamin D and increased laboratory testing for 25(OH)D in the general population,” they wrote.
It’s uncertain that “any putative benefits of screening would outweigh the increased burden and cost, and whether implementation of universal 25(OH)D screening would be feasible from a societal perspective,” Dr. Demay and coauthors added.
They noted that the influential US Preventive Services Task Force also has raised doubts about widespread use of vitamin D tests.
The USPSTF has a somewhat different take from the Endocrine Society. The task force in 2021 reiterated its view that there is not enough evidence to recommend for or against widespread vitamin D testing for adults. The task force gave this test an I grade, meaning there is insufficient evidence to weigh the risks and benefits. That’s the same grade the task force gave it in 2014.
The USPSTF uses a grade of D to recommend against use of a test or service.
In an interview with this news organization, John Wong, MD, vice chair of the USPSTF, reiterated his group’s call for more research into the potential benefits and harms of vitamin D screening.
One of the challenges in addressing this issue, Dr. Wong noted, has been the variability of test results. Therefore, efforts such as the CDC’s VDSCP in improving test quality may help in eventually building up the kind of evidence base needed for the task force to offer a more definitive judgment on the tests, he said.
Wong acknowledged it must be frustrating for clinicians and patients to hear that experts don’t have the evidence needed to make a broad call about whether routine vitamin D tests are beneficial.
“We really would like to have that evidence because we recognize that it’s an important health question to help everybody in this nation stay healthy and live longer,” Dr. Wong said.
A version of this article appeared on Medscape.com.
Some vitamin D tests may give misleading results despite progress made in recent years to improve the quality of these assays, according to the US Centers for Disease Control and Prevention (CDC).
Otoe Sugahara manager of the CDC Vitamin D Standardization-Certification Program (VDSCP), presented an update of her group’s work at ENDO 2024, the Endocrine Society’s annual meeting in Boston.
“Though most vitamin D tests in our program have improved, there still remain some sample-specific inaccuracies. The CDC is working with program participants to address these situations,” Ms. Sugahara said in a statement released by the Endocrine Society.
For example, some assays measure other compounds besides 25-hydroxyvitamin D, which can falsely elevate results of some blood samples, Ms. Sugahara reported. Thus, some tests may be misclassified, with results seen as sufficient from samples that should have indicated a vitamin D deficiency.
“While most vitamin D tests are effective, it is important for healthcare providers to be aware of the potential inconsistencies associated with vitamin D tests to avoid misclassification of the patients,” Ms. Sugahara and coauthors said in an abstract provided by the Endocrine Society.
Ms. Sugahara’s report provided a snapshot of the state of longstanding efforts to improve the quality of a widely performed service in US healthcare: testing vitamin D levels.
These include an international collaboration that gave rise in 2010 to a vitamin D standardization program, from which the CDC’s VDSCP certification emerged. Among the leaders of these efforts was Christopher Sempos, PhD, then with the Office of Dietary Supplements at the National Institutes of Health.
Many clinicians may not be aware of the concerns about the accuracy of vitamin D tests that led to the drive for standardization, Dr. Sempos, now retired, said in an interview. And, in his view, it’s something that busy practitioners should not have to consider.
“They have literally thousands of diseases they have to be able to recognize and diagnose,” Dr. Sempos said. “They should be able to count on the laboratory system to give them accurate and precise data.”
‘Nudging’ Toward Better Results
The CDC’s certification program gives labs and companies detailed information about the analytical accuracy and precision of their vitamin D tests.
This feedback has paid off with improved results, Andy Hoofnagle, MD, PhD, professor of laboratory medicine and pathology at the University of Washington in Seattle, told this news organization. It helps by “nudging manufacturers in the right direction,” he said.
“Some manufacturers reformulated, others recalibrated, which is a lot of effort on their part, so that when the patient get a number, it actually means the right thing,” said Dr. Hoofnagle, who is also chair of the Accuracy-Based Programs Committee of the College of American Pathologists.
“There are still many immunoassays on the market that aren’t giving the correct results, unfortunately, but the standardization certification program has really pushed the field in the right direction,” he said.
US scientists use two main types of technologies to measure vitamin D in the blood, Ms. Sugahara said. One is mass spectrometry, which separately measures 25-hydroxyvitamin D2 and D3 and sums the values. The other type, immunoassay, measures both compounds at the same time and reports one result for total 25-hydroxyvitamin D.
At the ENDO 2024 meeting, Ms. Sugahara reported generally positive trends seen in the VDSCP. For example, the program looks at specific tests’ bias, or the deviation of test results from the true value, as determined with the CDC’s reference method for vitamin D.
Average calibration bias was less than 1% for all assays in the VDSCP in 2022, Ms. Sugahara said. The average calibration bias for immunoassays was 0.86%, and for assays using mass spectrometry, it was 0.55%, Ms. Sugahara reported.
These are improved results compared with 2019 data, in which mass spectrometry–based assays had a mean bias of 1.9% and immunoassays had a mean bias of 2.4%, the CDC told this news organization in an email exchange.
The CDC said the VDSCP supports laboratories and researchers from around the world, including ones based in the US, China, Australia, Japan, and Korea.
