User login
Radiologically Isolated Syndrome: A condition that often precedes an MS diagnosis in children
Naila Makhani, MD completed medical school training at the University of British Columbia (Vancouver, Canada). This was followed by a residency in child neurology and fellowship in MS and other demyelinating diseases at the University of Toronto and The Hospital for Sick Children (Toronto, Canada). Concurrent with fellowship training, Dr. Makhani obtained a Masters’ degree in public health from Harvard University. Dr. Makhani is the Director of the Pediatric MS Program at Yale and the lead investigator of a multi-center international study examining outcomes following the radiologically isolated syndrome in children.
Q1. Could you please provide an overview of Radiologically Isolated Syndrome ?
A1. Radiologically Isolated Syndrome (RIS) was first described in adults in 2009. Since then it has also been increasingly recognized and diagnosed in children. RIS is diagnosed after an MRI of the brain that the patient has sought for reasons other than suspected multiple sclerosis-- for instance, for evaluation of head trauma or headache. However, unexpectedly or incidentally, the patient’s MRI shows the typical findings that we see in multiple sclerosis, even in the absence of any typical clinical symptoms. RIS is generally considered a rare syndrome.
Q2. You created Yale Medicine’s Pediatric Multiple Sclerosis program which advocates for the eradication of MS. What criteria defines a rare disease? Does RIS meet these criteria? And if so, how?
A2. The criteria for a rare disease vary, depending on the reference. In the US, a rare disease is defined as a condition that affects fewer than 200,000 people, in total, across the country. By contrast, in Europe, a disease is considered rare if it affects fewer than one in every 2,000 people within the country’s population.
In the case of RIS, especially in children, we suspect that this is a rare condition, but we don't know for sure, as there have been very few population-based studies. There is one large study that was conducted in Europe that found one case of RIS among approximately 5,000 otherwise healthy children, who were between 7 and 14 years of age. I think that's our best estimate of the overall prevalence of RIS in children. Using that finding, it likely would qualify as a rare condition, although, as I said, we really don't know for sure, as the prevalence may vary among different populations or age groups.
Q3. How do you investigate and manage RIS in children? What are some of the challenges?
A3. For children with radiologically isolated syndrome, we usually undertake a comprehensive workup. This includes a detailed clinical neurological exam to ensure that there are no abnormalities that would, for instance, suggest a misdiagnosis of multiple sclerosis or an alternative diagnosis. In addition to the brain MRI, we usually obtain an MRI of the spinal cord to determine whether there is any spinal cord involvement. We also obtain blood tests. We often analyze spinal fluid as well, primarily to exclude other alternative processes that may explain the MRI findings. A key challenge in this field is that there are currently no formal guidelines for the investigation and management of children with RIS. Collaborations within the pediatric MS community are needed to develop such consensus approaches to standardize care.
Q4. What are the most significant risk factors that indicate children with RIS could one day develop multiple sclerosis?
A4.This is an area of active research within our group. So far, we've found that approximately 42% of children with RIS develop multiple sclerosis in the future; on average, two years following their first abnormal MRI. Therefore, this is a high-risk group for developing multiple sclerosis in the future. Thus far, we've determined that in children with RIS, it is the presence of abnormal spinal cord imaging and an abnormality in spinal fluid – namely, the presence of oligoclonal bands – that are likely the predictors of whether these children could develop MS in the future. a child’s possible development
Q5. Based on your recent studies, are there data in children highlighting the potential for higher prevalence in one population over another?
A5. Thus far, population-based studies assessing RIS, especially in children, have been rare and thus far have not identified particular subgroups with increased prevalence. We do know that the prevalence of multiple sclerosis varies across different age groups and across gender. Whether such associations are also present for RIS is an area of active research.
de Mol CL, Bruijstens AL, Jansen PR, Dremmen M, Wong Y, van der Lugt A, White T, Neuteboom RF.Mult Scler. 2021 Oct;27(11):1790-1793. doi: 10.1177/1352458521989220. Epub 2021 Jan 22.PMID: 33480814
2. Radiologically isolated syndrome in children: Clinical and radiologic outcomes.
Makhani N, Lebrun C, Siva A, Brassat D, Carra Dallière C, de Seze J, Du W, Durand Dubief F, Kantarci O, Langille M, Narula S, Pelletier J, Rojas JI, Shapiro ED, Stone RT, Tintoré M, Uygunoglu U, Vermersch P, Wassmer E, Okuda DT, Pelletier D.Neurol Neuroimmunol Neuroinflamm. 2017 Sep 25;4(6):e395. doi: 10.1212/NXI.0000000000000395. eCollection 2017 Nov.PMID: 28959703
Makhani N, Lebrun C, Siva A, Narula S, Wassmer E, Brassat D, Brenton JN, Cabre P, Carra Dallière C, de Seze J, Durand Dubief F, Inglese M, Langille M, Mathey G, Neuteboom RF, Pelletier J, Pohl D, Reich DS, Ignacio Rojas J, Shabanova V, Shapiro ED, Stone RT, Tenembaum S, Tintoré M, Uygunoglu U, Vargas W, Venkateswaren S, Vermersch P, Kantarci O, Okuda DT, Pelletier D; Observatoire Francophone de la Sclérose en Plaques (OFSEP), Société Francophone de la Sclérose en Plaques (SFSEP), the Radiologically Isolated Syndrome Consortium (RISC) and the Pediatric Radiologically Isolated Syndrome Consortium (PARIS).Mult Scler J Exp Transl Clin. 2019 Mar 20;5(1):2055217319836664. doi: 10.1177/2055217319836664. eCollection 2019 Jan-Mar.PMID: 30915227
Naila Makhani, MD completed medical school training at the University of British Columbia (Vancouver, Canada). This was followed by a residency in child neurology and fellowship in MS and other demyelinating diseases at the University of Toronto and The Hospital for Sick Children (Toronto, Canada). Concurrent with fellowship training, Dr. Makhani obtained a Masters’ degree in public health from Harvard University. Dr. Makhani is the Director of the Pediatric MS Program at Yale and the lead investigator of a multi-center international study examining outcomes following the radiologically isolated syndrome in children.
Q1. Could you please provide an overview of Radiologically Isolated Syndrome ?
A1. Radiologically Isolated Syndrome (RIS) was first described in adults in 2009. Since then it has also been increasingly recognized and diagnosed in children. RIS is diagnosed after an MRI of the brain that the patient has sought for reasons other than suspected multiple sclerosis-- for instance, for evaluation of head trauma or headache. However, unexpectedly or incidentally, the patient’s MRI shows the typical findings that we see in multiple sclerosis, even in the absence of any typical clinical symptoms. RIS is generally considered a rare syndrome.
Q2. You created Yale Medicine’s Pediatric Multiple Sclerosis program which advocates for the eradication of MS. What criteria defines a rare disease? Does RIS meet these criteria? And if so, how?
A2. The criteria for a rare disease vary, depending on the reference. In the US, a rare disease is defined as a condition that affects fewer than 200,000 people, in total, across the country. By contrast, in Europe, a disease is considered rare if it affects fewer than one in every 2,000 people within the country’s population.
In the case of RIS, especially in children, we suspect that this is a rare condition, but we don't know for sure, as there have been very few population-based studies. There is one large study that was conducted in Europe that found one case of RIS among approximately 5,000 otherwise healthy children, who were between 7 and 14 years of age. I think that's our best estimate of the overall prevalence of RIS in children. Using that finding, it likely would qualify as a rare condition, although, as I said, we really don't know for sure, as the prevalence may vary among different populations or age groups.
Q3. How do you investigate and manage RIS in children? What are some of the challenges?
A3. For children with radiologically isolated syndrome, we usually undertake a comprehensive workup. This includes a detailed clinical neurological exam to ensure that there are no abnormalities that would, for instance, suggest a misdiagnosis of multiple sclerosis or an alternative diagnosis. In addition to the brain MRI, we usually obtain an MRI of the spinal cord to determine whether there is any spinal cord involvement. We also obtain blood tests. We often analyze spinal fluid as well, primarily to exclude other alternative processes that may explain the MRI findings. A key challenge in this field is that there are currently no formal guidelines for the investigation and management of children with RIS. Collaborations within the pediatric MS community are needed to develop such consensus approaches to standardize care.
Q4. What are the most significant risk factors that indicate children with RIS could one day develop multiple sclerosis?
A4.This is an area of active research within our group. So far, we've found that approximately 42% of children with RIS develop multiple sclerosis in the future; on average, two years following their first abnormal MRI. Therefore, this is a high-risk group for developing multiple sclerosis in the future. Thus far, we've determined that in children with RIS, it is the presence of abnormal spinal cord imaging and an abnormality in spinal fluid – namely, the presence of oligoclonal bands – that are likely the predictors of whether these children could develop MS in the future. a child’s possible development
Q5. Based on your recent studies, are there data in children highlighting the potential for higher prevalence in one population over another?
A5. Thus far, population-based studies assessing RIS, especially in children, have been rare and thus far have not identified particular subgroups with increased prevalence. We do know that the prevalence of multiple sclerosis varies across different age groups and across gender. Whether such associations are also present for RIS is an area of active research.
Naila Makhani, MD completed medical school training at the University of British Columbia (Vancouver, Canada). This was followed by a residency in child neurology and fellowship in MS and other demyelinating diseases at the University of Toronto and The Hospital for Sick Children (Toronto, Canada). Concurrent with fellowship training, Dr. Makhani obtained a Masters’ degree in public health from Harvard University. Dr. Makhani is the Director of the Pediatric MS Program at Yale and the lead investigator of a multi-center international study examining outcomes following the radiologically isolated syndrome in children.
Q1. Could you please provide an overview of Radiologically Isolated Syndrome ?
A1. Radiologically Isolated Syndrome (RIS) was first described in adults in 2009. Since then it has also been increasingly recognized and diagnosed in children. RIS is diagnosed after an MRI of the brain that the patient has sought for reasons other than suspected multiple sclerosis-- for instance, for evaluation of head trauma or headache. However, unexpectedly or incidentally, the patient’s MRI shows the typical findings that we see in multiple sclerosis, even in the absence of any typical clinical symptoms. RIS is generally considered a rare syndrome.
Q2. You created Yale Medicine’s Pediatric Multiple Sclerosis program which advocates for the eradication of MS. What criteria defines a rare disease? Does RIS meet these criteria? And if so, how?
A2. The criteria for a rare disease vary, depending on the reference. In the US, a rare disease is defined as a condition that affects fewer than 200,000 people, in total, across the country. By contrast, in Europe, a disease is considered rare if it affects fewer than one in every 2,000 people within the country’s population.
In the case of RIS, especially in children, we suspect that this is a rare condition, but we don't know for sure, as there have been very few population-based studies. There is one large study that was conducted in Europe that found one case of RIS among approximately 5,000 otherwise healthy children, who were between 7 and 14 years of age. I think that's our best estimate of the overall prevalence of RIS in children. Using that finding, it likely would qualify as a rare condition, although, as I said, we really don't know for sure, as the prevalence may vary among different populations or age groups.
Q3. How do you investigate and manage RIS in children? What are some of the challenges?
A3. For children with radiologically isolated syndrome, we usually undertake a comprehensive workup. This includes a detailed clinical neurological exam to ensure that there are no abnormalities that would, for instance, suggest a misdiagnosis of multiple sclerosis or an alternative diagnosis. In addition to the brain MRI, we usually obtain an MRI of the spinal cord to determine whether there is any spinal cord involvement. We also obtain blood tests. We often analyze spinal fluid as well, primarily to exclude other alternative processes that may explain the MRI findings. A key challenge in this field is that there are currently no formal guidelines for the investigation and management of children with RIS. Collaborations within the pediatric MS community are needed to develop such consensus approaches to standardize care.
Q4. What are the most significant risk factors that indicate children with RIS could one day develop multiple sclerosis?
A4.This is an area of active research within our group. So far, we've found that approximately 42% of children with RIS develop multiple sclerosis in the future; on average, two years following their first abnormal MRI. Therefore, this is a high-risk group for developing multiple sclerosis in the future. Thus far, we've determined that in children with RIS, it is the presence of abnormal spinal cord imaging and an abnormality in spinal fluid – namely, the presence of oligoclonal bands – that are likely the predictors of whether these children could develop MS in the future. a child’s possible development
Q5. Based on your recent studies, are there data in children highlighting the potential for higher prevalence in one population over another?
A5. Thus far, population-based studies assessing RIS, especially in children, have been rare and thus far have not identified particular subgroups with increased prevalence. We do know that the prevalence of multiple sclerosis varies across different age groups and across gender. Whether such associations are also present for RIS is an area of active research.
de Mol CL, Bruijstens AL, Jansen PR, Dremmen M, Wong Y, van der Lugt A, White T, Neuteboom RF.Mult Scler. 2021 Oct;27(11):1790-1793. doi: 10.1177/1352458521989220. Epub 2021 Jan 22.PMID: 33480814
2. Radiologically isolated syndrome in children: Clinical and radiologic outcomes.
Makhani N, Lebrun C, Siva A, Brassat D, Carra Dallière C, de Seze J, Du W, Durand Dubief F, Kantarci O, Langille M, Narula S, Pelletier J, Rojas JI, Shapiro ED, Stone RT, Tintoré M, Uygunoglu U, Vermersch P, Wassmer E, Okuda DT, Pelletier D.Neurol Neuroimmunol Neuroinflamm. 2017 Sep 25;4(6):e395. doi: 10.1212/NXI.0000000000000395. eCollection 2017 Nov.PMID: 28959703
Makhani N, Lebrun C, Siva A, Narula S, Wassmer E, Brassat D, Brenton JN, Cabre P, Carra Dallière C, de Seze J, Durand Dubief F, Inglese M, Langille M, Mathey G, Neuteboom RF, Pelletier J, Pohl D, Reich DS, Ignacio Rojas J, Shabanova V, Shapiro ED, Stone RT, Tenembaum S, Tintoré M, Uygunoglu U, Vargas W, Venkateswaren S, Vermersch P, Kantarci O, Okuda DT, Pelletier D; Observatoire Francophone de la Sclérose en Plaques (OFSEP), Société Francophone de la Sclérose en Plaques (SFSEP), the Radiologically Isolated Syndrome Consortium (RISC) and the Pediatric Radiologically Isolated Syndrome Consortium (PARIS).Mult Scler J Exp Transl Clin. 2019 Mar 20;5(1):2055217319836664. doi: 10.1177/2055217319836664. eCollection 2019 Jan-Mar.PMID: 30915227
de Mol CL, Bruijstens AL, Jansen PR, Dremmen M, Wong Y, van der Lugt A, White T, Neuteboom RF.Mult Scler. 2021 Oct;27(11):1790-1793. doi: 10.1177/1352458521989220. Epub 2021 Jan 22.PMID: 33480814
2. Radiologically isolated syndrome in children: Clinical and radiologic outcomes.
Makhani N, Lebrun C, Siva A, Brassat D, Carra Dallière C, de Seze J, Du W, Durand Dubief F, Kantarci O, Langille M, Narula S, Pelletier J, Rojas JI, Shapiro ED, Stone RT, Tintoré M, Uygunoglu U, Vermersch P, Wassmer E, Okuda DT, Pelletier D.Neurol Neuroimmunol Neuroinflamm. 2017 Sep 25;4(6):e395. doi: 10.1212/NXI.0000000000000395. eCollection 2017 Nov.PMID: 28959703
Makhani N, Lebrun C, Siva A, Narula S, Wassmer E, Brassat D, Brenton JN, Cabre P, Carra Dallière C, de Seze J, Durand Dubief F, Inglese M, Langille M, Mathey G, Neuteboom RF, Pelletier J, Pohl D, Reich DS, Ignacio Rojas J, Shabanova V, Shapiro ED, Stone RT, Tenembaum S, Tintoré M, Uygunoglu U, Vargas W, Venkateswaren S, Vermersch P, Kantarci O, Okuda DT, Pelletier D; Observatoire Francophone de la Sclérose en Plaques (OFSEP), Société Francophone de la Sclérose en Plaques (SFSEP), the Radiologically Isolated Syndrome Consortium (RISC) and the Pediatric Radiologically Isolated Syndrome Consortium (PARIS).Mult Scler J Exp Transl Clin. 2019 Mar 20;5(1):2055217319836664. doi: 10.1177/2055217319836664. eCollection 2019 Jan-Mar.PMID: 30915227
Early-Stage NSCLC Highlights From ESMO 2021
Benjamin Cooper, MD, director of Proton Therapy services at NYU Langone Health, shares key findings from early-stage non-small cell lung cancer (NSCLC) trials presented at the 2021 ESMO Congress.
Dr Cooper begins with the LungART trial, which evaluated whether postoperative radiotherapy (PORT) would benefit patients with completely resected NSCLC and mediastinal N2 involvement. Use of PORT reduced the risk of mediastinal relapse but did not show significant impact on disease-free survival (DFS).
Next, he turns to findings from the COAST trial, which compared durvalumab monotherapy, durvalumab plus oleclumab, and durvalumab plus monalizumab in patients with locally advanced, unresectable stage III NSCLC. Both combination regimens increased the objective response rate and significantly improved progression-free survival (PFS) vs durvalumab alone.
Dr Cooper also reviews sites of disease relapse and post-relapse treatment from IMpower010, which evaluated atezolizumab versus best supportive care after adjuvant chemotherapy in patients with resected stage IB-IIIA NSCLC. Similar patterns of relapse were seen across study arms, but patients with PD-L1 levels of 50% or higher experienced greatest DFS benefits.
Lastly, Dr Cooper highlights GEMSTONE-301, which tested the novel anti-PD-L1 drug sugemalimab in patients with unresectable, stage III NSCLC who did not progress after concurrent or sequential radiotherapy. There was a statistically significant and clinically meaningful PFS improvement in patients receiving sugemalimab compared to placebo.
--
Benjamin Cooper, MD, Assistant Professor, Department of Radiation Oncology, Director, Proton Therapy Services, NYU Grossman School of Medicine, New York, New York
Benjamin Cooper, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AstraZeneca.
Benjamin Cooper, MD, director of Proton Therapy services at NYU Langone Health, shares key findings from early-stage non-small cell lung cancer (NSCLC) trials presented at the 2021 ESMO Congress.
Dr Cooper begins with the LungART trial, which evaluated whether postoperative radiotherapy (PORT) would benefit patients with completely resected NSCLC and mediastinal N2 involvement. Use of PORT reduced the risk of mediastinal relapse but did not show significant impact on disease-free survival (DFS).
Next, he turns to findings from the COAST trial, which compared durvalumab monotherapy, durvalumab plus oleclumab, and durvalumab plus monalizumab in patients with locally advanced, unresectable stage III NSCLC. Both combination regimens increased the objective response rate and significantly improved progression-free survival (PFS) vs durvalumab alone.
Dr Cooper also reviews sites of disease relapse and post-relapse treatment from IMpower010, which evaluated atezolizumab versus best supportive care after adjuvant chemotherapy in patients with resected stage IB-IIIA NSCLC. Similar patterns of relapse were seen across study arms, but patients with PD-L1 levels of 50% or higher experienced greatest DFS benefits.
Lastly, Dr Cooper highlights GEMSTONE-301, which tested the novel anti-PD-L1 drug sugemalimab in patients with unresectable, stage III NSCLC who did not progress after concurrent or sequential radiotherapy. There was a statistically significant and clinically meaningful PFS improvement in patients receiving sugemalimab compared to placebo.
--
Benjamin Cooper, MD, Assistant Professor, Department of Radiation Oncology, Director, Proton Therapy Services, NYU Grossman School of Medicine, New York, New York
Benjamin Cooper, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AstraZeneca.
Benjamin Cooper, MD, director of Proton Therapy services at NYU Langone Health, shares key findings from early-stage non-small cell lung cancer (NSCLC) trials presented at the 2021 ESMO Congress.
Dr Cooper begins with the LungART trial, which evaluated whether postoperative radiotherapy (PORT) would benefit patients with completely resected NSCLC and mediastinal N2 involvement. Use of PORT reduced the risk of mediastinal relapse but did not show significant impact on disease-free survival (DFS).
Next, he turns to findings from the COAST trial, which compared durvalumab monotherapy, durvalumab plus oleclumab, and durvalumab plus monalizumab in patients with locally advanced, unresectable stage III NSCLC. Both combination regimens increased the objective response rate and significantly improved progression-free survival (PFS) vs durvalumab alone.
Dr Cooper also reviews sites of disease relapse and post-relapse treatment from IMpower010, which evaluated atezolizumab versus best supportive care after adjuvant chemotherapy in patients with resected stage IB-IIIA NSCLC. Similar patterns of relapse were seen across study arms, but patients with PD-L1 levels of 50% or higher experienced greatest DFS benefits.
Lastly, Dr Cooper highlights GEMSTONE-301, which tested the novel anti-PD-L1 drug sugemalimab in patients with unresectable, stage III NSCLC who did not progress after concurrent or sequential radiotherapy. There was a statistically significant and clinically meaningful PFS improvement in patients receiving sugemalimab compared to placebo.
--
Benjamin Cooper, MD, Assistant Professor, Department of Radiation Oncology, Director, Proton Therapy Services, NYU Grossman School of Medicine, New York, New York
Benjamin Cooper, MD, has disclosed the following relevant financial relationships:
Serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for: AstraZeneca.

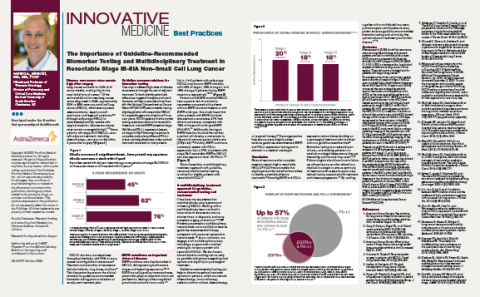
The Importance of Guideline-Recommended Biomarker Testing and Multidisciplinary Treatment in Resectable Stage IB-IIIA Non-Small Cell Lung Cancer
In this article, Gerard A. Silvestri, MD, MS, FCCP discusses guideline-recommended testing and multidisciplinary care for resectable non-small cell lung cancer.
Neither the editors of CHEST Physician® nor the Editorial Advisory Board nor the reporting staff contributed to this content.
In this article, Gerard A. Silvestri, MD, MS, FCCP discusses guideline-recommended testing and multidisciplinary care for resectable non-small cell lung cancer.
Neither the editors of CHEST Physician® nor the Editorial Advisory Board nor the reporting staff contributed to this content.
In this article, Gerard A. Silvestri, MD, MS, FCCP discusses guideline-recommended testing and multidisciplinary care for resectable non-small cell lung cancer.
Neither the editors of CHEST Physician® nor the Editorial Advisory Board nor the reporting staff contributed to this content.
