User login
Official Newspaper of the American College of Surgeons
Winners and losers under bold Trump plan to slash drug rebate deals
Few consumers have heard of the secret, business-to-business payments that the Trump administration wants to ban in an attempt to control drug costs.
But the administration’s plan for drug rebates, announced Jan. 31, would end the pharmaceutical business as usual, shift billions in revenue and cause far-reaching, unforeseen change, say health policy authorities.
In pointed language sure to anger middlemen who benefit from the deals, administration officials proposed banning rebates paid by drug companies to ensure coverage for their products under Medicare and Medicaid plans.
“A shadowy system of kickbacks,” was how Health and Human Services Secretary Alex Azar described the current system in a speech on Feb. 1.
The proposal is a regulatory change applying only to Medicare plans for seniors and managed Medicaid plans for low-income people. But private insurers, who often take cues from government programs, might make a similar shift, administration officials said.
Drug rebates are essentially discounts off the list price. Outlawing them would divert $29 billion in rebates now paid to insurers and pharmacy benefit managers into “seniors’ pocketbooks at the pharmacy counter,” Azar said.
The measure already faces fierce opposition from some in the industry and is unlikely to be implemented as presented or by the proposed 2020 effective date, health policy analysts said.
In any event, it’s hardly a pure win for seniors or patients in general. Consumers are unlikely to collect the full benefit of eliminated rebates.
At the same time, the change would produce uncertain ricochets, including higher drug-plan premiums for consumers, that would produce new winners and losers across the economy.
“It is the most significant proposal that the administration has introduced so far” to try to control drug prices, said Rachel Sachs, a law professor at Washington University in St. Louis. “But I’m struck by the uncertainty that the administration has in what the effects would be.”
Chronically ill patients who take lots of expensive medicine
The list price for many brand-name medicines has doubled or tripled in recent years. But virtually the only ones affected by the full increases are the many patients who pay cash or whose out-of-pocket payments are based on the posted price.
By banning rebates, the administration says its intention is to ensure discounts are passed all the way to the patient instead of the middlemen, the so-called pharmacy benefit managers or PBMs. That means consumers using expensive drugs might see their out-of-pocket costs go down.
If rebates were eliminated for commercial insurance, where deductibles and out-of-pocket costs are generally much higher, chronically ill patients could benefit much more.
Drug companies
Ending rebates would give the administration a drug-policy “win” that doesn’t directly threaten pharmaceutical company profits.
“We applaud the administration for taking steps to reform the rebate system” Stephen Ubl, CEO of PhRMA, the main lobby for branded drugs, said after the proposal came out.
The change might also slow the soaring list-price increases that have become a publicity nightmare for the industry. When list prices pop by 5% or 10% each year, drugmakers pay part of the proceeds to insurers and PBMs in the form of rebates to guarantee health-plan coverage.
No one is claiming that eliminating rebates would stop escalating list prices, even if all insurers adopted the practice. But some believe it would remove an important factor.
Pharmacy benefit managers
PBMs reap billions of dollars in rebate revenue in return for putting particular products on lists of covered drugs. The administration is essentially proposing to make those payments illegal, at least for Medicare and Medicaid plans.
PBMs, which claim they control costs by negotiating with drugmakers, might have to go back to their roots – processing pharmacy claims for a fee. After recent industry consolidation into a few enormous companies, on the other hand, they might have the market power to charge very high fees, replacing much of the lost rebate revenue.
PBMs “are concerned” that the move “would increase drug costs and force Medicare beneficiaries to pay higher premiums and out-of-pocket expenses,” said JC Scott, CEO of the Pharmaceutical Care Management Association, the PBM lobby.
Insurance companies
Insurers, who often receive rebates directly, could also be hurt financially.
“From the start, the focus on rebates has been a distraction from the real issue – the problem is the price” of the drugs, said Matt Eyles, CEO of America’s Health Insurance Plans, a trade group. “We are not middlemen – we are your bargaining power, working hard to negotiate lower prices.”
Patients without chronic conditions and high drug costs
Lower out-of-pocket costs at the pharmacy counter would be financed, at least in part, by higher premiums for Medicare and Medicaid plans paid by consumers and the government. Premiums for Medicare Part D plans could rise from $3.20 to $5.64 per month, according to consultants hired by the Department of Health and Human Services.
“There is likely to be a wide variation in how much savings people see based on the drugs they take and the point-of-sale discounts that are negotiated,” said Elizabeth Carpenter, policy practice director at Avalere, a consultancy.
Consumers who don’t need expensive drugs every month could see insurance costs go up slightly without getting the benefits of lower out-of-pocket expense for purchased drugs.
Other policy changes giving health plans more negotiating power against drugmakers would keep a lid on premium increases, administration officials argue.
Kaiser Health News is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
Few consumers have heard of the secret, business-to-business payments that the Trump administration wants to ban in an attempt to control drug costs.
But the administration’s plan for drug rebates, announced Jan. 31, would end the pharmaceutical business as usual, shift billions in revenue and cause far-reaching, unforeseen change, say health policy authorities.
In pointed language sure to anger middlemen who benefit from the deals, administration officials proposed banning rebates paid by drug companies to ensure coverage for their products under Medicare and Medicaid plans.
“A shadowy system of kickbacks,” was how Health and Human Services Secretary Alex Azar described the current system in a speech on Feb. 1.
The proposal is a regulatory change applying only to Medicare plans for seniors and managed Medicaid plans for low-income people. But private insurers, who often take cues from government programs, might make a similar shift, administration officials said.
Drug rebates are essentially discounts off the list price. Outlawing them would divert $29 billion in rebates now paid to insurers and pharmacy benefit managers into “seniors’ pocketbooks at the pharmacy counter,” Azar said.
The measure already faces fierce opposition from some in the industry and is unlikely to be implemented as presented or by the proposed 2020 effective date, health policy analysts said.
In any event, it’s hardly a pure win for seniors or patients in general. Consumers are unlikely to collect the full benefit of eliminated rebates.
At the same time, the change would produce uncertain ricochets, including higher drug-plan premiums for consumers, that would produce new winners and losers across the economy.
“It is the most significant proposal that the administration has introduced so far” to try to control drug prices, said Rachel Sachs, a law professor at Washington University in St. Louis. “But I’m struck by the uncertainty that the administration has in what the effects would be.”
Chronically ill patients who take lots of expensive medicine
The list price for many brand-name medicines has doubled or tripled in recent years. But virtually the only ones affected by the full increases are the many patients who pay cash or whose out-of-pocket payments are based on the posted price.
By banning rebates, the administration says its intention is to ensure discounts are passed all the way to the patient instead of the middlemen, the so-called pharmacy benefit managers or PBMs. That means consumers using expensive drugs might see their out-of-pocket costs go down.
If rebates were eliminated for commercial insurance, where deductibles and out-of-pocket costs are generally much higher, chronically ill patients could benefit much more.
Drug companies
Ending rebates would give the administration a drug-policy “win” that doesn’t directly threaten pharmaceutical company profits.
“We applaud the administration for taking steps to reform the rebate system” Stephen Ubl, CEO of PhRMA, the main lobby for branded drugs, said after the proposal came out.
The change might also slow the soaring list-price increases that have become a publicity nightmare for the industry. When list prices pop by 5% or 10% each year, drugmakers pay part of the proceeds to insurers and PBMs in the form of rebates to guarantee health-plan coverage.
No one is claiming that eliminating rebates would stop escalating list prices, even if all insurers adopted the practice. But some believe it would remove an important factor.
Pharmacy benefit managers
PBMs reap billions of dollars in rebate revenue in return for putting particular products on lists of covered drugs. The administration is essentially proposing to make those payments illegal, at least for Medicare and Medicaid plans.
PBMs, which claim they control costs by negotiating with drugmakers, might have to go back to their roots – processing pharmacy claims for a fee. After recent industry consolidation into a few enormous companies, on the other hand, they might have the market power to charge very high fees, replacing much of the lost rebate revenue.
PBMs “are concerned” that the move “would increase drug costs and force Medicare beneficiaries to pay higher premiums and out-of-pocket expenses,” said JC Scott, CEO of the Pharmaceutical Care Management Association, the PBM lobby.
Insurance companies
Insurers, who often receive rebates directly, could also be hurt financially.
“From the start, the focus on rebates has been a distraction from the real issue – the problem is the price” of the drugs, said Matt Eyles, CEO of America’s Health Insurance Plans, a trade group. “We are not middlemen – we are your bargaining power, working hard to negotiate lower prices.”
Patients without chronic conditions and high drug costs
Lower out-of-pocket costs at the pharmacy counter would be financed, at least in part, by higher premiums for Medicare and Medicaid plans paid by consumers and the government. Premiums for Medicare Part D plans could rise from $3.20 to $5.64 per month, according to consultants hired by the Department of Health and Human Services.
“There is likely to be a wide variation in how much savings people see based on the drugs they take and the point-of-sale discounts that are negotiated,” said Elizabeth Carpenter, policy practice director at Avalere, a consultancy.
Consumers who don’t need expensive drugs every month could see insurance costs go up slightly without getting the benefits of lower out-of-pocket expense for purchased drugs.
Other policy changes giving health plans more negotiating power against drugmakers would keep a lid on premium increases, administration officials argue.
Kaiser Health News is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
Few consumers have heard of the secret, business-to-business payments that the Trump administration wants to ban in an attempt to control drug costs.
But the administration’s plan for drug rebates, announced Jan. 31, would end the pharmaceutical business as usual, shift billions in revenue and cause far-reaching, unforeseen change, say health policy authorities.
In pointed language sure to anger middlemen who benefit from the deals, administration officials proposed banning rebates paid by drug companies to ensure coverage for their products under Medicare and Medicaid plans.
“A shadowy system of kickbacks,” was how Health and Human Services Secretary Alex Azar described the current system in a speech on Feb. 1.
The proposal is a regulatory change applying only to Medicare plans for seniors and managed Medicaid plans for low-income people. But private insurers, who often take cues from government programs, might make a similar shift, administration officials said.
Drug rebates are essentially discounts off the list price. Outlawing them would divert $29 billion in rebates now paid to insurers and pharmacy benefit managers into “seniors’ pocketbooks at the pharmacy counter,” Azar said.
The measure already faces fierce opposition from some in the industry and is unlikely to be implemented as presented or by the proposed 2020 effective date, health policy analysts said.
In any event, it’s hardly a pure win for seniors or patients in general. Consumers are unlikely to collect the full benefit of eliminated rebates.
At the same time, the change would produce uncertain ricochets, including higher drug-plan premiums for consumers, that would produce new winners and losers across the economy.
“It is the most significant proposal that the administration has introduced so far” to try to control drug prices, said Rachel Sachs, a law professor at Washington University in St. Louis. “But I’m struck by the uncertainty that the administration has in what the effects would be.”
Chronically ill patients who take lots of expensive medicine
The list price for many brand-name medicines has doubled or tripled in recent years. But virtually the only ones affected by the full increases are the many patients who pay cash or whose out-of-pocket payments are based on the posted price.
By banning rebates, the administration says its intention is to ensure discounts are passed all the way to the patient instead of the middlemen, the so-called pharmacy benefit managers or PBMs. That means consumers using expensive drugs might see their out-of-pocket costs go down.
If rebates were eliminated for commercial insurance, where deductibles and out-of-pocket costs are generally much higher, chronically ill patients could benefit much more.
Drug companies
Ending rebates would give the administration a drug-policy “win” that doesn’t directly threaten pharmaceutical company profits.
“We applaud the administration for taking steps to reform the rebate system” Stephen Ubl, CEO of PhRMA, the main lobby for branded drugs, said after the proposal came out.
The change might also slow the soaring list-price increases that have become a publicity nightmare for the industry. When list prices pop by 5% or 10% each year, drugmakers pay part of the proceeds to insurers and PBMs in the form of rebates to guarantee health-plan coverage.
No one is claiming that eliminating rebates would stop escalating list prices, even if all insurers adopted the practice. But some believe it would remove an important factor.
Pharmacy benefit managers
PBMs reap billions of dollars in rebate revenue in return for putting particular products on lists of covered drugs. The administration is essentially proposing to make those payments illegal, at least for Medicare and Medicaid plans.
PBMs, which claim they control costs by negotiating with drugmakers, might have to go back to their roots – processing pharmacy claims for a fee. After recent industry consolidation into a few enormous companies, on the other hand, they might have the market power to charge very high fees, replacing much of the lost rebate revenue.
PBMs “are concerned” that the move “would increase drug costs and force Medicare beneficiaries to pay higher premiums and out-of-pocket expenses,” said JC Scott, CEO of the Pharmaceutical Care Management Association, the PBM lobby.
