User login
Comments & Controversies
More on T. gondii
We reviewed the article by Dr. Torrey on Toxoplasma gondii (T. gondii) and schizophrenia (“Cats, toxoplasmosis, and psychosis: Understanding the risks,”
The natural history of toxoplasmosis is an extraordinary example of nature’s complexity. The life cycle of this parasite uses the nervous system of the mouse to increase its transmission. Behavior changes ranging from reduced cat urine avoidance and increased risk-taking are observed in mice infected with T. gondii.2 Chronic toxoplasmosis may also affect human behavior.3
Cats are fascinating, complex creatures. Of note, they produce a protein structurally like the secretion of the slow loris.4 The loris uses this brachial gland protein secretion as part of a defense strategy.5 Consideration of a possible toxic, neuroimmune role of these small mammal proteins in psychiatric disorders may open other avenues to explore.6
Our relationship to domesticated animals has been connected to serious diseases throughout human history.7 Severe acute respiratory syndrome and COVID-19 appear to be linked to animal reservoirs, mammals of the small animal trade, and the fur industry.8,9 The rapid development of vaccines for COVID-19 is commendable. In conditions with multifactorial causation, managing an infectious component is worthy of consideration.
With mounting evidence suggesting a link between T. gondii and schizophrenia, ASD, and other diseases, further epidemiological studies and pilot interventions offer value. Interventions, including encouraging keeping cats indoors only, cat immunization, and human treatment, could be implemented in high-risk families. Efficacy requires data collection. While not easy, collaborative work by psychiatrists, developmental pediatricians, veterinarians, and epidemiologists is encouraged.
Mark C. Chandler, MD
Triangle Neuropsychiatry
Durham, North Carolina
Michelle Douglass, PA-S2
Duke University Physician Assistant Program
Durham, North Carolina
References
1. Nayeri T, Sarvi S, Moosazadeh M, et al. Relationship between toxoplasmosis and autism: a systematic review and meta-analysis. Microb Pathog. 2020;147:104434. doi:10.1016/j.micpath.2020.104434
2. Kochanowsky JA, Koshy AA. Toxoplasma gondii. Curr Biol. 2018;28(14):R770-R771. doi:10.1016/j.cub.2018.05.035
3. Letcher S. Parasite mind control: how a single celled parasite carried in the cat intestine may be quietly tweaking our behavior. Scientific Kenyon: The Neuroscience Edition. 2018;22(1):4-11.
4. Scheib H, Nekaris KA, Rode-Margono J, et al. The toxicological intersection between allergen and toxin: a structural comparison of the cat dander allergenic protein Fel d1 and the slow loris brachial gland secretion protein. Toxins (Basel). 2020;12(2):86. doi:10.3390/toxins12020086
5. Nekaris KA, Moore RS, Rode EJ, et al. Mad, bad and dangerous to know: the biochemistry, ecology and evolution of slow loris venom. J Venom Anim Toxins Incl Trop Dis. 2013;19(1):21. doi:10.1186/1678-9199-19-21
6. Ligabue-Braun R. Hello, kitty: could cat allergy be a form of intoxication? J Venom Anim Toxins Incl Trop Dis. 2020;26:e20200051. doi:10.1590/1678-9199-JVATITD-2020-0051
7. Pearce-Duvet JM. The origin of human pathogens: evaluating the role of agriculture and domestic animals in the evolution of human disease. Biol Rev Camb Philos Soc. 2006;81(3):369-382. doi:10.1017/S1464793106007020
8. Jo WK, de Oliveira-Filho EF, Rasche A, et al. Potential zoonotic sources of SARS-CoV-2 infections. Transbound Emerg Dis. 2021;68(4):1824-1834. doi:10.1111/tbed.13872
9. Bell D, Roberton S, Hunter PR. Animal origins of SARS coronavirus: possible links with the international trade in small carnivores. Philos Trans R Soc Lond B Biol Sci. 2004;359(1447):1107-1114. doi:10.1098/rstb.2004.1492
Disclosures
The authors report no financial relationships with any companies whose products are mentioned in this letter, or with manufacturers of competing products.
Continue to: Pramipexole for MDD
Pramipexole for MDD
I appreciate Dr. Espejo’s recommendations for treating patients who experience limited response from initial antidepressant therapy (“Treating major depressive disorder after limited response to an initial agent,”
Jonathan R. Scarff, MD
Lexington VA Health Care System
Lexington, Kentucky
References
1. Tundo A, de Filippis R, De Crescenzo F. Pramipexole in the treatment of unipolar and bipolar depression. A systematic review and meta-analysis. Acta Psychiatr Scand. 2019;140(2):116-125.
2. Tundo A, Betrò S, Iommi M, et al. Efficacy and safety of 24-week pramipexole augmentation in patients with treatment resistant depression. A retrospective cohort study. Prog Neuropsychopharmacol Biol Psychiatry. 2022;112:110425. doi:10.1016/j.pnpbp.2021.110425
Disclosures
The author reports no financial relationships with any companies whose products are mentioned in this letter, or with manufacturers of competing products.
More on T. gondii
We reviewed the article by Dr. Torrey on Toxoplasma gondii (T. gondii) and schizophrenia (“Cats, toxoplasmosis, and psychosis: Understanding the risks,”
The natural history of toxoplasmosis is an extraordinary example of nature’s complexity. The life cycle of this parasite uses the nervous system of the mouse to increase its transmission. Behavior changes ranging from reduced cat urine avoidance and increased risk-taking are observed in mice infected with T. gondii.2 Chronic toxoplasmosis may also affect human behavior.3
Cats are fascinating, complex creatures. Of note, they produce a protein structurally like the secretion of the slow loris.4 The loris uses this brachial gland protein secretion as part of a defense strategy.5 Consideration of a possible toxic, neuroimmune role of these small mammal proteins in psychiatric disorders may open other avenues to explore.6
Our relationship to domesticated animals has been connected to serious diseases throughout human history.7 Severe acute respiratory syndrome and COVID-19 appear to be linked to animal reservoirs, mammals of the small animal trade, and the fur industry.8,9 The rapid development of vaccines for COVID-19 is commendable. In conditions with multifactorial causation, managing an infectious component is worthy of consideration.
With mounting evidence suggesting a link between T. gondii and schizophrenia, ASD, and other diseases, further epidemiological studies and pilot interventions offer value. Interventions, including encouraging keeping cats indoors only, cat immunization, and human treatment, could be implemented in high-risk families. Efficacy requires data collection. While not easy, collaborative work by psychiatrists, developmental pediatricians, veterinarians, and epidemiologists is encouraged.
Mark C. Chandler, MD
Triangle Neuropsychiatry
Durham, North Carolina
Michelle Douglass, PA-S2
Duke University Physician Assistant Program
Durham, North Carolina
References
1. Nayeri T, Sarvi S, Moosazadeh M, et al. Relationship between toxoplasmosis and autism: a systematic review and meta-analysis. Microb Pathog. 2020;147:104434. doi:10.1016/j.micpath.2020.104434
2. Kochanowsky JA, Koshy AA. Toxoplasma gondii. Curr Biol. 2018;28(14):R770-R771. doi:10.1016/j.cub.2018.05.035
3. Letcher S. Parasite mind control: how a single celled parasite carried in the cat intestine may be quietly tweaking our behavior. Scientific Kenyon: The Neuroscience Edition. 2018;22(1):4-11.
4. Scheib H, Nekaris KA, Rode-Margono J, et al. The toxicological intersection between allergen and toxin: a structural comparison of the cat dander allergenic protein Fel d1 and the slow loris brachial gland secretion protein. Toxins (Basel). 2020;12(2):86. doi:10.3390/toxins12020086
5. Nekaris KA, Moore RS, Rode EJ, et al. Mad, bad and dangerous to know: the biochemistry, ecology and evolution of slow loris venom. J Venom Anim Toxins Incl Trop Dis. 2013;19(1):21. doi:10.1186/1678-9199-19-21
6. Ligabue-Braun R. Hello, kitty: could cat allergy be a form of intoxication? J Venom Anim Toxins Incl Trop Dis. 2020;26:e20200051. doi:10.1590/1678-9199-JVATITD-2020-0051
7. Pearce-Duvet JM. The origin of human pathogens: evaluating the role of agriculture and domestic animals in the evolution of human disease. Biol Rev Camb Philos Soc. 2006;81(3):369-382. doi:10.1017/S1464793106007020
8. Jo WK, de Oliveira-Filho EF, Rasche A, et al. Potential zoonotic sources of SARS-CoV-2 infections. Transbound Emerg Dis. 2021;68(4):1824-1834. doi:10.1111/tbed.13872
9. Bell D, Roberton S, Hunter PR. Animal origins of SARS coronavirus: possible links with the international trade in small carnivores. Philos Trans R Soc Lond B Biol Sci. 2004;359(1447):1107-1114. doi:10.1098/rstb.2004.1492
Disclosures
The authors report no financial relationships with any companies whose products are mentioned in this letter, or with manufacturers of competing products.
Continue to: Pramipexole for MDD
Pramipexole for MDD
I appreciate Dr. Espejo’s recommendations for treating patients who experience limited response from initial antidepressant therapy (“Treating major depressive disorder after limited response to an initial agent,”
Jonathan R. Scarff, MD
Lexington VA Health Care System
Lexington, Kentucky
References
1. Tundo A, de Filippis R, De Crescenzo F. Pramipexole in the treatment of unipolar and bipolar depression. A systematic review and meta-analysis. Acta Psychiatr Scand. 2019;140(2):116-125.
2. Tundo A, Betrò S, Iommi M, et al. Efficacy and safety of 24-week pramipexole augmentation in patients with treatment resistant depression. A retrospective cohort study. Prog Neuropsychopharmacol Biol Psychiatry. 2022;112:110425. doi:10.1016/j.pnpbp.2021.110425
Disclosures
The author reports no financial relationships with any companies whose products are mentioned in this letter, or with manufacturers of competing products.
More on T. gondii
We reviewed the article by Dr. Torrey on Toxoplasma gondii (T. gondii) and schizophrenia (“Cats, toxoplasmosis, and psychosis: Understanding the risks,”
The natural history of toxoplasmosis is an extraordinary example of nature’s complexity. The life cycle of this parasite uses the nervous system of the mouse to increase its transmission. Behavior changes ranging from reduced cat urine avoidance and increased risk-taking are observed in mice infected with T. gondii.2 Chronic toxoplasmosis may also affect human behavior.3
Cats are fascinating, complex creatures. Of note, they produce a protein structurally like the secretion of the slow loris.4 The loris uses this brachial gland protein secretion as part of a defense strategy.5 Consideration of a possible toxic, neuroimmune role of these small mammal proteins in psychiatric disorders may open other avenues to explore.6
Our relationship to domesticated animals has been connected to serious diseases throughout human history.7 Severe acute respiratory syndrome and COVID-19 appear to be linked to animal reservoirs, mammals of the small animal trade, and the fur industry.8,9 The rapid development of vaccines for COVID-19 is commendable. In conditions with multifactorial causation, managing an infectious component is worthy of consideration.
With mounting evidence suggesting a link between T. gondii and schizophrenia, ASD, and other diseases, further epidemiological studies and pilot interventions offer value. Interventions, including encouraging keeping cats indoors only, cat immunization, and human treatment, could be implemented in high-risk families. Efficacy requires data collection. While not easy, collaborative work by psychiatrists, developmental pediatricians, veterinarians, and epidemiologists is encouraged.
Mark C. Chandler, MD
Triangle Neuropsychiatry
Durham, North Carolina
Michelle Douglass, PA-S2
Duke University Physician Assistant Program
Durham, North Carolina
References
1. Nayeri T, Sarvi S, Moosazadeh M, et al. Relationship between toxoplasmosis and autism: a systematic review and meta-analysis. Microb Pathog. 2020;147:104434. doi:10.1016/j.micpath.2020.104434
2. Kochanowsky JA, Koshy AA. Toxoplasma gondii. Curr Biol. 2018;28(14):R770-R771. doi:10.1016/j.cub.2018.05.035
3. Letcher S. Parasite mind control: how a single celled parasite carried in the cat intestine may be quietly tweaking our behavior. Scientific Kenyon: The Neuroscience Edition. 2018;22(1):4-11.
4. Scheib H, Nekaris KA, Rode-Margono J, et al. The toxicological intersection between allergen and toxin: a structural comparison of the cat dander allergenic protein Fel d1 and the slow loris brachial gland secretion protein. Toxins (Basel). 2020;12(2):86. doi:10.3390/toxins12020086
5. Nekaris KA, Moore RS, Rode EJ, et al. Mad, bad and dangerous to know: the biochemistry, ecology and evolution of slow loris venom. J Venom Anim Toxins Incl Trop Dis. 2013;19(1):21. doi:10.1186/1678-9199-19-21
6. Ligabue-Braun R. Hello, kitty: could cat allergy be a form of intoxication? J Venom Anim Toxins Incl Trop Dis. 2020;26:e20200051. doi:10.1590/1678-9199-JVATITD-2020-0051
7. Pearce-Duvet JM. The origin of human pathogens: evaluating the role of agriculture and domestic animals in the evolution of human disease. Biol Rev Camb Philos Soc. 2006;81(3):369-382. doi:10.1017/S1464793106007020
8. Jo WK, de Oliveira-Filho EF, Rasche A, et al. Potential zoonotic sources of SARS-CoV-2 infections. Transbound Emerg Dis. 2021;68(4):1824-1834. doi:10.1111/tbed.13872
9. Bell D, Roberton S, Hunter PR. Animal origins of SARS coronavirus: possible links with the international trade in small carnivores. Philos Trans R Soc Lond B Biol Sci. 2004;359(1447):1107-1114. doi:10.1098/rstb.2004.1492
Disclosures
The authors report no financial relationships with any companies whose products are mentioned in this letter, or with manufacturers of competing products.
Continue to: Pramipexole for MDD
Pramipexole for MDD
I appreciate Dr. Espejo’s recommendations for treating patients who experience limited response from initial antidepressant therapy (“Treating major depressive disorder after limited response to an initial agent,”
Jonathan R. Scarff, MD
Lexington VA Health Care System
Lexington, Kentucky
References
1. Tundo A, de Filippis R, De Crescenzo F. Pramipexole in the treatment of unipolar and bipolar depression. A systematic review and meta-analysis. Acta Psychiatr Scand. 2019;140(2):116-125.
2. Tundo A, Betrò S, Iommi M, et al. Efficacy and safety of 24-week pramipexole augmentation in patients with treatment resistant depression. A retrospective cohort study. Prog Neuropsychopharmacol Biol Psychiatry. 2022;112:110425. doi:10.1016/j.pnpbp.2021.110425
Disclosures
The author reports no financial relationships with any companies whose products are mentioned in this letter, or with manufacturers of competing products.
“Provider” Etymology is Unclear, but Still Wrong for Health Care
I am grateful for the opportunity to clarify and correct my recent commentary.1 I wrote that the word provider was first used to refer to health care professionals during the 1930s in Nazi Germany, when Jewish physicians were termed Behandler. The cited manuscript stated that Behandler was “freely translated” as “provider.”2 However, after reading social media comments that claimed this was a mistranslation, I sought to verify the translation.
Online German-English dictionaries yielded perplexing results. The dictionary Reverso translates Behandler as “dentist,” “practitioner,” or “therapist.”3 The Past Tenses Dictionary translates Behandler as “handlers.”4 Although a distasteful way to refer to a clinician-patient relationship, it still doesn’t translate as “provider.” The Collins and Cambridge dictionaries do not include Behandler, and the Langenscheidt dictionary does not provide a translation, instead noting that the translation “is missing” and that they are “verifying the word in question.”5-7 Conversely, Anbieter appears to be the commonly provided German translation for provider.
The author of the original manuscript acknowledged that although Behandler is not listed as a translation for provider, it “comes close.”2 He added that Behandler is not used anymore in German medicine because of the Nazi past (Saenger P, personal communication, February 9, 2022). A native German and Professor of German Studies at the University of Kentucky shared that “My best guess is that the term Behandler was used as a short form of Krankenbehandler, the designation for Jewish doctors in Nazi Germany who were still allowed to treat Jewish patients after withdrawal of their medical license. The best translations would be (health) practitioner or health care provider.” (Hobusch H, personal communication, 2022). However, Krankenbehandler has also been translated as “practitioner of the sick.”8
Given this ambiguity, it is ultimately unclear whether or to what extent Behandler can be translated as provider. Despite this uncertainty, my original argument remains unchanged. It is best to refer to all health care professionals (eg, psychotherapists, physicians, nurses, phlebotomists, pharmacists, physician assistants, social workers, physical therapists, dentists, optometrists) by their credentials. Overarching terms such as clinicians, practitioners, or health care professionals also are reasonable. This ensures accurate terminology, respects individuals’ unique training and degrees, and avoids confusion within multidisciplinary health care settings.
I thank Paul Saenger, MD, and Harald Höbusch, PhD, for their helpful insights, and those individuals who raised this concern on social media.
Correction: Scarff JR. What’s in a name? The problematic term “provider”. Fed Pract. 2021;38(10):446- 448. The translation of the German word Behandler is unclear; therefore, the word “provider” should not be directly associated with the Nazi regime and its treatment of Jewish physicians.1. Scarff JR. What’s in a name? The problematic term “provider”. Fed Pract. 2021;38(10):446-448. doi:10.12788/fp.0188
2. Saenger P. Jewish pediatricians in Nazi Germany: victims of persecution. Isr Med Assoc J. 2006;8(5):324-328.
3. Reverso German-English Dictionary. Behandler. Accessed March 16, 2022. https://dictionary.reverso.net/german-english /behandler/forced
4. Past Tenses. Translate behandler in English. Accessed March 4, 2022. https://pasttenses.com/behandler-german-english
5. Collins Reverso German-English Dictionary. Behandler. Accessed March 16, 2022. https://www.collinsdictionary.com /dictionary/german-english/behandeln
6. Cambridge Dictionary, German-–English Dictionary. Behandler. Accessed March 16, 2022. https://dictionary.cambridge.org /spellcheck/german-english/?q=behandler
7. Langenscheidt Dictionary, German-English. Behandler. Accessed March 4, 2022. https://en.langenscheidt .com/german-english/search?term=behandler&q _cat=%2Fgerman-english%2F
8. Von Villiez A [trans, Kummer I]. The disenfranchisement of Jewish physicians in Hamburg during National Socialism. In: Institut fur die Geschichte der Deutschen Juden [Institute for the history of German Jews]. Key Documents of German-Jewish History. Updated September 16, 2016. Accessed March 16, 2022. doi: 10.23691/jgo:article-156.en.v1
I am grateful for the opportunity to clarify and correct my recent commentary.1 I wrote that the word provider was first used to refer to health care professionals during the 1930s in Nazi Germany, when Jewish physicians were termed Behandler. The cited manuscript stated that Behandler was “freely translated” as “provider.”2 However, after reading social media comments that claimed this was a mistranslation, I sought to verify the translation.
