User login
-
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]


FDA Approves First Engineered Cell Therapy for a Solid Tumor
Afami-cel — the first engineered cell therapy for a solid tumor — is indicated specifically for adults with unresectable or metastatic synovial sarcoma who have received prior chemotherapy, are positive for several human leukocyte antigens (HLAs), and whose tumors express melanoma-associated antigen A4, as determined by FDA-authorized companion diagnostic devices.
The single-dose treatment targets solid tumors expressing melanoma-associated antigen A4, a protein highly expressed in synovial sarcoma.
Synovial sarcoma is a rare form of cancer, which affects about 1000 people in the US each year. Malignant cells develop and form a tumor in soft tissues, often in the extremities.
“Adults with metastatic synovial sarcoma, a life-threatening form of cancer, often face limited treatment options in addition to the risk of cancer spread or recurrence,” Nicole Verdun, MD, director of the Office of Therapeutic Products in the FDA’s Center for Biologics Evaluation and Research, said in the agency press release announcing the approval. “Today’s approval represents a significant milestone in the development of an innovative, safe and effective therapy for patients with this rare but potentially fatal disease.”
T-cell receptor therapy, like chimeric antigen receptor (CAR) T-cell (CAR-T) therapy, involves altering patient T cells to fight cancer. While CAR-T therapy inserts an artificial receptor to target a specific surface protein on cancer cells, the T-cell receptor therapy modifies existing receptors to recognize an array of antigens on the surface of cancer cells — a promising strategy for targeting solid tumors.
The accelerated approval of afami-cel was based on the phase 2 SPEARHEAD-1 trial in 44 patients with synovial sarcoma who received a single infusion of the therapy. The trial had enrolled 52 patients, but 8 did not receive afami-cel, including 3 who died and 1 who withdrew.
According to the FDA announcement, the overall response rate was 43.2%, with a median time to response of 4.9 weeks. The median duration of response was 6 months (95% CI, 4.6 months to not reached). Among patients who responded, 39% had a duration of response of 12 months or longer.
“These results suggest that a one-time treatment with afami-cel has the potential to extend life while allowing responders to go off chemotherapy,” said lead investigator Sandra D’Angelo, MD, a sarcoma specialist at Memorial Sloan Kettering Cancer Center in New York City, in a company press release.
The prescribing information includes a boxed warning for serious or fatal cytokine release syndrome.
The most common nonlaboratory adverse reactions, occurring in at least 20% of patients, included cytokine release syndrome, nausea, vomiting, fatigue, infections, pyrexia, constipation, dyspnea, tachycardia, hypotension, diarrhea, and edema. The most common grade 3 or 4 laboratory abnormalities, occurring in at least 20% of patients, included decreased lymphocyte count, neutrophil count, white cell blood count, red blood cell, and platelet count.
The recommended dose is between 2.68x109 to 10x109 MAGE-A4 T-cell receptor–positive T-cells. The FDA notice specifies not using a leukodepleting filter or prophylactic systemic corticosteroids.
The list price for the one-time therapy is $727,000, according to Fierce Pharma.
A version of this article first appeared on Medscape.com.
Afami-cel — the first engineered cell therapy for a solid tumor — is indicated specifically for adults with unresectable or metastatic synovial sarcoma who have received prior chemotherapy, are positive for several human leukocyte antigens (HLAs), and whose tumors express melanoma-associated antigen A4, as determined by FDA-authorized companion diagnostic devices.
The single-dose treatment targets solid tumors expressing melanoma-associated antigen A4, a protein highly expressed in synovial sarcoma.
Synovial sarcoma is a rare form of cancer, which affects about 1000 people in the US each year. Malignant cells develop and form a tumor in soft tissues, often in the extremities.
“Adults with metastatic synovial sarcoma, a life-threatening form of cancer, often face limited treatment options in addition to the risk of cancer spread or recurrence,” Nicole Verdun, MD, director of the Office of Therapeutic Products in the FDA’s Center for Biologics Evaluation and Research, said in the agency press release announcing the approval. “Today’s approval represents a significant milestone in the development of an innovative, safe and effective therapy for patients with this rare but potentially fatal disease.”
T-cell receptor therapy, like chimeric antigen receptor (CAR) T-cell (CAR-T) therapy, involves altering patient T cells to fight cancer. While CAR-T therapy inserts an artificial receptor to target a specific surface protein on cancer cells, the T-cell receptor therapy modifies existing receptors to recognize an array of antigens on the surface of cancer cells — a promising strategy for targeting solid tumors.
The accelerated approval of afami-cel was based on the phase 2 SPEARHEAD-1 trial in 44 patients with synovial sarcoma who received a single infusion of the therapy. The trial had enrolled 52 patients, but 8 did not receive afami-cel, including 3 who died and 1 who withdrew.
According to the FDA announcement, the overall response rate was 43.2%, with a median time to response of 4.9 weeks. The median duration of response was 6 months (95% CI, 4.6 months to not reached). Among patients who responded, 39% had a duration of response of 12 months or longer.
“These results suggest that a one-time treatment with afami-cel has the potential to extend life while allowing responders to go off chemotherapy,” said lead investigator Sandra D’Angelo, MD, a sarcoma specialist at Memorial Sloan Kettering Cancer Center in New York City, in a company press release.
The prescribing information includes a boxed warning for serious or fatal cytokine release syndrome.
The most common nonlaboratory adverse reactions, occurring in at least 20% of patients, included cytokine release syndrome, nausea, vomiting, fatigue, infections, pyrexia, constipation, dyspnea, tachycardia, hypotension, diarrhea, and edema. The most common grade 3 or 4 laboratory abnormalities, occurring in at least 20% of patients, included decreased lymphocyte count, neutrophil count, white cell blood count, red blood cell, and platelet count.
The recommended dose is between 2.68x109 to 10x109 MAGE-A4 T-cell receptor–positive T-cells. The FDA notice specifies not using a leukodepleting filter or prophylactic systemic corticosteroids.
The list price for the one-time therapy is $727,000, according to Fierce Pharma.
A version of this article first appeared on Medscape.com.
Afami-cel — the first engineered cell therapy for a solid tumor — is indicated specifically for adults with unresectable or metastatic synovial sarcoma who have received prior chemotherapy, are positive for several human leukocyte antigens (HLAs), and whose tumors express melanoma-associated antigen A4, as determined by FDA-authorized companion diagnostic devices.
The single-dose treatment targets solid tumors expressing melanoma-associated antigen A4, a protein highly expressed in synovial sarcoma.
Synovial sarcoma is a rare form of cancer, which affects about 1000 people in the US each year. Malignant cells develop and form a tumor in soft tissues, often in the extremities.
“Adults with metastatic synovial sarcoma, a life-threatening form of cancer, often face limited treatment options in addition to the risk of cancer spread or recurrence,” Nicole Verdun, MD, director of the Office of Therapeutic Products in the FDA’s Center for Biologics Evaluation and Research, said in the agency press release announcing the approval. “Today’s approval represents a significant milestone in the development of an innovative, safe and effective therapy for patients with this rare but potentially fatal disease.”
T-cell receptor therapy, like chimeric antigen receptor (CAR) T-cell (CAR-T) therapy, involves altering patient T cells to fight cancer. While CAR-T therapy inserts an artificial receptor to target a specific surface protein on cancer cells, the T-cell receptor therapy modifies existing receptors to recognize an array of antigens on the surface of cancer cells — a promising strategy for targeting solid tumors.
The accelerated approval of afami-cel was based on the phase 2 SPEARHEAD-1 trial in 44 patients with synovial sarcoma who received a single infusion of the therapy. The trial had enrolled 52 patients, but 8 did not receive afami-cel, including 3 who died and 1 who withdrew.
According to the FDA announcement, the overall response rate was 43.2%, with a median time to response of 4.9 weeks. The median duration of response was 6 months (95% CI, 4.6 months to not reached). Among patients who responded, 39% had a duration of response of 12 months or longer.
“These results suggest that a one-time treatment with afami-cel has the potential to extend life while allowing responders to go off chemotherapy,” said lead investigator Sandra D’Angelo, MD, a sarcoma specialist at Memorial Sloan Kettering Cancer Center in New York City, in a company press release.
The prescribing information includes a boxed warning for serious or fatal cytokine release syndrome.
The most common nonlaboratory adverse reactions, occurring in at least 20% of patients, included cytokine release syndrome, nausea, vomiting, fatigue, infections, pyrexia, constipation, dyspnea, tachycardia, hypotension, diarrhea, and edema. The most common grade 3 or 4 laboratory abnormalities, occurring in at least 20% of patients, included decreased lymphocyte count, neutrophil count, white cell blood count, red blood cell, and platelet count.
The recommended dose is between 2.68x109 to 10x109 MAGE-A4 T-cell receptor–positive T-cells. The FDA notice specifies not using a leukodepleting filter or prophylactic systemic corticosteroids.
The list price for the one-time therapy is $727,000, according to Fierce Pharma.
A version of this article first appeared on Medscape.com.
Wearables May Confirm Sleep Disruption Impact on Chronic Disease
Rapid eye movement (REM) sleep, deep sleep, and sleep irregularity were significantly associated with increased risk for a range of chronic diseases, based on a new study of > 6000 individuals.
“Most of what we think we know about sleep patterns in adults comes from either self-report surveys, which are widely used but have all sorts of problems with over- and under-estimating sleep duration and quality, or single-night sleep studies,” corresponding author Evan L. Brittain, MD, of Vanderbilt University, Nashville, Tennessee, said in an interview.
The single-night study yields the highest quality data but is limited by extrapolating a single night’s sleep to represent habitual sleep patterns, which is often not the case, he said. In the current study, published in Nature Medicine, “we had a unique opportunity to understand sleep using a large cohort of individuals using wearable devices that measure sleep duration, quality, and variability. The All of Us Research Program is the first to link wearables data to the electronic health record at scale and allowed us to study long-term, real-world sleep behavior,” Dr. Brittain said.
The timing of the study is important because the American Heart Association now recognizes sleep as a key component of heart health, and public awareness of the value of sleep is increasing, he added.
The researchers reviewed objectively measured, longitudinal sleep data from 6785 adults who used commercial wearable devices (Fitbit) linked to electronic health record data in the All of Us Research Program. The median age of the participants was 50.2 years, 71% were women, and 84% self-identified as White individuals. The median period of sleep monitoring was 4.5 years.
REM sleep and deep sleep were inversely associated with the odds of incident heart rhythm and heart rate abnormalities. A higher percentage of deep sleep was associated with reduced odds of atrial fibrillation (OR, 0.87), major depressive disorder (OR, 0.93), and anxiety disorder (OR, 0.94).
Increased irregular sleep was significantly associated with increased odds of incident obesity (OR, 1.49), hyperlipidemia (OR, 1.39), and hypertension (OR, 1.56), as well as major depressive disorder (OR, 1.75), anxiety disorder (OR, 1.55), and bipolar disorder (OR, 2.27).
The researchers also identified J-shaped associations between average daily sleep duration and hypertension (P for nonlinearity = .003), as well as major depressive disorder and generalized anxiety disorder (both P < .001).
The study was limited by several factors including the relatively young, White, and female study population. However, the results illustrate how sleep stages, duration, and regularity are associated with chronic disease development, and may inform evidence-based recommendations on healthy sleeping habits, the researchers wrote.
Findings Support Need for Sleep Consistency
“The biggest surprise for me was the impact of sleep variability of health,” Dr. Brittain told this news organization. “The more your sleep duration varies, the higher your risk of numerous chronic diseases across the entire spectrum of organ systems. Sleep duration and quality were also important but that was less surprising,” he said.
The clinical implications of the findings are that sleep duration, quality, and variability are all important, said Dr. Brittain. “To me, the easiest finding to translate into the clinic is the importance of reducing the variability of sleep duration as much as possible,” he said. For patients, that means explaining that they need to go to sleep and wake up at roughly the same time night to night, he said.
“Commercial wearable devices are not perfect compared with research grade devices, but our study showed that they nonetheless collect clinically relevant information,” Dr. Brittain added. “For patients who own a device, I have adopted the practice of reviewing my patients’ sleep and activity data which gives objective insight into behavior that is not always accurate through routine questioning,” he said.
As for other limitations, “Our cohort was limited to individuals who already owned a Fitbit; not surprisingly, these individuals differ from a random sample of the community in important ways, both demographic and behavioral, and our findings need to be validated in a more diverse population,” said Dr. Brittain.
Looking ahead, “we are interested in using commercial devices as a tool for sleep interventions to test the impact of improving sleep hygiene on chronic disease incidence, severity, and progression,” he said.
Device Data Will Evolve to Inform Patient Care
“With the increasing use of commercial wearable devices, it is crucial to identify and understand the data they can collect,” said Arianne K. Baldomero, MD, a pulmonologist and assistant professor of medicine at the University of Minnesota, Minneapolis, in an interview. “This study specifically analyzed sleep data from Fitbit devices among participants in the All of Us Research Program to assess sleep patterns and their association with chronic disease risk,” said Dr. Baldomero, who was not involved in the study.
The significant relationships between sleep patterns and risk for chronic diseases were not surprising, said Dr. Baldomero. The findings of an association between shorter sleep duration and greater sleep irregularity with obesity and sleep apnea validated previous studies in large-scale population surveys, she said. Findings from the current study also reflect data from the literature on sleep duration associated with hypertension, major depressive disorder, and generalized anxiety findings, she added.
“This study reinforces the importance of adequate sleep, typically around 7 hours per night, and suggests that insufficient or poor-quality sleep may be associated with chronic diseases,” Dr. Baldomero told this news organization. “Pulmonologists should remain vigilant about sleep-related issues, and consider further investigation and referrals to sleep specialty clinics for patients suspected of having sleep disturbances,” she said.
“What remains unclear is whether abnormal sleep patterns are a cause or an effect of chronic diseases,” Dr. Baldomero noted. “Additionally, it is essential to ensure that these devices accurately capture sleep patterns and continue to validate their data against gold standard measures of sleep disturbances,” she said.
The study was based on work that was partially funded by an unrestricted gift from Google, and the study itself was supported by National Institutes of Health. Dr. Brittain disclosed received research funds unrelated to this work from United Therapeutics. Dr. Baldomero had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Rapid eye movement (REM) sleep, deep sleep, and sleep irregularity were significantly associated with increased risk for a range of chronic diseases, based on a new study of > 6000 individuals.
“Most of what we think we know about sleep patterns in adults comes from either self-report surveys, which are widely used but have all sorts of problems with over- and under-estimating sleep duration and quality, or single-night sleep studies,” corresponding author Evan L. Brittain, MD, of Vanderbilt University, Nashville, Tennessee, said in an interview.
The single-night study yields the highest quality data but is limited by extrapolating a single night’s sleep to represent habitual sleep patterns, which is often not the case, he said. In the current study, published in Nature Medicine, “we had a unique opportunity to understand sleep using a large cohort of individuals using wearable devices that measure sleep duration, quality, and variability. The All of Us Research Program is the first to link wearables data to the electronic health record at scale and allowed us to study long-term, real-world sleep behavior,” Dr. Brittain said.
The timing of the study is important because the American Heart Association now recognizes sleep as a key component of heart health, and public awareness of the value of sleep is increasing, he added.
The researchers reviewed objectively measured, longitudinal sleep data from 6785 adults who used commercial wearable devices (Fitbit) linked to electronic health record data in the All of Us Research Program. The median age of the participants was 50.2 years, 71% were women, and 84% self-identified as White individuals. The median period of sleep monitoring was 4.5 years.
REM sleep and deep sleep were inversely associated with the odds of incident heart rhythm and heart rate abnormalities. A higher percentage of deep sleep was associated with reduced odds of atrial fibrillation (OR, 0.87), major depressive disorder (OR, 0.93), and anxiety disorder (OR, 0.94).
Increased irregular sleep was significantly associated with increased odds of incident obesity (OR, 1.49), hyperlipidemia (OR, 1.39), and hypertension (OR, 1.56), as well as major depressive disorder (OR, 1.75), anxiety disorder (OR, 1.55), and bipolar disorder (OR, 2.27).
The researchers also identified J-shaped associations between average daily sleep duration and hypertension (P for nonlinearity = .003), as well as major depressive disorder and generalized anxiety disorder (both P < .001).
The study was limited by several factors including the relatively young, White, and female study population. However, the results illustrate how sleep stages, duration, and regularity are associated with chronic disease development, and may inform evidence-based recommendations on healthy sleeping habits, the researchers wrote.
Findings Support Need for Sleep Consistency
“The biggest surprise for me was the impact of sleep variability of health,” Dr. Brittain told this news organization. “The more your sleep duration varies, the higher your risk of numerous chronic diseases across the entire spectrum of organ systems. Sleep duration and quality were also important but that was less surprising,” he said.
The clinical implications of the findings are that sleep duration, quality, and variability are all important, said Dr. Brittain. “To me, the easiest finding to translate into the clinic is the importance of reducing the variability of sleep duration as much as possible,” he said. For patients, that means explaining that they need to go to sleep and wake up at roughly the same time night to night, he said.
