User login
‘Excess’ deaths surging, but why?
This transcript has been edited for clarity.
“Excess deaths.” You’ve heard the phrase countless times by now. It is one of the myriad of previously esoteric epidemiology terms that the pandemic brought squarely into the zeitgeist.
As a sort of standard candle of the performance of a state or a region or a country in terms of health care, it has a lot of utility – if for nothing more than Monday-morning quarterbacking. But this week, I want to dig in on the concept a bit because, according to a new study, the excess death gap between the United States and Western Europe has never been higher.
You might imagine that the best way to figure this out is for some group of intelligent people to review each death and decide, somehow, whether it was expected or not. But aside from being impractical, this would end up being somewhat subjective. That older person who died from pneumonia – was that an expected death? Could it have been avoided?
Rather, the calculation of excess mortality relies on large numbers and statistical inference to compare an expected number of deaths with those that are observed.
The difference is excess mortality, even if you can never be sure whether any particular death was expected or not.
As always, however, the devil is in the details. What data do you use to define the expected number of deaths?
There are options here. Probably the most straightforward analysis uses past data from the country of interest. You look at annual deaths over some historical period of time and compare those numbers with the rates today. Two issues need to be accounted for here: population growth – a larger population will have more deaths, so you need to adjust the historical population with current levels, and demographic shifts – an older or more male population will have more deaths, so you need to adjust for that as well.
But provided you take care of those factors, you can estimate fairly well how many deaths you can expect to see in any given period of time.
Still, you should see right away that excess mortality is a relative concept. If you think that, just perhaps, the United States has some systematic failure to deliver care that has been stable and persistent over time, you wouldn’t capture that failing in an excess mortality calculation that uses U.S. historical data as the baseline.
The best way to get around that is to use data from other countries, and that’s just what this article – a rare single-author piece by Patrick Heuveline – does, calculating excess deaths in the United States by standardizing our mortality rates to the five largest Western European countries: the United Kingdom, France, Germany, Italy, and Spain.
Controlling for the differences in the demographics of that European population, here is the expected number of deaths in the United States over the past 5 years.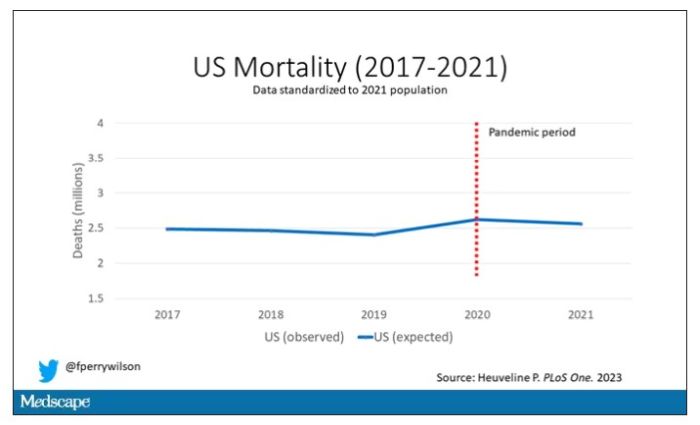
Note that there is a small uptick in expected deaths in 2020, reflecting the pandemic, which returns to baseline levels by 2021. This is because that’s what happened in Europe; by 2021, the excess mortality due to COVID-19 was quite low.
Here are the actual deaths in the US during that time.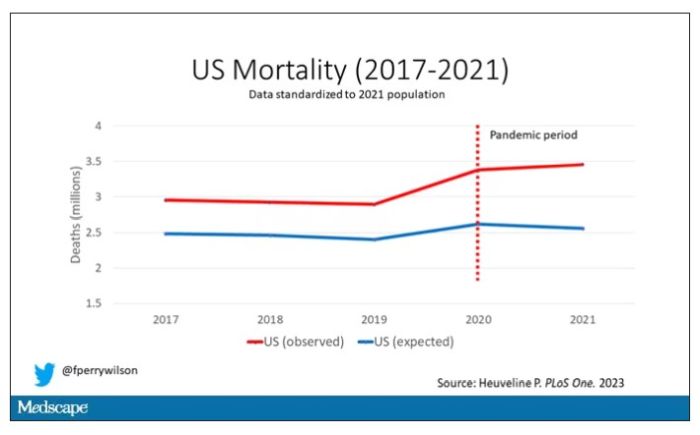
Highlighted here in green, then, is the excess mortality over time in the United States.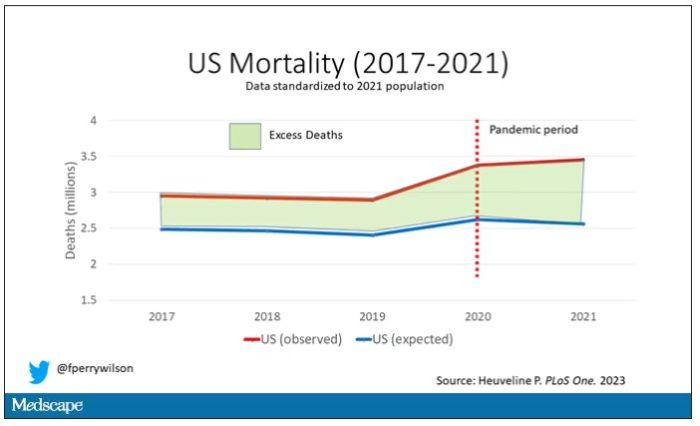
There are some fascinating and concerning findings here.
First of all, you can see that even before the pandemic, the United States has an excess mortality problem. This is not entirely a surprise; we’ve known that so-called “deaths of despair,” those due to alcohol abuse, drug overdoses, and suicide, are at an all-time high and tend to affect a “prime of life” population that would not otherwise be expected to die. In fact, fully 50% of the excess deaths in the United States occur in those between ages 15 and 64.
Excess deaths are also a concerning percentage of total deaths. In 2017, 17% of total deaths in the United States could be considered “excess.” In 2021, that number had doubled to 35%. Nearly 900,000 individuals in the United States died in 2021 who perhaps didn’t need to.
The obvious culprit to blame here is COVID, but COVID-associated excess deaths only explain about 50% of the excess we see in 2021. The rest reflect something even more concerning: a worsening of the failures of the past, perhaps exacerbated by the pandemic but not due to the virus itself.
Of course, we started this discussion acknowledging that the calculation of excess mortality is exquisitely dependent on how you model the expected number of deaths, and I’m sure some will take issue with the use of European numbers when applied to Americans. After all, Europe has, by and large, a robust public health service, socialized medicine, and healthcare that does not run the risk of bankrupting its citizens. How can we compare our outcomes to a place like that?
How indeed.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale University’s Clinical and Translational Research Accelerator in New Haven,Conn. He reported no relevant conflicts of interest.
A version of this article originally appeared on Medscape.com.
This transcript has been edited for clarity.
“Excess deaths.” You’ve heard the phrase countless times by now. It is one of the myriad of previously esoteric epidemiology terms that the pandemic brought squarely into the zeitgeist.
As a sort of standard candle of the performance of a state or a region or a country in terms of health care, it has a lot of utility – if for nothing more than Monday-morning quarterbacking. But this week, I want to dig in on the concept a bit because, according to a new study, the excess death gap between the United States and Western Europe has never been higher.
You might imagine that the best way to figure this out is for some group of intelligent people to review each death and decide, somehow, whether it was expected or not. But aside from being impractical, this would end up being somewhat subjective. That older person who died from pneumonia – was that an expected death? Could it have been avoided?
Rather, the calculation of excess mortality relies on large numbers and statistical inference to compare an expected number of deaths with those that are observed.
The difference is excess mortality, even if you can never be sure whether any particular death was expected or not.
As always, however, the devil is in the details. What data do you use to define the expected number of deaths?
There are options here. Probably the most straightforward analysis uses past data from the country of interest. You look at annual deaths over some historical period of time and compare those numbers with the rates today. Two issues need to be accounted for here: population growth – a larger population will have more deaths, so you need to adjust the historical population with current levels, and demographic shifts – an older or more male population will have more deaths, so you need to adjust for that as well.
But provided you take care of those factors, you can estimate fairly well how many deaths you can expect to see in any given period of time.
Still, you should see right away that excess mortality is a relative concept. If you think that, just perhaps, the United States has some systematic failure to deliver care that has been stable and persistent over time, you wouldn’t capture that failing in an excess mortality calculation that uses U.S. historical data as the baseline.
The best way to get around that is to use data from other countries, and that’s just what this article – a rare single-author piece by Patrick Heuveline – does, calculating excess deaths in the United States by standardizing our mortality rates to the five largest Western European countries: the United Kingdom, France, Germany, Italy, and Spain.
Controlling for the differences in the demographics of that European population, here is the expected number of deaths in the United States over the past 5 years.
Note that there is a small uptick in expected deaths in 2020, reflecting the pandemic, which returns to baseline levels by 2021. This is because that’s what happened in Europe; by 2021, the excess mortality due to COVID-19 was quite low.
Here are the actual deaths in the US during that time.
Highlighted here in green, then, is the excess mortality over time in the United States.
There are some fascinating and concerning findings here.
First of all, you can see that even before the pandemic, the United States has an excess mortality problem. This is not entirely a surprise; we’ve known that so-called “deaths of despair,” those due to alcohol abuse, drug overdoses, and suicide, are at an all-time high and tend to affect a “prime of life” population that would not otherwise be expected to die. In fact, fully 50% of the excess deaths in the United States occur in those between ages 15 and 64.
Excess deaths are also a concerning percentage of total deaths. In 2017, 17% of total deaths in the United States could be considered “excess.” In 2021, that number had doubled to 35%. Nearly 900,000 individuals in the United States died in 2021 who perhaps didn’t need to.
The obvious culprit to blame here is COVID, but COVID-associated excess deaths only explain about 50% of the excess we see in 2021. The rest reflect something even more concerning: a worsening of the failures of the past, perhaps exacerbated by the pandemic but not due to the virus itself.
Of course, we started this discussion acknowledging that the calculation of excess mortality is exquisitely dependent on how you model the expected number of deaths, and I’m sure some will take issue with the use of European numbers when applied to Americans. After all, Europe has, by and large, a robust public health service, socialized medicine, and healthcare that does not run the risk of bankrupting its citizens. How can we compare our outcomes to a place like that?
How indeed.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale University’s Clinical and Translational Research Accelerator in New Haven,Conn. He reported no relevant conflicts of interest.
A version of this article originally appeared on Medscape.com.
This transcript has been edited for clarity.
“Excess deaths.” You’ve heard the phrase countless times by now. It is one of the myriad of previously esoteric epidemiology terms that the pandemic brought squarely into the zeitgeist.
As a sort of standard candle of the performance of a state or a region or a country in terms of health care, it has a lot of utility – if for nothing more than Monday-morning quarterbacking. But this week, I want to dig in on the concept a bit because, according to a new study, the excess death gap between the United States and Western Europe has never been higher.
You might imagine that the best way to figure this out is for some group of intelligent people to review each death and decide, somehow, whether it was expected or not. But aside from being impractical, this would end up being somewhat subjective. That older person who died from pneumonia – was that an expected death? Could it have been avoided?
Rather, the calculation of excess mortality relies on large numbers and statistical inference to compare an expected number of deaths with those that are observed.
The difference is excess mortality, even if you can never be sure whether any particular death was expected or not.
As always, however, the devil is in the details. What data do you use to define the expected number of deaths?
There are options here. Probably the most straightforward analysis uses past data from the country of interest. You look at annual deaths over some historical period of time and compare those numbers with the rates today. Two issues need to be accounted for here: population growth – a larger population will have more deaths, so you need to adjust the historical population with current levels, and demographic shifts – an older or more male population will have more deaths, so you need to adjust for that as well.
But provided you take care of those factors, you can estimate fairly well how many deaths you can expect to see in any given period of time.
Still, you should see right away that excess mortality is a relative concept. If you think that, just perhaps, the United States has some systematic failure to deliver care that has been stable and persistent over time, you wouldn’t capture that failing in an excess mortality calculation that uses U.S. historical data as the baseline.
The best way to get around that is to use data from other countries, and that’s just what this article – a rare single-author piece by Patrick Heuveline – does, calculating excess deaths in the United States by standardizing our mortality rates to the five largest Western European countries: the United Kingdom, France, Germany, Italy, and Spain.
Controlling for the differences in the demographics of that European population, here is the expected number of deaths in the United States over the past 5 years.
Note that there is a small uptick in expected deaths in 2020, reflecting the pandemic, which returns to baseline levels by 2021. This is because that’s what happened in Europe; by 2021, the excess mortality due to COVID-19 was quite low.
Here are the actual deaths in the US during that time.
Highlighted here in green, then, is the excess mortality over time in the United States.
There are some fascinating and concerning findings here.
First of all, you can see that even before the pandemic, the United States has an excess mortality problem. This is not entirely a surprise; we’ve known that so-called “deaths of despair,” those due to alcohol abuse, drug overdoses, and suicide, are at an all-time high and tend to affect a “prime of life” population that would not otherwise be expected to die. In fact, fully 50% of the excess deaths in the United States occur in those between ages 15 and 64.
Excess deaths are also a concerning percentage of total deaths. In 2017, 17% of total deaths in the United States could be considered “excess.” In 2021, that number had doubled to 35%. Nearly 900,000 individuals in the United States died in 2021 who perhaps didn’t need to.
The obvious culprit to blame here is COVID, but COVID-associated excess deaths only explain about 50% of the excess we see in 2021. The rest reflect something even more concerning: a worsening of the failures of the past, perhaps exacerbated by the pandemic but not due to the virus itself.
Of course, we started this discussion acknowledging that the calculation of excess mortality is exquisitely dependent on how you model the expected number of deaths, and I’m sure some will take issue with the use of European numbers when applied to Americans. After all, Europe has, by and large, a robust public health service, socialized medicine, and healthcare that does not run the risk of bankrupting its citizens. How can we compare our outcomes to a place like that?
How indeed.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale University’s Clinical and Translational Research Accelerator in New Haven,Conn. He reported no relevant conflicts of interest.
A version of this article originally appeared on Medscape.com.
Celebrity death finally solved – with locks of hair
This transcript has been edited for clarity.
I’m going to open this week with a case.
A 56-year-old musician presents with diffuse abdominal pain, cramping, and jaundice. His medical history is notable for years of diffuse abdominal complaints, characterized by disabling bouts of diarrhea.
In addition to the jaundice, this acute illness was accompanied by fever as well as diffuse edema and ascites. The patient underwent several abdominal paracenteses to drain excess fluid. One consulting physician administered alcohol to relieve pain, to little avail.
The patient succumbed to his illness. An autopsy showed diffuse liver injury, as well as papillary necrosis of the kidneys. Notably, the nerves of his auditory canal were noted to be thickened, along with the bony part of the skull, consistent with Paget disease of the bone and explaining, potentially, why the talented musician had gone deaf at such a young age.
An interesting note on social history: The patient had apparently developed some feelings for the niece of that doctor who prescribed alcohol. Her name was Therese, perhaps mistranscribed as Elise, and it seems that he may have written this song for her.
We’re talking about this paper in Current Biology, by Tristan Begg and colleagues, which gives us a look into the very genome of what some would argue is the world’s greatest composer.
The ability to extract DNA from older specimens has transformed the fields of anthropology, archaeology, and history, and now, perhaps, musicology as well.
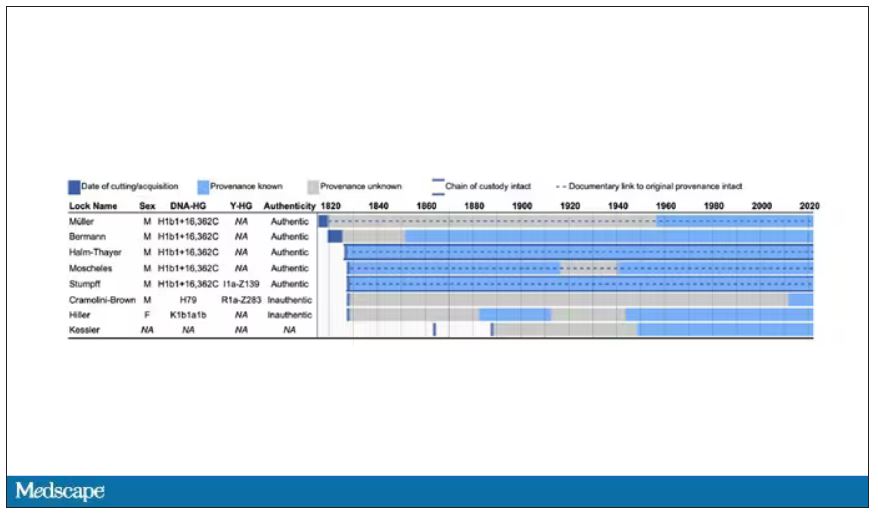
The researchers identified eight locks of hair in private and public collections, all attributed to the maestro.
Four of the samples had an intact chain of custody from the time the hair was cut. DNA sequencing on these four and an additional one of the eight locks came from the same individual, a male of European heritage.
The three locks with less documentation came from three other unrelated individuals. Interestingly, analysis of one of those hair samples – the so-called Hiller Lock – had shown high levels of lead, leading historians to speculate that lead poisoning could account for some of Beethoven’s symptoms.
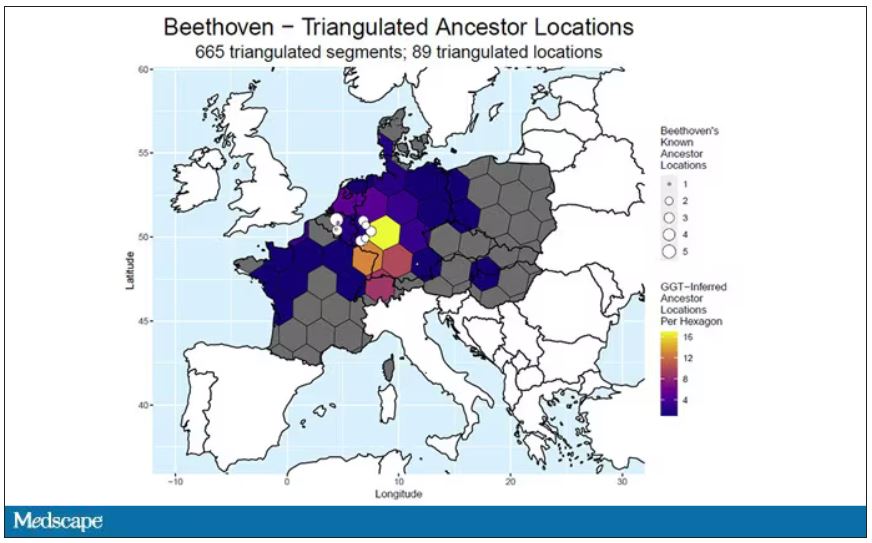
DNA analysis of that hair reveals it to have come from a woman likely of North African, Middle Eastern, or Jewish ancestry. We can no longer presume that plumbism was involved in Beethoven’s death. Beethoven’s ancestry turns out to be less exotic and maps quite well to ethnic German populations today.
In fact, there are van Beethovens alive as we speak, primarily in Belgium. Genealogic records suggest that these van Beethovens share a common ancestor with the virtuoso composer, a man by the name of Aert van Beethoven.
But the DNA reveals a scandal.
The Y-chromosome that Beethoven inherited was not Aert van Beethoven’s. Questions of Beethoven’s paternity have been raised before, but this evidence strongly suggests an extramarital paternity event, at least in the generations preceding his birth. That’s right – Beethoven may not have been a Beethoven.
With five locks now essentially certain to have come from Beethoven himself, the authors could use DNA analysis to try to explain three significant health problems he experienced throughout his life and death: his hearing loss, his terrible gastrointestinal issues, and his liver failure.
Let’s start with the most disappointing results, explanations for his hearing loss. No genetic cause was forthcoming, though the authors note that they have little to go on in regard to the genetic risk for otosclerosis, to which his hearing loss has often been attributed. Lead poisoning is, of course, possible here, though this report focuses only on genetics – there was no testing for lead – and as I mentioned, the lock that was strongly lead-positive in prior studies is almost certainly inauthentic.
What about his lifelong GI complaints? Some have suggested celiac disease or lactose intolerance as explanations. These can essentially be ruled out by the genetic analysis, which shows no risk alleles for celiac disease and the presence of the lactase-persistence gene which confers the ability to metabolize lactose throughout one’s life. IBS is harder to assess genetically, but for what it’s worth, he scored quite low on a polygenic risk score for the condition, in just the 9th percentile of risk. We should probably be looking elsewhere to explain the GI distress.
The genetic information bore much more fruit in regard to his liver disease. Remember that Beethoven’s autopsy showed cirrhosis. His polygenic risk score for liver cirrhosis puts him in the 96th percentile of risk. He was also heterozygous for two variants that can cause hereditary hemochromatosis. The risk for cirrhosis among those with these variants is increased by the use of alcohol. And historical accounts are quite clear that Beethoven consumed more than his share.
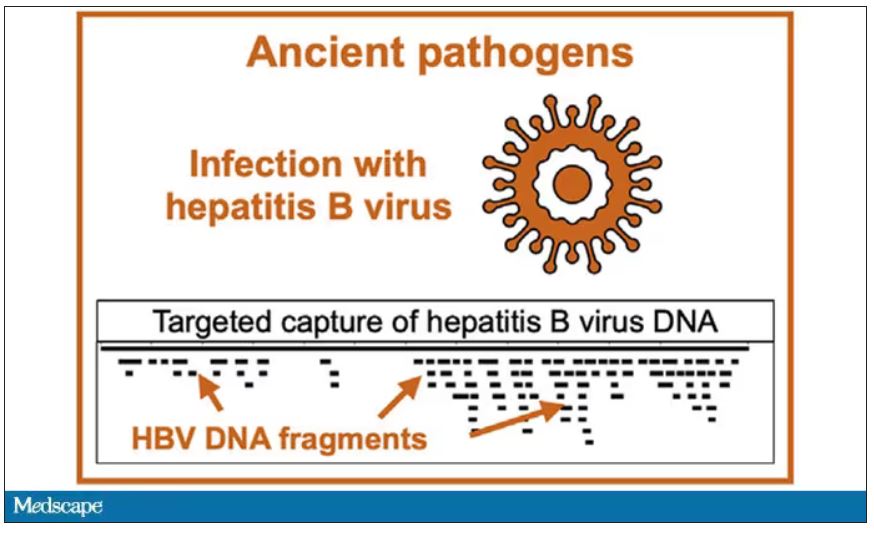
But it wasn’t just Beethoven’s DNA in these hair follicles. Analysis of a follicle from later in his life revealed the unmistakable presence of hepatitis B virus. Endemic in Europe at the time, this was a common cause of liver failure and is likely to have contributed to, if not directly caused, Beethoven’s demise.
It’s hard to read these results and not marvel at the fact that, two centuries after his death, our fascination with Beethoven has led us to probe every corner of his life – his letters, his writings, his medical records, and now his very DNA. What are we actually looking for? Is it relevant to us today what caused his hearing loss? His stomach troubles? Even his death? Will it help any patients in the future? I propose that what we are actually trying to understand is something ineffable: Genius of magnitude that is rarely seen in one or many lifetimes. And our scientific tools, as sharp as they may have become, are still far too blunt to probe the depths of that transcendence.
In any case, friends, no more of these sounds. Let us sing more cheerful songs, more full of joy.
For Medscape, I’m Perry Wilson.
Dr. Wilson is associate professor, department of medicine, and director, Clinical and Translational Research Accelerator, at Yale University, New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’m going to open this week with a case.
A 56-year-old musician presents with diffuse abdominal pain, cramping, and jaundice. His medical history is notable for years of diffuse abdominal complaints, characterized by disabling bouts of diarrhea.
In addition to the jaundice, this acute illness was accompanied by fever as well as diffuse edema and ascites. The patient underwent several abdominal paracenteses to drain excess fluid. One consulting physician administered alcohol to relieve pain, to little avail.
The patient succumbed to his illness. An autopsy showed diffuse liver injury, as well as papillary necrosis of the kidneys. Notably, the nerves of his auditory canal were noted to be thickened, along with the bony part of the skull, consistent with Paget disease of the bone and explaining, potentially, why the talented musician had gone deaf at such a young age.
An interesting note on social history: The patient had apparently developed some feelings for the niece of that doctor who prescribed alcohol. Her name was Therese, perhaps mistranscribed as Elise, and it seems that he may have written this song for her.
We’re talking about this paper in Current Biology, by Tristan Begg and colleagues, which gives us a look into the very genome of what some would argue is the world’s greatest composer.
The ability to extract DNA from older specimens has transformed the fields of anthropology, archaeology, and history, and now, perhaps, musicology as well.
The researchers identified eight locks of hair in private and public collections, all attributed to the maestro.
Four of the samples had an intact chain of custody from the time the hair was cut. DNA sequencing on these four and an additional one of the eight locks came from the same individual, a male of European heritage.
The three locks with less documentation came from three other unrelated individuals. Interestingly, analysis of one of those hair samples – the so-called Hiller Lock – had shown high levels of lead, leading historians to speculate that lead poisoning could account for some of Beethoven’s symptoms.
DNA analysis of that hair reveals it to have come from a woman likely of North African, Middle Eastern, or Jewish ancestry. We can no longer presume that plumbism was involved in Beethoven’s death. Beethoven’s ancestry turns out to be less exotic and maps quite well to ethnic German populations today.
In fact, there are van Beethovens alive as we speak, primarily in Belgium. Genealogic records suggest that these van Beethovens share a common ancestor with the virtuoso composer, a man by the name of Aert van Beethoven.
But the DNA reveals a scandal.
The Y-chromosome that Beethoven inherited was not Aert van Beethoven’s. Questions of Beethoven’s paternity have been raised before, but this evidence strongly suggests an extramarital paternity event, at least in the generations preceding his birth. That’s right – Beethoven may not have been a Beethoven.
