User login
The First Patient in the Veteran Affairs System to Receive Chimeric Antigen Receptors T-cell Therapy for Refractory Multiple Myeloma and the Role of Intravenous Immunoglobulin in the Prevention of Therapy-associated Infections
Background
In 3/2021, chimeric antigen receptor (CAR) T-cell therapy was approved for the treatment of multiple myeloma in adult patients with refractory disease. Currently, only the Veterans Affair (VA) center at the Tennessee Valley Healthcare System (TVHS) offers this treatment. Herein, we report a significant healthcare milestone in 2024 when the first patient received CAR T-cell therapy for multiple myeloma in the VA system. Additionally, the rate of hypogammaglobulinemia is the highest for CAR T-cell therapy using idecabtagene vicleucel compared to therapies using other antineoplastic agents (Wat et al, 2021). The complications of hypogammaglobulinemia can be mitigated by intravenous immunoglobulin (IVIG) treatment.
Case Presentation
A 75-year-old male veteran was diagnosed with IgA Kappa multiple myeloma and received induction therapy with bortezomib, lenalidomide, and dexamethasone in 2014. The patient underwent autologous stem cell transplant (SCT) in the same year. His disease recurred in 3/2019, and the patient was started on daratumumab and pomalidomide. He received another autologous SCT in 2/2021, to which he was refractory. The veteran then received treatment with daratumumab and ixazomib, followed by carfilzomib and cyclophosphamide. Starting in 9/2022, the patient also required regular IVIG treatment for hypogammaglobulinemia. He eventually received CAR T-cell therapy with idecabtagene vicleucel at THVS on 4/18/2024. The patient tolerated the treatment well and is undergoing routine disease monitoring. Following CAR T-cell therapy, his hypogammaglobulinemia persists with immunoglobulins level less than 500 mg/dL, and the veteran is still receiving supportive care IVIG.
Discussion
A population estimate of 1.3 million veterans are uninsured and can only access healthcare through the VA (Nelson et al, 2007). This case highlights the first patient to receive CAR T-cell therapy for multiple myeloma in the VA system, indicating that veterans now have access to this life-saving treatment. The rate of hypogammaglobulinemia following CAR T-cell therapy for multiple myeloma is as high as 41%, with an associated infection risk of 70%. Following CAR T-cell therapy with idecabtagene vicleucel, around 61% of patients will require IVIG treatment (Wat el al, 2021). Our case adds to this growing literature on the prevalence of IVIG treatment following CAR T-cell therapy in this patient population.
Background
In 3/2021, chimeric antigen receptor (CAR) T-cell therapy was approved for the treatment of multiple myeloma in adult patients with refractory disease. Currently, only the Veterans Affair (VA) center at the Tennessee Valley Healthcare System (TVHS) offers this treatment. Herein, we report a significant healthcare milestone in 2024 when the first patient received CAR T-cell therapy for multiple myeloma in the VA system. Additionally, the rate of hypogammaglobulinemia is the highest for CAR T-cell therapy using idecabtagene vicleucel compared to therapies using other antineoplastic agents (Wat et al, 2021). The complications of hypogammaglobulinemia can be mitigated by intravenous immunoglobulin (IVIG) treatment.
Case Presentation
A 75-year-old male veteran was diagnosed with IgA Kappa multiple myeloma and received induction therapy with bortezomib, lenalidomide, and dexamethasone in 2014. The patient underwent autologous stem cell transplant (SCT) in the same year. His disease recurred in 3/2019, and the patient was started on daratumumab and pomalidomide. He received another autologous SCT in 2/2021, to which he was refractory. The veteran then received treatment with daratumumab and ixazomib, followed by carfilzomib and cyclophosphamide. Starting in 9/2022, the patient also required regular IVIG treatment for hypogammaglobulinemia. He eventually received CAR T-cell therapy with idecabtagene vicleucel at THVS on 4/18/2024. The patient tolerated the treatment well and is undergoing routine disease monitoring. Following CAR T-cell therapy, his hypogammaglobulinemia persists with immunoglobulins level less than 500 mg/dL, and the veteran is still receiving supportive care IVIG.
Discussion
A population estimate of 1.3 million veterans are uninsured and can only access healthcare through the VA (Nelson et al, 2007). This case highlights the first patient to receive CAR T-cell therapy for multiple myeloma in the VA system, indicating that veterans now have access to this life-saving treatment. The rate of hypogammaglobulinemia following CAR T-cell therapy for multiple myeloma is as high as 41%, with an associated infection risk of 70%. Following CAR T-cell therapy with idecabtagene vicleucel, around 61% of patients will require IVIG treatment (Wat el al, 2021). Our case adds to this growing literature on the prevalence of IVIG treatment following CAR T-cell therapy in this patient population.
Background
In 3/2021, chimeric antigen receptor (CAR) T-cell therapy was approved for the treatment of multiple myeloma in adult patients with refractory disease. Currently, only the Veterans Affair (VA) center at the Tennessee Valley Healthcare System (TVHS) offers this treatment. Herein, we report a significant healthcare milestone in 2024 when the first patient received CAR T-cell therapy for multiple myeloma in the VA system. Additionally, the rate of hypogammaglobulinemia is the highest for CAR T-cell therapy using idecabtagene vicleucel compared to therapies using other antineoplastic agents (Wat et al, 2021). The complications of hypogammaglobulinemia can be mitigated by intravenous immunoglobulin (IVIG) treatment.
Case Presentation
A 75-year-old male veteran was diagnosed with IgA Kappa multiple myeloma and received induction therapy with bortezomib, lenalidomide, and dexamethasone in 2014. The patient underwent autologous stem cell transplant (SCT) in the same year. His disease recurred in 3/2019, and the patient was started on daratumumab and pomalidomide. He received another autologous SCT in 2/2021, to which he was refractory. The veteran then received treatment with daratumumab and ixazomib, followed by carfilzomib and cyclophosphamide. Starting in 9/2022, the patient also required regular IVIG treatment for hypogammaglobulinemia. He eventually received CAR T-cell therapy with idecabtagene vicleucel at THVS on 4/18/2024. The patient tolerated the treatment well and is undergoing routine disease monitoring. Following CAR T-cell therapy, his hypogammaglobulinemia persists with immunoglobulins level less than 500 mg/dL, and the veteran is still receiving supportive care IVIG.
Discussion
A population estimate of 1.3 million veterans are uninsured and can only access healthcare through the VA (Nelson et al, 2007). This case highlights the first patient to receive CAR T-cell therapy for multiple myeloma in the VA system, indicating that veterans now have access to this life-saving treatment. The rate of hypogammaglobulinemia following CAR T-cell therapy for multiple myeloma is as high as 41%, with an associated infection risk of 70%. Following CAR T-cell therapy with idecabtagene vicleucel, around 61% of patients will require IVIG treatment (Wat el al, 2021). Our case adds to this growing literature on the prevalence of IVIG treatment following CAR T-cell therapy in this patient population.
The emergence of postgraduate training programs for APPs in pulmonary and critical care
APP Intersection
Postgraduate training for advanced practice providers (APPs) has existed in one form or another since the genesis of the allied professions. They are typically referred to as residencies, fellowships, postgraduate programs, and transition-to-practice.
The desire and necessity for these programs has increased in the past decade with workforce changes; namely the increasing number of nurse practitioners (NPs) graduating with fewer years of experience at the bedside compared with previous eras, a similar decrease in patient contact hours for graduating PAs, the transition of physician colleagues from employers to employees and the subsequent change in priorities in training new graduate APPs, and resident work hour restrictions necessitating more APPs to staff inpatient units and work in various specialties.
The goal of these programs is to provide postgraduate training to physician assistants/associates (PAs) and NPs across myriad medical specialties to both newly graduated APPs and those looking to transition specialties. Current programs exist in family medicine, emergency medicine, urgent care, critical care medicine, pulmonary medicine, oncology, surgery, and various surgical subspecialties, to name a few. Program length is highly variable, though most programs advertise as lasting around 12 months, with varying ratios of clinical and didactic education. Postgraduate APP programs are largely advertise as salaried, benefitted positions, though usually at a rate below that of a so-called “direct hire” due to the protected learning time associated with the postgraduate training year.
Accreditation for these programs is still disjointed, although unifying efforts have been made as of late, and is currently available through the Advanced Practice Provider Fellowship Accreditation, Association of Postgraduate Physician Assistant Programs, ARC-PA, the Accreditation Commission for Education in Nursing, and the Consortium for Advanced Practice Providers. Other organizations, such as the Association of Post Graduate APRN Programs, host regular conferences to discuss the formulation of postgraduate APP education curricula and program development.
While accreditation offers guidance for fledgling programs, many utilize the standards published by the American College of Graduate Medical Education to ensure that appropriate clinical milestones are being met and that a common language among APPs and physicians who are involved in the evaluation of the postgraduate APP trainee is being used. Programs also seek to utilize other well-established curricula and certification programs published by various national and international organizations. A key distinction from physician postgraduate training is that there is currently no fiscal or legislative support for postgraduate APP programs; these issues have been cited as reasons for the limited scope and number of programs.
When starting APP Fellowship programs, it is important to consider why this would be beneficial to a specific division and health care organization. Usually, fellowship programs develop out of a need to train and retain APPs. It is no secret that turnover and retention of skilled APPs is a nationwide problem associated with significant costs to organizations. The ability to retain fellowship-trained APPs will result in cost savings due to the reduction in onboarding time and orientation costs, as these APP fellows finish their programs ready to be fully productive team members.
Additional considerations for the development of an APP fellowship include improving access to care and increasing the quality of the care provided. Fellowship programs encourage a smoother transition to practice by offering more support through education, closer evaluation, and frequent feedback, which improves competence and confidence of these providers. A supported APP is more likely to practice to the fullest extent of their license and have improved personal and professional satisfaction, leading to employee retention and better patient care.
When developing a budget for these types of programs, it is important to include the full-time equivalent (FTE) for the fellow, benefits, onboarding/licensure, simulations, and fellowship faculty costs.
Faculty compensation varies by institution but can include salary support, FTE reduction, and nonclinical appointments. Tracking metrics such as fellow billing, length of stay, and access to care during the fellowship year are helpful to highlight the benefit of these programs to the organization.
Initiating a program like those described may seem like a Herculean feat, but motivated individuals have been able to accomplish similar goals in both adequately and poorly resourced areas. For those aspiring to start a postgraduate APP program at their instruction, these authors suggest the following approach.
First, identify your institution’s need for such a program. Next, define your curriculum, evaluation process, and expectations. Then, create buy-in from stakeholders, including administrative and clinical personnel. Finally, focus on recruitment. Seeking accreditation may be challenging for new programs, but identifying the accreditation standard you plan to pursue early will pay dividends when the time comes for the program to apply. Those starting down this path should realistically expect an 18- to 24-month period between their first efforts and the start of the first class.
“APP Intersection” is a new quarterly column focusing on areas of interest for the entire chest medicine health care team.
APP Intersection
Postgraduate training for advanced practice providers (APPs) has existed in one form or another since the genesis of the allied professions. They are typically referred to as residencies, fellowships, postgraduate programs, and transition-to-practice.
The desire and necessity for these programs has increased in the past decade with workforce changes; namely the increasing number of nurse practitioners (NPs) graduating with fewer years of experience at the bedside compared with previous eras, a similar decrease in patient contact hours for graduating PAs, the transition of physician colleagues from employers to employees and the subsequent change in priorities in training new graduate APPs, and resident work hour restrictions necessitating more APPs to staff inpatient units and work in various specialties.
The goal of these programs is to provide postgraduate training to physician assistants/associates (PAs) and NPs across myriad medical specialties to both newly graduated APPs and those looking to transition specialties. Current programs exist in family medicine, emergency medicine, urgent care, critical care medicine, pulmonary medicine, oncology, surgery, and various surgical subspecialties, to name a few. Program length is highly variable, though most programs advertise as lasting around 12 months, with varying ratios of clinical and didactic education. Postgraduate APP programs are largely advertise as salaried, benefitted positions, though usually at a rate below that of a so-called “direct hire” due to the protected learning time associated with the postgraduate training year.
Accreditation for these programs is still disjointed, although unifying efforts have been made as of late, and is currently available through the Advanced Practice Provider Fellowship Accreditation, Association of Postgraduate Physician Assistant Programs, ARC-PA, the Accreditation Commission for Education in Nursing, and the Consortium for Advanced Practice Providers. Other organizations, such as the Association of Post Graduate APRN Programs, host regular conferences to discuss the formulation of postgraduate APP education curricula and program development.
While accreditation offers guidance for fledgling programs, many utilize the standards published by the American College of Graduate Medical Education to ensure that appropriate clinical milestones are being met and that a common language among APPs and physicians who are involved in the evaluation of the postgraduate APP trainee is being used. Programs also seek to utilize other well-established curricula and certification programs published by various national and international organizations. A key distinction from physician postgraduate training is that there is currently no fiscal or legislative support for postgraduate APP programs; these issues have been cited as reasons for the limited scope and number of programs.
When starting APP Fellowship programs, it is important to consider why this would be beneficial to a specific division and health care organization. Usually, fellowship programs develop out of a need to train and retain APPs. It is no secret that turnover and retention of skilled APPs is a nationwide problem associated with significant costs to organizations. The ability to retain fellowship-trained APPs will result in cost savings due to the reduction in onboarding time and orientation costs, as these APP fellows finish their programs ready to be fully productive team members.
Additional considerations for the development of an APP fellowship include improving access to care and increasing the quality of the care provided. Fellowship programs encourage a smoother transition to practice by offering more support through education, closer evaluation, and frequent feedback, which improves competence and confidence of these providers. A supported APP is more likely to practice to the fullest extent of their license and have improved personal and professional satisfaction, leading to employee retention and better patient care.
When developing a budget for these types of programs, it is important to include the full-time equivalent (FTE) for the fellow, benefits, onboarding/licensure, simulations, and fellowship faculty costs.
Faculty compensation varies by institution but can include salary support, FTE reduction, and nonclinical appointments. Tracking metrics such as fellow billing, length of stay, and access to care during the fellowship year are helpful to highlight the benefit of these programs to the organization.
Initiating a program like those described may seem like a Herculean feat, but motivated individuals have been able to accomplish similar goals in both adequately and poorly resourced areas. For those aspiring to start a postgraduate APP program at their instruction, these authors suggest the following approach.
First, identify your institution’s need for such a program. Next, define your curriculum, evaluation process, and expectations. Then, create buy-in from stakeholders, including administrative and clinical personnel. Finally, focus on recruitment. Seeking accreditation may be challenging for new programs, but identifying the accreditation standard you plan to pursue early will pay dividends when the time comes for the program to apply. Those starting down this path should realistically expect an 18- to 24-month period between their first efforts and the start of the first class.
“APP Intersection” is a new quarterly column focusing on areas of interest for the entire chest medicine health care team.
APP Intersection
Postgraduate training for advanced practice providers (APPs) has existed in one form or another since the genesis of the allied professions. They are typically referred to as residencies, fellowships, postgraduate programs, and transition-to-practice.
The desire and necessity for these programs has increased in the past decade with workforce changes; namely the increasing number of nurse practitioners (NPs) graduating with fewer years of experience at the bedside compared with previous eras, a similar decrease in patient contact hours for graduating PAs, the transition of physician colleagues from employers to employees and the subsequent change in priorities in training new graduate APPs, and resident work hour restrictions necessitating more APPs to staff inpatient units and work in various specialties.
The goal of these programs is to provide postgraduate training to physician assistants/associates (PAs) and NPs across myriad medical specialties to both newly graduated APPs and those looking to transition specialties. Current programs exist in family medicine, emergency medicine, urgent care, critical care medicine, pulmonary medicine, oncology, surgery, and various surgical subspecialties, to name a few. Program length is highly variable, though most programs advertise as lasting around 12 months, with varying ratios of clinical and didactic education. Postgraduate APP programs are largely advertise as salaried, benefitted positions, though usually at a rate below that of a so-called “direct hire” due to the protected learning time associated with the postgraduate training year.
Accreditation for these programs is still disjointed, although unifying efforts have been made as of late, and is currently available through the Advanced Practice Provider Fellowship Accreditation, Association of Postgraduate Physician Assistant Programs, ARC-PA, the Accreditation Commission for Education in Nursing, and the Consortium for Advanced Practice Providers. Other organizations, such as the Association of Post Graduate APRN Programs, host regular conferences to discuss the formulation of postgraduate APP education curricula and program development.
While accreditation offers guidance for fledgling programs, many utilize the standards published by the American College of Graduate Medical Education to ensure that appropriate clinical milestones are being met and that a common language among APPs and physicians who are involved in the evaluation of the postgraduate APP trainee is being used. Programs also seek to utilize other well-established curricula and certification programs published by various national and international organizations. A key distinction from physician postgraduate training is that there is currently no fiscal or legislative support for postgraduate APP programs; these issues have been cited as reasons for the limited scope and number of programs.
When starting APP Fellowship programs, it is important to consider why this would be beneficial to a specific division and health care organization. Usually, fellowship programs develop out of a need to train and retain APPs. It is no secret that turnover and retention of skilled APPs is a nationwide problem associated with significant costs to organizations. The ability to retain fellowship-trained APPs will result in cost savings due to the reduction in onboarding time and orientation costs, as these APP fellows finish their programs ready to be fully productive team members.
Additional considerations for the development of an APP fellowship include improving access to care and increasing the quality of the care provided. Fellowship programs encourage a smoother transition to practice by offering more support through education, closer evaluation, and frequent feedback, which improves competence and confidence of these providers. A supported APP is more likely to practice to the fullest extent of their license and have improved personal and professional satisfaction, leading to employee retention and better patient care.
When developing a budget for these types of programs, it is important to include the full-time equivalent (FTE) for the fellow, benefits, onboarding/licensure, simulations, and fellowship faculty costs.
Faculty compensation varies by institution but can include salary support, FTE reduction, and nonclinical appointments. Tracking metrics such as fellow billing, length of stay, and access to care during the fellowship year are helpful to highlight the benefit of these programs to the organization.
Initiating a program like those described may seem like a Herculean feat, but motivated individuals have been able to accomplish similar goals in both adequately and poorly resourced areas. For those aspiring to start a postgraduate APP program at their instruction, these authors suggest the following approach.
First, identify your institution’s need for such a program. Next, define your curriculum, evaluation process, and expectations. Then, create buy-in from stakeholders, including administrative and clinical personnel. Finally, focus on recruitment. Seeking accreditation may be challenging for new programs, but identifying the accreditation standard you plan to pursue early will pay dividends when the time comes for the program to apply. Those starting down this path should realistically expect an 18- to 24-month period between their first efforts and the start of the first class.
“APP Intersection” is a new quarterly column focusing on areas of interest for the entire chest medicine health care team.
A Learning Health System Approach to Long COVID Care
The Veterans Health Administration (VHA)—along with systems across the world—has spent the past 2 years continuously adapting to meet the emerging needs of persons infected with COVID-19. With the development of effective vaccines and global efforts to mitigate transmission, attention has now shifted to long COVID care as the need for further outpatient health care becomes increasingly apparent.1,2
Background
Multiple terms describe the lingering, multisystem sequelae of COVID-19 that last longer than 4 weeks: long COVID, postacute COVID-19 syndrome, post-COVID condition, postacute sequalae of COVID-19, and COVID long hauler.1,3 Common symptoms include fatigue, shortness of breath, cough, sleep disorders, brain fog or cognitive dysfunction, depression, anxiety, pain, and changes in taste or smell that impact a person’s functioning.4,5 The multisystem nature of the postacute course of COVID-19 necessitates an interdisciplinary approach to devise comprehensive and individualized care plans.6-9 Research is needed to better understand this postacute state (eg, prevalence, underlying effects, characteristics of those who experience long COVID) to establish and evaluate cost-effective treatment approaches.
Many patients who are experiencing symptoms beyond the acute course of COVID-19 have been referred to general outpatient clinics or home health, which may lack the capacity and knowledge of this novel disease to effectively manage complex long COVID cases.2,3 To address this growing need, clinicians and leadership across a variety of disciplines and settings in the VHA created a community of practice (CoP) to create a mechanism for cross-facility communication, identify gaps in long COVID care and research, and cocreate knowledge on best practices for care delivery.
In this spirit, we are embracing a learning health system (LHS) approach that uses rapid-cycle methods to integrate data and real-world experience to iteratively evaluate and adapt models of long COVID care.10 Our clinically identified and data-driven objective is to provide high value health care to patients with long COVID sequalae by creating a framework to learn about this novel condition and develop innovative care models. This article provides an overview of our emerging LHS approach to the study of long COVID care that is fostering innovation and adaptability within the VHA. We describe 3 aspects of our engagement approach central to LHS: the ongoing development of a long COVID CoP dedicated to iteratively informing the bidirectional cycle of data from practice to research, results of a broad environmental scan of VHA long COVID care, and results of a survey administered to CoP members to inform ongoing needs of the community and identify early successful outcomes from participation.
Learning Health System Approach
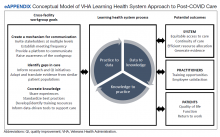
The VHA is one of the largest integrated health care systems in the United States serving more than 9 million veterans.11 Since 2017, the VHA has articulated a vision to become an LHS that informs and improves patient-centered care through practice-based and data-driven research (eAppendix).12 During the early COVID-19 pandemic, an LHS approach in the VHA was critical to rapidly establishing a data infrastructure for disease surveillance, coordinating data-driven solutions, leveraging use of technology, collaborating across the globe to identify best practices, and implementing systematic responses (eg, policies, workforce adjustments).
Our long COVID CoP was developed as clinical observations and ongoing conversations with stakeholders (eg, veterans, health care practitioners [HCPs], leadership) identified a need to effectively identify and treat the growing number of veterans with long COVID. This clinical issue is compounded by the limited but emerging evidence on the clinical presentation of prolonged COVID-19 symptoms, treatment, and subsequent care pathways. The VHA’s efforts and lessons learned within the lens of an LHS are applicable to other systems confronting the complex identification and management of patients with persistent and encumbering long COVID symptoms. The VHA is building upon the LHS approach to proactively prepare for and address future clinical or public health challenges that require cross-system and sector collaborations, expediency, inclusivity, and patient/family centeredness.11
Community of Practice
As of January 25, 2022, our workgroup consisted of 128 VHA employees representing 29 VHA medical centers. Members of the multidisciplinary workgroup have diverse backgrounds with HCPs from primary care (eg, physicians, nurse practitioners), rehabilitation (eg, physical therapists), specialty care (eg, pulmonologists, physiatrists), mental health (eg, psychologists), and complementary and integrated health/Whole Health services (eg, practitoners of services such as yoga, tai chi, mindfulness, acupuncture). Members also include clinical, operations, and research leadership at local, regional, and national VHA levels. Our first objective as a large, diverse group was to establish shared goals, which included: (1) determining efficient communication pathways; (2) identifying gaps in care or research; and (3) cocreating knowledge to provide solutions to identified gaps.
