User login
Research ties gout in women to comorbidities more than genetics
Comorbidities may play a greater role than genetics women with gout, although this appears not to be true for men, Nicholas Sumpter, MSc, of the University of Alabama at Birmingham said at the annual research symposium of the Gout, Hyperuricemia, and Crystal Associated Disease Network (G-CAN).
Mr. Sumpter was among the authors of a recent paper in Arthritis & Rheumatology that suggested that earlier gout onset involves the accumulation of certain allelic variants in men. This genetic risk was shared across multiple ancestral groups in the study, conducted with men of European and Polynesian ancestry, Mr. Sumpter and colleagues reported.
“There might be more than one factor in gout in men, but in women we’ve been getting at this idea that comorbidities are the big thing,” he said.
During his presentation, Mr. Sumpter offered a hypothesis that in men there might be a kind of “two-pronged attack,” with increases in serum urate linked to genetic risk, but comorbidities also playing a role. “But that may not be the case for women.”
In his presentation, Mr. Sumpter noted a paper published in March 2022 from his University of Alabama at Birmingham colleagues, Aakash V. Patel, MD, and Angelo L. Gaffo, MD. In the article, Dr. Patel and Dr. Gaffo delved into the challenges of treating women with gout given “the paucity of appropriately well-powered, randomized-controlled trials investigating the efficacy” of commonly used treatments.
“This poses major challenges for the management of female gout patients since they carry a greater burden of cardiovascular and renal morbidity, which is known to modulate the pathophysiology of gout; as such, conclusions regarding the efficacy of treatments for females cannot be extrapolated from investigative studies that are predominantly male,” they wrote, calling for increased efforts to enroll women in studies of treatments for this condition.
There’s increased interest in how gout affects women, including findings in a paper published in September in Arthritis & Rheumatology that found people with gout, especially women, appear to be at higher risk for poor COVID-19 outcomes, including hospitalization and death, regardless of COVID-19 vaccination status.
Gout has become more common in women, although this remains a condition that is far more likely to strike men.
The age-standardized prevalence of gout among women rose from 233.52 per 100,000 in 1990 to 253.49 in 2017, a gain of about 9%, according to a systematic analysis of the Global Burden of Disease Study.
That topped the roughly 5% gain seen for men in the same time frame, with the rate going from 747.48 per 100,000 to 790.90. With the aging of the global population, gout’s burden in terms of prevalence and disability is expected to increase.
Impact of obesity and healthy eating patterns
Obesity, or excess adiposity, appears to be of particular concern for women in terms of gout risk.
While obesity and genetic predisposition both are strongly associated with a higher risk of gout, the excess risk of both combined was higher than the sum of each, particularly among women, Natalie McCormick, PhD, of Massachusetts General Hospital, Boston, and coauthors reported in Annals of the Rheumatic Diseases.
These findings suggested that “addressing excess adiposity could prevent a large proportion of female gout cases in particular, as well as its cardiometabolic comorbidities, and the benefit could be greater in genetically predisposed women,” they wrote.
In general, there’s a need to re-examine the advice given by many clinicians in the past that people with gout, or those at risk for it, should follow a low-protein diet to avoid purines, Dr. McCormick said in an interview.
“Now we’re finding that a healthier diet that balances protein as well as fat intake can actually be better both for cardiovascular health and for gout prevention,” she said.
Dr. McCormick’s research on this topic includes a 2022 JAMA Internal Medicine article, and a 2021 article in Current Rheumatology Reports. In the latter article, Dr. McCormick and colleagues examined the benefits of changing habits for patients, such as following one of several well-established healthy eating patterns, including the Mediterranean and DASH diets.
With excess weight and associated cardiovascular and endocrine risks already elevated among people with gout, especially women, the “conventional low-purine (i.e., low-protein) approach to gout dietary guidance is neither helpful nor sustainable and may lead to detrimental effects related to worsening insulin resistance as a result of substitution of healthy proteins with unhealthy carbohydrates or fats,” they wrote. “Rather, by focusing our dietary recommendations on healthy eating patterns which have been proven to reduce cardiometabolic risk factors, as opposed to singular ‘good’ or ‘bad’ food items or groups, the beneficial effects of such diets on relevant gout endpoints should naturally follow for the majority of typical gout cases, mediated through changes in insulin resistance.”
Mr. Sumpter and Dr. McCormick had no competing interests to declare.
Comorbidities may play a greater role than genetics women with gout, although this appears not to be true for men, Nicholas Sumpter, MSc, of the University of Alabama at Birmingham said at the annual research symposium of the Gout, Hyperuricemia, and Crystal Associated Disease Network (G-CAN).
Mr. Sumpter was among the authors of a recent paper in Arthritis & Rheumatology that suggested that earlier gout onset involves the accumulation of certain allelic variants in men. This genetic risk was shared across multiple ancestral groups in the study, conducted with men of European and Polynesian ancestry, Mr. Sumpter and colleagues reported.
“There might be more than one factor in gout in men, but in women we’ve been getting at this idea that comorbidities are the big thing,” he said.
During his presentation, Mr. Sumpter offered a hypothesis that in men there might be a kind of “two-pronged attack,” with increases in serum urate linked to genetic risk, but comorbidities also playing a role. “But that may not be the case for women.”
In his presentation, Mr. Sumpter noted a paper published in March 2022 from his University of Alabama at Birmingham colleagues, Aakash V. Patel, MD, and Angelo L. Gaffo, MD. In the article, Dr. Patel and Dr. Gaffo delved into the challenges of treating women with gout given “the paucity of appropriately well-powered, randomized-controlled trials investigating the efficacy” of commonly used treatments.
“This poses major challenges for the management of female gout patients since they carry a greater burden of cardiovascular and renal morbidity, which is known to modulate the pathophysiology of gout; as such, conclusions regarding the efficacy of treatments for females cannot be extrapolated from investigative studies that are predominantly male,” they wrote, calling for increased efforts to enroll women in studies of treatments for this condition.
There’s increased interest in how gout affects women, including findings in a paper published in September in Arthritis & Rheumatology that found people with gout, especially women, appear to be at higher risk for poor COVID-19 outcomes, including hospitalization and death, regardless of COVID-19 vaccination status.
Gout has become more common in women, although this remains a condition that is far more likely to strike men.
The age-standardized prevalence of gout among women rose from 233.52 per 100,000 in 1990 to 253.49 in 2017, a gain of about 9%, according to a systematic analysis of the Global Burden of Disease Study.
That topped the roughly 5% gain seen for men in the same time frame, with the rate going from 747.48 per 100,000 to 790.90. With the aging of the global population, gout’s burden in terms of prevalence and disability is expected to increase.
Impact of obesity and healthy eating patterns
Obesity, or excess adiposity, appears to be of particular concern for women in terms of gout risk.
While obesity and genetic predisposition both are strongly associated with a higher risk of gout, the excess risk of both combined was higher than the sum of each, particularly among women, Natalie McCormick, PhD, of Massachusetts General Hospital, Boston, and coauthors reported in Annals of the Rheumatic Diseases.
These findings suggested that “addressing excess adiposity could prevent a large proportion of female gout cases in particular, as well as its cardiometabolic comorbidities, and the benefit could be greater in genetically predisposed women,” they wrote.
In general, there’s a need to re-examine the advice given by many clinicians in the past that people with gout, or those at risk for it, should follow a low-protein diet to avoid purines, Dr. McCormick said in an interview.
“Now we’re finding that a healthier diet that balances protein as well as fat intake can actually be better both for cardiovascular health and for gout prevention,” she said.
Dr. McCormick’s research on this topic includes a 2022 JAMA Internal Medicine article, and a 2021 article in Current Rheumatology Reports. In the latter article, Dr. McCormick and colleagues examined the benefits of changing habits for patients, such as following one of several well-established healthy eating patterns, including the Mediterranean and DASH diets.
With excess weight and associated cardiovascular and endocrine risks already elevated among people with gout, especially women, the “conventional low-purine (i.e., low-protein) approach to gout dietary guidance is neither helpful nor sustainable and may lead to detrimental effects related to worsening insulin resistance as a result of substitution of healthy proteins with unhealthy carbohydrates or fats,” they wrote. “Rather, by focusing our dietary recommendations on healthy eating patterns which have been proven to reduce cardiometabolic risk factors, as opposed to singular ‘good’ or ‘bad’ food items or groups, the beneficial effects of such diets on relevant gout endpoints should naturally follow for the majority of typical gout cases, mediated through changes in insulin resistance.”
Mr. Sumpter and Dr. McCormick had no competing interests to declare.
Comorbidities may play a greater role than genetics women with gout, although this appears not to be true for men, Nicholas Sumpter, MSc, of the University of Alabama at Birmingham said at the annual research symposium of the Gout, Hyperuricemia, and Crystal Associated Disease Network (G-CAN).
Mr. Sumpter was among the authors of a recent paper in Arthritis & Rheumatology that suggested that earlier gout onset involves the accumulation of certain allelic variants in men. This genetic risk was shared across multiple ancestral groups in the study, conducted with men of European and Polynesian ancestry, Mr. Sumpter and colleagues reported.
“There might be more than one factor in gout in men, but in women we’ve been getting at this idea that comorbidities are the big thing,” he said.
During his presentation, Mr. Sumpter offered a hypothesis that in men there might be a kind of “two-pronged attack,” with increases in serum urate linked to genetic risk, but comorbidities also playing a role. “But that may not be the case for women.”
In his presentation, Mr. Sumpter noted a paper published in March 2022 from his University of Alabama at Birmingham colleagues, Aakash V. Patel, MD, and Angelo L. Gaffo, MD. In the article, Dr. Patel and Dr. Gaffo delved into the challenges of treating women with gout given “the paucity of appropriately well-powered, randomized-controlled trials investigating the efficacy” of commonly used treatments.
“This poses major challenges for the management of female gout patients since they carry a greater burden of cardiovascular and renal morbidity, which is known to modulate the pathophysiology of gout; as such, conclusions regarding the efficacy of treatments for females cannot be extrapolated from investigative studies that are predominantly male,” they wrote, calling for increased efforts to enroll women in studies of treatments for this condition.
There’s increased interest in how gout affects women, including findings in a paper published in September in Arthritis & Rheumatology that found people with gout, especially women, appear to be at higher risk for poor COVID-19 outcomes, including hospitalization and death, regardless of COVID-19 vaccination status.
Gout has become more common in women, although this remains a condition that is far more likely to strike men.
The age-standardized prevalence of gout among women rose from 233.52 per 100,000 in 1990 to 253.49 in 2017, a gain of about 9%, according to a systematic analysis of the Global Burden of Disease Study.
That topped the roughly 5% gain seen for men in the same time frame, with the rate going from 747.48 per 100,000 to 790.90. With the aging of the global population, gout’s burden in terms of prevalence and disability is expected to increase.
Impact of obesity and healthy eating patterns
Obesity, or excess adiposity, appears to be of particular concern for women in terms of gout risk.
While obesity and genetic predisposition both are strongly associated with a higher risk of gout, the excess risk of both combined was higher than the sum of each, particularly among women, Natalie McCormick, PhD, of Massachusetts General Hospital, Boston, and coauthors reported in Annals of the Rheumatic Diseases.
These findings suggested that “addressing excess adiposity could prevent a large proportion of female gout cases in particular, as well as its cardiometabolic comorbidities, and the benefit could be greater in genetically predisposed women,” they wrote.
In general, there’s a need to re-examine the advice given by many clinicians in the past that people with gout, or those at risk for it, should follow a low-protein diet to avoid purines, Dr. McCormick said in an interview.
“Now we’re finding that a healthier diet that balances protein as well as fat intake can actually be better both for cardiovascular health and for gout prevention,” she said.
Dr. McCormick’s research on this topic includes a 2022 JAMA Internal Medicine article, and a 2021 article in Current Rheumatology Reports. In the latter article, Dr. McCormick and colleagues examined the benefits of changing habits for patients, such as following one of several well-established healthy eating patterns, including the Mediterranean and DASH diets.
With excess weight and associated cardiovascular and endocrine risks already elevated among people with gout, especially women, the “conventional low-purine (i.e., low-protein) approach to gout dietary guidance is neither helpful nor sustainable and may lead to detrimental effects related to worsening insulin resistance as a result of substitution of healthy proteins with unhealthy carbohydrates or fats,” they wrote. “Rather, by focusing our dietary recommendations on healthy eating patterns which have been proven to reduce cardiometabolic risk factors, as opposed to singular ‘good’ or ‘bad’ food items or groups, the beneficial effects of such diets on relevant gout endpoints should naturally follow for the majority of typical gout cases, mediated through changes in insulin resistance.”
Mr. Sumpter and Dr. McCormick had no competing interests to declare.
FROM G-CAN 2022
Worse COVID outcomes seen with gout, particularly in women
People with gout, especially women, appear to be at higher risk for poor COVID-19 outcomes, including hospitalization and death, regardless of COVID-19 vaccination status, researchers suggest.
“We found that the risks of SARS-CoV-2 infection, 30-day hospitalization, and 30-day death among individuals with gout were higher than the general population irrespective of the vaccination status,” lead study author Dongxing Xie, MD, PhD, Xiangya Hospital, Central South University, Changsha, China, and his colleagues write in their large population study. “This finding informs individuals with gout, especially women, that additional measures, even after vaccination, should be considered in order to mitigate the risk of SARS-CoV-2 infection and its severe sequelae.”
People with gout, the most common inflammatory arthritis, often have other conditions that are linked to higher risk for SARS-CoV-2 infection and poor outcomes as well, including obesity, cardiovascular disease, and chronic kidney disease, the authors write. And elevated serum urate may contribute to inflammation and possible COVID-19 complications. But unlike in the case of diseases such as lupus and rheumatoid arthritis, little is known about SARS-CoV-2 infection risk among patients with gout.
As reported in Arthritis & Rheumatology, Dr. Xie and his research team used the Health Improvement Network ([THIN], now called IQVIA Medical Research Database) repository of medical conditions, demographics, and other details of around 17 million people in the United Kingdom to estimate the risk for SARS-CoV-2 infection, hospitalization, and death in people with gout. They compared those outcomes with outcomes of people without gout and compared outcomes of vaccinated vs. nonvaccinated participants.
From December 2020 through October 2021, the researchers investigated the risk for SARS-CoV-2 breakthrough infection in vaccinated people between age 18 and 90 years who had gout and were hospitalized within 30 days after the infection diagnosis or who died within 30 days after the diagnosis. They compared these outcomes with the outcomes of people in the general population without gout after COVID-19 vaccination. They also compared the risk for SARS-CoV-2 infection and its severe outcomes between individuals with gout and the general population among unvaccinated people.
They weighted these comparisons on the basis of age, sex, body mass index, socioeconomic deprivation index score, region, and number of previous COVID-19 tests in one model. A more fully adjusted model also weighted the comparisons for lifestyle factors, comorbidities, medications, and healthcare utilization.
The vaccinated cohort consisted of 54,576 people with gout and 1,336,377 without gout from the general population. The unvaccinated cohort included 61,111 individuals with gout and 1,697,168 individuals without gout from the general population.
Women more likely to be hospitalized and die
The risk for breakthrough infection in the vaccinated cohort was significantly higher among people with gout than among those without gout in the general population, particularly for men, who had hazard ratios (HRs) ranging from 1.22 with a fully adjusted exposure score to 1.30 with a partially adjusted score, but this was not seen in women. The overall incidence of breakthrough infection per 1,000 person-months for these groups was 4.68 with gout vs. 3.76 without gout.
The researchers showed a similar pattern of a higher rate of hospitalizations for people with gout vs. without (0.42/1,000 person-months vs. 0.28); in this case, women had higher risks than did men, with HRs for women ranging from 1.55 with a fully adjusted exposure score to 1.91 with a partially adjusted score, compared with 1.22 and 1.43 for men, respectively.
People with gout had significantly higher mortality than did those without (0.06/1,000 person-months vs. 0.04), but the risk for death was only higher for women, with HRs calculated to be 2.23 in fully adjusted exposure scores and 3.01 in partially adjusted scores.
These same comparisons in the unvaccinated cohort all went in the same direction as did those in the vaccinated cohort but showed higher rates for infection (8.69/1,000 person-months vs. 6.89), hospitalization (2.57/1,000 person-months vs. 1.71), and death (0.65/1,000 person-months vs. 0.53). Similar sex-specific links between gout and risks for SARS-CoV-2 infection, hospitalization, and death were seen in the unvaccinated cohort.
Patients with gout and COVID-19 need close monitoring
Four experts who were not involved in the study encourage greater attention to the needs of patients with gout.
Pamela B. Davis, MD, PhD, research professor at Case Western Reserve University, Cleveland, told this news organization, “This study brings to attention yet another potentially vulnerable group for physicians to monitor closely if they are infected with SARS-CoV-2.
“It is not clear why women with gout are more vulnerable, but fewer women than men were in the cohort with gout, and the confidence intervals for the results in women were, in general, larger,” she said.
“The authors suggest that women with gout tend to be older and have more comorbidities than men with gout,” Dr. Davis added. “The excess risk diminishes when the model is fully adjusted for comorbidities, such as obesity, hypertension, or heart disease, suggesting that already-known antecedents of infection severity account for a great deal of the excess risk.”
Kevin D. Deane, MD, PhD, associate professor of medicine and chair in rheumatology research at the University of Colorado at Denver, Aurora, advises physicians to keep in mind other conditions linked with increased risk for severe COVID-19, including advanced age; heart, lung, or kidney problems; and autoimmune diseases.
“I would be very cautious about the finding that there was not a difference in outcomes in individuals with gout based on vaccination status,” he cautioned, urging clinicians to “still strongly recommend vaccines according to guidelines.”
Sarah E. Waldman, MD, associate clinical professor of infectious diseases at UC Davis Health in Sacramento, Calif., called the study interesting but not surprising.
“The reason for increased risk for COVID-19 infection among those with gout may have to do with their underlying inflammatory state. Additional research needs to be done on this topic.
“Retrospective population-based cohort studies can be difficult to interpret due to biases,” she added. Associations identified in this type of study do not determine causation.
“As the researchers noted, those with gout tend to have additional comorbidities as well as advanced age,” she said. “They may also seek medical care more often and be tested for SARS-CoV-2 more frequently.”
Dr. Waldman advises clinicians to counsel patients with gout about their potential increased infection risk and ways they can protect themselves, including COVID-19 vaccinations.
“The strong association between gout and COVID-19 infection could involve coexisting conditions such as diabetes, hypertension, cardiovascular disease, and chronic kidney disease,” Dr. Aung added.
Earlier studies show links between gout and severe COVID-19 outcomes
Lead author Kanon Jatuworapruk, MD, PhD, of Thammasat University in Pathumthani, Thailand, and his colleagues investigated characteristics and outcomes of people with gout who were hospitalized for COVID-19 between March 2020 and October 2021, using data from the COVID-19 Global Rheumatology Alliance registry.
“This cohort of people with gout and COVID-19 who were hospitalized had high frequencies of ventilatory support and death,” the authors write in ACR Open Rheumatology . “This suggests that patients with gout who were hospitalized for COVID-19 may be at risk of poor outcomes, perhaps related to known risk factors for poor outcomes, such as age and presence of comorbidity.”
In their study, the average age of the 163 patients was 63 years, and 85% were men. Most lived in the Western Pacific Region and North America, and 46% had two or more comorbidities, most commonly hypertension, cardiovascular disease, diabetes, chronic kidney disease, and obesity. The researchers found that:
- Sixty-eight percent of the cohort required supplemental oxygen or ventilatory support during hospitalization.
- Sixteen percent of deaths were related to COVID-19, with 73% of deaths occurring in people with two or more comorbidities.
Ruth K. Topless, assistant research fellow in the department of biochemistry at the University of Otago in Dunedin, New Zealand, is the lead author on a study she and her colleagues are conducting using the UK Biobank databases of 459,837 participants in the United Kingdom, including 15,871 people with gout, through April 6, 2021, to investigate whether gout is a risk factor for diagnosis of COVID-19 and COVID-19–related death.
“Gout is a risk factor for COVID-19-related death in the UK Biobank cohort, with an increased risk in women with gout, which was driven by risk factors independent of the metabolic comorbidities of gout,” the researchers conclude in The Lancet Rheumatology.
In their study, gout was linked with COVID-19 diagnosis (odds ratio, 1.20; 95% confidence interval, 1.11-1.29) but not with risk for COVID-19–related death in the group of patients with COVID-19 (OR, 1.20; 95% CI, 0.96-1.51). In the entire cohort, gout was linked with COVID-19–related death (OR, 1.29; 95% CI, 1.06-1.56); women with gout were at increased risk for COVID-19–related death (OR, 1.98; 95% CI, 1.34-2.94), but men with gout were not (OR, 1.16; 95% CI, 0.93-1.45). The risk for COVID-19 diagnosis was significant in the nonvaccinated group (OR, 1.21; 95% CI, 1.11-1.30) but not in the vaccinated group (OR, 1.09; 95% CI, 0.65-1.85).
Editorial authors join in recommending further related research
In a commentary in The Lancet Rheumatology about the UK Biobank and other related research, Christoffer B. Nissen, MD, of University Hospital of Southern Denmark in Sonderborg, and his co-authors call the Topless and colleagues study “an elegantly conducted analysis of data from the UK Biobank supporting the hypothesis that gout needs attention in patients with COVID-19.”
Further studies are needed to investigate to what degree a diagnosis of gout is a risk factor for COVID-19 and whether treatment modifies the risk of a severe disease course,” they write. “However, in the interim, the results of this study could be considered when risk stratifying patients with gout in view of vaccination recommendations and early treatment interventions.”
Each of the three studies received grant funding. Several of the authors of the studies report financial involvements with pharmaceutical companies. All outside experts commented by email and report no relevant financial involvements.
A version of this article first appeared on Medscape.com.
People with gout, especially women, appear to be at higher risk for poor COVID-19 outcomes, including hospitalization and death, regardless of COVID-19 vaccination status, researchers suggest.
“We found that the risks of SARS-CoV-2 infection, 30-day hospitalization, and 30-day death among individuals with gout were higher than the general population irrespective of the vaccination status,” lead study author Dongxing Xie, MD, PhD, Xiangya Hospital, Central South University, Changsha, China, and his colleagues write in their large population study. “This finding informs individuals with gout, especially women, that additional measures, even after vaccination, should be considered in order to mitigate the risk of SARS-CoV-2 infection and its severe sequelae.”
People with gout, the most common inflammatory arthritis, often have other conditions that are linked to higher risk for SARS-CoV-2 infection and poor outcomes as well, including obesity, cardiovascular disease, and chronic kidney disease, the authors write. And elevated serum urate may contribute to inflammation and possible COVID-19 complications. But unlike in the case of diseases such as lupus and rheumatoid arthritis, little is known about SARS-CoV-2 infection risk among patients with gout.
As reported in Arthritis & Rheumatology, Dr. Xie and his research team used the Health Improvement Network ([THIN], now called IQVIA Medical Research Database) repository of medical conditions, demographics, and other details of around 17 million people in the United Kingdom to estimate the risk for SARS-CoV-2 infection, hospitalization, and death in people with gout. They compared those outcomes with outcomes of people without gout and compared outcomes of vaccinated vs. nonvaccinated participants.
From December 2020 through October 2021, the researchers investigated the risk for SARS-CoV-2 breakthrough infection in vaccinated people between age 18 and 90 years who had gout and were hospitalized within 30 days after the infection diagnosis or who died within 30 days after the diagnosis. They compared these outcomes with the outcomes of people in the general population without gout after COVID-19 vaccination. They also compared the risk for SARS-CoV-2 infection and its severe outcomes between individuals with gout and the general population among unvaccinated people.
They weighted these comparisons on the basis of age, sex, body mass index, socioeconomic deprivation index score, region, and number of previous COVID-19 tests in one model. A more fully adjusted model also weighted the comparisons for lifestyle factors, comorbidities, medications, and healthcare utilization.
The vaccinated cohort consisted of 54,576 people with gout and 1,336,377 without gout from the general population. The unvaccinated cohort included 61,111 individuals with gout and 1,697,168 individuals without gout from the general population.