Insurance companies
Insurers, who often receive rebates directly, could also be hurt financially.
“From the start, the focus on rebates has been a distraction from the real issue – the problem is the price” of the drugs, said Matt Eyles, CEO of America’s Health Insurance Plans, a trade group. “We are not middlemen – we are your bargaining power, working hard to negotiate lower prices.”
Patients without chronic conditions and high drug costs
Lower out-of-pocket costs at the pharmacy counter would be financed, at least in part, by higher premiums for Medicare and Medicaid plans paid by consumers and the government. Premiums for Medicare Part D plans could rise from $3.20 to $5.64 per month, according to consultants hired by the Department of Health and Human Services.
“There is likely to be a wide variation in how much savings people see based on the drugs they take and the point-of-sale discounts that are negotiated,” said Elizabeth Carpenter, policy practice director at Avalere, a consultancy.
Consumers who don’t need expensive drugs every month could see insurance costs go up slightly without getting the benefits of lower out-of-pocket expense for purchased drugs.
Other policy changes giving health plans more negotiating power against drugmakers would keep a lid on premium increases, administration officials argue.
Kaiser Health News is a nonprofit national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
Gastrectomy does not alter benefit of new oral chemo in gastric cancer
SAN FRANCISCO – suggests a preplanned subgroup analysis of the global phase 3 randomized controlled TAGS trial. Results were reported at the 2019 GI Cancers Symposium.
“The standard of care for early-stage gastric cancer is surgery, which is the only potentially curative treatment,” noted lead investigator David H. Ilson, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York. “Forty percent of patients with metastatic disease have had a history of previous gastrectomy.”
The TAGS trial assessed efficacy of combined trifluridine and tipiracil (Lonsurf) among 507 patients with metastatic gastric or gastroesophageal junction cancer who had received at least two prior chemotherapy regimens. (This combination chemotherapy is currently approved in the United States as later-line therapy for metastatic colorectal cancer.)
Compared with placebo, trifluridine/tipiracil prolonged overall survival by 2.6 months in the subgroup who had previously undergone gastrectomy, a benefit slightly greater than the 2.1 months previously reported for the entire trial population (Lancet Oncol. 2018;19:1437-48). And although the gastrectomy subgroup experienced more grade 3 or 4 adverse events, they were not more likely to stop treatment because of toxicity.
“The data from this analysis reinforce the benefit for trifluridine/tipiracil as prolonging survival versus placebo, and this is regardless of prior gastrectomy,” Dr. Ilson summarized. “Hematologic adverse events, such as neutropenia and leukopenia, may have been somewhat more frequent among the trifluridine/tipiracil–treated patients with gastrectomy than in the overall population, but this did not result in more treatment discontinuations. Exposure to the drug was similar between patients with gastrectomy and those in the overall population.”
“Trifluridine/tipiracil is an effective treatment option with a manageable toxicity profile for patients with metastatic gastric cancer, regardless of prior gastrectomy status,” he concluded.
Still fit for treatment
The disconnect between toxicity and treatment discontinuation seen in the TAGS trial is not new, according to invited discussant Martine Extermann, MD, PhD, leader of the Senior Adult Oncology Program at the Moffitt Cancer Center in Tampa. Previous data among geriatric cancer patients have similarly shown that, despite substantial chemotherapy toxicity, by and large, there are only modest effects on health-related quality of life, performance status, and instrumental activities of daily living, she noted.
“The CTCAE [Common Terminology Criteria for Adverse Events] toxicity differences do not always translate into functional impact and treatment cessation. So this is only part of the picture. It’s a convenient part. It’s easily measurable. It’s well acknowledged as a measurement of side effects. But it does not tell the whole story. Quality of life and functional status add to the picture,” Dr. Extermann elaborated. “What the TAGS study is telling us is, despite a gastrectomy, these patients can be treated as a third-line treatment population for gastric cancer, which is not necessarily obvious to every oncologist.”
At the same time, she added that it would be helpful to have nutritional data on the study patients – and on all patients in similar trials, for that matter – because nutritional status is one of the components of the CRASH (Chemotherapy Risk Assessment Scale for High-Age Patients) score used to predict chemotherapy toxicity in older adults.
“I would support that [trifluridine/tipiracil] is an effective third-line chemotherapy in gastric cancer patients with or without prior gastrectomy, and this can be given safely,” Dr. Extermann concluded.
Study details
Patients in TAGS were randomized 2:1 to trifluridine/tipiracil (formerly TAS-102) or placebo, each added to best supportive care. (Trifluridine is a novel oral thymidine analogue, and tipiracil prevents trifluridine degradation.) Overall, 44% had undergone gastrectomy before entering the trial.
In the entire trial population, overall survival was 5.7 months with trifluridine/tipiracil and 3.6 months with placebo (hazard ratio, 0.69; 95% confidence interval, 0.56-0.85; P = .0006), as previously reported.
Among the subgroup who had undergone prior gastrectomy, overall survival was 6.0 months with trifluridine/tipiracil and 3.4 months with placebo (HR, 0.57; 95% CI, 0.41-0.79), Dr. Ilson reported at the symposium, which is sponsored by the American Gastroenterological Association, the American Society for Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology. The combination also netted better progression-free survival (2.2 vs. 1.8 months; HR, 0.48; 95% CI, 0.35-0.65). “These data mirror the data seen in the overall treatment population,” he commented.
In a multivariate analysis including all prespecified factors, prior gastrectomy was neither prognostic nor predictive. Moreover, the treatment effect size remained the same after adjustment for potential prognostic factors.
Exposure to trifluridine/tipiracil was similar for the gastrectomy subgroup and the entire trial population in terms of relative dose intensity, median number of cycles, and treatment duration.
The rate of grade 3 or 4 treatment-related adverse events with trifluridine/tipiracil in the gastrectomy subgroup, 64%, was higher than that in the entire trial population, 53%, but the rate of discontinuation because of any-grade adverse events was similar, at 10% and 13%, respectively.
The difference in grade 3 or 4 adverse events between the gastrectomy subgroup and the entire trial population was mainly driven by higher rates of neutropenia (44% vs. 34%) and leukopenia (14% vs. 9%) in the former.
Dr. Ilson disclosed that he has a consulting role with Amgen, Astellas, AstraZeneca, Bayer, Bristol-Myers Squibb, Lilly, Merck, Pieris, Roche/Genentech, and Taiho and that he receives research support from Taiho. The study was funded by Taiho Oncology and Taiho Pharmaceutical.
SOURCE: Ilson DH et al. 2019 GI Cancers Symposium, Abstract 3.
SAN FRANCISCO – suggests a preplanned subgroup analysis of the global phase 3 randomized controlled TAGS trial. Results were reported at the 2019 GI Cancers Symposium.
“The standard of care for early-stage gastric cancer is surgery, which is the only potentially curative treatment,” noted lead investigator David H. Ilson, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York. “Forty percent of patients with metastatic disease have had a history of previous gastrectomy.”
The TAGS trial assessed efficacy of combined trifluridine and tipiracil (Lonsurf) among 507 patients with metastatic gastric or gastroesophageal junction cancer who had received at least two prior chemotherapy regimens. (This combination chemotherapy is currently approved in the United States as later-line therapy for metastatic colorectal cancer.)
Compared with placebo, trifluridine/tipiracil prolonged overall survival by 2.6 months in the subgroup who had previously undergone gastrectomy, a benefit slightly greater than the 2.1 months previously reported for the entire trial population (Lancet Oncol. 2018;19:1437-48). And although the gastrectomy subgroup experienced more grade 3 or 4 adverse events, they were not more likely to stop treatment because of toxicity.
“The data from this analysis reinforce the benefit for trifluridine/tipiracil as prolonging survival versus placebo, and this is regardless of prior gastrectomy,” Dr. Ilson summarized. “Hematologic adverse events, such as neutropenia and leukopenia, may have been somewhat more frequent among the trifluridine/tipiracil–treated patients with gastrectomy than in the overall population, but this did not result in more treatment discontinuations. Exposure to the drug was similar between patients with gastrectomy and those in the overall population.”
“Trifluridine/tipiracil is an effective treatment option with a manageable toxicity profile for patients with metastatic gastric cancer, regardless of prior gastrectomy status,” he concluded.
Still fit for treatment
The disconnect between toxicity and treatment discontinuation seen in the TAGS trial is not new, according to invited discussant Martine Extermann, MD, PhD, leader of the Senior Adult Oncology Program at the Moffitt Cancer Center in Tampa. Previous data among geriatric cancer patients have similarly shown that, despite substantial chemotherapy toxicity, by and large, there are only modest effects on health-related quality of life, performance status, and instrumental activities of daily living, she noted.
“The CTCAE [Common Terminology Criteria for Adverse Events] toxicity differences do not always translate into functional impact and treatment cessation. So this is only part of the picture. It’s a convenient part. It’s easily measurable. It’s well acknowledged as a measurement of side effects. But it does not tell the whole story. Quality of life and functional status add to the picture,” Dr. Extermann elaborated. “What the TAGS study is telling us is, despite a gastrectomy, these patients can be treated as a third-line treatment population for gastric cancer, which is not necessarily obvious to every oncologist.”
At the same time, she added that it would be helpful to have nutritional data on the study patients – and on all patients in similar trials, for that matter – because nutritional status is one of the components of the CRASH (Chemotherapy Risk Assessment Scale for High-Age Patients) score used to predict chemotherapy toxicity in older adults.
“I would support that [trifluridine/tipiracil] is an effective third-line chemotherapy in gastric cancer patients with or without prior gastrectomy, and this can be given safely,” Dr. Extermann concluded.
Study details
Patients in TAGS were randomized 2:1 to trifluridine/tipiracil (formerly TAS-102) or placebo, each added to best supportive care. (Trifluridine is a novel oral thymidine analogue, and tipiracil prevents trifluridine degradation.) Overall, 44% had undergone gastrectomy before entering the trial.
In the entire trial population, overall survival was 5.7 months with trifluridine/tipiracil and 3.6 months with placebo (hazard ratio, 0.69; 95% confidence interval, 0.56-0.85; P = .0006), as previously reported.
Among the subgroup who had undergone prior gastrectomy, overall survival was 6.0 months with trifluridine/tipiracil and 3.4 months with placebo (HR, 0.57; 95% CI, 0.41-0.79), Dr. Ilson reported at the symposium, which is sponsored by the American Gastroenterological Association, the American Society for Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology. The combination also netted better progression-free survival (2.2 vs. 1.8 months; HR, 0.48; 95% CI, 0.35-0.65). “These data mirror the data seen in the overall treatment population,” he commented.
In a multivariate analysis including all prespecified factors, prior gastrectomy was neither prognostic nor predictive. Moreover, the treatment effect size remained the same after adjustment for potential prognostic factors.
Exposure to trifluridine/tipiracil was similar for the gastrectomy subgroup and the entire trial population in terms of relative dose intensity, median number of cycles, and treatment duration.
The rate of grade 3 or 4 treatment-related adverse events with trifluridine/tipiracil in the gastrectomy subgroup, 64%, was higher than that in the entire trial population, 53%, but the rate of discontinuation because of any-grade adverse events was similar, at 10% and 13%, respectively.
The difference in grade 3 or 4 adverse events between the gastrectomy subgroup and the entire trial population was mainly driven by higher rates of neutropenia (44% vs. 34%) and leukopenia (14% vs. 9%) in the former.
Dr. Ilson disclosed that he has a consulting role with Amgen, Astellas, AstraZeneca, Bayer, Bristol-Myers Squibb, Lilly, Merck, Pieris, Roche/Genentech, and Taiho and that he receives research support from Taiho. The study was funded by Taiho Oncology and Taiho Pharmaceutical.
SOURCE: Ilson DH et al. 2019 GI Cancers Symposium, Abstract 3.
SAN FRANCISCO – suggests a preplanned subgroup analysis of the global phase 3 randomized controlled TAGS trial. Results were reported at the 2019 GI Cancers Symposium.
“The standard of care for early-stage gastric cancer is surgery, which is the only potentially curative treatment,” noted lead investigator David H. Ilson, MD, PhD, of Memorial Sloan Kettering Cancer Center in New York. “Forty percent of patients with metastatic disease have had a history of previous gastrectomy.”
The TAGS trial assessed efficacy of combined trifluridine and tipiracil (Lonsurf) among 507 patients with metastatic gastric or gastroesophageal junction cancer who had received at least two prior chemotherapy regimens. (This combination chemotherapy is currently approved in the United States as later-line therapy for metastatic colorectal cancer.)