Call for Research
Vitamin D tests are widely administered despite questions about their benefit for people who do not appear likely to be deficient of it.
The Endocrine Society’s newly released practice guideline recommends against routine testing of blood vitamin D levels in the general population.
Laboratory testing has increased over the years owing to studies reporting associations between blood vitamin D [25(OH)D] levels and a variety of common disorders, including musculoskeletal, metabolic, cardiovascular, malignant, autoimmune, and infectious diseases, wrote Marie B. Demay, MD, of Harvard Medical School in Boston, and coauthors in the new guideline. It was published on June 3 in The Journal of Clinical Endocrinology & Metabolism.
‘”Although a causal link between serum 25(OH)D concentrations and many disorders has not been clearly established, these associations have led to widespread supplementation with vitamin D and increased laboratory testing for 25(OH)D in the general population,” they wrote.
It’s uncertain that “any putative benefits of screening would outweigh the increased burden and cost, and whether implementation of universal 25(OH)D screening would be feasible from a societal perspective,” Dr. Demay and coauthors added.
They noted that the influential US Preventive Services Task Force also has raised doubts about widespread use of vitamin D tests.
The USPSTF has a somewhat different take from the Endocrine Society. The task force in 2021 reiterated its view that there is not enough evidence to recommend for or against widespread vitamin D testing for adults. The task force gave this test an I grade, meaning there is insufficient evidence to weigh the risks and benefits. That’s the same grade the task force gave it in 2014.
The USPSTF uses a grade of D to recommend against use of a test or service.
In an interview with this news organization, John Wong, MD, vice chair of the USPSTF, reiterated his group’s call for more research into the potential benefits and harms of vitamin D screening.
One of the challenges in addressing this issue, Dr. Wong noted, has been the variability of test results. Therefore, efforts such as the CDC’s VDSCP in improving test quality may help in eventually building up the kind of evidence base needed for the task force to offer a more definitive judgment on the tests, he said.
Wong acknowledged it must be frustrating for clinicians and patients to hear that experts don’t have the evidence needed to make a broad call about whether routine vitamin D tests are beneficial.
“We really would like to have that evidence because we recognize that it’s an important health question to help everybody in this nation stay healthy and live longer,” Dr. Wong said.
A version of this article appeared on Medscape.com.
Some vitamin D tests may give misleading results despite progress made in recent years to improve the quality of these assays, according to the US Centers for Disease Control and Prevention (CDC).
Otoe Sugahara manager of the CDC Vitamin D Standardization-Certification Program (VDSCP), presented an update of her group’s work at ENDO 2024, the Endocrine Society’s annual meeting in Boston.
“Though most vitamin D tests in our program have improved, there still remain some sample-specific inaccuracies. The CDC is working with program participants to address these situations,” Ms. Sugahara said in a statement released by the Endocrine Society.
For example, some assays measure other compounds besides 25-hydroxyvitamin D, which can falsely elevate results of some blood samples, Ms. Sugahara reported. Thus, some tests may be misclassified, with results seen as sufficient from samples that should have indicated a vitamin D deficiency.
“While most vitamin D tests are effective, it is important for healthcare providers to be aware of the potential inconsistencies associated with vitamin D tests to avoid misclassification of the patients,” Ms. Sugahara and coauthors said in an abstract provided by the Endocrine Society.
Ms. Sugahara’s report provided a snapshot of the state of longstanding efforts to improve the quality of a widely performed service in US healthcare: testing vitamin D levels.
These include an international collaboration that gave rise in 2010 to a vitamin D standardization program, from which the CDC’s VDSCP certification emerged. Among the leaders of these efforts was Christopher Sempos, PhD, then with the Office of Dietary Supplements at the National Institutes of Health.
Many clinicians may not be aware of the concerns about the accuracy of vitamin D tests that led to the drive for standardization, Dr. Sempos, now retired, said in an interview. And, in his view, it’s something that busy practitioners should not have to consider.
“They have literally thousands of diseases they have to be able to recognize and diagnose,” Dr. Sempos said. “They should be able to count on the laboratory system to give them accurate and precise data.”
‘Nudging’ Toward Better Results
The CDC’s certification program gives labs and companies detailed information about the analytical accuracy and precision of their vitamin D tests.
This feedback has paid off with improved results, Andy Hoofnagle, MD, PhD, professor of laboratory medicine and pathology at the University of Washington in Seattle, told this news organization. It helps by “nudging manufacturers in the right direction,” he said.
“Some manufacturers reformulated, others recalibrated, which is a lot of effort on their part, so that when the patient get a number, it actually means the right thing,” said Dr. Hoofnagle, who is also chair of the Accuracy-Based Programs Committee of the College of American Pathologists.
“There are still many immunoassays on the market that aren’t giving the correct results, unfortunately, but the standardization certification program has really pushed the field in the right direction,” he said.
US scientists use two main types of technologies to measure vitamin D in the blood, Ms. Sugahara said. One is mass spectrometry, which separately measures 25-hydroxyvitamin D2 and D3 and sums the values. The other type, immunoassay, measures both compounds at the same time and reports one result for total 25-hydroxyvitamin D.
At the ENDO 2024 meeting, Ms. Sugahara reported generally positive trends seen in the VDSCP. For example, the program looks at specific tests’ bias, or the deviation of test results from the true value, as determined with the CDC’s reference method for vitamin D.
