User login
Widespread carboplatin, cisplatin shortages: NCCN survey
The survey, which included responses from 27 NCCN member institutions, revealed that 93% are experiencing a shortage of carboplatin and that 70% have reported a shortage of cisplatin.
“This is an unacceptable situation,” Robert W. Carlson, MD, NCCN’s chief executive offer, said in the statement released by the network.
“We are hearing from oncologists and pharmacists across the country who have to scramble to find appropriate alternatives for treating their patients with cancer right now,” Dr. Carlson said. And while the survey results show patients are still able to get lifesaving care, “it comes at a burden to our overtaxed medical facilities.”
The NCCN called on the federal government, the pharmaceutical industry, providers, and payers to take steps to “help mitigate any impacts” from this cancer drug shortage.
“We need to work together to improve the current situation and prevent it from happening again in the future,” Dr. Carlson stressed.
Carboplatin and cisplatin, which are frequently used together for systemic treatment, are highly effective therapies prescribed to treat many cancer types, including lung, breast, and prostate cancers, as well as leukemias and lymphomas. An estimated 500,000 new patients with cancer receive these agents each year.
The current survey, conducted over the last week of May, found that 100% of responding centers are able to continue to treat patients who need cisplatin without delays.
The same cannot be said for carboplatin: only 64% of centers said they are still able to continue treating all current patients receiving the platinum-based therapy. Among 19 responding centers, 20% reported that they were continuing carboplatin regimens for some but not all patients. And 16% reported treatment delays from having to obtain prior authorization for modified treatment plans, though none reported denials.
“Carboplatin has been in short supply for months but in the last 4 weeks has reached a critical stage,” according to one survey comment. “Without additional inventory many of our sites will be out of drug by early next week.”
In response to the survey question, “Is your center experiencing a shortage of carboplatin,” others made similar comments:
- “Current shipments from established manufacturers have been paused.”
- “The supply of carboplatin available is not meeting our demands.”
- “Without additional supply in early June, we will have to implement several shortage mitigation strategies.”
Survey respondents also addressed whether manufacturers or suppliers have provided any indication of when these drugs will become readily available again. For both drugs, about 60% of respondents said no. And for those who do receive updates, many noted that the “information is tentative and variable.”
Respondents indicated that other cancer agents, including methotrexate (67%) and 5FU (26%), are also in short supply at their centers.
The shortage and the uncertainty as to when it will end are forcing some centers to develop conservation and mitigation strategies.
The NCCN has broadly outlined how the federal government, the pharmaceutical industry, providers, and payers can help with prevention and mitigation. The NCCN has called on the federal government and the pharmaceutical industry to work to secure a steady supply of core anticancer drugs and has asked payers to “put patients first and provide flexible and efficient systems of providing coverage for alternative therapies replacing anti-cancer drugs that are unavailable or in shortage.”
Overall, the survey results “demonstrate the widespread impact of the chemotherapy shortage,” said Alyssa Schatz, MSW, senior director of policy and advocacy for NCCN. “We hope that by sharing this survey and calling for united action across the oncology community, we can come together to prevent future drug shortages and ensure quality, effective, equitable, and accessible cancer care for all.”
A version of this article first appeared on Medscape.com.
The survey, which included responses from 27 NCCN member institutions, revealed that 93% are experiencing a shortage of carboplatin and that 70% have reported a shortage of cisplatin.
“This is an unacceptable situation,” Robert W. Carlson, MD, NCCN’s chief executive offer, said in the statement released by the network.
“We are hearing from oncologists and pharmacists across the country who have to scramble to find appropriate alternatives for treating their patients with cancer right now,” Dr. Carlson said. And while the survey results show patients are still able to get lifesaving care, “it comes at a burden to our overtaxed medical facilities.”
The NCCN called on the federal government, the pharmaceutical industry, providers, and payers to take steps to “help mitigate any impacts” from this cancer drug shortage.
“We need to work together to improve the current situation and prevent it from happening again in the future,” Dr. Carlson stressed.
Carboplatin and cisplatin, which are frequently used together for systemic treatment, are highly effective therapies prescribed to treat many cancer types, including lung, breast, and prostate cancers, as well as leukemias and lymphomas. An estimated 500,000 new patients with cancer receive these agents each year.
The current survey, conducted over the last week of May, found that 100% of responding centers are able to continue to treat patients who need cisplatin without delays.
The same cannot be said for carboplatin: only 64% of centers said they are still able to continue treating all current patients receiving the platinum-based therapy. Among 19 responding centers, 20% reported that they were continuing carboplatin regimens for some but not all patients. And 16% reported treatment delays from having to obtain prior authorization for modified treatment plans, though none reported denials.
“Carboplatin has been in short supply for months but in the last 4 weeks has reached a critical stage,” according to one survey comment. “Without additional inventory many of our sites will be out of drug by early next week.”
In response to the survey question, “Is your center experiencing a shortage of carboplatin,” others made similar comments:
- “Current shipments from established manufacturers have been paused.”
- “The supply of carboplatin available is not meeting our demands.”
- “Without additional supply in early June, we will have to implement several shortage mitigation strategies.”
Survey respondents also addressed whether manufacturers or suppliers have provided any indication of when these drugs will become readily available again. For both drugs, about 60% of respondents said no. And for those who do receive updates, many noted that the “information is tentative and variable.”
Respondents indicated that other cancer agents, including methotrexate (67%) and 5FU (26%), are also in short supply at their centers.
The shortage and the uncertainty as to when it will end are forcing some centers to develop conservation and mitigation strategies.
The NCCN has broadly outlined how the federal government, the pharmaceutical industry, providers, and payers can help with prevention and mitigation. The NCCN has called on the federal government and the pharmaceutical industry to work to secure a steady supply of core anticancer drugs and has asked payers to “put patients first and provide flexible and efficient systems of providing coverage for alternative therapies replacing anti-cancer drugs that are unavailable or in shortage.”
Overall, the survey results “demonstrate the widespread impact of the chemotherapy shortage,” said Alyssa Schatz, MSW, senior director of policy and advocacy for NCCN. “We hope that by sharing this survey and calling for united action across the oncology community, we can come together to prevent future drug shortages and ensure quality, effective, equitable, and accessible cancer care for all.”
A version of this article first appeared on Medscape.com.
The survey, which included responses from 27 NCCN member institutions, revealed that 93% are experiencing a shortage of carboplatin and that 70% have reported a shortage of cisplatin.
“This is an unacceptable situation,” Robert W. Carlson, MD, NCCN’s chief executive offer, said in the statement released by the network.
“We are hearing from oncologists and pharmacists across the country who have to scramble to find appropriate alternatives for treating their patients with cancer right now,” Dr. Carlson said. And while the survey results show patients are still able to get lifesaving care, “it comes at a burden to our overtaxed medical facilities.”
The NCCN called on the federal government, the pharmaceutical industry, providers, and payers to take steps to “help mitigate any impacts” from this cancer drug shortage.
“We need to work together to improve the current situation and prevent it from happening again in the future,” Dr. Carlson stressed.
Carboplatin and cisplatin, which are frequently used together for systemic treatment, are highly effective therapies prescribed to treat many cancer types, including lung, breast, and prostate cancers, as well as leukemias and lymphomas. An estimated 500,000 new patients with cancer receive these agents each year.
The current survey, conducted over the last week of May, found that 100% of responding centers are able to continue to treat patients who need cisplatin without delays.
The same cannot be said for carboplatin: only 64% of centers said they are still able to continue treating all current patients receiving the platinum-based therapy. Among 19 responding centers, 20% reported that they were continuing carboplatin regimens for some but not all patients. And 16% reported treatment delays from having to obtain prior authorization for modified treatment plans, though none reported denials.
“Carboplatin has been in short supply for months but in the last 4 weeks has reached a critical stage,” according to one survey comment. “Without additional inventory many of our sites will be out of drug by early next week.”
In response to the survey question, “Is your center experiencing a shortage of carboplatin,” others made similar comments:
- “Current shipments from established manufacturers have been paused.”
- “The supply of carboplatin available is not meeting our demands.”
- “Without additional supply in early June, we will have to implement several shortage mitigation strategies.”
Survey respondents also addressed whether manufacturers or suppliers have provided any indication of when these drugs will become readily available again. For both drugs, about 60% of respondents said no. And for those who do receive updates, many noted that the “information is tentative and variable.”
Respondents indicated that other cancer agents, including methotrexate (67%) and 5FU (26%), are also in short supply at their centers.
The shortage and the uncertainty as to when it will end are forcing some centers to develop conservation and mitigation strategies.
The NCCN has broadly outlined how the federal government, the pharmaceutical industry, providers, and payers can help with prevention and mitigation. The NCCN has called on the federal government and the pharmaceutical industry to work to secure a steady supply of core anticancer drugs and has asked payers to “put patients first and provide flexible and efficient systems of providing coverage for alternative therapies replacing anti-cancer drugs that are unavailable or in shortage.”
Overall, the survey results “demonstrate the widespread impact of the chemotherapy shortage,” said Alyssa Schatz, MSW, senior director of policy and advocacy for NCCN. “We hope that by sharing this survey and calling for united action across the oncology community, we can come together to prevent future drug shortages and ensure quality, effective, equitable, and accessible cancer care for all.”
A version of this article first appeared on Medscape.com.
Emerging Treatment Options for Mantle Cell Lymphoma
Mantle cell lymphoma (MCL) is a rare, B-cell non-Hodgkin lymphoma whose biological heterogeneity has long challenged researchers and clinicians. There are no firmly-established therapies, and many individuals experience relapse even after successful treatment. There is a clear unmet need in MCL in the relapsed setting. In recent years, researchers have worked to address this need, demonstrating efficacy with covalent Bruton tyrosine kinase (BTK) inhibitors, led by ibrutinib, and anti-CD19 chimeric antigen receptor T-cell therapy. While these are helpful additions, relapse remains a challenge.
Fortunately, progress continues. Owing to encouraging results in recent trials, individuals with relapsed/refractory MCL are now experiencing clinical benefit from the noncovalent BTK inhibitor pirtrobrutinib. Investigational bispecific antibody (bsAb) therapy awaits in the wings.
Similarly, both younger and older patients with treatment naïve MCL could soon see improvement from the addition of BTK inhibitors to each age group’s standard treatment option. The following is a description of recent developments and their potential implications for practice.
One of the most exciting developments is the US Food and Drug Administration’s accelerated approval of pirtrobrutinib. A noncovalent BTK inhibitor, pirtrobrutinib has been found to have activity in individuals with MCL who have failed on multiple therapies, including standard BTK inhibitors. Pirtrobrutinib targets certain mutations in the BTK protein that are associated with resistance to covalent BTK inhibitors. In addition to resistance, some patients discontinue treatment with non-reversible BTK inhibitors because of intolerable toxicity.
Approval of pirtrobrutinib was based on an evaluation involving 120 individuals (median age 71) who were previously treated with a non-reversible BTK inhibitor. Two-thirds were previously treated with ibrutinib; 30% with acalabrutinib, and 8% zanubrutinib (some received more than one BTK inhibitor previously). The vast majority (83%) discontinued treatment due to refractory or progressive disease; 10% stopped due to toxicity; and the remainder halted treatment for other reasons.
Six in every 10 of the participants were classified on the MCL International Prognostic Index as intermediate; one-fourth were classified as high; and the remainder low. Patients received 200 mg of pirtrobrutinib once a day until disease either progressed or they experienced intolerable toxicity. Among the results:
Overall response rate was 50%; 13% responded completely
Median duration of response was 8.3 months
Duration of response rate at 6 months was 65%
Grade 3 or 4 abnormalities experienced by 10% or more of participants included decreased neutrophil counts, lymphocyte counts, and platelet counts
Further, bsAb therapy targeting CD20-CD3 is not yet approved but is showing promise as a potential therapy following BTK inhibitor failure. The treatment consists of an antibody containing two prongs. One is a CD20 protein that attaches to the lymphoma cell. The other is an anti-CD3 antibody that attaches to the T cell to bring the patient’s own T cells closer to the lymphoma to increase the cell kill.
Preliminary studies evaluating bsAbs in individuals with MCL, many of whom have failed on multiple other types of therapies, show a remarkably high response rate. In one such investigation, the bsAb glofitamab was given to 21 individuals as monotherapy following pretreatment with obinutuzumab. The regimen produced an overall response rate of 81% (n = 17) and a complete response rate of 68% (n = 14). Response was similar in participants who had and had not received prior BTK therapy. Among those who achieved a complete response, median duration was 2.4 months, and 12 of those who reached a complete response were still in remission at the study’s data cutoff point.
For younger individuals with treatment-naïve MCL, the current standard is chemotherapy and autologous stem-cell transplant (ASCT). For older individuals the standard is chemoimmunotherapy. The replacement or addition of the BTK inhibitor ibrutinib to these regimens is showing the promise of added clinical benefit in both age contingents.
Investigators presented results of the three-arm TRIANGLE trial at the 64th ASH Annual Meeting in December 2022. The study compared 1) chemotherapy followed by ASCT; 2) ibrutinib plus chemotherapy followed by ASCT and ibrutinib maintenance; and 3) ibrutinib plus chemotherapy followed by ibrutinib maintenance. Participants (n = 870) ≤ 65 years of age (median age 57) with previously untreated advanced-stage MCL were randomized to 1 of the 3 regimens. Investigators looked at overall response, complete response, and failure-free survival rates (FFS). Among the results:
Overall response rates were 98% in the 2 groups whose treatments included ibrutinib, versus 94% in the chemotherapy followed by ASCT group.
Complete response rates were 45% and 36%, respectively.
The non-ibrutinib regimen did not attain FFS superiority over ibrutinib plus chemotherapy, with a 3-year FFS rate of 72% and 86%, respectively (p=0.9979, hazard ratio [HR]: 1.77).
Ibrutinib plus chemotherapy was shown to be superior to chemotherapy/ASCT, with a 3-year FFS rate of 88% and 72%, respectively (p=0.0008, HR: 0.52).
The only adverse event differences of note occurred during maintenance treatment; there were significantly more grade 3-5 adverse events in the ibrutinib/chemotherapy/ASCT group, compared with the other 2 contingents.
Researchers noted in materials accompanying their presentation that, “It has been clearly demonstrated that the current standard high-dose regimen is not superior to the new ibrutinib-containing regimen without ASCT. More follow-up is needed to clarify the role of ASCT in the context of ibrutinib-containing treatment. However, the current results already support the use of ibrutinib in the first-line treatment of younger MCL patients.”
It also appears that ibrutinib added to standard chemoimmunotherapy can improve outcomes in older individuals with treatment-naïve MCL. In 2022, researchers published results from the international, randomized, double-blind, phase 3 SHINE trial. Participants (n = 523) were ≥ 65 years of age with previously untreated MCL and were randomized to receive either ibrutinib 560 mg daily or placebo added to chemoimmunotherapy consisting of bendamustine and rituximab every 4 weeks for 6 cycles. Individuals with a partial or complete response continued treatment every 8 weeks for up to 12 more doses. Investigators looked primarily at progression-free survival (PFS), as well as complete response, undetectable minimal residual disease, and time to worsening. Among the results:
116 participants (44%) in the ibrutinib group experienced disease progression or died, compared with 152 (58%) in the placebo contingent.
Median PFS was 80.6 months and 52.9 months, respectively.
PFS benefit was seen across most subgroups (patients categorized as high risk, and those with TP53 mutations did not benefit).
Complete response was seen in 66% and 58% of participants, respectively.
Undetectable minimal residual disease was observed in 62% and 57%, respectively.
Deaths attributed to disease progression or adverse events occurred in 22% and 28%, respectively.
Grade 3 or 4 adverse event incident rates were 82% and 77%, respectively.
Researchers noted that, “Given the shorter progression-free survival with current standard-care chemoimmunotherapy options, a prolongation of progression-free survival in response to primary therapy may provide patients with an improved opportunity for durable disease control in order to prevent or delay relapse.”
Data on the use of other BTK inhibitors as first-line treatment for MCL are forthcoming, including:
ECHO, a phase 3 trial assessing the efficacy of acalabrutinib versus placebo added to bendamustine and rituximab.
MANGROVE, a phase 3 study comparing zanubrutinib plus rituximab versus bendamustine plus rituximab.
ENRICH, a phase 2 study evaluating a chemotherapy-free option–ibrutinib and rituximab in older individuals.
OASIS, a randomized, phase 2 trial comparing ibrutinib/anti-CD20 antibodies (Ab) and Ibrutinib/anti-CD20 Ab/venetoclax given as fixed duration combinations.
The evolution of BTK inhibitors for relapsed MCL has great potential; further benefits continue to be explored.
