User login
VTE Diagnosis and Treatment
Despite the availability of effective thromboprophylaxis, the prevalence of venous thromboembolism (VTE) is increasing in the hospital setting. In 2008, the Fifth Annual Health Grades Patient Safety in American Hospitals Study reported on key patient safety incidents among nearly 41 million hospitalizations in the Medicare population between 2004 and 2006. Although many areas showed improvementincluding reduced rates of hospital‐related infections, postoperative bleeding, transfusion reactions, and other injuriesthe number of cases of postoperative VTE increased by 11% during this period.1
Even with optimal thromboprophylaxis, VTE will develop in some at‐risk patients. Early diagnosis and treatment of VTE is critical to reduce morbidity and mortality, but no single tool can definitively confirm its presence. Consequently, the detection of deep vein thrombosis (DVT) and pulmonary embolism (PE) requires a stepwise diagnostic strategy that combines clinical, biochemical, and imaging modalities.
In addition to outlining diagnostic strategies for DVT and PE, this article summarizes VTE treatment guidelines from various organizations and societies and discusses long‐term management strategies to prevent recurrent VTE and other complications.
Diagnosis of DVT
The clinical symptoms and signs of DVT are nonspecific and include unilateral calf, leg, or thigh swelling and pain. Despite the limited sensitivity and specificity of individual signs and symptoms of DVT, the combination of these variables can be useful in assessing the probability of VTE. Patients can be risk stratified according to the likelihood of DVT, as determined by implicit clinical judgment or by a validated prediction rule.2
Assessment of Clinical Probability
The Wells prediction rule is used in assessing the probability of DVT.3 It incorporates signs, symptoms, and risk factors of DVT to calculate a clinical probability rating. Specifically, 1 point is assigned to each of the following factors, if present:3
-
Active cancer (treatment ongoing, within 6 months, or palliative)
-
Calf swelling >3 cm asymptomatic side (measured 10 cm below tibial tuberosity)
-
Collateral superficial veins (nonvaricose)
-
Entire leg swelling
-
Localized tenderness along the distribution of the deep venous system
-
Paralysis, paresis, or recent plaster immobilization of the lower extremities
-
Pitting edema confined to the symptomatic leg
-
Recently bedridden more than 3 days or major surgery within 4 weeks
In addition, 2 points are subtracted if an alternative diagnosis is as likely as or more likely than DVT. In patients with symptoms in both legs, the more symptomatic leg is used.
Patients with low (score <1), moderate (score 1‐2), and high (score 3) pretest probability of DVT have been shown to have DVT prevalence rates of 3%, 17%, and 75%, respectively.3
D‐Dimer Testing
D‐dimer testing measures the small protein fragments remaining in the blood after a cross‐linked fibrin clot is degraded by fibrinolysis. A low clinical probability assessment combined with a negative result in a highly sensitive, enzyme‐linked immunosorbent assay (ELISA)‐based D‐dimer test can safely exclude DVT, with a negative predictive value of 99.1% (95% confidence interval [CI]; 96.7‐99.9).4
Due to its poor specificity, D‐dimer testing has limited utility in unselected inpatients, especially older patients and those who have undergone prolonged hospitalization.5 However, it is reasonable to obtain a highly sensitive, ELISA‐based D‐dimer test in carefully selected inpatients with a low pretest probability of DVT.5, 6 In such patients, a negative result indicates that DVT is highly unlikely, while a positive result indicates a need for further testing. D‐dimer testing is likely not helpful in moderate‐risk or high‐risk patients.
Diagnostic Imaging
For patients with a moderate to high pretest probability of DVT, ultrasound is recommended.6 Compression ultrasonography (CUS) is currently the preferred imaging tool in patients with suspected DVT because it is noninvasive, can be repeated serially, and offers high sensitivity (+90%) and high specificity (95%) for detecting proximal vein thrombosis.7, 8 If the clinical suspicion of DVT persists after an initial negative CUS study, imaging can be repeated after 3 to 7 days to detect the propagation of any thrombosis to the proximal veins. Limitations of CUS include poor visualization of deep iliac and pelvic veins and poor sensitivity in isolated or nonocclusive calf vein thrombi.2
Contrast venography was considered the gold standard for the detection of DVT of the lower extremity, but this modality is invasive, painful, and offers poor visualization of the deep femoral vein and the internal iliac vein. In addition, contrast venography is associated with an increased risk of new thrombosis, renal failure, and hypersensitivity reaction to contrast media. Consequently, contrast venography is currently used in symptomatic patients only when noninvasive testing is inconclusive or unavailable.2 Other second‐line diagnostic tools include computed tomography venography (CTV) and magnetic resonance venography (MRV).9
Diagnostic Strategy
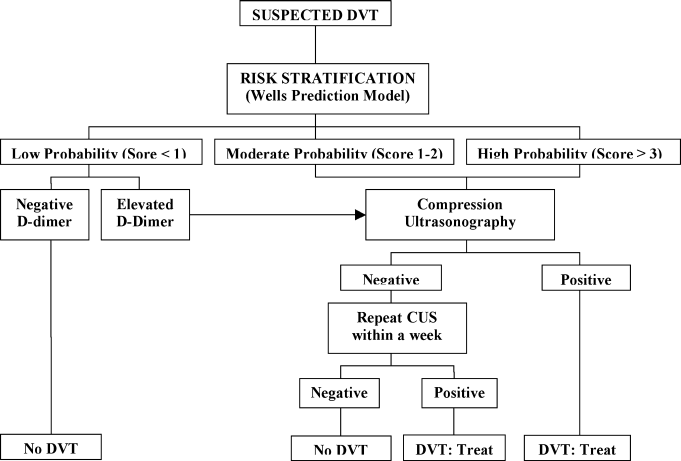
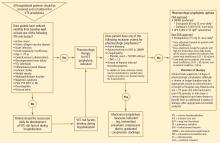
A diagnostic algorithm for DVT is presented in Figure 1. First, a validated clinical prediction scale such as the Wells prediction rule should be used to estimate the pretest probability of DVT, and the result of the clinical assessment should influence the choice and interpretation of subsequent testing.
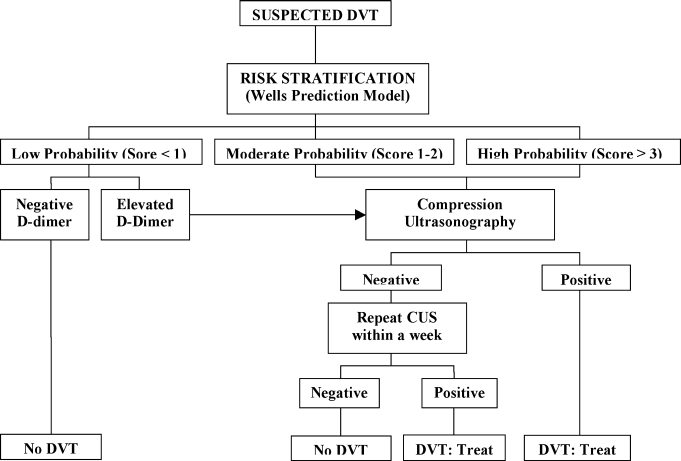
Abbreviations: CUS, compression ultrasonography; DVT, deep vein thrombosis (DVT).
Diagnosis of PE
Clinical symptoms and signs such as dyspnea, chest pain, tachycardia, tachypnea, and syncope raise the suspicion of PE. Individual signs and symptoms, however, cannot confirm or exclude acute PE, as they are neither sensitive nor specific.10 Furthermore, although the likelihood of PE increases with the number of predisposing risk factors, approximately 30% of PE cases are unprovoked or idiopathic, meaning that they occur in the absence of predisposing factors. Diagnosis, therefore, depends on an integrated strategy involving similar tools as those used in diagnosing DVT.
Assessing Clinical Probability
Wells et al.11 also developed a clinical prediction rule for the risk stratification of patients with suspected PE. In this model, 7 specified variables are assigned different scores: clinical signs and symptoms of DVT (3.0); lack of a likely alternative diagnosis (3.0); heart rate greater than 100 beats per minute (1.5); immobilization for more than 3 days or surgery in the previous 4 weeks (1.5); previous DVT/PE (1.5); hemoptysis (1.0); and malignancy (1.0). Although the Wells prediction rule initially categorized 3 levels of probability for PE (low, moderate, or high), a revised model uses a simplified, dichotomized approach to determine whether PE is likely (Wells score >4) or unlikely (4 Wells score).11 An independent, prospective observational study found that the Wells prediction model reliably risk‐stratified pretest probability in patients with suspected PE.12
For patients who are stratified into the low‐risk category, the pulmonary embolism rule‐out criteria (PERC) rule may be helpful in reducing unnecessary diagnostic testing for PE.13 The PERC rule consists of 8 variables designed to offer a pretest probability of PE of less than 1.8%, a probability at which further testing is unnecessary. If the clinical gestalt is that PE is unlikely and all of the following variables are present, further testing can be safely discontinued: (1) pulse <100; (2) age <50; (3) oxygen saturation (SaO2) >94%; (4) no unilateral leg swelling; (5) no hemoptysis; (6) no recent trauma or surgery; (7) no prior DVT or PE; and (8) no hormone use.13 In a large, multicenter study, these criteria combined with a gestalt interpretation of low risk were shown to select a subgroup of patients with a very low probability of PE (<2%).14
D‐Dimer Testing
Evidence suggests that the combination of a low clinical probability assessment and a normal result in a highly sensitive, ELISA‐based D‐dimer test can safely exclude PE in hospitalized patients.15 Due to the large number of comorbidities among hospitalized patients, however, this combination occurs in only approximately 10% of inpatients.15 D‐dimer levels may be elevated in patients with a variety of nonthrombotic conditions, and it is therefore most useful in the diagnosis of otherwise healthy patients who have symptoms of PE. D‐dimer testing is not appropriate in moderate‐risk or high‐risk patients.
Diagnostic Imaging
Computed tomography (CT) is a leading imaging modality for the exclusion or confirmation of PE, as well as for the detection of alternative diagnoses. The diagnostic algorithms endorsed by the European Society of Cardiology (ESC) rely on both single‐detector and multidetector CT. However, multidetector CT scanners are now preferred because, in contrast to single‐detector CT, they can detect pulmonary emboli in smaller pulmonary arteries.10 Because single‐detector CT has a limited sensitivity of approximately 70%, it must be used in conjunction with lower limb venous CUS.16 In contrast, multidetector CT angiography has high sensitivity (83%) and specificity (96%) for the detection of PE and does not require the additional use of lower limb venous CUS.16, 17
Diagnostic Strategy
The Christopher Study demonstrated the utility of a diagnostic algorithm that incorporates a dichotomized decision rule, D‐dimer testing, and CT. In this approach, PE is excluded in patients with an unlikely clinical probability score (Wells score 4) and a normal D‐dimer test result. In all other patients, CT is the sole imaging method used to make management decisions.18 However, in patients with massive pulmonary embolism, if CT angiography is not immediately available, selective pulmonary angiography has been performed to identify and localize the emboli before aggressive therapy is instituted (Figure 2).19 If the patient is critically ill (hypotensive, severely hypoxemic), empiric treatment is appropriate while diagnostic strategy is being formulated.
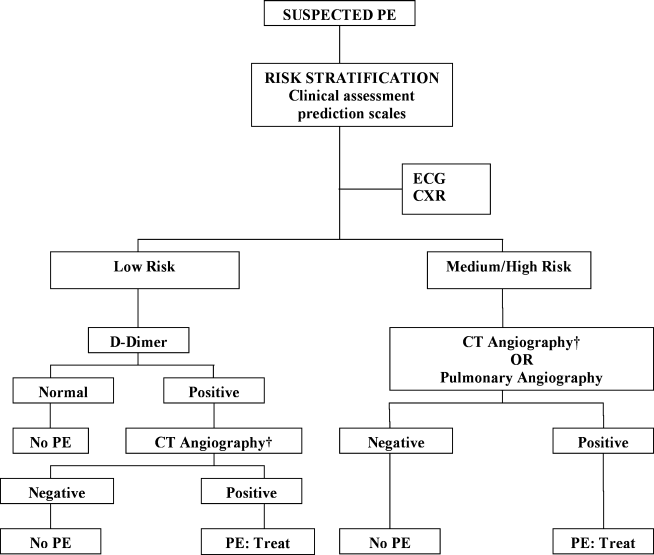
Abbreviations: CT, computed tomography; CXR, plain chest X‐ray; ECG, electrocardiogram; PE, pulmonary embolism. †CT angiography using multidetector instruments.
Treatment Options for VTE
For patients with VTE, the American College of Chest Physicians (ACCP) guidelines recommend initial treatment with low‐molecular‐weight heparin (LMWH), intravenous unfractionated heparin (UFH), or adjusted‐dose subcutaneous UFH, followed by at least 3 months of oral anticoagulation therapy.20
When VTE is diagnosed, anticoagulation should be initiated immediately unless contraindications are present. In addition, patients without contraindications to anticoagulation should receive treatment before diagnostic testing if such testing is delayed or if the clinical suspicion of VTE is high.20
Anticoagulant Treatment
For decades, parenteral administration of UFH for 5 to 7 days followed by long‐term warfarin therapy has been the conventional treatment of patients with VTE. Although UFH can be administered subcutaneously or by intravenous (IV) infusion, continuous IV infusion has been preferred because of superior dosing precision. The anticoagulation effect of intravenous UFH must be monitored to ensure a therapeutic activated partial thromboplastin time (aPTT). Consequently, the use of intravenous UFH requires frequent aPTT assessment and dose adjustment.20
Given their ease of use and improved pharmacokinetic and pharmacodynamic profiles, LMWHs have replaced UFH for the treatment of VTE in many institutions. Fondaparinux is also a safe and effective alternative to both intravenous UFH and LMWH in the treatment of VTE.20 It has a longer half‐life (15‐20 hours) than LMWH, permitting a once‐daily administration, and in patients with submassive PE, its efficacy and safety are comparable to UFH.21 Platelet count monitoring is not necessary with fondaparinux because it is given at weight‐adjusted doses, and only 1 case of heparin‐induced thrombocytopenia (HIT) has been reported.22 It is, however, contraindicated in renal failure with a creatinine clearance of <30 mL/minute.10
Warfarin is very effective in the long‐term management of VTE and should be started concurrently with rapid‐acting injectable anticoagulation therapy. Warfarin requires overlap with injectable anticoagulants for a minimum of 5 days until a therapeutic international normalized ratio (INR) has been achieved.20
Other Treatments
Most patients with VTE can be treated effectively with only anticoagulation therapy. However, in cases of massive PE (with or without systemic arterial hypotension and usually with significant hypoxemia not generally responsive to supplemental oxygen), removal of the occluding thrombus by thrombolytic agents, special clot‐removing catheters, or surgical procedures may be necessary to prevent or ameliorate shock and subsequent death.23 In other cases, such as when anticoagulants are ineffective or contraindicated, an inferior vena cava (IVC) filter may be an appropriate option for VTE treatment. Importantly, guidelines do not recommend filters in patients who can tolerate anticoagulation.
Permanent and retrievable IVC filters are effective at preventing PE and are generally associated with a low complication rate.24 However, nonfatal complications are relatively common with permanent IVC filters. One early complication is insertion‐site thrombosis, which occurs in about 10% of patients. Subsequent complications are more frequent and include recurrent DVT and post‐thrombotic syndrome (PTS), which occur in approximately 20% and 40% of patients, respectively. At 5 and 9 years, about 22% and 33% of the filters are occluded, regardless of the use and duration of anticoagulation.2527 To minimize these complications, retrievable filters have been increasingly used, but most filters are not retrieved and are subject to the same complications as permanent IVC filters.28
Catheter‐directed thrombolysis, with or without IVC filter placement, is safe and effective in treating acute DVT.29 Additional measures, such as the use of graduated compression stockings, can reduce the risk of developing PTS.20
Guideline Recommendations
Guidelines from the ACCP, the American College of Physicians (ACP), and the American Academy of Family Physicians (AAFP) address the treatment of VTE in a broad spectrum of patients. Additional guidelines provide recommendations for specific presentations or patient groups. For example, the ESC guidelines address the treatment of acute PE, and several groupsthe American Society of Clinical Oncology (ASCO), the National Comprehensive Cancer Network (NCCN), and the French Working Group (FWG)have published guidelines for the treatment of VTE in patients with cancer. The following sections summarize the most important recommendations from several of these organizations and societies.
ACCP Guidelines
The ACCP guideline recommendations are assigned grades of 1 or 2, denoting a stronger or weaker recommendation, as well as a grade of A, B, or C, indicating high‐quality evidence, moderate‐quality evidence, and low‐quality evidence, respectively. Physicians must supplement the guideline recommendations with informed clinical judgment to ensure proper use of treatment in at‐risk hospitalized patients.20
The 2008 ACCP guidelines suggest several options for the initial treatment of VTE, which are listed, along with acceptable dosing regimens, in Table 1.20, 30, 31 Fixed‐dose, unmonitored, subcutaneous UFH and fondaparinux are new Grade 1A additions to the 2008 update. In general, LMWH is preferred over intravenous UFH, except in patients with severe renal failure.20
| Initial Anticoagulation Therapy | Grade | Acceptable Treatment Regimen* |
|---|---|---|
| ||
| SC LMWH | 1A | Enoxaparin: 1 mg/kg every 12 hours or 1.5 mg/kg once daily; Dalteparin: 200 IU/kg once daily (can be administered out of hospital) |
| Intravenous UFH | 1A | Get baseline aPTT, PT, and platelet count; if no abnormalities, proceed with a weight‐based heparin infusion protocol such as: |
| Bolus of 80 U/kg, followed by an infusion of 18 U/kg per hour (treatment duration 72 days); check aPTT every 4‐6 hours and adjust according to the normogram; monitor platelet count every 3‐4 days for HIT | ||
| Monitored SC UFH; fixed‐dose, unmonitored, SC UFH | 1A; 1A | Initial dose of 333 U/kg, followed by a fixed dose of 250 U/kg every 12 hours (can be administered out of hospital) |
| SC fondaparinux | 1A | 5 mg (body weight <50 kg), 7.5 mg (body weight 50‐100 kg), or 10 mg (body weight >100 kg) once daily (treatment duration 72 days) |
| IVC filter if anticoagulation contraindicated | 1C | |
Warfarin should also be initiated on the same day as UFH or LMWH and adjusted to a target INR of 2.5 (range, 2.0‐3.0). Treatment with UFH or LMWH should be continued concomitantly for a minimum of 5 days and should not be discontinued until the INR has been over 2.0 for 24 hours. The ACCP guidelines also recommend systematic follow‐up of oral anticoagulation therapy.
ACP/AAFP Guidelines
In 2007, the ACP and the AAFP collaborated to develop joint guidelines for the management of VTE.32 Several of their key recommendations are the following:32
-
LMWH, rather than UFH, should be used whenever possible for the initial inpatient treatment of DVT
-
Either UFH or LMWH is appropriate for the initial treatment of PE
-
Anticoagulation should be continued for 3 to 6 months for VTE secondary to transient risk factors, and for more than 12 months for recurrent VTE
-
LMWH is safe and effective for the long‐term treatment of VTE in selected patients (and may be preferable for patients with cancer)
ESC Guidelines for the Treatment of PE
According to the 2008 ESC guidelines, anticoagulation with UFH, LMWH, or fondaparinux should be initiated immediately in patients with confirmed PE, as well as in those with a high or intermediate clinical probability of PE while the diagnostic workup is ongoing. Subcutaneous LMWH or fondaparinux is preferable to intravenous UFH for initial treatment in most patients. UFH, however, should be used in patients with a high risk of bleeding due to its capacity for reversal and short half‐life, as well as in those with severe renal dysfunction.10
According to the ESC, patients with high‐risk PE (presenting with cardiogenic shock or persistent arterial hypotension) should receive thrombolytic therapy as first‐line therapy. Hemodynamic and respiratory support is also necessary for these patients.10 Routine thrombolysis is not recommended in patients with non‐high‐risk PE, but it may be considered in select patients with intermediate‐risk PE (characterized by severe right ventricular dysfunction on echocardiography and/or myocardial injury), depending on the patient's risk of bleeding. Thrombolytic therapy should not be used in patients with low‐risk PE (presenting without shock, hypotension, right ventricular dysfunction, or myocardial injury).10
Like the ESC guidelines, the ACCP guidelines recommend against the use of thrombolytic therapy for the majority of patients with PE (Grade 1B), but they do recommend its use in patients with evidence of hemodynamic compromise and no major contraindications owing to bleeding risk (Grade 1B) and in certain other high‐risk patients (Grade 2B).20
The ESC states that pulmonary embolectomy has recently become a reasonable option for patients with massive, high‐risk PE and an absolute contraindication to thrombolysis, or in whom thrombolysis has failed, when appropriate expertise is available. In the past, it was performed as a last resort in patients with massive PE who were in shock and conferred a high risk of mortality (+50%). Recently, however, the procedure has been revived and performed immediately in patients with confirmed massive PE (with severe right ventricular dysfunction but before shock), with mortality rates of less than 10%.19 It should be noted, however, that the ACCP guidelines consider embolectomy a Grade 2C recommendation.20 Alternatively, catheter embolectomy or fragmentation of proximal pulmonary arterial clots may be considered as an alternative to surgical treatment in these patients.10
NCCN Guidelines: Oncology Patients
The NCCN has provided treatment algorithms for the management of DVT and PE in patients with cancer, which are available online at
FWG Guidelines: Oncology Patients
At the 2008 ASH annual meeting, the FWG presented updated guidelines for the treatment of VTE in cancer patients.34 The FWG guidelines contain the following key recommendations:
-
The treatment of VTE should be based on LMWH at curative doses for at least 3 months
-
During the initial treatment (up to 10 days), any approved drug (including LMWH, UFH, and fondaparinux) may be used
-
Beyond the first 10 days, VTE treatment should be based on LMWH at curative doses for at least 3 months and optimally 6 months, as validated with the following drugs and dosage regimens:
-
Dalteparin 200 IU/kg once daily for 1 month, then 150 IU/kg once daily
-
Enoxaparin 150 IU/kg (1.5 mg/kg) once daily
-
Tinzaparin 175 IU/kg once daily
-
Special treatment considerations include the following:
-
In severe renal impairment, UFH should be used and rapidly followed by a vitamin K agonist (VKA) for at least 3 months
-
In severe PE (representing hemodynamic failure), the indications and recommended uses of thrombolytic drugs in noncancer patients apply
-
In patients with an absolute contraindication to anticoagulation or VTE recurrence despite optimal anticoagulation, vena cava filters should be considered
-
In patients with intracranial malignancies, VTE treatment is the same as in cancer patients with nonintracranial tumors
The treatment of central venous catheter thrombosis requires the long‐term use of LMWH according to the FWG guidelines. In patients with severe renal failure, UFH with early VKA must be used as an alternative treatment. Regardless of the therapy used, treatment should be continued as long as the catheter is maintained.34
Long‐Term Management of VTE
The high rate of recurrent VTE after a first episode of DVT or PEapproximately 8% within 90 daysunderscores the importance of maintaining effective prophylaxis postdischarge.35 Inadequate prophylaxis following discharge from the hospital can have severe consequences. In a recent study of 10,744 patients who were discharged from the hospital following hip or knee replacement surgery, fewer than 1 in 5 received postdischarge thromboprophylaxis. The 3‐month risk of mortality was significantly lower among those who received thromboprophylaxis at discharge (adjusted hazard ratio, 0.34; 95% CI, 0.20‐0.57).36
Detailed patient education at the time of discharge may be one of the most effective ways to prevent or minimize the burden of long‐term complications such as PTS or recurrent VTE. Accordingly, proper discharge planning and postdischarge support, including an appropriate anticoagulant, are critical steps toward reducing mortality, morbidity, and healthcare costs.
PTS
As many as 50% of patients with VTE will develop PTS, a serious but preventable complication that leads to pain, swelling, and skin changes in the affected limb. Female gender, older age, higher body mass index (BMI), and DVT of the common femoral or iliac vein (vs. distal DVT) are associated with an increased risk of PTS.37 To prevent PTS in a patient who has had a symptomatic proximal DVT, current guidelines recommend the use of graduated elastic compression stockings with an ankle pressure of 30 to 40 mm Hg, if feasible. Compression therapy should start as soon as possible after the initiation of anticoagulation therapy and be encouraged for a minimum of 2 years.20
Recurrent VTE
After discontinuing anticoagulation, the risk of recurrent VTE increases steadily over time. In a recent long‐term study of patients with acute proximal DVT or PE, the risk of recurrent VTE was 11% after 1 year, 20% after 3 years, 30% after 5 years, and 40% after 10 years. In this study, risk factors for recurrent VTE included unprovoked initial VTE, thrombophilia, increasing age, and a shorter duration of anticoagulation (6 months or less).38 Another study identified residual venous thrombosis as an important risk factor for recurrent VTE.39 In addition, 1 meta‐analysis found that men had a 50% higher risk of recurrent VTE than women.40 Recurrent DVT events are associated with a 21% greater cost than the initial event, suggesting that recurrent VTE is a preventable drain on healthcare resources.41
Secondary Prevention
The risk of recurrent VTE is determined by the effectiveness of treatment for the acute episode of VTE and by the patient's intrinsic risk of thromboembolism. The ACCP recommends different durations of warfarin or LMWH anticoagulant therapy according to these features (Table 2).20 Attaching a high value to prevention of recurrent VTE and a lower value to the burden of long‐term anticoagulant treatment, the ACCP recommends long‐term treatment for patients with a first unprovoked proximal DVT, no risk factors for bleeding, and the ability to monitor the anticoagulant effectively (Grade 1A).20
| Clinical Features | Duration | Grade |
|---|---|---|
| ||
| First episode and transient risk factors | 3 months | 1A |
| Unprovoked episode | 3 months | 1A |
| Unprovoked proximal DVT with low bleed risk | Long‐term | 1A |
| Cancer | 3 to 6 months with LMWH; then with a VKA or a LMWH indefinitely or until cancer is resolved | 1A; 1C |
| Second unprovoked episode | Indefinite | 2A |
Transition to Outpatient Therapy
The use of outpatient LMWH has changed the course of long‐term anticoagulation therapy and is listed as the preferred option for anticoagulation in the ACCP guidelines.20 With the availability of subcutaneous LMWHs, patients with acute VTE no longer have to be hospitalized for the initiation of oral therapy. In addition, patients undergoing invasive procedures that require temporary discontinuation of warfarin can opt for bridge therapy with LMWH.42
Conclusions
The diagnosis of VTE is challenging and depends on the integration of clinical, biochemical, and imaging modalities. In the absence of contraindications, treatment should be initiated immediately after a diagnosis of VTE is confirmed. Anticoagulant therapy alone is sufficient for most patients, but some patients may require thrombolytics or other strategies. Various societies and organizations have issued recommendations regarding the optimal use of these therapies in specific patient populations. Following these recommendations carefully may reduce the risk of complications in patients with VTE.
- Health Grades, Inc. The Fifth Annual Health Grades Patient Safety in American Hospitals Study. Available at: http://www.healthgrades.com/media/dms/pdf/PatientSafetyInAmericanHospitalsStudy2008.pdf. Accessed August2009.
- , , , et al.Prevention of venous thromboembolism: American College of Chest Physicians evidence‐based clinical practice guidelines. 8th Edition.Chest2008;133(6 suppl):381S–453S.
- , , , et al.Value of assessment of pretest probability of deep‐vein thrombosis in clinical management.Lancet.1997;350(9094):1795–1798.
- , , , et al.Evaluation of D‐dimer in the diagnosis of suspected deep‐vein thrombosis.N Engl J Med.2003;349:1227–1235.
- , , , et al.Limitations of D‐dimer testing in unselected inpatients with suspected venous thromboembolism.Am J Med.2003;114(4):276–282.
- , , , et al.Current diagnosis of venous thromboembolism in primary care: a clinical practice guideline from the American Academy of Family Physicians and the American College of Physicians.Ann Fam Med.2007;5(1):57–62.
- , , .The role of venous ultrasonography in the diagnosis of suspected deep venous thrombosis and pulmonary embolism.Ann Intern Med.1998;129:1044–1049.
- , .Ultrasonography of leg veins in patients suspected of having pulmonary embolism.Ann Intern Med.1998;128:243–245.
- , .Role of computed tomography and magnetic resonance imaging for deep venous thrombosis and pulmonary embolism.Circulation.2004;109(12 suppl 1):I15–I21.
- , , , et al.Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology.Guidelines on the diagnosis and management of acute pulmonary embolism: the Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology (ESC).Eur Heart J.2008;29(18):2276–2315.
- , , , et al.Derivation of a simple clinical model to categorize patients probability of pulmonary embolism: increasing the models utility with the SimpliRED D‐dimer.Thromb Haemost.2000;83(3):416–420.
- , , , et al.Prospective validation of Wells criteria in the evaluation of patients with suspected pulmonary embolism.Ann Emerg Med.2004;44(5):503–510.
- , , , et al.Clinical criteria to prevent unnecessary diagnostic testing in emergency department patients with suspected pulmonary embolism.J Thromb Haemost.2004;2(8):1247–1255.
- , , , et al.Prospective multicenter evaluation of the pulmonary embolism rule‐out criteria.J Thromb Haemost.2008;6(5):772–780.
- , , , et al.A simple diagnostic strategy in hospitalized patients with clinically suspected pulmonary embolism.J Intern Med.2006;260(5):459–466.
- , .Is computed tomographic venography of lower limbs useful in suspected pulmonary embolism?Rev Med Suisse.2008;4(143):354,356–359.
- , , , et al.Multidetector computed tomography for acute pulmonary embolism.N Engl J Med.2006;354(22):2317–2327.
- , , , et al.Christopher Study Investigators.Effectiveness of managing suspected pulmonary embolism using an algorithm combining clinical probability, D‐dimer testing, and computed tomography.JAMA.2006;295(2):172–179.
- , , , .Acute pulmonary embolectomy: a contemporary approach.Circulation.2002;105:1416–1419.
- , , , et al.Antithrombotic therapy for venous thromboembolic disease: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines (8th Edition).Chest.2008;133(6 suppl):454S–545S.
- , , , et al.Subcutaneous fondaparinux versus unfractionated heparin in the initial treatment of pulmonary embolism.N Engl J Med.2003;349:1695–1702.
- .Heparin‐induced thrombocytopenia associated with fondaparinux.N Engl J Med.2007;356:2653–2655.
- , , , .Thrombolysis compared with heparin for the initial treatment of pulmonary embolism: a meta‐analysis of the randomized controlled trials.Circulation.2004;110:744–749.
- , , , et al.Ins and outs of inferior vena cava filters in patients with venous thromboembolism: the experience at Monash Medical Centre and review of the published reports.Intern Med J.2008;38(1):38–43.
- PREPIC Study Group.Eight‐year follow‐up of patients with permanent vena cava filters in the prevention of pulmonary embolism: the PREPIC (Prevention du Risque d'Embolie Pulmonaire par Interruption Cave) randomized study.Circulation.2005;112:416–422.
- , , , .Inferior vena cava filters: key considerations.Am J Med Sci.2005;330:82–87.
- , , , .Percutaneous inferior vena cava filters: follow‐up of 7 designs in 320 patients.Radiology.1993;188:851–856.
- , , , et al.Practice patterns and outcomes of retrievable vena cava filters in trauma patients: an AAST multicenter study.J Trauma.2007;62:17–24.
- , , , , , .Long‐term outcomes of catheter directed thrombolysis for lower extremity deep venous thrombosis without prophylactic inferior vena cava filter placement.J Vasc Surg.2007;45(5):992–997.
- , , , et al.Comparison of fixed‐dose weight‐adjusted unfractionated heparin and low‐molecular‐weight heparin for acute treatment of venous thromboembolism.JAMA.2006;296(8):935–942.
- Arixtra prescribing information. Last updated October 2008. Research Triangle Park, NC: GlaxoSmithKline. Available at: http://us.gsk.com/products/assets/us_arixtra.pdf. Accessed August2009.
- , , , et al.American College of Physicians;American Academy of Family Physicians Panel on Deep Venous Thrombosis/Pulmonary Embolism.Management of venous thromboembolism: a clinical practice guideline from the American College of Physicians and the American Academy of Family Physicians.Ann Intern Med.2007;146(3):204–210.
- National Comprehensive Cancer Network (NCCN). Venous thromboembolic disease. Practice Guidelines in Oncology. V.1.2009. Available at: http://www.nccn.org/professionals/physician_gls/PDF/vte.pdf. Accessed August2009.
- , , , et al. Guidelines for the treatment of venous thromboembolism in cancer patients: report from the French Working Group. Presented at the 50th Annual Meeting of the American College of Hematology; San Francisco, CA; December 6‐9, 2008. Abstract 1284.
- , , , et al.Predictors of recurrence after deep vein thrombosis and pulmonary embolism: a population‐based cohort study.Arch Intern Med.2000;160(6):761–768.
- , , , et al.Postdischarge thromboprophylaxis and mortality risk after hip‐or knee‐replacement surgery.CMAJ.2008;178(12):1545–1554.
- , , , et al.Determinants and time course of the postthrombotic syndrome after acute deep venous thrombosis.Ann Intern Med.2008;149(10):698–707.
- , , , et al.The risk of recurrent venous thromboembolism after discontinuing anticoagulation in patients with acute proximal deep vein thrombosis or pulmonary embolism. A prospective cohort study in 1,626 patients.Haematologica.2007;92(2):199–205.
- .Risk factors of recurrent venous thromboembolism: the role of residual vein thrombosis.Pathophysiol Haemost Thromb.2003/2004;33(5‐6):351–353.
- , , , et al.Effect of patient's sex on risk of recurrent venous thromboembolism: a meta‐analysis.Lancet.2006;368:371–378.
- , .Direct medical costs of venous thromboembolism and subsequent hospital readmission rates: an administrative claims analysis from 30 managed care organizations.J Manag Care Pharm.2007;13(6):475–486.
- , .Outpatient management of anticoagulation therapy.Am Fam Physician.2007;75:1031–1042.
Despite the availability of effective thromboprophylaxis, the prevalence of venous thromboembolism (VTE) is increasing in the hospital setting. In 2008, the Fifth Annual Health Grades Patient Safety in American Hospitals Study reported on key patient safety incidents among nearly 41 million hospitalizations in the Medicare population between 2004 and 2006. Although many areas showed improvementincluding reduced rates of hospital‐related infections, postoperative bleeding, transfusion reactions, and other injuriesthe number of cases of postoperative VTE increased by 11% during this period.1
Even with optimal thromboprophylaxis, VTE will develop in some at‐risk patients. Early diagnosis and treatment of VTE is critical to reduce morbidity and mortality, but no single tool can definitively confirm its presence. Consequently, the detection of deep vein thrombosis (DVT) and pulmonary embolism (PE) requires a stepwise diagnostic strategy that combines clinical, biochemical, and imaging modalities.
In addition to outlining diagnostic strategies for DVT and PE, this article summarizes VTE treatment guidelines from various organizations and societies and discusses long‐term management strategies to prevent recurrent VTE and other complications.
Diagnosis of DVT
The clinical symptoms and signs of DVT are nonspecific and include unilateral calf, leg, or thigh swelling and pain. Despite the limited sensitivity and specificity of individual signs and symptoms of DVT, the combination of these variables can be useful in assessing the probability of VTE. Patients can be risk stratified according to the likelihood of DVT, as determined by implicit clinical judgment or by a validated prediction rule.2
Assessment of Clinical Probability
The Wells prediction rule is used in assessing the probability of DVT.3 It incorporates signs, symptoms, and risk factors of DVT to calculate a clinical probability rating. Specifically, 1 point is assigned to each of the following factors, if present:3
-
Active cancer (treatment ongoing, within 6 months, or palliative)
-
Calf swelling >3 cm asymptomatic side (measured 10 cm below tibial tuberosity)
-
Collateral superficial veins (nonvaricose)
-
Entire leg swelling
-
Localized tenderness along the distribution of the deep venous system
-
Paralysis, paresis, or recent plaster immobilization of the lower extremities
-
Pitting edema confined to the symptomatic leg
-
Recently bedridden more than 3 days or major surgery within 4 weeks
In addition, 2 points are subtracted if an alternative diagnosis is as likely as or more likely than DVT. In patients with symptoms in both legs, the more symptomatic leg is used.
Patients with low (score <1), moderate (score 1‐2), and high (score 3) pretest probability of DVT have been shown to have DVT prevalence rates of 3%, 17%, and 75%, respectively.3
D‐Dimer Testing
D‐dimer testing measures the small protein fragments remaining in the blood after a cross‐linked fibrin clot is degraded by fibrinolysis. A low clinical probability assessment combined with a negative result in a highly sensitive, enzyme‐linked immunosorbent assay (ELISA)‐based D‐dimer test can safely exclude DVT, with a negative predictive value of 99.1% (95% confidence interval [CI]; 96.7‐99.9).4
Due to its poor specificity, D‐dimer testing has limited utility in unselected inpatients, especially older patients and those who have undergone prolonged hospitalization.5 However, it is reasonable to obtain a highly sensitive, ELISA‐based D‐dimer test in carefully selected inpatients with a low pretest probability of DVT.5, 6 In such patients, a negative result indicates that DVT is highly unlikely, while a positive result indicates a need for further testing. D‐dimer testing is likely not helpful in moderate‐risk or high‐risk patients.
Diagnostic Imaging
For patients with a moderate to high pretest probability of DVT, ultrasound is recommended.6 Compression ultrasonography (CUS) is currently the preferred imaging tool in patients with suspected DVT because it is noninvasive, can be repeated serially, and offers high sensitivity (+90%) and high specificity (95%) for detecting proximal vein thrombosis.7, 8 If the clinical suspicion of DVT persists after an initial negative CUS study, imaging can be repeated after 3 to 7 days to detect the propagation of any thrombosis to the proximal veins. Limitations of CUS include poor visualization of deep iliac and pelvic veins and poor sensitivity in isolated or nonocclusive calf vein thrombi.2
Contrast venography was considered the gold standard for the detection of DVT of the lower extremity, but this modality is invasive, painful, and offers poor visualization of the deep femoral vein and the internal iliac vein. In addition, contrast venography is associated with an increased risk of new thrombosis, renal failure, and hypersensitivity reaction to contrast media. Consequently, contrast venography is currently used in symptomatic patients only when noninvasive testing is inconclusive or unavailable.2 Other second‐line diagnostic tools include computed tomography venography (CTV) and magnetic resonance venography (MRV).9
Diagnostic Strategy
A diagnostic algorithm for DVT is presented in Figure 1. First, a validated clinical prediction scale such as the Wells prediction rule should be used to estimate the pretest probability of DVT, and the result of the clinical assessment should influence the choice and interpretation of subsequent testing.

Abbreviations: CUS, compression ultrasonography; DVT, deep vein thrombosis (DVT).
Diagnosis of PE
Clinical symptoms and signs such as dyspnea, chest pain, tachycardia, tachypnea, and syncope raise the suspicion of PE. Individual signs and symptoms, however, cannot confirm or exclude acute PE, as they are neither sensitive nor specific.10 Furthermore, although the likelihood of PE increases with the number of predisposing risk factors, approximately 30% of PE cases are unprovoked or idiopathic, meaning that they occur in the absence of predisposing factors. Diagnosis, therefore, depends on an integrated strategy involving similar tools as those used in diagnosing DVT.
Assessing Clinical Probability
Wells et al.11 also developed a clinical prediction rule for the risk stratification of patients with suspected PE. In this model, 7 specified variables are assigned different scores: clinical signs and symptoms of DVT (3.0); lack of a likely alternative diagnosis (3.0); heart rate greater than 100 beats per minute (1.5); immobilization for more than 3 days or surgery in the previous 4 weeks (1.5); previous DVT/PE (1.5); hemoptysis (1.0); and malignancy (1.0). Although the Wells prediction rule initially categorized 3 levels of probability for PE (low, moderate, or high), a revised model uses a simplified, dichotomized approach to determine whether PE is likely (Wells score >4) or unlikely (4 Wells score).11 An independent, prospective observational study found that the Wells prediction model reliably risk‐stratified pretest probability in patients with suspected PE.12
For patients who are stratified into the low‐risk category, the pulmonary embolism rule‐out criteria (PERC) rule may be helpful in reducing unnecessary diagnostic testing for PE.13 The PERC rule consists of 8 variables designed to offer a pretest probability of PE of less than 1.8%, a probability at which further testing is unnecessary. If the clinical gestalt is that PE is unlikely and all of the following variables are present, further testing can be safely discontinued: (1) pulse <100; (2) age <50; (3) oxygen saturation (SaO2) >94%; (4) no unilateral leg swelling; (5) no hemoptysis; (6) no recent trauma or surgery; (7) no prior DVT or PE; and (8) no hormone use.13 In a large, multicenter study, these criteria combined with a gestalt interpretation of low risk were shown to select a subgroup of patients with a very low probability of PE (<2%).14
D‐Dimer Testing
Evidence suggests that the combination of a low clinical probability assessment and a normal result in a highly sensitive, ELISA‐based D‐dimer test can safely exclude PE in hospitalized patients.15 Due to the large number of comorbidities among hospitalized patients, however, this combination occurs in only approximately 10% of inpatients.15 D‐dimer levels may be elevated in patients with a variety of nonthrombotic conditions, and it is therefore most useful in the diagnosis of otherwise healthy patients who have symptoms of PE. D‐dimer testing is not appropriate in moderate‐risk or high‐risk patients.
Diagnostic Imaging
Computed tomography (CT) is a leading imaging modality for the exclusion or confirmation of PE, as well as for the detection of alternative diagnoses. The diagnostic algorithms endorsed by the European Society of Cardiology (ESC) rely on both single‐detector and multidetector CT. However, multidetector CT scanners are now preferred because, in contrast to single‐detector CT, they can detect pulmonary emboli in smaller pulmonary arteries.10 Because single‐detector CT has a limited sensitivity of approximately 70%, it must be used in conjunction with lower limb venous CUS.16 In contrast, multidetector CT angiography has high sensitivity (83%) and specificity (96%) for the detection of PE and does not require the additional use of lower limb venous CUS.16, 17
Diagnostic Strategy
The Christopher Study demonstrated the utility of a diagnostic algorithm that incorporates a dichotomized decision rule, D‐dimer testing, and CT. In this approach, PE is excluded in patients with an unlikely clinical probability score (Wells score 4) and a normal D‐dimer test result. In all other patients, CT is the sole imaging method used to make management decisions.18 However, in patients with massive pulmonary embolism, if CT angiography is not immediately available, selective pulmonary angiography has been performed to identify and localize the emboli before aggressive therapy is instituted (Figure 2).19 If the patient is critically ill (hypotensive, severely hypoxemic), empiric treatment is appropriate while diagnostic strategy is being formulated.

Abbreviations: CT, computed tomography; CXR, plain chest X‐ray; ECG, electrocardiogram; PE, pulmonary embolism. †CT angiography using multidetector instruments.
Treatment Options for VTE
For patients with VTE, the American College of Chest Physicians (ACCP) guidelines recommend initial treatment with low‐molecular‐weight heparin (LMWH), intravenous unfractionated heparin (UFH), or adjusted‐dose subcutaneous UFH, followed by at least 3 months of oral anticoagulation therapy.20
When VTE is diagnosed, anticoagulation should be initiated immediately unless contraindications are present. In addition, patients without contraindications to anticoagulation should receive treatment before diagnostic testing if such testing is delayed or if the clinical suspicion of VTE is high.20
Anticoagulant Treatment
For decades, parenteral administration of UFH for 5 to 7 days followed by long‐term warfarin therapy has been the conventional treatment of patients with VTE. Although UFH can be administered subcutaneously or by intravenous (IV) infusion, continuous IV infusion has been preferred because of superior dosing precision. The anticoagulation effect of intravenous UFH must be monitored to ensure a therapeutic activated partial thromboplastin time (aPTT). Consequently, the use of intravenous UFH requires frequent aPTT assessment and dose adjustment.20
Given their ease of use and improved pharmacokinetic and pharmacodynamic profiles, LMWHs have replaced UFH for the treatment of VTE in many institutions. Fondaparinux is also a safe and effective alternative to both intravenous UFH and LMWH in the treatment of VTE.20 It has a longer half‐life (15‐20 hours) than LMWH, permitting a once‐daily administration, and in patients with submassive PE, its efficacy and safety are comparable to UFH.21 Platelet count monitoring is not necessary with fondaparinux because it is given at weight‐adjusted doses, and only 1 case of heparin‐induced thrombocytopenia (HIT) has been reported.22 It is, however, contraindicated in renal failure with a creatinine clearance of <30 mL/minute.10
Warfarin is very effective in the long‐term management of VTE and should be started concurrently with rapid‐acting injectable anticoagulation therapy. Warfarin requires overlap with injectable anticoagulants for a minimum of 5 days until a therapeutic international normalized ratio (INR) has been achieved.20
Other Treatments
Most patients with VTE can be treated effectively with only anticoagulation therapy. However, in cases of massive PE (with or without systemic arterial hypotension and usually with significant hypoxemia not generally responsive to supplemental oxygen), removal of the occluding thrombus by thrombolytic agents, special clot‐removing catheters, or surgical procedures may be necessary to prevent or ameliorate shock and subsequent death.23 In other cases, such as when anticoagulants are ineffective or contraindicated, an inferior vena cava (IVC) filter may be an appropriate option for VTE treatment. Importantly, guidelines do not recommend filters in patients who can tolerate anticoagulation.
Permanent and retrievable IVC filters are effective at preventing PE and are generally associated with a low complication rate.24 However, nonfatal complications are relatively common with permanent IVC filters. One early complication is insertion‐site thrombosis, which occurs in about 10% of patients. Subsequent complications are more frequent and include recurrent DVT and post‐thrombotic syndrome (PTS), which occur in approximately 20% and 40% of patients, respectively. At 5 and 9 years, about 22% and 33% of the filters are occluded, regardless of the use and duration of anticoagulation.2527 To minimize these complications, retrievable filters have been increasingly used, but most filters are not retrieved and are subject to the same complications as permanent IVC filters.28
Catheter‐directed thrombolysis, with or without IVC filter placement, is safe and effective in treating acute DVT.29 Additional measures, such as the use of graduated compression stockings, can reduce the risk of developing PTS.20
Guideline Recommendations
Guidelines from the ACCP, the American College of Physicians (ACP), and the American Academy of Family Physicians (AAFP) address the treatment of VTE in a broad spectrum of patients. Additional guidelines provide recommendations for specific presentations or patient groups. For example, the ESC guidelines address the treatment of acute PE, and several groupsthe American Society of Clinical Oncology (ASCO), the National Comprehensive Cancer Network (NCCN), and the French Working Group (FWG)have published guidelines for the treatment of VTE in patients with cancer. The following sections summarize the most important recommendations from several of these organizations and societies.
ACCP Guidelines
The ACCP guideline recommendations are assigned grades of 1 or 2, denoting a stronger or weaker recommendation, as well as a grade of A, B, or C, indicating high‐quality evidence, moderate‐quality evidence, and low‐quality evidence, respectively. Physicians must supplement the guideline recommendations with informed clinical judgment to ensure proper use of treatment in at‐risk hospitalized patients.20
The 2008 ACCP guidelines suggest several options for the initial treatment of VTE, which are listed, along with acceptable dosing regimens, in Table 1.20, 30, 31 Fixed‐dose, unmonitored, subcutaneous UFH and fondaparinux are new Grade 1A additions to the 2008 update. In general, LMWH is preferred over intravenous UFH, except in patients with severe renal failure.20
| Initial Anticoagulation Therapy | Grade | Acceptable Treatment Regimen* |
|---|---|---|
| ||
| SC LMWH | 1A | Enoxaparin: 1 mg/kg every 12 hours or 1.5 mg/kg once daily; Dalteparin: 200 IU/kg once daily (can be administered out of hospital) |
| Intravenous UFH | 1A | Get baseline aPTT, PT, and platelet count; if no abnormalities, proceed with a weight‐based heparin infusion protocol such as: |
| Bolus of 80 U/kg, followed by an infusion of 18 U/kg per hour (treatment duration 72 days); check aPTT every 4‐6 hours and adjust according to the normogram; monitor platelet count every 3‐4 days for HIT | ||
| Monitored SC UFH; fixed‐dose, unmonitored, SC UFH | 1A; 1A | Initial dose of 333 U/kg, followed by a fixed dose of 250 U/kg every 12 hours (can be administered out of hospital) |
| SC fondaparinux | 1A | 5 mg (body weight <50 kg), 7.5 mg (body weight 50‐100 kg), or 10 mg (body weight >100 kg) once daily (treatment duration 72 days) |
| IVC filter if anticoagulation contraindicated | 1C | |
Warfarin should also be initiated on the same day as UFH or LMWH and adjusted to a target INR of 2.5 (range, 2.0‐3.0). Treatment with UFH or LMWH should be continued concomitantly for a minimum of 5 days and should not be discontinued until the INR has been over 2.0 for 24 hours. The ACCP guidelines also recommend systematic follow‐up of oral anticoagulation therapy.
ACP/AAFP Guidelines
In 2007, the ACP and the AAFP collaborated to develop joint guidelines for the management of VTE.32 Several of their key recommendations are the following:32
-
LMWH, rather than UFH, should be used whenever possible for the initial inpatient treatment of DVT
-
Either UFH or LMWH is appropriate for the initial treatment of PE
-
Anticoagulation should be continued for 3 to 6 months for VTE secondary to transient risk factors, and for more than 12 months for recurrent VTE
-
LMWH is safe and effective for the long‐term treatment of VTE in selected patients (and may be preferable for patients with cancer)
ESC Guidelines for the Treatment of PE
According to the 2008 ESC guidelines, anticoagulation with UFH, LMWH, or fondaparinux should be initiated immediately in patients with confirmed PE, as well as in those with a high or intermediate clinical probability of PE while the diagnostic workup is ongoing. Subcutaneous LMWH or fondaparinux is preferable to intravenous UFH for initial treatment in most patients. UFH, however, should be used in patients with a high risk of bleeding due to its capacity for reversal and short half‐life, as well as in those with severe renal dysfunction.10
According to the ESC, patients with high‐risk PE (presenting with cardiogenic shock or persistent arterial hypotension) should receive thrombolytic therapy as first‐line therapy. Hemodynamic and respiratory support is also necessary for these patients.10 Routine thrombolysis is not recommended in patients with non‐high‐risk PE, but it may be considered in select patients with intermediate‐risk PE (characterized by severe right ventricular dysfunction on echocardiography and/or myocardial injury), depending on the patient's risk of bleeding. Thrombolytic therapy should not be used in patients with low‐risk PE (presenting without shock, hypotension, right ventricular dysfunction, or myocardial injury).10
Like the ESC guidelines, the ACCP guidelines recommend against the use of thrombolytic therapy for the majority of patients with PE (Grade 1B), but they do recommend its use in patients with evidence of hemodynamic compromise and no major contraindications owing to bleeding risk (Grade 1B) and in certain other high‐risk patients (Grade 2B).20
The ESC states that pulmonary embolectomy has recently become a reasonable option for patients with massive, high‐risk PE and an absolute contraindication to thrombolysis, or in whom thrombolysis has failed, when appropriate expertise is available. In the past, it was performed as a last resort in patients with massive PE who were in shock and conferred a high risk of mortality (+50%). Recently, however, the procedure has been revived and performed immediately in patients with confirmed massive PE (with severe right ventricular dysfunction but before shock), with mortality rates of less than 10%.19 It should be noted, however, that the ACCP guidelines consider embolectomy a Grade 2C recommendation.20 Alternatively, catheter embolectomy or fragmentation of proximal pulmonary arterial clots may be considered as an alternative to surgical treatment in these patients.10
NCCN Guidelines: Oncology Patients
The NCCN has provided treatment algorithms for the management of DVT and PE in patients with cancer, which are available online at
FWG Guidelines: Oncology Patients
At the 2008 ASH annual meeting, the FWG presented updated guidelines for the treatment of VTE in cancer patients.34 The FWG guidelines contain the following key recommendations:
-
The treatment of VTE should be based on LMWH at curative doses for at least 3 months
-
During the initial treatment (up to 10 days), any approved drug (including LMWH, UFH, and fondaparinux) may be used
-
Beyond the first 10 days, VTE treatment should be based on LMWH at curative doses for at least 3 months and optimally 6 months, as validated with the following drugs and dosage regimens:
-
Dalteparin 200 IU/kg once daily for 1 month, then 150 IU/kg once daily
-
Enoxaparin 150 IU/kg (1.5 mg/kg) once daily
-
Tinzaparin 175 IU/kg once daily
-
Special treatment considerations include the following:
-
In severe renal impairment, UFH should be used and rapidly followed by a vitamin K agonist (VKA) for at least 3 months
-
In severe PE (representing hemodynamic failure), the indications and recommended uses of thrombolytic drugs in noncancer patients apply
-
In patients with an absolute contraindication to anticoagulation or VTE recurrence despite optimal anticoagulation, vena cava filters should be considered
-
In patients with intracranial malignancies, VTE treatment is the same as in cancer patients with nonintracranial tumors
The treatment of central venous catheter thrombosis requires the long‐term use of LMWH according to the FWG guidelines. In patients with severe renal failure, UFH with early VKA must be used as an alternative treatment. Regardless of the therapy used, treatment should be continued as long as the catheter is maintained.34
Long‐Term Management of VTE
The high rate of recurrent VTE after a first episode of DVT or PEapproximately 8% within 90 daysunderscores the importance of maintaining effective prophylaxis postdischarge.35 Inadequate prophylaxis following discharge from the hospital can have severe consequences. In a recent study of 10,744 patients who were discharged from the hospital following hip or knee replacement surgery, fewer than 1 in 5 received postdischarge thromboprophylaxis. The 3‐month risk of mortality was significantly lower among those who received thromboprophylaxis at discharge (adjusted hazard ratio, 0.34; 95% CI, 0.20‐0.57).36
Detailed patient education at the time of discharge may be one of the most effective ways to prevent or minimize the burden of long‐term complications such as PTS or recurrent VTE. Accordingly, proper discharge planning and postdischarge support, including an appropriate anticoagulant, are critical steps toward reducing mortality, morbidity, and healthcare costs.
PTS
As many as 50% of patients with VTE will develop PTS, a serious but preventable complication that leads to pain, swelling, and skin changes in the affected limb. Female gender, older age, higher body mass index (BMI), and DVT of the common femoral or iliac vein (vs. distal DVT) are associated with an increased risk of PTS.37 To prevent PTS in a patient who has had a symptomatic proximal DVT, current guidelines recommend the use of graduated elastic compression stockings with an ankle pressure of 30 to 40 mm Hg, if feasible. Compression therapy should start as soon as possible after the initiation of anticoagulation therapy and be encouraged for a minimum of 2 years.20
Recurrent VTE
After discontinuing anticoagulation, the risk of recurrent VTE increases steadily over time. In a recent long‐term study of patients with acute proximal DVT or PE, the risk of recurrent VTE was 11% after 1 year, 20% after 3 years, 30% after 5 years, and 40% after 10 years. In this study, risk factors for recurrent VTE included unprovoked initial VTE, thrombophilia, increasing age, and a shorter duration of anticoagulation (6 months or less).38 Another study identified residual venous thrombosis as an important risk factor for recurrent VTE.39 In addition, 1 meta‐analysis found that men had a 50% higher risk of recurrent VTE than women.40 Recurrent DVT events are associated with a 21% greater cost than the initial event, suggesting that recurrent VTE is a preventable drain on healthcare resources.41
Secondary Prevention
The risk of recurrent VTE is determined by the effectiveness of treatment for the acute episode of VTE and by the patient's intrinsic risk of thromboembolism. The ACCP recommends different durations of warfarin or LMWH anticoagulant therapy according to these features (Table 2).20 Attaching a high value to prevention of recurrent VTE and a lower value to the burden of long‐term anticoagulant treatment, the ACCP recommends long‐term treatment for patients with a first unprovoked proximal DVT, no risk factors for bleeding, and the ability to monitor the anticoagulant effectively (Grade 1A).20
| Clinical Features | Duration | Grade |
|---|---|---|
| ||
| First episode and transient risk factors | 3 months | 1A |
| Unprovoked episode | 3 months | 1A |
| Unprovoked proximal DVT with low bleed risk | Long‐term | 1A |
| Cancer | 3 to 6 months with LMWH; then with a VKA or a LMWH indefinitely or until cancer is resolved | 1A; 1C |
| Second unprovoked episode | Indefinite | 2A |
Transition to Outpatient Therapy
The use of outpatient LMWH has changed the course of long‐term anticoagulation therapy and is listed as the preferred option for anticoagulation in the ACCP guidelines.20 With the availability of subcutaneous LMWHs, patients with acute VTE no longer have to be hospitalized for the initiation of oral therapy. In addition, patients undergoing invasive procedures that require temporary discontinuation of warfarin can opt for bridge therapy with LMWH.42
Conclusions
The diagnosis of VTE is challenging and depends on the integration of clinical, biochemical, and imaging modalities. In the absence of contraindications, treatment should be initiated immediately after a diagnosis of VTE is confirmed. Anticoagulant therapy alone is sufficient for most patients, but some patients may require thrombolytics or other strategies. Various societies and organizations have issued recommendations regarding the optimal use of these therapies in specific patient populations. Following these recommendations carefully may reduce the risk of complications in patients with VTE.
Despite the availability of effective thromboprophylaxis, the prevalence of venous thromboembolism (VTE) is increasing in the hospital setting. In 2008, the Fifth Annual Health Grades Patient Safety in American Hospitals Study reported on key patient safety incidents among nearly 41 million hospitalizations in the Medicare population between 2004 and 2006. Although many areas showed improvementincluding reduced rates of hospital‐related infections, postoperative bleeding, transfusion reactions, and other injuriesthe number of cases of postoperative VTE increased by 11% during this period.1
Even with optimal thromboprophylaxis, VTE will develop in some at‐risk patients. Early diagnosis and treatment of VTE is critical to reduce morbidity and mortality, but no single tool can definitively confirm its presence. Consequently, the detection of deep vein thrombosis (DVT) and pulmonary embolism (PE) requires a stepwise diagnostic strategy that combines clinical, biochemical, and imaging modalities.
In addition to outlining diagnostic strategies for DVT and PE, this article summarizes VTE treatment guidelines from various organizations and societies and discusses long‐term management strategies to prevent recurrent VTE and other complications.
Diagnosis of DVT
The clinical symptoms and signs of DVT are nonspecific and include unilateral calf, leg, or thigh swelling and pain. Despite the limited sensitivity and specificity of individual signs and symptoms of DVT, the combination of these variables can be useful in assessing the probability of VTE. Patients can be risk stratified according to the likelihood of DVT, as determined by implicit clinical judgment or by a validated prediction rule.2
Assessment of Clinical Probability
The Wells prediction rule is used in assessing the probability of DVT.3 It incorporates signs, symptoms, and risk factors of DVT to calculate a clinical probability rating. Specifically, 1 point is assigned to each of the following factors, if present:3
-
Active cancer (treatment ongoing, within 6 months, or palliative)
-
Calf swelling >3 cm asymptomatic side (measured 10 cm below tibial tuberosity)
-
Collateral superficial veins (nonvaricose)
-
Entire leg swelling
-
Localized tenderness along the distribution of the deep venous system
-
Paralysis, paresis, or recent plaster immobilization of the lower extremities
-
Pitting edema confined to the symptomatic leg
-
Recently bedridden more than 3 days or major surgery within 4 weeks
In addition, 2 points are subtracted if an alternative diagnosis is as likely as or more likely than DVT. In patients with symptoms in both legs, the more symptomatic leg is used.
Patients with low (score <1), moderate (score 1‐2), and high (score 3) pretest probability of DVT have been shown to have DVT prevalence rates of 3%, 17%, and 75%, respectively.3
D‐Dimer Testing
D‐dimer testing measures the small protein fragments remaining in the blood after a cross‐linked fibrin clot is degraded by fibrinolysis. A low clinical probability assessment combined with a negative result in a highly sensitive, enzyme‐linked immunosorbent assay (ELISA)‐based D‐dimer test can safely exclude DVT, with a negative predictive value of 99.1% (95% confidence interval [CI]; 96.7‐99.9).4
Due to its poor specificity, D‐dimer testing has limited utility in unselected inpatients, especially older patients and those who have undergone prolonged hospitalization.5 However, it is reasonable to obtain a highly sensitive, ELISA‐based D‐dimer test in carefully selected inpatients with a low pretest probability of DVT.5, 6 In such patients, a negative result indicates that DVT is highly unlikely, while a positive result indicates a need for further testing. D‐dimer testing is likely not helpful in moderate‐risk or high‐risk patients.
Diagnostic Imaging
For patients with a moderate to high pretest probability of DVT, ultrasound is recommended.6 Compression ultrasonography (CUS) is currently the preferred imaging tool in patients with suspected DVT because it is noninvasive, can be repeated serially, and offers high sensitivity (+90%) and high specificity (95%) for detecting proximal vein thrombosis.7, 8 If the clinical suspicion of DVT persists after an initial negative CUS study, imaging can be repeated after 3 to 7 days to detect the propagation of any thrombosis to the proximal veins. Limitations of CUS include poor visualization of deep iliac and pelvic veins and poor sensitivity in isolated or nonocclusive calf vein thrombi.2
Contrast venography was considered the gold standard for the detection of DVT of the lower extremity, but this modality is invasive, painful, and offers poor visualization of the deep femoral vein and the internal iliac vein. In addition, contrast venography is associated with an increased risk of new thrombosis, renal failure, and hypersensitivity reaction to contrast media. Consequently, contrast venography is currently used in symptomatic patients only when noninvasive testing is inconclusive or unavailable.2 Other second‐line diagnostic tools include computed tomography venography (CTV) and magnetic resonance venography (MRV).9
Diagnostic Strategy
A diagnostic algorithm for DVT is presented in Figure 1. First, a validated clinical prediction scale such as the Wells prediction rule should be used to estimate the pretest probability of DVT, and the result of the clinical assessment should influence the choice and interpretation of subsequent testing.

Abbreviations: CUS, compression ultrasonography; DVT, deep vein thrombosis (DVT).
Diagnosis of PE
Clinical symptoms and signs such as dyspnea, chest pain, tachycardia, tachypnea, and syncope raise the suspicion of PE. Individual signs and symptoms, however, cannot confirm or exclude acute PE, as they are neither sensitive nor specific.10 Furthermore, although the likelihood of PE increases with the number of predisposing risk factors, approximately 30% of PE cases are unprovoked or idiopathic, meaning that they occur in the absence of predisposing factors. Diagnosis, therefore, depends on an integrated strategy involving similar tools as those used in diagnosing DVT.
Assessing Clinical Probability
Wells et al.11 also developed a clinical prediction rule for the risk stratification of patients with suspected PE. In this model, 7 specified variables are assigned different scores: clinical signs and symptoms of DVT (3.0); lack of a likely alternative diagnosis (3.0); heart rate greater than 100 beats per minute (1.5); immobilization for more than 3 days or surgery in the previous 4 weeks (1.5); previous DVT/PE (1.5); hemoptysis (1.0); and malignancy (1.0). Although the Wells prediction rule initially categorized 3 levels of probability for PE (low, moderate, or high), a revised model uses a simplified, dichotomized approach to determine whether PE is likely (Wells score >4) or unlikely (4 Wells score).11 An independent, prospective observational study found that the Wells prediction model reliably risk‐stratified pretest probability in patients with suspected PE.12
For patients who are stratified into the low‐risk category, the pulmonary embolism rule‐out criteria (PERC) rule may be helpful in reducing unnecessary diagnostic testing for PE.13 The PERC rule consists of 8 variables designed to offer a pretest probability of PE of less than 1.8%, a probability at which further testing is unnecessary. If the clinical gestalt is that PE is unlikely and all of the following variables are present, further testing can be safely discontinued: (1) pulse <100; (2) age <50; (3) oxygen saturation (SaO2) >94%; (4) no unilateral leg swelling; (5) no hemoptysis; (6) no recent trauma or surgery; (7) no prior DVT or PE; and (8) no hormone use.13 In a large, multicenter study, these criteria combined with a gestalt interpretation of low risk were shown to select a subgroup of patients with a very low probability of PE (<2%).14
D‐Dimer Testing
Evidence suggests that the combination of a low clinical probability assessment and a normal result in a highly sensitive, ELISA‐based D‐dimer test can safely exclude PE in hospitalized patients.15 Due to the large number of comorbidities among hospitalized patients, however, this combination occurs in only approximately 10% of inpatients.15 D‐dimer levels may be elevated in patients with a variety of nonthrombotic conditions, and it is therefore most useful in the diagnosis of otherwise healthy patients who have symptoms of PE. D‐dimer testing is not appropriate in moderate‐risk or high‐risk patients.
Diagnostic Imaging
Computed tomography (CT) is a leading imaging modality for the exclusion or confirmation of PE, as well as for the detection of alternative diagnoses. The diagnostic algorithms endorsed by the European Society of Cardiology (ESC) rely on both single‐detector and multidetector CT. However, multidetector CT scanners are now preferred because, in contrast to single‐detector CT, they can detect pulmonary emboli in smaller pulmonary arteries.10 Because single‐detector CT has a limited sensitivity of approximately 70%, it must be used in conjunction with lower limb venous CUS.16 In contrast, multidetector CT angiography has high sensitivity (83%) and specificity (96%) for the detection of PE and does not require the additional use of lower limb venous CUS.16, 17
Diagnostic Strategy
The Christopher Study demonstrated the utility of a diagnostic algorithm that incorporates a dichotomized decision rule, D‐dimer testing, and CT. In this approach, PE is excluded in patients with an unlikely clinical probability score (Wells score 4) and a normal D‐dimer test result. In all other patients, CT is the sole imaging method used to make management decisions.18 However, in patients with massive pulmonary embolism, if CT angiography is not immediately available, selective pulmonary angiography has been performed to identify and localize the emboli before aggressive therapy is instituted (Figure 2).19 If the patient is critically ill (hypotensive, severely hypoxemic), empiric treatment is appropriate while diagnostic strategy is being formulated.