Average calibration bias was less than 1% for all assays in the VDSCP in 2022, Ms. Sugahara said. The average calibration bias for immunoassays was 0.86%, and for assays using mass spectrometry, it was 0.55%, Ms. Sugahara reported.
These are improved results compared with 2019 data, in which mass spectrometry–based assays had a mean bias of 1.9% and immunoassays had a mean bias of 2.4%, the CDC told this news organization in an email exchange.
The CDC said the VDSCP supports laboratories and researchers from around the world, including ones based in the US, China, Australia, Japan, and Korea.
Call for Research
Vitamin D tests are widely administered despite questions about their benefit for people who do not appear likely to be deficient of it.
The Endocrine Society’s newly released practice guideline recommends against routine testing of blood vitamin D levels in the general population.
Laboratory testing has increased over the years owing to studies reporting associations between blood vitamin D [25(OH)D] levels and a variety of common disorders, including musculoskeletal, metabolic, cardiovascular, malignant, autoimmune, and infectious diseases, wrote Marie B. Demay, MD, of Harvard Medical School in Boston, and coauthors in the new guideline. It was published on June 3 in The Journal of Clinical Endocrinology & Metabolism.
‘”Although a causal link between serum 25(OH)D concentrations and many disorders has not been clearly established, these associations have led to widespread supplementation with vitamin D and increased laboratory testing for 25(OH)D in the general population,” they wrote.
It’s uncertain that “any putative benefits of screening would outweigh the increased burden and cost, and whether implementation of universal 25(OH)D screening would be feasible from a societal perspective,” Dr. Demay and coauthors added.
They noted that the influential US Preventive Services Task Force also has raised doubts about widespread use of vitamin D tests.
The USPSTF has a somewhat different take from the Endocrine Society. The task force in 2021 reiterated its view that there is not enough evidence to recommend for or against widespread vitamin D testing for adults. The task force gave this test an I grade, meaning there is insufficient evidence to weigh the risks and benefits. That’s the same grade the task force gave it in 2014.
The USPSTF uses a grade of D to recommend against use of a test or service.
In an interview with this news organization, John Wong, MD, vice chair of the USPSTF, reiterated his group’s call for more research into the potential benefits and harms of vitamin D screening.
One of the challenges in addressing this issue, Dr. Wong noted, has been the variability of test results. Therefore, efforts such as the CDC’s VDSCP in improving test quality may help in eventually building up the kind of evidence base needed for the task force to offer a more definitive judgment on the tests, he said.
Wong acknowledged it must be frustrating for clinicians and patients to hear that experts don’t have the evidence needed to make a broad call about whether routine vitamin D tests are beneficial.
“We really would like to have that evidence because we recognize that it’s an important health question to help everybody in this nation stay healthy and live longer,” Dr. Wong said.
A version of this article appeared on Medscape.com.
Clear Coverage Preference for Humira Over Biosimilars Seen in Most Medicare Part D Plans
Despite the influx of adalimumab biosimilars entering the market in 2023, Humira remains on top.
As of January 2024, both high and low concentrations of Humira, the originator adalimumab product, are nearly universally covered by Medicare Part D plans, while only half of these plans covered adalimumab biosimilars, according to a new research letter published online on June 6, 2024, in JAMA.
Of the plans that covered both, only 1.5% had lower-tier placement for biosimilars.
“This study of formulary coverage helps explain limited uptake of adalimumab biosimilars,” wrote the authors, led by Matthew J. Klebanoff, MD, of the University of Pennsylvania, Philadelphia. “Subpar biosimilar adoption will not only undermine their potential to reduce spending but also may deter investments in biosimilar development.”
The analysis included the formulary and enrollment files for 5609 Medicare Part D plans, representing 44.4 million beneficiaries. Drug list prices and whole acquisition costs (WAC) were pulled from the Red Book database, which provides prices for prescription and over-the-counter drugs as well as medical devices and supplies.
Nearly all (98.9%) of Part D plans covered the high-concentration (100 mg/mL) version of adalimumab with a WAC of $6923. This higher concentration is the most popular formulation of the drug, making up an estimated 85% of prescriptions. By comparison, 26.8% of plans covered the high-concentration version of adalimumab-adaz (Hyrimoz), with a WAC 5% less than the reference product.
The unbranded version of adalimumab-adaz, sold at an 81% discount from the reference product, was covered by 13% of plans. Only 4.6% of plans covered high-concentration adalimumab-bwwd (Hadlima), manufactured by Samsung Bioepis.
In January 2024, no high-concentration adalimumab biosimilar had been granted interchangeability status by the US Food and Drug Administration (FDA). Adalimumab-ryvk (Simlandi) was the first biosimilar to receive this designation and was launched in late May 2024.
Coverage for the lower concentration of adalimumab was nearly universal (98.7% of plans). About half of the plans (50.7%) covered adalimumab-adbm (Cyltezo) at a 5% discount. Adalimumab-adbm (Boehringer Ingelheim) was the first interchangeable Humira biosimilar approved by the FDA, but it is only interchangeable with the less popular, lower concentration formulation of adalimumab.