Women more likely to be hospitalized and die
The risk for breakthrough infection in the vaccinated cohort was significantly higher among people with gout than among those without gout in the general population, particularly for men, who had hazard ratios (HRs) ranging from 1.22 with a fully adjusted exposure score to 1.30 with a partially adjusted score, but this was not seen in women. The overall incidence of breakthrough infection per 1,000 person-months for these groups was 4.68 with gout vs. 3.76 without gout.
The researchers showed a similar pattern of a higher rate of hospitalizations for people with gout vs. without (0.42/1,000 person-months vs. 0.28); in this case, women had higher risks than did men, with HRs for women ranging from 1.55 with a fully adjusted exposure score to 1.91 with a partially adjusted score, compared with 1.22 and 1.43 for men, respectively.
People with gout had significantly higher mortality than did those without (0.06/1,000 person-months vs. 0.04), but the risk for death was only higher for women, with HRs calculated to be 2.23 in fully adjusted exposure scores and 3.01 in partially adjusted scores.
These same comparisons in the unvaccinated cohort all went in the same direction as did those in the vaccinated cohort but showed higher rates for infection (8.69/1,000 person-months vs. 6.89), hospitalization (2.57/1,000 person-months vs. 1.71), and death (0.65/1,000 person-months vs. 0.53). Similar sex-specific links between gout and risks for SARS-CoV-2 infection, hospitalization, and death were seen in the unvaccinated cohort.
Patients with gout and COVID-19 need close monitoring
Four experts who were not involved in the study encourage greater attention to the needs of patients with gout.
Pamela B. Davis, MD, PhD, research professor at Case Western Reserve University, Cleveland, told this news organization, “This study brings to attention yet another potentially vulnerable group for physicians to monitor closely if they are infected with SARS-CoV-2.
“It is not clear why women with gout are more vulnerable, but fewer women than men were in the cohort with gout, and the confidence intervals for the results in women were, in general, larger,” she said.
“The authors suggest that women with gout tend to be older and have more comorbidities than men with gout,” Dr. Davis added. “The excess risk diminishes when the model is fully adjusted for comorbidities, such as obesity, hypertension, or heart disease, suggesting that already-known antecedents of infection severity account for a great deal of the excess risk.”
Kevin D. Deane, MD, PhD, associate professor of medicine and chair in rheumatology research at the University of Colorado at Denver, Aurora, advises physicians to keep in mind other conditions linked with increased risk for severe COVID-19, including advanced age; heart, lung, or kidney problems; and autoimmune diseases.
“I would be very cautious about the finding that there was not a difference in outcomes in individuals with gout based on vaccination status,” he cautioned, urging clinicians to “still strongly recommend vaccines according to guidelines.”
Sarah E. Waldman, MD, associate clinical professor of infectious diseases at UC Davis Health in Sacramento, Calif., called the study interesting but not surprising.
“The reason for increased risk for COVID-19 infection among those with gout may have to do with their underlying inflammatory state. Additional research needs to be done on this topic.
“Retrospective population-based cohort studies can be difficult to interpret due to biases,” she added. Associations identified in this type of study do not determine causation.
“As the researchers noted, those with gout tend to have additional comorbidities as well as advanced age,” she said. “They may also seek medical care more often and be tested for SARS-CoV-2 more frequently.”
Dr. Waldman advises clinicians to counsel patients with gout about their potential increased infection risk and ways they can protect themselves, including COVID-19 vaccinations.
“The strong association between gout and COVID-19 infection could involve coexisting conditions such as diabetes, hypertension, cardiovascular disease, and chronic kidney disease,” Dr. Aung added.
Earlier studies show links between gout and severe COVID-19 outcomes
Lead author Kanon Jatuworapruk, MD, PhD, of Thammasat University in Pathumthani, Thailand, and his colleagues investigated characteristics and outcomes of people with gout who were hospitalized for COVID-19 between March 2020 and October 2021, using data from the COVID-19 Global Rheumatology Alliance registry.
“This cohort of people with gout and COVID-19 who were hospitalized had high frequencies of ventilatory support and death,” the authors write in ACR Open Rheumatology . “This suggests that patients with gout who were hospitalized for COVID-19 may be at risk of poor outcomes, perhaps related to known risk factors for poor outcomes, such as age and presence of comorbidity.”
In their study, the average age of the 163 patients was 63 years, and 85% were men. Most lived in the Western Pacific Region and North America, and 46% had two or more comorbidities, most commonly hypertension, cardiovascular disease, diabetes, chronic kidney disease, and obesity. The researchers found that:
- Sixty-eight percent of the cohort required supplemental oxygen or ventilatory support during hospitalization.
- Sixteen percent of deaths were related to COVID-19, with 73% of deaths occurring in people with two or more comorbidities.
Ruth K. Topless, assistant research fellow in the department of biochemistry at the University of Otago in Dunedin, New Zealand, is the lead author on a study she and her colleagues are conducting using the UK Biobank databases of 459,837 participants in the United Kingdom, including 15,871 people with gout, through April 6, 2021, to investigate whether gout is a risk factor for diagnosis of COVID-19 and COVID-19–related death.
“Gout is a risk factor for COVID-19-related death in the UK Biobank cohort, with an increased risk in women with gout, which was driven by risk factors independent of the metabolic comorbidities of gout,” the researchers conclude in The Lancet Rheumatology.
In their study, gout was linked with COVID-19 diagnosis (odds ratio, 1.20; 95% confidence interval, 1.11-1.29) but not with risk for COVID-19–related death in the group of patients with COVID-19 (OR, 1.20; 95% CI, 0.96-1.51). In the entire cohort, gout was linked with COVID-19–related death (OR, 1.29; 95% CI, 1.06-1.56); women with gout were at increased risk for COVID-19–related death (OR, 1.98; 95% CI, 1.34-2.94), but men with gout were not (OR, 1.16; 95% CI, 0.93-1.45). The risk for COVID-19 diagnosis was significant in the nonvaccinated group (OR, 1.21; 95% CI, 1.11-1.30) but not in the vaccinated group (OR, 1.09; 95% CI, 0.65-1.85).
Editorial authors join in recommending further related research
In a commentary in The Lancet Rheumatology about the UK Biobank and other related research, Christoffer B. Nissen, MD, of University Hospital of Southern Denmark in Sonderborg, and his co-authors call the Topless and colleagues study “an elegantly conducted analysis of data from the UK Biobank supporting the hypothesis that gout needs attention in patients with COVID-19.”
Further studies are needed to investigate to what degree a diagnosis of gout is a risk factor for COVID-19 and whether treatment modifies the risk of a severe disease course,” they write. “However, in the interim, the results of this study could be considered when risk stratifying patients with gout in view of vaccination recommendations and early treatment interventions.”
Each of the three studies received grant funding. Several of the authors of the studies report financial involvements with pharmaceutical companies. All outside experts commented by email and report no relevant financial involvements.
A version of this article first appeared on Medscape.com.
People with gout, especially women, appear to be at higher risk for poor COVID-19 outcomes, including hospitalization and death, regardless of COVID-19 vaccination status, researchers suggest.
“We found that the risks of SARS-CoV-2 infection, 30-day hospitalization, and 30-day death among individuals with gout were higher than the general population irrespective of the vaccination status,” lead study author Dongxing Xie, MD, PhD, Xiangya Hospital, Central South University, Changsha, China, and his colleagues write in their large population study. “This finding informs individuals with gout, especially women, that additional measures, even after vaccination, should be considered in order to mitigate the risk of SARS-CoV-2 infection and its severe sequelae.”
People with gout, the most common inflammatory arthritis, often have other conditions that are linked to higher risk for SARS-CoV-2 infection and poor outcomes as well, including obesity, cardiovascular disease, and chronic kidney disease, the authors write. And elevated serum urate may contribute to inflammation and possible COVID-19 complications. But unlike in the case of diseases such as lupus and rheumatoid arthritis, little is known about SARS-CoV-2 infection risk among patients with gout.
As reported in Arthritis & Rheumatology, Dr. Xie and his research team used the Health Improvement Network ([THIN], now called IQVIA Medical Research Database) repository of medical conditions, demographics, and other details of around 17 million people in the United Kingdom to estimate the risk for SARS-CoV-2 infection, hospitalization, and death in people with gout. They compared those outcomes with outcomes of people without gout and compared outcomes of vaccinated vs. nonvaccinated participants.
From December 2020 through October 2021, the researchers investigated the risk for SARS-CoV-2 breakthrough infection in vaccinated people between age 18 and 90 years who had gout and were hospitalized within 30 days after the infection diagnosis or who died within 30 days after the diagnosis. They compared these outcomes with the outcomes of people in the general population without gout after COVID-19 vaccination. They also compared the risk for SARS-CoV-2 infection and its severe outcomes between individuals with gout and the general population among unvaccinated people.
They weighted these comparisons on the basis of age, sex, body mass index, socioeconomic deprivation index score, region, and number of previous COVID-19 tests in one model. A more fully adjusted model also weighted the comparisons for lifestyle factors, comorbidities, medications, and healthcare utilization.
The vaccinated cohort consisted of 54,576 people with gout and 1,336,377 without gout from the general population. The unvaccinated cohort included 61,111 individuals with gout and 1,697,168 individuals without gout from the general population.
Women more likely to be hospitalized and die
The risk for breakthrough infection in the vaccinated cohort was significantly higher among people with gout than among those without gout in the general population, particularly for men, who had hazard ratios (HRs) ranging from 1.22 with a fully adjusted exposure score to 1.30 with a partially adjusted score, but this was not seen in women. The overall incidence of breakthrough infection per 1,000 person-months for these groups was 4.68 with gout vs. 3.76 without gout.
The researchers showed a similar pattern of a higher rate of hospitalizations for people with gout vs. without (0.42/1,000 person-months vs. 0.28); in this case, women had higher risks than did men, with HRs for women ranging from 1.55 with a fully adjusted exposure score to 1.91 with a partially adjusted score, compared with 1.22 and 1.43 for men, respectively.
People with gout had significantly higher mortality than did those without (0.06/1,000 person-months vs. 0.04), but the risk for death was only higher for women, with HRs calculated to be 2.23 in fully adjusted exposure scores and 3.01 in partially adjusted scores.
These same comparisons in the unvaccinated cohort all went in the same direction as did those in the vaccinated cohort but showed higher rates for infection (8.69/1,000 person-months vs. 6.89), hospitalization (2.57/1,000 person-months vs. 1.71), and death (0.65/1,000 person-months vs. 0.53). Similar sex-specific links between gout and risks for SARS-CoV-2 infection, hospitalization, and death were seen in the unvaccinated cohort.
Patients with gout and COVID-19 need close monitoring
Four experts who were not involved in the study encourage greater attention to the needs of patients with gout.
Pamela B. Davis, MD, PhD, research professor at Case Western Reserve University, Cleveland, told this news organization, “This study brings to attention yet another potentially vulnerable group for physicians to monitor closely if they are infected with SARS-CoV-2.
“It is not clear why women with gout are more vulnerable, but fewer women than men were in the cohort with gout, and the confidence intervals for the results in women were, in general, larger,” she said.
“The authors suggest that women with gout tend to be older and have more comorbidities than men with gout,” Dr. Davis added. “The excess risk diminishes when the model is fully adjusted for comorbidities, such as obesity, hypertension, or heart disease, suggesting that already-known antecedents of infection severity account for a great deal of the excess risk.”
Kevin D. Deane, MD, PhD, associate professor of medicine and chair in rheumatology research at the University of Colorado at Denver, Aurora, advises physicians to keep in mind other conditions linked with increased risk for severe COVID-19, including advanced age; heart, lung, or kidney problems; and autoimmune diseases.
“I would be very cautious about the finding that there was not a difference in outcomes in individuals with gout based on vaccination status,” he cautioned, urging clinicians to “still strongly recommend vaccines according to guidelines.”
Sarah E. Waldman, MD, associate clinical professor of infectious diseases at UC Davis Health in Sacramento, Calif., called the study interesting but not surprising.
“The reason for increased risk for COVID-19 infection among those with gout may have to do with their underlying inflammatory state. Additional research needs to be done on this topic.
“Retrospective population-based cohort studies can be difficult to interpret due to biases,” she added. Associations identified in this type of study do not determine causation.
“As the researchers noted, those with gout tend to have additional comorbidities as well as advanced age,” she said. “They may also seek medical care more often and be tested for SARS-CoV-2 more frequently.”
Dr. Waldman advises clinicians to counsel patients with gout about their potential increased infection risk and ways they can protect themselves, including COVID-19 vaccinations.
“The strong association between gout and COVID-19 infection could involve coexisting conditions such as diabetes, hypertension, cardiovascular disease, and chronic kidney disease,” Dr. Aung added.
Earlier studies show links between gout and severe COVID-19 outcomes
Lead author Kanon Jatuworapruk, MD, PhD, of Thammasat University in Pathumthani, Thailand, and his colleagues investigated characteristics and outcomes of people with gout who were hospitalized for COVID-19 between March 2020 and October 2021, using data from the COVID-19 Global Rheumatology Alliance registry.
“This cohort of people with gout and COVID-19 who were hospitalized had high frequencies of ventilatory support and death,” the authors write in ACR Open Rheumatology . “This suggests that patients with gout who were hospitalized for COVID-19 may be at risk of poor outcomes, perhaps related to known risk factors for poor outcomes, such as age and presence of comorbidity.”
In their study, the average age of the 163 patients was 63 years, and 85% were men. Most lived in the Western Pacific Region and North America, and 46% had two or more comorbidities, most commonly hypertension, cardiovascular disease, diabetes, chronic kidney disease, and obesity. The researchers found that:
- Sixty-eight percent of the cohort required supplemental oxygen or ventilatory support during hospitalization.
- Sixteen percent of deaths were related to COVID-19, with 73% of deaths occurring in people with two or more comorbidities.
Ruth K. Topless, assistant research fellow in the department of biochemistry at the University of Otago in Dunedin, New Zealand, is the lead author on a study she and her colleagues are conducting using the UK Biobank databases of 459,837 participants in the United Kingdom, including 15,871 people with gout, through April 6, 2021, to investigate whether gout is a risk factor for diagnosis of COVID-19 and COVID-19–related death.
“Gout is a risk factor for COVID-19-related death in the UK Biobank cohort, with an increased risk in women with gout, which was driven by risk factors independent of the metabolic comorbidities of gout,” the researchers conclude in The Lancet Rheumatology.
In their study, gout was linked with COVID-19 diagnosis (odds ratio, 1.20; 95% confidence interval, 1.11-1.29) but not with risk for COVID-19–related death in the group of patients with COVID-19 (OR, 1.20; 95% CI, 0.96-1.51). In the entire cohort, gout was linked with COVID-19–related death (OR, 1.29; 95% CI, 1.06-1.56); women with gout were at increased risk for COVID-19–related death (OR, 1.98; 95% CI, 1.34-2.94), but men with gout were not (OR, 1.16; 95% CI, 0.93-1.45). The risk for COVID-19 diagnosis was significant in the nonvaccinated group (OR, 1.21; 95% CI, 1.11-1.30) but not in the vaccinated group (OR, 1.09; 95% CI, 0.65-1.85).
Editorial authors join in recommending further related research
In a commentary in The Lancet Rheumatology about the UK Biobank and other related research, Christoffer B. Nissen, MD, of University Hospital of Southern Denmark in Sonderborg, and his co-authors call the Topless and colleagues study “an elegantly conducted analysis of data from the UK Biobank supporting the hypothesis that gout needs attention in patients with COVID-19.”
Further studies are needed to investigate to what degree a diagnosis of gout is a risk factor for COVID-19 and whether treatment modifies the risk of a severe disease course,” they write. “However, in the interim, the results of this study could be considered when risk stratifying patients with gout in view of vaccination recommendations and early treatment interventions.”
Each of the three studies received grant funding. Several of the authors of the studies report financial involvements with pharmaceutical companies. All outside experts commented by email and report no relevant financial involvements.
A version of this article first appeared on Medscape.com.
Could vitamin C help reduce gout?
Could taking vitamin C help reduce the chances of developing gout? A new study sheds light on this possibility.
Gout is a form of inflammatory arthritis that has been on the rise in the United States in recent decades. Considered a lifestyle disease, some research has shown that instances of the condition have more than doubled in recent years as rates of obesity have skyrocketed. It’s caused by uric acid in the blood that builds up and crystallizes in the joints. Flare-ups are so intense that the joints can turn a cherry red and vibrate with intense – and sometimes seemingly intolerable – pain.
While there are effective treatments, many people fail to take their medications when they’re not in pain, and if the condition goes unchecked, it can get much worse and cause permanent damage to the joints.
“Gout can cause flare-ups that vary in frequency and severity; but sometimes when people aren’t experiencing them, they’re less likely to stay on top of their medications,” said Stephen Juraschek, MD, an assistant professor of medicine at Harvard Medical School, Boston.
That’s why lifestyle interventions are seen as particularly relevant to a disease like gout. Vitamin C, for example, has few side effects, and for those with higher levels of uric acid in the blood, it could reduce the likelihood of getting the condition. A recent study published in The American Journal of Clinical Nutrition found that people who were given 500 mg of vitamin C versus a placebo had a 12% reduced risk of getting gout. The study of over 14,000 male doctors showed that men who weren’t overweight had the most significant reduction in the risk of getting the condition. (Excess weight has been shown to increase the risk of gout.)
As part of the study, participants responded to a questionnaire that asked whether they had ever been diagnosed with gout. Other studies have shown that vitamin C reduced the levels of urate in people without gout and broke down uric crystals in the blood, but this study took it a step further to show that the supplement actually reduced the risk of getting the condition.
“In addition to lowering levels of uric acid in the body, it’s thought that vitamin C may also minimize the inflammatory response to urate crystals,” said Dr. Juraschek. That’s because when flare-ups develop in joints throughout the body, much of the painful irritation is caused by the immune system’s response as it fights to break down the crystals.
Dr. Juraschek said this likely wouldn’t change recommendations for patients with serious gout, but it could still have an impact.
“For individuals who were told that they have gout but have had fewer flare-ups, they might be more open to taking vitamin C,” he said.
Will Settle, 42, of Hilton Head, S.C., was not involved in the study, but he said he would be inclined to try most any safe preventive method. Gout runs in his family. His father and grandfather had it, and now, so does he. His flare-ups have slowed in recent years, which he said has a lot to do with his diet and lifestyle. He stopped eating seafood, started drinking more water, and stopped drinking as much alcohol – all of which he thinks has had a huge impact on the severity of his condition. (Both seafood and beer contain high levels of purines, which have been shown to increase the buildup of uric acid in the blood.) Mr. Settle said that other simple lifestyle changes like vitamin C would be an easy addition to his routine with few downsides. Plus, he hates having to take colchicine, a medication that’s meant to relieve pain but causes him intense diarrhea when he takes it.
“Anything to reduce my flare-ups without having to take colchicine,” he said.
But the jury is still out as to whether vitamin C will have any real benefits. Study coauthor Robert H. Shmerling, MD, is the former clinical chief of the division of rheumatology at Beth Israel Deaconess Medical Center, New York. He said the study shows that the effect of vitamin C in those undiagnosed with gout was rather modest. Also, vitamin C did not show a reduction in gout flare-ups in those who were already diagnosed with the condition. Not to mention that the study lacked diversity, as the people in it were all male and mostly white. Still, there’s little downside risk to taking vitamin C, and it might end up being worthwhile.
“Maybe it will turn out to be an effective treatment in those who are at high risk, but we’re not there yet,” he said.
Robert Terkeltaub, MD, chief of rheumatology at the Veterans Administration Medical Center in San Diego and a professor of medicine at the University of California, San Diego, said there’s an unmet need when it comes to tools for gout prevention.
“The disease impacts some 10 million Americans, and we need to better identify these individuals so we can intervene earlier,” he said.
While vitamin C had a small but significant association with fewer new cases of gout, it did not lower it in those who already had the disease, said Dr. Terkeltaub. What’s more, researchers didn’t measure the levels of uric acid in the blood, which would have painted a more accurate picture of whether vitamin C actually reduced it in the body.
“There remains no clarity on the potential role of vitamin C in either prevention or treatment of gout. That said, future research would be of interest,” he said.
Still, gout patients like Mr. Settle aren’t ruling it out. Anything to avoid the pain that, at times, makes it difficult for him to get out of bed. He’s seen the benefit that simple lifestyle changes can make, and he’s willing to try just about anything to live a normal, arthritis-free life.
“I’m always looking for simple ways to keep my flare-ups at bay,” he said.
A version of this article first appeared on WebMD.com.
Could taking vitamin C help reduce the chances of developing gout? A new study sheds light on this possibility.
Gout is a form of inflammatory arthritis that has been on the rise in the United States in recent decades. Considered a lifestyle disease, some research has shown that instances of the condition have more than doubled in recent years as rates of obesity have skyrocketed. It’s caused by uric acid in the blood that builds up and crystallizes in the joints. Flare-ups are so intense that the joints can turn a cherry red and vibrate with intense – and sometimes seemingly intolerable – pain.
While there are effective treatments, many people fail to take their medications when they’re not in pain, and if the condition goes unchecked, it can get much worse and cause permanent damage to the joints.
“Gout can cause flare-ups that vary in frequency and severity; but sometimes when people aren’t experiencing them, they’re less likely to stay on top of their medications,” said Stephen Juraschek, MD, an assistant professor of medicine at Harvard Medical School, Boston.
That’s why lifestyle interventions are seen as particularly relevant to a disease like gout. Vitamin C, for example, has few side effects, and for those with higher levels of uric acid in the blood, it could reduce the likelihood of getting the condition. A recent study published in The American Journal of Clinical Nutrition found that people who were given 500 mg of vitamin C versus a placebo had a 12% reduced risk of getting gout. The study of over 14,000 male doctors showed that men who weren’t overweight had the most significant reduction in the risk of getting the condition. (Excess weight has been shown to increase the risk of gout.)
As part of the study, participants responded to a questionnaire that asked whether they had ever been diagnosed with gout. Other studies have shown that vitamin C reduced the levels of urate in people without gout and broke down uric crystals in the blood, but this study took it a step further to show that the supplement actually reduced the risk of getting the condition.
“In addition to lowering levels of uric acid in the body, it’s thought that vitamin C may also minimize the inflammatory response to urate crystals,” said Dr. Juraschek. That’s because when flare-ups develop in joints throughout the body, much of the painful irritation is caused by the immune system’s response as it fights to break down the crystals.
Dr. Juraschek said this likely wouldn’t change recommendations for patients with serious gout, but it could still have an impact.
“For individuals who were told that they have gout but have had fewer flare-ups, they might be more open to taking vitamin C,” he said.
Will Settle, 42, of Hilton Head, S.C., was not involved in the study, but he said he would be inclined to try most any safe preventive method. Gout runs in his family. His father and grandfather had it, and now, so does he. His flare-ups have slowed in recent years, which he said has a lot to do with his diet and lifestyle. He stopped eating seafood, started drinking more water, and stopped drinking as much alcohol – all of which he thinks has had a huge impact on the severity of his condition. (Both seafood and beer contain high levels of purines, which have been shown to increase the buildup of uric acid in the blood.) Mr. Settle said that other simple lifestyle changes like vitamin C would be an easy addition to his routine with few downsides. Plus, he hates having to take colchicine, a medication that’s meant to relieve pain but causes him intense diarrhea when he takes it.
“Anything to reduce my flare-ups without having to take colchicine,” he said.
But the jury is still out as to whether vitamin C will have any real benefits. Study coauthor Robert H. Shmerling, MD, is the former clinical chief of the division of rheumatology at Beth Israel Deaconess Medical Center, New York. He said the study shows that the effect of vitamin C in those undiagnosed with gout was rather modest. Also, vitamin C did not show a reduction in gout flare-ups in those who were already diagnosed with the condition. Not to mention that the study lacked diversity, as the people in it were all male and mostly white. Still, there’s little downside risk to taking vitamin C, and it might end up being worthwhile.
“Maybe it will turn out to be an effective treatment in those who are at high risk, but we’re not there yet,” he said.
Robert Terkeltaub, MD, chief of rheumatology at the Veterans Administration Medical Center in San Diego and a professor of medicine at the University of California, San Diego, said there’s an unmet need when it comes to tools for gout prevention.
“The disease impacts some 10 million Americans, and we need to better identify these individuals so we can intervene earlier,” he said.