Compared with placebo, trifluridine/tipiracil prolonged overall survival by 2.6 months in the subgroup who had previously undergone gastrectomy, a benefit slightly greater than the 2.1 months previously reported for the entire trial population (Lancet Oncol. 2018;19:1437-48). And although the gastrectomy subgroup experienced more grade 3 or 4 adverse events, they were not more likely to stop treatment because of toxicity.
“The data from this analysis reinforce the benefit for trifluridine/tipiracil as prolonging survival versus placebo, and this is regardless of prior gastrectomy,” Dr. Ilson summarized. “Hematologic adverse events, such as neutropenia and leukopenia, may have been somewhat more frequent among the trifluridine/tipiracil–treated patients with gastrectomy than in the overall population, but this did not result in more treatment discontinuations. Exposure to the drug was similar between patients with gastrectomy and those in the overall population.”
“Trifluridine/tipiracil is an effective treatment option with a manageable toxicity profile for patients with metastatic gastric cancer, regardless of prior gastrectomy status,” he concluded.
Still fit for treatment
The disconnect between toxicity and treatment discontinuation seen in the TAGS trial is not new, according to invited discussant Martine Extermann, MD, PhD, leader of the Senior Adult Oncology Program at the Moffitt Cancer Center in Tampa. Previous data among geriatric cancer patients have similarly shown that, despite substantial chemotherapy toxicity, by and large, there are only modest effects on health-related quality of life, performance status, and instrumental activities of daily living, she noted.
“The CTCAE [Common Terminology Criteria for Adverse Events] toxicity differences do not always translate into functional impact and treatment cessation. So this is only part of the picture. It’s a convenient part. It’s easily measurable. It’s well acknowledged as a measurement of side effects. But it does not tell the whole story. Quality of life and functional status add to the picture,” Dr. Extermann elaborated. “What the TAGS study is telling us is, despite a gastrectomy, these patients can be treated as a third-line treatment population for gastric cancer, which is not necessarily obvious to every oncologist.”
At the same time, she added that it would be helpful to have nutritional data on the study patients – and on all patients in similar trials, for that matter – because nutritional status is one of the components of the CRASH (Chemotherapy Risk Assessment Scale for High-Age Patients) score used to predict chemotherapy toxicity in older adults.
“I would support that [trifluridine/tipiracil] is an effective third-line chemotherapy in gastric cancer patients with or without prior gastrectomy, and this can be given safely,” Dr. Extermann concluded.
Study details
Patients in TAGS were randomized 2:1 to trifluridine/tipiracil (formerly TAS-102) or placebo, each added to best supportive care. (Trifluridine is a novel oral thymidine analogue, and tipiracil prevents trifluridine degradation.) Overall, 44% had undergone gastrectomy before entering the trial.
In the entire trial population, overall survival was 5.7 months with trifluridine/tipiracil and 3.6 months with placebo (hazard ratio, 0.69; 95% confidence interval, 0.56-0.85; P = .0006), as previously reported.
Among the subgroup who had undergone prior gastrectomy, overall survival was 6.0 months with trifluridine/tipiracil and 3.4 months with placebo (HR, 0.57; 95% CI, 0.41-0.79), Dr. Ilson reported at the symposium, which is sponsored by the American Gastroenterological Association, the American Society for Clinical Oncology, the American Society for Radiation Oncology, and the Society of Surgical Oncology. The combination also netted better progression-free survival (2.2 vs. 1.8 months; HR, 0.48; 95% CI, 0.35-0.65). “These data mirror the data seen in the overall treatment population,” he commented.
In a multivariate analysis including all prespecified factors, prior gastrectomy was neither prognostic nor predictive. Moreover, the treatment effect size remained the same after adjustment for potential prognostic factors.
Exposure to trifluridine/tipiracil was similar for the gastrectomy subgroup and the entire trial population in terms of relative dose intensity, median number of cycles, and treatment duration.
The rate of grade 3 or 4 treatment-related adverse events with trifluridine/tipiracil in the gastrectomy subgroup, 64%, was higher than that in the entire trial population, 53%, but the rate of discontinuation because of any-grade adverse events was similar, at 10% and 13%, respectively.
The difference in grade 3 or 4 adverse events between the gastrectomy subgroup and the entire trial population was mainly driven by higher rates of neutropenia (44% vs. 34%) and leukopenia (14% vs. 9%) in the former.
Dr. Ilson disclosed that he has a consulting role with Amgen, Astellas, AstraZeneca, Bayer, Bristol-Myers Squibb, Lilly, Merck, Pieris, Roche/Genentech, and Taiho and that he receives research support from Taiho. The study was funded by Taiho Oncology and Taiho Pharmaceutical.
SOURCE: Ilson DH et al. 2019 GI Cancers Symposium, Abstract 3.
REPORTING FROM THE 2019 GI CANCERS SYMPOSIUM
Key clinical point: Patients with metastatic gastric cancer experience largely similar efficacy and safety outcomes with oral trifluridine/tipiracil regardless of prior gastrectomy.
Major finding: Compared with placebo, trifluridine/tipiracil improved overall survival in the gastrectomy subgroup (hazard ratio, 0.57) with a higher rate of grade 3/4 adverse events in that subgroup (64% vs. 53%) but similar rate of discontinuation because of adverse events (10% vs. 13%).
Study details: Preplanned subgroup analysis of a phase 3 randomized controlled trial (TAGS trial) among 507 patients with metastatic gastric or gastroesophageal junction cancer who had received at least two prior chemotherapy regimens.
Disclosures: Dr. Ilson disclosed that he has a consulting role with Amgen, Astellas, AstraZeneca, Bayer, Bristol-Myers Squibb, Lilly, Merck, Pieris, Roche/Genentech, and Taiho and that he receives research support from Taiho. The study was funded by Taiho Oncology and Taiho Pharmaceutical.
Source: Ilson DH et al. 2019 GI Cancers Symposium, Abstract 3.
Robotic sleeve gastrectomy may heighten organ space infection risk
While most outcomes are similar between robotic surgery and laparoscopic surgery for sleeve gastrectomy, the robotic approach carried a greater risk of organ space infection, according to the findings from a large clinical trial of more than 100,000 patients.
The study’s authors analyzed 107,726 sleeve gastrectomy operations in the Metabolic and Bariatric Surgery Association and Quality Improvement Program data registry (MBSAQIP), 7,385 of which were robotic sleeve gastrectomy (RSG). Peter William Lundberg, MD, and his coauthors of St. Luke’s University Health Network, Bethlehem, Pa., evaluated the safety of RSG vs. laparoscopic sleeve gastrectomy (LSG). The study was the first and largest comparing the two approaches to sleeve gastrectomy, the researchers noted.
“According to the MBSAQIP database, the robotic approach demonstrates a significantly higher rate of organ space infection while trending toward a lower rate of bleeding and 30-day reoperation and intervention,” Dr. Lundberg and his coauthors said.
Overall mortality was 0.07% in both groups (P = .49). The overall rates of significant adverse events were similar in both groups – 1.3% for LSG and 1.1% for RSG (P = .14) – as were bleeding rates – 0.5% and 0.4% (P = .003), respectively. The investigators characterized the slightly lower rates for RSG as “insignificant.”
RSG, however, had three times the rate of organ space infection than did the laparoscopic approach, 0.3% vs. 0.1% (P = .79). “Considering the enthusiasm with which robotics has been adopted by some bariatric surgeons, this is a sobering finding,” Dr. Lundberg noted.
The study determined that the use of staple-line reinforcement (SLR) alone significantly reduced the rate of bleeding regardless of approach by 31% on average (P = .0005). “This risk reduction was enhanced when SLR was combined with oversewing of the staple line,” Dr. Lundberg and his colleagues noted – an average reduction of 42% (P = .0009).
RSG took longer on average, 89 minutes vs. 63 minutes (P less than .0001), and the average length of stay was almost identical, 1.7 for RSG vs. 1.6 days for LSG. Reoperation rates within 30 days were also similar: 0.7% for RSG vs. 0.8% for LSG (P = .003).
“As surgeons continue to adopt and develop new technology, ongoing monitoring and reporting of safety and outcomes data are advised to maintain the high standards for outcomes in bariatric surgery,” Dr. Lundberg and his coauthors said.
The study researchers had no financial conflicts.
SOURCE: Lundberg PW et al. Surg Obes Relat Dis. 2018 Oct 25. doi:10.1016/j.soard.2018.10.015.
While most outcomes are similar between robotic surgery and laparoscopic surgery for sleeve gastrectomy, the robotic approach carried a greater risk of organ space infection, according to the findings from a large clinical trial of more than 100,000 patients.
The study’s authors analyzed 107,726 sleeve gastrectomy operations in the Metabolic and Bariatric Surgery Association and Quality Improvement Program data registry (MBSAQIP), 7,385 of which were robotic sleeve gastrectomy (RSG). Peter William Lundberg, MD, and his coauthors of St. Luke’s University Health Network, Bethlehem, Pa., evaluated the safety of RSG vs. laparoscopic sleeve gastrectomy (LSG). The study was the first and largest comparing the two approaches to sleeve gastrectomy, the researchers noted.
“According to the MBSAQIP database, the robotic approach demonstrates a significantly higher rate of organ space infection while trending toward a lower rate of bleeding and 30-day reoperation and intervention,” Dr. Lundberg and his coauthors said.
Overall mortality was 0.07% in both groups (P = .49). The overall rates of significant adverse events were similar in both groups – 1.3% for LSG and 1.1% for RSG (P = .14) – as were bleeding rates – 0.5% and 0.4% (P = .003), respectively. The investigators characterized the slightly lower rates for RSG as “insignificant.”
RSG, however, had three times the rate of organ space infection than did the laparoscopic approach, 0.3% vs. 0.1% (P = .79). “Considering the enthusiasm with which robotics has been adopted by some bariatric surgeons, this is a sobering finding,” Dr. Lundberg noted.
The study determined that the use of staple-line reinforcement (SLR) alone significantly reduced the rate of bleeding regardless of approach by 31% on average (P = .0005). “This risk reduction was enhanced when SLR was combined with oversewing of the staple line,” Dr. Lundberg and his colleagues noted – an average reduction of 42% (P = .0009).
RSG took longer on average, 89 minutes vs. 63 minutes (P less than .0001), and the average length of stay was almost identical, 1.7 for RSG vs. 1.6 days for LSG. Reoperation rates within 30 days were also similar: 0.7% for RSG vs. 0.8% for LSG (P = .003).
“As surgeons continue to adopt and develop new technology, ongoing monitoring and reporting of safety and outcomes data are advised to maintain the high standards for outcomes in bariatric surgery,” Dr. Lundberg and his coauthors said.
The study researchers had no financial conflicts.
SOURCE: Lundberg PW et al. Surg Obes Relat Dis. 2018 Oct 25. doi:10.1016/j.soard.2018.10.015.
While most outcomes are similar between robotic surgery and laparoscopic surgery for sleeve gastrectomy, the robotic approach carried a greater risk of organ space infection, according to the findings from a large clinical trial of more than 100,000 patients.
The study’s authors analyzed 107,726 sleeve gastrectomy operations in the Metabolic and Bariatric Surgery Association and Quality Improvement Program data registry (MBSAQIP), 7,385 of which were robotic sleeve gastrectomy (RSG). Peter William Lundberg, MD, and his coauthors of St. Luke’s University Health Network, Bethlehem, Pa., evaluated the safety of RSG vs. laparoscopic sleeve gastrectomy (LSG). The study was the first and largest comparing the two approaches to sleeve gastrectomy, the researchers noted.
“According to the MBSAQIP database, the robotic approach demonstrates a significantly higher rate of organ space infection while trending toward a lower rate of bleeding and 30-day reoperation and intervention,” Dr. Lundberg and his coauthors said.
Overall mortality was 0.07% in both groups (P = .49). The overall rates of significant adverse events were similar in both groups – 1.3% for LSG and 1.1% for RSG (P = .14) – as were bleeding rates – 0.5% and 0.4% (P = .003), respectively. The investigators characterized the slightly lower rates for RSG as “insignificant.”
RSG, however, had three times the rate of organ space infection than did the laparoscopic approach, 0.3% vs. 0.1% (P = .79). “Considering the enthusiasm with which robotics has been adopted by some bariatric surgeons, this is a sobering finding,” Dr. Lundberg noted.
The study determined that the use of staple-line reinforcement (SLR) alone significantly reduced the rate of bleeding regardless of approach by 31% on average (P = .0005). “This risk reduction was enhanced when SLR was combined with oversewing of the staple line,” Dr. Lundberg and his colleagues noted – an average reduction of 42% (P = .0009).
RSG took longer on average, 89 minutes vs. 63 minutes (P less than .0001), and the average length of stay was almost identical, 1.7 for RSG vs. 1.6 days for LSG. Reoperation rates within 30 days were also similar: 0.7% for RSG vs. 0.8% for LSG (P = .003).