Abbreviations: CT, computed tomography; CXR, plain chest X‐ray; ECG, electrocardiogram; PE, pulmonary embolism. †CT angiography using multidetector instruments.
Treatment Options for VTE
For patients with VTE, the American College of Chest Physicians (ACCP) guidelines recommend initial treatment with low‐molecular‐weight heparin (LMWH), intravenous unfractionated heparin (UFH), or adjusted‐dose subcutaneous UFH, followed by at least 3 months of oral anticoagulation therapy.20
When VTE is diagnosed, anticoagulation should be initiated immediately unless contraindications are present. In addition, patients without contraindications to anticoagulation should receive treatment before diagnostic testing if such testing is delayed or if the clinical suspicion of VTE is high.20
Anticoagulant Treatment
For decades, parenteral administration of UFH for 5 to 7 days followed by long‐term warfarin therapy has been the conventional treatment of patients with VTE. Although UFH can be administered subcutaneously or by intravenous (IV) infusion, continuous IV infusion has been preferred because of superior dosing precision. The anticoagulation effect of intravenous UFH must be monitored to ensure a therapeutic activated partial thromboplastin time (aPTT). Consequently, the use of intravenous UFH requires frequent aPTT assessment and dose adjustment.20
Given their ease of use and improved pharmacokinetic and pharmacodynamic profiles, LMWHs have replaced UFH for the treatment of VTE in many institutions. Fondaparinux is also a safe and effective alternative to both intravenous UFH and LMWH in the treatment of VTE.20 It has a longer half‐life (15‐20 hours) than LMWH, permitting a once‐daily administration, and in patients with submassive PE, its efficacy and safety are comparable to UFH.21 Platelet count monitoring is not necessary with fondaparinux because it is given at weight‐adjusted doses, and only 1 case of heparin‐induced thrombocytopenia (HIT) has been reported.22 It is, however, contraindicated in renal failure with a creatinine clearance of <30 mL/minute.10
Warfarin is very effective in the long‐term management of VTE and should be started concurrently with rapid‐acting injectable anticoagulation therapy. Warfarin requires overlap with injectable anticoagulants for a minimum of 5 days until a therapeutic international normalized ratio (INR) has been achieved.20
Other Treatments
Most patients with VTE can be treated effectively with only anticoagulation therapy. However, in cases of massive PE (with or without systemic arterial hypotension and usually with significant hypoxemia not generally responsive to supplemental oxygen), removal of the occluding thrombus by thrombolytic agents, special clot‐removing catheters, or surgical procedures may be necessary to prevent or ameliorate shock and subsequent death.23 In other cases, such as when anticoagulants are ineffective or contraindicated, an inferior vena cava (IVC) filter may be an appropriate option for VTE treatment. Importantly, guidelines do not recommend filters in patients who can tolerate anticoagulation.
Permanent and retrievable IVC filters are effective at preventing PE and are generally associated with a low complication rate.24 However, nonfatal complications are relatively common with permanent IVC filters. One early complication is insertion‐site thrombosis, which occurs in about 10% of patients. Subsequent complications are more frequent and include recurrent DVT and post‐thrombotic syndrome (PTS), which occur in approximately 20% and 40% of patients, respectively. At 5 and 9 years, about 22% and 33% of the filters are occluded, regardless of the use and duration of anticoagulation.2527 To minimize these complications, retrievable filters have been increasingly used, but most filters are not retrieved and are subject to the same complications as permanent IVC filters.28
Catheter‐directed thrombolysis, with or without IVC filter placement, is safe and effective in treating acute DVT.29 Additional measures, such as the use of graduated compression stockings, can reduce the risk of developing PTS.20
Guideline Recommendations
Guidelines from the ACCP, the American College of Physicians (ACP), and the American Academy of Family Physicians (AAFP) address the treatment of VTE in a broad spectrum of patients. Additional guidelines provide recommendations for specific presentations or patient groups. For example, the ESC guidelines address the treatment of acute PE, and several groupsthe American Society of Clinical Oncology (ASCO), the National Comprehensive Cancer Network (NCCN), and the French Working Group (FWG)have published guidelines for the treatment of VTE in patients with cancer. The following sections summarize the most important recommendations from several of these organizations and societies.
ACCP Guidelines
The ACCP guideline recommendations are assigned grades of 1 or 2, denoting a stronger or weaker recommendation, as well as a grade of A, B, or C, indicating high‐quality evidence, moderate‐quality evidence, and low‐quality evidence, respectively. Physicians must supplement the guideline recommendations with informed clinical judgment to ensure proper use of treatment in at‐risk hospitalized patients.20
The 2008 ACCP guidelines suggest several options for the initial treatment of VTE, which are listed, along with acceptable dosing regimens, in Table 1.20, 30, 31 Fixed‐dose, unmonitored, subcutaneous UFH and fondaparinux are new Grade 1A additions to the 2008 update. In general, LMWH is preferred over intravenous UFH, except in patients with severe renal failure.20
| Initial Anticoagulation Therapy | Grade | Acceptable Treatment Regimen* |
|---|---|---|
| ||
| SC LMWH | 1A | Enoxaparin: 1 mg/kg every 12 hours or 1.5 mg/kg once daily; Dalteparin: 200 IU/kg once daily (can be administered out of hospital) |
| Intravenous UFH | 1A | Get baseline aPTT, PT, and platelet count; if no abnormalities, proceed with a weight‐based heparin infusion protocol such as: |
| Bolus of 80 U/kg, followed by an infusion of 18 U/kg per hour (treatment duration 72 days); check aPTT every 4‐6 hours and adjust according to the normogram; monitor platelet count every 3‐4 days for HIT | ||
| Monitored SC UFH; fixed‐dose, unmonitored, SC UFH | 1A; 1A | Initial dose of 333 U/kg, followed by a fixed dose of 250 U/kg every 12 hours (can be administered out of hospital) |
| SC fondaparinux | 1A | 5 mg (body weight <50 kg), 7.5 mg (body weight 50‐100 kg), or 10 mg (body weight >100 kg) once daily (treatment duration 72 days) |
| IVC filter if anticoagulation contraindicated | 1C | |
Warfarin should also be initiated on the same day as UFH or LMWH and adjusted to a target INR of 2.5 (range, 2.0‐3.0). Treatment with UFH or LMWH should be continued concomitantly for a minimum of 5 days and should not be discontinued until the INR has been over 2.0 for 24 hours. The ACCP guidelines also recommend systematic follow‐up of oral anticoagulation therapy.
ACP/AAFP Guidelines
In 2007, the ACP and the AAFP collaborated to develop joint guidelines for the management of VTE.32 Several of their key recommendations are the following:32
-
LMWH, rather than UFH, should be used whenever possible for the initial inpatient treatment of DVT
-
Either UFH or LMWH is appropriate for the initial treatment of PE
-
Anticoagulation should be continued for 3 to 6 months for VTE secondary to transient risk factors, and for more than 12 months for recurrent VTE
-
LMWH is safe and effective for the long‐term treatment of VTE in selected patients (and may be preferable for patients with cancer)
ESC Guidelines for the Treatment of PE
According to the 2008 ESC guidelines, anticoagulation with UFH, LMWH, or fondaparinux should be initiated immediately in patients with confirmed PE, as well as in those with a high or intermediate clinical probability of PE while the diagnostic workup is ongoing. Subcutaneous LMWH or fondaparinux is preferable to intravenous UFH for initial treatment in most patients. UFH, however, should be used in patients with a high risk of bleeding due to its capacity for reversal and short half‐life, as well as in those with severe renal dysfunction.10
According to the ESC, patients with high‐risk PE (presenting with cardiogenic shock or persistent arterial hypotension) should receive thrombolytic therapy as first‐line therapy. Hemodynamic and respiratory support is also necessary for these patients.10 Routine thrombolysis is not recommended in patients with non‐high‐risk PE, but it may be considered in select patients with intermediate‐risk PE (characterized by severe right ventricular dysfunction on echocardiography and/or myocardial injury), depending on the patient's risk of bleeding. Thrombolytic therapy should not be used in patients with low‐risk PE (presenting without shock, hypotension, right ventricular dysfunction, or myocardial injury).10
Like the ESC guidelines, the ACCP guidelines recommend against the use of thrombolytic therapy for the majority of patients with PE (Grade 1B), but they do recommend its use in patients with evidence of hemodynamic compromise and no major contraindications owing to bleeding risk (Grade 1B) and in certain other high‐risk patients (Grade 2B).20
The ESC states that pulmonary embolectomy has recently become a reasonable option for patients with massive, high‐risk PE and an absolute contraindication to thrombolysis, or in whom thrombolysis has failed, when appropriate expertise is available. In the past, it was performed as a last resort in patients with massive PE who were in shock and conferred a high risk of mortality (+50%). Recently, however, the procedure has been revived and performed immediately in patients with confirmed massive PE (with severe right ventricular dysfunction but before shock), with mortality rates of less than 10%.19 It should be noted, however, that the ACCP guidelines consider embolectomy a Grade 2C recommendation.20 Alternatively, catheter embolectomy or fragmentation of proximal pulmonary arterial clots may be considered as an alternative to surgical treatment in these patients.10
NCCN Guidelines: Oncology Patients
The NCCN has provided treatment algorithms for the management of DVT and PE in patients with cancer, which are available online at
FWG Guidelines: Oncology Patients
At the 2008 ASH annual meeting, the FWG presented updated guidelines for the treatment of VTE in cancer patients.34 The FWG guidelines contain the following key recommendations:
-
The treatment of VTE should be based on LMWH at curative doses for at least 3 months
-
During the initial treatment (up to 10 days), any approved drug (including LMWH, UFH, and fondaparinux) may be used
-
Beyond the first 10 days, VTE treatment should be based on LMWH at curative doses for at least 3 months and optimally 6 months, as validated with the following drugs and dosage regimens:
-
Dalteparin 200 IU/kg once daily for 1 month, then 150 IU/kg once daily
-
Enoxaparin 150 IU/kg (1.5 mg/kg) once daily
-
Tinzaparin 175 IU/kg once daily
-
Special treatment considerations include the following:
-
In severe renal impairment, UFH should be used and rapidly followed by a vitamin K agonist (VKA) for at least 3 months
-
In severe PE (representing hemodynamic failure), the indications and recommended uses of thrombolytic drugs in noncancer patients apply
-
In patients with an absolute contraindication to anticoagulation or VTE recurrence despite optimal anticoagulation, vena cava filters should be considered
-
In patients with intracranial malignancies, VTE treatment is the same as in cancer patients with nonintracranial tumors
The treatment of central venous catheter thrombosis requires the long‐term use of LMWH according to the FWG guidelines. In patients with severe renal failure, UFH with early VKA must be used as an alternative treatment. Regardless of the therapy used, treatment should be continued as long as the catheter is maintained.34
Long‐Term Management of VTE
The high rate of recurrent VTE after a first episode of DVT or PEapproximately 8% within 90 daysunderscores the importance of maintaining effective prophylaxis postdischarge.35 Inadequate prophylaxis following discharge from the hospital can have severe consequences. In a recent study of 10,744 patients who were discharged from the hospital following hip or knee replacement surgery, fewer than 1 in 5 received postdischarge thromboprophylaxis. The 3‐month risk of mortality was significantly lower among those who received thromboprophylaxis at discharge (adjusted hazard ratio, 0.34; 95% CI, 0.20‐0.57).36
Detailed patient education at the time of discharge may be one of the most effective ways to prevent or minimize the burden of long‐term complications such as PTS or recurrent VTE. Accordingly, proper discharge planning and postdischarge support, including an appropriate anticoagulant, are critical steps toward reducing mortality, morbidity, and healthcare costs.
PTS
As many as 50% of patients with VTE will develop PTS, a serious but preventable complication that leads to pain, swelling, and skin changes in the affected limb. Female gender, older age, higher body mass index (BMI), and DVT of the common femoral or iliac vein (vs. distal DVT) are associated with an increased risk of PTS.37 To prevent PTS in a patient who has had a symptomatic proximal DVT, current guidelines recommend the use of graduated elastic compression stockings with an ankle pressure of 30 to 40 mm Hg, if feasible. Compression therapy should start as soon as possible after the initiation of anticoagulation therapy and be encouraged for a minimum of 2 years.20
Recurrent VTE
After discontinuing anticoagulation, the risk of recurrent VTE increases steadily over time. In a recent long‐term study of patients with acute proximal DVT or PE, the risk of recurrent VTE was 11% after 1 year, 20% after 3 years, 30% after 5 years, and 40% after 10 years. In this study, risk factors for recurrent VTE included unprovoked initial VTE, thrombophilia, increasing age, and a shorter duration of anticoagulation (6 months or less).38 Another study identified residual venous thrombosis as an important risk factor for recurrent VTE.39 In addition, 1 meta‐analysis found that men had a 50% higher risk of recurrent VTE than women.40 Recurrent DVT events are associated with a 21% greater cost than the initial event, suggesting that recurrent VTE is a preventable drain on healthcare resources.41
Secondary Prevention
The risk of recurrent VTE is determined by the effectiveness of treatment for the acute episode of VTE and by the patient's intrinsic risk of thromboembolism. The ACCP recommends different durations of warfarin or LMWH anticoagulant therapy according to these features (Table 2).20 Attaching a high value to prevention of recurrent VTE and a lower value to the burden of long‐term anticoagulant treatment, the ACCP recommends long‐term treatment for patients with a first unprovoked proximal DVT, no risk factors for bleeding, and the ability to monitor the anticoagulant effectively (Grade 1A).20
| Clinical Features | Duration | Grade |
|---|---|---|
| ||
| First episode and transient risk factors | 3 months | 1A |
| Unprovoked episode | 3 months | 1A |
| Unprovoked proximal DVT with low bleed risk | Long‐term | 1A |
| Cancer | 3 to 6 months with LMWH; then with a VKA or a LMWH indefinitely or until cancer is resolved | 1A; 1C |
| Second unprovoked episode | Indefinite | 2A |
Transition to Outpatient Therapy
The use of outpatient LMWH has changed the course of long‐term anticoagulation therapy and is listed as the preferred option for anticoagulation in the ACCP guidelines.20 With the availability of subcutaneous LMWHs, patients with acute VTE no longer have to be hospitalized for the initiation of oral therapy. In addition, patients undergoing invasive procedures that require temporary discontinuation of warfarin can opt for bridge therapy with LMWH.42
Conclusions
The diagnosis of VTE is challenging and depends on the integration of clinical, biochemical, and imaging modalities. In the absence of contraindications, treatment should be initiated immediately after a diagnosis of VTE is confirmed. Anticoagulant therapy alone is sufficient for most patients, but some patients may require thrombolytics or other strategies. Various societies and organizations have issued recommendations regarding the optimal use of these therapies in specific patient populations. Following these recommendations carefully may reduce the risk of complications in patients with VTE.
- Health Grades, Inc. The Fifth Annual Health Grades Patient Safety in American Hospitals Study. Available at: http://www.healthgrades.com/media/dms/pdf/PatientSafetyInAmericanHospitalsStudy2008.pdf. Accessed August2009.
- , , , et al.Prevention of venous thromboembolism: American College of Chest Physicians evidence‐based clinical practice guidelines. 8th Edition.Chest2008;133(6 suppl):381S–453S.
- , , , et al.Value of assessment of pretest probability of deep‐vein thrombosis in clinical management.Lancet.1997;350(9094):1795–1798.
- , , , et al.Evaluation of D‐dimer in the diagnosis of suspected deep‐vein thrombosis.N Engl J Med.2003;349:1227–1235.
- , , , et al.Limitations of D‐dimer testing in unselected inpatients with suspected venous thromboembolism.Am J Med.2003;114(4):276–282.
- , , , et al.Current diagnosis of venous thromboembolism in primary care: a clinical practice guideline from the American Academy of Family Physicians and the American College of Physicians.Ann Fam Med.2007;5(1):57–62.
- , , .The role of venous ultrasonography in the diagnosis of suspected deep venous thrombosis and pulmonary embolism.Ann Intern Med.1998;129:1044–1049.
- , .Ultrasonography of leg veins in patients suspected of having pulmonary embolism.Ann Intern Med.1998;128:243–245.
- , .Role of computed tomography and magnetic resonance imaging for deep venous thrombosis and pulmonary embolism.Circulation.2004;109(12 suppl 1):I15–I21.
- , , , et al.Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology.Guidelines on the diagnosis and management of acute pulmonary embolism: the Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology (ESC).Eur Heart J.2008;29(18):2276–2315.
- , , , et al.Derivation of a simple clinical model to categorize patients probability of pulmonary embolism: increasing the models utility with the SimpliRED D‐dimer.Thromb Haemost.2000;83(3):416–420.
- , , , et al.Prospective validation of Wells criteria in the evaluation of patients with suspected pulmonary embolism.Ann Emerg Med.2004;44(5):503–510.
- , , , et al.Clinical criteria to prevent unnecessary diagnostic testing in emergency department patients with suspected pulmonary embolism.J Thromb Haemost.2004;2(8):1247–1255.
- , , , et al.Prospective multicenter evaluation of the pulmonary embolism rule‐out criteria.J Thromb Haemost.2008;6(5):772–780.
- , , , et al.A simple diagnostic strategy in hospitalized patients with clinically suspected pulmonary embolism.J Intern Med.2006;260(5):459–466.
- , .Is computed tomographic venography of lower limbs useful in suspected pulmonary embolism?Rev Med Suisse.2008;4(143):354,356–359.
- , , , et al.Multidetector computed tomography for acute pulmonary embolism.N Engl J Med.2006;354(22):2317–2327.
- , , , et al.Christopher Study Investigators.Effectiveness of managing suspected pulmonary embolism using an algorithm combining clinical probability, D‐dimer testing, and computed tomography.JAMA.2006;295(2):172–179.
- , , , .Acute pulmonary embolectomy: a contemporary approach.Circulation.2002;105:1416–1419.
- , , , et al.Antithrombotic therapy for venous thromboembolic disease: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines (8th Edition).Chest.2008;133(6 suppl):454S–545S.
- , , , et al.Subcutaneous fondaparinux versus unfractionated heparin in the initial treatment of pulmonary embolism.N Engl J Med.2003;349:1695–1702.
- .Heparin‐induced thrombocytopenia associated with fondaparinux.N Engl J Med.2007;356:2653–2655.
- , , , .Thrombolysis compared with heparin for the initial treatment of pulmonary embolism: a meta‐analysis of the randomized controlled trials.Circulation.2004;110:744–749.
- , , , et al.Ins and outs of inferior vena cava filters in patients with venous thromboembolism: the experience at Monash Medical Centre and review of the published reports.Intern Med J.2008;38(1):38–43.
- PREPIC Study Group.Eight‐year follow‐up of patients with permanent vena cava filters in the prevention of pulmonary embolism: the PREPIC (Prevention du Risque d'Embolie Pulmonaire par Interruption Cave) randomized study.Circulation.2005;112:416–422.
- , , , .Inferior vena cava filters: key considerations.Am J Med Sci.2005;330:82–87.
- , , , .Percutaneous inferior vena cava filters: follow‐up of 7 designs in 320 patients.Radiology.1993;188:851–856.
- , , , et al.Practice patterns and outcomes of retrievable vena cava filters in trauma patients: an AAST multicenter study.J Trauma.2007;62:17–24.
- , , , , , .Long‐term outcomes of catheter directed thrombolysis for lower extremity deep venous thrombosis without prophylactic inferior vena cava filter placement.J Vasc Surg.2007;45(5):992–997.
- , , , et al.Comparison of fixed‐dose weight‐adjusted unfractionated heparin and low‐molecular‐weight heparin for acute treatment of venous thromboembolism.JAMA.2006;296(8):935–942.
- Arixtra prescribing information. Last updated October 2008. Research Triangle Park, NC: GlaxoSmithKline. Available at: http://us.gsk.com/products/assets/us_arixtra.pdf. Accessed August2009.
- , , , et al.American College of Physicians;American Academy of Family Physicians Panel on Deep Venous Thrombosis/Pulmonary Embolism.Management of venous thromboembolism: a clinical practice guideline from the American College of Physicians and the American Academy of Family Physicians.Ann Intern Med.2007;146(3):204–210.
- National Comprehensive Cancer Network (NCCN). Venous thromboembolic disease. Practice Guidelines in Oncology. V.1.2009. Available at: http://www.nccn.org/professionals/physician_gls/PDF/vte.pdf. Accessed August2009.
- , , , et al. Guidelines for the treatment of venous thromboembolism in cancer patients: report from the French Working Group. Presented at the 50th Annual Meeting of the American College of Hematology; San Francisco, CA; December 6‐9, 2008. Abstract 1284.
- , , , et al.Predictors of recurrence after deep vein thrombosis and pulmonary embolism: a population‐based cohort study.Arch Intern Med.2000;160(6):761–768.
- , , , et al.Postdischarge thromboprophylaxis and mortality risk after hip‐or knee‐replacement surgery.CMAJ.2008;178(12):1545–1554.
- , , , et al.Determinants and time course of the postthrombotic syndrome after acute deep venous thrombosis.Ann Intern Med.2008;149(10):698–707.
- , , , et al.The risk of recurrent venous thromboembolism after discontinuing anticoagulation in patients with acute proximal deep vein thrombosis or pulmonary embolism. A prospective cohort study in 1,626 patients.Haematologica.2007;92(2):199–205.
- .Risk factors of recurrent venous thromboembolism: the role of residual vein thrombosis.Pathophysiol Haemost Thromb.2003/2004;33(5‐6):351–353.
- , , , et al.Effect of patient's sex on risk of recurrent venous thromboembolism: a meta‐analysis.Lancet.2006;368:371–378.
- , .Direct medical costs of venous thromboembolism and subsequent hospital readmission rates: an administrative claims analysis from 30 managed care organizations.J Manag Care Pharm.2007;13(6):475–486.
- , .Outpatient management of anticoagulation therapy.Am Fam Physician.2007;75:1031–1042.
- Health Grades, Inc. The Fifth Annual Health Grades Patient Safety in American Hospitals Study. Available at: http://www.healthgrades.com/media/dms/pdf/PatientSafetyInAmericanHospitalsStudy2008.pdf. Accessed August2009.
- , , , et al.Prevention of venous thromboembolism: American College of Chest Physicians evidence‐based clinical practice guidelines. 8th Edition.Chest2008;133(6 suppl):381S–453S.
- , , , et al.Value of assessment of pretest probability of deep‐vein thrombosis in clinical management.Lancet.1997;350(9094):1795–1798.
- , , , et al.Evaluation of D‐dimer in the diagnosis of suspected deep‐vein thrombosis.N Engl J Med.2003;349:1227–1235.
- , , , et al.Limitations of D‐dimer testing in unselected inpatients with suspected venous thromboembolism.Am J Med.2003;114(4):276–282.
- , , , et al.Current diagnosis of venous thromboembolism in primary care: a clinical practice guideline from the American Academy of Family Physicians and the American College of Physicians.Ann Fam Med.2007;5(1):57–62.
- , , .The role of venous ultrasonography in the diagnosis of suspected deep venous thrombosis and pulmonary embolism.Ann Intern Med.1998;129:1044–1049.
- , .Ultrasonography of leg veins in patients suspected of having pulmonary embolism.Ann Intern Med.1998;128:243–245.
- , .Role of computed tomography and magnetic resonance imaging for deep venous thrombosis and pulmonary embolism.Circulation.2004;109(12 suppl 1):I15–I21.
- , , , et al.Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology.Guidelines on the diagnosis and management of acute pulmonary embolism: the Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology (ESC).Eur Heart J.2008;29(18):2276–2315.
- , , , et al.Derivation of a simple clinical model to categorize patients probability of pulmonary embolism: increasing the models utility with the SimpliRED D‐dimer.Thromb Haemost.2000;83(3):416–420.
- , , , et al.Prospective validation of Wells criteria in the evaluation of patients with suspected pulmonary embolism.Ann Emerg Med.2004;44(5):503–510.
- , , , et al.Clinical criteria to prevent unnecessary diagnostic testing in emergency department patients with suspected pulmonary embolism.J Thromb Haemost.2004;2(8):1247–1255.
- , , , et al.Prospective multicenter evaluation of the pulmonary embolism rule‐out criteria.J Thromb Haemost.2008;6(5):772–780.
- , , , et al.A simple diagnostic strategy in hospitalized patients with clinically suspected pulmonary embolism.J Intern Med.2006;260(5):459–466.
- , .Is computed tomographic venography of lower limbs useful in suspected pulmonary embolism?Rev Med Suisse.2008;4(143):354,356–359.
- , , , et al.Multidetector computed tomography for acute pulmonary embolism.N Engl J Med.2006;354(22):2317–2327.
- , , , et al.Christopher Study Investigators.Effectiveness of managing suspected pulmonary embolism using an algorithm combining clinical probability, D‐dimer testing, and computed tomography.JAMA.2006;295(2):172–179.
- , , , .Acute pulmonary embolectomy: a contemporary approach.Circulation.2002;105:1416–1419.
- , , , et al.Antithrombotic therapy for venous thromboembolic disease: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines (8th Edition).Chest.2008;133(6 suppl):454S–545S.
- , , , et al.Subcutaneous fondaparinux versus unfractionated heparin in the initial treatment of pulmonary embolism.N Engl J Med.2003;349:1695–1702.
- .Heparin‐induced thrombocytopenia associated with fondaparinux.N Engl J Med.2007;356:2653–2655.
- , , , .Thrombolysis compared with heparin for the initial treatment of pulmonary embolism: a meta‐analysis of the randomized controlled trials.Circulation.2004;110:744–749.
- , , , et al.Ins and outs of inferior vena cava filters in patients with venous thromboembolism: the experience at Monash Medical Centre and review of the published reports.Intern Med J.2008;38(1):38–43.
- PREPIC Study Group.Eight‐year follow‐up of patients with permanent vena cava filters in the prevention of pulmonary embolism: the PREPIC (Prevention du Risque d'Embolie Pulmonaire par Interruption Cave) randomized study.Circulation.2005;112:416–422.
- , , , .Inferior vena cava filters: key considerations.Am J Med Sci.2005;330:82–87.
- , , , .Percutaneous inferior vena cava filters: follow‐up of 7 designs in 320 patients.Radiology.1993;188:851–856.
- , , , et al.Practice patterns and outcomes of retrievable vena cava filters in trauma patients: an AAST multicenter study.J Trauma.2007;62:17–24.
- , , , , , .Long‐term outcomes of catheter directed thrombolysis for lower extremity deep venous thrombosis without prophylactic inferior vena cava filter placement.J Vasc Surg.2007;45(5):992–997.
- , , , et al.Comparison of fixed‐dose weight‐adjusted unfractionated heparin and low‐molecular‐weight heparin for acute treatment of venous thromboembolism.JAMA.2006;296(8):935–942.
- Arixtra prescribing information. Last updated October 2008. Research Triangle Park, NC: GlaxoSmithKline. Available at: http://us.gsk.com/products/assets/us_arixtra.pdf. Accessed August2009.
- , , , et al.American College of Physicians;American Academy of Family Physicians Panel on Deep Venous Thrombosis/Pulmonary Embolism.Management of venous thromboembolism: a clinical practice guideline from the American College of Physicians and the American Academy of Family Physicians.Ann Intern Med.2007;146(3):204–210.
- National Comprehensive Cancer Network (NCCN). Venous thromboembolic disease. Practice Guidelines in Oncology. V.1.2009. Available at: http://www.nccn.org/professionals/physician_gls/PDF/vte.pdf. Accessed August2009.
- , , , et al. Guidelines for the treatment of venous thromboembolism in cancer patients: report from the French Working Group. Presented at the 50th Annual Meeting of the American College of Hematology; San Francisco, CA; December 6‐9, 2008. Abstract 1284.
- , , , et al.Predictors of recurrence after deep vein thrombosis and pulmonary embolism: a population‐based cohort study.Arch Intern Med.2000;160(6):761–768.
- , , , et al.Postdischarge thromboprophylaxis and mortality risk after hip‐or knee‐replacement surgery.CMAJ.2008;178(12):1545–1554.
- , , , et al.Determinants and time course of the postthrombotic syndrome after acute deep venous thrombosis.Ann Intern Med.2008;149(10):698–707.
- , , , et al.The risk of recurrent venous thromboembolism after discontinuing anticoagulation in patients with acute proximal deep vein thrombosis or pulmonary embolism. A prospective cohort study in 1,626 patients.Haematologica.2007;92(2):199–205.
- .Risk factors of recurrent venous thromboembolism: the role of residual vein thrombosis.Pathophysiol Haemost Thromb.2003/2004;33(5‐6):351–353.
- , , , et al.Effect of patient's sex on risk of recurrent venous thromboembolism: a meta‐analysis.Lancet.2006;368:371–378.
- , .Direct medical costs of venous thromboembolism and subsequent hospital readmission rates: an administrative claims analysis from 30 managed care organizations.J Manag Care Pharm.2007;13(6):475–486.
- , .Outpatient management of anticoagulation therapy.Am Fam Physician.2007;75:1031–1042.
VTE Performance Measures and Strategies
Despite the availability of evidence‐based guidelines for the prevention of thromboembolic morbidity and mortality, venous thromboembolism (VTE) remains a pervasive threat to public health. Prophylaxis is underused for a variety of reasons, which were summarized in the first article of this Supplement. Overcoming these barriers and reducing the incidence of VTE has become a major priority for public health policy.
The Office of the Surgeon General released a report in September 2008 that reflects this sense of urgency and national focus by calling for a coordinated, multifaceted plan to reduce the incidence of VTE in the United States.1 The Surgeon General report is one of the latest in a string of national initiatives designed to improve outcomes in patients at risk of VTE. In the past several years, public and private agencies have launched a range of programs aimed at improving deficiencies in the awareness, prevention, and treatment of VTE in hospitalized patients (these are summarized in Table 1). New performance measures and improvement initiatives may reduce the discrepancies between recommendations and practice, ultimately improving patient outcomes. These measures may possibly become benchmarks for pay‐for‐performance initiatives or future hospital accreditation.
| Measure/Initiative | Description |
|---|---|
| |
| National Quality Forum/The Joint Commission (NQF/TJC) | Public reporting of hospital performance in 6 performance measures; will apply to all medical and surgical patients |
| Surgical Care Improvement Project (SCIP) | Two performance measures enacted with reimbursement implications; 2 outcomes measures |
| American Medical Association Physician Consortium for Performance Improvement (PCPI) | Medical societies collaborating to identify gaps in care and develop performance measures; 1 measure has been endorsed |
| Leapfrog Hospital Quality and Safety Survey | Web database allowing consumers to compare performance among participating hospitals; includes 2 NQF safe practices |
| TJC National Patient Safety Goals (NPSG) | Goals for solving patient safety problems; compliance required for Joint Commission accreditation, with online reporting of results (Quality Check website) |
| North American Thrombosis Forum (NATF) | Nonprofit organization addressing unmet needs related to VTE and other thrombotic disorders |
| American Venous Forum National Venous Screening Program | National VTE awareness campaign; promotes compliance with protocols |
Herein, we review a variety of VTE performance measures, including those from the National Quality Forum (NQF), The Joint Commission (TJC), and the Surgical Care Improvement Project (SCIP). To illustrate how performance measures may be applied in the hospital setting to improve patient care, performance improvement programs that have proven effective in select hospitals across the United States are described.
Performance Measures and Initiatives
National Quality Forum Performance Measures
The NQF and TJC (formerly known as the Joint Commission on Accreditation of Healthcare Organizations) have already enacted performance measures for pneumonia, heart failure, acute myocardial infarction (MI), and other conditions. Since 2005, the NQF and TJC have been collaborating to develop national consensus performance measures for the prevention and care of VTE. The VTE performance measures will apply to all medical and surgical patients and include process measures in the areas of prevention and treatment, as well as outcome measures. After pilot‐testing a range of measures for 3 years, TJC recommended 7 candidate measures in November 2007. In May 2008, the NQF endorsed 6 of these, embracing all TJC recommendations except one relating to the use and documentation of vena cava filter quality improvement (Table 2).2
|
| Risk assessment and prophylaxis |
| 1. Documentation of VTE risk/prophylaxis within 24 hours of hospital admission or surgery end‐time |
| 2. Documentation of VTE risk/prophylaxis within 24 hours after ICU admission, transfer to ICU, or surgery end‐time |
| Treatment |
| 3. Patients with VTE with overlap of parenteral and warfarin anticoagulation therapy for at least 5 days with an INR 2 before discontinuation of parenteral therapy; for > 5 days with an INR < 2 and discharged on overlap therapy; or discharged in < 5 days on overlap therapy |
| 4. Patients with VTE receiving UFH with dosages/platelet count monitoring by protocol or nomogram |
| 5. Patients with VTE or their caregivers are given written discharge instructions or other educational material addressing all of the following: follow‐up monitoring, compliance issues, dietary restrictions, and potential for adverse drug reactions and interactions |
| Outcomes |
| 6. Incidence of potentially preventable hospital‐acquired VTE measured by patients who received no VTE prophylaxis before VTE diagnosis |
The next step is for the NQF to develop a specification manual that defines which patients should be given prophylaxis using International Classification of Diseases, 9th edition (ICD‐9) codes and identifies which interventions are appropriate for each patient population. Current clinical guidelines provide important guidance for appropriate inclusion and exclusion criteria for medical and surgical prophylaxis, as well as evidence‐based recommendations for the treatment of VTE.3, 4
SCIP
The SCIP has a stated goal of reducing surgical complications by 25% by 2010.5 To accomplish this, the SCIP is targeting improvement in 4 areas: surgical‐site infection, cardiac events, postoperative pneumonia, and VTE prophylaxis. The SCIP performance measures for VTE prophylaxis in surgical patients are as follows:
-
Recommended VTE prophylaxis ordered during admission; and
-
Appropriate VTE prophylaxis received within 24 hours prior to surgical incision time to 24 hours after surgery end time.
After the success seen by a core group of hospitals who volunteered to participate, all Medicare‐accredited hospitals were required to submit SCIP data beginning with discharges in the first quarter of 2007 to obtain full reimbursement from the Centers for Medicare and Medicaid Services (CMS). Institutions can gauge whether they are in compliance with the SCIP VTE measures by answering a series of yes or no questions about whether prophylaxis has been ordered and received for specific patient groups and procedures. In a recent study, almost one‐half of all surgical patients at risk of VTE did not receive recommended and timely prophylaxis as specified by the SCIP performance measures.6
In addition to the 2 enacted SCIP performance measures for VTE prophylaxis, 2 outcome measures are under development. These measures address the rate at which intraoperative or postoperative pulmonary embolism (PE; SCIP VTE‐3) and deep vein thrombosis (DVT; SCIP VTE‐4) are diagnosed during the index hospitalization and within 30 days after surgery. If implemented, these measures will capture the efficacy of thromboprophylaxis.5
Other VTE Performance Initiatives
Several professional and consumer organizations are developing standards and compiling performance data for public reporting and other purposes:
-
The American Medical Association Physician Consortium for Performance Improvement (PCPI) comprises more than 100 national medical specialty and state medical societies working to identify gaps in care that can be addressed with evidence‐based medicine and formal performance measures. The PCPI has endorsed a measure requiring low‐molecular‐weight heparin (LMWH), low‐dose unfractionated heparin (UFH), adjusted‐dose warfarin, fondaparinux, or mechanical prophylaxis to be given within 24 hours prior to incision time or within 24 hours after surgery end‐time for adults undergoing a procedure for which prophylaxis is indicated.7
-
The Leapfrog Hospital Quality and Safety Survey hosts a searchable web‐based database that consumers can use to compare performance among participating hospitals in specific geographic regions. The Leapfrog survey includes NQF safe practices #28 (reduce occurrence of VTE) and #29 (ensure long‐term anticoagulation is effective and safe).8
-
TJC National Patient Safety Goals (NPSG) target specific improvements in patient safety by providing healthcare organizations with solutions to prevalent patient safety problems. Compliance is necessary for Joint Commission accreditation, and results are reported on the Quality Check website. NPSG Goal 3 is focused on improving the safety of medications, and Goal 3E specifically addresses patient harm associated with the use of anticoagulation therapy. The 2008 NPSG goals must be implemented by January 2009.2
-
The North American Thrombosis Forum (NATF), a nonprofit organization, was recently organized to address unmet needs in North America related to VTE and other thrombotic disorders. It is designed to complement existing organizations dealing with thrombosis‐related issues, with 5 major focus areas: basic translational research; clinical research; prevention and education; public policy; and advocacy. Each month, its website (
http://www.natfonline.org ) features several scientific papers dealing with venous and arterial thrombosis‐related issues. -
The American Venous Forum National Venous Screening Program is a national campaign designed to increase VTE awareness and promote the importance of compliance with prophylaxis protocols.9
As different organizations work to develop performance measures for VTE, conflicting standards have emerged. Although this remains a major challenge, the NQF is attempting to develop voluntary consensus standards that will harmonize VTE performance measures across all sites of care, including the acute medical, surgical, and oncology settings. Major clinical guidelines from the American College of Physicians (ACP), American College of Chest Physicians (ACCP), the American Society of Clinical Oncology (ASCO), the National Comprehensive Cancer Network (NCCN), the European Society of Cardiology (ESC), and other organizations provide data to support standardized, evidence‐based measures for VTE.
Implications of Performance Data
Hospital‐Level Performance Reporting
Performance results may affect an institution's ability to contract best rates with payors, obtain full reimbursement for services, and be eligible for bonus payments. For example, pay‐for‐reporting legislation from CMS provides targeted financial incentives to improve the rates at which hospitals report data on quality measures. The current legislation stipulates that hospitals must submit performance data, including data on compliance with the 2 SCIP‐VTE measures, or lose 2% of their annual CMS payment update. For a 500‐bed hospital with 80% occupancy and 50% CMS patients, failure to report data on SCIP‐VTE measures would result in an estimated annual loss of $2.6 million.10
In 2007, the first year of the CMS pay‐for‐reporting program, 93% of hospitals met the reporting goals. As penalties for nonreporting increase, an even higher compliance rate may be expected. CMS is proposing a new system that would withhold 5% of the base operating diagnosis‐related group payment from a hospital's budget; hospitals would be required to earn this back through reporting and meeting specific performance goals. Using a phase‐in system, CMS would reimburse 2.5% in the first year for pay‐for‐reporting and 2.5% for pay‐for‐performance. Ultimately, the full 5% bonus would be based on performance results.11
Performance ratings play a central role in hospital accreditation, which is critical for negotiating terms for tiered contracting arrangements with private insurers. In addition, hospital performance rankings are becoming more publicly accessible. TJC reports hospital performance in meeting the SCIP measures on its website (
Physician‐Level Performance Reporting
In new quality assessment programs, physicians will also be rewarded or penalized according to their individual performance. The CMS Physician Quality Reporting Initiative (PQRI) is a claims‐based, voluntary, pay‐for‐reporting initiative targeted to Medicare providers. The PQRI program currently pays physicians 2% of total charges for covered services in exchange for voluntary reporting, and it is moving toward results‐based reimbursement.12 The 2009 PQRI Measures List describes 186 quality measures, including 2 related to VTE:13
-
Quality Measure 23: Percentage of patients aged 18 years and older undergoing procedures for which VTE prophylaxis is indicated in all patients, who had an order for LMWH, low‐dose UFH, adjusted‐dose warfarin, fondaparinux, or mechanical prophylaxis to be given within 24 hours prior to incision time or within 24 hours after surgery end‐time; and
-
Quality Measure 31: Percentage of patients aged 18 years and older with a diagnosis of ischemic stroke or intracranial hemorrhage who received DVT prophylaxis by end of hospital day 2.
In the PQRI, physicians report quality measures on process and patient outcomes to CMS using G‐codes or current procedural terminology (CPT)‐II codes. Approximately one‐half of the 100,000 providers who submitted quality codes during the first PQRI reporting period (July 1 to December 31, 2007) qualified for the incentive payment, totaling $36 million.12
More stringent pay‐for‐performance initiatives that hold physicians personally accountable for performance results are being developed in the private sector. For example, the Consumer‐Purchaser Disclosure Project (CPDP) is a consumer‐advocacy group that aims to improve healthcare and lower costs by holding healthcare providers publicly accountable for their quality of treatment. The CPDP has partnered with the National Committee for Quality Assurance to develop guidelines for reporting NQF performance measures.14
VTE as a Nonreimbursable Never Event
In a program that began with hospital discharges on October 1, 2008, hospitals will not receive CMS payment for 12 selected conditions that were not present on admission and were caused by medical error. These hospital‐acquired conditions (HAC), commonly known as never events, include pressure ulcers, catheter‐associated urinary tract infections, postoperative infections, and other complications. Beginning in fiscal year 2009, CMS has added hospital‐acquired VTE following hip or knee replacement surgery as a nonreimbursable never event.15 While CMS acknowledges that prophylaxis will not prevent every occurrence of DVT/PE, they feel it is a reasonably preventable HAC.15 Similar policies are expanding to state and private payor programs that require neither the patient nor the payor to reimburse the hospital for care related to reasonably preventable complications.
Improving Performance and Patient Outcomes
Despite the growing volume of evidence supporting the use of thromboprophylaxis, its use remains inadequate. The consequences are clear: between 2004 and 2006, the number of cases of postoperative VTE increased by 11%.1 This lack of progress may be due to clinicians' lack of awareness of evidence‐based interventions and to hospitals' lack of protocols for the provision of high‐quality preventive treatment.1 Successful strategies for improving thromboprophylaxis and other VTE performance measures are urgently needed. Over the past several years, researchers have been evaluating the utility of different strategies for improving guideline compliance, such as computer‐aided decision‐making and auditing and feedback programs.
Several initiatives seem to have been successful. In one review, Tooher et al.16 found that computerized reminders are, in general, one of the most effective strategies for improving prescribing practice. Paper‐based systems are easier to ignore without a challenge, while electronic systems may force users to acknowledge alerts. Stand‐alone protocols and reminder systems at the point of care can improve prophylaxis rates by about 50%, and decision‐support systems that integrate orders for prophylaxis can increase rates by up to 85%. Importantly, education‐only programs have not been sufficiently effective.16
Regardless of the strategy chosen, Tooher et al identified.16 several general features that, when included as part of the initiative, increase the likelihood of program success:
-
A process for demonstrating the importance and relevance of VTE prophylaxis in the local clinical setting (eg, presenting findings of a local audit of current practice to clinical staff);
-
A process for improving clinician knowledge about VTE risk assessment and prophylaxis practice, such as through a continuing education program;
-
A method of reminding clinicians to assess patients for VTE risk, accompanied by aids to assist in the documentation of patient risk;
-
A process for assisting clinicians in prescribing the appropriate prophylaxis; and
-
A method for assessing the effectiveness of any changes and for refining local policy to further improve practice, such as through clinical audit and feedback.
Table 3 lists several resources and tools that may be useful when designing and implementing strategies to improve performance and quality of care for hospitalized patients at risk of VTE.
| Resource | Description |
|---|---|
| |
| Society of Hospital Medicine, VTE Resource Room* | A website with educational resources, prophylaxis and treatment algorithms, and sample VTE protocols for various patient populations |
| American Society of Clinical Oncology; VTE Prophylaxis Orders and Flow Sheet | A sheet to consult and fill out when prescribing pharmacologic VTE prophylaxis for cancer patients; includes justifications for use, contraindications, anticoagulant options and doses, and other important details |
| American College of Chest Physicians | A source of guidelines, clinical research, education, and other resources for building an evidence‐based VTE protocol |
| National Comprehensive Cancer Network, Clinical Practice GuidelinesVTE | A concise source of algorithms for VTE prophylaxis, diagnosis, and treatment in cancer patients; also includes tables detailing recommended prevention/treatment regimens and warnings/contraindications |
Case Studies in Performance Improvement
Several institutions have reported success stories and shared details of their quality improvement initiatives. Whether paper‐based, electronic, physician‐targeted, or pharmacist‐led, these programs were designed to meet the unique needs of each institution and can serve as models for other hospitals wishing to implement similar programs to improve VTE prophylaxis rates and patient outcomes.
Brigham and Women's Hospital, Boston, MA
In 2005, Kucher et al.17 published a landmark report illustrating the benefits of an electronic alert system in increasing thromboprophylaxis and reducing VTE rates among hospitalized patients. The randomized trial identified high‐risk patients who were not receiving prophylaxis and assigned them to the intervention group, in which the treating physician was alerted to the VTE risk (n = 1255), or to the control group, in which no alert was made (n = 1251). Compared with patients in the control arm, those in the intervention arm were more than twice as likely to receive mechanical or pharmacologic prophylaxis (14.5% vs. 33.5%) and 41% less likely to develop VTE within 90 days (P < 0.001).17
In 2008, this system was evaluated in a new cohort study to determine the ongoing effectiveness of electronic alerts in a real hospital setting.18 The following steps were taken:
-
Alerts were dispatched for all high‐risk cases; and
-
The responsible physician for each high‐risk patient not receiving prophylaxis was issued a single alert detailing the patient's risk and encouraging the use of thromboprophylaxis
During the study period, the use of prophylaxis increased by 50% (P < 0.001). Still, nearly two‐thirds of physicians ignored the electronic alerts.18 Thus, while computer alert systems are helpful, other strategies must be employed to further improve prophylaxis rates in high‐risk medical patients.18
Roswell Park Cancer Institute, Buffalo, NY
Roswell Park Cancer Institute (RPCI), a Comprehensive Cancer Center with 24,000 active patients, initiated an institute‐wide quality improvement initiative in 2006 to improve the rates of VTE prophylaxis for all adult inpatients.19 This initiative included efforts to improve compliance with NCCN guidelines on all medical services and follow guidelines in accordance with NCCN, surgical best practices, and published standards on all surgical services. To accomplish this objective, RPCI:
-
Implemented mandatory, computerized physician order entry forms;
-
Promoted VTE awareness via staff education, field in‐services, and seminars; and
-
Tracked compliance with manual audits of patient charts every 3 months.
When the initiative began in the fourth quarter of 2006, the rate of NCCN‐recommended VTE prophylaxis was 61% with the medical services and 86% with the surgical services. As of the second quarter of 2008, guideline compliance had increased to 90% and 100% with the medical and surgical services, respectively. Accompanying this increase in compliance was a corresponding decrease in the incidence of VTE, from 0.39% in the fourth quarter of 2006 to 0.08% in the second quarter of 2008 (P < 0.0001). The most pronounced reductions in VTE incidence were observed within the medical services and among outpatients.19
Hartford Hospital, Hartford, CT
Hartford Hospital is an 819‐bed acute‐care community hospital with 300 designated medical beds. In an effort to improve thromboprophylaxis rates among medical patients, the pharmacy, medicine, and information technology departments collaborated to develop an alert within the computerized prescriber‐order‐entry system that reminded clinicians to assess patients for VTE risk factors and the need for prophylaxis.20 When a patient met predefined criteria for VTE risk, the message was displayed until either mechanical or pharmacologic VTE prophylaxis was an active order on the patient's treatment profile (Figure 1). The program was implemented in conjunction with an extensive educational program targeting hospital staff, pharmacists, physicians, nurse practitioners, physician assistants, and nurses.20
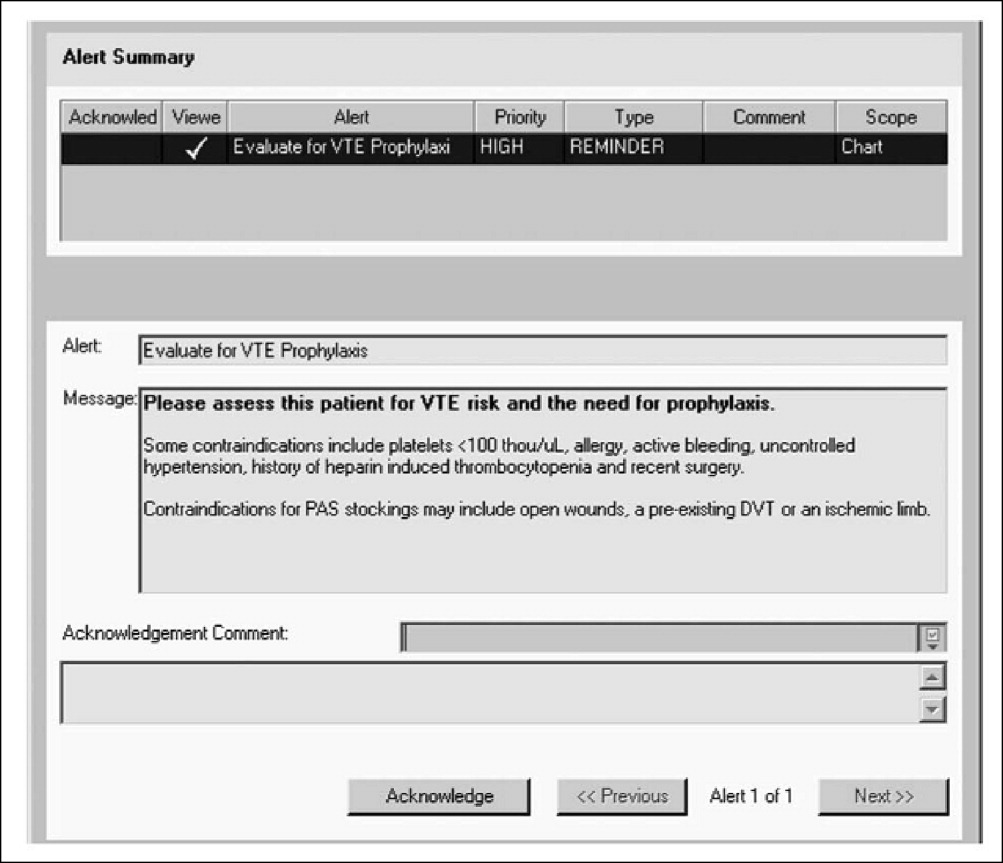
Compliance with institutional prophylaxis guidelines increased from 49% to 93% following implementation (P < 0.001). Interestingly, the initiative at Hartford Hospital was able to increase the use of mechanical prophylaxis among patients with a contradiction to pharmacologic therapy from 25% prior to the program to 100% after its implementation (P < 0.001).20
Saint Elizabeth's Hospital, Collinsville, IL
In 2008, Bauer et al.21 reported the benefits of a pharmacist‐led program for VTE prevention in Saint Elizabeth's Hospital, a 278‐bed hospital with more than 13,000 admissions per year. As part of the initiative, hospital pharmacists:
-
Received daily reports of all new admissions cross‐referenced with an accounting of patients currently prescribed UFH or LMWH;
-
Assessed the remaining patients at risk of VTE; and
-
Placed recommendations in patient charts in the form of a bold sticker alerting the physician to the patient's risk factors, their overall risk of VTE, and treatment recommendations (Figure 2).

The program ran 7 days per week, involved 1 pharmacist per day, and required an average of 4 hours per day. Patients in the maternity, nursery, pediatric, and psychiatric units were excluded from the program.
The program led to a significant increase in the use of VTE prophylaxis and a significant reduction in the rate of DVT (P < 0.002).21 These findings suggest that innovative programs tailored to the needs of individual institutions can dramatically increase thromboprophylaxis rates and decrease the incidence of VTE in at‐risk hospitalized patients.
Conclusions
VTE is a serious disease that leads to excess morbidity and mortality among hospitalized patients. The impact of hospital reporting on reimbursement and patient outcomes necessitates the adoption of strategies and protocols proven to enhance the management of VTE and improve patient outcomes. Several successful VTE initiatives have been described in the literature and can serve as models for institutions wishing to develop policies and procedures for preventing VTE. In addition, a number of online resources exist that can aid in the development of VTE protocols.
- U.S. Department of Health and Human Services. The surgeon general's call to action to prevent deep vein thrombosis and pulmonary embolism. Available at: http://www.surgeongeneral.gov/topics/deepvein/calltoaction/call‐to‐action‐on‐dvt‐2008.pdf. Accessed June2009.
- The Joint Commission. National consensus standards for prevention and care of venous thromboembolism (VTE). Last updated April 2009. Available at: http://www.jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/VTE.htm. Accessed June2009.
- , , , et al.Prevention of venous thromboembolism: American College of Chest Physicians evidence‐based clinical practice guidelines (8th edition).Chest2008;133(6 suppl):381S–453S.
- , , , , , ;American College of Chest Physicians.Antithrombotic therapy for venous thromboembolic disease: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines (8th edition).Chest.2008;133(6 suppl):454S–545S.
- The Joint Commission. Surgical Care Improvement Project Core Measure Set. Updated November 2008. Available at: http://www.jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/SCIP+Core+Measure+Set.htm. Accessed June2009.
- , , , et al.Are surgical patients at risk of venous thromboembolism current meeting the surgical care improvement performance target goal for appropriate and timely prophylaxis?Chest.2008;134:s46003.
- American Medical Association. Perioperative Care: Physician Performance Measurement Set. October 2006. Available at http://www.ama‐assn.org/ama1/pub/upload/mm/370/perioperativews1206.pdf. Accessed June2009.
- Leapfrog Group. The Leapfrog Group Hospital Quality and Safety Survey: What's New in the 2009 Survey (Version 5.1). Available at: https://leapfrog.medstat.com/pdf/final.pdf. Accessed June2009.
- , , , et al.Increasing awareness about venous disease: The American Venous Forum expands the National Venous Screening Program.J Vasc Surg.2008;48(2):394–399.
- Federal Register. Medicare Program; Proposed Changes to the Hospital Inpatient Prospective Payment Systems and Fiscal Year 2007 Rates. Available at: http://edocket.access.gpo.gov/2006/pdf/06‐3629.pdf. Accessed December 10,2008.
- Department of Health and Human Services, Centers for Medicare 241(3):397–415.
- , , , et al.Electronic alerts to prevent venous thromboembolism among hospitalized patients.N Engl J Med.2005;352(10):969–977.
- , , , et al.Electronic alerts for hospitalized high‐VTE risk patients not receiving prophylaxis: a cohort study.J Thromb Thrombolysis.2008;25(2):146–150.
- , , , et al. Improving compliance with guidelines for venous thromboembolism (VTE) prophylaxis significantly reduces VTE events. Presented at the 50th Annual Meeting of the American College of Hematology; San Francisco, CA; December 6‐9, 2008. Abstract 1288.
- .Development and implementation of a program to assess medical patients' need for venous thromboembolism prophylaxis.Am J Health Syst Pharm.2008;65(18):1755–1760.
- , , .Pharmacist‐led program to improve venous thromboembolism prophylaxis in a community hospital.Am J Health Syst Pharm.2008;65(17):1643–1647.
Despite the availability of evidence‐based guidelines for the prevention of thromboembolic morbidity and mortality, venous thromboembolism (VTE) remains a pervasive threat to public health. Prophylaxis is underused for a variety of reasons, which were summarized in the first article of this Supplement. Overcoming these barriers and reducing the incidence of VTE has become a major priority for public health policy.
The Office of the Surgeon General released a report in September 2008 that reflects this sense of urgency and national focus by calling for a coordinated, multifaceted plan to reduce the incidence of VTE in the United States.1 The Surgeon General report is one of the latest in a string of national initiatives designed to improve outcomes in patients at risk of VTE. In the past several years, public and private agencies have launched a range of programs aimed at improving deficiencies in the awareness, prevention, and treatment of VTE in hospitalized patients (these are summarized in Table 1). New performance measures and improvement initiatives may reduce the discrepancies between recommendations and practice, ultimately improving patient outcomes. These measures may possibly become benchmarks for pay‐for‐performance initiatives or future hospital accreditation.
| Measure/Initiative | Description |
|---|---|
| |
| National Quality Forum/The Joint Commission (NQF/TJC) | Public reporting of hospital performance in 6 performance measures; will apply to all medical and surgical patients |
| Surgical Care Improvement Project (SCIP) | Two performance measures enacted with reimbursement implications; 2 outcomes measures |
| American Medical Association Physician Consortium for Performance Improvement (PCPI) | Medical societies collaborating to identify gaps in care and develop performance measures; 1 measure has been endorsed |
| Leapfrog Hospital Quality and Safety Survey | Web database allowing consumers to compare performance among participating hospitals; includes 2 NQF safe practices |
| TJC National Patient Safety Goals (NPSG) | Goals for solving patient safety problems; compliance required for Joint Commission accreditation, with online reporting of results (Quality Check website) |
| North American Thrombosis Forum (NATF) | Nonprofit organization addressing unmet needs related to VTE and other thrombotic disorders |
| American Venous Forum National Venous Screening Program | National VTE awareness campaign; promotes compliance with protocols |
Herein, we review a variety of VTE performance measures, including those from the National Quality Forum (NQF), The Joint Commission (TJC), and the Surgical Care Improvement Project (SCIP). To illustrate how performance measures may be applied in the hospital setting to improve patient care, performance improvement programs that have proven effective in select hospitals across the United States are described.
Performance Measures and Initiatives
National Quality Forum Performance Measures
The NQF and TJC (formerly known as the Joint Commission on Accreditation of Healthcare Organizations) have already enacted performance measures for pneumonia, heart failure, acute myocardial infarction (MI), and other conditions. Since 2005, the NQF and TJC have been collaborating to develop national consensus performance measures for the prevention and care of VTE. The VTE performance measures will apply to all medical and surgical patients and include process measures in the areas of prevention and treatment, as well as outcome measures. After pilot‐testing a range of measures for 3 years, TJC recommended 7 candidate measures in November 2007. In May 2008, the NQF endorsed 6 of these, embracing all TJC recommendations except one relating to the use and documentation of vena cava filter quality improvement (Table 2).2
|
| Risk assessment and prophylaxis |
| 1. Documentation of VTE risk/prophylaxis within 24 hours of hospital admission or surgery end‐time |
| 2. Documentation of VTE risk/prophylaxis within 24 hours after ICU admission, transfer to ICU, or surgery end‐time |
| Treatment |
| 3. Patients with VTE with overlap of parenteral and warfarin anticoagulation therapy for at least 5 days with an INR 2 before discontinuation of parenteral therapy; for > 5 days with an INR < 2 and discharged on overlap therapy; or discharged in < 5 days on overlap therapy |
| 4. Patients with VTE receiving UFH with dosages/platelet count monitoring by protocol or nomogram |
| 5. Patients with VTE or their caregivers are given written discharge instructions or other educational material addressing all of the following: follow‐up monitoring, compliance issues, dietary restrictions, and potential for adverse drug reactions and interactions |
| Outcomes |
| 6. Incidence of potentially preventable hospital‐acquired VTE measured by patients who received no VTE prophylaxis before VTE diagnosis |
The next step is for the NQF to develop a specification manual that defines which patients should be given prophylaxis using International Classification of Diseases, 9th edition (ICD‐9) codes and identifies which interventions are appropriate for each patient population. Current clinical guidelines provide important guidance for appropriate inclusion and exclusion criteria for medical and surgical prophylaxis, as well as evidence‐based recommendations for the treatment of VTE.3, 4
SCIP
The SCIP has a stated goal of reducing surgical complications by 25% by 2010.5 To accomplish this, the SCIP is targeting improvement in 4 areas: surgical‐site infection, cardiac events, postoperative pneumonia, and VTE prophylaxis. The SCIP performance measures for VTE prophylaxis in surgical patients are as follows:
-
Recommended VTE prophylaxis ordered during admission; and
-
Appropriate VTE prophylaxis received within 24 hours prior to surgical incision time to 24 hours after surgery end time.
After the success seen by a core group of hospitals who volunteered to participate, all Medicare‐accredited hospitals were required to submit SCIP data beginning with discharges in the first quarter of 2007 to obtain full reimbursement from the Centers for Medicare and Medicaid Services (CMS). Institutions can gauge whether they are in compliance with the SCIP VTE measures by answering a series of yes or no questions about whether prophylaxis has been ordered and received for specific patient groups and procedures. In a recent study, almost one‐half of all surgical patients at risk of VTE did not receive recommended and timely prophylaxis as specified by the SCIP performance measures.6
In addition to the 2 enacted SCIP performance measures for VTE prophylaxis, 2 outcome measures are under development. These measures address the rate at which intraoperative or postoperative pulmonary embolism (PE; SCIP VTE‐3) and deep vein thrombosis (DVT; SCIP VTE‐4) are diagnosed during the index hospitalization and within 30 days after surgery. If implemented, these measures will capture the efficacy of thromboprophylaxis.5
Other VTE Performance Initiatives
Several professional and consumer organizations are developing standards and compiling performance data for public reporting and other purposes:
-
The American Medical Association Physician Consortium for Performance Improvement (PCPI) comprises more than 100 national medical specialty and state medical societies working to identify gaps in care that can be addressed with evidence‐based medicine and formal performance measures. The PCPI has endorsed a measure requiring low‐molecular‐weight heparin (LMWH), low‐dose unfractionated heparin (UFH), adjusted‐dose warfarin, fondaparinux, or mechanical prophylaxis to be given within 24 hours prior to incision time or within 24 hours after surgery end‐time for adults undergoing a procedure for which prophylaxis is indicated.7
-
The Leapfrog Hospital Quality and Safety Survey hosts a searchable web‐based database that consumers can use to compare performance among participating hospitals in specific geographic regions. The Leapfrog survey includes NQF safe practices #28 (reduce occurrence of VTE) and #29 (ensure long‐term anticoagulation is effective and safe).8
-
TJC National Patient Safety Goals (NPSG) target specific improvements in patient safety by providing healthcare organizations with solutions to prevalent patient safety problems. Compliance is necessary for Joint Commission accreditation, and results are reported on the Quality Check website. NPSG Goal 3 is focused on improving the safety of medications, and Goal 3E specifically addresses patient harm associated with the use of anticoagulation therapy. The 2008 NPSG goals must be implemented by January 2009.2
-
The North American Thrombosis Forum (NATF), a nonprofit organization, was recently organized to address unmet needs in North America related to VTE and other thrombotic disorders. It is designed to complement existing organizations dealing with thrombosis‐related issues, with 5 major focus areas: basic translational research; clinical research; prevention and education; public policy; and advocacy. Each month, its website (
http://www.natfonline.org ) features several scientific papers dealing with venous and arterial thrombosis‐related issues. -
The American Venous Forum National Venous Screening Program is a national campaign designed to increase VTE awareness and promote the importance of compliance with prophylaxis protocols.9
As different organizations work to develop performance measures for VTE, conflicting standards have emerged. Although this remains a major challenge, the NQF is attempting to develop voluntary consensus standards that will harmonize VTE performance measures across all sites of care, including the acute medical, surgical, and oncology settings. Major clinical guidelines from the American College of Physicians (ACP), American College of Chest Physicians (ACCP), the American Society of Clinical Oncology (ASCO), the National Comprehensive Cancer Network (NCCN), the European Society of Cardiology (ESC), and other organizations provide data to support standardized, evidence‐based measures for VTE.
Implications of Performance Data
Hospital‐Level Performance Reporting
Performance results may affect an institution's ability to contract best rates with payors, obtain full reimbursement for services, and be eligible for bonus payments. For example, pay‐for‐reporting legislation from CMS provides targeted financial incentives to improve the rates at which hospitals report data on quality measures. The current legislation stipulates that hospitals must submit performance data, including data on compliance with the 2 SCIP‐VTE measures, or lose 2% of their annual CMS payment update. For a 500‐bed hospital with 80% occupancy and 50% CMS patients, failure to report data on SCIP‐VTE measures would result in an estimated annual loss of $2.6 million.10
In 2007, the first year of the CMS pay‐for‐reporting program, 93% of hospitals met the reporting goals. As penalties for nonreporting increase, an even higher compliance rate may be expected. CMS is proposing a new system that would withhold 5% of the base operating diagnosis‐related group payment from a hospital's budget; hospitals would be required to earn this back through reporting and meeting specific performance goals. Using a phase‐in system, CMS would reimburse 2.5% in the first year for pay‐for‐reporting and 2.5% for pay‐for‐performance. Ultimately, the full 5% bonus would be based on performance results.11
Performance ratings play a central role in hospital accreditation, which is critical for negotiating terms for tiered contracting arrangements with private insurers. In addition, hospital performance rankings are becoming more publicly accessible. TJC reports hospital performance in meeting the SCIP measures on its website (
Physician‐Level Performance Reporting
In new quality assessment programs, physicians will also be rewarded or penalized according to their individual performance. The CMS Physician Quality Reporting Initiative (PQRI) is a claims‐based, voluntary, pay‐for‐reporting initiative targeted to Medicare providers. The PQRI program currently pays physicians 2% of total charges for covered services in exchange for voluntary reporting, and it is moving toward results‐based reimbursement.12 The 2009 PQRI Measures List describes 186 quality measures, including 2 related to VTE:13
-
Quality Measure 23: Percentage of patients aged 18 years and older undergoing procedures for which VTE prophylaxis is indicated in all patients, who had an order for LMWH, low‐dose UFH, adjusted‐dose warfarin, fondaparinux, or mechanical prophylaxis to be given within 24 hours prior to incision time or within 24 hours after surgery end‐time; and
-
Quality Measure 31: Percentage of patients aged 18 years and older with a diagnosis of ischemic stroke or intracranial hemorrhage who received DVT prophylaxis by end of hospital day 2.
In the PQRI, physicians report quality measures on process and patient outcomes to CMS using G‐codes or current procedural terminology (CPT)‐II codes. Approximately one‐half of the 100,000 providers who submitted quality codes during the first PQRI reporting period (July 1 to December 31, 2007) qualified for the incentive payment, totaling $36 million.12
More stringent pay‐for‐performance initiatives that hold physicians personally accountable for performance results are being developed in the private sector. For example, the Consumer‐Purchaser Disclosure Project (CPDP) is a consumer‐advocacy group that aims to improve healthcare and lower costs by holding healthcare providers publicly accountable for their quality of treatment. The CPDP has partnered with the National Committee for Quality Assurance to develop guidelines for reporting NQF performance measures.14
VTE as a Nonreimbursable Never Event
In a program that began with hospital discharges on October 1, 2008, hospitals will not receive CMS payment for 12 selected conditions that were not present on admission and were caused by medical error. These hospital‐acquired conditions (HAC), commonly known as never events, include pressure ulcers, catheter‐associated urinary tract infections, postoperative infections, and other complications. Beginning in fiscal year 2009, CMS has added hospital‐acquired VTE following hip or knee replacement surgery as a nonreimbursable never event.15 While CMS acknowledges that prophylaxis will not prevent every occurrence of DVT/PE, they feel it is a reasonably preventable HAC.15 Similar policies are expanding to state and private payor programs that require neither the patient nor the payor to reimburse the hospital for care related to reasonably preventable complications.
Improving Performance and Patient Outcomes
Despite the growing volume of evidence supporting the use of thromboprophylaxis, its use remains inadequate. The consequences are clear: between 2004 and 2006, the number of cases of postoperative VTE increased by 11%.1 This lack of progress may be due to clinicians' lack of awareness of evidence‐based interventions and to hospitals' lack of protocols for the provision of high‐quality preventive treatment.1 Successful strategies for improving thromboprophylaxis and other VTE performance measures are urgently needed. Over the past several years, researchers have been evaluating the utility of different strategies for improving guideline compliance, such as computer‐aided decision‐making and auditing and feedback programs.
Several initiatives seem to have been successful. In one review, Tooher et al.16 found that computerized reminders are, in general, one of the most effective strategies for improving prescribing practice. Paper‐based systems are easier to ignore without a challenge, while electronic systems may force users to acknowledge alerts. Stand‐alone protocols and reminder systems at the point of care can improve prophylaxis rates by about 50%, and decision‐support systems that integrate orders for prophylaxis can increase rates by up to 85%. Importantly, education‐only programs have not been sufficiently effective.16
Regardless of the strategy chosen, Tooher et al identified.16 several general features that, when included as part of the initiative, increase the likelihood of program success:
-
A process for demonstrating the importance and relevance of VTE prophylaxis in the local clinical setting (eg, presenting findings of a local audit of current practice to clinical staff);
-
A process for improving clinician knowledge about VTE risk assessment and prophylaxis practice, such as through a continuing education program;
-
A method of reminding clinicians to assess patients for VTE risk, accompanied by aids to assist in the documentation of patient risk;
-
A process for assisting clinicians in prescribing the appropriate prophylaxis; and
-
A method for assessing the effectiveness of any changes and for refining local policy to further improve practice, such as through clinical audit and feedback.
Table 3 lists several resources and tools that may be useful when designing and implementing strategies to improve performance and quality of care for hospitalized patients at risk of VTE.
| Resource | Description |
|---|---|
| |
| Society of Hospital Medicine, VTE Resource Room* | A website with educational resources, prophylaxis and treatment algorithms, and sample VTE protocols for various patient populations |
| American Society of Clinical Oncology; VTE Prophylaxis Orders and Flow Sheet | A sheet to consult and fill out when prescribing pharmacologic VTE prophylaxis for cancer patients; includes justifications for use, contraindications, anticoagulant options and doses, and other important details |
| American College of Chest Physicians | A source of guidelines, clinical research, education, and other resources for building an evidence‐based VTE protocol |
| National Comprehensive Cancer Network, Clinical Practice GuidelinesVTE | A concise source of algorithms for VTE prophylaxis, diagnosis, and treatment in cancer patients; also includes tables detailing recommended prevention/treatment regimens and warnings/contraindications |
Case Studies in Performance Improvement
Several institutions have reported success stories and shared details of their quality improvement initiatives. Whether paper‐based, electronic, physician‐targeted, or pharmacist‐led, these programs were designed to meet the unique needs of each institution and can serve as models for other hospitals wishing to implement similar programs to improve VTE prophylaxis rates and patient outcomes.
Brigham and Women's Hospital, Boston, MA
In 2005, Kucher et al.17 published a landmark report illustrating the benefits of an electronic alert system in increasing thromboprophylaxis and reducing VTE rates among hospitalized patients. The randomized trial identified high‐risk patients who were not receiving prophylaxis and assigned them to the intervention group, in which the treating physician was alerted to the VTE risk (n = 1255), or to the control group, in which no alert was made (n = 1251). Compared with patients in the control arm, those in the intervention arm were more than twice as likely to receive mechanical or pharmacologic prophylaxis (14.5% vs. 33.5%) and 41% less likely to develop VTE within 90 days (P < 0.001).17
In 2008, this system was evaluated in a new cohort study to determine the ongoing effectiveness of electronic alerts in a real hospital setting.18 The following steps were taken:
-
Alerts were dispatched for all high‐risk cases; and
-
The responsible physician for each high‐risk patient not receiving prophylaxis was issued a single alert detailing the patient's risk and encouraging the use of thromboprophylaxis
During the study period, the use of prophylaxis increased by 50% (P < 0.001). Still, nearly two‐thirds of physicians ignored the electronic alerts.18 Thus, while computer alert systems are helpful, other strategies must be employed to further improve prophylaxis rates in high‐risk medical patients.18
Roswell Park Cancer Institute, Buffalo, NY
Roswell Park Cancer Institute (RPCI), a Comprehensive Cancer Center with 24,000 active patients, initiated an institute‐wide quality improvement initiative in 2006 to improve the rates of VTE prophylaxis for all adult inpatients.19 This initiative included efforts to improve compliance with NCCN guidelines on all medical services and follow guidelines in accordance with NCCN, surgical best practices, and published standards on all surgical services. To accomplish this objective, RPCI:
-
Implemented mandatory, computerized physician order entry forms;
-
Promoted VTE awareness via staff education, field in‐services, and seminars; and
-
Tracked compliance with manual audits of patient charts every 3 months.
When the initiative began in the fourth quarter of 2006, the rate of NCCN‐recommended VTE prophylaxis was 61% with the medical services and 86% with the surgical services. As of the second quarter of 2008, guideline compliance had increased to 90% and 100% with the medical and surgical services, respectively. Accompanying this increase in compliance was a corresponding decrease in the incidence of VTE, from 0.39% in the fourth quarter of 2006 to 0.08% in the second quarter of 2008 (P < 0.0001). The most pronounced reductions in VTE incidence were observed within the medical services and among outpatients.19
Hartford Hospital, Hartford, CT
Hartford Hospital is an 819‐bed acute‐care community hospital with 300 designated medical beds. In an effort to improve thromboprophylaxis rates among medical patients, the pharmacy, medicine, and information technology departments collaborated to develop an alert within the computerized prescriber‐order‐entry system that reminded clinicians to assess patients for VTE risk factors and the need for prophylaxis.20 When a patient met predefined criteria for VTE risk, the message was displayed until either mechanical or pharmacologic VTE prophylaxis was an active order on the patient's treatment profile (Figure 1). The program was implemented in conjunction with an extensive educational program targeting hospital staff, pharmacists, physicians, nurse practitioners, physician assistants, and nurses.20

Compliance with institutional prophylaxis guidelines increased from 49% to 93% following implementation (P < 0.001). Interestingly, the initiative at Hartford Hospital was able to increase the use of mechanical prophylaxis among patients with a contradiction to pharmacologic therapy from 25% prior to the program to 100% after its implementation (P < 0.001).20
Saint Elizabeth's Hospital, Collinsville, IL
In 2008, Bauer et al.21 reported the benefits of a pharmacist‐led program for VTE prevention in Saint Elizabeth's Hospital, a 278‐bed hospital with more than 13,000 admissions per year. As part of the initiative, hospital pharmacists:
-
Received daily reports of all new admissions cross‐referenced with an accounting of patients currently prescribed UFH or LMWH;
-
Assessed the remaining patients at risk of VTE; and
-
Placed recommendations in patient charts in the form of a bold sticker alerting the physician to the patient's risk factors, their overall risk of VTE, and treatment recommendations (Figure 2).

The program ran 7 days per week, involved 1 pharmacist per day, and required an average of 4 hours per day. Patients in the maternity, nursery, pediatric, and psychiatric units were excluded from the program.
The program led to a significant increase in the use of VTE prophylaxis and a significant reduction in the rate of DVT (P < 0.002).21 These findings suggest that innovative programs tailored to the needs of individual institutions can dramatically increase thromboprophylaxis rates and decrease the incidence of VTE in at‐risk hospitalized patients.
Conclusions
VTE is a serious disease that leads to excess morbidity and mortality among hospitalized patients. The impact of hospital reporting on reimbursement and patient outcomes necessitates the adoption of strategies and protocols proven to enhance the management of VTE and improve patient outcomes. Several successful VTE initiatives have been described in the literature and can serve as models for institutions wishing to develop policies and procedures for preventing VTE. In addition, a number of online resources exist that can aid in the development of VTE protocols.
Despite the availability of evidence‐based guidelines for the prevention of thromboembolic morbidity and mortality, venous thromboembolism (VTE) remains a pervasive threat to public health. Prophylaxis is underused for a variety of reasons, which were summarized in the first article of this Supplement. Overcoming these barriers and reducing the incidence of VTE has become a major priority for public health policy.
The Office of the Surgeon General released a report in September 2008 that reflects this sense of urgency and national focus by calling for a coordinated, multifaceted plan to reduce the incidence of VTE in the United States.1 The Surgeon General report is one of the latest in a string of national initiatives designed to improve outcomes in patients at risk of VTE. In the past several years, public and private agencies have launched a range of programs aimed at improving deficiencies in the awareness, prevention, and treatment of VTE in hospitalized patients (these are summarized in Table 1). New performance measures and improvement initiatives may reduce the discrepancies between recommendations and practice, ultimately improving patient outcomes. These measures may possibly become benchmarks for pay‐for‐performance initiatives or future hospital accreditation.
| Measure/Initiative | Description |
|---|---|
| |
| National Quality Forum/The Joint Commission (NQF/TJC) | Public reporting of hospital performance in 6 performance measures; will apply to all medical and surgical patients |
| Surgical Care Improvement Project (SCIP) | Two performance measures enacted with reimbursement implications; 2 outcomes measures |
| American Medical Association Physician Consortium for Performance Improvement (PCPI) | Medical societies collaborating to identify gaps in care and develop performance measures; 1 measure has been endorsed |
| Leapfrog Hospital Quality and Safety Survey | Web database allowing consumers to compare performance among participating hospitals; includes 2 NQF safe practices |
| TJC National Patient Safety Goals (NPSG) | Goals for solving patient safety problems; compliance required for Joint Commission accreditation, with online reporting of results (Quality Check website) |
| North American Thrombosis Forum (NATF) | Nonprofit organization addressing unmet needs related to VTE and other thrombotic disorders |
| American Venous Forum National Venous Screening Program | National VTE awareness campaign; promotes compliance with protocols |
Herein, we review a variety of VTE performance measures, including those from the National Quality Forum (NQF), The Joint Commission (TJC), and the Surgical Care Improvement Project (SCIP). To illustrate how performance measures may be applied in the hospital setting to improve patient care, performance improvement programs that have proven effective in select hospitals across the United States are described.
Performance Measures and Initiatives
National Quality Forum Performance Measures
The NQF and TJC (formerly known as the Joint Commission on Accreditation of Healthcare Organizations) have already enacted performance measures for pneumonia, heart failure, acute myocardial infarction (MI), and other conditions. Since 2005, the NQF and TJC have been collaborating to develop national consensus performance measures for the prevention and care of VTE. The VTE performance measures will apply to all medical and surgical patients and include process measures in the areas of prevention and treatment, as well as outcome measures. After pilot‐testing a range of measures for 3 years, TJC recommended 7 candidate measures in November 2007. In May 2008, the NQF endorsed 6 of these, embracing all TJC recommendations except one relating to the use and documentation of vena cava filter quality improvement (Table 2).2
|
| Risk assessment and prophylaxis |
| 1. Documentation of VTE risk/prophylaxis within 24 hours of hospital admission or surgery end‐time |
| 2. Documentation of VTE risk/prophylaxis within 24 hours after ICU admission, transfer to ICU, or surgery end‐time |
| Treatment |
| 3. Patients with VTE with overlap of parenteral and warfarin anticoagulation therapy for at least 5 days with an INR 2 before discontinuation of parenteral therapy; for > 5 days with an INR < 2 and discharged on overlap therapy; or discharged in < 5 days on overlap therapy |
| 4. Patients with VTE receiving UFH with dosages/platelet count monitoring by protocol or nomogram |
| 5. Patients with VTE or their caregivers are given written discharge instructions or other educational material addressing all of the following: follow‐up monitoring, compliance issues, dietary restrictions, and potential for adverse drug reactions and interactions |
| Outcomes |
| 6. Incidence of potentially preventable hospital‐acquired VTE measured by patients who received no VTE prophylaxis before VTE diagnosis |
The next step is for the NQF to develop a specification manual that defines which patients should be given prophylaxis using International Classification of Diseases, 9th edition (ICD‐9) codes and identifies which interventions are appropriate for each patient population. Current clinical guidelines provide important guidance for appropriate inclusion and exclusion criteria for medical and surgical prophylaxis, as well as evidence‐based recommendations for the treatment of VTE.3, 4
SCIP
The SCIP has a stated goal of reducing surgical complications by 25% by 2010.5 To accomplish this, the SCIP is targeting improvement in 4 areas: surgical‐site infection, cardiac events, postoperative pneumonia, and VTE prophylaxis. The SCIP performance measures for VTE prophylaxis in surgical patients are as follows:
-
Recommended VTE prophylaxis ordered during admission; and
-
Appropriate VTE prophylaxis received within 24 hours prior to surgical incision time to 24 hours after surgery end time.
After the success seen by a core group of hospitals who volunteered to participate, all Medicare‐accredited hospitals were required to submit SCIP data beginning with discharges in the first quarter of 2007 to obtain full reimbursement from the Centers for Medicare and Medicaid Services (CMS). Institutions can gauge whether they are in compliance with the SCIP VTE measures by answering a series of yes or no questions about whether prophylaxis has been ordered and received for specific patient groups and procedures. In a recent study, almost one‐half of all surgical patients at risk of VTE did not receive recommended and timely prophylaxis as specified by the SCIP performance measures.6
In addition to the 2 enacted SCIP performance measures for VTE prophylaxis, 2 outcome measures are under development. These measures address the rate at which intraoperative or postoperative pulmonary embolism (PE; SCIP VTE‐3) and deep vein thrombosis (DVT; SCIP VTE‐4) are diagnosed during the index hospitalization and within 30 days after surgery. If implemented, these measures will capture the efficacy of thromboprophylaxis.5
Other VTE Performance Initiatives
Several professional and consumer organizations are developing standards and compiling performance data for public reporting and other purposes:
-
The American Medical Association Physician Consortium for Performance Improvement (PCPI) comprises more than 100 national medical specialty and state medical societies working to identify gaps in care that can be addressed with evidence‐based medicine and formal performance measures. The PCPI has endorsed a measure requiring low‐molecular‐weight heparin (LMWH), low‐dose unfractionated heparin (UFH), adjusted‐dose warfarin, fondaparinux, or mechanical prophylaxis to be given within 24 hours prior to incision time or within 24 hours after surgery end‐time for adults undergoing a procedure for which prophylaxis is indicated.7
-
The Leapfrog Hospital Quality and Safety Survey hosts a searchable web‐based database that consumers can use to compare performance among participating hospitals in specific geographic regions. The Leapfrog survey includes NQF safe practices #28 (reduce occurrence of VTE) and #29 (ensure long‐term anticoagulation is effective and safe).8
-
TJC National Patient Safety Goals (NPSG) target specific improvements in patient safety by providing healthcare organizations with solutions to prevalent patient safety problems. Compliance is necessary for Joint Commission accreditation, and results are reported on the Quality Check website. NPSG Goal 3 is focused on improving the safety of medications, and Goal 3E specifically addresses patient harm associated with the use of anticoagulation therapy. The 2008 NPSG goals must be implemented by January 2009.2
-
The North American Thrombosis Forum (NATF), a nonprofit organization, was recently organized to address unmet needs in North America related to VTE and other thrombotic disorders. It is designed to complement existing organizations dealing with thrombosis‐related issues, with 5 major focus areas: basic translational research; clinical research; prevention and education; public policy; and advocacy. Each month, its website (
http://www.natfonline.org ) features several scientific papers dealing with venous and arterial thrombosis‐related issues. -
The American Venous Forum National Venous Screening Program is a national campaign designed to increase VTE awareness and promote the importance of compliance with prophylaxis protocols.9
As different organizations work to develop performance measures for VTE, conflicting standards have emerged. Although this remains a major challenge, the NQF is attempting to develop voluntary consensus standards that will harmonize VTE performance measures across all sites of care, including the acute medical, surgical, and oncology settings. Major clinical guidelines from the American College of Physicians (ACP), American College of Chest Physicians (ACCP), the American Society of Clinical Oncology (ASCO), the National Comprehensive Cancer Network (NCCN), the European Society of Cardiology (ESC), and other organizations provide data to support standardized, evidence‐based measures for VTE.
Implications of Performance Data
Hospital‐Level Performance Reporting
Performance results may affect an institution's ability to contract best rates with payors, obtain full reimbursement for services, and be eligible for bonus payments. For example, pay‐for‐reporting legislation from CMS provides targeted financial incentives to improve the rates at which hospitals report data on quality measures. The current legislation stipulates that hospitals must submit performance data, including data on compliance with the 2 SCIP‐VTE measures, or lose 2% of their annual CMS payment update. For a 500‐bed hospital with 80% occupancy and 50% CMS patients, failure to report data on SCIP‐VTE measures would result in an estimated annual loss of $2.6 million.10
In 2007, the first year of the CMS pay‐for‐reporting program, 93% of hospitals met the reporting goals. As penalties for nonreporting increase, an even higher compliance rate may be expected. CMS is proposing a new system that would withhold 5% of the base operating diagnosis‐related group payment from a hospital's budget; hospitals would be required to earn this back through reporting and meeting specific performance goals. Using a phase‐in system, CMS would reimburse 2.5% in the first year for pay‐for‐reporting and 2.5% for pay‐for‐performance. Ultimately, the full 5% bonus would be based on performance results.11
Performance ratings play a central role in hospital accreditation, which is critical for negotiating terms for tiered contracting arrangements with private insurers. In addition, hospital performance rankings are becoming more publicly accessible. TJC reports hospital performance in meeting the SCIP measures on its website (
Physician‐Level Performance Reporting
In new quality assessment programs, physicians will also be rewarded or penalized according to their individual performance. The CMS Physician Quality Reporting Initiative (PQRI) is a claims‐based, voluntary, pay‐for‐reporting initiative targeted to Medicare providers. The PQRI program currently pays physicians 2% of total charges for covered services in exchange for voluntary reporting, and it is moving toward results‐based reimbursement.12 The 2009 PQRI Measures List describes 186 quality measures, including 2 related to VTE:13
-
Quality Measure 23: Percentage of patients aged 18 years and older undergoing procedures for which VTE prophylaxis is indicated in all patients, who had an order for LMWH, low‐dose UFH, adjusted‐dose warfarin, fondaparinux, or mechanical prophylaxis to be given within 24 hours prior to incision time or within 24 hours after surgery end‐time; and
-
Quality Measure 31: Percentage of patients aged 18 years and older with a diagnosis of ischemic stroke or intracranial hemorrhage who received DVT prophylaxis by end of hospital day 2.
In the PQRI, physicians report quality measures on process and patient outcomes to CMS using G‐codes or current procedural terminology (CPT)‐II codes. Approximately one‐half of the 100,000 providers who submitted quality codes during the first PQRI reporting period (July 1 to December 31, 2007) qualified for the incentive payment, totaling $36 million.12
More stringent pay‐for‐performance initiatives that hold physicians personally accountable for performance results are being developed in the private sector. For example, the Consumer‐Purchaser Disclosure Project (CPDP) is a consumer‐advocacy group that aims to improve healthcare and lower costs by holding healthcare providers publicly accountable for their quality of treatment. The CPDP has partnered with the National Committee for Quality Assurance to develop guidelines for reporting NQF performance measures.14
VTE as a Nonreimbursable Never Event
In a program that began with hospital discharges on October 1, 2008, hospitals will not receive CMS payment for 12 selected conditions that were not present on admission and were caused by medical error. These hospital‐acquired conditions (HAC), commonly known as never events, include pressure ulcers, catheter‐associated urinary tract infections, postoperative infections, and other complications. Beginning in fiscal year 2009, CMS has added hospital‐acquired VTE following hip or knee replacement surgery as a nonreimbursable never event.15 While CMS acknowledges that prophylaxis will not prevent every occurrence of DVT/PE, they feel it is a reasonably preventable HAC.15 Similar policies are expanding to state and private payor programs that require neither the patient nor the payor to reimburse the hospital for care related to reasonably preventable complications.
Improving Performance and Patient Outcomes
Despite the growing volume of evidence supporting the use of thromboprophylaxis, its use remains inadequate. The consequences are clear: between 2004 and 2006, the number of cases of postoperative VTE increased by 11%.1 This lack of progress may be due to clinicians' lack of awareness of evidence‐based interventions and to hospitals' lack of protocols for the provision of high‐quality preventive treatment.1 Successful strategies for improving thromboprophylaxis and other VTE performance measures are urgently needed. Over the past several years, researchers have been evaluating the utility of different strategies for improving guideline compliance, such as computer‐aided decision‐making and auditing and feedback programs.
Several initiatives seem to have been successful. In one review, Tooher et al.16 found that computerized reminders are, in general, one of the most effective strategies for improving prescribing practice. Paper‐based systems are easier to ignore without a challenge, while electronic systems may force users to acknowledge alerts. Stand‐alone protocols and reminder systems at the point of care can improve prophylaxis rates by about 50%, and decision‐support systems that integrate orders for prophylaxis can increase rates by up to 85%. Importantly, education‐only programs have not been sufficiently effective.16
Regardless of the strategy chosen, Tooher et al identified.16 several general features that, when included as part of the initiative, increase the likelihood of program success:
-
A process for demonstrating the importance and relevance of VTE prophylaxis in the local clinical setting (eg, presenting findings of a local audit of current practice to clinical staff);
-
A process for improving clinician knowledge about VTE risk assessment and prophylaxis practice, such as through a continuing education program;
-
A method of reminding clinicians to assess patients for VTE risk, accompanied by aids to assist in the documentation of patient risk;
-
A process for assisting clinicians in prescribing the appropriate prophylaxis; and
-
A method for assessing the effectiveness of any changes and for refining local policy to further improve practice, such as through clinical audit and feedback.
Table 3 lists several resources and tools that may be useful when designing and implementing strategies to improve performance and quality of care for hospitalized patients at risk of VTE.
| Resource | Description |
|---|---|
| |
| Society of Hospital Medicine, VTE Resource Room* | A website with educational resources, prophylaxis and treatment algorithms, and sample VTE protocols for various patient populations |
| American Society of Clinical Oncology; VTE Prophylaxis Orders and Flow Sheet | A sheet to consult and fill out when prescribing pharmacologic VTE prophylaxis for cancer patients; includes justifications for use, contraindications, anticoagulant options and doses, and other important details |
| American College of Chest Physicians | A source of guidelines, clinical research, education, and other resources for building an evidence‐based VTE protocol |
| National Comprehensive Cancer Network, Clinical Practice GuidelinesVTE | A concise source of algorithms for VTE prophylaxis, diagnosis, and treatment in cancer patients; also includes tables detailing recommended prevention/treatment regimens and warnings/contraindications |
Case Studies in Performance Improvement
Several institutions have reported success stories and shared details of their quality improvement initiatives. Whether paper‐based, electronic, physician‐targeted, or pharmacist‐led, these programs were designed to meet the unique needs of each institution and can serve as models for other hospitals wishing to implement similar programs to improve VTE prophylaxis rates and patient outcomes.
Brigham and Women's Hospital, Boston, MA
In 2005, Kucher et al.17 published a landmark report illustrating the benefits of an electronic alert system in increasing thromboprophylaxis and reducing VTE rates among hospitalized patients. The randomized trial identified high‐risk patients who were not receiving prophylaxis and assigned them to the intervention group, in which the treating physician was alerted to the VTE risk (n = 1255), or to the control group, in which no alert was made (n = 1251). Compared with patients in the control arm, those in the intervention arm were more than twice as likely to receive mechanical or pharmacologic prophylaxis (14.5% vs. 33.5%) and 41% less likely to develop VTE within 90 days (P < 0.001).17
In 2008, this system was evaluated in a new cohort study to determine the ongoing effectiveness of electronic alerts in a real hospital setting.18 The following steps were taken:
-
Alerts were dispatched for all high‐risk cases; and
-
The responsible physician for each high‐risk patient not receiving prophylaxis was issued a single alert detailing the patient's risk and encouraging the use of thromboprophylaxis
During the study period, the use of prophylaxis increased by 50% (P < 0.001). Still, nearly two‐thirds of physicians ignored the electronic alerts.18 Thus, while computer alert systems are helpful, other strategies must be employed to further improve prophylaxis rates in high‐risk medical patients.18
Roswell Park Cancer Institute, Buffalo, NY
Roswell Park Cancer Institute (RPCI), a Comprehensive Cancer Center with 24,000 active patients, initiated an institute‐wide quality improvement initiative in 2006 to improve the rates of VTE prophylaxis for all adult inpatients.19 This initiative included efforts to improve compliance with NCCN guidelines on all medical services and follow guidelines in accordance with NCCN, surgical best practices, and published standards on all surgical services. To accomplish this objective, RPCI:
-
Implemented mandatory, computerized physician order entry forms;
-
Promoted VTE awareness via staff education, field in‐services, and seminars; and
-
Tracked compliance with manual audits of patient charts every 3 months.
When the initiative began in the fourth quarter of 2006, the rate of NCCN‐recommended VTE prophylaxis was 61% with the medical services and 86% with the surgical services. As of the second quarter of 2008, guideline compliance had increased to 90% and 100% with the medical and surgical services, respectively. Accompanying this increase in compliance was a corresponding decrease in the incidence of VTE, from 0.39% in the fourth quarter of 2006 to 0.08% in the second quarter of 2008 (P < 0.0001). The most pronounced reductions in VTE incidence were observed within the medical services and among outpatients.19
Hartford Hospital, Hartford, CT
Hartford Hospital is an 819‐bed acute‐care community hospital with 300 designated medical beds. In an effort to improve thromboprophylaxis rates among medical patients, the pharmacy, medicine, and information technology departments collaborated to develop an alert within the computerized prescriber‐order‐entry system that reminded clinicians to assess patients for VTE risk factors and the need for prophylaxis.20 When a patient met predefined criteria for VTE risk, the message was displayed until either mechanical or pharmacologic VTE prophylaxis was an active order on the patient's treatment profile (Figure 1). The program was implemented in conjunction with an extensive educational program targeting hospital staff, pharmacists, physicians, nurse practitioners, physician assistants, and nurses.20

Compliance with institutional prophylaxis guidelines increased from 49% to 93% following implementation (P < 0.001). Interestingly, the initiative at Hartford Hospital was able to increase the use of mechanical prophylaxis among patients with a contradiction to pharmacologic therapy from 25% prior to the program to 100% after its implementation (P < 0.001).20
Saint Elizabeth's Hospital, Collinsville, IL
In 2008, Bauer et al.21 reported the benefits of a pharmacist‐led program for VTE prevention in Saint Elizabeth's Hospital, a 278‐bed hospital with more than 13,000 admissions per year. As part of the initiative, hospital pharmacists:
-
Received daily reports of all new admissions cross‐referenced with an accounting of patients currently prescribed UFH or LMWH;
-
Assessed the remaining patients at risk of VTE; and
-
Placed recommendations in patient charts in the form of a bold sticker alerting the physician to the patient's risk factors, their overall risk of VTE, and treatment recommendations (Figure 2).