Transforming Primary Care Clinical Learning Environments to Optimize Education, Outcomes, and Satisfaction
A broad consensus exists that US health care is now becoming more complex than at any other time in prior decades, potentially contributing to less than optimal outcomes, inadequate or unnecessary care, dissatisfied users, burned-out providers, and excessive costs.1 To reduce health system dysfunction, experts have looked to primary care to improve care continuity, coordination, and quality. The patient-centered medical home was designed to create environments where patients can access skilled professionals for both immediate and long-term needs across the health care spectrum, including nursing, pharmacy, social work, mental health, care coordinators, and educators.2
In 2010, the VHA of the Department of Veterans Affairs (VA) introduced a patient-centered model of primary care known as the patient-aligned care team (PACT). Each enrolled veteran is assigned to a PACT that is staffed by the enrollee’s personal provider, clinical staff, and appropriate professionals who work together to respond to patients in the context of their unique needs. In addition to the primary care provider (physician, physician assistant, or nurse practitioner), a nurse care manager, licensed vocational nurse or medical assistant, and an administrative professional, each PACT team is staffed by pharmacists, social workers, and mental health specialists. An especially important, and possibly unique, aspect of the VA PACT model is the integration of traditional primary care services with mental health access and care. This clinical interprofessional collaboration requires new educational strategies to effectively train a workforce qualified to work in, lead, and improve these settings.3
Although clinical environments are undergoing rapid change, curriculum for the health professions trainees has not adapted as quickly, even though it has been widely recognized that both should evolve concurrently.4 Curriculum emphasizing interprofessional practice, in particular, has been insufficiently implemented in educational settings.5 Static clinical learning environments pose a risk to future systems that will flounder without prepared professionals.6 Professional organizations, consensus groups, and medical education expert recommendations to implement interprofessional training environments have been met with relatively slow uptake in part because the challenges to implementation of scalable platforms for interprofessional clinical education are not trivial.7-9
This issue of Federal Practitioner introduces the first of 5 case studies that describe the implementation of instructional strategies designed and implemented by faculty, staff, and trainees. Each case embodies a unique approach to curriculum design and implementation that illustrates the collaborative innovation required to engage trainees with patients, with one another from differing professions, and with their faculty. The required flattening of the traditional hierarchy of staff in medical settings necessitates modification of clinical faculty and trainees skills. Didactic sessions are limited, and the focus is on experiential teaching and learning.10
As will be seen through the lenses of the cases presented in this series, the investments (including the time line to shift attitudes and change culture) required to achieve measurable outcomes are substantial. These investments not only are monetary, but also include addressing change management, conflict resolution, enhancement of communication skills, employee engagement, and leadership development.
The VA supports a comprehensive health system distributed throughout the nation with more than 1,000 points of care and more than 150 medical centers. Less recognized is that VA is the largest clinical learning platform in the US: More than 120,000 students and trainees enrolled in more than 40 different health professions and disciplines participate in VA clinical training programs annually.11 The VA has incorporated multiple innovative care designs, such as PACTs, along with educational and clinical leadership to create experiential workplace learning environments where structure, processes, and outcomes can be observed, adjusted, measured, and potentially duplicated.
This approach was key for the initial 5 of the current 7 Centers of Excellence in Primary Care Education (CoEPCEs) launched by VA in 2011, and from which the 5 cases in this series have evolved.12 The CoEPCE was developed as a demonstration project to show how to develop the interprofessional primary care curriculum for health professions that the PACT model requires. The CoEPCE, having trained more than 1,000 learners to date, has informed the PACT model to distinguish between PACTs whose mission is to provide clinical care from those that have the additional role of educating health professions trainees. The PACTS with this additional obligation are called interprofessional academic PACTs (iAPACTs). The iAPACTs incorporate features to accommodate clinical teaching and learning, including logistic challenges of scheduling, additional space requirements, faculty assignments, and affiliations with the academic institutions that sponsor the training programs.
Foundational concepts of the CoEPCE include those inherent in primary care, plus interprofessional practice where trainees of multiple professions are integrated into the care model to create a transformed workplace learning environment.13,14 Curricular domains of shared decision making among team members and their veteran patients, interprofessional collaboration, sustained relationships, and performance improvement are all required elements integral to the design and implementation of all CoEPCEs.13 This purposeful design provides clinical and educational infrastructure for interprofessional practice that simultaneously and seamlessly integrates both priorities of transforming clinical care and education.
The vision is to create the clinical learning environments necessary to produce the high-functioning individuals and teams needed to assure beneficial patient care outcomes as well as professional and personal satisfaction within the care team. The goal is to improve the PACT model of care in VA as a vehicle to enhance primary care services, to support changes in policy and practice that improve veterans’ care, safety, experience, health and well-being, and prepare a highly skilled future workforce for VA and for the nation as a whole.15
As all the cases in this series illustrate, the trainees are deeply embedded into clinical care and—very importantly—processes of patient care provision in consideration of all the patients care needs, using a holistic care model. As integrated team members, trainees from multiple professions learn with, from, and about one another as professionals and, as importantly, learn to appreciate the array of skills each brings to patient care, thus transforming their personal as well as professional learning experience. A highly relevant finding is that faculty and leadership—along with the trainees—have also learned, benefited, and transformed their thinking and attitudes, contributing to a cultural shift that is less hierarchical and more inclusive of all team members. A recently released external evaluation of the CoEPCE in their iAPACT environments indicates promising patterns of clinical outcomes with indications of improved staff satisfaction and less burnout. Better understanding of these innovations across and beyond the evaluated sites will be the topic of subsequent inquiries.16
These case studies demonstrate how education can be designed to advance the quality of care and improve the clinical teaching and learning environment, and educational outcomes. These cases are not intended to be recipes but rather exemplify the ingredients required to provide enough information and background to illustrate the transformational process. Superficially the cases may seem simple, but deeper examination reveals the complexity of confronting the challenges of day-to-day clinical work and redesigning both clinical and educational parameters.
These are real cases about real people working hard to revise a fragmented system and build a better future. The true purpose of these case studies is to inspire others to pursue educational modernization and excellence. In fact, there is no other satisfactory choice.
1. Dzau VJ, McClellan MB, McGinnis M, Finkelman EM, eds. Vital Directions for Health and Health Care: An Initiative of the National Academy of Medicine. https://nam.edu/initiatives/vital-directions-for-health-and-health-care. Published 2017. Accessed August 19, 2018.
2. US Department of Health and Human Services, Agency for Healthcare Research and Quality. Defining the PCMH. https://pcmh.ahrq.gov/page/defining-pcmh. Accessed August 19, 2018.
3. US Department of Veterans Affairs, Patient Care Services. Patient aligned care team (PACT) https://www.patientcare.va.gov/primarycare/PACT.asp. Updated September 22, 2016. Accessed August 19, 2018.
4. Gilman SC, Chokshi DA, Bowen JL, Rugen KW, Cox M. Connecting the dots: health professions education and delivery system redesign. Acad Med. 2014;89(8):1113-1116.
5. Josiah Macy Jr. Foundation. Conference recommendations: transforming patient care: aligning interprofessional education with clinical practice redesign. http://macyfoundation.org/docs/macy_pubs/TransformingPatientCare_ConferenceRec.pdf. Published January 2013. Accessed August 19, 2018.
6. Accreditation Council for Graduate Medical Education. Clinical learning environment review. https://www.acgme.org/What-We-Do/Initiatives/Clinical-Learning-Environment-Review-CLER. Accessed August 19, 2018.
7. Cox M, Cuff P, Brandt B, Reeves S, Zierler B. Measuring the impact of interprofessional education on collaborative practice and patient outcomes. J Interprof Care. 2016;30(1):1-3.
8. National Collaborative for Improving the Clinical Learning Environment. Envisioning the optimal interprofessional clinical learning environment: initial findings from an October 2017 NCICLE symposium. https://storage.googleapis.com/wzukusers/user-27661272documents/5a5e3933a1c1cKVwrfGy/NCICLE%2IP-CLE%20Symposium%20Findings_011218%20update.pdf. Published January 12, 2018. Accessed August 19, 2018.
9. Institute of Medicine of the National Academies. Interprofessional Education for Collaboration: Learning How to Improve Health from Interprofessional Models Across the Continuum of Education to Practice: Workshop Summary. https://doi.org/10.17226/13486. Published 2013. Accessed August 22, 2018.
10. Harada ND, Traylor L, Rugen KW, et al. Interprofessional transformation of clinical education: the first six years of the Veterans Affairs Centers of Excellence in Primary Care Education. J Interprof Care. 2018;20:1-9.
11. US Department of Veterans Affairs, Office of Academic Affiliations. 2017 statistics: health professions trainees. https://www.va.gov/OAA/docs/OAA_Statistics.pdf. Accessed August 19, 2018.
12. US Department of Veterans Affairs, Office of Academic Affiliations. VA centers of excellence in primary care education. https://www.va.gov/oaa/coepce. Updated July 24, 2018. Accessed August 19, 2018.
13. US Department of Veterans Affairs, Office of Academic Affiliations. Academic PACT. https://www.va.gov/oaa/apact. Updated April 3, 2018. Accessed August 19, 2018.
14. US Department of Veterans Affairs, Office of Academic Affiliations. VA academic PACT: a blueprint for primary care redesign in academic practice settings. https://www.va.gov/oaa/docs/VA_Academic_PACT_blueprint.pdf. Published July 29, 2013. Accessed August 19, 2018.
15. US Department of Veterans Affairs, Office of Academic Affiliations. Centers of Excellence in Primary Care Education. Compendium of five case studies: lessons for interprofessional teamwork in education and workplace learning environments 2011-2016. https://www.va.gov/OAA/docs/VACaseStudiesCoEPCE.pdf. Published 2017. Accessed August 19, 2018.
16. US Department of Veterans Affairs, Quality Enhancement Research Initiative. Action-oriented evaluation of interprofessional learning efforts in the CoEPCE and iA-PACT environments. https://www.queri.research.va.gov/about/factsheets/InterProfessional-PEI.pdf. Published June 2018. Accessed August 19, 2018.
A broad consensus exists that US health care is now becoming more complex than at any other time in prior decades, potentially contributing to less than optimal outcomes, inadequate or unnecessary care, dissatisfied users, burned-out providers, and excessive costs.1 To reduce health system dysfunction, experts have looked to primary care to improve care continuity, coordination, and quality. The patient-centered medical home was designed to create environments where patients can access skilled professionals for both immediate and long-term needs across the health care spectrum, including nursing, pharmacy, social work, mental health, care coordinators, and educators.2
In 2010, the VHA of the Department of Veterans Affairs (VA) introduced a patient-centered model of primary care known as the patient-aligned care team (PACT). Each enrolled veteran is assigned to a PACT that is staffed by the enrollee’s personal provider, clinical staff, and appropriate professionals who work together to respond to patients in the context of their unique needs. In addition to the primary care provider (physician, physician assistant, or nurse practitioner), a nurse care manager, licensed vocational nurse or medical assistant, and an administrative professional, each PACT team is staffed by pharmacists, social workers, and mental health specialists. An especially important, and possibly unique, aspect of the VA PACT model is the integration of traditional primary care services with mental health access and care. This clinical interprofessional collaboration requires new educational strategies to effectively train a workforce qualified to work in, lead, and improve these settings.3
Although clinical environments are undergoing rapid change, curriculum for the health professions trainees has not adapted as quickly, even though it has been widely recognized that both should evolve concurrently.4 Curriculum emphasizing interprofessional practice, in particular, has been insufficiently implemented in educational settings.5 Static clinical learning environments pose a risk to future systems that will flounder without prepared professionals.6 Professional organizations, consensus groups, and medical education expert recommendations to implement interprofessional training environments have been met with relatively slow uptake in part because the challenges to implementation of scalable platforms for interprofessional clinical education are not trivial.7-9
This issue of Federal Practitioner introduces the first of 5 case studies that describe the implementation of instructional strategies designed and implemented by faculty, staff, and trainees. Each case embodies a unique approach to curriculum design and implementation that illustrates the collaborative innovation required to engage trainees with patients, with one another from differing professions, and with their faculty. The required flattening of the traditional hierarchy of staff in medical settings necessitates modification of clinical faculty and trainees skills. Didactic sessions are limited, and the focus is on experiential teaching and learning.10
As will be seen through the lenses of the cases presented in this series, the investments (including the time line to shift attitudes and change culture) required to achieve measurable outcomes are substantial. These investments not only are monetary, but also include addressing change management, conflict resolution, enhancement of communication skills, employee engagement, and leadership development.
The VA supports a comprehensive health system distributed throughout the nation with more than 1,000 points of care and more than 150 medical centers. Less recognized is that VA is the largest clinical learning platform in the US: More than 120,000 students and trainees enrolled in more than 40 different health professions and disciplines participate in VA clinical training programs annually.11 The VA has incorporated multiple innovative care designs, such as PACTs, along with educational and clinical leadership to create experiential workplace learning environments where structure, processes, and outcomes can be observed, adjusted, measured, and potentially duplicated.
This approach was key for the initial 5 of the current 7 Centers of Excellence in Primary Care Education (CoEPCEs) launched by VA in 2011, and from which the 5 cases in this series have evolved.12 The CoEPCE was developed as a demonstration project to show how to develop the interprofessional primary care curriculum for health professions that the PACT model requires. The CoEPCE, having trained more than 1,000 learners to date, has informed the PACT model to distinguish between PACTs whose mission is to provide clinical care from those that have the additional role of educating health professions trainees. The PACTS with this additional obligation are called interprofessional academic PACTs (iAPACTs). The iAPACTs incorporate features to accommodate clinical teaching and learning, including logistic challenges of scheduling, additional space requirements, faculty assignments, and affiliations with the academic institutions that sponsor the training programs.
Foundational concepts of the CoEPCE include those inherent in primary care, plus interprofessional practice where trainees of multiple professions are integrated into the care model to create a transformed workplace learning environment.13,14 Curricular domains of shared decision making among team members and their veteran patients, interprofessional collaboration, sustained relationships, and performance improvement are all required elements integral to the design and implementation of all CoEPCEs.13 This purposeful design provides clinical and educational infrastructure for interprofessional practice that simultaneously and seamlessly integrates both priorities of transforming clinical care and education.
The vision is to create the clinical learning environments necessary to produce the high-functioning individuals and teams needed to assure beneficial patient care outcomes as well as professional and personal satisfaction within the care team. The goal is to improve the PACT model of care in VA as a vehicle to enhance primary care services, to support changes in policy and practice that improve veterans’ care, safety, experience, health and well-being, and prepare a highly skilled future workforce for VA and for the nation as a whole.15
As all the cases in this series illustrate, the trainees are deeply embedded into clinical care and—very importantly—processes of patient care provision in consideration of all the patients care needs, using a holistic care model. As integrated team members, trainees from multiple professions learn with, from, and about one another as professionals and, as importantly, learn to appreciate the array of skills each brings to patient care, thus transforming their personal as well as professional learning experience. A highly relevant finding is that faculty and leadership—along with the trainees—have also learned, benefited, and transformed their thinking and attitudes, contributing to a cultural shift that is less hierarchical and more inclusive of all team members. A recently released external evaluation of the CoEPCE in their iAPACT environments indicates promising patterns of clinical outcomes with indications of improved staff satisfaction and less burnout. Better understanding of these innovations across and beyond the evaluated sites will be the topic of subsequent inquiries.16
These case studies demonstrate how education can be designed to advance the quality of care and improve the clinical teaching and learning environment, and educational outcomes. These cases are not intended to be recipes but rather exemplify the ingredients required to provide enough information and background to illustrate the transformational process. Superficially the cases may seem simple, but deeper examination reveals the complexity of confronting the challenges of day-to-day clinical work and redesigning both clinical and educational parameters.
These are real cases about real people working hard to revise a fragmented system and build a better future. The true purpose of these case studies is to inspire others to pursue educational modernization and excellence. In fact, there is no other satisfactory choice.
A broad consensus exists that US health care is now becoming more complex than at any other time in prior decades, potentially contributing to less than optimal outcomes, inadequate or unnecessary care, dissatisfied users, burned-out providers, and excessive costs.1 To reduce health system dysfunction, experts have looked to primary care to improve care continuity, coordination, and quality. The patient-centered medical home was designed to create environments where patients can access skilled professionals for both immediate and long-term needs across the health care spectrum, including nursing, pharmacy, social work, mental health, care coordinators, and educators.2
In 2010, the VHA of the Department of Veterans Affairs (VA) introduced a patient-centered model of primary care known as the patient-aligned care team (PACT). Each enrolled veteran is assigned to a PACT that is staffed by the enrollee’s personal provider, clinical staff, and appropriate professionals who work together to respond to patients in the context of their unique needs. In addition to the primary care provider (physician, physician assistant, or nurse practitioner), a nurse care manager, licensed vocational nurse or medical assistant, and an administrative professional, each PACT team is staffed by pharmacists, social workers, and mental health specialists. An especially important, and possibly unique, aspect of the VA PACT model is the integration of traditional primary care services with mental health access and care. This clinical interprofessional collaboration requires new educational strategies to effectively train a workforce qualified to work in, lead, and improve these settings.3
Although clinical environments are undergoing rapid change, curriculum for the health professions trainees has not adapted as quickly, even though it has been widely recognized that both should evolve concurrently.4 Curriculum emphasizing interprofessional practice, in particular, has been insufficiently implemented in educational settings.5 Static clinical learning environments pose a risk to future systems that will flounder without prepared professionals.6 Professional organizations, consensus groups, and medical education expert recommendations to implement interprofessional training environments have been met with relatively slow uptake in part because the challenges to implementation of scalable platforms for interprofessional clinical education are not trivial.7-9
This issue of Federal Practitioner introduces the first of 5 case studies that describe the implementation of instructional strategies designed and implemented by faculty, staff, and trainees. Each case embodies a unique approach to curriculum design and implementation that illustrates the collaborative innovation required to engage trainees with patients, with one another from differing professions, and with their faculty. The required flattening of the traditional hierarchy of staff in medical settings necessitates modification of clinical faculty and trainees skills. Didactic sessions are limited, and the focus is on experiential teaching and learning.10
As will be seen through the lenses of the cases presented in this series, the investments (including the time line to shift attitudes and change culture) required to achieve measurable outcomes are substantial. These investments not only are monetary, but also include addressing change management, conflict resolution, enhancement of communication skills, employee engagement, and leadership development.
The VA supports a comprehensive health system distributed throughout the nation with more than 1,000 points of care and more than 150 medical centers. Less recognized is that VA is the largest clinical learning platform in the US: More than 120,000 students and trainees enrolled in more than 40 different health professions and disciplines participate in VA clinical training programs annually.11 The VA has incorporated multiple innovative care designs, such as PACTs, along with educational and clinical leadership to create experiential workplace learning environments where structure, processes, and outcomes can be observed, adjusted, measured, and potentially duplicated.
This approach was key for the initial 5 of the current 7 Centers of Excellence in Primary Care Education (CoEPCEs) launched by VA in 2011, and from which the 5 cases in this series have evolved.12 The CoEPCE was developed as a demonstration project to show how to develop the interprofessional primary care curriculum for health professions that the PACT model requires. The CoEPCE, having trained more than 1,000 learners to date, has informed the PACT model to distinguish between PACTs whose mission is to provide clinical care from those that have the additional role of educating health professions trainees. The PACTS with this additional obligation are called interprofessional academic PACTs (iAPACTs). The iAPACTs incorporate features to accommodate clinical teaching and learning, including logistic challenges of scheduling, additional space requirements, faculty assignments, and affiliations with the academic institutions that sponsor the training programs.
Foundational concepts of the CoEPCE include those inherent in primary care, plus interprofessional practice where trainees of multiple professions are integrated into the care model to create a transformed workplace learning environment.13,14 Curricular domains of shared decision making among team members and their veteran patients, interprofessional collaboration, sustained relationships, and performance improvement are all required elements integral to the design and implementation of all CoEPCEs.13 This purposeful design provides clinical and educational infrastructure for interprofessional practice that simultaneously and seamlessly integrates both priorities of transforming clinical care and education.
The vision is to create the clinical learning environments necessary to produce the high-functioning individuals and teams needed to assure beneficial patient care outcomes as well as professional and personal satisfaction within the care team. The goal is to improve the PACT model of care in VA as a vehicle to enhance primary care services, to support changes in policy and practice that improve veterans’ care, safety, experience, health and well-being, and prepare a highly skilled future workforce for VA and for the nation as a whole.15
As all the cases in this series illustrate, the trainees are deeply embedded into clinical care and—very importantly—processes of patient care provision in consideration of all the patients care needs, using a holistic care model. As integrated team members, trainees from multiple professions learn with, from, and about one another as professionals and, as importantly, learn to appreciate the array of skills each brings to patient care, thus transforming their personal as well as professional learning experience. A highly relevant finding is that faculty and leadership—along with the trainees—have also learned, benefited, and transformed their thinking and attitudes, contributing to a cultural shift that is less hierarchical and more inclusive of all team members. A recently released external evaluation of the CoEPCE in their iAPACT environments indicates promising patterns of clinical outcomes with indications of improved staff satisfaction and less burnout. Better understanding of these innovations across and beyond the evaluated sites will be the topic of subsequent inquiries.16
These case studies demonstrate how education can be designed to advance the quality of care and improve the clinical teaching and learning environment, and educational outcomes. These cases are not intended to be recipes but rather exemplify the ingredients required to provide enough information and background to illustrate the transformational process. Superficially the cases may seem simple, but deeper examination reveals the complexity of confronting the challenges of day-to-day clinical work and redesigning both clinical and educational parameters.
These are real cases about real people working hard to revise a fragmented system and build a better future. The true purpose of these case studies is to inspire others to pursue educational modernization and excellence. In fact, there is no other satisfactory choice.
1. Dzau VJ, McClellan MB, McGinnis M, Finkelman EM, eds. Vital Directions for Health and Health Care: An Initiative of the National Academy of Medicine. https://nam.edu/initiatives/vital-directions-for-health-and-health-care. Published 2017. Accessed August 19, 2018.
2. US Department of Health and Human Services, Agency for Healthcare Research and Quality. Defining the PCMH. https://pcmh.ahrq.gov/page/defining-pcmh. Accessed August 19, 2018.
3. US Department of Veterans Affairs, Patient Care Services. Patient aligned care team (PACT) https://www.patientcare.va.gov/primarycare/PACT.asp. Updated September 22, 2016. Accessed August 19, 2018.
4. Gilman SC, Chokshi DA, Bowen JL, Rugen KW, Cox M. Connecting the dots: health professions education and delivery system redesign. Acad Med. 2014;89(8):1113-1116.
5. Josiah Macy Jr. Foundation. Conference recommendations: transforming patient care: aligning interprofessional education with clinical practice redesign. http://macyfoundation.org/docs/macy_pubs/TransformingPatientCare_ConferenceRec.pdf. Published January 2013. Accessed August 19, 2018.
6. Accreditation Council for Graduate Medical Education. Clinical learning environment review. https://www.acgme.org/What-We-Do/Initiatives/Clinical-Learning-Environment-Review-CLER. Accessed August 19, 2018.
7. Cox M, Cuff P, Brandt B, Reeves S, Zierler B. Measuring the impact of interprofessional education on collaborative practice and patient outcomes. J Interprof Care. 2016;30(1):1-3.
8. National Collaborative for Improving the Clinical Learning Environment. Envisioning the optimal interprofessional clinical learning environment: initial findings from an October 2017 NCICLE symposium. https://storage.googleapis.com/wzukusers/user-27661272documents/5a5e3933a1c1cKVwrfGy/NCICLE%2IP-CLE%20Symposium%20Findings_011218%20update.pdf. Published January 12, 2018. Accessed August 19, 2018.