Mantle cell lymphoma (MCL) is a rare, B-cell non-Hodgkin lymphoma whose biological heterogeneity has long challenged researchers and clinicians. There are no firmly-established therapies, and many individuals experience relapse even after successful treatment. There is a clear unmet need in MCL in the relapsed setting. In recent years, researchers have worked to address this need, demonstrating efficacy with covalent Bruton tyrosine kinase (BTK) inhibitors, led by ibrutinib, and anti-CD19 chimeric antigen receptor T-cell therapy. While these are helpful additions, relapse remains a challenge.
Fortunately, progress continues. Owing to encouraging results in recent trials, individuals with relapsed/refractory MCL are now experiencing clinical benefit from the noncovalent BTK inhibitor pirtrobrutinib. Investigational bispecific antibody (bsAb) therapy awaits in the wings.
Similarly, both younger and older patients with treatment naïve MCL could soon see improvement from the addition of BTK inhibitors to each age group’s standard treatment option. The following is a description of recent developments and their potential implications for practice.
One of the most exciting developments is the US Food and Drug Administration’s accelerated approval of pirtrobrutinib. A noncovalent BTK inhibitor, pirtrobrutinib has been found to have activity in individuals with MCL who have failed on multiple therapies, including standard BTK inhibitors. Pirtrobrutinib targets certain mutations in the BTK protein that are associated with resistance to covalent BTK inhibitors. In addition to resistance, some patients discontinue treatment with non-reversible BTK inhibitors because of intolerable toxicity.
Approval of pirtrobrutinib was based on an evaluation involving 120 individuals (median age 71) who were previously treated with a non-reversible BTK inhibitor. Two-thirds were previously treated with ibrutinib; 30% with acalabrutinib, and 8% zanubrutinib (some received more than one BTK inhibitor previously). The vast majority (83%) discontinued treatment due to refractory or progressive disease; 10% stopped due to toxicity; and the remainder halted treatment for other reasons.
Six in every 10 of the participants were classified on the MCL International Prognostic Index as intermediate; one-fourth were classified as high; and the remainder low. Patients received 200 mg of pirtrobrutinib once a day until disease either progressed or they experienced intolerable toxicity. Among the results:
Overall response rate was 50%; 13% responded completely
Median duration of response was 8.3 months
Duration of response rate at 6 months was 65%
Grade 3 or 4 abnormalities experienced by 10% or more of participants included decreased neutrophil counts, lymphocyte counts, and platelet counts
Further, bsAb therapy targeting CD20-CD3 is not yet approved but is showing promise as a potential therapy following BTK inhibitor failure. The treatment consists of an antibody containing two prongs. One is a CD20 protein that attaches to the lymphoma cell. The other is an anti-CD3 antibody that attaches to the T cell to bring the patient’s own T cells closer to the lymphoma to increase the cell kill.
Preliminary studies evaluating bsAbs in individuals with MCL, many of whom have failed on multiple other types of therapies, show a remarkably high response rate. In one such investigation, the bsAb glofitamab was given to 21 individuals as monotherapy following pretreatment with obinutuzumab. The regimen produced an overall response rate of 81% (n = 17) and a complete response rate of 68% (n = 14). Response was similar in participants who had and had not received prior BTK therapy. Among those who achieved a complete response, median duration was 2.4 months, and 12 of those who reached a complete response were still in remission at the study’s data cutoff point.
For younger individuals with treatment-naïve MCL, the current standard is chemotherapy and autologous stem-cell transplant (ASCT). For older individuals the standard is chemoimmunotherapy. The replacement or addition of the BTK inhibitor ibrutinib to these regimens is showing the promise of added clinical benefit in both age contingents.
Investigators presented results of the three-arm TRIANGLE trial at the 64th ASH Annual Meeting in December 2022. The study compared 1) chemotherapy followed by ASCT; 2) ibrutinib plus chemotherapy followed by ASCT and ibrutinib maintenance; and 3) ibrutinib plus chemotherapy followed by ibrutinib maintenance. Participants (n = 870) ≤ 65 years of age (median age 57) with previously untreated advanced-stage MCL were randomized to 1 of the 3 regimens. Investigators looked at overall response, complete response, and failure-free survival rates (FFS). Among the results:
Overall response rates were 98% in the 2 groups whose treatments included ibrutinib, versus 94% in the chemotherapy followed by ASCT group.
Complete response rates were 45% and 36%, respectively.
The non-ibrutinib regimen did not attain FFS superiority over ibrutinib plus chemotherapy, with a 3-year FFS rate of 72% and 86%, respectively (p=0.9979, hazard ratio [HR]: 1.77).
Ibrutinib plus chemotherapy was shown to be superior to chemotherapy/ASCT, with a 3-year FFS rate of 88% and 72%, respectively (p=0.0008, HR: 0.52).
The only adverse event differences of note occurred during maintenance treatment; there were significantly more grade 3-5 adverse events in the ibrutinib/chemotherapy/ASCT group, compared with the other 2 contingents.
Researchers noted in materials accompanying their presentation that, “It has been clearly demonstrated that the current standard high-dose regimen is not superior to the new ibrutinib-containing regimen without ASCT. More follow-up is needed to clarify the role of ASCT in the context of ibrutinib-containing treatment. However, the current results already support the use of ibrutinib in the first-line treatment of younger MCL patients.”
It also appears that ibrutinib added to standard chemoimmunotherapy can improve outcomes in older individuals with treatment-naïve MCL. In 2022, researchers published results from the international, randomized, double-blind, phase 3 SHINE trial. Participants (n = 523) were ≥ 65 years of age with previously untreated MCL and were randomized to receive either ibrutinib 560 mg daily or placebo added to chemoimmunotherapy consisting of bendamustine and rituximab every 4 weeks for 6 cycles. Individuals with a partial or complete response continued treatment every 8 weeks for up to 12 more doses. Investigators looked primarily at progression-free survival (PFS), as well as complete response, undetectable minimal residual disease, and time to worsening. Among the results:
116 participants (44%) in the ibrutinib group experienced disease progression or died, compared with 152 (58%) in the placebo contingent.
Median PFS was 80.6 months and 52.9 months, respectively.
PFS benefit was seen across most subgroups (patients categorized as high risk, and those with TP53 mutations did not benefit).
Complete response was seen in 66% and 58% of participants, respectively.
Undetectable minimal residual disease was observed in 62% and 57%, respectively.
Deaths attributed to disease progression or adverse events occurred in 22% and 28%, respectively.
Grade 3 or 4 adverse event incident rates were 82% and 77%, respectively.
Researchers noted that, “Given the shorter progression-free survival with current standard-care chemoimmunotherapy options, a prolongation of progression-free survival in response to primary therapy may provide patients with an improved opportunity for durable disease control in order to prevent or delay relapse.”
Data on the use of other BTK inhibitors as first-line treatment for MCL are forthcoming, including:
ECHO, a phase 3 trial assessing the efficacy of acalabrutinib versus placebo added to bendamustine and rituximab.
MANGROVE, a phase 3 study comparing zanubrutinib plus rituximab versus bendamustine plus rituximab.
ENRICH, a phase 2 study evaluating a chemotherapy-free option–ibrutinib and rituximab in older individuals.
OASIS, a randomized, phase 2 trial comparing ibrutinib/anti-CD20 antibodies (Ab) and Ibrutinib/anti-CD20 Ab/venetoclax given as fixed duration combinations.
The evolution of BTK inhibitors for relapsed MCL has great potential; further benefits continue to be explored.
Mantle cell lymphoma (MCL) is a rare, B-cell non-Hodgkin lymphoma whose biological heterogeneity has long challenged researchers and clinicians. There are no firmly-established therapies, and many individuals experience relapse even after successful treatment. There is a clear unmet need in MCL in the relapsed setting. In recent years, researchers have worked to address this need, demonstrating efficacy with covalent Bruton tyrosine kinase (BTK) inhibitors, led by ibrutinib, and anti-CD19 chimeric antigen receptor T-cell therapy. While these are helpful additions, relapse remains a challenge.
Fortunately, progress continues. Owing to encouraging results in recent trials, individuals with relapsed/refractory MCL are now experiencing clinical benefit from the noncovalent BTK inhibitor pirtrobrutinib. Investigational bispecific antibody (bsAb) therapy awaits in the wings.
Similarly, both younger and older patients with treatment naïve MCL could soon see improvement from the addition of BTK inhibitors to each age group’s standard treatment option. The following is a description of recent developments and their potential implications for practice.
One of the most exciting developments is the US Food and Drug Administration’s accelerated approval of pirtrobrutinib. A noncovalent BTK inhibitor, pirtrobrutinib has been found to have activity in individuals with MCL who have failed on multiple therapies, including standard BTK inhibitors. Pirtrobrutinib targets certain mutations in the BTK protein that are associated with resistance to covalent BTK inhibitors. In addition to resistance, some patients discontinue treatment with non-reversible BTK inhibitors because of intolerable toxicity.
Approval of pirtrobrutinib was based on an evaluation involving 120 individuals (median age 71) who were previously treated with a non-reversible BTK inhibitor. Two-thirds were previously treated with ibrutinib; 30% with acalabrutinib, and 8% zanubrutinib (some received more than one BTK inhibitor previously). The vast majority (83%) discontinued treatment due to refractory or progressive disease; 10% stopped due to toxicity; and the remainder halted treatment for other reasons.
Six in every 10 of the participants were classified on the MCL International Prognostic Index as intermediate; one-fourth were classified as high; and the remainder low. Patients received 200 mg of pirtrobrutinib once a day until disease either progressed or they experienced intolerable toxicity. Among the results:
Overall response rate was 50%; 13% responded completely
Median duration of response was 8.3 months
Duration of response rate at 6 months was 65%
Grade 3 or 4 abnormalities experienced by 10% or more of participants included decreased neutrophil counts, lymphocyte counts, and platelet counts
Further, bsAb therapy targeting CD20-CD3 is not yet approved but is showing promise as a potential therapy following BTK inhibitor failure. The treatment consists of an antibody containing two prongs. One is a CD20 protein that attaches to the lymphoma cell. The other is an anti-CD3 antibody that attaches to the T cell to bring the patient’s own T cells closer to the lymphoma to increase the cell kill.
Preliminary studies evaluating bsAbs in individuals with MCL, many of whom have failed on multiple other types of therapies, show a remarkably high response rate. In one such investigation, the bsAb glofitamab was given to 21 individuals as monotherapy following pretreatment with obinutuzumab. The regimen produced an overall response rate of 81% (n = 17) and a complete response rate of 68% (n = 14). Response was similar in participants who had and had not received prior BTK therapy. Among those who achieved a complete response, median duration was 2.4 months, and 12 of those who reached a complete response were still in remission at the study’s data cutoff point.
For younger individuals with treatment-naïve MCL, the current standard is chemotherapy and autologous stem-cell transplant (ASCT). For older individuals the standard is chemoimmunotherapy. The replacement or addition of the BTK inhibitor ibrutinib to these regimens is showing the promise of added clinical benefit in both age contingents.
Investigators presented results of the three-arm TRIANGLE trial at the 64th ASH Annual Meeting in December 2022. The study compared 1) chemotherapy followed by ASCT; 2) ibrutinib plus chemotherapy followed by ASCT and ibrutinib maintenance; and 3) ibrutinib plus chemotherapy followed by ibrutinib maintenance. Participants (n = 870) ≤ 65 years of age (median age 57) with previously untreated advanced-stage MCL were randomized to 1 of the 3 regimens. Investigators looked at overall response, complete response, and failure-free survival rates (FFS). Among the results:
Overall response rates were 98% in the 2 groups whose treatments included ibrutinib, versus 94% in the chemotherapy followed by ASCT group.
Complete response rates were 45% and 36%, respectively.
The non-ibrutinib regimen did not attain FFS superiority over ibrutinib plus chemotherapy, with a 3-year FFS rate of 72% and 86%, respectively (p=0.9979, hazard ratio [HR]: 1.77).
Ibrutinib plus chemotherapy was shown to be superior to chemotherapy/ASCT, with a 3-year FFS rate of 88% and 72%, respectively (p=0.0008, HR: 0.52).
The only adverse event differences of note occurred during maintenance treatment; there were significantly more grade 3-5 adverse events in the ibrutinib/chemotherapy/ASCT group, compared with the other 2 contingents.
Researchers noted in materials accompanying their presentation that, “It has been clearly demonstrated that the current standard high-dose regimen is not superior to the new ibrutinib-containing regimen without ASCT. More follow-up is needed to clarify the role of ASCT in the context of ibrutinib-containing treatment. However, the current results already support the use of ibrutinib in the first-line treatment of younger MCL patients.”
It also appears that ibrutinib added to standard chemoimmunotherapy can improve outcomes in older individuals with treatment-naïve MCL. In 2022, researchers published results from the international, randomized, double-blind, phase 3 SHINE trial. Participants (n = 523) were ≥ 65 years of age with previously untreated MCL and were randomized to receive either ibrutinib 560 mg daily or placebo added to chemoimmunotherapy consisting of bendamustine and rituximab every 4 weeks for 6 cycles. Individuals with a partial or complete response continued treatment every 8 weeks for up to 12 more doses. Investigators looked primarily at progression-free survival (PFS), as well as complete response, undetectable minimal residual disease, and time to worsening. Among the results:
116 participants (44%) in the ibrutinib group experienced disease progression or died, compared with 152 (58%) in the placebo contingent.
Median PFS was 80.6 months and 52.9 months, respectively.
PFS benefit was seen across most subgroups (patients categorized as high risk, and those with TP53 mutations did not benefit).
Complete response was seen in 66% and 58% of participants, respectively.
Undetectable minimal residual disease was observed in 62% and 57%, respectively.
Deaths attributed to disease progression or adverse events occurred in 22% and 28%, respectively.
Grade 3 or 4 adverse event incident rates were 82% and 77%, respectively.
Researchers noted that, “Given the shorter progression-free survival with current standard-care chemoimmunotherapy options, a prolongation of progression-free survival in response to primary therapy may provide patients with an improved opportunity for durable disease control in order to prevent or delay relapse.”
Data on the use of other BTK inhibitors as first-line treatment for MCL are forthcoming, including:
ECHO, a phase 3 trial assessing the efficacy of acalabrutinib versus placebo added to bendamustine and rituximab.
MANGROVE, a phase 3 study comparing zanubrutinib plus rituximab versus bendamustine plus rituximab.
ENRICH, a phase 2 study evaluating a chemotherapy-free option–ibrutinib and rituximab in older individuals.
OASIS, a randomized, phase 2 trial comparing ibrutinib/anti-CD20 antibodies (Ab) and Ibrutinib/anti-CD20 Ab/venetoclax given as fixed duration combinations.
The evolution of BTK inhibitors for relapsed MCL has great potential; further benefits continue to be explored.
Number of cancer survivors with functional limitations doubled in 20 years
Vishal Patel, BS, a student at the Dell Medical School at The University of Texas at Austin, and colleagues identified 51,258 cancer survivors from the National Health Interview Survey, representing a weighted population of approximately 178.8 million from 1999 to 2018.
Most survivors were women (60.2%) and were at least 65 years old (55.4%). In 1999, 3.6 million weighted survivors reported functional limitation. In 2018, the number increased to 8.2 million, a 2.25-fold increase.
The number of survivors who reported no limitations also increased, but not by as much. That group grew 1.34-fold during the study period.
For context, “the 70% prevalence of functional limitation among survivors in 2018 is nearly twice that of the general population,” the authors wrote.
Patients surveyed on function
Functional limitation was defined as “self-reported difficulty performing any of 12 routine physical or social activities without assistance.” Examples of the activities included difficulty sitting for more than 2 hours, difficulty participating in social activities or difficulty pushing or pulling an object the size of a living room chair.
Over the 2 decades analyzed, the adjusted prevalence of functional limitation was highest among survivors of pancreatic cancer (80.3%) and lung cancer (76.5%). Prevalence was lowest for survivors of melanoma (62.2%), breast (61.8%) and prostate (59.5%) cancers.
Not just a result of living longer
Mr. Patel told this publication that one assumption people might make when they read these results is that people are just living longer with cancer and losing functional ability accordingly.
“But, in fact, we found that the youngest [– those less than 65 years–] actually contributed to this trend more than the oldest people, which means it’s not just [happening], because people are getting older,” he said.
Hispanic and Black individuals had disproportionately higher increases in functional limitation; percentage point increases over the 2 decades were 19.5 for Black people, 25.1 for Hispanic people and 12.5 for White people. There may be a couple of reasons for that, Mr. Patel noted.
Those who are Black or Hispanic tend to have less access to cancer survivorship care for reasons including insurance status and historic health care inequities, he noted.
“The other potential reason is that they have had less access to cancer care historically. And if, 20 years ago Black and Hispanic individuals didn’t have access to some chemotherapies, and now they do, maybe it’s the increased access to care that’s causing these functional limitations. Because chemotherapy can sometimes be very toxic. It may be sort of a catch-up toxicity,” he said.