The program ran 7 days per week, involved 1 pharmacist per day, and required an average of 4 hours per day. Patients in the maternity, nursery, pediatric, and psychiatric units were excluded from the program.
The program led to a significant increase in the use of VTE prophylaxis and a significant reduction in the rate of DVT (P < 0.002).21 These findings suggest that innovative programs tailored to the needs of individual institutions can dramatically increase thromboprophylaxis rates and decrease the incidence of VTE in at‐risk hospitalized patients.
Conclusions
VTE is a serious disease that leads to excess morbidity and mortality among hospitalized patients. The impact of hospital reporting on reimbursement and patient outcomes necessitates the adoption of strategies and protocols proven to enhance the management of VTE and improve patient outcomes. Several successful VTE initiatives have been described in the literature and can serve as models for institutions wishing to develop policies and procedures for preventing VTE. In addition, a number of online resources exist that can aid in the development of VTE protocols.
- U.S. Department of Health and Human Services. The surgeon general's call to action to prevent deep vein thrombosis and pulmonary embolism. Available at: http://www.surgeongeneral.gov/topics/deepvein/calltoaction/call‐to‐action‐on‐dvt‐2008.pdf. Accessed June2009.
- The Joint Commission. National consensus standards for prevention and care of venous thromboembolism (VTE). Last updated April 2009. Available at: http://www.jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/VTE.htm. Accessed June2009.
- , , , et al.Prevention of venous thromboembolism: American College of Chest Physicians evidence‐based clinical practice guidelines (8th edition).Chest2008;133(6 suppl):381S–453S.
- , , , , , ;American College of Chest Physicians.Antithrombotic therapy for venous thromboembolic disease: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines (8th edition).Chest.2008;133(6 suppl):454S–545S.
- The Joint Commission. Surgical Care Improvement Project Core Measure Set. Updated November 2008. Available at: http://www.jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/SCIP+Core+Measure+Set.htm. Accessed June2009.
- , , , et al.Are surgical patients at risk of venous thromboembolism current meeting the surgical care improvement performance target goal for appropriate and timely prophylaxis?Chest.2008;134:s46003.
- American Medical Association. Perioperative Care: Physician Performance Measurement Set. October 2006. Available at http://www.ama‐assn.org/ama1/pub/upload/mm/370/perioperativews1206.pdf. Accessed June2009.
- Leapfrog Group. The Leapfrog Group Hospital Quality and Safety Survey: What's New in the 2009 Survey (Version 5.1). Available at: https://leapfrog.medstat.com/pdf/final.pdf. Accessed June2009.
- , , , et al.Increasing awareness about venous disease: The American Venous Forum expands the National Venous Screening Program.J Vasc Surg.2008;48(2):394–399.
- Federal Register. Medicare Program; Proposed Changes to the Hospital Inpatient Prospective Payment Systems and Fiscal Year 2007 Rates. Available at: http://edocket.access.gpo.gov/2006/pdf/06‐3629.pdf. Accessed December 10,2008.
- Department of Health and Human Services, Centers for Medicare 241(3):397–415.
- , , , et al.Electronic alerts to prevent venous thromboembolism among hospitalized patients.N Engl J Med.2005;352(10):969–977.
- , , , et al.Electronic alerts for hospitalized high‐VTE risk patients not receiving prophylaxis: a cohort study.J Thromb Thrombolysis.2008;25(2):146–150.
- , , , et al. Improving compliance with guidelines for venous thromboembolism (VTE) prophylaxis significantly reduces VTE events. Presented at the 50th Annual Meeting of the American College of Hematology; San Francisco, CA; December 6‐9, 2008. Abstract 1288.
- .Development and implementation of a program to assess medical patients' need for venous thromboembolism prophylaxis.Am J Health Syst Pharm.2008;65(18):1755–1760.
- , , .Pharmacist‐led program to improve venous thromboembolism prophylaxis in a community hospital.Am J Health Syst Pharm.2008;65(17):1643–1647.
- U.S. Department of Health and Human Services. The surgeon general's call to action to prevent deep vein thrombosis and pulmonary embolism. Available at: http://www.surgeongeneral.gov/topics/deepvein/calltoaction/call‐to‐action‐on‐dvt‐2008.pdf. Accessed June2009.
- The Joint Commission. National consensus standards for prevention and care of venous thromboembolism (VTE). Last updated April 2009. Available at: http://www.jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/VTE.htm. Accessed June2009.
- , , , et al.Prevention of venous thromboembolism: American College of Chest Physicians evidence‐based clinical practice guidelines (8th edition).Chest2008;133(6 suppl):381S–453S.
- , , , , , ;American College of Chest Physicians.Antithrombotic therapy for venous thromboembolic disease: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines (8th edition).Chest.2008;133(6 suppl):454S–545S.
- The Joint Commission. Surgical Care Improvement Project Core Measure Set. Updated November 2008. Available at: http://www.jointcommission.org/PerformanceMeasurement/PerformanceMeasurement/SCIP+Core+Measure+Set.htm. Accessed June2009.
- , , , et al.Are surgical patients at risk of venous thromboembolism current meeting the surgical care improvement performance target goal for appropriate and timely prophylaxis?Chest.2008;134:s46003.
- American Medical Association. Perioperative Care: Physician Performance Measurement Set. October 2006. Available at http://www.ama‐assn.org/ama1/pub/upload/mm/370/perioperativews1206.pdf. Accessed June2009.
- Leapfrog Group. The Leapfrog Group Hospital Quality and Safety Survey: What's New in the 2009 Survey (Version 5.1). Available at: https://leapfrog.medstat.com/pdf/final.pdf. Accessed June2009.
- , , , et al.Increasing awareness about venous disease: The American Venous Forum expands the National Venous Screening Program.J Vasc Surg.2008;48(2):394–399.
- Federal Register. Medicare Program; Proposed Changes to the Hospital Inpatient Prospective Payment Systems and Fiscal Year 2007 Rates. Available at: http://edocket.access.gpo.gov/2006/pdf/06‐3629.pdf. Accessed December 10,2008.
- Department of Health and Human Services, Centers for Medicare 241(3):397–415.
- , , , et al.Electronic alerts to prevent venous thromboembolism among hospitalized patients.N Engl J Med.2005;352(10):969–977.
- , , , et al.Electronic alerts for hospitalized high‐VTE risk patients not receiving prophylaxis: a cohort study.J Thromb Thrombolysis.2008;25(2):146–150.
- , , , et al. Improving compliance with guidelines for venous thromboembolism (VTE) prophylaxis significantly reduces VTE events. Presented at the 50th Annual Meeting of the American College of Hematology; San Francisco, CA; December 6‐9, 2008. Abstract 1288.
- .Development and implementation of a program to assess medical patients' need for venous thromboembolism prophylaxis.Am J Health Syst Pharm.2008;65(18):1755–1760.
- , , .Pharmacist‐led program to improve venous thromboembolism prophylaxis in a community hospital.Am J Health Syst Pharm.2008;65(17):1643–1647.
VTE Risk Factors and Barriers
Venous thromboembolism (VTE) is a common and potentially devastating complication of medical illness and surgical intervention. Among patients discharged from acute‐care hospitals in 2003, more than 12 million (31%) had a moderate or high risk of VTE during hospitalization, including 11% at risk due to surgical procedures and 20% at risk due to medical illnesses.1 The incidence of VTEwhich can present as deep vein thrombosis (DVT) or pulmonary embolism (PE)is rising in hospitalized patients.2 Despite the availability of effective prophylaxis, VTE is the third most common cause of hospital‐related death and the most common preventable cause of hospital mortality.3
The clinical impact of VTE is significant. While total incidence, prevalence, and mortality rates of VTE are elusive, the annual incidence of DVT is thought to be as high as 2 million.4 The most serious complication of DVT is acute PE, which occurs in approximately 600,000 patients per year, one‐third of whom die.5 DVT may also be complicated by recurrent episodes of VTE and postthrombotic sequelae such as chronic venous stasis, venous ulceration, debilitating pain, and intractable edema.6 One prospective cohort study found that 30% of patients who had experienced a first episode of DVT developed recurrent VTE within 8 years, and the incidence of postthrombotic syndrome (PTS) was 23% after just 2 years.6
The economic burden of VTE is substantial, due both to the initial event and to the high rate of hospital readmission. The estimated average cost of DVT management (including initial acute care and 6 months of follow‐up care) is $10,072 per patient, and the corresponding cost for PE is $14,649.7 Hospital readmission occurs in 5% to 14% of patients, more than one‐half of whom are readmitted within 90 days.8 For patients with recurrent DVT or PE, the mean total hospitalization costs of readmission are $11,862 and $14,722, respectively.8
The clinical and economic burden of VTE can be significantly mitigated by the use of effective prophylaxis. Because VTE is difficult to diagnose antemortem, it is easier and safer to prevent with appropriate prophylaxis than to diagnose after it has occurred. Unfortunately, VTE prophylaxis is markedly underused, particularly among high‐risk, hospitalized medical patients who would most benefit from it.9
This article summarizes specific risk factors for VTE and provides guidance in identifying patients who may require thromboprophylaxis. Barriers to optimal VTE prophylaxis in the hospital setting will also be explored.
Methodology
For this article and the ones that follow, relevant literature was identified through a Medline search (January 1980 to December 2008) using the following search terms: venous thromboembolism, pulmonary embolism, deep vein thrombosis, epidemiology, risk factors, prophylaxis, mechanical prophylaxis, diagnosis, treatment, anticoagulants, monitoring, secondary prevention, guideline, adherence, treatment protocol, performance measure, and quality improvement. The bibliographies of all key texts were searched for additional relevant articles. The websites of the American College of Chest Physicians (ACCP), American Society of Clinical Oncology (ASCO), National Comprehensive Cancer Network (NCCN), and Society of Hospital Medicine (SHM) were also searched for annual meeting abstracts, position statements, and other key publications.
Evidence‐based clinical guidelines were identified through a search of the National Guideline Clearinghouse (
Pathogenesis of VTE
Venous thrombosis occurs as a result of at least 1 of 3 underlying factors: alterations in blood flow, vascular endothelial injury, and alterations in the constitution of the blood.10 Each potential underlying factor encompasses a wide range of risk factors and clinical scenarios. Alterations in venous blood flow can include several situations, including venous stasis, venous hypertension, and valvular incompetence. Endothelial injury can arise from shear stress, direct trauma, infection, hypertension, or other sources of endothelial damage. Hypercoagulability from alterations in the constitution of the blood may be due to antithrombin deficiency, cancer, surgery, pregnancy, or other risk factors. The presence of any of these factors indicates an elevated risk of VTE, and the presence of multiple factors further increases risk.10
Risk Factors for VTE
VTE can occur in a wide variety of clinical circumstances. Recognized risk factors for VTE include hospitalization for an acute medical illness, cardiovascular disease, pulmonary disease, major surgery, multiple trauma, obesity, and increasing age.10 Additional factors that place patients at increased risk of VTE (independent of age) include a history of prior VTE, known hypercoagulable states, active cancer, and acute infection.11 Hospital‐acquired risk factors such as immobility, acute illness, or medical interventions may lead to the development of VTE in these patients. Severity of illness must be factored into the risk assessment, and all patients need to be assessed for VTE risk at the time of hospital admission and daily thereafter if pharmacologic therapy is not initiated.
In a review of 1231 consecutive patients treated for acute DVT and/or PE, 96.3% had at least 1 risk factor for VTE, and more than one‐third (39%) had 3 or more risk factors (Table 1).10 The incidence of VTE in hospitalized patients is directly related to the number of risk factors present:10
-
1 risk factor: 11%
-
2 risk factors: 24%
-
3 risk factors: 36%
-
4 risk factors: 50%
-
5 risk factors: > 90%
| Risk Factor | Patients (%) |
|---|---|
| |
| Age 40 years | 88.5 |
| Obesity | 37.8 |
| History of VTE | 26.0 |
| Cancer | 22.3 |
| Bed rest 5 days | 12.0 |
| Major surgery | 11.2 |
| Congestive heart failure | 8.2 |
| Varicose veins | 5.8 |
| Fracture (hip or leg) | 3.7 |
| Estrogen treatment | 2.0 |
| Stroke | 1.8 |
| Multiple trauma | 1.1 |
| Childbirth | 1.1 |
| Myocardial infarction | 0.7 |
Current or Recent Prior Hospitalization
The risk of VTE is elevated among hospitalized patients. The prevalence of DVT varies across hospital specialties, reaching up to 80% in major trauma, spinal cord injury, and critical care.3 The Epidemiologic International Day for the Evaluation of Patients at Risk for Venous Thromboembolism in the Acute Hospital Care Setting (ENDORSE) study evaluated the prevalence of VTE risk factors in the acute hospital care setting.12 Among the 68,183 patients enrolled, more than one‐half (52%) were judged to be at risk of VTE. In a case‐control study examining 625 patients with a first lifetime VTE, confinement to a hospital (among other risk factors) was found to be an independent and important predictor of VTE (odds ratio [OR], 8.0; 95% confidence interval [CI], 4.514.2) (Figure 1).13 Recent hospitalization is also an important risk factor for VTE, and patients who are readmitted to the hospital should be considered moderate or high risk.13
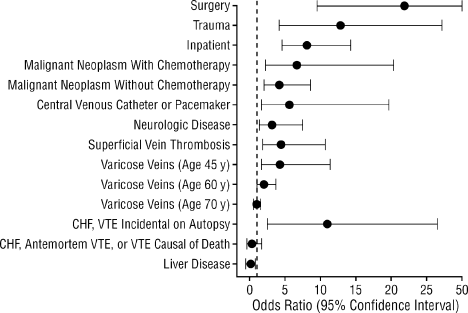
Age
Patient age must be considered when assessing VTE risk. VTE is predominantly a disease of older age, and age older than 75 years is an important risk factor for the condition.11 In general, patients older than 40 years have a significantly increased risk compared with younger patients, and the risk approximately doubles with each additional decade.10 Given the aging population, the prevalence of VTE and its complications are expected to increase.
Women of childbearing age experience VTE more frequently than men of the same age, due to pregnancy and exposure to contraceptive therapy.14 This risk, however, is modest compared with the risk among older patients. After age 45 years, the incidence of VTE increases markedly for both sexes, becoming more prominent in men.14 Compared with women, men also have an increased risk of recurrent VTE.15
Despite the effect of age on VTE risk, the risk among patients younger than 40 years may be underestimated because this subgroup has not been extensively studied. For reasons that are not well‐understood, the risk of VTE associated with heart failure is higher in patients younger than 40 years, and the relative risk of PE in patients with chronic obstructive pulmonary disease (COPD) is also higher in younger patients.16, 17
Cancer and Its Treatment
Cancer patients, on average, have twice the risk of VTE compared with noncancer patients.18 This risk, however, varies considerably by cancer type. According to an assessment of nearly 41 million hospitalized patients in the National Hospital Discharge Survey (NHDS), the relative risk of VTE varied from 1.02 in patients with bladder cancer to 4.34 in patients with cancer of the pancreas.18
VTE is one of the most common complications of cancer and cancer therapy, and it is the second leading cause of death among hospitalized cancer patients.19 Molecular mechanisms underlying thromboembolic events in cancer patients include tumor cell procoagulants, inflammatory cell cytokines, mediators of platelet adhesion, and tumor‐related stasis and endothelial damage.20 The clinical implications of these processes are severe. Cancer exacerbates the natural course of VTE, increasing the risk of recurrent VTE and major bleeding, and VTE worsens the prognosis of cancer, increasing the risk of death among cancer patients.
Various cancer therapiesincluding surgery, chemotherapy, hematopoietic stem cell transplantation, and even growth factor supportalso increase the risk of VTE, in part because extrinsic factors such as surgery or chemotherapy can intensify the hypercoagulable process.18, 2123 In the NHDS, cancer patients undergoing surgery had at least twice the risk of postoperative DVT and more than 3 times the risk of PE compared with noncancer patients undergoing similar procedures.18
Cancer is an independent predictor of thromboprophylaxis failure following surgery. The @RISTOS Project found that VTE was the most common cause of death among 2373 patients undergoing general, urologic, or gynecologic surgery for cancer.24 A multivariate analysis identified 5 independent risk factors for VTE after cancer surgery:
-
Previous VTE (OR, 5.98; 95% CI, 2.1316.80)
-
Anesthesia 2 hours (OR, 4.50; 95% CI, 1.0619.04)
-
Bed rest 4 days (OR, 4.37; 95% CI, 2.457.78)
-
Age 60 years (OR, 2.63; 95% CI, 1.215.71)
-
Advanced‐stage cancer (OR, 2.68; 95% CI, 1.375.24)
Cardiovascular Disease
The risk of VTE is pronounced among patients with cardiovascular disease. After stroke and coronary disease, VTE is the third most common cardiovascular disorder, and PE causes more deaths each year than myocardial infarction (MI).25 Several cardiovascular diseases, including hypertension, stroke, acute MI, and heart failure, are independently associated with VTE.10, 2627 Related disorders, including diabetes and the metabolic syndrome, also increase the risk of VTE.26, 28
Congestive heart failure (CHF) is a risk factor for VTE, and the severity of illness increases risk. In the DVT‐Free Prospective Registry, 13% of patients with ultrasound‐confirmed DVT had CHF.29 In a subgroup analysis of patients of the Prophylaxis in Medical Patients with Enoxaparin (MEDENOX) study, the incidence of VTE exceeded 20% in patients with New York Heart Association (NYHA) class IV heart failure, compared with 12% in patients with NYHA class III heart failure.30 Another study found that VTE risk increases as left ventricular ejection fraction (LVEF) decreases, with an LVEF of less than 20% associated with a VTE OR of 38.3 (95% CI, 9.6152.5).31
Infectious Disease
Acute infection may increase the relative risk of VTE by as much as 50% and is associated with VTE event rates of up to 26%.11 Acute infections may be associated with acute inflammation, adverse effects on cardiac or pulmonary function, and prolonged immobilization.30, 32, 33 Human immunodeficiency virus (HIV) patients may also have an increased risk of VTE due to a circulating lupus anticoagulant and/or the presence of acute infection.34
Obesity
The 2008 ACCP guideline update recognizes obesity, for the first time, as a risk factor for VTE.3 Obesity was 1 of the 5 most frequent comorbidities found in patients with DVT in the DVT‐Free Prospective Registry.29 It increases the risk of both incident and recurrent VTE, with every 1‐point increase in body mass index (BMI) increasing the risk of recurrent VTE by 4.4% (95% CI, 1.37.6%; P < 0.001).35
Pregnancy and Puerperium
Pregnancy, particularly the postpartum period, is associated with an increased risk of VTE in women, even though the absolute risk is small.36 Still, PE is one of the leading causes of maternal death following childbirth.10 Smoking, prior VTE, and inherited thrombophilias all increase the risk of VTE in pregnant women.10 The risk begins to rise in the first trimester, and when prophylaxis is needed, it should be started early in gestation.37
Pulmonary Disease
COPD is another risk factor for the development of VTE. COPD patients who develop VTE tend to be older, hospitalized in the intensive care unit (ICU), and on mechanical ventilation.38 In the DVT‐Free Prospective Registry, 12% of patients with ultrasound‐confirmed DVT had COPD.29
Trauma and Surgery
Injury to the body tissue, via trauma or surgery, stimulates the body's clotting mechanism and increases the risk of thromboembolic complications. During the perioperative period, the circulatory system must balance a variety of assaults: an immune response to surgical stress, prolonged immobilization during surgery and recovery, vasodilation associated with general or regional anesthesia, and hypercoagulability due to venous stasis and vascular injury.39 Renal transplant recipients have an increased risk of VTE due to a chronic hypercoagulable state.40 In surgery patients, perioperative complications such as dehydration and acute infection increase the risk of VTE beyond the risk associated with the surgical procedure itself.10
VTE risk is increased approximately 13‐fold by recent major trauma or lower‐extremity injury.13 In the absence of prophylaxis, the overall risk of VTE among patients undergoing major surgery is increased nearly 22‐fold.13 After controlling for the type of surgery, additional independent risk factors for VTE within 3 months of major surgery include:41, 42
-
Obesity
-
Central venous catheter placement
-
Malignancy
-
Smoking
-
Heart failure
-
Previous DVT
-
Prolonged immobility
-
Infection
Many surgical and medical inpatients share common risk factors, and without prophylaxis, the incidence of hospital‐acquired DVT ranges from 10% to 40% for both groups.3
Inherited or Acquired Risk Factors
VTE is a multifactorial disease, and recent evidence indicates that some heritable traits may be potent risk factors for VTE.43 Approximately 35% of patients with DVT will have at least 1 of 5 traits related to an inherited blood clotting disorder:43
-
Deficiencies in the anticoagulation factors protein C, protein S, or antithrombin, or
-
Mutations in the factor V and prothrombin genes, resulting in Factor V Leiden and prothrombin G20210A, respectively.
Certain inherited traits and genetic polymorphisms increase the risk of VTE by interacting with clinical risk factors such as contraceptive use, pregnancy, surgery, trauma, and cancer. One recent study found that oral estrogen therapy among women with the CYP3A5*1 allele was associated with a particularly high risk of VTE.44 Although widespread screening for inherited risk factors is not currently practical, future tools may incorporate genetic polymorphisms to more precisely identify patients who would benefit from aggressive prophylaxis.
Lifestyle Factors
Lifestyle factors have a significant effect on VTE risk. Smoking increases the risk of VTE by 20% to 30%, and a sedentary lifestyle also increases the risk of VTE.26, 45 In fact, women who exercise regularly and consume alcohol in moderation have one‐half the risk of VTE as women who have a sedentary lifestyle and drink little or no alcohol.42 For both men and women, a diet high in fruits, vegetables, and fish is associated with a lower lifetime risk of VTE.46
Medications
Medications may also increase the risk of VTE. In cancer patients with anemia, for example, the use of erythropoiesis‐stimulating agents such as recombinant erythropoietin and darbepoetin was recently shown to increase the risk of VTE by 57% (95% CI, 3187%) and increase mortality risk by 10% (95% CI, 120%).23 In addition, combination hormone replacement therapy in women is associated with a higher risk of VTE compared with estrogen monotherapy, and transdermal contraceptive systems more than double the risk of VTE compared with oral contraceptives (95% CI, 1.33.8).47, 48 Recent studies have also reported an increased risk of VTE with some psychiatric drugs, including amitriptyline, clozapine, olanzapine, and risperidone.4952
Thromboprophylaxis in the Hospital Setting
Despite the prevalence of risk factors and compelling evidence regarding the value of prophylaxis, VTE prophylaxis is suboptimal in hospitalized medical and surgical patients. In a study of 123,304 hospitalized patients who were determined to be at risk of VTE, only 13.3% received prophylaxis in accordance with ACCP guidelines.53 Compliance ranged from a high of 52.4% among patients undergoing orthopedic surgery to a low of 2.8% among patients undergoing neurosurgery.53 Results from several other large trials echo these findings (Table 2).12, 5456
| Trial | Patient Type | Total Patients | Patients at Risk of VTE (Based on ACCP Criteria) (%) | At‐Risk Patients Receiving Recommended Prophylaxis | |
|---|---|---|---|---|---|
| Medical Patients (%) | Surgical Patients (%) | ||||
| |||||
| IMPROVE | Medical patients | 15,156 | 52 | 61 | n/a |
| ENDORSE | Medical and surgical patients | 68,183 | 51.8 | 39.5 | 58.5 |
| ADHERE | Hospitalized heart failure patients | 155,073 | 46 | 30.6 | n/a |
| Amin et al.56 (2008) | Medical and surgical patients | 258,556 | 26.4 | 9.8 | 17.9 |
Reasons for Inadequate Prophylaxis
Researchers have identified a range of barriers to adequate VTE prophylaxis (Table 3).57 Some of these barriers are outlined below.
|
| Variability in clinician knowledge of risk assessment and appropriate prophylaxis |
| Lack of agreement with, and inconsistency between, guidelines in certain patient populations |
| Perceived lack of need |
| Concerns about adverse effects |
| Lack of hospital support systems and policies |
| Lack of established responsibilities for prophylaxis |
Underestimation of Risk of Clotting
VTE is often clinically silent, leading some physicians to mistakenly believe that it is rare.58 In hospitalized surgical patients, for example, the incidence of thromboembolic complications during a short postoperative stay may be low. Given that many cases of symptomatic VTE occur after hospital discharge, hospitalists and surgeons may be unaware of the true incidence of DVT.59
Overestimation of the Risk of Bleeding
Physicians may also overestimate the risk of possible side effects of prophylaxis, such as major bleeding or heparin‐induced thrombocytopenia (HIT).58 Fear of excess bleeding has been cited by physicians as a leading reason for their decision to withhold thromboprophylaxis from at‐risk hospitalized patients.60 Physicians are particularly fearful of complications among elderly patients, who are less likely to receive adequate prophylaxis than younger patients with a similar risk of VTE.61 When bleeding does occur, it rarely results in death. On the other hand, PE may account for as many as 10% of hospital deaths.9
Guideline Confusion and Complexity
Discrepancies between guidelines published by different medical societies contribute to confusion in choosing a management approach. The American Academy of Orthopedic Surgeons (AAOS), for example, describes aspirin alone as a reasonable choice for VTE prophylaxis in some patients, but the ACCP guidelines advise against the use of aspirin monotherapy.58 The cumbersome nature of multiple risk‐assessment and treatment algorithms can also be problematic.61 Furthermore, certain patient subgroups, such as those with cirrhosis, severe renal failure, and epidural catheters, have been excluded from randomized controlled trials, and the management of such patients is not straightforward.
Absence of Institutional Protocols and Information Technology Support
The lack of institution‐level guidance and support can have a detrimental effect on patient care. In a 2007 survey of 127 community hospitals, the prevalence of institutional protocols related to VTE was low: only 60% had protocols to encourage prophylaxis in at‐risk patients, 54% had guidelines to assist in appropriate drug selection, and 43% had guidelines for the dosing of prophylaxis regimens.62 A lack of systems for data collection and audit has also been identified as a barrier to the implementation of prophylaxis guidelines.57 Thus, hospitals need to adopt protocols such as:3
-
Written, institution‐wide thromboprophylaxis policies
-
Preprinted order forms and computer decision‐support systems
-
Policies specifying responsibilities for assessing VTE risk and prescribing prophylaxis
Conclusions
VTE is the most common preventable cause of hospital death, and prophylaxis is underused in hospitalized patients. Although VTE risk factors are numerous and complex, deciding whether to use prophylaxis need not be complicated. In general, elderly patients, medically‐ill patients, and patients undergoing surgery will benefit from prophylaxis, as well as those who are hospitalized for more than 1 night. Hospitalized patients with at least 1 risk factor should be considered for pharmacologic prophylaxis. In general, the risk of hospital‐acquired VTE greatly exceeds the risk of bleeding with prophylactic doses of anticoagulation. A patient's risk of VTE may change, and regular assessment of this risk should be mandated if pharmacologic therapy is not initiated at the time of admission.
Numerous barriers to the optimal use of VTE prophylaxis exist, and hospitals must implement systems changes and multidisciplinary approaches to overcome these barriers. The fourth article in this supplement provides detailed strategies for meeting VTE performance measures and overcoming barriers to the optimal use of prophylaxis.
- , , , , .Estimated annual numbers of US acute‐care hospital patients at risk for venous thromboembolism.Am J Hematol.2007;82(9):777–782.
- , , .Trends in the incidence of pulmonary embolism and deep venous thrombosis in hospitalized patients.Am J Cardiol.2005;95(12):1525–1526.
- , , , et al.Prevention of venous thromboembolism: American College of Chest Physicians evidence‐based clinical practice guidelines. 8th ed.Chest.2008;133(6 suppl):381S–453S.
- , .Management of deep vein thrombosis and pulmonary embolism. A statement for healthcare professionals. Council on Thrombosis (in consultation with the Council on Cardiovascular Radiology), American Heart Association.Circulation.1996;93(12)2212–2245.
- , , , et al.A population‐based perspective of the hospital incidence and case‐fatality rates of deep vein thrombosis and pulmonary embolism. The Worcester DVT Study.Arch Intern Med.1991;151(5):933–938.
- , , , et al.The long‐term clinical course of acute deep venous thrombosis.Ann Intern Med.1996;125(1):1–7.
- , .Direct medical cost of managing deep vein thrombosis according to the occurrence of complications.Pharmacoeconomics.2002;20(9):603–615.
- , .Direct medical costs of venous thromboembolism and subsequent hospital readmission rates: an administrative claims analysis from 30 managed care organizations.J Manag Care Pharm.2007;13(6):475–486.
- , , .Double trouble for 2,609 hospitalized medical patients who developed deep vein thrombosis: prophylaxis omitted more often and pulmonary embolism more frequent.Chest.2007;132(2):554–561.
- , .Risk factors for venous thromboembolism.Circulation.2003;107(23 suppl 1):I9–I16.
- , , , et al.Risk factors for VTE in hospitalized patients with acute medical illness: analysis of the MEDENOX Study.Arch Intern Med.2004;164(9):963–968.
- , , , et al.;ENDORSE Investigators.Venous thromboembolism risk and prophylaxis in the acute hospital care setting (ENDORSE study): a multinational cross‐sectional study.Lancet.2008;371(9610):387–394.
- , , , et al.Risk factors for deep vein thrombosis and pulmonary embolism: a population‐based case‐control study.Arch Intern Med.2000;160(6):809–815.
- , .Range of published estimates of venous thromboembolism incidence in young women.Contraception.2007;75(5):328–336.
- , , , , , .The risk of recurrent venous thromboembolism in men and women.N Engl J Med.2004;350(25):2558–2563.
- , , , et al.Risk of venous thromboembolism in patients hospitalized with heart failure.Am J Cardiol.2006;98(6):793–795.
- , , , et al.Pulmonary embolism and deep venous thrombosis in hospitalized adults with chronic obstructive pulmonary disease.J Cardiovasc Med.2007;8(4):253–257.
- , , , et al.Incidence of venous thromboembolism in patients hospitalized with cancer.Am J Med.2006;119(1):60–68.
- , , , et al.Causes of death in cancer patients.J Med.1975;6(1):61–64.
- , .Tissue factor and angiogenesis in cancer.Curr Opin Hematol.2002;9(5):401–406.
- , , , et al.Incidence of venous thromboembolism in patients with ovarian cancer undergoing platinum/paclitaxel‐containing first‐line chemotherapy: an exploratory analysis by the Arbeitsgemeinschaft Gynaekologische Onkologie Ovarian Cancer Study Group.J Clin Oncol.2008;26(16):2683–2689.
- , , , , , .The incidence of and risk factors for venous thromboembolism (VTE) and bleeding among 1514 patients undergoing hematopoietic stem cell transplantation: implications for VTE prevention.Blood.2008;112(3):504–510.
- , , , et al.Venous thromboembolism and mortality associated with recombinant erythropoietin and darbepoetin administration for the treatment of cancer‐associated anemia.JAMA.2008;299(8):914–924.
- , , , et al.A clinical outcome‐based prospective study on venous thromboembolism after cancer surgery: the @RISTOS project.Ann Surg.2006;243(1):89–95.
- .Pulmonary embolism in thrombolysis: a clarion call for international collaboration.J Am Coll Cardiol.1992;19:246–247.
- , , , et al.Cardiovascular risk factors and venous thromboembolism: a meta‐analysis.Circulation.2008;117(1):93–102.
- , , , , , .Family history of myocardial infarction is an independent risk factor for venous thromboembolism: the Tromsø study.J Thromb Haemost.2008;6(11):1851–1857.
- , , , et al.Abdominal obesity is essential for the risk of venous thromboembolism in the metabolic syndrome—the Tromsø study.J Thromb Haemost.2009;7(5):739–745.
- , .A prospective registry of 5,451 patients with ultrasound‐confirmed deep vein thrombosis.Am J Cardiol.2004;93(2):259–262.
- , , , et al.Prevention of venous thromboembolism in medical patients with enoxaparin; a subgroup analysis of the MEDENOX study.Blood Coagul Fibrinolysis.2003;14:341–348.
- , , .Congestive heart failure and outpatient risk of venous thromboembolism: a retrospective, case‐control study.J Clin Epidemiol.2001;54(8):810–816.
- , , , et al;ARTEMIS Investigators.Efficacy and safety of fondaparinux for the prevention of venous thromboembolism in older acute medical patients: randomised placebo controlled trial.BMJ.2006;332(7537):325–329.
- .Randomised, controlled trial of low‐dose heparin for prevention of fatal pulmonary embolism in patients with infectious diseases. The Heparin Prophylaxis Study Group.Lancet.1996;347(9012):1357–1361.
- , , , et al.HIV infection is a risk factor for venous thromboembolism.AIDS Patient Care STDS.2002;16(5):205–209.
- , , , et al.Overweight, obesity, and the risk of recurrent venous thromboembolism.Arch Intern Med.2008;168(15):1678–1683.
- .Venous thromboembolism in women.Vasc Med.2008;13(3):255–266.
- , , .Thrombosis during pregnancy and the postpartum period.Am J Obstet Gynecol.2005;193(1):216–219.
- , , , et al.Chronic obstructive pulmonary disease and deep vein thrombosis: a prevalent combination.J Thromb Thrombolysis.2008;26(1):35–40.
- , , .Venous thromboembolism in trauma: a local manifestation of systemic hypercoagulability?J Trauma.2003;54(2):224–231.
- , .Acquired hypercoagulable state in renal transplant recipients.Thromb Haemost.2004;91(4):646–654.
- , , , .Venous thromboembolism in patients undergoing surgery: low rates of prophylaxis and high rates of filter insertion.Thromb Haemost.2007;98(6):1220–1225.
- , , , , , .A 10‐year analysis of venous thromboembolism on the surgical service: the effect of practice guidelines for prophylaxis.Surgery.2008;144(1):3–11.
- , , , et al;EDITH Collaborative Study Group.Family history as a risk factor for venous thromboembolism.Thromb Res.2008;122(5):624–629.
- , , , et al;Estrogen and Thromboembolism Risk (ESTHER) Study Group.Synergism between oral estrogen therapy and cytochrome P450 3A5*1 allele on the risk of venous thromboembolism among postmenopausal women.J Clin Endocrinol Metab.2008;93(8):3082–3087.
- , , .The relationship between lifestyle factors and venous thromboembolism among women: a report from the MISS study.Br J Haematol.2009;144(2):234–240.
- , , , , .Greater fish, fruit, and vegetable intakes are related to lower incidence of venous thromboembolism: the Longitudinal Investigation of Thromboembolism Etiology.Circulation.2007;115(2):188–195.
- , , .Association between hormone replacement therapy and subsequent arterial and venous vascular events: a meta‐analysis.Eur Heart J.2008;29(16):2031–2041.
- , , , .Venous thromboembolism, myocardial infarction, and stroke among transdermal contraceptive system users.Obstet Gynecol.2007;109(2 Pt 1):339–346.
- , , , , , .Pulmonary thromboembolism associated with olanzapine and risperidone.J Emerg Med.2008;35(2):159–161.
- , , , .Associations between venous thromboembolism and antipsychotics. A study of the WHO database of adverse drug reactions.Drug Saf.2008;31(8):685–694.
- .Evaluating the association between clozapine and venous thromboembolism.Am J Health Syst Pharm.2008;65(19):1825–1829.
- , .Antidepressant drug use and risk of venous thromboembolism.Pharmacotherapy.2008;28(2):144–150.
- , , , .Hospitals' compliance with prophylaxis guidelines for venous thromboembolism.Am J Health Syst Pharm.2007;64(1):69–76.
- , , , et al.Venous thromboembolism prophylaxis in acutely ill hospitalized medical patients.Chest.2007;132:936–945.
- , , , et al.Adhere Scientific Advisory Committee and Investigators. Venous thromboembolism prophylaxis in hospitalized heart failure patients.J Card Fail.2008;14(2):127–132.
- , , , et al. Are hospitals following guidelines for VTE Prevention? The Venous Thromboembolism Study to Assess the Rate of Thromboprophylaxis. Presented at the 50th Annual Meeting of the American College of Hematology, San Francisco, CA, December 6–9, 2008. Abstract 1286.
- , , , et al.A systematic review of strategies to improve prophylaxis for venous thromboembolism in hospitals.Ann Surg.2005;241(3):397–415.
- .A need for a simplified approach to venous thromboembolism prophylaxis in acute medical inpatients.Int J Clin Pract.2007;61(2):336–340.
- .Barriers to the optimal use of anticoagulants after orthopaedic surgery.Arch Orthop Trauma Surg.2008 (Published online Oct, 8.). [http://dx.doi.org/DOI 10.1007/s00402‐008‐0765‐9]
- , , , , .Venous thrombosis in cancer patients: Insights from the FRONTLINE survey.Oncologist.2003;8:381–388.
- , , , et al.Incorrect use of thromboprophylaxis for venous thromboembolism in medical and surgical patients: results of a multicentric, observational and cross‐sectional study in Brazil.J Thromb Haemost.2006;4(6):1266–1270.
- , , , , .Survey of hospitals for guidelines, policies, and protocols for anticoagulants.Am J Health Syst Pharm.2007;64(11):1203–1208.
Venous thromboembolism (VTE) is a common and potentially devastating complication of medical illness and surgical intervention. Among patients discharged from acute‐care hospitals in 2003, more than 12 million (31%) had a moderate or high risk of VTE during hospitalization, including 11% at risk due to surgical procedures and 20% at risk due to medical illnesses.1 The incidence of VTEwhich can present as deep vein thrombosis (DVT) or pulmonary embolism (PE)is rising in hospitalized patients.2 Despite the availability of effective prophylaxis, VTE is the third most common cause of hospital‐related death and the most common preventable cause of hospital mortality.3
The clinical impact of VTE is significant. While total incidence, prevalence, and mortality rates of VTE are elusive, the annual incidence of DVT is thought to be as high as 2 million.4 The most serious complication of DVT is acute PE, which occurs in approximately 600,000 patients per year, one‐third of whom die.5 DVT may also be complicated by recurrent episodes of VTE and postthrombotic sequelae such as chronic venous stasis, venous ulceration, debilitating pain, and intractable edema.6 One prospective cohort study found that 30% of patients who had experienced a first episode of DVT developed recurrent VTE within 8 years, and the incidence of postthrombotic syndrome (PTS) was 23% after just 2 years.6
The economic burden of VTE is substantial, due both to the initial event and to the high rate of hospital readmission. The estimated average cost of DVT management (including initial acute care and 6 months of follow‐up care) is $10,072 per patient, and the corresponding cost for PE is $14,649.7 Hospital readmission occurs in 5% to 14% of patients, more than one‐half of whom are readmitted within 90 days.8 For patients with recurrent DVT or PE, the mean total hospitalization costs of readmission are $11,862 and $14,722, respectively.8
The clinical and economic burden of VTE can be significantly mitigated by the use of effective prophylaxis. Because VTE is difficult to diagnose antemortem, it is easier and safer to prevent with appropriate prophylaxis than to diagnose after it has occurred. Unfortunately, VTE prophylaxis is markedly underused, particularly among high‐risk, hospitalized medical patients who would most benefit from it.9
This article summarizes specific risk factors for VTE and provides guidance in identifying patients who may require thromboprophylaxis. Barriers to optimal VTE prophylaxis in the hospital setting will also be explored.
Methodology
For this article and the ones that follow, relevant literature was identified through a Medline search (January 1980 to December 2008) using the following search terms: venous thromboembolism, pulmonary embolism, deep vein thrombosis, epidemiology, risk factors, prophylaxis, mechanical prophylaxis, diagnosis, treatment, anticoagulants, monitoring, secondary prevention, guideline, adherence, treatment protocol, performance measure, and quality improvement. The bibliographies of all key texts were searched for additional relevant articles. The websites of the American College of Chest Physicians (ACCP), American Society of Clinical Oncology (ASCO), National Comprehensive Cancer Network (NCCN), and Society of Hospital Medicine (SHM) were also searched for annual meeting abstracts, position statements, and other key publications.
Evidence‐based clinical guidelines were identified through a search of the National Guideline Clearinghouse (
Pathogenesis of VTE
Venous thrombosis occurs as a result of at least 1 of 3 underlying factors: alterations in blood flow, vascular endothelial injury, and alterations in the constitution of the blood.10 Each potential underlying factor encompasses a wide range of risk factors and clinical scenarios. Alterations in venous blood flow can include several situations, including venous stasis, venous hypertension, and valvular incompetence. Endothelial injury can arise from shear stress, direct trauma, infection, hypertension, or other sources of endothelial damage. Hypercoagulability from alterations in the constitution of the blood may be due to antithrombin deficiency, cancer, surgery, pregnancy, or other risk factors. The presence of any of these factors indicates an elevated risk of VTE, and the presence of multiple factors further increases risk.10
Risk Factors for VTE
VTE can occur in a wide variety of clinical circumstances. Recognized risk factors for VTE include hospitalization for an acute medical illness, cardiovascular disease, pulmonary disease, major surgery, multiple trauma, obesity, and increasing age.10 Additional factors that place patients at increased risk of VTE (independent of age) include a history of prior VTE, known hypercoagulable states, active cancer, and acute infection.11 Hospital‐acquired risk factors such as immobility, acute illness, or medical interventions may lead to the development of VTE in these patients. Severity of illness must be factored into the risk assessment, and all patients need to be assessed for VTE risk at the time of hospital admission and daily thereafter if pharmacologic therapy is not initiated.
In a review of 1231 consecutive patients treated for acute DVT and/or PE, 96.3% had at least 1 risk factor for VTE, and more than one‐third (39%) had 3 or more risk factors (Table 1).10 The incidence of VTE in hospitalized patients is directly related to the number of risk factors present:10
-
1 risk factor: 11%
-
2 risk factors: 24%
-
3 risk factors: 36%
-
4 risk factors: 50%
-
5 risk factors: > 90%
| Risk Factor | Patients (%) |
|---|---|
| |
| Age 40 years | 88.5 |
| Obesity | 37.8 |
| History of VTE | 26.0 |
| Cancer | 22.3 |
| Bed rest 5 days | 12.0 |
| Major surgery | 11.2 |
| Congestive heart failure | 8.2 |
| Varicose veins | 5.8 |
| Fracture (hip or leg) | 3.7 |
| Estrogen treatment | 2.0 |
| Stroke | 1.8 |
| Multiple trauma | 1.1 |
| Childbirth | 1.1 |
| Myocardial infarction | 0.7 |
Current or Recent Prior Hospitalization
The risk of VTE is elevated among hospitalized patients. The prevalence of DVT varies across hospital specialties, reaching up to 80% in major trauma, spinal cord injury, and critical care.3 The Epidemiologic International Day for the Evaluation of Patients at Risk for Venous Thromboembolism in the Acute Hospital Care Setting (ENDORSE) study evaluated the prevalence of VTE risk factors in the acute hospital care setting.12 Among the 68,183 patients enrolled, more than one‐half (52%) were judged to be at risk of VTE. In a case‐control study examining 625 patients with a first lifetime VTE, confinement to a hospital (among other risk factors) was found to be an independent and important predictor of VTE (odds ratio [OR], 8.0; 95% confidence interval [CI], 4.514.2) (Figure 1).13 Recent hospitalization is also an important risk factor for VTE, and patients who are readmitted to the hospital should be considered moderate or high risk.13

Age
Patient age must be considered when assessing VTE risk. VTE is predominantly a disease of older age, and age older than 75 years is an important risk factor for the condition.11 In general, patients older than 40 years have a significantly increased risk compared with younger patients, and the risk approximately doubles with each additional decade.10 Given the aging population, the prevalence of VTE and its complications are expected to increase.
Women of childbearing age experience VTE more frequently than men of the same age, due to pregnancy and exposure to contraceptive therapy.14 This risk, however, is modest compared with the risk among older patients. After age 45 years, the incidence of VTE increases markedly for both sexes, becoming more prominent in men.14 Compared with women, men also have an increased risk of recurrent VTE.15
Despite the effect of age on VTE risk, the risk among patients younger than 40 years may be underestimated because this subgroup has not been extensively studied. For reasons that are not well‐understood, the risk of VTE associated with heart failure is higher in patients younger than 40 years, and the relative risk of PE in patients with chronic obstructive pulmonary disease (COPD) is also higher in younger patients.16, 17
Cancer and Its Treatment
Cancer patients, on average, have twice the risk of VTE compared with noncancer patients.18 This risk, however, varies considerably by cancer type. According to an assessment of nearly 41 million hospitalized patients in the National Hospital Discharge Survey (NHDS), the relative risk of VTE varied from 1.02 in patients with bladder cancer to 4.34 in patients with cancer of the pancreas.18
VTE is one of the most common complications of cancer and cancer therapy, and it is the second leading cause of death among hospitalized cancer patients.19 Molecular mechanisms underlying thromboembolic events in cancer patients include tumor cell procoagulants, inflammatory cell cytokines, mediators of platelet adhesion, and tumor‐related stasis and endothelial damage.20 The clinical implications of these processes are severe. Cancer exacerbates the natural course of VTE, increasing the risk of recurrent VTE and major bleeding, and VTE worsens the prognosis of cancer, increasing the risk of death among cancer patients.
Various cancer therapiesincluding surgery, chemotherapy, hematopoietic stem cell transplantation, and even growth factor supportalso increase the risk of VTE, in part because extrinsic factors such as surgery or chemotherapy can intensify the hypercoagulable process.18, 2123 In the NHDS, cancer patients undergoing surgery had at least twice the risk of postoperative DVT and more than 3 times the risk of PE compared with noncancer patients undergoing similar procedures.18
Cancer is an independent predictor of thromboprophylaxis failure following surgery. The @RISTOS Project found that VTE was the most common cause of death among 2373 patients undergoing general, urologic, or gynecologic surgery for cancer.24 A multivariate analysis identified 5 independent risk factors for VTE after cancer surgery:
-
Previous VTE (OR, 5.98; 95% CI, 2.1316.80)
-
Anesthesia 2 hours (OR, 4.50; 95% CI, 1.0619.04)
-
Bed rest 4 days (OR, 4.37; 95% CI, 2.457.78)
-
Age 60 years (OR, 2.63; 95% CI, 1.215.71)
-
Advanced‐stage cancer (OR, 2.68; 95% CI, 1.375.24)
Cardiovascular Disease
The risk of VTE is pronounced among patients with cardiovascular disease. After stroke and coronary disease, VTE is the third most common cardiovascular disorder, and PE causes more deaths each year than myocardial infarction (MI).25 Several cardiovascular diseases, including hypertension, stroke, acute MI, and heart failure, are independently associated with VTE.10, 2627 Related disorders, including diabetes and the metabolic syndrome, also increase the risk of VTE.26, 28
Congestive heart failure (CHF) is a risk factor for VTE, and the severity of illness increases risk. In the DVT‐Free Prospective Registry, 13% of patients with ultrasound‐confirmed DVT had CHF.29 In a subgroup analysis of patients of the Prophylaxis in Medical Patients with Enoxaparin (MEDENOX) study, the incidence of VTE exceeded 20% in patients with New York Heart Association (NYHA) class IV heart failure, compared with 12% in patients with NYHA class III heart failure.30 Another study found that VTE risk increases as left ventricular ejection fraction (LVEF) decreases, with an LVEF of less than 20% associated with a VTE OR of 38.3 (95% CI, 9.6152.5).31
Infectious Disease
Acute infection may increase the relative risk of VTE by as much as 50% and is associated with VTE event rates of up to 26%.11 Acute infections may be associated with acute inflammation, adverse effects on cardiac or pulmonary function, and prolonged immobilization.30, 32, 33 Human immunodeficiency virus (HIV) patients may also have an increased risk of VTE due to a circulating lupus anticoagulant and/or the presence of acute infection.34
Obesity
The 2008 ACCP guideline update recognizes obesity, for the first time, as a risk factor for VTE.3 Obesity was 1 of the 5 most frequent comorbidities found in patients with DVT in the DVT‐Free Prospective Registry.29 It increases the risk of both incident and recurrent VTE, with every 1‐point increase in body mass index (BMI) increasing the risk of recurrent VTE by 4.4% (95% CI, 1.37.6%; P < 0.001).35
Pregnancy and Puerperium
Pregnancy, particularly the postpartum period, is associated with an increased risk of VTE in women, even though the absolute risk is small.36 Still, PE is one of the leading causes of maternal death following childbirth.10 Smoking, prior VTE, and inherited thrombophilias all increase the risk of VTE in pregnant women.10 The risk begins to rise in the first trimester, and when prophylaxis is needed, it should be started early in gestation.37
Pulmonary Disease
COPD is another risk factor for the development of VTE. COPD patients who develop VTE tend to be older, hospitalized in the intensive care unit (ICU), and on mechanical ventilation.38 In the DVT‐Free Prospective Registry, 12% of patients with ultrasound‐confirmed DVT had COPD.29
Trauma and Surgery
Injury to the body tissue, via trauma or surgery, stimulates the body's clotting mechanism and increases the risk of thromboembolic complications. During the perioperative period, the circulatory system must balance a variety of assaults: an immune response to surgical stress, prolonged immobilization during surgery and recovery, vasodilation associated with general or regional anesthesia, and hypercoagulability due to venous stasis and vascular injury.39 Renal transplant recipients have an increased risk of VTE due to a chronic hypercoagulable state.40 In surgery patients, perioperative complications such as dehydration and acute infection increase the risk of VTE beyond the risk associated with the surgical procedure itself.10
VTE risk is increased approximately 13‐fold by recent major trauma or lower‐extremity injury.13 In the absence of prophylaxis, the overall risk of VTE among patients undergoing major surgery is increased nearly 22‐fold.13 After controlling for the type of surgery, additional independent risk factors for VTE within 3 months of major surgery include:41, 42
-
Obesity
-
Central venous catheter placement
-
Malignancy
-
Smoking
-
Heart failure
-
Previous DVT
-
Prolonged immobility
-
Infection
Many surgical and medical inpatients share common risk factors, and without prophylaxis, the incidence of hospital‐acquired DVT ranges from 10% to 40% for both groups.3
Inherited or Acquired Risk Factors
VTE is a multifactorial disease, and recent evidence indicates that some heritable traits may be potent risk factors for VTE.43 Approximately 35% of patients with DVT will have at least 1 of 5 traits related to an inherited blood clotting disorder:43
-
Deficiencies in the anticoagulation factors protein C, protein S, or antithrombin, or
-
Mutations in the factor V and prothrombin genes, resulting in Factor V Leiden and prothrombin G20210A, respectively.
Certain inherited traits and genetic polymorphisms increase the risk of VTE by interacting with clinical risk factors such as contraceptive use, pregnancy, surgery, trauma, and cancer. One recent study found that oral estrogen therapy among women with the CYP3A5*1 allele was associated with a particularly high risk of VTE.44 Although widespread screening for inherited risk factors is not currently practical, future tools may incorporate genetic polymorphisms to more precisely identify patients who would benefit from aggressive prophylaxis.
Lifestyle Factors
Lifestyle factors have a significant effect on VTE risk. Smoking increases the risk of VTE by 20% to 30%, and a sedentary lifestyle also increases the risk of VTE.26, 45 In fact, women who exercise regularly and consume alcohol in moderation have one‐half the risk of VTE as women who have a sedentary lifestyle and drink little or no alcohol.42 For both men and women, a diet high in fruits, vegetables, and fish is associated with a lower lifetime risk of VTE.46
Medications
Medications may also increase the risk of VTE. In cancer patients with anemia, for example, the use of erythropoiesis‐stimulating agents such as recombinant erythropoietin and darbepoetin was recently shown to increase the risk of VTE by 57% (95% CI, 3187%) and increase mortality risk by 10% (95% CI, 120%).23 In addition, combination hormone replacement therapy in women is associated with a higher risk of VTE compared with estrogen monotherapy, and transdermal contraceptive systems more than double the risk of VTE compared with oral contraceptives (95% CI, 1.33.8).47, 48 Recent studies have also reported an increased risk of VTE with some psychiatric drugs, including amitriptyline, clozapine, olanzapine, and risperidone.4952
Thromboprophylaxis in the Hospital Setting
Despite the prevalence of risk factors and compelling evidence regarding the value of prophylaxis, VTE prophylaxis is suboptimal in hospitalized medical and surgical patients. In a study of 123,304 hospitalized patients who were determined to be at risk of VTE, only 13.3% received prophylaxis in accordance with ACCP guidelines.53 Compliance ranged from a high of 52.4% among patients undergoing orthopedic surgery to a low of 2.8% among patients undergoing neurosurgery.53 Results from several other large trials echo these findings (Table 2).12, 5456
| Trial | Patient Type | Total Patients | Patients at Risk of VTE (Based on ACCP Criteria) (%) | At‐Risk Patients Receiving Recommended Prophylaxis | |
|---|---|---|---|---|---|
| Medical Patients (%) | Surgical Patients (%) | ||||
| |||||
| IMPROVE | Medical patients | 15,156 | 52 | 61 | n/a |
| ENDORSE | Medical and surgical patients | 68,183 | 51.8 | 39.5 | 58.5 |
| ADHERE | Hospitalized heart failure patients | 155,073 | 46 | 30.6 | n/a |
| Amin et al.56 (2008) | Medical and surgical patients | 258,556 | 26.4 | 9.8 | 17.9 |
Reasons for Inadequate Prophylaxis
Researchers have identified a range of barriers to adequate VTE prophylaxis (Table 3).57 Some of these barriers are outlined below.
|
| Variability in clinician knowledge of risk assessment and appropriate prophylaxis |
| Lack of agreement with, and inconsistency between, guidelines in certain patient populations |
| Perceived lack of need |
| Concerns about adverse effects |
| Lack of hospital support systems and policies |
| Lack of established responsibilities for prophylaxis |
Underestimation of Risk of Clotting
VTE is often clinically silent, leading some physicians to mistakenly believe that it is rare.58 In hospitalized surgical patients, for example, the incidence of thromboembolic complications during a short postoperative stay may be low. Given that many cases of symptomatic VTE occur after hospital discharge, hospitalists and surgeons may be unaware of the true incidence of DVT.59
Overestimation of the Risk of Bleeding
Physicians may also overestimate the risk of possible side effects of prophylaxis, such as major bleeding or heparin‐induced thrombocytopenia (HIT).58 Fear of excess bleeding has been cited by physicians as a leading reason for their decision to withhold thromboprophylaxis from at‐risk hospitalized patients.60 Physicians are particularly fearful of complications among elderly patients, who are less likely to receive adequate prophylaxis than younger patients with a similar risk of VTE.61 When bleeding does occur, it rarely results in death. On the other hand, PE may account for as many as 10% of hospital deaths.9
Guideline Confusion and Complexity
Discrepancies between guidelines published by different medical societies contribute to confusion in choosing a management approach. The American Academy of Orthopedic Surgeons (AAOS), for example, describes aspirin alone as a reasonable choice for VTE prophylaxis in some patients, but the ACCP guidelines advise against the use of aspirin monotherapy.58 The cumbersome nature of multiple risk‐assessment and treatment algorithms can also be problematic.61 Furthermore, certain patient subgroups, such as those with cirrhosis, severe renal failure, and epidural catheters, have been excluded from randomized controlled trials, and the management of such patients is not straightforward.
Absence of Institutional Protocols and Information Technology Support
The lack of institution‐level guidance and support can have a detrimental effect on patient care. In a 2007 survey of 127 community hospitals, the prevalence of institutional protocols related to VTE was low: only 60% had protocols to encourage prophylaxis in at‐risk patients, 54% had guidelines to assist in appropriate drug selection, and 43% had guidelines for the dosing of prophylaxis regimens.62 A lack of systems for data collection and audit has also been identified as a barrier to the implementation of prophylaxis guidelines.57 Thus, hospitals need to adopt protocols such as:3
-
Written, institution‐wide thromboprophylaxis policies
-
Preprinted order forms and computer decision‐support systems
-
Policies specifying responsibilities for assessing VTE risk and prescribing prophylaxis
Conclusions
VTE is the most common preventable cause of hospital death, and prophylaxis is underused in hospitalized patients. Although VTE risk factors are numerous and complex, deciding whether to use prophylaxis need not be complicated. In general, elderly patients, medically‐ill patients, and patients undergoing surgery will benefit from prophylaxis, as well as those who are hospitalized for more than 1 night. Hospitalized patients with at least 1 risk factor should be considered for pharmacologic prophylaxis. In general, the risk of hospital‐acquired VTE greatly exceeds the risk of bleeding with prophylactic doses of anticoagulation. A patient's risk of VTE may change, and regular assessment of this risk should be mandated if pharmacologic therapy is not initiated at the time of admission.
Numerous barriers to the optimal use of VTE prophylaxis exist, and hospitals must implement systems changes and multidisciplinary approaches to overcome these barriers. The fourth article in this supplement provides detailed strategies for meeting VTE performance measures and overcoming barriers to the optimal use of prophylaxis.
Venous thromboembolism (VTE) is a common and potentially devastating complication of medical illness and surgical intervention. Among patients discharged from acute‐care hospitals in 2003, more than 12 million (31%) had a moderate or high risk of VTE during hospitalization, including 11% at risk due to surgical procedures and 20% at risk due to medical illnesses.1 The incidence of VTEwhich can present as deep vein thrombosis (DVT) or pulmonary embolism (PE)is rising in hospitalized patients.2 Despite the availability of effective prophylaxis, VTE is the third most common cause of hospital‐related death and the most common preventable cause of hospital mortality.3
The clinical impact of VTE is significant. While total incidence, prevalence, and mortality rates of VTE are elusive, the annual incidence of DVT is thought to be as high as 2 million.4 The most serious complication of DVT is acute PE, which occurs in approximately 600,000 patients per year, one‐third of whom die.5 DVT may also be complicated by recurrent episodes of VTE and postthrombotic sequelae such as chronic venous stasis, venous ulceration, debilitating pain, and intractable edema.6 One prospective cohort study found that 30% of patients who had experienced a first episode of DVT developed recurrent VTE within 8 years, and the incidence of postthrombotic syndrome (PTS) was 23% after just 2 years.6
The economic burden of VTE is substantial, due both to the initial event and to the high rate of hospital readmission. The estimated average cost of DVT management (including initial acute care and 6 months of follow‐up care) is $10,072 per patient, and the corresponding cost for PE is $14,649.7 Hospital readmission occurs in 5% to 14% of patients, more than one‐half of whom are readmitted within 90 days.8 For patients with recurrent DVT or PE, the mean total hospitalization costs of readmission are $11,862 and $14,722, respectively.8
The clinical and economic burden of VTE can be significantly mitigated by the use of effective prophylaxis. Because VTE is difficult to diagnose antemortem, it is easier and safer to prevent with appropriate prophylaxis than to diagnose after it has occurred. Unfortunately, VTE prophylaxis is markedly underused, particularly among high‐risk, hospitalized medical patients who would most benefit from it.9
This article summarizes specific risk factors for VTE and provides guidance in identifying patients who may require thromboprophylaxis. Barriers to optimal VTE prophylaxis in the hospital setting will also be explored.
Methodology
For this article and the ones that follow, relevant literature was identified through a Medline search (January 1980 to December 2008) using the following search terms: venous thromboembolism, pulmonary embolism, deep vein thrombosis, epidemiology, risk factors, prophylaxis, mechanical prophylaxis, diagnosis, treatment, anticoagulants, monitoring, secondary prevention, guideline, adherence, treatment protocol, performance measure, and quality improvement. The bibliographies of all key texts were searched for additional relevant articles. The websites of the American College of Chest Physicians (ACCP), American Society of Clinical Oncology (ASCO), National Comprehensive Cancer Network (NCCN), and Society of Hospital Medicine (SHM) were also searched for annual meeting abstracts, position statements, and other key publications.
Evidence‐based clinical guidelines were identified through a search of the National Guideline Clearinghouse (
Pathogenesis of VTE
Venous thrombosis occurs as a result of at least 1 of 3 underlying factors: alterations in blood flow, vascular endothelial injury, and alterations in the constitution of the blood.10 Each potential underlying factor encompasses a wide range of risk factors and clinical scenarios. Alterations in venous blood flow can include several situations, including venous stasis, venous hypertension, and valvular incompetence. Endothelial injury can arise from shear stress, direct trauma, infection, hypertension, or other sources of endothelial damage. Hypercoagulability from alterations in the constitution of the blood may be due to antithrombin deficiency, cancer, surgery, pregnancy, or other risk factors. The presence of any of these factors indicates an elevated risk of VTE, and the presence of multiple factors further increases risk.10
Risk Factors for VTE
VTE can occur in a wide variety of clinical circumstances. Recognized risk factors for VTE include hospitalization for an acute medical illness, cardiovascular disease, pulmonary disease, major surgery, multiple trauma, obesity, and increasing age.10 Additional factors that place patients at increased risk of VTE (independent of age) include a history of prior VTE, known hypercoagulable states, active cancer, and acute infection.11 Hospital‐acquired risk factors such as immobility, acute illness, or medical interventions may lead to the development of VTE in these patients. Severity of illness must be factored into the risk assessment, and all patients need to be assessed for VTE risk at the time of hospital admission and daily thereafter if pharmacologic therapy is not initiated.
In a review of 1231 consecutive patients treated for acute DVT and/or PE, 96.3% had at least 1 risk factor for VTE, and more than one‐third (39%) had 3 or more risk factors (Table 1).10 The incidence of VTE in hospitalized patients is directly related to the number of risk factors present:10
-
1 risk factor: 11%
-
2 risk factors: 24%
-
3 risk factors: 36%
-
4 risk factors: 50%
-
5 risk factors: > 90%
| Risk Factor | Patients (%) |
|---|---|
| |
| Age 40 years | 88.5 |
| Obesity | 37.8 |
| History of VTE | 26.0 |
| Cancer | 22.3 |
| Bed rest 5 days | 12.0 |
| Major surgery | 11.2 |
| Congestive heart failure | 8.2 |
| Varicose veins | 5.8 |
| Fracture (hip or leg) | 3.7 |
| Estrogen treatment | 2.0 |
| Stroke | 1.8 |
| Multiple trauma | 1.1 |
| Childbirth | 1.1 |
| Myocardial infarction | 0.7 |
Current or Recent Prior Hospitalization
The risk of VTE is elevated among hospitalized patients. The prevalence of DVT varies across hospital specialties, reaching up to 80% in major trauma, spinal cord injury, and critical care.3 The Epidemiologic International Day for the Evaluation of Patients at Risk for Venous Thromboembolism in the Acute Hospital Care Setting (ENDORSE) study evaluated the prevalence of VTE risk factors in the acute hospital care setting.12 Among the 68,183 patients enrolled, more than one‐half (52%) were judged to be at risk of VTE. In a case‐control study examining 625 patients with a first lifetime VTE, confinement to a hospital (among other risk factors) was found to be an independent and important predictor of VTE (odds ratio [OR], 8.0; 95% confidence interval [CI], 4.514.2) (Figure 1).13 Recent hospitalization is also an important risk factor for VTE, and patients who are readmitted to the hospital should be considered moderate or high risk.13