“Commercial wearable devices are not perfect compared with research grade devices, but our study showed that they nonetheless collect clinically relevant information,” Dr. Brittain added. “For patients who own a device, I have adopted the practice of reviewing my patients’ sleep and activity data which gives objective insight into behavior that is not always accurate through routine questioning,” he said.
As for other limitations, “Our cohort was limited to individuals who already owned a Fitbit; not surprisingly, these individuals differ from a random sample of the community in important ways, both demographic and behavioral, and our findings need to be validated in a more diverse population,” said Dr. Brittain.
Looking ahead, “we are interested in using commercial devices as a tool for sleep interventions to test the impact of improving sleep hygiene on chronic disease incidence, severity, and progression,” he said.
Device Data Will Evolve to Inform Patient Care
“With the increasing use of commercial wearable devices, it is crucial to identify and understand the data they can collect,” said Arianne K. Baldomero, MD, a pulmonologist and assistant professor of medicine at the University of Minnesota, Minneapolis, in an interview. “This study specifically analyzed sleep data from Fitbit devices among participants in the All of Us Research Program to assess sleep patterns and their association with chronic disease risk,” said Dr. Baldomero, who was not involved in the study.
The significant relationships between sleep patterns and risk for chronic diseases were not surprising, said Dr. Baldomero. The findings of an association between shorter sleep duration and greater sleep irregularity with obesity and sleep apnea validated previous studies in large-scale population surveys, she said. Findings from the current study also reflect data from the literature on sleep duration associated with hypertension, major depressive disorder, and generalized anxiety findings, she added.
“This study reinforces the importance of adequate sleep, typically around 7 hours per night, and suggests that insufficient or poor-quality sleep may be associated with chronic diseases,” Dr. Baldomero told this news organization. “Pulmonologists should remain vigilant about sleep-related issues, and consider further investigation and referrals to sleep specialty clinics for patients suspected of having sleep disturbances,” she said.
“What remains unclear is whether abnormal sleep patterns are a cause or an effect of chronic diseases,” Dr. Baldomero noted. “Additionally, it is essential to ensure that these devices accurately capture sleep patterns and continue to validate their data against gold standard measures of sleep disturbances,” she said.
The study was based on work that was partially funded by an unrestricted gift from Google, and the study itself was supported by National Institutes of Health. Dr. Brittain disclosed received research funds unrelated to this work from United Therapeutics. Dr. Baldomero had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Rapid eye movement (REM) sleep, deep sleep, and sleep irregularity were significantly associated with increased risk for a range of chronic diseases, based on a new study of > 6000 individuals.
“Most of what we think we know about sleep patterns in adults comes from either self-report surveys, which are widely used but have all sorts of problems with over- and under-estimating sleep duration and quality, or single-night sleep studies,” corresponding author Evan L. Brittain, MD, of Vanderbilt University, Nashville, Tennessee, said in an interview.
The single-night study yields the highest quality data but is limited by extrapolating a single night’s sleep to represent habitual sleep patterns, which is often not the case, he said. In the current study, published in Nature Medicine, “we had a unique opportunity to understand sleep using a large cohort of individuals using wearable devices that measure sleep duration, quality, and variability. The All of Us Research Program is the first to link wearables data to the electronic health record at scale and allowed us to study long-term, real-world sleep behavior,” Dr. Brittain said.
The timing of the study is important because the American Heart Association now recognizes sleep as a key component of heart health, and public awareness of the value of sleep is increasing, he added.
The researchers reviewed objectively measured, longitudinal sleep data from 6785 adults who used commercial wearable devices (Fitbit) linked to electronic health record data in the All of Us Research Program. The median age of the participants was 50.2 years, 71% were women, and 84% self-identified as White individuals. The median period of sleep monitoring was 4.5 years.
REM sleep and deep sleep were inversely associated with the odds of incident heart rhythm and heart rate abnormalities. A higher percentage of deep sleep was associated with reduced odds of atrial fibrillation (OR, 0.87), major depressive disorder (OR, 0.93), and anxiety disorder (OR, 0.94).
Increased irregular sleep was significantly associated with increased odds of incident obesity (OR, 1.49), hyperlipidemia (OR, 1.39), and hypertension (OR, 1.56), as well as major depressive disorder (OR, 1.75), anxiety disorder (OR, 1.55), and bipolar disorder (OR, 2.27).
The researchers also identified J-shaped associations between average daily sleep duration and hypertension (P for nonlinearity = .003), as well as major depressive disorder and generalized anxiety disorder (both P < .001).
The study was limited by several factors including the relatively young, White, and female study population. However, the results illustrate how sleep stages, duration, and regularity are associated with chronic disease development, and may inform evidence-based recommendations on healthy sleeping habits, the researchers wrote.
Findings Support Need for Sleep Consistency
“The biggest surprise for me was the impact of sleep variability of health,” Dr. Brittain told this news organization. “The more your sleep duration varies, the higher your risk of numerous chronic diseases across the entire spectrum of organ systems. Sleep duration and quality were also important but that was less surprising,” he said.
The clinical implications of the findings are that sleep duration, quality, and variability are all important, said Dr. Brittain. “To me, the easiest finding to translate into the clinic is the importance of reducing the variability of sleep duration as much as possible,” he said. For patients, that means explaining that they need to go to sleep and wake up at roughly the same time night to night, he said.
“Commercial wearable devices are not perfect compared with research grade devices, but our study showed that they nonetheless collect clinically relevant information,” Dr. Brittain added. “For patients who own a device, I have adopted the practice of reviewing my patients’ sleep and activity data which gives objective insight into behavior that is not always accurate through routine questioning,” he said.
As for other limitations, “Our cohort was limited to individuals who already owned a Fitbit; not surprisingly, these individuals differ from a random sample of the community in important ways, both demographic and behavioral, and our findings need to be validated in a more diverse population,” said Dr. Brittain.
Looking ahead, “we are interested in using commercial devices as a tool for sleep interventions to test the impact of improving sleep hygiene on chronic disease incidence, severity, and progression,” he said.
Device Data Will Evolve to Inform Patient Care
“With the increasing use of commercial wearable devices, it is crucial to identify and understand the data they can collect,” said Arianne K. Baldomero, MD, a pulmonologist and assistant professor of medicine at the University of Minnesota, Minneapolis, in an interview. “This study specifically analyzed sleep data from Fitbit devices among participants in the All of Us Research Program to assess sleep patterns and their association with chronic disease risk,” said Dr. Baldomero, who was not involved in the study.
The significant relationships between sleep patterns and risk for chronic diseases were not surprising, said Dr. Baldomero. The findings of an association between shorter sleep duration and greater sleep irregularity with obesity and sleep apnea validated previous studies in large-scale population surveys, she said. Findings from the current study also reflect data from the literature on sleep duration associated with hypertension, major depressive disorder, and generalized anxiety findings, she added.
“This study reinforces the importance of adequate sleep, typically around 7 hours per night, and suggests that insufficient or poor-quality sleep may be associated with chronic diseases,” Dr. Baldomero told this news organization. “Pulmonologists should remain vigilant about sleep-related issues, and consider further investigation and referrals to sleep specialty clinics for patients suspected of having sleep disturbances,” she said.
“What remains unclear is whether abnormal sleep patterns are a cause or an effect of chronic diseases,” Dr. Baldomero noted. “Additionally, it is essential to ensure that these devices accurately capture sleep patterns and continue to validate their data against gold standard measures of sleep disturbances,” she said.
The study was based on work that was partially funded by an unrestricted gift from Google, and the study itself was supported by National Institutes of Health. Dr. Brittain disclosed received research funds unrelated to this work from United Therapeutics. Dr. Baldomero had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Ozempic Curbs Hunger – And Not Just for Food
This transcript has been edited for clarity.
If you’ve been paying attention only to the headlines, when you think of “Ozempic” you’ll think of a few things: a blockbuster weight loss drug or the tip of the spear of a completely new industry — why not? A drug so popular that the people it was invented for (those with diabetes) can’t even get it.
Ozempic and other GLP-1 receptor agonists are undeniable game changers. Insofar as obesity is the number-one public health risk in the United States, antiobesity drugs hold immense promise even if all they do is reduce obesity.
In 2023, an article in Scientific Reports presented data suggesting that people on Ozempic might be reducing their alcohol intake, not just their total calories.
A 2024 article in Molecular Psychiatry found that the drug might positively impact cannabis use disorder. An article from Brain Sciences suggests that the drug reduces compulsive shopping.
A picture is starting to form, a picture that suggests these drugs curb hunger both literally and figuratively. That GLP-1 receptor agonists like Ozempic and Mounjaro are fundamentally anticonsumption drugs. In a society that — some would argue — is plagued by overconsumption, these drugs might be just what the doctor ordered.
If only they could stop people from smoking.
Oh, wait — they can.
At least it seems they can, based on a new study appearing in Annals of Internal Medicine.
Before we get too excited, this is not a randomized trial. There actually was a small randomized trial of exenatide (Byetta), which is in the same class as Ozempic but probably a bit less potent, with promising results for smoking cessation. 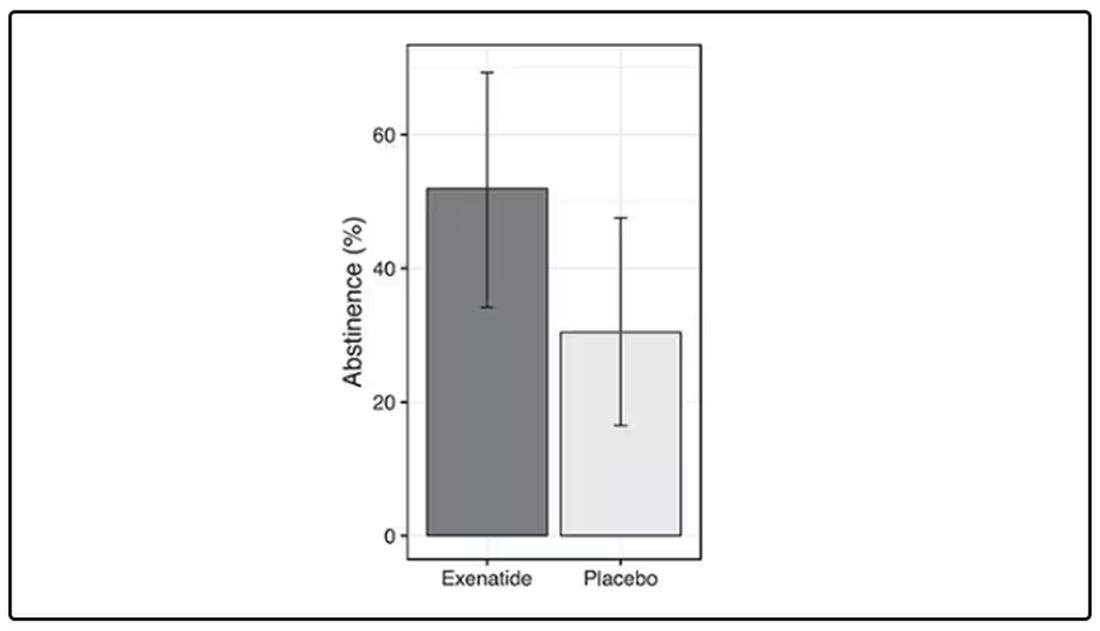
But Byetta is the weaker drug in this class; the market leader is Ozempic. So how can you figure out whether Ozempic can reduce smoking without doing a huge and expensive randomized trial? You can do what Nora Volkow and colleagues from the National Institute on Drug Abuse did: a target trial emulation study.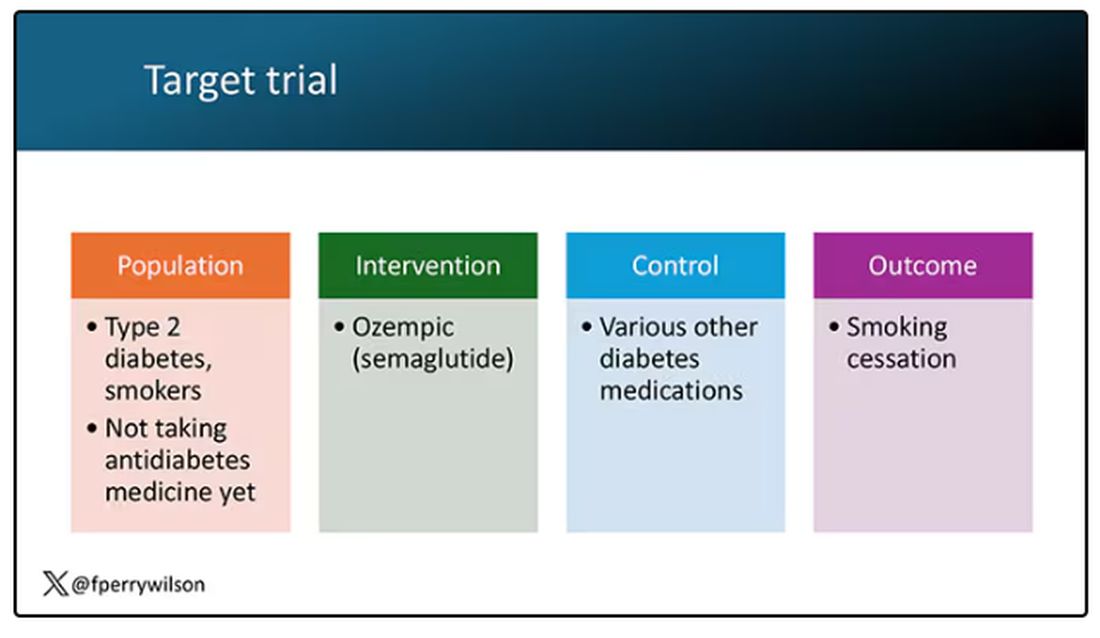
A target trial emulation study is more or less what it sounds like. First, you decide what your dream randomized controlled trial would be and you plan it all out in great detail. You define the population you would recruit, with all the relevant inclusion and exclusion criteria. You define the intervention and the control, and you define the outcome.
But you don’t actually do the trial. You could if someone would lend you $10-$50 million, but assuming you don’t have that lying around, you do the next best thing, which is to dig into a medical record database to find all the people who would be eligible for your imaginary trial. And you analyze them.
The authors wanted to study the effect of Ozempic on smoking among people with diabetes; that’s why all the comparator agents are antidiabetes drugs. They figured out whether these folks were smoking on the basis of a medical record diagnosis of tobacco use disorder before they started one of the drugs of interest. This code is fairly specific: If a patient has it, you can be pretty sure they are smoking. But it’s not very sensitive; not every smoker has this diagnostic code. This is an age-old limitation of using EHR data instead of asking patients, but it’s part of the tradeoff for not having to spend $50 million.
After applying all those inclusion and exclusion criteria, they have a defined population who could be in their dream trial. And, as luck would have it, some of those people really were treated with Ozempic and some really were treated with those other agents. Although decisions about what to prescribe were not randomized, the authors account for this confounding-by-indication using propensity-score matching. You can find a little explainer on propensity-score matching in an earlier column here. 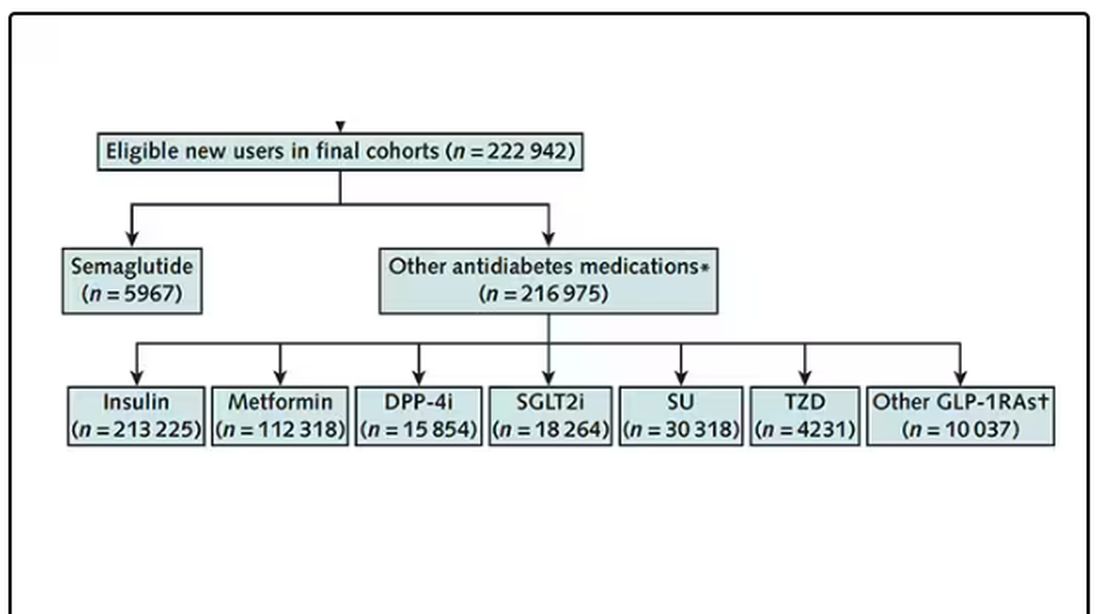
It’s easy enough, using the EHR, to figure out who has diabetes and who got which drug. But how do you know who quit smoking? Remember, everyone had a diagnosis code for tobacco use disorder prior to starting Ozempic or a comparator drug. The authors decided that if the patient had a medical visit where someone again coded tobacco-use disorder, they were still smoking. If someone prescribed smoking cessation meds like a nicotine patch or varenicline, they were obviously still smoking. If someone billed for tobacco-cessation counseling, the patient is still smoking. We’ll get back to the implications of this outcome definition in a minute.
