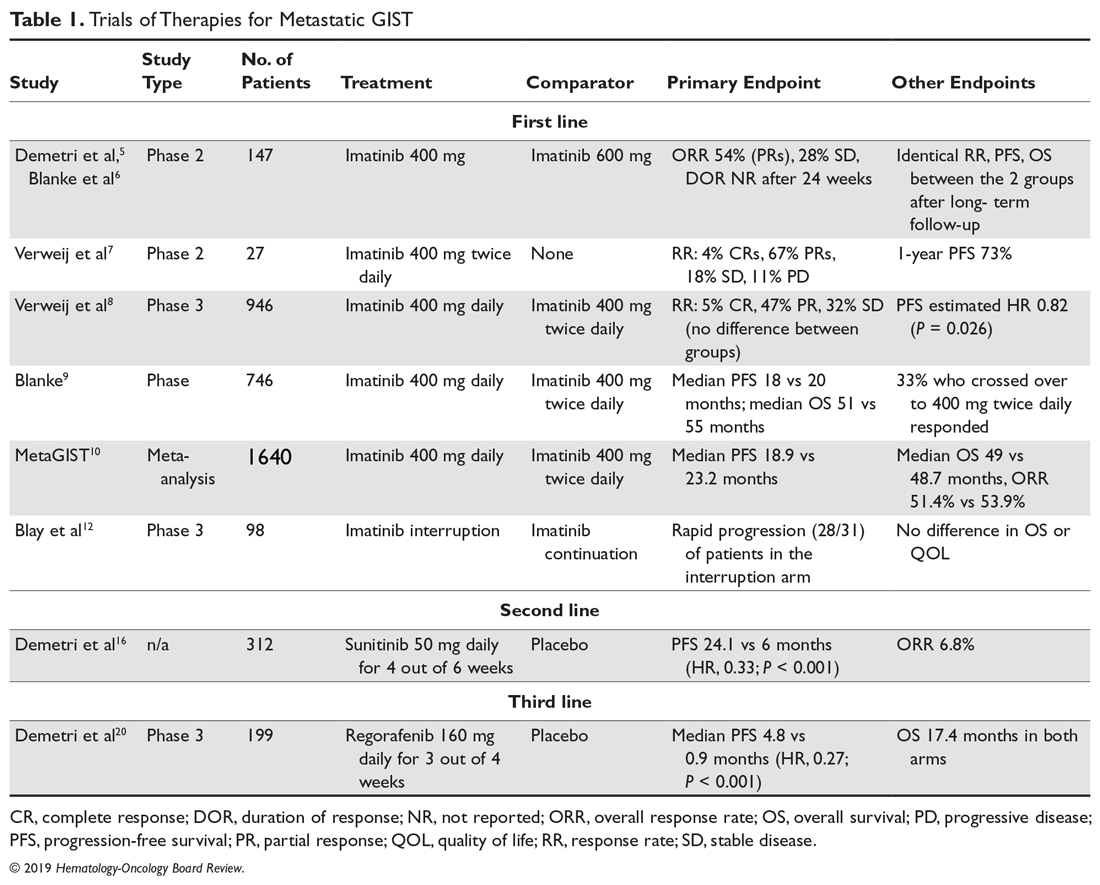
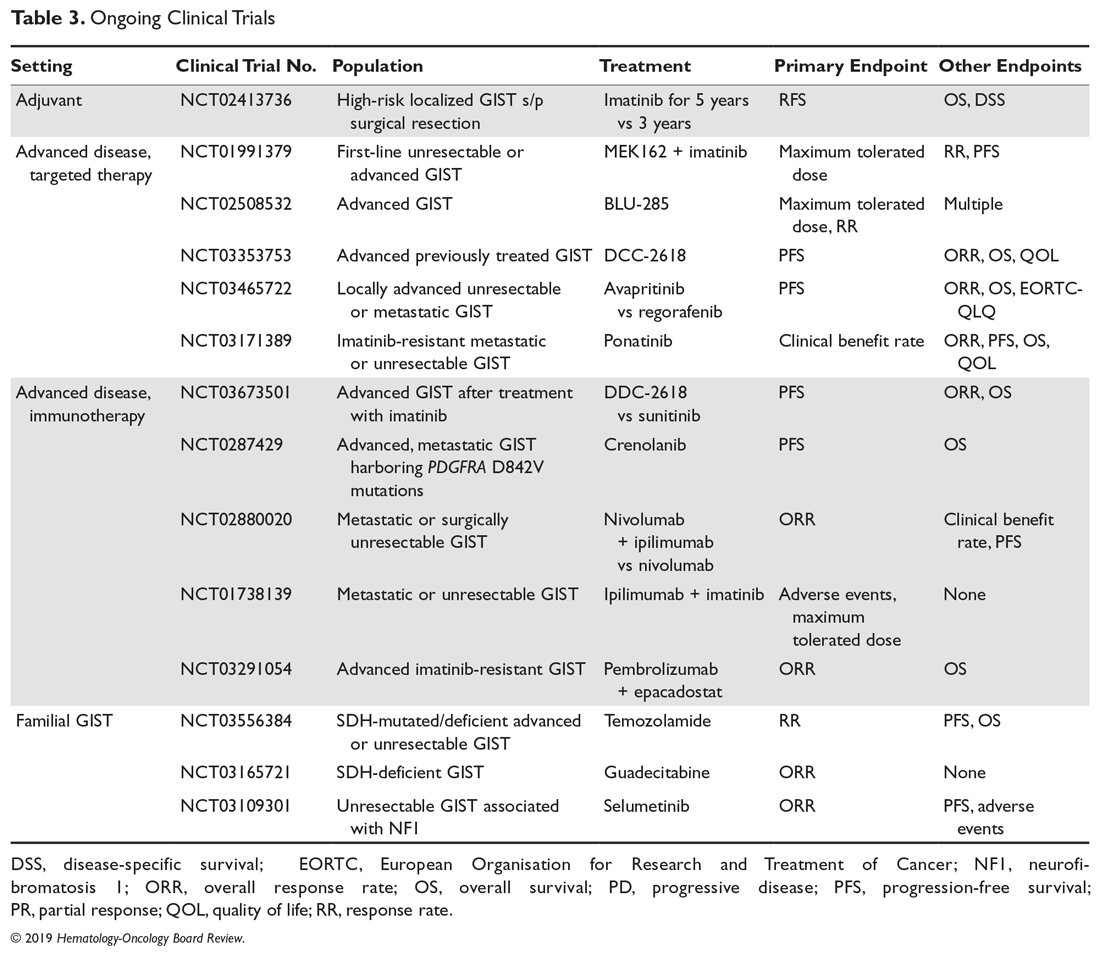
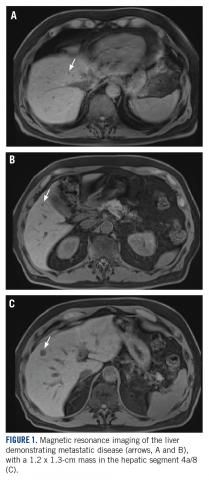
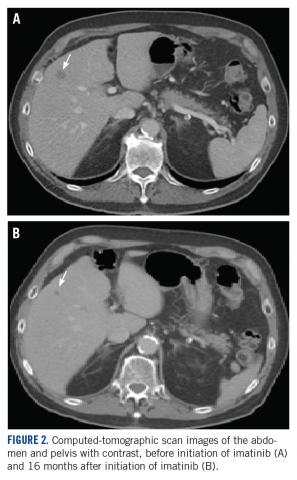
User login
Conference Coverage: ASCO 2019
Behind Olaratumab's Phase 3 Disappointment
ANNOUNCE, the phase 3 trial designed to confirm the clinical benefit of olaratumab in patients with advanced soft tissue sarcoma (STS), failed to meet its primary endpoint of overall survival (OS) in all STS histologies and the leiomyosarcoma population. The previous phase 1b/2 signal-finding study of olaratumab had achieved an unprecedented improvement in OS, and the US Food and Drug Administration (FDA) awarded olaratumab accelerated approval in October 2016. By December 2018, olaratumab received additional accelerated, conditional, and full approvals in more than 40 countries worldwide. William D. Tap, MD, chief of the Sarcoma Medical Oncology Service at Memorial Sloan Kettering Cancer Center in New York, presented the phase 3 results and provided some explanations for the findings during the plenary session at ASCO.
ANNOUNCE (NCT02451943), which was designed and enrolled prior to olaratumab receiving accelerated approval, opened in September 2015 and completed accrual 10 months later in July 2016. Investigators randomized and treated 509 patients with advanced STS not amenable to curative therapy, 258 patients in the olaratumab-doxorubicin arm and 251 in the placebo-doxorubicin arm. Most patients (46%) had leiomyosarcoma, followed by liposarcoma (18%), pleomorphic sarcoma (13%), and 24% of the patient population had 26 unique histologies. Three-quarters of the patients had no prior systemic therapy.
Results
As of the data cutoff on December 5, 2018, there were no survival differences in the intention-to-treat population, in the total STS population nor in the leiomyosarcoma subpopulation, with olaratumab-doxorubicin compared to placebo-doxorubicin. For the total STS population, median OS with olaratumab- doxorubicin was 20.4 months and with placebo-doxorubicin 19.7 months. “This is the highest survival rate described to date in any phase 3 sarcoma study,” Dr. Tap said. “It is of particular interest as ANNOUNCE did not mandate treatment in the first line.” In the leiomyosarcoma population, median OS was 21.6 months with olaratumab and 21.9 months with placebo. The secondary endpoints of progression-free survival (PFS), overall response rate, and disease control rate did not favor olaratumab either.
Investigators are examining the relationship between PDGFRα expression and OS in ANNOUNCE. PDGFRα-positive tumors tended to do worse with olaratumab than PDGFRα-negative tumors. The investigators noticed a 6-month difference in OS between these populations favoring PDGFRα-negative tumors. Additional biomarker analyses are ongoing.
A large and concerted effort is underway, Dr. Tap said, to understand the results of the ANNOUNCE study alone and in context with the phase 1b/2 study. “There are no noted discrepancies in study conduct or data integrity which could explain these findings or the differences between the two studies.”
Possible explanations
The designs of the phase 1b/2 and phase 3 studies had some important differences. The phase 1b/2 study was a small, open-label, US-centric study (10 sites) that did not include a placebo or subtype- specified analyses. Its primary endpoint was PFS, it did not have a loading dose of olaratumab, and it specified the timing of dexrazoxane administration after 300 mg/m2 of doxorubicin.
ANNOUNCE, on the other hand, was a large (n=509), international (110 study sites), double-blind, placebo-controlled trial that had outcomes evaluated in STS and leiomyosarcoma. Its primary endpoint was OS, it had a loading dose of olaratumab of 20 mg/kg, and there was no restriction as to the timing of dexrazoxane administration.
Dr. Tap pointed out that in ANNOUNCE it was difficult to predict or control for factors that may have had an unanticipated influence on outcomes, such as albumin levels as a surrogate for disease burden and behavior of PDGFRα status. It is possible, he said, that olaratumab has no activity in STS and that the phase 1b/2 results were due to, among other things, the small sample size, numerous represented histologies with disparate clinical behavior, and the effect of subtype-specific therapies on overall survival, given subsequently or even by chance. On the other hand, it is also possible, he said, that olaratumab has some activity in STS, with outcomes being affected by the heterogeneity of the study populations, differences in trial design, and the performance of the ANNOUNCE control arm. Whatever the case, he said, accelerated approval allowed patients to have access to a potentially life-prolonging drug with little added toxicity.
Discussion
In the expert discussion following the presentation, Jaap Verweij, MD, PhD, of Erasmus University Medical Center in Rotterdam, The Netherlands, congratulated the investigators for performing the study at an unprecedented pace. He commented that lumping STS subtypes together is problematic, as different histological subtypes behave as though they are different diseases. Small numbers of each tumor subtype and subtypes with slow tumor growth can impact trial outcomes. In the phase 1b/2 and phase 3 trials, 26 different subtypes were represented in each study. Dr. Verweij pointed out this could have made a big difference in the phase 1b/2 study, in which there were only 66 patients in each arm.
It is striking to note, he said, that without exception, phase 2 randomized studies in STS involving doxorubicin consistently overestimated and wrongly predicted PFS in the subsequent phase 3 studies. And the situation is similar for OS. The results of the ANNOUNCE study are no exception, he added. “Taken together, these studies indicate that phase 2 studies in soft tissue sarcomas, certainly those involving additions of drugs to doxorubicin, even if randomized, should be interpreted with great caution,” he said.
SOURCE: Tap WD, et al. J Clin Oncol 37, 2019 (suppl; abstr LBA3)
The study was sponsored by Eli Lilly and Company.
Dr. Tap reported research funding from Lilly and Dr. Verweij had nothing to report related to this study. Abstract coauthors disclosed numerous financial relationships, including consulting/advisory roles and/or research funding from Lilly, and several were employed by Lilly.
Addition of Temozolomide May Improve Outcomes in RMS
Investigators from the European Pediatric Soft Tissue Sarcoma Study Group (EpSSG) found that the addition of temozolomide (T) to vincristine and irinotecan (VI) may improve outcomes in adults and children with relapsed or refractory rhabdomyosarcoma (RMS). Principal investigator of the study, Anne Sophie Defachelles, MD, pediatric oncologist at the Centre Oscar Lambret in Lille, France, presented the results on behalf of the EpSSG.
The primary objective of the study was to evaluate the efficacy of VI and VIT regimens, defined as objective response (OR)—complete response (CR) plus partial response (PR)—after 2 cycles. Secondary objectives were progression-free survival (PFS), overall survival (OS), and safety in each arm, and the relative treatment effect of VIT compared to VI in terms of OR, survival, and safety.
The international, randomized (1:1), open-label, phase 2 trial (VIT-0910; NCT01355445) was conducted at 37 centers in 5 countries. Patients ages 6 months to 50 years with RMS were eligible. They could not have had prior irinotecan or temozolomide. A 2015 protocol amendment limited enrollment to patients at relapse and increased the enrollment goal by 40 patients. After the 2015 amendment, patients with refractory disease were no longer eligible.
From January 2012 to April 2018, investigators enrolled 120 patients, 60 on each arm. Two patients in the VI arm were not treated. Patients were a median age of 10.5 years in the VI arm and 12 years in the VIT arm, 92% (VI) and 87% (VIT) had relapsed disease, 8% (VI) and 13% (VIT) had refractory disease, and 55% (VI) and 68% (VIT) had metastatic disease at study entry.
Results
Patients achieved an OR rate of 44% (VIT) and 31% (VI) for the whole population, one-sided P value <.0001. The adjusted odds ratio for the whole population was 0.50, P=.09. PFS was 4.7 months (VIT) and 3.2 months (VI), “a nearly significant reduction in the risk of progression,” Dr. Defachelles noted. Median OS was 15.0 months (VIT) and 10.3 months (VI), which amounted to “a large and significant reduction in the risk of death,” she said. The adjusted hazard ratio was 0.55, P=.006.
Adverse events of grade 3 or higher were more frequent in the VIT arm, with hematologic toxicity the most frequent (81% for VIT, 59% for VI), followed by gastrointestinal adverse events. “VIT was significantly more toxic than VI,” Dr. Defachelles observed, “but the toxicity was manageable.”
“VIT is now the standard treatment in Europe for relapsed rhabdomyosarcoma and will be the control arm in the multiarm, multistage RMS study for relapsed patients,” she said.
In a discussion following the presentation, Lars M. Wagner, MD, of Cincinnati Children’s Hospital, pointed out that the study was not powered for the PFS and OS assessments. These were secondary objectives that should be considered exploratory. Therefore, he said, the outcome data is not conclusive. The role of temozolomide in RMS is also unclear, given recent negative results in patients with newly diagnosed metastatic RMS (Malempati et al, Cancer 2019). And he said it’s uncertain how these results apply to patients who received irinotecan upfront for RMS.
SOURCE: Defachelles AS, et al. J Clin Oncol 37, 2019 (suppl; abstr 10000)
The study was sponsored by Centre Oscar Lambret and SFCE (Société Française de Lutte contre les Cancers et Lucémies de l’Enfant et de l’Adolescent) served as collaborator.
Drs. Defachelles and Wagner had no relationships to disclose. A few coauthors had advisory/consulting or speaker roles for various commercial interests, including two for Merck (temozolomide).
Pazopanib Increases Pathologic Necrosis Rates in STS
Pazopanib added to a regimen of preoperative chemoradiation in non-rhabdomyosarcoma soft tissue sarcoma (NRSTS) significantly increased the rate of near-complete pathologic response in both children and adults with intermediate or high-risk disease. Pazopanib, a multitargeted receptor tyrosine kinase inhibitor, works in multiple signaling pathways involved in tumorigenesis— VEGFR-1, -2, -3, PDGFRα/β, and c-kit. A phase 3 study demonstrated significant improvement in progression-free survival (PFS) in advanced STS patients and was the basis for its approval in the US and elsewhere for treatment of this patient population. Preclinical data suggest synergy between pazopanib and cytotoxic chemotherapy, forming the rationale for the current trial with neoadjuvant pazopanib added to chemoradiation.
According to the investigators, the trial (ARST1321) is the first ever collaborative study codeveloped, written, and conducted by pediatric (Children’s Oncology Group) and adult (NRG Oncology) cancer cooperative groups (NCT02180867). Aaron R. Weiss, MD, of the Maine Medical Center in Portland and study cochair, presented the data for the chemotherapy arms at ASCO. The primary objectives of the study were to determine the feasibility of preoperative chemoradiation with or without pazopanib and to compare the rates of complete pathologic response in patients receiving radiation or chemoradiation with or without pazopanib. Pathologic necrosis rates of 90% or better have been found to be predictive of outcome in STS.
Patients with metastatic or non-metastatic NRSTS were eligible to enroll if they had initially unresectable extremity or trunk tumors with the expectation that they would be resectable after therapy. Patients had to be 2 years or older— there was no upper age limit—and had to be able to swallow a tablet whole. The dose-finding phase of the study determined the pediatric dose to be 350 mg/m2 and the adult dose to be 600 mg/m2, both taken orally and once daily. Patients in the chemotherapy cohort were then randomized to receive chemotherapy—ifosfamide and doxorubicin—with or without pazopanib. At 4 weeks, patients in both arms received preoperative radiotherapy (45 Gy in 25 fractions), and at week 13, surgery of the primary site if they did not have progressive disease. After surgery, patients received continuation therapy with or without pazopanib according to their randomization arm. Upon completion and recovery from the continuation therapy, patients could receive surgery/radiotherapy of their metastatic sites.
Results
As of the June 30, 2018, cutoff, 81 patients were enrolled on the chemotherapy arms: 42 in the pazopanib plus chemoradiation arm and 39 in the chemoradiation-only arm. Sixty-one percent of all patients were 18 years or older, and the median age was 20.3 years. Most patients (73%) did not have metastatic disease, and the major histologies represented were synovial sarcoma (49%) and undifferentiated pleomorphic sarcoma (25%).
At week 13, patients in the pazopanib arm showed significant improvement, with 14 (58%) of those evaluated having pathologic necrosis of at least 90%, compared with 4 (22%) in the chemoradiation-only arm (P=.02). The study was closed to further accrual.
Eighteen patients were not evaluable for pathologic response and 21 were pending pathologic evaluation at week 13. Radiographic response rates were not statistically significant on either arm. No complete responses (CR) were achieved in the pazopanib arm, but 14 patients (52%) achieved a partial response (PR) and 12 (44%) had stable disease (SD). In the chemoradiation-only arm, 2 patients (8%) achieved a CR, 12 (50%) a PR, and 8 (33%) SD. Fifteen patients in each arm were not evaluated for radiographic response.
The pazopanib arm experienced more febrile neutropenia and myelotoxicity during induction and continuation phases than the chemoradiation-only arm. In general, investigators indicated pazopanib combined with chemoradiation was well tolerated and no unexpected toxicities arose during the trial.
In the post-presentation discussion, Dr. Raphael E. Pollock, MD, PhD, of The Ohio State University, called it a tremendous challenge to interdigitate primary local therapies in systemic approaches, particularly in the neoadjuvant context. He pointed out that in an earlier study, a 95% to 100% necrosis level was needed to achieve a significant positive impact on outcomes and perhaps a subsequent prospective trial could determine the best level. He questioned whether the availability of only 60% of patient responses could affect the conclusions and whether the high number of toxicities (73.8% grade 3/4 with pazopanib) might be too high to consider the treatment for most patients, given the intensity of the regimen.
SOURCE: Weiss AR, et al. J Clin Oncol 37, 2019 (suppl; abstr 11002)
The study was sponsored by the National Cancer Institute.
Drs. Weiss and Pollock had no relationships with commercial interests to disclose. A few investigators disclosed advisory, consulting, or research roles with pharmaceutical companies, including one who received institutional research funding from Novartis (pazopanib).
Gemcitabine Plus Pazopanib a Potential Alternative in STS
In a phase 2 study of gemcitabine with pazopanib (G+P) or gemcitabine with docetaxel (G+D), investigators concluded the combination with pazopanib can be considered an alternative to that with docetaxel in select patients with advanced soft tissue sarcoma (STS). They reported similar progression-free survival (PFS) and rate of toxicity for the two regimens. Neeta Somaiah, MD, of the University of Texas MD Anderson Cancer Center in Houston, presented the findings of the investigator-initiated effort (NCT01593748) at ASCO.
The objective of the study, conducted at 10 centers across the United States, was to examine the activity of pazopanib when combined with gemcitabine as an alternative to the commonly used gemcitabine plus docetaxel regimen. Pazopanib is a multi-tyrosine kinase inhibitor with efficacy in non-adipocytic STS. Adult patients with metastatic or locally advanced non-adipocytic STS with ECOG performance of 0 or 1 were eligible. Patients had to have received prior anthracycline exposure unless it was contraindicated. The 1:1 randomization included stratification for pelvic radiation and leiomyosarcoma histology, which was felt to have a higher response rate with the pazopanib regimen.
The investigators enrolled 90 patients, 45 in each arm. Patients were a mean age of 56 years, and there was no difference in age or gender distribution between the arms. Patients with leiomyosarcoma (31% overall) or prior pelvic radiation (11% overall) were similar between the arms. The overall response rate using RECIST 1.1 criteria was partial response (PR) in 8 of 44 evaluable patients (18%) in the G+D arm and 5 of 43 evaluable patients (12%) in the G+P arm. Stable disease (SD) was observed in 21 patients (48%) in the G+D arm and 24 patients (56%) in the G+P arm. This amounted to a clinical benefit rate (PR + SD) of 66% and 68% for the G+D and G+P arms, respectively (Fisher’s exact test, P>.99). The median PFS was 4.1 months on both arms and the difference in median overall survival— 15.9 months in the G+D arm and 12.4 months in the G+P arm—was not statistically significant.
Adverse events (AEs) of grade 3 or higher occurred in 19.9% of patients on G+D and 20.6% on G+P. Serious AEs occurred in 33% (G+D) and 22% (G+P). Dose reductions were necessary in 80% of patients on G+P and doses were held in 93%. Dr. Somaiah explained that this may have been because the starting dose of gemcitabine and pazopanib (1000 mg/m2 of gemcitabine on days 1 and 8 and 800 mg of pazopanib) was “probably higher than what we should have started at.” The rate of doses held was also higher in the pazopanib arm (93%) compared with the docetaxel arm (58%). This was likely because pazopanib was a daily dosing, so if there was a toxicity it was more likely to be held than docetaxel, she observed. Grade 3 or higher toxicities occurring in 5% or more of patients in either arm consisted generally of cytopenias and fatigue. The G+P arm experienced a high amount of neutropenia, most likely because this arm did not receive granulocyte-colony stimulating factor (GCSF) support, as opposed to the G+D arm.
Dr. Somaiah pointed out that the 12% response rate for the G+P combination is similar to what has been previously presented and higher than single-agent gemcitabine or pazopanib, but not higher than the G+D combination. The PFS of 4.1 months was less than anticipated, she added, but it was similar on both arms. The investigators believe the G+P combination warrants further exploration.
SOURCE: Somaiah N, et al. J Clin Oncol 37, 2019 (suppl; abstr 11008)
The study was sponsored by the Medical University of South Carolina, with Novartis as collaborator.
Dr. Somaiah disclosed Advisory Board roles for Blueprint, Deciphera, and Bayer. Abstract coauthors disclosed advisory/consulting roles or research funding from various commercial interests, including Novartis (pazopanib) and Pfizer (gemcitabine).
rEECur Trial Finding Optimal Chemotherapy Regimen for Ewing Sarcoma
Interim results of the first and largest randomized trial in patients with refractory or recurrent Ewing sarcoma (ES), the rEECur trial, are guiding the way to finding the optimal chemotherapy regimen to treat the disease. Until now, there has been little prospective evidence and no randomized data to guide treatment choices in relapsed or refractory patients, and hence no real standard of care, according to the presentation at ASCO. Several molecularly targeted therapies are emerging, and they require a standardized chemotherapy backbone against which they can be tested.
The rEECur trial (ISRCTN36453794) is a multi-arm, multistage phase 2/3 “drop-a-loser” randomized trial designed to find the standard of care. The trial compares 4 chemotherapy regimens to each other and drops the least effective one after 50 patients per arm are enrolled and evaluated. The 3 remaining regimens continue until at least 75 patients on each arm are enrolled and evaluated, and then another arm would be dropped. The 2 remaining regimens continue to phase 3 evaluation. Four regimens are being tested at 8 centers in 17 countries: topotecan/ cyclophosphamide (TC), irinotecan/temozolomide (IT), gemcitabine/docetaxel (GD), and ifosfamide (IFOS). The primary objective is to identify the optimal regimen based on a balance between efficacy and toxicity. Martin G. McCabe, MB BChir, PhD, of the University of Manchester in the United Kingdom, presented the results on behalf of the investigators of the rEECur trial.
Results
Two hundred twenty patients 4 years or older and younger than 50 years with recurrent or refractory histologically confirmed ES of bone or soft tissue were randomized to receive GD (n=72) or TC, IT, or IFOS (n=148). Sixty-two GD patients and 123 TC/IT/IFOS patients were included in the primary outcome analysis. Patients were predominantly male (70%), with a median age of 19 years (range, 4 to 49). About two-thirds (67.3%) were post-pubertal. Most patients (85%) were primary refractory or experienced their first disease recurrence, and 89% had measurable disease.
Investigators assessed the primary outcome of objective response after 4 cycles of therapy and found 11% of patients treated with GD responded compared to 24% in the other 3 arms combined. When they subjected the data to Bayesian analysis, there was a 25% chance that the response rate in the GD arm was better than the response in Arm A, a 2% chance that it was better than Arm B, and a 3% chance that it was better than Arm C. Because this study was still blinded at the time of the presentation, investigators didn’t know which regimen constituted which arm. The probability that response favored GD, however, was low.
The investigators observed no surprising safety findings. Eighty-five percent of all patients experienced at least 1 adverse event. Most frequent grade 3‐5 events consisted of pneumonitis (50%, 60%), neutropenic fever (17%, 25%), and diarrhea (0, 12%) in GD and the combined 3 arms, respectively. Grade 3 events in the GD arm were lower than in the other 3 arms combined. There was 1 toxic death attributed to neutropenic sepsis in 1 of the 3 blinded arms.
Median progression-free survival (PFS) for all patients was approximately 5 months. Bayesian analysis suggested there was a low probability that GD was more effective than the other 3 arms: a 22% chance that GD was better than Arm A, a 3% chance that it was better than Arm B, and a 7% chance that it was better than Arm C. Bayesian analysis also suggested there was a probability that OS favored GD. Because the trial directs only the first 4 or 6 cycles of treatment and the patients receive more treatment after trial-directed therapy, investigators were not fully able to interpret this.
Data suggested GD is a less effective regimen than the other 3 regimens both by objective response rate and PFS, so GD has been dropped from the study. Investigators already had more than 75 evaluable patients in each of the 3 arms for the second interim analysis to take place. In a discussion following the presentation, Jayesh Desai, FRACP, of Peter MacCallum Cancer Centre in Melbourne, Australia, called this study a potentially practice-changing trial at this early stage, noting that the GD combination will be de-prioritized in practice based on these results.
SOURCE: McCabe MG, et al. J Clin Oncol 37, 2019 (suppl; abstr 11007)
The rEECur trial is sponsored by the University of Birmingham (UK) and received funding from the European Union’s Seventh Framework Programme under a grant agreement.
Dr. McCabe disclosed no conflicts of interest. Other authors disclosed consulting, advisory roles, or research funding from numerous pharmaceutical companies, including Lilly (gemcitabine) and Pfizer (irinotecan). Dr. Desai disclosed a consulting/advisory role and institutional research funding from Lilly.
Abemaciclib Meets Primary Endpoint in Phase 2 Trial of DDLS
The newer and more potent CDK4 inhibitor, abemaciclib, met its primary endpoint in the investigator-initiated, single-center, single-arm, phase 2 trial in patients with advanced progressive dedifferentiated liposarcoma (DDLS). Twenty-two patients (76%) achieved progression-free survival (PFS) at 12 weeks for a median PFS of 30 weeks. A subset of patients experienced prolonged clinical benefit, remaining on study with stable disease for over 900 days. The study (NCT02846987) was conducted at Memorial Sloan Kettering Cancer Center (MSKCC) in New York and Mark A. Dickson, MD, presented the results at ASCO.
Of three agents in the clinic with the potential to target CDK4 and CDK6—palbociclib, ribociclib, and abemaciclib— abemaciclib is more selective for CDK4 than CDK6. CDK4 amplification occurs in more than 90% of well-differentiated and dedifferentiated liposarcomas. Abemaciclib also has a different side effect profile, with less hematologic toxicity than the other 2 agents. The current study was considered positive if 15 patients or more of a 30-patient sample size were progression- free at 12 weeks.
Results
Thirty patients, 29 evaluable, with metastatic or recurrent DDLS were enrolled and treated with abemaciclib 200 mg orally twice daily between August 2016 and October 2018. Data cutoff for the presentation was the first week of May 2019. Patients were a median of 62 years, 60% were male, and half had no prior systemic treatment. Prior systemic treatments for those previously treated included doxorubicin, olaratumab, gemcitabine, docetaxel, ifosfamide, eribulin, and trabectedin. For 87%, the primary tumor was in their abdomen or retroperitoneum.
Toxicity was as expected with this class of agent, according to the investigators. The most common grades 2 and 3 toxicities, respectively, possibly related to the study drug, occurring in more than 1 patient included anemia (70%, 37%), thrombocytopenia (13%, 13%), neutropenia (43%, 17%), and lymphocyte count decreased (23%, 23%). Very few of these adverse events were grade 4—none for anemia, and 3% each for thrombocytopenia, neutropenia, and lymphocyte count decreased. Diarrhea of grades 2 and 3 occurred in 27% and 7% of patients, respectively, and was managed well with loperamide.
In addition to reaching the primary endpoint of 15 patients or more achieving PFS at 12 weeks, 1 patient had a confirmed partial response (PR) and another an unconfirmed PR. At data cutoff, 11 patients remained on study with stable disease or PR. The investigators conducted correlative studies that indicated all patients had CDK4 and MDM2 amplification with no loss of retinoblastoma tumor suppressor. They observed an inverse correlation between CDK4 amplification and PFS—the higher the level of CDK4 amplification, the shorter the PFS. They also found additional genomic alterations, including JUN, GLI1, ARID1A, TERT, and ATRX. TERT amplification was also associated with shorter PFS. Based on these findings, the investigators believe a phase 3 study of abemaciclib in DDLS is warranted.
Winette van der Graaf, MD, PhD, of the Netherlands Cancer Institute in Amsterdam, in the discussion following the presentation, concurred that it is certainly time for a multicenter phase 3 study of CDK4 inhibitors in DDLS, and a strong international collaboration is key to conducting such studies, particularly in rare cancers. On a critical note, Dr. van der Graaf expressed concern that no patient-reported outcomes were measured after 120 patients, including those in previous studies, were treated on palbociclib and abemaciclib. Given that the toxicities of the CDK4 inhibitors are quite different, she recommended including patient-reported outcomes in future studies using validated health-related quality-of-life instruments.
SOURCE: Dickson MA, et al. J Clin Oncol 37, 2019 (suppl; abstr 11004)
The study was sponsored by Memorial Sloan Kettering Cancer Center, with the study collaborator, Eli Lilly and Company.
Dr. Dickson disclosed research funding from Lilly, the company that provided the study drug. Dr. van der Graaf had no relevant relationships to disclose. Abstract coauthors had consulting/advisory roles or research funding from various companies, including Lilly.
nab-Sirolimus Provides Benefits in Advanced Malignant PEComa
In a prospective phase 2 study of nab-sirolimus in advanced malignant perivascular epithelioid cell tumor (PEComa), the mTOR inhibitor achieved an objective response rate (ORR) of 42% with an acceptable safety profile, despite using relatively high doses of nab-sirolimus compared to other mTOR inhibitors. Activation of the mTOR pathway is common in PEComa, and earlier case reports had indicated substantial clinical benefit with mTOR inhibitor treatment. nab-Sirolimus (ABI-009) is a novel intravenous mTOR inhibitor consisting of nanoparticles of albumin-bound sirolimus. It has significantly higher anti-tumor activity than oral mTOR inhibitors and greater mTOR target suppression at an equal dose. Andrew J. Wagner, MD, PhD, of the Dana-Farber Cancer Institute in Boston, presented the findings of AMPECT (NCT02494570)—Advanced Malignant PEComa Trial—at ASCO.
Investigators enrolled 34 patients 18 years or older with histologically confirmed malignant PEComa. Patients could not have had prior mTOR inhibitors. They received infusions of 100 mg/m2 nab-sirolimus on days 1 and 8 every 21 days until progression or unacceptable toxicity. Patients were a median age of 60 years and 44% were 65 or older; 82% were women, which is typical of the disease. Most patients (88%) had no prior systemic therapy for advanced PEComa.
Results
The drug was well tolerated, with toxicities similar to those of oral mTOR inhibitors. Treatment-related adverse events (TRAEs) occurring in 25% or more of patients were mostly grade 1 or 2 toxicities. Hematologic TRAEs included anemia (47%) and thrombocytopenia (32%) of any grade. Nonhematologic events of any grade included stomatitis/ mucositis (74%), dermatitis/rash (65%), fatigue (59%), nausea (47%), and diarrhea (38%), among others. A few grade 3 events occurred on study, most notably stomatitis/mucositis (18%). Severe adverse events (SAEs) were also uncommon, occurring in 7 of 34 patients (21%). Pneumonitis is common in orally administered mTOR inhibitors; 6 patients (18%) treated with nab-sirolimus had grade 1 or 2 pneumonitis.
Of the 31 evaluable patients, 13 (42%) had an objective response, all of which were partial responses (PR). Eleven (35%) had stable disease and 7 (23%) had progressive disease. The disease control rate, consisting of PR and stable disease, was 77%. The median duration of response had not been reached as of the data cutoff on May 10, 2019. At that time, it was 6.2 months (range, 1.5 to 27.7+). The median time to response was 1.4 months and the median progression-free survival (PFS) was 8.4 months. The PFS rate at 6 months was 61%. Three patients had received treatment for over a year and another 3 patients for more than 2 years.
Correlation with biomarkers
Of the 25 patients who had tissue suitable for next-generation sequencing, 9 had TSC2 mutations, 5 had TSC1 mutations, and 11 had neither mutation. Strikingly, 9 of 9 patients with TSC2 mutations developed a PR, while only 1 with a TSC1 mutation responded. One patient with no TSC1/2 mutation also responded and 2 patients with unknown mutational status responded. The investigators also analyzed pS6 status by immunohistochemistry—pS6 is a marker of mTOR hyperactivity. Twenty- five patient samples were available for analysis. Eight of 8 patients who were negative for pS6 staining did not have a response, while 10 of 17 (59%) who were pS6-positive had a PR.
In the discussion that followed, Winette van der Graaf, MD, of the Netherlands Cancer Institute in Amsterdam, noted that this study showed that biomarkers can be used for patient selection, although TSC2 mutations are not uniquely linked with response. She indicated a comparator with sirolimus would have been of great interest.
SOURCE: Wagner AJ, et al. J Clin Oncol 37, 2019 (suppl; abstr 11005).
The study was sponsored by Aadi Bioscience, Inc., and funded in part by a grant from the FDA Office of Orphan Products Development (OOPD).
Disclosures relevant to this presentation include contininstitutional research funding from Aadi Bioscience for Dr. Wagner and a few other abstract coauthors. Several coauthors are employed by Aadi Bioscience and have stock or other ownership interests. Dr. van der Graaf had nothing to disclose.
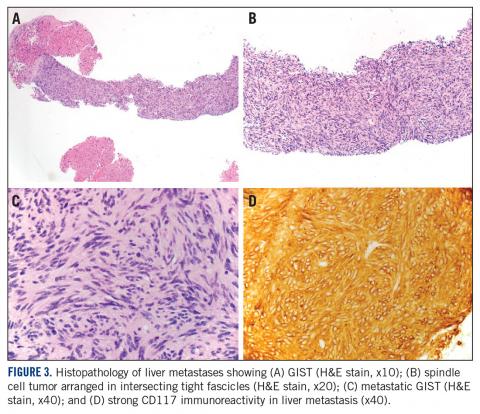
Cabozantinib Achieves Disease Control in GIST
The phase 2 EORTC 1317 trial, known as CaboGIST (NCT02216578), met its primary endpoint of progression-free survival (PFS) at 12 weeks in patients with metastatic gastrointestinal stromal tumor (GIST) treated with the tyrosine kinase inhibitor (TKI) cabozantinib. Twenty-four (58.5%) of the 41 patients in the primary study population, and 30 (60%) of the entire 50-patient population, were progression-free at 12 weeks. The study needed 21 patients to be progression- free for cabozantinib to warrant further exploration in GIST patients.
Cabozantinib is a multitargeted TKI inhibiting KIT, MET, AXL, and VEGFR2, which are potentially relevant targets in GIST. In patient-derived xenografts of GIST, cabozantinib demonstrated activity in imatinib-sensitive and -resistant models and inhibited tumor growth, proliferation, and angiogenesis. Additional preclinical experience suggested that cabozantinib could potentially be used as a potent MET inhibitor, overcoming upregulation of MET signaling that occurs with imatinib treatment of GIST, known as the kinase switch.
This investigator-initiated study had as its primary objective assessment of the safety and activity of cabozantinib in patients with metastatic GIST who had progressed on imatinib and sunitinib. The patients could not have been exposed to other KIT- or PDGFR-directed TKIs, such as regorafenib. Secondary objectives included the assessment of cabozantinib in different mutational subtypes of GIST. Patients received cabozantinib tablets once daily until they experienced no further clinical benefit or became intolerant to the drug or chose to discontinue therapy. Fifty patients started treatment between February 2017 and August 2018. All were evaluable for the primary endpoint, and one-third of patients contininstitutional cabozantinib treatment as of the database cutoff in January 2019.
Results
Patients were a median age of 63 years. Virtually all patients (92%) had prior surgery and only 8% had prior radiotherapy. The daily cabozantinib dose was a median 47.2 mg and duration of treatment was a median 20.4 weeks. No patient discontinued treatment due to toxicity, but 88% discontinued due to disease progression.
Safety signals were the same as for other indications in which cabozantinib is used. Almost all patients (94%) had at least 1 treatment-related adverse event of grades 1‐4, including diarrhea (74%), palmar-plantar erythrodysesthesia (58%), fatigue (46%), and hypertension (46%), which are typical of treatment with cabozantinib. Hematologic toxicities in this trial were clinically irrelevant, according to the investigators, consisting of small numbers of grades 2‐3 anemia, lymphopenia, white blood cell count abnormality, and neutropenia. Biochemical abnormalities included grades 3 and 4 hypophosphatemia, increased grades 3 and 4 gamma-glutamyl transferase, grade 3 hyponatremia, and grade 3 hypokalemia, in 8% or more of patients.
Overall survival was a median 14.4 months, with 16 patients still on treatment at the time of data cutoff. Twenty- four patients were progression-free at week 12, satisfying the study decision rule for clinical benefit. Median duration of PFS was 6.0 months. Seven patients (14%) achieved a confirmed partial response (PR) and 33 (66%) achieved stable disease (SD). Nine patients had progressive disease as their best response, 3 of whom had some clinical benefit. Forty patients (80%) experienced a clinical benefit of disease control (PR + SD).
An analysis of the relationship of genotype, duration, and RECIST response showed objective responses in patients with primary exon 11 mutations, with exon 9 mutations, and with exon 17 mutations, and in 2 patients without any known mutational information at the time of the presentation. Patients with stable disease were spread across all mutational subsets in the trial. The investigators suggested the definitive role of MET and AXL inhibition in GIST be assessed further in future clinical trials.
SOURCE: Schöffski P, et al. J Clin Oncol 37, 2019 (suppl; abstr 11006).
The study was sponsored by the European Organization for Research and Treatment of Cancer (EORTC).
Presenting author, Patrick Schöffski, MD, of KU Leuven and Leuven Cancer Institute in Belgium, disclosed institutional relationships with multiple pharmaceutical companies for consulting and research funding, including research funding from Exelixis, the developer of cabozantinib. No other abstract coauthor disclosed a relationship with Exelixis.
Larotectinib Effective in TRK Fusion Cancers
Pediatric patients with tropomyosin receptor kinase (TRK) fusions involving NTRK1, NTRK2, and NTRK3 genes had a high response rate with durable responses and a favorable safety profile when treated with larotrectinib, according to a presentation at ASCO. In this pediatric subset of children and adolescents from the SCOUT and NAVIGATE studies, the overall response rate (ORR) was 94%, with a 35% complete response (CR), 59% partial response (PR), and 6% stable disease as of the data cutoff at the end of July 2018.
TRK fusion cancer is a rare malignancy seen in a wide variety of adult and childhood tumor types. Among pediatric malignancies, infantile fibrosarcoma and congenital mesoblastic nephroma are rare, but have high NTRK gene fusion frequency. Other sarcomas and pediatric high-grade gliomas, for example, are less rare but have low NTRK gene fusion frequency. Larotrectinib, a first-in-class and the only selective TRK inhibitor, has high potency against the 3 NTRK genes that encode the neurotrophin receptors. It is highly selective and has limited inhibition of the other kinases. The US Food and Drug Administration approved larotrectinib for the treatment of patients with solid tumors harboring NTRK fusions. Cornelis Martinus van Tilburg, MD, of the Hopp Children’s Cancer Center, Heidelberg University Hospital, and German Cancer Research Center in Heidelberg, Germany, presented the findings.
Investigators enrolled 38 children and adolescents younger than 18 years from the SCOUT (NCT02637687) and NAVIGATE (NCT02576431) studies of larotrectinib who had non-central nervous system (CNS) TRK fusion cancers. Not all patients had the recommended phase 2 dose, Dr. van Tilburg pointed out, but most did. Hence, 29 of the 38 patients received the 100 mg/m2 twice-daily, phase 2 dose until progression, withdrawal, or unacceptable toxicity.
Patients were young, with a median age of 2.3 years (range, 0.1 to 14.0 years). Almost two-thirds (61%) had prior surgery, 11% had prior radiotherapy, and 68% had prior systemic therapy. For 12 patients, larotrectinib was their first systemic therapy. The predominant tumor types were infantile fibrosarcoma (47%) and other soft tissue sarcoma (42%). And 47% of patients had NTRK3 fusions with ETV6, most of which were infantile fibrosarcoma.
Efficacy
Thirty-four patients were evaluable, and 32 had a reduction in tumor size, for an ORR of 94%, CR of 35%, and PR of 59%. Two patients with infantile fibrosarcoma had pathologic CRs—after treatment, no fibroid tissue in the tumors could be found. Median time to response was 1.8 months, median duration of treatment was 10.24 months, and 33 of 38 patients (87%) remained on treatment or underwent surgery with curative intent. As of the data cutoff of July 30, 2018, the secondary endpoints were not yet reached. However, 84% of responders were estimated to have a response duration of a year or more, and progression-free and overall survival looked very promising, according to Dr. van Tilburg.
Adverse events were primarily grades 1 and 2. The grades 3 and 4 treatment-related adverse events were quite few and consisted of increased alanine aminotransferase, decreased neutrophil count, and nausea. Longer follow-up of the patient safety profile is required, particularly since NTRK has multiple roles in neurodevelopment. The investigators recommended that routine testing for NTRK gene fusions in pediatric patients with cancer be conducted in appropriate clinical contexts.
In a discussion after the presentation, Daniel Alexander Morgenstern, MB BChir, PhD, of Great Ormond Street Hospital, London, UK, said that in many ways, the NTRK inhibitors have become the new poster child for precision oncology in pediatrics because of “these really spectacular results” with larotrectinib [and entrectinib]. One of the questions he raised regarding larotrectinib was the issue of CNS penetration, since patients with CNS cancer were not enrolled in the trial and preclinical data suggest limited CNS penetration for larotrectinib.
SOURCE: van Tilburg CM, et al. J Clin Oncol 37, 2019 (suppl; abstr 10010).
The studies were funded by Loxo Oncology, Inc., and Bayer AG.
Disclosures relevant to this presentation include consulting or advisory roles for Bayer for Drs. van Tilburg and Morgenstern. A few coauthors also had consulting/advisory roles or research funding from various companies, including Loxo and Bayer.
Behind Olaratumab's Phase 3 Disappointment
ANNOUNCE, the phase 3 trial designed to confirm the clinical benefit of olaratumab in patients with advanced soft tissue sarcoma (STS), failed to meet its primary endpoint of overall survival (OS) in all STS histologies and the leiomyosarcoma population. The previous phase 1b/2 signal-finding study of olaratumab had achieved an unprecedented improvement in OS, and the US Food and Drug Administration (FDA) awarded olaratumab accelerated approval in October 2016. By December 2018, olaratumab received additional accelerated, conditional, and full approvals in more than 40 countries worldwide. William D. Tap, MD, chief of the Sarcoma Medical Oncology Service at Memorial Sloan Kettering Cancer Center in New York, presented the phase 3 results and provided some explanations for the findings during the plenary session at ASCO.
ANNOUNCE (NCT02451943), which was designed and enrolled prior to olaratumab receiving accelerated approval, opened in September 2015 and completed accrual 10 months later in July 2016. Investigators randomized and treated 509 patients with advanced STS not amenable to curative therapy, 258 patients in the olaratumab-doxorubicin arm and 251 in the placebo-doxorubicin arm. Most patients (46%) had leiomyosarcoma, followed by liposarcoma (18%), pleomorphic sarcoma (13%), and 24% of the patient population had 26 unique histologies. Three-quarters of the patients had no prior systemic therapy.
Results
As of the data cutoff on December 5, 2018, there were no survival differences in the intention-to-treat population, in the total STS population nor in the leiomyosarcoma subpopulation, with olaratumab-doxorubicin compared to placebo-doxorubicin. For the total STS population, median OS with olaratumab- doxorubicin was 20.4 months and with placebo-doxorubicin 19.7 months. “This is the highest survival rate described to date in any phase 3 sarcoma study,” Dr. Tap said. “It is of particular interest as ANNOUNCE did not mandate treatment in the first line.” In the leiomyosarcoma population, median OS was 21.6 months with olaratumab and 21.9 months with placebo. The secondary endpoints of progression-free survival (PFS), overall response rate, and disease control rate did not favor olaratumab either.
Investigators are examining the relationship between PDGFRα expression and OS in ANNOUNCE. PDGFRα-positive tumors tended to do worse with olaratumab than PDGFRα-negative tumors. The investigators noticed a 6-month difference in OS between these populations favoring PDGFRα-negative tumors. Additional biomarker analyses are ongoing.
A large and concerted effort is underway, Dr. Tap said, to understand the results of the ANNOUNCE study alone and in context with the phase 1b/2 study. “There are no noted discrepancies in study conduct or data integrity which could explain these findings or the differences between the two studies.”
Possible explanations
The designs of the phase 1b/2 and phase 3 studies had some important differences. The phase 1b/2 study was a small, open-label, US-centric study (10 sites) that did not include a placebo or subtype- specified analyses. Its primary endpoint was PFS, it did not have a loading dose of olaratumab, and it specified the timing of dexrazoxane administration after 300 mg/m2 of doxorubicin.
ANNOUNCE, on the other hand, was a large (n=509), international (110 study sites), double-blind, placebo-controlled trial that had outcomes evaluated in STS and leiomyosarcoma. Its primary endpoint was OS, it had a loading dose of olaratumab of 20 mg/kg, and there was no restriction as to the timing of dexrazoxane administration.
Dr. Tap pointed out that in ANNOUNCE it was difficult to predict or control for factors that may have had an unanticipated influence on outcomes, such as albumin levels as a surrogate for disease burden and behavior of PDGFRα status. It is possible, he said, that olaratumab has no activity in STS and that the phase 1b/2 results were due to, among other things, the small sample size, numerous represented histologies with disparate clinical behavior, and the effect of subtype-specific therapies on overall survival, given subsequently or even by chance. On the other hand, it is also possible, he said, that olaratumab has some activity in STS, with outcomes being affected by the heterogeneity of the study populations, differences in trial design, and the performance of the ANNOUNCE control arm. Whatever the case, he said, accelerated approval allowed patients to have access to a potentially life-prolonging drug with little added toxicity.
Discussion
In the expert discussion following the presentation, Jaap Verweij, MD, PhD, of Erasmus University Medical Center in Rotterdam, The Netherlands, congratulated the investigators for performing the study at an unprecedented pace. He commented that lumping STS subtypes together is problematic, as different histological subtypes behave as though they are different diseases. Small numbers of each tumor subtype and subtypes with slow tumor growth can impact trial outcomes. In the phase 1b/2 and phase 3 trials, 26 different subtypes were represented in each study. Dr. Verweij pointed out this could have made a big difference in the phase 1b/2 study, in which there were only 66 patients in each arm.
It is striking to note, he said, that without exception, phase 2 randomized studies in STS involving doxorubicin consistently overestimated and wrongly predicted PFS in the subsequent phase 3 studies. And the situation is similar for OS. The results of the ANNOUNCE study are no exception, he added. “Taken together, these studies indicate that phase 2 studies in soft tissue sarcomas, certainly those involving additions of drugs to doxorubicin, even if randomized, should be interpreted with great caution,” he said.
SOURCE: Tap WD, et al. J Clin Oncol 37, 2019 (suppl; abstr LBA3)
The study was sponsored by Eli Lilly and Company.
Dr. Tap reported research funding from Lilly and Dr. Verweij had nothing to report related to this study. Abstract coauthors disclosed numerous financial relationships, including consulting/advisory roles and/or research funding from Lilly, and several were employed by Lilly.
Addition of Temozolomide May Improve Outcomes in RMS
Investigators from the European Pediatric Soft Tissue Sarcoma Study Group (EpSSG) found that the addition of temozolomide (T) to vincristine and irinotecan (VI) may improve outcomes in adults and children with relapsed or refractory rhabdomyosarcoma (RMS). Principal investigator of the study, Anne Sophie Defachelles, MD, pediatric oncologist at the Centre Oscar Lambret in Lille, France, presented the results on behalf of the EpSSG.
The primary objective of the study was to evaluate the efficacy of VI and VIT regimens, defined as objective response (OR)—complete response (CR) plus partial response (PR)—after 2 cycles. Secondary objectives were progression-free survival (PFS), overall survival (OS), and safety in each arm, and the relative treatment effect of VIT compared to VI in terms of OR, survival, and safety.
The international, randomized (1:1), open-label, phase 2 trial (VIT-0910; NCT01355445) was conducted at 37 centers in 5 countries. Patients ages 6 months to 50 years with RMS were eligible. They could not have had prior irinotecan or temozolomide. A 2015 protocol amendment limited enrollment to patients at relapse and increased the enrollment goal by 40 patients. After the 2015 amendment, patients with refractory disease were no longer eligible.
From January 2012 to April 2018, investigators enrolled 120 patients, 60 on each arm. Two patients in the VI arm were not treated. Patients were a median age of 10.5 years in the VI arm and 12 years in the VIT arm, 92% (VI) and 87% (VIT) had relapsed disease, 8% (VI) and 13% (VIT) had refractory disease, and 55% (VI) and 68% (VIT) had metastatic disease at study entry.
Results
Patients achieved an OR rate of 44% (VIT) and 31% (VI) for the whole population, one-sided P value <.0001. The adjusted odds ratio for the whole population was 0.50, P=.09. PFS was 4.7 months (VIT) and 3.2 months (VI), “a nearly significant reduction in the risk of progression,” Dr. Defachelles noted. Median OS was 15.0 months (VIT) and 10.3 months (VI), which amounted to “a large and significant reduction in the risk of death,” she said. The adjusted hazard ratio was 0.55, P=.006.
Adverse events of grade 3 or higher were more frequent in the VIT arm, with hematologic toxicity the most frequent (81% for VIT, 59% for VI), followed by gastrointestinal adverse events. “VIT was significantly more toxic than VI,” Dr. Defachelles observed, “but the toxicity was manageable.”
“VIT is now the standard treatment in Europe for relapsed rhabdomyosarcoma and will be the control arm in the multiarm, multistage RMS study for relapsed patients,” she said.
In a discussion following the presentation, Lars M. Wagner, MD, of Cincinnati Children’s Hospital, pointed out that the study was not powered for the PFS and OS assessments. These were secondary objectives that should be considered exploratory. Therefore, he said, the outcome data is not conclusive. The role of temozolomide in RMS is also unclear, given recent negative results in patients with newly diagnosed metastatic RMS (Malempati et al, Cancer 2019). And he said it’s uncertain how these results apply to patients who received irinotecan upfront for RMS.
SOURCE: Defachelles AS, et al. J Clin Oncol 37, 2019 (suppl; abstr 10000)
The study was sponsored by Centre Oscar Lambret and SFCE (Société Française de Lutte contre les Cancers et Lucémies de l’Enfant et de l’Adolescent) served as collaborator.
Drs. Defachelles and Wagner had no relationships to disclose. A few coauthors had advisory/consulting or speaker roles for various commercial interests, including two for Merck (temozolomide).
Pazopanib Increases Pathologic Necrosis Rates in STS
Pazopanib added to a regimen of preoperative chemoradiation in non-rhabdomyosarcoma soft tissue sarcoma (NRSTS) significantly increased the rate of near-complete pathologic response in both children and adults with intermediate or high-risk disease. Pazopanib, a multitargeted receptor tyrosine kinase inhibitor, works in multiple signaling pathways involved in tumorigenesis— VEGFR-1, -2, -3, PDGFRα/β, and c-kit. A phase 3 study demonstrated significant improvement in progression-free survival (PFS) in advanced STS patients and was the basis for its approval in the US and elsewhere for treatment of this patient population. Preclinical data suggest synergy between pazopanib and cytotoxic chemotherapy, forming the rationale for the current trial with neoadjuvant pazopanib added to chemoradiation.
According to the investigators, the trial (ARST1321) is the first ever collaborative study codeveloped, written, and conducted by pediatric (Children’s Oncology Group) and adult (NRG Oncology) cancer cooperative groups (NCT02180867). Aaron R. Weiss, MD, of the Maine Medical Center in Portland and study cochair, presented the data for the chemotherapy arms at ASCO. The primary objectives of the study were to determine the feasibility of preoperative chemoradiation with or without pazopanib and to compare the rates of complete pathologic response in patients receiving radiation or chemoradiation with or without pazopanib. Pathologic necrosis rates of 90% or better have been found to be predictive of outcome in STS.
Patients with metastatic or non-metastatic NRSTS were eligible to enroll if they had initially unresectable extremity or trunk tumors with the expectation that they would be resectable after therapy. Patients had to be 2 years or older— there was no upper age limit—and had to be able to swallow a tablet whole. The dose-finding phase of the study determined the pediatric dose to be 350 mg/m2 and the adult dose to be 600 mg/m2, both taken orally and once daily. Patients in the chemotherapy cohort were then randomized to receive chemotherapy—ifosfamide and doxorubicin—with or without pazopanib. At 4 weeks, patients in both arms received preoperative radiotherapy (45 Gy in 25 fractions), and at week 13, surgery of the primary site if they did not have progressive disease. After surgery, patients received continuation therapy with or without pazopanib according to their randomization arm. Upon completion and recovery from the continuation therapy, patients could receive surgery/radiotherapy of their metastatic sites.
Results
As of the June 30, 2018, cutoff, 81 patients were enrolled on the chemotherapy arms: 42 in the pazopanib plus chemoradiation arm and 39 in the chemoradiation-only arm. Sixty-one percent of all patients were 18 years or older, and the median age was 20.3 years. Most patients (73%) did not have metastatic disease, and the major histologies represented were synovial sarcoma (49%) and undifferentiated pleomorphic sarcoma (25%).
At week 13, patients in the pazopanib arm showed significant improvement, with 14 (58%) of those evaluated having pathologic necrosis of at least 90%, compared with 4 (22%) in the chemoradiation-only arm (P=.02). The study was closed to further accrual.
Eighteen patients were not evaluable for pathologic response and 21 were pending pathologic evaluation at week 13. Radiographic response rates were not statistically significant on either arm. No complete responses (CR) were achieved in the pazopanib arm, but 14 patients (52%) achieved a partial response (PR) and 12 (44%) had stable disease (SD). In the chemoradiation-only arm, 2 patients (8%) achieved a CR, 12 (50%) a PR, and 8 (33%) SD. Fifteen patients in each arm were not evaluated for radiographic response.
The pazopanib arm experienced more febrile neutropenia and myelotoxicity during induction and continuation phases than the chemoradiation-only arm. In general, investigators indicated pazopanib combined with chemoradiation was well tolerated and no unexpected toxicities arose during the trial.
In the post-presentation discussion, Dr. Raphael E. Pollock, MD, PhD, of The Ohio State University, called it a tremendous challenge to interdigitate primary local therapies in systemic approaches, particularly in the neoadjuvant context. He pointed out that in an earlier study, a 95% to 100% necrosis level was needed to achieve a significant positive impact on outcomes and perhaps a subsequent prospective trial could determine the best level. He questioned whether the availability of only 60% of patient responses could affect the conclusions and whether the high number of toxicities (73.8% grade 3/4 with pazopanib) might be too high to consider the treatment for most patients, given the intensity of the regimen.
SOURCE: Weiss AR, et al. J Clin Oncol 37, 2019 (suppl; abstr 11002)
The study was sponsored by the National Cancer Institute.
Drs. Weiss and Pollock had no relationships with commercial interests to disclose. A few investigators disclosed advisory, consulting, or research roles with pharmaceutical companies, including one who received institutional research funding from Novartis (pazopanib).
Gemcitabine Plus Pazopanib a Potential Alternative in STS
In a phase 2 study of gemcitabine with pazopanib (G+P) or gemcitabine with docetaxel (G+D), investigators concluded the combination with pazopanib can be considered an alternative to that with docetaxel in select patients with advanced soft tissue sarcoma (STS). They reported similar progression-free survival (PFS) and rate of toxicity for the two regimens. Neeta Somaiah, MD, of the University of Texas MD Anderson Cancer Center in Houston, presented the findings of the investigator-initiated effort (NCT01593748) at ASCO.
The objective of the study, conducted at 10 centers across the United States, was to examine the activity of pazopanib when combined with gemcitabine as an alternative to the commonly used gemcitabine plus docetaxel regimen. Pazopanib is a multi-tyrosine kinase inhibitor with efficacy in non-adipocytic STS. Adult patients with metastatic or locally advanced non-adipocytic STS with ECOG performance of 0 or 1 were eligible. Patients had to have received prior anthracycline exposure unless it was contraindicated. The 1:1 randomization included stratification for pelvic radiation and leiomyosarcoma histology, which was felt to have a higher response rate with the pazopanib regimen.
The investigators enrolled 90 patients, 45 in each arm. Patients were a mean age of 56 years, and there was no difference in age or gender distribution between the arms. Patients with leiomyosarcoma (31% overall) or prior pelvic radiation (11% overall) were similar between the arms. The overall response rate using RECIST 1.1 criteria was partial response (PR) in 8 of 44 evaluable patients (18%) in the G+D arm and 5 of 43 evaluable patients (12%) in the G+P arm. Stable disease (SD) was observed in 21 patients (48%) in the G+D arm and 24 patients (56%) in the G+P arm. This amounted to a clinical benefit rate (PR + SD) of 66% and 68% for the G+D and G+P arms, respectively (Fisher’s exact test, P>.99). The median PFS was 4.1 months on both arms and the difference in median overall survival— 15.9 months in the G+D arm and 12.4 months in the G+P arm—was not statistically significant.
Adverse events (AEs) of grade 3 or higher occurred in 19.9% of patients on G+D and 20.6% on G+P. Serious AEs occurred in 33% (G+D) and 22% (G+P). Dose reductions were necessary in 80% of patients on G+P and doses were held in 93%. Dr. Somaiah explained that this may have been because the starting dose of gemcitabine and pazopanib (1000 mg/m2 of gemcitabine on days 1 and 8 and 800 mg of pazopanib) was “probably higher than what we should have started at.” The rate of doses held was also higher in the pazopanib arm (93%) compared with the docetaxel arm (58%). This was likely because pazopanib was a daily dosing, so if there was a toxicity it was more likely to be held than docetaxel, she observed. Grade 3 or higher toxicities occurring in 5% or more of patients in either arm consisted generally of cytopenias and fatigue. The G+P arm experienced a high amount of neutropenia, most likely because this arm did not receive granulocyte-colony stimulating factor (GCSF) support, as opposed to the G+D arm.
Dr. Somaiah pointed out that the 12% response rate for the G+P combination is similar to what has been previously presented and higher than single-agent gemcitabine or pazopanib, but not higher than the G+D combination. The PFS of 4.1 months was less than anticipated, she added, but it was similar on both arms. The investigators believe the G+P combination warrants further exploration.
SOURCE: Somaiah N, et al. J Clin Oncol 37, 2019 (suppl; abstr 11008)
The study was sponsored by the Medical University of South Carolina, with Novartis as collaborator.
Dr. Somaiah disclosed Advisory Board roles for Blueprint, Deciphera, and Bayer. Abstract coauthors disclosed advisory/consulting roles or research funding from various commercial interests, including Novartis (pazopanib) and Pfizer (gemcitabine).
rEECur Trial Finding Optimal Chemotherapy Regimen for Ewing Sarcoma
Interim results of the first and largest randomized trial in patients with refractory or recurrent Ewing sarcoma (ES), the rEECur trial, are guiding the way to finding the optimal chemotherapy regimen to treat the disease. Until now, there has been little prospective evidence and no randomized data to guide treatment choices in relapsed or refractory patients, and hence no real standard of care, according to the presentation at ASCO. Several molecularly targeted therapies are emerging, and they require a standardized chemotherapy backbone against which they can be tested.
The rEECur trial (ISRCTN36453794) is a multi-arm, multistage phase 2/3 “drop-a-loser” randomized trial designed to find the standard of care. The trial compares 4 chemotherapy regimens to each other and drops the least effective one after 50 patients per arm are enrolled and evaluated. The 3 remaining regimens continue until at least 75 patients on each arm are enrolled and evaluated, and then another arm would be dropped. The 2 remaining regimens continue to phase 3 evaluation. Four regimens are being tested at 8 centers in 17 countries: topotecan/ cyclophosphamide (TC), irinotecan/temozolomide (IT), gemcitabine/docetaxel (GD), and ifosfamide (IFOS). The primary objective is to identify the optimal regimen based on a balance between efficacy and toxicity. Martin G. McCabe, MB BChir, PhD, of the University of Manchester in the United Kingdom, presented the results on behalf of the investigators of the rEECur trial.
Results
Two hundred twenty patients 4 years or older and younger than 50 years with recurrent or refractory histologically confirmed ES of bone or soft tissue were randomized to receive GD (n=72) or TC, IT, or IFOS (n=148). Sixty-two GD patients and 123 TC/IT/IFOS patients were included in the primary outcome analysis. Patients were predominantly male (70%), with a median age of 19 years (range, 4 to 49). About two-thirds (67.3%) were post-pubertal. Most patients (85%) were primary refractory or experienced their first disease recurrence, and 89% had measurable disease.
Investigators assessed the primary outcome of objective response after 4 cycles of therapy and found 11% of patients treated with GD responded compared to 24% in the other 3 arms combined. When they subjected the data to Bayesian analysis, there was a 25% chance that the response rate in the GD arm was better than the response in Arm A, a 2% chance that it was better than Arm B, and a 3% chance that it was better than Arm C. Because this study was still blinded at the time of the presentation, investigators didn’t know which regimen constituted which arm. The probability that response favored GD, however, was low.
The investigators observed no surprising safety findings. Eighty-five percent of all patients experienced at least 1 adverse event. Most frequent grade 3‐5 events consisted of pneumonitis (50%, 60%), neutropenic fever (17%, 25%), and diarrhea (0, 12%) in GD and the combined 3 arms, respectively. Grade 3 events in the GD arm were lower than in the other 3 arms combined. There was 1 toxic death attributed to neutropenic sepsis in 1 of the 3 blinded arms.
Median progression-free survival (PFS) for all patients was approximately 5 months. Bayesian analysis suggested there was a low probability that GD was more effective than the other 3 arms: a 22% chance that GD was better than Arm A, a 3% chance that it was better than Arm B, and a 7% chance that it was better than Arm C. Bayesian analysis also suggested there was a probability that OS favored GD. Because the trial directs only the first 4 or 6 cycles of treatment and the patients receive more treatment after trial-directed therapy, investigators were not fully able to interpret this.
Data suggested GD is a less effective regimen than the other 3 regimens both by objective response rate and PFS, so GD has been dropped from the study. Investigators already had more than 75 evaluable patients in each of the 3 arms for the second interim analysis to take place. In a discussion following the presentation, Jayesh Desai, FRACP, of Peter MacCallum Cancer Centre in Melbourne, Australia, called this study a potentially practice-changing trial at this early stage, noting that the GD combination will be de-prioritized in practice based on these results.
SOURCE: McCabe MG, et al. J Clin Oncol 37, 2019 (suppl; abstr 11007)
The rEECur trial is sponsored by the University of Birmingham (UK) and received funding from the European Union’s Seventh Framework Programme under a grant agreement.
Dr. McCabe disclosed no conflicts of interest. Other authors disclosed consulting, advisory roles, or research funding from numerous pharmaceutical companies, including Lilly (gemcitabine) and Pfizer (irinotecan). Dr. Desai disclosed a consulting/advisory role and institutional research funding from Lilly.
Abemaciclib Meets Primary Endpoint in Phase 2 Trial of DDLS
The newer and more potent CDK4 inhibitor, abemaciclib, met its primary endpoint in the investigator-initiated, single-center, single-arm, phase 2 trial in patients with advanced progressive dedifferentiated liposarcoma (DDLS). Twenty-two patients (76%) achieved progression-free survival (PFS) at 12 weeks for a median PFS of 30 weeks. A subset of patients experienced prolonged clinical benefit, remaining on study with stable disease for over 900 days. The study (NCT02846987) was conducted at Memorial Sloan Kettering Cancer Center (MSKCC) in New York and Mark A. Dickson, MD, presented the results at ASCO.
Of three agents in the clinic with the potential to target CDK4 and CDK6—palbociclib, ribociclib, and abemaciclib— abemaciclib is more selective for CDK4 than CDK6. CDK4 amplification occurs in more than 90% of well-differentiated and dedifferentiated liposarcomas. Abemaciclib also has a different side effect profile, with less hematologic toxicity than the other 2 agents. The current study was considered positive if 15 patients or more of a 30-patient sample size were progression- free at 12 weeks.
Results
Thirty patients, 29 evaluable, with metastatic or recurrent DDLS were enrolled and treated with abemaciclib 200 mg orally twice daily between August 2016 and October 2018. Data cutoff for the presentation was the first week of May 2019. Patients were a median of 62 years, 60% were male, and half had no prior systemic treatment. Prior systemic treatments for those previously treated included doxorubicin, olaratumab, gemcitabine, docetaxel, ifosfamide, eribulin, and trabectedin. For 87%, the primary tumor was in their abdomen or retroperitoneum.
Toxicity was as expected with this class of agent, according to the investigators. The most common grades 2 and 3 toxicities, respectively, possibly related to the study drug, occurring in more than 1 patient included anemia (70%, 37%), thrombocytopenia (13%, 13%), neutropenia (43%, 17%), and lymphocyte count decreased (23%, 23%). Very few of these adverse events were grade 4—none for anemia, and 3% each for thrombocytopenia, neutropenia, and lymphocyte count decreased. Diarrhea of grades 2 and 3 occurred in 27% and 7% of patients, respectively, and was managed well with loperamide.
In addition to reaching the primary endpoint of 15 patients or more achieving PFS at 12 weeks, 1 patient had a confirmed partial response (PR) and another an unconfirmed PR. At data cutoff, 11 patients remained on study with stable disease or PR. The investigators conducted correlative studies that indicated all patients had CDK4 and MDM2 amplification with no loss of retinoblastoma tumor suppressor. They observed an inverse correlation between CDK4 amplification and PFS—the higher the level of CDK4 amplification, the shorter the PFS. They also found additional genomic alterations, including JUN, GLI1, ARID1A, TERT, and ATRX. TERT amplification was also associated with shorter PFS. Based on these findings, the investigators believe a phase 3 study of abemaciclib in DDLS is warranted.
Winette van der Graaf, MD, PhD, of the Netherlands Cancer Institute in Amsterdam, in the discussion following the presentation, concurred that it is certainly time for a multicenter phase 3 study of CDK4 inhibitors in DDLS, and a strong international collaboration is key to conducting such studies, particularly in rare cancers. On a critical note, Dr. van der Graaf expressed concern that no patient-reported outcomes were measured after 120 patients, including those in previous studies, were treated on palbociclib and abemaciclib. Given that the toxicities of the CDK4 inhibitors are quite different, she recommended including patient-reported outcomes in future studies using validated health-related quality-of-life instruments.
SOURCE: Dickson MA, et al. J Clin Oncol 37, 2019 (suppl; abstr 11004)
The study was sponsored by Memorial Sloan Kettering Cancer Center, with the study collaborator, Eli Lilly and Company.
Dr. Dickson disclosed research funding from Lilly, the company that provided the study drug. Dr. van der Graaf had no relevant relationships to disclose. Abstract coauthors had consulting/advisory roles or research funding from various companies, including Lilly.
nab-Sirolimus Provides Benefits in Advanced Malignant PEComa
In a prospective phase 2 study of nab-sirolimus in advanced malignant perivascular epithelioid cell tumor (PEComa), the mTOR inhibitor achieved an objective response rate (ORR) of 42% with an acceptable safety profile, despite using relatively high doses of nab-sirolimus compared to other mTOR inhibitors. Activation of the mTOR pathway is common in PEComa, and earlier case reports had indicated substantial clinical benefit with mTOR inhibitor treatment. nab-Sirolimus (ABI-009) is a novel intravenous mTOR inhibitor consisting of nanoparticles of albumin-bound sirolimus. It has significantly higher anti-tumor activity than oral mTOR inhibitors and greater mTOR target suppression at an equal dose. Andrew J. Wagner, MD, PhD, of the Dana-Farber Cancer Institute in Boston, presented the findings of AMPECT (NCT02494570)—Advanced Malignant PEComa Trial—at ASCO.
Investigators enrolled 34 patients 18 years or older with histologically confirmed malignant PEComa. Patients could not have had prior mTOR inhibitors. They received infusions of 100 mg/m2 nab-sirolimus on days 1 and 8 every 21 days until progression or unacceptable toxicity. Patients were a median age of 60 years and 44% were 65 or older; 82% were women, which is typical of the disease. Most patients (88%) had no prior systemic therapy for advanced PEComa.
Results
The drug was well tolerated, with toxicities similar to those of oral mTOR inhibitors. Treatment-related adverse events (TRAEs) occurring in 25% or more of patients were mostly grade 1 or 2 toxicities. Hematologic TRAEs included anemia (47%) and thrombocytopenia (32%) of any grade. Nonhematologic events of any grade included stomatitis/ mucositis (74%), dermatitis/rash (65%), fatigue (59%), nausea (47%), and diarrhea (38%), among others. A few grade 3 events occurred on study, most notably stomatitis/mucositis (18%). Severe adverse events (SAEs) were also uncommon, occurring in 7 of 34 patients (21%). Pneumonitis is common in orally administered mTOR inhibitors; 6 patients (18%) treated with nab-sirolimus had grade 1 or 2 pneumonitis.
Of the 31 evaluable patients, 13 (42%) had an objective response, all of which were partial responses (PR). Eleven (35%) had stable disease and 7 (23%) had progressive disease. The disease control rate, consisting of PR and stable disease, was 77%. The median duration of response had not been reached as of the data cutoff on May 10, 2019. At that time, it was 6.2 months (range, 1.5 to 27.7+). The median time to response was 1.4 months and the median progression-free survival (PFS) was 8.4 months. The PFS rate at 6 months was 61%. Three patients had received treatment for over a year and another 3 patients for more than 2 years.
Correlation with biomarkers
Of the 25 patients who had tissue suitable for next-generation sequencing, 9 had TSC2 mutations, 5 had TSC1 mutations, and 11 had neither mutation. Strikingly, 9 of 9 patients with TSC2 mutations developed a PR, while only 1 with a TSC1 mutation responded. One patient with no TSC1/2 mutation also responded and 2 patients with unknown mutational status responded. The investigators also analyzed pS6 status by immunohistochemistry—pS6 is a marker of mTOR hyperactivity. Twenty- five patient samples were available for analysis. Eight of 8 patients who were negative for pS6 staining did not have a response, while 10 of 17 (59%) who were pS6-positive had a PR.
In the discussion that followed, Winette van der Graaf, MD, of the Netherlands Cancer Institute in Amsterdam, noted that this study showed that biomarkers can be used for patient selection, although TSC2 mutations are not uniquely linked with response. She indicated a comparator with sirolimus would have been of great interest.
SOURCE: Wagner AJ, et al. J Clin Oncol 37, 2019 (suppl; abstr 11005).
The study was sponsored by Aadi Bioscience, Inc., and funded in part by a grant from the FDA Office of Orphan Products Development (OOPD).
Disclosures relevant to this presentation include contininstitutional research funding from Aadi Bioscience for Dr. Wagner and a few other abstract coauthors. Several coauthors are employed by Aadi Bioscience and have stock or other ownership interests. Dr. van der Graaf had nothing to disclose.
Cabozantinib Achieves Disease Control in GIST
The phase 2 EORTC 1317 trial, known as CaboGIST (NCT02216578), met its primary endpoint of progression-free survival (PFS) at 12 weeks in patients with metastatic gastrointestinal stromal tumor (GIST) treated with the tyrosine kinase inhibitor (TKI) cabozantinib. Twenty-four (58.5%) of the 41 patients in the primary study population, and 30 (60%) of the entire 50-patient population, were progression-free at 12 weeks. The study needed 21 patients to be progression- free for cabozantinib to warrant further exploration in GIST patients.
Cabozantinib is a multitargeted TKI inhibiting KIT, MET, AXL, and VEGFR2, which are potentially relevant targets in GIST. In patient-derived xenografts of GIST, cabozantinib demonstrated activity in imatinib-sensitive and -resistant models and inhibited tumor growth, proliferation, and angiogenesis. Additional preclinical experience suggested that cabozantinib could potentially be used as a potent MET inhibitor, overcoming upregulation of MET signaling that occurs with imatinib treatment of GIST, known as the kinase switch.
This investigator-initiated study had as its primary objective assessment of the safety and activity of cabozantinib in patients with metastatic GIST who had progressed on imatinib and sunitinib. The patients could not have been exposed to other KIT- or PDGFR-directed TKIs, such as regorafenib. Secondary objectives included the assessment of cabozantinib in different mutational subtypes of GIST. Patients received cabozantinib tablets once daily until they experienced no further clinical benefit or became intolerant to the drug or chose to discontinue therapy. Fifty patients started treatment between February 2017 and August 2018. All were evaluable for the primary endpoint, and one-third of patients contininstitutional cabozantinib treatment as of the database cutoff in January 2019.
Results
Patients were a median age of 63 years. Virtually all patients (92%) had prior surgery and only 8% had prior radiotherapy. The daily cabozantinib dose was a median 47.2 mg and duration of treatment was a median 20.4 weeks. No patient discontinued treatment due to toxicity, but 88% discontinued due to disease progression.
Safety signals were the same as for other indications in which cabozantinib is used. Almost all patients (94%) had at least 1 treatment-related adverse event of grades 1‐4, including diarrhea (74%), palmar-plantar erythrodysesthesia (58%), fatigue (46%), and hypertension (46%), which are typical of treatment with cabozantinib. Hematologic toxicities in this trial were clinically irrelevant, according to the investigators, consisting of small numbers of grades 2‐3 anemia, lymphopenia, white blood cell count abnormality, and neutropenia. Biochemical abnormalities included grades 3 and 4 hypophosphatemia, increased grades 3 and 4 gamma-glutamyl transferase, grade 3 hyponatremia, and grade 3 hypokalemia, in 8% or more of patients.
Overall survival was a median 14.4 months, with 16 patients still on treatment at the time of data cutoff. Twenty- four patients were progression-free at week 12, satisfying the study decision rule for clinical benefit. Median duration of PFS was 6.0 months. Seven patients (14%) achieved a confirmed partial response (PR) and 33 (66%) achieved stable disease (SD). Nine patients had progressive disease as their best response, 3 of whom had some clinical benefit. Forty patients (80%) experienced a clinical benefit of disease control (PR + SD).