With five locks now essentially certain to have come from Beethoven himself, the authors could use DNA analysis to try to explain three significant health problems he experienced throughout his life and death: his hearing loss, his terrible gastrointestinal issues, and his liver failure.
Let’s start with the most disappointing results, explanations for his hearing loss. No genetic cause was forthcoming, though the authors note that they have little to go on in regard to the genetic risk for otosclerosis, to which his hearing loss has often been attributed. Lead poisoning is, of course, possible here, though this report focuses only on genetics – there was no testing for lead – and as I mentioned, the lock that was strongly lead-positive in prior studies is almost certainly inauthentic.
What about his lifelong GI complaints? Some have suggested celiac disease or lactose intolerance as explanations. These can essentially be ruled out by the genetic analysis, which shows no risk alleles for celiac disease and the presence of the lactase-persistence gene which confers the ability to metabolize lactose throughout one’s life. IBS is harder to assess genetically, but for what it’s worth, he scored quite low on a polygenic risk score for the condition, in just the 9th percentile of risk. We should probably be looking elsewhere to explain the GI distress.
The genetic information bore much more fruit in regard to his liver disease. Remember that Beethoven’s autopsy showed cirrhosis. His polygenic risk score for liver cirrhosis puts him in the 96th percentile of risk. He was also heterozygous for two variants that can cause hereditary hemochromatosis. The risk for cirrhosis among those with these variants is increased by the use of alcohol. And historical accounts are quite clear that Beethoven consumed more than his share.
But it wasn’t just Beethoven’s DNA in these hair follicles. Analysis of a follicle from later in his life revealed the unmistakable presence of hepatitis B virus. Endemic in Europe at the time, this was a common cause of liver failure and is likely to have contributed to, if not directly caused, Beethoven’s demise.
It’s hard to read these results and not marvel at the fact that, two centuries after his death, our fascination with Beethoven has led us to probe every corner of his life – his letters, his writings, his medical records, and now his very DNA. What are we actually looking for? Is it relevant to us today what caused his hearing loss? His stomach troubles? Even his death? Will it help any patients in the future? I propose that what we are actually trying to understand is something ineffable: Genius of magnitude that is rarely seen in one or many lifetimes. And our scientific tools, as sharp as they may have become, are still far too blunt to probe the depths of that transcendence.
In any case, friends, no more of these sounds. Let us sing more cheerful songs, more full of joy.
For Medscape, I’m Perry Wilson.
Dr. Wilson is associate professor, department of medicine, and director, Clinical and Translational Research Accelerator, at Yale University, New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’m going to open this week with a case.
A 56-year-old musician presents with diffuse abdominal pain, cramping, and jaundice. His medical history is notable for years of diffuse abdominal complaints, characterized by disabling bouts of diarrhea.
In addition to the jaundice, this acute illness was accompanied by fever as well as diffuse edema and ascites. The patient underwent several abdominal paracenteses to drain excess fluid. One consulting physician administered alcohol to relieve pain, to little avail.
The patient succumbed to his illness. An autopsy showed diffuse liver injury, as well as papillary necrosis of the kidneys. Notably, the nerves of his auditory canal were noted to be thickened, along with the bony part of the skull, consistent with Paget disease of the bone and explaining, potentially, why the talented musician had gone deaf at such a young age.
An interesting note on social history: The patient had apparently developed some feelings for the niece of that doctor who prescribed alcohol. Her name was Therese, perhaps mistranscribed as Elise, and it seems that he may have written this song for her.
We’re talking about this paper in Current Biology, by Tristan Begg and colleagues, which gives us a look into the very genome of what some would argue is the world’s greatest composer.
The ability to extract DNA from older specimens has transformed the fields of anthropology, archaeology, and history, and now, perhaps, musicology as well.
The researchers identified eight locks of hair in private and public collections, all attributed to the maestro.
Four of the samples had an intact chain of custody from the time the hair was cut. DNA sequencing on these four and an additional one of the eight locks came from the same individual, a male of European heritage.
The three locks with less documentation came from three other unrelated individuals. Interestingly, analysis of one of those hair samples – the so-called Hiller Lock – had shown high levels of lead, leading historians to speculate that lead poisoning could account for some of Beethoven’s symptoms.
DNA analysis of that hair reveals it to have come from a woman likely of North African, Middle Eastern, or Jewish ancestry. We can no longer presume that plumbism was involved in Beethoven’s death. Beethoven’s ancestry turns out to be less exotic and maps quite well to ethnic German populations today.
In fact, there are van Beethovens alive as we speak, primarily in Belgium. Genealogic records suggest that these van Beethovens share a common ancestor with the virtuoso composer, a man by the name of Aert van Beethoven.
But the DNA reveals a scandal.
The Y-chromosome that Beethoven inherited was not Aert van Beethoven’s. Questions of Beethoven’s paternity have been raised before, but this evidence strongly suggests an extramarital paternity event, at least in the generations preceding his birth. That’s right – Beethoven may not have been a Beethoven.
With five locks now essentially certain to have come from Beethoven himself, the authors could use DNA analysis to try to explain three significant health problems he experienced throughout his life and death: his hearing loss, his terrible gastrointestinal issues, and his liver failure.
Let’s start with the most disappointing results, explanations for his hearing loss. No genetic cause was forthcoming, though the authors note that they have little to go on in regard to the genetic risk for otosclerosis, to which his hearing loss has often been attributed. Lead poisoning is, of course, possible here, though this report focuses only on genetics – there was no testing for lead – and as I mentioned, the lock that was strongly lead-positive in prior studies is almost certainly inauthentic.
What about his lifelong GI complaints? Some have suggested celiac disease or lactose intolerance as explanations. These can essentially be ruled out by the genetic analysis, which shows no risk alleles for celiac disease and the presence of the lactase-persistence gene which confers the ability to metabolize lactose throughout one’s life. IBS is harder to assess genetically, but for what it’s worth, he scored quite low on a polygenic risk score for the condition, in just the 9th percentile of risk. We should probably be looking elsewhere to explain the GI distress.
The genetic information bore much more fruit in regard to his liver disease. Remember that Beethoven’s autopsy showed cirrhosis. His polygenic risk score for liver cirrhosis puts him in the 96th percentile of risk. He was also heterozygous for two variants that can cause hereditary hemochromatosis. The risk for cirrhosis among those with these variants is increased by the use of alcohol. And historical accounts are quite clear that Beethoven consumed more than his share.
But it wasn’t just Beethoven’s DNA in these hair follicles. Analysis of a follicle from later in his life revealed the unmistakable presence of hepatitis B virus. Endemic in Europe at the time, this was a common cause of liver failure and is likely to have contributed to, if not directly caused, Beethoven’s demise.
It’s hard to read these results and not marvel at the fact that, two centuries after his death, our fascination with Beethoven has led us to probe every corner of his life – his letters, his writings, his medical records, and now his very DNA. What are we actually looking for? Is it relevant to us today what caused his hearing loss? His stomach troubles? Even his death? Will it help any patients in the future? I propose that what we are actually trying to understand is something ineffable: Genius of magnitude that is rarely seen in one or many lifetimes. And our scientific tools, as sharp as they may have become, are still far too blunt to probe the depths of that transcendence.
In any case, friends, no more of these sounds. Let us sing more cheerful songs, more full of joy.
For Medscape, I’m Perry Wilson.
Dr. Wilson is associate professor, department of medicine, and director, Clinical and Translational Research Accelerator, at Yale University, New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Joint effort: CBD not just innocent bystander in weed
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
I visited a legal cannabis dispensary in Massachusetts a few years ago, mostly to see what the hype was about. There I was, knowing basically nothing about pot, as the gentle stoner behind the counter explained to me the differences between the various strains. Acapulco Gold is buoyant and energizing; Purple Kush is sleepy, relaxed, dissociative. Here’s a strain that makes you feel nostalgic; here’s one that helps you focus. It was as complicated and as oddly specific as a fancy wine tasting – and, I had a feeling, about as reliable.
It’s a plant, after all, and though delta-9-tetrahydrocannabinol (THC) is the chemical responsible for its euphoric effects, it is far from the only substance in there.
The second most important compound in cannabis is cannabidiol, and most people will tell you that CBD is the gentle yin to THC’s paranoiac yang. Hence your local ganja barista reminding you that, if you don›t want all those anxiety-inducing side effects of THC, grab a strain with a nice CBD balance.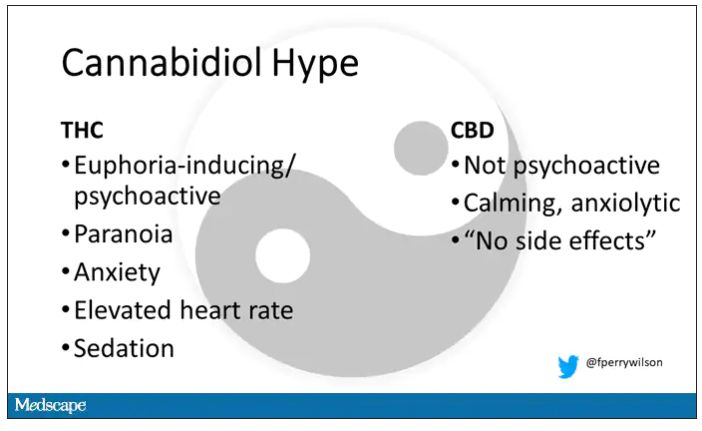
But is it true? A new study appearing in JAMA Network Open suggests, in fact, that it’s quite the opposite. This study is from Austin Zamarripa and colleagues, who clearly sit at the researcher cool kids table.
Eighteen adults who had abstained from marijuana use for at least a month participated in this trial (which is way more fun than anything we do in my lab at Yale). In random order, separated by at least a week, they ate some special brownies.
Condition one was a control brownie, condition two was a brownie containing 20 mg of THC, and condition three was a brownie containing 20 mg of THC and 640 mg of CBD. Participants were assigned each condition in random order, separated by at least a week.
A side note on doses for those of you who, like me, are not totally weed literate. A dose of 20 mg of THC is about a third of what you might find in a typical joint these days (though it’s about double the THC content of a joint in the ‘70s – I believe the technical term is “doobie”). And 640 mg of CBD is a decent dose, as 5 mg per kilogram is what some folks start with to achieve therapeutic effects.
Both THC and CBD interact with the cytochrome p450 system in the liver. This matters when you’re ingesting them instead of smoking them because you have first-pass metabolism to contend with. And, because of that p450 inhibition, it’s possible that CBD might actually increase the amount of THC that gets into your bloodstream from the brownie, or gummy, or pizza sauce, or whatever.
Let’s get to the results, starting with blood THC concentration. It’s not subtle. With CBD on board the THC concentration rises higher faster, with roughly double the area under the curve.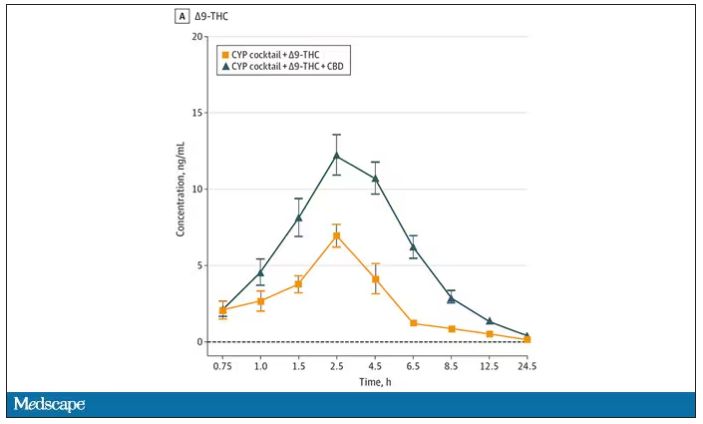
And, unsurprisingly, the subjective experience correlated with those higher levels. Individuals rated the “drug effect” higher with the combo. But, interestingly, the “pleasant” drug effect didn’t change much, while the unpleasant effects were substantially higher. No mitigation of THC anxiety here – quite the opposite. CBD made the anxiety worse.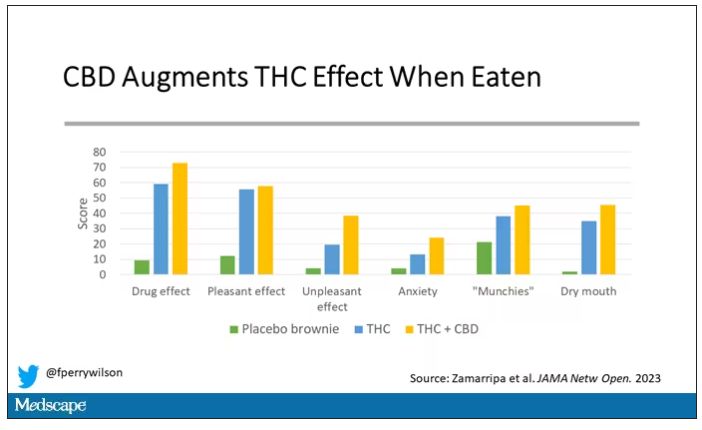
Cognitive effects were equally profound. Scores on a digit symbol substitution test and a paced serial addition task were all substantially worse when CBD was mixed with THC.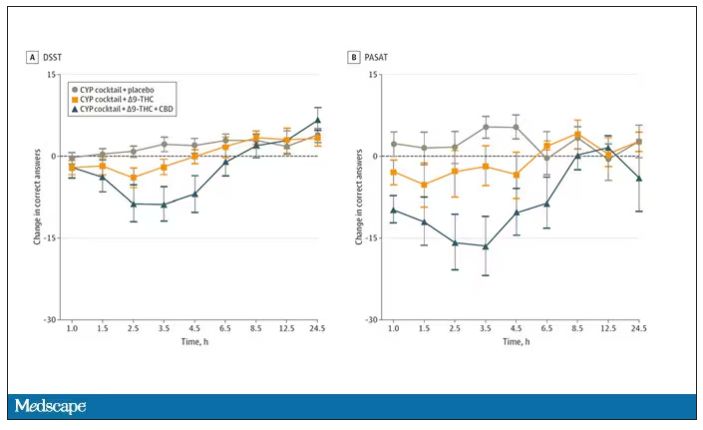
And for those of you who want some more objective measures, check out the heart rate. Despite the purported “calming” nature of CBD, heart rates were way higher when individuals were exposed to both chemicals.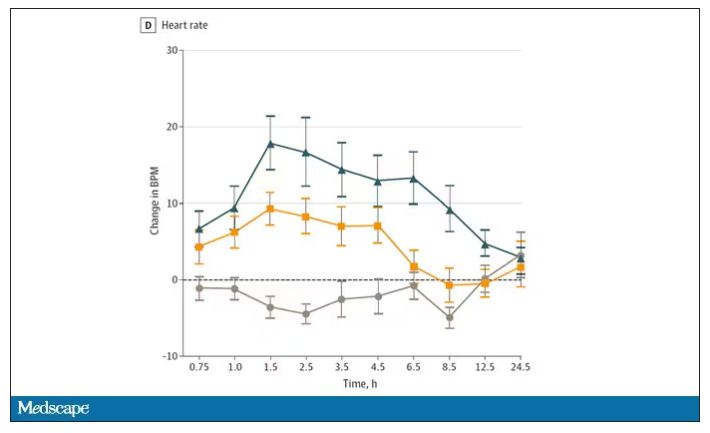
The picture here is quite clear, though the mechanism is not. At least when talking edibles, CBD enhances the effects of THC, and not necessarily for the better. It may be that CBD is competing with some of the proteins that metabolize THC, thus prolonging its effects. CBD may also directly inhibit those enzymes. But whatever the case, I think we can safely say the myth that CBD makes the effects of THC more mild or more tolerable is busted.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale University’s Clinical and Translational Research Accelerator in New Haven, Conn.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
I visited a legal cannabis dispensary in Massachusetts a few years ago, mostly to see what the hype was about. There I was, knowing basically nothing about pot, as the gentle stoner behind the counter explained to me the differences between the various strains. Acapulco Gold is buoyant and energizing; Purple Kush is sleepy, relaxed, dissociative. Here’s a strain that makes you feel nostalgic; here’s one that helps you focus. It was as complicated and as oddly specific as a fancy wine tasting – and, I had a feeling, about as reliable.
It’s a plant, after all, and though delta-9-tetrahydrocannabinol (THC) is the chemical responsible for its euphoric effects, it is far from the only substance in there.
The second most important compound in cannabis is cannabidiol, and most people will tell you that CBD is the gentle yin to THC’s paranoiac yang. Hence your local ganja barista reminding you that, if you don›t want all those anxiety-inducing side effects of THC, grab a strain with a nice CBD balance.
But is it true? A new study appearing in JAMA Network Open suggests, in fact, that it’s quite the opposite. This study is from Austin Zamarripa and colleagues, who clearly sit at the researcher cool kids table.
Eighteen adults who had abstained from marijuana use for at least a month participated in this trial (which is way more fun than anything we do in my lab at Yale). In random order, separated by at least a week, they ate some special brownies.
Condition one was a control brownie, condition two was a brownie containing 20 mg of THC, and condition three was a brownie containing 20 mg of THC and 640 mg of CBD. Participants were assigned each condition in random order, separated by at least a week.
A side note on doses for those of you who, like me, are not totally weed literate. A dose of 20 mg of THC is about a third of what you might find in a typical joint these days (though it’s about double the THC content of a joint in the ‘70s – I believe the technical term is “doobie”). And 640 mg of CBD is a decent dose, as 5 mg per kilogram is what some folks start with to achieve therapeutic effects.
Both THC and CBD interact with the cytochrome p450 system in the liver. This matters when you’re ingesting them instead of smoking them because you have first-pass metabolism to contend with. And, because of that p450 inhibition, it’s possible that CBD might actually increase the amount of THC that gets into your bloodstream from the brownie, or gummy, or pizza sauce, or whatever.
Let’s get to the results, starting with blood THC concentration. It’s not subtle. With CBD on board the THC concentration rises higher faster, with roughly double the area under the curve.
And, unsurprisingly, the subjective experience correlated with those higher levels. Individuals rated the “drug effect” higher with the combo. But, interestingly, the “pleasant” drug effect didn’t change much, while the unpleasant effects were substantially higher. No mitigation of THC anxiety here – quite the opposite. CBD made the anxiety worse.
Cognitive effects were equally profound. Scores on a digit symbol substitution test and a paced serial addition task were all substantially worse when CBD was mixed with THC.
And for those of you who want some more objective measures, check out the heart rate. Despite the purported “calming” nature of CBD, heart rates were way higher when individuals were exposed to both chemicals.
The picture here is quite clear, though the mechanism is not. At least when talking edibles, CBD enhances the effects of THC, and not necessarily for the better. It may be that CBD is competing with some of the proteins that metabolize THC, thus prolonging its effects. CBD may also directly inhibit those enzymes. But whatever the case, I think we can safely say the myth that CBD makes the effects of THC more mild or more tolerable is busted.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale University’s Clinical and Translational Research Accelerator in New Haven, Conn.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
I visited a legal cannabis dispensary in Massachusetts a few years ago, mostly to see what the hype was about. There I was, knowing basically nothing about pot, as the gentle stoner behind the counter explained to me the differences between the various strains. Acapulco Gold is buoyant and energizing; Purple Kush is sleepy, relaxed, dissociative. Here’s a strain that makes you feel nostalgic; here’s one that helps you focus. It was as complicated and as oddly specific as a fancy wine tasting – and, I had a feeling, about as reliable.
It’s a plant, after all, and though delta-9-tetrahydrocannabinol (THC) is the chemical responsible for its euphoric effects, it is far from the only substance in there.
The second most important compound in cannabis is cannabidiol, and most people will tell you that CBD is the gentle yin to THC’s paranoiac yang. Hence your local ganja barista reminding you that, if you don›t want all those anxiety-inducing side effects of THC, grab a strain with a nice CBD balance.
But is it true? A new study appearing in JAMA Network Open suggests, in fact, that it’s quite the opposite. This study is from Austin Zamarripa and colleagues, who clearly sit at the researcher cool kids table.
Eighteen adults who had abstained from marijuana use for at least a month participated in this trial (which is way more fun than anything we do in my lab at Yale). In random order, separated by at least a week, they ate some special brownies.
Condition one was a control brownie, condition two was a brownie containing 20 mg of THC, and condition three was a brownie containing 20 mg of THC and 640 mg of CBD. Participants were assigned each condition in random order, separated by at least a week.
A side note on doses for those of you who, like me, are not totally weed literate. A dose of 20 mg of THC is about a third of what you might find in a typical joint these days (though it’s about double the THC content of a joint in the ‘70s – I believe the technical term is “doobie”). And 640 mg of CBD is a decent dose, as 5 mg per kilogram is what some folks start with to achieve therapeutic effects.
Both THC and CBD interact with the cytochrome p450 system in the liver. This matters when you’re ingesting them instead of smoking them because you have first-pass metabolism to contend with. And, because of that p450 inhibition, it’s possible that CBD might actually increase the amount of THC that gets into your bloodstream from the brownie, or gummy, or pizza sauce, or whatever.
Let’s get to the results, starting with blood THC concentration. It’s not subtle. With CBD on board the THC concentration rises higher faster, with roughly double the area under the curve.
And, unsurprisingly, the subjective experience correlated with those higher levels. Individuals rated the “drug effect” higher with the combo. But, interestingly, the “pleasant” drug effect didn’t change much, while the unpleasant effects were substantially higher. No mitigation of THC anxiety here – quite the opposite. CBD made the anxiety worse.
Cognitive effects were equally profound. Scores on a digit symbol substitution test and a paced serial addition task were all substantially worse when CBD was mixed with THC.
And for those of you who want some more objective measures, check out the heart rate. Despite the purported “calming” nature of CBD, heart rates were way higher when individuals were exposed to both chemicals.
The picture here is quite clear, though the mechanism is not. At least when talking edibles, CBD enhances the effects of THC, and not necessarily for the better. It may be that CBD is competing with some of the proteins that metabolize THC, thus prolonging its effects. CBD may also directly inhibit those enzymes. But whatever the case, I think we can safely say the myth that CBD makes the effects of THC more mild or more tolerable is busted.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale University’s Clinical and Translational Research Accelerator in New Haven, Conn.
A version of this article first appeared on Medscape.com.
A new (old) drug joins the COVID fray, and guess what? It works
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
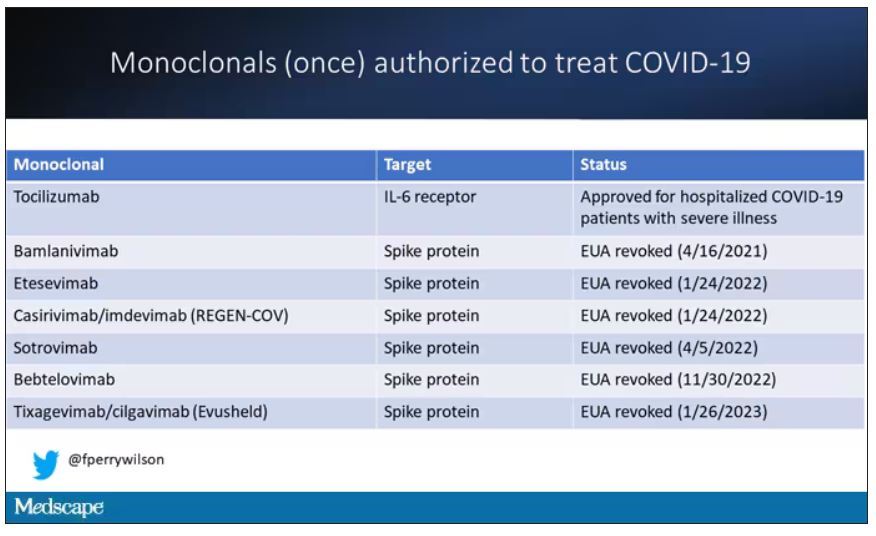
At this point, with the monoclonals found to be essentially useless, we are left with remdesivir with its modest efficacy and Paxlovid, which, for some reason, people don’t seem to be taking.
Part of the reason the monoclonals have failed lately is because of their specificity; they are homogeneous antibodies targeted toward a very specific epitope that may change from variant to variant. We need a broader therapeutic, one that has activity across all variants — maybe even one that has activity against all viruses? We’ve got one. Interferon.
The first mention of interferon as a potential COVID therapy was at the very start of the pandemic, so I’m sort of surprised that the first large, randomized trial is only being reported now in the New England Journal of Medicine.
Before we dig into the results, let’s talk mechanism. This is a trial of interferon-lambda, also known as interleukin-29.
The lambda interferons were only discovered in 2003. They differ from the more familiar interferons only in their cellular receptors; the downstream effects seem quite similar. As opposed to the cellular receptors for interferon alfa, which are widely expressed, the receptors for lambda are restricted to epithelial tissues. This makes it a good choice as a COVID treatment, since the virus also preferentially targets those epithelial cells.
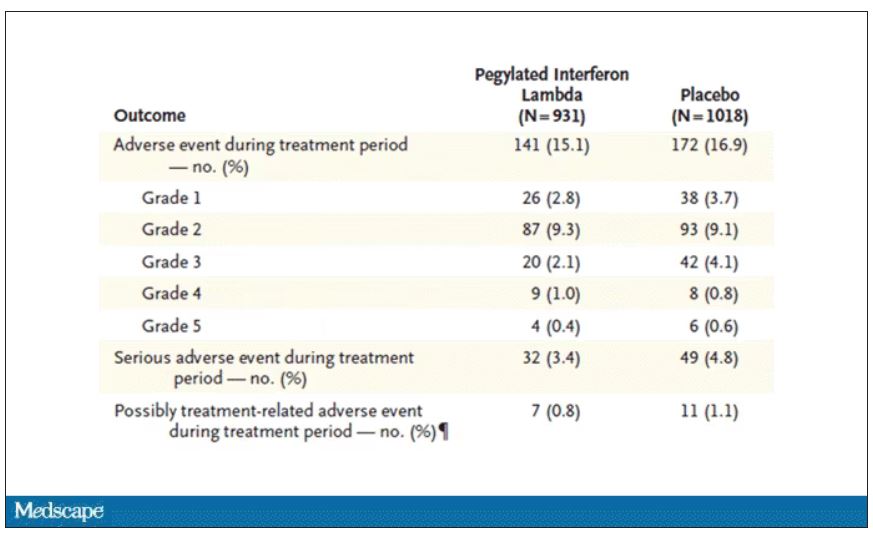
In this study, 1,951 participants from Brazil and Canada, but mostly Brazil, with new COVID infections who were not yet hospitalized were randomized to receive 180 mcg of interferon lambda or placebo.
