User login
Fracking sites tied to increased heart failure hospitalizations
Living near hydraulic fracturing is associated with increased risk of hospitalization in people with heart failure (HF), a new study from Pennsylvania suggests.
The link was strongest among those with more severe heart failure but patients with either HF phenotype showed this association of increased risk with exposure to fracking activities, according to the investigators, led by Tara P. McAlexander, PhD, MPH, Drexel University Dornsife School of Public Health in Philadelphia.
“Our understanding has expanded well beyond the famous Harvard Six Cities study to know that it’s not just a short-term uptick in air pollution that›s going to send someone to the hospital a couple days later,” said Dr. McAlexander in an interview, referring to the study conducted from the mid-1970s through 1991. “We know that people who live in these environments and are exposed for long periods of time may have long-term detrimental effects.”
Although questions remain about specific mechanisms and how best to assess exposure, the evidence is mounting in a way that is consistent with the biologic hypotheses of how fracking would adversely affect health, Dr. McAlexander said. “We have many studies now on adverse pregnancy and birth outcomes, and that’s just the tip of the iceberg.”
Pennsylvania is a hot spot for fracking, also known as unconventional natural gas development (UNGD), with more than 12,000 wells drilled in the Marcellus shale since 2004. The shale extends from upstate New York in the north to northeastern Kentucky and Tennessee in the south and covers about 72,000 square miles. Last year, Pennsylvania pledged $3 million to study clusters of rare pediatric cancers and asthma near fracking operations. A recent grand jury report concluded government officials failed to protect residents from the health effects of fracking.
Fracking involves a cascade of activities that can trigger neural circuitry, sympathetic activation, and inflammation – all well-known pathways that potentiate heart failure, said Sanjay Rajagopalan, MD, who has researched the health effects of air pollution for two decades and was not involved with the study.
“If you think about it, it’s like environmental perturbation on steroids in some ways where they are pulling the trigger from a variety of different ways: noise, air pollution, social displacement, psychosocial impacts, economic disparities. So it’s not at all surprising that they saw an association,” said Dr. Rajagopalan, chief of cardiovascular medicine at University Hospitals Harrington Heart & Vascular Institute and director of the Case Western Cardiovascular Research Institute, both in Cleveland, Ohio.
As reported in the Journal of the American College of Cardiology, Dr. McAlexander and colleagues at Johns Hopkins University, Baltimore, used electronic health data from the Geisinger Health System to identify 9,054 patients with heart failure seen between 2008 and 2015. Of these, 5,839 patients had an incident HF hospitalization and 3,215 served as controls. Geisinger operates 13 hospitals and two research centers in 45 of Pennsylvania’s 67 counties, serving more than 3 million of the state’s residents.
Patients’ residential addresses were used to identify latitude and longitude coordinates that were matched with 9,669 UNGD wells in Pennsylvania and the location of major and minor roadways. The researchers also calculated a measure of community socioeconomic deprivation.
The adjusted odds of hospitalization were higher for patients in the highest quartile of exposure for three of the four UNGD phases: pad preparation (odds ratio, 1.70; 95% confidence interval, 1.35-2.13), stimulation or the actual fracking (OR, 1.80; 95% CI, 1.35-2.40), and production (OR, 1.62; 95% CI, 1.07-2.45).
Dr. McAlexander said she initially thought the lack of association with drilling (OR, 0.97; 95% CI, 0.75-1.27) was a mistake but noted that the drilling metric reflects a shorter time period than, for example, 30 days needed to clear the well pad and bring in the necessary equipment.
Stronger associations between pad preparation, fracking, and production are also consistent with the known increases in air pollution, traffic, and noise associated with these phases.
Individuals with more severe HF had greater odds of hospitalization, but the effect sizes were generally comparable between HF with preserved versus reduced ejection fraction. For those with the highest exposure to fracking, the odds ratios for hospitalization reached 2.25 (95% CI, 1.56-3.25) and 2.09 (95% CI, 1.44-3.03), respectively.
Notably, patients who could be phenotyped versus those who could not were more likely to die, to be hospitalized for HF, and to have a higher Charlson Comorbidity Index and other relevant diagnoses like myocardial infarction.
“Clinicians need to be increasingly aware that the environments their patients are in are a huge factor in their disease progression and outlook,” McAlexander said. “We know that UNGD, specifically now, is something that could be impacting a heart failure patient’s survival.”
She also suggested that the findings may also spur more advocacy work and “across-silo” collaboration between clinicians and environmental researchers.
Dr. Rajagopalan said there is increasing recognition that physicians need to be aware of environmental health links as extreme events like the California and Oregon wildfires and coastal flooding become increasingly common. “Unfortunately, unconventional is becoming the new convention.”
The problem for many physicians, however, is just having enough bandwidth to get through the day and get enough learning to keep above water, he said. Artificial intelligence could be used to seed electronic medical records with other personalized information from a bevy of sources including smartphones and the internet of things, but fundamental changes are also needed in the educational process to emphasize the environment.
“It’s going to take a huge societal shift in the way we view commodities, what we consider healthy, etc, but it can happen very quickly because all it takes is a crisis like COVID-19 to bring people to their knees and make them understand how this is going to take over our lives over the next decade,” Dr. Rajagopalan said.
The scientific community has been calling for “good” epidemiologic studies on the health effects of fracking since the early 2010s, Barrak Alahmad, MBChB, MPH, Harvard T.H. Chan School of Public Health, and Haitham Khraishah, MD, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, point out in an accompanying editorial.
The current study applied “extensive and rigorous methods” involving both the design and statistical approach, including use of a negative control analysis to assess for sources of spurious causal inference, several sensitivity analyses, and controlled for a wide range of covariates.
“Their results were consistent and robust across all these measures,” the editorialists wrote. “Most importantly, the effect size is probably too large to be explained away by an unmeasured confounder.”
Dr. Alahmad and Dr. Khraishah call for advancements in exposure assessment, citing a recent study reporting that ambient particle radioactivity near unconventional oil and gas sites could induce adverse health effects. Other unmet needs include a better understanding of racial disparities in the impacts of fracking and a fine-tuning of cause-specific cardiovascular morbidity and mortality.
The study was supported by training grants from the National Institute of Environmental Health Sciences to Dr. McAlexander and principal investigator Brian Schwartz, MD. The authors, Dr. Rajagopalan, Dr. Alahmad, and Dr. Khraishah have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Living near hydraulic fracturing is associated with increased risk of hospitalization in people with heart failure (HF), a new study from Pennsylvania suggests.
The link was strongest among those with more severe heart failure but patients with either HF phenotype showed this association of increased risk with exposure to fracking activities, according to the investigators, led by Tara P. McAlexander, PhD, MPH, Drexel University Dornsife School of Public Health in Philadelphia.
“Our understanding has expanded well beyond the famous Harvard Six Cities study to know that it’s not just a short-term uptick in air pollution that›s going to send someone to the hospital a couple days later,” said Dr. McAlexander in an interview, referring to the study conducted from the mid-1970s through 1991. “We know that people who live in these environments and are exposed for long periods of time may have long-term detrimental effects.”
Although questions remain about specific mechanisms and how best to assess exposure, the evidence is mounting in a way that is consistent with the biologic hypotheses of how fracking would adversely affect health, Dr. McAlexander said. “We have many studies now on adverse pregnancy and birth outcomes, and that’s just the tip of the iceberg.”
Pennsylvania is a hot spot for fracking, also known as unconventional natural gas development (UNGD), with more than 12,000 wells drilled in the Marcellus shale since 2004. The shale extends from upstate New York in the north to northeastern Kentucky and Tennessee in the south and covers about 72,000 square miles. Last year, Pennsylvania pledged $3 million to study clusters of rare pediatric cancers and asthma near fracking operations. A recent grand jury report concluded government officials failed to protect residents from the health effects of fracking.
Fracking involves a cascade of activities that can trigger neural circuitry, sympathetic activation, and inflammation – all well-known pathways that potentiate heart failure, said Sanjay Rajagopalan, MD, who has researched the health effects of air pollution for two decades and was not involved with the study.
“If you think about it, it’s like environmental perturbation on steroids in some ways where they are pulling the trigger from a variety of different ways: noise, air pollution, social displacement, psychosocial impacts, economic disparities. So it’s not at all surprising that they saw an association,” said Dr. Rajagopalan, chief of cardiovascular medicine at University Hospitals Harrington Heart & Vascular Institute and director of the Case Western Cardiovascular Research Institute, both in Cleveland, Ohio.
As reported in the Journal of the American College of Cardiology, Dr. McAlexander and colleagues at Johns Hopkins University, Baltimore, used electronic health data from the Geisinger Health System to identify 9,054 patients with heart failure seen between 2008 and 2015. Of these, 5,839 patients had an incident HF hospitalization and 3,215 served as controls. Geisinger operates 13 hospitals and two research centers in 45 of Pennsylvania’s 67 counties, serving more than 3 million of the state’s residents.
Patients’ residential addresses were used to identify latitude and longitude coordinates that were matched with 9,669 UNGD wells in Pennsylvania and the location of major and minor roadways. The researchers also calculated a measure of community socioeconomic deprivation.
The adjusted odds of hospitalization were higher for patients in the highest quartile of exposure for three of the four UNGD phases: pad preparation (odds ratio, 1.70; 95% confidence interval, 1.35-2.13), stimulation or the actual fracking (OR, 1.80; 95% CI, 1.35-2.40), and production (OR, 1.62; 95% CI, 1.07-2.45).
Dr. McAlexander said she initially thought the lack of association with drilling (OR, 0.97; 95% CI, 0.75-1.27) was a mistake but noted that the drilling metric reflects a shorter time period than, for example, 30 days needed to clear the well pad and bring in the necessary equipment.
Stronger associations between pad preparation, fracking, and production are also consistent with the known increases in air pollution, traffic, and noise associated with these phases.
Individuals with more severe HF had greater odds of hospitalization, but the effect sizes were generally comparable between HF with preserved versus reduced ejection fraction. For those with the highest exposure to fracking, the odds ratios for hospitalization reached 2.25 (95% CI, 1.56-3.25) and 2.09 (95% CI, 1.44-3.03), respectively.
Notably, patients who could be phenotyped versus those who could not were more likely to die, to be hospitalized for HF, and to have a higher Charlson Comorbidity Index and other relevant diagnoses like myocardial infarction.
“Clinicians need to be increasingly aware that the environments their patients are in are a huge factor in their disease progression and outlook,” McAlexander said. “We know that UNGD, specifically now, is something that could be impacting a heart failure patient’s survival.”
She also suggested that the findings may also spur more advocacy work and “across-silo” collaboration between clinicians and environmental researchers.
Dr. Rajagopalan said there is increasing recognition that physicians need to be aware of environmental health links as extreme events like the California and Oregon wildfires and coastal flooding become increasingly common. “Unfortunately, unconventional is becoming the new convention.”
The problem for many physicians, however, is just having enough bandwidth to get through the day and get enough learning to keep above water, he said. Artificial intelligence could be used to seed electronic medical records with other personalized information from a bevy of sources including smartphones and the internet of things, but fundamental changes are also needed in the educational process to emphasize the environment.
“It’s going to take a huge societal shift in the way we view commodities, what we consider healthy, etc, but it can happen very quickly because all it takes is a crisis like COVID-19 to bring people to their knees and make them understand how this is going to take over our lives over the next decade,” Dr. Rajagopalan said.
The scientific community has been calling for “good” epidemiologic studies on the health effects of fracking since the early 2010s, Barrak Alahmad, MBChB, MPH, Harvard T.H. Chan School of Public Health, and Haitham Khraishah, MD, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, point out in an accompanying editorial.
The current study applied “extensive and rigorous methods” involving both the design and statistical approach, including use of a negative control analysis to assess for sources of spurious causal inference, several sensitivity analyses, and controlled for a wide range of covariates.
“Their results were consistent and robust across all these measures,” the editorialists wrote. “Most importantly, the effect size is probably too large to be explained away by an unmeasured confounder.”
Dr. Alahmad and Dr. Khraishah call for advancements in exposure assessment, citing a recent study reporting that ambient particle radioactivity near unconventional oil and gas sites could induce adverse health effects. Other unmet needs include a better understanding of racial disparities in the impacts of fracking and a fine-tuning of cause-specific cardiovascular morbidity and mortality.
The study was supported by training grants from the National Institute of Environmental Health Sciences to Dr. McAlexander and principal investigator Brian Schwartz, MD. The authors, Dr. Rajagopalan, Dr. Alahmad, and Dr. Khraishah have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Living near hydraulic fracturing is associated with increased risk of hospitalization in people with heart failure (HF), a new study from Pennsylvania suggests.
The link was strongest among those with more severe heart failure but patients with either HF phenotype showed this association of increased risk with exposure to fracking activities, according to the investigators, led by Tara P. McAlexander, PhD, MPH, Drexel University Dornsife School of Public Health in Philadelphia.
“Our understanding has expanded well beyond the famous Harvard Six Cities study to know that it’s not just a short-term uptick in air pollution that›s going to send someone to the hospital a couple days later,” said Dr. McAlexander in an interview, referring to the study conducted from the mid-1970s through 1991. “We know that people who live in these environments and are exposed for long periods of time may have long-term detrimental effects.”
Although questions remain about specific mechanisms and how best to assess exposure, the evidence is mounting in a way that is consistent with the biologic hypotheses of how fracking would adversely affect health, Dr. McAlexander said. “We have many studies now on adverse pregnancy and birth outcomes, and that’s just the tip of the iceberg.”
Pennsylvania is a hot spot for fracking, also known as unconventional natural gas development (UNGD), with more than 12,000 wells drilled in the Marcellus shale since 2004. The shale extends from upstate New York in the north to northeastern Kentucky and Tennessee in the south and covers about 72,000 square miles. Last year, Pennsylvania pledged $3 million to study clusters of rare pediatric cancers and asthma near fracking operations. A recent grand jury report concluded government officials failed to protect residents from the health effects of fracking.
Fracking involves a cascade of activities that can trigger neural circuitry, sympathetic activation, and inflammation – all well-known pathways that potentiate heart failure, said Sanjay Rajagopalan, MD, who has researched the health effects of air pollution for two decades and was not involved with the study.
“If you think about it, it’s like environmental perturbation on steroids in some ways where they are pulling the trigger from a variety of different ways: noise, air pollution, social displacement, psychosocial impacts, economic disparities. So it’s not at all surprising that they saw an association,” said Dr. Rajagopalan, chief of cardiovascular medicine at University Hospitals Harrington Heart & Vascular Institute and director of the Case Western Cardiovascular Research Institute, both in Cleveland, Ohio.
As reported in the Journal of the American College of Cardiology, Dr. McAlexander and colleagues at Johns Hopkins University, Baltimore, used electronic health data from the Geisinger Health System to identify 9,054 patients with heart failure seen between 2008 and 2015. Of these, 5,839 patients had an incident HF hospitalization and 3,215 served as controls. Geisinger operates 13 hospitals and two research centers in 45 of Pennsylvania’s 67 counties, serving more than 3 million of the state’s residents.
Patients’ residential addresses were used to identify latitude and longitude coordinates that were matched with 9,669 UNGD wells in Pennsylvania and the location of major and minor roadways. The researchers also calculated a measure of community socioeconomic deprivation.
The adjusted odds of hospitalization were higher for patients in the highest quartile of exposure for three of the four UNGD phases: pad preparation (odds ratio, 1.70; 95% confidence interval, 1.35-2.13), stimulation or the actual fracking (OR, 1.80; 95% CI, 1.35-2.40), and production (OR, 1.62; 95% CI, 1.07-2.45).
Dr. McAlexander said she initially thought the lack of association with drilling (OR, 0.97; 95% CI, 0.75-1.27) was a mistake but noted that the drilling metric reflects a shorter time period than, for example, 30 days needed to clear the well pad and bring in the necessary equipment.
Stronger associations between pad preparation, fracking, and production are also consistent with the known increases in air pollution, traffic, and noise associated with these phases.
Individuals with more severe HF had greater odds of hospitalization, but the effect sizes were generally comparable between HF with preserved versus reduced ejection fraction. For those with the highest exposure to fracking, the odds ratios for hospitalization reached 2.25 (95% CI, 1.56-3.25) and 2.09 (95% CI, 1.44-3.03), respectively.
Notably, patients who could be phenotyped versus those who could not were more likely to die, to be hospitalized for HF, and to have a higher Charlson Comorbidity Index and other relevant diagnoses like myocardial infarction.
“Clinicians need to be increasingly aware that the environments their patients are in are a huge factor in their disease progression and outlook,” McAlexander said. “We know that UNGD, specifically now, is something that could be impacting a heart failure patient’s survival.”
She also suggested that the findings may also spur more advocacy work and “across-silo” collaboration between clinicians and environmental researchers.
Dr. Rajagopalan said there is increasing recognition that physicians need to be aware of environmental health links as extreme events like the California and Oregon wildfires and coastal flooding become increasingly common. “Unfortunately, unconventional is becoming the new convention.”
The problem for many physicians, however, is just having enough bandwidth to get through the day and get enough learning to keep above water, he said. Artificial intelligence could be used to seed electronic medical records with other personalized information from a bevy of sources including smartphones and the internet of things, but fundamental changes are also needed in the educational process to emphasize the environment.
“It’s going to take a huge societal shift in the way we view commodities, what we consider healthy, etc, but it can happen very quickly because all it takes is a crisis like COVID-19 to bring people to their knees and make them understand how this is going to take over our lives over the next decade,” Dr. Rajagopalan said.
The scientific community has been calling for “good” epidemiologic studies on the health effects of fracking since the early 2010s, Barrak Alahmad, MBChB, MPH, Harvard T.H. Chan School of Public Health, and Haitham Khraishah, MD, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, point out in an accompanying editorial.
The current study applied “extensive and rigorous methods” involving both the design and statistical approach, including use of a negative control analysis to assess for sources of spurious causal inference, several sensitivity analyses, and controlled for a wide range of covariates.
“Their results were consistent and robust across all these measures,” the editorialists wrote. “Most importantly, the effect size is probably too large to be explained away by an unmeasured confounder.”
Dr. Alahmad and Dr. Khraishah call for advancements in exposure assessment, citing a recent study reporting that ambient particle radioactivity near unconventional oil and gas sites could induce adverse health effects. Other unmet needs include a better understanding of racial disparities in the impacts of fracking and a fine-tuning of cause-specific cardiovascular morbidity and mortality.
The study was supported by training grants from the National Institute of Environmental Health Sciences to Dr. McAlexander and principal investigator Brian Schwartz, MD. The authors, Dr. Rajagopalan, Dr. Alahmad, and Dr. Khraishah have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
COVID-19 and risk of clotting: ‘Be proactive about prevention’
The risk of arterial and venous thrombosis in patients with COVID-19 has been a major issue throughout the pandemic, and how best to manage this risk is the subject of a new review article.
The article, by Gregory Dr. Piazza, MD, and David A. Morrow, MD, Brigham and Women’s Hospital, Boston, was published online in JAMA on Nov. 23.
“Basically we’re saying: ‘Be proactive about prevention,’” Dr. Piazza told this news organization.
There is growing recognition among those on the frontline that there is an increased risk of thrombosis in COVID-19 patients, Dr. Piazza said. The risk is highest in patients in the intensive care unit, but the risk is also increased in patients hospitalized with COVID-19, even those not in ICU.
“We don’t really know what the risk is in nonhospitalized COVID-19 patients, but we think it’s much lower than in those who are hospitalized,” he said. “We are waiting for data on the optimal way of managing this increased risk of thrombosis in COVID patients, but for the time being, we believe a systematic way of addressing this risk is best, with every patient hospitalized with COVID-19 receiving some type of thromboprophylaxis. This would mainly be with anticoagulation, but in patients in whom anticoagulation is contraindicated, then mechanical methods could be used, such as pneumatic compression boots or compression stockings.”
The authors report thrombotic complication rates of 2.6% in noncritically ill hospitalized patients with COVID-19 and 35.3% in critically ill patients from a recent U.S. registry study.
Autopsy findings of microthrombi in multiple organ systems, including the lungs, heart, and kidneys, suggest that thrombosis may contribute to multisystem organ dysfunction in severe COVID-19, they note. Although the pathophysiology is not fully defined, prothrombotic abnormalities have been identified in patients with COVID-19, including elevated levels of D-dimer, fibrinogen, and factor VIII, they add.
“There are several major questions about which COVID-19 patients to treat with thromboprophylaxis, how to treat them in term of levels of anticoagulation, and there are many ongoing clinical trials to try and answer these questions,” Dr. Piazza commented. “We need results from these randomized trials to provide a better compass for COVID-19 patients at risk of clotting.”
At present, clinicians can follow two different sets of guidelines on the issue, one from the American College of Chest Physicians and the other from the International Society on Thrombosis and Hemostasis, the authors note.
“The ACCP guidelines are very conservative and basically follow the evidence base for medical patients, while the ISTH guidelines are more aggressive and recommend increased levels of anticoagulation in both ICU and hospitalized non-ICU patients and also extend prophylaxis after discharge,” Dr. Piazza said.
“There is quite a difference between the two sets of guidelines, which can be a point of confusion,” he added.
Dr. Piazza notes that at his center every hospitalized COVID patient who does not have a contraindication to anticoagulation receives a standard prophylactic dose of a once-daily low-molecular-weight heparin (for example, enoxaparin 40 mg). A once-daily product is used to minimize infection risk to staff.
While all COVID patients in the ICU should automatically receive some anticoagulation, the optimal dose is an area of active investigation, he explained. “There were several early reports of ICU patients developing blood clots despite receiving standard thromboprophylaxis so perhaps we need to use higher doses. There are trials underway looking at this, and we would advise enrolling patients into these trials.”
If patients can’t be enrolled into trials, and clinicians feel higher anticoagulation levels are needed, Dr. Piazza advises following the ISTH guidance, which allows an intermediate dose of low-molecular-weight heparin (up to 1 mg/kg enoxaparin).
“Some experts are suggesting even higher doses may be needed in some ICU patients, such as the full therapeutic dose, but I worry about the risk of bleeding with such a strategy,” he said.
Dr. Piazza says they do not routinely give anticoagulation after discharge, but if this is desired then patients could be switched to an oral agent, and some of the direct-acting oral anticoagulants are approved for prophylactic use in medically ill patients.
Dr. Piazza points out that whether thromboprophylaxis should be used for nonhospitalized COVID patients who have risk factors for clotting such as a prior history of thrombosis or obesity is a pressing question, and he encourages clinicians to enroll these patients in clinical trials evaluating this issue, such as the PREVENT-HD trial.
“If they can’t enroll patents in a trial, then they have to make a decision whether the patient is high-enough risk to justify off-label use of anticoagulant. There is a case to be made for this, but there is no evidence for or against such action at present,” he noted.
At this time, neither the ISTH nor ACCP recommend measuring D-dimer to screen for venous thromboembolism or to determine intensity of prophylaxis or treatment, the authors note.
“Ongoing investigation will determine optimal preventive regimens in COVID-19 in the intensive care unit, at hospital discharge, and in nonhospitalized patients at high risk for thrombosis,” they conclude.
Dr. Piazza reported grants from Bayer, Bristol Myers Squibb, Boston Scientific, Janssen, and Portola, and personal fees from Agile, Amgen, Pfizer, and the Prairie Education and Research Cooperative outside the submitted work. Dr. Morrow reported grants from Abbott Laboratories, Amgen, Anthos Therapeutics, Esai, GlaxoSmithKline, Takeda, and The Medicines Company; grants and personal fees from AstraZeneca, Merck, Novartis, and Roche Diagnostics; and personal fees from Bayer Pharma and InCarda outside the submitted work.