Quality of life beyond survivorship
Mr. Patel said the results seem to call for building on improved survival rates by tracking and improving function.
“It’s good to celebrate that there are more survivors. But now that we can keep people alive longer, maybe we can shift gears to improving their quality of life,” he said.
The more-than-doubling of functional limitations over 2 decades “is a very sobering trend,” he noted, while pointing out that the functional limitations applied to 8 million people in the United States – people whose needs are not being met.
There’s no sign of the trend stopping, he continued. “We saw no downward trend, only an upward trend.”
Increasingly, including functionality as an endpoint in cancer trials, in addition to improvements in mortality, is one place to start, he added.
“Our findings suggest an urgent need for care teams to understand and address function, for researchers to evaluate function as a core outcome in trials, and for health systems and policy makers to reimagine survivorship care, recognizing the burden of cancer and its treatment on physical, psychosocial, and cognitive function,” the authors wrote in their paper. Limitations of the study include the potential for recall bias, lack of cancer staging or treatment information, and the subjective perception of function.
A coauthor reported personal fees from Astellas, AstraZeneca, AAA, Blue Earth, Janssen, Lantheus, Myovant, Myriad Genetics, Novartis, Telix, and Sanofi, as well as grants from Pfizer and Bayer during the conduct of the study. No other disclosures were reported.
Vishal Patel, BS, a student at the Dell Medical School at The University of Texas at Austin, and colleagues identified 51,258 cancer survivors from the National Health Interview Survey, representing a weighted population of approximately 178.8 million from 1999 to 2018.
Most survivors were women (60.2%) and were at least 65 years old (55.4%). In 1999, 3.6 million weighted survivors reported functional limitation. In 2018, the number increased to 8.2 million, a 2.25-fold increase.
The number of survivors who reported no limitations also increased, but not by as much. That group grew 1.34-fold during the study period.
For context, “the 70% prevalence of functional limitation among survivors in 2018 is nearly twice that of the general population,” the authors wrote.
Patients surveyed on function
Functional limitation was defined as “self-reported difficulty performing any of 12 routine physical or social activities without assistance.” Examples of the activities included difficulty sitting for more than 2 hours, difficulty participating in social activities or difficulty pushing or pulling an object the size of a living room chair.
Over the 2 decades analyzed, the adjusted prevalence of functional limitation was highest among survivors of pancreatic cancer (80.3%) and lung cancer (76.5%). Prevalence was lowest for survivors of melanoma (62.2%), breast (61.8%) and prostate (59.5%) cancers.
Not just a result of living longer
Mr. Patel told this publication that one assumption people might make when they read these results is that people are just living longer with cancer and losing functional ability accordingly.
“But, in fact, we found that the youngest [– those less than 65 years–] actually contributed to this trend more than the oldest people, which means it’s not just [happening], because people are getting older,” he said.
Hispanic and Black individuals had disproportionately higher increases in functional limitation; percentage point increases over the 2 decades were 19.5 for Black people, 25.1 for Hispanic people and 12.5 for White people. There may be a couple of reasons for that, Mr. Patel noted.
Those who are Black or Hispanic tend to have less access to cancer survivorship care for reasons including insurance status and historic health care inequities, he noted.
“The other potential reason is that they have had less access to cancer care historically. And if, 20 years ago Black and Hispanic individuals didn’t have access to some chemotherapies, and now they do, maybe it’s the increased access to care that’s causing these functional limitations. Because chemotherapy can sometimes be very toxic. It may be sort of a catch-up toxicity,” he said.
Quality of life beyond survivorship
Mr. Patel said the results seem to call for building on improved survival rates by tracking and improving function.
“It’s good to celebrate that there are more survivors. But now that we can keep people alive longer, maybe we can shift gears to improving their quality of life,” he said.
The more-than-doubling of functional limitations over 2 decades “is a very sobering trend,” he noted, while pointing out that the functional limitations applied to 8 million people in the United States – people whose needs are not being met.
There’s no sign of the trend stopping, he continued. “We saw no downward trend, only an upward trend.”
Increasingly, including functionality as an endpoint in cancer trials, in addition to improvements in mortality, is one place to start, he added.
“Our findings suggest an urgent need for care teams to understand and address function, for researchers to evaluate function as a core outcome in trials, and for health systems and policy makers to reimagine survivorship care, recognizing the burden of cancer and its treatment on physical, psychosocial, and cognitive function,” the authors wrote in their paper. Limitations of the study include the potential for recall bias, lack of cancer staging or treatment information, and the subjective perception of function.
A coauthor reported personal fees from Astellas, AstraZeneca, AAA, Blue Earth, Janssen, Lantheus, Myovant, Myriad Genetics, Novartis, Telix, and Sanofi, as well as grants from Pfizer and Bayer during the conduct of the study. No other disclosures were reported.
Vishal Patel, BS, a student at the Dell Medical School at The University of Texas at Austin, and colleagues identified 51,258 cancer survivors from the National Health Interview Survey, representing a weighted population of approximately 178.8 million from 1999 to 2018.
Most survivors were women (60.2%) and were at least 65 years old (55.4%). In 1999, 3.6 million weighted survivors reported functional limitation. In 2018, the number increased to 8.2 million, a 2.25-fold increase.
The number of survivors who reported no limitations also increased, but not by as much. That group grew 1.34-fold during the study period.
For context, “the 70% prevalence of functional limitation among survivors in 2018 is nearly twice that of the general population,” the authors wrote.
Patients surveyed on function
Functional limitation was defined as “self-reported difficulty performing any of 12 routine physical or social activities without assistance.” Examples of the activities included difficulty sitting for more than 2 hours, difficulty participating in social activities or difficulty pushing or pulling an object the size of a living room chair.
Over the 2 decades analyzed, the adjusted prevalence of functional limitation was highest among survivors of pancreatic cancer (80.3%) and lung cancer (76.5%). Prevalence was lowest for survivors of melanoma (62.2%), breast (61.8%) and prostate (59.5%) cancers.
Not just a result of living longer
Mr. Patel told this publication that one assumption people might make when they read these results is that people are just living longer with cancer and losing functional ability accordingly.
“But, in fact, we found that the youngest [– those less than 65 years–] actually contributed to this trend more than the oldest people, which means it’s not just [happening], because people are getting older,” he said.
Hispanic and Black individuals had disproportionately higher increases in functional limitation; percentage point increases over the 2 decades were 19.5 for Black people, 25.1 for Hispanic people and 12.5 for White people. There may be a couple of reasons for that, Mr. Patel noted.
Those who are Black or Hispanic tend to have less access to cancer survivorship care for reasons including insurance status and historic health care inequities, he noted.
“The other potential reason is that they have had less access to cancer care historically. And if, 20 years ago Black and Hispanic individuals didn’t have access to some chemotherapies, and now they do, maybe it’s the increased access to care that’s causing these functional limitations. Because chemotherapy can sometimes be very toxic. It may be sort of a catch-up toxicity,” he said.
Quality of life beyond survivorship
Mr. Patel said the results seem to call for building on improved survival rates by tracking and improving function.
“It’s good to celebrate that there are more survivors. But now that we can keep people alive longer, maybe we can shift gears to improving their quality of life,” he said.
The more-than-doubling of functional limitations over 2 decades “is a very sobering trend,” he noted, while pointing out that the functional limitations applied to 8 million people in the United States – people whose needs are not being met.
There’s no sign of the trend stopping, he continued. “We saw no downward trend, only an upward trend.”
Increasingly, including functionality as an endpoint in cancer trials, in addition to improvements in mortality, is one place to start, he added.
“Our findings suggest an urgent need for care teams to understand and address function, for researchers to evaluate function as a core outcome in trials, and for health systems and policy makers to reimagine survivorship care, recognizing the burden of cancer and its treatment on physical, psychosocial, and cognitive function,” the authors wrote in their paper. Limitations of the study include the potential for recall bias, lack of cancer staging or treatment information, and the subjective perception of function.
A coauthor reported personal fees from Astellas, AstraZeneca, AAA, Blue Earth, Janssen, Lantheus, Myovant, Myriad Genetics, Novartis, Telix, and Sanofi, as well as grants from Pfizer and Bayer during the conduct of the study. No other disclosures were reported.
FROM JAMA ONCOLOGY
Patients With Newly Diagnosed Mantle Cell Lymphoma and the Relevance of Clinical Trials
What is the significance of the recent TRIANGLE study on mantle cell lymphoma (MCL)?
Dr. LaCasce: The TRIANGLE study is extremely important in previously untreated, transplant-eligible patients with MCL. The cutoff age for transplants varies by center and is between 60 and 75 years. In the absence of a TP53 mutation, we have typically used induction chemotherapy followed by autologous stem-cell transplant (ASCT), followed by 3 years of maintenance rituximab. Obviously, this is a lot of therapy.
The TRIANGLE study was a 3-arm study in which ibrutinib-containing therapy was compared with standard RCHOP/RDHAP followed by ASCT. Maintenance rituximab became standard of care midway through the trial and was added. In the first experimental arm, ibrutinib was combined with RCHOP and then given as maintenance for 2 years following ASCT. The second experimental arm included the same schedule of ibrutinib and omitted the ASCT.
The results are early, but what has been presented thus far, ibrutinib induction and maintenance with ASCT is clearly superior to the standard arm with ASCT. Although the data are not statistically mature, the failure-free survival of the 2 ibrutinib arms was similar, suggesting that transplant may not be necessary. Longer follow-up is necessary to confirm this conclusion and assess overall survival in all 3 arms.
If the results hold, ASCT could become a thing of the past or perhaps used in the second line. With the favorable activity of chimeric antigen receptor (CAR) T-cell therapy, however, it is unclear whether ASCT would be used in second line. Avoiding the sequential use of ASCT and CAR T-cell therapy is appealing given the stem-cell damage that can result. It is appealing to think about not using ASCT upfront, because ASCT increases the risk of myelodysplastic syndrome.
The TRIANGLE data are likely to change the frontline management of MCL. Although ibrutinib was the first Bruton tyrosine kinase (BTK) inhibitor approved in MCL and has obviously changed the field dramatically, it is significantly less well-tolerated than the next generation of drugs—acalabrutinib and zanubrutinib. I suspect these will be substituted for ibrutinib and we will see even more tolerable upfront regimens for patients with newly diagnosed MCL.
Have there been any disparities that you found in patients newly diagnosed with MCL regarding age, sex, or ethnicity?
Dr. LaCasce: MCL typically affects patients in their 60s. It is rare in young patients, and approximately 75% of the cases are male. If you look at the demographics, it is more common in White patients and less common in Hispanic and African American patients. In addition, there is an association with farming, which likely contribute to the demographics of patients with MCL.
What is your recommended approach to managing patients newly diagnosed with MCL in your day-to-day practice?
Dr. LaCasce: Management is a bit tricky right now because the TRIANGLE study is not part of any guidelines thus far. Therefore, most would argue the standard treatment continues to include ASCT upfront. There is an important, large randomized study (NCT03267433) going on in the United States that is assessing the role of ASCT in patients who are in MRD-negative complete remission at the end of induction therapy. These patients are randomized to ASCT plus maintenance rituximab versus maintenance rituximab alone.
We are still enrolling patients to participate in this study, which is addressing a different question than TRIANGLE. I think we will learn a lot from this study. For patients who are not interested in participating in this study, we talk about the risks and benefits of ASCT.
One or 2 years ago, I would have strongly encouraged patients who were appropriate candidates to consider transplant in first remission. With the TRIANGLE data, however, and now that we have CAR T-cell therapy, I think it is more important to tailor the recommendation to the individual patient. If a patient is reluctant about ASCT and the associated risks, I do not push it.
If patients want the most aggressive approach associated with the longest remissions, at this moment, before TRIANGLE findings have been adopted into guidelines, I continue to recommend ASCT. For patients who have TP53 mutation, however, we treat with typically less aggressive therapy, as this patient population does not benefit from ASCT. We look forward to more data incorporating BTK inhibitors upfront, particularly for this group of patients, who tend to have a more adverse prognosis.
Do you feel MCL data and clinical trials are important areas of focus for your colleagues?
Dr. LaCasce: Yes. I think it is a rapidly evolving field, which is really exciting. We are seeing data now from the bispecific antibodies in the relapsed/refractory setting. We also need more data using pirtobrutinib for patients who have had BTK inhibitors and compare pirtobrutinib (a non-covalent BTK inhibitor) with the covalent BTK inhibitors.
I would strongly encourage patients to participate in clinical trials so that we can better answer these important questions. When patients go online and read about MCL, they often see a median survival of 3 to 4 years, which is completely outdated. The overall prognosis of MCL has changed dramatically since I have been in the field. Hopefully, survival will continue to improve, and therapies will become more tolerable, as well.
What is the significance of the recent TRIANGLE study on mantle cell lymphoma (MCL)?
Dr. LaCasce: The TRIANGLE study is extremely important in previously untreated, transplant-eligible patients with MCL. The cutoff age for transplants varies by center and is between 60 and 75 years. In the absence of a TP53 mutation, we have typically used induction chemotherapy followed by autologous stem-cell transplant (ASCT), followed by 3 years of maintenance rituximab. Obviously, this is a lot of therapy.
The TRIANGLE study was a 3-arm study in which ibrutinib-containing therapy was compared with standard RCHOP/RDHAP followed by ASCT. Maintenance rituximab became standard of care midway through the trial and was added. In the first experimental arm, ibrutinib was combined with RCHOP and then given as maintenance for 2 years following ASCT. The second experimental arm included the same schedule of ibrutinib and omitted the ASCT.
The results are early, but what has been presented thus far, ibrutinib induction and maintenance with ASCT is clearly superior to the standard arm with ASCT. Although the data are not statistically mature, the failure-free survival of the 2 ibrutinib arms was similar, suggesting that transplant may not be necessary. Longer follow-up is necessary to confirm this conclusion and assess overall survival in all 3 arms.
If the results hold, ASCT could become a thing of the past or perhaps used in the second line. With the favorable activity of chimeric antigen receptor (CAR) T-cell therapy, however, it is unclear whether ASCT would be used in second line. Avoiding the sequential use of ASCT and CAR T-cell therapy is appealing given the stem-cell damage that can result. It is appealing to think about not using ASCT upfront, because ASCT increases the risk of myelodysplastic syndrome.
The TRIANGLE data are likely to change the frontline management of MCL. Although ibrutinib was the first Bruton tyrosine kinase (BTK) inhibitor approved in MCL and has obviously changed the field dramatically, it is significantly less well-tolerated than the next generation of drugs—acalabrutinib and zanubrutinib. I suspect these will be substituted for ibrutinib and we will see even more tolerable upfront regimens for patients with newly diagnosed MCL.
Have there been any disparities that you found in patients newly diagnosed with MCL regarding age, sex, or ethnicity?
Dr. LaCasce: MCL typically affects patients in their 60s. It is rare in young patients, and approximately 75% of the cases are male. If you look at the demographics, it is more common in White patients and less common in Hispanic and African American patients. In addition, there is an association with farming, which likely contribute to the demographics of patients with MCL.
What is your recommended approach to managing patients newly diagnosed with MCL in your day-to-day practice?
Dr. LaCasce: Management is a bit tricky right now because the TRIANGLE study is not part of any guidelines thus far. Therefore, most would argue the standard treatment continues to include ASCT upfront. There is an important, large randomized study (NCT03267433) going on in the United States that is assessing the role of ASCT in patients who are in MRD-negative complete remission at the end of induction therapy. These patients are randomized to ASCT plus maintenance rituximab versus maintenance rituximab alone.
We are still enrolling patients to participate in this study, which is addressing a different question than TRIANGLE. I think we will learn a lot from this study. For patients who are not interested in participating in this study, we talk about the risks and benefits of ASCT.
One or 2 years ago, I would have strongly encouraged patients who were appropriate candidates to consider transplant in first remission. With the TRIANGLE data, however, and now that we have CAR T-cell therapy, I think it is more important to tailor the recommendation to the individual patient. If a patient is reluctant about ASCT and the associated risks, I do not push it.
If patients want the most aggressive approach associated with the longest remissions, at this moment, before TRIANGLE findings have been adopted into guidelines, I continue to recommend ASCT. For patients who have TP53 mutation, however, we treat with typically less aggressive therapy, as this patient population does not benefit from ASCT. We look forward to more data incorporating BTK inhibitors upfront, particularly for this group of patients, who tend to have a more adverse prognosis.
Do you feel MCL data and clinical trials are important areas of focus for your colleagues?