9. Institute of Medicine of the National Academies. Interprofessional Education for Collaboration: Learning How to Improve Health from Interprofessional Models Across the Continuum of Education to Practice: Workshop Summary. https://doi.org/10.17226/13486. Published 2013. Accessed August 22, 2018.
10. Harada ND, Traylor L, Rugen KW, et al. Interprofessional transformation of clinical education: the first six years of the Veterans Affairs Centers of Excellence in Primary Care Education. J Interprof Care. 2018;20:1-9.
11. US Department of Veterans Affairs, Office of Academic Affiliations. 2017 statistics: health professions trainees. https://www.va.gov/OAA/docs/OAA_Statistics.pdf. Accessed August 19, 2018.
12. US Department of Veterans Affairs, Office of Academic Affiliations. VA centers of excellence in primary care education. https://www.va.gov/oaa/coepce. Updated July 24, 2018. Accessed August 19, 2018.
13. US Department of Veterans Affairs, Office of Academic Affiliations. Academic PACT. https://www.va.gov/oaa/apact. Updated April 3, 2018. Accessed August 19, 2018.
14. US Department of Veterans Affairs, Office of Academic Affiliations. VA academic PACT: a blueprint for primary care redesign in academic practice settings. https://www.va.gov/oaa/docs/VA_Academic_PACT_blueprint.pdf. Published July 29, 2013. Accessed August 19, 2018.
15. US Department of Veterans Affairs, Office of Academic Affiliations. Centers of Excellence in Primary Care Education. Compendium of five case studies: lessons for interprofessional teamwork in education and workplace learning environments 2011-2016. https://www.va.gov/OAA/docs/VACaseStudiesCoEPCE.pdf. Published 2017. Accessed August 19, 2018.
16. US Department of Veterans Affairs, Quality Enhancement Research Initiative. Action-oriented evaluation of interprofessional learning efforts in the CoEPCE and iA-PACT environments. https://www.queri.research.va.gov/about/factsheets/InterProfessional-PEI.pdf. Published June 2018. Accessed August 19, 2018.
1. Dzau VJ, McClellan MB, McGinnis M, Finkelman EM, eds. Vital Directions for Health and Health Care: An Initiative of the National Academy of Medicine. https://nam.edu/initiatives/vital-directions-for-health-and-health-care. Published 2017. Accessed August 19, 2018.
2. US Department of Health and Human Services, Agency for Healthcare Research and Quality. Defining the PCMH. https://pcmh.ahrq.gov/page/defining-pcmh. Accessed August 19, 2018.
3. US Department of Veterans Affairs, Patient Care Services. Patient aligned care team (PACT) https://www.patientcare.va.gov/primarycare/PACT.asp. Updated September 22, 2016. Accessed August 19, 2018.
4. Gilman SC, Chokshi DA, Bowen JL, Rugen KW, Cox M. Connecting the dots: health professions education and delivery system redesign. Acad Med. 2014;89(8):1113-1116.
5. Josiah Macy Jr. Foundation. Conference recommendations: transforming patient care: aligning interprofessional education with clinical practice redesign. http://macyfoundation.org/docs/macy_pubs/TransformingPatientCare_ConferenceRec.pdf. Published January 2013. Accessed August 19, 2018.
6. Accreditation Council for Graduate Medical Education. Clinical learning environment review. https://www.acgme.org/What-We-Do/Initiatives/Clinical-Learning-Environment-Review-CLER. Accessed August 19, 2018.
7. Cox M, Cuff P, Brandt B, Reeves S, Zierler B. Measuring the impact of interprofessional education on collaborative practice and patient outcomes. J Interprof Care. 2016;30(1):1-3.
8. National Collaborative for Improving the Clinical Learning Environment. Envisioning the optimal interprofessional clinical learning environment: initial findings from an October 2017 NCICLE symposium. https://storage.googleapis.com/wzukusers/user-27661272documents/5a5e3933a1c1cKVwrfGy/NCICLE%2IP-CLE%20Symposium%20Findings_011218%20update.pdf. Published January 12, 2018. Accessed August 19, 2018.
9. Institute of Medicine of the National Academies. Interprofessional Education for Collaboration: Learning How to Improve Health from Interprofessional Models Across the Continuum of Education to Practice: Workshop Summary. https://doi.org/10.17226/13486. Published 2013. Accessed August 22, 2018.
10. Harada ND, Traylor L, Rugen KW, et al. Interprofessional transformation of clinical education: the first six years of the Veterans Affairs Centers of Excellence in Primary Care Education. J Interprof Care. 2018;20:1-9.
11. US Department of Veterans Affairs, Office of Academic Affiliations. 2017 statistics: health professions trainees. https://www.va.gov/OAA/docs/OAA_Statistics.pdf. Accessed August 19, 2018.
12. US Department of Veterans Affairs, Office of Academic Affiliations. VA centers of excellence in primary care education. https://www.va.gov/oaa/coepce. Updated July 24, 2018. Accessed August 19, 2018.
13. US Department of Veterans Affairs, Office of Academic Affiliations. Academic PACT. https://www.va.gov/oaa/apact. Updated April 3, 2018. Accessed August 19, 2018.
14. US Department of Veterans Affairs, Office of Academic Affiliations. VA academic PACT: a blueprint for primary care redesign in academic practice settings. https://www.va.gov/oaa/docs/VA_Academic_PACT_blueprint.pdf. Published July 29, 2013. Accessed August 19, 2018.
15. US Department of Veterans Affairs, Office of Academic Affiliations. Centers of Excellence in Primary Care Education. Compendium of five case studies: lessons for interprofessional teamwork in education and workplace learning environments 2011-2016. https://www.va.gov/OAA/docs/VACaseStudiesCoEPCE.pdf. Published 2017. Accessed August 19, 2018.
16. US Department of Veterans Affairs, Quality Enhancement Research Initiative. Action-oriented evaluation of interprofessional learning efforts in the CoEPCE and iA-PACT environments. https://www.queri.research.va.gov/about/factsheets/InterProfessional-PEI.pdf. Published June 2018. Accessed August 19, 2018.
Prophylactic haloperidol does not improve survival in critically ill patients
Clinical question: Does prophylactic use of haloperidol in critically ill patients at high risk of delirium improve survival at 28 days?
Background: Delirium occurs frequently in critically ill patients and can lead to increased ICU length of stay, hospital length of stay, duration of mechanical ventilation, and mortality. Prior research into the use of prophylactic antipsychotic administration has yielded inconsistent results.
Study design: Double-blind, randomized, controlled trial.
Setting: 21 ICUs in the Netherlands, from July 2013 to March 2017.
Synopsis: A total of 1,789 critically ill adults with an anticipated ICU stay of at least 2 days were randomized to receive 1 mg of haloperidol, 2 mg of haloperidol, or a placebo three times daily. All study sites used “best practice” delirium prevention (for example, early mobilization, noise reduction, protocols aiming to prevent oversedation). The primary outcome was defined as the number of days patients survived in the 28 days following inclusion, and secondary outcome measures included number of days survived in 90 days, delirium incidence, number of delirium-free and coma-free days, duration of mechanical ventilation, and length of ICU and hospital stay. The 1-mg haloperidol group was stopped early because of futility. There was no significant difference between the 2-mg haloperidol group and the placebo group for the primary outcome (P = .93), or any of the secondary outcomes.Bottom line: In a population of critically ill patients at high risk of delirium, prophylactic haloperidol did not significantly improve 28-day survival, nor did it significantly reduce the incidence of delirium or length of stay.
Citation: van den Boogaard M et al. Effect of haloperidol on survival among critically ill adults with a high risk of delirium: The REDUCE randomized clinical trial. JAMA. 2018 Feb 20;319(7):680-90.
Dr. Winters is a hospitalist at Beth Israel Deaconess Medical Center, and instructor in medicine, Harvard Medical School, Boston.
Clinical question: Does prophylactic use of haloperidol in critically ill patients at high risk of delirium improve survival at 28 days?
Background: Delirium occurs frequently in critically ill patients and can lead to increased ICU length of stay, hospital length of stay, duration of mechanical ventilation, and mortality. Prior research into the use of prophylactic antipsychotic administration has yielded inconsistent results.
Study design: Double-blind, randomized, controlled trial.
Setting: 21 ICUs in the Netherlands, from July 2013 to March 2017.
Synopsis: A total of 1,789 critically ill adults with an anticipated ICU stay of at least 2 days were randomized to receive 1 mg of haloperidol, 2 mg of haloperidol, or a placebo three times daily. All study sites used “best practice” delirium prevention (for example, early mobilization, noise reduction, protocols aiming to prevent oversedation). The primary outcome was defined as the number of days patients survived in the 28 days following inclusion, and secondary outcome measures included number of days survived in 90 days, delirium incidence, number of delirium-free and coma-free days, duration of mechanical ventilation, and length of ICU and hospital stay. The 1-mg haloperidol group was stopped early because of futility. There was no significant difference between the 2-mg haloperidol group and the placebo group for the primary outcome (P = .93), or any of the secondary outcomes.Bottom line: In a population of critically ill patients at high risk of delirium, prophylactic haloperidol did not significantly improve 28-day survival, nor did it significantly reduce the incidence of delirium or length of stay.
Citation: van den Boogaard M et al. Effect of haloperidol on survival among critically ill adults with a high risk of delirium: The REDUCE randomized clinical trial. JAMA. 2018 Feb 20;319(7):680-90.
Dr. Winters is a hospitalist at Beth Israel Deaconess Medical Center, and instructor in medicine, Harvard Medical School, Boston.
Clinical question: Does prophylactic use of haloperidol in critically ill patients at high risk of delirium improve survival at 28 days?
Background: Delirium occurs frequently in critically ill patients and can lead to increased ICU length of stay, hospital length of stay, duration of mechanical ventilation, and mortality. Prior research into the use of prophylactic antipsychotic administration has yielded inconsistent results.
Study design: Double-blind, randomized, controlled trial.
Setting: 21 ICUs in the Netherlands, from July 2013 to March 2017.
Synopsis: A total of 1,789 critically ill adults with an anticipated ICU stay of at least 2 days were randomized to receive 1 mg of haloperidol, 2 mg of haloperidol, or a placebo three times daily. All study sites used “best practice” delirium prevention (for example, early mobilization, noise reduction, protocols aiming to prevent oversedation). The primary outcome was defined as the number of days patients survived in the 28 days following inclusion, and secondary outcome measures included number of days survived in 90 days, delirium incidence, number of delirium-free and coma-free days, duration of mechanical ventilation, and length of ICU and hospital stay. The 1-mg haloperidol group was stopped early because of futility. There was no significant difference between the 2-mg haloperidol group and the placebo group for the primary outcome (P = .93), or any of the secondary outcomes.Bottom line: In a population of critically ill patients at high risk of delirium, prophylactic haloperidol did not significantly improve 28-day survival, nor did it significantly reduce the incidence of delirium or length of stay.
Citation: van den Boogaard M et al. Effect of haloperidol on survival among critically ill adults with a high risk of delirium: The REDUCE randomized clinical trial. JAMA. 2018 Feb 20;319(7):680-90.
Dr. Winters is a hospitalist at Beth Israel Deaconess Medical Center, and instructor in medicine, Harvard Medical School, Boston.
Depression Screening and Treatment: A Missed Opportunity in Lung Cancer Care (FULL)
About Research in Context
In this article, the authors of recent scholarship have been asked to discuss the implications of their research on federal health care providers and specifically the veteran and active-duty service member patient populations. Because the article does not include new research and cannot be blinded, it has undergone an abbreviated peer review process. The original article can be found at Sullivan DR, Forsberg CW, Ganzini L, et al. Longitudinal changes in depression symptoms and survival among patients with lung cancer: a national cohort assessment. J Clin Oncol. 2016;34(33):3984-3991.
Although depression is common among patients with cancer, patients with lung cancer are at particularly high risk. The prevalence of major depressive disorder (MDD) among patients with cancer can be as high as 13%, whereas up to 44% of patients with lung cancer experience depression symptoms at some point following their cancer diagnosis.1-3 These estimates are consistently higher than those of other types of cancer, possibly related to the stigma of the disease and the associated morbidity and mortality that are its hallmarks.4-8 This potentially life-threatening cancer diagnosis often evokes psychological distress; however, additional stressors contribute to the development of depression, including the effects of chemotherapeutic agents, surgical procedures, radiotherapy, and the consequences of physical symptoms and paraneoplastic syndromes.
In addition to the crippling effects of comorbid depression on patients’ quality of life (QOL), severe and persistent depression among patients with cancer is associated with prolonged hospital stays, worse treatment adherence, physical distress and pain, and increased desire for hastened death.9-11 During treatment, depression can amplify physical symptoms and interfere with effective coping.12,13
Depression also is likely a significant factor for the risk of suicide, which is 4 times higher in patients with lung cancer than that of the general population.14 Most important, as our recent study demonstrated, depression that develops at cancer diagnosis or during cancer treatment may contribute to worse survival. This effect was strongest among patients with early stage disease, in other words, the patients who are most likely to achieve cure.3 This association with early stage disease also has been observed in a strictly veteran population from the northwest U.S.15
Another key finding of our study was the similar survival among patients who experienced a remission of their depression and those who were never depressed. This finding reinforces the importance of effective depression treatment, which has the potential to reduce depression-related mortality; however, depression treatment was not fully captured and could not be directly compared in our study. Unfortunately, comorbid depression often goes undiagnosed and untreated in cancer patients as they report unmet emotional needs and a desire for psychological support during and after completion of cancer treatment.16,17
Given the general lack of depression treatment that occurs in patients with cancer, the negative consequences of depression can be sustained well into survivorship—defined clinically as someone who is free of any sign of cancer for 5 years. Cancer survivors frequently report fatigue, mood disturbance, sleep disruption, pain, and cognitive limitations that significantly impact QOL and are associated with disability and increased health care use.18 These symptoms likely are intertwined with and contribute to the development and persistence of depression. The ramifications of untreated depression on long-term cancer survivor outcomes are not completely understood, as few high-quality studies of depression in cancer survivors exist. However, in a mixed group of patients with cancer, there was a 2-fold risk of mortality in survivors with depression symptoms when these patients were assessed from 1 to 10 years into survivorship.19 The impact of depression on cancer survivorship is an important aspect of cancer care that deserves significantly more attention from both a research and clinical perspective.
Special Considerations for Veterans
There is a higher prevalence of mental health diagnoses in veterans than that in the general population, and depressive disorders are the most common.20-22 According to the VA National Registry for Depression, 11% of veterans aged ≥ 65 years have a diagnosis of MDD, a rate more than twice that in the general population of a similar age.23 However, the actual rate of depression among veterans may be even higher, as studies suggest depression is underdiagnosed in the veteran population.24 In addition to depression, veterans experience other disabling psychological illnesses, such as posttraumatic stress disorder (PTSD) related to deployment and combat duty or combat-related injuries, such as traumatic brain injuries. The negative consequences of PTSD on cancer outcomes are largely unexplored, but PTSD can contribute to increased health care utilization and costs.25,26 A similar psychological construct, cancer-related posttraumatic stress (PTS), which develops as a result of a cancer diagnosis or treatment, is associated with missed medical appointments and procedures, which could impact survival.27
Depression Screening and Treatment
Given the negative consequences of comorbid mental illness, professional oncology societies have started developing guidelines regarding the assessments and care of patients with cancer who are experiencing symptoms of depression and/or anxiety.11,28,29 Among these, the American Society of Clinical Oncology (ASCO) has adapted the Pan-Canadian Practice Guideline on Screening, Assessment, and Care of Psychosocial Distress (Depression, Anxiety) in Adults With Cancer.28 Per ASCO, the target audience for these guidelines is health care providers (eg, medical, surgical, and radiation oncologists; psychiatrists; psychologists; primary care providers; nurses; and others involved in the delivery of care for adults with cancer) as well as patients with cancer and their family members and caregivers.28 These guidelines address the optimum screening, assessment, and psychosocial-supportive care interventions for adults with cancer who are identified as experiencing symptoms of depression. Among the most imperative recommendations are periodic assessments across the trajectory of cancer care, including after cure, as well as employing institutional and community resources for depression treatment.
In clinical practice in a VA setting, implementing these guidelines might involve various interventions. First, it is vital for providers to conduct depression screening during periodic health care encounters. Given the high prevalence of depression in patients with lung cancer, we suggest using the 9-item Patient Health Questionnaire (PHQ-9) as an initial screening tool.30 Unlike the abridged 2-item PHQ-2 commonly used in the VA, the PHQ-9 provides an assessment of the full range of depressive symptoms. An elevated PHQ-9 score (≥ 10) is consistent with a major depressive episode and should trigger next steps.30
Once clinically significant depression is identified, initiation of treatment should occur next. The VA is well suited to assist and support non-mental health clinicians—particularly primary care—in treatment initiation and monitoring. This model of partnership is frequently called collaborative care, or integrated care, and it is well positioned to help patients with lung cancer with concomitant depression. In the VA, this model of care is called primary care-mental healthintegration (PC-MHI). One PC-MHI resource is called TIDES (Translating Initiatives for Depression into Effective Solutions), and when a patient is referred, a mental health nurse care manager helps to track the patients’ antidepressant adherence and treatment response while reporting results to primary care clinicians, who are generally responsible for initiating and continuing the antidepressant prescription. For patients preferring nonpharmacologic approaches or for whom an antidepressant may be contraindicated, PC-MHI can provide other assistance. For example, psychologists working in PC-MHI are equipped to provide a brief course of cognitive behavioral therapy sessions, another first-line, evidence-based treatment for clinical depression.
Clinician follow-up to ensure patient adherence, response, and satisfaction, and to adjust treatment as needed is essential. Besides ongoing coordination with PC-MHI services, including mental health clinicians as part of multidisciplinary cancer clinics could offer substantial added value to patients’ comprehensive cancer care. Indeed, the initiation of multicomponent depression care has been shown to improve QOL and role functioning in patients with cancer.31 Besides the established benefits on QOL, patients with lung cancer who achieve depression symptom remission also may enjoy a significant survival benefit over patients whose depression symptoms remain untreated during lung cancer treatment as our study suggests.3
Conclusion
Depression is a common comorbid disease among patients with lung cancer with important negative implications for QOL and survival. When it occurs after a cancer diagnosis, depression is expected to impact all phases of a patient’s life through treatment and survivorshi —ultimately affecting long-term survival. Veterans may be at particularly high risk given the increased prevalence of mental illness, including depression and PTSD in this group compared with that of the general population. Early detection and prompt treatment can promote depression remission, prevent relapse, and reduce the eventual emotional and financial burden of the disease. This approach may ultimately diminish the prevalence and persistence of depression symptoms and decrease the associated negative effects of this disease on patients with lung cancer.
The importance of integrated systems of depression treatment for patients with cancer as part of comprehensive cancer care cannot be overstated. Development and implementation of these systems should be a priority of lung cancer clinicians and treatment centers. The integrated system within the VA is well positioned to be a leader in this area, and VA clinicians who care for patients with lung cancer are encouraged to take advantage of available mental health resources. Additional research is urgently needed to explore optimal implementation of depression screening and subsequent treatment delivery to improve cancer patient outcomes in VA and non-VA health care settings.
Overall, there is minimal evidence that depression treatment can improve lung cancer survival; however, the lack of high-quality studies is a considerable limitation. Given the significant impact of depression on survival among patients with lung cancer, additional funding and resources are urgently needed to combat this debilitating comorbid disease.
Acknowledgments
This project was supported in part by the National Cancer Institute of the National Institutes of Health under award K07CA190706 to Dr. Sullivan, a Career Development Award from the Veterans Health Administration Health Service Research and Development (CDA 14-428) to Dr. Teo and the HSR&D Center to Improve Veteran Involvement in Care (CIVIC) (CIN 13-404) at the VA Portland Health Care System.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The VA had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication. The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies.
Click here to read the digital edition.
1. Derogatis LR, Morrow GR, Fetting J, et al. The prevalence of psychiatric disorders among cancer patients. JAMA. 1983;249(6):751-757.
2. Walker J, Holm Hansen C, Martin P, et al. Prevalence of depression in adults with cancer: a systematic review. Ann Oncol. 2013;24(4):895-900.
3. Sullivan DR, Forsberg CW, Ganzini L, et al. Longitudinal changes in depression symptoms and survival among patients with lung cancer: a national cohort assessment. J Clin Oncol. 2016;34(33):3984-3991.
4. Linden W, Vodermaier A, Mackenzie R, Greig D. Anxiety and depression after cancer diagnosis: prevalence rates by cancer type, gender, and age. J Affect Disord. 2012;141(2-3):343-351.
5. Massie MJ. Prevalence of depression in patients with cancer. J Natl Cancer Inst Monogr. 2004;(32):57-71.
6. Brown Johnson CG, Brodsky JL, Cataldo JK. Lung cancer stigma, anxiety, depression, and quality of life. J Psychosoc Oncol. 2014;32(1):59-73.
7. Cataldo JK, Jahan TM, Pongquan VL. Lung cancer stigma, depression, and quality of life among ever and never smokers. Eur J Oncol Nurs. 2012;16(3):264-269.
8. Howlader N, Noone AM, Krapcho M, et al. SEER cancer statistics review, 1975-2010. https://seer.cancer.gov/archive/csr/1975_2010/. Revised February 21, 2014. Accessed July 12, 2017.
9. Li M, Boquiren V, Lo C, et al. Depression and anxiety in supportive oncology. In: Davis M, Feyer P, Ortner P, Zimmermann C, eds. Supportive Oncology. 1st ed. Philadelphia, PA: Elsevier; 2011:528-540.
10. Brown LF, Kroenke K, Theobald DE, Wu J, Tu W. The association of depression and anxiety with health-related quality of life in cancer patients with depression and/or pain. Psychooncology. 2010;19(7):734-741.
11. Lazenby M, Ercolano E, Grant M, Holland JC, Jacobsen PB, McCorkle R. Supporting Commission on Cancer-mandated psychosocial distress screening with implementation strategies. J Oncol Pract. 2015;11(3):e413-e420.
12. Mystakidou K, Tsilika E, Parpa E, Katsouda E, Galanos A, Vlahos L. Psychological distress of patients with advanced cancer: influence and contribution of pain severity and pain interference. Cancer Nurs. 2006;29(5):400-405.
13. Passik SD, Dugan W, McDonald MV, Rosenfeld B, Theobald DE, Edgerton S. Oncologists’ recognition of depression in their patients with cancer. J Clin Oncol. 1998;16(4):1594-1600.
14. Rahuma M, Kamel M, Nasar A, et al. Lung cancer patients have the highest malignancy-associated suicide rate in USA: a population based analysis. Am J Respir Crit Care Med. 2017;195:A6730.
15. Sullivan DR, Ganzini L, Duckart JP, et al. Treatment receipt and outcomes among lung cancer patients with depression. Clin Oncol (R Coll Radiol). 2014;26(1):25-31.
16. Merckaert I, Libert Y, Messin S, Milani M, Slachmuylder JL, Razavi D. Cancer patients’ desire for psychological support: prevalence and implications for screening patients psychological needs. Psychooncology. 2010;19(2):141-149.
