User login
A Veteran With a Solitary Pulmonary Nodule
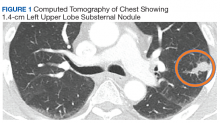
Case Presentation. A 69-year-old veteran presented with an intermittent, waxing and waning cough. He had never smoked and had no family history of lung cancer. His primary care physician ordered a chest radiograph, which revealed a nodular opacity within the lingula concerning for a parenchymal nodule. Further characterization with a chest computed tomography (CT) demonstrated a 1.4-cm left upper lobe subpleural nodule with small satellite nodules (Figure 1). Given these imaging findings, the patient was referred to the pulmonary clinic.
►Lauren Kearney, MD, Medical Resident, VA Boston Healthcare System (VABHS) and Boston Medical Center. What is the differential diagnosis of a solitary pulmonary nodule? What characteristics of the nodule do you consider to differentiate these diagnoses?
►Renda Wiener, MD, Pulmonary and Critical Care, VABHS, and Assistant Professor of Medicine, Boston University School of Medicine. Pulmonary nodules are well-defined lesions < 3 cm in diameter that are surrounded by lung parenchyma. Although cancer is a possibility (including primary lung cancers, metastatic cancers, or carcinoid tumors), most small nodules do not turn out to be malignant.1 Benign etiologies include infections, benign tumors, vascular malformations, and inflammatory conditions. Infectious causes of nodules are often granulomatous in nature, including fungi, Mycobacterium tuberculosis, and nontuberculous mycobacteria. Benign tumors are most commonly hamartomas, and these may be clearly distinguished based on imaging characteristics. Pulmonary arteriovenous malformations, hematomas, and infarcts may present as nodules as well. Inflammatory causes of nodules are important and relatively common, including granulomatosis with polyangiitis, rheumatoid arthritis, sarcoidosis, amyloidosis, and rounded atelectasis.
To distinguish benign from malignant etiologies, we look for several features of pulmonary nodules on imaging. Larger size, irregular borders, and upper lobe location all increase the likelihood of cancer, whereas solid attenuation and calcification make cancer less likely. One of the most reassuring findings that suggests a benign etiology is lack of growth over a period of surveillance; after 2 years without growth we typically consider a nodule benign.1 And of course, we also consider the patient’s symptoms and risk factors: weight loss, hemoptysis, a history of cigarette smoking or asbestos exposure, or family history of cancer all increase the likelihood of malignancy.
►Dr. Kearney. Given that the differential diagnosis is so broad, how do you think about the next step in evaluating a pulmonary nodule? How do you approach shared decision making with the patient?
►Dr. Wiener. The characteristics of the patient, the nodule, and the circumstances in which the nodule were discovered are all important to consider. Incidental pulmonary nodules are often found on chest imaging. The imaging characteristics of the nodule are important, as are the patient’s risk factors. A similarly appearing nodule can have very different implications if the patient is a never-smoker exposed to endemic fungi, or a long-time smoker enrolled in a lung cancer screening program. Consultation with a pulmonologist is often appropriate.
It’s important to note that we lack high-quality evidence on the optimal strategy to evaluate pulmonary nodules, and there is no single “right answer“ for all patients. For patients with a low risk of malignancy (< 5%-10%)—which comprises the majority of the incidental nodules discovered—we typically favor serial CT surveillance of the nodule over a period of a few years, whereas for patients at high risk of malignancy (> 65%), we favor early surgical resection if the patient is able to tolerate that. For patients with an intermediate risk of malignancy (~5%-65%), we might consider serial CT surveillance, positron emission tomography (PET) scan, or biopsy.1 The American College of Chest Physicians guidelines for pulmonary nodule evaluation recommend discussing with patients the different options and the trade-offs of these options in a shared decision-making process.1
►Dr. Kearney. The patient’s pulmonologist laid out options, including monitoring with serial CT scans, obtaining a PET scan, performing CT-guided needle biopsy, or referring for surgical excision. In this case, the patient elected to undergo CT-guided needle biopsy. Dr. Huang, can you discuss the pathology results?
►Qin Huang, MD, Pathology and Laboratory Medicine, VABHS, and Assistant Professor of Pathology, Harvard Medical School (HMS). The microscopic examination of the needle biopsy of the lung mass revealed rare clusters of atypical cells with crushed cells adjacent to an extensive area of necrosis with scarring. The atypical cells were suspicious for carcinoma. The Gomori methenamine silver (GMS) and periodic acid-Schiff (PAS) stains were negative for common bacterial and fungal microorganisms.
►Dr. Kearney. The tumor board, pulmonologist, and patient decide to move forward with video-assisted excisional biopsy with lymphadenectomy. Dr. Huang, can you interpret the pathology?
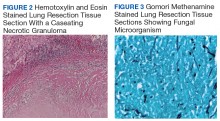
►Dr. Huang. Figure 2 showed an hemotoxylin and eosin (H&E)-stained lung resection tissue section with multiple caseating necrotic granulomas. No foreign bodies were identified. There was no evidence of malignancy. The GMS stain revealed a fungal microorganism oval with morphology typical of histoplasma capsulatum (Figure 3).
►Dr. Kearney. What are some of the different ways histoplasmosis can present? Which of these diagnoses fits this patient’s presentation?
►Judy Strymish, MD, Infectious Disease, VABHS, and Assistant Professor of Medicine, HMS. Most patients who inhale histoplasmosis spores develop asymptomatic or self-limited infection that is usually not detected. Patients at risk of symptomatic and clinically relevant disease include those who are immunocompromised, at extremes of ages, or exposed to larger inoculums. Acute pulmonary histoplasmosis can present with cough, shortness of breath, fever, chills, and less commonly, rheumatologic complaints such as erythema nodosum or erythema multiforme. Imaging often shows patchy infiltrates and enlarged mediastinal and hilar lymphadenopathy. Patients can go on to develop subacute or chronic pulmonary disease with focal opacities and mediastinal and hilar lymphadenopathy. Those with chronic disease can have cavitary lesions similar to patients with tuberculosis. Progressive disseminated histoplasmosis can develop in immunocompromised patients and disseminate through the reticuloendothelial system to other organs with the gastrointestinal tract, central nervous system, and adrenal glands.2
Pulmonary nodules are common incidental finding on chest imaging of patients who reside in histoplasmosis endemic regions, and they are often hard to differentiate from malignancies. There are 3 mediastinal manifestations: adenitis, granuloma, and fibrosis. Usually the syndromes are subclinical, but occasionally the nodes cause symptoms by impinging on other structures.2
This patient had a solitary pulmonary nodule with none of the associated features mentioned above. Pathology showed caseating granuloma and confirmed histoplasmosis.
►Dr. Kearney. Given the diagnosis of solitary histoplasmoma, how should this patient be managed?
►Dr. Strymish. The optimal therapy for histoplasmosis depends on the patient’s clinical syndrome. Most infections are self-limited and require no therapy. However, patients who are immunocompromised, exposed to large inoculum, and have progressive disease require antifungal treatment, usually with itraconazole for mild-to-moderate disease and a combination of azole therapy and amphotericin B with extensive disease. Patients with few solitary pulmonary nodules do not benefit from antifungal therapy as the nodule could represent quiescent disease that is unlikely to have clinical impact; in this case, the treatment would be higher risk than the nodule.3
►Dr. Kearney. While the discussion of the diagnosis is interesting, it is also important to acknowledge what the patient went through to arrive at this, an essentially benign diagnosis: 8 months, multiple imaging studies, and 2 invasive diagnostic procedures. Further, the patient had to grapple with the possibility of a diagnosis of cancer. Dr. Wiener, can you talk about the challenges in communicating with patients about pulmonary nodules when cancer is on the differential? What are some of the harms patients face and how can clinicians work to mitigate these harms?
►Dr. Wiener. My colleague Dr. Christopher Slatore of the Portland VA Medical Center and I studied communication about pulmonary nodules in a series of surveys and qualitative studies of patients with pulmonary nodules and the clinicians who take care of them. We found that there seems to be a disconnect between patients’ perceptions of pulmonary nodules and their clinicians, often due to inadequate communication about the nodule. Many clinicians indicated that they do not tell patients about the chance that a nodule may be cancer, because the clinicians know that cancer is unlikely (< 5% of incidentally detected pulmonary nodules turn out to be malignant), and they do not want to alarm patients unnecessarily. However, we found that patients almost immediately wondered about cancer when they learned about their pulmonary nodule, and without hearing explicitly from their clinician that cancer was unlikely, patients tended to overestimate the likelihood of a malignant nodule. Moreover, patients often were not told much about the evaluation plan for the nodule or the rationale for CT surveillance of small nodules instead of biopsy. This uncertainty about the risk of cancer and the plan for evaluating the nodule was difficult for some patients to live with; we found that about one-quarter of patients with a small pulmonary nodule experienced mild-moderate distress during the period of radiographic surveillance. Reassuringly, high-quality patient-clinician communication was associated with lower distress and higher adherence to pulmonary nodule evaluation.4
►Dr. Kearney. The patient was educated about his diagnosis of solitary histoplasmoma. Given that the patient was otherwise well appearing with no complicating factors, he was not treated with antifungal therapy. After an 8-month-long workup, the patient was relieved to receive a diagnosis that excluded cancer and did not require any further treatment. His case provides a good example of how to proceed in the workup of a solitary pulmonary nodule and on the importance of communication and shared decision making with our patients.
1. Gould MK, Donington J, Lynch WR, et al. Evaluation of individuals with pulmonary nodules: when is it lung cancer? Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2013;143(suppl 5):e93S-e120S.
2. Azar MM, Hage CA. Clinical perspectives in the diagnosis and management of histoplasmosis. Clin Chest Med. 2017;38(3):403-415.
3. Wheat LJ, Freifeld A, Kleiman MB, et al. Clinical practice guidelines for the management of patients with histoplasmosis: 2007 update by the Infectious Diseases Society of America. Clin Infect Dis. 2007;45(7):807-825.
4. Slatore CG, Wiener RS. Pulmonary nodules: a small problem for many, severe distress for some, and how to communicate about it. Chest. 2018;153(4):1004-1015.
Case Presentation. A 69-year-old veteran presented with an intermittent, waxing and waning cough. He had never smoked and had no family history of lung cancer. His primary care physician ordered a chest radiograph, which revealed a nodular opacity within the lingula concerning for a parenchymal nodule. Further characterization with a chest computed tomography (CT) demonstrated a 1.4-cm left upper lobe subpleural nodule with small satellite nodules (Figure 1). Given these imaging findings, the patient was referred to the pulmonary clinic.
►Lauren Kearney, MD, Medical Resident, VA Boston Healthcare System (VABHS) and Boston Medical Center. What is the differential diagnosis of a solitary pulmonary nodule? What characteristics of the nodule do you consider to differentiate these diagnoses?
►Renda Wiener, MD, Pulmonary and Critical Care, VABHS, and Assistant Professor of Medicine, Boston University School of Medicine. Pulmonary nodules are well-defined lesions < 3 cm in diameter that are surrounded by lung parenchyma. Although cancer is a possibility (including primary lung cancers, metastatic cancers, or carcinoid tumors), most small nodules do not turn out to be malignant.1 Benign etiologies include infections, benign tumors, vascular malformations, and inflammatory conditions. Infectious causes of nodules are often granulomatous in nature, including fungi, Mycobacterium tuberculosis, and nontuberculous mycobacteria. Benign tumors are most commonly hamartomas, and these may be clearly distinguished based on imaging characteristics. Pulmonary arteriovenous malformations, hematomas, and infarcts may present as nodules as well. Inflammatory causes of nodules are important and relatively common, including granulomatosis with polyangiitis, rheumatoid arthritis, sarcoidosis, amyloidosis, and rounded atelectasis.
To distinguish benign from malignant etiologies, we look for several features of pulmonary nodules on imaging. Larger size, irregular borders, and upper lobe location all increase the likelihood of cancer, whereas solid attenuation and calcification make cancer less likely. One of the most reassuring findings that suggests a benign etiology is lack of growth over a period of surveillance; after 2 years without growth we typically consider a nodule benign.1 And of course, we also consider the patient’s symptoms and risk factors: weight loss, hemoptysis, a history of cigarette smoking or asbestos exposure, or family history of cancer all increase the likelihood of malignancy.
►Dr. Kearney. Given that the differential diagnosis is so broad, how do you think about the next step in evaluating a pulmonary nodule? How do you approach shared decision making with the patient?
►Dr. Wiener. The characteristics of the patient, the nodule, and the circumstances in which the nodule were discovered are all important to consider. Incidental pulmonary nodules are often found on chest imaging. The imaging characteristics of the nodule are important, as are the patient’s risk factors. A similarly appearing nodule can have very different implications if the patient is a never-smoker exposed to endemic fungi, or a long-time smoker enrolled in a lung cancer screening program. Consultation with a pulmonologist is often appropriate.
It’s important to note that we lack high-quality evidence on the optimal strategy to evaluate pulmonary nodules, and there is no single “right answer“ for all patients. For patients with a low risk of malignancy (< 5%-10%)—which comprises the majority of the incidental nodules discovered—we typically favor serial CT surveillance of the nodule over a period of a few years, whereas for patients at high risk of malignancy (> 65%), we favor early surgical resection if the patient is able to tolerate that. For patients with an intermediate risk of malignancy (~5%-65%), we might consider serial CT surveillance, positron emission tomography (PET) scan, or biopsy.1 The American College of Chest Physicians guidelines for pulmonary nodule evaluation recommend discussing with patients the different options and the trade-offs of these options in a shared decision-making process.1
►Dr. Kearney. The patient’s pulmonologist laid out options, including monitoring with serial CT scans, obtaining a PET scan, performing CT-guided needle biopsy, or referring for surgical excision. In this case, the patient elected to undergo CT-guided needle biopsy. Dr. Huang, can you discuss the pathology results?
►Qin Huang, MD, Pathology and Laboratory Medicine, VABHS, and Assistant Professor of Pathology, Harvard Medical School (HMS). The microscopic examination of the needle biopsy of the lung mass revealed rare clusters of atypical cells with crushed cells adjacent to an extensive area of necrosis with scarring. The atypical cells were suspicious for carcinoma. The Gomori methenamine silver (GMS) and periodic acid-Schiff (PAS) stains were negative for common bacterial and fungal microorganisms.
►Dr. Kearney. The tumor board, pulmonologist, and patient decide to move forward with video-assisted excisional biopsy with lymphadenectomy. Dr. Huang, can you interpret the pathology?
►Dr. Huang. Figure 2 showed an hemotoxylin and eosin (H&E)-stained lung resection tissue section with multiple caseating necrotic granulomas. No foreign bodies were identified. There was no evidence of malignancy. The GMS stain revealed a fungal microorganism oval with morphology typical of histoplasma capsulatum (Figure 3).