Dr. LaCasce: Yes. I think it is a rapidly evolving field, which is really exciting. We are seeing data now from the bispecific antibodies in the relapsed/refractory setting. We also need more data using pirtobrutinib for patients who have had BTK inhibitors and compare pirtobrutinib (a non-covalent BTK inhibitor) with the covalent BTK inhibitors.
I would strongly encourage patients to participate in clinical trials so that we can better answer these important questions. When patients go online and read about MCL, they often see a median survival of 3 to 4 years, which is completely outdated. The overall prognosis of MCL has changed dramatically since I have been in the field. Hopefully, survival will continue to improve, and therapies will become more tolerable, as well.
What is the significance of the recent TRIANGLE study on mantle cell lymphoma (MCL)?
Dr. LaCasce: The TRIANGLE study is extremely important in previously untreated, transplant-eligible patients with MCL. The cutoff age for transplants varies by center and is between 60 and 75 years. In the absence of a TP53 mutation, we have typically used induction chemotherapy followed by autologous stem-cell transplant (ASCT), followed by 3 years of maintenance rituximab. Obviously, this is a lot of therapy.
The TRIANGLE study was a 3-arm study in which ibrutinib-containing therapy was compared with standard RCHOP/RDHAP followed by ASCT. Maintenance rituximab became standard of care midway through the trial and was added. In the first experimental arm, ibrutinib was combined with RCHOP and then given as maintenance for 2 years following ASCT. The second experimental arm included the same schedule of ibrutinib and omitted the ASCT.
The results are early, but what has been presented thus far, ibrutinib induction and maintenance with ASCT is clearly superior to the standard arm with ASCT. Although the data are not statistically mature, the failure-free survival of the 2 ibrutinib arms was similar, suggesting that transplant may not be necessary. Longer follow-up is necessary to confirm this conclusion and assess overall survival in all 3 arms.
If the results hold, ASCT could become a thing of the past or perhaps used in the second line. With the favorable activity of chimeric antigen receptor (CAR) T-cell therapy, however, it is unclear whether ASCT would be used in second line. Avoiding the sequential use of ASCT and CAR T-cell therapy is appealing given the stem-cell damage that can result. It is appealing to think about not using ASCT upfront, because ASCT increases the risk of myelodysplastic syndrome.
The TRIANGLE data are likely to change the frontline management of MCL. Although ibrutinib was the first Bruton tyrosine kinase (BTK) inhibitor approved in MCL and has obviously changed the field dramatically, it is significantly less well-tolerated than the next generation of drugs—acalabrutinib and zanubrutinib. I suspect these will be substituted for ibrutinib and we will see even more tolerable upfront regimens for patients with newly diagnosed MCL.
Have there been any disparities that you found in patients newly diagnosed with MCL regarding age, sex, or ethnicity?
Dr. LaCasce: MCL typically affects patients in their 60s. It is rare in young patients, and approximately 75% of the cases are male. If you look at the demographics, it is more common in White patients and less common in Hispanic and African American patients. In addition, there is an association with farming, which likely contribute to the demographics of patients with MCL.
What is your recommended approach to managing patients newly diagnosed with MCL in your day-to-day practice?
Dr. LaCasce: Management is a bit tricky right now because the TRIANGLE study is not part of any guidelines thus far. Therefore, most would argue the standard treatment continues to include ASCT upfront. There is an important, large randomized study (NCT03267433) going on in the United States that is assessing the role of ASCT in patients who are in MRD-negative complete remission at the end of induction therapy. These patients are randomized to ASCT plus maintenance rituximab versus maintenance rituximab alone.
We are still enrolling patients to participate in this study, which is addressing a different question than TRIANGLE. I think we will learn a lot from this study. For patients who are not interested in participating in this study, we talk about the risks and benefits of ASCT.
One or 2 years ago, I would have strongly encouraged patients who were appropriate candidates to consider transplant in first remission. With the TRIANGLE data, however, and now that we have CAR T-cell therapy, I think it is more important to tailor the recommendation to the individual patient. If a patient is reluctant about ASCT and the associated risks, I do not push it.
If patients want the most aggressive approach associated with the longest remissions, at this moment, before TRIANGLE findings have been adopted into guidelines, I continue to recommend ASCT. For patients who have TP53 mutation, however, we treat with typically less aggressive therapy, as this patient population does not benefit from ASCT. We look forward to more data incorporating BTK inhibitors upfront, particularly for this group of patients, who tend to have a more adverse prognosis.
Do you feel MCL data and clinical trials are important areas of focus for your colleagues?
Dr. LaCasce: Yes. I think it is a rapidly evolving field, which is really exciting. We are seeing data now from the bispecific antibodies in the relapsed/refractory setting. We also need more data using pirtobrutinib for patients who have had BTK inhibitors and compare pirtobrutinib (a non-covalent BTK inhibitor) with the covalent BTK inhibitors.
I would strongly encourage patients to participate in clinical trials so that we can better answer these important questions. When patients go online and read about MCL, they often see a median survival of 3 to 4 years, which is completely outdated. The overall prognosis of MCL has changed dramatically since I have been in the field. Hopefully, survival will continue to improve, and therapies will become more tolerable, as well.
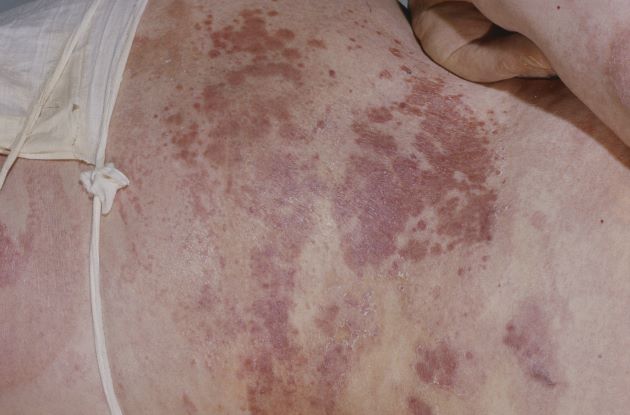
Three-month history of fever
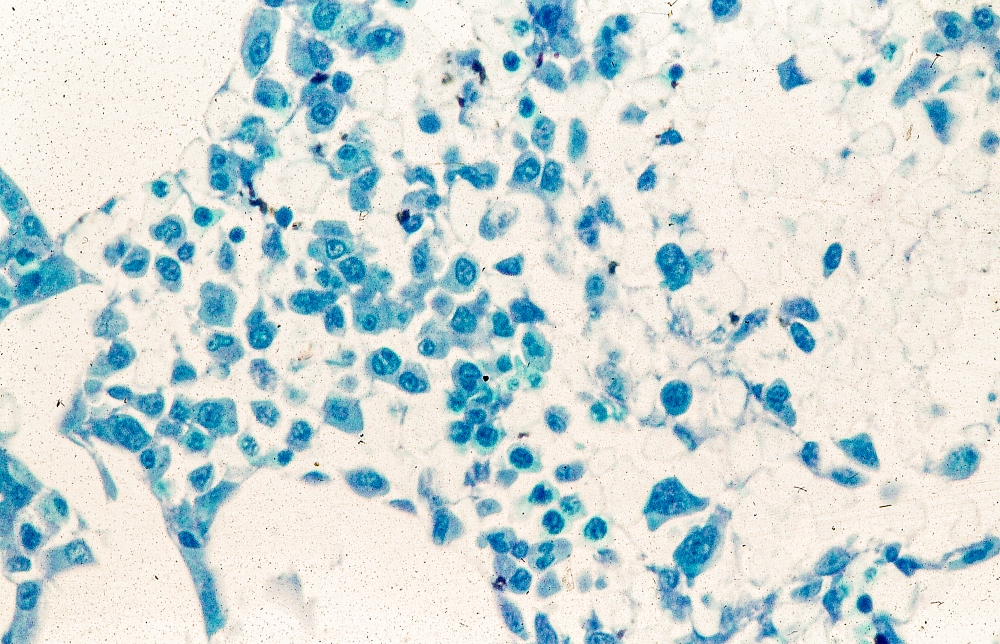
This patient's clinical presentation and laboratory findings are consistent with a diagnosis of pleomorphic mantle cell lymphoma (MCL).
MCL is a rare, clinically and biologically heterogeneous B-cell non-Hodgkin lymphoma. It accounts for approximately 5%-7% of all lymphomas. In North America and Europe, its incidence is akin to that of noncutaneous, peripheral T-cell lymphomas. The typical age at diagnosis is between 60 and 70 years. Approximately 70% of all cases are seen in men.
Little is known about risk factors for the development of MCL. Factors that have been associated with the development of other lymphomas (eg, familial risk, immunosuppression, other immune disorders, chemical and occupational exposures, and infectious agents) have not been convincingly identified as predisposing factors for MCL, with the possible exception of family history.
MCL is usually associated with reciprocal chromosomal translocation between chromosomes 11 and 14, t(11;14)(q13:q32), resulting in overexpression of cyclin D1, which plays a key role in tumor cell proliferation through cell-cycle dysregulation, chromosomal instability, and epigenetic regulation. Tumor cells (monoclonal B cells) express surface immunoglobulin, immunoglobulin M, or immunoglobulin D. Cells are usually CD5+ and pan B-cell antigen positive (eg, CD19, CD20, CD22) with no expression of CD10 and CD23. Histologic features include small-to-medium lymphocytes with scant cytoplasm, clumped chromatin, inconspicuous nucleoli, and prominent nuclear clefts. Cytologic subtypes include classic MCL, the blastoid variant (large cells, dispersed chromatin, and a high mitotic rate), and the pleomorphic variant (cells of varying size, although many are large, with pale cytoplasm, oval irregular nuclei, and prominent nucleoli). Blastoid and pleomorphic MCL typically have a more aggressive natural history and are associated with inferior clinical outcomes.
According to 2023 guidelines from the National Comprehensive Cancer Network (NCCN), an accurate pathologic diagnosis of the subtype is the most important initial step in the management of B-cell lymphomas, including pleomorphic MCL. The basic pathologic exam is the same for all subtypes, although additional testing may be needed in certain cases. An incisional or excisional lymph node biopsy is recommended. Fine-needle aspiration biopsy alone is typically not sufficient for the initial diagnosis of lymphoma; however, its diagnostic accuracy is significantly improved when it is used in combination with immunohistochemistry and flow cytometry. Immunohistochemistry is essential to differentiate MCL subtypes.
Essential workup procedures include a complete physical exam, with particular attention to node-bearing areas, including the Waldeyer ring, as well as the size of the liver and spleen, and assessment of performance status and B symptoms (fever, night sweats, unintentional weight loss). Laboratory studies should include complete blood count with differential, measurement of serum lactate dehydrogenase, hepatitis B virus testing, and a comprehensive metabolic panel. Required imaging studies include PET/CT (or chest/abdominal/pelvic CT with oral and intravenous contrast if PET/CT is not available) and multigated acquisition scanning or echocardiography when anthracyclines and anthracenedione-containing regimens are indicated.
A watch-and-wait approach may be appropriate for some patients with indolent MCL; however, patients with aggressive MCL, such as pleomorphic histology, require chemoimmunotherapy at diagnosis. For patients who relapse or achieve an incomplete response to first-line therapy, the NCCN guidelines recommend second-line treatment with a Bruton tyrosine kinase (BTK) inhibitor–containing regimen. Available BTK inhibitors include acalabrutinib, ibrutinib ± rituximab, zanubrutinib, and pirtobrutinib. Chemoimmunotherapy with lenalidomide + rituximab is another second-line option and may be particularly helpful for patients in whom a BTK inhibitor is contraindicated. Anti-CD19 CAR T-cell therapy is a recommended option for the third line and beyond.
Timothy J. Voorhees, MD, MSCR, Assistant Professor of Internal Medicine - Clinical, Division of Hematology, The Ohio State University James Comprehensive Cancer Center, Columbus, OH.
Timothy J. Voorhees, MD, MSCR, has disclosed the following relevant financial relationships:
Received research grant from: AstraZeneca; Morphosys; Incyte; Recordati.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
This patient's clinical presentation and laboratory findings are consistent with a diagnosis of pleomorphic mantle cell lymphoma (MCL).
MCL is a rare, clinically and biologically heterogeneous B-cell non-Hodgkin lymphoma. It accounts for approximately 5%-7% of all lymphomas. In North America and Europe, its incidence is akin to that of noncutaneous, peripheral T-cell lymphomas. The typical age at diagnosis is between 60 and 70 years. Approximately 70% of all cases are seen in men.
Little is known about risk factors for the development of MCL. Factors that have been associated with the development of other lymphomas (eg, familial risk, immunosuppression, other immune disorders, chemical and occupational exposures, and infectious agents) have not been convincingly identified as predisposing factors for MCL, with the possible exception of family history.
MCL is usually associated with reciprocal chromosomal translocation between chromosomes 11 and 14, t(11;14)(q13:q32), resulting in overexpression of cyclin D1, which plays a key role in tumor cell proliferation through cell-cycle dysregulation, chromosomal instability, and epigenetic regulation. Tumor cells (monoclonal B cells) express surface immunoglobulin, immunoglobulin M, or immunoglobulin D. Cells are usually CD5+ and pan B-cell antigen positive (eg, CD19, CD20, CD22) with no expression of CD10 and CD23. Histologic features include small-to-medium lymphocytes with scant cytoplasm, clumped chromatin, inconspicuous nucleoli, and prominent nuclear clefts. Cytologic subtypes include classic MCL, the blastoid variant (large cells, dispersed chromatin, and a high mitotic rate), and the pleomorphic variant (cells of varying size, although many are large, with pale cytoplasm, oval irregular nuclei, and prominent nucleoli). Blastoid and pleomorphic MCL typically have a more aggressive natural history and are associated with inferior clinical outcomes.
According to 2023 guidelines from the National Comprehensive Cancer Network (NCCN), an accurate pathologic diagnosis of the subtype is the most important initial step in the management of B-cell lymphomas, including pleomorphic MCL. The basic pathologic exam is the same for all subtypes, although additional testing may be needed in certain cases. An incisional or excisional lymph node biopsy is recommended. Fine-needle aspiration biopsy alone is typically not sufficient for the initial diagnosis of lymphoma; however, its diagnostic accuracy is significantly improved when it is used in combination with immunohistochemistry and flow cytometry. Immunohistochemistry is essential to differentiate MCL subtypes.
Essential workup procedures include a complete physical exam, with particular attention to node-bearing areas, including the Waldeyer ring, as well as the size of the liver and spleen, and assessment of performance status and B symptoms (fever, night sweats, unintentional weight loss). Laboratory studies should include complete blood count with differential, measurement of serum lactate dehydrogenase, hepatitis B virus testing, and a comprehensive metabolic panel. Required imaging studies include PET/CT (or chest/abdominal/pelvic CT with oral and intravenous contrast if PET/CT is not available) and multigated acquisition scanning or echocardiography when anthracyclines and anthracenedione-containing regimens are indicated.
A watch-and-wait approach may be appropriate for some patients with indolent MCL; however, patients with aggressive MCL, such as pleomorphic histology, require chemoimmunotherapy at diagnosis. For patients who relapse or achieve an incomplete response to first-line therapy, the NCCN guidelines recommend second-line treatment with a Bruton tyrosine kinase (BTK) inhibitor–containing regimen. Available BTK inhibitors include acalabrutinib, ibrutinib ± rituximab, zanubrutinib, and pirtobrutinib. Chemoimmunotherapy with lenalidomide + rituximab is another second-line option and may be particularly helpful for patients in whom a BTK inhibitor is contraindicated. Anti-CD19 CAR T-cell therapy is a recommended option for the third line and beyond.
Timothy J. Voorhees, MD, MSCR, Assistant Professor of Internal Medicine - Clinical, Division of Hematology, The Ohio State University James Comprehensive Cancer Center, Columbus, OH.
Timothy J. Voorhees, MD, MSCR, has disclosed the following relevant financial relationships:
Received research grant from: AstraZeneca; Morphosys; Incyte; Recordati.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
This patient's clinical presentation and laboratory findings are consistent with a diagnosis of pleomorphic mantle cell lymphoma (MCL).
MCL is a rare, clinically and biologically heterogeneous B-cell non-Hodgkin lymphoma. It accounts for approximately 5%-7% of all lymphomas. In North America and Europe, its incidence is akin to that of noncutaneous, peripheral T-cell lymphomas. The typical age at diagnosis is between 60 and 70 years. Approximately 70% of all cases are seen in men.
Little is known about risk factors for the development of MCL. Factors that have been associated with the development of other lymphomas (eg, familial risk, immunosuppression, other immune disorders, chemical and occupational exposures, and infectious agents) have not been convincingly identified as predisposing factors for MCL, with the possible exception of family history.