Age
Patient age must be considered when assessing VTE risk. VTE is predominantly a disease of older age, and age older than 75 years is an important risk factor for the condition.11 In general, patients older than 40 years have a significantly increased risk compared with younger patients, and the risk approximately doubles with each additional decade.10 Given the aging population, the prevalence of VTE and its complications are expected to increase.
Women of childbearing age experience VTE more frequently than men of the same age, due to pregnancy and exposure to contraceptive therapy.14 This risk, however, is modest compared with the risk among older patients. After age 45 years, the incidence of VTE increases markedly for both sexes, becoming more prominent in men.14 Compared with women, men also have an increased risk of recurrent VTE.15
Despite the effect of age on VTE risk, the risk among patients younger than 40 years may be underestimated because this subgroup has not been extensively studied. For reasons that are not well‐understood, the risk of VTE associated with heart failure is higher in patients younger than 40 years, and the relative risk of PE in patients with chronic obstructive pulmonary disease (COPD) is also higher in younger patients.16, 17
Cancer and Its Treatment
Cancer patients, on average, have twice the risk of VTE compared with noncancer patients.18 This risk, however, varies considerably by cancer type. According to an assessment of nearly 41 million hospitalized patients in the National Hospital Discharge Survey (NHDS), the relative risk of VTE varied from 1.02 in patients with bladder cancer to 4.34 in patients with cancer of the pancreas.18
VTE is one of the most common complications of cancer and cancer therapy, and it is the second leading cause of death among hospitalized cancer patients.19 Molecular mechanisms underlying thromboembolic events in cancer patients include tumor cell procoagulants, inflammatory cell cytokines, mediators of platelet adhesion, and tumor‐related stasis and endothelial damage.20 The clinical implications of these processes are severe. Cancer exacerbates the natural course of VTE, increasing the risk of recurrent VTE and major bleeding, and VTE worsens the prognosis of cancer, increasing the risk of death among cancer patients.
Various cancer therapiesincluding surgery, chemotherapy, hematopoietic stem cell transplantation, and even growth factor supportalso increase the risk of VTE, in part because extrinsic factors such as surgery or chemotherapy can intensify the hypercoagulable process.18, 2123 In the NHDS, cancer patients undergoing surgery had at least twice the risk of postoperative DVT and more than 3 times the risk of PE compared with noncancer patients undergoing similar procedures.18
Cancer is an independent predictor of thromboprophylaxis failure following surgery. The @RISTOS Project found that VTE was the most common cause of death among 2373 patients undergoing general, urologic, or gynecologic surgery for cancer.24 A multivariate analysis identified 5 independent risk factors for VTE after cancer surgery:
-
Previous VTE (OR, 5.98; 95% CI, 2.1316.80)
-
Anesthesia 2 hours (OR, 4.50; 95% CI, 1.0619.04)
-
Bed rest 4 days (OR, 4.37; 95% CI, 2.457.78)
-
Age 60 years (OR, 2.63; 95% CI, 1.215.71)
-
Advanced‐stage cancer (OR, 2.68; 95% CI, 1.375.24)
Cardiovascular Disease
The risk of VTE is pronounced among patients with cardiovascular disease. After stroke and coronary disease, VTE is the third most common cardiovascular disorder, and PE causes more deaths each year than myocardial infarction (MI).25 Several cardiovascular diseases, including hypertension, stroke, acute MI, and heart failure, are independently associated with VTE.10, 2627 Related disorders, including diabetes and the metabolic syndrome, also increase the risk of VTE.26, 28
Congestive heart failure (CHF) is a risk factor for VTE, and the severity of illness increases risk. In the DVT‐Free Prospective Registry, 13% of patients with ultrasound‐confirmed DVT had CHF.29 In a subgroup analysis of patients of the Prophylaxis in Medical Patients with Enoxaparin (MEDENOX) study, the incidence of VTE exceeded 20% in patients with New York Heart Association (NYHA) class IV heart failure, compared with 12% in patients with NYHA class III heart failure.30 Another study found that VTE risk increases as left ventricular ejection fraction (LVEF) decreases, with an LVEF of less than 20% associated with a VTE OR of 38.3 (95% CI, 9.6152.5).31
Infectious Disease
Acute infection may increase the relative risk of VTE by as much as 50% and is associated with VTE event rates of up to 26%.11 Acute infections may be associated with acute inflammation, adverse effects on cardiac or pulmonary function, and prolonged immobilization.30, 32, 33 Human immunodeficiency virus (HIV) patients may also have an increased risk of VTE due to a circulating lupus anticoagulant and/or the presence of acute infection.34
Obesity
The 2008 ACCP guideline update recognizes obesity, for the first time, as a risk factor for VTE.3 Obesity was 1 of the 5 most frequent comorbidities found in patients with DVT in the DVT‐Free Prospective Registry.29 It increases the risk of both incident and recurrent VTE, with every 1‐point increase in body mass index (BMI) increasing the risk of recurrent VTE by 4.4% (95% CI, 1.37.6%; P < 0.001).35
Pregnancy and Puerperium
Pregnancy, particularly the postpartum period, is associated with an increased risk of VTE in women, even though the absolute risk is small.36 Still, PE is one of the leading causes of maternal death following childbirth.10 Smoking, prior VTE, and inherited thrombophilias all increase the risk of VTE in pregnant women.10 The risk begins to rise in the first trimester, and when prophylaxis is needed, it should be started early in gestation.37
Pulmonary Disease
COPD is another risk factor for the development of VTE. COPD patients who develop VTE tend to be older, hospitalized in the intensive care unit (ICU), and on mechanical ventilation.38 In the DVT‐Free Prospective Registry, 12% of patients with ultrasound‐confirmed DVT had COPD.29
Trauma and Surgery
Injury to the body tissue, via trauma or surgery, stimulates the body's clotting mechanism and increases the risk of thromboembolic complications. During the perioperative period, the circulatory system must balance a variety of assaults: an immune response to surgical stress, prolonged immobilization during surgery and recovery, vasodilation associated with general or regional anesthesia, and hypercoagulability due to venous stasis and vascular injury.39 Renal transplant recipients have an increased risk of VTE due to a chronic hypercoagulable state.40 In surgery patients, perioperative complications such as dehydration and acute infection increase the risk of VTE beyond the risk associated with the surgical procedure itself.10
VTE risk is increased approximately 13‐fold by recent major trauma or lower‐extremity injury.13 In the absence of prophylaxis, the overall risk of VTE among patients undergoing major surgery is increased nearly 22‐fold.13 After controlling for the type of surgery, additional independent risk factors for VTE within 3 months of major surgery include:41, 42
-
Obesity
-
Central venous catheter placement
-
Malignancy
-
Smoking
-
Heart failure
-
Previous DVT
-
Prolonged immobility
-
Infection
Many surgical and medical inpatients share common risk factors, and without prophylaxis, the incidence of hospital‐acquired DVT ranges from 10% to 40% for both groups.3
Inherited or Acquired Risk Factors
VTE is a multifactorial disease, and recent evidence indicates that some heritable traits may be potent risk factors for VTE.43 Approximately 35% of patients with DVT will have at least 1 of 5 traits related to an inherited blood clotting disorder:43
-
Deficiencies in the anticoagulation factors protein C, protein S, or antithrombin, or
-
Mutations in the factor V and prothrombin genes, resulting in Factor V Leiden and prothrombin G20210A, respectively.
Certain inherited traits and genetic polymorphisms increase the risk of VTE by interacting with clinical risk factors such as contraceptive use, pregnancy, surgery, trauma, and cancer. One recent study found that oral estrogen therapy among women with the CYP3A5*1 allele was associated with a particularly high risk of VTE.44 Although widespread screening for inherited risk factors is not currently practical, future tools may incorporate genetic polymorphisms to more precisely identify patients who would benefit from aggressive prophylaxis.
Lifestyle Factors
Lifestyle factors have a significant effect on VTE risk. Smoking increases the risk of VTE by 20% to 30%, and a sedentary lifestyle also increases the risk of VTE.26, 45 In fact, women who exercise regularly and consume alcohol in moderation have one‐half the risk of VTE as women who have a sedentary lifestyle and drink little or no alcohol.42 For both men and women, a diet high in fruits, vegetables, and fish is associated with a lower lifetime risk of VTE.46
Medications
Medications may also increase the risk of VTE. In cancer patients with anemia, for example, the use of erythropoiesis‐stimulating agents such as recombinant erythropoietin and darbepoetin was recently shown to increase the risk of VTE by 57% (95% CI, 3187%) and increase mortality risk by 10% (95% CI, 120%).23 In addition, combination hormone replacement therapy in women is associated with a higher risk of VTE compared with estrogen monotherapy, and transdermal contraceptive systems more than double the risk of VTE compared with oral contraceptives (95% CI, 1.33.8).47, 48 Recent studies have also reported an increased risk of VTE with some psychiatric drugs, including amitriptyline, clozapine, olanzapine, and risperidone.4952
Thromboprophylaxis in the Hospital Setting
Despite the prevalence of risk factors and compelling evidence regarding the value of prophylaxis, VTE prophylaxis is suboptimal in hospitalized medical and surgical patients. In a study of 123,304 hospitalized patients who were determined to be at risk of VTE, only 13.3% received prophylaxis in accordance with ACCP guidelines.53 Compliance ranged from a high of 52.4% among patients undergoing orthopedic surgery to a low of 2.8% among patients undergoing neurosurgery.53 Results from several other large trials echo these findings (Table 2).12, 5456
| Trial | Patient Type | Total Patients | Patients at Risk of VTE (Based on ACCP Criteria) (%) | At‐Risk Patients Receiving Recommended Prophylaxis | |
|---|---|---|---|---|---|
| Medical Patients (%) | Surgical Patients (%) | ||||
| |||||
| IMPROVE | Medical patients | 15,156 | 52 | 61 | n/a |
| ENDORSE | Medical and surgical patients | 68,183 | 51.8 | 39.5 | 58.5 |
| ADHERE | Hospitalized heart failure patients | 155,073 | 46 | 30.6 | n/a |
| Amin et al.56 (2008) | Medical and surgical patients | 258,556 | 26.4 | 9.8 | 17.9 |
Reasons for Inadequate Prophylaxis
Researchers have identified a range of barriers to adequate VTE prophylaxis (Table 3).57 Some of these barriers are outlined below.
|
| Variability in clinician knowledge of risk assessment and appropriate prophylaxis |
| Lack of agreement with, and inconsistency between, guidelines in certain patient populations |
| Perceived lack of need |
| Concerns about adverse effects |
| Lack of hospital support systems and policies |
| Lack of established responsibilities for prophylaxis |
Underestimation of Risk of Clotting
VTE is often clinically silent, leading some physicians to mistakenly believe that it is rare.58 In hospitalized surgical patients, for example, the incidence of thromboembolic complications during a short postoperative stay may be low. Given that many cases of symptomatic VTE occur after hospital discharge, hospitalists and surgeons may be unaware of the true incidence of DVT.59
Overestimation of the Risk of Bleeding
Physicians may also overestimate the risk of possible side effects of prophylaxis, such as major bleeding or heparin‐induced thrombocytopenia (HIT).58 Fear of excess bleeding has been cited by physicians as a leading reason for their decision to withhold thromboprophylaxis from at‐risk hospitalized patients.60 Physicians are particularly fearful of complications among elderly patients, who are less likely to receive adequate prophylaxis than younger patients with a similar risk of VTE.61 When bleeding does occur, it rarely results in death. On the other hand, PE may account for as many as 10% of hospital deaths.9
Guideline Confusion and Complexity
Discrepancies between guidelines published by different medical societies contribute to confusion in choosing a management approach. The American Academy of Orthopedic Surgeons (AAOS), for example, describes aspirin alone as a reasonable choice for VTE prophylaxis in some patients, but the ACCP guidelines advise against the use of aspirin monotherapy.58 The cumbersome nature of multiple risk‐assessment and treatment algorithms can also be problematic.61 Furthermore, certain patient subgroups, such as those with cirrhosis, severe renal failure, and epidural catheters, have been excluded from randomized controlled trials, and the management of such patients is not straightforward.
Absence of Institutional Protocols and Information Technology Support
The lack of institution‐level guidance and support can have a detrimental effect on patient care. In a 2007 survey of 127 community hospitals, the prevalence of institutional protocols related to VTE was low: only 60% had protocols to encourage prophylaxis in at‐risk patients, 54% had guidelines to assist in appropriate drug selection, and 43% had guidelines for the dosing of prophylaxis regimens.62 A lack of systems for data collection and audit has also been identified as a barrier to the implementation of prophylaxis guidelines.57 Thus, hospitals need to adopt protocols such as:3
-
Written, institution‐wide thromboprophylaxis policies
-
Preprinted order forms and computer decision‐support systems
-
Policies specifying responsibilities for assessing VTE risk and prescribing prophylaxis
Conclusions
VTE is the most common preventable cause of hospital death, and prophylaxis is underused in hospitalized patients. Although VTE risk factors are numerous and complex, deciding whether to use prophylaxis need not be complicated. In general, elderly patients, medically‐ill patients, and patients undergoing surgery will benefit from prophylaxis, as well as those who are hospitalized for more than 1 night. Hospitalized patients with at least 1 risk factor should be considered for pharmacologic prophylaxis. In general, the risk of hospital‐acquired VTE greatly exceeds the risk of bleeding with prophylactic doses of anticoagulation. A patient's risk of VTE may change, and regular assessment of this risk should be mandated if pharmacologic therapy is not initiated at the time of admission.
Numerous barriers to the optimal use of VTE prophylaxis exist, and hospitals must implement systems changes and multidisciplinary approaches to overcome these barriers. The fourth article in this supplement provides detailed strategies for meeting VTE performance measures and overcoming barriers to the optimal use of prophylaxis.
- , , , , .Estimated annual numbers of US acute‐care hospital patients at risk for venous thromboembolism.Am J Hematol.2007;82(9):777–782.
- , , .Trends in the incidence of pulmonary embolism and deep venous thrombosis in hospitalized patients.Am J Cardiol.2005;95(12):1525–1526.
- , , , et al.Prevention of venous thromboembolism: American College of Chest Physicians evidence‐based clinical practice guidelines. 8th ed.Chest.2008;133(6 suppl):381S–453S.
- , .Management of deep vein thrombosis and pulmonary embolism. A statement for healthcare professionals. Council on Thrombosis (in consultation with the Council on Cardiovascular Radiology), American Heart Association.Circulation.1996;93(12)2212–2245.
- , , , et al.A population‐based perspective of the hospital incidence and case‐fatality rates of deep vein thrombosis and pulmonary embolism. The Worcester DVT Study.Arch Intern Med.1991;151(5):933–938.
- , , , et al.The long‐term clinical course of acute deep venous thrombosis.Ann Intern Med.1996;125(1):1–7.
- , .Direct medical cost of managing deep vein thrombosis according to the occurrence of complications.Pharmacoeconomics.2002;20(9):603–615.
- , .Direct medical costs of venous thromboembolism and subsequent hospital readmission rates: an administrative claims analysis from 30 managed care organizations.J Manag Care Pharm.2007;13(6):475–486.
- , , .Double trouble for 2,609 hospitalized medical patients who developed deep vein thrombosis: prophylaxis omitted more often and pulmonary embolism more frequent.Chest.2007;132(2):554–561.
- , .Risk factors for venous thromboembolism.Circulation.2003;107(23 suppl 1):I9–I16.
- , , , et al.Risk factors for VTE in hospitalized patients with acute medical illness: analysis of the MEDENOX Study.Arch Intern Med.2004;164(9):963–968.
- , , , et al.;ENDORSE Investigators.Venous thromboembolism risk and prophylaxis in the acute hospital care setting (ENDORSE study): a multinational cross‐sectional study.Lancet.2008;371(9610):387–394.
- , , , et al.Risk factors for deep vein thrombosis and pulmonary embolism: a population‐based case‐control study.Arch Intern Med.2000;160(6):809–815.
- , .Range of published estimates of venous thromboembolism incidence in young women.Contraception.2007;75(5):328–336.
- , , , , , .The risk of recurrent venous thromboembolism in men and women.N Engl J Med.2004;350(25):2558–2563.
- , , , et al.Risk of venous thromboembolism in patients hospitalized with heart failure.Am J Cardiol.2006;98(6):793–795.
- , , , et al.Pulmonary embolism and deep venous thrombosis in hospitalized adults with chronic obstructive pulmonary disease.J Cardiovasc Med.2007;8(4):253–257.
- , , , et al.Incidence of venous thromboembolism in patients hospitalized with cancer.Am J Med.2006;119(1):60–68.
- , , , et al.Causes of death in cancer patients.J Med.1975;6(1):61–64.
- , .Tissue factor and angiogenesis in cancer.Curr Opin Hematol.2002;9(5):401–406.
- , , , et al.Incidence of venous thromboembolism in patients with ovarian cancer undergoing platinum/paclitaxel‐containing first‐line chemotherapy: an exploratory analysis by the Arbeitsgemeinschaft Gynaekologische Onkologie Ovarian Cancer Study Group.J Clin Oncol.2008;26(16):2683–2689.
- , , , , , .The incidence of and risk factors for venous thromboembolism (VTE) and bleeding among 1514 patients undergoing hematopoietic stem cell transplantation: implications for VTE prevention.Blood.2008;112(3):504–510.
- , , , et al.Venous thromboembolism and mortality associated with recombinant erythropoietin and darbepoetin administration for the treatment of cancer‐associated anemia.JAMA.2008;299(8):914–924.
- , , , et al.A clinical outcome‐based prospective study on venous thromboembolism after cancer surgery: the @RISTOS project.Ann Surg.2006;243(1):89–95.
- .Pulmonary embolism in thrombolysis: a clarion call for international collaboration.J Am Coll Cardiol.1992;19:246–247.
- , , , et al.Cardiovascular risk factors and venous thromboembolism: a meta‐analysis.Circulation.2008;117(1):93–102.
- , , , , , .Family history of myocardial infarction is an independent risk factor for venous thromboembolism: the Tromsø study.J Thromb Haemost.2008;6(11):1851–1857.
- , , , et al.Abdominal obesity is essential for the risk of venous thromboembolism in the metabolic syndrome—the Tromsø study.J Thromb Haemost.2009;7(5):739–745.
- , .A prospective registry of 5,451 patients with ultrasound‐confirmed deep vein thrombosis.Am J Cardiol.2004;93(2):259–262.
- , , , et al.Prevention of venous thromboembolism in medical patients with enoxaparin; a subgroup analysis of the MEDENOX study.Blood Coagul Fibrinolysis.2003;14:341–348.
- , , .Congestive heart failure and outpatient risk of venous thromboembolism: a retrospective, case‐control study.J Clin Epidemiol.2001;54(8):810–816.
- , , , et al;ARTEMIS Investigators.Efficacy and safety of fondaparinux for the prevention of venous thromboembolism in older acute medical patients: randomised placebo controlled trial.BMJ.2006;332(7537):325–329.
- .Randomised, controlled trial of low‐dose heparin for prevention of fatal pulmonary embolism in patients with infectious diseases. The Heparin Prophylaxis Study Group.Lancet.1996;347(9012):1357–1361.
- , , , et al.HIV infection is a risk factor for venous thromboembolism.AIDS Patient Care STDS.2002;16(5):205–209.
- , , , et al.Overweight, obesity, and the risk of recurrent venous thromboembolism.Arch Intern Med.2008;168(15):1678–1683.
- .Venous thromboembolism in women.Vasc Med.2008;13(3):255–266.
- , , .Thrombosis during pregnancy and the postpartum period.Am J Obstet Gynecol.2005;193(1):216–219.
- , , , et al.Chronic obstructive pulmonary disease and deep vein thrombosis: a prevalent combination.J Thromb Thrombolysis.2008;26(1):35–40.
- , , .Venous thromboembolism in trauma: a local manifestation of systemic hypercoagulability?J Trauma.2003;54(2):224–231.
- , .Acquired hypercoagulable state in renal transplant recipients.Thromb Haemost.2004;91(4):646–654.
- , , , .Venous thromboembolism in patients undergoing surgery: low rates of prophylaxis and high rates of filter insertion.Thromb Haemost.2007;98(6):1220–1225.
- , , , , , .A 10‐year analysis of venous thromboembolism on the surgical service: the effect of practice guidelines for prophylaxis.Surgery.2008;144(1):3–11.
- , , , et al;EDITH Collaborative Study Group.Family history as a risk factor for venous thromboembolism.Thromb Res.2008;122(5):624–629.
- , , , et al;Estrogen and Thromboembolism Risk (ESTHER) Study Group.Synergism between oral estrogen therapy and cytochrome P450 3A5*1 allele on the risk of venous thromboembolism among postmenopausal women.J Clin Endocrinol Metab.2008;93(8):3082–3087.
- , , .The relationship between lifestyle factors and venous thromboembolism among women: a report from the MISS study.Br J Haematol.2009;144(2):234–240.
- , , , , .Greater fish, fruit, and vegetable intakes are related to lower incidence of venous thromboembolism: the Longitudinal Investigation of Thromboembolism Etiology.Circulation.2007;115(2):188–195.
- , , .Association between hormone replacement therapy and subsequent arterial and venous vascular events: a meta‐analysis.Eur Heart J.2008;29(16):2031–2041.
- , , , .Venous thromboembolism, myocardial infarction, and stroke among transdermal contraceptive system users.Obstet Gynecol.2007;109(2 Pt 1):339–346.
- , , , , , .Pulmonary thromboembolism associated with olanzapine and risperidone.J Emerg Med.2008;35(2):159–161.
- , , , .Associations between venous thromboembolism and antipsychotics. A study of the WHO database of adverse drug reactions.Drug Saf.2008;31(8):685–694.
- .Evaluating the association between clozapine and venous thromboembolism.Am J Health Syst Pharm.2008;65(19):1825–1829.
- , .Antidepressant drug use and risk of venous thromboembolism.Pharmacotherapy.2008;28(2):144–150.
- , , , .Hospitals' compliance with prophylaxis guidelines for venous thromboembolism.Am J Health Syst Pharm.2007;64(1):69–76.
- , , , et al.Venous thromboembolism prophylaxis in acutely ill hospitalized medical patients.Chest.2007;132:936–945.
- , , , et al.Adhere Scientific Advisory Committee and Investigators. Venous thromboembolism prophylaxis in hospitalized heart failure patients.J Card Fail.2008;14(2):127–132.
- , , , et al. Are hospitals following guidelines for VTE Prevention? The Venous Thromboembolism Study to Assess the Rate of Thromboprophylaxis. Presented at the 50th Annual Meeting of the American College of Hematology, San Francisco, CA, December 6–9, 2008. Abstract 1286.
- , , , et al.A systematic review of strategies to improve prophylaxis for venous thromboembolism in hospitals.Ann Surg.2005;241(3):397–415.
- .A need for a simplified approach to venous thromboembolism prophylaxis in acute medical inpatients.Int J Clin Pract.2007;61(2):336–340.
- .Barriers to the optimal use of anticoagulants after orthopaedic surgery.Arch Orthop Trauma Surg.2008 (Published online Oct, 8.). [http://dx.doi.org/DOI 10.1007/s00402‐008‐0765‐9]
- , , , , .Venous thrombosis in cancer patients: Insights from the FRONTLINE survey.Oncologist.2003;8:381–388.
- , , , et al.Incorrect use of thromboprophylaxis for venous thromboembolism in medical and surgical patients: results of a multicentric, observational and cross‐sectional study in Brazil.J Thromb Haemost.2006;4(6):1266–1270.
- , , , , .Survey of hospitals for guidelines, policies, and protocols for anticoagulants.Am J Health Syst Pharm.2007;64(11):1203–1208.
- , , , , .Estimated annual numbers of US acute‐care hospital patients at risk for venous thromboembolism.Am J Hematol.2007;82(9):777–782.
- , , .Trends in the incidence of pulmonary embolism and deep venous thrombosis in hospitalized patients.Am J Cardiol.2005;95(12):1525–1526.
- , , , et al.Prevention of venous thromboembolism: American College of Chest Physicians evidence‐based clinical practice guidelines. 8th ed.Chest.2008;133(6 suppl):381S–453S.
- , .Management of deep vein thrombosis and pulmonary embolism. A statement for healthcare professionals. Council on Thrombosis (in consultation with the Council on Cardiovascular Radiology), American Heart Association.Circulation.1996;93(12)2212–2245.
- , , , et al.A population‐based perspective of the hospital incidence and case‐fatality rates of deep vein thrombosis and pulmonary embolism. The Worcester DVT Study.Arch Intern Med.1991;151(5):933–938.
- , , , et al.The long‐term clinical course of acute deep venous thrombosis.Ann Intern Med.1996;125(1):1–7.
- , .Direct medical cost of managing deep vein thrombosis according to the occurrence of complications.Pharmacoeconomics.2002;20(9):603–615.
- , .Direct medical costs of venous thromboembolism and subsequent hospital readmission rates: an administrative claims analysis from 30 managed care organizations.J Manag Care Pharm.2007;13(6):475–486.
- , , .Double trouble for 2,609 hospitalized medical patients who developed deep vein thrombosis: prophylaxis omitted more often and pulmonary embolism more frequent.Chest.2007;132(2):554–561.
- , .Risk factors for venous thromboembolism.Circulation.2003;107(23 suppl 1):I9–I16.
- , , , et al.Risk factors for VTE in hospitalized patients with acute medical illness: analysis of the MEDENOX Study.Arch Intern Med.2004;164(9):963–968.
- , , , et al.;ENDORSE Investigators.Venous thromboembolism risk and prophylaxis in the acute hospital care setting (ENDORSE study): a multinational cross‐sectional study.Lancet.2008;371(9610):387–394.
- , , , et al.Risk factors for deep vein thrombosis and pulmonary embolism: a population‐based case‐control study.Arch Intern Med.2000;160(6):809–815.
- , .Range of published estimates of venous thromboembolism incidence in young women.Contraception.2007;75(5):328–336.
- , , , , , .The risk of recurrent venous thromboembolism in men and women.N Engl J Med.2004;350(25):2558–2563.
- , , , et al.Risk of venous thromboembolism in patients hospitalized with heart failure.Am J Cardiol.2006;98(6):793–795.
- , , , et al.Pulmonary embolism and deep venous thrombosis in hospitalized adults with chronic obstructive pulmonary disease.J Cardiovasc Med.2007;8(4):253–257.
- , , , et al.Incidence of venous thromboembolism in patients hospitalized with cancer.Am J Med.2006;119(1):60–68.
- , , , et al.Causes of death in cancer patients.J Med.1975;6(1):61–64.
- , .Tissue factor and angiogenesis in cancer.Curr Opin Hematol.2002;9(5):401–406.
- , , , et al.Incidence of venous thromboembolism in patients with ovarian cancer undergoing platinum/paclitaxel‐containing first‐line chemotherapy: an exploratory analysis by the Arbeitsgemeinschaft Gynaekologische Onkologie Ovarian Cancer Study Group.J Clin Oncol.2008;26(16):2683–2689.
- , , , , , .The incidence of and risk factors for venous thromboembolism (VTE) and bleeding among 1514 patients undergoing hematopoietic stem cell transplantation: implications for VTE prevention.Blood.2008;112(3):504–510.
- , , , et al.Venous thromboembolism and mortality associated with recombinant erythropoietin and darbepoetin administration for the treatment of cancer‐associated anemia.JAMA.2008;299(8):914–924.
- , , , et al.A clinical outcome‐based prospective study on venous thromboembolism after cancer surgery: the @RISTOS project.Ann Surg.2006;243(1):89–95.
- .Pulmonary embolism in thrombolysis: a clarion call for international collaboration.J Am Coll Cardiol.1992;19:246–247.
- , , , et al.Cardiovascular risk factors and venous thromboembolism: a meta‐analysis.Circulation.2008;117(1):93–102.
- , , , , , .Family history of myocardial infarction is an independent risk factor for venous thromboembolism: the Tromsø study.J Thromb Haemost.2008;6(11):1851–1857.
- , , , et al.Abdominal obesity is essential for the risk of venous thromboembolism in the metabolic syndrome—the Tromsø study.J Thromb Haemost.2009;7(5):739–745.
- , .A prospective registry of 5,451 patients with ultrasound‐confirmed deep vein thrombosis.Am J Cardiol.2004;93(2):259–262.
- , , , et al.Prevention of venous thromboembolism in medical patients with enoxaparin; a subgroup analysis of the MEDENOX study.Blood Coagul Fibrinolysis.2003;14:341–348.
- , , .Congestive heart failure and outpatient risk of venous thromboembolism: a retrospective, case‐control study.J Clin Epidemiol.2001;54(8):810–816.
- , , , et al;ARTEMIS Investigators.Efficacy and safety of fondaparinux for the prevention of venous thromboembolism in older acute medical patients: randomised placebo controlled trial.BMJ.2006;332(7537):325–329.
- .Randomised, controlled trial of low‐dose heparin for prevention of fatal pulmonary embolism in patients with infectious diseases. The Heparin Prophylaxis Study Group.Lancet.1996;347(9012):1357–1361.
- , , , et al.HIV infection is a risk factor for venous thromboembolism.AIDS Patient Care STDS.2002;16(5):205–209.
- , , , et al.Overweight, obesity, and the risk of recurrent venous thromboembolism.Arch Intern Med.2008;168(15):1678–1683.
- .Venous thromboembolism in women.Vasc Med.2008;13(3):255–266.
- , , .Thrombosis during pregnancy and the postpartum period.Am J Obstet Gynecol.2005;193(1):216–219.
- , , , et al.Chronic obstructive pulmonary disease and deep vein thrombosis: a prevalent combination.J Thromb Thrombolysis.2008;26(1):35–40.
- , , .Venous thromboembolism in trauma: a local manifestation of systemic hypercoagulability?J Trauma.2003;54(2):224–231.
- , .Acquired hypercoagulable state in renal transplant recipients.Thromb Haemost.2004;91(4):646–654.
- , , , .Venous thromboembolism in patients undergoing surgery: low rates of prophylaxis and high rates of filter insertion.Thromb Haemost.2007;98(6):1220–1225.
- , , , , , .A 10‐year analysis of venous thromboembolism on the surgical service: the effect of practice guidelines for prophylaxis.Surgery.2008;144(1):3–11.
- , , , et al;EDITH Collaborative Study Group.Family history as a risk factor for venous thromboembolism.Thromb Res.2008;122(5):624–629.
- , , , et al;Estrogen and Thromboembolism Risk (ESTHER) Study Group.Synergism between oral estrogen therapy and cytochrome P450 3A5*1 allele on the risk of venous thromboembolism among postmenopausal women.J Clin Endocrinol Metab.2008;93(8):3082–3087.
- , , .The relationship between lifestyle factors and venous thromboembolism among women: a report from the MISS study.Br J Haematol.2009;144(2):234–240.
- , , , , .Greater fish, fruit, and vegetable intakes are related to lower incidence of venous thromboembolism: the Longitudinal Investigation of Thromboembolism Etiology.Circulation.2007;115(2):188–195.
- , , .Association between hormone replacement therapy and subsequent arterial and venous vascular events: a meta‐analysis.Eur Heart J.2008;29(16):2031–2041.
- , , , .Venous thromboembolism, myocardial infarction, and stroke among transdermal contraceptive system users.Obstet Gynecol.2007;109(2 Pt 1):339–346.
- , , , , , .Pulmonary thromboembolism associated with olanzapine and risperidone.J Emerg Med.2008;35(2):159–161.
- , , , .Associations between venous thromboembolism and antipsychotics. A study of the WHO database of adverse drug reactions.Drug Saf.2008;31(8):685–694.
- .Evaluating the association between clozapine and venous thromboembolism.Am J Health Syst Pharm.2008;65(19):1825–1829.
- , .Antidepressant drug use and risk of venous thromboembolism.Pharmacotherapy.2008;28(2):144–150.
- , , , .Hospitals' compliance with prophylaxis guidelines for venous thromboembolism.Am J Health Syst Pharm.2007;64(1):69–76.
- , , , et al.Venous thromboembolism prophylaxis in acutely ill hospitalized medical patients.Chest.2007;132:936–945.
- , , , et al.Adhere Scientific Advisory Committee and Investigators. Venous thromboembolism prophylaxis in hospitalized heart failure patients.J Card Fail.2008;14(2):127–132.
- , , , et al. Are hospitals following guidelines for VTE Prevention? The Venous Thromboembolism Study to Assess the Rate of Thromboprophylaxis. Presented at the 50th Annual Meeting of the American College of Hematology, San Francisco, CA, December 6–9, 2008. Abstract 1286.
- , , , et al.A systematic review of strategies to improve prophylaxis for venous thromboembolism in hospitals.Ann Surg.2005;241(3):397–415.
- .A need for a simplified approach to venous thromboembolism prophylaxis in acute medical inpatients.Int J Clin Pract.2007;61(2):336–340.
- .Barriers to the optimal use of anticoagulants after orthopaedic surgery.Arch Orthop Trauma Surg.2008 (Published online Oct, 8.). [http://dx.doi.org/DOI 10.1007/s00402‐008‐0765‐9]
- , , , , .Venous thrombosis in cancer patients: Insights from the FRONTLINE survey.Oncologist.2003;8:381–388.
- , , , et al.Incorrect use of thromboprophylaxis for venous thromboembolism in medical and surgical patients: results of a multicentric, observational and cross‐sectional study in Brazil.J Thromb Haemost.2006;4(6):1266–1270.
- , , , , .Survey of hospitals for guidelines, policies, and protocols for anticoagulants.Am J Health Syst Pharm.2007;64(11):1203–1208.
Guidelines‐Based Thromboprophylaxis
Thromboprophylaxis with anticoagulants has proven benefits in hospitalized patients. Despite this, venous thromboembolism (VTE) prophylaxis is underused and VTE remains the leading cause of preventable hospital mortality.1 Medical patients have a particularly high risk; those who develop a deep vein thrombosis (DVT) are significantly less likely to have received prophylaxis prior to the diagnosis of DVT than nonmedical patients. Even within the high‐risk setting of the intensive care unit (ICU), medical patients receive thromboprophylaxis only two‐thirds as often as nonmedical patients.2
In this article we summarize the evidence concerning the various prophylaxis options, including current guideline recommendations for VTE prevention in medical and surgical patients. We also discuss strategies for thromboprophylaxis in special populations and potential complications of prophylaxis.
Efficacy of Prophylaxis in Medical Patients
Several meta‐analyses have demonstrated the marked benefits of anticoagulant prophylaxis in medical patients. Dentali et al3 conducted a meta‐analysis of 9 randomized controlled trials enrolling a total of 19,958 at‐risk hospitalized medical patients. The selected trials compared standard anticoagulant regimens with no treatment and only included studies with objectively documented and independently adjudicated outcomes. Compared with patients receiving placebo, those receiving thromboprophylaxis had significant reductions in any PE by 57% (95% CI, 0.26‐0.71; absolute risk reduction, 0.29%) and fatal pulmonary embolism (PE) by 62% (95% CI, 0.21‐0.69; absolute risk reduction, 0.25%), with a nonsignificant reduction in symptomatic DVT (relative risk [RR], 0.47; 95% CI, 0.22‐1.00) and a nonsignificant increase in major bleeding (RR, 1.32; 95% CI, 0.73‐2.37). The researchers concluded that anticoagulant prophylaxis is effective in preventing symptomatic VTE in medical patients, though the optimal duration of therapy is not yet defined.3
Another meta‐analysis focusing on subclinical DVT in acutely ill medical patients examined the therapeutic effects of various prophylaxis regimens. Overall, anticoagulant prophylaxis reduced the risk of any asymptomatic DVT (assessed by venogram or ultrasound) by 49% (95% CI, 0.39‐0.67) and asymptomatic proximal DVT by 55% (95% CI, 0.31‐0.65) compared with placebo (absolute risk reduction, 2.6% and 1.8%, respectively). Although prophylaxis was associated with a 0.5% absolute risk increase in major bleeding, the authors concluded that the benefits of prophylaxis outweighed the risks of bleeding.4
Anticoagulant Agents in the Prevention of VTE
Currently available anticoagulants for the prevention of VTE include unfractionated heparin (UFH), low‐molecular‐weight heparins (LMWHs), fondaparinux, and warfarin. These agents interrupt thrombus formation, either indirectly (through interaction with antithrombin) or directly (by inhibiting the action of thrombin). Each class of therapy has advantages and limitations. Table 1 lists common anticoagulant options for VTE prophylaxis, along with dosing information and other important information.510
| Prophylactic Dose | Warnings/Contraindications/Adverse Reactions | |
|---|---|---|
| ||
| Warfarin | 5 to 10 mg daily initially;* adjust dose based on INR; therapeutic INR goal: 2.5 (2‐3) | Warning: bleeding risk; requires frequent monitoring; contraindicated in patients for whom hazard of hemorrhage outweighs potential benefit (eg, in pregnant women) |
| UFH | 5000 IU every 8‐12 hours subcutaneously | Contraindicated in the presence of active bleeding, uncontrolled hypertension, or severe thrombocytopenia; monitor platelet count every 4‐7 days for HIT |
| Dalteparin | 5000 IU daily subcutaneously | Warning: spinal/epidural hematoma; monitor for signs and symptoms of neurological impairment. LMWHs should be used with caution in renal impairment; anti‐Xa monitoring and dose adjustments may be required. Follow prescribing information for dose adjustments and body weight‐based dosing. Most common adverse reactions: bleeding, anemia, thrombocytopenia, elevation of serum aminotransferase, diarrhea, and nausea |
| Enoxaparin | 40 mg daily subcutaneously; reduce to 30 mg daily in renal impairment | |
| Tinzaparin | 3500 IU daily subcutaneously | |
| Fondaparinux | 2.5 mg daily subcutaneously | Warning: spinal/epidural hematoma; monitor for signs and symptoms of neurological impairment; contraindicated in patients with severe renal impairment (creatinine clearance < 30 mL/minute) and in patients < 50 kg |
UFH
UFH, which is typically administered by subcutaneous injection, has the longest history as an anticoagulant in the prevention and treatment of VTE. It is an attractive option in patients with severe renal failure or those who may require a procedure in the near future. Although UFH is partially cleared by the kidney, its short half‐life can be perceived as a safety advantage in patients with severe renal impairment and an increased risk of bleeding. For most other patients, UFH holds several disadvantages compared with newer therapies, including the need for injections to be administered 3 times a day to be optimally effective, its effect on platelets, and its association with heparin‐induced thrombocytopenia (HIT).1 Given the costs of administration and potential complications, it is not less expensive than LMWHs, and it appears to be less cost‐effective.11
LMWHs
LMWHs have a higher bioavailability and longer half‐life than UFH, which translates to reliable anticoagulation levels when given subcutaneously on a weight‐based dosing schedule. Unlike UFH, LMWHs do not require laboratory tests to monitor the intensity of anticoagulation, except in special circumstances.1 The LMWHs dalteparin, enoxaparin, and tinzaparin are widely used for the prevention and treatment of VTE in the United States.
Two landmark clinical trials demonstrated the efficacy of appropriate thromboprophylaxis with LMWHs in reducing the burden of VTE in acutely ill, hospitalized medical patients. The Prophylaxis in Medical Patients with Enoxaparin (MEDENOX) trial and Prospective Evaluation of Dalteparin Efficacy for Prevention of VTE in Immobilized Patients Trial (PREVENT) demonstrated the benefits of enoxaparin and dalteparin, respectively, in reducing the risk of VTE. As shown in Table 2, thromboprophylaxis with these agents was associated with a 45% to 63% relative reduction in the risk of VTE compared with placebo.12, 13
| Trial | Number of Patients | Agent (vs. placebo) | Detection of VTE | Relative Risk Reduction | Number Needed to Treat |
|---|---|---|---|---|---|
| |||||
| MEDENOX | 866 | Enoxaparin 20 mg or 40 mg SC daily for 6‐14 days | Distal and proximal venographic DVT or documented PE | 63% (with 40 mg; 97.6 CI, 0.22‐0.63; P <0.001) | 10 |
| PREVENT | 3706 | Dalteparin 5000 IU SC daily for up to 14 days | CUS DVT, symptomatic VTE, and fatal PE | 45% (95% CI, 0.38‐0.80; P = 0.0015) | 45 |
| ARTEMIS | 849 | Fondaparinux 2.5 mg SC daily for 6‐14 days | Distal and proximal venographic DVT, symptomatic VTE, and fatal PE | 47% (95% CI, 0.077‐0.693) | 20 |
Pentasaccharides
Fondaparinux is a synthetic factor Xa antagonist that shares many features of LMWHs, including a high bioavailability and long half‐life. Fondaparinux does not require monitoring, but it is contraindicated in patients with renal failure (CrCl < 30 mL/minute) and in patients weighing less than 50 kg.1 Although PF4 antibodies have been associated with fondaparinux administration, this drug has not, to date, been associated with HIT.14 The Arixtra for Thromboembolism Prevention in a Medical Indications Study (ARTEMIS) trial demonstrated the advantage of fondaparinux over placebo in reducing the risk of VTE (Table 2).15 The American College of Chest Physicians (ACCP) guidelines state that fondaparinux appears to be as effective and safe as LMWH.1
Vitamin K Antagonists
Vitamin K antagonists (VKAs) such as warfarin inhibit the production of prothrombin, clotting factors VII, IX, and X, and the anticoagulants protein C and protein S. Warfarin is challenging to manage because of its narrow therapeutic window, its tendency to exhibit considerable variability in dose‐response, the time required to reach target international normalized ratio (INR), its potential for interaction with diet and concomitant medications, and its need for ongoing monitoring.5 Warfarin should usually be initiated within the same 24 hours as parenteral anticoagulation, with a goal of achieving INR results between 2.0 and 3.0. An initial dose of 5 to 10 mg for the first 1 or 2 days is appropriate for most patients, and subsequent dosing should be based on INR response.5 Warfarin prophylaxis is primarily used in patients in the US undergoing orthopedic surgery, including total hip replacement and hip and knee arthroplasty.1
Future Anticoagulants
New oral agents have the potential to improve the management of patients who have a moderate to high risk of thromboembolic disease.
Rivaroxaban
This oral factor Xa inhibitor is showing promise in patients undergoing major orthopedic surgery. A prespecified pooled analysis was performed on data from the four Regulation of Coagulation in Orthopedic Surgery to Prevent Deep Vein Thrombosis and Pulmonary Embolism (RECORD) trials to evaluate the effect of rivaroxaban on the composite of symptomatic VTE (DVT or PE) and death, and bleeding. In the analysis, patients undergoing hip or knee arthroplasty had a VTE rate of 0.8% with rivaroxaban vs. 1.6% with enoxaparin, the current gold standard for surgical prophylaxis (P < 0.001). Bleeding rates were not significantly different between treatment arms (P = 0.376).16
Apixaban
This oral, direct, reversible factor Xa inhibitor is under evaluation for the prevention and treatment of VTE. In the Apixaban Prophylaxis in Patients Undergoing Total Knee Replacement Surgery (APROPOS) study of patients undergoing knee replacement, apixaban had a lower composite rate of DVT, PE, and all‐cause mortality when compared with enoxaparin or warfarin.17 In the ADVANCE‐1 study of patients undergoing knee surgery, however, apixaban failed to meet criteria for noninferiority when compared with enoxaparin.18 Apixaban is now being evaluated for VTE prophylaxis in acutely ill medical patients.
Dabigatran
This oral direct thrombin inhibitor reversibly binds to free and fibrin‐bound thrombin. In the RE‐NOVATE trial, dabigatran was noninferior to enoxaparin in reducing the events of DVT, PE, and all‐cause mortality following total hip replacement surgery.19 In a Phase II dose‐ranging trial in patients with atrial fibrillation (Prevention of Embolic and Thrombotic Events in Patients with Persistent [AFPETRO]), dabigatran with or without aspirin was as effective as warfarin in reducing embolic events.20 In the RE‐MODEL study, dabigatran was as effective as enoxaparin in preventing VTE and all‐cause mortality following knee replacement surgery, but failed to show equivalence to a higher dose of enoxaparin in the RE‐MOBILIZE trial.21, 22 It should be noted that in the RE‐MODEL study, enoxaparin was not administered at the dosage recommended by the U.S. Food and Drug Administration (FDA) for knee replacement surgery.
Mechanical Prophylaxis
Mechanical methods of thromboprophylaxis include graduated compression stockings (GCS), intermittent pneumatic compression (IPC) devices, and the venous foot pump (VFP). Mechanical approaches to thromboprophylaxis should be used primarily in patients who have a high risk of bleeding or as an adjunct to pharmacotherapeutic prophylaxis.1 The ACCP guidelines summarize the advantages and limitations of mechanical prophylaxis in patients at risk of developing VTE (Table 3).1
| Advantages | Limitations |
|---|---|
| |
| Does not increase the risk of bleeding | Not as intensively studied as pharmacologic thromboprophylaxis (fewer studies and smaller) |
| Can be used in patients who have a high risk of bleeding | No established standards for size, pressure, or physiologic features |
| Efficacy has been demonstrated in a number of patient groups | Many specific mechanical devices have never been assessed in any clinical trial |
| May enhance the effectiveness of anticoagulant thromboprophylaxis | Almost all mechanical thromboprophylaxis trials were unblinded and therefore have a potential for bias |
| May reduce leg swelling | Are less effective in high‐risk groups than anticoagulant thromboprophylaxis |
| Greater effect in reducing calf DVT than proximal DVT | |
| Effect on PE and death unknown | |
| May reduce or delay the use of more effective anticoagulant thromboprophylaxis | |
| Compliance by patients and staff is often poor | |
| Trials may overestimate the protection compared with routine use | |
| Cost associated with purchase, storage, dispensing, and cleaning of the devices, as well as ensuring optimal compliance | |
When properly fitted, GCS increase venous blood return through external pressure, thereby reducing venous stasis. IPC devices or sequential compression devices are usually applied over compression stockings. In addition to improving venous blood flow, these devices stimulate endogenous fibrinolysis. Compliance is often a problem in medical patients, who may not use the devices properly. Furthermore, for patients with severe vascular insufficiency (ankle brachial index <0.05), IPC may worsen vascular insufficiency and digital gangrene.
Inferior vena cava (IVC) filters are barrier devices that may benefit patients with major bleeding risk in the acute VTE setting by preventing PE. These devices, however, do not prevent DVT and may promote further venous stasis and clotting below the device. Importantly, patients with HIT should not have IVC filters placed due to a very high thrombogenic state that could lead to limb ischemia or cerulea phlegmasia dolens.23
Thromboprophylaxis in Medical Patients
Duration
Although major trials support the use of short‐term prophylaxistypically 6 to 14 daysin‐hospital for acutely ill medical patients, the optimal duration of thromboprophylaxis in these patients is unclear.24 The Extended Clinical Prophylaxis in Acutely Ill Medical Patients (EXCLAIM) trial is the first randomized trial to evaluate the potential benefits of extended prophylaxis in acutely ill medical patients. In this study, 5101 hospitalized patients with varying levels of reduced mobility due to cancer, ischemic stroke, heart failure, respiratory failure, infection, and other acute medical conditions received open‐label enoxaparin 40 mg daily for a mean duration of 10 days. Patients were then randomly assigned to additional therapy with enoxaparin or placebo for a mean duration of 28 additional days.25 Preliminary findings from this trial suggest that high‐risk medical patients can benefit from extended thromboprophylaxis following hospital discharge, with significantly reduced VTE events (RR reduction, 44%; P = 0.0011). The benefits of thromboprophylaxis were apparent during the extended treatment period and persistent through 90 days.26
Guideline Recommendations
Incorrect use of thromboprophylaxis does not stem from a lack of evidence‐based recommendations. Within the past year, the ACCP, the American Society of Clinical Oncology (ASCO), and the National Comprehensive Cancer Network (NCCN) have published updated guidelines for thromboprophylaxis in hospitalized patients at risk of VTE.1, 27, 28 The 2008 ACCP guidelines include more than 700 recommendations for VTE risk assessment and management, to be implemented by a variety of physicians, including pulmonologists, cardiologists, cardiothoracic surgeons, and critical care medicine specialists.1
The ACCP guidelines organize prophylaxis recommendations on the basis of patient risk (Table 4).1 Risk assessment remains relatively subjective, however, and validated risk assessment models are not yet widely available. The prudent approach is to consider thromboprophylaxis for all hospitalized medically ill patients who do not have a specific contraindication.
| Levels of Risk | Approximate DVT Risk Without Thromboprophylaxis (%) | Suggested Thromboprophylaxis Options |
|---|---|---|
| ||
| Low risk | ||
| Minor surgery in mobile patients | < 10 | No specific thromboprophylaxis; early and aggressive ambulation |
| Medical patients who are fully mobile | ||
| Moderate risk | ||
| Most general, open gynecologic, or urologic surgery patients | 10‐40 | LMWH (at recommended doses), low‐dose UFH bid or tid, fondaparinux |
| Medical patients, bed rest or sick | ||
| Moderate VTE risk plus high bleeding risk | Mechanical thromboprophylaxis | |
| High risk | ||
| Hip or knee arthroplasty, HFS | 40‐80 | LMWH (at recommended doses), fondaparinux, oral vitamin K antagonist (INR 2‐3) |
| Major trauma, SCI | ||
| High VTE risk plus high bleeding risk | Mechanical thromboprophylaxis | |
Key evidence‐based recommendations regarding thromboprophylaxis for hospitalized, acutely ill patients include the following:1
-
Every hospital should develop a formal strategy to addresses VTE prophylaxis;
-
Aspirin alone is not recommended to prevent VTE for any patient group;
-
Mechanical methods of thromboprophylaxis should be used primarily for patients who have a high bleeding risk or possibly as an adjunct to anticoagulant thromboprophylaxis;
-
Thromboprophylaxis with LMWH, UFH, or fondaparinux is recommended for patients admitted to hospital with an acute medical illness (Note: fondaparinux is recommended, but not FDA‐approved, for this indication in the United States);
-
On admission to the ICU, all patients should be assessed for risk of VTE, and most should receive thromboprophylaxis;
-
All major trauma and all spinal‐cord injury patients should receive thromboprophylaxis.
Thromboprophylaxis in Surgical Patients
For hospitalized surgical patients, the ACCP guidelines indicate the importance of the type of surgery (eg, gynecologic, urologic, or neurologic) in determining the appropriate prophylaxis strategy. In general, routine thromboprophylaxis is recommended for patients undergoing major general, gynecologic, or orthopedic surgery, as well as bariatric and coronary artery bypass surgery.1 Some specific recommendations regarding thromboprophylaxis for surgical patients include the following:
-
Major general surgery: LMWH, low‐dose UFH, or fondaparinux;
-
Major gynecologic surgery and major open urologic procedures: LMWH, low‐dose UFH, fondaparinux, and/or a mechanical device;
-
Elective hip or knee arthroplasty: Anticoagulant therapy (LMWH, fondaparinux, or a VKA);
-
Hip‐fracture surgery: Fondaparinux, LMWH, a VKA, or low‐dose UFH;
-
Patients undergoing hip or knee arthroplasty or hip‐fracture surgery should receive thromboprophylaxis for a minimum of 10 days; for hip arthroplasty and hip‐fracture surgery, thromboprophylaxis should continue for more than 10 days and up to 35 days.
Although the ACCP guidelines recommend against aspirin monotherapy for any patient group, the American Academy of Orthopaedic Surgeons (AAOS) guidelines state that aspirin alone is an effective option in preventing VTE in standard‐risk patients who are undergoing hip or knee replacement surgery.29 However, evidence for aspirin monotherapy is currently limited.1
The 2008 ACCP guidelines include a new chapter on the perioperative management of patients receiving long‐term antithrombotic treatment who must undergo surgery or other invasive procedures. To minimize surgical bleeding, the ACCP recommends the temporary discontinuation of antithrombotic treatment immediately before and during surgery for most patients. Discontinuing antithrombotics can increase the risk of a thromboembolic event, but this risk must be weighed against the risk of bleeding.1 The guidelines also offer specific recommendations for the use of perioperative bridging therapy in patients receiving VKAs based on the risk of VTE and whether the patient has a mechanical heart valve or atrial fibrillation. Guidelines recommend discontinuing bridging anticoagulation 24 hours prior to surgery if therapeutic subcutaneous LMWH is the agent used and approximately 4 hours prior to surgery if intravenous UFH is the agent used.
Thromboprophylaxis in Special Populations
Care must be taken when using thromboprophylaxis in certain high‐risk populations. The following section provides recommendations regarding prophylaxis in the presence of cancer, pregnancy, renal insufficiency, and epidural anesthesia.
Cancer Patients
The ASCO and NCCN guidelines endorse the use of VTE prophylaxis with anticoagulants in all hospitalized patients with active cancer or suspicion of cancer in the absence of contraindications.27, 28 The ACCP guidelines restrict this recommendation to hospitalized cancer patients who are bedridden.1 Thromboprophylaxis should continue at least through the duration of the hospital stay. Acceptable subcutaneous regimens include fondaparinux, dalteparin, or enoxaparin at the doses presented in Table 1; if UFH is chosen, the dose should be 5000 units every 8 hours.
Cancer patients who are scheduled to undergo major surgery require a different prophylaxis strategy. Even with prophylaxis, cancer patients have a 2‐fold higher risk of postoperative VTE compared with noncancer patients and more than a 3‐fold higher risk of fatal PE.30 To manage this risk, the ASCO, NCCN, and ACCP guidelines recommend extended prophylaxis in patients undergoing major cancer surgery.1, 27, 28 Specific recommendations include the following:
-
All patients undergoing major surgical intervention for malignant disease should be considered for VTE prophylaxis with anticoagulants, with or without mechanical prophylaxis;
-
Thromboprophylaxis should be initiated prior to the start of surgery or as early as possible following surgery;
-
Mechanical interventions may supplement pharmacologic prophylaxis, especially in patients who have the highest risk;
-
Prophylaxis with a LMWH should be initiated 12 to 24 hours after the surgical procedure;
-
Continue prophylaxis at least 7 to 10 days postoperatively;
-
Consider prolonged prophylaxis (ie, up to 4 weeks) with a LMWH for high‐risk patients (eg, patients undergoing major abdominal or pelvic surgery, those with residual malignant disease after surgery, obese patients, and patients with a history of VTE).
Routine prophylaxis with anticoagulants is not recommended for most outpatients, except for those with high‐risk factors (eg, thrombogenic chemotherapy or a central venous catheter). The strategy of restricting thromboprophylaxis to cancer outpatients with specific indications, however, may miss an opportunity to reduce VTE in this vulnerable patient population. In the PROTECHT study, 1166 ambulatory cancer patients were randomly assigned to placebo or the LMWH nadroparin for the duration of their chemotherapy. Treatment with nadroparin reduced the rate of clinical thrombosis by 47.2% compared with placebo (3.9% vs. 2.1%; P = 0.033). The risk reduction was consistent across all measured events, including DVT, PE, stroke, and visceral venous thrombosis.31
Pregnancy
Prophylaxis should be considered in pregnant women with known risk factors for VTE such as prior VTE, thrombophilia, and a history of prolonged immobility. In addition, women with a moderate to high risk of VTE associated with a cesarean section should be considered for postpartum thromboprophylaxis. For example, 1 of the following regimens may be appropriate for high‐risk women following a cesarean section:32
-
UFH 5000 units subcutaneously every 12 hours until fully mobile;
-
LMWH subcutaneously once daily for 5 days (such as enoxaparin 20 mg daily).
For pregnant women already receiving anticoagulant prophylaxis (eg, for hypercoagulable state, structural heart disease, or prior DVT/PE), ACCP guidelines recommend discontinuing VKAs before 6 weeks of fetal gestation to minimize the risk of birth defects and miscarriage. In general, a LMWH should be substituted for VKAs as soon as pregnancy is confirmed or prior to conception in preparation for pregnancy, as VKAs cross the placental barrier, but LMWH and UFH do not.1, 33
Renal Insufficiency
The ACCP guidelines recommend that renal function be considered when making decisions about the use and/or dose of LMWHs and fondaparinux. Because these agents are eliminated primarily via renal clearance, changes in renal function can reduce drug clearance, prolong the half‐life, and increase plasma concentrations. Consequently, the risk of treatment‐related bleeding complications is elevated in patients with renal impairment.1 Depending on the circumstances, one of the following options should be considered1:
-
Avoid using an anticoagulant that bioaccumulates in the presence of renal impairment;
-
Use a lower dose of the agent;
-
Monitor the drug level or its anticoagulant effect.
In severe renal impairment (creatinine clearance < 30 mL/minute):710
-
The prophylactic dose of enoxaparin should be adjusted to 30 mg subcutaneously once daily; no specific dosing adjustments have been recommended for dalteparin or tinzaparin;
-
Fondaparinux is contraindicated.
Epidural Anesthesia
Neuraxial blockade has several advantages over systemic opioids, but the risk of spinal or epidural hematoma may be increased with the concomitant use of antithrombotic drugs. Therefore, these agents must be used cautiously in patients with neuraxial blockade.1 Guidelines from the American Society of Regional Anesthesia and Pain Medicine (ASRA) contain the following recommendations:34
-
Subcutaneous UFH: No contraindication, consider delaying heparin until after block if technical difficulty is anticipated;
-
LMWH: Since twice daily dosing may be associated with an increased risk of spinal hematoma, delay initiation of LMWH until at least 24 hours after surgery, regardless of anesthetic technique; for single daily dosing, administer the first dose of LMWH 6 to 8 hours postoperatively and second dose no sooner than 24 hours after the first dose;
-
Warfarin: Document normal INR after discontinuation (prior to neuraxial technique); remove catheter when INR 1.5 (initiation of therapy).
Complications of Thromboprophylaxis
Before initiating thromboprophylaxis, it is important to evaluate the risk of bleeding, and patients should be assessed for contraindications that could increase that risk. HIT should also be considered.
Bleeding Risk
The ACCP and ASCO guidelines emphasize the importance of weighing the potential benefits of thromboprophylaxis against the potential risks of bleeding in individual patients. According to the ACCP, the overall risk of bleeding with intravenous UFH in patients with VTE is less than 3%, and thromboprophylaxis has not been shown to increase the risk of bleeding compared with placebo in major clinical trials.13, 15, 35 However, bleeding risk may increase in older patients and with higher doses of heparin. Warfarin therapy can be monitored with an INR to reduce the risk of bleeding during thromboprophylaxis.1
Anticoagulation therapy may be contraindicated in patients with certain factors and conditions that increase the risk of bleeding. These include:
-
Clinically significant active or chronic bleeding;
-
Recent central nervous system or spinal surgery with increased risk of bleeding;
-
Thrombocytopenia (excluding HIT) or severe platelet dysfunction;
-
Abnormalities associated with clotting factors.
The NCCN provides specific contraindications to anticoagulation therapy for the prevention and treatment of VTE in cancer patients.28 These include:
-
Recent central nervous system bleed; intracranial, or spinal lesions at high risk of bleeding;
-
Active major bleeding (> 2 units transfused in 24 hours);
-
Chronic, clinically significant measurable bleeding for more than 48 hours;
-
Thrombocytopenia (platelets < 50,000/L);
-
Severe platelet dysfunction;
-
Recent major operation with high risk of bleeding;
-
Underlying coagulopathy (eg, clotting factor abnormalities or elevated prothrombin time or activated partial thromboplastin time [aPTT]);
-
Spinal anesthesia or lumbar puncture;
-
High risk of falls.
HIT
HIT is a serious complication that can occur as a result of exposure to heparin. It is an immune response that causes platelet activation and platelet aggregation, among other effects, and is capable of leading to severe thrombosis, amputation, or death.36 The incidence of HIT varies with subpopulations of patients and more commonly develops in patients receiving heparin in therapeutic doses. Early diagnosis (through an interpretation of clinical and laboratory information) is important to improve clinical outcomes, but difficult to achieve.36 The ACCP guidelines note that enzyme‐linked immunosorbent assay (ELISA)‐based tests for HIT are often falsely positive after surgery. As an alternative, serotonin‐release tests are more specific, although they are not as widely available.1
Substantial clinical evidence suggests that LMWH poses less of a risk of HIT than UFH. Martel et al,37 for example, conducted a meta‐analysis of 15 randomized and nonrandomized controlled trials (a total of 7287 patients) that included studies that compared prophylactic doses of UFH and LMWH and assessed postoperative or medical inpatients who received prophylaxis. The analysis revealed that the risk of HIT was 2.6% following UFH use compared with 0.2% following LMWH use.37 Despite the inclusion of UFH in the ASCO guidelines, ASCO acknowledges that a lower risk of HIT is one of the potential advantages of LMWH over UFH in cancer surgery prophylaxis.27 In addition, the recommendation to transition to outpatient therapy as soon as possible is an indirect way of stating a preference for LMWH. For cancer patients with established VTE, the recommendation is more direct: LMWH is clearly preferred over UFH for both initial and continuing antithrombotic therapy.27
Conclusions
Thromboprophylaxis should be considered in all hospitalized patients who have a risk of VTE. Anticoagulants are the mainstays of prophylaxis, and recent clinical trials have clearly demonstrated the efficacy of LMWHs and fondaparinux in preventing VTE. Each class of anticoagulant carries a number of side effects and contraindications, and frequent patient evaluation and monitoring may be required. This is especially true in those with renal impairment, for whom UFH may be a logical choice. A number of organizations have released guidelines for VTE prophylaxis that provide specific recommendations regarding thromboprophylaxis in special patient populations and scenarios.
- , , , et al.Prevention of venous thromboembolism: American College of Chest Physicians evidence‐based clinical practice guidelines. 8th Edition.Chest.2008;133(6 suppl):381S–453S.
- , , .Double trouble for 2,609 hospitalized medical patients who developed deep vein thrombosis: prophylaxis omitted more often and pulmonary embolism more frequent.Chest.2007;132(2):554–561.
- , , , , .Meta‐analysis: anticoagulant prophylaxis to prevent symptomatic venous thromboembolism in hospitalized medical patients.Ann Intern Med.2007;146(4):278–288.
- , , , , .Anticoagulant prophylaxis to prevent asymptomatic deep vein thrombosis in hospitalized medical patients: a systematic review and meta‐analysis.J Thromb Haemost.2008;6:405–414.
- , , , et al.Pharmacology and Management of the Vitamin K Antagonists. American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines. 8th Edition.Chest.2008;133:160S–198S.
- , .Outpatient management of anticoagulation therapy.Am Fam Physician.2007;75:1031–1042.
- Fragmin prescribing information.Last updated April 2007.Woodcliff Lake, NJ:Eisai, Inc.;2007.
- Lovenox prescribing information.Last updated 2008.Bridgewater, NJ:sanofi‐aventis U.S.;2008.
- Innohep prescribing information.Last updated July 2000.Wilmington, DE:DuPont Pharma;2000.
- Arixtra prescribing information.Last updated October 2008.Research Triangle Park, NC:GlaxoSmithKline;2008.
- , , , et al.Cost effectiveness of thromboprophylaxis with a low‐molecular‐weight heparin versus unfractionated heparin in acutely ill medical inpatients.Am J Manag Care.2004;10(9):632–642.
- , , , et al.A comparison of enoxaparin with placebo for the prevention of venous thromboembolism in acutely ill medical patients.N Engl J Med.1999;341(11):793–800.
- , , , et al.Randomized, placebo‐controlled trial of dalteparin for the prevention of venous thromboembolism in acutely ill medical patients.Circulation.2004;110:874–879.
- , , , et al.Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology. Guidelines on the diagnosis and management of acute pulmonary embolism: the Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology (ESC).Eur Heart J.2008;29(18):2276–2315.
- , , , et al.ARTEMIS Investigators. Efficacy and safety of fondaparinux for the prevention of venous thromboembolism in older acute medical patients: randomised placebo controlled trial.BMJ.2006;332(7537):325–329.
- , , , et al.A pooled analysis of four pivotal studies of rivaroxaban for the prevention of venous thromboembolism after orthopaedic surgery: effect on symptomatic venous thromboembolism, death, and bleeding.Blood.2008;112:19–20.
- , , , , , .The efficacy and safety of apixaban, an oral, direct factor Xa inhibitor, as thromboprophylaxis in patients following total knee replacement.J Thromb Haemost.2007;5:2368–2375.
- Bristol‐Myers Squibb and Pfizer. Bristol‐Myers Squibb and Pfizer provide update on apixaban clinical development program [Press release]. August 26, 2008. Available at: http://www.pfizer.com. Accessed August2009.
- , , , et al.Dabigatran etexilate versus enoxaparin for prevention of venous thromboembolism after total hip replacement: a randomized, double blind, non‐inferiority trial.Lancet.2007;370:949–956.
- , , , et al.Dabigatran with or without concomitant aspirin compared with warfarin alone in patients with non‐valvular atrial fibrillation (PETRO Study).Am J Cardiol.2007;100:1419–1426.
- , , , et al.Oral dabigatran etexilate vs. subcutaneous enoxaparin for the prevention of venous thromboembolism after total knee replacement: the RE‐MODEL randomized trial.J Thromb Haemost.2007;5:2178–2185.
- , , , et al.Dabigatran etexilate versus enoxaparin in preventing venous thromboembolism following total knee arthroplasty.J Thromb Haemost.2007;5(suppl 2):Abstract O‐W‐051.
- , , , .Treatment and prevention of heparin‐induced thrombocytopenia: American College of Chest Physicians evidence‐based clinical practice guidelines. 8th edition.Chest.2008;133:340–380.
- .Extended duration of thromboprophylaxis in acutely ill medical patients: optimizing therapy?J Thromb Haemost.2007;5:5–11.
- , , , et al.Extended‐duration thromboprophylaxis in acutely ill medical patients with recent reduced mobility: methodology for the EXCLAIM study.J Thromb Thrombolysis.2006;22:31–38.
- , , , et al.Extended‐duration venous thromboembolism prophylaxis in acutely ill medical patients with recent reduced mobility: the EXCLAIM study. 2007 Congress of the International Society on Thrombosis and Hemostasis; July 7–13,2007; Geneva, Switzerland. Abstract O‐S‐001.
- , , , et al.American Society of Clinical Oncology Guideline: recommendations for venous thromboembolism prophylaxis and treatment in patients with cancer.J Clin Oncol.2007;25:5490–5505.
- National Comprehensive Cancer Network (NCCN). Venous thromboembolic disease. Practice guidelines in oncology. V.1. 2009. Available at: http://www.nccn.org/professionals/physician_gls/PDF/vte.pdf. Accessed August2006.
- American Academy of Orthopaedic Surgeons Clinical Guideline on Prevention of Symptomatic Pulmonary Embolism in Patients Undergoing Total Hip or Knee Arthroplasty. May 18, 2007. Available at http://www.aaos.org/research/guidelines/PE_guideline.pdf. Accessed August2009.
- , , , et al.Incidence of venous thromboembolism in patients hospitalized with cancer.Am J Med.2006;119(1):60–68.
- , , , et al.A randomized double‐blind placebo‐controlled study on nadroparin for prophylaxis of thromboembolic events in cancer patients receiving chemotherapy: The PROTECHT Study.Blood.2008;112(11):Abstract 6.
- , , , et al.Prevention and treatment of venous thromboembolism (VTE) in obstetrics.J SOGC.2000;22(9):736–749.
- , , , et al.Management of venous thromboembolism: a clinical practice guideline from the American College of Physicians and the American Academy of Family Physicians.Am Fam Med.2007;5:74–80.
- , , , et al.Regional anesthesia in the anticoagulated patient: defining the risks (the second ASRA Consensus Conference on Neuraxial Anesthesia and Anticoagulation).Reg Anesth Pain Med.2003;28(3):172–197.
- , , , et al.Prevention of venous thromboembolism in medical patients with enoxaparin; a subgroup analysis of the MEDENOX study.Blood Coagul Fibrinolysis.2003;14:341–348.
- , .The laboratory diagnosis and clinical management of patients with HIT: an update.Semin Thromb Hemost.2008;34(1):86–93.
- , , .Risk for heparin‐induced thrombocytopenia with unfractionated and low‐molecular‐weight heparin thromboprophylaxis: a meta‐analysis.Blood.2005;106(8):2710–2715.
Thromboprophylaxis with anticoagulants has proven benefits in hospitalized patients. Despite this, venous thromboembolism (VTE) prophylaxis is underused and VTE remains the leading cause of preventable hospital mortality.1 Medical patients have a particularly high risk; those who develop a deep vein thrombosis (DVT) are significantly less likely to have received prophylaxis prior to the diagnosis of DVT than nonmedical patients. Even within the high‐risk setting of the intensive care unit (ICU), medical patients receive thromboprophylaxis only two‐thirds as often as nonmedical patients.2
In this article we summarize the evidence concerning the various prophylaxis options, including current guideline recommendations for VTE prevention in medical and surgical patients. We also discuss strategies for thromboprophylaxis in special populations and potential complications of prophylaxis.
Efficacy of Prophylaxis in Medical Patients
Several meta‐analyses have demonstrated the marked benefits of anticoagulant prophylaxis in medical patients. Dentali et al3 conducted a meta‐analysis of 9 randomized controlled trials enrolling a total of 19,958 at‐risk hospitalized medical patients. The selected trials compared standard anticoagulant regimens with no treatment and only included studies with objectively documented and independently adjudicated outcomes. Compared with patients receiving placebo, those receiving thromboprophylaxis had significant reductions in any PE by 57% (95% CI, 0.26‐0.71; absolute risk reduction, 0.29%) and fatal pulmonary embolism (PE) by 62% (95% CI, 0.21‐0.69; absolute risk reduction, 0.25%), with a nonsignificant reduction in symptomatic DVT (relative risk [RR], 0.47; 95% CI, 0.22‐1.00) and a nonsignificant increase in major bleeding (RR, 1.32; 95% CI, 0.73‐2.37). The researchers concluded that anticoagulant prophylaxis is effective in preventing symptomatic VTE in medical patients, though the optimal duration of therapy is not yet defined.3
Another meta‐analysis focusing on subclinical DVT in acutely ill medical patients examined the therapeutic effects of various prophylaxis regimens. Overall, anticoagulant prophylaxis reduced the risk of any asymptomatic DVT (assessed by venogram or ultrasound) by 49% (95% CI, 0.39‐0.67) and asymptomatic proximal DVT by 55% (95% CI, 0.31‐0.65) compared with placebo (absolute risk reduction, 2.6% and 1.8%, respectively). Although prophylaxis was associated with a 0.5% absolute risk increase in major bleeding, the authors concluded that the benefits of prophylaxis outweighed the risks of bleeding.4
Anticoagulant Agents in the Prevention of VTE
Currently available anticoagulants for the prevention of VTE include unfractionated heparin (UFH), low‐molecular‐weight heparins (LMWHs), fondaparinux, and warfarin. These agents interrupt thrombus formation, either indirectly (through interaction with antithrombin) or directly (by inhibiting the action of thrombin). Each class of therapy has advantages and limitations. Table 1 lists common anticoagulant options for VTE prophylaxis, along with dosing information and other important information.510
| Prophylactic Dose | Warnings/Contraindications/Adverse Reactions | |
|---|---|---|
| ||
| Warfarin | 5 to 10 mg daily initially;* adjust dose based on INR; therapeutic INR goal: 2.5 (2‐3) | Warning: bleeding risk; requires frequent monitoring; contraindicated in patients for whom hazard of hemorrhage outweighs potential benefit (eg, in pregnant women) |
| UFH | 5000 IU every 8‐12 hours subcutaneously | Contraindicated in the presence of active bleeding, uncontrolled hypertension, or severe thrombocytopenia; monitor platelet count every 4‐7 days for HIT |
| Dalteparin | 5000 IU daily subcutaneously | Warning: spinal/epidural hematoma; monitor for signs and symptoms of neurological impairment. LMWHs should be used with caution in renal impairment; anti‐Xa monitoring and dose adjustments may be required. Follow prescribing information for dose adjustments and body weight‐based dosing. Most common adverse reactions: bleeding, anemia, thrombocytopenia, elevation of serum aminotransferase, diarrhea, and nausea |
| Enoxaparin | 40 mg daily subcutaneously; reduce to 30 mg daily in renal impairment | |
| Tinzaparin | 3500 IU daily subcutaneously | |
| Fondaparinux | 2.5 mg daily subcutaneously | Warning: spinal/epidural hematoma; monitor for signs and symptoms of neurological impairment; contraindicated in patients with severe renal impairment (creatinine clearance < 30 mL/minute) and in patients < 50 kg |
UFH
UFH, which is typically administered by subcutaneous injection, has the longest history as an anticoagulant in the prevention and treatment of VTE. It is an attractive option in patients with severe renal failure or those who may require a procedure in the near future. Although UFH is partially cleared by the kidney, its short half‐life can be perceived as a safety advantage in patients with severe renal impairment and an increased risk of bleeding. For most other patients, UFH holds several disadvantages compared with newer therapies, including the need for injections to be administered 3 times a day to be optimally effective, its effect on platelets, and its association with heparin‐induced thrombocytopenia (HIT).1 Given the costs of administration and potential complications, it is not less expensive than LMWHs, and it appears to be less cost‐effective.11
LMWHs
LMWHs have a higher bioavailability and longer half‐life than UFH, which translates to reliable anticoagulation levels when given subcutaneously on a weight‐based dosing schedule. Unlike UFH, LMWHs do not require laboratory tests to monitor the intensity of anticoagulation, except in special circumstances.1 The LMWHs dalteparin, enoxaparin, and tinzaparin are widely used for the prevention and treatment of VTE in the United States.
Two landmark clinical trials demonstrated the efficacy of appropriate thromboprophylaxis with LMWHs in reducing the burden of VTE in acutely ill, hospitalized medical patients. The Prophylaxis in Medical Patients with Enoxaparin (MEDENOX) trial and Prospective Evaluation of Dalteparin Efficacy for Prevention of VTE in Immobilized Patients Trial (PREVENT) demonstrated the benefits of enoxaparin and dalteparin, respectively, in reducing the risk of VTE. As shown in Table 2, thromboprophylaxis with these agents was associated with a 45% to 63% relative reduction in the risk of VTE compared with placebo.12, 13
| Trial | Number of Patients | Agent (vs. placebo) | Detection of VTE | Relative Risk Reduction | Number Needed to Treat |
|---|---|---|---|---|---|
| |||||
| MEDENOX | 866 | Enoxaparin 20 mg or 40 mg SC daily for 6‐14 days | Distal and proximal venographic DVT or documented PE | 63% (with 40 mg; 97.6 CI, 0.22‐0.63; P <0.001) | 10 |
| PREVENT | 3706 | Dalteparin 5000 IU SC daily for up to 14 days | CUS DVT, symptomatic VTE, and fatal PE | 45% (95% CI, 0.38‐0.80; P = 0.0015) | 45 |
| ARTEMIS | 849 | Fondaparinux 2.5 mg SC daily for 6‐14 days | Distal and proximal venographic DVT, symptomatic VTE, and fatal PE | 47% (95% CI, 0.077‐0.693) | 20 |
Pentasaccharides
Fondaparinux is a synthetic factor Xa antagonist that shares many features of LMWHs, including a high bioavailability and long half‐life. Fondaparinux does not require monitoring, but it is contraindicated in patients with renal failure (CrCl < 30 mL/minute) and in patients weighing less than 50 kg.1 Although PF4 antibodies have been associated with fondaparinux administration, this drug has not, to date, been associated with HIT.14 The Arixtra for Thromboembolism Prevention in a Medical Indications Study (ARTEMIS) trial demonstrated the advantage of fondaparinux over placebo in reducing the risk of VTE (Table 2).15 The American College of Chest Physicians (ACCP) guidelines state that fondaparinux appears to be as effective and safe as LMWH.1
Vitamin K Antagonists
Vitamin K antagonists (VKAs) such as warfarin inhibit the production of prothrombin, clotting factors VII, IX, and X, and the anticoagulants protein C and protein S. Warfarin is challenging to manage because of its narrow therapeutic window, its tendency to exhibit considerable variability in dose‐response, the time required to reach target international normalized ratio (INR), its potential for interaction with diet and concomitant medications, and its need for ongoing monitoring.5 Warfarin should usually be initiated within the same 24 hours as parenteral anticoagulation, with a goal of achieving INR results between 2.0 and 3.0. An initial dose of 5 to 10 mg for the first 1 or 2 days is appropriate for most patients, and subsequent dosing should be based on INR response.5 Warfarin prophylaxis is primarily used in patients in the US undergoing orthopedic surgery, including total hip replacement and hip and knee arthroplasty.1
Future Anticoagulants
New oral agents have the potential to improve the management of patients who have a moderate to high risk of thromboembolic disease.
Rivaroxaban
This oral factor Xa inhibitor is showing promise in patients undergoing major orthopedic surgery. A prespecified pooled analysis was performed on data from the four Regulation of Coagulation in Orthopedic Surgery to Prevent Deep Vein Thrombosis and Pulmonary Embolism (RECORD) trials to evaluate the effect of rivaroxaban on the composite of symptomatic VTE (DVT or PE) and death, and bleeding. In the analysis, patients undergoing hip or knee arthroplasty had a VTE rate of 0.8% with rivaroxaban vs. 1.6% with enoxaparin, the current gold standard for surgical prophylaxis (P < 0.001). Bleeding rates were not significantly different between treatment arms (P = 0.376).16
Apixaban
This oral, direct, reversible factor Xa inhibitor is under evaluation for the prevention and treatment of VTE. In the Apixaban Prophylaxis in Patients Undergoing Total Knee Replacement Surgery (APROPOS) study of patients undergoing knee replacement, apixaban had a lower composite rate of DVT, PE, and all‐cause mortality when compared with enoxaparin or warfarin.17 In the ADVANCE‐1 study of patients undergoing knee surgery, however, apixaban failed to meet criteria for noninferiority when compared with enoxaparin.18 Apixaban is now being evaluated for VTE prophylaxis in acutely ill medical patients.
Dabigatran
This oral direct thrombin inhibitor reversibly binds to free and fibrin‐bound thrombin. In the RE‐NOVATE trial, dabigatran was noninferior to enoxaparin in reducing the events of DVT, PE, and all‐cause mortality following total hip replacement surgery.19 In a Phase II dose‐ranging trial in patients with atrial fibrillation (Prevention of Embolic and Thrombotic Events in Patients with Persistent [AFPETRO]), dabigatran with or without aspirin was as effective as warfarin in reducing embolic events.20 In the RE‐MODEL study, dabigatran was as effective as enoxaparin in preventing VTE and all‐cause mortality following knee replacement surgery, but failed to show equivalence to a higher dose of enoxaparin in the RE‐MOBILIZE trial.21, 22 It should be noted that in the RE‐MODEL study, enoxaparin was not administered at the dosage recommended by the U.S. Food and Drug Administration (FDA) for knee replacement surgery.
Mechanical Prophylaxis
Mechanical methods of thromboprophylaxis include graduated compression stockings (GCS), intermittent pneumatic compression (IPC) devices, and the venous foot pump (VFP). Mechanical approaches to thromboprophylaxis should be used primarily in patients who have a high risk of bleeding or as an adjunct to pharmacotherapeutic prophylaxis.1 The ACCP guidelines summarize the advantages and limitations of mechanical prophylaxis in patients at risk of developing VTE (Table 3).1
| Advantages | Limitations |
|---|---|
| |
| Does not increase the risk of bleeding | Not as intensively studied as pharmacologic thromboprophylaxis (fewer studies and smaller) |
| Can be used in patients who have a high risk of bleeding | No established standards for size, pressure, or physiologic features |
| Efficacy has been demonstrated in a number of patient groups | Many specific mechanical devices have never been assessed in any clinical trial |
| May enhance the effectiveness of anticoagulant thromboprophylaxis | Almost all mechanical thromboprophylaxis trials were unblinded and therefore have a potential for bias |
| May reduce leg swelling | Are less effective in high‐risk groups than anticoagulant thromboprophylaxis |
| Greater effect in reducing calf DVT than proximal DVT | |
| Effect on PE and death unknown | |
| May reduce or delay the use of more effective anticoagulant thromboprophylaxis | |
| Compliance by patients and staff is often poor | |
| Trials may overestimate the protection compared with routine use | |
| Cost associated with purchase, storage, dispensing, and cleaning of the devices, as well as ensuring optimal compliance | |
When properly fitted, GCS increase venous blood return through external pressure, thereby reducing venous stasis. IPC devices or sequential compression devices are usually applied over compression stockings. In addition to improving venous blood flow, these devices stimulate endogenous fibrinolysis. Compliance is often a problem in medical patients, who may not use the devices properly. Furthermore, for patients with severe vascular insufficiency (ankle brachial index <0.05), IPC may worsen vascular insufficiency and digital gangrene.
Inferior vena cava (IVC) filters are barrier devices that may benefit patients with major bleeding risk in the acute VTE setting by preventing PE. These devices, however, do not prevent DVT and may promote further venous stasis and clotting below the device. Importantly, patients with HIT should not have IVC filters placed due to a very high thrombogenic state that could lead to limb ischemia or cerulea phlegmasia dolens.23
Thromboprophylaxis in Medical Patients
Duration
Although major trials support the use of short‐term prophylaxistypically 6 to 14 daysin‐hospital for acutely ill medical patients, the optimal duration of thromboprophylaxis in these patients is unclear.24 The Extended Clinical Prophylaxis in Acutely Ill Medical Patients (EXCLAIM) trial is the first randomized trial to evaluate the potential benefits of extended prophylaxis in acutely ill medical patients. In this study, 5101 hospitalized patients with varying levels of reduced mobility due to cancer, ischemic stroke, heart failure, respiratory failure, infection, and other acute medical conditions received open‐label enoxaparin 40 mg daily for a mean duration of 10 days. Patients were then randomly assigned to additional therapy with enoxaparin or placebo for a mean duration of 28 additional days.25 Preliminary findings from this trial suggest that high‐risk medical patients can benefit from extended thromboprophylaxis following hospital discharge, with significantly reduced VTE events (RR reduction, 44%; P = 0.0011). The benefits of thromboprophylaxis were apparent during the extended treatment period and persistent through 90 days.26
Guideline Recommendations
Incorrect use of thromboprophylaxis does not stem from a lack of evidence‐based recommendations. Within the past year, the ACCP, the American Society of Clinical Oncology (ASCO), and the National Comprehensive Cancer Network (NCCN) have published updated guidelines for thromboprophylaxis in hospitalized patients at risk of VTE.1, 27, 28 The 2008 ACCP guidelines include more than 700 recommendations for VTE risk assessment and management, to be implemented by a variety of physicians, including pulmonologists, cardiologists, cardiothoracic surgeons, and critical care medicine specialists.1
The ACCP guidelines organize prophylaxis recommendations on the basis of patient risk (Table 4).1 Risk assessment remains relatively subjective, however, and validated risk assessment models are not yet widely available. The prudent approach is to consider thromboprophylaxis for all hospitalized medically ill patients who do not have a specific contraindication.
| Levels of Risk | Approximate DVT Risk Without Thromboprophylaxis (%) | Suggested Thromboprophylaxis Options |
|---|---|---|
| ||
| Low risk | ||
| Minor surgery in mobile patients | < 10 | No specific thromboprophylaxis; early and aggressive ambulation |
| Medical patients who are fully mobile | ||
| Moderate risk | ||
| Most general, open gynecologic, or urologic surgery patients | 10‐40 | LMWH (at recommended doses), low‐dose UFH bid or tid, fondaparinux |
| Medical patients, bed rest or sick | ||
| Moderate VTE risk plus high bleeding risk | Mechanical thromboprophylaxis | |
| High risk | ||
| Hip or knee arthroplasty, HFS | 40‐80 | LMWH (at recommended doses), fondaparinux, oral vitamin K antagonist (INR 2‐3) |
| Major trauma, SCI | ||
| High VTE risk plus high bleeding risk | Mechanical thromboprophylaxis | |
Key evidence‐based recommendations regarding thromboprophylaxis for hospitalized, acutely ill patients include the following:1
-
Every hospital should develop a formal strategy to addresses VTE prophylaxis;
-
Aspirin alone is not recommended to prevent VTE for any patient group;
-
Mechanical methods of thromboprophylaxis should be used primarily for patients who have a high bleeding risk or possibly as an adjunct to anticoagulant thromboprophylaxis;
-
Thromboprophylaxis with LMWH, UFH, or fondaparinux is recommended for patients admitted to hospital with an acute medical illness (Note: fondaparinux is recommended, but not FDA‐approved, for this indication in the United States);
-
On admission to the ICU, all patients should be assessed for risk of VTE, and most should receive thromboprophylaxis;
-
All major trauma and all spinal‐cord injury patients should receive thromboprophylaxis.
Thromboprophylaxis in Surgical Patients
For hospitalized surgical patients, the ACCP guidelines indicate the importance of the type of surgery (eg, gynecologic, urologic, or neurologic) in determining the appropriate prophylaxis strategy. In general, routine thromboprophylaxis is recommended for patients undergoing major general, gynecologic, or orthopedic surgery, as well as bariatric and coronary artery bypass surgery.1 Some specific recommendations regarding thromboprophylaxis for surgical patients include the following:
-
Major general surgery: LMWH, low‐dose UFH, or fondaparinux;
-
Major gynecologic surgery and major open urologic procedures: LMWH, low‐dose UFH, fondaparinux, and/or a mechanical device;
-
Elective hip or knee arthroplasty: Anticoagulant therapy (LMWH, fondaparinux, or a VKA);
-
Hip‐fracture surgery: Fondaparinux, LMWH, a VKA, or low‐dose UFH;
-
Patients undergoing hip or knee arthroplasty or hip‐fracture surgery should receive thromboprophylaxis for a minimum of 10 days; for hip arthroplasty and hip‐fracture surgery, thromboprophylaxis should continue for more than 10 days and up to 35 days.
Although the ACCP guidelines recommend against aspirin monotherapy for any patient group, the American Academy of Orthopaedic Surgeons (AAOS) guidelines state that aspirin alone is an effective option in preventing VTE in standard‐risk patients who are undergoing hip or knee replacement surgery.29 However, evidence for aspirin monotherapy is currently limited.1
The 2008 ACCP guidelines include a new chapter on the perioperative management of patients receiving long‐term antithrombotic treatment who must undergo surgery or other invasive procedures. To minimize surgical bleeding, the ACCP recommends the temporary discontinuation of antithrombotic treatment immediately before and during surgery for most patients. Discontinuing antithrombotics can increase the risk of a thromboembolic event, but this risk must be weighed against the risk of bleeding.1 The guidelines also offer specific recommendations for the use of perioperative bridging therapy in patients receiving VKAs based on the risk of VTE and whether the patient has a mechanical heart valve or atrial fibrillation. Guidelines recommend discontinuing bridging anticoagulation 24 hours prior to surgery if therapeutic subcutaneous LMWH is the agent used and approximately 4 hours prior to surgery if intravenous UFH is the agent used.
Thromboprophylaxis in Special Populations
Care must be taken when using thromboprophylaxis in certain high‐risk populations. The following section provides recommendations regarding prophylaxis in the presence of cancer, pregnancy, renal insufficiency, and epidural anesthesia.
Cancer Patients
The ASCO and NCCN guidelines endorse the use of VTE prophylaxis with anticoagulants in all hospitalized patients with active cancer or suspicion of cancer in the absence of contraindications.27, 28 The ACCP guidelines restrict this recommendation to hospitalized cancer patients who are bedridden.1 Thromboprophylaxis should continue at least through the duration of the hospital stay. Acceptable subcutaneous regimens include fondaparinux, dalteparin, or enoxaparin at the doses presented in Table 1; if UFH is chosen, the dose should be 5000 units every 8 hours.
Cancer patients who are scheduled to undergo major surgery require a different prophylaxis strategy. Even with prophylaxis, cancer patients have a 2‐fold higher risk of postoperative VTE compared with noncancer patients and more than a 3‐fold higher risk of fatal PE.30 To manage this risk, the ASCO, NCCN, and ACCP guidelines recommend extended prophylaxis in patients undergoing major cancer surgery.1, 27, 28 Specific recommendations include the following:
-
All patients undergoing major surgical intervention for malignant disease should be considered for VTE prophylaxis with anticoagulants, with or without mechanical prophylaxis;
-
Thromboprophylaxis should be initiated prior to the start of surgery or as early as possible following surgery;
-
Mechanical interventions may supplement pharmacologic prophylaxis, especially in patients who have the highest risk;
-
Prophylaxis with a LMWH should be initiated 12 to 24 hours after the surgical procedure;
-
Continue prophylaxis at least 7 to 10 days postoperatively;
-
Consider prolonged prophylaxis (ie, up to 4 weeks) with a LMWH for high‐risk patients (eg, patients undergoing major abdominal or pelvic surgery, those with residual malignant disease after surgery, obese patients, and patients with a history of VTE).
Routine prophylaxis with anticoagulants is not recommended for most outpatients, except for those with high‐risk factors (eg, thrombogenic chemotherapy or a central venous catheter). The strategy of restricting thromboprophylaxis to cancer outpatients with specific indications, however, may miss an opportunity to reduce VTE in this vulnerable patient population. In the PROTECHT study, 1166 ambulatory cancer patients were randomly assigned to placebo or the LMWH nadroparin for the duration of their chemotherapy. Treatment with nadroparin reduced the rate of clinical thrombosis by 47.2% compared with placebo (3.9% vs. 2.1%; P = 0.033). The risk reduction was consistent across all measured events, including DVT, PE, stroke, and visceral venous thrombosis.31
Pregnancy
Prophylaxis should be considered in pregnant women with known risk factors for VTE such as prior VTE, thrombophilia, and a history of prolonged immobility. In addition, women with a moderate to high risk of VTE associated with a cesarean section should be considered for postpartum thromboprophylaxis. For example, 1 of the following regimens may be appropriate for high‐risk women following a cesarean section:32
-
UFH 5000 units subcutaneously every 12 hours until fully mobile;
-
LMWH subcutaneously once daily for 5 days (such as enoxaparin 20 mg daily).
For pregnant women already receiving anticoagulant prophylaxis (eg, for hypercoagulable state, structural heart disease, or prior DVT/PE), ACCP guidelines recommend discontinuing VKAs before 6 weeks of fetal gestation to minimize the risk of birth defects and miscarriage. In general, a LMWH should be substituted for VKAs as soon as pregnancy is confirmed or prior to conception in preparation for pregnancy, as VKAs cross the placental barrier, but LMWH and UFH do not.1, 33
Renal Insufficiency
The ACCP guidelines recommend that renal function be considered when making decisions about the use and/or dose of LMWHs and fondaparinux. Because these agents are eliminated primarily via renal clearance, changes in renal function can reduce drug clearance, prolong the half‐life, and increase plasma concentrations. Consequently, the risk of treatment‐related bleeding complications is elevated in patients with renal impairment.1 Depending on the circumstances, one of the following options should be considered1:
-
Avoid using an anticoagulant that bioaccumulates in the presence of renal impairment;
-
Use a lower dose of the agent;
-
Monitor the drug level or its anticoagulant effect.
In severe renal impairment (creatinine clearance < 30 mL/minute):710
-
The prophylactic dose of enoxaparin should be adjusted to 30 mg subcutaneously once daily; no specific dosing adjustments have been recommended for dalteparin or tinzaparin;
-
Fondaparinux is contraindicated.
Epidural Anesthesia
Neuraxial blockade has several advantages over systemic opioids, but the risk of spinal or epidural hematoma may be increased with the concomitant use of antithrombotic drugs. Therefore, these agents must be used cautiously in patients with neuraxial blockade.1 Guidelines from the American Society of Regional Anesthesia and Pain Medicine (ASRA) contain the following recommendations:34
-
Subcutaneous UFH: No contraindication, consider delaying heparin until after block if technical difficulty is anticipated;
-
LMWH: Since twice daily dosing may be associated with an increased risk of spinal hematoma, delay initiation of LMWH until at least 24 hours after surgery, regardless of anesthetic technique; for single daily dosing, administer the first dose of LMWH 6 to 8 hours postoperatively and second dose no sooner than 24 hours after the first dose;
-
Warfarin: Document normal INR after discontinuation (prior to neuraxial technique); remove catheter when INR 1.5 (initiation of therapy).
Complications of Thromboprophylaxis
Before initiating thromboprophylaxis, it is important to evaluate the risk of bleeding, and patients should be assessed for contraindications that could increase that risk. HIT should also be considered.
Bleeding Risk
The ACCP and ASCO guidelines emphasize the importance of weighing the potential benefits of thromboprophylaxis against the potential risks of bleeding in individual patients. According to the ACCP, the overall risk of bleeding with intravenous UFH in patients with VTE is less than 3%, and thromboprophylaxis has not been shown to increase the risk of bleeding compared with placebo in major clinical trials.13, 15, 35 However, bleeding risk may increase in older patients and with higher doses of heparin. Warfarin therapy can be monitored with an INR to reduce the risk of bleeding during thromboprophylaxis.1
Anticoagulation therapy may be contraindicated in patients with certain factors and conditions that increase the risk of bleeding. These include:
-
Clinically significant active or chronic bleeding;
-
Recent central nervous system or spinal surgery with increased risk of bleeding;
-
Thrombocytopenia (excluding HIT) or severe platelet dysfunction;
-
Abnormalities associated with clotting factors.
The NCCN provides specific contraindications to anticoagulation therapy for the prevention and treatment of VTE in cancer patients.28 These include:
-
Recent central nervous system bleed; intracranial, or spinal lesions at high risk of bleeding;
-
Active major bleeding (> 2 units transfused in 24 hours);
-
Chronic, clinically significant measurable bleeding for more than 48 hours;
-
Thrombocytopenia (platelets < 50,000/L);
-
Severe platelet dysfunction;
-
Recent major operation with high risk of bleeding;
-
Underlying coagulopathy (eg, clotting factor abnormalities or elevated prothrombin time or activated partial thromboplastin time [aPTT]);
-
Spinal anesthesia or lumbar puncture;
-
High risk of falls.
HIT
HIT is a serious complication that can occur as a result of exposure to heparin. It is an immune response that causes platelet activation and platelet aggregation, among other effects, and is capable of leading to severe thrombosis, amputation, or death.36 The incidence of HIT varies with subpopulations of patients and more commonly develops in patients receiving heparin in therapeutic doses. Early diagnosis (through an interpretation of clinical and laboratory information) is important to improve clinical outcomes, but difficult to achieve.36 The ACCP guidelines note that enzyme‐linked immunosorbent assay (ELISA)‐based tests for HIT are often falsely positive after surgery. As an alternative, serotonin‐release tests are more specific, although they are not as widely available.1
Substantial clinical evidence suggests that LMWH poses less of a risk of HIT than UFH. Martel et al,37 for example, conducted a meta‐analysis of 15 randomized and nonrandomized controlled trials (a total of 7287 patients) that included studies that compared prophylactic doses of UFH and LMWH and assessed postoperative or medical inpatients who received prophylaxis. The analysis revealed that the risk of HIT was 2.6% following UFH use compared with 0.2% following LMWH use.37 Despite the inclusion of UFH in the ASCO guidelines, ASCO acknowledges that a lower risk of HIT is one of the potential advantages of LMWH over UFH in cancer surgery prophylaxis.27 In addition, the recommendation to transition to outpatient therapy as soon as possible is an indirect way of stating a preference for LMWH. For cancer patients with established VTE, the recommendation is more direct: LMWH is clearly preferred over UFH for both initial and continuing antithrombotic therapy.27
Conclusions
Thromboprophylaxis should be considered in all hospitalized patients who have a risk of VTE. Anticoagulants are the mainstays of prophylaxis, and recent clinical trials have clearly demonstrated the efficacy of LMWHs and fondaparinux in preventing VTE. Each class of anticoagulant carries a number of side effects and contraindications, and frequent patient evaluation and monitoring may be required. This is especially true in those with renal impairment, for whom UFH may be a logical choice. A number of organizations have released guidelines for VTE prophylaxis that provide specific recommendations regarding thromboprophylaxis in special patient populations and scenarios.
Thromboprophylaxis with anticoagulants has proven benefits in hospitalized patients. Despite this, venous thromboembolism (VTE) prophylaxis is underused and VTE remains the leading cause of preventable hospital mortality.1 Medical patients have a particularly high risk; those who develop a deep vein thrombosis (DVT) are significantly less likely to have received prophylaxis prior to the diagnosis of DVT than nonmedical patients. Even within the high‐risk setting of the intensive care unit (ICU), medical patients receive thromboprophylaxis only two‐thirds as often as nonmedical patients.2
In this article we summarize the evidence concerning the various prophylaxis options, including current guideline recommendations for VTE prevention in medical and surgical patients. We also discuss strategies for thromboprophylaxis in special populations and potential complications of prophylaxis.
Efficacy of Prophylaxis in Medical Patients
Several meta‐analyses have demonstrated the marked benefits of anticoagulant prophylaxis in medical patients. Dentali et al3 conducted a meta‐analysis of 9 randomized controlled trials enrolling a total of 19,958 at‐risk hospitalized medical patients. The selected trials compared standard anticoagulant regimens with no treatment and only included studies with objectively documented and independently adjudicated outcomes. Compared with patients receiving placebo, those receiving thromboprophylaxis had significant reductions in any PE by 57% (95% CI, 0.26‐0.71; absolute risk reduction, 0.29%) and fatal pulmonary embolism (PE) by 62% (95% CI, 0.21‐0.69; absolute risk reduction, 0.25%), with a nonsignificant reduction in symptomatic DVT (relative risk [RR], 0.47; 95% CI, 0.22‐1.00) and a nonsignificant increase in major bleeding (RR, 1.32; 95% CI, 0.73‐2.37). The researchers concluded that anticoagulant prophylaxis is effective in preventing symptomatic VTE in medical patients, though the optimal duration of therapy is not yet defined.3
Another meta‐analysis focusing on subclinical DVT in acutely ill medical patients examined the therapeutic effects of various prophylaxis regimens. Overall, anticoagulant prophylaxis reduced the risk of any asymptomatic DVT (assessed by venogram or ultrasound) by 49% (95% CI, 0.39‐0.67) and asymptomatic proximal DVT by 55% (95% CI, 0.31‐0.65) compared with placebo (absolute risk reduction, 2.6% and 1.8%, respectively). Although prophylaxis was associated with a 0.5% absolute risk increase in major bleeding, the authors concluded that the benefits of prophylaxis outweighed the risks of bleeding.4
Anticoagulant Agents in the Prevention of VTE
Currently available anticoagulants for the prevention of VTE include unfractionated heparin (UFH), low‐molecular‐weight heparins (LMWHs), fondaparinux, and warfarin. These agents interrupt thrombus formation, either indirectly (through interaction with antithrombin) or directly (by inhibiting the action of thrombin). Each class of therapy has advantages and limitations. Table 1 lists common anticoagulant options for VTE prophylaxis, along with dosing information and other important information.510
| Prophylactic Dose | Warnings/Contraindications/Adverse Reactions | |
|---|---|---|
| ||
| Warfarin | 5 to 10 mg daily initially;* adjust dose based on INR; therapeutic INR goal: 2.5 (2‐3) | Warning: bleeding risk; requires frequent monitoring; contraindicated in patients for whom hazard of hemorrhage outweighs potential benefit (eg, in pregnant women) |
| UFH | 5000 IU every 8‐12 hours subcutaneously | Contraindicated in the presence of active bleeding, uncontrolled hypertension, or severe thrombocytopenia; monitor platelet count every 4‐7 days for HIT |
| Dalteparin | 5000 IU daily subcutaneously | Warning: spinal/epidural hematoma; monitor for signs and symptoms of neurological impairment. LMWHs should be used with caution in renal impairment; anti‐Xa monitoring and dose adjustments may be required. Follow prescribing information for dose adjustments and body weight‐based dosing. Most common adverse reactions: bleeding, anemia, thrombocytopenia, elevation of serum aminotransferase, diarrhea, and nausea |
| Enoxaparin | 40 mg daily subcutaneously; reduce to 30 mg daily in renal impairment | |
| Tinzaparin | 3500 IU daily subcutaneously | |
| Fondaparinux | 2.5 mg daily subcutaneously | Warning: spinal/epidural hematoma; monitor for signs and symptoms of neurological impairment; contraindicated in patients with severe renal impairment (creatinine clearance < 30 mL/minute) and in patients < 50 kg |
UFH
UFH, which is typically administered by subcutaneous injection, has the longest history as an anticoagulant in the prevention and treatment of VTE. It is an attractive option in patients with severe renal failure or those who may require a procedure in the near future. Although UFH is partially cleared by the kidney, its short half‐life can be perceived as a safety advantage in patients with severe renal impairment and an increased risk of bleeding. For most other patients, UFH holds several disadvantages compared with newer therapies, including the need for injections to be administered 3 times a day to be optimally effective, its effect on platelets, and its association with heparin‐induced thrombocytopenia (HIT).1 Given the costs of administration and potential complications, it is not less expensive than LMWHs, and it appears to be less cost‐effective.11
LMWHs
LMWHs have a higher bioavailability and longer half‐life than UFH, which translates to reliable anticoagulation levels when given subcutaneously on a weight‐based dosing schedule. Unlike UFH, LMWHs do not require laboratory tests to monitor the intensity of anticoagulation, except in special circumstances.1 The LMWHs dalteparin, enoxaparin, and tinzaparin are widely used for the prevention and treatment of VTE in the United States.
Two landmark clinical trials demonstrated the efficacy of appropriate thromboprophylaxis with LMWHs in reducing the burden of VTE in acutely ill, hospitalized medical patients. The Prophylaxis in Medical Patients with Enoxaparin (MEDENOX) trial and Prospective Evaluation of Dalteparin Efficacy for Prevention of VTE in Immobilized Patients Trial (PREVENT) demonstrated the benefits of enoxaparin and dalteparin, respectively, in reducing the risk of VTE. As shown in Table 2, thromboprophylaxis with these agents was associated with a 45% to 63% relative reduction in the risk of VTE compared with placebo.12, 13
| Trial | Number of Patients | Agent (vs. placebo) | Detection of VTE | Relative Risk Reduction | Number Needed to Treat |
|---|---|---|---|---|---|
| |||||
| MEDENOX | 866 | Enoxaparin 20 mg or 40 mg SC daily for 6‐14 days | Distal and proximal venographic DVT or documented PE | 63% (with 40 mg; 97.6 CI, 0.22‐0.63; P <0.001) | 10 |
| PREVENT | 3706 | Dalteparin 5000 IU SC daily for up to 14 days | CUS DVT, symptomatic VTE, and fatal PE | 45% (95% CI, 0.38‐0.80; P = 0.0015) | 45 |
| ARTEMIS | 849 | Fondaparinux 2.5 mg SC daily for 6‐14 days | Distal and proximal venographic DVT, symptomatic VTE, and fatal PE | 47% (95% CI, 0.077‐0.693) | 20 |
Pentasaccharides
Fondaparinux is a synthetic factor Xa antagonist that shares many features of LMWHs, including a high bioavailability and long half‐life. Fondaparinux does not require monitoring, but it is contraindicated in patients with renal failure (CrCl < 30 mL/minute) and in patients weighing less than 50 kg.1 Although PF4 antibodies have been associated with fondaparinux administration, this drug has not, to date, been associated with HIT.14 The Arixtra for Thromboembolism Prevention in a Medical Indications Study (ARTEMIS) trial demonstrated the advantage of fondaparinux over placebo in reducing the risk of VTE (Table 2).15 The American College of Chest Physicians (ACCP) guidelines state that fondaparinux appears to be as effective and safe as LMWH.1
Vitamin K Antagonists
Vitamin K antagonists (VKAs) such as warfarin inhibit the production of prothrombin, clotting factors VII, IX, and X, and the anticoagulants protein C and protein S. Warfarin is challenging to manage because of its narrow therapeutic window, its tendency to exhibit considerable variability in dose‐response, the time required to reach target international normalized ratio (INR), its potential for interaction with diet and concomitant medications, and its need for ongoing monitoring.5 Warfarin should usually be initiated within the same 24 hours as parenteral anticoagulation, with a goal of achieving INR results between 2.0 and 3.0. An initial dose of 5 to 10 mg for the first 1 or 2 days is appropriate for most patients, and subsequent dosing should be based on INR response.5 Warfarin prophylaxis is primarily used in patients in the US undergoing orthopedic surgery, including total hip replacement and hip and knee arthroplasty.1
Future Anticoagulants
New oral agents have the potential to improve the management of patients who have a moderate to high risk of thromboembolic disease.
Rivaroxaban
This oral factor Xa inhibitor is showing promise in patients undergoing major orthopedic surgery. A prespecified pooled analysis was performed on data from the four Regulation of Coagulation in Orthopedic Surgery to Prevent Deep Vein Thrombosis and Pulmonary Embolism (RECORD) trials to evaluate the effect of rivaroxaban on the composite of symptomatic VTE (DVT or PE) and death, and bleeding. In the analysis, patients undergoing hip or knee arthroplasty had a VTE rate of 0.8% with rivaroxaban vs. 1.6% with enoxaparin, the current gold standard for surgical prophylaxis (P < 0.001). Bleeding rates were not significantly different between treatment arms (P = 0.376).16
Apixaban
This oral, direct, reversible factor Xa inhibitor is under evaluation for the prevention and treatment of VTE. In the Apixaban Prophylaxis in Patients Undergoing Total Knee Replacement Surgery (APROPOS) study of patients undergoing knee replacement, apixaban had a lower composite rate of DVT, PE, and all‐cause mortality when compared with enoxaparin or warfarin.17 In the ADVANCE‐1 study of patients undergoing knee surgery, however, apixaban failed to meet criteria for noninferiority when compared with enoxaparin.18 Apixaban is now being evaluated for VTE prophylaxis in acutely ill medical patients.
Dabigatran
This oral direct thrombin inhibitor reversibly binds to free and fibrin‐bound thrombin. In the RE‐NOVATE trial, dabigatran was noninferior to enoxaparin in reducing the events of DVT, PE, and all‐cause mortality following total hip replacement surgery.19 In a Phase II dose‐ranging trial in patients with atrial fibrillation (Prevention of Embolic and Thrombotic Events in Patients with Persistent [AFPETRO]), dabigatran with or without aspirin was as effective as warfarin in reducing embolic events.20 In the RE‐MODEL study, dabigatran was as effective as enoxaparin in preventing VTE and all‐cause mortality following knee replacement surgery, but failed to show equivalence to a higher dose of enoxaparin in the RE‐MOBILIZE trial.21, 22 It should be noted that in the RE‐MODEL study, enoxaparin was not administered at the dosage recommended by the U.S. Food and Drug Administration (FDA) for knee replacement surgery.
Mechanical Prophylaxis
Mechanical methods of thromboprophylaxis include graduated compression stockings (GCS), intermittent pneumatic compression (IPC) devices, and the venous foot pump (VFP). Mechanical approaches to thromboprophylaxis should be used primarily in patients who have a high risk of bleeding or as an adjunct to pharmacotherapeutic prophylaxis.1 The ACCP guidelines summarize the advantages and limitations of mechanical prophylaxis in patients at risk of developing VTE (Table 3).1
| Advantages | Limitations |
|---|---|
| |
| Does not increase the risk of bleeding | Not as intensively studied as pharmacologic thromboprophylaxis (fewer studies and smaller) |
| Can be used in patients who have a high risk of bleeding | No established standards for size, pressure, or physiologic features |
| Efficacy has been demonstrated in a number of patient groups | Many specific mechanical devices have never been assessed in any clinical trial |
| May enhance the effectiveness of anticoagulant thromboprophylaxis | Almost all mechanical thromboprophylaxis trials were unblinded and therefore have a potential for bias |
| May reduce leg swelling | Are less effective in high‐risk groups than anticoagulant thromboprophylaxis |
| Greater effect in reducing calf DVT than proximal DVT | |
| Effect on PE and death unknown | |
| May reduce or delay the use of more effective anticoagulant thromboprophylaxis | |
| Compliance by patients and staff is often poor | |
| Trials may overestimate the protection compared with routine use | |
| Cost associated with purchase, storage, dispensing, and cleaning of the devices, as well as ensuring optimal compliance | |
When properly fitted, GCS increase venous blood return through external pressure, thereby reducing venous stasis. IPC devices or sequential compression devices are usually applied over compression stockings. In addition to improving venous blood flow, these devices stimulate endogenous fibrinolysis. Compliance is often a problem in medical patients, who may not use the devices properly. Furthermore, for patients with severe vascular insufficiency (ankle brachial index <0.05), IPC may worsen vascular insufficiency and digital gangrene.
Inferior vena cava (IVC) filters are barrier devices that may benefit patients with major bleeding risk in the acute VTE setting by preventing PE. These devices, however, do not prevent DVT and may promote further venous stasis and clotting below the device. Importantly, patients with HIT should not have IVC filters placed due to a very high thrombogenic state that could lead to limb ischemia or cerulea phlegmasia dolens.23
Thromboprophylaxis in Medical Patients
Duration
Although major trials support the use of short‐term prophylaxistypically 6 to 14 daysin‐hospital for acutely ill medical patients, the optimal duration of thromboprophylaxis in these patients is unclear.24 The Extended Clinical Prophylaxis in Acutely Ill Medical Patients (EXCLAIM) trial is the first randomized trial to evaluate the potential benefits of extended prophylaxis in acutely ill medical patients. In this study, 5101 hospitalized patients with varying levels of reduced mobility due to cancer, ischemic stroke, heart failure, respiratory failure, infection, and other acute medical conditions received open‐label enoxaparin 40 mg daily for a mean duration of 10 days. Patients were then randomly assigned to additional therapy with enoxaparin or placebo for a mean duration of 28 additional days.25 Preliminary findings from this trial suggest that high‐risk medical patients can benefit from extended thromboprophylaxis following hospital discharge, with significantly reduced VTE events (RR reduction, 44%; P = 0.0011). The benefits of thromboprophylaxis were apparent during the extended treatment period and persistent through 90 days.26
Guideline Recommendations
Incorrect use of thromboprophylaxis does not stem from a lack of evidence‐based recommendations. Within the past year, the ACCP, the American Society of Clinical Oncology (ASCO), and the National Comprehensive Cancer Network (NCCN) have published updated guidelines for thromboprophylaxis in hospitalized patients at risk of VTE.1, 27, 28 The 2008 ACCP guidelines include more than 700 recommendations for VTE risk assessment and management, to be implemented by a variety of physicians, including pulmonologists, cardiologists, cardiothoracic surgeons, and critical care medicine specialists.1
The ACCP guidelines organize prophylaxis recommendations on the basis of patient risk (Table 4).1 Risk assessment remains relatively subjective, however, and validated risk assessment models are not yet widely available. The prudent approach is to consider thromboprophylaxis for all hospitalized medically ill patients who do not have a specific contraindication.
| Levels of Risk | Approximate DVT Risk Without Thromboprophylaxis (%) | Suggested Thromboprophylaxis Options |
|---|---|---|
| ||
| Low risk | ||
| Minor surgery in mobile patients | < 10 | No specific thromboprophylaxis; early and aggressive ambulation |
| Medical patients who are fully mobile | ||
| Moderate risk | ||
| Most general, open gynecologic, or urologic surgery patients | 10‐40 | LMWH (at recommended doses), low‐dose UFH bid or tid, fondaparinux |
| Medical patients, bed rest or sick | ||
| Moderate VTE risk plus high bleeding risk | Mechanical thromboprophylaxis | |
| High risk | ||
| Hip or knee arthroplasty, HFS | 40‐80 | LMWH (at recommended doses), fondaparinux, oral vitamin K antagonist (INR 2‐3) |
| Major trauma, SCI | ||
| High VTE risk plus high bleeding risk | Mechanical thromboprophylaxis | |
Key evidence‐based recommendations regarding thromboprophylaxis for hospitalized, acutely ill patients include the following:1
-
Every hospital should develop a formal strategy to addresses VTE prophylaxis;
-
Aspirin alone is not recommended to prevent VTE for any patient group;
-
Mechanical methods of thromboprophylaxis should be used primarily for patients who have a high bleeding risk or possibly as an adjunct to anticoagulant thromboprophylaxis;
-
Thromboprophylaxis with LMWH, UFH, or fondaparinux is recommended for patients admitted to hospital with an acute medical illness (Note: fondaparinux is recommended, but not FDA‐approved, for this indication in the United States);
-
On admission to the ICU, all patients should be assessed for risk of VTE, and most should receive thromboprophylaxis;
-
All major trauma and all spinal‐cord injury patients should receive thromboprophylaxis.
Thromboprophylaxis in Surgical Patients
For hospitalized surgical patients, the ACCP guidelines indicate the importance of the type of surgery (eg, gynecologic, urologic, or neurologic) in determining the appropriate prophylaxis strategy. In general, routine thromboprophylaxis is recommended for patients undergoing major general, gynecologic, or orthopedic surgery, as well as bariatric and coronary artery bypass surgery.1 Some specific recommendations regarding thromboprophylaxis for surgical patients include the following:
-
Major general surgery: LMWH, low‐dose UFH, or fondaparinux;
-
Major gynecologic surgery and major open urologic procedures: LMWH, low‐dose UFH, fondaparinux, and/or a mechanical device;
-
Elective hip or knee arthroplasty: Anticoagulant therapy (LMWH, fondaparinux, or a VKA);
-
Hip‐fracture surgery: Fondaparinux, LMWH, a VKA, or low‐dose UFH;
-
Patients undergoing hip or knee arthroplasty or hip‐fracture surgery should receive thromboprophylaxis for a minimum of 10 days; for hip arthroplasty and hip‐fracture surgery, thromboprophylaxis should continue for more than 10 days and up to 35 days.
Although the ACCP guidelines recommend against aspirin monotherapy for any patient group, the American Academy of Orthopaedic Surgeons (AAOS) guidelines state that aspirin alone is an effective option in preventing VTE in standard‐risk patients who are undergoing hip or knee replacement surgery.29 However, evidence for aspirin monotherapy is currently limited.1
The 2008 ACCP guidelines include a new chapter on the perioperative management of patients receiving long‐term antithrombotic treatment who must undergo surgery or other invasive procedures. To minimize surgical bleeding, the ACCP recommends the temporary discontinuation of antithrombotic treatment immediately before and during surgery for most patients. Discontinuing antithrombotics can increase the risk of a thromboembolic event, but this risk must be weighed against the risk of bleeding.1 The guidelines also offer specific recommendations for the use of perioperative bridging therapy in patients receiving VKAs based on the risk of VTE and whether the patient has a mechanical heart valve or atrial fibrillation. Guidelines recommend discontinuing bridging anticoagulation 24 hours prior to surgery if therapeutic subcutaneous LMWH is the agent used and approximately 4 hours prior to surgery if intravenous UFH is the agent used.
Thromboprophylaxis in Special Populations
Care must be taken when using thromboprophylaxis in certain high‐risk populations. The following section provides recommendations regarding prophylaxis in the presence of cancer, pregnancy, renal insufficiency, and epidural anesthesia.
Cancer Patients
The ASCO and NCCN guidelines endorse the use of VTE prophylaxis with anticoagulants in all hospitalized patients with active cancer or suspicion of cancer in the absence of contraindications.27, 28 The ACCP guidelines restrict this recommendation to hospitalized cancer patients who are bedridden.1 Thromboprophylaxis should continue at least through the duration of the hospital stay. Acceptable subcutaneous regimens include fondaparinux, dalteparin, or enoxaparin at the doses presented in Table 1; if UFH is chosen, the dose should be 5000 units every 8 hours.
Cancer patients who are scheduled to undergo major surgery require a different prophylaxis strategy. Even with prophylaxis, cancer patients have a 2‐fold higher risk of postoperative VTE compared with noncancer patients and more than a 3‐fold higher risk of fatal PE.30 To manage this risk, the ASCO, NCCN, and ACCP guidelines recommend extended prophylaxis in patients undergoing major cancer surgery.1, 27, 28 Specific recommendations include the following:
-
All patients undergoing major surgical intervention for malignant disease should be considered for VTE prophylaxis with anticoagulants, with or without mechanical prophylaxis;
-
Thromboprophylaxis should be initiated prior to the start of surgery or as early as possible following surgery;
-
Mechanical interventions may supplement pharmacologic prophylaxis, especially in patients who have the highest risk;
-
Prophylaxis with a LMWH should be initiated 12 to 24 hours after the surgical procedure;
-
Continue prophylaxis at least 7 to 10 days postoperatively;
-
Consider prolonged prophylaxis (ie, up to 4 weeks) with a LMWH for high‐risk patients (eg, patients undergoing major abdominal or pelvic surgery, those with residual malignant disease after surgery, obese patients, and patients with a history of VTE).
Routine prophylaxis with anticoagulants is not recommended for most outpatients, except for those with high‐risk factors (eg, thrombogenic chemotherapy or a central venous catheter). The strategy of restricting thromboprophylaxis to cancer outpatients with specific indications, however, may miss an opportunity to reduce VTE in this vulnerable patient population. In the PROTECHT study, 1166 ambulatory cancer patients were randomly assigned to placebo or the LMWH nadroparin for the duration of their chemotherapy. Treatment with nadroparin reduced the rate of clinical thrombosis by 47.2% compared with placebo (3.9% vs. 2.1%; P = 0.033). The risk reduction was consistent across all measured events, including DVT, PE, stroke, and visceral venous thrombosis.31
Pregnancy
Prophylaxis should be considered in pregnant women with known risk factors for VTE such as prior VTE, thrombophilia, and a history of prolonged immobility. In addition, women with a moderate to high risk of VTE associated with a cesarean section should be considered for postpartum thromboprophylaxis. For example, 1 of the following regimens may be appropriate for high‐risk women following a cesarean section:32
-
UFH 5000 units subcutaneously every 12 hours until fully mobile;
-
LMWH subcutaneously once daily for 5 days (such as enoxaparin 20 mg daily).
For pregnant women already receiving anticoagulant prophylaxis (eg, for hypercoagulable state, structural heart disease, or prior DVT/PE), ACCP guidelines recommend discontinuing VKAs before 6 weeks of fetal gestation to minimize the risk of birth defects and miscarriage. In general, a LMWH should be substituted for VKAs as soon as pregnancy is confirmed or prior to conception in preparation for pregnancy, as VKAs cross the placental barrier, but LMWH and UFH do not.1, 33
Renal Insufficiency
The ACCP guidelines recommend that renal function be considered when making decisions about the use and/or dose of LMWHs and fondaparinux. Because these agents are eliminated primarily via renal clearance, changes in renal function can reduce drug clearance, prolong the half‐life, and increase plasma concentrations. Consequently, the risk of treatment‐related bleeding complications is elevated in patients with renal impairment.1 Depending on the circumstances, one of the following options should be considered1:
-
Avoid using an anticoagulant that bioaccumulates in the presence of renal impairment;
-
Use a lower dose of the agent;
-
Monitor the drug level or its anticoagulant effect.
In severe renal impairment (creatinine clearance < 30 mL/minute):710
-
The prophylactic dose of enoxaparin should be adjusted to 30 mg subcutaneously once daily; no specific dosing adjustments have been recommended for dalteparin or tinzaparin;
-
Fondaparinux is contraindicated.
Epidural Anesthesia
Neuraxial blockade has several advantages over systemic opioids, but the risk of spinal or epidural hematoma may be increased with the concomitant use of antithrombotic drugs. Therefore, these agents must be used cautiously in patients with neuraxial blockade.1 Guidelines from the American Society of Regional Anesthesia and Pain Medicine (ASRA) contain the following recommendations:34
-
Subcutaneous UFH: No contraindication, consider delaying heparin until after block if technical difficulty is anticipated;
-
LMWH: Since twice daily dosing may be associated with an increased risk of spinal hematoma, delay initiation of LMWH until at least 24 hours after surgery, regardless of anesthetic technique; for single daily dosing, administer the first dose of LMWH 6 to 8 hours postoperatively and second dose no sooner than 24 hours after the first dose;
-
Warfarin: Document normal INR after discontinuation (prior to neuraxial technique); remove catheter when INR 1.5 (initiation of therapy).
Complications of Thromboprophylaxis
Before initiating thromboprophylaxis, it is important to evaluate the risk of bleeding, and patients should be assessed for contraindications that could increase that risk. HIT should also be considered.
Bleeding Risk
The ACCP and ASCO guidelines emphasize the importance of weighing the potential benefits of thromboprophylaxis against the potential risks of bleeding in individual patients. According to the ACCP, the overall risk of bleeding with intravenous UFH in patients with VTE is less than 3%, and thromboprophylaxis has not been shown to increase the risk of bleeding compared with placebo in major clinical trials.13, 15, 35 However, bleeding risk may increase in older patients and with higher doses of heparin. Warfarin therapy can be monitored with an INR to reduce the risk of bleeding during thromboprophylaxis.1
Anticoagulation therapy may be contraindicated in patients with certain factors and conditions that increase the risk of bleeding. These include:
-
Clinically significant active or chronic bleeding;
-
Recent central nervous system or spinal surgery with increased risk of bleeding;
-
Thrombocytopenia (excluding HIT) or severe platelet dysfunction;
-
Abnormalities associated with clotting factors.
The NCCN provides specific contraindications to anticoagulation therapy for the prevention and treatment of VTE in cancer patients.28 These include:
-
Recent central nervous system bleed; intracranial, or spinal lesions at high risk of bleeding;
-
Active major bleeding (> 2 units transfused in 24 hours);
-
Chronic, clinically significant measurable bleeding for more than 48 hours;
-
Thrombocytopenia (platelets < 50,000/L);
-
Severe platelet dysfunction;
-
Recent major operation with high risk of bleeding;
-
Underlying coagulopathy (eg, clotting factor abnormalities or elevated prothrombin time or activated partial thromboplastin time [aPTT]);
-
Spinal anesthesia or lumbar puncture;
-
High risk of falls.
HIT
HIT is a serious complication that can occur as a result of exposure to heparin. It is an immune response that causes platelet activation and platelet aggregation, among other effects, and is capable of leading to severe thrombosis, amputation, or death.36 The incidence of HIT varies with subpopulations of patients and more commonly develops in patients receiving heparin in therapeutic doses. Early diagnosis (through an interpretation of clinical and laboratory information) is important to improve clinical outcomes, but difficult to achieve.36 The ACCP guidelines note that enzyme‐linked immunosorbent assay (ELISA)‐based tests for HIT are often falsely positive after surgery. As an alternative, serotonin‐release tests are more specific, although they are not as widely available.1
Substantial clinical evidence suggests that LMWH poses less of a risk of HIT than UFH. Martel et al,37 for example, conducted a meta‐analysis of 15 randomized and nonrandomized controlled trials (a total of 7287 patients) that included studies that compared prophylactic doses of UFH and LMWH and assessed postoperative or medical inpatients who received prophylaxis. The analysis revealed that the risk of HIT was 2.6% following UFH use compared with 0.2% following LMWH use.37 Despite the inclusion of UFH in the ASCO guidelines, ASCO acknowledges that a lower risk of HIT is one of the potential advantages of LMWH over UFH in cancer surgery prophylaxis.27 In addition, the recommendation to transition to outpatient therapy as soon as possible is an indirect way of stating a preference for LMWH. For cancer patients with established VTE, the recommendation is more direct: LMWH is clearly preferred over UFH for both initial and continuing antithrombotic therapy.27
Conclusions
Thromboprophylaxis should be considered in all hospitalized patients who have a risk of VTE. Anticoagulants are the mainstays of prophylaxis, and recent clinical trials have clearly demonstrated the efficacy of LMWHs and fondaparinux in preventing VTE. Each class of anticoagulant carries a number of side effects and contraindications, and frequent patient evaluation and monitoring may be required. This is especially true in those with renal impairment, for whom UFH may be a logical choice. A number of organizations have released guidelines for VTE prophylaxis that provide specific recommendations regarding thromboprophylaxis in special patient populations and scenarios.
- , , , et al.Prevention of venous thromboembolism: American College of Chest Physicians evidence‐based clinical practice guidelines. 8th Edition.Chest.2008;133(6 suppl):381S–453S.
- , , .Double trouble for 2,609 hospitalized medical patients who developed deep vein thrombosis: prophylaxis omitted more often and pulmonary embolism more frequent.Chest.2007;132(2):554–561.
- , , , , .Meta‐analysis: anticoagulant prophylaxis to prevent symptomatic venous thromboembolism in hospitalized medical patients.Ann Intern Med.2007;146(4):278–288.
- , , , , .Anticoagulant prophylaxis to prevent asymptomatic deep vein thrombosis in hospitalized medical patients: a systematic review and meta‐analysis.J Thromb Haemost.2008;6:405–414.
- , , , et al.Pharmacology and Management of the Vitamin K Antagonists. American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines. 8th Edition.Chest.2008;133:160S–198S.
- , .Outpatient management of anticoagulation therapy.Am Fam Physician.2007;75:1031–1042.
- Fragmin prescribing information.Last updated April 2007.Woodcliff Lake, NJ:Eisai, Inc.;2007.
- Lovenox prescribing information.Last updated 2008.Bridgewater, NJ:sanofi‐aventis U.S.;2008.
- Innohep prescribing information.Last updated July 2000.Wilmington, DE:DuPont Pharma;2000.
- Arixtra prescribing information.Last updated October 2008.Research Triangle Park, NC:GlaxoSmithKline;2008.
- , , , et al.Cost effectiveness of thromboprophylaxis with a low‐molecular‐weight heparin versus unfractionated heparin in acutely ill medical inpatients.Am J Manag Care.2004;10(9):632–642.
- , , , et al.A comparison of enoxaparin with placebo for the prevention of venous thromboembolism in acutely ill medical patients.N Engl J Med.1999;341(11):793–800.
- , , , et al.Randomized, placebo‐controlled trial of dalteparin for the prevention of venous thromboembolism in acutely ill medical patients.Circulation.2004;110:874–879.
- , , , et al.Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology. Guidelines on the diagnosis and management of acute pulmonary embolism: the Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology (ESC).Eur Heart J.2008;29(18):2276–2315.
- , , , et al.ARTEMIS Investigators. Efficacy and safety of fondaparinux for the prevention of venous thromboembolism in older acute medical patients: randomised placebo controlled trial.BMJ.2006;332(7537):325–329.
- , , , et al.A pooled analysis of four pivotal studies of rivaroxaban for the prevention of venous thromboembolism after orthopaedic surgery: effect on symptomatic venous thromboembolism, death, and bleeding.Blood.2008;112:19–20.
- , , , , , .The efficacy and safety of apixaban, an oral, direct factor Xa inhibitor, as thromboprophylaxis in patients following total knee replacement.J Thromb Haemost.2007;5:2368–2375.
- Bristol‐Myers Squibb and Pfizer. Bristol‐Myers Squibb and Pfizer provide update on apixaban clinical development program [Press release]. August 26, 2008. Available at: http://www.pfizer.com. Accessed August2009.
- , , , et al.Dabigatran etexilate versus enoxaparin for prevention of venous thromboembolism after total hip replacement: a randomized, double blind, non‐inferiority trial.Lancet.2007;370:949–956.
- , , , et al.Dabigatran with or without concomitant aspirin compared with warfarin alone in patients with non‐valvular atrial fibrillation (PETRO Study).Am J Cardiol.2007;100:1419–1426.
- , , , et al.Oral dabigatran etexilate vs. subcutaneous enoxaparin for the prevention of venous thromboembolism after total knee replacement: the RE‐MODEL randomized trial.J Thromb Haemost.2007;5:2178–2185.
- , , , et al.Dabigatran etexilate versus enoxaparin in preventing venous thromboembolism following total knee arthroplasty.J Thromb Haemost.2007;5(suppl 2):Abstract O‐W‐051.
- , , , .Treatment and prevention of heparin‐induced thrombocytopenia: American College of Chest Physicians evidence‐based clinical practice guidelines. 8th edition.Chest.2008;133:340–380.
- .Extended duration of thromboprophylaxis in acutely ill medical patients: optimizing therapy?J Thromb Haemost.2007;5:5–11.
- , , , et al.Extended‐duration thromboprophylaxis in acutely ill medical patients with recent reduced mobility: methodology for the EXCLAIM study.J Thromb Thrombolysis.2006;22:31–38.
- , , , et al.Extended‐duration venous thromboembolism prophylaxis in acutely ill medical patients with recent reduced mobility: the EXCLAIM study. 2007 Congress of the International Society on Thrombosis and Hemostasis; July 7–13,2007; Geneva, Switzerland. Abstract O‐S‐001.
- , , , et al.American Society of Clinical Oncology Guideline: recommendations for venous thromboembolism prophylaxis and treatment in patients with cancer.J Clin Oncol.2007;25:5490–5505.
- National Comprehensive Cancer Network (NCCN). Venous thromboembolic disease. Practice guidelines in oncology. V.1. 2009. Available at: http://www.nccn.org/professionals/physician_gls/PDF/vte.pdf. Accessed August2006.
- American Academy of Orthopaedic Surgeons Clinical Guideline on Prevention of Symptomatic Pulmonary Embolism in Patients Undergoing Total Hip or Knee Arthroplasty. May 18, 2007. Available at http://www.aaos.org/research/guidelines/PE_guideline.pdf. Accessed August2009.
- , , , et al.Incidence of venous thromboembolism in patients hospitalized with cancer.Am J Med.2006;119(1):60–68.
- , , , et al.A randomized double‐blind placebo‐controlled study on nadroparin for prophylaxis of thromboembolic events in cancer patients receiving chemotherapy: The PROTECHT Study.Blood.2008;112(11):Abstract 6.
- , , , et al.Prevention and treatment of venous thromboembolism (VTE) in obstetrics.J SOGC.2000;22(9):736–749.
- , , , et al.Management of venous thromboembolism: a clinical practice guideline from the American College of Physicians and the American Academy of Family Physicians.Am Fam Med.2007;5:74–80.
- , , , et al.Regional anesthesia in the anticoagulated patient: defining the risks (the second ASRA Consensus Conference on Neuraxial Anesthesia and Anticoagulation).Reg Anesth Pain Med.2003;28(3):172–197.
- , , , et al.Prevention of venous thromboembolism in medical patients with enoxaparin; a subgroup analysis of the MEDENOX study.Blood Coagul Fibrinolysis.2003;14:341–348.
- , .The laboratory diagnosis and clinical management of patients with HIT: an update.Semin Thromb Hemost.2008;34(1):86–93.
- , , .Risk for heparin‐induced thrombocytopenia with unfractionated and low‐molecular‐weight heparin thromboprophylaxis: a meta‐analysis.Blood.2005;106(8):2710–2715.
- , , , et al.Prevention of venous thromboembolism: American College of Chest Physicians evidence‐based clinical practice guidelines. 8th Edition.Chest.2008;133(6 suppl):381S–453S.
- , , .Double trouble for 2,609 hospitalized medical patients who developed deep vein thrombosis: prophylaxis omitted more often and pulmonary embolism more frequent.Chest.2007;132(2):554–561.
- , , , , .Meta‐analysis: anticoagulant prophylaxis to prevent symptomatic venous thromboembolism in hospitalized medical patients.Ann Intern Med.2007;146(4):278–288.
- , , , , .Anticoagulant prophylaxis to prevent asymptomatic deep vein thrombosis in hospitalized medical patients: a systematic review and meta‐analysis.J Thromb Haemost.2008;6:405–414.
- , , , et al.Pharmacology and Management of the Vitamin K Antagonists. American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines. 8th Edition.Chest.2008;133:160S–198S.
- , .Outpatient management of anticoagulation therapy.Am Fam Physician.2007;75:1031–1042.
- Fragmin prescribing information.Last updated April 2007.Woodcliff Lake, NJ:Eisai, Inc.;2007.
- Lovenox prescribing information.Last updated 2008.Bridgewater, NJ:sanofi‐aventis U.S.;2008.
- Innohep prescribing information.Last updated July 2000.Wilmington, DE:DuPont Pharma;2000.
- Arixtra prescribing information.Last updated October 2008.Research Triangle Park, NC:GlaxoSmithKline;2008.
- , , , et al.Cost effectiveness of thromboprophylaxis with a low‐molecular‐weight heparin versus unfractionated heparin in acutely ill medical inpatients.Am J Manag Care.2004;10(9):632–642.
- , , , et al.A comparison of enoxaparin with placebo for the prevention of venous thromboembolism in acutely ill medical patients.N Engl J Med.1999;341(11):793–800.
- , , , et al.Randomized, placebo‐controlled trial of dalteparin for the prevention of venous thromboembolism in acutely ill medical patients.Circulation.2004;110:874–879.
- , , , et al.Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology. Guidelines on the diagnosis and management of acute pulmonary embolism: the Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology (ESC).Eur Heart J.2008;29(18):2276–2315.
- , , , et al.ARTEMIS Investigators. Efficacy and safety of fondaparinux for the prevention of venous thromboembolism in older acute medical patients: randomised placebo controlled trial.BMJ.2006;332(7537):325–329.
- , , , et al.A pooled analysis of four pivotal studies of rivaroxaban for the prevention of venous thromboembolism after orthopaedic surgery: effect on symptomatic venous thromboembolism, death, and bleeding.Blood.2008;112:19–20.
- , , , , , .The efficacy and safety of apixaban, an oral, direct factor Xa inhibitor, as thromboprophylaxis in patients following total knee replacement.J Thromb Haemost.2007;5:2368–2375.
- Bristol‐Myers Squibb and Pfizer. Bristol‐Myers Squibb and Pfizer provide update on apixaban clinical development program [Press release]. August 26, 2008. Available at: http://www.pfizer.com. Accessed August2009.
- , , , et al.Dabigatran etexilate versus enoxaparin for prevention of venous thromboembolism after total hip replacement: a randomized, double blind, non‐inferiority trial.Lancet.2007;370:949–956.
- , , , et al.Dabigatran with or without concomitant aspirin compared with warfarin alone in patients with non‐valvular atrial fibrillation (PETRO Study).Am J Cardiol.2007;100:1419–1426.
- , , , et al.Oral dabigatran etexilate vs. subcutaneous enoxaparin for the prevention of venous thromboembolism after total knee replacement: the RE‐MODEL randomized trial.J Thromb Haemost.2007;5:2178–2185.
- , , , et al.Dabigatran etexilate versus enoxaparin in preventing venous thromboembolism following total knee arthroplasty.J Thromb Haemost.2007;5(suppl 2):Abstract O‐W‐051.
- , , , .Treatment and prevention of heparin‐induced thrombocytopenia: American College of Chest Physicians evidence‐based clinical practice guidelines. 8th edition.Chest.2008;133:340–380.
- .Extended duration of thromboprophylaxis in acutely ill medical patients: optimizing therapy?J Thromb Haemost.2007;5:5–11.
- , , , et al.Extended‐duration thromboprophylaxis in acutely ill medical patients with recent reduced mobility: methodology for the EXCLAIM study.J Thromb Thrombolysis.2006;22:31–38.
- , , , et al.Extended‐duration venous thromboembolism prophylaxis in acutely ill medical patients with recent reduced mobility: the EXCLAIM study. 2007 Congress of the International Society on Thrombosis and Hemostasis; July 7–13,2007; Geneva, Switzerland. Abstract O‐S‐001.
- , , , et al.American Society of Clinical Oncology Guideline: recommendations for venous thromboembolism prophylaxis and treatment in patients with cancer.J Clin Oncol.2007;25:5490–5505.
- National Comprehensive Cancer Network (NCCN). Venous thromboembolic disease. Practice guidelines in oncology. V.1. 2009. Available at: http://www.nccn.org/professionals/physician_gls/PDF/vte.pdf. Accessed August2006.
- American Academy of Orthopaedic Surgeons Clinical Guideline on Prevention of Symptomatic Pulmonary Embolism in Patients Undergoing Total Hip or Knee Arthroplasty. May 18, 2007. Available at http://www.aaos.org/research/guidelines/PE_guideline.pdf. Accessed August2009.
- , , , et al.Incidence of venous thromboembolism in patients hospitalized with cancer.Am J Med.2006;119(1):60–68.
- , , , et al.A randomized double‐blind placebo‐controlled study on nadroparin for prophylaxis of thromboembolic events in cancer patients receiving chemotherapy: The PROTECHT Study.Blood.2008;112(11):Abstract 6.
- , , , et al.Prevention and treatment of venous thromboembolism (VTE) in obstetrics.J SOGC.2000;22(9):736–749.
- , , , et al.Management of venous thromboembolism: a clinical practice guideline from the American College of Physicians and the American Academy of Family Physicians.Am Fam Med.2007;5:74–80.
- , , , et al.Regional anesthesia in the anticoagulated patient: defining the risks (the second ASRA Consensus Conference on Neuraxial Anesthesia and Anticoagulation).Reg Anesth Pain Med.2003;28(3):172–197.
- , , , et al.Prevention of venous thromboembolism in medical patients with enoxaparin; a subgroup analysis of the MEDENOX study.Blood Coagul Fibrinolysis.2003;14:341–348.
- , .The laboratory diagnosis and clinical management of patients with HIT: an update.Semin Thromb Hemost.2008;34(1):86–93.
- , , .Risk for heparin‐induced thrombocytopenia with unfractionated and low‐molecular‐weight heparin thromboprophylaxis: a meta‐analysis.Blood.2005;106(8):2710–2715.
When are vasoactive agents indicated in acute heart failure?
Case
A 72-year-old retired nurse with known nonischemic dilated cardiomyopathy with an ejection fraction of approximately 20% and status-post cardiac resynchronization therapy presents to the emergency department with dyspnea with minimal activity, three-pillow orthopnea, and paroxysmal nocturnal dyspnea.
She had been hospitalized twice during the past 60 days for similar symptoms. Her medications included losartan (20 mg po q daily), carvedilol (3.125 mg twice daily), spironolactone (25 mg daily), digoxin (0.125 mg daily), and furosemide (80 mg twice daily). Vital signs are notable for a blood pressure of 90/50 mmHg and an irregular pulse of 90 beats per minute. Physical examination is notable for marked jugular venous distension, lungs clear to auscultation bilaterally, biventricular heaves, a markedly displaced left ventricular point of maximal impulse, and a prominent S3 gallop.
Despite treatment with intravenous furosemide and temporary withdrawal of carvedilol, the patient remains symptomatic with persistent jugular venous distension.
Should she be given a vasoactive agent?
Overview
Acute heart failure syndrome (AHFS), defined as a gradual or rapid change in heart failure signs and symptoms, is the most common cause of hospitalization in the United States1. It is associated with an average in-hospital mortality of 4% to 5%, a 30-day mortality of 7% to11%, and a one-year mortality of 33%2.
In patients with previously established myocardial dysfunction, AHFS commonly reflects exacerbation of symptoms after a period of stability. The clinical presentation and severity of AHFS may range from mild volume overload to life-threatening cardiogenic shock and multi-organ failure unresponsive to pharmacologic therapy.2
Initial management of AHFS depends on definition of the patient’s hemodynamic profile. To guide initial therapy, classify patients into one of four hemodynamic profiles during a brief bedside assessment that relies on evaluation of filling pressures (wet or dry) and adequacy of perfusion (hot or cold) (see figure 1).3
Treating volume overload or elevated filling pressures generally begins with diuretics. Diuretics have been shown to provide symptomatic relief, though they have not yet been proven safe.4 Initial treatment can include a loop diuretic at a dose higher than the patient’s chronic dose, with intravenous dosing offering greater bio-absorption and rapidity in onset of action.5 If perfusion is inadequate, escalate therapy beyond diuretics to include vasoactive agents.
Review of the Data
The use of vasoactive medications is largely based on anecdotal experiences and physiologic assumptions rather than on adequately powered prospective randomized controlled trials.6 Vasoactive therapy includes vasodilator and inotropic support and is generally limited for use in patients with advanced disease not responding to standard medical treatment and diuresis. The physiologic premise rests in the expected improvement in ventricular filling pressures and cardiac output with reduction in afterload and/or preload. Vasodilators counteract vascular constriction, reducing both preload and afterload. Positive inotropic agents amplify cardiac output by increasing the strength of myocardial contraction.
Vasodilators
The Heart Failure Society of America (HFSA) 2006 Comprehensive Heart Failure Practice Guidelines state, “In the absence of symptomatic hypotension, intravenous vasodilators (nitroglycerin, nitroprusside, or nesiritide) may be considered as an addition to diuretic therapy for rapid improvement of congestive symptoms.”7 The clinical utility of nesiritide remains in question with clinical and hemodynamic improvement demonstrated in three randomized trials 8-10; but tempered against meta-analyses 11-12 of selected trials, demonstrating a non-significant trend toward increased kidney dysfunction and death within 30 days (35/485 [7.2%] vs. 15/377 [4.0%] patients; risk ratio from meta-analyses, 1.74; 95% confidence interval, 0.97-3.12; p=0.059). In a randomized trial of 489 in-patients with dyspnea at rest from AHFS, treatment with three hours of intravenous nesiritide resulted in a significant improvement in dyspnea compared with placebo (p=0.03). Similar improvement was observed with intravenous nitroglycerin and did not differ statistically from that observed with nesiritide.8 Nitroprusside, an attractive option among those with hypertension and cardiogenic pulmonary edema, is limited by the need for invasive hemodynamic monitoring and potential for either cyanide toxicity or worsening myocardial ischemia.
Inotropes
Again, there is little evidence from adequately powered randomized controlled trials guiding the use of inotropes. Their use is generally limited to the following indications (see figure 2): (1) Short-term treatment for AHFS that is unresponsive to adequate doses of diuretics and especially when associated with systemic hypotension, (2) Bridge to recovery (as following myocarditis) or to definitive treatment (as with transplant), or (3) For palliation when symptomatic relief is the agreed upon goal.13 The HFSA 2006 guideline states: “Intravenous inotropes may be considered to relieve symptoms and improve end-organ function in patients with advanced HF characterized by left ventricle dilation, reduced left ventricular ejection fraction, and diminished peripheral perfusion or end-organ dysfunction, particularly if these patients have marginal systolic blood pressure, have symptomatic hypotension despite adequate filling pressures, or are unresponsive to, or intolerant of, intravenous vasodilators.”7
Dobutamine and milrinone are the most commonly used IV inotropes for the treatment of AHFS and increase contractility by increasing intracellular levels of cyclic adenylate monophosphate (cAMP). Dobutamine is a catechlamine agonist that increases cAMP production through stimulation of adenylate cyclase. Milrinone selectively inhibits phosphodiesterase III, which catalyzes the breakdown of cAMP.
Despite their frequent use when traditional treatments have failed, the data supporting the use of dobutamine and milrinone is limited. The largest registry of patients with AHFS to date associated excess mortality with intravenous inotrope use compared to nitroglycerin or nesiritide.14 In a study population of 255 patients randomized to receive either intravenous nesiritide or intravenous dobutamine, Burger et al.15 demonstrated that dobutamine significantly increased the mean number of ventricular tachycardia events per 24 hours (p=0.001), suggesting increased arrhythmogenicity associated with inotrope use. Nonetheless, in a randomized trial of 15 patients admitted with AHFS, functional class improved in six of eight dobutamine-treated patients, but in only two of seven patients treated with placebo, suggesting clinical improvement as a consequence of inotropic stimulation.16 Unverferth et al. demonstrated a similar sustained functional improvement up to 10 weeks following a 72-hour infusion of intravenous dobutamine. 17
The Outcomes of a Prospective Trial of Intravenous Milrinone for Exacerbations of Chronic Heart Failure trial (OPTIME-CHF), randomized 951 patients with AHFS to receive either intravenous milrinone or placebo within 48 hours of hospitalization.18 Compared to placebo, milrinone was associated with a significant increase in sustained hypotension requiring intervention (10.7% vs. 3.2%; p<.001) and new atrial arrhythmias (4.6% vs. 1.5%; p=0.004), along with a non-significant trend toward increased mortality (3.8% vs. 2.3%; p=0.19). However, as measured by a visual analog scale, milrinone-treated patients reported feeling better than those treated with placebo at 30 days post-randomization (p=0.02).
Although there are not randomized data comparing the efficacy of milrinone and dobutamine in AHFS, a retrospective analysis of 329 patients compared the hemodynamic and clinical effects of these two inotropes.19 Milrinone consistently was associated with a more favorable hemodynamic response, including lower systemic vascular resistance (p=0.01); lower pulmonary artery wedge pressure (p<0.001); larger percentage increase in cardiac index (p=0.03); and larger percentage decrease in pulmonary vascular resistance (p=0.0001). In-hospital mortality (dobutamine 7.8% vs. milrinone 10%) was not significantly different.
Conclusion
Clearly, vasoactive and inotropic agents are available when AHFS is refractory to traditional diuresis and may offer short-term symptomatic relief, palliation in the context of end-of-life care, or bridge to recovery or more definitive treatment. Unfortunately, sufficient and robust evidence that supports the safety and efficacy of such agents is lacking and their use is largely guided by historical practices, clinical experience, and anticipation of theoretic physiologic changes. While adequately powered prospective randomized data emerge, newer agents such as vasopressin receptor antagonists, cardiac myosin activators, calcium sensitizers, and adenosine-receptor antagonists will offer additional pharmacologic options.20 When continued pharmacologic support becomes ineffective, device therapy is available to aid in the treatment of AHFS and includes ultrafiltration to reduce filling pressures and intra-aortic balloon pump counterpulsation or left ventricular assist device placement for pharmacologically resistant cardiogenic shock.21
Back to the Case
Despite maximal medical therapy for her chronic heart failure and biventricular pacing, the patient continued to have markedly limited functional status and repeated hospitalizations for AHFS. Given her advanced age and poor nutritional status, she was not a candidate for cardiac transplantation or placement of a left ventricular assist device. To allow for palliative tailored therapy, right heart catheterization was performed. Right heart catheterization revealed elevated filling pressures, as follows: right atrium, 20 mmHg; pulmonary artery, 63/34 mmHg (mean 47 mmHg); and pulmonary capillary wedge, 29 mmHg. Her mixed venous oxygen saturation was only 41% with a calculated cardiac output of 2.9 liters per minute and cardiac index of 2 liters per minute per meter squared.
As she expressed symptomatic relief as her goal, she was started on intravenous milrinone at 0.2 micrograms per kilogram per minute. This was done with the understanding her symptoms would likely would improve, at the expense of worsening longevity and prognosis. With uptitration of her intravenous milrinone and a continuous infusion of furosemide, she demonstrated the following filling pressures within 24 hours: right atrium, 18 mmHg; pulmonary artery, 63/33 mmHg (mean 43 mmHg); and pulmonary capillary wedge, 24. Importantly, her mixed venous oxygen saturation improved to 68% with a calculated cardiac output of 3.4 liters per minute and cardiac index of 2.4 liters per minute per meter squared. These favorable hemodynamic changes were accompanied by modest improvement in symptoms. After continued intravenous diuresis, she was transitioned back to an oral diuretic regimen and was ultimately discharged to home with a continuous infusion of milrinone for palliation. TH
Drs. Vaishnava, McKean, Nohria, and Baughman are from Brigham and Women’s Hospital and Harvard Medical School in Boston, Mass.
REFERENCES:
- Thom T, Haase N, Rosamond W, et al. Heart disease and stroke statistics – 2006 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2006;113:85-151.
- Iong P, Vowinckel E, Liu PP, Gong Y, Tu JV. Prognosis and determinants of survival in patients newly hospitalized for heart failure: a population-based study. Arch Intern Med. 2002;162:1689-94.
- Nohria A, Lewis EF, Stevenson LW. Medical management of advanced heart failure. JAMA. 2002;287;628-40.
- Faris R, Flather MD, Purcell H, et al. Diuretics for heart failure. Cochrane Database Syst Rev. 2006;1;CD003838.
- Wang DJ and Gottlieb SS. Diuretics: Still the mainstay of treatment. Crit Care Med. 2008;36(Suppl.):S89-S94.
- Fares WH. Management of acute decompensated heart failure in an evidence-based era: What is the evidence behind the current standard of care? Heart & Lung. 2008;37(3):173-8.
- Adams KF, Lindenfield J, Arnold J, et al. Executive summary: HFSA 2006 comprehensive heart failure practice guidelines. J Card Fail. 2006;12:10-38.
- Publication Committee for the VMAC Investigators (Vasodilatation in the Management of Acute CHF). Intravenous nesiritide vs nitroglycerin for treatment of decompensated congestive heart failure: a randomized controlled trial. JAMA. 2002;297:1531-40.
- Peacock WF, Enerman CL, Silver MA, on behalf of the PROACTION Study Group. Am J Emerg Med. 2005;23:327-31.
- Cotter G, Metzkor E, Kaluski E, et al. Randomized trial of high-dose isosorbide dinitrate plus low-dose furosemide versus high-dose furosemide plus low-dose isosorbide dinitrate in severe pulmonary edema. Lancet. 1998;351:389-93.
- Sackner-Bernstein JD, Kowalski M, Fox M, Aaronson K. Short-term risk of death after treatment with nesiritide for decompensated heart failure: a pooled analysis of randomized controlled trials. JAMA. 2005;293:1900-5.
- Sackner-Bernstein JD, Skopicki HA, Aaronson K. Risk of worsening renal function with nesiritide in patients with acutely decompensated heart failure. Circulation. 2005;111:1487-91.
- Felker GM and O’Connor CM. Inotropic therapy for heart failure: An evidence-based approach. American Heart Journal. 2001; 142:393-401.
- Abrahm WT, Adams KF, Fonarow GC, et al. In-hospital mortality in patients with acute decompensated heart failure requiring intravenous vasoactive medications: an analysis from the Acute Decompensated Heart Failure National Registry (ADHERE). J Am Coll Cardiol. 2005;46:57-64.
- Burger AJ, Houton DP, LeJemtel T, et al. Effect of nesiritide and dobutamine on ventricular arrhythmias in the treatment of patients with acutely decompensated congestive heart failure: the PRECEDENT study. American Heart Journal. 2002;144:1102-8.
- Liang CS, Sherman LG, Doherty JU, et al. Sustained improvement of cardiac function in patients with congestive heart failure after short-term infusion of dobutamine. Circulation. 1984;69:113-9.
- Unverferth DV, Magorien RD, Lewis RP, et al. Long-term benefit of dobutamine in patients with congestive cardiomyopathy. American Heart Journal. 1980;100:622-30.
- Cuffe MS, Califf RM, Adams KF Jr, et al. Outcomes of a Prospective Trial of Intravenous Milrinone for Exacerbations of Chronic Heart Failure (OPTIME-CHF) Investigators. Short-term intravenous milrinone for acute exacerbation of chronic heart failure: a randomized controlled trial. JAMA. 2002;287:1541-7.
- Yamani MH, Haji SA, Starling RC, et al. Comparison of dobutamine-based and milrinone-based therapy for advanced decompensated congestive heart failure: Hemodynamic efficacy, clinical outcome, and economic impact. American Heart Journal. 2001;142:998-1002.
- Shin, DD, Brandimarte F, De Luca L, et al. Review of current and investigational pharmacologic agents for acute heart failure syndromes. Am J Cardiol. 2007;99(suppl):4A-23A.
- Kale P and Fang JC. Devices in acute heart failure. Crit Care Med. 2008;36(Suppl.):S121-128.
Case
A 72-year-old retired nurse with known nonischemic dilated cardiomyopathy with an ejection fraction of approximately 20% and status-post cardiac resynchronization therapy presents to the emergency department with dyspnea with minimal activity, three-pillow orthopnea, and paroxysmal nocturnal dyspnea.
She had been hospitalized twice during the past 60 days for similar symptoms. Her medications included losartan (20 mg po q daily), carvedilol (3.125 mg twice daily), spironolactone (25 mg daily), digoxin (0.125 mg daily), and furosemide (80 mg twice daily). Vital signs are notable for a blood pressure of 90/50 mmHg and an irregular pulse of 90 beats per minute. Physical examination is notable for marked jugular venous distension, lungs clear to auscultation bilaterally, biventricular heaves, a markedly displaced left ventricular point of maximal impulse, and a prominent S3 gallop.
Despite treatment with intravenous furosemide and temporary withdrawal of carvedilol, the patient remains symptomatic with persistent jugular venous distension.
Should she be given a vasoactive agent?
Overview
Acute heart failure syndrome (AHFS), defined as a gradual or rapid change in heart failure signs and symptoms, is the most common cause of hospitalization in the United States1. It is associated with an average in-hospital mortality of 4% to 5%, a 30-day mortality of 7% to11%, and a one-year mortality of 33%2.
In patients with previously established myocardial dysfunction, AHFS commonly reflects exacerbation of symptoms after a period of stability. The clinical presentation and severity of AHFS may range from mild volume overload to life-threatening cardiogenic shock and multi-organ failure unresponsive to pharmacologic therapy.2
Initial management of AHFS depends on definition of the patient’s hemodynamic profile. To guide initial therapy, classify patients into one of four hemodynamic profiles during a brief bedside assessment that relies on evaluation of filling pressures (wet or dry) and adequacy of perfusion (hot or cold) (see figure 1).3
Treating volume overload or elevated filling pressures generally begins with diuretics. Diuretics have been shown to provide symptomatic relief, though they have not yet been proven safe.4 Initial treatment can include a loop diuretic at a dose higher than the patient’s chronic dose, with intravenous dosing offering greater bio-absorption and rapidity in onset of action.5 If perfusion is inadequate, escalate therapy beyond diuretics to include vasoactive agents.
Review of the Data
The use of vasoactive medications is largely based on anecdotal experiences and physiologic assumptions rather than on adequately powered prospective randomized controlled trials.6 Vasoactive therapy includes vasodilator and inotropic support and is generally limited for use in patients with advanced disease not responding to standard medical treatment and diuresis. The physiologic premise rests in the expected improvement in ventricular filling pressures and cardiac output with reduction in afterload and/or preload. Vasodilators counteract vascular constriction, reducing both preload and afterload. Positive inotropic agents amplify cardiac output by increasing the strength of myocardial contraction.
Vasodilators
The Heart Failure Society of America (HFSA) 2006 Comprehensive Heart Failure Practice Guidelines state, “In the absence of symptomatic hypotension, intravenous vasodilators (nitroglycerin, nitroprusside, or nesiritide) may be considered as an addition to diuretic therapy for rapid improvement of congestive symptoms.”7 The clinical utility of nesiritide remains in question with clinical and hemodynamic improvement demonstrated in three randomized trials 8-10; but tempered against meta-analyses 11-12 of selected trials, demonstrating a non-significant trend toward increased kidney dysfunction and death within 30 days (35/485 [7.2%] vs. 15/377 [4.0%] patients; risk ratio from meta-analyses, 1.74; 95% confidence interval, 0.97-3.12; p=0.059). In a randomized trial of 489 in-patients with dyspnea at rest from AHFS, treatment with three hours of intravenous nesiritide resulted in a significant improvement in dyspnea compared with placebo (p=0.03). Similar improvement was observed with intravenous nitroglycerin and did not differ statistically from that observed with nesiritide.8 Nitroprusside, an attractive option among those with hypertension and cardiogenic pulmonary edema, is limited by the need for invasive hemodynamic monitoring and potential for either cyanide toxicity or worsening myocardial ischemia.
Inotropes
Again, there is little evidence from adequately powered randomized controlled trials guiding the use of inotropes. Their use is generally limited to the following indications (see figure 2): (1) Short-term treatment for AHFS that is unresponsive to adequate doses of diuretics and especially when associated with systemic hypotension, (2) Bridge to recovery (as following myocarditis) or to definitive treatment (as with transplant), or (3) For palliation when symptomatic relief is the agreed upon goal.13 The HFSA 2006 guideline states: “Intravenous inotropes may be considered to relieve symptoms and improve end-organ function in patients with advanced HF characterized by left ventricle dilation, reduced left ventricular ejection fraction, and diminished peripheral perfusion or end-organ dysfunction, particularly if these patients have marginal systolic blood pressure, have symptomatic hypotension despite adequate filling pressures, or are unresponsive to, or intolerant of, intravenous vasodilators.”7
Dobutamine and milrinone are the most commonly used IV inotropes for the treatment of AHFS and increase contractility by increasing intracellular levels of cyclic adenylate monophosphate (cAMP). Dobutamine is a catechlamine agonist that increases cAMP production through stimulation of adenylate cyclase. Milrinone selectively inhibits phosphodiesterase III, which catalyzes the breakdown of cAMP.
Despite their frequent use when traditional treatments have failed, the data supporting the use of dobutamine and milrinone is limited. The largest registry of patients with AHFS to date associated excess mortality with intravenous inotrope use compared to nitroglycerin or nesiritide.14 In a study population of 255 patients randomized to receive either intravenous nesiritide or intravenous dobutamine, Burger et al.15 demonstrated that dobutamine significantly increased the mean number of ventricular tachycardia events per 24 hours (p=0.001), suggesting increased arrhythmogenicity associated with inotrope use. Nonetheless, in a randomized trial of 15 patients admitted with AHFS, functional class improved in six of eight dobutamine-treated patients, but in only two of seven patients treated with placebo, suggesting clinical improvement as a consequence of inotropic stimulation.16 Unverferth et al. demonstrated a similar sustained functional improvement up to 10 weeks following a 72-hour infusion of intravenous dobutamine. 17
The Outcomes of a Prospective Trial of Intravenous Milrinone for Exacerbations of Chronic Heart Failure trial (OPTIME-CHF), randomized 951 patients with AHFS to receive either intravenous milrinone or placebo within 48 hours of hospitalization.18 Compared to placebo, milrinone was associated with a significant increase in sustained hypotension requiring intervention (10.7% vs. 3.2%; p<.001) and new atrial arrhythmias (4.6% vs. 1.5%; p=0.004), along with a non-significant trend toward increased mortality (3.8% vs. 2.3%; p=0.19). However, as measured by a visual analog scale, milrinone-treated patients reported feeling better than those treated with placebo at 30 days post-randomization (p=0.02).
Although there are not randomized data comparing the efficacy of milrinone and dobutamine in AHFS, a retrospective analysis of 329 patients compared the hemodynamic and clinical effects of these two inotropes.19 Milrinone consistently was associated with a more favorable hemodynamic response, including lower systemic vascular resistance (p=0.01); lower pulmonary artery wedge pressure (p<0.001); larger percentage increase in cardiac index (p=0.03); and larger percentage decrease in pulmonary vascular resistance (p=0.0001). In-hospital mortality (dobutamine 7.8% vs. milrinone 10%) was not significantly different.
Conclusion
Clearly, vasoactive and inotropic agents are available when AHFS is refractory to traditional diuresis and may offer short-term symptomatic relief, palliation in the context of end-of-life care, or bridge to recovery or more definitive treatment. Unfortunately, sufficient and robust evidence that supports the safety and efficacy of such agents is lacking and their use is largely guided by historical practices, clinical experience, and anticipation of theoretic physiologic changes. While adequately powered prospective randomized data emerge, newer agents such as vasopressin receptor antagonists, cardiac myosin activators, calcium sensitizers, and adenosine-receptor antagonists will offer additional pharmacologic options.20 When continued pharmacologic support becomes ineffective, device therapy is available to aid in the treatment of AHFS and includes ultrafiltration to reduce filling pressures and intra-aortic balloon pump counterpulsation or left ventricular assist device placement for pharmacologically resistant cardiogenic shock.21
Back to the Case
Despite maximal medical therapy for her chronic heart failure and biventricular pacing, the patient continued to have markedly limited functional status and repeated hospitalizations for AHFS. Given her advanced age and poor nutritional status, she was not a candidate for cardiac transplantation or placement of a left ventricular assist device. To allow for palliative tailored therapy, right heart catheterization was performed. Right heart catheterization revealed elevated filling pressures, as follows: right atrium, 20 mmHg; pulmonary artery, 63/34 mmHg (mean 47 mmHg); and pulmonary capillary wedge, 29 mmHg. Her mixed venous oxygen saturation was only 41% with a calculated cardiac output of 2.9 liters per minute and cardiac index of 2 liters per minute per meter squared.
As she expressed symptomatic relief as her goal, she was started on intravenous milrinone at 0.2 micrograms per kilogram per minute. This was done with the understanding her symptoms would likely would improve, at the expense of worsening longevity and prognosis. With uptitration of her intravenous milrinone and a continuous infusion of furosemide, she demonstrated the following filling pressures within 24 hours: right atrium, 18 mmHg; pulmonary artery, 63/33 mmHg (mean 43 mmHg); and pulmonary capillary wedge, 24. Importantly, her mixed venous oxygen saturation improved to 68% with a calculated cardiac output of 3.4 liters per minute and cardiac index of 2.4 liters per minute per meter squared. These favorable hemodynamic changes were accompanied by modest improvement in symptoms. After continued intravenous diuresis, she was transitioned back to an oral diuretic regimen and was ultimately discharged to home with a continuous infusion of milrinone for palliation. TH
Drs. Vaishnava, McKean, Nohria, and Baughman are from Brigham and Women’s Hospital and Harvard Medical School in Boston, Mass.
REFERENCES:
- Thom T, Haase N, Rosamond W, et al. Heart disease and stroke statistics – 2006 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2006;113:85-151.
- Iong P, Vowinckel E, Liu PP, Gong Y, Tu JV. Prognosis and determinants of survival in patients newly hospitalized for heart failure: a population-based study. Arch Intern Med. 2002;162:1689-94.
- Nohria A, Lewis EF, Stevenson LW. Medical management of advanced heart failure. JAMA. 2002;287;628-40.
- Faris R, Flather MD, Purcell H, et al. Diuretics for heart failure. Cochrane Database Syst Rev. 2006;1;CD003838.
- Wang DJ and Gottlieb SS. Diuretics: Still the mainstay of treatment. Crit Care Med. 2008;36(Suppl.):S89-S94.
- Fares WH. Management of acute decompensated heart failure in an evidence-based era: What is the evidence behind the current standard of care? Heart & Lung. 2008;37(3):173-8.
- Adams KF, Lindenfield J, Arnold J, et al. Executive summary: HFSA 2006 comprehensive heart failure practice guidelines. J Card Fail. 2006;12:10-38.
- Publication Committee for the VMAC Investigators (Vasodilatation in the Management of Acute CHF). Intravenous nesiritide vs nitroglycerin for treatment of decompensated congestive heart failure: a randomized controlled trial. JAMA. 2002;297:1531-40.
- Peacock WF, Enerman CL, Silver MA, on behalf of the PROACTION Study Group. Am J Emerg Med. 2005;23:327-31.
- Cotter G, Metzkor E, Kaluski E, et al. Randomized trial of high-dose isosorbide dinitrate plus low-dose furosemide versus high-dose furosemide plus low-dose isosorbide dinitrate in severe pulmonary edema. Lancet. 1998;351:389-93.
- Sackner-Bernstein JD, Kowalski M, Fox M, Aaronson K. Short-term risk of death after treatment with nesiritide for decompensated heart failure: a pooled analysis of randomized controlled trials. JAMA. 2005;293:1900-5.
- Sackner-Bernstein JD, Skopicki HA, Aaronson K. Risk of worsening renal function with nesiritide in patients with acutely decompensated heart failure. Circulation. 2005;111:1487-91.
- Felker GM and O’Connor CM. Inotropic therapy for heart failure: An evidence-based approach. American Heart Journal. 2001; 142:393-401.
- Abrahm WT, Adams KF, Fonarow GC, et al. In-hospital mortality in patients with acute decompensated heart failure requiring intravenous vasoactive medications: an analysis from the Acute Decompensated Heart Failure National Registry (ADHERE). J Am Coll Cardiol. 2005;46:57-64.
- Burger AJ, Houton DP, LeJemtel T, et al. Effect of nesiritide and dobutamine on ventricular arrhythmias in the treatment of patients with acutely decompensated congestive heart failure: the PRECEDENT study. American Heart Journal. 2002;144:1102-8.
- Liang CS, Sherman LG, Doherty JU, et al. Sustained improvement of cardiac function in patients with congestive heart failure after short-term infusion of dobutamine. Circulation. 1984;69:113-9.
- Unverferth DV, Magorien RD, Lewis RP, et al. Long-term benefit of dobutamine in patients with congestive cardiomyopathy. American Heart Journal. 1980;100:622-30.
- Cuffe MS, Califf RM, Adams KF Jr, et al. Outcomes of a Prospective Trial of Intravenous Milrinone for Exacerbations of Chronic Heart Failure (OPTIME-CHF) Investigators. Short-term intravenous milrinone for acute exacerbation of chronic heart failure: a randomized controlled trial. JAMA. 2002;287:1541-7.
- Yamani MH, Haji SA, Starling RC, et al. Comparison of dobutamine-based and milrinone-based therapy for advanced decompensated congestive heart failure: Hemodynamic efficacy, clinical outcome, and economic impact. American Heart Journal. 2001;142:998-1002.
- Shin, DD, Brandimarte F, De Luca L, et al. Review of current and investigational pharmacologic agents for acute heart failure syndromes. Am J Cardiol. 2007;99(suppl):4A-23A.
- Kale P and Fang JC. Devices in acute heart failure. Crit Care Med. 2008;36(Suppl.):S121-128.
Case
A 72-year-old retired nurse with known nonischemic dilated cardiomyopathy with an ejection fraction of approximately 20% and status-post cardiac resynchronization therapy presents to the emergency department with dyspnea with minimal activity, three-pillow orthopnea, and paroxysmal nocturnal dyspnea.
She had been hospitalized twice during the past 60 days for similar symptoms. Her medications included losartan (20 mg po q daily), carvedilol (3.125 mg twice daily), spironolactone (25 mg daily), digoxin (0.125 mg daily), and furosemide (80 mg twice daily). Vital signs are notable for a blood pressure of 90/50 mmHg and an irregular pulse of 90 beats per minute. Physical examination is notable for marked jugular venous distension, lungs clear to auscultation bilaterally, biventricular heaves, a markedly displaced left ventricular point of maximal impulse, and a prominent S3 gallop.
Despite treatment with intravenous furosemide and temporary withdrawal of carvedilol, the patient remains symptomatic with persistent jugular venous distension.
Should she be given a vasoactive agent?
Overview
Acute heart failure syndrome (AHFS), defined as a gradual or rapid change in heart failure signs and symptoms, is the most common cause of hospitalization in the United States1. It is associated with an average in-hospital mortality of 4% to 5%, a 30-day mortality of 7% to11%, and a one-year mortality of 33%2.
In patients with previously established myocardial dysfunction, AHFS commonly reflects exacerbation of symptoms after a period of stability. The clinical presentation and severity of AHFS may range from mild volume overload to life-threatening cardiogenic shock and multi-organ failure unresponsive to pharmacologic therapy.2
Initial management of AHFS depends on definition of the patient’s hemodynamic profile. To guide initial therapy, classify patients into one of four hemodynamic profiles during a brief bedside assessment that relies on evaluation of filling pressures (wet or dry) and adequacy of perfusion (hot or cold) (see figure 1).3
Treating volume overload or elevated filling pressures generally begins with diuretics. Diuretics have been shown to provide symptomatic relief, though they have not yet been proven safe.4 Initial treatment can include a loop diuretic at a dose higher than the patient’s chronic dose, with intravenous dosing offering greater bio-absorption and rapidity in onset of action.5 If perfusion is inadequate, escalate therapy beyond diuretics to include vasoactive agents.
Review of the Data
The use of vasoactive medications is largely based on anecdotal experiences and physiologic assumptions rather than on adequately powered prospective randomized controlled trials.6 Vasoactive therapy includes vasodilator and inotropic support and is generally limited for use in patients with advanced disease not responding to standard medical treatment and diuresis. The physiologic premise rests in the expected improvement in ventricular filling pressures and cardiac output with reduction in afterload and/or preload. Vasodilators counteract vascular constriction, reducing both preload and afterload. Positive inotropic agents amplify cardiac output by increasing the strength of myocardial contraction.
Vasodilators
The Heart Failure Society of America (HFSA) 2006 Comprehensive Heart Failure Practice Guidelines state, “In the absence of symptomatic hypotension, intravenous vasodilators (nitroglycerin, nitroprusside, or nesiritide) may be considered as an addition to diuretic therapy for rapid improvement of congestive symptoms.”7 The clinical utility of nesiritide remains in question with clinical and hemodynamic improvement demonstrated in three randomized trials 8-10; but tempered against meta-analyses 11-12 of selected trials, demonstrating a non-significant trend toward increased kidney dysfunction and death within 30 days (35/485 [7.2%] vs. 15/377 [4.0%] patients; risk ratio from meta-analyses, 1.74; 95% confidence interval, 0.97-3.12; p=0.059). In a randomized trial of 489 in-patients with dyspnea at rest from AHFS, treatment with three hours of intravenous nesiritide resulted in a significant improvement in dyspnea compared with placebo (p=0.03). Similar improvement was observed with intravenous nitroglycerin and did not differ statistically from that observed with nesiritide.8 Nitroprusside, an attractive option among those with hypertension and cardiogenic pulmonary edema, is limited by the need for invasive hemodynamic monitoring and potential for either cyanide toxicity or worsening myocardial ischemia.
Inotropes
Again, there is little evidence from adequately powered randomized controlled trials guiding the use of inotropes. Their use is generally limited to the following indications (see figure 2): (1) Short-term treatment for AHFS that is unresponsive to adequate doses of diuretics and especially when associated with systemic hypotension, (2) Bridge to recovery (as following myocarditis) or to definitive treatment (as with transplant), or (3) For palliation when symptomatic relief is the agreed upon goal.13 The HFSA 2006 guideline states: “Intravenous inotropes may be considered to relieve symptoms and improve end-organ function in patients with advanced HF characterized by left ventricle dilation, reduced left ventricular ejection fraction, and diminished peripheral perfusion or end-organ dysfunction, particularly if these patients have marginal systolic blood pressure, have symptomatic hypotension despite adequate filling pressures, or are unresponsive to, or intolerant of, intravenous vasodilators.”7
Dobutamine and milrinone are the most commonly used IV inotropes for the treatment of AHFS and increase contractility by increasing intracellular levels of cyclic adenylate monophosphate (cAMP). Dobutamine is a catechlamine agonist that increases cAMP production through stimulation of adenylate cyclase. Milrinone selectively inhibits phosphodiesterase III, which catalyzes the breakdown of cAMP.
Despite their frequent use when traditional treatments have failed, the data supporting the use of dobutamine and milrinone is limited. The largest registry of patients with AHFS to date associated excess mortality with intravenous inotrope use compared to nitroglycerin or nesiritide.14 In a study population of 255 patients randomized to receive either intravenous nesiritide or intravenous dobutamine, Burger et al.15 demonstrated that dobutamine significantly increased the mean number of ventricular tachycardia events per 24 hours (p=0.001), suggesting increased arrhythmogenicity associated with inotrope use. Nonetheless, in a randomized trial of 15 patients admitted with AHFS, functional class improved in six of eight dobutamine-treated patients, but in only two of seven patients treated with placebo, suggesting clinical improvement as a consequence of inotropic stimulation.16 Unverferth et al. demonstrated a similar sustained functional improvement up to 10 weeks following a 72-hour infusion of intravenous dobutamine. 17
The Outcomes of a Prospective Trial of Intravenous Milrinone for Exacerbations of Chronic Heart Failure trial (OPTIME-CHF), randomized 951 patients with AHFS to receive either intravenous milrinone or placebo within 48 hours of hospitalization.18 Compared to placebo, milrinone was associated with a significant increase in sustained hypotension requiring intervention (10.7% vs. 3.2%; p<.001) and new atrial arrhythmias (4.6% vs. 1.5%; p=0.004), along with a non-significant trend toward increased mortality (3.8% vs. 2.3%; p=0.19). However, as measured by a visual analog scale, milrinone-treated patients reported feeling better than those treated with placebo at 30 days post-randomization (p=0.02).
Although there are not randomized data comparing the efficacy of milrinone and dobutamine in AHFS, a retrospective analysis of 329 patients compared the hemodynamic and clinical effects of these two inotropes.19 Milrinone consistently was associated with a more favorable hemodynamic response, including lower systemic vascular resistance (p=0.01); lower pulmonary artery wedge pressure (p<0.001); larger percentage increase in cardiac index (p=0.03); and larger percentage decrease in pulmonary vascular resistance (p=0.0001). In-hospital mortality (dobutamine 7.8% vs. milrinone 10%) was not significantly different.
Conclusion
Clearly, vasoactive and inotropic agents are available when AHFS is refractory to traditional diuresis and may offer short-term symptomatic relief, palliation in the context of end-of-life care, or bridge to recovery or more definitive treatment. Unfortunately, sufficient and robust evidence that supports the safety and efficacy of such agents is lacking and their use is largely guided by historical practices, clinical experience, and anticipation of theoretic physiologic changes. While adequately powered prospective randomized data emerge, newer agents such as vasopressin receptor antagonists, cardiac myosin activators, calcium sensitizers, and adenosine-receptor antagonists will offer additional pharmacologic options.20 When continued pharmacologic support becomes ineffective, device therapy is available to aid in the treatment of AHFS and includes ultrafiltration to reduce filling pressures and intra-aortic balloon pump counterpulsation or left ventricular assist device placement for pharmacologically resistant cardiogenic shock.21
Back to the Case
Despite maximal medical therapy for her chronic heart failure and biventricular pacing, the patient continued to have markedly limited functional status and repeated hospitalizations for AHFS. Given her advanced age and poor nutritional status, she was not a candidate for cardiac transplantation or placement of a left ventricular assist device. To allow for palliative tailored therapy, right heart catheterization was performed. Right heart catheterization revealed elevated filling pressures, as follows: right atrium, 20 mmHg; pulmonary artery, 63/34 mmHg (mean 47 mmHg); and pulmonary capillary wedge, 29 mmHg. Her mixed venous oxygen saturation was only 41% with a calculated cardiac output of 2.9 liters per minute and cardiac index of 2 liters per minute per meter squared.
As she expressed symptomatic relief as her goal, she was started on intravenous milrinone at 0.2 micrograms per kilogram per minute. This was done with the understanding her symptoms would likely would improve, at the expense of worsening longevity and prognosis. With uptitration of her intravenous milrinone and a continuous infusion of furosemide, she demonstrated the following filling pressures within 24 hours: right atrium, 18 mmHg; pulmonary artery, 63/33 mmHg (mean 43 mmHg); and pulmonary capillary wedge, 24. Importantly, her mixed venous oxygen saturation improved to 68% with a calculated cardiac output of 3.4 liters per minute and cardiac index of 2.4 liters per minute per meter squared. These favorable hemodynamic changes were accompanied by modest improvement in symptoms. After continued intravenous diuresis, she was transitioned back to an oral diuretic regimen and was ultimately discharged to home with a continuous infusion of milrinone for palliation. TH
Drs. Vaishnava, McKean, Nohria, and Baughman are from Brigham and Women’s Hospital and Harvard Medical School in Boston, Mass.
REFERENCES:
- Thom T, Haase N, Rosamond W, et al. Heart disease and stroke statistics – 2006 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2006;113:85-151.
- Iong P, Vowinckel E, Liu PP, Gong Y, Tu JV. Prognosis and determinants of survival in patients newly hospitalized for heart failure: a population-based study. Arch Intern Med. 2002;162:1689-94.
- Nohria A, Lewis EF, Stevenson LW. Medical management of advanced heart failure. JAMA. 2002;287;628-40.
- Faris R, Flather MD, Purcell H, et al. Diuretics for heart failure. Cochrane Database Syst Rev. 2006;1;CD003838.
- Wang DJ and Gottlieb SS. Diuretics: Still the mainstay of treatment. Crit Care Med. 2008;36(Suppl.):S89-S94.
- Fares WH. Management of acute decompensated heart failure in an evidence-based era: What is the evidence behind the current standard of care? Heart & Lung. 2008;37(3):173-8.
- Adams KF, Lindenfield J, Arnold J, et al. Executive summary: HFSA 2006 comprehensive heart failure practice guidelines. J Card Fail. 2006;12:10-38.
- Publication Committee for the VMAC Investigators (Vasodilatation in the Management of Acute CHF). Intravenous nesiritide vs nitroglycerin for treatment of decompensated congestive heart failure: a randomized controlled trial. JAMA. 2002;297:1531-40.
- Peacock WF, Enerman CL, Silver MA, on behalf of the PROACTION Study Group. Am J Emerg Med. 2005;23:327-31.
- Cotter G, Metzkor E, Kaluski E, et al. Randomized trial of high-dose isosorbide dinitrate plus low-dose furosemide versus high-dose furosemide plus low-dose isosorbide dinitrate in severe pulmonary edema. Lancet. 1998;351:389-93.
- Sackner-Bernstein JD, Kowalski M, Fox M, Aaronson K. Short-term risk of death after treatment with nesiritide for decompensated heart failure: a pooled analysis of randomized controlled trials. JAMA. 2005;293:1900-5.
- Sackner-Bernstein JD, Skopicki HA, Aaronson K. Risk of worsening renal function with nesiritide in patients with acutely decompensated heart failure. Circulation. 2005;111:1487-91.
- Felker GM and O’Connor CM. Inotropic therapy for heart failure: An evidence-based approach. American Heart Journal. 2001; 142:393-401.
- Abrahm WT, Adams KF, Fonarow GC, et al. In-hospital mortality in patients with acute decompensated heart failure requiring intravenous vasoactive medications: an analysis from the Acute Decompensated Heart Failure National Registry (ADHERE). J Am Coll Cardiol. 2005;46:57-64.
- Burger AJ, Houton DP, LeJemtel T, et al. Effect of nesiritide and dobutamine on ventricular arrhythmias in the treatment of patients with acutely decompensated congestive heart failure: the PRECEDENT study. American Heart Journal. 2002;144:1102-8.
- Liang CS, Sherman LG, Doherty JU, et al. Sustained improvement of cardiac function in patients with congestive heart failure after short-term infusion of dobutamine. Circulation. 1984;69:113-9.
- Unverferth DV, Magorien RD, Lewis RP, et al. Long-term benefit of dobutamine in patients with congestive cardiomyopathy. American Heart Journal. 1980;100:622-30.
- Cuffe MS, Califf RM, Adams KF Jr, et al. Outcomes of a Prospective Trial of Intravenous Milrinone for Exacerbations of Chronic Heart Failure (OPTIME-CHF) Investigators. Short-term intravenous milrinone for acute exacerbation of chronic heart failure: a randomized controlled trial. JAMA. 2002;287:1541-7.
- Yamani MH, Haji SA, Starling RC, et al. Comparison of dobutamine-based and milrinone-based therapy for advanced decompensated congestive heart failure: Hemodynamic efficacy, clinical outcome, and economic impact. American Heart Journal. 2001;142:998-1002.
- Shin, DD, Brandimarte F, De Luca L, et al. Review of current and investigational pharmacologic agents for acute heart failure syndromes. Am J Cardiol. 2007;99(suppl):4A-23A.
- Kale P and Fang JC. Devices in acute heart failure. Crit Care Med. 2008;36(Suppl.):S121-128.
In reply: VTE prevention in major orthopedic surgery
Editor's Note: This letter concerns an article in a Cleveland Clinic Journal of Medicine supplement (Preventing Venous Thromboembolism Throughout the Continuum of Care) distributed to only a portion of the Journal's regular readership, owing to the terms of the grant supporting the supplement.
In Reply: We appreciate the comments by Drs. Fishmann and Boyd, but we strongly disagree with their suggestion that aspirin monotherapy is an appropriate option for the prevention of venous thromboembolism (VTE) following major orthopedic surgery.
As discussed in our original article,1 multiple large-scale clinical trials in patients undergoing elective hip arthroplasty, knee arthroplasty, or hip fracture surgery have demonstrated the thromboprophylactic efficacy of warfarin, unfractionated heparin, low-molecular-weight heparin (LMWH), fondaparinux, and oral direct thrombin inhibitors. The relative risk reduction with these agents has been greater than 50% in most studies. In contrast, in a large meta-analysis of VTE prophylaxis following total hip replacement, which included data from 56 randomized trials published between 1966 and 1993, aspirin was not beneficial in preventing DVT.14
The largest prospective randomized trial comparing aspirin with placebo for VTE prevention was conducted between 1992 and 1998 among 17,444 patients in five countries.5 It involved 13,356 patients requiring hip fracture surgery and 4,088 patients requiring elective hip arthroplasty. Patients were randomized to receive aspirin 160 mg/day or placebo for 35 days. However, additional forms of VTE prophylaxis were allowed if deemed necessary by the clinician. In fact, 26% of patients received LMWH in addition to aspirin, and dual therapy was probably more common in those patients at highest thromboembolic risk. As such, the 36% relative risk reduction in VTE ascribed to aspirin should be viewed with caution. Further, this is a smaller risk reduction than that observed in trials of other anticoagulant agents.
A large, well-designed, randomized clinical trial comparing aspirin to LMWH or fondaparinux remains to be conducted.
Dr. Fishmann cites a small study of patients undergoing knee arthroplasty who received spinal anesthesia and intermittent calf compression devices.7 In this underpowered study, 275 patients were randomized to receive aspirin 325 mg twice daily or enoxaparin 30 mg twice daily for 3 weeks. The overall DVT rates were 14.1% in the enoxaparin group vs 17.8% in the aspirin group (P = .27).7 Patients who received aspirin had significantly more postoperative drainage than those randomized to enoxaparin. In addition, the protocol for scheduling enoxaparin 48 hours postoperatively is not consistent with recommendations of the American College of Chest Physicians (ACCP) and may have reduced the efficacy of enoxaparin.
The other evidence in support of aspirin cited by Dr. Fishmann includes an editorial,9 an uncontrolled retrospective analysis,8 a single-center retrospective review,10 and a review article.6 Although there is evidence that the use of aspirin is probably associated with a modest reduction in postoperative VTE risk, it has been unequivocally surpassed in efficacy by other anticoagulants.
Both the latest (2004) ACCP guidelines on VTE2 and the 2006 International Consensus Statement on VTE prevention and treatment15 advise against aspirin monotherapy as VTE prophylaxis in any patient groups. It is likely that the upcoming 2008 ACCP guidelines will also advocate against using aspirin as well.
Lastly, the most recent guideline from the American Academy of Orthopaedic Surgeons advocating aspirin as monotherapy11 is based on the assumption that the major important clinical end point in the orthopedic surgery patient is clinical pulmonary embolism, an end point that was not included as a lone primary end point in any of the modern randomized controlled studies in major orthopedic surgery. This represents a flawed logic for the development of evidence-based guideline recommendations, and this recommendation has not been advocated by well-respected bodies such as the ACCP and the international groups that developed the International Consensus Statement. Furthermore, if this practice is going to be advocated by the American Academy of Orthopaedic Surgeons, then large rigorously designed randomized trials must be conducted to compare aspirin to currently available anticoagulants, and the type of joint surgery should be clearly defined.
- Deitelzweig SB, McKean SC, Amin AN, Brotman DJ, Jaffer AK, Spyropoulos AC. Prevention of venous thromboembolism in the orthopedic surgery patient. Cleve Clin J Med 2008; 75(suppl 3):S27–S36.
- Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 2004; 126(3 suppl):338S–400S.
- Geerts WH, Heit JA, Clagett GP, et al. Prevention of venous thromboembolism. Chest 2001; 119(1 suppl):132S–175S.
- Zimlich RH, Fulbright BM, Friedman RJ. Current status of anticoagulation therapy after total hip and total knee arthroplasty. J Am Acad Orthop Surg 1996; 4:54–62.
- PEP Trial Collaborative Group. Prevention of pulmonary embolism and deep vein thrombosis with low dose aspirin: Pulmonary Embolism Prevention (PEP) trial. Lancet 2000; 355:1295–1302.
- Berend KR, Lombardi AV. Multimodal venous thromboembolic disease prevention for patients undergoing primary or revision total joint arthroplasty: the role of aspirin. Am J Orthop 2006; 35:24–29.
- Westrich GH, Bottner F, Windsor RE, Laskin RS, Haas SB, Sculco TP. VenaFlow plus Lovenox vs VenaFlow plus aspirin for thromboembolic disease prophylaxis in total knee arthroplasty. J Arthroplasty 2006; 21(6 suppl 2):139–143.
- Lotke PA, Lonner JH. The benefit of aspirin chemoprophylaxis for thromboembolism after total knee arthroplasty. Clin Orthop Relat Res 2006; 452:175–180.
- Callaghan JJ, Dorr LD, Engh GA, et al. Prophylaxis for thromboembolic disease: recommendations from the American College of Chest Physicians—are they appropriate for orthopaedic surgery? J Arthroplasty 2005; 20:273–274.
- Dorr LD, Gendelman V, Maheshwari AV, Boutary M, Wan Z, Long WT. Multimodal thromboprophylaxis for total hip and knee arthroplasty based on risk assessment. J Bone Joint Surg Am 2007; 89:2648–2657.
- American Academy of Orthopaedic Surgeons Clinical Guideline on Prevention of Symptomatic Pulmonary Embolism in Patients Undergoing Total Hip or Knee Arthroplasty: Summary of Recommendations. http://www.aaos.org/Research/guidelines/PE_summary.pdf. Accessed April 16, 2008.
- Parvizi J, Ghanem E, Joshi A, Sharkey PF, Hozack WJ, Rothman RH. Does “excessive” anticoagulation predispose to periprosthetic infection? J Arthroplasty 2007; 22(6 suppl 2):24–28.
- Bern M, Deshmukh RV, Nelson R, et al. Low-dose warfarin coupled with lower leg compression is effective prophylaxis against thromboembolic disease after hip arthroplasty. J Arthroplasty 2007; 22:644–650.
- Imperiale TF, Speroff T. A meta-analysis of methods to prevent venous thromboembolism following total hip replacement. JAMA 1994; 271:1780–1785.
- Cardiovascular Disease Educational and Research Trust; Cyprus Cardiovascular Disease Educational and Research Trust; European Venous Forum; International Surgical Thrombosis Forum; International Union of Angiology; Union Internationale de Phlébologie. Prevention and treatment of venous thromboembolism. International Consensus Statement (guidelines according to scientific evidence). Int Angiol 2006; 25:101–161.
Editor's Note: This letter concerns an article in a Cleveland Clinic Journal of Medicine supplement (Preventing Venous Thromboembolism Throughout the Continuum of Care) distributed to only a portion of the Journal's regular readership, owing to the terms of the grant supporting the supplement.
In Reply: We appreciate the comments by Drs. Fishmann and Boyd, but we strongly disagree with their suggestion that aspirin monotherapy is an appropriate option for the prevention of venous thromboembolism (VTE) following major orthopedic surgery.
As discussed in our original article,1 multiple large-scale clinical trials in patients undergoing elective hip arthroplasty, knee arthroplasty, or hip fracture surgery have demonstrated the thromboprophylactic efficacy of warfarin, unfractionated heparin, low-molecular-weight heparin (LMWH), fondaparinux, and oral direct thrombin inhibitors. The relative risk reduction with these agents has been greater than 50% in most studies. In contrast, in a large meta-analysis of VTE prophylaxis following total hip replacement, which included data from 56 randomized trials published between 1966 and 1993, aspirin was not beneficial in preventing DVT.14
The largest prospective randomized trial comparing aspirin with placebo for VTE prevention was conducted between 1992 and 1998 among 17,444 patients in five countries.5 It involved 13,356 patients requiring hip fracture surgery and 4,088 patients requiring elective hip arthroplasty. Patients were randomized to receive aspirin 160 mg/day or placebo for 35 days. However, additional forms of VTE prophylaxis were allowed if deemed necessary by the clinician. In fact, 26% of patients received LMWH in addition to aspirin, and dual therapy was probably more common in those patients at highest thromboembolic risk. As such, the 36% relative risk reduction in VTE ascribed to aspirin should be viewed with caution. Further, this is a smaller risk reduction than that observed in trials of other anticoagulant agents.
A large, well-designed, randomized clinical trial comparing aspirin to LMWH or fondaparinux remains to be conducted.
Dr. Fishmann cites a small study of patients undergoing knee arthroplasty who received spinal anesthesia and intermittent calf compression devices.7 In this underpowered study, 275 patients were randomized to receive aspirin 325 mg twice daily or enoxaparin 30 mg twice daily for 3 weeks. The overall DVT rates were 14.1% in the enoxaparin group vs 17.8% in the aspirin group (P = .27).7 Patients who received aspirin had significantly more postoperative drainage than those randomized to enoxaparin. In addition, the protocol for scheduling enoxaparin 48 hours postoperatively is not consistent with recommendations of the American College of Chest Physicians (ACCP) and may have reduced the efficacy of enoxaparin.
The other evidence in support of aspirin cited by Dr. Fishmann includes an editorial,9 an uncontrolled retrospective analysis,8 a single-center retrospective review,10 and a review article.6 Although there is evidence that the use of aspirin is probably associated with a modest reduction in postoperative VTE risk, it has been unequivocally surpassed in efficacy by other anticoagulants.
Both the latest (2004) ACCP guidelines on VTE2 and the 2006 International Consensus Statement on VTE prevention and treatment15 advise against aspirin monotherapy as VTE prophylaxis in any patient groups. It is likely that the upcoming 2008 ACCP guidelines will also advocate against using aspirin as well.
Lastly, the most recent guideline from the American Academy of Orthopaedic Surgeons advocating aspirin as monotherapy11 is based on the assumption that the major important clinical end point in the orthopedic surgery patient is clinical pulmonary embolism, an end point that was not included as a lone primary end point in any of the modern randomized controlled studies in major orthopedic surgery. This represents a flawed logic for the development of evidence-based guideline recommendations, and this recommendation has not been advocated by well-respected bodies such as the ACCP and the international groups that developed the International Consensus Statement. Furthermore, if this practice is going to be advocated by the American Academy of Orthopaedic Surgeons, then large rigorously designed randomized trials must be conducted to compare aspirin to currently available anticoagulants, and the type of joint surgery should be clearly defined.
Editor's Note: This letter concerns an article in a Cleveland Clinic Journal of Medicine supplement (Preventing Venous Thromboembolism Throughout the Continuum of Care) distributed to only a portion of the Journal's regular readership, owing to the terms of the grant supporting the supplement.
In Reply: We appreciate the comments by Drs. Fishmann and Boyd, but we strongly disagree with their suggestion that aspirin monotherapy is an appropriate option for the prevention of venous thromboembolism (VTE) following major orthopedic surgery.
As discussed in our original article,1 multiple large-scale clinical trials in patients undergoing elective hip arthroplasty, knee arthroplasty, or hip fracture surgery have demonstrated the thromboprophylactic efficacy of warfarin, unfractionated heparin, low-molecular-weight heparin (LMWH), fondaparinux, and oral direct thrombin inhibitors. The relative risk reduction with these agents has been greater than 50% in most studies. In contrast, in a large meta-analysis of VTE prophylaxis following total hip replacement, which included data from 56 randomized trials published between 1966 and 1993, aspirin was not beneficial in preventing DVT.14
The largest prospective randomized trial comparing aspirin with placebo for VTE prevention was conducted between 1992 and 1998 among 17,444 patients in five countries.5 It involved 13,356 patients requiring hip fracture surgery and 4,088 patients requiring elective hip arthroplasty. Patients were randomized to receive aspirin 160 mg/day or placebo for 35 days. However, additional forms of VTE prophylaxis were allowed if deemed necessary by the clinician. In fact, 26% of patients received LMWH in addition to aspirin, and dual therapy was probably more common in those patients at highest thromboembolic risk. As such, the 36% relative risk reduction in VTE ascribed to aspirin should be viewed with caution. Further, this is a smaller risk reduction than that observed in trials of other anticoagulant agents.
A large, well-designed, randomized clinical trial comparing aspirin to LMWH or fondaparinux remains to be conducted.
Dr. Fishmann cites a small study of patients undergoing knee arthroplasty who received spinal anesthesia and intermittent calf compression devices.7 In this underpowered study, 275 patients were randomized to receive aspirin 325 mg twice daily or enoxaparin 30 mg twice daily for 3 weeks. The overall DVT rates were 14.1% in the enoxaparin group vs 17.8% in the aspirin group (P = .27).7 Patients who received aspirin had significantly more postoperative drainage than those randomized to enoxaparin. In addition, the protocol for scheduling enoxaparin 48 hours postoperatively is not consistent with recommendations of the American College of Chest Physicians (ACCP) and may have reduced the efficacy of enoxaparin.
The other evidence in support of aspirin cited by Dr. Fishmann includes an editorial,9 an uncontrolled retrospective analysis,8 a single-center retrospective review,10 and a review article.6 Although there is evidence that the use of aspirin is probably associated with a modest reduction in postoperative VTE risk, it has been unequivocally surpassed in efficacy by other anticoagulants.
Both the latest (2004) ACCP guidelines on VTE2 and the 2006 International Consensus Statement on VTE prevention and treatment15 advise against aspirin monotherapy as VTE prophylaxis in any patient groups. It is likely that the upcoming 2008 ACCP guidelines will also advocate against using aspirin as well.
Lastly, the most recent guideline from the American Academy of Orthopaedic Surgeons advocating aspirin as monotherapy11 is based on the assumption that the major important clinical end point in the orthopedic surgery patient is clinical pulmonary embolism, an end point that was not included as a lone primary end point in any of the modern randomized controlled studies in major orthopedic surgery. This represents a flawed logic for the development of evidence-based guideline recommendations, and this recommendation has not been advocated by well-respected bodies such as the ACCP and the international groups that developed the International Consensus Statement. Furthermore, if this practice is going to be advocated by the American Academy of Orthopaedic Surgeons, then large rigorously designed randomized trials must be conducted to compare aspirin to currently available anticoagulants, and the type of joint surgery should be clearly defined.
- Deitelzweig SB, McKean SC, Amin AN, Brotman DJ, Jaffer AK, Spyropoulos AC. Prevention of venous thromboembolism in the orthopedic surgery patient. Cleve Clin J Med 2008; 75(suppl 3):S27–S36.
- Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 2004; 126(3 suppl):338S–400S.
- Geerts WH, Heit JA, Clagett GP, et al. Prevention of venous thromboembolism. Chest 2001; 119(1 suppl):132S–175S.
- Zimlich RH, Fulbright BM, Friedman RJ. Current status of anticoagulation therapy after total hip and total knee arthroplasty. J Am Acad Orthop Surg 1996; 4:54–62.
- PEP Trial Collaborative Group. Prevention of pulmonary embolism and deep vein thrombosis with low dose aspirin: Pulmonary Embolism Prevention (PEP) trial. Lancet 2000; 355:1295–1302.
- Berend KR, Lombardi AV. Multimodal venous thromboembolic disease prevention for patients undergoing primary or revision total joint arthroplasty: the role of aspirin. Am J Orthop 2006; 35:24–29.
- Westrich GH, Bottner F, Windsor RE, Laskin RS, Haas SB, Sculco TP. VenaFlow plus Lovenox vs VenaFlow plus aspirin for thromboembolic disease prophylaxis in total knee arthroplasty. J Arthroplasty 2006; 21(6 suppl 2):139–143.
- Lotke PA, Lonner JH. The benefit of aspirin chemoprophylaxis for thromboembolism after total knee arthroplasty. Clin Orthop Relat Res 2006; 452:175–180.
- Callaghan JJ, Dorr LD, Engh GA, et al. Prophylaxis for thromboembolic disease: recommendations from the American College of Chest Physicians—are they appropriate for orthopaedic surgery? J Arthroplasty 2005; 20:273–274.
- Dorr LD, Gendelman V, Maheshwari AV, Boutary M, Wan Z, Long WT. Multimodal thromboprophylaxis for total hip and knee arthroplasty based on risk assessment. J Bone Joint Surg Am 2007; 89:2648–2657.
- American Academy of Orthopaedic Surgeons Clinical Guideline on Prevention of Symptomatic Pulmonary Embolism in Patients Undergoing Total Hip or Knee Arthroplasty: Summary of Recommendations. http://www.aaos.org/Research/guidelines/PE_summary.pdf. Accessed April 16, 2008.
- Parvizi J, Ghanem E, Joshi A, Sharkey PF, Hozack WJ, Rothman RH. Does “excessive” anticoagulation predispose to periprosthetic infection? J Arthroplasty 2007; 22(6 suppl 2):24–28.
- Bern M, Deshmukh RV, Nelson R, et al. Low-dose warfarin coupled with lower leg compression is effective prophylaxis against thromboembolic disease after hip arthroplasty. J Arthroplasty 2007; 22:644–650.
- Imperiale TF, Speroff T. A meta-analysis of methods to prevent venous thromboembolism following total hip replacement. JAMA 1994; 271:1780–1785.
- Cardiovascular Disease Educational and Research Trust; Cyprus Cardiovascular Disease Educational and Research Trust; European Venous Forum; International Surgical Thrombosis Forum; International Union of Angiology; Union Internationale de Phlébologie. Prevention and treatment of venous thromboembolism. International Consensus Statement (guidelines according to scientific evidence). Int Angiol 2006; 25:101–161.
- Deitelzweig SB, McKean SC, Amin AN, Brotman DJ, Jaffer AK, Spyropoulos AC. Prevention of venous thromboembolism in the orthopedic surgery patient. Cleve Clin J Med 2008; 75(suppl 3):S27–S36.
- Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 2004; 126(3 suppl):338S–400S.
- Geerts WH, Heit JA, Clagett GP, et al. Prevention of venous thromboembolism. Chest 2001; 119(1 suppl):132S–175S.
- Zimlich RH, Fulbright BM, Friedman RJ. Current status of anticoagulation therapy after total hip and total knee arthroplasty. J Am Acad Orthop Surg 1996; 4:54–62.
- PEP Trial Collaborative Group. Prevention of pulmonary embolism and deep vein thrombosis with low dose aspirin: Pulmonary Embolism Prevention (PEP) trial. Lancet 2000; 355:1295–1302.
- Berend KR, Lombardi AV. Multimodal venous thromboembolic disease prevention for patients undergoing primary or revision total joint arthroplasty: the role of aspirin. Am J Orthop 2006; 35:24–29.
- Westrich GH, Bottner F, Windsor RE, Laskin RS, Haas SB, Sculco TP. VenaFlow plus Lovenox vs VenaFlow plus aspirin for thromboembolic disease prophylaxis in total knee arthroplasty. J Arthroplasty 2006; 21(6 suppl 2):139–143.
- Lotke PA, Lonner JH. The benefit of aspirin chemoprophylaxis for thromboembolism after total knee arthroplasty. Clin Orthop Relat Res 2006; 452:175–180.
- Callaghan JJ, Dorr LD, Engh GA, et al. Prophylaxis for thromboembolic disease: recommendations from the American College of Chest Physicians—are they appropriate for orthopaedic surgery? J Arthroplasty 2005; 20:273–274.
- Dorr LD, Gendelman V, Maheshwari AV, Boutary M, Wan Z, Long WT. Multimodal thromboprophylaxis for total hip and knee arthroplasty based on risk assessment. J Bone Joint Surg Am 2007; 89:2648–2657.
- American Academy of Orthopaedic Surgeons Clinical Guideline on Prevention of Symptomatic Pulmonary Embolism in Patients Undergoing Total Hip or Knee Arthroplasty: Summary of Recommendations. http://www.aaos.org/Research/guidelines/PE_summary.pdf. Accessed April 16, 2008.
- Parvizi J, Ghanem E, Joshi A, Sharkey PF, Hozack WJ, Rothman RH. Does “excessive” anticoagulation predispose to periprosthetic infection? J Arthroplasty 2007; 22(6 suppl 2):24–28.
- Bern M, Deshmukh RV, Nelson R, et al. Low-dose warfarin coupled with lower leg compression is effective prophylaxis against thromboembolic disease after hip arthroplasty. J Arthroplasty 2007; 22:644–650.
- Imperiale TF, Speroff T. A meta-analysis of methods to prevent venous thromboembolism following total hip replacement. JAMA 1994; 271:1780–1785.
- Cardiovascular Disease Educational and Research Trust; Cyprus Cardiovascular Disease Educational and Research Trust; European Venous Forum; International Surgical Thrombosis Forum; International Union of Angiology; Union Internationale de Phlébologie. Prevention and treatment of venous thromboembolism. International Consensus Statement (guidelines according to scientific evidence). Int Angiol 2006; 25:101–161.
Prevention of venous thromboembolism in the hospitalized medical patient
The need for prophylaxis of venous thromboembolism (VTE) in hospitalized acutely ill medical patients is well established. Without prophylaxis, hospitalized medical patients develop VTE at a rate of 5% to 15%.1–3 Moreover, pulmonary embolism (PE) occurs more frequently in hospitalized medical patients than in nonmedical patients, and is a leading cause of sudden death in hospitalized medical patients.4,5 Without appropriate prophylaxis, 1 in 20 hospitalized medical patients may suffer a fatal PE.4
PROPHYLAXIS IN MEDICAL PATIENTS: UNDERUSED AND OFTEN INAPPROPRIATE
Despite these risks and the clear indications for VTE prophylaxis in hospitalized medical patients, prophylaxis of VTE is omitted more often in these patients than in hospitalized surgical patients.5 Even when prophylaxis is given, it is often used inappropriately in the medical population. So concludes a recent analysis of data from 196,104 patients with acute medical conditions who were discharged from 227 US hospitals from January 2002 to September 2005.6 Criteria for inclusion in the analysis were patient age of 40 years or older, a hospital stay of 6 days or greater, and an absence of contraindications to anticoagulation. Appropriate prophylaxis was defined in accordance with the Sixth American College of Chest Physicians (ACCP) Consensus Conference on Antithrombotic Therapy.7
The analysis revealed an overall VTE prophylaxis rate of 61.8%, but the rate of appropriate prophylaxis was only 33.9%, meaning that two-thirds of discharged patients did not receive prophylaxis in accordance with ACCP guidelines. When temporal trends were analyzed according to groups based on patients’ diagnosis at admission (acute myocardial infarction, severe lung disease, ischemic stroke, cancer, heart failure, or trauma), the rate of appropriate prophylaxis remained essentially flat from the beginning to the end of the study period for virtually all diagnosis groups.6
Similar findings have emerged from the International Medical Prevention Registry on Venous Thromboembolism (IMPROVE), an ongoing international registry of acutely ill medical patients.8 Data from the first 15,156 patients, enrolled from July 2002 through September 2006, reveal that 50% of patients received pharmacologic and/or mechanical VTE prophylaxis in the hospital, and only 60% of patients who met established criteria for VTE prophylaxis actually received it.
Analysis of the US portion of the IMPROVE data shows that 54% of the US patient sample received some form of VTE prophylaxis; 22% of US patients received intermittent pneumatic compression, 21% received unfractionated heparin (UFH), 14% received low-molecular-weight heparin (LMWH), and 3% wore compression stockings.8 Thus, despite a paucity of data supporting a benefit of intermittent pneumatic compression in this population,9 it was the most frequently used form of prophylaxis in US patients.
CLINICAL TRIALS OF PHARMACOLOGIC PROPHYLAXIS IN MEDICAL PATIENTS
The Prophylaxis in Medical Patients with Enoxaparin (MEDENOX) trial1 randomized 1,102 hospitalized patients to one of two doses of the LMWH enoxaparin (20 mg or 40 mg once daily subcutaneously) or placebo for 6 to 14 days. Compared with placebo, the 40-mg dose of enoxaparin was associated with a 63% reduction in risk of VTE over 3 months of follow-up (P < .001) (Figure 1).
The Prospective Evaluation of Dalteparin Efficacy for Prevention of VTE in Immobilized Patients Trial (PREVENT)2 was a multicenter, randomized, double-blind study comparing the LMWH dalteparin (5,000 IU daily given subcutaneously for 14 days) with placebo in 3,706 acutely ill medical patients. Over 90 days of follow-up, the risk of VTE was reduced by 44% in patients assigned to dalteparin compared with those assigned to placebo (P = .0015) (Figure 1).
The Arixtra for Thromboembolism Prevention in a Medical Indications Study (ARTEMIS)3 randomized 849 medical patients 60 years or older to 6 to 14 days of therapy with the selective factor Xa inhibitor fondaparinux (2.5 mg once daily subcutaneously) or placebo. Compared with the placebo group, fondaparinux recipients had a 47% lower risk of developing VTE by day 15 (P = .029) (Figure 1).
Fewer events and fatal PEs, but no effect on all-cause mortality
A recent meta-analysis by Dentali et al10 further demonstrates the efficacy of anticoagulant therapy for preventing symptomatic VTE in hospitalized medical patients. This analysis included several other trials in addition to the three reviewed above,1–3 for a total of nine randomized studies (seven of which were dou-ble-blind) comprising 19,958 patients. Across the nine studies, anticoagulant prophylaxis was clearly superior to placebo in preventing fatal PE (relative risk, 0.38 [95% CI, 0.21 to 0.69]). There was a strong trend toward a reduction in symptomatic deep vein thrombosis (DVT) with prophylaxis but no effect on all-cause mortality. The meta-analysis also provided reassurance that prophylaxis does not increase the rate of major bleeding.
HOW DO THE PROPHYLAXIS OPTIONS STACK UP?
What the ACCP recommends
Current ACCP guidelines recommend the use of either LMWH or low-dose UFH (5,000 U subcutaneously two or three times daily) as a Grade 1A recommendation for VTE prophylaxis in patients with medical conditions and risk factors for VTE.9 This represents the guidelines’ highest level of recommendation, ie, one that is based on randomized controlled trials (RCTs) without important limitations. In contrast, the 2006 International Consensus Statement, developed as a collaborative effort among expert bodies on VTE, specified a more narrow dosing recommendation for UFH in this patient population (5,000 U three times daily, not twice daily) as well as specifying 40 mg once daily as the recommended dose of enoxaparin and 5,000 IU once daily as the recommended dose of dalteparin.11
For medical patients with risk factors for VTE who have a contraindication to anticoagulant prophylaxis, the ACCP guidelines recommend the use of graduated compression stockings or intermittent pneumatic compression devices as a Grade 1C+ recommendation (“no RCTs but strong RCT results can be unequivocally extrapolated, or overwhelming evidence from observational studies”9).
Current ACCP guidelines do not address the use of fondaparinux in their recommendations for VTE prophylaxis in medical patients.
Getting a handle on bleeding risk
Patient characteristics that exclude pharmacologic thromboprophylaxis due to bleeding risk are generally limited to active bleeding or coagulopathy, as demonstrated by a platelet count less than 50,000 cells/µL or an international normalized ratio greater than 1.5. Additionally, bleeding risk should be carefully assessed if an invasive procedure is planned during a patient’s hospital stay.
It is worth noting that the anticoagulant doses used for VTE prophylaxis are a fraction of those used for treatment of VTE. Thus, if a patient would be treated with full-dose anticoagulation if VTE developed, then that patient should be eligible for VTE prophylaxis.
Because the use of mechanical forms of prophylaxis in medical patients is not truly evidence-based, mechanical prophylaxis should be reserved for medical patients who have a contraindication to anticoagulants, or for use in combination with anticoagulants in patients at very high risk of VTE.
UFH vs LMWH
Two meta-analyses have compared UFH with LMWH for VTE prevention in medical patients.12,13 In a recent analysis that included 10 trials directly comparing the two therapies, 14 trials comparing UFH with control, and 11 trials comparing LMWH with control, Wein et al found a lower risk of DVT with LMWH than with UFH (relative risk, 0.68 [95% CI, 0.52 to 0.88]) but no difference between the therapies in mortality or bleeding risk.12 In an earlier and smaller analysis, Mismetti et al found no significant differences between UFH and LMWH in preventing DVT or death but did find a significant reduction in major bleeding episodes with LMWH versus three-times-daily UFH (52% relative reduction; P = .049).13
Randomized trials also reveal that enoxaparin 40 mg once daily is as efficacious as UFH 5,000 U three times daily for VTE prevention in medical patients.14,15 The above analysis by Wein et al12 and an additional meta-analysis by King and colleagues16 found that three-times-daily dosing of UFH is more efficacious than twice-daily dosing of UFH, but at the expense of more bleeding, including major bleeding.
Economic considerations
Because of differences in drug acquisition costs between UFH and the LMWH agents, several economic evaluations have compared the use of these therapies for prophylaxis in medical patients at risk of VTE.
In an analysis of hospital costs for medical patients receiving VTE prophylaxis from more than 330 US hospitals for the period 2001–2004, Burleigh et al found that mean total hospital costs were higher for patients who received UFH than for those who received LMWH ($7,615 vs $6,866) even though mean drug costs were higher for LMWH ($791 vs $569 for UFH).17 A reduction in hospital length of stay appeared to contribute to the overall savings with LMWH; other contributors may have included costs associated with heparin-induced thrombocytopenia (HIT) in UFH recipients or the extra nursing time required for administering UFH in two or three daily doses.
Leykum et al used a decision analysis model to estimate the economic effect of substituting enoxaparin for UFH in hospitalized medical patients for whom VTE prophylaxis is indicated.18 Cost data were based on Medicare reimbursement rates as well as drug and laboratory costs for a multi-institutional health system. The model assumed HIT incidence rates of 2.7% with UFH and 0.3% with enoxaparin. It also assumed the cost of a daily dose to be $4 for UFH versus $84 for enoxaparin. From the payer perspective, the model showed that substituting enoxaparin for UFH would reduce the overall cost of care by $28.61 per day on a per-patient basis, despite enoxaparin’s higher acquisition cost, and would save $4,550 per quality-adjusted life-year by reducing the incidence of HIT.
Another cost analysis confirms the association between HIT and increased hospital costs. Creekmore et al retrospectively analyzed data from 10,121 adult medical patients who received VTE prophylaxis at the University of Utah Hospital in Salt Lake City from August 2000 to November 2004.19 They found that an admission during which HIT developed incurred a mean cost of $56,364, compared with $15,231 for an admission without HIT. Because LMWH was associated with a lower incidence of HIT compared with UFH (0.084% vs 0.51%, respectively), LMWH reduced the incremental cost of VTE prophylaxis by $13.88 per patient compared with UFH.
THE EXCLAIM TRIAL: IS THERE A ROLE FOR EXTENDED PROPHYLAXIS?
Although the previously discussed studies have clearly demonstrated the benefit of in-hospital VTE prophylaxis for acutely ill medical patients, none has rigorously examined extended-duration out-of-hospital prophylaxis in these patients. This represents an important gap in the literature, since a substantial proportion of VTE develops in the outpatient setting within 3 months of a hospitalization, and most outpatient VTE episodes occur within 1 month of a preceding hospitalization.20
To begin to fill this gap, the Extended Clinical Prophylaxis in Acutely Ill Medical Patients (EXCLAIM) trial was conducted to compare extended-duration LMWH prophylaxis with a standard LMWH prophylaxis regimen in acutely ill medical patients using a prospective, multicenter, randomized, double-blind, placebo-controlled design.21
Patients and study design
Patients were eligible for enrollment if they were aged 40 years or older and had recent immobilization (≤ 3 days), a predefined acute medical illness, and either level 1 mobility (total bed rest or sedentary state) or level 2 mobility (level 1 with bathroom privileges). The predefined acute medical illnesses consisted of New York Heart Association class III/IV heart failure, acute respiratory insufficiency, or other acute medical conditions, including post-acute ischemic stroke, acute infection without septic shock, and active cancer.
All patients received open-label enoxaparin 40 mg subcutaneously once daily for 10 ± 4 days, after which they were randomized to either enoxaparin 40 mg subcutaneously once daily or placebo for an additional 28 ± 4 days.
The primary efficacy end point was the incidence of VTE events, defined as asymptomatic DVT documented by mandatory ultrasonography at the end of the double-blind treatment period (28 ± 4 days) or as symptomatic DVT, symptomatic PE, or fatal PE at any time during the double-blind period. Symptomatic DVT was confirmed by objective tests; PE was confirmed by ventilation-perfusion scan, computed tomography, angiography, or autopsy.
Secondary efficacy end points were mortality at the end of the double-blind period, at 3 months, and at 6 months, as well as the incidence of VTE at 3 months.
The primary safety outcome measure was the incidence of major hemorrhage during the double-blind period; secondary safety measures were rates of major and minor hemorrhage, minor hemorrhage, HIT, and serious adverse events.
Population amended at planned interim analysis
After approximately half of the patients were enrolled, a planned and blinded interim analysis for futility concluded that the study was unlikely to show a statistically significant advantage of enoxaparin over placebo. The trial’s steering committee followed the suggestion of its data safety monitoring board to redefine the inclusion criteria to refocus enrollment on patients with a high risk of VTE. A blinded analysis was performed to identify this subgroup.
The resulting amended inclusion criteria were the same as above except that level 2 mobility had to be accompanied by at least one of three additional high-risk criteria: (1) age greater than 75 years, (2) history of prior VTE, or (3) diagnosis of cancer.
The trial’s main exclusion criteria were evidence of active bleeding, a contraindication to anticoagulation, receipt of prophylactic LMWH or UFH more than 72 hours prior to enrollment, treatment with an oral anticoagulant within 72 hours of enrollment, major surgery within the prior 3 months, cerebral stroke with bleeding, and persistent renal failure (creatinine clearance < 30 mL/min).