An analysis of the relationship of genotype, duration, and RECIST response showed objective responses in patients with primary exon 11 mutations, with exon 9 mutations, and with exon 17 mutations, and in 2 patients without any known mutational information at the time of the presentation. Patients with stable disease were spread across all mutational subsets in the trial. The investigators suggested the definitive role of MET and AXL inhibition in GIST be assessed further in future clinical trials.
SOURCE: Schöffski P, et al. J Clin Oncol 37, 2019 (suppl; abstr 11006).
The study was sponsored by the European Organization for Research and Treatment of Cancer (EORTC).
Presenting author, Patrick Schöffski, MD, of KU Leuven and Leuven Cancer Institute in Belgium, disclosed institutional relationships with multiple pharmaceutical companies for consulting and research funding, including research funding from Exelixis, the developer of cabozantinib. No other abstract coauthor disclosed a relationship with Exelixis.
Larotectinib Effective in TRK Fusion Cancers
Pediatric patients with tropomyosin receptor kinase (TRK) fusions involving NTRK1, NTRK2, and NTRK3 genes had a high response rate with durable responses and a favorable safety profile when treated with larotrectinib, according to a presentation at ASCO. In this pediatric subset of children and adolescents from the SCOUT and NAVIGATE studies, the overall response rate (ORR) was 94%, with a 35% complete response (CR), 59% partial response (PR), and 6% stable disease as of the data cutoff at the end of July 2018.
TRK fusion cancer is a rare malignancy seen in a wide variety of adult and childhood tumor types. Among pediatric malignancies, infantile fibrosarcoma and congenital mesoblastic nephroma are rare, but have high NTRK gene fusion frequency. Other sarcomas and pediatric high-grade gliomas, for example, are less rare but have low NTRK gene fusion frequency. Larotrectinib, a first-in-class and the only selective TRK inhibitor, has high potency against the 3 NTRK genes that encode the neurotrophin receptors. It is highly selective and has limited inhibition of the other kinases. The US Food and Drug Administration approved larotrectinib for the treatment of patients with solid tumors harboring NTRK fusions. Cornelis Martinus van Tilburg, MD, of the Hopp Children’s Cancer Center, Heidelberg University Hospital, and German Cancer Research Center in Heidelberg, Germany, presented the findings.
Investigators enrolled 38 children and adolescents younger than 18 years from the SCOUT (NCT02637687) and NAVIGATE (NCT02576431) studies of larotrectinib who had non-central nervous system (CNS) TRK fusion cancers. Not all patients had the recommended phase 2 dose, Dr. van Tilburg pointed out, but most did. Hence, 29 of the 38 patients received the 100 mg/m2 twice-daily, phase 2 dose until progression, withdrawal, or unacceptable toxicity.
Patients were young, with a median age of 2.3 years (range, 0.1 to 14.0 years). Almost two-thirds (61%) had prior surgery, 11% had prior radiotherapy, and 68% had prior systemic therapy. For 12 patients, larotrectinib was their first systemic therapy. The predominant tumor types were infantile fibrosarcoma (47%) and other soft tissue sarcoma (42%). And 47% of patients had NTRK3 fusions with ETV6, most of which were infantile fibrosarcoma.
Efficacy
Thirty-four patients were evaluable, and 32 had a reduction in tumor size, for an ORR of 94%, CR of 35%, and PR of 59%. Two patients with infantile fibrosarcoma had pathologic CRs—after treatment, no fibroid tissue in the tumors could be found. Median time to response was 1.8 months, median duration of treatment was 10.24 months, and 33 of 38 patients (87%) remained on treatment or underwent surgery with curative intent. As of the data cutoff of July 30, 2018, the secondary endpoints were not yet reached. However, 84% of responders were estimated to have a response duration of a year or more, and progression-free and overall survival looked very promising, according to Dr. van Tilburg.
Adverse events were primarily grades 1 and 2. The grades 3 and 4 treatment-related adverse events were quite few and consisted of increased alanine aminotransferase, decreased neutrophil count, and nausea. Longer follow-up of the patient safety profile is required, particularly since NTRK has multiple roles in neurodevelopment. The investigators recommended that routine testing for NTRK gene fusions in pediatric patients with cancer be conducted in appropriate clinical contexts.
In a discussion after the presentation, Daniel Alexander Morgenstern, MB BChir, PhD, of Great Ormond Street Hospital, London, UK, said that in many ways, the NTRK inhibitors have become the new poster child for precision oncology in pediatrics because of “these really spectacular results” with larotrectinib [and entrectinib]. One of the questions he raised regarding larotrectinib was the issue of CNS penetration, since patients with CNS cancer were not enrolled in the trial and preclinical data suggest limited CNS penetration for larotrectinib.
SOURCE: van Tilburg CM, et al. J Clin Oncol 37, 2019 (suppl; abstr 10010).
The studies were funded by Loxo Oncology, Inc., and Bayer AG.
Disclosures relevant to this presentation include consulting or advisory roles for Bayer for Drs. van Tilburg and Morgenstern. A few coauthors also had consulting/advisory roles or research funding from various companies, including Loxo and Bayer.
Behind Olaratumab's Phase 3 Disappointment
ANNOUNCE, the phase 3 trial designed to confirm the clinical benefit of olaratumab in patients with advanced soft tissue sarcoma (STS), failed to meet its primary endpoint of overall survival (OS) in all STS histologies and the leiomyosarcoma population. The previous phase 1b/2 signal-finding study of olaratumab had achieved an unprecedented improvement in OS, and the US Food and Drug Administration (FDA) awarded olaratumab accelerated approval in October 2016. By December 2018, olaratumab received additional accelerated, conditional, and full approvals in more than 40 countries worldwide. William D. Tap, MD, chief of the Sarcoma Medical Oncology Service at Memorial Sloan Kettering Cancer Center in New York, presented the phase 3 results and provided some explanations for the findings during the plenary session at ASCO.
ANNOUNCE (NCT02451943), which was designed and enrolled prior to olaratumab receiving accelerated approval, opened in September 2015 and completed accrual 10 months later in July 2016. Investigators randomized and treated 509 patients with advanced STS not amenable to curative therapy, 258 patients in the olaratumab-doxorubicin arm and 251 in the placebo-doxorubicin arm. Most patients (46%) had leiomyosarcoma, followed by liposarcoma (18%), pleomorphic sarcoma (13%), and 24% of the patient population had 26 unique histologies. Three-quarters of the patients had no prior systemic therapy.
Results
As of the data cutoff on December 5, 2018, there were no survival differences in the intention-to-treat population, in the total STS population nor in the leiomyosarcoma subpopulation, with olaratumab-doxorubicin compared to placebo-doxorubicin. For the total STS population, median OS with olaratumab- doxorubicin was 20.4 months and with placebo-doxorubicin 19.7 months. “This is the highest survival rate described to date in any phase 3 sarcoma study,” Dr. Tap said. “It is of particular interest as ANNOUNCE did not mandate treatment in the first line.” In the leiomyosarcoma population, median OS was 21.6 months with olaratumab and 21.9 months with placebo. The secondary endpoints of progression-free survival (PFS), overall response rate, and disease control rate did not favor olaratumab either.
Investigators are examining the relationship between PDGFRα expression and OS in ANNOUNCE. PDGFRα-positive tumors tended to do worse with olaratumab than PDGFRα-negative tumors. The investigators noticed a 6-month difference in OS between these populations favoring PDGFRα-negative tumors. Additional biomarker analyses are ongoing.
A large and concerted effort is underway, Dr. Tap said, to understand the results of the ANNOUNCE study alone and in context with the phase 1b/2 study. “There are no noted discrepancies in study conduct or data integrity which could explain these findings or the differences between the two studies.”
Possible explanations
The designs of the phase 1b/2 and phase 3 studies had some important differences. The phase 1b/2 study was a small, open-label, US-centric study (10 sites) that did not include a placebo or subtype- specified analyses. Its primary endpoint was PFS, it did not have a loading dose of olaratumab, and it specified the timing of dexrazoxane administration after 300 mg/m2 of doxorubicin.
ANNOUNCE, on the other hand, was a large (n=509), international (110 study sites), double-blind, placebo-controlled trial that had outcomes evaluated in STS and leiomyosarcoma. Its primary endpoint was OS, it had a loading dose of olaratumab of 20 mg/kg, and there was no restriction as to the timing of dexrazoxane administration.
Dr. Tap pointed out that in ANNOUNCE it was difficult to predict or control for factors that may have had an unanticipated influence on outcomes, such as albumin levels as a surrogate for disease burden and behavior of PDGFRα status. It is possible, he said, that olaratumab has no activity in STS and that the phase 1b/2 results were due to, among other things, the small sample size, numerous represented histologies with disparate clinical behavior, and the effect of subtype-specific therapies on overall survival, given subsequently or even by chance. On the other hand, it is also possible, he said, that olaratumab has some activity in STS, with outcomes being affected by the heterogeneity of the study populations, differences in trial design, and the performance of the ANNOUNCE control arm. Whatever the case, he said, accelerated approval allowed patients to have access to a potentially life-prolonging drug with little added toxicity.
Discussion
In the expert discussion following the presentation, Jaap Verweij, MD, PhD, of Erasmus University Medical Center in Rotterdam, The Netherlands, congratulated the investigators for performing the study at an unprecedented pace. He commented that lumping STS subtypes together is problematic, as different histological subtypes behave as though they are different diseases. Small numbers of each tumor subtype and subtypes with slow tumor growth can impact trial outcomes. In the phase 1b/2 and phase 3 trials, 26 different subtypes were represented in each study. Dr. Verweij pointed out this could have made a big difference in the phase 1b/2 study, in which there were only 66 patients in each arm.
It is striking to note, he said, that without exception, phase 2 randomized studies in STS involving doxorubicin consistently overestimated and wrongly predicted PFS in the subsequent phase 3 studies. And the situation is similar for OS. The results of the ANNOUNCE study are no exception, he added. “Taken together, these studies indicate that phase 2 studies in soft tissue sarcomas, certainly those involving additions of drugs to doxorubicin, even if randomized, should be interpreted with great caution,” he said.
SOURCE: Tap WD, et al. J Clin Oncol 37, 2019 (suppl; abstr LBA3)
The study was sponsored by Eli Lilly and Company.
Dr. Tap reported research funding from Lilly and Dr. Verweij had nothing to report related to this study. Abstract coauthors disclosed numerous financial relationships, including consulting/advisory roles and/or research funding from Lilly, and several were employed by Lilly.
Addition of Temozolomide May Improve Outcomes in RMS
Investigators from the European Pediatric Soft Tissue Sarcoma Study Group (EpSSG) found that the addition of temozolomide (T) to vincristine and irinotecan (VI) may improve outcomes in adults and children with relapsed or refractory rhabdomyosarcoma (RMS). Principal investigator of the study, Anne Sophie Defachelles, MD, pediatric oncologist at the Centre Oscar Lambret in Lille, France, presented the results on behalf of the EpSSG.
The primary objective of the study was to evaluate the efficacy of VI and VIT regimens, defined as objective response (OR)—complete response (CR) plus partial response (PR)—after 2 cycles. Secondary objectives were progression-free survival (PFS), overall survival (OS), and safety in each arm, and the relative treatment effect of VIT compared to VI in terms of OR, survival, and safety.
The international, randomized (1:1), open-label, phase 2 trial (VIT-0910; NCT01355445) was conducted at 37 centers in 5 countries. Patients ages 6 months to 50 years with RMS were eligible. They could not have had prior irinotecan or temozolomide. A 2015 protocol amendment limited enrollment to patients at relapse and increased the enrollment goal by 40 patients. After the 2015 amendment, patients with refractory disease were no longer eligible.
From January 2012 to April 2018, investigators enrolled 120 patients, 60 on each arm. Two patients in the VI arm were not treated. Patients were a median age of 10.5 years in the VI arm and 12 years in the VIT arm, 92% (VI) and 87% (VIT) had relapsed disease, 8% (VI) and 13% (VIT) had refractory disease, and 55% (VI) and 68% (VIT) had metastatic disease at study entry.
Results
Patients achieved an OR rate of 44% (VIT) and 31% (VI) for the whole population, one-sided P value <.0001. The adjusted odds ratio for the whole population was 0.50, P=.09. PFS was 4.7 months (VIT) and 3.2 months (VI), “a nearly significant reduction in the risk of progression,” Dr. Defachelles noted. Median OS was 15.0 months (VIT) and 10.3 months (VI), which amounted to “a large and significant reduction in the risk of death,” she said. The adjusted hazard ratio was 0.55, P=.006.
Adverse events of grade 3 or higher were more frequent in the VIT arm, with hematologic toxicity the most frequent (81% for VIT, 59% for VI), followed by gastrointestinal adverse events. “VIT was significantly more toxic than VI,” Dr. Defachelles observed, “but the toxicity was manageable.”
“VIT is now the standard treatment in Europe for relapsed rhabdomyosarcoma and will be the control arm in the multiarm, multistage RMS study for relapsed patients,” she said.
In a discussion following the presentation, Lars M. Wagner, MD, of Cincinnati Children’s Hospital, pointed out that the study was not powered for the PFS and OS assessments. These were secondary objectives that should be considered exploratory. Therefore, he said, the outcome data is not conclusive. The role of temozolomide in RMS is also unclear, given recent negative results in patients with newly diagnosed metastatic RMS (Malempati et al, Cancer 2019). And he said it’s uncertain how these results apply to patients who received irinotecan upfront for RMS.
SOURCE: Defachelles AS, et al. J Clin Oncol 37, 2019 (suppl; abstr 10000)
The study was sponsored by Centre Oscar Lambret and SFCE (Société Française de Lutte contre les Cancers et Lucémies de l’Enfant et de l’Adolescent) served as collaborator.
Drs. Defachelles and Wagner had no relationships to disclose. A few coauthors had advisory/consulting or speaker roles for various commercial interests, including two for Merck (temozolomide).
Pazopanib Increases Pathologic Necrosis Rates in STS
Pazopanib added to a regimen of preoperative chemoradiation in non-rhabdomyosarcoma soft tissue sarcoma (NRSTS) significantly increased the rate of near-complete pathologic response in both children and adults with intermediate or high-risk disease. Pazopanib, a multitargeted receptor tyrosine kinase inhibitor, works in multiple signaling pathways involved in tumorigenesis— VEGFR-1, -2, -3, PDGFRα/β, and c-kit. A phase 3 study demonstrated significant improvement in progression-free survival (PFS) in advanced STS patients and was the basis for its approval in the US and elsewhere for treatment of this patient population. Preclinical data suggest synergy between pazopanib and cytotoxic chemotherapy, forming the rationale for the current trial with neoadjuvant pazopanib added to chemoradiation.
According to the investigators, the trial (ARST1321) is the first ever collaborative study codeveloped, written, and conducted by pediatric (Children’s Oncology Group) and adult (NRG Oncology) cancer cooperative groups (NCT02180867). Aaron R. Weiss, MD, of the Maine Medical Center in Portland and study cochair, presented the data for the chemotherapy arms at ASCO. The primary objectives of the study were to determine the feasibility of preoperative chemoradiation with or without pazopanib and to compare the rates of complete pathologic response in patients receiving radiation or chemoradiation with or without pazopanib. Pathologic necrosis rates of 90% or better have been found to be predictive of outcome in STS.
Patients with metastatic or non-metastatic NRSTS were eligible to enroll if they had initially unresectable extremity or trunk tumors with the expectation that they would be resectable after therapy. Patients had to be 2 years or older— there was no upper age limit—and had to be able to swallow a tablet whole. The dose-finding phase of the study determined the pediatric dose to be 350 mg/m2 and the adult dose to be 600 mg/m2, both taken orally and once daily. Patients in the chemotherapy cohort were then randomized to receive chemotherapy—ifosfamide and doxorubicin—with or without pazopanib. At 4 weeks, patients in both arms received preoperative radiotherapy (45 Gy in 25 fractions), and at week 13, surgery of the primary site if they did not have progressive disease. After surgery, patients received continuation therapy with or without pazopanib according to their randomization arm. Upon completion and recovery from the continuation therapy, patients could receive surgery/radiotherapy of their metastatic sites.
Results
As of the June 30, 2018, cutoff, 81 patients were enrolled on the chemotherapy arms: 42 in the pazopanib plus chemoradiation arm and 39 in the chemoradiation-only arm. Sixty-one percent of all patients were 18 years or older, and the median age was 20.3 years. Most patients (73%) did not have metastatic disease, and the major histologies represented were synovial sarcoma (49%) and undifferentiated pleomorphic sarcoma (25%).
At week 13, patients in the pazopanib arm showed significant improvement, with 14 (58%) of those evaluated having pathologic necrosis of at least 90%, compared with 4 (22%) in the chemoradiation-only arm (P=.02). The study was closed to further accrual.
Eighteen patients were not evaluable for pathologic response and 21 were pending pathologic evaluation at week 13. Radiographic response rates were not statistically significant on either arm. No complete responses (CR) were achieved in the pazopanib arm, but 14 patients (52%) achieved a partial response (PR) and 12 (44%) had stable disease (SD). In the chemoradiation-only arm, 2 patients (8%) achieved a CR, 12 (50%) a PR, and 8 (33%) SD. Fifteen patients in each arm were not evaluated for radiographic response.
The pazopanib arm experienced more febrile neutropenia and myelotoxicity during induction and continuation phases than the chemoradiation-only arm. In general, investigators indicated pazopanib combined with chemoradiation was well tolerated and no unexpected toxicities arose during the trial.
In the post-presentation discussion, Dr. Raphael E. Pollock, MD, PhD, of The Ohio State University, called it a tremendous challenge to interdigitate primary local therapies in systemic approaches, particularly in the neoadjuvant context. He pointed out that in an earlier study, a 95% to 100% necrosis level was needed to achieve a significant positive impact on outcomes and perhaps a subsequent prospective trial could determine the best level. He questioned whether the availability of only 60% of patient responses could affect the conclusions and whether the high number of toxicities (73.8% grade 3/4 with pazopanib) might be too high to consider the treatment for most patients, given the intensity of the regimen.
SOURCE: Weiss AR, et al. J Clin Oncol 37, 2019 (suppl; abstr 11002)
The study was sponsored by the National Cancer Institute.
Drs. Weiss and Pollock had no relationships with commercial interests to disclose. A few investigators disclosed advisory, consulting, or research roles with pharmaceutical companies, including one who received institutional research funding from Novartis (pazopanib).
Gemcitabine Plus Pazopanib a Potential Alternative in STS
In a phase 2 study of gemcitabine with pazopanib (G+P) or gemcitabine with docetaxel (G+D), investigators concluded the combination with pazopanib can be considered an alternative to that with docetaxel in select patients with advanced soft tissue sarcoma (STS). They reported similar progression-free survival (PFS) and rate of toxicity for the two regimens. Neeta Somaiah, MD, of the University of Texas MD Anderson Cancer Center in Houston, presented the findings of the investigator-initiated effort (NCT01593748) at ASCO.
The objective of the study, conducted at 10 centers across the United States, was to examine the activity of pazopanib when combined with gemcitabine as an alternative to the commonly used gemcitabine plus docetaxel regimen. Pazopanib is a multi-tyrosine kinase inhibitor with efficacy in non-adipocytic STS. Adult patients with metastatic or locally advanced non-adipocytic STS with ECOG performance of 0 or 1 were eligible. Patients had to have received prior anthracycline exposure unless it was contraindicated. The 1:1 randomization included stratification for pelvic radiation and leiomyosarcoma histology, which was felt to have a higher response rate with the pazopanib regimen.
The investigators enrolled 90 patients, 45 in each arm. Patients were a mean age of 56 years, and there was no difference in age or gender distribution between the arms. Patients with leiomyosarcoma (31% overall) or prior pelvic radiation (11% overall) were similar between the arms. The overall response rate using RECIST 1.1 criteria was partial response (PR) in 8 of 44 evaluable patients (18%) in the G+D arm and 5 of 43 evaluable patients (12%) in the G+P arm. Stable disease (SD) was observed in 21 patients (48%) in the G+D arm and 24 patients (56%) in the G+P arm. This amounted to a clinical benefit rate (PR + SD) of 66% and 68% for the G+D and G+P arms, respectively (Fisher’s exact test, P>.99). The median PFS was 4.1 months on both arms and the difference in median overall survival— 15.9 months in the G+D arm and 12.4 months in the G+P arm—was not statistically significant.
Adverse events (AEs) of grade 3 or higher occurred in 19.9% of patients on G+D and 20.6% on G+P. Serious AEs occurred in 33% (G+D) and 22% (G+P). Dose reductions were necessary in 80% of patients on G+P and doses were held in 93%. Dr. Somaiah explained that this may have been because the starting dose of gemcitabine and pazopanib (1000 mg/m2 of gemcitabine on days 1 and 8 and 800 mg of pazopanib) was “probably higher than what we should have started at.” The rate of doses held was also higher in the pazopanib arm (93%) compared with the docetaxel arm (58%). This was likely because pazopanib was a daily dosing, so if there was a toxicity it was more likely to be held than docetaxel, she observed. Grade 3 or higher toxicities occurring in 5% or more of patients in either arm consisted generally of cytopenias and fatigue. The G+P arm experienced a high amount of neutropenia, most likely because this arm did not receive granulocyte-colony stimulating factor (GCSF) support, as opposed to the G+D arm.
Dr. Somaiah pointed out that the 12% response rate for the G+P combination is similar to what has been previously presented and higher than single-agent gemcitabine or pazopanib, but not higher than the G+D combination. The PFS of 4.1 months was less than anticipated, she added, but it was similar on both arms. The investigators believe the G+P combination warrants further exploration.
SOURCE: Somaiah N, et al. J Clin Oncol 37, 2019 (suppl; abstr 11008)
The study was sponsored by the Medical University of South Carolina, with Novartis as collaborator.
Dr. Somaiah disclosed Advisory Board roles for Blueprint, Deciphera, and Bayer. Abstract coauthors disclosed advisory/consulting roles or research funding from various commercial interests, including Novartis (pazopanib) and Pfizer (gemcitabine).
rEECur Trial Finding Optimal Chemotherapy Regimen for Ewing Sarcoma
Interim results of the first and largest randomized trial in patients with refractory or recurrent Ewing sarcoma (ES), the rEECur trial, are guiding the way to finding the optimal chemotherapy regimen to treat the disease. Until now, there has been little prospective evidence and no randomized data to guide treatment choices in relapsed or refractory patients, and hence no real standard of care, according to the presentation at ASCO. Several molecularly targeted therapies are emerging, and they require a standardized chemotherapy backbone against which they can be tested.
The rEECur trial (ISRCTN36453794) is a multi-arm, multistage phase 2/3 “drop-a-loser” randomized trial designed to find the standard of care. The trial compares 4 chemotherapy regimens to each other and drops the least effective one after 50 patients per arm are enrolled and evaluated. The 3 remaining regimens continue until at least 75 patients on each arm are enrolled and evaluated, and then another arm would be dropped. The 2 remaining regimens continue to phase 3 evaluation. Four regimens are being tested at 8 centers in 17 countries: topotecan/ cyclophosphamide (TC), irinotecan/temozolomide (IT), gemcitabine/docetaxel (GD), and ifosfamide (IFOS). The primary objective is to identify the optimal regimen based on a balance between efficacy and toxicity. Martin G. McCabe, MB BChir, PhD, of the University of Manchester in the United Kingdom, presented the results on behalf of the investigators of the rEECur trial.
Results
Two hundred twenty patients 4 years or older and younger than 50 years with recurrent or refractory histologically confirmed ES of bone or soft tissue were randomized to receive GD (n=72) or TC, IT, or IFOS (n=148). Sixty-two GD patients and 123 TC/IT/IFOS patients were included in the primary outcome analysis. Patients were predominantly male (70%), with a median age of 19 years (range, 4 to 49). About two-thirds (67.3%) were post-pubertal. Most patients (85%) were primary refractory or experienced their first disease recurrence, and 89% had measurable disease.
Investigators assessed the primary outcome of objective response after 4 cycles of therapy and found 11% of patients treated with GD responded compared to 24% in the other 3 arms combined. When they subjected the data to Bayesian analysis, there was a 25% chance that the response rate in the GD arm was better than the response in Arm A, a 2% chance that it was better than Arm B, and a 3% chance that it was better than Arm C. Because this study was still blinded at the time of the presentation, investigators didn’t know which regimen constituted which arm. The probability that response favored GD, however, was low.
The investigators observed no surprising safety findings. Eighty-five percent of all patients experienced at least 1 adverse event. Most frequent grade 3‐5 events consisted of pneumonitis (50%, 60%), neutropenic fever (17%, 25%), and diarrhea (0, 12%) in GD and the combined 3 arms, respectively. Grade 3 events in the GD arm were lower than in the other 3 arms combined. There was 1 toxic death attributed to neutropenic sepsis in 1 of the 3 blinded arms.
Median progression-free survival (PFS) for all patients was approximately 5 months. Bayesian analysis suggested there was a low probability that GD was more effective than the other 3 arms: a 22% chance that GD was better than Arm A, a 3% chance that it was better than Arm B, and a 7% chance that it was better than Arm C. Bayesian analysis also suggested there was a probability that OS favored GD. Because the trial directs only the first 4 or 6 cycles of treatment and the patients receive more treatment after trial-directed therapy, investigators were not fully able to interpret this.
Data suggested GD is a less effective regimen than the other 3 regimens both by objective response rate and PFS, so GD has been dropped from the study. Investigators already had more than 75 evaluable patients in each of the 3 arms for the second interim analysis to take place. In a discussion following the presentation, Jayesh Desai, FRACP, of Peter MacCallum Cancer Centre in Melbourne, Australia, called this study a potentially practice-changing trial at this early stage, noting that the GD combination will be de-prioritized in practice based on these results.
SOURCE: McCabe MG, et al. J Clin Oncol 37, 2019 (suppl; abstr 11007)
The rEECur trial is sponsored by the University of Birmingham (UK) and received funding from the European Union’s Seventh Framework Programme under a grant agreement.
Dr. McCabe disclosed no conflicts of interest. Other authors disclosed consulting, advisory roles, or research funding from numerous pharmaceutical companies, including Lilly (gemcitabine) and Pfizer (irinotecan). Dr. Desai disclosed a consulting/advisory role and institutional research funding from Lilly.
Abemaciclib Meets Primary Endpoint in Phase 2 Trial of DDLS
The newer and more potent CDK4 inhibitor, abemaciclib, met its primary endpoint in the investigator-initiated, single-center, single-arm, phase 2 trial in patients with advanced progressive dedifferentiated liposarcoma (DDLS). Twenty-two patients (76%) achieved progression-free survival (PFS) at 12 weeks for a median PFS of 30 weeks. A subset of patients experienced prolonged clinical benefit, remaining on study with stable disease for over 900 days. The study (NCT02846987) was conducted at Memorial Sloan Kettering Cancer Center (MSKCC) in New York and Mark A. Dickson, MD, presented the results at ASCO.
Of three agents in the clinic with the potential to target CDK4 and CDK6—palbociclib, ribociclib, and abemaciclib— abemaciclib is more selective for CDK4 than CDK6. CDK4 amplification occurs in more than 90% of well-differentiated and dedifferentiated liposarcomas. Abemaciclib also has a different side effect profile, with less hematologic toxicity than the other 2 agents. The current study was considered positive if 15 patients or more of a 30-patient sample size were progression- free at 12 weeks.
Results
Thirty patients, 29 evaluable, with metastatic or recurrent DDLS were enrolled and treated with abemaciclib 200 mg orally twice daily between August 2016 and October 2018. Data cutoff for the presentation was the first week of May 2019. Patients were a median of 62 years, 60% were male, and half had no prior systemic treatment. Prior systemic treatments for those previously treated included doxorubicin, olaratumab, gemcitabine, docetaxel, ifosfamide, eribulin, and trabectedin. For 87%, the primary tumor was in their abdomen or retroperitoneum.
Toxicity was as expected with this class of agent, according to the investigators. The most common grades 2 and 3 toxicities, respectively, possibly related to the study drug, occurring in more than 1 patient included anemia (70%, 37%), thrombocytopenia (13%, 13%), neutropenia (43%, 17%), and lymphocyte count decreased (23%, 23%). Very few of these adverse events were grade 4—none for anemia, and 3% each for thrombocytopenia, neutropenia, and lymphocyte count decreased. Diarrhea of grades 2 and 3 occurred in 27% and 7% of patients, respectively, and was managed well with loperamide.
In addition to reaching the primary endpoint of 15 patients or more achieving PFS at 12 weeks, 1 patient had a confirmed partial response (PR) and another an unconfirmed PR. At data cutoff, 11 patients remained on study with stable disease or PR. The investigators conducted correlative studies that indicated all patients had CDK4 and MDM2 amplification with no loss of retinoblastoma tumor suppressor. They observed an inverse correlation between CDK4 amplification and PFS—the higher the level of CDK4 amplification, the shorter the PFS. They also found additional genomic alterations, including JUN, GLI1, ARID1A, TERT, and ATRX. TERT amplification was also associated with shorter PFS. Based on these findings, the investigators believe a phase 3 study of abemaciclib in DDLS is warranted.
Winette van der Graaf, MD, PhD, of the Netherlands Cancer Institute in Amsterdam, in the discussion following the presentation, concurred that it is certainly time for a multicenter phase 3 study of CDK4 inhibitors in DDLS, and a strong international collaboration is key to conducting such studies, particularly in rare cancers. On a critical note, Dr. van der Graaf expressed concern that no patient-reported outcomes were measured after 120 patients, including those in previous studies, were treated on palbociclib and abemaciclib. Given that the toxicities of the CDK4 inhibitors are quite different, she recommended including patient-reported outcomes in future studies using validated health-related quality-of-life instruments.
SOURCE: Dickson MA, et al. J Clin Oncol 37, 2019 (suppl; abstr 11004)
The study was sponsored by Memorial Sloan Kettering Cancer Center, with the study collaborator, Eli Lilly and Company.
Dr. Dickson disclosed research funding from Lilly, the company that provided the study drug. Dr. van der Graaf had no relevant relationships to disclose. Abstract coauthors had consulting/advisory roles or research funding from various companies, including Lilly.
nab-Sirolimus Provides Benefits in Advanced Malignant PEComa
In a prospective phase 2 study of nab-sirolimus in advanced malignant perivascular epithelioid cell tumor (PEComa), the mTOR inhibitor achieved an objective response rate (ORR) of 42% with an acceptable safety profile, despite using relatively high doses of nab-sirolimus compared to other mTOR inhibitors. Activation of the mTOR pathway is common in PEComa, and earlier case reports had indicated substantial clinical benefit with mTOR inhibitor treatment. nab-Sirolimus (ABI-009) is a novel intravenous mTOR inhibitor consisting of nanoparticles of albumin-bound sirolimus. It has significantly higher anti-tumor activity than oral mTOR inhibitors and greater mTOR target suppression at an equal dose. Andrew J. Wagner, MD, PhD, of the Dana-Farber Cancer Institute in Boston, presented the findings of AMPECT (NCT02494570)—Advanced Malignant PEComa Trial—at ASCO.
Investigators enrolled 34 patients 18 years or older with histologically confirmed malignant PEComa. Patients could not have had prior mTOR inhibitors. They received infusions of 100 mg/m2 nab-sirolimus on days 1 and 8 every 21 days until progression or unacceptable toxicity. Patients were a median age of 60 years and 44% were 65 or older; 82% were women, which is typical of the disease. Most patients (88%) had no prior systemic therapy for advanced PEComa.
Results
The drug was well tolerated, with toxicities similar to those of oral mTOR inhibitors. Treatment-related adverse events (TRAEs) occurring in 25% or more of patients were mostly grade 1 or 2 toxicities. Hematologic TRAEs included anemia (47%) and thrombocytopenia (32%) of any grade. Nonhematologic events of any grade included stomatitis/ mucositis (74%), dermatitis/rash (65%), fatigue (59%), nausea (47%), and diarrhea (38%), among others. A few grade 3 events occurred on study, most notably stomatitis/mucositis (18%). Severe adverse events (SAEs) were also uncommon, occurring in 7 of 34 patients (21%). Pneumonitis is common in orally administered mTOR inhibitors; 6 patients (18%) treated with nab-sirolimus had grade 1 or 2 pneumonitis.
Of the 31 evaluable patients, 13 (42%) had an objective response, all of which were partial responses (PR). Eleven (35%) had stable disease and 7 (23%) had progressive disease. The disease control rate, consisting of PR and stable disease, was 77%. The median duration of response had not been reached as of the data cutoff on May 10, 2019. At that time, it was 6.2 months (range, 1.5 to 27.7+). The median time to response was 1.4 months and the median progression-free survival (PFS) was 8.4 months. The PFS rate at 6 months was 61%. Three patients had received treatment for over a year and another 3 patients for more than 2 years.
Correlation with biomarkers
Of the 25 patients who had tissue suitable for next-generation sequencing, 9 had TSC2 mutations, 5 had TSC1 mutations, and 11 had neither mutation. Strikingly, 9 of 9 patients with TSC2 mutations developed a PR, while only 1 with a TSC1 mutation responded. One patient with no TSC1/2 mutation also responded and 2 patients with unknown mutational status responded. The investigators also analyzed pS6 status by immunohistochemistry—pS6 is a marker of mTOR hyperactivity. Twenty- five patient samples were available for analysis. Eight of 8 patients who were negative for pS6 staining did not have a response, while 10 of 17 (59%) who were pS6-positive had a PR.
In the discussion that followed, Winette van der Graaf, MD, of the Netherlands Cancer Institute in Amsterdam, noted that this study showed that biomarkers can be used for patient selection, although TSC2 mutations are not uniquely linked with response. She indicated a comparator with sirolimus would have been of great interest.
SOURCE: Wagner AJ, et al. J Clin Oncol 37, 2019 (suppl; abstr 11005).
The study was sponsored by Aadi Bioscience, Inc., and funded in part by a grant from the FDA Office of Orphan Products Development (OOPD).
Disclosures relevant to this presentation include contininstitutional research funding from Aadi Bioscience for Dr. Wagner and a few other abstract coauthors. Several coauthors are employed by Aadi Bioscience and have stock or other ownership interests. Dr. van der Graaf had nothing to disclose.
Cabozantinib Achieves Disease Control in GIST
The phase 2 EORTC 1317 trial, known as CaboGIST (NCT02216578), met its primary endpoint of progression-free survival (PFS) at 12 weeks in patients with metastatic gastrointestinal stromal tumor (GIST) treated with the tyrosine kinase inhibitor (TKI) cabozantinib. Twenty-four (58.5%) of the 41 patients in the primary study population, and 30 (60%) of the entire 50-patient population, were progression-free at 12 weeks. The study needed 21 patients to be progression- free for cabozantinib to warrant further exploration in GIST patients.
Cabozantinib is a multitargeted TKI inhibiting KIT, MET, AXL, and VEGFR2, which are potentially relevant targets in GIST. In patient-derived xenografts of GIST, cabozantinib demonstrated activity in imatinib-sensitive and -resistant models and inhibited tumor growth, proliferation, and angiogenesis. Additional preclinical experience suggested that cabozantinib could potentially be used as a potent MET inhibitor, overcoming upregulation of MET signaling that occurs with imatinib treatment of GIST, known as the kinase switch.
This investigator-initiated study had as its primary objective assessment of the safety and activity of cabozantinib in patients with metastatic GIST who had progressed on imatinib and sunitinib. The patients could not have been exposed to other KIT- or PDGFR-directed TKIs, such as regorafenib. Secondary objectives included the assessment of cabozantinib in different mutational subtypes of GIST. Patients received cabozantinib tablets once daily until they experienced no further clinical benefit or became intolerant to the drug or chose to discontinue therapy. Fifty patients started treatment between February 2017 and August 2018. All were evaluable for the primary endpoint, and one-third of patients contininstitutional cabozantinib treatment as of the database cutoff in January 2019.
Results
Patients were a median age of 63 years. Virtually all patients (92%) had prior surgery and only 8% had prior radiotherapy. The daily cabozantinib dose was a median 47.2 mg and duration of treatment was a median 20.4 weeks. No patient discontinued treatment due to toxicity, but 88% discontinued due to disease progression.
Safety signals were the same as for other indications in which cabozantinib is used. Almost all patients (94%) had at least 1 treatment-related adverse event of grades 1‐4, including diarrhea (74%), palmar-plantar erythrodysesthesia (58%), fatigue (46%), and hypertension (46%), which are typical of treatment with cabozantinib. Hematologic toxicities in this trial were clinically irrelevant, according to the investigators, consisting of small numbers of grades 2‐3 anemia, lymphopenia, white blood cell count abnormality, and neutropenia. Biochemical abnormalities included grades 3 and 4 hypophosphatemia, increased grades 3 and 4 gamma-glutamyl transferase, grade 3 hyponatremia, and grade 3 hypokalemia, in 8% or more of patients.
Overall survival was a median 14.4 months, with 16 patients still on treatment at the time of data cutoff. Twenty- four patients were progression-free at week 12, satisfying the study decision rule for clinical benefit. Median duration of PFS was 6.0 months. Seven patients (14%) achieved a confirmed partial response (PR) and 33 (66%) achieved stable disease (SD). Nine patients had progressive disease as their best response, 3 of whom had some clinical benefit. Forty patients (80%) experienced a clinical benefit of disease control (PR + SD).
An analysis of the relationship of genotype, duration, and RECIST response showed objective responses in patients with primary exon 11 mutations, with exon 9 mutations, and with exon 17 mutations, and in 2 patients without any known mutational information at the time of the presentation. Patients with stable disease were spread across all mutational subsets in the trial. The investigators suggested the definitive role of MET and AXL inhibition in GIST be assessed further in future clinical trials.
SOURCE: Schöffski P, et al. J Clin Oncol 37, 2019 (suppl; abstr 11006).
The study was sponsored by the European Organization for Research and Treatment of Cancer (EORTC).
Presenting author, Patrick Schöffski, MD, of KU Leuven and Leuven Cancer Institute in Belgium, disclosed institutional relationships with multiple pharmaceutical companies for consulting and research funding, including research funding from Exelixis, the developer of cabozantinib. No other abstract coauthor disclosed a relationship with Exelixis.
Larotectinib Effective in TRK Fusion Cancers
Pediatric patients with tropomyosin receptor kinase (TRK) fusions involving NTRK1, NTRK2, and NTRK3 genes had a high response rate with durable responses and a favorable safety profile when treated with larotrectinib, according to a presentation at ASCO. In this pediatric subset of children and adolescents from the SCOUT and NAVIGATE studies, the overall response rate (ORR) was 94%, with a 35% complete response (CR), 59% partial response (PR), and 6% stable disease as of the data cutoff at the end of July 2018.
TRK fusion cancer is a rare malignancy seen in a wide variety of adult and childhood tumor types. Among pediatric malignancies, infantile fibrosarcoma and congenital mesoblastic nephroma are rare, but have high NTRK gene fusion frequency. Other sarcomas and pediatric high-grade gliomas, for example, are less rare but have low NTRK gene fusion frequency. Larotrectinib, a first-in-class and the only selective TRK inhibitor, has high potency against the 3 NTRK genes that encode the neurotrophin receptors. It is highly selective and has limited inhibition of the other kinases. The US Food and Drug Administration approved larotrectinib for the treatment of patients with solid tumors harboring NTRK fusions. Cornelis Martinus van Tilburg, MD, of the Hopp Children’s Cancer Center, Heidelberg University Hospital, and German Cancer Research Center in Heidelberg, Germany, presented the findings.
Investigators enrolled 38 children and adolescents younger than 18 years from the SCOUT (NCT02637687) and NAVIGATE (NCT02576431) studies of larotrectinib who had non-central nervous system (CNS) TRK fusion cancers. Not all patients had the recommended phase 2 dose, Dr. van Tilburg pointed out, but most did. Hence, 29 of the 38 patients received the 100 mg/m2 twice-daily, phase 2 dose until progression, withdrawal, or unacceptable toxicity.
Patients were young, with a median age of 2.3 years (range, 0.1 to 14.0 years). Almost two-thirds (61%) had prior surgery, 11% had prior radiotherapy, and 68% had prior systemic therapy. For 12 patients, larotrectinib was their first systemic therapy. The predominant tumor types were infantile fibrosarcoma (47%) and other soft tissue sarcoma (42%). And 47% of patients had NTRK3 fusions with ETV6, most of which were infantile fibrosarcoma.
Efficacy
Thirty-four patients were evaluable, and 32 had a reduction in tumor size, for an ORR of 94%, CR of 35%, and PR of 59%. Two patients with infantile fibrosarcoma had pathologic CRs—after treatment, no fibroid tissue in the tumors could be found. Median time to response was 1.8 months, median duration of treatment was 10.24 months, and 33 of 38 patients (87%) remained on treatment or underwent surgery with curative intent. As of the data cutoff of July 30, 2018, the secondary endpoints were not yet reached. However, 84% of responders were estimated to have a response duration of a year or more, and progression-free and overall survival looked very promising, according to Dr. van Tilburg.
Adverse events were primarily grades 1 and 2. The grades 3 and 4 treatment-related adverse events were quite few and consisted of increased alanine aminotransferase, decreased neutrophil count, and nausea. Longer follow-up of the patient safety profile is required, particularly since NTRK has multiple roles in neurodevelopment. The investigators recommended that routine testing for NTRK gene fusions in pediatric patients with cancer be conducted in appropriate clinical contexts.
In a discussion after the presentation, Daniel Alexander Morgenstern, MB BChir, PhD, of Great Ormond Street Hospital, London, UK, said that in many ways, the NTRK inhibitors have become the new poster child for precision oncology in pediatrics because of “these really spectacular results” with larotrectinib [and entrectinib]. One of the questions he raised regarding larotrectinib was the issue of CNS penetration, since patients with CNS cancer were not enrolled in the trial and preclinical data suggest limited CNS penetration for larotrectinib.
SOURCE: van Tilburg CM, et al. J Clin Oncol 37, 2019 (suppl; abstr 10010).
The studies were funded by Loxo Oncology, Inc., and Bayer AG.
Disclosures relevant to this presentation include consulting or advisory roles for Bayer for Drs. van Tilburg and Morgenstern. A few coauthors also had consulting/advisory roles or research funding from various companies, including Loxo and Bayer.
Gastrointestinal Stromal Tumors: Management of Localized Disease
Gastrointestinal stromal tumors (GISTs) are the most common mesenchymal tumor of the gastrointestinal (GI) tract and arise from the interstitial cells of Cajal of the myenteric plexus. These tumors are rare, with about 1 case per 100,000 persons diagnosed in the United States annually, but may be incidentally discovered in up to 1 in 5 autopsy specimens of older adults.1,2 Epidemiologic risk factors include increasing age, with a peak incidence between age 60 and 65 years, male gender, black race, and non-Hispanic white ethnicity. Germline predisposition can also increase the risk of developing GISTs; molecular drivers of GIST include gain-of-function mutations in the KIT proto-oncogene and platelet-derived growth factor receptor α (PDGFRA) gene, which both encode structurally similar tyrosine kinase receptors; germline mutations of succinate dehydrogenase (SDH) subunit genes; and mutations associated with neurofibromatosis type 1.
GISTs most commonly involve the stomach, followed by the small intestine, but can arise anywhere within the GI tract (esophagus, colon, rectum, and anus). They can also develop outside the GI tract, arising from the mesentery, omentum, and retroperitoneum. The majority of cases are localized or locoregional, whereas about 20% are metastatic at presentation.1 GISTs can occur in children, adolescents, and young adults. Pediatric GISTs represent a distinct subset marked by female predominance and gastric origin, are often multifocal, can sometimes have lymph node involvement, and typically lack mutations in the KIT and PDGFRA genes.
This review is the first of 2 articles focusing on the diagnosis and management of GISTs. Here, we review the evaluation and diagnosis of GISTs along with management of localized disease. Management of advanced disease is reviewed in a separate article.
Case Presentation
A 64-year-old African American man with progressive iron deficiency and abdominal discomfort undergoes upper and lower endoscopy and is found to have a bulging mass within his abdominal cavity. He undergoes a computed tomography (CT) evaluation of the chest, abdomen, and pelvis with contrast, which reveals the presence of a 10-cm gastric mass, with no other lesions identified. He undergoes surgical resection of the mass and presents for review of his pathology and to discuss his treatment plan.
What histopathologic features are consistent with GIST?
What factors are used for risk stratification and to predict likelihood of recurrence?
Clinical Presentation and Diagnosis
Most patients present with symptoms of overt or occult GI bleeding or abdominal discomfort, but a significant proportion of GISTs are discovered incidentally. Lymph node involvement is not typical, except for GISTs occurring in children and/or with rare syndromes. Most syndromic GISTs are multifocal and multicentric. After surgical resection, GISTs usually recur or metastasize within the abdominal cavity, including the omentum, peritoneum, or liver. These tumors rarely spread to the lungs, brain, or bones; when tumor spread does occur, it tends to be in heavily pre-treated patients with advanced disease who have been on multiple lines of therapy for a long duration of time.
The diagnosis usually can be made by histopathology. Specimens can be obtained by endoscopic ultrasound (EUS)– or CT-guided methods, the latter of which carries a very small risk of contamination from percutaneous biopsy. In terms of morphology, GISTs can be spindle cell, epithelioid, or mixed neoplasms. Epithelioid tumors are more commonly seen in the stomach and are often PDGFRA-mutated or SDH-deficient. The differential diagnosis includes other soft-tissue GI wall tumors such as leiomyosarcomas/leiomyomas, germ cell tumors, lymphomas, fibromatosis, and neuroendocrine and neurogenic tumors. A unique feature of GISTs that differentiates them from leiomyomas is near universal expression of CD117 by immunohistochemistry (IHC); this characteristic has allowed pathologists and providers to accurately distinguish true GISTs from other GI mesenchymal tumors.3 Recently, DOG1 (discovered on GIST1) immunoreactivity has been found to be helpful in identifying patients with CD117-negative GISTs. Initially identified through gene expression analysis of GISTs, DOG1 IHC can identify the common mutant c-Kit-driven CD117-positive GISTs as well as the rare CD117-negative GISTs, which are often driven by mutated PDGFRA.4 Importantly, IHC for KIT and DOG1 are not surrogates for mutational status, nor are they predictive of tyrosine kinase inhibitor (TKI) sensitivity. If IHC of a tumor specimen is CD117- and DOG1-negative, the specimen can be sent for KIT and PDGFRA mutational analysis to confirm the diagnosis. If analysis reveals that these genes are wild-type, then IHC staining for SDH B (SDHB) should follow to assess for an SDH-deficient GIST (negative staining).
Risk Stratification for Recurrence
The clinical behavior of GISTs can be variable. Some are indolent, while others behave more aggressively, with a greater malignant potential and a higher propensity to recur and metastasize. Clinical and pathologic features can provide important prognostic information that allows providers to risk-stratify patients. Various institutions have assessed prognostic variables for GISTs. In 2001, the National Institutes of Health (NIH) held a GIST workshop that proposed an approach to estimating metastatic risk based on tumor size and mitotic index (NIH or Fletcher criteria).5 Joensuu et al later proposed a modification of the NIH risk classification to include tumor location and tumor rupture (modified NIH criteria or Joensuu criteria).6-8 Similarly, the Armed Forces Institute of Pathology (AFIP) identified tumor site as a prognostic factor, with gastric GISTs having the best prognosis (AFIP-Miettinen criteria).9-11 Tabular schemes were designed which stratified patients into discrete groups with ranges for mitotic rate and tumor size. Nomograms for ease of use were then constructed utilizing a bimodal mitotic rate and included tumor site and size.12 Finally, contour maps were developed, which have the advantage of evaluating mitotic rate and tumor size as continuous nonlinear variables and also include tumor site and rupture (associated with a high risk of peritoneal metastasis) separately, further improving risk assessment. These contour maps have been validated against pooled data from 10 series (2560 patients).13 High-risk features identified from these studies include tumor location, size, mitotic rate and tumor rupture and are now used for deciding on the use of adjuvant imatinib and as requirements to enter clinical trials assessing adjuvant therapy for resected GISTs.
Case Continued
The patient’s operative and pathology reports indicate that the tumor is a spindle cell neoplasm of the stomach that is positive for CD117, DOG1, and CD34 and negative for smooth muscle actin and S-100, consistent with a diagnosis of GIST. Resection margins are negative. There are 10 mitoses per 50 high-power fields (HPF). Per the operative report, there was no intraoperative or intraperitoneal tumor rupture. Thus, while his GIST was gastric, which generally has a more favorable prognosis, the tumor harbors high-risk features based on its size and mitotic index.
What further testing should be requested?
Molecular Alterations
It is recommended that a mutational analysis be performed as part of the diagnostic work-up of all GISTs.14 Mutational analysis can provide prognostic and predictive information for sensitivity to imatinib and should be considered standard of care. It may also be useful for confirming a GIST diagnosis, or, if negative, lead to further evaluation with an IHC stain for SDHB. The c-Kit receptor is a member of the tyrosine kinase family and, through direct interactions with stem cell factor (SCF), can upregulate the PI3K/AKT/mTOR, Ras/Raf/MEK/ERK, and JAK-STAT pathways, resulting in transcription and translation of genes that enhance cell growth and survival.15 The cell of origin of GISTs, the interstitial cells of Cajal, are dependent on the SCF–c-Kit interaction for development.16 Likewise, the large majority of GISTs (about 70%) are driven by upregulation and constitutive activation of c-Kit, which is normally autoinhibited. About 80% of KIT mutations involve exon 11; these GISTs are most often associated with a gastric location and are associated with a favorable recurrence-free survival (RFS) rate with surgery alone.17KIT exon 9 mutations are much less common, encompassing only about 10% of GIST KIT mutations, and GISTs with these mutations are more likely to arise from the small bowel.17
About 8% of GISTs harbor gain-of-function PDGFRA driver mutations rendering constitutively active PDGFRA.18PDGFRA mutations are mutually exclusive from KIT mutations, and PDGFRA-mutated tumors most often occur in the stomach. PDGFRA mutations generally are associated with a lower mitotic rate and gastric location. Identification of the PDGFRA D842V mutation on exon 18, which is the most common, is important, as it is associated with imatinib resistance, and these patients should not be offered imatinib.19
Several other mutations associated with GISTs outside of the KIT and PDGFRA spectrum have been identified. About 10% of GISTs are wildtype for KIT and PDGFRA, and not all KIT/PDGFRA-wildtype GISTs are imatinib-sensitive and/or respond to other TKIs.18 These tumors may harbor aberrations in SDH and NF1, or less commonly, BRAF V600E, FGFR, and NTRK.20,21 SDH subunits B, C and D play a role in the Krebs cycle and electron transport chain. Germline mutations in these SDH subunits can result in the Carney-Stratakis syndrome characterized by the dyad of multifocal GISTs and multicentric paragangliomas.22 This syndrome is most likely to manifest in the pediatric or young adult population. In contradistinction is the Carney triad, which is associated with acquired loss of function of the SDHC gene due to promoter hypermethylation. This syndrome classically occurs in young women and is characterized by an indolent-behaving triad of multicentric GISTs, non-adrenal paragangliomas, and pulmonary chondromas.23 Like PDGFRA D842V–mutated GISTs, SDH-deficient and NF1-associated GISTs are considered imatinib resistant, and these patients should not be offered imatinib therapy.14
Case Continued
The patient’s GIST is found to harbor a KIT exon 11 single codon deletion. He appears anxious and asks to have everything done to prevent his GIST from coming back and to improve his lifespan.
What are the next steps in the management of this patient?
Management
A multidisciplinary team approach to the management of all GISTs is essential and includes input from radiology, gastroenterology, pathology, medical and surgical oncology, nuclear medicine, and nursing.
Surgical Resection
Small esophagogastric and duodenal GISTs ≤ 2 cm can be asymptomatic and managed with serial endoscopic surveillance, typically every 6 to 12 months, with biopsies if the tumors increase in size. GISTs larger than 2 cm require surgical resection, with resection of the full pseudocapsule and an R0 resection, if possible, since larger GISTs carry a higher risk of growth and recurrence. If an R0 resection would lead to significant morbidity or functional sequelae, an R1 may suffice. Rectal GISTs are an exception, where microscopic margins have been shown to be associated with an increased risk of local failure.24 It is important to explore the abdomen thoroughly for peritoneal, rectovaginal, and vesicular implants and metastasis to the liver. A formal lymph node dissection is not necessary because lymph nodes are rarely involved and should only be removed when clinically suspicious. Tumor rupture must be avoided. A laparoscopic approach should only be considered for smaller tumors, since there is a risk of tumor rupture with larger tumors.14
When is adjuvant imatinib indicated?
Adjuvant Imatinib
Among patients with local or locally advanced GISTs, the risk of death from recurrence with surgery alone can be high, with a historical 5-year overall survival (OS) of about 35%.25 As a result, multiple studies have assessed the benefit of adjuvant imatinib, which is now considered standard of care for patients with imatinib-sensitive, high-risk GISTs. In addition to inhibiting BCR-ABL, imatinib mesylate inhibits multiple other receptor tyrosine kinases, including PDGFR, SCF and c-Kit. As a result, imatinib has demonstrated in vitro inhibition of cell proliferation and apoptosis and clinical activity against GISTs expressing CD117.26 Importantly, adjuvant imatinib should only be offered to patients with imatinib-sensitive mutations, such as KIT exon 11 and KIT exon 9 mutations. Adjuvant imatinib should not be offered to patients with imatinib-insensitive mutations such as PDGFR 842V, NF1, or BRAF-related or SDH-deficient GISTs.
The ACOSOG Z9000 was the first study of adjuvant imatinib in patients with resected GISTs.25 This was a single-arm, phase 2 study involving 106 patients with surgically resected GISTs deemed high-risk for recurrence, defined as size > 10 cm, tumor rupture, or up to 4 peritoneal implants. Patients were treated with imatinib 400 mg daily for 1 year. The primary and secondary endpoints were OS and RFS, respectively. Long-term follow-up of this study demonstrated 1-, 3-, and 5-year OS of 99%, 97%, and 83%, and 1-, 3-, and 5-year RFS of 96%, 60%, and 40%, which compared favorably with historical controls. In a multivariable analysis, increasing tumor size, small bowel location, KIT exon 9 mutation, high mitotic rate, and older age were independent risk factors for a poor RFS.25 It is important to note that the benefit of adjuvant imatinib waned after discontinuation of therapy, creating a rationale to study adjuvant imatinib for longer periods of time.
As a result of the promising phase 2 data, ACOSOG opened a phase 3 randomized trial (Z9001) comparing 1 year of adjuvant imatinib to placebo among patients with surgically resected GISTs that were > 3 cm in size and that stained positive for CD117 on pathology. The trial accrued 713 patients and was stopped early at a planned interim analysis, which revealed a 1-year RFS of 98% for imatinib versus 83% for placebo (hazard ratio [HR], 0.35; P < 0.001). The 1-year OS did not differ between the 2 arms (92.2% vs 99.7%; HR, 0.66; P = 0.47).27 When comparing the 2 arms, imatinib was associated with a higher RFS among patients with a KIT exon 11 deletion, but not among patients with other KIT mutation types, PDGFRA mutations, or who were KIT/PDGFRA wildtype.28 Imatinib was granted approval by the US Food and Drug Administration (FDA) for the adjuvant treatment of high-risk GISTs based on the results of the ACOSOG Z9001 trial.
The EORTC 62024 study was a randomized placebo-controlled trial assessing the benefit of 2 years of adjuvant imatinib.29 Patients had to be considered intermediate or high risk per the 2002 NIH consensus classification to be eligible. The trial enrolled 918 patients. The 5-year OS rate, the original primary endpoint, did not differ between the 2 groups (100% vs 99%). The 3-year and 5-year RFS rates, secondary endpoints, were significantly longer among patients treated with imatinib (84% vs 66% and 69% vs 63%, respectively). Again, it was noted that the benefit of imatinib waned over time after treatment discontinuation.
The Scandinavian Sarcoma Group (SSG XVIII) trial was a prospective randomized phase 3 trial that compared 3 years versus 1 year of adjuvant imatinib.30 Patients had to be enrolled within 12 weeks of the postoperative period and had to have GISTs that were CD117-positive and with a high estimated risk of recurrence, per the modified NIH consensus criteria (size > 10 cm, > 10 mitoses per 50 HPF, diameter > 5 cm with mitotic count > 5, or tumor rupture before or at surgery). Three years of adjuvant imatinib was associated with a 54% reduction in the hazard for recurrence at 5 years (65.6% vs 47.9%; HR, 0.46; P < 0.001) and a 55% reduction in the hazard for death at 5 years (OS 92% vs 81.7%; HR, 0.45; P = 0.02). Based on the results of this study, the FDA granted approval for the use of 3 years of adjuvant imatinib in patients with high-risk resected GISTs.
The observation that a longer duration of adjuvant imatinib was associated with superior RFS and OS led to studies to further explore longer durations of adjuvant imatinib. The PERSIST-5 (Postresection Evaluation of Recurrence-free Survival for Gastrointestinal Stromal Tumors With 5 Years of Adjuvant Imatinib) was a multicenter, single-arm, phase 2 prospective study of adjuvant imatinib with a primary endpoint of RFS after 5 years.31 Patients had to have an intermediate or high risk of recurrence, which included GISTs at any site > 2 cm with > 5 mitoses per 50 HPF or nongastric GISTs that were ≥ 5 cm. With 91 patients enrolled, the estimated 5-year RFS was 90% and the OS was 95%. Of note, about half of the patients stopped treatment early due to a variety of reasons, including patient choice or adverse events. Importantly, there were no recurrences in patients with imatinib-sensitive mutations while on therapy. We know that in patients at high risk of relapse, adjuvant imatinib delays recurrence and improves survival, but whether any patients are cured, or their survival curves are just shifted to the right, is unknown. Only longer follow-up of existing studies, and the results of newer trials utilizing longer durations of adjuvant treatment, will help to determine the real value of adjuvant therapy for GIST patients.32 Based on this study, it would be reasonable to discuss a longer duration of imatinib with patients deemed to be at very high risk of recurrence and who are tolerating therapy well. We are awaiting the data from the randomized phase 3 Scandinavian Sarcoma Group XII trial comparing 5 years versus 3 years of adjuvant imatinib therapy, and from the French ImadGIST trial of adjuvant imatinib for 3 versus 6 years. A summary of the aforementioned key adjuvant trials is shown in the Table.
When imatinib is commenced, careful monitoring for treatment toxicities and drug interactions should ensue in order to improve compliance. Dose density should be maintained if possible, as retrospective studies suggest suboptimal plasma levels are associated with a worse outcome.33
When should neoadjuvant imatinib be considered?
Neoadjuvant Imatinib
Neoadjuvant imatinib should be considered for patients requiring total gastrectomy, esophagectomy, or abdominoperineal resection of the rectum in order to reduce tumor size, limit subsequent surgical morbidity, mitigate tumor bleeding and rupture, and aid with organ preservation. Patients with rectal GISTs that may otherwise warrant an abdominoperineal resection should be offered a trial of imatinib in the neoadjuvant setting. There is no evidence for the use of any other TKI aside from imatinib in the neoadjuvant or adjuvant setting. With neoadjuvant imatinib, it is difficult to accurately assess the mitotic rate in the resected tumor specimen.
The RTOG 0132/ACRIN 6665 trial was a prospective phase 2 study evaluating the efficacy of imatinib 600 mg daily in the perioperative setting.34 The trial enrolled 50 patients, 30 with primary GISTs (group A) and 22 with recurrent metastatic GISTs (group B). Based on data from the metastatic setting revealing a time to treatment response of about 2.5 months, patients were treated with 8 to 12 weeks of preoperative imatinib followed by 2 years of adjuvant imatinib. Imatinib was stopped 24 hours preoperatively and resumed as soon as possible postoperatively. In group A, 7% of patients achieved a partial response (PR), 83% achieved stable disease, and 2-year progression-free survival (PFS) and OS were 83% and 93%, respectively. In group B, 4.5% of patients achieved a PR, 91% achieved stable disease, and 4.5% experienced progressive disease in the preoperative period; the 2-year PFS and OS were 77% and 91%, respectively. The results of this trial demonstrated the feasibility of using perioperative imatinib with minimal effects on surgical outcomes and set the rationale to use neoadjuvant imatinib in select patients with borderline resectable or rectal GISTs. Another EORTC pooled analysis from 10 sarcoma centers revealed that after a median of 10 months of neoadjuvant imatinib, 83.2% of patients achieved an R0 resection and only 1% progressed during treatment.35 After a median follow-up of 46 months, the 5-year disease-free survival and OS were 65% and 87%, respectively.
Mutational testing should be performed beforehand to ensure the tumor is imatinib-sensitive. If a KIT exon 9 mutation is identified, then 400 mg twice daily should be considered (given the benefit seen with 800 mg imatinib for advanced GIST patients), although there are no studies to confirm this practice. Neoadjuvant imatinib is recommended for a total of 6 to 12 months to ensure maximal tumor debulking, but with very close monitoring and surgical input for disease resistance and growth.14 Imatinib should be stopped 1 to 2 days preoperatively and resumed once the patient has recovered from surgery for a total of 3 years (pre-/postoperatively combined). Neoadjuvant therapy has been shown to be safe and effective, but there have been no randomized trials to assess survival.
What is appropriate surveillance for resected GISTs?
Surveillance
There have been no randomized studies to guide the management of surveillance after surgical resection and adjuvant therapy. There is no known optimal follow-up schedule, but several have been proposed.13,36 Among high-risk patients, it is suggested to image every 3 to 6 months during adjuvant therapy, followed by every 3 months for 2 years after discontinuing therapy, then every 6 months for another 3 years and annually thereafter for an additional 5 years. High-risk patients usually relapse within 1 to 3 years after finishing adjuvant therapy, while low-risk patients can relapse later given that their disease can be slower growing. It has been recommended that low-risk patients undergo imaging every 6 months for 5 years, with follow-up individualized thereafter. Very-low-risk patients may not require more than annual imaging. Because most relapses occur within the peritoneum or liver, imaging should encompass the abdomen and pelvis. Surveillance imaging usually consists of CT scans of the abdomen and pelvis. MRI scans can be utilized for patients at lower risk or who are out several years in order to avoid excess radiation exposure. MRI is also specifically helpful for rectal and esophageal lesions. Chest CT or chest radiograph and bone scan are not routinely required for follow-up.
Case Conclusion
The patient receives adjuvant imatinib and experiences grade 2 myalgias, periorbital edema, and macrocytic anemia, which result in imatinib discontinuation after 3 years of treatment. He is seen every 3 to 6 months and a contrast CT abdomen and pelvis is obtained every 6 months for 5 years. During this 5-year follow-up period, he does not have any clinical or radiographic evidence of disease recurrence.
Further follow-up of this patient is presented in the second article in this 2-part review of management of GISTs.
Key Points
- GISTs are the most common mesenchymal neoplasms of the GI tract and can occasionally occur in extragastrointestinal locations as well.
- GISTs encompass a heterogeneous family of tumor subsets with different natural histories, mutations, and TKI responsiveness.
- Surgery is the mainstay of treatment for localized GISTs, with cure rates greater than 50%.
- For very small (< 2 cm) esophagogastric GISTs, endoscopic ultrasound evaluation and follow-up is recommended.
- For tumors ≥ 2 cm, biopsy and excision is the standard approach.
- For localized GISTs, complete surgical resection (R0) is standard treatment, with no lymphadenectomy for clinically negative lymph nodes.
- Mutational analysis should be considered standard of practice. It can be helpful for confirming the diagnosis and can be predictive and prognostic in determining specific TKI therapy and dose.
- Adjuvant imatinib at a dose of 400 mg for 3 years is standard of care for GISTs that are at high risk of relapse and are imatinib-sensitive, and it is the only TKI approved for adjuvant therapy. Patients with PDGFRA D842V, NF1, BRAF or SDH-deficient GISTs should not receive adjuvant imatinib therapy.
- Neoadjuvant therapy can be utilized for sites where extensive resection would lead to significant morbidity. It should be given for 6 to 12 months, but patients need to be monitored closely for tumor growth.
1. Ma GL, Murphy JD, Martinez ME et al. Epidemiology of gastrointestinal stromal tumors in the era of histology codes: results of a population-based study. Cancer Epidemiol Biomarkers Prev. 2015;24:298-302.
2. Agaimy A, Wunsch PH, Hofstaedter F, et al. Minute gastric sclerosing stromal tumors (GIST tumorlets) are common in adults and frequently show c-KIT mutations. Am J Surg Pathol. 2007;31:113-120.
3. Miettinen M, Sobin LH, Sarlomo-Rikala M. Immunohistochemical spectrum of GISTs at different sites and their differential diagnosis with a reference to CD117 (KIT). Mod Pathol. 2000;13:1134-1142.
4. West RB, Corless CL, Chen X, et al. The novel marker, DOG1, is expressed ubiquitously in gastrointestinal stromal tumors irrespective of KIT or PDGFRA mutational status. Am J Pathol. 2004;165:107-113.
5. Fletcher CD, Berman JJ, Corless C, et al. Diagnosis of gastrointestinal stromal tumors: a consensus approach. Int J Surg Pathol. 2002;10:81-89.
6. Joensuu H. Risk stratification of patients diagnosed with gastrointestinal stromal tumor. Hum Pathol. 2008;39:1411-1419.
7. Hohenberger P, Ronellenfitsch U, Oladeji O, et al. Pattern of recurrence in patients with ruptured primary gastrointestinal stromal tumor. Br J Surg. 2010;97:1854-1859.
8. Holmenbakk T, Bjerkehagen B, Boye K, et al. Definition and clinical significance of tumor rupture in gastrointestinal stromal tumours of the small intestine. Br J Surg. 2016;103:684-691.
9. Emory TS, Sobin LH, Lukes L, et al. Prognosis of gastrointestinal smooth-muscle (stromal) tumors: dependence on anatomic site. Am J Surg Pathol. 1999;23:82-87.
10. Miettinen M, Makhlouf H, Sobin LH, Lasota J. Gastrointestinal stromal tumors of the jejunum and ileum: a clinicopathologic, immunohistochemical, and molecular genetic study of 906 cases before imatinib with long-term follow-up. Am J Surg Pathol. 2006;30:477-489.
11. Miettinen M, Sobin LH, Lasota J. Gastrointestinal stromal tumors of the stomach: a clinicopathologic, immunohistochemical, and molecular genetic study of 1765 cases with long-term follow-up. Am J Surg Pathol. 2005;29:52-68.
12. Gold JS, Gonen M, Gutierrez A, et al. Development and validation of a prognostic nomogram for recurrence-free survival after complete surgical resection of localized primary gastrointestinal stromal tumour: a retrospective analysis. Lancet Oncol. 2009;10:1045-1052.
13. Joensuu H, Vehtari A, Rihimaki J et al. Risk of recurrence of gastrointestinal stromal tumor after surgery: an analysis of pooled population-based cohorts. Lancet Oncol. 2012;13:265-274.
14. Casali PG, Abecassis N, Bauer S, et al. Gastrointestinal stromal tumours: ESMO-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow up. Ann Oncol. 2018;29(Supplement_4): iv267.
15. Jing L, Yan-Ling W, Bing-Jia C, et al. The c-kit receptor-mediated signal transduction and tumor-related diseases. Int J Biol Sci. 2013;9:435-443.
16. Hirota S, Isozaki K, Moriyama Y, et al. Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science. 1998;279:577-580.
17. Joensuu H, Rutkowski P, Nishida T, et al. KIT and PDGFRA mutations and the risk of GI stromal tumor recurrence. J Clin Oncol. 2015;33:634-642.
18. Corless CL, Fletcher JA, Heinrich MC. Biology of gastrointestinal stromal tumors. J Clin Oncol. 2004;22:3813-3825.
19. Heinrich MC, Corless CL, Demetri GD, et al. Kinase mutations and imatinib response in patients with metastatic gastrointestinal stromal tumor. J Clin Oncol. 2003;21:4342-4349.
20. Huss S, Pasternack H, Ihle MA, et al. Clinicopathological and molecular features of a large cohort of gastrointestinal stromal tumors (GISTs) and review of the literature: BRAF mutations in KIT/PDGFRA wild-type GISTs are rare events. Hum Pathol. 2017;62:206-214.
21. Shi E, Chmielecki J, Tang CM, et al. FGFR1 and NTRK3 actionable alterations in “Wild-Type” gastrointestinal stromal tumors. J Transl Med. 2016;14:339.
22. Carney JA, Stratakis CA. Familial paraganglioma and gastric stromal sarcoma: a new syndrome distinct from the Carney triad. Am J Med Genet. 2002;108:132-139.
23. Carney JA. Gastric stromal sarcoma, pulmonary chondroma, and extra-adrenal paraganglioma (Carney Triad): natural history, adrenocortical component, and possible familial occurrence. Mayo Clin Proc. 1999;74:543-552.
24. Jakob J, Mussi C, Ronellenfitsch U, et al. Gastrointestinal stromal tumor of the rectum: results of surgical and multimodality therapy in the era of imatinib. Ann Surg Oncol. 2013;20:586-592.
25. DeMatteo RP, Ballman KV, Antonescu CR, et al. Long-term results of adjuvant imatinib mesylate in localized, high-risk, primary gastrointestinal stromal tumor (GIST): ACOSOG Z9000 (Alliance) intergroup phase 2 trial. Ann Surg. 2013;258:422-429.
26. Gleevac (imatinib) [package insert]. East Hanover, NJ: Novartis Pharmaceuticals; 2016.
27. DeMatteo RP, Ballman KV, Antonescu CR, et al. Placebo-controlled randomized trial of adjuvant imatinib mesylate following the resection of localized, primary gastrointestinal stromal tumor (GIST). Lancet. 2009;373:1097-1104.
28. Corless CL, Ballman KV, Antonescu CR, et al. Pathologic and molecular features correlate with long-term outcome after adjuvant therapy of resected primary GI stromal tumor: the ACOSOG Z9001 trial. J Clin Oncol. 2014;32:1563-1570.
29. Casali PG, Le Cesne A, Poveda Velasco A, et al. Imatinib failure-free survival (IFS) in patients with localized gastrointestinal stromal tumors (GIST) treated with adjuvant imatinib (IM): the EORTC/AGITG/FSG/GEIS/ISG randomized controlled phase III trial. J Clin Oncol. 2013;31. Abstract 10500.
30. Joensuu H, Eriksson M, Sundby HK, et al. One vs three years of adjuvant imatinib for operable gastrointestinal stromal tumor: a randomized trial. JAMA. 2012;307:1265-1272.
31. Raut CP, Espat NJ, Maki RG, et al. Efficacy and tolerability of 5-year adjuvant imatinib treatment for patients with resected intermediate- or high-risk primary gastrointestinal stromal tumor: The PERSIST-5 Clinical Trial. JAMA Oncol. 2018: e184060.
32. Benjamin RS, Casali PG. Adjuvant imatinib for GI stromal tumors: when and for how long? J Clin Oncol. 2016;34:215-218.
33. Demetri GD, Wang Y, Wehrle E, et al. Imatinib plasma levels are correlated with clinical benefit in patients with unresectable/metastatic gastrointestinal stromal tumors. J Clin Oncol. 2009;27:3141-3147.
34. Eisenberg BL, Harris J, Blanke CD, et al. Phase II trial of neoadjuvant/adjuvant imatinib mesylate (IM) for advanced primary and metastatic/recurrent operable gastrointestinal stromal tumor (GIST): early results of RTOG 0132/ACRIN 6665. J Surg Oncol. 2009;99:42-47.
35. Rutkowski P, Gronchi A, Hohenberger P, et al. Neoadjuvant imatinib in locally advanced gastrointestinal stromal tumors (GIST): the EORTC STBSG experience. Ann Surg Oncol. 2013;20:2937-2943.
36. Joensuu H, Martin-Broto J, Nishida T, et al. Follow-up strategies for patients with gastrointestinal stromal tumour treated with or without adjuvant imatinib after surgery. Eur J Cancer. 2015;51:1611-1617.
Gastrointestinal stromal tumors (GISTs) are the most common mesenchymal tumor of the gastrointestinal (GI) tract and arise from the interstitial cells of Cajal of the myenteric plexus. These tumors are rare, with about 1 case per 100,000 persons diagnosed in the United States annually, but may be incidentally discovered in up to 1 in 5 autopsy specimens of older adults.1,2 Epidemiologic risk factors include increasing age, with a peak incidence between age 60 and 65 years, male gender, black race, and non-Hispanic white ethnicity. Germline predisposition can also increase the risk of developing GISTs; molecular drivers of GIST include gain-of-function mutations in the KIT proto-oncogene and platelet-derived growth factor receptor α (PDGFRA) gene, which both encode structurally similar tyrosine kinase receptors; germline mutations of succinate dehydrogenase (SDH) subunit genes; and mutations associated with neurofibromatosis type 1.
GISTs most commonly involve the stomach, followed by the small intestine, but can arise anywhere within the GI tract (esophagus, colon, rectum, and anus). They can also develop outside the GI tract, arising from the mesentery, omentum, and retroperitoneum. The majority of cases are localized or locoregional, whereas about 20% are metastatic at presentation.1 GISTs can occur in children, adolescents, and young adults. Pediatric GISTs represent a distinct subset marked by female predominance and gastric origin, are often multifocal, can sometimes have lymph node involvement, and typically lack mutations in the KIT and PDGFRA genes.
This review is the first of 2 articles focusing on the diagnosis and management of GISTs. Here, we review the evaluation and diagnosis of GISTs along with management of localized disease. Management of advanced disease is reviewed in a separate article.
Case Presentation
A 64-year-old African American man with progressive iron deficiency and abdominal discomfort undergoes upper and lower endoscopy and is found to have a bulging mass within his abdominal cavity. He undergoes a computed tomography (CT) evaluation of the chest, abdomen, and pelvis with contrast, which reveals the presence of a 10-cm gastric mass, with no other lesions identified. He undergoes surgical resection of the mass and presents for review of his pathology and to discuss his treatment plan.
What histopathologic features are consistent with GIST?
What factors are used for risk stratification and to predict likelihood of recurrence?
Clinical Presentation and Diagnosis
Most patients present with symptoms of overt or occult GI bleeding or abdominal discomfort, but a significant proportion of GISTs are discovered incidentally. Lymph node involvement is not typical, except for GISTs occurring in children and/or with rare syndromes. Most syndromic GISTs are multifocal and multicentric. After surgical resection, GISTs usually recur or metastasize within the abdominal cavity, including the omentum, peritoneum, or liver. These tumors rarely spread to the lungs, brain, or bones; when tumor spread does occur, it tends to be in heavily pre-treated patients with advanced disease who have been on multiple lines of therapy for a long duration of time.
The diagnosis usually can be made by histopathology. Specimens can be obtained by endoscopic ultrasound (EUS)– or CT-guided methods, the latter of which carries a very small risk of contamination from percutaneous biopsy. In terms of morphology, GISTs can be spindle cell, epithelioid, or mixed neoplasms. Epithelioid tumors are more commonly seen in the stomach and are often PDGFRA-mutated or SDH-deficient. The differential diagnosis includes other soft-tissue GI wall tumors such as leiomyosarcomas/leiomyomas, germ cell tumors, lymphomas, fibromatosis, and neuroendocrine and neurogenic tumors. A unique feature of GISTs that differentiates them from leiomyomas is near universal expression of CD117 by immunohistochemistry (IHC); this characteristic has allowed pathologists and providers to accurately distinguish true GISTs from other GI mesenchymal tumors.3 Recently, DOG1 (discovered on GIST1) immunoreactivity has been found to be helpful in identifying patients with CD117-negative GISTs. Initially identified through gene expression analysis of GISTs, DOG1 IHC can identify the common mutant c-Kit-driven CD117-positive GISTs as well as the rare CD117-negative GISTs, which are often driven by mutated PDGFRA.4 Importantly, IHC for KIT and DOG1 are not surrogates for mutational status, nor are they predictive of tyrosine kinase inhibitor (TKI) sensitivity. If IHC of a tumor specimen is CD117- and DOG1-negative, the specimen can be sent for KIT and PDGFRA mutational analysis to confirm the diagnosis. If analysis reveals that these genes are wild-type, then IHC staining for SDH B (SDHB) should follow to assess for an SDH-deficient GIST (negative staining).
Risk Stratification for Recurrence
The clinical behavior of GISTs can be variable. Some are indolent, while others behave more aggressively, with a greater malignant potential and a higher propensity to recur and metastasize. Clinical and pathologic features can provide important prognostic information that allows providers to risk-stratify patients. Various institutions have assessed prognostic variables for GISTs. In 2001, the National Institutes of Health (NIH) held a GIST workshop that proposed an approach to estimating metastatic risk based on tumor size and mitotic index (NIH or Fletcher criteria).5 Joensuu et al later proposed a modification of the NIH risk classification to include tumor location and tumor rupture (modified NIH criteria or Joensuu criteria).6-8 Similarly, the Armed Forces Institute of Pathology (AFIP) identified tumor site as a prognostic factor, with gastric GISTs having the best prognosis (AFIP-Miettinen criteria).9-11 Tabular schemes were designed which stratified patients into discrete groups with ranges for mitotic rate and tumor size. Nomograms for ease of use were then constructed utilizing a bimodal mitotic rate and included tumor site and size.12 Finally, contour maps were developed, which have the advantage of evaluating mitotic rate and tumor size as continuous nonlinear variables and also include tumor site and rupture (associated with a high risk of peritoneal metastasis) separately, further improving risk assessment. These contour maps have been validated against pooled data from 10 series (2560 patients).13 High-risk features identified from these studies include tumor location, size, mitotic rate and tumor rupture and are now used for deciding on the use of adjuvant imatinib and as requirements to enter clinical trials assessing adjuvant therapy for resected GISTs.