Communication Mechanisms
Our first goal was to create an efficient mechanism for cross-facility communication. The initial CoP was formed in April 2021 and the first virtual meeting focused on reaching a consensus regarding the best way to communicate and proceed. We agreed to convene weekly at a consistent time, created a standard agenda template, and elected a lead facilitator of meeting proceedings. In addition, a member of the CoP recorded and took extensive meeting notes, which were later distributed to the entire CoP to accommodate varying schedules and ability to attend live meetings. Approximately 20 to 30 participants attend the meetings in real-time.
To consolidate working documents, information, and resources in one location, we created a platform to communicate via a Microsoft Teams channel. All CoP members are given access to the folders and allowed to add to the growing library of resources. Resources include clinical assessment and note templates for electronic documentation of care, site-specific process maps, relevant literature on screening and interventions identified by practice members, and meeting notes along with the recordings. A chat feature alerts CoP members to questions posed by other members. Any resources or information shared on the chat discussion are curated by CoP leaders to disseminate to all members. Importantly, this platform allowed us to communicate efficiently within the VHA organization by creating a centralized space for documents and the ability to correspond with all or select members of the CoP. Additional VHA employees can easily be referred and request access.
To increase awareness of the CoP, expand reach, and diversify perspectives, every participant was encouraged to invite colleagues and stakeholders with interest or experience in long COVID care to join. While patients are not included in this CoP, we are working closely with the VHA user experience workgroup (many members overlap) that is gathering patient and caregiver perspectives on their COVID-19 experience and long COVID care. Concurrently, CoP members and leadership facilitate communication and set up formal collaborations with other non-VHA health care systems to create an intersystem network of collaboration for long COVID care. This approach further enhances the speed at which we can work together to share lessons learned and stay up-to-date on emerging evidence surrounding long COVID care.
Identifying Gaps in Care and Research
Our second goal was to identify gaps in care or knowledge to inform future research and quality improvement initiatives, while also creating a foundation to cocreate knowledge about safe, effective care management of the novel long COVID sequelae. To translate knowledge, we must first identify and understand the gaps between the current, best available evidence and current care practices or policies impacting that delivery.13 As such, the structured meeting agenda and facilitated meeting discussions focused on understanding current clinical decision making and the evidence base. We shared VHA evidence synthesis reports and living rapid reviews on complications following COVID-19 illness (ie, major organ damage and posthospitalization health care use) that provided an objective evidence base on common long COVID complications.14,15
Since long COVID is a novel condition, we drew from literature in similar patient populations and translated that information in the context of our current knowledge of this unique syndrome. For example, we discussed the predominant and persistent symptom of fatigue post-COVID.5 In particular, the CoP discussed challenges in identifying and treating post-COVID fatigue, which is often a vague symptom with multiple or interacting etiologies that require a comprehensive, interdisciplinary approach. As such, we reviewed, adapted, and translated identification and treatment strategies from the literature on chronic fatigue syndrome to patients with post-COVID syndrome.16,17 We continue to work collaboratively and engage the appropriate stakeholders to provide input on the gaps to prioritize targeting.
Cocreate Knowledge
Our third goal was to cocreate knowledge regarding the care of patients with long COVID. To accomplish this, our structured meetings and communication pathways invited members to share experiences on the who (delivers and receives care), what (type of care or HCPs), when (identification of post-COVID and access), and how (eg, telehealth) of care to patients post-COVID. As part of the workgroup, we identified and shared resources on standardized, facility-level practices to reduce variability across the VHA system. These resources included intake/assessment forms, care processes, and batteries of tests/measures used for screening and assessment. The knowledge obtained from outside the CoP and cocreated within is being used to inform data-driven tools to support and evaluate care for patients with long COVID. As such, members of the workgroup are in the formative stages of participating in quality improvement innovation pilots to test technologies and processes designed to improve and validate long COVID care pathways. These technologies include screening tools, clinical decision support tools, and population health management technologies. In addition, we are developing a formal collaboration with the VHA Office of Research and Development to create standardized intake forms across VHA long COVID clinics to facilitate both clinical monitoring and research.
Surveys
The US Department of Veterans Affairs Central Office collaborated with our workgroup to draft an initial set of survey questions designed to understand how each VHA facility defines, identifies, and provides care to veterans experiencing post-COVID sequalae. The 41-question survey was distributed through regional directors and chief medical officers at 139 VHA facilities in August 2021. One hundred nineteen responses (86%) were received. Sixteen facilities indicated they had established programs and 26 facilities were considering a program. Our CoP had representation from the 16 facilities with established programs indicating the deep and well-connected nature of our grassroots efforts to bring together stakeholders to learn as part of a CoP.
A separate, follow-up survey generated responses from 18 facilities and identified the need to capture evolving innovations and to develop smaller workstreams (eg, best practices, electronic documentation templates, pathway for referrals, veteran engagement, outcome measures). The survey not only exposed ongoing challenges to providing long COVID care, but importantly, outlined the ways in which CoP members were leveraging community knowledge and resources to inform innovations and processes of care changes at their specific sites. Fourteen of 18 facilities with long COVID programs in place explicitly identified the CoP as a resource they have found most beneficial when employing such innovations. Specific innovations reported included changes in care delivery, engagement in active outreach with veterans and local facility, and infrastructure development to sustain local long COVID clinics (Table).
Future Directions
Our CoP strives to contribute to an evidence base for long COVID care. At the system level, the CoP has the potential to impact access and continuity of care by identifying appropriate processes and ensuring that VHA patients receive outreach and an opportunity for post-COVID care. Comprehensive care requires input from HCP, clinical leadership, and operations levels. In this sense, our CoP provides an opportunity for diverse stakeholders to come together, discuss barriers to screening and delivering post-COVID care, and create an action plan to remove or lessen such barriers.18 Part of the process to remove barriers is to identify and support efficient resource allocation. Our CoP has worked to address issues in resource allocation (eg, space, personnel) for post-COVID care. For example, one facility is currently implementing interdisciplinary virtual post-COVID care. Another facility identified and restructured working assignments for psychologists who served in different capacities throughout the system to fill the need within the long COVID team.
At the HCP level, the CoP is currently developing workshops, media campaigns, written clinical resources, skills training, publications, and webinars/seminars with continuing medical education credits.19 The CoP may also provide learning and growth opportunities, such as clinical or VHA operational fellowships and research grants.
We are still in the formative stages of post-COVID care and future efforts will explore patient-centered outcomes. We are drawing on the Centers for Disease Control and Prevention’s guidance for evaluating patients with long COVID symptoms and examining the feasibility within VHA, as well as patient perspectives on post-COVID sequalae, to ensure we are selecting assessments that measure patient-centered constructs.18
Conclusions
A VHA-wide LHS approach is identifying issues related to the identification, delivery, and evaluation of long COVID care. This long COVID CoP has developed an infrastructure for communication, identified gaps in care, and cocreated knowledge related to best current practices for post-COVID care. This work is contributing to systemwide LHS efforts dedicated to creating a culture of quality care and innovation and is a process that is transferrable to other areas of care in the VHA, as well as other health care systems. The LHS approach continues to be highly relevant as we persist through the COVID-19 pandemic and reimagine a postpandemic world.
Acknowledg
We thank all the members of the Veterans Health Administration long COVID Community of Practice who participate in the meetings and contribute to the sharing and spread of knowledge.
1. Sivan M, Halpin S, Hollingworth L, Snook N, Hickman K, Clifton I. Development of an integrated rehabilitation pathway for individuals recovering from COVID-19 in the community. J Rehabil Med. 2020;52(8):jrm00089. doi:10.2340/16501977-2727
2. Understanding the long-term health effects of COVID-19. EClinicalMedicine. 2020;26:100586. doi:10.1016/j.eclinm.2020.100586
3. Greenhalgh T, Knight M, A’Court C, Buxton M, Husain L. Management of post-acute covid-19 in primary care. BMJ. Published online August 11, 2020:m3026. doi:10.1136/bmj.m3026
4. Iwua CJ, Iwu CD, Wiysonge CS. The occurrence of long COVID: a rapid review. Pan Afr Med J. 2021;38. doi:10.11604/pamj.2021.38.65.27366
5. Carfì A, Bernabei R, Landi F; Gemelli Against COVID-19 Post-Acute Care Study Group. Persistent symptoms in patients after acute COVID-19. JAMA. 2020;324(6):603-605. doi:10.1001/jama.2020.12603
6. Gemelli Against COVID-19 Post-Acute Care Study Group. Post-COVID-19 global health strategies: the need for an interdisciplinary approach. Aging Clin Exp Res. 2020;32(8):1613-1620. doi:10.1007/s40520-020-01616-x
7. Xie Y, Xu E, Bowe B, Al-Aly Z. Long-term cardiovascular outcomes of COVID-19. Nat Med. 2022;28:583-590. doi:10.1038/s41591-022-01689-3
8. Al-Aly Z, Xie Y, Bowe B. High-dimensional characterization of post-acute sequelae of COVID-19. Nature. 2021;594:259-264. doi:10.1038/s41586-021-03553-9
9. Ayoubkhani D, Bermingham C, Pouwels KB, et al. Trajectory of long covid symptoms after covid-19 vaccination: community based cohort study. BMJ. 2022;377:e069676. doi:10.1136/bmj-2021-069676
10. Institute of Medicine (US) Roundtable on Evidence-Based Medicine, Olsen L, Aisner D, McGinnis JM, eds. The Learning Healthcare System: Workshop Summary. Washington (DC): National Academies Press (US); 2007. doi:10.17226/11903
11. Romanelli RJ, Azar KMJ, Sudat S, Hung D, Frosch DL, Pressman AR. Learning health system in crisis: lessons from the COVID-19 pandemic. Mayo Clin Proc Innov Qual Outcomes. 2021;5(1):171-176. doi:10.1016/j.mayocpiqo.2020.10.004
12. Atkins D, Kilbourne AM, Shulkin D. Moving from discovery to system-wide change: the role of research in a learning health care system: experience from three decades of health systems research in the Veterans Health Administration. Annu Rev Public Health. 2017;38:467-487. doi:10.1146/annurev-publhealth-031816-044255
13. Kitson A, Straus SE. The knowledge-to-action cycle: identifying the gaps. CMAJ. 2010;182(2):E73-77. doi:10.1503/cmaj.081231
14. Greer N, Bart B, Billington C, et al. COVID-19 post-acute care major organ damage: a living rapid review. Updated September 2021. Accessed May 31, 2022. https://www.hsrd.research.va.gov/publications/esp/covid-organ-damage.pdf
15. Sharpe JA, Burke C, Gordon AM, et al. COVID-19 post-hospitalization health care utilization: a living review. Updated February 2022. Accessed May 31, 2022. https://www.hsrd.research.va.gov/publications/esp/covid19-post-hosp.pdf
16. Bested AC, Marshall LM. Review of Myalgic Encephalomyelitis/chronic fatigue syndrome: an evidence-based approach to diagnosis and management by clinicians. Rev Environ Health. 2015;30(4):223-249. doi:10.1515/reveh-2015-0026
17. Yancey JR, Thomas SM. Chronic fatigue syndrome: diagnosis and treatment. Am Fam Physician. 2012;86(8):741-746.
18. Kotter JP, Cohen DS. Change Leadership The Kotter Collection. Harvard Business Review Press; 2014.
19. Brownson RC, Eyler AA, Harris JK, Moore JB, Tabak RG. Getting the word out: new approaches for disseminating public health science. J Public Health Manag Pract. 2018;24(2):102-111. doi:10.1097/PHH.0000000000000673
The Veterans Health Administration (VHA)—along with systems across the world—has spent the past 2 years continuously adapting to meet the emerging needs of persons infected with COVID-19. With the development of effective vaccines and global efforts to mitigate transmission, attention has now shifted to long COVID care as the need for further outpatient health care becomes increasingly apparent.1,2
Background
Multiple terms describe the lingering, multisystem sequelae of COVID-19 that last longer than 4 weeks: long COVID, postacute COVID-19 syndrome, post-COVID condition, postacute sequalae of COVID-19, and COVID long hauler.1,3 Common symptoms include fatigue, shortness of breath, cough, sleep disorders, brain fog or cognitive dysfunction, depression, anxiety, pain, and changes in taste or smell that impact a person’s functioning.4,5 The multisystem nature of the postacute course of COVID-19 necessitates an interdisciplinary approach to devise comprehensive and individualized care plans.6-9 Research is needed to better understand this postacute state (eg, prevalence, underlying effects, characteristics of those who experience long COVID) to establish and evaluate cost-effective treatment approaches.
Many patients who are experiencing symptoms beyond the acute course of COVID-19 have been referred to general outpatient clinics or home health, which may lack the capacity and knowledge of this novel disease to effectively manage complex long COVID cases.2,3 To address this growing need, clinicians and leadership across a variety of disciplines and settings in the VHA created a community of practice (CoP) to create a mechanism for cross-facility communication, identify gaps in long COVID care and research, and cocreate knowledge on best practices for care delivery.
In this spirit, we are embracing a learning health system (LHS) approach that uses rapid-cycle methods to integrate data and real-world experience to iteratively evaluate and adapt models of long COVID care.10 Our clinically identified and data-driven objective is to provide high value health care to patients with long COVID sequalae by creating a framework to learn about this novel condition and develop innovative care models. This article provides an overview of our emerging LHS approach to the study of long COVID care that is fostering innovation and adaptability within the VHA. We describe 3 aspects of our engagement approach central to LHS: the ongoing development of a long COVID CoP dedicated to iteratively informing the bidirectional cycle of data from practice to research, results of a broad environmental scan of VHA long COVID care, and results of a survey administered to CoP members to inform ongoing needs of the community and identify early successful outcomes from participation.
Learning Health System Approach
The VHA is one of the largest integrated health care systems in the United States serving more than 9 million veterans.11 Since 2017, the VHA has articulated a vision to become an LHS that informs and improves patient-centered care through practice-based and data-driven research (eAppendix).12 During the early COVID-19 pandemic, an LHS approach in the VHA was critical to rapidly establishing a data infrastructure for disease surveillance, coordinating data-driven solutions, leveraging use of technology, collaborating across the globe to identify best practices, and implementing systematic responses (eg, policies, workforce adjustments).
Our long COVID CoP was developed as clinical observations and ongoing conversations with stakeholders (eg, veterans, health care practitioners [HCPs], leadership) identified a need to effectively identify and treat the growing number of veterans with long COVID. This clinical issue is compounded by the limited but emerging evidence on the clinical presentation of prolonged COVID-19 symptoms, treatment, and subsequent care pathways. The VHA’s efforts and lessons learned within the lens of an LHS are applicable to other systems confronting the complex identification and management of patients with persistent and encumbering long COVID symptoms. The VHA is building upon the LHS approach to proactively prepare for and address future clinical or public health challenges that require cross-system and sector collaborations, expediency, inclusivity, and patient/family centeredness.11
Community of Practice
As of January 25, 2022, our workgroup consisted of 128 VHA employees representing 29 VHA medical centers. Members of the multidisciplinary workgroup have diverse backgrounds with HCPs from primary care (eg, physicians, nurse practitioners), rehabilitation (eg, physical therapists), specialty care (eg, pulmonologists, physiatrists), mental health (eg, psychologists), and complementary and integrated health/Whole Health services (eg, practitoners of services such as yoga, tai chi, mindfulness, acupuncture). Members also include clinical, operations, and research leadership at local, regional, and national VHA levels. Our first objective as a large, diverse group was to establish shared goals, which included: (1) determining efficient communication pathways; (2) identifying gaps in care or research; and (3) cocreating knowledge to provide solutions to identified gaps.
Communication Mechanisms
Our first goal was to create an efficient mechanism for cross-facility communication. The initial CoP was formed in April 2021 and the first virtual meeting focused on reaching a consensus regarding the best way to communicate and proceed. We agreed to convene weekly at a consistent time, created a standard agenda template, and elected a lead facilitator of meeting proceedings. In addition, a member of the CoP recorded and took extensive meeting notes, which were later distributed to the entire CoP to accommodate varying schedules and ability to attend live meetings. Approximately 20 to 30 participants attend the meetings in real-time.
To consolidate working documents, information, and resources in one location, we created a platform to communicate via a Microsoft Teams channel. All CoP members are given access to the folders and allowed to add to the growing library of resources. Resources include clinical assessment and note templates for electronic documentation of care, site-specific process maps, relevant literature on screening and interventions identified by practice members, and meeting notes along with the recordings. A chat feature alerts CoP members to questions posed by other members. Any resources or information shared on the chat discussion are curated by CoP leaders to disseminate to all members. Importantly, this platform allowed us to communicate efficiently within the VHA organization by creating a centralized space for documents and the ability to correspond with all or select members of the CoP. Additional VHA employees can easily be referred and request access.
To increase awareness of the CoP, expand reach, and diversify perspectives, every participant was encouraged to invite colleagues and stakeholders with interest or experience in long COVID care to join. While patients are not included in this CoP, we are working closely with the VHA user experience workgroup (many members overlap) that is gathering patient and caregiver perspectives on their COVID-19 experience and long COVID care. Concurrently, CoP members and leadership facilitate communication and set up formal collaborations with other non-VHA health care systems to create an intersystem network of collaboration for long COVID care. This approach further enhances the speed at which we can work together to share lessons learned and stay up-to-date on emerging evidence surrounding long COVID care.
Identifying Gaps in Care and Research
Our second goal was to identify gaps in care or knowledge to inform future research and quality improvement initiatives, while also creating a foundation to cocreate knowledge about safe, effective care management of the novel long COVID sequelae. To translate knowledge, we must first identify and understand the gaps between the current, best available evidence and current care practices or policies impacting that delivery.13 As such, the structured meeting agenda and facilitated meeting discussions focused on understanding current clinical decision making and the evidence base. We shared VHA evidence synthesis reports and living rapid reviews on complications following COVID-19 illness (ie, major organ damage and posthospitalization health care use) that provided an objective evidence base on common long COVID complications.14,15
Since long COVID is a novel condition, we drew from literature in similar patient populations and translated that information in the context of our current knowledge of this unique syndrome. For example, we discussed the predominant and persistent symptom of fatigue post-COVID.5 In particular, the CoP discussed challenges in identifying and treating post-COVID fatigue, which is often a vague symptom with multiple or interacting etiologies that require a comprehensive, interdisciplinary approach. As such, we reviewed, adapted, and translated identification and treatment strategies from the literature on chronic fatigue syndrome to patients with post-COVID syndrome.16,17 We continue to work collaboratively and engage the appropriate stakeholders to provide input on the gaps to prioritize targeting.
Cocreate Knowledge
Our third goal was to cocreate knowledge regarding the care of patients with long COVID. To accomplish this, our structured meetings and communication pathways invited members to share experiences on the who (delivers and receives care), what (type of care or HCPs), when (identification of post-COVID and access), and how (eg, telehealth) of care to patients post-COVID. As part of the workgroup, we identified and shared resources on standardized, facility-level practices to reduce variability across the VHA system. These resources included intake/assessment forms, care processes, and batteries of tests/measures used for screening and assessment. The knowledge obtained from outside the CoP and cocreated within is being used to inform data-driven tools to support and evaluate care for patients with long COVID. As such, members of the workgroup are in the formative stages of participating in quality improvement innovation pilots to test technologies and processes designed to improve and validate long COVID care pathways. These technologies include screening tools, clinical decision support tools, and population health management technologies. In addition, we are developing a formal collaboration with the VHA Office of Research and Development to create standardized intake forms across VHA long COVID clinics to facilitate both clinical monitoring and research.
Surveys
The US Department of Veterans Affairs Central Office collaborated with our workgroup to draft an initial set of survey questions designed to understand how each VHA facility defines, identifies, and provides care to veterans experiencing post-COVID sequalae. The 41-question survey was distributed through regional directors and chief medical officers at 139 VHA facilities in August 2021. One hundred nineteen responses (86%) were received. Sixteen facilities indicated they had established programs and 26 facilities were considering a program. Our CoP had representation from the 16 facilities with established programs indicating the deep and well-connected nature of our grassroots efforts to bring together stakeholders to learn as part of a CoP.
A separate, follow-up survey generated responses from 18 facilities and identified the need to capture evolving innovations and to develop smaller workstreams (eg, best practices, electronic documentation templates, pathway for referrals, veteran engagement, outcome measures). The survey not only exposed ongoing challenges to providing long COVID care, but importantly, outlined the ways in which CoP members were leveraging community knowledge and resources to inform innovations and processes of care changes at their specific sites. Fourteen of 18 facilities with long COVID programs in place explicitly identified the CoP as a resource they have found most beneficial when employing such innovations. Specific innovations reported included changes in care delivery, engagement in active outreach with veterans and local facility, and infrastructure development to sustain local long COVID clinics (Table).
Future Directions
Our CoP strives to contribute to an evidence base for long COVID care. At the system level, the CoP has the potential to impact access and continuity of care by identifying appropriate processes and ensuring that VHA patients receive outreach and an opportunity for post-COVID care. Comprehensive care requires input from HCP, clinical leadership, and operations levels. In this sense, our CoP provides an opportunity for diverse stakeholders to come together, discuss barriers to screening and delivering post-COVID care, and create an action plan to remove or lessen such barriers.18 Part of the process to remove barriers is to identify and support efficient resource allocation. Our CoP has worked to address issues in resource allocation (eg, space, personnel) for post-COVID care. For example, one facility is currently implementing interdisciplinary virtual post-COVID care. Another facility identified and restructured working assignments for psychologists who served in different capacities throughout the system to fill the need within the long COVID team.
At the HCP level, the CoP is currently developing workshops, media campaigns, written clinical resources, skills training, publications, and webinars/seminars with continuing medical education credits.19 The CoP may also provide learning and growth opportunities, such as clinical or VHA operational fellowships and research grants.
We are still in the formative stages of post-COVID care and future efforts will explore patient-centered outcomes. We are drawing on the Centers for Disease Control and Prevention’s guidance for evaluating patients with long COVID symptoms and examining the feasibility within VHA, as well as patient perspectives on post-COVID sequalae, to ensure we are selecting assessments that measure patient-centered constructs.18
Conclusions
A VHA-wide LHS approach is identifying issues related to the identification, delivery, and evaluation of long COVID care. This long COVID CoP has developed an infrastructure for communication, identified gaps in care, and cocreated knowledge related to best current practices for post-COVID care. This work is contributing to systemwide LHS efforts dedicated to creating a culture of quality care and innovation and is a process that is transferrable to other areas of care in the VHA, as well as other health care systems. The LHS approach continues to be highly relevant as we persist through the COVID-19 pandemic and reimagine a postpandemic world.