While vitamin C had a small but significant association with fewer new cases of gout, it did not lower it in those who already had the disease, said Dr. Terkeltaub. What’s more, researchers didn’t measure the levels of uric acid in the blood, which would have painted a more accurate picture of whether vitamin C actually reduced it in the body.
“There remains no clarity on the potential role of vitamin C in either prevention or treatment of gout. That said, future research would be of interest,” he said.
Still, gout patients like Mr. Settle aren’t ruling it out. Anything to avoid the pain that, at times, makes it difficult for him to get out of bed. He’s seen the benefit that simple lifestyle changes can make, and he’s willing to try just about anything to live a normal, arthritis-free life.
“I’m always looking for simple ways to keep my flare-ups at bay,” he said.
A version of this article first appeared on WebMD.com.
Could taking vitamin C help reduce the chances of developing gout? A new study sheds light on this possibility.
Gout is a form of inflammatory arthritis that has been on the rise in the United States in recent decades. Considered a lifestyle disease, some research has shown that instances of the condition have more than doubled in recent years as rates of obesity have skyrocketed. It’s caused by uric acid in the blood that builds up and crystallizes in the joints. Flare-ups are so intense that the joints can turn a cherry red and vibrate with intense – and sometimes seemingly intolerable – pain.
While there are effective treatments, many people fail to take their medications when they’re not in pain, and if the condition goes unchecked, it can get much worse and cause permanent damage to the joints.
“Gout can cause flare-ups that vary in frequency and severity; but sometimes when people aren’t experiencing them, they’re less likely to stay on top of their medications,” said Stephen Juraschek, MD, an assistant professor of medicine at Harvard Medical School, Boston.
That’s why lifestyle interventions are seen as particularly relevant to a disease like gout. Vitamin C, for example, has few side effects, and for those with higher levels of uric acid in the blood, it could reduce the likelihood of getting the condition. A recent study published in The American Journal of Clinical Nutrition found that people who were given 500 mg of vitamin C versus a placebo had a 12% reduced risk of getting gout. The study of over 14,000 male doctors showed that men who weren’t overweight had the most significant reduction in the risk of getting the condition. (Excess weight has been shown to increase the risk of gout.)
As part of the study, participants responded to a questionnaire that asked whether they had ever been diagnosed with gout. Other studies have shown that vitamin C reduced the levels of urate in people without gout and broke down uric crystals in the blood, but this study took it a step further to show that the supplement actually reduced the risk of getting the condition.
“In addition to lowering levels of uric acid in the body, it’s thought that vitamin C may also minimize the inflammatory response to urate crystals,” said Dr. Juraschek. That’s because when flare-ups develop in joints throughout the body, much of the painful irritation is caused by the immune system’s response as it fights to break down the crystals.
Dr. Juraschek said this likely wouldn’t change recommendations for patients with serious gout, but it could still have an impact.
“For individuals who were told that they have gout but have had fewer flare-ups, they might be more open to taking vitamin C,” he said.
Will Settle, 42, of Hilton Head, S.C., was not involved in the study, but he said he would be inclined to try most any safe preventive method. Gout runs in his family. His father and grandfather had it, and now, so does he. His flare-ups have slowed in recent years, which he said has a lot to do with his diet and lifestyle. He stopped eating seafood, started drinking more water, and stopped drinking as much alcohol – all of which he thinks has had a huge impact on the severity of his condition. (Both seafood and beer contain high levels of purines, which have been shown to increase the buildup of uric acid in the blood.) Mr. Settle said that other simple lifestyle changes like vitamin C would be an easy addition to his routine with few downsides. Plus, he hates having to take colchicine, a medication that’s meant to relieve pain but causes him intense diarrhea when he takes it.
“Anything to reduce my flare-ups without having to take colchicine,” he said.
But the jury is still out as to whether vitamin C will have any real benefits. Study coauthor Robert H. Shmerling, MD, is the former clinical chief of the division of rheumatology at Beth Israel Deaconess Medical Center, New York. He said the study shows that the effect of vitamin C in those undiagnosed with gout was rather modest. Also, vitamin C did not show a reduction in gout flare-ups in those who were already diagnosed with the condition. Not to mention that the study lacked diversity, as the people in it were all male and mostly white. Still, there’s little downside risk to taking vitamin C, and it might end up being worthwhile.
“Maybe it will turn out to be an effective treatment in those who are at high risk, but we’re not there yet,” he said.
Robert Terkeltaub, MD, chief of rheumatology at the Veterans Administration Medical Center in San Diego and a professor of medicine at the University of California, San Diego, said there’s an unmet need when it comes to tools for gout prevention.
“The disease impacts some 10 million Americans, and we need to better identify these individuals so we can intervene earlier,” he said.
While vitamin C had a small but significant association with fewer new cases of gout, it did not lower it in those who already had the disease, said Dr. Terkeltaub. What’s more, researchers didn’t measure the levels of uric acid in the blood, which would have painted a more accurate picture of whether vitamin C actually reduced it in the body.
“There remains no clarity on the potential role of vitamin C in either prevention or treatment of gout. That said, future research would be of interest,” he said.
Still, gout patients like Mr. Settle aren’t ruling it out. Anything to avoid the pain that, at times, makes it difficult for him to get out of bed. He’s seen the benefit that simple lifestyle changes can make, and he’s willing to try just about anything to live a normal, arthritis-free life.
“I’m always looking for simple ways to keep my flare-ups at bay,” he said.
A version of this article first appeared on WebMD.com.
FROM THE AMERICAN JOURNAL OF CLINICAL NUTRITION
OMERACT continues to set standards on research outcomes, enhancing the patient voice
Clinical research in rheumatology was suffering from an identity crisis of sorts 40 years ago. A lack of consensus across continents resulted in differing views about clinical outcome measures and judgments about treatments.
Patients were not allowed to be the generating source of a clinical outcome, according to Peter Tugwell, MSc, MD. “The only outcomes that were acceptable were clinician assessments, blood tests, and imaging,” said Dr. Tugwell, professor of medicine, epidemiology, and public health at the University of Ottawa (Ont.) and a practicing rheumatologist at Ottawa Hospital.
Clinicians were coming to different conclusions about patient responses to treatment when managing rheumatoid arthritis in clinical practice.
OMERACT sought to address this lack of uniformity. This international group, formed in 1992, leverages stakeholder groups to improve outcome measurement in rheumatology endpoints through a consensus-building, data-driven format.
It was originally known as “Outcome Measures in Rheumatoid Arthritis Clinical Trials,” but its leaders have since broadened its scope to “Outcome Measures in Rheumatology.” Over the years, it has evolved into an international network that assesses measurement across a wide variety of intervention studies. Now 30 years old, the network spans 40 active working groups and has influenced work in patient outcomes across 500 peer-reviewed publications.
The network meets every 2 years to address what is always a challenging agenda, said Dr. Tugwell, one of its founding members and chair. “There’s lots of strong opinions.” Participating in the discussions are individuals from all stages of seniority in rheumatology and clinical epidemiology, patient research partners, industry, approval agencies, and many countries who are committed to the spirit of OMERACT.
“The secret to our success has been getting world leaders to come together and have those discussions, work them through, and identify common ground in such a way that the approval agencies accept these outcome measures in clinical trials,” he added.
“My impression was the founders perceived a problem in the early 1990s and devised a consensus method in an attempt to quantify clinical parameters to define disease activity in rheumatoid arthritis – an important first step to do clinical trials and allow comparisons between them,” said Patricia Woo, CBE, FMedSci, FRCP, emeritus professor of pediatric rheumatology and previous head of the Centre for Paediatric and Adolescent Rheumatology at UCL, London. At that time, even disease definitions varied between the United States and Europe and other parts of the world, said Dr. Woo, who is not a part of OMERACT. “This was especially true for pediatric rheumatology.”
Fusing the continental divide
OMERACT arose from a need to streamline clinical outcome measures in rheumatology. Research papers during the 1980s demonstrated a lack of coherence in managing patients with rheumatoid arthritis in routine practice. In addition, the measures used to define clinical endpoints in clinical trials operated in silos – they were either too specific to a certain trial, overlapped with other concepts, or didn’t reflect changes in treatment.
Approval agencies in Europe and North America were approving only outcomes measures developed by their respective researchers. This was also true of patients they tested on. “This seemed crazy,” Dr. Tugwell said.
Dr. Tugwell was involved in the Cochrane collaboration, which conducts systematic reviews of best evidence across the world that assesses the magnitude of benefits versus harms.
To achieve this goal, “you need to pull studies from around the world,” he said. Maarten Boers, MD, PhD, a rheumatologist (and later professor of clinical epidemiology at Amsterdam University Medical Center) from the Netherlands, spent a year in Ontario, Canada, to train as a clinical epidemiologist. Together, Dr. Tugwell and Dr. Boers began discussing options to develop more streamlined outcome measures.
They initiated the first OMERACT conference in Maastricht, the Netherlands, in 1992. The Food and Drug Administration and European Medicines Agency participated, along with leaders of outcomes measurement in Europe and in North America.
Discussions centered on methods to develop outcomes in a meaningful fashion. During the first meeting, North American and European approval agencies agreed to accept each other’s studies and endpoints and patient reported outcomes.
Agreement was achieved on a preliminary set of outcome domains and measures that later became known as the WHO-ILAR (World Health Organization–International League of Associations for Rheumatology) core set. The set included seven outcome domains: tender joints, swollen joints, pain, physician global assessment, patient global assessment, physical disability, and acute phase reactants, and one additional outcome domain for studies lasting 1 year or more: radiographs of the joints.
“A proactive program was planned to test not only the validity of these endpoints, but also the methods for their measurement. This was the start of a continuing process,” OMERACT members said in a joint statement for this article. Meetings have since taken place every 2 years.

OMERACT accomplishments
OMERACT now requires buy-in from four continents: Asia, Australia, Europe, and North America.
Its leaders have developed an explicit process for gaining endorsement of core outcome domains and instrument measurement sets. To fully capture the possibilities of “what to measure,” i.e., “measurable aspects of health conditions,” OMERACT has developed a framework of concepts, core areas, and outcome domains. The key concepts are pathophysiology (with a core area termed “manifestations/abnormalities”) and impact (with core areas of “death/lifespan,” and “life impact,” and the optional area of “societal/resource use”). An outcome domain defines an element of a core area to measure the effects of a treatment, such as blood markers, pain intensity, physical function, or emotional well-being.
A core outcome domain set is developed by agreeing to at least one outcome domain within one of the three core areas. Subsequently, a core outcome measurement set is developed by agreeing to at least one applicable measurement instrument for each core outcome domain. This requires documentation of validity, summarized under three metrics: truth, discrimination, and feasibility.
OMERACT’s handbook provides tutelage on establishing and implementing core outcomes, and several workbooks offer guidance on developing core outcome domain sets, selecting instruments for core outcome measurement sets, and OMERACT methodology.
All this work has led to widespread adoption.
Approval agencies have accepted OMERACT’s filter and methods advances, which have been adopted by many research groups in rheumatology and among nonrheumatology research groups. Organizations such as the U.S. National Institutes of Health’s National Institute of Neurological Disorders and Stroke have sought its advice.
Its core outcomes have been adopted and used for approval in the great majority of studies on rheumatoid arthritis, Dr. Tugwell said.
Several BMJ articles underscore the influence and uptake of OMERACT’s core outcome set. One 2017 paper, which analyzed 273 randomized trials of rheumatoid arthritis drug treatments on ClinicalTrials.gov, found that the WHO-ILAR arthritis core outcome set was reported in 81% of the studies. “The adoption of a core outcome set has the potential to increase consistency in outcomes measured across trials and ensure that trials are more likely to measure appropriate outcomes,” the authors concluded.
Since the initial 1992 meeting, OMERACT has broadened its focus from rheumatoid arthritis to 25 other musculoskeletal conditions.
For example, other OMERACT conferences have led to consensus on core sets of measures for osteoarthritis and osteoporosis, psoriasis/psoriatic arthritis, psychosocial measures, and a core set of data for cost-effectiveness evaluations.
‘Speed is a limitation’
OMERACT is a bottom-up volunteer organization. It doesn’t represent any official organization of any clinical society. “We’ve not asked to be adopted by the American College of Rheumatology, EULAR [European Alliance of Associations for Rheumatology], or other international organizations,” Dr. Tugwell said. It offers a chance for patients, users, and doers of research to work together to agree on rigorous criteria accepted by the approval agencies and take the necessary time to work things through.
This is not a fast process, usually taking 4-6 years to initiate and establish an outcome domain set, he emphasized. “It would be beneficial to do it faster if we had the resources to meet every year. The fact is we’re a volunteer organization that meets every 2 years.”
Speed is a limitation, he acknowledged, but it’s an acceptable trade-off for doing things correctly.
The group has faced other challenges during the COVID-19 pandemic, pivoting to a virtual format that had benefits and limitations.
In one respect, moving to a virtual meeting increased uptake in participation and voting, Dr. Tugwell said. Patient participants with severe rheumatoid arthritis no longer faced the challenges of travel. “On the other hand, we didn’t have the same opportunity to achieve common ground virtually,” he said. “Where there are strong disagreements, I’m a great believer that people need to know one another. There needs to be relationship building.”
OMERACT’s emerging leader program has been a cornerstone of its in-person meetings, engaging young rheumatologists to interact with some of the leaders of outcome measurement. The virtual format dampened this process somewhat, eliminating those important “café chats” between the stakeholders.
The hope is to bring people face-to-face once more at the next meeting in May 2023. The agenda will focus on relationship building, identifying controversial areas, and bringing younger people to develop relationships, Dr. Tugwell said. OMERACT will retain a virtual option for the worldwide voting, “which will allow for more buy-in from so many more people,” he added.
A consensus on pain
The onus of developing outcome measures that move with the times is sometimes too great for one group to manage. In 2018, OMERACT became a part of the Red Hat Group (RHG), an organization conceived at the COMET (Core Outcome Measures in Effectiveness Trials) VII meeting in Amsterdam.
RHG aims to improve the choice of outcomes in health research. It includes eight groups: COMET; OMERACT; the Cochrane Skin Core Outcome Set Initiative; Grading of Recommendations, Assessment, Development and Evaluations; Center for Medical Technology Policy; COnsensus-based Standards for the selection of health Measurement Instruments; Clinical Data Interchange Standards Consortium; and Standardized Outcomes in Nephrology.
The collaboration between groups offers a “very interesting interface between consensus building as well as hard evidence,” Dr. Tugwell said. The focus goes beyond rheumatology to other clinical areas of common interest, exploring how one classifies outcome domains in terms of symptoms, life impact, or death.
Pain is an important common denominator that the RHG has evaluated.
“We believe it’s too general. We’re trying to define pain across all Red Hat Groups because it’s clear that the research community has all these different scales for defining pain severity,” Dr. Tugwell said. “We have to find a way to make ruthless decisions and rules for doing it. And of course, it has to be transparent.”
Looking ahead
As part of its ongoing work, OMERACT is evaluating the robustness of instruments that rheumatologists use as outcome measures in clinical trials, which can be a laborious process. The OMERACT Filter 2.0, part of the latest iteration of the handbook, offers strong guidance for researchers but needs a long-term strategy and key methodological support. “To that end, we set up a technical advisory group to help people in the instrument selection work and that remains an ongoing process,” OMERACT leaders said in their joint statement.
OMERACT is looking at opportunities to create benchmark processes for developing core sets outside of rheumatology and a methodology around outcome measures such as contextual factors, composites, and surrogates.
It will also be taking a step back to solicit opinions from the approval agencies represented by the OMERACT membership on the OMERACT handbook.
The goal is to make sure the handbook aligns with everyone else’s approval and labeling requirements.
OMERACT’s patient participants bring important perspectives
OMERACT over the years has sought to become a more patient-centered group. Patients have been involved in OMERACT activities since its sixth meeting, forming an independent, yet integrated, group within the network. They have their own steering committee and produced and helped to update a glossary for OMERACT patients and professionals.
Catherine (McGowan) Hofstetter, who was diagnosed with rheumatoid arthritis 30 years ago, chairs OMERACT’s Patient Research Partners Support Team. In a Q&A, she discussed the importance of patient voices and OMERACT’s plans to further educate and include patients in the dialogue on outcomes.
Question: Have patients always been a part of OMERACT meetings?
Answer: Patients have been involved with OMERACT since 2002. The patient voice adds relevance to all the work that OMERACT does. You can’t begin to talk about outcomes unless there is a patient at the table with lived experience.
Q: Can you cite a few examples of how the patient voice enriches the conversation on outcomes research?
A: Outcomes and priorities that are important to patients are often completely different than those of the clinician. For instance, a work outcome is important to someone who doesn’t have any medical insurance or disability insurance, so that you can ensure that there is food on the table and a roof over your head. Or it may be important to someone because the employment provides medical and disability insurance to provide security for them and their family. These are two different perspectives on work and therefore work priorities and outcomes.
Q: What have been some of the challenges of getting patients to participate?
A: Training patients is one challenge. OMERACT’s work has a very steep learning curve, and while the basics are the same between the groups in terms of looking at what we measure and how we measure it, the nuances of different working groups require a lot of time and energy to be comfortable enough with the work, and then be confident enough to bring your perspective and lived experience to the table. It’s also a very accomplished group, which can be quite intimidating. Self-disclosure is a very personal and intimate undertaking that requires patience, compassion, and respect.
Q: Are there any plans to enhance patient engagement?
A: When we had OMERACT 2020 it was a virtual conference that took place over about 6 months. We had far more patient research partners [PRPs] participate than we have ever had at any OMERACT face-to-face meeting. There is a desire and passion on the part of patients to lend their voices to the work. The working groups meet virtually throughout the year to advance their agendas, and PRPs are a part of each of the working groups.
Hopefully, we can start working toward including more voices at the conferences by enabling a hybrid model. The PRP Support Team will begin engaging patients this fall with education, mentoring, and team-building exercises so by the time we meet in person in May 2023, they will have enough background knowledge and information to give them the confidence that will enhance their experience at the face-to-face meeting.
We also need to ensure that those patients who want to stay engaged can. This means that the education and training should continue long after the face-to-face meeting is over. We need to build capacity in the PRP group and look to succession planning and be a resource to working groups struggling to find PRPs to work with them on a longer-term basis.
Clinical research in rheumatology was suffering from an identity crisis of sorts 40 years ago. A lack of consensus across continents resulted in differing views about clinical outcome measures and judgments about treatments.
Patients were not allowed to be the generating source of a clinical outcome, according to Peter Tugwell, MSc, MD. “The only outcomes that were acceptable were clinician assessments, blood tests, and imaging,” said Dr. Tugwell, professor of medicine, epidemiology, and public health at the University of Ottawa (Ont.) and a practicing rheumatologist at Ottawa Hospital.
Clinicians were coming to different conclusions about patient responses to treatment when managing rheumatoid arthritis in clinical practice.
OMERACT sought to address this lack of uniformity. This international group, formed in 1992, leverages stakeholder groups to improve outcome measurement in rheumatology endpoints through a consensus-building, data-driven format.
It was originally known as “Outcome Measures in Rheumatoid Arthritis Clinical Trials,” but its leaders have since broadened its scope to “Outcome Measures in Rheumatology.” Over the years, it has evolved into an international network that assesses measurement across a wide variety of intervention studies. Now 30 years old, the network spans 40 active working groups and has influenced work in patient outcomes across 500 peer-reviewed publications.
The network meets every 2 years to address what is always a challenging agenda, said Dr. Tugwell, one of its founding members and chair. “There’s lots of strong opinions.” Participating in the discussions are individuals from all stages of seniority in rheumatology and clinical epidemiology, patient research partners, industry, approval agencies, and many countries who are committed to the spirit of OMERACT.
“The secret to our success has been getting world leaders to come together and have those discussions, work them through, and identify common ground in such a way that the approval agencies accept these outcome measures in clinical trials,” he added.
“My impression was the founders perceived a problem in the early 1990s and devised a consensus method in an attempt to quantify clinical parameters to define disease activity in rheumatoid arthritis – an important first step to do clinical trials and allow comparisons between them,” said Patricia Woo, CBE, FMedSci, FRCP, emeritus professor of pediatric rheumatology and previous head of the Centre for Paediatric and Adolescent Rheumatology at UCL, London. At that time, even disease definitions varied between the United States and Europe and other parts of the world, said Dr. Woo, who is not a part of OMERACT. “This was especially true for pediatric rheumatology.”
Fusing the continental divide
OMERACT arose from a need to streamline clinical outcome measures in rheumatology. Research papers during the 1980s demonstrated a lack of coherence in managing patients with rheumatoid arthritis in routine practice. In addition, the measures used to define clinical endpoints in clinical trials operated in silos – they were either too specific to a certain trial, overlapped with other concepts, or didn’t reflect changes in treatment.
Approval agencies in Europe and North America were approving only outcomes measures developed by their respective researchers. This was also true of patients they tested on. “This seemed crazy,” Dr. Tugwell said.
Dr. Tugwell was involved in the Cochrane collaboration, which conducts systematic reviews of best evidence across the world that assesses the magnitude of benefits versus harms.
To achieve this goal, “you need to pull studies from around the world,” he said. Maarten Boers, MD, PhD, a rheumatologist (and later professor of clinical epidemiology at Amsterdam University Medical Center) from the Netherlands, spent a year in Ontario, Canada, to train as a clinical epidemiologist. Together, Dr. Tugwell and Dr. Boers began discussing options to develop more streamlined outcome measures.
They initiated the first OMERACT conference in Maastricht, the Netherlands, in 1992. The Food and Drug Administration and European Medicines Agency participated, along with leaders of outcomes measurement in Europe and in North America.
Discussions centered on methods to develop outcomes in a meaningful fashion. During the first meeting, North American and European approval agencies agreed to accept each other’s studies and endpoints and patient reported outcomes.
Agreement was achieved on a preliminary set of outcome domains and measures that later became known as the WHO-ILAR (World Health Organization–International League of Associations for Rheumatology) core set. The set included seven outcome domains: tender joints, swollen joints, pain, physician global assessment, patient global assessment, physical disability, and acute phase reactants, and one additional outcome domain for studies lasting 1 year or more: radiographs of the joints.
“A proactive program was planned to test not only the validity of these endpoints, but also the methods for their measurement. This was the start of a continuing process,” OMERACT members said in a joint statement for this article. Meetings have since taken place every 2 years.

OMERACT accomplishments
OMERACT now requires buy-in from four continents: Asia, Australia, Europe, and North America.
Its leaders have developed an explicit process for gaining endorsement of core outcome domains and instrument measurement sets. To fully capture the possibilities of “what to measure,” i.e., “measurable aspects of health conditions,” OMERACT has developed a framework of concepts, core areas, and outcome domains. The key concepts are pathophysiology (with a core area termed “manifestations/abnormalities”) and impact (with core areas of “death/lifespan,” and “life impact,” and the optional area of “societal/resource use”). An outcome domain defines an element of a core area to measure the effects of a treatment, such as blood markers, pain intensity, physical function, or emotional well-being.
A core outcome domain set is developed by agreeing to at least one outcome domain within one of the three core areas. Subsequently, a core outcome measurement set is developed by agreeing to at least one applicable measurement instrument for each core outcome domain. This requires documentation of validity, summarized under three metrics: truth, discrimination, and feasibility.
OMERACT’s handbook provides tutelage on establishing and implementing core outcomes, and several workbooks offer guidance on developing core outcome domain sets, selecting instruments for core outcome measurement sets, and OMERACT methodology.
All this work has led to widespread adoption.
Approval agencies have accepted OMERACT’s filter and methods advances, which have been adopted by many research groups in rheumatology and among nonrheumatology research groups. Organizations such as the U.S. National Institutes of Health’s National Institute of Neurological Disorders and Stroke have sought its advice.
Its core outcomes have been adopted and used for approval in the great majority of studies on rheumatoid arthritis, Dr. Tugwell said.
Several BMJ articles underscore the influence and uptake of OMERACT’s core outcome set. One 2017 paper, which analyzed 273 randomized trials of rheumatoid arthritis drug treatments on ClinicalTrials.gov, found that the WHO-ILAR arthritis core outcome set was reported in 81% of the studies. “The adoption of a core outcome set has the potential to increase consistency in outcomes measured across trials and ensure that trials are more likely to measure appropriate outcomes,” the authors concluded.