This was a relatively current COVID trial, as you can see from the participant characteristics. The majority had been vaccinated, and nearly half of the infections were during the Omicron phase of the pandemic.
If you just want to cut to the chase, interferon worked.
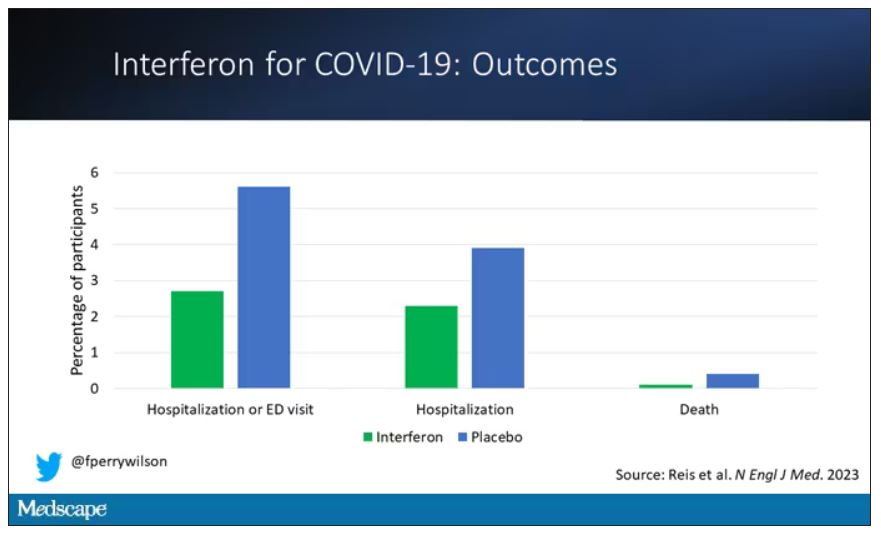
The primary outcome – hospitalization or a prolonged emergency room visit for COVID – was 50% lower in the interferon group.
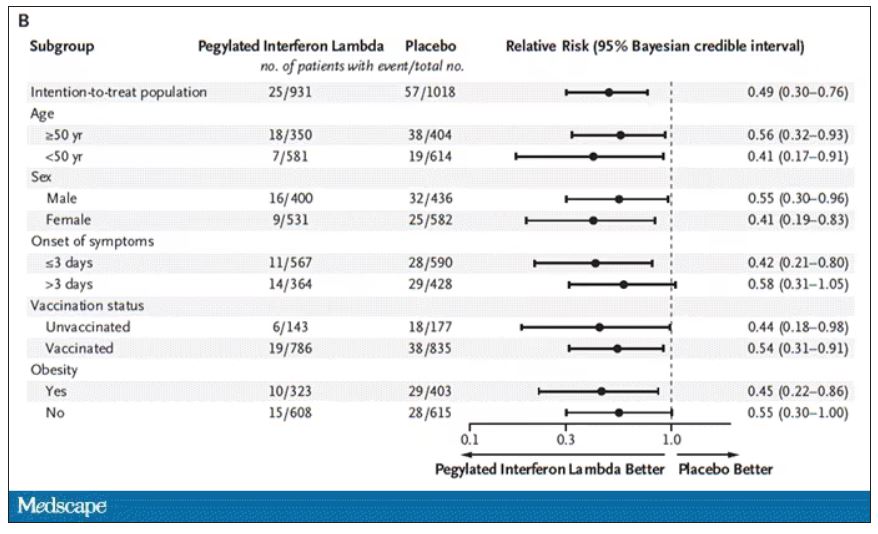
Key secondary outcomes, including death from COVID, were lower in the interferon group as well. These effects persisted across most of the subgroups I was looking out for.
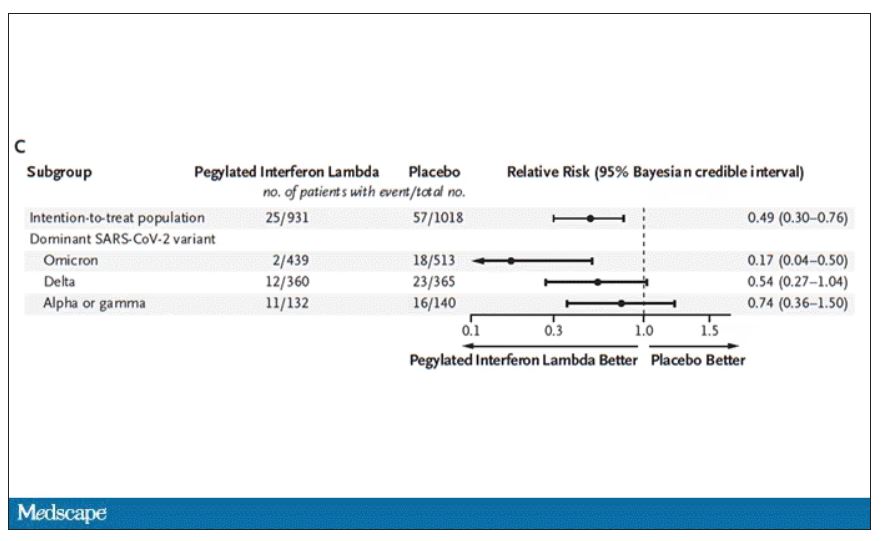
Interferon seemed to help those who were already vaccinated and those who were unvaccinated. There’s a hint that it works better within the first few days of symptoms, which isn’t surprising; we’ve seen this for many of the therapeutics, including Paxlovid. Time is of the essence. Encouragingly, the effect was a bit more pronounced among those infected with Omicron.
Of course, if you have any experience with interferon, you know that the side effects can be pretty rough. In the bad old days when we treated hepatitis C infection with interferon, patients would get their injections on Friday in anticipation of being essentially out of commission with flu-like symptoms through the weekend. But we don’t see much evidence of adverse events in this trial, maybe due to the greater specificity of interferon lambda.
Putting it all together, the state of play for interferons in COVID may be changing. To date, the FDA has not recommended the use of interferon alfa or -beta for COVID-19, citing some data that they are ineffective or even harmful in hospitalized patients with COVID. Interferon lambda is not FDA approved and thus not even available in the United States. But the reason it has not been approved is that there has not been a large, well-conducted interferon lambda trial. Now there is. Will this study be enough to prompt an emergency use authorization? The elephant in the room, of course, is Paxlovid, which at this point has a longer safety track record and, importantly, is oral. I’d love to see a head-to-head trial. Short of that, I tend to be in favor of having more options on the table.
Dr. Perry Wilson is associate professor, department of medicine, and director, Clinical and Translational Research Accelerator, at Yale University, New Haven, Conn. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
At this point, with the monoclonals found to be essentially useless, we are left with remdesivir with its modest efficacy and Paxlovid, which, for some reason, people don’t seem to be taking.
Part of the reason the monoclonals have failed lately is because of their specificity; they are homogeneous antibodies targeted toward a very specific epitope that may change from variant to variant. We need a broader therapeutic, one that has activity across all variants — maybe even one that has activity against all viruses? We’ve got one. Interferon.
The first mention of interferon as a potential COVID therapy was at the very start of the pandemic, so I’m sort of surprised that the first large, randomized trial is only being reported now in the New England Journal of Medicine.
Before we dig into the results, let’s talk mechanism. This is a trial of interferon-lambda, also known as interleukin-29.
The lambda interferons were only discovered in 2003. They differ from the more familiar interferons only in their cellular receptors; the downstream effects seem quite similar. As opposed to the cellular receptors for interferon alfa, which are widely expressed, the receptors for lambda are restricted to epithelial tissues. This makes it a good choice as a COVID treatment, since the virus also preferentially targets those epithelial cells.
In this study, 1,951 participants from Brazil and Canada, but mostly Brazil, with new COVID infections who were not yet hospitalized were randomized to receive 180 mcg of interferon lambda or placebo.
This was a relatively current COVID trial, as you can see from the participant characteristics. The majority had been vaccinated, and nearly half of the infections were during the Omicron phase of the pandemic.
If you just want to cut to the chase, interferon worked.
The primary outcome – hospitalization or a prolonged emergency room visit for COVID – was 50% lower in the interferon group.
Key secondary outcomes, including death from COVID, were lower in the interferon group as well. These effects persisted across most of the subgroups I was looking out for.
Interferon seemed to help those who were already vaccinated and those who were unvaccinated. There’s a hint that it works better within the first few days of symptoms, which isn’t surprising; we’ve seen this for many of the therapeutics, including Paxlovid. Time is of the essence. Encouragingly, the effect was a bit more pronounced among those infected with Omicron.
Of course, if you have any experience with interferon, you know that the side effects can be pretty rough. In the bad old days when we treated hepatitis C infection with interferon, patients would get their injections on Friday in anticipation of being essentially out of commission with flu-like symptoms through the weekend. But we don’t see much evidence of adverse events in this trial, maybe due to the greater specificity of interferon lambda.
Putting it all together, the state of play for interferons in COVID may be changing. To date, the FDA has not recommended the use of interferon alfa or -beta for COVID-19, citing some data that they are ineffective or even harmful in hospitalized patients with COVID. Interferon lambda is not FDA approved and thus not even available in the United States. But the reason it has not been approved is that there has not been a large, well-conducted interferon lambda trial. Now there is. Will this study be enough to prompt an emergency use authorization? The elephant in the room, of course, is Paxlovid, which at this point has a longer safety track record and, importantly, is oral. I’d love to see a head-to-head trial. Short of that, I tend to be in favor of having more options on the table.
Dr. Perry Wilson is associate professor, department of medicine, and director, Clinical and Translational Research Accelerator, at Yale University, New Haven, Conn. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
At this point, with the monoclonals found to be essentially useless, we are left with remdesivir with its modest efficacy and Paxlovid, which, for some reason, people don’t seem to be taking.
Part of the reason the monoclonals have failed lately is because of their specificity; they are homogeneous antibodies targeted toward a very specific epitope that may change from variant to variant. We need a broader therapeutic, one that has activity across all variants — maybe even one that has activity against all viruses? We’ve got one. Interferon.
The first mention of interferon as a potential COVID therapy was at the very start of the pandemic, so I’m sort of surprised that the first large, randomized trial is only being reported now in the New England Journal of Medicine.
Before we dig into the results, let’s talk mechanism. This is a trial of interferon-lambda, also known as interleukin-29.
The lambda interferons were only discovered in 2003. They differ from the more familiar interferons only in their cellular receptors; the downstream effects seem quite similar. As opposed to the cellular receptors for interferon alfa, which are widely expressed, the receptors for lambda are restricted to epithelial tissues. This makes it a good choice as a COVID treatment, since the virus also preferentially targets those epithelial cells.
In this study, 1,951 participants from Brazil and Canada, but mostly Brazil, with new COVID infections who were not yet hospitalized were randomized to receive 180 mcg of interferon lambda or placebo.
This was a relatively current COVID trial, as you can see from the participant characteristics. The majority had been vaccinated, and nearly half of the infections were during the Omicron phase of the pandemic.
If you just want to cut to the chase, interferon worked.
The primary outcome – hospitalization or a prolonged emergency room visit for COVID – was 50% lower in the interferon group.
Key secondary outcomes, including death from COVID, were lower in the interferon group as well. These effects persisted across most of the subgroups I was looking out for.
Interferon seemed to help those who were already vaccinated and those who were unvaccinated. There’s a hint that it works better within the first few days of symptoms, which isn’t surprising; we’ve seen this for many of the therapeutics, including Paxlovid. Time is of the essence. Encouragingly, the effect was a bit more pronounced among those infected with Omicron.
Of course, if you have any experience with interferon, you know that the side effects can be pretty rough. In the bad old days when we treated hepatitis C infection with interferon, patients would get their injections on Friday in anticipation of being essentially out of commission with flu-like symptoms through the weekend. But we don’t see much evidence of adverse events in this trial, maybe due to the greater specificity of interferon lambda.
Putting it all together, the state of play for interferons in COVID may be changing. To date, the FDA has not recommended the use of interferon alfa or -beta for COVID-19, citing some data that they are ineffective or even harmful in hospitalized patients with COVID. Interferon lambda is not FDA approved and thus not even available in the United States. But the reason it has not been approved is that there has not been a large, well-conducted interferon lambda trial. Now there is. Will this study be enough to prompt an emergency use authorization? The elephant in the room, of course, is Paxlovid, which at this point has a longer safety track record and, importantly, is oral. I’d love to see a head-to-head trial. Short of that, I tend to be in favor of having more options on the table.
Dr. Perry Wilson is associate professor, department of medicine, and director, Clinical and Translational Research Accelerator, at Yale University, New Haven, Conn. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Spikes out: A COVID mystery
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
To date, it has been a mystery, like “Glass Onion.” And in the spirit of all the great mysteries, to get to the bottom of this, we’ll need to round up the usual suspects.
Appearing in Circulation, a new study does a great job of systematically evaluating multiple hypotheses linking vaccination to myocarditis, and eliminating them, Poirot-style, one by one until only one remains. We’ll get there.
But first, let’s review the suspects. Why do the mRNA vaccines cause myocarditis in a small subset of people?
There are a few leading candidates.
Number one: antibody responses. There are two flavors here. The quantitative hypothesis suggests that some people simply generate too many antibodies to the vaccine, leading to increased inflammation and heart damage.
The qualitative hypothesis suggests that maybe it’s the nature of the antibodies generated rather than the amount; they might cross-react with some protein on the surface of heart cells for instance.
Or maybe it is driven by T-cell responses, which, of course, are independent of antibody levels.
There’s the idea that myocarditis is due to excessive cytokine release – sort of like what we see in the multisystem inflammatory syndrome in children.
Or it could be due to the viral antigens themselves – the spike protein the mRNA codes for that is generated after vaccination.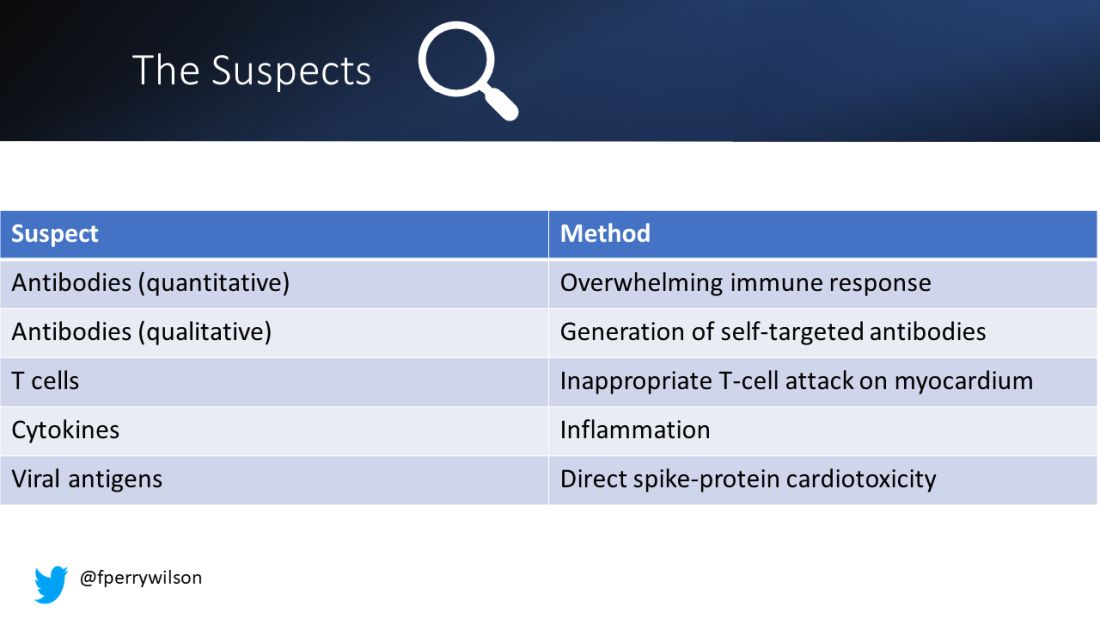
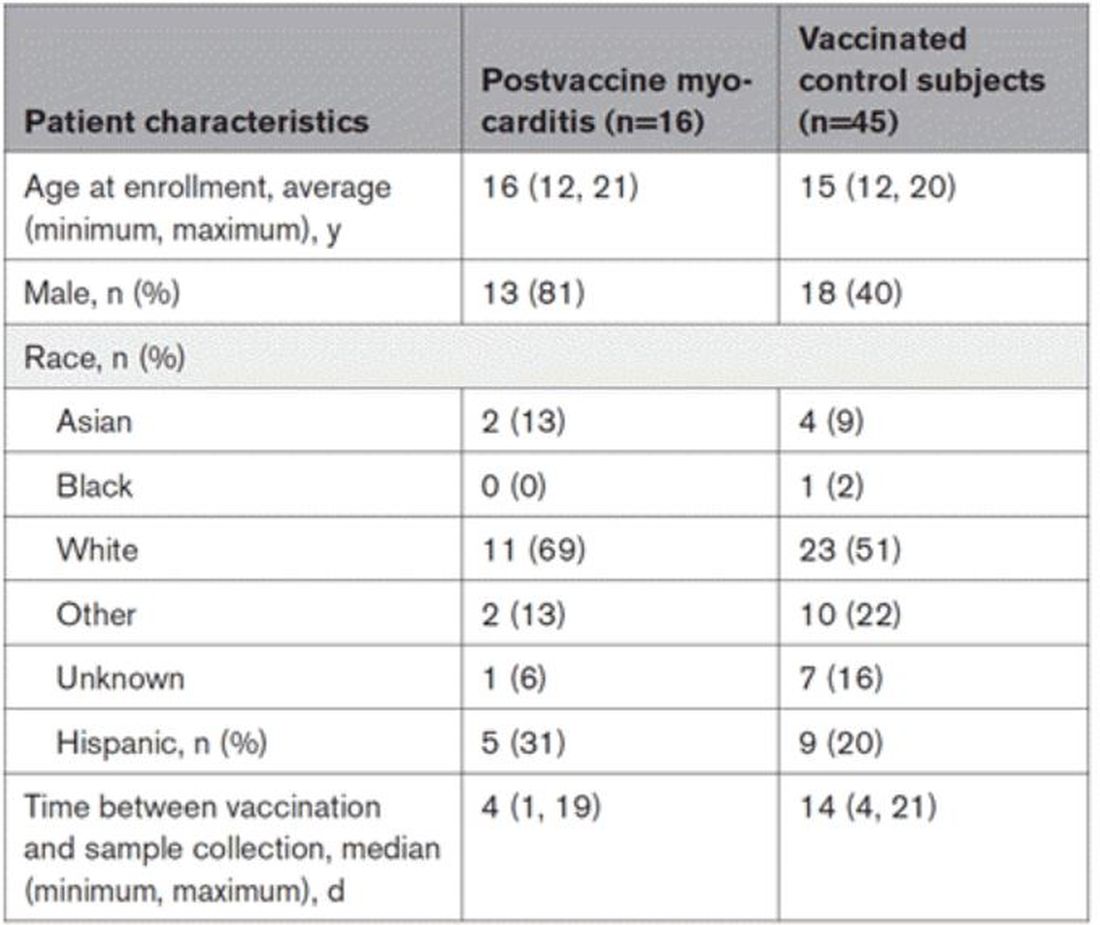
To tease all these possibilities apart, researchers led by Lael Yonker at Mass General performed a case-control study. Sixteen children with postvaccine myocarditis were matched by age to 45 control children who had been vaccinated without complications.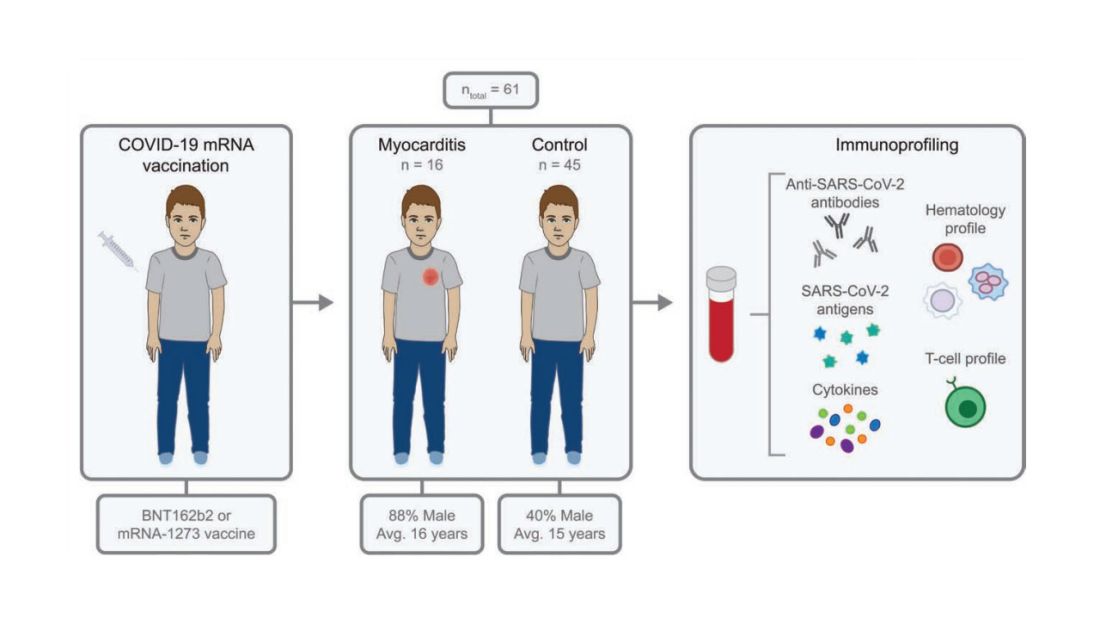
The matching was OK, but as you can see here, there were more boys in the myocarditis group, and the time from vaccination was a bit shorter in that group as well. We’ll keep that in mind as we go through the results.
OK, let’s start eliminating suspects.
First, quantitative antibodies. Seems unlikely. Absolute antibody titers were really no different in the myocarditis vs. the control group.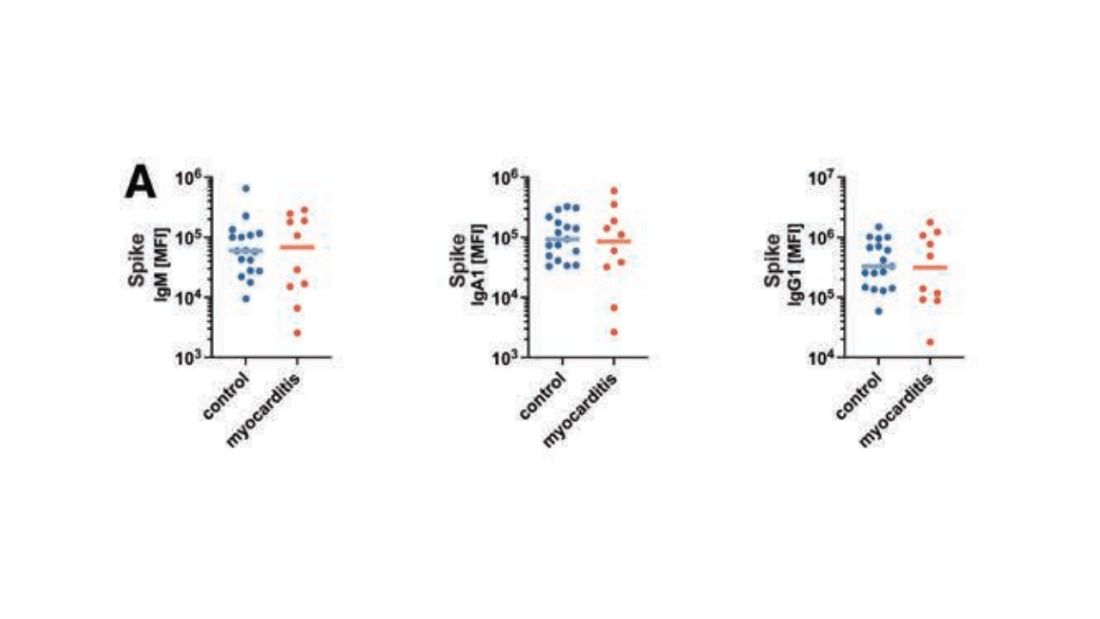
What about the quality of the antibodies? Would the kids with myocarditis have more self-recognizing antibodies present? It doesn’t appear so. Autoantibody levels were similar in the two groups.
Take antibodies off the list.
T-cell responses come next, and, again, no major differences here, save for one specific T-cell subtype that was moderately elevated in the myocarditis group. Not what I would call a smoking gun, frankly.