A version of this article originally appeared on Medscape.com.
The risk of arterial and venous thrombosis in patients with COVID-19 has been a major issue throughout the pandemic, and how best to manage this risk is the subject of a new review article.
The article, by Gregory Dr. Piazza, MD, and David A. Morrow, MD, Brigham and Women’s Hospital, Boston, was published online in JAMA on Nov. 23.
“Basically we’re saying: ‘Be proactive about prevention,’” Dr. Piazza told this news organization.
There is growing recognition among those on the frontline that there is an increased risk of thrombosis in COVID-19 patients, Dr. Piazza said. The risk is highest in patients in the intensive care unit, but the risk is also increased in patients hospitalized with COVID-19, even those not in ICU.
“We don’t really know what the risk is in nonhospitalized COVID-19 patients, but we think it’s much lower than in those who are hospitalized,” he said. “We are waiting for data on the optimal way of managing this increased risk of thrombosis in COVID patients, but for the time being, we believe a systematic way of addressing this risk is best, with every patient hospitalized with COVID-19 receiving some type of thromboprophylaxis. This would mainly be with anticoagulation, but in patients in whom anticoagulation is contraindicated, then mechanical methods could be used, such as pneumatic compression boots or compression stockings.”
The authors report thrombotic complication rates of 2.6% in noncritically ill hospitalized patients with COVID-19 and 35.3% in critically ill patients from a recent U.S. registry study.
Autopsy findings of microthrombi in multiple organ systems, including the lungs, heart, and kidneys, suggest that thrombosis may contribute to multisystem organ dysfunction in severe COVID-19, they note. Although the pathophysiology is not fully defined, prothrombotic abnormalities have been identified in patients with COVID-19, including elevated levels of D-dimer, fibrinogen, and factor VIII, they add.
“There are several major questions about which COVID-19 patients to treat with thromboprophylaxis, how to treat them in term of levels of anticoagulation, and there are many ongoing clinical trials to try and answer these questions,” Dr. Piazza commented. “We need results from these randomized trials to provide a better compass for COVID-19 patients at risk of clotting.”
At present, clinicians can follow two different sets of guidelines on the issue, one from the American College of Chest Physicians and the other from the International Society on Thrombosis and Hemostasis, the authors note.
“The ACCP guidelines are very conservative and basically follow the evidence base for medical patients, while the ISTH guidelines are more aggressive and recommend increased levels of anticoagulation in both ICU and hospitalized non-ICU patients and also extend prophylaxis after discharge,” Dr. Piazza said.
“There is quite a difference between the two sets of guidelines, which can be a point of confusion,” he added.
Dr. Piazza notes that at his center every hospitalized COVID patient who does not have a contraindication to anticoagulation receives a standard prophylactic dose of a once-daily low-molecular-weight heparin (for example, enoxaparin 40 mg). A once-daily product is used to minimize infection risk to staff.
While all COVID patients in the ICU should automatically receive some anticoagulation, the optimal dose is an area of active investigation, he explained. “There were several early reports of ICU patients developing blood clots despite receiving standard thromboprophylaxis so perhaps we need to use higher doses. There are trials underway looking at this, and we would advise enrolling patients into these trials.”
If patients can’t be enrolled into trials, and clinicians feel higher anticoagulation levels are needed, Dr. Piazza advises following the ISTH guidance, which allows an intermediate dose of low-molecular-weight heparin (up to 1 mg/kg enoxaparin).
“Some experts are suggesting even higher doses may be needed in some ICU patients, such as the full therapeutic dose, but I worry about the risk of bleeding with such a strategy,” he said.
Dr. Piazza says they do not routinely give anticoagulation after discharge, but if this is desired then patients could be switched to an oral agent, and some of the direct-acting oral anticoagulants are approved for prophylactic use in medically ill patients.
Dr. Piazza points out that whether thromboprophylaxis should be used for nonhospitalized COVID patients who have risk factors for clotting such as a prior history of thrombosis or obesity is a pressing question, and he encourages clinicians to enroll these patients in clinical trials evaluating this issue, such as the PREVENT-HD trial.
“If they can’t enroll patents in a trial, then they have to make a decision whether the patient is high-enough risk to justify off-label use of anticoagulant. There is a case to be made for this, but there is no evidence for or against such action at present,” he noted.
At this time, neither the ISTH nor ACCP recommend measuring D-dimer to screen for venous thromboembolism or to determine intensity of prophylaxis or treatment, the authors note.
“Ongoing investigation will determine optimal preventive regimens in COVID-19 in the intensive care unit, at hospital discharge, and in nonhospitalized patients at high risk for thrombosis,” they conclude.
Dr. Piazza reported grants from Bayer, Bristol Myers Squibb, Boston Scientific, Janssen, and Portola, and personal fees from Agile, Amgen, Pfizer, and the Prairie Education and Research Cooperative outside the submitted work. Dr. Morrow reported grants from Abbott Laboratories, Amgen, Anthos Therapeutics, Esai, GlaxoSmithKline, Takeda, and The Medicines Company; grants and personal fees from AstraZeneca, Merck, Novartis, and Roche Diagnostics; and personal fees from Bayer Pharma and InCarda outside the submitted work.
A version of this article originally appeared on Medscape.com.
The risk of arterial and venous thrombosis in patients with COVID-19 has been a major issue throughout the pandemic, and how best to manage this risk is the subject of a new review article.
The article, by Gregory Dr. Piazza, MD, and David A. Morrow, MD, Brigham and Women’s Hospital, Boston, was published online in JAMA on Nov. 23.
“Basically we’re saying: ‘Be proactive about prevention,’” Dr. Piazza told this news organization.
There is growing recognition among those on the frontline that there is an increased risk of thrombosis in COVID-19 patients, Dr. Piazza said. The risk is highest in patients in the intensive care unit, but the risk is also increased in patients hospitalized with COVID-19, even those not in ICU.
“We don’t really know what the risk is in nonhospitalized COVID-19 patients, but we think it’s much lower than in those who are hospitalized,” he said. “We are waiting for data on the optimal way of managing this increased risk of thrombosis in COVID patients, but for the time being, we believe a systematic way of addressing this risk is best, with every patient hospitalized with COVID-19 receiving some type of thromboprophylaxis. This would mainly be with anticoagulation, but in patients in whom anticoagulation is contraindicated, then mechanical methods could be used, such as pneumatic compression boots or compression stockings.”
The authors report thrombotic complication rates of 2.6% in noncritically ill hospitalized patients with COVID-19 and 35.3% in critically ill patients from a recent U.S. registry study.
Autopsy findings of microthrombi in multiple organ systems, including the lungs, heart, and kidneys, suggest that thrombosis may contribute to multisystem organ dysfunction in severe COVID-19, they note. Although the pathophysiology is not fully defined, prothrombotic abnormalities have been identified in patients with COVID-19, including elevated levels of D-dimer, fibrinogen, and factor VIII, they add.
“There are several major questions about which COVID-19 patients to treat with thromboprophylaxis, how to treat them in term of levels of anticoagulation, and there are many ongoing clinical trials to try and answer these questions,” Dr. Piazza commented. “We need results from these randomized trials to provide a better compass for COVID-19 patients at risk of clotting.”
At present, clinicians can follow two different sets of guidelines on the issue, one from the American College of Chest Physicians and the other from the International Society on Thrombosis and Hemostasis, the authors note.
“The ACCP guidelines are very conservative and basically follow the evidence base for medical patients, while the ISTH guidelines are more aggressive and recommend increased levels of anticoagulation in both ICU and hospitalized non-ICU patients and also extend prophylaxis after discharge,” Dr. Piazza said.
“There is quite a difference between the two sets of guidelines, which can be a point of confusion,” he added.
Dr. Piazza notes that at his center every hospitalized COVID patient who does not have a contraindication to anticoagulation receives a standard prophylactic dose of a once-daily low-molecular-weight heparin (for example, enoxaparin 40 mg). A once-daily product is used to minimize infection risk to staff.
While all COVID patients in the ICU should automatically receive some anticoagulation, the optimal dose is an area of active investigation, he explained. “There were several early reports of ICU patients developing blood clots despite receiving standard thromboprophylaxis so perhaps we need to use higher doses. There are trials underway looking at this, and we would advise enrolling patients into these trials.”
If patients can’t be enrolled into trials, and clinicians feel higher anticoagulation levels are needed, Dr. Piazza advises following the ISTH guidance, which allows an intermediate dose of low-molecular-weight heparin (up to 1 mg/kg enoxaparin).
“Some experts are suggesting even higher doses may be needed in some ICU patients, such as the full therapeutic dose, but I worry about the risk of bleeding with such a strategy,” he said.
Dr. Piazza says they do not routinely give anticoagulation after discharge, but if this is desired then patients could be switched to an oral agent, and some of the direct-acting oral anticoagulants are approved for prophylactic use in medically ill patients.
Dr. Piazza points out that whether thromboprophylaxis should be used for nonhospitalized COVID patients who have risk factors for clotting such as a prior history of thrombosis or obesity is a pressing question, and he encourages clinicians to enroll these patients in clinical trials evaluating this issue, such as the PREVENT-HD trial.
“If they can’t enroll patents in a trial, then they have to make a decision whether the patient is high-enough risk to justify off-label use of anticoagulant. There is a case to be made for this, but there is no evidence for or against such action at present,” he noted.
At this time, neither the ISTH nor ACCP recommend measuring D-dimer to screen for venous thromboembolism or to determine intensity of prophylaxis or treatment, the authors note.
“Ongoing investigation will determine optimal preventive regimens in COVID-19 in the intensive care unit, at hospital discharge, and in nonhospitalized patients at high risk for thrombosis,” they conclude.
Dr. Piazza reported grants from Bayer, Bristol Myers Squibb, Boston Scientific, Janssen, and Portola, and personal fees from Agile, Amgen, Pfizer, and the Prairie Education and Research Cooperative outside the submitted work. Dr. Morrow reported grants from Abbott Laboratories, Amgen, Anthos Therapeutics, Esai, GlaxoSmithKline, Takeda, and The Medicines Company; grants and personal fees from AstraZeneca, Merck, Novartis, and Roche Diagnostics; and personal fees from Bayer Pharma and InCarda outside the submitted work.
A version of this article originally appeared on Medscape.com.
Infant’s COVID-19–related myocardial injury reversed
Reports of signs of heart failure in adults with COVID-19 have been rare – just four such cases have been published since the outbreak started in China – and now a team of pediatric cardiologists in New York have reported a case of acute but reversible myocardial injury in an infant with COVID-19.
and right upper lobe atelectasis.
The 2-month-old infant went home after more than 2 weeks in the hospital with no apparent lingering cardiac effects of the illness and not needing any oral heart failure medications, Madhu Sharma, MD, of the Children’s Hospital and Montefiore in New York and colleagues reported in JACC Case Reports. With close follow-up, the child’s left ventricle size and systolic function have remained normal and mitral regurgitation resolved. The case report didn’t mention the infant’s gender.
But before the straightforward postdischarge course emerged, the infant was in a precarious state, and Dr. Sharma and her team were challenged to diagnose the underlying causes.
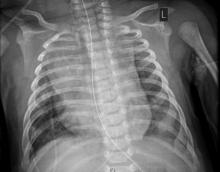
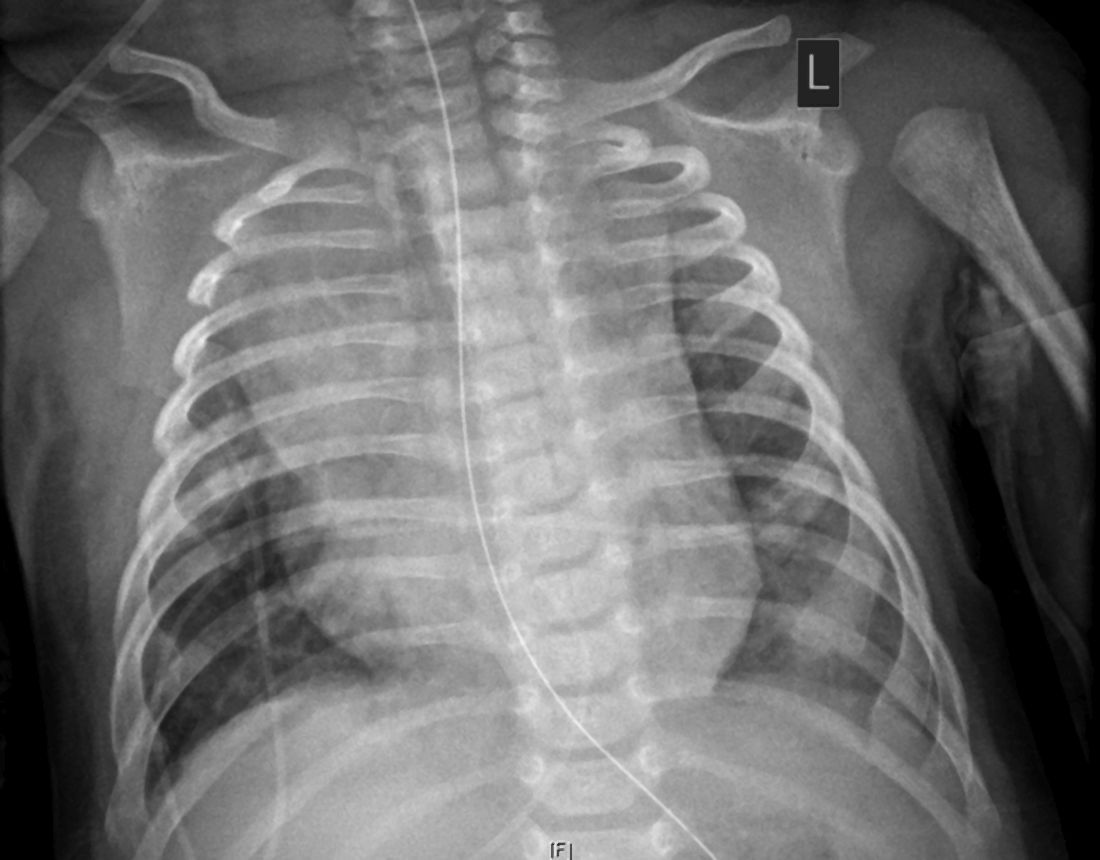
The child, who was born about 7 weeks premature, first came to the hospital having turned blue after choking on food. Nonrebreather mask ventilation was initiated in the ED, and an examination detected a holosystolic murmur. A test for COVID-19 was negative, but a later test was positive, and a chest x-ray exhibited cardiomegaly and signs of fluid and inflammation in the lungs.
An electrocardiogram detected sinus tachycardia, ST-segment depression and other anomalies in cardiac function. Further investigation with a transthoracic ECG showed severely depressed left ventricle systolic function with an ejection fraction of 30%, severe mitral regurgitation, and normal right ventricular systolic function.
Treatment included remdesivir and intravenous antibiotics. Through the hospital course, the patient was extubated to noninvasive ventilation, reintubated, put on intravenous steroid (methylprednisolone) and low-molecular-weight heparin, extubated, and tested throughout for cardiac function.
By day 14, left ventricle size and function normalized, and while the mitral regurgitation remained severe, it improved later without HF therapies. Left ventricle ejection fraction had recovered to 60%, and key cardiac biomarkers had normalized. On day 16, milrinone was discontinued, and the care team determined the patient no longer needed oral heart failure therapies.
“Most children with COVID-19 are either asymptomatic or have mild symptoms, but our case shows the potential for reversible myocardial injury in infants with COVID-19,” said Dr. Sharma. “Testing for COVID-19 in children presenting with signs and symptoms of heart failure is very important as we learn more about the impact of this virus.”
Dr. Sharma and coauthors have no relevant financial relationships to disclose.
SOURCE: Sharma M et al. JACC Case Rep. 2020. doi: 10.1016/j.jaccas.2020.09.031.
Reports of signs of heart failure in adults with COVID-19 have been rare – just four such cases have been published since the outbreak started in China – and now a team of pediatric cardiologists in New York have reported a case of acute but reversible myocardial injury in an infant with COVID-19.
and right upper lobe atelectasis.
The 2-month-old infant went home after more than 2 weeks in the hospital with no apparent lingering cardiac effects of the illness and not needing any oral heart failure medications, Madhu Sharma, MD, of the Children’s Hospital and Montefiore in New York and colleagues reported in JACC Case Reports. With close follow-up, the child’s left ventricle size and systolic function have remained normal and mitral regurgitation resolved. The case report didn’t mention the infant’s gender.
But before the straightforward postdischarge course emerged, the infant was in a precarious state, and Dr. Sharma and her team were challenged to diagnose the underlying causes.
The child, who was born about 7 weeks premature, first came to the hospital having turned blue after choking on food. Nonrebreather mask ventilation was initiated in the ED, and an examination detected a holosystolic murmur. A test for COVID-19 was negative, but a later test was positive, and a chest x-ray exhibited cardiomegaly and signs of fluid and inflammation in the lungs.
An electrocardiogram detected sinus tachycardia, ST-segment depression and other anomalies in cardiac function. Further investigation with a transthoracic ECG showed severely depressed left ventricle systolic function with an ejection fraction of 30%, severe mitral regurgitation, and normal right ventricular systolic function.
Treatment included remdesivir and intravenous antibiotics. Through the hospital course, the patient was extubated to noninvasive ventilation, reintubated, put on intravenous steroid (methylprednisolone) and low-molecular-weight heparin, extubated, and tested throughout for cardiac function.
By day 14, left ventricle size and function normalized, and while the mitral regurgitation remained severe, it improved later without HF therapies. Left ventricle ejection fraction had recovered to 60%, and key cardiac biomarkers had normalized. On day 16, milrinone was discontinued, and the care team determined the patient no longer needed oral heart failure therapies.
“Most children with COVID-19 are either asymptomatic or have mild symptoms, but our case shows the potential for reversible myocardial injury in infants with COVID-19,” said Dr. Sharma. “Testing for COVID-19 in children presenting with signs and symptoms of heart failure is very important as we learn more about the impact of this virus.”
Dr. Sharma and coauthors have no relevant financial relationships to disclose.
SOURCE: Sharma M et al. JACC Case Rep. 2020. doi: 10.1016/j.jaccas.2020.09.031.
Reports of signs of heart failure in adults with COVID-19 have been rare – just four such cases have been published since the outbreak started in China – and now a team of pediatric cardiologists in New York have reported a case of acute but reversible myocardial injury in an infant with COVID-19.
and right upper lobe atelectasis.
The 2-month-old infant went home after more than 2 weeks in the hospital with no apparent lingering cardiac effects of the illness and not needing any oral heart failure medications, Madhu Sharma, MD, of the Children’s Hospital and Montefiore in New York and colleagues reported in JACC Case Reports. With close follow-up, the child’s left ventricle size and systolic function have remained normal and mitral regurgitation resolved. The case report didn’t mention the infant’s gender.
But before the straightforward postdischarge course emerged, the infant was in a precarious state, and Dr. Sharma and her team were challenged to diagnose the underlying causes.
The child, who was born about 7 weeks premature, first came to the hospital having turned blue after choking on food. Nonrebreather mask ventilation was initiated in the ED, and an examination detected a holosystolic murmur. A test for COVID-19 was negative, but a later test was positive, and a chest x-ray exhibited cardiomegaly and signs of fluid and inflammation in the lungs.
An electrocardiogram detected sinus tachycardia, ST-segment depression and other anomalies in cardiac function. Further investigation with a transthoracic ECG showed severely depressed left ventricle systolic function with an ejection fraction of 30%, severe mitral regurgitation, and normal right ventricular systolic function.
Treatment included remdesivir and intravenous antibiotics. Through the hospital course, the patient was extubated to noninvasive ventilation, reintubated, put on intravenous steroid (methylprednisolone) and low-molecular-weight heparin, extubated, and tested throughout for cardiac function.
By day 14, left ventricle size and function normalized, and while the mitral regurgitation remained severe, it improved later without HF therapies. Left ventricle ejection fraction had recovered to 60%, and key cardiac biomarkers had normalized. On day 16, milrinone was discontinued, and the care team determined the patient no longer needed oral heart failure therapies.
“Most children with COVID-19 are either asymptomatic or have mild symptoms, but our case shows the potential for reversible myocardial injury in infants with COVID-19,” said Dr. Sharma. “Testing for COVID-19 in children presenting with signs and symptoms of heart failure is very important as we learn more about the impact of this virus.”
Dr. Sharma and coauthors have no relevant financial relationships to disclose.
SOURCE: Sharma M et al. JACC Case Rep. 2020. doi: 10.1016/j.jaccas.2020.09.031.
FROM JACC CASE REPORTS
Key clinical point: Children presenting with COVID-19 should be tested for heart failure.
Major finding: A 2-month-old infant with COVID-19 had acute but reversible myocardial injury.
Study details: Single case report.
Disclosures: Dr. Sharma, MD, has no relevant financial relationships to disclose.
Source: Sharma M et al. JACC Case Rep. 2020. doi: 10.1016/j.jaccas.2020.09.031.
Sedentary postmenopausal women have higher heart failure risk
The more time older women spent sitting or lying down, the more likely their risk of hospitalization for heart failure, based on data from more than 80,000 postmenopausal women.
The 2018 Physical Activity Guidelines show evidence of the impact of physical activity on reducing heart failure risk, but the association between activity, sedentary behavior (SB) and heart failure (HF) in older women in particular has not been well studied, wrote Michael J. LaMonte, PhD, MPH, of the State University of New York at Buffalo, and colleagues in a study published in Circulation: Heart Failure. “Given the high prevalence of prolonged sedentary time among U.S. adults aged 65 and older, among whom HF burden is substantial, understanding the role SB has in HF development is relevant to future HF prevention strategies,” the researchers wrote.
The researchers identified 80,982 women aged 50-79 years who were enrolled in the Women’s Health Initiative Observational Study, had no known HF, and could walk at least one block unassisted. The average follow-up period was 9 years, and a total of 1,402 women were hospitalized for heart failure during the period of time they were observed.
The time spent sedentary (combined sitting or lying down) was divided into tertiles of 6.5 hours or less, 6.6-9.5 hours, and more than 9.5 hours. Time spent sitting was divided into tertiles of 4.5 hours or less; 4.6-8.5 hours; and more than 8.5 hours.
Heart failure risk goes up with more down time
After controlling for multiple variables including age, race, education, income, smoking status alcohol use, menopausal hormone therapy, and hysterectomy status, the researchers found that patients in the second tertile for sedentary behavior had a significantly increased heart failure risk than patients in the first tertile for sedentary behavior. This risk was even greater for patients falling in the third tertile for sedentary behavior. Odds ratios were 1.00 (referent), 1.15, and 1.42 for the lowest to highest tertiles for total sedentary behavior, respectively, and 1.00 (referent), 1.14, and 1.54 for sitting (P < .001 for both total sedentary behavior and sitting only).
The trends remained significant after controlling for comorbidities including MI and coronary revascularization, and the associations were similar among categories of women with additional HF risk factors, including body mass index, diabetes, hypertension, and coronary heart disease.
Notably, the association between hours spent sitting or lying down and HF risk persisted even in women who met recommended activity levels, the researchers wrote.
The study findings were limited by the use of self-reports and by the inability to evaluate SB patterns or SB and HF subtypes, the researchers noted. However, the results were strengthened by the large sample size, use of time-varying SB exposure, and extensive controlling, and the data support the risk of increased SB on adverse cardiovascular outcomes.
“Results of this study underscore the need for effective strategies to reduce daily SB time, in addition to increasing recreational physical activity, as part of population efforts for HF prevention,” they concluded.