Case Continued
The patient’s operative and pathology reports indicate that the tumor is a spindle cell neoplasm of the stomach that is positive for CD117, DOG1, and CD34 and negative for smooth muscle actin and S-100, consistent with a diagnosis of GIST. Resection margins are negative. There are 10 mitoses per 50 high-power fields (HPF). Per the operative report, there was no intraoperative or intraperitoneal tumor rupture. Thus, while his GIST was gastric, which generally has a more favorable prognosis, the tumor harbors high-risk features based on its size and mitotic index.
What further testing should be requested?
Molecular Alterations
It is recommended that a mutational analysis be performed as part of the diagnostic work-up of all GISTs.14 Mutational analysis can provide prognostic and predictive information for sensitivity to imatinib and should be considered standard of care. It may also be useful for confirming a GIST diagnosis, or, if negative, lead to further evaluation with an IHC stain for SDHB. The c-Kit receptor is a member of the tyrosine kinase family and, through direct interactions with stem cell factor (SCF), can upregulate the PI3K/AKT/mTOR, Ras/Raf/MEK/ERK, and JAK-STAT pathways, resulting in transcription and translation of genes that enhance cell growth and survival.15 The cell of origin of GISTs, the interstitial cells of Cajal, are dependent on the SCF–c-Kit interaction for development.16 Likewise, the large majority of GISTs (about 70%) are driven by upregulation and constitutive activation of c-Kit, which is normally autoinhibited. About 80% of KIT mutations involve exon 11; these GISTs are most often associated with a gastric location and are associated with a favorable recurrence-free survival (RFS) rate with surgery alone.17KIT exon 9 mutations are much less common, encompassing only about 10% of GIST KIT mutations, and GISTs with these mutations are more likely to arise from the small bowel.17
About 8% of GISTs harbor gain-of-function PDGFRA driver mutations rendering constitutively active PDGFRA.18PDGFRA mutations are mutually exclusive from KIT mutations, and PDGFRA-mutated tumors most often occur in the stomach. PDGFRA mutations generally are associated with a lower mitotic rate and gastric location. Identification of the PDGFRA D842V mutation on exon 18, which is the most common, is important, as it is associated with imatinib resistance, and these patients should not be offered imatinib.19
Several other mutations associated with GISTs outside of the KIT and PDGFRA spectrum have been identified. About 10% of GISTs are wildtype for KIT and PDGFRA, and not all KIT/PDGFRA-wildtype GISTs are imatinib-sensitive and/or respond to other TKIs.18 These tumors may harbor aberrations in SDH and NF1, or less commonly, BRAF V600E, FGFR, and NTRK.20,21 SDH subunits B, C and D play a role in the Krebs cycle and electron transport chain. Germline mutations in these SDH subunits can result in the Carney-Stratakis syndrome characterized by the dyad of multifocal GISTs and multicentric paragangliomas.22 This syndrome is most likely to manifest in the pediatric or young adult population. In contradistinction is the Carney triad, which is associated with acquired loss of function of the SDHC gene due to promoter hypermethylation. This syndrome classically occurs in young women and is characterized by an indolent-behaving triad of multicentric GISTs, non-adrenal paragangliomas, and pulmonary chondromas.23 Like PDGFRA D842V–mutated GISTs, SDH-deficient and NF1-associated GISTs are considered imatinib resistant, and these patients should not be offered imatinib therapy.14
Case Continued
The patient’s GIST is found to harbor a KIT exon 11 single codon deletion. He appears anxious and asks to have everything done to prevent his GIST from coming back and to improve his lifespan.
What are the next steps in the management of this patient?
Management
A multidisciplinary team approach to the management of all GISTs is essential and includes input from radiology, gastroenterology, pathology, medical and surgical oncology, nuclear medicine, and nursing.
Surgical Resection
Small esophagogastric and duodenal GISTs ≤ 2 cm can be asymptomatic and managed with serial endoscopic surveillance, typically every 6 to 12 months, with biopsies if the tumors increase in size. GISTs larger than 2 cm require surgical resection, with resection of the full pseudocapsule and an R0 resection, if possible, since larger GISTs carry a higher risk of growth and recurrence. If an R0 resection would lead to significant morbidity or functional sequelae, an R1 may suffice. Rectal GISTs are an exception, where microscopic margins have been shown to be associated with an increased risk of local failure.24 It is important to explore the abdomen thoroughly for peritoneal, rectovaginal, and vesicular implants and metastasis to the liver. A formal lymph node dissection is not necessary because lymph nodes are rarely involved and should only be removed when clinically suspicious. Tumor rupture must be avoided. A laparoscopic approach should only be considered for smaller tumors, since there is a risk of tumor rupture with larger tumors.14
When is adjuvant imatinib indicated?
Adjuvant Imatinib
Among patients with local or locally advanced GISTs, the risk of death from recurrence with surgery alone can be high, with a historical 5-year overall survival (OS) of about 35%.25 As a result, multiple studies have assessed the benefit of adjuvant imatinib, which is now considered standard of care for patients with imatinib-sensitive, high-risk GISTs. In addition to inhibiting BCR-ABL, imatinib mesylate inhibits multiple other receptor tyrosine kinases, including PDGFR, SCF and c-Kit. As a result, imatinib has demonstrated in vitro inhibition of cell proliferation and apoptosis and clinical activity against GISTs expressing CD117.26 Importantly, adjuvant imatinib should only be offered to patients with imatinib-sensitive mutations, such as KIT exon 11 and KIT exon 9 mutations. Adjuvant imatinib should not be offered to patients with imatinib-insensitive mutations such as PDGFR 842V, NF1, or BRAF-related or SDH-deficient GISTs.
The ACOSOG Z9000 was the first study of adjuvant imatinib in patients with resected GISTs.25 This was a single-arm, phase 2 study involving 106 patients with surgically resected GISTs deemed high-risk for recurrence, defined as size > 10 cm, tumor rupture, or up to 4 peritoneal implants. Patients were treated with imatinib 400 mg daily for 1 year. The primary and secondary endpoints were OS and RFS, respectively. Long-term follow-up of this study demonstrated 1-, 3-, and 5-year OS of 99%, 97%, and 83%, and 1-, 3-, and 5-year RFS of 96%, 60%, and 40%, which compared favorably with historical controls. In a multivariable analysis, increasing tumor size, small bowel location, KIT exon 9 mutation, high mitotic rate, and older age were independent risk factors for a poor RFS.25 It is important to note that the benefit of adjuvant imatinib waned after discontinuation of therapy, creating a rationale to study adjuvant imatinib for longer periods of time.
As a result of the promising phase 2 data, ACOSOG opened a phase 3 randomized trial (Z9001) comparing 1 year of adjuvant imatinib to placebo among patients with surgically resected GISTs that were > 3 cm in size and that stained positive for CD117 on pathology. The trial accrued 713 patients and was stopped early at a planned interim analysis, which revealed a 1-year RFS of 98% for imatinib versus 83% for placebo (hazard ratio [HR], 0.35; P < 0.001). The 1-year OS did not differ between the 2 arms (92.2% vs 99.7%; HR, 0.66; P = 0.47).27 When comparing the 2 arms, imatinib was associated with a higher RFS among patients with a KIT exon 11 deletion, but not among patients with other KIT mutation types, PDGFRA mutations, or who were KIT/PDGFRA wildtype.28 Imatinib was granted approval by the US Food and Drug Administration (FDA) for the adjuvant treatment of high-risk GISTs based on the results of the ACOSOG Z9001 trial.
The EORTC 62024 study was a randomized placebo-controlled trial assessing the benefit of 2 years of adjuvant imatinib.29 Patients had to be considered intermediate or high risk per the 2002 NIH consensus classification to be eligible. The trial enrolled 918 patients. The 5-year OS rate, the original primary endpoint, did not differ between the 2 groups (100% vs 99%). The 3-year and 5-year RFS rates, secondary endpoints, were significantly longer among patients treated with imatinib (84% vs 66% and 69% vs 63%, respectively). Again, it was noted that the benefit of imatinib waned over time after treatment discontinuation.
The Scandinavian Sarcoma Group (SSG XVIII) trial was a prospective randomized phase 3 trial that compared 3 years versus 1 year of adjuvant imatinib.30 Patients had to be enrolled within 12 weeks of the postoperative period and had to have GISTs that were CD117-positive and with a high estimated risk of recurrence, per the modified NIH consensus criteria (size > 10 cm, > 10 mitoses per 50 HPF, diameter > 5 cm with mitotic count > 5, or tumor rupture before or at surgery). Three years of adjuvant imatinib was associated with a 54% reduction in the hazard for recurrence at 5 years (65.6% vs 47.9%; HR, 0.46; P < 0.001) and a 55% reduction in the hazard for death at 5 years (OS 92% vs 81.7%; HR, 0.45; P = 0.02). Based on the results of this study, the FDA granted approval for the use of 3 years of adjuvant imatinib in patients with high-risk resected GISTs.
The observation that a longer duration of adjuvant imatinib was associated with superior RFS and OS led to studies to further explore longer durations of adjuvant imatinib. The PERSIST-5 (Postresection Evaluation of Recurrence-free Survival for Gastrointestinal Stromal Tumors With 5 Years of Adjuvant Imatinib) was a multicenter, single-arm, phase 2 prospective study of adjuvant imatinib with a primary endpoint of RFS after 5 years.31 Patients had to have an intermediate or high risk of recurrence, which included GISTs at any site > 2 cm with > 5 mitoses per 50 HPF or nongastric GISTs that were ≥ 5 cm. With 91 patients enrolled, the estimated 5-year RFS was 90% and the OS was 95%. Of note, about half of the patients stopped treatment early due to a variety of reasons, including patient choice or adverse events. Importantly, there were no recurrences in patients with imatinib-sensitive mutations while on therapy. We know that in patients at high risk of relapse, adjuvant imatinib delays recurrence and improves survival, but whether any patients are cured, or their survival curves are just shifted to the right, is unknown. Only longer follow-up of existing studies, and the results of newer trials utilizing longer durations of adjuvant treatment, will help to determine the real value of adjuvant therapy for GIST patients.32 Based on this study, it would be reasonable to discuss a longer duration of imatinib with patients deemed to be at very high risk of recurrence and who are tolerating therapy well. We are awaiting the data from the randomized phase 3 Scandinavian Sarcoma Group XII trial comparing 5 years versus 3 years of adjuvant imatinib therapy, and from the French ImadGIST trial of adjuvant imatinib for 3 versus 6 years. A summary of the aforementioned key adjuvant trials is shown in the Table.
When imatinib is commenced, careful monitoring for treatment toxicities and drug interactions should ensue in order to improve compliance. Dose density should be maintained if possible, as retrospective studies suggest suboptimal plasma levels are associated with a worse outcome.33
When should neoadjuvant imatinib be considered?
Neoadjuvant Imatinib
Neoadjuvant imatinib should be considered for patients requiring total gastrectomy, esophagectomy, or abdominoperineal resection of the rectum in order to reduce tumor size, limit subsequent surgical morbidity, mitigate tumor bleeding and rupture, and aid with organ preservation. Patients with rectal GISTs that may otherwise warrant an abdominoperineal resection should be offered a trial of imatinib in the neoadjuvant setting. There is no evidence for the use of any other TKI aside from imatinib in the neoadjuvant or adjuvant setting. With neoadjuvant imatinib, it is difficult to accurately assess the mitotic rate in the resected tumor specimen.
The RTOG 0132/ACRIN 6665 trial was a prospective phase 2 study evaluating the efficacy of imatinib 600 mg daily in the perioperative setting.34 The trial enrolled 50 patients, 30 with primary GISTs (group A) and 22 with recurrent metastatic GISTs (group B). Based on data from the metastatic setting revealing a time to treatment response of about 2.5 months, patients were treated with 8 to 12 weeks of preoperative imatinib followed by 2 years of adjuvant imatinib. Imatinib was stopped 24 hours preoperatively and resumed as soon as possible postoperatively. In group A, 7% of patients achieved a partial response (PR), 83% achieved stable disease, and 2-year progression-free survival (PFS) and OS were 83% and 93%, respectively. In group B, 4.5% of patients achieved a PR, 91% achieved stable disease, and 4.5% experienced progressive disease in the preoperative period; the 2-year PFS and OS were 77% and 91%, respectively. The results of this trial demonstrated the feasibility of using perioperative imatinib with minimal effects on surgical outcomes and set the rationale to use neoadjuvant imatinib in select patients with borderline resectable or rectal GISTs. Another EORTC pooled analysis from 10 sarcoma centers revealed that after a median of 10 months of neoadjuvant imatinib, 83.2% of patients achieved an R0 resection and only 1% progressed during treatment.35 After a median follow-up of 46 months, the 5-year disease-free survival and OS were 65% and 87%, respectively.
Mutational testing should be performed beforehand to ensure the tumor is imatinib-sensitive. If a KIT exon 9 mutation is identified, then 400 mg twice daily should be considered (given the benefit seen with 800 mg imatinib for advanced GIST patients), although there are no studies to confirm this practice. Neoadjuvant imatinib is recommended for a total of 6 to 12 months to ensure maximal tumor debulking, but with very close monitoring and surgical input for disease resistance and growth.14 Imatinib should be stopped 1 to 2 days preoperatively and resumed once the patient has recovered from surgery for a total of 3 years (pre-/postoperatively combined). Neoadjuvant therapy has been shown to be safe and effective, but there have been no randomized trials to assess survival.
What is appropriate surveillance for resected GISTs?
Surveillance
There have been no randomized studies to guide the management of surveillance after surgical resection and adjuvant therapy. There is no known optimal follow-up schedule, but several have been proposed.13,36 Among high-risk patients, it is suggested to image every 3 to 6 months during adjuvant therapy, followed by every 3 months for 2 years after discontinuing therapy, then every 6 months for another 3 years and annually thereafter for an additional 5 years. High-risk patients usually relapse within 1 to 3 years after finishing adjuvant therapy, while low-risk patients can relapse later given that their disease can be slower growing. It has been recommended that low-risk patients undergo imaging every 6 months for 5 years, with follow-up individualized thereafter. Very-low-risk patients may not require more than annual imaging. Because most relapses occur within the peritoneum or liver, imaging should encompass the abdomen and pelvis. Surveillance imaging usually consists of CT scans of the abdomen and pelvis. MRI scans can be utilized for patients at lower risk or who are out several years in order to avoid excess radiation exposure. MRI is also specifically helpful for rectal and esophageal lesions. Chest CT or chest radiograph and bone scan are not routinely required for follow-up.
Case Conclusion
The patient receives adjuvant imatinib and experiences grade 2 myalgias, periorbital edema, and macrocytic anemia, which result in imatinib discontinuation after 3 years of treatment. He is seen every 3 to 6 months and a contrast CT abdomen and pelvis is obtained every 6 months for 5 years. During this 5-year follow-up period, he does not have any clinical or radiographic evidence of disease recurrence.
Further follow-up of this patient is presented in the second article in this 2-part review of management of GISTs.
Key Points
- GISTs are the most common mesenchymal neoplasms of the GI tract and can occasionally occur in extragastrointestinal locations as well.
- GISTs encompass a heterogeneous family of tumor subsets with different natural histories, mutations, and TKI responsiveness.
- Surgery is the mainstay of treatment for localized GISTs, with cure rates greater than 50%.
- For very small (< 2 cm) esophagogastric GISTs, endoscopic ultrasound evaluation and follow-up is recommended.
- For tumors ≥ 2 cm, biopsy and excision is the standard approach.
- For localized GISTs, complete surgical resection (R0) is standard treatment, with no lymphadenectomy for clinically negative lymph nodes.
- Mutational analysis should be considered standard of practice. It can be helpful for confirming the diagnosis and can be predictive and prognostic in determining specific TKI therapy and dose.
- Adjuvant imatinib at a dose of 400 mg for 3 years is standard of care for GISTs that are at high risk of relapse and are imatinib-sensitive, and it is the only TKI approved for adjuvant therapy. Patients with PDGFRA D842V, NF1, BRAF or SDH-deficient GISTs should not receive adjuvant imatinib therapy.
- Neoadjuvant therapy can be utilized for sites where extensive resection would lead to significant morbidity. It should be given for 6 to 12 months, but patients need to be monitored closely for tumor growth.
Gastrointestinal stromal tumors (GISTs) are the most common mesenchymal tumor of the gastrointestinal (GI) tract and arise from the interstitial cells of Cajal of the myenteric plexus. These tumors are rare, with about 1 case per 100,000 persons diagnosed in the United States annually, but may be incidentally discovered in up to 1 in 5 autopsy specimens of older adults.1,2 Epidemiologic risk factors include increasing age, with a peak incidence between age 60 and 65 years, male gender, black race, and non-Hispanic white ethnicity. Germline predisposition can also increase the risk of developing GISTs; molecular drivers of GIST include gain-of-function mutations in the KIT proto-oncogene and platelet-derived growth factor receptor α (PDGFRA) gene, which both encode structurally similar tyrosine kinase receptors; germline mutations of succinate dehydrogenase (SDH) subunit genes; and mutations associated with neurofibromatosis type 1.
GISTs most commonly involve the stomach, followed by the small intestine, but can arise anywhere within the GI tract (esophagus, colon, rectum, and anus). They can also develop outside the GI tract, arising from the mesentery, omentum, and retroperitoneum. The majority of cases are localized or locoregional, whereas about 20% are metastatic at presentation.1 GISTs can occur in children, adolescents, and young adults. Pediatric GISTs represent a distinct subset marked by female predominance and gastric origin, are often multifocal, can sometimes have lymph node involvement, and typically lack mutations in the KIT and PDGFRA genes.
This review is the first of 2 articles focusing on the diagnosis and management of GISTs. Here, we review the evaluation and diagnosis of GISTs along with management of localized disease. Management of advanced disease is reviewed in a separate article.
Case Presentation
A 64-year-old African American man with progressive iron deficiency and abdominal discomfort undergoes upper and lower endoscopy and is found to have a bulging mass within his abdominal cavity. He undergoes a computed tomography (CT) evaluation of the chest, abdomen, and pelvis with contrast, which reveals the presence of a 10-cm gastric mass, with no other lesions identified. He undergoes surgical resection of the mass and presents for review of his pathology and to discuss his treatment plan.
What histopathologic features are consistent with GIST?
What factors are used for risk stratification and to predict likelihood of recurrence?
Clinical Presentation and Diagnosis
Most patients present with symptoms of overt or occult GI bleeding or abdominal discomfort, but a significant proportion of GISTs are discovered incidentally. Lymph node involvement is not typical, except for GISTs occurring in children and/or with rare syndromes. Most syndromic GISTs are multifocal and multicentric. After surgical resection, GISTs usually recur or metastasize within the abdominal cavity, including the omentum, peritoneum, or liver. These tumors rarely spread to the lungs, brain, or bones; when tumor spread does occur, it tends to be in heavily pre-treated patients with advanced disease who have been on multiple lines of therapy for a long duration of time.
The diagnosis usually can be made by histopathology. Specimens can be obtained by endoscopic ultrasound (EUS)– or CT-guided methods, the latter of which carries a very small risk of contamination from percutaneous biopsy. In terms of morphology, GISTs can be spindle cell, epithelioid, or mixed neoplasms. Epithelioid tumors are more commonly seen in the stomach and are often PDGFRA-mutated or SDH-deficient. The differential diagnosis includes other soft-tissue GI wall tumors such as leiomyosarcomas/leiomyomas, germ cell tumors, lymphomas, fibromatosis, and neuroendocrine and neurogenic tumors. A unique feature of GISTs that differentiates them from leiomyomas is near universal expression of CD117 by immunohistochemistry (IHC); this characteristic has allowed pathologists and providers to accurately distinguish true GISTs from other GI mesenchymal tumors.3 Recently, DOG1 (discovered on GIST1) immunoreactivity has been found to be helpful in identifying patients with CD117-negative GISTs. Initially identified through gene expression analysis of GISTs, DOG1 IHC can identify the common mutant c-Kit-driven CD117-positive GISTs as well as the rare CD117-negative GISTs, which are often driven by mutated PDGFRA.4 Importantly, IHC for KIT and DOG1 are not surrogates for mutational status, nor are they predictive of tyrosine kinase inhibitor (TKI) sensitivity. If IHC of a tumor specimen is CD117- and DOG1-negative, the specimen can be sent for KIT and PDGFRA mutational analysis to confirm the diagnosis. If analysis reveals that these genes are wild-type, then IHC staining for SDH B (SDHB) should follow to assess for an SDH-deficient GIST (negative staining).
Risk Stratification for Recurrence
The clinical behavior of GISTs can be variable. Some are indolent, while others behave more aggressively, with a greater malignant potential and a higher propensity to recur and metastasize. Clinical and pathologic features can provide important prognostic information that allows providers to risk-stratify patients. Various institutions have assessed prognostic variables for GISTs. In 2001, the National Institutes of Health (NIH) held a GIST workshop that proposed an approach to estimating metastatic risk based on tumor size and mitotic index (NIH or Fletcher criteria).5 Joensuu et al later proposed a modification of the NIH risk classification to include tumor location and tumor rupture (modified NIH criteria or Joensuu criteria).6-8 Similarly, the Armed Forces Institute of Pathology (AFIP) identified tumor site as a prognostic factor, with gastric GISTs having the best prognosis (AFIP-Miettinen criteria).9-11 Tabular schemes were designed which stratified patients into discrete groups with ranges for mitotic rate and tumor size. Nomograms for ease of use were then constructed utilizing a bimodal mitotic rate and included tumor site and size.12 Finally, contour maps were developed, which have the advantage of evaluating mitotic rate and tumor size as continuous nonlinear variables and also include tumor site and rupture (associated with a high risk of peritoneal metastasis) separately, further improving risk assessment. These contour maps have been validated against pooled data from 10 series (2560 patients).13 High-risk features identified from these studies include tumor location, size, mitotic rate and tumor rupture and are now used for deciding on the use of adjuvant imatinib and as requirements to enter clinical trials assessing adjuvant therapy for resected GISTs.
Case Continued
The patient’s operative and pathology reports indicate that the tumor is a spindle cell neoplasm of the stomach that is positive for CD117, DOG1, and CD34 and negative for smooth muscle actin and S-100, consistent with a diagnosis of GIST. Resection margins are negative. There are 10 mitoses per 50 high-power fields (HPF). Per the operative report, there was no intraoperative or intraperitoneal tumor rupture. Thus, while his GIST was gastric, which generally has a more favorable prognosis, the tumor harbors high-risk features based on its size and mitotic index.
What further testing should be requested?
Molecular Alterations
It is recommended that a mutational analysis be performed as part of the diagnostic work-up of all GISTs.14 Mutational analysis can provide prognostic and predictive information for sensitivity to imatinib and should be considered standard of care. It may also be useful for confirming a GIST diagnosis, or, if negative, lead to further evaluation with an IHC stain for SDHB. The c-Kit receptor is a member of the tyrosine kinase family and, through direct interactions with stem cell factor (SCF), can upregulate the PI3K/AKT/mTOR, Ras/Raf/MEK/ERK, and JAK-STAT pathways, resulting in transcription and translation of genes that enhance cell growth and survival.15 The cell of origin of GISTs, the interstitial cells of Cajal, are dependent on the SCF–c-Kit interaction for development.16 Likewise, the large majority of GISTs (about 70%) are driven by upregulation and constitutive activation of c-Kit, which is normally autoinhibited. About 80% of KIT mutations involve exon 11; these GISTs are most often associated with a gastric location and are associated with a favorable recurrence-free survival (RFS) rate with surgery alone.17KIT exon 9 mutations are much less common, encompassing only about 10% of GIST KIT mutations, and GISTs with these mutations are more likely to arise from the small bowel.17
About 8% of GISTs harbor gain-of-function PDGFRA driver mutations rendering constitutively active PDGFRA.18PDGFRA mutations are mutually exclusive from KIT mutations, and PDGFRA-mutated tumors most often occur in the stomach. PDGFRA mutations generally are associated with a lower mitotic rate and gastric location. Identification of the PDGFRA D842V mutation on exon 18, which is the most common, is important, as it is associated with imatinib resistance, and these patients should not be offered imatinib.19
Several other mutations associated with GISTs outside of the KIT and PDGFRA spectrum have been identified. About 10% of GISTs are wildtype for KIT and PDGFRA, and not all KIT/PDGFRA-wildtype GISTs are imatinib-sensitive and/or respond to other TKIs.18 These tumors may harbor aberrations in SDH and NF1, or less commonly, BRAF V600E, FGFR, and NTRK.20,21 SDH subunits B, C and D play a role in the Krebs cycle and electron transport chain. Germline mutations in these SDH subunits can result in the Carney-Stratakis syndrome characterized by the dyad of multifocal GISTs and multicentric paragangliomas.22 This syndrome is most likely to manifest in the pediatric or young adult population. In contradistinction is the Carney triad, which is associated with acquired loss of function of the SDHC gene due to promoter hypermethylation. This syndrome classically occurs in young women and is characterized by an indolent-behaving triad of multicentric GISTs, non-adrenal paragangliomas, and pulmonary chondromas.23 Like PDGFRA D842V–mutated GISTs, SDH-deficient and NF1-associated GISTs are considered imatinib resistant, and these patients should not be offered imatinib therapy.14
Case Continued
The patient’s GIST is found to harbor a KIT exon 11 single codon deletion. He appears anxious and asks to have everything done to prevent his GIST from coming back and to improve his lifespan.
What are the next steps in the management of this patient?
Management
A multidisciplinary team approach to the management of all GISTs is essential and includes input from radiology, gastroenterology, pathology, medical and surgical oncology, nuclear medicine, and nursing.
Surgical Resection
Small esophagogastric and duodenal GISTs ≤ 2 cm can be asymptomatic and managed with serial endoscopic surveillance, typically every 6 to 12 months, with biopsies if the tumors increase in size. GISTs larger than 2 cm require surgical resection, with resection of the full pseudocapsule and an R0 resection, if possible, since larger GISTs carry a higher risk of growth and recurrence. If an R0 resection would lead to significant morbidity or functional sequelae, an R1 may suffice. Rectal GISTs are an exception, where microscopic margins have been shown to be associated with an increased risk of local failure.24 It is important to explore the abdomen thoroughly for peritoneal, rectovaginal, and vesicular implants and metastasis to the liver. A formal lymph node dissection is not necessary because lymph nodes are rarely involved and should only be removed when clinically suspicious. Tumor rupture must be avoided. A laparoscopic approach should only be considered for smaller tumors, since there is a risk of tumor rupture with larger tumors.14
When is adjuvant imatinib indicated?
Adjuvant Imatinib
Among patients with local or locally advanced GISTs, the risk of death from recurrence with surgery alone can be high, with a historical 5-year overall survival (OS) of about 35%.25 As a result, multiple studies have assessed the benefit of adjuvant imatinib, which is now considered standard of care for patients with imatinib-sensitive, high-risk GISTs. In addition to inhibiting BCR-ABL, imatinib mesylate inhibits multiple other receptor tyrosine kinases, including PDGFR, SCF and c-Kit. As a result, imatinib has demonstrated in vitro inhibition of cell proliferation and apoptosis and clinical activity against GISTs expressing CD117.26 Importantly, adjuvant imatinib should only be offered to patients with imatinib-sensitive mutations, such as KIT exon 11 and KIT exon 9 mutations. Adjuvant imatinib should not be offered to patients with imatinib-insensitive mutations such as PDGFR 842V, NF1, or BRAF-related or SDH-deficient GISTs.
The ACOSOG Z9000 was the first study of adjuvant imatinib in patients with resected GISTs.25 This was a single-arm, phase 2 study involving 106 patients with surgically resected GISTs deemed high-risk for recurrence, defined as size > 10 cm, tumor rupture, or up to 4 peritoneal implants. Patients were treated with imatinib 400 mg daily for 1 year. The primary and secondary endpoints were OS and RFS, respectively. Long-term follow-up of this study demonstrated 1-, 3-, and 5-year OS of 99%, 97%, and 83%, and 1-, 3-, and 5-year RFS of 96%, 60%, and 40%, which compared favorably with historical controls. In a multivariable analysis, increasing tumor size, small bowel location, KIT exon 9 mutation, high mitotic rate, and older age were independent risk factors for a poor RFS.25 It is important to note that the benefit of adjuvant imatinib waned after discontinuation of therapy, creating a rationale to study adjuvant imatinib for longer periods of time.
As a result of the promising phase 2 data, ACOSOG opened a phase 3 randomized trial (Z9001) comparing 1 year of adjuvant imatinib to placebo among patients with surgically resected GISTs that were > 3 cm in size and that stained positive for CD117 on pathology. The trial accrued 713 patients and was stopped early at a planned interim analysis, which revealed a 1-year RFS of 98% for imatinib versus 83% for placebo (hazard ratio [HR], 0.35; P < 0.001). The 1-year OS did not differ between the 2 arms (92.2% vs 99.7%; HR, 0.66; P = 0.47).27 When comparing the 2 arms, imatinib was associated with a higher RFS among patients with a KIT exon 11 deletion, but not among patients with other KIT mutation types, PDGFRA mutations, or who were KIT/PDGFRA wildtype.28 Imatinib was granted approval by the US Food and Drug Administration (FDA) for the adjuvant treatment of high-risk GISTs based on the results of the ACOSOG Z9001 trial.
The EORTC 62024 study was a randomized placebo-controlled trial assessing the benefit of 2 years of adjuvant imatinib.29 Patients had to be considered intermediate or high risk per the 2002 NIH consensus classification to be eligible. The trial enrolled 918 patients. The 5-year OS rate, the original primary endpoint, did not differ between the 2 groups (100% vs 99%). The 3-year and 5-year RFS rates, secondary endpoints, were significantly longer among patients treated with imatinib (84% vs 66% and 69% vs 63%, respectively). Again, it was noted that the benefit of imatinib waned over time after treatment discontinuation.
The Scandinavian Sarcoma Group (SSG XVIII) trial was a prospective randomized phase 3 trial that compared 3 years versus 1 year of adjuvant imatinib.30 Patients had to be enrolled within 12 weeks of the postoperative period and had to have GISTs that were CD117-positive and with a high estimated risk of recurrence, per the modified NIH consensus criteria (size > 10 cm, > 10 mitoses per 50 HPF, diameter > 5 cm with mitotic count > 5, or tumor rupture before or at surgery). Three years of adjuvant imatinib was associated with a 54% reduction in the hazard for recurrence at 5 years (65.6% vs 47.9%; HR, 0.46; P < 0.001) and a 55% reduction in the hazard for death at 5 years (OS 92% vs 81.7%; HR, 0.45; P = 0.02). Based on the results of this study, the FDA granted approval for the use of 3 years of adjuvant imatinib in patients with high-risk resected GISTs.
The observation that a longer duration of adjuvant imatinib was associated with superior RFS and OS led to studies to further explore longer durations of adjuvant imatinib. The PERSIST-5 (Postresection Evaluation of Recurrence-free Survival for Gastrointestinal Stromal Tumors With 5 Years of Adjuvant Imatinib) was a multicenter, single-arm, phase 2 prospective study of adjuvant imatinib with a primary endpoint of RFS after 5 years.31 Patients had to have an intermediate or high risk of recurrence, which included GISTs at any site > 2 cm with > 5 mitoses per 50 HPF or nongastric GISTs that were ≥ 5 cm. With 91 patients enrolled, the estimated 5-year RFS was 90% and the OS was 95%. Of note, about half of the patients stopped treatment early due to a variety of reasons, including patient choice or adverse events. Importantly, there were no recurrences in patients with imatinib-sensitive mutations while on therapy. We know that in patients at high risk of relapse, adjuvant imatinib delays recurrence and improves survival, but whether any patients are cured, or their survival curves are just shifted to the right, is unknown. Only longer follow-up of existing studies, and the results of newer trials utilizing longer durations of adjuvant treatment, will help to determine the real value of adjuvant therapy for GIST patients.32 Based on this study, it would be reasonable to discuss a longer duration of imatinib with patients deemed to be at very high risk of recurrence and who are tolerating therapy well. We are awaiting the data from the randomized phase 3 Scandinavian Sarcoma Group XII trial comparing 5 years versus 3 years of adjuvant imatinib therapy, and from the French ImadGIST trial of adjuvant imatinib for 3 versus 6 years. A summary of the aforementioned key adjuvant trials is shown in the Table.
When imatinib is commenced, careful monitoring for treatment toxicities and drug interactions should ensue in order to improve compliance. Dose density should be maintained if possible, as retrospective studies suggest suboptimal plasma levels are associated with a worse outcome.33
When should neoadjuvant imatinib be considered?
Neoadjuvant Imatinib
Neoadjuvant imatinib should be considered for patients requiring total gastrectomy, esophagectomy, or abdominoperineal resection of the rectum in order to reduce tumor size, limit subsequent surgical morbidity, mitigate tumor bleeding and rupture, and aid with organ preservation. Patients with rectal GISTs that may otherwise warrant an abdominoperineal resection should be offered a trial of imatinib in the neoadjuvant setting. There is no evidence for the use of any other TKI aside from imatinib in the neoadjuvant or adjuvant setting. With neoadjuvant imatinib, it is difficult to accurately assess the mitotic rate in the resected tumor specimen.
The RTOG 0132/ACRIN 6665 trial was a prospective phase 2 study evaluating the efficacy of imatinib 600 mg daily in the perioperative setting.34 The trial enrolled 50 patients, 30 with primary GISTs (group A) and 22 with recurrent metastatic GISTs (group B). Based on data from the metastatic setting revealing a time to treatment response of about 2.5 months, patients were treated with 8 to 12 weeks of preoperative imatinib followed by 2 years of adjuvant imatinib. Imatinib was stopped 24 hours preoperatively and resumed as soon as possible postoperatively. In group A, 7% of patients achieved a partial response (PR), 83% achieved stable disease, and 2-year progression-free survival (PFS) and OS were 83% and 93%, respectively. In group B, 4.5% of patients achieved a PR, 91% achieved stable disease, and 4.5% experienced progressive disease in the preoperative period; the 2-year PFS and OS were 77% and 91%, respectively. The results of this trial demonstrated the feasibility of using perioperative imatinib with minimal effects on surgical outcomes and set the rationale to use neoadjuvant imatinib in select patients with borderline resectable or rectal GISTs. Another EORTC pooled analysis from 10 sarcoma centers revealed that after a median of 10 months of neoadjuvant imatinib, 83.2% of patients achieved an R0 resection and only 1% progressed during treatment.35 After a median follow-up of 46 months, the 5-year disease-free survival and OS were 65% and 87%, respectively.
Mutational testing should be performed beforehand to ensure the tumor is imatinib-sensitive. If a KIT exon 9 mutation is identified, then 400 mg twice daily should be considered (given the benefit seen with 800 mg imatinib for advanced GIST patients), although there are no studies to confirm this practice. Neoadjuvant imatinib is recommended for a total of 6 to 12 months to ensure maximal tumor debulking, but with very close monitoring and surgical input for disease resistance and growth.14 Imatinib should be stopped 1 to 2 days preoperatively and resumed once the patient has recovered from surgery for a total of 3 years (pre-/postoperatively combined). Neoadjuvant therapy has been shown to be safe and effective, but there have been no randomized trials to assess survival.
What is appropriate surveillance for resected GISTs?
Surveillance
There have been no randomized studies to guide the management of surveillance after surgical resection and adjuvant therapy. There is no known optimal follow-up schedule, but several have been proposed.13,36 Among high-risk patients, it is suggested to image every 3 to 6 months during adjuvant therapy, followed by every 3 months for 2 years after discontinuing therapy, then every 6 months for another 3 years and annually thereafter for an additional 5 years. High-risk patients usually relapse within 1 to 3 years after finishing adjuvant therapy, while low-risk patients can relapse later given that their disease can be slower growing. It has been recommended that low-risk patients undergo imaging every 6 months for 5 years, with follow-up individualized thereafter. Very-low-risk patients may not require more than annual imaging. Because most relapses occur within the peritoneum or liver, imaging should encompass the abdomen and pelvis. Surveillance imaging usually consists of CT scans of the abdomen and pelvis. MRI scans can be utilized for patients at lower risk or who are out several years in order to avoid excess radiation exposure. MRI is also specifically helpful for rectal and esophageal lesions. Chest CT or chest radiograph and bone scan are not routinely required for follow-up.
Case Conclusion
The patient receives adjuvant imatinib and experiences grade 2 myalgias, periorbital edema, and macrocytic anemia, which result in imatinib discontinuation after 3 years of treatment. He is seen every 3 to 6 months and a contrast CT abdomen and pelvis is obtained every 6 months for 5 years. During this 5-year follow-up period, he does not have any clinical or radiographic evidence of disease recurrence.
Further follow-up of this patient is presented in the second article in this 2-part review of management of GISTs.
Key Points
- GISTs are the most common mesenchymal neoplasms of the GI tract and can occasionally occur in extragastrointestinal locations as well.
- GISTs encompass a heterogeneous family of tumor subsets with different natural histories, mutations, and TKI responsiveness.
- Surgery is the mainstay of treatment for localized GISTs, with cure rates greater than 50%.
- For very small (< 2 cm) esophagogastric GISTs, endoscopic ultrasound evaluation and follow-up is recommended.
- For tumors ≥ 2 cm, biopsy and excision is the standard approach.
- For localized GISTs, complete surgical resection (R0) is standard treatment, with no lymphadenectomy for clinically negative lymph nodes.
- Mutational analysis should be considered standard of practice. It can be helpful for confirming the diagnosis and can be predictive and prognostic in determining specific TKI therapy and dose.
- Adjuvant imatinib at a dose of 400 mg for 3 years is standard of care for GISTs that are at high risk of relapse and are imatinib-sensitive, and it is the only TKI approved for adjuvant therapy. Patients with PDGFRA D842V, NF1, BRAF or SDH-deficient GISTs should not receive adjuvant imatinib therapy.
- Neoadjuvant therapy can be utilized for sites where extensive resection would lead to significant morbidity. It should be given for 6 to 12 months, but patients need to be monitored closely for tumor growth.
1. Ma GL, Murphy JD, Martinez ME et al. Epidemiology of gastrointestinal stromal tumors in the era of histology codes: results of a population-based study. Cancer Epidemiol Biomarkers Prev. 2015;24:298-302.
2. Agaimy A, Wunsch PH, Hofstaedter F, et al. Minute gastric sclerosing stromal tumors (GIST tumorlets) are common in adults and frequently show c-KIT mutations. Am J Surg Pathol. 2007;31:113-120.
3. Miettinen M, Sobin LH, Sarlomo-Rikala M. Immunohistochemical spectrum of GISTs at different sites and their differential diagnosis with a reference to CD117 (KIT). Mod Pathol. 2000;13:1134-1142.
4. West RB, Corless CL, Chen X, et al. The novel marker, DOG1, is expressed ubiquitously in gastrointestinal stromal tumors irrespective of KIT or PDGFRA mutational status. Am J Pathol. 2004;165:107-113.
5. Fletcher CD, Berman JJ, Corless C, et al. Diagnosis of gastrointestinal stromal tumors: a consensus approach. Int J Surg Pathol. 2002;10:81-89.
6. Joensuu H. Risk stratification of patients diagnosed with gastrointestinal stromal tumor. Hum Pathol. 2008;39:1411-1419.
7. Hohenberger P, Ronellenfitsch U, Oladeji O, et al. Pattern of recurrence in patients with ruptured primary gastrointestinal stromal tumor. Br J Surg. 2010;97:1854-1859.
8. Holmenbakk T, Bjerkehagen B, Boye K, et al. Definition and clinical significance of tumor rupture in gastrointestinal stromal tumours of the small intestine. Br J Surg. 2016;103:684-691.
9. Emory TS, Sobin LH, Lukes L, et al. Prognosis of gastrointestinal smooth-muscle (stromal) tumors: dependence on anatomic site. Am J Surg Pathol. 1999;23:82-87.
10. Miettinen M, Makhlouf H, Sobin LH, Lasota J. Gastrointestinal stromal tumors of the jejunum and ileum: a clinicopathologic, immunohistochemical, and molecular genetic study of 906 cases before imatinib with long-term follow-up. Am J Surg Pathol. 2006;30:477-489.
11. Miettinen M, Sobin LH, Lasota J. Gastrointestinal stromal tumors of the stomach: a clinicopathologic, immunohistochemical, and molecular genetic study of 1765 cases with long-term follow-up. Am J Surg Pathol. 2005;29:52-68.
12. Gold JS, Gonen M, Gutierrez A, et al. Development and validation of a prognostic nomogram for recurrence-free survival after complete surgical resection of localized primary gastrointestinal stromal tumour: a retrospective analysis. Lancet Oncol. 2009;10:1045-1052.
13. Joensuu H, Vehtari A, Rihimaki J et al. Risk of recurrence of gastrointestinal stromal tumor after surgery: an analysis of pooled population-based cohorts. Lancet Oncol. 2012;13:265-274.
14. Casali PG, Abecassis N, Bauer S, et al. Gastrointestinal stromal tumours: ESMO-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow up. Ann Oncol. 2018;29(Supplement_4): iv267.
15. Jing L, Yan-Ling W, Bing-Jia C, et al. The c-kit receptor-mediated signal transduction and tumor-related diseases. Int J Biol Sci. 2013;9:435-443.
16. Hirota S, Isozaki K, Moriyama Y, et al. Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science. 1998;279:577-580.
17. Joensuu H, Rutkowski P, Nishida T, et al. KIT and PDGFRA mutations and the risk of GI stromal tumor recurrence. J Clin Oncol. 2015;33:634-642.
18. Corless CL, Fletcher JA, Heinrich MC. Biology of gastrointestinal stromal tumors. J Clin Oncol. 2004;22:3813-3825.
19. Heinrich MC, Corless CL, Demetri GD, et al. Kinase mutations and imatinib response in patients with metastatic gastrointestinal stromal tumor. J Clin Oncol. 2003;21:4342-4349.
20. Huss S, Pasternack H, Ihle MA, et al. Clinicopathological and molecular features of a large cohort of gastrointestinal stromal tumors (GISTs) and review of the literature: BRAF mutations in KIT/PDGFRA wild-type GISTs are rare events. Hum Pathol. 2017;62:206-214.
21. Shi E, Chmielecki J, Tang CM, et al. FGFR1 and NTRK3 actionable alterations in “Wild-Type” gastrointestinal stromal tumors. J Transl Med. 2016;14:339.
22. Carney JA, Stratakis CA. Familial paraganglioma and gastric stromal sarcoma: a new syndrome distinct from the Carney triad. Am J Med Genet. 2002;108:132-139.
23. Carney JA. Gastric stromal sarcoma, pulmonary chondroma, and extra-adrenal paraganglioma (Carney Triad): natural history, adrenocortical component, and possible familial occurrence. Mayo Clin Proc. 1999;74:543-552.
24. Jakob J, Mussi C, Ronellenfitsch U, et al. Gastrointestinal stromal tumor of the rectum: results of surgical and multimodality therapy in the era of imatinib. Ann Surg Oncol. 2013;20:586-592.
25. DeMatteo RP, Ballman KV, Antonescu CR, et al. Long-term results of adjuvant imatinib mesylate in localized, high-risk, primary gastrointestinal stromal tumor (GIST): ACOSOG Z9000 (Alliance) intergroup phase 2 trial. Ann Surg. 2013;258:422-429.
26. Gleevac (imatinib) [package insert]. East Hanover, NJ: Novartis Pharmaceuticals; 2016.
27. DeMatteo RP, Ballman KV, Antonescu CR, et al. Placebo-controlled randomized trial of adjuvant imatinib mesylate following the resection of localized, primary gastrointestinal stromal tumor (GIST). Lancet. 2009;373:1097-1104.
28. Corless CL, Ballman KV, Antonescu CR, et al. Pathologic and molecular features correlate with long-term outcome after adjuvant therapy of resected primary GI stromal tumor: the ACOSOG Z9001 trial. J Clin Oncol. 2014;32:1563-1570.
29. Casali PG, Le Cesne A, Poveda Velasco A, et al. Imatinib failure-free survival (IFS) in patients with localized gastrointestinal stromal tumors (GIST) treated with adjuvant imatinib (IM): the EORTC/AGITG/FSG/GEIS/ISG randomized controlled phase III trial. J Clin Oncol. 2013;31. Abstract 10500.
30. Joensuu H, Eriksson M, Sundby HK, et al. One vs three years of adjuvant imatinib for operable gastrointestinal stromal tumor: a randomized trial. JAMA. 2012;307:1265-1272.
31. Raut CP, Espat NJ, Maki RG, et al. Efficacy and tolerability of 5-year adjuvant imatinib treatment for patients with resected intermediate- or high-risk primary gastrointestinal stromal tumor: The PERSIST-5 Clinical Trial. JAMA Oncol. 2018: e184060.
32. Benjamin RS, Casali PG. Adjuvant imatinib for GI stromal tumors: when and for how long? J Clin Oncol. 2016;34:215-218.
33. Demetri GD, Wang Y, Wehrle E, et al. Imatinib plasma levels are correlated with clinical benefit in patients with unresectable/metastatic gastrointestinal stromal tumors. J Clin Oncol. 2009;27:3141-3147.
34. Eisenberg BL, Harris J, Blanke CD, et al. Phase II trial of neoadjuvant/adjuvant imatinib mesylate (IM) for advanced primary and metastatic/recurrent operable gastrointestinal stromal tumor (GIST): early results of RTOG 0132/ACRIN 6665. J Surg Oncol. 2009;99:42-47.
35. Rutkowski P, Gronchi A, Hohenberger P, et al. Neoadjuvant imatinib in locally advanced gastrointestinal stromal tumors (GIST): the EORTC STBSG experience. Ann Surg Oncol. 2013;20:2937-2943.
36. Joensuu H, Martin-Broto J, Nishida T, et al. Follow-up strategies for patients with gastrointestinal stromal tumour treated with or without adjuvant imatinib after surgery. Eur J Cancer. 2015;51:1611-1617.
1. Ma GL, Murphy JD, Martinez ME et al. Epidemiology of gastrointestinal stromal tumors in the era of histology codes: results of a population-based study. Cancer Epidemiol Biomarkers Prev. 2015;24:298-302.
2. Agaimy A, Wunsch PH, Hofstaedter F, et al. Minute gastric sclerosing stromal tumors (GIST tumorlets) are common in adults and frequently show c-KIT mutations. Am J Surg Pathol. 2007;31:113-120.
3. Miettinen M, Sobin LH, Sarlomo-Rikala M. Immunohistochemical spectrum of GISTs at different sites and their differential diagnosis with a reference to CD117 (KIT). Mod Pathol. 2000;13:1134-1142.
4. West RB, Corless CL, Chen X, et al. The novel marker, DOG1, is expressed ubiquitously in gastrointestinal stromal tumors irrespective of KIT or PDGFRA mutational status. Am J Pathol. 2004;165:107-113.
5. Fletcher CD, Berman JJ, Corless C, et al. Diagnosis of gastrointestinal stromal tumors: a consensus approach. Int J Surg Pathol. 2002;10:81-89.
6. Joensuu H. Risk stratification of patients diagnosed with gastrointestinal stromal tumor. Hum Pathol. 2008;39:1411-1419.
7. Hohenberger P, Ronellenfitsch U, Oladeji O, et al. Pattern of recurrence in patients with ruptured primary gastrointestinal stromal tumor. Br J Surg. 2010;97:1854-1859.
8. Holmenbakk T, Bjerkehagen B, Boye K, et al. Definition and clinical significance of tumor rupture in gastrointestinal stromal tumours of the small intestine. Br J Surg. 2016;103:684-691.
9. Emory TS, Sobin LH, Lukes L, et al. Prognosis of gastrointestinal smooth-muscle (stromal) tumors: dependence on anatomic site. Am J Surg Pathol. 1999;23:82-87.
10. Miettinen M, Makhlouf H, Sobin LH, Lasota J. Gastrointestinal stromal tumors of the jejunum and ileum: a clinicopathologic, immunohistochemical, and molecular genetic study of 906 cases before imatinib with long-term follow-up. Am J Surg Pathol. 2006;30:477-489.
11. Miettinen M, Sobin LH, Lasota J. Gastrointestinal stromal tumors of the stomach: a clinicopathologic, immunohistochemical, and molecular genetic study of 1765 cases with long-term follow-up. Am J Surg Pathol. 2005;29:52-68.
12. Gold JS, Gonen M, Gutierrez A, et al. Development and validation of a prognostic nomogram for recurrence-free survival after complete surgical resection of localized primary gastrointestinal stromal tumour: a retrospective analysis. Lancet Oncol. 2009;10:1045-1052.
13. Joensuu H, Vehtari A, Rihimaki J et al. Risk of recurrence of gastrointestinal stromal tumor after surgery: an analysis of pooled population-based cohorts. Lancet Oncol. 2012;13:265-274.
14. Casali PG, Abecassis N, Bauer S, et al. Gastrointestinal stromal tumours: ESMO-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow up. Ann Oncol. 2018;29(Supplement_4): iv267.
15. Jing L, Yan-Ling W, Bing-Jia C, et al. The c-kit receptor-mediated signal transduction and tumor-related diseases. Int J Biol Sci. 2013;9:435-443.
16. Hirota S, Isozaki K, Moriyama Y, et al. Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science. 1998;279:577-580.
17. Joensuu H, Rutkowski P, Nishida T, et al. KIT and PDGFRA mutations and the risk of GI stromal tumor recurrence. J Clin Oncol. 2015;33:634-642.
18. Corless CL, Fletcher JA, Heinrich MC. Biology of gastrointestinal stromal tumors. J Clin Oncol. 2004;22:3813-3825.
19. Heinrich MC, Corless CL, Demetri GD, et al. Kinase mutations and imatinib response in patients with metastatic gastrointestinal stromal tumor. J Clin Oncol. 2003;21:4342-4349.
20. Huss S, Pasternack H, Ihle MA, et al. Clinicopathological and molecular features of a large cohort of gastrointestinal stromal tumors (GISTs) and review of the literature: BRAF mutations in KIT/PDGFRA wild-type GISTs are rare events. Hum Pathol. 2017;62:206-214.
21. Shi E, Chmielecki J, Tang CM, et al. FGFR1 and NTRK3 actionable alterations in “Wild-Type” gastrointestinal stromal tumors. J Transl Med. 2016;14:339.
22. Carney JA, Stratakis CA. Familial paraganglioma and gastric stromal sarcoma: a new syndrome distinct from the Carney triad. Am J Med Genet. 2002;108:132-139.
23. Carney JA. Gastric stromal sarcoma, pulmonary chondroma, and extra-adrenal paraganglioma (Carney Triad): natural history, adrenocortical component, and possible familial occurrence. Mayo Clin Proc. 1999;74:543-552.
24. Jakob J, Mussi C, Ronellenfitsch U, et al. Gastrointestinal stromal tumor of the rectum: results of surgical and multimodality therapy in the era of imatinib. Ann Surg Oncol. 2013;20:586-592.
25. DeMatteo RP, Ballman KV, Antonescu CR, et al. Long-term results of adjuvant imatinib mesylate in localized, high-risk, primary gastrointestinal stromal tumor (GIST): ACOSOG Z9000 (Alliance) intergroup phase 2 trial. Ann Surg. 2013;258:422-429.
26. Gleevac (imatinib) [package insert]. East Hanover, NJ: Novartis Pharmaceuticals; 2016.
27. DeMatteo RP, Ballman KV, Antonescu CR, et al. Placebo-controlled randomized trial of adjuvant imatinib mesylate following the resection of localized, primary gastrointestinal stromal tumor (GIST). Lancet. 2009;373:1097-1104.
28. Corless CL, Ballman KV, Antonescu CR, et al. Pathologic and molecular features correlate with long-term outcome after adjuvant therapy of resected primary GI stromal tumor: the ACOSOG Z9001 trial. J Clin Oncol. 2014;32:1563-1570.
29. Casali PG, Le Cesne A, Poveda Velasco A, et al. Imatinib failure-free survival (IFS) in patients with localized gastrointestinal stromal tumors (GIST) treated with adjuvant imatinib (IM): the EORTC/AGITG/FSG/GEIS/ISG randomized controlled phase III trial. J Clin Oncol. 2013;31. Abstract 10500.
30. Joensuu H, Eriksson M, Sundby HK, et al. One vs three years of adjuvant imatinib for operable gastrointestinal stromal tumor: a randomized trial. JAMA. 2012;307:1265-1272.
31. Raut CP, Espat NJ, Maki RG, et al. Efficacy and tolerability of 5-year adjuvant imatinib treatment for patients with resected intermediate- or high-risk primary gastrointestinal stromal tumor: The PERSIST-5 Clinical Trial. JAMA Oncol. 2018: e184060.
32. Benjamin RS, Casali PG. Adjuvant imatinib for GI stromal tumors: when and for how long? J Clin Oncol. 2016;34:215-218.
33. Demetri GD, Wang Y, Wehrle E, et al. Imatinib plasma levels are correlated with clinical benefit in patients with unresectable/metastatic gastrointestinal stromal tumors. J Clin Oncol. 2009;27:3141-3147.
34. Eisenberg BL, Harris J, Blanke CD, et al. Phase II trial of neoadjuvant/adjuvant imatinib mesylate (IM) for advanced primary and metastatic/recurrent operable gastrointestinal stromal tumor (GIST): early results of RTOG 0132/ACRIN 6665. J Surg Oncol. 2009;99:42-47.
35. Rutkowski P, Gronchi A, Hohenberger P, et al. Neoadjuvant imatinib in locally advanced gastrointestinal stromal tumors (GIST): the EORTC STBSG experience. Ann Surg Oncol. 2013;20:2937-2943.
36. Joensuu H, Martin-Broto J, Nishida T, et al. Follow-up strategies for patients with gastrointestinal stromal tumour treated with or without adjuvant imatinib after surgery. Eur J Cancer. 2015;51:1611-1617.
Gastrointestinal Stromal Tumors: Management of Advanced Disease
Most advanced gastrointestinal stromal tumors (GISTs) are due to a recurrence of localized disease, with only a small minority presenting with metastatic disease.1 Compared with chemotherapy, tyrosine kinase inhibitors (TKIs) have significantly improved the natural history of the disease, with median overall survival (OS) increasing from less than 1 year to about 5 years and approximately 1 in 5 patients achieving long-term survival.2 In addition, newer drugs in development and in clinical trials appear promising and have the potential to improve outcomes even further. This article reviews current evidence on options for treating metastatic or recurrent GISTs and GISTs that have progressed following initial therapy. The evaluation and diagnosis of GIST along with management of localized disease are reviewed in a separate article.
Case Presentation
A 64-year-old African American man underwent surgical resection of a 10-cm gastric mass, which pathology reported was positive for CD117, DOG1, and CD34 and negative for smooth muscle actin and S-100, consistent with a diagnosis of GIST. There were 10 mitoses per 50 HPF, and there was no intraoperative or intraperitoneal tumor rupture. The patient was treated with adjuvant imatinib, which was discontinued after 3 years due to grade 2 myalgias, periorbital edema, and macrocytic anemia. Surveillance included office visits every 3 to 6 months and a contrast CT abdomen and pelvis every 6 months. For the past 5 years, he has not had any clinical or radiographic evidence of disease recurrence. New imaging reveals multiple liver metastases and peritoneal implants. He feels fatigued and has lost about 10 lb since his last visit. He is 5 years out from his initial diagnosis and 2 years out from last receiving imatinib. His original tumor harbored a KIT exon 11 deletion.
What treatment should you recommend now?
Imatinib for Advanced GISTs
Before the first report of the efficacy of imatinib for metastatic GISTs in 2002, patients with advanced unresectable or metastatic GISTs were routinely treated with doxorubicin-based chemotherapy regimens, which were largely ineffective, with response rates (RRs) of around 5% and a median overall survival (OS) of less than 1 year.3,4 In 2002 a landmark phase 2 study revealed imatinib’s significant efficacy profile in advanced or metastatic GISTs, resulting in its approval by the US Food and Drug Administration (FDA).5 In this study, 147 patients with CD117-positive GISTs were randomly assigned to receive daily imatinib 400 mg or 600 mg for up to 36 months. The RRs were similar between the 2 groups (68.5% vs 67.6%), with a median time to response of 12 weeks and median duration of response of 118 days. Results of this study were much more favorable when compared to doxorubicin, rendering imatinib the new standard of care for advanced GISTs. A long-term follow-up of this study after a median of 63 months confirmed near identical RRs, progression-free survival (PFS), and median survival of 57 months among the 2 groups.6
Imatinib Daily Dosing
Although 400 mg of daily imatinib proved to be efficacious, it was unclear if a dose-response relationship existed for imatinib. An EORTC phase 2 study demonstrated a benefit of using a higher dose of imatinib at 400 mg twice daily, producing a RR of 71% (4% complete , 67% partial) and 1-year PFS of 73%, which appeared favorable compared with once-daily dosing and set the framework for larger phase 3 studies.7 Two phase 3 studies compared imatinib 400 mg once daily versus twice daily (until disease progression or unacceptable toxicity) among patients with CD117-positive advanced or metastatic GISTs. These studies were eventually combined into a meta-analysis (metaGIST) to compare RR, PFS and OS between the treatment groups. Both studies allowed cross-over to the 800 mg dose for patients who progressed on 400 mg daily.
The first study, conducted jointly by the EORTC, Italian Sarcoma Group, and Australasian Gastro-Intestinal Trials Group (EU-AUS),8 randomly assigned 946 patients to 400 mg once daily or twice daily. There were no differences in response rates between the groups, but the twice-daily group had a predicted 18% reduction in the hazard for progression compared with the once-daily group (estimated HR, 0.82; P = 0.026), which came at the expense of greater toxicities warranting dose reductions (60%) and treatment interruptions (64%). Cross-over to high-dose imatinib was feasible and safe, producing a partial response in 2%, stable disease in 27%, and a median PFS of 81 days. The second study was an intergroup study conducted jointly by SWOG, CALGB, NCI-C, and ECOG (S0033, US-CDN), with a nearly identical study design as the EU-AUS trial.9 The trial enrolled 746 patients. After a median follow up of 4.5 years, the median PFS and OS were not statistically different (18 vs 20 months and 55 vs 51 months, respectively). There were also no differences in response rates. One third of patients initially placed on the once-daily arm who crossed over after progression achieved a treatment response or stable disease.
The combined EU-AUS and US-CDN analysis (metaGIST) included 1640 patients with a median age of 60 years and 58% of whom were men; 818 and 822 patients were assigned to the 400 mg and 800 mg total daily doses, respectively.10 The median follow-up was 37.5 months. There were no differences in OS (49 vs 48.7 months), median PFS (18.9 vs 23.2 months), or overall response rates (51.4% vs 53.9%). Patients who had crossed over (n = 347) to the 800 mg total daily dose arm had a 7.7-month average PFS while on the higher daily dose. An analysis was performed on 377 patients in the EU-AUS trial assessing the impact of mutational status on clinical outcomes among imatinib-treated patients. KIT exon 9 activating mutations were found to be a significant independent prognostic factor for death when compared with KIT exon 11 mutations. However, the adverse prognostic value of KIT exon 9 mutations was partially overcome with higher doses of imatinib, as those who received 800 mg total had a significantly better PFS, with a 61% relative risk reduction, than those who received 400 mg. Altogether, it was concluded that imatinib 400 mg once daily should be the standard-of-care first-line treatment for advanced or metastatic GISTs, unless a KIT exon 9 mutation is present, in which case imatinib 800 mg should be considered, if 400 mg is well tolerated. In addition, patients treated with frontline imatinib at 400 mg once daily, if tolerated well, should be considered for imatinib 800 mg upon progression of disease.
Despite there being problems with secondary resistance, significant progress has occurred in the treatment of metastatic disease over a short period of time. Prior to 2000, median OS for patients with metastatic GISTs was 9 months. With the introduction of imatinib and other TKIs, the median OS has increased to 5 years, with an estimated 10-year OS rate of approximately 20%.2
Imatinib Interruption
Since at this point, imatinib was a well-established standard of care for advanced GISTs, it was questioned whether imatinib therapy could be interrupted. At this time, treatment interruption in a stop-and-go fashion was deemed feasible in other metastatic solid tumors such as colorectal cancer (OPTIMOX1).11 The BFR French trial showed that stopping imatinib therapy in patients who had a response or stable disease after 1, 3, or 5 years was generally followed by relatively rapid tumor progression (approximately 50% of patients within 6 months), even when tumors were previously removed.12 Therefore, it is recommended that treatment in the metastatic setting should be continued indefinitely, unless there is disease progression. Hence, unlike with colorectal cancer or chronic myelogenous leukemia, as of now there is no role for imatinib interruption in metastatic GISTs.
Case Continued
The patient is started on imatinib 400 mg daily, and overall he tolerates therapy well. Interval CT imaging reveals a treatment response. Two years later, imaging reveals an increase in the tumor size and density with a new nodule present within a preexisting mass. There are no clinical trials in the area.
What defines tumor progression?
Disease Progression
When GISTs are responding to treatment, on imaging the tumors can become more cystic and less dense but with an increase in size. In addition, tumor progression may not always be associated with increased size—increased density of the tumor or a nodule within a mass that may indicate progression. If CT imaging is equivocal for progression, positron emission tomography (PET) can play a role in identifying true progression. It is critically important that tumor size and density are carefully assessed when performing interval imaging. Of note, radiofrequency ablation, cryotherapy, or chemoembolization can be used for symptomatic liver metastases or oligometastatic disease. When evaluating for progression, one needs to ask patients about compliance (ie, maintaining dose intensity related to side effects of therapy as well as the financial burden of treatment—copay toxicity).
What are mechanisms of secondary imatinib resistance?
Imatinib resistance can be subtle in patients with GISTs, manifesting with new nodular, enhancing foci enclosed within a preexisting mass (resistant clonal nodule), or can be clinically or radiographically overt.13 Imatinib resistance occurs through multiple mechanisms including acquisition of secondary activating KIT mutations in the intracellular ATP-binding domain (exons 13 and 14) and the activation loop (exons 17 and 18).14
What are the treatment options for this patient?
Second-line Therapy
Sunitinib malate is a multitargeted TKI that not only targets c-Kit and PDGFRA, but also has anti-angiogenic activity through inhibition of vascular endothelial growth factor receptors (VEGFR). Sunitinib gained FDA approval for the second-line treatment of advanced GISTs based on an international double-blind trial that randomized 312 patients with imatinib-resistant metastatic GISTs in a 2:1 fashion to receive sunitinib 50 mg daily for 4 weeks on and 2 weeks off or placebo.15,16 The trial was unblinded early at the planned interim analysis, which revealed a marked benefit, producing a 66% reduction in the hazard risk of progression (27.3 vs 6.4 weeks, HR, 0.33; P < 0.001). The most common treatment-related adverse events were fatigue, diarrhea, skin discoloration, nausea, and hand-foot syndrome. Another open-label phase 2 study assessed a continuous dosing schema of sunitinib 37.5 mg daily, which has been shown to be effective with less toxicity.17 Among the 60 patients enrolled, the primary endpoint of clinical benefit rate at 24 weeks was reached in 53%, which consisted of 13% partial responses and 40% stable disease. Most toxicities were grade 1 or 2 and easily manageable through standard interventions. This has been recommended as an alternative to the initial scheduled regimen.18 Part of sunitinib’s success is its activity against GISTs harboring secondary KIT exon 13 and 14 mutations, and possibly its anti-angiogenic activity.19 Sunitinib is particularly efficacious among GISTs harboring KIT exon 9 mutations.
Third-line Therapy
Patients who have progressed on prior imatinib and sunitinib can receive third-line regorafenib, a multi-TKI that differs chemically from sorafenib by a fluorouracil group (fluoro-sorafenib). FDA approval of regorafenib was based on the phase 3 GRID (GIST Regorafenib In progressive Disease) multicenter international trial.20 This trial randomly assigned 199 patients in a 2:1 fashion to receive regorafenib 160 mg daily for 21 days out of 28-day cycles plus best supportive care (BSC) versus placebo plus BSC. Cross-over was allowed. Regorafenib significantly reduced the hazard risk of progression by 73% compared with placebo (4.8 vs 0.9 months; HR, 0.27; P < 0.001). There was no difference in OS, which may be because of cross-over (median OS, 17.4 months in both arms). As a result, regorafenib is now considered standard third-line treatment for patients with metastatic GISTs. It has a less favorable toxicity profile than imatinib, with hand-foot syndrome, transaminitis, hypertension and fatigue being the most common treatment toxicities. In order to avoid noncompliance, it is recommended to start at 80 mg and carefully titrate upwards to the 160 mg dose.
A list of landmark studies for advanced GISTs is provided in Table 1.
A summary of FDA-approved drugs for treating GISTs is provided in Table 2.
Clinical Trials
Clinical trial enrollment should be considered for all patients with advanced or unresectable GISTs throughout their treatment continuum. Owing to significant advances in genomic profiling through next-generation sequencing, multiple driver mutations have recently been identified, and targeted therapies are being explored in clinical trials.21 For example, the neurotrophic receptor tyrosine kinase (NTRK) gene appears to be mutated in a small number of advanced GISTs, and these can respond to the highly selective TRK inhibitor larotrectinib.22 Additionally, ongoing studies are assessing immunotherapies for sporadic GISTs and treatment for familial GISTs (Table 3). Some notable studies include those assessing the efficacy of agents that target KIT and PDGFR secondary mutations, including avapritinib (BLU-285) and DCC-2618, MEK inhibitors, and the multi-kinase inhibitor crenolanib for GISTs harboring the imatinib-resistant PDGFRA D842V mutation. There are also studies utilizing checkpoint inhibitors alone or in combination with imatinib.
Case Conclusion
Given the patient’s progression on imatinib, he is started on second-line sunitinib malate. He experiences grade 1 fatigue and hand-foot syndrome, which are managed supportively. After he has been on sunitinib for approximately 8 months, his disease progresses. He subsequently undergoes genomic profiling of his tumor and starts BLU-285 on a clinical trial.
Key Points
- For advanced and metastatic disease, TKIs have substantially improved the prognosis of KIT mutated GISTs, with 3 FDA-approved drugs: imatinib, sunitinib, and regorafenib. Imatinib 400 mg is the standard-of-care frontline therapy for locally advanced, unresectable, or metastatic imatinib-sensitive GISTs. If a patient has a KIT exon 9 mutation and 400 mg is well-tolerated, increasing to 800 mg is recommended. Imatinib should be continued indefinitely unless there is intolerance, a specific patient request for interruption, or progression of disease.
- When there is progression of disease in a patient with a sensitive mutation on 400 mg of imatinib, the dose can be increased to 800 mg.
- For patients who are imatinib-intolerant or have progression, standard second line is sunitinib.
- For patients who further progress or are sunitinib-intolerant, regorafenib is the standard third-line treatment.
- There needs to be close attention to side effects, drug and food interactions, and patient copay costs in order to maintain patient compliance while on TKI therapy.
- There are still major limitations in the systemic treatment of GISTs marked by their inherent genetic heterogeneity and secondary resistance. Continued translational and clinical research is needed in order to improve treatment for patients who develop secondary resistance or who have less common primary resistant mutations. Patients are encouraged to participate in clinical trials of new therapies.
Summary
GISTs are the most common mesenchymal tumors of the GI tract. They comprise an expanding landscape of tumors that are heterogenous in terms of natural history, mutations, and response to systemic treatments. The mainstay of treatment for localized GISTs is surgical resection followed by at least 3-years of adjuvant imatinib for patients with high-risk features who are imatinib-sensitive. Patients with GISTs harboring resistance mutations such as PDGFRA D842V or with SDH-deficient or NF1-associated GISTs should not receive adjuvant imatinib. Patients with more advanced GISTs and/or in difficult to resect sites harboring a sensitive mutation can be considered for neoadjuvant imatinib. Those with metastatic GISTs can receive first-, second-, and third-line imatinib, sunitinib, or regorafenib, respectively. Clinical trial enrollment should be encouraged for patients whose GISTs harbor primary imatinib-resistant mutations, and those with advanced or unresectable GISTs with secondary resistance.
1. Ma GL, Murphy JD, Martinez ME et al. Epidemiology of gastrointestinal stromal tumors in the era of histology codes: results of a population-based study. Cancer Epidemiol Biomarkers Prev. 2015;24:298-302.
2. Heinrich MC, Rankin C, Blanke CD, et al. Correlation of long-term results of imatinib in advanced gastrointestinal stromal tumors with next-generation sequencing results: analysis of phase 3 SWOG Intergroup Trial S0033. JAMA Oncol. 2017;3:944-952.
3. DeMatteo RP, Lewis JJ, Leung D, et al. Two hundred gastrointestinal stromal tumors recurrence patterns and prognostic factors for survival. Ann Surg. 2000;231:51-58.
4. Goss GA, Merriam P, Manola J, et al. Clinical and pathological characteristics of gastrointestinal stromal tumors (GIST). Prog Proc Am Soc Clin Oncol. 2000;19:599a.
5. Demetri GD, von Mehren M, Blanke CD, et al. Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N Engl J Med. 2002; 347:472-480.
6. Blanke CD, Demetri GD, von Mehren M, et al. Long-term results from a randomized phase ii trial of standard- versus higher-dose imatinib mesylate for patients with unresectable or metastatic gastrointestinal stromal tumors expressing KIT. J Clin Oncol. 2008;26:620-625.
7. Verweij J, van Oosterom A, Blay JY, et al. Imatinib mesylate (STI-571 Glivec, Gleevac) is an active agent for gastrointestinal stromal tumours, but does not yield responses in other soft-tissue sarcomas that are unselected for a molecular target. Results from an EORTC Soft Tissue and Bone Sarcoma Group phase II study. Eur J Cancer. 2003;39:2006-2011.
8. Verweij J, Casali PG, Zalcberg J, et al. Progression-free survival in gastrointestinal stromal tumours with high-dose imatinib: randomized trial. Lancet. 2004;364:1127-1134.
9. Blanke CD, Rankin C, Demetri GD, et al. Phase III randomized, intergroup trial assessing imatinib mesylate at two dose levels in patients with unresectable or metastatic gastrointestinal stromal tumors expressing the kit receptor tyrosine kinase: S0033. J Clin Oncol. 2008;26:626-632.
10. Gastrointestinal Stromal Tumor Meta-Analysis Group (MetaGIST). Comparison of two doses of imatinib for the treatment of unresectable or metastatic gastrointestinal stromal tumors: a meta-analysis of 1,640 patients. J Clin Oncol. 2010;28:1247-1253.
11. Tournigand C, Cervantes A, Figer A, et al. OPTIMOX1: a randomized study of FOLFOX4 or FOLFOX7 with oxaliplatin in a stop-and-Go fashion in advanced colorectal cancer –a GERCOR study. J Clin Oncol. 2006;24:394-400.
12. Blay JV, Cesne AL, Ray-Coquard I, et al. Prospective multicentric randomized phase iii study of imatinib in patients with advanced gastrointestinal stromal tumors comparing interruption versus continuation of treatment beyond 1 year: The French Sarcoma Group. J Clin Oncol. 2007;25:1107-1113.
13. Desai J, Shankar S, Heinrich MC, et al. Clonal evolution of resistance to imatinib in patients with metastatic gastrointestinal stromal tumors. Clin Cancer Res. 2007;13(18 Pt 1): 5398-5405.
14. Gramza AW, Corless CL, Heinrich MC. Resistance to tyrosine kinase inhibitors in gastrointestinal stromal tumors. Clin Cancer Res. 2009;15:7510-7518.
15. Sutent (sunitinib malate) [package insert]. New York, NY: Pfizer Labs; 2017.
16. Demetri GD, van Oosterom AT, Garrett CR, et al. Efficacy and safety of sunitinib in patients with advanced gastrointestinal stromal tumour after failure of imatinib: a randomized controlled trial. Lancet. 2006;368:1329-1338.
17. George S, Blay JY, Casali PG, et al. Clinical evaluation of continuous daily dosing of sunitinib malate in patients with advanced gastrointestinal stromal tumour after imatinib failure. Eur J Cancer. 2009;45:1959-1968.
18. Brennan MF, Antonescu CR, Maki RG. Management of Soft Tissue Sarcomas. Switzerland: Springer International Publishing; 2013.
19. Heinrich MC, Maki RG, Corless CL, et al. Primary and secondary kinase genotypes correlate with the biological and clinical activity of sunitinib in imatinib-resistant gastrointestinal stromal tumors. J Clin Oncol. 2008;26:5352-5359.
20. Demetri GD, Reichardt P, Kang YK, et al. Efficacy and safety of regorafenib for advanced gastrointestinal stromal tumours after failure of imatinib and sunitinib (GRID): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet. 2013;381:295-302.
21. Wilky BA, Villalobos VM. Emerging role for precision therapy through next-generation sequencing for sarcomas. JCO Precision Oncology. 2018;2:1-4.
22. Drilon A, Laetsch TW, Kummar S, et al. Efficacy of larotrectinib in trk fusion-positive cancers in adults and children. N Engl J Med. 2018;378:731-739.
Most advanced gastrointestinal stromal tumors (GISTs) are due to a recurrence of localized disease, with only a small minority presenting with metastatic disease.1 Compared with chemotherapy, tyrosine kinase inhibitors (TKIs) have significantly improved the natural history of the disease, with median overall survival (OS) increasing from less than 1 year to about 5 years and approximately 1 in 5 patients achieving long-term survival.2 In addition, newer drugs in development and in clinical trials appear promising and have the potential to improve outcomes even further. This article reviews current evidence on options for treating metastatic or recurrent GISTs and GISTs that have progressed following initial therapy. The evaluation and diagnosis of GIST along with management of localized disease are reviewed in a separate article.
Case Presentation
A 64-year-old African American man underwent surgical resection of a 10-cm gastric mass, which pathology reported was positive for CD117, DOG1, and CD34 and negative for smooth muscle actin and S-100, consistent with a diagnosis of GIST. There were 10 mitoses per 50 HPF, and there was no intraoperative or intraperitoneal tumor rupture. The patient was treated with adjuvant imatinib, which was discontinued after 3 years due to grade 2 myalgias, periorbital edema, and macrocytic anemia. Surveillance included office visits every 3 to 6 months and a contrast CT abdomen and pelvis every 6 months. For the past 5 years, he has not had any clinical or radiographic evidence of disease recurrence. New imaging reveals multiple liver metastases and peritoneal implants. He feels fatigued and has lost about 10 lb since his last visit. He is 5 years out from his initial diagnosis and 2 years out from last receiving imatinib. His original tumor harbored a KIT exon 11 deletion.
What treatment should you recommend now?
Imatinib for Advanced GISTs
Before the first report of the efficacy of imatinib for metastatic GISTs in 2002, patients with advanced unresectable or metastatic GISTs were routinely treated with doxorubicin-based chemotherapy regimens, which were largely ineffective, with response rates (RRs) of around 5% and a median overall survival (OS) of less than 1 year.3,4 In 2002 a landmark phase 2 study revealed imatinib’s significant efficacy profile in advanced or metastatic GISTs, resulting in its approval by the US Food and Drug Administration (FDA).5 In this study, 147 patients with CD117-positive GISTs were randomly assigned to receive daily imatinib 400 mg or 600 mg for up to 36 months. The RRs were similar between the 2 groups (68.5% vs 67.6%), with a median time to response of 12 weeks and median duration of response of 118 days. Results of this study were much more favorable when compared to doxorubicin, rendering imatinib the new standard of care for advanced GISTs. A long-term follow-up of this study after a median of 63 months confirmed near identical RRs, progression-free survival (PFS), and median survival of 57 months among the 2 groups.6
Imatinib Daily Dosing
Although 400 mg of daily imatinib proved to be efficacious, it was unclear if a dose-response relationship existed for imatinib. An EORTC phase 2 study demonstrated a benefit of using a higher dose of imatinib at 400 mg twice daily, producing a RR of 71% (4% complete , 67% partial) and 1-year PFS of 73%, which appeared favorable compared with once-daily dosing and set the framework for larger phase 3 studies.7 Two phase 3 studies compared imatinib 400 mg once daily versus twice daily (until disease progression or unacceptable toxicity) among patients with CD117-positive advanced or metastatic GISTs. These studies were eventually combined into a meta-analysis (metaGIST) to compare RR, PFS and OS between the treatment groups. Both studies allowed cross-over to the 800 mg dose for patients who progressed on 400 mg daily.
The first study, conducted jointly by the EORTC, Italian Sarcoma Group, and Australasian Gastro-Intestinal Trials Group (EU-AUS),8 randomly assigned 946 patients to 400 mg once daily or twice daily. There were no differences in response rates between the groups, but the twice-daily group had a predicted 18% reduction in the hazard for progression compared with the once-daily group (estimated HR, 0.82; P = 0.026), which came at the expense of greater toxicities warranting dose reductions (60%) and treatment interruptions (64%). Cross-over to high-dose imatinib was feasible and safe, producing a partial response in 2%, stable disease in 27%, and a median PFS of 81 days. The second study was an intergroup study conducted jointly by SWOG, CALGB, NCI-C, and ECOG (S0033, US-CDN), with a nearly identical study design as the EU-AUS trial.9 The trial enrolled 746 patients. After a median follow up of 4.5 years, the median PFS and OS were not statistically different (18 vs 20 months and 55 vs 51 months, respectively). There were also no differences in response rates. One third of patients initially placed on the once-daily arm who crossed over after progression achieved a treatment response or stable disease.