Online German-English dictionaries yielded perplexing results. The dictionary Reverso translates Behandler as “dentist,” “practitioner,” or “therapist.”3 The Past Tenses Dictionary translates Behandler as “handlers.”4 Although a distasteful way to refer to a clinician-patient relationship, it still doesn’t translate as “provider.” The Collins and Cambridge dictionaries do not include Behandler, and the Langenscheidt dictionary does not provide a translation, instead noting that the translation “is missing” and that they are “verifying the word in question.”5-7 Conversely, Anbieter appears to be the commonly provided German translation for provider.
The author of the original manuscript acknowledged that although Behandler is not listed as a translation for provider, it “comes close.”2 He added that Behandler is not used anymore in German medicine because of the Nazi past (Saenger P, personal communication, February 9, 2022). A native German and Professor of German Studies at the University of Kentucky shared that “My best guess is that the term Behandler was used as a short form of Krankenbehandler, the designation for Jewish doctors in Nazi Germany who were still allowed to treat Jewish patients after withdrawal of their medical license. The best translations would be (health) practitioner or health care provider.” (Hobusch H, personal communication, 2022). However, Krankenbehandler has also been translated as “practitioner of the sick.”8
Given this ambiguity, it is ultimately unclear whether or to what extent Behandler can be translated as provider. Despite this uncertainty, my original argument remains unchanged. It is best to refer to all health care professionals (eg, psychotherapists, physicians, nurses, phlebotomists, pharmacists, physician assistants, social workers, physical therapists, dentists, optometrists) by their credentials. Overarching terms such as clinicians, practitioners, or health care professionals also are reasonable. This ensures accurate terminology, respects individuals’ unique training and degrees, and avoids confusion within multidisciplinary health care settings.
I thank Paul Saenger, MD, and Harald Höbusch, PhD, for their helpful insights, and those individuals who raised this concern on social media.
Correction: Scarff JR. What’s in a name? The problematic term “provider”. Fed Pract. 2021;38(10):446- 448. The translation of the German word Behandler is unclear; therefore, the word “provider” should not be directly associated with the Nazi regime and its treatment of Jewish physicians.I am grateful for the opportunity to clarify and correct my recent commentary.1 I wrote that the word provider was first used to refer to health care professionals during the 1930s in Nazi Germany, when Jewish physicians were termed Behandler. The cited manuscript stated that Behandler was “freely translated” as “provider.”2 However, after reading social media comments that claimed this was a mistranslation, I sought to verify the translation.
Online German-English dictionaries yielded perplexing results. The dictionary Reverso translates Behandler as “dentist,” “practitioner,” or “therapist.”3 The Past Tenses Dictionary translates Behandler as “handlers.”4 Although a distasteful way to refer to a clinician-patient relationship, it still doesn’t translate as “provider.” The Collins and Cambridge dictionaries do not include Behandler, and the Langenscheidt dictionary does not provide a translation, instead noting that the translation “is missing” and that they are “verifying the word in question.”5-7 Conversely, Anbieter appears to be the commonly provided German translation for provider.
The author of the original manuscript acknowledged that although Behandler is not listed as a translation for provider, it “comes close.”2 He added that Behandler is not used anymore in German medicine because of the Nazi past (Saenger P, personal communication, February 9, 2022). A native German and Professor of German Studies at the University of Kentucky shared that “My best guess is that the term Behandler was used as a short form of Krankenbehandler, the designation for Jewish doctors in Nazi Germany who were still allowed to treat Jewish patients after withdrawal of their medical license. The best translations would be (health) practitioner or health care provider.” (Hobusch H, personal communication, 2022). However, Krankenbehandler has also been translated as “practitioner of the sick.”8
Given this ambiguity, it is ultimately unclear whether or to what extent Behandler can be translated as provider. Despite this uncertainty, my original argument remains unchanged. It is best to refer to all health care professionals (eg, psychotherapists, physicians, nurses, phlebotomists, pharmacists, physician assistants, social workers, physical therapists, dentists, optometrists) by their credentials. Overarching terms such as clinicians, practitioners, or health care professionals also are reasonable. This ensures accurate terminology, respects individuals’ unique training and degrees, and avoids confusion within multidisciplinary health care settings.
I thank Paul Saenger, MD, and Harald Höbusch, PhD, for their helpful insights, and those individuals who raised this concern on social media.
Correction: Scarff JR. What’s in a name? The problematic term “provider”. Fed Pract. 2021;38(10):446- 448. The translation of the German word Behandler is unclear; therefore, the word “provider” should not be directly associated with the Nazi regime and its treatment of Jewish physicians.1. Scarff JR. What’s in a name? The problematic term “provider”. Fed Pract. 2021;38(10):446-448. doi:10.12788/fp.0188
2. Saenger P. Jewish pediatricians in Nazi Germany: victims of persecution. Isr Med Assoc J. 2006;8(5):324-328.
3. Reverso German-English Dictionary. Behandler. Accessed March 16, 2022. https://dictionary.reverso.net/german-english /behandler/forced
4. Past Tenses. Translate behandler in English. Accessed March 4, 2022. https://pasttenses.com/behandler-german-english
5. Collins Reverso German-English Dictionary. Behandler. Accessed March 16, 2022. https://www.collinsdictionary.com /dictionary/german-english/behandeln
6. Cambridge Dictionary, German-–English Dictionary. Behandler. Accessed March 16, 2022. https://dictionary.cambridge.org /spellcheck/german-english/?q=behandler
7. Langenscheidt Dictionary, German-English. Behandler. Accessed March 4, 2022. https://en.langenscheidt .com/german-english/search?term=behandler&q _cat=%2Fgerman-english%2F
8. Von Villiez A [trans, Kummer I]. The disenfranchisement of Jewish physicians in Hamburg during National Socialism. In: Institut fur die Geschichte der Deutschen Juden [Institute for the history of German Jews]. Key Documents of German-Jewish History. Updated September 16, 2016. Accessed March 16, 2022. doi: 10.23691/jgo:article-156.en.v1
1. Scarff JR. What’s in a name? The problematic term “provider”. Fed Pract. 2021;38(10):446-448. doi:10.12788/fp.0188
2. Saenger P. Jewish pediatricians in Nazi Germany: victims of persecution. Isr Med Assoc J. 2006;8(5):324-328.
3. Reverso German-English Dictionary. Behandler. Accessed March 16, 2022. https://dictionary.reverso.net/german-english /behandler/forced
4. Past Tenses. Translate behandler in English. Accessed March 4, 2022. https://pasttenses.com/behandler-german-english
5. Collins Reverso German-English Dictionary. Behandler. Accessed March 16, 2022. https://www.collinsdictionary.com /dictionary/german-english/behandeln
6. Cambridge Dictionary, German-–English Dictionary. Behandler. Accessed March 16, 2022. https://dictionary.cambridge.org /spellcheck/german-english/?q=behandler
7. Langenscheidt Dictionary, German-English. Behandler. Accessed March 4, 2022. https://en.langenscheidt .com/german-english/search?term=behandler&q _cat=%2Fgerman-english%2F
8. Von Villiez A [trans, Kummer I]. The disenfranchisement of Jewish physicians in Hamburg during National Socialism. In: Institut fur die Geschichte der Deutschen Juden [Institute for the history of German Jews]. Key Documents of German-Jewish History. Updated September 16, 2016. Accessed March 16, 2022. doi: 10.23691/jgo:article-156.en.v1
Intermittent fasting: What to tell patients
Intermittent fasting is the purposeful, restricted intake of food (and sometimes water), usually for health or religious reasons. Common forms are alternative-day fasting or time-restricted fasting, with variable ratios of days or hours for fasting and eating/drinking.1 For example, fasting during Ramadan, the ninth month of the Islamic calendar, occurs from dawn to sunset, for a variable duration due to latitude and seasonal shifts.2 Clinicians are likely to care for a patient who occasionally fasts. While there are potential benefits of fasting, clinicians need to consider the implications for patients who fast, particularly those receiving psychotropic medications.
Potential benefits for weight loss, mood
Some research suggests fasting is popular and may have benefits for an individual’s physical and mental health. In a 2020 online poll (N = 1,241), 24% of respondents said they had tried intermittent fasting, and 87% said the practice was very effective (50%) or somewhat effective (37%) in helping them lose weight.3 While more randomized control trials are needed to examine the practice’s effectiveness in promoting and maintaining weight loss, fasting has been linked to better glucose control in both humans and animals, and patients may have better adherence with fasting compared to caloric restriction alone.1 Improved mood, alertness, tranquility, and sometimes euphoria have been documented among individuals who fast, but these benefits may not be sustained.4 A prospective study of 462 participants who fasted during Ramadan found the practice reduced depression in patients with diabetes, possibly due to mindfulness, decreased inflammation from improved insulin sensitivity, and/or social cohesion.5
Be aware of the potential risks
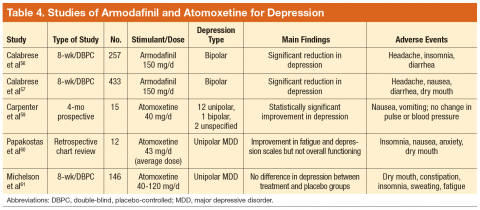
Fasting may either improve or destabilize mood in people with bipolar disorder by disrupting circadian rhythm and sleep.2 Fasting might exacerbate underlying eating disorders.2 Increased dehydration escalates the risk for orthostatic hypotension, which might require discontinuing clozapine.6 Hypotension and toxicity might arise during lithium pharmacotherapy. The Table4 summarizes things to consider when caring for a patient who fasts while receiving pharmacotherapy.

Provide patients with guidance
Advise patients not to fast if you believe it might exacerbate their mental illness, and encourage them to discuss with their primary care physicians any potential worsening of physical illnesses.2 When caring for a patient who fasts for religious reasons, consider consulting with the patient’s religious leaders.2 If patients choose to fast, monitor them for mood destabilization and/or medication adverse effects. If possible, avoid altering drug treatment regimens during fasting, and carefully monitor whenever a pharmaceutical change is necessary. When appropriate, the use of long-acting injectable medications may minimize adverse effects while maintaining mood stability. Encourage patients who fast to ensure they remain hydrated and practice sleep hygiene while they fast.7
1. Dong TA, Sandesara PB, Dhindsa DS, et al. Intermittent fasting: a heart healthy dietary pattern? Am J Med. 2020;133(8):901-907.
2. Fond G, Macgregor A, Leboyer M, et al. Fasting in mood disorders: neurobiology and effectiveness. A review of the literature. Psychiatry Res. 2013;209(3):253-258.
3. Ballard J. Americans say this popular diet is effective and inexpensive. YouGov. February 24, 2020. Accessed January 6, 2022. https://today.yougov.com/topics/food/articles-reports/2020/02/24/most-effective-diet-intermittent-fasting-poll
4. Furqan Z, Awaad R, Kurdyak P, et al. Considerations for clinicians treating Muslim patients with psychiatric disorders during Ramadan. Lancet Psychiatry. 2019;6(7):556-557.
5. Al-Ozairi E, AlAwadhi MM, Al-Ozairi A, et al. A prospective study of the effect of fasting during the month of Ramadan on depression and diabetes distress in people with type 2 diabetes. Diabet Res Clin Pract. 2019;153:145-149.
6. Chehovich C, Demler TL, Leppien E. Impact of Ramadan fasting on medical and psychiatric health. Int Clin Psychopharmacol. 2019;34(6):317-322.
7. Farooq S, Nazar Z, Akhtar J, et al. Effect of fasting during Ramadan on serum lithium level and mental state in bipolar affective disorder. Int Clin Psychopharmacol. 2010;25(6):323-327.
Intermittent fasting is the purposeful, restricted intake of food (and sometimes water), usually for health or religious reasons. Common forms are alternative-day fasting or time-restricted fasting, with variable ratios of days or hours for fasting and eating/drinking.1 For example, fasting during Ramadan, the ninth month of the Islamic calendar, occurs from dawn to sunset, for a variable duration due to latitude and seasonal shifts.2 Clinicians are likely to care for a patient who occasionally fasts. While there are potential benefits of fasting, clinicians need to consider the implications for patients who fast, particularly those receiving psychotropic medications.
Potential benefits for weight loss, mood
Some research suggests fasting is popular and may have benefits for an individual’s physical and mental health. In a 2020 online poll (N = 1,241), 24% of respondents said they had tried intermittent fasting, and 87% said the practice was very effective (50%) or somewhat effective (37%) in helping them lose weight.3 While more randomized control trials are needed to examine the practice’s effectiveness in promoting and maintaining weight loss, fasting has been linked to better glucose control in both humans and animals, and patients may have better adherence with fasting compared to caloric restriction alone.1 Improved mood, alertness, tranquility, and sometimes euphoria have been documented among individuals who fast, but these benefits may not be sustained.4 A prospective study of 462 participants who fasted during Ramadan found the practice reduced depression in patients with diabetes, possibly due to mindfulness, decreased inflammation from improved insulin sensitivity, and/or social cohesion.5
Be aware of the potential risks
Fasting may either improve or destabilize mood in people with bipolar disorder by disrupting circadian rhythm and sleep.2 Fasting might exacerbate underlying eating disorders.2 Increased dehydration escalates the risk for orthostatic hypotension, which might require discontinuing clozapine.6 Hypotension and toxicity might arise during lithium pharmacotherapy. The Table4 summarizes things to consider when caring for a patient who fasts while receiving pharmacotherapy.

Provide patients with guidance
Advise patients not to fast if you believe it might exacerbate their mental illness, and encourage them to discuss with their primary care physicians any potential worsening of physical illnesses.2 When caring for a patient who fasts for religious reasons, consider consulting with the patient’s religious leaders.2 If patients choose to fast, monitor them for mood destabilization and/or medication adverse effects. If possible, avoid altering drug treatment regimens during fasting, and carefully monitor whenever a pharmaceutical change is necessary. When appropriate, the use of long-acting injectable medications may minimize adverse effects while maintaining mood stability. Encourage patients who fast to ensure they remain hydrated and practice sleep hygiene while they fast.7
Intermittent fasting is the purposeful, restricted intake of food (and sometimes water), usually for health or religious reasons. Common forms are alternative-day fasting or time-restricted fasting, with variable ratios of days or hours for fasting and eating/drinking.1 For example, fasting during Ramadan, the ninth month of the Islamic calendar, occurs from dawn to sunset, for a variable duration due to latitude and seasonal shifts.2 Clinicians are likely to care for a patient who occasionally fasts. While there are potential benefits of fasting, clinicians need to consider the implications for patients who fast, particularly those receiving psychotropic medications.
Potential benefits for weight loss, mood
Some research suggests fasting is popular and may have benefits for an individual’s physical and mental health. In a 2020 online poll (N = 1,241), 24% of respondents said they had tried intermittent fasting, and 87% said the practice was very effective (50%) or somewhat effective (37%) in helping them lose weight.3 While more randomized control trials are needed to examine the practice’s effectiveness in promoting and maintaining weight loss, fasting has been linked to better glucose control in both humans and animals, and patients may have better adherence with fasting compared to caloric restriction alone.1 Improved mood, alertness, tranquility, and sometimes euphoria have been documented among individuals who fast, but these benefits may not be sustained.4 A prospective study of 462 participants who fasted during Ramadan found the practice reduced depression in patients with diabetes, possibly due to mindfulness, decreased inflammation from improved insulin sensitivity, and/or social cohesion.5
Be aware of the potential risks
Fasting may either improve or destabilize mood in people with bipolar disorder by disrupting circadian rhythm and sleep.2 Fasting might exacerbate underlying eating disorders.2 Increased dehydration escalates the risk for orthostatic hypotension, which might require discontinuing clozapine.6 Hypotension and toxicity might arise during lithium pharmacotherapy. The Table4 summarizes things to consider when caring for a patient who fasts while receiving pharmacotherapy.

Provide patients with guidance
Advise patients not to fast if you believe it might exacerbate their mental illness, and encourage them to discuss with their primary care physicians any potential worsening of physical illnesses.2 When caring for a patient who fasts for religious reasons, consider consulting with the patient’s religious leaders.2 If patients choose to fast, monitor them for mood destabilization and/or medication adverse effects. If possible, avoid altering drug treatment regimens during fasting, and carefully monitor whenever a pharmaceutical change is necessary. When appropriate, the use of long-acting injectable medications may minimize adverse effects while maintaining mood stability. Encourage patients who fast to ensure they remain hydrated and practice sleep hygiene while they fast.7
1. Dong TA, Sandesara PB, Dhindsa DS, et al. Intermittent fasting: a heart healthy dietary pattern? Am J Med. 2020;133(8):901-907.
2. Fond G, Macgregor A, Leboyer M, et al. Fasting in mood disorders: neurobiology and effectiveness. A review of the literature. Psychiatry Res. 2013;209(3):253-258.
3. Ballard J. Americans say this popular diet is effective and inexpensive. YouGov. February 24, 2020. Accessed January 6, 2022. https://today.yougov.com/topics/food/articles-reports/2020/02/24/most-effective-diet-intermittent-fasting-poll
4. Furqan Z, Awaad R, Kurdyak P, et al. Considerations for clinicians treating Muslim patients with psychiatric disorders during Ramadan. Lancet Psychiatry. 2019;6(7):556-557.
5. Al-Ozairi E, AlAwadhi MM, Al-Ozairi A, et al. A prospective study of the effect of fasting during the month of Ramadan on depression and diabetes distress in people with type 2 diabetes. Diabet Res Clin Pract. 2019;153:145-149.
6. Chehovich C, Demler TL, Leppien E. Impact of Ramadan fasting on medical and psychiatric health. Int Clin Psychopharmacol. 2019;34(6):317-322.
7. Farooq S, Nazar Z, Akhtar J, et al. Effect of fasting during Ramadan on serum lithium level and mental state in bipolar affective disorder. Int Clin Psychopharmacol. 2010;25(6):323-327.
1. Dong TA, Sandesara PB, Dhindsa DS, et al. Intermittent fasting: a heart healthy dietary pattern? Am J Med. 2020;133(8):901-907.
2. Fond G, Macgregor A, Leboyer M, et al. Fasting in mood disorders: neurobiology and effectiveness. A review of the literature. Psychiatry Res. 2013;209(3):253-258.
3. Ballard J. Americans say this popular diet is effective and inexpensive. YouGov. February 24, 2020. Accessed January 6, 2022. https://today.yougov.com/topics/food/articles-reports/2020/02/24/most-effective-diet-intermittent-fasting-poll
4. Furqan Z, Awaad R, Kurdyak P, et al. Considerations for clinicians treating Muslim patients with psychiatric disorders during Ramadan. Lancet Psychiatry. 2019;6(7):556-557.
5. Al-Ozairi E, AlAwadhi MM, Al-Ozairi A, et al. A prospective study of the effect of fasting during the month of Ramadan on depression and diabetes distress in people with type 2 diabetes. Diabet Res Clin Pract. 2019;153:145-149.
6. Chehovich C, Demler TL, Leppien E. Impact of Ramadan fasting on medical and psychiatric health. Int Clin Psychopharmacol. 2019;34(6):317-322.
7. Farooq S, Nazar Z, Akhtar J, et al. Effect of fasting during Ramadan on serum lithium level and mental state in bipolar affective disorder. Int Clin Psychopharmacol. 2010;25(6):323-327.
What’s in a Name? The Problematic Term “Provider”
Health care has been dramatically transformed and influenced by medical and technological advances, insurance companies, state and federal legislation, and medical ethics. Amid these changes, including crises such as the ongoing coronavirus pandemic, earning the trust of patients to care for their mental and physical health remains a priority and a privilege.