All other biosimilars were covered by less than 5% of Medicare Part D plans, even with some having a WAC 86% below Humira.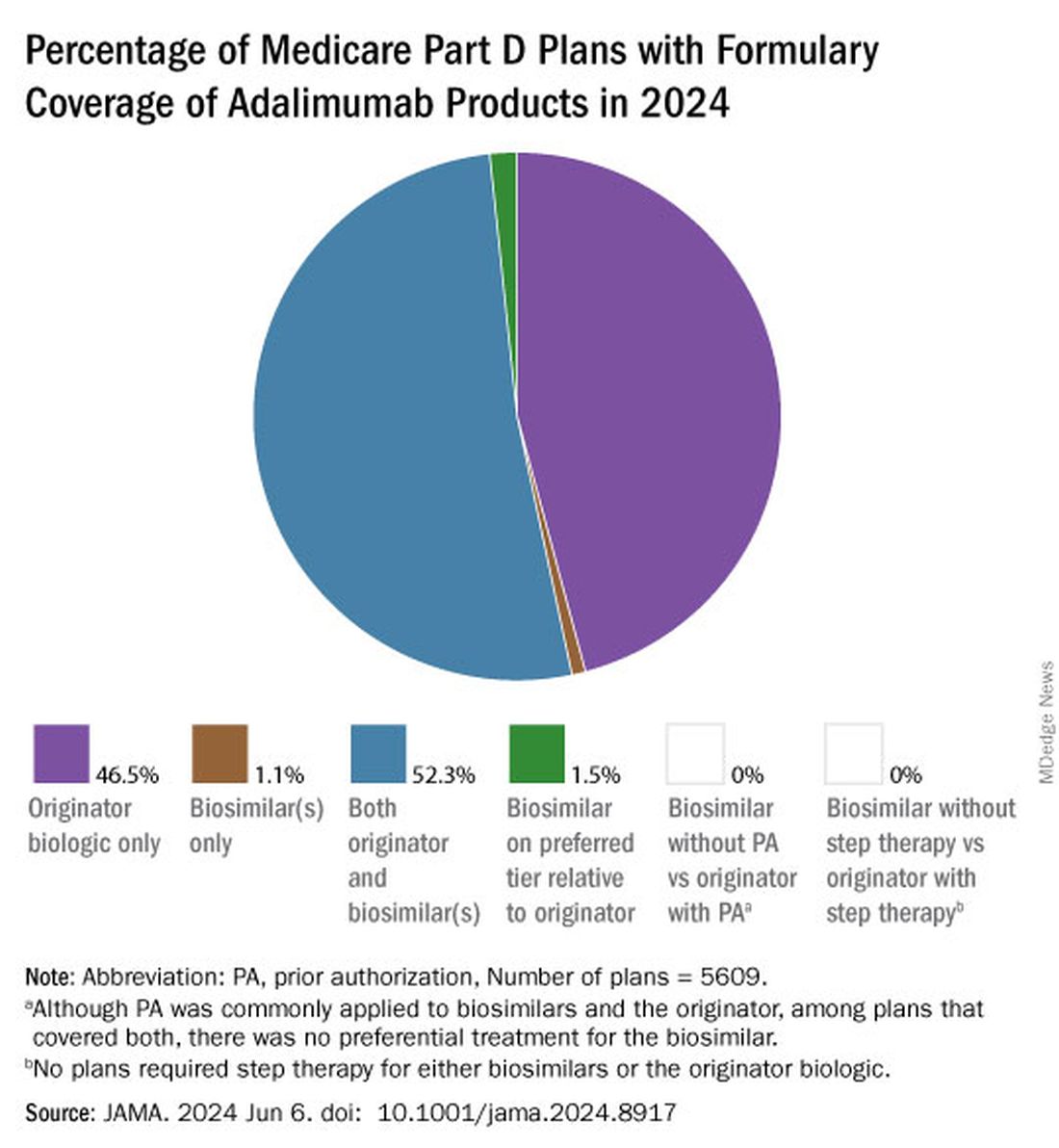
Few plans (1.5%) had biosimilars on preferred tiers compared with the reference product, and no plans used prior authorization to incentivize use of biosimilars. Most plans preferred the higher-priced version of adalimumab biosimilars, which appeals to pharmacy benefit managers who can therefore receive higher rebates, the authors noted.
“Ultimately, biosimilars’ true effect on spending will depend not on their list price but rather on their net price (after rebates) and their influence on originator biologics’ net price,” they wrote. They pointed to the 38% drop in Humira’s annual net price at the end of 2023 compared with the prior year.
“Despite this price decrease, biosimilars offer far greater potential savings: Several adalimumab biosimilars have list prices that are less than half of Humira’s net price,” the authors continued, and encouraged policy makers to mandate coverage for these lower-priced options.
Dr. Klebanoff was supported by a grant from the Health Resources and Services Administration. Two coauthors were supported by a grant from the National Institute on Aging. One author reported receiving consulting fees from AbbVie, which manufactures Humira.
A version of this article appeared on Medscape.com .
Despite the influx of adalimumab biosimilars entering the market in 2023, Humira remains on top.