►Dr. Kearney. What are some of the different ways histoplasmosis can present? Which of these diagnoses fits this patient’s presentation?
►Judy Strymish, MD, Infectious Disease, VABHS, and Assistant Professor of Medicine, HMS. Most patients who inhale histoplasmosis spores develop asymptomatic or self-limited infection that is usually not detected. Patients at risk of symptomatic and clinically relevant disease include those who are immunocompromised, at extremes of ages, or exposed to larger inoculums. Acute pulmonary histoplasmosis can present with cough, shortness of breath, fever, chills, and less commonly, rheumatologic complaints such as erythema nodosum or erythema multiforme. Imaging often shows patchy infiltrates and enlarged mediastinal and hilar lymphadenopathy. Patients can go on to develop subacute or chronic pulmonary disease with focal opacities and mediastinal and hilar lymphadenopathy. Those with chronic disease can have cavitary lesions similar to patients with tuberculosis. Progressive disseminated histoplasmosis can develop in immunocompromised patients and disseminate through the reticuloendothelial system to other organs with the gastrointestinal tract, central nervous system, and adrenal glands.2
Pulmonary nodules are common incidental finding on chest imaging of patients who reside in histoplasmosis endemic regions, and they are often hard to differentiate from malignancies. There are 3 mediastinal manifestations: adenitis, granuloma, and fibrosis. Usually the syndromes are subclinical, but occasionally the nodes cause symptoms by impinging on other structures.2
This patient had a solitary pulmonary nodule with none of the associated features mentioned above. Pathology showed caseating granuloma and confirmed histoplasmosis.
►Dr. Kearney. Given the diagnosis of solitary histoplasmoma, how should this patient be managed?
►Dr. Strymish. The optimal therapy for histoplasmosis depends on the patient’s clinical syndrome. Most infections are self-limited and require no therapy. However, patients who are immunocompromised, exposed to large inoculum, and have progressive disease require antifungal treatment, usually with itraconazole for mild-to-moderate disease and a combination of azole therapy and amphotericin B with extensive disease. Patients with few solitary pulmonary nodules do not benefit from antifungal therapy as the nodule could represent quiescent disease that is unlikely to have clinical impact; in this case, the treatment would be higher risk than the nodule.3
►Dr. Kearney. While the discussion of the diagnosis is interesting, it is also important to acknowledge what the patient went through to arrive at this, an essentially benign diagnosis: 8 months, multiple imaging studies, and 2 invasive diagnostic procedures. Further, the patient had to grapple with the possibility of a diagnosis of cancer. Dr. Wiener, can you talk about the challenges in communicating with patients about pulmonary nodules when cancer is on the differential? What are some of the harms patients face and how can clinicians work to mitigate these harms?
►Dr. Wiener. My colleague Dr. Christopher Slatore of the Portland VA Medical Center and I studied communication about pulmonary nodules in a series of surveys and qualitative studies of patients with pulmonary nodules and the clinicians who take care of them. We found that there seems to be a disconnect between patients’ perceptions of pulmonary nodules and their clinicians, often due to inadequate communication about the nodule. Many clinicians indicated that they do not tell patients about the chance that a nodule may be cancer, because the clinicians know that cancer is unlikely (< 5% of incidentally detected pulmonary nodules turn out to be malignant), and they do not want to alarm patients unnecessarily. However, we found that patients almost immediately wondered about cancer when they learned about their pulmonary nodule, and without hearing explicitly from their clinician that cancer was unlikely, patients tended to overestimate the likelihood of a malignant nodule. Moreover, patients often were not told much about the evaluation plan for the nodule or the rationale for CT surveillance of small nodules instead of biopsy. This uncertainty about the risk of cancer and the plan for evaluating the nodule was difficult for some patients to live with; we found that about one-quarter of patients with a small pulmonary nodule experienced mild-moderate distress during the period of radiographic surveillance. Reassuringly, high-quality patient-clinician communication was associated with lower distress and higher adherence to pulmonary nodule evaluation.4
►Dr. Kearney. The patient was educated about his diagnosis of solitary histoplasmoma. Given that the patient was otherwise well appearing with no complicating factors, he was not treated with antifungal therapy. After an 8-month-long workup, the patient was relieved to receive a diagnosis that excluded cancer and did not require any further treatment. His case provides a good example of how to proceed in the workup of a solitary pulmonary nodule and on the importance of communication and shared decision making with our patients.
Case Presentation. A 69-year-old veteran presented with an intermittent, waxing and waning cough. He had never smoked and had no family history of lung cancer. His primary care physician ordered a chest radiograph, which revealed a nodular opacity within the lingula concerning for a parenchymal nodule. Further characterization with a chest computed tomography (CT) demonstrated a 1.4-cm left upper lobe subpleural nodule with small satellite nodules (Figure 1). Given these imaging findings, the patient was referred to the pulmonary clinic.
►Lauren Kearney, MD, Medical Resident, VA Boston Healthcare System (VABHS) and Boston Medical Center. What is the differential diagnosis of a solitary pulmonary nodule? What characteristics of the nodule do you consider to differentiate these diagnoses?
►Renda Wiener, MD, Pulmonary and Critical Care, VABHS, and Assistant Professor of Medicine, Boston University School of Medicine. Pulmonary nodules are well-defined lesions < 3 cm in diameter that are surrounded by lung parenchyma. Although cancer is a possibility (including primary lung cancers, metastatic cancers, or carcinoid tumors), most small nodules do not turn out to be malignant.1 Benign etiologies include infections, benign tumors, vascular malformations, and inflammatory conditions. Infectious causes of nodules are often granulomatous in nature, including fungi, Mycobacterium tuberculosis, and nontuberculous mycobacteria. Benign tumors are most commonly hamartomas, and these may be clearly distinguished based on imaging characteristics. Pulmonary arteriovenous malformations, hematomas, and infarcts may present as nodules as well. Inflammatory causes of nodules are important and relatively common, including granulomatosis with polyangiitis, rheumatoid arthritis, sarcoidosis, amyloidosis, and rounded atelectasis.
To distinguish benign from malignant etiologies, we look for several features of pulmonary nodules on imaging. Larger size, irregular borders, and upper lobe location all increase the likelihood of cancer, whereas solid attenuation and calcification make cancer less likely. One of the most reassuring findings that suggests a benign etiology is lack of growth over a period of surveillance; after 2 years without growth we typically consider a nodule benign.1 And of course, we also consider the patient’s symptoms and risk factors: weight loss, hemoptysis, a history of cigarette smoking or asbestos exposure, or family history of cancer all increase the likelihood of malignancy.
►Dr. Kearney. Given that the differential diagnosis is so broad, how do you think about the next step in evaluating a pulmonary nodule? How do you approach shared decision making with the patient?
►Dr. Wiener. The characteristics of the patient, the nodule, and the circumstances in which the nodule were discovered are all important to consider. Incidental pulmonary nodules are often found on chest imaging. The imaging characteristics of the nodule are important, as are the patient’s risk factors. A similarly appearing nodule can have very different implications if the patient is a never-smoker exposed to endemic fungi, or a long-time smoker enrolled in a lung cancer screening program. Consultation with a pulmonologist is often appropriate.
It’s important to note that we lack high-quality evidence on the optimal strategy to evaluate pulmonary nodules, and there is no single “right answer“ for all patients. For patients with a low risk of malignancy (< 5%-10%)—which comprises the majority of the incidental nodules discovered—we typically favor serial CT surveillance of the nodule over a period of a few years, whereas for patients at high risk of malignancy (> 65%), we favor early surgical resection if the patient is able to tolerate that. For patients with an intermediate risk of malignancy (~5%-65%), we might consider serial CT surveillance, positron emission tomography (PET) scan, or biopsy.1 The American College of Chest Physicians guidelines for pulmonary nodule evaluation recommend discussing with patients the different options and the trade-offs of these options in a shared decision-making process.1
►Dr. Kearney. The patient’s pulmonologist laid out options, including monitoring with serial CT scans, obtaining a PET scan, performing CT-guided needle biopsy, or referring for surgical excision. In this case, the patient elected to undergo CT-guided needle biopsy. Dr. Huang, can you discuss the pathology results?
►Qin Huang, MD, Pathology and Laboratory Medicine, VABHS, and Assistant Professor of Pathology, Harvard Medical School (HMS). The microscopic examination of the needle biopsy of the lung mass revealed rare clusters of atypical cells with crushed cells adjacent to an extensive area of necrosis with scarring. The atypical cells were suspicious for carcinoma. The Gomori methenamine silver (GMS) and periodic acid-Schiff (PAS) stains were negative for common bacterial and fungal microorganisms.
►Dr. Kearney. The tumor board, pulmonologist, and patient decide to move forward with video-assisted excisional biopsy with lymphadenectomy. Dr. Huang, can you interpret the pathology?
►Dr. Huang. Figure 2 showed an hemotoxylin and eosin (H&E)-stained lung resection tissue section with multiple caseating necrotic granulomas. No foreign bodies were identified. There was no evidence of malignancy. The GMS stain revealed a fungal microorganism oval with morphology typical of histoplasma capsulatum (Figure 3).
►Dr. Kearney. What are some of the different ways histoplasmosis can present? Which of these diagnoses fits this patient’s presentation?
►Judy Strymish, MD, Infectious Disease, VABHS, and Assistant Professor of Medicine, HMS. Most patients who inhale histoplasmosis spores develop asymptomatic or self-limited infection that is usually not detected. Patients at risk of symptomatic and clinically relevant disease include those who are immunocompromised, at extremes of ages, or exposed to larger inoculums. Acute pulmonary histoplasmosis can present with cough, shortness of breath, fever, chills, and less commonly, rheumatologic complaints such as erythema nodosum or erythema multiforme. Imaging often shows patchy infiltrates and enlarged mediastinal and hilar lymphadenopathy. Patients can go on to develop subacute or chronic pulmonary disease with focal opacities and mediastinal and hilar lymphadenopathy. Those with chronic disease can have cavitary lesions similar to patients with tuberculosis. Progressive disseminated histoplasmosis can develop in immunocompromised patients and disseminate through the reticuloendothelial system to other organs with the gastrointestinal tract, central nervous system, and adrenal glands.2
Pulmonary nodules are common incidental finding on chest imaging of patients who reside in histoplasmosis endemic regions, and they are often hard to differentiate from malignancies. There are 3 mediastinal manifestations: adenitis, granuloma, and fibrosis. Usually the syndromes are subclinical, but occasionally the nodes cause symptoms by impinging on other structures.2
This patient had a solitary pulmonary nodule with none of the associated features mentioned above. Pathology showed caseating granuloma and confirmed histoplasmosis.
►Dr. Kearney. Given the diagnosis of solitary histoplasmoma, how should this patient be managed?
►Dr. Strymish. The optimal therapy for histoplasmosis depends on the patient’s clinical syndrome. Most infections are self-limited and require no therapy. However, patients who are immunocompromised, exposed to large inoculum, and have progressive disease require antifungal treatment, usually with itraconazole for mild-to-moderate disease and a combination of azole therapy and amphotericin B with extensive disease. Patients with few solitary pulmonary nodules do not benefit from antifungal therapy as the nodule could represent quiescent disease that is unlikely to have clinical impact; in this case, the treatment would be higher risk than the nodule.3
►Dr. Kearney. While the discussion of the diagnosis is interesting, it is also important to acknowledge what the patient went through to arrive at this, an essentially benign diagnosis: 8 months, multiple imaging studies, and 2 invasive diagnostic procedures. Further, the patient had to grapple with the possibility of a diagnosis of cancer. Dr. Wiener, can you talk about the challenges in communicating with patients about pulmonary nodules when cancer is on the differential? What are some of the harms patients face and how can clinicians work to mitigate these harms?
►Dr. Wiener. My colleague Dr. Christopher Slatore of the Portland VA Medical Center and I studied communication about pulmonary nodules in a series of surveys and qualitative studies of patients with pulmonary nodules and the clinicians who take care of them. We found that there seems to be a disconnect between patients’ perceptions of pulmonary nodules and their clinicians, often due to inadequate communication about the nodule. Many clinicians indicated that they do not tell patients about the chance that a nodule may be cancer, because the clinicians know that cancer is unlikely (< 5% of incidentally detected pulmonary nodules turn out to be malignant), and they do not want to alarm patients unnecessarily. However, we found that patients almost immediately wondered about cancer when they learned about their pulmonary nodule, and without hearing explicitly from their clinician that cancer was unlikely, patients tended to overestimate the likelihood of a malignant nodule. Moreover, patients often were not told much about the evaluation plan for the nodule or the rationale for CT surveillance of small nodules instead of biopsy. This uncertainty about the risk of cancer and the plan for evaluating the nodule was difficult for some patients to live with; we found that about one-quarter of patients with a small pulmonary nodule experienced mild-moderate distress during the period of radiographic surveillance. Reassuringly, high-quality patient-clinician communication was associated with lower distress and higher adherence to pulmonary nodule evaluation.4
►Dr. Kearney. The patient was educated about his diagnosis of solitary histoplasmoma. Given that the patient was otherwise well appearing with no complicating factors, he was not treated with antifungal therapy. After an 8-month-long workup, the patient was relieved to receive a diagnosis that excluded cancer and did not require any further treatment. His case provides a good example of how to proceed in the workup of a solitary pulmonary nodule and on the importance of communication and shared decision making with our patients.
1. Gould MK, Donington J, Lynch WR, et al. Evaluation of individuals with pulmonary nodules: when is it lung cancer? Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2013;143(suppl 5):e93S-e120S.
2. Azar MM, Hage CA. Clinical perspectives in the diagnosis and management of histoplasmosis. Clin Chest Med. 2017;38(3):403-415.
3. Wheat LJ, Freifeld A, Kleiman MB, et al. Clinical practice guidelines for the management of patients with histoplasmosis: 2007 update by the Infectious Diseases Society of America. Clin Infect Dis. 2007;45(7):807-825.
4. Slatore CG, Wiener RS. Pulmonary nodules: a small problem for many, severe distress for some, and how to communicate about it. Chest. 2018;153(4):1004-1015.
1. Gould MK, Donington J, Lynch WR, et al. Evaluation of individuals with pulmonary nodules: when is it lung cancer? Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2013;143(suppl 5):e93S-e120S.
2. Azar MM, Hage CA. Clinical perspectives in the diagnosis and management of histoplasmosis. Clin Chest Med. 2017;38(3):403-415.
3. Wheat LJ, Freifeld A, Kleiman MB, et al. Clinical practice guidelines for the management of patients with histoplasmosis: 2007 update by the Infectious Diseases Society of America. Clin Infect Dis. 2007;45(7):807-825.
4. Slatore CG, Wiener RS. Pulmonary nodules: a small problem for many, severe distress for some, and how to communicate about it. Chest. 2018;153(4):1004-1015.