Results
The amended study population included 5,105 patients, 5,049 of whom received open-label enoxaparin. Of this group, 2,013 were randomized to active extended prophylaxis with enoxparain and 2,027 to placebo. Baseline characteristics, including level of mobility, were similar between the two groups.
The efficacy of extended prophylaxis with enoxaparin was enduring, as the cumulative incidence of VTE events at day 90 was significantly lower in enoxaparin recipients than in placebo recipients (3.0% vs 5.2%; relative reduction of 42%; P = .0115).
There was no difference in all-cause mortality at 6 months between the enoxaparin and placebo groups (10.1% vs 8.9%, respectively; P = .179).
Safety. Major hemorrhage was significantly more frequent in the enoxaparin arm, occurring in 0.60% of enoxaparin recipients compared with 0.15% of placebo recipients (P = .019). Minor bleeding was also more common with enoxaparin (5.20% vs 3.70%; P = .024).
Conclusions
The EXCLAIM trial found that an extended-duration (38-day) enoxaparin regimen significantly reduced the overall incidence of VTE relative to a 10-day enoxaparin regimen in acutely ill medical patients with reduced mobility. At the same time, the extended regimen was associated with a significant increase in the rate of major bleeding, although the incidence of major bleeding was low. The investigators concluded that the net clinical effect of extended-duration prophylaxis with enoxaparin is favorable, as only 46 patients would need to be treated to prevent one VTE event, whereas 224 patients would need to be treated to result in one major bleeding event.21
For this reason, it is reasonable to consider extended prophylaxis for hospitalized medical patients after identifying these patients’ risk factors. In keeping with the trial’s amended inclusion criteria, patients older than age 75 and those with cancer or prior VTE should receive special consideration for extended prophylaxis.
RECOMMENDED APPROACH TO VTE PREVENTION IN HOSPITALIZED MEDICAL PATIENTS
- All hospitalized medical patients should be screened at the time of admission, and patients at risk for VTE should receive prophylaxis.
- All patients with reduced mobility and one or more other risk factors for VTE are candidates for prophylaxis.
- Patients should be reassessed daily for the development of VTE risk factors during their hospitalization if risk factors are absent on admission.
- If screening or reassessment reveals any VTE risk factors, pharmacologic prophylaxis is the mainstay of therapy. If exclusion criteria for pharmacologic prophylaxis are present, mechanical prophylaxis with graduated compression stockings and intermittent compression devices should be used. For very high-risk medical patients without a contraindication to anticoagulants, combination prophylaxis with both an anticoagulant and mechanical devices is preferred.
- In this patient population, LMWH agents are preferred as pharmacologic prophylaxis over UFH and over fondaparinux (which is not currently approved by the US Food and Drug Administration for this population).
- If UFH is to be used in this patient population, 5,000 U three times daily is the preferred dosage.
- Extended pharmacologic prophylaxis should be considered in patients older than age 75 and in patients with a cancer diagnosis or a prior VTE episode.
DISCUSSION: ADDITIONAL PERSPECTIVES FROM THE AUTHORS
Dr. Jaffer: Dr. Spyropoulos, are there any guidelines, other than those from the ACCP, that speak to VTE prophylaxis in hospitalized medical patients? If so, what are their take-home messages and how do they differ from the ACCP guidelines?
Dr. Spyropoulos: I was part of the group that developed the International Consensus Statement (ICS) published in International Angiology in 2006,11 which is more recent than the latest ACCP guidelines, which were published in 2004. The ICS drew on much of the same data that the ACCP did, but we did an updated review of clinical trials.
For VTE prophylaxis in hospitalized medical patients, the ICS recommendations are more specific with regard to the type, dose, and dosing frequency of anticoagulant agents. First, they specify doses for both LMWH agents in this patient setting: 40 mg once daily for enoxaparin, and 5,000 IU once daily for dalteparin.
The ICS document also states that if UFH is the choice for prophylaxis, a regimen of 5,000 U three times daily should be considered. In the past year alone, two analyses suggest that three-times-daily dosing of UFH in medical patients provides superior efficacy to twice-daily dosing, although perhaps at the expense of more bleeding episodes.12,16 It is important to remember that no large placebo-controlled trial supports the efficacy of a UFH regimen of 5,000 U twice daily in this population.
Finally, the ICS document states that fondaparinux 2.5 mg once daily is a viable option for prophylaxis in medical patients, based on the ARTEMIS trial,3 even though this represents an off-label use.
Dr. Jaffer: Real-world use of VTE prophylaxis is far from optimal, especially in medical patients, and this is partly a result of system-of-care issues. I’d like to conclude by asking each of my colleagues to offer your perspectives on how your own institutions have improved their systems of care to promote better use of VTE prophylaxis and what lessons might be shared with others. Dr. McKean, you work at Brigham and Women’s Hospital, which recently reported impressive results with an electronic alert system designed to increase clinicians’ consideration of VTE risk assessment and use of prophylaxis.24 Please tell us about that study and the alert system.
Dr. McKean: Despite many educational initiatives at Brigham and Women’s Hospital, there were still some patients at high risk for VTE who were not receiving appropriate prophylaxis. What Dr. Samuel Goldhaber and his colleagues wanted to determine was whether changing the system of care could result in a reduced incidence of VTE.24 They devised a computer software program linked to the patient database that used eight common risk factors to determine each hospitalized patient’s risk profile for VTE. Each risk factor was weighted according to a point scale, with major risk factors (cancer, prior VTE, or hypercoagulability) assigned 3 points, the intermediate risk factor of surgery assigned 2 points, and minor risk factors (advanced age, obesity, immobility, or use of hormone replacement therapy or oral contraceptives) assigned 1 point. For patients with a total risk score of 4 or greater, the computer screen generates a color-coded VTE risk alert that requires the physician to acknowledge the alert and choose one of three options: order prophylaxis as appropriate, review a 60-page document on the computer to learn more about prophylaxis, or do nothing.
The study found that hospitalized patients who were randomized to treatment under the computer alert system were significantly more likely to receive VTE prophylaxis and significantly less likely to develop VTE than were patients randomized to a control group. The alert system reduced the risk of DVT or PE at 90 days by 41% in patients considered to be at high risk. It was particularly interesting that the incidence of VTE was lower in the intervention group even when physicians chose not to use prophylaxis, which suggests that simply having this alert system in place improved outcomes, perhaps by raising awareness of the risk of VTE.24
Additional studies are needed to better understand physicians’ behavior and determine why they seem to have a disproportionate fear of the risk of bleeding relative to the risk of clotting, including fatal PE, because that is really the heart of the matter. When patients are not given prophylaxis, often it is because of fear of bleeding. It is not clear, however, why some of these patients did not receive mechanical devices as an alternative form of prophylaxis, but this seems to be the case worldwide, as shown recently by the multinational ENDORSE study.22 Meanwhile, as we await studies to better understand physician perceptions and behaviors regarding prophylaxis, we need to work hard to change the system of care.
Dr. Deitelzweig: Over the past couple of years, the Ochsner Clinic has grown from a one-hospital teaching organization to a seven-hospital system with a mix of closed and open medical staff. The challenge is how to take a process that worked well in the one center, where appropriate prophylaxis was used about 90% of the time, and transfer it to the other centers in the larger system. We have endorsed several types of performance tools, such as the change-acceleration processes used by General Electric. The aim is to share a vision of heightening awareness. To do that, we have worked to mobilize the key stakeholders, at least half of them, to build algorithms that they all will endorse. It is easier said than done, however, and we have found it essential to involve both physicians and non-physician colleagues from pharmacy and nursing who have political and organizational clout.
Dr. Brotman: At Johns Hopkins, I took a bit more draconian approach to this issue because I thought that hospitalists often knew that they should be using VTE prophylaxis but sometimes weren’t, and I am not convinced that clinicians always look at prompts. So we came up with a system that incorporates both billing and documentation simultaneously. We put a hard stop on users’ documentation so that they could not sign off on a note or bill for their care until they checked off the kind of VTE prophylaxis they were using. Since hospitalists ultimately care about billing for their work, this system has at least ensured that everybody has considered and documented VTE prophylaxis on a daily basis. There are other hard stops that can be implemented in computer order-entry systems as well, and we are considering ways to roll them out on a broader scale.
However, all of these systems can have problems because patient situations change from day to day. For instance, VTE prophylaxis is not necessarily indicated in a 38-year-old ambulatory patient who comes in with a sickle cell crisis, but you will need to reconsider if the patient ends up in acute chest syndrome in the intensive care unit. I do not yet have a good way to ensure that this is being done on a daily basis with all patients.
Dr. Amin: At the University of California, Irvine, we implemented an electronic alert system, but we locked users in so that they could not complete their admission orders until they answered questions about VTE prevention. This practice increased our VTE prophylaxis rates tremendously. Because we are a level I trauma center, we allow users to bypass the screens one time, but the next time they log in, even to get a simple lab result, they have to answer the questions about VTE prevention.
With any system you develop, you also have to continue with the education process, because clinicians sometimes get into bad habits or simply forget things.
Dr. Spyropolous: At Lovelace Medical Center, we didn’t have the sophistication of an electronic order-entry system, but we had an experienced clinical pharmacist (the director of inpatient pharmacy) who helped to develop and champion VTE prevention guidelines that have then been used throughout the system in close conjunction with our hospitalists’ rounds. This system has been used successfully for the past 7 years.
- Samama MM, Cohen AT, Darmon JY, et al. A comparison of enoxaparin with placebo for the prevention of venous thrombo-embolism in acutely ill medical patients. Prophylaxis in Medical Patients with Enoxaparin Study Group. N Engl J Med 1999; 341:793–800.
- Leizorovicz A, Cohen AT, Turpie AG, et al. Randomized, placebo-controlled trial of dalteparin for the prevention of venous thromboembolism in acutely ill medical patients. Circulation 2004; 110:874–879.
- Cohen AT, Davidson BL, Gallus AS, et al. Efficacy and safety of fondaparinux for the prevention of venous thromboembolism in older acute medical patients: randomised placebo controlled trial. BMJ 2006; 332:325–329.
- Baglin TP, White K, Charles A. Fatal pulmonary embolism in hos-pitalised medical patients. J Clin Pathol 1997; 50:609–610.
- Piazza G, Seddighzadeh A, Goldhaber SZ. Double trouble for 2,609 hospitalized medical patients who developed deep vein thrombosis: prophylaxis omitted more often and pulmonary embolism more frequent. Chest 2007; 132:554–561.
- Amin A, Stemkowski S, Lin J, Yang G. Thromboprophylaxis rates in US medical centers: success or failure? J Thromb Haemost 2007; 5:1610–1616.
- Geerts WH, Heit JA, Clagett GP, et al. Prevention of venous thromboembolism. Chest 2001; 119(1 Suppl):132S–175S.
- Tapson VF, Decousus H, Pini M, et al. Venous thromboembolism prophylaxis in acutely ill hospitalized medical patients: findings from the International Medical Prevention Registry on Venous Thromboembolism. Chest 2007; 132:936–945.
- Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thrombo-embolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 2004; 126(3 Suppl):338S–400S.
- Dentali F, Douketis JD, Gianni M, Lim W, Crowther MA. Meta-analysis: anticoagulant prophylaxis to prevent symptomatic venous thromboembolism in hospitalized medical patients. Ann Intern Med 2007; 146:278–288.
- Cardiovascular Disease Educational and Research Trust; Cyprus Cardiovascular Disease Educational and Research Trust; European Venous Forum; International Surgical Thrombosis Forum; International Union of Angiology; Union Internationale de Phlébologie. Prevention and treatment of venous thromboembolism. International Consensus Statement (guidelines according to scientific evidence). Int Angiol 2006; 25:101–161.
- Wein L, Wein S, Haas SJ, Shaw J, Krum H. Pharmacological venous thromboembolism prophylaxis in hospitalized medical patients: a meta-analysis of randomized controlled trials. Arch Intern Med 2007; 167:1476–1486.
- Mismetti P, Laporte-Simitsidis S, Tardy B, et al. Prevention of venous thromboembolism in internal medicine with unfractionated or low-molecular-weight heparins: a meta-analysis of randomised clinical trials. Thromb Haemost 2000; 83:14–19.
- Lechler E, Schramm W, Flosbach CW. The venous thrombotic risk in non-surgical patients: epidemiological data and efficacy/safety profile of a low-molecular-weight heparin (enoxaparin). The Prime Study Group. Haemostasis 1996; 26(Suppl 2):49–56.
- Kleber FX, Witt C, Vogel G, et al; the PRINCE Study Group. Randomized comparison of enoxaparin with unfractionated heparin for the prevention of venous thromboembolism in medical patients with heart failure or severe respiratory disease. Am Heart J 2003; 145:614–621.
- King CS, Holley AB, Jackson JL, Shorr AF, Moores LK. Twice vs three times daily heparin dosing for thromboembolism prophylaxis in the general medical population: a meta-analysis. Chest 2007; 131:507–516.
- Burleigh E, Wang C, Foster D, et al. Thromboprophylaxis in medically ill patients at risk for venous thromboembolism. Am J Health Syst Pharm 2006; 63(20 Suppl 6):S23–S29.
- Leykum L, Pugh J, Diuguid D, Papadopoulos K. Cost utility of substituting enoxaparin for unfractionated heparin for prophylaxis of venous thrombosis in the hospitalized medical patient. J Hosp Med 2006; 1:168–176.
- Creekmore FM, Oderda GM, Pendleton RC, Brixner DI. Incidence and economic implications of heparin-induced thrombocytopenia in medical patients receiving prophylaxis for venous thromboembolism. Pharmacotherapy 2006; 26:1438–1445.
- Spencer FA, Lessard D, Emery C, Reed G, Goldberg RJ. Venous thromboembolism in the outpatient setting. Arch Intern Med 2007; 167:1471–1475.
- Hull RD, Schellong SM Tapson VF, et al. Extended-duration venous thromboembolism (VTE) prophylaxis in acutely ill medical patients with recent reduced mobility: the EXCLAIM study. Presentation at: International Society on Thrombosis and Haemo-stasis XXIst Congress; July 6–12, 2007; Geneva, Switzerland.
- Cohen AT, Tapson VF, Bergmann JF, et al. Venous thromboembolism risk and prophylaxis in the acute hospital care setting (ENDORSE study): a multinational cross-sectional study. Lancet 2008; 371:387–394.
- Kahn SR, Panju A, Geerts W, et al; CURVE study investigators. Multicenter evaluation of the use of venous thromboembolism prophylaxis in acutely ill medical patients in Canada. Thromb Res 2007; 119:145–155.
- Kucher N, Koo S, Quiroz R, et al. Electronic alerts to prevent thromboembolism among hospitalized patients. N Engl J Med 2005; 352:969–977.
- Anderson FA Jr, Wheeler HB, Goldberg RJ, et al. A population-based perspective of the hospital incidence and case-fatality rates of deep vein thrombosis and pulmonary embolism. The Worcester DVT Study. Arch Intern Med 1991; 151:933–938.
- Howell MD, Geraci JM, Knowlton AA. Congestive heart failure and outpatient risk of venous thromboembolism: a retrospective, case-control study. J Clin Epidemiol 2001; 54:810–816.
- Smeeth L, Cook C, Thomas S, Hall AJ, Hubbard R, Vallance P. Risk of deep vein thrombosis and pulmonary embolism after acute infection in a community setting. Lancet 2006; 367:1075–1079.
- Alikhan R, Cohen AT, Combe S, et al. Prevention of venous thromboembolism in medical patients with enoxaparin: a subgroup analysis of the MEDENOX study. Blood Coagul Firbinolysis 2003; 14:341–346.
- Anderson FA Jr, Spencer FA. Risk factors for venous thromboembolism. Circulation 2003; 107(23 Suppl 1):I9–I16.
- Greinacher A, Warkentin TE. Recognition, treatment, and prevention of heparin-induced thrombocytopenia: review and update. Thromb Res 2006; 118:165–176.
- Martel N, Lee J, Wells PS. Risk for heparin-induced thrombocytopenia with unfractionated and low-molecular-weight heparin thromboprophylaxis: a meta-analysis. Blood 2005; 106:2710–2715.
- Sanderink GJ, Guimart CG, Ozoux ML, Jariwala NU, Shukla UA, Boutouyrie BX. Pharmacokinetics and pharmacodynamics of the prophylactic dose of enoxaparin once daily over 4 days in patients with renal impairment. Thromb Res 2002; 105:225–231.
- Douketis J, Cook D, Zytaruk N, et al. Dalteparin thromboprophylaxis in critically ill patients with severe renal insufficiency: the DIRECT study [abstract]. J Thromb Haemost 2007; 5(Suppl 2): P-S-680.
- Scholten DJ, Hoedema RM, Scholten SE. A comparison of two different prophylactic dose regimens of low molecular weight heparin in bariatric surgery. Obes Surg 2002; 12:19–24.
The need for prophylaxis of venous thromboembolism (VTE) in hospitalized acutely ill medical patients is well established. Without prophylaxis, hospitalized medical patients develop VTE at a rate of 5% to 15%.1–3 Moreover, pulmonary embolism (PE) occurs more frequently in hospitalized medical patients than in nonmedical patients, and is a leading cause of sudden death in hospitalized medical patients.4,5 Without appropriate prophylaxis, 1 in 20 hospitalized medical patients may suffer a fatal PE.4
PROPHYLAXIS IN MEDICAL PATIENTS: UNDERUSED AND OFTEN INAPPROPRIATE
Despite these risks and the clear indications for VTE prophylaxis in hospitalized medical patients, prophylaxis of VTE is omitted more often in these patients than in hospitalized surgical patients.5 Even when prophylaxis is given, it is often used inappropriately in the medical population. So concludes a recent analysis of data from 196,104 patients with acute medical conditions who were discharged from 227 US hospitals from January 2002 to September 2005.6 Criteria for inclusion in the analysis were patient age of 40 years or older, a hospital stay of 6 days or greater, and an absence of contraindications to anticoagulation. Appropriate prophylaxis was defined in accordance with the Sixth American College of Chest Physicians (ACCP) Consensus Conference on Antithrombotic Therapy.7
The analysis revealed an overall VTE prophylaxis rate of 61.8%, but the rate of appropriate prophylaxis was only 33.9%, meaning that two-thirds of discharged patients did not receive prophylaxis in accordance with ACCP guidelines. When temporal trends were analyzed according to groups based on patients’ diagnosis at admission (acute myocardial infarction, severe lung disease, ischemic stroke, cancer, heart failure, or trauma), the rate of appropriate prophylaxis remained essentially flat from the beginning to the end of the study period for virtually all diagnosis groups.6
Similar findings have emerged from the International Medical Prevention Registry on Venous Thromboembolism (IMPROVE), an ongoing international registry of acutely ill medical patients.8 Data from the first 15,156 patients, enrolled from July 2002 through September 2006, reveal that 50% of patients received pharmacologic and/or mechanical VTE prophylaxis in the hospital, and only 60% of patients who met established criteria for VTE prophylaxis actually received it.
Analysis of the US portion of the IMPROVE data shows that 54% of the US patient sample received some form of VTE prophylaxis; 22% of US patients received intermittent pneumatic compression, 21% received unfractionated heparin (UFH), 14% received low-molecular-weight heparin (LMWH), and 3% wore compression stockings.8 Thus, despite a paucity of data supporting a benefit of intermittent pneumatic compression in this population,9 it was the most frequently used form of prophylaxis in US patients.
CLINICAL TRIALS OF PHARMACOLOGIC PROPHYLAXIS IN MEDICAL PATIENTS
The Prophylaxis in Medical Patients with Enoxaparin (MEDENOX) trial1 randomized 1,102 hospitalized patients to one of two doses of the LMWH enoxaparin (20 mg or 40 mg once daily subcutaneously) or placebo for 6 to 14 days. Compared with placebo, the 40-mg dose of enoxaparin was associated with a 63% reduction in risk of VTE over 3 months of follow-up (P < .001) (Figure 1).
The Prospective Evaluation of Dalteparin Efficacy for Prevention of VTE in Immobilized Patients Trial (PREVENT)2 was a multicenter, randomized, double-blind study comparing the LMWH dalteparin (5,000 IU daily given subcutaneously for 14 days) with placebo in 3,706 acutely ill medical patients. Over 90 days of follow-up, the risk of VTE was reduced by 44% in patients assigned to dalteparin compared with those assigned to placebo (P = .0015) (Figure 1).
The Arixtra for Thromboembolism Prevention in a Medical Indications Study (ARTEMIS)3 randomized 849 medical patients 60 years or older to 6 to 14 days of therapy with the selective factor Xa inhibitor fondaparinux (2.5 mg once daily subcutaneously) or placebo. Compared with the placebo group, fondaparinux recipients had a 47% lower risk of developing VTE by day 15 (P = .029) (Figure 1).
Fewer events and fatal PEs, but no effect on all-cause mortality
A recent meta-analysis by Dentali et al10 further demonstrates the efficacy of anticoagulant therapy for preventing symptomatic VTE in hospitalized medical patients. This analysis included several other trials in addition to the three reviewed above,1–3 for a total of nine randomized studies (seven of which were dou-ble-blind) comprising 19,958 patients. Across the nine studies, anticoagulant prophylaxis was clearly superior to placebo in preventing fatal PE (relative risk, 0.38 [95% CI, 0.21 to 0.69]). There was a strong trend toward a reduction in symptomatic deep vein thrombosis (DVT) with prophylaxis but no effect on all-cause mortality. The meta-analysis also provided reassurance that prophylaxis does not increase the rate of major bleeding.
HOW DO THE PROPHYLAXIS OPTIONS STACK UP?
What the ACCP recommends
Current ACCP guidelines recommend the use of either LMWH or low-dose UFH (5,000 U subcutaneously two or three times daily) as a Grade 1A recommendation for VTE prophylaxis in patients with medical conditions and risk factors for VTE.9 This represents the guidelines’ highest level of recommendation, ie, one that is based on randomized controlled trials (RCTs) without important limitations. In contrast, the 2006 International Consensus Statement, developed as a collaborative effort among expert bodies on VTE, specified a more narrow dosing recommendation for UFH in this patient population (5,000 U three times daily, not twice daily) as well as specifying 40 mg once daily as the recommended dose of enoxaparin and 5,000 IU once daily as the recommended dose of dalteparin.11
For medical patients with risk factors for VTE who have a contraindication to anticoagulant prophylaxis, the ACCP guidelines recommend the use of graduated compression stockings or intermittent pneumatic compression devices as a Grade 1C+ recommendation (“no RCTs but strong RCT results can be unequivocally extrapolated, or overwhelming evidence from observational studies”9).
Current ACCP guidelines do not address the use of fondaparinux in their recommendations for VTE prophylaxis in medical patients.
Getting a handle on bleeding risk
Patient characteristics that exclude pharmacologic thromboprophylaxis due to bleeding risk are generally limited to active bleeding or coagulopathy, as demonstrated by a platelet count less than 50,000 cells/µL or an international normalized ratio greater than 1.5. Additionally, bleeding risk should be carefully assessed if an invasive procedure is planned during a patient’s hospital stay.
It is worth noting that the anticoagulant doses used for VTE prophylaxis are a fraction of those used for treatment of VTE. Thus, if a patient would be treated with full-dose anticoagulation if VTE developed, then that patient should be eligible for VTE prophylaxis.
Because the use of mechanical forms of prophylaxis in medical patients is not truly evidence-based, mechanical prophylaxis should be reserved for medical patients who have a contraindication to anticoagulants, or for use in combination with anticoagulants in patients at very high risk of VTE.
UFH vs LMWH
Two meta-analyses have compared UFH with LMWH for VTE prevention in medical patients.12,13 In a recent analysis that included 10 trials directly comparing the two therapies, 14 trials comparing UFH with control, and 11 trials comparing LMWH with control, Wein et al found a lower risk of DVT with LMWH than with UFH (relative risk, 0.68 [95% CI, 0.52 to 0.88]) but no difference between the therapies in mortality or bleeding risk.12 In an earlier and smaller analysis, Mismetti et al found no significant differences between UFH and LMWH in preventing DVT or death but did find a significant reduction in major bleeding episodes with LMWH versus three-times-daily UFH (52% relative reduction; P = .049).13
Randomized trials also reveal that enoxaparin 40 mg once daily is as efficacious as UFH 5,000 U three times daily for VTE prevention in medical patients.14,15 The above analysis by Wein et al12 and an additional meta-analysis by King and colleagues16 found that three-times-daily dosing of UFH is more efficacious than twice-daily dosing of UFH, but at the expense of more bleeding, including major bleeding.
Economic considerations
Because of differences in drug acquisition costs between UFH and the LMWH agents, several economic evaluations have compared the use of these therapies for prophylaxis in medical patients at risk of VTE.
In an analysis of hospital costs for medical patients receiving VTE prophylaxis from more than 330 US hospitals for the period 2001–2004, Burleigh et al found that mean total hospital costs were higher for patients who received UFH than for those who received LMWH ($7,615 vs $6,866) even though mean drug costs were higher for LMWH ($791 vs $569 for UFH).17 A reduction in hospital length of stay appeared to contribute to the overall savings with LMWH; other contributors may have included costs associated with heparin-induced thrombocytopenia (HIT) in UFH recipients or the extra nursing time required for administering UFH in two or three daily doses.
Leykum et al used a decision analysis model to estimate the economic effect of substituting enoxaparin for UFH in hospitalized medical patients for whom VTE prophylaxis is indicated.18 Cost data were based on Medicare reimbursement rates as well as drug and laboratory costs for a multi-institutional health system. The model assumed HIT incidence rates of 2.7% with UFH and 0.3% with enoxaparin. It also assumed the cost of a daily dose to be $4 for UFH versus $84 for enoxaparin. From the payer perspective, the model showed that substituting enoxaparin for UFH would reduce the overall cost of care by $28.61 per day on a per-patient basis, despite enoxaparin’s higher acquisition cost, and would save $4,550 per quality-adjusted life-year by reducing the incidence of HIT.
Another cost analysis confirms the association between HIT and increased hospital costs. Creekmore et al retrospectively analyzed data from 10,121 adult medical patients who received VTE prophylaxis at the University of Utah Hospital in Salt Lake City from August 2000 to November 2004.19 They found that an admission during which HIT developed incurred a mean cost of $56,364, compared with $15,231 for an admission without HIT. Because LMWH was associated with a lower incidence of HIT compared with UFH (0.084% vs 0.51%, respectively), LMWH reduced the incremental cost of VTE prophylaxis by $13.88 per patient compared with UFH.
THE EXCLAIM TRIAL: IS THERE A ROLE FOR EXTENDED PROPHYLAXIS?
Although the previously discussed studies have clearly demonstrated the benefit of in-hospital VTE prophylaxis for acutely ill medical patients, none has rigorously examined extended-duration out-of-hospital prophylaxis in these patients. This represents an important gap in the literature, since a substantial proportion of VTE develops in the outpatient setting within 3 months of a hospitalization, and most outpatient VTE episodes occur within 1 month of a preceding hospitalization.20
To begin to fill this gap, the Extended Clinical Prophylaxis in Acutely Ill Medical Patients (EXCLAIM) trial was conducted to compare extended-duration LMWH prophylaxis with a standard LMWH prophylaxis regimen in acutely ill medical patients using a prospective, multicenter, randomized, double-blind, placebo-controlled design.21
Patients and study design
Patients were eligible for enrollment if they were aged 40 years or older and had recent immobilization (≤ 3 days), a predefined acute medical illness, and either level 1 mobility (total bed rest or sedentary state) or level 2 mobility (level 1 with bathroom privileges). The predefined acute medical illnesses consisted of New York Heart Association class III/IV heart failure, acute respiratory insufficiency, or other acute medical conditions, including post-acute ischemic stroke, acute infection without septic shock, and active cancer.
All patients received open-label enoxaparin 40 mg subcutaneously once daily for 10 ± 4 days, after which they were randomized to either enoxaparin 40 mg subcutaneously once daily or placebo for an additional 28 ± 4 days.
The primary efficacy end point was the incidence of VTE events, defined as asymptomatic DVT documented by mandatory ultrasonography at the end of the double-blind treatment period (28 ± 4 days) or as symptomatic DVT, symptomatic PE, or fatal PE at any time during the double-blind period. Symptomatic DVT was confirmed by objective tests; PE was confirmed by ventilation-perfusion scan, computed tomography, angiography, or autopsy.
Secondary efficacy end points were mortality at the end of the double-blind period, at 3 months, and at 6 months, as well as the incidence of VTE at 3 months.
The primary safety outcome measure was the incidence of major hemorrhage during the double-blind period; secondary safety measures were rates of major and minor hemorrhage, minor hemorrhage, HIT, and serious adverse events.
Population amended at planned interim analysis
After approximately half of the patients were enrolled, a planned and blinded interim analysis for futility concluded that the study was unlikely to show a statistically significant advantage of enoxaparin over placebo. The trial’s steering committee followed the suggestion of its data safety monitoring board to redefine the inclusion criteria to refocus enrollment on patients with a high risk of VTE. A blinded analysis was performed to identify this subgroup.
The resulting amended inclusion criteria were the same as above except that level 2 mobility had to be accompanied by at least one of three additional high-risk criteria: (1) age greater than 75 years, (2) history of prior VTE, or (3) diagnosis of cancer.
The trial’s main exclusion criteria were evidence of active bleeding, a contraindication to anticoagulation, receipt of prophylactic LMWH or UFH more than 72 hours prior to enrollment, treatment with an oral anticoagulant within 72 hours of enrollment, major surgery within the prior 3 months, cerebral stroke with bleeding, and persistent renal failure (creatinine clearance < 30 mL/min).