Clinicians know the value of a physically active lifestyle for heart health, said lead author Dr. LaMonte in a statement accompanying the study’s release. “However, our study clearly shows that we also need to increase efforts to reduce daily sedentary time and encourage adults to frequently interrupt their sedentary time. This does not necessarily require an extended bout of physical activity; it might simply be standing up for 5 minutes or standing and moving one’s feet in place.
“We do not have sufficient evidence on the best approach to recommend for interrupting sedentary time. However, accumulating data suggest that habitual activities such as steps taken during household and other activities of daily living are an important aspect of cardiovascular disease prevention and healthy aging,” Dr. LaMonte added.
Promote more movement and less sitting
“This is the first study to assess sedentary time and the risk for incident heart failure hospitalization in postmenopausal women,” said Robert H. Hopkins Jr., MD, of the University of Arkansas for Medical Sciences, Little Rock, in an interview.
“Heart failure is the cause of approximately 35% of cardiovascular mortalities in women, and sedentary behaviors are common in older adults,” he noted.
Kashif J. Piracha, MD, of Houston Methodist Willowbrook Hospital, agreed that there is a lack of existing data looking at the relationship between sedentary behavior and the risk of the development of heart failure in postmenopausal women. In an interview, he cited this as a reason “it was important to conduct this study.”
Dr. Hopkins added that he was not surprised by the study results “There are a number of studies which have demonstrated reduction in risk for heart failure in men and in combined populations of men and women with increased physical activity.” There are fewer data (but similar outcomes) in studies of men with increased levels of sedentary behaviors, he said.
“This study adds one more reason that other clinicians in primary care and me need to encourage our older patients to get up and move,” said Dr. Hopkins, who also serves on the editorial advisory board of Internal Medicine News. “Many of us have focused our efforts in the past on achieving exercise goals and this study provides a foundation for a recommendation that ‘it is not just about exercise;’ we need to also encourage our patients to minimize their time in sedentary pursuits in addition to exercise if we are to optimize their health into older age.”
Dr. Hopkins noted that the large size of the study was a strength, but the observational design and use of patient surveys were limitations.
“We need further studies to better tease out whether there are risk differences in different sedentary behavior patterns, whether this applies across heart failure with reduced ejection fraction versus heart failure with preserved ejection fraction, and whether there are additional ways we can mitigate these risks as our society ages,” he said.
Findings differ from California Men’s Health Study’s
“The results corroborate the fact that there is less risk of heart failure in physically active patients,” Dr. Piracha noted.
The message for clinicians is to encourage postmenopausal female patients to engage in physical activity as much as possible, said Dr. Piracha. “Also, it appears that in this population, even with good physical activity, prolonged sedentary behavior of more than 8.5 hours a day was still associated with a higher risk of incident HF hospitalization. Therefore, a case can be made to focus on carrying out physical activity with an intensity that can be sustained for longer, rather than shorter periods of time.”
Notably, the finding of increased HF hospitalization in women who reported high amounts of physical activity but were still sedentary for more than 8.5 hours a day “is contrary to what was seen in the California Men’s Health Study.” In that study, “men with high physical activity levels who also had prolonged sitting time did not have increased risk of HF hospitalization,” Dr. Piracha noted. “Further research is needed to elucidate what hormonal or other factors contribute to this difference.”
The new study was supported by the National Heart, Lung, and Blood Institute. The researchers had no financial conflicts to disclose. Dr. Hopkins and Dr. Piracha had no financial conflicts to disclose.
SOURCE: LaMonte MJ et al. Circ Heart Fail. 2020 Nov 24. doi: 10.1161/CIRCHEARTFAILURE.120.007508.
The more time older women spent sitting or lying down, the more likely their risk of hospitalization for heart failure, based on data from more than 80,000 postmenopausal women.
The 2018 Physical Activity Guidelines show evidence of the impact of physical activity on reducing heart failure risk, but the association between activity, sedentary behavior (SB) and heart failure (HF) in older women in particular has not been well studied, wrote Michael J. LaMonte, PhD, MPH, of the State University of New York at Buffalo, and colleagues in a study published in Circulation: Heart Failure. “Given the high prevalence of prolonged sedentary time among U.S. adults aged 65 and older, among whom HF burden is substantial, understanding the role SB has in HF development is relevant to future HF prevention strategies,” the researchers wrote.
The researchers identified 80,982 women aged 50-79 years who were enrolled in the Women’s Health Initiative Observational Study, had no known HF, and could walk at least one block unassisted. The average follow-up period was 9 years, and a total of 1,402 women were hospitalized for heart failure during the period of time they were observed.
The time spent sedentary (combined sitting or lying down) was divided into tertiles of 6.5 hours or less, 6.6-9.5 hours, and more than 9.5 hours. Time spent sitting was divided into tertiles of 4.5 hours or less; 4.6-8.5 hours; and more than 8.5 hours.
Heart failure risk goes up with more down time
After controlling for multiple variables including age, race, education, income, smoking status alcohol use, menopausal hormone therapy, and hysterectomy status, the researchers found that patients in the second tertile for sedentary behavior had a significantly increased heart failure risk than patients in the first tertile for sedentary behavior. This risk was even greater for patients falling in the third tertile for sedentary behavior. Odds ratios were 1.00 (referent), 1.15, and 1.42 for the lowest to highest tertiles for total sedentary behavior, respectively, and 1.00 (referent), 1.14, and 1.54 for sitting (P < .001 for both total sedentary behavior and sitting only).
The trends remained significant after controlling for comorbidities including MI and coronary revascularization, and the associations were similar among categories of women with additional HF risk factors, including body mass index, diabetes, hypertension, and coronary heart disease.
Notably, the association between hours spent sitting or lying down and HF risk persisted even in women who met recommended activity levels, the researchers wrote.
The study findings were limited by the use of self-reports and by the inability to evaluate SB patterns or SB and HF subtypes, the researchers noted. However, the results were strengthened by the large sample size, use of time-varying SB exposure, and extensive controlling, and the data support the risk of increased SB on adverse cardiovascular outcomes.
“Results of this study underscore the need for effective strategies to reduce daily SB time, in addition to increasing recreational physical activity, as part of population efforts for HF prevention,” they concluded.
Clinicians know the value of a physically active lifestyle for heart health, said lead author Dr. LaMonte in a statement accompanying the study’s release. “However, our study clearly shows that we also need to increase efforts to reduce daily sedentary time and encourage adults to frequently interrupt their sedentary time. This does not necessarily require an extended bout of physical activity; it might simply be standing up for 5 minutes or standing and moving one’s feet in place.
“We do not have sufficient evidence on the best approach to recommend for interrupting sedentary time. However, accumulating data suggest that habitual activities such as steps taken during household and other activities of daily living are an important aspect of cardiovascular disease prevention and healthy aging,” Dr. LaMonte added.
Promote more movement and less sitting
“This is the first study to assess sedentary time and the risk for incident heart failure hospitalization in postmenopausal women,” said Robert H. Hopkins Jr., MD, of the University of Arkansas for Medical Sciences, Little Rock, in an interview.
“Heart failure is the cause of approximately 35% of cardiovascular mortalities in women, and sedentary behaviors are common in older adults,” he noted.
Kashif J. Piracha, MD, of Houston Methodist Willowbrook Hospital, agreed that there is a lack of existing data looking at the relationship between sedentary behavior and the risk of the development of heart failure in postmenopausal women. In an interview, he cited this as a reason “it was important to conduct this study.”
Dr. Hopkins added that he was not surprised by the study results “There are a number of studies which have demonstrated reduction in risk for heart failure in men and in combined populations of men and women with increased physical activity.” There are fewer data (but similar outcomes) in studies of men with increased levels of sedentary behaviors, he said.
“This study adds one more reason that other clinicians in primary care and me need to encourage our older patients to get up and move,” said Dr. Hopkins, who also serves on the editorial advisory board of Internal Medicine News. “Many of us have focused our efforts in the past on achieving exercise goals and this study provides a foundation for a recommendation that ‘it is not just about exercise;’ we need to also encourage our patients to minimize their time in sedentary pursuits in addition to exercise if we are to optimize their health into older age.”
Dr. Hopkins noted that the large size of the study was a strength, but the observational design and use of patient surveys were limitations.
“We need further studies to better tease out whether there are risk differences in different sedentary behavior patterns, whether this applies across heart failure with reduced ejection fraction versus heart failure with preserved ejection fraction, and whether there are additional ways we can mitigate these risks as our society ages,” he said.
Findings differ from California Men’s Health Study’s
“The results corroborate the fact that there is less risk of heart failure in physically active patients,” Dr. Piracha noted.
The message for clinicians is to encourage postmenopausal female patients to engage in physical activity as much as possible, said Dr. Piracha. “Also, it appears that in this population, even with good physical activity, prolonged sedentary behavior of more than 8.5 hours a day was still associated with a higher risk of incident HF hospitalization. Therefore, a case can be made to focus on carrying out physical activity with an intensity that can be sustained for longer, rather than shorter periods of time.”
Notably, the finding of increased HF hospitalization in women who reported high amounts of physical activity but were still sedentary for more than 8.5 hours a day “is contrary to what was seen in the California Men’s Health Study.” In that study, “men with high physical activity levels who also had prolonged sitting time did not have increased risk of HF hospitalization,” Dr. Piracha noted. “Further research is needed to elucidate what hormonal or other factors contribute to this difference.”
The new study was supported by the National Heart, Lung, and Blood Institute. The researchers had no financial conflicts to disclose. Dr. Hopkins and Dr. Piracha had no financial conflicts to disclose.
SOURCE: LaMonte MJ et al. Circ Heart Fail. 2020 Nov 24. doi: 10.1161/CIRCHEARTFAILURE.120.007508.
The more time older women spent sitting or lying down, the more likely their risk of hospitalization for heart failure, based on data from more than 80,000 postmenopausal women.
The 2018 Physical Activity Guidelines show evidence of the impact of physical activity on reducing heart failure risk, but the association between activity, sedentary behavior (SB) and heart failure (HF) in older women in particular has not been well studied, wrote Michael J. LaMonte, PhD, MPH, of the State University of New York at Buffalo, and colleagues in a study published in Circulation: Heart Failure. “Given the high prevalence of prolonged sedentary time among U.S. adults aged 65 and older, among whom HF burden is substantial, understanding the role SB has in HF development is relevant to future HF prevention strategies,” the researchers wrote.
The researchers identified 80,982 women aged 50-79 years who were enrolled in the Women’s Health Initiative Observational Study, had no known HF, and could walk at least one block unassisted. The average follow-up period was 9 years, and a total of 1,402 women were hospitalized for heart failure during the period of time they were observed.
The time spent sedentary (combined sitting or lying down) was divided into tertiles of 6.5 hours or less, 6.6-9.5 hours, and more than 9.5 hours. Time spent sitting was divided into tertiles of 4.5 hours or less; 4.6-8.5 hours; and more than 8.5 hours.
Heart failure risk goes up with more down time
After controlling for multiple variables including age, race, education, income, smoking status alcohol use, menopausal hormone therapy, and hysterectomy status, the researchers found that patients in the second tertile for sedentary behavior had a significantly increased heart failure risk than patients in the first tertile for sedentary behavior. This risk was even greater for patients falling in the third tertile for sedentary behavior. Odds ratios were 1.00 (referent), 1.15, and 1.42 for the lowest to highest tertiles for total sedentary behavior, respectively, and 1.00 (referent), 1.14, and 1.54 for sitting (P < .001 for both total sedentary behavior and sitting only).
The trends remained significant after controlling for comorbidities including MI and coronary revascularization, and the associations were similar among categories of women with additional HF risk factors, including body mass index, diabetes, hypertension, and coronary heart disease.
Notably, the association between hours spent sitting or lying down and HF risk persisted even in women who met recommended activity levels, the researchers wrote.
The study findings were limited by the use of self-reports and by the inability to evaluate SB patterns or SB and HF subtypes, the researchers noted. However, the results were strengthened by the large sample size, use of time-varying SB exposure, and extensive controlling, and the data support the risk of increased SB on adverse cardiovascular outcomes.
“Results of this study underscore the need for effective strategies to reduce daily SB time, in addition to increasing recreational physical activity, as part of population efforts for HF prevention,” they concluded.
Clinicians know the value of a physically active lifestyle for heart health, said lead author Dr. LaMonte in a statement accompanying the study’s release. “However, our study clearly shows that we also need to increase efforts to reduce daily sedentary time and encourage adults to frequently interrupt their sedentary time. This does not necessarily require an extended bout of physical activity; it might simply be standing up for 5 minutes or standing and moving one’s feet in place.
“We do not have sufficient evidence on the best approach to recommend for interrupting sedentary time. However, accumulating data suggest that habitual activities such as steps taken during household and other activities of daily living are an important aspect of cardiovascular disease prevention and healthy aging,” Dr. LaMonte added.
Promote more movement and less sitting
“This is the first study to assess sedentary time and the risk for incident heart failure hospitalization in postmenopausal women,” said Robert H. Hopkins Jr., MD, of the University of Arkansas for Medical Sciences, Little Rock, in an interview.
“Heart failure is the cause of approximately 35% of cardiovascular mortalities in women, and sedentary behaviors are common in older adults,” he noted.
Kashif J. Piracha, MD, of Houston Methodist Willowbrook Hospital, agreed that there is a lack of existing data looking at the relationship between sedentary behavior and the risk of the development of heart failure in postmenopausal women. In an interview, he cited this as a reason “it was important to conduct this study.”
Dr. Hopkins added that he was not surprised by the study results “There are a number of studies which have demonstrated reduction in risk for heart failure in men and in combined populations of men and women with increased physical activity.” There are fewer data (but similar outcomes) in studies of men with increased levels of sedentary behaviors, he said.
“This study adds one more reason that other clinicians in primary care and me need to encourage our older patients to get up and move,” said Dr. Hopkins, who also serves on the editorial advisory board of Internal Medicine News. “Many of us have focused our efforts in the past on achieving exercise goals and this study provides a foundation for a recommendation that ‘it is not just about exercise;’ we need to also encourage our patients to minimize their time in sedentary pursuits in addition to exercise if we are to optimize their health into older age.”
Dr. Hopkins noted that the large size of the study was a strength, but the observational design and use of patient surveys were limitations.
“We need further studies to better tease out whether there are risk differences in different sedentary behavior patterns, whether this applies across heart failure with reduced ejection fraction versus heart failure with preserved ejection fraction, and whether there are additional ways we can mitigate these risks as our society ages,” he said.
Findings differ from California Men’s Health Study’s
“The results corroborate the fact that there is less risk of heart failure in physically active patients,” Dr. Piracha noted.
The message for clinicians is to encourage postmenopausal female patients to engage in physical activity as much as possible, said Dr. Piracha. “Also, it appears that in this population, even with good physical activity, prolonged sedentary behavior of more than 8.5 hours a day was still associated with a higher risk of incident HF hospitalization. Therefore, a case can be made to focus on carrying out physical activity with an intensity that can be sustained for longer, rather than shorter periods of time.”
Notably, the finding of increased HF hospitalization in women who reported high amounts of physical activity but were still sedentary for more than 8.5 hours a day “is contrary to what was seen in the California Men’s Health Study.” In that study, “men with high physical activity levels who also had prolonged sitting time did not have increased risk of HF hospitalization,” Dr. Piracha noted. “Further research is needed to elucidate what hormonal or other factors contribute to this difference.”
The new study was supported by the National Heart, Lung, and Blood Institute. The researchers had no financial conflicts to disclose. Dr. Hopkins and Dr. Piracha had no financial conflicts to disclose.
SOURCE: LaMonte MJ et al. Circ Heart Fail. 2020 Nov 24. doi: 10.1161/CIRCHEARTFAILURE.120.007508.
FROM CIRCULATION: HEART FAILURE
Finerenone’s heart benefits hold up in T2D patients without CVD
Finerenone, the first nonsteroidal mineralocorticoid receptor antagonist to complete a phase 3 trial, showed cardiovascular benefits in patients with type 2 diabetes and chronic kidney disease, regardless of whether they entered the study with a history of cardiovascular disease, in follow-up analyses of the FIDELIO-DKD trial, which included 5,674 patients.
“Finerenone demonstrated benefits for primary and secondary cardiovascular disease protection,” said Gerasimos Filippatos, MD, at the American Heart Association scientific sessions. Finerenone treatment cut the rate of cardiovascular death, nonfatal MI or stroke, or heart failure hospitalization, when compared with placebo, by a relative 15% among patients with a history of cardiovascular disease (CVD), and by a relative 14% in patients without this history, differences that met a statistical test for consistency. But the absolute, drug-associated increments in benefit over placebo differed between the two CVD subgroups because of a sharp underlying difference in event rates.
In contrast, the analyses reported by Dr. Filippatos and associates from the FIDELIO-DKD study showed significant heterogeneity based on the presence or absence of CVD for the study’s primary endpoint, a composite renal metric that tallied the combined rate of death from renal causes, renal failure, or a sustained drop in estimated glomerular filtration rate of at least 40%. Researchers enrolled patients into FIDELIO-DKD based on having type 2 diabetes (T2D) and chronic kidney disease (CKD). The prevalence of a history of CVD was 46%.
Among patients with a history of CVD, the composite adverse CVD outcome occurred at a rate of 8.5/100 patient-years in patients on placebo and in 7.18/100 patients years among those on finerenone during a median of 2.6 years of follow-up, a 1.32/100–patient-year absolute between-group difference. Among patients in a primary prevention setting, incident CVD event rates during follow-up were roughly half that in the secondary prevention patients. The upshot was that, in the placebo group, the rate was 3.92/100 patient- years, and in those on finerenone was 3.43/100 patient-years, a 0.49/100–patient-year absolute difference.
CVD history produced heterogeneity for the primary endpoint
In the analysis that focused on the study’s primary, renal endpoint, among patients identified as having CVD at study entry, the outcome occurred at a rate of 9.06/100 patient-years in the placebo subgroup and at a rate of 6.6/100 patient years in those who received finerenone, a significant 30% relative risk reduction and an absolute between-group difference of 2.46/100 patient-years.
In contrast, among patients without a CVD history, the composite renal endpoint occurred at a rate of 9.1/100 patient-years in the placebo patients and 8.42/100 patient-years in those on finerenone, a 6% relative risk reduction that was not significant, and a 0.68/100–patient-year absolute difference. This disparity in the primary event rate between the two treatment arms reached statistical significance (P = .016), the investigators reported in the published version of the report in Circulation that simultaneously appeared online.
“The totality of evidence suggests that finerenone could be used in patients with T2D with or without a history of CVD,” explained Dr. Filippatos in an interview. “The P-interaction for the composite kidney outcome is significant, but it is not corrected for multiple testing; therefore, it might be a false-chance finding and must be interpreted cautiously.
Furthermore, in another prespecified kidney composite outcome the results were consistent in patients with and without a history of CVD. In sum, all the FIDELIO-DKD analyses so far are “suggestive of a beneficial effect in patients without a history of CVD.”
Despite these patients receiving guideline directed therapies, “there remains a high unmet medical need in patients with T2D and CKD,” added Dr. Filippatos, professor of cardiology at the University of Athens. “We use multiple treatments for patients with heart failure, and we should use the same mindset for treating patients with T2D and CKD. The costs of dialysis and kidney transplant are very high, so it is important to consider options that slow progression of CKD in these patients.”
In FIDELIO-DKD, virtually all patients were on background therapy with a renin-angiotensin-system (RAS) inhibitor, so the trial’s results suggest that treatment should at least involve dual therapy with finerenone and a RAS inhibitor. Fewer than 5% were on background therapy with a sodium-glucose cotransporter 2 (SGLT2) inhibitor, a drug class recently established as another key agent for treating CKD in patients with T2D, setting up the prospect for triple therapy, although this approach has not yet undergone prospective testing.
Combining RAS inhibition, finerenone, and an SGLT2 inhibitor is “potentially a marriage made in diabetes heaven,” commented Deepak L. Bhatt, MD, a professor of medicine at Harvard Medical School, Boston, who has not participated in finerenone studies.
Finerenone looks better for safety
Regardless of subgroup analyses based on history of CVD, the findings from all patients enrolled in FIDELIO-DKD were positive for the both the primary renal outcome and key secondary outcome of composite CVD events. In the total randomized cohort, treatment with finerenone on top of optimized treatment with an ACE inhibitor or angiotensin receptor blocker (RAS inhibition) led to a significant 18% relative risk reduction, compared with placebo, for the primary renal endpoint, and a significant 14% relative drop in the key secondary CVD outcome. Those results were published in October in the New England Journal of Medicine.
For treating patients with T2D and CKD ,finerenone overall “looks like a major advance,” Dr. Bhatt said in an interview.
In addition to the positive efficacy results, several experts also focused on what they saw as superior safety of finerenone in the trial, compared with the historical safety of the steroidal mineralocorticoid receptor antagonists (MRAs) now in use: spironolactone and eplerenone.
“I’m a big believer in spironolactone, but it has issues with side effects, and eplerenone never seemed to catch on,” said Dr. Bhatt, who is also executive director of interventional cardiovascular programs at Brigham and Women’s Hospital in Boston.
“A lot of physicians like these MRAs, but acknowledge that side effects have kept these drugs from being used to the extent they should.” The existing MRAs, especially spironolactone, have become a key drug class for treating heart failure with reduced ejection fraction (and, some claim, for also treating heart failure with preserved ejection fraction), as well as treatment-resistant hypertension and primary aldosteronism. By design, FIDELIO-DKD did not enroll patients with heart failure because treatment with an MRA is indicated for those with heart failure with reduced ejection fraction.
The spironolactone adverse effect that generates the greatest concern is hyperkalemia. During his discussion of FIDELIO-DKD as designated discussant, Christoph Wanner, MD, noted a recent study in which the incidence of hyperkalemia severe enough to cause study discontinuation was 23% among patients treated with spironolactone for heart failure, which contrasts with the 2.3% rate in FIDELIO-DKD among finerenone recipients. This hyperkalemia incidence from finerenone also improved on the historical performance of other drugs, like aliskiren (Tekturna), said Dr. Wanner, professor and head of nephrology at the University of Würzburg (Germany).
The FIDELIO-DKD results place finerenone alongside the RAS- and SGLT2-inhibitor drug classes as appropriate treatments for most patients with T2D and CKD. “We have entered a new era of effective treatment for diabetic kidney disease,” Dr. Wanner declared.
“The overall safety profile of finerenone looked better, including hyperkalemia,” said Dr. Bhatt. “Hyperkalemia with spironolactone is not necessarily as bad as the perception. With careful monitoring of spironolactone, the hyperkalemia is manageable. But the perception is that it’s bad, and along with gynecomastia it’s a real killer.”
While some dismiss gynecomastia as a major concern (for men) with spironolactone treatment, “if medical students learn one thing about spironolactone, it’s that it can cause gynecomastia,” adding to the negative image that the approved MRAs carry, Dr. Bhatt said.
“The hyperkalemia was manageable. This is very important because of past problems with potassium when using spironolactone,” Dr. Filippatos said. Finerenone also looks “more cardiorenal protective” than the steroidal MRAs, exerting renal benefits in FIDELIO-DKD never previously described for a steroidal MRA.
Some of the uncertainty about the efficacy of finerenone in patients with a history of cardiovascular disease will lift when results are available in about another year from the FIGARO-DKD pivotal trial of finerenone, which enrolled more than 7,000 patients with T2D and CKD (entry criteria very similar to FIDELIO-CKD). A big difference is that FIGARO-DKD has a composite CVD event metric as its primary endpoint, and includes hospitalization for heart failure as one facet of the composite.