17. Harrison JD, Young JM, Price MA, Butow PN, Solomon MJ. What are the unmet supportive care needs of people with cancer? A systematic review. Support Care Cancer. 2009;17(8):1117-1128.
18. Wu HS, Harden JK. Symptom burden and quality of life in survivorship: a review of the literature. Cancer Nurs. 2015;38(1):E29-E54.
19. Mols F, Husson O, Roukema JA, van de Poll-Franse LV. Depressive symptoms are a risk factor for all-cause mortality: results from a prospective population-based study among 3,080 cancer survivors from the PROFILES registry. J Cancer Surviv. 2013;7(3):484-492.
20. Hoge CW, Castro CA, Messer SC, McGurk D, Cotting DI, Koffman RL. Combat duty in Iraq and Afghanistan, mental health problems, and barriers to care. N Engl J Med. 2004;351(1):13-22.
21. Fortney JC, Curran GM, Hunt JB, et al. Prevalence of probable mental disorders and help-seeking behaviors among veteran and non-veteran community college students. Gen Hosp Psychiatry. 2016;38:99-104.
22. Pickett T, Rothman D, Crawford EF, Brancu M, Fairbank JA, Kudler HS. Mental health among military personnel and veterans. N C Med J. 2015;76(5):299-306.
23. U.S. Department of Veterans Affairs, Veterans Health Administration. One in ten older vets is depressed. https://www.va.gov/health/NewsFeatures/20110624a.asp. Updated April 17, 2015. Accessed July 12, 2017.
24. Fontana A, Rosenheck R. Treatment-seeking veterans of Iraq and Afghanistan: comparison with veterans of previous wars. J Nerv Ment Dis. 2008;196(7):513-521.
25. Kessler RC. Posttraumatic stress disorder: the burden to the individual and to society. J Clin Psychiatry. 2000;61(suppl 5):4-12; discussion, 13-14.
26. Kartha A, Brower V, Saitz R, Samet JH, Keane TM, Liebschutz J. The impact of trauma exposure and post-traumatic stress disorder on healthcare utilization among primary care patients. Med Care. 2008;46(4):388-393.
27. National Cancer Institute. Cancer-related post-traumatic stress (PDQ®)–Patient version. https://www.cancer.gov/about-cancer/coping/survivorship/new-normal/ptsd-pdq. Updated July 7, 2015. Accessed July 12, 2017.
28. Andersen BL, DeRubeis RJ, Berman BS, et al; American Society of Clinical Oncology. Screening, assessment, and care of anxiety and depressive symptoms in adults with cancer: an American Society of Clinical Oncology guideline adaptation. J Clin Oncol. 2014;32(15):1605-1619.
29. Howell D, Keller-Olaman S, Oliver TK, et al. A pan-Canadian practice guideline and algorithm: screening, assessment, and supportive care of adults with cancer-related fatigue. Curr Oncol. 2013;20(3):e233-e246.
30. Kroenke K, Wu J, Bair MJ, Krebs EE, Damush TM, Tu W. Reciprocal relationship between pain and depression: a 12-month longitudinal analysis in primary care. J Pain. 2011;12(9):964-973.
31. Walker J, Hansen CH, Martin P, et al; SMaRT (Symptom Management Research Trials) Oncology-3 Team. Integrated collaborative care for major depression comorbid with a poor prognosis cancer (SMaRT oncology-3): a multicentre randomised controlled trial in patients with lung cancer. Lancet Oncol. 2014;15(10):1168-1176.
About Research in Context
In this article, the authors of recent scholarship have been asked to discuss the implications of their research on federal health care providers and specifically the veteran and active-duty service member patient populations. Because the article does not include new research and cannot be blinded, it has undergone an abbreviated peer review process. The original article can be found at Sullivan DR, Forsberg CW, Ganzini L, et al. Longitudinal changes in depression symptoms and survival among patients with lung cancer: a national cohort assessment. J Clin Oncol. 2016;34(33):3984-3991.
Although depression is common among patients with cancer, patients with lung cancer are at particularly high risk. The prevalence of major depressive disorder (MDD) among patients with cancer can be as high as 13%, whereas up to 44% of patients with lung cancer experience depression symptoms at some point following their cancer diagnosis.1-3 These estimates are consistently higher than those of other types of cancer, possibly related to the stigma of the disease and the associated morbidity and mortality that are its hallmarks.4-8 This potentially life-threatening cancer diagnosis often evokes psychological distress; however, additional stressors contribute to the development of depression, including the effects of chemotherapeutic agents, surgical procedures, radiotherapy, and the consequences of physical symptoms and paraneoplastic syndromes.
In addition to the crippling effects of comorbid depression on patients’ quality of life (QOL), severe and persistent depression among patients with cancer is associated with prolonged hospital stays, worse treatment adherence, physical distress and pain, and increased desire for hastened death.9-11 During treatment, depression can amplify physical symptoms and interfere with effective coping.12,13
Depression also is likely a significant factor for the risk of suicide, which is 4 times higher in patients with lung cancer than that of the general population.14 Most important, as our recent study demonstrated, depression that develops at cancer diagnosis or during cancer treatment may contribute to worse survival. This effect was strongest among patients with early stage disease, in other words, the patients who are most likely to achieve cure.3 This association with early stage disease also has been observed in a strictly veteran population from the northwest U.S.15
Another key finding of our study was the similar survival among patients who experienced a remission of their depression and those who were never depressed. This finding reinforces the importance of effective depression treatment, which has the potential to reduce depression-related mortality; however, depression treatment was not fully captured and could not be directly compared in our study. Unfortunately, comorbid depression often goes undiagnosed and untreated in cancer patients as they report unmet emotional needs and a desire for psychological support during and after completion of cancer treatment.16,17
Given the general lack of depression treatment that occurs in patients with cancer, the negative consequences of depression can be sustained well into survivorship—defined clinically as someone who is free of any sign of cancer for 5 years. Cancer survivors frequently report fatigue, mood disturbance, sleep disruption, pain, and cognitive limitations that significantly impact QOL and are associated with disability and increased health care use.18 These symptoms likely are intertwined with and contribute to the development and persistence of depression. The ramifications of untreated depression on long-term cancer survivor outcomes are not completely understood, as few high-quality studies of depression in cancer survivors exist. However, in a mixed group of patients with cancer, there was a 2-fold risk of mortality in survivors with depression symptoms when these patients were assessed from 1 to 10 years into survivorship.19 The impact of depression on cancer survivorship is an important aspect of cancer care that deserves significantly more attention from both a research and clinical perspective.
Special Considerations for Veterans
There is a higher prevalence of mental health diagnoses in veterans than that in the general population, and depressive disorders are the most common.20-22 According to the VA National Registry for Depression, 11% of veterans aged ≥ 65 years have a diagnosis of MDD, a rate more than twice that in the general population of a similar age.23 However, the actual rate of depression among veterans may be even higher, as studies suggest depression is underdiagnosed in the veteran population.24 In addition to depression, veterans experience other disabling psychological illnesses, such as posttraumatic stress disorder (PTSD) related to deployment and combat duty or combat-related injuries, such as traumatic brain injuries. The negative consequences of PTSD on cancer outcomes are largely unexplored, but PTSD can contribute to increased health care utilization and costs.25,26 A similar psychological construct, cancer-related posttraumatic stress (PTS), which develops as a result of a cancer diagnosis or treatment, is associated with missed medical appointments and procedures, which could impact survival.27
Depression Screening and Treatment
Given the negative consequences of comorbid mental illness, professional oncology societies have started developing guidelines regarding the assessments and care of patients with cancer who are experiencing symptoms of depression and/or anxiety.11,28,29 Among these, the American Society of Clinical Oncology (ASCO) has adapted the Pan-Canadian Practice Guideline on Screening, Assessment, and Care of Psychosocial Distress (Depression, Anxiety) in Adults With Cancer.28 Per ASCO, the target audience for these guidelines is health care providers (eg, medical, surgical, and radiation oncologists; psychiatrists; psychologists; primary care providers; nurses; and others involved in the delivery of care for adults with cancer) as well as patients with cancer and their family members and caregivers.28 These guidelines address the optimum screening, assessment, and psychosocial-supportive care interventions for adults with cancer who are identified as experiencing symptoms of depression. Among the most imperative recommendations are periodic assessments across the trajectory of cancer care, including after cure, as well as employing institutional and community resources for depression treatment.
In clinical practice in a VA setting, implementing these guidelines might involve various interventions. First, it is vital for providers to conduct depression screening during periodic health care encounters. Given the high prevalence of depression in patients with lung cancer, we suggest using the 9-item Patient Health Questionnaire (PHQ-9) as an initial screening tool.30 Unlike the abridged 2-item PHQ-2 commonly used in the VA, the PHQ-9 provides an assessment of the full range of depressive symptoms. An elevated PHQ-9 score (≥ 10) is consistent with a major depressive episode and should trigger next steps.30
Once clinically significant depression is identified, initiation of treatment should occur next. The VA is well suited to assist and support non-mental health clinicians—particularly primary care—in treatment initiation and monitoring. This model of partnership is frequently called collaborative care, or integrated care, and it is well positioned to help patients with lung cancer with concomitant depression. In the VA, this model of care is called primary care-mental healthintegration (PC-MHI). One PC-MHI resource is called TIDES (Translating Initiatives for Depression into Effective Solutions), and when a patient is referred, a mental health nurse care manager helps to track the patients’ antidepressant adherence and treatment response while reporting results to primary care clinicians, who are generally responsible for initiating and continuing the antidepressant prescription. For patients preferring nonpharmacologic approaches or for whom an antidepressant may be contraindicated, PC-MHI can provide other assistance. For example, psychologists working in PC-MHI are equipped to provide a brief course of cognitive behavioral therapy sessions, another first-line, evidence-based treatment for clinical depression.
Clinician follow-up to ensure patient adherence, response, and satisfaction, and to adjust treatment as needed is essential. Besides ongoing coordination with PC-MHI services, including mental health clinicians as part of multidisciplinary cancer clinics could offer substantial added value to patients’ comprehensive cancer care. Indeed, the initiation of multicomponent depression care has been shown to improve QOL and role functioning in patients with cancer.31 Besides the established benefits on QOL, patients with lung cancer who achieve depression symptom remission also may enjoy a significant survival benefit over patients whose depression symptoms remain untreated during lung cancer treatment as our study suggests.3
Conclusion
Depression is a common comorbid disease among patients with lung cancer with important negative implications for QOL and survival. When it occurs after a cancer diagnosis, depression is expected to impact all phases of a patient’s life through treatment and survivorshi —ultimately affecting long-term survival. Veterans may be at particularly high risk given the increased prevalence of mental illness, including depression and PTSD in this group compared with that of the general population. Early detection and prompt treatment can promote depression remission, prevent relapse, and reduce the eventual emotional and financial burden of the disease. This approach may ultimately diminish the prevalence and persistence of depression symptoms and decrease the associated negative effects of this disease on patients with lung cancer.
The importance of integrated systems of depression treatment for patients with cancer as part of comprehensive cancer care cannot be overstated. Development and implementation of these systems should be a priority of lung cancer clinicians and treatment centers. The integrated system within the VA is well positioned to be a leader in this area, and VA clinicians who care for patients with lung cancer are encouraged to take advantage of available mental health resources. Additional research is urgently needed to explore optimal implementation of depression screening and subsequent treatment delivery to improve cancer patient outcomes in VA and non-VA health care settings.
Overall, there is minimal evidence that depression treatment can improve lung cancer survival; however, the lack of high-quality studies is a considerable limitation. Given the significant impact of depression on survival among patients with lung cancer, additional funding and resources are urgently needed to combat this debilitating comorbid disease.
Acknowledgments
This project was supported in part by the National Cancer Institute of the National Institutes of Health under award K07CA190706 to Dr. Sullivan, a Career Development Award from the Veterans Health Administration Health Service Research and Development (CDA 14-428) to Dr. Teo and the HSR&D Center to Improve Veteran Involvement in Care (CIVIC) (CIN 13-404) at the VA Portland Health Care System.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The VA had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication. The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies.
Click here to read the digital edition.
About Research in Context
In this article, the authors of recent scholarship have been asked to discuss the implications of their research on federal health care providers and specifically the veteran and active-duty service member patient populations. Because the article does not include new research and cannot be blinded, it has undergone an abbreviated peer review process. The original article can be found at Sullivan DR, Forsberg CW, Ganzini L, et al. Longitudinal changes in depression symptoms and survival among patients with lung cancer: a national cohort assessment. J Clin Oncol. 2016;34(33):3984-3991.
Although depression is common among patients with cancer, patients with lung cancer are at particularly high risk. The prevalence of major depressive disorder (MDD) among patients with cancer can be as high as 13%, whereas up to 44% of patients with lung cancer experience depression symptoms at some point following their cancer diagnosis.1-3 These estimates are consistently higher than those of other types of cancer, possibly related to the stigma of the disease and the associated morbidity and mortality that are its hallmarks.4-8 This potentially life-threatening cancer diagnosis often evokes psychological distress; however, additional stressors contribute to the development of depression, including the effects of chemotherapeutic agents, surgical procedures, radiotherapy, and the consequences of physical symptoms and paraneoplastic syndromes.
In addition to the crippling effects of comorbid depression on patients’ quality of life (QOL), severe and persistent depression among patients with cancer is associated with prolonged hospital stays, worse treatment adherence, physical distress and pain, and increased desire for hastened death.9-11 During treatment, depression can amplify physical symptoms and interfere with effective coping.12,13
Depression also is likely a significant factor for the risk of suicide, which is 4 times higher in patients with lung cancer than that of the general population.14 Most important, as our recent study demonstrated, depression that develops at cancer diagnosis or during cancer treatment may contribute to worse survival. This effect was strongest among patients with early stage disease, in other words, the patients who are most likely to achieve cure.3 This association with early stage disease also has been observed in a strictly veteran population from the northwest U.S.15
Another key finding of our study was the similar survival among patients who experienced a remission of their depression and those who were never depressed. This finding reinforces the importance of effective depression treatment, which has the potential to reduce depression-related mortality; however, depression treatment was not fully captured and could not be directly compared in our study. Unfortunately, comorbid depression often goes undiagnosed and untreated in cancer patients as they report unmet emotional needs and a desire for psychological support during and after completion of cancer treatment.16,17
Given the general lack of depression treatment that occurs in patients with cancer, the negative consequences of depression can be sustained well into survivorship—defined clinically as someone who is free of any sign of cancer for 5 years. Cancer survivors frequently report fatigue, mood disturbance, sleep disruption, pain, and cognitive limitations that significantly impact QOL and are associated with disability and increased health care use.18 These symptoms likely are intertwined with and contribute to the development and persistence of depression. The ramifications of untreated depression on long-term cancer survivor outcomes are not completely understood, as few high-quality studies of depression in cancer survivors exist. However, in a mixed group of patients with cancer, there was a 2-fold risk of mortality in survivors with depression symptoms when these patients were assessed from 1 to 10 years into survivorship.19 The impact of depression on cancer survivorship is an important aspect of cancer care that deserves significantly more attention from both a research and clinical perspective.
Special Considerations for Veterans
There is a higher prevalence of mental health diagnoses in veterans than that in the general population, and depressive disorders are the most common.20-22 According to the VA National Registry for Depression, 11% of veterans aged ≥ 65 years have a diagnosis of MDD, a rate more than twice that in the general population of a similar age.23 However, the actual rate of depression among veterans may be even higher, as studies suggest depression is underdiagnosed in the veteran population.24 In addition to depression, veterans experience other disabling psychological illnesses, such as posttraumatic stress disorder (PTSD) related to deployment and combat duty or combat-related injuries, such as traumatic brain injuries. The negative consequences of PTSD on cancer outcomes are largely unexplored, but PTSD can contribute to increased health care utilization and costs.25,26 A similar psychological construct, cancer-related posttraumatic stress (PTS), which develops as a result of a cancer diagnosis or treatment, is associated with missed medical appointments and procedures, which could impact survival.27
Depression Screening and Treatment
Given the negative consequences of comorbid mental illness, professional oncology societies have started developing guidelines regarding the assessments and care of patients with cancer who are experiencing symptoms of depression and/or anxiety.11,28,29 Among these, the American Society of Clinical Oncology (ASCO) has adapted the Pan-Canadian Practice Guideline on Screening, Assessment, and Care of Psychosocial Distress (Depression, Anxiety) in Adults With Cancer.28 Per ASCO, the target audience for these guidelines is health care providers (eg, medical, surgical, and radiation oncologists; psychiatrists; psychologists; primary care providers; nurses; and others involved in the delivery of care for adults with cancer) as well as patients with cancer and their family members and caregivers.28 These guidelines address the optimum screening, assessment, and psychosocial-supportive care interventions for adults with cancer who are identified as experiencing symptoms of depression. Among the most imperative recommendations are periodic assessments across the trajectory of cancer care, including after cure, as well as employing institutional and community resources for depression treatment.
In clinical practice in a VA setting, implementing these guidelines might involve various interventions. First, it is vital for providers to conduct depression screening during periodic health care encounters. Given the high prevalence of depression in patients with lung cancer, we suggest using the 9-item Patient Health Questionnaire (PHQ-9) as an initial screening tool.30 Unlike the abridged 2-item PHQ-2 commonly used in the VA, the PHQ-9 provides an assessment of the full range of depressive symptoms. An elevated PHQ-9 score (≥ 10) is consistent with a major depressive episode and should trigger next steps.30
Once clinically significant depression is identified, initiation of treatment should occur next. The VA is well suited to assist and support non-mental health clinicians—particularly primary care—in treatment initiation and monitoring. This model of partnership is frequently called collaborative care, or integrated care, and it is well positioned to help patients with lung cancer with concomitant depression. In the VA, this model of care is called primary care-mental healthintegration (PC-MHI). One PC-MHI resource is called TIDES (Translating Initiatives for Depression into Effective Solutions), and when a patient is referred, a mental health nurse care manager helps to track the patients’ antidepressant adherence and treatment response while reporting results to primary care clinicians, who are generally responsible for initiating and continuing the antidepressant prescription. For patients preferring nonpharmacologic approaches or for whom an antidepressant may be contraindicated, PC-MHI can provide other assistance. For example, psychologists working in PC-MHI are equipped to provide a brief course of cognitive behavioral therapy sessions, another first-line, evidence-based treatment for clinical depression.
Clinician follow-up to ensure patient adherence, response, and satisfaction, and to adjust treatment as needed is essential. Besides ongoing coordination with PC-MHI services, including mental health clinicians as part of multidisciplinary cancer clinics could offer substantial added value to patients’ comprehensive cancer care. Indeed, the initiation of multicomponent depression care has been shown to improve QOL and role functioning in patients with cancer.31 Besides the established benefits on QOL, patients with lung cancer who achieve depression symptom remission also may enjoy a significant survival benefit over patients whose depression symptoms remain untreated during lung cancer treatment as our study suggests.3
Conclusion
Depression is a common comorbid disease among patients with lung cancer with important negative implications for QOL and survival. When it occurs after a cancer diagnosis, depression is expected to impact all phases of a patient’s life through treatment and survivorshi —ultimately affecting long-term survival. Veterans may be at particularly high risk given the increased prevalence of mental illness, including depression and PTSD in this group compared with that of the general population. Early detection and prompt treatment can promote depression remission, prevent relapse, and reduce the eventual emotional and financial burden of the disease. This approach may ultimately diminish the prevalence and persistence of depression symptoms and decrease the associated negative effects of this disease on patients with lung cancer.
The importance of integrated systems of depression treatment for patients with cancer as part of comprehensive cancer care cannot be overstated. Development and implementation of these systems should be a priority of lung cancer clinicians and treatment centers. The integrated system within the VA is well positioned to be a leader in this area, and VA clinicians who care for patients with lung cancer are encouraged to take advantage of available mental health resources. Additional research is urgently needed to explore optimal implementation of depression screening and subsequent treatment delivery to improve cancer patient outcomes in VA and non-VA health care settings.
Overall, there is minimal evidence that depression treatment can improve lung cancer survival; however, the lack of high-quality studies is a considerable limitation. Given the significant impact of depression on survival among patients with lung cancer, additional funding and resources are urgently needed to combat this debilitating comorbid disease.
Acknowledgments
This project was supported in part by the National Cancer Institute of the National Institutes of Health under award K07CA190706 to Dr. Sullivan, a Career Development Award from the Veterans Health Administration Health Service Research and Development (CDA 14-428) to Dr. Teo and the HSR&D Center to Improve Veteran Involvement in Care (CIVIC) (CIN 13-404) at the VA Portland Health Care System.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The VA had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication. The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies.
Click here to read the digital edition.
1. Derogatis LR, Morrow GR, Fetting J, et al. The prevalence of psychiatric disorders among cancer patients. JAMA. 1983;249(6):751-757.
2. Walker J, Holm Hansen C, Martin P, et al. Prevalence of depression in adults with cancer: a systematic review. Ann Oncol. 2013;24(4):895-900.
3. Sullivan DR, Forsberg CW, Ganzini L, et al. Longitudinal changes in depression symptoms and survival among patients with lung cancer: a national cohort assessment. J Clin Oncol. 2016;34(33):3984-3991.
4. Linden W, Vodermaier A, Mackenzie R, Greig D. Anxiety and depression after cancer diagnosis: prevalence rates by cancer type, gender, and age. J Affect Disord. 2012;141(2-3):343-351.
5. Massie MJ. Prevalence of depression in patients with cancer. J Natl Cancer Inst Monogr. 2004;(32):57-71.
6. Brown Johnson CG, Brodsky JL, Cataldo JK. Lung cancer stigma, anxiety, depression, and quality of life. J Psychosoc Oncol. 2014;32(1):59-73.
7. Cataldo JK, Jahan TM, Pongquan VL. Lung cancer stigma, depression, and quality of life among ever and never smokers. Eur J Oncol Nurs. 2012;16(3):264-269.
8. Howlader N, Noone AM, Krapcho M, et al. SEER cancer statistics review, 1975-2010. https://seer.cancer.gov/archive/csr/1975_2010/. Revised February 21, 2014. Accessed July 12, 2017.
9. Li M, Boquiren V, Lo C, et al. Depression and anxiety in supportive oncology. In: Davis M, Feyer P, Ortner P, Zimmermann C, eds. Supportive Oncology. 1st ed. Philadelphia, PA: Elsevier; 2011:528-540.
10. Brown LF, Kroenke K, Theobald DE, Wu J, Tu W. The association of depression and anxiety with health-related quality of life in cancer patients with depression and/or pain. Psychooncology. 2010;19(7):734-741.
11. Lazenby M, Ercolano E, Grant M, Holland JC, Jacobsen PB, McCorkle R. Supporting Commission on Cancer-mandated psychosocial distress screening with implementation strategies. J Oncol Pract. 2015;11(3):e413-e420.
12. Mystakidou K, Tsilika E, Parpa E, Katsouda E, Galanos A, Vlahos L. Psychological distress of patients with advanced cancer: influence and contribution of pain severity and pain interference. Cancer Nurs. 2006;29(5):400-405.