The combined EU-AUS and US-CDN analysis (metaGIST) included 1640 patients with a median age of 60 years and 58% of whom were men; 818 and 822 patients were assigned to the 400 mg and 800 mg total daily doses, respectively.10 The median follow-up was 37.5 months. There were no differences in OS (49 vs 48.7 months), median PFS (18.9 vs 23.2 months), or overall response rates (51.4% vs 53.9%). Patients who had crossed over (n = 347) to the 800 mg total daily dose arm had a 7.7-month average PFS while on the higher daily dose. An analysis was performed on 377 patients in the EU-AUS trial assessing the impact of mutational status on clinical outcomes among imatinib-treated patients. KIT exon 9 activating mutations were found to be a significant independent prognostic factor for death when compared with KIT exon 11 mutations. However, the adverse prognostic value of KIT exon 9 mutations was partially overcome with higher doses of imatinib, as those who received 800 mg total had a significantly better PFS, with a 61% relative risk reduction, than those who received 400 mg. Altogether, it was concluded that imatinib 400 mg once daily should be the standard-of-care first-line treatment for advanced or metastatic GISTs, unless a KIT exon 9 mutation is present, in which case imatinib 800 mg should be considered, if 400 mg is well tolerated. In addition, patients treated with frontline imatinib at 400 mg once daily, if tolerated well, should be considered for imatinib 800 mg upon progression of disease.
Despite there being problems with secondary resistance, significant progress has occurred in the treatment of metastatic disease over a short period of time. Prior to 2000, median OS for patients with metastatic GISTs was 9 months. With the introduction of imatinib and other TKIs, the median OS has increased to 5 years, with an estimated 10-year OS rate of approximately 20%.2
Imatinib Interruption
Since at this point, imatinib was a well-established standard of care for advanced GISTs, it was questioned whether imatinib therapy could be interrupted. At this time, treatment interruption in a stop-and-go fashion was deemed feasible in other metastatic solid tumors such as colorectal cancer (OPTIMOX1).11 The BFR French trial showed that stopping imatinib therapy in patients who had a response or stable disease after 1, 3, or 5 years was generally followed by relatively rapid tumor progression (approximately 50% of patients within 6 months), even when tumors were previously removed.12 Therefore, it is recommended that treatment in the metastatic setting should be continued indefinitely, unless there is disease progression. Hence, unlike with colorectal cancer or chronic myelogenous leukemia, as of now there is no role for imatinib interruption in metastatic GISTs.
Case Continued
The patient is started on imatinib 400 mg daily, and overall he tolerates therapy well. Interval CT imaging reveals a treatment response. Two years later, imaging reveals an increase in the tumor size and density with a new nodule present within a preexisting mass. There are no clinical trials in the area.
What defines tumor progression?
Disease Progression
When GISTs are responding to treatment, on imaging the tumors can become more cystic and less dense but with an increase in size. In addition, tumor progression may not always be associated with increased size—increased density of the tumor or a nodule within a mass that may indicate progression. If CT imaging is equivocal for progression, positron emission tomography (PET) can play a role in identifying true progression. It is critically important that tumor size and density are carefully assessed when performing interval imaging. Of note, radiofrequency ablation, cryotherapy, or chemoembolization can be used for symptomatic liver metastases or oligometastatic disease. When evaluating for progression, one needs to ask patients about compliance (ie, maintaining dose intensity related to side effects of therapy as well as the financial burden of treatment—copay toxicity).
What are mechanisms of secondary imatinib resistance?
Imatinib resistance can be subtle in patients with GISTs, manifesting with new nodular, enhancing foci enclosed within a preexisting mass (resistant clonal nodule), or can be clinically or radiographically overt.13 Imatinib resistance occurs through multiple mechanisms including acquisition of secondary activating KIT mutations in the intracellular ATP-binding domain (exons 13 and 14) and the activation loop (exons 17 and 18).14
What are the treatment options for this patient?
Second-line Therapy
Sunitinib malate is a multitargeted TKI that not only targets c-Kit and PDGFRA, but also has anti-angiogenic activity through inhibition of vascular endothelial growth factor receptors (VEGFR). Sunitinib gained FDA approval for the second-line treatment of advanced GISTs based on an international double-blind trial that randomized 312 patients with imatinib-resistant metastatic GISTs in a 2:1 fashion to receive sunitinib 50 mg daily for 4 weeks on and 2 weeks off or placebo.15,16 The trial was unblinded early at the planned interim analysis, which revealed a marked benefit, producing a 66% reduction in the hazard risk of progression (27.3 vs 6.4 weeks, HR, 0.33; P < 0.001). The most common treatment-related adverse events were fatigue, diarrhea, skin discoloration, nausea, and hand-foot syndrome. Another open-label phase 2 study assessed a continuous dosing schema of sunitinib 37.5 mg daily, which has been shown to be effective with less toxicity.17 Among the 60 patients enrolled, the primary endpoint of clinical benefit rate at 24 weeks was reached in 53%, which consisted of 13% partial responses and 40% stable disease. Most toxicities were grade 1 or 2 and easily manageable through standard interventions. This has been recommended as an alternative to the initial scheduled regimen.18 Part of sunitinib’s success is its activity against GISTs harboring secondary KIT exon 13 and 14 mutations, and possibly its anti-angiogenic activity.19 Sunitinib is particularly efficacious among GISTs harboring KIT exon 9 mutations.
Third-line Therapy
Patients who have progressed on prior imatinib and sunitinib can receive third-line regorafenib, a multi-TKI that differs chemically from sorafenib by a fluorouracil group (fluoro-sorafenib). FDA approval of regorafenib was based on the phase 3 GRID (GIST Regorafenib In progressive Disease) multicenter international trial.20 This trial randomly assigned 199 patients in a 2:1 fashion to receive regorafenib 160 mg daily for 21 days out of 28-day cycles plus best supportive care (BSC) versus placebo plus BSC. Cross-over was allowed. Regorafenib significantly reduced the hazard risk of progression by 73% compared with placebo (4.8 vs 0.9 months; HR, 0.27; P < 0.001). There was no difference in OS, which may be because of cross-over (median OS, 17.4 months in both arms). As a result, regorafenib is now considered standard third-line treatment for patients with metastatic GISTs. It has a less favorable toxicity profile than imatinib, with hand-foot syndrome, transaminitis, hypertension and fatigue being the most common treatment toxicities. In order to avoid noncompliance, it is recommended to start at 80 mg and carefully titrate upwards to the 160 mg dose.
A list of landmark studies for advanced GISTs is provided in Table 1.
A summary of FDA-approved drugs for treating GISTs is provided in Table 2.
Clinical Trials
Clinical trial enrollment should be considered for all patients with advanced or unresectable GISTs throughout their treatment continuum. Owing to significant advances in genomic profiling through next-generation sequencing, multiple driver mutations have recently been identified, and targeted therapies are being explored in clinical trials.21 For example, the neurotrophic receptor tyrosine kinase (NTRK) gene appears to be mutated in a small number of advanced GISTs, and these can respond to the highly selective TRK inhibitor larotrectinib.22 Additionally, ongoing studies are assessing immunotherapies for sporadic GISTs and treatment for familial GISTs (Table 3). Some notable studies include those assessing the efficacy of agents that target KIT and PDGFR secondary mutations, including avapritinib (BLU-285) and DCC-2618, MEK inhibitors, and the multi-kinase inhibitor crenolanib for GISTs harboring the imatinib-resistant PDGFRA D842V mutation. There are also studies utilizing checkpoint inhibitors alone or in combination with imatinib.
Case Conclusion
Given the patient’s progression on imatinib, he is started on second-line sunitinib malate. He experiences grade 1 fatigue and hand-foot syndrome, which are managed supportively. After he has been on sunitinib for approximately 8 months, his disease progresses. He subsequently undergoes genomic profiling of his tumor and starts BLU-285 on a clinical trial.
Key Points
- For advanced and metastatic disease, TKIs have substantially improved the prognosis of KIT mutated GISTs, with 3 FDA-approved drugs: imatinib, sunitinib, and regorafenib. Imatinib 400 mg is the standard-of-care frontline therapy for locally advanced, unresectable, or metastatic imatinib-sensitive GISTs. If a patient has a KIT exon 9 mutation and 400 mg is well-tolerated, increasing to 800 mg is recommended. Imatinib should be continued indefinitely unless there is intolerance, a specific patient request for interruption, or progression of disease.
- When there is progression of disease in a patient with a sensitive mutation on 400 mg of imatinib, the dose can be increased to 800 mg.
- For patients who are imatinib-intolerant or have progression, standard second line is sunitinib.
- For patients who further progress or are sunitinib-intolerant, regorafenib is the standard third-line treatment.
- There needs to be close attention to side effects, drug and food interactions, and patient copay costs in order to maintain patient compliance while on TKI therapy.
- There are still major limitations in the systemic treatment of GISTs marked by their inherent genetic heterogeneity and secondary resistance. Continued translational and clinical research is needed in order to improve treatment for patients who develop secondary resistance or who have less common primary resistant mutations. Patients are encouraged to participate in clinical trials of new therapies.
Summary
GISTs are the most common mesenchymal tumors of the GI tract. They comprise an expanding landscape of tumors that are heterogenous in terms of natural history, mutations, and response to systemic treatments. The mainstay of treatment for localized GISTs is surgical resection followed by at least 3-years of adjuvant imatinib for patients with high-risk features who are imatinib-sensitive. Patients with GISTs harboring resistance mutations such as PDGFRA D842V or with SDH-deficient or NF1-associated GISTs should not receive adjuvant imatinib. Patients with more advanced GISTs and/or in difficult to resect sites harboring a sensitive mutation can be considered for neoadjuvant imatinib. Those with metastatic GISTs can receive first-, second-, and third-line imatinib, sunitinib, or regorafenib, respectively. Clinical trial enrollment should be encouraged for patients whose GISTs harbor primary imatinib-resistant mutations, and those with advanced or unresectable GISTs with secondary resistance.
Most advanced gastrointestinal stromal tumors (GISTs) are due to a recurrence of localized disease, with only a small minority presenting with metastatic disease.1 Compared with chemotherapy, tyrosine kinase inhibitors (TKIs) have significantly improved the natural history of the disease, with median overall survival (OS) increasing from less than 1 year to about 5 years and approximately 1 in 5 patients achieving long-term survival.2 In addition, newer drugs in development and in clinical trials appear promising and have the potential to improve outcomes even further. This article reviews current evidence on options for treating metastatic or recurrent GISTs and GISTs that have progressed following initial therapy. The evaluation and diagnosis of GIST along with management of localized disease are reviewed in a separate article.
Case Presentation
A 64-year-old African American man underwent surgical resection of a 10-cm gastric mass, which pathology reported was positive for CD117, DOG1, and CD34 and negative for smooth muscle actin and S-100, consistent with a diagnosis of GIST. There were 10 mitoses per 50 HPF, and there was no intraoperative or intraperitoneal tumor rupture. The patient was treated with adjuvant imatinib, which was discontinued after 3 years due to grade 2 myalgias, periorbital edema, and macrocytic anemia. Surveillance included office visits every 3 to 6 months and a contrast CT abdomen and pelvis every 6 months. For the past 5 years, he has not had any clinical or radiographic evidence of disease recurrence. New imaging reveals multiple liver metastases and peritoneal implants. He feels fatigued and has lost about 10 lb since his last visit. He is 5 years out from his initial diagnosis and 2 years out from last receiving imatinib. His original tumor harbored a KIT exon 11 deletion.
What treatment should you recommend now?
Imatinib for Advanced GISTs
Before the first report of the efficacy of imatinib for metastatic GISTs in 2002, patients with advanced unresectable or metastatic GISTs were routinely treated with doxorubicin-based chemotherapy regimens, which were largely ineffective, with response rates (RRs) of around 5% and a median overall survival (OS) of less than 1 year.3,4 In 2002 a landmark phase 2 study revealed imatinib’s significant efficacy profile in advanced or metastatic GISTs, resulting in its approval by the US Food and Drug Administration (FDA).5 In this study, 147 patients with CD117-positive GISTs were randomly assigned to receive daily imatinib 400 mg or 600 mg for up to 36 months. The RRs were similar between the 2 groups (68.5% vs 67.6%), with a median time to response of 12 weeks and median duration of response of 118 days. Results of this study were much more favorable when compared to doxorubicin, rendering imatinib the new standard of care for advanced GISTs. A long-term follow-up of this study after a median of 63 months confirmed near identical RRs, progression-free survival (PFS), and median survival of 57 months among the 2 groups.6
Imatinib Daily Dosing
Although 400 mg of daily imatinib proved to be efficacious, it was unclear if a dose-response relationship existed for imatinib. An EORTC phase 2 study demonstrated a benefit of using a higher dose of imatinib at 400 mg twice daily, producing a RR of 71% (4% complete , 67% partial) and 1-year PFS of 73%, which appeared favorable compared with once-daily dosing and set the framework for larger phase 3 studies.7 Two phase 3 studies compared imatinib 400 mg once daily versus twice daily (until disease progression or unacceptable toxicity) among patients with CD117-positive advanced or metastatic GISTs. These studies were eventually combined into a meta-analysis (metaGIST) to compare RR, PFS and OS between the treatment groups. Both studies allowed cross-over to the 800 mg dose for patients who progressed on 400 mg daily.
The first study, conducted jointly by the EORTC, Italian Sarcoma Group, and Australasian Gastro-Intestinal Trials Group (EU-AUS),8 randomly assigned 946 patients to 400 mg once daily or twice daily. There were no differences in response rates between the groups, but the twice-daily group had a predicted 18% reduction in the hazard for progression compared with the once-daily group (estimated HR, 0.82; P = 0.026), which came at the expense of greater toxicities warranting dose reductions (60%) and treatment interruptions (64%). Cross-over to high-dose imatinib was feasible and safe, producing a partial response in 2%, stable disease in 27%, and a median PFS of 81 days. The second study was an intergroup study conducted jointly by SWOG, CALGB, NCI-C, and ECOG (S0033, US-CDN), with a nearly identical study design as the EU-AUS trial.9 The trial enrolled 746 patients. After a median follow up of 4.5 years, the median PFS and OS were not statistically different (18 vs 20 months and 55 vs 51 months, respectively). There were also no differences in response rates. One third of patients initially placed on the once-daily arm who crossed over after progression achieved a treatment response or stable disease.
The combined EU-AUS and US-CDN analysis (metaGIST) included 1640 patients with a median age of 60 years and 58% of whom were men; 818 and 822 patients were assigned to the 400 mg and 800 mg total daily doses, respectively.10 The median follow-up was 37.5 months. There were no differences in OS (49 vs 48.7 months), median PFS (18.9 vs 23.2 months), or overall response rates (51.4% vs 53.9%). Patients who had crossed over (n = 347) to the 800 mg total daily dose arm had a 7.7-month average PFS while on the higher daily dose. An analysis was performed on 377 patients in the EU-AUS trial assessing the impact of mutational status on clinical outcomes among imatinib-treated patients. KIT exon 9 activating mutations were found to be a significant independent prognostic factor for death when compared with KIT exon 11 mutations. However, the adverse prognostic value of KIT exon 9 mutations was partially overcome with higher doses of imatinib, as those who received 800 mg total had a significantly better PFS, with a 61% relative risk reduction, than those who received 400 mg. Altogether, it was concluded that imatinib 400 mg once daily should be the standard-of-care first-line treatment for advanced or metastatic GISTs, unless a KIT exon 9 mutation is present, in which case imatinib 800 mg should be considered, if 400 mg is well tolerated. In addition, patients treated with frontline imatinib at 400 mg once daily, if tolerated well, should be considered for imatinib 800 mg upon progression of disease.
Despite there being problems with secondary resistance, significant progress has occurred in the treatment of metastatic disease over a short period of time. Prior to 2000, median OS for patients with metastatic GISTs was 9 months. With the introduction of imatinib and other TKIs, the median OS has increased to 5 years, with an estimated 10-year OS rate of approximately 20%.2
Imatinib Interruption
Since at this point, imatinib was a well-established standard of care for advanced GISTs, it was questioned whether imatinib therapy could be interrupted. At this time, treatment interruption in a stop-and-go fashion was deemed feasible in other metastatic solid tumors such as colorectal cancer (OPTIMOX1).11 The BFR French trial showed that stopping imatinib therapy in patients who had a response or stable disease after 1, 3, or 5 years was generally followed by relatively rapid tumor progression (approximately 50% of patients within 6 months), even when tumors were previously removed.12 Therefore, it is recommended that treatment in the metastatic setting should be continued indefinitely, unless there is disease progression. Hence, unlike with colorectal cancer or chronic myelogenous leukemia, as of now there is no role for imatinib interruption in metastatic GISTs.
Case Continued
The patient is started on imatinib 400 mg daily, and overall he tolerates therapy well. Interval CT imaging reveals a treatment response. Two years later, imaging reveals an increase in the tumor size and density with a new nodule present within a preexisting mass. There are no clinical trials in the area.
What defines tumor progression?
Disease Progression
When GISTs are responding to treatment, on imaging the tumors can become more cystic and less dense but with an increase in size. In addition, tumor progression may not always be associated with increased size—increased density of the tumor or a nodule within a mass that may indicate progression. If CT imaging is equivocal for progression, positron emission tomography (PET) can play a role in identifying true progression. It is critically important that tumor size and density are carefully assessed when performing interval imaging. Of note, radiofrequency ablation, cryotherapy, or chemoembolization can be used for symptomatic liver metastases or oligometastatic disease. When evaluating for progression, one needs to ask patients about compliance (ie, maintaining dose intensity related to side effects of therapy as well as the financial burden of treatment—copay toxicity).
What are mechanisms of secondary imatinib resistance?
Imatinib resistance can be subtle in patients with GISTs, manifesting with new nodular, enhancing foci enclosed within a preexisting mass (resistant clonal nodule), or can be clinically or radiographically overt.13 Imatinib resistance occurs through multiple mechanisms including acquisition of secondary activating KIT mutations in the intracellular ATP-binding domain (exons 13 and 14) and the activation loop (exons 17 and 18).14
What are the treatment options for this patient?
Second-line Therapy
Sunitinib malate is a multitargeted TKI that not only targets c-Kit and PDGFRA, but also has anti-angiogenic activity through inhibition of vascular endothelial growth factor receptors (VEGFR). Sunitinib gained FDA approval for the second-line treatment of advanced GISTs based on an international double-blind trial that randomized 312 patients with imatinib-resistant metastatic GISTs in a 2:1 fashion to receive sunitinib 50 mg daily for 4 weeks on and 2 weeks off or placebo.15,16 The trial was unblinded early at the planned interim analysis, which revealed a marked benefit, producing a 66% reduction in the hazard risk of progression (27.3 vs 6.4 weeks, HR, 0.33; P < 0.001). The most common treatment-related adverse events were fatigue, diarrhea, skin discoloration, nausea, and hand-foot syndrome. Another open-label phase 2 study assessed a continuous dosing schema of sunitinib 37.5 mg daily, which has been shown to be effective with less toxicity.17 Among the 60 patients enrolled, the primary endpoint of clinical benefit rate at 24 weeks was reached in 53%, which consisted of 13% partial responses and 40% stable disease. Most toxicities were grade 1 or 2 and easily manageable through standard interventions. This has been recommended as an alternative to the initial scheduled regimen.18 Part of sunitinib’s success is its activity against GISTs harboring secondary KIT exon 13 and 14 mutations, and possibly its anti-angiogenic activity.19 Sunitinib is particularly efficacious among GISTs harboring KIT exon 9 mutations.
Third-line Therapy
Patients who have progressed on prior imatinib and sunitinib can receive third-line regorafenib, a multi-TKI that differs chemically from sorafenib by a fluorouracil group (fluoro-sorafenib). FDA approval of regorafenib was based on the phase 3 GRID (GIST Regorafenib In progressive Disease) multicenter international trial.20 This trial randomly assigned 199 patients in a 2:1 fashion to receive regorafenib 160 mg daily for 21 days out of 28-day cycles plus best supportive care (BSC) versus placebo plus BSC. Cross-over was allowed. Regorafenib significantly reduced the hazard risk of progression by 73% compared with placebo (4.8 vs 0.9 months; HR, 0.27; P < 0.001). There was no difference in OS, which may be because of cross-over (median OS, 17.4 months in both arms). As a result, regorafenib is now considered standard third-line treatment for patients with metastatic GISTs. It has a less favorable toxicity profile than imatinib, with hand-foot syndrome, transaminitis, hypertension and fatigue being the most common treatment toxicities. In order to avoid noncompliance, it is recommended to start at 80 mg and carefully titrate upwards to the 160 mg dose.
A list of landmark studies for advanced GISTs is provided in Table 1.
A summary of FDA-approved drugs for treating GISTs is provided in Table 2.
Clinical Trials
Clinical trial enrollment should be considered for all patients with advanced or unresectable GISTs throughout their treatment continuum. Owing to significant advances in genomic profiling through next-generation sequencing, multiple driver mutations have recently been identified, and targeted therapies are being explored in clinical trials.21 For example, the neurotrophic receptor tyrosine kinase (NTRK) gene appears to be mutated in a small number of advanced GISTs, and these can respond to the highly selective TRK inhibitor larotrectinib.22 Additionally, ongoing studies are assessing immunotherapies for sporadic GISTs and treatment for familial GISTs (Table 3). Some notable studies include those assessing the efficacy of agents that target KIT and PDGFR secondary mutations, including avapritinib (BLU-285) and DCC-2618, MEK inhibitors, and the multi-kinase inhibitor crenolanib for GISTs harboring the imatinib-resistant PDGFRA D842V mutation. There are also studies utilizing checkpoint inhibitors alone or in combination with imatinib.
Case Conclusion
Given the patient’s progression on imatinib, he is started on second-line sunitinib malate. He experiences grade 1 fatigue and hand-foot syndrome, which are managed supportively. After he has been on sunitinib for approximately 8 months, his disease progresses. He subsequently undergoes genomic profiling of his tumor and starts BLU-285 on a clinical trial.
Key Points
- For advanced and metastatic disease, TKIs have substantially improved the prognosis of KIT mutated GISTs, with 3 FDA-approved drugs: imatinib, sunitinib, and regorafenib. Imatinib 400 mg is the standard-of-care frontline therapy for locally advanced, unresectable, or metastatic imatinib-sensitive GISTs. If a patient has a KIT exon 9 mutation and 400 mg is well-tolerated, increasing to 800 mg is recommended. Imatinib should be continued indefinitely unless there is intolerance, a specific patient request for interruption, or progression of disease.
- When there is progression of disease in a patient with a sensitive mutation on 400 mg of imatinib, the dose can be increased to 800 mg.
- For patients who are imatinib-intolerant or have progression, standard second line is sunitinib.
- For patients who further progress or are sunitinib-intolerant, regorafenib is the standard third-line treatment.
- There needs to be close attention to side effects, drug and food interactions, and patient copay costs in order to maintain patient compliance while on TKI therapy.
- There are still major limitations in the systemic treatment of GISTs marked by their inherent genetic heterogeneity and secondary resistance. Continued translational and clinical research is needed in order to improve treatment for patients who develop secondary resistance or who have less common primary resistant mutations. Patients are encouraged to participate in clinical trials of new therapies.
Summary
GISTs are the most common mesenchymal tumors of the GI tract. They comprise an expanding landscape of tumors that are heterogenous in terms of natural history, mutations, and response to systemic treatments. The mainstay of treatment for localized GISTs is surgical resection followed by at least 3-years of adjuvant imatinib for patients with high-risk features who are imatinib-sensitive. Patients with GISTs harboring resistance mutations such as PDGFRA D842V or with SDH-deficient or NF1-associated GISTs should not receive adjuvant imatinib. Patients with more advanced GISTs and/or in difficult to resect sites harboring a sensitive mutation can be considered for neoadjuvant imatinib. Those with metastatic GISTs can receive first-, second-, and third-line imatinib, sunitinib, or regorafenib, respectively. Clinical trial enrollment should be encouraged for patients whose GISTs harbor primary imatinib-resistant mutations, and those with advanced or unresectable GISTs with secondary resistance.
1. Ma GL, Murphy JD, Martinez ME et al. Epidemiology of gastrointestinal stromal tumors in the era of histology codes: results of a population-based study. Cancer Epidemiol Biomarkers Prev. 2015;24:298-302.
2. Heinrich MC, Rankin C, Blanke CD, et al. Correlation of long-term results of imatinib in advanced gastrointestinal stromal tumors with next-generation sequencing results: analysis of phase 3 SWOG Intergroup Trial S0033. JAMA Oncol. 2017;3:944-952.
3. DeMatteo RP, Lewis JJ, Leung D, et al. Two hundred gastrointestinal stromal tumors recurrence patterns and prognostic factors for survival. Ann Surg. 2000;231:51-58.
4. Goss GA, Merriam P, Manola J, et al. Clinical and pathological characteristics of gastrointestinal stromal tumors (GIST). Prog Proc Am Soc Clin Oncol. 2000;19:599a.
5. Demetri GD, von Mehren M, Blanke CD, et al. Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N Engl J Med. 2002; 347:472-480.
6. Blanke CD, Demetri GD, von Mehren M, et al. Long-term results from a randomized phase ii trial of standard- versus higher-dose imatinib mesylate for patients with unresectable or metastatic gastrointestinal stromal tumors expressing KIT. J Clin Oncol. 2008;26:620-625.
7. Verweij J, van Oosterom A, Blay JY, et al. Imatinib mesylate (STI-571 Glivec, Gleevac) is an active agent for gastrointestinal stromal tumours, but does not yield responses in other soft-tissue sarcomas that are unselected for a molecular target. Results from an EORTC Soft Tissue and Bone Sarcoma Group phase II study. Eur J Cancer. 2003;39:2006-2011.
8. Verweij J, Casali PG, Zalcberg J, et al. Progression-free survival in gastrointestinal stromal tumours with high-dose imatinib: randomized trial. Lancet. 2004;364:1127-1134.
9. Blanke CD, Rankin C, Demetri GD, et al. Phase III randomized, intergroup trial assessing imatinib mesylate at two dose levels in patients with unresectable or metastatic gastrointestinal stromal tumors expressing the kit receptor tyrosine kinase: S0033. J Clin Oncol. 2008;26:626-632.
10. Gastrointestinal Stromal Tumor Meta-Analysis Group (MetaGIST). Comparison of two doses of imatinib for the treatment of unresectable or metastatic gastrointestinal stromal tumors: a meta-analysis of 1,640 patients. J Clin Oncol. 2010;28:1247-1253.
11. Tournigand C, Cervantes A, Figer A, et al. OPTIMOX1: a randomized study of FOLFOX4 or FOLFOX7 with oxaliplatin in a stop-and-Go fashion in advanced colorectal cancer –a GERCOR study. J Clin Oncol. 2006;24:394-400.
12. Blay JV, Cesne AL, Ray-Coquard I, et al. Prospective multicentric randomized phase iii study of imatinib in patients with advanced gastrointestinal stromal tumors comparing interruption versus continuation of treatment beyond 1 year: The French Sarcoma Group. J Clin Oncol. 2007;25:1107-1113.
13. Desai J, Shankar S, Heinrich MC, et al. Clonal evolution of resistance to imatinib in patients with metastatic gastrointestinal stromal tumors. Clin Cancer Res. 2007;13(18 Pt 1): 5398-5405.
14. Gramza AW, Corless CL, Heinrich MC. Resistance to tyrosine kinase inhibitors in gastrointestinal stromal tumors. Clin Cancer Res. 2009;15:7510-7518.
15. Sutent (sunitinib malate) [package insert]. New York, NY: Pfizer Labs; 2017.
16. Demetri GD, van Oosterom AT, Garrett CR, et al. Efficacy and safety of sunitinib in patients with advanced gastrointestinal stromal tumour after failure of imatinib: a randomized controlled trial. Lancet. 2006;368:1329-1338.
17. George S, Blay JY, Casali PG, et al. Clinical evaluation of continuous daily dosing of sunitinib malate in patients with advanced gastrointestinal stromal tumour after imatinib failure. Eur J Cancer. 2009;45:1959-1968.
18. Brennan MF, Antonescu CR, Maki RG. Management of Soft Tissue Sarcomas. Switzerland: Springer International Publishing; 2013.
19. Heinrich MC, Maki RG, Corless CL, et al. Primary and secondary kinase genotypes correlate with the biological and clinical activity of sunitinib in imatinib-resistant gastrointestinal stromal tumors. J Clin Oncol. 2008;26:5352-5359.
20. Demetri GD, Reichardt P, Kang YK, et al. Efficacy and safety of regorafenib for advanced gastrointestinal stromal tumours after failure of imatinib and sunitinib (GRID): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet. 2013;381:295-302.
21. Wilky BA, Villalobos VM. Emerging role for precision therapy through next-generation sequencing for sarcomas. JCO Precision Oncology. 2018;2:1-4.
22. Drilon A, Laetsch TW, Kummar S, et al. Efficacy of larotrectinib in trk fusion-positive cancers in adults and children. N Engl J Med. 2018;378:731-739.
1. Ma GL, Murphy JD, Martinez ME et al. Epidemiology of gastrointestinal stromal tumors in the era of histology codes: results of a population-based study. Cancer Epidemiol Biomarkers Prev. 2015;24:298-302.
2. Heinrich MC, Rankin C, Blanke CD, et al. Correlation of long-term results of imatinib in advanced gastrointestinal stromal tumors with next-generation sequencing results: analysis of phase 3 SWOG Intergroup Trial S0033. JAMA Oncol. 2017;3:944-952.
3. DeMatteo RP, Lewis JJ, Leung D, et al. Two hundred gastrointestinal stromal tumors recurrence patterns and prognostic factors for survival. Ann Surg. 2000;231:51-58.
4. Goss GA, Merriam P, Manola J, et al. Clinical and pathological characteristics of gastrointestinal stromal tumors (GIST). Prog Proc Am Soc Clin Oncol. 2000;19:599a.
5. Demetri GD, von Mehren M, Blanke CD, et al. Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N Engl J Med. 2002; 347:472-480.
6. Blanke CD, Demetri GD, von Mehren M, et al. Long-term results from a randomized phase ii trial of standard- versus higher-dose imatinib mesylate for patients with unresectable or metastatic gastrointestinal stromal tumors expressing KIT. J Clin Oncol. 2008;26:620-625.
7. Verweij J, van Oosterom A, Blay JY, et al. Imatinib mesylate (STI-571 Glivec, Gleevac) is an active agent for gastrointestinal stromal tumours, but does not yield responses in other soft-tissue sarcomas that are unselected for a molecular target. Results from an EORTC Soft Tissue and Bone Sarcoma Group phase II study. Eur J Cancer. 2003;39:2006-2011.
8. Verweij J, Casali PG, Zalcberg J, et al. Progression-free survival in gastrointestinal stromal tumours with high-dose imatinib: randomized trial. Lancet. 2004;364:1127-1134.
9. Blanke CD, Rankin C, Demetri GD, et al. Phase III randomized, intergroup trial assessing imatinib mesylate at two dose levels in patients with unresectable or metastatic gastrointestinal stromal tumors expressing the kit receptor tyrosine kinase: S0033. J Clin Oncol. 2008;26:626-632.
10. Gastrointestinal Stromal Tumor Meta-Analysis Group (MetaGIST). Comparison of two doses of imatinib for the treatment of unresectable or metastatic gastrointestinal stromal tumors: a meta-analysis of 1,640 patients. J Clin Oncol. 2010;28:1247-1253.
11. Tournigand C, Cervantes A, Figer A, et al. OPTIMOX1: a randomized study of FOLFOX4 or FOLFOX7 with oxaliplatin in a stop-and-Go fashion in advanced colorectal cancer –a GERCOR study. J Clin Oncol. 2006;24:394-400.
12. Blay JV, Cesne AL, Ray-Coquard I, et al. Prospective multicentric randomized phase iii study of imatinib in patients with advanced gastrointestinal stromal tumors comparing interruption versus continuation of treatment beyond 1 year: The French Sarcoma Group. J Clin Oncol. 2007;25:1107-1113.
13. Desai J, Shankar S, Heinrich MC, et al. Clonal evolution of resistance to imatinib in patients with metastatic gastrointestinal stromal tumors. Clin Cancer Res. 2007;13(18 Pt 1): 5398-5405.
14. Gramza AW, Corless CL, Heinrich MC. Resistance to tyrosine kinase inhibitors in gastrointestinal stromal tumors. Clin Cancer Res. 2009;15:7510-7518.
15. Sutent (sunitinib malate) [package insert]. New York, NY: Pfizer Labs; 2017.
16. Demetri GD, van Oosterom AT, Garrett CR, et al. Efficacy and safety of sunitinib in patients with advanced gastrointestinal stromal tumour after failure of imatinib: a randomized controlled trial. Lancet. 2006;368:1329-1338.
17. George S, Blay JY, Casali PG, et al. Clinical evaluation of continuous daily dosing of sunitinib malate in patients with advanced gastrointestinal stromal tumour after imatinib failure. Eur J Cancer. 2009;45:1959-1968.
18. Brennan MF, Antonescu CR, Maki RG. Management of Soft Tissue Sarcomas. Switzerland: Springer International Publishing; 2013.
19. Heinrich MC, Maki RG, Corless CL, et al. Primary and secondary kinase genotypes correlate with the biological and clinical activity of sunitinib in imatinib-resistant gastrointestinal stromal tumors. J Clin Oncol. 2008;26:5352-5359.
20. Demetri GD, Reichardt P, Kang YK, et al. Efficacy and safety of regorafenib for advanced gastrointestinal stromal tumours after failure of imatinib and sunitinib (GRID): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet. 2013;381:295-302.
21. Wilky BA, Villalobos VM. Emerging role for precision therapy through next-generation sequencing for sarcomas. JCO Precision Oncology. 2018;2:1-4.
22. Drilon A, Laetsch TW, Kummar S, et al. Efficacy of larotrectinib in trk fusion-positive cancers in adults and children. N Engl J Med. 2018;378:731-739.
Recurrence of a small gastric gastrointestinal stromal tumor with high mitotic index
Gastrointestinal stromal tumor (GIST) is the most common soft tissue sarcoma of the gastrointestinal tract, usually arising from the interstitial cells of Cajal or similar cells in the outer wall of the gastrointestinal tract.1,2 Most GISTs have an activating mutation in KIT or platelet-derived growth factor receptor alpha (PDGFRα). Tumor size, mitotic rate, and anatomic site are the most common pathological features used to risk stratify GIST tumors.3-10 It is important to note when using such risk calculators that preoperative imatinib before determining tumor characteristics (such as mitoses per 50 high-power fields [hpf]) often changes the relevant parameters so that the same risk calculations may not apply. Tumors with a mitotic rate ≤5 mitoses per 50 hpf and a size ≤5 cm in greatest dimension have a lower recurrence rate after resection than tumors with a mitotic rate >5 mitoses per 50 hpf and a size >10 cm, and larger tumors can have a recurrence rate of up to 86%.11,12 Findings from a large observational study have suggested that the prognosis of gastric GIST in Korea and Japan may be more favorable compared with that in Western countries.13
The primary treatment of a localized primary GIST is surgical excision, but a cure is limited by recurrence.14,15 Imatinib is useful in the treatment of metastatic or recurrent GIST, and adjuvant treatment with imatinib after surgery has been shown to improve progression-free and overall survival in some cases.3,16-18 Responses to adjuvant imatinib depend on tumor sensitivity to the drug and the risk of recurrence. Drug sensitivity is largely dependent on the presence of mutations in KIT or PDGFRα.3,18 Recurrence risk is highly dependent on tumor size, tumor site, tumor rupture, and mitotic index.1,3,5,6,8,9,18,19 Findings on the use of gene expression patterns to predict recurrence risk have also been reported.20-27 However, recurrence risk is poorly understood for categories in which there are few cases with known outcomes, such as very small gastric GIST with a high mitotic index. For example, few cases of gastric GIST have been reported with a tumor size ≤2 cm, a mitotic rate >5 mitoses per 50 hpf, and adequate clinical follow-up. In such cases, it is difficult to assess the risk of recurrence.6 We report here the long-term outcome of a patient with a 1.8-cm gastric GIST with a mitotic index of 36 mitoses per 50 hpf and a KIT exon 11 mutation.
Case Presentation and Summary
A 69-year-old man presented with periumbilical and epigastric pain of 6-month duration. His medical history was notable for hyperlipidemia, hypertension, coronary angioplasty, and spinal surgery. He had a 40 pack-year smoking history and consumed 2 to 4 alcoholic drinks per day. The results of a physical examination were unremarkable. A computed tomographic (CT) scan showed no abnormalities. An esophagogastroduodenoscopy (EGD) revealed gastric ulcers. He was treated successfully with omeprazole 20 mg by mouth daily.
A month later, a follow-up EGD revealed a 1.8 x 1.5-cm submucosal mass 3 cm from the gastroesophageal junction. The patient underwent a fundus wedge resection, and a submucosal mass 1.8 cm in greatest dimension was removed. Pathologic examination revealed a GIST, spindle cell type, with a mitotic rate of 36 mitoses per 50 hpf with negative margins. Immunohistochemistry was positive for CD117. An exon 11 deletion (KVV558-560NV) was present in KIT. The patient’s risk of recurrence was unclear, and his follow-up included CT scans of the abdomen and pelvis every 3 to 4 months for the first 2 years, then every 6 months for the next 2.5 years.
A CT scan about 3.5 years after primary resection revealed small nonspecific liver hypodensities that became more prominent during the next year. About 5 years after primary resection, magnetic resonance imaging (MRI) revealed several liver lesions, the largest of which measured 1.3 cm in greatest dimension. The patient’s liver metastases were readily identified by MRI (Figure 1) and CT imaging (Figure 2A).
Discussion
Small gastric GISTs are sometimes found by endoscopy performed for unrelated reasons. Recent data suggest that the incidence of gastric GIST may be higher than previously thought. In a Japanese study of patients with gastric cancer in which 100 stomachs were systematically examined pathologically, 50 microscopic GISTs were found in 35 patients.28 Most small gastric GISTs have a low mitotic index. Few cases have been described with a high mitotic index. In a study of 1765 cases of GIST of the stomach, 8 patients had a tumor size less than 2 cm and a mitotic index greater than 5. Of those, only 6 patients had long-term follow-up, and 3 were alive without disease at 2, 17, and 20 years of follow-up.7 These limited data make it impossible to predict outcomes in patients with small gastric GIST with a high mitotic index.
For patients who are at high risk of recurrence after surgery, 3 years of adjuvant imatinib treatment compared with 1 year has been shown to improve overall survival and is the current standard of care.10,17 A study comparing 5 and 3 years of imatinib is ongoing to establish whether a longer period of adjuvant treatment is warranted. In patients with metastatic GIST, lifelong imatinib until lack of benefit is considered optimal treatment.10 All patients should undergo KIT mutation analysis. Those with the PDGFRα D842V mutation, SDH (succinate dehydrogenase) deficiency, or neurofibromatosis-related GIST should not receive adjuvant imatinib.
This case has several unusual features. The small tumor size with a very high mitotic rate is rare. Such cases have not been reported in large numbers and have therefore not been reliably incorporated into risk prediction algorithms. In addition, despite a high mitotic index, the tumor was not FDG avid on PET imaging. The diagnosis of GIST is strongly supported by the KIT mutation and response to imatinib. This particular KIT mutation in larger GISTs is associated with aggressive disease. The present case adds to the data on the biology of small gastric GISTs with a high mitotic index and suggests the mitotic index in these tumors may be a more important predictor than size. TSJ
Acknowlegement
The authors thank Michael Franklin, MS, for editorial assistance, and Sabrina Porter for media edits.
aDepartment of Medicine, University of Minnesota Medical School; bDepartment of Laboratory Medicine and Pathology, University of Minnesota Medical School; and cMasonic Cancer Center, University of Minnesota Medical School, Minneapolis, Minnesota.
Disclosures
The authors report no disclosures or conflicts of interest. This article was originally published in The Journal of Community and Supportive Oncology JCSO. 2018;16(3):e163-e166. ©Frontline Medical Communications. doi:10.12788/jcso.0402. It is reproduced with permission from the copyright owner. Further reproduction prohibited without permission.
1. Corless CL, Barnett CM, Heinrich MC. Gastrointestinal stromal tumours: origin and molecular oncology. Nat Rev Cancer. 2011;11(12):865-878.
2. Hirota S, Isozaki K, Moriyama Y, Hashimoto K, Nishida T, Ishiguro S, et al. Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science. 1998;279(5350):577-580.
3. Corless CL, Ballman KV, Antonescu CR, Kolesnikova V, Maki RG, Pisters PW, et al. Pathologic and molecular features correlate with long-term outcome after adjuvant therapy of resected primary GI stromal tumor: the ACOSOG Z9001 trial. J Clin Oncol. 2014;32(15):1563-1570.
4. Huang J, Zheng DL, Qin FS, Cheng N, Chen H, Wan BB, et al. Genetic and epigenetic silencing of SCARA5 may contribute to human hepatocellular carcinoma by activating FAK signaling. J Clin Invest. 2010;120(1):223-241.
5. Joensuu H, Vehtari A, Riihimaki J, Nishida T, Steigen SE, Brabec P, et al. Risk of recurrence of gastrointestinal stromal tumour after surgery: an analysis of pooled population-based cohorts. Lancet Oncol. 2012;13(3):265-274.
6. Miettinen M, Lasota J. Gastrointestinal stromal tumors: review on morphology, molecular pathology, prognosis, and differential diagnosis. Arch Pathol Lab Med. 2006;130(10):1466-1478.
7. Miettinen M, Sobin LH, Lasota J. Gastrointestinal stromal tumors of the stomach: a clinicopathologic, immunohistochemical, and molecular genetic study of 1765 cases with long-term follow-up. Am J Surg Pathol. 2005;29(1):52-68.
8. Patel S. Navigating risk stratification systems for the management of patients with GIST. Ann Surg Oncol. 2011;18(6):1698-1704.
9. Rossi S, Miceli R, Messerini L, Bearzi I, Mazzoleni G, Capella C, et al. Natural history of imatinib-naive GISTs: a retrospective analysis of 929 cases with long-term follow-up and development of a survival nomogram based on mitotic index and size as continuous variables. Am J Surg Pathol. 2011;35(11):1646-1656.
10. National Comprehensive Cancer Network. Sarcoma. https://www.nccn.org/professionals/physician_gls/default.aspx#age. Accessed March 27, 2018.
11. Fletcher CD, Berman JJ, Corless C, Gorstein F, Lasota J, Longley BJ, et al. Diagnosis of gastrointestinal stromal tumors: a consensus approach. Int J Surg Pathol. 2002;10(2):81-89.
12. Huang HY, Li CF, Huang WW, Hu TH, Lin CN, Uen YH, et al. A modification of NIH consensus criteria to better distinguish the highly lethal subset of primary localized gastrointestinal stromal tumors: a subdivision of the original high-risk group on the basis of outcome. Surgery. 2007;141(6):748-756.
13. Kim MC, Yook JH, Yang HK, Lee HJ, Sohn TS, Hyung WJ, et al. Long-term surgical outcome of 1057 gastric GISTs according to 7th UICC/AJCC TNM system: multicenter observational study from Korea and Japan. Medicine (Baltimore). 2015;94(41):e1526.
14. Casali PG, Blay JY; ESMO/CONTICANET/EUROBONET Consensus Panel of experts. Soft tissue sarcomas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2010;21(Suppl 5):v198-v203.
15. Joensuu H, DeMatteo RP. The management of gastrointestinal stromal tumors: a model for targeted and multidisciplinary therapy of malignancy. Annu Rev Med. 2012;63:247-258.
16. Dematteo RP, Ballman KV, Antonescu CR, Maki RG, Pisters PW, Demetri GD, et al. Adjuvant imatinib mesylate after resection of localised, primary gastrointestinal stromal tumour: a randomised, double-blind, placebo-controlled trial. Lancet. 2009;373(9669):1097-1104.
17. Joensuu H, Eriksson M, Sundby Hall K, Hartmann JT, Pink D, Schütte J, et al. One vs three years of adjuvant imatinib for operable gastrointestinal stromal tumor: a randomized trial. JAMA. 2012;307(12):1265-1272.
18. Joensuu H, Rutkowski P, Nishida T, Steigen SE, Brabec P, Plank L, et al. KIT and PDGFRA mutations and the risk of GI stromal tumor recurrence. J Clin Oncol. 2015;33(6):634-642.
19. Fletcher CD, Berman JJ, Corless C, Gorstein F, Lasota J, Longley BJ, et al. Diagnosis of gastrointestinal stromal tumors: A consensus approach. Hum Pathol. 2002;33(5):459-465.
20. Antonescu CR, Viale A, Sarran L, Tschernyavsky SJ, Gonen M, Segal NH, et al. Gene expression in gastrointestinal stromal tumors is distinguished by KIT genotype and anatomic site. Clin Cancer Res. 2004;10(10):3282-3290.
21. Arne G, Kristiansson E, Nerman O, Kindblom LG, Ahlman H, Nilsson B, et al. Expression profiling of GIST: CD133 is associated with KIT exon 11 mutations, gastric location and poor prognosis. Int J Cancer. 2011;129(5):1149-1161.
22. Bertucci F, Finetti P, Ostrowski J, Kim WK, Kim H, Pantaleo MA, et al. Genomic Grade Index predicts postoperative clinical outcome of GIST. Br J Cancer. 2012;107(8):1433-1441.
23. Koon N, Schneider-Stock R, Sarlomo-Rikala M, Lasota J, Smolkin M, Petroni G, et al. Molecular targets for tumour progression in gastrointestinal stromal tumours. Gut. 2004;53(2):235-240.
24. Lagarde P, Perot G, Kauffmann A, Brulard C, Dapremont V, Hostein I, et al. Mitotic checkpoints and chromosome instability are strong predictors of clinical outcome in gastrointestinal stromal tumors. Clin Cancer Res. 2012;18(3):826-838.
25. Skubitz KM, Geschwind K, Xu WW, Koopmeiners JS, Skubitz AP. Gene expression identifies heterogeneity of metastatic behavior among gastrointestinal stromal tumors. J Transl Med. 2016;14:51.
26. Yamaguchi U, Nakayama R, Honda K, Ichikawa H, Haseqawa T, Shitashige M, et al. Distinct gene expression-defined classes of gastrointestinal stromal tumor. J Clin Oncol. 2008;26(25):4100-4108.
27. Ylipaa A, Hunt KK, Yang J, Lazar AJ, Torres KE, Lev DC, et al. Integrative genomic characterization and a genomic staging system for gastrointestinal stromal tumors. Cancer. 2011;117(2):380-389.
28. Kawanowa K, Sakuma Y, Sakurai S, Hishima T, Iwasaki Y, Saito K, et al. High incidence of microscopic gastrointestinal stromal tumors in the stomach. Hum Pathol. 2006;37(12):1527-1535.
Gastrointestinal stromal tumor (GIST) is the most common soft tissue sarcoma of the gastrointestinal tract, usually arising from the interstitial cells of Cajal or similar cells in the outer wall of the gastrointestinal tract.1,2 Most GISTs have an activating mutation in KIT or platelet-derived growth factor receptor alpha (PDGFRα). Tumor size, mitotic rate, and anatomic site are the most common pathological features used to risk stratify GIST tumors.3-10 It is important to note when using such risk calculators that preoperative imatinib before determining tumor characteristics (such as mitoses per 50 high-power fields [hpf]) often changes the relevant parameters so that the same risk calculations may not apply. Tumors with a mitotic rate ≤5 mitoses per 50 hpf and a size ≤5 cm in greatest dimension have a lower recurrence rate after resection than tumors with a mitotic rate >5 mitoses per 50 hpf and a size >10 cm, and larger tumors can have a recurrence rate of up to 86%.11,12 Findings from a large observational study have suggested that the prognosis of gastric GIST in Korea and Japan may be more favorable compared with that in Western countries.13
The primary treatment of a localized primary GIST is surgical excision, but a cure is limited by recurrence.14,15 Imatinib is useful in the treatment of metastatic or recurrent GIST, and adjuvant treatment with imatinib after surgery has been shown to improve progression-free and overall survival in some cases.3,16-18 Responses to adjuvant imatinib depend on tumor sensitivity to the drug and the risk of recurrence. Drug sensitivity is largely dependent on the presence of mutations in KIT or PDGFRα.3,18 Recurrence risk is highly dependent on tumor size, tumor site, tumor rupture, and mitotic index.1,3,5,6,8,9,18,19 Findings on the use of gene expression patterns to predict recurrence risk have also been reported.20-27 However, recurrence risk is poorly understood for categories in which there are few cases with known outcomes, such as very small gastric GIST with a high mitotic index. For example, few cases of gastric GIST have been reported with a tumor size ≤2 cm, a mitotic rate >5 mitoses per 50 hpf, and adequate clinical follow-up. In such cases, it is difficult to assess the risk of recurrence.6 We report here the long-term outcome of a patient with a 1.8-cm gastric GIST with a mitotic index of 36 mitoses per 50 hpf and a KIT exon 11 mutation.
Case Presentation and Summary
A 69-year-old man presented with periumbilical and epigastric pain of 6-month duration. His medical history was notable for hyperlipidemia, hypertension, coronary angioplasty, and spinal surgery. He had a 40 pack-year smoking history and consumed 2 to 4 alcoholic drinks per day. The results of a physical examination were unremarkable. A computed tomographic (CT) scan showed no abnormalities. An esophagogastroduodenoscopy (EGD) revealed gastric ulcers. He was treated successfully with omeprazole 20 mg by mouth daily.
A month later, a follow-up EGD revealed a 1.8 x 1.5-cm submucosal mass 3 cm from the gastroesophageal junction. The patient underwent a fundus wedge resection, and a submucosal mass 1.8 cm in greatest dimension was removed. Pathologic examination revealed a GIST, spindle cell type, with a mitotic rate of 36 mitoses per 50 hpf with negative margins. Immunohistochemistry was positive for CD117. An exon 11 deletion (KVV558-560NV) was present in KIT. The patient’s risk of recurrence was unclear, and his follow-up included CT scans of the abdomen and pelvis every 3 to 4 months for the first 2 years, then every 6 months for the next 2.5 years.
A CT scan about 3.5 years after primary resection revealed small nonspecific liver hypodensities that became more prominent during the next year. About 5 years after primary resection, magnetic resonance imaging (MRI) revealed several liver lesions, the largest of which measured 1.3 cm in greatest dimension. The patient’s liver metastases were readily identified by MRI (Figure 1) and CT imaging (Figure 2A).
Discussion
Small gastric GISTs are sometimes found by endoscopy performed for unrelated reasons. Recent data suggest that the incidence of gastric GIST may be higher than previously thought. In a Japanese study of patients with gastric cancer in which 100 stomachs were systematically examined pathologically, 50 microscopic GISTs were found in 35 patients.28 Most small gastric GISTs have a low mitotic index. Few cases have been described with a high mitotic index. In a study of 1765 cases of GIST of the stomach, 8 patients had a tumor size less than 2 cm and a mitotic index greater than 5. Of those, only 6 patients had long-term follow-up, and 3 were alive without disease at 2, 17, and 20 years of follow-up.7 These limited data make it impossible to predict outcomes in patients with small gastric GIST with a high mitotic index.
For patients who are at high risk of recurrence after surgery, 3 years of adjuvant imatinib treatment compared with 1 year has been shown to improve overall survival and is the current standard of care.10,17 A study comparing 5 and 3 years of imatinib is ongoing to establish whether a longer period of adjuvant treatment is warranted. In patients with metastatic GIST, lifelong imatinib until lack of benefit is considered optimal treatment.10 All patients should undergo KIT mutation analysis. Those with the PDGFRα D842V mutation, SDH (succinate dehydrogenase) deficiency, or neurofibromatosis-related GIST should not receive adjuvant imatinib.
This case has several unusual features. The small tumor size with a very high mitotic rate is rare. Such cases have not been reported in large numbers and have therefore not been reliably incorporated into risk prediction algorithms. In addition, despite a high mitotic index, the tumor was not FDG avid on PET imaging. The diagnosis of GIST is strongly supported by the KIT mutation and response to imatinib. This particular KIT mutation in larger GISTs is associated with aggressive disease. The present case adds to the data on the biology of small gastric GISTs with a high mitotic index and suggests the mitotic index in these tumors may be a more important predictor than size. TSJ
Acknowlegement
The authors thank Michael Franklin, MS, for editorial assistance, and Sabrina Porter for media edits.
aDepartment of Medicine, University of Minnesota Medical School; bDepartment of Laboratory Medicine and Pathology, University of Minnesota Medical School; and cMasonic Cancer Center, University of Minnesota Medical School, Minneapolis, Minnesota.
Disclosures
The authors report no disclosures or conflicts of interest. This article was originally published in The Journal of Community and Supportive Oncology JCSO. 2018;16(3):e163-e166. ©Frontline Medical Communications. doi:10.12788/jcso.0402. It is reproduced with permission from the copyright owner. Further reproduction prohibited without permission.
Gastrointestinal stromal tumor (GIST) is the most common soft tissue sarcoma of the gastrointestinal tract, usually arising from the interstitial cells of Cajal or similar cells in the outer wall of the gastrointestinal tract.1,2 Most GISTs have an activating mutation in KIT or platelet-derived growth factor receptor alpha (PDGFRα). Tumor size, mitotic rate, and anatomic site are the most common pathological features used to risk stratify GIST tumors.3-10 It is important to note when using such risk calculators that preoperative imatinib before determining tumor characteristics (such as mitoses per 50 high-power fields [hpf]) often changes the relevant parameters so that the same risk calculations may not apply. Tumors with a mitotic rate ≤5 mitoses per 50 hpf and a size ≤5 cm in greatest dimension have a lower recurrence rate after resection than tumors with a mitotic rate >5 mitoses per 50 hpf and a size >10 cm, and larger tumors can have a recurrence rate of up to 86%.11,12 Findings from a large observational study have suggested that the prognosis of gastric GIST in Korea and Japan may be more favorable compared with that in Western countries.13
The primary treatment of a localized primary GIST is surgical excision, but a cure is limited by recurrence.14,15 Imatinib is useful in the treatment of metastatic or recurrent GIST, and adjuvant treatment with imatinib after surgery has been shown to improve progression-free and overall survival in some cases.3,16-18 Responses to adjuvant imatinib depend on tumor sensitivity to the drug and the risk of recurrence. Drug sensitivity is largely dependent on the presence of mutations in KIT or PDGFRα.3,18 Recurrence risk is highly dependent on tumor size, tumor site, tumor rupture, and mitotic index.1,3,5,6,8,9,18,19 Findings on the use of gene expression patterns to predict recurrence risk have also been reported.20-27 However, recurrence risk is poorly understood for categories in which there are few cases with known outcomes, such as very small gastric GIST with a high mitotic index. For example, few cases of gastric GIST have been reported with a tumor size ≤2 cm, a mitotic rate >5 mitoses per 50 hpf, and adequate clinical follow-up. In such cases, it is difficult to assess the risk of recurrence.6 We report here the long-term outcome of a patient with a 1.8-cm gastric GIST with a mitotic index of 36 mitoses per 50 hpf and a KIT exon 11 mutation.
Case Presentation and Summary
A 69-year-old man presented with periumbilical and epigastric pain of 6-month duration. His medical history was notable for hyperlipidemia, hypertension, coronary angioplasty, and spinal surgery. He had a 40 pack-year smoking history and consumed 2 to 4 alcoholic drinks per day. The results of a physical examination were unremarkable. A computed tomographic (CT) scan showed no abnormalities. An esophagogastroduodenoscopy (EGD) revealed gastric ulcers. He was treated successfully with omeprazole 20 mg by mouth daily.
A month later, a follow-up EGD revealed a 1.8 x 1.5-cm submucosal mass 3 cm from the gastroesophageal junction. The patient underwent a fundus wedge resection, and a submucosal mass 1.8 cm in greatest dimension was removed. Pathologic examination revealed a GIST, spindle cell type, with a mitotic rate of 36 mitoses per 50 hpf with negative margins. Immunohistochemistry was positive for CD117. An exon 11 deletion (KVV558-560NV) was present in KIT. The patient’s risk of recurrence was unclear, and his follow-up included CT scans of the abdomen and pelvis every 3 to 4 months for the first 2 years, then every 6 months for the next 2.5 years.
A CT scan about 3.5 years after primary resection revealed small nonspecific liver hypodensities that became more prominent during the next year. About 5 years after primary resection, magnetic resonance imaging (MRI) revealed several liver lesions, the largest of which measured 1.3 cm in greatest dimension. The patient’s liver metastases were readily identified by MRI (Figure 1) and CT imaging (Figure 2A).
Discussion
Small gastric GISTs are sometimes found by endoscopy performed for unrelated reasons. Recent data suggest that the incidence of gastric GIST may be higher than previously thought. In a Japanese study of patients with gastric cancer in which 100 stomachs were systematically examined pathologically, 50 microscopic GISTs were found in 35 patients.28 Most small gastric GISTs have a low mitotic index. Few cases have been described with a high mitotic index. In a study of 1765 cases of GIST of the stomach, 8 patients had a tumor size less than 2 cm and a mitotic index greater than 5. Of those, only 6 patients had long-term follow-up, and 3 were alive without disease at 2, 17, and 20 years of follow-up.7 These limited data make it impossible to predict outcomes in patients with small gastric GIST with a high mitotic index.
For patients who are at high risk of recurrence after surgery, 3 years of adjuvant imatinib treatment compared with 1 year has been shown to improve overall survival and is the current standard of care.10,17 A study comparing 5 and 3 years of imatinib is ongoing to establish whether a longer period of adjuvant treatment is warranted. In patients with metastatic GIST, lifelong imatinib until lack of benefit is considered optimal treatment.10 All patients should undergo KIT mutation analysis. Those with the PDGFRα D842V mutation, SDH (succinate dehydrogenase) deficiency, or neurofibromatosis-related GIST should not receive adjuvant imatinib.
This case has several unusual features. The small tumor size with a very high mitotic rate is rare. Such cases have not been reported in large numbers and have therefore not been reliably incorporated into risk prediction algorithms. In addition, despite a high mitotic index, the tumor was not FDG avid on PET imaging. The diagnosis of GIST is strongly supported by the KIT mutation and response to imatinib. This particular KIT mutation in larger GISTs is associated with aggressive disease. The present case adds to the data on the biology of small gastric GISTs with a high mitotic index and suggests the mitotic index in these tumors may be a more important predictor than size. TSJ
Acknowlegement
The authors thank Michael Franklin, MS, for editorial assistance, and Sabrina Porter for media edits.
aDepartment of Medicine, University of Minnesota Medical School; bDepartment of Laboratory Medicine and Pathology, University of Minnesota Medical School; and cMasonic Cancer Center, University of Minnesota Medical School, Minneapolis, Minnesota.
Disclosures
The authors report no disclosures or conflicts of interest. This article was originally published in The Journal of Community and Supportive Oncology JCSO. 2018;16(3):e163-e166. ©Frontline Medical Communications. doi:10.12788/jcso.0402. It is reproduced with permission from the copyright owner. Further reproduction prohibited without permission.
1. Corless CL, Barnett CM, Heinrich MC. Gastrointestinal stromal tumours: origin and molecular oncology. Nat Rev Cancer. 2011;11(12):865-878.
2. Hirota S, Isozaki K, Moriyama Y, Hashimoto K, Nishida T, Ishiguro S, et al. Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science. 1998;279(5350):577-580.
3. Corless CL, Ballman KV, Antonescu CR, Kolesnikova V, Maki RG, Pisters PW, et al. Pathologic and molecular features correlate with long-term outcome after adjuvant therapy of resected primary GI stromal tumor: the ACOSOG Z9001 trial. J Clin Oncol. 2014;32(15):1563-1570.
4. Huang J, Zheng DL, Qin FS, Cheng N, Chen H, Wan BB, et al. Genetic and epigenetic silencing of SCARA5 may contribute to human hepatocellular carcinoma by activating FAK signaling. J Clin Invest. 2010;120(1):223-241.
5. Joensuu H, Vehtari A, Riihimaki J, Nishida T, Steigen SE, Brabec P, et al. Risk of recurrence of gastrointestinal stromal tumour after surgery: an analysis of pooled population-based cohorts. Lancet Oncol. 2012;13(3):265-274.
6. Miettinen M, Lasota J. Gastrointestinal stromal tumors: review on morphology, molecular pathology, prognosis, and differential diagnosis. Arch Pathol Lab Med. 2006;130(10):1466-1478.
7. Miettinen M, Sobin LH, Lasota J. Gastrointestinal stromal tumors of the stomach: a clinicopathologic, immunohistochemical, and molecular genetic study of 1765 cases with long-term follow-up. Am J Surg Pathol. 2005;29(1):52-68.
8. Patel S. Navigating risk stratification systems for the management of patients with GIST. Ann Surg Oncol. 2011;18(6):1698-1704.
9. Rossi S, Miceli R, Messerini L, Bearzi I, Mazzoleni G, Capella C, et al. Natural history of imatinib-naive GISTs: a retrospective analysis of 929 cases with long-term follow-up and development of a survival nomogram based on mitotic index and size as continuous variables. Am J Surg Pathol. 2011;35(11):1646-1656.
10. National Comprehensive Cancer Network. Sarcoma. https://www.nccn.org/professionals/physician_gls/default.aspx#age. Accessed March 27, 2018.
11. Fletcher CD, Berman JJ, Corless C, Gorstein F, Lasota J, Longley BJ, et al. Diagnosis of gastrointestinal stromal tumors: a consensus approach. Int J Surg Pathol. 2002;10(2):81-89.
12. Huang HY, Li CF, Huang WW, Hu TH, Lin CN, Uen YH, et al. A modification of NIH consensus criteria to better distinguish the highly lethal subset of primary localized gastrointestinal stromal tumors: a subdivision of the original high-risk group on the basis of outcome. Surgery. 2007;141(6):748-756.
13. Kim MC, Yook JH, Yang HK, Lee HJ, Sohn TS, Hyung WJ, et al. Long-term surgical outcome of 1057 gastric GISTs according to 7th UICC/AJCC TNM system: multicenter observational study from Korea and Japan. Medicine (Baltimore). 2015;94(41):e1526.
14. Casali PG, Blay JY; ESMO/CONTICANET/EUROBONET Consensus Panel of experts. Soft tissue sarcomas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2010;21(Suppl 5):v198-v203.
15. Joensuu H, DeMatteo RP. The management of gastrointestinal stromal tumors: a model for targeted and multidisciplinary therapy of malignancy. Annu Rev Med. 2012;63:247-258.
16. Dematteo RP, Ballman KV, Antonescu CR, Maki RG, Pisters PW, Demetri GD, et al. Adjuvant imatinib mesylate after resection of localised, primary gastrointestinal stromal tumour: a randomised, double-blind, placebo-controlled trial. Lancet. 2009;373(9669):1097-1104.
17. Joensuu H, Eriksson M, Sundby Hall K, Hartmann JT, Pink D, Schütte J, et al. One vs three years of adjuvant imatinib for operable gastrointestinal stromal tumor: a randomized trial. JAMA. 2012;307(12):1265-1272.
18. Joensuu H, Rutkowski P, Nishida T, Steigen SE, Brabec P, Plank L, et al. KIT and PDGFRA mutations and the risk of GI stromal tumor recurrence. J Clin Oncol. 2015;33(6):634-642.
19. Fletcher CD, Berman JJ, Corless C, Gorstein F, Lasota J, Longley BJ, et al. Diagnosis of gastrointestinal stromal tumors: A consensus approach. Hum Pathol. 2002;33(5):459-465.
20. Antonescu CR, Viale A, Sarran L, Tschernyavsky SJ, Gonen M, Segal NH, et al. Gene expression in gastrointestinal stromal tumors is distinguished by KIT genotype and anatomic site. Clin Cancer Res. 2004;10(10):3282-3290.
21. Arne G, Kristiansson E, Nerman O, Kindblom LG, Ahlman H, Nilsson B, et al. Expression profiling of GIST: CD133 is associated with KIT exon 11 mutations, gastric location and poor prognosis. Int J Cancer. 2011;129(5):1149-1161.
22. Bertucci F, Finetti P, Ostrowski J, Kim WK, Kim H, Pantaleo MA, et al. Genomic Grade Index predicts postoperative clinical outcome of GIST. Br J Cancer. 2012;107(8):1433-1441.
23. Koon N, Schneider-Stock R, Sarlomo-Rikala M, Lasota J, Smolkin M, Petroni G, et al. Molecular targets for tumour progression in gastrointestinal stromal tumours. Gut. 2004;53(2):235-240.
24. Lagarde P, Perot G, Kauffmann A, Brulard C, Dapremont V, Hostein I, et al. Mitotic checkpoints and chromosome instability are strong predictors of clinical outcome in gastrointestinal stromal tumors. Clin Cancer Res. 2012;18(3):826-838.
25. Skubitz KM, Geschwind K, Xu WW, Koopmeiners JS, Skubitz AP. Gene expression identifies heterogeneity of metastatic behavior among gastrointestinal stromal tumors. J Transl Med. 2016;14:51.
26. Yamaguchi U, Nakayama R, Honda K, Ichikawa H, Haseqawa T, Shitashige M, et al. Distinct gene expression-defined classes of gastrointestinal stromal tumor. J Clin Oncol. 2008;26(25):4100-4108.
27. Ylipaa A, Hunt KK, Yang J, Lazar AJ, Torres KE, Lev DC, et al. Integrative genomic characterization and a genomic staging system for gastrointestinal stromal tumors. Cancer. 2011;117(2):380-389.
28. Kawanowa K, Sakuma Y, Sakurai S, Hishima T, Iwasaki Y, Saito K, et al. High incidence of microscopic gastrointestinal stromal tumors in the stomach. Hum Pathol. 2006;37(12):1527-1535.
1. Corless CL, Barnett CM, Heinrich MC. Gastrointestinal stromal tumours: origin and molecular oncology. Nat Rev Cancer. 2011;11(12):865-878.
2. Hirota S, Isozaki K, Moriyama Y, Hashimoto K, Nishida T, Ishiguro S, et al. Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science. 1998;279(5350):577-580.
3. Corless CL, Ballman KV, Antonescu CR, Kolesnikova V, Maki RG, Pisters PW, et al. Pathologic and molecular features correlate with long-term outcome after adjuvant therapy of resected primary GI stromal tumor: the ACOSOG Z9001 trial. J Clin Oncol. 2014;32(15):1563-1570.
4. Huang J, Zheng DL, Qin FS, Cheng N, Chen H, Wan BB, et al. Genetic and epigenetic silencing of SCARA5 may contribute to human hepatocellular carcinoma by activating FAK signaling. J Clin Invest. 2010;120(1):223-241.
5. Joensuu H, Vehtari A, Riihimaki J, Nishida T, Steigen SE, Brabec P, et al. Risk of recurrence of gastrointestinal stromal tumour after surgery: an analysis of pooled population-based cohorts. Lancet Oncol. 2012;13(3):265-274.
6. Miettinen M, Lasota J. Gastrointestinal stromal tumors: review on morphology, molecular pathology, prognosis, and differential diagnosis. Arch Pathol Lab Med. 2006;130(10):1466-1478.
7. Miettinen M, Sobin LH, Lasota J. Gastrointestinal stromal tumors of the stomach: a clinicopathologic, immunohistochemical, and molecular genetic study of 1765 cases with long-term follow-up. Am J Surg Pathol. 2005;29(1):52-68.
8. Patel S. Navigating risk stratification systems for the management of patients with GIST. Ann Surg Oncol. 2011;18(6):1698-1704.
9. Rossi S, Miceli R, Messerini L, Bearzi I, Mazzoleni G, Capella C, et al. Natural history of imatinib-naive GISTs: a retrospective analysis of 929 cases with long-term follow-up and development of a survival nomogram based on mitotic index and size as continuous variables. Am J Surg Pathol. 2011;35(11):1646-1656.
10. National Comprehensive Cancer Network. Sarcoma. https://www.nccn.org/professionals/physician_gls/default.aspx#age. Accessed March 27, 2018.
11. Fletcher CD, Berman JJ, Corless C, Gorstein F, Lasota J, Longley BJ, et al. Diagnosis of gastrointestinal stromal tumors: a consensus approach. Int J Surg Pathol. 2002;10(2):81-89.
12. Huang HY, Li CF, Huang WW, Hu TH, Lin CN, Uen YH, et al. A modification of NIH consensus criteria to better distinguish the highly lethal subset of primary localized gastrointestinal stromal tumors: a subdivision of the original high-risk group on the basis of outcome. Surgery. 2007;141(6):748-756.
13. Kim MC, Yook JH, Yang HK, Lee HJ, Sohn TS, Hyung WJ, et al. Long-term surgical outcome of 1057 gastric GISTs according to 7th UICC/AJCC TNM system: multicenter observational study from Korea and Japan. Medicine (Baltimore). 2015;94(41):e1526.
14. Casali PG, Blay JY; ESMO/CONTICANET/EUROBONET Consensus Panel of experts. Soft tissue sarcomas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2010;21(Suppl 5):v198-v203.
15. Joensuu H, DeMatteo RP. The management of gastrointestinal stromal tumors: a model for targeted and multidisciplinary therapy of malignancy. Annu Rev Med. 2012;63:247-258.
16. Dematteo RP, Ballman KV, Antonescu CR, Maki RG, Pisters PW, Demetri GD, et al. Adjuvant imatinib mesylate after resection of localised, primary gastrointestinal stromal tumour: a randomised, double-blind, placebo-controlled trial. Lancet. 2009;373(9669):1097-1104.
17. Joensuu H, Eriksson M, Sundby Hall K, Hartmann JT, Pink D, Schütte J, et al. One vs three years of adjuvant imatinib for operable gastrointestinal stromal tumor: a randomized trial. JAMA. 2012;307(12):1265-1272.
18. Joensuu H, Rutkowski P, Nishida T, Steigen SE, Brabec P, Plank L, et al. KIT and PDGFRA mutations and the risk of GI stromal tumor recurrence. J Clin Oncol. 2015;33(6):634-642.
19. Fletcher CD, Berman JJ, Corless C, Gorstein F, Lasota J, Longley BJ, et al. Diagnosis of gastrointestinal stromal tumors: A consensus approach. Hum Pathol. 2002;33(5):459-465.
20. Antonescu CR, Viale A, Sarran L, Tschernyavsky SJ, Gonen M, Segal NH, et al. Gene expression in gastrointestinal stromal tumors is distinguished by KIT genotype and anatomic site. Clin Cancer Res. 2004;10(10):3282-3290.
21. Arne G, Kristiansson E, Nerman O, Kindblom LG, Ahlman H, Nilsson B, et al. Expression profiling of GIST: CD133 is associated with KIT exon 11 mutations, gastric location and poor prognosis. Int J Cancer. 2011;129(5):1149-1161.
22. Bertucci F, Finetti P, Ostrowski J, Kim WK, Kim H, Pantaleo MA, et al. Genomic Grade Index predicts postoperative clinical outcome of GIST. Br J Cancer. 2012;107(8):1433-1441.
23. Koon N, Schneider-Stock R, Sarlomo-Rikala M, Lasota J, Smolkin M, Petroni G, et al. Molecular targets for tumour progression in gastrointestinal stromal tumours. Gut. 2004;53(2):235-240.
24. Lagarde P, Perot G, Kauffmann A, Brulard C, Dapremont V, Hostein I, et al. Mitotic checkpoints and chromosome instability are strong predictors of clinical outcome in gastrointestinal stromal tumors. Clin Cancer Res. 2012;18(3):826-838.
25. Skubitz KM, Geschwind K, Xu WW, Koopmeiners JS, Skubitz AP. Gene expression identifies heterogeneity of metastatic behavior among gastrointestinal stromal tumors. J Transl Med. 2016;14:51.
26. Yamaguchi U, Nakayama R, Honda K, Ichikawa H, Haseqawa T, Shitashige M, et al. Distinct gene expression-defined classes of gastrointestinal stromal tumor. J Clin Oncol. 2008;26(25):4100-4108.
27. Ylipaa A, Hunt KK, Yang J, Lazar AJ, Torres KE, Lev DC, et al. Integrative genomic characterization and a genomic staging system for gastrointestinal stromal tumors. Cancer. 2011;117(2):380-389.
28. Kawanowa K, Sakuma Y, Sakurai S, Hishima T, Iwasaki Y, Saito K, et al. High incidence of microscopic gastrointestinal stromal tumors in the stomach. Hum Pathol. 2006;37(12):1527-1535.
Rapid drug alteration a bust in metastatic GIST
CHICAGO – For patients with gastrointestinal stromal tumor (GIST) with KIT mutations conferring resistance to imatinib, a strategy of rapid alteration of drugs with complementary activity against KIT mutations is feasible but has thus far failed to yield significant clinical benefits, investigators said.
There were no objective responses among 12 patients treated continuously with 3 days of sunitinib (Sutent) followed by 4 days of regorafenib (Stivarga), and although 4 patients had stable disease in the short term, in each case the disease progressed within 16 weeks, reported Cesar Serrano-Garcia, MD, from the Dana-Farber Cancer Institute and Brigham and Women’s Hospital in Boston, and his colleagues.
“Drug exposure is critical to effectively target specific resistant subpopulations and low exposure may have contributed to the lack of efficacy in this cohort,” they wrote in a poster presented at the annual meeting of the American Society of Clinical Oncology.
The investigators noted that the main mechanism of resistance to imatinib (Gleevec) in GIST is polyclonal emergence of KIT secondary mutations. They then theorized that rapid alteration of sunitinib with regorafenib, which both have complementary activity against different KIT resistance mutations, could be a novel therapeutic strategy for controlling imatinib-resistant disease.
Both agents are active against KIT and platelet-derived growth factor receptor alpha (PDGFR-alpha). Sunitinib has stronger activity against ATP-binding pocket mutations, and regorafenib is more effective against activation loop oncoproteins, the investigators explained.
They conducted a phase Ib trial to evaluate the safety and preliminary efficacy of the strategy in patients with metastatic GIST that had advanced on therapy with all established protocols. The trial had a standard 3+3 design to determine the recommended phase 2 dose; a total of 14 patients were enrolled, but only 12 received one or more complete cycles.
The median patient age was 63.5%. Nine patients had Eastern Cooperative Oncology Group performance status of 0, and five had an ECOG status of 1. The patients had received a median of four prior lines of therapy, and all had received at least three lines.
The primary mutations were at KIT exon 11 in eight patients, exon 9 in five patients, and a KIT/PDGFR-alpha wild type in one patient.
Of the 12 patients who received one or more complete cycles, 7 were treated with sunitinib 37.5 mg daily for 3 days, followed by regorafenib 120 mg daily for 4 days. There were no dose-limiting toxicities in this group. The median number of cycles delivered was 2 (range 1-4).
The other five patients were treated with sunitinib at the same 37.5 mg daily dose for 3 days, followed immediately by regorafenib 160 mg daily for 4 days. There were two dose-limiting toxicities in this group, both grade 3 hypophosphatemia, one of which was refractory to phosphorous replacement.
Antitumor activity according to Response Evaluation Criteria in Solid Tumors version 1.1 included four cases of stable disease at the time of the efficacy analysis, and eight cases of disease progression. The median progression-free survival was 1.9 months. As noted before, there were no complete or partial responses among the 12 patients.
A pharmacokinetic profile at cycle 1 showed that neither drug reached its reported active blood drug levels.
The patients appeared to tolerate the treatment well, with grade 1 or 2 fatigue in all patients being the most common adverse events. Grade 3 or 4 events included hand-foot syndrome, hypertension, and hypophosphatemia in two patients each.
As noted, the authors acknowledged that low drug exposure levels may explain the lack of any responses in this cohort.
“Therapeutic strategies based on KIT inhibition remain crucial in GIST patients progressing to multiple lines,” they wrote.
The study was supported by an ASCO Young Investigator Award, Pfizer, and Bayer. Dr. Serrano-Garcia disclosed honoraria from Bayer, a consulting or advisory role for Deciphera, research funding from Bayer and Deciphera, and travel accommodations and expenses from Pfizer.
SOURCE: Serrano-Garcia C et al. ASCO 2018, Abstract 11510.
CHICAGO – For patients with gastrointestinal stromal tumor (GIST) with KIT mutations conferring resistance to imatinib, a strategy of rapid alteration of drugs with complementary activity against KIT mutations is feasible but has thus far failed to yield significant clinical benefits, investigators said.
There were no objective responses among 12 patients treated continuously with 3 days of sunitinib (Sutent) followed by 4 days of regorafenib (Stivarga), and although 4 patients had stable disease in the short term, in each case the disease progressed within 16 weeks, reported Cesar Serrano-Garcia, MD, from the Dana-Farber Cancer Institute and Brigham and Women’s Hospital in Boston, and his colleagues.
“Drug exposure is critical to effectively target specific resistant subpopulations and low exposure may have contributed to the lack of efficacy in this cohort,” they wrote in a poster presented at the annual meeting of the American Society of Clinical Oncology.
The investigators noted that the main mechanism of resistance to imatinib (Gleevec) in GIST is polyclonal emergence of KIT secondary mutations. They then theorized that rapid alteration of sunitinib with regorafenib, which both have complementary activity against different KIT resistance mutations, could be a novel therapeutic strategy for controlling imatinib-resistant disease.
Both agents are active against KIT and platelet-derived growth factor receptor alpha (PDGFR-alpha). Sunitinib has stronger activity against ATP-binding pocket mutations, and regorafenib is more effective against activation loop oncoproteins, the investigators explained.