Acknowledg
We thank all the members of the Veterans Health Administration long COVID Community of Practice who participate in the meetings and contribute to the sharing and spread of knowledge.
The Veterans Health Administration (VHA)—along with systems across the world—has spent the past 2 years continuously adapting to meet the emerging needs of persons infected with COVID-19. With the development of effective vaccines and global efforts to mitigate transmission, attention has now shifted to long COVID care as the need for further outpatient health care becomes increasingly apparent.1,2
Background
Multiple terms describe the lingering, multisystem sequelae of COVID-19 that last longer than 4 weeks: long COVID, postacute COVID-19 syndrome, post-COVID condition, postacute sequalae of COVID-19, and COVID long hauler.1,3 Common symptoms include fatigue, shortness of breath, cough, sleep disorders, brain fog or cognitive dysfunction, depression, anxiety, pain, and changes in taste or smell that impact a person’s functioning.4,5 The multisystem nature of the postacute course of COVID-19 necessitates an interdisciplinary approach to devise comprehensive and individualized care plans.6-9 Research is needed to better understand this postacute state (eg, prevalence, underlying effects, characteristics of those who experience long COVID) to establish and evaluate cost-effective treatment approaches.
Many patients who are experiencing symptoms beyond the acute course of COVID-19 have been referred to general outpatient clinics or home health, which may lack the capacity and knowledge of this novel disease to effectively manage complex long COVID cases.2,3 To address this growing need, clinicians and leadership across a variety of disciplines and settings in the VHA created a community of practice (CoP) to create a mechanism for cross-facility communication, identify gaps in long COVID care and research, and cocreate knowledge on best practices for care delivery.
In this spirit, we are embracing a learning health system (LHS) approach that uses rapid-cycle methods to integrate data and real-world experience to iteratively evaluate and adapt models of long COVID care.10 Our clinically identified and data-driven objective is to provide high value health care to patients with long COVID sequalae by creating a framework to learn about this novel condition and develop innovative care models. This article provides an overview of our emerging LHS approach to the study of long COVID care that is fostering innovation and adaptability within the VHA. We describe 3 aspects of our engagement approach central to LHS: the ongoing development of a long COVID CoP dedicated to iteratively informing the bidirectional cycle of data from practice to research, results of a broad environmental scan of VHA long COVID care, and results of a survey administered to CoP members to inform ongoing needs of the community and identify early successful outcomes from participation.
Learning Health System Approach
The VHA is one of the largest integrated health care systems in the United States serving more than 9 million veterans.11 Since 2017, the VHA has articulated a vision to become an LHS that informs and improves patient-centered care through practice-based and data-driven research (eAppendix).12 During the early COVID-19 pandemic, an LHS approach in the VHA was critical to rapidly establishing a data infrastructure for disease surveillance, coordinating data-driven solutions, leveraging use of technology, collaborating across the globe to identify best practices, and implementing systematic responses (eg, policies, workforce adjustments).
Our long COVID CoP was developed as clinical observations and ongoing conversations with stakeholders (eg, veterans, health care practitioners [HCPs], leadership) identified a need to effectively identify and treat the growing number of veterans with long COVID. This clinical issue is compounded by the limited but emerging evidence on the clinical presentation of prolonged COVID-19 symptoms, treatment, and subsequent care pathways. The VHA’s efforts and lessons learned within the lens of an LHS are applicable to other systems confronting the complex identification and management of patients with persistent and encumbering long COVID symptoms. The VHA is building upon the LHS approach to proactively prepare for and address future clinical or public health challenges that require cross-system and sector collaborations, expediency, inclusivity, and patient/family centeredness.11
Community of Practice
As of January 25, 2022, our workgroup consisted of 128 VHA employees representing 29 VHA medical centers. Members of the multidisciplinary workgroup have diverse backgrounds with HCPs from primary care (eg, physicians, nurse practitioners), rehabilitation (eg, physical therapists), specialty care (eg, pulmonologists, physiatrists), mental health (eg, psychologists), and complementary and integrated health/Whole Health services (eg, practitoners of services such as yoga, tai chi, mindfulness, acupuncture). Members also include clinical, operations, and research leadership at local, regional, and national VHA levels. Our first objective as a large, diverse group was to establish shared goals, which included: (1) determining efficient communication pathways; (2) identifying gaps in care or research; and (3) cocreating knowledge to provide solutions to identified gaps.
Communication Mechanisms
Our first goal was to create an efficient mechanism for cross-facility communication. The initial CoP was formed in April 2021 and the first virtual meeting focused on reaching a consensus regarding the best way to communicate and proceed. We agreed to convene weekly at a consistent time, created a standard agenda template, and elected a lead facilitator of meeting proceedings. In addition, a member of the CoP recorded and took extensive meeting notes, which were later distributed to the entire CoP to accommodate varying schedules and ability to attend live meetings. Approximately 20 to 30 participants attend the meetings in real-time.
To consolidate working documents, information, and resources in one location, we created a platform to communicate via a Microsoft Teams channel. All CoP members are given access to the folders and allowed to add to the growing library of resources. Resources include clinical assessment and note templates for electronic documentation of care, site-specific process maps, relevant literature on screening and interventions identified by practice members, and meeting notes along with the recordings. A chat feature alerts CoP members to questions posed by other members. Any resources or information shared on the chat discussion are curated by CoP leaders to disseminate to all members. Importantly, this platform allowed us to communicate efficiently within the VHA organization by creating a centralized space for documents and the ability to correspond with all or select members of the CoP. Additional VHA employees can easily be referred and request access.
To increase awareness of the CoP, expand reach, and diversify perspectives, every participant was encouraged to invite colleagues and stakeholders with interest or experience in long COVID care to join. While patients are not included in this CoP, we are working closely with the VHA user experience workgroup (many members overlap) that is gathering patient and caregiver perspectives on their COVID-19 experience and long COVID care. Concurrently, CoP members and leadership facilitate communication and set up formal collaborations with other non-VHA health care systems to create an intersystem network of collaboration for long COVID care. This approach further enhances the speed at which we can work together to share lessons learned and stay up-to-date on emerging evidence surrounding long COVID care.
Identifying Gaps in Care and Research
Our second goal was to identify gaps in care or knowledge to inform future research and quality improvement initiatives, while also creating a foundation to cocreate knowledge about safe, effective care management of the novel long COVID sequelae. To translate knowledge, we must first identify and understand the gaps between the current, best available evidence and current care practices or policies impacting that delivery.13 As such, the structured meeting agenda and facilitated meeting discussions focused on understanding current clinical decision making and the evidence base. We shared VHA evidence synthesis reports and living rapid reviews on complications following COVID-19 illness (ie, major organ damage and posthospitalization health care use) that provided an objective evidence base on common long COVID complications.14,15
Since long COVID is a novel condition, we drew from literature in similar patient populations and translated that information in the context of our current knowledge of this unique syndrome. For example, we discussed the predominant and persistent symptom of fatigue post-COVID.5 In particular, the CoP discussed challenges in identifying and treating post-COVID fatigue, which is often a vague symptom with multiple or interacting etiologies that require a comprehensive, interdisciplinary approach. As such, we reviewed, adapted, and translated identification and treatment strategies from the literature on chronic fatigue syndrome to patients with post-COVID syndrome.16,17 We continue to work collaboratively and engage the appropriate stakeholders to provide input on the gaps to prioritize targeting.
Cocreate Knowledge
Our third goal was to cocreate knowledge regarding the care of patients with long COVID. To accomplish this, our structured meetings and communication pathways invited members to share experiences on the who (delivers and receives care), what (type of care or HCPs), when (identification of post-COVID and access), and how (eg, telehealth) of care to patients post-COVID. As part of the workgroup, we identified and shared resources on standardized, facility-level practices to reduce variability across the VHA system. These resources included intake/assessment forms, care processes, and batteries of tests/measures used for screening and assessment. The knowledge obtained from outside the CoP and cocreated within is being used to inform data-driven tools to support and evaluate care for patients with long COVID. As such, members of the workgroup are in the formative stages of participating in quality improvement innovation pilots to test technologies and processes designed to improve and validate long COVID care pathways. These technologies include screening tools, clinical decision support tools, and population health management technologies. In addition, we are developing a formal collaboration with the VHA Office of Research and Development to create standardized intake forms across VHA long COVID clinics to facilitate both clinical monitoring and research.
Surveys
The US Department of Veterans Affairs Central Office collaborated with our workgroup to draft an initial set of survey questions designed to understand how each VHA facility defines, identifies, and provides care to veterans experiencing post-COVID sequalae. The 41-question survey was distributed through regional directors and chief medical officers at 139 VHA facilities in August 2021. One hundred nineteen responses (86%) were received. Sixteen facilities indicated they had established programs and 26 facilities were considering a program. Our CoP had representation from the 16 facilities with established programs indicating the deep and well-connected nature of our grassroots efforts to bring together stakeholders to learn as part of a CoP.
A separate, follow-up survey generated responses from 18 facilities and identified the need to capture evolving innovations and to develop smaller workstreams (eg, best practices, electronic documentation templates, pathway for referrals, veteran engagement, outcome measures). The survey not only exposed ongoing challenges to providing long COVID care, but importantly, outlined the ways in which CoP members were leveraging community knowledge and resources to inform innovations and processes of care changes at their specific sites. Fourteen of 18 facilities with long COVID programs in place explicitly identified the CoP as a resource they have found most beneficial when employing such innovations. Specific innovations reported included changes in care delivery, engagement in active outreach with veterans and local facility, and infrastructure development to sustain local long COVID clinics (Table).
Future Directions
Our CoP strives to contribute to an evidence base for long COVID care. At the system level, the CoP has the potential to impact access and continuity of care by identifying appropriate processes and ensuring that VHA patients receive outreach and an opportunity for post-COVID care. Comprehensive care requires input from HCP, clinical leadership, and operations levels. In this sense, our CoP provides an opportunity for diverse stakeholders to come together, discuss barriers to screening and delivering post-COVID care, and create an action plan to remove or lessen such barriers.18 Part of the process to remove barriers is to identify and support efficient resource allocation. Our CoP has worked to address issues in resource allocation (eg, space, personnel) for post-COVID care. For example, one facility is currently implementing interdisciplinary virtual post-COVID care. Another facility identified and restructured working assignments for psychologists who served in different capacities throughout the system to fill the need within the long COVID team.
At the HCP level, the CoP is currently developing workshops, media campaigns, written clinical resources, skills training, publications, and webinars/seminars with continuing medical education credits.19 The CoP may also provide learning and growth opportunities, such as clinical or VHA operational fellowships and research grants.
We are still in the formative stages of post-COVID care and future efforts will explore patient-centered outcomes. We are drawing on the Centers for Disease Control and Prevention’s guidance for evaluating patients with long COVID symptoms and examining the feasibility within VHA, as well as patient perspectives on post-COVID sequalae, to ensure we are selecting assessments that measure patient-centered constructs.18
Conclusions
A VHA-wide LHS approach is identifying issues related to the identification, delivery, and evaluation of long COVID care. This long COVID CoP has developed an infrastructure for communication, identified gaps in care, and cocreated knowledge related to best current practices for post-COVID care. This work is contributing to systemwide LHS efforts dedicated to creating a culture of quality care and innovation and is a process that is transferrable to other areas of care in the VHA, as well as other health care systems. The LHS approach continues to be highly relevant as we persist through the COVID-19 pandemic and reimagine a postpandemic world.
Acknowledg
We thank all the members of the Veterans Health Administration long COVID Community of Practice who participate in the meetings and contribute to the sharing and spread of knowledge.
1. Sivan M, Halpin S, Hollingworth L, Snook N, Hickman K, Clifton I. Development of an integrated rehabilitation pathway for individuals recovering from COVID-19 in the community. J Rehabil Med. 2020;52(8):jrm00089. doi:10.2340/16501977-2727
2. Understanding the long-term health effects of COVID-19. EClinicalMedicine. 2020;26:100586. doi:10.1016/j.eclinm.2020.100586
3. Greenhalgh T, Knight M, A’Court C, Buxton M, Husain L. Management of post-acute covid-19 in primary care. BMJ. Published online August 11, 2020:m3026. doi:10.1136/bmj.m3026
4. Iwua CJ, Iwu CD, Wiysonge CS. The occurrence of long COVID: a rapid review. Pan Afr Med J. 2021;38. doi:10.11604/pamj.2021.38.65.27366
5. Carfì A, Bernabei R, Landi F; Gemelli Against COVID-19 Post-Acute Care Study Group. Persistent symptoms in patients after acute COVID-19. JAMA. 2020;324(6):603-605. doi:10.1001/jama.2020.12603
6. Gemelli Against COVID-19 Post-Acute Care Study Group. Post-COVID-19 global health strategies: the need for an interdisciplinary approach. Aging Clin Exp Res. 2020;32(8):1613-1620. doi:10.1007/s40520-020-01616-x
7. Xie Y, Xu E, Bowe B, Al-Aly Z. Long-term cardiovascular outcomes of COVID-19. Nat Med. 2022;28:583-590. doi:10.1038/s41591-022-01689-3
8. Al-Aly Z, Xie Y, Bowe B. High-dimensional characterization of post-acute sequelae of COVID-19. Nature. 2021;594:259-264. doi:10.1038/s41586-021-03553-9
9. Ayoubkhani D, Bermingham C, Pouwels KB, et al. Trajectory of long covid symptoms after covid-19 vaccination: community based cohort study. BMJ. 2022;377:e069676. doi:10.1136/bmj-2021-069676
10. Institute of Medicine (US) Roundtable on Evidence-Based Medicine, Olsen L, Aisner D, McGinnis JM, eds. The Learning Healthcare System: Workshop Summary. Washington (DC): National Academies Press (US); 2007. doi:10.17226/11903
11. Romanelli RJ, Azar KMJ, Sudat S, Hung D, Frosch DL, Pressman AR. Learning health system in crisis: lessons from the COVID-19 pandemic. Mayo Clin Proc Innov Qual Outcomes. 2021;5(1):171-176. doi:10.1016/j.mayocpiqo.2020.10.004
12. Atkins D, Kilbourne AM, Shulkin D. Moving from discovery to system-wide change: the role of research in a learning health care system: experience from three decades of health systems research in the Veterans Health Administration. Annu Rev Public Health. 2017;38:467-487. doi:10.1146/annurev-publhealth-031816-044255
13. Kitson A, Straus SE. The knowledge-to-action cycle: identifying the gaps. CMAJ. 2010;182(2):E73-77. doi:10.1503/cmaj.081231
14. Greer N, Bart B, Billington C, et al. COVID-19 post-acute care major organ damage: a living rapid review. Updated September 2021. Accessed May 31, 2022. https://www.hsrd.research.va.gov/publications/esp/covid-organ-damage.pdf
15. Sharpe JA, Burke C, Gordon AM, et al. COVID-19 post-hospitalization health care utilization: a living review. Updated February 2022. Accessed May 31, 2022. https://www.hsrd.research.va.gov/publications/esp/covid19-post-hosp.pdf
16. Bested AC, Marshall LM. Review of Myalgic Encephalomyelitis/chronic fatigue syndrome: an evidence-based approach to diagnosis and management by clinicians. Rev Environ Health. 2015;30(4):223-249. doi:10.1515/reveh-2015-0026
17. Yancey JR, Thomas SM. Chronic fatigue syndrome: diagnosis and treatment. Am Fam Physician. 2012;86(8):741-746.
18. Kotter JP, Cohen DS. Change Leadership The Kotter Collection. Harvard Business Review Press; 2014.
19. Brownson RC, Eyler AA, Harris JK, Moore JB, Tabak RG. Getting the word out: new approaches for disseminating public health science. J Public Health Manag Pract. 2018;24(2):102-111. doi:10.1097/PHH.0000000000000673
1. Sivan M, Halpin S, Hollingworth L, Snook N, Hickman K, Clifton I. Development of an integrated rehabilitation pathway for individuals recovering from COVID-19 in the community. J Rehabil Med. 2020;52(8):jrm00089. doi:10.2340/16501977-2727
2. Understanding the long-term health effects of COVID-19. EClinicalMedicine. 2020;26:100586. doi:10.1016/j.eclinm.2020.100586
3. Greenhalgh T, Knight M, A’Court C, Buxton M, Husain L. Management of post-acute covid-19 in primary care. BMJ. Published online August 11, 2020:m3026. doi:10.1136/bmj.m3026
4. Iwua CJ, Iwu CD, Wiysonge CS. The occurrence of long COVID: a rapid review. Pan Afr Med J. 2021;38. doi:10.11604/pamj.2021.38.65.27366
5. Carfì A, Bernabei R, Landi F; Gemelli Against COVID-19 Post-Acute Care Study Group. Persistent symptoms in patients after acute COVID-19. JAMA. 2020;324(6):603-605. doi:10.1001/jama.2020.12603
6. Gemelli Against COVID-19 Post-Acute Care Study Group. Post-COVID-19 global health strategies: the need for an interdisciplinary approach. Aging Clin Exp Res. 2020;32(8):1613-1620. doi:10.1007/s40520-020-01616-x
7. Xie Y, Xu E, Bowe B, Al-Aly Z. Long-term cardiovascular outcomes of COVID-19. Nat Med. 2022;28:583-590. doi:10.1038/s41591-022-01689-3
8. Al-Aly Z, Xie Y, Bowe B. High-dimensional characterization of post-acute sequelae of COVID-19. Nature. 2021;594:259-264. doi:10.1038/s41586-021-03553-9
9. Ayoubkhani D, Bermingham C, Pouwels KB, et al. Trajectory of long covid symptoms after covid-19 vaccination: community based cohort study. BMJ. 2022;377:e069676. doi:10.1136/bmj-2021-069676
10. Institute of Medicine (US) Roundtable on Evidence-Based Medicine, Olsen L, Aisner D, McGinnis JM, eds. The Learning Healthcare System: Workshop Summary. Washington (DC): National Academies Press (US); 2007. doi:10.17226/11903
11. Romanelli RJ, Azar KMJ, Sudat S, Hung D, Frosch DL, Pressman AR. Learning health system in crisis: lessons from the COVID-19 pandemic. Mayo Clin Proc Innov Qual Outcomes. 2021;5(1):171-176. doi:10.1016/j.mayocpiqo.2020.10.004
12. Atkins D, Kilbourne AM, Shulkin D. Moving from discovery to system-wide change: the role of research in a learning health care system: experience from three decades of health systems research in the Veterans Health Administration. Annu Rev Public Health. 2017;38:467-487. doi:10.1146/annurev-publhealth-031816-044255
13. Kitson A, Straus SE. The knowledge-to-action cycle: identifying the gaps. CMAJ. 2010;182(2):E73-77. doi:10.1503/cmaj.081231
14. Greer N, Bart B, Billington C, et al. COVID-19 post-acute care major organ damage: a living rapid review. Updated September 2021. Accessed May 31, 2022. https://www.hsrd.research.va.gov/publications/esp/covid-organ-damage.pdf
15. Sharpe JA, Burke C, Gordon AM, et al. COVID-19 post-hospitalization health care utilization: a living review. Updated February 2022. Accessed May 31, 2022. https://www.hsrd.research.va.gov/publications/esp/covid19-post-hosp.pdf
16. Bested AC, Marshall LM. Review of Myalgic Encephalomyelitis/chronic fatigue syndrome: an evidence-based approach to diagnosis and management by clinicians. Rev Environ Health. 2015;30(4):223-249. doi:10.1515/reveh-2015-0026
17. Yancey JR, Thomas SM. Chronic fatigue syndrome: diagnosis and treatment. Am Fam Physician. 2012;86(8):741-746.
18. Kotter JP, Cohen DS. Change Leadership The Kotter Collection. Harvard Business Review Press; 2014.
19. Brownson RC, Eyler AA, Harris JK, Moore JB, Tabak RG. Getting the word out: new approaches for disseminating public health science. J Public Health Manag Pract. 2018;24(2):102-111. doi:10.1097/PHH.0000000000000673
Treatment and Management of Patients With Non-Small Cell Lung Cancer (FULL)
Comorbidities
Joshua M. Bauml, MD, Corporal Michael J. Crescenz VAMC, Philadelphia, PA. One of the
In addition, kidney dysfunction is quite common as a result of comorbid cardiovascular and hypertensive diseases. Kidney dysfunction can negatively impact our ability to administer both cisplatin and other systemic therapies.
Millie Das, MD, Palo Alto Health Care System, CA. Another major comorbidity for a lot of our veterans is COPD (chronic obstructive pulmonary disease). It doesn’t complicate the chemotherapy choice, but it affects surgical candidacy for those patients who present with early stage disease. Many times if you obtain pulmonary function tests in patients with COPD, the tests are abnormal and can prohibit safe surgical resection. These are patients that I see in the clinic and refer for definitive radiation, usually SABR (stereotactic ablative radiotherapy)/SBRT (stereotactic body radiation therapy), at a local radiation facility that can offer specialized radiation treatment.
Dr. Bauml. The fact that the VA has so many patients who require stereotactic radiosurgery for their early stage lung cancer represents an opportunity. There is a newly opened study that is evaluating SBRT vs surgery for these early stage lung cancer patients within the VA system. That study model has previously failed in multiple health care settings, but the VA is uniquely suited to answer this question.
Kelly A. Tammaro, PharmD, BCOP, Boston VA Healthcare System, MA. I would add heart failure patients or patients who have cardiac comorbidities and fluid restrictions. These restrictions can affect hydration that is needed for cisplatin, for example, as well as final volumes used to mix other chemotherapeutic agents with narrow concentration maximums, such as etoposide.
Julie Beck, RN, MSN, MPH, APRN-BC, VA Connecticut Healthcare System West Haven Campus. As a lung cancer navigator, I find that psychosocial comorbidities are an impediment to getting patients to diagnosis and treatment. Patients will miss appointments because they don’t have rides or will be reluctant to get imaging or other diagnostic testing because of anxiety or because it triggers PTSD (posttraumatic stress disorder) or because they are concerned about cost.
Dr. Das. I couldn’t agree more.
Dr. Bauml. It’s a great point.
Ms. Beck. You have to think outside the box with this patient population. We treat patients from as far away as Western Massachusetts. We have a dedicated oncology social worker who helps to arrange transportation. We have our CLC ( community living center), which is a rehabilitation and hospice unit but is also a resource for patients who live alone or far away and are getting an aggressive daily treatment regimen such as combined chemotherapy and radiation. We admit some patients to the CLC during their treatment to ensure that they get their treatment on time, maintain their nutritional status, and to provide emotional support. This is not an acute medical bed. Patients will sometimes go home on the weekend, but the support of the CLC increases the chance that they will get through their treatment safely.
Cancer care requires a lot of handholding. We often have to make multiple telephone calls to persuade our patients to get imaging or biopsies. Some of our patients require admission following biopsy because they live alone and have no one to drive them home following the procedure.