A Veteran With Fibromyalgia Presenting With Dyspnea
Case Presentation. A 64-year-old US Army veteran with a history of colorectal cancer, melanoma, and fibrinolytic presented with dyspnea to VA Boston Healthcare System (VABHS). Seven years prior to the current presentation, at the time of her diagnosis of colorectal cancer, the patient was found to be HIV negative but to have a positive purified protein derivative (PPD) test. She was treated with isoniazid (INH) therapy for 9 months. Sputum cultures collected prior to initiation of therapy grew Mycobacterium avium complex (MAC) in 1 of 3 samples, with these results reported several months after initiation of therapy. She was a never smoker with no known travel or exposure. At the time of the current presentation, her medications included bupropion, levothyroxine, capsaicin, cyclobenzaprine, ibuprofen, and acetaminophen.
►Lakshmana Swamy, MD, Chief Medical Resident, VABHS and Boston Medical Center. Dr. Monach, this patient is on a variety of pain medications and has a diagnosis of fibromyalgia. This diagnosis often frustrates doctors and patients alike. Can you tell us about fibromyalgia from the rheumatologist’s perspective and what you think of her current treatment regimen?
►Paul A. Monach, MD, PhD, Chief, Section of Rheumatology, VABHS and Associate Professor of Medicine, Boston University School of Medicine. Fibromyalgia is a syndrome of chronic widespread pain without known pathology in the musculoskeletal system. It is thought to be caused by chronic dysfunction of pain-processing pathways in the central nervous system (CNS). It is often accompanied by other somatic symptoms such as chronic fatigue, irritable bowel syndrome, and bladder pain. It is a common condition, affecting up to 5% of otherwise healthy women. It is particularly common in persons with chronic nonrestorative sleep or posttraumatic stress disorder from a wide range of causes. However, it also is more common in persons with autoimmune inflammatory diseases, such as lupus, Sjögren syndrome, or rheumatoid arthritis. Concern for one of these diseases is the main reason to consider referring a patient for evaluation by a rheumatologist. Often rheumatologists participate in the management of fibromyalgia. A patient should be given appropriate expectations by the referring physician.
Effectiveness of treatment varies widely among patients. Nonpharmacologic approaches such as aerobic exercise, cognitive behavioral therapy, and tai chi have support from clinical trials, and yoga and aquatherapy also are widely used.1,2 The classes of drugs used are the same as for neuropathic pain: tricyclics, including cyclobenzaprine; serotonin and norepinephrine reuptake inhibitors (SNRIs); and gabapentinoids. In contrast, nonsteroidal anti-inflammatory drugs and opioids are ineffective unless there is a superimposed mechanical or inflammatory cause in the periphery. The key point is that continuation of any treatment should be based entirely on the patient’s own assessment of benefit.
►Dr. Swamy. Seven years later, the patient returned to her primary care provider, reporting increased dyspnea on exertion as well as significant fatigue. She was referred to the pulmonary department and had repeat computed tomography (CT) scans of the chest, which indicated persistent right middle lobe (RML) bronchiectasis. She then underwent bronchoscopy with a subsequent bronchoalveolar lavage (BAL) culture growing MAC. Dr. Fine, please interpret the baseline and follow-up CT scans and help us understand the significance of the MAC on sputum and BAL cultures.
►Alan Fine, MD, Section of Pulmonary and Critical Care, VABHS and Professor of Medicine, Boston University School of Medicine. Prior to this presentation, the patient had a pleural-based area of fibrosis with possible associated RML bronchiectasis. This appears to be a postinflammatory process without classic features of malignant or metastatic disease. She then had a sputum, which grew MAC in only 1 of 3 samples and in liquid media only. Importantly, the sputum was not smear positive. All of this suggests a low organism burden. One possibility is that this could reflect colonization with MAC; it is not uncommon for patients with underlying chronic changes in their lung to grow MAC, and it is often difficult to tell whether it is indicative of active disease. Structural lung disease, such as bronchiectasis, predisposes a patient to MAC, but chronic MAC also may cause bronchiectasis. This chicken-and-egg scenario comes up frequently. She may have a MAC infection, but as she is HIV negative and asymptomatic, there is no urgent indication to treat, especially as the burden of therapy is not insignificant.
►Dr. Swamy. Do we need to worry about Mycobacterium tuberculosis (MTB)?
►Dr. Fine. Although she was previously PPD positive, she had already completed 1 year of isoniazid (INH) therapy, making active MTB less likely. From an infection control standpoint, it is important to distinguish MAC from MTB. The former is not contagious, and there is no need for airborne isolation.
►Dr. Swamy. Dr. Fine, where does MAC come from? Does it commonly cause disease?
►Dr. Fine. In the environment, MAC is nearly ubiquitous , especially in water and soil. In one study, 20% of showerheads were positive for MAC; when patients are infected, we may suggest changing/bleaching the showerhead, but there are no definitive recommendations.3 Because MAC is so common in the environment, it is unlikely that measures to target MAC colonization will be clinically meaningful. On the other hand, the incidence of nontuberculous mycobacterial infections is increasing across the US, and it may be a common and frequently underdiagnosed cause of chronic cough, especially in postmenopausal women.
►Dr. Swamy. Four years prior to the current presentation, the patient developed a cough after an upper respiratory tract infection that persisted for more than 2 weeks. Given her history, she underwent a repeat chest CT, which noted a slight increase in nodularity and ground-glass opacity restricted to the RML. She also reported dyspnea on exertion and was referred to the pulmonary medicine department. By the time she arrived, her dyspnea had largely resolved, but she reported persistent fatigue without other systemic symptoms, such as fevers or chills. Dr. Fine, does MAC explain this patient’s dyspnea?
►Dr. Fine. As her pulmonary symptoms resolved in a short period of time with only azithromycin, it is very unlikely that her symptoms were related to her prior disease. The MAC infection is not likely to cause dyspnea on exertion and fatigue and should be worked up more broadly before attributing it to MAC. In view of this, it would not be unreasonable to follow her clinically and see her again in 6 to 8 weeks. In this context, we also should consider the untoward impact of repeated radiation exposure derived from multiple CT scans. When a patient has an abnormality on CT scan, it often leads to further scans even if the symptoms do not match the previous findings, as in this case.
►Dr. Swamy. Given her ongoing fatigue and systemic symptoms (morning stiffness of the shoulders, legs, and thighs, and leg cramps), she was referred to the rheumatology department where the physical examination revealed muscle tenderness in her proximal arms and legs with normal strength, tender points at the elbows and medial side of the bilateral knees, significant tenderness of lower legs, and no synovitis.
Dr. Monach, can you walk us through your approach to this patient? Are we seeing manifestations of fibromyalgia? What diagnoses concerns you and how would you proceed?
►Dr. Monach. The history and exam are most helpful in raising or reducing suspicion for an underlying inflammatory disease. Areas of tenderness described in her case are typical of fibromyalgia, although it can be difficult to interpret symptoms in the hip girdle and shoulder girdle because objective findings are often absent on exam in patients with inflammatory arthritis or bursitis. Similarly, tenderness at sites of tendon insertion (enthuses) without objective abnormalities is common in different forms of spondyloarthritis, so tenderness at the elbow, knee, lateral hip, and low back can be difficult to interpret. What this patient is lacking is prominent subjective or objective findings in the joints most commonly affected in rheumatoid arthritis and lupus: wrists, hands, ankles, and feet.
►Dr. Swamy. Initial laboratory data include an erythrocyte sedimentation rate of 79 with a normal C-reactive protein. A tentative diagnosis of polymyalgia rheumatic is made with consideration of a trial treatment of prednisone.
Dr. Monach, this patient has an indolent infection and is about to be given glucocorticoids. Could you describe the situations in which you feel that glucocorticoids cause a relative immunosuppression?
►Dr. Monach. Glucocorticoids are considered safe in a patient whose infection is not intrinsically dangerous or who has started appropriate antibiotics for that infection. Although all toxicities of glucocorticoids are dose dependent, the long-standing assertion that doses below 10 mg to 15 mg do not increase risk of infection is contradicted by data published in the past 10 to 15 years, with the caveat that these patients were on long-term treatment.
►Dr. Swamy. The patient was started on prednisone 15 mg per day for 15 days. She returned to the clinic after 1 week of prednisone troutment and noted “significant improvement in fatigue, morning stiffness of shoulders, thighs, leg, back is better, leg cramps resolved, shooting pain in many joints resolved.” Further laboratory results were notable for a negative rheumatoid factor, negative antinuclear antibody, and a cyclic citrullinated peptide of 60. A presumptive diagnosis of rheumatoid arthritis (RA) was made and plaquenil 200 mg twice daily was started.
Dr. Monach, can you explain why RA comes up now on serology but was not considered initially? Why does this presentation fit RA, and was her response to treatment typical? How does this fit in with her previous diagnosis of fibromyalgia? Was that just an atypical, indolent presentation of RA?
►Dr. Monach. Though her presentation is atypical for RA, in elderly patients, RA can present with symptoms resembling polymyalgia rheumatica. The question is whether she had RA all along (in which case “elderly onset” would not apply) or had fibromyalgia and developed RA more recently. The response to empiric glucocorticoid therapy is helpful, since fibromyalgia should not improve with prednisone even in a patient with RA unless treatment of RA would allow better sleep and ability to exercise. Rheumatoid arthritis typically responds very well to prednisone in the 5-mg to 15-mg range.
►Dr. Swamy. Given the new diagnosis of an inflammatory arthritis requiring immunosuppression, bronchoscopy with BAL is performed to evaluate for the presence of MAC. These cultures were positive for MAC.
Dr. Fine, does the positive BAL culture indicate an active MAC infection?
►Dr. Fine. Yes, based on these updated data, the patient has an active MAC infection. Active infection is defined as symptoms or imaging consistent with the diagnosis, supporting microbiology data (either 2 sputum or 1 BAL sample growing MAC) and the exclusion of other causes. Previously, this patient grew MAC in just one expectorated sputum; this did not meet the microbiologic criteria. Now sputum has grown in the BAL sample; along with the CT imaging, this is enough to diagnosis active MAC infection.
Treatment for MAC must consider the details of each case. First, this is not an emergency; treatment decisions should be made with the rheumatologist to consider the planned immunosuppression. For example, we must consider potential drug interactions. A specific point should be made of the use of tumor necrosis factor (TNF)-α inhibition, which data indicate can reactivate TB and may inhibit mechanisms that restrain mycobacterial disease. Serious cases of MAC infection have been reported in the literature in the setting of TNF-α inhibition.5,6 Despite these concerns, there is not a contraindication to using these therapies from the perspective of the active MAC disease. All of these decisions will impact the need to commit the patient to MAC therapy.
►Dr. Swamy. Dr. Fine, what do you consider prior to initiating MAC therapy?
► Dr. Fine. The decision to pursue MAC therapy should not be taken lightly. Therapy often entails prolonged multidrug regimens, usually spanning more than a year, with frequent adverse effects. Outside of very specific cases, such as TNF-β inhibition, MAC is rarely a life-threatening disease, so the benefit may be limited. Treatment for MAC is certainly unlikely to be fruitful without a diligent and motivated patient able to handle the high and prolonged pill burden. Of note, it is also important to keep this patient up-to-date with influenza and pneumonia vaccination given her structural lung disease.
►Dr. Swamy. The decision is made to treat MAC with azithromycin, rifampin, and ethambutol. The disease is noted to be nonfibrocavitary. The patient underwent monthly liver function test monitoring and visual acuity testing, which were unremarkable. Dr. Fine, can you describe the phenotypes of nontuberculous mycobacterial (NTM) disease?
►Dr. Fine. There are 3 main phenotypes of NTM.3 First, we see the elderly man with preexisting lung disease—usually chronic obstructive pulmonary disease—with fibrocavitary and/or reticulonodular appearance. Second, we see the slim, elderly woman often without any preexisting lung disease presenting with focal bronchiectasis and nodular lesions in right middle lobe and lingula—the Lady Windermere syndrome. This eponym is derived from Oscar Wilde’s play “Lady Windermere’s Fan, a Play About a Good Woman,” and was first associated with this disease in 1992.7 At the time, it was thought that the voluntary suppression of cough led to poorly draining lung regions, vulnerable to engraftment by atypical mycobacteria. Infection with atypical mycobacteria are associated with this population; however, it is no longer thought to be due to the voluntary suppression of cough.7,8 Third, we do occasionally see atypical presentations, such as focal masses and solitary nodules.
►Dr. Swamy. At 1-year follow-up she successfully completed MAC therapy and noted ongoing control of rheumatoid symptoms.
1. Bernardy K, Klose P, Welsch P, Häuser W. Efficacy, acceptability and safety of cognitive behavioural therapies in fibromyalgia syndrome—a systematic review and meta-analysis of randomized controlled trials. Eur J Pain. 2018;22(2):242-260.
2. Geneen LJ, Moore RA, Clarke C, Martin D, Colvin LA, Smith BH. Physical activity and exercise for chronic pain in adults: an overview of Cochrane Reviews. Cochrane Database Syst Rev. 2017;4:CD011279.
3. Aksamit TR, Philley JV, Griffith DE. Nontuberculous mycobacterial (NTM) lung disease: the top ten essentials. Respir Med. 2014;108(3):417-425.
4. Aucott JN. Glucocorticoids and infection. Endocrinol Metab Clin North Am. 1994;23(3):655-670.
5. Curtis JR, Yang S, Patkar NM, et al. Risk of hospitalized bacterial infections associated with biologic treatment among US veterans with rheumatoid arthritis. Arthritis Care Res (Hoboken). 2014;66(7):990-997.
6. Lane MA, McDonald JR, Zeringue AL, et al. TNF-α antagonist use and risk of hospitalization for infection in a national cohort of veterans with rheumatoid arthritis. Medicine (Baltimore). 2011;90(2):139-145.
7. Reich JM, Johnson RE. Mycobacterium avium complex pulmonary disease presenting as an isolated lingular or middle lobe pattern. The Lady Windermere syndrome. Chest. 1992;101(6):1605-1609.
8. Kasthoori JJ, Liam CK, Wastie ML. Lady Windermere syndrome: an inappropriate eponym for an increasingly important condition. Singapore Med J. 2008;49(2):e47-e49.
Case Presentation. A 64-year-old US Army veteran with a history of colorectal cancer, melanoma, and fibrinolytic presented with dyspnea to VA Boston Healthcare System (VABHS). Seven years prior to the current presentation, at the time of her diagnosis of colorectal cancer, the patient was found to be HIV negative but to have a positive purified protein derivative (PPD) test. She was treated with isoniazid (INH) therapy for 9 months. Sputum cultures collected prior to initiation of therapy grew Mycobacterium avium complex (MAC) in 1 of 3 samples, with these results reported several months after initiation of therapy. She was a never smoker with no known travel or exposure. At the time of the current presentation, her medications included bupropion, levothyroxine, capsaicin, cyclobenzaprine, ibuprofen, and acetaminophen.