FIDELIO-DKD was sponsored by Bayer. Dr. Filippatos has been a lecturer on behalf of, served as a researcher for, or both for Bayer and also for Amgen, Boehringer Ingelheim, Medtronic, Novartis, Servier, and Vifor. Dr. Bhatt has received research funding from Bayer and also from several other companies, and he also is an adviser to several companies. Dr. Wanner has received honoraria from Bayer, and also from AstraZeneca, Boehringer Ingelheim, FMC, Gilead, GlaxoSmithKline, Lilly, and Merck.
Finerenone, the first nonsteroidal mineralocorticoid receptor antagonist to complete a phase 3 trial, showed cardiovascular benefits in patients with type 2 diabetes and chronic kidney disease, regardless of whether they entered the study with a history of cardiovascular disease, in follow-up analyses of the FIDELIO-DKD trial, which included 5,674 patients.
“Finerenone demonstrated benefits for primary and secondary cardiovascular disease protection,” said Gerasimos Filippatos, MD, at the American Heart Association scientific sessions. Finerenone treatment cut the rate of cardiovascular death, nonfatal MI or stroke, or heart failure hospitalization, when compared with placebo, by a relative 15% among patients with a history of cardiovascular disease (CVD), and by a relative 14% in patients without this history, differences that met a statistical test for consistency. But the absolute, drug-associated increments in benefit over placebo differed between the two CVD subgroups because of a sharp underlying difference in event rates.
In contrast, the analyses reported by Dr. Filippatos and associates from the FIDELIO-DKD study showed significant heterogeneity based on the presence or absence of CVD for the study’s primary endpoint, a composite renal metric that tallied the combined rate of death from renal causes, renal failure, or a sustained drop in estimated glomerular filtration rate of at least 40%. Researchers enrolled patients into FIDELIO-DKD based on having type 2 diabetes (T2D) and chronic kidney disease (CKD). The prevalence of a history of CVD was 46%.
Among patients with a history of CVD, the composite adverse CVD outcome occurred at a rate of 8.5/100 patient-years in patients on placebo and in 7.18/100 patients years among those on finerenone during a median of 2.6 years of follow-up, a 1.32/100–patient-year absolute between-group difference. Among patients in a primary prevention setting, incident CVD event rates during follow-up were roughly half that in the secondary prevention patients. The upshot was that, in the placebo group, the rate was 3.92/100 patient- years, and in those on finerenone was 3.43/100 patient-years, a 0.49/100–patient-year absolute difference.
CVD history produced heterogeneity for the primary endpoint
In the analysis that focused on the study’s primary, renal endpoint, among patients identified as having CVD at study entry, the outcome occurred at a rate of 9.06/100 patient-years in the placebo subgroup and at a rate of 6.6/100 patient years in those who received finerenone, a significant 30% relative risk reduction and an absolute between-group difference of 2.46/100 patient-years.
In contrast, among patients without a CVD history, the composite renal endpoint occurred at a rate of 9.1/100 patient-years in the placebo patients and 8.42/100 patient-years in those on finerenone, a 6% relative risk reduction that was not significant, and a 0.68/100–patient-year absolute difference. This disparity in the primary event rate between the two treatment arms reached statistical significance (P = .016), the investigators reported in the published version of the report in Circulation that simultaneously appeared online.
“The totality of evidence suggests that finerenone could be used in patients with T2D with or without a history of CVD,” explained Dr. Filippatos in an interview. “The P-interaction for the composite kidney outcome is significant, but it is not corrected for multiple testing; therefore, it might be a false-chance finding and must be interpreted cautiously.
Furthermore, in another prespecified kidney composite outcome the results were consistent in patients with and without a history of CVD. In sum, all the FIDELIO-DKD analyses so far are “suggestive of a beneficial effect in patients without a history of CVD.”
Despite these patients receiving guideline directed therapies, “there remains a high unmet medical need in patients with T2D and CKD,” added Dr. Filippatos, professor of cardiology at the University of Athens. “We use multiple treatments for patients with heart failure, and we should use the same mindset for treating patients with T2D and CKD. The costs of dialysis and kidney transplant are very high, so it is important to consider options that slow progression of CKD in these patients.”
In FIDELIO-DKD, virtually all patients were on background therapy with a renin-angiotensin-system (RAS) inhibitor, so the trial’s results suggest that treatment should at least involve dual therapy with finerenone and a RAS inhibitor. Fewer than 5% were on background therapy with a sodium-glucose cotransporter 2 (SGLT2) inhibitor, a drug class recently established as another key agent for treating CKD in patients with T2D, setting up the prospect for triple therapy, although this approach has not yet undergone prospective testing.
Combining RAS inhibition, finerenone, and an SGLT2 inhibitor is “potentially a marriage made in diabetes heaven,” commented Deepak L. Bhatt, MD, a professor of medicine at Harvard Medical School, Boston, who has not participated in finerenone studies.
Finerenone looks better for safety
Regardless of subgroup analyses based on history of CVD, the findings from all patients enrolled in FIDELIO-DKD were positive for the both the primary renal outcome and key secondary outcome of composite CVD events. In the total randomized cohort, treatment with finerenone on top of optimized treatment with an ACE inhibitor or angiotensin receptor blocker (RAS inhibition) led to a significant 18% relative risk reduction, compared with placebo, for the primary renal endpoint, and a significant 14% relative drop in the key secondary CVD outcome. Those results were published in October in the New England Journal of Medicine.
For treating patients with T2D and CKD ,finerenone overall “looks like a major advance,” Dr. Bhatt said in an interview.
In addition to the positive efficacy results, several experts also focused on what they saw as superior safety of finerenone in the trial, compared with the historical safety of the steroidal mineralocorticoid receptor antagonists (MRAs) now in use: spironolactone and eplerenone.
“I’m a big believer in spironolactone, but it has issues with side effects, and eplerenone never seemed to catch on,” said Dr. Bhatt, who is also executive director of interventional cardiovascular programs at Brigham and Women’s Hospital in Boston.
“A lot of physicians like these MRAs, but acknowledge that side effects have kept these drugs from being used to the extent they should.” The existing MRAs, especially spironolactone, have become a key drug class for treating heart failure with reduced ejection fraction (and, some claim, for also treating heart failure with preserved ejection fraction), as well as treatment-resistant hypertension and primary aldosteronism. By design, FIDELIO-DKD did not enroll patients with heart failure because treatment with an MRA is indicated for those with heart failure with reduced ejection fraction.
The spironolactone adverse effect that generates the greatest concern is hyperkalemia. During his discussion of FIDELIO-DKD as designated discussant, Christoph Wanner, MD, noted a recent study in which the incidence of hyperkalemia severe enough to cause study discontinuation was 23% among patients treated with spironolactone for heart failure, which contrasts with the 2.3% rate in FIDELIO-DKD among finerenone recipients. This hyperkalemia incidence from finerenone also improved on the historical performance of other drugs, like aliskiren (Tekturna), said Dr. Wanner, professor and head of nephrology at the University of Würzburg (Germany).
The FIDELIO-DKD results place finerenone alongside the RAS- and SGLT2-inhibitor drug classes as appropriate treatments for most patients with T2D and CKD. “We have entered a new era of effective treatment for diabetic kidney disease,” Dr. Wanner declared.
“The overall safety profile of finerenone looked better, including hyperkalemia,” said Dr. Bhatt. “Hyperkalemia with spironolactone is not necessarily as bad as the perception. With careful monitoring of spironolactone, the hyperkalemia is manageable. But the perception is that it’s bad, and along with gynecomastia it’s a real killer.”
While some dismiss gynecomastia as a major concern (for men) with spironolactone treatment, “if medical students learn one thing about spironolactone, it’s that it can cause gynecomastia,” adding to the negative image that the approved MRAs carry, Dr. Bhatt said.
“The hyperkalemia was manageable. This is very important because of past problems with potassium when using spironolactone,” Dr. Filippatos said. Finerenone also looks “more cardiorenal protective” than the steroidal MRAs, exerting renal benefits in FIDELIO-DKD never previously described for a steroidal MRA.
Some of the uncertainty about the efficacy of finerenone in patients with a history of cardiovascular disease will lift when results are available in about another year from the FIGARO-DKD pivotal trial of finerenone, which enrolled more than 7,000 patients with T2D and CKD (entry criteria very similar to FIDELIO-CKD). A big difference is that FIGARO-DKD has a composite CVD event metric as its primary endpoint, and includes hospitalization for heart failure as one facet of the composite.
FIDELIO-DKD was sponsored by Bayer. Dr. Filippatos has been a lecturer on behalf of, served as a researcher for, or both for Bayer and also for Amgen, Boehringer Ingelheim, Medtronic, Novartis, Servier, and Vifor. Dr. Bhatt has received research funding from Bayer and also from several other companies, and he also is an adviser to several companies. Dr. Wanner has received honoraria from Bayer, and also from AstraZeneca, Boehringer Ingelheim, FMC, Gilead, GlaxoSmithKline, Lilly, and Merck.
Finerenone, the first nonsteroidal mineralocorticoid receptor antagonist to complete a phase 3 trial, showed cardiovascular benefits in patients with type 2 diabetes and chronic kidney disease, regardless of whether they entered the study with a history of cardiovascular disease, in follow-up analyses of the FIDELIO-DKD trial, which included 5,674 patients.
“Finerenone demonstrated benefits for primary and secondary cardiovascular disease protection,” said Gerasimos Filippatos, MD, at the American Heart Association scientific sessions. Finerenone treatment cut the rate of cardiovascular death, nonfatal MI or stroke, or heart failure hospitalization, when compared with placebo, by a relative 15% among patients with a history of cardiovascular disease (CVD), and by a relative 14% in patients without this history, differences that met a statistical test for consistency. But the absolute, drug-associated increments in benefit over placebo differed between the two CVD subgroups because of a sharp underlying difference in event rates.
In contrast, the analyses reported by Dr. Filippatos and associates from the FIDELIO-DKD study showed significant heterogeneity based on the presence or absence of CVD for the study’s primary endpoint, a composite renal metric that tallied the combined rate of death from renal causes, renal failure, or a sustained drop in estimated glomerular filtration rate of at least 40%. Researchers enrolled patients into FIDELIO-DKD based on having type 2 diabetes (T2D) and chronic kidney disease (CKD). The prevalence of a history of CVD was 46%.
Among patients with a history of CVD, the composite adverse CVD outcome occurred at a rate of 8.5/100 patient-years in patients on placebo and in 7.18/100 patients years among those on finerenone during a median of 2.6 years of follow-up, a 1.32/100–patient-year absolute between-group difference. Among patients in a primary prevention setting, incident CVD event rates during follow-up were roughly half that in the secondary prevention patients. The upshot was that, in the placebo group, the rate was 3.92/100 patient- years, and in those on finerenone was 3.43/100 patient-years, a 0.49/100–patient-year absolute difference.
CVD history produced heterogeneity for the primary endpoint
In the analysis that focused on the study’s primary, renal endpoint, among patients identified as having CVD at study entry, the outcome occurred at a rate of 9.06/100 patient-years in the placebo subgroup and at a rate of 6.6/100 patient years in those who received finerenone, a significant 30% relative risk reduction and an absolute between-group difference of 2.46/100 patient-years.
In contrast, among patients without a CVD history, the composite renal endpoint occurred at a rate of 9.1/100 patient-years in the placebo patients and 8.42/100 patient-years in those on finerenone, a 6% relative risk reduction that was not significant, and a 0.68/100–patient-year absolute difference. This disparity in the primary event rate between the two treatment arms reached statistical significance (P = .016), the investigators reported in the published version of the report in Circulation that simultaneously appeared online.
“The totality of evidence suggests that finerenone could be used in patients with T2D with or without a history of CVD,” explained Dr. Filippatos in an interview. “The P-interaction for the composite kidney outcome is significant, but it is not corrected for multiple testing; therefore, it might be a false-chance finding and must be interpreted cautiously.
Furthermore, in another prespecified kidney composite outcome the results were consistent in patients with and without a history of CVD. In sum, all the FIDELIO-DKD analyses so far are “suggestive of a beneficial effect in patients without a history of CVD.”
Despite these patients receiving guideline directed therapies, “there remains a high unmet medical need in patients with T2D and CKD,” added Dr. Filippatos, professor of cardiology at the University of Athens. “We use multiple treatments for patients with heart failure, and we should use the same mindset for treating patients with T2D and CKD. The costs of dialysis and kidney transplant are very high, so it is important to consider options that slow progression of CKD in these patients.”
In FIDELIO-DKD, virtually all patients were on background therapy with a renin-angiotensin-system (RAS) inhibitor, so the trial’s results suggest that treatment should at least involve dual therapy with finerenone and a RAS inhibitor. Fewer than 5% were on background therapy with a sodium-glucose cotransporter 2 (SGLT2) inhibitor, a drug class recently established as another key agent for treating CKD in patients with T2D, setting up the prospect for triple therapy, although this approach has not yet undergone prospective testing.
Combining RAS inhibition, finerenone, and an SGLT2 inhibitor is “potentially a marriage made in diabetes heaven,” commented Deepak L. Bhatt, MD, a professor of medicine at Harvard Medical School, Boston, who has not participated in finerenone studies.
Finerenone looks better for safety
Regardless of subgroup analyses based on history of CVD, the findings from all patients enrolled in FIDELIO-DKD were positive for the both the primary renal outcome and key secondary outcome of composite CVD events. In the total randomized cohort, treatment with finerenone on top of optimized treatment with an ACE inhibitor or angiotensin receptor blocker (RAS inhibition) led to a significant 18% relative risk reduction, compared with placebo, for the primary renal endpoint, and a significant 14% relative drop in the key secondary CVD outcome. Those results were published in October in the New England Journal of Medicine.
For treating patients with T2D and CKD ,finerenone overall “looks like a major advance,” Dr. Bhatt said in an interview.
In addition to the positive efficacy results, several experts also focused on what they saw as superior safety of finerenone in the trial, compared with the historical safety of the steroidal mineralocorticoid receptor antagonists (MRAs) now in use: spironolactone and eplerenone.
“I’m a big believer in spironolactone, but it has issues with side effects, and eplerenone never seemed to catch on,” said Dr. Bhatt, who is also executive director of interventional cardiovascular programs at Brigham and Women’s Hospital in Boston.
“A lot of physicians like these MRAs, but acknowledge that side effects have kept these drugs from being used to the extent they should.” The existing MRAs, especially spironolactone, have become a key drug class for treating heart failure with reduced ejection fraction (and, some claim, for also treating heart failure with preserved ejection fraction), as well as treatment-resistant hypertension and primary aldosteronism. By design, FIDELIO-DKD did not enroll patients with heart failure because treatment with an MRA is indicated for those with heart failure with reduced ejection fraction.
The spironolactone adverse effect that generates the greatest concern is hyperkalemia. During his discussion of FIDELIO-DKD as designated discussant, Christoph Wanner, MD, noted a recent study in which the incidence of hyperkalemia severe enough to cause study discontinuation was 23% among patients treated with spironolactone for heart failure, which contrasts with the 2.3% rate in FIDELIO-DKD among finerenone recipients. This hyperkalemia incidence from finerenone also improved on the historical performance of other drugs, like aliskiren (Tekturna), said Dr. Wanner, professor and head of nephrology at the University of Würzburg (Germany).
The FIDELIO-DKD results place finerenone alongside the RAS- and SGLT2-inhibitor drug classes as appropriate treatments for most patients with T2D and CKD. “We have entered a new era of effective treatment for diabetic kidney disease,” Dr. Wanner declared.
“The overall safety profile of finerenone looked better, including hyperkalemia,” said Dr. Bhatt. “Hyperkalemia with spironolactone is not necessarily as bad as the perception. With careful monitoring of spironolactone, the hyperkalemia is manageable. But the perception is that it’s bad, and along with gynecomastia it’s a real killer.”
While some dismiss gynecomastia as a major concern (for men) with spironolactone treatment, “if medical students learn one thing about spironolactone, it’s that it can cause gynecomastia,” adding to the negative image that the approved MRAs carry, Dr. Bhatt said.
“The hyperkalemia was manageable. This is very important because of past problems with potassium when using spironolactone,” Dr. Filippatos said. Finerenone also looks “more cardiorenal protective” than the steroidal MRAs, exerting renal benefits in FIDELIO-DKD never previously described for a steroidal MRA.
Some of the uncertainty about the efficacy of finerenone in patients with a history of cardiovascular disease will lift when results are available in about another year from the FIGARO-DKD pivotal trial of finerenone, which enrolled more than 7,000 patients with T2D and CKD (entry criteria very similar to FIDELIO-CKD). A big difference is that FIGARO-DKD has a composite CVD event metric as its primary endpoint, and includes hospitalization for heart failure as one facet of the composite.
FIDELIO-DKD was sponsored by Bayer. Dr. Filippatos has been a lecturer on behalf of, served as a researcher for, or both for Bayer and also for Amgen, Boehringer Ingelheim, Medtronic, Novartis, Servier, and Vifor. Dr. Bhatt has received research funding from Bayer and also from several other companies, and he also is an adviser to several companies. Dr. Wanner has received honoraria from Bayer, and also from AstraZeneca, Boehringer Ingelheim, FMC, Gilead, GlaxoSmithKline, Lilly, and Merck.
FROM AHA 2020
New HCM guidelines make shared decision-making top priority
Greater involvement of the patient and family in decision-making, clarity on the role of genetic testing and parameters for team-oriented care, and use of high-volume specialty centers are cornerstones of the first update in almost a decade of the American Heart Association/American College of Cardiology guideline for patients with hypertrophic cardiomyopathy (HCM).
The update lists 133 recommendations for HCM care in six categories: shared decision-making; role of high-volume HCM centers; diagnosis, initial evaluation, and follow-up; risk assessment and prevention of sudden cardiac death (SCD); management of HCM; and lifestyle considerations for patients.
“The guideline puts the patient front and center in the shared decision-making process and emphasizes the importance of incorporating patient’s lifestyle choices and preferences when making complex, life-altering decisions,” writing committee vice chair Seema Mital, MD, of the University of Toronto and the Hospital for Sick Children, also in Toronto, said in an interview.
The fully updated guideline, authored by a joint committee of the AHA and ACC with input from other specialty societies, has been published online in the Journal of the American College of Cardiology. It replaces the 2011 guideline.
Another key component of the update is the strong recommendation to utilize multidisciplinary care, said Matthew W. Martinez, MD, a writing committee member and sports cardiologists at Morristown (N.J.) Medical Center. “This is not only as a part of shared decision-making, but really in care for the patients,” he said, “that there’s a level of expertise that is provided by centers of excellence who handle HCM, and we did lay out some recommendations with regards to surgery, imaging, interventionists, and management with electrophysiology, and the care of athletes with potential for HCM and pregnant women.”
The update ranks recommendations by class of recommendation (COR), ranging from strong benefit much greater than risk to harm with risk exceeding benefit, and level of evidence (LOE). The recommendation for shared decision making, for example, carries at COR of 1, the highest rating, and a mid-level LOE of B-NR, meaning from nonrandomized studies. Patients who need septal reduction therapy (SRT) should be referred to a comprehensive or primary HCM center – a recommendation with a COR of 1 but an LOE of C-LD, meaning there are limited data.
From diagnosis to follow-up
The most extensive list of recommendations falls under the category covering diagnosis, initial evaluation and follow-up. They include a three-generation family history as part of the initial diagnostic assessment (COR, 1; LOE, B-NR), high-level recommendations for use of transthoracic echocardiogram in the initial work-up, every 1 or 2 years or when the patient’s status changes in confirmed cases, as well as parameters for using other imaging and diagnostic tests. Cardiovascular MRI, for example, is indicated when echocardiography is inconclusive (COR, 1; LOE, B-NR) and in other scenarios. When echocardiography is inconclusive but cardiac MRI isn’t available, cardiac CT is an option, albeit at a lower level of evidence (COR, 2b; LOE, C-LD).
Heart rhythm assessment has a high level of recommendation in multiple scenarios, even in first-degree relatives of HCM patients. Invasive hemodynamic assessment is in order for candidates of SRT whose left ventricular (LV) outflow tract obstruction status is unknown. This category also sets parameters for angiography, and exercise stress testing.
The most extensive recommendations for diagnosis and follow-up cover genetic testing; it consists of nine high-level recommendations.
“The guideline highlights not only the importance of genetic testing of an affected patient and genetic screening of family members, but also emphasizes ongoing reassessment of variant classification as this may evolve with time and change how we recommend ongoing family screening,” Dr. Mital noted.
“The guideline proposes initiating screening of family members at the earliest regardless of age given HCM can manifest at any age in affected families,” she added.
The guideline notes that the usefulness of genetic testing to evaluate the risk of sudden cardiac death (SCD) is uncertain. There’s even guidance for implementing those test results. Further testing is recommended for patients who are genotype positive and phenotype negative for HCM (COR, 1; LOE, B-NR). Those same patients may participate in competitive sports (COR, 2a; LOE, C-LD), but a pacemaker isn’t recommended as a primary prevention (COR, 3 [no benefit]; LOE, B-NR).
Risk evaluation and prevention
For SCD risk evaluation and prevention, the guideline spells out five components for the initial and follow-up evaluations (COR, 1; LOE, B-NR). That includes maximal LV wall thickness, ejection fraction, and LV apical aneurysm. The section include multiple recommendations for patient selection for placement of an implantable cardioverter-defibrillator (ICD). For example, it’s recommended for patient’s who’ve had a heart attack or sustained ventricular tachycardia (COR, 1; LOE, B-NR), but not so much for patients without risk factors or for participating in sports (COR, 3 [harm]; LOE, B-NR). The guideline even provides recommendations for selecting an ICD.
Management recommendations address when medical therapy is indicated, including which therapies are indicated for specific scenarios, as well as higher level interventions such as SRT for severely symptomatic patients with obstructive HCM (COR, 2b; LOE, C-LD) and surgical myectomy with ablation in patients with HCM and atrial fibrillation (COR, 2a; LOE, B-NR). This section also provides recommendations for managing patients with HCM and ventricular arrhythmias or advanced heart failure.
The guideline also includes a host of lifestyle considerations. Mild to moderate exercise is beneficial (COR, 1; LOE, B-NR), but athletes with HCM should consult with an “expert provider” (COR, 1; LOE, C, meaning based on expert opinion). Truck drivers, pilots and people who do strenuous physical labor with HCM should meet specific standards.
These recommendations again emphasize the role of shared decision-making, said Dr. Martinez. “It’s not a cookie-cutter discussion. It is taking all of the information, incorporating what the patient’s needs are, and then making sure you appropriately tell them what are the risks of exercising and not exercising. I have as many discussions through the day about what the risks of exercise are as I do the risks of not exercising.”
Refining nomenclature, pathophysiology
The writing committee addressed the nomenclature for HCM. The use of HCM to describe increased LV wall thickness linked to systemic diseases or secondary to LV hypertrophy “can lead to confusion,” the committee stated, so other cardiac or systemic causes of LV hypertrophy shouldn’t be labeled HCM. Other etiologies can cause secondary LV hypertrophy that can overlap with HCM; clinical markers and testing can help differentiate these mimickers from HCM. When echocardiography is inconclusive, cardiovascular MRI is indicated (COR, 1; LOE, B-NR).
The guideline update also provides clarity on the pathophysiology of HCM: It consists of dynamic LV outflow tract obstruction, mitral regurgitation, diastolic dysfunction, myocardial ischemia, arrhythmias, or autonomic dysfunction. “For a given patient with HCM, the clinical outcome may be dominated by one of these components or may be the result of a complex interplay,” the guideline states. The clinical evaluation should consider all these conditions.