Since the initial 1992 meeting, OMERACT has broadened its focus from rheumatoid arthritis to 25 other musculoskeletal conditions.
For example, other OMERACT conferences have led to consensus on core sets of measures for osteoarthritis and osteoporosis, psoriasis/psoriatic arthritis, psychosocial measures, and a core set of data for cost-effectiveness evaluations.
‘Speed is a limitation’
OMERACT is a bottom-up volunteer organization. It doesn’t represent any official organization of any clinical society. “We’ve not asked to be adopted by the American College of Rheumatology, EULAR [European Alliance of Associations for Rheumatology], or other international organizations,” Dr. Tugwell said. It offers a chance for patients, users, and doers of research to work together to agree on rigorous criteria accepted by the approval agencies and take the necessary time to work things through.
This is not a fast process, usually taking 4-6 years to initiate and establish an outcome domain set, he emphasized. “It would be beneficial to do it faster if we had the resources to meet every year. The fact is we’re a volunteer organization that meets every 2 years.”
Speed is a limitation, he acknowledged, but it’s an acceptable trade-off for doing things correctly.
The group has faced other challenges during the COVID-19 pandemic, pivoting to a virtual format that had benefits and limitations.
In one respect, moving to a virtual meeting increased uptake in participation and voting, Dr. Tugwell said. Patient participants with severe rheumatoid arthritis no longer faced the challenges of travel. “On the other hand, we didn’t have the same opportunity to achieve common ground virtually,” he said. “Where there are strong disagreements, I’m a great believer that people need to know one another. There needs to be relationship building.”
OMERACT’s emerging leader program has been a cornerstone of its in-person meetings, engaging young rheumatologists to interact with some of the leaders of outcome measurement. The virtual format dampened this process somewhat, eliminating those important “café chats” between the stakeholders.
The hope is to bring people face-to-face once more at the next meeting in May 2023. The agenda will focus on relationship building, identifying controversial areas, and bringing younger people to develop relationships, Dr. Tugwell said. OMERACT will retain a virtual option for the worldwide voting, “which will allow for more buy-in from so many more people,” he added.
A consensus on pain
The onus of developing outcome measures that move with the times is sometimes too great for one group to manage. In 2018, OMERACT became a part of the Red Hat Group (RHG), an organization conceived at the COMET (Core Outcome Measures in Effectiveness Trials) VII meeting in Amsterdam.
RHG aims to improve the choice of outcomes in health research. It includes eight groups: COMET; OMERACT; the Cochrane Skin Core Outcome Set Initiative; Grading of Recommendations, Assessment, Development and Evaluations; Center for Medical Technology Policy; COnsensus-based Standards for the selection of health Measurement Instruments; Clinical Data Interchange Standards Consortium; and Standardized Outcomes in Nephrology.
The collaboration between groups offers a “very interesting interface between consensus building as well as hard evidence,” Dr. Tugwell said. The focus goes beyond rheumatology to other clinical areas of common interest, exploring how one classifies outcome domains in terms of symptoms, life impact, or death.
Pain is an important common denominator that the RHG has evaluated.
“We believe it’s too general. We’re trying to define pain across all Red Hat Groups because it’s clear that the research community has all these different scales for defining pain severity,” Dr. Tugwell said. “We have to find a way to make ruthless decisions and rules for doing it. And of course, it has to be transparent.”
Looking ahead
As part of its ongoing work, OMERACT is evaluating the robustness of instruments that rheumatologists use as outcome measures in clinical trials, which can be a laborious process. The OMERACT Filter 2.0, part of the latest iteration of the handbook, offers strong guidance for researchers but needs a long-term strategy and key methodological support. “To that end, we set up a technical advisory group to help people in the instrument selection work and that remains an ongoing process,” OMERACT leaders said in their joint statement.
OMERACT is looking at opportunities to create benchmark processes for developing core sets outside of rheumatology and a methodology around outcome measures such as contextual factors, composites, and surrogates.
It will also be taking a step back to solicit opinions from the approval agencies represented by the OMERACT membership on the OMERACT handbook.
The goal is to make sure the handbook aligns with everyone else’s approval and labeling requirements.
OMERACT’s patient participants bring important perspectives
OMERACT over the years has sought to become a more patient-centered group. Patients have been involved in OMERACT activities since its sixth meeting, forming an independent, yet integrated, group within the network. They have their own steering committee and produced and helped to update a glossary for OMERACT patients and professionals.
Catherine (McGowan) Hofstetter, who was diagnosed with rheumatoid arthritis 30 years ago, chairs OMERACT’s Patient Research Partners Support Team. In a Q&A, she discussed the importance of patient voices and OMERACT’s plans to further educate and include patients in the dialogue on outcomes.
Question: Have patients always been a part of OMERACT meetings?
Answer: Patients have been involved with OMERACT since 2002. The patient voice adds relevance to all the work that OMERACT does. You can’t begin to talk about outcomes unless there is a patient at the table with lived experience.
Q: Can you cite a few examples of how the patient voice enriches the conversation on outcomes research?
A: Outcomes and priorities that are important to patients are often completely different than those of the clinician. For instance, a work outcome is important to someone who doesn’t have any medical insurance or disability insurance, so that you can ensure that there is food on the table and a roof over your head. Or it may be important to someone because the employment provides medical and disability insurance to provide security for them and their family. These are two different perspectives on work and therefore work priorities and outcomes.
Q: What have been some of the challenges of getting patients to participate?
A: Training patients is one challenge. OMERACT’s work has a very steep learning curve, and while the basics are the same between the groups in terms of looking at what we measure and how we measure it, the nuances of different working groups require a lot of time and energy to be comfortable enough with the work, and then be confident enough to bring your perspective and lived experience to the table. It’s also a very accomplished group, which can be quite intimidating. Self-disclosure is a very personal and intimate undertaking that requires patience, compassion, and respect.
Q: Are there any plans to enhance patient engagement?
A: When we had OMERACT 2020 it was a virtual conference that took place over about 6 months. We had far more patient research partners [PRPs] participate than we have ever had at any OMERACT face-to-face meeting. There is a desire and passion on the part of patients to lend their voices to the work. The working groups meet virtually throughout the year to advance their agendas, and PRPs are a part of each of the working groups.
Hopefully, we can start working toward including more voices at the conferences by enabling a hybrid model. The PRP Support Team will begin engaging patients this fall with education, mentoring, and team-building exercises so by the time we meet in person in May 2023, they will have enough background knowledge and information to give them the confidence that will enhance their experience at the face-to-face meeting.
We also need to ensure that those patients who want to stay engaged can. This means that the education and training should continue long after the face-to-face meeting is over. We need to build capacity in the PRP group and look to succession planning and be a resource to working groups struggling to find PRPs to work with them on a longer-term basis.
Clinical research in rheumatology was suffering from an identity crisis of sorts 40 years ago. A lack of consensus across continents resulted in differing views about clinical outcome measures and judgments about treatments.
Patients were not allowed to be the generating source of a clinical outcome, according to Peter Tugwell, MSc, MD. “The only outcomes that were acceptable were clinician assessments, blood tests, and imaging,” said Dr. Tugwell, professor of medicine, epidemiology, and public health at the University of Ottawa (Ont.) and a practicing rheumatologist at Ottawa Hospital.
Clinicians were coming to different conclusions about patient responses to treatment when managing rheumatoid arthritis in clinical practice.
OMERACT sought to address this lack of uniformity. This international group, formed in 1992, leverages stakeholder groups to improve outcome measurement in rheumatology endpoints through a consensus-building, data-driven format.
It was originally known as “Outcome Measures in Rheumatoid Arthritis Clinical Trials,” but its leaders have since broadened its scope to “Outcome Measures in Rheumatology.” Over the years, it has evolved into an international network that assesses measurement across a wide variety of intervention studies. Now 30 years old, the network spans 40 active working groups and has influenced work in patient outcomes across 500 peer-reviewed publications.
The network meets every 2 years to address what is always a challenging agenda, said Dr. Tugwell, one of its founding members and chair. “There’s lots of strong opinions.” Participating in the discussions are individuals from all stages of seniority in rheumatology and clinical epidemiology, patient research partners, industry, approval agencies, and many countries who are committed to the spirit of OMERACT.
“The secret to our success has been getting world leaders to come together and have those discussions, work them through, and identify common ground in such a way that the approval agencies accept these outcome measures in clinical trials,” he added.
“My impression was the founders perceived a problem in the early 1990s and devised a consensus method in an attempt to quantify clinical parameters to define disease activity in rheumatoid arthritis – an important first step to do clinical trials and allow comparisons between them,” said Patricia Woo, CBE, FMedSci, FRCP, emeritus professor of pediatric rheumatology and previous head of the Centre for Paediatric and Adolescent Rheumatology at UCL, London. At that time, even disease definitions varied between the United States and Europe and other parts of the world, said Dr. Woo, who is not a part of OMERACT. “This was especially true for pediatric rheumatology.”
Fusing the continental divide
OMERACT arose from a need to streamline clinical outcome measures in rheumatology. Research papers during the 1980s demonstrated a lack of coherence in managing patients with rheumatoid arthritis in routine practice. In addition, the measures used to define clinical endpoints in clinical trials operated in silos – they were either too specific to a certain trial, overlapped with other concepts, or didn’t reflect changes in treatment.
Approval agencies in Europe and North America were approving only outcomes measures developed by their respective researchers. This was also true of patients they tested on. “This seemed crazy,” Dr. Tugwell said.
Dr. Tugwell was involved in the Cochrane collaboration, which conducts systematic reviews of best evidence across the world that assesses the magnitude of benefits versus harms.
To achieve this goal, “you need to pull studies from around the world,” he said. Maarten Boers, MD, PhD, a rheumatologist (and later professor of clinical epidemiology at Amsterdam University Medical Center) from the Netherlands, spent a year in Ontario, Canada, to train as a clinical epidemiologist. Together, Dr. Tugwell and Dr. Boers began discussing options to develop more streamlined outcome measures.
They initiated the first OMERACT conference in Maastricht, the Netherlands, in 1992. The Food and Drug Administration and European Medicines Agency participated, along with leaders of outcomes measurement in Europe and in North America.
Discussions centered on methods to develop outcomes in a meaningful fashion. During the first meeting, North American and European approval agencies agreed to accept each other’s studies and endpoints and patient reported outcomes.
Agreement was achieved on a preliminary set of outcome domains and measures that later became known as the WHO-ILAR (World Health Organization–International League of Associations for Rheumatology) core set. The set included seven outcome domains: tender joints, swollen joints, pain, physician global assessment, patient global assessment, physical disability, and acute phase reactants, and one additional outcome domain for studies lasting 1 year or more: radiographs of the joints.
“A proactive program was planned to test not only the validity of these endpoints, but also the methods for their measurement. This was the start of a continuing process,” OMERACT members said in a joint statement for this article. Meetings have since taken place every 2 years.

OMERACT accomplishments
OMERACT now requires buy-in from four continents: Asia, Australia, Europe, and North America.
Its leaders have developed an explicit process for gaining endorsement of core outcome domains and instrument measurement sets. To fully capture the possibilities of “what to measure,” i.e., “measurable aspects of health conditions,” OMERACT has developed a framework of concepts, core areas, and outcome domains. The key concepts are pathophysiology (with a core area termed “manifestations/abnormalities”) and impact (with core areas of “death/lifespan,” and “life impact,” and the optional area of “societal/resource use”). An outcome domain defines an element of a core area to measure the effects of a treatment, such as blood markers, pain intensity, physical function, or emotional well-being.
A core outcome domain set is developed by agreeing to at least one outcome domain within one of the three core areas. Subsequently, a core outcome measurement set is developed by agreeing to at least one applicable measurement instrument for each core outcome domain. This requires documentation of validity, summarized under three metrics: truth, discrimination, and feasibility.
OMERACT’s handbook provides tutelage on establishing and implementing core outcomes, and several workbooks offer guidance on developing core outcome domain sets, selecting instruments for core outcome measurement sets, and OMERACT methodology.
All this work has led to widespread adoption.
Approval agencies have accepted OMERACT’s filter and methods advances, which have been adopted by many research groups in rheumatology and among nonrheumatology research groups. Organizations such as the U.S. National Institutes of Health’s National Institute of Neurological Disorders and Stroke have sought its advice.
Its core outcomes have been adopted and used for approval in the great majority of studies on rheumatoid arthritis, Dr. Tugwell said.
Several BMJ articles underscore the influence and uptake of OMERACT’s core outcome set. One 2017 paper, which analyzed 273 randomized trials of rheumatoid arthritis drug treatments on ClinicalTrials.gov, found that the WHO-ILAR arthritis core outcome set was reported in 81% of the studies. “The adoption of a core outcome set has the potential to increase consistency in outcomes measured across trials and ensure that trials are more likely to measure appropriate outcomes,” the authors concluded.
Since the initial 1992 meeting, OMERACT has broadened its focus from rheumatoid arthritis to 25 other musculoskeletal conditions.
For example, other OMERACT conferences have led to consensus on core sets of measures for osteoarthritis and osteoporosis, psoriasis/psoriatic arthritis, psychosocial measures, and a core set of data for cost-effectiveness evaluations.
‘Speed is a limitation’
OMERACT is a bottom-up volunteer organization. It doesn’t represent any official organization of any clinical society. “We’ve not asked to be adopted by the American College of Rheumatology, EULAR [European Alliance of Associations for Rheumatology], or other international organizations,” Dr. Tugwell said. It offers a chance for patients, users, and doers of research to work together to agree on rigorous criteria accepted by the approval agencies and take the necessary time to work things through.
This is not a fast process, usually taking 4-6 years to initiate and establish an outcome domain set, he emphasized. “It would be beneficial to do it faster if we had the resources to meet every year. The fact is we’re a volunteer organization that meets every 2 years.”
Speed is a limitation, he acknowledged, but it’s an acceptable trade-off for doing things correctly.
The group has faced other challenges during the COVID-19 pandemic, pivoting to a virtual format that had benefits and limitations.
In one respect, moving to a virtual meeting increased uptake in participation and voting, Dr. Tugwell said. Patient participants with severe rheumatoid arthritis no longer faced the challenges of travel. “On the other hand, we didn’t have the same opportunity to achieve common ground virtually,” he said. “Where there are strong disagreements, I’m a great believer that people need to know one another. There needs to be relationship building.”
OMERACT’s emerging leader program has been a cornerstone of its in-person meetings, engaging young rheumatologists to interact with some of the leaders of outcome measurement. The virtual format dampened this process somewhat, eliminating those important “café chats” between the stakeholders.
The hope is to bring people face-to-face once more at the next meeting in May 2023. The agenda will focus on relationship building, identifying controversial areas, and bringing younger people to develop relationships, Dr. Tugwell said. OMERACT will retain a virtual option for the worldwide voting, “which will allow for more buy-in from so many more people,” he added.
A consensus on pain
The onus of developing outcome measures that move with the times is sometimes too great for one group to manage. In 2018, OMERACT became a part of the Red Hat Group (RHG), an organization conceived at the COMET (Core Outcome Measures in Effectiveness Trials) VII meeting in Amsterdam.
RHG aims to improve the choice of outcomes in health research. It includes eight groups: COMET; OMERACT; the Cochrane Skin Core Outcome Set Initiative; Grading of Recommendations, Assessment, Development and Evaluations; Center for Medical Technology Policy; COnsensus-based Standards for the selection of health Measurement Instruments; Clinical Data Interchange Standards Consortium; and Standardized Outcomes in Nephrology.
The collaboration between groups offers a “very interesting interface between consensus building as well as hard evidence,” Dr. Tugwell said. The focus goes beyond rheumatology to other clinical areas of common interest, exploring how one classifies outcome domains in terms of symptoms, life impact, or death.
Pain is an important common denominator that the RHG has evaluated.
“We believe it’s too general. We’re trying to define pain across all Red Hat Groups because it’s clear that the research community has all these different scales for defining pain severity,” Dr. Tugwell said. “We have to find a way to make ruthless decisions and rules for doing it. And of course, it has to be transparent.”
Looking ahead
As part of its ongoing work, OMERACT is evaluating the robustness of instruments that rheumatologists use as outcome measures in clinical trials, which can be a laborious process. The OMERACT Filter 2.0, part of the latest iteration of the handbook, offers strong guidance for researchers but needs a long-term strategy and key methodological support. “To that end, we set up a technical advisory group to help people in the instrument selection work and that remains an ongoing process,” OMERACT leaders said in their joint statement.
OMERACT is looking at opportunities to create benchmark processes for developing core sets outside of rheumatology and a methodology around outcome measures such as contextual factors, composites, and surrogates.
It will also be taking a step back to solicit opinions from the approval agencies represented by the OMERACT membership on the OMERACT handbook.
The goal is to make sure the handbook aligns with everyone else’s approval and labeling requirements.
OMERACT’s patient participants bring important perspectives
OMERACT over the years has sought to become a more patient-centered group. Patients have been involved in OMERACT activities since its sixth meeting, forming an independent, yet integrated, group within the network. They have their own steering committee and produced and helped to update a glossary for OMERACT patients and professionals.
Catherine (McGowan) Hofstetter, who was diagnosed with rheumatoid arthritis 30 years ago, chairs OMERACT’s Patient Research Partners Support Team. In a Q&A, she discussed the importance of patient voices and OMERACT’s plans to further educate and include patients in the dialogue on outcomes.
Question: Have patients always been a part of OMERACT meetings?
Answer: Patients have been involved with OMERACT since 2002. The patient voice adds relevance to all the work that OMERACT does. You can’t begin to talk about outcomes unless there is a patient at the table with lived experience.
Q: Can you cite a few examples of how the patient voice enriches the conversation on outcomes research?
A: Outcomes and priorities that are important to patients are often completely different than those of the clinician. For instance, a work outcome is important to someone who doesn’t have any medical insurance or disability insurance, so that you can ensure that there is food on the table and a roof over your head. Or it may be important to someone because the employment provides medical and disability insurance to provide security for them and their family. These are two different perspectives on work and therefore work priorities and outcomes.
Q: What have been some of the challenges of getting patients to participate?
A: Training patients is one challenge. OMERACT’s work has a very steep learning curve, and while the basics are the same between the groups in terms of looking at what we measure and how we measure it, the nuances of different working groups require a lot of time and energy to be comfortable enough with the work, and then be confident enough to bring your perspective and lived experience to the table. It’s also a very accomplished group, which can be quite intimidating. Self-disclosure is a very personal and intimate undertaking that requires patience, compassion, and respect.
Q: Are there any plans to enhance patient engagement?
A: When we had OMERACT 2020 it was a virtual conference that took place over about 6 months. We had far more patient research partners [PRPs] participate than we have ever had at any OMERACT face-to-face meeting. There is a desire and passion on the part of patients to lend their voices to the work. The working groups meet virtually throughout the year to advance their agendas, and PRPs are a part of each of the working groups.
Hopefully, we can start working toward including more voices at the conferences by enabling a hybrid model. The PRP Support Team will begin engaging patients this fall with education, mentoring, and team-building exercises so by the time we meet in person in May 2023, they will have enough background knowledge and information to give them the confidence that will enhance their experience at the face-to-face meeting.
We also need to ensure that those patients who want to stay engaged can. This means that the education and training should continue long after the face-to-face meeting is over. We need to build capacity in the PRP group and look to succession planning and be a resource to working groups struggling to find PRPs to work with them on a longer-term basis.
ALL-HEART: No benefit of allopurinol in ischemic heart disease
Allopurinol, a drug commonly used to treat gout, provided no benefit in terms of reducing cardiovascular (CV) events in patients with ischemic heart disease, new randomized trial results show.
Treatment of these patients without gout with 600 mg of allopurinol daily had no effect on composite primary endpoint outcomes, including nonfatal MI, nonfatal stroke, or CV death.
“ALL-HEART is the first large, prospective, randomized trial of the effect of allopurinol on major cardiovascular outcomes in patients with ischemic heart disease and provides robust evidence on the role of allopurinol in these patients,” principal investigator Isla Shelagh Mackenzie, MBChB (Honors), PhD, University of Dundee (Scotland), concluded at a press conference.
Their results suggest allopurinol should not be recommended for secondary prevention of events in this group, Dr. Mackenzie said. Although it remains an important treatment for gout, she added, “other avenues for treatment of ischemic heart disease should be explored in future.”
Results of the ALL-HEART (Allopurinol and Cardiovascular Outcomes in Ischemic Heart Disease) trial were presented at the annual congress of the European Society of Cardiology.
Gout treatment
Allopurinol is a xanthine oxidase inhibitor and acts by reducing serum uric acid levels and oxidative stress. Treatment is generally well tolerated, Dr. Mackenzie noted in her presentation, but some patients develop a rash, which can in some cases be serious or even fatal, progressing to Stevens-Johnson syndrome or toxic epidermal necrolysis, “particularly in certain ethnicities.” If rash develops, the advice is to stop treatment immediately.
“The importance of serum uric acid levels in cardiovascular disease is controversial, and there have been different reports over the years of how important they may be,” Dr. Mackenzie explained.
Observational studies have shown variable results, whereas intervention trials, most with fewer than 100 participants, have suggested potential improvements in factors such as blood pressure, endothelial function, left ventricular hypertrophy, or carotid intima-media thickness. Some have reported benefits in acute coronary syndrome and coronary artery bypass grafting, but others have not, she said. A previous study by their own group suggested an improvement in chest pain and exercise time in patients with chronic stable angina and documented coronary artery disease (CAD).
“So, until now, there have been no large prospective randomized trials of the effects of allopurinol on major cardiovascular outcomes in patients with ischemic heart disease,” Dr. Mackenzie said, and this was the aim of ALL-HEART.
ALL-HEART was a prospective, randomized, open-label, blinded-endpoint, multicenter trial. Patients with ischemia heart disease but no history of gout were recruited from 424 general practices across the United Kingdom, starting in February 2014 and with follow-up ending in September 2021. Participants were randomly assigned 1:1 to receive 600 mg of allopurinol daily or usual care.
“It was a decentralized trial, so the follow-up was largely remote after the first 6 weeks, and that included using record linkage data collected from centralized NHS [National Health Service] databases for hospitalizations and deaths in Scotland and England,” she said. The average follow-up was 4.8 years.
During that time, 258 (9.0%) participants in the allopurinol group and 76 (2.6%) in usual care withdrew from follow-up. By the end of the trial, 57.4% of patients in the allopurinol arm withdrew from randomized treatment.
Mean serum uric acid levels dropped from 0.34 mmol/L at baseline to 0.18 mmol/L at 6 weeks of treatment, “so we can see that the treatment was effective at lowering uric acid,” she noted.
In total, there were 5,721 patients in the final intention-to-treat analysis, and 639 patients had a first primary event.
For the primary outcome of nonfatal MI, nonfatal stroke, and cardiovascular death, there was no difference between the groups, the researchers reported, with a hazard ratio of 1.04 (95% confidence interval, 0.89-1.21; P = .65). Similarly, in secondary analyses, there were no differences in any of the component endpoints making up the primary outcome (nonfatal MI: HR, 0.97; 95% CI, 0.78-1.21; P = .81; nonfatal stroke: HR, 1.20; 95% CI, 0.89-1.60; P = .23; cardiovascular death: HR, 1.10; 95% CI, 0.85-1.43; P = .48), or in all-cause mortality (HR, 1.02; 95% CI, 0.87-1.20; P = .77), between the two groups, Dr. Mackenzie noted, “so a definitively neutral trial all round.”
In addition, no differences were seen in prespecified subgroups, including age, sex, estimated glomerular filtration rate, or diabetes, MI, heart failure, peripheral arterial disease, stroke, and stroke or transient ischemic attack at baseline.
There were also no significant effects on quality of life outcomes. Cost-effectiveness analyses are ongoing, although no differences are expected there, Dr. Mackenzie noted.
In terms of safety, incident cancers and all-cause mortality did not differ between groups. Serious adverse events were also similar between groups, Dr. Mackenzie said, “and there were no fatal treatment-related SAEs [serious adverse events] in the study.”
Another negative antioxidant trial
Invited discussant for the presentation, Leslie Cho, MD, of the Cleveland Clinic said that ALL-HEART, while an excellent trial with a pragmatic design, constitutes yet another negative antioxidant trial.