Results
The amended study population included 5,105 patients, 5,049 of whom received open-label enoxaparin. Of this group, 2,013 were randomized to active extended prophylaxis with enoxparain and 2,027 to placebo. Baseline characteristics, including level of mobility, were similar between the two groups.
The efficacy of extended prophylaxis with enoxaparin was enduring, as the cumulative incidence of VTE events at day 90 was significantly lower in enoxaparin recipients than in placebo recipients (3.0% vs 5.2%; relative reduction of 42%; P = .0115).
There was no difference in all-cause mortality at 6 months between the enoxaparin and placebo groups (10.1% vs 8.9%, respectively; P = .179).
Safety. Major hemorrhage was significantly more frequent in the enoxaparin arm, occurring in 0.60% of enoxaparin recipients compared with 0.15% of placebo recipients (P = .019). Minor bleeding was also more common with enoxaparin (5.20% vs 3.70%; P = .024).
Conclusions
The EXCLAIM trial found that an extended-duration (38-day) enoxaparin regimen significantly reduced the overall incidence of VTE relative to a 10-day enoxaparin regimen in acutely ill medical patients with reduced mobility. At the same time, the extended regimen was associated with a significant increase in the rate of major bleeding, although the incidence of major bleeding was low. The investigators concluded that the net clinical effect of extended-duration prophylaxis with enoxaparin is favorable, as only 46 patients would need to be treated to prevent one VTE event, whereas 224 patients would need to be treated to result in one major bleeding event.21
For this reason, it is reasonable to consider extended prophylaxis for hospitalized medical patients after identifying these patients’ risk factors. In keeping with the trial’s amended inclusion criteria, patients older than age 75 and those with cancer or prior VTE should receive special consideration for extended prophylaxis.
RECOMMENDED APPROACH TO VTE PREVENTION IN HOSPITALIZED MEDICAL PATIENTS
- All hospitalized medical patients should be screened at the time of admission, and patients at risk for VTE should receive prophylaxis.
- All patients with reduced mobility and one or more other risk factors for VTE are candidates for prophylaxis.
- Patients should be reassessed daily for the development of VTE risk factors during their hospitalization if risk factors are absent on admission.
- If screening or reassessment reveals any VTE risk factors, pharmacologic prophylaxis is the mainstay of therapy. If exclusion criteria for pharmacologic prophylaxis are present, mechanical prophylaxis with graduated compression stockings and intermittent compression devices should be used. For very high-risk medical patients without a contraindication to anticoagulants, combination prophylaxis with both an anticoagulant and mechanical devices is preferred.
- In this patient population, LMWH agents are preferred as pharmacologic prophylaxis over UFH and over fondaparinux (which is not currently approved by the US Food and Drug Administration for this population).
- If UFH is to be used in this patient population, 5,000 U three times daily is the preferred dosage.
- Extended pharmacologic prophylaxis should be considered in patients older than age 75 and in patients with a cancer diagnosis or a prior VTE episode.
DISCUSSION: ADDITIONAL PERSPECTIVES FROM THE AUTHORS
Dr. Jaffer: Dr. Spyropoulos, are there any guidelines, other than those from the ACCP, that speak to VTE prophylaxis in hospitalized medical patients? If so, what are their take-home messages and how do they differ from the ACCP guidelines?
Dr. Spyropoulos: I was part of the group that developed the International Consensus Statement (ICS) published in International Angiology in 2006,11 which is more recent than the latest ACCP guidelines, which were published in 2004. The ICS drew on much of the same data that the ACCP did, but we did an updated review of clinical trials.
For VTE prophylaxis in hospitalized medical patients, the ICS recommendations are more specific with regard to the type, dose, and dosing frequency of anticoagulant agents. First, they specify doses for both LMWH agents in this patient setting: 40 mg once daily for enoxaparin, and 5,000 IU once daily for dalteparin.
The ICS document also states that if UFH is the choice for prophylaxis, a regimen of 5,000 U three times daily should be considered. In the past year alone, two analyses suggest that three-times-daily dosing of UFH in medical patients provides superior efficacy to twice-daily dosing, although perhaps at the expense of more bleeding episodes.12,16 It is important to remember that no large placebo-controlled trial supports the efficacy of a UFH regimen of 5,000 U twice daily in this population.
Finally, the ICS document states that fondaparinux 2.5 mg once daily is a viable option for prophylaxis in medical patients, based on the ARTEMIS trial,3 even though this represents an off-label use.
Dr. Jaffer: Real-world use of VTE prophylaxis is far from optimal, especially in medical patients, and this is partly a result of system-of-care issues. I’d like to conclude by asking each of my colleagues to offer your perspectives on how your own institutions have improved their systems of care to promote better use of VTE prophylaxis and what lessons might be shared with others. Dr. McKean, you work at Brigham and Women’s Hospital, which recently reported impressive results with an electronic alert system designed to increase clinicians’ consideration of VTE risk assessment and use of prophylaxis.24 Please tell us about that study and the alert system.
Dr. McKean: Despite many educational initiatives at Brigham and Women’s Hospital, there were still some patients at high risk for VTE who were not receiving appropriate prophylaxis. What Dr. Samuel Goldhaber and his colleagues wanted to determine was whether changing the system of care could result in a reduced incidence of VTE.24 They devised a computer software program linked to the patient database that used eight common risk factors to determine each hospitalized patient’s risk profile for VTE. Each risk factor was weighted according to a point scale, with major risk factors (cancer, prior VTE, or hypercoagulability) assigned 3 points, the intermediate risk factor of surgery assigned 2 points, and minor risk factors (advanced age, obesity, immobility, or use of hormone replacement therapy or oral contraceptives) assigned 1 point. For patients with a total risk score of 4 or greater, the computer screen generates a color-coded VTE risk alert that requires the physician to acknowledge the alert and choose one of three options: order prophylaxis as appropriate, review a 60-page document on the computer to learn more about prophylaxis, or do nothing.
The study found that hospitalized patients who were randomized to treatment under the computer alert system were significantly more likely to receive VTE prophylaxis and significantly less likely to develop VTE than were patients randomized to a control group. The alert system reduced the risk of DVT or PE at 90 days by 41% in patients considered to be at high risk. It was particularly interesting that the incidence of VTE was lower in the intervention group even when physicians chose not to use prophylaxis, which suggests that simply having this alert system in place improved outcomes, perhaps by raising awareness of the risk of VTE.24
Additional studies are needed to better understand physicians’ behavior and determine why they seem to have a disproportionate fear of the risk of bleeding relative to the risk of clotting, including fatal PE, because that is really the heart of the matter. When patients are not given prophylaxis, often it is because of fear of bleeding. It is not clear, however, why some of these patients did not receive mechanical devices as an alternative form of prophylaxis, but this seems to be the case worldwide, as shown recently by the multinational ENDORSE study.22 Meanwhile, as we await studies to better understand physician perceptions and behaviors regarding prophylaxis, we need to work hard to change the system of care.
Dr. Deitelzweig: Over the past couple of years, the Ochsner Clinic has grown from a one-hospital teaching organization to a seven-hospital system with a mix of closed and open medical staff. The challenge is how to take a process that worked well in the one center, where appropriate prophylaxis was used about 90% of the time, and transfer it to the other centers in the larger system. We have endorsed several types of performance tools, such as the change-acceleration processes used by General Electric. The aim is to share a vision of heightening awareness. To do that, we have worked to mobilize the key stakeholders, at least half of them, to build algorithms that they all will endorse. It is easier said than done, however, and we have found it essential to involve both physicians and non-physician colleagues from pharmacy and nursing who have political and organizational clout.
Dr. Brotman: At Johns Hopkins, I took a bit more draconian approach to this issue because I thought that hospitalists often knew that they should be using VTE prophylaxis but sometimes weren’t, and I am not convinced that clinicians always look at prompts. So we came up with a system that incorporates both billing and documentation simultaneously. We put a hard stop on users’ documentation so that they could not sign off on a note or bill for their care until they checked off the kind of VTE prophylaxis they were using. Since hospitalists ultimately care about billing for their work, this system has at least ensured that everybody has considered and documented VTE prophylaxis on a daily basis. There are other hard stops that can be implemented in computer order-entry systems as well, and we are considering ways to roll them out on a broader scale.
However, all of these systems can have problems because patient situations change from day to day. For instance, VTE prophylaxis is not necessarily indicated in a 38-year-old ambulatory patient who comes in with a sickle cell crisis, but you will need to reconsider if the patient ends up in acute chest syndrome in the intensive care unit. I do not yet have a good way to ensure that this is being done on a daily basis with all patients.
Dr. Amin: At the University of California, Irvine, we implemented an electronic alert system, but we locked users in so that they could not complete their admission orders until they answered questions about VTE prevention. This practice increased our VTE prophylaxis rates tremendously. Because we are a level I trauma center, we allow users to bypass the screens one time, but the next time they log in, even to get a simple lab result, they have to answer the questions about VTE prevention.
With any system you develop, you also have to continue with the education process, because clinicians sometimes get into bad habits or simply forget things.
Dr. Spyropolous: At Lovelace Medical Center, we didn’t have the sophistication of an electronic order-entry system, but we had an experienced clinical pharmacist (the director of inpatient pharmacy) who helped to develop and champion VTE prevention guidelines that have then been used throughout the system in close conjunction with our hospitalists’ rounds. This system has been used successfully for the past 7 years.
The need for prophylaxis of venous thromboembolism (VTE) in hospitalized acutely ill medical patients is well established. Without prophylaxis, hospitalized medical patients develop VTE at a rate of 5% to 15%.1–3 Moreover, pulmonary embolism (PE) occurs more frequently in hospitalized medical patients than in nonmedical patients, and is a leading cause of sudden death in hospitalized medical patients.4,5 Without appropriate prophylaxis, 1 in 20 hospitalized medical patients may suffer a fatal PE.4
PROPHYLAXIS IN MEDICAL PATIENTS: UNDERUSED AND OFTEN INAPPROPRIATE
Despite these risks and the clear indications for VTE prophylaxis in hospitalized medical patients, prophylaxis of VTE is omitted more often in these patients than in hospitalized surgical patients.5 Even when prophylaxis is given, it is often used inappropriately in the medical population. So concludes a recent analysis of data from 196,104 patients with acute medical conditions who were discharged from 227 US hospitals from January 2002 to September 2005.6 Criteria for inclusion in the analysis were patient age of 40 years or older, a hospital stay of 6 days or greater, and an absence of contraindications to anticoagulation. Appropriate prophylaxis was defined in accordance with the Sixth American College of Chest Physicians (ACCP) Consensus Conference on Antithrombotic Therapy.7
The analysis revealed an overall VTE prophylaxis rate of 61.8%, but the rate of appropriate prophylaxis was only 33.9%, meaning that two-thirds of discharged patients did not receive prophylaxis in accordance with ACCP guidelines. When temporal trends were analyzed according to groups based on patients’ diagnosis at admission (acute myocardial infarction, severe lung disease, ischemic stroke, cancer, heart failure, or trauma), the rate of appropriate prophylaxis remained essentially flat from the beginning to the end of the study period for virtually all diagnosis groups.6
Similar findings have emerged from the International Medical Prevention Registry on Venous Thromboembolism (IMPROVE), an ongoing international registry of acutely ill medical patients.8 Data from the first 15,156 patients, enrolled from July 2002 through September 2006, reveal that 50% of patients received pharmacologic and/or mechanical VTE prophylaxis in the hospital, and only 60% of patients who met established criteria for VTE prophylaxis actually received it.
Analysis of the US portion of the IMPROVE data shows that 54% of the US patient sample received some form of VTE prophylaxis; 22% of US patients received intermittent pneumatic compression, 21% received unfractionated heparin (UFH), 14% received low-molecular-weight heparin (LMWH), and 3% wore compression stockings.8 Thus, despite a paucity of data supporting a benefit of intermittent pneumatic compression in this population,9 it was the most frequently used form of prophylaxis in US patients.
CLINICAL TRIALS OF PHARMACOLOGIC PROPHYLAXIS IN MEDICAL PATIENTS
The Prophylaxis in Medical Patients with Enoxaparin (MEDENOX) trial1 randomized 1,102 hospitalized patients to one of two doses of the LMWH enoxaparin (20 mg or 40 mg once daily subcutaneously) or placebo for 6 to 14 days. Compared with placebo, the 40-mg dose of enoxaparin was associated with a 63% reduction in risk of VTE over 3 months of follow-up (P < .001) (Figure 1).
The Prospective Evaluation of Dalteparin Efficacy for Prevention of VTE in Immobilized Patients Trial (PREVENT)2 was a multicenter, randomized, double-blind study comparing the LMWH dalteparin (5,000 IU daily given subcutaneously for 14 days) with placebo in 3,706 acutely ill medical patients. Over 90 days of follow-up, the risk of VTE was reduced by 44% in patients assigned to dalteparin compared with those assigned to placebo (P = .0015) (Figure 1).
The Arixtra for Thromboembolism Prevention in a Medical Indications Study (ARTEMIS)3 randomized 849 medical patients 60 years or older to 6 to 14 days of therapy with the selective factor Xa inhibitor fondaparinux (2.5 mg once daily subcutaneously) or placebo. Compared with the placebo group, fondaparinux recipients had a 47% lower risk of developing VTE by day 15 (P = .029) (Figure 1).
Fewer events and fatal PEs, but no effect on all-cause mortality
A recent meta-analysis by Dentali et al10 further demonstrates the efficacy of anticoagulant therapy for preventing symptomatic VTE in hospitalized medical patients. This analysis included several other trials in addition to the three reviewed above,1–3 for a total of nine randomized studies (seven of which were dou-ble-blind) comprising 19,958 patients. Across the nine studies, anticoagulant prophylaxis was clearly superior to placebo in preventing fatal PE (relative risk, 0.38 [95% CI, 0.21 to 0.69]). There was a strong trend toward a reduction in symptomatic deep vein thrombosis (DVT) with prophylaxis but no effect on all-cause mortality. The meta-analysis also provided reassurance that prophylaxis does not increase the rate of major bleeding.
HOW DO THE PROPHYLAXIS OPTIONS STACK UP?
What the ACCP recommends
Current ACCP guidelines recommend the use of either LMWH or low-dose UFH (5,000 U subcutaneously two or three times daily) as a Grade 1A recommendation for VTE prophylaxis in patients with medical conditions and risk factors for VTE.9 This represents the guidelines’ highest level of recommendation, ie, one that is based on randomized controlled trials (RCTs) without important limitations. In contrast, the 2006 International Consensus Statement, developed as a collaborative effort among expert bodies on VTE, specified a more narrow dosing recommendation for UFH in this patient population (5,000 U three times daily, not twice daily) as well as specifying 40 mg once daily as the recommended dose of enoxaparin and 5,000 IU once daily as the recommended dose of dalteparin.11
For medical patients with risk factors for VTE who have a contraindication to anticoagulant prophylaxis, the ACCP guidelines recommend the use of graduated compression stockings or intermittent pneumatic compression devices as a Grade 1C+ recommendation (“no RCTs but strong RCT results can be unequivocally extrapolated, or overwhelming evidence from observational studies”9).
Current ACCP guidelines do not address the use of fondaparinux in their recommendations for VTE prophylaxis in medical patients.
Getting a handle on bleeding risk
Patient characteristics that exclude pharmacologic thromboprophylaxis due to bleeding risk are generally limited to active bleeding or coagulopathy, as demonstrated by a platelet count less than 50,000 cells/µL or an international normalized ratio greater than 1.5. Additionally, bleeding risk should be carefully assessed if an invasive procedure is planned during a patient’s hospital stay.
It is worth noting that the anticoagulant doses used for VTE prophylaxis are a fraction of those used for treatment of VTE. Thus, if a patient would be treated with full-dose anticoagulation if VTE developed, then that patient should be eligible for VTE prophylaxis.
Because the use of mechanical forms of prophylaxis in medical patients is not truly evidence-based, mechanical prophylaxis should be reserved for medical patients who have a contraindication to anticoagulants, or for use in combination with anticoagulants in patients at very high risk of VTE.
UFH vs LMWH
Two meta-analyses have compared UFH with LMWH for VTE prevention in medical patients.12,13 In a recent analysis that included 10 trials directly comparing the two therapies, 14 trials comparing UFH with control, and 11 trials comparing LMWH with control, Wein et al found a lower risk of DVT with LMWH than with UFH (relative risk, 0.68 [95% CI, 0.52 to 0.88]) but no difference between the therapies in mortality or bleeding risk.12 In an earlier and smaller analysis, Mismetti et al found no significant differences between UFH and LMWH in preventing DVT or death but did find a significant reduction in major bleeding episodes with LMWH versus three-times-daily UFH (52% relative reduction; P = .049).13
Randomized trials also reveal that enoxaparin 40 mg once daily is as efficacious as UFH 5,000 U three times daily for VTE prevention in medical patients.14,15 The above analysis by Wein et al12 and an additional meta-analysis by King and colleagues16 found that three-times-daily dosing of UFH is more efficacious than twice-daily dosing of UFH, but at the expense of more bleeding, including major bleeding.
Economic considerations
Because of differences in drug acquisition costs between UFH and the LMWH agents, several economic evaluations have compared the use of these therapies for prophylaxis in medical patients at risk of VTE.
In an analysis of hospital costs for medical patients receiving VTE prophylaxis from more than 330 US hospitals for the period 2001–2004, Burleigh et al found that mean total hospital costs were higher for patients who received UFH than for those who received LMWH ($7,615 vs $6,866) even though mean drug costs were higher for LMWH ($791 vs $569 for UFH).17 A reduction in hospital length of stay appeared to contribute to the overall savings with LMWH; other contributors may have included costs associated with heparin-induced thrombocytopenia (HIT) in UFH recipients or the extra nursing time required for administering UFH in two or three daily doses.
Leykum et al used a decision analysis model to estimate the economic effect of substituting enoxaparin for UFH in hospitalized medical patients for whom VTE prophylaxis is indicated.18 Cost data were based on Medicare reimbursement rates as well as drug and laboratory costs for a multi-institutional health system. The model assumed HIT incidence rates of 2.7% with UFH and 0.3% with enoxaparin. It also assumed the cost of a daily dose to be $4 for UFH versus $84 for enoxaparin. From the payer perspective, the model showed that substituting enoxaparin for UFH would reduce the overall cost of care by $28.61 per day on a per-patient basis, despite enoxaparin’s higher acquisition cost, and would save $4,550 per quality-adjusted life-year by reducing the incidence of HIT.
Another cost analysis confirms the association between HIT and increased hospital costs. Creekmore et al retrospectively analyzed data from 10,121 adult medical patients who received VTE prophylaxis at the University of Utah Hospital in Salt Lake City from August 2000 to November 2004.19 They found that an admission during which HIT developed incurred a mean cost of $56,364, compared with $15,231 for an admission without HIT. Because LMWH was associated with a lower incidence of HIT compared with UFH (0.084% vs 0.51%, respectively), LMWH reduced the incremental cost of VTE prophylaxis by $13.88 per patient compared with UFH.
THE EXCLAIM TRIAL: IS THERE A ROLE FOR EXTENDED PROPHYLAXIS?
Although the previously discussed studies have clearly demonstrated the benefit of in-hospital VTE prophylaxis for acutely ill medical patients, none has rigorously examined extended-duration out-of-hospital prophylaxis in these patients. This represents an important gap in the literature, since a substantial proportion of VTE develops in the outpatient setting within 3 months of a hospitalization, and most outpatient VTE episodes occur within 1 month of a preceding hospitalization.20
To begin to fill this gap, the Extended Clinical Prophylaxis in Acutely Ill Medical Patients (EXCLAIM) trial was conducted to compare extended-duration LMWH prophylaxis with a standard LMWH prophylaxis regimen in acutely ill medical patients using a prospective, multicenter, randomized, double-blind, placebo-controlled design.21
Patients and study design
Patients were eligible for enrollment if they were aged 40 years or older and had recent immobilization (≤ 3 days), a predefined acute medical illness, and either level 1 mobility (total bed rest or sedentary state) or level 2 mobility (level 1 with bathroom privileges). The predefined acute medical illnesses consisted of New York Heart Association class III/IV heart failure, acute respiratory insufficiency, or other acute medical conditions, including post-acute ischemic stroke, acute infection without septic shock, and active cancer.
All patients received open-label enoxaparin 40 mg subcutaneously once daily for 10 ± 4 days, after which they were randomized to either enoxaparin 40 mg subcutaneously once daily or placebo for an additional 28 ± 4 days.
The primary efficacy end point was the incidence of VTE events, defined as asymptomatic DVT documented by mandatory ultrasonography at the end of the double-blind treatment period (28 ± 4 days) or as symptomatic DVT, symptomatic PE, or fatal PE at any time during the double-blind period. Symptomatic DVT was confirmed by objective tests; PE was confirmed by ventilation-perfusion scan, computed tomography, angiography, or autopsy.
Secondary efficacy end points were mortality at the end of the double-blind period, at 3 months, and at 6 months, as well as the incidence of VTE at 3 months.
The primary safety outcome measure was the incidence of major hemorrhage during the double-blind period; secondary safety measures were rates of major and minor hemorrhage, minor hemorrhage, HIT, and serious adverse events.
Population amended at planned interim analysis
After approximately half of the patients were enrolled, a planned and blinded interim analysis for futility concluded that the study was unlikely to show a statistically significant advantage of enoxaparin over placebo. The trial’s steering committee followed the suggestion of its data safety monitoring board to redefine the inclusion criteria to refocus enrollment on patients with a high risk of VTE. A blinded analysis was performed to identify this subgroup.
The resulting amended inclusion criteria were the same as above except that level 2 mobility had to be accompanied by at least one of three additional high-risk criteria: (1) age greater than 75 years, (2) history of prior VTE, or (3) diagnosis of cancer.
The trial’s main exclusion criteria were evidence of active bleeding, a contraindication to anticoagulation, receipt of prophylactic LMWH or UFH more than 72 hours prior to enrollment, treatment with an oral anticoagulant within 72 hours of enrollment, major surgery within the prior 3 months, cerebral stroke with bleeding, and persistent renal failure (creatinine clearance < 30 mL/min).


Results
The amended study population included 5,105 patients, 5,049 of whom received open-label enoxaparin. Of this group, 2,013 were randomized to active extended prophylaxis with enoxparain and 2,027 to placebo. Baseline characteristics, including level of mobility, were similar between the two groups.
The efficacy of extended prophylaxis with enoxaparin was enduring, as the cumulative incidence of VTE events at day 90 was significantly lower in enoxaparin recipients than in placebo recipients (3.0% vs 5.2%; relative reduction of 42%; P = .0115).
There was no difference in all-cause mortality at 6 months between the enoxaparin and placebo groups (10.1% vs 8.9%, respectively; P = .179).
Safety. Major hemorrhage was significantly more frequent in the enoxaparin arm, occurring in 0.60% of enoxaparin recipients compared with 0.15% of placebo recipients (P = .019). Minor bleeding was also more common with enoxaparin (5.20% vs 3.70%; P = .024).
Conclusions
The EXCLAIM trial found that an extended-duration (38-day) enoxaparin regimen significantly reduced the overall incidence of VTE relative to a 10-day enoxaparin regimen in acutely ill medical patients with reduced mobility. At the same time, the extended regimen was associated with a significant increase in the rate of major bleeding, although the incidence of major bleeding was low. The investigators concluded that the net clinical effect of extended-duration prophylaxis with enoxaparin is favorable, as only 46 patients would need to be treated to prevent one VTE event, whereas 224 patients would need to be treated to result in one major bleeding event.21
For this reason, it is reasonable to consider extended prophylaxis for hospitalized medical patients after identifying these patients’ risk factors. In keeping with the trial’s amended inclusion criteria, patients older than age 75 and those with cancer or prior VTE should receive special consideration for extended prophylaxis.
RECOMMENDED APPROACH TO VTE PREVENTION IN HOSPITALIZED MEDICAL PATIENTS
- All hospitalized medical patients should be screened at the time of admission, and patients at risk for VTE should receive prophylaxis.
- All patients with reduced mobility and one or more other risk factors for VTE are candidates for prophylaxis.
- Patients should be reassessed daily for the development of VTE risk factors during their hospitalization if risk factors are absent on admission.
- If screening or reassessment reveals any VTE risk factors, pharmacologic prophylaxis is the mainstay of therapy. If exclusion criteria for pharmacologic prophylaxis are present, mechanical prophylaxis with graduated compression stockings and intermittent compression devices should be used. For very high-risk medical patients without a contraindication to anticoagulants, combination prophylaxis with both an anticoagulant and mechanical devices is preferred.
- In this patient population, LMWH agents are preferred as pharmacologic prophylaxis over UFH and over fondaparinux (which is not currently approved by the US Food and Drug Administration for this population).
- If UFH is to be used in this patient population, 5,000 U three times daily is the preferred dosage.
- Extended pharmacologic prophylaxis should be considered in patients older than age 75 and in patients with a cancer diagnosis or a prior VTE episode.
DISCUSSION: ADDITIONAL PERSPECTIVES FROM THE AUTHORS
Dr. Jaffer: Dr. Spyropoulos, are there any guidelines, other than those from the ACCP, that speak to VTE prophylaxis in hospitalized medical patients? If so, what are their take-home messages and how do they differ from the ACCP guidelines?
Dr. Spyropoulos: I was part of the group that developed the International Consensus Statement (ICS) published in International Angiology in 2006,11 which is more recent than the latest ACCP guidelines, which were published in 2004. The ICS drew on much of the same data that the ACCP did, but we did an updated review of clinical trials.
For VTE prophylaxis in hospitalized medical patients, the ICS recommendations are more specific with regard to the type, dose, and dosing frequency of anticoagulant agents. First, they specify doses for both LMWH agents in this patient setting: 40 mg once daily for enoxaparin, and 5,000 IU once daily for dalteparin.
The ICS document also states that if UFH is the choice for prophylaxis, a regimen of 5,000 U three times daily should be considered. In the past year alone, two analyses suggest that three-times-daily dosing of UFH in medical patients provides superior efficacy to twice-daily dosing, although perhaps at the expense of more bleeding episodes.12,16 It is important to remember that no large placebo-controlled trial supports the efficacy of a UFH regimen of 5,000 U twice daily in this population.
Finally, the ICS document states that fondaparinux 2.5 mg once daily is a viable option for prophylaxis in medical patients, based on the ARTEMIS trial,3 even though this represents an off-label use.
Dr. Jaffer: Real-world use of VTE prophylaxis is far from optimal, especially in medical patients, and this is partly a result of system-of-care issues. I’d like to conclude by asking each of my colleagues to offer your perspectives on how your own institutions have improved their systems of care to promote better use of VTE prophylaxis and what lessons might be shared with others. Dr. McKean, you work at Brigham and Women’s Hospital, which recently reported impressive results with an electronic alert system designed to increase clinicians’ consideration of VTE risk assessment and use of prophylaxis.24 Please tell us about that study and the alert system.
Dr. McKean: Despite many educational initiatives at Brigham and Women’s Hospital, there were still some patients at high risk for VTE who were not receiving appropriate prophylaxis. What Dr. Samuel Goldhaber and his colleagues wanted to determine was whether changing the system of care could result in a reduced incidence of VTE.24 They devised a computer software program linked to the patient database that used eight common risk factors to determine each hospitalized patient’s risk profile for VTE. Each risk factor was weighted according to a point scale, with major risk factors (cancer, prior VTE, or hypercoagulability) assigned 3 points, the intermediate risk factor of surgery assigned 2 points, and minor risk factors (advanced age, obesity, immobility, or use of hormone replacement therapy or oral contraceptives) assigned 1 point. For patients with a total risk score of 4 or greater, the computer screen generates a color-coded VTE risk alert that requires the physician to acknowledge the alert and choose one of three options: order prophylaxis as appropriate, review a 60-page document on the computer to learn more about prophylaxis, or do nothing.
The study found that hospitalized patients who were randomized to treatment under the computer alert system were significantly more likely to receive VTE prophylaxis and significantly less likely to develop VTE than were patients randomized to a control group. The alert system reduced the risk of DVT or PE at 90 days by 41% in patients considered to be at high risk. It was particularly interesting that the incidence of VTE was lower in the intervention group even when physicians chose not to use prophylaxis, which suggests that simply having this alert system in place improved outcomes, perhaps by raising awareness of the risk of VTE.24
Additional studies are needed to better understand physicians’ behavior and determine why they seem to have a disproportionate fear of the risk of bleeding relative to the risk of clotting, including fatal PE, because that is really the heart of the matter. When patients are not given prophylaxis, often it is because of fear of bleeding. It is not clear, however, why some of these patients did not receive mechanical devices as an alternative form of prophylaxis, but this seems to be the case worldwide, as shown recently by the multinational ENDORSE study.22 Meanwhile, as we await studies to better understand physician perceptions and behaviors regarding prophylaxis, we need to work hard to change the system of care.
Dr. Deitelzweig: Over the past couple of years, the Ochsner Clinic has grown from a one-hospital teaching organization to a seven-hospital system with a mix of closed and open medical staff. The challenge is how to take a process that worked well in the one center, where appropriate prophylaxis was used about 90% of the time, and transfer it to the other centers in the larger system. We have endorsed several types of performance tools, such as the change-acceleration processes used by General Electric. The aim is to share a vision of heightening awareness. To do that, we have worked to mobilize the key stakeholders, at least half of them, to build algorithms that they all will endorse. It is easier said than done, however, and we have found it essential to involve both physicians and non-physician colleagues from pharmacy and nursing who have political and organizational clout.
Dr. Brotman: At Johns Hopkins, I took a bit more draconian approach to this issue because I thought that hospitalists often knew that they should be using VTE prophylaxis but sometimes weren’t, and I am not convinced that clinicians always look at prompts. So we came up with a system that incorporates both billing and documentation simultaneously. We put a hard stop on users’ documentation so that they could not sign off on a note or bill for their care until they checked off the kind of VTE prophylaxis they were using. Since hospitalists ultimately care about billing for their work, this system has at least ensured that everybody has considered and documented VTE prophylaxis on a daily basis. There are other hard stops that can be implemented in computer order-entry systems as well, and we are considering ways to roll them out on a broader scale.
However, all of these systems can have problems because patient situations change from day to day. For instance, VTE prophylaxis is not necessarily indicated in a 38-year-old ambulatory patient who comes in with a sickle cell crisis, but you will need to reconsider if the patient ends up in acute chest syndrome in the intensive care unit. I do not yet have a good way to ensure that this is being done on a daily basis with all patients.
Dr. Amin: At the University of California, Irvine, we implemented an electronic alert system, but we locked users in so that they could not complete their admission orders until they answered questions about VTE prevention. This practice increased our VTE prophylaxis rates tremendously. Because we are a level I trauma center, we allow users to bypass the screens one time, but the next time they log in, even to get a simple lab result, they have to answer the questions about VTE prevention.
With any system you develop, you also have to continue with the education process, because clinicians sometimes get into bad habits or simply forget things.
Dr. Spyropolous: At Lovelace Medical Center, we didn’t have the sophistication of an electronic order-entry system, but we had an experienced clinical pharmacist (the director of inpatient pharmacy) who helped to develop and champion VTE prevention guidelines that have then been used throughout the system in close conjunction with our hospitalists’ rounds. This system has been used successfully for the past 7 years.
- Samama MM, Cohen AT, Darmon JY, et al. A comparison of enoxaparin with placebo for the prevention of venous thrombo-embolism in acutely ill medical patients. Prophylaxis in Medical Patients with Enoxaparin Study Group. N Engl J Med 1999; 341:793–800.
- Leizorovicz A, Cohen AT, Turpie AG, et al. Randomized, placebo-controlled trial of dalteparin for the prevention of venous thromboembolism in acutely ill medical patients. Circulation 2004; 110:874–879.
- Cohen AT, Davidson BL, Gallus AS, et al. Efficacy and safety of fondaparinux for the prevention of venous thromboembolism in older acute medical patients: randomised placebo controlled trial. BMJ 2006; 332:325–329.
- Baglin TP, White K, Charles A. Fatal pulmonary embolism in hos-pitalised medical patients. J Clin Pathol 1997; 50:609–610.
- Piazza G, Seddighzadeh A, Goldhaber SZ. Double trouble for 2,609 hospitalized medical patients who developed deep vein thrombosis: prophylaxis omitted more often and pulmonary embolism more frequent. Chest 2007; 132:554–561.
- Amin A, Stemkowski S, Lin J, Yang G. Thromboprophylaxis rates in US medical centers: success or failure? J Thromb Haemost 2007; 5:1610–1616.
- Geerts WH, Heit JA, Clagett GP, et al. Prevention of venous thromboembolism. Chest 2001; 119(1 Suppl):132S–175S.
- Tapson VF, Decousus H, Pini M, et al. Venous thromboembolism prophylaxis in acutely ill hospitalized medical patients: findings from the International Medical Prevention Registry on Venous Thromboembolism. Chest 2007; 132:936–945.
- Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thrombo-embolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 2004; 126(3 Suppl):338S–400S.
- Dentali F, Douketis JD, Gianni M, Lim W, Crowther MA. Meta-analysis: anticoagulant prophylaxis to prevent symptomatic venous thromboembolism in hospitalized medical patients. Ann Intern Med 2007; 146:278–288.
- Cardiovascular Disease Educational and Research Trust; Cyprus Cardiovascular Disease Educational and Research Trust; European Venous Forum; International Surgical Thrombosis Forum; International Union of Angiology; Union Internationale de Phlébologie. Prevention and treatment of venous thromboembolism. International Consensus Statement (guidelines according to scientific evidence). Int Angiol 2006; 25:101–161.
- Wein L, Wein S, Haas SJ, Shaw J, Krum H. Pharmacological venous thromboembolism prophylaxis in hospitalized medical patients: a meta-analysis of randomized controlled trials. Arch Intern Med 2007; 167:1476–1486.
- Mismetti P, Laporte-Simitsidis S, Tardy B, et al. Prevention of venous thromboembolism in internal medicine with unfractionated or low-molecular-weight heparins: a meta-analysis of randomised clinical trials. Thromb Haemost 2000; 83:14–19.
- Lechler E, Schramm W, Flosbach CW. The venous thrombotic risk in non-surgical patients: epidemiological data and efficacy/safety profile of a low-molecular-weight heparin (enoxaparin). The Prime Study Group. Haemostasis 1996; 26(Suppl 2):49–56.
- Kleber FX, Witt C, Vogel G, et al; the PRINCE Study Group. Randomized comparison of enoxaparin with unfractionated heparin for the prevention of venous thromboembolism in medical patients with heart failure or severe respiratory disease. Am Heart J 2003; 145:614–621.
- King CS, Holley AB, Jackson JL, Shorr AF, Moores LK. Twice vs three times daily heparin dosing for thromboembolism prophylaxis in the general medical population: a meta-analysis. Chest 2007; 131:507–516.
- Burleigh E, Wang C, Foster D, et al. Thromboprophylaxis in medically ill patients at risk for venous thromboembolism. Am J Health Syst Pharm 2006; 63(20 Suppl 6):S23–S29.
- Leykum L, Pugh J, Diuguid D, Papadopoulos K. Cost utility of substituting enoxaparin for unfractionated heparin for prophylaxis of venous thrombosis in the hospitalized medical patient. J Hosp Med 2006; 1:168–176.
- Creekmore FM, Oderda GM, Pendleton RC, Brixner DI. Incidence and economic implications of heparin-induced thrombocytopenia in medical patients receiving prophylaxis for venous thromboembolism. Pharmacotherapy 2006; 26:1438–1445.
- Spencer FA, Lessard D, Emery C, Reed G, Goldberg RJ. Venous thromboembolism in the outpatient setting. Arch Intern Med 2007; 167:1471–1475.
- Hull RD, Schellong SM Tapson VF, et al. Extended-duration venous thromboembolism (VTE) prophylaxis in acutely ill medical patients with recent reduced mobility: the EXCLAIM study. Presentation at: International Society on Thrombosis and Haemo-stasis XXIst Congress; July 6–12, 2007; Geneva, Switzerland.
- Cohen AT, Tapson VF, Bergmann JF, et al. Venous thromboembolism risk and prophylaxis in the acute hospital care setting (ENDORSE study): a multinational cross-sectional study. Lancet 2008; 371:387–394.
- Kahn SR, Panju A, Geerts W, et al; CURVE study investigators. Multicenter evaluation of the use of venous thromboembolism prophylaxis in acutely ill medical patients in Canada. Thromb Res 2007; 119:145–155.
- Kucher N, Koo S, Quiroz R, et al. Electronic alerts to prevent thromboembolism among hospitalized patients. N Engl J Med 2005; 352:969–977.
- Anderson FA Jr, Wheeler HB, Goldberg RJ, et al. A population-based perspective of the hospital incidence and case-fatality rates of deep vein thrombosis and pulmonary embolism. The Worcester DVT Study. Arch Intern Med 1991; 151:933–938.
- Howell MD, Geraci JM, Knowlton AA. Congestive heart failure and outpatient risk of venous thromboembolism: a retrospective, case-control study. J Clin Epidemiol 2001; 54:810–816.
- Smeeth L, Cook C, Thomas S, Hall AJ, Hubbard R, Vallance P. Risk of deep vein thrombosis and pulmonary embolism after acute infection in a community setting. Lancet 2006; 367:1075–1079.
- Alikhan R, Cohen AT, Combe S, et al. Prevention of venous thromboembolism in medical patients with enoxaparin: a subgroup analysis of the MEDENOX study. Blood Coagul Firbinolysis 2003; 14:341–346.
- Anderson FA Jr, Spencer FA. Risk factors for venous thromboembolism. Circulation 2003; 107(23 Suppl 1):I9–I16.
- Greinacher A, Warkentin TE. Recognition, treatment, and prevention of heparin-induced thrombocytopenia: review and update. Thromb Res 2006; 118:165–176.
- Martel N, Lee J, Wells PS. Risk for heparin-induced thrombocytopenia with unfractionated and low-molecular-weight heparin thromboprophylaxis: a meta-analysis. Blood 2005; 106:2710–2715.
- Sanderink GJ, Guimart CG, Ozoux ML, Jariwala NU, Shukla UA, Boutouyrie BX. Pharmacokinetics and pharmacodynamics of the prophylactic dose of enoxaparin once daily over 4 days in patients with renal impairment. Thromb Res 2002; 105:225–231.
- Douketis J, Cook D, Zytaruk N, et al. Dalteparin thromboprophylaxis in critically ill patients with severe renal insufficiency: the DIRECT study [abstract]. J Thromb Haemost 2007; 5(Suppl 2): P-S-680.
- Scholten DJ, Hoedema RM, Scholten SE. A comparison of two different prophylactic dose regimens of low molecular weight heparin in bariatric surgery. Obes Surg 2002; 12:19–24.
- Samama MM, Cohen AT, Darmon JY, et al. A comparison of enoxaparin with placebo for the prevention of venous thrombo-embolism in acutely ill medical patients. Prophylaxis in Medical Patients with Enoxaparin Study Group. N Engl J Med 1999; 341:793–800.
- Leizorovicz A, Cohen AT, Turpie AG, et al. Randomized, placebo-controlled trial of dalteparin for the prevention of venous thromboembolism in acutely ill medical patients. Circulation 2004; 110:874–879.
- Cohen AT, Davidson BL, Gallus AS, et al. Efficacy and safety of fondaparinux for the prevention of venous thromboembolism in older acute medical patients: randomised placebo controlled trial. BMJ 2006; 332:325–329.
- Baglin TP, White K, Charles A. Fatal pulmonary embolism in hos-pitalised medical patients. J Clin Pathol 1997; 50:609–610.
- Piazza G, Seddighzadeh A, Goldhaber SZ. Double trouble for 2,609 hospitalized medical patients who developed deep vein thrombosis: prophylaxis omitted more often and pulmonary embolism more frequent. Chest 2007; 132:554–561.
- Amin A, Stemkowski S, Lin J, Yang G. Thromboprophylaxis rates in US medical centers: success or failure? J Thromb Haemost 2007; 5:1610–1616.
- Geerts WH, Heit JA, Clagett GP, et al. Prevention of venous thromboembolism. Chest 2001; 119(1 Suppl):132S–175S.
- Tapson VF, Decousus H, Pini M, et al. Venous thromboembolism prophylaxis in acutely ill hospitalized medical patients: findings from the International Medical Prevention Registry on Venous Thromboembolism. Chest 2007; 132:936–945.
- Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thrombo-embolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 2004; 126(3 Suppl):338S–400S.
- Dentali F, Douketis JD, Gianni M, Lim W, Crowther MA. Meta-analysis: anticoagulant prophylaxis to prevent symptomatic venous thromboembolism in hospitalized medical patients. Ann Intern Med 2007; 146:278–288.
- Cardiovascular Disease Educational and Research Trust; Cyprus Cardiovascular Disease Educational and Research Trust; European Venous Forum; International Surgical Thrombosis Forum; International Union of Angiology; Union Internationale de Phlébologie. Prevention and treatment of venous thromboembolism. International Consensus Statement (guidelines according to scientific evidence). Int Angiol 2006; 25:101–161.
- Wein L, Wein S, Haas SJ, Shaw J, Krum H. Pharmacological venous thromboembolism prophylaxis in hospitalized medical patients: a meta-analysis of randomized controlled trials. Arch Intern Med 2007; 167:1476–1486.
- Mismetti P, Laporte-Simitsidis S, Tardy B, et al. Prevention of venous thromboembolism in internal medicine with unfractionated or low-molecular-weight heparins: a meta-analysis of randomised clinical trials. Thromb Haemost 2000; 83:14–19.
- Lechler E, Schramm W, Flosbach CW. The venous thrombotic risk in non-surgical patients: epidemiological data and efficacy/safety profile of a low-molecular-weight heparin (enoxaparin). The Prime Study Group. Haemostasis 1996; 26(Suppl 2):49–56.
- Kleber FX, Witt C, Vogel G, et al; the PRINCE Study Group. Randomized comparison of enoxaparin with unfractionated heparin for the prevention of venous thromboembolism in medical patients with heart failure or severe respiratory disease. Am Heart J 2003; 145:614–621.
- King CS, Holley AB, Jackson JL, Shorr AF, Moores LK. Twice vs three times daily heparin dosing for thromboembolism prophylaxis in the general medical population: a meta-analysis. Chest 2007; 131:507–516.
- Burleigh E, Wang C, Foster D, et al. Thromboprophylaxis in medically ill patients at risk for venous thromboembolism. Am J Health Syst Pharm 2006; 63(20 Suppl 6):S23–S29.
- Leykum L, Pugh J, Diuguid D, Papadopoulos K. Cost utility of substituting enoxaparin for unfractionated heparin for prophylaxis of venous thrombosis in the hospitalized medical patient. J Hosp Med 2006; 1:168–176.
- Creekmore FM, Oderda GM, Pendleton RC, Brixner DI. Incidence and economic implications of heparin-induced thrombocytopenia in medical patients receiving prophylaxis for venous thromboembolism. Pharmacotherapy 2006; 26:1438–1445.
- Spencer FA, Lessard D, Emery C, Reed G, Goldberg RJ. Venous thromboembolism in the outpatient setting. Arch Intern Med 2007; 167:1471–1475.
- Hull RD, Schellong SM Tapson VF, et al. Extended-duration venous thromboembolism (VTE) prophylaxis in acutely ill medical patients with recent reduced mobility: the EXCLAIM study. Presentation at: International Society on Thrombosis and Haemo-stasis XXIst Congress; July 6–12, 2007; Geneva, Switzerland.
- Cohen AT, Tapson VF, Bergmann JF, et al. Venous thromboembolism risk and prophylaxis in the acute hospital care setting (ENDORSE study): a multinational cross-sectional study. Lancet 2008; 371:387–394.
- Kahn SR, Panju A, Geerts W, et al; CURVE study investigators. Multicenter evaluation of the use of venous thromboembolism prophylaxis in acutely ill medical patients in Canada. Thromb Res 2007; 119:145–155.
- Kucher N, Koo S, Quiroz R, et al. Electronic alerts to prevent thromboembolism among hospitalized patients. N Engl J Med 2005; 352:969–977.
- Anderson FA Jr, Wheeler HB, Goldberg RJ, et al. A population-based perspective of the hospital incidence and case-fatality rates of deep vein thrombosis and pulmonary embolism. The Worcester DVT Study. Arch Intern Med 1991; 151:933–938.
- Howell MD, Geraci JM, Knowlton AA. Congestive heart failure and outpatient risk of venous thromboembolism: a retrospective, case-control study. J Clin Epidemiol 2001; 54:810–816.
- Smeeth L, Cook C, Thomas S, Hall AJ, Hubbard R, Vallance P. Risk of deep vein thrombosis and pulmonary embolism after acute infection in a community setting. Lancet 2006; 367:1075–1079.
- Alikhan R, Cohen AT, Combe S, et al. Prevention of venous thromboembolism in medical patients with enoxaparin: a subgroup analysis of the MEDENOX study. Blood Coagul Firbinolysis 2003; 14:341–346.
- Anderson FA Jr, Spencer FA. Risk factors for venous thromboembolism. Circulation 2003; 107(23 Suppl 1):I9–I16.
- Greinacher A, Warkentin TE. Recognition, treatment, and prevention of heparin-induced thrombocytopenia: review and update. Thromb Res 2006; 118:165–176.
- Martel N, Lee J, Wells PS. Risk for heparin-induced thrombocytopenia with unfractionated and low-molecular-weight heparin thromboprophylaxis: a meta-analysis. Blood 2005; 106:2710–2715.
- Sanderink GJ, Guimart CG, Ozoux ML, Jariwala NU, Shukla UA, Boutouyrie BX. Pharmacokinetics and pharmacodynamics of the prophylactic dose of enoxaparin once daily over 4 days in patients with renal impairment. Thromb Res 2002; 105:225–231.
- Douketis J, Cook D, Zytaruk N, et al. Dalteparin thromboprophylaxis in critically ill patients with severe renal insufficiency: the DIRECT study [abstract]. J Thromb Haemost 2007; 5(Suppl 2): P-S-680.
- Scholten DJ, Hoedema RM, Scholten SE. A comparison of two different prophylactic dose regimens of low molecular weight heparin in bariatric surgery. Obes Surg 2002; 12:19–24.
Prevention of venous thromboembolism in the cancer surgery patient
Venous thromboembolism (VTE) is a major complication of cancer, occurring in 4% to 20% of patients,1 and is one of the leading causes of death in cancer patients, although these figures are believed to be underestimates, given the low autopsy rates among cancer patients.2 In hospitalized cancer patients specifically, VTE is the second leading cause of death.3,4 The risk of VTE in cancer patients undergoing surgery is three to five times greater than that in surgical patients without cancer.4 Moreover, cancer patients with symptomatic deep vein thrombosis (DVT) exhibit a high risk of recurrent VTE that may persist for many years after the index event.5
VTE PREVENTION POSES PARTICULAR CHALLENGES IN CANCER PATIENTS
Until recently, data on VTE prevention specific to cancer patients have been sparse. Cancer patients have represented only a small subset (< 20%) of participants in most of the largest clinical trials of VTE prophylaxis. Until the past 2 or 3 years, clinicians largely have had to extrapolate their approach to VTE prophylaxis in cancer patients from data in patients without cancer, bearing in mind that cancer patients are among the populations at highest risk of developing VTE.
High rates of VTE, even with prophylaxis
Further insights have come from the @RISTOS project, a Web-based prospective registry of patients undergoing general, urologic, or gynecologic surgery for cancer at multiple centers in Italy.8 Of the 2,372 patients tracked in this study, 82% received in-hospital VTE prophylaxis and 31% received prophylaxis following discharge. Despite this relatively high frequency of prophylaxis, however, the incidence of clinically overt VTE was 2.1% and the incidence of fatal VTE was 0.8%. Notably, most VTE events occurred after hospital discharge, and VTE was the most common cause of 30-day postoperative death in this registry.
RISK FACTORS: CANCER TYPE AND TREATMENT LOOM LARGE
Both the type and stage of a patient’s cancer are important in assessing the risk of VTE. For men, cancers of the prostate, colon, brain, and lung have been associated with an increased risk of VTE. Among women, cancers of the breast, ovary, and lung have been especially implicated as risk factors for VTE.9,10
The type of cancer therapy also influences VTE risk:
- Surgery. Among patients who undergo cancer-related surgery, the rate of proximal DVT is 10% to 20%, the rate of clinically evident PE is 4% to 10%, and the incidence of fatal PE is 0.2% to 5%.8,11
- Systemic treatments, including chemotherapy and hormone therapy, are also associated with an increased risk of VTE.12–15
- Central venous catheters. Approximately 4% of cancer patients who have central venous catheters placed develop clinically relevant VTE.16,17
In addition to the above risks related to cancer treatments, the following have been identified as risk factors for VTE in surgical oncology patients:
- Age greater than 40 years (risk also increases steeply after age 60 and again after age 75)
- Cancer procoagulants
- Thrombophilia
- Length and complications of cancer surgery (ie, often involving tissue trauma and immobilization)
- Debilitation and slow recovery.
Another risk factor worth noting is perioperative transfusion, as illustrated in a recent study of 14,104 adults undergoing colorectal cancer resection.18 The overall incidence of VTE in these patients was 1.0%, and the risk of death was nearly four times as great in patients who developed VTE as in those who did not. Notably, the need for transfusion was a marker of increased risk of VTE, particularly in women: women who received perioperative transfusions had almost double the risk of developing VTE compared with women who did not receive transfusions (P = .004).
CLINICAL TRIALS OF PROPHYLAXIS IN CANCER SURGERY PATIENTS
LMWH vs UFH for in-hospital prophylaxis
Two large randomized, double-blind trials have compared low-molecular-weight heparin (LMWH) with low-dose unfractionated heparin (UFH) for VTE prophylaxis in surgical patients with cancer—the Enoxaparin and Cancer (ENOXACAN) study19 and the Canadian Colorectal Surgery DVT Prophylaxis Trial.20 Patients in these studies underwent surgery for abdominal or pelvic cancer (mostly colorectal cancer). Both studies compared 40 mg of the LMWH enoxaparin given once daily with 5,000 U of UFH given three times daily for 7 to 10 days postoperatively. Outcome measures were the presence of DVT determined by venography on day 7 to 10 and the incidence of symptomatic VTE. Rates of VTE were statistically equivalent between the two treatment arms in both ENOXACAN (14.7% with LMWH vs 18.2% with UFH) and the Canadian Colorectal Surgery study (9.4% with both therapies), as were rates of major bleeding (4.1% with LMWH vs 2.9% with UFH in ENOXACAN; 2.7% with LMWH vs 1.5% with UFH in the Canadian study).
These findings are consistent with a 2001 meta-analysis by Mismetti et al of all available randomized trials comparing LMWH with placebo or with UFH for VTE prophylaxis in general surgery.21 This analysis found no differences in rates of asymptomatic DVT, clinical PE, clinical thromboembolism, death, major hemorrhage, total hemorrhage, wound hematoma, or need for transfusion between LMWH and UFH in patients undergoing either cancer-related surgery or surgery not related to cancer.
Fondaparinux for in-hospital prophylaxis
Subgroup analysis of the large randomized trial known as PEGASUS22 sheds some light on the efficacy of the factor Xa inhibitor fondaparinux relative to LMWH for thromboprophylaxis in cancer surgery patients. PEGASUS compared fondaparinux 2.5 mg once daily with the LMWH dalteparin 5,000 IU once daily for 5 to 9 days in patients undergoing high-risk abdominal surgery. Among the study’s 1,408 patients undergoing surgery for cancer, rates of VTE were 4.7% in the fondaparinux group compared with 7.7% in the LMWH group, a relative risk reduction of 38.6% with fondaparinux (95% CI, 6.7% to 59.6%). In contrast, in the rest of the PEGASUS population (patients undergoing abdominal surgery for reasons other than cancer), LMWH was nonsignificantly more efficacious at preventing VTE than was fondaparinux. Rates of major bleeding in this cancer subgroup were comparable between the two treatments.
Extended prophylaxis
Two additional randomized trials have evaluated extended prophylaxis with LMWH in surgical cancer patients—ENOXACAN II23 and the Fragmin After Major Abdominal Surgery (FAME) study.24
In ENOXACAN II, patients undergoing surgery for abdominal or pelvic cancer first received 6 to 10 days of prophylaxis with enoxaparin 40 mg once daily and then were randomized in a double-blind fashion to an additional 21 days of enoxaparin or placebo.23 Among 332 patients in the intent-to-treat analysis, the rate of VTE at the end of the double-blind phase was reduced from 12.0% with placebo to 4.8% with extended-duration enoxaparin (P = .02), an effect that was maintained at 3-month follow-up (P = .01). There was no significant difference between the two groups in rates of major bleeding events or any bleeding events.
In FAME, patients received 5,000 IU of dalteparin once daily for 1 week following major abdominal surgery and then were randomized in open-label fashion to either placebo or extended prophylaxis with dalteparin for 3 more weeks; a subanalysis examined outcomes in the 198 FAME participants whose abdominal surgery was for cancer.24 Among these 198 cancer surgery patients, the rate of venography-documented VTE at 4 weeks was reduced from 19.6% with placebo to 8.8% with extended-duration dalteparin, a relative reduction of 55% (P = .03). The rate of proximal DVT was reduced from 10.4% to 2.2% with extended prophylaxis, a relative reduction of 79% (P = .02).
The number needed to treat with extended LMWH prophylaxis to prevent one VTE event was 14 in ENOXACAN II23 and 9 in the FAME subanalysis of cancer surgery patients.24
New systematic review of relevant trials
Leonardi et al recently published a systematic review of 26 randomized controlled trials of DVT prophylaxis in 7,639 cancer surgery patients.25 They found the overall incidence of DVT to be 12.7% in those who received pharmacologic prophylaxis compared with 35.2% in controls. They also found high-dose LMWH therapy (> 3,400 U daily) to be associated with a significantly lower incidence of DVT than low-dose LMWH therapy (≤ 3,400 U daily) (7.9% vs 14.5%, respectively; P < .01). No differences were demonstrated between LMWH and UFH in preventing DVT, DVT location, or bleeding. Bleeding complications requiring discontinuation of pharmacologic prophylaxis occurred in 3% of patients overall.
Implications of HIT
The sequelae of heparin-induced thrombocytopenia (HIT) can have major consequences for cancer surgery patients. The incidence of HIT is markedly lower with LMWH than with UFH, as demonstrated in a nested case-control study by Creekmore et al.26 These researchers also found that the average cost of an admission during which HIT developed was nearly four times as great as the average cost of an admission during which UFH or LMWH was given without development of HIT ($56,364 vs $15,231; P < .001).
EVIDENCE IN SPECIFIC ONCOLOGIC POPULATIONS
Most of the patients in the trials reviewed above underwent abdominal surgery for malignancy. Although studies of VTE prophylaxis in patients undergoing nonabdominal cancer surgery are relatively few, some data are available for a few other specific oncologic populations, as reviewed below.
Surgery for gynecologic cancer
There is a paucity of randomized controlled trials or prospective observational studies on VTE and its prevention in the gynecologic cancer surgery population. Based on small historical studies, the postoperative risk of VTE in this population varies from 12% to 35%.27,28 Twice-daily administration of UFH 5,000 U appears to be ineffective as VTE prophylaxis in this population, but increasing the frequency to three times daily reduces VTE risk by 50% to 60% compared with placebo. Once-daily LMWH is comparable to three-times-daily UFH in efficacy and safety in this population.
A systematic Cochrane review of eight randomized controlled trials in patients undergoing major gynecologic surgery revealed that heparin prophylaxis (either UFH or LMWH) reduces the risk of DVT by 70% compared with no prophylaxis, with an identical risk reduction specifically among women with malignancy (odds ratio, 0.30; 95% CI, 0.10 to 0.89).29 This review found no evidence that anticoagulation reduces the risk of PE following major gynecologic surgery. LMWH and UFH were similar in efficacy for preventing DVT and had a comparable risk of bleeding complications.
Surgery for urologic cancer
The risk of VTE and the benefits of thromboprophylaxis also are poorly studied in patients undergoing surgery for urologic cancer.
The risk of VTE varies with the type of urologic surgery and the method used to diagnose VTE. For instance, patients undergoing radical retropubic prostatectomy have been reported to develop DVT at rates of 1% to 3%, PE at rates of 1% to 3%, and fatal PE at a rate of 0.6%, whereas the incidences of these events are somewhat higher in patients undergoing cystectomy: 8% for DVT, 2% to 4% for PE, and 2% for fatal PE. Radiologic diagnosis of thromboembolism in pelvic surgery patients has yielded higher incidences, with DVT rates of 21% to 51% and PE rates of 11% to 22%.30
Small studies suggest that prophylaxis with either low-dose UFH or LMWH is both effective in reducing VTE risk and safe in urologic cancer surgery patients, although pharmacologic prophylaxis poses a possible increased risk of pelvic hematoma and lymphocele formation in this population.30
Neurosurgery
Most neurosurgical procedures are performed for malignancies. The risk of venography-confirmed VTE in patients undergoing neurosurgery is approximately 30% to 40%.31,32 Likewise, the risks of intracranial or intraspinal hemorrhage in these patients are high. For this reason, mechanical methods of VTE prophylaxis are preferred in these patients. The use of anticoagulant prophylaxis remains controversial in this setting, although more recent data suggest that it might be safer than previously recognized.
GUIDELINES FOR VTE PROPHYLAXIS IN THE CANCER SURGERY PATIENT
American College of Chest Physicians
National Comprehensive Cancer Network
The National Comprehensive Cancer Network (NCCN) recently published clinical practice guidelines on venous thromboembolic disease in cancer patients.35 The defined at-risk population for these guidelines is the adult cancer inpatient with a diagnosis of (or clinical suspicion for) cancer. The guidelines recommend prophylactic anticoagulation (category 1 recommendation) with or without a sequential compression device as initial prophylaxis, unless the patient has a relative contraindication to anticoagulation, in which case mechanical prophylaxis (sequential compression device or graduated compression stockings) is recommended. (A category 1 recommendation indicates “uniform NCCN consensus, based on high-level evidence.”)
The NCCN guidelines include a specific recommended risk-factor assessment, which includes noting the patient’s age (VTE risk increases beginning at age 40 and then steeply again at age 75), any prior VTE, the presence of familial thrombophilia or active cancer, the use of medications associated with increased VTE risk (chemotherapy, exogenous estrogen compounds, and thalidomide or lenalidomide), and a number of other risk factors for VTE as outlined in the prior two articles in this supplement. The NCCN guidelines explicitly call for assessment of modifiable risk factors for VTE (ie, smoking or other tobacco use, obesity, and a low level of activity or lack of exercise) and call for active patient education on these factors.
American Society of Clinical Oncology
Our recommended algorithm
LINGERING CHALLENGE OF UNDERUTILIZATION
Despite this consensus on ways to reduce thromboembolic risk in this population and the clear evidence of the benefit of VTE prophylaxis in patients with cancer, data from several registries confirm a persistently low utilization of prophylaxis in patients with cancer.36–38 The global Fundamental Research in Oncology and Thrombosis (FRONTLINE) study surveyed 3,891 clinicians who treat cancer patients regarding their practices with respect to VTE in those patients.36 The survey found that only 52% of respondents routinely used thromboprophylaxis for their surgical patients with cancer. More striking, however, was the finding that most respondents routinely considered thrombo-prophylaxis in only 5% of their medical oncology patients. These data are echoed by findings of other retrospective medical record reviews in patients undergoing major abdominal or abdominothoracic surgery (in many cases for cancer), with VTE prophylaxis rates ranging from 38% to 75%.37,38
SUMMARY
Patients undergoing surgery for cancer have an increased risk of VTE and fatal PE, even when thromboprophylaxis is used. Nevertheless, prophylaxis with either LMWH or UFH does reduce venographic VTE event rates in these patients. If UFH is chosen for prophylaxis, a three-times-daily regimen should be used in this population. In specific surgical cancer populations, especially those undergoing abdominal surgery, out-of-hospital prophylaxis with once-daily LMWH is warranted. Current registries reveal that compliance with established guidelines for VTE prophylaxis in this population is low.