As of January 2024, both high and low concentrations of Humira, the originator adalimumab product, are nearly universally covered by Medicare Part D plans, while only half of these plans covered adalimumab biosimilars, according to a new research letter published online on June 6, 2024, in JAMA.
Of the plans that covered both, only 1.5% had lower-tier placement for biosimilars.
“This study of formulary coverage helps explain limited uptake of adalimumab biosimilars,” wrote the authors, led by Matthew J. Klebanoff, MD, of the University of Pennsylvania, Philadelphia. “Subpar biosimilar adoption will not only undermine their potential to reduce spending but also may deter investments in biosimilar development.”
The analysis included the formulary and enrollment files for 5609 Medicare Part D plans, representing 44.4 million beneficiaries. Drug list prices and whole acquisition costs (WAC) were pulled from the Red Book database, which provides prices for prescription and over-the-counter drugs as well as medical devices and supplies.
Nearly all (98.9%) of Part D plans covered the high-concentration (100 mg/mL) version of adalimumab with a WAC of $6923. This higher concentration is the most popular formulation of the drug, making up an estimated 85% of prescriptions. By comparison, 26.8% of plans covered the high-concentration version of adalimumab-adaz (Hyrimoz), with a WAC 5% less than the reference product.
The unbranded version of adalimumab-adaz, sold at an 81% discount from the reference product, was covered by 13% of plans. Only 4.6% of plans covered high-concentration adalimumab-bwwd (Hadlima), manufactured by Samsung Bioepis.
In January 2024, no high-concentration adalimumab biosimilar had been granted interchangeability status by the US Food and Drug Administration (FDA). Adalimumab-ryvk (Simlandi) was the first biosimilar to receive this designation and was launched in late May 2024.
Coverage for the lower concentration of adalimumab was nearly universal (98.7% of plans). About half of the plans (50.7%) covered adalimumab-adbm (Cyltezo) at a 5% discount. Adalimumab-adbm (Boehringer Ingelheim) was the first interchangeable Humira biosimilar approved by the FDA, but it is only interchangeable with the less popular, lower concentration formulation of adalimumab.
All other biosimilars were covered by less than 5% of Medicare Part D plans, even with some having a WAC 86% below Humira.
Few plans (1.5%) had biosimilars on preferred tiers compared with the reference product, and no plans used prior authorization to incentivize use of biosimilars. Most plans preferred the higher-priced version of adalimumab biosimilars, which appeals to pharmacy benefit managers who can therefore receive higher rebates, the authors noted.
“Ultimately, biosimilars’ true effect on spending will depend not on their list price but rather on their net price (after rebates) and their influence on originator biologics’ net price,” they wrote. They pointed to the 38% drop in Humira’s annual net price at the end of 2023 compared with the prior year.
“Despite this price decrease, biosimilars offer far greater potential savings: Several adalimumab biosimilars have list prices that are less than half of Humira’s net price,” the authors continued, and encouraged policy makers to mandate coverage for these lower-priced options.
Dr. Klebanoff was supported by a grant from the Health Resources and Services Administration. Two coauthors were supported by a grant from the National Institute on Aging. One author reported receiving consulting fees from AbbVie, which manufactures Humira.
A version of this article appeared on Medscape.com .
Despite the influx of adalimumab biosimilars entering the market in 2023, Humira remains on top.
As of January 2024, both high and low concentrations of Humira, the originator adalimumab product, are nearly universally covered by Medicare Part D plans, while only half of these plans covered adalimumab biosimilars, according to a new research letter published online on June 6, 2024, in JAMA.
Of the plans that covered both, only 1.5% had lower-tier placement for biosimilars.
“This study of formulary coverage helps explain limited uptake of adalimumab biosimilars,” wrote the authors, led by Matthew J. Klebanoff, MD, of the University of Pennsylvania, Philadelphia. “Subpar biosimilar adoption will not only undermine their potential to reduce spending but also may deter investments in biosimilar development.”
The analysis included the formulary and enrollment files for 5609 Medicare Part D plans, representing 44.4 million beneficiaries. Drug list prices and whole acquisition costs (WAC) were pulled from the Red Book database, which provides prices for prescription and over-the-counter drugs as well as medical devices and supplies.
Nearly all (98.9%) of Part D plans covered the high-concentration (100 mg/mL) version of adalimumab with a WAC of $6923. This higher concentration is the most popular formulation of the drug, making up an estimated 85% of prescriptions. By comparison, 26.8% of plans covered the high-concentration version of adalimumab-adaz (Hyrimoz), with a WAC 5% less than the reference product.
The unbranded version of adalimumab-adaz, sold at an 81% discount from the reference product, was covered by 13% of plans. Only 4.6% of plans covered high-concentration adalimumab-bwwd (Hadlima), manufactured by Samsung Bioepis.
In January 2024, no high-concentration adalimumab biosimilar had been granted interchangeability status by the US Food and Drug Administration (FDA). Adalimumab-ryvk (Simlandi) was the first biosimilar to receive this designation and was launched in late May 2024.
Coverage for the lower concentration of adalimumab was nearly universal (98.7% of plans). About half of the plans (50.7%) covered adalimumab-adbm (Cyltezo) at a 5% discount. Adalimumab-adbm (Boehringer Ingelheim) was the first interchangeable Humira biosimilar approved by the FDA, but it is only interchangeable with the less popular, lower concentration formulation of adalimumab.