13. Passik SD, Dugan W, McDonald MV, Rosenfeld B, Theobald DE, Edgerton S. Oncologists’ recognition of depression in their patients with cancer. J Clin Oncol. 1998;16(4):1594-1600.
14. Rahuma M, Kamel M, Nasar A, et al. Lung cancer patients have the highest malignancy-associated suicide rate in USA: a population based analysis. Am J Respir Crit Care Med. 2017;195:A6730.
15. Sullivan DR, Ganzini L, Duckart JP, et al. Treatment receipt and outcomes among lung cancer patients with depression. Clin Oncol (R Coll Radiol). 2014;26(1):25-31.
16. Merckaert I, Libert Y, Messin S, Milani M, Slachmuylder JL, Razavi D. Cancer patients’ desire for psychological support: prevalence and implications for screening patients psychological needs. Psychooncology. 2010;19(2):141-149.
17. Harrison JD, Young JM, Price MA, Butow PN, Solomon MJ. What are the unmet supportive care needs of people with cancer? A systematic review. Support Care Cancer. 2009;17(8):1117-1128.
18. Wu HS, Harden JK. Symptom burden and quality of life in survivorship: a review of the literature. Cancer Nurs. 2015;38(1):E29-E54.
19. Mols F, Husson O, Roukema JA, van de Poll-Franse LV. Depressive symptoms are a risk factor for all-cause mortality: results from a prospective population-based study among 3,080 cancer survivors from the PROFILES registry. J Cancer Surviv. 2013;7(3):484-492.
20. Hoge CW, Castro CA, Messer SC, McGurk D, Cotting DI, Koffman RL. Combat duty in Iraq and Afghanistan, mental health problems, and barriers to care. N Engl J Med. 2004;351(1):13-22.
21. Fortney JC, Curran GM, Hunt JB, et al. Prevalence of probable mental disorders and help-seeking behaviors among veteran and non-veteran community college students. Gen Hosp Psychiatry. 2016;38:99-104.
22. Pickett T, Rothman D, Crawford EF, Brancu M, Fairbank JA, Kudler HS. Mental health among military personnel and veterans. N C Med J. 2015;76(5):299-306.
23. U.S. Department of Veterans Affairs, Veterans Health Administration. One in ten older vets is depressed. https://www.va.gov/health/NewsFeatures/20110624a.asp. Updated April 17, 2015. Accessed July 12, 2017.
24. Fontana A, Rosenheck R. Treatment-seeking veterans of Iraq and Afghanistan: comparison with veterans of previous wars. J Nerv Ment Dis. 2008;196(7):513-521.
25. Kessler RC. Posttraumatic stress disorder: the burden to the individual and to society. J Clin Psychiatry. 2000;61(suppl 5):4-12; discussion, 13-14.
26. Kartha A, Brower V, Saitz R, Samet JH, Keane TM, Liebschutz J. The impact of trauma exposure and post-traumatic stress disorder on healthcare utilization among primary care patients. Med Care. 2008;46(4):388-393.
27. National Cancer Institute. Cancer-related post-traumatic stress (PDQ®)–Patient version. https://www.cancer.gov/about-cancer/coping/survivorship/new-normal/ptsd-pdq. Updated July 7, 2015. Accessed July 12, 2017.
28. Andersen BL, DeRubeis RJ, Berman BS, et al; American Society of Clinical Oncology. Screening, assessment, and care of anxiety and depressive symptoms in adults with cancer: an American Society of Clinical Oncology guideline adaptation. J Clin Oncol. 2014;32(15):1605-1619.
29. Howell D, Keller-Olaman S, Oliver TK, et al. A pan-Canadian practice guideline and algorithm: screening, assessment, and supportive care of adults with cancer-related fatigue. Curr Oncol. 2013;20(3):e233-e246.
30. Kroenke K, Wu J, Bair MJ, Krebs EE, Damush TM, Tu W. Reciprocal relationship between pain and depression: a 12-month longitudinal analysis in primary care. J Pain. 2011;12(9):964-973.
31. Walker J, Hansen CH, Martin P, et al; SMaRT (Symptom Management Research Trials) Oncology-3 Team. Integrated collaborative care for major depression comorbid with a poor prognosis cancer (SMaRT oncology-3): a multicentre randomised controlled trial in patients with lung cancer. Lancet Oncol. 2014;15(10):1168-1176.
1. Derogatis LR, Morrow GR, Fetting J, et al. The prevalence of psychiatric disorders among cancer patients. JAMA. 1983;249(6):751-757.
2. Walker J, Holm Hansen C, Martin P, et al. Prevalence of depression in adults with cancer: a systematic review. Ann Oncol. 2013;24(4):895-900.
3. Sullivan DR, Forsberg CW, Ganzini L, et al. Longitudinal changes in depression symptoms and survival among patients with lung cancer: a national cohort assessment. J Clin Oncol. 2016;34(33):3984-3991.
4. Linden W, Vodermaier A, Mackenzie R, Greig D. Anxiety and depression after cancer diagnosis: prevalence rates by cancer type, gender, and age. J Affect Disord. 2012;141(2-3):343-351.
5. Massie MJ. Prevalence of depression in patients with cancer. J Natl Cancer Inst Monogr. 2004;(32):57-71.
6. Brown Johnson CG, Brodsky JL, Cataldo JK. Lung cancer stigma, anxiety, depression, and quality of life. J Psychosoc Oncol. 2014;32(1):59-73.
7. Cataldo JK, Jahan TM, Pongquan VL. Lung cancer stigma, depression, and quality of life among ever and never smokers. Eur J Oncol Nurs. 2012;16(3):264-269.
8. Howlader N, Noone AM, Krapcho M, et al. SEER cancer statistics review, 1975-2010. https://seer.cancer.gov/archive/csr/1975_2010/. Revised February 21, 2014. Accessed July 12, 2017.
9. Li M, Boquiren V, Lo C, et al. Depression and anxiety in supportive oncology. In: Davis M, Feyer P, Ortner P, Zimmermann C, eds. Supportive Oncology. 1st ed. Philadelphia, PA: Elsevier; 2011:528-540.
10. Brown LF, Kroenke K, Theobald DE, Wu J, Tu W. The association of depression and anxiety with health-related quality of life in cancer patients with depression and/or pain. Psychooncology. 2010;19(7):734-741.
11. Lazenby M, Ercolano E, Grant M, Holland JC, Jacobsen PB, McCorkle R. Supporting Commission on Cancer-mandated psychosocial distress screening with implementation strategies. J Oncol Pract. 2015;11(3):e413-e420.
12. Mystakidou K, Tsilika E, Parpa E, Katsouda E, Galanos A, Vlahos L. Psychological distress of patients with advanced cancer: influence and contribution of pain severity and pain interference. Cancer Nurs. 2006;29(5):400-405.
13. Passik SD, Dugan W, McDonald MV, Rosenfeld B, Theobald DE, Edgerton S. Oncologists’ recognition of depression in their patients with cancer. J Clin Oncol. 1998;16(4):1594-1600.
14. Rahuma M, Kamel M, Nasar A, et al. Lung cancer patients have the highest malignancy-associated suicide rate in USA: a population based analysis. Am J Respir Crit Care Med. 2017;195:A6730.
15. Sullivan DR, Ganzini L, Duckart JP, et al. Treatment receipt and outcomes among lung cancer patients with depression. Clin Oncol (R Coll Radiol). 2014;26(1):25-31.
16. Merckaert I, Libert Y, Messin S, Milani M, Slachmuylder JL, Razavi D. Cancer patients’ desire for psychological support: prevalence and implications for screening patients psychological needs. Psychooncology. 2010;19(2):141-149.
17. Harrison JD, Young JM, Price MA, Butow PN, Solomon MJ. What are the unmet supportive care needs of people with cancer? A systematic review. Support Care Cancer. 2009;17(8):1117-1128.
18. Wu HS, Harden JK. Symptom burden and quality of life in survivorship: a review of the literature. Cancer Nurs. 2015;38(1):E29-E54.
19. Mols F, Husson O, Roukema JA, van de Poll-Franse LV. Depressive symptoms are a risk factor for all-cause mortality: results from a prospective population-based study among 3,080 cancer survivors from the PROFILES registry. J Cancer Surviv. 2013;7(3):484-492.
20. Hoge CW, Castro CA, Messer SC, McGurk D, Cotting DI, Koffman RL. Combat duty in Iraq and Afghanistan, mental health problems, and barriers to care. N Engl J Med. 2004;351(1):13-22.
21. Fortney JC, Curran GM, Hunt JB, et al. Prevalence of probable mental disorders and help-seeking behaviors among veteran and non-veteran community college students. Gen Hosp Psychiatry. 2016;38:99-104.
22. Pickett T, Rothman D, Crawford EF, Brancu M, Fairbank JA, Kudler HS. Mental health among military personnel and veterans. N C Med J. 2015;76(5):299-306.
23. U.S. Department of Veterans Affairs, Veterans Health Administration. One in ten older vets is depressed. https://www.va.gov/health/NewsFeatures/20110624a.asp. Updated April 17, 2015. Accessed July 12, 2017.
24. Fontana A, Rosenheck R. Treatment-seeking veterans of Iraq and Afghanistan: comparison with veterans of previous wars. J Nerv Ment Dis. 2008;196(7):513-521.
25. Kessler RC. Posttraumatic stress disorder: the burden to the individual and to society. J Clin Psychiatry. 2000;61(suppl 5):4-12; discussion, 13-14.
26. Kartha A, Brower V, Saitz R, Samet JH, Keane TM, Liebschutz J. The impact of trauma exposure and post-traumatic stress disorder on healthcare utilization among primary care patients. Med Care. 2008;46(4):388-393.
27. National Cancer Institute. Cancer-related post-traumatic stress (PDQ®)–Patient version. https://www.cancer.gov/about-cancer/coping/survivorship/new-normal/ptsd-pdq. Updated July 7, 2015. Accessed July 12, 2017.
28. Andersen BL, DeRubeis RJ, Berman BS, et al; American Society of Clinical Oncology. Screening, assessment, and care of anxiety and depressive symptoms in adults with cancer: an American Society of Clinical Oncology guideline adaptation. J Clin Oncol. 2014;32(15):1605-1619.
29. Howell D, Keller-Olaman S, Oliver TK, et al. A pan-Canadian practice guideline and algorithm: screening, assessment, and supportive care of adults with cancer-related fatigue. Curr Oncol. 2013;20(3):e233-e246.
30. Kroenke K, Wu J, Bair MJ, Krebs EE, Damush TM, Tu W. Reciprocal relationship between pain and depression: a 12-month longitudinal analysis in primary care. J Pain. 2011;12(9):964-973.
31. Walker J, Hansen CH, Martin P, et al; SMaRT (Symptom Management Research Trials) Oncology-3 Team. Integrated collaborative care for major depression comorbid with a poor prognosis cancer (SMaRT oncology-3): a multicentre randomised controlled trial in patients with lung cancer. Lancet Oncol. 2014;15(10):1168-1176.
Expanded approval for daratumumab in multiple myeloma
In November 2016, the US Food and Drug Administration expanded the approval of daratumumab for patients with multiple myeloma. The monoclonal antibody, which targets CD38, a protein that is highly expressed on the surface of multiple myeloma cells, was previously granted approval by the agency as a single agent for the treatment of patients who had received at least three previous therapies.
The current approval was for the use of daratumumab in two different combination regimens for the treatment of patients who have received one previous line of treatment. On the basis of improved progression-free survival (PFS), demonstrated in two randomized, open-label, phase 3 trials, daratumumab can now be used in combination with the immunomodulatory agent lenalidomide and dexamethasone, or the proteasome inhibitor bortezomib and dexamethasone, both standard therapies for the treatment of multiple myeloma.
In the POLLUX trial, 569 patients with relapsed/refractory multiple myeloma were randomized 1:1 to receive daratumumab in combination with lenalidomide-dexamethasone or lenalidomide-dexamethasone alone. The CASTOR trial randomized 498 patients with relapsed/refractory multiple myeloma 1:1 to daratumumab in combination with bortezomib-dexamethasone, or bortezomib-dexamethasone alone.
The eligibility and exclusion criteria for both trials were similar; patients had received at least one previous line of therapy, had documented progressive disease according to International Myeloma Working Group criteria, and had measurable disease on the basis of urine and/or serum assessments or serum-free, light-chain assay.
Patients with a neutrophil count of ≤1,000 cells/mm3, hemoglobin level of ≤7.5 g/dL, platelet count of <75,000 cells/mm3, creatinine clearance of ≤20 mL/min per 1.73m2 body surface area (or <30 mL/min in the POLLUX trial), alanine aminotransferase or aspartate aminotransferase level ≥2.5 times the upper limit of normal (ULN) range, bilirubin level of ≥1.5 or more times the ULN range, disease refractory to bortezomib or lenalidomide, and unacceptable side effects from bortezomib or lenalidomide, were ineligible for these studies. In addition, patients with grade 2 or higher peripheral neuropathy or neuropathic pain, were excluded from the CASTOR study.
Randomization was stratified according to International Staging System disease stage at the time of screening (stage I, II or III, with higher stage indicating more severe disease), number of previous lines of therapy (1 vs 2, or 3 vs >3), and previous receipt of lenalidomide or bortezomib.
In the CASTOR trial, patients received up to eight 21-day cycles of bortezomib, administered subcutaneously at a dose of 1.3 mg/m2 on days 1, 4, 8, and 11 of cycles 1-8, and dexamethasone, administered orally or intravenously at a dose of 20 mg on days 1, 2, 4, 5, 8, 9, 11, and 12 for a total dose of 160 mg per cycle. Daratumumab was administered at a dose of 16 mg/kg intravenously once weekly on days 1, 8, and 15 during cycles 1 to 3, once every 3 weeks on day 1 of cycles 4-8, and once every 4 weeks thereafter.
In the POLLUX trial, patients were treated in 28-day cycles. Daratumumab was administered at the same dose as in the CASTOR trial, but on days 1, 8, 15 and 22 for 8 weeks during cycles 1 and 2, every 2 weeks on days 1 and 15 for 16 weeks during cycles 3 through 7, and every 4 weeks from then onwards. Lenalidomide was administered at a dose of 25 mg orally on days 1-21 of each cycle, and dexamethasone at a dose of 20 mg before infusion and 20 mg the following day.
The combination of daratumumab with lenalidomide-dexamethasone demonstrated a substantial improvement in PFS, compared with lenalidomide-dexamethasone alone (estimated PFS not yet reached vs 18.4 months, respectively; HR, 0.37; P < .0001), representing a 63% reduction in the risk of disease progression or death. Meanwhile, there was a 61% reduction in the risk of disease progression or death for the combination of daratumumab with bortezomib-dexamethasone in the CASTOR trial (estimated PFS not yet reached vs 7.2 months; HR: 0.39; P < .0001). The PFS benefit was observed across all prespecified subgroups in both studies.
In the CASTOR trial, over a median follow-up of 7.4 months, the overall response rate (ORR) was 82.9% for the combination arm, compared with 63.2% for the bortezomib-dexamethasone arm (P < .001), with a very good partial response (VGPR) or better rate of 59.2% compared with 29.1%, and a complete response (CR) rate of 19.2% compared with 9%. In the POLLUX trial, over a median follow-up of 13.5 months, ORR was 92.9% for the combination arm, compared with 76.4% for lenalidomide-dexamethasone, with a VGPR or better rate of 75.8% versus 44% and a CR rate of 43.1% versus 19.2%.
Overall, the safety profile for both combinations was consistent with what is usually observed with daratumumab monotherapy and lenalidomide-dexamethasone or bortezomib-dexamethasone combinations. The most frequently reported adverse events (AEs) were similar in both studies and included infusion reactions, diarrhea, and upper respiratory tract infection. In the POLLUX trial they also included nausea, fatigue, pyrexia, muscle spasm, cough, and dyspnea, whereas in the CASTOR trial patients also frequently experienced peripheral edema.
The most common grade 3/4 AEs in both trials were neutropenia (51.9% vs 37% in the POLLUX trial and 12.8 vs 4.2% in the CASTOR trial), thrombocytopenia (12.7% vs 13.5% and 45.3% vs 32.9%, respectively), and anemia (12.4% vs 19.6% and 14.4% vs 16%, respectively). The percentage of patients who discontinued treatment due to AEs was similar in both groups across the two studies; in the CASTOR trial discontinuations resulted most commonly from peripheral sensory neuropathy and pneumonia, while in the POLLUX trial, from pneumonia, pulmonary embolism and deterioration in general physical health.
The recommended dose for daratumumab in both combination regimens is 16 mg/kg intravenously, calculated on actual body weight. The dosing schedules begin with weekly administration during weeks 1-8 (when used in combination with lenalidomide-dexamethasone) and weeks 1-9 (for use with the bortezomib-dexamethasone combination), decreasing to every 2 weeks between weeks 9 and 24 or 10 and 24, respectively, and progressing to every 4 weeks from week 25 onward until disease progression and unacceptable toxicity.
Daratumumab is marketed as Darzalex by Janssen Biotech Inc. Neutropenia and thrombocytopenia have been added to the list of warnings and precautions for the prescribing information for these new indications. Complete blood cell count should be monitored periodically during treatment and daratumumab administration delayed to allow recovery of neutrophils or platelets. Supportive care with growth factors or transfusion should be considered in the event of neutropenia or thrombocytopenia, respectively.
1. Darzalex (daratumumab) injection, for intravenous use. Prescribing information. Janssen Biotech Inc. https://www.darzalexhcp.com/shared/product/darzalex/darzalex-prescribing-information.pdf. Released November 2016. Accessed January 8, 2017.
2. Palumbo A, Chanan-Khan A, Weisel K, et al. Daratumumab, bortezomib, and dexamethasone for multiple myeloma. N Engl J Med. 2016;375:754-766.
3. Dimopoulos MA, Oriol A, Nahi H, et al. Daratumumab, lenalidomide, and dexamethasone for multiple myeloma. N Engl J Med. 2016;375:1319-1331.
In November 2016, the US Food and Drug Administration expanded the approval of daratumumab for patients with multiple myeloma. The monoclonal antibody, which targets CD38, a protein that is highly expressed on the surface of multiple myeloma cells, was previously granted approval by the agency as a single agent for the treatment of patients who had received at least three previous therapies.
The current approval was for the use of daratumumab in two different combination regimens for the treatment of patients who have received one previous line of treatment. On the basis of improved progression-free survival (PFS), demonstrated in two randomized, open-label, phase 3 trials, daratumumab can now be used in combination with the immunomodulatory agent lenalidomide and dexamethasone, or the proteasome inhibitor bortezomib and dexamethasone, both standard therapies for the treatment of multiple myeloma.
In the POLLUX trial, 569 patients with relapsed/refractory multiple myeloma were randomized 1:1 to receive daratumumab in combination with lenalidomide-dexamethasone or lenalidomide-dexamethasone alone. The CASTOR trial randomized 498 patients with relapsed/refractory multiple myeloma 1:1 to daratumumab in combination with bortezomib-dexamethasone, or bortezomib-dexamethasone alone.
The eligibility and exclusion criteria for both trials were similar; patients had received at least one previous line of therapy, had documented progressive disease according to International Myeloma Working Group criteria, and had measurable disease on the basis of urine and/or serum assessments or serum-free, light-chain assay.
Patients with a neutrophil count of ≤1,000 cells/mm3, hemoglobin level of ≤7.5 g/dL, platelet count of <75,000 cells/mm3, creatinine clearance of ≤20 mL/min per 1.73m2 body surface area (or <30 mL/min in the POLLUX trial), alanine aminotransferase or aspartate aminotransferase level ≥2.5 times the upper limit of normal (ULN) range, bilirubin level of ≥1.5 or more times the ULN range, disease refractory to bortezomib or lenalidomide, and unacceptable side effects from bortezomib or lenalidomide, were ineligible for these studies. In addition, patients with grade 2 or higher peripheral neuropathy or neuropathic pain, were excluded from the CASTOR study.
Randomization was stratified according to International Staging System disease stage at the time of screening (stage I, II or III, with higher stage indicating more severe disease), number of previous lines of therapy (1 vs 2, or 3 vs >3), and previous receipt of lenalidomide or bortezomib.
In the CASTOR trial, patients received up to eight 21-day cycles of bortezomib, administered subcutaneously at a dose of 1.3 mg/m2 on days 1, 4, 8, and 11 of cycles 1-8, and dexamethasone, administered orally or intravenously at a dose of 20 mg on days 1, 2, 4, 5, 8, 9, 11, and 12 for a total dose of 160 mg per cycle. Daratumumab was administered at a dose of 16 mg/kg intravenously once weekly on days 1, 8, and 15 during cycles 1 to 3, once every 3 weeks on day 1 of cycles 4-8, and once every 4 weeks thereafter.
In the POLLUX trial, patients were treated in 28-day cycles. Daratumumab was administered at the same dose as in the CASTOR trial, but on days 1, 8, 15 and 22 for 8 weeks during cycles 1 and 2, every 2 weeks on days 1 and 15 for 16 weeks during cycles 3 through 7, and every 4 weeks from then onwards. Lenalidomide was administered at a dose of 25 mg orally on days 1-21 of each cycle, and dexamethasone at a dose of 20 mg before infusion and 20 mg the following day.
The combination of daratumumab with lenalidomide-dexamethasone demonstrated a substantial improvement in PFS, compared with lenalidomide-dexamethasone alone (estimated PFS not yet reached vs 18.4 months, respectively; HR, 0.37; P < .0001), representing a 63% reduction in the risk of disease progression or death. Meanwhile, there was a 61% reduction in the risk of disease progression or death for the combination of daratumumab with bortezomib-dexamethasone in the CASTOR trial (estimated PFS not yet reached vs 7.2 months; HR: 0.39; P < .0001). The PFS benefit was observed across all prespecified subgroups in both studies.
In the CASTOR trial, over a median follow-up of 7.4 months, the overall response rate (ORR) was 82.9% for the combination arm, compared with 63.2% for the bortezomib-dexamethasone arm (P < .001), with a very good partial response (VGPR) or better rate of 59.2% compared with 29.1%, and a complete response (CR) rate of 19.2% compared with 9%. In the POLLUX trial, over a median follow-up of 13.5 months, ORR was 92.9% for the combination arm, compared with 76.4% for lenalidomide-dexamethasone, with a VGPR or better rate of 75.8% versus 44% and a CR rate of 43.1% versus 19.2%.
Overall, the safety profile for both combinations was consistent with what is usually observed with daratumumab monotherapy and lenalidomide-dexamethasone or bortezomib-dexamethasone combinations. The most frequently reported adverse events (AEs) were similar in both studies and included infusion reactions, diarrhea, and upper respiratory tract infection. In the POLLUX trial they also included nausea, fatigue, pyrexia, muscle spasm, cough, and dyspnea, whereas in the CASTOR trial patients also frequently experienced peripheral edema.
The most common grade 3/4 AEs in both trials were neutropenia (51.9% vs 37% in the POLLUX trial and 12.8 vs 4.2% in the CASTOR trial), thrombocytopenia (12.7% vs 13.5% and 45.3% vs 32.9%, respectively), and anemia (12.4% vs 19.6% and 14.4% vs 16%, respectively). The percentage of patients who discontinued treatment due to AEs was similar in both groups across the two studies; in the CASTOR trial discontinuations resulted most commonly from peripheral sensory neuropathy and pneumonia, while in the POLLUX trial, from pneumonia, pulmonary embolism and deterioration in general physical health.