Let’s talk about the results, which are pretty intriguing. 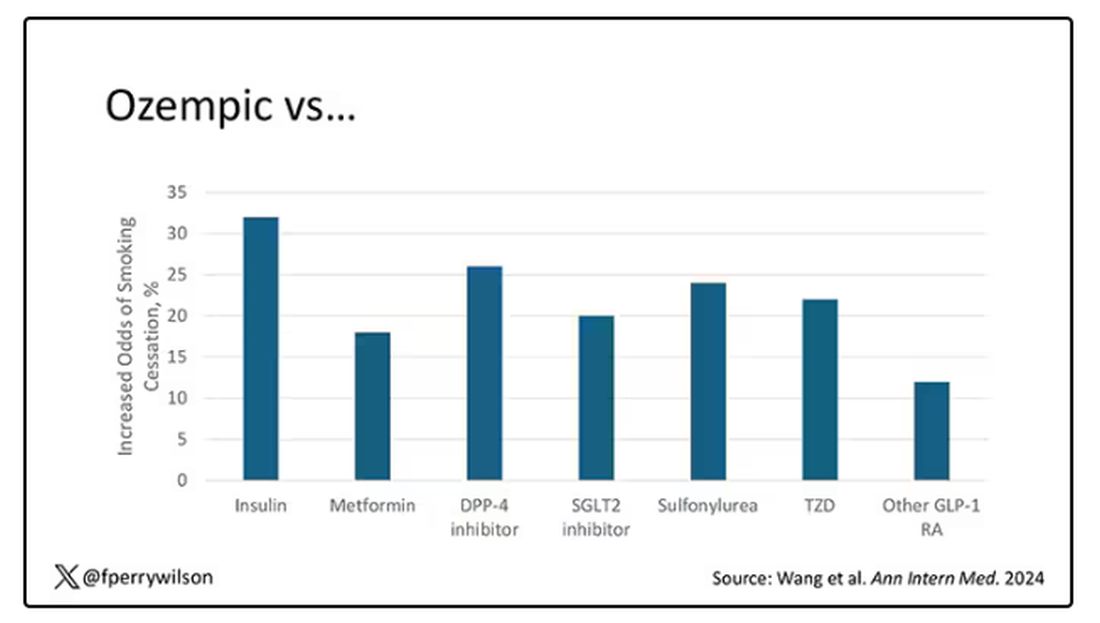
When Ozempic is compared with insulin among smokers with diabetes, those on Ozempic were about 30% more likely to quit smoking. They were about 18% more likely to quit smoking than those who took metformin. They were even slightly more likely to quit smoking than those on other GLP-1 receptor antagonists, though I should note that Mounjaro, which is probably the more potent GLP-1 drug in terms of weight loss, was not among the comparators.
This is pretty impressive for a drug that was not designed to be a smoking cessation drug. It speaks to this emerging idea that these drugs do more than curb appetite by slowing down gastric emptying or something. They work in the brain, modulating some of the reward circuitry that keeps us locked into our bad habits.
There are, of course, some caveats. As I pointed out, this study captured the idea of “still smoking” through the use of administrative codes in the EHR and prescription of smoking cessation aids. You could see similar results if taking Ozempic makes people less likely to address their smoking at all; maybe they shut down the doctor before they even talk about it, or there is too much to discuss during these visits to even get to the subject of smoking. You could also see results like this if people taking Ozempic had fewer visits overall, but the authors showed that that, at least, was not the case.
I’m inclined to believe that this effect is real, simply because we keep seeing signals from multiple sources. If that turns out to be the case, these new “weight loss” drugs may prove to be much more than that; they may turn out to be the drugs that can finally save us from ourselves.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
If you’ve been paying attention only to the headlines, when you think of “Ozempic” you’ll think of a few things: a blockbuster weight loss drug or the tip of the spear of a completely new industry — why not? A drug so popular that the people it was invented for (those with diabetes) can’t even get it.
Ozempic and other GLP-1 receptor agonists are undeniable game changers. Insofar as obesity is the number-one public health risk in the United States, antiobesity drugs hold immense promise even if all they do is reduce obesity.
In 2023, an article in Scientific Reports presented data suggesting that people on Ozempic might be reducing their alcohol intake, not just their total calories.
A 2024 article in Molecular Psychiatry found that the drug might positively impact cannabis use disorder. An article from Brain Sciences suggests that the drug reduces compulsive shopping.
A picture is starting to form, a picture that suggests these drugs curb hunger both literally and figuratively. That GLP-1 receptor agonists like Ozempic and Mounjaro are fundamentally anticonsumption drugs. In a society that — some would argue — is plagued by overconsumption, these drugs might be just what the doctor ordered.
If only they could stop people from smoking.
Oh, wait — they can.
At least it seems they can, based on a new study appearing in Annals of Internal Medicine.
Before we get too excited, this is not a randomized trial. There actually was a small randomized trial of exenatide (Byetta), which is in the same class as Ozempic but probably a bit less potent, with promising results for smoking cessation. 
But Byetta is the weaker drug in this class; the market leader is Ozempic. So how can you figure out whether Ozempic can reduce smoking without doing a huge and expensive randomized trial? You can do what Nora Volkow and colleagues from the National Institute on Drug Abuse did: a target trial emulation study.
A target trial emulation study is more or less what it sounds like. First, you decide what your dream randomized controlled trial would be and you plan it all out in great detail. You define the population you would recruit, with all the relevant inclusion and exclusion criteria. You define the intervention and the control, and you define the outcome.
But you don’t actually do the trial. You could if someone would lend you $10-$50 million, but assuming you don’t have that lying around, you do the next best thing, which is to dig into a medical record database to find all the people who would be eligible for your imaginary trial. And you analyze them.
The authors wanted to study the effect of Ozempic on smoking among people with diabetes; that’s why all the comparator agents are antidiabetes drugs. They figured out whether these folks were smoking on the basis of a medical record diagnosis of tobacco use disorder before they started one of the drugs of interest. This code is fairly specific: If a patient has it, you can be pretty sure they are smoking. But it’s not very sensitive; not every smoker has this diagnostic code. This is an age-old limitation of using EHR data instead of asking patients, but it’s part of the tradeoff for not having to spend $50 million.
After applying all those inclusion and exclusion criteria, they have a defined population who could be in their dream trial. And, as luck would have it, some of those people really were treated with Ozempic and some really were treated with those other agents. Although decisions about what to prescribe were not randomized, the authors account for this confounding-by-indication using propensity-score matching. You can find a little explainer on propensity-score matching in an earlier column here. 
It’s easy enough, using the EHR, to figure out who has diabetes and who got which drug. But how do you know who quit smoking? Remember, everyone had a diagnosis code for tobacco use disorder prior to starting Ozempic or a comparator drug. The authors decided that if the patient had a medical visit where someone again coded tobacco-use disorder, they were still smoking. If someone prescribed smoking cessation meds like a nicotine patch or varenicline, they were obviously still smoking. If someone billed for tobacco-cessation counseling, the patient is still smoking. We’ll get back to the implications of this outcome definition in a minute.
Let’s talk about the results, which are pretty intriguing. 
When Ozempic is compared with insulin among smokers with diabetes, those on Ozempic were about 30% more likely to quit smoking. They were about 18% more likely to quit smoking than those who took metformin. They were even slightly more likely to quit smoking than those on other GLP-1 receptor antagonists, though I should note that Mounjaro, which is probably the more potent GLP-1 drug in terms of weight loss, was not among the comparators.
This is pretty impressive for a drug that was not designed to be a smoking cessation drug. It speaks to this emerging idea that these drugs do more than curb appetite by slowing down gastric emptying or something. They work in the brain, modulating some of the reward circuitry that keeps us locked into our bad habits.
There are, of course, some caveats. As I pointed out, this study captured the idea of “still smoking” through the use of administrative codes in the EHR and prescription of smoking cessation aids. You could see similar results if taking Ozempic makes people less likely to address their smoking at all; maybe they shut down the doctor before they even talk about it, or there is too much to discuss during these visits to even get to the subject of smoking. You could also see results like this if people taking Ozempic had fewer visits overall, but the authors showed that that, at least, was not the case.
I’m inclined to believe that this effect is real, simply because we keep seeing signals from multiple sources. If that turns out to be the case, these new “weight loss” drugs may prove to be much more than that; they may turn out to be the drugs that can finally save us from ourselves.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
If you’ve been paying attention only to the headlines, when you think of “Ozempic” you’ll think of a few things: a blockbuster weight loss drug or the tip of the spear of a completely new industry — why not? A drug so popular that the people it was invented for (those with diabetes) can’t even get it.
Ozempic and other GLP-1 receptor agonists are undeniable game changers. Insofar as obesity is the number-one public health risk in the United States, antiobesity drugs hold immense promise even if all they do is reduce obesity.
In 2023, an article in Scientific Reports presented data suggesting that people on Ozempic might be reducing their alcohol intake, not just their total calories.
A 2024 article in Molecular Psychiatry found that the drug might positively impact cannabis use disorder. An article from Brain Sciences suggests that the drug reduces compulsive shopping.
A picture is starting to form, a picture that suggests these drugs curb hunger both literally and figuratively. That GLP-1 receptor agonists like Ozempic and Mounjaro are fundamentally anticonsumption drugs. In a society that — some would argue — is plagued by overconsumption, these drugs might be just what the doctor ordered.
If only they could stop people from smoking.
Oh, wait — they can.
At least it seems they can, based on a new study appearing in Annals of Internal Medicine.
Before we get too excited, this is not a randomized trial. There actually was a small randomized trial of exenatide (Byetta), which is in the same class as Ozempic but probably a bit less potent, with promising results for smoking cessation. 
But Byetta is the weaker drug in this class; the market leader is Ozempic. So how can you figure out whether Ozempic can reduce smoking without doing a huge and expensive randomized trial? You can do what Nora Volkow and colleagues from the National Institute on Drug Abuse did: a target trial emulation study.
A target trial emulation study is more or less what it sounds like. First, you decide what your dream randomized controlled trial would be and you plan it all out in great detail. You define the population you would recruit, with all the relevant inclusion and exclusion criteria. You define the intervention and the control, and you define the outcome.
But you don’t actually do the trial. You could if someone would lend you $10-$50 million, but assuming you don’t have that lying around, you do the next best thing, which is to dig into a medical record database to find all the people who would be eligible for your imaginary trial. And you analyze them.
The authors wanted to study the effect of Ozempic on smoking among people with diabetes; that’s why all the comparator agents are antidiabetes drugs. They figured out whether these folks were smoking on the basis of a medical record diagnosis of tobacco use disorder before they started one of the drugs of interest. This code is fairly specific: If a patient has it, you can be pretty sure they are smoking. But it’s not very sensitive; not every smoker has this diagnostic code. This is an age-old limitation of using EHR data instead of asking patients, but it’s part of the tradeoff for not having to spend $50 million.
After applying all those inclusion and exclusion criteria, they have a defined population who could be in their dream trial. And, as luck would have it, some of those people really were treated with Ozempic and some really were treated with those other agents. Although decisions about what to prescribe were not randomized, the authors account for this confounding-by-indication using propensity-score matching. You can find a little explainer on propensity-score matching in an earlier column here. 
It’s easy enough, using the EHR, to figure out who has diabetes and who got which drug. But how do you know who quit smoking? Remember, everyone had a diagnosis code for tobacco use disorder prior to starting Ozempic or a comparator drug. The authors decided that if the patient had a medical visit where someone again coded tobacco-use disorder, they were still smoking. If someone prescribed smoking cessation meds like a nicotine patch or varenicline, they were obviously still smoking. If someone billed for tobacco-cessation counseling, the patient is still smoking. We’ll get back to the implications of this outcome definition in a minute.
Let’s talk about the results, which are pretty intriguing. 
When Ozempic is compared with insulin among smokers with diabetes, those on Ozempic were about 30% more likely to quit smoking. They were about 18% more likely to quit smoking than those who took metformin. They were even slightly more likely to quit smoking than those on other GLP-1 receptor antagonists, though I should note that Mounjaro, which is probably the more potent GLP-1 drug in terms of weight loss, was not among the comparators.
This is pretty impressive for a drug that was not designed to be a smoking cessation drug. It speaks to this emerging idea that these drugs do more than curb appetite by slowing down gastric emptying or something. They work in the brain, modulating some of the reward circuitry that keeps us locked into our bad habits.
There are, of course, some caveats. As I pointed out, this study captured the idea of “still smoking” through the use of administrative codes in the EHR and prescription of smoking cessation aids. You could see similar results if taking Ozempic makes people less likely to address their smoking at all; maybe they shut down the doctor before they even talk about it, or there is too much to discuss during these visits to even get to the subject of smoking. You could also see results like this if people taking Ozempic had fewer visits overall, but the authors showed that that, at least, was not the case.
I’m inclined to believe that this effect is real, simply because we keep seeing signals from multiple sources. If that turns out to be the case, these new “weight loss” drugs may prove to be much more than that; they may turn out to be the drugs that can finally save us from ourselves.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Vasculopathy Can Vary in Patients With Idiopathic Pulmonary Arterial Hypertension
Approximately half of adults with idiopathic pulmonary arterial hypertension (IPAH) had nonplexiform vasculopathy characterized in part by severe pulmonary microvascular remodeling, based on data from 50 individuals.
The clinical phenotype of IPAH was historically described as a rapidly progressive rare disease in young women and characterized by plexiform lesions, wrote Esther J. Nossent, MD, of Amsterdam University Medical Centers, Amsterdam, the Netherlands, and colleagues. However, the patient population with IPAH has become older and predominantly men, and the nature of vascular phenotypes and histologic patterns in patients with contemporary IPAH has not been well studied, the researchers said.
In a cross-sectional study published in CHEST, the researchers reviewed lung histology data from 50 adults with IPAH that had been assessed by two experienced pathologists. The mean age of the patients was 52 years and 58% were women. Based on a histopathologic evaluation, 24 patients had nonplexiform vasculopathy (48%) and 26 had plexiform vasculopathy (52%). Notably, microvascular remodeling involving arterioles and venules was substantial in patients with nonplexiform vasculopathy but mild or absent in those with plexiform vasculopathy, the researchers wrote.
The researchers also compared the clinical characteristics of patients with plexiform vs nonplexiform vasculopathy. Hemodynamic parameters were similar in both patient groups. However, those with nonplexiform vasculopathy were significantly older than those with plexiform vasculopathy (60 years vs 44 years), were more likely to be men (67% vs 20%), and had a lower diffusing capacity of the lungs for carbon monoxide (DLCO) at diagnosis (all P < .001). Patients with nonplexiform vasculopathy also were significantly more likely than those with plexiform vasculopathy to have a history of smoking (P = .03). Genetic testing revealed no mutations in established PAH genes in the nonplexiform group.
Low DLCO has been associated with worse outcomes regardless of hemodynamic response, the researchers noted. In the current study, “a DLCO of < 45% almost perfectly identified patients with nonplexiform vasculopathy with prominent pulmonary microvascular disease,” they said.
The findings were limited by several factors, including the small study population and the higher frequency of surgical lung biopsies in the nonplexiform group vs the plexiform group, which is not part of the general workup of patients with IPAH, the researchers noted.
, they said. However, the results suggest that differences between patients with IPAH with plexiform vasculopathy and those with nonplexiform vasculopathy could ultimately inform targeted treatment strategies.
“Recognizing these clinical phenotypes allows revisiting current datasets to understand better the potential future clinical consequences of the vascular phenotypes for treatment response and clinical outcome,” the researchers concluded.
Findings May Inform More Targeted Therapy
“Any investigation that adds substantive insight into a complex disease that can translate into a better understanding of clinical patient phenotypes and eventually into improved treatments and patient outcomes has relevance at any time,” Paul Forfia, MD, professor of medicine at the Lewis Katz School of Medicine at Temple University, Philadelphia, said in an interview.
“There is focus on the antiproliferative forms of pulmonary arterial hypertension–specific therapy, and the results of the current study may have implications to these therapies,” said Dr. Forfia, who was not involved in the current study.
“In the current study, the investigators show that 48% of patients that were traditionally categorized as IPAH had a vascular phenotype that is not considered ‘typical’ or classic for IPAH,” Dr. Forfia told this news organization. “These findings highlight a significant heterogeneity of the pulmonary vascular phenotype within IPAH, which raises the question of whether the nonplexiform patient would be less responsive to the novel, antiproliferative forms of therapy,” he said.