She pointed to three problems with this study and antioxidant trials in general. “First, the problem is with the antioxidant,” a xanthine oxidase inhibitor. “Xanthine oxidase is not a major trigger of oxidative stress. In a field of major players,” including nitric oxide, uncoupled endothelial nitric oxide synthase, and mitochondria myeloperoxidase, Dr. Cho said, “xanthine oxidase is a minor player.”
“Moreover, 57% of the patients stopped taking allopurinol, and rightfully so,” she said. Patients were receiving optimal medical therapies, many of which are also antioxidants, including statins, ACE inhibitors, angiotensin receptor blockers, and beta-blockers.
Second, the patient population was older, with an average age of 72 years. “This makes the ALL-HEART study a chronic angina study, chronic CAD study, one of the oldest modern day CAD trials. If you look at LoDoCo or ISCHEMIA trials, the average age is 63.” Patients also had established disease, many with previous revascularization.
The final issue seen with this trial, and all antioxidant trials, is that patient selection is not based on oxidative stress or antioxidant level. “The antioxidant trials have been disappointing at best. There is clear and convincing evidence that oxidative stress is involved in the pathogenesis of atherosclerosis, and yet study after study of antioxidant trials have been negative,” she said.
“Currently, there is no reliable measurement of global level of oxidative stress,” Dr. Cho noted. “Moreover, dose response was not tested, and if we cannot test the baseline antioxidant stress level of patients, we also cannot measure the effect of treatment on the global oxidative stress.”
So, “is there no hope for antioxidant trials?” she asked. Three factors will be required for future success, she said. “No. 1, selecting the right patient at the right time. No. 2, a reliable biomarker to measure oxidative stress to guide who should get therapy, and if the therapy is working. And lastly, targeted therapies that work on major triggers of oxidative stress.”
Also commenting on the results, B. Hadley Wilson, MD, executive vice chair of the Sanger Heart & Vascular Institute/Atrium Health, clinical professor of medicine at the University of North Carolina at Chapel Hill, and vice president of the American College of Cardiology, called ALL-HEART “an important and interesting study.”
“For years, cardiologists and others have been interested in allopurinol as an anti-inflammatory, xanthine oxidase inhibitor ... to prevent coronary ischemic events,” he said in an interview.
But this was a well-designed, well-conducted study, and “unfortunately there was no improvement in the primary outcome, no reduction in major cardiovascular events like myocardial infarction or stroke or cardiovascular death,” Dr. Wilson said. “So, it’s a bit of a disappointment that it’s not there as an important medication to help us with these patients with ischemic heart disease, but it’s also an important question answered — that we need to look at treatments for ischemic heart disease other than allopurinol.”
The trial was supported by the National Institute for Health and Care Research Health Technology Assessment Program in the United Kingdom. Dr. Mackenzie reported research contracts to her institution from NIHR HTA for this work, and other disclosures related to other work. Dr. Cho and Dr. Wilson reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
Allopurinol, a drug commonly used to treat gout, provided no benefit in terms of reducing cardiovascular (CV) events in patients with ischemic heart disease, new randomized trial results show.
Treatment of these patients without gout with 600 mg of allopurinol daily had no effect on composite primary endpoint outcomes, including nonfatal MI, nonfatal stroke, or CV death.
“ALL-HEART is the first large, prospective, randomized trial of the effect of allopurinol on major cardiovascular outcomes in patients with ischemic heart disease and provides robust evidence on the role of allopurinol in these patients,” principal investigator Isla Shelagh Mackenzie, MBChB (Honors), PhD, University of Dundee (Scotland), concluded at a press conference.
Their results suggest allopurinol should not be recommended for secondary prevention of events in this group, Dr. Mackenzie said. Although it remains an important treatment for gout, she added, “other avenues for treatment of ischemic heart disease should be explored in future.”
Results of the ALL-HEART (Allopurinol and Cardiovascular Outcomes in Ischemic Heart Disease) trial were presented at the annual congress of the European Society of Cardiology.
Gout treatment
Allopurinol is a xanthine oxidase inhibitor and acts by reducing serum uric acid levels and oxidative stress. Treatment is generally well tolerated, Dr. Mackenzie noted in her presentation, but some patients develop a rash, which can in some cases be serious or even fatal, progressing to Stevens-Johnson syndrome or toxic epidermal necrolysis, “particularly in certain ethnicities.” If rash develops, the advice is to stop treatment immediately.
“The importance of serum uric acid levels in cardiovascular disease is controversial, and there have been different reports over the years of how important they may be,” Dr. Mackenzie explained.
Observational studies have shown variable results, whereas intervention trials, most with fewer than 100 participants, have suggested potential improvements in factors such as blood pressure, endothelial function, left ventricular hypertrophy, or carotid intima-media thickness. Some have reported benefits in acute coronary syndrome and coronary artery bypass grafting, but others have not, she said. A previous study by their own group suggested an improvement in chest pain and exercise time in patients with chronic stable angina and documented coronary artery disease (CAD).
“So, until now, there have been no large prospective randomized trials of the effects of allopurinol on major cardiovascular outcomes in patients with ischemic heart disease,” Dr. Mackenzie said, and this was the aim of ALL-HEART.
ALL-HEART was a prospective, randomized, open-label, blinded-endpoint, multicenter trial. Patients with ischemia heart disease but no history of gout were recruited from 424 general practices across the United Kingdom, starting in February 2014 and with follow-up ending in September 2021. Participants were randomly assigned 1:1 to receive 600 mg of allopurinol daily or usual care.
“It was a decentralized trial, so the follow-up was largely remote after the first 6 weeks, and that included using record linkage data collected from centralized NHS [National Health Service] databases for hospitalizations and deaths in Scotland and England,” she said. The average follow-up was 4.8 years.
During that time, 258 (9.0%) participants in the allopurinol group and 76 (2.6%) in usual care withdrew from follow-up. By the end of the trial, 57.4% of patients in the allopurinol arm withdrew from randomized treatment.
Mean serum uric acid levels dropped from 0.34 mmol/L at baseline to 0.18 mmol/L at 6 weeks of treatment, “so we can see that the treatment was effective at lowering uric acid,” she noted.
In total, there were 5,721 patients in the final intention-to-treat analysis, and 639 patients had a first primary event.
For the primary outcome of nonfatal MI, nonfatal stroke, and cardiovascular death, there was no difference between the groups, the researchers reported, with a hazard ratio of 1.04 (95% confidence interval, 0.89-1.21; P = .65). Similarly, in secondary analyses, there were no differences in any of the component endpoints making up the primary outcome (nonfatal MI: HR, 0.97; 95% CI, 0.78-1.21; P = .81; nonfatal stroke: HR, 1.20; 95% CI, 0.89-1.60; P = .23; cardiovascular death: HR, 1.10; 95% CI, 0.85-1.43; P = .48), or in all-cause mortality (HR, 1.02; 95% CI, 0.87-1.20; P = .77), between the two groups, Dr. Mackenzie noted, “so a definitively neutral trial all round.”
In addition, no differences were seen in prespecified subgroups, including age, sex, estimated glomerular filtration rate, or diabetes, MI, heart failure, peripheral arterial disease, stroke, and stroke or transient ischemic attack at baseline.
There were also no significant effects on quality of life outcomes. Cost-effectiveness analyses are ongoing, although no differences are expected there, Dr. Mackenzie noted.
In terms of safety, incident cancers and all-cause mortality did not differ between groups. Serious adverse events were also similar between groups, Dr. Mackenzie said, “and there were no fatal treatment-related SAEs [serious adverse events] in the study.”
Another negative antioxidant trial
Invited discussant for the presentation, Leslie Cho, MD, of the Cleveland Clinic said that ALL-HEART, while an excellent trial with a pragmatic design, constitutes yet another negative antioxidant trial.
She pointed to three problems with this study and antioxidant trials in general. “First, the problem is with the antioxidant,” a xanthine oxidase inhibitor. “Xanthine oxidase is not a major trigger of oxidative stress. In a field of major players,” including nitric oxide, uncoupled endothelial nitric oxide synthase, and mitochondria myeloperoxidase, Dr. Cho said, “xanthine oxidase is a minor player.”
“Moreover, 57% of the patients stopped taking allopurinol, and rightfully so,” she said. Patients were receiving optimal medical therapies, many of which are also antioxidants, including statins, ACE inhibitors, angiotensin receptor blockers, and beta-blockers.
Second, the patient population was older, with an average age of 72 years. “This makes the ALL-HEART study a chronic angina study, chronic CAD study, one of the oldest modern day CAD trials. If you look at LoDoCo or ISCHEMIA trials, the average age is 63.” Patients also had established disease, many with previous revascularization.
The final issue seen with this trial, and all antioxidant trials, is that patient selection is not based on oxidative stress or antioxidant level. “The antioxidant trials have been disappointing at best. There is clear and convincing evidence that oxidative stress is involved in the pathogenesis of atherosclerosis, and yet study after study of antioxidant trials have been negative,” she said.
“Currently, there is no reliable measurement of global level of oxidative stress,” Dr. Cho noted. “Moreover, dose response was not tested, and if we cannot test the baseline antioxidant stress level of patients, we also cannot measure the effect of treatment on the global oxidative stress.”
So, “is there no hope for antioxidant trials?” she asked. Three factors will be required for future success, she said. “No. 1, selecting the right patient at the right time. No. 2, a reliable biomarker to measure oxidative stress to guide who should get therapy, and if the therapy is working. And lastly, targeted therapies that work on major triggers of oxidative stress.”
Also commenting on the results, B. Hadley Wilson, MD, executive vice chair of the Sanger Heart & Vascular Institute/Atrium Health, clinical professor of medicine at the University of North Carolina at Chapel Hill, and vice president of the American College of Cardiology, called ALL-HEART “an important and interesting study.”
“For years, cardiologists and others have been interested in allopurinol as an anti-inflammatory, xanthine oxidase inhibitor ... to prevent coronary ischemic events,” he said in an interview.
But this was a well-designed, well-conducted study, and “unfortunately there was no improvement in the primary outcome, no reduction in major cardiovascular events like myocardial infarction or stroke or cardiovascular death,” Dr. Wilson said. “So, it’s a bit of a disappointment that it’s not there as an important medication to help us with these patients with ischemic heart disease, but it’s also an important question answered — that we need to look at treatments for ischemic heart disease other than allopurinol.”
The trial was supported by the National Institute for Health and Care Research Health Technology Assessment Program in the United Kingdom. Dr. Mackenzie reported research contracts to her institution from NIHR HTA for this work, and other disclosures related to other work. Dr. Cho and Dr. Wilson reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
Allopurinol, a drug commonly used to treat gout, provided no benefit in terms of reducing cardiovascular (CV) events in patients with ischemic heart disease, new randomized trial results show.
Treatment of these patients without gout with 600 mg of allopurinol daily had no effect on composite primary endpoint outcomes, including nonfatal MI, nonfatal stroke, or CV death.
“ALL-HEART is the first large, prospective, randomized trial of the effect of allopurinol on major cardiovascular outcomes in patients with ischemic heart disease and provides robust evidence on the role of allopurinol in these patients,” principal investigator Isla Shelagh Mackenzie, MBChB (Honors), PhD, University of Dundee (Scotland), concluded at a press conference.
Their results suggest allopurinol should not be recommended for secondary prevention of events in this group, Dr. Mackenzie said. Although it remains an important treatment for gout, she added, “other avenues for treatment of ischemic heart disease should be explored in future.”
Results of the ALL-HEART (Allopurinol and Cardiovascular Outcomes in Ischemic Heart Disease) trial were presented at the annual congress of the European Society of Cardiology.
Gout treatment
Allopurinol is a xanthine oxidase inhibitor and acts by reducing serum uric acid levels and oxidative stress. Treatment is generally well tolerated, Dr. Mackenzie noted in her presentation, but some patients develop a rash, which can in some cases be serious or even fatal, progressing to Stevens-Johnson syndrome or toxic epidermal necrolysis, “particularly in certain ethnicities.” If rash develops, the advice is to stop treatment immediately.
“The importance of serum uric acid levels in cardiovascular disease is controversial, and there have been different reports over the years of how important they may be,” Dr. Mackenzie explained.
Observational studies have shown variable results, whereas intervention trials, most with fewer than 100 participants, have suggested potential improvements in factors such as blood pressure, endothelial function, left ventricular hypertrophy, or carotid intima-media thickness. Some have reported benefits in acute coronary syndrome and coronary artery bypass grafting, but others have not, she said. A previous study by their own group suggested an improvement in chest pain and exercise time in patients with chronic stable angina and documented coronary artery disease (CAD).
“So, until now, there have been no large prospective randomized trials of the effects of allopurinol on major cardiovascular outcomes in patients with ischemic heart disease,” Dr. Mackenzie said, and this was the aim of ALL-HEART.
ALL-HEART was a prospective, randomized, open-label, blinded-endpoint, multicenter trial. Patients with ischemia heart disease but no history of gout were recruited from 424 general practices across the United Kingdom, starting in February 2014 and with follow-up ending in September 2021. Participants were randomly assigned 1:1 to receive 600 mg of allopurinol daily or usual care.
“It was a decentralized trial, so the follow-up was largely remote after the first 6 weeks, and that included using record linkage data collected from centralized NHS [National Health Service] databases for hospitalizations and deaths in Scotland and England,” she said. The average follow-up was 4.8 years.
During that time, 258 (9.0%) participants in the allopurinol group and 76 (2.6%) in usual care withdrew from follow-up. By the end of the trial, 57.4% of patients in the allopurinol arm withdrew from randomized treatment.
Mean serum uric acid levels dropped from 0.34 mmol/L at baseline to 0.18 mmol/L at 6 weeks of treatment, “so we can see that the treatment was effective at lowering uric acid,” she noted.
In total, there were 5,721 patients in the final intention-to-treat analysis, and 639 patients had a first primary event.
For the primary outcome of nonfatal MI, nonfatal stroke, and cardiovascular death, there was no difference between the groups, the researchers reported, with a hazard ratio of 1.04 (95% confidence interval, 0.89-1.21; P = .65). Similarly, in secondary analyses, there were no differences in any of the component endpoints making up the primary outcome (nonfatal MI: HR, 0.97; 95% CI, 0.78-1.21; P = .81; nonfatal stroke: HR, 1.20; 95% CI, 0.89-1.60; P = .23; cardiovascular death: HR, 1.10; 95% CI, 0.85-1.43; P = .48), or in all-cause mortality (HR, 1.02; 95% CI, 0.87-1.20; P = .77), between the two groups, Dr. Mackenzie noted, “so a definitively neutral trial all round.”
In addition, no differences were seen in prespecified subgroups, including age, sex, estimated glomerular filtration rate, or diabetes, MI, heart failure, peripheral arterial disease, stroke, and stroke or transient ischemic attack at baseline.
There were also no significant effects on quality of life outcomes. Cost-effectiveness analyses are ongoing, although no differences are expected there, Dr. Mackenzie noted.
In terms of safety, incident cancers and all-cause mortality did not differ between groups. Serious adverse events were also similar between groups, Dr. Mackenzie said, “and there were no fatal treatment-related SAEs [serious adverse events] in the study.”
Another negative antioxidant trial
Invited discussant for the presentation, Leslie Cho, MD, of the Cleveland Clinic said that ALL-HEART, while an excellent trial with a pragmatic design, constitutes yet another negative antioxidant trial.
She pointed to three problems with this study and antioxidant trials in general. “First, the problem is with the antioxidant,” a xanthine oxidase inhibitor. “Xanthine oxidase is not a major trigger of oxidative stress. In a field of major players,” including nitric oxide, uncoupled endothelial nitric oxide synthase, and mitochondria myeloperoxidase, Dr. Cho said, “xanthine oxidase is a minor player.”
“Moreover, 57% of the patients stopped taking allopurinol, and rightfully so,” she said. Patients were receiving optimal medical therapies, many of which are also antioxidants, including statins, ACE inhibitors, angiotensin receptor blockers, and beta-blockers.
Second, the patient population was older, with an average age of 72 years. “This makes the ALL-HEART study a chronic angina study, chronic CAD study, one of the oldest modern day CAD trials. If you look at LoDoCo or ISCHEMIA trials, the average age is 63.” Patients also had established disease, many with previous revascularization.
The final issue seen with this trial, and all antioxidant trials, is that patient selection is not based on oxidative stress or antioxidant level. “The antioxidant trials have been disappointing at best. There is clear and convincing evidence that oxidative stress is involved in the pathogenesis of atherosclerosis, and yet study after study of antioxidant trials have been negative,” she said.
“Currently, there is no reliable measurement of global level of oxidative stress,” Dr. Cho noted. “Moreover, dose response was not tested, and if we cannot test the baseline antioxidant stress level of patients, we also cannot measure the effect of treatment on the global oxidative stress.”
So, “is there no hope for antioxidant trials?” she asked. Three factors will be required for future success, she said. “No. 1, selecting the right patient at the right time. No. 2, a reliable biomarker to measure oxidative stress to guide who should get therapy, and if the therapy is working. And lastly, targeted therapies that work on major triggers of oxidative stress.”
Also commenting on the results, B. Hadley Wilson, MD, executive vice chair of the Sanger Heart & Vascular Institute/Atrium Health, clinical professor of medicine at the University of North Carolina at Chapel Hill, and vice president of the American College of Cardiology, called ALL-HEART “an important and interesting study.”
“For years, cardiologists and others have been interested in allopurinol as an anti-inflammatory, xanthine oxidase inhibitor ... to prevent coronary ischemic events,” he said in an interview.
But this was a well-designed, well-conducted study, and “unfortunately there was no improvement in the primary outcome, no reduction in major cardiovascular events like myocardial infarction or stroke or cardiovascular death,” Dr. Wilson said. “So, it’s a bit of a disappointment that it’s not there as an important medication to help us with these patients with ischemic heart disease, but it’s also an important question answered — that we need to look at treatments for ischemic heart disease other than allopurinol.”
The trial was supported by the National Institute for Health and Care Research Health Technology Assessment Program in the United Kingdom. Dr. Mackenzie reported research contracts to her institution from NIHR HTA for this work, and other disclosures related to other work. Dr. Cho and Dr. Wilson reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM ESC CONGRESS 2022
Low urate limits for gout questioned in study
Lower limits on serum urate levels applied in gout management may be based on a misreading of data on mortality risks, researchers say.
Low urate levels may not in themselves pose a risk of death but may be a sign of some other illness, said Joshua F. Baker, MD, MSCE, associate professor of rheumatology and epidemiology at the University of Pennsylvania in Philadelphia.
“It points us towards being more reassured that we can be aggressive in treating gout without a concern about long-term effects for our patients,” he said in an interview. He and colleagues published their findings online in Arthritis & Rheumatology.
Previous research has linked high levels of urate with excessive fat and low levels of urate with loss of skeletal muscle mass. And epidemiologic studies have shown a U-shaped relationship between urate levels and mortality, suggesting that very high and very low levels of urate could be harmful.
Based on this correlation, and the theory that urate could have antioxidant benefits, some professional societies have recommended not lowering urate levels below a defined threshold when treating gout. The European Alliance of Associations for Rheumatology has recommended a lower limit of 3 mg/dL.
But the evidence doesn’t entirely support this caution. For example, in a clinical trial of pegloticase (Krystexxa) in patients with refractory gout, patients whose mean serum urate dropped below 2 mg/dL did not die in higher proportions than patients with higher urate levels.
To better understand the risk of low urate, Dr. Baker and colleagues analyzed data on 13,979 participants in the National Health and Nutrition Examination Survey (NHANES) during 1999-2006. The dataset included whole-body dual-energy x-ray absorptiometry (DXA) body composition measures as well as urate levels.
The researchers argue this measurement reveals more about a person’s overall health than body mass index (BMI), which doesn’t distinguish between mass from fat and mass from muscle.
They defined low lean body mass, or sarcopenia, as an appendicular lean mass index relative to fat mass index z score of –1. And they defined low urate as less than 2.5 mg/dL in women and less than 3.5 mg/dL in men.
They found that 29% of people with low urate had low lean body mass, compared with 16% of people with normal urate levels. The difference was statistically significant (P = .001).
They found an association between low urate and increased mortality (hazard ratio, 1.61; 95% confidence interval, 1.14-2.28; P = .008). But that association lost its statistical significance when the researchers adjusted for body composition and weight loss (HR, 1.30; 95% CI, 0.92-1.85; P = .13).
Dr. Baker thinks the association between elevated mortality and low urate can be explained by conditions such as cancer or lung inflammation that might on one hand increase the risk of death and on the other hand lower urate levels by lowering muscle mass. “Low uric acid levels are observed in people who have lost weight for unhealthy reasons, and that can explain relationships with long-term outcomes,” he said.
Proportions of muscle and fat could not account for the risk of mortality associated with high levels of urate, the researchers found. Those participants with urate levels above 5.7 mg/dL had a higher risk of death with higher levels of urate, and this persisted even after statistical adjustment for body composition.
The study sheds light on an important area of controversy, said Mehdi Fini, MD, of the department of medicine at the University of Colorado at Denver, Aurora, who was not involved in the research.
But body composition does not entirely explain the relationship between urate and mortality, he told this news organization. Medications used to lower urate can cause side effects that might increase mortality, he said.
Also, he said, it’s important to understand the role of comorbidities. He cited evidence that low urate is associated with renal, cardiovascular, and pulmonary conditions. Safe levels of urate might differ depending on these factors. So rather than applying the same target serum level to all patients, perhaps researchers should investigate whether lowering urate by a percentage of the patient’s current level is safer and more effective, he suggested.
He agreed with an editorial that also appeared in Arthritis & Rheumatology saying that there is no evidence for a benefit in lowering urate much below 5 mg/dL. “No matter what, I think we should just be careful,” Dr. Fini said.
Dr. Fini and Dr. Baker report no relevant financial relationships. Dr. Baker acknowledged support from a VA Clinical Science Research & Development Merit Award and a Rehabilitation R&D Merit Award.
A version of this article first appeared on Medscape.com.
Lower limits on serum urate levels applied in gout management may be based on a misreading of data on mortality risks, researchers say.
Low urate levels may not in themselves pose a risk of death but may be a sign of some other illness, said Joshua F. Baker, MD, MSCE, associate professor of rheumatology and epidemiology at the University of Pennsylvania in Philadelphia.
“It points us towards being more reassured that we can be aggressive in treating gout without a concern about long-term effects for our patients,” he said in an interview. He and colleagues published their findings online in Arthritis & Rheumatology.
Previous research has linked high levels of urate with excessive fat and low levels of urate with loss of skeletal muscle mass. And epidemiologic studies have shown a U-shaped relationship between urate levels and mortality, suggesting that very high and very low levels of urate could be harmful.
Based on this correlation, and the theory that urate could have antioxidant benefits, some professional societies have recommended not lowering urate levels below a defined threshold when treating gout. The European Alliance of Associations for Rheumatology has recommended a lower limit of 3 mg/dL.
But the evidence doesn’t entirely support this caution. For example, in a clinical trial of pegloticase (Krystexxa) in patients with refractory gout, patients whose mean serum urate dropped below 2 mg/dL did not die in higher proportions than patients with higher urate levels.
To better understand the risk of low urate, Dr. Baker and colleagues analyzed data on 13,979 participants in the National Health and Nutrition Examination Survey (NHANES) during 1999-2006. The dataset included whole-body dual-energy x-ray absorptiometry (DXA) body composition measures as well as urate levels.
The researchers argue this measurement reveals more about a person’s overall health than body mass index (BMI), which doesn’t distinguish between mass from fat and mass from muscle.
They defined low lean body mass, or sarcopenia, as an appendicular lean mass index relative to fat mass index z score of –1. And they defined low urate as less than 2.5 mg/dL in women and less than 3.5 mg/dL in men.
They found that 29% of people with low urate had low lean body mass, compared with 16% of people with normal urate levels. The difference was statistically significant (P = .001).
They found an association between low urate and increased mortality (hazard ratio, 1.61; 95% confidence interval, 1.14-2.28; P = .008). But that association lost its statistical significance when the researchers adjusted for body composition and weight loss (HR, 1.30; 95% CI, 0.92-1.85; P = .13).