►Lakshmana Swamy, MD, Chief Medical Resident, VABHS and Boston Medical Center. Dr. Monach, this patient is on a variety of pain medications and has a diagnosis of fibromyalgia. This diagnosis often frustrates doctors and patients alike. Can you tell us about fibromyalgia from the rheumatologist’s perspective and what you think of her current treatment regimen?
►Paul A. Monach, MD, PhD, Chief, Section of Rheumatology, VABHS and Associate Professor of Medicine, Boston University School of Medicine. Fibromyalgia is a syndrome of chronic widespread pain without known pathology in the musculoskeletal system. It is thought to be caused by chronic dysfunction of pain-processing pathways in the central nervous system (CNS). It is often accompanied by other somatic symptoms such as chronic fatigue, irritable bowel syndrome, and bladder pain. It is a common condition, affecting up to 5% of otherwise healthy women. It is particularly common in persons with chronic nonrestorative sleep or posttraumatic stress disorder from a wide range of causes. However, it also is more common in persons with autoimmune inflammatory diseases, such as lupus, Sjögren syndrome, or rheumatoid arthritis. Concern for one of these diseases is the main reason to consider referring a patient for evaluation by a rheumatologist. Often rheumatologists participate in the management of fibromyalgia. A patient should be given appropriate expectations by the referring physician.
Effectiveness of treatment varies widely among patients. Nonpharmacologic approaches such as aerobic exercise, cognitive behavioral therapy, and tai chi have support from clinical trials, and yoga and aquatherapy also are widely used.1,2 The classes of drugs used are the same as for neuropathic pain: tricyclics, including cyclobenzaprine; serotonin and norepinephrine reuptake inhibitors (SNRIs); and gabapentinoids. In contrast, nonsteroidal anti-inflammatory drugs and opioids are ineffective unless there is a superimposed mechanical or inflammatory cause in the periphery. The key point is that continuation of any treatment should be based entirely on the patient’s own assessment of benefit.
►Dr. Swamy. Seven years later, the patient returned to her primary care provider, reporting increased dyspnea on exertion as well as significant fatigue. She was referred to the pulmonary department and had repeat computed tomography (CT) scans of the chest, which indicated persistent right middle lobe (RML) bronchiectasis. She then underwent bronchoscopy with a subsequent bronchoalveolar lavage (BAL) culture growing MAC. Dr. Fine, please interpret the baseline and follow-up CT scans and help us understand the significance of the MAC on sputum and BAL cultures.
►Alan Fine, MD, Section of Pulmonary and Critical Care, VABHS and Professor of Medicine, Boston University School of Medicine. Prior to this presentation, the patient had a pleural-based area of fibrosis with possible associated RML bronchiectasis. This appears to be a postinflammatory process without classic features of malignant or metastatic disease. She then had a sputum, which grew MAC in only 1 of 3 samples and in liquid media only. Importantly, the sputum was not smear positive. All of this suggests a low organism burden. One possibility is that this could reflect colonization with MAC; it is not uncommon for patients with underlying chronic changes in their lung to grow MAC, and it is often difficult to tell whether it is indicative of active disease. Structural lung disease, such as bronchiectasis, predisposes a patient to MAC, but chronic MAC also may cause bronchiectasis. This chicken-and-egg scenario comes up frequently. She may have a MAC infection, but as she is HIV negative and asymptomatic, there is no urgent indication to treat, especially as the burden of therapy is not insignificant.
►Dr. Swamy. Do we need to worry about Mycobacterium tuberculosis (MTB)?
►Dr. Fine. Although she was previously PPD positive, she had already completed 1 year of isoniazid (INH) therapy, making active MTB less likely. From an infection control standpoint, it is important to distinguish MAC from MTB. The former is not contagious, and there is no need for airborne isolation.
►Dr. Swamy. Dr. Fine, where does MAC come from? Does it commonly cause disease?
►Dr. Fine. In the environment, MAC is nearly ubiquitous , especially in water and soil. In one study, 20% of showerheads were positive for MAC; when patients are infected, we may suggest changing/bleaching the showerhead, but there are no definitive recommendations.3 Because MAC is so common in the environment, it is unlikely that measures to target MAC colonization will be clinically meaningful. On the other hand, the incidence of nontuberculous mycobacterial infections is increasing across the US, and it may be a common and frequently underdiagnosed cause of chronic cough, especially in postmenopausal women.
►Dr. Swamy. Four years prior to the current presentation, the patient developed a cough after an upper respiratory tract infection that persisted for more than 2 weeks. Given her history, she underwent a repeat chest CT, which noted a slight increase in nodularity and ground-glass opacity restricted to the RML. She also reported dyspnea on exertion and was referred to the pulmonary medicine department. By the time she arrived, her dyspnea had largely resolved, but she reported persistent fatigue without other systemic symptoms, such as fevers or chills. Dr. Fine, does MAC explain this patient’s dyspnea?
►Dr. Fine. As her pulmonary symptoms resolved in a short period of time with only azithromycin, it is very unlikely that her symptoms were related to her prior disease. The MAC infection is not likely to cause dyspnea on exertion and fatigue and should be worked up more broadly before attributing it to MAC. In view of this, it would not be unreasonable to follow her clinically and see her again in 6 to 8 weeks. In this context, we also should consider the untoward impact of repeated radiation exposure derived from multiple CT scans. When a patient has an abnormality on CT scan, it often leads to further scans even if the symptoms do not match the previous findings, as in this case.
►Dr. Swamy. Given her ongoing fatigue and systemic symptoms (morning stiffness of the shoulders, legs, and thighs, and leg cramps), she was referred to the rheumatology department where the physical examination revealed muscle tenderness in her proximal arms and legs with normal strength, tender points at the elbows and medial side of the bilateral knees, significant tenderness of lower legs, and no synovitis.
Dr. Monach, can you walk us through your approach to this patient? Are we seeing manifestations of fibromyalgia? What diagnoses concerns you and how would you proceed?
►Dr. Monach. The history and exam are most helpful in raising or reducing suspicion for an underlying inflammatory disease. Areas of tenderness described in her case are typical of fibromyalgia, although it can be difficult to interpret symptoms in the hip girdle and shoulder girdle because objective findings are often absent on exam in patients with inflammatory arthritis or bursitis. Similarly, tenderness at sites of tendon insertion (enthuses) without objective abnormalities is common in different forms of spondyloarthritis, so tenderness at the elbow, knee, lateral hip, and low back can be difficult to interpret. What this patient is lacking is prominent subjective or objective findings in the joints most commonly affected in rheumatoid arthritis and lupus: wrists, hands, ankles, and feet.
►Dr. Swamy. Initial laboratory data include an erythrocyte sedimentation rate of 79 with a normal C-reactive protein. A tentative diagnosis of polymyalgia rheumatic is made with consideration of a trial treatment of prednisone.
Dr. Monach, this patient has an indolent infection and is about to be given glucocorticoids. Could you describe the situations in which you feel that glucocorticoids cause a relative immunosuppression?
►Dr. Monach. Glucocorticoids are considered safe in a patient whose infection is not intrinsically dangerous or who has started appropriate antibiotics for that infection. Although all toxicities of glucocorticoids are dose dependent, the long-standing assertion that doses below 10 mg to 15 mg do not increase risk of infection is contradicted by data published in the past 10 to 15 years, with the caveat that these patients were on long-term treatment.
►Dr. Swamy. The patient was started on prednisone 15 mg per day for 15 days. She returned to the clinic after 1 week of prednisone troutment and noted “significant improvement in fatigue, morning stiffness of shoulders, thighs, leg, back is better, leg cramps resolved, shooting pain in many joints resolved.” Further laboratory results were notable for a negative rheumatoid factor, negative antinuclear antibody, and a cyclic citrullinated peptide of 60. A presumptive diagnosis of rheumatoid arthritis (RA) was made and plaquenil 200 mg twice daily was started.
Dr. Monach, can you explain why RA comes up now on serology but was not considered initially? Why does this presentation fit RA, and was her response to treatment typical? How does this fit in with her previous diagnosis of fibromyalgia? Was that just an atypical, indolent presentation of RA?
►Dr. Monach. Though her presentation is atypical for RA, in elderly patients, RA can present with symptoms resembling polymyalgia rheumatica. The question is whether she had RA all along (in which case “elderly onset” would not apply) or had fibromyalgia and developed RA more recently. The response to empiric glucocorticoid therapy is helpful, since fibromyalgia should not improve with prednisone even in a patient with RA unless treatment of RA would allow better sleep and ability to exercise. Rheumatoid arthritis typically responds very well to prednisone in the 5-mg to 15-mg range.
►Dr. Swamy. Given the new diagnosis of an inflammatory arthritis requiring immunosuppression, bronchoscopy with BAL is performed to evaluate for the presence of MAC. These cultures were positive for MAC.
Dr. Fine, does the positive BAL culture indicate an active MAC infection?
►Dr. Fine. Yes, based on these updated data, the patient has an active MAC infection. Active infection is defined as symptoms or imaging consistent with the diagnosis, supporting microbiology data (either 2 sputum or 1 BAL sample growing MAC) and the exclusion of other causes. Previously, this patient grew MAC in just one expectorated sputum; this did not meet the microbiologic criteria. Now sputum has grown in the BAL sample; along with the CT imaging, this is enough to diagnosis active MAC infection.
Treatment for MAC must consider the details of each case. First, this is not an emergency; treatment decisions should be made with the rheumatologist to consider the planned immunosuppression. For example, we must consider potential drug interactions. A specific point should be made of the use of tumor necrosis factor (TNF)-α inhibition, which data indicate can reactivate TB and may inhibit mechanisms that restrain mycobacterial disease. Serious cases of MAC infection have been reported in the literature in the setting of TNF-α inhibition.5,6 Despite these concerns, there is not a contraindication to using these therapies from the perspective of the active MAC disease. All of these decisions will impact the need to commit the patient to MAC therapy.
►Dr. Swamy. Dr. Fine, what do you consider prior to initiating MAC therapy?
► Dr. Fine. The decision to pursue MAC therapy should not be taken lightly. Therapy often entails prolonged multidrug regimens, usually spanning more than a year, with frequent adverse effects. Outside of very specific cases, such as TNF-β inhibition, MAC is rarely a life-threatening disease, so the benefit may be limited. Treatment for MAC is certainly unlikely to be fruitful without a diligent and motivated patient able to handle the high and prolonged pill burden. Of note, it is also important to keep this patient up-to-date with influenza and pneumonia vaccination given her structural lung disease.
►Dr. Swamy. The decision is made to treat MAC with azithromycin, rifampin, and ethambutol. The disease is noted to be nonfibrocavitary. The patient underwent monthly liver function test monitoring and visual acuity testing, which were unremarkable. Dr. Fine, can you describe the phenotypes of nontuberculous mycobacterial (NTM) disease?
►Dr. Fine. There are 3 main phenotypes of NTM.3 First, we see the elderly man with preexisting lung disease—usually chronic obstructive pulmonary disease—with fibrocavitary and/or reticulonodular appearance. Second, we see the slim, elderly woman often without any preexisting lung disease presenting with focal bronchiectasis and nodular lesions in right middle lobe and lingula—the Lady Windermere syndrome. This eponym is derived from Oscar Wilde’s play “Lady Windermere’s Fan, a Play About a Good Woman,” and was first associated with this disease in 1992.7 At the time, it was thought that the voluntary suppression of cough led to poorly draining lung regions, vulnerable to engraftment by atypical mycobacteria. Infection with atypical mycobacteria are associated with this population; however, it is no longer thought to be due to the voluntary suppression of cough.7,8 Third, we do occasionally see atypical presentations, such as focal masses and solitary nodules.
►Dr. Swamy. At 1-year follow-up she successfully completed MAC therapy and noted ongoing control of rheumatoid symptoms.
Case Presentation. A 64-year-old US Army veteran with a history of colorectal cancer, melanoma, and fibrinolytic presented with dyspnea to VA Boston Healthcare System (VABHS). Seven years prior to the current presentation, at the time of her diagnosis of colorectal cancer, the patient was found to be HIV negative but to have a positive purified protein derivative (PPD) test. She was treated with isoniazid (INH) therapy for 9 months. Sputum cultures collected prior to initiation of therapy grew Mycobacterium avium complex (MAC) in 1 of 3 samples, with these results reported several months after initiation of therapy. She was a never smoker with no known travel or exposure. At the time of the current presentation, her medications included bupropion, levothyroxine, capsaicin, cyclobenzaprine, ibuprofen, and acetaminophen.
►Lakshmana Swamy, MD, Chief Medical Resident, VABHS and Boston Medical Center. Dr. Monach, this patient is on a variety of pain medications and has a diagnosis of fibromyalgia. This diagnosis often frustrates doctors and patients alike. Can you tell us about fibromyalgia from the rheumatologist’s perspective and what you think of her current treatment regimen?
►Paul A. Monach, MD, PhD, Chief, Section of Rheumatology, VABHS and Associate Professor of Medicine, Boston University School of Medicine. Fibromyalgia is a syndrome of chronic widespread pain without known pathology in the musculoskeletal system. It is thought to be caused by chronic dysfunction of pain-processing pathways in the central nervous system (CNS). It is often accompanied by other somatic symptoms such as chronic fatigue, irritable bowel syndrome, and bladder pain. It is a common condition, affecting up to 5% of otherwise healthy women. It is particularly common in persons with chronic nonrestorative sleep or posttraumatic stress disorder from a wide range of causes. However, it also is more common in persons with autoimmune inflammatory diseases, such as lupus, Sjögren syndrome, or rheumatoid arthritis. Concern for one of these diseases is the main reason to consider referring a patient for evaluation by a rheumatologist. Often rheumatologists participate in the management of fibromyalgia. A patient should be given appropriate expectations by the referring physician.
Effectiveness of treatment varies widely among patients. Nonpharmacologic approaches such as aerobic exercise, cognitive behavioral therapy, and tai chi have support from clinical trials, and yoga and aquatherapy also are widely used.1,2 The classes of drugs used are the same as for neuropathic pain: tricyclics, including cyclobenzaprine; serotonin and norepinephrine reuptake inhibitors (SNRIs); and gabapentinoids. In contrast, nonsteroidal anti-inflammatory drugs and opioids are ineffective unless there is a superimposed mechanical or inflammatory cause in the periphery. The key point is that continuation of any treatment should be based entirely on the patient’s own assessment of benefit.
►Dr. Swamy. Seven years later, the patient returned to her primary care provider, reporting increased dyspnea on exertion as well as significant fatigue. She was referred to the pulmonary department and had repeat computed tomography (CT) scans of the chest, which indicated persistent right middle lobe (RML) bronchiectasis. She then underwent bronchoscopy with a subsequent bronchoalveolar lavage (BAL) culture growing MAC. Dr. Fine, please interpret the baseline and follow-up CT scans and help us understand the significance of the MAC on sputum and BAL cultures.