“As surgeons continue to adopt and develop new technology, ongoing monitoring and reporting of safety and outcomes data are advised to maintain the high standards for outcomes in bariatric surgery,” Dr. Lundberg and his coauthors said.
The study researchers had no financial conflicts.
SOURCE: Lundberg PW et al. Surg Obes Relat Dis. 2018 Oct 25. doi:10.1016/j.soard.2018.10.015.
FROM SURGERY FOR OBESITY AND RELATED DISEASES
Key clinical point: Robotic sleeve gastrectomy carries a higher risk of organ space infection than does the laparoscopic approach.
Major finding: Rate of OSI was 0.3% with RSG and 0.1% with laparoscopic surgery.
Study details: An analysis of 107,726 patients who had sleeve gastrectomy in 2016 in the Metabolic and Bariatric Surgery Accreditation and Quality Improvement Program registry.
Disclosures: Dr. Lundberg and his coauthors reported having no conflicts.
Source: Lundberg PW et al. Surg Obes Related Dis. 2018 Oct. 25. doi:10.1016/j.soard.2018.10.015.
Endometriosis surgery: Women can expect years-long benefits
LAS VEGAS – according to a survey study from the University of Pittsburgh.
The work was likely the first to assess long-term outcomes after laparoscopic endometriosis excision with a disease-specific questionnaire, the Endometriosis Health Profile-30 (EHP-30). The findings should reassure both surgeons and patients. “I really feel these results can help us as endometriosis providers” to counsel women, said lead investigator Nicole M. Donnellan, MD, a gynecologic surgeon at the University of Pittsburgh.
Surgery “offers lasting improvement in all quality of life domains ... measured by the EHP-30”: pain; control/powerlessness; emotional well-being; social support; and self-image, with supplemental questions about work, sexual function, and other matters. Because “definitive surgery was not associated with improved outcomes when compared with fertility-sparing surgery ... fertility preservation should continue to be offered as first-line surgery for treatment of symptomatic disease,” Dr. Donnellan and her team concluded at a meeting sponsored by AAGL.
Surgery is the gold standard for endometriosis, but there just hasn’t been much data on long-term outcomes until now, especially with a potent questionnaire like the EHP-30. The gap left surgeons in the lurch on what to tell women how they’ll do, especially because results from previous, shorter, and less-rigorous studies have been mixed. The Pittsburgh results mean that competent surgeons can breathe easier and be confident in telling women what to expect.
The team administered EHP-30 to 61 women before surgery and at 4 weeks postoperatively; 45 patients (74%) had fertility-sparing excisions, 7 (11%) had hysterectomy with adnexa preservation, and 9 (15%) had hysterectomy with bilateral salpingo-oophorectomy. The women were contacted again in 2017 to fill out the survey anywhere from 3 to 7 years after their operation; 45 women agreed, a response rate of 74%.
There was a definitive, statistically significant reduction in scores across all five domains of the survey, both at 4 weeks and out to 7 years, and the improvements did not vary by endometriosis stage or the type of surgery women had.
The overall score – a combination of the five domains – fell from a preoperative median of 50 points out of a possible 100, with 100 being the worst possible score, to a median of about 20 points 4 weeks after surgery, and a median of about 10 points at long-term follow-up. Pain scores fell about the same amount; the greatest improvements were on questions that focused on sense of control and empowerment.
At long-term follow-up, overall scores improved a median of 43 points in women with American Society for Reproductive Medicine stage 1 endometriosis and 28 points among women with stage 4 disease (P = .705). Although the differences were not statistically significant, women with stage 1 disease generally reported the greatest improvements, except on the control and empowerment scale, where women reported the same improvement across all four stages, about 50 points out of 100.
Long-term score improvements were pretty much identical among women who had fertility-sparing surgery and those who had hysterectomies, with, for instance, both groups reporting about a 33-point improvement in pain scores. The two groups separated out only on emotional well-being scores, a 38-point improvement in the hysterectomy group versus 21 points, but the difference was not statistically significant (P = .525).
The long-term results remained the same when eight women who had subsequent gynecologic surgery were excluded.
In the end, the take home is that “all of these women improved,” Dr. Donnellan said.
The investigators didn’t report any disclosures.
SOURCE: Donnellan NM et al. 2018 AAGL Global Congress, Abstract 82.
LAS VEGAS – according to a survey study from the University of Pittsburgh.
The work was likely the first to assess long-term outcomes after laparoscopic endometriosis excision with a disease-specific questionnaire, the Endometriosis Health Profile-30 (EHP-30). The findings should reassure both surgeons and patients. “I really feel these results can help us as endometriosis providers” to counsel women, said lead investigator Nicole M. Donnellan, MD, a gynecologic surgeon at the University of Pittsburgh.
Surgery “offers lasting improvement in all quality of life domains ... measured by the EHP-30”: pain; control/powerlessness; emotional well-being; social support; and self-image, with supplemental questions about work, sexual function, and other matters. Because “definitive surgery was not associated with improved outcomes when compared with fertility-sparing surgery ... fertility preservation should continue to be offered as first-line surgery for treatment of symptomatic disease,” Dr. Donnellan and her team concluded at a meeting sponsored by AAGL.
Surgery is the gold standard for endometriosis, but there just hasn’t been much data on long-term outcomes until now, especially with a potent questionnaire like the EHP-30. The gap left surgeons in the lurch on what to tell women how they’ll do, especially because results from previous, shorter, and less-rigorous studies have been mixed. The Pittsburgh results mean that competent surgeons can breathe easier and be confident in telling women what to expect.
The team administered EHP-30 to 61 women before surgery and at 4 weeks postoperatively; 45 patients (74%) had fertility-sparing excisions, 7 (11%) had hysterectomy with adnexa preservation, and 9 (15%) had hysterectomy with bilateral salpingo-oophorectomy. The women were contacted again in 2017 to fill out the survey anywhere from 3 to 7 years after their operation; 45 women agreed, a response rate of 74%.
There was a definitive, statistically significant reduction in scores across all five domains of the survey, both at 4 weeks and out to 7 years, and the improvements did not vary by endometriosis stage or the type of surgery women had.
The overall score – a combination of the five domains – fell from a preoperative median of 50 points out of a possible 100, with 100 being the worst possible score, to a median of about 20 points 4 weeks after surgery, and a median of about 10 points at long-term follow-up. Pain scores fell about the same amount; the greatest improvements were on questions that focused on sense of control and empowerment.
At long-term follow-up, overall scores improved a median of 43 points in women with American Society for Reproductive Medicine stage 1 endometriosis and 28 points among women with stage 4 disease (P = .705). Although the differences were not statistically significant, women with stage 1 disease generally reported the greatest improvements, except on the control and empowerment scale, where women reported the same improvement across all four stages, about 50 points out of 100.
Long-term score improvements were pretty much identical among women who had fertility-sparing surgery and those who had hysterectomies, with, for instance, both groups reporting about a 33-point improvement in pain scores. The two groups separated out only on emotional well-being scores, a 38-point improvement in the hysterectomy group versus 21 points, but the difference was not statistically significant (P = .525).
The long-term results remained the same when eight women who had subsequent gynecologic surgery were excluded.
In the end, the take home is that “all of these women improved,” Dr. Donnellan said.
The investigators didn’t report any disclosures.
SOURCE: Donnellan NM et al. 2018 AAGL Global Congress, Abstract 82.
LAS VEGAS – according to a survey study from the University of Pittsburgh.
The work was likely the first to assess long-term outcomes after laparoscopic endometriosis excision with a disease-specific questionnaire, the Endometriosis Health Profile-30 (EHP-30). The findings should reassure both surgeons and patients. “I really feel these results can help us as endometriosis providers” to counsel women, said lead investigator Nicole M. Donnellan, MD, a gynecologic surgeon at the University of Pittsburgh.
Surgery “offers lasting improvement in all quality of life domains ... measured by the EHP-30”: pain; control/powerlessness; emotional well-being; social support; and self-image, with supplemental questions about work, sexual function, and other matters. Because “definitive surgery was not associated with improved outcomes when compared with fertility-sparing surgery ... fertility preservation should continue to be offered as first-line surgery for treatment of symptomatic disease,” Dr. Donnellan and her team concluded at a meeting sponsored by AAGL.
Surgery is the gold standard for endometriosis, but there just hasn’t been much data on long-term outcomes until now, especially with a potent questionnaire like the EHP-30. The gap left surgeons in the lurch on what to tell women how they’ll do, especially because results from previous, shorter, and less-rigorous studies have been mixed. The Pittsburgh results mean that competent surgeons can breathe easier and be confident in telling women what to expect.
The team administered EHP-30 to 61 women before surgery and at 4 weeks postoperatively; 45 patients (74%) had fertility-sparing excisions, 7 (11%) had hysterectomy with adnexa preservation, and 9 (15%) had hysterectomy with bilateral salpingo-oophorectomy. The women were contacted again in 2017 to fill out the survey anywhere from 3 to 7 years after their operation; 45 women agreed, a response rate of 74%.
There was a definitive, statistically significant reduction in scores across all five domains of the survey, both at 4 weeks and out to 7 years, and the improvements did not vary by endometriosis stage or the type of surgery women had.
The overall score – a combination of the five domains – fell from a preoperative median of 50 points out of a possible 100, with 100 being the worst possible score, to a median of about 20 points 4 weeks after surgery, and a median of about 10 points at long-term follow-up. Pain scores fell about the same amount; the greatest improvements were on questions that focused on sense of control and empowerment.
At long-term follow-up, overall scores improved a median of 43 points in women with American Society for Reproductive Medicine stage 1 endometriosis and 28 points among women with stage 4 disease (P = .705). Although the differences were not statistically significant, women with stage 1 disease generally reported the greatest improvements, except on the control and empowerment scale, where women reported the same improvement across all four stages, about 50 points out of 100.
Long-term score improvements were pretty much identical among women who had fertility-sparing surgery and those who had hysterectomies, with, for instance, both groups reporting about a 33-point improvement in pain scores. The two groups separated out only on emotional well-being scores, a 38-point improvement in the hysterectomy group versus 21 points, but the difference was not statistically significant (P = .525).
The long-term results remained the same when eight women who had subsequent gynecologic surgery were excluded.
In the end, the take home is that “all of these women improved,” Dr. Donnellan said.
The investigators didn’t report any disclosures.
SOURCE: Donnellan NM et al. 2018 AAGL Global Congress, Abstract 82.
REPORTING FROM THE AAGL GLOBAL CONGRESS
Key clinical point: Endometriosis excision improves quality of life for at least 7 years, even when women have conservative, fertility-sparing surgery.
Major finding: The overall score on the Endometriosis Health Profile-30 fell from a preoperative median of 50 points out of a possible 100, with 100 being the worst possible score, to a median of about 20 points 4 weeks after surgery, and a median of about 10 points at the 7-year follow-up.
Study details: A review of 61 cases
Disclosures: The investigators didn’t report any disclosures.
Source: Donnellan NM et al. 2018 AAGL Global Congress, Abstract 82.
Miscarriage after myomectomy depends on fibroid number, uterine incisions
LAS VEGAS – according to a review of 252 cases at Northwestern University, Chicago.
Surgeons feel terrible when a woman loses a pregnancy after fibroid treatment, and wonder if they “caused it, or if it was just a bad uterus or a bad initial pathology,” said lead investigator Laura M. Glaser, MD, an ob.gyn. in private practice in Lake Forest, Ill.
Her study, which was presented at a meeting sponsored by AAGL, suggests that miscarriage occurs mostly from complex pathology, as indicated by the number of fibroids and the degree of uterine cutting needed to remove them. The team reviewed outcomes among women who conceived after treatment; 28 had robotic-assisted myomectomies; 208 had open, abdominal myomectomies; and 16 had uterine fibroid embolization (UFE). Miscarriage was defined as pregnancy loss before 24 weeks.
After the researchers adjusted for age, body mass index, and parity, there were no statistically significant differences in miscarriage rates among the three groups (31% after UFE, 29% after robotic myomectomy, and 22% after abdominal myomectomy).
Open cases had the largest dominant fibroid at a mean of 8.5 cm, the most fibroids removed at 4.5, and the highest rate of cavity entry, 42%. Even so, at 22%, open cases were the least likely to miscarry.
Uterine size, specimen weight, time from procedure to pregnancy, and fibroid location didn’t seem to matter otherwise. The only risk factors that reached statistical significance were among women who had myomectomies; an increasing number of uterine cuts (odds ratio, 1.558; P = .004) and fibroids removed (OR, 1.11; P = .033) increased the odds of miscarriage.