It is troubling that federal health care agencies, in addition to hospitals, clinics, pharmacies, insurance companies, and administrators, often use the term provider when referring to clinicians on the multidisciplinary health care treatment team, which has become the predominant model for health care delivery. The word provider does not originate in the health care arena but from the world of commerce and contains no reference to professionalism or therapeutic relationships.1 Therefore, it should be replaced with more appropriate terminology that acknowledges clinicians’ roles and expertise and values our unique relationship with patients.
Why Is Provider a Problem?
First, the origin of the term provider is deplorable. During its ascent to power in the 1930s, the Nazi Party promoted the devaluation and exclusion of Jews in German society, including the medical community. Due to its eugenics campaign, the Nazi Party first targeted pediatrics, a specialty in which nearly half of its practitioners were Jewish.2 Beginning with female pediatricians, all Jewish physicians were redesignated as Behandler (provider) instead of Arzt (doctor).2 This is the first documented demeaning of physicians as providers in modern history. Jewish doctors were soon restricted to treating only Jewish patients and were further persecuted during the Holocaust. Knowing this background, what health care organization would use a term once associated with Nazi ideology?3
Second, using provider changes the treatment relationship. The nomenclature shift in the United States also seems to have originated in political and legislative circles. Although the reasons for this shift are unclear, the terminology became more pervasive after the government first used the term provider in Title XIX of the 1965 Social Security Amendments that established Medicare and Medicaid. Paydarfar and Schwartz noted it was used “in the sense of a contractor being paid for delivering any health-related products and services.”4 Ironically, a 1967 medical student health organization grant proposal discussed the role of a patient advocate in facilitating communication between “health care provider and patient.”5 A journalist for the New York Times used the word to describe a 1970 New York Senate debate surrounding the sale of Medicaid bills to collection agencies, but it is unclear whether the senators themselves used the term.6 Provider was later used in the National Health Planning and Resource Development Act of 1974.7
Ultimately, the adaptation of this terminology led to medicine being thought of only as a business, a commoditization of care, and reinforced by referring to patients as consumers, clients, or customers.3 This terminology suggests that the clinician-patient relationship is a commercial transaction based on a market concept where patients are consumers to be serviced.1,8 Emphasis is placed on following algorithms and treating symptoms rather than patients.9 Despite a goal of minimizing cost, a mismatched referral to a provider may actually compromise patient safety and cost-effectiveness due to missed diagnoses or excessive diagnostic testing.10
In addition to government, other nonclinical entities (eg, insurance companies, advocacy groups) and some clinicians may prefer the generic term provider. Besides health care commoditization, reasons may include convenience, simplifying health care nomenclature, or removing distinctions among health care professionals to reduce costs and/or increase autonomy.
However, our value as health care professionals is not simply what we can “provide.”11 We seek to know patients as people, putting their needs ahead of ours.1 We serve as confidants and advocates and not merely providers of medications, tests, or procedures.11 This personalized nature of health care depends on trust and professionalism rather than dispassionate delivery of commoditized services.1 Using traditional terminology acknowledges the true nature of the treatment relationship—one that is established not on market concepts but on medical ethics of autonomy, justice, beneficence, and nonmaleficence.
Third, provider is inaccurate and potentially disrespectful and harmful. The word doctor is derived from Latin doctus or docere, meaning to teach or instruct—a valued function in our interactions with patients, families, students, and colleagues.12,13 In contrast, provider refers to commercial transactions or the provision of shelter, food, and love within families and communities.1,14
Although there are no studies assessing the impact of this terminology on individual clinicians, the term provider may have a negative impact on both individual clinicians and on the health care system. Health care professionals may feel they are being disrespected by being portrayed as dispensers of services rather than as individuals.13,15 Furthermore, provider does not acknowledge the specialized training and qualifications of multidisciplinary treatment team members. The historical and theoretical foundation, degrees awarded, and scopes of practice for physicians, physician assistants, nurse practitioners, dentists, psychologists, optometrists, physical therapists, or social workers are different yet valuable, and their expertise and accomplishment should be recognized.
The use of this term has potential for causing moral injury and reduced self-worth, sense of purpose, and meaning in our daily work; this could threaten satisfaction and commitment and lead to demoralization and burnout.1,16 It may impair effective team dynamics, as it makes no reference to professional values and may lead patients and clinicians to place lower value on professionalism and conduct.10 It may negatively impact primary care specialties by propagating the connotation that primary care is simple care and promoting low compensation, lagging recruitment, and diminished respect.10 Finally, it is detrimental to patients by changing the nature of the relationship and failing to evoke the compassion and support that sick people (that is, patients) need and deserve.3
Last, use of this term can mislead patients. By law, a health care provider is defined as “a doctor of medicine or osteopathy who is authorized to practice medicine or surgery… or any other person determined by the Secretary [of Labor] to be capable of providing health care services,” which includes podiatrists, dentists, clinical psychologists, optometrists, chiropractors, nurse practitioners, nurse-midwives, clinical social workers, and physician assistants.17
When clinicians are categorized as providers rather than by their degrees and roles/responsibilities, patients may assume that all team members have equal training, interchangeable skills, and uniform expertise and knowledge and may conclude they can receive the same level of care from anyone.8,10 Potential for confusion is increased by the nearly ubiquitous white laboratory coat in clinical settings and doctoral degrees attainable in different health care disciplines (eg, medicine, nursing, psychology, pharmacy, physical therapy). Patients deserve to know who does what on the team of professionals who care for them and may not be fully informed when requesting or receiving treatment if they are not provided important information, such as a clinician’s title, training, and scope of practice.8,16
Reversing the Trend
Increasing awareness among patients, their families, health professions students, and health care colleagues and administrators of the importance of traditional nomenclature is a first step in reversing this trend or mitigating its impact. If an overarching generic term is required, then health care professional, clinician, or practitioner are preferred.10,12 Fifteen years ago, the Southern California Permanente Medical Group prohibited the use of the word provider to describe physicians, and its editorial style deemed it cold and institutional.16 Many, but not all, state, regional, or national medical associations and journals avoid provider in their names or titles.
I am encouraged that this journal—drawing its audience from several government health care agencies—is named Federal Practitioner rather than Federal Provider. This is reasonable and accurate, as practitioner refers to the practice of a profession, usually associated with health care.
I hope other professions can resist this trend. Lawyers are not considered legal aid providers, and teachers are not called knowledge providers.3 We do not refer to airline pilots as air transportation providers or musicians as instrument-playing melody providers. Many veterans likely would be offended if they were referred to as Constitution support and defense providers rather than by the military branch-specific titles that they earned through dedication, training, and sacrifice. The individuals in these examples demonstrate commitment to representing clients, educating students, flying passengers, playing instruments, or ensuring national defense. As health care professionals, our commitment to treating patients is equally important.4
Language matters when it comes to people feeling respected and achieving their full potential.1 I encourage government health care agencies to stop referring to us as providers and resume using traditional nomenclature. This will demonstrate genuine respect for us, transparency for the patients we serve, and recognition that caring for the sick is a calling, not a commodity.
Dedication
The author dedicates this article to his father John E. Scarff, Jr, a physician and United States Army veteran.
1. Beasley JW, Roberts RG, Goroll AH. Promoting trust and morale by changing how the word provider is used: encouraging specificity and transparency. JAMA. 2021;325(23):2343-2344. doi:10.1001/jama.2021.6046
2. Saenger P. Jewish pediatricians in Nazi Germany: victims of persecution. Isr Med Assoc J. 2006;8(5):324-328.
3. Nasrallah HA. We are physicians, not providers, and we treat patients, not clients! Curr Psychiatr. 2020;19(2):5-7,29.
4. Paydarfar D, Schwartz WJ. A piece of my mind. Dear provider. JAMA. 2011;305(20):2046-2047. doi:10.1001/jama.2011.702
5. Student Health Organization. Grant Proposal of Student Health Organization. Summer Project of the South Bronx, 1967. Albert Einstein College of Medicine, unpublished.
6. Clines FX. Doctors face ban on sale of bills. New York Times. February 25, 1970:51
7. The National Health Planning and Resource Development Act of 1974. 42 USC § 300 (1975).
8. American Academy of Family Physicians. Provider, use of term (Position paper). Published 2018. Accessed September 22, 2021. https://www.aafp.org/about/policies/all/provider.html
9. Sanniec K, Gellis M. I am not a medical provider; I am a doctor. Aesthet Surg J. 2013;33(5):749-750. doi:10.1177/1090820X13487017
10. Goroll AH. Eliminating the term primary care “provider”: consequences of language for the future of primary care. JAMA. 2016;315(17):1833-1834. doi:10.1001/jama.2016.2329
11. Lee BY. Time to stop labeling physicians as providers. Published May 5, 2019. Accessed September 22, 2021. https://www.forbes.com/sites/brucelee/2019/05/05/time-to-stop-labeling-physicians-as-providers/?sh=7edfc865118e
12. Dhand S, Carbone WJ. Physicians are not providers: An open letter to the AMA and medical boards. Published November 30, 2015. Accessed September 22, 2021. https://www.kevinmd.com/blog/2015/11/physicians-are-not-providers-an-open-letter-to-the-ama-and-medical-boards.html
13. Al-Agba N. If you call me a provider, I will assume you are a Nazi. Published February 8, 2019. Accessed September 22, 2021. https://thedeductible.com/2019/02/08/if-you-call-me-a-provider-i-will-assume-you-are-a-nazi
14. Weiss JM. Physician or surgeon, but not “provider.” Published February 5, 2020. Accessed September 22, 2021. https://www.psychologytoday.com/us/blog/women-in-medicine/202002/physician-or-surgeon-not-provider
15. Liao L. Providers or professionals: how our conceptions of physician as machine or person lead to burnout. Med Teach. 2021;43(2):234-236. doi:10.1080/0142159X.2020.1769049
16. Weiss J. ‘Physician’ not ‘provider’ is better for doctor and patient. Published December 16, 2019. Accessed September 22, 2021. https://permanente.org/physician-not-provider-is-better-for-doctor-and-patient
17. Definition of Health Care Provider. 29 CFR § 825.125 (1993).
Health care has been dramatically transformed and influenced by medical and technological advances, insurance companies, state and federal legislation, and medical ethics. Amid these changes, including crises such as the ongoing coronavirus pandemic, earning the trust of patients to care for their mental and physical health remains a priority and a privilege.
It is troubling that federal health care agencies, in addition to hospitals, clinics, pharmacies, insurance companies, and administrators, often use the term provider when referring to clinicians on the multidisciplinary health care treatment team, which has become the predominant model for health care delivery. The word provider does not originate in the health care arena but from the world of commerce and contains no reference to professionalism or therapeutic relationships.1 Therefore, it should be replaced with more appropriate terminology that acknowledges clinicians’ roles and expertise and values our unique relationship with patients.
Why Is Provider a Problem?
First, the origin of the term provider is deplorable. During its ascent to power in the 1930s, the Nazi Party promoted the devaluation and exclusion of Jews in German society, including the medical community. Due to its eugenics campaign, the Nazi Party first targeted pediatrics, a specialty in which nearly half of its practitioners were Jewish.2 Beginning with female pediatricians, all Jewish physicians were redesignated as Behandler (provider) instead of Arzt (doctor).2 This is the first documented demeaning of physicians as providers in modern history. Jewish doctors were soon restricted to treating only Jewish patients and were further persecuted during the Holocaust. Knowing this background, what health care organization would use a term once associated with Nazi ideology?3
Second, using provider changes the treatment relationship. The nomenclature shift in the United States also seems to have originated in political and legislative circles. Although the reasons for this shift are unclear, the terminology became more pervasive after the government first used the term provider in Title XIX of the 1965 Social Security Amendments that established Medicare and Medicaid. Paydarfar and Schwartz noted it was used “in the sense of a contractor being paid for delivering any health-related products and services.”4 Ironically, a 1967 medical student health organization grant proposal discussed the role of a patient advocate in facilitating communication between “health care provider and patient.”5 A journalist for the New York Times used the word to describe a 1970 New York Senate debate surrounding the sale of Medicaid bills to collection agencies, but it is unclear whether the senators themselves used the term.6 Provider was later used in the National Health Planning and Resource Development Act of 1974.7
Ultimately, the adaptation of this terminology led to medicine being thought of only as a business, a commoditization of care, and reinforced by referring to patients as consumers, clients, or customers.3 This terminology suggests that the clinician-patient relationship is a commercial transaction based on a market concept where patients are consumers to be serviced.1,8 Emphasis is placed on following algorithms and treating symptoms rather than patients.9 Despite a goal of minimizing cost, a mismatched referral to a provider may actually compromise patient safety and cost-effectiveness due to missed diagnoses or excessive diagnostic testing.10
In addition to government, other nonclinical entities (eg, insurance companies, advocacy groups) and some clinicians may prefer the generic term provider. Besides health care commoditization, reasons may include convenience, simplifying health care nomenclature, or removing distinctions among health care professionals to reduce costs and/or increase autonomy.
However, our value as health care professionals is not simply what we can “provide.”11 We seek to know patients as people, putting their needs ahead of ours.1 We serve as confidants and advocates and not merely providers of medications, tests, or procedures.11 This personalized nature of health care depends on trust and professionalism rather than dispassionate delivery of commoditized services.1 Using traditional terminology acknowledges the true nature of the treatment relationship—one that is established not on market concepts but on medical ethics of autonomy, justice, beneficence, and nonmaleficence.
Third, provider is inaccurate and potentially disrespectful and harmful. The word doctor is derived from Latin doctus or docere, meaning to teach or instruct—a valued function in our interactions with patients, families, students, and colleagues.12,13 In contrast, provider refers to commercial transactions or the provision of shelter, food, and love within families and communities.1,14
Although there are no studies assessing the impact of this terminology on individual clinicians, the term provider may have a negative impact on both individual clinicians and on the health care system. Health care professionals may feel they are being disrespected by being portrayed as dispensers of services rather than as individuals.13,15 Furthermore, provider does not acknowledge the specialized training and qualifications of multidisciplinary treatment team members. The historical and theoretical foundation, degrees awarded, and scopes of practice for physicians, physician assistants, nurse practitioners, dentists, psychologists, optometrists, physical therapists, or social workers are different yet valuable, and their expertise and accomplishment should be recognized.
The use of this term has potential for causing moral injury and reduced self-worth, sense of purpose, and meaning in our daily work; this could threaten satisfaction and commitment and lead to demoralization and burnout.1,16 It may impair effective team dynamics, as it makes no reference to professional values and may lead patients and clinicians to place lower value on professionalism and conduct.10 It may negatively impact primary care specialties by propagating the connotation that primary care is simple care and promoting low compensation, lagging recruitment, and diminished respect.10 Finally, it is detrimental to patients by changing the nature of the relationship and failing to evoke the compassion and support that sick people (that is, patients) need and deserve.3
Last, use of this term can mislead patients. By law, a health care provider is defined as “a doctor of medicine or osteopathy who is authorized to practice medicine or surgery… or any other person determined by the Secretary [of Labor] to be capable of providing health care services,” which includes podiatrists, dentists, clinical psychologists, optometrists, chiropractors, nurse practitioners, nurse-midwives, clinical social workers, and physician assistants.17
When clinicians are categorized as providers rather than by their degrees and roles/responsibilities, patients may assume that all team members have equal training, interchangeable skills, and uniform expertise and knowledge and may conclude they can receive the same level of care from anyone.8,10 Potential for confusion is increased by the nearly ubiquitous white laboratory coat in clinical settings and doctoral degrees attainable in different health care disciplines (eg, medicine, nursing, psychology, pharmacy, physical therapy). Patients deserve to know who does what on the team of professionals who care for them and may not be fully informed when requesting or receiving treatment if they are not provided important information, such as a clinician’s title, training, and scope of practice.8,16
Reversing the Trend
Increasing awareness among patients, their families, health professions students, and health care colleagues and administrators of the importance of traditional nomenclature is a first step in reversing this trend or mitigating its impact. If an overarching generic term is required, then health care professional, clinician, or practitioner are preferred.10,12 Fifteen years ago, the Southern California Permanente Medical Group prohibited the use of the word provider to describe physicians, and its editorial style deemed it cold and institutional.16 Many, but not all, state, regional, or national medical associations and journals avoid provider in their names or titles.
I am encouraged that this journal—drawing its audience from several government health care agencies—is named Federal Practitioner rather than Federal Provider. This is reasonable and accurate, as practitioner refers to the practice of a profession, usually associated with health care.
I hope other professions can resist this trend. Lawyers are not considered legal aid providers, and teachers are not called knowledge providers.3 We do not refer to airline pilots as air transportation providers or musicians as instrument-playing melody providers. Many veterans likely would be offended if they were referred to as Constitution support and defense providers rather than by the military branch-specific titles that they earned through dedication, training, and sacrifice. The individuals in these examples demonstrate commitment to representing clients, educating students, flying passengers, playing instruments, or ensuring national defense. As health care professionals, our commitment to treating patients is equally important.4
Language matters when it comes to people feeling respected and achieving their full potential.1 I encourage government health care agencies to stop referring to us as providers and resume using traditional nomenclature. This will demonstrate genuine respect for us, transparency for the patients we serve, and recognition that caring for the sick is a calling, not a commodity.
Dedication
The author dedicates this article to his father John E. Scarff, Jr, a physician and United States Army veteran.
Health care has been dramatically transformed and influenced by medical and technological advances, insurance companies, state and federal legislation, and medical ethics. Amid these changes, including crises such as the ongoing coronavirus pandemic, earning the trust of patients to care for their mental and physical health remains a priority and a privilege.
It is troubling that federal health care agencies, in addition to hospitals, clinics, pharmacies, insurance companies, and administrators, often use the term provider when referring to clinicians on the multidisciplinary health care treatment team, which has become the predominant model for health care delivery. The word provider does not originate in the health care arena but from the world of commerce and contains no reference to professionalism or therapeutic relationships.1 Therefore, it should be replaced with more appropriate terminology that acknowledges clinicians’ roles and expertise and values our unique relationship with patients.
Why Is Provider a Problem?