►Alan Fine, MD, Section of Pulmonary and Critical Care, VABHS and Professor of Medicine, Boston University School of Medicine. Prior to this presentation, the patient had a pleural-based area of fibrosis with possible associated RML bronchiectasis. This appears to be a postinflammatory process without classic features of malignant or metastatic disease. She then had a sputum, which grew MAC in only 1 of 3 samples and in liquid media only. Importantly, the sputum was not smear positive. All of this suggests a low organism burden. One possibility is that this could reflect colonization with MAC; it is not uncommon for patients with underlying chronic changes in their lung to grow MAC, and it is often difficult to tell whether it is indicative of active disease. Structural lung disease, such as bronchiectasis, predisposes a patient to MAC, but chronic MAC also may cause bronchiectasis. This chicken-and-egg scenario comes up frequently. She may have a MAC infection, but as she is HIV negative and asymptomatic, there is no urgent indication to treat, especially as the burden of therapy is not insignificant.
►Dr. Swamy. Do we need to worry about Mycobacterium tuberculosis (MTB)?
►Dr. Fine. Although she was previously PPD positive, she had already completed 1 year of isoniazid (INH) therapy, making active MTB less likely. From an infection control standpoint, it is important to distinguish MAC from MTB. The former is not contagious, and there is no need for airborne isolation.
►Dr. Swamy. Dr. Fine, where does MAC come from? Does it commonly cause disease?
►Dr. Fine. In the environment, MAC is nearly ubiquitous , especially in water and soil. In one study, 20% of showerheads were positive for MAC; when patients are infected, we may suggest changing/bleaching the showerhead, but there are no definitive recommendations.3 Because MAC is so common in the environment, it is unlikely that measures to target MAC colonization will be clinically meaningful. On the other hand, the incidence of nontuberculous mycobacterial infections is increasing across the US, and it may be a common and frequently underdiagnosed cause of chronic cough, especially in postmenopausal women.
►Dr. Swamy. Four years prior to the current presentation, the patient developed a cough after an upper respiratory tract infection that persisted for more than 2 weeks. Given her history, she underwent a repeat chest CT, which noted a slight increase in nodularity and ground-glass opacity restricted to the RML. She also reported dyspnea on exertion and was referred to the pulmonary medicine department. By the time she arrived, her dyspnea had largely resolved, but she reported persistent fatigue without other systemic symptoms, such as fevers or chills. Dr. Fine, does MAC explain this patient’s dyspnea?
►Dr. Fine. As her pulmonary symptoms resolved in a short period of time with only azithromycin, it is very unlikely that her symptoms were related to her prior disease. The MAC infection is not likely to cause dyspnea on exertion and fatigue and should be worked up more broadly before attributing it to MAC. In view of this, it would not be unreasonable to follow her clinically and see her again in 6 to 8 weeks. In this context, we also should consider the untoward impact of repeated radiation exposure derived from multiple CT scans. When a patient has an abnormality on CT scan, it often leads to further scans even if the symptoms do not match the previous findings, as in this case.
►Dr. Swamy. Given her ongoing fatigue and systemic symptoms (morning stiffness of the shoulders, legs, and thighs, and leg cramps), she was referred to the rheumatology department where the physical examination revealed muscle tenderness in her proximal arms and legs with normal strength, tender points at the elbows and medial side of the bilateral knees, significant tenderness of lower legs, and no synovitis.
Dr. Monach, can you walk us through your approach to this patient? Are we seeing manifestations of fibromyalgia? What diagnoses concerns you and how would you proceed?
►Dr. Monach. The history and exam are most helpful in raising or reducing suspicion for an underlying inflammatory disease. Areas of tenderness described in her case are typical of fibromyalgia, although it can be difficult to interpret symptoms in the hip girdle and shoulder girdle because objective findings are often absent on exam in patients with inflammatory arthritis or bursitis. Similarly, tenderness at sites of tendon insertion (enthuses) without objective abnormalities is common in different forms of spondyloarthritis, so tenderness at the elbow, knee, lateral hip, and low back can be difficult to interpret. What this patient is lacking is prominent subjective or objective findings in the joints most commonly affected in rheumatoid arthritis and lupus: wrists, hands, ankles, and feet.
►Dr. Swamy. Initial laboratory data include an erythrocyte sedimentation rate of 79 with a normal C-reactive protein. A tentative diagnosis of polymyalgia rheumatic is made with consideration of a trial treatment of prednisone.
Dr. Monach, this patient has an indolent infection and is about to be given glucocorticoids. Could you describe the situations in which you feel that glucocorticoids cause a relative immunosuppression?
►Dr. Monach. Glucocorticoids are considered safe in a patient whose infection is not intrinsically dangerous or who has started appropriate antibiotics for that infection. Although all toxicities of glucocorticoids are dose dependent, the long-standing assertion that doses below 10 mg to 15 mg do not increase risk of infection is contradicted by data published in the past 10 to 15 years, with the caveat that these patients were on long-term treatment.
►Dr. Swamy. The patient was started on prednisone 15 mg per day for 15 days. She returned to the clinic after 1 week of prednisone troutment and noted “significant improvement in fatigue, morning stiffness of shoulders, thighs, leg, back is better, leg cramps resolved, shooting pain in many joints resolved.” Further laboratory results were notable for a negative rheumatoid factor, negative antinuclear antibody, and a cyclic citrullinated peptide of 60. A presumptive diagnosis of rheumatoid arthritis (RA) was made and plaquenil 200 mg twice daily was started.
Dr. Monach, can you explain why RA comes up now on serology but was not considered initially? Why does this presentation fit RA, and was her response to treatment typical? How does this fit in with her previous diagnosis of fibromyalgia? Was that just an atypical, indolent presentation of RA?
►Dr. Monach. Though her presentation is atypical for RA, in elderly patients, RA can present with symptoms resembling polymyalgia rheumatica. The question is whether she had RA all along (in which case “elderly onset” would not apply) or had fibromyalgia and developed RA more recently. The response to empiric glucocorticoid therapy is helpful, since fibromyalgia should not improve with prednisone even in a patient with RA unless treatment of RA would allow better sleep and ability to exercise. Rheumatoid arthritis typically responds very well to prednisone in the 5-mg to 15-mg range.
►Dr. Swamy. Given the new diagnosis of an inflammatory arthritis requiring immunosuppression, bronchoscopy with BAL is performed to evaluate for the presence of MAC. These cultures were positive for MAC.
Dr. Fine, does the positive BAL culture indicate an active MAC infection?
►Dr. Fine. Yes, based on these updated data, the patient has an active MAC infection. Active infection is defined as symptoms or imaging consistent with the diagnosis, supporting microbiology data (either 2 sputum or 1 BAL sample growing MAC) and the exclusion of other causes. Previously, this patient grew MAC in just one expectorated sputum; this did not meet the microbiologic criteria. Now sputum has grown in the BAL sample; along with the CT imaging, this is enough to diagnosis active MAC infection.
Treatment for MAC must consider the details of each case. First, this is not an emergency; treatment decisions should be made with the rheumatologist to consider the planned immunosuppression. For example, we must consider potential drug interactions. A specific point should be made of the use of tumor necrosis factor (TNF)-α inhibition, which data indicate can reactivate TB and may inhibit mechanisms that restrain mycobacterial disease. Serious cases of MAC infection have been reported in the literature in the setting of TNF-α inhibition.5,6 Despite these concerns, there is not a contraindication to using these therapies from the perspective of the active MAC disease. All of these decisions will impact the need to commit the patient to MAC therapy.
►Dr. Swamy. Dr. Fine, what do you consider prior to initiating MAC therapy?
► Dr. Fine. The decision to pursue MAC therapy should not be taken lightly. Therapy often entails prolonged multidrug regimens, usually spanning more than a year, with frequent adverse effects. Outside of very specific cases, such as TNF-β inhibition, MAC is rarely a life-threatening disease, so the benefit may be limited. Treatment for MAC is certainly unlikely to be fruitful without a diligent and motivated patient able to handle the high and prolonged pill burden. Of note, it is also important to keep this patient up-to-date with influenza and pneumonia vaccination given her structural lung disease.
►Dr. Swamy. The decision is made to treat MAC with azithromycin, rifampin, and ethambutol. The disease is noted to be nonfibrocavitary. The patient underwent monthly liver function test monitoring and visual acuity testing, which were unremarkable. Dr. Fine, can you describe the phenotypes of nontuberculous mycobacterial (NTM) disease?
►Dr. Fine. There are 3 main phenotypes of NTM.3 First, we see the elderly man with preexisting lung disease—usually chronic obstructive pulmonary disease—with fibrocavitary and/or reticulonodular appearance. Second, we see the slim, elderly woman often without any preexisting lung disease presenting with focal bronchiectasis and nodular lesions in right middle lobe and lingula—the Lady Windermere syndrome. This eponym is derived from Oscar Wilde’s play “Lady Windermere’s Fan, a Play About a Good Woman,” and was first associated with this disease in 1992.7 At the time, it was thought that the voluntary suppression of cough led to poorly draining lung regions, vulnerable to engraftment by atypical mycobacteria. Infection with atypical mycobacteria are associated with this population; however, it is no longer thought to be due to the voluntary suppression of cough.7,8 Third, we do occasionally see atypical presentations, such as focal masses and solitary nodules.
►Dr. Swamy. At 1-year follow-up she successfully completed MAC therapy and noted ongoing control of rheumatoid symptoms.
1. Bernardy K, Klose P, Welsch P, Häuser W. Efficacy, acceptability and safety of cognitive behavioural therapies in fibromyalgia syndrome—a systematic review and meta-analysis of randomized controlled trials. Eur J Pain. 2018;22(2):242-260.
2. Geneen LJ, Moore RA, Clarke C, Martin D, Colvin LA, Smith BH. Physical activity and exercise for chronic pain in adults: an overview of Cochrane Reviews. Cochrane Database Syst Rev. 2017;4:CD011279.
3. Aksamit TR, Philley JV, Griffith DE. Nontuberculous mycobacterial (NTM) lung disease: the top ten essentials. Respir Med. 2014;108(3):417-425.
4. Aucott JN. Glucocorticoids and infection. Endocrinol Metab Clin North Am. 1994;23(3):655-670.
5. Curtis JR, Yang S, Patkar NM, et al. Risk of hospitalized bacterial infections associated with biologic treatment among US veterans with rheumatoid arthritis. Arthritis Care Res (Hoboken). 2014;66(7):990-997.
6. Lane MA, McDonald JR, Zeringue AL, et al. TNF-α antagonist use and risk of hospitalization for infection in a national cohort of veterans with rheumatoid arthritis. Medicine (Baltimore). 2011;90(2):139-145.
7. Reich JM, Johnson RE. Mycobacterium avium complex pulmonary disease presenting as an isolated lingular or middle lobe pattern. The Lady Windermere syndrome. Chest. 1992;101(6):1605-1609.
8. Kasthoori JJ, Liam CK, Wastie ML. Lady Windermere syndrome: an inappropriate eponym for an increasingly important condition. Singapore Med J. 2008;49(2):e47-e49.
1. Bernardy K, Klose P, Welsch P, Häuser W. Efficacy, acceptability and safety of cognitive behavioural therapies in fibromyalgia syndrome—a systematic review and meta-analysis of randomized controlled trials. Eur J Pain. 2018;22(2):242-260.
2. Geneen LJ, Moore RA, Clarke C, Martin D, Colvin LA, Smith BH. Physical activity and exercise for chronic pain in adults: an overview of Cochrane Reviews. Cochrane Database Syst Rev. 2017;4:CD011279.
3. Aksamit TR, Philley JV, Griffith DE. Nontuberculous mycobacterial (NTM) lung disease: the top ten essentials. Respir Med. 2014;108(3):417-425.
4. Aucott JN. Glucocorticoids and infection. Endocrinol Metab Clin North Am. 1994;23(3):655-670.
5. Curtis JR, Yang S, Patkar NM, et al. Risk of hospitalized bacterial infections associated with biologic treatment among US veterans with rheumatoid arthritis. Arthritis Care Res (Hoboken). 2014;66(7):990-997.
6. Lane MA, McDonald JR, Zeringue AL, et al. TNF-α antagonist use and risk of hospitalization for infection in a national cohort of veterans with rheumatoid arthritis. Medicine (Baltimore). 2011;90(2):139-145.
7. Reich JM, Johnson RE. Mycobacterium avium complex pulmonary disease presenting as an isolated lingular or middle lobe pattern. The Lady Windermere syndrome. Chest. 1992;101(6):1605-1609.
8. Kasthoori JJ, Liam CK, Wastie ML. Lady Windermere syndrome: an inappropriate eponym for an increasingly important condition. Singapore Med J. 2008;49(2):e47-e49.
Boston VA Medical Forum: HIV-Positive Veteran With Progressive Visual Changes
►Lakshmana Swamy, MD, chief medical resident, VA Boston Healthcare System (VABHS) and Boston Medical Center. Dr. Serrao, when you hear about vision changes in a patient with HIV, what differential diagnosis is generated? What epidemiologic or historical factors can help distinguish these entities?
►Richard Serrao, MD, Infectious Disease Service, VABHS and assistant professor of medicine, Boston University School of Medicine. The differential diagnoses for vision changes in a patient with HIV is based on the overall immunosuppression of the patient: the lower the patient’s CD4 count, the higher the number of etiologies.1 The portions of the visual pathway as well as the pattern of vision loss are useful in narrowing the differential. For example, monocular visual disturbances with dermatomal vesicles within the ophthalmic division of the trigeminal nerve strongly implicates varicella zoster retinitis or keratitis; abducens nerve palsy could suggest granulomatous basilar meningitis from cryptococcosis. Likewise, ongoing fevers in an advanced AIDS patient with concomitant colitis, hepatitis, and pneumonitis is strongly suspicious for cytomegalovirus (CMV) retinitis with wide dissemination.
Geographic epidemiologic factors can suggest pathogens more prevalent to certain regions of the world, such as histoplasma chorioretinitis in a resident of the central and eastern U.S. or tuberculosis in a returning traveler. Likewise, a cat owner or one who consumes steak tartare increases the likelihood for toxoplasma retinochoroiditis, or syphilis in men who have sex with men (MSM) in the U.S. given that the majority of new cases occur in this patient population. Other clues one should consider include the presence of splinter hemorrhages in the extremities in an intravenous drug user, raising the possibility of embolic endophthalmitis from bacterial or fungal endocarditis. A variety of other diagnoses can certainly occur as a result of drug treatment (uveitis from rifampin, for example), immune reconstitution from HAART, infections with other HIV-associated pathogens, such as Pneumocystis jiroveci, and many non-HIV-related ocular diseases.
►Dr. Swamy. Dr. Butler, what concerns do you have when you hear about an HIV-infected patient with vision loss from the ophthalmology perspective?