Cytokines give us a bit more to chew on. Levels of interleukin (IL)-8, IL-6, tumor necrosis factor (TNF)-alpha, and IL-10 were all substantially higher in the kids with myocarditis.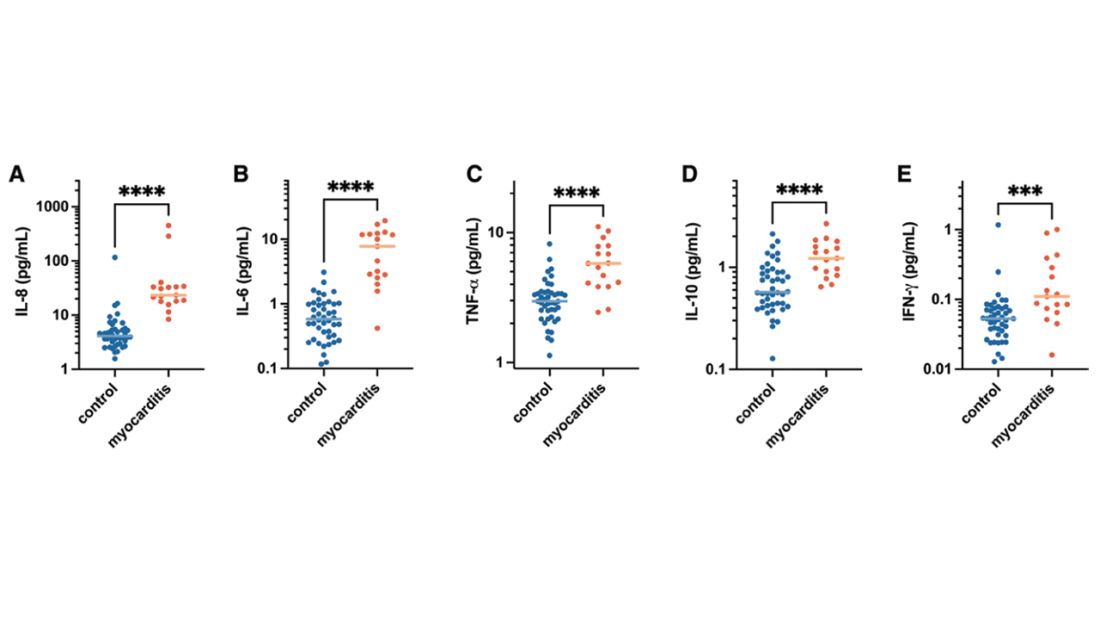
But the thing about cytokines is that they are not particularly specific. OK, kids with myocarditis have more systemic inflammation than kids without; that’s not really surprising. It still leaves us with the question of what is causing all this inflammation? Who is the arch-villain? The kingpin? The don?
It’s the analyses of antigens – the protein products of vaccination – that may hold the key here.
In 12 out of 16 kids with myocarditis, the researchers were able to measure free spike protein in the blood – that is to say spike protein, not bound by antispike antibodies.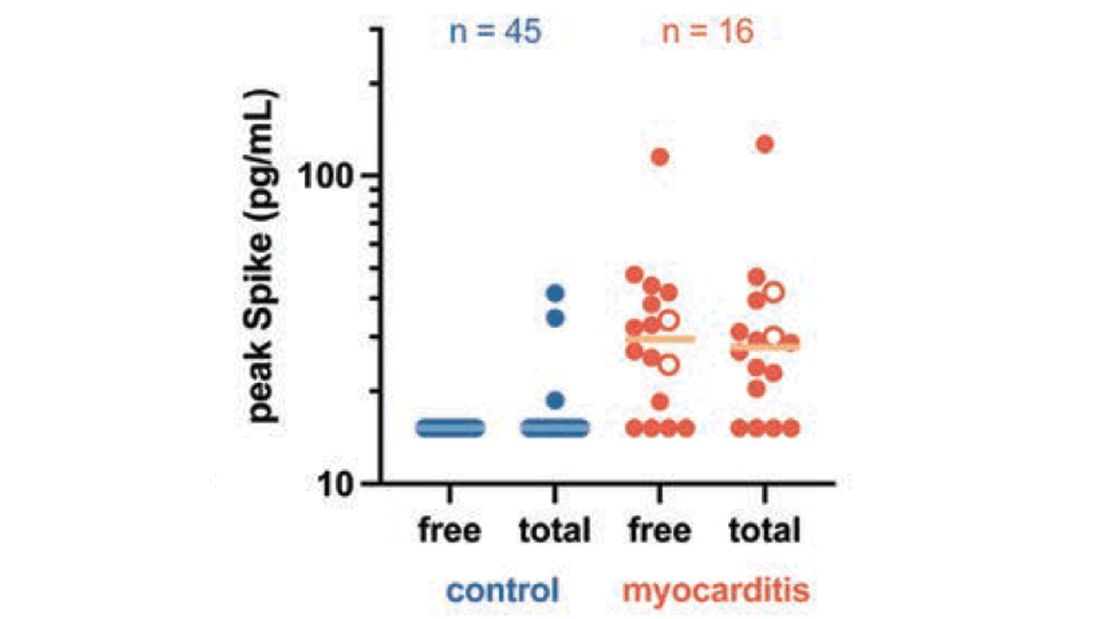
These free spikes were present in – wait for it – zero of the 45 control patients. That makes spike protein itself our prime suspect. J’accuse free spike protein!
Of course, all good detectives need to wrap up the case with a good story: How was it all done?
And here’s where we could use Agatha Christie’s help. How could this all work? The vaccine gets injected; mRNA is taken up into cells, where spike protein is generated and released, generating antibody and T-cell responses all the while. Those responses rapidly clear that spike protein from the system – this has been demonstrated in multiple studies – in adults, at least. But in some small number of people, apparently, spike protein is not cleared. Why? It makes no damn sense. Compels me, though. Some have suggested that inadvertent intravenous injection of vaccine, compared with the appropriate intramuscular route, might distribute the vaccine to sites with less immune surveillance. But that is definitely not proven yet.
We are on the path for sure, but this is, as Benoit Blanc would say, a twisted web – and we are not finished untangling it. Not yet.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here. He tweets @fperrywilson and his new book, “How Medicine Works and When It Doesn’t,” is available for preorder now. He reports no conflicts of interest.
A version of this article first appeared on Medscape.com.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
To date, it has been a mystery, like “Glass Onion.” And in the spirit of all the great mysteries, to get to the bottom of this, we’ll need to round up the usual suspects.
Appearing in Circulation, a new study does a great job of systematically evaluating multiple hypotheses linking vaccination to myocarditis, and eliminating them, Poirot-style, one by one until only one remains. We’ll get there.
But first, let’s review the suspects. Why do the mRNA vaccines cause myocarditis in a small subset of people?
There are a few leading candidates.
Number one: antibody responses. There are two flavors here. The quantitative hypothesis suggests that some people simply generate too many antibodies to the vaccine, leading to increased inflammation and heart damage.
The qualitative hypothesis suggests that maybe it’s the nature of the antibodies generated rather than the amount; they might cross-react with some protein on the surface of heart cells for instance.
Or maybe it is driven by T-cell responses, which, of course, are independent of antibody levels.
There’s the idea that myocarditis is due to excessive cytokine release – sort of like what we see in the multisystem inflammatory syndrome in children.
Or it could be due to the viral antigens themselves – the spike protein the mRNA codes for that is generated after vaccination.
To tease all these possibilities apart, researchers led by Lael Yonker at Mass General performed a case-control study. Sixteen children with postvaccine myocarditis were matched by age to 45 control children who had been vaccinated without complications.
The matching was OK, but as you can see here, there were more boys in the myocarditis group, and the time from vaccination was a bit shorter in that group as well. We’ll keep that in mind as we go through the results.
OK, let’s start eliminating suspects.
First, quantitative antibodies. Seems unlikely. Absolute antibody titers were really no different in the myocarditis vs. the control group.
What about the quality of the antibodies? Would the kids with myocarditis have more self-recognizing antibodies present? It doesn’t appear so. Autoantibody levels were similar in the two groups.
Take antibodies off the list.
T-cell responses come next, and, again, no major differences here, save for one specific T-cell subtype that was moderately elevated in the myocarditis group. Not what I would call a smoking gun, frankly.
Cytokines give us a bit more to chew on. Levels of interleukin (IL)-8, IL-6, tumor necrosis factor (TNF)-alpha, and IL-10 were all substantially higher in the kids with myocarditis.
But the thing about cytokines is that they are not particularly specific. OK, kids with myocarditis have more systemic inflammation than kids without; that’s not really surprising. It still leaves us with the question of what is causing all this inflammation? Who is the arch-villain? The kingpin? The don?
It’s the analyses of antigens – the protein products of vaccination – that may hold the key here.
In 12 out of 16 kids with myocarditis, the researchers were able to measure free spike protein in the blood – that is to say spike protein, not bound by antispike antibodies.
These free spikes were present in – wait for it – zero of the 45 control patients. That makes spike protein itself our prime suspect. J’accuse free spike protein!
Of course, all good detectives need to wrap up the case with a good story: How was it all done?
And here’s where we could use Agatha Christie’s help. How could this all work? The vaccine gets injected; mRNA is taken up into cells, where spike protein is generated and released, generating antibody and T-cell responses all the while. Those responses rapidly clear that spike protein from the system – this has been demonstrated in multiple studies – in adults, at least. But in some small number of people, apparently, spike protein is not cleared. Why? It makes no damn sense. Compels me, though. Some have suggested that inadvertent intravenous injection of vaccine, compared with the appropriate intramuscular route, might distribute the vaccine to sites with less immune surveillance. But that is definitely not proven yet.
We are on the path for sure, but this is, as Benoit Blanc would say, a twisted web – and we are not finished untangling it. Not yet.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here. He tweets @fperrywilson and his new book, “How Medicine Works and When It Doesn’t,” is available for preorder now. He reports no conflicts of interest.
A version of this article first appeared on Medscape.com.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
To date, it has been a mystery, like “Glass Onion.” And in the spirit of all the great mysteries, to get to the bottom of this, we’ll need to round up the usual suspects.
Appearing in Circulation, a new study does a great job of systematically evaluating multiple hypotheses linking vaccination to myocarditis, and eliminating them, Poirot-style, one by one until only one remains. We’ll get there.
But first, let’s review the suspects. Why do the mRNA vaccines cause myocarditis in a small subset of people?
There are a few leading candidates.
Number one: antibody responses. There are two flavors here. The quantitative hypothesis suggests that some people simply generate too many antibodies to the vaccine, leading to increased inflammation and heart damage.
The qualitative hypothesis suggests that maybe it’s the nature of the antibodies generated rather than the amount; they might cross-react with some protein on the surface of heart cells for instance.
Or maybe it is driven by T-cell responses, which, of course, are independent of antibody levels.
There’s the idea that myocarditis is due to excessive cytokine release – sort of like what we see in the multisystem inflammatory syndrome in children.
Or it could be due to the viral antigens themselves – the spike protein the mRNA codes for that is generated after vaccination.
To tease all these possibilities apart, researchers led by Lael Yonker at Mass General performed a case-control study. Sixteen children with postvaccine myocarditis were matched by age to 45 control children who had been vaccinated without complications.
The matching was OK, but as you can see here, there were more boys in the myocarditis group, and the time from vaccination was a bit shorter in that group as well. We’ll keep that in mind as we go through the results.
OK, let’s start eliminating suspects.
First, quantitative antibodies. Seems unlikely. Absolute antibody titers were really no different in the myocarditis vs. the control group.
What about the quality of the antibodies? Would the kids with myocarditis have more self-recognizing antibodies present? It doesn’t appear so. Autoantibody levels were similar in the two groups.
Take antibodies off the list.
T-cell responses come next, and, again, no major differences here, save for one specific T-cell subtype that was moderately elevated in the myocarditis group. Not what I would call a smoking gun, frankly.
Cytokines give us a bit more to chew on. Levels of interleukin (IL)-8, IL-6, tumor necrosis factor (TNF)-alpha, and IL-10 were all substantially higher in the kids with myocarditis.
But the thing about cytokines is that they are not particularly specific. OK, kids with myocarditis have more systemic inflammation than kids without; that’s not really surprising. It still leaves us with the question of what is causing all this inflammation? Who is the arch-villain? The kingpin? The don?
It’s the analyses of antigens – the protein products of vaccination – that may hold the key here.
In 12 out of 16 kids with myocarditis, the researchers were able to measure free spike protein in the blood – that is to say spike protein, not bound by antispike antibodies.
These free spikes were present in – wait for it – zero of the 45 control patients. That makes spike protein itself our prime suspect. J’accuse free spike protein!
Of course, all good detectives need to wrap up the case with a good story: How was it all done?
And here’s where we could use Agatha Christie’s help. How could this all work? The vaccine gets injected; mRNA is taken up into cells, where spike protein is generated and released, generating antibody and T-cell responses all the while. Those responses rapidly clear that spike protein from the system – this has been demonstrated in multiple studies – in adults, at least. But in some small number of people, apparently, spike protein is not cleared. Why? It makes no damn sense. Compels me, though. Some have suggested that inadvertent intravenous injection of vaccine, compared with the appropriate intramuscular route, might distribute the vaccine to sites with less immune surveillance. But that is definitely not proven yet.
We are on the path for sure, but this is, as Benoit Blanc would say, a twisted web – and we are not finished untangling it. Not yet.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. His science communication work can be found in the Huffington Post, on NPR, and here. He tweets @fperrywilson and his new book, “How Medicine Works and When It Doesn’t,” is available for preorder now. He reports no conflicts of interest.
A version of this article first appeared on Medscape.com.
Debating the clinical trial upending colonoscopy practices
This transcript has been edited for clarity.
F. Perry Wilson, MD, MSCE: Hello, and thank you for joining us today for what promises to be a lively discussion about screening for colon cancer.
My name is Perry Wilson. I’m an associate professor of medicine and director of the Clinical and Translational Research Accelerator at the Yale School of Medicine. My new book, “How Medicine Works and When It Doesn’t: Learning Who to Trust to Get and Stay Healthy,” is available for pre-order now anywhere that books are sold.
I’m joined by two wonderful experts. Dr. David Johnson is a professor of medicine and the chief of gastroenterology at the Eastern Virginia School of Medicine. He is the past president of the American College of Gastroenterology. And I’m very encouraged to see that he’s won a Distinguished Educator Award for his efforts in gastroenterology.
I’m also joined by Dr Kenny Lin. He’s a frequent contributor to Medscape and WebMD. He’s a family physician and public health consultant from Lancaster, Pa., and deputy editor of the American Family Physician journal. He’s also a teacher of residents and students at Lancaster General Health and the Penn Medicine Family Medicine Residency program.
So, we have two great educators with us today to hopefully help teach us something about colon cancer and colon cancer screening. Thank you for joining me today.
David A. Johnson, MD: Thanks for having us.
Kenneth W. Lin, MD, MPH: Good to be here.
Dr. Wilson: Colon cancer is the second leading cause of cancer mortality in the United States. A little over 50,000 people die every year in the United States due to colon cancer.
A month ago, I would have said that there was a pretty broad consensus, at least from my perspective, that people should be getting colonoscopies. That’s certainly what we tell our patients.
Then a paper came out in the New England Journal of Medicine, a very prestigious journal, that has caused a lot of consternation online and led to my receiving a lot questions from patients and their family members. Today,
Dr Johnson, can you give us a brief overview of what this trial was about?
Dr. Johnson: This was a randomized trial looking at screening colonoscopy versus no screening test whatsoever. They looked at the outcomes of prevention of cancer and the prevention of colon cancer–related death.
The short answer was that it was disappointing as it relates to colonoscopy. The study looked at patients from four European countries, with data from three of them (Norway, Poland, and Sweden) ultimately analyzed in this report in NEJM. It got a lot of attention because it surprised a lot of people by saying maybe colonoscopy wasn’t quite as good as we thought it was.
They tried to correct that by only looking at the numbers of patients who got their colonoscopy screening, which still showed value, but it was less than that we’ve seen before. There’s lots of reasons for that, which we’ll discuss shortly.
An invitation to a screening
Dr. Wilson: This was a bit of an interesting trial design. I think I’m correct, Dr Lin, that this was the first randomized trial of screening colonoscopy. But they didn’t really randomize people to get a colonoscopy versus not get a colonoscopy. Can you tell us why this differed from that study design, which I’d have thought would be simpler way of assessing this?
Dr. Lin: It’s definitely an important point to highlight about the study. What investigators did was randomize patients to receive an invitation to get a screening colonoscopy. When the trial was set up, they randomized people before they were asked whether they wanted to participate in the study. If you did it the other way around, by first asking them whether they wanted to be in the study and then randomizing them, you would have been assured that more of them probably would have gotten the colonoscopy.
But in this case, they were more interested in figuring out the real-life results of having a national program that invited patients to receive screening colonoscopy. Because we know that everyone that you recommend to get a colonoscopy doesn’t necessarily want to do that, forgets to do it, or something happens that prevents their actually getting it.
When it comes to measuring the effectiveness of the colonoscopy, it perhaps wasn’t the greatest type of study to do that. But I think it did provide some information about what would happen if you invited people to get colonoscopy, in terms of how many would do it and the results overall for that population.
Lower participation numbers than expected
Dr. Wilson: Dr. Johnson, the data show that 42% of people who were in that invitation arm followed through and got their colonoscopy. You’re a gastroenterologist. Does that seem low or about right? Do about half of people who should get a colonoscopy end up getting one?
Dr. Johnson: No, it’s low. In the United States, those numbers are probably in the 70% range. Certainly, the test doesn’t work for people who don’t get the test performed. So, if 42% of those randomized to receive an invitation to get the colonoscopy got one, that really means the majority of patients never got the test.
Dr. Wilson: Certainly, we wouldn’t expect impressive results if they don’t get the test. But on the other hand, I imagine that people who choose to get the test when they’re invited are sort of a different breed. Perhaps they’re more health conscious or living in other healthy ways. Is that something we should worry about when we look at these results?
Dr. Johnson: I don’t think you can stratify based on this study. Factors like ethnicities and diet weren’t really explained. The key element that will hopefully have the major take-home impact is quality. It’s not just the test. It’s how the test is done.
The key results
Dr. Wilson: Let’s start with the big picture. This was a study looking at everyone invited; not the subgroup of people who got the colonoscopy, but the real randomized study population.
Dr. Lin, the study did show that the invited group had a lower risk of colon cancer over the next 10 years. That’s a good thing, I imagine.
Dr. Lin: I think that’s a significant benefit. Initially in the first few years, they had more colon cancers diagnosed. But that’s probably because those were cancers that were already existing and couldn’t be prevented by the test.
But then over the years the curves crossed, and by the end of the average follow-up of 10 years, there was a significantly lower rate of colon cancers being detected. That’s as you would expect, because you’re finding polyps and removing them before they became colon cancer.
Dr. Wilson: Dr. Johnson, is that the natural history of colon cancer? It starts out as a polyp that maybe can be easily removed and doesn’t require more therapy. Is that why screening colonoscopy is helpful?
Dr. Johnson: The ultimate goal of screening is prevention of cancer, rather than detection of cancer. That occurs by identification and complete removal of the polyps that we find that are precancerous. The key is, first, detection, and second, resection. Adequate resection comes down to some very significant issues of quality, which are questions that I’d raised about this study, and we can talk about momentarily.
Dr. Wilson: Absolutely. Let me first go through the two other big findings in this study.
The fact that there were fewer cases of colon cancer over 10 years seems good. But colon cancer mortality was not significantly different in the two groups. Now, of course, we know that not everyone got a colonoscopy. I would have expected though, if you had less colon cancer, you’d have less death from colon cancer.
Dr. Lin, what might explain this disconnect?
Dr. Lin: I think there are a couple of possible explanations.
One explanation is that they just didn’t follow the people long enough. Colon cancer takes a long time to go from an adenoma to cancer, and from cancer to something that would cause the patient’s death. You may need to follow them for longer than the 10 years that most of these patients were followed to see that benefit. I think there probably will be benefit after a while, because if you are removing colon cancers that otherwise would have progressed and metastasized, you often see a benefit.
We also have to consider the other possibility that not all the polyps removed necessarily were going to progress to advanced cancer. Therefore, you weren’t seeing the death benefit because not every polyp that was removed was necessarily going to cause health consequences.
In colonoscopy, quality is key to success
Dr. Wilson: You’re removing things and have no way of knowing in advance which are the bad ones and which aren’t.
Dr. Johnson, you’ve mentioned several times now that the quality of colonoscopy matters here. So, I’m intuiting that it’s not one-size-fits-all, that it’s not all the same. What do you mean by quality of colonoscopy, and what was it in the NEJM study?
Dr. Johnson: Quality colonoscopy is the quality of the whole process. It starts with the warm-up, if you will, and the clean out for the procedure. That allows the colonoscopist to be able to identify precancerous polyps, which we call adenomas (there are other precancerous polyps called sessile serrated lesions).
The identification of adenomas is extremely important. Even a small increase in the detection of those precancerous polyps has benefits. Well-performed studies looking at large databases show that a small, 1% increase in the adenoma detection leads to a 3% decrease in colon cancer and a 5% decrease in colon cancer–related death. There’s a huge array of effect when we talk about small increases in the adenoma detection rate.
Now, let’s go back to this study in NEJM.
If we base quality on the physician performing the colonoscopy, and say that the colonoscopy is achieving the act of getting all the way around the colon, but not all physicians in the study were able to do that, it starts to raise the question about quality, because adenoma detection is so important. Earlier reports from this group [Nordic-European Initiative on Colorectal Cancer Study Group] have shown that the adenoma detection rates have been way below the national thresholds. So, this raises the question of whether they found the polyp, and then whether they resected the polyp. They also don’t tell us where these cancers were. It is about the colonoscopy quality. It’s not the instrument. It’s the process.
An overview of other screening tools
Dr. Wilson: Dr. Lin, colonoscopy, which requires prep and anesthesia, is not the only colon cancer screening method we have. In fact, there are a bunch. I think we’re on board saying it’s probably better to detect colon cancer early than not detect it. But what are our other options aside from colonoscopy that can allow for early detection of colon cancer?
Dr. Lin: For most of my career, there were three options that I presented patients with. The first was the fecal test, which used to be in the form of initial hemoccult tests. These have been mostly replaced by fecal immunochemical testing. But they’re both just basically looking for the presence of blood in the stool. Anyone who has a positive test would be referred for a diagnostic colonoscopy.
The other test besides colonoscopy, which has been largely phased out in the United States, although it is still very much used in Canada and much of Europe, is flexible sigmoidoscopy. Until this study, the tests supported by randomized controlled trials were the fecal tests and flexible sigmoidoscopy.
Interestingly, there was a recent systematic review of flexible sigmoidoscopy looking at four trials and their effects over 15 years. They showed not only a reduction in colon cancer, but also a reduction in colon cancer mortality, and even a small reduction in all-cause mortality.
I believe three out of the four trials were done where the patients were consented and then randomized, so they had a higher uptake of the procedure.
But when you compare this with the colonoscopy trial, it really isn’t that impressive. You would expect a much larger benefit, because obviously you’re looking at the entire colon. But you really didn’t see that. It was, at best, maybe equivalent to sigmoidoscopy, but not a whole lot better.
Dr. Johnson: Perry, you mentioned sedation. It’s important to understand that this particular cohort of patients are from Norway, Sweden, and Poland, where it’s very much the norm to not get sedation for your colonoscopy. Any of the [audience] who have had colonoscopy will tell you that they are not ones to say, “Don’t give me sedation.” The rate of sedation is around 11% in Norway, maybe 23% in Sweden, and around 45% in Poland. So, the examiner and the patient were never really super comfortable.
I’ve done 50,000 colonoscopies in my career, and many nonsedated. We know that taking time increases the finding of polyps and the adequate identification and resection. So, that ability to perform at a high quality is very much impacted when the patients aren’t comfortable.
Dr. Wilson: Dr. Johnson, we brought up flexible sigmoidoscopy. For the patients watching whose doctors are talking to them about screening colonoscopy, what’s the difference?
Dr. Johnson: Flexible sigmoidoscopy is just a short scope examination, in which you see about one-third of the colon. I’ve been in the field for 45 years, and during that time we’ve seen that there’s a progressive increase in the development of cancers above that bottom third of the colon to the higher end, the two-thirds of the colon that you would miss without doing a full colonoscopy. Also, flexible sigmoidoscopy typically does not get covered for sedation.
Again, if you do the exam and find something, then you’re going to have to come back and do an adequate resection with a colonoscopy. So, one-stop-shopping colon cancer screening is not about detection of cancer, it’s about prevention of cancer, and that’s what colonoscopy does.
Patients want convenience, but at what cost?
Dr. Wilson: Dr. Lin, how are your patients in your family practice handling this study? Have conversations changed around colon cancer screening? What are people asking about these days?
Dr. Lin: I don’t think the conversations have changed in my practice that much. When patients ask about this study, we do discuss the limitations, that it wasn’t designed to assess the maximum benefit of getting a colonoscopy because the majority of people assigned to that group didn’t get colonoscopy.
But I think it is an opportunity in primary care to consider the way we present the options to patients. Because I would guess that a majority of primary care physicians, when they present the options, would say colonoscopy is the gold standard and recommend their patients get it. And they only offer fecal testing to patients who don’t want the colonoscopy or really refuse.
That hasn’t been my practice. I’m usually more agnostic, because there are both harms and benefits. If you get a fecal test, the chance of you having a complication from colonoscopy is automatically lower because most of those people will not get colonoscopy. Now obviously, the complications with colonoscopy are pretty rare and usually self-limited, but they do exist. If you’re doing lots and lots of these, eventually you’ll see them. Probably all primary care physicians have patients who’ve had a complication from colonoscopy and may or may not have regretted it depending on how information was presented.
But I feel like this study reinforces my feeling that we ought to be presenting these, and not saying one is superior or inferior to the other. Instead, I’d base it on what the patient’s priorities are. But I feel like this study reinforces my feeling that we ought to be presenting these, and not saying one is superior or inferior to the other. Instead, I’d base it on what the patient’s priorities are. Is your priority finding every single cancer? Do you want to know exactly what the benefit is? I think with colonoscopy, we’re still trying to figure out exactly what the benefit is. Whereas we can say it pretty confidently for fecal tests because we have those randomized trials.