MCL is usually associated with reciprocal chromosomal translocation between chromosomes 11 and 14, t(11;14)(q13:q32), resulting in overexpression of cyclin D1, which plays a key role in tumor cell proliferation through cell-cycle dysregulation, chromosomal instability, and epigenetic regulation. Tumor cells (monoclonal B cells) express surface immunoglobulin, immunoglobulin M, or immunoglobulin D. Cells are usually CD5+ and pan B-cell antigen positive (eg, CD19, CD20, CD22) with no expression of CD10 and CD23. Histologic features include small-to-medium lymphocytes with scant cytoplasm, clumped chromatin, inconspicuous nucleoli, and prominent nuclear clefts. Cytologic subtypes include classic MCL, the blastoid variant (large cells, dispersed chromatin, and a high mitotic rate), and the pleomorphic variant (cells of varying size, although many are large, with pale cytoplasm, oval irregular nuclei, and prominent nucleoli). Blastoid and pleomorphic MCL typically have a more aggressive natural history and are associated with inferior clinical outcomes.
According to 2023 guidelines from the National Comprehensive Cancer Network (NCCN), an accurate pathologic diagnosis of the subtype is the most important initial step in the management of B-cell lymphomas, including pleomorphic MCL. The basic pathologic exam is the same for all subtypes, although additional testing may be needed in certain cases. An incisional or excisional lymph node biopsy is recommended. Fine-needle aspiration biopsy alone is typically not sufficient for the initial diagnosis of lymphoma; however, its diagnostic accuracy is significantly improved when it is used in combination with immunohistochemistry and flow cytometry. Immunohistochemistry is essential to differentiate MCL subtypes.
Essential workup procedures include a complete physical exam, with particular attention to node-bearing areas, including the Waldeyer ring, as well as the size of the liver and spleen, and assessment of performance status and B symptoms (fever, night sweats, unintentional weight loss). Laboratory studies should include complete blood count with differential, measurement of serum lactate dehydrogenase, hepatitis B virus testing, and a comprehensive metabolic panel. Required imaging studies include PET/CT (or chest/abdominal/pelvic CT with oral and intravenous contrast if PET/CT is not available) and multigated acquisition scanning or echocardiography when anthracyclines and anthracenedione-containing regimens are indicated.
A watch-and-wait approach may be appropriate for some patients with indolent MCL; however, patients with aggressive MCL, such as pleomorphic histology, require chemoimmunotherapy at diagnosis. For patients who relapse or achieve an incomplete response to first-line therapy, the NCCN guidelines recommend second-line treatment with a Bruton tyrosine kinase (BTK) inhibitor–containing regimen. Available BTK inhibitors include acalabrutinib, ibrutinib ± rituximab, zanubrutinib, and pirtobrutinib. Chemoimmunotherapy with lenalidomide + rituximab is another second-line option and may be particularly helpful for patients in whom a BTK inhibitor is contraindicated. Anti-CD19 CAR T-cell therapy is a recommended option for the third line and beyond.
Timothy J. Voorhees, MD, MSCR, Assistant Professor of Internal Medicine - Clinical, Division of Hematology, The Ohio State University James Comprehensive Cancer Center, Columbus, OH.
Timothy J. Voorhees, MD, MSCR, has disclosed the following relevant financial relationships:
Received research grant from: AstraZeneca; Morphosys; Incyte; Recordati.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
A 62-year-old man with no significant past medical history presents with a 3-month history of fever, night sweats, upper abdominal pain and bloating, and unintentional weight loss. He does not currently take any medications. His height and weight are 6 ft 2 in and 171 lb (BMI 22).
Physical examination reveals generalized lymphadenopathy and splenomegaly. Subsequently, an excisional lymph node biopsy is performed. Histologic examination of the specimen reveals sheets of mostly large cells of varying sizes, with nuclear overlap and extensive necrosis. Cytology findings include large lymphocytes with pale cytoplasm, clumped chromatin, oval irregular nuclei, and prominent nucleoli. Pertinent findings from immunohistochemical staining include the presence of t(11:14), Ki67 > 30%, CD5 and CD20 positivity, and CD10 and CD23 negativity. Centroblasts are absent.
MCL Treatment
Upfront Transplants in Patients With Mantle Cell Lymphoma
What is your outlook on the role of upfront autologous stem cell transplant (ASCT) for patients with mantle cell lymphoma (MCL)?
Dr. Barrientos: Most of the data that we have for upfront ASCT for young patients in frontline therapy come from the era when we did not use rituximab, and the data have not kept up with the pace of all the recent advances. Rituximab has changed the way we approach maintenance therapy after induction therapy. No randomized controlled trial data (in regimens that incorporate rituximab and cytarabine) have demonstrated a benefit in overall survival (OS) with ASCT in the modern era.
There is a lot to consider for every patient with MCL before we start therapy or discuss upfront transplant. MCL is one of these non-Hodgkin lymphomas that unfortunately can be aggressive in some patients depending on their prognostic markers and particular clinical features of the disease. Some patients have a more indolent form, whereas others have a more aggressive presentation at the time of diagnosis. The disease is heterogeneous and will respond differently to certain regimens. For example, patients with MCL who have a high proliferation rate, blastoid morphology, multiple chromosomal aberrations, complex karyotype, and/or the presence of tumor suppressor protein P53 (TP53) mutation will likely have a more aggressive course. Fitness for transplant is also an important consideration regardless of age; that is, a patient with comorbid end-stage chronic kidney or liver disease will not be able to tolerate a transplant.
Even with optimal therapy that incorporates rituximab and cytarabine, pursuing a transplant does not necessarily benefit survival in patients with a known TP53 mutation, as these patients typically experience increased toxicity without improved OS. We know they will not respond well, and we should discuss the available data so that the patients can make a sound decision and consider participation in a clinical trial that incorporates novel agents. Another type of mutation—cyclin-dependent kinase inhibitor 2A (CDKN2A)—also has lower OS. Concurrent deletion of CDKN2A and TP53 aberration (deletion and/or mutation) are known to be associated with lower OS given their chemoresistant nature. Patients with these genetic mutations should not be offered standard ASCT, but rather they should be identified early on and prioritized to participate in clinical trials.
Importantly, the role of upfront ASCT is changing right now, based on a recent trial that was presented at the latest American Society of Hematology meeting in 2022. The TRIANGLE trial demonstrated the addition of ibrutinib (a first-generation Bruton tyrosine kinase [BTK] inhibitor) to standard chemoimmunotherapy induction and 2 years of ibrutinib maintenance can improve outcomes vs standard chemoimmunotherapy induction and ASCT alone for younger patients with MCL. However, longer follow-up is needed to fully elucidate the role of ASCT in the era of BTK inhibitors when incorporated early on into the treatment paradigm.
The TRIANGLE trial was an international, randomized 3-arm phase 3 trial (EudraCT-no. 2014-001363-12) for young (up to 65 years) fit patients with histologically confirmed, untreated, advanced stage II-IV MCL. In the control arm A, patients received an alternating R-CHOP/R-DHAP induction followed by myeloablative consolidation (ASCT). In arm A+I, ibrutinib was added to the R-CHOP cycles (560 mg day 1-19) and was applied as maintenance (continuous dosing) for 2 years. In arm I, the same induction and maintenance was applied but high-dose consolidation and ASCT was skipped. A rituximab maintenance (single doses every 2 months for up to 3 years) was allowed to be added in all study arms according to national clinical routine.
The study showed that failure-free survival at 3 years was 72% with chemotherapy alone, 86% with ibrutinib alone, and 88% with ibrutinib plus ASCT. However, the ibrutinib plus ASCT group seemed to have much more toxicity, comorbidities, and other complications from the transplant. The OS data are not mature yet, but looking at the available data, ibrutinib alone might be more beneficial to our patients— not only in terms of efficacy, but also in tolerability and response, with less toxicity over time.
To put things in perspective, we did not have good salvage therapies a decade ago. At the time ASCT was incorporated, it was a good option that allowed numerous patients to achieve a deep response with durable remission duration. Before ibrutinib was approved, the overall response rate for the best salvage therapies was not as encouraging as the initial therapy and, with each relapse, the duration of response shortened. When ibrutinib came along, the overall response rate improved significantly. But again, these patients had relapsed/refractory disease. Researchers have been investigating what would happen if we used such a drug in earlier lines of therapy. Can we get better outcomes? Can we get patients in remission longer, similar to what we have seen with ASCT, but without the ASCT?
There has never been a single modern trial that has demonstrated that transplant improves survival. Transplantation can improve progression-free survival, but not OS. For a disease for which we do not have a cure, if we can keep patients in remission with a good salvage therapy and give them a better quality of life, without subjecting them to an ASCT, then I might choose that. New targeted agents and novel therapies are in clinical development all the time, so the future is bright for patients with this diagnosis. Given the novel salvage therapies in the pipeline, we may be able to no longer recommend ASCT upfront for most patients soon.
Can you share more about the potential benefits of using salvage therapies over ASCT, and particularly any promising newer agents in the salvage therapy setting?
Dr. Barrientos: Recently we had the FDA approval of pirtobrutinib—a noncovalently bound BTK inhibitor—for patients with relapsed/refractory MCL in whom at least 2 lines of systemic therapy had failed, including another BTK inhibitor. In the trial that led to the accelerated approval, pirtobrutinib-treated patients showed an overall response rate of 50% in those who received the drug at 200 mg daily (n = 120); most of the responses were partial responses. The efficacy of other novel drugs are being studied in patients with MCL. For example, ROR1 (receptor tyrosine kinase–like orphan receptor 1) inhibitors and BTK degraders are currently in clinical trials. Chimeric antigen receptor (CAR) T-cell therapy targeting CD19 has been approved for the treatment of adult patients with relapsed or refractory MCL, and this may be an option for some patients.
Multiple novel agents might be able to salvage our patients without subjecting them to an upfront transplant. My hope is to get away from using the intense chemotherapy regimens that might cause myelosuppression, infection risk, or other toxicities, and try to stay with the novel agents. We need to do better for our patients.
Based on the data we now have, until there is a trial that demonstrates a higher OS rate with ASCT, it is hard for me to tell a patient to blindly pursue ASCT without learning more about all the available options. If you have access to a good salvage therapy, especially with all these new promising agents, a patient might be able to stay in remission without having ASCT, which can still have an increased risk of morbidity.
Are there certain patient groups that should never be considered for ASCT?
Dr. Barrientos: Younger patients with the CDKN2A gene—which represents about 22% of patients—and those who have a TP53 mutation should not be considered for a standard transplant because they have a worse outcome independent of the treatment. I would also include complex karyotype patients because of the same nature of the chromosomal aberrations. The more genetic aberrations that a patient has, the more likelihood that any chemotherapy will damage the DNA further and create a more aggressive clone. Instead, I would recommend that young patients in this category participate in a clinical trial with novel agents.
With novel therapies in the pipeline, the availability of CAR T, and now the bispecific antibodies such as blinatumomab and HexAbs coming along, the number of patients who may opt out of ASCT may increase. I have a long discussion with my patients. The more educated they are, the better it is for the patient. At the end of the day, the most important thing for me, with any therapy, is: how does the patient feel? Because if we cannot cure a patient or provide a survival advantage, I do not want to give that patient something that will decrease their quality of life. I would rather keep the patient in some sort of stable disease remission, comfortable, and having a good quality of life. That is my goal for anyone who cannot be cured. Now if it is a curable disease, like a diffuse large cell lymphoma or a Burkitt’s lymphoma, then it is a different story. But for people with MCL, a disease that you cannot cure, or chronic lymphocytic leukemia or follicular lymphoma, then it becomes a different discussion. Undetectable minimal residual disease correlates with longer remission durations, but sometimes trying to achieve that, you can actually do a lot of harm to some patients.
Are there any other conversations you have with your patients in day-to-day practice?
Dr. Barrientos: I always tell my patients to be on top of the age-appropriate cancer screening recommendations. For example, they should see a dermatologist once a year. Men should make sure that their prostate is checked. I recommend women get breast mammograms, Pap smears, and most importantly to avoid smoking—and that includes vaping. It is important to lead a healthy life to minimize the risk of secondary malignancies.
For risk of infections, I recommend to all my patients to be up to date on their vaccinations, such as pneumonia if they are older than 65, Shingrix for prevention of reactivation of varicella or chickenpox, and the flu shot once a year. I also recommend the COVID-19 vaccine even now, as our patients with blood disorders might have a harder time fighting COVID-19 infection. I always tell my patients to please reach out to us because we can discuss the use of antivirals such as Paxlovid (nirmatrelvir/ritonavir), and if they are sick, then they can get remdesivir in the hospital.
I want to touch on health literacy and disparities for a moment. I have some younger patients who are Latin or Black with uncontrolled hypertension or diabetes, even at a young age, and do not realize that I can treat their cancer into remission, but if their blood glucose is in the 500 range, they could die from their diabetes. So talking with patients about their overall health is important. Survivorship issues are important, especially if patients are diagnosed at a young age. We have known for a long time that chemotherapy can create cardiac events, arrhythmias, and heart disease. Therefore, I always tell patients with metabolic syndrome to try to exercise and eat healthy. Patients should get an electrocardiogram and see an internist at least once a year to make sure their cholesterol is well controlled. I think now we are being more cognizant that many complications can happen even 10 years after cancer treatment.
What is your outlook on the role of upfront autologous stem cell transplant (ASCT) for patients with mantle cell lymphoma (MCL)?
Dr. Barrientos: Most of the data that we have for upfront ASCT for young patients in frontline therapy come from the era when we did not use rituximab, and the data have not kept up with the pace of all the recent advances. Rituximab has changed the way we approach maintenance therapy after induction therapy. No randomized controlled trial data (in regimens that incorporate rituximab and cytarabine) have demonstrated a benefit in overall survival (OS) with ASCT in the modern era.
There is a lot to consider for every patient with MCL before we start therapy or discuss upfront transplant. MCL is one of these non-Hodgkin lymphomas that unfortunately can be aggressive in some patients depending on their prognostic markers and particular clinical features of the disease. Some patients have a more indolent form, whereas others have a more aggressive presentation at the time of diagnosis. The disease is heterogeneous and will respond differently to certain regimens. For example, patients with MCL who have a high proliferation rate, blastoid morphology, multiple chromosomal aberrations, complex karyotype, and/or the presence of tumor suppressor protein P53 (TP53) mutation will likely have a more aggressive course. Fitness for transplant is also an important consideration regardless of age; that is, a patient with comorbid end-stage chronic kidney or liver disease will not be able to tolerate a transplant.
Even with optimal therapy that incorporates rituximab and cytarabine, pursuing a transplant does not necessarily benefit survival in patients with a known TP53 mutation, as these patients typically experience increased toxicity without improved OS. We know they will not respond well, and we should discuss the available data so that the patients can make a sound decision and consider participation in a clinical trial that incorporates novel agents. Another type of mutation—cyclin-dependent kinase inhibitor 2A (CDKN2A)—also has lower OS. Concurrent deletion of CDKN2A and TP53 aberration (deletion and/or mutation) are known to be associated with lower OS given their chemoresistant nature. Patients with these genetic mutations should not be offered standard ASCT, but rather they should be identified early on and prioritized to participate in clinical trials.
Importantly, the role of upfront ASCT is changing right now, based on a recent trial that was presented at the latest American Society of Hematology meeting in 2022. The TRIANGLE trial demonstrated the addition of ibrutinib (a first-generation Bruton tyrosine kinase [BTK] inhibitor) to standard chemoimmunotherapy induction and 2 years of ibrutinib maintenance can improve outcomes vs standard chemoimmunotherapy induction and ASCT alone for younger patients with MCL. However, longer follow-up is needed to fully elucidate the role of ASCT in the era of BTK inhibitors when incorporated early on into the treatment paradigm.
The TRIANGLE trial was an international, randomized 3-arm phase 3 trial (EudraCT-no. 2014-001363-12) for young (up to 65 years) fit patients with histologically confirmed, untreated, advanced stage II-IV MCL. In the control arm A, patients received an alternating R-CHOP/R-DHAP induction followed by myeloablative consolidation (ASCT). In arm A+I, ibrutinib was added to the R-CHOP cycles (560 mg day 1-19) and was applied as maintenance (continuous dosing) for 2 years. In arm I, the same induction and maintenance was applied but high-dose consolidation and ASCT was skipped. A rituximab maintenance (single doses every 2 months for up to 3 years) was allowed to be added in all study arms according to national clinical routine.
The study showed that failure-free survival at 3 years was 72% with chemotherapy alone, 86% with ibrutinib alone, and 88% with ibrutinib plus ASCT. However, the ibrutinib plus ASCT group seemed to have much more toxicity, comorbidities, and other complications from the transplant. The OS data are not mature yet, but looking at the available data, ibrutinib alone might be more beneficial to our patients— not only in terms of efficacy, but also in tolerability and response, with less toxicity over time.