The recommended dose for daratumumab in both combination regimens is 16 mg/kg intravenously, calculated on actual body weight. The dosing schedules begin with weekly administration during weeks 1-8 (when used in combination with lenalidomide-dexamethasone) and weeks 1-9 (for use with the bortezomib-dexamethasone combination), decreasing to every 2 weeks between weeks 9 and 24 or 10 and 24, respectively, and progressing to every 4 weeks from week 25 onward until disease progression and unacceptable toxicity.
Daratumumab is marketed as Darzalex by Janssen Biotech Inc. Neutropenia and thrombocytopenia have been added to the list of warnings and precautions for the prescribing information for these new indications. Complete blood cell count should be monitored periodically during treatment and daratumumab administration delayed to allow recovery of neutrophils or platelets. Supportive care with growth factors or transfusion should be considered in the event of neutropenia or thrombocytopenia, respectively.
In November 2016, the US Food and Drug Administration expanded the approval of daratumumab for patients with multiple myeloma. The monoclonal antibody, which targets CD38, a protein that is highly expressed on the surface of multiple myeloma cells, was previously granted approval by the agency as a single agent for the treatment of patients who had received at least three previous therapies.
The current approval was for the use of daratumumab in two different combination regimens for the treatment of patients who have received one previous line of treatment. On the basis of improved progression-free survival (PFS), demonstrated in two randomized, open-label, phase 3 trials, daratumumab can now be used in combination with the immunomodulatory agent lenalidomide and dexamethasone, or the proteasome inhibitor bortezomib and dexamethasone, both standard therapies for the treatment of multiple myeloma.
In the POLLUX trial, 569 patients with relapsed/refractory multiple myeloma were randomized 1:1 to receive daratumumab in combination with lenalidomide-dexamethasone or lenalidomide-dexamethasone alone. The CASTOR trial randomized 498 patients with relapsed/refractory multiple myeloma 1:1 to daratumumab in combination with bortezomib-dexamethasone, or bortezomib-dexamethasone alone.
The eligibility and exclusion criteria for both trials were similar; patients had received at least one previous line of therapy, had documented progressive disease according to International Myeloma Working Group criteria, and had measurable disease on the basis of urine and/or serum assessments or serum-free, light-chain assay.
Patients with a neutrophil count of ≤1,000 cells/mm3, hemoglobin level of ≤7.5 g/dL, platelet count of <75,000 cells/mm3, creatinine clearance of ≤20 mL/min per 1.73m2 body surface area (or <30 mL/min in the POLLUX trial), alanine aminotransferase or aspartate aminotransferase level ≥2.5 times the upper limit of normal (ULN) range, bilirubin level of ≥1.5 or more times the ULN range, disease refractory to bortezomib or lenalidomide, and unacceptable side effects from bortezomib or lenalidomide, were ineligible for these studies. In addition, patients with grade 2 or higher peripheral neuropathy or neuropathic pain, were excluded from the CASTOR study.
Randomization was stratified according to International Staging System disease stage at the time of screening (stage I, II or III, with higher stage indicating more severe disease), number of previous lines of therapy (1 vs 2, or 3 vs >3), and previous receipt of lenalidomide or bortezomib.
In the CASTOR trial, patients received up to eight 21-day cycles of bortezomib, administered subcutaneously at a dose of 1.3 mg/m2 on days 1, 4, 8, and 11 of cycles 1-8, and dexamethasone, administered orally or intravenously at a dose of 20 mg on days 1, 2, 4, 5, 8, 9, 11, and 12 for a total dose of 160 mg per cycle. Daratumumab was administered at a dose of 16 mg/kg intravenously once weekly on days 1, 8, and 15 during cycles 1 to 3, once every 3 weeks on day 1 of cycles 4-8, and once every 4 weeks thereafter.
In the POLLUX trial, patients were treated in 28-day cycles. Daratumumab was administered at the same dose as in the CASTOR trial, but on days 1, 8, 15 and 22 for 8 weeks during cycles 1 and 2, every 2 weeks on days 1 and 15 for 16 weeks during cycles 3 through 7, and every 4 weeks from then onwards. Lenalidomide was administered at a dose of 25 mg orally on days 1-21 of each cycle, and dexamethasone at a dose of 20 mg before infusion and 20 mg the following day.
The combination of daratumumab with lenalidomide-dexamethasone demonstrated a substantial improvement in PFS, compared with lenalidomide-dexamethasone alone (estimated PFS not yet reached vs 18.4 months, respectively; HR, 0.37; P < .0001), representing a 63% reduction in the risk of disease progression or death. Meanwhile, there was a 61% reduction in the risk of disease progression or death for the combination of daratumumab with bortezomib-dexamethasone in the CASTOR trial (estimated PFS not yet reached vs 7.2 months; HR: 0.39; P < .0001). The PFS benefit was observed across all prespecified subgroups in both studies.
In the CASTOR trial, over a median follow-up of 7.4 months, the overall response rate (ORR) was 82.9% for the combination arm, compared with 63.2% for the bortezomib-dexamethasone arm (P < .001), with a very good partial response (VGPR) or better rate of 59.2% compared with 29.1%, and a complete response (CR) rate of 19.2% compared with 9%. In the POLLUX trial, over a median follow-up of 13.5 months, ORR was 92.9% for the combination arm, compared with 76.4% for lenalidomide-dexamethasone, with a VGPR or better rate of 75.8% versus 44% and a CR rate of 43.1% versus 19.2%.
Overall, the safety profile for both combinations was consistent with what is usually observed with daratumumab monotherapy and lenalidomide-dexamethasone or bortezomib-dexamethasone combinations. The most frequently reported adverse events (AEs) were similar in both studies and included infusion reactions, diarrhea, and upper respiratory tract infection. In the POLLUX trial they also included nausea, fatigue, pyrexia, muscle spasm, cough, and dyspnea, whereas in the CASTOR trial patients also frequently experienced peripheral edema.
The most common grade 3/4 AEs in both trials were neutropenia (51.9% vs 37% in the POLLUX trial and 12.8 vs 4.2% in the CASTOR trial), thrombocytopenia (12.7% vs 13.5% and 45.3% vs 32.9%, respectively), and anemia (12.4% vs 19.6% and 14.4% vs 16%, respectively). The percentage of patients who discontinued treatment due to AEs was similar in both groups across the two studies; in the CASTOR trial discontinuations resulted most commonly from peripheral sensory neuropathy and pneumonia, while in the POLLUX trial, from pneumonia, pulmonary embolism and deterioration in general physical health.
The recommended dose for daratumumab in both combination regimens is 16 mg/kg intravenously, calculated on actual body weight. The dosing schedules begin with weekly administration during weeks 1-8 (when used in combination with lenalidomide-dexamethasone) and weeks 1-9 (for use with the bortezomib-dexamethasone combination), decreasing to every 2 weeks between weeks 9 and 24 or 10 and 24, respectively, and progressing to every 4 weeks from week 25 onward until disease progression and unacceptable toxicity.
Daratumumab is marketed as Darzalex by Janssen Biotech Inc. Neutropenia and thrombocytopenia have been added to the list of warnings and precautions for the prescribing information for these new indications. Complete blood cell count should be monitored periodically during treatment and daratumumab administration delayed to allow recovery of neutrophils or platelets. Supportive care with growth factors or transfusion should be considered in the event of neutropenia or thrombocytopenia, respectively.
1. Darzalex (daratumumab) injection, for intravenous use. Prescribing information. Janssen Biotech Inc. https://www.darzalexhcp.com/shared/product/darzalex/darzalex-prescribing-information.pdf. Released November 2016. Accessed January 8, 2017.
2. Palumbo A, Chanan-Khan A, Weisel K, et al. Daratumumab, bortezomib, and dexamethasone for multiple myeloma. N Engl J Med. 2016;375:754-766.
3. Dimopoulos MA, Oriol A, Nahi H, et al. Daratumumab, lenalidomide, and dexamethasone for multiple myeloma. N Engl J Med. 2016;375:1319-1331.
1. Darzalex (daratumumab) injection, for intravenous use. Prescribing information. Janssen Biotech Inc. https://www.darzalexhcp.com/shared/product/darzalex/darzalex-prescribing-information.pdf. Released November 2016. Accessed January 8, 2017.
2. Palumbo A, Chanan-Khan A, Weisel K, et al. Daratumumab, bortezomib, and dexamethasone for multiple myeloma. N Engl J Med. 2016;375:754-766.
3. Dimopoulos MA, Oriol A, Nahi H, et al. Daratumumab, lenalidomide, and dexamethasone for multiple myeloma. N Engl J Med. 2016;375:1319-1331.
Multifaceted pharmacist intervention may reduce postdischarge ED visits and readmissions
Clinical question: Can a multifaceted intervention by a clinical pharmacist reduce the rate of ED visits and readmission over the subsequent 180 days?
Background: The period following an inpatient admission contains many potential risks for patients, among them the risk for adverse drug events. Approximately 45% of readmissions from adverse drug reactions are thought to be avoidable.
Study design: Multicentered, single-blinded, randomized, control trial, from September 2013 to April 2015.
Setting: Four acute inpatient hospitals in Denmark.
Synopsis: 1,467 adult patients being admitted for an acute hospitalization on a minimum of five medications were randomized to receive usual care, a basic intervention (medication review by a clinical pharmacist), or an extended intervention (medication review, three motivational interviews, and follow-up with the primary care physician, pharmacy and, if appropriate, nursing home by a clinical pharmacist). The primary endpoints were readmission within 30 days or 180 days, ED visits within 180 days, and a composite endpoint of readmission or ED visit within 180 days post discharge. For these endpoints, the basic intervention group had no statistically significant difference from the usual-care group. The extended intervention group had significantly lower rates of readmission within 30 days and 180 days, as well as the primary composite endpoint compared to the usual-care group (P less than .05 for all comparisons). For the extended intervention, the number needed to treat for the main composite endpoint was 12.
Bottom line: For patients admitted to the hospital, an extended intervention by a clinical pharmacist resulted in a significant reduction in readmissions.
Citation: Ravn-Nielsen LV et al. Effect of an in-hospital multifaceted clinical pharmacist intervention on the risk of readmission. JAMA Intern Med. 2018;178(3):375-82.
Dr. Biddick is a hospitalist at Beth Israel Deaconess Medical Center, and instructor in medicine, Harvard Medical School, Boston.
Clinical question: Can a multifaceted intervention by a clinical pharmacist reduce the rate of ED visits and readmission over the subsequent 180 days?
Background: The period following an inpatient admission contains many potential risks for patients, among them the risk for adverse drug events. Approximately 45% of readmissions from adverse drug reactions are thought to be avoidable.
Study design: Multicentered, single-blinded, randomized, control trial, from September 2013 to April 2015.
Setting: Four acute inpatient hospitals in Denmark.
Synopsis: 1,467 adult patients being admitted for an acute hospitalization on a minimum of five medications were randomized to receive usual care, a basic intervention (medication review by a clinical pharmacist), or an extended intervention (medication review, three motivational interviews, and follow-up with the primary care physician, pharmacy and, if appropriate, nursing home by a clinical pharmacist). The primary endpoints were readmission within 30 days or 180 days, ED visits within 180 days, and a composite endpoint of readmission or ED visit within 180 days post discharge. For these endpoints, the basic intervention group had no statistically significant difference from the usual-care group. The extended intervention group had significantly lower rates of readmission within 30 days and 180 days, as well as the primary composite endpoint compared to the usual-care group (P less than .05 for all comparisons). For the extended intervention, the number needed to treat for the main composite endpoint was 12.
Bottom line: For patients admitted to the hospital, an extended intervention by a clinical pharmacist resulted in a significant reduction in readmissions.
Citation: Ravn-Nielsen LV et al. Effect of an in-hospital multifaceted clinical pharmacist intervention on the risk of readmission. JAMA Intern Med. 2018;178(3):375-82.
Dr. Biddick is a hospitalist at Beth Israel Deaconess Medical Center, and instructor in medicine, Harvard Medical School, Boston.
Clinical question: Can a multifaceted intervention by a clinical pharmacist reduce the rate of ED visits and readmission over the subsequent 180 days?
Background: The period following an inpatient admission contains many potential risks for patients, among them the risk for adverse drug events. Approximately 45% of readmissions from adverse drug reactions are thought to be avoidable.
Study design: Multicentered, single-blinded, randomized, control trial, from September 2013 to April 2015.
Setting: Four acute inpatient hospitals in Denmark.
Synopsis: 1,467 adult patients being admitted for an acute hospitalization on a minimum of five medications were randomized to receive usual care, a basic intervention (medication review by a clinical pharmacist), or an extended intervention (medication review, three motivational interviews, and follow-up with the primary care physician, pharmacy and, if appropriate, nursing home by a clinical pharmacist). The primary endpoints were readmission within 30 days or 180 days, ED visits within 180 days, and a composite endpoint of readmission or ED visit within 180 days post discharge. For these endpoints, the basic intervention group had no statistically significant difference from the usual-care group. The extended intervention group had significantly lower rates of readmission within 30 days and 180 days, as well as the primary composite endpoint compared to the usual-care group (P less than .05 for all comparisons). For the extended intervention, the number needed to treat for the main composite endpoint was 12.
Bottom line: For patients admitted to the hospital, an extended intervention by a clinical pharmacist resulted in a significant reduction in readmissions.
Citation: Ravn-Nielsen LV et al. Effect of an in-hospital multifaceted clinical pharmacist intervention on the risk of readmission. JAMA Intern Med. 2018;178(3):375-82.
Dr. Biddick is a hospitalist at Beth Israel Deaconess Medical Center, and instructor in medicine, Harvard Medical School, Boston.
Balanced fluid resuscitation vs. saline does not decrease hospital-free days
Clinical question: Does balanced crystalloid fluid improve outcomes versus saline in noncritically ill patients who are hospitalized?
Background: Prior research has raised concerns about a connection between intravenous saline administration and adverse outcomes. However, this work has been limited to patients in the ICU and operative room settings.
Study design: Single-center, unblinded, multiple crossover (clustered randomization) trial.
Setting: A tertiary-care, academic medical center, from January 2016 to April 2017.
Synopsis: This study enrolled 13,347 adult patients receiving a minimum of 500 cc of intravenous fluid in the emergency department. Participants were randomized to receive either normal saline or balanced crystalloid fluid (lactated Ringer’s solution or Plasma-Lyte A). The study authors found no significant difference between the two groups in the primary outcome of hospital-free days (P = .41), or in several of the secondary outcomes including acute kidney injury stage 2 or higher (P = .14) and in-hospital mortality (P = .36). The balanced crystalloid fluid group did have a significantly lower incidence of a composite secondary outcome of major adverse kidney events (P = .01). However, given the primary and other secondary outcome findings, and concerns that composite outcomes lack patient centeredness, an accompanying editorial urged caution against changing clinical practice based on this finding.
Bottom line: There was no significant difference in hospital-free days for noncritically ill patients receiving IV fluids in the ED between those treated with saline and balanced crystalloid fluid.
Citation: Self WH et al. Balanced crystalloids versus saline in noncritically ill adults. N Eng J Med. 2018;378:819-28.
Dr. Biddick is a hospitalist at Beth Israel Deaconess Medical Center, and instructor in medicine, Harvard Medical School, Boston.
Clinical question: Does balanced crystalloid fluid improve outcomes versus saline in noncritically ill patients who are hospitalized?
Background: Prior research has raised concerns about a connection between intravenous saline administration and adverse outcomes. However, this work has been limited to patients in the ICU and operative room settings.
Study design: Single-center, unblinded, multiple crossover (clustered randomization) trial.
Setting: A tertiary-care, academic medical center, from January 2016 to April 2017.
Synopsis: This study enrolled 13,347 adult patients receiving a minimum of 500 cc of intravenous fluid in the emergency department. Participants were randomized to receive either normal saline or balanced crystalloid fluid (lactated Ringer’s solution or Plasma-Lyte A). The study authors found no significant difference between the two groups in the primary outcome of hospital-free days (P = .41), or in several of the secondary outcomes including acute kidney injury stage 2 or higher (P = .14) and in-hospital mortality (P = .36). The balanced crystalloid fluid group did have a significantly lower incidence of a composite secondary outcome of major adverse kidney events (P = .01). However, given the primary and other secondary outcome findings, and concerns that composite outcomes lack patient centeredness, an accompanying editorial urged caution against changing clinical practice based on this finding.
Bottom line: There was no significant difference in hospital-free days for noncritically ill patients receiving IV fluids in the ED between those treated with saline and balanced crystalloid fluid.
Citation: Self WH et al. Balanced crystalloids versus saline in noncritically ill adults. N Eng J Med. 2018;378:819-28.
Dr. Biddick is a hospitalist at Beth Israel Deaconess Medical Center, and instructor in medicine, Harvard Medical School, Boston.
Clinical question: Does balanced crystalloid fluid improve outcomes versus saline in noncritically ill patients who are hospitalized?
Background: Prior research has raised concerns about a connection between intravenous saline administration and adverse outcomes. However, this work has been limited to patients in the ICU and operative room settings.
Study design: Single-center, unblinded, multiple crossover (clustered randomization) trial.
Setting: A tertiary-care, academic medical center, from January 2016 to April 2017.
Synopsis: This study enrolled 13,347 adult patients receiving a minimum of 500 cc of intravenous fluid in the emergency department. Participants were randomized to receive either normal saline or balanced crystalloid fluid (lactated Ringer’s solution or Plasma-Lyte A). The study authors found no significant difference between the two groups in the primary outcome of hospital-free days (P = .41), or in several of the secondary outcomes including acute kidney injury stage 2 or higher (P = .14) and in-hospital mortality (P = .36). The balanced crystalloid fluid group did have a significantly lower incidence of a composite secondary outcome of major adverse kidney events (P = .01). However, given the primary and other secondary outcome findings, and concerns that composite outcomes lack patient centeredness, an accompanying editorial urged caution against changing clinical practice based on this finding.
Bottom line: There was no significant difference in hospital-free days for noncritically ill patients receiving IV fluids in the ED between those treated with saline and balanced crystalloid fluid.
Citation: Self WH et al. Balanced crystalloids versus saline in noncritically ill adults. N Eng J Med. 2018;378:819-28.
Dr. Biddick is a hospitalist at Beth Israel Deaconess Medical Center, and instructor in medicine, Harvard Medical School, Boston.
Ketamine Plus Memantine-Based Multimodality Treatment of Chronic Refractory Migraine
Dr. Charles is Clinical Associate Professor Neurology, Rutgers–New Jersey Medical School, Newark, NJ; Neurology Attending, Holy Name Medical Center, Teaneck, NJ (jacharlesmd@gmail.com).
Dr. Gallo is Interventional Radiology Attending, Holy Name Medical Center, Teaneck, NJ (Vgallo83@gmail.com).
DISCLOSURES
The authors have no financial relationships to disclose relevant to the manuscript. There was no sponsorship of, or funding for, the study.
Dr. Charles designed and conceptualized the study; analyzed study data and performed the statistical analysis; and drafted the manuscript for intellectual content. Dr. Gallo had a major role in the acquisition of interventional sphenopalatine ganglion data.
ABSTRACT
Objective
Chronic refractory migraine patients who failed repetitive dihydroergotamine/dopamine infusion protocols and conventional preventives were treated with repeated low-dose ketamine-based parenteral protocols, followed by memantine-based preventive therapy, and observed for immediate reduction in pain intensity and headache frequency.
Methods
Ten patients were treated at an outpatient infusion center for 2 to 5 sequential days with AM and PM courses of intravenous diphenhydramine, prochlorperazine, and dihydroergotamine. A daily sphenopalatine ganglion block and low-dose intramuscular ketamine were given midday between treatments, with dexamethasone given on the last infusion day. The Numeric Pain Rating Scale was measured after infusion. Carryover effect was assessed 1 month and 2 months after infusion by headache frequency while being treated with memantine and various other preventive and abortive therapies.
Results
Reduction in headache pain of 71% was achieved at the end of the infusion period. Sedation was the only adverse effect. Decreased headache frequency persisted beyond the infusion period, with an 88.6% reduction in headache days per month at 1 month and a 79.4% reduction in headache days per month at 2 months, without adverse effects.
Conclusions
Data indicate that 1) repetitive low-dose, ketamine-based parenteral therapy, followed by memantine-based preventive therapy, reduced refractory headache pain and 2) the decremental effect on headache frequency persisted beyond the infusion period. Our results support the hypothesis that multimechanistic therapies might be better than single-modality treatment. More studies, with a larger patient population, are needed to confirm whether these multimodality ketamine/memantine therapies should become the preferred approach for these extremely disabled patients.
Chronic refractory migraine (CRM) degrades function and quality of life despite elimination of triggers and adequate trials of acute and preventive medicines that have established efficacy. This definition requires that patients with chronic migraine fail adequate trials of preventive drugs, alone or in combination, in at least 2 of 4 drug classes, including beta blockers, anticonvulsants, tricyclic antidepressants, onabotulinumtoxin A, and calcium-channel blockers. Patients must also fail adequate trials of abortive medicines, including both a triptan and dihydroergotamine (DHE), intranasal or injectable formulation, and either a nonsteroidal anti-inflammatory drug or a combination analgesic, unless contraindicated.1-4
In 1986, Raskin published a nonrandomized, nonblinded study of 2 treatments for intractable migraine in which repetitive inpatient intravenous (IV) DHE, administered in the hospital, was statistically more effective than IV diazepam in terminating cycles of intractable migraine.5 Most headache specialists have adopted the so-called Raskin protocol, as originally described or in any of several variations, as cornerstone therapy for CRM, chronic migraine, and prolonged status migrainosus.6 However, DHE-based infusion protocols do not always effectively reset the brain’s pain modulatory pathways in chronic migraine immediately posttreatment and might not induce a meaningful carryover effect.
We present 10 patients with CRM who met criteria for refractory migraine, including failure to terminate their headache with repetitive DHE/prochlorperazine/diphenhydramine/ketorolac/dexamethasone IV protocols, with or without sporadic administration of a sphenopalatine ganglion block. We treated these patients multimechanistically with repetitive IV DHE, a dopamine antagonist, an antihistamine, sphenopalatine ganglion (SPG) block, and low-dose ketamine, plus last-infusion-day dexamethasone, followed by outpatient oral memantine. Subsequently, we observed them for 2 months.
Ketamine is a phencyclidine derivative introduced the early 1960s as an IV anesthetic. Low-dose ketamine has been used successfully in the treatment of chronic pain. Today, increased interest in the application of low-dose ketamine includes cancer pain; treatment and prevention of acute and chronic pain, with and without neuropathic analgesia; fibromyalgia; complex regional pain; and migraine.7,8 The effectiveness of ketamine in different pain disorders may arise through different pathways and/or by way of activity at various receptor systems. Effects arise predominantly by noncompetitive antagonism of the glutamate N-methyl-D-aspartate (NMDA ) receptor.7,8
Memantine also is an NMDA receptor antagonist that is used effectively as an oral agent in CRM.9
METHODS
Patients enrolled in this prospective study had CRM for periods ranging from 1 to 2 years. All had daily headache that could not be terminated with repetitive DHE/prochlorperazine/diphenhydramine/ketorolac/dexamethasone IV protocols with or without sporadic administration of an SPG block. Age ranged from 18 and 68 years; all patients were female. Patients were excluded if they had known coronary artery disease, uncontrolled hypertension, or peripheral arterial disease; a history of stroke, transient ischemic attack, or pregnancy; impaired liver or renal function; smoked a tobacco product; or were taking a protease inhibitor or macrolide antibiotic.