►Nicholas Butler, MD, Ophthalmology Service, Uveitis and Ocular Immunology, VABHS and assistant professor of ophthalmology, Harvard Medical School. Of course, patients with HIV suffer from common causes of vision loss—cataract, glaucoma, diabetes, macular degeneration, for instance—just like those without HIV infection. If there is no significant immunodeficiency, then the patient’s HIV status would be less relevant, and these more common causes of vision loss should be pursued. My first task would be to determine the patient’s most recent CD4 T-cell count.
Assuming an HIV-positive individual is experiencing visual symptoms related to his/her underlying HIV infection (especially in the setting of CD4 counts < 200 cells/mm3), ocular opportunistic infections (OOI) come to mind first. Despite a reduction in incidence of 75% to 80% in the HAART-era, CMV retinitis remains the most common OOI in patients with AIDS and carries the greatest risk of ocular morbidity.2 In fact, based on enrollment data for the Longitudinal Study of the Ocular Complications of AIDS (LSOCA), the prevalence of CMV retinitis among patients with AIDS is more than 20-fold higher than all other ocular complications of AIDS (OOIs and ocular neoplastic disease), including Kaposi sarcoma, lymphoma, herpes zoster ophthalmicus, ocular syphilis, ocular toxoplasma, necrotizing herpetic retinitis, cryptococcal choroiditis, and pneumocystis choroiditis.3 Beyond ocular opportunistic infections, the most common retinal finding in HIV-positive people is HIV retinopathy, nonspecific microvascular findings in the retina affecting nearly 70% of those with advanced HIV disease. Fortunately, HIV retinopathy is generally asymptomatic.4
►Dr. Swamy. Thank you for those explanations. Based on Dr. Serrao’s differential, it is worth noting that this patient is MSM. He was evaluated in urgent care with the initial examination showing a temperature of 98.0° F, pulse 83 beats per minute, and blood pressure 110/70 mm Hg. The eye exam showed no injection with normal extraocular movements. Initial laboratory data were notable for a CD4 count of 730 cells/mm3 with fewer than 20 HIV viral copies/mL. Cytomegalovirus immunoglobulin G (IgG) was positive, and immunoglobulin M (IgM) was negative. A Lyme antibody was positive with negative IgM and IgG by Western blot. Additional tests can be seen in Tables 1 and 2. The patient has good immunologic and virologic control. How does this change your thinking about the case?
►Dr. Serrao. His CD4 count is well above 350, increasing the likelihood of a relatively uncomplicated course and treatment. Cytomegalovirus antibodies reflect prior infection. As CMV generally does not manifest with disease of any variety (including CMV retinitis) at this high CD4 count, one can presume he does not have CMV retinitis as a cause for his visual changes. CMV retinitis occurs mainly when substantial CD4 depletion has occurred (typically less than 50 cells/mm3). A positive Lyme antibody screen, not specific to Lyme, can be falsely positive in other treponema diseases (eg, Treponema pallidum, the etiologic organism of syphilis) as evidenced by negative confirmatory Western blot IgG and IgM. Antineutrophil cystoplasmic antibodies, lysozyme, angiotensin-converting enzyme, rapid plasma reagin (RPR), herpes simplex virus, toxoplasma are generally included in the workup for the differential of uveitis, retinitis, choroiditis, etc.
►Dr. Swamy. Based on the visual changes, the patient was referred for urgent ophthalmologic evaluation. Dr. Butler, when should a generalist consider urgent ophthalmology referral?
►Dr. Butler. In general, all patients with acute (and significant) vision loss should be referred immediately to an ophthalmologist. The challenge for the general practitioner is determining the true extent of the reported vision loss. If possible, some assessment of visual acuity should be obtained, testing each eye independently and with the correct glasses correction (ie, the patient’s distance glasses if the test object is 12 feet or more from the patient or their reading glasses if the test object is held inside arm’s length). If the general practitioner does not have access to an eye chart or near card, any assessment of vision with an appropriate description will be useful (eg, the patient can quickly count fingers at 15 feet in the unaffected eye, but the eye with reported vision loss cannot reliably count fingers outside of 2 feet). Additional ocular symptoms associated with the vision loss, such as pain, redness, photophobia, new flashes or floaters, increase the urgency of the referral. The threshold for referral for any ocular complaint is lower compared with that of the general population for those with evidence of immunodeficiency, such as for this patient with HIV. Any CD4 count < 200 cells/mm3 should raise the practitioner’s concern for an ocular opportunistic infection, with the greatest concern with CD4 counts < 50 cells/mm3.
►Dr. Swamy. The patient underwent further testing in the ophthalmology clinic. Dr. Butler, can you please interpret the funduscopic exam?
►Dr. Butler. Both eyes demonstrate findings (microaneurysms and small dot-blot hemorrhages) consistent with moderate nonproliferative diabetic retinopathy (Figure 1A, white arrows). HIV-associated retinopathy could produce similar findings, but it is not generally seen with CD4 counts > 200 cells/mm3. Additionally, in the left eye, there is a diffuse patch of retinal whitening (retinitis) associated with the inferotemporal vascular arcades (Figure 1B, white arrows). The entire area involved is poorly circumscribed and the whitening is subtle in areas. Overlying some areas of deeper, ground-glass whitening there are scattered, punctate white spots (Figure 1B, green arrows). Wickremasinghe and colleagues described this pattern of retinitis and suggested that it had a high positive-predictive value in the diagnosis of ocular syphilis.5
►Dr. Swamy. The patient then underwent fluorescein angiography and optical coherence tomography (OCT). Dr. Butler, what did the fluorescein angiography show?
►Dr. Butler. The fluorescein angiogram in both eyes revealed leakage of dye consistent with diabetic retinopathy, with the right eye (OD) worse than the left (OS). Additionally, the areas of active retinitis in the left eye displayed gradual staining with leopard-spot changes, along with late leakage of fluorescein dye, indicating vasculopathy in the infected area (Figure 2, arrows). The patient also underwent OCT in the left eye (images not displayed) demonstrating vitreous cells (vitritis), patches of inner retinal thickening with hyperreflectivity, and hyperreflective nodules at the level of the retinal pigment epithelium with overlying photoreceptor disruption. These OCT findings are fairly stereotypic for syphilitic chorioretinitis.6
►Dr. Swamy. Based on the ophthalmic findings, a diagnosis of ocular syphilis was made. Dr. Serrao, what should internists consider as they evaluate and manage a patient with ocular syphilis?
►Dr. Serrao. Although isolated ocular involvement from syphilis is possible, the majority of patients (up to 85%) with HIV can present with concomitant central nervous system infection and about 30% present with symptomatic neurosyphilis (a typical late manifestation of this disease) that reflects the aggressiveness, accelerated course and propensity for wide dissemination of syphilis in this patient population.7
The presence of concomitant cutaneous rashes should prompt universal precautions, because transmission can occur via skin to skin contact. Clinicians should watch for the Jarisch-Herxheimer reaction during treatment, a syndrome of fever, myalgias, and headache, which results from circulating cytokines produced because of rapidly dying spirochetes that could mimic a penicillin drug reaction, yet is treated supportively.
As syphilis is sexually acquired, clinicians should test for coexistent sexually transmitted infections, vaccinate for those that are preventable (eg, hepatitis B), notify sexual partners via assistance from local departments of public health, and assess for coexistent drug use and offer counseling in order to optimize risk reduction. Special attention should be paid to virologic control of HIV since some studies have shown an increase in the propensity for breakthrough HIV viremia while on effective ART.9 This should warrant counseling for ongoing optimal ART adherence and close monitoring in the follow-up visits with a provider specialized in the treatment of syphilis and HIV.
►Dr. Swamy. A lumbar puncture is performed with the results listed in Table 2. Dr. Serrao, is the CSF consistent with neurosyphilis? What would you do next?
►Dr. Serrao. The lumbar puncture is inflammatory with a lymphocytic predominance, consistent with active ocular/neurosyphilis. The CSF Venereal Disease Research Laboratory test is specific but not sensitive so a negative value does not rule out the presence of central nervous system infection.10 The CSF fluorescent treponemal antibody (CSF FTA-ABS) is sensitive but not specific. In this case, the ocular findings, positive serum RPR, CSF lymphocytic predominance, and CSF FTA ABS strongly supports the diagnosis of ocular/early neurosyphilis in a patient with HIV infection in whom early aggressive treatment is warranted to prevent rapid progression/potential loss of vision.11
►Dr. Swamy. Dr. Butler, how does syphilis behave in the eye as compared to other infectious or inflammatory diseases? Do visual symptoms respond well to treatment?
►Dr. Butler. As opposed to the dramatic reduction in rates and severity of CMV retinitis, HAART has had a negligible effect on ocular syphilis in the setting of HIV coinfection; in fact, rates of syphilis, including ocular syphilis, are currently surging world-wide, and HIV coinfection portends a worse prognosis.12 This is especially true among gay men. More so, there appears to be no correlation between CD4 count and incidence of developing ocular syphilis, as opposed to CMV retinitis, which occurs far more frequently in those with CD4 counts < 50 cells/mm3. In keeping with its epithet as one of the “Great Imitators,” syphilis can affect virtually every tissue of the eye—conjunctiva, sclera, cornea, iris, lens, vitreous, retina, choroid, optic nerve—unlike other OOI, such as CMV or toxoplasma, which generally hone to the retina. Nonetheless, various findings and patterns on clinical exam and ancillary testing, such as the more recently described punctate inner retinitis (as seen in our patient) and the more classic acute syphilitic posterior placoid chorioretinitis, carry high specificity for ocular syphilis.13
Patients with ocular syphilis should be treated according to neurosyphilis treatment protocols. In general, these patients respond very well to treatment with resolution of the ocular findings and recovery of complete, or nearly so, visual function, as long as an excessive delay between diagnosis and proper treatment does not occur.14
►Dr. Swamy. Following this testing, the patient completed 14 days of IV penicillin with resolution of symptoms. He had no further vision complaints. He was started on Triumeq (abacavir, dolutegravir, and lamivudine) with good adherence to therapy. Dr. Serrao, in 2016 the CDC released a clinical advisory about ocular syphilis. Can you tell us about why this is an important diagnosis to be aware of today?
►Dr. Serrao. As with any disease, the epidemiologic characteristics of an infection like syphilis allow the clinician to more carefully entertain such a diagnosis in any one individual by improving the index of suspicion for a particular disease. Awareness of an increase in ocular syphilis in HIV positive MSM allows for a more timely assessment and subsequent treatment with the goal of preventing loss of vision.15
1. Cunningham ET Jr, Margolis TP. Ocular manifestations of HIV infection. N Engl J Med. 1998;339(4):236-244.
2. Holtzer CD, Jacobson MA, Hadley WK, et al. Decline in the rate of specific opportunistic infections at San Francisco General Hospital, 1994-1997. AIDS. 1998;12(14):1931-1933.
3. Gangaputra S, Drye L, Vaidya V, Thorne JE, Jabs DA, Lyon AT. Non-cytomegalovirus ocular opportunistic infections in patients with acquired immunodeficiency syndrome. Am J Ophthalmol. 2013;155(2):206-212.e205.
4. Jabs DA, Van Natta ML, Holbrook JT, et al. Longitudinal study of the ocular complications of AIDS: 1. Ocular diagnoses at enrollment. Ophthalmology. 2007;114(4):780-786.
5. Wickremasinghe S, Ling C, Stawell R, Yeoh J, Hall A, Zamir E. Syphilitic punctate inner retinitis in immunocompetent gay men. Ophthalmology. 2009;116(6):1195-1200.
6. Burkholder BM, Leung TG, Ostheimer TA, Butler NJ, Thorne JE, Dunn JP. Spectral domain optical coherence tomography findings in acute syphilitic posterior placoid chorioretinitis. J Ophthalmic Inflamm Infect. 2014;4(1):2.
7. Musher DM, Hamill RJ, Baughn RE. Effect of human immunodeficiency virus (HIV) infection on the course of syphilis and on the response to treatment. Ann Intern Med. 1990;113(11):872-881.
8. Lukehart SA, Hook EW 3rd, Baker-Zander SA, Collier AC, Critchlow CW, Handsfield HH. Invasion of the central nervous system by Treponema pallidum: implications for diagnosis and treatment. Ann Intern Med. 1988;109(11):855-862.
9. Golden MR, Marra CM, Holmes KK. Update on syphilis: resurgence of an old problem. JAMA. 2003;290(11):1510-1514.
10. Marra CM, Tantalo LC, Maxwell CL, Ho EL, Sahi SK, Jones T. The rapid plasma reagin test cannot replace the venereal disease research laboratory test for neurosyphilis diagnosis. Sex Transm Dis. 2012;39(6):453-457.
11. Harding AS, Ghanem KG. The performance of cerebrospinal fluid treponemal-specific antibody tests in neurosyphilis: a systematic review. Sex Transm Dis. 2012;39(4):291-297.
12. Butler NJ, Thorne JE. Current status of HIV infection and ocular disease. Curr Opin Ophthalmol. 2012;23(6):517-522.
13. Gass JD, Braunstein RA, Chenoweth RG. Acute syphilitic posterior placoid chorioretinitis. Ophthalmology. 1990;97(10):1288-1297.
14. Davis JL. Ocular syphilis. Curr Opin Ophthalmol. 2014;25(6):513-518.
15. Clinical Advisory: Ocular Syphilis in the United States. https://www.cdc.gov/std/syphilis/clinicaladvisoryos2015.htm. Accessed September 11, 2017.
►Lakshmana Swamy, MD, chief medical resident, VA Boston Healthcare System (VABHS) and Boston Medical Center. Dr. Serrao, when you hear about vision changes in a patient with HIV, what differential diagnosis is generated? What epidemiologic or historical factors can help distinguish these entities?
►Richard Serrao, MD, Infectious Disease Service, VABHS and assistant professor of medicine, Boston University School of Medicine. The differential diagnoses for vision changes in a patient with HIV is based on the overall immunosuppression of the patient: the lower the patient’s CD4 count, the higher the number of etiologies.1 The portions of the visual pathway as well as the pattern of vision loss are useful in narrowing the differential. For example, monocular visual disturbances with dermatomal vesicles within the ophthalmic division of the trigeminal nerve strongly implicates varicella zoster retinitis or keratitis; abducens nerve palsy could suggest granulomatous basilar meningitis from cryptococcosis. Likewise, ongoing fevers in an advanced AIDS patient with concomitant colitis, hepatitis, and pneumonitis is strongly suspicious for cytomegalovirus (CMV) retinitis with wide dissemination.