First, the origin of the term provider is deplorable. During its ascent to power in the 1930s, the Nazi Party promoted the devaluation and exclusion of Jews in German society, including the medical community. Due to its eugenics campaign, the Nazi Party first targeted pediatrics, a specialty in which nearly half of its practitioners were Jewish.2 Beginning with female pediatricians, all Jewish physicians were redesignated as Behandler (provider) instead of Arzt (doctor).2 This is the first documented demeaning of physicians as providers in modern history. Jewish doctors were soon restricted to treating only Jewish patients and were further persecuted during the Holocaust. Knowing this background, what health care organization would use a term once associated with Nazi ideology?3
Second, using provider changes the treatment relationship. The nomenclature shift in the United States also seems to have originated in political and legislative circles. Although the reasons for this shift are unclear, the terminology became more pervasive after the government first used the term provider in Title XIX of the 1965 Social Security Amendments that established Medicare and Medicaid. Paydarfar and Schwartz noted it was used “in the sense of a contractor being paid for delivering any health-related products and services.”4 Ironically, a 1967 medical student health organization grant proposal discussed the role of a patient advocate in facilitating communication between “health care provider and patient.”5 A journalist for the New York Times used the word to describe a 1970 New York Senate debate surrounding the sale of Medicaid bills to collection agencies, but it is unclear whether the senators themselves used the term.6 Provider was later used in the National Health Planning and Resource Development Act of 1974.7
Ultimately, the adaptation of this terminology led to medicine being thought of only as a business, a commoditization of care, and reinforced by referring to patients as consumers, clients, or customers.3 This terminology suggests that the clinician-patient relationship is a commercial transaction based on a market concept where patients are consumers to be serviced.1,8 Emphasis is placed on following algorithms and treating symptoms rather than patients.9 Despite a goal of minimizing cost, a mismatched referral to a provider may actually compromise patient safety and cost-effectiveness due to missed diagnoses or excessive diagnostic testing.10
In addition to government, other nonclinical entities (eg, insurance companies, advocacy groups) and some clinicians may prefer the generic term provider. Besides health care commoditization, reasons may include convenience, simplifying health care nomenclature, or removing distinctions among health care professionals to reduce costs and/or increase autonomy.
However, our value as health care professionals is not simply what we can “provide.”11 We seek to know patients as people, putting their needs ahead of ours.1 We serve as confidants and advocates and not merely providers of medications, tests, or procedures.11 This personalized nature of health care depends on trust and professionalism rather than dispassionate delivery of commoditized services.1 Using traditional terminology acknowledges the true nature of the treatment relationship—one that is established not on market concepts but on medical ethics of autonomy, justice, beneficence, and nonmaleficence.
Third, provider is inaccurate and potentially disrespectful and harmful. The word doctor is derived from Latin doctus or docere, meaning to teach or instruct—a valued function in our interactions with patients, families, students, and colleagues.12,13 In contrast, provider refers to commercial transactions or the provision of shelter, food, and love within families and communities.1,14
Although there are no studies assessing the impact of this terminology on individual clinicians, the term provider may have a negative impact on both individual clinicians and on the health care system. Health care professionals may feel they are being disrespected by being portrayed as dispensers of services rather than as individuals.13,15 Furthermore, provider does not acknowledge the specialized training and qualifications of multidisciplinary treatment team members. The historical and theoretical foundation, degrees awarded, and scopes of practice for physicians, physician assistants, nurse practitioners, dentists, psychologists, optometrists, physical therapists, or social workers are different yet valuable, and their expertise and accomplishment should be recognized.
The use of this term has potential for causing moral injury and reduced self-worth, sense of purpose, and meaning in our daily work; this could threaten satisfaction and commitment and lead to demoralization and burnout.1,16 It may impair effective team dynamics, as it makes no reference to professional values and may lead patients and clinicians to place lower value on professionalism and conduct.10 It may negatively impact primary care specialties by propagating the connotation that primary care is simple care and promoting low compensation, lagging recruitment, and diminished respect.10 Finally, it is detrimental to patients by changing the nature of the relationship and failing to evoke the compassion and support that sick people (that is, patients) need and deserve.3
Last, use of this term can mislead patients. By law, a health care provider is defined as “a doctor of medicine or osteopathy who is authorized to practice medicine or surgery… or any other person determined by the Secretary [of Labor] to be capable of providing health care services,” which includes podiatrists, dentists, clinical psychologists, optometrists, chiropractors, nurse practitioners, nurse-midwives, clinical social workers, and physician assistants.17
When clinicians are categorized as providers rather than by their degrees and roles/responsibilities, patients may assume that all team members have equal training, interchangeable skills, and uniform expertise and knowledge and may conclude they can receive the same level of care from anyone.8,10 Potential for confusion is increased by the nearly ubiquitous white laboratory coat in clinical settings and doctoral degrees attainable in different health care disciplines (eg, medicine, nursing, psychology, pharmacy, physical therapy). Patients deserve to know who does what on the team of professionals who care for them and may not be fully informed when requesting or receiving treatment if they are not provided important information, such as a clinician’s title, training, and scope of practice.8,16
Reversing the Trend
Increasing awareness among patients, their families, health professions students, and health care colleagues and administrators of the importance of traditional nomenclature is a first step in reversing this trend or mitigating its impact. If an overarching generic term is required, then health care professional, clinician, or practitioner are preferred.10,12 Fifteen years ago, the Southern California Permanente Medical Group prohibited the use of the word provider to describe physicians, and its editorial style deemed it cold and institutional.16 Many, but not all, state, regional, or national medical associations and journals avoid provider in their names or titles.
I am encouraged that this journal—drawing its audience from several government health care agencies—is named Federal Practitioner rather than Federal Provider. This is reasonable and accurate, as practitioner refers to the practice of a profession, usually associated with health care.
I hope other professions can resist this trend. Lawyers are not considered legal aid providers, and teachers are not called knowledge providers.3 We do not refer to airline pilots as air transportation providers or musicians as instrument-playing melody providers. Many veterans likely would be offended if they were referred to as Constitution support and defense providers rather than by the military branch-specific titles that they earned through dedication, training, and sacrifice. The individuals in these examples demonstrate commitment to representing clients, educating students, flying passengers, playing instruments, or ensuring national defense. As health care professionals, our commitment to treating patients is equally important.4
Language matters when it comes to people feeling respected and achieving their full potential.1 I encourage government health care agencies to stop referring to us as providers and resume using traditional nomenclature. This will demonstrate genuine respect for us, transparency for the patients we serve, and recognition that caring for the sick is a calling, not a commodity.
Dedication
The author dedicates this article to his father John E. Scarff, Jr, a physician and United States Army veteran.
1. Beasley JW, Roberts RG, Goroll AH. Promoting trust and morale by changing how the word provider is used: encouraging specificity and transparency. JAMA. 2021;325(23):2343-2344. doi:10.1001/jama.2021.6046
2. Saenger P. Jewish pediatricians in Nazi Germany: victims of persecution. Isr Med Assoc J. 2006;8(5):324-328.
3. Nasrallah HA. We are physicians, not providers, and we treat patients, not clients! Curr Psychiatr. 2020;19(2):5-7,29.
4. Paydarfar D, Schwartz WJ. A piece of my mind. Dear provider. JAMA. 2011;305(20):2046-2047. doi:10.1001/jama.2011.702
5. Student Health Organization. Grant Proposal of Student Health Organization. Summer Project of the South Bronx, 1967. Albert Einstein College of Medicine, unpublished.
6. Clines FX. Doctors face ban on sale of bills. New York Times. February 25, 1970:51
7. The National Health Planning and Resource Development Act of 1974. 42 USC § 300 (1975).
8. American Academy of Family Physicians. Provider, use of term (Position paper). Published 2018. Accessed September 22, 2021. https://www.aafp.org/about/policies/all/provider.html
9. Sanniec K, Gellis M. I am not a medical provider; I am a doctor. Aesthet Surg J. 2013;33(5):749-750. doi:10.1177/1090820X13487017
10. Goroll AH. Eliminating the term primary care “provider”: consequences of language for the future of primary care. JAMA. 2016;315(17):1833-1834. doi:10.1001/jama.2016.2329
11. Lee BY. Time to stop labeling physicians as providers. Published May 5, 2019. Accessed September 22, 2021. https://www.forbes.com/sites/brucelee/2019/05/05/time-to-stop-labeling-physicians-as-providers/?sh=7edfc865118e
12. Dhand S, Carbone WJ. Physicians are not providers: An open letter to the AMA and medical boards. Published November 30, 2015. Accessed September 22, 2021. https://www.kevinmd.com/blog/2015/11/physicians-are-not-providers-an-open-letter-to-the-ama-and-medical-boards.html
13. Al-Agba N. If you call me a provider, I will assume you are a Nazi. Published February 8, 2019. Accessed September 22, 2021. https://thedeductible.com/2019/02/08/if-you-call-me-a-provider-i-will-assume-you-are-a-nazi
14. Weiss JM. Physician or surgeon, but not “provider.” Published February 5, 2020. Accessed September 22, 2021. https://www.psychologytoday.com/us/blog/women-in-medicine/202002/physician-or-surgeon-not-provider
15. Liao L. Providers or professionals: how our conceptions of physician as machine or person lead to burnout. Med Teach. 2021;43(2):234-236. doi:10.1080/0142159X.2020.1769049
16. Weiss J. ‘Physician’ not ‘provider’ is better for doctor and patient. Published December 16, 2019. Accessed September 22, 2021. https://permanente.org/physician-not-provider-is-better-for-doctor-and-patient
17. Definition of Health Care Provider. 29 CFR § 825.125 (1993).
1. Beasley JW, Roberts RG, Goroll AH. Promoting trust and morale by changing how the word provider is used: encouraging specificity and transparency. JAMA. 2021;325(23):2343-2344. doi:10.1001/jama.2021.6046
2. Saenger P. Jewish pediatricians in Nazi Germany: victims of persecution. Isr Med Assoc J. 2006;8(5):324-328.
3. Nasrallah HA. We are physicians, not providers, and we treat patients, not clients! Curr Psychiatr. 2020;19(2):5-7,29.
4. Paydarfar D, Schwartz WJ. A piece of my mind. Dear provider. JAMA. 2011;305(20):2046-2047. doi:10.1001/jama.2011.702
5. Student Health Organization. Grant Proposal of Student Health Organization. Summer Project of the South Bronx, 1967. Albert Einstein College of Medicine, unpublished.
6. Clines FX. Doctors face ban on sale of bills. New York Times. February 25, 1970:51
7. The National Health Planning and Resource Development Act of 1974. 42 USC § 300 (1975).
8. American Academy of Family Physicians. Provider, use of term (Position paper). Published 2018. Accessed September 22, 2021. https://www.aafp.org/about/policies/all/provider.html
9. Sanniec K, Gellis M. I am not a medical provider; I am a doctor. Aesthet Surg J. 2013;33(5):749-750. doi:10.1177/1090820X13487017
10. Goroll AH. Eliminating the term primary care “provider”: consequences of language for the future of primary care. JAMA. 2016;315(17):1833-1834. doi:10.1001/jama.2016.2329
11. Lee BY. Time to stop labeling physicians as providers. Published May 5, 2019. Accessed September 22, 2021. https://www.forbes.com/sites/brucelee/2019/05/05/time-to-stop-labeling-physicians-as-providers/?sh=7edfc865118e
12. Dhand S, Carbone WJ. Physicians are not providers: An open letter to the AMA and medical boards. Published November 30, 2015. Accessed September 22, 2021. https://www.kevinmd.com/blog/2015/11/physicians-are-not-providers-an-open-letter-to-the-ama-and-medical-boards.html
13. Al-Agba N. If you call me a provider, I will assume you are a Nazi. Published February 8, 2019. Accessed September 22, 2021. https://thedeductible.com/2019/02/08/if-you-call-me-a-provider-i-will-assume-you-are-a-nazi
14. Weiss JM. Physician or surgeon, but not “provider.” Published February 5, 2020. Accessed September 22, 2021. https://www.psychologytoday.com/us/blog/women-in-medicine/202002/physician-or-surgeon-not-provider
15. Liao L. Providers or professionals: how our conceptions of physician as machine or person lead to burnout. Med Teach. 2021;43(2):234-236. doi:10.1080/0142159X.2020.1769049
16. Weiss J. ‘Physician’ not ‘provider’ is better for doctor and patient. Published December 16, 2019. Accessed September 22, 2021. https://permanente.org/physician-not-provider-is-better-for-doctor-and-patient
17. Definition of Health Care Provider. 29 CFR § 825.125 (1993).
The ABCDs of treating tardive dyskinesia
Tardive dyskinesia (TD)—involuntary movement persisting for >1 month—is often caused by exposure to dopamine receptor–blocking agents such as antipsychotics.1 The pathophysiology of TD is attributed to dopamine receptor hypersensitivity and upregulation of dopamine receptors in response to chronic receptor blockade, although striatal dysfunction, oxidative stress, and gamma-aminobutyric acid (GABA) dysfunction may play a role.1 Because discontinuing the antipsychotic may not improve the patient’s TD symptoms and may worsen mood or psychosis, clinicians often prescribe adjunctive agents to reduce TD symptoms while continuing the antipsychotic. Clinicians can use the mnemonic ABCD to help recall 4 evidence-based treatments for TD.
Amantadine is an N-methyl-
Ginkgo Biloba contains antioxidant properties that may help reduce TD symptoms by alleviating oxidative stress. In a meta-analysis of 3 randomized controlled trials from China (N = 299), ginkgo biloba extract, 240 mg/d, significantly improved symptoms of TD compared with placebo.3 Ginkgo biloba has an antiplatelet effect and therefore should not be used in patients with an increased bleeding risk.
Clonazepam. Several small studies have examined the use of this GABA agonist for TD. In a study of 19 patients with TD, researchers found a symptom reduction of up to 35% with doses up to 4.5 mg/d.4 However, many studies have had small sample sizes or poor methodology. A 2018 Cochrane review recommended using other agents before considering clonazepam for TD because this medication has uncertain efficacy in treating TD, and it can cause sedation and dependence.5
Deutetrabenazine and valbenazine, the only FDA-approved treatments for TD, are vesicular monoamine transporter 2 (VMAT2) inhibitors, which inhibit dopamine release and decrease dopamine receptor hypersensitivity.6 In a 12-week, randomized, double-blind, placebo-controlled study of 117 patients with moderate-to-severe TD, those who received deutetrabenazine (up to 48 mg/d) had a significant mean reduction in AIMS score (3 points) compared with placebo.7 In the 1-year KINECT 3 study, 124 patients with TD who received valbenazine, 40 or 80 mg/d, had significant mean reductions in AIMS scores of 3.0 and 4.8 points, respectively.8 Adverse effects of these medications include somnolence, headache, akathisia, urinary tract infection, worsening mood, and suicidality. Tetrabenazine is another VMAT2 inhibitor that may be effective in doses up to 150 mg/d, but its off-label use is limited by the need for frequent dosing and a risk for suicidality.6
Other adjunctive treatments, such as vitamin B6, vitamin E, zonisamide, and levetiracetam, might offer some benefit in TD.6 However, further evidence is needed to support including these interventions in treatment guidelines.
1. Elkurd MT, Bahroo L. Keeping up with the clinical advances: tardive dyskinesia. CNS Spectr. 2019;24(suppl 1):70-81.
2. Pappa S, Tsouli S, Apostolu G, et al. Effects of amantadine on tardive dyskinesia: a randomized, double-blind, placebo-controlled study. Clin Neuropharmacol. 2010;33(6):271-275.
3. Zheng W, Xiang YQ, Ng CH, et al. Extract of ginkgo biloba for tardive dyskinesia: meta-analysis of randomized controlled trials. Pharmacopsychiatry. 2016;49(3):107-111.
4. Thaker GK, Nguyen JA, Strauss ME, et al. Clonazepam treatment of tardive dyskinesia: a practical GABAmimetic strategy. Am J Psychiatry. 1990;147(4):445-451.
5. Bergman H, Bhoopathi PS, Soares-Weiser K. Benzodiazepines for antipsychotic-induced tardive dyskinesia. Cochrane Database Syst Rev. 2018;1:CD000205.
6. Sreeram V, Shagufta S, Kagadkar F. Role of vesicular monoamine transporter 2 inhibitors in tardive dyskinesia management. Cureus. 2019;11(8):e5471. doi: 10.7759/cureus.5471.
7. Fernandez HH, Factor SA, Hauser RA. Randomized controlled trial of deutetrabenazine for tardive dyskinesia. The ARM-TD study. Neurology. 2017;88(21):2003-2010.
8. Factor SA, Remington G, Comella CL, et al. The effects of valbenazine in participants with tardive dyskinesia: results of the 1-year KINECT 3 extension study. J Clin Psychiatry. 2017;78(9):1344-1350.
Tardive dyskinesia (TD)—involuntary movement persisting for >1 month—is often caused by exposure to dopamine receptor–blocking agents such as antipsychotics.1 The pathophysiology of TD is attributed to dopamine receptor hypersensitivity and upregulation of dopamine receptors in response to chronic receptor blockade, although striatal dysfunction, oxidative stress, and gamma-aminobutyric acid (GABA) dysfunction may play a role.1 Because discontinuing the antipsychotic may not improve the patient’s TD symptoms and may worsen mood or psychosis, clinicians often prescribe adjunctive agents to reduce TD symptoms while continuing the antipsychotic. Clinicians can use the mnemonic ABCD to help recall 4 evidence-based treatments for TD.
Amantadine is an N-methyl-
Ginkgo Biloba contains antioxidant properties that may help reduce TD symptoms by alleviating oxidative stress. In a meta-analysis of 3 randomized controlled trials from China (N = 299), ginkgo biloba extract, 240 mg/d, significantly improved symptoms of TD compared with placebo.3 Ginkgo biloba has an antiplatelet effect and therefore should not be used in patients with an increased bleeding risk.
Clonazepam. Several small studies have examined the use of this GABA agonist for TD. In a study of 19 patients with TD, researchers found a symptom reduction of up to 35% with doses up to 4.5 mg/d.4 However, many studies have had small sample sizes or poor methodology. A 2018 Cochrane review recommended using other agents before considering clonazepam for TD because this medication has uncertain efficacy in treating TD, and it can cause sedation and dependence.5
Deutetrabenazine and valbenazine, the only FDA-approved treatments for TD, are vesicular monoamine transporter 2 (VMAT2) inhibitors, which inhibit dopamine release and decrease dopamine receptor hypersensitivity.6 In a 12-week, randomized, double-blind, placebo-controlled study of 117 patients with moderate-to-severe TD, those who received deutetrabenazine (up to 48 mg/d) had a significant mean reduction in AIMS score (3 points) compared with placebo.7 In the 1-year KINECT 3 study, 124 patients with TD who received valbenazine, 40 or 80 mg/d, had significant mean reductions in AIMS scores of 3.0 and 4.8 points, respectively.8 Adverse effects of these medications include somnolence, headache, akathisia, urinary tract infection, worsening mood, and suicidality. Tetrabenazine is another VMAT2 inhibitor that may be effective in doses up to 150 mg/d, but its off-label use is limited by the need for frequent dosing and a risk for suicidality.6
Other adjunctive treatments, such as vitamin B6, vitamin E, zonisamide, and levetiracetam, might offer some benefit in TD.6 However, further evidence is needed to support including these interventions in treatment guidelines.
Tardive dyskinesia (TD)—involuntary movement persisting for >1 month—is often caused by exposure to dopamine receptor–blocking agents such as antipsychotics.1 The pathophysiology of TD is attributed to dopamine receptor hypersensitivity and upregulation of dopamine receptors in response to chronic receptor blockade, although striatal dysfunction, oxidative stress, and gamma-aminobutyric acid (GABA) dysfunction may play a role.1 Because discontinuing the antipsychotic may not improve the patient’s TD symptoms and may worsen mood or psychosis, clinicians often prescribe adjunctive agents to reduce TD symptoms while continuing the antipsychotic. Clinicians can use the mnemonic ABCD to help recall 4 evidence-based treatments for TD.
Amantadine is an N-methyl-
Ginkgo Biloba contains antioxidant properties that may help reduce TD symptoms by alleviating oxidative stress. In a meta-analysis of 3 randomized controlled trials from China (N = 299), ginkgo biloba extract, 240 mg/d, significantly improved symptoms of TD compared with placebo.3 Ginkgo biloba has an antiplatelet effect and therefore should not be used in patients with an increased bleeding risk.