More than 40% of women in the UFE group had previous fibroid surgery, versus 5% among women who had myomectomies. UFE women also were far more likely to have had a previous birth (50% versus 17%), but less likely to have subserosal fibroids (13% versus 33%), and their dominant fibroid was a few centimeters smaller.
Subjects were in their mid-30s, on average, with a mean body mass index of about 28 kg/m2. Just over 40% of the women who had myomectomies were white, versus 19% of women who had UFE.
There was no outside funding for the work, and the investigators didn’t have any disclosures.
SOURCE: Glaser LM et al. 2018 AAGL Global Congress, Abstract 160
LAS VEGAS – according to a review of 252 cases at Northwestern University, Chicago.
Surgeons feel terrible when a woman loses a pregnancy after fibroid treatment, and wonder if they “caused it, or if it was just a bad uterus or a bad initial pathology,” said lead investigator Laura M. Glaser, MD, an ob.gyn. in private practice in Lake Forest, Ill.
Her study, which was presented at a meeting sponsored by AAGL, suggests that miscarriage occurs mostly from complex pathology, as indicated by the number of fibroids and the degree of uterine cutting needed to remove them. The team reviewed outcomes among women who conceived after treatment; 28 had robotic-assisted myomectomies; 208 had open, abdominal myomectomies; and 16 had uterine fibroid embolization (UFE). Miscarriage was defined as pregnancy loss before 24 weeks.
After the researchers adjusted for age, body mass index, and parity, there were no statistically significant differences in miscarriage rates among the three groups (31% after UFE, 29% after robotic myomectomy, and 22% after abdominal myomectomy).
Open cases had the largest dominant fibroid at a mean of 8.5 cm, the most fibroids removed at 4.5, and the highest rate of cavity entry, 42%. Even so, at 22%, open cases were the least likely to miscarry.
Uterine size, specimen weight, time from procedure to pregnancy, and fibroid location didn’t seem to matter otherwise. The only risk factors that reached statistical significance were among women who had myomectomies; an increasing number of uterine cuts (odds ratio, 1.558; P = .004) and fibroids removed (OR, 1.11; P = .033) increased the odds of miscarriage.
More than 40% of women in the UFE group had previous fibroid surgery, versus 5% among women who had myomectomies. UFE women also were far more likely to have had a previous birth (50% versus 17%), but less likely to have subserosal fibroids (13% versus 33%), and their dominant fibroid was a few centimeters smaller.
Subjects were in their mid-30s, on average, with a mean body mass index of about 28 kg/m2. Just over 40% of the women who had myomectomies were white, versus 19% of women who had UFE.
There was no outside funding for the work, and the investigators didn’t have any disclosures.
SOURCE: Glaser LM et al. 2018 AAGL Global Congress, Abstract 160
LAS VEGAS – according to a review of 252 cases at Northwestern University, Chicago.
Surgeons feel terrible when a woman loses a pregnancy after fibroid treatment, and wonder if they “caused it, or if it was just a bad uterus or a bad initial pathology,” said lead investigator Laura M. Glaser, MD, an ob.gyn. in private practice in Lake Forest, Ill.
Her study, which was presented at a meeting sponsored by AAGL, suggests that miscarriage occurs mostly from complex pathology, as indicated by the number of fibroids and the degree of uterine cutting needed to remove them. The team reviewed outcomes among women who conceived after treatment; 28 had robotic-assisted myomectomies; 208 had open, abdominal myomectomies; and 16 had uterine fibroid embolization (UFE). Miscarriage was defined as pregnancy loss before 24 weeks.
After the researchers adjusted for age, body mass index, and parity, there were no statistically significant differences in miscarriage rates among the three groups (31% after UFE, 29% after robotic myomectomy, and 22% after abdominal myomectomy).
Open cases had the largest dominant fibroid at a mean of 8.5 cm, the most fibroids removed at 4.5, and the highest rate of cavity entry, 42%. Even so, at 22%, open cases were the least likely to miscarry.
Uterine size, specimen weight, time from procedure to pregnancy, and fibroid location didn’t seem to matter otherwise. The only risk factors that reached statistical significance were among women who had myomectomies; an increasing number of uterine cuts (odds ratio, 1.558; P = .004) and fibroids removed (OR, 1.11; P = .033) increased the odds of miscarriage.
More than 40% of women in the UFE group had previous fibroid surgery, versus 5% among women who had myomectomies. UFE women also were far more likely to have had a previous birth (50% versus 17%), but less likely to have subserosal fibroids (13% versus 33%), and their dominant fibroid was a few centimeters smaller.
Subjects were in their mid-30s, on average, with a mean body mass index of about 28 kg/m2. Just over 40% of the women who had myomectomies were white, versus 19% of women who had UFE.
There was no outside funding for the work, and the investigators didn’t have any disclosures.
SOURCE: Glaser LM et al. 2018 AAGL Global Congress, Abstract 160
REPORTING FROM AAGL GLOBAL CONGRESS
Key clinical point: The number of uterine incisions and fibroids removed increase the risk of miscarriage after fibroid treatment, not the type of procedure.
Major finding: After adjusting for age, body mass index, and parity, there were no statistically significant differences in miscarriage rates between the three groups (31% after uterine fibroid embolization; 29% after robotic myomectomy, and 22% after open abdominal myomectomy).
Study details: Review of 252 cases
Disclosures: There was no outside funding for the work, and the investigators didn’t have any disclosures.
Source: Glaser LM et al. 2018 AAGL Global Congress, Abstract 160
No change in postoperative pain with restrictive opioid protocol
Opioid prescriptions after gynecologic surgery can be significantly reduced without impacting postoperative pain scores or complication rates, according to a paper published in JAMA Network Open.
A tertiary care comprehensive care center implemented an ultrarestrictive opioid prescription protocol (UROPP) then evaluated the outcomes in a case-control study involving 605 women undergoing gynecologic surgery, compared with 626 controls treated before implementation of the new protocol.
The ultrarestrictive protocol was prompted by frequent inquiries from patients who had used very little of their prescribed opioids after surgery and wanted to know what to do with the unused pills.
The new protocol involved a short preoperative counseling session about postoperative pain management. Following that, ambulatory surgery, minimally invasive surgery, or laparotomy patients were prescribed a 7-day supply of nonopioid pain relief. Laparotomy patients were also prescribed a 3-day supply of an oral opioid.
Any patients who required more than five opioid doses in the 24 hours before discharge were also prescribed a 3-day supply of opioid pain medication as needed, and all patients had the option of requesting an additional 3-day opioid refill.
Researchers saw no significant differences between the two groups in mean postoperative pain scores 2 weeks after surgery, and a similar number of patients in each group requested an opioid refill. There was also no significant difference in the number of postoperative complications between groups.
Implementation of the ultrarestrictive protocol was associated with significant declines in the mean number of opioid pills prescribed dropped from 31.7 to 3.5 in all surgical cases, from 43.6 to 12.1 in the laparotomy group, from 38.4 to 1.3 in the minimally invasive surgery group, and from 13.9 to 0.2 in patients who underwent ambulatory surgery.
“These data suggest that the implementation of a UROPP in a large surgical service is feasible and safe and was associated with a significantly decreased number of opioids dispensed during the perioperative period, particularly among opioid-naive patients,” wrote Jaron Mark, MD, of the department of gynecologic oncology at Roswell Park Comprehensive Cancer Center, Buffalo, N.Y., and his coauthors. “The opioid-sparing effect was also marked and statistically significant in the laparotomy group, where most patients remained physically active and recovered well with no negative sequelae or elevated pain score after surgery.”
The researchers also noted that patients who were discharged home with an opioid prescription were more likely to call and request a refill within 30 days, compared with patients who did not receive opioids at discharge.
The study was supported by the Roswell Park Comprehensive Cancer Center, the National Cancer Institute and the Roswell Park Alliance Foundation. Two authors reported receiving fees and nonfinancial support from the private sector unrelated to the study.
SOURCE: Mark J et al. JAMA Netw Open. 2018 Dec 7. doi: 10.1001/jamanetworkopen.2018.5452.
The ultrarestrictive postoperative opioid prescribing protocol described in this study is a promising strategy for reducing opioid prescribing without increasing pain and limiting the potential for diversion and misuse of opioids. An important element of this protocol is the preoperative counseling, because setting patient expectations is likely to be an important factor in improving postoperative outcomes.
It is also worth noting that this study focused on patients undergoing major and minor gynecologic surgery, so more research is needed to explore these outcomes particularly among patients undergoing procedures that may be associated with a higher risk of persistent postoperative pain and/or opioid use. It is also a management strategy explored in patients at low risk of chronic postoperative opioid use, but a similar pathway should be developed and explored in more high-risk patients.
Dr. Jennifer M. Hah is from the department of anesthesiology, perioperative, and pain management at Stanford University (Calif.). These comments are taken from an accompanying editorial (JAMA Network Open. 2018 Dec 7. doi: 10.1001/jamanetworkopen.2018.5432). No conflicts of interest were reported.
The ultrarestrictive postoperative opioid prescribing protocol described in this study is a promising strategy for reducing opioid prescribing without increasing pain and limiting the potential for diversion and misuse of opioids. An important element of this protocol is the preoperative counseling, because setting patient expectations is likely to be an important factor in improving postoperative outcomes.
It is also worth noting that this study focused on patients undergoing major and minor gynecologic surgery, so more research is needed to explore these outcomes particularly among patients undergoing procedures that may be associated with a higher risk of persistent postoperative pain and/or opioid use. It is also a management strategy explored in patients at low risk of chronic postoperative opioid use, but a similar pathway should be developed and explored in more high-risk patients.
Dr. Jennifer M. Hah is from the department of anesthesiology, perioperative, and pain management at Stanford University (Calif.). These comments are taken from an accompanying editorial (JAMA Network Open. 2018 Dec 7. doi: 10.1001/jamanetworkopen.2018.5432). No conflicts of interest were reported.
The ultrarestrictive postoperative opioid prescribing protocol described in this study is a promising strategy for reducing opioid prescribing without increasing pain and limiting the potential for diversion and misuse of opioids. An important element of this protocol is the preoperative counseling, because setting patient expectations is likely to be an important factor in improving postoperative outcomes.
It is also worth noting that this study focused on patients undergoing major and minor gynecologic surgery, so more research is needed to explore these outcomes particularly among patients undergoing procedures that may be associated with a higher risk of persistent postoperative pain and/or opioid use. It is also a management strategy explored in patients at low risk of chronic postoperative opioid use, but a similar pathway should be developed and explored in more high-risk patients.
Dr. Jennifer M. Hah is from the department of anesthesiology, perioperative, and pain management at Stanford University (Calif.). These comments are taken from an accompanying editorial (JAMA Network Open. 2018 Dec 7. doi: 10.1001/jamanetworkopen.2018.5432). No conflicts of interest were reported.
Opioid prescriptions after gynecologic surgery can be significantly reduced without impacting postoperative pain scores or complication rates, according to a paper published in JAMA Network Open.
A tertiary care comprehensive care center implemented an ultrarestrictive opioid prescription protocol (UROPP) then evaluated the outcomes in a case-control study involving 605 women undergoing gynecologic surgery, compared with 626 controls treated before implementation of the new protocol.
The ultrarestrictive protocol was prompted by frequent inquiries from patients who had used very little of their prescribed opioids after surgery and wanted to know what to do with the unused pills.
The new protocol involved a short preoperative counseling session about postoperative pain management. Following that, ambulatory surgery, minimally invasive surgery, or laparotomy patients were prescribed a 7-day supply of nonopioid pain relief. Laparotomy patients were also prescribed a 3-day supply of an oral opioid.
Any patients who required more than five opioid doses in the 24 hours before discharge were also prescribed a 3-day supply of opioid pain medication as needed, and all patients had the option of requesting an additional 3-day opioid refill.
Researchers saw no significant differences between the two groups in mean postoperative pain scores 2 weeks after surgery, and a similar number of patients in each group requested an opioid refill. There was also no significant difference in the number of postoperative complications between groups.
Implementation of the ultrarestrictive protocol was associated with significant declines in the mean number of opioid pills prescribed dropped from 31.7 to 3.5 in all surgical cases, from 43.6 to 12.1 in the laparotomy group, from 38.4 to 1.3 in the minimally invasive surgery group, and from 13.9 to 0.2 in patients who underwent ambulatory surgery.
“These data suggest that the implementation of a UROPP in a large surgical service is feasible and safe and was associated with a significantly decreased number of opioids dispensed during the perioperative period, particularly among opioid-naive patients,” wrote Jaron Mark, MD, of the department of gynecologic oncology at Roswell Park Comprehensive Cancer Center, Buffalo, N.Y., and his coauthors. “The opioid-sparing effect was also marked and statistically significant in the laparotomy group, where most patients remained physically active and recovered well with no negative sequelae or elevated pain score after surgery.”