Dr. Baker thinks the association between elevated mortality and low urate can be explained by conditions such as cancer or lung inflammation that might on one hand increase the risk of death and on the other hand lower urate levels by lowering muscle mass. “Low uric acid levels are observed in people who have lost weight for unhealthy reasons, and that can explain relationships with long-term outcomes,” he said.
Proportions of muscle and fat could not account for the risk of mortality associated with high levels of urate, the researchers found. Those participants with urate levels above 5.7 mg/dL had a higher risk of death with higher levels of urate, and this persisted even after statistical adjustment for body composition.
The study sheds light on an important area of controversy, said Mehdi Fini, MD, of the department of medicine at the University of Colorado at Denver, Aurora, who was not involved in the research.
But body composition does not entirely explain the relationship between urate and mortality, he told this news organization. Medications used to lower urate can cause side effects that might increase mortality, he said.
Also, he said, it’s important to understand the role of comorbidities. He cited evidence that low urate is associated with renal, cardiovascular, and pulmonary conditions. Safe levels of urate might differ depending on these factors. So rather than applying the same target serum level to all patients, perhaps researchers should investigate whether lowering urate by a percentage of the patient’s current level is safer and more effective, he suggested.
He agreed with an editorial that also appeared in Arthritis & Rheumatology saying that there is no evidence for a benefit in lowering urate much below 5 mg/dL. “No matter what, I think we should just be careful,” Dr. Fini said.
Dr. Fini and Dr. Baker report no relevant financial relationships. Dr. Baker acknowledged support from a VA Clinical Science Research & Development Merit Award and a Rehabilitation R&D Merit Award.
A version of this article first appeared on Medscape.com.
Lower limits on serum urate levels applied in gout management may be based on a misreading of data on mortality risks, researchers say.
Low urate levels may not in themselves pose a risk of death but may be a sign of some other illness, said Joshua F. Baker, MD, MSCE, associate professor of rheumatology and epidemiology at the University of Pennsylvania in Philadelphia.
“It points us towards being more reassured that we can be aggressive in treating gout without a concern about long-term effects for our patients,” he said in an interview. He and colleagues published their findings online in Arthritis & Rheumatology.
Previous research has linked high levels of urate with excessive fat and low levels of urate with loss of skeletal muscle mass. And epidemiologic studies have shown a U-shaped relationship between urate levels and mortality, suggesting that very high and very low levels of urate could be harmful.
Based on this correlation, and the theory that urate could have antioxidant benefits, some professional societies have recommended not lowering urate levels below a defined threshold when treating gout. The European Alliance of Associations for Rheumatology has recommended a lower limit of 3 mg/dL.
But the evidence doesn’t entirely support this caution. For example, in a clinical trial of pegloticase (Krystexxa) in patients with refractory gout, patients whose mean serum urate dropped below 2 mg/dL did not die in higher proportions than patients with higher urate levels.
To better understand the risk of low urate, Dr. Baker and colleagues analyzed data on 13,979 participants in the National Health and Nutrition Examination Survey (NHANES) during 1999-2006. The dataset included whole-body dual-energy x-ray absorptiometry (DXA) body composition measures as well as urate levels.
The researchers argue this measurement reveals more about a person’s overall health than body mass index (BMI), which doesn’t distinguish between mass from fat and mass from muscle.
They defined low lean body mass, or sarcopenia, as an appendicular lean mass index relative to fat mass index z score of –1. And they defined low urate as less than 2.5 mg/dL in women and less than 3.5 mg/dL in men.
They found that 29% of people with low urate had low lean body mass, compared with 16% of people with normal urate levels. The difference was statistically significant (P = .001).
They found an association between low urate and increased mortality (hazard ratio, 1.61; 95% confidence interval, 1.14-2.28; P = .008). But that association lost its statistical significance when the researchers adjusted for body composition and weight loss (HR, 1.30; 95% CI, 0.92-1.85; P = .13).
Dr. Baker thinks the association between elevated mortality and low urate can be explained by conditions such as cancer or lung inflammation that might on one hand increase the risk of death and on the other hand lower urate levels by lowering muscle mass. “Low uric acid levels are observed in people who have lost weight for unhealthy reasons, and that can explain relationships with long-term outcomes,” he said.
Proportions of muscle and fat could not account for the risk of mortality associated with high levels of urate, the researchers found. Those participants with urate levels above 5.7 mg/dL had a higher risk of death with higher levels of urate, and this persisted even after statistical adjustment for body composition.
The study sheds light on an important area of controversy, said Mehdi Fini, MD, of the department of medicine at the University of Colorado at Denver, Aurora, who was not involved in the research.
But body composition does not entirely explain the relationship between urate and mortality, he told this news organization. Medications used to lower urate can cause side effects that might increase mortality, he said.
Also, he said, it’s important to understand the role of comorbidities. He cited evidence that low urate is associated with renal, cardiovascular, and pulmonary conditions. Safe levels of urate might differ depending on these factors. So rather than applying the same target serum level to all patients, perhaps researchers should investigate whether lowering urate by a percentage of the patient’s current level is safer and more effective, he suggested.
He agreed with an editorial that also appeared in Arthritis & Rheumatology saying that there is no evidence for a benefit in lowering urate much below 5 mg/dL. “No matter what, I think we should just be careful,” Dr. Fini said.
Dr. Fini and Dr. Baker report no relevant financial relationships. Dr. Baker acknowledged support from a VA Clinical Science Research & Development Merit Award and a Rehabilitation R&D Merit Award.
A version of this article first appeared on Medscape.com.
FROM ARTHRITIS & RHEUMATOLOGY
Ultrasound helps predict gout flares over the next year
Adding ultrasound (US) to the clinical exam helps predict the likelihood of future gout flares, results of a prospective, observational study conducted in Italy suggest.
“Baseline US findings indicative of MSU [monosodium urate] burden and US-detected inflammation are independent predictors of gout flares over 12 months,” lead author Edoardo Cipolletta, MD, of the rheumatology unit, department of clinical and molecular sciences at Marche Polytechnic University in Ancona, Italy, and colleagues wrote in Rheumatology.
“We demonstrated that US findings provided an additional value over clinical data in estimating the risk of flares. Moreover, we reported an association between US findings at a joint and the occurrence of gout flares at the same joint,” they added.
Predicting risk of flares and reducing their occurrence are two main challenges in managing gout, the authors wrote. US can be used to scan multiple joints and is widely used in Europe as a low-cost, radiation-free imaging tool that’s easily integrated into clinical practice.
To investigate whether US can predict gout flares, the researchers enrolled 81 consecutive adult patients with gout in the study between April 2019 and March 2021 at one academic rheumatology treatment site in Italy and followed them for 12 months. The authors compared cases (who developed at least one flare within 12 months of the baseline visit) with controls (who self-reported no gout flares over that period).
Patients diagnosed with other inflammatory arthritis and those with coexisting calcium pyrophosphate deposition disease were excluded from the study.
The 71 participants who completed the study were, on average, in their early 60s, and in both groups, all but one were male. At the baseline visit, all had been on stable urate-lowering therapy for at least 6 months and had not had any gout flares in 4 weeks. The mean gout duration was 7 years in the case group and 8 years in controls.
At baseline, all participants underwent physical examination and US of elbows, wrists, second metacarpophalangeal joints, knees, ankles, and first metatarsophalangeal joints by a member of the research team who was blinded to the clinical and laboratory data.
Clinical assessments were scheduled at baseline and at 6-month intervals, and all participants were evaluated by a second researcher who was blinded to US findings.
During follow-up visits, participants were asked to report any gout flare, considered to meet at least three of four criteria: patient-defined flare, pain at rest score higher than 3 on a 0-10 scale, at least one swollen joint, and at least one warm joint. Patients not reaching their target serum urate goal received escalated urate-lowering therapy dosage and anti-inflammatory prophylaxis.
The US indicators of MSU deposits – aggregates, double contour sign, and tophi – were recorded as present or absent. The power Doppler signal was scored from 0 through 4, and summated scores for each US finding were calculated.
Over 12 months, the researchers found:
- Thirty (42.3%) patients had at least one flare, with a median of 2.0 flares. Patients with flares had higher a US median total MSU score (5.0 vs. 2.0; P = .01) and power Doppler signal (3.0 vs. 0; P < .01) than controls.
- In multivariate analysis, baseline US scores indicating MSU deposits and US-detected inflammation were significantly linked with the occurrence of flares. The adjusted odds ratio for total MSU score was 1.75 (95% confidence interval, 1.26-2.43) and for power Doppler score was 1.63 (95% CI, 1.12-2.40).
- Also in a multivariate analysis, baseline US scores indicating MSU deposits and US-detected inflammation were significantly linked with the number of flares. The incidence risk ratio for total MSU score adjusted was 1.17 (95% CI, 1.08-1.26) and for power Doppler score was 1.29 (95% CI, 1.19-1.40).
Four rheumatologists welcome findings
Gout remains the most common cause of inflammatory arthritis and a significant reason for hospital visits, noted Narender Annapureddy, MD, associate professor of medicine at Vanderbilt University Medical Center in Nashville, Tenn..
“The study adds to the growing utility of musculoskeletal ultrasound in rheumatology practices to treat various diseases,” he said. “Data that could provide risk prediction for gout flares would be associated with significant benefits in terms of reducing ED visits, hospital admission, and lost work productivity.”
One study limitation, Dr. Annapureddy mentioned, was the single experienced US reader, “which may limit generalizability of results at this time, at least in the United States.”
Yeohan Song, MD, an instructor at Ohio State University Wexner Medical Center, Columbus, integrates US into his practice.
“In gout management, musculoskeletal ultrasound is a useful adjunct to the clinical exam and laboratory markers, particularly [in patients] with recurrent flares despite guideline-directed target serum urate levels,” he said.
Sara K. Tedeschi, MD, MPH, assistant professor of medicine at Harvard Medical School, Boston, pointed out that the US protocol in the study involved imaging knees, ankles, first metatarsophalangeal joints, elbows, wrists, and second metacarpophalangeal joints, and took around 30 minutes to complete.
“That would not be practical in the United States due to time constraints in most rheumatology clinics,” she said.
“The authors report that a ‘reduced scanning protocol’ of the bilateral knees, ankles, and first metatarsophalangeal joints demonstrated similar predictive ability as the full protocol,” she added, “although scanning six joints still might not be feasible during a typical return patient clinic visit in the United States.”
Philip Chu, MD, clinical associate at Duke University, Durham, N.C., uses diagnostic US to help differentiate borderline gout cases from other arthropathies.
“A baseline scan, a follow-up scan before deciding to stop prophylaxis, or a follow-up scan in the setting of recurrent gout flares despite reaching goal serum uric acid, may be cost-effective time points to perform diagnostic US,” he advised.
“Unfortunately,” he added, “reimbursement for diagnostic US has been decreasing over the years, which makes it challenging to increase diagnostic US to the [frequency of its use] in Europe.”
Asked how most gout care being provided by primary care doctors in the United States affects gout management, Dr. Chu said: “Depending on which guidelines one follows for treating gout – from the American College of Rheumatology or the American College of Physicians – one may be more or less likely to start urate-lowering therapy after the first gout flare.”
“Understanding MSU burden in each patient, or even seeing active inflammation at these sites by increased Doppler signal, may change the threshold for physicians to initiate therapy,” he added.
The study received no funding. Three study authors reported financial involvements with pharmaceutical companies. Dr. Cipolletta, Dr. Annapureddy, Dr. Song, Dr. Tedeschi, and Dr. Chu reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Adding ultrasound (US) to the clinical exam helps predict the likelihood of future gout flares, results of a prospective, observational study conducted in Italy suggest.
“Baseline US findings indicative of MSU [monosodium urate] burden and US-detected inflammation are independent predictors of gout flares over 12 months,” lead author Edoardo Cipolletta, MD, of the rheumatology unit, department of clinical and molecular sciences at Marche Polytechnic University in Ancona, Italy, and colleagues wrote in Rheumatology.
“We demonstrated that US findings provided an additional value over clinical data in estimating the risk of flares. Moreover, we reported an association between US findings at a joint and the occurrence of gout flares at the same joint,” they added.
Predicting risk of flares and reducing their occurrence are two main challenges in managing gout, the authors wrote. US can be used to scan multiple joints and is widely used in Europe as a low-cost, radiation-free imaging tool that’s easily integrated into clinical practice.
To investigate whether US can predict gout flares, the researchers enrolled 81 consecutive adult patients with gout in the study between April 2019 and March 2021 at one academic rheumatology treatment site in Italy and followed them for 12 months. The authors compared cases (who developed at least one flare within 12 months of the baseline visit) with controls (who self-reported no gout flares over that period).
Patients diagnosed with other inflammatory arthritis and those with coexisting calcium pyrophosphate deposition disease were excluded from the study.
The 71 participants who completed the study were, on average, in their early 60s, and in both groups, all but one were male. At the baseline visit, all had been on stable urate-lowering therapy for at least 6 months and had not had any gout flares in 4 weeks. The mean gout duration was 7 years in the case group and 8 years in controls.
At baseline, all participants underwent physical examination and US of elbows, wrists, second metacarpophalangeal joints, knees, ankles, and first metatarsophalangeal joints by a member of the research team who was blinded to the clinical and laboratory data.
Clinical assessments were scheduled at baseline and at 6-month intervals, and all participants were evaluated by a second researcher who was blinded to US findings.
During follow-up visits, participants were asked to report any gout flare, considered to meet at least three of four criteria: patient-defined flare, pain at rest score higher than 3 on a 0-10 scale, at least one swollen joint, and at least one warm joint. Patients not reaching their target serum urate goal received escalated urate-lowering therapy dosage and anti-inflammatory prophylaxis.
The US indicators of MSU deposits – aggregates, double contour sign, and tophi – were recorded as present or absent. The power Doppler signal was scored from 0 through 4, and summated scores for each US finding were calculated.
Over 12 months, the researchers found:
- Thirty (42.3%) patients had at least one flare, with a median of 2.0 flares. Patients with flares had higher a US median total MSU score (5.0 vs. 2.0; P = .01) and power Doppler signal (3.0 vs. 0; P < .01) than controls.
- In multivariate analysis, baseline US scores indicating MSU deposits and US-detected inflammation were significantly linked with the occurrence of flares. The adjusted odds ratio for total MSU score was 1.75 (95% confidence interval, 1.26-2.43) and for power Doppler score was 1.63 (95% CI, 1.12-2.40).
- Also in a multivariate analysis, baseline US scores indicating MSU deposits and US-detected inflammation were significantly linked with the number of flares. The incidence risk ratio for total MSU score adjusted was 1.17 (95% CI, 1.08-1.26) and for power Doppler score was 1.29 (95% CI, 1.19-1.40).
Four rheumatologists welcome findings
Gout remains the most common cause of inflammatory arthritis and a significant reason for hospital visits, noted Narender Annapureddy, MD, associate professor of medicine at Vanderbilt University Medical Center in Nashville, Tenn..
“The study adds to the growing utility of musculoskeletal ultrasound in rheumatology practices to treat various diseases,” he said. “Data that could provide risk prediction for gout flares would be associated with significant benefits in terms of reducing ED visits, hospital admission, and lost work productivity.”
One study limitation, Dr. Annapureddy mentioned, was the single experienced US reader, “which may limit generalizability of results at this time, at least in the United States.”
Yeohan Song, MD, an instructor at Ohio State University Wexner Medical Center, Columbus, integrates US into his practice.
“In gout management, musculoskeletal ultrasound is a useful adjunct to the clinical exam and laboratory markers, particularly [in patients] with recurrent flares despite guideline-directed target serum urate levels,” he said.
Sara K. Tedeschi, MD, MPH, assistant professor of medicine at Harvard Medical School, Boston, pointed out that the US protocol in the study involved imaging knees, ankles, first metatarsophalangeal joints, elbows, wrists, and second metacarpophalangeal joints, and took around 30 minutes to complete.
“That would not be practical in the United States due to time constraints in most rheumatology clinics,” she said.
“The authors report that a ‘reduced scanning protocol’ of the bilateral knees, ankles, and first metatarsophalangeal joints demonstrated similar predictive ability as the full protocol,” she added, “although scanning six joints still might not be feasible during a typical return patient clinic visit in the United States.”
Philip Chu, MD, clinical associate at Duke University, Durham, N.C., uses diagnostic US to help differentiate borderline gout cases from other arthropathies.
“A baseline scan, a follow-up scan before deciding to stop prophylaxis, or a follow-up scan in the setting of recurrent gout flares despite reaching goal serum uric acid, may be cost-effective time points to perform diagnostic US,” he advised.
“Unfortunately,” he added, “reimbursement for diagnostic US has been decreasing over the years, which makes it challenging to increase diagnostic US to the [frequency of its use] in Europe.”
Asked how most gout care being provided by primary care doctors in the United States affects gout management, Dr. Chu said: “Depending on which guidelines one follows for treating gout – from the American College of Rheumatology or the American College of Physicians – one may be more or less likely to start urate-lowering therapy after the first gout flare.”
“Understanding MSU burden in each patient, or even seeing active inflammation at these sites by increased Doppler signal, may change the threshold for physicians to initiate therapy,” he added.
The study received no funding. Three study authors reported financial involvements with pharmaceutical companies. Dr. Cipolletta, Dr. Annapureddy, Dr. Song, Dr. Tedeschi, and Dr. Chu reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Adding ultrasound (US) to the clinical exam helps predict the likelihood of future gout flares, results of a prospective, observational study conducted in Italy suggest.
“Baseline US findings indicative of MSU [monosodium urate] burden and US-detected inflammation are independent predictors of gout flares over 12 months,” lead author Edoardo Cipolletta, MD, of the rheumatology unit, department of clinical and molecular sciences at Marche Polytechnic University in Ancona, Italy, and colleagues wrote in Rheumatology.
“We demonstrated that US findings provided an additional value over clinical data in estimating the risk of flares. Moreover, we reported an association between US findings at a joint and the occurrence of gout flares at the same joint,” they added.
Predicting risk of flares and reducing their occurrence are two main challenges in managing gout, the authors wrote. US can be used to scan multiple joints and is widely used in Europe as a low-cost, radiation-free imaging tool that’s easily integrated into clinical practice.
To investigate whether US can predict gout flares, the researchers enrolled 81 consecutive adult patients with gout in the study between April 2019 and March 2021 at one academic rheumatology treatment site in Italy and followed them for 12 months. The authors compared cases (who developed at least one flare within 12 months of the baseline visit) with controls (who self-reported no gout flares over that period).
Patients diagnosed with other inflammatory arthritis and those with coexisting calcium pyrophosphate deposition disease were excluded from the study.
The 71 participants who completed the study were, on average, in their early 60s, and in both groups, all but one were male. At the baseline visit, all had been on stable urate-lowering therapy for at least 6 months and had not had any gout flares in 4 weeks. The mean gout duration was 7 years in the case group and 8 years in controls.
At baseline, all participants underwent physical examination and US of elbows, wrists, second metacarpophalangeal joints, knees, ankles, and first metatarsophalangeal joints by a member of the research team who was blinded to the clinical and laboratory data.
Clinical assessments were scheduled at baseline and at 6-month intervals, and all participants were evaluated by a second researcher who was blinded to US findings.
During follow-up visits, participants were asked to report any gout flare, considered to meet at least three of four criteria: patient-defined flare, pain at rest score higher than 3 on a 0-10 scale, at least one swollen joint, and at least one warm joint. Patients not reaching their target serum urate goal received escalated urate-lowering therapy dosage and anti-inflammatory prophylaxis.
The US indicators of MSU deposits – aggregates, double contour sign, and tophi – were recorded as present or absent. The power Doppler signal was scored from 0 through 4, and summated scores for each US finding were calculated.
Over 12 months, the researchers found:
- Thirty (42.3%) patients had at least one flare, with a median of 2.0 flares. Patients with flares had higher a US median total MSU score (5.0 vs. 2.0; P = .01) and power Doppler signal (3.0 vs. 0; P < .01) than controls.
- In multivariate analysis, baseline US scores indicating MSU deposits and US-detected inflammation were significantly linked with the occurrence of flares. The adjusted odds ratio for total MSU score was 1.75 (95% confidence interval, 1.26-2.43) and for power Doppler score was 1.63 (95% CI, 1.12-2.40).
- Also in a multivariate analysis, baseline US scores indicating MSU deposits and US-detected inflammation were significantly linked with the number of flares. The incidence risk ratio for total MSU score adjusted was 1.17 (95% CI, 1.08-1.26) and for power Doppler score was 1.29 (95% CI, 1.19-1.40).
Four rheumatologists welcome findings
Gout remains the most common cause of inflammatory arthritis and a significant reason for hospital visits, noted Narender Annapureddy, MD, associate professor of medicine at Vanderbilt University Medical Center in Nashville, Tenn..
“The study adds to the growing utility of musculoskeletal ultrasound in rheumatology practices to treat various diseases,” he said. “Data that could provide risk prediction for gout flares would be associated with significant benefits in terms of reducing ED visits, hospital admission, and lost work productivity.”
One study limitation, Dr. Annapureddy mentioned, was the single experienced US reader, “which may limit generalizability of results at this time, at least in the United States.”
Yeohan Song, MD, an instructor at Ohio State University Wexner Medical Center, Columbus, integrates US into his practice.
“In gout management, musculoskeletal ultrasound is a useful adjunct to the clinical exam and laboratory markers, particularly [in patients] with recurrent flares despite guideline-directed target serum urate levels,” he said.
Sara K. Tedeschi, MD, MPH, assistant professor of medicine at Harvard Medical School, Boston, pointed out that the US protocol in the study involved imaging knees, ankles, first metatarsophalangeal joints, elbows, wrists, and second metacarpophalangeal joints, and took around 30 minutes to complete.
“That would not be practical in the United States due to time constraints in most rheumatology clinics,” she said.
“The authors report that a ‘reduced scanning protocol’ of the bilateral knees, ankles, and first metatarsophalangeal joints demonstrated similar predictive ability as the full protocol,” she added, “although scanning six joints still might not be feasible during a typical return patient clinic visit in the United States.”
Philip Chu, MD, clinical associate at Duke University, Durham, N.C., uses diagnostic US to help differentiate borderline gout cases from other arthropathies.
“A baseline scan, a follow-up scan before deciding to stop prophylaxis, or a follow-up scan in the setting of recurrent gout flares despite reaching goal serum uric acid, may be cost-effective time points to perform diagnostic US,” he advised.
“Unfortunately,” he added, “reimbursement for diagnostic US has been decreasing over the years, which makes it challenging to increase diagnostic US to the [frequency of its use] in Europe.”
Asked how most gout care being provided by primary care doctors in the United States affects gout management, Dr. Chu said: “Depending on which guidelines one follows for treating gout – from the American College of Rheumatology or the American College of Physicians – one may be more or less likely to start urate-lowering therapy after the first gout flare.”
“Understanding MSU burden in each patient, or even seeing active inflammation at these sites by increased Doppler signal, may change the threshold for physicians to initiate therapy,” he added.
The study received no funding. Three study authors reported financial involvements with pharmaceutical companies. Dr. Cipolletta, Dr. Annapureddy, Dr. Song, Dr. Tedeschi, and Dr. Chu reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM RHEUMATOLOGY
Black Americans’ high gout rate stems from social causes
Gout prevalence is more common in Black Americans than White Americans, and the disparity in prevalence is attributable to social determinants of health, according to a recently published article in JAMA Network Open.
“There has been evidence from recent cohort studies in the U.S. that was suggesting that the prevalence and incidence [of gout] was growing among non-White populations,” said Natalie McCormick, PhD, the study’s lead author and postdoctoral research fellow in medicine in the division of rheumatology, allergy, and immunology at Massachusetts General Hospital and Harvard Medical School, both in Boston. “We wanted to do this at the general population level to see how generalizable [that evidence] is.”
Alvin Wells, MD, PhD, director of the department of rheumatology at Advocate Aurora Medical Group, Franklin, Wisc., noted the findings highlight inequities in care for patients with gout that could be improved with greater emphasis on educating patients about their condition.
“I think that what this shows is that in the U.S. ... there still are some disparities in treating gout,” said Dr. Wells, who was not involved with the study. “And that we have ways to mitigate that, with not only aggressive therapy, but also with other tools like counseling patients. At the end of the day, people all want to be educated about the disease.”