Clonazepam. Several small studies have examined the use of this GABA agonist for TD. In a study of 19 patients with TD, researchers found a symptom reduction of up to 35% with doses up to 4.5 mg/d.4 However, many studies have had small sample sizes or poor methodology. A 2018 Cochrane review recommended using other agents before considering clonazepam for TD because this medication has uncertain efficacy in treating TD, and it can cause sedation and dependence.5
Deutetrabenazine and valbenazine, the only FDA-approved treatments for TD, are vesicular monoamine transporter 2 (VMAT2) inhibitors, which inhibit dopamine release and decrease dopamine receptor hypersensitivity.6 In a 12-week, randomized, double-blind, placebo-controlled study of 117 patients with moderate-to-severe TD, those who received deutetrabenazine (up to 48 mg/d) had a significant mean reduction in AIMS score (3 points) compared with placebo.7 In the 1-year KINECT 3 study, 124 patients with TD who received valbenazine, 40 or 80 mg/d, had significant mean reductions in AIMS scores of 3.0 and 4.8 points, respectively.8 Adverse effects of these medications include somnolence, headache, akathisia, urinary tract infection, worsening mood, and suicidality. Tetrabenazine is another VMAT2 inhibitor that may be effective in doses up to 150 mg/d, but its off-label use is limited by the need for frequent dosing and a risk for suicidality.6
Other adjunctive treatments, such as vitamin B6, vitamin E, zonisamide, and levetiracetam, might offer some benefit in TD.6 However, further evidence is needed to support including these interventions in treatment guidelines.
1. Elkurd MT, Bahroo L. Keeping up with the clinical advances: tardive dyskinesia. CNS Spectr. 2019;24(suppl 1):70-81.
2. Pappa S, Tsouli S, Apostolu G, et al. Effects of amantadine on tardive dyskinesia: a randomized, double-blind, placebo-controlled study. Clin Neuropharmacol. 2010;33(6):271-275.
3. Zheng W, Xiang YQ, Ng CH, et al. Extract of ginkgo biloba for tardive dyskinesia: meta-analysis of randomized controlled trials. Pharmacopsychiatry. 2016;49(3):107-111.
4. Thaker GK, Nguyen JA, Strauss ME, et al. Clonazepam treatment of tardive dyskinesia: a practical GABAmimetic strategy. Am J Psychiatry. 1990;147(4):445-451.
5. Bergman H, Bhoopathi PS, Soares-Weiser K. Benzodiazepines for antipsychotic-induced tardive dyskinesia. Cochrane Database Syst Rev. 2018;1:CD000205.
6. Sreeram V, Shagufta S, Kagadkar F. Role of vesicular monoamine transporter 2 inhibitors in tardive dyskinesia management. Cureus. 2019;11(8):e5471. doi: 10.7759/cureus.5471.
7. Fernandez HH, Factor SA, Hauser RA. Randomized controlled trial of deutetrabenazine for tardive dyskinesia. The ARM-TD study. Neurology. 2017;88(21):2003-2010.
8. Factor SA, Remington G, Comella CL, et al. The effects of valbenazine in participants with tardive dyskinesia: results of the 1-year KINECT 3 extension study. J Clin Psychiatry. 2017;78(9):1344-1350.
1. Elkurd MT, Bahroo L. Keeping up with the clinical advances: tardive dyskinesia. CNS Spectr. 2019;24(suppl 1):70-81.
2. Pappa S, Tsouli S, Apostolu G, et al. Effects of amantadine on tardive dyskinesia: a randomized, double-blind, placebo-controlled study. Clin Neuropharmacol. 2010;33(6):271-275.
3. Zheng W, Xiang YQ, Ng CH, et al. Extract of ginkgo biloba for tardive dyskinesia: meta-analysis of randomized controlled trials. Pharmacopsychiatry. 2016;49(3):107-111.
4. Thaker GK, Nguyen JA, Strauss ME, et al. Clonazepam treatment of tardive dyskinesia: a practical GABAmimetic strategy. Am J Psychiatry. 1990;147(4):445-451.
5. Bergman H, Bhoopathi PS, Soares-Weiser K. Benzodiazepines for antipsychotic-induced tardive dyskinesia. Cochrane Database Syst Rev. 2018;1:CD000205.
6. Sreeram V, Shagufta S, Kagadkar F. Role of vesicular monoamine transporter 2 inhibitors in tardive dyskinesia management. Cureus. 2019;11(8):e5471. doi: 10.7759/cureus.5471.
7. Fernandez HH, Factor SA, Hauser RA. Randomized controlled trial of deutetrabenazine for tardive dyskinesia. The ARM-TD study. Neurology. 2017;88(21):2003-2010.
8. Factor SA, Remington G, Comella CL, et al. The effects of valbenazine in participants with tardive dyskinesia: results of the 1-year KINECT 3 extension study. J Clin Psychiatry. 2017;78(9):1344-1350.
Orthorexia Nervosa: An Obsession With Healthy Eating
First named by Steven Bratman in 1997, orthorexia nervosa (ON) from the Greek ortho, meaning correct, and orexi, meaning appetite, is classified as an unspecified feeding and eating disorder in the Diagnostic and Statistical Manual of Mental Disorders 5th edition (DSM-5).1,2
Hypothetical Case
Mr. P is a 30-year-old male who presented to the mental health clinic with his wife. The patient recounted that he had wanted to “be healthy” since childhood and has focused on exercise and proper diet, but anxiety about diet and food intake have steadily increased. Two years ago, he adopted a vegetarian diet by progressively eliminating several foods and food groups from his diet. He now feels “proud” to eat certain organically grown fruits, vegetables, nuts, beans, and drink only fruit or vegetable juice.
His wife stated that he spent between 3 and 5 hours daily preparing food or talking to friends and family about “correct foods to eat.” He also believed that errors in dietary habits caused physical or mental illnesses. He reported significant guilt and shame whenever he “slips up” on his dietary regimen and eats anything containing seafood, beef, or pork products, which he corrects by a day of fasting. His wife was frustrated because he refused to go to restaurants and started declining offers from friends to eat dinner at their homes unless he could bring his prepared food. He describes feeling “annoyed” when he sees other people eating fast food or meat.
Mr. P reported no significant medical or surgical history. His family history was significant for anxiety in his mother. He used to drink alcohol socially but ceased a few years ago due to its carbohydrate content. He never smoked or used illicit drugs.
A mental status exam revealed a thin male who appeared his stated age. He was cooperative, casually dressed, and made fair eye contact. He spoke clearly with an anxious tone and appropriate rate and volume. His affect was congruent with stated anxious mood. He was alert, awake, and oriented to person, place, and time. He reported no paranoia, auditory or visual hallucinations, and suicidal or homicidal ideation.
A physical exam revealed a thin male in no distress who measured 5 feet 10 inches tall and weighed 145 pounds, which yielded a body mass index of 20.8. His vitals included temperature of 98° F, blood pressure 115/76, pulse 74, and oxygen saturation 98% on room air. The remaining physical examination revealed no abnormalities. A complete blood count, thyroid function, urinalysis, and urine drug screens were within normal limits. Comprehensive metabolic profile revealed decreased sodium of 130 meq/L. Electrocardiogram revealed bradycardia.
An ON diagnosis is made primarily through a clinical interview. Collateral information from individuals familiar with the patient can be helpful. Experts have proposed and recently revised criteria for ON (Table). Although the ORTO-15 assessment tool may assist with diagnosis, the tool does not substitute for the clinical interview.
Discussion
There is no reliable measure of prevalence of ON, though Varga and colleagues initially estimated ON to occur in 6.9% of the general population, and ON may occur more frequently in health care professionals and performance artists.3 However, these may be overestimates, as the assessment tool used in the study does not adequately separate people with healthy eating habits from those with ON.4,5
Most prevalence studies were conducted in Europe and Turkey, and prevalence of ON may differ in the U.S. population. A recent assessment determined a prevalence of about 1%, similar to that of other eating disorders.5 No study has reported a correlation between ON and gender, but a survey of 448 college students in the U.S. (mean age 22 years) reported highest ON tendencies in Hispanic/Latino and overweight/obese students.6
Relationship to Other Illnesses
There is significant debate whether ON is a single syndrome, a variance of other syndromes, or a behavioral and culturally influenced attitude.7,8 Although ON may lead to or be comorbid with anorexia nervosa (AN) or obsessive-compulsive disorder (OCD), subtle differences exist between ON and these conditions.
To meet DSM-5 diagnostic criteria for AN, patients must weigh below minimally normal weight for their height and age, have an intense fear of gaining weight or becoming fat, and have a disturbed experience of their weight or body shape or cannot recognize the severity of the low weight.2 In contrast, an individual with ON may possess normal or low-normal weight. Patients with AN focus on food quantity, while patients with ON tend to focus on food quality. As summarized by Bratman, “People are ashamed of their anorexia, but they actively evangelize their orthorexia. People with anorexia skip meals; people with orthorexia do not (unless they are fasting). Those with anorexia focus only on avoiding foods, while those with orthorexia both avoid foods they think are bad and embrace foods they think are super-healthy.”9
Similarities between ON and OCD include anxiety, a need to exert control, and perfectionism. However, patients with OCD tend to report distress from compulsive behavior and a desire to change, thus exhibiting insight into their illness.8,10 Similarities between obsessive-compulsive personality disorder (OCPD) and ON include perfectionism, rigid thinking, excessive devotion, hypermorality, and a preoccupation with details and perceived rules.11
While no studies have yet described ON as a feature of somatoform disorders, some experts have hypothesized that preoccupation with illness in a patient with somatization disorder may engender a preoccupation with food and diet as a way to combat either real or perceived illness.11 Finally, there is a report of ON associated with the prodromal phase of schizophrenia, and the development of ON may increase risk for future psychotic disorders.11,12
Pathophysiology
The exact cause of ON is unknown, though it is likely multifactorial. Individuals with ON have neurocognitive deficits similar to those seen in patients with AN and OCD, including impairments in set-shifting (flexible problem solving), external attention, and working memory.11,13 Given these cognitive deficits as well as similar symptomatology, there may be analogous brain dysfunction in patients with ON and AN or OCD. Neuroimaging studies of patients with AN have revealed dysregulation of dopamine transmission in the reward circuitry of the ventral striatum and the food regulatory mechanism in the hypothalamus.14
Dysmorphology of and dysfunction in neural circuitry, particularly the cortico-striato-thalamo-cortical pathway, have been implicated in OCD.15 Neuroimaging studies have revealed increased volume and activation of the orbitofrontal cortex, which may be associated with obsessions and difficulty with extinction recall.14,15 In contrast, decreased volume and activity of the thalamus may impair its ability to inhibit the orbitofrontal cortex.15,16 Decreased volume and activity of the cingulate gyrus may be associated with difficulty in error monitoring and fear conditioning, while overactivation of the parietal lobe and cerebellum may be associated with compulsive behaviors.15,16
Risk Factors
Factors that contribute to the development of AN and possibly ON include development of food preferences, inherited differences in taste perception, food neophobia or pickiness, being premorbidly overweight or obese, parental feeding practices, and a history of parental eating disorders.14 One survey associated orthorexic tendencies with perfectionism, appearance orientation, overweight preoccupation, self-classified weight, and fearful and dismissing attachment styles.17 Significant predictors of ON included overweight preoccupation, appearance orientation, and a history of an eating disorder.17
Treatment
In contrast to patients with AN, patients with ON may be easily amenable to treatment, given their pursuit of and emphasis on wellness.18 Experts recommend a multidisciplinary team approach that includes physicians, psychotherapists, and dieticians.11 Treatment may be undertaken in an outpatient setting, but hospitalization for refeeding is recommended in cases with significant weight loss or malnourishment.11 Physical examination and laboratory studies are warranted, as excessive dietary restrictions can lead to weight loss and medical complications similar to those seen in AN, including osteopenia, anemia, hyponatremia, pancytopenia, bradycardia, and even pneumothorax and pneumomediastinum.19-21
There are no reported studies exploring the efficacy of psychotherapy or psychotropic medications for patients with ON. However, several treatments have been proposed given the symptom overlap with AN. Serotonin reuptake inhibitors may be beneficial for anxiety and obsessive-compulsive traits.18 However, patients with ON may refuse medications as unnatural substances.18
Cognitive behavioral therapy may be beneficial to address perfectionism and cognitive distortions, and exposure and response prevention may reduce obsessive-compulsive behaviors.11 Relaxation therapy may reduce mealtime anxiety. Psychoeducation may correct inaccurate beliefs about food groups, purity, and preparation, but it may induce emotional stress for the patient with ON.11
Conclusion
Orthorexia nervosa is perhaps best summarized as an obsession with healthy eating with associated restrictive behaviors. However, the attempt to attain optimum health through attention to diet may lead to malnourishment, loss of relationships, and poor quality of life.11 It is a little-understood disorder with uncertain etiology, imprecise assessment tools, and no formal diagnostic criteria or classification. Orthorexic characteristics vary from normal to pathologic in degree, and making a diagnosis remains a clinical judgment.22 Further research is needed to develop valid diagnostic tools and determining whether ON should be classified as a unique illness or a variation of other eating or anxiety disorders. Further research also may identify the etiology of ON, thus enabling targeted multidisciplinary treatment.
1. Bratman S. Health food junkie. Yoga J. 1997;136:42-50.
2. American Psychiatric Association. Feeding and eating disorders. In: Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington, VA: American Psychiatric Publishing; 2013:329-354.
3. Varga M, Dukay-Szabó S, Túry F, van Furth EF. Evidence and gaps in the literature on orthorexia nervosa. Eat Weight Disord. 2013;18(2):103-111.
4. Donini LM, Marsili D, Graziani MP, Imbriale M, Cannella C. Orthorexia nervosa: validation of a diagnosis questionnaire. Eat Weight Disord. 2005;10(2):e28-e32.
5. Dunn TM, Gibbs J, Whitney N, Starosta A. Prevalence of orthorexia nervosa is less than 1 %: data from a US sample. Eat Weight Disord. 2016;22(1):185-192.
6. Bundros J, Clifford D, Silliman K, Neyman Morris M. Prevalence of orthorexia nervosa among college students based on Bratman’s test and associated tendencies. Appetite. 2016;101:86-94.
7. Vandereycken W. Media hype, diagnostic fad or genuine disorder? Professionals’ opinions about night eating syndrome, orthorexia, muscle dysmorphia, and emetophobia. Eat Disord. 2011;19(2):145-155.
8. Dell’Osso L, Abelli M, Carpita B, et al. Historical evolution of the concept of anorexia nervosa and relationships with orthorexia nervosa, autism, and obsessive-compulsive spectrum. Neuropsychiatr Dis Treat. 2016;12:1651-1660.
9. Bratman S. Orthorexia: an update. http://www.orthorexia.com/orthorexia-an-update. Updated October 5, 2015. Accessed April 18, 2017.
10. Dunn TM, Bratman S. On orthorexia nervosa: a review of the literature and proposed diagnostic criteria. Eat Behav. 2016;21:11-17.
11. Koven NS, Abry AW. The clinical basis of orthorexia nervosa: emerging perspectives. Neuropsychiatr Dis Treat. 2015;11:385-394.
12. Saddichha S, Babu GN, Chandra P. Orthorexia nervosa presenting as prodrome of schizophrenia. Schizophr Res. 2012;134(1):110.
13. Koven NS, Senbonmatsu R. A neuropsychological evaluation of orthorexia nervosa. Open J Psychiatry. 2013;3(2):214-222.
14. Gorwood P, Blanchet-Collet C, Chartrel N, et al. New insights in anorexia nervosa. Front Neurosci. 2016;10:256.
15. Milad MR, Rauch SL. Obsessive-compulsive disorder: beyond segregated cortico-striatal pathways. Trends Cogn Sci. 2012;16(1):43-51.
16. Tang W, Zhu Q, Gong X, Zhu C, Wang Y, Chen S. Cortico-striato-thalamo-cortical circuit abnormalities in obsessive-compulsive disorder: A voxel-based morphometric and fMRI study of the whole brain. Behav Brain Res. 2016;313:17-22.
17. Barnes MA, Caltabiano ML. The interrelationship between orthorexia nervosa, perfectionism, body image and attachment style. Eat Weight Disord. 2017;22(1):177-184.
18. Mathieu J. What is orthorexia? J Am Diet Assoc. 2005;105(10):1510-1512.
19. Catalina Zamora ML, Bote Bonaechea B, García Sánchez F, Ríos Rial B. Orthorexia nervosa. A new eating behavior disorder? [in Spanish]. Actas Esp Psiquiatr. 2005;33(1):66-68.
20. Moroze RM, Dunn TM, Craig Holland J, Yager J, Weintraub P. Microthinking about micronutrients: a case of transition from obsessions about healthy eating to near-fatal “orthorexia nervosa” and proposed diagnostic criteria. Psychosomatics. 2015;56(4):397-403.
21. Park SW, Kim JY, Go GJ, Jeon ES, Pyo HJ, Kwon YJ. Orthorexia nervosa with hyponatremia, subcutaneous emphysema, pneumomediastimum, pneumothorax, and pancytopenia. Electrolyte Blood Press. 2011;9(1):32-37.
22. Mogallapu RNG, Aynampudi AR, Scarff JR, Lippmann S. Orthorexia nervosa. The Kentucky Psychiatrist. 2012;22(3):3-6.
First named by Steven Bratman in 1997, orthorexia nervosa (ON) from the Greek ortho, meaning correct, and orexi, meaning appetite, is classified as an unspecified feeding and eating disorder in the Diagnostic and Statistical Manual of Mental Disorders 5th edition (DSM-5).1,2
Hypothetical Case
Mr. P is a 30-year-old male who presented to the mental health clinic with his wife. The patient recounted that he had wanted to “be healthy” since childhood and has focused on exercise and proper diet, but anxiety about diet and food intake have steadily increased. Two years ago, he adopted a vegetarian diet by progressively eliminating several foods and food groups from his diet. He now feels “proud” to eat certain organically grown fruits, vegetables, nuts, beans, and drink only fruit or vegetable juice.
His wife stated that he spent between 3 and 5 hours daily preparing food or talking to friends and family about “correct foods to eat.” He also believed that errors in dietary habits caused physical or mental illnesses. He reported significant guilt and shame whenever he “slips up” on his dietary regimen and eats anything containing seafood, beef, or pork products, which he corrects by a day of fasting. His wife was frustrated because he refused to go to restaurants and started declining offers from friends to eat dinner at their homes unless he could bring his prepared food. He describes feeling “annoyed” when he sees other people eating fast food or meat.
Mr. P reported no significant medical or surgical history. His family history was significant for anxiety in his mother. He used to drink alcohol socially but ceased a few years ago due to its carbohydrate content. He never smoked or used illicit drugs.
A mental status exam revealed a thin male who appeared his stated age. He was cooperative, casually dressed, and made fair eye contact. He spoke clearly with an anxious tone and appropriate rate and volume. His affect was congruent with stated anxious mood. He was alert, awake, and oriented to person, place, and time. He reported no paranoia, auditory or visual hallucinations, and suicidal or homicidal ideation.