The researchers also noted that patients who were discharged home with an opioid prescription were more likely to call and request a refill within 30 days, compared with patients who did not receive opioids at discharge.
The study was supported by the Roswell Park Comprehensive Cancer Center, the National Cancer Institute and the Roswell Park Alliance Foundation. Two authors reported receiving fees and nonfinancial support from the private sector unrelated to the study.
SOURCE: Mark J et al. JAMA Netw Open. 2018 Dec 7. doi: 10.1001/jamanetworkopen.2018.5452.
Opioid prescriptions after gynecologic surgery can be significantly reduced without impacting postoperative pain scores or complication rates, according to a paper published in JAMA Network Open.
A tertiary care comprehensive care center implemented an ultrarestrictive opioid prescription protocol (UROPP) then evaluated the outcomes in a case-control study involving 605 women undergoing gynecologic surgery, compared with 626 controls treated before implementation of the new protocol.
The ultrarestrictive protocol was prompted by frequent inquiries from patients who had used very little of their prescribed opioids after surgery and wanted to know what to do with the unused pills.
The new protocol involved a short preoperative counseling session about postoperative pain management. Following that, ambulatory surgery, minimally invasive surgery, or laparotomy patients were prescribed a 7-day supply of nonopioid pain relief. Laparotomy patients were also prescribed a 3-day supply of an oral opioid.
Any patients who required more than five opioid doses in the 24 hours before discharge were also prescribed a 3-day supply of opioid pain medication as needed, and all patients had the option of requesting an additional 3-day opioid refill.
Researchers saw no significant differences between the two groups in mean postoperative pain scores 2 weeks after surgery, and a similar number of patients in each group requested an opioid refill. There was also no significant difference in the number of postoperative complications between groups.
Implementation of the ultrarestrictive protocol was associated with significant declines in the mean number of opioid pills prescribed dropped from 31.7 to 3.5 in all surgical cases, from 43.6 to 12.1 in the laparotomy group, from 38.4 to 1.3 in the minimally invasive surgery group, and from 13.9 to 0.2 in patients who underwent ambulatory surgery.
“These data suggest that the implementation of a UROPP in a large surgical service is feasible and safe and was associated with a significantly decreased number of opioids dispensed during the perioperative period, particularly among opioid-naive patients,” wrote Jaron Mark, MD, of the department of gynecologic oncology at Roswell Park Comprehensive Cancer Center, Buffalo, N.Y., and his coauthors. “The opioid-sparing effect was also marked and statistically significant in the laparotomy group, where most patients remained physically active and recovered well with no negative sequelae or elevated pain score after surgery.”
The researchers also noted that patients who were discharged home with an opioid prescription were more likely to call and request a refill within 30 days, compared with patients who did not receive opioids at discharge.
The study was supported by the Roswell Park Comprehensive Cancer Center, the National Cancer Institute and the Roswell Park Alliance Foundation. Two authors reported receiving fees and nonfinancial support from the private sector unrelated to the study.
SOURCE: Mark J et al. JAMA Netw Open. 2018 Dec 7. doi: 10.1001/jamanetworkopen.2018.5452.
Key clinical point: A ultrarestrictive postoperative opioid protocol is not associated with higher postoperative pain scores.
Major finding: The protocol achieves significant reductions in opioid use.
Study details: A case-control study in 1,231 women undergoing gynecologic surgery.
Disclosures: The study was supported by the Roswell Park Comprehensive Cancer Center, the National Cancer Institute, and the Roswell Park Alliance Foundation. Two authors reported receiving fees and nonfinancial support from the private sector unrelated to the study.
Source: Mark J et al. JAMA Netw Open. 2018 Dec 7. doi: 10.1001/jamanetworkopen.2018.5452.
EHR stress linked to burnout
Physicians who experience stress related to the use of health information technology are twice as likely to experience burnout.
Rebekah Gardner, MD, of Brown University in Providence, R.I., and her colleagues surveyed all 4,197 Rhode Island physicians in 2017 to learn how the use of electronic health records affected their practices and their job satisfaction.
Just over a quarter (25.0%) of 1,792 respondents reported burnout. Among electronic health record users (91% of respondents), 70% reported health IT-related stress (J Am Med Inform Assoc. doi: 10.1093/jamia/ocy145).
“After adjustment, physicians reporting poor/marginal time for documentation had 2.8 times the odds of burnout (95% confidence interval, 2.0-4.1; P less than .0001) compared to those reporting sufficient time,” according to the researchers.
The team looked at three stress-related variables: whether the EHR adds to the frustration of one’s day; whether physicians felt they had sufficient time for documentation; and the amount of time spent on the EHR at home. Variables were measured on a four- or five-point scale depending on the question related to the specific stress variable.
Almost two-thirds (64.2%) of respondents “agreed” or “strongly agreed” that EHRs add to the frustration of their day.
“It was the most commonly cited HIT-related stress measure in almost every specialty, with the highest prevalence among emergency physicians (77.6%),” the investigators wrote.
More than a third of physicians (37.7%) reported “moderately high” or “excessive” time spent on EHRs at home; this metric was the most commonly cited stress measure among pediatricians (63.6%).
Nearly half (46.4%) of physicians reported “poor” or “marginal” sufficiency of time for documentation.
“Presence of any 1 of the HIT-related stress measures was associated with approximately twice the odds of burnout among physician respondents,” Dr. Gardner and her colleagues noted, adding that “measuring and addressing HIT-related stress is an important step in reducing workforce burden and improving the care of our patients.”
To alleviate burnout, the authors recommended increased use of scribes, use of medical assistants to help create a more team-based documentation function, improved EHR training, providing more time during the day for documentation, and streamlining documentation expectations, with certain culture shifts needed in some cases (i.e., banning work-related email and clinical tasks for vacationing physicians).
SOURCE: Gardner R et al. J Am Med Inform Assoc. doi: 10.1093/jamia/ocy145.
Physicians who experience stress related to the use of health information technology are twice as likely to experience burnout.
Rebekah Gardner, MD, of Brown University in Providence, R.I., and her colleagues surveyed all 4,197 Rhode Island physicians in 2017 to learn how the use of electronic health records affected their practices and their job satisfaction.
Just over a quarter (25.0%) of 1,792 respondents reported burnout. Among electronic health record users (91% of respondents), 70% reported health IT-related stress (J Am Med Inform Assoc. doi: 10.1093/jamia/ocy145).
“After adjustment, physicians reporting poor/marginal time for documentation had 2.8 times the odds of burnout (95% confidence interval, 2.0-4.1; P less than .0001) compared to those reporting sufficient time,” according to the researchers.
The team looked at three stress-related variables: whether the EHR adds to the frustration of one’s day; whether physicians felt they had sufficient time for documentation; and the amount of time spent on the EHR at home. Variables were measured on a four- or five-point scale depending on the question related to the specific stress variable.
Almost two-thirds (64.2%) of respondents “agreed” or “strongly agreed” that EHRs add to the frustration of their day.
“It was the most commonly cited HIT-related stress measure in almost every specialty, with the highest prevalence among emergency physicians (77.6%),” the investigators wrote.
More than a third of physicians (37.7%) reported “moderately high” or “excessive” time spent on EHRs at home; this metric was the most commonly cited stress measure among pediatricians (63.6%).
Nearly half (46.4%) of physicians reported “poor” or “marginal” sufficiency of time for documentation.
“Presence of any 1 of the HIT-related stress measures was associated with approximately twice the odds of burnout among physician respondents,” Dr. Gardner and her colleagues noted, adding that “measuring and addressing HIT-related stress is an important step in reducing workforce burden and improving the care of our patients.”
To alleviate burnout, the authors recommended increased use of scribes, use of medical assistants to help create a more team-based documentation function, improved EHR training, providing more time during the day for documentation, and streamlining documentation expectations, with certain culture shifts needed in some cases (i.e., banning work-related email and clinical tasks for vacationing physicians).
SOURCE: Gardner R et al. J Am Med Inform Assoc. doi: 10.1093/jamia/ocy145.
Physicians who experience stress related to the use of health information technology are twice as likely to experience burnout.
Rebekah Gardner, MD, of Brown University in Providence, R.I., and her colleagues surveyed all 4,197 Rhode Island physicians in 2017 to learn how the use of electronic health records affected their practices and their job satisfaction.
Just over a quarter (25.0%) of 1,792 respondents reported burnout. Among electronic health record users (91% of respondents), 70% reported health IT-related stress (J Am Med Inform Assoc. doi: 10.1093/jamia/ocy145).
“After adjustment, physicians reporting poor/marginal time for documentation had 2.8 times the odds of burnout (95% confidence interval, 2.0-4.1; P less than .0001) compared to those reporting sufficient time,” according to the researchers.
The team looked at three stress-related variables: whether the EHR adds to the frustration of one’s day; whether physicians felt they had sufficient time for documentation; and the amount of time spent on the EHR at home. Variables were measured on a four- or five-point scale depending on the question related to the specific stress variable.
Almost two-thirds (64.2%) of respondents “agreed” or “strongly agreed” that EHRs add to the frustration of their day.
“It was the most commonly cited HIT-related stress measure in almost every specialty, with the highest prevalence among emergency physicians (77.6%),” the investigators wrote.
More than a third of physicians (37.7%) reported “moderately high” or “excessive” time spent on EHRs at home; this metric was the most commonly cited stress measure among pediatricians (63.6%).
Nearly half (46.4%) of physicians reported “poor” or “marginal” sufficiency of time for documentation.
“Presence of any 1 of the HIT-related stress measures was associated with approximately twice the odds of burnout among physician respondents,” Dr. Gardner and her colleagues noted, adding that “measuring and addressing HIT-related stress is an important step in reducing workforce burden and improving the care of our patients.”
To alleviate burnout, the authors recommended increased use of scribes, use of medical assistants to help create a more team-based documentation function, improved EHR training, providing more time during the day for documentation, and streamlining documentation expectations, with certain culture shifts needed in some cases (i.e., banning work-related email and clinical tasks for vacationing physicians).
SOURCE: Gardner R et al. J Am Med Inform Assoc. doi: 10.1093/jamia/ocy145.
FROM JAMIA
Key clinical point: EHR-related stress is a measurable predictor of physician burnout.
Major finding: Seventy percent of EHR users who responded to a survey reported stress related to the use of health information technology.
Study details: Survey of all 4,197 physicians in Rhode Island; 1,792 (43%) responded.
Disclosures: The Rhode Island Department of Health funded the study. The authors reported no conflicts of interest.
Source: Gardner R et al. J Am Med Inform Assoc. doi: 10.1093/jamia/ocy145.
Black women more likely to have open hysterectomies
Black women were more likely than white women to undergo open hysterectomy, according to a recent analysis of national surgical data by Amy L. Alexander, MD, MS, of Northwestern University, Chicago, and her colleagues.
Even after the researchers controlled for many factors that might influence surgical approach, such as comorbidities and body mass index, black women had an odds ratio of 2.02 to receive open, rather than laparoscopic, hysterectomy (95% confidence interval, 1.85-2.20), according to a study in Obstetrics & Gynecology.
The analysis of the targeted hysterectomy file in the National Surgical Quality Improvement Program (NSQIP) database showed that, of 15,316 women who had hysterectomy for nonmalignant indications, the 25% who were black also were more likely to have major complications with open procedures. Such complications as sepsis, wound dehiscence, prolonged intubation, and death were seen in 4% of the black women, versus 2% of the white women receiving open hysterectomy (P less than .001). Minor complications, such as urinary tract infections, superficial wound infections, and blood transfusions, also were more common for black women having open procedures (11% vs. 7%; P less than .001).
The study used a large national database with detailed information about comorbidities and patient characteristics to look at racial disparities in surgical route and complications for the second-most-common surgical procedure women receive on the United States. The results, said Dr. Alexander and her coauthors, confirm and extend previous work showing these disparities.
Black women are known to have more diabetes and hypertension, as well as higher rates of obesity, compared with white women, wrote Dr. Alexander and her coauthors. Even after they controlled for all these variables, black women still had significantly higher odds of having complications from hysterectomy: The odds ratios for major and minor complications were 1.56 and 1.27, respectively.
Uterine weight was included and tracked as a binary variable, with large uteri considered those weighing 250 g or more. Making uterine weight a binary, rather than continuous or categorical variable, didn’t significantly change results, and realistically mirrors a surgeon’s assessment of a uterus as “large” or “small” when making treatment decisions, Dr. Alexander and her coauthors said.