Dr. Tammaro. Boston has a similar model. We have a social worker who is highly dedicated and is able address our patients needs immediately. We also have many patients with PTSD and other psychological comorbidities, and depending on the severity, may require admission for their treatment to avoid the overwhelming nature of the ambulatory setting. For those who have to travel long distances for treatment we the Huntington House, which is housing located next door to our ambulatory campus. This accommodation can be used by our patients and their caregivers. We also have long term care facilities and a hospice unit located at our Brockton facility.
Ms. Beck. In West Haven, we have both palliative care and health psychology providers embedded in our clinic. They assist with symptom management and issues related to coping with diagnosis, anxiety, sleep, pain, smoking cessation, and lifestyle changes. We have also been offering pet therapy through our social work team, which has been very helpful for many of our patients.
Dr. Bauml. Mental health issues also can affect the choice of the type of treatment. Patients who have severe claustrophobia associated with their PTSD may have difficulty undergoing radiation. This can impact their ability to comply with therapy, and we have to adjust the treatment accordingly. For instance, I have a patient who has a known brain metastasis that was treated with definitive intent, but this gentleman gets highly agitated doing a brain magnetic resonance image (MRI). Instead we have had to follow him with serial computed tomography (CAT) scans, which is suboptimal. We have discussed that, but the distress that it causes him is simply not worth it.
Dr. Das. In some instances, we have had to use IV sedation for some of our patients with severe claustrophobia just to be able to get them through a positron emission tomography (PET) scan as part of their staging workup. We discuss these types of challenging cases in a multidisciplinary setting in our thoracic tumor board in order to brainstorm and figure out a realistic plan with our radiology and anesthesia colleagues, with the goal of getting the patient through the necessary tests in order to establish a treatment recommendation.
Due to underlying mental health or other health issues, some of our patients may also have difficulty with breath holding or with following other necessary instructions during their radiation treatments. We sometimes have to get creative on an individual basis in order to help a patient get through the needed treatment.
We have a dedicated psychologist and social worker who are embedded in our clinics and work closely with the oncology providers to offer strategies that can help our patients comply and complete the recommended treatment plan.
Rural Care
Dr. Bauml. One of the questions that comes up frequently when you have a patient who is remote is the type of treatment that you can administer. It’s difficult to administer a weekly therapy if somebody’s traveling 3 hours to see you every time. That can play into your decision making as you’re choosing a chemotherapy. If there are equivalent treatment regimens and one involves visits every 3 weeks and one involves weekly visits, well, that will help sway your decision making after discussion with the patient.
We often have to balance things. For instance, when I give someone carboplatin and paclitaxel, my preference is to administer it weekly with 3 weeks on and 1 week off. However, if a patient tells me, “You know, I do not want to come in once a week,” then I will discuss with them my concern for the increased adverse effects (AEs) with the every-3-week dosing. We will do it and then watch them closely. Of course, this gets even more complicated when you consider the fact that many of these patients have multiple medical comorbidities, so you’d like to administer the treatments in the least toxic way possible.
Ms. Beck. We have overcome some of those challenges by partnering with the primary care doctors. We are very close to our primary care colleagues in Massachusetts. They will order labs for the patient the day before the patient's appointment, so if the patient has a long drive, we already have their lab work; and they are ready to go when they get here for their treatment. The nursing staff is very aware of who needs to get on a shuttle back to Massachusetts. For some patients, we will have them stay overnight before their treatment.
Precision Oncology
Dr. Tammaro. In Boston, we have integrated Precision Oncology to be part of clinical practice, which we started with metastatic lung cancer patients. The VA Precision Oncology Program (POP) began at our healthcare center. We had to evaluate the genetic testing platforms, the accuracy of the results, and amount of tissue necessary for the laboratories. We have since succeeded in sending high-quality samples to the laboratories that generate accurate results. However, for your standard mutation panel for identifying therapy for first line treatment in lung cancer, we still use our local send out laboratory.
The POP has rolled out nationwide, and it is another clinical tool, especially for patients who have already failed multiple lines of therapy. When we send for a precision oncology consult, the “N of 1” report provides annotation. The report will generate a review of relevant literature and provide available abstracts or phase 1 or 2 trials that support a targeted therapy against potential point mutation for your patient.
The POP also has a research component, known as Re-POP. The goal is to open bucket trials that assess targeted therapy off label. Re-POP allows us to recontact these patients in the future to say, “You had your tissue sent through precision oncology, and you were diagnosed with a certain point mutation. Now we have a clinical trial that’s available. Would you be interested?” The plan is to have those clinical trials open and available to our patients when we receive the results from precision oncology.
I have used POP for 2 metastatic prostate cancer patient who exhausted all lines of therapy in hopes to identify a potential BCRA 1/2 mutation in order for us to use a PARP inhibitor. Unfortunately, neither harbored this mutation. Precision oncology does not perform immunohistochemistry, therefore identifying HER-2 or PD-L1 status for example, would need to be done through your local laboratory. I have found POP to be helpful in identifying a patients potential therapeutic option after progression on first/second line therapy, by sending tissue to POP initially or at the time of relapse.
Dr. Das. In our clinical practice at the Palo Alto VA, we follow the National Comprehensive Cancer Network (NCCN) guidelines, and we routinely evaluate for the presence of an EGFR mutation and also for ALK and ROS1 translocations in all lung cancer patients with nonsquamous histology. We send our molecular testing through Quest Diagnostics (Madison, NJ), and we usually get results back within a week or so.
For those patients who do not have any of those targetable gene alterations, we will go ahead and send for next-generation sequencing through POP, which allows testing of a much broader gene panel. Those results can take about a month or so to come back. I usually don’t wait for these results in order to get someone started on treatment. For patients without EGFR, ALK, or ROS1 found on initial testing, I will go ahead and start them on IV systemic chemotherapy. It is often very useful when you do get the next-generation sequencing results back, since in almost all cases, a gene alteration can be detected and is provided in the accompanying report. In a large subset of lung cancer cases, a gene alteration is seen in KRAS, for which we still do not have an effective targeted therapy. Despite this, I still find it useful to obtain the results because we generally feel that the driving genetic alterations occur mutually exclusive of one another. When we do see KRAS reported from a patient’s tumor specimen, we’re not generally looking for other types of mutations, so I find it helpful to know what is the alteration that is driving the growth of a patient’s tumor. The trend moving forward is to perform next-generation sequencing on all tumor specimens regardless of tumor type or histology, which can hopefully enable us to get to the bottom of what the driving genetic alteration is and to see if there are any targeted treatment approaches that can be offered to the patient.
In a few lung cancer cases, I have seen alterations in HER2 and BRAF that have been detected and reported using a next-generation sequencing platform. Just recently the FDA approved the BRAF-directed therapies of dabrafenib and trametinib for patients with lung cancer who are found to have a BRAF V600E mutation. It is hoped that as the FDA continues to provide approvals for targeted drugs in patients with lung cancer, the VA formulary will be able to offer these therapies to our veteran patients with the ultimate goal of providing treatment that has increased efficacy and less toxicity compared to conventional IV chemotherapy.
One of my frustrations earlier on was when we did find these more rare targetable mutations, I would run into problems with the VA formulary in allowing me to prescribe certain targeted therapies. In many cases, if the drug was not FDA-approved for lung cancer, I was told that I couldn’t use it and would have to go through the appeal process, which was quite onerous. Moving forward, we are seeing more and more data and trials with newer targeted agents in lung cancer, leading to new FDA approvals. With these approvals, I think it will be easier to be able to offer these targeted therapies to our patients.
Dr. Bauml. One of the issues that arises when we’re discussing even the FDA-approved therapies, is that many of these targeted therapies are relatively rare, and they’re especially rare amongst veterans. Now others have mentioned BRAF and HER2, and these do have some overexpression and mutations that occur among smokers. But the more common targetable genetic aberrations, EGFR, ALK, and ROS1 are more common amongst never-smokers. Given the high prevalence of tobacco use among veterans, these changes are rare. The incidence of ALK translocation is 3% to 7%. The incidence amongst veterans is likely much lower than that, given the tobacco abuse—to the point that I actually had a patient who had an ALK translocation; and of course, I prescribed the patient crizotinib. This was prior to the ALEX Trial and alectinib data. I prescribed crizotinib and was told it wasn’t on the formulary. Initially I was surprised, but when I said, “Well, look, when was the last time someone within our VA has prescribed crizotinib?” The answer was never.
This is the difficulty: As we enter this era of molecularly targetable therapy, the way we structure our formularies and the way that we review these data is going to have to change. This year at the American Society of Clinical Oncology (ASCO) meeting there were some very exciting lung cancer abstracts that evaluated ado-trastuzumab emtansine, which is an antibody drug conjugate currently approved for the treatment of HER2 overexpressing breast cancer. The abstracts showed response rates of up to 40% in lung cancer with the administration of this drug in HER2-mutated lung cancer. The HER2-amplified still had a response rate of 20%, which given the toxicity profile of this agent, is quite appealing. Being able to explore these early phase studies, as was described through the personalized medicine pathway, is, a great step forward for VA care.
Dr. Tammaro. The PBM in collaboration with the POP Advisory Board, are developing different levels of evidence to support the use of targeted medications identified to be potential therapy in those diagnosed with a point mutation. Even if a medication does not have an FDA approval, it has to have some evidence to support its use in a particular cancer. If you identify a point mutation or biomarker in a patient and provide evidence to supports its use within that particular disease state, the VA pharmacy could approve its use based off of that evidence. VA pharmacy would not require an actual FDA approval for that indication.
What the VISNs, PBM, and precision oncology are trying to do is determine the level of evidence that we have to support or approve use of a targeted therapy. We are definitely moving forward and changing the horizon on how we actually treat our patients after they’ve gone through first-line therapy. We are trying to figure out where these point mutations come in, the line of the therapy, and how we actually treat these cancers. Pharmacy is making a step forward in conjunction with Michael Kelley, MD, the National Program Director for Oncology, Specialty Care Services, whose group is establishing those guidelines.
Dr. Bauml. I don’t mean to downplay the difficulty of that process. This is a huge, difficult process. One only needs to look at the long line of failed trials looking at PI3 kinase inhibitors to show that just knowing that a mutation exists does not necessarily mean that a targeted therapy works in that space.
Drawing that line is really complicated, both within the VA and, indeed, outside of the VA. It’s a really complicated process, and understanding the implications of different mutations is only going to get more complicated. Of course, now we have things like NTRK and even rarer genetic aberrations that are going to affect not only lung cancer, but also a wide range of malignancies.
Promising Research
Dr. Bauml. The pathways that are emerging as clear driver mutations for which we have available therapies, at least within lung cancer, are MET exon 14, RET, and NTRK. I am also intrigued by the emerging data in the HER2 space.
Dr. Das. The other therapy that has been getting a lot of press is immunotherapy, of course. And I’ve been seeing many really good responders to immunotherapy within the veteran population that I treat. It is felt that degree of PD-L1 expression correlates with responsiveness to the immune check point inhibitors that are being used in lung cancer, and we are tending to see higher rates of PD-L1 expression in patients who are prior or current smokers who have a higher overall tumor mutation burden.
I see patients both at Stanford and at the Palo Alto VA, and I have noticed that the patients that I have been treating at the VA tend to have higher levels of PD-L1 expression with better responses to the immunotherapy drugs, probably because most of the VA patients are former or current smokers. And, another interesting observation is that these veteran patients are, for whatever reason, having a lower incidence of some of the autoimmune AEs seen with these immune checkpoint inhibitors. I have been keeping an eye out for more data and information to support these observations I have had in my clinical practice and I specifically attended ASCO this year to learn more about what others have seen and studied with immune check point inhibition in lung cancer. We are learning now that PD-L1 is not a perfect marker for predicting response to the checkpoint inhibitors and the other immunotherapeutic agents, and there is a great deal of research going on to try to figure out what other biomarkers could be useful and which patients are most likely to benefit from these drugs.
I was excited to hear about the combination of nivolumab and ipilimumab that is being tested in both mesothelioma and in small-cell lung cancer where we really don’t have as many treatment options as we have in non-small cell lung cancer. That data was quite exciting, and interestingly, there does not seem to be a correlation with PD-L1 expression and responsiveness to treatment with the immunotherapeutic agents in those histologic subtypes. The story is still unfolding, and we await additional data to help guide us in our treatment decisions.
Dr. Tammaro. Immunotherapy is the new fad in oncology. We have just scheduled our first patient for first-line therapy due to PD-L1 tumor proportion score is > 50%. Recently, at ASCO KEYNOTE-021 researchers looked at using pembrolizumab in combination with carboplatin plus pemetrexed chemotherapy for first-line metastatic non-squamous NSCLC. The research suggested that patients treated with pembrolizumab + chemotherapy continued to derive a higher overall response rate and progression free survival when compare with those on chemotherapy alone despite a low or no PD-L1 tumor expression.
It’s very interesting that many clinical trials that we’re evaluating are now using some type of checkpoint inhibitor up front with cytotoxic chemotherapy. If they are positive trials, this could change how patients are treated up front.
Dr. Bauml. There was some really interesting data that were presented at ASCO this year by Matthew Hellmann, MD, which evaluated the predictive nature of PD-L1 vs tumor mutation burden and other biomarkers, including gene expression profiling. In this particular abstract, the PD-L1 and tumor mutation burden really do function as orthogonal biomarkers such that a patient who has high PD-L1 and high tumor mutation burden is the most likely to respond. Patients who are really low for both are unlikely to respond. We really need better biomarkers for immunotherapy, though. PD-L1 has a lot of limitations, namely, it is dynamic, so over time it changes. So I can do a biopsy at one point, then treat the patient and the PD-L1 may change.
More importantly, it’s heterogeneous. There was this great paper by McLaughlin and colleagues in JAMA Oncology (2016) who described a patient who had a small tumor biopsy. They took a micrograph of the tumor and showed that one part of the micrograph was completely floridly PD-L1 positive. At another site of the same biopsy it was completely stone-cold negative, which is humbling when you think about the fact that we stick small needles into tumors and make clinical decisions on the basis of that.
The KEYNOTE-024 study evaluated pembrolizumab vs chemotherapy in high PD-L1 expressers. It’s a very exciting study, but at the end of the day even in this highly select patient population, the response rate to immunotherapy was only about 50%, which is not the sort of biomarker-driven response that we’re used to seeing with our EGFR inhibitors. That’s really what we want to get to. More important even than that is being able to say the negative predictive value. One of the reasons that we’re probably seeing more responses among veterans is that we know that patients who are veterans who have high tobacco exposure have a higher tumor mutation burden. I’m surprised to hear about the immune-related AEs, actually, because one of the things that was reported this year at ASCO was some data that showed that patients who have immune-related AEs are more likely to have a better outcome, which is an interesting biomarker of response.
Dr. Das. I heard that as well, and I found that to be really interesting. The patients that I’ve had on nivolumab for over a year are doing very well. These are stage IV patients who have essentially had complete responses to treatment and have not had any or have had very minor immune-related AEs to date.
Overall, these are a small numbers of patients, but I have been curious to see why that might be the case. Anecdotally, my colleagues and I who treat patients at Stanford have seen significantly higher rates of grades 3 and 4 pneumonitis and other autoimmune toxicities, such as myocarditis and enterocolitis, in those lung cancer patients who are light or never-smokers treated with immune checkpoint inhibitors.
Dr. Bauml. I really feel that PD-L1 as a biomarker has significant limitations. I certainly hope that in at least 2 or 3 years we’re not going to be talking about PD-L1 anymore. I’m hopeful that we’ll be able to use better predictive biomarkers, such as mutational burden and gene expression profiling. In the data in head and neck that was presented this year at ASCO, patients who were low for both gene expression profiling and mutational burden had a very low response (Haddad et al, ASCO 2017).
That’s really what you want to be. You want to be able to say, “Here’s a person who will not benefit from this therapy.” From there you can identify, based upon these biomarkers, the combination that is going to be best for this person. Is it chemoimmunotherapy or combination immunotherapy with CTLA4, or another checkpoint blockade? That is really the way that we’re going to be able to fine-tune this, because the toxicity is substantial for some treatments, like the nivolumab/ipilimumab combination. Using them in a biomarker-blind fashion is just scary to me, honestly.
Managing Adverse Reactions
Dr. Tammaro. The increasing amount of oral chemotherapy has posed a significant challenge. As a clinical oncology pharmacist, it was difficult to grasp the most effective way to follow all these patients and ensure adherence, adverse drug event reporting/significance and adequate follow up. When patients are receiving IV chemotherapy, we know we will see them, we are assured compliance and are able to assess side effects in a timely manner. When we give oral chemotherapy, the tables are turned, where the responsibility is now on the patient. We are now depending on the patient to ensure they are taking the medication correctly and we may not see AEs if the patient misses an appointment or feels as though they are bothering the provider by calling.
In 2012, we started an oral oncology clinic here at the VA in Boston that I found to be extremely effective. When you’re sending a patient home with an oral chemotherapy, you have to make sure that you are counseling them correctly and encourage them to call at any time if they are experiencing any type of AE. One of the newest issues we have been seeing is bleeding with ibrutinib, especially in those patients on anticoagulation therapies.
A general strategy we employee for oral chemotherapy is to start at half dose and titrate slowly. This method has been effective in identifying AEs and preventing delays in therapy. We do this for the majority of oral chemotherapy. Patients are given a 2 week supply to start and then are reassessed on follow up for escalation to the target dose. We do not place refills on oral oncology prescriptions. They are instructed to call 10 days prior to running out if they are not scheduled to come in for an appointment. Having consistent dialogue with our patients allows us to assess for adherence, AEs, and tolerability. The other advantage to this clinic is ensuring our patients have someone to speak to at all times and answer all their questions. Direct lines of communication is what most of our patients are appreciative of when paying gratitude to the clinic.
Ms. Beck. We have an oral chemotherapy clinic staffed by dedicated oncology pharmacists. Patients meet with the pharmacist and have education prior to starting a new oral chemotherapy. They will then be followed by both the oncology provider and the pharmacist.
Dr. Das. One of the challenges we also face is with so many of our patients living so far away. When our patients do have AEs that require hospitalization, it can be very tricky to really get a sense for how they are being managed at the outside community (non-VA) facility. Sharing of electronic medical records can be a challenge in these cases, and I worry that the care teams at the more remote hospitals may not be as familiar with the newer cancer treatments and the toxicities associated with them, such as the autoimmune AEs associated with many of the immune checkpoint inhibitors.
I provide patients with pocket cards to keep in their wallets with my contact information and the name of the drug that they are getting because not all patients can remember or even pronounce the names of the drugs and may not be able to tell their local treating physician and care team what they are getting. I have been getting more frequent phone calls from emergency department physicians and hospitalists from the local communities where many of our veterans live, because they want guidance on how best to approach treatment for our patients when they show up with an AE related to their cancer treatment.
At times, the presenting symptoms may be vague or nonspecific, but for our patients being treated with immunotherapy, we always have to keep in mind the possibility of immune-related AEs because we know that prompt initiation of steroids is critical in these cases and can really help the patients feel better quickly.
Dr. Tammaro. You bring up a valid point. Our pharmacists meet with all the patients on checkpoint inhibitors. Specifically, when we started using ipilimumab it was uncharted territory for our team. We put together take home medication bag that included hydrocortisone cream, methylprednisolone dose pak, dipheydramine, and loperamide. This was utilized for all patients and specific attention was given to patients who lived far away from an emergency room. This bag system was accompanied by “what to do if I have this symptom” handout that outlined which medication to take depending on the severity of the AE. A direct line phone line to the oncology pharmacy also was supplied.
With the evolution to the PD-L1s and the anti-PD inhibitors, we haven’t seen the same level of AEs. Patients go home with wallet cards that includes our staff contact numbers/pagers. The wallet card also serves as information to a treating provider if the patient presents outside the VA, to ensure they understand the severity of a potential autoimmune AE, such as diarrhea.
Another challenge is shared-care patients. We have patients coming from outside hospitals, and at times they want to use this pharmacy like a CVS, and it just doesn’t operate that way. We want to collaborate with others. Most shared care patients present to our service for oral chemotherapy because the veteran just can’t afford the copays. So, we will see the patient concurrently. They can still see their outside hospital physician as well, but they have to fax us the laboratory results and progress notes on a monthly basis (or longer depending on where they are in there therapy). Before we fill their medications, we talk to the patients, the same way we would treat a veteran who was getting their oral chemotherapy here. In addition, they need to be seen by the VA physician at least every 3 months. We want our veterans to feel comfortable with the cancer care and help them out as best as we can.
Click here to read the digital edition.
Comorbidities
Joshua M. Bauml, MD, Corporal Michael J. Crescenz VAMC, Philadelphia, PA. One of the
In addition, kidney dysfunction is quite common as a result of comorbid cardiovascular and hypertensive diseases. Kidney dysfunction can negatively impact our ability to administer both cisplatin and other systemic therapies.
Millie Das, MD, Palo Alto Health Care System, CA. Another major comorbidity for a lot of our veterans is COPD (chronic obstructive pulmonary disease). It doesn’t complicate the chemotherapy choice, but it affects surgical candidacy for those patients who present with early stage disease. Many times if you obtain pulmonary function tests in patients with COPD, the tests are abnormal and can prohibit safe surgical resection. These are patients that I see in the clinic and refer for definitive radiation, usually SABR (stereotactic ablative radiotherapy)/SBRT (stereotactic body radiation therapy), at a local radiation facility that can offer specialized radiation treatment.
Dr. Bauml. The fact that the VA has so many patients who require stereotactic radiosurgery for their early stage lung cancer represents an opportunity. There is a newly opened study that is evaluating SBRT vs surgery for these early stage lung cancer patients within the VA system. That study model has previously failed in multiple health care settings, but the VA is uniquely suited to answer this question.
Kelly A. Tammaro, PharmD, BCOP, Boston VA Healthcare System, MA. I would add heart failure patients or patients who have cardiac comorbidities and fluid restrictions. These restrictions can affect hydration that is needed for cisplatin, for example, as well as final volumes used to mix other chemotherapeutic agents with narrow concentration maximums, such as etoposide.
Julie Beck, RN, MSN, MPH, APRN-BC, VA Connecticut Healthcare System West Haven Campus. As a lung cancer navigator, I find that psychosocial comorbidities are an impediment to getting patients to diagnosis and treatment. Patients will miss appointments because they don’t have rides or will be reluctant to get imaging or other diagnostic testing because of anxiety or because it triggers PTSD (posttraumatic stress disorder) or because they are concerned about cost.
Dr. Das. I couldn’t agree more.
Dr. Bauml. It’s a great point.