A physical exam revealed a thin male in no distress who measured 5 feet 10 inches tall and weighed 145 pounds, which yielded a body mass index of 20.8. His vitals included temperature of 98° F, blood pressure 115/76, pulse 74, and oxygen saturation 98% on room air. The remaining physical examination revealed no abnormalities. A complete blood count, thyroid function, urinalysis, and urine drug screens were within normal limits. Comprehensive metabolic profile revealed decreased sodium of 130 meq/L. Electrocardiogram revealed bradycardia.
An ON diagnosis is made primarily through a clinical interview. Collateral information from individuals familiar with the patient can be helpful. Experts have proposed and recently revised criteria for ON (Table). Although the ORTO-15 assessment tool may assist with diagnosis, the tool does not substitute for the clinical interview.
Discussion
There is no reliable measure of prevalence of ON, though Varga and colleagues initially estimated ON to occur in 6.9% of the general population, and ON may occur more frequently in health care professionals and performance artists.3 However, these may be overestimates, as the assessment tool used in the study does not adequately separate people with healthy eating habits from those with ON.4,5
Most prevalence studies were conducted in Europe and Turkey, and prevalence of ON may differ in the U.S. population. A recent assessment determined a prevalence of about 1%, similar to that of other eating disorders.5 No study has reported a correlation between ON and gender, but a survey of 448 college students in the U.S. (mean age 22 years) reported highest ON tendencies in Hispanic/Latino and overweight/obese students.6
Relationship to Other Illnesses
There is significant debate whether ON is a single syndrome, a variance of other syndromes, or a behavioral and culturally influenced attitude.7,8 Although ON may lead to or be comorbid with anorexia nervosa (AN) or obsessive-compulsive disorder (OCD), subtle differences exist between ON and these conditions.
To meet DSM-5 diagnostic criteria for AN, patients must weigh below minimally normal weight for their height and age, have an intense fear of gaining weight or becoming fat, and have a disturbed experience of their weight or body shape or cannot recognize the severity of the low weight.2 In contrast, an individual with ON may possess normal or low-normal weight. Patients with AN focus on food quantity, while patients with ON tend to focus on food quality. As summarized by Bratman, “People are ashamed of their anorexia, but they actively evangelize their orthorexia. People with anorexia skip meals; people with orthorexia do not (unless they are fasting). Those with anorexia focus only on avoiding foods, while those with orthorexia both avoid foods they think are bad and embrace foods they think are super-healthy.”9
Similarities between ON and OCD include anxiety, a need to exert control, and perfectionism. However, patients with OCD tend to report distress from compulsive behavior and a desire to change, thus exhibiting insight into their illness.8,10 Similarities between obsessive-compulsive personality disorder (OCPD) and ON include perfectionism, rigid thinking, excessive devotion, hypermorality, and a preoccupation with details and perceived rules.11
While no studies have yet described ON as a feature of somatoform disorders, some experts have hypothesized that preoccupation with illness in a patient with somatization disorder may engender a preoccupation with food and diet as a way to combat either real or perceived illness.11 Finally, there is a report of ON associated with the prodromal phase of schizophrenia, and the development of ON may increase risk for future psychotic disorders.11,12
Pathophysiology
The exact cause of ON is unknown, though it is likely multifactorial. Individuals with ON have neurocognitive deficits similar to those seen in patients with AN and OCD, including impairments in set-shifting (flexible problem solving), external attention, and working memory.11,13 Given these cognitive deficits as well as similar symptomatology, there may be analogous brain dysfunction in patients with ON and AN or OCD. Neuroimaging studies of patients with AN have revealed dysregulation of dopamine transmission in the reward circuitry of the ventral striatum and the food regulatory mechanism in the hypothalamus.14
Dysmorphology of and dysfunction in neural circuitry, particularly the cortico-striato-thalamo-cortical pathway, have been implicated in OCD.15 Neuroimaging studies have revealed increased volume and activation of the orbitofrontal cortex, which may be associated with obsessions and difficulty with extinction recall.14,15 In contrast, decreased volume and activity of the thalamus may impair its ability to inhibit the orbitofrontal cortex.15,16 Decreased volume and activity of the cingulate gyrus may be associated with difficulty in error monitoring and fear conditioning, while overactivation of the parietal lobe and cerebellum may be associated with compulsive behaviors.15,16
Risk Factors
Factors that contribute to the development of AN and possibly ON include development of food preferences, inherited differences in taste perception, food neophobia or pickiness, being premorbidly overweight or obese, parental feeding practices, and a history of parental eating disorders.14 One survey associated orthorexic tendencies with perfectionism, appearance orientation, overweight preoccupation, self-classified weight, and fearful and dismissing attachment styles.17 Significant predictors of ON included overweight preoccupation, appearance orientation, and a history of an eating disorder.17
Treatment
In contrast to patients with AN, patients with ON may be easily amenable to treatment, given their pursuit of and emphasis on wellness.18 Experts recommend a multidisciplinary team approach that includes physicians, psychotherapists, and dieticians.11 Treatment may be undertaken in an outpatient setting, but hospitalization for refeeding is recommended in cases with significant weight loss or malnourishment.11 Physical examination and laboratory studies are warranted, as excessive dietary restrictions can lead to weight loss and medical complications similar to those seen in AN, including osteopenia, anemia, hyponatremia, pancytopenia, bradycardia, and even pneumothorax and pneumomediastinum.19-21
There are no reported studies exploring the efficacy of psychotherapy or psychotropic medications for patients with ON. However, several treatments have been proposed given the symptom overlap with AN. Serotonin reuptake inhibitors may be beneficial for anxiety and obsessive-compulsive traits.18 However, patients with ON may refuse medications as unnatural substances.18
Cognitive behavioral therapy may be beneficial to address perfectionism and cognitive distortions, and exposure and response prevention may reduce obsessive-compulsive behaviors.11 Relaxation therapy may reduce mealtime anxiety. Psychoeducation may correct inaccurate beliefs about food groups, purity, and preparation, but it may induce emotional stress for the patient with ON.11
Conclusion
Orthorexia nervosa is perhaps best summarized as an obsession with healthy eating with associated restrictive behaviors. However, the attempt to attain optimum health through attention to diet may lead to malnourishment, loss of relationships, and poor quality of life.11 It is a little-understood disorder with uncertain etiology, imprecise assessment tools, and no formal diagnostic criteria or classification. Orthorexic characteristics vary from normal to pathologic in degree, and making a diagnosis remains a clinical judgment.22 Further research is needed to develop valid diagnostic tools and determining whether ON should be classified as a unique illness or a variation of other eating or anxiety disorders. Further research also may identify the etiology of ON, thus enabling targeted multidisciplinary treatment.
First named by Steven Bratman in 1997, orthorexia nervosa (ON) from the Greek ortho, meaning correct, and orexi, meaning appetite, is classified as an unspecified feeding and eating disorder in the Diagnostic and Statistical Manual of Mental Disorders 5th edition (DSM-5).1,2
Hypothetical Case
Mr. P is a 30-year-old male who presented to the mental health clinic with his wife. The patient recounted that he had wanted to “be healthy” since childhood and has focused on exercise and proper diet, but anxiety about diet and food intake have steadily increased. Two years ago, he adopted a vegetarian diet by progressively eliminating several foods and food groups from his diet. He now feels “proud” to eat certain organically grown fruits, vegetables, nuts, beans, and drink only fruit or vegetable juice.
His wife stated that he spent between 3 and 5 hours daily preparing food or talking to friends and family about “correct foods to eat.” He also believed that errors in dietary habits caused physical or mental illnesses. He reported significant guilt and shame whenever he “slips up” on his dietary regimen and eats anything containing seafood, beef, or pork products, which he corrects by a day of fasting. His wife was frustrated because he refused to go to restaurants and started declining offers from friends to eat dinner at their homes unless he could bring his prepared food. He describes feeling “annoyed” when he sees other people eating fast food or meat.
Mr. P reported no significant medical or surgical history. His family history was significant for anxiety in his mother. He used to drink alcohol socially but ceased a few years ago due to its carbohydrate content. He never smoked or used illicit drugs.
A mental status exam revealed a thin male who appeared his stated age. He was cooperative, casually dressed, and made fair eye contact. He spoke clearly with an anxious tone and appropriate rate and volume. His affect was congruent with stated anxious mood. He was alert, awake, and oriented to person, place, and time. He reported no paranoia, auditory or visual hallucinations, and suicidal or homicidal ideation.
A physical exam revealed a thin male in no distress who measured 5 feet 10 inches tall and weighed 145 pounds, which yielded a body mass index of 20.8. His vitals included temperature of 98° F, blood pressure 115/76, pulse 74, and oxygen saturation 98% on room air. The remaining physical examination revealed no abnormalities. A complete blood count, thyroid function, urinalysis, and urine drug screens were within normal limits. Comprehensive metabolic profile revealed decreased sodium of 130 meq/L. Electrocardiogram revealed bradycardia.
An ON diagnosis is made primarily through a clinical interview. Collateral information from individuals familiar with the patient can be helpful. Experts have proposed and recently revised criteria for ON (Table). Although the ORTO-15 assessment tool may assist with diagnosis, the tool does not substitute for the clinical interview.
Discussion
There is no reliable measure of prevalence of ON, though Varga and colleagues initially estimated ON to occur in 6.9% of the general population, and ON may occur more frequently in health care professionals and performance artists.3 However, these may be overestimates, as the assessment tool used in the study does not adequately separate people with healthy eating habits from those with ON.4,5
Most prevalence studies were conducted in Europe and Turkey, and prevalence of ON may differ in the U.S. population. A recent assessment determined a prevalence of about 1%, similar to that of other eating disorders.5 No study has reported a correlation between ON and gender, but a survey of 448 college students in the U.S. (mean age 22 years) reported highest ON tendencies in Hispanic/Latino and overweight/obese students.6
Relationship to Other Illnesses
There is significant debate whether ON is a single syndrome, a variance of other syndromes, or a behavioral and culturally influenced attitude.7,8 Although ON may lead to or be comorbid with anorexia nervosa (AN) or obsessive-compulsive disorder (OCD), subtle differences exist between ON and these conditions.
To meet DSM-5 diagnostic criteria for AN, patients must weigh below minimally normal weight for their height and age, have an intense fear of gaining weight or becoming fat, and have a disturbed experience of their weight or body shape or cannot recognize the severity of the low weight.2 In contrast, an individual with ON may possess normal or low-normal weight. Patients with AN focus on food quantity, while patients with ON tend to focus on food quality. As summarized by Bratman, “People are ashamed of their anorexia, but they actively evangelize their orthorexia. People with anorexia skip meals; people with orthorexia do not (unless they are fasting). Those with anorexia focus only on avoiding foods, while those with orthorexia both avoid foods they think are bad and embrace foods they think are super-healthy.”9
Similarities between ON and OCD include anxiety, a need to exert control, and perfectionism. However, patients with OCD tend to report distress from compulsive behavior and a desire to change, thus exhibiting insight into their illness.8,10 Similarities between obsessive-compulsive personality disorder (OCPD) and ON include perfectionism, rigid thinking, excessive devotion, hypermorality, and a preoccupation with details and perceived rules.11
While no studies have yet described ON as a feature of somatoform disorders, some experts have hypothesized that preoccupation with illness in a patient with somatization disorder may engender a preoccupation with food and diet as a way to combat either real or perceived illness.11 Finally, there is a report of ON associated with the prodromal phase of schizophrenia, and the development of ON may increase risk for future psychotic disorders.11,12
Pathophysiology
The exact cause of ON is unknown, though it is likely multifactorial. Individuals with ON have neurocognitive deficits similar to those seen in patients with AN and OCD, including impairments in set-shifting (flexible problem solving), external attention, and working memory.11,13 Given these cognitive deficits as well as similar symptomatology, there may be analogous brain dysfunction in patients with ON and AN or OCD. Neuroimaging studies of patients with AN have revealed dysregulation of dopamine transmission in the reward circuitry of the ventral striatum and the food regulatory mechanism in the hypothalamus.14
Dysmorphology of and dysfunction in neural circuitry, particularly the cortico-striato-thalamo-cortical pathway, have been implicated in OCD.15 Neuroimaging studies have revealed increased volume and activation of the orbitofrontal cortex, which may be associated with obsessions and difficulty with extinction recall.14,15 In contrast, decreased volume and activity of the thalamus may impair its ability to inhibit the orbitofrontal cortex.15,16 Decreased volume and activity of the cingulate gyrus may be associated with difficulty in error monitoring and fear conditioning, while overactivation of the parietal lobe and cerebellum may be associated with compulsive behaviors.15,16
Risk Factors
Factors that contribute to the development of AN and possibly ON include development of food preferences, inherited differences in taste perception, food neophobia or pickiness, being premorbidly overweight or obese, parental feeding practices, and a history of parental eating disorders.14 One survey associated orthorexic tendencies with perfectionism, appearance orientation, overweight preoccupation, self-classified weight, and fearful and dismissing attachment styles.17 Significant predictors of ON included overweight preoccupation, appearance orientation, and a history of an eating disorder.17
Treatment
In contrast to patients with AN, patients with ON may be easily amenable to treatment, given their pursuit of and emphasis on wellness.18 Experts recommend a multidisciplinary team approach that includes physicians, psychotherapists, and dieticians.11 Treatment may be undertaken in an outpatient setting, but hospitalization for refeeding is recommended in cases with significant weight loss or malnourishment.11 Physical examination and laboratory studies are warranted, as excessive dietary restrictions can lead to weight loss and medical complications similar to those seen in AN, including osteopenia, anemia, hyponatremia, pancytopenia, bradycardia, and even pneumothorax and pneumomediastinum.19-21
There are no reported studies exploring the efficacy of psychotherapy or psychotropic medications for patients with ON. However, several treatments have been proposed given the symptom overlap with AN. Serotonin reuptake inhibitors may be beneficial for anxiety and obsessive-compulsive traits.18 However, patients with ON may refuse medications as unnatural substances.18
Cognitive behavioral therapy may be beneficial to address perfectionism and cognitive distortions, and exposure and response prevention may reduce obsessive-compulsive behaviors.11 Relaxation therapy may reduce mealtime anxiety. Psychoeducation may correct inaccurate beliefs about food groups, purity, and preparation, but it may induce emotional stress for the patient with ON.11
Conclusion
Orthorexia nervosa is perhaps best summarized as an obsession with healthy eating with associated restrictive behaviors. However, the attempt to attain optimum health through attention to diet may lead to malnourishment, loss of relationships, and poor quality of life.11 It is a little-understood disorder with uncertain etiology, imprecise assessment tools, and no formal diagnostic criteria or classification. Orthorexic characteristics vary from normal to pathologic in degree, and making a diagnosis remains a clinical judgment.22 Further research is needed to develop valid diagnostic tools and determining whether ON should be classified as a unique illness or a variation of other eating or anxiety disorders. Further research also may identify the etiology of ON, thus enabling targeted multidisciplinary treatment.
1. Bratman S. Health food junkie. Yoga J. 1997;136:42-50.
2. American Psychiatric Association. Feeding and eating disorders. In: Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington, VA: American Psychiatric Publishing; 2013:329-354.
3. Varga M, Dukay-Szabó S, Túry F, van Furth EF. Evidence and gaps in the literature on orthorexia nervosa. Eat Weight Disord. 2013;18(2):103-111.
4. Donini LM, Marsili D, Graziani MP, Imbriale M, Cannella C. Orthorexia nervosa: validation of a diagnosis questionnaire. Eat Weight Disord. 2005;10(2):e28-e32.
5. Dunn TM, Gibbs J, Whitney N, Starosta A. Prevalence of orthorexia nervosa is less than 1 %: data from a US sample. Eat Weight Disord. 2016;22(1):185-192.
6. Bundros J, Clifford D, Silliman K, Neyman Morris M. Prevalence of orthorexia nervosa among college students based on Bratman’s test and associated tendencies. Appetite. 2016;101:86-94.
7. Vandereycken W. Media hype, diagnostic fad or genuine disorder? Professionals’ opinions about night eating syndrome, orthorexia, muscle dysmorphia, and emetophobia. Eat Disord. 2011;19(2):145-155.
8. Dell’Osso L, Abelli M, Carpita B, et al. Historical evolution of the concept of anorexia nervosa and relationships with orthorexia nervosa, autism, and obsessive-compulsive spectrum. Neuropsychiatr Dis Treat. 2016;12:1651-1660.
9. Bratman S. Orthorexia: an update. http://www.orthorexia.com/orthorexia-an-update. Updated October 5, 2015. Accessed April 18, 2017.
10. Dunn TM, Bratman S. On orthorexia nervosa: a review of the literature and proposed diagnostic criteria. Eat Behav. 2016;21:11-17.
11. Koven NS, Abry AW. The clinical basis of orthorexia nervosa: emerging perspectives. Neuropsychiatr Dis Treat. 2015;11:385-394.
12. Saddichha S, Babu GN, Chandra P. Orthorexia nervosa presenting as prodrome of schizophrenia. Schizophr Res. 2012;134(1):110.
13. Koven NS, Senbonmatsu R. A neuropsychological evaluation of orthorexia nervosa. Open J Psychiatry. 2013;3(2):214-222.
14. Gorwood P, Blanchet-Collet C, Chartrel N, et al. New insights in anorexia nervosa. Front Neurosci. 2016;10:256.
15. Milad MR, Rauch SL. Obsessive-compulsive disorder: beyond segregated cortico-striatal pathways. Trends Cogn Sci. 2012;16(1):43-51.
16. Tang W, Zhu Q, Gong X, Zhu C, Wang Y, Chen S. Cortico-striato-thalamo-cortical circuit abnormalities in obsessive-compulsive disorder: A voxel-based morphometric and fMRI study of the whole brain. Behav Brain Res. 2016;313:17-22.
17. Barnes MA, Caltabiano ML. The interrelationship between orthorexia nervosa, perfectionism, body image and attachment style. Eat Weight Disord. 2017;22(1):177-184.
18. Mathieu J. What is orthorexia? J Am Diet Assoc. 2005;105(10):1510-1512.
19. Catalina Zamora ML, Bote Bonaechea B, García Sánchez F, Ríos Rial B. Orthorexia nervosa. A new eating behavior disorder? [in Spanish]. Actas Esp Psiquiatr. 2005;33(1):66-68.
20. Moroze RM, Dunn TM, Craig Holland J, Yager J, Weintraub P. Microthinking about micronutrients: a case of transition from obsessions about healthy eating to near-fatal “orthorexia nervosa” and proposed diagnostic criteria. Psychosomatics. 2015;56(4):397-403.
21. Park SW, Kim JY, Go GJ, Jeon ES, Pyo HJ, Kwon YJ. Orthorexia nervosa with hyponatremia, subcutaneous emphysema, pneumomediastimum, pneumothorax, and pancytopenia. Electrolyte Blood Press. 2011;9(1):32-37.
22. Mogallapu RNG, Aynampudi AR, Scarff JR, Lippmann S. Orthorexia nervosa. The Kentucky Psychiatrist. 2012;22(3):3-6.
1. Bratman S. Health food junkie. Yoga J. 1997;136:42-50.
2. American Psychiatric Association. Feeding and eating disorders. In: Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington, VA: American Psychiatric Publishing; 2013:329-354.