This update also provides “clear separation” between care of HCM with and without obstruction, Dr. Martinez said. “The role of advanced therapies and referrals with advanced treatment options such as heart transplantation or CRT therapy in this group is different than before, recognizing that people with obstruction have symptoms that may be similar to those without obstruction, and the individual should be [thoroughly] investigated to make sure that you can discern between those two groups to make appropriate recommendations.”
The guideline was developed in collaboration with and endorsed by the American Association for Thoracic Surgery, American Society of Echocardiography, Heart Failure Society of America, Heart Rhythm Society, Society for Cardiovascular Angiography and Interventions, and Society for Cardiovascular Magnetic Resonance. It’s also been endorsed by the Pediatric & Congenital Electrophysiology Society.
Dr. Mital and Dr. Martinez have no relevant financial relationships to disclose.
SOURCE: Mital S et al. J Am Coll Cardiol. 2020 Nov 20. doi: 10.1016/j.jacc.2020.08.044.
Greater involvement of the patient and family in decision-making, clarity on the role of genetic testing and parameters for team-oriented care, and use of high-volume specialty centers are cornerstones of the first update in almost a decade of the American Heart Association/American College of Cardiology guideline for patients with hypertrophic cardiomyopathy (HCM).
The update lists 133 recommendations for HCM care in six categories: shared decision-making; role of high-volume HCM centers; diagnosis, initial evaluation, and follow-up; risk assessment and prevention of sudden cardiac death (SCD); management of HCM; and lifestyle considerations for patients.
“The guideline puts the patient front and center in the shared decision-making process and emphasizes the importance of incorporating patient’s lifestyle choices and preferences when making complex, life-altering decisions,” writing committee vice chair Seema Mital, MD, of the University of Toronto and the Hospital for Sick Children, also in Toronto, said in an interview.
The fully updated guideline, authored by a joint committee of the AHA and ACC with input from other specialty societies, has been published online in the Journal of the American College of Cardiology. It replaces the 2011 guideline.
Another key component of the update is the strong recommendation to utilize multidisciplinary care, said Matthew W. Martinez, MD, a writing committee member and sports cardiologists at Morristown (N.J.) Medical Center. “This is not only as a part of shared decision-making, but really in care for the patients,” he said, “that there’s a level of expertise that is provided by centers of excellence who handle HCM, and we did lay out some recommendations with regards to surgery, imaging, interventionists, and management with electrophysiology, and the care of athletes with potential for HCM and pregnant women.”
The update ranks recommendations by class of recommendation (COR), ranging from strong benefit much greater than risk to harm with risk exceeding benefit, and level of evidence (LOE). The recommendation for shared decision making, for example, carries at COR of 1, the highest rating, and a mid-level LOE of B-NR, meaning from nonrandomized studies. Patients who need septal reduction therapy (SRT) should be referred to a comprehensive or primary HCM center – a recommendation with a COR of 1 but an LOE of C-LD, meaning there are limited data.
From diagnosis to follow-up
The most extensive list of recommendations falls under the category covering diagnosis, initial evaluation and follow-up. They include a three-generation family history as part of the initial diagnostic assessment (COR, 1; LOE, B-NR), high-level recommendations for use of transthoracic echocardiogram in the initial work-up, every 1 or 2 years or when the patient’s status changes in confirmed cases, as well as parameters for using other imaging and diagnostic tests. Cardiovascular MRI, for example, is indicated when echocardiography is inconclusive (COR, 1; LOE, B-NR) and in other scenarios. When echocardiography is inconclusive but cardiac MRI isn’t available, cardiac CT is an option, albeit at a lower level of evidence (COR, 2b; LOE, C-LD).
Heart rhythm assessment has a high level of recommendation in multiple scenarios, even in first-degree relatives of HCM patients. Invasive hemodynamic assessment is in order for candidates of SRT whose left ventricular (LV) outflow tract obstruction status is unknown. This category also sets parameters for angiography, and exercise stress testing.
The most extensive recommendations for diagnosis and follow-up cover genetic testing; it consists of nine high-level recommendations.
“The guideline highlights not only the importance of genetic testing of an affected patient and genetic screening of family members, but also emphasizes ongoing reassessment of variant classification as this may evolve with time and change how we recommend ongoing family screening,” Dr. Mital noted.
“The guideline proposes initiating screening of family members at the earliest regardless of age given HCM can manifest at any age in affected families,” she added.
The guideline notes that the usefulness of genetic testing to evaluate the risk of sudden cardiac death (SCD) is uncertain. There’s even guidance for implementing those test results. Further testing is recommended for patients who are genotype positive and phenotype negative for HCM (COR, 1; LOE, B-NR). Those same patients may participate in competitive sports (COR, 2a; LOE, C-LD), but a pacemaker isn’t recommended as a primary prevention (COR, 3 [no benefit]; LOE, B-NR).
Risk evaluation and prevention
For SCD risk evaluation and prevention, the guideline spells out five components for the initial and follow-up evaluations (COR, 1; LOE, B-NR). That includes maximal LV wall thickness, ejection fraction, and LV apical aneurysm. The section include multiple recommendations for patient selection for placement of an implantable cardioverter-defibrillator (ICD). For example, it’s recommended for patient’s who’ve had a heart attack or sustained ventricular tachycardia (COR, 1; LOE, B-NR), but not so much for patients without risk factors or for participating in sports (COR, 3 [harm]; LOE, B-NR). The guideline even provides recommendations for selecting an ICD.
Management recommendations address when medical therapy is indicated, including which therapies are indicated for specific scenarios, as well as higher level interventions such as SRT for severely symptomatic patients with obstructive HCM (COR, 2b; LOE, C-LD) and surgical myectomy with ablation in patients with HCM and atrial fibrillation (COR, 2a; LOE, B-NR). This section also provides recommendations for managing patients with HCM and ventricular arrhythmias or advanced heart failure.
The guideline also includes a host of lifestyle considerations. Mild to moderate exercise is beneficial (COR, 1; LOE, B-NR), but athletes with HCM should consult with an “expert provider” (COR, 1; LOE, C, meaning based on expert opinion). Truck drivers, pilots and people who do strenuous physical labor with HCM should meet specific standards.
These recommendations again emphasize the role of shared decision-making, said Dr. Martinez. “It’s not a cookie-cutter discussion. It is taking all of the information, incorporating what the patient’s needs are, and then making sure you appropriately tell them what are the risks of exercising and not exercising. I have as many discussions through the day about what the risks of exercise are as I do the risks of not exercising.”
Refining nomenclature, pathophysiology
The writing committee addressed the nomenclature for HCM. The use of HCM to describe increased LV wall thickness linked to systemic diseases or secondary to LV hypertrophy “can lead to confusion,” the committee stated, so other cardiac or systemic causes of LV hypertrophy shouldn’t be labeled HCM. Other etiologies can cause secondary LV hypertrophy that can overlap with HCM; clinical markers and testing can help differentiate these mimickers from HCM. When echocardiography is inconclusive, cardiovascular MRI is indicated (COR, 1; LOE, B-NR).
The guideline update also provides clarity on the pathophysiology of HCM: It consists of dynamic LV outflow tract obstruction, mitral regurgitation, diastolic dysfunction, myocardial ischemia, arrhythmias, or autonomic dysfunction. “For a given patient with HCM, the clinical outcome may be dominated by one of these components or may be the result of a complex interplay,” the guideline states. The clinical evaluation should consider all these conditions.
This update also provides “clear separation” between care of HCM with and without obstruction, Dr. Martinez said. “The role of advanced therapies and referrals with advanced treatment options such as heart transplantation or CRT therapy in this group is different than before, recognizing that people with obstruction have symptoms that may be similar to those without obstruction, and the individual should be [thoroughly] investigated to make sure that you can discern between those two groups to make appropriate recommendations.”
The guideline was developed in collaboration with and endorsed by the American Association for Thoracic Surgery, American Society of Echocardiography, Heart Failure Society of America, Heart Rhythm Society, Society for Cardiovascular Angiography and Interventions, and Society for Cardiovascular Magnetic Resonance. It’s also been endorsed by the Pediatric & Congenital Electrophysiology Society.
Dr. Mital and Dr. Martinez have no relevant financial relationships to disclose.
SOURCE: Mital S et al. J Am Coll Cardiol. 2020 Nov 20. doi: 10.1016/j.jacc.2020.08.044.
Greater involvement of the patient and family in decision-making, clarity on the role of genetic testing and parameters for team-oriented care, and use of high-volume specialty centers are cornerstones of the first update in almost a decade of the American Heart Association/American College of Cardiology guideline for patients with hypertrophic cardiomyopathy (HCM).
The update lists 133 recommendations for HCM care in six categories: shared decision-making; role of high-volume HCM centers; diagnosis, initial evaluation, and follow-up; risk assessment and prevention of sudden cardiac death (SCD); management of HCM; and lifestyle considerations for patients.
“The guideline puts the patient front and center in the shared decision-making process and emphasizes the importance of incorporating patient’s lifestyle choices and preferences when making complex, life-altering decisions,” writing committee vice chair Seema Mital, MD, of the University of Toronto and the Hospital for Sick Children, also in Toronto, said in an interview.
The fully updated guideline, authored by a joint committee of the AHA and ACC with input from other specialty societies, has been published online in the Journal of the American College of Cardiology. It replaces the 2011 guideline.
Another key component of the update is the strong recommendation to utilize multidisciplinary care, said Matthew W. Martinez, MD, a writing committee member and sports cardiologists at Morristown (N.J.) Medical Center. “This is not only as a part of shared decision-making, but really in care for the patients,” he said, “that there’s a level of expertise that is provided by centers of excellence who handle HCM, and we did lay out some recommendations with regards to surgery, imaging, interventionists, and management with electrophysiology, and the care of athletes with potential for HCM and pregnant women.”
The update ranks recommendations by class of recommendation (COR), ranging from strong benefit much greater than risk to harm with risk exceeding benefit, and level of evidence (LOE). The recommendation for shared decision making, for example, carries at COR of 1, the highest rating, and a mid-level LOE of B-NR, meaning from nonrandomized studies. Patients who need septal reduction therapy (SRT) should be referred to a comprehensive or primary HCM center – a recommendation with a COR of 1 but an LOE of C-LD, meaning there are limited data.
From diagnosis to follow-up
The most extensive list of recommendations falls under the category covering diagnosis, initial evaluation and follow-up. They include a three-generation family history as part of the initial diagnostic assessment (COR, 1; LOE, B-NR), high-level recommendations for use of transthoracic echocardiogram in the initial work-up, every 1 or 2 years or when the patient’s status changes in confirmed cases, as well as parameters for using other imaging and diagnostic tests. Cardiovascular MRI, for example, is indicated when echocardiography is inconclusive (COR, 1; LOE, B-NR) and in other scenarios. When echocardiography is inconclusive but cardiac MRI isn’t available, cardiac CT is an option, albeit at a lower level of evidence (COR, 2b; LOE, C-LD).
Heart rhythm assessment has a high level of recommendation in multiple scenarios, even in first-degree relatives of HCM patients. Invasive hemodynamic assessment is in order for candidates of SRT whose left ventricular (LV) outflow tract obstruction status is unknown. This category also sets parameters for angiography, and exercise stress testing.
The most extensive recommendations for diagnosis and follow-up cover genetic testing; it consists of nine high-level recommendations.
“The guideline highlights not only the importance of genetic testing of an affected patient and genetic screening of family members, but also emphasizes ongoing reassessment of variant classification as this may evolve with time and change how we recommend ongoing family screening,” Dr. Mital noted.
“The guideline proposes initiating screening of family members at the earliest regardless of age given HCM can manifest at any age in affected families,” she added.
The guideline notes that the usefulness of genetic testing to evaluate the risk of sudden cardiac death (SCD) is uncertain. There’s even guidance for implementing those test results. Further testing is recommended for patients who are genotype positive and phenotype negative for HCM (COR, 1; LOE, B-NR). Those same patients may participate in competitive sports (COR, 2a; LOE, C-LD), but a pacemaker isn’t recommended as a primary prevention (COR, 3 [no benefit]; LOE, B-NR).
Risk evaluation and prevention
For SCD risk evaluation and prevention, the guideline spells out five components for the initial and follow-up evaluations (COR, 1; LOE, B-NR). That includes maximal LV wall thickness, ejection fraction, and LV apical aneurysm. The section include multiple recommendations for patient selection for placement of an implantable cardioverter-defibrillator (ICD). For example, it’s recommended for patient’s who’ve had a heart attack or sustained ventricular tachycardia (COR, 1; LOE, B-NR), but not so much for patients without risk factors or for participating in sports (COR, 3 [harm]; LOE, B-NR). The guideline even provides recommendations for selecting an ICD.
Management recommendations address when medical therapy is indicated, including which therapies are indicated for specific scenarios, as well as higher level interventions such as SRT for severely symptomatic patients with obstructive HCM (COR, 2b; LOE, C-LD) and surgical myectomy with ablation in patients with HCM and atrial fibrillation (COR, 2a; LOE, B-NR). This section also provides recommendations for managing patients with HCM and ventricular arrhythmias or advanced heart failure.
The guideline also includes a host of lifestyle considerations. Mild to moderate exercise is beneficial (COR, 1; LOE, B-NR), but athletes with HCM should consult with an “expert provider” (COR, 1; LOE, C, meaning based on expert opinion). Truck drivers, pilots and people who do strenuous physical labor with HCM should meet specific standards.
These recommendations again emphasize the role of shared decision-making, said Dr. Martinez. “It’s not a cookie-cutter discussion. It is taking all of the information, incorporating what the patient’s needs are, and then making sure you appropriately tell them what are the risks of exercising and not exercising. I have as many discussions through the day about what the risks of exercise are as I do the risks of not exercising.”
Refining nomenclature, pathophysiology
The writing committee addressed the nomenclature for HCM. The use of HCM to describe increased LV wall thickness linked to systemic diseases or secondary to LV hypertrophy “can lead to confusion,” the committee stated, so other cardiac or systemic causes of LV hypertrophy shouldn’t be labeled HCM. Other etiologies can cause secondary LV hypertrophy that can overlap with HCM; clinical markers and testing can help differentiate these mimickers from HCM. When echocardiography is inconclusive, cardiovascular MRI is indicated (COR, 1; LOE, B-NR).
The guideline update also provides clarity on the pathophysiology of HCM: It consists of dynamic LV outflow tract obstruction, mitral regurgitation, diastolic dysfunction, myocardial ischemia, arrhythmias, or autonomic dysfunction. “For a given patient with HCM, the clinical outcome may be dominated by one of these components or may be the result of a complex interplay,” the guideline states. The clinical evaluation should consider all these conditions.
This update also provides “clear separation” between care of HCM with and without obstruction, Dr. Martinez said. “The role of advanced therapies and referrals with advanced treatment options such as heart transplantation or CRT therapy in this group is different than before, recognizing that people with obstruction have symptoms that may be similar to those without obstruction, and the individual should be [thoroughly] investigated to make sure that you can discern between those two groups to make appropriate recommendations.”
The guideline was developed in collaboration with and endorsed by the American Association for Thoracic Surgery, American Society of Echocardiography, Heart Failure Society of America, Heart Rhythm Society, Society for Cardiovascular Angiography and Interventions, and Society for Cardiovascular Magnetic Resonance. It’s also been endorsed by the Pediatric & Congenital Electrophysiology Society.
Dr. Mital and Dr. Martinez have no relevant financial relationships to disclose.
SOURCE: Mital S et al. J Am Coll Cardiol. 2020 Nov 20. doi: 10.1016/j.jacc.2020.08.044.
Myocarditis rare, macrophage infiltration common at COVID autopsy
An international autopsy study of 21 patients who died from COVID-19 has shown the presence of multifocal lymphocytic myocarditis in three patients (14%). In an additional six patients, focally increased interstitial T-lymphocytes within the myocardium were noted, with only focal or no myocyte injury.
However, increased interstitial macrophage infiltration, possibly related to cytokine infiltration, was seen in 86% of patients.
“One way to think about this is that, if these patients were having biopsies and not autopsies, there would be myocardial injury in the patients with myocarditis, even after they recovered. But with interstitial macrophages, there may or may not be any injury,” said cardiovascular pathologist James R. Stone, MD, PhD, Massachusetts General Hospital, Boston.
Dr. Stone and colleagues from Mass General, two hospitals in Italy, the University of Amsterdam, and the Mayo Clinic in Rochester, Minn., conducted the autopsies in March and April. The results were published in the October 14 issue of the European Heart Journal.
Their technique was rigorous: a median of 20 full-thickness blocks of myocardium were examined histologically (range, 5-29 blocks).
The presence of myocarditis, defined by the presence of multiple foci of inflammation with associated myocyte injury, was determined, and the inflammatory cell composition analyzed by immunohistochemistry.
“I think one of the take-homes from this study is that you have to do a thorough sampling of the heart in order to exclude myocardial injury. You cannot exclude myocarditis with just a biopsy or two,” said Dr. Stone in an interview.
“We looked at multiple different sections of tissue preserved in paraffin for every case and found only 14% had myocarditis. The vast majority of autopsies done on patients dying from COVID-19 have short-changed the autopsy and not been done in a way to exclude myocarditis,” he added.
For all patients, COVID-19 was the underlying cause of death, but the mechanisms of death were acute respiratory distress syndrome in 15, viral pneumonia in 4, cardiogenic shock in 1, and cardiac arrest in 1. Seven patients had a history of cardiovascular disease, including atrial fibrillation in four, coronary artery disease in three, left ventricular hypertrophy in one, and previous valve replacement in one. A total of 16 had hypertension, 7 had diabetes mellitus, and 1 had chronic obstructive pulmonary disease. In four cases, mild pericarditis was present. Acute myocyte injury in the right ventricle, most probably from strain or overload, was also present in four cases.
A nonsignificant trend was seen toward higher serum troponin levels in the patients with myocarditis compared with those without myocarditis. There were no reports of disrupted coronary artery plaques, coronary artery aneurysms, or large pulmonary emboli.
Macrophage infiltration rather than myocarditis, myocardial injury?
The study sheds more light on previous cardiac magnetic resonance (CMR) imaging findings that have suggested that many patients who recover from COVID-19 show signs suggestive of myocarditis. These earlier studies include a recent one in competitive athletes and the earlier Puntmann and colleagues study of relatively young COVID-19 patients, which showed ongoing myocardial involvement in a majority of patients.
“It would not surprise me if some or all of the cardiac MR changes seen in some of these recent imaging studies are due to the macrophages,” said Dr. Stone.
“What we saw was not a routine pathology by any means. It was a huge amount of macrophages, higher that what we saw in SARS and more similar to a study published in 2007 that looked at patients with bacterial sepsis,” said Dr. Stone.
In an older study of SARS patients, 35% had the virus detected in myocardial tissue by polymerase chain reaction. In that subset, the degree of myocardial macrophage infiltrate was comparable to that seen in 86% of the COVID-19 cases described in this series.
Another possibility is that the macrophage infiltration reflects underlying disease rather than COVID-19. All but one of the patients had known underlying medical conditions associated with cardiac remodeling, said Nikolaos G. Frangogiannis, MD, a cardiologist who studies the mechanisms of cardiac injury, repair, and remodeling.
Frangogiannis, from Albert Einstein College of Medicine, New York, wrote an editorial that accompanied the autopsy study.
“The problem with this finding of increased macrophage infiltration is that it’s very hard to interpret because as we age, and especially in a less healthy population, the numbers and the density of macrophages in the heart increase, so it’s impossible to interpret as an effect of the infection itself unless you have an appropriate control population that matches the same characteristics, which is almost impossible to ask for,” he said.
“I’ve observed since the beginning of the pandemic that there seemed to be some people who wanted every single case to be myocarditis and others who had a bias toward not wanting COVID-19 to be a cause of myocarditis. I think what we’re seeing is it’s not either/or for anything with this virus, it’s a bit of everything,” said Dr. Stone.
Dr. Stone and Dr. Frangogiannis reported no conflict of interest.
A version of this article originally appeared on Medscape.com.
An international autopsy study of 21 patients who died from COVID-19 has shown the presence of multifocal lymphocytic myocarditis in three patients (14%). In an additional six patients, focally increased interstitial T-lymphocytes within the myocardium were noted, with only focal or no myocyte injury.
However, increased interstitial macrophage infiltration, possibly related to cytokine infiltration, was seen in 86% of patients.
“One way to think about this is that, if these patients were having biopsies and not autopsies, there would be myocardial injury in the patients with myocarditis, even after they recovered. But with interstitial macrophages, there may or may not be any injury,” said cardiovascular pathologist James R. Stone, MD, PhD, Massachusetts General Hospital, Boston.
Dr. Stone and colleagues from Mass General, two hospitals in Italy, the University of Amsterdam, and the Mayo Clinic in Rochester, Minn., conducted the autopsies in March and April. The results were published in the October 14 issue of the European Heart Journal.
Their technique was rigorous: a median of 20 full-thickness blocks of myocardium were examined histologically (range, 5-29 blocks).
The presence of myocarditis, defined by the presence of multiple foci of inflammation with associated myocyte injury, was determined, and the inflammatory cell composition analyzed by immunohistochemistry.
“I think one of the take-homes from this study is that you have to do a thorough sampling of the heart in order to exclude myocardial injury. You cannot exclude myocarditis with just a biopsy or two,” said Dr. Stone in an interview.
“We looked at multiple different sections of tissue preserved in paraffin for every case and found only 14% had myocarditis. The vast majority of autopsies done on patients dying from COVID-19 have short-changed the autopsy and not been done in a way to exclude myocarditis,” he added.
For all patients, COVID-19 was the underlying cause of death, but the mechanisms of death were acute respiratory distress syndrome in 15, viral pneumonia in 4, cardiogenic shock in 1, and cardiac arrest in 1. Seven patients had a history of cardiovascular disease, including atrial fibrillation in four, coronary artery disease in three, left ventricular hypertrophy in one, and previous valve replacement in one. A total of 16 had hypertension, 7 had diabetes mellitus, and 1 had chronic obstructive pulmonary disease. In four cases, mild pericarditis was present. Acute myocyte injury in the right ventricle, most probably from strain or overload, was also present in four cases.
A nonsignificant trend was seen toward higher serum troponin levels in the patients with myocarditis compared with those without myocarditis. There were no reports of disrupted coronary artery plaques, coronary artery aneurysms, or large pulmonary emboli.
Macrophage infiltration rather than myocarditis, myocardial injury?
The study sheds more light on previous cardiac magnetic resonance (CMR) imaging findings that have suggested that many patients who recover from COVID-19 show signs suggestive of myocarditis. These earlier studies include a recent one in competitive athletes and the earlier Puntmann and colleagues study of relatively young COVID-19 patients, which showed ongoing myocardial involvement in a majority of patients.
“It would not surprise me if some or all of the cardiac MR changes seen in some of these recent imaging studies are due to the macrophages,” said Dr. Stone.
“What we saw was not a routine pathology by any means. It was a huge amount of macrophages, higher that what we saw in SARS and more similar to a study published in 2007 that looked at patients with bacterial sepsis,” said Dr. Stone.
In an older study of SARS patients, 35% had the virus detected in myocardial tissue by polymerase chain reaction. In that subset, the degree of myocardial macrophage infiltrate was comparable to that seen in 86% of the COVID-19 cases described in this series.