Ms. Beck. You have to think outside the box with this patient population. We treat patients from as far away as Western Massachusetts. We have a dedicated oncology social worker who helps to arrange transportation. We have our CLC ( community living center), which is a rehabilitation and hospice unit but is also a resource for patients who live alone or far away and are getting an aggressive daily treatment regimen such as combined chemotherapy and radiation. We admit some patients to the CLC during their treatment to ensure that they get their treatment on time, maintain their nutritional status, and to provide emotional support. This is not an acute medical bed. Patients will sometimes go home on the weekend, but the support of the CLC increases the chance that they will get through their treatment safely.
Cancer care requires a lot of handholding. We often have to make multiple telephone calls to persuade our patients to get imaging or biopsies. Some of our patients require admission following biopsy because they live alone and have no one to drive them home following the procedure.
Dr. Tammaro. Boston has a similar model. We have a social worker who is highly dedicated and is able address our patients needs immediately. We also have many patients with PTSD and other psychological comorbidities, and depending on the severity, may require admission for their treatment to avoid the overwhelming nature of the ambulatory setting. For those who have to travel long distances for treatment we the Huntington House, which is housing located next door to our ambulatory campus. This accommodation can be used by our patients and their caregivers. We also have long term care facilities and a hospice unit located at our Brockton facility.
Ms. Beck. In West Haven, we have both palliative care and health psychology providers embedded in our clinic. They assist with symptom management and issues related to coping with diagnosis, anxiety, sleep, pain, smoking cessation, and lifestyle changes. We have also been offering pet therapy through our social work team, which has been very helpful for many of our patients.
Dr. Bauml. Mental health issues also can affect the choice of the type of treatment. Patients who have severe claustrophobia associated with their PTSD may have difficulty undergoing radiation. This can impact their ability to comply with therapy, and we have to adjust the treatment accordingly. For instance, I have a patient who has a known brain metastasis that was treated with definitive intent, but this gentleman gets highly agitated doing a brain magnetic resonance image (MRI). Instead we have had to follow him with serial computed tomography (CAT) scans, which is suboptimal. We have discussed that, but the distress that it causes him is simply not worth it.
Dr. Das. In some instances, we have had to use IV sedation for some of our patients with severe claustrophobia just to be able to get them through a positron emission tomography (PET) scan as part of their staging workup. We discuss these types of challenging cases in a multidisciplinary setting in our thoracic tumor board in order to brainstorm and figure out a realistic plan with our radiology and anesthesia colleagues, with the goal of getting the patient through the necessary tests in order to establish a treatment recommendation.
Due to underlying mental health or other health issues, some of our patients may also have difficulty with breath holding or with following other necessary instructions during their radiation treatments. We sometimes have to get creative on an individual basis in order to help a patient get through the needed treatment.
We have a dedicated psychologist and social worker who are embedded in our clinics and work closely with the oncology providers to offer strategies that can help our patients comply and complete the recommended treatment plan.
Rural Care
Dr. Bauml. One of the questions that comes up frequently when you have a patient who is remote is the type of treatment that you can administer. It’s difficult to administer a weekly therapy if somebody’s traveling 3 hours to see you every time. That can play into your decision making as you’re choosing a chemotherapy. If there are equivalent treatment regimens and one involves visits every 3 weeks and one involves weekly visits, well, that will help sway your decision making after discussion with the patient.
We often have to balance things. For instance, when I give someone carboplatin and paclitaxel, my preference is to administer it weekly with 3 weeks on and 1 week off. However, if a patient tells me, “You know, I do not want to come in once a week,” then I will discuss with them my concern for the increased adverse effects (AEs) with the every-3-week dosing. We will do it and then watch them closely. Of course, this gets even more complicated when you consider the fact that many of these patients have multiple medical comorbidities, so you’d like to administer the treatments in the least toxic way possible.
Ms. Beck. We have overcome some of those challenges by partnering with the primary care doctors. We are very close to our primary care colleagues in Massachusetts. They will order labs for the patient the day before the patient's appointment, so if the patient has a long drive, we already have their lab work; and they are ready to go when they get here for their treatment. The nursing staff is very aware of who needs to get on a shuttle back to Massachusetts. For some patients, we will have them stay overnight before their treatment.
Precision Oncology
Dr. Tammaro. In Boston, we have integrated Precision Oncology to be part of clinical practice, which we started with metastatic lung cancer patients. The VA Precision Oncology Program (POP) began at our healthcare center. We had to evaluate the genetic testing platforms, the accuracy of the results, and amount of tissue necessary for the laboratories. We have since succeeded in sending high-quality samples to the laboratories that generate accurate results. However, for your standard mutation panel for identifying therapy for first line treatment in lung cancer, we still use our local send out laboratory.
The POP has rolled out nationwide, and it is another clinical tool, especially for patients who have already failed multiple lines of therapy. When we send for a precision oncology consult, the “N of 1” report provides annotation. The report will generate a review of relevant literature and provide available abstracts or phase 1 or 2 trials that support a targeted therapy against potential point mutation for your patient.
The POP also has a research component, known as Re-POP. The goal is to open bucket trials that assess targeted therapy off label. Re-POP allows us to recontact these patients in the future to say, “You had your tissue sent through precision oncology, and you were diagnosed with a certain point mutation. Now we have a clinical trial that’s available. Would you be interested?” The plan is to have those clinical trials open and available to our patients when we receive the results from precision oncology.
I have used POP for 2 metastatic prostate cancer patient who exhausted all lines of therapy in hopes to identify a potential BCRA 1/2 mutation in order for us to use a PARP inhibitor. Unfortunately, neither harbored this mutation. Precision oncology does not perform immunohistochemistry, therefore identifying HER-2 or PD-L1 status for example, would need to be done through your local laboratory. I have found POP to be helpful in identifying a patients potential therapeutic option after progression on first/second line therapy, by sending tissue to POP initially or at the time of relapse.
Dr. Das. In our clinical practice at the Palo Alto VA, we follow the National Comprehensive Cancer Network (NCCN) guidelines, and we routinely evaluate for the presence of an EGFR mutation and also for ALK and ROS1 translocations in all lung cancer patients with nonsquamous histology. We send our molecular testing through Quest Diagnostics (Madison, NJ), and we usually get results back within a week or so.
For those patients who do not have any of those targetable gene alterations, we will go ahead and send for next-generation sequencing through POP, which allows testing of a much broader gene panel. Those results can take about a month or so to come back. I usually don’t wait for these results in order to get someone started on treatment. For patients without EGFR, ALK, or ROS1 found on initial testing, I will go ahead and start them on IV systemic chemotherapy. It is often very useful when you do get the next-generation sequencing results back, since in almost all cases, a gene alteration can be detected and is provided in the accompanying report. In a large subset of lung cancer cases, a gene alteration is seen in KRAS, for which we still do not have an effective targeted therapy. Despite this, I still find it useful to obtain the results because we generally feel that the driving genetic alterations occur mutually exclusive of one another. When we do see KRAS reported from a patient’s tumor specimen, we’re not generally looking for other types of mutations, so I find it helpful to know what is the alteration that is driving the growth of a patient’s tumor. The trend moving forward is to perform next-generation sequencing on all tumor specimens regardless of tumor type or histology, which can hopefully enable us to get to the bottom of what the driving genetic alteration is and to see if there are any targeted treatment approaches that can be offered to the patient.
In a few lung cancer cases, I have seen alterations in HER2 and BRAF that have been detected and reported using a next-generation sequencing platform. Just recently the FDA approved the BRAF-directed therapies of dabrafenib and trametinib for patients with lung cancer who are found to have a BRAF V600E mutation. It is hoped that as the FDA continues to provide approvals for targeted drugs in patients with lung cancer, the VA formulary will be able to offer these therapies to our veteran patients with the ultimate goal of providing treatment that has increased efficacy and less toxicity compared to conventional IV chemotherapy.
One of my frustrations earlier on was when we did find these more rare targetable mutations, I would run into problems with the VA formulary in allowing me to prescribe certain targeted therapies. In many cases, if the drug was not FDA-approved for lung cancer, I was told that I couldn’t use it and would have to go through the appeal process, which was quite onerous. Moving forward, we are seeing more and more data and trials with newer targeted agents in lung cancer, leading to new FDA approvals. With these approvals, I think it will be easier to be able to offer these targeted therapies to our patients.
Dr. Bauml. One of the issues that arises when we’re discussing even the FDA-approved therapies, is that many of these targeted therapies are relatively rare, and they’re especially rare amongst veterans. Now others have mentioned BRAF and HER2, and these do have some overexpression and mutations that occur among smokers. But the more common targetable genetic aberrations, EGFR, ALK, and ROS1 are more common amongst never-smokers. Given the high prevalence of tobacco use among veterans, these changes are rare. The incidence of ALK translocation is 3% to 7%. The incidence amongst veterans is likely much lower than that, given the tobacco abuse—to the point that I actually had a patient who had an ALK translocation; and of course, I prescribed the patient crizotinib. This was prior to the ALEX Trial and alectinib data. I prescribed crizotinib and was told it wasn’t on the formulary. Initially I was surprised, but when I said, “Well, look, when was the last time someone within our VA has prescribed crizotinib?” The answer was never.
This is the difficulty: As we enter this era of molecularly targetable therapy, the way we structure our formularies and the way that we review these data is going to have to change. This year at the American Society of Clinical Oncology (ASCO) meeting there were some very exciting lung cancer abstracts that evaluated ado-trastuzumab emtansine, which is an antibody drug conjugate currently approved for the treatment of HER2 overexpressing breast cancer. The abstracts showed response rates of up to 40% in lung cancer with the administration of this drug in HER2-mutated lung cancer. The HER2-amplified still had a response rate of 20%, which given the toxicity profile of this agent, is quite appealing. Being able to explore these early phase studies, as was described through the personalized medicine pathway, is, a great step forward for VA care.
Dr. Tammaro. The PBM in collaboration with the POP Advisory Board, are developing different levels of evidence to support the use of targeted medications identified to be potential therapy in those diagnosed with a point mutation. Even if a medication does not have an FDA approval, it has to have some evidence to support its use in a particular cancer. If you identify a point mutation or biomarker in a patient and provide evidence to supports its use within that particular disease state, the VA pharmacy could approve its use based off of that evidence. VA pharmacy would not require an actual FDA approval for that indication.
What the VISNs, PBM, and precision oncology are trying to do is determine the level of evidence that we have to support or approve use of a targeted therapy. We are definitely moving forward and changing the horizon on how we actually treat our patients after they’ve gone through first-line therapy. We are trying to figure out where these point mutations come in, the line of the therapy, and how we actually treat these cancers. Pharmacy is making a step forward in conjunction with Michael Kelley, MD, the National Program Director for Oncology, Specialty Care Services, whose group is establishing those guidelines.
Dr. Bauml. I don’t mean to downplay the difficulty of that process. This is a huge, difficult process. One only needs to look at the long line of failed trials looking at PI3 kinase inhibitors to show that just knowing that a mutation exists does not necessarily mean that a targeted therapy works in that space.
Drawing that line is really complicated, both within the VA and, indeed, outside of the VA. It’s a really complicated process, and understanding the implications of different mutations is only going to get more complicated. Of course, now we have things like NTRK and even rarer genetic aberrations that are going to affect not only lung cancer, but also a wide range of malignancies.
Promising Research
Dr. Bauml. The pathways that are emerging as clear driver mutations for which we have available therapies, at least within lung cancer, are MET exon 14, RET, and NTRK. I am also intrigued by the emerging data in the HER2 space.
Dr. Das. The other therapy that has been getting a lot of press is immunotherapy, of course. And I’ve been seeing many really good responders to immunotherapy within the veteran population that I treat. It is felt that degree of PD-L1 expression correlates with responsiveness to the immune check point inhibitors that are being used in lung cancer, and we are tending to see higher rates of PD-L1 expression in patients who are prior or current smokers who have a higher overall tumor mutation burden.
I see patients both at Stanford and at the Palo Alto VA, and I have noticed that the patients that I have been treating at the VA tend to have higher levels of PD-L1 expression with better responses to the immunotherapy drugs, probably because most of the VA patients are former or current smokers. And, another interesting observation is that these veteran patients are, for whatever reason, having a lower incidence of some of the autoimmune AEs seen with these immune checkpoint inhibitors. I have been keeping an eye out for more data and information to support these observations I have had in my clinical practice and I specifically attended ASCO this year to learn more about what others have seen and studied with immune check point inhibition in lung cancer. We are learning now that PD-L1 is not a perfect marker for predicting response to the checkpoint inhibitors and the other immunotherapeutic agents, and there is a great deal of research going on to try to figure out what other biomarkers could be useful and which patients are most likely to benefit from these drugs.
I was excited to hear about the combination of nivolumab and ipilimumab that is being tested in both mesothelioma and in small-cell lung cancer where we really don’t have as many treatment options as we have in non-small cell lung cancer. That data was quite exciting, and interestingly, there does not seem to be a correlation with PD-L1 expression and responsiveness to treatment with the immunotherapeutic agents in those histologic subtypes. The story is still unfolding, and we await additional data to help guide us in our treatment decisions.
Dr. Tammaro. Immunotherapy is the new fad in oncology. We have just scheduled our first patient for first-line therapy due to PD-L1 tumor proportion score is > 50%. Recently, at ASCO KEYNOTE-021 researchers looked at using pembrolizumab in combination with carboplatin plus pemetrexed chemotherapy for first-line metastatic non-squamous NSCLC. The research suggested that patients treated with pembrolizumab + chemotherapy continued to derive a higher overall response rate and progression free survival when compare with those on chemotherapy alone despite a low or no PD-L1 tumor expression.
It’s very interesting that many clinical trials that we’re evaluating are now using some type of checkpoint inhibitor up front with cytotoxic chemotherapy. If they are positive trials, this could change how patients are treated up front.
Dr. Bauml. There was some really interesting data that were presented at ASCO this year by Matthew Hellmann, MD, which evaluated the predictive nature of PD-L1 vs tumor mutation burden and other biomarkers, including gene expression profiling. In this particular abstract, the PD-L1 and tumor mutation burden really do function as orthogonal biomarkers such that a patient who has high PD-L1 and high tumor mutation burden is the most likely to respond. Patients who are really low for both are unlikely to respond. We really need better biomarkers for immunotherapy, though. PD-L1 has a lot of limitations, namely, it is dynamic, so over time it changes. So I can do a biopsy at one point, then treat the patient and the PD-L1 may change.
More importantly, it’s heterogeneous. There was this great paper by McLaughlin and colleagues in JAMA Oncology (2016) who described a patient who had a small tumor biopsy. They took a micrograph of the tumor and showed that one part of the micrograph was completely floridly PD-L1 positive. At another site of the same biopsy it was completely stone-cold negative, which is humbling when you think about the fact that we stick small needles into tumors and make clinical decisions on the basis of that.
The KEYNOTE-024 study evaluated pembrolizumab vs chemotherapy in high PD-L1 expressers. It’s a very exciting study, but at the end of the day even in this highly select patient population, the response rate to immunotherapy was only about 50%, which is not the sort of biomarker-driven response that we’re used to seeing with our EGFR inhibitors. That’s really what we want to get to. More important even than that is being able to say the negative predictive value. One of the reasons that we’re probably seeing more responses among veterans is that we know that patients who are veterans who have high tobacco exposure have a higher tumor mutation burden. I’m surprised to hear about the immune-related AEs, actually, because one of the things that was reported this year at ASCO was some data that showed that patients who have immune-related AEs are more likely to have a better outcome, which is an interesting biomarker of response.
Dr. Das. I heard that as well, and I found that to be really interesting. The patients that I’ve had on nivolumab for over a year are doing very well. These are stage IV patients who have essentially had complete responses to treatment and have not had any or have had very minor immune-related AEs to date.
Overall, these are a small numbers of patients, but I have been curious to see why that might be the case. Anecdotally, my colleagues and I who treat patients at Stanford have seen significantly higher rates of grades 3 and 4 pneumonitis and other autoimmune toxicities, such as myocarditis and enterocolitis, in those lung cancer patients who are light or never-smokers treated with immune checkpoint inhibitors.
Dr. Bauml. I really feel that PD-L1 as a biomarker has significant limitations. I certainly hope that in at least 2 or 3 years we’re not going to be talking about PD-L1 anymore. I’m hopeful that we’ll be able to use better predictive biomarkers, such as mutational burden and gene expression profiling. In the data in head and neck that was presented this year at ASCO, patients who were low for both gene expression profiling and mutational burden had a very low response (Haddad et al, ASCO 2017).
That’s really what you want to be. You want to be able to say, “Here’s a person who will not benefit from this therapy.” From there you can identify, based upon these biomarkers, the combination that is going to be best for this person. Is it chemoimmunotherapy or combination immunotherapy with CTLA4, or another checkpoint blockade? That is really the way that we’re going to be able to fine-tune this, because the toxicity is substantial for some treatments, like the nivolumab/ipilimumab combination. Using them in a biomarker-blind fashion is just scary to me, honestly.
Managing Adverse Reactions
Dr. Tammaro. The increasing amount of oral chemotherapy has posed a significant challenge. As a clinical oncology pharmacist, it was difficult to grasp the most effective way to follow all these patients and ensure adherence, adverse drug event reporting/significance and adequate follow up. When patients are receiving IV chemotherapy, we know we will see them, we are assured compliance and are able to assess side effects in a timely manner. When we give oral chemotherapy, the tables are turned, where the responsibility is now on the patient. We are now depending on the patient to ensure they are taking the medication correctly and we may not see AEs if the patient misses an appointment or feels as though they are bothering the provider by calling.
In 2012, we started an oral oncology clinic here at the VA in Boston that I found to be extremely effective. When you’re sending a patient home with an oral chemotherapy, you have to make sure that you are counseling them correctly and encourage them to call at any time if they are experiencing any type of AE. One of the newest issues we have been seeing is bleeding with ibrutinib, especially in those patients on anticoagulation therapies.
A general strategy we employee for oral chemotherapy is to start at half dose and titrate slowly. This method has been effective in identifying AEs and preventing delays in therapy. We do this for the majority of oral chemotherapy. Patients are given a 2 week supply to start and then are reassessed on follow up for escalation to the target dose. We do not place refills on oral oncology prescriptions. They are instructed to call 10 days prior to running out if they are not scheduled to come in for an appointment. Having consistent dialogue with our patients allows us to assess for adherence, AEs, and tolerability. The other advantage to this clinic is ensuring our patients have someone to speak to at all times and answer all their questions. Direct lines of communication is what most of our patients are appreciative of when paying gratitude to the clinic.
Ms. Beck. We have an oral chemotherapy clinic staffed by dedicated oncology pharmacists. Patients meet with the pharmacist and have education prior to starting a new oral chemotherapy. They will then be followed by both the oncology provider and the pharmacist.
Dr. Das. One of the challenges we also face is with so many of our patients living so far away. When our patients do have AEs that require hospitalization, it can be very tricky to really get a sense for how they are being managed at the outside community (non-VA) facility. Sharing of electronic medical records can be a challenge in these cases, and I worry that the care teams at the more remote hospitals may not be as familiar with the newer cancer treatments and the toxicities associated with them, such as the autoimmune AEs associated with many of the immune checkpoint inhibitors.
I provide patients with pocket cards to keep in their wallets with my contact information and the name of the drug that they are getting because not all patients can remember or even pronounce the names of the drugs and may not be able to tell their local treating physician and care team what they are getting. I have been getting more frequent phone calls from emergency department physicians and hospitalists from the local communities where many of our veterans live, because they want guidance on how best to approach treatment for our patients when they show up with an AE related to their cancer treatment.
At times, the presenting symptoms may be vague or nonspecific, but for our patients being treated with immunotherapy, we always have to keep in mind the possibility of immune-related AEs because we know that prompt initiation of steroids is critical in these cases and can really help the patients feel better quickly.
Dr. Tammaro. You bring up a valid point. Our pharmacists meet with all the patients on checkpoint inhibitors. Specifically, when we started using ipilimumab it was uncharted territory for our team. We put together take home medication bag that included hydrocortisone cream, methylprednisolone dose pak, dipheydramine, and loperamide. This was utilized for all patients and specific attention was given to patients who lived far away from an emergency room. This bag system was accompanied by “what to do if I have this symptom” handout that outlined which medication to take depending on the severity of the AE. A direct line phone line to the oncology pharmacy also was supplied.
With the evolution to the PD-L1s and the anti-PD inhibitors, we haven’t seen the same level of AEs. Patients go home with wallet cards that includes our staff contact numbers/pagers. The wallet card also serves as information to a treating provider if the patient presents outside the VA, to ensure they understand the severity of a potential autoimmune AE, such as diarrhea.
Another challenge is shared-care patients. We have patients coming from outside hospitals, and at times they want to use this pharmacy like a CVS, and it just doesn’t operate that way. We want to collaborate with others. Most shared care patients present to our service for oral chemotherapy because the veteran just can’t afford the copays. So, we will see the patient concurrently. They can still see their outside hospital physician as well, but they have to fax us the laboratory results and progress notes on a monthly basis (or longer depending on where they are in there therapy). Before we fill their medications, we talk to the patients, the same way we would treat a veteran who was getting their oral chemotherapy here. In addition, they need to be seen by the VA physician at least every 3 months. We want our veterans to feel comfortable with the cancer care and help them out as best as we can.
Click here to read the digital edition.
Comorbidities
Joshua M. Bauml, MD, Corporal Michael J. Crescenz VAMC, Philadelphia, PA. One of the
In addition, kidney dysfunction is quite common as a result of comorbid cardiovascular and hypertensive diseases. Kidney dysfunction can negatively impact our ability to administer both cisplatin and other systemic therapies.
Millie Das, MD, Palo Alto Health Care System, CA. Another major comorbidity for a lot of our veterans is COPD (chronic obstructive pulmonary disease). It doesn’t complicate the chemotherapy choice, but it affects surgical candidacy for those patients who present with early stage disease. Many times if you obtain pulmonary function tests in patients with COPD, the tests are abnormal and can prohibit safe surgical resection. These are patients that I see in the clinic and refer for definitive radiation, usually SABR (stereotactic ablative radiotherapy)/SBRT (stereotactic body radiation therapy), at a local radiation facility that can offer specialized radiation treatment.
Dr. Bauml. The fact that the VA has so many patients who require stereotactic radiosurgery for their early stage lung cancer represents an opportunity. There is a newly opened study that is evaluating SBRT vs surgery for these early stage lung cancer patients within the VA system. That study model has previously failed in multiple health care settings, but the VA is uniquely suited to answer this question.
Kelly A. Tammaro, PharmD, BCOP, Boston VA Healthcare System, MA. I would add heart failure patients or patients who have cardiac comorbidities and fluid restrictions. These restrictions can affect hydration that is needed for cisplatin, for example, as well as final volumes used to mix other chemotherapeutic agents with narrow concentration maximums, such as etoposide.