They conducted a phase Ib trial to evaluate the safety and preliminary efficacy of the strategy in patients with metastatic GIST that had advanced on therapy with all established protocols. The trial had a standard 3+3 design to determine the recommended phase 2 dose; a total of 14 patients were enrolled, but only 12 received one or more complete cycles.
The median patient age was 63.5%. Nine patients had Eastern Cooperative Oncology Group performance status of 0, and five had an ECOG status of 1. The patients had received a median of four prior lines of therapy, and all had received at least three lines.
The primary mutations were at KIT exon 11 in eight patients, exon 9 in five patients, and a KIT/PDGFR-alpha wild type in one patient.
Of the 12 patients who received one or more complete cycles, 7 were treated with sunitinib 37.5 mg daily for 3 days, followed by regorafenib 120 mg daily for 4 days. There were no dose-limiting toxicities in this group. The median number of cycles delivered was 2 (range 1-4).
The other five patients were treated with sunitinib at the same 37.5 mg daily dose for 3 days, followed immediately by regorafenib 160 mg daily for 4 days. There were two dose-limiting toxicities in this group, both grade 3 hypophosphatemia, one of which was refractory to phosphorous replacement.
Antitumor activity according to Response Evaluation Criteria in Solid Tumors version 1.1 included four cases of stable disease at the time of the efficacy analysis, and eight cases of disease progression. The median progression-free survival was 1.9 months. As noted before, there were no complete or partial responses among the 12 patients.
A pharmacokinetic profile at cycle 1 showed that neither drug reached its reported active blood drug levels.
The patients appeared to tolerate the treatment well, with grade 1 or 2 fatigue in all patients being the most common adverse events. Grade 3 or 4 events included hand-foot syndrome, hypertension, and hypophosphatemia in two patients each.
As noted, the authors acknowledged that low drug exposure levels may explain the lack of any responses in this cohort.
“Therapeutic strategies based on KIT inhibition remain crucial in GIST patients progressing to multiple lines,” they wrote.
The study was supported by an ASCO Young Investigator Award, Pfizer, and Bayer. Dr. Serrano-Garcia disclosed honoraria from Bayer, a consulting or advisory role for Deciphera, research funding from Bayer and Deciphera, and travel accommodations and expenses from Pfizer.
SOURCE: Serrano-Garcia C et al. ASCO 2018, Abstract 11510.
CHICAGO – For patients with gastrointestinal stromal tumor (GIST) with KIT mutations conferring resistance to imatinib, a strategy of rapid alteration of drugs with complementary activity against KIT mutations is feasible but has thus far failed to yield significant clinical benefits, investigators said.
There were no objective responses among 12 patients treated continuously with 3 days of sunitinib (Sutent) followed by 4 days of regorafenib (Stivarga), and although 4 patients had stable disease in the short term, in each case the disease progressed within 16 weeks, reported Cesar Serrano-Garcia, MD, from the Dana-Farber Cancer Institute and Brigham and Women’s Hospital in Boston, and his colleagues.
“Drug exposure is critical to effectively target specific resistant subpopulations and low exposure may have contributed to the lack of efficacy in this cohort,” they wrote in a poster presented at the annual meeting of the American Society of Clinical Oncology.
The investigators noted that the main mechanism of resistance to imatinib (Gleevec) in GIST is polyclonal emergence of KIT secondary mutations. They then theorized that rapid alteration of sunitinib with regorafenib, which both have complementary activity against different KIT resistance mutations, could be a novel therapeutic strategy for controlling imatinib-resistant disease.
Both agents are active against KIT and platelet-derived growth factor receptor alpha (PDGFR-alpha). Sunitinib has stronger activity against ATP-binding pocket mutations, and regorafenib is more effective against activation loop oncoproteins, the investigators explained.
They conducted a phase Ib trial to evaluate the safety and preliminary efficacy of the strategy in patients with metastatic GIST that had advanced on therapy with all established protocols. The trial had a standard 3+3 design to determine the recommended phase 2 dose; a total of 14 patients were enrolled, but only 12 received one or more complete cycles.
The median patient age was 63.5%. Nine patients had Eastern Cooperative Oncology Group performance status of 0, and five had an ECOG status of 1. The patients had received a median of four prior lines of therapy, and all had received at least three lines.
The primary mutations were at KIT exon 11 in eight patients, exon 9 in five patients, and a KIT/PDGFR-alpha wild type in one patient.
Of the 12 patients who received one or more complete cycles, 7 were treated with sunitinib 37.5 mg daily for 3 days, followed by regorafenib 120 mg daily for 4 days. There were no dose-limiting toxicities in this group. The median number of cycles delivered was 2 (range 1-4).
The other five patients were treated with sunitinib at the same 37.5 mg daily dose for 3 days, followed immediately by regorafenib 160 mg daily for 4 days. There were two dose-limiting toxicities in this group, both grade 3 hypophosphatemia, one of which was refractory to phosphorous replacement.
Antitumor activity according to Response Evaluation Criteria in Solid Tumors version 1.1 included four cases of stable disease at the time of the efficacy analysis, and eight cases of disease progression. The median progression-free survival was 1.9 months. As noted before, there were no complete or partial responses among the 12 patients.
A pharmacokinetic profile at cycle 1 showed that neither drug reached its reported active blood drug levels.
The patients appeared to tolerate the treatment well, with grade 1 or 2 fatigue in all patients being the most common adverse events. Grade 3 or 4 events included hand-foot syndrome, hypertension, and hypophosphatemia in two patients each.
As noted, the authors acknowledged that low drug exposure levels may explain the lack of any responses in this cohort.
“Therapeutic strategies based on KIT inhibition remain crucial in GIST patients progressing to multiple lines,” they wrote.
The study was supported by an ASCO Young Investigator Award, Pfizer, and Bayer. Dr. Serrano-Garcia disclosed honoraria from Bayer, a consulting or advisory role for Deciphera, research funding from Bayer and Deciphera, and travel accommodations and expenses from Pfizer.
SOURCE: Serrano-Garcia C et al. ASCO 2018, Abstract 11510.
REPORTING FROM ASCO 2018
Key clinical point: The strategy of rapid alteration of drugs to overcome mutations conferring imatinib resistance in gastrointestinal stromal tumor (GIST) was feasible but ineffective.
Major finding: There were no objective responses among 12 patients treated with the strategy.
Study details: A phase Ib clinical trial in 12 patients with heavily pretreated metastatic GIST.
Disclosures: The study was supported by an ASCO Young Investigator Award, Pfizer, and Bayer. Dr. Serrano-Garcia disclosed honoraria from Bayer, a consulting or advisory role for Deciphera, research funding from Bayer and Deciphera, and travel accommodations and expenses from Pfizer.
Source: Serrano-Garcia C et al. ASCO 2018, Abstract 11510.
ctDNA profiles pre- and posttreatment KIT mutations in GIST
CHICAGO – Detection of circulating tumor DNA (ctDNA), aka “liquid biopsy,” may serve as a noninvasive marker for disease heterogeneity and aid in the assessment of clinical responses to therapy for patients with gastrointestinal stromal tumors (GIST), according to investigators.
In a phase 1 trial of the investigational agent DCC-2618, a pan-KIT/platelet-derived growth factor receptor alpha (PDGFRA) switch control inhibitor, identification of ctDNA by next-generation sequencing (NGS) was accomplished in the majority of patients, with findings that support the need for a broad-spectrum KIT inhibitor for patients with GIST resistant to imatinib (Gleevec), reported Suzanne George, MD, of Dana-Farber Cancer Institute in Boston, and her colleagues.
“This data demonstrates for the first time that the distribution of resistance mutations in KIT across exons 13, 14, 17, and 18 or a combination thereof is similar in 2nd, 3rd, and 4th-line patients,” they wrote in a poster presented at the annual meeting of the American Society of Clinical Oncology.
The dose-finding and escalation trial included baseline evaluations of KIT/PDGFRA mutations with both ctDNA and fresh tumor biopsy, and ctDNA measurements during treatment.
Biopsy detected 68 KIT mutations at baseline in 81 patients, and ctDNA detected 75 mutations in 95 patients. Some patients had multiple mutations within one exon.
An analysis of mutations by response showed that of 73 patients with detectable KIT mutations by ctDNA at baseline, 35 became KIT ctDNA negative during at least one treatment time point. Of this group, 8 had a partial response (PR) and 27 had stable disease (SD). In all, 57 of the 73 patients had a more than 50% reduction in KIT mutation allele frequency (MAF).
Some patients with stable disease remained KIT negative out to 60 weeks following the first DCC-2618 dose.
The investigators also looked at ctDNA at baseline (21 patients) and post treatment (20 patients) in those who had a PR as their best response. Ten of these patients had KIT mutations detected at baseline, and of this group, eight became KIT negative after treatment; one had no detectable mutations in one exon and one exon with an MAF less than .1%. No posttreatment samples were available for the remaining patient.
There were preliminary data suggesting that DCC-2618 in the second line could be more efficacious than sunitinib (Sutent) in the same setting, and that in KIT-driven GIST DCC-2618 may provide more benefit in the second line compared with the fourth or subsequent lines of therapy, the authors stated.
“The mutational profile of KIT in tumors and plasma at baseline in GIST patients supports the need for a broad spectrum KIT inhibitor in all post-imatinib lines of therapy,” they wrote.
The trial is supported by Deciphera Pharmaceuticals. Dr. George disclosed stock or other ownership in Abbott Laboratories and Abbvie, consulting/advising for AstraZeneca, Blueprint Medicines, and Deciphera, and institutional research funding from Ariad, Bayer, Blueprint Medicine, Deciphera, Novartis, and Pfizer.
SOURCE: George S et al. ASCO 2018. Abstract 11511.
CHICAGO – Detection of circulating tumor DNA (ctDNA), aka “liquid biopsy,” may serve as a noninvasive marker for disease heterogeneity and aid in the assessment of clinical responses to therapy for patients with gastrointestinal stromal tumors (GIST), according to investigators.
In a phase 1 trial of the investigational agent DCC-2618, a pan-KIT/platelet-derived growth factor receptor alpha (PDGFRA) switch control inhibitor, identification of ctDNA by next-generation sequencing (NGS) was accomplished in the majority of patients, with findings that support the need for a broad-spectrum KIT inhibitor for patients with GIST resistant to imatinib (Gleevec), reported Suzanne George, MD, of Dana-Farber Cancer Institute in Boston, and her colleagues.
“This data demonstrates for the first time that the distribution of resistance mutations in KIT across exons 13, 14, 17, and 18 or a combination thereof is similar in 2nd, 3rd, and 4th-line patients,” they wrote in a poster presented at the annual meeting of the American Society of Clinical Oncology.
The dose-finding and escalation trial included baseline evaluations of KIT/PDGFRA mutations with both ctDNA and fresh tumor biopsy, and ctDNA measurements during treatment.
Biopsy detected 68 KIT mutations at baseline in 81 patients, and ctDNA detected 75 mutations in 95 patients. Some patients had multiple mutations within one exon.
An analysis of mutations by response showed that of 73 patients with detectable KIT mutations by ctDNA at baseline, 35 became KIT ctDNA negative during at least one treatment time point. Of this group, 8 had a partial response (PR) and 27 had stable disease (SD). In all, 57 of the 73 patients had a more than 50% reduction in KIT mutation allele frequency (MAF).
Some patients with stable disease remained KIT negative out to 60 weeks following the first DCC-2618 dose.
The investigators also looked at ctDNA at baseline (21 patients) and post treatment (20 patients) in those who had a PR as their best response. Ten of these patients had KIT mutations detected at baseline, and of this group, eight became KIT negative after treatment; one had no detectable mutations in one exon and one exon with an MAF less than .1%. No posttreatment samples were available for the remaining patient.
There were preliminary data suggesting that DCC-2618 in the second line could be more efficacious than sunitinib (Sutent) in the same setting, and that in KIT-driven GIST DCC-2618 may provide more benefit in the second line compared with the fourth or subsequent lines of therapy, the authors stated.
“The mutational profile of KIT in tumors and plasma at baseline in GIST patients supports the need for a broad spectrum KIT inhibitor in all post-imatinib lines of therapy,” they wrote.
The trial is supported by Deciphera Pharmaceuticals. Dr. George disclosed stock or other ownership in Abbott Laboratories and Abbvie, consulting/advising for AstraZeneca, Blueprint Medicines, and Deciphera, and institutional research funding from Ariad, Bayer, Blueprint Medicine, Deciphera, Novartis, and Pfizer.
SOURCE: George S et al. ASCO 2018. Abstract 11511.
CHICAGO – Detection of circulating tumor DNA (ctDNA), aka “liquid biopsy,” may serve as a noninvasive marker for disease heterogeneity and aid in the assessment of clinical responses to therapy for patients with gastrointestinal stromal tumors (GIST), according to investigators.
In a phase 1 trial of the investigational agent DCC-2618, a pan-KIT/platelet-derived growth factor receptor alpha (PDGFRA) switch control inhibitor, identification of ctDNA by next-generation sequencing (NGS) was accomplished in the majority of patients, with findings that support the need for a broad-spectrum KIT inhibitor for patients with GIST resistant to imatinib (Gleevec), reported Suzanne George, MD, of Dana-Farber Cancer Institute in Boston, and her colleagues.
“This data demonstrates for the first time that the distribution of resistance mutations in KIT across exons 13, 14, 17, and 18 or a combination thereof is similar in 2nd, 3rd, and 4th-line patients,” they wrote in a poster presented at the annual meeting of the American Society of Clinical Oncology.
The dose-finding and escalation trial included baseline evaluations of KIT/PDGFRA mutations with both ctDNA and fresh tumor biopsy, and ctDNA measurements during treatment.
Biopsy detected 68 KIT mutations at baseline in 81 patients, and ctDNA detected 75 mutations in 95 patients. Some patients had multiple mutations within one exon.
An analysis of mutations by response showed that of 73 patients with detectable KIT mutations by ctDNA at baseline, 35 became KIT ctDNA negative during at least one treatment time point. Of this group, 8 had a partial response (PR) and 27 had stable disease (SD). In all, 57 of the 73 patients had a more than 50% reduction in KIT mutation allele frequency (MAF).
Some patients with stable disease remained KIT negative out to 60 weeks following the first DCC-2618 dose.
The investigators also looked at ctDNA at baseline (21 patients) and post treatment (20 patients) in those who had a PR as their best response. Ten of these patients had KIT mutations detected at baseline, and of this group, eight became KIT negative after treatment; one had no detectable mutations in one exon and one exon with an MAF less than .1%. No posttreatment samples were available for the remaining patient.
There were preliminary data suggesting that DCC-2618 in the second line could be more efficacious than sunitinib (Sutent) in the same setting, and that in KIT-driven GIST DCC-2618 may provide more benefit in the second line compared with the fourth or subsequent lines of therapy, the authors stated.
“The mutational profile of KIT in tumors and plasma at baseline in GIST patients supports the need for a broad spectrum KIT inhibitor in all post-imatinib lines of therapy,” they wrote.
The trial is supported by Deciphera Pharmaceuticals. Dr. George disclosed stock or other ownership in Abbott Laboratories and Abbvie, consulting/advising for AstraZeneca, Blueprint Medicines, and Deciphera, and institutional research funding from Ariad, Bayer, Blueprint Medicine, Deciphera, Novartis, and Pfizer.
SOURCE: George S et al. ASCO 2018. Abstract 11511.
REPORTING FROM ASCO 2018
Key clinical point: Circulating tumor DNA can be used for mutational profiling and responses assessment in patients with advanced imatinib-resistant GIST.
Major finding: Of 73 patients with detectable KIT mutations by ctDNA at baseline, 35 became KIT ctDNA negative during at least one treatment time point.
Study details: Subanalyses from a phase 1 trial of DCC-2618.
Disclosures: The trial is supported by Deciphera Pharmaceuticals. Dr. George disclosed stock or other ownership in Abbott Laboratories and Abbvie, consulting/advising for AstraZeneca, Blueprint Medicines, and Deciphera, and institutional research funding from Ariad, Bayer, Blueprint Medicine, Deciphera, Novartis, and Pfizer.
Source: George S et al. ASCO 2018. Abstract 11511.
Novel TKI PLX9486 showed efficacy against KIT mutations in GIST
CHICAGO – A combination of the investigational agent PLX9486 with another novel tyrosine kinase inhibitor (TKI) showed some efficacy against a range of primary and secondary KIT mutations in patients with gastrointestinal stromal tumor (GIST), the results of a phase 1 dose escalation study have suggested.
Among 39 patients with GIST who had progressed on imatinib and other TKIs, the rates of clinical benefit at 16 weeks were 64% for 11 patients treated with PLX8486 monotherapy at a dose of 1,000 mg daily and 67% for 9 patients treated with PLX9486 and the investigational TKI pexidartinib.
One patient in the 1,000 mg monotherapy group had a partial response on interim analysis. The median progression-free survival in this dose group was 6 months, which was “significantly better than at lower doses,” reported Andrew J. Wagner, MD, PhD, from the Dana-Farber Cancer Institute in Boston and his colleagues.
“The combination of PLX9486 with either pexidartinib or sunitinib is generally well tolerated and toxicities are typically grade 1 or 2 in nature and reversible,” they wrote in a poster presented at the annual meeting of the American Society of Clinical Oncology.
PLX9486 is an inhibitor of KIT primary mutations in exons 9 and 11 and secondary resistance mutations in exons 17 and 18. Compared with other KIT-targeted TKIs, PLX9486 has complementary selectivity for mutant forms of KIT with a greater than 150-fold selectivity for mutant versus wild-type KIT, the investigators explained.
“Combinations of PLX9486 with either pexidartinib (PLX3397) or sunitinib potentially inhibit and address all common primary and secondary KIT mutations,” they wrote.
The investigators conducted a phase 1, open-label, dose-escalation study with two parts. The first part was designed to study the safety and pharmacokinetics of single-agent PLX9486 and established a maximum tolerated dose (MTD) for phase 2 studies. The second part was designed to study the drug as a single agent at the recommended phase 2 dose in GIST and other solid tumors with KIT mutations and also in combination with either pexidartinib or sunitinib in patients with GIST.
They found that single-agent PLX9486 was well tolerated at all doses tested (250, 300, 350, 500, and 1,000 mg daily) and that it selectively inhibited a spectrum of KIT mutations, “including difficult to treat exon 17/18 activation loop variants.”
The combination of PLX9486 at 500 mg and pexidartinib 600 mg was associated with three partial responses and a clinical benefit rate of 67%, with a PFS on interim analysis of 6 months.
The efficacy of single agent PLX9486 was suggested by circulating tumor DNA studies, which showed reductions in circulating tumor DNA levels of exons 11 and 17/18, which reflected the selectivity profile of the TKI.
In the PLX9486 dose escalation phase, there were three cases of grade 3 or 4 toxicities, including one case each of fatigue, creatinine phosphokinase increase, and hypophosphatemia.
The combination of PLX9486 and pexidartinib was associated with grade 1 or 2 adverse events, including hair color changes in five patients; fatigue and decreased appetite in four patients each; anemia, diarrhea, nausea, alanine aminotransferase increase, and aspartate aminotransferase increase in three patients each; and weight loss, maculopapular rash, and hypertension in two patients each.
At the time of the poster presentation, the sunitinib cohort was still accruing, and interim efficacy data were not available.
“Given these interim results, it is anticipated that the selectivity profile and potency of PLX9486 + sunitinib combination will achieve broader and more durable coverage of primary and secondary KIT mutations,” Dr. Wagner and his associates wrote.
SOURCE: Wagner AJ et al. ASCO 2018, Abstract 11509.
CHICAGO – A combination of the investigational agent PLX9486 with another novel tyrosine kinase inhibitor (TKI) showed some efficacy against a range of primary and secondary KIT mutations in patients with gastrointestinal stromal tumor (GIST), the results of a phase 1 dose escalation study have suggested.
Among 39 patients with GIST who had progressed on imatinib and other TKIs, the rates of clinical benefit at 16 weeks were 64% for 11 patients treated with PLX8486 monotherapy at a dose of 1,000 mg daily and 67% for 9 patients treated with PLX9486 and the investigational TKI pexidartinib.
One patient in the 1,000 mg monotherapy group had a partial response on interim analysis. The median progression-free survival in this dose group was 6 months, which was “significantly better than at lower doses,” reported Andrew J. Wagner, MD, PhD, from the Dana-Farber Cancer Institute in Boston and his colleagues.
“The combination of PLX9486 with either pexidartinib or sunitinib is generally well tolerated and toxicities are typically grade 1 or 2 in nature and reversible,” they wrote in a poster presented at the annual meeting of the American Society of Clinical Oncology.
PLX9486 is an inhibitor of KIT primary mutations in exons 9 and 11 and secondary resistance mutations in exons 17 and 18. Compared with other KIT-targeted TKIs, PLX9486 has complementary selectivity for mutant forms of KIT with a greater than 150-fold selectivity for mutant versus wild-type KIT, the investigators explained.
“Combinations of PLX9486 with either pexidartinib (PLX3397) or sunitinib potentially inhibit and address all common primary and secondary KIT mutations,” they wrote.
The investigators conducted a phase 1, open-label, dose-escalation study with two parts. The first part was designed to study the safety and pharmacokinetics of single-agent PLX9486 and established a maximum tolerated dose (MTD) for phase 2 studies. The second part was designed to study the drug as a single agent at the recommended phase 2 dose in GIST and other solid tumors with KIT mutations and also in combination with either pexidartinib or sunitinib in patients with GIST.
They found that single-agent PLX9486 was well tolerated at all doses tested (250, 300, 350, 500, and 1,000 mg daily) and that it selectively inhibited a spectrum of KIT mutations, “including difficult to treat exon 17/18 activation loop variants.”
The combination of PLX9486 at 500 mg and pexidartinib 600 mg was associated with three partial responses and a clinical benefit rate of 67%, with a PFS on interim analysis of 6 months.
The efficacy of single agent PLX9486 was suggested by circulating tumor DNA studies, which showed reductions in circulating tumor DNA levels of exons 11 and 17/18, which reflected the selectivity profile of the TKI.
In the PLX9486 dose escalation phase, there were three cases of grade 3 or 4 toxicities, including one case each of fatigue, creatinine phosphokinase increase, and hypophosphatemia.
The combination of PLX9486 and pexidartinib was associated with grade 1 or 2 adverse events, including hair color changes in five patients; fatigue and decreased appetite in four patients each; anemia, diarrhea, nausea, alanine aminotransferase increase, and aspartate aminotransferase increase in three patients each; and weight loss, maculopapular rash, and hypertension in two patients each.
At the time of the poster presentation, the sunitinib cohort was still accruing, and interim efficacy data were not available.
“Given these interim results, it is anticipated that the selectivity profile and potency of PLX9486 + sunitinib combination will achieve broader and more durable coverage of primary and secondary KIT mutations,” Dr. Wagner and his associates wrote.
SOURCE: Wagner AJ et al. ASCO 2018, Abstract 11509.
CHICAGO – A combination of the investigational agent PLX9486 with another novel tyrosine kinase inhibitor (TKI) showed some efficacy against a range of primary and secondary KIT mutations in patients with gastrointestinal stromal tumor (GIST), the results of a phase 1 dose escalation study have suggested.
Among 39 patients with GIST who had progressed on imatinib and other TKIs, the rates of clinical benefit at 16 weeks were 64% for 11 patients treated with PLX8486 monotherapy at a dose of 1,000 mg daily and 67% for 9 patients treated with PLX9486 and the investigational TKI pexidartinib.
One patient in the 1,000 mg monotherapy group had a partial response on interim analysis. The median progression-free survival in this dose group was 6 months, which was “significantly better than at lower doses,” reported Andrew J. Wagner, MD, PhD, from the Dana-Farber Cancer Institute in Boston and his colleagues.
“The combination of PLX9486 with either pexidartinib or sunitinib is generally well tolerated and toxicities are typically grade 1 or 2 in nature and reversible,” they wrote in a poster presented at the annual meeting of the American Society of Clinical Oncology.
PLX9486 is an inhibitor of KIT primary mutations in exons 9 and 11 and secondary resistance mutations in exons 17 and 18. Compared with other KIT-targeted TKIs, PLX9486 has complementary selectivity for mutant forms of KIT with a greater than 150-fold selectivity for mutant versus wild-type KIT, the investigators explained.
“Combinations of PLX9486 with either pexidartinib (PLX3397) or sunitinib potentially inhibit and address all common primary and secondary KIT mutations,” they wrote.
The investigators conducted a phase 1, open-label, dose-escalation study with two parts. The first part was designed to study the safety and pharmacokinetics of single-agent PLX9486 and established a maximum tolerated dose (MTD) for phase 2 studies. The second part was designed to study the drug as a single agent at the recommended phase 2 dose in GIST and other solid tumors with KIT mutations and also in combination with either pexidartinib or sunitinib in patients with GIST.
They found that single-agent PLX9486 was well tolerated at all doses tested (250, 300, 350, 500, and 1,000 mg daily) and that it selectively inhibited a spectrum of KIT mutations, “including difficult to treat exon 17/18 activation loop variants.”
The combination of PLX9486 at 500 mg and pexidartinib 600 mg was associated with three partial responses and a clinical benefit rate of 67%, with a PFS on interim analysis of 6 months.
The efficacy of single agent PLX9486 was suggested by circulating tumor DNA studies, which showed reductions in circulating tumor DNA levels of exons 11 and 17/18, which reflected the selectivity profile of the TKI.
In the PLX9486 dose escalation phase, there were three cases of grade 3 or 4 toxicities, including one case each of fatigue, creatinine phosphokinase increase, and hypophosphatemia.
The combination of PLX9486 and pexidartinib was associated with grade 1 or 2 adverse events, including hair color changes in five patients; fatigue and decreased appetite in four patients each; anemia, diarrhea, nausea, alanine aminotransferase increase, and aspartate aminotransferase increase in three patients each; and weight loss, maculopapular rash, and hypertension in two patients each.
At the time of the poster presentation, the sunitinib cohort was still accruing, and interim efficacy data were not available.
“Given these interim results, it is anticipated that the selectivity profile and potency of PLX9486 + sunitinib combination will achieve broader and more durable coverage of primary and secondary KIT mutations,” Dr. Wagner and his associates wrote.
SOURCE: Wagner AJ et al. ASCO 2018, Abstract 11509.
PRESENTED AT ASCO 2018
Key clinical point: The novel tyrosine kinase inhibitor PLX9486 showed activity against resistance mutations in gastrointestinal stromal tumors.
Major finding: The combination of PLX9486 at 500 mg and pexidartinib 600 mg was associated with three partial responses and a clinical benefit rate of 67% with a PFS on interim analysis of 6 months.
Study details: Phase 1 dose-escalation, safety and pharmacokinetics study in 39 patients with GIST, four with adenocarcinomas, and one with follicular lymphoma.
Disclosures: The study was sponsored by Plexxikon. Dr. Wagner disclosed consulting or advisory roles with Prime Therapeutics, Lilly, and Loxo Oncology, as well as having received institutional research funding from AADi, Celldex Therapeutics, Daiichi Sankyo, Karyopharm Therapeutics, Lilly, and Plexxikon.
Source: Wagner AJ et al. ASCO 2018, Abstract 11509.
Adult soft tissue sarcoma: Professional resources from the National Cancer Institute
Adult Soft Tissue Sarcoma Treatment (PDQ®)–Health Professional Version
General Information About Adult Soft Tissue Sarcoma
Incidence and Mortality
Estimated new cases and deaths from soft tissue sarcoma in the United States in 2018:[1]
- New cases: 13,040.
- Deaths: 5,150.
Soft tissue sarcomas are malignant tumors that arise in any of the mesodermal tissues of the extremities (50%), trunk and retroperitoneum (40%), or head and neck (10%). The reported international incidence rates range from 1.8 to 5 per 100,000 individuals per year.[2]
Risk Factors and Genetic Factors
The risk of sporadic soft tissue sarcomas is increased by previous radiation therapy and, in the case of lymphangiosarcoma, by chronic lymphedema. The chemicals Thorotrast (thorium dioxide), vinyl chloride, and arsenic are also established carcinogens for hepatic angiosarcomas.[3-5]
Soft tissue sarcomas occur with greater frequency in patients with the following inherited syndromes:[3-5]
- Nevoid basal cell carcinoma syndrome (Gorlin syndrome: PTC gene mutation).
- Gardner syndrome (APC mutation).
- Li-Fraumeni syndrome (p53 mutation).
- Tuberous sclerosis (Bourneville disease: TSC1 or TSC2 mutation).
- von Recklinghausen disease (neurofibromatosis type 1: NF1 mutation).
- Werner syndrome (adult progeria: WRN mutation).
Diagnosis
Soft tissue sarcomas may be heterogeneous, so adequate tissue should be obtained via either core-needle or incisional biopsy for microscopic examination to determine histologic type and tumor grade. Careful planning of the initial biopsy is important to avoid compromising subsequent curative resection. Since the selection of treatment is determined by the grade of the tumor, it is essential to have a careful review of the biopsy tissue by a pathologist who is experienced in diagnosing sarcomas. Complete staging and treatment planning by a multidisciplinary team of cancer specialists is required to determine the optimal treatment for patients with this disease.
There is evidence that at least some favorable clinical outcomes may be associated with referral to a specialized sarcoma treatment center. In a population-based consecutive series of 375 soft tissue sarcoma patients in Sweden, local recurrence rates of resected tumors were higher in patients who were not referred to the specialized center: in 35 of 78 (45%) patients not referred; in 24 of 102 (24%) patients referred after initial surgery or incisional biopsy; and in 36 of 195 (18%) patients referred before any surgical procedure (P = .0001 for the difference between those never referred vs. those referred before any surgical procedure).[6][Level of evidence: 3iDii] However, there were no statistically significant differences in death from sarcoma between the groups of patients.
Prognostic Factors
The prognosis for patients with adult soft tissue sarcomas depends on several factors, including:[3-5,7,8]
- Patient’s age.
- Size, sarcoma subtype, histologic grade, mitotic activity, and stage of the tumor.
Factors associated with a poorer prognosis include the following:[9]
- Age older than 60 years.
- Tumors larger than 5 cm in greatest dimension.
- High-grade histology with high mitotic activity.
- Positive margins after resection.[10]
Although low-grade tumors are usually curable by surgery alone, higher-grade sarcomas (as determined by the mitotic index and by the presence of hemorrhage and necrosis) are associated with higher local-treatment failure rates and increased metastatic potential.
Surveillance for Relapse
A retrospective review included 174 consecutive patients with a soft tissue sarcoma of the limb who underwent follow-up by oncologists at a single center from 2003 to 2009.[11] The rate and site of recurrence and mode of detection were analyzed. Eighty-two patients (47%) experienced relapse. Isolated local recurrences occurred in 26 patients and local relapse with synchronous pulmonary metastases occurred in 5 patients. Local recurrences were detected clinically in 30 of the 31 patients; magnetic resonance imaging identified only one local recurrence. Twenty-eight patients developed isolated lung metastases; in 9 patients, the lung metastases were amenable to resections, 7 of whom were free of disease after treatment. Lung metastases were detected by chest x-ray in 19 patients, by computed tomography scanning in 3 patients, and clinically in 11 patients. Twenty-three patients developed nonpulmonary metastases. More than 80% of the relapses occurred in the first 2 years of follow-up; however, later recurrences were also observed.[11][Level of evidence: 3iiDi] This study supports imaging surveillance for detection of lung metastases, whereas local recurrences at the primary site were usually detected by clinical examination. The impact of picking up metastases from overall survival or quality-of-life data is unknown.
Related Summaries
Other PDQ summaries containing information about soft tissue sarcoma include:
References
- American Cancer Society: Cancer Facts and Figures 2018. Atlanta, Ga: American Cancer Society, 2018. Available online. Last accessed January 5, 2018.
- Wibmer C, Leithner A, Zielonke N, et al.: Increasing incidence rates of soft tissue sarcomas? A population-based epidemiologic study and literature review. Ann Oncol 21 (5): 1106-11, 2010. [PUBMED Abstract]
- Singer S, Nielsen T, Antonescu CR: Molecular biology of soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1522-32.
- Singer S, Maki RG, O'Sullivan B: Soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1533-77.
- Malawer MM, Helman LJ, O'Sullivan B: Sarcomas of bone. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1578-1609.
- Gustafson P, Dreinhöfer KE, Rydholm A: Soft tissue sarcoma should be treated at a tumor center. A comparison of quality of surgery in 375 patients. Acta Orthop Scand 65 (1): 47-50, 1994. [PUBMED Abstract]
- Coindre JM, Terrier P, Guillou L, et al.: Predictive value of grade for metastasis development in the main histologic types of adult soft tissue sarcomas: a study of 1240 patients from the French Federation of Cancer Centers Sarcoma Group. Cancer 91 (10): 1914-26, 2001. [PUBMED Abstract]
- Kasper B, Ouali M, van Glabbeke M, et al.: Prognostic factors in adolescents and young adults (AYA) with high risk soft tissue sarcoma (STS) treated by adjuvant chemotherapy: a study based on pooled European Organisation for Research and Treatment of Cancer (EORTC) clinical trials 62771 and 62931. Eur J Cancer 49 (2): 449-56, 2013. [PUBMED Abstract]
- Vraa S, Keller J, Nielsen OS, et al.: Prognostic factors in soft tissue sarcomas: the Aarhus experience. Eur J Cancer 34 (12): 1876-82, 1998. [PUBMED Abstract]
- Trovik LH, Ovrebo K, Almquist M, et al.: Adjuvant radiotherapy in retroperitoneal sarcomas. A Scandinavian Sarcoma Group study of 97 patients. Acta Oncol 53 (9): 1165-72, 2014. [PUBMED Abstract]
- Rothermundt C, Whelan JS, Dileo P, et al.: What is the role of routine follow-up for localised limb soft tissue sarcomas? A retrospective analysis of 174 patients. Br J Cancer 110 (10): 2420-6, 2014. [PUBMED Abstract]
Cellular Classification of Adult Soft Tissue Sarcoma
Soft tissue sarcomas are classified histologically according to the soft tissue cell of origin. Additional studies, including electron microscopy, specialized immunohistochemistry, flow cytometry, cytogenetics, and tissue culture studies may allow identification of particular subtypes within the major histologic categories. For example, S100 antigen suggests neural sheath origin, cytokeratin suggests epithelioid or synovial cell origin, and factor VIII-related antigen suggests endothelial origin. Likewise, some subtypes of sarcomas have characteristic genetic markers, but these markers are not generally used in the routine clinical setting (e.g., translocation t(X;18)(p11;q11) in synovial sarcomas and translocation t(12;16)(q13;p11) in myxoid and round-cell sarcomas).[1-3]
The histologic grade reflects the metastatic potential of these tumors more accurately than the classic cellular classification listed below. Pathologists assign a grade based on the number of mitoses per high-powered field, the presence of necrosis, cellular and nuclear morphology, and the degree of cellularity; discordance among expert pathologists regarding tumor grade, and even histologic subtype, can be substantial.[4]
The World Health Organization lists the following cell types in its classification of soft tissue sarcomas:[5,6]
- Adipocytic tumors.
- Dedifferentiated liposarcoma.*
- Myxoid/round cell liposarcoma.
- Pleomorphic liposarcoma.
- Fibroblastic/myofibroblastic tumors.
- Fibrosarcoma.**
- Myxofibrosarcoma, low grade.
- Low-grade fibromyxoid sarcoma.
- Sclerosing epithelioid fibrosarcoma.
- So-called fibrohistiocytic tumors.
- Undifferentiated pleomorphic sarcoma/malignant fibrous histiocytoma (including pleomorphic, giant cell, myxoid/high-grade myxofibrosarcoma, and inflammatory forms).
- Smooth muscle tumors.
- Leiomyosarcoma.
- Skeletal muscle tumors.
- Rhabdomyosarcoma (embryonal, alveolar, and pleomorphic forms).
- Vascular tumors.
- Epithelioid hemangioendothelioma.
- Angiosarcoma, deep.***
- Tumors of peripheral nerves.
- Malignant peripheral nerve sheath tumor.
- Chondro-osseous tumors.
- Extraskeletal chondrosarcoma (mesenchymal and other variants).
- Extraskeletal osteosarcoma.
- Tumors of uncertain differentiation.
- Synovial sarcoma.
- Epithelioid sarcoma.
- Alveolar soft part sarcoma.
- Clear cell sarcoma of soft tissue.
- Extraskeletal myxoid chondrosarcoma.
- Primitive neuroectodermal tumor/extraskeletal Ewing tumor.
- Desmoplastic small round cell tumor.
- Extrarenal rhabdoid tumor.
- Undifferentiated sarcoma; sarcoma, not otherwise specified.
[Note: *It is recognized that dedifferentiated liposarcoma primarily arises in the context of deep atypical lipomatous tumor/well-differentiated liposarcoma, a sarcoma of intermediate malignancy because of the lack of metastatic capacity. **The category of fibrosarcoma can be inclusive of fibrosarcomatous differentiation in dermatofibrosarcoma protuberans. ***Cutaneous angiosarcoma may be difficult to stage using the American Joint Committee on Cancer system. (Refer to the PDQ summary on Gastrointestinal Stromal Tumors for more information.)]
References
- Singer S, Nielsen T, Antonescu CR: Molecular biology of soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1522-32.
- Singer S, Maki RG, O'Sullivan B: Soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1533-77.
- Malawer MM, Helman LJ, O'Sullivan B: Sarcomas of bone. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1578-1609.
- Alvegård TA, Berg NO: Histopathology peer review of high-grade soft tissue sarcoma: the Scandinavian Sarcoma Group experience. J Clin Oncol 7 (12): 1845-51, 1989. [PUBMED Abstract]
- Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-6.
- Brodowicz T, Schwameis E, Widder J, et al.: Intensified Adjuvant IFADIC Chemotherapy for Adult Soft Tissue Sarcoma: A Prospective Randomized Feasibility Trial. Sarcoma 4 (4): 151-60, 2000. [PUBMED Abstract]
Stage Information for Adult Soft Tissue Sarcoma
Note: The American Joint Committee on Cancer (AJCC) has published the 8th edition of the AJCC Cancer Staging Manual, which includes revisions to the staging for this disease. Implementation of the 8th edition began in January 2018. The PDQ Adult Treatment Editorial Board, which maintains this summary, is reviewing the revised staging and will make appropriate changes as needed.
Staging has an important role in determining the most effective treatment for soft tissue sarcoma. Clinical staging involves magnetic resonance imaging (MRI) or computed tomography (CT) of the primary tumor area and a chest CT to look for metastasis to the lung (the most common site of distant spread). An abdominal CT scan is done in the case of retroperitoneal sarcomas because the liver may be the site of initial clinical metastasis for these tumors.
The stage is determined by the size of the tumor, the histologic grade, and whether there is spread to lymph nodes or distant sites. Intracompartmental or extracompartmental extension of extremity sarcomas is also important for surgical decision making. For complete staging, a thorough review of all biopsy specimens (including those from the primary tumor, lymph nodes, or other suspicious lesions) is essential. CT scan of the chest is recommended for sarcomas larger than 5 cm (T2) or with moderate to poor differentiation (grades 2–4). Nodal involvement is rare, occurring in fewer than 3% of patients with sarcoma.[1]
Lymph node involvement in soft tissue sarcomas of adulthood is rare but is somewhat more frequent in some subtypes (e.g., rhabdomyosarcoma, vascular sarcomas, clear cell sarcomas, and epithelioid sarcomas) when they are high grade.[2] Because treatment decisions are predicated on pathology staging, patients should be staged before, and again after, any neoadjuvant therapy. The assessment of tumor grade can be affected in either direction, but more frequently decreased because of differential cellular loss related to the neoadjuvant chemotherapy or radiation.[3] Grade, which is based on cellular differentiation, mitotic rate, and extent of necrosis, should be recorded for all soft tissue sarcomas. A three-grade system (G1–G3) is preferred. (See below.)
The AJCC has designated staging by the four criteria of tumor size, nodal status, metastasis, and grade (TNMG).[3] The characteristic molecular markers of some sarcomas are not formally incorporated in the staging system pending further evaluation of their impact on prognosis. Recurrent sarcomas are restaged using the same system as for primary tumors with the specification that the tumor is recurrent.
Definitions of TNM and Grade
| aReprinted with permission from AJCC: Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-8. | |
| bSuperficial tumor is located exclusively above the superficial fascia without invasion of the fascia; deep tumor is located either exclusively beneath the superficial fascia, superficial to the fascia with invasion of or through the fascia, or both superficial yet beneath the fascia. | |
| TX | Primary tumor cannot be assessed. |
| T0 | No evidence of primary tumor. |
| T1 | Tumor ≤5 cm in greatest dimension. (Size should be regarded as a continuous variable, and the measurement should be provided.) |
| T1a | Superficial tumor.b |
| T1b | Deep tumor.b |
| T2 | Tumor >5 cm in greatest dimension.b |
| T2a | Superficial tumor.b |
| T2b | Deep tumor. |
| aReprinted with permission from AJCC: Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-8. | |
| bPresence of positive nodes (N1) in M0 tumors is considered Stage III. | |
| NX | Regional lymph nodes cannot be assessed. |
| N0 | No regional lymph node metastasis. |
| N1b | Regional lymph node metastasis. |
| aReprinted with permission from AJCC: Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-8. | |
| M0 | No distant metastasis. |
| M1 | Distant metastasis. |
| aReprinted with permission from AJCC: Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-8. | ||||
| Stage IA | T1a | N0 | M0 | G1, GX |
| T1b | N0 | M0 | G1, GX | |
| Stage IB | T2a | N0 | M0 | G1, GX |
| T2b | N0 | M0 | G1, GX | |
| Stage IIA | T1a | N0 | M0 | G2, G3 |
| T1b | N0 | M0 | G2, G3 | |
| Stage IIB | T2a | N0 | M0 | G2 |
| T2b | N0 | M0 | G2 | |
| Stage III | T2a, T2b | N0 | M0 | G3 |
| Any T | N1 | M0 | Any G | |
| Stage IV | Any T | Any N | M1 | Any G |
Neurovascular and bone invasion are indicators of poor prognosis, but they are not incorporated into the formal staging system.
References
- Fong Y, Coit DG, Woodruff JM, et al.: Lymph node metastasis from soft tissue sarcoma in adults. Analysis of data from a prospective database of 1772 sarcoma patients. Ann Surg 217 (1): 72-7, 1993. [PUBMED Abstract]
- Mazeron JJ, Suit HD: Lymph nodes as sites of metastases from sarcomas of soft tissue. Cancer 60 (8): 1800-8, 1987. [PUBMED Abstract]
- Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-6.
Treatment Option Overview
Multimodality Approach
In most cases, a combined modality approach of preoperative radiation therapy (preRT) or postoperative radiation therapy (PORT) is used, rather than the radical surgical procedures, such as amputation, that were used in the past. It may even be possible to use surgery without PORT in selected cases. For example, a case series was reported from a specialized sarcoma treatment referral center in which 74 selected patients with primary extremity and trunk tumors 5 cm or smaller were found to have no histologic involvement of the surgical margins. The patients were observed without radiation therapy, and the estimated local recurrence rate after 10 years was 11%.[1][Level of evidence: 3iiiDiv] The role of chemotherapy is not as well defined as is the role for radiation therapy. Because of the evolving nature of the treatment options for this disease, patients should be considered when available. Information about ongoing clinical trials is available from the NCI website.
Role of Surgery
Surgical resection is the mainstay of therapy for soft tissue sarcomas. When feasible, wide-margin function–sparing surgical excision is the cornerstone of effective treatment for extremity tumors. This may be facilitated by soft tissue reconstructive surgery, which generally permits wider margins than those obtained when the surgical plan involves direct closure of the excision site.[2] Cutting into the tumor mass or shelling out the gross tumor along the plane of the pseudocapsule of compressed tumor cells and reactive tissue that often surrounds soft tissue sarcomas are associated with an elevated risk of local recurrence. Even high-grade, soft tissue sarcomas of the extremities can usually be effectively treated while preserving the limb with combined-modality treatment consisting of preRT or PORT to reduce local recurrence. (Refer to the Role of Radiation Therapy section of this summary for more information.)
Only one small, single-institution, randomized trial has directly compared amputation to limb-sparing surgery for soft tissue sarcomas of the extremities.[3] In a 2:1 randomization ratio, 27 patients with high-grade extremity sarcomas were assigned to a wide excision plus PORT (45 Gy–50 Gy to the wide local excision area, and a total of 60 Gy–70 Gy to the tumor bed over 6–7 weeks), and 16 were assigned to amputation at or above the joint proximal to the tumor. Both groups received adjuvant chemotherapy (i.e., doxorubicin, cyclophosphamide, and high-dose methotrexate). At 63 months, with a median follow-up of 56 months, there were four local recurrences in the 27 patients who underwent limb-sparing surgery and no recurrences in the 16 patients who underwent amputation P2 = .12. Overall survival (OS) rates were not statistically significantly different (actuarial 5-year survival rate, 83% vs. 88%, P2 = .99).[3][Level of evidence: 1iiA]
Local control of high-grade soft tissue sarcomas of the trunk and the head and neck can be achieved with surgery in combination with radiation therapy.[4] It may be possible to use surgery without PORT in selected cases. For example, a case series was reported from a specialized sarcoma treatment referral center in which 74 selected patients with primary extremity and trunk tumors 5 cm or smaller were found to have no histologic involvement of the surgical margins.[1] They were observed without radiation therapy, and the estimated local recurrence rate after 10 years was 11%.[1][Level of evidence: 3iiiDiv] The role of chemotherapy is not as well defined as is the role of radiation therapy. Because of the evolving nature of the treatment options for this disease, patients should be offered the option of clinical trials when available.
Effective treatment of retroperitoneal sarcomas requires removal of all gross disease while sparing adjacent viscera not invaded by tumor. The prognosis for patients with high-grade retroperitoneal sarcomas is less favorable than for patients with tumors at other sites, partly because of the difficulty in completely resecting these tumors and the dose-limiting toxicity of high-dose radiation therapy on visceral organs.[5-8]
In the setting of distant metastasis, surgery may be associated with long-term, disease-free survival in patients with pulmonary metastasis and optimal underlying disease biology (i.e., patients with a limited number of metastases and slow nodule growth) who have undergone or are undergoing complete resection of the primary tumor.[9-11] It is not clear to what degree the favorable outcomes are attributable to the efficacy of surgery or the careful selection of patients based on factors that are associated with less-virulent disease.
Role of Radiation Therapy
Radiation plays an important role in limb-sparing therapy. Pre- and postoperative external-beam radiation therapies (EBRT), as well as brachytherapy, have been shown to decrease the risk of local recurrence. They have not been shown to increase OS but are used to avoid amputation for all but the most locally advanced tumors or for limbs seriously compromised by vascular disease, where acceptable functional preservation is not possible. In the case of EBRT, irradiation of the entire limb circumference is avoided to preserve vascular and nerve structures that are critical to function and preservation of the limb.
PORT
PORT has been tested in a single-institution, randomized trial of 141 patients with extremity sarcomas who were treated with limb-sparing surgery. Patients with high-grade tumors (n = 91) also received adjuvant chemotherapy (i.e., five 28-day cycles of doxorubicin and cyclophosphamide). All patients were randomly assigned to receive radiation (45 Gy to a wide field, plus a tumor-bed boost of 18 Gy over 6–7 weeks), concurrent with chemotherapy in the case of high-grade tumors versus no radiation.[12] At up to 12 years of follow-up, there was one local recurrence in the 70 patients randomly assigned to receive radiation versus 17 recurrences in the 71 control patients (P = .0001), with similar reduction in risk of local recurrence for both high- and low-grade tumors. However, there was no difference in OS between the radiation and control groups.[12][Level of evidence: 1iiDiii] Global quality of life was similar in the two groups, but the radiation therapy group had substantially worse functional deficits resulting from reduced strength and joint motion as well as increased edema.
To limit acute toxicity with preRT, smaller fields and lower doses are generally given than is the case with PORT. PreRT has been directly compared with PORT for extremity soft tissue sarcomas in a multicenter randomized trial.[13-15] Designed to include 266 patients, the trial was stopped early after 190 patients had been accrued because of an increase in wound complications in the preRT group. The scheduled radiation in the preRT group was a wide field of 50 Gy in 2-Gy fractions (first phase of the trial) with an additional 16 Gy to 20 Gy to the tumor bed and a 2-cm margin (second phase of the trial) only if tumor cells were found at the surgical margins.
Patients in the PORT group were scheduled to receive radiation during both phases of the trial. The wound-complication rates were 35% versus 17% in the preRT and PORT groups, respectively (P = .01). In addition, limb function at 6 weeks after surgery was worse in the preRT group (P = .01).[13] At 5 years, the two groups had similar local control rates (93% vs. 92%) and OS (73% vs. 67%, P = .48).[14] Of the 129 patients evaluated for limb function at 21 to 27 months after surgery (n = 73 for preRT and n = 56 for PORT), limb function was similar in both groups, but there was a statistical trend for less fibrosis in the preRT group (P = .07).[15]
Brachytherapy
Brachytherapy has also been investigated as an adjuvant therapy for soft tissue sarcomas. Although it has possible advantages of convenience and less radiation to normal surrounding tissue relative to EBRT, the two treatment strategies have not been directly compared in terms of efficacy or morbidity. However, adjuvant brachytherapy has been compared with surgery without radiation. The time interval between preRT and surgical excision in extremity soft tissue sarcoma had minimal influence on the development of wound complications. Four- or 5-week intervals showed equivalent complication rates between patients who did or did not develop wound complications, suggesting an optimal interval to reduce potential complications.[16]
In a single-institution trial, 164 patients with sarcomas of the extremity or superficial trunk were randomly assigned during surgery, if all gross tumor could be excised, to receive an iridium Ir 192 implant (delivering 42 Gy–45 Gy over 4–6 days; 78 patients) or to a control arm of no radiation (86 patients).[17,18] Some of the patients with high-grade tumors received adjuvant doxorubicin-based chemotherapy if they were thought to be at a high risk for metastasis (34 patients in each study arm). With a median follow-up of 76 months, the 5-year actuarial local recurrence rates were 18% and 31% in the brachytherapy and control arms, respectively (P = .04). This difference was limited to patients with high-grade tumors. There was no discernible difference in sarcoma-specific survival rates between the brachytherapy and control arms (84% and 81%, respectively; P = .65), and there was no difference in the high tumor-grade group.[17][Level of evidence: 1iiDiii] The rates of clinically important wound complications (e.g., need for operative revision or repeated seroma drainage, wound separation, large hematomas, or purulent infection) were 24% and 14% in the radiation and control arms, respectively (P = .13); wound reoperation rates were 10% and 0%, respectively (P = .006).[18]
Intensity-modulated radiation therapy
Intensity-modulated radiation therapy (IMRT) has been used to deliver preRT or PORT to patients with extremity soft tissue sarcomas in an effort to spare the femur, joints, and selected other normal tissues from the full prescription dose and to maintain local control while potentially reducing radiation therapy-related morbidity. Initial single-institution reports suggest that high rates of local control with some reduction in morbidity are possible with this technique.[19,20] Retrospective comparison of IMRT compared with 3-dimensional, conformal radiation therapy demonstrates that local recurrence for primary soft tissue sarcomas of the extremity was worse in the non-IMRT group.[21][Level of evidence: 3iiiDiv]
Surgery and radiation therapy
In some tumors of the extremities or trunk, surgery alone can be performed without the use of radiation. Evidence for this approach is limited to single-institution, relatively small, case series [1,22,23] or analysis of outcomes in the National Cancer Institute's Surveillance, Epidemiology, and End Results (SEER) tumor registry.[24] However, these comparisons suffer from low statistical power and differential evaluability rates that could have introduced bias.[1] Patient selection factors may vary among surgeons. In general, this approach is considered in patients with low-grade tumors of the extremity or superficial trunk that are 5 cm or smaller in diameter (T1) and have microscopically negative surgical margins; long-term local tumor control is about 90% in such patients.[25]
A patterns-of-care study using SEER data was queried to identify patients undergoing surgery for truncal and extremity soft tissue sarcomas from 2004 to 2009.[26] Of 5,075 patients, 50% received radiation therapy. Radiation was considered to be underused in a significant portion of patients undergoing treatment for soft tissue sarcoma in the United States. Although routine radiation therapy is not recommended for stage I patients, 25% of them still underwent radiation. Even though routine radiation therapy is recommended for patients with stage II and III tumors, only 60% of them underwent radiation. On multivariate analysis, predictors of radiation therapy included age younger than 50 years (odds ratio [OR], 1.57; 95% confidence interval [CI], 1.28–1.91), malignant fibrous histiocytoma histology (OR, 1.47; 95% CI, 1.3–1.92), T2 classification (OR, 1.88; 95% CI, 1.60–2.20), and G3 (OR, 6.27; 95% CI, 5.10–7.72). Patients with stage III soft tissue sarcoma who received radiation therapy showed improved disease-specific survival at 5 years compared with those who did not (68% vs. 46%, P < .001).[26][Level of evidence: 3iDii]
On occasion, surgical excision cannot be performed in the initial management of soft tissue sarcomas because the morbidity would be unacceptable or nearby critical organs make complete resection impossible. In such circumstances, radiation has been used as the primary therapy.[27] However, this must be considered a treatment of last resort. Experience is limited to retrospective case series from single centers.[27][Level of evidence: 3iiiDiv]
Role of Adjuvant or Neoadjuvant Chemotherapy for Clinically Localized Tumors
The role of adjuvant chemotherapy is not completely clear. The investigation of its use falls into two categories or generations—pre- and post-ifosfamide regimens. In discussions with a patient, any potential benefits should be considered in the context of the short- and long-term toxicities of the chemotherapy.
First-generation trials (preifosfamide)
Several prospective, randomized trials were unable to determine conclusively whether doxorubicin-based adjuvant chemotherapy benefits adults with resectable soft tissue sarcomas. The majority of these studies accrued small numbers of patients and did not demonstrate a metastasis-free survival or an OS benefit for adjuvant chemotherapy.[4] A small study of adjuvant chemotherapy showed a positive effect on both disease-free survival (DFS) and OS in patients treated with postoperative chemotherapy.[28] There was wide interstudy variability among the reported trials, including differences in therapeutic regimens, drug doses, sample size, tumor site, and histologic grade.
A quantitative meta-analysis of updated data from 1,568 individual patients in 14 trials of doxorubicin-based adjuvant therapy showed an absolute benefit from adjuvant therapy of 6% for a local relapse-free interval (95% CI, 1%–10%), 10% for a distant relapse-free interval (95% CI, 5%–15%), and 10% for recurrence-free survival (95% CI, 5%–15%). A statistically significant OS benefit at 10 years was not detected: absolute difference 4% (95% CI, -1%–+9%).[29,30][Level of evidence: 1iiDii] However, only a small proportion of patients in this meta-analysis were treated with ifosfamide, an agent with demonstrated activity against soft tissue sarcoma. In addition, a subset analysis suggested that patients with sarcomas of the extremities may have benefited from adjuvant chemotherapy (hazard ratio [HR] for death, 0.8, P = .029), but there was no clear evidence that patients with extremity sarcomas had outcomes that were statistically significantly different from the outcomes of patients with tumors at other sites (P = .58).[30]
Second-generation trials (postifosfamide)
Subsequent chemotherapy trials were performed using anthracycline and ifosfamide combinations in patients who primarily had extremity or truncal soft tissue sarcomas. The data are conflicting, and the issue is still not settled. In a small feasibility study, 59 patients with high-risk, soft tissue sarcomas, 58 of whom had an extremity or the trunk as the primary site, underwent primary resection plus PORT and were randomly assigned to observation versus a dose-dense regimen of six 14-day courses of ifosfamide, dacarbazine (DTIC), and doxorubicin (IFADIC regimen) with granulocyte colony-stimulating factor (G-CSF) bone marrow support and mesna uroprotection.[31] There were no statistically significant differences in OS or relapse-free survival (RFS), but the study was severely underpowered.
In a second trial performed by the Italian National Council for Research, high-risk patients were treated with local therapy (i.e., wide resection plus preRT or PORT, or amputation as clinically necessary) and were then randomly assigned to observation versus five 21-day cycles of 4-epidoxorubicin (epirubicin) plus ifosfamide (with mesna and G-CSF).[28,32] Based on power calculations, the planned study size was 190 patients, but the trial was stopped after 104 patients had been entered because an interim analysis revealed a statistically significant (P = .001) difference in DFS favoring the chemotherapy arm. By the time of the initial peer-reviewed report of the study, the DFS still favored the chemotherapy group (median DFS of 48 months vs. 16 months), but the P value had risen to .04.[28]
Although there was no difference in metastasis-free survival at the time of the report, there was an improvement in median OS (75 months vs. 46 months, P = .03). However, at the follow-up report (at a median of 89.6 months in a range of 56–119 months), OS differences were no longer statistically significant (58.5% vs. 43.1% [P = .07]). The DFS difference had also lost statistical significance (47.2% vs. 16.0% [P = .09]).[32] In summary, the trial was underpowered because it was stopped early, and the early promising results that led to stopping the trial diminished as the trial matured.
In a third, underpowered, single-center trial, 88 patients with high-risk, soft tissue sarcomas (64 of whom had extremity or truncal primary tumors) underwent surgery (with or without radiation) and were then randomly assigned to receive four 21-day cycles of chemotherapy (epirubicin [n = 26] or epirubicin plus ifosfamide [n = 19]) versus no adjuvant chemotherapy (n = 43).[33] The trial was closed prematurely because of a slow accrual rate. After a median follow-up of 94 months, the 5-year DFS in the chemotherapy and control arms was 69% versus 44%, respectively (P = .01); the 5-year OS rates were 72% versus 47% (P = .06). All of the benefit associated with chemotherapy appeared restricted to the 19 patients who received epirubicin plus ifosfamide.
In yet another underpowered trial, 137 patients with high-risk, soft tissue sarcomas (93% with extremity or truncal primary tumors) who met the eligibility criteria were randomly assigned to undergo surgical resection (with or without radiation) or to receive three preoperative 21-day cycles of doxorubicin plus ifosfamide.[34] This multicenter European Organization for Research and Treatment of Cancer trial (EORTC-62874) was closed because of slow accrual and results that were not promising enough to continue. With a median follow-up of 7.3 years, the 5-year DFS in the surgery alone and chemotherapy plus surgery arms was 52% and 56%, respectively (P = .35); and OS was 64% and 65%, respectively (P = .22).
These last four trials have been combined with the 14 first-generation trials in a trial-level meta-analysis.[35] Of the 18 randomized trials of patients with resectable soft tissue sarcomas, five trials used a combination of doxorubicin (50–90 mg/m2 per cycle) plus ifosfamide (1,500–5,000 mg/m2 per cycle). The remaining 13 trials used doxorubicin (50–70 mg/m2 per cycle) alone or with other drugs. The absolute risk reduction in local recurrence rates associated with any chemotherapy added to local therapy was 4 percentage points (95% CI, 0%–7%), and it was 5 percentage points (95% CI, 1%–12%) when ifosfamide was combined with doxorubicin. The absolute reduction in overall mortality was 6 percentage points with any chemotherapy (95% CI, 2%–11%; [i.e., a reduction from 46%–40%]), 11 percentage points for doxorubicin plus ifosfamide (95% CI, 3%–19%; [i.e., a reduction from 41%–30%]), and 5 percentage points for doxorubicin without ifosfamide.[35][Level of evidence: 1iiA]
An additional multicenter randomized trial (EORTC-62931 [NCT00002641]), the largest trial reported to date using adjuvant doxorubicin (75 mg/m2) plus ifosfamide (5,000 mg/m2), was subsequently published in abstract form and was not included in the above meta-analysis.[36] The results differed from those reported in the meta-analysis.[35] After local therapy, 351 patients were randomly assigned to five 21-day cycles of adjuvant therapy versus observation. The trial was stopped for futility because the 5-year RFS was 52% in both arms. OS was 64% in the chemotherapy arm versus 69% in the observation arm. In a subsequent abstract, the EORTC investigators reported a combined analysis of this trial and their previous trial (EORTC-62771) [37] of adjuvant cyclophosphamide plus doxorubicin plus DTIC (CYVADIC), representing the two largest trials of adjuvant therapy for adult soft tissue sarcoma in the literature.[38] The combined analysis showed no improvement in either RFS or OS associated with adjuvant chemotherapy.[38][Level of evidence: 1iiA]
In summary, the impact of adjuvant chemotherapy on survival is not clear but is likely to be small in absolute magnitude. Therefore, in discussions with a patient, any potential benefits should be considered in the context of the short- and long-term toxicities of the chemotherapy.
Role of regional hyperthermia
The use of regional hyperthermia to enhance the local effects of systemic chemotherapy in the neoadjuvant and adjuvant setting is under investigation. In a multicenter phase III trial, 341 patients with high-risk (tumor ≥5 cm, grade 2–3, and deep to fascia), soft tissue sarcomas (149 extremity tumors and 192 nonextremity tumors) were randomly allocated to receive four 21-day cycles of chemotherapy (etoposide 125 mg/m2 on days 1 and 4; ifosfamide 1,500 mg/m2 on days 1–4; doxorubicin 50 mg/m2 on day 1) with or without regional hyperthermia both before and after local therapy.[39] Approximately 11% of the patients were being treated for recurrent tumors. The regional hyperthermia was designed to produce tumor temperatures of 42°C for 60 minutes and was given on days 1 and 4 of each chemotherapy cycle. After the first four cycles of chemotherapy, definitive surgical excision of the tumor was performed, if possible, followed by radiation therapy, if indicated (i.e., a 52.7 Gy median dose delivered), and then the last four cycles of chemotherapy plus or minus hyperthermia. Three of the nine treatment centers with particular expertise in hyperthermia treated 91% of the patients in the trial.
The median duration of follow-up was 34 months. Local progression occurred in 56 patients in the hyperthermia group and 76 patients in the control group. The relative HR for local progression or death was 0.58 (95% CI, 0.41–0.84), with an absolute difference at 2 years of 15% (76% vs. 61%; 95% CI of the difference 6–26). The decreased risk of local progression or death was seen in both extremity and nonextremity tumors. However, hyperthermia had no effect on distant failure rates nor was there a statistically significant effect on OS (HR, .88, 95% CI, 0.64–1.21; P = .43).[39][Level of evidence: 1iiDiii] There was a higher rate of grade 3 to 4 leucopenia in the hyperthermia group: 77.6% versus 63.5% (P = .005). Since a large proportion of the patients were treated at centers with special expertise, there is no certainty that the finding can be generalized to apply to other settings.
Role of isolated limb perfusion
Isolated limb perfusion is under investigation as a means to deliver high doses of chemotherapy and permit limb salvage in unresectable primary or recurrent extremity soft tissue sarcomas that would otherwise require amputation, in the opinion of the surgeon.[40,41] Common drugs used in the procedure are TNF-alpha, melphalan, and interferon-gamma. Experience is limited to case series with response rates and reported avoidance of amputation as the outcome.[40,41][Level of evidence: 3iiiDiv] The technique requires specialized expertise to avoid severe local and systemic toxicity including systemic effects of TNF-alpha. The technique has not been directly compared with standard approaches using combined systemic and local therapy.
Role of chemotherapy for advanced disease
Doxorubicin is a mainstay of systemic therapy in the management of locally advanced and metastatic soft tissue sarcoma. Pegylated liposomal encapsulated doxorubicin is a formulation of doxorubicin designed to prolong the half-life of circulating doxorubicin and slow the release of active drugs.[42] The changed pharmacokinetics result in less myelosuppression and possibly less cardiotoxic effects, but there is a substantial incidence of hypersensitivity-like reactions and hand-foot syndrome. Its clinical activity relative to unencapsulated doxorubicin is not clear.[42][Level of evidence: 3iiiDiv] Other drugs that are thought to have clinical activity as single agents are ifosfamide, epirubicin, gemcitabine, and paclitaxel.[43-46][Level of Evidence: 3iiiDiv] Their clinical activity relative to single-agent doxorubicin is not clear, and they are not known to have superior activity.
There is controversy about the clinical benefit of adding other drugs to doxorubicin as a single agent. A systematic evidence review and meta-analysis conducted by the Cochrane Collaboration summarized the eight randomized trials reported from 1976 to 1995.[47] No additional randomized trials had been reported or were known to be in progress between 1995 and the 2002 literature search. Single-agent doxorubicin had been compared with a variety of doxorubicin-containing combinations that included vincristine, vindesine, cyclophosphamide, streptozotocin, mitomycin-C, cisplatin, and/or ifosfamide. Combination regimens consistently caused more nausea and hematologic toxicity. However, the better response rates associated with combination therapy were marginal and depended on the statistical model used (fixed effects model ORresponse = 1.29; 95% CI, 1.03–1.60, P = .03; random effects model ORresp = 1.26; 95% CI, 0.96–1.67, P = .10) There was no statistically significant difference in the 1- (ORmortality = 0.87; 95% CI, 0.73–1.05, P = .14) or 2-year mortality rates (ORmortality = 0.84; 95% CI, 0.67–1.06, P = .13).
These results were very similar even when the analyses were restricted to the four trials that used DTIC and/or ifosfamide as part of the combination regimen with doxorubicin agents that were postulated to have greater activity than the others tested. A subsequent meta-analysis of all three published randomized trials of chemotherapy regimens that contained ifosfamide versus those that did not came to similar conclusions: tumor response rates were better when the regimen included ifosfamide (RRresponse = 1.52; 95% CI, 1.11–2.08), but mortality at 1 year was not (RRmortality = 0.98; 95% CI, 0.85–1.13).[48][Level of evidence: 1iiDiv]. Therefore, response rate was a poor surrogate for OS. Quality-of-life outcomes were not reported in any of the above-mentioned randomized trials, but toxicity was worse when agents were added to doxorubicin.
References
- Pisters PW, Pollock RE, Lewis VO, et al.: Long-term results of prospective trial of surgery alone with selective use of radiation for patients with T1 extremity and trunk soft tissue sarcomas. Ann Surg 246 (4): 675-81; discussion 681-2, 2007. [PUBMED Abstract]
- Lohman RF, Nabawi AS, Reece GP, et al.: Soft tissue sarcoma of the upper extremity: a 5-year experience at two institutions emphasizing the role of soft tissue flap reconstruction. Cancer 94 (8): 2256-64, 2002. [PUBMED Abstract]
- Rosenberg SA, Tepper J, Glatstein E, et al.: The treatment of soft-tissue sarcomas of the extremities: prospective randomized evaluations of (1) limb-sparing surgery plus radiation therapy compared with amputation and (2) the role of adjuvant chemotherapy. Ann Surg 196 (3): 305-15, 1982. [PUBMED Abstract]
- O'Byrne K, Steward WP: The role of adjuvant chemotherapy in the treatment of adult soft tissue sarcomas. Crit Rev Oncol Hematol 27 (3): 221-7, 1998. [PUBMED Abstract]
- Singer S, Nielsen T, Antonescu CR: Molecular biology of soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1522-32.
- Singer S, Maki RG, O'Sullivan B: Soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1533-77.
- Malawer MM, Helman LJ, O'Sullivan B: Sarcomas of bone. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1578-1609.
- Lewis JJ, Leung D, Woodruff JM, et al.: Retroperitoneal soft-tissue sarcoma: analysis of 500 patients treated and followed at a single institution. Ann Surg 228 (3): 355-65, 1998. [PUBMED Abstract]
- van Geel AN, Pastorino U, Jauch KW, et al.: Surgical treatment of lung metastases: The European Organization for Research and Treatment of Cancer-Soft Tissue and Bone Sarcoma Group study of 255 patients. Cancer 77 (4): 675-82, 1996. [PUBMED Abstract]
- Casson AG, Putnam JB, Natarajan G, et al.: Five-year survival after pulmonary metastasectomy for adult soft tissue sarcoma. Cancer 69 (3): 662-8, 1992. [PUBMED Abstract]
- Putnam JB Jr, Roth JA: Surgical treatment for pulmonary metastases from sarcoma. Hematol Oncol Clin North Am 9 (4): 869-87, 1995. [PUBMED Abstract]
- Yang JC, Chang AE, Baker AR, et al.: Randomized prospective study of the benefit of adjuvant radiation therapy in the treatment of soft tissue sarcomas of the extremity. J Clin Oncol 16 (1): 197-203, 1998. [PUBMED Abstract]
- O'Sullivan B, Davis AM, Turcotte R, et al.: Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: a randomised trial. Lancet 359 (9325): 2235-41, 2002. [PUBMED Abstract]
- O'Sullivan B, Davis A, Turcotte R, et al.: Five-year results of a randomized phase III trial of pre-operative vs post-operative radiotherapy in extremity soft tissue sarcoma. [Abstract] J Clin Oncol 22 (Suppl 14): A-9007, 819s, 2004.
- Davis AM, O'Sullivan B, Turcotte R, et al.: Late radiation morbidity following randomization to preoperative versus postoperative radiotherapy in extremity soft tissue sarcoma. Radiother Oncol 75 (1): 48-53, 2005. [PUBMED Abstract]
- Griffin AM, Dickie CI, Catton CN, et al.: The influence of time interval between preoperative radiation and surgical resection on the development of wound healing complications in extremity soft tissue sarcoma. Ann Surg Oncol 22 (9): 2824-30, 2015. [PUBMED Abstract]
- Pisters PW, Harrison LB, Leung DH, et al.: Long-term results of a prospective randomized trial of adjuvant brachytherapy in soft tissue sarcoma. J Clin Oncol 14 (3): 859-68, 1996. [PUBMED Abstract]
- Alektiar KM, Zelefsky MJ, Brennan MF: Morbidity of adjuvant brachytherapy in soft tissue sarcoma of the extremity and superficial trunk. Int J Radiat Oncol Biol Phys 47 (5): 1273-9, 2000. [PUBMED Abstract]
- Alektiar KM, Brennan MF, Healey JH, et al.: Impact of intensity-modulated radiation therapy on local control in primary soft-tissue sarcoma of the extremity. J Clin Oncol 26 (20): 3440-4, 2008. [PUBMED Abstract]
- Alektiar KM, Brennan MF, Singer S: Local control comparison of adjuvant brachytherapy to intensity-modulated radiotherapy in primary high-grade sarcoma of the extremity. Cancer 117 (14): 3229-34, 2011. [PUBMED Abstract]
- Folkert MR, Singer S, Brennan MF, et al.: Comparison of local recurrence with conventional and intensity-modulated radiation therapy for primary soft-tissue sarcomas of the extremity. J Clin Oncol 32 (29): 3236-41, 2014. [PUBMED Abstract]
- Fabrizio PL, Stafford SL, Pritchard DJ: Extremity soft-tissue sarcomas selectively treated with surgery alone. Int J Radiat Oncol Biol Phys 48 (1): 227-32, 2000. [PUBMED Abstract]
- Rydholm A, Gustafson P, Rööser B, et al.: Limb-sparing surgery without radiotherapy based on anatomic location of soft tissue sarcoma. J Clin Oncol 9 (10): 1757-65, 1991. [PUBMED Abstract]
- Al-Refaie WB, Habermann EB, Jensen EH, et al.: Surgery alone is adequate treatment for early stage soft tissue sarcoma of the extremity. Br J Surg 97 (5): 707-13, 2010. [PUBMED Abstract]
- Rydholm A: Surgery without radiotherapy in soft tissue sarcoma. Acta Orthop Scand Suppl 273: 117-9, 1997. [PUBMED Abstract]
- Bagaria SP, Ashman JB, Daugherty LC, et al.: Compliance with National Comprehensive Cancer Network guidelines in the use of radiation therapy for extremity and superficial trunk soft tissue sarcoma in the United States. J Surg Oncol 109 (7): 633-8, 2014. [PUBMED Abstract]
- Kepka L, DeLaney TF, Suit HD, et al.: Results of radiation therapy for unresected soft-tissue sarcomas. Int J Radiat Oncol Biol Phys 63 (3): 852-9, 2005. [PUBMED Abstract]
- Frustaci S, Gherlinzoni F, De Paoli A, et al.: Adjuvant chemotherapy for adult soft tissue sarcomas of the extremities and girdles: results of the Italian randomized cooperative trial. J Clin Oncol 19 (5): 1238-47, 2001. [PUBMED Abstract]
- Adjuvant chemotherapy for localised resectable soft-tissue sarcoma of adults: meta-analysis of individual data. Sarcoma Meta-analysis Collaboration. Lancet 350 (9092): 1647-54, 1997. [PUBMED Abstract]
- Sarcoma Meta-analysis Collaboration (SMAC): Adjuvant chemotherapy for localised resectable soft tissue sarcoma in adults. Cochrane Database Syst Rev (4): CD001419, 2000. [PUBMED Abstract]
- Brodowicz T, Schwameis E, Widder J, et al.: Intensified Adjuvant IFADIC Chemotherapy for Adult Soft Tissue Sarcoma: A Prospective Randomized Feasibility Trial. Sarcoma 4 (4): 151-60, 2000. [PUBMED Abstract]
- Frustaci S, De Paoli A, Bidoli E, et al.: Ifosfamide in the adjuvant therapy of soft tissue sarcomas. Oncology 65 (Suppl 2): 80-4, 2003. [PUBMED Abstract]
- Petrioli R, Coratti A, Correale P, et al.: Adjuvant epirubicin with or without Ifosfamide for adult soft-tissue sarcoma. Am J Clin Oncol 25 (5): 468-73, 2002. [PUBMED Abstract]
- Gortzak E, Azzarelli A, Buesa J, et al.: A randomised phase II study on neo-adjuvant chemotherapy for 'high-risk' adult soft-tissue sarcoma. Eur J Cancer 37 (9): 1096-103, 2001. [PUBMED Abstract]
- Pervaiz N, Colterjohn N, Farrokhyar F, et al.: A systematic meta-analysis of randomized controlled trials of adjuvant chemotherapy for localized resectable soft-tissue sarcoma. Cancer 113 (3): 573-81, 2008. [PUBMED Abstract]
- Woll PJ, van Glabbeke M, Hohenberger P, et al.: Adjuvant chemotherapy (CT) with doxorubicin and ifosfamide in resected soft tissue sarcoma (STS): Interim analysis of a randomised phase III trial. [Abstract] J Clin Oncol 25 (Suppl 18): A-10008, 2007.
- Bramwell V, Rouesse J, Steward W, et al.: Adjuvant CYVADIC chemotherapy for adult soft tissue sarcoma--reduced local recurrence but no improvement in survival: a study of the European Organization for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group. J Clin Oncol 12 (6): 1137-49, 1994. [PUBMED Abstract]
- Le Cesne A, Van Glabbeke M, Woll PJ, et al.: The end of adjuvant chemotherapy (adCT) era with doxorubicin-based regimen in resected high-grade soft tissue sarcoma (STS): pooled analysis of the two STBSG-EORTC phase III clinical trials. [Abstract] J Clin Oncol 26 (Suppl 15): A-10525, 2008.
- Issels RD, Lindner LH, Verweij J, et al.: Neo-adjuvant chemotherapy alone or with regional hyperthermia for localised high-risk soft-tissue sarcoma: a randomised phase 3 multicentre study. Lancet Oncol 11 (6): 561-70, 2010. [PUBMED Abstract]
- Eggermont AM, de Wilt JH, ten Hagen TL: Current uses of isolated limb perfusion in the clinic and a model system for new strategies. Lancet Oncol 4 (7): 429-37, 2003. [PUBMED Abstract]
- Bonvalot S, Laplanche A, Lejeune F, et al.: Limb salvage with isolated perfusion for soft tissue sarcoma: could less TNF-alpha be better? Ann Oncol 16 (7): 1061-8, 2005. [PUBMED Abstract]
- Grenader T, Goldberg A, Hadas-Halperin I, et al.: Long-term response to pegylated liposomal doxorubicin in patients with metastatic soft tissue sarcomas. Anticancer Drugs 20 (1): 15-20, 2009. [PUBMED Abstract]
- Lorigan P, Verweij J, Papai Z, et al.: Phase III trial of two investigational schedules of ifosfamide compared with standard-dose doxorubicin in advanced or metastatic soft tissue sarcoma: a European Organisation for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group Study. J Clin Oncol 25 (21): 3144-50, 2007. [PUBMED Abstract]
- Nielsen OS, Dombernowsky P, Mouridsen H, et al.: High-dose epirubicin is not an alternative to standard-dose doxorubicin in the treatment of advanced soft tissue sarcomas. A study of the EORTC soft tissue and bone sarcoma group. Br J Cancer 78 (12): 1634-9, 1998. [PUBMED Abstract]
- Maki RG, Wathen JK, Patel SR, et al.: Randomized phase II study of gemcitabine and docetaxel compared with gemcitabine alone in patients with metastatic soft tissue sarcomas: results of sarcoma alliance for research through collaboration study 002 [corrected]. J Clin Oncol 25 (19): 2755-63, 2007. [PUBMED Abstract]
- Okuno S, Ryan LM, Edmonson JH, et al.: Phase II trial of gemcitabine in patients with advanced sarcomas (E1797): a trial of the Eastern Cooperative Oncology Group. Cancer 97 (8): 1969-73, 2003. [PUBMED Abstract]
- Bramwell VH, Anderson D, Charette ML, et al.: Doxorubicin-based chemotherapy for the palliative treatment of adult patients with locally advanced or metastatic soft tissue sarcoma. Cochrane Database Syst Rev (3): CD003293, 2003. [PUBMED Abstract]
- Verma S, Younus J, Stys-Norman D, et al.: Meta-analysis of ifosfamide-based combination chemotherapy in advanced soft tissue sarcoma. Cancer Treat Rev 34 (4): 339-47, 2008. [PUBMED Abstract]
Stage I Adult Soft Tissue Sarcoma
Refer to the Treatment Option Overview section of this summary for a more detailed discussion of the roles of surgery and radiation therapy.