The new findings are quite interesting but not surprising, Dr. Forfia said. “The World Symposia diagnostic groupings for pulmonary hypertension are a very important and necessary form of categorization and differentiation amongst forms of PH [pulmonary hypertension], and these groupings make a best attempt based on available evidence to separate patients of varying PH pathophysiology, both in terms of diagnosis and in how PH patients are treated,” he explained.
“However, clinical experts in PH have known that subphenotypes of PH pathophysiology exist within group I PAH, as well as in PH related to left heart disease (group 2), chronic respiratory disease (group 3), and chronic thromboembolic disease (group 4),” he said.
Findings from the current study reinforce the importance of clinical and physiological phenotyping of each patient, which can help in terms of therapy selection and in managing expectations in response to therapy, Dr. Forfia added.
“Perhaps the most evident and important clinical implication from the current study is to remind clinicians treating patients with PH that heterogeneity exists within the vascular phenotype and clinical makeup of patients even within the same type of PAH,” Dr. Forfia said. “With this insight, clinicians are more informed and thus more apt to consider nuances in the diagnosis, treatment, and expectations for treatment response within PAH,” he said.
Dr. Forfia also highlighted the potential implications of the association between cigarette smoking and the nonplexiform vascular phenotype. “This association was present in the absence of radiographic evidence of emphysema and raises the provocative notion that cigarette smoking may lead to pulmonary vascular abnormalities, perhaps even PAH, in patients without a diagnosis of emphysema,” he said.
“An important limitation from the current study is that the vascular phenotypes observed within their cohort of IPAH patients were obtained from histopathology specimens at the time of autopsy, explant at the time of lung transplantation, and surgical lung biopsy spanning over a 22-year period,” Dr. Forfia noted. Additional research is needed to explore how vascular phenotypic differences can be appreciated in the absence of histopathology and how these differences could impact therapy selection and patient outcomes, he said.
The study received no outside funding. Dr. Nossent disclosed receiving speaker fees from Janssen, MSD, and United Therapeutics/Ferrer and consulting fees from Janssen and United Therapeutics/Ferrer. Dr. Forfia had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Approximately half of adults with idiopathic pulmonary arterial hypertension (IPAH) had nonplexiform vasculopathy characterized in part by severe pulmonary microvascular remodeling, based on data from 50 individuals.
The clinical phenotype of IPAH was historically described as a rapidly progressive rare disease in young women and characterized by plexiform lesions, wrote Esther J. Nossent, MD, of Amsterdam University Medical Centers, Amsterdam, the Netherlands, and colleagues. However, the patient population with IPAH has become older and predominantly men, and the nature of vascular phenotypes and histologic patterns in patients with contemporary IPAH has not been well studied, the researchers said.
In a cross-sectional study published in CHEST, the researchers reviewed lung histology data from 50 adults with IPAH that had been assessed by two experienced pathologists. The mean age of the patients was 52 years and 58% were women. Based on a histopathologic evaluation, 24 patients had nonplexiform vasculopathy (48%) and 26 had plexiform vasculopathy (52%). Notably, microvascular remodeling involving arterioles and venules was substantial in patients with nonplexiform vasculopathy but mild or absent in those with plexiform vasculopathy, the researchers wrote.
The researchers also compared the clinical characteristics of patients with plexiform vs nonplexiform vasculopathy. Hemodynamic parameters were similar in both patient groups. However, those with nonplexiform vasculopathy were significantly older than those with plexiform vasculopathy (60 years vs 44 years), were more likely to be men (67% vs 20%), and had a lower diffusing capacity of the lungs for carbon monoxide (DLCO) at diagnosis (all P < .001). Patients with nonplexiform vasculopathy also were significantly more likely than those with plexiform vasculopathy to have a history of smoking (P = .03). Genetic testing revealed no mutations in established PAH genes in the nonplexiform group.
Low DLCO has been associated with worse outcomes regardless of hemodynamic response, the researchers noted. In the current study, “a DLCO of < 45% almost perfectly identified patients with nonplexiform vasculopathy with prominent pulmonary microvascular disease,” they said.
The findings were limited by several factors, including the small study population and the higher frequency of surgical lung biopsies in the nonplexiform group vs the plexiform group, which is not part of the general workup of patients with IPAH, the researchers noted.
, they said. However, the results suggest that differences between patients with IPAH with plexiform vasculopathy and those with nonplexiform vasculopathy could ultimately inform targeted treatment strategies.
“Recognizing these clinical phenotypes allows revisiting current datasets to understand better the potential future clinical consequences of the vascular phenotypes for treatment response and clinical outcome,” the researchers concluded.
Findings May Inform More Targeted Therapy
“Any investigation that adds substantive insight into a complex disease that can translate into a better understanding of clinical patient phenotypes and eventually into improved treatments and patient outcomes has relevance at any time,” Paul Forfia, MD, professor of medicine at the Lewis Katz School of Medicine at Temple University, Philadelphia, said in an interview.
“There is focus on the antiproliferative forms of pulmonary arterial hypertension–specific therapy, and the results of the current study may have implications to these therapies,” said Dr. Forfia, who was not involved in the current study.
“In the current study, the investigators show that 48% of patients that were traditionally categorized as IPAH had a vascular phenotype that is not considered ‘typical’ or classic for IPAH,” Dr. Forfia told this news organization. “These findings highlight a significant heterogeneity of the pulmonary vascular phenotype within IPAH, which raises the question of whether the nonplexiform patient would be less responsive to the novel, antiproliferative forms of therapy,” he said.
The new findings are quite interesting but not surprising, Dr. Forfia said. “The World Symposia diagnostic groupings for pulmonary hypertension are a very important and necessary form of categorization and differentiation amongst forms of PH [pulmonary hypertension], and these groupings make a best attempt based on available evidence to separate patients of varying PH pathophysiology, both in terms of diagnosis and in how PH patients are treated,” he explained.
“However, clinical experts in PH have known that subphenotypes of PH pathophysiology exist within group I PAH, as well as in PH related to left heart disease (group 2), chronic respiratory disease (group 3), and chronic thromboembolic disease (group 4),” he said.
Findings from the current study reinforce the importance of clinical and physiological phenotyping of each patient, which can help in terms of therapy selection and in managing expectations in response to therapy, Dr. Forfia added.
“Perhaps the most evident and important clinical implication from the current study is to remind clinicians treating patients with PH that heterogeneity exists within the vascular phenotype and clinical makeup of patients even within the same type of PAH,” Dr. Forfia said. “With this insight, clinicians are more informed and thus more apt to consider nuances in the diagnosis, treatment, and expectations for treatment response within PAH,” he said.
Dr. Forfia also highlighted the potential implications of the association between cigarette smoking and the nonplexiform vascular phenotype. “This association was present in the absence of radiographic evidence of emphysema and raises the provocative notion that cigarette smoking may lead to pulmonary vascular abnormalities, perhaps even PAH, in patients without a diagnosis of emphysema,” he said.
“An important limitation from the current study is that the vascular phenotypes observed within their cohort of IPAH patients were obtained from histopathology specimens at the time of autopsy, explant at the time of lung transplantation, and surgical lung biopsy spanning over a 22-year period,” Dr. Forfia noted. Additional research is needed to explore how vascular phenotypic differences can be appreciated in the absence of histopathology and how these differences could impact therapy selection and patient outcomes, he said.
The study received no outside funding. Dr. Nossent disclosed receiving speaker fees from Janssen, MSD, and United Therapeutics/Ferrer and consulting fees from Janssen and United Therapeutics/Ferrer. Dr. Forfia had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Approximately half of adults with idiopathic pulmonary arterial hypertension (IPAH) had nonplexiform vasculopathy characterized in part by severe pulmonary microvascular remodeling, based on data from 50 individuals.
The clinical phenotype of IPAH was historically described as a rapidly progressive rare disease in young women and characterized by plexiform lesions, wrote Esther J. Nossent, MD, of Amsterdam University Medical Centers, Amsterdam, the Netherlands, and colleagues. However, the patient population with IPAH has become older and predominantly men, and the nature of vascular phenotypes and histologic patterns in patients with contemporary IPAH has not been well studied, the researchers said.
In a cross-sectional study published in CHEST, the researchers reviewed lung histology data from 50 adults with IPAH that had been assessed by two experienced pathologists. The mean age of the patients was 52 years and 58% were women. Based on a histopathologic evaluation, 24 patients had nonplexiform vasculopathy (48%) and 26 had plexiform vasculopathy (52%). Notably, microvascular remodeling involving arterioles and venules was substantial in patients with nonplexiform vasculopathy but mild or absent in those with plexiform vasculopathy, the researchers wrote.
The researchers also compared the clinical characteristics of patients with plexiform vs nonplexiform vasculopathy. Hemodynamic parameters were similar in both patient groups. However, those with nonplexiform vasculopathy were significantly older than those with plexiform vasculopathy (60 years vs 44 years), were more likely to be men (67% vs 20%), and had a lower diffusing capacity of the lungs for carbon monoxide (DLCO) at diagnosis (all P < .001). Patients with nonplexiform vasculopathy also were significantly more likely than those with plexiform vasculopathy to have a history of smoking (P = .03). Genetic testing revealed no mutations in established PAH genes in the nonplexiform group.
Low DLCO has been associated with worse outcomes regardless of hemodynamic response, the researchers noted. In the current study, “a DLCO of < 45% almost perfectly identified patients with nonplexiform vasculopathy with prominent pulmonary microvascular disease,” they said.
The findings were limited by several factors, including the small study population and the higher frequency of surgical lung biopsies in the nonplexiform group vs the plexiform group, which is not part of the general workup of patients with IPAH, the researchers noted.
, they said. However, the results suggest that differences between patients with IPAH with plexiform vasculopathy and those with nonplexiform vasculopathy could ultimately inform targeted treatment strategies.
“Recognizing these clinical phenotypes allows revisiting current datasets to understand better the potential future clinical consequences of the vascular phenotypes for treatment response and clinical outcome,” the researchers concluded.
Findings May Inform More Targeted Therapy
“Any investigation that adds substantive insight into a complex disease that can translate into a better understanding of clinical patient phenotypes and eventually into improved treatments and patient outcomes has relevance at any time,” Paul Forfia, MD, professor of medicine at the Lewis Katz School of Medicine at Temple University, Philadelphia, said in an interview.
“There is focus on the antiproliferative forms of pulmonary arterial hypertension–specific therapy, and the results of the current study may have implications to these therapies,” said Dr. Forfia, who was not involved in the current study.
“In the current study, the investigators show that 48% of patients that were traditionally categorized as IPAH had a vascular phenotype that is not considered ‘typical’ or classic for IPAH,” Dr. Forfia told this news organization. “These findings highlight a significant heterogeneity of the pulmonary vascular phenotype within IPAH, which raises the question of whether the nonplexiform patient would be less responsive to the novel, antiproliferative forms of therapy,” he said.
The new findings are quite interesting but not surprising, Dr. Forfia said. “The World Symposia diagnostic groupings for pulmonary hypertension are a very important and necessary form of categorization and differentiation amongst forms of PH [pulmonary hypertension], and these groupings make a best attempt based on available evidence to separate patients of varying PH pathophysiology, both in terms of diagnosis and in how PH patients are treated,” he explained.
“However, clinical experts in PH have known that subphenotypes of PH pathophysiology exist within group I PAH, as well as in PH related to left heart disease (group 2), chronic respiratory disease (group 3), and chronic thromboembolic disease (group 4),” he said.
Findings from the current study reinforce the importance of clinical and physiological phenotyping of each patient, which can help in terms of therapy selection and in managing expectations in response to therapy, Dr. Forfia added.
“Perhaps the most evident and important clinical implication from the current study is to remind clinicians treating patients with PH that heterogeneity exists within the vascular phenotype and clinical makeup of patients even within the same type of PAH,” Dr. Forfia said. “With this insight, clinicians are more informed and thus more apt to consider nuances in the diagnosis, treatment, and expectations for treatment response within PAH,” he said.
Dr. Forfia also highlighted the potential implications of the association between cigarette smoking and the nonplexiform vascular phenotype. “This association was present in the absence of radiographic evidence of emphysema and raises the provocative notion that cigarette smoking may lead to pulmonary vascular abnormalities, perhaps even PAH, in patients without a diagnosis of emphysema,” he said.
“An important limitation from the current study is that the vascular phenotypes observed within their cohort of IPAH patients were obtained from histopathology specimens at the time of autopsy, explant at the time of lung transplantation, and surgical lung biopsy spanning over a 22-year period,” Dr. Forfia noted. Additional research is needed to explore how vascular phenotypic differences can be appreciated in the absence of histopathology and how these differences could impact therapy selection and patient outcomes, he said.
The study received no outside funding. Dr. Nossent disclosed receiving speaker fees from Janssen, MSD, and United Therapeutics/Ferrer and consulting fees from Janssen and United Therapeutics/Ferrer. Dr. Forfia had no financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
The Last 30 Days: How Oncologists’ Choices Affect End-of-Life Cancer Care
TOPLINE:
Patients treated by oncologists in the top quartile for end-of-life prescribing behavior were almost four and a half times more likely to receive end-of-life therapy than those treated by these specialists in the bottom quartile.
METHODOLOGY:
- Researchers analyzed data from the Surveillance, Epidemiology, and End Results (SEER)-Medicare database, focusing on patients who died of cancer between 2012 and 2017.
- A total of 17,609 patients with breast, lung, colorectal, or prostate cancer were included, treated by 960 oncologists across 388 practices.
- Patients were required to have had at least one systemic cancer therapy claim in the last 180 days of life, with the treating oncologist identified on the basis of the therapy claim closest to the time of death.
- The study used multilevel models to estimate oncologists’ rates of providing cancer therapy in the last 30 days of life, adjusting for patient characteristics and practice variation.
- Functional status was assessed on the basis of paid claims for durable medical equipment in the last 60 months of life, with scores categorized as 0, 1, ≥ 2, or unknown.
TAKEAWAY:
- Oncologists in the 95th percentile for high end-of-life prescribing behavior had a 45% adjusted rate of treating patients in the last 30 days of life, compared with 17% among those in the 5th percentile.
- Patients treated by high end-of-life prescribing oncologists had over four times higher odds of receiving systemic therapy in the last 30 days of life (odds ratio [OR], 4.42; 95% CI, 4.00-4.89).
- Higher end-of-life prescribing oncologists also had a higher proportion of patients hospitalized in the last 30 days of life than low prescribers (58% vs 51.9%).
- No significant association was found between oncologist prescribing behavior and patient race or ethnicity, except for Black patients who had lower odds of receiving treatment (OR, 0.77; P < .001).
IN PRACTICE:
“Given calls to rein in overutilization of end-of-life six to eight cancer therapies, our findings highlight an underappreciated area for further research: How treatment discontinuation before death is shaped by oncologists’ unique treatment propensities. Elucidating the reasons for this remarkable variability in oncologist treatment behavior could inform efforts to reduce end-of-life cancer treatment overutilization,” wrote the authors of the study.
SOURCE:
The study was led by Login S. George, PhD, Institute for Health, Health Care Policy and Aging Research, Rutgers University in New Brunswick, New Jersey. It was published online in Cancer.
LIMITATIONS:
The study’s reliance on SEER-Medicare data may limit the generalizability of the findings to patients with Medicare Advantage, private insurance, or Medicaid, as well as younger patients. The lack of data on patient preferences and other health characteristics could confound the results. The study focused on systemic therapies and may not be generalizable to other treatments such as clinical trial drugs, oral therapies, surgery, or radiation. The data from 2012 to 2017 may not reflect more recent trends in cancer treatment.
DISCLOSURES:
The study was supported by grants from the National Cancer Institute and the Rutgers Cancer Institute of New Jersey. George disclosed receiving grants from these organizations. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Patients treated by oncologists in the top quartile for end-of-life prescribing behavior were almost four and a half times more likely to receive end-of-life therapy than those treated by these specialists in the bottom quartile.
METHODOLOGY:
- Researchers analyzed data from the Surveillance, Epidemiology, and End Results (SEER)-Medicare database, focusing on patients who died of cancer between 2012 and 2017.
- A total of 17,609 patients with breast, lung, colorectal, or prostate cancer were included, treated by 960 oncologists across 388 practices.
- Patients were required to have had at least one systemic cancer therapy claim in the last 180 days of life, with the treating oncologist identified on the basis of the therapy claim closest to the time of death.
- The study used multilevel models to estimate oncologists’ rates of providing cancer therapy in the last 30 days of life, adjusting for patient characteristics and practice variation.
- Functional status was assessed on the basis of paid claims for durable medical equipment in the last 60 months of life, with scores categorized as 0, 1, ≥ 2, or unknown.
TAKEAWAY:
- Oncologists in the 95th percentile for high end-of-life prescribing behavior had a 45% adjusted rate of treating patients in the last 30 days of life, compared with 17% among those in the 5th percentile.
- Patients treated by high end-of-life prescribing oncologists had over four times higher odds of receiving systemic therapy in the last 30 days of life (odds ratio [OR], 4.42; 95% CI, 4.00-4.89).
- Higher end-of-life prescribing oncologists also had a higher proportion of patients hospitalized in the last 30 days of life than low prescribers (58% vs 51.9%).