Julie Beck, RN, MSN, MPH, APRN-BC, VA Connecticut Healthcare System West Haven Campus. As a lung cancer navigator, I find that psychosocial comorbidities are an impediment to getting patients to diagnosis and treatment. Patients will miss appointments because they don’t have rides or will be reluctant to get imaging or other diagnostic testing because of anxiety or because it triggers PTSD (posttraumatic stress disorder) or because they are concerned about cost.
Dr. Das. I couldn’t agree more.
Dr. Bauml. It’s a great point.
Ms. Beck. You have to think outside the box with this patient population. We treat patients from as far away as Western Massachusetts. We have a dedicated oncology social worker who helps to arrange transportation. We have our CLC ( community living center), which is a rehabilitation and hospice unit but is also a resource for patients who live alone or far away and are getting an aggressive daily treatment regimen such as combined chemotherapy and radiation. We admit some patients to the CLC during their treatment to ensure that they get their treatment on time, maintain their nutritional status, and to provide emotional support. This is not an acute medical bed. Patients will sometimes go home on the weekend, but the support of the CLC increases the chance that they will get through their treatment safely.
Cancer care requires a lot of handholding. We often have to make multiple telephone calls to persuade our patients to get imaging or biopsies. Some of our patients require admission following biopsy because they live alone and have no one to drive them home following the procedure.
Dr. Tammaro. Boston has a similar model. We have a social worker who is highly dedicated and is able address our patients needs immediately. We also have many patients with PTSD and other psychological comorbidities, and depending on the severity, may require admission for their treatment to avoid the overwhelming nature of the ambulatory setting. For those who have to travel long distances for treatment we the Huntington House, which is housing located next door to our ambulatory campus. This accommodation can be used by our patients and their caregivers. We also have long term care facilities and a hospice unit located at our Brockton facility.
Ms. Beck. In West Haven, we have both palliative care and health psychology providers embedded in our clinic. They assist with symptom management and issues related to coping with diagnosis, anxiety, sleep, pain, smoking cessation, and lifestyle changes. We have also been offering pet therapy through our social work team, which has been very helpful for many of our patients.
Dr. Bauml. Mental health issues also can affect the choice of the type of treatment. Patients who have severe claustrophobia associated with their PTSD may have difficulty undergoing radiation. This can impact their ability to comply with therapy, and we have to adjust the treatment accordingly. For instance, I have a patient who has a known brain metastasis that was treated with definitive intent, but this gentleman gets highly agitated doing a brain magnetic resonance image (MRI). Instead we have had to follow him with serial computed tomography (CAT) scans, which is suboptimal. We have discussed that, but the distress that it causes him is simply not worth it.
Dr. Das. In some instances, we have had to use IV sedation for some of our patients with severe claustrophobia just to be able to get them through a positron emission tomography (PET) scan as part of their staging workup. We discuss these types of challenging cases in a multidisciplinary setting in our thoracic tumor board in order to brainstorm and figure out a realistic plan with our radiology and anesthesia colleagues, with the goal of getting the patient through the necessary tests in order to establish a treatment recommendation.
Due to underlying mental health or other health issues, some of our patients may also have difficulty with breath holding or with following other necessary instructions during their radiation treatments. We sometimes have to get creative on an individual basis in order to help a patient get through the needed treatment.
We have a dedicated psychologist and social worker who are embedded in our clinics and work closely with the oncology providers to offer strategies that can help our patients comply and complete the recommended treatment plan.
Rural Care
Dr. Bauml. One of the questions that comes up frequently when you have a patient who is remote is the type of treatment that you can administer. It’s difficult to administer a weekly therapy if somebody’s traveling 3 hours to see you every time. That can play into your decision making as you’re choosing a chemotherapy. If there are equivalent treatment regimens and one involves visits every 3 weeks and one involves weekly visits, well, that will help sway your decision making after discussion with the patient.
We often have to balance things. For instance, when I give someone carboplatin and paclitaxel, my preference is to administer it weekly with 3 weeks on and 1 week off. However, if a patient tells me, “You know, I do not want to come in once a week,” then I will discuss with them my concern for the increased adverse effects (AEs) with the every-3-week dosing. We will do it and then watch them closely. Of course, this gets even more complicated when you consider the fact that many of these patients have multiple medical comorbidities, so you’d like to administer the treatments in the least toxic way possible.
Ms. Beck. We have overcome some of those challenges by partnering with the primary care doctors. We are very close to our primary care colleagues in Massachusetts. They will order labs for the patient the day before the patient's appointment, so if the patient has a long drive, we already have their lab work; and they are ready to go when they get here for their treatment. The nursing staff is very aware of who needs to get on a shuttle back to Massachusetts. For some patients, we will have them stay overnight before their treatment.
Precision Oncology
Dr. Tammaro. In Boston, we have integrated Precision Oncology to be part of clinical practice, which we started with metastatic lung cancer patients. The VA Precision Oncology Program (POP) began at our healthcare center. We had to evaluate the genetic testing platforms, the accuracy of the results, and amount of tissue necessary for the laboratories. We have since succeeded in sending high-quality samples to the laboratories that generate accurate results. However, for your standard mutation panel for identifying therapy for first line treatment in lung cancer, we still use our local send out laboratory.
The POP has rolled out nationwide, and it is another clinical tool, especially for patients who have already failed multiple lines of therapy. When we send for a precision oncology consult, the “N of 1” report provides annotation. The report will generate a review of relevant literature and provide available abstracts or phase 1 or 2 trials that support a targeted therapy against potential point mutation for your patient.
The POP also has a research component, known as Re-POP. The goal is to open bucket trials that assess targeted therapy off label. Re-POP allows us to recontact these patients in the future to say, “You had your tissue sent through precision oncology, and you were diagnosed with a certain point mutation. Now we have a clinical trial that’s available. Would you be interested?” The plan is to have those clinical trials open and available to our patients when we receive the results from precision oncology.
I have used POP for 2 metastatic prostate cancer patient who exhausted all lines of therapy in hopes to identify a potential BCRA 1/2 mutation in order for us to use a PARP inhibitor. Unfortunately, neither harbored this mutation. Precision oncology does not perform immunohistochemistry, therefore identifying HER-2 or PD-L1 status for example, would need to be done through your local laboratory. I have found POP to be helpful in identifying a patients potential therapeutic option after progression on first/second line therapy, by sending tissue to POP initially or at the time of relapse.
Dr. Das. In our clinical practice at the Palo Alto VA, we follow the National Comprehensive Cancer Network (NCCN) guidelines, and we routinely evaluate for the presence of an EGFR mutation and also for ALK and ROS1 translocations in all lung cancer patients with nonsquamous histology. We send our molecular testing through Quest Diagnostics (Madison, NJ), and we usually get results back within a week or so.
For those patients who do not have any of those targetable gene alterations, we will go ahead and send for next-generation sequencing through POP, which allows testing of a much broader gene panel. Those results can take about a month or so to come back. I usually don’t wait for these results in order to get someone started on treatment. For patients without EGFR, ALK, or ROS1 found on initial testing, I will go ahead and start them on IV systemic chemotherapy. It is often very useful when you do get the next-generation sequencing results back, since in almost all cases, a gene alteration can be detected and is provided in the accompanying report. In a large subset of lung cancer cases, a gene alteration is seen in KRAS, for which we still do not have an effective targeted therapy. Despite this, I still find it useful to obtain the results because we generally feel that the driving genetic alterations occur mutually exclusive of one another. When we do see KRAS reported from a patient’s tumor specimen, we’re not generally looking for other types of mutations, so I find it helpful to know what is the alteration that is driving the growth of a patient’s tumor. The trend moving forward is to perform next-generation sequencing on all tumor specimens regardless of tumor type or histology, which can hopefully enable us to get to the bottom of what the driving genetic alteration is and to see if there are any targeted treatment approaches that can be offered to the patient.
In a few lung cancer cases, I have seen alterations in HER2 and BRAF that have been detected and reported using a next-generation sequencing platform. Just recently the FDA approved the BRAF-directed therapies of dabrafenib and trametinib for patients with lung cancer who are found to have a BRAF V600E mutation. It is hoped that as the FDA continues to provide approvals for targeted drugs in patients with lung cancer, the VA formulary will be able to offer these therapies to our veteran patients with the ultimate goal of providing treatment that has increased efficacy and less toxicity compared to conventional IV chemotherapy.
One of my frustrations earlier on was when we did find these more rare targetable mutations, I would run into problems with the VA formulary in allowing me to prescribe certain targeted therapies. In many cases, if the drug was not FDA-approved for lung cancer, I was told that I couldn’t use it and would have to go through the appeal process, which was quite onerous. Moving forward, we are seeing more and more data and trials with newer targeted agents in lung cancer, leading to new FDA approvals. With these approvals, I think it will be easier to be able to offer these targeted therapies to our patients.
Dr. Bauml. One of the issues that arises when we’re discussing even the FDA-approved therapies, is that many of these targeted therapies are relatively rare, and they’re especially rare amongst veterans. Now others have mentioned BRAF and HER2, and these do have some overexpression and mutations that occur among smokers. But the more common targetable genetic aberrations, EGFR, ALK, and ROS1 are more common amongst never-smokers. Given the high prevalence of tobacco use among veterans, these changes are rare. The incidence of ALK translocation is 3% to 7%. The incidence amongst veterans is likely much lower than that, given the tobacco abuse—to the point that I actually had a patient who had an ALK translocation; and of course, I prescribed the patient crizotinib. This was prior to the ALEX Trial and alectinib data. I prescribed crizotinib and was told it wasn’t on the formulary. Initially I was surprised, but when I said, “Well, look, when was the last time someone within our VA has prescribed crizotinib?” The answer was never.
This is the difficulty: As we enter this era of molecularly targetable therapy, the way we structure our formularies and the way that we review these data is going to have to change. This year at the American Society of Clinical Oncology (ASCO) meeting there were some very exciting lung cancer abstracts that evaluated ado-trastuzumab emtansine, which is an antibody drug conjugate currently approved for the treatment of HER2 overexpressing breast cancer. The abstracts showed response rates of up to 40% in lung cancer with the administration of this drug in HER2-mutated lung cancer. The HER2-amplified still had a response rate of 20%, which given the toxicity profile of this agent, is quite appealing. Being able to explore these early phase studies, as was described through the personalized medicine pathway, is, a great step forward for VA care.
Dr. Tammaro. The PBM in collaboration with the POP Advisory Board, are developing different levels of evidence to support the use of targeted medications identified to be potential therapy in those diagnosed with a point mutation. Even if a medication does not have an FDA approval, it has to have some evidence to support its use in a particular cancer. If you identify a point mutation or biomarker in a patient and provide evidence to supports its use within that particular disease state, the VA pharmacy could approve its use based off of that evidence. VA pharmacy would not require an actual FDA approval for that indication.
What the VISNs, PBM, and precision oncology are trying to do is determine the level of evidence that we have to support or approve use of a targeted therapy. We are definitely moving forward and changing the horizon on how we actually treat our patients after they’ve gone through first-line therapy. We are trying to figure out where these point mutations come in, the line of the therapy, and how we actually treat these cancers. Pharmacy is making a step forward in conjunction with Michael Kelley, MD, the National Program Director for Oncology, Specialty Care Services, whose group is establishing those guidelines.
Dr. Bauml. I don’t mean to downplay the difficulty of that process. This is a huge, difficult process. One only needs to look at the long line of failed trials looking at PI3 kinase inhibitors to show that just knowing that a mutation exists does not necessarily mean that a targeted therapy works in that space.
Drawing that line is really complicated, both within the VA and, indeed, outside of the VA. It’s a really complicated process, and understanding the implications of different mutations is only going to get more complicated. Of course, now we have things like NTRK and even rarer genetic aberrations that are going to affect not only lung cancer, but also a wide range of malignancies.
Promising Research
Dr. Bauml. The pathways that are emerging as clear driver mutations for which we have available therapies, at least within lung cancer, are MET exon 14, RET, and NTRK. I am also intrigued by the emerging data in the HER2 space.
Dr. Das. The other therapy that has been getting a lot of press is immunotherapy, of course. And I’ve been seeing many really good responders to immunotherapy within the veteran population that I treat. It is felt that degree of PD-L1 expression correlates with responsiveness to the immune check point inhibitors that are being used in lung cancer, and we are tending to see higher rates of PD-L1 expression in patients who are prior or current smokers who have a higher overall tumor mutation burden.
I see patients both at Stanford and at the Palo Alto VA, and I have noticed that the patients that I have been treating at the VA tend to have higher levels of PD-L1 expression with better responses to the immunotherapy drugs, probably because most of the VA patients are former or current smokers. And, another interesting observation is that these veteran patients are, for whatever reason, having a lower incidence of some of the autoimmune AEs seen with these immune checkpoint inhibitors. I have been keeping an eye out for more data and information to support these observations I have had in my clinical practice and I specifically attended ASCO this year to learn more about what others have seen and studied with immune check point inhibition in lung cancer. We are learning now that PD-L1 is not a perfect marker for predicting response to the checkpoint inhibitors and the other immunotherapeutic agents, and there is a great deal of research going on to try to figure out what other biomarkers could be useful and which patients are most likely to benefit from these drugs.
I was excited to hear about the combination of nivolumab and ipilimumab that is being tested in both mesothelioma and in small-cell lung cancer where we really don’t have as many treatment options as we have in non-small cell lung cancer. That data was quite exciting, and interestingly, there does not seem to be a correlation with PD-L1 expression and responsiveness to treatment with the immunotherapeutic agents in those histologic subtypes. The story is still unfolding, and we await additional data to help guide us in our treatment decisions.
Dr. Tammaro. Immunotherapy is the new fad in oncology. We have just scheduled our first patient for first-line therapy due to PD-L1 tumor proportion score is > 50%. Recently, at ASCO KEYNOTE-021 researchers looked at using pembrolizumab in combination with carboplatin plus pemetrexed chemotherapy for first-line metastatic non-squamous NSCLC. The research suggested that patients treated with pembrolizumab + chemotherapy continued to derive a higher overall response rate and progression free survival when compare with those on chemotherapy alone despite a low or no PD-L1 tumor expression.
It’s very interesting that many clinical trials that we’re evaluating are now using some type of checkpoint inhibitor up front with cytotoxic chemotherapy. If they are positive trials, this could change how patients are treated up front.
Dr. Bauml. There was some really interesting data that were presented at ASCO this year by Matthew Hellmann, MD, which evaluated the predictive nature of PD-L1 vs tumor mutation burden and other biomarkers, including gene expression profiling. In this particular abstract, the PD-L1 and tumor mutation burden really do function as orthogonal biomarkers such that a patient who has high PD-L1 and high tumor mutation burden is the most likely to respond. Patients who are really low for both are unlikely to respond. We really need better biomarkers for immunotherapy, though. PD-L1 has a lot of limitations, namely, it is dynamic, so over time it changes. So I can do a biopsy at one point, then treat the patient and the PD-L1 may change.
More importantly, it’s heterogeneous. There was this great paper by McLaughlin and colleagues in JAMA Oncology (2016) who described a patient who had a small tumor biopsy. They took a micrograph of the tumor and showed that one part of the micrograph was completely floridly PD-L1 positive. At another site of the same biopsy it was completely stone-cold negative, which is humbling when you think about the fact that we stick small needles into tumors and make clinical decisions on the basis of that.
The KEYNOTE-024 study evaluated pembrolizumab vs chemotherapy in high PD-L1 expressers. It’s a very exciting study, but at the end of the day even in this highly select patient population, the response rate to immunotherapy was only about 50%, which is not the sort of biomarker-driven response that we’re used to seeing with our EGFR inhibitors. That’s really what we want to get to. More important even than that is being able to say the negative predictive value. One of the reasons that we’re probably seeing more responses among veterans is that we know that patients who are veterans who have high tobacco exposure have a higher tumor mutation burden. I’m surprised to hear about the immune-related AEs, actually, because one of the things that was reported this year at ASCO was some data that showed that patients who have immune-related AEs are more likely to have a better outcome, which is an interesting biomarker of response.
Dr. Das. I heard that as well, and I found that to be really interesting. The patients that I’ve had on nivolumab for over a year are doing very well. These are stage IV patients who have essentially had complete responses to treatment and have not had any or have had very minor immune-related AEs to date.
Overall, these are a small numbers of patients, but I have been curious to see why that might be the case. Anecdotally, my colleagues and I who treat patients at Stanford have seen significantly higher rates of grades 3 and 4 pneumonitis and other autoimmune toxicities, such as myocarditis and enterocolitis, in those lung cancer patients who are light or never-smokers treated with immune checkpoint inhibitors.
Dr. Bauml. I really feel that PD-L1 as a biomarker has significant limitations. I certainly hope that in at least 2 or 3 years we’re not going to be talking about PD-L1 anymore. I’m hopeful that we’ll be able to use better predictive biomarkers, such as mutational burden and gene expression profiling. In the data in head and neck that was presented this year at ASCO, patients who were low for both gene expression profiling and mutational burden had a very low response (Haddad et al, ASCO 2017).
That’s really what you want to be. You want to be able to say, “Here’s a person who will not benefit from this therapy.” From there you can identify, based upon these biomarkers, the combination that is going to be best for this person. Is it chemoimmunotherapy or combination immunotherapy with CTLA4, or another checkpoint blockade? That is really the way that we’re going to be able to fine-tune this, because the toxicity is substantial for some treatments, like the nivolumab/ipilimumab combination. Using them in a biomarker-blind fashion is just scary to me, honestly.
Managing Adverse Reactions
Dr. Tammaro. The increasing amount of oral chemotherapy has posed a significant challenge. As a clinical oncology pharmacist, it was difficult to grasp the most effective way to follow all these patients and ensure adherence, adverse drug event reporting/significance and adequate follow up. When patients are receiving IV chemotherapy, we know we will see them, we are assured compliance and are able to assess side effects in a timely manner. When we give oral chemotherapy, the tables are turned, where the responsibility is now on the patient. We are now depending on the patient to ensure they are taking the medication correctly and we may not see AEs if the patient misses an appointment or feels as though they are bothering the provider by calling.
In 2012, we started an oral oncology clinic here at the VA in Boston that I found to be extremely effective. When you’re sending a patient home with an oral chemotherapy, you have to make sure that you are counseling them correctly and encourage them to call at any time if they are experiencing any type of AE. One of the newest issues we have been seeing is bleeding with ibrutinib, especially in those patients on anticoagulation therapies.
A general strategy we employee for oral chemotherapy is to start at half dose and titrate slowly. This method has been effective in identifying AEs and preventing delays in therapy. We do this for the majority of oral chemotherapy. Patients are given a 2 week supply to start and then are reassessed on follow up for escalation to the target dose. We do not place refills on oral oncology prescriptions. They are instructed to call 10 days prior to running out if they are not scheduled to come in for an appointment. Having consistent dialogue with our patients allows us to assess for adherence, AEs, and tolerability. The other advantage to this clinic is ensuring our patients have someone to speak to at all times and answer all their questions. Direct lines of communication is what most of our patients are appreciative of when paying gratitude to the clinic.
Ms. Beck. We have an oral chemotherapy clinic staffed by dedicated oncology pharmacists. Patients meet with the pharmacist and have education prior to starting a new oral chemotherapy. They will then be followed by both the oncology provider and the pharmacist.
Dr. Das. One of the challenges we also face is with so many of our patients living so far away. When our patients do have AEs that require hospitalization, it can be very tricky to really get a sense for how they are being managed at the outside community (non-VA) facility. Sharing of electronic medical records can be a challenge in these cases, and I worry that the care teams at the more remote hospitals may not be as familiar with the newer cancer treatments and the toxicities associated with them, such as the autoimmune AEs associated with many of the immune checkpoint inhibitors.
I provide patients with pocket cards to keep in their wallets with my contact information and the name of the drug that they are getting because not all patients can remember or even pronounce the names of the drugs and may not be able to tell their local treating physician and care team what they are getting. I have been getting more frequent phone calls from emergency department physicians and hospitalists from the local communities where many of our veterans live, because they want guidance on how best to approach treatment for our patients when they show up with an AE related to their cancer treatment.
At times, the presenting symptoms may be vague or nonspecific, but for our patients being treated with immunotherapy, we always have to keep in mind the possibility of immune-related AEs because we know that prompt initiation of steroids is critical in these cases and can really help the patients feel better quickly.
Dr. Tammaro. You bring up a valid point. Our pharmacists meet with all the patients on checkpoint inhibitors. Specifically, when we started using ipilimumab it was uncharted territory for our team. We put together take home medication bag that included hydrocortisone cream, methylprednisolone dose pak, dipheydramine, and loperamide. This was utilized for all patients and specific attention was given to patients who lived far away from an emergency room. This bag system was accompanied by “what to do if I have this symptom” handout that outlined which medication to take depending on the severity of the AE. A direct line phone line to the oncology pharmacy also was supplied.
With the evolution to the PD-L1s and the anti-PD inhibitors, we haven’t seen the same level of AEs. Patients go home with wallet cards that includes our staff contact numbers/pagers. The wallet card also serves as information to a treating provider if the patient presents outside the VA, to ensure they understand the severity of a potential autoimmune AE, such as diarrhea.
Another challenge is shared-care patients. We have patients coming from outside hospitals, and at times they want to use this pharmacy like a CVS, and it just doesn’t operate that way. We want to collaborate with others. Most shared care patients present to our service for oral chemotherapy because the veteran just can’t afford the copays. So, we will see the patient concurrently. They can still see their outside hospital physician as well, but they have to fax us the laboratory results and progress notes on a monthly basis (or longer depending on where they are in there therapy). Before we fill their medications, we talk to the patients, the same way we would treat a veteran who was getting their oral chemotherapy here. In addition, they need to be seen by the VA physician at least every 3 months. We want our veterans to feel comfortable with the cancer care and help them out as best as we can.
Click here to read the digital edition.
Advanced practice nurses and physician assistants are not the same
Looking across a hospital ward, emergency department, or primary care clinic aligned side by side, you may not see any differences between an advanced practice nurse (APN) or physician assistant (PA). However, if you took a closer look at their education programs and credentialing, you would find considerable differences.
Although both professions hold advanced degrees, the approach to patient care differs, as well as the training they receive, including different models of practice. The APN is trained according to the nursing model, while the PA attends programs that are more in line with the medical model. The APN has a patient-centered model, while the PA adheres to a disease-centered model. Consequently, their approach to caring for the same patient population differs in viewpoint and philosophy.
Entry into the APN programs requires a nursing degree or related field from an accredited college or university. The curriculum includes coursework in health care policy, advocacy, outcomes, advanced assessment, diagnosis, and practice skills as well as, pharmacology, pathophysiology, and a final capstone project.
There are six specialty APN tracks including pediatrics, women’s/gender health, family practice, adult-gerontology, psychiatric, and neonatal. Additionally, there are three additional advanced practice registered nurses tracks: certified nurse anesthesia, certified nurse midwife, and clinical nurse leader. In addition to academic hours, there is a minimum of 1,000 supervised, direct patient care clinical hours in a variety of locations covering all populations specific to the identified specialty.