Dr. Jaffer: Dr. Amin, based on your study on thrombo-prophylaxis rates in US medical centers, will you comment on rates of prophylaxis for cancer surgery patients?
Dr. Amin: The overall study included approximately 200,000 medical patients and about 80,000 surgical patients enrolled over more than a 3-year period between 2002 and 2005.39,40 Our goal was to assess rates of prophylaxis and, when it was provided, whether it was appropriate (in terms of type, dosage, and duration) based on the ACCP guidelines. A subanalysis assessed medical cancer patients and surgical cancer patients separately. Medical cancer patients received thromboprophylaxis 56% of the time but received appropriate prophylaxis only 28% of the time. Among surgical cancer patients, appropriate prophylaxis was given only about 24% of the time for those undergoing gynecologic surgery and about 12% of the time for those undergoing neurosurgery. These percentages are consistent with data from other national registries, such as the IMPROVE registry, which documented prophylaxis rates on the order of 45% in medical patients with cancer.41 We also analyzed the data according to individual practitioners and found that medical oncologists use prophylaxis about 25% of the time, which is relatively consistent with other providers, such as internists and surgeons.
So there is a huge opportunity to improve rates of prophylaxis for this group of patients that national guidelines say are at high risk. Why is prophylaxis so underutilized in the cancer population? One factor may be a misperception about the risk of bleeding with anticoagulants. Yet several studies have shown that the rate of bleeding from prophylaxis is extremely low, whether LMWH or UFH is used, so more awareness of actual bleeding risk is needed. Another factor is the obvious focus among internists and oncologists on treating the patient, with perhaps a reduced consideration of prophylaxis and prevention. A third factor may be a concern about thrombocytopenia. However, in our study of prophylaxis rates in US medical centers, we excluded patients who had thrombocytopenia, yet rates of prophylaxis were still low. Nothing in the literature indicates that anticoagulants cannot be used in patients with platelet counts of 50,000 to 150,000 cells/µL or higher, so this suggests that we need to do more education.
Dr. Jaffer: Dr. Brotman, can you tell us more about how clinicians in practice should use prophylaxis in their neurosurgery patients, such as those undergoing craniotomy or spine surgery for cancer? What is the safest and most efficacious way to prevent DVT in these patients?
Dr. Brotman: First, it’s important to recognize that some sort of prophylaxis needs to be used. Neurosurgery patients are at an extremely high risk for thromboembolic events, and such events are often fatal in these patients. Having said that, the jury is still out on whether the prophylaxis in these patients should be compression devices or anticoagulation. This gives physicians some latitude in their decisions. They can decide not to use pharmacologic prophylaxis so long as they use pneumatic compression devices consistently, perhaps even starting during the operation and certainly throughout hospitalization when the patient is immobilized.
Certainly, the concerns about using full-dose anticoagulation in the immediate postoperative setting in neurosurgery patients are valid. Yet these patients are at very high risk for thromboembolic events, and if we take too cautious an approach to prophylaxis in the immediate perioperative setting, more patients are going to have thromboembolic events, at which point management decisions become much more difficult. The risk of intracranial bleeding with anticoagulation to treat a patient who develops a DVT at postoperative day 10 will certainly be higher than it would have been with lower-dose perioperative prophylactic anticoagulation. Plus, if you put in a filter at that point, the outcomes tend to be poor. Therefore, I believe there is some degree of risk that we should be willing to take with regard to perioperative bleeding, even in neurosurgery patients.
Dr. McKean: I’d like to make a point about combination prophylaxis. At many institutions, compression stockings and sequential compression devices are used preoperatively and intraoperatively, and then pharmacologic prophylaxis—for example, twice-daily UFH—is used postoperatively. There is concern that these patients are hypercoagulable, and most clinicians believe that mechanical prophylaxis alone, even with sequential compression devices plus compression stockings, is not aggressive enough in these high-risk patients.
Dr. Jaffer: Dr. Spyropoulos, what is the optimal duration of pharmacologic prophylaxis for cancer surgery patients?
Dr. Spyropoulos: First let’s consider in-hospital prophylaxis. The supportive data for in-hospital prophylaxis are strong, and the duration of therapy used in the major in-hospital prophylaxis trials was 7 to 10 days. With regard to extended prophylaxis, we have at least two moderately sized randomized controlled trials, ENOXACAN II23 and the substudy of FAME,24 that demonstrated that extending prophylaxis with LMWH at doses of 3,400 U once daily (5,000 IU of dalteparin; 40 mg of enoxaparin) reduces VTE risk at postoperative day 30. Also, recent data from the @RISTOS registry show that in cancer surgery patients, especially those having abdominal or pelvic procedures, the leading cause of 30-day mortality was VTE.8 This registry also shows that despite prophylaxis, the rate of symptomatic VTE can be as high as 2%, with the rate of fatal VTE approaching 1%. Thus, in cancer patients undergoing abdominal or pelvic surgery, physicians should strongly consider prophylaxis of up to 30 days’ duration.
Dr. Jaffer: One striking finding from the @RISTOS registry was that 40% of VTE events in these cancer surgery patients occurred after postoperative day 21. This really underscores the need to consider prophylaxis for at least 4 weeks in these patients in real-world practice.
Dr. Brotman: The other striking finding from that registry was that the in-hospital prophylaxis rate was quite high, about 80%, and the rate of extended prophylaxis approached 35%. These are rates that are rarely achieved in clinical practice. Yet despite these high levels of prophylaxis, patients in this registry still had a high incidence of morbidity and mortality from VTE. This suggests that we need to improve our out-of-hospital VTE prevention paradigms.
Dr. Jaffer: Dr. Deitelzweig, oncologists and internists are often unsure about whether their ambulatory cancer patients who are receiving chemotherapy should be on any form of prophylaxis. What is your opinion?
Dr. Deitelzweig: That question comes up regularly because these patients are encountered across many medical specialties. At this point, all of the large organizations, including ASCO and NCCN, are advocating that prophylaxis is not indicated for such patients.
- Lyman GH, Khorana AA, Falanga A, et al. American Society of Clinical Oncology guideline: recommendations for venous thromboembolism prophylaxis and treatment in patients with cancer. J Clin Oncol 2007; 25:5490–5505.
- Khorana AA, Francis CW, Culakova E, et al. Thromboembolism is a leading cause of death in cancer patients receiving outpatient chemotherapy. J Thromb Haemost 2007; 5:632–634.
- Ambrus JL, Ambrus CM, Mink IB, Pickren JW. Causes of death in cancer patients. J Med 1975; 6:61–64.
- Donati MB. Cancer and thrombosis. Haemostasis 1994; 24:128–131.
- Prandoni P, Lensing AW, Cogo A, et al. The long-term clinical course of acute deep venous thrombosis. Ann Intern Med 1996; 125:1–7.
- Haas S, Wolf H, Kakkar AK, Fareed J, Encke A. Prevention of fatal pulmonary embolism and mortality in surgical patients: a randomized double-blind comparison of LMWH with unfractionated heparin. Thromb Haemost 2005; 94:814–819.
- Kakkar AK, Haas S, Wolf H, Encke A. Evaluation of perioperative fatal pulmonary embolism and death in cancer surgical patients: the MC-4 cancer substudy. Thromb Haemost 2005; 94:867–871.
- Agnelli G, Bolis G, Capussotti L, et al. A clinical outcome-based prospective study on venous thromboembolism after cancer surgery: the @RISTOS project. Ann Surg 2006; 243:89–95.
- Levitan N, Dowlati A, Remick SC, et al. Rates of initial and recurrent thromboembolic disease among patients with malignancy versus those without malignancy: risk analysis using Medicare claims data. Medicine (Baltimore) 1999; 78:285–291.
- Levine M, Gent M, Hirsh J, et al. A comparison of low-molecular-weight heparin administered primarily at home with unfractionated heparin administered in the hospital for proximal deep-vein thrombosis. N Engl J Med 1996; 334:677–681.
- Bergqvist D. Risk of venous thromboembolism in patients undergoing cancer surgery and options for thromboprophylaxis. J Surg Oncol 2007; 95:167–174.
- Heit JA, Silverstein MD, Mohr DN, et al. Risk factors for deep vein thrombosis and pulmonary embolism: a population-based case-control study. Arch Intern Med 2000; 160:809–815.
- Sallah S, Wan JY, Nguyen NP. Venous thrombosis in patients with solid tumors: determination of frequency and characteristics. Thromb Haemost 2002; 87:575–579.
- Kröger K, Weiland D, Ose C, et al. Risk factors for venous thromboembolic events in cancer patients. Ann Oncol 2006; 17:297–303.
- Blom JW, Vanderschoot JP, Oostindiër MJ, et al. Incidence of venous thrombosis in a large cohort of 66,329 cancer patients: results of a record linkage study. J Thromb Haemost 2006; 4:529–535.
- Couban S, Simpson DR, Barnett MJ, et al. A randomized multicenter comparison of bone marrow and peripheral blood in recipients of matched sibling allogeneic transplants for myeloid malignancies. Blood 2002; 100:1525–1531.
- Walshe LJ, Malak SF, Eagan J, Sepkowitz KA. Complication rates among cancer patients with peripherally inserted central catheters. J Clin Oncol 2002; 20:3276–3281.
- Nilsson KR, Berenholtz SM, Garrett-Mayer E, et al. Association between venous thromboembolism and perioperative allogeneic transfusion. Arch Surg 2007; 142:126–133.
- Efficacy and safety of enoxaparin versus unfractionated heparin for prevention of deep vein thrombosis in elective cancer surgery: a double-blind randomized multicentre trial with venographic assessment. ENOXACAN Study Group. Br J Surg 1997; 84:1099–1103.
- McLeod RS, Geerts WH, Sniderman KW, et al. Subcutaneous heparin versus low-molecular-weight heparin as thromboprophylaxis in patients undergoing colorectal surgery: results of the Canadian Colorectal DVT Prophylaxis Trial: a randomized, double-blind trial. Ann Surg 2001; 233:438–444.
- Mismetti P, Laporte S, Darmon JY, Buchmüller A, Decousus H. Meta-analysis of low molecular weight heparin in the prevention of venous thromboembolism in general surgery. Br J Surg 2001; 88:913–930.
- Agnelli G, Bergqvist D, Cohen AT, et al, on behalf of the PEGASUS investigators. Randomized clinical trial of postoperative fondaparinux versus perioperative dalteparin for prevention of venous thromboembolism in high-risk abdominal surgery. Br J Surg 2005; 92:1212–1220.
- Bergqvist D, Agnelli G, Cohen AT, et al. Duration of prophylaxis against venous thromboembolism with enoxaparin after surgery for cancer. N Engl J Med 2002; 346:975–980.
- Rasmussen MS, Wille-Jorgensen P, Jorgensen LN, et al. Prolonged thromboprophylaxis with low molecular weight heparin (dalteparin) following major abdominal surgery for malignancy [abstract 186]. Blood 2003; 102:56a.
- Leonardi MJ, McGory ML, Ko CY. A systematic review of deep venous thrombosis prophylaxis in cancer patients: implications for improving quality. Ann Surg Oncol 2007; 14:929–936.
- Creekmore FM, Oderda GM, Pendleton RC, Brixner DI. Incidence and economic implications of heparin-induced thrombocytopenia in medical patients receiving prophylaxis for venous thromboembolism. Pharmacotherapy 2006; 26:1438–1445.
- Walsh JJ, Bonnar J, Wright FW. A study of pulmonary embolism and deep leg vein thrombosis after major gynaecological surgery using labeled fibrinogen-phlebography and lung scanning. J Obstet Gynaecol Br Commonw 1974; 81:311–316.
- Clarke-Pearson DL, Synan IS, Coleman RE, et al. The natural history of postoperative venous thromboemboli in gynecologic oncology: a prospective study of 382 patients. Am J Obstet Gynecol 1984; 148:1051–1054.
- Oates-Whitehead RM, D’Angelo A, Mol B. Anticoagulant and aspirin prophylaxis for preventing thromboembolism after major gynaecological surgery. Cochrane Database Syst Rev 2003; (4):CD003679.
- Kibel AS, Loughlin KR. Pathogenesis and prophylaxis of postoperative thromboembolic disease in urological pelvic surgery. J Urol 1995; 153:1763–1774.
- Agnelli G, Piovella F, Buoncristiani P, et al. Enoxaparin plus compression stockings compared with compression stockings alone in the prevention of venous thromboembolism after elective neurosurgery. N Engl J Med 1998; 339:80–85.
- Semrad TJ, O’Donnell R, Wun T, et al. Epidemiology of venous thromboembolism in 9489 patients with malignant glioma. J Neurosurg 2007; 106:601–608.
- Iorio A, Agnelli G. Low-molecular-weight and unfractionated heparin for prevention of venous thromboembolism in neurosurgery: a meta-analysis. Arch Intern Med 2000; 160:2327–2332.
- Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 2004; 126(3 Suppl):338S–400S.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology. Venous thromboembolic disease. V.1.2007. http://www.nccn.org/professionals/physician_gls/PDF/vte.pdf. Accessed December 5, 2007.
- Kakkar AK, Levine M, Pinedo HM, Wolff R, Wong J. Venous thrombosis in cancer patients: insights from the FRONTLINE survey. Oncologist 2003; 8:381–388.
- Stratton MA, Anderson FA, Bussey HI, et al. Prevention of venous thromboembolism: adherence to the 1995 American College of Chest Physicians consensus guidelines for surgical patients. Arch Intern Med 2000; 160:334–340.
- Bratzler DW, Raskob GE, Murray CK, Bumpus LJ, Piatt DS. Underuse of venous thromboembolism prophylaxis for general surgery patients: physician practices in the community hospital setting. Arch Intern Med 1998; 158:1909–1912.
- Amin A, Stemkowski S, Lin J, et al. Thromboprophylaxis rates in US medical centers: success or failure? J Thromb Haemost 2007; 5:1610–1616.
- Amin A, Stemkowski S, Lin J, Yang G. Thromboprophylaxis in US hospitals: adherence to the 6th American College of Chest Physicians’ recommendations for at-risk medical and surgical patients. Abstract presented at: 41st Midyear Clinical Meeting of the American Society of Health-System Pharmacists; December 3–7, 2006; Anaheim, CA.
- Tapson VF, Decousus H, Pini M, et al. Venous thromboembolism prophylaxis in acutely ill hospitalized medical patients: findings from the International Medical Prevention Registry on Venous Thromboembolism. Chest 2007; 132:936–945.
- Clarke-Pearson DL, Synan IS, Dodge R, et al. A randomized trial of low-dose heparin and intermittent pneumatic calf compression for the prevention of deep venous thrombosis after gynecologic oncology surgery. Am J Obstet Gynecol 1993; 168:1146–1154.
- Einstein MH, Pritts EA, Hartenbach EM. Venous thromboembolism prevention in gynecologic cancer surgery: a systematic review. Gynecol Oncol 2007; 105:813–819.
- Clarke-Pearson DL, Synan IS, Hinshaw WM, Coleman RE, Creasman WT. Prevention of postoperative venous thromboembolism by external pneumatic calf compression in patients with gynecologic malignancy. Obstet Gynecol 1984; 63:92–98.
- Ruff RL, Posner JB. Incidence and treatment of peripheral venous thrombosis in patients with glioma. Ann Neurol 1983; 13:334–336.
- Levin JM, Schiff D, Loeffler JS, Fine HA, Black PM, Wen PY. Complications of therapy for venous thromboembolic disease in patients with brain tumors. Neurology 1993; 43:1111–1114.
Venous thromboembolism (VTE) is a major complication of cancer, occurring in 4% to 20% of patients,1 and is one of the leading causes of death in cancer patients, although these figures are believed to be underestimates, given the low autopsy rates among cancer patients.2 In hospitalized cancer patients specifically, VTE is the second leading cause of death.3,4 The risk of VTE in cancer patients undergoing surgery is three to five times greater than that in surgical patients without cancer.4 Moreover, cancer patients with symptomatic deep vein thrombosis (DVT) exhibit a high risk of recurrent VTE that may persist for many years after the index event.5
VTE PREVENTION POSES PARTICULAR CHALLENGES IN CANCER PATIENTS
Until recently, data on VTE prevention specific to cancer patients have been sparse. Cancer patients have represented only a small subset (< 20%) of participants in most of the largest clinical trials of VTE prophylaxis. Until the past 2 or 3 years, clinicians largely have had to extrapolate their approach to VTE prophylaxis in cancer patients from data in patients without cancer, bearing in mind that cancer patients are among the populations at highest risk of developing VTE.
High rates of VTE, even with prophylaxis
Further insights have come from the @RISTOS project, a Web-based prospective registry of patients undergoing general, urologic, or gynecologic surgery for cancer at multiple centers in Italy.8 Of the 2,372 patients tracked in this study, 82% received in-hospital VTE prophylaxis and 31% received prophylaxis following discharge. Despite this relatively high frequency of prophylaxis, however, the incidence of clinically overt VTE was 2.1% and the incidence of fatal VTE was 0.8%. Notably, most VTE events occurred after hospital discharge, and VTE was the most common cause of 30-day postoperative death in this registry.
RISK FACTORS: CANCER TYPE AND TREATMENT LOOM LARGE
Both the type and stage of a patient’s cancer are important in assessing the risk of VTE. For men, cancers of the prostate, colon, brain, and lung have been associated with an increased risk of VTE. Among women, cancers of the breast, ovary, and lung have been especially implicated as risk factors for VTE.9,10
The type of cancer therapy also influences VTE risk:
- Surgery. Among patients who undergo cancer-related surgery, the rate of proximal DVT is 10% to 20%, the rate of clinically evident PE is 4% to 10%, and the incidence of fatal PE is 0.2% to 5%.8,11
- Systemic treatments, including chemotherapy and hormone therapy, are also associated with an increased risk of VTE.12–15
- Central venous catheters. Approximately 4% of cancer patients who have central venous catheters placed develop clinically relevant VTE.16,17
In addition to the above risks related to cancer treatments, the following have been identified as risk factors for VTE in surgical oncology patients:
- Age greater than 40 years (risk also increases steeply after age 60 and again after age 75)
- Cancer procoagulants
- Thrombophilia
- Length and complications of cancer surgery (ie, often involving tissue trauma and immobilization)
- Debilitation and slow recovery.
Another risk factor worth noting is perioperative transfusion, as illustrated in a recent study of 14,104 adults undergoing colorectal cancer resection.18 The overall incidence of VTE in these patients was 1.0%, and the risk of death was nearly four times as great in patients who developed VTE as in those who did not. Notably, the need for transfusion was a marker of increased risk of VTE, particularly in women: women who received perioperative transfusions had almost double the risk of developing VTE compared with women who did not receive transfusions (P = .004).
CLINICAL TRIALS OF PROPHYLAXIS IN CANCER SURGERY PATIENTS
LMWH vs UFH for in-hospital prophylaxis
Two large randomized, double-blind trials have compared low-molecular-weight heparin (LMWH) with low-dose unfractionated heparin (UFH) for VTE prophylaxis in surgical patients with cancer—the Enoxaparin and Cancer (ENOXACAN) study19 and the Canadian Colorectal Surgery DVT Prophylaxis Trial.20 Patients in these studies underwent surgery for abdominal or pelvic cancer (mostly colorectal cancer). Both studies compared 40 mg of the LMWH enoxaparin given once daily with 5,000 U of UFH given three times daily for 7 to 10 days postoperatively. Outcome measures were the presence of DVT determined by venography on day 7 to 10 and the incidence of symptomatic VTE. Rates of VTE were statistically equivalent between the two treatment arms in both ENOXACAN (14.7% with LMWH vs 18.2% with UFH) and the Canadian Colorectal Surgery study (9.4% with both therapies), as were rates of major bleeding (4.1% with LMWH vs 2.9% with UFH in ENOXACAN; 2.7% with LMWH vs 1.5% with UFH in the Canadian study).
These findings are consistent with a 2001 meta-analysis by Mismetti et al of all available randomized trials comparing LMWH with placebo or with UFH for VTE prophylaxis in general surgery.21 This analysis found no differences in rates of asymptomatic DVT, clinical PE, clinical thromboembolism, death, major hemorrhage, total hemorrhage, wound hematoma, or need for transfusion between LMWH and UFH in patients undergoing either cancer-related surgery or surgery not related to cancer.
Fondaparinux for in-hospital prophylaxis
Subgroup analysis of the large randomized trial known as PEGASUS22 sheds some light on the efficacy of the factor Xa inhibitor fondaparinux relative to LMWH for thromboprophylaxis in cancer surgery patients. PEGASUS compared fondaparinux 2.5 mg once daily with the LMWH dalteparin 5,000 IU once daily for 5 to 9 days in patients undergoing high-risk abdominal surgery. Among the study’s 1,408 patients undergoing surgery for cancer, rates of VTE were 4.7% in the fondaparinux group compared with 7.7% in the LMWH group, a relative risk reduction of 38.6% with fondaparinux (95% CI, 6.7% to 59.6%). In contrast, in the rest of the PEGASUS population (patients undergoing abdominal surgery for reasons other than cancer), LMWH was nonsignificantly more efficacious at preventing VTE than was fondaparinux. Rates of major bleeding in this cancer subgroup were comparable between the two treatments.
Extended prophylaxis
Two additional randomized trials have evaluated extended prophylaxis with LMWH in surgical cancer patients—ENOXACAN II23 and the Fragmin After Major Abdominal Surgery (FAME) study.24
In ENOXACAN II, patients undergoing surgery for abdominal or pelvic cancer first received 6 to 10 days of prophylaxis with enoxaparin 40 mg once daily and then were randomized in a double-blind fashion to an additional 21 days of enoxaparin or placebo.23 Among 332 patients in the intent-to-treat analysis, the rate of VTE at the end of the double-blind phase was reduced from 12.0% with placebo to 4.8% with extended-duration enoxaparin (P = .02), an effect that was maintained at 3-month follow-up (P = .01). There was no significant difference between the two groups in rates of major bleeding events or any bleeding events.
In FAME, patients received 5,000 IU of dalteparin once daily for 1 week following major abdominal surgery and then were randomized in open-label fashion to either placebo or extended prophylaxis with dalteparin for 3 more weeks; a subanalysis examined outcomes in the 198 FAME participants whose abdominal surgery was for cancer.24 Among these 198 cancer surgery patients, the rate of venography-documented VTE at 4 weeks was reduced from 19.6% with placebo to 8.8% with extended-duration dalteparin, a relative reduction of 55% (P = .03). The rate of proximal DVT was reduced from 10.4% to 2.2% with extended prophylaxis, a relative reduction of 79% (P = .02).
The number needed to treat with extended LMWH prophylaxis to prevent one VTE event was 14 in ENOXACAN II23 and 9 in the FAME subanalysis of cancer surgery patients.24
New systematic review of relevant trials
Leonardi et al recently published a systematic review of 26 randomized controlled trials of DVT prophylaxis in 7,639 cancer surgery patients.25 They found the overall incidence of DVT to be 12.7% in those who received pharmacologic prophylaxis compared with 35.2% in controls. They also found high-dose LMWH therapy (> 3,400 U daily) to be associated with a significantly lower incidence of DVT than low-dose LMWH therapy (≤ 3,400 U daily) (7.9% vs 14.5%, respectively; P < .01). No differences were demonstrated between LMWH and UFH in preventing DVT, DVT location, or bleeding. Bleeding complications requiring discontinuation of pharmacologic prophylaxis occurred in 3% of patients overall.
Implications of HIT
The sequelae of heparin-induced thrombocytopenia (HIT) can have major consequences for cancer surgery patients. The incidence of HIT is markedly lower with LMWH than with UFH, as demonstrated in a nested case-control study by Creekmore et al.26 These researchers also found that the average cost of an admission during which HIT developed was nearly four times as great as the average cost of an admission during which UFH or LMWH was given without development of HIT ($56,364 vs $15,231; P < .001).
EVIDENCE IN SPECIFIC ONCOLOGIC POPULATIONS
Most of the patients in the trials reviewed above underwent abdominal surgery for malignancy. Although studies of VTE prophylaxis in patients undergoing nonabdominal cancer surgery are relatively few, some data are available for a few other specific oncologic populations, as reviewed below.
Surgery for gynecologic cancer
There is a paucity of randomized controlled trials or prospective observational studies on VTE and its prevention in the gynecologic cancer surgery population. Based on small historical studies, the postoperative risk of VTE in this population varies from 12% to 35%.27,28 Twice-daily administration of UFH 5,000 U appears to be ineffective as VTE prophylaxis in this population, but increasing the frequency to three times daily reduces VTE risk by 50% to 60% compared with placebo. Once-daily LMWH is comparable to three-times-daily UFH in efficacy and safety in this population.
A systematic Cochrane review of eight randomized controlled trials in patients undergoing major gynecologic surgery revealed that heparin prophylaxis (either UFH or LMWH) reduces the risk of DVT by 70% compared with no prophylaxis, with an identical risk reduction specifically among women with malignancy (odds ratio, 0.30; 95% CI, 0.10 to 0.89).29 This review found no evidence that anticoagulation reduces the risk of PE following major gynecologic surgery. LMWH and UFH were similar in efficacy for preventing DVT and had a comparable risk of bleeding complications.
Surgery for urologic cancer
The risk of VTE and the benefits of thromboprophylaxis also are poorly studied in patients undergoing surgery for urologic cancer.
The risk of VTE varies with the type of urologic surgery and the method used to diagnose VTE. For instance, patients undergoing radical retropubic prostatectomy have been reported to develop DVT at rates of 1% to 3%, PE at rates of 1% to 3%, and fatal PE at a rate of 0.6%, whereas the incidences of these events are somewhat higher in patients undergoing cystectomy: 8% for DVT, 2% to 4% for PE, and 2% for fatal PE. Radiologic diagnosis of thromboembolism in pelvic surgery patients has yielded higher incidences, with DVT rates of 21% to 51% and PE rates of 11% to 22%.30
Small studies suggest that prophylaxis with either low-dose UFH or LMWH is both effective in reducing VTE risk and safe in urologic cancer surgery patients, although pharmacologic prophylaxis poses a possible increased risk of pelvic hematoma and lymphocele formation in this population.30
Neurosurgery
Most neurosurgical procedures are performed for malignancies. The risk of venography-confirmed VTE in patients undergoing neurosurgery is approximately 30% to 40%.31,32 Likewise, the risks of intracranial or intraspinal hemorrhage in these patients are high. For this reason, mechanical methods of VTE prophylaxis are preferred in these patients. The use of anticoagulant prophylaxis remains controversial in this setting, although more recent data suggest that it might be safer than previously recognized.
GUIDELINES FOR VTE PROPHYLAXIS IN THE CANCER SURGERY PATIENT
American College of Chest Physicians
National Comprehensive Cancer Network
The National Comprehensive Cancer Network (NCCN) recently published clinical practice guidelines on venous thromboembolic disease in cancer patients.35 The defined at-risk population for these guidelines is the adult cancer inpatient with a diagnosis of (or clinical suspicion for) cancer. The guidelines recommend prophylactic anticoagulation (category 1 recommendation) with or without a sequential compression device as initial prophylaxis, unless the patient has a relative contraindication to anticoagulation, in which case mechanical prophylaxis (sequential compression device or graduated compression stockings) is recommended. (A category 1 recommendation indicates “uniform NCCN consensus, based on high-level evidence.”)
The NCCN guidelines include a specific recommended risk-factor assessment, which includes noting the patient’s age (VTE risk increases beginning at age 40 and then steeply again at age 75), any prior VTE, the presence of familial thrombophilia or active cancer, the use of medications associated with increased VTE risk (chemotherapy, exogenous estrogen compounds, and thalidomide or lenalidomide), and a number of other risk factors for VTE as outlined in the prior two articles in this supplement. The NCCN guidelines explicitly call for assessment of modifiable risk factors for VTE (ie, smoking or other tobacco use, obesity, and a low level of activity or lack of exercise) and call for active patient education on these factors.
American Society of Clinical Oncology
Our recommended algorithm
LINGERING CHALLENGE OF UNDERUTILIZATION
Despite this consensus on ways to reduce thromboembolic risk in this population and the clear evidence of the benefit of VTE prophylaxis in patients with cancer, data from several registries confirm a persistently low utilization of prophylaxis in patients with cancer.36–38 The global Fundamental Research in Oncology and Thrombosis (FRONTLINE) study surveyed 3,891 clinicians who treat cancer patients regarding their practices with respect to VTE in those patients.36 The survey found that only 52% of respondents routinely used thromboprophylaxis for their surgical patients with cancer. More striking, however, was the finding that most respondents routinely considered thrombo-prophylaxis in only 5% of their medical oncology patients. These data are echoed by findings of other retrospective medical record reviews in patients undergoing major abdominal or abdominothoracic surgery (in many cases for cancer), with VTE prophylaxis rates ranging from 38% to 75%.37,38
SUMMARY
Patients undergoing surgery for cancer have an increased risk of VTE and fatal PE, even when thromboprophylaxis is used. Nevertheless, prophylaxis with either LMWH or UFH does reduce venographic VTE event rates in these patients. If UFH is chosen for prophylaxis, a three-times-daily regimen should be used in this population. In specific surgical cancer populations, especially those undergoing abdominal surgery, out-of-hospital prophylaxis with once-daily LMWH is warranted. Current registries reveal that compliance with established guidelines for VTE prophylaxis in this population is low.


Dr. Jaffer: Dr. Amin, based on your study on thrombo-prophylaxis rates in US medical centers, will you comment on rates of prophylaxis for cancer surgery patients?
Dr. Amin: The overall study included approximately 200,000 medical patients and about 80,000 surgical patients enrolled over more than a 3-year period between 2002 and 2005.39,40 Our goal was to assess rates of prophylaxis and, when it was provided, whether it was appropriate (in terms of type, dosage, and duration) based on the ACCP guidelines. A subanalysis assessed medical cancer patients and surgical cancer patients separately. Medical cancer patients received thromboprophylaxis 56% of the time but received appropriate prophylaxis only 28% of the time. Among surgical cancer patients, appropriate prophylaxis was given only about 24% of the time for those undergoing gynecologic surgery and about 12% of the time for those undergoing neurosurgery. These percentages are consistent with data from other national registries, such as the IMPROVE registry, which documented prophylaxis rates on the order of 45% in medical patients with cancer.41 We also analyzed the data according to individual practitioners and found that medical oncologists use prophylaxis about 25% of the time, which is relatively consistent with other providers, such as internists and surgeons.
So there is a huge opportunity to improve rates of prophylaxis for this group of patients that national guidelines say are at high risk. Why is prophylaxis so underutilized in the cancer population? One factor may be a misperception about the risk of bleeding with anticoagulants. Yet several studies have shown that the rate of bleeding from prophylaxis is extremely low, whether LMWH or UFH is used, so more awareness of actual bleeding risk is needed. Another factor is the obvious focus among internists and oncologists on treating the patient, with perhaps a reduced consideration of prophylaxis and prevention. A third factor may be a concern about thrombocytopenia. However, in our study of prophylaxis rates in US medical centers, we excluded patients who had thrombocytopenia, yet rates of prophylaxis were still low. Nothing in the literature indicates that anticoagulants cannot be used in patients with platelet counts of 50,000 to 150,000 cells/µL or higher, so this suggests that we need to do more education.
Dr. Jaffer: Dr. Brotman, can you tell us more about how clinicians in practice should use prophylaxis in their neurosurgery patients, such as those undergoing craniotomy or spine surgery for cancer? What is the safest and most efficacious way to prevent DVT in these patients?
Dr. Brotman: First, it’s important to recognize that some sort of prophylaxis needs to be used. Neurosurgery patients are at an extremely high risk for thromboembolic events, and such events are often fatal in these patients. Having said that, the jury is still out on whether the prophylaxis in these patients should be compression devices or anticoagulation. This gives physicians some latitude in their decisions. They can decide not to use pharmacologic prophylaxis so long as they use pneumatic compression devices consistently, perhaps even starting during the operation and certainly throughout hospitalization when the patient is immobilized.
Certainly, the concerns about using full-dose anticoagulation in the immediate postoperative setting in neurosurgery patients are valid. Yet these patients are at very high risk for thromboembolic events, and if we take too cautious an approach to prophylaxis in the immediate perioperative setting, more patients are going to have thromboembolic events, at which point management decisions become much more difficult. The risk of intracranial bleeding with anticoagulation to treat a patient who develops a DVT at postoperative day 10 will certainly be higher than it would have been with lower-dose perioperative prophylactic anticoagulation. Plus, if you put in a filter at that point, the outcomes tend to be poor. Therefore, I believe there is some degree of risk that we should be willing to take with regard to perioperative bleeding, even in neurosurgery patients.
Dr. McKean: I’d like to make a point about combination prophylaxis. At many institutions, compression stockings and sequential compression devices are used preoperatively and intraoperatively, and then pharmacologic prophylaxis—for example, twice-daily UFH—is used postoperatively. There is concern that these patients are hypercoagulable, and most clinicians believe that mechanical prophylaxis alone, even with sequential compression devices plus compression stockings, is not aggressive enough in these high-risk patients.
Dr. Jaffer: Dr. Spyropoulos, what is the optimal duration of pharmacologic prophylaxis for cancer surgery patients?
Dr. Spyropoulos: First let’s consider in-hospital prophylaxis. The supportive data for in-hospital prophylaxis are strong, and the duration of therapy used in the major in-hospital prophylaxis trials was 7 to 10 days. With regard to extended prophylaxis, we have at least two moderately sized randomized controlled trials, ENOXACAN II23 and the substudy of FAME,24 that demonstrated that extending prophylaxis with LMWH at doses of 3,400 U once daily (5,000 IU of dalteparin; 40 mg of enoxaparin) reduces VTE risk at postoperative day 30. Also, recent data from the @RISTOS registry show that in cancer surgery patients, especially those having abdominal or pelvic procedures, the leading cause of 30-day mortality was VTE.8 This registry also shows that despite prophylaxis, the rate of symptomatic VTE can be as high as 2%, with the rate of fatal VTE approaching 1%. Thus, in cancer patients undergoing abdominal or pelvic surgery, physicians should strongly consider prophylaxis of up to 30 days’ duration.
Dr. Jaffer: One striking finding from the @RISTOS registry was that 40% of VTE events in these cancer surgery patients occurred after postoperative day 21. This really underscores the need to consider prophylaxis for at least 4 weeks in these patients in real-world practice.
Dr. Brotman: The other striking finding from that registry was that the in-hospital prophylaxis rate was quite high, about 80%, and the rate of extended prophylaxis approached 35%. These are rates that are rarely achieved in clinical practice. Yet despite these high levels of prophylaxis, patients in this registry still had a high incidence of morbidity and mortality from VTE. This suggests that we need to improve our out-of-hospital VTE prevention paradigms.
Dr. Jaffer: Dr. Deitelzweig, oncologists and internists are often unsure about whether their ambulatory cancer patients who are receiving chemotherapy should be on any form of prophylaxis. What is your opinion?
Dr. Deitelzweig: That question comes up regularly because these patients are encountered across many medical specialties. At this point, all of the large organizations, including ASCO and NCCN, are advocating that prophylaxis is not indicated for such patients.
Venous thromboembolism (VTE) is a major complication of cancer, occurring in 4% to 20% of patients,1 and is one of the leading causes of death in cancer patients, although these figures are believed to be underestimates, given the low autopsy rates among cancer patients.2 In hospitalized cancer patients specifically, VTE is the second leading cause of death.3,4 The risk of VTE in cancer patients undergoing surgery is three to five times greater than that in surgical patients without cancer.4 Moreover, cancer patients with symptomatic deep vein thrombosis (DVT) exhibit a high risk of recurrent VTE that may persist for many years after the index event.5
VTE PREVENTION POSES PARTICULAR CHALLENGES IN CANCER PATIENTS
Until recently, data on VTE prevention specific to cancer patients have been sparse. Cancer patients have represented only a small subset (< 20%) of participants in most of the largest clinical trials of VTE prophylaxis. Until the past 2 or 3 years, clinicians largely have had to extrapolate their approach to VTE prophylaxis in cancer patients from data in patients without cancer, bearing in mind that cancer patients are among the populations at highest risk of developing VTE.
High rates of VTE, even with prophylaxis
Further insights have come from the @RISTOS project, a Web-based prospective registry of patients undergoing general, urologic, or gynecologic surgery for cancer at multiple centers in Italy.8 Of the 2,372 patients tracked in this study, 82% received in-hospital VTE prophylaxis and 31% received prophylaxis following discharge. Despite this relatively high frequency of prophylaxis, however, the incidence of clinically overt VTE was 2.1% and the incidence of fatal VTE was 0.8%. Notably, most VTE events occurred after hospital discharge, and VTE was the most common cause of 30-day postoperative death in this registry.
RISK FACTORS: CANCER TYPE AND TREATMENT LOOM LARGE
Both the type and stage of a patient’s cancer are important in assessing the risk of VTE. For men, cancers of the prostate, colon, brain, and lung have been associated with an increased risk of VTE. Among women, cancers of the breast, ovary, and lung have been especially implicated as risk factors for VTE.9,10
The type of cancer therapy also influences VTE risk:
- Surgery. Among patients who undergo cancer-related surgery, the rate of proximal DVT is 10% to 20%, the rate of clinically evident PE is 4% to 10%, and the incidence of fatal PE is 0.2% to 5%.8,11
- Systemic treatments, including chemotherapy and hormone therapy, are also associated with an increased risk of VTE.12–15
- Central venous catheters. Approximately 4% of cancer patients who have central venous catheters placed develop clinically relevant VTE.16,17
In addition to the above risks related to cancer treatments, the following have been identified as risk factors for VTE in surgical oncology patients:
- Age greater than 40 years (risk also increases steeply after age 60 and again after age 75)
- Cancer procoagulants
- Thrombophilia
- Length and complications of cancer surgery (ie, often involving tissue trauma and immobilization)
- Debilitation and slow recovery.
Another risk factor worth noting is perioperative transfusion, as illustrated in a recent study of 14,104 adults undergoing colorectal cancer resection.18 The overall incidence of VTE in these patients was 1.0%, and the risk of death was nearly four times as great in patients who developed VTE as in those who did not. Notably, the need for transfusion was a marker of increased risk of VTE, particularly in women: women who received perioperative transfusions had almost double the risk of developing VTE compared with women who did not receive transfusions (P = .004).
CLINICAL TRIALS OF PROPHYLAXIS IN CANCER SURGERY PATIENTS
LMWH vs UFH for in-hospital prophylaxis
Two large randomized, double-blind trials have compared low-molecular-weight heparin (LMWH) with low-dose unfractionated heparin (UFH) for VTE prophylaxis in surgical patients with cancer—the Enoxaparin and Cancer (ENOXACAN) study19 and the Canadian Colorectal Surgery DVT Prophylaxis Trial.20 Patients in these studies underwent surgery for abdominal or pelvic cancer (mostly colorectal cancer). Both studies compared 40 mg of the LMWH enoxaparin given once daily with 5,000 U of UFH given three times daily for 7 to 10 days postoperatively. Outcome measures were the presence of DVT determined by venography on day 7 to 10 and the incidence of symptomatic VTE. Rates of VTE were statistically equivalent between the two treatment arms in both ENOXACAN (14.7% with LMWH vs 18.2% with UFH) and the Canadian Colorectal Surgery study (9.4% with both therapies), as were rates of major bleeding (4.1% with LMWH vs 2.9% with UFH in ENOXACAN; 2.7% with LMWH vs 1.5% with UFH in the Canadian study).
These findings are consistent with a 2001 meta-analysis by Mismetti et al of all available randomized trials comparing LMWH with placebo or with UFH for VTE prophylaxis in general surgery.21 This analysis found no differences in rates of asymptomatic DVT, clinical PE, clinical thromboembolism, death, major hemorrhage, total hemorrhage, wound hematoma, or need for transfusion between LMWH and UFH in patients undergoing either cancer-related surgery or surgery not related to cancer.
Fondaparinux for in-hospital prophylaxis
Subgroup analysis of the large randomized trial known as PEGASUS22 sheds some light on the efficacy of the factor Xa inhibitor fondaparinux relative to LMWH for thromboprophylaxis in cancer surgery patients. PEGASUS compared fondaparinux 2.5 mg once daily with the LMWH dalteparin 5,000 IU once daily for 5 to 9 days in patients undergoing high-risk abdominal surgery. Among the study’s 1,408 patients undergoing surgery for cancer, rates of VTE were 4.7% in the fondaparinux group compared with 7.7% in the LMWH group, a relative risk reduction of 38.6% with fondaparinux (95% CI, 6.7% to 59.6%). In contrast, in the rest of the PEGASUS population (patients undergoing abdominal surgery for reasons other than cancer), LMWH was nonsignificantly more efficacious at preventing VTE than was fondaparinux. Rates of major bleeding in this cancer subgroup were comparable between the two treatments.
Extended prophylaxis
Two additional randomized trials have evaluated extended prophylaxis with LMWH in surgical cancer patients—ENOXACAN II23 and the Fragmin After Major Abdominal Surgery (FAME) study.24
In ENOXACAN II, patients undergoing surgery for abdominal or pelvic cancer first received 6 to 10 days of prophylaxis with enoxaparin 40 mg once daily and then were randomized in a double-blind fashion to an additional 21 days of enoxaparin or placebo.23 Among 332 patients in the intent-to-treat analysis, the rate of VTE at the end of the double-blind phase was reduced from 12.0% with placebo to 4.8% with extended-duration enoxaparin (P = .02), an effect that was maintained at 3-month follow-up (P = .01). There was no significant difference between the two groups in rates of major bleeding events or any bleeding events.
In FAME, patients received 5,000 IU of dalteparin once daily for 1 week following major abdominal surgery and then were randomized in open-label fashion to either placebo or extended prophylaxis with dalteparin for 3 more weeks; a subanalysis examined outcomes in the 198 FAME participants whose abdominal surgery was for cancer.24 Among these 198 cancer surgery patients, the rate of venography-documented VTE at 4 weeks was reduced from 19.6% with placebo to 8.8% with extended-duration dalteparin, a relative reduction of 55% (P = .03). The rate of proximal DVT was reduced from 10.4% to 2.2% with extended prophylaxis, a relative reduction of 79% (P = .02).
The number needed to treat with extended LMWH prophylaxis to prevent one VTE event was 14 in ENOXACAN II23 and 9 in the FAME subanalysis of cancer surgery patients.24
New systematic review of relevant trials
Leonardi et al recently published a systematic review of 26 randomized controlled trials of DVT prophylaxis in 7,639 cancer surgery patients.25 They found the overall incidence of DVT to be 12.7% in those who received pharmacologic prophylaxis compared with 35.2% in controls. They also found high-dose LMWH therapy (> 3,400 U daily) to be associated with a significantly lower incidence of DVT than low-dose LMWH therapy (≤ 3,400 U daily) (7.9% vs 14.5%, respectively; P < .01). No differences were demonstrated between LMWH and UFH in preventing DVT, DVT location, or bleeding. Bleeding complications requiring discontinuation of pharmacologic prophylaxis occurred in 3% of patients overall.
Implications of HIT
The sequelae of heparin-induced thrombocytopenia (HIT) can have major consequences for cancer surgery patients. The incidence of HIT is markedly lower with LMWH than with UFH, as demonstrated in a nested case-control study by Creekmore et al.26 These researchers also found that the average cost of an admission during which HIT developed was nearly four times as great as the average cost of an admission during which UFH or LMWH was given without development of HIT ($56,364 vs $15,231; P < .001).
EVIDENCE IN SPECIFIC ONCOLOGIC POPULATIONS
Most of the patients in the trials reviewed above underwent abdominal surgery for malignancy. Although studies of VTE prophylaxis in patients undergoing nonabdominal cancer surgery are relatively few, some data are available for a few other specific oncologic populations, as reviewed below.
Surgery for gynecologic cancer
There is a paucity of randomized controlled trials or prospective observational studies on VTE and its prevention in the gynecologic cancer surgery population. Based on small historical studies, the postoperative risk of VTE in this population varies from 12% to 35%.27,28 Twice-daily administration of UFH 5,000 U appears to be ineffective as VTE prophylaxis in this population, but increasing the frequency to three times daily reduces VTE risk by 50% to 60% compared with placebo. Once-daily LMWH is comparable to three-times-daily UFH in efficacy and safety in this population.
A systematic Cochrane review of eight randomized controlled trials in patients undergoing major gynecologic surgery revealed that heparin prophylaxis (either UFH or LMWH) reduces the risk of DVT by 70% compared with no prophylaxis, with an identical risk reduction specifically among women with malignancy (odds ratio, 0.30; 95% CI, 0.10 to 0.89).29 This review found no evidence that anticoagulation reduces the risk of PE following major gynecologic surgery. LMWH and UFH were similar in efficacy for preventing DVT and had a comparable risk of bleeding complications.
Surgery for urologic cancer
The risk of VTE and the benefits of thromboprophylaxis also are poorly studied in patients undergoing surgery for urologic cancer.
The risk of VTE varies with the type of urologic surgery and the method used to diagnose VTE. For instance, patients undergoing radical retropubic prostatectomy have been reported to develop DVT at rates of 1% to 3%, PE at rates of 1% to 3%, and fatal PE at a rate of 0.6%, whereas the incidences of these events are somewhat higher in patients undergoing cystectomy: 8% for DVT, 2% to 4% for PE, and 2% for fatal PE. Radiologic diagnosis of thromboembolism in pelvic surgery patients has yielded higher incidences, with DVT rates of 21% to 51% and PE rates of 11% to 22%.30
Small studies suggest that prophylaxis with either low-dose UFH or LMWH is both effective in reducing VTE risk and safe in urologic cancer surgery patients, although pharmacologic prophylaxis poses a possible increased risk of pelvic hematoma and lymphocele formation in this population.30
Neurosurgery
Most neurosurgical procedures are performed for malignancies. The risk of venography-confirmed VTE in patients undergoing neurosurgery is approximately 30% to 40%.31,32 Likewise, the risks of intracranial or intraspinal hemorrhage in these patients are high. For this reason, mechanical methods of VTE prophylaxis are preferred in these patients. The use of anticoagulant prophylaxis remains controversial in this setting, although more recent data suggest that it might be safer than previously recognized.
GUIDELINES FOR VTE PROPHYLAXIS IN THE CANCER SURGERY PATIENT
American College of Chest Physicians
National Comprehensive Cancer Network
The National Comprehensive Cancer Network (NCCN) recently published clinical practice guidelines on venous thromboembolic disease in cancer patients.35 The defined at-risk population for these guidelines is the adult cancer inpatient with a diagnosis of (or clinical suspicion for) cancer. The guidelines recommend prophylactic anticoagulation (category 1 recommendation) with or without a sequential compression device as initial prophylaxis, unless the patient has a relative contraindication to anticoagulation, in which case mechanical prophylaxis (sequential compression device or graduated compression stockings) is recommended. (A category 1 recommendation indicates “uniform NCCN consensus, based on high-level evidence.”)
The NCCN guidelines include a specific recommended risk-factor assessment, which includes noting the patient’s age (VTE risk increases beginning at age 40 and then steeply again at age 75), any prior VTE, the presence of familial thrombophilia or active cancer, the use of medications associated with increased VTE risk (chemotherapy, exogenous estrogen compounds, and thalidomide or lenalidomide), and a number of other risk factors for VTE as outlined in the prior two articles in this supplement. The NCCN guidelines explicitly call for assessment of modifiable risk factors for VTE (ie, smoking or other tobacco use, obesity, and a low level of activity or lack of exercise) and call for active patient education on these factors.
American Society of Clinical Oncology
Our recommended algorithm
LINGERING CHALLENGE OF UNDERUTILIZATION
Despite this consensus on ways to reduce thromboembolic risk in this population and the clear evidence of the benefit of VTE prophylaxis in patients with cancer, data from several registries confirm a persistently low utilization of prophylaxis in patients with cancer.36–38 The global Fundamental Research in Oncology and Thrombosis (FRONTLINE) study surveyed 3,891 clinicians who treat cancer patients regarding their practices with respect to VTE in those patients.36 The survey found that only 52% of respondents routinely used thromboprophylaxis for their surgical patients with cancer. More striking, however, was the finding that most respondents routinely considered thrombo-prophylaxis in only 5% of their medical oncology patients. These data are echoed by findings of other retrospective medical record reviews in patients undergoing major abdominal or abdominothoracic surgery (in many cases for cancer), with VTE prophylaxis rates ranging from 38% to 75%.37,38
SUMMARY
Patients undergoing surgery for cancer have an increased risk of VTE and fatal PE, even when thromboprophylaxis is used. Nevertheless, prophylaxis with either LMWH or UFH does reduce venographic VTE event rates in these patients. If UFH is chosen for prophylaxis, a three-times-daily regimen should be used in this population. In specific surgical cancer populations, especially those undergoing abdominal surgery, out-of-hospital prophylaxis with once-daily LMWH is warranted. Current registries reveal that compliance with established guidelines for VTE prophylaxis in this population is low.


Dr. Jaffer: Dr. Amin, based on your study on thrombo-prophylaxis rates in US medical centers, will you comment on rates of prophylaxis for cancer surgery patients?
Dr. Amin: The overall study included approximately 200,000 medical patients and about 80,000 surgical patients enrolled over more than a 3-year period between 2002 and 2005.39,40 Our goal was to assess rates of prophylaxis and, when it was provided, whether it was appropriate (in terms of type, dosage, and duration) based on the ACCP guidelines. A subanalysis assessed medical cancer patients and surgical cancer patients separately. Medical cancer patients received thromboprophylaxis 56% of the time but received appropriate prophylaxis only 28% of the time. Among surgical cancer patients, appropriate prophylaxis was given only about 24% of the time for those undergoing gynecologic surgery and about 12% of the time for those undergoing neurosurgery. These percentages are consistent with data from other national registries, such as the IMPROVE registry, which documented prophylaxis rates on the order of 45% in medical patients with cancer.41 We also analyzed the data according to individual practitioners and found that medical oncologists use prophylaxis about 25% of the time, which is relatively consistent with other providers, such as internists and surgeons.
So there is a huge opportunity to improve rates of prophylaxis for this group of patients that national guidelines say are at high risk. Why is prophylaxis so underutilized in the cancer population? One factor may be a misperception about the risk of bleeding with anticoagulants. Yet several studies have shown that the rate of bleeding from prophylaxis is extremely low, whether LMWH or UFH is used, so more awareness of actual bleeding risk is needed. Another factor is the obvious focus among internists and oncologists on treating the patient, with perhaps a reduced consideration of prophylaxis and prevention. A third factor may be a concern about thrombocytopenia. However, in our study of prophylaxis rates in US medical centers, we excluded patients who had thrombocytopenia, yet rates of prophylaxis were still low. Nothing in the literature indicates that anticoagulants cannot be used in patients with platelet counts of 50,000 to 150,000 cells/µL or higher, so this suggests that we need to do more education.
Dr. Jaffer: Dr. Brotman, can you tell us more about how clinicians in practice should use prophylaxis in their neurosurgery patients, such as those undergoing craniotomy or spine surgery for cancer? What is the safest and most efficacious way to prevent DVT in these patients?
Dr. Brotman: First, it’s important to recognize that some sort of prophylaxis needs to be used. Neurosurgery patients are at an extremely high risk for thromboembolic events, and such events are often fatal in these patients. Having said that, the jury is still out on whether the prophylaxis in these patients should be compression devices or anticoagulation. This gives physicians some latitude in their decisions. They can decide not to use pharmacologic prophylaxis so long as they use pneumatic compression devices consistently, perhaps even starting during the operation and certainly throughout hospitalization when the patient is immobilized.
Certainly, the concerns about using full-dose anticoagulation in the immediate postoperative setting in neurosurgery patients are valid. Yet these patients are at very high risk for thromboembolic events, and if we take too cautious an approach to prophylaxis in the immediate perioperative setting, more patients are going to have thromboembolic events, at which point management decisions become much more difficult. The risk of intracranial bleeding with anticoagulation to treat a patient who develops a DVT at postoperative day 10 will certainly be higher than it would have been with lower-dose perioperative prophylactic anticoagulation. Plus, if you put in a filter at that point, the outcomes tend to be poor. Therefore, I believe there is some degree of risk that we should be willing to take with regard to perioperative bleeding, even in neurosurgery patients.
Dr. McKean: I’d like to make a point about combination prophylaxis. At many institutions, compression stockings and sequential compression devices are used preoperatively and intraoperatively, and then pharmacologic prophylaxis—for example, twice-daily UFH—is used postoperatively. There is concern that these patients are hypercoagulable, and most clinicians believe that mechanical prophylaxis alone, even with sequential compression devices plus compression stockings, is not aggressive enough in these high-risk patients.
Dr. Jaffer: Dr. Spyropoulos, what is the optimal duration of pharmacologic prophylaxis for cancer surgery patients?
Dr. Spyropoulos: First let’s consider in-hospital prophylaxis. The supportive data for in-hospital prophylaxis are strong, and the duration of therapy used in the major in-hospital prophylaxis trials was 7 to 10 days. With regard to extended prophylaxis, we have at least two moderately sized randomized controlled trials, ENOXACAN II23 and the substudy of FAME,24 that demonstrated that extending prophylaxis with LMWH at doses of 3,400 U once daily (5,000 IU of dalteparin; 40 mg of enoxaparin) reduces VTE risk at postoperative day 30. Also, recent data from the @RISTOS registry show that in cancer surgery patients, especially those having abdominal or pelvic procedures, the leading cause of 30-day mortality was VTE.8 This registry also shows that despite prophylaxis, the rate of symptomatic VTE can be as high as 2%, with the rate of fatal VTE approaching 1%. Thus, in cancer patients undergoing abdominal or pelvic surgery, physicians should strongly consider prophylaxis of up to 30 days’ duration.
Dr. Jaffer: One striking finding from the @RISTOS registry was that 40% of VTE events in these cancer surgery patients occurred after postoperative day 21. This really underscores the need to consider prophylaxis for at least 4 weeks in these patients in real-world practice.
Dr. Brotman: The other striking finding from that registry was that the in-hospital prophylaxis rate was quite high, about 80%, and the rate of extended prophylaxis approached 35%. These are rates that are rarely achieved in clinical practice. Yet despite these high levels of prophylaxis, patients in this registry still had a high incidence of morbidity and mortality from VTE. This suggests that we need to improve our out-of-hospital VTE prevention paradigms.
Dr. Jaffer: Dr. Deitelzweig, oncologists and internists are often unsure about whether their ambulatory cancer patients who are receiving chemotherapy should be on any form of prophylaxis. What is your opinion?
Dr. Deitelzweig: That question comes up regularly because these patients are encountered across many medical specialties. At this point, all of the large organizations, including ASCO and NCCN, are advocating that prophylaxis is not indicated for such patients.
- Lyman GH, Khorana AA, Falanga A, et al. American Society of Clinical Oncology guideline: recommendations for venous thromboembolism prophylaxis and treatment in patients with cancer. J Clin Oncol 2007; 25:5490–5505.
- Khorana AA, Francis CW, Culakova E, et al. Thromboembolism is a leading cause of death in cancer patients receiving outpatient chemotherapy. J Thromb Haemost 2007; 5:632–634.
- Ambrus JL, Ambrus CM, Mink IB, Pickren JW. Causes of death in cancer patients. J Med 1975; 6:61–64.
- Donati MB. Cancer and thrombosis. Haemostasis 1994; 24:128–131.
- Prandoni P, Lensing AW, Cogo A, et al. The long-term clinical course of acute deep venous thrombosis. Ann Intern Med 1996; 125:1–7.
- Haas S, Wolf H, Kakkar AK, Fareed J, Encke A. Prevention of fatal pulmonary embolism and mortality in surgical patients: a randomized double-blind comparison of LMWH with unfractionated heparin. Thromb Haemost 2005; 94:814–819.
- Kakkar AK, Haas S, Wolf H, Encke A. Evaluation of perioperative fatal pulmonary embolism and death in cancer surgical patients: the MC-4 cancer substudy. Thromb Haemost 2005; 94:867–871.
- Agnelli G, Bolis G, Capussotti L, et al. A clinical outcome-based prospective study on venous thromboembolism after cancer surgery: the @RISTOS project. Ann Surg 2006; 243:89–95.
- Levitan N, Dowlati A, Remick SC, et al. Rates of initial and recurrent thromboembolic disease among patients with malignancy versus those without malignancy: risk analysis using Medicare claims data. Medicine (Baltimore) 1999; 78:285–291.
- Levine M, Gent M, Hirsh J, et al. A comparison of low-molecular-weight heparin administered primarily at home with unfractionated heparin administered in the hospital for proximal deep-vein thrombosis. N Engl J Med 1996; 334:677–681.
- Bergqvist D. Risk of venous thromboembolism in patients undergoing cancer surgery and options for thromboprophylaxis. J Surg Oncol 2007; 95:167–174.
- Heit JA, Silverstein MD, Mohr DN, et al. Risk factors for deep vein thrombosis and pulmonary embolism: a population-based case-control study. Arch Intern Med 2000; 160:809–815.
- Sallah S, Wan JY, Nguyen NP. Venous thrombosis in patients with solid tumors: determination of frequency and characteristics. Thromb Haemost 2002; 87:575–579.
- Kröger K, Weiland D, Ose C, et al. Risk factors for venous thromboembolic events in cancer patients. Ann Oncol 2006; 17:297–303.
- Blom JW, Vanderschoot JP, Oostindiër MJ, et al. Incidence of venous thrombosis in a large cohort of 66,329 cancer patients: results of a record linkage study. J Thromb Haemost 2006; 4:529–535.
- Couban S, Simpson DR, Barnett MJ, et al. A randomized multicenter comparison of bone marrow and peripheral blood in recipients of matched sibling allogeneic transplants for myeloid malignancies. Blood 2002; 100:1525–1531.
- Walshe LJ, Malak SF, Eagan J, Sepkowitz KA. Complication rates among cancer patients with peripherally inserted central catheters. J Clin Oncol 2002; 20:3276–3281.
- Nilsson KR, Berenholtz SM, Garrett-Mayer E, et al. Association between venous thromboembolism and perioperative allogeneic transfusion. Arch Surg 2007; 142:126–133.
- Efficacy and safety of enoxaparin versus unfractionated heparin for prevention of deep vein thrombosis in elective cancer surgery: a double-blind randomized multicentre trial with venographic assessment. ENOXACAN Study Group. Br J Surg 1997; 84:1099–1103.
- McLeod RS, Geerts WH, Sniderman KW, et al. Subcutaneous heparin versus low-molecular-weight heparin as thromboprophylaxis in patients undergoing colorectal surgery: results of the Canadian Colorectal DVT Prophylaxis Trial: a randomized, double-blind trial. Ann Surg 2001; 233:438–444.
- Mismetti P, Laporte S, Darmon JY, Buchmüller A, Decousus H. Meta-analysis of low molecular weight heparin in the prevention of venous thromboembolism in general surgery. Br J Surg 2001; 88:913–930.
- Agnelli G, Bergqvist D, Cohen AT, et al, on behalf of the PEGASUS investigators. Randomized clinical trial of postoperative fondaparinux versus perioperative dalteparin for prevention of venous thromboembolism in high-risk abdominal surgery. Br J Surg 2005; 92:1212–1220.
- Bergqvist D, Agnelli G, Cohen AT, et al. Duration of prophylaxis against venous thromboembolism with enoxaparin after surgery for cancer. N Engl J Med 2002; 346:975–980.
- Rasmussen MS, Wille-Jorgensen P, Jorgensen LN, et al. Prolonged thromboprophylaxis with low molecular weight heparin (dalteparin) following major abdominal surgery for malignancy [abstract 186]. Blood 2003; 102:56a.
- Leonardi MJ, McGory ML, Ko CY. A systematic review of deep venous thrombosis prophylaxis in cancer patients: implications for improving quality. Ann Surg Oncol 2007; 14:929–936.
- Creekmore FM, Oderda GM, Pendleton RC, Brixner DI. Incidence and economic implications of heparin-induced thrombocytopenia in medical patients receiving prophylaxis for venous thromboembolism. Pharmacotherapy 2006; 26:1438–1445.
- Walsh JJ, Bonnar J, Wright FW. A study of pulmonary embolism and deep leg vein thrombosis after major gynaecological surgery using labeled fibrinogen-phlebography and lung scanning. J Obstet Gynaecol Br Commonw 1974; 81:311–316.
- Clarke-Pearson DL, Synan IS, Coleman RE, et al. The natural history of postoperative venous thromboemboli in gynecologic oncology: a prospective study of 382 patients. Am J Obstet Gynecol 1984; 148:1051–1054.
- Oates-Whitehead RM, D’Angelo A, Mol B. Anticoagulant and aspirin prophylaxis for preventing thromboembolism after major gynaecological surgery. Cochrane Database Syst Rev 2003; (4):CD003679.
- Kibel AS, Loughlin KR. Pathogenesis and prophylaxis of postoperative thromboembolic disease in urological pelvic surgery. J Urol 1995; 153:1763–1774.
- Agnelli G, Piovella F, Buoncristiani P, et al. Enoxaparin plus compression stockings compared with compression stockings alone in the prevention of venous thromboembolism after elective neurosurgery. N Engl J Med 1998; 339:80–85.
- Semrad TJ, O’Donnell R, Wun T, et al. Epidemiology of venous thromboembolism in 9489 patients with malignant glioma. J Neurosurg 2007; 106:601–608.
- Iorio A, Agnelli G. Low-molecular-weight and unfractionated heparin for prevention of venous thromboembolism in neurosurgery: a meta-analysis. Arch Intern Med 2000; 160:2327–2332.
- Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 2004; 126(3 Suppl):338S–400S.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology. Venous thromboembolic disease. V.1.2007. http://www.nccn.org/professionals/physician_gls/PDF/vte.pdf. Accessed December 5, 2007.
- Kakkar AK, Levine M, Pinedo HM, Wolff R, Wong J. Venous thrombosis in cancer patients: insights from the FRONTLINE survey. Oncologist 2003; 8:381–388.
- Stratton MA, Anderson FA, Bussey HI, et al. Prevention of venous thromboembolism: adherence to the 1995 American College of Chest Physicians consensus guidelines for surgical patients. Arch Intern Med 2000; 160:334–340.
- Bratzler DW, Raskob GE, Murray CK, Bumpus LJ, Piatt DS. Underuse of venous thromboembolism prophylaxis for general surgery patients: physician practices in the community hospital setting. Arch Intern Med 1998; 158:1909–1912.
- Amin A, Stemkowski S, Lin J, et al. Thromboprophylaxis rates in US medical centers: success or failure? J Thromb Haemost 2007; 5:1610–1616.
- Amin A, Stemkowski S, Lin J, Yang G. Thromboprophylaxis in US hospitals: adherence to the 6th American College of Chest Physicians’ recommendations for at-risk medical and surgical patients. Abstract presented at: 41st Midyear Clinical Meeting of the American Society of Health-System Pharmacists; December 3–7, 2006; Anaheim, CA.
- Tapson VF, Decousus H, Pini M, et al. Venous thromboembolism prophylaxis in acutely ill hospitalized medical patients: findings from the International Medical Prevention Registry on Venous Thromboembolism. Chest 2007; 132:936–945.
- Clarke-Pearson DL, Synan IS, Dodge R, et al. A randomized trial of low-dose heparin and intermittent pneumatic calf compression for the prevention of deep venous thrombosis after gynecologic oncology surgery. Am J Obstet Gynecol 1993; 168:1146–1154.
- Einstein MH, Pritts EA, Hartenbach EM. Venous thromboembolism prevention in gynecologic cancer surgery: a systematic review. Gynecol Oncol 2007; 105:813–819.
- Clarke-Pearson DL, Synan IS, Hinshaw WM, Coleman RE, Creasman WT. Prevention of postoperative venous thromboembolism by external pneumatic calf compression in patients with gynecologic malignancy. Obstet Gynecol 1984; 63:92–98.
- Ruff RL, Posner JB. Incidence and treatment of peripheral venous thrombosis in patients with glioma. Ann Neurol 1983; 13:334–336.
- Levin JM, Schiff D, Loeffler JS, Fine HA, Black PM, Wen PY. Complications of therapy for venous thromboembolic disease in patients with brain tumors. Neurology 1993; 43:1111–1114.
- Lyman GH, Khorana AA, Falanga A, et al. American Society of Clinical Oncology guideline: recommendations for venous thromboembolism prophylaxis and treatment in patients with cancer. J Clin Oncol 2007; 25:5490–5505.
- Khorana AA, Francis CW, Culakova E, et al. Thromboembolism is a leading cause of death in cancer patients receiving outpatient chemotherapy. J Thromb Haemost 2007; 5:632–634.
- Ambrus JL, Ambrus CM, Mink IB, Pickren JW. Causes of death in cancer patients. J Med 1975; 6:61–64.
- Donati MB. Cancer and thrombosis. Haemostasis 1994; 24:128–131.
- Prandoni P, Lensing AW, Cogo A, et al. The long-term clinical course of acute deep venous thrombosis. Ann Intern Med 1996; 125:1–7.
- Haas S, Wolf H, Kakkar AK, Fareed J, Encke A. Prevention of fatal pulmonary embolism and mortality in surgical patients: a randomized double-blind comparison of LMWH with unfractionated heparin. Thromb Haemost 2005; 94:814–819.
- Kakkar AK, Haas S, Wolf H, Encke A. Evaluation of perioperative fatal pulmonary embolism and death in cancer surgical patients: the MC-4 cancer substudy. Thromb Haemost 2005; 94:867–871.
- Agnelli G, Bolis G, Capussotti L, et al. A clinical outcome-based prospective study on venous thromboembolism after cancer surgery: the @RISTOS project. Ann Surg 2006; 243:89–95.
- Levitan N, Dowlati A, Remick SC, et al. Rates of initial and recurrent thromboembolic disease among patients with malignancy versus those without malignancy: risk analysis using Medicare claims data. Medicine (Baltimore) 1999; 78:285–291.
- Levine M, Gent M, Hirsh J, et al. A comparison of low-molecular-weight heparin administered primarily at home with unfractionated heparin administered in the hospital for proximal deep-vein thrombosis. N Engl J Med 1996; 334:677–681.
- Bergqvist D. Risk of venous thromboembolism in patients undergoing cancer surgery and options for thromboprophylaxis. J Surg Oncol 2007; 95:167–174.
- Heit JA, Silverstein MD, Mohr DN, et al. Risk factors for deep vein thrombosis and pulmonary embolism: a population-based case-control study. Arch Intern Med 2000; 160:809–815.
- Sallah S, Wan JY, Nguyen NP. Venous thrombosis in patients with solid tumors: determination of frequency and characteristics. Thromb Haemost 2002; 87:575–579.
- Kröger K, Weiland D, Ose C, et al. Risk factors for venous thromboembolic events in cancer patients. Ann Oncol 2006; 17:297–303.
- Blom JW, Vanderschoot JP, Oostindiër MJ, et al. Incidence of venous thrombosis in a large cohort of 66,329 cancer patients: results of a record linkage study. J Thromb Haemost 2006; 4:529–535.
- Couban S, Simpson DR, Barnett MJ, et al. A randomized multicenter comparison of bone marrow and peripheral blood in recipients of matched sibling allogeneic transplants for myeloid malignancies. Blood 2002; 100:1525–1531.
- Walshe LJ, Malak SF, Eagan J, Sepkowitz KA. Complication rates among cancer patients with peripherally inserted central catheters. J Clin Oncol 2002; 20:3276–3281.
- Nilsson KR, Berenholtz SM, Garrett-Mayer E, et al. Association between venous thromboembolism and perioperative allogeneic transfusion. Arch Surg 2007; 142:126–133.
- Efficacy and safety of enoxaparin versus unfractionated heparin for prevention of deep vein thrombosis in elective cancer surgery: a double-blind randomized multicentre trial with venographic assessment. ENOXACAN Study Group. Br J Surg 1997; 84:1099–1103.
- McLeod RS, Geerts WH, Sniderman KW, et al. Subcutaneous heparin versus low-molecular-weight heparin as thromboprophylaxis in patients undergoing colorectal surgery: results of the Canadian Colorectal DVT Prophylaxis Trial: a randomized, double-blind trial. Ann Surg 2001; 233:438–444.
- Mismetti P, Laporte S, Darmon JY, Buchmüller A, Decousus H. Meta-analysis of low molecular weight heparin in the prevention of venous thromboembolism in general surgery. Br J Surg 2001; 88:913–930.
- Agnelli G, Bergqvist D, Cohen AT, et al, on behalf of the PEGASUS investigators. Randomized clinical trial of postoperative fondaparinux versus perioperative dalteparin for prevention of venous thromboembolism in high-risk abdominal surgery. Br J Surg 2005; 92:1212–1220.
- Bergqvist D, Agnelli G, Cohen AT, et al. Duration of prophylaxis against venous thromboembolism with enoxaparin after surgery for cancer. N Engl J Med 2002; 346:975–980.
- Rasmussen MS, Wille-Jorgensen P, Jorgensen LN, et al. Prolonged thromboprophylaxis with low molecular weight heparin (dalteparin) following major abdominal surgery for malignancy [abstract 186]. Blood 2003; 102:56a.
- Leonardi MJ, McGory ML, Ko CY. A systematic review of deep venous thrombosis prophylaxis in cancer patients: implications for improving quality. Ann Surg Oncol 2007; 14:929–936.
- Creekmore FM, Oderda GM, Pendleton RC, Brixner DI. Incidence and economic implications of heparin-induced thrombocytopenia in medical patients receiving prophylaxis for venous thromboembolism. Pharmacotherapy 2006; 26:1438–1445.
- Walsh JJ, Bonnar J, Wright FW. A study of pulmonary embolism and deep leg vein thrombosis after major gynaecological surgery using labeled fibrinogen-phlebography and lung scanning. J Obstet Gynaecol Br Commonw 1974; 81:311–316.
- Clarke-Pearson DL, Synan IS, Coleman RE, et al. The natural history of postoperative venous thromboemboli in gynecologic oncology: a prospective study of 382 patients. Am J Obstet Gynecol 1984; 148:1051–1054.
- Oates-Whitehead RM, D’Angelo A, Mol B. Anticoagulant and aspirin prophylaxis for preventing thromboembolism after major gynaecological surgery. Cochrane Database Syst Rev 2003; (4):CD003679.
- Kibel AS, Loughlin KR. Pathogenesis and prophylaxis of postoperative thromboembolic disease in urological pelvic surgery. J Urol 1995; 153:1763–1774.
- Agnelli G, Piovella F, Buoncristiani P, et al. Enoxaparin plus compression stockings compared with compression stockings alone in the prevention of venous thromboembolism after elective neurosurgery. N Engl J Med 1998; 339:80–85.
- Semrad TJ, O’Donnell R, Wun T, et al. Epidemiology of venous thromboembolism in 9489 patients with malignant glioma. J Neurosurg 2007; 106:601–608.
- Iorio A, Agnelli G. Low-molecular-weight and unfractionated heparin for prevention of venous thromboembolism in neurosurgery: a meta-analysis. Arch Intern Med 2000; 160:2327–2332.
- Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 2004; 126(3 Suppl):338S–400S.
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology. Venous thromboembolic disease. V.1.2007. http://www.nccn.org/professionals/physician_gls/PDF/vte.pdf. Accessed December 5, 2007.
- Kakkar AK, Levine M, Pinedo HM, Wolff R, Wong J. Venous thrombosis in cancer patients: insights from the FRONTLINE survey. Oncologist 2003; 8:381–388.
- Stratton MA, Anderson FA, Bussey HI, et al. Prevention of venous thromboembolism: adherence to the 1995 American College of Chest Physicians consensus guidelines for surgical patients. Arch Intern Med 2000; 160:334–340.
- Bratzler DW, Raskob GE, Murray CK, Bumpus LJ, Piatt DS. Underuse of venous thromboembolism prophylaxis for general surgery patients: physician practices in the community hospital setting. Arch Intern Med 1998; 158:1909–1912.
- Amin A, Stemkowski S, Lin J, et al. Thromboprophylaxis rates in US medical centers: success or failure? J Thromb Haemost 2007; 5:1610–1616.
- Amin A, Stemkowski S, Lin J, Yang G. Thromboprophylaxis in US hospitals: adherence to the 6th American College of Chest Physicians’ recommendations for at-risk medical and surgical patients. Abstract presented at: 41st Midyear Clinical Meeting of the American Society of Health-System Pharmacists; December 3–7, 2006; Anaheim, CA.
- Tapson VF, Decousus H, Pini M, et al. Venous thromboembolism prophylaxis in acutely ill hospitalized medical patients: findings from the International Medical Prevention Registry on Venous Thromboembolism. Chest 2007; 132:936–945.
- Clarke-Pearson DL, Synan IS, Dodge R, et al. A randomized trial of low-dose heparin and intermittent pneumatic calf compression for the prevention of deep venous thrombosis after gynecologic oncology surgery. Am J Obstet Gynecol 1993; 168:1146–1154.
- Einstein MH, Pritts EA, Hartenbach EM. Venous thromboembolism prevention in gynecologic cancer surgery: a systematic review. Gynecol Oncol 2007; 105:813–819.
- Clarke-Pearson DL, Synan IS, Hinshaw WM, Coleman RE, Creasman WT. Prevention of postoperative venous thromboembolism by external pneumatic calf compression in patients with gynecologic malignancy. Obstet Gynecol 1984; 63:92–98.
- Ruff RL, Posner JB. Incidence and treatment of peripheral venous thrombosis in patients with glioma. Ann Neurol 1983; 13:334–336.
- Levin JM, Schiff D, Loeffler JS, Fine HA, Black PM, Wen PY. Complications of therapy for venous thromboembolic disease in patients with brain tumors. Neurology 1993; 43:1111–1114.
Prevention of venous thromboembolism in the orthopedic surgery patient
Nearly half of orthopedic surgery patients do not receive appropriate prophylaxis for venous thromboembolism (VTE), as defined by American College of Chest Physicians (ACCP) consensus guidelines, according to a recent analysis of a nationwide database of hospital admissions.1 Even in teaching hospitals, compliance with consensus guidelines for thromboprophylaxis is suboptimal. In a study of adherence to the ACCP guidelines for VTE prevention among 1,907 surgical patients at 10 teaching hospitals, only 45.2% of hip fracture patients received optimal VTE prophylaxis.2 Rates of optimal prophylaxis were higher among patients undergoing hip arthroplasty and knee arthroplasty—84.3% and 75.9%, respectively—but were still in need of improvement.2
GROWING INTEREST IN POSTOPERATIVE VTE PROPHYLAXIS AS A QUALITY INDICATOR
As noted in the introductory article in this supplement, the Joint Commission on Accreditation of Healthcare Organizations has taken notice of these shortcomings and has proposed national consensus standards for VTE prevention and treatment.3 Among its proposed standards are two related to risk assessment and prophylaxis: whether risk assessment/prophylaxis is ordered within 24 hours of hospital admission and within 24 hours of transfer to the intensive care unit.
Other quality-monitoring initiatives are focused specifically on VTE in the surgical population. The Surgical Care Improvement Project (SCIP) has approved two quality measures with respect to VTE prevention: (1) the proportion of surgical patients for whom recommended VTE prophylaxis is ordered, and (2) the proportion of patients who receive appropriate VTE prophylaxis (based on ACCP guideline recommendations) within 24 hours before or after surgery.4
In the future, two other VTE-related quality measures from SCIP may be implemented by the Centers for Medicare and Medicaid Services: (1) how often intra- or postoperative pulmonary embolism (PE) is diagnosed during the index hospitalization and within 30 days of surgery, and (2) how often intra- or postoperative deep vein thrombosis (DVT) is diagnosed during the index hospitalization and within 30 days of surgery.5
VTE RISK IN ORTHOPEDIC SURGERY
Surgical patients can be stratified into four VTE risk levels—low, moderate, high, and highest—based on age, surgery type, surgery duration, duration of immobilization, and other risk factors.6 For patients undergoing orthopedic surgery, these levels may be defined according to the following patient and surgical characteristics:
- Low risk—surgery duration of less than 30 minutes, age less than 40 years, repair of small fractures
- Moderate risk—age of 40 to 60 years, arthroscopy or repair of lower leg fractures, postoperative plaster cast
- High risk—age greater than 60 years, or age 40 to 60 years with additional VTE risk factors, or immobilization for greater than 4 days
- Highest risk—hip or knee arthroplasty, hip fracture repair, repair of open lower leg fractures, major trauma or spinal cord injury, or multiple risk factors for VTE (age > 40 years, prior VTE, cancer, or hypercoagulable state).
For patients in the low-risk category, no specific prophylaxis is indicated beyond early and aggressive ambulation.6 For those in all other risk categories, prophylaxis with pharmacologic anticoagulant agents and/or mechanical devices is indicated, as reviewed below.
All major orthopedic procedures confer highest risk level
Notably, the “highest risk” category includes any patient undergoing hip or knee arthroplasty or hip fracture repair. Among orthopedic surgery patients in this highest-risk category, rates of VTE events in the absence of prophylaxis are as follows:6
- Calf DVT, 40% to 80%
- Proximal DVT, 10% to 20%
- Clinical PE, 4% to 10%
- Fatal PE, 0.2% to 5%.
Hip replacement poses greater risk than knee replacement
Within this overall highest-risk category, thromboembolic risk in the absence of prophylaxis differs among procedures. Although patients undergoing hip replacement and those undergoing knee replacement have similar rates of DVT of any type,6,7 hip replacement is associated with higher rates of the more clinically important events, specifically proximal DVT and PE. In the absence of prophylaxis, proximal DVT occurs in 23% to 36% of hip replacement patients as opposed to 9% to 20% of knee replacement patients; similarly, PE occurs in 0.7% to 30% of hip replacement patients as compared with 1.8% to 7.0% of knee replacement patients.6,7
What about bleeding risk?
For many orthopedic surgeons, the risk of bleeding as a result of anticoagulant prophylaxis of VTE looms larger than the risk of VTE itself. This is likely because bleeding, when it does occur, is likely to occur more acutely than VTE does and may directly compromise the result of the operation. For this reason, orthopedic surgeons may be more likely to actually witness bleeding events than VTE events (especially fatal PEs) while their patients are still under their care, leading to a misperception of the relative risks of anticoagulation-related bleeding and thromboembolism.
In reality, rates of major bleeding with pharmacologic prophylaxis of VTE are a tiny fraction of the above-listed rates of VTE events in the absence of prophylaxis in patients undergoing major orthopedic surgery. Reported 30-day rates of major bleeding in patients receiving VTE prophylaxis with heparins range from 0.2% to 1.7%; these rates barely differ from the rates among placebo recipients in the same VTE prophylaxis trials, which range from 0.2% to 1.5%.8,9 Additionally, within the continuum of risk of major bleeding from various medical interventions, VTE prophylaxis with heparins is one of the lowest-risk interventions, posing far less risk than, for example, the use of warfarin in ischemic stroke patients or in patients older than 75 years.
PHARMACOLOGIC OPTIONS FOR VTE PROPHYLAXIS IN ORTHOPEDIC SURGERY
As reviewed in the introductory article of this supplement, the arsenal of anticoagulants for use in VTE prophylaxis includes low-dose unfractionated heparin (UFH), low-molecular-weight heparin (LMWH) agents such as dalteparin and enoxaparin, and the factor Xa inhibitor fondaparinux. A few additional comments about these and other anticoagulant options is warranted in the specific context of orthopedic surgery.
Fondaparinux. Because most of its formal US indications are for use as VTE prophylaxis in major orthopedic surgery—including hip replacement, knee replacement, and hip fracture repair—fondaparinux has been studied more widely in orthopedic surgery patients than in the other populations reviewed earlier in this supplement. Nevertheless, its use even in these settings has remained somewhat limited. This may be because of concerns over possible increased bleeding risk relative to some other anticoagulants. Because of bleeding risk, fondaparinux is contraindicated in patients who weigh less than 50 kg, and its package insert recommends caution when it is used in the elderly due to an increased risk of bleeding in patients aged 65 or older. Additionally, the Pentasaccharide in Major Knee Surgery (PENTAMAKS) study found fondaparinux to be associated with a significantly higher incidence of major bleeding compared with enoxaparin (2.1% vs 0.2%; P = .006) in major knee surgery, although it was superior to enoxaparin in preventing VTE.10 Other possible reasons for slow adoption of fondaparinux include its long half-life, which results in a sustained antithrombotic effect, its lack of easy reversibility, and a contraindication in patients with renal insufficiency.11
Limited role for UFH. Low-dose UFH has a more limited role in orthopedic surgery than in other settings requiring VTE prophylaxis, as current ACCP guidelines for VTE prevention recognize it only as a possible option in hip fracture surgery and state that it is not to be considered as sole prophylaxis in patients undergoing hip or knee replacement.6
Warfarin. Although not indicated for use in other VTE prophylaxis settings, the vitamin K antagonist warfarin is recommended as an option for all three major orthopedic surgery indications—knee replacement, hip replacement, and hip fracture repair.6
The key to effective prophylaxis with warfarin is achieving the appropriate intensity of anticoagulation. In two separate analyses, Hylek et al demonstrated a balance between safety and efficacy with warfarin therapy targeted to an international normalized ratio (INR) of 2.0 to 3.0.12,13 An INR greater than 4.0 greatly increased the risk of intracranial hemorrhage, whereas thrombosis was not effectively prevented with an INR less than 2.0.12,13 This latter point should be stressed to orthopedic surgeons, who sometimes aim for INR values below 2.0.
Although anticoagulation clinics are superior to usual care at maintaining the INR within the window of 2.0 to 3.0, only about one-third of patients nationally who take warfarin receive care in such clinics.14 Even with optimal care in anticoagulation clinics, some patients will still receive subtherapeutic or supertherapeutic levels of warfarin, which is one of this agent’s limitations.
Aspirin not recommended as sole agent. Although aspirin is still used as thromboprophylaxis in orthopedic surgery patients, current ACCP guidelines recommend against its use as the sole means of VTE prophylaxis in any patient group.6 The limitations of the evidence for aspirin in this setting are illustrated by the Pulmonary Embolism Prevention study, a multicenter randomized trial in patients undergoing hip fracture (n = 13,356) or hip/knee replacement (n = 4,088).15 Patients received aspirin 160 mg/day or placebo for 5 weeks, starting preoperatively, and were evaluated for outcomes at day 35. Among the hip fracture patients, the rate of symptomatic DVT was lower in the aspirin group than in the placebo group (1.0% vs 1.5%; P = .03), as was the rate of PE (0.7% vs 1.2%, respectively; P = .002), but there was no significant difference in outcomes between the groups among the patients undergoing hip or knee replacement. Notably, 40% of patients in the study also received UFH or LMWH. Further confounding the results, some patients received nonpharmacologic VTE prophylaxis modalities, and others received nonsteroidal anti-inflammatory drugs other than aspirin.
Heparin-induced thrombocytopenia. As noted earlier in this supplement, the incidence of heparin-induced thrombocytopenia (HIT) is markedly higher in patients who receive UFH than in those who receive LMWH. This difference in frequency, which constitutes about a sixfold to eightfold differential, is due to the relationship between standard heparin and platelet factor IV, which can induce formation of IgG antibodies.16 A 50% or greater reduction in platelet count in heparin recipients should prompt consideration of HIT.
Oral direct thrombin inhibitors. Although the oral direct thrombin inhibitor ximelagatran was rejected for approval by the US Food and Drug Administration (FDA) and recently withdrawn from the market worldwide as a result of hepatic risks, other oral direct thrombin inhibitors are in phase 3 studies for use in orthopedic surgery and may be commercially available options for postoperative VTE prophylaxis before long.
GUIDELINES FOR VTE PROPHYLAXIS IN ORTHOPEDIC SURGERY
The ACCP guidelines referred to throughout this article are widely recognized as a practice standard for VTE prevention and treatment, and have been regularly updated throughout recent decades. The most recent version, issued in 2004, is formally known as the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy.6 Key orthopedic surgery-related recommendations and notable changes from the previous version of the guidelines, issued in 2001, are outlined below, along with pertinent supportive or illustrative studies.
Hip replacement surgery
In a change from the previous guidelines, the Seventh ACCP Conference recommends extended prophylaxis, for up to 28 to 35 days after surgery, for patients undergoing hip replacement or hip fracture surgery. For hip replacement surgery, this is a Grade 1A recommendation for prophylaxis with either LMWH or warfarin and a Grade 1C+ recommendation (“no RCTs but strong RCT results can be unequivocally extrapolated, or overwhelming evidence from observational studies”) for prophylaxis with fondaparinux.6
Knee replacement surgery
The same three anticoagulant options that received Grade 1A recommendations for patients undergoing total hip replacement—LMWH, fondaparinux, and adjusted-dose warfarin—are also given Grade 1A recommendations as routine thromboprophylaxis in patients undergoing elective knee replacement (see Table 1 for dosing). In addition, optimal use of intermittent pneumatic compression devices is recommended as an alternative option to anticoagulant prophylaxis in these patients (Grade 1B, indicating a “strong recommendation” based on RCTs with important limitations). Use of UFH as the sole agent for prophylaxis is recommended against.6
For both hip and knee replacement surgery, the Seventh ACCP Conference does not endorse superiority of any one of its three recommended prophylaxis options—LMWH, fondaparinux, and adjusted-dose warfarin—over the other two. However, at least four large randomized trials have directly compared LMWH and adjusted-dose warfarin in the setting of arthroplasty—two in total hip replacement surgery19,20 and two in total knee replacement surgery.21,22 Each of these four studies found LMWH to be significantly more effective than warfarin in preventing VTE. In three of the four trials, there was no significant difference between the therapies in rates of major bleeding.19,21,22 In the remaining trial, which was conducted in hip replacement surgery patients and compared postoperative warfarin with dalteparin initiated either immediately before or early after surgery, patients who received preoperative dalteparin initiation (but not those who received postoperative dalteparin initiation) had an increased rate of major bleeding compared with warfarin recipients (P = .01).20
Hip fracture surgery
The supportive evidence for anticoagulant prophylaxis in hip fracture surgery is less robust than that in hip and knee replacement surgery. As a result, only fondaparinux has a Grade 1A recommendation as routine prophylaxis in patients undergoing hip fracture surgery. Options with less definitive recommendations are LMWH (Grade 1C+), low-dose UFH (Grade 1B), and adjusted-dose warfarin (Grade 2B, indicating a “weak recommendation” based on RCTs with important limitations) (see Table 1 for dosing of all agents).6
These differing recommendations are supported by the double-blind Pentasaccharide in Hip Fracture Surgery Study (PENTHIFRA) of 1,711 consecutive patients undergoing surgery for hip fracture repair.23 Patients were randomized to at least 5 days of fondaparinux 2.5 mg once daily, initiated postoperatively, or enoxaparin 40 mg once daily, initiated preoperatively. The incidence of DVT or PE by postoperative day 11 was 8.3% in the fondaparinux arm versus 19.1% in the enoxaparin arm, a statistically significant difference (P < .001) in favor of fondaparinux. There were no differences between the groups in rates of death or clinically relevant bleeding.
As noted above, the newly added recommendation in the Seventh ACCP Conference for extended prophylaxis, for up to 28 to 35 days after surgery, applies to patients undergoing hip fracture surgery as well as those undergoing hip replacement surgery. In the setting of hip fracture repair, extended prophylaxis is a Grade 1A recommendation with the use of fondaparinux and a Grade 1C+ recommendation with the use of either LMWH or adjusted-dose warfarin.6
Lower extremity fractures and trauma
Although lower extremity fractures are very common, the risk of DVT has been poorly studied in this setting. For patients with isolated lower extremity fractures, the Seventh ACCP Conference recommends that clinicians not use thromboprophylaxis routinely (Grade 2A, indicating an “intermediate-strength recommendation” based on RCTs without important limitations).6
Trauma patients, in contrast, are well recognized as being at very high risk for DVT and PE. The Seventh ACCP Conference gives a Grade 1A recommendation to thromboprophylaxis for all trauma patients who have at least one risk factor for VTE. LMWH is recommended (Grade 1A) as the agent of choice for this purpose, provided there are no contraindications to its use, and should be administered as soon as safely possible. Mechanical modalities are reserved for trauma patients with active bleeding or high risk for hemorrhage (Grade 1B). The guidelines recommend against use of inferior vena cava (IVC) filters as primary thromboprophylaxis in trauma patients (Grade 1C, indicating an “intermediate-strength recommendation” based on observational studies).6
Use of ultrasonography
Duplex ultrasonographic screening is recommended in orthopedic trauma patients who are at high risk for VTE and have received suboptimal or no prophylaxis (Grade 1C). In contrast, the Seventh ACCP Conference recommends against routine use of duplex ultrasonography to screen for VTE at hospital discharge in asymptomatic patients following major orthopedic surgery (Grade 1A).6
Knee arthroscopy
Arthroscopic knee procedures are increasing in frequency and raise the specter of a potential role for thromboprophylaxis. However, the clinical diagnosis of DVT is unreliable, and even diagnosis by ultrasonography is unreliable following knee arthroscopy, as interpreting scans of veins below the knee is challenging in this setting.24
The Seventh ACCP Conference recommends that clinicians not use routine thromboprophylaxis, other than early mobilization, for patients who undergo knee arthroscopy (Grade 2B). However, for arthroscopy patients who have inherent risk factors for VTE or who undergo a prolonged or complicated arthroscopy procedure, thromboprophylaxis with LMWH is suggested (Grade 2B).6
RECOMMENDED APPROACH TO VTE PROPHYLAXIS IN ORTHOPEDIC SURGERY
Drawing on the ACCP guidelines and the evidence reviewed above, we have outlined our evidence-based recommendations for pharmacologic VTE prophylaxis in patients undergoing orthopedic surgery, as presented in Table 1. All patients undergoing major orthopedic surgical procedures (ie, procedures other than arthroscopy) should routinely receive anticoagulant prophylaxis unless they have contraindications to anticoagulation. Recommended agents and their duration of use vary according to the type of surgery, as detailed in Table 1.
Extended-duration prophylaxis is recommended for patients undergoing total hip replacement and hip fracture surgery. Aspirin is not recommended as the sole agent for prophylaxis in any orthopedic surgery setting.
Importance of a postoperative prophylaxis protocol
In addition to these broad pharmacologic recommendations, it is important that a postoperative VTE prophylaxis protocol be in place at all hospitals.
At the Ochsner Medical Center in New Orleans, where one of us (S.B.D.) practices, postoperative orders include antithrombotic therapy for surgical patients, starting with placement of thigh-high antiembolism stockings on both legs on the day of surgery for patients undergoing hip replacement and on postoperative day 1 in those undergoing knee replacement. Plantar pneumatic compression devices are applied to both legs in the recovery room and kept on except when the patient is walking. The hospitalist team dictates further anticoagulation orders. If extended prophylaxis is prescribed, the discharge planner sets up drug delivery and reimbursement, provides a LMWH discharge kit, and teaches the patient to self-inject. If there is concern about increasing swelling at the surgical site while anticoagulant therapy continues, the protocol calls for prompt notification of the responsible physician. To minimize the risk that spinal or epidural hematomas will develop, all agents that increase bleeding propensity should be recognized and ordered accordingly.