All other biosimilars were covered by less than 5% of Medicare Part D plans, even with some having a WAC 86% below Humira.
Few plans (1.5%) had biosimilars on preferred tiers compared with the reference product, and no plans used prior authorization to incentivize use of biosimilars. Most plans preferred the higher-priced version of adalimumab biosimilars, which appeals to pharmacy benefit managers who can therefore receive higher rebates, the authors noted.
“Ultimately, biosimilars’ true effect on spending will depend not on their list price but rather on their net price (after rebates) and their influence on originator biologics’ net price,” they wrote. They pointed to the 38% drop in Humira’s annual net price at the end of 2023 compared with the prior year.
“Despite this price decrease, biosimilars offer far greater potential savings: Several adalimumab biosimilars have list prices that are less than half of Humira’s net price,” the authors continued, and encouraged policy makers to mandate coverage for these lower-priced options.
Dr. Klebanoff was supported by a grant from the Health Resources and Services Administration. Two coauthors were supported by a grant from the National Institute on Aging. One author reported receiving consulting fees from AbbVie, which manufactures Humira.
A version of this article appeared on Medscape.com .
FROM JAMA
Would Making Tuition Free Address the Primary Care Shortage?
This transcript has been edited for clarity.
Would free medical school encourage more students to pursue primary care? Overpriced medical training in this country definitely contributes to burnout and the physician shortage, and all that debt may influence what type of specialty somebody goes into.
Assumptions Behind Free Tuition Initiatives
Now, this question is based on an assumption that there already is a large group of medical students who want to go into primary care, and the reason they’re not or the reason they’re reluctant to is because of tuition. If you take that stress away, you fix the problem.
This was at least part of the assumption made when NYU announced free tuition for all medical students back in 2018. One goal was to encourage more students to go into primary care. The other was to broaden the application pool to include more diverse students from different socioeconomic and racial backgrounds. We›ll get back to that.
Quick numbers. In NYU’s 2022 match, the first tuition-free class, about 25% of students matched into a primary care specialty— internal medicine or pediatrics — and there were zero matches into a family medicine residency. I can’t find the data, but anecdotally that 25% is a slight increase from prior years.
Primary Care Match Rates Post–Tuition Waiver
There’s some fine print to consider. Some of the residents who matched into internal medicine or pediatric programs may subspecialize and not work in outpatient medicine at all. Also, a majority of students matched in major urban areas such as New York, Boston, or Los Angeles. We know that historically, people tend to work where they train, so that’s not looking too great for recruitment to rural communities or underserved areas.
There is some hope. In 2024, slightly more students from NYU matched into primary care specialties, including to family medicine. This is amidst a new record in family med residency matches nationwide. Rightfully, in the beginning, NYU was applauded for this tuition-free decision, but there was some criticism about who should be prioritized for financial assistance. Consider a future surgeon from a wealthy family vs a first-generation student committed to rural primary care.
NYU said, “No, equality for all.” Even acclaimed physician and bioethicist, Dr Ezekiel Emanuel weighed in and suggested “forgiving medical school debt for students who commit to a career in primary care in an underserved area. Two years of service for each year of free tuition.” At least this would allow resources to be focused only on building a primary care pool.
Also, after the tuition-free announcement, applications to NYU increased by almost 50%, and from underrepresented groups, 100%. Not that surprising. The average MCAT score and GPA also increased, but the acceptance rate stayed at around 2%.
How much difference will this tuition-free program really make in the future? Time will tell, but I do think that NYU set an important precedent here. Medical schools should be critically looking at where tuition money goes and what financial incentives could be used to attract a more diverse student body, with more hopefully going into primary care.
Let’s take a look at NYU’s Grossman Long Island campus. They have an accelerated 3-year program, tuition-free, primary care focused, and 67% of their graduates went into primary care.
In California, Kaiser Permanente School of Medicine, which is also tuition-free and focused, had 38% of their graduates go to primary care.
What are these programs doing differently? Well, they’re tuition-free, they have focused tracks, and they have enough accredited sites so that students can get a realistic and broad look of what it’s actually like to practice primary care.
Attracting Med Students to Primary Care
This leads to a broader question: How do we create an environment beyond tuition that encourages more students to go into internal medicine, pediatrics, or family medicine?
Right now, across those three specialties, the average salary is $250,000, which is lower than in other subspecialties. There’s a high amount of administrative workload, loss of autonomy, and plenty of burnout. You want to get more students to go into primary care? We need to fix primary care.
That involves many factors. Get ready for this. I actually had to make a list based on what I’ve read in articles and heard from my colleagues.
If we want to attract more students to primary care, we need to talk about:
- Improving reimbursement;
- Better mental health support;
- Highlighting the importance primary care plays in public health;
- Expanding care teams;
- Creating more medical students and training sites in rural and underserved areas;
- Expanding the use of telehealth services;
- Creating early exposure programs for high school and college students; and
- Paying attention to how local policies and statistics, such as crime, housing, and abortion bans, may push people away from practicing in certain areas or states.
Clearly, this is a large number of considerations that goes far beyond the altruistic tuition-free gifts.