The Bureau of Labor Statistics defines the role of physician assistant as follows: “Physician assistants practice medicine under the supervision of physicians and surgeons. PAs are formally trained to provide diagnostic, therapeutic, and preventive health care services, as delegated by a physician.” The physician assistant program is a master’s prepared education.
School requirements include completing 2 years of pre-physician assistant undergraduate studies prior to applying to the School of Biomedical Sciences. Many programs have a 200-hour health care experience requirement, which can be either paid or unpaid. However, unlike the APN program, this is not required by all PA programs, but it is strongly encouraged.
Accredited PA programs require completing a 3-year graduate program that includes clinical rotations and results in a Master of Science in Physician Assistant Studies. Physician assistant programs typically involve 1,000 classroom hours and 2,000 or more hours in a clinical setting. The course work focuses on biochemistry, pathology, anatomy and physiology, ethics, and biology.
Both the APN and PA practices are regulated by the state through licensure laws and policy that determine the scope of practice and allow prescriptive authority.
Both programs began in 1965 in response to a shortage of primary care physicians, yet each program took a different route to address this need. According to the May 2017 Bureau of Labor Statistics, there were more than 109,000 physician assistants and more than 166,000 nurse practitioners practicing in the United States.
With the enactment of the Affordable Care Act in 2010, the mandate for APN’s and PA’s to lead patient-centered medical homes continued to grow to meet the demand. Both roles provide direct patient care under the sponsorship of a physician, yet both roles have gained a greater level of independence as state and federal requirements have relaxed restrictive physician collaboration and oversight rules, which has allowed both roles to practice at the highest level of their training. These relaxed restrictions come at a time when a growing physician shortage is met by increased demands placed on the health care system.
Ms. Thew is a certified family nurse practitioner in the division of adolescent medicine at the Medical College of Wisconsin, Milwaukee. Email her at pdnews@mdedge.com
Looking across a hospital ward, emergency department, or primary care clinic aligned side by side, you may not see any differences between an advanced practice nurse (APN) or physician assistant (PA). However, if you took a closer look at their education programs and credentialing, you would find considerable differences.
Although both professions hold advanced degrees, the approach to patient care differs, as well as the training they receive, including different models of practice. The APN is trained according to the nursing model, while the PA attends programs that are more in line with the medical model. The APN has a patient-centered model, while the PA adheres to a disease-centered model. Consequently, their approach to caring for the same patient population differs in viewpoint and philosophy.
Entry into the APN programs requires a nursing degree or related field from an accredited college or university. The curriculum includes coursework in health care policy, advocacy, outcomes, advanced assessment, diagnosis, and practice skills as well as, pharmacology, pathophysiology, and a final capstone project.
There are six specialty APN tracks including pediatrics, women’s/gender health, family practice, adult-gerontology, psychiatric, and neonatal. Additionally, there are three additional advanced practice registered nurses tracks: certified nurse anesthesia, certified nurse midwife, and clinical nurse leader. In addition to academic hours, there is a minimum of 1,000 supervised, direct patient care clinical hours in a variety of locations covering all populations specific to the identified specialty.
The Bureau of Labor Statistics defines the role of physician assistant as follows: “Physician assistants practice medicine under the supervision of physicians and surgeons. PAs are formally trained to provide diagnostic, therapeutic, and preventive health care services, as delegated by a physician.” The physician assistant program is a master’s prepared education.
School requirements include completing 2 years of pre-physician assistant undergraduate studies prior to applying to the School of Biomedical Sciences. Many programs have a 200-hour health care experience requirement, which can be either paid or unpaid. However, unlike the APN program, this is not required by all PA programs, but it is strongly encouraged.
Accredited PA programs require completing a 3-year graduate program that includes clinical rotations and results in a Master of Science in Physician Assistant Studies. Physician assistant programs typically involve 1,000 classroom hours and 2,000 or more hours in a clinical setting. The course work focuses on biochemistry, pathology, anatomy and physiology, ethics, and biology.
Both the APN and PA practices are regulated by the state through licensure laws and policy that determine the scope of practice and allow prescriptive authority.
Both programs began in 1965 in response to a shortage of primary care physicians, yet each program took a different route to address this need. According to the May 2017 Bureau of Labor Statistics, there were more than 109,000 physician assistants and more than 166,000 nurse practitioners practicing in the United States.
With the enactment of the Affordable Care Act in 2010, the mandate for APN’s and PA’s to lead patient-centered medical homes continued to grow to meet the demand. Both roles provide direct patient care under the sponsorship of a physician, yet both roles have gained a greater level of independence as state and federal requirements have relaxed restrictive physician collaboration and oversight rules, which has allowed both roles to practice at the highest level of their training. These relaxed restrictions come at a time when a growing physician shortage is met by increased demands placed on the health care system.
Ms. Thew is a certified family nurse practitioner in the division of adolescent medicine at the Medical College of Wisconsin, Milwaukee. Email her at pdnews@mdedge.com
Looking across a hospital ward, emergency department, or primary care clinic aligned side by side, you may not see any differences between an advanced practice nurse (APN) or physician assistant (PA). However, if you took a closer look at their education programs and credentialing, you would find considerable differences.
Although both professions hold advanced degrees, the approach to patient care differs, as well as the training they receive, including different models of practice. The APN is trained according to the nursing model, while the PA attends programs that are more in line with the medical model. The APN has a patient-centered model, while the PA adheres to a disease-centered model. Consequently, their approach to caring for the same patient population differs in viewpoint and philosophy.
Entry into the APN programs requires a nursing degree or related field from an accredited college or university. The curriculum includes coursework in health care policy, advocacy, outcomes, advanced assessment, diagnosis, and practice skills as well as, pharmacology, pathophysiology, and a final capstone project.
There are six specialty APN tracks including pediatrics, women’s/gender health, family practice, adult-gerontology, psychiatric, and neonatal. Additionally, there are three additional advanced practice registered nurses tracks: certified nurse anesthesia, certified nurse midwife, and clinical nurse leader. In addition to academic hours, there is a minimum of 1,000 supervised, direct patient care clinical hours in a variety of locations covering all populations specific to the identified specialty.
The Bureau of Labor Statistics defines the role of physician assistant as follows: “Physician assistants practice medicine under the supervision of physicians and surgeons. PAs are formally trained to provide diagnostic, therapeutic, and preventive health care services, as delegated by a physician.” The physician assistant program is a master’s prepared education.
School requirements include completing 2 years of pre-physician assistant undergraduate studies prior to applying to the School of Biomedical Sciences. Many programs have a 200-hour health care experience requirement, which can be either paid or unpaid. However, unlike the APN program, this is not required by all PA programs, but it is strongly encouraged.
Accredited PA programs require completing a 3-year graduate program that includes clinical rotations and results in a Master of Science in Physician Assistant Studies. Physician assistant programs typically involve 1,000 classroom hours and 2,000 or more hours in a clinical setting. The course work focuses on biochemistry, pathology, anatomy and physiology, ethics, and biology.
Both the APN and PA practices are regulated by the state through licensure laws and policy that determine the scope of practice and allow prescriptive authority.
Both programs began in 1965 in response to a shortage of primary care physicians, yet each program took a different route to address this need. According to the May 2017 Bureau of Labor Statistics, there were more than 109,000 physician assistants and more than 166,000 nurse practitioners practicing in the United States.
With the enactment of the Affordable Care Act in 2010, the mandate for APN’s and PA’s to lead patient-centered medical homes continued to grow to meet the demand. Both roles provide direct patient care under the sponsorship of a physician, yet both roles have gained a greater level of independence as state and federal requirements have relaxed restrictive physician collaboration and oversight rules, which has allowed both roles to practice at the highest level of their training. These relaxed restrictions come at a time when a growing physician shortage is met by increased demands placed on the health care system.
Ms. Thew is a certified family nurse practitioner in the division of adolescent medicine at the Medical College of Wisconsin, Milwaukee. Email her at pdnews@mdedge.com
Interdisciplinary Geriatric Difficult Case Conference: Innovative Education Across the Continuum
From Wheaton Franciscan Healthcare (Ms. Fedel), Aspirus (Ms. Hackbarth), and Aurora Health Care (Mr. Malsch and Ms. Pagel).
Abstract
- Background: There is a nationwide shortage of geriatric prepared providers. Caring for complex older adults is challenging.
- Objective: To develop an efficient and affordable way to educate members of the interdisciplinary team involved in the care of geriatric patients.
- Methods: A team from 3 area health systems developed a plan to present monthly case studies via teleconference. Cases are presented by a direct caregiver using the Wisconsin Star Method to facilitate analysis of the case. A geriatric expert and another member of the team presents teaching points, and questions are elicited and discussed.
- Results: The team has completed 18 consecutive monthly teleconferences. Participant satisfaction has been favorable. Participation on the call has increased approximately 300% since the initiation of the program.
- Conclusion: The case teleconference provides an accessible and affordable educational forum that provides learners an opportunity to improve their knowledge in care of older adults.
The number of older adults in the United States will nearly double between 2005 and 2030 [1] as the baby boom generation begins turning 65 and as life expectancy for older Americans increases. The Institute of Medicine’s (IOM) landmark report Retooling for an Aging America: Building the Health Care Workforce states that “unless action is taken immediately, the health care workforce will lack the capacity (in both size and ability) to meet the needs of older patients in the future [1].” One of their recommendations is to explore ways to widen the duties and responsibilities of workers at various levels of training. More health care providers need to be trained in the basics of geriatric care and should be capable of caring for older patients.
Team-based care is becoming more prevalent. Care delivered by interdisciplinary teams have been shown to improve patient outcomes [2]. A team led by one of the authors (PF) developed an intervention to increase the geriatric and teamwork competencies of interdisciplinary teams who serve patients throughout Wisconsin. The Interdisciplinary Geriatric Difficult Case Conference Call (IGDCC) is sponsored monthly by 3 Wisconsin health systems. The purpose is to provide opportunities to discuss clinical cases, to learn from one another and from experts, and to elevate the level of geriatric care in the states of Wisconsin, Michigan, and beyond. Each month a difficult case is presented by a clinician involved in that patient’s care. Time is allotted for participants to ask questions, and teaching points are shared by a clinical expert to highlight concepts and provide additional context. The IGDCC is meant to be a joint learning exercise to explore a specific difficult patient situation and learn skills and knowledge to improve care and transitions for older adults. The conference call is not a critique of the care, but rather an opportunity to jointly learn from the challenging situations all experience.
Background
The IGDCC was created by four members of 3 health systems in Wisconsin: Wheaton Franciscan Healthcare, Aspirus, and Aurora Health Care. The health systems serve and partially overlap on a broad geographic and demographic area of Wisconsin. The 4 members collaborated on numerous projects in the past, including Nurses Improving Case for Health System Elders (NICHE) implementation [3]. A common concern among the team is the management of challenging geriatric clinical patients and having a prepared workforce to meet those challenges.
Problem/Issue
As mentioned above, the older adult population is increasing, and these statistics are reflected in our service area [4]. Exacerbating these demographic changes is a shortage of health care workers in all disciplines, inadequate geriatric training, and the increased prevalence of multiple chronic conditions. Older adults also have higher rates of 30-day readmissions as well as higher rates of functional decline and medical errors during hospital stays [5,6]. Effective interprofessional teamwork is essential for the delivery of high-quality patient care in an increasingly complex health environment [7]. The IOM’s Future of Nursing report recommends that nurses, who represent the largest segment of the US health workforce, should achieve higher levels of training and be full partners in redesigning health care [8]. Unfortunately, effective care is hampered by poor coordination, limited communication, boundary infringement, and lack of understanding of roles [9]. Meta-analyses have demonstrated that there is a positive relationship between team training interventions and outcomes [10,11].
Objectives
The objective of the IGDCC is to elevate the level of geriatric care in the region by providing an accessible and affordable forum for the education of health care workers involved in the care of our most vulnerable population. To meet this challenge, the 4 founding members of IGDCC utilized the Aurora Health Care Geriatric Fellow’s Most Difficult Case (GFMCC) conference format as a model [12,13]. All disciplines are encouraged to participate, with announcements sent out via the leadership at the participating hospital systems. Participants have the option to call into the conference and teleconference via their own personal telephone and computer; in addition, each participating hospital system frequently hosts an open forum teleconference room where participants also may join a group.
Conference Components
The team uses the Wisconsin Star Method framework for presentation and discussion of the case. The Star Method, developed by Timothy Howell, enables clinical data about a person to be mapped out onto a single field with 5 domains: medications, medical, behavioral, personal, and social [14], creating a visual representation of the complicated and interacting physical, emotional, and social issues of older adults (Figure). By becoming comfortable using this method, the learner can use a similar approach in their clinical practice to address the needs of the patient in a holistic manner.
The case call concludes with expert teaching points from both a geriatric expert and a member of the interdisciplinary team. The interdisciplinary team member is chosen based on the key issues raised by the case. For example, cases that are made complex due to polypharmacy and adverse drug reactions might have a pharmacist presenting pertinent take-home message for the learner. In addition, geriatric teaching experts (ie, a geriatrician or advanced practice geriatric nursing specialist) provide the learner with insights that they can apply to their future practice. Often times the teaching points consist of an analysis of the various geriatric syndromes and how they can be managed in the complex older adult.
Implementation
Implementation of the IGDCC is coordinated by an oversight team with representation from each of the 3 sponsoring health systems. The oversight team currently includes 4 members: 3 geriatric clinical nurse specialists and a geriatric service line administrator. The team is responsible for:
- Planning the conference call schedule
- Making arrangements for case presenters and experts to contribute teaching points
- Registering participants and sharing written materials with participants
- Publicizing and encouraging attendance
- Soliciting feedback for continual improvement
- Exploring and implementing new ways to maximize learning.
Team members share duties and rotate case presentations. The Aurora and Wheaton Franciscan systems provide the geriatric specialists who provide the expert teaching points. The Aspirus system provides the conference line and webinar application and supports publicity and evaluations. All 3 systems are supported by a geriatric clinical nurse specialist who identifies and helps prepare presenters, case presentations, and call participants. Over time, the conference call format has evolved into a webinar format, allowing participants to either phone into the call for audio only or participate via both audio and visual. The visual allows participants to watch on their computer screens while the case is presented using the Star Method. During the call, a member of the oversight team adds clinical details by typing into a Word template of a blank star, adding information for each of the 5 domains in real-time as the case is discussed. Another member of the team facilitates the call, introducing presenters and experts, describing the Star Method, and offering “housekeeping” announcements. The facilitator also watches the timing to make certain the agenda is followed and the call begins and ends on time. During the call, another member of the team updates the attendance spreadsheet and makes a recording of each session.
Some participating facilities reserve a meeting room and project the webinar onto a screen for shared viewing. One of the participating sites has done this quite successfully with a growing group of participants coming together to watch the case during their lunch hour. This allows an opportunity for group discussion—when the conference call is on “mute” so as not to disrupt learners at other locations.
Measurement/Analysis
Attendance has steadily increased. In CY2015 from January to September, the mean attendance per month was 29.1 (mode, 17). The maximum per month was 62 (September 2015). The program enjoyed a boost in attendance beginning in July 2015 when Nurses Improving Care of Healthsystem Elders (NICHE) [3] began promoting the call-in opportunity to its NICHE Coordinators at member health systems. In June 2015, the technology was improved to allow for recorded sessions, and the recordings are growing in popularity from 2 listeners per month in July 2014 to 23 listeners per month in September 2015.
Lessons Learned
In comparing the IGDCC with similar conference call educational offerings, the team found that the program was unique in 2 areas. First, in addition to having a rich discussion in the care of frail older adults with experts in the field, the team also sought to help our staff learn how to present a difficult case to their peers. Three of our 4 committee members are geriatric clinical nurse specialists (a fourth is a clinical nurse specialist from Aspirus who assists periodically) who have been able to mentor, guide, and encourage interdisciplinary team members to present a challenging case. Many presenters had never presented a difficult case in this format. Presenters found the process fun and rewarding and have offered to present cases again in the future.
A second unique feature was utilizing the Wisconsin Star Method rather than focusing on a typical medical model framework for discussing a challenging case. The Star Method allows participants to increase their proficiency in providing comprehensive care while being more confident and mindful in addressing the complicated interacting physical, emotional and social issues of older adults [13].
A monthly post-call debriefing with committee members to review the strengths and weakness of the call was key to growing the program. The committee was able to critically review the process of the call, review participant surveys and discuss next steps. Adding a webinar approach, automatic email notification of calls, participant electronic survey, recording the call, and the addition of offering contact hours were some of the action items that were a result of monthly debriefing calls.
The team also found the 3-system collaboration to be beneficial. Aspirus has a large rural population, and Wheaton and Aurora have a diverse population, and each adds to the participant’s experience. Each IGDCC was rotated between the systems, which did not put the burden on any one health system. An annual call assignment listing was maintained for noting which system was responsible for the case each month and whether the geriatric expert was assigned/confirmed. Identifying the committee’s individual and collective group expertise was helpful in the overall project planning. The committee also developed a standard presenter guide and template and an expert teaching guide so the monthly IGDCC were consistent.
Challenges
The committee did not have a budget. Participation on the committee was in-kind funding from each system. Aspirus used its electronic system in place at the time to support the project. Interactive conference call education platform can be challenging with multiple participants on an open line who may not mute their phone. Often times, when a group of participants are calling in from one phone line it is difficult to know how many people are attending the IGDCC. It can be challenging at times to facilitate the call during the discussion component as participants occasionally talk over each other.
Current Status/Future Directions
The team has completed 18 consecutive monthly IGDCCs. Our participation rate has tripled. Participant satisfaction remains favorable. The team is now offering 1 contact hour to participants, and our invitations to participate have been extended to national health care groups. Challenging cases will be presented from community sources outside the hospital. Focusing attention on elevating the level of geriatric care in our region using a community educational approach will give us new opportunities for collaborating on best practice in multiple settings across the care continuum.
Acknowledgment: The planning team acknowledges Evalyn Michira, MSN, RN, PHN, AGCNS-BC, for her assistance in call presentations.
Corresponding author: Margie Hackbarth, MBA, margie.hackbarth@aspirus.org.
Financial disclosures: none.
1. Institute of Medicine. Retooling for an aging America: Building the health care workforce. Washington, DC: National Academies Press; 2008.
2. Mitchell P, Wynia M, Golden R, et al. Core principles and values of effective team-based health care. Discussion paper. Washington, DC; Institute of Medicine; 2012.
3. Nurses Improving Care for Healthsystem Elders. Accessed 1 Dec 2015 at www.nicheprogram.org/.
4. Wisconsin Department of Health Services. Southeastern region population report: 1 Jul 2013. Accessed 16 Feb 2015 at www.dhs.wisconsin.gov/sites/default/files/legacy/population/13data/southeastern.pdf.
5. From the Centers for Disease Control and Prevention. Public health and aging: trends in aging--United States and worldwide. JAMA 2003;289:1371–3.
6. Hall MJ, DeFrances CJ, Williams SN, et al. National Hospital Discharge Survey: 2007 summary. Natl Health Stat Report 2010;(29):1–20, 24.
7. Nembhard IM, Edmondson AC. Making it safe: The effects of leader inclusiveness and professional status on psychological safety and improvement efforts in health care teams. J Organiz Behav 2006; 27:941–66.
8. Institute of Medicine. The future of nursing: leading change, advancing health. National Academies Press; 2011.
9. Reeves S, Zwarenstein M, Goldman et al. Interprofessional education: effects on professional practice and health care outcomes. Cochrane Database Syst Rev 2013;3:CD002213.
10. Salas E, Diaz Granados D, Klein C, et al. Does team training improve team performance? A meta-analysis. Hum Factors 2008;50:903–33.
11. Strasser DD, Burridge AB, Falconer JA, et al. Toward spanning the quality chasm: an examination of team functioning measures. Arch Phys Med Rehabil 2014;95:2220–3.
12. Roche VM, Torregosa H, Howell T, Malone ML. Establishing a treatment plan for an elder with a complex and incomplete medical history and multiple medical providers, diagnoses, and medications. Ann Long-Term Care 2012;20(9).
13. Roche VM, Arnouville J, Danto-Nocton ES, et al. Optimal management of an older patient with multiple comorbidities and a complex psychosocial history. Ann Long-Term Care 2011;19(9).
14. Wisconsin Geriatric Psychiatry Initiative. The Wisconsin Star Method. Accessed 19 Jan 2015 at wgpi.wisc.edu/wisconsin-star-method/.
From Wheaton Franciscan Healthcare (Ms. Fedel), Aspirus (Ms. Hackbarth), and Aurora Health Care (Mr. Malsch and Ms. Pagel).
Abstract
- Background: There is a nationwide shortage of geriatric prepared providers. Caring for complex older adults is challenging.
- Objective: To develop an efficient and affordable way to educate members of the interdisciplinary team involved in the care of geriatric patients.
- Methods: A team from 3 area health systems developed a plan to present monthly case studies via teleconference. Cases are presented by a direct caregiver using the Wisconsin Star Method to facilitate analysis of the case. A geriatric expert and another member of the team presents teaching points, and questions are elicited and discussed.
- Results: The team has completed 18 consecutive monthly teleconferences. Participant satisfaction has been favorable. Participation on the call has increased approximately 300% since the initiation of the program.
- Conclusion: The case teleconference provides an accessible and affordable educational forum that provides learners an opportunity to improve their knowledge in care of older adults.
The number of older adults in the United States will nearly double between 2005 and 2030 [1] as the baby boom generation begins turning 65 and as life expectancy for older Americans increases. The Institute of Medicine’s (IOM) landmark report Retooling for an Aging America: Building the Health Care Workforce states that “unless action is taken immediately, the health care workforce will lack the capacity (in both size and ability) to meet the needs of older patients in the future [1].” One of their recommendations is to explore ways to widen the duties and responsibilities of workers at various levels of training. More health care providers need to be trained in the basics of geriatric care and should be capable of caring for older patients.
Team-based care is becoming more prevalent. Care delivered by interdisciplinary teams have been shown to improve patient outcomes [2]. A team led by one of the authors (PF) developed an intervention to increase the geriatric and teamwork competencies of interdisciplinary teams who serve patients throughout Wisconsin. The Interdisciplinary Geriatric Difficult Case Conference Call (IGDCC) is sponsored monthly by 3 Wisconsin health systems. The purpose is to provide opportunities to discuss clinical cases, to learn from one another and from experts, and to elevate the level of geriatric care in the states of Wisconsin, Michigan, and beyond. Each month a difficult case is presented by a clinician involved in that patient’s care. Time is allotted for participants to ask questions, and teaching points are shared by a clinical expert to highlight concepts and provide additional context. The IGDCC is meant to be a joint learning exercise to explore a specific difficult patient situation and learn skills and knowledge to improve care and transitions for older adults. The conference call is not a critique of the care, but rather an opportunity to jointly learn from the challenging situations all experience.