SUMMARY
VTE in patients undergoing major orthopedic surgery is a serious health problem that is highly preventable, yet VTE prophylaxis remains underused in this patient population. Despite the availability of practice guidelines for VTE prevention in the orthopedic surgery setting, recommendations are not widely implemented in clinical practice. Recommended prophylactic options differ somewhat among various orthopedic procedures, and the supportive evidence differs for various anticoagulant options.
DISCUSSION: ADDITIONAL PERSPECTIVES FROM THE AUTHORS
Dr. Jaffer: The ACCP recommends against the routine use of aspirin as primary prophylaxis against VTE in major orthopedic surgery, yet orthopedic surgeons across the country still continue to use aspirin in this setting. What are your thoughts on this, Dr. McKean?
Dr. McKean: We agree with the ACCP’s recommendation against aspirin as primary VTE prophylaxis in orthopedic patients. The percentage of US knee arthroplasty patients who develop VTE after receiving no prophylaxis at all is roughly 64%; this percentage declines only slightly (to 56%) for knee arthroplasty patients who receive prophylaxis with aspirin.25 Since we clearly want to reduce VTE risk as much as possible, I would not use aspirin alone. I would use it only if the patient were already on aspirin, but then I would add either LMWH or fondaparinux.
Dr. Jaffer: Warfarin is another agent that is widely used for prophylaxis in major orthopedic surgery. In fact, the large registries of VTE prevention in major orthopedic surgery suggest that the use of warfarin may be slightly higher than the use of LMWH. If clinicians choose to use warfarin in their practice, what are your recommendations, Dr. Deitelzweig?
Dr. Deitelzweig: As primary prophylaxis for orthopedic surgery patients, warfarin must be dosed to achieve an INR of 2.0 to 3.0; the need for a value in this range is unequivocal. This is a challenging target to attain in the hospital setting.
Dr. Brotman: A study I was involved with a few years ago suggested that warfarin may be inadequate for VTE prevention in the first few days after orthopedic surgery.26 Orthopedic surgeons at the Cleveland Clinic, where I was practicing at the time, routinely used systematic ultrasonography to assess for thrombosis on postoperative day 2 or 3 following hip or knee arthroplasty, so we conducted a secondary analysis of a case-control study in these ultrasonographically screened arthroplasty patients to assess rates of early VTE and look for any associations with the type of prophylaxis used. We found that there was about a tenfold increase in the risk of VTE, both distal and proximal, on postoperative day 2 or 3 among patients who received warfarin compared with those who received LMWH. We concluded that warfarin’s delayed antithrombotic effects may not provide sufficient VTE prophylaxis in the immediate postoperative setting.26
Dr. Deitelzweig: That’s a good point. Although it’s important to achieve a therapeutic level of warfarin, we now have evidence that it takes some time to achieve that level, and in the interim, bad things can happen to patients.
Dr. Jaffer: Orthopedic surgery encompasses several types of procedures. Dr. Amin, which specific orthopedic surgery patients stand to benefit from extended prophylaxis, and how long should extended prophylaxis last?
Dr. Amin: Major orthopedic surgery comprises hip fracture repair, total hip replacement, and total knee replacement. For hip fracture, there are strong data to support the use of extended prophylaxis with fondaparinux 2.5 mg/day, which showed about an 88% relative reduction in the risk of symptomatic VTE compared with standard-duration fondaparinux (6 to 8 days) followed by matching placebo for the extended phase.27 The total duration of fondaparinux therapy in the extended-duration arm was 4 to 5 weeks.
Likewise, data support extended prophylaxis in hip arthroplasty patients, for whom the recommended duration is also 4 to 5 weeks. The systematic review by Hull et al17 demonstrated a 0.41 relative risk of DVT with extended-duration LMWH prophylaxis versus placebo in hip replacement patients (Figure 1), which was a highly statistically significant result.
In contrast, we do not yet have good data to support extended prophylaxis for patients undergoing total knee replacement, which is a bit surprising. In this setting, prophylaxis is recommended for 7 to 14 days but not beyond that.
Dr. Jaffer: Arthroscopy is probably the most common orthopedic procedure performed in the United States today. Dr. Brotman, what is the role of prophylaxis in patients undergoing arthroscopy?
Dr. Brotman: Minor surgery such as arthroscopy can typically be performed safely without routine prophylaxis, other than having the patient ambulate as soon as possible after the procedure. There may be exceptions to this rule, however. I believe that there is potentially a role for pharmacologic prophylaxis in arthroscopy patients who have major risk factors for VTE, such as a personal history of VTE, or who are not expected to become mobile again in a normal rapid fashion after the operation, but prophylaxis has not been studied systematically in such patients.
Dr. Jaffer: Dr. Spyropoulos, there are several new anticoagulants in the pipeline, specifically agents such as the oral direct factor Xa inhibitors and the direct thrombin inhibitors. What do recent clinical trials suggest with regard to the efficacy of these two drug classes for thromboprophylaxis in major orthopedic surgery?
Dr. Spyropoulos: The agents with the most available data are the oral direct factor Xa inhibitors apixaban and rivaroxaban and the oral direct thrombin inhibitor dabigatran. For prophylaxis in orthopedic surgery populations, phase 2 studies have been completed for apixaban and phase 3 trials have been completed for rivaroxaban and dabigatran.
It appears that the factor Xa inhibitors, apixaban and rivaroxaban, are efficacious in comparison with both adjusted-dose warfarin and LMWH, which is the gold standard for this group of patients.28,29 So these indeed appear to be promising agents. Rivaroxaban has been submitted to European regulatory agencies for approval for the prevention of VTE in patients undergoing major orthopedic surgery, and its developer plans to submit it to the FDA in 2008 for a similar indication in the United States.
The data are more equivocal with dabigatran. There have been several positive phase 3 studies in orthopedic surgery comparing two dabigatran dosing schemes, 150 and 220 mg once daily, with the European regimen of enoxaparin (40 mg once daily),30 but a recent study that compared these doses with the North American enoxaparin regimen (30 mg twice daily) failed to meet the criteria for noninferiority.31 Further clinical trial development is necessary for dabigatran, although in January 2008 the European Medicines Agency recommended its marketing approval for thromboprophylaxis in patients undergoing orthopedic procedures.32
I believe that in the next 3 to 5 years our armamentarium will see the addition of at least one, if not more, of these new agents that offer the promise of oral anticoagulation with highly predictable pharmacokinetics and pharmacodynamics and no need for monitoring.
- Yu HT, Dylan ML, Lin J, Dubois RW. Hospitals’ compliance with prophylaxis guidelines for venous thromboembolism. Am J Health Syst Pharm 2007; 64:69–76.
- Stratton MA, Anderson FA, Bussey HI, et al. Prevention of venous thromboembolism: adherence to the 1995 ACCP consensus guidelines for surgical patients. Arch Intern Med 2000; 160:334–340.
- National Consensus Standards for Prevention and Care of Venous Thromboembolism (VTE). The Joint Commission Web site. http://www.jointcommission.org/PerformanceMeasurement/Perform anceMeasurement/VTE.htm. Accessed January 8, 2008.
- Surgical Care Improvement Project. MedQIC Web site. http://www.medqic.org/scip. Accessed January 8, 2008.
- SCIP process and outcome measures, October 2005. MedQIC Web site. http://www.medqic.org. Accessed January 1, 2007.
- Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 2004; 126(3 Suppl):338S–400S.
- Zimlich RH, Fulbright BM, Friedman RJ. Current status of anticoagulation therapy after total hip and total knee arthroplasty.J Am Acad Orthop Surg 1996; 4:54–62.
- Planes A, Vochelle N, Mazas F, et al. Prevention of postoperative venous thrombosis: a randomized trial comparing unfractionated heparin with low molecular weight heparin in patients undergoing total hip replacement. Thromb Haemost 1988; 60:407–410.
- Colwell CW Jr, Spiro TE, Trowbridge AA, et al. Efficacy and safety of enoxaparin versus unfractionated heparin for prevention of deep venous thrombosis after elective knee arthroplasty. Clin Orthop Relat Res 1995; 321:19–27.
- Bauer KA, Eriksson BI, Lassen MR, Turpie AG. Fondaparinux compared with enoxaparin for the prevention of venous thromboembolism after elective major knee surgery. N Engl J Med 2001; 345:1305–1310.
- Turpie AGG. Pentasaccharide Org31540/SR90107A clinical trials update: lessons for practice. Am Heart J 2001; 142(Suppl):S9–S15.
- Hylek EM, Singer DE. Risk factors for intracranial hemorrhage in outpatients taking warfarin. Ann Intern Med 1994; 120:897–902.
- Hylek EM, Skates SJ, Sheehan MA, Singer DE. An analysis of the lowest effective intensity of prophylactic anticoagulation for patients with nonrheumatic atrial fibrillation. N Engl J Med 1996; 335:540–546.
- Samsa GP, Matchar DB, Goldstein LB, et al. Quality of anticoagulation management among patients with atrial fibrillation: review of medical records from 2 communities. Arch Intern Med 2000; 160:967–973.
- PEP Trial Collaborative Group. Prevention of pulmonary embolism and deep vein thrombosis with low dose aspirin: Pulmonary Embolism Prevention (PEP) trial. Lancet 2000; 355:1295–1302.
- Warkentin TE. Heparin-induced thrombocytopenia: pathogenesis and management. Br J Haemotol 2003; 121:535–555.
- Hull RD, Pineo GF, Stein PD, et al. Extended out-of-hospital low-molecular-weight heparin prophylaxis against deep venous thrombosis in patients after elective hip arthroplasty: a systematic review. Ann Intern Med 2001; 135:858–869.
- Haentjens P, De Groote K, Annemans L. Prolonged enoxaparin therapy to prevent venous thromboembolism after primary hip or knee replacement: a cost-utility analysis. Arch Orthop Trauma Surg 2004; 124:507–517.
- Colwell CW Jr, Collis DK, Paulson R, et al. Comparison of enoxaparin and warfarin for the prevention of venous thromboembolic disease after total hip arthroplasty: evaluation during hospitalization and three months after discharge. J Bone Joint Surg Am 1999; 81:932–940.
- Hull RD, Pineo GF, Francis C, et al. Low-molecular-weight heparin prophylaxis using dalteparin in close proximity to surgery vs warfarin in hip arthroplasty patients: a double-blind, randomized comparison. Arch Intern Med 2000; 160:2199–2207.
- Leclerc JR, Geerts WH, Desjardins L, et al. Prevention of venous thromboembolism after knee arthroplasty: a randomized, double-blind trial comparing enoxaparin with warfarin. Ann Intern Med 1996; 124:619–626.
- Fitzgerald RH Jr, Spiro TE, Trowbridge AA, et al. Prevention of venous thromboembolic disease following primary total knee arthroplasty: a randomized, multicenter, open-label, parallel-group comparison of enoxaparin and warfarin. J Bone Joint Surg Am 2001; 83-A:900–906.
- Eriksson BI, Bauer KA, Lassen MR, Turpie AG, Steering Committee of the Pentasaccharide in Hip-Fracture Surgery Study. Fondaparinux compared with enoxaparin for the prevention of venous thromboembolism after hip-fracture surgery. N Engl J Med 2001; 345:1298–1304.
- Demers C, Marcoux S, Ginsberg JS, Laroche F, Cloutier R, Poulin J. Incidence of venographically proved deep vein thrombosis after knee arthroscopy. Arch Intern Med 1998; 158:47–50.
- Geerts WH, Heit JA, Clagett GP, et al. Prevention of venous thromboembolism. Chest 2001; 119(1 Suppl):132S–175S.
- Brotman DJ, Jaffer AK, Hurbanek JG, Morra N. Warfarin prophylaxis and venous thromboembolism in the first 5 days following hip and knee arthroplasty. Thromb Haemost 2004; 92:1012–1017.
- Eriksson BI, Lassen MR; Pentasaccharide in Hip-Fracture Surgery Plus Investigators. Duration of prophylaxis against venous thromboembolism with fondaparinux after hip fracture surgery: a multicenter, randomized, placebo-controlled, double-blind study. Arch Intern Med 2003; 163:1337–1342.
- The Botticelli Investigators. Late-breaking clinical trial: a dose-finding study of the oral direct factor Xa inhibitor apixaban in the treatment of patients with acute symptomatic deep vein thrombosis [abstract]. Presented at the 21st Congress of the International Society on Thrombosis and Haemostasis; July 2007; Geneva, Switzerland.
- Fisher WD, Eriksson BI, Bauer KA, et al. Rivaroxaban for thromboprophylaxis after orthopaedic surgery: pooled analysis of two studies. Thromb Haemost 2007; 97:931–937.
- Haas S. New oral Xa and IIa inhibitors: updates on clinical trial results. J Thromb Thrombolysis 2008; 25:52–60.
- Friedman RJ, Caprini JA, Comp PC, et al. Dabigatran etexilate vs enoxaparin in preventing venous thromboembolism following total knee arthroplasty. Presented at: 2007 Congress of the International Society on Thrombosis and Haemostasis; July 7–13, 2007; Geneva, Switzerland.
- Committee for Medicinal Products for Human Use summary of positive opinion for Pradaxa [news release]. London, UK: European Medicines Agency. January 24, 2008. http://www.emea.europa.eu/pdfs/human/ opinion/Pradaxa_3503008en.pdf. Accessed February 21, 2008.
- Goldhaber SZ, Grodstein F, Stampfer MJ. A prospective study of risk factors for pulmonary embolism in women. JAMA 1997; 277:642–645.
- Turpie AGG, Bauer KA, Eriksson BI, Lassen MR, for the Steering Committees of the Pentasaccharide Orthopedic Prophylaxis Studies. Fondaparinux vs enoxaparin for the prevention of venous thromboembolism in major orthopedic surgery. Arch Intern Med 2002; 162:1833–1840.
- Stein PD, Beemath A, Matta F, et al. Clinical characteristics of patients with acute pulmonary embolism: data from PIOPED II. Am J Med 2007; 120:871–879.
- Goldhaber SZ. Diagnosis of acute pulmonary embolism: always be vigilant. Am J Med 2007; 120:827–828.
- American Academy of Orthopaedic Surgeons Clinical Guideline on Prevention of Symptomatic Pulmonary Embolism in Patients Undergoing Total Hip or Knee Arthroplasty: Summary of Recommendations. http://www.aaos.org/Research/guidelines/PE_ summary.pdf. Accessed December 10, 2007.
Nearly half of orthopedic surgery patients do not receive appropriate prophylaxis for venous thromboembolism (VTE), as defined by American College of Chest Physicians (ACCP) consensus guidelines, according to a recent analysis of a nationwide database of hospital admissions.1 Even in teaching hospitals, compliance with consensus guidelines for thromboprophylaxis is suboptimal. In a study of adherence to the ACCP guidelines for VTE prevention among 1,907 surgical patients at 10 teaching hospitals, only 45.2% of hip fracture patients received optimal VTE prophylaxis.2 Rates of optimal prophylaxis were higher among patients undergoing hip arthroplasty and knee arthroplasty—84.3% and 75.9%, respectively—but were still in need of improvement.2
GROWING INTEREST IN POSTOPERATIVE VTE PROPHYLAXIS AS A QUALITY INDICATOR
As noted in the introductory article in this supplement, the Joint Commission on Accreditation of Healthcare Organizations has taken notice of these shortcomings and has proposed national consensus standards for VTE prevention and treatment.3 Among its proposed standards are two related to risk assessment and prophylaxis: whether risk assessment/prophylaxis is ordered within 24 hours of hospital admission and within 24 hours of transfer to the intensive care unit.
Other quality-monitoring initiatives are focused specifically on VTE in the surgical population. The Surgical Care Improvement Project (SCIP) has approved two quality measures with respect to VTE prevention: (1) the proportion of surgical patients for whom recommended VTE prophylaxis is ordered, and (2) the proportion of patients who receive appropriate VTE prophylaxis (based on ACCP guideline recommendations) within 24 hours before or after surgery.4
In the future, two other VTE-related quality measures from SCIP may be implemented by the Centers for Medicare and Medicaid Services: (1) how often intra- or postoperative pulmonary embolism (PE) is diagnosed during the index hospitalization and within 30 days of surgery, and (2) how often intra- or postoperative deep vein thrombosis (DVT) is diagnosed during the index hospitalization and within 30 days of surgery.5
VTE RISK IN ORTHOPEDIC SURGERY
Surgical patients can be stratified into four VTE risk levels—low, moderate, high, and highest—based on age, surgery type, surgery duration, duration of immobilization, and other risk factors.6 For patients undergoing orthopedic surgery, these levels may be defined according to the following patient and surgical characteristics:
- Low risk—surgery duration of less than 30 minutes, age less than 40 years, repair of small fractures
- Moderate risk—age of 40 to 60 years, arthroscopy or repair of lower leg fractures, postoperative plaster cast
- High risk—age greater than 60 years, or age 40 to 60 years with additional VTE risk factors, or immobilization for greater than 4 days
- Highest risk—hip or knee arthroplasty, hip fracture repair, repair of open lower leg fractures, major trauma or spinal cord injury, or multiple risk factors for VTE (age > 40 years, prior VTE, cancer, or hypercoagulable state).
For patients in the low-risk category, no specific prophylaxis is indicated beyond early and aggressive ambulation.6 For those in all other risk categories, prophylaxis with pharmacologic anticoagulant agents and/or mechanical devices is indicated, as reviewed below.
All major orthopedic procedures confer highest risk level
Notably, the “highest risk” category includes any patient undergoing hip or knee arthroplasty or hip fracture repair. Among orthopedic surgery patients in this highest-risk category, rates of VTE events in the absence of prophylaxis are as follows:6
- Calf DVT, 40% to 80%
- Proximal DVT, 10% to 20%
- Clinical PE, 4% to 10%
- Fatal PE, 0.2% to 5%.
Hip replacement poses greater risk than knee replacement
Within this overall highest-risk category, thromboembolic risk in the absence of prophylaxis differs among procedures. Although patients undergoing hip replacement and those undergoing knee replacement have similar rates of DVT of any type,6,7 hip replacement is associated with higher rates of the more clinically important events, specifically proximal DVT and PE. In the absence of prophylaxis, proximal DVT occurs in 23% to 36% of hip replacement patients as opposed to 9% to 20% of knee replacement patients; similarly, PE occurs in 0.7% to 30% of hip replacement patients as compared with 1.8% to 7.0% of knee replacement patients.6,7
What about bleeding risk?
For many orthopedic surgeons, the risk of bleeding as a result of anticoagulant prophylaxis of VTE looms larger than the risk of VTE itself. This is likely because bleeding, when it does occur, is likely to occur more acutely than VTE does and may directly compromise the result of the operation. For this reason, orthopedic surgeons may be more likely to actually witness bleeding events than VTE events (especially fatal PEs) while their patients are still under their care, leading to a misperception of the relative risks of anticoagulation-related bleeding and thromboembolism.
In reality, rates of major bleeding with pharmacologic prophylaxis of VTE are a tiny fraction of the above-listed rates of VTE events in the absence of prophylaxis in patients undergoing major orthopedic surgery. Reported 30-day rates of major bleeding in patients receiving VTE prophylaxis with heparins range from 0.2% to 1.7%; these rates barely differ from the rates among placebo recipients in the same VTE prophylaxis trials, which range from 0.2% to 1.5%.8,9 Additionally, within the continuum of risk of major bleeding from various medical interventions, VTE prophylaxis with heparins is one of the lowest-risk interventions, posing far less risk than, for example, the use of warfarin in ischemic stroke patients or in patients older than 75 years.
PHARMACOLOGIC OPTIONS FOR VTE PROPHYLAXIS IN ORTHOPEDIC SURGERY
As reviewed in the introductory article of this supplement, the arsenal of anticoagulants for use in VTE prophylaxis includes low-dose unfractionated heparin (UFH), low-molecular-weight heparin (LMWH) agents such as dalteparin and enoxaparin, and the factor Xa inhibitor fondaparinux. A few additional comments about these and other anticoagulant options is warranted in the specific context of orthopedic surgery.
Fondaparinux. Because most of its formal US indications are for use as VTE prophylaxis in major orthopedic surgery—including hip replacement, knee replacement, and hip fracture repair—fondaparinux has been studied more widely in orthopedic surgery patients than in the other populations reviewed earlier in this supplement. Nevertheless, its use even in these settings has remained somewhat limited. This may be because of concerns over possible increased bleeding risk relative to some other anticoagulants. Because of bleeding risk, fondaparinux is contraindicated in patients who weigh less than 50 kg, and its package insert recommends caution when it is used in the elderly due to an increased risk of bleeding in patients aged 65 or older. Additionally, the Pentasaccharide in Major Knee Surgery (PENTAMAKS) study found fondaparinux to be associated with a significantly higher incidence of major bleeding compared with enoxaparin (2.1% vs 0.2%; P = .006) in major knee surgery, although it was superior to enoxaparin in preventing VTE.10 Other possible reasons for slow adoption of fondaparinux include its long half-life, which results in a sustained antithrombotic effect, its lack of easy reversibility, and a contraindication in patients with renal insufficiency.11
Limited role for UFH. Low-dose UFH has a more limited role in orthopedic surgery than in other settings requiring VTE prophylaxis, as current ACCP guidelines for VTE prevention recognize it only as a possible option in hip fracture surgery and state that it is not to be considered as sole prophylaxis in patients undergoing hip or knee replacement.6
Warfarin. Although not indicated for use in other VTE prophylaxis settings, the vitamin K antagonist warfarin is recommended as an option for all three major orthopedic surgery indications—knee replacement, hip replacement, and hip fracture repair.6
The key to effective prophylaxis with warfarin is achieving the appropriate intensity of anticoagulation. In two separate analyses, Hylek et al demonstrated a balance between safety and efficacy with warfarin therapy targeted to an international normalized ratio (INR) of 2.0 to 3.0.12,13 An INR greater than 4.0 greatly increased the risk of intracranial hemorrhage, whereas thrombosis was not effectively prevented with an INR less than 2.0.12,13 This latter point should be stressed to orthopedic surgeons, who sometimes aim for INR values below 2.0.
Although anticoagulation clinics are superior to usual care at maintaining the INR within the window of 2.0 to 3.0, only about one-third of patients nationally who take warfarin receive care in such clinics.14 Even with optimal care in anticoagulation clinics, some patients will still receive subtherapeutic or supertherapeutic levels of warfarin, which is one of this agent’s limitations.
Aspirin not recommended as sole agent. Although aspirin is still used as thromboprophylaxis in orthopedic surgery patients, current ACCP guidelines recommend against its use as the sole means of VTE prophylaxis in any patient group.6 The limitations of the evidence for aspirin in this setting are illustrated by the Pulmonary Embolism Prevention study, a multicenter randomized trial in patients undergoing hip fracture (n = 13,356) or hip/knee replacement (n = 4,088).15 Patients received aspirin 160 mg/day or placebo for 5 weeks, starting preoperatively, and were evaluated for outcomes at day 35. Among the hip fracture patients, the rate of symptomatic DVT was lower in the aspirin group than in the placebo group (1.0% vs 1.5%; P = .03), as was the rate of PE (0.7% vs 1.2%, respectively; P = .002), but there was no significant difference in outcomes between the groups among the patients undergoing hip or knee replacement. Notably, 40% of patients in the study also received UFH or LMWH. Further confounding the results, some patients received nonpharmacologic VTE prophylaxis modalities, and others received nonsteroidal anti-inflammatory drugs other than aspirin.
Heparin-induced thrombocytopenia. As noted earlier in this supplement, the incidence of heparin-induced thrombocytopenia (HIT) is markedly higher in patients who receive UFH than in those who receive LMWH. This difference in frequency, which constitutes about a sixfold to eightfold differential, is due to the relationship between standard heparin and platelet factor IV, which can induce formation of IgG antibodies.16 A 50% or greater reduction in platelet count in heparin recipients should prompt consideration of HIT.
Oral direct thrombin inhibitors. Although the oral direct thrombin inhibitor ximelagatran was rejected for approval by the US Food and Drug Administration (FDA) and recently withdrawn from the market worldwide as a result of hepatic risks, other oral direct thrombin inhibitors are in phase 3 studies for use in orthopedic surgery and may be commercially available options for postoperative VTE prophylaxis before long.
GUIDELINES FOR VTE PROPHYLAXIS IN ORTHOPEDIC SURGERY
The ACCP guidelines referred to throughout this article are widely recognized as a practice standard for VTE prevention and treatment, and have been regularly updated throughout recent decades. The most recent version, issued in 2004, is formally known as the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy.6 Key orthopedic surgery-related recommendations and notable changes from the previous version of the guidelines, issued in 2001, are outlined below, along with pertinent supportive or illustrative studies.
Hip replacement surgery
In a change from the previous guidelines, the Seventh ACCP Conference recommends extended prophylaxis, for up to 28 to 35 days after surgery, for patients undergoing hip replacement or hip fracture surgery. For hip replacement surgery, this is a Grade 1A recommendation for prophylaxis with either LMWH or warfarin and a Grade 1C+ recommendation (“no RCTs but strong RCT results can be unequivocally extrapolated, or overwhelming evidence from observational studies”) for prophylaxis with fondaparinux.6
Knee replacement surgery
The same three anticoagulant options that received Grade 1A recommendations for patients undergoing total hip replacement—LMWH, fondaparinux, and adjusted-dose warfarin—are also given Grade 1A recommendations as routine thromboprophylaxis in patients undergoing elective knee replacement (see Table 1 for dosing). In addition, optimal use of intermittent pneumatic compression devices is recommended as an alternative option to anticoagulant prophylaxis in these patients (Grade 1B, indicating a “strong recommendation” based on RCTs with important limitations). Use of UFH as the sole agent for prophylaxis is recommended against.6
For both hip and knee replacement surgery, the Seventh ACCP Conference does not endorse superiority of any one of its three recommended prophylaxis options—LMWH, fondaparinux, and adjusted-dose warfarin—over the other two. However, at least four large randomized trials have directly compared LMWH and adjusted-dose warfarin in the setting of arthroplasty—two in total hip replacement surgery19,20 and two in total knee replacement surgery.21,22 Each of these four studies found LMWH to be significantly more effective than warfarin in preventing VTE. In three of the four trials, there was no significant difference between the therapies in rates of major bleeding.19,21,22 In the remaining trial, which was conducted in hip replacement surgery patients and compared postoperative warfarin with dalteparin initiated either immediately before or early after surgery, patients who received preoperative dalteparin initiation (but not those who received postoperative dalteparin initiation) had an increased rate of major bleeding compared with warfarin recipients (P = .01).20
Hip fracture surgery
The supportive evidence for anticoagulant prophylaxis in hip fracture surgery is less robust than that in hip and knee replacement surgery. As a result, only fondaparinux has a Grade 1A recommendation as routine prophylaxis in patients undergoing hip fracture surgery. Options with less definitive recommendations are LMWH (Grade 1C+), low-dose UFH (Grade 1B), and adjusted-dose warfarin (Grade 2B, indicating a “weak recommendation” based on RCTs with important limitations) (see Table 1 for dosing of all agents).6
These differing recommendations are supported by the double-blind Pentasaccharide in Hip Fracture Surgery Study (PENTHIFRA) of 1,711 consecutive patients undergoing surgery for hip fracture repair.23 Patients were randomized to at least 5 days of fondaparinux 2.5 mg once daily, initiated postoperatively, or enoxaparin 40 mg once daily, initiated preoperatively. The incidence of DVT or PE by postoperative day 11 was 8.3% in the fondaparinux arm versus 19.1% in the enoxaparin arm, a statistically significant difference (P < .001) in favor of fondaparinux. There were no differences between the groups in rates of death or clinically relevant bleeding.
As noted above, the newly added recommendation in the Seventh ACCP Conference for extended prophylaxis, for up to 28 to 35 days after surgery, applies to patients undergoing hip fracture surgery as well as those undergoing hip replacement surgery. In the setting of hip fracture repair, extended prophylaxis is a Grade 1A recommendation with the use of fondaparinux and a Grade 1C+ recommendation with the use of either LMWH or adjusted-dose warfarin.6
Lower extremity fractures and trauma
Although lower extremity fractures are very common, the risk of DVT has been poorly studied in this setting. For patients with isolated lower extremity fractures, the Seventh ACCP Conference recommends that clinicians not use thromboprophylaxis routinely (Grade 2A, indicating an “intermediate-strength recommendation” based on RCTs without important limitations).6
Trauma patients, in contrast, are well recognized as being at very high risk for DVT and PE. The Seventh ACCP Conference gives a Grade 1A recommendation to thromboprophylaxis for all trauma patients who have at least one risk factor for VTE. LMWH is recommended (Grade 1A) as the agent of choice for this purpose, provided there are no contraindications to its use, and should be administered as soon as safely possible. Mechanical modalities are reserved for trauma patients with active bleeding or high risk for hemorrhage (Grade 1B). The guidelines recommend against use of inferior vena cava (IVC) filters as primary thromboprophylaxis in trauma patients (Grade 1C, indicating an “intermediate-strength recommendation” based on observational studies).6
Use of ultrasonography
Duplex ultrasonographic screening is recommended in orthopedic trauma patients who are at high risk for VTE and have received suboptimal or no prophylaxis (Grade 1C). In contrast, the Seventh ACCP Conference recommends against routine use of duplex ultrasonography to screen for VTE at hospital discharge in asymptomatic patients following major orthopedic surgery (Grade 1A).6
Knee arthroscopy
Arthroscopic knee procedures are increasing in frequency and raise the specter of a potential role for thromboprophylaxis. However, the clinical diagnosis of DVT is unreliable, and even diagnosis by ultrasonography is unreliable following knee arthroscopy, as interpreting scans of veins below the knee is challenging in this setting.24
The Seventh ACCP Conference recommends that clinicians not use routine thromboprophylaxis, other than early mobilization, for patients who undergo knee arthroscopy (Grade 2B). However, for arthroscopy patients who have inherent risk factors for VTE or who undergo a prolonged or complicated arthroscopy procedure, thromboprophylaxis with LMWH is suggested (Grade 2B).6
RECOMMENDED APPROACH TO VTE PROPHYLAXIS IN ORTHOPEDIC SURGERY
Drawing on the ACCP guidelines and the evidence reviewed above, we have outlined our evidence-based recommendations for pharmacologic VTE prophylaxis in patients undergoing orthopedic surgery, as presented in Table 1. All patients undergoing major orthopedic surgical procedures (ie, procedures other than arthroscopy) should routinely receive anticoagulant prophylaxis unless they have contraindications to anticoagulation. Recommended agents and their duration of use vary according to the type of surgery, as detailed in Table 1.
Extended-duration prophylaxis is recommended for patients undergoing total hip replacement and hip fracture surgery. Aspirin is not recommended as the sole agent for prophylaxis in any orthopedic surgery setting.
Importance of a postoperative prophylaxis protocol
In addition to these broad pharmacologic recommendations, it is important that a postoperative VTE prophylaxis protocol be in place at all hospitals.
At the Ochsner Medical Center in New Orleans, where one of us (S.B.D.) practices, postoperative orders include antithrombotic therapy for surgical patients, starting with placement of thigh-high antiembolism stockings on both legs on the day of surgery for patients undergoing hip replacement and on postoperative day 1 in those undergoing knee replacement. Plantar pneumatic compression devices are applied to both legs in the recovery room and kept on except when the patient is walking. The hospitalist team dictates further anticoagulation orders. If extended prophylaxis is prescribed, the discharge planner sets up drug delivery and reimbursement, provides a LMWH discharge kit, and teaches the patient to self-inject. If there is concern about increasing swelling at the surgical site while anticoagulant therapy continues, the protocol calls for prompt notification of the responsible physician. To minimize the risk that spinal or epidural hematomas will develop, all agents that increase bleeding propensity should be recognized and ordered accordingly.



SUMMARY
VTE in patients undergoing major orthopedic surgery is a serious health problem that is highly preventable, yet VTE prophylaxis remains underused in this patient population. Despite the availability of practice guidelines for VTE prevention in the orthopedic surgery setting, recommendations are not widely implemented in clinical practice. Recommended prophylactic options differ somewhat among various orthopedic procedures, and the supportive evidence differs for various anticoagulant options.
DISCUSSION: ADDITIONAL PERSPECTIVES FROM THE AUTHORS
Dr. Jaffer: The ACCP recommends against the routine use of aspirin as primary prophylaxis against VTE in major orthopedic surgery, yet orthopedic surgeons across the country still continue to use aspirin in this setting. What are your thoughts on this, Dr. McKean?
Dr. McKean: We agree with the ACCP’s recommendation against aspirin as primary VTE prophylaxis in orthopedic patients. The percentage of US knee arthroplasty patients who develop VTE after receiving no prophylaxis at all is roughly 64%; this percentage declines only slightly (to 56%) for knee arthroplasty patients who receive prophylaxis with aspirin.25 Since we clearly want to reduce VTE risk as much as possible, I would not use aspirin alone. I would use it only if the patient were already on aspirin, but then I would add either LMWH or fondaparinux.
Dr. Jaffer: Warfarin is another agent that is widely used for prophylaxis in major orthopedic surgery. In fact, the large registries of VTE prevention in major orthopedic surgery suggest that the use of warfarin may be slightly higher than the use of LMWH. If clinicians choose to use warfarin in their practice, what are your recommendations, Dr. Deitelzweig?
Dr. Deitelzweig: As primary prophylaxis for orthopedic surgery patients, warfarin must be dosed to achieve an INR of 2.0 to 3.0; the need for a value in this range is unequivocal. This is a challenging target to attain in the hospital setting.
Dr. Brotman: A study I was involved with a few years ago suggested that warfarin may be inadequate for VTE prevention in the first few days after orthopedic surgery.26 Orthopedic surgeons at the Cleveland Clinic, where I was practicing at the time, routinely used systematic ultrasonography to assess for thrombosis on postoperative day 2 or 3 following hip or knee arthroplasty, so we conducted a secondary analysis of a case-control study in these ultrasonographically screened arthroplasty patients to assess rates of early VTE and look for any associations with the type of prophylaxis used. We found that there was about a tenfold increase in the risk of VTE, both distal and proximal, on postoperative day 2 or 3 among patients who received warfarin compared with those who received LMWH. We concluded that warfarin’s delayed antithrombotic effects may not provide sufficient VTE prophylaxis in the immediate postoperative setting.26
Dr. Deitelzweig: That’s a good point. Although it’s important to achieve a therapeutic level of warfarin, we now have evidence that it takes some time to achieve that level, and in the interim, bad things can happen to patients.
Dr. Jaffer: Orthopedic surgery encompasses several types of procedures. Dr. Amin, which specific orthopedic surgery patients stand to benefit from extended prophylaxis, and how long should extended prophylaxis last?
Dr. Amin: Major orthopedic surgery comprises hip fracture repair, total hip replacement, and total knee replacement. For hip fracture, there are strong data to support the use of extended prophylaxis with fondaparinux 2.5 mg/day, which showed about an 88% relative reduction in the risk of symptomatic VTE compared with standard-duration fondaparinux (6 to 8 days) followed by matching placebo for the extended phase.27 The total duration of fondaparinux therapy in the extended-duration arm was 4 to 5 weeks.
Likewise, data support extended prophylaxis in hip arthroplasty patients, for whom the recommended duration is also 4 to 5 weeks. The systematic review by Hull et al17 demonstrated a 0.41 relative risk of DVT with extended-duration LMWH prophylaxis versus placebo in hip replacement patients (Figure 1), which was a highly statistically significant result.
In contrast, we do not yet have good data to support extended prophylaxis for patients undergoing total knee replacement, which is a bit surprising. In this setting, prophylaxis is recommended for 7 to 14 days but not beyond that.
Dr. Jaffer: Arthroscopy is probably the most common orthopedic procedure performed in the United States today. Dr. Brotman, what is the role of prophylaxis in patients undergoing arthroscopy?
Dr. Brotman: Minor surgery such as arthroscopy can typically be performed safely without routine prophylaxis, other than having the patient ambulate as soon as possible after the procedure. There may be exceptions to this rule, however. I believe that there is potentially a role for pharmacologic prophylaxis in arthroscopy patients who have major risk factors for VTE, such as a personal history of VTE, or who are not expected to become mobile again in a normal rapid fashion after the operation, but prophylaxis has not been studied systematically in such patients.
Dr. Jaffer: Dr. Spyropoulos, there are several new anticoagulants in the pipeline, specifically agents such as the oral direct factor Xa inhibitors and the direct thrombin inhibitors. What do recent clinical trials suggest with regard to the efficacy of these two drug classes for thromboprophylaxis in major orthopedic surgery?
Dr. Spyropoulos: The agents with the most available data are the oral direct factor Xa inhibitors apixaban and rivaroxaban and the oral direct thrombin inhibitor dabigatran. For prophylaxis in orthopedic surgery populations, phase 2 studies have been completed for apixaban and phase 3 trials have been completed for rivaroxaban and dabigatran.
It appears that the factor Xa inhibitors, apixaban and rivaroxaban, are efficacious in comparison with both adjusted-dose warfarin and LMWH, which is the gold standard for this group of patients.28,29 So these indeed appear to be promising agents. Rivaroxaban has been submitted to European regulatory agencies for approval for the prevention of VTE in patients undergoing major orthopedic surgery, and its developer plans to submit it to the FDA in 2008 for a similar indication in the United States.
The data are more equivocal with dabigatran. There have been several positive phase 3 studies in orthopedic surgery comparing two dabigatran dosing schemes, 150 and 220 mg once daily, with the European regimen of enoxaparin (40 mg once daily),30 but a recent study that compared these doses with the North American enoxaparin regimen (30 mg twice daily) failed to meet the criteria for noninferiority.31 Further clinical trial development is necessary for dabigatran, although in January 2008 the European Medicines Agency recommended its marketing approval for thromboprophylaxis in patients undergoing orthopedic procedures.32
I believe that in the next 3 to 5 years our armamentarium will see the addition of at least one, if not more, of these new agents that offer the promise of oral anticoagulation with highly predictable pharmacokinetics and pharmacodynamics and no need for monitoring.
Nearly half of orthopedic surgery patients do not receive appropriate prophylaxis for venous thromboembolism (VTE), as defined by American College of Chest Physicians (ACCP) consensus guidelines, according to a recent analysis of a nationwide database of hospital admissions.1 Even in teaching hospitals, compliance with consensus guidelines for thromboprophylaxis is suboptimal. In a study of adherence to the ACCP guidelines for VTE prevention among 1,907 surgical patients at 10 teaching hospitals, only 45.2% of hip fracture patients received optimal VTE prophylaxis.2 Rates of optimal prophylaxis were higher among patients undergoing hip arthroplasty and knee arthroplasty—84.3% and 75.9%, respectively—but were still in need of improvement.2
GROWING INTEREST IN POSTOPERATIVE VTE PROPHYLAXIS AS A QUALITY INDICATOR
As noted in the introductory article in this supplement, the Joint Commission on Accreditation of Healthcare Organizations has taken notice of these shortcomings and has proposed national consensus standards for VTE prevention and treatment.3 Among its proposed standards are two related to risk assessment and prophylaxis: whether risk assessment/prophylaxis is ordered within 24 hours of hospital admission and within 24 hours of transfer to the intensive care unit.
Other quality-monitoring initiatives are focused specifically on VTE in the surgical population. The Surgical Care Improvement Project (SCIP) has approved two quality measures with respect to VTE prevention: (1) the proportion of surgical patients for whom recommended VTE prophylaxis is ordered, and (2) the proportion of patients who receive appropriate VTE prophylaxis (based on ACCP guideline recommendations) within 24 hours before or after surgery.4
In the future, two other VTE-related quality measures from SCIP may be implemented by the Centers for Medicare and Medicaid Services: (1) how often intra- or postoperative pulmonary embolism (PE) is diagnosed during the index hospitalization and within 30 days of surgery, and (2) how often intra- or postoperative deep vein thrombosis (DVT) is diagnosed during the index hospitalization and within 30 days of surgery.5
VTE RISK IN ORTHOPEDIC SURGERY
Surgical patients can be stratified into four VTE risk levels—low, moderate, high, and highest—based on age, surgery type, surgery duration, duration of immobilization, and other risk factors.6 For patients undergoing orthopedic surgery, these levels may be defined according to the following patient and surgical characteristics:
- Low risk—surgery duration of less than 30 minutes, age less than 40 years, repair of small fractures
- Moderate risk—age of 40 to 60 years, arthroscopy or repair of lower leg fractures, postoperative plaster cast
- High risk—age greater than 60 years, or age 40 to 60 years with additional VTE risk factors, or immobilization for greater than 4 days
- Highest risk—hip or knee arthroplasty, hip fracture repair, repair of open lower leg fractures, major trauma or spinal cord injury, or multiple risk factors for VTE (age > 40 years, prior VTE, cancer, or hypercoagulable state).
For patients in the low-risk category, no specific prophylaxis is indicated beyond early and aggressive ambulation.6 For those in all other risk categories, prophylaxis with pharmacologic anticoagulant agents and/or mechanical devices is indicated, as reviewed below.
All major orthopedic procedures confer highest risk level
Notably, the “highest risk” category includes any patient undergoing hip or knee arthroplasty or hip fracture repair. Among orthopedic surgery patients in this highest-risk category, rates of VTE events in the absence of prophylaxis are as follows:6
- Calf DVT, 40% to 80%
- Proximal DVT, 10% to 20%
- Clinical PE, 4% to 10%
- Fatal PE, 0.2% to 5%.
Hip replacement poses greater risk than knee replacement
Within this overall highest-risk category, thromboembolic risk in the absence of prophylaxis differs among procedures. Although patients undergoing hip replacement and those undergoing knee replacement have similar rates of DVT of any type,6,7 hip replacement is associated with higher rates of the more clinically important events, specifically proximal DVT and PE. In the absence of prophylaxis, proximal DVT occurs in 23% to 36% of hip replacement patients as opposed to 9% to 20% of knee replacement patients; similarly, PE occurs in 0.7% to 30% of hip replacement patients as compared with 1.8% to 7.0% of knee replacement patients.6,7
What about bleeding risk?
For many orthopedic surgeons, the risk of bleeding as a result of anticoagulant prophylaxis of VTE looms larger than the risk of VTE itself. This is likely because bleeding, when it does occur, is likely to occur more acutely than VTE does and may directly compromise the result of the operation. For this reason, orthopedic surgeons may be more likely to actually witness bleeding events than VTE events (especially fatal PEs) while their patients are still under their care, leading to a misperception of the relative risks of anticoagulation-related bleeding and thromboembolism.
In reality, rates of major bleeding with pharmacologic prophylaxis of VTE are a tiny fraction of the above-listed rates of VTE events in the absence of prophylaxis in patients undergoing major orthopedic surgery. Reported 30-day rates of major bleeding in patients receiving VTE prophylaxis with heparins range from 0.2% to 1.7%; these rates barely differ from the rates among placebo recipients in the same VTE prophylaxis trials, which range from 0.2% to 1.5%.8,9 Additionally, within the continuum of risk of major bleeding from various medical interventions, VTE prophylaxis with heparins is one of the lowest-risk interventions, posing far less risk than, for example, the use of warfarin in ischemic stroke patients or in patients older than 75 years.
PHARMACOLOGIC OPTIONS FOR VTE PROPHYLAXIS IN ORTHOPEDIC SURGERY
As reviewed in the introductory article of this supplement, the arsenal of anticoagulants for use in VTE prophylaxis includes low-dose unfractionated heparin (UFH), low-molecular-weight heparin (LMWH) agents such as dalteparin and enoxaparin, and the factor Xa inhibitor fondaparinux. A few additional comments about these and other anticoagulant options is warranted in the specific context of orthopedic surgery.
Fondaparinux. Because most of its formal US indications are for use as VTE prophylaxis in major orthopedic surgery—including hip replacement, knee replacement, and hip fracture repair—fondaparinux has been studied more widely in orthopedic surgery patients than in the other populations reviewed earlier in this supplement. Nevertheless, its use even in these settings has remained somewhat limited. This may be because of concerns over possible increased bleeding risk relative to some other anticoagulants. Because of bleeding risk, fondaparinux is contraindicated in patients who weigh less than 50 kg, and its package insert recommends caution when it is used in the elderly due to an increased risk of bleeding in patients aged 65 or older. Additionally, the Pentasaccharide in Major Knee Surgery (PENTAMAKS) study found fondaparinux to be associated with a significantly higher incidence of major bleeding compared with enoxaparin (2.1% vs 0.2%; P = .006) in major knee surgery, although it was superior to enoxaparin in preventing VTE.10 Other possible reasons for slow adoption of fondaparinux include its long half-life, which results in a sustained antithrombotic effect, its lack of easy reversibility, and a contraindication in patients with renal insufficiency.11
Limited role for UFH. Low-dose UFH has a more limited role in orthopedic surgery than in other settings requiring VTE prophylaxis, as current ACCP guidelines for VTE prevention recognize it only as a possible option in hip fracture surgery and state that it is not to be considered as sole prophylaxis in patients undergoing hip or knee replacement.6
Warfarin. Although not indicated for use in other VTE prophylaxis settings, the vitamin K antagonist warfarin is recommended as an option for all three major orthopedic surgery indications—knee replacement, hip replacement, and hip fracture repair.6
The key to effective prophylaxis with warfarin is achieving the appropriate intensity of anticoagulation. In two separate analyses, Hylek et al demonstrated a balance between safety and efficacy with warfarin therapy targeted to an international normalized ratio (INR) of 2.0 to 3.0.12,13 An INR greater than 4.0 greatly increased the risk of intracranial hemorrhage, whereas thrombosis was not effectively prevented with an INR less than 2.0.12,13 This latter point should be stressed to orthopedic surgeons, who sometimes aim for INR values below 2.0.
Although anticoagulation clinics are superior to usual care at maintaining the INR within the window of 2.0 to 3.0, only about one-third of patients nationally who take warfarin receive care in such clinics.14 Even with optimal care in anticoagulation clinics, some patients will still receive subtherapeutic or supertherapeutic levels of warfarin, which is one of this agent’s limitations.
Aspirin not recommended as sole agent. Although aspirin is still used as thromboprophylaxis in orthopedic surgery patients, current ACCP guidelines recommend against its use as the sole means of VTE prophylaxis in any patient group.6 The limitations of the evidence for aspirin in this setting are illustrated by the Pulmonary Embolism Prevention study, a multicenter randomized trial in patients undergoing hip fracture (n = 13,356) or hip/knee replacement (n = 4,088).15 Patients received aspirin 160 mg/day or placebo for 5 weeks, starting preoperatively, and were evaluated for outcomes at day 35. Among the hip fracture patients, the rate of symptomatic DVT was lower in the aspirin group than in the placebo group (1.0% vs 1.5%; P = .03), as was the rate of PE (0.7% vs 1.2%, respectively; P = .002), but there was no significant difference in outcomes between the groups among the patients undergoing hip or knee replacement. Notably, 40% of patients in the study also received UFH or LMWH. Further confounding the results, some patients received nonpharmacologic VTE prophylaxis modalities, and others received nonsteroidal anti-inflammatory drugs other than aspirin.
Heparin-induced thrombocytopenia. As noted earlier in this supplement, the incidence of heparin-induced thrombocytopenia (HIT) is markedly higher in patients who receive UFH than in those who receive LMWH. This difference in frequency, which constitutes about a sixfold to eightfold differential, is due to the relationship between standard heparin and platelet factor IV, which can induce formation of IgG antibodies.16 A 50% or greater reduction in platelet count in heparin recipients should prompt consideration of HIT.
Oral direct thrombin inhibitors. Although the oral direct thrombin inhibitor ximelagatran was rejected for approval by the US Food and Drug Administration (FDA) and recently withdrawn from the market worldwide as a result of hepatic risks, other oral direct thrombin inhibitors are in phase 3 studies for use in orthopedic surgery and may be commercially available options for postoperative VTE prophylaxis before long.
GUIDELINES FOR VTE PROPHYLAXIS IN ORTHOPEDIC SURGERY
The ACCP guidelines referred to throughout this article are widely recognized as a practice standard for VTE prevention and treatment, and have been regularly updated throughout recent decades. The most recent version, issued in 2004, is formally known as the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy.6 Key orthopedic surgery-related recommendations and notable changes from the previous version of the guidelines, issued in 2001, are outlined below, along with pertinent supportive or illustrative studies.
Hip replacement surgery
In a change from the previous guidelines, the Seventh ACCP Conference recommends extended prophylaxis, for up to 28 to 35 days after surgery, for patients undergoing hip replacement or hip fracture surgery. For hip replacement surgery, this is a Grade 1A recommendation for prophylaxis with either LMWH or warfarin and a Grade 1C+ recommendation (“no RCTs but strong RCT results can be unequivocally extrapolated, or overwhelming evidence from observational studies”) for prophylaxis with fondaparinux.6
Knee replacement surgery
The same three anticoagulant options that received Grade 1A recommendations for patients undergoing total hip replacement—LMWH, fondaparinux, and adjusted-dose warfarin—are also given Grade 1A recommendations as routine thromboprophylaxis in patients undergoing elective knee replacement (see Table 1 for dosing). In addition, optimal use of intermittent pneumatic compression devices is recommended as an alternative option to anticoagulant prophylaxis in these patients (Grade 1B, indicating a “strong recommendation” based on RCTs with important limitations). Use of UFH as the sole agent for prophylaxis is recommended against.6
For both hip and knee replacement surgery, the Seventh ACCP Conference does not endorse superiority of any one of its three recommended prophylaxis options—LMWH, fondaparinux, and adjusted-dose warfarin—over the other two. However, at least four large randomized trials have directly compared LMWH and adjusted-dose warfarin in the setting of arthroplasty—two in total hip replacement surgery19,20 and two in total knee replacement surgery.21,22 Each of these four studies found LMWH to be significantly more effective than warfarin in preventing VTE. In three of the four trials, there was no significant difference between the therapies in rates of major bleeding.19,21,22 In the remaining trial, which was conducted in hip replacement surgery patients and compared postoperative warfarin with dalteparin initiated either immediately before or early after surgery, patients who received preoperative dalteparin initiation (but not those who received postoperative dalteparin initiation) had an increased rate of major bleeding compared with warfarin recipients (P = .01).20
Hip fracture surgery
The supportive evidence for anticoagulant prophylaxis in hip fracture surgery is less robust than that in hip and knee replacement surgery. As a result, only fondaparinux has a Grade 1A recommendation as routine prophylaxis in patients undergoing hip fracture surgery. Options with less definitive recommendations are LMWH (Grade 1C+), low-dose UFH (Grade 1B), and adjusted-dose warfarin (Grade 2B, indicating a “weak recommendation” based on RCTs with important limitations) (see Table 1 for dosing of all agents).6
These differing recommendations are supported by the double-blind Pentasaccharide in Hip Fracture Surgery Study (PENTHIFRA) of 1,711 consecutive patients undergoing surgery for hip fracture repair.23 Patients were randomized to at least 5 days of fondaparinux 2.5 mg once daily, initiated postoperatively, or enoxaparin 40 mg once daily, initiated preoperatively. The incidence of DVT or PE by postoperative day 11 was 8.3% in the fondaparinux arm versus 19.1% in the enoxaparin arm, a statistically significant difference (P < .001) in favor of fondaparinux. There were no differences between the groups in rates of death or clinically relevant bleeding.
As noted above, the newly added recommendation in the Seventh ACCP Conference for extended prophylaxis, for up to 28 to 35 days after surgery, applies to patients undergoing hip fracture surgery as well as those undergoing hip replacement surgery. In the setting of hip fracture repair, extended prophylaxis is a Grade 1A recommendation with the use of fondaparinux and a Grade 1C+ recommendation with the use of either LMWH or adjusted-dose warfarin.6
Lower extremity fractures and trauma
Although lower extremity fractures are very common, the risk of DVT has been poorly studied in this setting. For patients with isolated lower extremity fractures, the Seventh ACCP Conference recommends that clinicians not use thromboprophylaxis routinely (Grade 2A, indicating an “intermediate-strength recommendation” based on RCTs without important limitations).6
Trauma patients, in contrast, are well recognized as being at very high risk for DVT and PE. The Seventh ACCP Conference gives a Grade 1A recommendation to thromboprophylaxis for all trauma patients who have at least one risk factor for VTE. LMWH is recommended (Grade 1A) as the agent of choice for this purpose, provided there are no contraindications to its use, and should be administered as soon as safely possible. Mechanical modalities are reserved for trauma patients with active bleeding or high risk for hemorrhage (Grade 1B). The guidelines recommend against use of inferior vena cava (IVC) filters as primary thromboprophylaxis in trauma patients (Grade 1C, indicating an “intermediate-strength recommendation” based on observational studies).6
Use of ultrasonography
Duplex ultrasonographic screening is recommended in orthopedic trauma patients who are at high risk for VTE and have received suboptimal or no prophylaxis (Grade 1C). In contrast, the Seventh ACCP Conference recommends against routine use of duplex ultrasonography to screen for VTE at hospital discharge in asymptomatic patients following major orthopedic surgery (Grade 1A).6
Knee arthroscopy
Arthroscopic knee procedures are increasing in frequency and raise the specter of a potential role for thromboprophylaxis. However, the clinical diagnosis of DVT is unreliable, and even diagnosis by ultrasonography is unreliable following knee arthroscopy, as interpreting scans of veins below the knee is challenging in this setting.24
The Seventh ACCP Conference recommends that clinicians not use routine thromboprophylaxis, other than early mobilization, for patients who undergo knee arthroscopy (Grade 2B). However, for arthroscopy patients who have inherent risk factors for VTE or who undergo a prolonged or complicated arthroscopy procedure, thromboprophylaxis with LMWH is suggested (Grade 2B).6
RECOMMENDED APPROACH TO VTE PROPHYLAXIS IN ORTHOPEDIC SURGERY
Drawing on the ACCP guidelines and the evidence reviewed above, we have outlined our evidence-based recommendations for pharmacologic VTE prophylaxis in patients undergoing orthopedic surgery, as presented in Table 1. All patients undergoing major orthopedic surgical procedures (ie, procedures other than arthroscopy) should routinely receive anticoagulant prophylaxis unless they have contraindications to anticoagulation. Recommended agents and their duration of use vary according to the type of surgery, as detailed in Table 1.
Extended-duration prophylaxis is recommended for patients undergoing total hip replacement and hip fracture surgery. Aspirin is not recommended as the sole agent for prophylaxis in any orthopedic surgery setting.
Importance of a postoperative prophylaxis protocol
In addition to these broad pharmacologic recommendations, it is important that a postoperative VTE prophylaxis protocol be in place at all hospitals.
At the Ochsner Medical Center in New Orleans, where one of us (S.B.D.) practices, postoperative orders include antithrombotic therapy for surgical patients, starting with placement of thigh-high antiembolism stockings on both legs on the day of surgery for patients undergoing hip replacement and on postoperative day 1 in those undergoing knee replacement. Plantar pneumatic compression devices are applied to both legs in the recovery room and kept on except when the patient is walking. The hospitalist team dictates further anticoagulation orders. If extended prophylaxis is prescribed, the discharge planner sets up drug delivery and reimbursement, provides a LMWH discharge kit, and teaches the patient to self-inject. If there is concern about increasing swelling at the surgical site while anticoagulant therapy continues, the protocol calls for prompt notification of the responsible physician. To minimize the risk that spinal or epidural hematomas will develop, all agents that increase bleeding propensity should be recognized and ordered accordingly.



SUMMARY
VTE in patients undergoing major orthopedic surgery is a serious health problem that is highly preventable, yet VTE prophylaxis remains underused in this patient population. Despite the availability of practice guidelines for VTE prevention in the orthopedic surgery setting, recommendations are not widely implemented in clinical practice. Recommended prophylactic options differ somewhat among various orthopedic procedures, and the supportive evidence differs for various anticoagulant options.
DISCUSSION: ADDITIONAL PERSPECTIVES FROM THE AUTHORS
Dr. Jaffer: The ACCP recommends against the routine use of aspirin as primary prophylaxis against VTE in major orthopedic surgery, yet orthopedic surgeons across the country still continue to use aspirin in this setting. What are your thoughts on this, Dr. McKean?
Dr. McKean: We agree with the ACCP’s recommendation against aspirin as primary VTE prophylaxis in orthopedic patients. The percentage of US knee arthroplasty patients who develop VTE after receiving no prophylaxis at all is roughly 64%; this percentage declines only slightly (to 56%) for knee arthroplasty patients who receive prophylaxis with aspirin.25 Since we clearly want to reduce VTE risk as much as possible, I would not use aspirin alone. I would use it only if the patient were already on aspirin, but then I would add either LMWH or fondaparinux.
Dr. Jaffer: Warfarin is another agent that is widely used for prophylaxis in major orthopedic surgery. In fact, the large registries of VTE prevention in major orthopedic surgery suggest that the use of warfarin may be slightly higher than the use of LMWH. If clinicians choose to use warfarin in their practice, what are your recommendations, Dr. Deitelzweig?
Dr. Deitelzweig: As primary prophylaxis for orthopedic surgery patients, warfarin must be dosed to achieve an INR of 2.0 to 3.0; the need for a value in this range is unequivocal. This is a challenging target to attain in the hospital setting.
Dr. Brotman: A study I was involved with a few years ago suggested that warfarin may be inadequate for VTE prevention in the first few days after orthopedic surgery.26 Orthopedic surgeons at the Cleveland Clinic, where I was practicing at the time, routinely used systematic ultrasonography to assess for thrombosis on postoperative day 2 or 3 following hip or knee arthroplasty, so we conducted a secondary analysis of a case-control study in these ultrasonographically screened arthroplasty patients to assess rates of early VTE and look for any associations with the type of prophylaxis used. We found that there was about a tenfold increase in the risk of VTE, both distal and proximal, on postoperative day 2 or 3 among patients who received warfarin compared with those who received LMWH. We concluded that warfarin’s delayed antithrombotic effects may not provide sufficient VTE prophylaxis in the immediate postoperative setting.26
Dr. Deitelzweig: That’s a good point. Although it’s important to achieve a therapeutic level of warfarin, we now have evidence that it takes some time to achieve that level, and in the interim, bad things can happen to patients.
Dr. Jaffer: Orthopedic surgery encompasses several types of procedures. Dr. Amin, which specific orthopedic surgery patients stand to benefit from extended prophylaxis, and how long should extended prophylaxis last?
Dr. Amin: Major orthopedic surgery comprises hip fracture repair, total hip replacement, and total knee replacement. For hip fracture, there are strong data to support the use of extended prophylaxis with fondaparinux 2.5 mg/day, which showed about an 88% relative reduction in the risk of symptomatic VTE compared with standard-duration fondaparinux (6 to 8 days) followed by matching placebo for the extended phase.27 The total duration of fondaparinux therapy in the extended-duration arm was 4 to 5 weeks.
Likewise, data support extended prophylaxis in hip arthroplasty patients, for whom the recommended duration is also 4 to 5 weeks. The systematic review by Hull et al17 demonstrated a 0.41 relative risk of DVT with extended-duration LMWH prophylaxis versus placebo in hip replacement patients (Figure 1), which was a highly statistically significant result.
In contrast, we do not yet have good data to support extended prophylaxis for patients undergoing total knee replacement, which is a bit surprising. In this setting, prophylaxis is recommended for 7 to 14 days but not beyond that.
Dr. Jaffer: Arthroscopy is probably the most common orthopedic procedure performed in the United States today. Dr. Brotman, what is the role of prophylaxis in patients undergoing arthroscopy?
Dr. Brotman: Minor surgery such as arthroscopy can typically be performed safely without routine prophylaxis, other than having the patient ambulate as soon as possible after the procedure. There may be exceptions to this rule, however. I believe that there is potentially a role for pharmacologic prophylaxis in arthroscopy patients who have major risk factors for VTE, such as a personal history of VTE, or who are not expected to become mobile again in a normal rapid fashion after the operation, but prophylaxis has not been studied systematically in such patients.
Dr. Jaffer: Dr. Spyropoulos, there are several new anticoagulants in the pipeline, specifically agents such as the oral direct factor Xa inhibitors and the direct thrombin inhibitors. What do recent clinical trials suggest with regard to the efficacy of these two drug classes for thromboprophylaxis in major orthopedic surgery?
Dr. Spyropoulos: The agents with the most available data are the oral direct factor Xa inhibitors apixaban and rivaroxaban and the oral direct thrombin inhibitor dabigatran. For prophylaxis in orthopedic surgery populations, phase 2 studies have been completed for apixaban and phase 3 trials have been completed for rivaroxaban and dabigatran.
It appears that the factor Xa inhibitors, apixaban and rivaroxaban, are efficacious in comparison with both adjusted-dose warfarin and LMWH, which is the gold standard for this group of patients.28,29 So these indeed appear to be promising agents. Rivaroxaban has been submitted to European regulatory agencies for approval for the prevention of VTE in patients undergoing major orthopedic surgery, and its developer plans to submit it to the FDA in 2008 for a similar indication in the United States.
The data are more equivocal with dabigatran. There have been several positive phase 3 studies in orthopedic surgery comparing two dabigatran dosing schemes, 150 and 220 mg once daily, with the European regimen of enoxaparin (40 mg once daily),30 but a recent study that compared these doses with the North American enoxaparin regimen (30 mg twice daily) failed to meet the criteria for noninferiority.31 Further clinical trial development is necessary for dabigatran, although in January 2008 the European Medicines Agency recommended its marketing approval for thromboprophylaxis in patients undergoing orthopedic procedures.32
I believe that in the next 3 to 5 years our armamentarium will see the addition of at least one, if not more, of these new agents that offer the promise of oral anticoagulation with highly predictable pharmacokinetics and pharmacodynamics and no need for monitoring.
- Yu HT, Dylan ML, Lin J, Dubois RW. Hospitals’ compliance with prophylaxis guidelines for venous thromboembolism. Am J Health Syst Pharm 2007; 64:69–76.
- Stratton MA, Anderson FA, Bussey HI, et al. Prevention of venous thromboembolism: adherence to the 1995 ACCP consensus guidelines for surgical patients. Arch Intern Med 2000; 160:334–340.
- National Consensus Standards for Prevention and Care of Venous Thromboembolism (VTE). The Joint Commission Web site. http://www.jointcommission.org/PerformanceMeasurement/Perform anceMeasurement/VTE.htm. Accessed January 8, 2008.
- Surgical Care Improvement Project. MedQIC Web site. http://www.medqic.org/scip. Accessed January 8, 2008.
- SCIP process and outcome measures, October 2005. MedQIC Web site. http://www.medqic.org. Accessed January 1, 2007.
- Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 2004; 126(3 Suppl):338S–400S.
- Zimlich RH, Fulbright BM, Friedman RJ. Current status of anticoagulation therapy after total hip and total knee arthroplasty.J Am Acad Orthop Surg 1996; 4:54–62.
- Planes A, Vochelle N, Mazas F, et al. Prevention of postoperative venous thrombosis: a randomized trial comparing unfractionated heparin with low molecular weight heparin in patients undergoing total hip replacement. Thromb Haemost 1988; 60:407–410.
- Colwell CW Jr, Spiro TE, Trowbridge AA, et al. Efficacy and safety of enoxaparin versus unfractionated heparin for prevention of deep venous thrombosis after elective knee arthroplasty. Clin Orthop Relat Res 1995; 321:19–27.
- Bauer KA, Eriksson BI, Lassen MR, Turpie AG. Fondaparinux compared with enoxaparin for the prevention of venous thromboembolism after elective major knee surgery. N Engl J Med 2001; 345:1305–1310.
- Turpie AGG. Pentasaccharide Org31540/SR90107A clinical trials update: lessons for practice. Am Heart J 2001; 142(Suppl):S9–S15.
- Hylek EM, Singer DE. Risk factors for intracranial hemorrhage in outpatients taking warfarin. Ann Intern Med 1994; 120:897–902.
- Hylek EM, Skates SJ, Sheehan MA, Singer DE. An analysis of the lowest effective intensity of prophylactic anticoagulation for patients with nonrheumatic atrial fibrillation. N Engl J Med 1996; 335:540–546.
- Samsa GP, Matchar DB, Goldstein LB, et al. Quality of anticoagulation management among patients with atrial fibrillation: review of medical records from 2 communities. Arch Intern Med 2000; 160:967–973.
- PEP Trial Collaborative Group. Prevention of pulmonary embolism and deep vein thrombosis with low dose aspirin: Pulmonary Embolism Prevention (PEP) trial. Lancet 2000; 355:1295–1302.
- Warkentin TE. Heparin-induced thrombocytopenia: pathogenesis and management. Br J Haemotol 2003; 121:535–555.
- Hull RD, Pineo GF, Stein PD, et al. Extended out-of-hospital low-molecular-weight heparin prophylaxis against deep venous thrombosis in patients after elective hip arthroplasty: a systematic review. Ann Intern Med 2001; 135:858–869.
- Haentjens P, De Groote K, Annemans L. Prolonged enoxaparin therapy to prevent venous thromboembolism after primary hip or knee replacement: a cost-utility analysis. Arch Orthop Trauma Surg 2004; 124:507–517.
- Colwell CW Jr, Collis DK, Paulson R, et al. Comparison of enoxaparin and warfarin for the prevention of venous thromboembolic disease after total hip arthroplasty: evaluation during hospitalization and three months after discharge. J Bone Joint Surg Am 1999; 81:932–940.
- Hull RD, Pineo GF, Francis C, et al. Low-molecular-weight heparin prophylaxis using dalteparin in close proximity to surgery vs warfarin in hip arthroplasty patients: a double-blind, randomized comparison. Arch Intern Med 2000; 160:2199–2207.
- Leclerc JR, Geerts WH, Desjardins L, et al. Prevention of venous thromboembolism after knee arthroplasty: a randomized, double-blind trial comparing enoxaparin with warfarin. Ann Intern Med 1996; 124:619–626.
- Fitzgerald RH Jr, Spiro TE, Trowbridge AA, et al. Prevention of venous thromboembolic disease following primary total knee arthroplasty: a randomized, multicenter, open-label, parallel-group comparison of enoxaparin and warfarin. J Bone Joint Surg Am 2001; 83-A:900–906.
- Eriksson BI, Bauer KA, Lassen MR, Turpie AG, Steering Committee of the Pentasaccharide in Hip-Fracture Surgery Study. Fondaparinux compared with enoxaparin for the prevention of venous thromboembolism after hip-fracture surgery. N Engl J Med 2001; 345:1298–1304.
- Demers C, Marcoux S, Ginsberg JS, Laroche F, Cloutier R, Poulin J. Incidence of venographically proved deep vein thrombosis after knee arthroscopy. Arch Intern Med 1998; 158:47–50.
- Geerts WH, Heit JA, Clagett GP, et al. Prevention of venous thromboembolism. Chest 2001; 119(1 Suppl):132S–175S.
- Brotman DJ, Jaffer AK, Hurbanek JG, Morra N. Warfarin prophylaxis and venous thromboembolism in the first 5 days following hip and knee arthroplasty. Thromb Haemost 2004; 92:1012–1017.
- Eriksson BI, Lassen MR; Pentasaccharide in Hip-Fracture Surgery Plus Investigators. Duration of prophylaxis against venous thromboembolism with fondaparinux after hip fracture surgery: a multicenter, randomized, placebo-controlled, double-blind study. Arch Intern Med 2003; 163:1337–1342.
- The Botticelli Investigators. Late-breaking clinical trial: a dose-finding study of the oral direct factor Xa inhibitor apixaban in the treatment of patients with acute symptomatic deep vein thrombosis [abstract]. Presented at the 21st Congress of the International Society on Thrombosis and Haemostasis; July 2007; Geneva, Switzerland.
- Fisher WD, Eriksson BI, Bauer KA, et al. Rivaroxaban for thromboprophylaxis after orthopaedic surgery: pooled analysis of two studies. Thromb Haemost 2007; 97:931–937.
- Haas S. New oral Xa and IIa inhibitors: updates on clinical trial results. J Thromb Thrombolysis 2008; 25:52–60.
- Friedman RJ, Caprini JA, Comp PC, et al. Dabigatran etexilate vs enoxaparin in preventing venous thromboembolism following total knee arthroplasty. Presented at: 2007 Congress of the International Society on Thrombosis and Haemostasis; July 7–13, 2007; Geneva, Switzerland.
- Committee for Medicinal Products for Human Use summary of positive opinion for Pradaxa [news release]. London, UK: European Medicines Agency. January 24, 2008. http://www.emea.europa.eu/pdfs/human/ opinion/Pradaxa_3503008en.pdf. Accessed February 21, 2008.
- Goldhaber SZ, Grodstein F, Stampfer MJ. A prospective study of risk factors for pulmonary embolism in women. JAMA 1997; 277:642–645.
- Turpie AGG, Bauer KA, Eriksson BI, Lassen MR, for the Steering Committees of the Pentasaccharide Orthopedic Prophylaxis Studies. Fondaparinux vs enoxaparin for the prevention of venous thromboembolism in major orthopedic surgery. Arch Intern Med 2002; 162:1833–1840.
- Stein PD, Beemath A, Matta F, et al. Clinical characteristics of patients with acute pulmonary embolism: data from PIOPED II. Am J Med 2007; 120:871–879.
- Goldhaber SZ. Diagnosis of acute pulmonary embolism: always be vigilant. Am J Med 2007; 120:827–828.
- American Academy of Orthopaedic Surgeons Clinical Guideline on Prevention of Symptomatic Pulmonary Embolism in Patients Undergoing Total Hip or Knee Arthroplasty: Summary of Recommendations. http://www.aaos.org/Research/guidelines/PE_ summary.pdf. Accessed December 10, 2007.
- Yu HT, Dylan ML, Lin J, Dubois RW. Hospitals’ compliance with prophylaxis guidelines for venous thromboembolism. Am J Health Syst Pharm 2007; 64:69–76.
- Stratton MA, Anderson FA, Bussey HI, et al. Prevention of venous thromboembolism: adherence to the 1995 ACCP consensus guidelines for surgical patients. Arch Intern Med 2000; 160:334–340.
- National Consensus Standards for Prevention and Care of Venous Thromboembolism (VTE). The Joint Commission Web site. http://www.jointcommission.org/PerformanceMeasurement/Perform anceMeasurement/VTE.htm. Accessed January 8, 2008.
- Surgical Care Improvement Project. MedQIC Web site. http://www.medqic.org/scip. Accessed January 8, 2008.
- SCIP process and outcome measures, October 2005. MedQIC Web site. http://www.medqic.org. Accessed January 1, 2007.
- Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 2004; 126(3 Suppl):338S–400S.
- Zimlich RH, Fulbright BM, Friedman RJ. Current status of anticoagulation therapy after total hip and total knee arthroplasty.J Am Acad Orthop Surg 1996; 4:54–62.
- Planes A, Vochelle N, Mazas F, et al. Prevention of postoperative venous thrombosis: a randomized trial comparing unfractionated heparin with low molecular weight heparin in patients undergoing total hip replacement. Thromb Haemost 1988; 60:407–410.
- Colwell CW Jr, Spiro TE, Trowbridge AA, et al. Efficacy and safety of enoxaparin versus unfractionated heparin for prevention of deep venous thrombosis after elective knee arthroplasty. Clin Orthop Relat Res 1995; 321:19–27.
- Bauer KA, Eriksson BI, Lassen MR, Turpie AG. Fondaparinux compared with enoxaparin for the prevention of venous thromboembolism after elective major knee surgery. N Engl J Med 2001; 345:1305–1310.
- Turpie AGG. Pentasaccharide Org31540/SR90107A clinical trials update: lessons for practice. Am Heart J 2001; 142(Suppl):S9–S15.
- Hylek EM, Singer DE. Risk factors for intracranial hemorrhage in outpatients taking warfarin. Ann Intern Med 1994; 120:897–902.
- Hylek EM, Skates SJ, Sheehan MA, Singer DE. An analysis of the lowest effective intensity of prophylactic anticoagulation for patients with nonrheumatic atrial fibrillation. N Engl J Med 1996; 335:540–546.
- Samsa GP, Matchar DB, Goldstein LB, et al. Quality of anticoagulation management among patients with atrial fibrillation: review of medical records from 2 communities. Arch Intern Med 2000; 160:967–973.
- PEP Trial Collaborative Group. Prevention of pulmonary embolism and deep vein thrombosis with low dose aspirin: Pulmonary Embolism Prevention (PEP) trial. Lancet 2000; 355:1295–1302.
- Warkentin TE. Heparin-induced thrombocytopenia: pathogenesis and management. Br J Haemotol 2003; 121:535–555.
- Hull RD, Pineo GF, Stein PD, et al. Extended out-of-hospital low-molecular-weight heparin prophylaxis against deep venous thrombosis in patients after elective hip arthroplasty: a systematic review. Ann Intern Med 2001; 135:858–869.
- Haentjens P, De Groote K, Annemans L. Prolonged enoxaparin therapy to prevent venous thromboembolism after primary hip or knee replacement: a cost-utility analysis. Arch Orthop Trauma Surg 2004; 124:507–517.
- Colwell CW Jr, Collis DK, Paulson R, et al. Comparison of enoxaparin and warfarin for the prevention of venous thromboembolic disease after total hip arthroplasty: evaluation during hospitalization and three months after discharge. J Bone Joint Surg Am 1999; 81:932–940.
- Hull RD, Pineo GF, Francis C, et al. Low-molecular-weight heparin prophylaxis using dalteparin in close proximity to surgery vs warfarin in hip arthroplasty patients: a double-blind, randomized comparison. Arch Intern Med 2000; 160:2199–2207.
- Leclerc JR, Geerts WH, Desjardins L, et al. Prevention of venous thromboembolism after knee arthroplasty: a randomized, double-blind trial comparing enoxaparin with warfarin. Ann Intern Med 1996; 124:619–626.
- Fitzgerald RH Jr, Spiro TE, Trowbridge AA, et al. Prevention of venous thromboembolic disease following primary total knee arthroplasty: a randomized, multicenter, open-label, parallel-group comparison of enoxaparin and warfarin. J Bone Joint Surg Am 2001; 83-A:900–906.
- Eriksson BI, Bauer KA, Lassen MR, Turpie AG, Steering Committee of the Pentasaccharide in Hip-Fracture Surgery Study. Fondaparinux compared with enoxaparin for the prevention of venous thromboembolism after hip-fracture surgery. N Engl J Med 2001; 345:1298–1304.
- Demers C, Marcoux S, Ginsberg JS, Laroche F, Cloutier R, Poulin J. Incidence of venographically proved deep vein thrombosis after knee arthroscopy. Arch Intern Med 1998; 158:47–50.
- Geerts WH, Heit JA, Clagett GP, et al. Prevention of venous thromboembolism. Chest 2001; 119(1 Suppl):132S–175S.
- Brotman DJ, Jaffer AK, Hurbanek JG, Morra N. Warfarin prophylaxis and venous thromboembolism in the first 5 days following hip and knee arthroplasty. Thromb Haemost 2004; 92:1012–1017.
- Eriksson BI, Lassen MR; Pentasaccharide in Hip-Fracture Surgery Plus Investigators. Duration of prophylaxis against venous thromboembolism with fondaparinux after hip fracture surgery: a multicenter, randomized, placebo-controlled, double-blind study. Arch Intern Med 2003; 163:1337–1342.
- The Botticelli Investigators. Late-breaking clinical trial: a dose-finding study of the oral direct factor Xa inhibitor apixaban in the treatment of patients with acute symptomatic deep vein thrombosis [abstract]. Presented at the 21st Congress of the International Society on Thrombosis and Haemostasis; July 2007; Geneva, Switzerland.
- Fisher WD, Eriksson BI, Bauer KA, et al. Rivaroxaban for thromboprophylaxis after orthopaedic surgery: pooled analysis of two studies. Thromb Haemost 2007; 97:931–937.
- Haas S. New oral Xa and IIa inhibitors: updates on clinical trial results. J Thromb Thrombolysis 2008; 25:52–60.
- Friedman RJ, Caprini JA, Comp PC, et al. Dabigatran etexilate vs enoxaparin in preventing venous thromboembolism following total knee arthroplasty. Presented at: 2007 Congress of the International Society on Thrombosis and Haemostasis; July 7–13, 2007; Geneva, Switzerland.
- Committee for Medicinal Products for Human Use summary of positive opinion for Pradaxa [news release]. London, UK: European Medicines Agency. January 24, 2008. http://www.emea.europa.eu/pdfs/human/ opinion/Pradaxa_3503008en.pdf. Accessed February 21, 2008.
- Goldhaber SZ, Grodstein F, Stampfer MJ. A prospective study of risk factors for pulmonary embolism in women. JAMA 1997; 277:642–645.
- Turpie AGG, Bauer KA, Eriksson BI, Lassen MR, for the Steering Committees of the Pentasaccharide Orthopedic Prophylaxis Studies. Fondaparinux vs enoxaparin for the prevention of venous thromboembolism in major orthopedic surgery. Arch Intern Med 2002; 162:1833–1840.
- Stein PD, Beemath A, Matta F, et al. Clinical characteristics of patients with acute pulmonary embolism: data from PIOPED II. Am J Med 2007; 120:871–879.
- Goldhaber SZ. Diagnosis of acute pulmonary embolism: always be vigilant. Am J Med 2007; 120:827–828.
- American Academy of Orthopaedic Surgeons Clinical Guideline on Prevention of Symptomatic Pulmonary Embolism in Patients Undergoing Total Hip or Knee Arthroplasty: Summary of Recommendations. http://www.aaos.org/Research/guidelines/PE_ summary.pdf. Accessed December 10, 2007.