Because the median weight of uteri from black women was more than double the weight of those from white women (262 g vs. 123 g), the investigators also performed an analysis looking just at women with uterine weight less than 250 g, to ensure that uterine weight alone was not accounting for much of the disparity. In this analysis, the black patients still had an adjusted odds ratio of 1.84 for receiving an open procedure.
“Some of the postoperative complications experienced by black women are likely attributable to the fact that black women are more likely to undergo an open hysterectomy,” noted Dr. Alexander and her colleagues. “However, because black race is still associated with a higher odds of complications, even when adjusting for hysterectomy route, there are other contributing factors that warrant further investigation.” Among these factors, they said, may be access to care and quality of care while hospitalized.
The study’s strengths include the use of the NSQIP database’s prospectively collected data to construct the cohort study; the data base included “important patient-level factors such as uterine size, obesity, and comorbidities not previously available in other secondary data set studies,” noted Dr. Alexander and her colleagues. But the possibility of unmeasured bias persists, they said, and such variables as regional practice patterns and surgeon experience and procedure volume could not be detected from the NSQIP data on hand.
“This study suggests that an important step to reduce the disparity in route of surgery and postoperative complications is to increase access to and use of minimally invasive surgery,” wrote Dr. Alexander and her coauthors.
The study was funded by the National Institutes of Health. Dr. Alexander and her colleagues reported no conflicts of interest.
koakes@mdedge.com
SOURCE: Alexander AL et al. Obstet Gynecol. 2018 Dec 4. doi: 10.1097/AOG.0000000000002990
The discrepancies reported by Dr. Alexander and her colleagues between black and white women undergoing hysterectomy for nonmalignant reasons mirror discrepancies found for other oncologic procedures, including colectomy and prostatectomy.
Findings of the current study show that black women continue to be less likely to receive more modern, minimally invasive procedures, and that outcomes are worse for these women, even after controlling for factors that may contribute to complications.
The American College of Obstetricians and Gynecologists (ACOG) has looked at this issue. In a 2015 opinion, an ACOG committee looked at three categories of factors contributing to racial disparities in women’s health care. Patient-level factors include such things as genetics, medical comorbidities, and patient preferences and adherence. Health care system–level factors include insurance status and geographic barriers to accessing care, while stereotyping and implicit bias on the part of practitioners constitute the third set of factors.
All these factors are likely in play for the discrepancies seen in hysterectomy rates. Those who care for black women need to understand the long, shameful history of how black Americans were mistreated, undertreated, and used for medical experimentation without consent. This past shapes present care and contributes to the difficulty black patients have trusting today’s health care systems.
The important question today is how individual health care providers will address their own biases, learn from the past, and move forward to do better for our black patients.
Shanna N. Wingo, MD , is an ob.gyn. and a gynecologic oncologist in private practice in Phoenix. She had no potential conflicts of interest. These remarks were drawn from an editorial accompanying the study by Dr. Alexander et al.( Obstet Gynecol. 2018 Dec 4;133[1]:4-5 ).
The discrepancies reported by Dr. Alexander and her colleagues between black and white women undergoing hysterectomy for nonmalignant reasons mirror discrepancies found for other oncologic procedures, including colectomy and prostatectomy.
Findings of the current study show that black women continue to be less likely to receive more modern, minimally invasive procedures, and that outcomes are worse for these women, even after controlling for factors that may contribute to complications.
The American College of Obstetricians and Gynecologists (ACOG) has looked at this issue. In a 2015 opinion, an ACOG committee looked at three categories of factors contributing to racial disparities in women’s health care. Patient-level factors include such things as genetics, medical comorbidities, and patient preferences and adherence. Health care system–level factors include insurance status and geographic barriers to accessing care, while stereotyping and implicit bias on the part of practitioners constitute the third set of factors.
All these factors are likely in play for the discrepancies seen in hysterectomy rates. Those who care for black women need to understand the long, shameful history of how black Americans were mistreated, undertreated, and used for medical experimentation without consent. This past shapes present care and contributes to the difficulty black patients have trusting today’s health care systems.
The important question today is how individual health care providers will address their own biases, learn from the past, and move forward to do better for our black patients.
Shanna N. Wingo, MD , is an ob.gyn. and a gynecologic oncologist in private practice in Phoenix. She had no potential conflicts of interest. These remarks were drawn from an editorial accompanying the study by Dr. Alexander et al.( Obstet Gynecol. 2018 Dec 4;133[1]:4-5 ).
The discrepancies reported by Dr. Alexander and her colleagues between black and white women undergoing hysterectomy for nonmalignant reasons mirror discrepancies found for other oncologic procedures, including colectomy and prostatectomy.
Findings of the current study show that black women continue to be less likely to receive more modern, minimally invasive procedures, and that outcomes are worse for these women, even after controlling for factors that may contribute to complications.
The American College of Obstetricians and Gynecologists (ACOG) has looked at this issue. In a 2015 opinion, an ACOG committee looked at three categories of factors contributing to racial disparities in women’s health care. Patient-level factors include such things as genetics, medical comorbidities, and patient preferences and adherence. Health care system–level factors include insurance status and geographic barriers to accessing care, while stereotyping and implicit bias on the part of practitioners constitute the third set of factors.
All these factors are likely in play for the discrepancies seen in hysterectomy rates. Those who care for black women need to understand the long, shameful history of how black Americans were mistreated, undertreated, and used for medical experimentation without consent. This past shapes present care and contributes to the difficulty black patients have trusting today’s health care systems.
The important question today is how individual health care providers will address their own biases, learn from the past, and move forward to do better for our black patients.
Shanna N. Wingo, MD , is an ob.gyn. and a gynecologic oncologist in private practice in Phoenix. She had no potential conflicts of interest. These remarks were drawn from an editorial accompanying the study by Dr. Alexander et al.( Obstet Gynecol. 2018 Dec 4;133[1]:4-5 ).
Black women were more likely than white women to undergo open hysterectomy, according to a recent analysis of national surgical data by Amy L. Alexander, MD, MS, of Northwestern University, Chicago, and her colleagues.
Even after the researchers controlled for many factors that might influence surgical approach, such as comorbidities and body mass index, black women had an odds ratio of 2.02 to receive open, rather than laparoscopic, hysterectomy (95% confidence interval, 1.85-2.20), according to a study in Obstetrics & Gynecology.
The analysis of the targeted hysterectomy file in the National Surgical Quality Improvement Program (NSQIP) database showed that, of 15,316 women who had hysterectomy for nonmalignant indications, the 25% who were black also were more likely to have major complications with open procedures. Such complications as sepsis, wound dehiscence, prolonged intubation, and death were seen in 4% of the black women, versus 2% of the white women receiving open hysterectomy (P less than .001). Minor complications, such as urinary tract infections, superficial wound infections, and blood transfusions, also were more common for black women having open procedures (11% vs. 7%; P less than .001).
The study used a large national database with detailed information about comorbidities and patient characteristics to look at racial disparities in surgical route and complications for the second-most-common surgical procedure women receive on the United States. The results, said Dr. Alexander and her coauthors, confirm and extend previous work showing these disparities.
Black women are known to have more diabetes and hypertension, as well as higher rates of obesity, compared with white women, wrote Dr. Alexander and her coauthors. Even after they controlled for all these variables, black women still had significantly higher odds of having complications from hysterectomy: The odds ratios for major and minor complications were 1.56 and 1.27, respectively.
Uterine weight was included and tracked as a binary variable, with large uteri considered those weighing 250 g or more. Making uterine weight a binary, rather than continuous or categorical variable, didn’t significantly change results, and realistically mirrors a surgeon’s assessment of a uterus as “large” or “small” when making treatment decisions, Dr. Alexander and her coauthors said.
Because the median weight of uteri from black women was more than double the weight of those from white women (262 g vs. 123 g), the investigators also performed an analysis looking just at women with uterine weight less than 250 g, to ensure that uterine weight alone was not accounting for much of the disparity. In this analysis, the black patients still had an adjusted odds ratio of 1.84 for receiving an open procedure.
“Some of the postoperative complications experienced by black women are likely attributable to the fact that black women are more likely to undergo an open hysterectomy,” noted Dr. Alexander and her colleagues. “However, because black race is still associated with a higher odds of complications, even when adjusting for hysterectomy route, there are other contributing factors that warrant further investigation.” Among these factors, they said, may be access to care and quality of care while hospitalized.
The study’s strengths include the use of the NSQIP database’s prospectively collected data to construct the cohort study; the data base included “important patient-level factors such as uterine size, obesity, and comorbidities not previously available in other secondary data set studies,” noted Dr. Alexander and her colleagues. But the possibility of unmeasured bias persists, they said, and such variables as regional practice patterns and surgeon experience and procedure volume could not be detected from the NSQIP data on hand.
“This study suggests that an important step to reduce the disparity in route of surgery and postoperative complications is to increase access to and use of minimally invasive surgery,” wrote Dr. Alexander and her coauthors.
The study was funded by the National Institutes of Health. Dr. Alexander and her colleagues reported no conflicts of interest.
koakes@mdedge.com
SOURCE: Alexander AL et al. Obstet Gynecol. 2018 Dec 4. doi: 10.1097/AOG.0000000000002990
Black women were more likely than white women to undergo open hysterectomy, according to a recent analysis of national surgical data by Amy L. Alexander, MD, MS, of Northwestern University, Chicago, and her colleagues.
Even after the researchers controlled for many factors that might influence surgical approach, such as comorbidities and body mass index, black women had an odds ratio of 2.02 to receive open, rather than laparoscopic, hysterectomy (95% confidence interval, 1.85-2.20), according to a study in Obstetrics & Gynecology.
The analysis of the targeted hysterectomy file in the National Surgical Quality Improvement Program (NSQIP) database showed that, of 15,316 women who had hysterectomy for nonmalignant indications, the 25% who were black also were more likely to have major complications with open procedures. Such complications as sepsis, wound dehiscence, prolonged intubation, and death were seen in 4% of the black women, versus 2% of the white women receiving open hysterectomy (P less than .001). Minor complications, such as urinary tract infections, superficial wound infections, and blood transfusions, also were more common for black women having open procedures (11% vs. 7%; P less than .001).
The study used a large national database with detailed information about comorbidities and patient characteristics to look at racial disparities in surgical route and complications for the second-most-common surgical procedure women receive on the United States. The results, said Dr. Alexander and her coauthors, confirm and extend previous work showing these disparities.
Black women are known to have more diabetes and hypertension, as well as higher rates of obesity, compared with white women, wrote Dr. Alexander and her coauthors. Even after they controlled for all these variables, black women still had significantly higher odds of having complications from hysterectomy: The odds ratios for major and minor complications were 1.56 and 1.27, respectively.
Uterine weight was included and tracked as a binary variable, with large uteri considered those weighing 250 g or more. Making uterine weight a binary, rather than continuous or categorical variable, didn’t significantly change results, and realistically mirrors a surgeon’s assessment of a uterus as “large” or “small” when making treatment decisions, Dr. Alexander and her coauthors said.
Because the median weight of uteri from black women was more than double the weight of those from white women (262 g vs. 123 g), the investigators also performed an analysis looking just at women with uterine weight less than 250 g, to ensure that uterine weight alone was not accounting for much of the disparity. In this analysis, the black patients still had an adjusted odds ratio of 1.84 for receiving an open procedure.
“Some of the postoperative complications experienced by black women are likely attributable to the fact that black women are more likely to undergo an open hysterectomy,” noted Dr. Alexander and her colleagues. “However, because black race is still associated with a higher odds of complications, even when adjusting for hysterectomy route, there are other contributing factors that warrant further investigation.” Among these factors, they said, may be access to care and quality of care while hospitalized.
The study’s strengths include the use of the NSQIP database’s prospectively collected data to construct the cohort study; the data base included “important patient-level factors such as uterine size, obesity, and comorbidities not previously available in other secondary data set studies,” noted Dr. Alexander and her colleagues. But the possibility of unmeasured bias persists, they said, and such variables as regional practice patterns and surgeon experience and procedure volume could not be detected from the NSQIP data on hand.
“This study suggests that an important step to reduce the disparity in route of surgery and postoperative complications is to increase access to and use of minimally invasive surgery,” wrote Dr. Alexander and her coauthors.
The study was funded by the National Institutes of Health. Dr. Alexander and her colleagues reported no conflicts of interest.
koakes@mdedge.com
SOURCE: Alexander AL et al. Obstet Gynecol. 2018 Dec 4. doi: 10.1097/AOG.0000000000002990
FROM OBSTETRICS & GYNECOLOGY
Key clinical point: Black women were more likely than white women were to have open hysterectomies.
Major finding:
Study details: Analysis of prospectively collected hysterectomy data of 15,136 women in the NSQIP database.