Dr. Wilson: Dr. Johnson, I think patients who are watching need to know, first of all, that if they do the fecal test route, a positive fecal test does lead to colonoscopy. In some sense, all roads lead to colonoscopy once you have a positive screening test. So, I can certainly see the value of just sort of skipping to that point. But what about this risk-versus-benefit relationship? Colonoscopy, albeit a relatively safe procedure, is still a procedure. There is some risk associated with it. If we can get the same benefit from yearly fecal immunochemical testing, is that a better choice potentially, at least for patients at average risk?
Dr. Johnson: The stool-based testing is really more effective for detection of cancer. That’s not screening, where the entire goal is the prevention of cancer. The fecal-based testing, including the stool-based DNA testing, misses the majority of precancerous polyps. And the fecal immunochemical tests, which Dr. Lin just mentioned, misses virtually all of them. We really want to get to the prevention of cancer, meaning identification and removal of polyps, not just screening for cancer.
Dr. Wilson: Do you see anything on the horizon that could unseat colonoscopy as, to quote Dr. Lin, the potential gold standard for screening for colon cancer?
Dr. Johnson: I think not on the horizon for identification and removal of polyps. That’s really the gold standard. Technology continues to advance. We’ll see what happens. But on the short and intermediate horizon, colonoscopy is going to be needed.
We are finding that some patients are starting to acquiesce to stool-based testing because they can do it at home. Maybe they don’t have to do a prep. We’re talking about screening only here, not about the follow-up of patients who have a family history, patients who have colitis, patients who have had colon polyps, or other reasons. Stool-based testing is not an option for the follow-up of those patients.
Convenience testing, in the face of COVID, also has thrown a wrench into things. Patients may have wanted to stay home and do these tests. Again, we need to be proactive, not reactive. We want to prevent cancer, not detect it.
Changing advice in the face of younger screening thresholds
Dr. Wilson: Dr. Lin, I’m 42 years old. I don’t believe I’m at any increased risk of colon cancer based on my family history or other risk factors. I’m 3 years away from when the U.S. Preventive Services Task Force tells me I should potentially consider starting to screen for colon cancer. That recommendation has recently been moved down from 50 years old to 45 years old. So, it’s on my mind as I approach that age. What do you advise younger patients approaching 45 right now in terms of screening for colon cancer?
Dr. Lin: For patients with the risk factors that Dr. Johnson mentioned, I would recommend screening colonoscopy as the initial test.
Assuming you don’t have those risk factors, I present it as we have a couple of different fecal tests. There’s the traditional one that just looks for blood. Then there’s the newer one that also adds DNA, which is more sensitive for colorectal cancer, but a little less specific, which is a problem just because there are more false positives.
But you need to compare that with colonoscopy, which you only need to get done ideally every 10 years if there are no findings. That is more complete. And theoretically, as we’ve been talking about, it would also prevent as well as detect early cancers.
So, I think it’s really down to your preference in terms of how the various factors that come into play, such as convenience of the test and your level of concern about cancer. I do tell patients that family history of cancer is not terribly predictive of whether you get it or not. A lot of people unfortunately who develop colorectal cancer have no previous family history. Diet will come into play to some extent. There are some things that point to increased risk for colorectal cancer if you have a diet high in red meat and things like that. But ultimately, it really is up to the patient. I lay out the options, and whatever they choose, I’m happy to pursue.
But the most important thing is that they do some test, because doing no test is not going to help anyone. I do agree with the notion that the best test is the test that gets done.
Dr. Wilson: Absolutely. I think the NEJM study supports that, even when we’re talking about colonoscopy.
Dr. Johnson, you’ve had some criticisms about the NEJM study, and I think they make sense. At the same time, as this is the first randomized trial of colonoscopy, it’s kind of the only data we have. Are we going to get better data? Are there other studies going on out there that might help shed some light on what’s turning out to be a complicated issue?
Dr. Johnson: Yes, there are ongoing studies. They’re not taking place within the United States, because you couldn’t get through a no-screening option trial. There are comparative studies that are probably still 5 years away looking at stool-based testing.
But again, we have to recognize that if you do these alternative tests that were eloquently discussed by Dr. Lin, and not the colonoscopy, which would be every 10 years with high-quality performance, that you have to annualize or do them in sequence. It’s important that you follow up on those with regularity. It’s not just a one-time test every 10 years for these individual tests.
And any of the time that those tests are ordered, the patient should be instructed that if it’s positive you need a colonoscopy. We’re seeing a lot of slippage on that front for the stool-based testing. Convenience is not the answer. It’s getting the job done.
Dr. Wilson: Would you agree, Dr. Johnson, that for patients that really don’t want to do the colonoscopy for one reason or another, and you’ve done your best in explaining what you think the risks and benefits are, that you’d rather have them get something than nothing?
Dr. Johnson: Absolutely. It comes down to what I recommend and then what you decide. But I still make the point explicit: If we’ve gone through those checkpoints and it’s positive, we agree that you understand that colonoscopy is the next step.
Final take-home messages
Dr. Wilson: Dr. Lin, I’ll turn the last word over to you, as the person who is probably discussing the choice of screening modalities more than any of us, before someone would get referred to someone like Dr. Johnson. What’s your final take-home message about the NEJM study and the state of colon cancer screening in the United States?
Dr. Lin: My take-home points about the study are that there were some limitations, but it is good to finally have a randomized trial of colonoscopy screening 2 decades after we really started doing that in the United States. It won’t immediately change – nor do I think it should – the way we practice and discuss different options. I think that some of Dr. Johnson’s points about making sure that whoever’s doing the colonoscopies for your practices is doing it in a high-quality way are really important. Just as it’s important, if you’re doing the fecal tests, to make sure that all patients who have positives get expeditiously referred for colonoscopy.
Dr. Johnson: Perry, I’d like to make one concluding comment as the gastroenterology expert in this discussion. I’ve had countless questions about this study from my patients and my peers. I tell them the following: Don’t let the headlines mislead you.
When you look at this study, the instrument is not so much the question. We know that getting the test is the first step in colon cancer screening. But we also know that getting the test done, with the highest-quality providers and the best-quality performance, is really the key to optimizing the true value of colonoscopy for colon cancer prevention.
So please don’t lose sight of this when reading the headlines in the media around this study. We really need to analyze the true characteristics of what we call a quality performance, because that’s what drives success and that’s what prevents colon cancer.
Dr. Wilson: Dr. Johnson and Dr. Lin, thank you very much. I appreciate you spending time with me here today and wish you all the best.
I guess I’ll sum up by saying that if you’re getting a colonoscopy, make sure it’s a good one. But do get screened.
This video originally appeared on WebMD. A transcript appeared on Medscape.com.
This transcript has been edited for clarity.
F. Perry Wilson, MD, MSCE: Hello, and thank you for joining us today for what promises to be a lively discussion about screening for colon cancer.
My name is Perry Wilson. I’m an associate professor of medicine and director of the Clinical and Translational Research Accelerator at the Yale School of Medicine. My new book, “How Medicine Works and When It Doesn’t: Learning Who to Trust to Get and Stay Healthy,” is available for pre-order now anywhere that books are sold.
I’m joined by two wonderful experts. Dr. David Johnson is a professor of medicine and the chief of gastroenterology at the Eastern Virginia School of Medicine. He is the past president of the American College of Gastroenterology. And I’m very encouraged to see that he’s won a Distinguished Educator Award for his efforts in gastroenterology.
I’m also joined by Dr Kenny Lin. He’s a frequent contributor to Medscape and WebMD. He’s a family physician and public health consultant from Lancaster, Pa., and deputy editor of the American Family Physician journal. He’s also a teacher of residents and students at Lancaster General Health and the Penn Medicine Family Medicine Residency program.
So, we have two great educators with us today to hopefully help teach us something about colon cancer and colon cancer screening. Thank you for joining me today.
David A. Johnson, MD: Thanks for having us.
Kenneth W. Lin, MD, MPH: Good to be here.
Dr. Wilson: Colon cancer is the second leading cause of cancer mortality in the United States. A little over 50,000 people die every year in the United States due to colon cancer.
A month ago, I would have said that there was a pretty broad consensus, at least from my perspective, that people should be getting colonoscopies. That’s certainly what we tell our patients.
Then a paper came out in the New England Journal of Medicine, a very prestigious journal, that has caused a lot of consternation online and led to my receiving a lot questions from patients and their family members. Today,
Dr Johnson, can you give us a brief overview of what this trial was about?
Dr. Johnson: This was a randomized trial looking at screening colonoscopy versus no screening test whatsoever. They looked at the outcomes of prevention of cancer and the prevention of colon cancer–related death.
The short answer was that it was disappointing as it relates to colonoscopy. The study looked at patients from four European countries, with data from three of them (Norway, Poland, and Sweden) ultimately analyzed in this report in NEJM. It got a lot of attention because it surprised a lot of people by saying maybe colonoscopy wasn’t quite as good as we thought it was.
They tried to correct that by only looking at the numbers of patients who got their colonoscopy screening, which still showed value, but it was less than that we’ve seen before. There’s lots of reasons for that, which we’ll discuss shortly.
An invitation to a screening
Dr. Wilson: This was a bit of an interesting trial design. I think I’m correct, Dr Lin, that this was the first randomized trial of screening colonoscopy. But they didn’t really randomize people to get a colonoscopy versus not get a colonoscopy. Can you tell us why this differed from that study design, which I’d have thought would be simpler way of assessing this?
Dr. Lin: It’s definitely an important point to highlight about the study. What investigators did was randomize patients to receive an invitation to get a screening colonoscopy. When the trial was set up, they randomized people before they were asked whether they wanted to participate in the study. If you did it the other way around, by first asking them whether they wanted to be in the study and then randomizing them, you would have been assured that more of them probably would have gotten the colonoscopy.
But in this case, they were more interested in figuring out the real-life results of having a national program that invited patients to receive screening colonoscopy. Because we know that everyone that you recommend to get a colonoscopy doesn’t necessarily want to do that, forgets to do it, or something happens that prevents their actually getting it.
When it comes to measuring the effectiveness of the colonoscopy, it perhaps wasn’t the greatest type of study to do that. But I think it did provide some information about what would happen if you invited people to get colonoscopy, in terms of how many would do it and the results overall for that population.
Lower participation numbers than expected
Dr. Wilson: Dr. Johnson, the data show that 42% of people who were in that invitation arm followed through and got their colonoscopy. You’re a gastroenterologist. Does that seem low or about right? Do about half of people who should get a colonoscopy end up getting one?
Dr. Johnson: No, it’s low. In the United States, those numbers are probably in the 70% range. Certainly, the test doesn’t work for people who don’t get the test performed. So, if 42% of those randomized to receive an invitation to get the colonoscopy got one, that really means the majority of patients never got the test.
Dr. Wilson: Certainly, we wouldn’t expect impressive results if they don’t get the test. But on the other hand, I imagine that people who choose to get the test when they’re invited are sort of a different breed. Perhaps they’re more health conscious or living in other healthy ways. Is that something we should worry about when we look at these results?
Dr. Johnson: I don’t think you can stratify based on this study. Factors like ethnicities and diet weren’t really explained. The key element that will hopefully have the major take-home impact is quality. It’s not just the test. It’s how the test is done.
The key results
Dr. Wilson: Let’s start with the big picture. This was a study looking at everyone invited; not the subgroup of people who got the colonoscopy, but the real randomized study population.
Dr. Lin, the study did show that the invited group had a lower risk of colon cancer over the next 10 years. That’s a good thing, I imagine.
Dr. Lin: I think that’s a significant benefit. Initially in the first few years, they had more colon cancers diagnosed. But that’s probably because those were cancers that were already existing and couldn’t be prevented by the test.
But then over the years the curves crossed, and by the end of the average follow-up of 10 years, there was a significantly lower rate of colon cancers being detected. That’s as you would expect, because you’re finding polyps and removing them before they became colon cancer.
Dr. Wilson: Dr. Johnson, is that the natural history of colon cancer? It starts out as a polyp that maybe can be easily removed and doesn’t require more therapy. Is that why screening colonoscopy is helpful?
Dr. Johnson: The ultimate goal of screening is prevention of cancer, rather than detection of cancer. That occurs by identification and complete removal of the polyps that we find that are precancerous. The key is, first, detection, and second, resection. Adequate resection comes down to some very significant issues of quality, which are questions that I’d raised about this study, and we can talk about momentarily.
Dr. Wilson: Absolutely. Let me first go through the two other big findings in this study.
The fact that there were fewer cases of colon cancer over 10 years seems good. But colon cancer mortality was not significantly different in the two groups. Now, of course, we know that not everyone got a colonoscopy. I would have expected though, if you had less colon cancer, you’d have less death from colon cancer.
Dr. Lin, what might explain this disconnect?
Dr. Lin: I think there are a couple of possible explanations.
One explanation is that they just didn’t follow the people long enough. Colon cancer takes a long time to go from an adenoma to cancer, and from cancer to something that would cause the patient’s death. You may need to follow them for longer than the 10 years that most of these patients were followed to see that benefit. I think there probably will be benefit after a while, because if you are removing colon cancers that otherwise would have progressed and metastasized, you often see a benefit.
We also have to consider the other possibility that not all the polyps removed necessarily were going to progress to advanced cancer. Therefore, you weren’t seeing the death benefit because not every polyp that was removed was necessarily going to cause health consequences.
In colonoscopy, quality is key to success
Dr. Wilson: You’re removing things and have no way of knowing in advance which are the bad ones and which aren’t.
Dr. Johnson, you’ve mentioned several times now that the quality of colonoscopy matters here. So, I’m intuiting that it’s not one-size-fits-all, that it’s not all the same. What do you mean by quality of colonoscopy, and what was it in the NEJM study?
Dr. Johnson: Quality colonoscopy is the quality of the whole process. It starts with the warm-up, if you will, and the clean out for the procedure. That allows the colonoscopist to be able to identify precancerous polyps, which we call adenomas (there are other precancerous polyps called sessile serrated lesions).
The identification of adenomas is extremely important. Even a small increase in the detection of those precancerous polyps has benefits. Well-performed studies looking at large databases show that a small, 1% increase in the adenoma detection leads to a 3% decrease in colon cancer and a 5% decrease in colon cancer–related death. There’s a huge array of effect when we talk about small increases in the adenoma detection rate.
Now, let’s go back to this study in NEJM.
If we base quality on the physician performing the colonoscopy, and say that the colonoscopy is achieving the act of getting all the way around the colon, but not all physicians in the study were able to do that, it starts to raise the question about quality, because adenoma detection is so important. Earlier reports from this group [Nordic-European Initiative on Colorectal Cancer Study Group] have shown that the adenoma detection rates have been way below the national thresholds. So, this raises the question of whether they found the polyp, and then whether they resected the polyp. They also don’t tell us where these cancers were. It is about the colonoscopy quality. It’s not the instrument. It’s the process.
An overview of other screening tools
Dr. Wilson: Dr. Lin, colonoscopy, which requires prep and anesthesia, is not the only colon cancer screening method we have. In fact, there are a bunch. I think we’re on board saying it’s probably better to detect colon cancer early than not detect it. But what are our other options aside from colonoscopy that can allow for early detection of colon cancer?
Dr. Lin: For most of my career, there were three options that I presented patients with. The first was the fecal test, which used to be in the form of initial hemoccult tests. These have been mostly replaced by fecal immunochemical testing. But they’re both just basically looking for the presence of blood in the stool. Anyone who has a positive test would be referred for a diagnostic colonoscopy.
The other test besides colonoscopy, which has been largely phased out in the United States, although it is still very much used in Canada and much of Europe, is flexible sigmoidoscopy. Until this study, the tests supported by randomized controlled trials were the fecal tests and flexible sigmoidoscopy.
Interestingly, there was a recent systematic review of flexible sigmoidoscopy looking at four trials and their effects over 15 years. They showed not only a reduction in colon cancer, but also a reduction in colon cancer mortality, and even a small reduction in all-cause mortality.
I believe three out of the four trials were done where the patients were consented and then randomized, so they had a higher uptake of the procedure.
But when you compare this with the colonoscopy trial, it really isn’t that impressive. You would expect a much larger benefit, because obviously you’re looking at the entire colon. But you really didn’t see that. It was, at best, maybe equivalent to sigmoidoscopy, but not a whole lot better.
Dr. Johnson: Perry, you mentioned sedation. It’s important to understand that this particular cohort of patients are from Norway, Sweden, and Poland, where it’s very much the norm to not get sedation for your colonoscopy. Any of the [audience] who have had colonoscopy will tell you that they are not ones to say, “Don’t give me sedation.” The rate of sedation is around 11% in Norway, maybe 23% in Sweden, and around 45% in Poland. So, the examiner and the patient were never really super comfortable.
I’ve done 50,000 colonoscopies in my career, and many nonsedated. We know that taking time increases the finding of polyps and the adequate identification and resection. So, that ability to perform at a high quality is very much impacted when the patients aren’t comfortable.
Dr. Wilson: Dr. Johnson, we brought up flexible sigmoidoscopy. For the patients watching whose doctors are talking to them about screening colonoscopy, what’s the difference?
Dr. Johnson: Flexible sigmoidoscopy is just a short scope examination, in which you see about one-third of the colon. I’ve been in the field for 45 years, and during that time we’ve seen that there’s a progressive increase in the development of cancers above that bottom third of the colon to the higher end, the two-thirds of the colon that you would miss without doing a full colonoscopy. Also, flexible sigmoidoscopy typically does not get covered for sedation.
Again, if you do the exam and find something, then you’re going to have to come back and do an adequate resection with a colonoscopy. So, one-stop-shopping colon cancer screening is not about detection of cancer, it’s about prevention of cancer, and that’s what colonoscopy does.
Patients want convenience, but at what cost?
Dr. Wilson: Dr. Lin, how are your patients in your family practice handling this study? Have conversations changed around colon cancer screening? What are people asking about these days?
Dr. Lin: I don’t think the conversations have changed in my practice that much. When patients ask about this study, we do discuss the limitations, that it wasn’t designed to assess the maximum benefit of getting a colonoscopy because the majority of people assigned to that group didn’t get colonoscopy.
But I think it is an opportunity in primary care to consider the way we present the options to patients. Because I would guess that a majority of primary care physicians, when they present the options, would say colonoscopy is the gold standard and recommend their patients get it. And they only offer fecal testing to patients who don’t want the colonoscopy or really refuse.
That hasn’t been my practice. I’m usually more agnostic, because there are both harms and benefits. If you get a fecal test, the chance of you having a complication from colonoscopy is automatically lower because most of those people will not get colonoscopy. Now obviously, the complications with colonoscopy are pretty rare and usually self-limited, but they do exist. If you’re doing lots and lots of these, eventually you’ll see them. Probably all primary care physicians have patients who’ve had a complication from colonoscopy and may or may not have regretted it depending on how information was presented.
But I feel like this study reinforces my feeling that we ought to be presenting these, and not saying one is superior or inferior to the other. Instead, I’d base it on what the patient’s priorities are. But I feel like this study reinforces my feeling that we ought to be presenting these, and not saying one is superior or inferior to the other. Instead, I’d base it on what the patient’s priorities are. Is your priority finding every single cancer? Do you want to know exactly what the benefit is? I think with colonoscopy, we’re still trying to figure out exactly what the benefit is. Whereas we can say it pretty confidently for fecal tests because we have those randomized trials.
Dr. Wilson: Dr. Johnson, I think patients who are watching need to know, first of all, that if they do the fecal test route, a positive fecal test does lead to colonoscopy. In some sense, all roads lead to colonoscopy once you have a positive screening test. So, I can certainly see the value of just sort of skipping to that point. But what about this risk-versus-benefit relationship? Colonoscopy, albeit a relatively safe procedure, is still a procedure. There is some risk associated with it. If we can get the same benefit from yearly fecal immunochemical testing, is that a better choice potentially, at least for patients at average risk?
Dr. Johnson: The stool-based testing is really more effective for detection of cancer. That’s not screening, where the entire goal is the prevention of cancer. The fecal-based testing, including the stool-based DNA testing, misses the majority of precancerous polyps. And the fecal immunochemical tests, which Dr. Lin just mentioned, misses virtually all of them. We really want to get to the prevention of cancer, meaning identification and removal of polyps, not just screening for cancer.
Dr. Wilson: Do you see anything on the horizon that could unseat colonoscopy as, to quote Dr. Lin, the potential gold standard for screening for colon cancer?
Dr. Johnson: I think not on the horizon for identification and removal of polyps. That’s really the gold standard. Technology continues to advance. We’ll see what happens. But on the short and intermediate horizon, colonoscopy is going to be needed.
We are finding that some patients are starting to acquiesce to stool-based testing because they can do it at home. Maybe they don’t have to do a prep. We’re talking about screening only here, not about the follow-up of patients who have a family history, patients who have colitis, patients who have had colon polyps, or other reasons. Stool-based testing is not an option for the follow-up of those patients.
Convenience testing, in the face of COVID, also has thrown a wrench into things. Patients may have wanted to stay home and do these tests. Again, we need to be proactive, not reactive. We want to prevent cancer, not detect it.
Changing advice in the face of younger screening thresholds
Dr. Wilson: Dr. Lin, I’m 42 years old. I don’t believe I’m at any increased risk of colon cancer based on my family history or other risk factors. I’m 3 years away from when the U.S. Preventive Services Task Force tells me I should potentially consider starting to screen for colon cancer. That recommendation has recently been moved down from 50 years old to 45 years old. So, it’s on my mind as I approach that age. What do you advise younger patients approaching 45 right now in terms of screening for colon cancer?
Dr. Lin: For patients with the risk factors that Dr. Johnson mentioned, I would recommend screening colonoscopy as the initial test.
Assuming you don’t have those risk factors, I present it as we have a couple of different fecal tests. There’s the traditional one that just looks for blood. Then there’s the newer one that also adds DNA, which is more sensitive for colorectal cancer, but a little less specific, which is a problem just because there are more false positives.
But you need to compare that with colonoscopy, which you only need to get done ideally every 10 years if there are no findings. That is more complete. And theoretically, as we’ve been talking about, it would also prevent as well as detect early cancers.
So, I think it’s really down to your preference in terms of how the various factors that come into play, such as convenience of the test and your level of concern about cancer. I do tell patients that family history of cancer is not terribly predictive of whether you get it or not. A lot of people unfortunately who develop colorectal cancer have no previous family history. Diet will come into play to some extent. There are some things that point to increased risk for colorectal cancer if you have a diet high in red meat and things like that. But ultimately, it really is up to the patient. I lay out the options, and whatever they choose, I’m happy to pursue.
But the most important thing is that they do some test, because doing no test is not going to help anyone. I do agree with the notion that the best test is the test that gets done.
Dr. Wilson: Absolutely. I think the NEJM study supports that, even when we’re talking about colonoscopy.
Dr. Johnson, you’ve had some criticisms about the NEJM study, and I think they make sense. At the same time, as this is the first randomized trial of colonoscopy, it’s kind of the only data we have. Are we going to get better data? Are there other studies going on out there that might help shed some light on what’s turning out to be a complicated issue?
Dr. Johnson: Yes, there are ongoing studies. They’re not taking place within the United States, because you couldn’t get through a no-screening option trial. There are comparative studies that are probably still 5 years away looking at stool-based testing.
But again, we have to recognize that if you do these alternative tests that were eloquently discussed by Dr. Lin, and not the colonoscopy, which would be every 10 years with high-quality performance, that you have to annualize or do them in sequence. It’s important that you follow up on those with regularity. It’s not just a one-time test every 10 years for these individual tests.
And any of the time that those tests are ordered, the patient should be instructed that if it’s positive you need a colonoscopy. We’re seeing a lot of slippage on that front for the stool-based testing. Convenience is not the answer. It’s getting the job done.
Dr. Wilson: Would you agree, Dr. Johnson, that for patients that really don’t want to do the colonoscopy for one reason or another, and you’ve done your best in explaining what you think the risks and benefits are, that you’d rather have them get something than nothing?
Dr. Johnson: Absolutely. It comes down to what I recommend and then what you decide. But I still make the point explicit: If we’ve gone through those checkpoints and it’s positive, we agree that you understand that colonoscopy is the next step.
Final take-home messages
Dr. Wilson: Dr. Lin, I’ll turn the last word over to you, as the person who is probably discussing the choice of screening modalities more than any of us, before someone would get referred to someone like Dr. Johnson. What’s your final take-home message about the NEJM study and the state of colon cancer screening in the United States?
Dr. Lin: My take-home points about the study are that there were some limitations, but it is good to finally have a randomized trial of colonoscopy screening 2 decades after we really started doing that in the United States. It won’t immediately change – nor do I think it should – the way we practice and discuss different options. I think that some of Dr. Johnson’s points about making sure that whoever’s doing the colonoscopies for your practices is doing it in a high-quality way are really important. Just as it’s important, if you’re doing the fecal tests, to make sure that all patients who have positives get expeditiously referred for colonoscopy.
Dr. Johnson: Perry, I’d like to make one concluding comment as the gastroenterology expert in this discussion. I’ve had countless questions about this study from my patients and my peers. I tell them the following: Don’t let the headlines mislead you.
When you look at this study, the instrument is not so much the question. We know that getting the test is the first step in colon cancer screening. But we also know that getting the test done, with the highest-quality providers and the best-quality performance, is really the key to optimizing the true value of colonoscopy for colon cancer prevention.
So please don’t lose sight of this when reading the headlines in the media around this study. We really need to analyze the true characteristics of what we call a quality performance, because that’s what drives success and that’s what prevents colon cancer.
Dr. Wilson: Dr. Johnson and Dr. Lin, thank you very much. I appreciate you spending time with me here today and wish you all the best.
I guess I’ll sum up by saying that if you’re getting a colonoscopy, make sure it’s a good one. But do get screened.
This video originally appeared on WebMD. A transcript appeared on Medscape.com.
This transcript has been edited for clarity.
F. Perry Wilson, MD, MSCE: Hello, and thank you for joining us today for what promises to be a lively discussion about screening for colon cancer.