- No significant association was found between oncologist prescribing behavior and patient race or ethnicity, except for Black patients who had lower odds of receiving treatment (OR, 0.77; P < .001).
IN PRACTICE:
“Given calls to rein in overutilization of end-of-life six to eight cancer therapies, our findings highlight an underappreciated area for further research: How treatment discontinuation before death is shaped by oncologists’ unique treatment propensities. Elucidating the reasons for this remarkable variability in oncologist treatment behavior could inform efforts to reduce end-of-life cancer treatment overutilization,” wrote the authors of the study.
SOURCE:
The study was led by Login S. George, PhD, Institute for Health, Health Care Policy and Aging Research, Rutgers University in New Brunswick, New Jersey. It was published online in Cancer.
LIMITATIONS:
The study’s reliance on SEER-Medicare data may limit the generalizability of the findings to patients with Medicare Advantage, private insurance, or Medicaid, as well as younger patients. The lack of data on patient preferences and other health characteristics could confound the results. The study focused on systemic therapies and may not be generalizable to other treatments such as clinical trial drugs, oral therapies, surgery, or radiation. The data from 2012 to 2017 may not reflect more recent trends in cancer treatment.
DISCLOSURES:
The study was supported by grants from the National Cancer Institute and the Rutgers Cancer Institute of New Jersey. George disclosed receiving grants from these organizations. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Patients treated by oncologists in the top quartile for end-of-life prescribing behavior were almost four and a half times more likely to receive end-of-life therapy than those treated by these specialists in the bottom quartile.
METHODOLOGY:
- Researchers analyzed data from the Surveillance, Epidemiology, and End Results (SEER)-Medicare database, focusing on patients who died of cancer between 2012 and 2017.
- A total of 17,609 patients with breast, lung, colorectal, or prostate cancer were included, treated by 960 oncologists across 388 practices.
- Patients were required to have had at least one systemic cancer therapy claim in the last 180 days of life, with the treating oncologist identified on the basis of the therapy claim closest to the time of death.
- The study used multilevel models to estimate oncologists’ rates of providing cancer therapy in the last 30 days of life, adjusting for patient characteristics and practice variation.
- Functional status was assessed on the basis of paid claims for durable medical equipment in the last 60 months of life, with scores categorized as 0, 1, ≥ 2, or unknown.
TAKEAWAY:
- Oncologists in the 95th percentile for high end-of-life prescribing behavior had a 45% adjusted rate of treating patients in the last 30 days of life, compared with 17% among those in the 5th percentile.
- Patients treated by high end-of-life prescribing oncologists had over four times higher odds of receiving systemic therapy in the last 30 days of life (odds ratio [OR], 4.42; 95% CI, 4.00-4.89).
- Higher end-of-life prescribing oncologists also had a higher proportion of patients hospitalized in the last 30 days of life than low prescribers (58% vs 51.9%).
- No significant association was found between oncologist prescribing behavior and patient race or ethnicity, except for Black patients who had lower odds of receiving treatment (OR, 0.77; P < .001).
IN PRACTICE:
“Given calls to rein in overutilization of end-of-life six to eight cancer therapies, our findings highlight an underappreciated area for further research: How treatment discontinuation before death is shaped by oncologists’ unique treatment propensities. Elucidating the reasons for this remarkable variability in oncologist treatment behavior could inform efforts to reduce end-of-life cancer treatment overutilization,” wrote the authors of the study.
SOURCE:
The study was led by Login S. George, PhD, Institute for Health, Health Care Policy and Aging Research, Rutgers University in New Brunswick, New Jersey. It was published online in Cancer.
LIMITATIONS:
The study’s reliance on SEER-Medicare data may limit the generalizability of the findings to patients with Medicare Advantage, private insurance, or Medicaid, as well as younger patients. The lack of data on patient preferences and other health characteristics could confound the results. The study focused on systemic therapies and may not be generalizable to other treatments such as clinical trial drugs, oral therapies, surgery, or radiation. The data from 2012 to 2017 may not reflect more recent trends in cancer treatment.
DISCLOSURES:
The study was supported by grants from the National Cancer Institute and the Rutgers Cancer Institute of New Jersey. George disclosed receiving grants from these organizations. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Ancient Viruses in Our DNA Hold Clues to Cancer Treatment
according to a fascinating new study in Science Advances. Targeting these viral remnants still lingering in our DNA could lead to more effective cancer treatment with fewer side effects, the researchers said.
The study “gives a better understanding of how gene regulation can be impacted by these ancient retroviral sequences,” said Dixie Mager, PhD, scientist emeritus at the Terry Fox Laboratory at the British Columbia Cancer Research Institute, Vancouver, British Columbia, Canada. (Mager was not involved in the study.)
Long thought to be “junk” DNA with no biologic function, “endogenous retroviruses,” which have mutated over time and lost their ability to create the virus, are now known to regulate genes — allowing some genes to turn on and off. Research in recent years suggests they may play a role in diseases like cancer.
But scientists weren’t exactly sure what that role was, said senior study author Edward Chuong, PhD, a genome biologist at the University of Colorado Boulder.
Most studies have looked at whether endogenous retroviruses code for proteins that influence cancer. But these ancient viral strands usually don’t code for proteins at all.
Dr. Chuong took a different approach. Inspired by scientists who’ve studied how viral remnants regulate positive processes (immunity, brain development, or placenta development), he and his team explored whether some might regulate genes that, once activated, help cancer thrive.
Borrowing from epigenomic analysis data (data on molecules that alter gene expression) for 21 cancers mapped by the Cancer Genome Atlas, the researchers identified 19 virus-derived DNA sequences that bind to regulatory proteins more in cancer cells than in healthy cells. All of these could potentially act as gene regulators that promote cancer.
The researchers homed in on one sequence, called LTR10, because it showed especially high activity in several cancers, including lung and colorectal cancer. This DNA segment comes from a virus that entered our ancestors’ genome 30 million years ago, and it’s activated in a third of colorectal cancers.
Using the gene editing technology clustered regularly interspaced short palindromic repeats (CRISPR), Dr. Chuong’s team silenced LTR10 in colorectal cancer cells, altering the gene sequence so it couldn’t bind to regulatory proteins. Doing so dampened the activity of nearby cancer-promoting genes.
“They still behaved like cancer cells,” Dr. Chuong said. But “it made the cancer cells more susceptible to radiation. That would imply that the presence of that viral ‘switch’ actually helped those cancer cells survive radiation therapy.”
Previously, two studies had found that viral regulators play a role in promoting two types of cancer: Leukemia and prostate cancer. The new study shows these two cases weren’t flukes. All 21 cancers they looked at had at least one of those 19 viral elements, presumably working as cancer enhancers.
The study also identified what activates LTR10 to make it promote cancer. The culprit is a regulator protein called mitogen-activated protein (MAP) kinase, which is overactivated in about 40% of all human cancers.
Some cancer drugs — MAP kinase inhibitors — already target MAP kinase, and they’re often the first ones prescribed when a patient is diagnosed with cancer, Dr. Chuong said. As with many cancer treatments, doctors don’t know why they work, just that they do.
“By understanding the mechanisms in the cell, we might be able to make them work better or further optimize their treatment,” he said.
“MAP kinase inhibitors are really like a sledgehammer to the cell,” Dr. Chuong said — meaning they affect many cellular processes, not just those related to cancer.
“If we’re able to say that these viral switches are what’s important, then that could potentially help us develop a more targeted therapy that uses something like CRISPR to silence these viral elements,” he said. Or it could help providers choose a MAP kinase inhibitor from among the dozens available best suited to treat an individual patient and avoid side effects.
Still, whether the findings translate to real cancer patients remains to be seen. “It’s very, very hard to go the final step of showing in a patient that these actually make a difference in the cancer,” Dr. Mager said.
More lab research, human trials, and at least a few years will be needed before this discovery could help treat cancer. “Directly targeting these elements as a therapy would be at least 5 years out,” Dr. Chuong said, “partly because that application would rely on CRISPR epigenome editing technology that is still being developed for clinical use.”
A version of this article first appeared on Medscape.com.
according to a fascinating new study in Science Advances. Targeting these viral remnants still lingering in our DNA could lead to more effective cancer treatment with fewer side effects, the researchers said.
The study “gives a better understanding of how gene regulation can be impacted by these ancient retroviral sequences,” said Dixie Mager, PhD, scientist emeritus at the Terry Fox Laboratory at the British Columbia Cancer Research Institute, Vancouver, British Columbia, Canada. (Mager was not involved in the study.)
Long thought to be “junk” DNA with no biologic function, “endogenous retroviruses,” which have mutated over time and lost their ability to create the virus, are now known to regulate genes — allowing some genes to turn on and off. Research in recent years suggests they may play a role in diseases like cancer.
But scientists weren’t exactly sure what that role was, said senior study author Edward Chuong, PhD, a genome biologist at the University of Colorado Boulder.
Most studies have looked at whether endogenous retroviruses code for proteins that influence cancer. But these ancient viral strands usually don’t code for proteins at all.
Dr. Chuong took a different approach. Inspired by scientists who’ve studied how viral remnants regulate positive processes (immunity, brain development, or placenta development), he and his team explored whether some might regulate genes that, once activated, help cancer thrive.
Borrowing from epigenomic analysis data (data on molecules that alter gene expression) for 21 cancers mapped by the Cancer Genome Atlas, the researchers identified 19 virus-derived DNA sequences that bind to regulatory proteins more in cancer cells than in healthy cells. All of these could potentially act as gene regulators that promote cancer.
The researchers homed in on one sequence, called LTR10, because it showed especially high activity in several cancers, including lung and colorectal cancer. This DNA segment comes from a virus that entered our ancestors’ genome 30 million years ago, and it’s activated in a third of colorectal cancers.
Using the gene editing technology clustered regularly interspaced short palindromic repeats (CRISPR), Dr. Chuong’s team silenced LTR10 in colorectal cancer cells, altering the gene sequence so it couldn’t bind to regulatory proteins. Doing so dampened the activity of nearby cancer-promoting genes.
“They still behaved like cancer cells,” Dr. Chuong said. But “it made the cancer cells more susceptible to radiation. That would imply that the presence of that viral ‘switch’ actually helped those cancer cells survive radiation therapy.”
Previously, two studies had found that viral regulators play a role in promoting two types of cancer: Leukemia and prostate cancer. The new study shows these two cases weren’t flukes. All 21 cancers they looked at had at least one of those 19 viral elements, presumably working as cancer enhancers.
The study also identified what activates LTR10 to make it promote cancer. The culprit is a regulator protein called mitogen-activated protein (MAP) kinase, which is overactivated in about 40% of all human cancers.
Some cancer drugs — MAP kinase inhibitors — already target MAP kinase, and they’re often the first ones prescribed when a patient is diagnosed with cancer, Dr. Chuong said. As with many cancer treatments, doctors don’t know why they work, just that they do.
“By understanding the mechanisms in the cell, we might be able to make them work better or further optimize their treatment,” he said.
“MAP kinase inhibitors are really like a sledgehammer to the cell,” Dr. Chuong said — meaning they affect many cellular processes, not just those related to cancer.
“If we’re able to say that these viral switches are what’s important, then that could potentially help us develop a more targeted therapy that uses something like CRISPR to silence these viral elements,” he said. Or it could help providers choose a MAP kinase inhibitor from among the dozens available best suited to treat an individual patient and avoid side effects.
Still, whether the findings translate to real cancer patients remains to be seen. “It’s very, very hard to go the final step of showing in a patient that these actually make a difference in the cancer,” Dr. Mager said.
More lab research, human trials, and at least a few years will be needed before this discovery could help treat cancer. “Directly targeting these elements as a therapy would be at least 5 years out,” Dr. Chuong said, “partly because that application would rely on CRISPR epigenome editing technology that is still being developed for clinical use.”
A version of this article first appeared on Medscape.com.
according to a fascinating new study in Science Advances. Targeting these viral remnants still lingering in our DNA could lead to more effective cancer treatment with fewer side effects, the researchers said.
The study “gives a better understanding of how gene regulation can be impacted by these ancient retroviral sequences,” said Dixie Mager, PhD, scientist emeritus at the Terry Fox Laboratory at the British Columbia Cancer Research Institute, Vancouver, British Columbia, Canada. (Mager was not involved in the study.)
Long thought to be “junk” DNA with no biologic function, “endogenous retroviruses,” which have mutated over time and lost their ability to create the virus, are now known to regulate genes — allowing some genes to turn on and off. Research in recent years suggests they may play a role in diseases like cancer.
But scientists weren’t exactly sure what that role was, said senior study author Edward Chuong, PhD, a genome biologist at the University of Colorado Boulder.
Most studies have looked at whether endogenous retroviruses code for proteins that influence cancer. But these ancient viral strands usually don’t code for proteins at all.
Dr. Chuong took a different approach. Inspired by scientists who’ve studied how viral remnants regulate positive processes (immunity, brain development, or placenta development), he and his team explored whether some might regulate genes that, once activated, help cancer thrive.
Borrowing from epigenomic analysis data (data on molecules that alter gene expression) for 21 cancers mapped by the Cancer Genome Atlas, the researchers identified 19 virus-derived DNA sequences that bind to regulatory proteins more in cancer cells than in healthy cells. All of these could potentially act as gene regulators that promote cancer.
The researchers homed in on one sequence, called LTR10, because it showed especially high activity in several cancers, including lung and colorectal cancer. This DNA segment comes from a virus that entered our ancestors’ genome 30 million years ago, and it’s activated in a third of colorectal cancers.
Using the gene editing technology clustered regularly interspaced short palindromic repeats (CRISPR), Dr. Chuong’s team silenced LTR10 in colorectal cancer cells, altering the gene sequence so it couldn’t bind to regulatory proteins. Doing so dampened the activity of nearby cancer-promoting genes.
“They still behaved like cancer cells,” Dr. Chuong said. But “it made the cancer cells more susceptible to radiation. That would imply that the presence of that viral ‘switch’ actually helped those cancer cells survive radiation therapy.”
Previously, two studies had found that viral regulators play a role in promoting two types of cancer: Leukemia and prostate cancer. The new study shows these two cases weren’t flukes. All 21 cancers they looked at had at least one of those 19 viral elements, presumably working as cancer enhancers.
The study also identified what activates LTR10 to make it promote cancer. The culprit is a regulator protein called mitogen-activated protein (MAP) kinase, which is overactivated in about 40% of all human cancers.
Some cancer drugs — MAP kinase inhibitors — already target MAP kinase, and they’re often the first ones prescribed when a patient is diagnosed with cancer, Dr. Chuong said. As with many cancer treatments, doctors don’t know why they work, just that they do.
“By understanding the mechanisms in the cell, we might be able to make them work better or further optimize their treatment,” he said.
“MAP kinase inhibitors are really like a sledgehammer to the cell,” Dr. Chuong said — meaning they affect many cellular processes, not just those related to cancer.
“If we’re able to say that these viral switches are what’s important, then that could potentially help us develop a more targeted therapy that uses something like CRISPR to silence these viral elements,” he said. Or it could help providers choose a MAP kinase inhibitor from among the dozens available best suited to treat an individual patient and avoid side effects.
Still, whether the findings translate to real cancer patients remains to be seen. “It’s very, very hard to go the final step of showing in a patient that these actually make a difference in the cancer,” Dr. Mager said.
More lab research, human trials, and at least a few years will be needed before this discovery could help treat cancer. “Directly targeting these elements as a therapy would be at least 5 years out,” Dr. Chuong said, “partly because that application would rely on CRISPR epigenome editing technology that is still being developed for clinical use.”
A version of this article first appeared on Medscape.com.
FROM SCIENCE ADVANCES
Insurers’ Rules and AI for Preauthorization: ‘Ethically Nuts,’ Says Ethicist
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the Division of Medical Ethics at New York University Grossman School of Medicine in New York City.
There are many things screwy with our healthcare system. Many of you [reading] this are dealing with bureaucracy, paperwork, all sorts of constraints, restraints, and requirements that sometimes make the practice of medicine, or even nursing, difficult.
I don’t think I’ve seen anything screwier, from a moral point of view, than the system we have that allows for preauthorization by third-party payers, or insurers, in order to give care to patients. It’s pretty clear that a third-party payer has a conflict of interest. It’s simple: They don’t want to spend money.
Their goal as profit-making companies is to reduce what it is that they’re going to authorize. That clearly is driving how the preauthorization process works. or somebody saying, this is the standard of care and this is what ought to happen.
We’re letting the people who have the pocketbooks and the wallets have prior approval of what the doctor thinks is correct. That is really not the way to practice medicine.
We now have more evidence about what really is going on. A doctor was recently interviewed by ProPublica and said that she had worked for Cigna as a reviewer. Basically, the message she got from that insurer was to speed it up, go fast, and basically “deny, deny, deny” when she got requests. Those are her words, not mine.
We get a peek under the tent of how this works, and Dr. Day is basically saying she had to leave because she just didn’t feel that it was evidence-driven. It was driven by concerns about who’s going to lose money or make money.
If you want to check to see whether something is appropriate, the question becomes, who ought to do prior review?