Disclosures: The study was funded by the National Institutes of Health. Dr. Alexander and her coauthors reported no conflicts of interest.
Source: Alexander AL et al. Obstet Gynecol. 2018 Dec 4. doi: 10.1097/AOG0000000000002990.
Healthcare.gov activity surged in last week of open enrollment
A surge in activity during the last week of open enrollment at Healthcare.gov more than doubled the number of plans selected for the season, according to the Centers for Medicare & Medicaid Services.
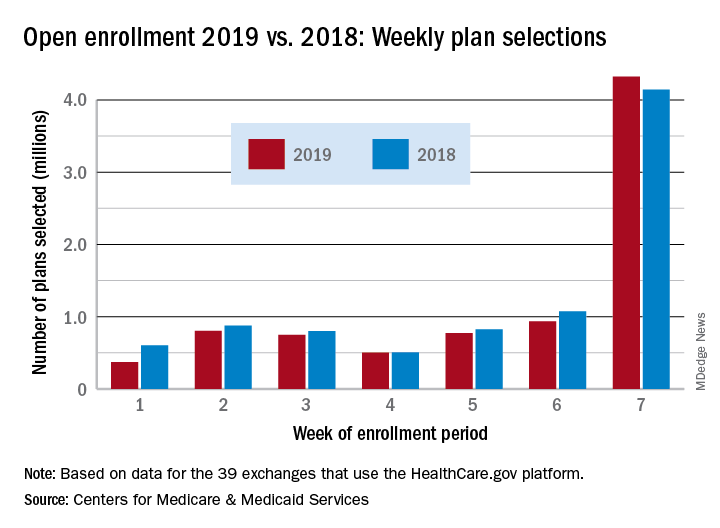
Over 4.32 million plans were selected during week 7 (Dec. 9-15) of enrollment for the 2019 coverage year, exceeding the 4.13 million selected over the previous 6 weeks and bringing the total to 8.45 million, the CMS reported. During week 6 (Dec. 3-9), which was previously the busiest week of the year, 943,000 plans were selected by residents of the 39 states that use the Healthcare.gov platform.
This year’s week 7 total also topped the comparable number from last year’s enrollment period for the first time and closed the gap in cumulative selections from 11.7% after 6 weeks to 4.2%. Last year, a total of 8.82 million plans were selected for the 2018 coverage year, CMS said, while also noting that the data for this year “are preliminary and do not represent final 2019 Exchange Open Enrollment figures.”
CMS Administrator Seema Verma addressed the drop from 2018 to 2019: “With the lowest unemployment rate in 50 years, it’s possible that more Americans have employer based coverage, and don’t need exchange plans.” The CMS also estimated that “approximately 100,000 current exchange enrollees in Virginia will be eligible for” Medicaid now that the state has expanded its Medicaid population.
A surge in activity during the last week of open enrollment at Healthcare.gov more than doubled the number of plans selected for the season, according to the Centers for Medicare & Medicaid Services.

Over 4.32 million plans were selected during week 7 (Dec. 9-15) of enrollment for the 2019 coverage year, exceeding the 4.13 million selected over the previous 6 weeks and bringing the total to 8.45 million, the CMS reported. During week 6 (Dec. 3-9), which was previously the busiest week of the year, 943,000 plans were selected by residents of the 39 states that use the Healthcare.gov platform.
This year’s week 7 total also topped the comparable number from last year’s enrollment period for the first time and closed the gap in cumulative selections from 11.7% after 6 weeks to 4.2%. Last year, a total of 8.82 million plans were selected for the 2018 coverage year, CMS said, while also noting that the data for this year “are preliminary and do not represent final 2019 Exchange Open Enrollment figures.”
CMS Administrator Seema Verma addressed the drop from 2018 to 2019: “With the lowest unemployment rate in 50 years, it’s possible that more Americans have employer based coverage, and don’t need exchange plans.” The CMS also estimated that “approximately 100,000 current exchange enrollees in Virginia will be eligible for” Medicaid now that the state has expanded its Medicaid population.
A surge in activity during the last week of open enrollment at Healthcare.gov more than doubled the number of plans selected for the season, according to the Centers for Medicare & Medicaid Services.

Over 4.32 million plans were selected during week 7 (Dec. 9-15) of enrollment for the 2019 coverage year, exceeding the 4.13 million selected over the previous 6 weeks and bringing the total to 8.45 million, the CMS reported. During week 6 (Dec. 3-9), which was previously the busiest week of the year, 943,000 plans were selected by residents of the 39 states that use the Healthcare.gov platform.
This year’s week 7 total also topped the comparable number from last year’s enrollment period for the first time and closed the gap in cumulative selections from 11.7% after 6 weeks to 4.2%. Last year, a total of 8.82 million plans were selected for the 2018 coverage year, CMS said, while also noting that the data for this year “are preliminary and do not represent final 2019 Exchange Open Enrollment figures.”
CMS Administrator Seema Verma addressed the drop from 2018 to 2019: “With the lowest unemployment rate in 50 years, it’s possible that more Americans have employer based coverage, and don’t need exchange plans.” The CMS also estimated that “approximately 100,000 current exchange enrollees in Virginia will be eligible for” Medicaid now that the state has expanded its Medicaid population.
Next legal ruling on ACA could come on New Year’s Eve
As Democratic governors scramble to appeal a Dec. 14 ruling that essentially struck down the Affordable Care Act, a reprieve keeping the law intact could come by the end of the year.
The ACA remains in effect until Dec. 31 after Judge Reed O’Connor of the U.S. District Court for the Northern District of Texas ruled that the 2017 tax law that zeroed out penalties beginning in 2019 for not carrying health insurance effectively rendered the entire ACA unconstitutional.
On Dec. 17, a group of Democratic attorneys general “asked Judge O’Connor to clarify that his ruling does not presently affect any rights or responsibilities under the ACA until appellate review is complete, or alternatively to stay his ruling,” Timothy S. Jost, emeritus professor at the Washington and Lee School of Law, in Lexington, Va., said during a Dec. 18 press teleconference hosted by the Commonwealth Fund. “They also asked him to certify the case for an immediate appeal and to do so by the end of this week.”
Mr. Jost noted that Judge O’Connor asked the plaintiffs and the U.S. Department of Health & Human Services to respond to this request by Dec. 21.
“I expect him to rule, probably, on New Year’s Eve on what happens next,” Mr. Jost said.
If Judge O’Connor refuses to stay his order, the group of Democratic attorneys general is expected to appeal to the Fifth Circuit Court of Appeals for such a stay, Mr. Jost said. He noted that the Fifth Circuit is one of the more conservative courts – with five Trump administration appointees and five judges appointed by previous Democratic administrations.
In general, Mr. Jost said that he does not expect Judge O’Connor’s ruling to stand.
“The decision is so clearly wrong, however, that I believe there is a good chance that it will be reversed,” he said. “If the Fifth Circuit does, I think it is very unlikely the Supreme Court take the case.” He added that if the Fifth Circuit upholds Judge O’Connor’s ruling, the court would take the case and “very likely reverse at least 5-4 and quite possibly 6-3 on at least the issue of severability.”
Mr. Jost added that he expects the case to drag into 2020 and possibly 2021.
Democrats – with their new majority in the House of Representatives – are likely to intervene legislatively in early in 2019 but are unlikely to be successful at getting the Republican-led Senate to pass “feel-good” legislation that would protect those with preexisting conditions, something Mr. Jost said “cannot be re-created” short of reenacting the entire ACA given the complex processes and subsidies that make coverage of preexisting conditions possible.
Both chambers of Congress could work together on something as simple as reinstating the penalty – even if it were set at just $1 – to coming up with something more comprehensive, but that would be extremely challenging to make happen and to get President Trump to sign off on, he said.
As Democratic governors scramble to appeal a Dec. 14 ruling that essentially struck down the Affordable Care Act, a reprieve keeping the law intact could come by the end of the year.
The ACA remains in effect until Dec. 31 after Judge Reed O’Connor of the U.S. District Court for the Northern District of Texas ruled that the 2017 tax law that zeroed out penalties beginning in 2019 for not carrying health insurance effectively rendered the entire ACA unconstitutional.
On Dec. 17, a group of Democratic attorneys general “asked Judge O’Connor to clarify that his ruling does not presently affect any rights or responsibilities under the ACA until appellate review is complete, or alternatively to stay his ruling,” Timothy S. Jost, emeritus professor at the Washington and Lee School of Law, in Lexington, Va., said during a Dec. 18 press teleconference hosted by the Commonwealth Fund. “They also asked him to certify the case for an immediate appeal and to do so by the end of this week.”
Mr. Jost noted that Judge O’Connor asked the plaintiffs and the U.S. Department of Health & Human Services to respond to this request by Dec. 21.
“I expect him to rule, probably, on New Year’s Eve on what happens next,” Mr. Jost said.
If Judge O’Connor refuses to stay his order, the group of Democratic attorneys general is expected to appeal to the Fifth Circuit Court of Appeals for such a stay, Mr. Jost said. He noted that the Fifth Circuit is one of the more conservative courts – with five Trump administration appointees and five judges appointed by previous Democratic administrations.
In general, Mr. Jost said that he does not expect Judge O’Connor’s ruling to stand.
“The decision is so clearly wrong, however, that I believe there is a good chance that it will be reversed,” he said. “If the Fifth Circuit does, I think it is very unlikely the Supreme Court take the case.” He added that if the Fifth Circuit upholds Judge O’Connor’s ruling, the court would take the case and “very likely reverse at least 5-4 and quite possibly 6-3 on at least the issue of severability.”
Mr. Jost added that he expects the case to drag into 2020 and possibly 2021.
Democrats – with their new majority in the House of Representatives – are likely to intervene legislatively in early in 2019 but are unlikely to be successful at getting the Republican-led Senate to pass “feel-good” legislation that would protect those with preexisting conditions, something Mr. Jost said “cannot be re-created” short of reenacting the entire ACA given the complex processes and subsidies that make coverage of preexisting conditions possible.
Both chambers of Congress could work together on something as simple as reinstating the penalty – even if it were set at just $1 – to coming up with something more comprehensive, but that would be extremely challenging to make happen and to get President Trump to sign off on, he said.
As Democratic governors scramble to appeal a Dec. 14 ruling that essentially struck down the Affordable Care Act, a reprieve keeping the law intact could come by the end of the year.
The ACA remains in effect until Dec. 31 after Judge Reed O’Connor of the U.S. District Court for the Northern District of Texas ruled that the 2017 tax law that zeroed out penalties beginning in 2019 for not carrying health insurance effectively rendered the entire ACA unconstitutional.
On Dec. 17, a group of Democratic attorneys general “asked Judge O’Connor to clarify that his ruling does not presently affect any rights or responsibilities under the ACA until appellate review is complete, or alternatively to stay his ruling,” Timothy S. Jost, emeritus professor at the Washington and Lee School of Law, in Lexington, Va., said during a Dec. 18 press teleconference hosted by the Commonwealth Fund. “They also asked him to certify the case for an immediate appeal and to do so by the end of this week.”
Mr. Jost noted that Judge O’Connor asked the plaintiffs and the U.S. Department of Health & Human Services to respond to this request by Dec. 21.
“I expect him to rule, probably, on New Year’s Eve on what happens next,” Mr. Jost said.
If Judge O’Connor refuses to stay his order, the group of Democratic attorneys general is expected to appeal to the Fifth Circuit Court of Appeals for such a stay, Mr. Jost said. He noted that the Fifth Circuit is one of the more conservative courts – with five Trump administration appointees and five judges appointed by previous Democratic administrations.
In general, Mr. Jost said that he does not expect Judge O’Connor’s ruling to stand.
“The decision is so clearly wrong, however, that I believe there is a good chance that it will be reversed,” he said. “If the Fifth Circuit does, I think it is very unlikely the Supreme Court take the case.” He added that if the Fifth Circuit upholds Judge O’Connor’s ruling, the court would take the case and “very likely reverse at least 5-4 and quite possibly 6-3 on at least the issue of severability.”
Mr. Jost added that he expects the case to drag into 2020 and possibly 2021.
Democrats – with their new majority in the House of Representatives – are likely to intervene legislatively in early in 2019 but are unlikely to be successful at getting the Republican-led Senate to pass “feel-good” legislation that would protect those with preexisting conditions, something Mr. Jost said “cannot be re-created” short of reenacting the entire ACA given the complex processes and subsidies that make coverage of preexisting conditions possible.
Both chambers of Congress could work together on something as simple as reinstating the penalty – even if it were set at just $1 – to coming up with something more comprehensive, but that would be extremely challenging to make happen and to get President Trump to sign off on, he said.