Another possibility is that the macrophage infiltration reflects underlying disease rather than COVID-19. All but one of the patients had known underlying medical conditions associated with cardiac remodeling, said Nikolaos G. Frangogiannis, MD, a cardiologist who studies the mechanisms of cardiac injury, repair, and remodeling.
Frangogiannis, from Albert Einstein College of Medicine, New York, wrote an editorial that accompanied the autopsy study.
“The problem with this finding of increased macrophage infiltration is that it’s very hard to interpret because as we age, and especially in a less healthy population, the numbers and the density of macrophages in the heart increase, so it’s impossible to interpret as an effect of the infection itself unless you have an appropriate control population that matches the same characteristics, which is almost impossible to ask for,” he said.
“I’ve observed since the beginning of the pandemic that there seemed to be some people who wanted every single case to be myocarditis and others who had a bias toward not wanting COVID-19 to be a cause of myocarditis. I think what we’re seeing is it’s not either/or for anything with this virus, it’s a bit of everything,” said Dr. Stone.
Dr. Stone and Dr. Frangogiannis reported no conflict of interest.
A version of this article originally appeared on Medscape.com.
An international autopsy study of 21 patients who died from COVID-19 has shown the presence of multifocal lymphocytic myocarditis in three patients (14%). In an additional six patients, focally increased interstitial T-lymphocytes within the myocardium were noted, with only focal or no myocyte injury.
However, increased interstitial macrophage infiltration, possibly related to cytokine infiltration, was seen in 86% of patients.
“One way to think about this is that, if these patients were having biopsies and not autopsies, there would be myocardial injury in the patients with myocarditis, even after they recovered. But with interstitial macrophages, there may or may not be any injury,” said cardiovascular pathologist James R. Stone, MD, PhD, Massachusetts General Hospital, Boston.
Dr. Stone and colleagues from Mass General, two hospitals in Italy, the University of Amsterdam, and the Mayo Clinic in Rochester, Minn., conducted the autopsies in March and April. The results were published in the October 14 issue of the European Heart Journal.
Their technique was rigorous: a median of 20 full-thickness blocks of myocardium were examined histologically (range, 5-29 blocks).
The presence of myocarditis, defined by the presence of multiple foci of inflammation with associated myocyte injury, was determined, and the inflammatory cell composition analyzed by immunohistochemistry.
“I think one of the take-homes from this study is that you have to do a thorough sampling of the heart in order to exclude myocardial injury. You cannot exclude myocarditis with just a biopsy or two,” said Dr. Stone in an interview.
“We looked at multiple different sections of tissue preserved in paraffin for every case and found only 14% had myocarditis. The vast majority of autopsies done on patients dying from COVID-19 have short-changed the autopsy and not been done in a way to exclude myocarditis,” he added.
For all patients, COVID-19 was the underlying cause of death, but the mechanisms of death were acute respiratory distress syndrome in 15, viral pneumonia in 4, cardiogenic shock in 1, and cardiac arrest in 1. Seven patients had a history of cardiovascular disease, including atrial fibrillation in four, coronary artery disease in three, left ventricular hypertrophy in one, and previous valve replacement in one. A total of 16 had hypertension, 7 had diabetes mellitus, and 1 had chronic obstructive pulmonary disease. In four cases, mild pericarditis was present. Acute myocyte injury in the right ventricle, most probably from strain or overload, was also present in four cases.
A nonsignificant trend was seen toward higher serum troponin levels in the patients with myocarditis compared with those without myocarditis. There were no reports of disrupted coronary artery plaques, coronary artery aneurysms, or large pulmonary emboli.
Macrophage infiltration rather than myocarditis, myocardial injury?
The study sheds more light on previous cardiac magnetic resonance (CMR) imaging findings that have suggested that many patients who recover from COVID-19 show signs suggestive of myocarditis. These earlier studies include a recent one in competitive athletes and the earlier Puntmann and colleagues study of relatively young COVID-19 patients, which showed ongoing myocardial involvement in a majority of patients.
“It would not surprise me if some or all of the cardiac MR changes seen in some of these recent imaging studies are due to the macrophages,” said Dr. Stone.
“What we saw was not a routine pathology by any means. It was a huge amount of macrophages, higher that what we saw in SARS and more similar to a study published in 2007 that looked at patients with bacterial sepsis,” said Dr. Stone.
In an older study of SARS patients, 35% had the virus detected in myocardial tissue by polymerase chain reaction. In that subset, the degree of myocardial macrophage infiltrate was comparable to that seen in 86% of the COVID-19 cases described in this series.
Another possibility is that the macrophage infiltration reflects underlying disease rather than COVID-19. All but one of the patients had known underlying medical conditions associated with cardiac remodeling, said Nikolaos G. Frangogiannis, MD, a cardiologist who studies the mechanisms of cardiac injury, repair, and remodeling.
Frangogiannis, from Albert Einstein College of Medicine, New York, wrote an editorial that accompanied the autopsy study.
“The problem with this finding of increased macrophage infiltration is that it’s very hard to interpret because as we age, and especially in a less healthy population, the numbers and the density of macrophages in the heart increase, so it’s impossible to interpret as an effect of the infection itself unless you have an appropriate control population that matches the same characteristics, which is almost impossible to ask for,” he said.
“I’ve observed since the beginning of the pandemic that there seemed to be some people who wanted every single case to be myocarditis and others who had a bias toward not wanting COVID-19 to be a cause of myocarditis. I think what we’re seeing is it’s not either/or for anything with this virus, it’s a bit of everything,” said Dr. Stone.
Dr. Stone and Dr. Frangogiannis reported no conflict of interest.
A version of this article originally appeared on Medscape.com.
First SGLT1/2 inhibitor shows ‘spectacular’ phase 3 safety and efficacy in T2D
Sotagliflozin, a novel type of sodium-glucose cotransporter inhibitor, showed the diverse benefits this drug class provides along some new twists in a pair of international pivotal trials that together enrolled nearly 12,000 patients with type 2 diabetes.
Unprecedented benefits were seen for the first time with a drug, sotagliflozin (Zynquista) that produces both sodium-glucose cotransporter 2 inhibition as well as SGLT1 inhibition.
They included a big reduction in both MIs and strokes; an ability to meaningfully reduce hyperglycemia in patients with severe renal dysfunction with an estimated glomerular filtration rate (eGFR) of 25-29 mL/min per 1.73 m2; an ability to safely and effectively start in patients still hospitalized (but stable) for an acute heart failure episode; and a striking 37% relative risk reduction in cardiovascular death, heart failure hospitalizations, or an urgent outpatient visit for heart failure in 739 of the patients enrolled in both trials who had heart failure with preserved ejection fraction (HFpEF).
These studies produced for the first time evidence from controlled, prospective, randomized trials that a drug could improve the outcome of HFpEF patients.
All these novel outcomes came on top of the usual benefits clinicians have generally seen across the SGLT2 inhibitors already on the U.S. market: reductions in cardiovascular death and heart failure hospitalizations among all patients with type 2 diabetes, preservation of renal function, and hemoglobin A1c lowering among T2D patients with eGFR levels of at least 30 mL/min per 1.73 m2.
“The data look spectacular,” summed up Deepak L. Bhatt, MD, who presented the results from the two trials, SOLOIST-WHF and SCORED, in talks at the virtual scientific sessions of the American Heart Association.
“I think sotagliflozin has the potential to be the best in class” based on the several added attributes shown in the two trials, he said in an interview. “We’ve shown that it is very safe, well tolerated, and effective.”
The primary results were a significant 33% relative risk reduction with sotagliflozin treatment, compared with placebo in the rate of total cardiovascular deaths, hospitalizations for heart failure, or urgent outpatient visits for heart failure during just over 9 months of median follow-up among patients with T2D recently hospitalized for heart failure in SOLOIST-WFH. And a significant 26% relative risk reduction with sotagliflozin for the same endpoint after a median follow-up of just over 14 months in SCORED, which enrolled patients with T2D and chronic kidney disease.
“Sotagliflozin adds to the SGLT2 inhibitor story,” and the SOLOIST-WHF results “may shift our focus to vulnerable, acute heart failure patients with an opportunity to treat during the transition phase,” when these patients leave the hospital, commented Jane E. Wilcox, MD, the study’s designated discussant and a heart failure cardiologist at Northwestern Medicine in Chicago.
A dual SGLT inhibitor
What sets sotagliflozin apart from the SGLT2 inhibitors is that it not only inhibits that protein but also SGTL1, which primarily resides in the gastrointestinal tract and is the main route for gut absorption of glucose. Dr. Bhatt said that he was unaware of any other SGLT1/2 inhibitors currently in advanced clinical testing.
The activity of sotagliflozin against the SGLT1 protein likely explains its ability to cut A1c levels in patients with severe renal dysfunction, a condition that stymies glucose lowering by SGLT2 inhibitors. In SCORED, which randomized 10,584 patients with T2D at 750 study sites in 44 countries, 813 patients (8%) had an eGFR of 25-29 mL/min per 1.73 m2 at enrollment. Sotagliflozin treatment led to an average 0.6% cut in A1c in this subgroup, and by the same average amount among the patients with GFRs of 30-60 mL/min per 1.73 m2.
“This is a huge finding for endocrinologists and primary care physicians” who treat patients with T2D who have severe renal dysfunction, said Dr. Bhatt, a professor of medicine at Harvard Medical School in Boston. “It’s a good enough reason by itself to approve this drug.”
The same mechanism may also be behind another unexpected finding in SCORED. Treatment with sotagliflozin cut the rate of total episodes of cardiovascular death, nonfatal MI, or nonfatal stroke by an absolute 1.6%, compared with placebo, and by a relative 23%. This benefit was largely driven by a 32% relative risk reduction total in MIs, and a 34% relative risk reduction in total stroke, both significant differences.
“No SGLT2 inhibitor has shown a reduction in stroke, and the MI signals have been mixed. The sizable MI and stroke effects are unique to sotagliflozin,” compared with the SGLT2 inhibitors, and likely reflect one or more mechanisms that result from blocked gut SGLT1 and a cut in GI glucose uptake, said Dr. Bhatt. “Probably some novel mechanism we don’t fully understand.”
First-ever HFpEF benefit
In contrast to these two benefits that are probably unique to drugs that inhibit the SGLT1 protein, sotagliflozin showed two other notable and unprecedented benefits that are likely generalizable to the SGLT2 inhibitors.
First is the striking benefit for HFpEF. Neither SOLOIST, which enrolled 1,222 patients with T2D and just hospitalized for worsening heart failure, nor SCORED, which enrolled patients with T2D and chronic kidney disease based exclusively on an eGFR of 25-60 mL/min per 1.73 m2, excluded patients with HFpEF, defined as heart failure patients with a left ventricular ejection fraction of at least 50%. The two studies together included a total of 739 of these patients, and they split fairly evenly between treatment with sotagliflozin or placebo.
The combined analysis showed that the incidence rate for the primary endpoint in both SOLOIST and SCORED was 59% with placebo and 39% with sotagliflozin, an absolute event reduction of 11.6 events/100 patient-years, and a significant 37% relative risk reduction, with a number needed to treat to prevent 1 event per year event of 9.
Although this observation comes from a nonprespecified combined analysis, “to me this result seems real, and I think it’s a class effect that I’m willing to extrapolate to the SGLT2 inhibitors,” Dr. Bhatt said. “It will change my practice,” he added, by spurring him to more aggressively prescribe an SGLT2 inhibitor to a patient with T2D and HFpEF.
“I think there has been some hesitation to use SGLT2 inhibitors in T2D patients with HFpEF” because of the paucity of data in this population, even though labeling and society recommendations do not rule it out. “I hope this finding will move that needle, and also generally improve SGLT2 inhibitor uptake, which has been low,” he said.
Also safe soon after acute heart failure decompensation
The other finding likely generalizable to SGLT2 inhibitors stems from the design of SOLOIST-WHF, which tested the efficacy and safety of starting sotagliflozin in patients with T2D as soon as they were stable after hospitalization for acute heart failure decompensation.
“Showing safety and efficacy when started in the hospital is pretty meaningful, because its tells patients that this drug is important and they should stay on it,” which should improve adherence, predicted Dr. Bhatt, who is also executive director of Interventional Cardiovascular Programs at Brigham and Women’s Hospital in Boston. “That’s the ultimate treatment path to prevent patients from falling through the cracks” and failing to receive an SGLT2 inhibitor.
SOLOIST-WHF enrolled patients hospitalized for worsening heart failure who also required intravenous diuretic treatment but had become stable enough to transition to an oral diuretic and come off oxygen. During a median follow-up of just over 9 months (both SOLOIST-WHF and SCORED ended sooner than planned because of a change in drug company sponsorship), treatment with sotagliflozin cut the primary endpoint by a relative 33%, compared with placebo, and with an absolute reduction of 25 events per 100 patient-years for a number needed to treat of 4. Sotagliflozin produced a strikingly high level of treatment efficiency driven by the high event rate in these recently decompensated patients. The benefit also appeared quickly, with a significant cut in events discernible within 28 days.
Extrapolating this finding to the SGLT2 inhibitors is “not a huge leap of faith,” Dr. Bhatt said.
“There is a role for sotagliflozin in acute heart failure. It showed benefit in these high-risk, transition-phase patients,” said Dr. Wilcox.
Simultaneously with Dr. Bhatt’s presentation, results of SOLOIST-WHF and SCORED were published online in the New England Journal of Medicine.
The trials were sponsored initially by Sanofi, and more recently by Lexicon. Dr. Bhatt has received research funding from both companies, and also from several other companies. He also is an adviser to several companies. Dr. Wilcox has been a consultant to Boehringer Ingelheim and Medtronic.
Sotagliflozin, a novel type of sodium-glucose cotransporter inhibitor, showed the diverse benefits this drug class provides along some new twists in a pair of international pivotal trials that together enrolled nearly 12,000 patients with type 2 diabetes.
Unprecedented benefits were seen for the first time with a drug, sotagliflozin (Zynquista) that produces both sodium-glucose cotransporter 2 inhibition as well as SGLT1 inhibition.
They included a big reduction in both MIs and strokes; an ability to meaningfully reduce hyperglycemia in patients with severe renal dysfunction with an estimated glomerular filtration rate (eGFR) of 25-29 mL/min per 1.73 m2; an ability to safely and effectively start in patients still hospitalized (but stable) for an acute heart failure episode; and a striking 37% relative risk reduction in cardiovascular death, heart failure hospitalizations, or an urgent outpatient visit for heart failure in 739 of the patients enrolled in both trials who had heart failure with preserved ejection fraction (HFpEF).
These studies produced for the first time evidence from controlled, prospective, randomized trials that a drug could improve the outcome of HFpEF patients.
All these novel outcomes came on top of the usual benefits clinicians have generally seen across the SGLT2 inhibitors already on the U.S. market: reductions in cardiovascular death and heart failure hospitalizations among all patients with type 2 diabetes, preservation of renal function, and hemoglobin A1c lowering among T2D patients with eGFR levels of at least 30 mL/min per 1.73 m2.
“The data look spectacular,” summed up Deepak L. Bhatt, MD, who presented the results from the two trials, SOLOIST-WHF and SCORED, in talks at the virtual scientific sessions of the American Heart Association.
“I think sotagliflozin has the potential to be the best in class” based on the several added attributes shown in the two trials, he said in an interview. “We’ve shown that it is very safe, well tolerated, and effective.”
The primary results were a significant 33% relative risk reduction with sotagliflozin treatment, compared with placebo in the rate of total cardiovascular deaths, hospitalizations for heart failure, or urgent outpatient visits for heart failure during just over 9 months of median follow-up among patients with T2D recently hospitalized for heart failure in SOLOIST-WFH. And a significant 26% relative risk reduction with sotagliflozin for the same endpoint after a median follow-up of just over 14 months in SCORED, which enrolled patients with T2D and chronic kidney disease.
“Sotagliflozin adds to the SGLT2 inhibitor story,” and the SOLOIST-WHF results “may shift our focus to vulnerable, acute heart failure patients with an opportunity to treat during the transition phase,” when these patients leave the hospital, commented Jane E. Wilcox, MD, the study’s designated discussant and a heart failure cardiologist at Northwestern Medicine in Chicago.
A dual SGLT inhibitor
What sets sotagliflozin apart from the SGLT2 inhibitors is that it not only inhibits that protein but also SGTL1, which primarily resides in the gastrointestinal tract and is the main route for gut absorption of glucose. Dr. Bhatt said that he was unaware of any other SGLT1/2 inhibitors currently in advanced clinical testing.
The activity of sotagliflozin against the SGLT1 protein likely explains its ability to cut A1c levels in patients with severe renal dysfunction, a condition that stymies glucose lowering by SGLT2 inhibitors. In SCORED, which randomized 10,584 patients with T2D at 750 study sites in 44 countries, 813 patients (8%) had an eGFR of 25-29 mL/min per 1.73 m2 at enrollment. Sotagliflozin treatment led to an average 0.6% cut in A1c in this subgroup, and by the same average amount among the patients with GFRs of 30-60 mL/min per 1.73 m2.
“This is a huge finding for endocrinologists and primary care physicians” who treat patients with T2D who have severe renal dysfunction, said Dr. Bhatt, a professor of medicine at Harvard Medical School in Boston. “It’s a good enough reason by itself to approve this drug.”
The same mechanism may also be behind another unexpected finding in SCORED. Treatment with sotagliflozin cut the rate of total episodes of cardiovascular death, nonfatal MI, or nonfatal stroke by an absolute 1.6%, compared with placebo, and by a relative 23%. This benefit was largely driven by a 32% relative risk reduction total in MIs, and a 34% relative risk reduction in total stroke, both significant differences.
“No SGLT2 inhibitor has shown a reduction in stroke, and the MI signals have been mixed. The sizable MI and stroke effects are unique to sotagliflozin,” compared with the SGLT2 inhibitors, and likely reflect one or more mechanisms that result from blocked gut SGLT1 and a cut in GI glucose uptake, said Dr. Bhatt. “Probably some novel mechanism we don’t fully understand.”
First-ever HFpEF benefit
In contrast to these two benefits that are probably unique to drugs that inhibit the SGLT1 protein, sotagliflozin showed two other notable and unprecedented benefits that are likely generalizable to the SGLT2 inhibitors.
First is the striking benefit for HFpEF. Neither SOLOIST, which enrolled 1,222 patients with T2D and just hospitalized for worsening heart failure, nor SCORED, which enrolled patients with T2D and chronic kidney disease based exclusively on an eGFR of 25-60 mL/min per 1.73 m2, excluded patients with HFpEF, defined as heart failure patients with a left ventricular ejection fraction of at least 50%. The two studies together included a total of 739 of these patients, and they split fairly evenly between treatment with sotagliflozin or placebo.
The combined analysis showed that the incidence rate for the primary endpoint in both SOLOIST and SCORED was 59% with placebo and 39% with sotagliflozin, an absolute event reduction of 11.6 events/100 patient-years, and a significant 37% relative risk reduction, with a number needed to treat to prevent 1 event per year event of 9.
Although this observation comes from a nonprespecified combined analysis, “to me this result seems real, and I think it’s a class effect that I’m willing to extrapolate to the SGLT2 inhibitors,” Dr. Bhatt said. “It will change my practice,” he added, by spurring him to more aggressively prescribe an SGLT2 inhibitor to a patient with T2D and HFpEF.
“I think there has been some hesitation to use SGLT2 inhibitors in T2D patients with HFpEF” because of the paucity of data in this population, even though labeling and society recommendations do not rule it out. “I hope this finding will move that needle, and also generally improve SGLT2 inhibitor uptake, which has been low,” he said.
Also safe soon after acute heart failure decompensation
The other finding likely generalizable to SGLT2 inhibitors stems from the design of SOLOIST-WHF, which tested the efficacy and safety of starting sotagliflozin in patients with T2D as soon as they were stable after hospitalization for acute heart failure decompensation.
“Showing safety and efficacy when started in the hospital is pretty meaningful, because its tells patients that this drug is important and they should stay on it,” which should improve adherence, predicted Dr. Bhatt, who is also executive director of Interventional Cardiovascular Programs at Brigham and Women’s Hospital in Boston. “That’s the ultimate treatment path to prevent patients from falling through the cracks” and failing to receive an SGLT2 inhibitor.
SOLOIST-WHF enrolled patients hospitalized for worsening heart failure who also required intravenous diuretic treatment but had become stable enough to transition to an oral diuretic and come off oxygen. During a median follow-up of just over 9 months (both SOLOIST-WHF and SCORED ended sooner than planned because of a change in drug company sponsorship), treatment with sotagliflozin cut the primary endpoint by a relative 33%, compared with placebo, and with an absolute reduction of 25 events per 100 patient-years for a number needed to treat of 4. Sotagliflozin produced a strikingly high level of treatment efficiency driven by the high event rate in these recently decompensated patients. The benefit also appeared quickly, with a significant cut in events discernible within 28 days.
Extrapolating this finding to the SGLT2 inhibitors is “not a huge leap of faith,” Dr. Bhatt said.
“There is a role for sotagliflozin in acute heart failure. It showed benefit in these high-risk, transition-phase patients,” said Dr. Wilcox.
Simultaneously with Dr. Bhatt’s presentation, results of SOLOIST-WHF and SCORED were published online in the New England Journal of Medicine.
The trials were sponsored initially by Sanofi, and more recently by Lexicon. Dr. Bhatt has received research funding from both companies, and also from several other companies. He also is an adviser to several companies. Dr. Wilcox has been a consultant to Boehringer Ingelheim and Medtronic.
Sotagliflozin, a novel type of sodium-glucose cotransporter inhibitor, showed the diverse benefits this drug class provides along some new twists in a pair of international pivotal trials that together enrolled nearly 12,000 patients with type 2 diabetes.
Unprecedented benefits were seen for the first time with a drug, sotagliflozin (Zynquista) that produces both sodium-glucose cotransporter 2 inhibition as well as SGLT1 inhibition.
They included a big reduction in both MIs and strokes; an ability to meaningfully reduce hyperglycemia in patients with severe renal dysfunction with an estimated glomerular filtration rate (eGFR) of 25-29 mL/min per 1.73 m2; an ability to safely and effectively start in patients still hospitalized (but stable) for an acute heart failure episode; and a striking 37% relative risk reduction in cardiovascular death, heart failure hospitalizations, or an urgent outpatient visit for heart failure in 739 of the patients enrolled in both trials who had heart failure with preserved ejection fraction (HFpEF).
These studies produced for the first time evidence from controlled, prospective, randomized trials that a drug could improve the outcome of HFpEF patients.
All these novel outcomes came on top of the usual benefits clinicians have generally seen across the SGLT2 inhibitors already on the U.S. market: reductions in cardiovascular death and heart failure hospitalizations among all patients with type 2 diabetes, preservation of renal function, and hemoglobin A1c lowering among T2D patients with eGFR levels of at least 30 mL/min per 1.73 m2.
“The data look spectacular,” summed up Deepak L. Bhatt, MD, who presented the results from the two trials, SOLOIST-WHF and SCORED, in talks at the virtual scientific sessions of the American Heart Association.
“I think sotagliflozin has the potential to be the best in class” based on the several added attributes shown in the two trials, he said in an interview. “We’ve shown that it is very safe, well tolerated, and effective.”
The primary results were a significant 33% relative risk reduction with sotagliflozin treatment, compared with placebo in the rate of total cardiovascular deaths, hospitalizations for heart failure, or urgent outpatient visits for heart failure during just over 9 months of median follow-up among patients with T2D recently hospitalized for heart failure in SOLOIST-WFH. And a significant 26% relative risk reduction with sotagliflozin for the same endpoint after a median follow-up of just over 14 months in SCORED, which enrolled patients with T2D and chronic kidney disease.