Look, it’s no surprise we have a physician shortage that affects multiple specialties, but it is alarming that by 2034, there’s going to be an estimated shortage of 50,000 primary care doctors.
Stay Tuned
What do you all think? Is free tuition enough to actually move the needle in the long term, or should NYU have made a more focused gift? Comment below.
I know what you’re all wondering why I didn’t talk about tuition waivers in terms of diversifying the student body. That’s because that deserves its own video. Stay tuned for part 2.
Dr. Patel is a pediatric hospitalist, television producer, media contributor, and digital health enthusiast. He splits his time between New York City and San Francisco, as he is on faculty at Columbia University/Morgan Stanley Children’s Hospital and UCSF Benioff Children’s Hospital. He hosts The Hospitalist Retort video blog on Medscape.
Dr. Patel has disclosed the following relevant financial relationships: serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for Medumo Inc.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Would free medical school encourage more students to pursue primary care? Overpriced medical training in this country definitely contributes to burnout and the physician shortage, and all that debt may influence what type of specialty somebody goes into.
Assumptions Behind Free Tuition Initiatives
Now, this question is based on an assumption that there already is a large group of medical students who want to go into primary care, and the reason they’re not or the reason they’re reluctant to is because of tuition. If you take that stress away, you fix the problem.
This was at least part of the assumption made when NYU announced free tuition for all medical students back in 2018. One goal was to encourage more students to go into primary care. The other was to broaden the application pool to include more diverse students from different socioeconomic and racial backgrounds. We›ll get back to that.
Quick numbers. In NYU’s 2022 match, the first tuition-free class, about 25% of students matched into a primary care specialty— internal medicine or pediatrics — and there were zero matches into a family medicine residency. I can’t find the data, but anecdotally that 25% is a slight increase from prior years.
Primary Care Match Rates Post–Tuition Waiver
There’s some fine print to consider. Some of the residents who matched into internal medicine or pediatric programs may subspecialize and not work in outpatient medicine at all. Also, a majority of students matched in major urban areas such as New York, Boston, or Los Angeles. We know that historically, people tend to work where they train, so that’s not looking too great for recruitment to rural communities or underserved areas.
There is some hope. In 2024, slightly more students from NYU matched into primary care specialties, including to family medicine. This is amidst a new record in family med residency matches nationwide. Rightfully, in the beginning, NYU was applauded for this tuition-free decision, but there was some criticism about who should be prioritized for financial assistance. Consider a future surgeon from a wealthy family vs a first-generation student committed to rural primary care.
NYU said, “No, equality for all.” Even acclaimed physician and bioethicist, Dr Ezekiel Emanuel weighed in and suggested “forgiving medical school debt for students who commit to a career in primary care in an underserved area. Two years of service for each year of free tuition.” At least this would allow resources to be focused only on building a primary care pool.
Also, after the tuition-free announcement, applications to NYU increased by almost 50%, and from underrepresented groups, 100%. Not that surprising. The average MCAT score and GPA also increased, but the acceptance rate stayed at around 2%.
How much difference will this tuition-free program really make in the future? Time will tell, but I do think that NYU set an important precedent here. Medical schools should be critically looking at where tuition money goes and what financial incentives could be used to attract a more diverse student body, with more hopefully going into primary care.
Let’s take a look at NYU’s Grossman Long Island campus. They have an accelerated 3-year program, tuition-free, primary care focused, and 67% of their graduates went into primary care.
In California, Kaiser Permanente School of Medicine, which is also tuition-free and focused, had 38% of their graduates go to primary care.
What are these programs doing differently? Well, they’re tuition-free, they have focused tracks, and they have enough accredited sites so that students can get a realistic and broad look of what it’s actually like to practice primary care.
Attracting Med Students to Primary Care
This leads to a broader question: How do we create an environment beyond tuition that encourages more students to go into internal medicine, pediatrics, or family medicine?
Right now, across those three specialties, the average salary is $250,000, which is lower than in other subspecialties. There’s a high amount of administrative workload, loss of autonomy, and plenty of burnout. You want to get more students to go into primary care? We need to fix primary care.
That involves many factors. Get ready for this. I actually had to make a list based on what I’ve read in articles and heard from my colleagues.
If we want to attract more students to primary care, we need to talk about:
- Improving reimbursement;
- Better mental health support;
- Highlighting the importance primary care plays in public health;
- Expanding care teams;
- Creating more medical students and training sites in rural and underserved areas;
- Expanding the use of telehealth services;
- Creating early exposure programs for high school and college students; and
- Paying attention to how local policies and statistics, such as crime, housing, and abortion bans, may push people away from practicing in certain areas or states.
Clearly, this is a large number of considerations that goes far beyond the altruistic tuition-free gifts.
Look, it’s no surprise we have a physician shortage that affects multiple specialties, but it is alarming that by 2034, there’s going to be an estimated shortage of 50,000 primary care doctors.
Stay Tuned
What do you all think? Is free tuition enough to actually move the needle in the long term, or should NYU have made a more focused gift? Comment below.
I know what you’re all wondering why I didn’t talk about tuition waivers in terms of diversifying the student body. That’s because that deserves its own video. Stay tuned for part 2.