Geographic epidemiologic factors can suggest pathogens more prevalent to certain regions of the world, such as histoplasma chorioretinitis in a resident of the central and eastern U.S. or tuberculosis in a returning traveler. Likewise, a cat owner or one who consumes steak tartare increases the likelihood for toxoplasma retinochoroiditis, or syphilis in men who have sex with men (MSM) in the U.S. given that the majority of new cases occur in this patient population. Other clues one should consider include the presence of splinter hemorrhages in the extremities in an intravenous drug user, raising the possibility of embolic endophthalmitis from bacterial or fungal endocarditis. A variety of other diagnoses can certainly occur as a result of drug treatment (uveitis from rifampin, for example), immune reconstitution from HAART, infections with other HIV-associated pathogens, such as Pneumocystis jiroveci, and many non-HIV-related ocular diseases.
►Dr. Swamy. Dr. Butler, what concerns do you have when you hear about an HIV-infected patient with vision loss from the ophthalmology perspective?
►Nicholas Butler, MD, Ophthalmology Service, Uveitis and Ocular Immunology, VABHS and assistant professor of ophthalmology, Harvard Medical School. Of course, patients with HIV suffer from common causes of vision loss—cataract, glaucoma, diabetes, macular degeneration, for instance—just like those without HIV infection. If there is no significant immunodeficiency, then the patient’s HIV status would be less relevant, and these more common causes of vision loss should be pursued. My first task would be to determine the patient’s most recent CD4 T-cell count.
Assuming an HIV-positive individual is experiencing visual symptoms related to his/her underlying HIV infection (especially in the setting of CD4 counts < 200 cells/mm3), ocular opportunistic infections (OOI) come to mind first. Despite a reduction in incidence of 75% to 80% in the HAART-era, CMV retinitis remains the most common OOI in patients with AIDS and carries the greatest risk of ocular morbidity.2 In fact, based on enrollment data for the Longitudinal Study of the Ocular Complications of AIDS (LSOCA), the prevalence of CMV retinitis among patients with AIDS is more than 20-fold higher than all other ocular complications of AIDS (OOIs and ocular neoplastic disease), including Kaposi sarcoma, lymphoma, herpes zoster ophthalmicus, ocular syphilis, ocular toxoplasma, necrotizing herpetic retinitis, cryptococcal choroiditis, and pneumocystis choroiditis.3 Beyond ocular opportunistic infections, the most common retinal finding in HIV-positive people is HIV retinopathy, nonspecific microvascular findings in the retina affecting nearly 70% of those with advanced HIV disease. Fortunately, HIV retinopathy is generally asymptomatic.4
►Dr. Swamy. Thank you for those explanations. Based on Dr. Serrao’s differential, it is worth noting that this patient is MSM. He was evaluated in urgent care with the initial examination showing a temperature of 98.0° F, pulse 83 beats per minute, and blood pressure 110/70 mm Hg. The eye exam showed no injection with normal extraocular movements. Initial laboratory data were notable for a CD4 count of 730 cells/mm3 with fewer than 20 HIV viral copies/mL. Cytomegalovirus immunoglobulin G (IgG) was positive, and immunoglobulin M (IgM) was negative. A Lyme antibody was positive with negative IgM and IgG by Western blot. Additional tests can be seen in Tables 1 and 2. The patient has good immunologic and virologic control. How does this change your thinking about the case?
►Dr. Serrao. His CD4 count is well above 350, increasing the likelihood of a relatively uncomplicated course and treatment. Cytomegalovirus antibodies reflect prior infection. As CMV generally does not manifest with disease of any variety (including CMV retinitis) at this high CD4 count, one can presume he does not have CMV retinitis as a cause for his visual changes. CMV retinitis occurs mainly when substantial CD4 depletion has occurred (typically less than 50 cells/mm3). A positive Lyme antibody screen, not specific to Lyme, can be falsely positive in other treponema diseases (eg, Treponema pallidum, the etiologic organism of syphilis) as evidenced by negative confirmatory Western blot IgG and IgM. Antineutrophil cystoplasmic antibodies, lysozyme, angiotensin-converting enzyme, rapid plasma reagin (RPR), herpes simplex virus, toxoplasma are generally included in the workup for the differential of uveitis, retinitis, choroiditis, etc.
►Dr. Swamy. Based on the visual changes, the patient was referred for urgent ophthalmologic evaluation. Dr. Butler, when should a generalist consider urgent ophthalmology referral?
►Dr. Butler. In general, all patients with acute (and significant) vision loss should be referred immediately to an ophthalmologist. The challenge for the general practitioner is determining the true extent of the reported vision loss. If possible, some assessment of visual acuity should be obtained, testing each eye independently and with the correct glasses correction (ie, the patient’s distance glasses if the test object is 12 feet or more from the patient or their reading glasses if the test object is held inside arm’s length). If the general practitioner does not have access to an eye chart or near card, any assessment of vision with an appropriate description will be useful (eg, the patient can quickly count fingers at 15 feet in the unaffected eye, but the eye with reported vision loss cannot reliably count fingers outside of 2 feet). Additional ocular symptoms associated with the vision loss, such as pain, redness, photophobia, new flashes or floaters, increase the urgency of the referral. The threshold for referral for any ocular complaint is lower compared with that of the general population for those with evidence of immunodeficiency, such as for this patient with HIV. Any CD4 count < 200 cells/mm3 should raise the practitioner’s concern for an ocular opportunistic infection, with the greatest concern with CD4 counts < 50 cells/mm3.
►Dr. Swamy. The patient underwent further testing in the ophthalmology clinic. Dr. Butler, can you please interpret the funduscopic exam?
►Dr. Butler. Both eyes demonstrate findings (microaneurysms and small dot-blot hemorrhages) consistent with moderate nonproliferative diabetic retinopathy (Figure 1A, white arrows). HIV-associated retinopathy could produce similar findings, but it is not generally seen with CD4 counts > 200 cells/mm3. Additionally, in the left eye, there is a diffuse patch of retinal whitening (retinitis) associated with the inferotemporal vascular arcades (Figure 1B, white arrows). The entire area involved is poorly circumscribed and the whitening is subtle in areas. Overlying some areas of deeper, ground-glass whitening there are scattered, punctate white spots (Figure 1B, green arrows). Wickremasinghe and colleagues described this pattern of retinitis and suggested that it had a high positive-predictive value in the diagnosis of ocular syphilis.5
►Dr. Swamy. The patient then underwent fluorescein angiography and optical coherence tomography (OCT). Dr. Butler, what did the fluorescein angiography show?
►Dr. Butler. The fluorescein angiogram in both eyes revealed leakage of dye consistent with diabetic retinopathy, with the right eye (OD) worse than the left (OS). Additionally, the areas of active retinitis in the left eye displayed gradual staining with leopard-spot changes, along with late leakage of fluorescein dye, indicating vasculopathy in the infected area (Figure 2, arrows). The patient also underwent OCT in the left eye (images not displayed) demonstrating vitreous cells (vitritis), patches of inner retinal thickening with hyperreflectivity, and hyperreflective nodules at the level of the retinal pigment epithelium with overlying photoreceptor disruption. These OCT findings are fairly stereotypic for syphilitic chorioretinitis.6
►Dr. Swamy. Based on the ophthalmic findings, a diagnosis of ocular syphilis was made. Dr. Serrao, what should internists consider as they evaluate and manage a patient with ocular syphilis?
►Dr. Serrao. Although isolated ocular involvement from syphilis is possible, the majority of patients (up to 85%) with HIV can present with concomitant central nervous system infection and about 30% present with symptomatic neurosyphilis (a typical late manifestation of this disease) that reflects the aggressiveness, accelerated course and propensity for wide dissemination of syphilis in this patient population.7
The presence of concomitant cutaneous rashes should prompt universal precautions, because transmission can occur via skin to skin contact. Clinicians should watch for the Jarisch-Herxheimer reaction during treatment, a syndrome of fever, myalgias, and headache, which results from circulating cytokines produced because of rapidly dying spirochetes that could mimic a penicillin drug reaction, yet is treated supportively.
As syphilis is sexually acquired, clinicians should test for coexistent sexually transmitted infections, vaccinate for those that are preventable (eg, hepatitis B), notify sexual partners via assistance from local departments of public health, and assess for coexistent drug use and offer counseling in order to optimize risk reduction. Special attention should be paid to virologic control of HIV since some studies have shown an increase in the propensity for breakthrough HIV viremia while on effective ART.9 This should warrant counseling for ongoing optimal ART adherence and close monitoring in the follow-up visits with a provider specialized in the treatment of syphilis and HIV.
►Dr. Swamy. A lumbar puncture is performed with the results listed in Table 2. Dr. Serrao, is the CSF consistent with neurosyphilis? What would you do next?
►Dr. Serrao. The lumbar puncture is inflammatory with a lymphocytic predominance, consistent with active ocular/neurosyphilis. The CSF Venereal Disease Research Laboratory test is specific but not sensitive so a negative value does not rule out the presence of central nervous system infection.10 The CSF fluorescent treponemal antibody (CSF FTA-ABS) is sensitive but not specific. In this case, the ocular findings, positive serum RPR, CSF lymphocytic predominance, and CSF FTA ABS strongly supports the diagnosis of ocular/early neurosyphilis in a patient with HIV infection in whom early aggressive treatment is warranted to prevent rapid progression/potential loss of vision.11
►Dr. Swamy. Dr. Butler, how does syphilis behave in the eye as compared to other infectious or inflammatory diseases? Do visual symptoms respond well to treatment?
►Dr. Butler. As opposed to the dramatic reduction in rates and severity of CMV retinitis, HAART has had a negligible effect on ocular syphilis in the setting of HIV coinfection; in fact, rates of syphilis, including ocular syphilis, are currently surging world-wide, and HIV coinfection portends a worse prognosis.12 This is especially true among gay men. More so, there appears to be no correlation between CD4 count and incidence of developing ocular syphilis, as opposed to CMV retinitis, which occurs far more frequently in those with CD4 counts < 50 cells/mm3. In keeping with its epithet as one of the “Great Imitators,” syphilis can affect virtually every tissue of the eye—conjunctiva, sclera, cornea, iris, lens, vitreous, retina, choroid, optic nerve—unlike other OOI, such as CMV or toxoplasma, which generally hone to the retina. Nonetheless, various findings and patterns on clinical exam and ancillary testing, such as the more recently described punctate inner retinitis (as seen in our patient) and the more classic acute syphilitic posterior placoid chorioretinitis, carry high specificity for ocular syphilis.13
Patients with ocular syphilis should be treated according to neurosyphilis treatment protocols. In general, these patients respond very well to treatment with resolution of the ocular findings and recovery of complete, or nearly so, visual function, as long as an excessive delay between diagnosis and proper treatment does not occur.14
►Dr. Swamy. Following this testing, the patient completed 14 days of IV penicillin with resolution of symptoms. He had no further vision complaints. He was started on Triumeq (abacavir, dolutegravir, and lamivudine) with good adherence to therapy. Dr. Serrao, in 2016 the CDC released a clinical advisory about ocular syphilis. Can you tell us about why this is an important diagnosis to be aware of today?
►Dr. Serrao. As with any disease, the epidemiologic characteristics of an infection like syphilis allow the clinician to more carefully entertain such a diagnosis in any one individual by improving the index of suspicion for a particular disease. Awareness of an increase in ocular syphilis in HIV positive MSM allows for a more timely assessment and subsequent treatment with the goal of preventing loss of vision.15
►Lakshmana Swamy, MD, chief medical resident, VA Boston Healthcare System (VABHS) and Boston Medical Center. Dr. Serrao, when you hear about vision changes in a patient with HIV, what differential diagnosis is generated? What epidemiologic or historical factors can help distinguish these entities?
►Richard Serrao, MD, Infectious Disease Service, VABHS and assistant professor of medicine, Boston University School of Medicine. The differential diagnoses for vision changes in a patient with HIV is based on the overall immunosuppression of the patient: the lower the patient’s CD4 count, the higher the number of etiologies.1 The portions of the visual pathway as well as the pattern of vision loss are useful in narrowing the differential. For example, monocular visual disturbances with dermatomal vesicles within the ophthalmic division of the trigeminal nerve strongly implicates varicella zoster retinitis or keratitis; abducens nerve palsy could suggest granulomatous basilar meningitis from cryptococcosis. Likewise, ongoing fevers in an advanced AIDS patient with concomitant colitis, hepatitis, and pneumonitis is strongly suspicious for cytomegalovirus (CMV) retinitis with wide dissemination.
Geographic epidemiologic factors can suggest pathogens more prevalent to certain regions of the world, such as histoplasma chorioretinitis in a resident of the central and eastern U.S. or tuberculosis in a returning traveler. Likewise, a cat owner or one who consumes steak tartare increases the likelihood for toxoplasma retinochoroiditis, or syphilis in men who have sex with men (MSM) in the U.S. given that the majority of new cases occur in this patient population. Other clues one should consider include the presence of splinter hemorrhages in the extremities in an intravenous drug user, raising the possibility of embolic endophthalmitis from bacterial or fungal endocarditis. A variety of other diagnoses can certainly occur as a result of drug treatment (uveitis from rifampin, for example), immune reconstitution from HAART, infections with other HIV-associated pathogens, such as Pneumocystis jiroveci, and many non-HIV-related ocular diseases.
►Dr. Swamy. Dr. Butler, what concerns do you have when you hear about an HIV-infected patient with vision loss from the ophthalmology perspective?
►Nicholas Butler, MD, Ophthalmology Service, Uveitis and Ocular Immunology, VABHS and assistant professor of ophthalmology, Harvard Medical School. Of course, patients with HIV suffer from common causes of vision loss—cataract, glaucoma, diabetes, macular degeneration, for instance—just like those without HIV infection. If there is no significant immunodeficiency, then the patient’s HIV status would be less relevant, and these more common causes of vision loss should be pursued. My first task would be to determine the patient’s most recent CD4 T-cell count.
Assuming an HIV-positive individual is experiencing visual symptoms related to his/her underlying HIV infection (especially in the setting of CD4 counts < 200 cells/mm3), ocular opportunistic infections (OOI) come to mind first. Despite a reduction in incidence of 75% to 80% in the HAART-era, CMV retinitis remains the most common OOI in patients with AIDS and carries the greatest risk of ocular morbidity.2 In fact, based on enrollment data for the Longitudinal Study of the Ocular Complications of AIDS (LSOCA), the prevalence of CMV retinitis among patients with AIDS is more than 20-fold higher than all other ocular complications of AIDS (OOIs and ocular neoplastic disease), including Kaposi sarcoma, lymphoma, herpes zoster ophthalmicus, ocular syphilis, ocular toxoplasma, necrotizing herpetic retinitis, cryptococcal choroiditis, and pneumocystis choroiditis.3 Beyond ocular opportunistic infections, the most common retinal finding in HIV-positive people is HIV retinopathy, nonspecific microvascular findings in the retina affecting nearly 70% of those with advanced HIV disease. Fortunately, HIV retinopathy is generally asymptomatic.4
►Dr. Swamy. Thank you for those explanations. Based on Dr. Serrao’s differential, it is worth noting that this patient is MSM. He was evaluated in urgent care with the initial examination showing a temperature of 98.0° F, pulse 83 beats per minute, and blood pressure 110/70 mm Hg. The eye exam showed no injection with normal extraocular movements. Initial laboratory data were notable for a CD4 count of 730 cells/mm3 with fewer than 20 HIV viral copies/mL. Cytomegalovirus immunoglobulin G (IgG) was positive, and immunoglobulin M (IgM) was negative. A Lyme antibody was positive with negative IgM and IgG by Western blot. Additional tests can be seen in Tables 1 and 2. The patient has good immunologic and virologic control. How does this change your thinking about the case?