Who does it now? Sometimes doctors. Sometimes nurses who aren’t in the specialty where the request is coming in for preapproval. I’ve even seen situations where some companies use nurses in other countries, such as the Philippines, to do preapproval. They send them information, like a clip, to use to deny things that basically is boilerplate language, whatever the request is.
Looming up now, some insurers are starting to think, well, maybe artificial intelligence could do it. Just review the written request, trigger certain responses on the part of the artificial intelligence — it can deny the claims just as well as a human — and maybe it’s even cheaper to set up that system for the insurer.
This is ethically nuts. We need to have a system where doctors’ judgments drive what patients get. You listen to doctors, as I do, about preapproval access and they say patients sometimes give up trying to get what they think is needed. Continuity of care is interrupted if they have to keep making requests all the time.
There are adverse events when the thing that the doctor thought was most appropriate isn’t approved and something else is used that is less safe or less efficacious. It isn’t in patient interest to have the person with the wallet saying, this is what we think you need, and then having unqualified people or even automated intelligence with no accountability and no transparency get involved in preauthorization.
This system costs us money because middlemen are doing all this work. It basically becomes one of the huge scandals, in my view, of our health system, that doctors don’t ultimately decide what the patient needs. A preauthorizing third party or robot, without transparency, without accountability, and behind closed doors second-guesses what’s going on.
I’m Art Caplan at the Division of Medical Ethics at the New York University Grossman School of Medicine.
Arthur L. Caplan, Director, Division of Medical Ethics, New York University Langone Medical Center, New York, New York, has disclosed the following relevant financial relationships: Served as a director, officer, partner, employee, advisor, consultant, or trustee for Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position). Serves as a contributing author and advisor for Medscape.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the Division of Medical Ethics at New York University Grossman School of Medicine in New York City.
There are many things screwy with our healthcare system. Many of you [reading] this are dealing with bureaucracy, paperwork, all sorts of constraints, restraints, and requirements that sometimes make the practice of medicine, or even nursing, difficult.
I don’t think I’ve seen anything screwier, from a moral point of view, than the system we have that allows for preauthorization by third-party payers, or insurers, in order to give care to patients. It’s pretty clear that a third-party payer has a conflict of interest. It’s simple: They don’t want to spend money.
Their goal as profit-making companies is to reduce what it is that they’re going to authorize. That clearly is driving how the preauthorization process works. or somebody saying, this is the standard of care and this is what ought to happen.
We’re letting the people who have the pocketbooks and the wallets have prior approval of what the doctor thinks is correct. That is really not the way to practice medicine.
We now have more evidence about what really is going on. A doctor was recently interviewed by ProPublica and said that she had worked for Cigna as a reviewer. Basically, the message she got from that insurer was to speed it up, go fast, and basically “deny, deny, deny” when she got requests. Those are her words, not mine.
We get a peek under the tent of how this works, and Dr. Day is basically saying she had to leave because she just didn’t feel that it was evidence-driven. It was driven by concerns about who’s going to lose money or make money.
If you want to check to see whether something is appropriate, the question becomes, who ought to do prior review?
Who does it now? Sometimes doctors. Sometimes nurses who aren’t in the specialty where the request is coming in for preapproval. I’ve even seen situations where some companies use nurses in other countries, such as the Philippines, to do preapproval. They send them information, like a clip, to use to deny things that basically is boilerplate language, whatever the request is.
Looming up now, some insurers are starting to think, well, maybe artificial intelligence could do it. Just review the written request, trigger certain responses on the part of the artificial intelligence — it can deny the claims just as well as a human — and maybe it’s even cheaper to set up that system for the insurer.
This is ethically nuts. We need to have a system where doctors’ judgments drive what patients get. You listen to doctors, as I do, about preapproval access and they say patients sometimes give up trying to get what they think is needed. Continuity of care is interrupted if they have to keep making requests all the time.
There are adverse events when the thing that the doctor thought was most appropriate isn’t approved and something else is used that is less safe or less efficacious. It isn’t in patient interest to have the person with the wallet saying, this is what we think you need, and then having unqualified people or even automated intelligence with no accountability and no transparency get involved in preauthorization.
This system costs us money because middlemen are doing all this work. It basically becomes one of the huge scandals, in my view, of our health system, that doctors don’t ultimately decide what the patient needs. A preauthorizing third party or robot, without transparency, without accountability, and behind closed doors second-guesses what’s going on.
I’m Art Caplan at the Division of Medical Ethics at the New York University Grossman School of Medicine.
Arthur L. Caplan, Director, Division of Medical Ethics, New York University Langone Medical Center, New York, New York, has disclosed the following relevant financial relationships: Served as a director, officer, partner, employee, advisor, consultant, or trustee for Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position). Serves as a contributing author and advisor for Medscape.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the Division of Medical Ethics at New York University Grossman School of Medicine in New York City.
There are many things screwy with our healthcare system. Many of you [reading] this are dealing with bureaucracy, paperwork, all sorts of constraints, restraints, and requirements that sometimes make the practice of medicine, or even nursing, difficult.
I don’t think I’ve seen anything screwier, from a moral point of view, than the system we have that allows for preauthorization by third-party payers, or insurers, in order to give care to patients. It’s pretty clear that a third-party payer has a conflict of interest. It’s simple: They don’t want to spend money.
Their goal as profit-making companies is to reduce what it is that they’re going to authorize. That clearly is driving how the preauthorization process works. or somebody saying, this is the standard of care and this is what ought to happen.
We’re letting the people who have the pocketbooks and the wallets have prior approval of what the doctor thinks is correct. That is really not the way to practice medicine.
We now have more evidence about what really is going on. A doctor was recently interviewed by ProPublica and said that she had worked for Cigna as a reviewer. Basically, the message she got from that insurer was to speed it up, go fast, and basically “deny, deny, deny” when she got requests. Those are her words, not mine.
We get a peek under the tent of how this works, and Dr. Day is basically saying she had to leave because she just didn’t feel that it was evidence-driven. It was driven by concerns about who’s going to lose money or make money.
If you want to check to see whether something is appropriate, the question becomes, who ought to do prior review?
Who does it now? Sometimes doctors. Sometimes nurses who aren’t in the specialty where the request is coming in for preapproval. I’ve even seen situations where some companies use nurses in other countries, such as the Philippines, to do preapproval. They send them information, like a clip, to use to deny things that basically is boilerplate language, whatever the request is.
Looming up now, some insurers are starting to think, well, maybe artificial intelligence could do it. Just review the written request, trigger certain responses on the part of the artificial intelligence — it can deny the claims just as well as a human — and maybe it’s even cheaper to set up that system for the insurer.
This is ethically nuts. We need to have a system where doctors’ judgments drive what patients get. You listen to doctors, as I do, about preapproval access and they say patients sometimes give up trying to get what they think is needed. Continuity of care is interrupted if they have to keep making requests all the time.
There are adverse events when the thing that the doctor thought was most appropriate isn’t approved and something else is used that is less safe or less efficacious. It isn’t in patient interest to have the person with the wallet saying, this is what we think you need, and then having unqualified people or even automated intelligence with no accountability and no transparency get involved in preauthorization.
This system costs us money because middlemen are doing all this work. It basically becomes one of the huge scandals, in my view, of our health system, that doctors don’t ultimately decide what the patient needs. A preauthorizing third party or robot, without transparency, without accountability, and behind closed doors second-guesses what’s going on.
I’m Art Caplan at the Division of Medical Ethics at the New York University Grossman School of Medicine.
Arthur L. Caplan, Director, Division of Medical Ethics, New York University Langone Medical Center, New York, New York, has disclosed the following relevant financial relationships: Served as a director, officer, partner, employee, advisor, consultant, or trustee for Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position). Serves as a contributing author and advisor for Medscape.
A version of this article first appeared on Medscape.com.
Could Resistin Predict Death and Disease Severity in PAH?
Resistin, a cytokine expressed in adipocytes, has been associated with poor clinical outcomes in heart failure and cardiovascular disease, Li Gao, MD, of Johns Hopkins University, Baltimore, Maryland, and colleagues wrote. While mouse studies have shown that human resistin drives pulmonary vascular remodeling and the development of PAH, the role of resistin as a biomarker for PAH remains unclear.
In a study published in Respiratory Research, the researchers reviewed biospecimens and clinical and genetic data from 1121 adults with PAH, 808 with idiopathic PAH (IPAH), and 313 with scleroderma-associated PAH (SSc-PAH). They examined the associations between serum resistin levels and PAH outcomes in multivariate regression models, using machine-learning algorithms to develop models to predict mortality.
Resistin levels were significantly higher in all patients with PAH and patients with the two subtypes than in control participants (all P < .0001). Resistin was also associated with significant discriminative properties, with area under the curve (AUC) measures of 0.84, 0.82, and 0.91 for PAH overall, IPAH, and SSc-PAH, respectively.
Elevated resistin levels (defined as > 4.54 ng/mL) were significantly associated with an increased risk for death (hazard ratio, 2.6; P < .0087) as well as with older age and shorter distance on the 6-minute walk test (P = .001 for both) and reduced cardiac capacity based on the New York Heart Association functional class (P < .014).
Survival models derived from machine learning confirmed the prognostic value of resistin for mortality in PAH as seen in the random forest model, with an AUC of 0.70. “When we used the AUC values of the ROC curve as criteria to evaluate how well resistin levels discerned the presence of PAH, all three tests had excellent discriminative ability (AUCs were 0.84, 0.82, and 0.91 for all PAH, IPAH, and SSc-PAH, respectively),” the researchers wrote.
The researchers also evaluated three RETN genetic variants (rs7408174, rs3219175, and rs3745367) for a specific association with serum resistin levels and measures of PAH severity. Resistin levels were highest among individuals who were carriers of either the rs3219175 or rs3745367 mutation, the researchers noted.
The findings were limited by several factors, including missing data on the 6-minute walk test from several centers, which led to the elimination of that item from the survival analysis. Other limitations included the inability to control for PAH therapy at the time of assessment and the collection of serum at a different time from other clinical variables.
However, “our study provides evidence to support the use of circulating biomarkers as objective and accessible tools for noninvasive PAH risk stratification,” the researchers said. Additional research is needed to strengthen the association, but the findings suggest that resistin represents a novel biomarker for PAH prognostication and risk stratification and may have implications for the development of new treatments.
Biomarker Research Expands Diagnosis and Treatment Horizons
“It is a dynamic time in PAH research and clinical management, given the recent approval and use of the BMP/TGF beta balancing agent sotatercept (Winrevair) as an effective agent to target the molecular origins of this disease,” Stephen Chan, MD, professor of medicine and director of the Vascular Medicine Institute at the University of Pittsburgh, Pittsburgh, Pennsylvania, said in an interview.
The growing number of medications that can be used to treat patients with PAH will likely be more effective if patients are identified and treated early, said Dr. Chan, who was not involved in the study.
However, the time to diagnosis for patients with PAH is still more than 3 years from the start of symptoms, he said. Factors contributing to the delay include the requirement of an invasive cardiac catheterization procedure to make the final diagnosis, the status of PAH as a borderline orphan disease, and the often nonspecific nature of the initial symptoms of PAH.
Consequently, “there is an unmet need to develop effective and preferably noninvasive tools to aid in early diagnosis of PAH,” Dr. Chan added.
The power of the study is in the number of patients included, as much of previous PAH research has involved small studies of patients that could not be replicated or did not generalize to the larger patient population, Dr. Chan said.
The use of the PAH Biobank allows researchers to access a larger population of patients with PAH. “With that in mind, it is not surprising that some markers would emerge as potentially powerful and clinically meaningful,” he said.
“Currently, we do not have a reliable blood-based biomarker that we use in clinical PAH practice, although there are emerging studies that suggest other markers such as metabolites, RNA molecules, and proteins that may serve in the same capacity. If these studies turn out to be reproducible, generalizable, and specific to PAH in larger populations, measuring resistin could be helpful in making early diagnosis, particularly in areas that do not have invasive catheterization facilities (and globally) and for nonspecialists who are puzzled about the nonspecificity of initial symptoms of PAH,” Dr. Chan said.
Resistin could also be incorporated into existing risk stratification scores, such as the REVEAL risk score, that are already used in PAH clinical practice as guidance for when and how to use currently approved medications, he added.
Limitations of the study included the focus only on resistin alone, not in combination with other molecules that might perform better. Also, no independent validation cohort was used, he noted. “While PAH Biobank certainly offered larger numbers than we typically see, we would have to see validation in large independent cohorts for us to be convinced that measurements of resistin should be used in clinical practice.”
Resistin is not specific to PAH, which makes interpretation of the results more complicated, said Dr. Chan. “In this study, the authors used a smaller healthy control cohort of 50 patients as a comparison to their PAH cohort. However, they did not compare their PAH cohort with other cohorts that represent these other ‘resistin-relevant diseases’ and thus do not know whether they can distinguish PAH from any of these other diseases based on simply the resistin levels.” The frequency of comorbidities in patients with PAH, such as obesity, other inflammatory diseases, and cardiovascular disease, could confound the resistin levels.
The study was supported by the National Institutes of Health. Neither the researchers nor Dr. Chan had financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Resistin, a cytokine expressed in adipocytes, has been associated with poor clinical outcomes in heart failure and cardiovascular disease, Li Gao, MD, of Johns Hopkins University, Baltimore, Maryland, and colleagues wrote. While mouse studies have shown that human resistin drives pulmonary vascular remodeling and the development of PAH, the role of resistin as a biomarker for PAH remains unclear.
In a study published in Respiratory Research, the researchers reviewed biospecimens and clinical and genetic data from 1121 adults with PAH, 808 with idiopathic PAH (IPAH), and 313 with scleroderma-associated PAH (SSc-PAH). They examined the associations between serum resistin levels and PAH outcomes in multivariate regression models, using machine-learning algorithms to develop models to predict mortality.
Resistin levels were significantly higher in all patients with PAH and patients with the two subtypes than in control participants (all P < .0001). Resistin was also associated with significant discriminative properties, with area under the curve (AUC) measures of 0.84, 0.82, and 0.91 for PAH overall, IPAH, and SSc-PAH, respectively.
Elevated resistin levels (defined as > 4.54 ng/mL) were significantly associated with an increased risk for death (hazard ratio, 2.6; P < .0087) as well as with older age and shorter distance on the 6-minute walk test (P = .001 for both) and reduced cardiac capacity based on the New York Heart Association functional class (P < .014).
Survival models derived from machine learning confirmed the prognostic value of resistin for mortality in PAH as seen in the random forest model, with an AUC of 0.70. “When we used the AUC values of the ROC curve as criteria to evaluate how well resistin levels discerned the presence of PAH, all three tests had excellent discriminative ability (AUCs were 0.84, 0.82, and 0.91 for all PAH, IPAH, and SSc-PAH, respectively),” the researchers wrote.
The researchers also evaluated three RETN genetic variants (rs7408174, rs3219175, and rs3745367) for a specific association with serum resistin levels and measures of PAH severity. Resistin levels were highest among individuals who were carriers of either the rs3219175 or rs3745367 mutation, the researchers noted.
The findings were limited by several factors, including missing data on the 6-minute walk test from several centers, which led to the elimination of that item from the survival analysis. Other limitations included the inability to control for PAH therapy at the time of assessment and the collection of serum at a different time from other clinical variables.
However, “our study provides evidence to support the use of circulating biomarkers as objective and accessible tools for noninvasive PAH risk stratification,” the researchers said. Additional research is needed to strengthen the association, but the findings suggest that resistin represents a novel biomarker for PAH prognostication and risk stratification and may have implications for the development of new treatments.
Biomarker Research Expands Diagnosis and Treatment Horizons
“It is a dynamic time in PAH research and clinical management, given the recent approval and use of the BMP/TGF beta balancing agent sotatercept (Winrevair) as an effective agent to target the molecular origins of this disease,” Stephen Chan, MD, professor of medicine and director of the Vascular Medicine Institute at the University of Pittsburgh, Pittsburgh, Pennsylvania, said in an interview.
The growing number of medications that can be used to treat patients with PAH will likely be more effective if patients are identified and treated early, said Dr. Chan, who was not involved in the study.
However, the time to diagnosis for patients with PAH is still more than 3 years from the start of symptoms, he said. Factors contributing to the delay include the requirement of an invasive cardiac catheterization procedure to make the final diagnosis, the status of PAH as a borderline orphan disease, and the often nonspecific nature of the initial symptoms of PAH.
Consequently, “there is an unmet need to develop effective and preferably noninvasive tools to aid in early diagnosis of PAH,” Dr. Chan added.
The power of the study is in the number of patients included, as much of previous PAH research has involved small studies of patients that could not be replicated or did not generalize to the larger patient population, Dr. Chan said.
The use of the PAH Biobank allows researchers to access a larger population of patients with PAH. “With that in mind, it is not surprising that some markers would emerge as potentially powerful and clinically meaningful,” he said.
“Currently, we do not have a reliable blood-based biomarker that we use in clinical PAH practice, although there are emerging studies that suggest other markers such as metabolites, RNA molecules, and proteins that may serve in the same capacity. If these studies turn out to be reproducible, generalizable, and specific to PAH in larger populations, measuring resistin could be helpful in making early diagnosis, particularly in areas that do not have invasive catheterization facilities (and globally) and for nonspecialists who are puzzled about the nonspecificity of initial symptoms of PAH,” Dr. Chan said.