To put things in perspective, we did not have good salvage therapies a decade ago. At the time ASCT was incorporated, it was a good option that allowed numerous patients to achieve a deep response with durable remission duration. Before ibrutinib was approved, the overall response rate for the best salvage therapies was not as encouraging as the initial therapy and, with each relapse, the duration of response shortened. When ibrutinib came along, the overall response rate improved significantly. But again, these patients had relapsed/refractory disease. Researchers have been investigating what would happen if we used such a drug in earlier lines of therapy. Can we get better outcomes? Can we get patients in remission longer, similar to what we have seen with ASCT, but without the ASCT?
There has never been a single modern trial that has demonstrated that transplant improves survival. Transplantation can improve progression-free survival, but not OS. For a disease for which we do not have a cure, if we can keep patients in remission with a good salvage therapy and give them a better quality of life, without subjecting them to an ASCT, then I might choose that. New targeted agents and novel therapies are in clinical development all the time, so the future is bright for patients with this diagnosis. Given the novel salvage therapies in the pipeline, we may be able to no longer recommend ASCT upfront for most patients soon.
Can you share more about the potential benefits of using salvage therapies over ASCT, and particularly any promising newer agents in the salvage therapy setting?
Dr. Barrientos: Recently we had the FDA approval of pirtobrutinib—a noncovalently bound BTK inhibitor—for patients with relapsed/refractory MCL in whom at least 2 lines of systemic therapy had failed, including another BTK inhibitor. In the trial that led to the accelerated approval, pirtobrutinib-treated patients showed an overall response rate of 50% in those who received the drug at 200 mg daily (n = 120); most of the responses were partial responses. The efficacy of other novel drugs are being studied in patients with MCL. For example, ROR1 (receptor tyrosine kinase–like orphan receptor 1) inhibitors and BTK degraders are currently in clinical trials. Chimeric antigen receptor (CAR) T-cell therapy targeting CD19 has been approved for the treatment of adult patients with relapsed or refractory MCL, and this may be an option for some patients.
Multiple novel agents might be able to salvage our patients without subjecting them to an upfront transplant. My hope is to get away from using the intense chemotherapy regimens that might cause myelosuppression, infection risk, or other toxicities, and try to stay with the novel agents. We need to do better for our patients.
Based on the data we now have, until there is a trial that demonstrates a higher OS rate with ASCT, it is hard for me to tell a patient to blindly pursue ASCT without learning more about all the available options. If you have access to a good salvage therapy, especially with all these new promising agents, a patient might be able to stay in remission without having ASCT, which can still have an increased risk of morbidity.
Are there certain patient groups that should never be considered for ASCT?
Dr. Barrientos: Younger patients with the CDKN2A gene—which represents about 22% of patients—and those who have a TP53 mutation should not be considered for a standard transplant because they have a worse outcome independent of the treatment. I would also include complex karyotype patients because of the same nature of the chromosomal aberrations. The more genetic aberrations that a patient has, the more likelihood that any chemotherapy will damage the DNA further and create a more aggressive clone. Instead, I would recommend that young patients in this category participate in a clinical trial with novel agents.
With novel therapies in the pipeline, the availability of CAR T, and now the bispecific antibodies such as blinatumomab and HexAbs coming along, the number of patients who may opt out of ASCT may increase. I have a long discussion with my patients. The more educated they are, the better it is for the patient. At the end of the day, the most important thing for me, with any therapy, is: how does the patient feel? Because if we cannot cure a patient or provide a survival advantage, I do not want to give that patient something that will decrease their quality of life. I would rather keep the patient in some sort of stable disease remission, comfortable, and having a good quality of life. That is my goal for anyone who cannot be cured. Now if it is a curable disease, like a diffuse large cell lymphoma or a Burkitt’s lymphoma, then it is a different story. But for people with MCL, a disease that you cannot cure, or chronic lymphocytic leukemia or follicular lymphoma, then it becomes a different discussion. Undetectable minimal residual disease correlates with longer remission durations, but sometimes trying to achieve that, you can actually do a lot of harm to some patients.
Are there any other conversations you have with your patients in day-to-day practice?
Dr. Barrientos: I always tell my patients to be on top of the age-appropriate cancer screening recommendations. For example, they should see a dermatologist once a year. Men should make sure that their prostate is checked. I recommend women get breast mammograms, Pap smears, and most importantly to avoid smoking—and that includes vaping. It is important to lead a healthy life to minimize the risk of secondary malignancies.
For risk of infections, I recommend to all my patients to be up to date on their vaccinations, such as pneumonia if they are older than 65, Shingrix for prevention of reactivation of varicella or chickenpox, and the flu shot once a year. I also recommend the COVID-19 vaccine even now, as our patients with blood disorders might have a harder time fighting COVID-19 infection. I always tell my patients to please reach out to us because we can discuss the use of antivirals such as Paxlovid (nirmatrelvir/ritonavir), and if they are sick, then they can get remdesivir in the hospital.
I want to touch on health literacy and disparities for a moment. I have some younger patients who are Latin or Black with uncontrolled hypertension or diabetes, even at a young age, and do not realize that I can treat their cancer into remission, but if their blood glucose is in the 500 range, they could die from their diabetes. So talking with patients about their overall health is important. Survivorship issues are important, especially if patients are diagnosed at a young age. We have known for a long time that chemotherapy can create cardiac events, arrhythmias, and heart disease. Therefore, I always tell patients with metabolic syndrome to try to exercise and eat healthy. Patients should get an electrocardiogram and see an internist at least once a year to make sure their cholesterol is well controlled. I think now we are being more cognizant that many complications can happen even 10 years after cancer treatment.
What is your outlook on the role of upfront autologous stem cell transplant (ASCT) for patients with mantle cell lymphoma (MCL)?
Dr. Barrientos: Most of the data that we have for upfront ASCT for young patients in frontline therapy come from the era when we did not use rituximab, and the data have not kept up with the pace of all the recent advances. Rituximab has changed the way we approach maintenance therapy after induction therapy. No randomized controlled trial data (in regimens that incorporate rituximab and cytarabine) have demonstrated a benefit in overall survival (OS) with ASCT in the modern era.
There is a lot to consider for every patient with MCL before we start therapy or discuss upfront transplant. MCL is one of these non-Hodgkin lymphomas that unfortunately can be aggressive in some patients depending on their prognostic markers and particular clinical features of the disease. Some patients have a more indolent form, whereas others have a more aggressive presentation at the time of diagnosis. The disease is heterogeneous and will respond differently to certain regimens. For example, patients with MCL who have a high proliferation rate, blastoid morphology, multiple chromosomal aberrations, complex karyotype, and/or the presence of tumor suppressor protein P53 (TP53) mutation will likely have a more aggressive course. Fitness for transplant is also an important consideration regardless of age; that is, a patient with comorbid end-stage chronic kidney or liver disease will not be able to tolerate a transplant.
Even with optimal therapy that incorporates rituximab and cytarabine, pursuing a transplant does not necessarily benefit survival in patients with a known TP53 mutation, as these patients typically experience increased toxicity without improved OS. We know they will not respond well, and we should discuss the available data so that the patients can make a sound decision and consider participation in a clinical trial that incorporates novel agents. Another type of mutation—cyclin-dependent kinase inhibitor 2A (CDKN2A)—also has lower OS. Concurrent deletion of CDKN2A and TP53 aberration (deletion and/or mutation) are known to be associated with lower OS given their chemoresistant nature. Patients with these genetic mutations should not be offered standard ASCT, but rather they should be identified early on and prioritized to participate in clinical trials.
Importantly, the role of upfront ASCT is changing right now, based on a recent trial that was presented at the latest American Society of Hematology meeting in 2022. The TRIANGLE trial demonstrated the addition of ibrutinib (a first-generation Bruton tyrosine kinase [BTK] inhibitor) to standard chemoimmunotherapy induction and 2 years of ibrutinib maintenance can improve outcomes vs standard chemoimmunotherapy induction and ASCT alone for younger patients with MCL. However, longer follow-up is needed to fully elucidate the role of ASCT in the era of BTK inhibitors when incorporated early on into the treatment paradigm.
The TRIANGLE trial was an international, randomized 3-arm phase 3 trial (EudraCT-no. 2014-001363-12) for young (up to 65 years) fit patients with histologically confirmed, untreated, advanced stage II-IV MCL. In the control arm A, patients received an alternating R-CHOP/R-DHAP induction followed by myeloablative consolidation (ASCT). In arm A+I, ibrutinib was added to the R-CHOP cycles (560 mg day 1-19) and was applied as maintenance (continuous dosing) for 2 years. In arm I, the same induction and maintenance was applied but high-dose consolidation and ASCT was skipped. A rituximab maintenance (single doses every 2 months for up to 3 years) was allowed to be added in all study arms according to national clinical routine.
The study showed that failure-free survival at 3 years was 72% with chemotherapy alone, 86% with ibrutinib alone, and 88% with ibrutinib plus ASCT. However, the ibrutinib plus ASCT group seemed to have much more toxicity, comorbidities, and other complications from the transplant. The OS data are not mature yet, but looking at the available data, ibrutinib alone might be more beneficial to our patients— not only in terms of efficacy, but also in tolerability and response, with less toxicity over time.
To put things in perspective, we did not have good salvage therapies a decade ago. At the time ASCT was incorporated, it was a good option that allowed numerous patients to achieve a deep response with durable remission duration. Before ibrutinib was approved, the overall response rate for the best salvage therapies was not as encouraging as the initial therapy and, with each relapse, the duration of response shortened. When ibrutinib came along, the overall response rate improved significantly. But again, these patients had relapsed/refractory disease. Researchers have been investigating what would happen if we used such a drug in earlier lines of therapy. Can we get better outcomes? Can we get patients in remission longer, similar to what we have seen with ASCT, but without the ASCT?
There has never been a single modern trial that has demonstrated that transplant improves survival. Transplantation can improve progression-free survival, but not OS. For a disease for which we do not have a cure, if we can keep patients in remission with a good salvage therapy and give them a better quality of life, without subjecting them to an ASCT, then I might choose that. New targeted agents and novel therapies are in clinical development all the time, so the future is bright for patients with this diagnosis. Given the novel salvage therapies in the pipeline, we may be able to no longer recommend ASCT upfront for most patients soon.
Can you share more about the potential benefits of using salvage therapies over ASCT, and particularly any promising newer agents in the salvage therapy setting?
Dr. Barrientos: Recently we had the FDA approval of pirtobrutinib—a noncovalently bound BTK inhibitor—for patients with relapsed/refractory MCL in whom at least 2 lines of systemic therapy had failed, including another BTK inhibitor. In the trial that led to the accelerated approval, pirtobrutinib-treated patients showed an overall response rate of 50% in those who received the drug at 200 mg daily (n = 120); most of the responses were partial responses. The efficacy of other novel drugs are being studied in patients with MCL. For example, ROR1 (receptor tyrosine kinase–like orphan receptor 1) inhibitors and BTK degraders are currently in clinical trials. Chimeric antigen receptor (CAR) T-cell therapy targeting CD19 has been approved for the treatment of adult patients with relapsed or refractory MCL, and this may be an option for some patients.
Multiple novel agents might be able to salvage our patients without subjecting them to an upfront transplant. My hope is to get away from using the intense chemotherapy regimens that might cause myelosuppression, infection risk, or other toxicities, and try to stay with the novel agents. We need to do better for our patients.
Based on the data we now have, until there is a trial that demonstrates a higher OS rate with ASCT, it is hard for me to tell a patient to blindly pursue ASCT without learning more about all the available options. If you have access to a good salvage therapy, especially with all these new promising agents, a patient might be able to stay in remission without having ASCT, which can still have an increased risk of morbidity.
Are there certain patient groups that should never be considered for ASCT?
Dr. Barrientos: Younger patients with the CDKN2A gene—which represents about 22% of patients—and those who have a TP53 mutation should not be considered for a standard transplant because they have a worse outcome independent of the treatment. I would also include complex karyotype patients because of the same nature of the chromosomal aberrations. The more genetic aberrations that a patient has, the more likelihood that any chemotherapy will damage the DNA further and create a more aggressive clone. Instead, I would recommend that young patients in this category participate in a clinical trial with novel agents.
With novel therapies in the pipeline, the availability of CAR T, and now the bispecific antibodies such as blinatumomab and HexAbs coming along, the number of patients who may opt out of ASCT may increase. I have a long discussion with my patients. The more educated they are, the better it is for the patient. At the end of the day, the most important thing for me, with any therapy, is: how does the patient feel? Because if we cannot cure a patient or provide a survival advantage, I do not want to give that patient something that will decrease their quality of life. I would rather keep the patient in some sort of stable disease remission, comfortable, and having a good quality of life. That is my goal for anyone who cannot be cured. Now if it is a curable disease, like a diffuse large cell lymphoma or a Burkitt’s lymphoma, then it is a different story. But for people with MCL, a disease that you cannot cure, or chronic lymphocytic leukemia or follicular lymphoma, then it becomes a different discussion. Undetectable minimal residual disease correlates with longer remission durations, but sometimes trying to achieve that, you can actually do a lot of harm to some patients.
Are there any other conversations you have with your patients in day-to-day practice?
Dr. Barrientos: I always tell my patients to be on top of the age-appropriate cancer screening recommendations. For example, they should see a dermatologist once a year. Men should make sure that their prostate is checked. I recommend women get breast mammograms, Pap smears, and most importantly to avoid smoking—and that includes vaping. It is important to lead a healthy life to minimize the risk of secondary malignancies.
For risk of infections, I recommend to all my patients to be up to date on their vaccinations, such as pneumonia if they are older than 65, Shingrix for prevention of reactivation of varicella or chickenpox, and the flu shot once a year. I also recommend the COVID-19 vaccine even now, as our patients with blood disorders might have a harder time fighting COVID-19 infection. I always tell my patients to please reach out to us because we can discuss the use of antivirals such as Paxlovid (nirmatrelvir/ritonavir), and if they are sick, then they can get remdesivir in the hospital.
I want to touch on health literacy and disparities for a moment. I have some younger patients who are Latin or Black with uncontrolled hypertension or diabetes, even at a young age, and do not realize that I can treat their cancer into remission, but if their blood glucose is in the 500 range, they could die from their diabetes. So talking with patients about their overall health is important. Survivorship issues are important, especially if patients are diagnosed at a young age. We have known for a long time that chemotherapy can create cardiac events, arrhythmias, and heart disease. Therefore, I always tell patients with metabolic syndrome to try to exercise and eat healthy. Patients should get an electrocardiogram and see an internist at least once a year to make sure their cholesterol is well controlled. I think now we are being more cognizant that many complications can happen even 10 years after cancer treatment.
Erythema extent predicts death in cutaneous GVHD
“There is value in collecting erythema serially over time as a continuous variable on a scale of 0%-100%” to identify high-risk patients for prophylactic and preemptive treatment, say investigators led by dermatologist Emily Baumrin, MD, director of the GVHD clinic at the University of Pennsylvania, Philadelphia.
They report a study of more than 300 patients with ccGVHD, which found that the extent of skin erythema strongly predicted the risk for death from GVHD.
Of the 267 patients with cutaneous GVHD at baseline, 103 patients died, the majority without a relapse of their blood cancer.
With additional research, erythema body surface area (BSA) should be “introduced as an outcome measure in clinical practice and trials,” they conclude.
At the moment, the NIH Skin Score is commonly used for risk assessment in cutaneous GVHD, but the researchers found that erythema BSA out-predicts this score.
The investigators explain that the NIH Skin Score does incorporate erythema surface area, but it does so as a categorical variable, not a continuous variable. Among other additional factors, it also includes assessments of skin sclerosis, which the investigators found was not associated with GVHD mortality.
Overall, the composite score waters down the weight given to erythema BSA because the score is “driven by stable sclerotic features, and erythema changes are missed,” they explain.
The study was published online in JAMA Dermatology.
Study details
The study included 469 patients with chronic GVHD (cGVHD), of whom 267 (57%) had cutaneous cGVHD at enrollment and 89 (19%) developed skin involvement subsequently.
All of the patients were on systemic immunosuppression for GVHD after allogeneic stem cell transplants for various blood cancers.
They were enrolled from 2007 through 2012 at nine U.S. medical centers – all members of the Chronic Graft Versus Host Disease Consortium – and they were followed until 2018.
Erythema BSA and NIH Skin Score were assessed at baseline and then every 3-6 months. Erythema was the first manifestation of skin involvement in the majority of patients, with a median surface area involvement of 11% at baseline.
The study team found that the extent of erythema at first follow-up visit was associated with both nonrelapse mortality (hazard ratio, 1.33 per 10% BSA increase; P < .001) and overall survival (HR, 1.28 per 10% BSA increase; P < .001), whereas extent of sclerotic skin involvement was not associated with either.