Dr. Patel is a pediatric hospitalist, television producer, media contributor, and digital health enthusiast. He splits his time between New York City and San Francisco, as he is on faculty at Columbia University/Morgan Stanley Children’s Hospital and UCSF Benioff Children’s Hospital. He hosts The Hospitalist Retort video blog on Medscape.
Dr. Patel has disclosed the following relevant financial relationships: serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for Medumo Inc.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Would free medical school encourage more students to pursue primary care? Overpriced medical training in this country definitely contributes to burnout and the physician shortage, and all that debt may influence what type of specialty somebody goes into.
Assumptions Behind Free Tuition Initiatives
Now, this question is based on an assumption that there already is a large group of medical students who want to go into primary care, and the reason they’re not or the reason they’re reluctant to is because of tuition. If you take that stress away, you fix the problem.
This was at least part of the assumption made when NYU announced free tuition for all medical students back in 2018. One goal was to encourage more students to go into primary care. The other was to broaden the application pool to include more diverse students from different socioeconomic and racial backgrounds. We›ll get back to that.
Quick numbers. In NYU’s 2022 match, the first tuition-free class, about 25% of students matched into a primary care specialty— internal medicine or pediatrics — and there were zero matches into a family medicine residency. I can’t find the data, but anecdotally that 25% is a slight increase from prior years.
Primary Care Match Rates Post–Tuition Waiver
There’s some fine print to consider. Some of the residents who matched into internal medicine or pediatric programs may subspecialize and not work in outpatient medicine at all. Also, a majority of students matched in major urban areas such as New York, Boston, or Los Angeles. We know that historically, people tend to work where they train, so that’s not looking too great for recruitment to rural communities or underserved areas.
There is some hope. In 2024, slightly more students from NYU matched into primary care specialties, including to family medicine. This is amidst a new record in family med residency matches nationwide. Rightfully, in the beginning, NYU was applauded for this tuition-free decision, but there was some criticism about who should be prioritized for financial assistance. Consider a future surgeon from a wealthy family vs a first-generation student committed to rural primary care.
NYU said, “No, equality for all.” Even acclaimed physician and bioethicist, Dr Ezekiel Emanuel weighed in and suggested “forgiving medical school debt for students who commit to a career in primary care in an underserved area. Two years of service for each year of free tuition.” At least this would allow resources to be focused only on building a primary care pool.
Also, after the tuition-free announcement, applications to NYU increased by almost 50%, and from underrepresented groups, 100%. Not that surprising. The average MCAT score and GPA also increased, but the acceptance rate stayed at around 2%.
How much difference will this tuition-free program really make in the future? Time will tell, but I do think that NYU set an important precedent here. Medical schools should be critically looking at where tuition money goes and what financial incentives could be used to attract a more diverse student body, with more hopefully going into primary care.
Let’s take a look at NYU’s Grossman Long Island campus. They have an accelerated 3-year program, tuition-free, primary care focused, and 67% of their graduates went into primary care.
In California, Kaiser Permanente School of Medicine, which is also tuition-free and focused, had 38% of their graduates go to primary care.
What are these programs doing differently? Well, they’re tuition-free, they have focused tracks, and they have enough accredited sites so that students can get a realistic and broad look of what it’s actually like to practice primary care.
Attracting Med Students to Primary Care
This leads to a broader question: How do we create an environment beyond tuition that encourages more students to go into internal medicine, pediatrics, or family medicine?
Right now, across those three specialties, the average salary is $250,000, which is lower than in other subspecialties. There’s a high amount of administrative workload, loss of autonomy, and plenty of burnout. You want to get more students to go into primary care? We need to fix primary care.
That involves many factors. Get ready for this. I actually had to make a list based on what I’ve read in articles and heard from my colleagues.
If we want to attract more students to primary care, we need to talk about:
- Improving reimbursement;
- Better mental health support;
- Highlighting the importance primary care plays in public health;
- Expanding care teams;
- Creating more medical students and training sites in rural and underserved areas;
- Expanding the use of telehealth services;
- Creating early exposure programs for high school and college students; and
- Paying attention to how local policies and statistics, such as crime, housing, and abortion bans, may push people away from practicing in certain areas or states.
Clearly, this is a large number of considerations that goes far beyond the altruistic tuition-free gifts.
Look, it’s no surprise we have a physician shortage that affects multiple specialties, but it is alarming that by 2034, there’s going to be an estimated shortage of 50,000 primary care doctors.
Stay Tuned
What do you all think? Is free tuition enough to actually move the needle in the long term, or should NYU have made a more focused gift? Comment below.
I know what you’re all wondering why I didn’t talk about tuition waivers in terms of diversifying the student body. That’s because that deserves its own video. Stay tuned for part 2.
Dr. Patel is a pediatric hospitalist, television producer, media contributor, and digital health enthusiast. He splits his time between New York City and San Francisco, as he is on faculty at Columbia University/Morgan Stanley Children’s Hospital and UCSF Benioff Children’s Hospital. He hosts The Hospitalist Retort video blog on Medscape.
Dr. Patel has disclosed the following relevant financial relationships: serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for Medumo Inc.
A version of this article appeared on Medscape.com.