Resistin could also be incorporated into existing risk stratification scores, such as the REVEAL risk score, that are already used in PAH clinical practice as guidance for when and how to use currently approved medications, he added.
Limitations of the study included the focus only on resistin alone, not in combination with other molecules that might perform better. Also, no independent validation cohort was used, he noted. “While PAH Biobank certainly offered larger numbers than we typically see, we would have to see validation in large independent cohorts for us to be convinced that measurements of resistin should be used in clinical practice.”
Resistin is not specific to PAH, which makes interpretation of the results more complicated, said Dr. Chan. “In this study, the authors used a smaller healthy control cohort of 50 patients as a comparison to their PAH cohort. However, they did not compare their PAH cohort with other cohorts that represent these other ‘resistin-relevant diseases’ and thus do not know whether they can distinguish PAH from any of these other diseases based on simply the resistin levels.” The frequency of comorbidities in patients with PAH, such as obesity, other inflammatory diseases, and cardiovascular disease, could confound the resistin levels.
The study was supported by the National Institutes of Health. Neither the researchers nor Dr. Chan had financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
Resistin, a cytokine expressed in adipocytes, has been associated with poor clinical outcomes in heart failure and cardiovascular disease, Li Gao, MD, of Johns Hopkins University, Baltimore, Maryland, and colleagues wrote. While mouse studies have shown that human resistin drives pulmonary vascular remodeling and the development of PAH, the role of resistin as a biomarker for PAH remains unclear.
In a study published in Respiratory Research, the researchers reviewed biospecimens and clinical and genetic data from 1121 adults with PAH, 808 with idiopathic PAH (IPAH), and 313 with scleroderma-associated PAH (SSc-PAH). They examined the associations between serum resistin levels and PAH outcomes in multivariate regression models, using machine-learning algorithms to develop models to predict mortality.
Resistin levels were significantly higher in all patients with PAH and patients with the two subtypes than in control participants (all P < .0001). Resistin was also associated with significant discriminative properties, with area under the curve (AUC) measures of 0.84, 0.82, and 0.91 for PAH overall, IPAH, and SSc-PAH, respectively.
Elevated resistin levels (defined as > 4.54 ng/mL) were significantly associated with an increased risk for death (hazard ratio, 2.6; P < .0087) as well as with older age and shorter distance on the 6-minute walk test (P = .001 for both) and reduced cardiac capacity based on the New York Heart Association functional class (P < .014).
Survival models derived from machine learning confirmed the prognostic value of resistin for mortality in PAH as seen in the random forest model, with an AUC of 0.70. “When we used the AUC values of the ROC curve as criteria to evaluate how well resistin levels discerned the presence of PAH, all three tests had excellent discriminative ability (AUCs were 0.84, 0.82, and 0.91 for all PAH, IPAH, and SSc-PAH, respectively),” the researchers wrote.
The researchers also evaluated three RETN genetic variants (rs7408174, rs3219175, and rs3745367) for a specific association with serum resistin levels and measures of PAH severity. Resistin levels were highest among individuals who were carriers of either the rs3219175 or rs3745367 mutation, the researchers noted.
The findings were limited by several factors, including missing data on the 6-minute walk test from several centers, which led to the elimination of that item from the survival analysis. Other limitations included the inability to control for PAH therapy at the time of assessment and the collection of serum at a different time from other clinical variables.
However, “our study provides evidence to support the use of circulating biomarkers as objective and accessible tools for noninvasive PAH risk stratification,” the researchers said. Additional research is needed to strengthen the association, but the findings suggest that resistin represents a novel biomarker for PAH prognostication and risk stratification and may have implications for the development of new treatments.
Biomarker Research Expands Diagnosis and Treatment Horizons
“It is a dynamic time in PAH research and clinical management, given the recent approval and use of the BMP/TGF beta balancing agent sotatercept (Winrevair) as an effective agent to target the molecular origins of this disease,” Stephen Chan, MD, professor of medicine and director of the Vascular Medicine Institute at the University of Pittsburgh, Pittsburgh, Pennsylvania, said in an interview.
The growing number of medications that can be used to treat patients with PAH will likely be more effective if patients are identified and treated early, said Dr. Chan, who was not involved in the study.
However, the time to diagnosis for patients with PAH is still more than 3 years from the start of symptoms, he said. Factors contributing to the delay include the requirement of an invasive cardiac catheterization procedure to make the final diagnosis, the status of PAH as a borderline orphan disease, and the often nonspecific nature of the initial symptoms of PAH.
Consequently, “there is an unmet need to develop effective and preferably noninvasive tools to aid in early diagnosis of PAH,” Dr. Chan added.
The power of the study is in the number of patients included, as much of previous PAH research has involved small studies of patients that could not be replicated or did not generalize to the larger patient population, Dr. Chan said.
The use of the PAH Biobank allows researchers to access a larger population of patients with PAH. “With that in mind, it is not surprising that some markers would emerge as potentially powerful and clinically meaningful,” he said.
“Currently, we do not have a reliable blood-based biomarker that we use in clinical PAH practice, although there are emerging studies that suggest other markers such as metabolites, RNA molecules, and proteins that may serve in the same capacity. If these studies turn out to be reproducible, generalizable, and specific to PAH in larger populations, measuring resistin could be helpful in making early diagnosis, particularly in areas that do not have invasive catheterization facilities (and globally) and for nonspecialists who are puzzled about the nonspecificity of initial symptoms of PAH,” Dr. Chan said.
Resistin could also be incorporated into existing risk stratification scores, such as the REVEAL risk score, that are already used in PAH clinical practice as guidance for when and how to use currently approved medications, he added.
Limitations of the study included the focus only on resistin alone, not in combination with other molecules that might perform better. Also, no independent validation cohort was used, he noted. “While PAH Biobank certainly offered larger numbers than we typically see, we would have to see validation in large independent cohorts for us to be convinced that measurements of resistin should be used in clinical practice.”
Resistin is not specific to PAH, which makes interpretation of the results more complicated, said Dr. Chan. “In this study, the authors used a smaller healthy control cohort of 50 patients as a comparison to their PAH cohort. However, they did not compare their PAH cohort with other cohorts that represent these other ‘resistin-relevant diseases’ and thus do not know whether they can distinguish PAH from any of these other diseases based on simply the resistin levels.” The frequency of comorbidities in patients with PAH, such as obesity, other inflammatory diseases, and cardiovascular disease, could confound the resistin levels.
The study was supported by the National Institutes of Health. Neither the researchers nor Dr. Chan had financial conflicts to disclose.
A version of this article first appeared on Medscape.com.
New Study Says Your Sedentary Lifestyle Is Killing You
TOPLINE:
METHODOLOGY:
- Researchers evaluated the association between PA and ST with the risk for mortality in 5836 middle-aged and older Australian adults (mean age, 56.4 years; 45% men) from the Australian Diabetes, Obesity and Lifestyle Study.
- The Physical Activity and Sitting Time Balance Index (PASTBI) was calculated by dividing the total duration of daily PA by the duration of daily ST.
- Participants were categorized into quartiles on the basis of their PASTBI score, ranging from low PA/high ST to high PA/low ST.
- The primary outcome was all-cause mortality.
TAKEAWAY:
- During a median follow-up time of 14.3 years, 885 (15%) all-cause deaths were reported.
- The risk for all-cause mortality was 47% higher in participants with lower engagement in PA and higher ST (low PASTBI) than those with higher engagement in PA and lower ST (high PASTBI; adjusted hazard ratio, 1.47; 95% confidence interval, 1.21-1.79).
IN PRACTICE:
“The utility of the PASTBI in identifying relationships with mortality risk further highlights the importance of achieving a healthier balance in the dual health behaviors of PA [physical activity] and ST [sitting time],” the authors wrote.
SOURCE:
The study was led by Roslin Botlero, MBBS, MPH, PhD, of the School of Public Health and Preventive Medicine at Monash University in Melbourne, Australia. It was published online in the American Journal of Preventive Medicine.
LIMITATIONS:
The study relied on self-reported data for PA and ST, which may have introduced recall or reporting bias. The generalizability of the findings is restricted to a specific set of self-reported questionnaires. Even after adjustment for several potential confounders, other unmeasured or unknown confounders may have influenced the association between PASTBI and all-cause mortality.
DISCLOSURES:
The Australian Diabetes, Obesity and Lifestyle Study was sponsored by the National Health and Medical Research Council, the Australian Government Department of Health and Aged Care, and others. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Researchers evaluated the association between PA and ST with the risk for mortality in 5836 middle-aged and older Australian adults (mean age, 56.4 years; 45% men) from the Australian Diabetes, Obesity and Lifestyle Study.
- The Physical Activity and Sitting Time Balance Index (PASTBI) was calculated by dividing the total duration of daily PA by the duration of daily ST.
- Participants were categorized into quartiles on the basis of their PASTBI score, ranging from low PA/high ST to high PA/low ST.
- The primary outcome was all-cause mortality.
TAKEAWAY:
- During a median follow-up time of 14.3 years, 885 (15%) all-cause deaths were reported.
- The risk for all-cause mortality was 47% higher in participants with lower engagement in PA and higher ST (low PASTBI) than those with higher engagement in PA and lower ST (high PASTBI; adjusted hazard ratio, 1.47; 95% confidence interval, 1.21-1.79).
IN PRACTICE:
“The utility of the PASTBI in identifying relationships with mortality risk further highlights the importance of achieving a healthier balance in the dual health behaviors of PA [physical activity] and ST [sitting time],” the authors wrote.
SOURCE:
The study was led by Roslin Botlero, MBBS, MPH, PhD, of the School of Public Health and Preventive Medicine at Monash University in Melbourne, Australia. It was published online in the American Journal of Preventive Medicine.
LIMITATIONS:
The study relied on self-reported data for PA and ST, which may have introduced recall or reporting bias. The generalizability of the findings is restricted to a specific set of self-reported questionnaires. Even after adjustment for several potential confounders, other unmeasured or unknown confounders may have influenced the association between PASTBI and all-cause mortality.
DISCLOSURES:
The Australian Diabetes, Obesity and Lifestyle Study was sponsored by the National Health and Medical Research Council, the Australian Government Department of Health and Aged Care, and others. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Researchers evaluated the association between PA and ST with the risk for mortality in 5836 middle-aged and older Australian adults (mean age, 56.4 years; 45% men) from the Australian Diabetes, Obesity and Lifestyle Study.
- The Physical Activity and Sitting Time Balance Index (PASTBI) was calculated by dividing the total duration of daily PA by the duration of daily ST.
- Participants were categorized into quartiles on the basis of their PASTBI score, ranging from low PA/high ST to high PA/low ST.
- The primary outcome was all-cause mortality.
TAKEAWAY:
- During a median follow-up time of 14.3 years, 885 (15%) all-cause deaths were reported.
- The risk for all-cause mortality was 47% higher in participants with lower engagement in PA and higher ST (low PASTBI) than those with higher engagement in PA and lower ST (high PASTBI; adjusted hazard ratio, 1.47; 95% confidence interval, 1.21-1.79).
IN PRACTICE:
“The utility of the PASTBI in identifying relationships with mortality risk further highlights the importance of achieving a healthier balance in the dual health behaviors of PA [physical activity] and ST [sitting time],” the authors wrote.
SOURCE:
The study was led by Roslin Botlero, MBBS, MPH, PhD, of the School of Public Health and Preventive Medicine at Monash University in Melbourne, Australia. It was published online in the American Journal of Preventive Medicine.
LIMITATIONS:
The study relied on self-reported data for PA and ST, which may have introduced recall or reporting bias. The generalizability of the findings is restricted to a specific set of self-reported questionnaires. Even after adjustment for several potential confounders, other unmeasured or unknown confounders may have influenced the association between PASTBI and all-cause mortality.
DISCLOSURES:
The Australian Diabetes, Obesity and Lifestyle Study was sponsored by the National Health and Medical Research Council, the Australian Government Department of Health and Aged Care, and others. The authors declared no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Changing the tumor board conversation: Immunotherapy in resectable NSCLC
Without a doubt, immunotherapy has transformed the treatment landscape of non-small cell lung cancer (NSCLC) and enhanced survival rates across the different stages of disease. High recurrence rates following complete surgical resection prompted the study of immune checkpoint inhibitors (ICI) in earlier, operable stages of disease. This shift toward early application of ICI reflects the larger trend toward merging precision oncology with lung cancer staging. The resulting complexity in treatment and decision making creates systemic and logistical challenges that will require health care systems to adapt and improve.
Adjuvant immunotherapy for NSCLC
Prior to recent approvals for adjuvant immunotherapy, it was standard to give chemotherapy following resection of stage IB-IIIA disease, which offered a statistically nonsignificant survival gain. Recurrence in these patients is believed to be related to postsurgical micrometastasis. The utilization of alternative mechanisms to prevent recurrence is increasingly more common.
Atezolizumab, a PD-L1 inhibitor, is currently approved as first-line adjuvant treatment following chemotherapy in post-NSCLC resection patients with PD-L1 scores ≥1%. This category one recommendation by the National Comprehensive Cancer Network (NCCN) is based on results from the IMpower010 trial, which randomized patients to Atezolizumab vs best supportive care. All were early-stage NSCLC, stage IB-IIIA, who underwent resection followed by platinum-based chemotherapy. Statistically significant benefits were found in disease-free survival (DFS) with a trend toward overall survival.1
The PEARLS/KEYNOTE-091 trial evaluated another PD-L1 inhibitor, Pembrolizumab, as adjuvant therapy. Its design largely mirrored the IMPower010 study, but it differed in that the ICI was administered with or without chemotherapy following resection in patients with stage IB-IIIA NSCLC. Improvements in DFS were found in the overall population, leading to FDA approval for adjuvant therapy in 2023.2
These approvals require changes to the management of operable NSCLC. Until recently, it was not routine to send surgical specimens for additional testing because adjuvant treatment meant chemotherapy only. However, it is now essential that all surgically resected malignant tissue be sent for genomic sequencing and PD-L1 testing. Selecting the next form of therapy, whether it is an ICI or targeted drug therapy, depends on it.
From a surgical perspective, quality surgery with accurate nodal staging is crucial. The surgical findings can determine and identify those who are candidates for adjuvant immunotherapy. For these same reasons, it is helpful to advise surgeons preoperatively that targeted adjuvant therapy is being considered after resection.
Neoadjuvant immunotherapy for NSCLC
ICIs have also been used as neoadjuvant treatment for operable NSCLC. In 2021, the Checkmate-816 trial evaluated Nivolumab with platinum doublet chemotherapy prior to resection of stage IB-IIIa NSCLC. When compared with chemotherapy alone, there were significant improvements in EFS, MPR, and time to death or distant metastasis (TTDM) out to 3 years. At a median follow-up time of 41.4 months, only 28% in the nivolumab group had recurrence postsurgery compared with 42% in the chemotherapy-alone group.3 As a result, certain patients who are likely to receive adjuvant chemotherapy may additionally receive neoadjuvant immunotherapy with chemotherapy before surgical resection. In 2023, the KEYNOTE-671 study demonstrated that neoadjuvant Pembrolizumab and chemotherapy in patients with resectable stage II-IIIb (N2 stage) NSCLC improved EFS. At a median follow-up of 25.2 months, the EFS was 62.4% in the Pembrolizumab group vs 40.6% in the placebo group (P < .001).4
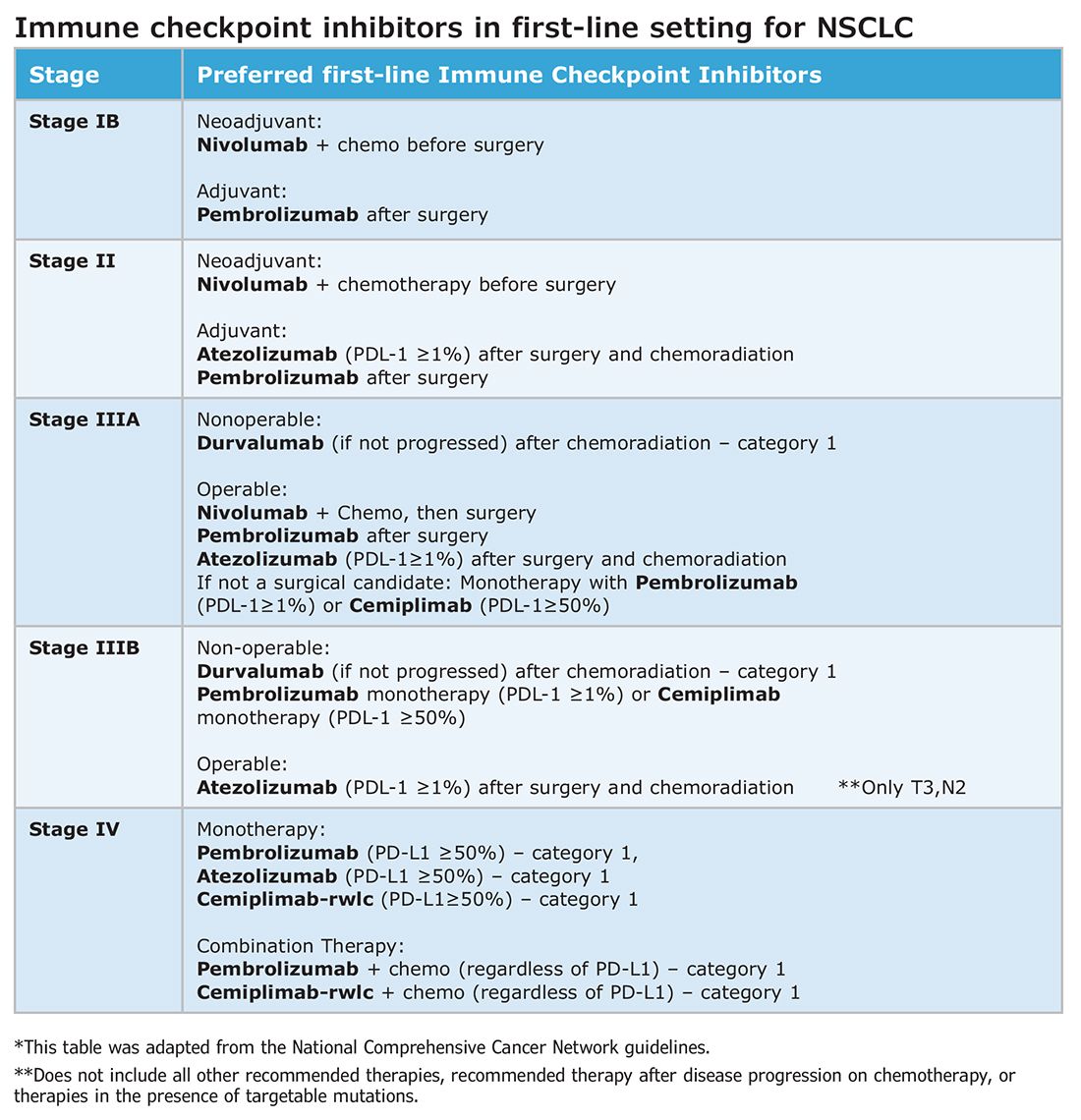
Such changes in treatment options mean patients should be discussed first and simultaneous referrals to oncology and surgery should occur in early-stage NSCLC. Up-front genomic phenotyping and PD-L1 testing may assist in decision making. High PD-L1 levels correlate better with response.