Participants in the study were predominantly White. The investigators note that “BSA assessments of erythema may be less reliable in patients with darker skin.”
The work was funded by the Department of Veterans Affairs and the National Institutes of Health. Dr. Baumrin had no disclosures; one coauthor is an employee of CorEvitas, and two others reported grants/adviser fees from several companies, including Janssen, Mallinckrodt, and Pfizer.
A version of this article first appeared on Medscape.com.
“There is value in collecting erythema serially over time as a continuous variable on a scale of 0%-100%” to identify high-risk patients for prophylactic and preemptive treatment, say investigators led by dermatologist Emily Baumrin, MD, director of the GVHD clinic at the University of Pennsylvania, Philadelphia.
They report a study of more than 300 patients with ccGVHD, which found that the extent of skin erythema strongly predicted the risk for death from GVHD.
Of the 267 patients with cutaneous GVHD at baseline, 103 patients died, the majority without a relapse of their blood cancer.
With additional research, erythema body surface area (BSA) should be “introduced as an outcome measure in clinical practice and trials,” they conclude.
At the moment, the NIH Skin Score is commonly used for risk assessment in cutaneous GVHD, but the researchers found that erythema BSA out-predicts this score.
The investigators explain that the NIH Skin Score does incorporate erythema surface area, but it does so as a categorical variable, not a continuous variable. Among other additional factors, it also includes assessments of skin sclerosis, which the investigators found was not associated with GVHD mortality.
Overall, the composite score waters down the weight given to erythema BSA because the score is “driven by stable sclerotic features, and erythema changes are missed,” they explain.
The study was published online in JAMA Dermatology.
Study details
The study included 469 patients with chronic GVHD (cGVHD), of whom 267 (57%) had cutaneous cGVHD at enrollment and 89 (19%) developed skin involvement subsequently.
All of the patients were on systemic immunosuppression for GVHD after allogeneic stem cell transplants for various blood cancers.
They were enrolled from 2007 through 2012 at nine U.S. medical centers – all members of the Chronic Graft Versus Host Disease Consortium – and they were followed until 2018.
Erythema BSA and NIH Skin Score were assessed at baseline and then every 3-6 months. Erythema was the first manifestation of skin involvement in the majority of patients, with a median surface area involvement of 11% at baseline.
The study team found that the extent of erythema at first follow-up visit was associated with both nonrelapse mortality (hazard ratio, 1.33 per 10% BSA increase; P < .001) and overall survival (HR, 1.28 per 10% BSA increase; P < .001), whereas extent of sclerotic skin involvement was not associated with either.
Participants in the study were predominantly White. The investigators note that “BSA assessments of erythema may be less reliable in patients with darker skin.”
The work was funded by the Department of Veterans Affairs and the National Institutes of Health. Dr. Baumrin had no disclosures; one coauthor is an employee of CorEvitas, and two others reported grants/adviser fees from several companies, including Janssen, Mallinckrodt, and Pfizer.
A version of this article first appeared on Medscape.com.
“There is value in collecting erythema serially over time as a continuous variable on a scale of 0%-100%” to identify high-risk patients for prophylactic and preemptive treatment, say investigators led by dermatologist Emily Baumrin, MD, director of the GVHD clinic at the University of Pennsylvania, Philadelphia.
They report a study of more than 300 patients with ccGVHD, which found that the extent of skin erythema strongly predicted the risk for death from GVHD.
Of the 267 patients with cutaneous GVHD at baseline, 103 patients died, the majority without a relapse of their blood cancer.
With additional research, erythema body surface area (BSA) should be “introduced as an outcome measure in clinical practice and trials,” they conclude.
At the moment, the NIH Skin Score is commonly used for risk assessment in cutaneous GVHD, but the researchers found that erythema BSA out-predicts this score.
The investigators explain that the NIH Skin Score does incorporate erythema surface area, but it does so as a categorical variable, not a continuous variable. Among other additional factors, it also includes assessments of skin sclerosis, which the investigators found was not associated with GVHD mortality.
Overall, the composite score waters down the weight given to erythema BSA because the score is “driven by stable sclerotic features, and erythema changes are missed,” they explain.
The study was published online in JAMA Dermatology.
Study details
The study included 469 patients with chronic GVHD (cGVHD), of whom 267 (57%) had cutaneous cGVHD at enrollment and 89 (19%) developed skin involvement subsequently.
All of the patients were on systemic immunosuppression for GVHD after allogeneic stem cell transplants for various blood cancers.
They were enrolled from 2007 through 2012 at nine U.S. medical centers – all members of the Chronic Graft Versus Host Disease Consortium – and they were followed until 2018.
Erythema BSA and NIH Skin Score were assessed at baseline and then every 3-6 months. Erythema was the first manifestation of skin involvement in the majority of patients, with a median surface area involvement of 11% at baseline.
The study team found that the extent of erythema at first follow-up visit was associated with both nonrelapse mortality (hazard ratio, 1.33 per 10% BSA increase; P < .001) and overall survival (HR, 1.28 per 10% BSA increase; P < .001), whereas extent of sclerotic skin involvement was not associated with either.
Participants in the study were predominantly White. The investigators note that “BSA assessments of erythema may be less reliable in patients with darker skin.”
The work was funded by the Department of Veterans Affairs and the National Institutes of Health. Dr. Baumrin had no disclosures; one coauthor is an employee of CorEvitas, and two others reported grants/adviser fees from several companies, including Janssen, Mallinckrodt, and Pfizer.
A version of this article first appeared on Medscape.com.
The Evolving Role for Transplantation in Mantle Cell Lymphoma
Mantle cell lymphoma (MCL) has served as a paradigm of progress among the non-Hodgkin lymphomas over the past 30 years. It was originally defined within the Kiel classification as centrocytic lymphoma, then renamed MCL once the characteristic translocation and resulting cyclin D1 overexpression were identified. These diagnostic markers allowed for the characterization of MCL subtypes as well as the initiation of MCL-focused clinical trials which, in turn, led to regulatory approval of more effective regimens, new therapeutic agents, and an improvement in overall survival (OS) from around 3 years to more than 10 years for many patients.
Despite this progress, virtually all patients relapse, and a cure remains elusive for most. In younger (< 65 to 70 years), medically-fit patients who are transplant-eligible and have symptomatic MCL, a standard of care has been induction chemoimmunotherapy containing high-dose cytarabine followed by ASCT consolidation. For example, a clinical trial of R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, prednisone) alternating with R-DHAP (rituximab, dexamethasone, high-dose cytarabine, cisplatin; 3 cycles each) showed a significant benefit over R-CHOP x 6 cycles; at a median follow-up of 10.6 years, the time-to-treatment failure was 8.4 v 3.9 years. In another trial, all patients received induction R-DHAP (with cisplatin or an alternative platinum agent) x 4 cycles followed by ASCT. Those patients randomized to post-ASCT maintenance rituximab for 3 years had significantly improved, 4-year progression-free survival (PFS) as compared with observation only (83% vs 64%, p < 0.001); maintenance also significantly improved OS.
Although ASCT consolidation followed by maintenance became widely adopted on the basis of these and other clinical trials, important questions remain:
First, MCL is biologically and clinically quite heterogeneous. Several prognostic tools such as the MCL International Prognostic Index (MIPI) scoring system and biomarkers are available to define lower- versus higher-risk subtypes, but none is routinely used for treatment planning. About 15% of MCL patients present with a highly-aggressive blastoid or pleomorphic variant that usually carries a TP53 mutation or deletion. Given the short survival and limited benefit from dose-intensive chemotherapy and ASCT in TP53-mutated MCL, should transplant be avoided in these patients?
Second, if deep remission is achieved following front-line therapy, defined as positron emission tomography (PET) negative and measurable residual disease (MRD) negative, will high-dose chemotherapy and ASCT provide additional benefits or only toxicity? This question is being addressed by the ongoing ECOG 4151 study, a risk-adapted trial in which post-induction MRD-negative patients are randomized to standard ASCT consolidation plus maintenance rituximab vs maintenance only.
Bruton tyrosine kinase inhibitors (BTKi) are now among the most used agents for relapsed MCL. Recent clinical trials testing the integration of a BTKi into first- or second-line therapy have shown increased response rates and variable clinical outcomes and toxicities for the combinations, depending upon the chemotherapy- and non-chemotherapy backbones utilized, as well as the BTKi. Combinations with the BCL2 inhibitor venetoclax plus chemotherapy or BTKi are also showing promise.
The activity of BTKi in MCL led the European MCL Network (EMCL) to design the 3-arm TRIANGLE study to analyze the potential of ibrutinib to improve outcomes when given in conjunction with standard ASCT consolidation, and the ability to replace the need for ASCT. The TRIANGLE results were presented by Dr. Martin Dreyling in the Plenary Session at the December 2022 American Society of Hematology (ASH) Annual Meeting. Transplant-eligible MCL patients < 65 years of age were randomized to the EMCL’s established front-line therapy of alternating R-CHOP/R-DHAP plus ASCT; the same regimen plus oral ibrutinib given with the R-CHOP induction cycles and then post-ASCT ibrutinib maintenance therapy for 2 years (Arm A+I); or the A+I regimen minus ASCT (Arm I). Maintenance rituximab was allowed in each arm, on the basis of the treating centers’ institutional guidelines. Overall, 54%-58% of patients in each study arm received rituximab maintenance, with no differential benefit in efficacy noted for those so treated.
The results showed that 94%-98% of patients responded by the end of induction (defined as R-chemo and ASCT), with complete remissions in 36%-45% (from computerized tomography imaging, not PET scan). With a median follow-up of 31 months, failure-free survival (FFS; the primary study endpoint) was significantly improved for A+I vs A (3 year FFS of 88% vs 72%, respectively; p = 0.0008). In a subgroup analysis, FFS was notably improved for A+I in patients with high-level TP53 overexpression by immunohistochemistry. Toxicity did not differ during the induction and ASCT periods among the 3 arms regarding cytopenia, gastrointestinal disorders, and infections. However, neutropenia and infections were increased in the ibrutinib-containing arms during maintenance therapy—especially for Arm A+I.
The authors concluded that ASCT plus ibrutinib (Arm A+I) is superior to ASCT only (Arm A), and that Arm A is not superior to ibrutinib without ASCT (Arm I). No decision can yet be made regarding A+I versus I for which FFS to date remains very similar; however, the authors favor ibrutinib without ASCT due to lower toxicity. OS is trending to favor the ibrutinib arms, but longer follow-up will be needed to fully assess.
Should ASCT consolidation now be replaced by ibrutinib-containing induction R-CHOP/R-DHAP and maintenance ibrutinib, with or without maintenance rituximab? A definitive answer will require the fully-published TRIANGLE results, as well as ongoing analysis with longer follow-up. However, it seems very likely that ASCT indeed will be replaced by the new approach. TP53-mutated MCL should be treated with ibrutinib plus R-CHOP/R-DHAP and ibrutinib maintenance as validated in this trial.
Many centers have begun using a second-generation BTKi, acalabrutinib or zanubrutinib, rather than ibrutinib due to equivalent response rates with more favorable side effect profiles and fewer treatment discontinuations. Caution is warranted regarding simply adding a BTKi to one’s favored MCL induction regimen and foregoing ASCT—pending additional studies and the safety of such alternative approaches.
These are indeed exciting times of therapeutic progress, as they have been improving outcomes and providing longer survival outcomes for MCL patients. Targeted agents facilitate this shift to less intensive and chemotherapy-free regimens that provide enhanced response and mitigate short- and longer-term toxicities. More results will be forthcoming for MRD as a treatment endpoint, guiding maintenance therapy, and for risk-adapted treatment of newly-diagnosed and relapsing patients (based upon MCL subtype and biomarker profiles). Enrolling patients into clinical trials is strongly encouraged as the best mechanism to help answer emerging questions in the field and open the pathway to continued progress.
Mantle cell lymphoma (MCL) has served as a paradigm of progress among the non-Hodgkin lymphomas over the past 30 years. It was originally defined within the Kiel classification as centrocytic lymphoma, then renamed MCL once the characteristic translocation and resulting cyclin D1 overexpression were identified. These diagnostic markers allowed for the characterization of MCL subtypes as well as the initiation of MCL-focused clinical trials which, in turn, led to regulatory approval of more effective regimens, new therapeutic agents, and an improvement in overall survival (OS) from around 3 years to more than 10 years for many patients.
Despite this progress, virtually all patients relapse, and a cure remains elusive for most. In younger (< 65 to 70 years), medically-fit patients who are transplant-eligible and have symptomatic MCL, a standard of care has been induction chemoimmunotherapy containing high-dose cytarabine followed by ASCT consolidation. For example, a clinical trial of R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, prednisone) alternating with R-DHAP (rituximab, dexamethasone, high-dose cytarabine, cisplatin; 3 cycles each) showed a significant benefit over R-CHOP x 6 cycles; at a median follow-up of 10.6 years, the time-to-treatment failure was 8.4 v 3.9 years. In another trial, all patients received induction R-DHAP (with cisplatin or an alternative platinum agent) x 4 cycles followed by ASCT. Those patients randomized to post-ASCT maintenance rituximab for 3 years had significantly improved, 4-year progression-free survival (PFS) as compared with observation only (83% vs 64%, p < 0.001); maintenance also significantly improved OS.
Although ASCT consolidation followed by maintenance became widely adopted on the basis of these and other clinical trials, important questions remain:
First, MCL is biologically and clinically quite heterogeneous. Several prognostic tools such as the MCL International Prognostic Index (MIPI) scoring system and biomarkers are available to define lower- versus higher-risk subtypes, but none is routinely used for treatment planning. About 15% of MCL patients present with a highly-aggressive blastoid or pleomorphic variant that usually carries a TP53 mutation or deletion. Given the short survival and limited benefit from dose-intensive chemotherapy and ASCT in TP53-mutated MCL, should transplant be avoided in these patients?
Second, if deep remission is achieved following front-line therapy, defined as positron emission tomography (PET) negative and measurable residual disease (MRD) negative, will high-dose chemotherapy and ASCT provide additional benefits or only toxicity? This question is being addressed by the ongoing ECOG 4151 study, a risk-adapted trial in which post-induction MRD-negative patients are randomized to standard ASCT consolidation plus maintenance rituximab vs maintenance only.
Bruton tyrosine kinase inhibitors (BTKi) are now among the most used agents for relapsed MCL. Recent clinical trials testing the integration of a BTKi into first- or second-line therapy have shown increased response rates and variable clinical outcomes and toxicities for the combinations, depending upon the chemotherapy- and non-chemotherapy backbones utilized, as well as the BTKi. Combinations with the BCL2 inhibitor venetoclax plus chemotherapy or BTKi are also showing promise.
The activity of BTKi in MCL led the European MCL Network (EMCL) to design the 3-arm TRIANGLE study to analyze the potential of ibrutinib to improve outcomes when given in conjunction with standard ASCT consolidation, and the ability to replace the need for ASCT. The TRIANGLE results were presented by Dr. Martin Dreyling in the Plenary Session at the December 2022 American Society of Hematology (ASH) Annual Meeting. Transplant-eligible MCL patients < 65 years of age were randomized to the EMCL’s established front-line therapy of alternating R-CHOP/R-DHAP plus ASCT; the same regimen plus oral ibrutinib given with the R-CHOP induction cycles and then post-ASCT ibrutinib maintenance therapy for 2 years (Arm A+I); or the A+I regimen minus ASCT (Arm I). Maintenance rituximab was allowed in each arm, on the basis of the treating centers’ institutional guidelines. Overall, 54%-58% of patients in each study arm received rituximab maintenance, with no differential benefit in efficacy noted for those so treated.
The results showed that 94%-98% of patients responded by the end of induction (defined as R-chemo and ASCT), with complete remissions in 36%-45% (from computerized tomography imaging, not PET scan). With a median follow-up of 31 months, failure-free survival (FFS; the primary study endpoint) was significantly improved for A+I vs A (3 year FFS of 88% vs 72%, respectively; p = 0.0008). In a subgroup analysis, FFS was notably improved for A+I in patients with high-level TP53 overexpression by immunohistochemistry. Toxicity did not differ during the induction and ASCT periods among the 3 arms regarding cytopenia, gastrointestinal disorders, and infections. However, neutropenia and infections were increased in the ibrutinib-containing arms during maintenance therapy—especially for Arm A+I.
The authors concluded that ASCT plus ibrutinib (Arm A+I) is superior to ASCT only (Arm A), and that Arm A is not superior to ibrutinib without ASCT (Arm I). No decision can yet be made regarding A+I versus I for which FFS to date remains very similar; however, the authors favor ibrutinib without ASCT due to lower toxicity. OS is trending to favor the ibrutinib arms, but longer follow-up will be needed to fully assess.