“Sotagliflozin adds to the SGLT2 inhibitor story,” and the SOLOIST-WHF results “may shift our focus to vulnerable, acute heart failure patients with an opportunity to treat during the transition phase,” when these patients leave the hospital, commented Jane E. Wilcox, MD, the study’s designated discussant and a heart failure cardiologist at Northwestern Medicine in Chicago.
A dual SGLT inhibitor
What sets sotagliflozin apart from the SGLT2 inhibitors is that it not only inhibits that protein but also SGTL1, which primarily resides in the gastrointestinal tract and is the main route for gut absorption of glucose. Dr. Bhatt said that he was unaware of any other SGLT1/2 inhibitors currently in advanced clinical testing.
The activity of sotagliflozin against the SGLT1 protein likely explains its ability to cut A1c levels in patients with severe renal dysfunction, a condition that stymies glucose lowering by SGLT2 inhibitors. In SCORED, which randomized 10,584 patients with T2D at 750 study sites in 44 countries, 813 patients (8%) had an eGFR of 25-29 mL/min per 1.73 m2 at enrollment. Sotagliflozin treatment led to an average 0.6% cut in A1c in this subgroup, and by the same average amount among the patients with GFRs of 30-60 mL/min per 1.73 m2.
“This is a huge finding for endocrinologists and primary care physicians” who treat patients with T2D who have severe renal dysfunction, said Dr. Bhatt, a professor of medicine at Harvard Medical School in Boston. “It’s a good enough reason by itself to approve this drug.”
The same mechanism may also be behind another unexpected finding in SCORED. Treatment with sotagliflozin cut the rate of total episodes of cardiovascular death, nonfatal MI, or nonfatal stroke by an absolute 1.6%, compared with placebo, and by a relative 23%. This benefit was largely driven by a 32% relative risk reduction total in MIs, and a 34% relative risk reduction in total stroke, both significant differences.
“No SGLT2 inhibitor has shown a reduction in stroke, and the MI signals have been mixed. The sizable MI and stroke effects are unique to sotagliflozin,” compared with the SGLT2 inhibitors, and likely reflect one or more mechanisms that result from blocked gut SGLT1 and a cut in GI glucose uptake, said Dr. Bhatt. “Probably some novel mechanism we don’t fully understand.”
First-ever HFpEF benefit
In contrast to these two benefits that are probably unique to drugs that inhibit the SGLT1 protein, sotagliflozin showed two other notable and unprecedented benefits that are likely generalizable to the SGLT2 inhibitors.
First is the striking benefit for HFpEF. Neither SOLOIST, which enrolled 1,222 patients with T2D and just hospitalized for worsening heart failure, nor SCORED, which enrolled patients with T2D and chronic kidney disease based exclusively on an eGFR of 25-60 mL/min per 1.73 m2, excluded patients with HFpEF, defined as heart failure patients with a left ventricular ejection fraction of at least 50%. The two studies together included a total of 739 of these patients, and they split fairly evenly between treatment with sotagliflozin or placebo.
The combined analysis showed that the incidence rate for the primary endpoint in both SOLOIST and SCORED was 59% with placebo and 39% with sotagliflozin, an absolute event reduction of 11.6 events/100 patient-years, and a significant 37% relative risk reduction, with a number needed to treat to prevent 1 event per year event of 9.
Although this observation comes from a nonprespecified combined analysis, “to me this result seems real, and I think it’s a class effect that I’m willing to extrapolate to the SGLT2 inhibitors,” Dr. Bhatt said. “It will change my practice,” he added, by spurring him to more aggressively prescribe an SGLT2 inhibitor to a patient with T2D and HFpEF.
“I think there has been some hesitation to use SGLT2 inhibitors in T2D patients with HFpEF” because of the paucity of data in this population, even though labeling and society recommendations do not rule it out. “I hope this finding will move that needle, and also generally improve SGLT2 inhibitor uptake, which has been low,” he said.
Also safe soon after acute heart failure decompensation
The other finding likely generalizable to SGLT2 inhibitors stems from the design of SOLOIST-WHF, which tested the efficacy and safety of starting sotagliflozin in patients with T2D as soon as they were stable after hospitalization for acute heart failure decompensation.
“Showing safety and efficacy when started in the hospital is pretty meaningful, because its tells patients that this drug is important and they should stay on it,” which should improve adherence, predicted Dr. Bhatt, who is also executive director of Interventional Cardiovascular Programs at Brigham and Women’s Hospital in Boston. “That’s the ultimate treatment path to prevent patients from falling through the cracks” and failing to receive an SGLT2 inhibitor.
SOLOIST-WHF enrolled patients hospitalized for worsening heart failure who also required intravenous diuretic treatment but had become stable enough to transition to an oral diuretic and come off oxygen. During a median follow-up of just over 9 months (both SOLOIST-WHF and SCORED ended sooner than planned because of a change in drug company sponsorship), treatment with sotagliflozin cut the primary endpoint by a relative 33%, compared with placebo, and with an absolute reduction of 25 events per 100 patient-years for a number needed to treat of 4. Sotagliflozin produced a strikingly high level of treatment efficiency driven by the high event rate in these recently decompensated patients. The benefit also appeared quickly, with a significant cut in events discernible within 28 days.
Extrapolating this finding to the SGLT2 inhibitors is “not a huge leap of faith,” Dr. Bhatt said.
“There is a role for sotagliflozin in acute heart failure. It showed benefit in these high-risk, transition-phase patients,” said Dr. Wilcox.
Simultaneously with Dr. Bhatt’s presentation, results of SOLOIST-WHF and SCORED were published online in the New England Journal of Medicine.
The trials were sponsored initially by Sanofi, and more recently by Lexicon. Dr. Bhatt has received research funding from both companies, and also from several other companies. He also is an adviser to several companies. Dr. Wilcox has been a consultant to Boehringer Ingelheim and Medtronic.
FROM AHA 2020
Empagliflozin favorably reshaped left ventricles in HFrEF patients
Treatment with the SGLT2 inhibitor empagliflozin led to significant reductions in both left ventricular end systolic and diastolic volumes in two independent randomized studies of patients with heart failure with reduced ejection fraction.
These results provide important new evidence that one way a drug from this class exerts its beneficial effects on cardiovascular outcomes in these patients is by producing favorable left-ventricular remodeling.
One of the two studies involved only patients with heart failure with reduced ejection fraction (HFrEF) with diabetes and examined treatment impact after 36 weeks. The second study focused exclusively on HFrEF patients without diabetes and followed patients for 6 months. Both studies also generated additional significant evidence of favorable left-ventricular effects.
“The results of these two new trials are incredibly important, as they tell cardiologists one of the mechanisms by which SGLT2 [sodium glucose co-transporter 2] inhibitors reduce heart failure hospitalizations and cardiovascular death,” said Mark C. Petrie, MBChB, professor at the Institute of Cardiovascular & Medical Sciences at the University of Glasgow, and principal investigator for one of the two studies.
“Many cardiologists want to know mechanisms as well as clinical benefit. These remodeling data showing that these drugs reduce the size of abnormally large hearts [and] are also very important for patients,” Dr. Petrie said in an interview. “There have been more than 50 publications on potential mechanisms of benefit of SGLT2 inhibitors in HFrEF, but these are the first randomized, mechanistic data.”
Mechanistic clues follow large cardiovascular outcome trials
Results from a large randomized trial, EMPEROR-Reduced, recently showed that treatment with empagliflozin (Jardiance) on top of standard HFrEF treatment led to significant benefits in patients with or without type 2 diabetes (T2D), compared with placebo, for major cardiovascular and renal endpoints, including the combination of cardiovascular death or hospitalization for heart failure. And results from a second large randomized trial, DAPA-HF, showed similar results with a different drug from the same class, dapagliflozin (Farxiga), in an earlier report.
But while these reports led to quick uptake of these two drugs for the treatment of patients with HFrEF, the means by which these agents exert their HFrEF benefits have been unclear.
“Our study identifies why this drug [empagliflozin] is effective – because it improves heart function, something that has not been understood until now,” Carlos G. Santos-Gallego, MD, lead investigator for the second new report, said in a written statement. “Many doctors are afraid of prescribing a drug they do not understand, and our findings will help clinicians feel more comfortable giving this to patients once approved.”
On the strength of the DAPA-HF results, dapagliflozin received a revised U.S. label in May 2020 that added the indication for treating patients with HFrEF regardless of the whether patients also have T2D, the original indication for prescribing the drug. Many experts anticipate that a similar addition to the label for empagliflozin will soon occur.
EMPA-TROPISM examines patients with no T2D
The single-center study reported by Dr. Santos-Gallego randomized 84 patients with HFrEF and no diabetes to standard treatment with empagliflozin or placebo and measured several parameters in 80 patients who completed the planned 6 months of treatment. The primary endpoints were the changes in both left ventricular end systolic and diastolic volume from baseline in the empagliflozin-treated patients compared with patients on placebo, measured by cardiac MR.
The results showed an average reduction of end systolic volume of 26.6 mL from baseline compared with a small rise in the placebo patients, and an average drop in end diastolic volume of 25.1 mL from baseline compared again with a small increase in the controls. Both differences were statistically significant, reported the senior author of the study, Juan J. Badimon, PhD, in a talk at the virtual scientific sessions of the American Heart Association. Concurrently, the results were published online in the Journal of the American College of Cardiology.
Results from the EMPA-TROPISM study also showed several other significant benefits from empagliflozin treatment, both to left ventricular shape and function as well as to other measures of patient well being. The drug regimen led to an increase in left ventricular ejection fraction, a decrease in left ventricular mass, reduced myocardial fibrosis and aortic stiffness, increased peak oxygen consumption, an increased distance traveled in a 6-minute walk test, and improved quality of life, said Dr. Badimon, professor of medicine and director of the Atherothrombosis Research Unit at the Cardiovascular Institute at the Icahn School of Medicine at Mount Sinai in New York.
SUGAR-DM-HF enrolled only T2D patients
The second study, SUGAR-DM-HF, randomized 105 patients with HFrEF and T2D to treatment with empagliflozin or placebo at any of 15 centers in Scotland, with 92 patients completing the full 36 weeks on treatment. One of the study’s two primary endpoints was the change in left ventricular end systolic volume index, which dropped by an average of 7.9 mL/m2 in patients who received empagliflozin and by 1.5 mL/m2 in the controls, a significant average between-group difference of 6.0 mL/m2, reported Matthew M.Y. Lee, MBChB, at the same meeting.
However, the second primary endpoint, change in left ventricular global longitudinal strain, showed no significant difference in effect on empagliflozin compared with placebo, said Dr. Lee, a cardiologist at the University of Glasgow. Concurrently with his report the results appeared in an article published online in Circulation.
The results also showed a significant drop in left ventricular end diastolic volume index from baseline compared with the control patients, with an average between-group difference in the reduction from baseline of 8.2 mL/m2.
“Reverse cardiac remodeling is a mechanism by which SGLT2 inhibitors reduce heart failure hospitalizations and cardiovascular mortality,” Dr. Lee concluded during his presentation at the meeting.
Although the findings from both studies together provide strong evidence for an effect by empagliflozin on left ventricular shape and function, neither study provides much insight into how this drug exerts these effects. The authors of both studies agreed on several potential explanations, including reductions in cardiac preload and afterload that could reduce left ventricular stretch and volume; a change triggered in myocardial energetics that switches from a metabolism mostly dependent on glucose to one more geared to using fatty acids, ketone bodies, and branched chain amino acids; and a possible drug-induced reduction in oxidative stress and inflammation.
SUGAR-DM-HF was sponsored by a grant from Boehringer Ingelheim, the company that along with Eli Lilly markets empagliflozin (Jardiance). Dr. Lee had no disclosures. Dr. Petrie has been a consultant to Boehringer Ingelheim and Eli Lilly and to several other companies. EMPA-TROPISM was sponsored by a grant from Boehringer Ingelheim. Dr. Badimon and Dr. Santos-Gallego had no disclosures.
Treatment with the SGLT2 inhibitor empagliflozin led to significant reductions in both left ventricular end systolic and diastolic volumes in two independent randomized studies of patients with heart failure with reduced ejection fraction.
These results provide important new evidence that one way a drug from this class exerts its beneficial effects on cardiovascular outcomes in these patients is by producing favorable left-ventricular remodeling.
One of the two studies involved only patients with heart failure with reduced ejection fraction (HFrEF) with diabetes and examined treatment impact after 36 weeks. The second study focused exclusively on HFrEF patients without diabetes and followed patients for 6 months. Both studies also generated additional significant evidence of favorable left-ventricular effects.
“The results of these two new trials are incredibly important, as they tell cardiologists one of the mechanisms by which SGLT2 [sodium glucose co-transporter 2] inhibitors reduce heart failure hospitalizations and cardiovascular death,” said Mark C. Petrie, MBChB, professor at the Institute of Cardiovascular & Medical Sciences at the University of Glasgow, and principal investigator for one of the two studies.
“Many cardiologists want to know mechanisms as well as clinical benefit. These remodeling data showing that these drugs reduce the size of abnormally large hearts [and] are also very important for patients,” Dr. Petrie said in an interview. “There have been more than 50 publications on potential mechanisms of benefit of SGLT2 inhibitors in HFrEF, but these are the first randomized, mechanistic data.”
Mechanistic clues follow large cardiovascular outcome trials
Results from a large randomized trial, EMPEROR-Reduced, recently showed that treatment with empagliflozin (Jardiance) on top of standard HFrEF treatment led to significant benefits in patients with or without type 2 diabetes (T2D), compared with placebo, for major cardiovascular and renal endpoints, including the combination of cardiovascular death or hospitalization for heart failure. And results from a second large randomized trial, DAPA-HF, showed similar results with a different drug from the same class, dapagliflozin (Farxiga), in an earlier report.
But while these reports led to quick uptake of these two drugs for the treatment of patients with HFrEF, the means by which these agents exert their HFrEF benefits have been unclear.
“Our study identifies why this drug [empagliflozin] is effective – because it improves heart function, something that has not been understood until now,” Carlos G. Santos-Gallego, MD, lead investigator for the second new report, said in a written statement. “Many doctors are afraid of prescribing a drug they do not understand, and our findings will help clinicians feel more comfortable giving this to patients once approved.”
On the strength of the DAPA-HF results, dapagliflozin received a revised U.S. label in May 2020 that added the indication for treating patients with HFrEF regardless of the whether patients also have T2D, the original indication for prescribing the drug. Many experts anticipate that a similar addition to the label for empagliflozin will soon occur.
EMPA-TROPISM examines patients with no T2D
The single-center study reported by Dr. Santos-Gallego randomized 84 patients with HFrEF and no diabetes to standard treatment with empagliflozin or placebo and measured several parameters in 80 patients who completed the planned 6 months of treatment. The primary endpoints were the changes in both left ventricular end systolic and diastolic volume from baseline in the empagliflozin-treated patients compared with patients on placebo, measured by cardiac MR.
The results showed an average reduction of end systolic volume of 26.6 mL from baseline compared with a small rise in the placebo patients, and an average drop in end diastolic volume of 25.1 mL from baseline compared again with a small increase in the controls. Both differences were statistically significant, reported the senior author of the study, Juan J. Badimon, PhD, in a talk at the virtual scientific sessions of the American Heart Association. Concurrently, the results were published online in the Journal of the American College of Cardiology.
Results from the EMPA-TROPISM study also showed several other significant benefits from empagliflozin treatment, both to left ventricular shape and function as well as to other measures of patient well being. The drug regimen led to an increase in left ventricular ejection fraction, a decrease in left ventricular mass, reduced myocardial fibrosis and aortic stiffness, increased peak oxygen consumption, an increased distance traveled in a 6-minute walk test, and improved quality of life, said Dr. Badimon, professor of medicine and director of the Atherothrombosis Research Unit at the Cardiovascular Institute at the Icahn School of Medicine at Mount Sinai in New York.
SUGAR-DM-HF enrolled only T2D patients
The second study, SUGAR-DM-HF, randomized 105 patients with HFrEF and T2D to treatment with empagliflozin or placebo at any of 15 centers in Scotland, with 92 patients completing the full 36 weeks on treatment. One of the study’s two primary endpoints was the change in left ventricular end systolic volume index, which dropped by an average of 7.9 mL/m2 in patients who received empagliflozin and by 1.5 mL/m2 in the controls, a significant average between-group difference of 6.0 mL/m2, reported Matthew M.Y. Lee, MBChB, at the same meeting.
However, the second primary endpoint, change in left ventricular global longitudinal strain, showed no significant difference in effect on empagliflozin compared with placebo, said Dr. Lee, a cardiologist at the University of Glasgow. Concurrently with his report the results appeared in an article published online in Circulation.
The results also showed a significant drop in left ventricular end diastolic volume index from baseline compared with the control patients, with an average between-group difference in the reduction from baseline of 8.2 mL/m2.
“Reverse cardiac remodeling is a mechanism by which SGLT2 inhibitors reduce heart failure hospitalizations and cardiovascular mortality,” Dr. Lee concluded during his presentation at the meeting.
Although the findings from both studies together provide strong evidence for an effect by empagliflozin on left ventricular shape and function, neither study provides much insight into how this drug exerts these effects. The authors of both studies agreed on several potential explanations, including reductions in cardiac preload and afterload that could reduce left ventricular stretch and volume; a change triggered in myocardial energetics that switches from a metabolism mostly dependent on glucose to one more geared to using fatty acids, ketone bodies, and branched chain amino acids; and a possible drug-induced reduction in oxidative stress and inflammation.
SUGAR-DM-HF was sponsored by a grant from Boehringer Ingelheim, the company that along with Eli Lilly markets empagliflozin (Jardiance). Dr. Lee had no disclosures. Dr. Petrie has been a consultant to Boehringer Ingelheim and Eli Lilly and to several other companies. EMPA-TROPISM was sponsored by a grant from Boehringer Ingelheim. Dr. Badimon and Dr. Santos-Gallego had no disclosures.
Treatment with the SGLT2 inhibitor empagliflozin led to significant reductions in both left ventricular end systolic and diastolic volumes in two independent randomized studies of patients with heart failure with reduced ejection fraction.
These results provide important new evidence that one way a drug from this class exerts its beneficial effects on cardiovascular outcomes in these patients is by producing favorable left-ventricular remodeling.
One of the two studies involved only patients with heart failure with reduced ejection fraction (HFrEF) with diabetes and examined treatment impact after 36 weeks. The second study focused exclusively on HFrEF patients without diabetes and followed patients for 6 months. Both studies also generated additional significant evidence of favorable left-ventricular effects.
“The results of these two new trials are incredibly important, as they tell cardiologists one of the mechanisms by which SGLT2 [sodium glucose co-transporter 2] inhibitors reduce heart failure hospitalizations and cardiovascular death,” said Mark C. Petrie, MBChB, professor at the Institute of Cardiovascular & Medical Sciences at the University of Glasgow, and principal investigator for one of the two studies.
“Many cardiologists want to know mechanisms as well as clinical benefit. These remodeling data showing that these drugs reduce the size of abnormally large hearts [and] are also very important for patients,” Dr. Petrie said in an interview. “There have been more than 50 publications on potential mechanisms of benefit of SGLT2 inhibitors in HFrEF, but these are the first randomized, mechanistic data.”
Mechanistic clues follow large cardiovascular outcome trials
Results from a large randomized trial, EMPEROR-Reduced, recently showed that treatment with empagliflozin (Jardiance) on top of standard HFrEF treatment led to significant benefits in patients with or without type 2 diabetes (T2D), compared with placebo, for major cardiovascular and renal endpoints, including the combination of cardiovascular death or hospitalization for heart failure. And results from a second large randomized trial, DAPA-HF, showed similar results with a different drug from the same class, dapagliflozin (Farxiga), in an earlier report.
But while these reports led to quick uptake of these two drugs for the treatment of patients with HFrEF, the means by which these agents exert their HFrEF benefits have been unclear.
“Our study identifies why this drug [empagliflozin] is effective – because it improves heart function, something that has not been understood until now,” Carlos G. Santos-Gallego, MD, lead investigator for the second new report, said in a written statement. “Many doctors are afraid of prescribing a drug they do not understand, and our findings will help clinicians feel more comfortable giving this to patients once approved.”
On the strength of the DAPA-HF results, dapagliflozin received a revised U.S. label in May 2020 that added the indication for treating patients with HFrEF regardless of the whether patients also have T2D, the original indication for prescribing the drug. Many experts anticipate that a similar addition to the label for empagliflozin will soon occur.
EMPA-TROPISM examines patients with no T2D
The single-center study reported by Dr. Santos-Gallego randomized 84 patients with HFrEF and no diabetes to standard treatment with empagliflozin or placebo and measured several parameters in 80 patients who completed the planned 6 months of treatment. The primary endpoints were the changes in both left ventricular end systolic and diastolic volume from baseline in the empagliflozin-treated patients compared with patients on placebo, measured by cardiac MR.
The results showed an average reduction of end systolic volume of 26.6 mL from baseline compared with a small rise in the placebo patients, and an average drop in end diastolic volume of 25.1 mL from baseline compared again with a small increase in the controls. Both differences were statistically significant, reported the senior author of the study, Juan J. Badimon, PhD, in a talk at the virtual scientific sessions of the American Heart Association. Concurrently, the results were published online in the Journal of the American College of Cardiology.
Results from the EMPA-TROPISM study also showed several other significant benefits from empagliflozin treatment, both to left ventricular shape and function as well as to other measures of patient well being. The drug regimen led to an increase in left ventricular ejection fraction, a decrease in left ventricular mass, reduced myocardial fibrosis and aortic stiffness, increased peak oxygen consumption, an increased distance traveled in a 6-minute walk test, and improved quality of life, said Dr. Badimon, professor of medicine and director of the Atherothrombosis Research Unit at the Cardiovascular Institute at the Icahn School of Medicine at Mount Sinai in New York.
SUGAR-DM-HF enrolled only T2D patients
The second study, SUGAR-DM-HF, randomized 105 patients with HFrEF and T2D to treatment with empagliflozin or placebo at any of 15 centers in Scotland, with 92 patients completing the full 36 weeks on treatment. One of the study’s two primary endpoints was the change in left ventricular end systolic volume index, which dropped by an average of 7.9 mL/m2 in patients who received empagliflozin and by 1.5 mL/m2 in the controls, a significant average between-group difference of 6.0 mL/m2, reported Matthew M.Y. Lee, MBChB, at the same meeting.
However, the second primary endpoint, change in left ventricular global longitudinal strain, showed no significant difference in effect on empagliflozin compared with placebo, said Dr. Lee, a cardiologist at the University of Glasgow. Concurrently with his report the results appeared in an article published online in Circulation.
The results also showed a significant drop in left ventricular end diastolic volume index from baseline compared with the control patients, with an average between-group difference in the reduction from baseline of 8.2 mL/m2.
“Reverse cardiac remodeling is a mechanism by which SGLT2 inhibitors reduce heart failure hospitalizations and cardiovascular mortality,” Dr. Lee concluded during his presentation at the meeting.
Although the findings from both studies together provide strong evidence for an effect by empagliflozin on left ventricular shape and function, neither study provides much insight into how this drug exerts these effects. The authors of both studies agreed on several potential explanations, including reductions in cardiac preload and afterload that could reduce left ventricular stretch and volume; a change triggered in myocardial energetics that switches from a metabolism mostly dependent on glucose to one more geared to using fatty acids, ketone bodies, and branched chain amino acids; and a possible drug-induced reduction in oxidative stress and inflammation.