My name is Perry Wilson. I’m an associate professor of medicine and director of the Clinical and Translational Research Accelerator at the Yale School of Medicine. My new book, “How Medicine Works and When It Doesn’t: Learning Who to Trust to Get and Stay Healthy,” is available for pre-order now anywhere that books are sold.
I’m joined by two wonderful experts. Dr. David Johnson is a professor of medicine and the chief of gastroenterology at the Eastern Virginia School of Medicine. He is the past president of the American College of Gastroenterology. And I’m very encouraged to see that he’s won a Distinguished Educator Award for his efforts in gastroenterology.
I’m also joined by Dr Kenny Lin. He’s a frequent contributor to Medscape and WebMD. He’s a family physician and public health consultant from Lancaster, Pa., and deputy editor of the American Family Physician journal. He’s also a teacher of residents and students at Lancaster General Health and the Penn Medicine Family Medicine Residency program.
So, we have two great educators with us today to hopefully help teach us something about colon cancer and colon cancer screening. Thank you for joining me today.
David A. Johnson, MD: Thanks for having us.
Kenneth W. Lin, MD, MPH: Good to be here.
Dr. Wilson: Colon cancer is the second leading cause of cancer mortality in the United States. A little over 50,000 people die every year in the United States due to colon cancer.
A month ago, I would have said that there was a pretty broad consensus, at least from my perspective, that people should be getting colonoscopies. That’s certainly what we tell our patients.
Then a paper came out in the New England Journal of Medicine, a very prestigious journal, that has caused a lot of consternation online and led to my receiving a lot questions from patients and their family members. Today,
Dr Johnson, can you give us a brief overview of what this trial was about?
Dr. Johnson: This was a randomized trial looking at screening colonoscopy versus no screening test whatsoever. They looked at the outcomes of prevention of cancer and the prevention of colon cancer–related death.
The short answer was that it was disappointing as it relates to colonoscopy. The study looked at patients from four European countries, with data from three of them (Norway, Poland, and Sweden) ultimately analyzed in this report in NEJM. It got a lot of attention because it surprised a lot of people by saying maybe colonoscopy wasn’t quite as good as we thought it was.
They tried to correct that by only looking at the numbers of patients who got their colonoscopy screening, which still showed value, but it was less than that we’ve seen before. There’s lots of reasons for that, which we’ll discuss shortly.
An invitation to a screening
Dr. Wilson: This was a bit of an interesting trial design. I think I’m correct, Dr Lin, that this was the first randomized trial of screening colonoscopy. But they didn’t really randomize people to get a colonoscopy versus not get a colonoscopy. Can you tell us why this differed from that study design, which I’d have thought would be simpler way of assessing this?
Dr. Lin: It’s definitely an important point to highlight about the study. What investigators did was randomize patients to receive an invitation to get a screening colonoscopy. When the trial was set up, they randomized people before they were asked whether they wanted to participate in the study. If you did it the other way around, by first asking them whether they wanted to be in the study and then randomizing them, you would have been assured that more of them probably would have gotten the colonoscopy.
But in this case, they were more interested in figuring out the real-life results of having a national program that invited patients to receive screening colonoscopy. Because we know that everyone that you recommend to get a colonoscopy doesn’t necessarily want to do that, forgets to do it, or something happens that prevents their actually getting it.
When it comes to measuring the effectiveness of the colonoscopy, it perhaps wasn’t the greatest type of study to do that. But I think it did provide some information about what would happen if you invited people to get colonoscopy, in terms of how many would do it and the results overall for that population.
Lower participation numbers than expected
Dr. Wilson: Dr. Johnson, the data show that 42% of people who were in that invitation arm followed through and got their colonoscopy. You’re a gastroenterologist. Does that seem low or about right? Do about half of people who should get a colonoscopy end up getting one?
Dr. Johnson: No, it’s low. In the United States, those numbers are probably in the 70% range. Certainly, the test doesn’t work for people who don’t get the test performed. So, if 42% of those randomized to receive an invitation to get the colonoscopy got one, that really means the majority of patients never got the test.
Dr. Wilson: Certainly, we wouldn’t expect impressive results if they don’t get the test. But on the other hand, I imagine that people who choose to get the test when they’re invited are sort of a different breed. Perhaps they’re more health conscious or living in other healthy ways. Is that something we should worry about when we look at these results?
Dr. Johnson: I don’t think you can stratify based on this study. Factors like ethnicities and diet weren’t really explained. The key element that will hopefully have the major take-home impact is quality. It’s not just the test. It’s how the test is done.
The key results
Dr. Wilson: Let’s start with the big picture. This was a study looking at everyone invited; not the subgroup of people who got the colonoscopy, but the real randomized study population.
Dr. Lin, the study did show that the invited group had a lower risk of colon cancer over the next 10 years. That’s a good thing, I imagine.
Dr. Lin: I think that’s a significant benefit. Initially in the first few years, they had more colon cancers diagnosed. But that’s probably because those were cancers that were already existing and couldn’t be prevented by the test.
But then over the years the curves crossed, and by the end of the average follow-up of 10 years, there was a significantly lower rate of colon cancers being detected. That’s as you would expect, because you’re finding polyps and removing them before they became colon cancer.
Dr. Wilson: Dr. Johnson, is that the natural history of colon cancer? It starts out as a polyp that maybe can be easily removed and doesn’t require more therapy. Is that why screening colonoscopy is helpful?
Dr. Johnson: The ultimate goal of screening is prevention of cancer, rather than detection of cancer. That occurs by identification and complete removal of the polyps that we find that are precancerous. The key is, first, detection, and second, resection. Adequate resection comes down to some very significant issues of quality, which are questions that I’d raised about this study, and we can talk about momentarily.
Dr. Wilson: Absolutely. Let me first go through the two other big findings in this study.
The fact that there were fewer cases of colon cancer over 10 years seems good. But colon cancer mortality was not significantly different in the two groups. Now, of course, we know that not everyone got a colonoscopy. I would have expected though, if you had less colon cancer, you’d have less death from colon cancer.
Dr. Lin, what might explain this disconnect?
Dr. Lin: I think there are a couple of possible explanations.
One explanation is that they just didn’t follow the people long enough. Colon cancer takes a long time to go from an adenoma to cancer, and from cancer to something that would cause the patient’s death. You may need to follow them for longer than the 10 years that most of these patients were followed to see that benefit. I think there probably will be benefit after a while, because if you are removing colon cancers that otherwise would have progressed and metastasized, you often see a benefit.
We also have to consider the other possibility that not all the polyps removed necessarily were going to progress to advanced cancer. Therefore, you weren’t seeing the death benefit because not every polyp that was removed was necessarily going to cause health consequences.
In colonoscopy, quality is key to success
Dr. Wilson: You’re removing things and have no way of knowing in advance which are the bad ones and which aren’t.
Dr. Johnson, you’ve mentioned several times now that the quality of colonoscopy matters here. So, I’m intuiting that it’s not one-size-fits-all, that it’s not all the same. What do you mean by quality of colonoscopy, and what was it in the NEJM study?
Dr. Johnson: Quality colonoscopy is the quality of the whole process. It starts with the warm-up, if you will, and the clean out for the procedure. That allows the colonoscopist to be able to identify precancerous polyps, which we call adenomas (there are other precancerous polyps called sessile serrated lesions).
The identification of adenomas is extremely important. Even a small increase in the detection of those precancerous polyps has benefits. Well-performed studies looking at large databases show that a small, 1% increase in the adenoma detection leads to a 3% decrease in colon cancer and a 5% decrease in colon cancer–related death. There’s a huge array of effect when we talk about small increases in the adenoma detection rate.
Now, let’s go back to this study in NEJM.
If we base quality on the physician performing the colonoscopy, and say that the colonoscopy is achieving the act of getting all the way around the colon, but not all physicians in the study were able to do that, it starts to raise the question about quality, because adenoma detection is so important. Earlier reports from this group [Nordic-European Initiative on Colorectal Cancer Study Group] have shown that the adenoma detection rates have been way below the national thresholds. So, this raises the question of whether they found the polyp, and then whether they resected the polyp. They also don’t tell us where these cancers were. It is about the colonoscopy quality. It’s not the instrument. It’s the process.
An overview of other screening tools
Dr. Wilson: Dr. Lin, colonoscopy, which requires prep and anesthesia, is not the only colon cancer screening method we have. In fact, there are a bunch. I think we’re on board saying it’s probably better to detect colon cancer early than not detect it. But what are our other options aside from colonoscopy that can allow for early detection of colon cancer?
Dr. Lin: For most of my career, there were three options that I presented patients with. The first was the fecal test, which used to be in the form of initial hemoccult tests. These have been mostly replaced by fecal immunochemical testing. But they’re both just basically looking for the presence of blood in the stool. Anyone who has a positive test would be referred for a diagnostic colonoscopy.
The other test besides colonoscopy, which has been largely phased out in the United States, although it is still very much used in Canada and much of Europe, is flexible sigmoidoscopy. Until this study, the tests supported by randomized controlled trials were the fecal tests and flexible sigmoidoscopy.
Interestingly, there was a recent systematic review of flexible sigmoidoscopy looking at four trials and their effects over 15 years. They showed not only a reduction in colon cancer, but also a reduction in colon cancer mortality, and even a small reduction in all-cause mortality.
I believe three out of the four trials were done where the patients were consented and then randomized, so they had a higher uptake of the procedure.
But when you compare this with the colonoscopy trial, it really isn’t that impressive. You would expect a much larger benefit, because obviously you’re looking at the entire colon. But you really didn’t see that. It was, at best, maybe equivalent to sigmoidoscopy, but not a whole lot better.
Dr. Johnson: Perry, you mentioned sedation. It’s important to understand that this particular cohort of patients are from Norway, Sweden, and Poland, where it’s very much the norm to not get sedation for your colonoscopy. Any of the [audience] who have had colonoscopy will tell you that they are not ones to say, “Don’t give me sedation.” The rate of sedation is around 11% in Norway, maybe 23% in Sweden, and around 45% in Poland. So, the examiner and the patient were never really super comfortable.
I’ve done 50,000 colonoscopies in my career, and many nonsedated. We know that taking time increases the finding of polyps and the adequate identification and resection. So, that ability to perform at a high quality is very much impacted when the patients aren’t comfortable.
Dr. Wilson: Dr. Johnson, we brought up flexible sigmoidoscopy. For the patients watching whose doctors are talking to them about screening colonoscopy, what’s the difference?
Dr. Johnson: Flexible sigmoidoscopy is just a short scope examination, in which you see about one-third of the colon. I’ve been in the field for 45 years, and during that time we’ve seen that there’s a progressive increase in the development of cancers above that bottom third of the colon to the higher end, the two-thirds of the colon that you would miss without doing a full colonoscopy. Also, flexible sigmoidoscopy typically does not get covered for sedation.
Again, if you do the exam and find something, then you’re going to have to come back and do an adequate resection with a colonoscopy. So, one-stop-shopping colon cancer screening is not about detection of cancer, it’s about prevention of cancer, and that’s what colonoscopy does.
Patients want convenience, but at what cost?
Dr. Wilson: Dr. Lin, how are your patients in your family practice handling this study? Have conversations changed around colon cancer screening? What are people asking about these days?
Dr. Lin: I don’t think the conversations have changed in my practice that much. When patients ask about this study, we do discuss the limitations, that it wasn’t designed to assess the maximum benefit of getting a colonoscopy because the majority of people assigned to that group didn’t get colonoscopy.
But I think it is an opportunity in primary care to consider the way we present the options to patients. Because I would guess that a majority of primary care physicians, when they present the options, would say colonoscopy is the gold standard and recommend their patients get it. And they only offer fecal testing to patients who don’t want the colonoscopy or really refuse.
That hasn’t been my practice. I’m usually more agnostic, because there are both harms and benefits. If you get a fecal test, the chance of you having a complication from colonoscopy is automatically lower because most of those people will not get colonoscopy. Now obviously, the complications with colonoscopy are pretty rare and usually self-limited, but they do exist. If you’re doing lots and lots of these, eventually you’ll see them. Probably all primary care physicians have patients who’ve had a complication from colonoscopy and may or may not have regretted it depending on how information was presented.
But I feel like this study reinforces my feeling that we ought to be presenting these, and not saying one is superior or inferior to the other. Instead, I’d base it on what the patient’s priorities are. But I feel like this study reinforces my feeling that we ought to be presenting these, and not saying one is superior or inferior to the other. Instead, I’d base it on what the patient’s priorities are. Is your priority finding every single cancer? Do you want to know exactly what the benefit is? I think with colonoscopy, we’re still trying to figure out exactly what the benefit is. Whereas we can say it pretty confidently for fecal tests because we have those randomized trials.
Dr. Wilson: Dr. Johnson, I think patients who are watching need to know, first of all, that if they do the fecal test route, a positive fecal test does lead to colonoscopy. In some sense, all roads lead to colonoscopy once you have a positive screening test. So, I can certainly see the value of just sort of skipping to that point. But what about this risk-versus-benefit relationship? Colonoscopy, albeit a relatively safe procedure, is still a procedure. There is some risk associated with it. If we can get the same benefit from yearly fecal immunochemical testing, is that a better choice potentially, at least for patients at average risk?
Dr. Johnson: The stool-based testing is really more effective for detection of cancer. That’s not screening, where the entire goal is the prevention of cancer. The fecal-based testing, including the stool-based DNA testing, misses the majority of precancerous polyps. And the fecal immunochemical tests, which Dr. Lin just mentioned, misses virtually all of them. We really want to get to the prevention of cancer, meaning identification and removal of polyps, not just screening for cancer.
Dr. Wilson: Do you see anything on the horizon that could unseat colonoscopy as, to quote Dr. Lin, the potential gold standard for screening for colon cancer?
Dr. Johnson: I think not on the horizon for identification and removal of polyps. That’s really the gold standard. Technology continues to advance. We’ll see what happens. But on the short and intermediate horizon, colonoscopy is going to be needed.
We are finding that some patients are starting to acquiesce to stool-based testing because they can do it at home. Maybe they don’t have to do a prep. We’re talking about screening only here, not about the follow-up of patients who have a family history, patients who have colitis, patients who have had colon polyps, or other reasons. Stool-based testing is not an option for the follow-up of those patients.
Convenience testing, in the face of COVID, also has thrown a wrench into things. Patients may have wanted to stay home and do these tests. Again, we need to be proactive, not reactive. We want to prevent cancer, not detect it.
Changing advice in the face of younger screening thresholds
Dr. Wilson: Dr. Lin, I’m 42 years old. I don’t believe I’m at any increased risk of colon cancer based on my family history or other risk factors. I’m 3 years away from when the U.S. Preventive Services Task Force tells me I should potentially consider starting to screen for colon cancer. That recommendation has recently been moved down from 50 years old to 45 years old. So, it’s on my mind as I approach that age. What do you advise younger patients approaching 45 right now in terms of screening for colon cancer?
Dr. Lin: For patients with the risk factors that Dr. Johnson mentioned, I would recommend screening colonoscopy as the initial test.
Assuming you don’t have those risk factors, I present it as we have a couple of different fecal tests. There’s the traditional one that just looks for blood. Then there’s the newer one that also adds DNA, which is more sensitive for colorectal cancer, but a little less specific, which is a problem just because there are more false positives.
But you need to compare that with colonoscopy, which you only need to get done ideally every 10 years if there are no findings. That is more complete. And theoretically, as we’ve been talking about, it would also prevent as well as detect early cancers.
So, I think it’s really down to your preference in terms of how the various factors that come into play, such as convenience of the test and your level of concern about cancer. I do tell patients that family history of cancer is not terribly predictive of whether you get it or not. A lot of people unfortunately who develop colorectal cancer have no previous family history. Diet will come into play to some extent. There are some things that point to increased risk for colorectal cancer if you have a diet high in red meat and things like that. But ultimately, it really is up to the patient. I lay out the options, and whatever they choose, I’m happy to pursue.
But the most important thing is that they do some test, because doing no test is not going to help anyone. I do agree with the notion that the best test is the test that gets done.
Dr. Wilson: Absolutely. I think the NEJM study supports that, even when we’re talking about colonoscopy.
Dr. Johnson, you’ve had some criticisms about the NEJM study, and I think they make sense. At the same time, as this is the first randomized trial of colonoscopy, it’s kind of the only data we have. Are we going to get better data? Are there other studies going on out there that might help shed some light on what’s turning out to be a complicated issue?
Dr. Johnson: Yes, there are ongoing studies. They’re not taking place within the United States, because you couldn’t get through a no-screening option trial. There are comparative studies that are probably still 5 years away looking at stool-based testing.
But again, we have to recognize that if you do these alternative tests that were eloquently discussed by Dr. Lin, and not the colonoscopy, which would be every 10 years with high-quality performance, that you have to annualize or do them in sequence. It’s important that you follow up on those with regularity. It’s not just a one-time test every 10 years for these individual tests.
And any of the time that those tests are ordered, the patient should be instructed that if it’s positive you need a colonoscopy. We’re seeing a lot of slippage on that front for the stool-based testing. Convenience is not the answer. It’s getting the job done.
Dr. Wilson: Would you agree, Dr. Johnson, that for patients that really don’t want to do the colonoscopy for one reason or another, and you’ve done your best in explaining what you think the risks and benefits are, that you’d rather have them get something than nothing?
Dr. Johnson: Absolutely. It comes down to what I recommend and then what you decide. But I still make the point explicit: If we’ve gone through those checkpoints and it’s positive, we agree that you understand that colonoscopy is the next step.
Final take-home messages
Dr. Wilson: Dr. Lin, I’ll turn the last word over to you, as the person who is probably discussing the choice of screening modalities more than any of us, before someone would get referred to someone like Dr. Johnson. What’s your final take-home message about the NEJM study and the state of colon cancer screening in the United States?
Dr. Lin: My take-home points about the study are that there were some limitations, but it is good to finally have a randomized trial of colonoscopy screening 2 decades after we really started doing that in the United States. It won’t immediately change – nor do I think it should – the way we practice and discuss different options. I think that some of Dr. Johnson’s points about making sure that whoever’s doing the colonoscopies for your practices is doing it in a high-quality way are really important. Just as it’s important, if you’re doing the fecal tests, to make sure that all patients who have positives get expeditiously referred for colonoscopy.
Dr. Johnson: Perry, I’d like to make one concluding comment as the gastroenterology expert in this discussion. I’ve had countless questions about this study from my patients and my peers. I tell them the following: Don’t let the headlines mislead you.
When you look at this study, the instrument is not so much the question. We know that getting the test is the first step in colon cancer screening. But we also know that getting the test done, with the highest-quality providers and the best-quality performance, is really the key to optimizing the true value of colonoscopy for colon cancer prevention.
So please don’t lose sight of this when reading the headlines in the media around this study. We really need to analyze the true characteristics of what we call a quality performance, because that’s what drives success and that’s what prevents colon cancer.
Dr. Wilson: Dr. Johnson and Dr. Lin, thank you very much. I appreciate you spending time with me here today and wish you all the best.
I guess I’ll sum up by saying that if you’re getting a colonoscopy, make sure it’s a good one. But do get screened.
This video originally appeared on WebMD. A transcript appeared on Medscape.com.
Mindfulness, exercise strike out in memory trial
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
We are coming to the end of the year, which always makes me think about getting older. Much like the search for the fountain of youth, many promising leads have ultimately led to dead ends. And yet, I had high hopes for a trial that focused on two cornerstones of wellness – exercise and mindfulness – to address the subjective loss of memory that comes with aging. Alas, meditation and exercise do not appear to be the fountain of youth.
I’m talking about this study, appearing in JAMA, known as the MEDEX trial.
It’s a clever design: a 2 x 2 factorial randomized trial where participants could be randomized to a mindfulness intervention, an exercise intervention, both, or neither.
In this manner, you can test multiple hypotheses exploiting a shared control group. Or as a mentor of mine used to say, you get two trials for the price of one and a half.
The participants were older adults, aged 65-84, living in the community. They had to be relatively sedentary at baseline and not engaging in mindfulness practices. They had to subjectively report some memory or concentration issues but had to be cognitively intact, based on a standard dementia screening test. In other words, these are your average older people who are worried that they aren’t as sharp as they used to be.
The interventions themselves were fairly intense. The exercise group had instructor-led sessions for 90 minutes twice a week for the first 6 months of the study, once a week thereafter. And participants were encouraged to exercise at home such that they had a total of 300 minutes of weekly exercise.
The mindfulness program was characterized by eight weekly classes of 2.5 hours each as well as a half-day retreat to teach the tenets of mindfulness and meditation, with monthly refreshers thereafter. Participants were instructed to meditate for 60 minutes a day in addition to the classes.
For the 144 people who were randomized to both meditation and exercise, this trial amounted to something of a part-time job. So you might think that adherence to the interventions was low, but apparently that’s not the case. Attendance to the mindfulness classes was over 90%, and over 80% for the exercise classes. And diary-based reporting of home efforts was also pretty good.
The control group wasn’t left to their own devices. Recognizing that the community aspect of exercise or mindfulness classes might convey a benefit independent of the actual exercise or mindfulness, the control group met on a similar schedule to discuss health education, but no mention of exercise or mindfulness occurred in that setting.
The primary outcome was change in memory and executive function scores across a battery of neuropsychologic testing, but the story is told in just a few pictures.
Memory scores improved in all three groups – mindfulness, exercise, and health education – over time. Cognitive composite score improved in all three groups similarly. There was no synergistic effect of mindfulness and exercise either. Basically, everyone got a bit better.
But the study did way more than look at scores on tests. Researchers used MRI to measure brain anatomic outcomes as well. And the surprising thing is that virtually none of these outcomes were different between the groups either.
Hippocampal volume decreased a bit in all the groups. Dorsolateral prefrontal cortex volume was flat. There was no change in scores measuring tasks of daily living.
When you see negative results like this, right away you worry that the intervention wasn’t properly delivered. Were these people really exercising and meditating? Well, the authors showed that individuals randomized to exercise, at least, had less sleep latency, greater aerobic fitness, and greater strength. So we know something was happening.
They then asked, would the people in the exercise group with the greatest changes in those physiologic parameters show some improvement in cognitive parameters? In other words, we know you were exercising because you got stronger and are sleeping better; is your memory better? The answer? Surprisingly, still no. Even in that honestly somewhat cherry-picked group, the interventions had no effect.
Could it be that the control was inappropriate, that the “health education” intervention was actually so helpful that it obscured the benefits of exercise and meditation? After all, cognitive scores did improve in all groups. The authors doubt it. They say they think the improvement in cognitive scores reflects the fact that patients had learned a bit about how to take the tests. This is pretty common in the neuropsychiatric literature.
So here we are and I just want to say, well, shoot. This is not the result I wanted. And I think the reason I’m so disappointed is because aging and the loss of cognitive faculties that comes with aging are just sort of scary. We are all looking for some control over that fear, and how nice it would be to be able to tell ourselves not to worry – that we won’t have those problems as we get older because we exercise, or meditate, or drink red wine, or don’t drink wine, or whatever. And while I have no doubt that staying healthier physically will keep you healthier mentally, it may take more than one simple thing to move the needle.
Dr. Wilson is associate professor, department of medicine, and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
We are coming to the end of the year, which always makes me think about getting older. Much like the search for the fountain of youth, many promising leads have ultimately led to dead ends. And yet, I had high hopes for a trial that focused on two cornerstones of wellness – exercise and mindfulness – to address the subjective loss of memory that comes with aging. Alas, meditation and exercise do not appear to be the fountain of youth.
I’m talking about this study, appearing in JAMA, known as the MEDEX trial.
It’s a clever design: a 2 x 2 factorial randomized trial where participants could be randomized to a mindfulness intervention, an exercise intervention, both, or neither.
In this manner, you can test multiple hypotheses exploiting a shared control group. Or as a mentor of mine used to say, you get two trials for the price of one and a half.
The participants were older adults, aged 65-84, living in the community. They had to be relatively sedentary at baseline and not engaging in mindfulness practices. They had to subjectively report some memory or concentration issues but had to be cognitively intact, based on a standard dementia screening test. In other words, these are your average older people who are worried that they aren’t as sharp as they used to be.
The interventions themselves were fairly intense. The exercise group had instructor-led sessions for 90 minutes twice a week for the first 6 months of the study, once a week thereafter. And participants were encouraged to exercise at home such that they had a total of 300 minutes of weekly exercise.
The mindfulness program was characterized by eight weekly classes of 2.5 hours each as well as a half-day retreat to teach the tenets of mindfulness and meditation, with monthly refreshers thereafter. Participants were instructed to meditate for 60 minutes a day in addition to the classes.
For the 144 people who were randomized to both meditation and exercise, this trial amounted to something of a part-time job. So you might think that adherence to the interventions was low, but apparently that’s not the case. Attendance to the mindfulness classes was over 90%, and over 80% for the exercise classes. And diary-based reporting of home efforts was also pretty good.
The control group wasn’t left to their own devices. Recognizing that the community aspect of exercise or mindfulness classes might convey a benefit independent of the actual exercise or mindfulness, the control group met on a similar schedule to discuss health education, but no mention of exercise or mindfulness occurred in that setting.
The primary outcome was change in memory and executive function scores across a battery of neuropsychologic testing, but the story is told in just a few pictures.
Memory scores improved in all three groups – mindfulness, exercise, and health education – over time. Cognitive composite score improved in all three groups similarly. There was no synergistic effect of mindfulness and exercise either. Basically, everyone got a bit better.
But the study did way more than look at scores on tests. Researchers used MRI to measure brain anatomic outcomes as well. And the surprising thing is that virtually none of these outcomes were different between the groups either.
Hippocampal volume decreased a bit in all the groups. Dorsolateral prefrontal cortex volume was flat. There was no change in scores measuring tasks of daily living.
When you see negative results like this, right away you worry that the intervention wasn’t properly delivered. Were these people really exercising and meditating? Well, the authors showed that individuals randomized to exercise, at least, had less sleep latency, greater aerobic fitness, and greater strength. So we know something was happening.