Low-grade soft tissue sarcomas have little metastatic potential, but they have a propensity to recur locally. Accordingly, surgical excision with negative tissue margins of 1 cm to 2 cm or larger in all directions is the treatment of choice for patients with these early-stage sarcomas.[1-3] The Mohs surgical technique may be considered as an alternative to wide surgical excision for the very rare, small, well-differentiated primary sarcomas of the skin when cosmetic results are considered to be important, as margins can be assured with minimal normal tissue removal.[4]
Carefully executed high-dose radiation therapy using a shrinking-field technique may be beneficial for unresectable tumors or for resectable tumors in which a high likelihood of residual disease is thought to be present when margins are judged to be inadequate, and when wider resection would require either an amputation or the removal of a vital organ.[5] Because of the low metastatic potential of these tumors, chemotherapy is usually not given.[6,7]
Standard treatment options:
- Surgical excision of tumors 5 cm or smaller in diameter with negative tissue margins in all directions.[8-12]
- Surgical excision with preoperative radiation therapy (preRT) or postoperative radiation therapy (PORT). Radiation decreases the risk of local recurrence but has not been shown to increase overall survival.[13-16]
- If the tumor is unresectable, high-dose preRT may be used.[17]
- For tumors of the retroperitoneum, trunk, and head and neck, the following are options:
- Surgical resection with the option of PORT if negative margins cannot be obtained. Wide margins are unusual in these sites, and radiation therapy is usually advocated for trunk and head and neck primary sites.[18]
- PreRT followed by maximal surgical resection. Radiation therapy may be used in sarcomas of the trunk and head and neck to maximize local control because of the inability to obtain wide surgical margins.[19]
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- Singer S, Nielsen T, Antonescu CR: Molecular biology of soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1522-32.
- Singer S, Maki RG, O'Sullivan B: Soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1533-77.
- Malawer MM, Helman LJ, O'Sullivan B: Sarcomas of bone. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1578-1609.
- Fish FS: Soft tissue sarcomas in dermatology. Dermatol Surg 22 (3): 268-73, 1996. [PUBMED Abstract]
- Temple WJ, Temple CL, Arthur K, et al.: Prospective cohort study of neoadjuvant treatment in conservative surgery of soft tissue sarcomas. Ann Surg Oncol 4 (7): 586-90, 1997 Oct-Nov. [PUBMED Abstract]
- Sarcoma Meta-analysis Collaboration (SMAC): Adjuvant chemotherapy for localised resectable soft tissue sarcoma in adults. Cochrane Database Syst Rev (4): CD001419, 2000. [PUBMED Abstract]
- Pervaiz N, Colterjohn N, Farrokhyar F, et al.: A systematic meta-analysis of randomized controlled trials of adjuvant chemotherapy for localized resectable soft-tissue sarcoma. Cancer 113 (3): 573-81, 2008. [PUBMED Abstract]
- Al-Refaie WB, Habermann EB, Jensen EH, et al.: Surgery alone is adequate treatment for early stage soft tissue sarcoma of the extremity. Br J Surg 97 (5): 707-13, 2010. [PUBMED Abstract]
- Pisters PW, Pollock RE, Lewis VO, et al.: Long-term results of prospective trial of surgery alone with selective use of radiation for patients with T1 extremity and trunk soft tissue sarcomas. Ann Surg 246 (4): 675-81; discussion 681-2, 2007. [PUBMED Abstract]
- Fabrizio PL, Stafford SL, Pritchard DJ: Extremity soft-tissue sarcomas selectively treated with surgery alone. Int J Radiat Oncol Biol Phys 48 (1): 227-32, 2000. [PUBMED Abstract]
- Rydholm A, Gustafson P, Rööser B, et al.: Limb-sparing surgery without radiotherapy based on anatomic location of soft tissue sarcoma. J Clin Oncol 9 (10): 1757-65, 1991. [PUBMED Abstract]
- Rydholm A: Surgery without radiotherapy in soft tissue sarcoma. Acta Orthop Scand Suppl 273: 117-9, 1997. [PUBMED Abstract]
- Yang JC, Chang AE, Baker AR, et al.: Randomized prospective study of the benefit of adjuvant radiation therapy in the treatment of soft tissue sarcomas of the extremity. J Clin Oncol 16 (1): 197-203, 1998. [PUBMED Abstract]
- O'Sullivan B, Davis AM, Turcotte R, et al.: Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: a randomised trial. Lancet 359 (9325): 2235-41, 2002. [PUBMED Abstract]
- O'Sullivan B, Davis A, Turcotte R, et al.: Five-year results of a randomized phase III trial of pre-operative vs post-operative radiotherapy in extremity soft tissue sarcoma. [Abstract] J Clin Oncol 22 (Suppl 14): A-9007, 819s, 2004.
- Davis AM, O'Sullivan B, Turcotte R, et al.: Late radiation morbidity following randomization to preoperative versus postoperative radiotherapy in extremity soft tissue sarcoma. Radiother Oncol 75 (1): 48-53, 2005. [PUBMED Abstract]
- Kepka L, DeLaney TF, Suit HD, et al.: Results of radiation therapy for unresected soft-tissue sarcomas. Int J Radiat Oncol Biol Phys 63 (3): 852-9, 2005. [PUBMED Abstract]
- Brennan MF, Singer S, Maki RG: Sarcomas of the soft tissue and bone. In: DeVita VT Jr, Hellman S, Rosenberg SA, eds.: Cancer: Principles and Practice of Oncology. Vols. 1 & 2. 8th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2008, pp 1741-1833.
- Baldini EH, Wang D, Haas RL, et al.: Treatment Guidelines for Preoperative Radiation Therapy for Retroperitoneal Sarcoma: Preliminary Consensus of an International Expert Panel. Int J Radiat Oncol Biol Phys 92 (3): 602-12, 2015. [PUBMED Abstract]
Stage II and Node-Negative Stage III Adult Soft Tissue Sarcoma
Refer to the Treatment Option Overview section of this summary for a more detailed discussion of the roles of surgery, radiation therapy, and chemotherapy.
High-grade localized soft tissue sarcomas have an increased potential for local recurrence and metastasis. For sarcomas of the extremities, local control comparable to that obtained with amputation may be achieved with limb-sparing surgery that involves wide local excision in combination with preoperative radiation therapy (preRT) or postoperative radiation therapy (PORT).
Complete surgical resection is often difficult for sarcomas of the retroperitoneum because of their large size before detection and anatomical location.[1,2] As opposed to soft tissue sarcomas of the extremities, local recurrence is the most common cause of death in patients with retroperitoneal soft tissue sarcomas. Complete surgical resection (i.e., removal of the entire gross tumor) is the most important factor in preventing local recurrence and, in many instances, requires resection of adjacent viscera. For retroperitoneal sarcomas, retrospective comparison of surgery alone versus preRT review suggests that preRT is associated with improved local recurrence-free survival, but not disease-free survival.[3]
Standard treatment options:
- Surgical excision with preRT or PORT. Radiation decreases the risk of local recurrence but has not been shown to increase overall survival.[4-8]
- Surgical excision with negative tissue margins in all directions. This approach is generally restricted to low-grade tumors ( ≤5 cm in diameter) of the extremities or superficial trunk with microscopically negative surgical tumor margins.[9-13]
- If the tumor is unresectable, high-dose radiation therapy may be used, but poor local control is likely to result.[14]
- In some situations, radiation therapy and/or chemotherapy may be used before surgery in an attempt to convert a marginally resectable tumor to one that can be adequately resected with limb preservation; this treatment may be followed by PORT.
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- Heslin MJ, Lewis JJ, Nadler E, et al.: Prognostic factors associated with long-term survival for retroperitoneal sarcoma: implications for management. J Clin Oncol 15 (8): 2832-9, 1997. [PUBMED Abstract]
- Jaques DP, Coit DG, Hajdu SI, et al.: Management of primary and recurrent soft-tissue sarcoma of the retroperitoneum. Ann Surg 212 (1): 51-9, 1990. [PUBMED Abstract]
- Kelly KJ, Yoon SS, Kuk D, et al.: Comparison of Perioperative Radiation Therapy and Surgery Versus Surgery Alone in 204 Patients With Primary Retroperitoneal Sarcoma: A Retrospective 2-Institution Study. Ann Surg 262 (1): 156-62, 2015. [PUBMED Abstract]
- Yang JC, Chang AE, Baker AR, et al.: Randomized prospective study of the benefit of adjuvant radiation therapy in the treatment of soft tissue sarcomas of the extremity. J Clin Oncol 16 (1): 197-203, 1998. [PUBMED Abstract]
- Rosenberg SA, Tepper J, Glatstein E, et al.: The treatment of soft-tissue sarcomas of the extremities: prospective randomized evaluations of (1) limb-sparing surgery plus radiation therapy compared with amputation and (2) the role of adjuvant chemotherapy. Ann Surg 196 (3): 305-15, 1982. [PUBMED Abstract]
- O'Sullivan B, Davis AM, Turcotte R, et al.: Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: a randomised trial. Lancet 359 (9325): 2235-41, 2002. [PUBMED Abstract]
- O'Sullivan B, Davis A, Turcotte R, et al.: Five-year results of a randomized phase III trial of pre-operative vs post-operative radiotherapy in extremity soft tissue sarcoma. [Abstract] J Clin Oncol 22 (Suppl 14): A-9007, 819s, 2004.
- Davis AM, O'Sullivan B, Turcotte R, et al.: Late radiation morbidity following randomization to preoperative versus postoperative radiotherapy in extremity soft tissue sarcoma. Radiother Oncol 75 (1): 48-53, 2005. [PUBMED Abstract]
- Al-Refaie WB, Habermann EB, Jensen EH, et al.: Surgery alone is adequate treatment for early stage soft tissue sarcoma of the extremity. Br J Surg 97 (5): 707-13, 2010. [PUBMED Abstract]
- Pisters PW, Pollock RE, Lewis VO, et al.: Long-term results of prospective trial of surgery alone with selective use of radiation for patients with T1 extremity and trunk soft tissue sarcomas. Ann Surg 246 (4): 675-81; discussion 681-2, 2007. [PUBMED Abstract]
- Fabrizio PL, Stafford SL, Pritchard DJ: Extremity soft-tissue sarcomas selectively treated with surgery alone. Int J Radiat Oncol Biol Phys 48 (1): 227-32, 2000. [PUBMED Abstract]
- Rydholm A, Gustafson P, Rööser B, et al.: Limb-sparing surgery without radiotherapy based on anatomic location of soft tissue sarcoma. J Clin Oncol 9 (10): 1757-65, 1991. [PUBMED Abstract]
- Rydholm A: Surgery without radiotherapy in soft tissue sarcoma. Acta Orthop Scand Suppl 273: 117-9, 1997. [PUBMED Abstract]
- Kepka L, DeLaney TF, Suit HD, et al.: Results of radiation therapy for unresected soft-tissue sarcomas. Int J Radiat Oncol Biol Phys 63 (3): 852-9, 2005. [PUBMED Abstract]
Advanced Stage III (N1) Adult Soft Tissue Sarcoma
Refer to the Treatment Option Overview section of this summary for a more detailed discussion of the roles of surgery, radiation therapy, and chemotherapy.
Regional lymph node involvement by soft tissue sarcomas of adulthood is very infrequent. However, sarcoma types that more commonly spread to lymph nodes include high-grade rhabdomyosarcoma, vascular sarcomas, and epithelioid sarcomas.[1]
Standard treatment options:
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- Mazeron JJ, Suit HD: Lymph nodes as sites of metastases from sarcomas of soft tissue. Cancer 60 (8): 1800-8, 1987. [PUBMED Abstract]
- Watson DI, Coventry BJ, Langlois SL, et al.: Soft-tissue sarcoma of the extremity. Experience with limb-sparing surgery. Med J Aust 160 (7): 412-6, 1994. [PUBMED Abstract]
- Cormier JN, Huang X, Xing Y, et al.: Cohort analysis of patients with localized, high-risk, extremity soft tissue sarcoma treated at two cancer centers: chemotherapy-associated outcomes. J Clin Oncol 22 (22): 4567-74, 2004. [PUBMED Abstract]
- O'Byrne K, Steward WP: The role of adjuvant chemotherapy in the treatment of adult soft tissue sarcomas. Crit Rev Oncol Hematol 27 (3): 221-7, 1998. [PUBMED Abstract]
- Adjuvant chemotherapy for localised resectable soft-tissue sarcoma of adults: meta-analysis of individual data. Sarcoma Meta-analysis Collaboration. Lancet 350 (9092): 1647-54, 1997. [PUBMED Abstract]
Stage IV Adult Soft Tissue Sarcoma
Refer to the Treatment Option Overview section of this summary for a more detailed discussion of the roles of surgery, radiation therapy, and chemotherapy.
In the setting of lung metastasis, resection of metastatic tumors may be associated with long-term disease-free survival in patients selected for optimal underlying disease biology (i.e., patients with a limited number of metastases and slow tumor growth).[1-3] It is not clear to what degree the favorable outcomes are attributable to the efficacy of surgery or to careful selection of patients based upon factors that are associated with less-virulent disease.[1-3] The value of resection of hepatic metastases is unclear.
As noted in the Treatment Option Overview section above, doxorubicin is the standard systemic therapy in the management of metastatic sarcomas.[4,5] Other drugs that may have clinical activity as single agents are ifosfamide, epirubicin, gemcitabine, and paclitaxel.[6-9] Their clinical activity relative to single-agent doxorubicin is not clear, and they are not known to have superior activity. There is controversy about whether adding drugs to doxorubicin offers clinical benefit beyond what is achieved by doxorubicin as a single agent. To avoid severe toxicity in older patients, sequential use of single agents may be the preferred strategy for palliation.
A randomized study assessed whether dose intensification of doxorubicin with ifosfamide improved the survival of patients with advanced soft-tissue sarcoma compared with doxorubicin alone.[10] Two hundred twenty-eight patients were randomly assigned to receive doxorubicin, and 227 patients were randomly assigned to receive doxorubicin and ifosfamide. Median follow-up was 56 months (interquartile range [IQR], 31–77) in the doxorubicin-only group and 59 months (IQR, 36–72) in the combination group.
There was no significant difference in overall survival (OS) between groups (median OS, 12.8 months; 95.5% confidence interval [CI], 10.5–14.3 in the doxorubicin-alone group vs. 14.3 months; range, 12.5–16.5 months in the doxorubicin and ifosfamide group; hazard ratio [HR], 0.83; 95.5% CI 0.67–1.03; stratified log-rank test P = .076). Median progression-free survival was significantly higher for the doxorubicin and ifosfamide group (7.4 months; 95% CI, 6.6-8.3) than for the doxorubicin-alone group (4.6 months; range, 2.9–5.6 months; HR, 0.74; 95% CI, 0.60–0.90; stratified log-rank test P = .003). More patients in the doxorubicin and ifosfamide group than in the doxorubicin-alone group had an overall response (60 [26%] of 227 patients vs. 31 [14%] of 228; P < .0006). The most common grade 3 and 4 toxic effects, which were all more common with doxorubicin and ifosfamide than with doxorubicin alone, were leucopenia (97 [43%] of 224 patients vs. 40 [18%] of 223 patients), neutropenia (93 [42%] vs. 83 [37%]), febrile neutropenia (103 (46%) vs. 30 [13%]), anemia (78 [35%] vs. 10 [5%]), and thrombocytopenia (75 [33%]) vs. 1 [<1%]).[10][Level of evidence: 1iiA] Treatment intensification with doxorubicin and ifosfamide for palliation of advanced soft tissue sarcoma is not indicated.
Standard treatment options
- Chemotherapy.
- Single-agent chemotherapy, with subsequent single agents for disease regrowth.[4-6,8,9,11] Doxorubicin is generally the first-line agent. Ifosfamide also has substantial single-agent activity.
- Doxorubicin-based combination chemotherapy. A variety of regimens have been used, but none has been proven to increase OS compared with doxorubicin alone.[4,5] There is some evidence that the addition of ifosfamide increases response rates (but not survival). Toxicity is increased with the addition of drugs to doxorubicin. No quality-of-life studies have been reported in comparisons of single-agent therapy versus combination therapy.
- Resection of pulmonary lesions may be performed if the primary tumor is under control.[1-3]
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- van Geel AN, Pastorino U, Jauch KW, et al.: Surgical treatment of lung metastases: The European Organization for Research and Treatment of Cancer-Soft Tissue and Bone Sarcoma Group study of 255 patients. Cancer 77 (4): 675-82, 1996. [PUBMED Abstract]
- Casson AG, Putnam JB, Natarajan G, et al.: Five-year survival after pulmonary metastasectomy for adult soft tissue sarcoma. Cancer 69 (3): 662-8, 1992. [PUBMED Abstract]
- Putnam JB Jr, Roth JA: Surgical treatment for pulmonary metastases from sarcoma. Hematol Oncol Clin North Am 9 (4): 869-87, 1995. [PUBMED Abstract]
- Bramwell VH, Anderson D, Charette ML, et al.: Doxorubicin-based chemotherapy for the palliative treatment of adult patients with locally advanced or metastatic soft tissue sarcoma. Cochrane Database Syst Rev (3): CD003293, 2003. [PUBMED Abstract]
- Verma S, Younus J, Stys-Norman D, et al.: Meta-analysis of ifosfamide-based combination chemotherapy in advanced soft tissue sarcoma. Cancer Treat Rev 34 (4): 339-47, 2008. [PUBMED Abstract]
- Lorigan P, Verweij J, Papai Z, et al.: Phase III trial of two investigational schedules of ifosfamide compared with standard-dose doxorubicin in advanced or metastatic soft tissue sarcoma: a European Organisation for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group Study. J Clin Oncol 25 (21): 3144-50, 2007. [PUBMED Abstract]
- Nielsen OS, Dombernowsky P, Mouridsen H, et al.: High-dose epirubicin is not an alternative to standard-dose doxorubicin in the treatment of advanced soft tissue sarcomas. A study of the EORTC soft tissue and bone sarcoma group. Br J Cancer 78 (12): 1634-9, 1998. [PUBMED Abstract]
- Maki RG, Wathen JK, Patel SR, et al.: Randomized phase II study of gemcitabine and docetaxel compared with gemcitabine alone in patients with metastatic soft tissue sarcomas: results of sarcoma alliance for research through collaboration study 002 [corrected]. J Clin Oncol 25 (19): 2755-63, 2007. [PUBMED Abstract]
- Okuno S, Ryan LM, Edmonson JH, et al.: Phase II trial of gemcitabine in patients with advanced sarcomas (E1797): a trial of the Eastern Cooperative Oncology Group. Cancer 97 (8): 1969-73, 2003. [PUBMED Abstract]
- Judson I, Verweij J, Gelderblom H, et al.: Doxorubicin alone versus intensified doxorubicin plus ifosfamide for first-line treatment of advanced or metastatic soft-tissue sarcoma: a randomised controlled phase 3 trial. Lancet Oncol 15 (4): 415-23, 2014. [PUBMED Abstract]
- Grenader T, Goldberg A, Hadas-Halperin I, et al.: Long-term response to pegylated liposomal doxorubicin in patients with metastatic soft tissue sarcomas. Anticancer Drugs 20 (1): 15-20, 2009. [PUBMED Abstract]
Recurrent Adult Soft Tissue Sarcoma
Treatment of patients with recurrent soft tissue sarcoma depends on the type of initial presentation and treatment. Patients who develop a local recurrence often can be treated by local therapy: surgical excision plus radiation therapy after previous minimal therapy or amputation after previous aggressive treatment.[1-7] Resection of limited pulmonary metastases may be associated with favorable disease-free survival.[8-10][Level of evidence: 3iiiDiv] However, the contribution of selection factors, such as low tumor burden, slow tumor growth, and long disease-free interval, to these favorable outcomes is not known.
There is no standard chemotherapy for recurrent soft tissue sarcomas that have progressed after doxorubicin as a single agent or in combination with other agents that have clinical activity, such as ifosfamide, epirubicin, gemcitabine, and paclitaxel. Any of these agents not previously administered to the patient may be used sequentially at the time of recurrence or progression.[11-14][Level of Evidence: 3iiiDiv] None of these agents has been shown to increase overall survival in this setting, therefore, clinical trials are an appropriate option.
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- Singer S, Nielsen T, Antonescu CR: Molecular biology of soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1522-32.
- Singer S, Maki RG, O'Sullivan B: Soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1533-77.
- Malawer MM, Helman LJ, O'Sullivan B: Sarcomas of bone. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1578-1609.
- Midis GP, Pollock RE, Chen NP, et al.: Locally recurrent soft tissue sarcoma of the extremities. Surgery 123 (6): 666-71, 1998. [PUBMED Abstract]
- Essner R, Selch M, Eilber FR: Reirradiation for extremity soft tissue sarcomas. Local control and complications. Cancer 67 (11): 2813-7, 1991. [PUBMED Abstract]
- Singer S, Antman K, Corson JM, et al.: Long-term salvageability for patients with locally recurrent soft-tissue sarcomas. Arch Surg 127 (5): 548-53; discussion 553-4, 1992. [PUBMED Abstract]
- Lewis JJ, Leung D, Heslin M, et al.: Association of local recurrence with subsequent survival in extremity soft tissue sarcoma. J Clin Oncol 15 (2): 646-52, 1997. [PUBMED Abstract]
- van Geel AN, Pastorino U, Jauch KW, et al.: Surgical treatment of lung metastases: The European Organization for Research and Treatment of Cancer-Soft Tissue and Bone Sarcoma Group study of 255 patients. Cancer 77 (4): 675-82, 1996. [PUBMED Abstract]
- Casson AG, Putnam JB, Natarajan G, et al.: Five-year survival after pulmonary metastasectomy for adult soft tissue sarcoma. Cancer 69 (3): 662-8, 1992. [PUBMED Abstract]
- Putnam JB Jr, Roth JA: Surgical treatment for pulmonary metastases from sarcoma. Hematol Oncol Clin North Am 9 (4): 869-87, 1995. [PUBMED Abstract]
- Lorigan P, Verweij J, Papai Z, et al.: Phase III trial of two investigational schedules of ifosfamide compared with standard-dose doxorubicin in advanced or metastatic soft tissue sarcoma: a European Organisation for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group Study. J Clin Oncol 25 (21): 3144-50, 2007. [PUBMED Abstract]
- Nielsen OS, Dombernowsky P, Mouridsen H, et al.: High-dose epirubicin is not an alternative to standard-dose doxorubicin in the treatment of advanced soft tissue sarcomas. A study of the EORTC soft tissue and bone sarcoma group. Br J Cancer 78 (12): 1634-9, 1998. [PUBMED Abstract]
- Maki RG, Wathen JK, Patel SR, et al.: Randomized phase II study of gemcitabine and docetaxel compared with gemcitabine alone in patients with metastatic soft tissue sarcomas: results of sarcoma alliance for research through collaboration study 002 [corrected]. J Clin Oncol 25 (19): 2755-63, 2007. [PUBMED Abstract]
- Okuno S, Ryan LM, Edmonson JH, et al.: Phase II trial of gemcitabine in patients with advanced sarcomas (E1797): a trial of the Eastern Cooperative Oncology Group. Cancer 97 (8): 1969-73, 2003. [PUBMED Abstract]
Changes to This Summary (02/01/2018)
The PDQ cancer information summaries are reviewed regularly and updated as new information becomes available. This section describes the latest changes made to this summary as of the date above.
General Information About Adult Soft Tissue Sarcoma
Updated statistics with estimated new cases and deaths for 2018 (cited American Cancer Society as reference 1).
Stage Information for Adult Soft Tissue Sarcoma
An editorial change was made to this section.
This summary is written and maintained by the PDQ Adult Treatment Editorial Board, which is editorially independent of NCI. The summary reflects an independent review of the literature and does not represent a policy statement of NCI or NIH. More information about summary policies and the role of the PDQ Editorial Boards in maintaining the PDQ summaries can be found on the About This PDQ Summary and PDQ® - NCI's Comprehensive Cancer Database pages.
About This PDQ Summary
Purpose of This Summary
This PDQ cancer information summary for health professionals provides comprehensive, peer-reviewed, evidence-based information about the treatment of adult soft tissue sarcoma. It is intended as a resource to inform and assist clinicians who care for cancer patients. It does not provide formal guidelines or recommendations for making health care decisions.
Reviewers and Updates
This summary is reviewed regularly and updated as necessary by the PDQ Adult Treatment Editorial Board, which is editorially independent of the National Cancer Institute (NCI). The summary reflects an independent review of the literature and does not represent a policy statement of NCI or the National Institutes of Health (NIH).
Board members review recently published articles each month to determine whether an article should:
- be discussed at a meeting,
- be cited with text, or
- replace or update an existing article that is already cited.
Changes to the summaries are made through a consensus process in which Board members evaluate the strength of the evidence in the published articles and determine how the article should be included in the summary.
The lead reviewers for Adult Soft Tissue Sarcoma Treatment are:
- Russell S. Berman, MD (New York University School of Medicine)
- Minh Tam Truong, MD (Boston University Medical Center)
Any comments or questions about the summary content should be submitted to Cancer.gov through the NCI website's Email Us. Do not contact the individual Board Members with questions or comments about the summaries. Board members will not respond to individual inquiries.
Levels of Evidence
Some of the reference citations in this summary are accompanied by a level-of-evidence designation. These designations are intended to help readers assess the strength of the evidence supporting the use of specific interventions or approaches. The PDQ Adult Treatment Editorial Board uses a formal evidence ranking system in developing its level-of-evidence designations.
Permission to Use This Summary
PDQ is a registered trademark. Although the content of PDQ documents can be used freely as text, it cannot be identified as an NCI PDQ cancer information summary unless it is presented in its entirety and is regularly updated. However, an author would be permitted to write a sentence such as “NCI’s PDQ cancer information summary about breast cancer prevention states the risks succinctly: [include excerpt from the summary].”
The preferred citation for this PDQ summary is:
PDQ® Adult Treatment Editorial Board. PDQ Adult Soft Tissue Sarcoma Treatment. Bethesda, MD: National Cancer Institute. Updated <MM/DD/YYYY>. Available at: https://www.cancer.gov/types/soft-tissue-sarcoma/hp/adult-soft-tissue-treatment-pdq. Accessed <MM/DD/YYYY>. [PMID: 26389481]
Images in this summary are used with permission of the author(s), artist, and/or publisher for use within the PDQ summaries only. Permission to use images outside the context of PDQ information must be obtained from the owner(s) and cannot be granted by the National Cancer Institute. Information about using the illustrations in this summary, along with many other cancer-related images, is available in Visuals Online, a collection of over 2,000 scientific images.
Disclaimer
Based on the strength of the available evidence, treatment options may be described as either “standard” or “under clinical evaluation.” These classifications should not be used as a basis for insurance reimbursement determinations. More information on insurance coverage is available on Cancer.gov on the Managing Cancer Care page.
Contact Us
More information about contacting us or receiving help with the Cancer.gov website can be found on our Contact Us for Help page. Questions can also be submitted to Cancer.gov through the website’s Email Us.
Updated: February 1, 2018
Source URL: https://www.cancer.gov/publishedcontent/syndication/2127.htm
Source Agency: National Cancer Institute (NCI)
Captured Date: 2013-09-14 09:00:45.0
Adult Soft Tissue Sarcoma Treatment (PDQ®)–Health Professional Version
General Information About Adult Soft Tissue Sarcoma
Incidence and Mortality
Estimated new cases and deaths from soft tissue sarcoma in the United States in 2018:[1]
- New cases: 13,040.
- Deaths: 5,150.
Soft tissue sarcomas are malignant tumors that arise in any of the mesodermal tissues of the extremities (50%), trunk and retroperitoneum (40%), or head and neck (10%). The reported international incidence rates range from 1.8 to 5 per 100,000 individuals per year.[2]
Risk Factors and Genetic Factors
The risk of sporadic soft tissue sarcomas is increased by previous radiation therapy and, in the case of lymphangiosarcoma, by chronic lymphedema. The chemicals Thorotrast (thorium dioxide), vinyl chloride, and arsenic are also established carcinogens for hepatic angiosarcomas.[3-5]
Soft tissue sarcomas occur with greater frequency in patients with the following inherited syndromes:[3-5]
- Nevoid basal cell carcinoma syndrome (Gorlin syndrome: PTC gene mutation).
- Gardner syndrome (APC mutation).
- Li-Fraumeni syndrome (p53 mutation).
- Tuberous sclerosis (Bourneville disease: TSC1 or TSC2 mutation).
- von Recklinghausen disease (neurofibromatosis type 1: NF1 mutation).
- Werner syndrome (adult progeria: WRN mutation).
Diagnosis
Soft tissue sarcomas may be heterogeneous, so adequate tissue should be obtained via either core-needle or incisional biopsy for microscopic examination to determine histologic type and tumor grade. Careful planning of the initial biopsy is important to avoid compromising subsequent curative resection. Since the selection of treatment is determined by the grade of the tumor, it is essential to have a careful review of the biopsy tissue by a pathologist who is experienced in diagnosing sarcomas. Complete staging and treatment planning by a multidisciplinary team of cancer specialists is required to determine the optimal treatment for patients with this disease.
There is evidence that at least some favorable clinical outcomes may be associated with referral to a specialized sarcoma treatment center. In a population-based consecutive series of 375 soft tissue sarcoma patients in Sweden, local recurrence rates of resected tumors were higher in patients who were not referred to the specialized center: in 35 of 78 (45%) patients not referred; in 24 of 102 (24%) patients referred after initial surgery or incisional biopsy; and in 36 of 195 (18%) patients referred before any surgical procedure (P = .0001 for the difference between those never referred vs. those referred before any surgical procedure).[6][Level of evidence: 3iDii] However, there were no statistically significant differences in death from sarcoma between the groups of patients.
Prognostic Factors
The prognosis for patients with adult soft tissue sarcomas depends on several factors, including:[3-5,7,8]
- Patient’s age.
- Size, sarcoma subtype, histologic grade, mitotic activity, and stage of the tumor.
Factors associated with a poorer prognosis include the following:[9]
- Age older than 60 years.
- Tumors larger than 5 cm in greatest dimension.
- High-grade histology with high mitotic activity.
- Positive margins after resection.[10]
Although low-grade tumors are usually curable by surgery alone, higher-grade sarcomas (as determined by the mitotic index and by the presence of hemorrhage and necrosis) are associated with higher local-treatment failure rates and increased metastatic potential.
Surveillance for Relapse
A retrospective review included 174 consecutive patients with a soft tissue sarcoma of the limb who underwent follow-up by oncologists at a single center from 2003 to 2009.[11] The rate and site of recurrence and mode of detection were analyzed. Eighty-two patients (47%) experienced relapse. Isolated local recurrences occurred in 26 patients and local relapse with synchronous pulmonary metastases occurred in 5 patients. Local recurrences were detected clinically in 30 of the 31 patients; magnetic resonance imaging identified only one local recurrence. Twenty-eight patients developed isolated lung metastases; in 9 patients, the lung metastases were amenable to resections, 7 of whom were free of disease after treatment. Lung metastases were detected by chest x-ray in 19 patients, by computed tomography scanning in 3 patients, and clinically in 11 patients. Twenty-three patients developed nonpulmonary metastases. More than 80% of the relapses occurred in the first 2 years of follow-up; however, later recurrences were also observed.[11][Level of evidence: 3iiDi] This study supports imaging surveillance for detection of lung metastases, whereas local recurrences at the primary site were usually detected by clinical examination. The impact of picking up metastases from overall survival or quality-of-life data is unknown.
Related Summaries
Other PDQ summaries containing information about soft tissue sarcoma include:
References
- American Cancer Society: Cancer Facts and Figures 2018. Atlanta, Ga: American Cancer Society, 2018. Available online. Last accessed January 5, 2018.
- Wibmer C, Leithner A, Zielonke N, et al.: Increasing incidence rates of soft tissue sarcomas? A population-based epidemiologic study and literature review. Ann Oncol 21 (5): 1106-11, 2010. [PUBMED Abstract]
- Singer S, Nielsen T, Antonescu CR: Molecular biology of soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1522-32.
- Singer S, Maki RG, O'Sullivan B: Soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1533-77.
- Malawer MM, Helman LJ, O'Sullivan B: Sarcomas of bone. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1578-1609.
- Gustafson P, Dreinhöfer KE, Rydholm A: Soft tissue sarcoma should be treated at a tumor center. A comparison of quality of surgery in 375 patients. Acta Orthop Scand 65 (1): 47-50, 1994. [PUBMED Abstract]
- Coindre JM, Terrier P, Guillou L, et al.: Predictive value of grade for metastasis development in the main histologic types of adult soft tissue sarcomas: a study of 1240 patients from the French Federation of Cancer Centers Sarcoma Group. Cancer 91 (10): 1914-26, 2001. [PUBMED Abstract]
- Kasper B, Ouali M, van Glabbeke M, et al.: Prognostic factors in adolescents and young adults (AYA) with high risk soft tissue sarcoma (STS) treated by adjuvant chemotherapy: a study based on pooled European Organisation for Research and Treatment of Cancer (EORTC) clinical trials 62771 and 62931. Eur J Cancer 49 (2): 449-56, 2013. [PUBMED Abstract]
- Vraa S, Keller J, Nielsen OS, et al.: Prognostic factors in soft tissue sarcomas: the Aarhus experience. Eur J Cancer 34 (12): 1876-82, 1998. [PUBMED Abstract]
- Trovik LH, Ovrebo K, Almquist M, et al.: Adjuvant radiotherapy in retroperitoneal sarcomas. A Scandinavian Sarcoma Group study of 97 patients. Acta Oncol 53 (9): 1165-72, 2014. [PUBMED Abstract]
- Rothermundt C, Whelan JS, Dileo P, et al.: What is the role of routine follow-up for localised limb soft tissue sarcomas? A retrospective analysis of 174 patients. Br J Cancer 110 (10): 2420-6, 2014. [PUBMED Abstract]
Cellular Classification of Adult Soft Tissue Sarcoma
Soft tissue sarcomas are classified histologically according to the soft tissue cell of origin. Additional studies, including electron microscopy, specialized immunohistochemistry, flow cytometry, cytogenetics, and tissue culture studies may allow identification of particular subtypes within the major histologic categories. For example, S100 antigen suggests neural sheath origin, cytokeratin suggests epithelioid or synovial cell origin, and factor VIII-related antigen suggests endothelial origin. Likewise, some subtypes of sarcomas have characteristic genetic markers, but these markers are not generally used in the routine clinical setting (e.g., translocation t(X;18)(p11;q11) in synovial sarcomas and translocation t(12;16)(q13;p11) in myxoid and round-cell sarcomas).[1-3]
The histologic grade reflects the metastatic potential of these tumors more accurately than the classic cellular classification listed below. Pathologists assign a grade based on the number of mitoses per high-powered field, the presence of necrosis, cellular and nuclear morphology, and the degree of cellularity; discordance among expert pathologists regarding tumor grade, and even histologic subtype, can be substantial.[4]
The World Health Organization lists the following cell types in its classification of soft tissue sarcomas:[5,6]
- Adipocytic tumors.
- Dedifferentiated liposarcoma.*
- Myxoid/round cell liposarcoma.
- Pleomorphic liposarcoma.
- Fibroblastic/myofibroblastic tumors.
- Fibrosarcoma.**
- Myxofibrosarcoma, low grade.
- Low-grade fibromyxoid sarcoma.
- Sclerosing epithelioid fibrosarcoma.
- So-called fibrohistiocytic tumors.
- Undifferentiated pleomorphic sarcoma/malignant fibrous histiocytoma (including pleomorphic, giant cell, myxoid/high-grade myxofibrosarcoma, and inflammatory forms).
- Smooth muscle tumors.
- Leiomyosarcoma.
- Skeletal muscle tumors.
- Rhabdomyosarcoma (embryonal, alveolar, and pleomorphic forms).
- Vascular tumors.
- Epithelioid hemangioendothelioma.
- Angiosarcoma, deep.***
- Tumors of peripheral nerves.
- Malignant peripheral nerve sheath tumor.
- Chondro-osseous tumors.
- Extraskeletal chondrosarcoma (mesenchymal and other variants).
- Extraskeletal osteosarcoma.
- Tumors of uncertain differentiation.
- Synovial sarcoma.
- Epithelioid sarcoma.
- Alveolar soft part sarcoma.
- Clear cell sarcoma of soft tissue.
- Extraskeletal myxoid chondrosarcoma.
- Primitive neuroectodermal tumor/extraskeletal Ewing tumor.
- Desmoplastic small round cell tumor.
- Extrarenal rhabdoid tumor.
- Undifferentiated sarcoma; sarcoma, not otherwise specified.
[Note: *It is recognized that dedifferentiated liposarcoma primarily arises in the context of deep atypical lipomatous tumor/well-differentiated liposarcoma, a sarcoma of intermediate malignancy because of the lack of metastatic capacity. **The category of fibrosarcoma can be inclusive of fibrosarcomatous differentiation in dermatofibrosarcoma protuberans. ***Cutaneous angiosarcoma may be difficult to stage using the American Joint Committee on Cancer system. (Refer to the PDQ summary on Gastrointestinal Stromal Tumors for more information.)]
References
- Singer S, Nielsen T, Antonescu CR: Molecular biology of soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1522-32.
- Singer S, Maki RG, O'Sullivan B: Soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1533-77.
- Malawer MM, Helman LJ, O'Sullivan B: Sarcomas of bone. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1578-1609.
- Alvegård TA, Berg NO: Histopathology peer review of high-grade soft tissue sarcoma: the Scandinavian Sarcoma Group experience. J Clin Oncol 7 (12): 1845-51, 1989. [PUBMED Abstract]
- Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-6.
- Brodowicz T, Schwameis E, Widder J, et al.: Intensified Adjuvant IFADIC Chemotherapy for Adult Soft Tissue Sarcoma: A Prospective Randomized Feasibility Trial. Sarcoma 4 (4): 151-60, 2000. [PUBMED Abstract]
Stage Information for Adult Soft Tissue Sarcoma
Note: The American Joint Committee on Cancer (AJCC) has published the 8th edition of the AJCC Cancer Staging Manual, which includes revisions to the staging for this disease. Implementation of the 8th edition began in January 2018. The PDQ Adult Treatment Editorial Board, which maintains this summary, is reviewing the revised staging and will make appropriate changes as needed.
Staging has an important role in determining the most effective treatment for soft tissue sarcoma. Clinical staging involves magnetic resonance imaging (MRI) or computed tomography (CT) of the primary tumor area and a chest CT to look for metastasis to the lung (the most common site of distant spread). An abdominal CT scan is done in the case of retroperitoneal sarcomas because the liver may be the site of initial clinical metastasis for these tumors.
The stage is determined by the size of the tumor, the histologic grade, and whether there is spread to lymph nodes or distant sites. Intracompartmental or extracompartmental extension of extremity sarcomas is also important for surgical decision making. For complete staging, a thorough review of all biopsy specimens (including those from the primary tumor, lymph nodes, or other suspicious lesions) is essential. CT scan of the chest is recommended for sarcomas larger than 5 cm (T2) or with moderate to poor differentiation (grades 2–4). Nodal involvement is rare, occurring in fewer than 3% of patients with sarcoma.[1]
Lymph node involvement in soft tissue sarcomas of adulthood is rare but is somewhat more frequent in some subtypes (e.g., rhabdomyosarcoma, vascular sarcomas, clear cell sarcomas, and epithelioid sarcomas) when they are high grade.[2] Because treatment decisions are predicated on pathology staging, patients should be staged before, and again after, any neoadjuvant therapy. The assessment of tumor grade can be affected in either direction, but more frequently decreased because of differential cellular loss related to the neoadjuvant chemotherapy or radiation.[3] Grade, which is based on cellular differentiation, mitotic rate, and extent of necrosis, should be recorded for all soft tissue sarcomas. A three-grade system (G1–G3) is preferred. (See below.)
The AJCC has designated staging by the four criteria of tumor size, nodal status, metastasis, and grade (TNMG).[3] The characteristic molecular markers of some sarcomas are not formally incorporated in the staging system pending further evaluation of their impact on prognosis. Recurrent sarcomas are restaged using the same system as for primary tumors with the specification that the tumor is recurrent.
Definitions of TNM and Grade
| aReprinted with permission from AJCC: Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-8. | |
| bSuperficial tumor is located exclusively above the superficial fascia without invasion of the fascia; deep tumor is located either exclusively beneath the superficial fascia, superficial to the fascia with invasion of or through the fascia, or both superficial yet beneath the fascia. | |
| TX | Primary tumor cannot be assessed. |
| T0 | No evidence of primary tumor. |
| T1 | Tumor ≤5 cm in greatest dimension. (Size should be regarded as a continuous variable, and the measurement should be provided.) |
| T1a | Superficial tumor.b |
| T1b | Deep tumor.b |
| T2 | Tumor >5 cm in greatest dimension.b |
| T2a | Superficial tumor.b |
| T2b | Deep tumor. |
| aReprinted with permission from AJCC: Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-8. | |
| bPresence of positive nodes (N1) in M0 tumors is considered Stage III. | |
| NX | Regional lymph nodes cannot be assessed. |
| N0 | No regional lymph node metastasis. |
| N1b | Regional lymph node metastasis. |
| aReprinted with permission from AJCC: Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-8. | |
| M0 | No distant metastasis. |
| M1 | Distant metastasis. |
| aReprinted with permission from AJCC: Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-8. | ||||
| Stage IA | T1a | N0 | M0 | G1, GX |
| T1b | N0 | M0 | G1, GX | |
| Stage IB | T2a | N0 | M0 | G1, GX |
| T2b | N0 | M0 | G1, GX | |
| Stage IIA | T1a | N0 | M0 | G2, G3 |
| T1b | N0 | M0 | G2, G3 | |
| Stage IIB | T2a | N0 | M0 | G2 |
| T2b | N0 | M0 | G2 | |
| Stage III | T2a, T2b | N0 | M0 | G3 |
| Any T | N1 | M0 | Any G | |
| Stage IV | Any T | Any N | M1 | Any G |
Neurovascular and bone invasion are indicators of poor prognosis, but they are not incorporated into the formal staging system.
References
- Fong Y, Coit DG, Woodruff JM, et al.: Lymph node metastasis from soft tissue sarcoma in adults. Analysis of data from a prospective database of 1772 sarcoma patients. Ann Surg 217 (1): 72-7, 1993. [PUBMED Abstract]
- Mazeron JJ, Suit HD: Lymph nodes as sites of metastases from sarcomas of soft tissue. Cancer 60 (8): 1800-8, 1987. [PUBMED Abstract]
- Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-6.
Treatment Option Overview
Multimodality Approach
In most cases, a combined modality approach of preoperative radiation therapy (preRT) or postoperative radiation therapy (PORT) is used, rather than the radical surgical procedures, such as amputation, that were used in the past. It may even be possible to use surgery without PORT in selected cases. For example, a case series was reported from a specialized sarcoma treatment referral center in which 74 selected patients with primary extremity and trunk tumors 5 cm or smaller were found to have no histologic involvement of the surgical margins. The patients were observed without radiation therapy, and the estimated local recurrence rate after 10 years was 11%.[1][Level of evidence: 3iiiDiv] The role of chemotherapy is not as well defined as is the role for radiation therapy. Because of the evolving nature of the treatment options for this disease, patients should be considered when available. Information about ongoing clinical trials is available from the NCI website.
Role of Surgery
Surgical resection is the mainstay of therapy for soft tissue sarcomas. When feasible, wide-margin function–sparing surgical excision is the cornerstone of effective treatment for extremity tumors. This may be facilitated by soft tissue reconstructive surgery, which generally permits wider margins than those obtained when the surgical plan involves direct closure of the excision site.[2] Cutting into the tumor mass or shelling out the gross tumor along the plane of the pseudocapsule of compressed tumor cells and reactive tissue that often surrounds soft tissue sarcomas are associated with an elevated risk of local recurrence. Even high-grade, soft tissue sarcomas of the extremities can usually be effectively treated while preserving the limb with combined-modality treatment consisting of preRT or PORT to reduce local recurrence. (Refer to the Role of Radiation Therapy section of this summary for more information.)
Only one small, single-institution, randomized trial has directly compared amputation to limb-sparing surgery for soft tissue sarcomas of the extremities.[3] In a 2:1 randomization ratio, 27 patients with high-grade extremity sarcomas were assigned to a wide excision plus PORT (45 Gy–50 Gy to the wide local excision area, and a total of 60 Gy–70 Gy to the tumor bed over 6–7 weeks), and 16 were assigned to amputation at or above the joint proximal to the tumor. Both groups received adjuvant chemotherapy (i.e., doxorubicin, cyclophosphamide, and high-dose methotrexate). At 63 months, with a median follow-up of 56 months, there were four local recurrences in the 27 patients who underwent limb-sparing surgery and no recurrences in the 16 patients who underwent amputation P2 = .12. Overall survival (OS) rates were not statistically significantly different (actuarial 5-year survival rate, 83% vs. 88%, P2 = .99).[3][Level of evidence: 1iiA]
Local control of high-grade soft tissue sarcomas of the trunk and the head and neck can be achieved with surgery in combination with radiation therapy.[4] It may be possible to use surgery without PORT in selected cases. For example, a case series was reported from a specialized sarcoma treatment referral center in which 74 selected patients with primary extremity and trunk tumors 5 cm or smaller were found to have no histologic involvement of the surgical margins.[1] They were observed without radiation therapy, and the estimated local recurrence rate after 10 years was 11%.[1][Level of evidence: 3iiiDiv] The role of chemotherapy is not as well defined as is the role of radiation therapy. Because of the evolving nature of the treatment options for this disease, patients should be offered the option of clinical trials when available.
Effective treatment of retroperitoneal sarcomas requires removal of all gross disease while sparing adjacent viscera not invaded by tumor. The prognosis for patients with high-grade retroperitoneal sarcomas is less favorable than for patients with tumors at other sites, partly because of the difficulty in completely resecting these tumors and the dose-limiting toxicity of high-dose radiation therapy on visceral organs.[5-8]
In the setting of distant metastasis, surgery may be associated with long-term, disease-free survival in patients with pulmonary metastasis and optimal underlying disease biology (i.e., patients with a limited number of metastases and slow nodule growth) who have undergone or are undergoing complete resection of the primary tumor.[9-11] It is not clear to what degree the favorable outcomes are attributable to the efficacy of surgery or the careful selection of patients based on factors that are associated with less-virulent disease.
Role of Radiation Therapy
Radiation plays an important role in limb-sparing therapy. Pre- and postoperative external-beam radiation therapies (EBRT), as well as brachytherapy, have been shown to decrease the risk of local recurrence. They have not been shown to increase OS but are used to avoid amputation for all but the most locally advanced tumors or for limbs seriously compromised by vascular disease, where acceptable functional preservation is not possible. In the case of EBRT, irradiation of the entire limb circumference is avoided to preserve vascular and nerve structures that are critical to function and preservation of the limb.
PORT
PORT has been tested in a single-institution, randomized trial of 141 patients with extremity sarcomas who were treated with limb-sparing surgery. Patients with high-grade tumors (n = 91) also received adjuvant chemotherapy (i.e., five 28-day cycles of doxorubicin and cyclophosphamide). All patients were randomly assigned to receive radiation (45 Gy to a wide field, plus a tumor-bed boost of 18 Gy over 6–7 weeks), concurrent with chemotherapy in the case of high-grade tumors versus no radiation.[12] At up to 12 years of follow-up, there was one local recurrence in the 70 patients randomly assigned to receive radiation versus 17 recurrences in the 71 control patients (P = .0001), with similar reduction in risk of local recurrence for both high- and low-grade tumors. However, there was no difference in OS between the radiation and control groups.[12][Level of evidence: 1iiDiii] Global quality of life was similar in the two groups, but the radiation therapy group had substantially worse functional deficits resulting from reduced strength and joint motion as well as increased edema.
To limit acute toxicity with preRT, smaller fields and lower doses are generally given than is the case with PORT. PreRT has been directly compared with PORT for extremity soft tissue sarcomas in a multicenter randomized trial.[13-15] Designed to include 266 patients, the trial was stopped early after 190 patients had been accrued because of an increase in wound complications in the preRT group. The scheduled radiation in the preRT group was a wide field of 50 Gy in 2-Gy fractions (first phase of the trial) with an additional 16 Gy to 20 Gy to the tumor bed and a 2-cm margin (second phase of the trial) only if tumor cells were found at the surgical margins.
Patients in the PORT group were scheduled to receive radiation during both phases of the trial. The wound-complication rates were 35% versus 17% in the preRT and PORT groups, respectively (P = .01). In addition, limb function at 6 weeks after surgery was worse in the preRT group (P = .01).[13] At 5 years, the two groups had similar local control rates (93% vs. 92%) and OS (73% vs. 67%, P = .48).[14] Of the 129 patients evaluated for limb function at 21 to 27 months after surgery (n = 73 for preRT and n = 56 for PORT), limb function was similar in both groups, but there was a statistical trend for less fibrosis in the preRT group (P = .07).[15]
Brachytherapy
Brachytherapy has also been investigated as an adjuvant therapy for soft tissue sarcomas. Although it has possible advantages of convenience and less radiation to normal surrounding tissue relative to EBRT, the two treatment strategies have not been directly compared in terms of efficacy or morbidity. However, adjuvant brachytherapy has been compared with surgery without radiation. The time interval between preRT and surgical excision in extremity soft tissue sarcoma had minimal influence on the development of wound complications. Four- or 5-week intervals showed equivalent complication rates between patients who did or did not develop wound complications, suggesting an optimal interval to reduce potential complications.[16]
In a single-institution trial, 164 patients with sarcomas of the extremity or superficial trunk were randomly assigned during surgery, if all gross tumor could be excised, to receive an iridium Ir 192 implant (delivering 42 Gy–45 Gy over 4–6 days; 78 patients) or to a control arm of no radiation (86 patients).[17,18] Some of the patients with high-grade tumors received adjuvant doxorubicin-based chemotherapy if they were thought to be at a high risk for metastasis (34 patients in each study arm). With a median follow-up of 76 months, the 5-year actuarial local recurrence rates were 18% and 31% in the brachytherapy and control arms, respectively (P = .04). This difference was limited to patients with high-grade tumors. There was no discernible difference in sarcoma-specific survival rates between the brachytherapy and control arms (84% and 81%, respectively; P = .65), and there was no difference in the high tumor-grade group.[17][Level of evidence: 1iiDiii] The rates of clinically important wound complications (e.g., need for operative revision or repeated seroma drainage, wound separation, large hematomas, or purulent infection) were 24% and 14% in the radiation and control arms, respectively (P = .13); wound reoperation rates were 10% and 0%, respectively (P = .006).[18]
Intensity-modulated radiation therapy
Intensity-modulated radiation therapy (IMRT) has been used to deliver preRT or PORT to patients with extremity soft tissue sarcomas in an effort to spare the femur, joints, and selected other normal tissues from the full prescription dose and to maintain local control while potentially reducing radiation therapy-related morbidity. Initial single-institution reports suggest that high rates of local control with some reduction in morbidity are possible with this technique.[19,20] Retrospective comparison of IMRT compared with 3-dimensional, conformal radiation therapy demonstrates that local recurrence for primary soft tissue sarcomas of the extremity was worse in the non-IMRT group.[21][Level of evidence: 3iiiDiv]
Surgery and radiation therapy
In some tumors of the extremities or trunk, surgery alone can be performed without the use of radiation. Evidence for this approach is limited to single-institution, relatively small, case series [1,22,23] or analysis of outcomes in the National Cancer Institute's Surveillance, Epidemiology, and End Results (SEER) tumor registry.[24] However, these comparisons suffer from low statistical power and differential evaluability rates that could have introduced bias.[1] Patient selection factors may vary among surgeons. In general, this approach is considered in patients with low-grade tumors of the extremity or superficial trunk that are 5 cm or smaller in diameter (T1) and have microscopically negative surgical margins; long-term local tumor control is about 90% in such patients.[25]
A patterns-of-care study using SEER data was queried to identify patients undergoing surgery for truncal and extremity soft tissue sarcomas from 2004 to 2009.[26] Of 5,075 patients, 50% received radiation therapy. Radiation was considered to be underused in a significant portion of patients undergoing treatment for soft tissue sarcoma in the United States. Although routine radiation therapy is not recommended for stage I patients, 25% of them still underwent radiation. Even though routine radiation therapy is recommended for patients with stage II and III tumors, only 60% of them underwent radiation. On multivariate analysis, predictors of radiation therapy included age younger than 50 years (odds ratio [OR], 1.57; 95% confidence interval [CI], 1.28–1.91), malignant fibrous histiocytoma histology (OR, 1.47; 95% CI, 1.3–1.92), T2 classification (OR, 1.88; 95% CI, 1.60–2.20), and G3 (OR, 6.27; 95% CI, 5.10–7.72). Patients with stage III soft tissue sarcoma who received radiation therapy showed improved disease-specific survival at 5 years compared with those who did not (68% vs. 46%, P < .001).[26][Level of evidence: 3iDii]
On occasion, surgical excision cannot be performed in the initial management of soft tissue sarcomas because the morbidity would be unacceptable or nearby critical organs make complete resection impossible. In such circumstances, radiation has been used as the primary therapy.[27] However, this must be considered a treatment of last resort. Experience is limited to retrospective case series from single centers.[27][Level of evidence: 3iiiDiv]
Role of Adjuvant or Neoadjuvant Chemotherapy for Clinically Localized Tumors
The role of adjuvant chemotherapy is not completely clear. The investigation of its use falls into two categories or generations—pre- and post-ifosfamide regimens. In discussions with a patient, any potential benefits should be considered in the context of the short- and long-term toxicities of the chemotherapy.
First-generation trials (preifosfamide)
Several prospective, randomized trials were unable to determine conclusively whether doxorubicin-based adjuvant chemotherapy benefits adults with resectable soft tissue sarcomas. The majority of these studies accrued small numbers of patients and did not demonstrate a metastasis-free survival or an OS benefit for adjuvant chemotherapy.[4] A small study of adjuvant chemotherapy showed a positive effect on both disease-free survival (DFS) and OS in patients treated with postoperative chemotherapy.[28] There was wide interstudy variability among the reported trials, including differences in therapeutic regimens, drug doses, sample size, tumor site, and histologic grade.
A quantitative meta-analysis of updated data from 1,568 individual patients in 14 trials of doxorubicin-based adjuvant therapy showed an absolute benefit from adjuvant therapy of 6% for a local relapse-free interval (95% CI, 1%–10%), 10% for a distant relapse-free interval (95% CI, 5%–15%), and 10% for recurrence-free survival (95% CI, 5%–15%). A statistically significant OS benefit at 10 years was not detected: absolute difference 4% (95% CI, -1%–+9%).[29,30][Level of evidence: 1iiDii] However, only a small proportion of patients in this meta-analysis were treated with ifosfamide, an agent with demonstrated activity against soft tissue sarcoma. In addition, a subset analysis suggested that patients with sarcomas of the extremities may have benefited from adjuvant chemotherapy (hazard ratio [HR] for death, 0.8, P = .029), but there was no clear evidence that patients with extremity sarcomas had outcomes that were statistically significantly different from the outcomes of patients with tumors at other sites (P = .58).[30]
Second-generation trials (postifosfamide)
Subsequent chemotherapy trials were performed using anthracycline and ifosfamide combinations in patients who primarily had extremity or truncal soft tissue sarcomas. The data are conflicting, and the issue is still not settled. In a small feasibility study, 59 patients with high-risk, soft tissue sarcomas, 58 of whom had an extremity or the trunk as the primary site, underwent primary resection plus PORT and were randomly assigned to observation versus a dose-dense regimen of six 14-day courses of ifosfamide, dacarbazine (DTIC), and doxorubicin (IFADIC regimen) with granulocyte colony-stimulating factor (G-CSF) bone marrow support and mesna uroprotection.[31] There were no statistically significant differences in OS or relapse-free survival (RFS), but the study was severely underpowered.
In a second trial performed by the Italian National Council for Research, high-risk patients were treated with local therapy (i.e., wide resection plus preRT or PORT, or amputation as clinically necessary) and were then randomly assigned to observation versus five 21-day cycles of 4-epidoxorubicin (epirubicin) plus ifosfamide (with mesna and G-CSF).[28,32] Based on power calculations, the planned study size was 190 patients, but the trial was stopped after 104 patients had been entered because an interim analysis revealed a statistically significant (P = .001) difference in DFS favoring the chemotherapy arm. By the time of the initial peer-reviewed report of the study, the DFS still favored the chemotherapy group (median DFS of 48 months vs. 16 months), but the P value had risen to .04.[28]
Although there was no difference in metastasis-free survival at the time of the report, there was an improvement in median OS (75 months vs. 46 months, P = .03). However, at the follow-up report (at a median of 89.6 months in a range of 56–119 months), OS differences were no longer statistically significant (58.5% vs. 43.1% [P = .07]). The DFS difference had also lost statistical significance (47.2% vs. 16.0% [P = .09]).[32] In summary, the trial was underpowered because it was stopped early, and the early promising results that led to stopping the trial diminished as the trial matured.
In a third, underpowered, single-center trial, 88 patients with high-risk, soft tissue sarcomas (64 of whom had extremity or truncal primary tumors) underwent surgery (with or without radiation) and were then randomly assigned to receive four 21-day cycles of chemotherapy (epirubicin [n = 26] or epirubicin plus ifosfamide [n = 19]) versus no adjuvant chemotherapy (n = 43).[33] The trial was closed prematurely because of a slow accrual rate. After a median follow-up of 94 months, the 5-year DFS in the chemotherapy and control arms was 69% versus 44%, respectively (P = .01); the 5-year OS rates were 72% versus 47% (P = .06). All of the benefit associated with chemotherapy appeared restricted to the 19 patients who received epirubicin plus ifosfamide.
In yet another underpowered trial, 137 patients with high-risk, soft tissue sarcomas (93% with extremity or truncal primary tumors) who met the eligibility criteria were randomly assigned to undergo surgical resection (with or without radiation) or to receive three preoperative 21-day cycles of doxorubicin plus ifosfamide.[34] This multicenter European Organization for Research and Treatment of Cancer trial (EORTC-62874) was closed because of slow accrual and results that were not promising enough to continue. With a median follow-up of 7.3 years, the 5-year DFS in the surgery alone and chemotherapy plus surgery arms was 52% and 56%, respectively (P = .35); and OS was 64% and 65%, respectively (P = .22).
These last four trials have been combined with the 14 first-generation trials in a trial-level meta-analysis.[35] Of the 18 randomized trials of patients with resectable soft tissue sarcomas, five trials used a combination of doxorubicin (50–90 mg/m2 per cycle) plus ifosfamide (1,500–5,000 mg/m2 per cycle). The remaining 13 trials used doxorubicin (50–70 mg/m2 per cycle) alone or with other drugs. The absolute risk reduction in local recurrence rates associated with any chemotherapy added to local therapy was 4 percentage points (95% CI, 0%–7%), and it was 5 percentage points (95% CI, 1%–12%) when ifosfamide was combined with doxorubicin. The absolute reduction in overall mortality was 6 percentage points with any chemotherapy (95% CI, 2%–11%; [i.e., a reduction from 46%–40%]), 11 percentage points for doxorubicin plus ifosfamide (95% CI, 3%–19%; [i.e., a reduction from 41%–30%]), and 5 percentage points for doxorubicin without ifosfamide.[35][Level of evidence: 1iiA]
An additional multicenter randomized trial (EORTC-62931 [NCT00002641]), the largest trial reported to date using adjuvant doxorubicin (75 mg/m2) plus ifosfamide (5,000 mg/m2), was subsequently published in abstract form and was not included in the above meta-analysis.[36] The results differed from those reported in the meta-analysis.[35] After local therapy, 351 patients were randomly assigned to five 21-day cycles of adjuvant therapy versus observation. The trial was stopped for futility because the 5-year RFS was 52% in both arms. OS was 64% in the chemotherapy arm versus 69% in the observation arm. In a subsequent abstract, the EORTC investigators reported a combined analysis of this trial and their previous trial (EORTC-62771) [37] of adjuvant cyclophosphamide plus doxorubicin plus DTIC (CYVADIC), representing the two largest trials of adjuvant therapy for adult soft tissue sarcoma in the literature.[38] The combined analysis showed no improvement in either RFS or OS associated with adjuvant chemotherapy.[38][Level of evidence: 1iiA]
In summary, the impact of adjuvant chemotherapy on survival is not clear but is likely to be small in absolute magnitude. Therefore, in discussions with a patient, any potential benefits should be considered in the context of the short- and long-term toxicities of the chemotherapy.
Role of regional hyperthermia
The use of regional hyperthermia to enhance the local effects of systemic chemotherapy in the neoadjuvant and adjuvant setting is under investigation. In a multicenter phase III trial, 341 patients with high-risk (tumor ≥5 cm, grade 2–3, and deep to fascia), soft tissue sarcomas (149 extremity tumors and 192 nonextremity tumors) were randomly allocated to receive four 21-day cycles of chemotherapy (etoposide 125 mg/m2 on days 1 and 4; ifosfamide 1,500 mg/m2 on days 1–4; doxorubicin 50 mg/m2 on day 1) with or without regional hyperthermia both before and after local therapy.[39] Approximately 11% of the patients were being treated for recurrent tumors. The regional hyperthermia was designed to produce tumor temperatures of 42°C for 60 minutes and was given on days 1 and 4 of each chemotherapy cycle. After the first four cycles of chemotherapy, definitive surgical excision of the tumor was performed, if possible, followed by radiation therapy, if indicated (i.e., a 52.7 Gy median dose delivered), and then the last four cycles of chemotherapy plus or minus hyperthermia. Three of the nine treatment centers with particular expertise in hyperthermia treated 91% of the patients in the trial.
The median duration of follow-up was 34 months. Local progression occurred in 56 patients in the hyperthermia group and 76 patients in the control group. The relative HR for local progression or death was 0.58 (95% CI, 0.41–0.84), with an absolute difference at 2 years of 15% (76% vs. 61%; 95% CI of the difference 6–26). The decreased risk of local progression or death was seen in both extremity and nonextremity tumors. However, hyperthermia had no effect on distant failure rates nor was there a statistically significant effect on OS (HR, .88, 95% CI, 0.64–1.21; P = .43).[39][Level of evidence: 1iiDiii] There was a higher rate of grade 3 to 4 leucopenia in the hyperthermia group: 77.6% versus 63.5% (P = .005). Since a large proportion of the patients were treated at centers with special expertise, there is no certainty that the finding can be generalized to apply to other settings.
Role of isolated limb perfusion
Isolated limb perfusion is under investigation as a means to deliver high doses of chemotherapy and permit limb salvage in unresectable primary or recurrent extremity soft tissue sarcomas that would otherwise require amputation, in the opinion of the surgeon.[40,41] Common drugs used in the procedure are TNF-alpha, melphalan, and interferon-gamma. Experience is limited to case series with response rates and reported avoidance of amputation as the outcome.[40,41][Level of evidence: 3iiiDiv] The technique requires specialized expertise to avoid severe local and systemic toxicity including systemic effects of TNF-alpha. The technique has not been directly compared with standard approaches using combined systemic and local therapy.
Role of chemotherapy for advanced disease
Doxorubicin is a mainstay of systemic therapy in the management of locally advanced and metastatic soft tissue sarcoma. Pegylated liposomal encapsulated doxorubicin is a formulation of doxorubicin designed to prolong the half-life of circulating doxorubicin and slow the release of active drugs.[42] The changed pharmacokinetics result in less myelosuppression and possibly less cardiotoxic effects, but there is a substantial incidence of hypersensitivity-like reactions and hand-foot syndrome. Its clinical activity relative to unencapsulated doxorubicin is not clear.[42][Level of evidence: 3iiiDiv] Other drugs that are thought to have clinical activity as single agents are ifosfamide, epirubicin, gemcitabine, and paclitaxel.[43-46][Level of Evidence: 3iiiDiv] Their clinical activity relative to single-agent doxorubicin is not clear, and they are not known to have superior activity.
There is controversy about the clinical benefit of adding other drugs to doxorubicin as a single agent. A systematic evidence review and meta-analysis conducted by the Cochrane Collaboration summarized the eight randomized trials reported from 1976 to 1995.[47] No additional randomized trials had been reported or were known to be in progress between 1995 and the 2002 literature search. Single-agent doxorubicin had been compared with a variety of doxorubicin-containing combinations that included vincristine, vindesine, cyclophosphamide, streptozotocin, mitomycin-C, cisplatin, and/or ifosfamide. Combination regimens consistently caused more nausea and hematologic toxicity. However, the better response rates associated with combination therapy were marginal and depended on the statistical model used (fixed effects model ORresponse = 1.29; 95% CI, 1.03–1.60, P = .03; random effects model ORresp = 1.26; 95% CI, 0.96–1.67, P = .10) There was no statistically significant difference in the 1- (ORmortality = 0.87; 95% CI, 0.73–1.05, P = .14) or 2-year mortality rates (ORmortality = 0.84; 95% CI, 0.67–1.06, P = .13).
These results were very similar even when the analyses were restricted to the four trials that used DTIC and/or ifosfamide as part of the combination regimen with doxorubicin agents that were postulated to have greater activity than the others tested. A subsequent meta-analysis of all three published randomized trials of chemotherapy regimens that contained ifosfamide versus those that did not came to similar conclusions: tumor response rates were better when the regimen included ifosfamide (RRresponse = 1.52; 95% CI, 1.11–2.08), but mortality at 1 year was not (RRmortality = 0.98; 95% CI, 0.85–1.13).[48][Level of evidence: 1iiDiv]. Therefore, response rate was a poor surrogate for OS. Quality-of-life outcomes were not reported in any of the above-mentioned randomized trials, but toxicity was worse when agents were added to doxorubicin.
References
- Pisters PW, Pollock RE, Lewis VO, et al.: Long-term results of prospective trial of surgery alone with selective use of radiation for patients with T1 extremity and trunk soft tissue sarcomas. Ann Surg 246 (4): 675-81; discussion 681-2, 2007. [PUBMED Abstract]
- Lohman RF, Nabawi AS, Reece GP, et al.: Soft tissue sarcoma of the upper extremity: a 5-year experience at two institutions emphasizing the role of soft tissue flap reconstruction. Cancer 94 (8): 2256-64, 2002. [PUBMED Abstract]
- Rosenberg SA, Tepper J, Glatstein E, et al.: The treatment of soft-tissue sarcomas of the extremities: prospective randomized evaluations of (1) limb-sparing surgery plus radiation therapy compared with amputation and (2) the role of adjuvant chemotherapy. Ann Surg 196 (3): 305-15, 1982. [PUBMED Abstract]
- O'Byrne K, Steward WP: The role of adjuvant chemotherapy in the treatment of adult soft tissue sarcomas. Crit Rev Oncol Hematol 27 (3): 221-7, 1998. [PUBMED Abstract]
- Singer S, Nielsen T, Antonescu CR: Molecular biology of soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1522-32.
- Singer S, Maki RG, O'Sullivan B: Soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1533-77.
- Malawer MM, Helman LJ, O'Sullivan B: Sarcomas of bone. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1578-1609.
- Lewis JJ, Leung D, Woodruff JM, et al.: Retroperitoneal soft-tissue sarcoma: analysis of 500 patients treated and followed at a single institution. Ann Surg 228 (3): 355-65, 1998. [PUBMED Abstract]
- van Geel AN, Pastorino U, Jauch KW, et al.: Surgical treatment of lung metastases: The European Organization for Research and Treatment of Cancer-Soft Tissue and Bone Sarcoma Group study of 255 patients. Cancer 77 (4): 675-82, 1996. [PUBMED Abstract]
- Casson AG, Putnam JB, Natarajan G, et al.: Five-year survival after pulmonary metastasectomy for adult soft tissue sarcoma. Cancer 69 (3): 662-8, 1992. [PUBMED Abstract]
- Putnam JB Jr, Roth JA: Surgical treatment for pulmonary metastases from sarcoma. Hematol Oncol Clin North Am 9 (4): 869-87, 1995. [PUBMED Abstract]
- Yang JC, Chang AE, Baker AR, et al.: Randomized prospective study of the benefit of adjuvant radiation therapy in the treatment of soft tissue sarcomas of the extremity. J Clin Oncol 16 (1): 197-203, 1998. [PUBMED Abstract]
- O'Sullivan B, Davis AM, Turcotte R, et al.: Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: a randomised trial. Lancet 359 (9325): 2235-41, 2002. [PUBMED Abstract]
- O'Sullivan B, Davis A, Turcotte R, et al.: Five-year results of a randomized phase III trial of pre-operative vs post-operative radiotherapy in extremity soft tissue sarcoma. [Abstract] J Clin Oncol 22 (Suppl 14): A-9007, 819s, 2004.
- Davis AM, O'Sullivan B, Turcotte R, et al.: Late radiation morbidity following randomization to preoperative versus postoperative radiotherapy in extremity soft tissue sarcoma. Radiother Oncol 75 (1): 48-53, 2005. [PUBMED Abstract]
- Griffin AM, Dickie CI, Catton CN, et al.: The influence of time interval between preoperative radiation and surgical resection on the development of wound healing complications in extremity soft tissue sarcoma. Ann Surg Oncol 22 (9): 2824-30, 2015. [PUBMED Abstract]
- Pisters PW, Harrison LB, Leung DH, et al.: Long-term results of a prospective randomized trial of adjuvant brachytherapy in soft tissue sarcoma. J Clin Oncol 14 (3): 859-68, 1996. [PUBMED Abstract]
- Alektiar KM, Zelefsky MJ, Brennan MF: Morbidity of adjuvant brachytherapy in soft tissue sarcoma of the extremity and superficial trunk. Int J Radiat Oncol Biol Phys 47 (5): 1273-9, 2000. [PUBMED Abstract]
- Alektiar KM, Brennan MF, Healey JH, et al.: Impact of intensity-modulated radiation therapy on local control in primary soft-tissue sarcoma of the extremity. J Clin Oncol 26 (20): 3440-4, 2008. [PUBMED Abstract]
- Alektiar KM, Brennan MF, Singer S: Local control comparison of adjuvant brachytherapy to intensity-modulated radiotherapy in primary high-grade sarcoma of the extremity. Cancer 117 (14): 3229-34, 2011. [PUBMED Abstract]
- Folkert MR, Singer S, Brennan MF, et al.: Comparison of local recurrence with conventional and intensity-modulated radiation therapy for primary soft-tissue sarcomas of the extremity. J Clin Oncol 32 (29): 3236-41, 2014. [PUBMED Abstract]
- Fabrizio PL, Stafford SL, Pritchard DJ: Extremity soft-tissue sarcomas selectively treated with surgery alone. Int J Radiat Oncol Biol Phys 48 (1): 227-32, 2000. [PUBMED Abstract]
- Rydholm A, Gustafson P, Rööser B, et al.: Limb-sparing surgery without radiotherapy based on anatomic location of soft tissue sarcoma. J Clin Oncol 9 (10): 1757-65, 1991. [PUBMED Abstract]
- Al-Refaie WB, Habermann EB, Jensen EH, et al.: Surgery alone is adequate treatment for early stage soft tissue sarcoma of the extremity. Br J Surg 97 (5): 707-13, 2010. [PUBMED Abstract]
- Rydholm A: Surgery without radiotherapy in soft tissue sarcoma. Acta Orthop Scand Suppl 273: 117-9, 1997. [PUBMED Abstract]
- Bagaria SP, Ashman JB, Daugherty LC, et al.: Compliance with National Comprehensive Cancer Network guidelines in the use of radiation therapy for extremity and superficial trunk soft tissue sarcoma in the United States. J Surg Oncol 109 (7): 633-8, 2014. [PUBMED Abstract]
- Kepka L, DeLaney TF, Suit HD, et al.: Results of radiation therapy for unresected soft-tissue sarcomas. Int J Radiat Oncol Biol Phys 63 (3): 852-9, 2005. [PUBMED Abstract]
- Frustaci S, Gherlinzoni F, De Paoli A, et al.: Adjuvant chemotherapy for adult soft tissue sarcomas of the extremities and girdles: results of the Italian randomized cooperative trial. J Clin Oncol 19 (5): 1238-47, 2001. [PUBMED Abstract]
- Adjuvant chemotherapy for localised resectable soft-tissue sarcoma of adults: meta-analysis of individual data. Sarcoma Meta-analysis Collaboration. Lancet 350 (9092): 1647-54, 1997. [PUBMED Abstract]
- Sarcoma Meta-analysis Collaboration (SMAC): Adjuvant chemotherapy for localised resectable soft tissue sarcoma in adults. Cochrane Database Syst Rev (4): CD001419, 2000. [PUBMED Abstract]
- Brodowicz T, Schwameis E, Widder J, et al.: Intensified Adjuvant IFADIC Chemotherapy for Adult Soft Tissue Sarcoma: A Prospective Randomized Feasibility Trial. Sarcoma 4 (4): 151-60, 2000. [PUBMED Abstract]
- Frustaci S, De Paoli A, Bidoli E, et al.: Ifosfamide in the adjuvant therapy of soft tissue sarcomas. Oncology 65 (Suppl 2): 80-4, 2003. [PUBMED Abstract]
- Petrioli R, Coratti A, Correale P, et al.: Adjuvant epirubicin with or without Ifosfamide for adult soft-tissue sarcoma. Am J Clin Oncol 25 (5): 468-73, 2002. [PUBMED Abstract]
- Gortzak E, Azzarelli A, Buesa J, et al.: A randomised phase II study on neo-adjuvant chemotherapy for 'high-risk' adult soft-tissue sarcoma. Eur J Cancer 37 (9): 1096-103, 2001. [PUBMED Abstract]
- Pervaiz N, Colterjohn N, Farrokhyar F, et al.: A systematic meta-analysis of randomized controlled trials of adjuvant chemotherapy for localized resectable soft-tissue sarcoma. Cancer 113 (3): 573-81, 2008. [PUBMED Abstract]
- Woll PJ, van Glabbeke M, Hohenberger P, et al.: Adjuvant chemotherapy (CT) with doxorubicin and ifosfamide in resected soft tissue sarcoma (STS): Interim analysis of a randomised phase III trial. [Abstract] J Clin Oncol 25 (Suppl 18): A-10008, 2007.
- Bramwell V, Rouesse J, Steward W, et al.: Adjuvant CYVADIC chemotherapy for adult soft tissue sarcoma--reduced local recurrence but no improvement in survival: a study of the European Organization for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group. J Clin Oncol 12 (6): 1137-49, 1994. [PUBMED Abstract]
- Le Cesne A, Van Glabbeke M, Woll PJ, et al.: The end of adjuvant chemotherapy (adCT) era with doxorubicin-based regimen in resected high-grade soft tissue sarcoma (STS): pooled analysis of the two STBSG-EORTC phase III clinical trials. [Abstract] J Clin Oncol 26 (Suppl 15): A-10525, 2008.
- Issels RD, Lindner LH, Verweij J, et al.: Neo-adjuvant chemotherapy alone or with regional hyperthermia for localised high-risk soft-tissue sarcoma: a randomised phase 3 multicentre study. Lancet Oncol 11 (6): 561-70, 2010. [PUBMED Abstract]
- Eggermont AM, de Wilt JH, ten Hagen TL: Current uses of isolated limb perfusion in the clinic and a model system for new strategies. Lancet Oncol 4 (7): 429-37, 2003. [PUBMED Abstract]
- Bonvalot S, Laplanche A, Lejeune F, et al.: Limb salvage with isolated perfusion for soft tissue sarcoma: could less TNF-alpha be better? Ann Oncol 16 (7): 1061-8, 2005. [PUBMED Abstract]
- Grenader T, Goldberg A, Hadas-Halperin I, et al.: Long-term response to pegylated liposomal doxorubicin in patients with metastatic soft tissue sarcomas. Anticancer Drugs 20 (1): 15-20, 2009. [PUBMED Abstract]
- Lorigan P, Verweij J, Papai Z, et al.: Phase III trial of two investigational schedules of ifosfamide compared with standard-dose doxorubicin in advanced or metastatic soft tissue sarcoma: a European Organisation for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group Study. J Clin Oncol 25 (21): 3144-50, 2007. [PUBMED Abstract]
- Nielsen OS, Dombernowsky P, Mouridsen H, et al.: High-dose epirubicin is not an alternative to standard-dose doxorubicin in the treatment of advanced soft tissue sarcomas. A study of the EORTC soft tissue and bone sarcoma group. Br J Cancer 78 (12): 1634-9, 1998. [PUBMED Abstract]
- Maki RG, Wathen JK, Patel SR, et al.: Randomized phase II study of gemcitabine and docetaxel compared with gemcitabine alone in patients with metastatic soft tissue sarcomas: results of sarcoma alliance for research through collaboration study 002 [corrected]. J Clin Oncol 25 (19): 2755-63, 2007. [PUBMED Abstract]
- Okuno S, Ryan LM, Edmonson JH, et al.: Phase II trial of gemcitabine in patients with advanced sarcomas (E1797): a trial of the Eastern Cooperative Oncology Group. Cancer 97 (8): 1969-73, 2003. [PUBMED Abstract]
- Bramwell VH, Anderson D, Charette ML, et al.: Doxorubicin-based chemotherapy for the palliative treatment of adult patients with locally advanced or metastatic soft tissue sarcoma. Cochrane Database Syst Rev (3): CD003293, 2003. [PUBMED Abstract]
- Verma S, Younus J, Stys-Norman D, et al.: Meta-analysis of ifosfamide-based combination chemotherapy in advanced soft tissue sarcoma. Cancer Treat Rev 34 (4): 339-47, 2008. [PUBMED Abstract]
Stage I Adult Soft Tissue Sarcoma
Refer to the Treatment Option Overview section of this summary for a more detailed discussion of the roles of surgery and radiation therapy.