►Dr. Serrao. His CD4 count is well above 350, increasing the likelihood of a relatively uncomplicated course and treatment. Cytomegalovirus antibodies reflect prior infection. As CMV generally does not manifest with disease of any variety (including CMV retinitis) at this high CD4 count, one can presume he does not have CMV retinitis as a cause for his visual changes. CMV retinitis occurs mainly when substantial CD4 depletion has occurred (typically less than 50 cells/mm3). A positive Lyme antibody screen, not specific to Lyme, can be falsely positive in other treponema diseases (eg, Treponema pallidum, the etiologic organism of syphilis) as evidenced by negative confirmatory Western blot IgG and IgM. Antineutrophil cystoplasmic antibodies, lysozyme, angiotensin-converting enzyme, rapid plasma reagin (RPR), herpes simplex virus, toxoplasma are generally included in the workup for the differential of uveitis, retinitis, choroiditis, etc.
►Dr. Swamy. Based on the visual changes, the patient was referred for urgent ophthalmologic evaluation. Dr. Butler, when should a generalist consider urgent ophthalmology referral?
►Dr. Butler. In general, all patients with acute (and significant) vision loss should be referred immediately to an ophthalmologist. The challenge for the general practitioner is determining the true extent of the reported vision loss. If possible, some assessment of visual acuity should be obtained, testing each eye independently and with the correct glasses correction (ie, the patient’s distance glasses if the test object is 12 feet or more from the patient or their reading glasses if the test object is held inside arm’s length). If the general practitioner does not have access to an eye chart or near card, any assessment of vision with an appropriate description will be useful (eg, the patient can quickly count fingers at 15 feet in the unaffected eye, but the eye with reported vision loss cannot reliably count fingers outside of 2 feet). Additional ocular symptoms associated with the vision loss, such as pain, redness, photophobia, new flashes or floaters, increase the urgency of the referral. The threshold for referral for any ocular complaint is lower compared with that of the general population for those with evidence of immunodeficiency, such as for this patient with HIV. Any CD4 count < 200 cells/mm3 should raise the practitioner’s concern for an ocular opportunistic infection, with the greatest concern with CD4 counts < 50 cells/mm3.
►Dr. Swamy. The patient underwent further testing in the ophthalmology clinic. Dr. Butler, can you please interpret the funduscopic exam?
►Dr. Butler. Both eyes demonstrate findings (microaneurysms and small dot-blot hemorrhages) consistent with moderate nonproliferative diabetic retinopathy (Figure 1A, white arrows). HIV-associated retinopathy could produce similar findings, but it is not generally seen with CD4 counts > 200 cells/mm3. Additionally, in the left eye, there is a diffuse patch of retinal whitening (retinitis) associated with the inferotemporal vascular arcades (Figure 1B, white arrows). The entire area involved is poorly circumscribed and the whitening is subtle in areas. Overlying some areas of deeper, ground-glass whitening there are scattered, punctate white spots (Figure 1B, green arrows). Wickremasinghe and colleagues described this pattern of retinitis and suggested that it had a high positive-predictive value in the diagnosis of ocular syphilis.5
►Dr. Swamy. The patient then underwent fluorescein angiography and optical coherence tomography (OCT). Dr. Butler, what did the fluorescein angiography show?
►Dr. Butler. The fluorescein angiogram in both eyes revealed leakage of dye consistent with diabetic retinopathy, with the right eye (OD) worse than the left (OS). Additionally, the areas of active retinitis in the left eye displayed gradual staining with leopard-spot changes, along with late leakage of fluorescein dye, indicating vasculopathy in the infected area (Figure 2, arrows). The patient also underwent OCT in the left eye (images not displayed) demonstrating vitreous cells (vitritis), patches of inner retinal thickening with hyperreflectivity, and hyperreflective nodules at the level of the retinal pigment epithelium with overlying photoreceptor disruption. These OCT findings are fairly stereotypic for syphilitic chorioretinitis.6
►Dr. Swamy. Based on the ophthalmic findings, a diagnosis of ocular syphilis was made. Dr. Serrao, what should internists consider as they evaluate and manage a patient with ocular syphilis?
►Dr. Serrao. Although isolated ocular involvement from syphilis is possible, the majority of patients (up to 85%) with HIV can present with concomitant central nervous system infection and about 30% present with symptomatic neurosyphilis (a typical late manifestation of this disease) that reflects the aggressiveness, accelerated course and propensity for wide dissemination of syphilis in this patient population.7
The presence of concomitant cutaneous rashes should prompt universal precautions, because transmission can occur via skin to skin contact. Clinicians should watch for the Jarisch-Herxheimer reaction during treatment, a syndrome of fever, myalgias, and headache, which results from circulating cytokines produced because of rapidly dying spirochetes that could mimic a penicillin drug reaction, yet is treated supportively.
As syphilis is sexually acquired, clinicians should test for coexistent sexually transmitted infections, vaccinate for those that are preventable (eg, hepatitis B), notify sexual partners via assistance from local departments of public health, and assess for coexistent drug use and offer counseling in order to optimize risk reduction. Special attention should be paid to virologic control of HIV since some studies have shown an increase in the propensity for breakthrough HIV viremia while on effective ART.9 This should warrant counseling for ongoing optimal ART adherence and close monitoring in the follow-up visits with a provider specialized in the treatment of syphilis and HIV.
►Dr. Swamy. A lumbar puncture is performed with the results listed in Table 2. Dr. Serrao, is the CSF consistent with neurosyphilis? What would you do next?
►Dr. Serrao. The lumbar puncture is inflammatory with a lymphocytic predominance, consistent with active ocular/neurosyphilis. The CSF Venereal Disease Research Laboratory test is specific but not sensitive so a negative value does not rule out the presence of central nervous system infection.10 The CSF fluorescent treponemal antibody (CSF FTA-ABS) is sensitive but not specific. In this case, the ocular findings, positive serum RPR, CSF lymphocytic predominance, and CSF FTA ABS strongly supports the diagnosis of ocular/early neurosyphilis in a patient with HIV infection in whom early aggressive treatment is warranted to prevent rapid progression/potential loss of vision.11
►Dr. Swamy. Dr. Butler, how does syphilis behave in the eye as compared to other infectious or inflammatory diseases? Do visual symptoms respond well to treatment?
►Dr. Butler. As opposed to the dramatic reduction in rates and severity of CMV retinitis, HAART has had a negligible effect on ocular syphilis in the setting of HIV coinfection; in fact, rates of syphilis, including ocular syphilis, are currently surging world-wide, and HIV coinfection portends a worse prognosis.12 This is especially true among gay men. More so, there appears to be no correlation between CD4 count and incidence of developing ocular syphilis, as opposed to CMV retinitis, which occurs far more frequently in those with CD4 counts < 50 cells/mm3. In keeping with its epithet as one of the “Great Imitators,” syphilis can affect virtually every tissue of the eye—conjunctiva, sclera, cornea, iris, lens, vitreous, retina, choroid, optic nerve—unlike other OOI, such as CMV or toxoplasma, which generally hone to the retina. Nonetheless, various findings and patterns on clinical exam and ancillary testing, such as the more recently described punctate inner retinitis (as seen in our patient) and the more classic acute syphilitic posterior placoid chorioretinitis, carry high specificity for ocular syphilis.13
Patients with ocular syphilis should be treated according to neurosyphilis treatment protocols. In general, these patients respond very well to treatment with resolution of the ocular findings and recovery of complete, or nearly so, visual function, as long as an excessive delay between diagnosis and proper treatment does not occur.14
►Dr. Swamy. Following this testing, the patient completed 14 days of IV penicillin with resolution of symptoms. He had no further vision complaints. He was started on Triumeq (abacavir, dolutegravir, and lamivudine) with good adherence to therapy. Dr. Serrao, in 2016 the CDC released a clinical advisory about ocular syphilis. Can you tell us about why this is an important diagnosis to be aware of today?
►Dr. Serrao. As with any disease, the epidemiologic characteristics of an infection like syphilis allow the clinician to more carefully entertain such a diagnosis in any one individual by improving the index of suspicion for a particular disease. Awareness of an increase in ocular syphilis in HIV positive MSM allows for a more timely assessment and subsequent treatment with the goal of preventing loss of vision.15
1. Cunningham ET Jr, Margolis TP. Ocular manifestations of HIV infection. N Engl J Med. 1998;339(4):236-244.
2. Holtzer CD, Jacobson MA, Hadley WK, et al. Decline in the rate of specific opportunistic infections at San Francisco General Hospital, 1994-1997. AIDS. 1998;12(14):1931-1933.
3. Gangaputra S, Drye L, Vaidya V, Thorne JE, Jabs DA, Lyon AT. Non-cytomegalovirus ocular opportunistic infections in patients with acquired immunodeficiency syndrome. Am J Ophthalmol. 2013;155(2):206-212.e205.
4. Jabs DA, Van Natta ML, Holbrook JT, et al. Longitudinal study of the ocular complications of AIDS: 1. Ocular diagnoses at enrollment. Ophthalmology. 2007;114(4):780-786.
5. Wickremasinghe S, Ling C, Stawell R, Yeoh J, Hall A, Zamir E. Syphilitic punctate inner retinitis in immunocompetent gay men. Ophthalmology. 2009;116(6):1195-1200.
6. Burkholder BM, Leung TG, Ostheimer TA, Butler NJ, Thorne JE, Dunn JP. Spectral domain optical coherence tomography findings in acute syphilitic posterior placoid chorioretinitis. J Ophthalmic Inflamm Infect. 2014;4(1):2.
7. Musher DM, Hamill RJ, Baughn RE. Effect of human immunodeficiency virus (HIV) infection on the course of syphilis and on the response to treatment. Ann Intern Med. 1990;113(11):872-881.
8. Lukehart SA, Hook EW 3rd, Baker-Zander SA, Collier AC, Critchlow CW, Handsfield HH. Invasion of the central nervous system by Treponema pallidum: implications for diagnosis and treatment. Ann Intern Med. 1988;109(11):855-862.
9. Golden MR, Marra CM, Holmes KK. Update on syphilis: resurgence of an old problem. JAMA. 2003;290(11):1510-1514.
10. Marra CM, Tantalo LC, Maxwell CL, Ho EL, Sahi SK, Jones T. The rapid plasma reagin test cannot replace the venereal disease research laboratory test for neurosyphilis diagnosis. Sex Transm Dis. 2012;39(6):453-457.
11. Harding AS, Ghanem KG. The performance of cerebrospinal fluid treponemal-specific antibody tests in neurosyphilis: a systematic review. Sex Transm Dis. 2012;39(4):291-297.
12. Butler NJ, Thorne JE. Current status of HIV infection and ocular disease. Curr Opin Ophthalmol. 2012;23(6):517-522.
13. Gass JD, Braunstein RA, Chenoweth RG. Acute syphilitic posterior placoid chorioretinitis. Ophthalmology. 1990;97(10):1288-1297.
14. Davis JL. Ocular syphilis. Curr Opin Ophthalmol. 2014;25(6):513-518.
15. Clinical Advisory: Ocular Syphilis in the United States. https://www.cdc.gov/std/syphilis/clinicaladvisoryos2015.htm. Accessed September 11, 2017.
1. Cunningham ET Jr, Margolis TP. Ocular manifestations of HIV infection. N Engl J Med. 1998;339(4):236-244.
2. Holtzer CD, Jacobson MA, Hadley WK, et al. Decline in the rate of specific opportunistic infections at San Francisco General Hospital, 1994-1997. AIDS. 1998;12(14):1931-1933.
3. Gangaputra S, Drye L, Vaidya V, Thorne JE, Jabs DA, Lyon AT. Non-cytomegalovirus ocular opportunistic infections in patients with acquired immunodeficiency syndrome. Am J Ophthalmol. 2013;155(2):206-212.e205.
4. Jabs DA, Van Natta ML, Holbrook JT, et al. Longitudinal study of the ocular complications of AIDS: 1. Ocular diagnoses at enrollment. Ophthalmology. 2007;114(4):780-786.
5. Wickremasinghe S, Ling C, Stawell R, Yeoh J, Hall A, Zamir E. Syphilitic punctate inner retinitis in immunocompetent gay men. Ophthalmology. 2009;116(6):1195-1200.
6. Burkholder BM, Leung TG, Ostheimer TA, Butler NJ, Thorne JE, Dunn JP. Spectral domain optical coherence tomography findings in acute syphilitic posterior placoid chorioretinitis. J Ophthalmic Inflamm Infect. 2014;4(1):2.
7. Musher DM, Hamill RJ, Baughn RE. Effect of human immunodeficiency virus (HIV) infection on the course of syphilis and on the response to treatment. Ann Intern Med. 1990;113(11):872-881.
8. Lukehart SA, Hook EW 3rd, Baker-Zander SA, Collier AC, Critchlow CW, Handsfield HH. Invasion of the central nervous system by Treponema pallidum: implications for diagnosis and treatment. Ann Intern Med. 1988;109(11):855-862.
9. Golden MR, Marra CM, Holmes KK. Update on syphilis: resurgence of an old problem. JAMA. 2003;290(11):1510-1514.
10. Marra CM, Tantalo LC, Maxwell CL, Ho EL, Sahi SK, Jones T. The rapid plasma reagin test cannot replace the venereal disease research laboratory test for neurosyphilis diagnosis. Sex Transm Dis. 2012;39(6):453-457.
11. Harding AS, Ghanem KG. The performance of cerebrospinal fluid treponemal-specific antibody tests in neurosyphilis: a systematic review. Sex Transm Dis. 2012;39(4):291-297.
12. Butler NJ, Thorne JE. Current status of HIV infection and ocular disease. Curr Opin Ophthalmol. 2012;23(6):517-522.
13. Gass JD, Braunstein RA, Chenoweth RG. Acute syphilitic posterior placoid chorioretinitis. Ophthalmology. 1990;97(10):1288-1297.
14. Davis JL. Ocular syphilis. Curr Opin Ophthalmol. 2014;25(6):513-518.
15. Clinical Advisory: Ocular Syphilis in the United States. https://www.cdc.gov/std/syphilis/clinicaladvisoryos2015.htm. Accessed September 11, 2017.