They then asked, would the people in the exercise group with the greatest changes in those physiologic parameters show some improvement in cognitive parameters? In other words, we know you were exercising because you got stronger and are sleeping better; is your memory better? The answer? Surprisingly, still no. Even in that honestly somewhat cherry-picked group, the interventions had no effect.
Could it be that the control was inappropriate, that the “health education” intervention was actually so helpful that it obscured the benefits of exercise and meditation? After all, cognitive scores did improve in all groups. The authors doubt it. They say they think the improvement in cognitive scores reflects the fact that patients had learned a bit about how to take the tests. This is pretty common in the neuropsychiatric literature.
So here we are and I just want to say, well, shoot. This is not the result I wanted. And I think the reason I’m so disappointed is because aging and the loss of cognitive faculties that comes with aging are just sort of scary. We are all looking for some control over that fear, and how nice it would be to be able to tell ourselves not to worry – that we won’t have those problems as we get older because we exercise, or meditate, or drink red wine, or don’t drink wine, or whatever. And while I have no doubt that staying healthier physically will keep you healthier mentally, it may take more than one simple thing to move the needle.
Dr. Wilson is associate professor, department of medicine, and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
We are coming to the end of the year, which always makes me think about getting older. Much like the search for the fountain of youth, many promising leads have ultimately led to dead ends. And yet, I had high hopes for a trial that focused on two cornerstones of wellness – exercise and mindfulness – to address the subjective loss of memory that comes with aging. Alas, meditation and exercise do not appear to be the fountain of youth.
I’m talking about this study, appearing in JAMA, known as the MEDEX trial.
It’s a clever design: a 2 x 2 factorial randomized trial where participants could be randomized to a mindfulness intervention, an exercise intervention, both, or neither.
In this manner, you can test multiple hypotheses exploiting a shared control group. Or as a mentor of mine used to say, you get two trials for the price of one and a half.
The participants were older adults, aged 65-84, living in the community. They had to be relatively sedentary at baseline and not engaging in mindfulness practices. They had to subjectively report some memory or concentration issues but had to be cognitively intact, based on a standard dementia screening test. In other words, these are your average older people who are worried that they aren’t as sharp as they used to be.
The interventions themselves were fairly intense. The exercise group had instructor-led sessions for 90 minutes twice a week for the first 6 months of the study, once a week thereafter. And participants were encouraged to exercise at home such that they had a total of 300 minutes of weekly exercise.
The mindfulness program was characterized by eight weekly classes of 2.5 hours each as well as a half-day retreat to teach the tenets of mindfulness and meditation, with monthly refreshers thereafter. Participants were instructed to meditate for 60 minutes a day in addition to the classes.
For the 144 people who were randomized to both meditation and exercise, this trial amounted to something of a part-time job. So you might think that adherence to the interventions was low, but apparently that’s not the case. Attendance to the mindfulness classes was over 90%, and over 80% for the exercise classes. And diary-based reporting of home efforts was also pretty good.
The control group wasn’t left to their own devices. Recognizing that the community aspect of exercise or mindfulness classes might convey a benefit independent of the actual exercise or mindfulness, the control group met on a similar schedule to discuss health education, but no mention of exercise or mindfulness occurred in that setting.
The primary outcome was change in memory and executive function scores across a battery of neuropsychologic testing, but the story is told in just a few pictures.
Memory scores improved in all three groups – mindfulness, exercise, and health education – over time. Cognitive composite score improved in all three groups similarly. There was no synergistic effect of mindfulness and exercise either. Basically, everyone got a bit better.
But the study did way more than look at scores on tests. Researchers used MRI to measure brain anatomic outcomes as well. And the surprising thing is that virtually none of these outcomes were different between the groups either.
Hippocampal volume decreased a bit in all the groups. Dorsolateral prefrontal cortex volume was flat. There was no change in scores measuring tasks of daily living.
When you see negative results like this, right away you worry that the intervention wasn’t properly delivered. Were these people really exercising and meditating? Well, the authors showed that individuals randomized to exercise, at least, had less sleep latency, greater aerobic fitness, and greater strength. So we know something was happening.
They then asked, would the people in the exercise group with the greatest changes in those physiologic parameters show some improvement in cognitive parameters? In other words, we know you were exercising because you got stronger and are sleeping better; is your memory better? The answer? Surprisingly, still no. Even in that honestly somewhat cherry-picked group, the interventions had no effect.
Could it be that the control was inappropriate, that the “health education” intervention was actually so helpful that it obscured the benefits of exercise and meditation? After all, cognitive scores did improve in all groups. The authors doubt it. They say they think the improvement in cognitive scores reflects the fact that patients had learned a bit about how to take the tests. This is pretty common in the neuropsychiatric literature.
So here we are and I just want to say, well, shoot. This is not the result I wanted. And I think the reason I’m so disappointed is because aging and the loss of cognitive faculties that comes with aging are just sort of scary. We are all looking for some control over that fear, and how nice it would be to be able to tell ourselves not to worry – that we won’t have those problems as we get older because we exercise, or meditate, or drink red wine, or don’t drink wine, or whatever. And while I have no doubt that staying healthier physically will keep you healthier mentally, it may take more than one simple thing to move the needle.
Dr. Wilson is associate professor, department of medicine, and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
How a cheap liver drug may be the key to preventing COVID
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
As soon as the pandemic started, the search was on for a medication that could stave off infection, or at least the worst consequences of infection.
One that would be cheap to make, safe, easy to distribute, and, ideally, was already available. The search had a quest-like quality, like something from a fairy tale. Society, poisoned by COVID, would find the antidote out there, somewhere, if we looked hard enough.
You know the story. There were some pretty dramatic failures: hydroxychloroquine, ivermectin. There were some successes, like dexamethasone.
I’m not here today to tell you that the antidote has been found – no, it takes large randomized trials to figure that out. But
How do you make a case that an existing drug – UDCA, in this case – might be useful to prevent or treat COVID? In contrast to prior basic-science studies, like the original ivermectin study, which essentially took a bunch of cells and virus in a tube filled with varying concentrations of the antiparasitic agent, the authors of this paper appearing in Nature give us multiple, complementary lines of evidence. Let me walk you through it.
All good science starts with a biologically plausible hypothesis. In this case, the authors recognized that SARS-CoV-2, in all its variants, requires the presence of the ACE2 receptor on the surface of cells to bind.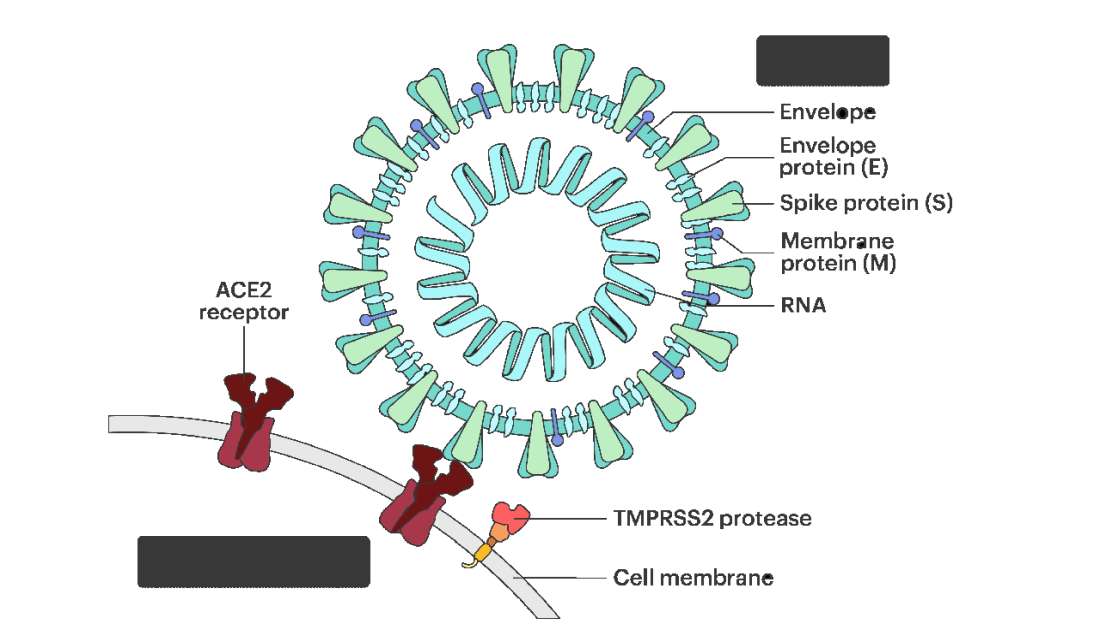
That is the doorway to infection. Vaccines and antibodies block the key to this door, the spike protein and its receptor binding domain. But what if you could get rid of the doors altogether?
The authors first showed that ACE2 expression is controlled by a certain transcription factor known as the farnesoid X receptor, or FXR. Reducing the binding of FXR should therefore reduce ACE2 expression.
As luck would have it, UDCA – Actigall – reduces the levels of FXR and thus the expression of ACE2 in cells.
Okay. So we have a drug that can reduce ACE2, and we know that ACE2 is necessary for the virus to infect cells. Would UDCA prevent viral infection?
They started with test tubes, showing that cells were less likely to be infected by SARS-CoV-2 in the presence of UDCA at concentrations similar to what humans achieve in their blood after standard dosing. The red staining here is spike protein; you can see that it is markedly lower in the cells exposed to UDCA.
So far, so good. But test tubes aren’t people. So they moved up to mice and Syrian golden hamsters. These cute fellows are quite susceptible to human COVID and have been a model organism in countless studies
Mice and hamsters treated with UDCA in the presence of littermates with COVID infections were less likely to become infected themselves compared with mice not so treated. They also showed that mice and hamsters treated with UDCA had lower levels of ACE2 in their nasal passages.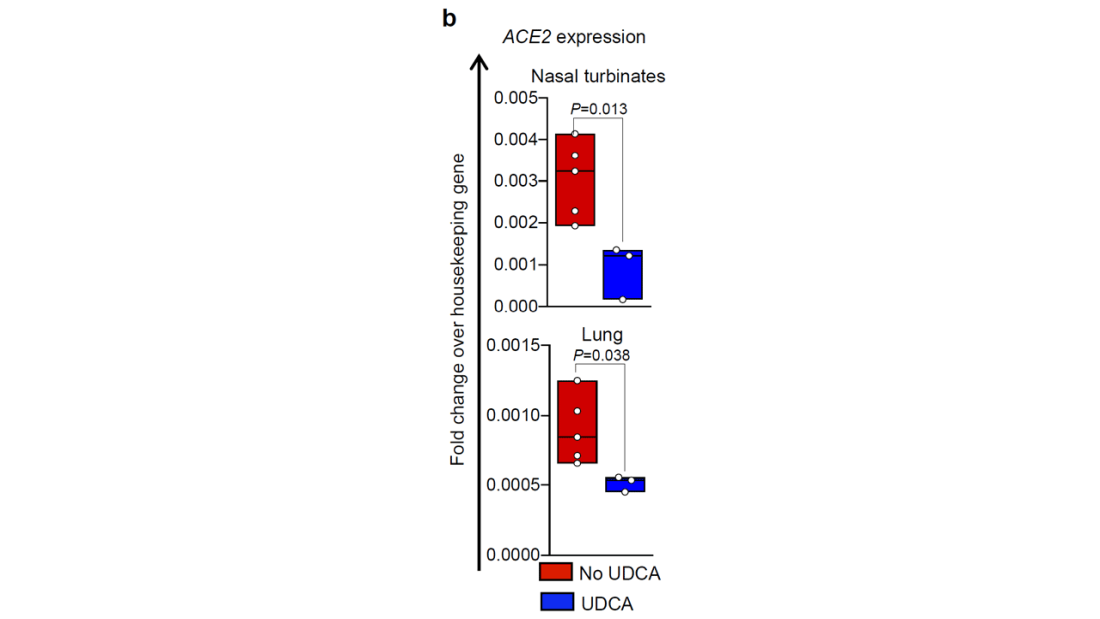
Of course, mice aren’t humans either. So the researchers didn’t stop there.
To determine the effects of UDCA on human tissue, they utilized perfused human lungs that had been declined for transplantation. The lungs were perfused with a special fluid to keep them viable, and were mechanically ventilated. One lung was exposed to UDCA and the other served as a control. The authors were able to show that ACE2 levels went down in the exposed lung. And, importantly, when samples of tissue from both lungs were exposed to SARS-CoV-2, the lung tissue exposed to UDCA had lower levels of viral infection.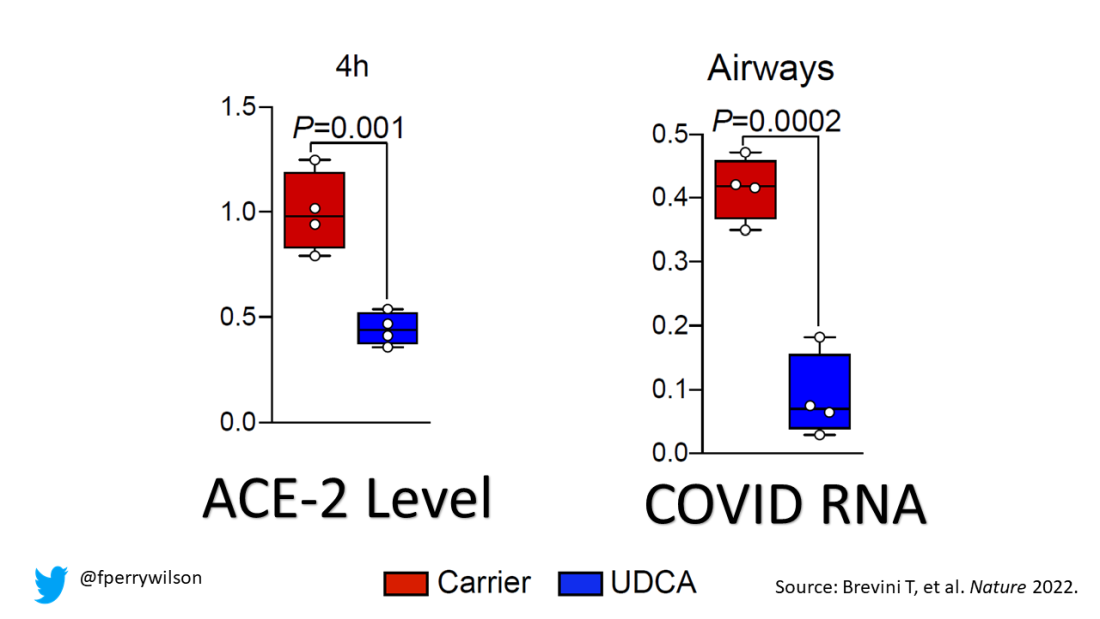
They didn’t stop there.
Eight human volunteers were recruited to take UDCA for 5 days. ACE2 levels in the nasal passages went down over the course of treatment. They confirmed those results from a proteomics dataset with several hundred people who had received UDCA for clinical reasons. Treated individuals had lower ACE2 levels.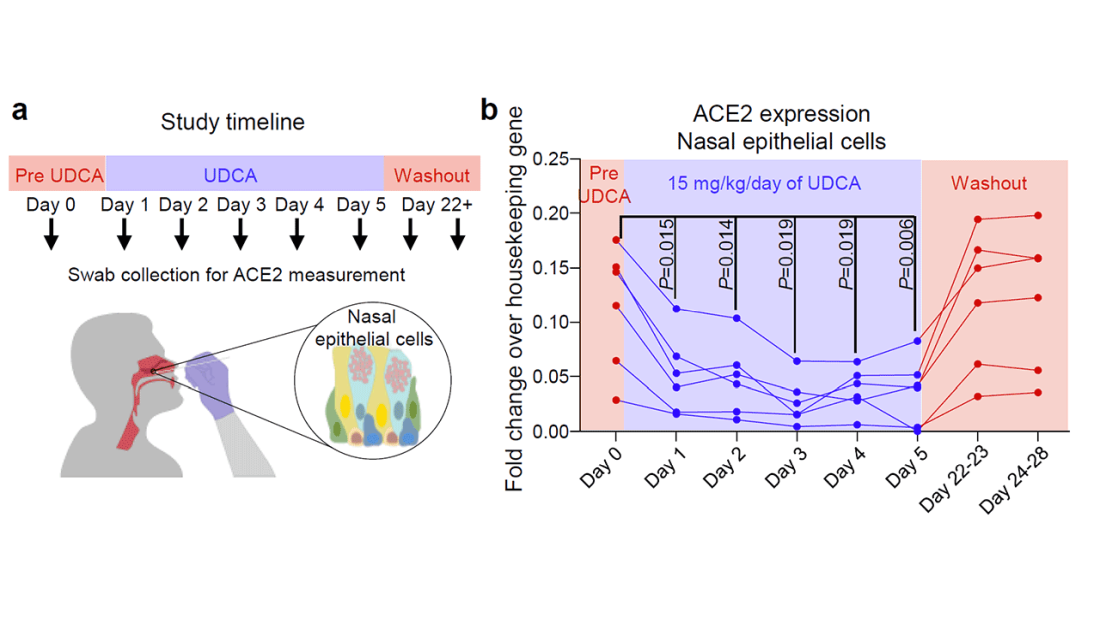
Finally, they looked at the epidemiologic effect. They examined a dataset that contained information on over 1,000 patients with liver disease who had contracted COVID-19, 31 of whom had been receiving UDCA. Even after adjustment for baseline differences, those receiving UDCA were less likely to be hospitalized, require an ICU, or die.
Okay, we’ll stop there. Reading this study, all I could think was, Yes! This is how you generate evidence that you have a drug that might work – step by careful step.
But let’s be careful as well. Does this study show that taking Actigall will prevent COVID? Of course not. It doesn’t show that it will treat COVID either. But I bring it up because the rigor of this study stands in contrast to those that generated huge enthusiasm earlier in the pandemic only to let us down in randomized trials. If there has been a drug out there this whole time which will prevent or treat COVID, this is how we’ll find it. The next step? Test it in a randomized trial.
For Medscape, I’m Perry Wilson.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. He disclosed no relevant financial relationships.
A version of this video transcript first appeared on Medscape.com.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
As soon as the pandemic started, the search was on for a medication that could stave off infection, or at least the worst consequences of infection.
One that would be cheap to make, safe, easy to distribute, and, ideally, was already available. The search had a quest-like quality, like something from a fairy tale. Society, poisoned by COVID, would find the antidote out there, somewhere, if we looked hard enough.
You know the story. There were some pretty dramatic failures: hydroxychloroquine, ivermectin. There were some successes, like dexamethasone.
I’m not here today to tell you that the antidote has been found – no, it takes large randomized trials to figure that out. But
How do you make a case that an existing drug – UDCA, in this case – might be useful to prevent or treat COVID? In contrast to prior basic-science studies, like the original ivermectin study, which essentially took a bunch of cells and virus in a tube filled with varying concentrations of the antiparasitic agent, the authors of this paper appearing in Nature give us multiple, complementary lines of evidence. Let me walk you through it.
All good science starts with a biologically plausible hypothesis. In this case, the authors recognized that SARS-CoV-2, in all its variants, requires the presence of the ACE2 receptor on the surface of cells to bind.
That is the doorway to infection. Vaccines and antibodies block the key to this door, the spike protein and its receptor binding domain. But what if you could get rid of the doors altogether?
The authors first showed that ACE2 expression is controlled by a certain transcription factor known as the farnesoid X receptor, or FXR. Reducing the binding of FXR should therefore reduce ACE2 expression.
As luck would have it, UDCA – Actigall – reduces the levels of FXR and thus the expression of ACE2 in cells.
Okay. So we have a drug that can reduce ACE2, and we know that ACE2 is necessary for the virus to infect cells. Would UDCA prevent viral infection?
They started with test tubes, showing that cells were less likely to be infected by SARS-CoV-2 in the presence of UDCA at concentrations similar to what humans achieve in their blood after standard dosing. The red staining here is spike protein; you can see that it is markedly lower in the cells exposed to UDCA.
So far, so good. But test tubes aren’t people. So they moved up to mice and Syrian golden hamsters. These cute fellows are quite susceptible to human COVID and have been a model organism in countless studies
Mice and hamsters treated with UDCA in the presence of littermates with COVID infections were less likely to become infected themselves compared with mice not so treated. They also showed that mice and hamsters treated with UDCA had lower levels of ACE2 in their nasal passages.
Of course, mice aren’t humans either. So the researchers didn’t stop there.
To determine the effects of UDCA on human tissue, they utilized perfused human lungs that had been declined for transplantation. The lungs were perfused with a special fluid to keep them viable, and were mechanically ventilated. One lung was exposed to UDCA and the other served as a control. The authors were able to show that ACE2 levels went down in the exposed lung. And, importantly, when samples of tissue from both lungs were exposed to SARS-CoV-2, the lung tissue exposed to UDCA had lower levels of viral infection.
They didn’t stop there.
Eight human volunteers were recruited to take UDCA for 5 days. ACE2 levels in the nasal passages went down over the course of treatment. They confirmed those results from a proteomics dataset with several hundred people who had received UDCA for clinical reasons. Treated individuals had lower ACE2 levels.
Finally, they looked at the epidemiologic effect. They examined a dataset that contained information on over 1,000 patients with liver disease who had contracted COVID-19, 31 of whom had been receiving UDCA. Even after adjustment for baseline differences, those receiving UDCA were less likely to be hospitalized, require an ICU, or die.
Okay, we’ll stop there. Reading this study, all I could think was, Yes! This is how you generate evidence that you have a drug that might work – step by careful step.
But let’s be careful as well. Does this study show that taking Actigall will prevent COVID? Of course not. It doesn’t show that it will treat COVID either. But I bring it up because the rigor of this study stands in contrast to those that generated huge enthusiasm earlier in the pandemic only to let us down in randomized trials. If there has been a drug out there this whole time which will prevent or treat COVID, this is how we’ll find it. The next step? Test it in a randomized trial.
For Medscape, I’m Perry Wilson.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. He disclosed no relevant financial relationships.
A version of this video transcript first appeared on Medscape.com.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
As soon as the pandemic started, the search was on for a medication that could stave off infection, or at least the worst consequences of infection.
One that would be cheap to make, safe, easy to distribute, and, ideally, was already available. The search had a quest-like quality, like something from a fairy tale. Society, poisoned by COVID, would find the antidote out there, somewhere, if we looked hard enough.
You know the story. There were some pretty dramatic failures: hydroxychloroquine, ivermectin. There were some successes, like dexamethasone.
I’m not here today to tell you that the antidote has been found – no, it takes large randomized trials to figure that out. But
How do you make a case that an existing drug – UDCA, in this case – might be useful to prevent or treat COVID? In contrast to prior basic-science studies, like the original ivermectin study, which essentially took a bunch of cells and virus in a tube filled with varying concentrations of the antiparasitic agent, the authors of this paper appearing in Nature give us multiple, complementary lines of evidence. Let me walk you through it.
All good science starts with a biologically plausible hypothesis. In this case, the authors recognized that SARS-CoV-2, in all its variants, requires the presence of the ACE2 receptor on the surface of cells to bind.
That is the doorway to infection. Vaccines and antibodies block the key to this door, the spike protein and its receptor binding domain. But what if you could get rid of the doors altogether?
The authors first showed that ACE2 expression is controlled by a certain transcription factor known as the farnesoid X receptor, or FXR. Reducing the binding of FXR should therefore reduce ACE2 expression.
As luck would have it, UDCA – Actigall – reduces the levels of FXR and thus the expression of ACE2 in cells.
Okay. So we have a drug that can reduce ACE2, and we know that ACE2 is necessary for the virus to infect cells. Would UDCA prevent viral infection?
They started with test tubes, showing that cells were less likely to be infected by SARS-CoV-2 in the presence of UDCA at concentrations similar to what humans achieve in their blood after standard dosing. The red staining here is spike protein; you can see that it is markedly lower in the cells exposed to UDCA.
So far, so good. But test tubes aren’t people. So they moved up to mice and Syrian golden hamsters. These cute fellows are quite susceptible to human COVID and have been a model organism in countless studies
Mice and hamsters treated with UDCA in the presence of littermates with COVID infections were less likely to become infected themselves compared with mice not so treated. They also showed that mice and hamsters treated with UDCA had lower levels of ACE2 in their nasal passages.
Of course, mice aren’t humans either. So the researchers didn’t stop there.
To determine the effects of UDCA on human tissue, they utilized perfused human lungs that had been declined for transplantation. The lungs were perfused with a special fluid to keep them viable, and were mechanically ventilated. One lung was exposed to UDCA and the other served as a control. The authors were able to show that ACE2 levels went down in the exposed lung. And, importantly, when samples of tissue from both lungs were exposed to SARS-CoV-2, the lung tissue exposed to UDCA had lower levels of viral infection.
They didn’t stop there.
Eight human volunteers were recruited to take UDCA for 5 days. ACE2 levels in the nasal passages went down over the course of treatment. They confirmed those results from a proteomics dataset with several hundred people who had received UDCA for clinical reasons. Treated individuals had lower ACE2 levels.
Finally, they looked at the epidemiologic effect. They examined a dataset that contained information on over 1,000 patients with liver disease who had contracted COVID-19, 31 of whom had been receiving UDCA. Even after adjustment for baseline differences, those receiving UDCA were less likely to be hospitalized, require an ICU, or die.
Okay, we’ll stop there. Reading this study, all I could think was, Yes! This is how you generate evidence that you have a drug that might work – step by careful step.
But let’s be careful as well. Does this study show that taking Actigall will prevent COVID? Of course not. It doesn’t show that it will treat COVID either. But I bring it up because the rigor of this study stands in contrast to those that generated huge enthusiasm earlier in the pandemic only to let us down in randomized trials. If there has been a drug out there this whole time which will prevent or treat COVID, this is how we’ll find it. The next step? Test it in a randomized trial.