Approval by the institutional review board was unnecessary because all drugs and procedures are FDA-approved and have published evidence-based efficacy for migraine and other diseases.
The Numeric Pain Rating Scale (NPRS; a scale of 0 to 10) was utilized to rate the intensity of pain from the beginning of the infusion to the end of the multiday infusion protocol, when the catheter was removed. All patients but 1 were treated for 5 days; for the 1 exception, treatment was terminated after 48 hours because of a scheduling conflict. The observational follow‐up periods for assessment of outcomes were 1 month and 2 months post-infusion.
Patients started the study with a baseline NPRS of 9 or 10. They were treated at the institution’s headache outpatient infusion center. In the morning, patients received, by sequential IV infusion, diphenhydramine, 50 mg; prochlorperazine, 10 mg; and DHE, 1 mg. They then received a midday SPG block under fluoroscopic guidance and ketamine, 0.45 mg/kg intramuscularly (IM), given in the post-anesthesia care unit. In the late afternoon, the patients received diphenhydramine, 50 mg; prochlorperazine, 10 mg; and DHE, 0.5 mg, in the Headache Outpatient Infusion Center. Patients were discharged to home by 6 PM. They received IV dexamethasone, 20 mg, on the last day of therapy.
Oral preventive agents were continued and abortives were temporarily discontinued during infusion therapy. Oral memantine was used immediately before, during, and, in all cases, after infusion, at a daily dosage that ranged from 10 mg BID to 28 mg, once-daily extended release.
RESULTS
Therapies were well-tolerated by all patients. On the last day of treatment, the entire cohort (N = 10) demonstrated an average of 71% (mean standard deviation [SD], 10.1%) reduction in pain intensity. The average reduction in headache days per month at 1 month was 88.6% (mean SD, 6.24%) and at 2 months was 79.4% (mean SD, 17.13%) (Table). Adverse effects were mild temporary sedation from ketamine. Pulse oximetry revealed no abnormal decrease in O2 saturation. All patients reported marked overall reduction in headache disability at the end of the infusion protocol. Self-administered abortive therapies posttreatment were more efficacious than they were pretreatment. All patients indicated less headache disability overall by the end of the 2-month observation period.
Table. Chronic Refractory Migraine Baseline Data and Treatment Resultsa
Name | Age (y) | Sex | Treatment Duration (days) | Baseline NPRS | Post-treatment NPRS | One Month Follow-upb | Two Month Follow-upb |
SL | 45 | F | 5 | 10 | 2 | 3 | 3 |
RR | 44 | F | 5 | 9 | 1 | 1 | 3 |
MP | 41 | F | 5 | 10 | 4 | 3 | 6 |
AP | 35 | F | 5 | 10 | 3 | 8 | 15 |
SW | 27 | F | 5 | 10 | 2 | 6 | 12 |
HC | 47 | F | 5 | 10 | 4 | 4 | 6 |
KK | 56 | F | 5 | 10 | 3 | 3 | 8 |
MG | 53 | F | 5 | 9 | 4 | 2 | 3 |
DM | 68 | F | 2 | 9 | 2 | 2 | 4 |
AO | 18 | F | 5 | 9 | 3 | 2 | 2 |
aAll patients had daily headache at initiation of treatment.
bHeadache days/month.
NPRS, Numeric Pain Rating Scale.
DISCUSSION
In our study of 10 patients with CRM who had daily headache treated repetitively in an outpatient infusion center with multimodality therapies, including sub-anesthetic doses of ketamine, all patients experienced marked reduction in headache pain intensity, with a whole-group average reduction of 71% by the end of infusion treatment. During post-infusion observation, all patients continued various preventive therapies, including memantine. At 1 month, the average reduction in headache frequency was 88.6%. Two months post-infusion, the average reduction in headache frequency was 79.4%. Adverse effects were minimal. Overall, the treatment was found to be safe and efficacious. All patients felt less headache disability after 2 months.
Because the protocol was administered comfortably in the Headache Outpatient Infusion Center, the inconvenience and higher cost of inpatient parenteral treatment were avoided. Ketamine, 0.45 mg/kg IM is a sub-anesthetic dose with proven efficacy in treating migraine without adverse effects in an outpatient setting.8 Low-dose ketamine obviated the need for anesthesia personnel and precautions. Temporary sedation was the only adverse effect. Ketamine was administered by a nurse in the post-anesthesia care unit while patients were under observation with conventional measurement of vital signs and pulse oximetry. Memantine, also an NMDA receptor antagonist, is postulated to prolong the NMDA antagonism of ketamine.
Inpatient and outpatient continuous IV DHE and repetitive IV DHE, often combined with dopamine antagonists in controlled and comparator studies, have demonstrated equal effectiveness for the treatment of chronic migraine.5,10,11 Our patients failed these therapies. This raises the question: Should our combined multimodality, ketamine-based approach be standard parenteral therapy for CRM?
In a recent study of continuous inpatient single-modality IV ketamine, a less-impressive carryover effect was obtained, with 23% to 50% 1-month sustained responders.12 Multimechanistic treatment superiority over monotherapy is legendary in the treatment of cancer and human immunodeficiency infection. Sumatriptan plus naproxen sodium as a single tablet for acute treatment of migraine resulted in more favorable clinical benefit compared with either monotherapy, with an acceptable, well-tolerated adverse effect profile. Because multiple pathogenic mechanisms putatively are involved in generation of the migraine symptom complex, multimechanism-targeted therapy may confer advantages over individual monotherapy. Drugs in 2 classes of migraine pharmacotherapy—triptans and nonsteroidal anti-inflammatory drugs —target distinct aspects of the vascular and inflammatory processes hypothesized to underlie migraine.13
Although combination therapy for CRM has not been systematically studied in randomized trials, clinical experience suggests that a rational approach to CRM treatment, utilizing a combination of treatments, may be effective when monotherapy has failed.14 During the infusion protocol, we re-set the trigeminovascular pain pathways 1) by repetitively blocking NMDA receptors (with ketamine), dopamine receptors (with prochlorperazine), and histamine receptors (with diphenhydramine); 2) by lidocaine anesthetic block of the sphenopalatine ganglia; and, on the last day of the protocol, 3) administering 1 large dose of IV dexamethasone to help prevent recurrence.15 NMDA blockade continued with oral outpatient memantine.
Virtually all patients were taking other preventives during the pretreatment period and 2-month observation period, including topiramate, venlafaxine, beta blockers, candesartan, zonisamide, onabotulinumtoxin A, neuromodulation (Cefaly Technology), and transcranial magnetic stimulation (springTMS®). Self-administered abortives were more effective in the 2-month observational period; these included IM/IV DHE; oral, spray, and subcutaneous triptans; IM ketorolac; diclofenac buffered solution; and transcranial magnetic stimulation (springTMS®). The cornerstone strategy of our treatment group that was a constant was the use of low-dose IM sub-anesthetic ketamine at a dosage of 0.45 mg/kg/d and the use of oral memantine during the follow-up observation period, at dosages ranging from 10 mg BID to 28 mg, once-daily extended release.
Limitations of this study design are:
- lack of a control group
- lack of subject randomization for comparative outcomes
- patients remaining on a variety of prophylactic regimens
- patients permitted to take any rescue therapy.
The effect of repetitive SPG block cannot be teased out of the efficacy data, but many of our patients had a poor or temporary response to infrequent sporadic SPG blocks prior to participating in our protocol.
Many migraineurs who seek care in a headache clinic are refractory to treatment, despite advances in headache therapy; refractory migraine was found in 5.1% of these patients.16 In this small series of patients, we demonstrated immediate relief and a significant 2-month carryover effect with our multimodality parenteral protocol. Larger, controlled studies are needed to further explore this protocol with repetitive DHE, diphenhydramine, prochlorperazine, SPG block, and low-dose IM ketamine, followed by outpatient memantine. Such studies would determine whether our protocol should be utilized as a primary treatment, instead of the conventional DHE-based Raskin and modified Raskin protocols.
Although this is a small series of patients, lack of adverse effects and impressive results should give credence to utilizing our protocol as treatment for this extremely debilitated, often desperate subset of headache patients. Data indicate that, whereas ketamine combined with other therapies immediately reduced refractory headache pain, the ameliorating effect of ketamine on CRM headache frequency and pain in our protocol persisted beyond the infusion period. This phenomenon indicates a disease-modulating role for ketamine in refractory migraine pain, possibly by means of desensitization of NMDA receptors in the trigeminal nucleus caudalis—desensitization that continued with the NMDA receptor antagonist memantine and/or restoration of inhibitory sensory control in the brain.
CONCLUSION
Our results support the hypothesis that multimechanistic therapies, including low-dose IM ketamine and memantine, might be better than single-modality treatment in this debilitated, refractory population. Future studies, with larger patient populations, are needed to confirm whether these multimodality ketamine/memantine-inclusive therapies should become the preferred approach for these extremely disabled patients.
REFERENCES
1. Goadsby PJ, Schoenen J, Ferrari MD, Silberstein SD, Dodick DW. Towards a definition of intractable headache for use in clinical practice and trials. Cephalalgia. 2006;26(9):1168-1170.
2. Schulman EA, Lipton R, Peterlin BL, Levin M, Grosberg BM. Commentary from the Refractory Headache Special Interest Section on defining the pharmacologically intractable headache for clinical trials and clinical practice. Headache. 2010;50(10):1637-1639.
3. Martelletti P, Jensen RH, Antal A, et al. Neuromodulation of chronic headaches: position statement from the European Headache Federation. J Headache Pain. 2013;14:86.
4. Dodick DW, Turkel CC, DeGryse RE, et al; PREEMPT Chronic Migraine Study Group. OnabotulinumtoxinA for treatment of chronic migraine: pooled results from the double-blind, randomized, placebo-controlled phases of the PREEMPT clinical program. Headache. 2010;50(6):921-936.
5. Raskin NH. Repetitive intravenous dihydroergotamine as therapy for intractable migraine. Neurology. 1986;36(7):995‐997.
6. Charles JA, von Dohln P. Outpatient home-based continuous intravenous dihydroergotamine therapy for intractable migraine. Headache. 2010;50(5):852-860.
7. Sigtermans M, Noppers I, Sarton E, et al. An observational study on the effect of S+-ketamine on chronic pain versus experimental acute pain in complex regional pain syndrome type 1 patients. Eur J Pain. 2010;14(3):302-307.
8. Krusz J, Cagle J, Hall S. Intramuscular (IM) ketamine for treating headache and pain flare-ups in the clinic. J Pain. 2008;9(4):30.
9. Bigal M Rapoport A, Sheftell F, Tepper D, Tepper S. Memantine in the preventive treatment of refractory migraine. Headache. 2008;48(9):1337-1342.
10. Ford RG, Ford KT. Continuous intravenous dihydroergotamine for treatment of intractable headache. Headache. 1997;37(3):129‐136.
11. Boudreau G, Aghai E, Marchand L, Langlois M. Outpatient intravenous dihydroergotamine for probable medication overuse headache. Headache Care. 2006;3(1):45‐49.
12. Pomeroy JL, Marmura MJ, Nahas SJ, Viscusi ER. Ketamine infusions for treatment refractory headache. Headache. 2017;57(2):276-282.
13. Brandes JL, Kudrow D, Stark SR, et al. Sumatriptan-naproxen for acute treatment of migraine: a randomized trial. JAMA. 2007;297(13):1443-1454.
14. Peterlin BL, Calhoun AH, Siegel S, Mathew NT. Rational combination therapy in refractory migraine. Headache. 2008;48(6):805-819.
15. Innes G, Macphail I, Dillon EC, Metcalfe C, Gao M. Dexamethasone prevents relapse after emergency department treatment of acute migraine: a randomized clinical trial. CJEM. 2015;1(1):26-33.
16. Irimia P, Palma JA, Fernandez-Torron R, Martinez-Vila E. Refractory migraine in a headache clinic population. BMC Neurol. 2011;11:94.
Dr. Charles is Clinical Associate Professor Neurology, Rutgers–New Jersey Medical School, Newark, NJ; Neurology Attending, Holy Name Medical Center, Teaneck, NJ (jacharlesmd@gmail.com).
Dr. Gallo is Interventional Radiology Attending, Holy Name Medical Center, Teaneck, NJ (Vgallo83@gmail.com).
DISCLOSURES
The authors have no financial relationships to disclose relevant to the manuscript. There was no sponsorship of, or funding for, the study.
Dr. Charles designed and conceptualized the study; analyzed study data and performed the statistical analysis; and drafted the manuscript for intellectual content. Dr. Gallo had a major role in the acquisition of interventional sphenopalatine ganglion data.
ABSTRACT
Objective
Chronic refractory migraine patients who failed repetitive dihydroergotamine/dopamine infusion protocols and conventional preventives were treated with repeated low-dose ketamine-based parenteral protocols, followed by memantine-based preventive therapy, and observed for immediate reduction in pain intensity and headache frequency.
Methods
Ten patients were treated at an outpatient infusion center for 2 to 5 sequential days with AM and PM courses of intravenous diphenhydramine, prochlorperazine, and dihydroergotamine. A daily sphenopalatine ganglion block and low-dose intramuscular ketamine were given midday between treatments, with dexamethasone given on the last infusion day. The Numeric Pain Rating Scale was measured after infusion. Carryover effect was assessed 1 month and 2 months after infusion by headache frequency while being treated with memantine and various other preventive and abortive therapies.
Results
Reduction in headache pain of 71% was achieved at the end of the infusion period. Sedation was the only adverse effect. Decreased headache frequency persisted beyond the infusion period, with an 88.6% reduction in headache days per month at 1 month and a 79.4% reduction in headache days per month at 2 months, without adverse effects.
Conclusions
Data indicate that 1) repetitive low-dose, ketamine-based parenteral therapy, followed by memantine-based preventive therapy, reduced refractory headache pain and 2) the decremental effect on headache frequency persisted beyond the infusion period. Our results support the hypothesis that multimechanistic therapies might be better than single-modality treatment. More studies, with a larger patient population, are needed to confirm whether these multimodality ketamine/memantine therapies should become the preferred approach for these extremely disabled patients.
Chronic refractory migraine (CRM) degrades function and quality of life despite elimination of triggers and adequate trials of acute and preventive medicines that have established efficacy. This definition requires that patients with chronic migraine fail adequate trials of preventive drugs, alone or in combination, in at least 2 of 4 drug classes, including beta blockers, anticonvulsants, tricyclic antidepressants, onabotulinumtoxin A, and calcium-channel blockers. Patients must also fail adequate trials of abortive medicines, including both a triptan and dihydroergotamine (DHE), intranasal or injectable formulation, and either a nonsteroidal anti-inflammatory drug or a combination analgesic, unless contraindicated.1-4
In 1986, Raskin published a nonrandomized, nonblinded study of 2 treatments for intractable migraine in which repetitive inpatient intravenous (IV) DHE, administered in the hospital, was statistically more effective than IV diazepam in terminating cycles of intractable migraine.5 Most headache specialists have adopted the so-called Raskin protocol, as originally described or in any of several variations, as cornerstone therapy for CRM, chronic migraine, and prolonged status migrainosus.6 However, DHE-based infusion protocols do not always effectively reset the brain’s pain modulatory pathways in chronic migraine immediately posttreatment and might not induce a meaningful carryover effect.
We present 10 patients with CRM who met criteria for refractory migraine, including failure to terminate their headache with repetitive DHE/prochlorperazine/diphenhydramine/ketorolac/dexamethasone IV protocols, with or without sporadic administration of a sphenopalatine ganglion block. We treated these patients multimechanistically with repetitive IV DHE, a dopamine antagonist, an antihistamine, sphenopalatine ganglion (SPG) block, and low-dose ketamine, plus last-infusion-day dexamethasone, followed by outpatient oral memantine. Subsequently, we observed them for 2 months.
Ketamine is a phencyclidine derivative introduced the early 1960s as an IV anesthetic. Low-dose ketamine has been used successfully in the treatment of chronic pain. Today, increased interest in the application of low-dose ketamine includes cancer pain; treatment and prevention of acute and chronic pain, with and without neuropathic analgesia; fibromyalgia; complex regional pain; and migraine.7,8 The effectiveness of ketamine in different pain disorders may arise through different pathways and/or by way of activity at various receptor systems. Effects arise predominantly by noncompetitive antagonism of the glutamate N-methyl-D-aspartate (NMDA ) receptor.7,8
Memantine also is an NMDA receptor antagonist that is used effectively as an oral agent in CRM.9
METHODS
Patients enrolled in this prospective study had CRM for periods ranging from 1 to 2 years. All had daily headache that could not be terminated with repetitive DHE/prochlorperazine/diphenhydramine/ketorolac/dexamethasone IV protocols with or without sporadic administration of an SPG block. Age ranged from 18 and 68 years; all patients were female. Patients were excluded if they had known coronary artery disease, uncontrolled hypertension, or peripheral arterial disease; a history of stroke, transient ischemic attack, or pregnancy; impaired liver or renal function; smoked a tobacco product; or were taking a protease inhibitor or macrolide antibiotic.
Approval by the institutional review board was unnecessary because all drugs and procedures are FDA-approved and have published evidence-based efficacy for migraine and other diseases.
The Numeric Pain Rating Scale (NPRS; a scale of 0 to 10) was utilized to rate the intensity of pain from the beginning of the infusion to the end of the multiday infusion protocol, when the catheter was removed. All patients but 1 were treated for 5 days; for the 1 exception, treatment was terminated after 48 hours because of a scheduling conflict. The observational follow‐up periods for assessment of outcomes were 1 month and 2 months post-infusion.
Patients started the study with a baseline NPRS of 9 or 10. They were treated at the institution’s headache outpatient infusion center. In the morning, patients received, by sequential IV infusion, diphenhydramine, 50 mg; prochlorperazine, 10 mg; and DHE, 1 mg. They then received a midday SPG block under fluoroscopic guidance and ketamine, 0.45 mg/kg intramuscularly (IM), given in the post-anesthesia care unit. In the late afternoon, the patients received diphenhydramine, 50 mg; prochlorperazine, 10 mg; and DHE, 0.5 mg, in the Headache Outpatient Infusion Center. Patients were discharged to home by 6 PM. They received IV dexamethasone, 20 mg, on the last day of therapy.
Oral preventive agents were continued and abortives were temporarily discontinued during infusion therapy. Oral memantine was used immediately before, during, and, in all cases, after infusion, at a daily dosage that ranged from 10 mg BID to 28 mg, once-daily extended release.
RESULTS
Therapies were well-tolerated by all patients. On the last day of treatment, the entire cohort (N = 10) demonstrated an average of 71% (mean standard deviation [SD], 10.1%) reduction in pain intensity. The average reduction in headache days per month at 1 month was 88.6% (mean SD, 6.24%) and at 2 months was 79.4% (mean SD, 17.13%) (Table). Adverse effects were mild temporary sedation from ketamine. Pulse oximetry revealed no abnormal decrease in O2 saturation. All patients reported marked overall reduction in headache disability at the end of the infusion protocol. Self-administered abortive therapies posttreatment were more efficacious than they were pretreatment. All patients indicated less headache disability overall by the end of the 2-month observation period.
Table. Chronic Refractory Migraine Baseline Data and Treatment Resultsa
Name | Age (y) | Sex | Treatment Duration (days) | Baseline NPRS | Post-treatment NPRS | One Month Follow-upb | Two Month Follow-upb |
SL | 45 | F | 5 | 10 | 2 | 3 | 3 |
RR | 44 | F | 5 | 9 | 1 | 1 | 3 |
MP | 41 | F | 5 | 10 | 4 | 3 | 6 |
AP | 35 | F | 5 | 10 | 3 | 8 | 15 |
SW | 27 | F | 5 | 10 | 2 | 6 | 12 |
HC | 47 | F | 5 | 10 | 4 | 4 | 6 |
KK | 56 | F | 5 | 10 | 3 | 3 | 8 |
MG | 53 | F | 5 | 9 | 4 | 2 | 3 |
DM | 68 | F | 2 | 9 | 2 | 2 | 4 |
AO | 18 | F | 5 | 9 | 3 | 2 | 2 |
aAll patients had daily headache at initiation of treatment.
bHeadache days/month.
NPRS, Numeric Pain Rating Scale.
DISCUSSION
In our study of 10 patients with CRM who had daily headache treated repetitively in an outpatient infusion center with multimodality therapies, including sub-anesthetic doses of ketamine, all patients experienced marked reduction in headache pain intensity, with a whole-group average reduction of 71% by the end of infusion treatment. During post-infusion observation, all patients continued various preventive therapies, including memantine. At 1 month, the average reduction in headache frequency was 88.6%. Two months post-infusion, the average reduction in headache frequency was 79.4%. Adverse effects were minimal. Overall, the treatment was found to be safe and efficacious. All patients felt less headache disability after 2 months.
Because the protocol was administered comfortably in the Headache Outpatient Infusion Center, the inconvenience and higher cost of inpatient parenteral treatment were avoided. Ketamine, 0.45 mg/kg IM is a sub-anesthetic dose with proven efficacy in treating migraine without adverse effects in an outpatient setting.8 Low-dose ketamine obviated the need for anesthesia personnel and precautions. Temporary sedation was the only adverse effect. Ketamine was administered by a nurse in the post-anesthesia care unit while patients were under observation with conventional measurement of vital signs and pulse oximetry. Memantine, also an NMDA receptor antagonist, is postulated to prolong the NMDA antagonism of ketamine.
Inpatient and outpatient continuous IV DHE and repetitive IV DHE, often combined with dopamine antagonists in controlled and comparator studies, have demonstrated equal effectiveness for the treatment of chronic migraine.5,10,11 Our patients failed these therapies. This raises the question: Should our combined multimodality, ketamine-based approach be standard parenteral therapy for CRM?
In a recent study of continuous inpatient single-modality IV ketamine, a less-impressive carryover effect was obtained, with 23% to 50% 1-month sustained responders.12 Multimechanistic treatment superiority over monotherapy is legendary in the treatment of cancer and human immunodeficiency infection. Sumatriptan plus naproxen sodium as a single tablet for acute treatment of migraine resulted in more favorable clinical benefit compared with either monotherapy, with an acceptable, well-tolerated adverse effect profile. Because multiple pathogenic mechanisms putatively are involved in generation of the migraine symptom complex, multimechanism-targeted therapy may confer advantages over individual monotherapy. Drugs in 2 classes of migraine pharmacotherapy—triptans and nonsteroidal anti-inflammatory drugs —target distinct aspects of the vascular and inflammatory processes hypothesized to underlie migraine.13
Although combination therapy for CRM has not been systematically studied in randomized trials, clinical experience suggests that a rational approach to CRM treatment, utilizing a combination of treatments, may be effective when monotherapy has failed.14 During the infusion protocol, we re-set the trigeminovascular pain pathways 1) by repetitively blocking NMDA receptors (with ketamine), dopamine receptors (with prochlorperazine), and histamine receptors (with diphenhydramine); 2) by lidocaine anesthetic block of the sphenopalatine ganglia; and, on the last day of the protocol, 3) administering 1 large dose of IV dexamethasone to help prevent recurrence.15 NMDA blockade continued with oral outpatient memantine.