Should ASCT consolidation now be replaced by ibrutinib-containing induction R-CHOP/R-DHAP and maintenance ibrutinib, with or without maintenance rituximab? A definitive answer will require the fully-published TRIANGLE results, as well as ongoing analysis with longer follow-up. However, it seems very likely that ASCT indeed will be replaced by the new approach. TP53-mutated MCL should be treated with ibrutinib plus R-CHOP/R-DHAP and ibrutinib maintenance as validated in this trial.
Many centers have begun using a second-generation BTKi, acalabrutinib or zanubrutinib, rather than ibrutinib due to equivalent response rates with more favorable side effect profiles and fewer treatment discontinuations. Caution is warranted regarding simply adding a BTKi to one’s favored MCL induction regimen and foregoing ASCT—pending additional studies and the safety of such alternative approaches.
These are indeed exciting times of therapeutic progress, as they have been improving outcomes and providing longer survival outcomes for MCL patients. Targeted agents facilitate this shift to less intensive and chemotherapy-free regimens that provide enhanced response and mitigate short- and longer-term toxicities. More results will be forthcoming for MRD as a treatment endpoint, guiding maintenance therapy, and for risk-adapted treatment of newly-diagnosed and relapsing patients (based upon MCL subtype and biomarker profiles). Enrolling patients into clinical trials is strongly encouraged as the best mechanism to help answer emerging questions in the field and open the pathway to continued progress.
Mantle cell lymphoma (MCL) has served as a paradigm of progress among the non-Hodgkin lymphomas over the past 30 years. It was originally defined within the Kiel classification as centrocytic lymphoma, then renamed MCL once the characteristic translocation and resulting cyclin D1 overexpression were identified. These diagnostic markers allowed for the characterization of MCL subtypes as well as the initiation of MCL-focused clinical trials which, in turn, led to regulatory approval of more effective regimens, new therapeutic agents, and an improvement in overall survival (OS) from around 3 years to more than 10 years for many patients.
Despite this progress, virtually all patients relapse, and a cure remains elusive for most. In younger (< 65 to 70 years), medically-fit patients who are transplant-eligible and have symptomatic MCL, a standard of care has been induction chemoimmunotherapy containing high-dose cytarabine followed by ASCT consolidation. For example, a clinical trial of R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, prednisone) alternating with R-DHAP (rituximab, dexamethasone, high-dose cytarabine, cisplatin; 3 cycles each) showed a significant benefit over R-CHOP x 6 cycles; at a median follow-up of 10.6 years, the time-to-treatment failure was 8.4 v 3.9 years. In another trial, all patients received induction R-DHAP (with cisplatin or an alternative platinum agent) x 4 cycles followed by ASCT. Those patients randomized to post-ASCT maintenance rituximab for 3 years had significantly improved, 4-year progression-free survival (PFS) as compared with observation only (83% vs 64%, p < 0.001); maintenance also significantly improved OS.
Although ASCT consolidation followed by maintenance became widely adopted on the basis of these and other clinical trials, important questions remain:
First, MCL is biologically and clinically quite heterogeneous. Several prognostic tools such as the MCL International Prognostic Index (MIPI) scoring system and biomarkers are available to define lower- versus higher-risk subtypes, but none is routinely used for treatment planning. About 15% of MCL patients present with a highly-aggressive blastoid or pleomorphic variant that usually carries a TP53 mutation or deletion. Given the short survival and limited benefit from dose-intensive chemotherapy and ASCT in TP53-mutated MCL, should transplant be avoided in these patients?
Second, if deep remission is achieved following front-line therapy, defined as positron emission tomography (PET) negative and measurable residual disease (MRD) negative, will high-dose chemotherapy and ASCT provide additional benefits or only toxicity? This question is being addressed by the ongoing ECOG 4151 study, a risk-adapted trial in which post-induction MRD-negative patients are randomized to standard ASCT consolidation plus maintenance rituximab vs maintenance only.
Bruton tyrosine kinase inhibitors (BTKi) are now among the most used agents for relapsed MCL. Recent clinical trials testing the integration of a BTKi into first- or second-line therapy have shown increased response rates and variable clinical outcomes and toxicities for the combinations, depending upon the chemotherapy- and non-chemotherapy backbones utilized, as well as the BTKi. Combinations with the BCL2 inhibitor venetoclax plus chemotherapy or BTKi are also showing promise.
The activity of BTKi in MCL led the European MCL Network (EMCL) to design the 3-arm TRIANGLE study to analyze the potential of ibrutinib to improve outcomes when given in conjunction with standard ASCT consolidation, and the ability to replace the need for ASCT. The TRIANGLE results were presented by Dr. Martin Dreyling in the Plenary Session at the December 2022 American Society of Hematology (ASH) Annual Meeting. Transplant-eligible MCL patients < 65 years of age were randomized to the EMCL’s established front-line therapy of alternating R-CHOP/R-DHAP plus ASCT; the same regimen plus oral ibrutinib given with the R-CHOP induction cycles and then post-ASCT ibrutinib maintenance therapy for 2 years (Arm A+I); or the A+I regimen minus ASCT (Arm I). Maintenance rituximab was allowed in each arm, on the basis of the treating centers’ institutional guidelines. Overall, 54%-58% of patients in each study arm received rituximab maintenance, with no differential benefit in efficacy noted for those so treated.
The results showed that 94%-98% of patients responded by the end of induction (defined as R-chemo and ASCT), with complete remissions in 36%-45% (from computerized tomography imaging, not PET scan). With a median follow-up of 31 months, failure-free survival (FFS; the primary study endpoint) was significantly improved for A+I vs A (3 year FFS of 88% vs 72%, respectively; p = 0.0008). In a subgroup analysis, FFS was notably improved for A+I in patients with high-level TP53 overexpression by immunohistochemistry. Toxicity did not differ during the induction and ASCT periods among the 3 arms regarding cytopenia, gastrointestinal disorders, and infections. However, neutropenia and infections were increased in the ibrutinib-containing arms during maintenance therapy—especially for Arm A+I.
The authors concluded that ASCT plus ibrutinib (Arm A+I) is superior to ASCT only (Arm A), and that Arm A is not superior to ibrutinib without ASCT (Arm I). No decision can yet be made regarding A+I versus I for which FFS to date remains very similar; however, the authors favor ibrutinib without ASCT due to lower toxicity. OS is trending to favor the ibrutinib arms, but longer follow-up will be needed to fully assess.
Should ASCT consolidation now be replaced by ibrutinib-containing induction R-CHOP/R-DHAP and maintenance ibrutinib, with or without maintenance rituximab? A definitive answer will require the fully-published TRIANGLE results, as well as ongoing analysis with longer follow-up. However, it seems very likely that ASCT indeed will be replaced by the new approach. TP53-mutated MCL should be treated with ibrutinib plus R-CHOP/R-DHAP and ibrutinib maintenance as validated in this trial.
Many centers have begun using a second-generation BTKi, acalabrutinib or zanubrutinib, rather than ibrutinib due to equivalent response rates with more favorable side effect profiles and fewer treatment discontinuations. Caution is warranted regarding simply adding a BTKi to one’s favored MCL induction regimen and foregoing ASCT—pending additional studies and the safety of such alternative approaches.
These are indeed exciting times of therapeutic progress, as they have been improving outcomes and providing longer survival outcomes for MCL patients. Targeted agents facilitate this shift to less intensive and chemotherapy-free regimens that provide enhanced response and mitigate short- and longer-term toxicities. More results will be forthcoming for MRD as a treatment endpoint, guiding maintenance therapy, and for risk-adapted treatment of newly-diagnosed and relapsing patients (based upon MCL subtype and biomarker profiles). Enrolling patients into clinical trials is strongly encouraged as the best mechanism to help answer emerging questions in the field and open the pathway to continued progress.
Intermittent abdominal pain
Mantle cell lymphoma (MCL) is an aggressive form of non-Hodgkin lymphoma characterized by the proliferation of CD5-positive B cells within the mantle zone that surrounds normal germinal center follicles. MCL is a rare disease that most commonly presents in adult men (male to female ratio > 2:1) in the fifth and sixth decades of life. Individuals diagnosed with MCL typically present with constitutional symptoms, such as weight loss, night sweats, persistent fever, and fatigue. Approximately 25% of cases present with extranodal involvement with the bone marrow; peripheral blood and gastrointestinal tract are most often involved. In patients with extensive node involvement in the gastrointestinal tract, additional symptoms at presentation often include abdominal pain, abdominal fullness, and bloating. Skin involvement in MCL is rare and usually indicates widespread disease.
According to the guidelines of the World Health Organization–European Organization for Research and Treatment of Cancer, a diagnosis of MCL is established on the basis of the morphologic examination findings and immunophenotyping.
Immunohistochemically, expression of cyclin D1 in normal lymphoid cells is very low and often undetectable; only hairy cell leukemia shows moderate expression of cyclin D1. Therefore, positive immunohistochemistry for cyclin D1 is pathognomonic for MCL. Increased expression of cyclin D1 protein leads to dysregulation of the cell cycle and stimulates uncontrolled cell proliferation. It is also indirect evidence of the chromosomal translocation (11;14)(q13;q32) on the CCND1 gene, which is detected in 95% of cases of MCL. In addition, negative expression of antigens may also help to differentiate MCL from other lymphomas. MCL does not usually express the antigens that are associated with germinal centers, such as CD10, CD23, and BCL6. Thus, these antigens can be used to distinguish MCL from B-cell lymphomas of germinal center origin, including follicular lymphoma, Burkitt lymphoma, and diffuse large B-cell lymphoma.
The National Comprehensive Cancer Network recommends chemotherapy followed by radiation for stage I or II disease. In general, patients with advanced-stage disease benefit from systemic chemotherapy. Because MCL is clinically heterogeneous, treatment may require adjustment on the basis of the patient's age, underlying comorbidities, and underlying MCL biology such as TP53 mutations. During induction therapy, prophylaxis and monitoring for tumor lysis syndrome is strongly recommended to be considered. Before treatment, hepatitis B virus testing is recommended because of an increased risk for viral reactivation with use of immunotherapy regimens for treatment.
Timothy J. Voorhees, MD, MSCR, Assistant Professor of Internal Medicine - Clinical, Division of Hematology, The Ohio State University James Comprehensive Cancer Center, Columbus, OH.
Timothy J. Voorhees, MD, MSCR, has disclosed the following relevant financial relationships:
Received research grant from: AstraZeneca; Morphosys; Incyte; Recordati.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
Mantle cell lymphoma (MCL) is an aggressive form of non-Hodgkin lymphoma characterized by the proliferation of CD5-positive B cells within the mantle zone that surrounds normal germinal center follicles. MCL is a rare disease that most commonly presents in adult men (male to female ratio > 2:1) in the fifth and sixth decades of life. Individuals diagnosed with MCL typically present with constitutional symptoms, such as weight loss, night sweats, persistent fever, and fatigue. Approximately 25% of cases present with extranodal involvement with the bone marrow; peripheral blood and gastrointestinal tract are most often involved. In patients with extensive node involvement in the gastrointestinal tract, additional symptoms at presentation often include abdominal pain, abdominal fullness, and bloating. Skin involvement in MCL is rare and usually indicates widespread disease.
According to the guidelines of the World Health Organization–European Organization for Research and Treatment of Cancer, a diagnosis of MCL is established on the basis of the morphologic examination findings and immunophenotyping.
Immunohistochemically, expression of cyclin D1 in normal lymphoid cells is very low and often undetectable; only hairy cell leukemia shows moderate expression of cyclin D1. Therefore, positive immunohistochemistry for cyclin D1 is pathognomonic for MCL. Increased expression of cyclin D1 protein leads to dysregulation of the cell cycle and stimulates uncontrolled cell proliferation. It is also indirect evidence of the chromosomal translocation (11;14)(q13;q32) on the CCND1 gene, which is detected in 95% of cases of MCL. In addition, negative expression of antigens may also help to differentiate MCL from other lymphomas. MCL does not usually express the antigens that are associated with germinal centers, such as CD10, CD23, and BCL6. Thus, these antigens can be used to distinguish MCL from B-cell lymphomas of germinal center origin, including follicular lymphoma, Burkitt lymphoma, and diffuse large B-cell lymphoma.
The National Comprehensive Cancer Network recommends chemotherapy followed by radiation for stage I or II disease. In general, patients with advanced-stage disease benefit from systemic chemotherapy. Because MCL is clinically heterogeneous, treatment may require adjustment on the basis of the patient's age, underlying comorbidities, and underlying MCL biology such as TP53 mutations. During induction therapy, prophylaxis and monitoring for tumor lysis syndrome is strongly recommended to be considered. Before treatment, hepatitis B virus testing is recommended because of an increased risk for viral reactivation with use of immunotherapy regimens for treatment.
Timothy J. Voorhees, MD, MSCR, Assistant Professor of Internal Medicine - Clinical, Division of Hematology, The Ohio State University James Comprehensive Cancer Center, Columbus, OH.
Timothy J. Voorhees, MD, MSCR, has disclosed the following relevant financial relationships:
Received research grant from: AstraZeneca; Morphosys; Incyte; Recordati.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
Mantle cell lymphoma (MCL) is an aggressive form of non-Hodgkin lymphoma characterized by the proliferation of CD5-positive B cells within the mantle zone that surrounds normal germinal center follicles. MCL is a rare disease that most commonly presents in adult men (male to female ratio > 2:1) in the fifth and sixth decades of life. Individuals diagnosed with MCL typically present with constitutional symptoms, such as weight loss, night sweats, persistent fever, and fatigue. Approximately 25% of cases present with extranodal involvement with the bone marrow; peripheral blood and gastrointestinal tract are most often involved. In patients with extensive node involvement in the gastrointestinal tract, additional symptoms at presentation often include abdominal pain, abdominal fullness, and bloating. Skin involvement in MCL is rare and usually indicates widespread disease.
According to the guidelines of the World Health Organization–European Organization for Research and Treatment of Cancer, a diagnosis of MCL is established on the basis of the morphologic examination findings and immunophenotyping.
Immunohistochemically, expression of cyclin D1 in normal lymphoid cells is very low and often undetectable; only hairy cell leukemia shows moderate expression of cyclin D1. Therefore, positive immunohistochemistry for cyclin D1 is pathognomonic for MCL. Increased expression of cyclin D1 protein leads to dysregulation of the cell cycle and stimulates uncontrolled cell proliferation. It is also indirect evidence of the chromosomal translocation (11;14)(q13;q32) on the CCND1 gene, which is detected in 95% of cases of MCL. In addition, negative expression of antigens may also help to differentiate MCL from other lymphomas. MCL does not usually express the antigens that are associated with germinal centers, such as CD10, CD23, and BCL6. Thus, these antigens can be used to distinguish MCL from B-cell lymphomas of germinal center origin, including follicular lymphoma, Burkitt lymphoma, and diffuse large B-cell lymphoma.
The National Comprehensive Cancer Network recommends chemotherapy followed by radiation for stage I or II disease. In general, patients with advanced-stage disease benefit from systemic chemotherapy. Because MCL is clinically heterogeneous, treatment may require adjustment on the basis of the patient's age, underlying comorbidities, and underlying MCL biology such as TP53 mutations. During induction therapy, prophylaxis and monitoring for tumor lysis syndrome is strongly recommended to be considered. Before treatment, hepatitis B virus testing is recommended because of an increased risk for viral reactivation with use of immunotherapy regimens for treatment.
Timothy J. Voorhees, MD, MSCR, Assistant Professor of Internal Medicine - Clinical, Division of Hematology, The Ohio State University James Comprehensive Cancer Center, Columbus, OH.
Timothy J. Voorhees, MD, MSCR, has disclosed the following relevant financial relationships:
Received research grant from: AstraZeneca; Morphosys; Incyte; Recordati.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
A 69-year-old man presents for an evaluation for a 3-month history of generalized intermittent abdominal pain with occasional dark blood with bowel movements. He does not report experiencing any fever, chills, diarrhea, or obstructive symptoms. However, he does note a 10-lb weight loss over the past few months. He underwent routine screening colonoscopy 5 years ago, which was unremarkable. Complete blood count reveals normocytic anemia. CT of the chest, abdomen, and pelvis demonstrated extensive mesenteric lymphadenopathy and bilateral axillary lymphadenopathy. Physical examination reveals an enlarged right inguinal lymph node, diffuse cutaneous erythematous plaques, and nodules with irregular borders on the upper back. Lesion diameters range from 0.5 to 1.5 cm, with the largest having a central ulceration.
A biopsy of one of the skin lesions was performed. Histopathologic examination demonstrated diffuse lymphoid infiltrate composed predominately of small, mature lymphocytes. Immunohistochemistry showed expression of cyclin D1, CD5, CD20, SOX11, and BCL2. Lesions were negative for CD10, CD23, and BCL6.