Greater prevalence disappears with adjustment for socioclinical factors
The cross-sectional analysis involved data from U.S. adult participants in the National Health and Nutrition Examination Survey (NHANES) from 2007 to 2016 who self-reported Black or White race.
Investigators considered factors such as excess body mass index (BMI), chronic kidney disease (defined as estimated glomerular filtration rate less than 60 mL/min per 1.73 m2), poverty, poor-quality diet, lower educational level, alcohol consumption, and diuretic use in their analysis.
Dr. McCormick and coinvestigators included a total of 18,693 participants, consisting of 3,304 Black women, 6,195 White women, 3,085 Black men, and 6,109 White men.
They determined that the age-standardized prevalence of gout was 3.5% (95% confidence interval, 2.7%-4.3%) in Black women and 2.0% (95% CI, 1.5% - 2.5%) in White women (age-adjusted odds ratio, 1.81; 95% CI, 1.29-2.53). They calculated that the prevalence was 7.0% (95% CI, 6.2%-7.9%) in Black men and 5.4% (95% CI, 4.7%-6.2%) in White men (age-adjusted OR, 1.26; 95% CI, 1.02-1.55). They found similar differences in the prevalence of hyperuricemia between Black and White Americans.
The increased prevalence of gout in Black Americans, compared with White Americans, does not arise from genetics, according to McCormick. “Our conclusion was that it was due to social determinants of health,” she said. “When we adjusted for all socioclinical risk factors, the racial differences in gout and hyperuricemia prevalence disappeared. Importantly, stepwise regression analysis showed the two biggest drivers of the racial difference in gout prevalence among women were poverty itself, and excess BMI, which can be influenced by poverty.”
Dr. McCormick pointed out that in contrast to the current data, there was no racial difference in the prevalence of gout approximately 2 decades earlier, looking at data from the 1988-1994 NHANES III.
Given the findings, which included the fact that significantly more Black women and men were currently taking diuretics, compared with their White counterparts, Dr. McCormick pointed out clinicians should give more thought to medical therapies prescribed for conditions like high blood pressure to patients with gout or at risk for gout.
“One thing we found was that diuretic use was a driver” of gout, Dr. McCormick said. A prescriber “may want to consider different therapies that present a lower risk of gout if someone has hypertension. There could be greater consideration for prescribing alternatives to diuretics.”
More patient education and rheumatology referrals needed
An impediment to providing that education to patients with gout is unconscious bias on the part of the primary care provider, Dr. Wells said.
“It is about what your perspectives are and what you bring to the table,” he explained. “If you saw [a patient] who looked like someone in your family, that person will be treated differently [than someone who does not look like a family member]. That is where the whole concept [of unconscious bias] comes in.”
Primary care providers need to adopt a holistic approach to gout management that involves counseling about good nutrition, smoking cessation, regular exercise, and limiting alcohol consumption, in addition to medication adherence. Primary care providers may have a bias in treating their Black patients, failing to devote sufficient time and attention to assist them in getting their disease under control, he said.
“Gout should be just like any other chronic disease,” Dr. Wells said. “You need to have a target in mind, and you and your patient need to work together to get to that target. When [patients] end up in rheumatology offices, it is almost too late. I think the take-home message here is that in 2022 ... for any patient who has gout, that patient probably needs to be seen by a rheumatologist because, indeed, with aggressive therapy, preventive therapy, [and] education, and if they are on the right medications, they won’t end up with these crippling joints that we see all the time.”
Dr. McCormick and Dr. Wells disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Gout prevalence is more common in Black Americans than White Americans, and the disparity in prevalence is attributable to social determinants of health, according to a recently published article in JAMA Network Open.
“There has been evidence from recent cohort studies in the U.S. that was suggesting that the prevalence and incidence [of gout] was growing among non-White populations,” said Natalie McCormick, PhD, the study’s lead author and postdoctoral research fellow in medicine in the division of rheumatology, allergy, and immunology at Massachusetts General Hospital and Harvard Medical School, both in Boston. “We wanted to do this at the general population level to see how generalizable [that evidence] is.”
Alvin Wells, MD, PhD, director of the department of rheumatology at Advocate Aurora Medical Group, Franklin, Wisc., noted the findings highlight inequities in care for patients with gout that could be improved with greater emphasis on educating patients about their condition.
“I think that what this shows is that in the U.S. ... there still are some disparities in treating gout,” said Dr. Wells, who was not involved with the study. “And that we have ways to mitigate that, with not only aggressive therapy, but also with other tools like counseling patients. At the end of the day, people all want to be educated about the disease.”
Greater prevalence disappears with adjustment for socioclinical factors
The cross-sectional analysis involved data from U.S. adult participants in the National Health and Nutrition Examination Survey (NHANES) from 2007 to 2016 who self-reported Black or White race.
Investigators considered factors such as excess body mass index (BMI), chronic kidney disease (defined as estimated glomerular filtration rate less than 60 mL/min per 1.73 m2), poverty, poor-quality diet, lower educational level, alcohol consumption, and diuretic use in their analysis.
Dr. McCormick and coinvestigators included a total of 18,693 participants, consisting of 3,304 Black women, 6,195 White women, 3,085 Black men, and 6,109 White men.
They determined that the age-standardized prevalence of gout was 3.5% (95% confidence interval, 2.7%-4.3%) in Black women and 2.0% (95% CI, 1.5% - 2.5%) in White women (age-adjusted odds ratio, 1.81; 95% CI, 1.29-2.53). They calculated that the prevalence was 7.0% (95% CI, 6.2%-7.9%) in Black men and 5.4% (95% CI, 4.7%-6.2%) in White men (age-adjusted OR, 1.26; 95% CI, 1.02-1.55). They found similar differences in the prevalence of hyperuricemia between Black and White Americans.
The increased prevalence of gout in Black Americans, compared with White Americans, does not arise from genetics, according to McCormick. “Our conclusion was that it was due to social determinants of health,” she said. “When we adjusted for all socioclinical risk factors, the racial differences in gout and hyperuricemia prevalence disappeared. Importantly, stepwise regression analysis showed the two biggest drivers of the racial difference in gout prevalence among women were poverty itself, and excess BMI, which can be influenced by poverty.”
Dr. McCormick pointed out that in contrast to the current data, there was no racial difference in the prevalence of gout approximately 2 decades earlier, looking at data from the 1988-1994 NHANES III.
Given the findings, which included the fact that significantly more Black women and men were currently taking diuretics, compared with their White counterparts, Dr. McCormick pointed out clinicians should give more thought to medical therapies prescribed for conditions like high blood pressure to patients with gout or at risk for gout.
“One thing we found was that diuretic use was a driver” of gout, Dr. McCormick said. A prescriber “may want to consider different therapies that present a lower risk of gout if someone has hypertension. There could be greater consideration for prescribing alternatives to diuretics.”
More patient education and rheumatology referrals needed
An impediment to providing that education to patients with gout is unconscious bias on the part of the primary care provider, Dr. Wells said.
“It is about what your perspectives are and what you bring to the table,” he explained. “If you saw [a patient] who looked like someone in your family, that person will be treated differently [than someone who does not look like a family member]. That is where the whole concept [of unconscious bias] comes in.”
Primary care providers need to adopt a holistic approach to gout management that involves counseling about good nutrition, smoking cessation, regular exercise, and limiting alcohol consumption, in addition to medication adherence. Primary care providers may have a bias in treating their Black patients, failing to devote sufficient time and attention to assist them in getting their disease under control, he said.
“Gout should be just like any other chronic disease,” Dr. Wells said. “You need to have a target in mind, and you and your patient need to work together to get to that target. When [patients] end up in rheumatology offices, it is almost too late. I think the take-home message here is that in 2022 ... for any patient who has gout, that patient probably needs to be seen by a rheumatologist because, indeed, with aggressive therapy, preventive therapy, [and] education, and if they are on the right medications, they won’t end up with these crippling joints that we see all the time.”
Dr. McCormick and Dr. Wells disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Gout prevalence is more common in Black Americans than White Americans, and the disparity in prevalence is attributable to social determinants of health, according to a recently published article in JAMA Network Open.
“There has been evidence from recent cohort studies in the U.S. that was suggesting that the prevalence and incidence [of gout] was growing among non-White populations,” said Natalie McCormick, PhD, the study’s lead author and postdoctoral research fellow in medicine in the division of rheumatology, allergy, and immunology at Massachusetts General Hospital and Harvard Medical School, both in Boston. “We wanted to do this at the general population level to see how generalizable [that evidence] is.”
Alvin Wells, MD, PhD, director of the department of rheumatology at Advocate Aurora Medical Group, Franklin, Wisc., noted the findings highlight inequities in care for patients with gout that could be improved with greater emphasis on educating patients about their condition.
“I think that what this shows is that in the U.S. ... there still are some disparities in treating gout,” said Dr. Wells, who was not involved with the study. “And that we have ways to mitigate that, with not only aggressive therapy, but also with other tools like counseling patients. At the end of the day, people all want to be educated about the disease.”
Greater prevalence disappears with adjustment for socioclinical factors
The cross-sectional analysis involved data from U.S. adult participants in the National Health and Nutrition Examination Survey (NHANES) from 2007 to 2016 who self-reported Black or White race.
Investigators considered factors such as excess body mass index (BMI), chronic kidney disease (defined as estimated glomerular filtration rate less than 60 mL/min per 1.73 m2), poverty, poor-quality diet, lower educational level, alcohol consumption, and diuretic use in their analysis.
Dr. McCormick and coinvestigators included a total of 18,693 participants, consisting of 3,304 Black women, 6,195 White women, 3,085 Black men, and 6,109 White men.
They determined that the age-standardized prevalence of gout was 3.5% (95% confidence interval, 2.7%-4.3%) in Black women and 2.0% (95% CI, 1.5% - 2.5%) in White women (age-adjusted odds ratio, 1.81; 95% CI, 1.29-2.53). They calculated that the prevalence was 7.0% (95% CI, 6.2%-7.9%) in Black men and 5.4% (95% CI, 4.7%-6.2%) in White men (age-adjusted OR, 1.26; 95% CI, 1.02-1.55). They found similar differences in the prevalence of hyperuricemia between Black and White Americans.
The increased prevalence of gout in Black Americans, compared with White Americans, does not arise from genetics, according to McCormick. “Our conclusion was that it was due to social determinants of health,” she said. “When we adjusted for all socioclinical risk factors, the racial differences in gout and hyperuricemia prevalence disappeared. Importantly, stepwise regression analysis showed the two biggest drivers of the racial difference in gout prevalence among women were poverty itself, and excess BMI, which can be influenced by poverty.”
Dr. McCormick pointed out that in contrast to the current data, there was no racial difference in the prevalence of gout approximately 2 decades earlier, looking at data from the 1988-1994 NHANES III.
Given the findings, which included the fact that significantly more Black women and men were currently taking diuretics, compared with their White counterparts, Dr. McCormick pointed out clinicians should give more thought to medical therapies prescribed for conditions like high blood pressure to patients with gout or at risk for gout.
“One thing we found was that diuretic use was a driver” of gout, Dr. McCormick said. A prescriber “may want to consider different therapies that present a lower risk of gout if someone has hypertension. There could be greater consideration for prescribing alternatives to diuretics.”
More patient education and rheumatology referrals needed
An impediment to providing that education to patients with gout is unconscious bias on the part of the primary care provider, Dr. Wells said.
“It is about what your perspectives are and what you bring to the table,” he explained. “If you saw [a patient] who looked like someone in your family, that person will be treated differently [than someone who does not look like a family member]. That is where the whole concept [of unconscious bias] comes in.”
Primary care providers need to adopt a holistic approach to gout management that involves counseling about good nutrition, smoking cessation, regular exercise, and limiting alcohol consumption, in addition to medication adherence. Primary care providers may have a bias in treating their Black patients, failing to devote sufficient time and attention to assist them in getting their disease under control, he said.
“Gout should be just like any other chronic disease,” Dr. Wells said. “You need to have a target in mind, and you and your patient need to work together to get to that target. When [patients] end up in rheumatology offices, it is almost too late. I think the take-home message here is that in 2022 ... for any patient who has gout, that patient probably needs to be seen by a rheumatologist because, indeed, with aggressive therapy, preventive therapy, [and] education, and if they are on the right medications, they won’t end up with these crippling joints that we see all the time.”
Dr. McCormick and Dr. Wells disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Gout flares linked to transient jump in MI, stroke risk
There is evidence that gout and heart disease are mechanistically linked by inflammation and patients with gout are at elevated risk for cardiovascular disease (CVD). But do gout flares, on their own, affect short-term risk for CV events? A new analysis based on records from British medical practices suggests that might be the case.
Risk for myocardial infarction or stroke climbed in the weeks after individual gout flare-ups in the study’s more than 60,000 patients with a recent gout diagnosis. The jump in risk, significant but small in absolute terms, held for about 4 months in the case-control study before going away.
A sensitivity analysis that excluded patients who already had CVD when their gout was diagnosed yielded similar results.
The observational study isn’t able to show that gout flares themselves transiently raise the risk for MI or stroke, but it’s enough to send a cautionary message to physicians who care for patients with gout, rheumatologist Abhishek Abhishek, PhD, Nottingham (England) City Hospital, said in an interview.
In such patients who also have conditions like hypertension, diabetes, or dyslipidemia, or a history of heart disease, he said, it’s important “to manage risk factors really aggressively, knowing that when these patients have a gout flare, there’s a temporary increase in risk of a cardiovascular event.”
Managing their absolute CV risk – whether with drug therapy, lifestyle changes, or other interventions – should help limit the transient jump in risk for MI or stroke following a gout flare, proposed Dr. Abhishek, who is senior author on the study published in JAMA, with lead author Edoardo Cipolletta, MD, also from Nottingham City Hospital.
First robust evidence
The case-control study, which involved more than 60,000 patients with a recent gout diagnosis, some who went on to have MI or stroke, looked at rates of such events at different time intervals after gout flares. Those who experienced such events showed a more than 90% increased likelihood of a gout flare-up in the preceding 60 days, a greater than 50% chance of a flare between 60 and 120 days before the event, but no increased likelihood prior to 120 days before the event.
Such a link between gout flares and CV events “has been suspected but never proven,” observed rheumatologist Hyon K. Choi, MD, Harvard Medical School, Boston, who was not associated with the analysis. “This is the first time it has actually been shown in a robust way,” he said in an interview.
The study suggests a “likely causative relationship” between gout flares and CV events, but – as the published report noted – has limitations like any observational study, said Dr. Choi, who also directs the Gout & Crystal Arthropathy Center at Massachusetts General Hospital, Boston. “Hopefully, this can be replicated in other cohorts.”
The analysis controlled for a number of relevant potential confounders, he noted, but couldn’t account for all issues that could argue against gout flares as a direct cause of the MIs and strokes.
Gout attacks are a complex experience with a range of potential indirect effects on CV risk, Dr. Choi observed. They can immobilize patients, possibly raising their risk for thrombotic events, for example. They can be exceptionally painful, which causes stress and can lead to frequent or chronic use of glucocorticoids or NSAIDs, all of which can exacerbate high blood pressure and possibly worsen CV risk.
A unique insight
The timing of gout flares relative to acute vascular events hasn’t been fully explored, observed an accompanying editorial. The current study’s “unique insight,” it stated, “is that disease activity from gout was associated with an incremental increase in risk for acute vascular events during the time period immediately following the gout flare.”
Although the study is observational, a “large body of evidence from animal and human research, mechanistic insights, and clinical interventions” support an association between flares and vascular events and “make a causal link eminently reasonable,” stated the editorialists, Jeffrey L. Anderson, MD, and Kirk U. Knowlton, MD, both with Intermountain Medical Center, Salt Lake City, Utah.
The findings, they wrote, “should alert clinicians and patients to the increased cardiovascular risk in the weeks beginning after a gout flare and should focus attention on optimizing preventive measures.” Those can include “lifestyle measures and standard risk-factor control including adherence to diet, statins, anti-inflammatory drugs (e.g., aspirin, colchicine), smoking cessation, diabetic and blood pressure control, and antithrombotic medications as indicated.”
Dr. Choi said the current results argue for more liberal use of colchicine, and for preferring colchicine over other anti-inflammatories, in patients with gout and traditional CV risk factors, given multiple randomized trials supporting the drug’s use in such cases. “If you use colchicine, you are covering their heart disease risk as well as their gout. It’s two birds with one stone.”
Nested case-control study
The investigators accessed electronic health records from 96,153 patients with recently diagnosed gout in England from 1997 to 2020; the cohort’s mean age was about 76 years, and 69% of participants were men. They matched 10,475 patients with at least one CV event to 52,099 others who didn’t have such an event by age, sex, and time from gout diagnosis. In each matched set of patients, those not experiencing a CV event were assigned a flare-to-event interval based on their matching with patients who did experience such an event.
Those with CV events, compared with patients without an event, had a greater than 90% increased likelihood of experiencing a gout flare-up in the 60 days preceding the event, a more than 50% greater chance of a flare-up 60-120 days before the CV event, but no increased likelihood more than 120 days before the event.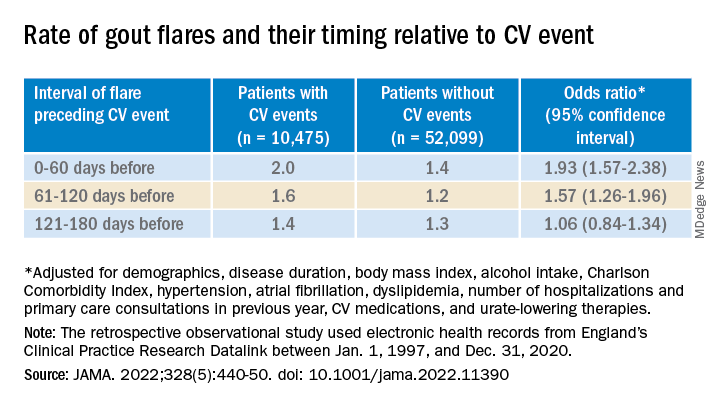
A self-controlled case series based on the same overall cohort with gout yielded similar results while sidestepping any potential for residual confounding, an inherent concern with any case–control analysis, the report notes. It involved 1,421 patients with one or more gout flare and at least one MI or stroke after the diagnosis of gout.
Among that cohort, the CV-event incidence rate ratio, adjusted for age and season of the year, by time interval after a gout flare, was 1.89 (95% confidence interval, 1.54-2.30) at 0-60 days, 1.64 (95% CI, 1.45-1.86) at 61-120 days, and1.29 (95% CI, 1.02-1.64) at 121-180 days.
Also similar, the report noted, were results of several sensitivity analyses, including one that excluded patients with confirmed CVD before their gout diagnosis; another that left out patients at low to moderate CV risk; and one that considered only gout flares treated with colchicine, corticosteroids, or NSAIDs.
The incremental CV event risks observed after flares in the study were small, which “has implications for both cost effectiveness and clinical relevance,” observed Dr. Anderson and Dr. Knowlton.
“An alternative to universal augmentation of cardiovascular risk prevention with therapies among patients with gout flares,” they wrote, would be “to further stratify risk by defining a group at highest near-term risk.” Such interventions could potentially be guided by markers of CV risk such as, for example, levels of high-sensitivity C-reactive protein or lipoprotein(a), or plaque burden on coronary-artery calcium scans.
Dr. Abhishek, Dr. Cipolletta, and the other authors reported no competing interests. Dr. Choi disclosed research support from Ironwood and Horizon; and consulting fees from Ironwood, Selecta, Horizon, Takeda, Kowa, and Vaxart. Dr. Anderson disclosed receiving grants to his institution from Novartis and Milestone.
A version of this article first appeared on Medscape.com.
There is evidence that gout and heart disease are mechanistically linked by inflammation and patients with gout are at elevated risk for cardiovascular disease (CVD). But do gout flares, on their own, affect short-term risk for CV events? A new analysis based on records from British medical practices suggests that might be the case.
Risk for myocardial infarction or stroke climbed in the weeks after individual gout flare-ups in the study’s more than 60,000 patients with a recent gout diagnosis. The jump in risk, significant but small in absolute terms, held for about 4 months in the case-control study before going away.
A sensitivity analysis that excluded patients who already had CVD when their gout was diagnosed yielded similar results.
The observational study isn’t able to show that gout flares themselves transiently raise the risk for MI or stroke, but it’s enough to send a cautionary message to physicians who care for patients with gout, rheumatologist Abhishek Abhishek, PhD, Nottingham (England) City Hospital, said in an interview.
In such patients who also have conditions like hypertension, diabetes, or dyslipidemia, or a history of heart disease, he said, it’s important “to manage risk factors really aggressively, knowing that when these patients have a gout flare, there’s a temporary increase in risk of a cardiovascular event.”
Managing their absolute CV risk – whether with drug therapy, lifestyle changes, or other interventions – should help limit the transient jump in risk for MI or stroke following a gout flare, proposed Dr. Abhishek, who is senior author on the study published in JAMA, with lead author Edoardo Cipolletta, MD, also from Nottingham City Hospital.
First robust evidence
The case-control study, which involved more than 60,000 patients with a recent gout diagnosis, some who went on to have MI or stroke, looked at rates of such events at different time intervals after gout flares. Those who experienced such events showed a more than 90% increased likelihood of a gout flare-up in the preceding 60 days, a greater than 50% chance of a flare between 60 and 120 days before the event, but no increased likelihood prior to 120 days before the event.
Such a link between gout flares and CV events “has been suspected but never proven,” observed rheumatologist Hyon K. Choi, MD, Harvard Medical School, Boston, who was not associated with the analysis. “This is the first time it has actually been shown in a robust way,” he said in an interview.
The study suggests a “likely causative relationship” between gout flares and CV events, but – as the published report noted – has limitations like any observational study, said Dr. Choi, who also directs the Gout & Crystal Arthropathy Center at Massachusetts General Hospital, Boston. “Hopefully, this can be replicated in other cohorts.”
The analysis controlled for a number of relevant potential confounders, he noted, but couldn’t account for all issues that could argue against gout flares as a direct cause of the MIs and strokes.
Gout attacks are a complex experience with a range of potential indirect effects on CV risk, Dr. Choi observed. They can immobilize patients, possibly raising their risk for thrombotic events, for example. They can be exceptionally painful, which causes stress and can lead to frequent or chronic use of glucocorticoids or NSAIDs, all of which can exacerbate high blood pressure and possibly worsen CV risk.
A unique insight
The timing of gout flares relative to acute vascular events hasn’t been fully explored, observed an accompanying editorial. The current study’s “unique insight,” it stated, “is that disease activity from gout was associated with an incremental increase in risk for acute vascular events during the time period immediately following the gout flare.”
Although the study is observational, a “large body of evidence from animal and human research, mechanistic insights, and clinical interventions” support an association between flares and vascular events and “make a causal link eminently reasonable,” stated the editorialists, Jeffrey L. Anderson, MD, and Kirk U. Knowlton, MD, both with Intermountain Medical Center, Salt Lake City, Utah.
The findings, they wrote, “should alert clinicians and patients to the increased cardiovascular risk in the weeks beginning after a gout flare and should focus attention on optimizing preventive measures.” Those can include “lifestyle measures and standard risk-factor control including adherence to diet, statins, anti-inflammatory drugs (e.g., aspirin, colchicine), smoking cessation, diabetic and blood pressure control, and antithrombotic medications as indicated.”
Dr. Choi said the current results argue for more liberal use of colchicine, and for preferring colchicine over other anti-inflammatories, in patients with gout and traditional CV risk factors, given multiple randomized trials supporting the drug’s use in such cases. “If you use colchicine, you are covering their heart disease risk as well as their gout. It’s two birds with one stone.”
Nested case-control study
The investigators accessed electronic health records from 96,153 patients with recently diagnosed gout in England from 1997 to 2020; the cohort’s mean age was about 76 years, and 69% of participants were men. They matched 10,475 patients with at least one CV event to 52,099 others who didn’t have such an event by age, sex, and time from gout diagnosis. In each matched set of patients, those not experiencing a CV event were assigned a flare-to-event interval based on their matching with patients who did experience such an event.
Those with CV events, compared with patients without an event, had a greater than 90% increased likelihood of experiencing a gout flare-up in the 60 days preceding the event, a more than 50% greater chance of a flare-up 60-120 days before the CV event, but no increased likelihood more than 120 days before the event.