When an ICI-chemotherapy combination is given up front for newly diagnosed NSCLC, there is the potential for large reductions in tumor size and lymph node burden. Although the NCCN does not recommend ICIs to induce resectability, a patient originally deemed inoperable could theoretically become a surgical candidate with neoadjuvant ICI treatment. There is also the potential for toxicity, which could increase the risk of surgery when it does occur. Such scenarios will require frequent tumor board discussions so plans can be adjusted in real time to optimize outcomes as clinical circumstances change.
Perioperative immunotherapy for NSCLC
It is clear that both neoadjuvant and adjuvant immunotherapy can improve outcomes for patients with resectable NSCLC. The combination of neoadjuvant with adjuvant immunotherapy/chemotherapy is currently being studied. Two recent phase III clinical trials, NEOTORCH and AEGAEN, have found statistical improvements in EFS and MPR with this approach.5,6 These studies have not found their way into the NCCN guidelines yet but are sure to be considered in future iterations. Once adopted, the tumor board at each institution will have more options to choose from but many more decisions to make.
References
1. Felip E, Altorki N, Zhou C, et al. Adjuvant atezolizumab after adjuvant chemotherapy in resected stage IB-IIIA non-small-cell lung cancer (IMpower010): a randomised, multicentre, open-label, phase 3 trial. Lancet. 2021;398(10308):1344-1357. [Published correction appears in Lancet. 2021 Nov 6;398(10312):1686.]
2. O’Brien M, Paz-Ares L, Marreaud S, et al. Pembrolizumab versus placebo as adjuvant therapy for completely resected stage IB-IIIA non-small-cell lung cancer (PEARLS/KEYNOTE-091): an interim analysis of a randomised, triple-blind, phase 3 trial. Lancet Oncol. 2022;23(10):1274-1286.
3. Forde PM, Spicer J, Lu S, et al. Neoadjuvant nivolumab plus chemotherapy in resectable lung cancer. N Engl J Med. 2022;386(21):1973-1985.
4. Wakelee H, Liberman M, Kato T, et al. Perioperative pembrolizumab for early-stage non-small-cell lung cancer. N Engl J Med. 2023;389(6):491-503.
5. Lu S, Zhang W, Wu L, et al. Perioperative toripalimab plus chemotherapy for patients with resectable non-small cell lung cancer: the neotorch randomized clinical trial. JAMA. 2024;331(3):201-211.
6. Heymach JV, Harpole D, Mitsudomi T, et al. Perioperative durvalumab for resectable non-small-cell lung cancer. N Engl J Med. 2023;389(18):1672-1684.
Without a doubt, immunotherapy has transformed the treatment landscape of non-small cell lung cancer (NSCLC) and enhanced survival rates across the different stages of disease. High recurrence rates following complete surgical resection prompted the study of immune checkpoint inhibitors (ICI) in earlier, operable stages of disease. This shift toward early application of ICI reflects the larger trend toward merging precision oncology with lung cancer staging. The resulting complexity in treatment and decision making creates systemic and logistical challenges that will require health care systems to adapt and improve.
Adjuvant immunotherapy for NSCLC
Prior to recent approvals for adjuvant immunotherapy, it was standard to give chemotherapy following resection of stage IB-IIIA disease, which offered a statistically nonsignificant survival gain. Recurrence in these patients is believed to be related to postsurgical micrometastasis. The utilization of alternative mechanisms to prevent recurrence is increasingly more common.
Atezolizumab, a PD-L1 inhibitor, is currently approved as first-line adjuvant treatment following chemotherapy in post-NSCLC resection patients with PD-L1 scores ≥1%. This category one recommendation by the National Comprehensive Cancer Network (NCCN) is based on results from the IMpower010 trial, which randomized patients to Atezolizumab vs best supportive care. All were early-stage NSCLC, stage IB-IIIA, who underwent resection followed by platinum-based chemotherapy. Statistically significant benefits were found in disease-free survival (DFS) with a trend toward overall survival.1
The PEARLS/KEYNOTE-091 trial evaluated another PD-L1 inhibitor, Pembrolizumab, as adjuvant therapy. Its design largely mirrored the IMPower010 study, but it differed in that the ICI was administered with or without chemotherapy following resection in patients with stage IB-IIIA NSCLC. Improvements in DFS were found in the overall population, leading to FDA approval for adjuvant therapy in 2023.2
These approvals require changes to the management of operable NSCLC. Until recently, it was not routine to send surgical specimens for additional testing because adjuvant treatment meant chemotherapy only. However, it is now essential that all surgically resected malignant tissue be sent for genomic sequencing and PD-L1 testing. Selecting the next form of therapy, whether it is an ICI or targeted drug therapy, depends on it.
From a surgical perspective, quality surgery with accurate nodal staging is crucial. The surgical findings can determine and identify those who are candidates for adjuvant immunotherapy. For these same reasons, it is helpful to advise surgeons preoperatively that targeted adjuvant therapy is being considered after resection.
Neoadjuvant immunotherapy for NSCLC
ICIs have also been used as neoadjuvant treatment for operable NSCLC. In 2021, the Checkmate-816 trial evaluated Nivolumab with platinum doublet chemotherapy prior to resection of stage IB-IIIa NSCLC. When compared with chemotherapy alone, there were significant improvements in EFS, MPR, and time to death or distant metastasis (TTDM) out to 3 years. At a median follow-up time of 41.4 months, only 28% in the nivolumab group had recurrence postsurgery compared with 42% in the chemotherapy-alone group.3 As a result, certain patients who are likely to receive adjuvant chemotherapy may additionally receive neoadjuvant immunotherapy with chemotherapy before surgical resection. In 2023, the KEYNOTE-671 study demonstrated that neoadjuvant Pembrolizumab and chemotherapy in patients with resectable stage II-IIIb (N2 stage) NSCLC improved EFS. At a median follow-up of 25.2 months, the EFS was 62.4% in the Pembrolizumab group vs 40.6% in the placebo group (P < .001).4

Such changes in treatment options mean patients should be discussed first and simultaneous referrals to oncology and surgery should occur in early-stage NSCLC. Up-front genomic phenotyping and PD-L1 testing may assist in decision making. High PD-L1 levels correlate better with response.
When an ICI-chemotherapy combination is given up front for newly diagnosed NSCLC, there is the potential for large reductions in tumor size and lymph node burden. Although the NCCN does not recommend ICIs to induce resectability, a patient originally deemed inoperable could theoretically become a surgical candidate with neoadjuvant ICI treatment. There is also the potential for toxicity, which could increase the risk of surgery when it does occur. Such scenarios will require frequent tumor board discussions so plans can be adjusted in real time to optimize outcomes as clinical circumstances change.
Perioperative immunotherapy for NSCLC
It is clear that both neoadjuvant and adjuvant immunotherapy can improve outcomes for patients with resectable NSCLC. The combination of neoadjuvant with adjuvant immunotherapy/chemotherapy is currently being studied. Two recent phase III clinical trials, NEOTORCH and AEGAEN, have found statistical improvements in EFS and MPR with this approach.5,6 These studies have not found their way into the NCCN guidelines yet but are sure to be considered in future iterations. Once adopted, the tumor board at each institution will have more options to choose from but many more decisions to make.
References
1. Felip E, Altorki N, Zhou C, et al. Adjuvant atezolizumab after adjuvant chemotherapy in resected stage IB-IIIA non-small-cell lung cancer (IMpower010): a randomised, multicentre, open-label, phase 3 trial. Lancet. 2021;398(10308):1344-1357. [Published correction appears in Lancet. 2021 Nov 6;398(10312):1686.]
2. O’Brien M, Paz-Ares L, Marreaud S, et al. Pembrolizumab versus placebo as adjuvant therapy for completely resected stage IB-IIIA non-small-cell lung cancer (PEARLS/KEYNOTE-091): an interim analysis of a randomised, triple-blind, phase 3 trial. Lancet Oncol. 2022;23(10):1274-1286.
3. Forde PM, Spicer J, Lu S, et al. Neoadjuvant nivolumab plus chemotherapy in resectable lung cancer. N Engl J Med. 2022;386(21):1973-1985.
4. Wakelee H, Liberman M, Kato T, et al. Perioperative pembrolizumab for early-stage non-small-cell lung cancer. N Engl J Med. 2023;389(6):491-503.
5. Lu S, Zhang W, Wu L, et al. Perioperative toripalimab plus chemotherapy for patients with resectable non-small cell lung cancer: the neotorch randomized clinical trial. JAMA. 2024;331(3):201-211.
6. Heymach JV, Harpole D, Mitsudomi T, et al. Perioperative durvalumab for resectable non-small-cell lung cancer. N Engl J Med. 2023;389(18):1672-1684.
Without a doubt, immunotherapy has transformed the treatment landscape of non-small cell lung cancer (NSCLC) and enhanced survival rates across the different stages of disease. High recurrence rates following complete surgical resection prompted the study of immune checkpoint inhibitors (ICI) in earlier, operable stages of disease. This shift toward early application of ICI reflects the larger trend toward merging precision oncology with lung cancer staging. The resulting complexity in treatment and decision making creates systemic and logistical challenges that will require health care systems to adapt and improve.
Adjuvant immunotherapy for NSCLC
Prior to recent approvals for adjuvant immunotherapy, it was standard to give chemotherapy following resection of stage IB-IIIA disease, which offered a statistically nonsignificant survival gain. Recurrence in these patients is believed to be related to postsurgical micrometastasis. The utilization of alternative mechanisms to prevent recurrence is increasingly more common.
Atezolizumab, a PD-L1 inhibitor, is currently approved as first-line adjuvant treatment following chemotherapy in post-NSCLC resection patients with PD-L1 scores ≥1%. This category one recommendation by the National Comprehensive Cancer Network (NCCN) is based on results from the IMpower010 trial, which randomized patients to Atezolizumab vs best supportive care. All were early-stage NSCLC, stage IB-IIIA, who underwent resection followed by platinum-based chemotherapy. Statistically significant benefits were found in disease-free survival (DFS) with a trend toward overall survival.1
The PEARLS/KEYNOTE-091 trial evaluated another PD-L1 inhibitor, Pembrolizumab, as adjuvant therapy. Its design largely mirrored the IMPower010 study, but it differed in that the ICI was administered with or without chemotherapy following resection in patients with stage IB-IIIA NSCLC. Improvements in DFS were found in the overall population, leading to FDA approval for adjuvant therapy in 2023.2
These approvals require changes to the management of operable NSCLC. Until recently, it was not routine to send surgical specimens for additional testing because adjuvant treatment meant chemotherapy only. However, it is now essential that all surgically resected malignant tissue be sent for genomic sequencing and PD-L1 testing. Selecting the next form of therapy, whether it is an ICI or targeted drug therapy, depends on it.
From a surgical perspective, quality surgery with accurate nodal staging is crucial. The surgical findings can determine and identify those who are candidates for adjuvant immunotherapy. For these same reasons, it is helpful to advise surgeons preoperatively that targeted adjuvant therapy is being considered after resection.
Neoadjuvant immunotherapy for NSCLC
ICIs have also been used as neoadjuvant treatment for operable NSCLC. In 2021, the Checkmate-816 trial evaluated Nivolumab with platinum doublet chemotherapy prior to resection of stage IB-IIIa NSCLC. When compared with chemotherapy alone, there were significant improvements in EFS, MPR, and time to death or distant metastasis (TTDM) out to 3 years. At a median follow-up time of 41.4 months, only 28% in the nivolumab group had recurrence postsurgery compared with 42% in the chemotherapy-alone group.3 As a result, certain patients who are likely to receive adjuvant chemotherapy may additionally receive neoadjuvant immunotherapy with chemotherapy before surgical resection. In 2023, the KEYNOTE-671 study demonstrated that neoadjuvant Pembrolizumab and chemotherapy in patients with resectable stage II-IIIb (N2 stage) NSCLC improved EFS. At a median follow-up of 25.2 months, the EFS was 62.4% in the Pembrolizumab group vs 40.6% in the placebo group (P < .001).4

Such changes in treatment options mean patients should be discussed first and simultaneous referrals to oncology and surgery should occur in early-stage NSCLC. Up-front genomic phenotyping and PD-L1 testing may assist in decision making. High PD-L1 levels correlate better with response.
When an ICI-chemotherapy combination is given up front for newly diagnosed NSCLC, there is the potential for large reductions in tumor size and lymph node burden. Although the NCCN does not recommend ICIs to induce resectability, a patient originally deemed inoperable could theoretically become a surgical candidate with neoadjuvant ICI treatment. There is also the potential for toxicity, which could increase the risk of surgery when it does occur. Such scenarios will require frequent tumor board discussions so plans can be adjusted in real time to optimize outcomes as clinical circumstances change.
Perioperative immunotherapy for NSCLC
It is clear that both neoadjuvant and adjuvant immunotherapy can improve outcomes for patients with resectable NSCLC. The combination of neoadjuvant with adjuvant immunotherapy/chemotherapy is currently being studied. Two recent phase III clinical trials, NEOTORCH and AEGAEN, have found statistical improvements in EFS and MPR with this approach.5,6 These studies have not found their way into the NCCN guidelines yet but are sure to be considered in future iterations. Once adopted, the tumor board at each institution will have more options to choose from but many more decisions to make.
References
1. Felip E, Altorki N, Zhou C, et al. Adjuvant atezolizumab after adjuvant chemotherapy in resected stage IB-IIIA non-small-cell lung cancer (IMpower010): a randomised, multicentre, open-label, phase 3 trial. Lancet. 2021;398(10308):1344-1357. [Published correction appears in Lancet. 2021 Nov 6;398(10312):1686.]
2. O’Brien M, Paz-Ares L, Marreaud S, et al. Pembrolizumab versus placebo as adjuvant therapy for completely resected stage IB-IIIA non-small-cell lung cancer (PEARLS/KEYNOTE-091): an interim analysis of a randomised, triple-blind, phase 3 trial. Lancet Oncol. 2022;23(10):1274-1286.
3. Forde PM, Spicer J, Lu S, et al. Neoadjuvant nivolumab plus chemotherapy in resectable lung cancer. N Engl J Med. 2022;386(21):1973-1985.
4. Wakelee H, Liberman M, Kato T, et al. Perioperative pembrolizumab for early-stage non-small-cell lung cancer. N Engl J Med. 2023;389(6):491-503.
5. Lu S, Zhang W, Wu L, et al. Perioperative toripalimab plus chemotherapy for patients with resectable non-small cell lung cancer: the neotorch randomized clinical trial. JAMA. 2024;331(3):201-211.
6. Heymach JV, Harpole D, Mitsudomi T, et al. Perioperative durvalumab for resectable non-small-cell lung cancer. N Engl J Med. 2023;389(18):1672-1684.