Virtually all patients were taking other preventives during the pretreatment period and 2-month observation period, including topiramate, venlafaxine, beta blockers, candesartan, zonisamide, onabotulinumtoxin A, neuromodulation (Cefaly Technology), and transcranial magnetic stimulation (springTMS®). Self-administered abortives were more effective in the 2-month observational period; these included IM/IV DHE; oral, spray, and subcutaneous triptans; IM ketorolac; diclofenac buffered solution; and transcranial magnetic stimulation (springTMS®). The cornerstone strategy of our treatment group that was a constant was the use of low-dose IM sub-anesthetic ketamine at a dosage of 0.45 mg/kg/d and the use of oral memantine during the follow-up observation period, at dosages ranging from 10 mg BID to 28 mg, once-daily extended release.
Limitations of this study design are:
- lack of a control group
- lack of subject randomization for comparative outcomes
- patients remaining on a variety of prophylactic regimens
- patients permitted to take any rescue therapy.
The effect of repetitive SPG block cannot be teased out of the efficacy data, but many of our patients had a poor or temporary response to infrequent sporadic SPG blocks prior to participating in our protocol.
Many migraineurs who seek care in a headache clinic are refractory to treatment, despite advances in headache therapy; refractory migraine was found in 5.1% of these patients.16 In this small series of patients, we demonstrated immediate relief and a significant 2-month carryover effect with our multimodality parenteral protocol. Larger, controlled studies are needed to further explore this protocol with repetitive DHE, diphenhydramine, prochlorperazine, SPG block, and low-dose IM ketamine, followed by outpatient memantine. Such studies would determine whether our protocol should be utilized as a primary treatment, instead of the conventional DHE-based Raskin and modified Raskin protocols.
Although this is a small series of patients, lack of adverse effects and impressive results should give credence to utilizing our protocol as treatment for this extremely debilitated, often desperate subset of headache patients. Data indicate that, whereas ketamine combined with other therapies immediately reduced refractory headache pain, the ameliorating effect of ketamine on CRM headache frequency and pain in our protocol persisted beyond the infusion period. This phenomenon indicates a disease-modulating role for ketamine in refractory migraine pain, possibly by means of desensitization of NMDA receptors in the trigeminal nucleus caudalis—desensitization that continued with the NMDA receptor antagonist memantine and/or restoration of inhibitory sensory control in the brain.
CONCLUSION
Our results support the hypothesis that multimechanistic therapies, including low-dose IM ketamine and memantine, might be better than single-modality treatment in this debilitated, refractory population. Future studies, with larger patient populations, are needed to confirm whether these multimodality ketamine/memantine-inclusive therapies should become the preferred approach for these extremely disabled patients.
REFERENCES
1. Goadsby PJ, Schoenen J, Ferrari MD, Silberstein SD, Dodick DW. Towards a definition of intractable headache for use in clinical practice and trials. Cephalalgia. 2006;26(9):1168-1170.
2. Schulman EA, Lipton R, Peterlin BL, Levin M, Grosberg BM. Commentary from the Refractory Headache Special Interest Section on defining the pharmacologically intractable headache for clinical trials and clinical practice. Headache. 2010;50(10):1637-1639.
3. Martelletti P, Jensen RH, Antal A, et al. Neuromodulation of chronic headaches: position statement from the European Headache Federation. J Headache Pain. 2013;14:86.
4. Dodick DW, Turkel CC, DeGryse RE, et al; PREEMPT Chronic Migraine Study Group. OnabotulinumtoxinA for treatment of chronic migraine: pooled results from the double-blind, randomized, placebo-controlled phases of the PREEMPT clinical program. Headache. 2010;50(6):921-936.
5. Raskin NH. Repetitive intravenous dihydroergotamine as therapy for intractable migraine. Neurology. 1986;36(7):995‐997.
6. Charles JA, von Dohln P. Outpatient home-based continuous intravenous dihydroergotamine therapy for intractable migraine. Headache. 2010;50(5):852-860.
7. Sigtermans M, Noppers I, Sarton E, et al. An observational study on the effect of S+-ketamine on chronic pain versus experimental acute pain in complex regional pain syndrome type 1 patients. Eur J Pain. 2010;14(3):302-307.
8. Krusz J, Cagle J, Hall S. Intramuscular (IM) ketamine for treating headache and pain flare-ups in the clinic. J Pain. 2008;9(4):30.
9. Bigal M Rapoport A, Sheftell F, Tepper D, Tepper S. Memantine in the preventive treatment of refractory migraine. Headache. 2008;48(9):1337-1342.
10. Ford RG, Ford KT. Continuous intravenous dihydroergotamine for treatment of intractable headache. Headache. 1997;37(3):129‐136.
11. Boudreau G, Aghai E, Marchand L, Langlois M. Outpatient intravenous dihydroergotamine for probable medication overuse headache. Headache Care. 2006;3(1):45‐49.
12. Pomeroy JL, Marmura MJ, Nahas SJ, Viscusi ER. Ketamine infusions for treatment refractory headache. Headache. 2017;57(2):276-282.
13. Brandes JL, Kudrow D, Stark SR, et al. Sumatriptan-naproxen for acute treatment of migraine: a randomized trial. JAMA. 2007;297(13):1443-1454.
14. Peterlin BL, Calhoun AH, Siegel S, Mathew NT. Rational combination therapy in refractory migraine. Headache. 2008;48(6):805-819.
15. Innes G, Macphail I, Dillon EC, Metcalfe C, Gao M. Dexamethasone prevents relapse after emergency department treatment of acute migraine: a randomized clinical trial. CJEM. 2015;1(1):26-33.
16. Irimia P, Palma JA, Fernandez-Torron R, Martinez-Vila E. Refractory migraine in a headache clinic population. BMC Neurol. 2011;11:94.
Dr. Charles is Clinical Associate Professor Neurology, Rutgers–New Jersey Medical School, Newark, NJ; Neurology Attending, Holy Name Medical Center, Teaneck, NJ (jacharlesmd@gmail.com).
Dr. Gallo is Interventional Radiology Attending, Holy Name Medical Center, Teaneck, NJ (Vgallo83@gmail.com).
DISCLOSURES
The authors have no financial relationships to disclose relevant to the manuscript. There was no sponsorship of, or funding for, the study.
Dr. Charles designed and conceptualized the study; analyzed study data and performed the statistical analysis; and drafted the manuscript for intellectual content. Dr. Gallo had a major role in the acquisition of interventional sphenopalatine ganglion data.
ABSTRACT
Objective
Chronic refractory migraine patients who failed repetitive dihydroergotamine/dopamine infusion protocols and conventional preventives were treated with repeated low-dose ketamine-based parenteral protocols, followed by memantine-based preventive therapy, and observed for immediate reduction in pain intensity and headache frequency.
Methods
Ten patients were treated at an outpatient infusion center for 2 to 5 sequential days with AM and PM courses of intravenous diphenhydramine, prochlorperazine, and dihydroergotamine. A daily sphenopalatine ganglion block and low-dose intramuscular ketamine were given midday between treatments, with dexamethasone given on the last infusion day. The Numeric Pain Rating Scale was measured after infusion. Carryover effect was assessed 1 month and 2 months after infusion by headache frequency while being treated with memantine and various other preventive and abortive therapies.
Results
Reduction in headache pain of 71% was achieved at the end of the infusion period. Sedation was the only adverse effect. Decreased headache frequency persisted beyond the infusion period, with an 88.6% reduction in headache days per month at 1 month and a 79.4% reduction in headache days per month at 2 months, without adverse effects.
Conclusions
Data indicate that 1) repetitive low-dose, ketamine-based parenteral therapy, followed by memantine-based preventive therapy, reduced refractory headache pain and 2) the decremental effect on headache frequency persisted beyond the infusion period. Our results support the hypothesis that multimechanistic therapies might be better than single-modality treatment. More studies, with a larger patient population, are needed to confirm whether these multimodality ketamine/memantine therapies should become the preferred approach for these extremely disabled patients.
Chronic refractory migraine (CRM) degrades function and quality of life despite elimination of triggers and adequate trials of acute and preventive medicines that have established efficacy. This definition requires that patients with chronic migraine fail adequate trials of preventive drugs, alone or in combination, in at least 2 of 4 drug classes, including beta blockers, anticonvulsants, tricyclic antidepressants, onabotulinumtoxin A, and calcium-channel blockers. Patients must also fail adequate trials of abortive medicines, including both a triptan and dihydroergotamine (DHE), intranasal or injectable formulation, and either a nonsteroidal anti-inflammatory drug or a combination analgesic, unless contraindicated.1-4
In 1986, Raskin published a nonrandomized, nonblinded study of 2 treatments for intractable migraine in which repetitive inpatient intravenous (IV) DHE, administered in the hospital, was statistically more effective than IV diazepam in terminating cycles of intractable migraine.5 Most headache specialists have adopted the so-called Raskin protocol, as originally described or in any of several variations, as cornerstone therapy for CRM, chronic migraine, and prolonged status migrainosus.6 However, DHE-based infusion protocols do not always effectively reset the brain’s pain modulatory pathways in chronic migraine immediately posttreatment and might not induce a meaningful carryover effect.
We present 10 patients with CRM who met criteria for refractory migraine, including failure to terminate their headache with repetitive DHE/prochlorperazine/diphenhydramine/ketorolac/dexamethasone IV protocols, with or without sporadic administration of a sphenopalatine ganglion block. We treated these patients multimechanistically with repetitive IV DHE, a dopamine antagonist, an antihistamine, sphenopalatine ganglion (SPG) block, and low-dose ketamine, plus last-infusion-day dexamethasone, followed by outpatient oral memantine. Subsequently, we observed them for 2 months.
Ketamine is a phencyclidine derivative introduced the early 1960s as an IV anesthetic. Low-dose ketamine has been used successfully in the treatment of chronic pain. Today, increased interest in the application of low-dose ketamine includes cancer pain; treatment and prevention of acute and chronic pain, with and without neuropathic analgesia; fibromyalgia; complex regional pain; and migraine.7,8 The effectiveness of ketamine in different pain disorders may arise through different pathways and/or by way of activity at various receptor systems. Effects arise predominantly by noncompetitive antagonism of the glutamate N-methyl-D-aspartate (NMDA ) receptor.7,8
Memantine also is an NMDA receptor antagonist that is used effectively as an oral agent in CRM.9
METHODS
Patients enrolled in this prospective study had CRM for periods ranging from 1 to 2 years. All had daily headache that could not be terminated with repetitive DHE/prochlorperazine/diphenhydramine/ketorolac/dexamethasone IV protocols with or without sporadic administration of an SPG block. Age ranged from 18 and 68 years; all patients were female. Patients were excluded if they had known coronary artery disease, uncontrolled hypertension, or peripheral arterial disease; a history of stroke, transient ischemic attack, or pregnancy; impaired liver or renal function; smoked a tobacco product; or were taking a protease inhibitor or macrolide antibiotic.
Approval by the institutional review board was unnecessary because all drugs and procedures are FDA-approved and have published evidence-based efficacy for migraine and other diseases.
The Numeric Pain Rating Scale (NPRS; a scale of 0 to 10) was utilized to rate the intensity of pain from the beginning of the infusion to the end of the multiday infusion protocol, when the catheter was removed. All patients but 1 were treated for 5 days; for the 1 exception, treatment was terminated after 48 hours because of a scheduling conflict. The observational follow‐up periods for assessment of outcomes were 1 month and 2 months post-infusion.
Patients started the study with a baseline NPRS of 9 or 10. They were treated at the institution’s headache outpatient infusion center. In the morning, patients received, by sequential IV infusion, diphenhydramine, 50 mg; prochlorperazine, 10 mg; and DHE, 1 mg. They then received a midday SPG block under fluoroscopic guidance and ketamine, 0.45 mg/kg intramuscularly (IM), given in the post-anesthesia care unit. In the late afternoon, the patients received diphenhydramine, 50 mg; prochlorperazine, 10 mg; and DHE, 0.5 mg, in the Headache Outpatient Infusion Center. Patients were discharged to home by 6 PM. They received IV dexamethasone, 20 mg, on the last day of therapy.
Oral preventive agents were continued and abortives were temporarily discontinued during infusion therapy. Oral memantine was used immediately before, during, and, in all cases, after infusion, at a daily dosage that ranged from 10 mg BID to 28 mg, once-daily extended release.
RESULTS
Therapies were well-tolerated by all patients. On the last day of treatment, the entire cohort (N = 10) demonstrated an average of 71% (mean standard deviation [SD], 10.1%) reduction in pain intensity. The average reduction in headache days per month at 1 month was 88.6% (mean SD, 6.24%) and at 2 months was 79.4% (mean SD, 17.13%) (Table). Adverse effects were mild temporary sedation from ketamine. Pulse oximetry revealed no abnormal decrease in O2 saturation. All patients reported marked overall reduction in headache disability at the end of the infusion protocol. Self-administered abortive therapies posttreatment were more efficacious than they were pretreatment. All patients indicated less headache disability overall by the end of the 2-month observation period.
Table. Chronic Refractory Migraine Baseline Data and Treatment Resultsa
Name | Age (y) | Sex | Treatment Duration (days) | Baseline NPRS | Post-treatment NPRS | One Month Follow-upb | Two Month Follow-upb |
SL | 45 | F | 5 | 10 | 2 | 3 | 3 |
RR | 44 | F | 5 | 9 | 1 | 1 | 3 |
MP | 41 | F | 5 | 10 | 4 | 3 | 6 |
AP | 35 | F | 5 | 10 | 3 | 8 | 15 |
SW | 27 | F | 5 | 10 | 2 | 6 | 12 |
HC | 47 | F | 5 | 10 | 4 | 4 | 6 |
KK | 56 | F | 5 | 10 | 3 | 3 | 8 |
MG | 53 | F | 5 | 9 | 4 | 2 | 3 |
DM | 68 | F | 2 | 9 | 2 | 2 | 4 |
AO | 18 | F | 5 | 9 | 3 | 2 | 2 |
aAll patients had daily headache at initiation of treatment.
bHeadache days/month.
NPRS, Numeric Pain Rating Scale.
DISCUSSION
In our study of 10 patients with CRM who had daily headache treated repetitively in an outpatient infusion center with multimodality therapies, including sub-anesthetic doses of ketamine, all patients experienced marked reduction in headache pain intensity, with a whole-group average reduction of 71% by the end of infusion treatment. During post-infusion observation, all patients continued various preventive therapies, including memantine. At 1 month, the average reduction in headache frequency was 88.6%. Two months post-infusion, the average reduction in headache frequency was 79.4%. Adverse effects were minimal. Overall, the treatment was found to be safe and efficacious. All patients felt less headache disability after 2 months.
Because the protocol was administered comfortably in the Headache Outpatient Infusion Center, the inconvenience and higher cost of inpatient parenteral treatment were avoided. Ketamine, 0.45 mg/kg IM is a sub-anesthetic dose with proven efficacy in treating migraine without adverse effects in an outpatient setting.8 Low-dose ketamine obviated the need for anesthesia personnel and precautions. Temporary sedation was the only adverse effect. Ketamine was administered by a nurse in the post-anesthesia care unit while patients were under observation with conventional measurement of vital signs and pulse oximetry. Memantine, also an NMDA receptor antagonist, is postulated to prolong the NMDA antagonism of ketamine.
Inpatient and outpatient continuous IV DHE and repetitive IV DHE, often combined with dopamine antagonists in controlled and comparator studies, have demonstrated equal effectiveness for the treatment of chronic migraine.5,10,11 Our patients failed these therapies. This raises the question: Should our combined multimodality, ketamine-based approach be standard parenteral therapy for CRM?
In a recent study of continuous inpatient single-modality IV ketamine, a less-impressive carryover effect was obtained, with 23% to 50% 1-month sustained responders.12 Multimechanistic treatment superiority over monotherapy is legendary in the treatment of cancer and human immunodeficiency infection. Sumatriptan plus naproxen sodium as a single tablet for acute treatment of migraine resulted in more favorable clinical benefit compared with either monotherapy, with an acceptable, well-tolerated adverse effect profile. Because multiple pathogenic mechanisms putatively are involved in generation of the migraine symptom complex, multimechanism-targeted therapy may confer advantages over individual monotherapy. Drugs in 2 classes of migraine pharmacotherapy—triptans and nonsteroidal anti-inflammatory drugs —target distinct aspects of the vascular and inflammatory processes hypothesized to underlie migraine.13
Although combination therapy for CRM has not been systematically studied in randomized trials, clinical experience suggests that a rational approach to CRM treatment, utilizing a combination of treatments, may be effective when monotherapy has failed.14 During the infusion protocol, we re-set the trigeminovascular pain pathways 1) by repetitively blocking NMDA receptors (with ketamine), dopamine receptors (with prochlorperazine), and histamine receptors (with diphenhydramine); 2) by lidocaine anesthetic block of the sphenopalatine ganglia; and, on the last day of the protocol, 3) administering 1 large dose of IV dexamethasone to help prevent recurrence.15 NMDA blockade continued with oral outpatient memantine.
Virtually all patients were taking other preventives during the pretreatment period and 2-month observation period, including topiramate, venlafaxine, beta blockers, candesartan, zonisamide, onabotulinumtoxin A, neuromodulation (Cefaly Technology), and transcranial magnetic stimulation (springTMS®). Self-administered abortives were more effective in the 2-month observational period; these included IM/IV DHE; oral, spray, and subcutaneous triptans; IM ketorolac; diclofenac buffered solution; and transcranial magnetic stimulation (springTMS®). The cornerstone strategy of our treatment group that was a constant was the use of low-dose IM sub-anesthetic ketamine at a dosage of 0.45 mg/kg/d and the use of oral memantine during the follow-up observation period, at dosages ranging from 10 mg BID to 28 mg, once-daily extended release.
Limitations of this study design are:
- lack of a control group
- lack of subject randomization for comparative outcomes
- patients remaining on a variety of prophylactic regimens
- patients permitted to take any rescue therapy.
The effect of repetitive SPG block cannot be teased out of the efficacy data, but many of our patients had a poor or temporary response to infrequent sporadic SPG blocks prior to participating in our protocol.
Many migraineurs who seek care in a headache clinic are refractory to treatment, despite advances in headache therapy; refractory migraine was found in 5.1% of these patients.16 In this small series of patients, we demonstrated immediate relief and a significant 2-month carryover effect with our multimodality parenteral protocol. Larger, controlled studies are needed to further explore this protocol with repetitive DHE, diphenhydramine, prochlorperazine, SPG block, and low-dose IM ketamine, followed by outpatient memantine. Such studies would determine whether our protocol should be utilized as a primary treatment, instead of the conventional DHE-based Raskin and modified Raskin protocols.
Although this is a small series of patients, lack of adverse effects and impressive results should give credence to utilizing our protocol as treatment for this extremely debilitated, often desperate subset of headache patients. Data indicate that, whereas ketamine combined with other therapies immediately reduced refractory headache pain, the ameliorating effect of ketamine on CRM headache frequency and pain in our protocol persisted beyond the infusion period. This phenomenon indicates a disease-modulating role for ketamine in refractory migraine pain, possibly by means of desensitization of NMDA receptors in the trigeminal nucleus caudalis—desensitization that continued with the NMDA receptor antagonist memantine and/or restoration of inhibitory sensory control in the brain.
CONCLUSION
Our results support the hypothesis that multimechanistic therapies, including low-dose IM ketamine and memantine, might be better than single-modality treatment in this debilitated, refractory population. Future studies, with larger patient populations, are needed to confirm whether these multimodality ketamine/memantine-inclusive therapies should become the preferred approach for these extremely disabled patients.
REFERENCES
1. Goadsby PJ, Schoenen J, Ferrari MD, Silberstein SD, Dodick DW. Towards a definition of intractable headache for use in clinical practice and trials. Cephalalgia. 2006;26(9):1168-1170.
2. Schulman EA, Lipton R, Peterlin BL, Levin M, Grosberg BM. Commentary from the Refractory Headache Special Interest Section on defining the pharmacologically intractable headache for clinical trials and clinical practice. Headache. 2010;50(10):1637-1639.
3. Martelletti P, Jensen RH, Antal A, et al. Neuromodulation of chronic headaches: position statement from the European Headache Federation. J Headache Pain. 2013;14:86.
4. Dodick DW, Turkel CC, DeGryse RE, et al; PREEMPT Chronic Migraine Study Group. OnabotulinumtoxinA for treatment of chronic migraine: pooled results from the double-blind, randomized, placebo-controlled phases of the PREEMPT clinical program. Headache. 2010;50(6):921-936.
5. Raskin NH. Repetitive intravenous dihydroergotamine as therapy for intractable migraine. Neurology. 1986;36(7):995‐997.
6. Charles JA, von Dohln P. Outpatient home-based continuous intravenous dihydroergotamine therapy for intractable migraine. Headache. 2010;50(5):852-860.
7. Sigtermans M, Noppers I, Sarton E, et al. An observational study on the effect of S+-ketamine on chronic pain versus experimental acute pain in complex regional pain syndrome type 1 patients. Eur J Pain. 2010;14(3):302-307.
8. Krusz J, Cagle J, Hall S. Intramuscular (IM) ketamine for treating headache and pain flare-ups in the clinic. J Pain. 2008;9(4):30.
9. Bigal M Rapoport A, Sheftell F, Tepper D, Tepper S. Memantine in the preventive treatment of refractory migraine. Headache. 2008;48(9):1337-1342.
10. Ford RG, Ford KT. Continuous intravenous dihydroergotamine for treatment of intractable headache. Headache. 1997;37(3):129‐136.
11. Boudreau G, Aghai E, Marchand L, Langlois M. Outpatient intravenous dihydroergotamine for probable medication overuse headache. Headache Care. 2006;3(1):45‐49.
12. Pomeroy JL, Marmura MJ, Nahas SJ, Viscusi ER. Ketamine infusions for treatment refractory headache. Headache. 2017;57(2):276-282.
13. Brandes JL, Kudrow D, Stark SR, et al. Sumatriptan-naproxen for acute treatment of migraine: a randomized trial. JAMA. 2007;297(13):1443-1454.
14. Peterlin BL, Calhoun AH, Siegel S, Mathew NT. Rational combination therapy in refractory migraine. Headache. 2008;48(6):805-819.
15. Innes G, Macphail I, Dillon EC, Metcalfe C, Gao M. Dexamethasone prevents relapse after emergency department treatment of acute migraine: a randomized clinical trial. CJEM. 2015;1(1):26-33.
16. Irimia P, Palma JA, Fernandez-Torron R, Martinez-Vila E. Refractory migraine in a headache clinic population. BMC Neurol. 2011;11:94.