For Medscape, I’m Perry Wilson.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and director of Yale’s Clinical and Translational Research Accelerator. He disclosed no relevant financial relationships.
A version of this video transcript first appeared on Medscape.com.
The surprising failure of vitamin D in deficient kids
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
My basic gripe is that you’ve got all these observational studies linking lower levels of vitamin D to everything from dementia to falls to cancer to COVID infection, and then you do a big randomized trial of supplementation and don’t see an effect.
And the explanation is that vitamin D is not necessarily the thing causing these bad outcomes; it’s a bystander – a canary in the coal mine. Your vitamin D level is a marker of your lifestyle; it’s higher in people who eat healthier foods, who exercise, and who spend more time out in the sun.
And yet ... if you were to ask me whether supplementing vitamin D in children with vitamin D deficiency would help them grow better and be healthier, I probably would have been on board for the idea.
And, it looks like, I would have been wrong.
Yes, it’s another negative randomized trial of vitamin D supplementation to add to the seemingly ever-growing body of literature suggesting that your money is better spent on a day at the park rather than buying D3 from your local GNC.
We are talking about this study, appearing in JAMA Pediatrics.
Briefly, 8,851 children from around Ulaanbaatar, Mongolia, were randomized to receive 14,000 international units of vitamin D3 or placebo every week for 3 years.
Before we get into the results of the study, I need to point out that this part of Mongolia has a high rate of vitamin D deficiency. Beyond that, a prior observational study by these authors had shown that lower vitamin D levels were linked to the risk of acquiring latent tuberculosis infection in this area. Other studies have linked vitamin D deficiency with poorer growth metrics in children. Given the global scourge that is TB (around 2 million deaths a year) and childhood malnutrition (around 10% of children around the world), vitamin D supplementation is incredibly attractive as a public health intervention. It is relatively low on side effects and, importantly, it is cheap – and thus scalable.
Back to the study. These kids had pretty poor vitamin D levels at baseline; 95% of them were deficient, based on the accepted standard of levels less than 20 ng/mL. Over 30% were severely deficient, with levels less than 10 ng/mL.
The initial purpose of this study was to see if supplementation would prevent TB, but that analysis, which was published a few months ago, was negative. Vitamin D levels went up dramatically in the intervention group – they were taking their pills – but there was no difference in the rate of latent TB infection, active TB, other respiratory infections, or even serum interferon gamma levels.
Nothing.
But to be fair, the TB seroconversion rate was lower than expected, potentially leading to an underpowered study.
Which brings us to the just-published analysis which moves away from infectious disease to something where vitamin D should have some stronger footing: growth.
Would the kids who were randomized to vitamin D, those same kids who got their vitamin D levels into the normal range over 3 years of supplementation, grow more or grow better than the kids who didn’t?
And, unfortunately, the answer is still no.
At the end of follow-up, height z scores were not different between the groups. BMI z scores were not different between the groups. Pubertal development was not different between the groups. This was true not only overall, but across various subgroups, including analyses of those kids who had vitamin D levels less than 10 ng/mL to start with.
So, what’s going on? There are two very broad possibilities we can endorse. First, there’s the idea that vitamin D supplementation simply doesn’t do much for health. This is supported, now, by a long string of large clinical trials that show no effect across a variety of disease states and predisease states. In other words, the observational data linking low vitamin D to bad outcomes is correlation, not causation.
Or we can take the tack of some vitamin D apologists and decide that this trial just got it wrong. Perhaps the dose wasn’t given correctly, or 3 years isn’t long enough to see a real difference, or the growth metrics were wrong, or vitamin D needs to be given alongside something else to really work and so on. This is fine; no study is perfect and there is always something to criticize, believe me. But we need to be careful not to fall into the baby-and-bathwater fallacy. Just because we think a study could have done something better, or differently, doesn’t mean we can ignore all the results. And as each new randomized trial of vitamin D supplementation comes out, it’s getting harder and harder to believe that these trialists keep getting their methods wrong. Maybe they are just testing something that doesn’t work.
What to do? Well, it should be obvious. If low vitamin D levels are linked to TB rates and poor growth but supplementation doesn’t fix the problem, then we have to fix what is upstream of the problem. We need to boost vitamin D levels not through supplements, but through nutrition, exercise, activity, and getting outside. That’s a randomized trial you can sign me up for any day.
Dr. Wilson is associate professor, department of medicine, Yale University, New Haven, Conn. He reported no relevant conflicts of interest.
A version of this video transcript first appeared on Medscape.com.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
My basic gripe is that you’ve got all these observational studies linking lower levels of vitamin D to everything from dementia to falls to cancer to COVID infection, and then you do a big randomized trial of supplementation and don’t see an effect.
And the explanation is that vitamin D is not necessarily the thing causing these bad outcomes; it’s a bystander – a canary in the coal mine. Your vitamin D level is a marker of your lifestyle; it’s higher in people who eat healthier foods, who exercise, and who spend more time out in the sun.
And yet ... if you were to ask me whether supplementing vitamin D in children with vitamin D deficiency would help them grow better and be healthier, I probably would have been on board for the idea.
And, it looks like, I would have been wrong.
Yes, it’s another negative randomized trial of vitamin D supplementation to add to the seemingly ever-growing body of literature suggesting that your money is better spent on a day at the park rather than buying D3 from your local GNC.
We are talking about this study, appearing in JAMA Pediatrics.
Briefly, 8,851 children from around Ulaanbaatar, Mongolia, were randomized to receive 14,000 international units of vitamin D3 or placebo every week for 3 years.
Before we get into the results of the study, I need to point out that this part of Mongolia has a high rate of vitamin D deficiency. Beyond that, a prior observational study by these authors had shown that lower vitamin D levels were linked to the risk of acquiring latent tuberculosis infection in this area. Other studies have linked vitamin D deficiency with poorer growth metrics in children. Given the global scourge that is TB (around 2 million deaths a year) and childhood malnutrition (around 10% of children around the world), vitamin D supplementation is incredibly attractive as a public health intervention. It is relatively low on side effects and, importantly, it is cheap – and thus scalable.
Back to the study. These kids had pretty poor vitamin D levels at baseline; 95% of them were deficient, based on the accepted standard of levels less than 20 ng/mL. Over 30% were severely deficient, with levels less than 10 ng/mL.
The initial purpose of this study was to see if supplementation would prevent TB, but that analysis, which was published a few months ago, was negative. Vitamin D levels went up dramatically in the intervention group – they were taking their pills – but there was no difference in the rate of latent TB infection, active TB, other respiratory infections, or even serum interferon gamma levels.
Nothing.
But to be fair, the TB seroconversion rate was lower than expected, potentially leading to an underpowered study.
Which brings us to the just-published analysis which moves away from infectious disease to something where vitamin D should have some stronger footing: growth.
Would the kids who were randomized to vitamin D, those same kids who got their vitamin D levels into the normal range over 3 years of supplementation, grow more or grow better than the kids who didn’t?
And, unfortunately, the answer is still no.
At the end of follow-up, height z scores were not different between the groups. BMI z scores were not different between the groups. Pubertal development was not different between the groups. This was true not only overall, but across various subgroups, including analyses of those kids who had vitamin D levels less than 10 ng/mL to start with.
So, what’s going on? There are two very broad possibilities we can endorse. First, there’s the idea that vitamin D supplementation simply doesn’t do much for health. This is supported, now, by a long string of large clinical trials that show no effect across a variety of disease states and predisease states. In other words, the observational data linking low vitamin D to bad outcomes is correlation, not causation.
Or we can take the tack of some vitamin D apologists and decide that this trial just got it wrong. Perhaps the dose wasn’t given correctly, or 3 years isn’t long enough to see a real difference, or the growth metrics were wrong, or vitamin D needs to be given alongside something else to really work and so on. This is fine; no study is perfect and there is always something to criticize, believe me. But we need to be careful not to fall into the baby-and-bathwater fallacy. Just because we think a study could have done something better, or differently, doesn’t mean we can ignore all the results. And as each new randomized trial of vitamin D supplementation comes out, it’s getting harder and harder to believe that these trialists keep getting their methods wrong. Maybe they are just testing something that doesn’t work.
What to do? Well, it should be obvious. If low vitamin D levels are linked to TB rates and poor growth but supplementation doesn’t fix the problem, then we have to fix what is upstream of the problem. We need to boost vitamin D levels not through supplements, but through nutrition, exercise, activity, and getting outside. That’s a randomized trial you can sign me up for any day.
Dr. Wilson is associate professor, department of medicine, Yale University, New Haven, Conn. He reported no relevant conflicts of interest.
A version of this video transcript first appeared on Medscape.com.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
My basic gripe is that you’ve got all these observational studies linking lower levels of vitamin D to everything from dementia to falls to cancer to COVID infection, and then you do a big randomized trial of supplementation and don’t see an effect.
And the explanation is that vitamin D is not necessarily the thing causing these bad outcomes; it’s a bystander – a canary in the coal mine. Your vitamin D level is a marker of your lifestyle; it’s higher in people who eat healthier foods, who exercise, and who spend more time out in the sun.
And yet ... if you were to ask me whether supplementing vitamin D in children with vitamin D deficiency would help them grow better and be healthier, I probably would have been on board for the idea.
And, it looks like, I would have been wrong.
Yes, it’s another negative randomized trial of vitamin D supplementation to add to the seemingly ever-growing body of literature suggesting that your money is better spent on a day at the park rather than buying D3 from your local GNC.
We are talking about this study, appearing in JAMA Pediatrics.
Briefly, 8,851 children from around Ulaanbaatar, Mongolia, were randomized to receive 14,000 international units of vitamin D3 or placebo every week for 3 years.
Before we get into the results of the study, I need to point out that this part of Mongolia has a high rate of vitamin D deficiency. Beyond that, a prior observational study by these authors had shown that lower vitamin D levels were linked to the risk of acquiring latent tuberculosis infection in this area. Other studies have linked vitamin D deficiency with poorer growth metrics in children. Given the global scourge that is TB (around 2 million deaths a year) and childhood malnutrition (around 10% of children around the world), vitamin D supplementation is incredibly attractive as a public health intervention. It is relatively low on side effects and, importantly, it is cheap – and thus scalable.
Back to the study. These kids had pretty poor vitamin D levels at baseline; 95% of them were deficient, based on the accepted standard of levels less than 20 ng/mL. Over 30% were severely deficient, with levels less than 10 ng/mL.
The initial purpose of this study was to see if supplementation would prevent TB, but that analysis, which was published a few months ago, was negative. Vitamin D levels went up dramatically in the intervention group – they were taking their pills – but there was no difference in the rate of latent TB infection, active TB, other respiratory infections, or even serum interferon gamma levels.
Nothing.
But to be fair, the TB seroconversion rate was lower than expected, potentially leading to an underpowered study.
Which brings us to the just-published analysis which moves away from infectious disease to something where vitamin D should have some stronger footing: growth.
Would the kids who were randomized to vitamin D, those same kids who got their vitamin D levels into the normal range over 3 years of supplementation, grow more or grow better than the kids who didn’t?
And, unfortunately, the answer is still no.
At the end of follow-up, height z scores were not different between the groups. BMI z scores were not different between the groups. Pubertal development was not different between the groups. This was true not only overall, but across various subgroups, including analyses of those kids who had vitamin D levels less than 10 ng/mL to start with.
So, what’s going on? There are two very broad possibilities we can endorse. First, there’s the idea that vitamin D supplementation simply doesn’t do much for health. This is supported, now, by a long string of large clinical trials that show no effect across a variety of disease states and predisease states. In other words, the observational data linking low vitamin D to bad outcomes is correlation, not causation.
Or we can take the tack of some vitamin D apologists and decide that this trial just got it wrong. Perhaps the dose wasn’t given correctly, or 3 years isn’t long enough to see a real difference, or the growth metrics were wrong, or vitamin D needs to be given alongside something else to really work and so on. This is fine; no study is perfect and there is always something to criticize, believe me. But we need to be careful not to fall into the baby-and-bathwater fallacy. Just because we think a study could have done something better, or differently, doesn’t mean we can ignore all the results. And as each new randomized trial of vitamin D supplementation comes out, it’s getting harder and harder to believe that these trialists keep getting their methods wrong. Maybe they are just testing something that doesn’t work.
What to do? Well, it should be obvious. If low vitamin D levels are linked to TB rates and poor growth but supplementation doesn’t fix the problem, then we have to fix what is upstream of the problem. We need to boost vitamin D levels not through supplements, but through nutrition, exercise, activity, and getting outside. That’s a randomized trial you can sign me up for any day.
Dr. Wilson is associate professor, department of medicine, Yale University, New Haven, Conn. He reported no relevant conflicts of interest.
A version of this video transcript first appeared on Medscape.com.
Love them or hate them, masks in schools work
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
On March 26, 2022, Hawaii became the last state in the United States to lift its indoor mask mandate. By the time the current school year started, there were essentially no public school mask mandates either.
Whether you viewed the mask as an emblem of stalwart defiance against a rampaging virus, or a scarlet letter emblematic of the overreaches of public policy, you probably aren’t seeing them much anymore.
And yet, the debate about masks still rages. Who was right, who was wrong? Who trusted science, and what does the science even say? If we brought our country into marriage counseling, would we be told it is time to move on? To look forward, not backward? To plan for our bright future together?
Perhaps. But this question isn’t really moot just because masks have largely disappeared in the United States. Variants may emerge that lead to more infection waves – and other pandemics may occur in the future. And so I think it is important to discuss a study that, with quite rigorous analysis, attempts to answer the following question: Did masking in schools lower students’ and teachers’ risk of COVID?
We are talking about this study, appearing in the New England Journal of Medicine. The short version goes like this.
Researchers had access to two important sources of data. One – an accounting of all the teachers and students (more than 300,000 of them) in 79 public, noncharter school districts in Eastern Massachusetts who tested positive for COVID every week. Two – the date that each of those school districts lifted their mask mandates or (in the case of two districts) didn’t.
Right away, I’m sure you’re thinking of potential issues. Districts that kept masks even when the statewide ban was lifted are likely quite a bit different from districts that dropped masks right away. You’re right, of course – hold on to that thought; we’ll get there.
But first – the big question – would districts that kept their masks on longer do better when it comes to the rate of COVID infection?
When everyone was masking, COVID case rates were pretty similar. Statewide mandates are lifted in late February – and most school districts remove their mandates within a few weeks – the black line are the two districts (Boston and Chelsea) where mask mandates remained in place.
Prior to the mask mandate lifting, you see very similar COVID rates in districts that would eventually remove the mandate and those that would not, with a bit of noise around the initial Omicron wave which saw just a huge amount of people get infected.
And then, after the mandate was lifted, separation. Districts that held on to masks longer had lower rates of COVID infection.
In all, over the 15-weeks of the study, there were roughly 12,000 extra cases of COVID in the mask-free school districts, which corresponds to about 35% of the total COVID burden during that time. And, yes, kids do well with COVID – on average. But 12,000 extra cases is enough to translate into a significant number of important clinical outcomes – think hospitalizations and post-COVID syndromes. And of course, maybe most importantly, missed school days. Positive kids were not allowed in class no matter what district they were in.
Okay – I promised we’d address confounders. This was not a cluster-randomized trial, where some school districts had their mandates removed based on the vicissitudes of a virtual coin flip, as much as many of us would have been interested to see that. The decision to remove masks was up to the various school boards – and they had a lot of pressure on them from many different directions. But all we need to worry about is whether any of those things that pressure a school board to keep masks on would ALSO lead to fewer COVID cases. That’s how confounders work, and how you can get false results in a study like this.
And yes – districts that kept the masks on longer were different than those who took them right off. But check out how they were different.
The districts that kept masks on longer had more low-income students. More Black and Latino students. More students per classroom. These are all risk factors that increase the risk of COVID infection. In other words, the confounding here goes in the opposite direction of the results. If anything, these factors should make you more certain that masking works.
The authors also adjusted for other factors – the community transmission of COVID-19, vaccination rates, school district sizes, and so on. No major change in the results.
One concern I addressed to Dr. Ellie Murray, the biostatistician on the study – could districts that removed masks simply have been testing more to compensate, leading to increased capturing of cases?
If anything, the schools that kept masks on were testing more than the schools that took them off – again that would tend to imply that the results are even stronger than what was reported.
Is this a perfect study? Of course not – it’s one study, it’s from one state. And the relatively large effects from keeping masks on for one or 2 weeks require us to really embrace the concept of exponential growth of infections, but, if COVID has taught us anything, it is that small changes in initial conditions can have pretty big effects.
My daughter, who goes to a public school here in Connecticut, unmasked, was home with COVID this past week. She’s fine. But you know what? She missed a week of school. I worked from home to be with her – though I didn’t test positive. And that is a real cost to both of us that I think we need to consider when we consider the value of masks. Yes, they’re annoying – but if they keep kids in school, might they be worth it? Perhaps not for now, as cases aren’t surging. But in the future, be it a particularly concerning variant, or a whole new pandemic, we should not discount the simple, cheap, and apparently beneficial act of wearing masks to decrease transmission.
Dr. Perry Wilson is an associate professor of medicine and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
On March 26, 2022, Hawaii became the last state in the United States to lift its indoor mask mandate. By the time the current school year started, there were essentially no public school mask mandates either.
Whether you viewed the mask as an emblem of stalwart defiance against a rampaging virus, or a scarlet letter emblematic of the overreaches of public policy, you probably aren’t seeing them much anymore.
And yet, the debate about masks still rages. Who was right, who was wrong? Who trusted science, and what does the science even say? If we brought our country into marriage counseling, would we be told it is time to move on? To look forward, not backward? To plan for our bright future together?
Perhaps. But this question isn’t really moot just because masks have largely disappeared in the United States. Variants may emerge that lead to more infection waves – and other pandemics may occur in the future. And so I think it is important to discuss a study that, with quite rigorous analysis, attempts to answer the following question: Did masking in schools lower students’ and teachers’ risk of COVID?
We are talking about this study, appearing in the New England Journal of Medicine. The short version goes like this.
Researchers had access to two important sources of data. One – an accounting of all the teachers and students (more than 300,000 of them) in 79 public, noncharter school districts in Eastern Massachusetts who tested positive for COVID every week. Two – the date that each of those school districts lifted their mask mandates or (in the case of two districts) didn’t.
Right away, I’m sure you’re thinking of potential issues. Districts that kept masks even when the statewide ban was lifted are likely quite a bit different from districts that dropped masks right away. You’re right, of course – hold on to that thought; we’ll get there.
But first – the big question – would districts that kept their masks on longer do better when it comes to the rate of COVID infection?
When everyone was masking, COVID case rates were pretty similar. Statewide mandates are lifted in late February – and most school districts remove their mandates within a few weeks – the black line are the two districts (Boston and Chelsea) where mask mandates remained in place.
Prior to the mask mandate lifting, you see very similar COVID rates in districts that would eventually remove the mandate and those that would not, with a bit of noise around the initial Omicron wave which saw just a huge amount of people get infected.
And then, after the mandate was lifted, separation. Districts that held on to masks longer had lower rates of COVID infection.
In all, over the 15-weeks of the study, there were roughly 12,000 extra cases of COVID in the mask-free school districts, which corresponds to about 35% of the total COVID burden during that time. And, yes, kids do well with COVID – on average. But 12,000 extra cases is enough to translate into a significant number of important clinical outcomes – think hospitalizations and post-COVID syndromes. And of course, maybe most importantly, missed school days. Positive kids were not allowed in class no matter what district they were in.
Okay – I promised we’d address confounders. This was not a cluster-randomized trial, where some school districts had their mandates removed based on the vicissitudes of a virtual coin flip, as much as many of us would have been interested to see that. The decision to remove masks was up to the various school boards – and they had a lot of pressure on them from many different directions. But all we need to worry about is whether any of those things that pressure a school board to keep masks on would ALSO lead to fewer COVID cases. That’s how confounders work, and how you can get false results in a study like this.
And yes – districts that kept the masks on longer were different than those who took them right off. But check out how they were different.
The districts that kept masks on longer had more low-income students. More Black and Latino students. More students per classroom. These are all risk factors that increase the risk of COVID infection. In other words, the confounding here goes in the opposite direction of the results. If anything, these factors should make you more certain that masking works.
The authors also adjusted for other factors – the community transmission of COVID-19, vaccination rates, school district sizes, and so on. No major change in the results.
One concern I addressed to Dr. Ellie Murray, the biostatistician on the study – could districts that removed masks simply have been testing more to compensate, leading to increased capturing of cases?
If anything, the schools that kept masks on were testing more than the schools that took them off – again that would tend to imply that the results are even stronger than what was reported.
Is this a perfect study? Of course not – it’s one study, it’s from one state. And the relatively large effects from keeping masks on for one or 2 weeks require us to really embrace the concept of exponential growth of infections, but, if COVID has taught us anything, it is that small changes in initial conditions can have pretty big effects.
My daughter, who goes to a public school here in Connecticut, unmasked, was home with COVID this past week. She’s fine. But you know what? She missed a week of school. I worked from home to be with her – though I didn’t test positive. And that is a real cost to both of us that I think we need to consider when we consider the value of masks. Yes, they’re annoying – but if they keep kids in school, might they be worth it? Perhaps not for now, as cases aren’t surging. But in the future, be it a particularly concerning variant, or a whole new pandemic, we should not discount the simple, cheap, and apparently beneficial act of wearing masks to decrease transmission.
Dr. Perry Wilson is an associate professor of medicine and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr. F. Perry Wilson of the Yale School of Medicine.
On March 26, 2022, Hawaii became the last state in the United States to lift its indoor mask mandate. By the time the current school year started, there were essentially no public school mask mandates either.
Whether you viewed the mask as an emblem of stalwart defiance against a rampaging virus, or a scarlet letter emblematic of the overreaches of public policy, you probably aren’t seeing them much anymore.
And yet, the debate about masks still rages. Who was right, who was wrong? Who trusted science, and what does the science even say? If we brought our country into marriage counseling, would we be told it is time to move on? To look forward, not backward? To plan for our bright future together?
Perhaps. But this question isn’t really moot just because masks have largely disappeared in the United States. Variants may emerge that lead to more infection waves – and other pandemics may occur in the future. And so I think it is important to discuss a study that, with quite rigorous analysis, attempts to answer the following question: Did masking in schools lower students’ and teachers’ risk of COVID?
We are talking about this study, appearing in the New England Journal of Medicine. The short version goes like this.
Researchers had access to two important sources of data. One – an accounting of all the teachers and students (more than 300,000 of them) in 79 public, noncharter school districts in Eastern Massachusetts who tested positive for COVID every week. Two – the date that each of those school districts lifted their mask mandates or (in the case of two districts) didn’t.
Right away, I’m sure you’re thinking of potential issues. Districts that kept masks even when the statewide ban was lifted are likely quite a bit different from districts that dropped masks right away. You’re right, of course – hold on to that thought; we’ll get there.
But first – the big question – would districts that kept their masks on longer do better when it comes to the rate of COVID infection?
When everyone was masking, COVID case rates were pretty similar. Statewide mandates are lifted in late February – and most school districts remove their mandates within a few weeks – the black line are the two districts (Boston and Chelsea) where mask mandates remained in place.
Prior to the mask mandate lifting, you see very similar COVID rates in districts that would eventually remove the mandate and those that would not, with a bit of noise around the initial Omicron wave which saw just a huge amount of people get infected.
And then, after the mandate was lifted, separation. Districts that held on to masks longer had lower rates of COVID infection.
In all, over the 15-weeks of the study, there were roughly 12,000 extra cases of COVID in the mask-free school districts, which corresponds to about 35% of the total COVID burden during that time. And, yes, kids do well with COVID – on average. But 12,000 extra cases is enough to translate into a significant number of important clinical outcomes – think hospitalizations and post-COVID syndromes. And of course, maybe most importantly, missed school days. Positive kids were not allowed in class no matter what district they were in.
Okay – I promised we’d address confounders. This was not a cluster-randomized trial, where some school districts had their mandates removed based on the vicissitudes of a virtual coin flip, as much as many of us would have been interested to see that. The decision to remove masks was up to the various school boards – and they had a lot of pressure on them from many different directions. But all we need to worry about is whether any of those things that pressure a school board to keep masks on would ALSO lead to fewer COVID cases. That’s how confounders work, and how you can get false results in a study like this.
And yes – districts that kept the masks on longer were different than those who took them right off. But check out how they were different.
The districts that kept masks on longer had more low-income students. More Black and Latino students. More students per classroom. These are all risk factors that increase the risk of COVID infection. In other words, the confounding here goes in the opposite direction of the results. If anything, these factors should make you more certain that masking works.
The authors also adjusted for other factors – the community transmission of COVID-19, vaccination rates, school district sizes, and so on. No major change in the results.
One concern I addressed to Dr. Ellie Murray, the biostatistician on the study – could districts that removed masks simply have been testing more to compensate, leading to increased capturing of cases?
If anything, the schools that kept masks on were testing more than the schools that took them off – again that would tend to imply that the results are even stronger than what was reported.
Is this a perfect study? Of course not – it’s one study, it’s from one state. And the relatively large effects from keeping masks on for one or 2 weeks require us to really embrace the concept of exponential growth of infections, but, if COVID has taught us anything, it is that small changes in initial conditions can have pretty big effects.
My daughter, who goes to a public school here in Connecticut, unmasked, was home with COVID this past week. She’s fine. But you know what? She missed a week of school. I worked from home to be with her – though I didn’t test positive. And that is a real cost to both of us that I think we need to consider when we consider the value of masks. Yes, they’re annoying – but if they keep kids in school, might they be worth it? Perhaps not for now, as cases aren’t surging. But in the future, be it a particularly concerning variant, or a whole new pandemic, we should not discount the simple, cheap, and apparently beneficial act of wearing masks to decrease transmission.
Dr. Perry Wilson is an associate professor of medicine and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.