Background
The IGDCC was created by four members of 3 health systems in Wisconsin: Wheaton Franciscan Healthcare, Aspirus, and Aurora Health Care. The health systems serve and partially overlap on a broad geographic and demographic area of Wisconsin. The 4 members collaborated on numerous projects in the past, including Nurses Improving Case for Health System Elders (NICHE) implementation [3]. A common concern among the team is the management of challenging geriatric clinical patients and having a prepared workforce to meet those challenges.
Problem/Issue
As mentioned above, the older adult population is increasing, and these statistics are reflected in our service area [4]. Exacerbating these demographic changes is a shortage of health care workers in all disciplines, inadequate geriatric training, and the increased prevalence of multiple chronic conditions. Older adults also have higher rates of 30-day readmissions as well as higher rates of functional decline and medical errors during hospital stays [5,6]. Effective interprofessional teamwork is essential for the delivery of high-quality patient care in an increasingly complex health environment [7]. The IOM’s Future of Nursing report recommends that nurses, who represent the largest segment of the US health workforce, should achieve higher levels of training and be full partners in redesigning health care [8]. Unfortunately, effective care is hampered by poor coordination, limited communication, boundary infringement, and lack of understanding of roles [9]. Meta-analyses have demonstrated that there is a positive relationship between team training interventions and outcomes [10,11].
Objectives
The objective of the IGDCC is to elevate the level of geriatric care in the region by providing an accessible and affordable forum for the education of health care workers involved in the care of our most vulnerable population. To meet this challenge, the 4 founding members of IGDCC utilized the Aurora Health Care Geriatric Fellow’s Most Difficult Case (GFMCC) conference format as a model [12,13]. All disciplines are encouraged to participate, with announcements sent out via the leadership at the participating hospital systems. Participants have the option to call into the conference and teleconference via their own personal telephone and computer; in addition, each participating hospital system frequently hosts an open forum teleconference room where participants also may join a group.
Conference Components
The team uses the Wisconsin Star Method framework for presentation and discussion of the case. The Star Method, developed by Timothy Howell, enables clinical data about a person to be mapped out onto a single field with 5 domains: medications, medical, behavioral, personal, and social [14], creating a visual representation of the complicated and interacting physical, emotional, and social issues of older adults (Figure). By becoming comfortable using this method, the learner can use a similar approach in their clinical practice to address the needs of the patient in a holistic manner.
The case call concludes with expert teaching points from both a geriatric expert and a member of the interdisciplinary team. The interdisciplinary team member is chosen based on the key issues raised by the case. For example, cases that are made complex due to polypharmacy and adverse drug reactions might have a pharmacist presenting pertinent take-home message for the learner. In addition, geriatric teaching experts (ie, a geriatrician or advanced practice geriatric nursing specialist) provide the learner with insights that they can apply to their future practice. Often times the teaching points consist of an analysis of the various geriatric syndromes and how they can be managed in the complex older adult.
Implementation
Implementation of the IGDCC is coordinated by an oversight team with representation from each of the 3 sponsoring health systems. The oversight team currently includes 4 members: 3 geriatric clinical nurse specialists and a geriatric service line administrator. The team is responsible for:
- Planning the conference call schedule
- Making arrangements for case presenters and experts to contribute teaching points
- Registering participants and sharing written materials with participants
- Publicizing and encouraging attendance
- Soliciting feedback for continual improvement
- Exploring and implementing new ways to maximize learning.
Team members share duties and rotate case presentations. The Aurora and Wheaton Franciscan systems provide the geriatric specialists who provide the expert teaching points. The Aspirus system provides the conference line and webinar application and supports publicity and evaluations. All 3 systems are supported by a geriatric clinical nurse specialist who identifies and helps prepare presenters, case presentations, and call participants. Over time, the conference call format has evolved into a webinar format, allowing participants to either phone into the call for audio only or participate via both audio and visual. The visual allows participants to watch on their computer screens while the case is presented using the Star Method. During the call, a member of the oversight team adds clinical details by typing into a Word template of a blank star, adding information for each of the 5 domains in real-time as the case is discussed. Another member of the team facilitates the call, introducing presenters and experts, describing the Star Method, and offering “housekeeping” announcements. The facilitator also watches the timing to make certain the agenda is followed and the call begins and ends on time. During the call, another member of the team updates the attendance spreadsheet and makes a recording of each session.
Some participating facilities reserve a meeting room and project the webinar onto a screen for shared viewing. One of the participating sites has done this quite successfully with a growing group of participants coming together to watch the case during their lunch hour. This allows an opportunity for group discussion—when the conference call is on “mute” so as not to disrupt learners at other locations.
Measurement/Analysis
Attendance has steadily increased. In CY2015 from January to September, the mean attendance per month was 29.1 (mode, 17). The maximum per month was 62 (September 2015). The program enjoyed a boost in attendance beginning in July 2015 when Nurses Improving Care of Healthsystem Elders (NICHE) [3] began promoting the call-in opportunity to its NICHE Coordinators at member health systems. In June 2015, the technology was improved to allow for recorded sessions, and the recordings are growing in popularity from 2 listeners per month in July 2014 to 23 listeners per month in September 2015.
Lessons Learned
In comparing the IGDCC with similar conference call educational offerings, the team found that the program was unique in 2 areas. First, in addition to having a rich discussion in the care of frail older adults with experts in the field, the team also sought to help our staff learn how to present a difficult case to their peers. Three of our 4 committee members are geriatric clinical nurse specialists (a fourth is a clinical nurse specialist from Aspirus who assists periodically) who have been able to mentor, guide, and encourage interdisciplinary team members to present a challenging case. Many presenters had never presented a difficult case in this format. Presenters found the process fun and rewarding and have offered to present cases again in the future.
A second unique feature was utilizing the Wisconsin Star Method rather than focusing on a typical medical model framework for discussing a challenging case. The Star Method allows participants to increase their proficiency in providing comprehensive care while being more confident and mindful in addressing the complicated interacting physical, emotional and social issues of older adults [13].
A monthly post-call debriefing with committee members to review the strengths and weakness of the call was key to growing the program. The committee was able to critically review the process of the call, review participant surveys and discuss next steps. Adding a webinar approach, automatic email notification of calls, participant electronic survey, recording the call, and the addition of offering contact hours were some of the action items that were a result of monthly debriefing calls.
The team also found the 3-system collaboration to be beneficial. Aspirus has a large rural population, and Wheaton and Aurora have a diverse population, and each adds to the participant’s experience. Each IGDCC was rotated between the systems, which did not put the burden on any one health system. An annual call assignment listing was maintained for noting which system was responsible for the case each month and whether the geriatric expert was assigned/confirmed. Identifying the committee’s individual and collective group expertise was helpful in the overall project planning. The committee also developed a standard presenter guide and template and an expert teaching guide so the monthly IGDCC were consistent.
Challenges
The committee did not have a budget. Participation on the committee was in-kind funding from each system. Aspirus used its electronic system in place at the time to support the project. Interactive conference call education platform can be challenging with multiple participants on an open line who may not mute their phone. Often times, when a group of participants are calling in from one phone line it is difficult to know how many people are attending the IGDCC. It can be challenging at times to facilitate the call during the discussion component as participants occasionally talk over each other.
Current Status/Future Directions
The team has completed 18 consecutive monthly IGDCCs. Our participation rate has tripled. Participant satisfaction remains favorable. The team is now offering 1 contact hour to participants, and our invitations to participate have been extended to national health care groups. Challenging cases will be presented from community sources outside the hospital. Focusing attention on elevating the level of geriatric care in our region using a community educational approach will give us new opportunities for collaborating on best practice in multiple settings across the care continuum.
Acknowledgment: The planning team acknowledges Evalyn Michira, MSN, RN, PHN, AGCNS-BC, for her assistance in call presentations.
Corresponding author: Margie Hackbarth, MBA, margie.hackbarth@aspirus.org.
Financial disclosures: none.
From Wheaton Franciscan Healthcare (Ms. Fedel), Aspirus (Ms. Hackbarth), and Aurora Health Care (Mr. Malsch and Ms. Pagel).
Abstract
- Background: There is a nationwide shortage of geriatric prepared providers. Caring for complex older adults is challenging.
- Objective: To develop an efficient and affordable way to educate members of the interdisciplinary team involved in the care of geriatric patients.
- Methods: A team from 3 area health systems developed a plan to present monthly case studies via teleconference. Cases are presented by a direct caregiver using the Wisconsin Star Method to facilitate analysis of the case. A geriatric expert and another member of the team presents teaching points, and questions are elicited and discussed.
- Results: The team has completed 18 consecutive monthly teleconferences. Participant satisfaction has been favorable. Participation on the call has increased approximately 300% since the initiation of the program.
- Conclusion: The case teleconference provides an accessible and affordable educational forum that provides learners an opportunity to improve their knowledge in care of older adults.
The number of older adults in the United States will nearly double between 2005 and 2030 [1] as the baby boom generation begins turning 65 and as life expectancy for older Americans increases. The Institute of Medicine’s (IOM) landmark report Retooling for an Aging America: Building the Health Care Workforce states that “unless action is taken immediately, the health care workforce will lack the capacity (in both size and ability) to meet the needs of older patients in the future [1].” One of their recommendations is to explore ways to widen the duties and responsibilities of workers at various levels of training. More health care providers need to be trained in the basics of geriatric care and should be capable of caring for older patients.
Team-based care is becoming more prevalent. Care delivered by interdisciplinary teams have been shown to improve patient outcomes [2]. A team led by one of the authors (PF) developed an intervention to increase the geriatric and teamwork competencies of interdisciplinary teams who serve patients throughout Wisconsin. The Interdisciplinary Geriatric Difficult Case Conference Call (IGDCC) is sponsored monthly by 3 Wisconsin health systems. The purpose is to provide opportunities to discuss clinical cases, to learn from one another and from experts, and to elevate the level of geriatric care in the states of Wisconsin, Michigan, and beyond. Each month a difficult case is presented by a clinician involved in that patient’s care. Time is allotted for participants to ask questions, and teaching points are shared by a clinical expert to highlight concepts and provide additional context. The IGDCC is meant to be a joint learning exercise to explore a specific difficult patient situation and learn skills and knowledge to improve care and transitions for older adults. The conference call is not a critique of the care, but rather an opportunity to jointly learn from the challenging situations all experience.
Background
The IGDCC was created by four members of 3 health systems in Wisconsin: Wheaton Franciscan Healthcare, Aspirus, and Aurora Health Care. The health systems serve and partially overlap on a broad geographic and demographic area of Wisconsin. The 4 members collaborated on numerous projects in the past, including Nurses Improving Case for Health System Elders (NICHE) implementation [3]. A common concern among the team is the management of challenging geriatric clinical patients and having a prepared workforce to meet those challenges.
Problem/Issue
As mentioned above, the older adult population is increasing, and these statistics are reflected in our service area [4]. Exacerbating these demographic changes is a shortage of health care workers in all disciplines, inadequate geriatric training, and the increased prevalence of multiple chronic conditions. Older adults also have higher rates of 30-day readmissions as well as higher rates of functional decline and medical errors during hospital stays [5,6]. Effective interprofessional teamwork is essential for the delivery of high-quality patient care in an increasingly complex health environment [7]. The IOM’s Future of Nursing report recommends that nurses, who represent the largest segment of the US health workforce, should achieve higher levels of training and be full partners in redesigning health care [8]. Unfortunately, effective care is hampered by poor coordination, limited communication, boundary infringement, and lack of understanding of roles [9]. Meta-analyses have demonstrated that there is a positive relationship between team training interventions and outcomes [10,11].
Objectives
The objective of the IGDCC is to elevate the level of geriatric care in the region by providing an accessible and affordable forum for the education of health care workers involved in the care of our most vulnerable population. To meet this challenge, the 4 founding members of IGDCC utilized the Aurora Health Care Geriatric Fellow’s Most Difficult Case (GFMCC) conference format as a model [12,13]. All disciplines are encouraged to participate, with announcements sent out via the leadership at the participating hospital systems. Participants have the option to call into the conference and teleconference via their own personal telephone and computer; in addition, each participating hospital system frequently hosts an open forum teleconference room where participants also may join a group.
Conference Components
The team uses the Wisconsin Star Method framework for presentation and discussion of the case. The Star Method, developed by Timothy Howell, enables clinical data about a person to be mapped out onto a single field with 5 domains: medications, medical, behavioral, personal, and social [14], creating a visual representation of the complicated and interacting physical, emotional, and social issues of older adults (Figure). By becoming comfortable using this method, the learner can use a similar approach in their clinical practice to address the needs of the patient in a holistic manner.
The case call concludes with expert teaching points from both a geriatric expert and a member of the interdisciplinary team. The interdisciplinary team member is chosen based on the key issues raised by the case. For example, cases that are made complex due to polypharmacy and adverse drug reactions might have a pharmacist presenting pertinent take-home message for the learner. In addition, geriatric teaching experts (ie, a geriatrician or advanced practice geriatric nursing specialist) provide the learner with insights that they can apply to their future practice. Often times the teaching points consist of an analysis of the various geriatric syndromes and how they can be managed in the complex older adult.
Implementation
Implementation of the IGDCC is coordinated by an oversight team with representation from each of the 3 sponsoring health systems. The oversight team currently includes 4 members: 3 geriatric clinical nurse specialists and a geriatric service line administrator. The team is responsible for:
- Planning the conference call schedule
- Making arrangements for case presenters and experts to contribute teaching points
- Registering participants and sharing written materials with participants
- Publicizing and encouraging attendance
- Soliciting feedback for continual improvement
- Exploring and implementing new ways to maximize learning.
Team members share duties and rotate case presentations. The Aurora and Wheaton Franciscan systems provide the geriatric specialists who provide the expert teaching points. The Aspirus system provides the conference line and webinar application and supports publicity and evaluations. All 3 systems are supported by a geriatric clinical nurse specialist who identifies and helps prepare presenters, case presentations, and call participants. Over time, the conference call format has evolved into a webinar format, allowing participants to either phone into the call for audio only or participate via both audio and visual. The visual allows participants to watch on their computer screens while the case is presented using the Star Method. During the call, a member of the oversight team adds clinical details by typing into a Word template of a blank star, adding information for each of the 5 domains in real-time as the case is discussed. Another member of the team facilitates the call, introducing presenters and experts, describing the Star Method, and offering “housekeeping” announcements. The facilitator also watches the timing to make certain the agenda is followed and the call begins and ends on time. During the call, another member of the team updates the attendance spreadsheet and makes a recording of each session.
Some participating facilities reserve a meeting room and project the webinar onto a screen for shared viewing. One of the participating sites has done this quite successfully with a growing group of participants coming together to watch the case during their lunch hour. This allows an opportunity for group discussion—when the conference call is on “mute” so as not to disrupt learners at other locations.
Measurement/Analysis
Attendance has steadily increased. In CY2015 from January to September, the mean attendance per month was 29.1 (mode, 17). The maximum per month was 62 (September 2015). The program enjoyed a boost in attendance beginning in July 2015 when Nurses Improving Care of Healthsystem Elders (NICHE) [3] began promoting the call-in opportunity to its NICHE Coordinators at member health systems. In June 2015, the technology was improved to allow for recorded sessions, and the recordings are growing in popularity from 2 listeners per month in July 2014 to 23 listeners per month in September 2015.
Lessons Learned
In comparing the IGDCC with similar conference call educational offerings, the team found that the program was unique in 2 areas. First, in addition to having a rich discussion in the care of frail older adults with experts in the field, the team also sought to help our staff learn how to present a difficult case to their peers. Three of our 4 committee members are geriatric clinical nurse specialists (a fourth is a clinical nurse specialist from Aspirus who assists periodically) who have been able to mentor, guide, and encourage interdisciplinary team members to present a challenging case. Many presenters had never presented a difficult case in this format. Presenters found the process fun and rewarding and have offered to present cases again in the future.
A second unique feature was utilizing the Wisconsin Star Method rather than focusing on a typical medical model framework for discussing a challenging case. The Star Method allows participants to increase their proficiency in providing comprehensive care while being more confident and mindful in addressing the complicated interacting physical, emotional and social issues of older adults [13].
A monthly post-call debriefing with committee members to review the strengths and weakness of the call was key to growing the program. The committee was able to critically review the process of the call, review participant surveys and discuss next steps. Adding a webinar approach, automatic email notification of calls, participant electronic survey, recording the call, and the addition of offering contact hours were some of the action items that were a result of monthly debriefing calls.
The team also found the 3-system collaboration to be beneficial. Aspirus has a large rural population, and Wheaton and Aurora have a diverse population, and each adds to the participant’s experience. Each IGDCC was rotated between the systems, which did not put the burden on any one health system. An annual call assignment listing was maintained for noting which system was responsible for the case each month and whether the geriatric expert was assigned/confirmed. Identifying the committee’s individual and collective group expertise was helpful in the overall project planning. The committee also developed a standard presenter guide and template and an expert teaching guide so the monthly IGDCC were consistent.
Challenges
The committee did not have a budget. Participation on the committee was in-kind funding from each system. Aspirus used its electronic system in place at the time to support the project. Interactive conference call education platform can be challenging with multiple participants on an open line who may not mute their phone. Often times, when a group of participants are calling in from one phone line it is difficult to know how many people are attending the IGDCC. It can be challenging at times to facilitate the call during the discussion component as participants occasionally talk over each other.
Current Status/Future Directions
The team has completed 18 consecutive monthly IGDCCs. Our participation rate has tripled. Participant satisfaction remains favorable. The team is now offering 1 contact hour to participants, and our invitations to participate have been extended to national health care groups. Challenging cases will be presented from community sources outside the hospital. Focusing attention on elevating the level of geriatric care in our region using a community educational approach will give us new opportunities for collaborating on best practice in multiple settings across the care continuum.
Acknowledgment: The planning team acknowledges Evalyn Michira, MSN, RN, PHN, AGCNS-BC, for her assistance in call presentations.
Corresponding author: Margie Hackbarth, MBA, margie.hackbarth@aspirus.org.
Financial disclosures: none.
1. Institute of Medicine. Retooling for an aging America: Building the health care workforce. Washington, DC: National Academies Press; 2008.
2. Mitchell P, Wynia M, Golden R, et al. Core principles and values of effective team-based health care. Discussion paper. Washington, DC; Institute of Medicine; 2012.
3. Nurses Improving Care for Healthsystem Elders. Accessed 1 Dec 2015 at www.nicheprogram.org/.
4. Wisconsin Department of Health Services. Southeastern region population report: 1 Jul 2013. Accessed 16 Feb 2015 at www.dhs.wisconsin.gov/sites/default/files/legacy/population/13data/southeastern.pdf.
5. From the Centers for Disease Control and Prevention. Public health and aging: trends in aging--United States and worldwide. JAMA 2003;289:1371–3.
6. Hall MJ, DeFrances CJ, Williams SN, et al. National Hospital Discharge Survey: 2007 summary. Natl Health Stat Report 2010;(29):1–20, 24.
7. Nembhard IM, Edmondson AC. Making it safe: The effects of leader inclusiveness and professional status on psychological safety and improvement efforts in health care teams. J Organiz Behav 2006; 27:941–66.
8. Institute of Medicine. The future of nursing: leading change, advancing health. National Academies Press; 2011.
9. Reeves S, Zwarenstein M, Goldman et al. Interprofessional education: effects on professional practice and health care outcomes. Cochrane Database Syst Rev 2013;3:CD002213.
10. Salas E, Diaz Granados D, Klein C, et al. Does team training improve team performance? A meta-analysis. Hum Factors 2008;50:903–33.
11. Strasser DD, Burridge AB, Falconer JA, et al. Toward spanning the quality chasm: an examination of team functioning measures. Arch Phys Med Rehabil 2014;95:2220–3.
12. Roche VM, Torregosa H, Howell T, Malone ML. Establishing a treatment plan for an elder with a complex and incomplete medical history and multiple medical providers, diagnoses, and medications. Ann Long-Term Care 2012;20(9).
13. Roche VM, Arnouville J, Danto-Nocton ES, et al. Optimal management of an older patient with multiple comorbidities and a complex psychosocial history. Ann Long-Term Care 2011;19(9).
14. Wisconsin Geriatric Psychiatry Initiative. The Wisconsin Star Method. Accessed 19 Jan 2015 at wgpi.wisc.edu/wisconsin-star-method/.
1. Institute of Medicine. Retooling for an aging America: Building the health care workforce. Washington, DC: National Academies Press; 2008.
2. Mitchell P, Wynia M, Golden R, et al. Core principles and values of effective team-based health care. Discussion paper. Washington, DC; Institute of Medicine; 2012.
3. Nurses Improving Care for Healthsystem Elders. Accessed 1 Dec 2015 at www.nicheprogram.org/.
4. Wisconsin Department of Health Services. Southeastern region population report: 1 Jul 2013. Accessed 16 Feb 2015 at www.dhs.wisconsin.gov/sites/default/files/legacy/population/13data/southeastern.pdf.
5. From the Centers for Disease Control and Prevention. Public health and aging: trends in aging--United States and worldwide. JAMA 2003;289:1371–3.
6. Hall MJ, DeFrances CJ, Williams SN, et al. National Hospital Discharge Survey: 2007 summary. Natl Health Stat Report 2010;(29):1–20, 24.
7. Nembhard IM, Edmondson AC. Making it safe: The effects of leader inclusiveness and professional status on psychological safety and improvement efforts in health care teams. J Organiz Behav 2006; 27:941–66.
8. Institute of Medicine. The future of nursing: leading change, advancing health. National Academies Press; 2011.
9. Reeves S, Zwarenstein M, Goldman et al. Interprofessional education: effects on professional practice and health care outcomes. Cochrane Database Syst Rev 2013;3:CD002213.
10. Salas E, Diaz Granados D, Klein C, et al. Does team training improve team performance? A meta-analysis. Hum Factors 2008;50:903–33.
11. Strasser DD, Burridge AB, Falconer JA, et al. Toward spanning the quality chasm: an examination of team functioning measures. Arch Phys Med Rehabil 2014;95:2220–3.
12. Roche VM, Torregosa H, Howell T, Malone ML. Establishing a treatment plan for an elder with a complex and incomplete medical history and multiple medical providers, diagnoses, and medications. Ann Long-Term Care 2012;20(9).
13. Roche VM, Arnouville J, Danto-Nocton ES, et al. Optimal management of an older patient with multiple comorbidities and a complex psychosocial history. Ann Long-Term Care 2011;19(9).
14. Wisconsin Geriatric Psychiatry Initiative. The Wisconsin Star Method. Accessed 19 Jan 2015 at wgpi.wisc.edu/wisconsin-star-method/.