3. Varga M, Dukay-Szabó S, Túry F, van Furth EF. Evidence and gaps in the literature on orthorexia nervosa. Eat Weight Disord. 2013;18(2):103-111.
4. Donini LM, Marsili D, Graziani MP, Imbriale M, Cannella C. Orthorexia nervosa: validation of a diagnosis questionnaire. Eat Weight Disord. 2005;10(2):e28-e32.
5. Dunn TM, Gibbs J, Whitney N, Starosta A. Prevalence of orthorexia nervosa is less than 1 %: data from a US sample. Eat Weight Disord. 2016;22(1):185-192.
6. Bundros J, Clifford D, Silliman K, Neyman Morris M. Prevalence of orthorexia nervosa among college students based on Bratman’s test and associated tendencies. Appetite. 2016;101:86-94.
7. Vandereycken W. Media hype, diagnostic fad or genuine disorder? Professionals’ opinions about night eating syndrome, orthorexia, muscle dysmorphia, and emetophobia. Eat Disord. 2011;19(2):145-155.
8. Dell’Osso L, Abelli M, Carpita B, et al. Historical evolution of the concept of anorexia nervosa and relationships with orthorexia nervosa, autism, and obsessive-compulsive spectrum. Neuropsychiatr Dis Treat. 2016;12:1651-1660.
9. Bratman S. Orthorexia: an update. http://www.orthorexia.com/orthorexia-an-update. Updated October 5, 2015. Accessed April 18, 2017.
10. Dunn TM, Bratman S. On orthorexia nervosa: a review of the literature and proposed diagnostic criteria. Eat Behav. 2016;21:11-17.
11. Koven NS, Abry AW. The clinical basis of orthorexia nervosa: emerging perspectives. Neuropsychiatr Dis Treat. 2015;11:385-394.
12. Saddichha S, Babu GN, Chandra P. Orthorexia nervosa presenting as prodrome of schizophrenia. Schizophr Res. 2012;134(1):110.
13. Koven NS, Senbonmatsu R. A neuropsychological evaluation of orthorexia nervosa. Open J Psychiatry. 2013;3(2):214-222.
14. Gorwood P, Blanchet-Collet C, Chartrel N, et al. New insights in anorexia nervosa. Front Neurosci. 2016;10:256.
15. Milad MR, Rauch SL. Obsessive-compulsive disorder: beyond segregated cortico-striatal pathways. Trends Cogn Sci. 2012;16(1):43-51.
16. Tang W, Zhu Q, Gong X, Zhu C, Wang Y, Chen S. Cortico-striato-thalamo-cortical circuit abnormalities in obsessive-compulsive disorder: A voxel-based morphometric and fMRI study of the whole brain. Behav Brain Res. 2016;313:17-22.
17. Barnes MA, Caltabiano ML. The interrelationship between orthorexia nervosa, perfectionism, body image and attachment style. Eat Weight Disord. 2017;22(1):177-184.
18. Mathieu J. What is orthorexia? J Am Diet Assoc. 2005;105(10):1510-1512.
19. Catalina Zamora ML, Bote Bonaechea B, García Sánchez F, Ríos Rial B. Orthorexia nervosa. A new eating behavior disorder? [in Spanish]. Actas Esp Psiquiatr. 2005;33(1):66-68.
20. Moroze RM, Dunn TM, Craig Holland J, Yager J, Weintraub P. Microthinking about micronutrients: a case of transition from obsessions about healthy eating to near-fatal “orthorexia nervosa” and proposed diagnostic criteria. Psychosomatics. 2015;56(4):397-403.
21. Park SW, Kim JY, Go GJ, Jeon ES, Pyo HJ, Kwon YJ. Orthorexia nervosa with hyponatremia, subcutaneous emphysema, pneumomediastimum, pneumothorax, and pancytopenia. Electrolyte Blood Press. 2011;9(1):32-37.
22. Mogallapu RNG, Aynampudi AR, Scarff JR, Lippmann S. Orthorexia nervosa. The Kentucky Psychiatrist. 2012;22(3):3-6.
A Review of Psychostimulants for Adults With Depression
Depression is a common condition that significantly impairs social and occupational functioning. Many patients do not respond to first-line pharmacotherapies and are considered to have treatment-resistant depression (TRD). These patients may benefit from augmentation of their antidepressant to reduce depression. Multiple medications have demonstrated various degrees of efficacy for augmentation, including psychostimulants. This article reviews studies of psychostimulants as augmentation agents for TRD and discusses risks, offers advice, and makes recommendations for clinicians who prescribe stimulants.
Background
Major depressive disorder (MDD) is a common psychiatric condition that significantly impairs quality of life.1 It is a recurrent illness, averaging 2 relapses per decade. The probability of recurrence increases with the number of depressive episodes.2,3 A patient who experiences major depressive episodes alternating with euthymia has unipolar depression; whereas one who experiences major depressive episodes alternating with episodes of mania or hypomania has bipolar depression.4
Despite adequate dose and duration of pharmacotherapy, many individuals with unipolar or bipolar depression do not achieve and sustain remission.5 Remission rates decrease and relapse rates increase with subsequent failed antidepressant trials.6 It is difficult to identify factors that predict treatment resistance, but one review of antidepressant studies found that patients who did not demonstrate a response within 3 weeks of medication initiation were less likely to respond after a longer duration.7
Treatment-resistant depression is commonly, but not universally, defined as lack of response after trials of 2 or more antidepressants with different mechanisms of action for sufficient duration.5 This definition will be used here as well. Other definitions have proposed stages of TRD, but these require further study to evaluate their reliability and predictive utility.8 Due to lack of consensus regarding the definition of TRD, it is not possible to determine the exact prevalence of TRD.
Patients with TRD may benefit from augmentation of their medication regimen. Augmentation with lithium has yielded conflicting results, and its efficacy with newer antidepressants is not well studied.9-12 Triiodothyronine, buspirone, and pindolol have demonstrated some efficacy when added to serotonin reuptake inhibitors (SRIs).10,12,13 Second-generation antipsychotic drugs, antidepressant drug combinations, omega-3 fatty acids, S-adenosyl methionine (SAMe), and L-methylfolate have demonstrated some efficacy in some studies as well.12,14-23 In patients with depression who have not responded to these strategies, psychostimulant augmentation may be appropriate.
Methods
A literature search was conducted following an algorithmic approach in the MEDLINE/PubMed database for studies in English from January 1985 to August 2014 of stimulants as augmenting agents for depression, using the Medical Subject Headings stimulant, depression, and augmentation, combined with an AND operator. The search was limited to adult humans and excluded case reports and letters, to identify studies with stronger evidence. Also excluded were studies using caffeine (to augment electroconvulsive therapy for depression) and pemoline as the sole augmenting stimulant as well as studies of patients with comorbid mental health diagnoses and studies that initiated stimulants and antidepressants simultaneously to assess antidepressant response.
This review organized results by stimulant rather than by depression type, even though some studies used > 1 stimulant or recruited patients with different types of depression. Although prevalence, prognosis, and monotherapy differ for unipolar and bipolar depression, psychostimulants target similar symptoms, despite augmenting different monotherapies in unipolar and bipolar depression. Therefore, no distinction is made between assessing studies of stimulants for unipolar and bipolar depression.
Results
A total of 70 articles were identified, and 31 studies met inclusion criteria (Figure). Of the studies included, 12 were double-blind, placebo-controlled (DBPC) trials and 19 were retrospective chart reviews or open studies. Most studies evaluated depression, using validated scales, such as the Hamilton Depression Rating Scale, Montgomery-Asberg Depression Rating Scale, Clinical Global Impressions of Severity, Inventory of Depressive Symptoms, Carroll Depression Rating Scale, Global Assessment of Functioning, Quick Inventory of Depressive Symptomatology, or the Psychiatric Symptom Assessment Scale. Study details are provided in Tables 1 to 4.
Dextroamphetamine and Methylphenidate
Dextroamphetamine and methylphenidate are indicated for the treatment of attention-deficit/hyperactivity disorder (ADHD) and exert their effects by inhibiting uptake of norepinephrine and dopamine.24 In one chart review, patients received dextroamphetamine or methylphenidate augmentation of monoamine oxidase inhibitors (MAOIs) alone or with concurrent tricyclic antidepressants; the majority reported decreased depression.25 In an openlabel trial, dextroamphetamine was titrated to efficacy in patients who were receiving an MAOI with or without pemoline.26 Nearly 80% of patients reported long-lasting improvement in depression. In an open-label trial, all patients reported decreased depression when methylphenidate was added to SRIs; however, no scales were used.27
In a case series, patients with both major depression and persistent depressive disorder (dysthymia) experienced a substantial, quick, and sustained response to dextroamphetamine or methylphenidate augmentation.28 Addition of lisdexamfetamine significantly reduced depressive symptoms in individuals with inadequate response to escitalopram.29 Patients with full or partial remission of depression noted improved executive function and residual depressive symptoms after lisdexamfetamine was added to SRI monotherapy.30 In a trial in which patients received dexamphetamine or methylphenidate as monotherapy or augmentation, 30% to 34% of patients reported mood improvement, but 36% reported no improvement.31 In an extension study, low-dose psychostimulants quickly diminished melancholia.32
Methylphenidate was safe and effective in patients with bipolar depression receiving treatment for 1 to 5 years; 44% evidenced significant improvement.33 When offered to patients with bipolar depression, patients receiving methylphenidate or dextroamphetamine reported less depression or sedation and did not develop tolerance, mania, or misuse.34 A case series concluded that methylphenidate addition to mood stabilizers was generally effective and safe.35
However, not all preparations of methylphenidate have demonstrated efficacy. In one study, osmotic controlledrelease oral system (OROS) methylphenidate improved apathy and fatigue but not overall depression.36 Although OROS methylphenidate similarly failed to demonstrate statistically significant efficacy in another study, more responders were documented in the treatment group.37
Although this review focuses on stimulants as augmenting agents in patients with depression, it is worth noting the limited number of studies evaluating stimulants’ effect on depression in patients with traumatic brain injury. This observation is of concern, as these conditions are frequently comorbid in returning veterans. One study noted that methylphenidate was an effective monotherapy for depression; whereas another study found that methylphenidate monotherapy reduced depression as well as sertraline, was better tolerated, and improved fatigue and cognition.38,39
Modafinil and Armodafinil
Modafinil and armodafinil (the R-enantiomer of modafinil) are indicated for improving wakefulness in individuals with narcolepsy, obstructive sleep apnea, and shift work sleep disorder by modulating glutamate, gamma amino-butyric acid, and histamine.40,41 Although they increase extracellular dopamine concentrations, they do not cause an increase in dopamine release and may have less misuse potential than that of dextroamphetamine and methylphenidate.40,41 In a study of 7 patients with unipolar or bipolar depression, all patients achieved full or partial remission with minimal adverse effects (AEs).42 In a prospective study, 41% of patients reported only mild depression or full remission with modafinil augmentation.43
Multiple trials and a pooled analysis noted decreased depression and fatigue and improved cognition in patients receiving modafinil augmentation compared with mood stabilizers or antidepressants.44-49 Modafinil is a useful adjunct for partial responders to SRIs, resulting in rapid mood improvement and decreased fatigue.50-54 However, in one study, modafinil did not demonstrate efficacy compared with placebo. This result was attributed to premature study termination after 2 modafinil-treated patients developed suicidal ideation.55 A post hoc analysis found no difference in frequency of suicidal ideation between groups.
Two DBPC studies evaluated armodafinil in patients with bipolar depression. In both studies it was added to a mood-stabilizing agent (lithium, valproate, aripiprazole, olanzapine, lamotrigine, risperidone, or ziprasidone), and patients receiving armodafinil reported significant reductions
in depression.56,57
Atomoxetine
Atomoxetine is a norepinephrine reuptake inhibitor indicated for the treatment of ADHD and is considered to have no misuse potential due to lack of dopamine modulation.58 In one study, 15 patients received atomoxetine added to their antidepressant, and 60% experienced significant symptom reduction.59 A chart review noted decreases in fatigue and depression when atomoxetine was added to an SRI, mirtazapine, or amitriptyline.60 However, in a DBPC trial, atomoxetine did not lead to significant changes in depression.61
Discussion
There is a limited amount of high-quality evidence to support psychostimulant augmentation, as noted by the relatively few DBPC trials, most of short duration. The evidence supports their efficacy primarily for unipolar depression, as 14 studies evaluated patients with unipolar depression, whereas only 7 studies evaluated patients with bipolar depression. The remaining studies recruited patients with both depression types. Collectively, modafinil and armodafinil have the most evidence in DBPC trials.
There are relatively few DBPC trials with high power and sufficient duration for dextroamphetamine and methylphenidate preparations. This discovery is surprising, considering the duration that these medications have been available. However, several chart reviews and open-label trials provided some evidence to support their use in patients without a history of substance misuse or cardiac conditions.62 Osmotic controlled- release oral system methylphenidate seems to be ineffective, and the efficacy of atomoxetine for augmentation
is uncertain.
Precautions
Prescribing physicians who offer stimulants should consider potential AEs, such as psychosis, anorexia, anxiety, insomnia, mood changes (eg, anger), misuse, addiction, mania, and cardiovascular problems. Psychostimulants have been implicated in precipitating psychosis.63,64 However, in a 12-month study of 250 adults with ADHD, 73 reported AEs, and only 31 discontinued the stimulant. Adverse effects leading to discontinuation included mood instability (n = 7), agitation (n = 6), irritability (n = 4), or decreased appetite (n = 4).65
Although associated with the risks of anorexia and insomnia in patients with ADHD, methylphenidate rapidly improved daytime sleepiness and mood, and—paradoxically—appetite and nighttime sleep in medically ill elderly patients with depression.66 Misuse or abuse of methylphenidate and dextroamphetamine were noted in 23% of patients referred for substance misuse.67 Nonetheless, little evidence exists that these drugs possess significant misuse potential in patients taking them as prescribed. As a prodrug, lisdexamfetamine is hypothesized to have less abuse potential compared with dextroamphetamine and methylphenidate, but it carries the same prescribing and monitoring precautions.68 Risks related to stimulant usage extend to manic symptoms.69 Patients with bipolar disorder should not receive stimulants if they have a history of stimulant-induced mania, rapid cycling, or psychosis.70
Long-term cardiovascular safety data exist for dextroamphetamine and methylphenidate but are limited or unavailable for modafinil, armodafinil, and atomoxetine. A retrospective cohort study found no significant increase in the number of cardiac events in patients receiving dextroamphetamine,
methylphenidate, or atomoxetine for an average of 1 year compared with controls.71 Another cohort study of > 44,000 patients found that initiation of
methylphenidate was associated with increased risk of sudden death or arrhythmia, but the risk was attributed to an unmeasured confounding factor, as the authors found a negative correlation between methylphenidate dose and all cardiovascular events.72
Recent practice guidelines recommend that before prescribing stimulants, clinicians should perform a physical examination (including heart and lung auscultation), obtain vital signs and height and weight, and request an electrocardiogram in case of abnormal findings on a cardiovascular examination or in case of a personal or family history of heart disease. Before offering atomoxetine, clinicians should evaluate the patient for a history of liver disease (and check liver function studies in case of a positive history). Clinicians should also assess risk of self-harm prior to initiating psychostimulant therapy.73 Throughout treatment, clinicians should evaluate the patient for changes in blood pressure, pulse, weight or mood, as well as the development of dependence or misuse. Urine toxicology testing is recommended for dextroamphetamine and methylphenidate to screen for adherence and diversion.
Limitations
Using only PubMed and MEDLINE databases limited the search to articles published in English after 1985, excluding letters and case reports to identify studies with higher evidence (the studies were not weighted based on study design). In addition, the studies had certain limitations. These include a limited number of DBPC trials, most were of short duration. It is also difficult to compare studies due to various rating scales used and concurrent
medication regimens of study subjects. These limitations raise questions surrounding the long-term efficacy of stimulants, and there is no consensus for how long a stimulant should be continued if beneficial. Longer, higherpowered, DBPC trials are warranted to determine longterm efficacy and safety of stimulant augmentation.62
Conclusion
For patients with depression who have not responded to other augmentation strategies, psychostimulants may be offered to improve mood, energy, and concentration. For clinicians considering stimulant augmentation, modafinil and armodafinil are reasonable choices given their efficacy in double-blind, placebo-controlled trials and lower risk of misuse. Dextroamphetamine (particularly lisdexamphetamine) and methylphenidate may be appropriate for patients who have not benefited from or tolerated modafinil or armodafinil, provided these patients do not have a medical history of cardiac disease or current substance use.
Osmotic controlled-release oral system methylphenidate seems to be ineffective as an augmenting agent. The efficacy of atomoxetine for augmentation is questionable, but atomoxetine could be offered if other stimulants were contraindicated, ineffective, or poorly tolerated. Both OROS methylphenidate and atomoxetine should be evaluated in additional trials before they can be recommended as augmentation therapies. Certain psychostimulants may be appropriate and reasonable adjunctive pharmacotherapies for patients with unipolar or bipolar depression who have failed other augmentation strategies, for patients who have significant fatigue or cognitive complaints, or for elderly patients with melancholic or somatic features of depression.
Acknowledgements
The authors thank Maureen Humphrey-Shelton and Kathy Thomas for their help in obtaining references.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Kessler RC, Berglund P, Demler O, Jin R, Merikangas KR, Walters EE. Lifetime prevalence and age-of-onset distribution of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):593-602.
2. Solomon DA, Keller MB, Leon AC, et al. Multiple recurrences of major depressive disorder. Am J Psychiatry. 2000;157(2):229-233.
3. Katon WJ, Fan MY, Lin EH, Unützer J. Depressive symptom deterioration in a large
primary care-based elderly cohort. Am J Geriatr Psychiatry. 2006;14(3):246-254.
4. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington, VA: American Psychiatric Association; 2013.
5. McIntyre RS, Filteau M-J, Martin L, et al. Treatment-resistant depression: Definitions, review of the evidence, and algorithmic approach. J Affect Disord. 2014;156:1-7.
6. Gaynes BN, Warden D, Trivedi MH, Wisniewski SR, Fava M, Rush AJ. What did STAR*D teach us? Results from a large-scale, practical, clinical trial for patients with depression. Focus. 2012;10(4):510-517.
7. Kudlow PA, Cha DS, McIntyre RS. Predicting treatment response in major depressive disorder: The impact of early symptomatic improvement. Can J Psychiatry. 2012;57(12):782-788.
8. Ruhé HG, van Rooijen G, Spijker J, Peeters FP, Schene AH. Staging methods for treatment resistant depression. A systematic review. J Affect Disord. 2012;137(1-3):35-45.
9. Bauer M, Dopfmer S. Lithium augmentation treatment-resistant depression: Metaanalysis of placebo-controlled studies. J Clin Psychopharmacol. 1999;19(5):427-434.
10. Nierenberg AA, Fava M, Trivedi MH, et al. A comparison of lithium and T(3) augmentation following two failed medication treatments for depression: A STAR*D report. Am J Psychiatry. 2006;163(9):1519-1530.