Low-grade soft tissue sarcomas have little metastatic potential, but they have a propensity to recur locally. Accordingly, surgical excision with negative tissue margins of 1 cm to 2 cm or larger in all directions is the treatment of choice for patients with these early-stage sarcomas.[1-3] The Mohs surgical technique may be considered as an alternative to wide surgical excision for the very rare, small, well-differentiated primary sarcomas of the skin when cosmetic results are considered to be important, as margins can be assured with minimal normal tissue removal.[4]
Carefully executed high-dose radiation therapy using a shrinking-field technique may be beneficial for unresectable tumors or for resectable tumors in which a high likelihood of residual disease is thought to be present when margins are judged to be inadequate, and when wider resection would require either an amputation or the removal of a vital organ.[5] Because of the low metastatic potential of these tumors, chemotherapy is usually not given.[6,7]
Standard treatment options:
- Surgical excision of tumors 5 cm or smaller in diameter with negative tissue margins in all directions.[8-12]
- Surgical excision with preoperative radiation therapy (preRT) or postoperative radiation therapy (PORT). Radiation decreases the risk of local recurrence but has not been shown to increase overall survival.[13-16]
- If the tumor is unresectable, high-dose preRT may be used.[17]
- For tumors of the retroperitoneum, trunk, and head and neck, the following are options:
- Surgical resection with the option of PORT if negative margins cannot be obtained. Wide margins are unusual in these sites, and radiation therapy is usually advocated for trunk and head and neck primary sites.[18]
- PreRT followed by maximal surgical resection. Radiation therapy may be used in sarcomas of the trunk and head and neck to maximize local control because of the inability to obtain wide surgical margins.[19]
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- Singer S, Nielsen T, Antonescu CR: Molecular biology of soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1522-32.
- Singer S, Maki RG, O'Sullivan B: Soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1533-77.
- Malawer MM, Helman LJ, O'Sullivan B: Sarcomas of bone. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1578-1609.
- Fish FS: Soft tissue sarcomas in dermatology. Dermatol Surg 22 (3): 268-73, 1996. [PUBMED Abstract]
- Temple WJ, Temple CL, Arthur K, et al.: Prospective cohort study of neoadjuvant treatment in conservative surgery of soft tissue sarcomas. Ann Surg Oncol 4 (7): 586-90, 1997 Oct-Nov. [PUBMED Abstract]
- Sarcoma Meta-analysis Collaboration (SMAC): Adjuvant chemotherapy for localised resectable soft tissue sarcoma in adults. Cochrane Database Syst Rev (4): CD001419, 2000. [PUBMED Abstract]
- Pervaiz N, Colterjohn N, Farrokhyar F, et al.: A systematic meta-analysis of randomized controlled trials of adjuvant chemotherapy for localized resectable soft-tissue sarcoma. Cancer 113 (3): 573-81, 2008. [PUBMED Abstract]
- Al-Refaie WB, Habermann EB, Jensen EH, et al.: Surgery alone is adequate treatment for early stage soft tissue sarcoma of the extremity. Br J Surg 97 (5): 707-13, 2010. [PUBMED Abstract]
- Pisters PW, Pollock RE, Lewis VO, et al.: Long-term results of prospective trial of surgery alone with selective use of radiation for patients with T1 extremity and trunk soft tissue sarcomas. Ann Surg 246 (4): 675-81; discussion 681-2, 2007. [PUBMED Abstract]
- Fabrizio PL, Stafford SL, Pritchard DJ: Extremity soft-tissue sarcomas selectively treated with surgery alone. Int J Radiat Oncol Biol Phys 48 (1): 227-32, 2000. [PUBMED Abstract]
- Rydholm A, Gustafson P, Rööser B, et al.: Limb-sparing surgery without radiotherapy based on anatomic location of soft tissue sarcoma. J Clin Oncol 9 (10): 1757-65, 1991. [PUBMED Abstract]
- Rydholm A: Surgery without radiotherapy in soft tissue sarcoma. Acta Orthop Scand Suppl 273: 117-9, 1997. [PUBMED Abstract]
- Yang JC, Chang AE, Baker AR, et al.: Randomized prospective study of the benefit of adjuvant radiation therapy in the treatment of soft tissue sarcomas of the extremity. J Clin Oncol 16 (1): 197-203, 1998. [PUBMED Abstract]
- O'Sullivan B, Davis AM, Turcotte R, et al.: Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: a randomised trial. Lancet 359 (9325): 2235-41, 2002. [PUBMED Abstract]
- O'Sullivan B, Davis A, Turcotte R, et al.: Five-year results of a randomized phase III trial of pre-operative vs post-operative radiotherapy in extremity soft tissue sarcoma. [Abstract] J Clin Oncol 22 (Suppl 14): A-9007, 819s, 2004.
- Davis AM, O'Sullivan B, Turcotte R, et al.: Late radiation morbidity following randomization to preoperative versus postoperative radiotherapy in extremity soft tissue sarcoma. Radiother Oncol 75 (1): 48-53, 2005. [PUBMED Abstract]
- Kepka L, DeLaney TF, Suit HD, et al.: Results of radiation therapy for unresected soft-tissue sarcomas. Int J Radiat Oncol Biol Phys 63 (3): 852-9, 2005. [PUBMED Abstract]
- Brennan MF, Singer S, Maki RG: Sarcomas of the soft tissue and bone. In: DeVita VT Jr, Hellman S, Rosenberg SA, eds.: Cancer: Principles and Practice of Oncology. Vols. 1 & 2. 8th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2008, pp 1741-1833.
- Baldini EH, Wang D, Haas RL, et al.: Treatment Guidelines for Preoperative Radiation Therapy for Retroperitoneal Sarcoma: Preliminary Consensus of an International Expert Panel. Int J Radiat Oncol Biol Phys 92 (3): 602-12, 2015. [PUBMED Abstract]
Stage II and Node-Negative Stage III Adult Soft Tissue Sarcoma
Refer to the Treatment Option Overview section of this summary for a more detailed discussion of the roles of surgery, radiation therapy, and chemotherapy.
High-grade localized soft tissue sarcomas have an increased potential for local recurrence and metastasis. For sarcomas of the extremities, local control comparable to that obtained with amputation may be achieved with limb-sparing surgery that involves wide local excision in combination with preoperative radiation therapy (preRT) or postoperative radiation therapy (PORT).
Complete surgical resection is often difficult for sarcomas of the retroperitoneum because of their large size before detection and anatomical location.[1,2] As opposed to soft tissue sarcomas of the extremities, local recurrence is the most common cause of death in patients with retroperitoneal soft tissue sarcomas. Complete surgical resection (i.e., removal of the entire gross tumor) is the most important factor in preventing local recurrence and, in many instances, requires resection of adjacent viscera. For retroperitoneal sarcomas, retrospective comparison of surgery alone versus preRT review suggests that preRT is associated with improved local recurrence-free survival, but not disease-free survival.[3]
Standard treatment options:
- Surgical excision with preRT or PORT. Radiation decreases the risk of local recurrence but has not been shown to increase overall survival.[4-8]
- Surgical excision with negative tissue margins in all directions. This approach is generally restricted to low-grade tumors ( ≤5 cm in diameter) of the extremities or superficial trunk with microscopically negative surgical tumor margins.[9-13]
- If the tumor is unresectable, high-dose radiation therapy may be used, but poor local control is likely to result.[14]
- In some situations, radiation therapy and/or chemotherapy may be used before surgery in an attempt to convert a marginally resectable tumor to one that can be adequately resected with limb preservation; this treatment may be followed by PORT.
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- Heslin MJ, Lewis JJ, Nadler E, et al.: Prognostic factors associated with long-term survival for retroperitoneal sarcoma: implications for management. J Clin Oncol 15 (8): 2832-9, 1997. [PUBMED Abstract]
- Jaques DP, Coit DG, Hajdu SI, et al.: Management of primary and recurrent soft-tissue sarcoma of the retroperitoneum. Ann Surg 212 (1): 51-9, 1990. [PUBMED Abstract]
- Kelly KJ, Yoon SS, Kuk D, et al.: Comparison of Perioperative Radiation Therapy and Surgery Versus Surgery Alone in 204 Patients With Primary Retroperitoneal Sarcoma: A Retrospective 2-Institution Study. Ann Surg 262 (1): 156-62, 2015. [PUBMED Abstract]
- Yang JC, Chang AE, Baker AR, et al.: Randomized prospective study of the benefit of adjuvant radiation therapy in the treatment of soft tissue sarcomas of the extremity. J Clin Oncol 16 (1): 197-203, 1998. [PUBMED Abstract]
- Rosenberg SA, Tepper J, Glatstein E, et al.: The treatment of soft-tissue sarcomas of the extremities: prospective randomized evaluations of (1) limb-sparing surgery plus radiation therapy compared with amputation and (2) the role of adjuvant chemotherapy. Ann Surg 196 (3): 305-15, 1982. [PUBMED Abstract]
- O'Sullivan B, Davis AM, Turcotte R, et al.: Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: a randomised trial. Lancet 359 (9325): 2235-41, 2002. [PUBMED Abstract]
- O'Sullivan B, Davis A, Turcotte R, et al.: Five-year results of a randomized phase III trial of pre-operative vs post-operative radiotherapy in extremity soft tissue sarcoma. [Abstract] J Clin Oncol 22 (Suppl 14): A-9007, 819s, 2004.
- Davis AM, O'Sullivan B, Turcotte R, et al.: Late radiation morbidity following randomization to preoperative versus postoperative radiotherapy in extremity soft tissue sarcoma. Radiother Oncol 75 (1): 48-53, 2005. [PUBMED Abstract]
- Al-Refaie WB, Habermann EB, Jensen EH, et al.: Surgery alone is adequate treatment for early stage soft tissue sarcoma of the extremity. Br J Surg 97 (5): 707-13, 2010. [PUBMED Abstract]
- Pisters PW, Pollock RE, Lewis VO, et al.: Long-term results of prospective trial of surgery alone with selective use of radiation for patients with T1 extremity and trunk soft tissue sarcomas. Ann Surg 246 (4): 675-81; discussion 681-2, 2007. [PUBMED Abstract]
- Fabrizio PL, Stafford SL, Pritchard DJ: Extremity soft-tissue sarcomas selectively treated with surgery alone. Int J Radiat Oncol Biol Phys 48 (1): 227-32, 2000. [PUBMED Abstract]
- Rydholm A, Gustafson P, Rööser B, et al.: Limb-sparing surgery without radiotherapy based on anatomic location of soft tissue sarcoma. J Clin Oncol 9 (10): 1757-65, 1991. [PUBMED Abstract]
- Rydholm A: Surgery without radiotherapy in soft tissue sarcoma. Acta Orthop Scand Suppl 273: 117-9, 1997. [PUBMED Abstract]
- Kepka L, DeLaney TF, Suit HD, et al.: Results of radiation therapy for unresected soft-tissue sarcomas. Int J Radiat Oncol Biol Phys 63 (3): 852-9, 2005. [PUBMED Abstract]
Advanced Stage III (N1) Adult Soft Tissue Sarcoma
Refer to the Treatment Option Overview section of this summary for a more detailed discussion of the roles of surgery, radiation therapy, and chemotherapy.
Regional lymph node involvement by soft tissue sarcomas of adulthood is very infrequent. However, sarcoma types that more commonly spread to lymph nodes include high-grade rhabdomyosarcoma, vascular sarcomas, and epithelioid sarcomas.[1]
Standard treatment options:
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- Mazeron JJ, Suit HD: Lymph nodes as sites of metastases from sarcomas of soft tissue. Cancer 60 (8): 1800-8, 1987. [PUBMED Abstract]
- Watson DI, Coventry BJ, Langlois SL, et al.: Soft-tissue sarcoma of the extremity. Experience with limb-sparing surgery. Med J Aust 160 (7): 412-6, 1994. [PUBMED Abstract]
- Cormier JN, Huang X, Xing Y, et al.: Cohort analysis of patients with localized, high-risk, extremity soft tissue sarcoma treated at two cancer centers: chemotherapy-associated outcomes. J Clin Oncol 22 (22): 4567-74, 2004. [PUBMED Abstract]
- O'Byrne K, Steward WP: The role of adjuvant chemotherapy in the treatment of adult soft tissue sarcomas. Crit Rev Oncol Hematol 27 (3): 221-7, 1998. [PUBMED Abstract]
- Adjuvant chemotherapy for localised resectable soft-tissue sarcoma of adults: meta-analysis of individual data. Sarcoma Meta-analysis Collaboration. Lancet 350 (9092): 1647-54, 1997. [PUBMED Abstract]
Stage IV Adult Soft Tissue Sarcoma
Refer to the Treatment Option Overview section of this summary for a more detailed discussion of the roles of surgery, radiation therapy, and chemotherapy.
In the setting of lung metastasis, resection of metastatic tumors may be associated with long-term disease-free survival in patients selected for optimal underlying disease biology (i.e., patients with a limited number of metastases and slow tumor growth).[1-3] It is not clear to what degree the favorable outcomes are attributable to the efficacy of surgery or to careful selection of patients based upon factors that are associated with less-virulent disease.[1-3] The value of resection of hepatic metastases is unclear.
As noted in the Treatment Option Overview section above, doxorubicin is the standard systemic therapy in the management of metastatic sarcomas.[4,5] Other drugs that may have clinical activity as single agents are ifosfamide, epirubicin, gemcitabine, and paclitaxel.[6-9] Their clinical activity relative to single-agent doxorubicin is not clear, and they are not known to have superior activity. There is controversy about whether adding drugs to doxorubicin offers clinical benefit beyond what is achieved by doxorubicin as a single agent. To avoid severe toxicity in older patients, sequential use of single agents may be the preferred strategy for palliation.
A randomized study assessed whether dose intensification of doxorubicin with ifosfamide improved the survival of patients with advanced soft-tissue sarcoma compared with doxorubicin alone.[10] Two hundred twenty-eight patients were randomly assigned to receive doxorubicin, and 227 patients were randomly assigned to receive doxorubicin and ifosfamide. Median follow-up was 56 months (interquartile range [IQR], 31–77) in the doxorubicin-only group and 59 months (IQR, 36–72) in the combination group.
There was no significant difference in overall survival (OS) between groups (median OS, 12.8 months; 95.5% confidence interval [CI], 10.5–14.3 in the doxorubicin-alone group vs. 14.3 months; range, 12.5–16.5 months in the doxorubicin and ifosfamide group; hazard ratio [HR], 0.83; 95.5% CI 0.67–1.03; stratified log-rank test P = .076). Median progression-free survival was significantly higher for the doxorubicin and ifosfamide group (7.4 months; 95% CI, 6.6-8.3) than for the doxorubicin-alone group (4.6 months; range, 2.9–5.6 months; HR, 0.74; 95% CI, 0.60–0.90; stratified log-rank test P = .003). More patients in the doxorubicin and ifosfamide group than in the doxorubicin-alone group had an overall response (60 [26%] of 227 patients vs. 31 [14%] of 228; P < .0006). The most common grade 3 and 4 toxic effects, which were all more common with doxorubicin and ifosfamide than with doxorubicin alone, were leucopenia (97 [43%] of 224 patients vs. 40 [18%] of 223 patients), neutropenia (93 [42%] vs. 83 [37%]), febrile neutropenia (103 (46%) vs. 30 [13%]), anemia (78 [35%] vs. 10 [5%]), and thrombocytopenia (75 [33%]) vs. 1 [<1%]).[10][Level of evidence: 1iiA] Treatment intensification with doxorubicin and ifosfamide for palliation of advanced soft tissue sarcoma is not indicated.
Standard treatment options
- Chemotherapy.
- Single-agent chemotherapy, with subsequent single agents for disease regrowth.[4-6,8,9,11] Doxorubicin is generally the first-line agent. Ifosfamide also has substantial single-agent activity.
- Doxorubicin-based combination chemotherapy. A variety of regimens have been used, but none has been proven to increase OS compared with doxorubicin alone.[4,5] There is some evidence that the addition of ifosfamide increases response rates (but not survival). Toxicity is increased with the addition of drugs to doxorubicin. No quality-of-life studies have been reported in comparisons of single-agent therapy versus combination therapy.
- Resection of pulmonary lesions may be performed if the primary tumor is under control.[1-3]
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- van Geel AN, Pastorino U, Jauch KW, et al.: Surgical treatment of lung metastases: The European Organization for Research and Treatment of Cancer-Soft Tissue and Bone Sarcoma Group study of 255 patients. Cancer 77 (4): 675-82, 1996. [PUBMED Abstract]
- Casson AG, Putnam JB, Natarajan G, et al.: Five-year survival after pulmonary metastasectomy for adult soft tissue sarcoma. Cancer 69 (3): 662-8, 1992. [PUBMED Abstract]
- Putnam JB Jr, Roth JA: Surgical treatment for pulmonary metastases from sarcoma. Hematol Oncol Clin North Am 9 (4): 869-87, 1995. [PUBMED Abstract]
- Bramwell VH, Anderson D, Charette ML, et al.: Doxorubicin-based chemotherapy for the palliative treatment of adult patients with locally advanced or metastatic soft tissue sarcoma. Cochrane Database Syst Rev (3): CD003293, 2003. [PUBMED Abstract]
- Verma S, Younus J, Stys-Norman D, et al.: Meta-analysis of ifosfamide-based combination chemotherapy in advanced soft tissue sarcoma. Cancer Treat Rev 34 (4): 339-47, 2008. [PUBMED Abstract]
- Lorigan P, Verweij J, Papai Z, et al.: Phase III trial of two investigational schedules of ifosfamide compared with standard-dose doxorubicin in advanced or metastatic soft tissue sarcoma: a European Organisation for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group Study. J Clin Oncol 25 (21): 3144-50, 2007. [PUBMED Abstract]
- Nielsen OS, Dombernowsky P, Mouridsen H, et al.: High-dose epirubicin is not an alternative to standard-dose doxorubicin in the treatment of advanced soft tissue sarcomas. A study of the EORTC soft tissue and bone sarcoma group. Br J Cancer 78 (12): 1634-9, 1998. [PUBMED Abstract]
- Maki RG, Wathen JK, Patel SR, et al.: Randomized phase II study of gemcitabine and docetaxel compared with gemcitabine alone in patients with metastatic soft tissue sarcomas: results of sarcoma alliance for research through collaboration study 002 [corrected]. J Clin Oncol 25 (19): 2755-63, 2007. [PUBMED Abstract]
- Okuno S, Ryan LM, Edmonson JH, et al.: Phase II trial of gemcitabine in patients with advanced sarcomas (E1797): a trial of the Eastern Cooperative Oncology Group. Cancer 97 (8): 1969-73, 2003. [PUBMED Abstract]
- Judson I, Verweij J, Gelderblom H, et al.: Doxorubicin alone versus intensified doxorubicin plus ifosfamide for first-line treatment of advanced or metastatic soft-tissue sarcoma: a randomised controlled phase 3 trial. Lancet Oncol 15 (4): 415-23, 2014. [PUBMED Abstract]
- Grenader T, Goldberg A, Hadas-Halperin I, et al.: Long-term response to pegylated liposomal doxorubicin in patients with metastatic soft tissue sarcomas. Anticancer Drugs 20 (1): 15-20, 2009. [PUBMED Abstract]
Recurrent Adult Soft Tissue Sarcoma
Treatment of patients with recurrent soft tissue sarcoma depends on the type of initial presentation and treatment. Patients who develop a local recurrence often can be treated by local therapy: surgical excision plus radiation therapy after previous minimal therapy or amputation after previous aggressive treatment.[1-7] Resection of limited pulmonary metastases may be associated with favorable disease-free survival.[8-10][Level of evidence: 3iiiDiv] However, the contribution of selection factors, such as low tumor burden, slow tumor growth, and long disease-free interval, to these favorable outcomes is not known.
There is no standard chemotherapy for recurrent soft tissue sarcomas that have progressed after doxorubicin as a single agent or in combination with other agents that have clinical activity, such as ifosfamide, epirubicin, gemcitabine, and paclitaxel. Any of these agents not previously administered to the patient may be used sequentially at the time of recurrence or progression.[11-14][Level of Evidence: 3iiiDiv] None of these agents has been shown to increase overall survival in this setting, therefore, clinical trials are an appropriate option.
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- Singer S, Nielsen T, Antonescu CR: Molecular biology of soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1522-32.
- Singer S, Maki RG, O'Sullivan B: Soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1533-77.
- Malawer MM, Helman LJ, O'Sullivan B: Sarcomas of bone. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1578-1609.
- Midis GP, Pollock RE, Chen NP, et al.: Locally recurrent soft tissue sarcoma of the extremities. Surgery 123 (6): 666-71, 1998. [PUBMED Abstract]
- Essner R, Selch M, Eilber FR: Reirradiation for extremity soft tissue sarcomas. Local control and complications. Cancer 67 (11): 2813-7, 1991. [PUBMED Abstract]
- Singer S, Antman K, Corson JM, et al.: Long-term salvageability for patients with locally recurrent soft-tissue sarcomas. Arch Surg 127 (5): 548-53; discussion 553-4, 1992. [PUBMED Abstract]
- Lewis JJ, Leung D, Heslin M, et al.: Association of local recurrence with subsequent survival in extremity soft tissue sarcoma. J Clin Oncol 15 (2): 646-52, 1997. [PUBMED Abstract]
- van Geel AN, Pastorino U, Jauch KW, et al.: Surgical treatment of lung metastases: The European Organization for Research and Treatment of Cancer-Soft Tissue and Bone Sarcoma Group study of 255 patients. Cancer 77 (4): 675-82, 1996. [PUBMED Abstract]
- Casson AG, Putnam JB, Natarajan G, et al.: Five-year survival after pulmonary metastasectomy for adult soft tissue sarcoma. Cancer 69 (3): 662-8, 1992. [PUBMED Abstract]
- Putnam JB Jr, Roth JA: Surgical treatment for pulmonary metastases from sarcoma. Hematol Oncol Clin North Am 9 (4): 869-87, 1995. [PUBMED Abstract]
- Lorigan P, Verweij J, Papai Z, et al.: Phase III trial of two investigational schedules of ifosfamide compared with standard-dose doxorubicin in advanced or metastatic soft tissue sarcoma: a European Organisation for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group Study. J Clin Oncol 25 (21): 3144-50, 2007. [PUBMED Abstract]
- Nielsen OS, Dombernowsky P, Mouridsen H, et al.: High-dose epirubicin is not an alternative to standard-dose doxorubicin in the treatment of advanced soft tissue sarcomas. A study of the EORTC soft tissue and bone sarcoma group. Br J Cancer 78 (12): 1634-9, 1998. [PUBMED Abstract]
- Maki RG, Wathen JK, Patel SR, et al.: Randomized phase II study of gemcitabine and docetaxel compared with gemcitabine alone in patients with metastatic soft tissue sarcomas: results of sarcoma alliance for research through collaboration study 002 [corrected]. J Clin Oncol 25 (19): 2755-63, 2007. [PUBMED Abstract]
- Okuno S, Ryan LM, Edmonson JH, et al.: Phase II trial of gemcitabine in patients with advanced sarcomas (E1797): a trial of the Eastern Cooperative Oncology Group. Cancer 97 (8): 1969-73, 2003. [PUBMED Abstract]
Changes to This Summary (02/01/2018)
The PDQ cancer information summaries are reviewed regularly and updated as new information becomes available. This section describes the latest changes made to this summary as of the date above.
General Information About Adult Soft Tissue Sarcoma
Updated statistics with estimated new cases and deaths for 2018 (cited American Cancer Society as reference 1).
Stage Information for Adult Soft Tissue Sarcoma
An editorial change was made to this section.
This summary is written and maintained by the PDQ Adult Treatment Editorial Board, which is editorially independent of NCI. The summary reflects an independent review of the literature and does not represent a policy statement of NCI or NIH. More information about summary policies and the role of the PDQ Editorial Boards in maintaining the PDQ summaries can be found on the About This PDQ Summary and PDQ® - NCI's Comprehensive Cancer Database pages.
About This PDQ Summary
Purpose of This Summary
This PDQ cancer information summary for health professionals provides comprehensive, peer-reviewed, evidence-based information about the treatment of adult soft tissue sarcoma. It is intended as a resource to inform and assist clinicians who care for cancer patients. It does not provide formal guidelines or recommendations for making health care decisions.
Reviewers and Updates
This summary is reviewed regularly and updated as necessary by the PDQ Adult Treatment Editorial Board, which is editorially independent of the National Cancer Institute (NCI). The summary reflects an independent review of the literature and does not represent a policy statement of NCI or the National Institutes of Health (NIH).
Board members review recently published articles each month to determine whether an article should:
- be discussed at a meeting,
- be cited with text, or
- replace or update an existing article that is already cited.
Changes to the summaries are made through a consensus process in which Board members evaluate the strength of the evidence in the published articles and determine how the article should be included in the summary.
The lead reviewers for Adult Soft Tissue Sarcoma Treatment are:
- Russell S. Berman, MD (New York University School of Medicine)
- Minh Tam Truong, MD (Boston University Medical Center)
Any comments or questions about the summary content should be submitted to Cancer.gov through the NCI website's Email Us. Do not contact the individual Board Members with questions or comments about the summaries. Board members will not respond to individual inquiries.
Levels of Evidence
Some of the reference citations in this summary are accompanied by a level-of-evidence designation. These designations are intended to help readers assess the strength of the evidence supporting the use of specific interventions or approaches. The PDQ Adult Treatment Editorial Board uses a formal evidence ranking system in developing its level-of-evidence designations.
Permission to Use This Summary
PDQ is a registered trademark. Although the content of PDQ documents can be used freely as text, it cannot be identified as an NCI PDQ cancer information summary unless it is presented in its entirety and is regularly updated. However, an author would be permitted to write a sentence such as “NCI’s PDQ cancer information summary about breast cancer prevention states the risks succinctly: [include excerpt from the summary].”
The preferred citation for this PDQ summary is:
PDQ® Adult Treatment Editorial Board. PDQ Adult Soft Tissue Sarcoma Treatment. Bethesda, MD: National Cancer Institute. Updated <MM/DD/YYYY>. Available at: https://www.cancer.gov/types/soft-tissue-sarcoma/hp/adult-soft-tissue-treatment-pdq. Accessed <MM/DD/YYYY>. [PMID: 26389481]
Images in this summary are used with permission of the author(s), artist, and/or publisher for use within the PDQ summaries only. Permission to use images outside the context of PDQ information must be obtained from the owner(s) and cannot be granted by the National Cancer Institute. Information about using the illustrations in this summary, along with many other cancer-related images, is available in Visuals Online, a collection of over 2,000 scientific images.
Disclaimer
Based on the strength of the available evidence, treatment options may be described as either “standard” or “under clinical evaluation.” These classifications should not be used as a basis for insurance reimbursement determinations. More information on insurance coverage is available on Cancer.gov on the Managing Cancer Care page.
Contact Us
More information about contacting us or receiving help with the Cancer.gov website can be found on our Contact Us for Help page. Questions can also be submitted to Cancer.gov through the website’s Email Us.
Updated: February 1, 2018
Source URL: https://www.cancer.gov/publishedcontent/syndication/2127.htm
Source Agency: National Cancer Institute (NCI)
Captured Date: 2013-09-14 09:00:45.0
Adult Soft Tissue Sarcoma Treatment (PDQ®)–Health Professional Version
General Information About Adult Soft Tissue Sarcoma
Incidence and Mortality
Estimated new cases and deaths from soft tissue sarcoma in the United States in 2018:[1]
- New cases: 13,040.
- Deaths: 5,150.
Soft tissue sarcomas are malignant tumors that arise in any of the mesodermal tissues of the extremities (50%), trunk and retroperitoneum (40%), or head and neck (10%). The reported international incidence rates range from 1.8 to 5 per 100,000 individuals per year.[2]
Risk Factors and Genetic Factors
The risk of sporadic soft tissue sarcomas is increased by previous radiation therapy and, in the case of lymphangiosarcoma, by chronic lymphedema. The chemicals Thorotrast (thorium dioxide), vinyl chloride, and arsenic are also established carcinogens for hepatic angiosarcomas.[3-5]
Soft tissue sarcomas occur with greater frequency in patients with the following inherited syndromes:[3-5]
- Nevoid basal cell carcinoma syndrome (Gorlin syndrome: PTC gene mutation).
- Gardner syndrome (APC mutation).
- Li-Fraumeni syndrome (p53 mutation).
- Tuberous sclerosis (Bourneville disease: TSC1 or TSC2 mutation).
- von Recklinghausen disease (neurofibromatosis type 1: NF1 mutation).
- Werner syndrome (adult progeria: WRN mutation).
Diagnosis
Soft tissue sarcomas may be heterogeneous, so adequate tissue should be obtained via either core-needle or incisional biopsy for microscopic examination to determine histologic type and tumor grade. Careful planning of the initial biopsy is important to avoid compromising subsequent curative resection. Since the selection of treatment is determined by the grade of the tumor, it is essential to have a careful review of the biopsy tissue by a pathologist who is experienced in diagnosing sarcomas. Complete staging and treatment planning by a multidisciplinary team of cancer specialists is required to determine the optimal treatment for patients with this disease.
There is evidence that at least some favorable clinical outcomes may be associated with referral to a specialized sarcoma treatment center. In a population-based consecutive series of 375 soft tissue sarcoma patients in Sweden, local recurrence rates of resected tumors were higher in patients who were not referred to the specialized center: in 35 of 78 (45%) patients not referred; in 24 of 102 (24%) patients referred after initial surgery or incisional biopsy; and in 36 of 195 (18%) patients referred before any surgical procedure (P = .0001 for the difference between those never referred vs. those referred before any surgical procedure).[6][Level of evidence: 3iDii] However, there were no statistically significant differences in death from sarcoma between the groups of patients.
Prognostic Factors
The prognosis for patients with adult soft tissue sarcomas depends on several factors, including:[3-5,7,8]
- Patient’s age.
- Size, sarcoma subtype, histologic grade, mitotic activity, and stage of the tumor.
Factors associated with a poorer prognosis include the following:[9]
- Age older than 60 years.
- Tumors larger than 5 cm in greatest dimension.
- High-grade histology with high mitotic activity.
- Positive margins after resection.[10]
Although low-grade tumors are usually curable by surgery alone, higher-grade sarcomas (as determined by the mitotic index and by the presence of hemorrhage and necrosis) are associated with higher local-treatment failure rates and increased metastatic potential.
Surveillance for Relapse
A retrospective review included 174 consecutive patients with a soft tissue sarcoma of the limb who underwent follow-up by oncologists at a single center from 2003 to 2009.[11] The rate and site of recurrence and mode of detection were analyzed. Eighty-two patients (47%) experienced relapse. Isolated local recurrences occurred in 26 patients and local relapse with synchronous pulmonary metastases occurred in 5 patients. Local recurrences were detected clinically in 30 of the 31 patients; magnetic resonance imaging identified only one local recurrence. Twenty-eight patients developed isolated lung metastases; in 9 patients, the lung metastases were amenable to resections, 7 of whom were free of disease after treatment. Lung metastases were detected by chest x-ray in 19 patients, by computed tomography scanning in 3 patients, and clinically in 11 patients. Twenty-three patients developed nonpulmonary metastases. More than 80% of the relapses occurred in the first 2 years of follow-up; however, later recurrences were also observed.[11][Level of evidence: 3iiDi] This study supports imaging surveillance for detection of lung metastases, whereas local recurrences at the primary site were usually detected by clinical examination. The impact of picking up metastases from overall survival or quality-of-life data is unknown.
Related Summaries
Other PDQ summaries containing information about soft tissue sarcoma include:
References
- American Cancer Society: Cancer Facts and Figures 2018. Atlanta, Ga: American Cancer Society, 2018. Available online. Last accessed January 5, 2018.
- Wibmer C, Leithner A, Zielonke N, et al.: Increasing incidence rates of soft tissue sarcomas? A population-based epidemiologic study and literature review. Ann Oncol 21 (5): 1106-11, 2010. [PUBMED Abstract]
- Singer S, Nielsen T, Antonescu CR: Molecular biology of soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1522-32.
- Singer S, Maki RG, O'Sullivan B: Soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1533-77.
- Malawer MM, Helman LJ, O'Sullivan B: Sarcomas of bone. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1578-1609.
- Gustafson P, Dreinhöfer KE, Rydholm A: Soft tissue sarcoma should be treated at a tumor center. A comparison of quality of surgery in 375 patients. Acta Orthop Scand 65 (1): 47-50, 1994. [PUBMED Abstract]
- Coindre JM, Terrier P, Guillou L, et al.: Predictive value of grade for metastasis development in the main histologic types of adult soft tissue sarcomas: a study of 1240 patients from the French Federation of Cancer Centers Sarcoma Group. Cancer 91 (10): 1914-26, 2001. [PUBMED Abstract]
- Kasper B, Ouali M, van Glabbeke M, et al.: Prognostic factors in adolescents and young adults (AYA) with high risk soft tissue sarcoma (STS) treated by adjuvant chemotherapy: a study based on pooled European Organisation for Research and Treatment of Cancer (EORTC) clinical trials 62771 and 62931. Eur J Cancer 49 (2): 449-56, 2013. [PUBMED Abstract]
- Vraa S, Keller J, Nielsen OS, et al.: Prognostic factors in soft tissue sarcomas: the Aarhus experience. Eur J Cancer 34 (12): 1876-82, 1998. [PUBMED Abstract]
- Trovik LH, Ovrebo K, Almquist M, et al.: Adjuvant radiotherapy in retroperitoneal sarcomas. A Scandinavian Sarcoma Group study of 97 patients. Acta Oncol 53 (9): 1165-72, 2014. [PUBMED Abstract]
- Rothermundt C, Whelan JS, Dileo P, et al.: What is the role of routine follow-up for localised limb soft tissue sarcomas? A retrospective analysis of 174 patients. Br J Cancer 110 (10): 2420-6, 2014. [PUBMED Abstract]
Cellular Classification of Adult Soft Tissue Sarcoma
Soft tissue sarcomas are classified histologically according to the soft tissue cell of origin. Additional studies, including electron microscopy, specialized immunohistochemistry, flow cytometry, cytogenetics, and tissue culture studies may allow identification of particular subtypes within the major histologic categories. For example, S100 antigen suggests neural sheath origin, cytokeratin suggests epithelioid or synovial cell origin, and factor VIII-related antigen suggests endothelial origin. Likewise, some subtypes of sarcomas have characteristic genetic markers, but these markers are not generally used in the routine clinical setting (e.g., translocation t(X;18)(p11;q11) in synovial sarcomas and translocation t(12;16)(q13;p11) in myxoid and round-cell sarcomas).[1-3]
The histologic grade reflects the metastatic potential of these tumors more accurately than the classic cellular classification listed below. Pathologists assign a grade based on the number of mitoses per high-powered field, the presence of necrosis, cellular and nuclear morphology, and the degree of cellularity; discordance among expert pathologists regarding tumor grade, and even histologic subtype, can be substantial.[4]
The World Health Organization lists the following cell types in its classification of soft tissue sarcomas:[5,6]
- Adipocytic tumors.
- Dedifferentiated liposarcoma.*
- Myxoid/round cell liposarcoma.
- Pleomorphic liposarcoma.
- Fibroblastic/myofibroblastic tumors.
- Fibrosarcoma.**
- Myxofibrosarcoma, low grade.
- Low-grade fibromyxoid sarcoma.
- Sclerosing epithelioid fibrosarcoma.
- So-called fibrohistiocytic tumors.
- Undifferentiated pleomorphic sarcoma/malignant fibrous histiocytoma (including pleomorphic, giant cell, myxoid/high-grade myxofibrosarcoma, and inflammatory forms).
- Smooth muscle tumors.
- Leiomyosarcoma.
- Skeletal muscle tumors.
- Rhabdomyosarcoma (embryonal, alveolar, and pleomorphic forms).
- Vascular tumors.
- Epithelioid hemangioendothelioma.
- Angiosarcoma, deep.***
- Tumors of peripheral nerves.
- Malignant peripheral nerve sheath tumor.
- Chondro-osseous tumors.
- Extraskeletal chondrosarcoma (mesenchymal and other variants).
- Extraskeletal osteosarcoma.
- Tumors of uncertain differentiation.
- Synovial sarcoma.
- Epithelioid sarcoma.
- Alveolar soft part sarcoma.
- Clear cell sarcoma of soft tissue.
- Extraskeletal myxoid chondrosarcoma.
- Primitive neuroectodermal tumor/extraskeletal Ewing tumor.
- Desmoplastic small round cell tumor.
- Extrarenal rhabdoid tumor.
- Undifferentiated sarcoma; sarcoma, not otherwise specified.
[Note: *It is recognized that dedifferentiated liposarcoma primarily arises in the context of deep atypical lipomatous tumor/well-differentiated liposarcoma, a sarcoma of intermediate malignancy because of the lack of metastatic capacity. **The category of fibrosarcoma can be inclusive of fibrosarcomatous differentiation in dermatofibrosarcoma protuberans. ***Cutaneous angiosarcoma may be difficult to stage using the American Joint Committee on Cancer system. (Refer to the PDQ summary on Gastrointestinal Stromal Tumors for more information.)]
References
- Singer S, Nielsen T, Antonescu CR: Molecular biology of soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1522-32.
- Singer S, Maki RG, O'Sullivan B: Soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1533-77.
- Malawer MM, Helman LJ, O'Sullivan B: Sarcomas of bone. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1578-1609.
- Alvegård TA, Berg NO: Histopathology peer review of high-grade soft tissue sarcoma: the Scandinavian Sarcoma Group experience. J Clin Oncol 7 (12): 1845-51, 1989. [PUBMED Abstract]
- Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-6.
- Brodowicz T, Schwameis E, Widder J, et al.: Intensified Adjuvant IFADIC Chemotherapy for Adult Soft Tissue Sarcoma: A Prospective Randomized Feasibility Trial. Sarcoma 4 (4): 151-60, 2000. [PUBMED Abstract]
Stage Information for Adult Soft Tissue Sarcoma
Note: The American Joint Committee on Cancer (AJCC) has published the 8th edition of the AJCC Cancer Staging Manual, which includes revisions to the staging for this disease. Implementation of the 8th edition began in January 2018. The PDQ Adult Treatment Editorial Board, which maintains this summary, is reviewing the revised staging and will make appropriate changes as needed.
Staging has an important role in determining the most effective treatment for soft tissue sarcoma. Clinical staging involves magnetic resonance imaging (MRI) or computed tomography (CT) of the primary tumor area and a chest CT to look for metastasis to the lung (the most common site of distant spread). An abdominal CT scan is done in the case of retroperitoneal sarcomas because the liver may be the site of initial clinical metastasis for these tumors.
The stage is determined by the size of the tumor, the histologic grade, and whether there is spread to lymph nodes or distant sites. Intracompartmental or extracompartmental extension of extremity sarcomas is also important for surgical decision making. For complete staging, a thorough review of all biopsy specimens (including those from the primary tumor, lymph nodes, or other suspicious lesions) is essential. CT scan of the chest is recommended for sarcomas larger than 5 cm (T2) or with moderate to poor differentiation (grades 2–4). Nodal involvement is rare, occurring in fewer than 3% of patients with sarcoma.[1]
Lymph node involvement in soft tissue sarcomas of adulthood is rare but is somewhat more frequent in some subtypes (e.g., rhabdomyosarcoma, vascular sarcomas, clear cell sarcomas, and epithelioid sarcomas) when they are high grade.[2] Because treatment decisions are predicated on pathology staging, patients should be staged before, and again after, any neoadjuvant therapy. The assessment of tumor grade can be affected in either direction, but more frequently decreased because of differential cellular loss related to the neoadjuvant chemotherapy or radiation.[3] Grade, which is based on cellular differentiation, mitotic rate, and extent of necrosis, should be recorded for all soft tissue sarcomas. A three-grade system (G1–G3) is preferred. (See below.)
The AJCC has designated staging by the four criteria of tumor size, nodal status, metastasis, and grade (TNMG).[3] The characteristic molecular markers of some sarcomas are not formally incorporated in the staging system pending further evaluation of their impact on prognosis. Recurrent sarcomas are restaged using the same system as for primary tumors with the specification that the tumor is recurrent.
Definitions of TNM and Grade
| aReprinted with permission from AJCC: Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-8. | |
| bSuperficial tumor is located exclusively above the superficial fascia without invasion of the fascia; deep tumor is located either exclusively beneath the superficial fascia, superficial to the fascia with invasion of or through the fascia, or both superficial yet beneath the fascia. | |
| TX | Primary tumor cannot be assessed. |
| T0 | No evidence of primary tumor. |
| T1 | Tumor ≤5 cm in greatest dimension. (Size should be regarded as a continuous variable, and the measurement should be provided.) |
| T1a | Superficial tumor.b |
| T1b | Deep tumor.b |
| T2 | Tumor >5 cm in greatest dimension.b |
| T2a | Superficial tumor.b |
| T2b | Deep tumor. |
| aReprinted with permission from AJCC: Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-8. | |
| bPresence of positive nodes (N1) in M0 tumors is considered Stage III. | |
| NX | Regional lymph nodes cannot be assessed. |
| N0 | No regional lymph node metastasis. |
| N1b | Regional lymph node metastasis. |
| aReprinted with permission from AJCC: Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-8. | |
| M0 | No distant metastasis. |
| M1 | Distant metastasis. |
| aReprinted with permission from AJCC: Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-8. | ||||
| Stage IA | T1a | N0 | M0 | G1, GX |
| T1b | N0 | M0 | G1, GX | |
| Stage IB | T2a | N0 | M0 | G1, GX |
| T2b | N0 | M0 | G1, GX | |
| Stage IIA | T1a | N0 | M0 | G2, G3 |
| T1b | N0 | M0 | G2, G3 | |
| Stage IIB | T2a | N0 | M0 | G2 |
| T2b | N0 | M0 | G2 | |
| Stage III | T2a, T2b | N0 | M0 | G3 |
| Any T | N1 | M0 | Any G | |
| Stage IV | Any T | Any N | M1 | Any G |
Neurovascular and bone invasion are indicators of poor prognosis, but they are not incorporated into the formal staging system.
References
- Fong Y, Coit DG, Woodruff JM, et al.: Lymph node metastasis from soft tissue sarcoma in adults. Analysis of data from a prospective database of 1772 sarcoma patients. Ann Surg 217 (1): 72-7, 1993. [PUBMED Abstract]
- Mazeron JJ, Suit HD: Lymph nodes as sites of metastases from sarcomas of soft tissue. Cancer 60 (8): 1800-8, 1987. [PUBMED Abstract]
- Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, et al., eds.: AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer, 2010, pp 291-6.
Treatment Option Overview
Multimodality Approach
In most cases, a combined modality approach of preoperative radiation therapy (preRT) or postoperative radiation therapy (PORT) is used, rather than the radical surgical procedures, such as amputation, that were used in the past. It may even be possible to use surgery without PORT in selected cases. For example, a case series was reported from a specialized sarcoma treatment referral center in which 74 selected patients with primary extremity and trunk tumors 5 cm or smaller were found to have no histologic involvement of the surgical margins. The patients were observed without radiation therapy, and the estimated local recurrence rate after 10 years was 11%.[1][Level of evidence: 3iiiDiv] The role of chemotherapy is not as well defined as is the role for radiation therapy. Because of the evolving nature of the treatment options for this disease, patients should be considered when available. Information about ongoing clinical trials is available from the NCI website.
Role of Surgery
Surgical resection is the mainstay of therapy for soft tissue sarcomas. When feasible, wide-margin function–sparing surgical excision is the cornerstone of effective treatment for extremity tumors. This may be facilitated by soft tissue reconstructive surgery, which generally permits wider margins than those obtained when the surgical plan involves direct closure of the excision site.[2] Cutting into the tumor mass or shelling out the gross tumor along the plane of the pseudocapsule of compressed tumor cells and reactive tissue that often surrounds soft tissue sarcomas are associated with an elevated risk of local recurrence. Even high-grade, soft tissue sarcomas of the extremities can usually be effectively treated while preserving the limb with combined-modality treatment consisting of preRT or PORT to reduce local recurrence. (Refer to the Role of Radiation Therapy section of this summary for more information.)
Only one small, single-institution, randomized trial has directly compared amputation to limb-sparing surgery for soft tissue sarcomas of the extremities.[3] In a 2:1 randomization ratio, 27 patients with high-grade extremity sarcomas were assigned to a wide excision plus PORT (45 Gy–50 Gy to the wide local excision area, and a total of 60 Gy–70 Gy to the tumor bed over 6–7 weeks), and 16 were assigned to amputation at or above the joint proximal to the tumor. Both groups received adjuvant chemotherapy (i.e., doxorubicin, cyclophosphamide, and high-dose methotrexate). At 63 months, with a median follow-up of 56 months, there were four local recurrences in the 27 patients who underwent limb-sparing surgery and no recurrences in the 16 patients who underwent amputation P2 = .12. Overall survival (OS) rates were not statistically significantly different (actuarial 5-year survival rate, 83% vs. 88%, P2 = .99).[3][Level of evidence: 1iiA]
Local control of high-grade soft tissue sarcomas of the trunk and the head and neck can be achieved with surgery in combination with radiation therapy.[4] It may be possible to use surgery without PORT in selected cases. For example, a case series was reported from a specialized sarcoma treatment referral center in which 74 selected patients with primary extremity and trunk tumors 5 cm or smaller were found to have no histologic involvement of the surgical margins.[1] They were observed without radiation therapy, and the estimated local recurrence rate after 10 years was 11%.[1][Level of evidence: 3iiiDiv] The role of chemotherapy is not as well defined as is the role of radiation therapy. Because of the evolving nature of the treatment options for this disease, patients should be offered the option of clinical trials when available.
Effective treatment of retroperitoneal sarcomas requires removal of all gross disease while sparing adjacent viscera not invaded by tumor. The prognosis for patients with high-grade retroperitoneal sarcomas is less favorable than for patients with tumors at other sites, partly because of the difficulty in completely resecting these tumors and the dose-limiting toxicity of high-dose radiation therapy on visceral organs.[5-8]
In the setting of distant metastasis, surgery may be associated with long-term, disease-free survival in patients with pulmonary metastasis and optimal underlying disease biology (i.e., patients with a limited number of metastases and slow nodule growth) who have undergone or are undergoing complete resection of the primary tumor.[9-11] It is not clear to what degree the favorable outcomes are attributable to the efficacy of surgery or the careful selection of patients based on factors that are associated with less-virulent disease.
Role of Radiation Therapy
Radiation plays an important role in limb-sparing therapy. Pre- and postoperative external-beam radiation therapies (EBRT), as well as brachytherapy, have been shown to decrease the risk of local recurrence. They have not been shown to increase OS but are used to avoid amputation for all but the most locally advanced tumors or for limbs seriously compromised by vascular disease, where acceptable functional preservation is not possible. In the case of EBRT, irradiation of the entire limb circumference is avoided to preserve vascular and nerve structures that are critical to function and preservation of the limb.
PORT
PORT has been tested in a single-institution, randomized trial of 141 patients with extremity sarcomas who were treated with limb-sparing surgery. Patients with high-grade tumors (n = 91) also received adjuvant chemotherapy (i.e., five 28-day cycles of doxorubicin and cyclophosphamide). All patients were randomly assigned to receive radiation (45 Gy to a wide field, plus a tumor-bed boost of 18 Gy over 6–7 weeks), concurrent with chemotherapy in the case of high-grade tumors versus no radiation.[12] At up to 12 years of follow-up, there was one local recurrence in the 70 patients randomly assigned to receive radiation versus 17 recurrences in the 71 control patients (P = .0001), with similar reduction in risk of local recurrence for both high- and low-grade tumors. However, there was no difference in OS between the radiation and control groups.[12][Level of evidence: 1iiDiii] Global quality of life was similar in the two groups, but the radiation therapy group had substantially worse functional deficits resulting from reduced strength and joint motion as well as increased edema.
To limit acute toxicity with preRT, smaller fields and lower doses are generally given than is the case with PORT. PreRT has been directly compared with PORT for extremity soft tissue sarcomas in a multicenter randomized trial.[13-15] Designed to include 266 patients, the trial was stopped early after 190 patients had been accrued because of an increase in wound complications in the preRT group. The scheduled radiation in the preRT group was a wide field of 50 Gy in 2-Gy fractions (first phase of the trial) with an additional 16 Gy to 20 Gy to the tumor bed and a 2-cm margin (second phase of the trial) only if tumor cells were found at the surgical margins.
Patients in the PORT group were scheduled to receive radiation during both phases of the trial. The wound-complication rates were 35% versus 17% in the preRT and PORT groups, respectively (P = .01). In addition, limb function at 6 weeks after surgery was worse in the preRT group (P = .01).[13] At 5 years, the two groups had similar local control rates (93% vs. 92%) and OS (73% vs. 67%, P = .48).[14] Of the 129 patients evaluated for limb function at 21 to 27 months after surgery (n = 73 for preRT and n = 56 for PORT), limb function was similar in both groups, but there was a statistical trend for less fibrosis in the preRT group (P = .07).[15]
Brachytherapy
Brachytherapy has also been investigated as an adjuvant therapy for soft tissue sarcomas. Although it has possible advantages of convenience and less radiation to normal surrounding tissue relative to EBRT, the two treatment strategies have not been directly compared in terms of efficacy or morbidity. However, adjuvant brachytherapy has been compared with surgery without radiation. The time interval between preRT and surgical excision in extremity soft tissue sarcoma had minimal influence on the development of wound complications. Four- or 5-week intervals showed equivalent complication rates between patients who did or did not develop wound complications, suggesting an optimal interval to reduce potential complications.[16]
In a single-institution trial, 164 patients with sarcomas of the extremity or superficial trunk were randomly assigned during surgery, if all gross tumor could be excised, to receive an iridium Ir 192 implant (delivering 42 Gy–45 Gy over 4–6 days; 78 patients) or to a control arm of no radiation (86 patients).[17,18] Some of the patients with high-grade tumors received adjuvant doxorubicin-based chemotherapy if they were thought to be at a high risk for metastasis (34 patients in each study arm). With a median follow-up of 76 months, the 5-year actuarial local recurrence rates were 18% and 31% in the brachytherapy and control arms, respectively (P = .04). This difference was limited to patients with high-grade tumors. There was no discernible difference in sarcoma-specific survival rates between the brachytherapy and control arms (84% and 81%, respectively; P = .65), and there was no difference in the high tumor-grade group.[17][Level of evidence: 1iiDiii] The rates of clinically important wound complications (e.g., need for operative revision or repeated seroma drainage, wound separation, large hematomas, or purulent infection) were 24% and 14% in the radiation and control arms, respectively (P = .13); wound reoperation rates were 10% and 0%, respectively (P = .006).[18]
Intensity-modulated radiation therapy
Intensity-modulated radiation therapy (IMRT) has been used to deliver preRT or PORT to patients with extremity soft tissue sarcomas in an effort to spare the femur, joints, and selected other normal tissues from the full prescription dose and to maintain local control while potentially reducing radiation therapy-related morbidity. Initial single-institution reports suggest that high rates of local control with some reduction in morbidity are possible with this technique.[19,20] Retrospective comparison of IMRT compared with 3-dimensional, conformal radiation therapy demonstrates that local recurrence for primary soft tissue sarcomas of the extremity was worse in the non-IMRT group.[21][Level of evidence: 3iiiDiv]
Surgery and radiation therapy
In some tumors of the extremities or trunk, surgery alone can be performed without the use of radiation. Evidence for this approach is limited to single-institution, relatively small, case series [1,22,23] or analysis of outcomes in the National Cancer Institute's Surveillance, Epidemiology, and End Results (SEER) tumor registry.[24] However, these comparisons suffer from low statistical power and differential evaluability rates that could have introduced bias.[1] Patient selection factors may vary among surgeons. In general, this approach is considered in patients with low-grade tumors of the extremity or superficial trunk that are 5 cm or smaller in diameter (T1) and have microscopically negative surgical margins; long-term local tumor control is about 90% in such patients.[25]
A patterns-of-care study using SEER data was queried to identify patients undergoing surgery for truncal and extremity soft tissue sarcomas from 2004 to 2009.[26] Of 5,075 patients, 50% received radiation therapy. Radiation was considered to be underused in a significant portion of patients undergoing treatment for soft tissue sarcoma in the United States. Although routine radiation therapy is not recommended for stage I patients, 25% of them still underwent radiation. Even though routine radiation therapy is recommended for patients with stage II and III tumors, only 60% of them underwent radiation. On multivariate analysis, predictors of radiation therapy included age younger than 50 years (odds ratio [OR], 1.57; 95% confidence interval [CI], 1.28–1.91), malignant fibrous histiocytoma histology (OR, 1.47; 95% CI, 1.3–1.92), T2 classification (OR, 1.88; 95% CI, 1.60–2.20), and G3 (OR, 6.27; 95% CI, 5.10–7.72). Patients with stage III soft tissue sarcoma who received radiation therapy showed improved disease-specific survival at 5 years compared with those who did not (68% vs. 46%, P < .001).[26][Level of evidence: 3iDii]
On occasion, surgical excision cannot be performed in the initial management of soft tissue sarcomas because the morbidity would be unacceptable or nearby critical organs make complete resection impossible. In such circumstances, radiation has been used as the primary therapy.[27] However, this must be considered a treatment of last resort. Experience is limited to retrospective case series from single centers.[27][Level of evidence: 3iiiDiv]
Role of Adjuvant or Neoadjuvant Chemotherapy for Clinically Localized Tumors
The role of adjuvant chemotherapy is not completely clear. The investigation of its use falls into two categories or generations—pre- and post-ifosfamide regimens. In discussions with a patient, any potential benefits should be considered in the context of the short- and long-term toxicities of the chemotherapy.
First-generation trials (preifosfamide)
Several prospective, randomized trials were unable to determine conclusively whether doxorubicin-based adjuvant chemotherapy benefits adults with resectable soft tissue sarcomas. The majority of these studies accrued small numbers of patients and did not demonstrate a metastasis-free survival or an OS benefit for adjuvant chemotherapy.[4] A small study of adjuvant chemotherapy showed a positive effect on both disease-free survival (DFS) and OS in patients treated with postoperative chemotherapy.[28] There was wide interstudy variability among the reported trials, including differences in therapeutic regimens, drug doses, sample size, tumor site, and histologic grade.
A quantitative meta-analysis of updated data from 1,568 individual patients in 14 trials of doxorubicin-based adjuvant therapy showed an absolute benefit from adjuvant therapy of 6% for a local relapse-free interval (95% CI, 1%–10%), 10% for a distant relapse-free interval (95% CI, 5%–15%), and 10% for recurrence-free survival (95% CI, 5%–15%). A statistically significant OS benefit at 10 years was not detected: absolute difference 4% (95% CI, -1%–+9%).[29,30][Level of evidence: 1iiDii] However, only a small proportion of patients in this meta-analysis were treated with ifosfamide, an agent with demonstrated activity against soft tissue sarcoma. In addition, a subset analysis suggested that patients with sarcomas of the extremities may have benefited from adjuvant chemotherapy (hazard ratio [HR] for death, 0.8, P = .029), but there was no clear evidence that patients with extremity sarcomas had outcomes that were statistically significantly different from the outcomes of patients with tumors at other sites (P = .58).[30]
Second-generation trials (postifosfamide)
Subsequent chemotherapy trials were performed using anthracycline and ifosfamide combinations in patients who primarily had extremity or truncal soft tissue sarcomas. The data are conflicting, and the issue is still not settled. In a small feasibility study, 59 patients with high-risk, soft tissue sarcomas, 58 of whom had an extremity or the trunk as the primary site, underwent primary resection plus PORT and were randomly assigned to observation versus a dose-dense regimen of six 14-day courses of ifosfamide, dacarbazine (DTIC), and doxorubicin (IFADIC regimen) with granulocyte colony-stimulating factor (G-CSF) bone marrow support and mesna uroprotection.[31] There were no statistically significant differences in OS or relapse-free survival (RFS), but the study was severely underpowered.
In a second trial performed by the Italian National Council for Research, high-risk patients were treated with local therapy (i.e., wide resection plus preRT or PORT, or amputation as clinically necessary) and were then randomly assigned to observation versus five 21-day cycles of 4-epidoxorubicin (epirubicin) plus ifosfamide (with mesna and G-CSF).[28,32] Based on power calculations, the planned study size was 190 patients, but the trial was stopped after 104 patients had been entered because an interim analysis revealed a statistically significant (P = .001) difference in DFS favoring the chemotherapy arm. By the time of the initial peer-reviewed report of the study, the DFS still favored the chemotherapy group (median DFS of 48 months vs. 16 months), but the P value had risen to .04.[28]
Although there was no difference in metastasis-free survival at the time of the report, there was an improvement in median OS (75 months vs. 46 months, P = .03). However, at the follow-up report (at a median of 89.6 months in a range of 56–119 months), OS differences were no longer statistically significant (58.5% vs. 43.1% [P = .07]). The DFS difference had also lost statistical significance (47.2% vs. 16.0% [P = .09]).[32] In summary, the trial was underpowered because it was stopped early, and the early promising results that led to stopping the trial diminished as the trial matured.
In a third, underpowered, single-center trial, 88 patients with high-risk, soft tissue sarcomas (64 of whom had extremity or truncal primary tumors) underwent surgery (with or without radiation) and were then randomly assigned to receive four 21-day cycles of chemotherapy (epirubicin [n = 26] or epirubicin plus ifosfamide [n = 19]) versus no adjuvant chemotherapy (n = 43).[33] The trial was closed prematurely because of a slow accrual rate. After a median follow-up of 94 months, the 5-year DFS in the chemotherapy and control arms was 69% versus 44%, respectively (P = .01); the 5-year OS rates were 72% versus 47% (P = .06). All of the benefit associated with chemotherapy appeared restricted to the 19 patients who received epirubicin plus ifosfamide.
In yet another underpowered trial, 137 patients with high-risk, soft tissue sarcomas (93% with extremity or truncal primary tumors) who met the eligibility criteria were randomly assigned to undergo surgical resection (with or without radiation) or to receive three preoperative 21-day cycles of doxorubicin plus ifosfamide.[34] This multicenter European Organization for Research and Treatment of Cancer trial (EORTC-62874) was closed because of slow accrual and results that were not promising enough to continue. With a median follow-up of 7.3 years, the 5-year DFS in the surgery alone and chemotherapy plus surgery arms was 52% and 56%, respectively (P = .35); and OS was 64% and 65%, respectively (P = .22).
These last four trials have been combined with the 14 first-generation trials in a trial-level meta-analysis.[35] Of the 18 randomized trials of patients with resectable soft tissue sarcomas, five trials used a combination of doxorubicin (50–90 mg/m2 per cycle) plus ifosfamide (1,500–5,000 mg/m2 per cycle). The remaining 13 trials used doxorubicin (50–70 mg/m2 per cycle) alone or with other drugs. The absolute risk reduction in local recurrence rates associated with any chemotherapy added to local therapy was 4 percentage points (95% CI, 0%–7%), and it was 5 percentage points (95% CI, 1%–12%) when ifosfamide was combined with doxorubicin. The absolute reduction in overall mortality was 6 percentage points with any chemotherapy (95% CI, 2%–11%; [i.e., a reduction from 46%–40%]), 11 percentage points for doxorubicin plus ifosfamide (95% CI, 3%–19%; [i.e., a reduction from 41%–30%]), and 5 percentage points for doxorubicin without ifosfamide.[35][Level of evidence: 1iiA]
An additional multicenter randomized trial (EORTC-62931 [NCT00002641]), the largest trial reported to date using adjuvant doxorubicin (75 mg/m2) plus ifosfamide (5,000 mg/m2), was subsequently published in abstract form and was not included in the above meta-analysis.[36] The results differed from those reported in the meta-analysis.[35] After local therapy, 351 patients were randomly assigned to five 21-day cycles of adjuvant therapy versus observation. The trial was stopped for futility because the 5-year RFS was 52% in both arms. OS was 64% in the chemotherapy arm versus 69% in the observation arm. In a subsequent abstract, the EORTC investigators reported a combined analysis of this trial and their previous trial (EORTC-62771) [37] of adjuvant cyclophosphamide plus doxorubicin plus DTIC (CYVADIC), representing the two largest trials of adjuvant therapy for adult soft tissue sarcoma in the literature.[38] The combined analysis showed no improvement in either RFS or OS associated with adjuvant chemotherapy.[38][Level of evidence: 1iiA]
In summary, the impact of adjuvant chemotherapy on survival is not clear but is likely to be small in absolute magnitude. Therefore, in discussions with a patient, any potential benefits should be considered in the context of the short- and long-term toxicities of the chemotherapy.
Role of regional hyperthermia
The use of regional hyperthermia to enhance the local effects of systemic chemotherapy in the neoadjuvant and adjuvant setting is under investigation. In a multicenter phase III trial, 341 patients with high-risk (tumor ≥5 cm, grade 2–3, and deep to fascia), soft tissue sarcomas (149 extremity tumors and 192 nonextremity tumors) were randomly allocated to receive four 21-day cycles of chemotherapy (etoposide 125 mg/m2 on days 1 and 4; ifosfamide 1,500 mg/m2 on days 1–4; doxorubicin 50 mg/m2 on day 1) with or without regional hyperthermia both before and after local therapy.[39] Approximately 11% of the patients were being treated for recurrent tumors. The regional hyperthermia was designed to produce tumor temperatures of 42°C for 60 minutes and was given on days 1 and 4 of each chemotherapy cycle. After the first four cycles of chemotherapy, definitive surgical excision of the tumor was performed, if possible, followed by radiation therapy, if indicated (i.e., a 52.7 Gy median dose delivered), and then the last four cycles of chemotherapy plus or minus hyperthermia. Three of the nine treatment centers with particular expertise in hyperthermia treated 91% of the patients in the trial.
The median duration of follow-up was 34 months. Local progression occurred in 56 patients in the hyperthermia group and 76 patients in the control group. The relative HR for local progression or death was 0.58 (95% CI, 0.41–0.84), with an absolute difference at 2 years of 15% (76% vs. 61%; 95% CI of the difference 6–26). The decreased risk of local progression or death was seen in both extremity and nonextremity tumors. However, hyperthermia had no effect on distant failure rates nor was there a statistically significant effect on OS (HR, .88, 95% CI, 0.64–1.21; P = .43).[39][Level of evidence: 1iiDiii] There was a higher rate of grade 3 to 4 leucopenia in the hyperthermia group: 77.6% versus 63.5% (P = .005). Since a large proportion of the patients were treated at centers with special expertise, there is no certainty that the finding can be generalized to apply to other settings.
Role of isolated limb perfusion
Isolated limb perfusion is under investigation as a means to deliver high doses of chemotherapy and permit limb salvage in unresectable primary or recurrent extremity soft tissue sarcomas that would otherwise require amputation, in the opinion of the surgeon.[40,41] Common drugs used in the procedure are TNF-alpha, melphalan, and interferon-gamma. Experience is limited to case series with response rates and reported avoidance of amputation as the outcome.[40,41][Level of evidence: 3iiiDiv] The technique requires specialized expertise to avoid severe local and systemic toxicity including systemic effects of TNF-alpha. The technique has not been directly compared with standard approaches using combined systemic and local therapy.
Role of chemotherapy for advanced disease
Doxorubicin is a mainstay of systemic therapy in the management of locally advanced and metastatic soft tissue sarcoma. Pegylated liposomal encapsulated doxorubicin is a formulation of doxorubicin designed to prolong the half-life of circulating doxorubicin and slow the release of active drugs.[42] The changed pharmacokinetics result in less myelosuppression and possibly less cardiotoxic effects, but there is a substantial incidence of hypersensitivity-like reactions and hand-foot syndrome. Its clinical activity relative to unencapsulated doxorubicin is not clear.[42][Level of evidence: 3iiiDiv] Other drugs that are thought to have clinical activity as single agents are ifosfamide, epirubicin, gemcitabine, and paclitaxel.[43-46][Level of Evidence: 3iiiDiv] Their clinical activity relative to single-agent doxorubicin is not clear, and they are not known to have superior activity.
There is controversy about the clinical benefit of adding other drugs to doxorubicin as a single agent. A systematic evidence review and meta-analysis conducted by the Cochrane Collaboration summarized the eight randomized trials reported from 1976 to 1995.[47] No additional randomized trials had been reported or were known to be in progress between 1995 and the 2002 literature search. Single-agent doxorubicin had been compared with a variety of doxorubicin-containing combinations that included vincristine, vindesine, cyclophosphamide, streptozotocin, mitomycin-C, cisplatin, and/or ifosfamide. Combination regimens consistently caused more nausea and hematologic toxicity. However, the better response rates associated with combination therapy were marginal and depended on the statistical model used (fixed effects model ORresponse = 1.29; 95% CI, 1.03–1.60, P = .03; random effects model ORresp = 1.26; 95% CI, 0.96–1.67, P = .10) There was no statistically significant difference in the 1- (ORmortality = 0.87; 95% CI, 0.73–1.05, P = .14) or 2-year mortality rates (ORmortality = 0.84; 95% CI, 0.67–1.06, P = .13).
These results were very similar even when the analyses were restricted to the four trials that used DTIC and/or ifosfamide as part of the combination regimen with doxorubicin agents that were postulated to have greater activity than the others tested. A subsequent meta-analysis of all three published randomized trials of chemotherapy regimens that contained ifosfamide versus those that did not came to similar conclusions: tumor response rates were better when the regimen included ifosfamide (RRresponse = 1.52; 95% CI, 1.11–2.08), but mortality at 1 year was not (RRmortality = 0.98; 95% CI, 0.85–1.13).[48][Level of evidence: 1iiDiv]. Therefore, response rate was a poor surrogate for OS. Quality-of-life outcomes were not reported in any of the above-mentioned randomized trials, but toxicity was worse when agents were added to doxorubicin.
References
- Pisters PW, Pollock RE, Lewis VO, et al.: Long-term results of prospective trial of surgery alone with selective use of radiation for patients with T1 extremity and trunk soft tissue sarcomas. Ann Surg 246 (4): 675-81; discussion 681-2, 2007. [PUBMED Abstract]
- Lohman RF, Nabawi AS, Reece GP, et al.: Soft tissue sarcoma of the upper extremity: a 5-year experience at two institutions emphasizing the role of soft tissue flap reconstruction. Cancer 94 (8): 2256-64, 2002. [PUBMED Abstract]
- Rosenberg SA, Tepper J, Glatstein E, et al.: The treatment of soft-tissue sarcomas of the extremities: prospective randomized evaluations of (1) limb-sparing surgery plus radiation therapy compared with amputation and (2) the role of adjuvant chemotherapy. Ann Surg 196 (3): 305-15, 1982. [PUBMED Abstract]
- O'Byrne K, Steward WP: The role of adjuvant chemotherapy in the treatment of adult soft tissue sarcomas. Crit Rev Oncol Hematol 27 (3): 221-7, 1998. [PUBMED Abstract]
- Singer S, Nielsen T, Antonescu CR: Molecular biology of soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1522-32.
- Singer S, Maki RG, O'Sullivan B: Soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1533-77.
- Malawer MM, Helman LJ, O'Sullivan B: Sarcomas of bone. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1578-1609.
- Lewis JJ, Leung D, Woodruff JM, et al.: Retroperitoneal soft-tissue sarcoma: analysis of 500 patients treated and followed at a single institution. Ann Surg 228 (3): 355-65, 1998. [PUBMED Abstract]
- van Geel AN, Pastorino U, Jauch KW, et al.: Surgical treatment of lung metastases: The European Organization for Research and Treatment of Cancer-Soft Tissue and Bone Sarcoma Group study of 255 patients. Cancer 77 (4): 675-82, 1996. [PUBMED Abstract]
- Casson AG, Putnam JB, Natarajan G, et al.: Five-year survival after pulmonary metastasectomy for adult soft tissue sarcoma. Cancer 69 (3): 662-8, 1992. [PUBMED Abstract]
- Putnam JB Jr, Roth JA: Surgical treatment for pulmonary metastases from sarcoma. Hematol Oncol Clin North Am 9 (4): 869-87, 1995. [PUBMED Abstract]
- Yang JC, Chang AE, Baker AR, et al.: Randomized prospective study of the benefit of adjuvant radiation therapy in the treatment of soft tissue sarcomas of the extremity. J Clin Oncol 16 (1): 197-203, 1998. [PUBMED Abstract]
- O'Sullivan B, Davis AM, Turcotte R, et al.: Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: a randomised trial. Lancet 359 (9325): 2235-41, 2002. [PUBMED Abstract]
- O'Sullivan B, Davis A, Turcotte R, et al.: Five-year results of a randomized phase III trial of pre-operative vs post-operative radiotherapy in extremity soft tissue sarcoma. [Abstract] J Clin Oncol 22 (Suppl 14): A-9007, 819s, 2004.
- Davis AM, O'Sullivan B, Turcotte R, et al.: Late radiation morbidity following randomization to preoperative versus postoperative radiotherapy in extremity soft tissue sarcoma. Radiother Oncol 75 (1): 48-53, 2005. [PUBMED Abstract]
- Griffin AM, Dickie CI, Catton CN, et al.: The influence of time interval between preoperative radiation and surgical resection on the development of wound healing complications in extremity soft tissue sarcoma. Ann Surg Oncol 22 (9): 2824-30, 2015. [PUBMED Abstract]
- Pisters PW, Harrison LB, Leung DH, et al.: Long-term results of a prospective randomized trial of adjuvant brachytherapy in soft tissue sarcoma. J Clin Oncol 14 (3): 859-68, 1996. [PUBMED Abstract]
- Alektiar KM, Zelefsky MJ, Brennan MF: Morbidity of adjuvant brachytherapy in soft tissue sarcoma of the extremity and superficial trunk. Int J Radiat Oncol Biol Phys 47 (5): 1273-9, 2000. [PUBMED Abstract]
- Alektiar KM, Brennan MF, Healey JH, et al.: Impact of intensity-modulated radiation therapy on local control in primary soft-tissue sarcoma of the extremity. J Clin Oncol 26 (20): 3440-4, 2008. [PUBMED Abstract]
- Alektiar KM, Brennan MF, Singer S: Local control comparison of adjuvant brachytherapy to intensity-modulated radiotherapy in primary high-grade sarcoma of the extremity. Cancer 117 (14): 3229-34, 2011. [PUBMED Abstract]
- Folkert MR, Singer S, Brennan MF, et al.: Comparison of local recurrence with conventional and intensity-modulated radiation therapy for primary soft-tissue sarcomas of the extremity. J Clin Oncol 32 (29): 3236-41, 2014. [PUBMED Abstract]
- Fabrizio PL, Stafford SL, Pritchard DJ: Extremity soft-tissue sarcomas selectively treated with surgery alone. Int J Radiat Oncol Biol Phys 48 (1): 227-32, 2000. [PUBMED Abstract]
- Rydholm A, Gustafson P, Rööser B, et al.: Limb-sparing surgery without radiotherapy based on anatomic location of soft tissue sarcoma. J Clin Oncol 9 (10): 1757-65, 1991. [PUBMED Abstract]
- Al-Refaie WB, Habermann EB, Jensen EH, et al.: Surgery alone is adequate treatment for early stage soft tissue sarcoma of the extremity. Br J Surg 97 (5): 707-13, 2010. [PUBMED Abstract]
- Rydholm A: Surgery without radiotherapy in soft tissue sarcoma. Acta Orthop Scand Suppl 273: 117-9, 1997. [PUBMED Abstract]
- Bagaria SP, Ashman JB, Daugherty LC, et al.: Compliance with National Comprehensive Cancer Network guidelines in the use of radiation therapy for extremity and superficial trunk soft tissue sarcoma in the United States. J Surg Oncol 109 (7): 633-8, 2014. [PUBMED Abstract]
- Kepka L, DeLaney TF, Suit HD, et al.: Results of radiation therapy for unresected soft-tissue sarcomas. Int J Radiat Oncol Biol Phys 63 (3): 852-9, 2005. [PUBMED Abstract]
- Frustaci S, Gherlinzoni F, De Paoli A, et al.: Adjuvant chemotherapy for adult soft tissue sarcomas of the extremities and girdles: results of the Italian randomized cooperative trial. J Clin Oncol 19 (5): 1238-47, 2001. [PUBMED Abstract]
- Adjuvant chemotherapy for localised resectable soft-tissue sarcoma of adults: meta-analysis of individual data. Sarcoma Meta-analysis Collaboration. Lancet 350 (9092): 1647-54, 1997. [PUBMED Abstract]
- Sarcoma Meta-analysis Collaboration (SMAC): Adjuvant chemotherapy for localised resectable soft tissue sarcoma in adults. Cochrane Database Syst Rev (4): CD001419, 2000. [PUBMED Abstract]
- Brodowicz T, Schwameis E, Widder J, et al.: Intensified Adjuvant IFADIC Chemotherapy for Adult Soft Tissue Sarcoma: A Prospective Randomized Feasibility Trial. Sarcoma 4 (4): 151-60, 2000. [PUBMED Abstract]
- Frustaci S, De Paoli A, Bidoli E, et al.: Ifosfamide in the adjuvant therapy of soft tissue sarcomas. Oncology 65 (Suppl 2): 80-4, 2003. [PUBMED Abstract]
- Petrioli R, Coratti A, Correale P, et al.: Adjuvant epirubicin with or without Ifosfamide for adult soft-tissue sarcoma. Am J Clin Oncol 25 (5): 468-73, 2002. [PUBMED Abstract]
- Gortzak E, Azzarelli A, Buesa J, et al.: A randomised phase II study on neo-adjuvant chemotherapy for 'high-risk' adult soft-tissue sarcoma. Eur J Cancer 37 (9): 1096-103, 2001. [PUBMED Abstract]
- Pervaiz N, Colterjohn N, Farrokhyar F, et al.: A systematic meta-analysis of randomized controlled trials of adjuvant chemotherapy for localized resectable soft-tissue sarcoma. Cancer 113 (3): 573-81, 2008. [PUBMED Abstract]
- Woll PJ, van Glabbeke M, Hohenberger P, et al.: Adjuvant chemotherapy (CT) with doxorubicin and ifosfamide in resected soft tissue sarcoma (STS): Interim analysis of a randomised phase III trial. [Abstract] J Clin Oncol 25 (Suppl 18): A-10008, 2007.
- Bramwell V, Rouesse J, Steward W, et al.: Adjuvant CYVADIC chemotherapy for adult soft tissue sarcoma--reduced local recurrence but no improvement in survival: a study of the European Organization for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group. J Clin Oncol 12 (6): 1137-49, 1994. [PUBMED Abstract]
- Le Cesne A, Van Glabbeke M, Woll PJ, et al.: The end of adjuvant chemotherapy (adCT) era with doxorubicin-based regimen in resected high-grade soft tissue sarcoma (STS): pooled analysis of the two STBSG-EORTC phase III clinical trials. [Abstract] J Clin Oncol 26 (Suppl 15): A-10525, 2008.
- Issels RD, Lindner LH, Verweij J, et al.: Neo-adjuvant chemotherapy alone or with regional hyperthermia for localised high-risk soft-tissue sarcoma: a randomised phase 3 multicentre study. Lancet Oncol 11 (6): 561-70, 2010. [PUBMED Abstract]
- Eggermont AM, de Wilt JH, ten Hagen TL: Current uses of isolated limb perfusion in the clinic and a model system for new strategies. Lancet Oncol 4 (7): 429-37, 2003. [PUBMED Abstract]
- Bonvalot S, Laplanche A, Lejeune F, et al.: Limb salvage with isolated perfusion for soft tissue sarcoma: could less TNF-alpha be better? Ann Oncol 16 (7): 1061-8, 2005. [PUBMED Abstract]
- Grenader T, Goldberg A, Hadas-Halperin I, et al.: Long-term response to pegylated liposomal doxorubicin in patients with metastatic soft tissue sarcomas. Anticancer Drugs 20 (1): 15-20, 2009. [PUBMED Abstract]
- Lorigan P, Verweij J, Papai Z, et al.: Phase III trial of two investigational schedules of ifosfamide compared with standard-dose doxorubicin in advanced or metastatic soft tissue sarcoma: a European Organisation for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group Study. J Clin Oncol 25 (21): 3144-50, 2007. [PUBMED Abstract]
- Nielsen OS, Dombernowsky P, Mouridsen H, et al.: High-dose epirubicin is not an alternative to standard-dose doxorubicin in the treatment of advanced soft tissue sarcomas. A study of the EORTC soft tissue and bone sarcoma group. Br J Cancer 78 (12): 1634-9, 1998. [PUBMED Abstract]
- Maki RG, Wathen JK, Patel SR, et al.: Randomized phase II study of gemcitabine and docetaxel compared with gemcitabine alone in patients with metastatic soft tissue sarcomas: results of sarcoma alliance for research through collaboration study 002 [corrected]. J Clin Oncol 25 (19): 2755-63, 2007. [PUBMED Abstract]
- Okuno S, Ryan LM, Edmonson JH, et al.: Phase II trial of gemcitabine in patients with advanced sarcomas (E1797): a trial of the Eastern Cooperative Oncology Group. Cancer 97 (8): 1969-73, 2003. [PUBMED Abstract]
- Bramwell VH, Anderson D, Charette ML, et al.: Doxorubicin-based chemotherapy for the palliative treatment of adult patients with locally advanced or metastatic soft tissue sarcoma. Cochrane Database Syst Rev (3): CD003293, 2003. [PUBMED Abstract]
- Verma S, Younus J, Stys-Norman D, et al.: Meta-analysis of ifosfamide-based combination chemotherapy in advanced soft tissue sarcoma. Cancer Treat Rev 34 (4): 339-47, 2008. [PUBMED Abstract]
Stage I Adult Soft Tissue Sarcoma
Refer to the Treatment Option Overview section of this summary for a more detailed discussion of the roles of surgery and radiation therapy.