A self-controlled case series based on the same overall cohort with gout yielded similar results while sidestepping any potential for residual confounding, an inherent concern with any case–control analysis, the report notes. It involved 1,421 patients with one or more gout flare and at least one MI or stroke after the diagnosis of gout.
Among that cohort, the CV-event incidence rate ratio, adjusted for age and season of the year, by time interval after a gout flare, was 1.89 (95% confidence interval, 1.54-2.30) at 0-60 days, 1.64 (95% CI, 1.45-1.86) at 61-120 days, and1.29 (95% CI, 1.02-1.64) at 121-180 days.
Also similar, the report noted, were results of several sensitivity analyses, including one that excluded patients with confirmed CVD before their gout diagnosis; another that left out patients at low to moderate CV risk; and one that considered only gout flares treated with colchicine, corticosteroids, or NSAIDs.
The incremental CV event risks observed after flares in the study were small, which “has implications for both cost effectiveness and clinical relevance,” observed Dr. Anderson and Dr. Knowlton.
“An alternative to universal augmentation of cardiovascular risk prevention with therapies among patients with gout flares,” they wrote, would be “to further stratify risk by defining a group at highest near-term risk.” Such interventions could potentially be guided by markers of CV risk such as, for example, levels of high-sensitivity C-reactive protein or lipoprotein(a), or plaque burden on coronary-artery calcium scans.
Dr. Abhishek, Dr. Cipolletta, and the other authors reported no competing interests. Dr. Choi disclosed research support from Ironwood and Horizon; and consulting fees from Ironwood, Selecta, Horizon, Takeda, Kowa, and Vaxart. Dr. Anderson disclosed receiving grants to his institution from Novartis and Milestone.
A version of this article first appeared on Medscape.com.
There is evidence that gout and heart disease are mechanistically linked by inflammation and patients with gout are at elevated risk for cardiovascular disease (CVD). But do gout flares, on their own, affect short-term risk for CV events? A new analysis based on records from British medical practices suggests that might be the case.
Risk for myocardial infarction or stroke climbed in the weeks after individual gout flare-ups in the study’s more than 60,000 patients with a recent gout diagnosis. The jump in risk, significant but small in absolute terms, held for about 4 months in the case-control study before going away.
A sensitivity analysis that excluded patients who already had CVD when their gout was diagnosed yielded similar results.
The observational study isn’t able to show that gout flares themselves transiently raise the risk for MI or stroke, but it’s enough to send a cautionary message to physicians who care for patients with gout, rheumatologist Abhishek Abhishek, PhD, Nottingham (England) City Hospital, said in an interview.
In such patients who also have conditions like hypertension, diabetes, or dyslipidemia, or a history of heart disease, he said, it’s important “to manage risk factors really aggressively, knowing that when these patients have a gout flare, there’s a temporary increase in risk of a cardiovascular event.”
Managing their absolute CV risk – whether with drug therapy, lifestyle changes, or other interventions – should help limit the transient jump in risk for MI or stroke following a gout flare, proposed Dr. Abhishek, who is senior author on the study published in JAMA, with lead author Edoardo Cipolletta, MD, also from Nottingham City Hospital.
First robust evidence
The case-control study, which involved more than 60,000 patients with a recent gout diagnosis, some who went on to have MI or stroke, looked at rates of such events at different time intervals after gout flares. Those who experienced such events showed a more than 90% increased likelihood of a gout flare-up in the preceding 60 days, a greater than 50% chance of a flare between 60 and 120 days before the event, but no increased likelihood prior to 120 days before the event.
Such a link between gout flares and CV events “has been suspected but never proven,” observed rheumatologist Hyon K. Choi, MD, Harvard Medical School, Boston, who was not associated with the analysis. “This is the first time it has actually been shown in a robust way,” he said in an interview.
The study suggests a “likely causative relationship” between gout flares and CV events, but – as the published report noted – has limitations like any observational study, said Dr. Choi, who also directs the Gout & Crystal Arthropathy Center at Massachusetts General Hospital, Boston. “Hopefully, this can be replicated in other cohorts.”
The analysis controlled for a number of relevant potential confounders, he noted, but couldn’t account for all issues that could argue against gout flares as a direct cause of the MIs and strokes.
Gout attacks are a complex experience with a range of potential indirect effects on CV risk, Dr. Choi observed. They can immobilize patients, possibly raising their risk for thrombotic events, for example. They can be exceptionally painful, which causes stress and can lead to frequent or chronic use of glucocorticoids or NSAIDs, all of which can exacerbate high blood pressure and possibly worsen CV risk.
A unique insight
The timing of gout flares relative to acute vascular events hasn’t been fully explored, observed an accompanying editorial. The current study’s “unique insight,” it stated, “is that disease activity from gout was associated with an incremental increase in risk for acute vascular events during the time period immediately following the gout flare.”
Although the study is observational, a “large body of evidence from animal and human research, mechanistic insights, and clinical interventions” support an association between flares and vascular events and “make a causal link eminently reasonable,” stated the editorialists, Jeffrey L. Anderson, MD, and Kirk U. Knowlton, MD, both with Intermountain Medical Center, Salt Lake City, Utah.
The findings, they wrote, “should alert clinicians and patients to the increased cardiovascular risk in the weeks beginning after a gout flare and should focus attention on optimizing preventive measures.” Those can include “lifestyle measures and standard risk-factor control including adherence to diet, statins, anti-inflammatory drugs (e.g., aspirin, colchicine), smoking cessation, diabetic and blood pressure control, and antithrombotic medications as indicated.”
Dr. Choi said the current results argue for more liberal use of colchicine, and for preferring colchicine over other anti-inflammatories, in patients with gout and traditional CV risk factors, given multiple randomized trials supporting the drug’s use in such cases. “If you use colchicine, you are covering their heart disease risk as well as their gout. It’s two birds with one stone.”
Nested case-control study
The investigators accessed electronic health records from 96,153 patients with recently diagnosed gout in England from 1997 to 2020; the cohort’s mean age was about 76 years, and 69% of participants were men. They matched 10,475 patients with at least one CV event to 52,099 others who didn’t have such an event by age, sex, and time from gout diagnosis. In each matched set of patients, those not experiencing a CV event were assigned a flare-to-event interval based on their matching with patients who did experience such an event.
Those with CV events, compared with patients without an event, had a greater than 90% increased likelihood of experiencing a gout flare-up in the 60 days preceding the event, a more than 50% greater chance of a flare-up 60-120 days before the CV event, but no increased likelihood more than 120 days before the event.
A self-controlled case series based on the same overall cohort with gout yielded similar results while sidestepping any potential for residual confounding, an inherent concern with any case–control analysis, the report notes. It involved 1,421 patients with one or more gout flare and at least one MI or stroke after the diagnosis of gout.
Among that cohort, the CV-event incidence rate ratio, adjusted for age and season of the year, by time interval after a gout flare, was 1.89 (95% confidence interval, 1.54-2.30) at 0-60 days, 1.64 (95% CI, 1.45-1.86) at 61-120 days, and1.29 (95% CI, 1.02-1.64) at 121-180 days.
Also similar, the report noted, were results of several sensitivity analyses, including one that excluded patients with confirmed CVD before their gout diagnosis; another that left out patients at low to moderate CV risk; and one that considered only gout flares treated with colchicine, corticosteroids, or NSAIDs.
The incremental CV event risks observed after flares in the study were small, which “has implications for both cost effectiveness and clinical relevance,” observed Dr. Anderson and Dr. Knowlton.
“An alternative to universal augmentation of cardiovascular risk prevention with therapies among patients with gout flares,” they wrote, would be “to further stratify risk by defining a group at highest near-term risk.” Such interventions could potentially be guided by markers of CV risk such as, for example, levels of high-sensitivity C-reactive protein or lipoprotein(a), or plaque burden on coronary-artery calcium scans.
Dr. Abhishek, Dr. Cipolletta, and the other authors reported no competing interests. Dr. Choi disclosed research support from Ironwood and Horizon; and consulting fees from Ironwood, Selecta, Horizon, Takeda, Kowa, and Vaxart. Dr. Anderson disclosed receiving grants to his institution from Novartis and Milestone.
A version of this article first appeared on Medscape.com.
FROM JAMA
Benzbromarone tops febuxostat for gout?
Benzbromarone is not approved in the United States because of concerns of acute liver injury but is approved in several other countries, including China, Brazil, and New Zealand.
“The results suggest that low dosing of benzbromarone may warrant stronger consideration as a safe and effective therapy to achieve serum urate target in gout without moderate chronic kidney disease,” the study team writes.
“Benzbromarone is severely hepatotoxic in some individuals and unlikely to ever gain approval in the United States,” one of the study’s investigators, Robert Terkeltaub, MD, professor of medicine, University of California, San Diego, told this news organization.
However, this study “illustrates the value and impact of uricosuric therapy in general in gout, including potentially as an initial urate-lowering monotherapy strategy, and the sheer number of subjects reaching urate target with low-dose uricosuric monotherapy was impressive,” Dr. Terkeltaub said.
The study was published online in Arthritis & Rheumatology.
“Renal uric acid underexcretion is the chief mechanism driving hyperuricemia in gout, yet the standard urate-lowering therapy recommendation is first-line xanthine oxidase inhibition irrespective of the cause of hyperuricemia,” the study team explains in their article.
Their prospective, randomized, single-center, open-labeled trial was conducted at the Gout Clinic of the Affiliated Hospital of Qingdao University, China.
A total of 196 relatively young healthy men with gout and uric acid underexcretion were randomly assigned to receive low-dose benzbromarone (25 mg/d) or low-dose febuxostat (20 mg/d) for 12 weeks.
Renal uric acid underexcretion was defined as fractional excretion of urate less than 5.5% and uric acid excretion less than or equal to 600 mg/d/1.73 m2.
A “major aspect” of this comparative effectiveness trial was its specific focus on gout-associated renal uric acid underexcretion, where the uricosuric targeted the dominant abnormality promoting the hyperuricemia, Dr. Terkeltaub told this news organization.
In addition, all participants received daily urine alkalinization with oral sodium bicarbonate. “This is not always done in clinical practice, nor in clinical trials of uricosuric agents,” Dr. Terkeltaub said.
The results showed that more participants in the benzbromarone group achieved the serum urate target of less than 6 mg/dL, compared with those in the febuxostat group (primary endpoint, 61% vs. 32%, P < .001).
Adverse events, including gout flares and urolithiasis, did not differ significantly between the two groups, with the exception of more transaminase elevation in the febuxostat group (15% vs. 4%; P = .008).
“We did not find severe hepatotoxicity with low-dose benzbromarone, but ethnic background may affect drug responses, and severe hepatotoxicity of benzbromarone has rarely been reported in Asia,” the authors write.
The incidence of urolithiasis was numerically, but not significantly, higher in the benzbromarone group (5% vs. 2%).
This study found no significant changes in participants’ triglyceride levels, though a previous study suggested febuxostat could increase serum triglycerides.
The investigators caution that the study only included patients who had baseline serum urate levels ranging from 8.0 to 10 mg/dL, who were relatively young and with few comorbidities.
The authors further noted that the “... results may not be generalizable to patients with higher serum urate levels or impaired kidney function, as well [as] patients from other geographical regions, age, and ethnicity groups. The study only included men, and the findings may not be generalizable to women with gout.”
‘Very useful’ in select cases
Weighing in on the results, Valderilio Feijó Azevedo, MD, PhD, adjunct professor of rheumatology, Federal University of Paraná, Brazil, noted that in some specific clinical circumstances, benzbromarone has been “a very useful medication, alone or combined, to treat gout patients.”
“We have great experience with the drug in Brazil. However, it is not used to treat all patients. Patients must be very well-selected in our clinical practice,” Dr. Azevedo said in an interview.
“For most patients, benzbromarone is effective for those who have failed to achieve serum uric acid goals with allopurinol treatment. We do not use it to treat patients with asymptomatic hyperuricemia. In general, we avoid patients with hepatic dysfunction due to previous hepatotoxicity reports. In every patient, we do active monitoring of enzymes,” Dr. Azevedo explained.
“We also avoid using it in patients with severe kidney disease. However, we have used it in some patients with estimated glomerular filtration rate less than 30. We also avoid dosage over 200 mg per day. On average, we use 100 mg per day combined with allopurinol or alone,” said Dr. Azevedo, who was not involved with the study.
Also weighing in, Michael Pillinger, MD, rheumatologist at NYU Langone Health, noted that while benzbromarone is not used in the United States, “in many parts of the world, it is used and is felt to be effective.” Dr. Pillinger was not associated with this current research.
This study, Dr. Pillinger said, “does underline the fact that an alternative drug that lowers urate by promoting urate excretion, if it could gain [U.S. Food and Drug Association] approval and if it were safe, could present a viable new option for therapy.”
He added, “If one conclusion to the study is that determining the basis of hyperuricemia is helpful in guiding benzbromarone use, that implies an additional layer of effort for physicians and patients in a disease that is already notoriously known for patient noncompliance – and in a case where febuxostat and allopurinol will work for both overproducers and underexcreters and would not need this additional assessment.”
The study was sponsored by Shandong Provincial Key Research and Development Plan, the National Natural Science Foundation of China, and Shandong Provincial Science Foundation for Outstanding Youth Scholarship. Dr. Terkeltaub was supported by the National Institutes of Health and the VA Research Service. Dr. Terkeltaub has received research funding from AstraZeneca, and has consulted with Horizon, Selecta, SOBI, Dyve BioSciences, Fortress, AstraZeneca, Allena, Fortress Biotech, and LG Life Sciences. Dr. Azevedo and Dr. Pillinger have no reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Benzbromarone is not approved in the United States because of concerns of acute liver injury but is approved in several other countries, including China, Brazil, and New Zealand.
“The results suggest that low dosing of benzbromarone may warrant stronger consideration as a safe and effective therapy to achieve serum urate target in gout without moderate chronic kidney disease,” the study team writes.
“Benzbromarone is severely hepatotoxic in some individuals and unlikely to ever gain approval in the United States,” one of the study’s investigators, Robert Terkeltaub, MD, professor of medicine, University of California, San Diego, told this news organization.
However, this study “illustrates the value and impact of uricosuric therapy in general in gout, including potentially as an initial urate-lowering monotherapy strategy, and the sheer number of subjects reaching urate target with low-dose uricosuric monotherapy was impressive,” Dr. Terkeltaub said.
The study was published online in Arthritis & Rheumatology.
“Renal uric acid underexcretion is the chief mechanism driving hyperuricemia in gout, yet the standard urate-lowering therapy recommendation is first-line xanthine oxidase inhibition irrespective of the cause of hyperuricemia,” the study team explains in their article.
Their prospective, randomized, single-center, open-labeled trial was conducted at the Gout Clinic of the Affiliated Hospital of Qingdao University, China.
A total of 196 relatively young healthy men with gout and uric acid underexcretion were randomly assigned to receive low-dose benzbromarone (25 mg/d) or low-dose febuxostat (20 mg/d) for 12 weeks.
Renal uric acid underexcretion was defined as fractional excretion of urate less than 5.5% and uric acid excretion less than or equal to 600 mg/d/1.73 m2.
A “major aspect” of this comparative effectiveness trial was its specific focus on gout-associated renal uric acid underexcretion, where the uricosuric targeted the dominant abnormality promoting the hyperuricemia, Dr. Terkeltaub told this news organization.
In addition, all participants received daily urine alkalinization with oral sodium bicarbonate. “This is not always done in clinical practice, nor in clinical trials of uricosuric agents,” Dr. Terkeltaub said.
The results showed that more participants in the benzbromarone group achieved the serum urate target of less than 6 mg/dL, compared with those in the febuxostat group (primary endpoint, 61% vs. 32%, P < .001).
Adverse events, including gout flares and urolithiasis, did not differ significantly between the two groups, with the exception of more transaminase elevation in the febuxostat group (15% vs. 4%; P = .008).
“We did not find severe hepatotoxicity with low-dose benzbromarone, but ethnic background may affect drug responses, and severe hepatotoxicity of benzbromarone has rarely been reported in Asia,” the authors write.
The incidence of urolithiasis was numerically, but not significantly, higher in the benzbromarone group (5% vs. 2%).
This study found no significant changes in participants’ triglyceride levels, though a previous study suggested febuxostat could increase serum triglycerides.
The investigators caution that the study only included patients who had baseline serum urate levels ranging from 8.0 to 10 mg/dL, who were relatively young and with few comorbidities.
The authors further noted that the “... results may not be generalizable to patients with higher serum urate levels or impaired kidney function, as well [as] patients from other geographical regions, age, and ethnicity groups. The study only included men, and the findings may not be generalizable to women with gout.”
‘Very useful’ in select cases
Weighing in on the results, Valderilio Feijó Azevedo, MD, PhD, adjunct professor of rheumatology, Federal University of Paraná, Brazil, noted that in some specific clinical circumstances, benzbromarone has been “a very useful medication, alone or combined, to treat gout patients.”
“We have great experience with the drug in Brazil. However, it is not used to treat all patients. Patients must be very well-selected in our clinical practice,” Dr. Azevedo said in an interview.
“For most patients, benzbromarone is effective for those who have failed to achieve serum uric acid goals with allopurinol treatment. We do not use it to treat patients with asymptomatic hyperuricemia. In general, we avoid patients with hepatic dysfunction due to previous hepatotoxicity reports. In every patient, we do active monitoring of enzymes,” Dr. Azevedo explained.
“We also avoid using it in patients with severe kidney disease. However, we have used it in some patients with estimated glomerular filtration rate less than 30. We also avoid dosage over 200 mg per day. On average, we use 100 mg per day combined with allopurinol or alone,” said Dr. Azevedo, who was not involved with the study.
Also weighing in, Michael Pillinger, MD, rheumatologist at NYU Langone Health, noted that while benzbromarone is not used in the United States, “in many parts of the world, it is used and is felt to be effective.” Dr. Pillinger was not associated with this current research.
This study, Dr. Pillinger said, “does underline the fact that an alternative drug that lowers urate by promoting urate excretion, if it could gain [U.S. Food and Drug Association] approval and if it were safe, could present a viable new option for therapy.”
He added, “If one conclusion to the study is that determining the basis of hyperuricemia is helpful in guiding benzbromarone use, that implies an additional layer of effort for physicians and patients in a disease that is already notoriously known for patient noncompliance – and in a case where febuxostat and allopurinol will work for both overproducers and underexcreters and would not need this additional assessment.”
The study was sponsored by Shandong Provincial Key Research and Development Plan, the National Natural Science Foundation of China, and Shandong Provincial Science Foundation for Outstanding Youth Scholarship. Dr. Terkeltaub was supported by the National Institutes of Health and the VA Research Service. Dr. Terkeltaub has received research funding from AstraZeneca, and has consulted with Horizon, Selecta, SOBI, Dyve BioSciences, Fortress, AstraZeneca, Allena, Fortress Biotech, and LG Life Sciences. Dr. Azevedo and Dr. Pillinger have no reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Benzbromarone is not approved in the United States because of concerns of acute liver injury but is approved in several other countries, including China, Brazil, and New Zealand.
“The results suggest that low dosing of benzbromarone may warrant stronger consideration as a safe and effective therapy to achieve serum urate target in gout without moderate chronic kidney disease,” the study team writes.
“Benzbromarone is severely hepatotoxic in some individuals and unlikely to ever gain approval in the United States,” one of the study’s investigators, Robert Terkeltaub, MD, professor of medicine, University of California, San Diego, told this news organization.
However, this study “illustrates the value and impact of uricosuric therapy in general in gout, including potentially as an initial urate-lowering monotherapy strategy, and the sheer number of subjects reaching urate target with low-dose uricosuric monotherapy was impressive,” Dr. Terkeltaub said.
The study was published online in Arthritis & Rheumatology.
“Renal uric acid underexcretion is the chief mechanism driving hyperuricemia in gout, yet the standard urate-lowering therapy recommendation is first-line xanthine oxidase inhibition irrespective of the cause of hyperuricemia,” the study team explains in their article.
Their prospective, randomized, single-center, open-labeled trial was conducted at the Gout Clinic of the Affiliated Hospital of Qingdao University, China.
A total of 196 relatively young healthy men with gout and uric acid underexcretion were randomly assigned to receive low-dose benzbromarone (25 mg/d) or low-dose febuxostat (20 mg/d) for 12 weeks.
Renal uric acid underexcretion was defined as fractional excretion of urate less than 5.5% and uric acid excretion less than or equal to 600 mg/d/1.73 m2.
A “major aspect” of this comparative effectiveness trial was its specific focus on gout-associated renal uric acid underexcretion, where the uricosuric targeted the dominant abnormality promoting the hyperuricemia, Dr. Terkeltaub told this news organization.
In addition, all participants received daily urine alkalinization with oral sodium bicarbonate. “This is not always done in clinical practice, nor in clinical trials of uricosuric agents,” Dr. Terkeltaub said.
The results showed that more participants in the benzbromarone group achieved the serum urate target of less than 6 mg/dL, compared with those in the febuxostat group (primary endpoint, 61% vs. 32%, P < .001).
Adverse events, including gout flares and urolithiasis, did not differ significantly between the two groups, with the exception of more transaminase elevation in the febuxostat group (15% vs. 4%; P = .008).
“We did not find severe hepatotoxicity with low-dose benzbromarone, but ethnic background may affect drug responses, and severe hepatotoxicity of benzbromarone has rarely been reported in Asia,” the authors write.
The incidence of urolithiasis was numerically, but not significantly, higher in the benzbromarone group (5% vs. 2%).
This study found no significant changes in participants’ triglyceride levels, though a previous study suggested febuxostat could increase serum triglycerides.
The investigators caution that the study only included patients who had baseline serum urate levels ranging from 8.0 to 10 mg/dL, who were relatively young and with few comorbidities.
The authors further noted that the “... results may not be generalizable to patients with higher serum urate levels or impaired kidney function, as well [as] patients from other geographical regions, age, and ethnicity groups. The study only included men, and the findings may not be generalizable to women with gout.”
‘Very useful’ in select cases
Weighing in on the results, Valderilio Feijó Azevedo, MD, PhD, adjunct professor of rheumatology, Federal University of Paraná, Brazil, noted that in some specific clinical circumstances, benzbromarone has been “a very useful medication, alone or combined, to treat gout patients.”
“We have great experience with the drug in Brazil. However, it is not used to treat all patients. Patients must be very well-selected in our clinical practice,” Dr. Azevedo said in an interview.
“For most patients, benzbromarone is effective for those who have failed to achieve serum uric acid goals with allopurinol treatment. We do not use it to treat patients with asymptomatic hyperuricemia. In general, we avoid patients with hepatic dysfunction due to previous hepatotoxicity reports. In every patient, we do active monitoring of enzymes,” Dr. Azevedo explained.
“We also avoid using it in patients with severe kidney disease. However, we have used it in some patients with estimated glomerular filtration rate less than 30. We also avoid dosage over 200 mg per day. On average, we use 100 mg per day combined with allopurinol or alone,” said Dr. Azevedo, who was not involved with the study.
Also weighing in, Michael Pillinger, MD, rheumatologist at NYU Langone Health, noted that while benzbromarone is not used in the United States, “in many parts of the world, it is used and is felt to be effective.” Dr. Pillinger was not associated with this current research.
This study, Dr. Pillinger said, “does underline the fact that an alternative drug that lowers urate by promoting urate excretion, if it could gain [U.S. Food and Drug Association] approval and if it were safe, could present a viable new option for therapy.”
He added, “If one conclusion to the study is that determining the basis of hyperuricemia is helpful in guiding benzbromarone use, that implies an additional layer of effort for physicians and patients in a disease that is already notoriously known for patient noncompliance – and in a case where febuxostat and allopurinol will work for both overproducers and underexcreters and would not need this additional assessment.”
The study was sponsored by Shandong Provincial Key Research and Development Plan, the National Natural Science Foundation of China, and Shandong Provincial Science Foundation for Outstanding Youth Scholarship. Dr. Terkeltaub was supported by the National Institutes of Health and the VA Research Service. Dr. Terkeltaub has received research funding from AstraZeneca, and has consulted with Horizon, Selecta, SOBI, Dyve BioSciences, Fortress, AstraZeneca, Allena, Fortress Biotech, and LG Life Sciences. Dr. Azevedo and Dr. Pillinger have no reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.