SUGAR-DM-HF was sponsored by a grant from Boehringer Ingelheim, the company that along with Eli Lilly markets empagliflozin (Jardiance). Dr. Lee had no disclosures. Dr. Petrie has been a consultant to Boehringer Ingelheim and Eli Lilly and to several other companies. EMPA-TROPISM was sponsored by a grant from Boehringer Ingelheim. Dr. Badimon and Dr. Santos-Gallego had no disclosures.
FROM AHA 2020
Intravenous iron reduces HF readmissions: AFFIRM-AHF
Iron supplementation reduces heart failure (HF) readmissions in iron-deficient patients hospitalized for acute HF, according to results of the AFFIRM-AHF trial.
After 52 weeks, intravenous ferric carboxymaltose (Ferinject) reduced the risk of total HF hospitalizations and cardiovascular (CV) death by 21% compared with placebo (293 vs 372 events; rate ratio [RR] 0.79; 95% CI, 0.62 - 1.01).
Although the composite primary endpoint failed to achieve statistical significance, it was driven by a significant 26% reduction in the risk of total HF hospital readmissions (P = .013) without an effect on CV mortality (P =.809).
Because the management and follow-up of patients was affected by the COVID-19 pandemic, a prespecified sensitivity analysis was performed that censored patients in each country at the date when its first COVID-19 patient was reported, explained principal investigator Piotr Ponikowski, MD, PhD, Wroclaw Medical University, Wroclaw, Poland.
That analysis revealed a significant 30% reduction in total HF readmissions (P = .005) in patients receiving ferric carboxymaltose (FCM), as well as significant benefits on the primary composite and secondary endpoints.
Notably, 80% of patients required only one or two injections and HF hospitalizations were reduced irrespective of anemia status.
“Iron deficiency should be searched in patients hospitalized with acute heart failure — assessed using a simple blood test — and is now an important therapeutic target,” Ponikowski said at the virtual American Heart Association (AHA) Scientific Sessions 2020.
The results were also published simultaneously in The Lancet.
Iron deficiency is present in up to 70% of patients with acute HF and a predictor of poor outcome, independent of anemia and ejection fraction, he noted.
The FAIR-HF, CONFIRM-HF, and EFFECT-HF trials demonstrated that IV iron supplementation improves exercise capacity, symptoms, and quality of life in iron-deficient HF patients.
However, no such benefit was seen with oral IV in the IRONOUT trial. “So it seems if we are to replace iron, it needs to be done using intravenous therapy,” said John McMurray, MD, University of Glasgow, Scotland, who was invited to discuss the results.
He observed that the reduction in HF hospitalizations in AFFIRM-AHF were relatively modest and that the trial was never expected to show a benefit on CV mortality. Also, the COVID-19 sensitivity analysis providing more convincing effects is a valid approach and one recommended by regulators.
Further, the findings are supported by independent evidence in chronic kidney disease, from the PIVOTAL trial, that intravenous iron reduces HF hospitalizations, McMurray said.
“The million-dollar question, of course, is what will the results of this study mean for the guidelines: I think they probably will change the guidelines,” he said. “Certainly, I hope they will change the US guidelines, which have really given a very lukewarm recommendation for intravenous iron and I think that should probably be stronger.”
In a class IIb recommendation, the 2017 American College of Cardiology/AHA/Heart Failure Society of America heart failure guidelines say intravenous iron “might be reasonable” to improve functional status and quality of life in New York Heart Association class II and III patients with iron deficiency.
The 2016 European Society of Cardiology guidelines include a class IIa recommendation that IV iron “should be considered” in iron-deficient patients with symptomatic HF with reduced ejection fraction.
“This is the first large-scale [trial] of IV supplementation that could potentially change the way we approach patients, particularly those with hospitalized heart failure,” past AHA president Clyde Yancy, MD, MSc, Northwestern University Feinberg School of Medicine in Chicago, said during an earlier press briefing.
He pointed out that clinicians have been circumspect about the early IV iron data. “I have to congratulate you because you’ve changed the narrative,” Yancy said. “We have to start thinking about iron deficiency; we have to think about how we incorporate this in treatment protocols.”
Press briefing panelist Marc Pfeffer, MD, PhD, Brigham and Women’s Hospital and Harvard Medical School in Boston, acknowledged he was among those circumspect.
“I’m no longer a skeptic and I want to congratulate them for showing it’s a risk factor,” he said. “It’s one thing to have a risk factor; it’s another to be a modifiable risk factor and I think that’s what’s so exciting about this.”
The double-blind, phase 4 AFFIRM-AHF trial randomly assigned 1132 patients to receive a bolus injection of ferric carboxymaltose or normal saline before hospital discharge for an acute HF episode. Subsequent treatment was given, as needed, up to 24 weeks post-randomization.
At admission, all patients had left ventricular ejection fractions less than 50% and iron deficiency (serum ferritin <100 ng/mL or serum ferritin 100-299 ng/mL if transferrin saturation <20%).
The modified intention-to-treat (mITT) analysis included 558 FCM patients and 550 controls in whom study treatment was started and for whom at least one post-randomization value was available.
Press briefing discussant Nancy Sweitzer, MD, PhD, director of the University of Arizona’s Sarver Heart Center in Tucson, said AFFIRM-AHF is an “important trial likely to change guidelines” and “targeted one of the highest risk populations we have in heart failure.”
Patients with iron deficiency tend to be elderly with more comorbidities, have longer hospital lengths of stay, and higher readmission rates. “So impacting hospitalizations in this population is incredibly impactful,” she said.
“Awareness and assessment of iron deficiency are an important part of inpatient care of patients with ejection fractions less than or equal to 50% and acute decompensated heart failure, and I think all of us in the community need to pay much more attention to this issue.”
As with any new therapy, there are implementation challenges such as how to monitor patients and deliver the therapy in a cost-effective way, Sweitzer said.
The trial focused on the most vulnerable period for HF patients, but these patients should be rechecked every 3 to 4 months for iron deficiency, Ponikowski observed during the briefing.
“This is a modifiable risk factor,” he said. “We only need to remember, we only need to assess it, and we have a very, very simple tool in our hands. We just need to measure two biomarkers, transferrin saturation and ferritin — that’s all.”
Unanswered questions include the mechanism behind the reduction in hospitalization, the relationship of benefit to hemoglobin levels, and whether there is a differential benefit based on age, presence of ischemia, or sex, especially as women tend to be more severely affected by iron deficiency, Sweitzer said.
During the formal presentation, Ponikowski said the primary endpoint was consistent in subgroup analyses across baseline hemoglobin, estimated glomerular filtration rate, and N-terminal pro-brain natriuretic peptide levels, HF etiology, ejection fraction, and whether HF was diagnosed prior to the index hospitalization.
Treatment with FCM was safe, with no significant differences between the FCM and placebo groups in serious adverse events (45% vs 51%) or adverse events leading to study discontinuation (18% vs 17%), he reported. The most common adverse events were cardiac disorders (40.1% vs 44.3%) and infections (18.2% vs 22%).
AFFIRM-AHF is the first of three ongoing mortality and morbidity trials in heart failure with intravenous ferric carboxymaltose; the others are FAIR-HF2 and HEART-FID. Additional insights are also expected next year on intravenous iron isomaltoside from the Scottish-based IRONMAN trial in 1300 HF patients with iron deficiency.
The study was sponsored by Vifor International. Ponikowski has received research grants and personal fees from Vifor Pharma; and personal fees from Amgen, Bayer, Novartis, Abbott Vascular, Boehringer Ingelheim, Merck, Pfizer, Servier, AstraZeneca, Berlin Chemie, Cibiem, Renal Guard Solutions Bristol-Myers Squibb, and Impulse Dynamics.
Pfeffer reported honoraria from AstraZeneca, Corvidia, GlaxoSmithKline, Jazz, MyoKardia, Novartis, Roche, Sanofi, and Servier; other relationships with DalCor and Novo Nordisk; research grants from Novartis; and an ownership interest in DalCor. Sweitzer reported research payments from Merck and Novartis; and consulting fees from Myocardia.
McMurray reported relationships with Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Cytokinetics, Novartis, and Servier. Yancy reported a relationship with Abbott and JAMA Network.
Lancet. Published online November 13, 2020. Full text
American Heart Association Scientific Sessions 2020: Presented November 13, 2020.
A version of this article originally appeared on Medscape.com.
Iron supplementation reduces heart failure (HF) readmissions in iron-deficient patients hospitalized for acute HF, according to results of the AFFIRM-AHF trial.
After 52 weeks, intravenous ferric carboxymaltose (Ferinject) reduced the risk of total HF hospitalizations and cardiovascular (CV) death by 21% compared with placebo (293 vs 372 events; rate ratio [RR] 0.79; 95% CI, 0.62 - 1.01).
Although the composite primary endpoint failed to achieve statistical significance, it was driven by a significant 26% reduction in the risk of total HF hospital readmissions (P = .013) without an effect on CV mortality (P =.809).
Because the management and follow-up of patients was affected by the COVID-19 pandemic, a prespecified sensitivity analysis was performed that censored patients in each country at the date when its first COVID-19 patient was reported, explained principal investigator Piotr Ponikowski, MD, PhD, Wroclaw Medical University, Wroclaw, Poland.
That analysis revealed a significant 30% reduction in total HF readmissions (P = .005) in patients receiving ferric carboxymaltose (FCM), as well as significant benefits on the primary composite and secondary endpoints.
Notably, 80% of patients required only one or two injections and HF hospitalizations were reduced irrespective of anemia status.
“Iron deficiency should be searched in patients hospitalized with acute heart failure — assessed using a simple blood test — and is now an important therapeutic target,” Ponikowski said at the virtual American Heart Association (AHA) Scientific Sessions 2020.
The results were also published simultaneously in The Lancet.
Iron deficiency is present in up to 70% of patients with acute HF and a predictor of poor outcome, independent of anemia and ejection fraction, he noted.
The FAIR-HF, CONFIRM-HF, and EFFECT-HF trials demonstrated that IV iron supplementation improves exercise capacity, symptoms, and quality of life in iron-deficient HF patients.
However, no such benefit was seen with oral IV in the IRONOUT trial. “So it seems if we are to replace iron, it needs to be done using intravenous therapy,” said John McMurray, MD, University of Glasgow, Scotland, who was invited to discuss the results.
He observed that the reduction in HF hospitalizations in AFFIRM-AHF were relatively modest and that the trial was never expected to show a benefit on CV mortality. Also, the COVID-19 sensitivity analysis providing more convincing effects is a valid approach and one recommended by regulators.
Further, the findings are supported by independent evidence in chronic kidney disease, from the PIVOTAL trial, that intravenous iron reduces HF hospitalizations, McMurray said.
“The million-dollar question, of course, is what will the results of this study mean for the guidelines: I think they probably will change the guidelines,” he said. “Certainly, I hope they will change the US guidelines, which have really given a very lukewarm recommendation for intravenous iron and I think that should probably be stronger.”
In a class IIb recommendation, the 2017 American College of Cardiology/AHA/Heart Failure Society of America heart failure guidelines say intravenous iron “might be reasonable” to improve functional status and quality of life in New York Heart Association class II and III patients with iron deficiency.
The 2016 European Society of Cardiology guidelines include a class IIa recommendation that IV iron “should be considered” in iron-deficient patients with symptomatic HF with reduced ejection fraction.
“This is the first large-scale [trial] of IV supplementation that could potentially change the way we approach patients, particularly those with hospitalized heart failure,” past AHA president Clyde Yancy, MD, MSc, Northwestern University Feinberg School of Medicine in Chicago, said during an earlier press briefing.
He pointed out that clinicians have been circumspect about the early IV iron data. “I have to congratulate you because you’ve changed the narrative,” Yancy said. “We have to start thinking about iron deficiency; we have to think about how we incorporate this in treatment protocols.”
Press briefing panelist Marc Pfeffer, MD, PhD, Brigham and Women’s Hospital and Harvard Medical School in Boston, acknowledged he was among those circumspect.
“I’m no longer a skeptic and I want to congratulate them for showing it’s a risk factor,” he said. “It’s one thing to have a risk factor; it’s another to be a modifiable risk factor and I think that’s what’s so exciting about this.”
The double-blind, phase 4 AFFIRM-AHF trial randomly assigned 1132 patients to receive a bolus injection of ferric carboxymaltose or normal saline before hospital discharge for an acute HF episode. Subsequent treatment was given, as needed, up to 24 weeks post-randomization.
At admission, all patients had left ventricular ejection fractions less than 50% and iron deficiency (serum ferritin <100 ng/mL or serum ferritin 100-299 ng/mL if transferrin saturation <20%).
The modified intention-to-treat (mITT) analysis included 558 FCM patients and 550 controls in whom study treatment was started and for whom at least one post-randomization value was available.
Press briefing discussant Nancy Sweitzer, MD, PhD, director of the University of Arizona’s Sarver Heart Center in Tucson, said AFFIRM-AHF is an “important trial likely to change guidelines” and “targeted one of the highest risk populations we have in heart failure.”
Patients with iron deficiency tend to be elderly with more comorbidities, have longer hospital lengths of stay, and higher readmission rates. “So impacting hospitalizations in this population is incredibly impactful,” she said.
“Awareness and assessment of iron deficiency are an important part of inpatient care of patients with ejection fractions less than or equal to 50% and acute decompensated heart failure, and I think all of us in the community need to pay much more attention to this issue.”
As with any new therapy, there are implementation challenges such as how to monitor patients and deliver the therapy in a cost-effective way, Sweitzer said.
The trial focused on the most vulnerable period for HF patients, but these patients should be rechecked every 3 to 4 months for iron deficiency, Ponikowski observed during the briefing.
“This is a modifiable risk factor,” he said. “We only need to remember, we only need to assess it, and we have a very, very simple tool in our hands. We just need to measure two biomarkers, transferrin saturation and ferritin — that’s all.”
Unanswered questions include the mechanism behind the reduction in hospitalization, the relationship of benefit to hemoglobin levels, and whether there is a differential benefit based on age, presence of ischemia, or sex, especially as women tend to be more severely affected by iron deficiency, Sweitzer said.
During the formal presentation, Ponikowski said the primary endpoint was consistent in subgroup analyses across baseline hemoglobin, estimated glomerular filtration rate, and N-terminal pro-brain natriuretic peptide levels, HF etiology, ejection fraction, and whether HF was diagnosed prior to the index hospitalization.
Treatment with FCM was safe, with no significant differences between the FCM and placebo groups in serious adverse events (45% vs 51%) or adverse events leading to study discontinuation (18% vs 17%), he reported. The most common adverse events were cardiac disorders (40.1% vs 44.3%) and infections (18.2% vs 22%).
AFFIRM-AHF is the first of three ongoing mortality and morbidity trials in heart failure with intravenous ferric carboxymaltose; the others are FAIR-HF2 and HEART-FID. Additional insights are also expected next year on intravenous iron isomaltoside from the Scottish-based IRONMAN trial in 1300 HF patients with iron deficiency.
The study was sponsored by Vifor International. Ponikowski has received research grants and personal fees from Vifor Pharma; and personal fees from Amgen, Bayer, Novartis, Abbott Vascular, Boehringer Ingelheim, Merck, Pfizer, Servier, AstraZeneca, Berlin Chemie, Cibiem, Renal Guard Solutions Bristol-Myers Squibb, and Impulse Dynamics.
Pfeffer reported honoraria from AstraZeneca, Corvidia, GlaxoSmithKline, Jazz, MyoKardia, Novartis, Roche, Sanofi, and Servier; other relationships with DalCor and Novo Nordisk; research grants from Novartis; and an ownership interest in DalCor. Sweitzer reported research payments from Merck and Novartis; and consulting fees from Myocardia.
McMurray reported relationships with Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Cytokinetics, Novartis, and Servier. Yancy reported a relationship with Abbott and JAMA Network.
Lancet. Published online November 13, 2020. Full text
American Heart Association Scientific Sessions 2020: Presented November 13, 2020.
A version of this article originally appeared on Medscape.com.
Iron supplementation reduces heart failure (HF) readmissions in iron-deficient patients hospitalized for acute HF, according to results of the AFFIRM-AHF trial.
After 52 weeks, intravenous ferric carboxymaltose (Ferinject) reduced the risk of total HF hospitalizations and cardiovascular (CV) death by 21% compared with placebo (293 vs 372 events; rate ratio [RR] 0.79; 95% CI, 0.62 - 1.01).
Although the composite primary endpoint failed to achieve statistical significance, it was driven by a significant 26% reduction in the risk of total HF hospital readmissions (P = .013) without an effect on CV mortality (P =.809).
Because the management and follow-up of patients was affected by the COVID-19 pandemic, a prespecified sensitivity analysis was performed that censored patients in each country at the date when its first COVID-19 patient was reported, explained principal investigator Piotr Ponikowski, MD, PhD, Wroclaw Medical University, Wroclaw, Poland.
That analysis revealed a significant 30% reduction in total HF readmissions (P = .005) in patients receiving ferric carboxymaltose (FCM), as well as significant benefits on the primary composite and secondary endpoints.
Notably, 80% of patients required only one or two injections and HF hospitalizations were reduced irrespective of anemia status.
“Iron deficiency should be searched in patients hospitalized with acute heart failure — assessed using a simple blood test — and is now an important therapeutic target,” Ponikowski said at the virtual American Heart Association (AHA) Scientific Sessions 2020.
The results were also published simultaneously in The Lancet.
Iron deficiency is present in up to 70% of patients with acute HF and a predictor of poor outcome, independent of anemia and ejection fraction, he noted.
The FAIR-HF, CONFIRM-HF, and EFFECT-HF trials demonstrated that IV iron supplementation improves exercise capacity, symptoms, and quality of life in iron-deficient HF patients.
However, no such benefit was seen with oral IV in the IRONOUT trial. “So it seems if we are to replace iron, it needs to be done using intravenous therapy,” said John McMurray, MD, University of Glasgow, Scotland, who was invited to discuss the results.
He observed that the reduction in HF hospitalizations in AFFIRM-AHF were relatively modest and that the trial was never expected to show a benefit on CV mortality. Also, the COVID-19 sensitivity analysis providing more convincing effects is a valid approach and one recommended by regulators.
Further, the findings are supported by independent evidence in chronic kidney disease, from the PIVOTAL trial, that intravenous iron reduces HF hospitalizations, McMurray said.
“The million-dollar question, of course, is what will the results of this study mean for the guidelines: I think they probably will change the guidelines,” he said. “Certainly, I hope they will change the US guidelines, which have really given a very lukewarm recommendation for intravenous iron and I think that should probably be stronger.”
In a class IIb recommendation, the 2017 American College of Cardiology/AHA/Heart Failure Society of America heart failure guidelines say intravenous iron “might be reasonable” to improve functional status and quality of life in New York Heart Association class II and III patients with iron deficiency.
The 2016 European Society of Cardiology guidelines include a class IIa recommendation that IV iron “should be considered” in iron-deficient patients with symptomatic HF with reduced ejection fraction.
“This is the first large-scale [trial] of IV supplementation that could potentially change the way we approach patients, particularly those with hospitalized heart failure,” past AHA president Clyde Yancy, MD, MSc, Northwestern University Feinberg School of Medicine in Chicago, said during an earlier press briefing.
He pointed out that clinicians have been circumspect about the early IV iron data. “I have to congratulate you because you’ve changed the narrative,” Yancy said. “We have to start thinking about iron deficiency; we have to think about how we incorporate this in treatment protocols.”
Press briefing panelist Marc Pfeffer, MD, PhD, Brigham and Women’s Hospital and Harvard Medical School in Boston, acknowledged he was among those circumspect.
“I’m no longer a skeptic and I want to congratulate them for showing it’s a risk factor,” he said. “It’s one thing to have a risk factor; it’s another to be a modifiable risk factor and I think that’s what’s so exciting about this.”
The double-blind, phase 4 AFFIRM-AHF trial randomly assigned 1132 patients to receive a bolus injection of ferric carboxymaltose or normal saline before hospital discharge for an acute HF episode. Subsequent treatment was given, as needed, up to 24 weeks post-randomization.
At admission, all patients had left ventricular ejection fractions less than 50% and iron deficiency (serum ferritin <100 ng/mL or serum ferritin 100-299 ng/mL if transferrin saturation <20%).
The modified intention-to-treat (mITT) analysis included 558 FCM patients and 550 controls in whom study treatment was started and for whom at least one post-randomization value was available.
Press briefing discussant Nancy Sweitzer, MD, PhD, director of the University of Arizona’s Sarver Heart Center in Tucson, said AFFIRM-AHF is an “important trial likely to change guidelines” and “targeted one of the highest risk populations we have in heart failure.”
Patients with iron deficiency tend to be elderly with more comorbidities, have longer hospital lengths of stay, and higher readmission rates. “So impacting hospitalizations in this population is incredibly impactful,” she said.
“Awareness and assessment of iron deficiency are an important part of inpatient care of patients with ejection fractions less than or equal to 50% and acute decompensated heart failure, and I think all of us in the community need to pay much more attention to this issue.”
As with any new therapy, there are implementation challenges such as how to monitor patients and deliver the therapy in a cost-effective way, Sweitzer said.
The trial focused on the most vulnerable period for HF patients, but these patients should be rechecked every 3 to 4 months for iron deficiency, Ponikowski observed during the briefing.
“This is a modifiable risk factor,” he said. “We only need to remember, we only need to assess it, and we have a very, very simple tool in our hands. We just need to measure two biomarkers, transferrin saturation and ferritin — that’s all.”
Unanswered questions include the mechanism behind the reduction in hospitalization, the relationship of benefit to hemoglobin levels, and whether there is a differential benefit based on age, presence of ischemia, or sex, especially as women tend to be more severely affected by iron deficiency, Sweitzer said.
During the formal presentation, Ponikowski said the primary endpoint was consistent in subgroup analyses across baseline hemoglobin, estimated glomerular filtration rate, and N-terminal pro-brain natriuretic peptide levels, HF etiology, ejection fraction, and whether HF was diagnosed prior to the index hospitalization.
Treatment with FCM was safe, with no significant differences between the FCM and placebo groups in serious adverse events (45% vs 51%) or adverse events leading to study discontinuation (18% vs 17%), he reported. The most common adverse events were cardiac disorders (40.1% vs 44.3%) and infections (18.2% vs 22%).
AFFIRM-AHF is the first of three ongoing mortality and morbidity trials in heart failure with intravenous ferric carboxymaltose; the others are FAIR-HF2 and HEART-FID. Additional insights are also expected next year on intravenous iron isomaltoside from the Scottish-based IRONMAN trial in 1300 HF patients with iron deficiency.
The study was sponsored by Vifor International. Ponikowski has received research grants and personal fees from Vifor Pharma; and personal fees from Amgen, Bayer, Novartis, Abbott Vascular, Boehringer Ingelheim, Merck, Pfizer, Servier, AstraZeneca, Berlin Chemie, Cibiem, Renal Guard Solutions Bristol-Myers Squibb, and Impulse Dynamics.
Pfeffer reported honoraria from AstraZeneca, Corvidia, GlaxoSmithKline, Jazz, MyoKardia, Novartis, Roche, Sanofi, and Servier; other relationships with DalCor and Novo Nordisk; research grants from Novartis; and an ownership interest in DalCor. Sweitzer reported research payments from Merck and Novartis; and consulting fees from Myocardia.
McMurray reported relationships with Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Cytokinetics, Novartis, and Servier. Yancy reported a relationship with Abbott and JAMA Network.
Lancet. Published online November 13, 2020. Full text
American Heart Association Scientific Sessions 2020: Presented November 13, 2020.
A version of this article originally appeared on Medscape.com.
FROM AHA 2020