Low-grade soft tissue sarcomas have little metastatic potential, but they have a propensity to recur locally. Accordingly, surgical excision with negative tissue margins of 1 cm to 2 cm or larger in all directions is the treatment of choice for patients with these early-stage sarcomas.[1-3] The Mohs surgical technique may be considered as an alternative to wide surgical excision for the very rare, small, well-differentiated primary sarcomas of the skin when cosmetic results are considered to be important, as margins can be assured with minimal normal tissue removal.[4]
Carefully executed high-dose radiation therapy using a shrinking-field technique may be beneficial for unresectable tumors or for resectable tumors in which a high likelihood of residual disease is thought to be present when margins are judged to be inadequate, and when wider resection would require either an amputation or the removal of a vital organ.[5] Because of the low metastatic potential of these tumors, chemotherapy is usually not given.[6,7]
Standard treatment options:
- Surgical excision of tumors 5 cm or smaller in diameter with negative tissue margins in all directions.[8-12]
- Surgical excision with preoperative radiation therapy (preRT) or postoperative radiation therapy (PORT). Radiation decreases the risk of local recurrence but has not been shown to increase overall survival.[13-16]
- If the tumor is unresectable, high-dose preRT may be used.[17]
- For tumors of the retroperitoneum, trunk, and head and neck, the following are options:
- Surgical resection with the option of PORT if negative margins cannot be obtained. Wide margins are unusual in these sites, and radiation therapy is usually advocated for trunk and head and neck primary sites.[18]
- PreRT followed by maximal surgical resection. Radiation therapy may be used in sarcomas of the trunk and head and neck to maximize local control because of the inability to obtain wide surgical margins.[19]
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- Singer S, Nielsen T, Antonescu CR: Molecular biology of soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1522-32.
- Singer S, Maki RG, O'Sullivan B: Soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1533-77.
- Malawer MM, Helman LJ, O'Sullivan B: Sarcomas of bone. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1578-1609.
- Fish FS: Soft tissue sarcomas in dermatology. Dermatol Surg 22 (3): 268-73, 1996. [PUBMED Abstract]
- Temple WJ, Temple CL, Arthur K, et al.: Prospective cohort study of neoadjuvant treatment in conservative surgery of soft tissue sarcomas. Ann Surg Oncol 4 (7): 586-90, 1997 Oct-Nov. [PUBMED Abstract]
- Sarcoma Meta-analysis Collaboration (SMAC): Adjuvant chemotherapy for localised resectable soft tissue sarcoma in adults. Cochrane Database Syst Rev (4): CD001419, 2000. [PUBMED Abstract]
- Pervaiz N, Colterjohn N, Farrokhyar F, et al.: A systematic meta-analysis of randomized controlled trials of adjuvant chemotherapy for localized resectable soft-tissue sarcoma. Cancer 113 (3): 573-81, 2008. [PUBMED Abstract]
- Al-Refaie WB, Habermann EB, Jensen EH, et al.: Surgery alone is adequate treatment for early stage soft tissue sarcoma of the extremity. Br J Surg 97 (5): 707-13, 2010. [PUBMED Abstract]
- Pisters PW, Pollock RE, Lewis VO, et al.: Long-term results of prospective trial of surgery alone with selective use of radiation for patients with T1 extremity and trunk soft tissue sarcomas. Ann Surg 246 (4): 675-81; discussion 681-2, 2007. [PUBMED Abstract]
- Fabrizio PL, Stafford SL, Pritchard DJ: Extremity soft-tissue sarcomas selectively treated with surgery alone. Int J Radiat Oncol Biol Phys 48 (1): 227-32, 2000. [PUBMED Abstract]
- Rydholm A, Gustafson P, Rööser B, et al.: Limb-sparing surgery without radiotherapy based on anatomic location of soft tissue sarcoma. J Clin Oncol 9 (10): 1757-65, 1991. [PUBMED Abstract]
- Rydholm A: Surgery without radiotherapy in soft tissue sarcoma. Acta Orthop Scand Suppl 273: 117-9, 1997. [PUBMED Abstract]
- Yang JC, Chang AE, Baker AR, et al.: Randomized prospective study of the benefit of adjuvant radiation therapy in the treatment of soft tissue sarcomas of the extremity. J Clin Oncol 16 (1): 197-203, 1998. [PUBMED Abstract]
- O'Sullivan B, Davis AM, Turcotte R, et al.: Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: a randomised trial. Lancet 359 (9325): 2235-41, 2002. [PUBMED Abstract]
- O'Sullivan B, Davis A, Turcotte R, et al.: Five-year results of a randomized phase III trial of pre-operative vs post-operative radiotherapy in extremity soft tissue sarcoma. [Abstract] J Clin Oncol 22 (Suppl 14): A-9007, 819s, 2004.
- Davis AM, O'Sullivan B, Turcotte R, et al.: Late radiation morbidity following randomization to preoperative versus postoperative radiotherapy in extremity soft tissue sarcoma. Radiother Oncol 75 (1): 48-53, 2005. [PUBMED Abstract]
- Kepka L, DeLaney TF, Suit HD, et al.: Results of radiation therapy for unresected soft-tissue sarcomas. Int J Radiat Oncol Biol Phys 63 (3): 852-9, 2005. [PUBMED Abstract]
- Brennan MF, Singer S, Maki RG: Sarcomas of the soft tissue and bone. In: DeVita VT Jr, Hellman S, Rosenberg SA, eds.: Cancer: Principles and Practice of Oncology. Vols. 1 & 2. 8th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2008, pp 1741-1833.
- Baldini EH, Wang D, Haas RL, et al.: Treatment Guidelines for Preoperative Radiation Therapy for Retroperitoneal Sarcoma: Preliminary Consensus of an International Expert Panel. Int J Radiat Oncol Biol Phys 92 (3): 602-12, 2015. [PUBMED Abstract]
Stage II and Node-Negative Stage III Adult Soft Tissue Sarcoma
Refer to the Treatment Option Overview section of this summary for a more detailed discussion of the roles of surgery, radiation therapy, and chemotherapy.
High-grade localized soft tissue sarcomas have an increased potential for local recurrence and metastasis. For sarcomas of the extremities, local control comparable to that obtained with amputation may be achieved with limb-sparing surgery that involves wide local excision in combination with preoperative radiation therapy (preRT) or postoperative radiation therapy (PORT).
Complete surgical resection is often difficult for sarcomas of the retroperitoneum because of their large size before detection and anatomical location.[1,2] As opposed to soft tissue sarcomas of the extremities, local recurrence is the most common cause of death in patients with retroperitoneal soft tissue sarcomas. Complete surgical resection (i.e., removal of the entire gross tumor) is the most important factor in preventing local recurrence and, in many instances, requires resection of adjacent viscera. For retroperitoneal sarcomas, retrospective comparison of surgery alone versus preRT review suggests that preRT is associated with improved local recurrence-free survival, but not disease-free survival.[3]
Standard treatment options:
- Surgical excision with preRT or PORT. Radiation decreases the risk of local recurrence but has not been shown to increase overall survival.[4-8]
- Surgical excision with negative tissue margins in all directions. This approach is generally restricted to low-grade tumors ( ≤5 cm in diameter) of the extremities or superficial trunk with microscopically negative surgical tumor margins.[9-13]
- If the tumor is unresectable, high-dose radiation therapy may be used, but poor local control is likely to result.[14]
- In some situations, radiation therapy and/or chemotherapy may be used before surgery in an attempt to convert a marginally resectable tumor to one that can be adequately resected with limb preservation; this treatment may be followed by PORT.
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- Heslin MJ, Lewis JJ, Nadler E, et al.: Prognostic factors associated with long-term survival for retroperitoneal sarcoma: implications for management. J Clin Oncol 15 (8): 2832-9, 1997. [PUBMED Abstract]
- Jaques DP, Coit DG, Hajdu SI, et al.: Management of primary and recurrent soft-tissue sarcoma of the retroperitoneum. Ann Surg 212 (1): 51-9, 1990. [PUBMED Abstract]
- Kelly KJ, Yoon SS, Kuk D, et al.: Comparison of Perioperative Radiation Therapy and Surgery Versus Surgery Alone in 204 Patients With Primary Retroperitoneal Sarcoma: A Retrospective 2-Institution Study. Ann Surg 262 (1): 156-62, 2015. [PUBMED Abstract]
- Yang JC, Chang AE, Baker AR, et al.: Randomized prospective study of the benefit of adjuvant radiation therapy in the treatment of soft tissue sarcomas of the extremity. J Clin Oncol 16 (1): 197-203, 1998. [PUBMED Abstract]
- Rosenberg SA, Tepper J, Glatstein E, et al.: The treatment of soft-tissue sarcomas of the extremities: prospective randomized evaluations of (1) limb-sparing surgery plus radiation therapy compared with amputation and (2) the role of adjuvant chemotherapy. Ann Surg 196 (3): 305-15, 1982. [PUBMED Abstract]
- O'Sullivan B, Davis AM, Turcotte R, et al.: Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: a randomised trial. Lancet 359 (9325): 2235-41, 2002. [PUBMED Abstract]
- O'Sullivan B, Davis A, Turcotte R, et al.: Five-year results of a randomized phase III trial of pre-operative vs post-operative radiotherapy in extremity soft tissue sarcoma. [Abstract] J Clin Oncol 22 (Suppl 14): A-9007, 819s, 2004.
- Davis AM, O'Sullivan B, Turcotte R, et al.: Late radiation morbidity following randomization to preoperative versus postoperative radiotherapy in extremity soft tissue sarcoma. Radiother Oncol 75 (1): 48-53, 2005. [PUBMED Abstract]
- Al-Refaie WB, Habermann EB, Jensen EH, et al.: Surgery alone is adequate treatment for early stage soft tissue sarcoma of the extremity. Br J Surg 97 (5): 707-13, 2010. [PUBMED Abstract]
- Pisters PW, Pollock RE, Lewis VO, et al.: Long-term results of prospective trial of surgery alone with selective use of radiation for patients with T1 extremity and trunk soft tissue sarcomas. Ann Surg 246 (4): 675-81; discussion 681-2, 2007. [PUBMED Abstract]
- Fabrizio PL, Stafford SL, Pritchard DJ: Extremity soft-tissue sarcomas selectively treated with surgery alone. Int J Radiat Oncol Biol Phys 48 (1): 227-32, 2000. [PUBMED Abstract]
- Rydholm A, Gustafson P, Rööser B, et al.: Limb-sparing surgery without radiotherapy based on anatomic location of soft tissue sarcoma. J Clin Oncol 9 (10): 1757-65, 1991. [PUBMED Abstract]
- Rydholm A: Surgery without radiotherapy in soft tissue sarcoma. Acta Orthop Scand Suppl 273: 117-9, 1997. [PUBMED Abstract]
- Kepka L, DeLaney TF, Suit HD, et al.: Results of radiation therapy for unresected soft-tissue sarcomas. Int J Radiat Oncol Biol Phys 63 (3): 852-9, 2005. [PUBMED Abstract]
Advanced Stage III (N1) Adult Soft Tissue Sarcoma
Refer to the Treatment Option Overview section of this summary for a more detailed discussion of the roles of surgery, radiation therapy, and chemotherapy.
Regional lymph node involvement by soft tissue sarcomas of adulthood is very infrequent. However, sarcoma types that more commonly spread to lymph nodes include high-grade rhabdomyosarcoma, vascular sarcomas, and epithelioid sarcomas.[1]
Standard treatment options:
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- Mazeron JJ, Suit HD: Lymph nodes as sites of metastases from sarcomas of soft tissue. Cancer 60 (8): 1800-8, 1987. [PUBMED Abstract]
- Watson DI, Coventry BJ, Langlois SL, et al.: Soft-tissue sarcoma of the extremity. Experience with limb-sparing surgery. Med J Aust 160 (7): 412-6, 1994. [PUBMED Abstract]
- Cormier JN, Huang X, Xing Y, et al.: Cohort analysis of patients with localized, high-risk, extremity soft tissue sarcoma treated at two cancer centers: chemotherapy-associated outcomes. J Clin Oncol 22 (22): 4567-74, 2004. [PUBMED Abstract]
- O'Byrne K, Steward WP: The role of adjuvant chemotherapy in the treatment of adult soft tissue sarcomas. Crit Rev Oncol Hematol 27 (3): 221-7, 1998. [PUBMED Abstract]
- Adjuvant chemotherapy for localised resectable soft-tissue sarcoma of adults: meta-analysis of individual data. Sarcoma Meta-analysis Collaboration. Lancet 350 (9092): 1647-54, 1997. [PUBMED Abstract]
Stage IV Adult Soft Tissue Sarcoma
Refer to the Treatment Option Overview section of this summary for a more detailed discussion of the roles of surgery, radiation therapy, and chemotherapy.
In the setting of lung metastasis, resection of metastatic tumors may be associated with long-term disease-free survival in patients selected for optimal underlying disease biology (i.e., patients with a limited number of metastases and slow tumor growth).[1-3] It is not clear to what degree the favorable outcomes are attributable to the efficacy of surgery or to careful selection of patients based upon factors that are associated with less-virulent disease.[1-3] The value of resection of hepatic metastases is unclear.
As noted in the Treatment Option Overview section above, doxorubicin is the standard systemic therapy in the management of metastatic sarcomas.[4,5] Other drugs that may have clinical activity as single agents are ifosfamide, epirubicin, gemcitabine, and paclitaxel.[6-9] Their clinical activity relative to single-agent doxorubicin is not clear, and they are not known to have superior activity. There is controversy about whether adding drugs to doxorubicin offers clinical benefit beyond what is achieved by doxorubicin as a single agent. To avoid severe toxicity in older patients, sequential use of single agents may be the preferred strategy for palliation.
A randomized study assessed whether dose intensification of doxorubicin with ifosfamide improved the survival of patients with advanced soft-tissue sarcoma compared with doxorubicin alone.[10] Two hundred twenty-eight patients were randomly assigned to receive doxorubicin, and 227 patients were randomly assigned to receive doxorubicin and ifosfamide. Median follow-up was 56 months (interquartile range [IQR], 31–77) in the doxorubicin-only group and 59 months (IQR, 36–72) in the combination group.
There was no significant difference in overall survival (OS) between groups (median OS, 12.8 months; 95.5% confidence interval [CI], 10.5–14.3 in the doxorubicin-alone group vs. 14.3 months; range, 12.5–16.5 months in the doxorubicin and ifosfamide group; hazard ratio [HR], 0.83; 95.5% CI 0.67–1.03; stratified log-rank test P = .076). Median progression-free survival was significantly higher for the doxorubicin and ifosfamide group (7.4 months; 95% CI, 6.6-8.3) than for the doxorubicin-alone group (4.6 months; range, 2.9–5.6 months; HR, 0.74; 95% CI, 0.60–0.90; stratified log-rank test P = .003). More patients in the doxorubicin and ifosfamide group than in the doxorubicin-alone group had an overall response (60 [26%] of 227 patients vs. 31 [14%] of 228; P < .0006). The most common grade 3 and 4 toxic effects, which were all more common with doxorubicin and ifosfamide than with doxorubicin alone, were leucopenia (97 [43%] of 224 patients vs. 40 [18%] of 223 patients), neutropenia (93 [42%] vs. 83 [37%]), febrile neutropenia (103 (46%) vs. 30 [13%]), anemia (78 [35%] vs. 10 [5%]), and thrombocytopenia (75 [33%]) vs. 1 [<1%]).[10][Level of evidence: 1iiA] Treatment intensification with doxorubicin and ifosfamide for palliation of advanced soft tissue sarcoma is not indicated.
Standard treatment options
- Chemotherapy.
- Single-agent chemotherapy, with subsequent single agents for disease regrowth.[4-6,8,9,11] Doxorubicin is generally the first-line agent. Ifosfamide also has substantial single-agent activity.
- Doxorubicin-based combination chemotherapy. A variety of regimens have been used, but none has been proven to increase OS compared with doxorubicin alone.[4,5] There is some evidence that the addition of ifosfamide increases response rates (but not survival). Toxicity is increased with the addition of drugs to doxorubicin. No quality-of-life studies have been reported in comparisons of single-agent therapy versus combination therapy.
- Resection of pulmonary lesions may be performed if the primary tumor is under control.[1-3]
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- van Geel AN, Pastorino U, Jauch KW, et al.: Surgical treatment of lung metastases: The European Organization for Research and Treatment of Cancer-Soft Tissue and Bone Sarcoma Group study of 255 patients. Cancer 77 (4): 675-82, 1996. [PUBMED Abstract]
- Casson AG, Putnam JB, Natarajan G, et al.: Five-year survival after pulmonary metastasectomy for adult soft tissue sarcoma. Cancer 69 (3): 662-8, 1992. [PUBMED Abstract]
- Putnam JB Jr, Roth JA: Surgical treatment for pulmonary metastases from sarcoma. Hematol Oncol Clin North Am 9 (4): 869-87, 1995. [PUBMED Abstract]
- Bramwell VH, Anderson D, Charette ML, et al.: Doxorubicin-based chemotherapy for the palliative treatment of adult patients with locally advanced or metastatic soft tissue sarcoma. Cochrane Database Syst Rev (3): CD003293, 2003. [PUBMED Abstract]
- Verma S, Younus J, Stys-Norman D, et al.: Meta-analysis of ifosfamide-based combination chemotherapy in advanced soft tissue sarcoma. Cancer Treat Rev 34 (4): 339-47, 2008. [PUBMED Abstract]
- Lorigan P, Verweij J, Papai Z, et al.: Phase III trial of two investigational schedules of ifosfamide compared with standard-dose doxorubicin in advanced or metastatic soft tissue sarcoma: a European Organisation for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group Study. J Clin Oncol 25 (21): 3144-50, 2007. [PUBMED Abstract]
- Nielsen OS, Dombernowsky P, Mouridsen H, et al.: High-dose epirubicin is not an alternative to standard-dose doxorubicin in the treatment of advanced soft tissue sarcomas. A study of the EORTC soft tissue and bone sarcoma group. Br J Cancer 78 (12): 1634-9, 1998. [PUBMED Abstract]
- Maki RG, Wathen JK, Patel SR, et al.: Randomized phase II study of gemcitabine and docetaxel compared with gemcitabine alone in patients with metastatic soft tissue sarcomas: results of sarcoma alliance for research through collaboration study 002 [corrected]. J Clin Oncol 25 (19): 2755-63, 2007. [PUBMED Abstract]
- Okuno S, Ryan LM, Edmonson JH, et al.: Phase II trial of gemcitabine in patients with advanced sarcomas (E1797): a trial of the Eastern Cooperative Oncology Group. Cancer 97 (8): 1969-73, 2003. [PUBMED Abstract]
- Judson I, Verweij J, Gelderblom H, et al.: Doxorubicin alone versus intensified doxorubicin plus ifosfamide for first-line treatment of advanced or metastatic soft-tissue sarcoma: a randomised controlled phase 3 trial. Lancet Oncol 15 (4): 415-23, 2014. [PUBMED Abstract]
- Grenader T, Goldberg A, Hadas-Halperin I, et al.: Long-term response to pegylated liposomal doxorubicin in patients with metastatic soft tissue sarcomas. Anticancer Drugs 20 (1): 15-20, 2009. [PUBMED Abstract]
Recurrent Adult Soft Tissue Sarcoma
Treatment of patients with recurrent soft tissue sarcoma depends on the type of initial presentation and treatment. Patients who develop a local recurrence often can be treated by local therapy: surgical excision plus radiation therapy after previous minimal therapy or amputation after previous aggressive treatment.[1-7] Resection of limited pulmonary metastases may be associated with favorable disease-free survival.[8-10][Level of evidence: 3iiiDiv] However, the contribution of selection factors, such as low tumor burden, slow tumor growth, and long disease-free interval, to these favorable outcomes is not known.
There is no standard chemotherapy for recurrent soft tissue sarcomas that have progressed after doxorubicin as a single agent or in combination with other agents that have clinical activity, such as ifosfamide, epirubicin, gemcitabine, and paclitaxel. Any of these agents not previously administered to the patient may be used sequentially at the time of recurrence or progression.[11-14][Level of Evidence: 3iiiDiv] None of these agents has been shown to increase overall survival in this setting, therefore, clinical trials are an appropriate option.
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- Singer S, Nielsen T, Antonescu CR: Molecular biology of soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1522-32.
- Singer S, Maki RG, O'Sullivan B: Soft tissue sarcoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1533-77.
- Malawer MM, Helman LJ, O'Sullivan B: Sarcomas of bone. In: DeVita VT Jr, Lawrence TS, Rosenberg SA: Cancer: Principles and Practice of Oncology. 9th ed. Philadelphia, Pa: Lippincott Williams & Wilkins, 2011, pp 1578-1609.
- Midis GP, Pollock RE, Chen NP, et al.: Locally recurrent soft tissue sarcoma of the extremities. Surgery 123 (6): 666-71, 1998. [PUBMED Abstract]
- Essner R, Selch M, Eilber FR: Reirradiation for extremity soft tissue sarcomas. Local control and complications. Cancer 67 (11): 2813-7, 1991. [PUBMED Abstract]
- Singer S, Antman K, Corson JM, et al.: Long-term salvageability for patients with locally recurrent soft-tissue sarcomas. Arch Surg 127 (5): 548-53; discussion 553-4, 1992. [PUBMED Abstract]
- Lewis JJ, Leung D, Heslin M, et al.: Association of local recurrence with subsequent survival in extremity soft tissue sarcoma. J Clin Oncol 15 (2): 646-52, 1997. [PUBMED Abstract]
- van Geel AN, Pastorino U, Jauch KW, et al.: Surgical treatment of lung metastases: The European Organization for Research and Treatment of Cancer-Soft Tissue and Bone Sarcoma Group study of 255 patients. Cancer 77 (4): 675-82, 1996. [PUBMED Abstract]
- Casson AG, Putnam JB, Natarajan G, et al.: Five-year survival after pulmonary metastasectomy for adult soft tissue sarcoma. Cancer 69 (3): 662-8, 1992. [PUBMED Abstract]
- Putnam JB Jr, Roth JA: Surgical treatment for pulmonary metastases from sarcoma. Hematol Oncol Clin North Am 9 (4): 869-87, 1995. [PUBMED Abstract]
- Lorigan P, Verweij J, Papai Z, et al.: Phase III trial of two investigational schedules of ifosfamide compared with standard-dose doxorubicin in advanced or metastatic soft tissue sarcoma: a European Organisation for Research and Treatment of Cancer Soft Tissue and Bone Sarcoma Group Study. J Clin Oncol 25 (21): 3144-50, 2007. [PUBMED Abstract]
- Nielsen OS, Dombernowsky P, Mouridsen H, et al.: High-dose epirubicin is not an alternative to standard-dose doxorubicin in the treatment of advanced soft tissue sarcomas. A study of the EORTC soft tissue and bone sarcoma group. Br J Cancer 78 (12): 1634-9, 1998. [PUBMED Abstract]
- Maki RG, Wathen JK, Patel SR, et al.: Randomized phase II study of gemcitabine and docetaxel compared with gemcitabine alone in patients with metastatic soft tissue sarcomas: results of sarcoma alliance for research through collaboration study 002 [corrected]. J Clin Oncol 25 (19): 2755-63, 2007. [PUBMED Abstract]
- Okuno S, Ryan LM, Edmonson JH, et al.: Phase II trial of gemcitabine in patients with advanced sarcomas (E1797): a trial of the Eastern Cooperative Oncology Group. Cancer 97 (8): 1969-73, 2003. [PUBMED Abstract]
Changes to This Summary (02/01/2018)
The PDQ cancer information summaries are reviewed regularly and updated as new information becomes available. This section describes the latest changes made to this summary as of the date above.
General Information About Adult Soft Tissue Sarcoma
Updated statistics with estimated new cases and deaths for 2018 (cited American Cancer Society as reference 1).
Stage Information for Adult Soft Tissue Sarcoma
An editorial change was made to this section.
This summary is written and maintained by the PDQ Adult Treatment Editorial Board, which is editorially independent of NCI. The summary reflects an independent review of the literature and does not represent a policy statement of NCI or NIH. More information about summary policies and the role of the PDQ Editorial Boards in maintaining the PDQ summaries can be found on the About This PDQ Summary and PDQ® - NCI's Comprehensive Cancer Database pages.
About This PDQ Summary
Purpose of This Summary
This PDQ cancer information summary for health professionals provides comprehensive, peer-reviewed, evidence-based information about the treatment of adult soft tissue sarcoma. It is intended as a resource to inform and assist clinicians who care for cancer patients. It does not provide formal guidelines or recommendations for making health care decisions.
Reviewers and Updates
This summary is reviewed regularly and updated as necessary by the PDQ Adult Treatment Editorial Board, which is editorially independent of the National Cancer Institute (NCI). The summary reflects an independent review of the literature and does not represent a policy statement of NCI or the National Institutes of Health (NIH).
Board members review recently published articles each month to determine whether an article should:
- be discussed at a meeting,
- be cited with text, or
- replace or update an existing article that is already cited.
Changes to the summaries are made through a consensus process in which Board members evaluate the strength of the evidence in the published articles and determine how the article should be included in the summary.
The lead reviewers for Adult Soft Tissue Sarcoma Treatment are:
- Russell S. Berman, MD (New York University School of Medicine)
- Minh Tam Truong, MD (Boston University Medical Center)
Any comments or questions about the summary content should be submitted to Cancer.gov through the NCI website's Email Us. Do not contact the individual Board Members with questions or comments about the summaries. Board members will not respond to individual inquiries.
Levels of Evidence
Some of the reference citations in this summary are accompanied by a level-of-evidence designation. These designations are intended to help readers assess the strength of the evidence supporting the use of specific interventions or approaches. The PDQ Adult Treatment Editorial Board uses a formal evidence ranking system in developing its level-of-evidence designations.
Permission to Use This Summary
PDQ is a registered trademark. Although the content of PDQ documents can be used freely as text, it cannot be identified as an NCI PDQ cancer information summary unless it is presented in its entirety and is regularly updated. However, an author would be permitted to write a sentence such as “NCI’s PDQ cancer information summary about breast cancer prevention states the risks succinctly: [include excerpt from the summary].”
The preferred citation for this PDQ summary is:
PDQ® Adult Treatment Editorial Board. PDQ Adult Soft Tissue Sarcoma Treatment. Bethesda, MD: National Cancer Institute. Updated <MM/DD/YYYY>. Available at: https://www.cancer.gov/types/soft-tissue-sarcoma/hp/adult-soft-tissue-treatment-pdq. Accessed <MM/DD/YYYY>. [PMID: 26389481]
Images in this summary are used with permission of the author(s), artist, and/or publisher for use within the PDQ summaries only. Permission to use images outside the context of PDQ information must be obtained from the owner(s) and cannot be granted by the National Cancer Institute. Information about using the illustrations in this summary, along with many other cancer-related images, is available in Visuals Online, a collection of over 2,000 scientific images.
Disclaimer
Based on the strength of the available evidence, treatment options may be described as either “standard” or “under clinical evaluation.” These classifications should not be used as a basis for insurance reimbursement determinations. More information on insurance coverage is available on Cancer.gov on the Managing Cancer Care page.
Contact Us
More information about contacting us or receiving help with the Cancer.gov website can be found on our Contact Us for Help page. Questions can also be submitted to Cancer.gov through the website’s Email Us.
Updated: February 1, 2018
Source URL: https://www.cancer.gov/publishedcontent/syndication/2127.htm
Source Agency: National Cancer Institute (NCI)
Captured Date: 2013-09-14 09:00:45.0
Dasatinib activity prominent in subset of GIST patients
Dasatinib might have activity in some subsets of patients with imatinib-resistant gastrointestinal stromal tumors (GISTs), investigators have reported.
The tyrosine kinase inhibitor had a 29% rate of 6-month progression-free survival (PFS) in a nonrandomized, 50-patient study.
That PFS rate was well above the 10% threshold that would have constituted evidence of inactive treatment, but it “fell just short of our goal” of 30% that would have been considered evidence of drug activity, wrote Scott M. Schuetze, MD, PhD, of the department of internal medicine, University of Michigan, Ann Arbor, and his coauthors. The report was published in JAMA Oncology.
It was also higher than the 16% 6-month PFS rate reported in a randomized trial of sunitinib, which was approved for imatinib-resistant GIST treatment in 2006. However, it was lower than the 38% 6-month PFS rate reported for regorafenib, which was approved in 2013 for that indication, the researchers noted.
Exploratory analyses did identify a few biomarker-driven subsets that might particularly benefit from dasatinib therapy. Notably, the 6-month PFS rate was 50% for patients with tumors expressing phosphorylated SRC.
While intriguing, the results of the exploratory analyses are hampered by the small number of patients enrolled in the trial; only 14 patients in the study had phosphorylated SRC.
“Further studies should explore whether activated SRC is a prognostic biomarker of more indolent disease, or is a predictive biomarker of response to tyrosine kinase therapy,” the researchers wrote.
Patients in the study had imatinib refractory GIST. They received dasatinib 70 mg twice daily. They were enrolled in 2008-2009 and followed for at least 5 years.
In addition to previously receiving imatinib, most enrollees (80%) had already been treated with sunitinib as well. The study started before the approval of sunitinib in GIST, but after the approval of regorafenib, the investigators noted.
“Preclinical research suggested that dasatinib had higher potency against mutations in the activation domain of KIT and PDGFRA than imatinib and sunitinib,” the authors recounted.
This trial did provide some evidence in support of that preclinical data: One patient with a specific mutation in PDGFRA exhibited prolonged tumor control.
Bristol-Myers Squibb provided funding for the trial and dasatinib. Dr. Schuetze reported disclosures related to Novartis, Amgen, Janssen, Daiichi-Sankyo, Eli Lilly, and AB Science.
SOURCE: Schuetze SM et al. 2018 Apr 26. doi: 10.1001/jamaoncol.2018.0601.
Dasatinib might have activity in some subsets of patients with imatinib-resistant gastrointestinal stromal tumors (GISTs), investigators have reported.
The tyrosine kinase inhibitor had a 29% rate of 6-month progression-free survival (PFS) in a nonrandomized, 50-patient study.
That PFS rate was well above the 10% threshold that would have constituted evidence of inactive treatment, but it “fell just short of our goal” of 30% that would have been considered evidence of drug activity, wrote Scott M. Schuetze, MD, PhD, of the department of internal medicine, University of Michigan, Ann Arbor, and his coauthors. The report was published in JAMA Oncology.
It was also higher than the 16% 6-month PFS rate reported in a randomized trial of sunitinib, which was approved for imatinib-resistant GIST treatment in 2006. However, it was lower than the 38% 6-month PFS rate reported for regorafenib, which was approved in 2013 for that indication, the researchers noted.
Exploratory analyses did identify a few biomarker-driven subsets that might particularly benefit from dasatinib therapy. Notably, the 6-month PFS rate was 50% for patients with tumors expressing phosphorylated SRC.
While intriguing, the results of the exploratory analyses are hampered by the small number of patients enrolled in the trial; only 14 patients in the study had phosphorylated SRC.
“Further studies should explore whether activated SRC is a prognostic biomarker of more indolent disease, or is a predictive biomarker of response to tyrosine kinase therapy,” the researchers wrote.
Patients in the study had imatinib refractory GIST. They received dasatinib 70 mg twice daily. They were enrolled in 2008-2009 and followed for at least 5 years.
In addition to previously receiving imatinib, most enrollees (80%) had already been treated with sunitinib as well. The study started before the approval of sunitinib in GIST, but after the approval of regorafenib, the investigators noted.
“Preclinical research suggested that dasatinib had higher potency against mutations in the activation domain of KIT and PDGFRA than imatinib and sunitinib,” the authors recounted.
This trial did provide some evidence in support of that preclinical data: One patient with a specific mutation in PDGFRA exhibited prolonged tumor control.
Bristol-Myers Squibb provided funding for the trial and dasatinib. Dr. Schuetze reported disclosures related to Novartis, Amgen, Janssen, Daiichi-Sankyo, Eli Lilly, and AB Science.
SOURCE: Schuetze SM et al. 2018 Apr 26. doi: 10.1001/jamaoncol.2018.0601.
Dasatinib might have activity in some subsets of patients with imatinib-resistant gastrointestinal stromal tumors (GISTs), investigators have reported.
The tyrosine kinase inhibitor had a 29% rate of 6-month progression-free survival (PFS) in a nonrandomized, 50-patient study.
That PFS rate was well above the 10% threshold that would have constituted evidence of inactive treatment, but it “fell just short of our goal” of 30% that would have been considered evidence of drug activity, wrote Scott M. Schuetze, MD, PhD, of the department of internal medicine, University of Michigan, Ann Arbor, and his coauthors. The report was published in JAMA Oncology.
It was also higher than the 16% 6-month PFS rate reported in a randomized trial of sunitinib, which was approved for imatinib-resistant GIST treatment in 2006. However, it was lower than the 38% 6-month PFS rate reported for regorafenib, which was approved in 2013 for that indication, the researchers noted.
Exploratory analyses did identify a few biomarker-driven subsets that might particularly benefit from dasatinib therapy. Notably, the 6-month PFS rate was 50% for patients with tumors expressing phosphorylated SRC.
While intriguing, the results of the exploratory analyses are hampered by the small number of patients enrolled in the trial; only 14 patients in the study had phosphorylated SRC.
“Further studies should explore whether activated SRC is a prognostic biomarker of more indolent disease, or is a predictive biomarker of response to tyrosine kinase therapy,” the researchers wrote.
Patients in the study had imatinib refractory GIST. They received dasatinib 70 mg twice daily. They were enrolled in 2008-2009 and followed for at least 5 years.
In addition to previously receiving imatinib, most enrollees (80%) had already been treated with sunitinib as well. The study started before the approval of sunitinib in GIST, but after the approval of regorafenib, the investigators noted.
“Preclinical research suggested that dasatinib had higher potency against mutations in the activation domain of KIT and PDGFRA than imatinib and sunitinib,” the authors recounted.
This trial did provide some evidence in support of that preclinical data: One patient with a specific mutation in PDGFRA exhibited prolonged tumor control.
Bristol-Myers Squibb provided funding for the trial and dasatinib. Dr. Schuetze reported disclosures related to Novartis, Amgen, Janssen, Daiichi-Sankyo, Eli Lilly, and AB Science.
SOURCE: Schuetze SM et al. 2018 Apr 26. doi: 10.1001/jamaoncol.2018.0601.
FROM JAMA ONCOLOGY
Key clinical point: The efficacy of dasatinib in imatinib-resistant GIST was just short of what investigators considered evidence of an active drug.
Major finding: The estimated rate of 6-month progression-free survival was 29% overall, though it was 50% in one biomarker-defined patient subset.
Study details: A nonrandomized single-arm study of 50 patients with GIST treated with dasatinib 70 mg twice daily.
Disclosures: Bristol-Myers Squibb provided funding for the trial and dasatinib. Dr. Schuetze reported disclosures related to Novartis, Amgen, Janssen, Daiichi-Sankyo, Eli Lilly, and AB Science.
Source: Schuetze SM et al. JAMA Oncol. 2018 Apr 26. doi: 10.1001/jamaoncol.2018.0601.
Onodera’s Prognostic Nutritional Index in soft tissue sarcoma patients as a predictor of wound complications
Background The ability to predict a wound complication after radiation therapy and surgery for soft tissue sarcomas remains difficult. Preoperative nutritional status, as determined by Onodera’s Prognostic Nutritional Index (OPNI), has been a predictor of complications in patients undergoing gastrointestinal surgery. However, the role OPNI has in predicting wound complications for soft tissue sarcoma remains unknown.
Objective To evaluate the role OPNI has in predicting wound complication in patients treated with radiation and surgery for soft tissue sarcomas.
Methods OPNI was calculated based on the published formula OPNI = (10*albumin level [g/dL]) + (0.005*total lymphocyte count). The albumin level and total lymphocyte counts closest to the index operation were chosen. Major and minor wound complications were identified. A receiver operating curve was calculated to identify a cut-off point value for OPNI and for age based on the best combination of sensitivity and specificity.
Results 44 patients were included in the study. Patients with an OPNI of <45.4 had a 7.5-times increased risk of a wound complication (P = .005; 95% confidence interval [CI], 1.8-31.0). An OPNI of <45.4 had a sensitivity of 62% and specificity of 82% of predicting a wound complication. Being older than 73 years was associated with a 6.8-times increased risk of wound complications (P = .01; 95% CI, 1.6-28.7).
Limitations Small sample size for patients with a rare condition
Conclusion An OPNI of <45.4 and being older than 73 years are strong predictors of which patients will have a wound complication after radiation therapy for soft tissue sarcomas. Preoperative nutritional status could be an important modifiable factor to help decrease wound complications.
Wound complications after pre- or post-operative radiation for soft tissue sarcomas are well established.1 The ability to predict who will have a wound complication remains difficult. Some studies have looked at risk factors such as smoking, and the preoperative nutritional status of patients has been identified as a risk factor for wound complication in patients with elective orthopedic surgical procedures.2 One validated method of measuring preoperative nutritional status in patients with gastrointestinal malignant tumors has been with Onodera’s Prognostic Nutritional Index (OPNI). It uses the patient’s preoperative albumin (g/dL) and absolute lymphocyte values (per mm3). The prognostic value of the OPNI has been demonstrated in patients with colorectal, esophageal, and gastric cancers, and has been shown to be prognostic for postoperative wound healing and overall prognosis.3-5 In this study, we investigate the significance of preoperative nutritional status, measured by OPNI, as a predictor of wound complications in patients treated with pre- or postoperative radiation for soft tissue sarcoma.
Methods
After receiving Institutional Review Board approval for the study, we conducted a retrospective review of consecutive patients treated during July 2012-April 2016 for a soft tissue sarcoma by the orthopedic oncology division at Cooper University Hospital in Camden, New Jersey. Inclusion criteria were patients with biopsy-proven soft tissue sarcoma, who were older than 18 years, had received pre- or postoperative radiation, and who had a recorded preoperative albumin and total lymphocyte count. A minimum follow-up of 3 months was required to assess for postoperative wound complications. Exclusion criteria included patients who had a bone sarcoma, had not received radiation therapy, or had a missing preoperative albumin or total lymphocyte count.
All of the surgeries were performed by 2 fellowshiptrained orthopedic oncologists. Patients received either pre- or postoperative radiation therapy by multiple radiation oncologists.
The OPNI was calculated based on the published formula OPNI = (10*albumin level [g/dL]) + (0.005*total lymphocyte count [per mm3]). The albumin level and total lymphocyte counts closest to the index operation were chosen.
Demographic information including gender, age at diagnosis, height, and weight were recorded. Data related to the patients’ pathologic diagnosis, stage at presentation, radiation therapy, and surgical resection were collected. A minor wound complication was defined as a wound problem that did not require operative intervention. Major wound complication was defined as a complication requiring operative intervention with or without flap reconstruction. Wound complications occurring within the 3-month postoperative period were considered.
Univariate and multiple variable analysis was performed. A P value <.05 was considered significant. A receiver operating curve as well as recursive partitioning was performed for OPNI and age to determine the best cut-off point to use in the analysis. The Sobel test was used to evaluate mediation. All statistical analysis was performed using SAS v9.4 and JMP10. (SAS Institute, Cary, NC).
Results
In all, 44 patients (28 men, 16 women) were included in the study. Their mean age was 61.2 years (range, 19-94). The average size of the tumors was 8.5 cm in greatest dimension (range, 1.2-27.4 cm), and all of the patients had nonmetastatic disease at the time of surgical resection; 37 patients had R0 resections, and 7 patients had a positive margin from an outside hospital, but obtained R0 resections on a subsequent resection (Table 1 and Table 2). In all, 30 patients received preoperative radiation, 14 patients received postoperative radiation, 32 patients received external beam radiation, 8 received Cyberknife treatment, and information for 4 patients was not unavailable. Mean preoperative external beam radiation and Cyberknife dose was 4,931 Gy and 3,750 Gy, respectively. Mean postoperative external beam and Cyberknife radiation dose was 6,077 Gy and 4,000 Gy, respectively. When evaluating radiation dose delivered between those who had wound complications and those who did not, there was no significant difference (Table 3).
Of the total, 13 patients had a wound complication (30%). Ten patients had preoperative radiation, and 3 had postoperative radiation. Ten patients had major wound complications requiring a combined 27 surgeries. Three patients had minor wound complications, which resolved with conservative management. One patient had a major wound complication in the group that had an initial R1 resection.
The OPNI was calculated based on the aforementioned formula. When the univariate analysis was performed, only age and OPNI were statistically significant. Patients older than 72.6 years had a 6.8 times higher risk of a wound complication (P = .01; 95% confidence interval [CI], 1.6-28.7). When the OPNI value of 45.4 was used as the threshold, a patient with a preoperative OPNI value of <45.4 had a 7.5 times increased risk of developing a wound complication (P = .005; 95% CI, 1.8-31.0).
When the receiver operating curve and recursive partitioning was performed, an OPNI value of 45.4 showed a sensitivity of 62% and specificity of 82% in predicting wound complications (Figure 1).
When a multiple variable analysis was performed, OPNI and age were not statistically significant (P = .06 and P = .11, respectively). A test for mediation was performed, and the OPNI seemed to mediate the effect age has on wound complications, accounting for 36% of the total effect (Sobel test statistic, 1.79; P = .07).
Discussion
Wound complications after pre- and postoperative radiation for soft tissue sarcomas are well known. The best study to date to demonstrate that relationship was a randomized controlled trial performed in Canada, which showed that preoperative radiation resulted in 37% wound complications, compared with 17% for postoperative radiation.6 In that study, of the wound complications in both radiation types, more than 50%-60% required a secondary surgical procedure, designating it as a major wound complication. Other variables that have been shown to contribute to wound complications include being older than 40 years and/or having large tumors, diabetes, peripheral vascular disease, and begin a smoker.7-10
In our study, we applied OPNI to orthopedic oncology and showed that the patient’s age and preoperative nutritional status were significant predictors of developing a wound complication. An OPNI of <45.4 increased the chance of a wound complication by 7.5 times. Being older than 73 years increased the risk of a wound complication by 6.8 times. Most of these wound complications were major and required surgical intervention.
In general surgical oncology, the evaluation of nutritional status has had a significant impact on the care of patients, especially for those patients undergoing gastrointestinal surgery. The OPNI was initially designed to assess the nutritional and immunological statuses of patients undergoing gastrointestinal surgery.11 Preoperative OPNI has been shown to be a good predictor of postoperative complications and survival in patients with colorectal cancer, malignant mesothelioma, hepatocellular carcinoma and in patients who undergo total gastrectomy.12-15 Chen and colleagues evaluated the significance of OPNI in patients with colorectal cancer. They found an optimal cut-off value of 45. An OPNI value <45 has a sensitivity and specificity of 85% and 69%, respectively, in predicting 5-year overall survival.16 Hong and colleagues noted that an OPNI cut-off value of 52.6 as a predictor of overall survival.17
Poor preoperative nutritional status has been shown to have a negative impact on wound healing. In patients who underwent emergency laparotomy, a low OPNI had significantly higher rates of wound dehiscence and infection.18 This happens because protein deficiency leads to decreased wound tensile strength, decreased T-cell function, decreased phagocytic activity, which ultimately diminish the patient’s ability to heal and defend against wound infections.19-21
In soft tissue sarcoma patients, poor preoperative nutritional status is further compromised by radiation therapy to the wound. Gu and colleagues showed that radiation to wounds in mice showed early inhibition of the inflammatory phase, injury and inhibition of fibroblasts, and collagen formation, and then prolonged re-epithelialization.22 This “double hit” with radiation onto host tissue that is already nutritionally compromised could be an important cause of why wound complications occur at such high rates in our soft tissue sarcoma patients.
There are several limitations to this study. First, the study has a small sample size, which was a direct result of the number of patients who were excluded because an OPNI value could not be calculated for them. Second, we could not determine if the OPNI was more valuable in patients who underwent pre- or postoperative radiation. This study did not look at other nutritional indices such as prealbumin and vitamin levels. Third, the radiation was provided by different providers, so technique was variable, but the patients received nearly equivalent doses and variability in technique is likely limited. Fourth, we were not able to meaningfully analyze the role of chemotherapy in this patient population because there was a significant heterogeneity of patients receiving pre- and postoperative chemotherapy.
Our findings strongly suggest that a preoperative OPNI of <45.4 and being older than 73 years are strong predictors of patients who will experience a wound complication after radiation therapy for soft tissue sarcomas. This study has led us to start measuring preoperative albumin levels and assess complete metabolic panels. Our goal is to identify patients who are at high risk of wound complication and perform interventions to improve nutrition, then to study whether the interventions help lower the rates of wound complications. TSJ
Correspondence
References
1. Ormsby MV, Hilaris BS, Nori D, Brennan MF. Wound complications of adjuvant radiation therapy in patients with soft-tissue sarcomas. Ann Surg. 1989;210(1):93-99.
2. Greene KA, Wilde AH, Stulberg BN. Preoperative nutritional status of total joint patients: relationship to postoperative wound complications. J Arthroplasty. 1991;6(4):321-325.
3. Nozoe T, Kimura Y, Ishida M, Saeki H, Korenaga D, Sugimachi K. Correlation of pre-operative nutritional condition with post-operative complications in surgical treatment for oesophageal carcinoma. Eur J Surg Oncol. 2002;28(4):396-400.
4. Nozoe T, Kohno M, Iguchi T, et al. The prognostic nutritional index can be a prognostic indicator in colorectal carcinoma. Surg Today. 2012;42(6):532-535.
5. Nozoe T, Ninomiya M, Maeda T, Matsukuma A, Nakashima H, Ezaki T. Prognostic nutritional index: a tool to predict the biological aggressiveness of gastric carcinoma. Surg Today. 2010;40(5):440-443.
6. O’Sullivan B, Davis AM, Turcotte R, Bell R, Catton C, Chabot P, et al. Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: a randomised trial. Lancet. 2002;359(9325):2235-2241.
7. Peat BG, Bell RS, Davis A, et al. Wound-healing complications after soft-tissue sarcoma surgery. Plast Reconstr Surg. 1994;93(5):980-987.
8. Kunisada T, Ngan SY, Powell G, Choong PF. Wound complications following pre-operative radiotherapy for soft tissue sarcoma. Eur J Surg Oncol. 2002;28(1):75-79.
9. Saddegh MK, Bauer HC. Wound complication in surgery of soft tissue sarcoma: analysis of 103 consecutive patients managed without adjuvant therapy. Clin Orthop Relat Res. 1993;289:247-253.
10. Tseng JF, Ballo MT, Langstein HN, et al. The effect of preoperative radiotherapy and reconstructive surgery on wound complications after resection of extremity soft-tissue sarcomas. Ann Surg Oncol. 2006;13(9):1209-1215.
11. Smale BF, Mullen JL, Buzby GP, Rosato EF. The efficacy of nutritional assessment and support in cancer surgery. Cancer. 1981;47(10):2375-2381.
12. Mohri Y, Inoue Y, Tanaka K, Hiro J, Uchida K, Kusunoki M. Prognostic nutritional index predicts postoperative outcome in colorectal cancer. World J Surg. 2013;37(11):2688-2692.
13. Jiang N, Deng JY, Ding XW, et al. Prognostic nutritional index predicts postoperative complications and long-term outcomes of gastric cancer. World J Gastroenterol. 2014;20(30):10537-10544.
14. Pinato DJ, North BV, Sharma R. A novel, externally validated inflammation-based prognostic algorithm in hepatocellular carcinoma: the prognostic nutritional index (PNI). Brit J Cancer. 2012;106(8):1439-1445.
15. Yao ZH, Tian GY, Wan YY, et al. Prognostic nutritional index predicts outcomes of malignant pleural mesothelioma. J Cancer Res Clin Oncol. 2013;139(12):2117-2123.
16. Jian-Hui C, Iskandar EA, Cai Sh I, et al. Significance of Onodera’s prognostic nutritional index in patients with colorectal cancer: a large cohort study in a single Chinese institution. Tumour Biol. 2016;37(3):3277-3283.
17. Hong S, Zhou T, Fang W, et al. The prognostic nutritional index (PNI) predicts overall survival of small-cell lung cancer patients. Tumour Biol. 2015;36(5):3389-9337.
18. Mohil RS, Agarwal A, Singh N, Arora J, Bhatnagar D. Does nutritional status play a role in patients undergoing emergency laparotomy? E Spen Eur E J Clin Nutr Metab. 2008;3(5):e226-e231.
19. Kay SP, Moreland JR, Schmitter E. Nutritional status and wound healing in lower extremity amputations. Clin Orthop Relat Res. 1987;(217):253-256.
20. Dickhaut SC, DeLee JC, Page CP. Nutritional status: importance in predicting wound-healing after amputation. J Bone Joint Surg Am. 1984;66(1):71-75.
21. Casey J, Flinn WR, Yao JS, Fahey V, Pawlowski J, Bergan JJ. Correlation of immune and nutritional status with wound complications in patients undergoing vascular operations. Surgery. 1983;93(6):822-827.
22. Gu Q, Wang D, Cui C, Gao Y, Xia G, Cui X. Effects of radiation on wound healing. J Environ Pathol Toxicol Oncol. 1998;17(2):117-123.
Background The ability to predict a wound complication after radiation therapy and surgery for soft tissue sarcomas remains difficult. Preoperative nutritional status, as determined by Onodera’s Prognostic Nutritional Index (OPNI), has been a predictor of complications in patients undergoing gastrointestinal surgery. However, the role OPNI has in predicting wound complications for soft tissue sarcoma remains unknown.
Objective To evaluate the role OPNI has in predicting wound complication in patients treated with radiation and surgery for soft tissue sarcomas.
Methods OPNI was calculated based on the published formula OPNI = (10*albumin level [g/dL]) + (0.005*total lymphocyte count). The albumin level and total lymphocyte counts closest to the index operation were chosen. Major and minor wound complications were identified. A receiver operating curve was calculated to identify a cut-off point value for OPNI and for age based on the best combination of sensitivity and specificity.
Results 44 patients were included in the study. Patients with an OPNI of <45.4 had a 7.5-times increased risk of a wound complication (P = .005; 95% confidence interval [CI], 1.8-31.0). An OPNI of <45.4 had a sensitivity of 62% and specificity of 82% of predicting a wound complication. Being older than 73 years was associated with a 6.8-times increased risk of wound complications (P = .01; 95% CI, 1.6-28.7).
Limitations Small sample size for patients with a rare condition
Conclusion An OPNI of <45.4 and being older than 73 years are strong predictors of which patients will have a wound complication after radiation therapy for soft tissue sarcomas. Preoperative nutritional status could be an important modifiable factor to help decrease wound complications.
Wound complications after pre- or post-operative radiation for soft tissue sarcomas are well established.1 The ability to predict who will have a wound complication remains difficult. Some studies have looked at risk factors such as smoking, and the preoperative nutritional status of patients has been identified as a risk factor for wound complication in patients with elective orthopedic surgical procedures.2 One validated method of measuring preoperative nutritional status in patients with gastrointestinal malignant tumors has been with Onodera’s Prognostic Nutritional Index (OPNI). It uses the patient’s preoperative albumin (g/dL) and absolute lymphocyte values (per mm3). The prognostic value of the OPNI has been demonstrated in patients with colorectal, esophageal, and gastric cancers, and has been shown to be prognostic for postoperative wound healing and overall prognosis.3-5 In this study, we investigate the significance of preoperative nutritional status, measured by OPNI, as a predictor of wound complications in patients treated with pre- or postoperative radiation for soft tissue sarcoma.
Methods
After receiving Institutional Review Board approval for the study, we conducted a retrospective review of consecutive patients treated during July 2012-April 2016 for a soft tissue sarcoma by the orthopedic oncology division at Cooper University Hospital in Camden, New Jersey. Inclusion criteria were patients with biopsy-proven soft tissue sarcoma, who were older than 18 years, had received pre- or postoperative radiation, and who had a recorded preoperative albumin and total lymphocyte count. A minimum follow-up of 3 months was required to assess for postoperative wound complications. Exclusion criteria included patients who had a bone sarcoma, had not received radiation therapy, or had a missing preoperative albumin or total lymphocyte count.
All of the surgeries were performed by 2 fellowshiptrained orthopedic oncologists. Patients received either pre- or postoperative radiation therapy by multiple radiation oncologists.
The OPNI was calculated based on the published formula OPNI = (10*albumin level [g/dL]) + (0.005*total lymphocyte count [per mm3]). The albumin level and total lymphocyte counts closest to the index operation were chosen.
Demographic information including gender, age at diagnosis, height, and weight were recorded. Data related to the patients’ pathologic diagnosis, stage at presentation, radiation therapy, and surgical resection were collected. A minor wound complication was defined as a wound problem that did not require operative intervention. Major wound complication was defined as a complication requiring operative intervention with or without flap reconstruction. Wound complications occurring within the 3-month postoperative period were considered.
Univariate and multiple variable analysis was performed. A P value <.05 was considered significant. A receiver operating curve as well as recursive partitioning was performed for OPNI and age to determine the best cut-off point to use in the analysis. The Sobel test was used to evaluate mediation. All statistical analysis was performed using SAS v9.4 and JMP10. (SAS Institute, Cary, NC).
Results
In all, 44 patients (28 men, 16 women) were included in the study. Their mean age was 61.2 years (range, 19-94). The average size of the tumors was 8.5 cm in greatest dimension (range, 1.2-27.4 cm), and all of the patients had nonmetastatic disease at the time of surgical resection; 37 patients had R0 resections, and 7 patients had a positive margin from an outside hospital, but obtained R0 resections on a subsequent resection (Table 1 and Table 2). In all, 30 patients received preoperative radiation, 14 patients received postoperative radiation, 32 patients received external beam radiation, 8 received Cyberknife treatment, and information for 4 patients was not unavailable. Mean preoperative external beam radiation and Cyberknife dose was 4,931 Gy and 3,750 Gy, respectively. Mean postoperative external beam and Cyberknife radiation dose was 6,077 Gy and 4,000 Gy, respectively. When evaluating radiation dose delivered between those who had wound complications and those who did not, there was no significant difference (Table 3).
Of the total, 13 patients had a wound complication (30%). Ten patients had preoperative radiation, and 3 had postoperative radiation. Ten patients had major wound complications requiring a combined 27 surgeries. Three patients had minor wound complications, which resolved with conservative management. One patient had a major wound complication in the group that had an initial R1 resection.
The OPNI was calculated based on the aforementioned formula. When the univariate analysis was performed, only age and OPNI were statistically significant. Patients older than 72.6 years had a 6.8 times higher risk of a wound complication (P = .01; 95% confidence interval [CI], 1.6-28.7). When the OPNI value of 45.4 was used as the threshold, a patient with a preoperative OPNI value of <45.4 had a 7.5 times increased risk of developing a wound complication (P = .005; 95% CI, 1.8-31.0).
When the receiver operating curve and recursive partitioning was performed, an OPNI value of 45.4 showed a sensitivity of 62% and specificity of 82% in predicting wound complications (Figure 1).
When a multiple variable analysis was performed, OPNI and age were not statistically significant (P = .06 and P = .11, respectively). A test for mediation was performed, and the OPNI seemed to mediate the effect age has on wound complications, accounting for 36% of the total effect (Sobel test statistic, 1.79; P = .07).
Discussion
Wound complications after pre- and postoperative radiation for soft tissue sarcomas are well known. The best study to date to demonstrate that relationship was a randomized controlled trial performed in Canada, which showed that preoperative radiation resulted in 37% wound complications, compared with 17% for postoperative radiation.6 In that study, of the wound complications in both radiation types, more than 50%-60% required a secondary surgical procedure, designating it as a major wound complication. Other variables that have been shown to contribute to wound complications include being older than 40 years and/or having large tumors, diabetes, peripheral vascular disease, and begin a smoker.7-10
In our study, we applied OPNI to orthopedic oncology and showed that the patient’s age and preoperative nutritional status were significant predictors of developing a wound complication. An OPNI of <45.4 increased the chance of a wound complication by 7.5 times. Being older than 73 years increased the risk of a wound complication by 6.8 times. Most of these wound complications were major and required surgical intervention.
In general surgical oncology, the evaluation of nutritional status has had a significant impact on the care of patients, especially for those patients undergoing gastrointestinal surgery. The OPNI was initially designed to assess the nutritional and immunological statuses of patients undergoing gastrointestinal surgery.11 Preoperative OPNI has been shown to be a good predictor of postoperative complications and survival in patients with colorectal cancer, malignant mesothelioma, hepatocellular carcinoma and in patients who undergo total gastrectomy.12-15 Chen and colleagues evaluated the significance of OPNI in patients with colorectal cancer. They found an optimal cut-off value of 45. An OPNI value <45 has a sensitivity and specificity of 85% and 69%, respectively, in predicting 5-year overall survival.16 Hong and colleagues noted that an OPNI cut-off value of 52.6 as a predictor of overall survival.17
Poor preoperative nutritional status has been shown to have a negative impact on wound healing. In patients who underwent emergency laparotomy, a low OPNI had significantly higher rates of wound dehiscence and infection.18 This happens because protein deficiency leads to decreased wound tensile strength, decreased T-cell function, decreased phagocytic activity, which ultimately diminish the patient’s ability to heal and defend against wound infections.19-21
In soft tissue sarcoma patients, poor preoperative nutritional status is further compromised by radiation therapy to the wound. Gu and colleagues showed that radiation to wounds in mice showed early inhibition of the inflammatory phase, injury and inhibition of fibroblasts, and collagen formation, and then prolonged re-epithelialization.22 This “double hit” with radiation onto host tissue that is already nutritionally compromised could be an important cause of why wound complications occur at such high rates in our soft tissue sarcoma patients.
There are several limitations to this study. First, the study has a small sample size, which was a direct result of the number of patients who were excluded because an OPNI value could not be calculated for them. Second, we could not determine if the OPNI was more valuable in patients who underwent pre- or postoperative radiation. This study did not look at other nutritional indices such as prealbumin and vitamin levels. Third, the radiation was provided by different providers, so technique was variable, but the patients received nearly equivalent doses and variability in technique is likely limited. Fourth, we were not able to meaningfully analyze the role of chemotherapy in this patient population because there was a significant heterogeneity of patients receiving pre- and postoperative chemotherapy.
Our findings strongly suggest that a preoperative OPNI of <45.4 and being older than 73 years are strong predictors of patients who will experience a wound complication after radiation therapy for soft tissue sarcomas. This study has led us to start measuring preoperative albumin levels and assess complete metabolic panels. Our goal is to identify patients who are at high risk of wound complication and perform interventions to improve nutrition, then to study whether the interventions help lower the rates of wound complications. TSJ
Correspondence
References
1. Ormsby MV, Hilaris BS, Nori D, Brennan MF. Wound complications of adjuvant radiation therapy in patients with soft-tissue sarcomas. Ann Surg. 1989;210(1):93-99.
2. Greene KA, Wilde AH, Stulberg BN. Preoperative nutritional status of total joint patients: relationship to postoperative wound complications. J Arthroplasty. 1991;6(4):321-325.
3. Nozoe T, Kimura Y, Ishida M, Saeki H, Korenaga D, Sugimachi K. Correlation of pre-operative nutritional condition with post-operative complications in surgical treatment for oesophageal carcinoma. Eur J Surg Oncol. 2002;28(4):396-400.
4. Nozoe T, Kohno M, Iguchi T, et al. The prognostic nutritional index can be a prognostic indicator in colorectal carcinoma. Surg Today. 2012;42(6):532-535.
5. Nozoe T, Ninomiya M, Maeda T, Matsukuma A, Nakashima H, Ezaki T. Prognostic nutritional index: a tool to predict the biological aggressiveness of gastric carcinoma. Surg Today. 2010;40(5):440-443.
6. O’Sullivan B, Davis AM, Turcotte R, Bell R, Catton C, Chabot P, et al. Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: a randomised trial. Lancet. 2002;359(9325):2235-2241.
7. Peat BG, Bell RS, Davis A, et al. Wound-healing complications after soft-tissue sarcoma surgery. Plast Reconstr Surg. 1994;93(5):980-987.
8. Kunisada T, Ngan SY, Powell G, Choong PF. Wound complications following pre-operative radiotherapy for soft tissue sarcoma. Eur J Surg Oncol. 2002;28(1):75-79.
9. Saddegh MK, Bauer HC. Wound complication in surgery of soft tissue sarcoma: analysis of 103 consecutive patients managed without adjuvant therapy. Clin Orthop Relat Res. 1993;289:247-253.
10. Tseng JF, Ballo MT, Langstein HN, et al. The effect of preoperative radiotherapy and reconstructive surgery on wound complications after resection of extremity soft-tissue sarcomas. Ann Surg Oncol. 2006;13(9):1209-1215.
11. Smale BF, Mullen JL, Buzby GP, Rosato EF. The efficacy of nutritional assessment and support in cancer surgery. Cancer. 1981;47(10):2375-2381.
12. Mohri Y, Inoue Y, Tanaka K, Hiro J, Uchida K, Kusunoki M. Prognostic nutritional index predicts postoperative outcome in colorectal cancer. World J Surg. 2013;37(11):2688-2692.
13. Jiang N, Deng JY, Ding XW, et al. Prognostic nutritional index predicts postoperative complications and long-term outcomes of gastric cancer. World J Gastroenterol. 2014;20(30):10537-10544.
14. Pinato DJ, North BV, Sharma R. A novel, externally validated inflammation-based prognostic algorithm in hepatocellular carcinoma: the prognostic nutritional index (PNI). Brit J Cancer. 2012;106(8):1439-1445.
15. Yao ZH, Tian GY, Wan YY, et al. Prognostic nutritional index predicts outcomes of malignant pleural mesothelioma. J Cancer Res Clin Oncol. 2013;139(12):2117-2123.
16. Jian-Hui C, Iskandar EA, Cai Sh I, et al. Significance of Onodera’s prognostic nutritional index in patients with colorectal cancer: a large cohort study in a single Chinese institution. Tumour Biol. 2016;37(3):3277-3283.
17. Hong S, Zhou T, Fang W, et al. The prognostic nutritional index (PNI) predicts overall survival of small-cell lung cancer patients. Tumour Biol. 2015;36(5):3389-9337.
18. Mohil RS, Agarwal A, Singh N, Arora J, Bhatnagar D. Does nutritional status play a role in patients undergoing emergency laparotomy? E Spen Eur E J Clin Nutr Metab. 2008;3(5):e226-e231.
19. Kay SP, Moreland JR, Schmitter E. Nutritional status and wound healing in lower extremity amputations. Clin Orthop Relat Res. 1987;(217):253-256.
20. Dickhaut SC, DeLee JC, Page CP. Nutritional status: importance in predicting wound-healing after amputation. J Bone Joint Surg Am. 1984;66(1):71-75.
21. Casey J, Flinn WR, Yao JS, Fahey V, Pawlowski J, Bergan JJ. Correlation of immune and nutritional status with wound complications in patients undergoing vascular operations. Surgery. 1983;93(6):822-827.
22. Gu Q, Wang D, Cui C, Gao Y, Xia G, Cui X. Effects of radiation on wound healing. J Environ Pathol Toxicol Oncol. 1998;17(2):117-123.
Background The ability to predict a wound complication after radiation therapy and surgery for soft tissue sarcomas remains difficult. Preoperative nutritional status, as determined by Onodera’s Prognostic Nutritional Index (OPNI), has been a predictor of complications in patients undergoing gastrointestinal surgery. However, the role OPNI has in predicting wound complications for soft tissue sarcoma remains unknown.
Objective To evaluate the role OPNI has in predicting wound complication in patients treated with radiation and surgery for soft tissue sarcomas.
Methods OPNI was calculated based on the published formula OPNI = (10*albumin level [g/dL]) + (0.005*total lymphocyte count). The albumin level and total lymphocyte counts closest to the index operation were chosen. Major and minor wound complications were identified. A receiver operating curve was calculated to identify a cut-off point value for OPNI and for age based on the best combination of sensitivity and specificity.
Results 44 patients were included in the study. Patients with an OPNI of <45.4 had a 7.5-times increased risk of a wound complication (P = .005; 95% confidence interval [CI], 1.8-31.0). An OPNI of <45.4 had a sensitivity of 62% and specificity of 82% of predicting a wound complication. Being older than 73 years was associated with a 6.8-times increased risk of wound complications (P = .01; 95% CI, 1.6-28.7).
Limitations Small sample size for patients with a rare condition
Conclusion An OPNI of <45.4 and being older than 73 years are strong predictors of which patients will have a wound complication after radiation therapy for soft tissue sarcomas. Preoperative nutritional status could be an important modifiable factor to help decrease wound complications.
Wound complications after pre- or post-operative radiation for soft tissue sarcomas are well established.1 The ability to predict who will have a wound complication remains difficult. Some studies have looked at risk factors such as smoking, and the preoperative nutritional status of patients has been identified as a risk factor for wound complication in patients with elective orthopedic surgical procedures.2 One validated method of measuring preoperative nutritional status in patients with gastrointestinal malignant tumors has been with Onodera’s Prognostic Nutritional Index (OPNI). It uses the patient’s preoperative albumin (g/dL) and absolute lymphocyte values (per mm3). The prognostic value of the OPNI has been demonstrated in patients with colorectal, esophageal, and gastric cancers, and has been shown to be prognostic for postoperative wound healing and overall prognosis.3-5 In this study, we investigate the significance of preoperative nutritional status, measured by OPNI, as a predictor of wound complications in patients treated with pre- or postoperative radiation for soft tissue sarcoma.
Methods
After receiving Institutional Review Board approval for the study, we conducted a retrospective review of consecutive patients treated during July 2012-April 2016 for a soft tissue sarcoma by the orthopedic oncology division at Cooper University Hospital in Camden, New Jersey. Inclusion criteria were patients with biopsy-proven soft tissue sarcoma, who were older than 18 years, had received pre- or postoperative radiation, and who had a recorded preoperative albumin and total lymphocyte count. A minimum follow-up of 3 months was required to assess for postoperative wound complications. Exclusion criteria included patients who had a bone sarcoma, had not received radiation therapy, or had a missing preoperative albumin or total lymphocyte count.
All of the surgeries were performed by 2 fellowshiptrained orthopedic oncologists. Patients received either pre- or postoperative radiation therapy by multiple radiation oncologists.
The OPNI was calculated based on the published formula OPNI = (10*albumin level [g/dL]) + (0.005*total lymphocyte count [per mm3]). The albumin level and total lymphocyte counts closest to the index operation were chosen.
Demographic information including gender, age at diagnosis, height, and weight were recorded. Data related to the patients’ pathologic diagnosis, stage at presentation, radiation therapy, and surgical resection were collected. A minor wound complication was defined as a wound problem that did not require operative intervention. Major wound complication was defined as a complication requiring operative intervention with or without flap reconstruction. Wound complications occurring within the 3-month postoperative period were considered.
Univariate and multiple variable analysis was performed. A P value <.05 was considered significant. A receiver operating curve as well as recursive partitioning was performed for OPNI and age to determine the best cut-off point to use in the analysis. The Sobel test was used to evaluate mediation. All statistical analysis was performed using SAS v9.4 and JMP10. (SAS Institute, Cary, NC).
Results
In all, 44 patients (28 men, 16 women) were included in the study. Their mean age was 61.2 years (range, 19-94). The average size of the tumors was 8.5 cm in greatest dimension (range, 1.2-27.4 cm), and all of the patients had nonmetastatic disease at the time of surgical resection; 37 patients had R0 resections, and 7 patients had a positive margin from an outside hospital, but obtained R0 resections on a subsequent resection (Table 1 and Table 2). In all, 30 patients received preoperative radiation, 14 patients received postoperative radiation, 32 patients received external beam radiation, 8 received Cyberknife treatment, and information for 4 patients was not unavailable. Mean preoperative external beam radiation and Cyberknife dose was 4,931 Gy and 3,750 Gy, respectively. Mean postoperative external beam and Cyberknife radiation dose was 6,077 Gy and 4,000 Gy, respectively. When evaluating radiation dose delivered between those who had wound complications and those who did not, there was no significant difference (Table 3).
Of the total, 13 patients had a wound complication (30%). Ten patients had preoperative radiation, and 3 had postoperative radiation. Ten patients had major wound complications requiring a combined 27 surgeries. Three patients had minor wound complications, which resolved with conservative management. One patient had a major wound complication in the group that had an initial R1 resection.
The OPNI was calculated based on the aforementioned formula. When the univariate analysis was performed, only age and OPNI were statistically significant. Patients older than 72.6 years had a 6.8 times higher risk of a wound complication (P = .01; 95% confidence interval [CI], 1.6-28.7). When the OPNI value of 45.4 was used as the threshold, a patient with a preoperative OPNI value of <45.4 had a 7.5 times increased risk of developing a wound complication (P = .005; 95% CI, 1.8-31.0).
When the receiver operating curve and recursive partitioning was performed, an OPNI value of 45.4 showed a sensitivity of 62% and specificity of 82% in predicting wound complications (Figure 1).
When a multiple variable analysis was performed, OPNI and age were not statistically significant (P = .06 and P = .11, respectively). A test for mediation was performed, and the OPNI seemed to mediate the effect age has on wound complications, accounting for 36% of the total effect (Sobel test statistic, 1.79; P = .07).
Discussion
Wound complications after pre- and postoperative radiation for soft tissue sarcomas are well known. The best study to date to demonstrate that relationship was a randomized controlled trial performed in Canada, which showed that preoperative radiation resulted in 37% wound complications, compared with 17% for postoperative radiation.6 In that study, of the wound complications in both radiation types, more than 50%-60% required a secondary surgical procedure, designating it as a major wound complication. Other variables that have been shown to contribute to wound complications include being older than 40 years and/or having large tumors, diabetes, peripheral vascular disease, and begin a smoker.7-10
In our study, we applied OPNI to orthopedic oncology and showed that the patient’s age and preoperative nutritional status were significant predictors of developing a wound complication. An OPNI of <45.4 increased the chance of a wound complication by 7.5 times. Being older than 73 years increased the risk of a wound complication by 6.8 times. Most of these wound complications were major and required surgical intervention.
In general surgical oncology, the evaluation of nutritional status has had a significant impact on the care of patients, especially for those patients undergoing gastrointestinal surgery. The OPNI was initially designed to assess the nutritional and immunological statuses of patients undergoing gastrointestinal surgery.11 Preoperative OPNI has been shown to be a good predictor of postoperative complications and survival in patients with colorectal cancer, malignant mesothelioma, hepatocellular carcinoma and in patients who undergo total gastrectomy.12-15 Chen and colleagues evaluated the significance of OPNI in patients with colorectal cancer. They found an optimal cut-off value of 45. An OPNI value <45 has a sensitivity and specificity of 85% and 69%, respectively, in predicting 5-year overall survival.16 Hong and colleagues noted that an OPNI cut-off value of 52.6 as a predictor of overall survival.17
Poor preoperative nutritional status has been shown to have a negative impact on wound healing. In patients who underwent emergency laparotomy, a low OPNI had significantly higher rates of wound dehiscence and infection.18 This happens because protein deficiency leads to decreased wound tensile strength, decreased T-cell function, decreased phagocytic activity, which ultimately diminish the patient’s ability to heal and defend against wound infections.19-21
In soft tissue sarcoma patients, poor preoperative nutritional status is further compromised by radiation therapy to the wound. Gu and colleagues showed that radiation to wounds in mice showed early inhibition of the inflammatory phase, injury and inhibition of fibroblasts, and collagen formation, and then prolonged re-epithelialization.22 This “double hit” with radiation onto host tissue that is already nutritionally compromised could be an important cause of why wound complications occur at such high rates in our soft tissue sarcoma patients.
There are several limitations to this study. First, the study has a small sample size, which was a direct result of the number of patients who were excluded because an OPNI value could not be calculated for them. Second, we could not determine if the OPNI was more valuable in patients who underwent pre- or postoperative radiation. This study did not look at other nutritional indices such as prealbumin and vitamin levels. Third, the radiation was provided by different providers, so technique was variable, but the patients received nearly equivalent doses and variability in technique is likely limited. Fourth, we were not able to meaningfully analyze the role of chemotherapy in this patient population because there was a significant heterogeneity of patients receiving pre- and postoperative chemotherapy.
Our findings strongly suggest that a preoperative OPNI of <45.4 and being older than 73 years are strong predictors of patients who will experience a wound complication after radiation therapy for soft tissue sarcomas. This study has led us to start measuring preoperative albumin levels and assess complete metabolic panels. Our goal is to identify patients who are at high risk of wound complication and perform interventions to improve nutrition, then to study whether the interventions help lower the rates of wound complications. TSJ
Correspondence
References
1. Ormsby MV, Hilaris BS, Nori D, Brennan MF. Wound complications of adjuvant radiation therapy in patients with soft-tissue sarcomas. Ann Surg. 1989;210(1):93-99.
2. Greene KA, Wilde AH, Stulberg BN. Preoperative nutritional status of total joint patients: relationship to postoperative wound complications. J Arthroplasty. 1991;6(4):321-325.
3. Nozoe T, Kimura Y, Ishida M, Saeki H, Korenaga D, Sugimachi K. Correlation of pre-operative nutritional condition with post-operative complications in surgical treatment for oesophageal carcinoma. Eur J Surg Oncol. 2002;28(4):396-400.
4. Nozoe T, Kohno M, Iguchi T, et al. The prognostic nutritional index can be a prognostic indicator in colorectal carcinoma. Surg Today. 2012;42(6):532-535.
5. Nozoe T, Ninomiya M, Maeda T, Matsukuma A, Nakashima H, Ezaki T. Prognostic nutritional index: a tool to predict the biological aggressiveness of gastric carcinoma. Surg Today. 2010;40(5):440-443.
6. O’Sullivan B, Davis AM, Turcotte R, Bell R, Catton C, Chabot P, et al. Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: a randomised trial. Lancet. 2002;359(9325):2235-2241.
7. Peat BG, Bell RS, Davis A, et al. Wound-healing complications after soft-tissue sarcoma surgery. Plast Reconstr Surg. 1994;93(5):980-987.
8. Kunisada T, Ngan SY, Powell G, Choong PF. Wound complications following pre-operative radiotherapy for soft tissue sarcoma. Eur J Surg Oncol. 2002;28(1):75-79.
9. Saddegh MK, Bauer HC. Wound complication in surgery of soft tissue sarcoma: analysis of 103 consecutive patients managed without adjuvant therapy. Clin Orthop Relat Res. 1993;289:247-253.
10. Tseng JF, Ballo MT, Langstein HN, et al. The effect of preoperative radiotherapy and reconstructive surgery on wound complications after resection of extremity soft-tissue sarcomas. Ann Surg Oncol. 2006;13(9):1209-1215.
11. Smale BF, Mullen JL, Buzby GP, Rosato EF. The efficacy of nutritional assessment and support in cancer surgery. Cancer. 1981;47(10):2375-2381.
12. Mohri Y, Inoue Y, Tanaka K, Hiro J, Uchida K, Kusunoki M. Prognostic nutritional index predicts postoperative outcome in colorectal cancer. World J Surg. 2013;37(11):2688-2692.
13. Jiang N, Deng JY, Ding XW, et al. Prognostic nutritional index predicts postoperative complications and long-term outcomes of gastric cancer. World J Gastroenterol. 2014;20(30):10537-10544.
14. Pinato DJ, North BV, Sharma R. A novel, externally validated inflammation-based prognostic algorithm in hepatocellular carcinoma: the prognostic nutritional index (PNI). Brit J Cancer. 2012;106(8):1439-1445.
15. Yao ZH, Tian GY, Wan YY, et al. Prognostic nutritional index predicts outcomes of malignant pleural mesothelioma. J Cancer Res Clin Oncol. 2013;139(12):2117-2123.
16. Jian-Hui C, Iskandar EA, Cai Sh I, et al. Significance of Onodera’s prognostic nutritional index in patients with colorectal cancer: a large cohort study in a single Chinese institution. Tumour Biol. 2016;37(3):3277-3283.
17. Hong S, Zhou T, Fang W, et al. The prognostic nutritional index (PNI) predicts overall survival of small-cell lung cancer patients. Tumour Biol. 2015;36(5):3389-9337.
18. Mohil RS, Agarwal A, Singh N, Arora J, Bhatnagar D. Does nutritional status play a role in patients undergoing emergency laparotomy? E Spen Eur E J Clin Nutr Metab. 2008;3(5):e226-e231.
19. Kay SP, Moreland JR, Schmitter E. Nutritional status and wound healing in lower extremity amputations. Clin Orthop Relat Res. 1987;(217):253-256.
20. Dickhaut SC, DeLee JC, Page CP. Nutritional status: importance in predicting wound-healing after amputation. J Bone Joint Surg Am. 1984;66(1):71-75.
21. Casey J, Flinn WR, Yao JS, Fahey V, Pawlowski J, Bergan JJ. Correlation of immune and nutritional status with wound complications in patients undergoing vascular operations. Surgery. 1983;93(6):822-827.
22. Gu Q, Wang D, Cui C, Gao Y, Xia G, Cui X. Effects of radiation on wound healing. J Environ Pathol Toxicol Oncol. 1998;17(2):117-123.