11. Nierenberg AA, Papakostas GI, Petersen T, et al. Lithium augmentation of nortriptyline
for subjects resistant to multiple antidepressants. J Clin Psychopharmacol. 2003;23(1):92-95.
12. Connolly KR, Thase ME. If at first you don’t succeed: A review of the evidence for antidepressant augmentation, combination, and switching strategies. Drugs. 2011;71(1):43-64.
13. Trivedi MH, Fava M, Wisniewski SR, et al; STAR*D Study Team. Medication augmentation after the failure of SSRIs for depression. N Engl J Med. 2006;354(12):1243-1252.
14. Papakostas GI, Shelton RC, Smith J, Fava M. Augmentation of antidepressants with atypical antipsychotic medications for treatment resistant major depressive disorder: A meta-analysis. J Clin Psychiatry. 2007;68(6):826-831.
15. Mahmoud RA, Pandina GJ, Turkoz I, et al. Risperidone for treatment-refractory major depressive disorder: A randomized trial. Ann Intern Med. 2007;147(9):593-602.
16. Barbee JG, Conrad EJ, Jamhour NJ. The effectiveness of olanzapine, risperidone, quetiapine, and ziprasidone as augmentation agents in treatment resistant depressive disorder. J Clin Psychiatry. 2004;65(7):975-981.
17. Fatemi SH, Emamian ES, Kist DA. Venlafaxine and bupropion combination therapy in a case of treatment-resistant depression. Ann Pharmacother.1999;33(6):701-703.
18. Carpenter LL, Yasman S, Price LH. A double-blind, placebo-controlled study of antidepressant augmentation with mirtazapine. Biol Psychiatry. 2002;51(2):183-188.
19. Hannan N, Hamzah Z, Akinpeloye HO, Meagher D. Venlafaxine-mirtazapine combination therapy in the treatment of persistent depressive illness. J Psychopharmacol. 2007;21(2):161-164.
20. McGrath PJ, Stewart JW, Fava M, et al. Tranylcypromine versus venlafaxine plus mirtazapine following three failed antidepressant medication trials for depression: A STAR*D report. Am J Psychiatry. 2006;163(9):1531-1541.
21. Blier P, Ward HE, Tremblay P, Laberge L, Hébert C, Bergeron R. Combination of antidepressant medications from treatment initiation for major depressive disorder: A double-blind randomized study. Am J Psychiatry. 2010;167(3):281-288.
22. Papakostas GI, Mischoulon D, Shyu I, Alpert JE, Fava M. S-adenosyl methionine (SAMe) augmentation of serotonin reuptake inhibitors for antidepressant nonresponders with major depressive disorder: A double blind randomized clinical trial. Am J Psychiatry. 2010;167(8):942-948.
23. Papakostas GI, Shelton RC, Zajecka JM, et al. L-methylfolate as adjunctive therapy
for SSRI-resistant major depression: Results of two randomized, double-blind,
parallel-sequential trials. Am J Psychiatry. 2012;169(12):1267-1274.
24. Korston TR. Drugs of abuse. In: Katzung BG, ed. Basic and Clinical Pharmacology. 9th ed. New York, NY: McGraw-Hill; 2004:521-523.
25. Feighner JP, Herbstein J, Damlouji N. Combined MAOI, TCA, and direct stimulant therapy of treatment-resistant depression. J Clin Psychiatry. 1985;46(6):206-209.
26. Fawcett J, Kravitz HM, Zajecka JM, Schaff MR. CNS stimulant potentiation of monoamine oxidase inhibitors in treatment-refractory depression. J Clin Psychopharmacol. 1991;11(2):127-132.
27. Stoll AL, Pillay SS, Diamond L, Workum SB, Cole JO. Methylphenidate augmentation of serotonin selective reuptake inhibitors: A case series. J Clin Psychiatry. 1996;57(2):72-76.
28. Masand PS, Anand VS, Tanquary JF. Psychostimulant augmentation of second generation antidepressants: A case series. Depress Anxiety. 1998;7(2):89-91.
29. Trivedi MH, Cutler AJ, Richards C, et al. A randomized control trial of the efficacy and safety of lisdesxamfetamine dimesylate as augmentation therapy in adults with residual symptoms of major depressive disorder after treatment with escitalopram. J Clin Psychiatry. 2013;74(8):802-809.
30. Madhoo M, Keefe RS, Roth RM, et al. Lisdexamfetamine dimesylate augmentation in adults with persistent executive dysfunction after partial or full remission of major depressive disorder. Neuropsychopharmacology. 2014;39(6):1388-1398.
31. Parker G, Brotchie H. Do the old psychostimulant drugs have a role in managing treatment-resistant depression. Acta Psychiatr Scand. 2010;121(4):308-314.
32. Parker G, Brotchie H, McClure G, Fletcher K. Psychostimulants for managing unipolar and bipolar treatment-resistant melancholic depression: A medium term evaluation of cost benefits. J Affect Disord. 2013;151(1):360-364.
33. Lydon E, El-Mallakh RS. Naturalistic long-term use of methylphenidate in bipolar disorder. J Clin Psychopharmacol. 2006;26(5):516-518.
34. Carlson PJ, Merlock MC, Suppes T. Adjunctive stimulant use in patients with bipolar disorder: Treatment of residual depression and sedation. Bipolar Disord. 2004;6(5):416-420.
35. El-Mallakh RS. An open study of methylphenidate in bipolar depression. Bipolar Disord. 2000;2(1):56-59.
36. Ravindran AV, Kennedy SH, O’Donovan MC, Fallu A, Camacho F, Binder CE. Osmotic-release oral system methylphenidate augmentation of antidepressant monotherapy in major depressive disorder: Results of a double-blind, randomized, placebo-controlled trial. J Clin Psychiatry. 2008;69(1):87-94.
37. Patkar AA, Masand PS, Pae CU, et al. A randomized, double-blind, placebocontrolled
trial of augmentation with an extended release formulation of methylphenidate in outpatients with treatment-resistant depression. J Clin Psychopharmacol. 2006;26(6):653-656.
38. Lee H, Kim SW, Kim JM, Shin IS, Yang SJ, Yoon JS Comparing effects of methylphenidate, sertraline, and placebo on neuropsychiatric sequelae in patients with
traumatic brain injury. Hum Psychopharmacol. 2005;20(2):97-104.
39. Gualtieri CT, Evans RW. Stimulant treatment for the neurobehavioural sequelae of traumatic brain injury. Brain Inj. 1988;2(4):273-290.
40. Provigil [package insert]. North Wales, PA: Cephalon Inc; 2015.
41. Nuvigil [package insert]. Frazer, PA: Cephalon, Inc; 2013.
42. Menza MA, Kaufman KR, Castellanos A. Modafinil augmentation of antidepressant treatment in depression. J Clin Psychiatry. 2000;61(5):378-381.
43. Markovitz PJ, Wagner S. An open-label trial of modafinil augmentation in patients with partial response to antidepressant therapy. J Clin Psychopharmacol. 2003;23(2):207-209.
44. Fernandes PP, Petty F. Modafinil for remitted bipolar depression with hypersomnia. Ann Pharmacother. 2003;37(12):1807-1809.
45. Nasr S. Modafinil as adjunctive therapy in depressed outpatients. Ann Clin Psychiatry. 2004;16(3):133-138.
46. DeBattista C, Lembke A, Solvason HB, Ghebremichael R, Poirier J. A prospective trial of modafinil as an adjunctive treatment of major depression. J Clin Psychopharmacol. 2004;24(1):87-90.
47. Nasr S, Wendt B, Steiner K. Absence of mood switch with and tolerance to modafinil: A replication study from a large private practice. J Affect Disord. 2006;95(1-3):111-114.
48. DeBattista C, Doghramji K, Menza MA, Rosenthal MH, Fieve RR; Modafinil in Depression Study Group. Adjunct modafinil for the short-term treatment of fatigue and sleepiness in patients with major depressive disorder: A preliminary doubleblind, placebo-controlled study. J Clin Psychiatry. 2003;64(9):1057-1064.
49. Frye MA, Grunze H, Suppes T, et al. A placebo-controlled evaluation of adjunctive modafinil in the treatment of bipolar depression. Am J Psychiatry. 2007;164(8):1242-1249.
50. Fava M, Thase ME, DeBattista C, Doghramji K, Arora S, Hughes RJ. Modafinil augmentation of selective serotonin reuptake inhibitor therapy in MDD partial responders with persistent fatigue and sleepiness. Ann Clin Psychiatry. 2007;19(3):153-159.
51. Thase ME, Fava M, DeBattista C, Arora S, Hughes RJ. Modafinil augmentation of SSRI therapy in patients with major depressive disorder and excessive sleepiness and fatigue: A 12-week, open-label, extension study. CNS Spectr. 2006;11(2):93-102.
52. Fava M, Thase ME, DeBattista C. A multicenter, placebo-controlled study of modafinil augmentation in partial responders to selective serotonin reuptake inhibitors with persistent fatigue and sleepiness. J Clin Psychiatry. 2005;66(1):85-93.
53. Abolfazli R, Hosseini M, Ghanizadeh A, et al. Double-blind randomized parallelgroup clinical trial of efficacy of the combination fluoxetine plus modafinil versus fluoxetine plus placebo in the treatment of major depression. Depress Anxiety. 2011;28(4):297-302.
54. Rasmussen NA, Schrøder P, Olsen LR, Brødsgaard M, Undén M, Bech P. Modafinil augmentation in depressed patients with partial response to antidepressants: A pilot study on self-reported symptoms covered by the Major Depression Inventory (MDI) and the Symptom Checklist (SCL-92). Nord J Psychiatry. 2005;59(3):173-178.
55. Dunlop BW, Crits-Christoph P, Evans DL, et al. Coadministration of modafinil and a selective serotonin reuptake inhibitor from the initiation of treatment of major depressive disorder with fatigue and sleepiness: A double-blind, placebocontrolled study. J Clin Psychopharmacol. 2007;27(6):614-619.
56. Calabrese JR, Ketter TA, Youakim JM, Tiller JM, Yang R, Frye MA. Adjunctive armodafinil
for major depressive episodes associated with bipolar I disorder: A randomized multicenter, double-blind, placebo-controlled, proof-of-concept study. J Clin Psychiatry. 2010;71(10):1363-1370.
57. Calabrese JR, Frye MA, Yang R, Ketter TA; Armodafinil Treatment Trial Study Network. Efficacy and safety of adjunctive armodafinil in adults with major depressive episodes associated with bipolar I disorder: A randomized, double-blind, placebo-controlled, multicenter trial. J Clin Psychiatry. 2014;75(10):1054-1061.
58. Strattera [package insert]. Indianapolis, IN. Lilly; 2015.
59. Carpenter LL, Milosavljevic N, Schecter JM, Tyrka AR, Price LH. Augmentation with open-label atomoxetine for partial or nonresponse to antidepressants. J Clin Psychiatry. 2005;66(10):1234-1238.
60. Papakostas GI, Petersen TJ, Burns AM, Fava M. Adjunctive atomoxetine for residual
fatigue in major depressive disorder. J Psychiatr Res. 2006;40(4):370-373.
61. Michelson D, Adler LA, Amsterdam JD, et al. Addition of atomoxetine for depression
incompletely responsive to sertraline: A randomized, double-blind, placebocontrolled study. J Clin Psychiatry. 2007;68(4):582-587.
62. Corp SA, Gitlin MJ, Altshuler LL. A review of the use of stimulants and stimulant alternatives in treating bipolar depression and major depressive disorder. J Clin Psychiatry. 2014;75(9):1010-1018.
63. Kraemer M, Uekermann J, Wiltfang J, Kis B. Methylphenidate-induced psychosis in adult attention-deficit/hyperactivity disorder: Report of 3 new cases and review of the literature. Clin Neuropharmacol. 2010;33(4):204-206.
64. Berman SM, Kuczenski R, McCracken JT, London ED. Potential adverse effects of amphetamine treatment on brain and behavior: A review. Mol Psychiatry. 2009;14(2):123-142.
65. Fredriksen M, Dahl AA, Martinsen EW, Klungsøyr O, Haavik J, Peleikis DE. Effectiveness of one-year pharmacological treatment of adult attention-deficit/hyperactivity disorder (ADHD): An open-label prospective study of time in treatment, dose, side-effects and comorbidity. Eur Neuropsychopharmacol. 2014;24(12):1873-1874.
66. Hardy SE. Methylphenidate for the treatment of depressive symptoms, including fatigue and apathy, in medically ill older adults and terminally ill adults. Am J Geriatr Pharmacother. 2009;7(1):34-59.
67. Williams RJ, Goodale LA, Shay-Fiddler MA, Gloster SP, Chang SY. Methylphenidate and dextroamphetamine abuse in substance-abusing adolescents. Am J Addict. 2004;13(4):381-389.
68. Madaan V, Kolli V, Bestha DP, Shah MJ. Update on optimal use of lisdexamfetamine in the treatment of ADHD. Neuropsychiatr Dis Treat. 2013;9:977-983.
69. Ross RG. Psychotic and manic-like symptoms during stimulant treatment of attention deficit hyperactivity disorder. Am J Psychiatry. 2006;163(7):1149-1152.
70. Dell’Osso B, Ketter TA. Use of adjunctive stimulants in adult bipolar depression. Int J Neuropsychopharmacol. 2013;16(1):55-68.
71. Habel LA, Cooper WO, Sox CM, et al. ADHD medications and risk of serious cardiovascular events in young and middle-aged adults. JAMA. 2011;306(24):2673-2683.
72. Schelleman H, Bilker WB, Kimmel SE, et al. Methylphenidate and risk of serious cardiovascular events in adults. Am J Psychiatry. 2012;169(2):178-185.
73. Bolea-Alamañac B, Nutt DJ, Adamou M, et al; British Association for Psychopharmacology. Evidence-based guidelines for the pharmacological management of attention deficit hyperactivity disorder: Update on recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2014;28(3):179-203.
74. Moher D, Liberati A, Tetzlaff J, Altman DG; The PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009;6(6):e1000097.
Depression is a common condition that significantly impairs social and occupational functioning. Many patients do not respond to first-line pharmacotherapies and are considered to have treatment-resistant depression (TRD). These patients may benefit from augmentation of their antidepressant to reduce depression. Multiple medications have demonstrated various degrees of efficacy for augmentation, including psychostimulants. This article reviews studies of psychostimulants as augmentation agents for TRD and discusses risks, offers advice, and makes recommendations for clinicians who prescribe stimulants.
Background
Major depressive disorder (MDD) is a common psychiatric condition that significantly impairs quality of life.1 It is a recurrent illness, averaging 2 relapses per decade. The probability of recurrence increases with the number of depressive episodes.2,3 A patient who experiences major depressive episodes alternating with euthymia has unipolar depression; whereas one who experiences major depressive episodes alternating with episodes of mania or hypomania has bipolar depression.4
Despite adequate dose and duration of pharmacotherapy, many individuals with unipolar or bipolar depression do not achieve and sustain remission.5 Remission rates decrease and relapse rates increase with subsequent failed antidepressant trials.6 It is difficult to identify factors that predict treatment resistance, but one review of antidepressant studies found that patients who did not demonstrate a response within 3 weeks of medication initiation were less likely to respond after a longer duration.7
Treatment-resistant depression is commonly, but not universally, defined as lack of response after trials of 2 or more antidepressants with different mechanisms of action for sufficient duration.5 This definition will be used here as well. Other definitions have proposed stages of TRD, but these require further study to evaluate their reliability and predictive utility.8 Due to lack of consensus regarding the definition of TRD, it is not possible to determine the exact prevalence of TRD.
Patients with TRD may benefit from augmentation of their medication regimen. Augmentation with lithium has yielded conflicting results, and its efficacy with newer antidepressants is not well studied.9-12 Triiodothyronine, buspirone, and pindolol have demonstrated some efficacy when added to serotonin reuptake inhibitors (SRIs).10,12,13 Second-generation antipsychotic drugs, antidepressant drug combinations, omega-3 fatty acids, S-adenosyl methionine (SAMe), and L-methylfolate have demonstrated some efficacy in some studies as well.12,14-23 In patients with depression who have not responded to these strategies, psychostimulant augmentation may be appropriate.
Methods
A literature search was conducted following an algorithmic approach in the MEDLINE/PubMed database for studies in English from January 1985 to August 2014 of stimulants as augmenting agents for depression, using the Medical Subject Headings stimulant, depression, and augmentation, combined with an AND operator. The search was limited to adult humans and excluded case reports and letters, to identify studies with stronger evidence. Also excluded were studies using caffeine (to augment electroconvulsive therapy for depression) and pemoline as the sole augmenting stimulant as well as studies of patients with comorbid mental health diagnoses and studies that initiated stimulants and antidepressants simultaneously to assess antidepressant response.
This review organized results by stimulant rather than by depression type, even though some studies used > 1 stimulant or recruited patients with different types of depression. Although prevalence, prognosis, and monotherapy differ for unipolar and bipolar depression, psychostimulants target similar symptoms, despite augmenting different monotherapies in unipolar and bipolar depression. Therefore, no distinction is made between assessing studies of stimulants for unipolar and bipolar depression.
Results
A total of 70 articles were identified, and 31 studies met inclusion criteria (Figure). Of the studies included, 12 were double-blind, placebo-controlled (DBPC) trials and 19 were retrospective chart reviews or open studies. Most studies evaluated depression, using validated scales, such as the Hamilton Depression Rating Scale, Montgomery-Asberg Depression Rating Scale, Clinical Global Impressions of Severity, Inventory of Depressive Symptoms, Carroll Depression Rating Scale, Global Assessment of Functioning, Quick Inventory of Depressive Symptomatology, or the Psychiatric Symptom Assessment Scale. Study details are provided in Tables 1 to 4.
Dextroamphetamine and Methylphenidate
Dextroamphetamine and methylphenidate are indicated for the treatment of attention-deficit/hyperactivity disorder (ADHD) and exert their effects by inhibiting uptake of norepinephrine and dopamine.24 In one chart review, patients received dextroamphetamine or methylphenidate augmentation of monoamine oxidase inhibitors (MAOIs) alone or with concurrent tricyclic antidepressants; the majority reported decreased depression.25 In an openlabel trial, dextroamphetamine was titrated to efficacy in patients who were receiving an MAOI with or without pemoline.26 Nearly 80% of patients reported long-lasting improvement in depression. In an open-label trial, all patients reported decreased depression when methylphenidate was added to SRIs; however, no scales were used.27
In a case series, patients with both major depression and persistent depressive disorder (dysthymia) experienced a substantial, quick, and sustained response to dextroamphetamine or methylphenidate augmentation.28 Addition of lisdexamfetamine significantly reduced depressive symptoms in individuals with inadequate response to escitalopram.29 Patients with full or partial remission of depression noted improved executive function and residual depressive symptoms after lisdexamfetamine was added to SRI monotherapy.30 In a trial in which patients received dexamphetamine or methylphenidate as monotherapy or augmentation, 30% to 34% of patients reported mood improvement, but 36% reported no improvement.31 In an extension study, low-dose psychostimulants quickly diminished melancholia.32
Methylphenidate was safe and effective in patients with bipolar depression receiving treatment for 1 to 5 years; 44% evidenced significant improvement.33 When offered to patients with bipolar depression, patients receiving methylphenidate or dextroamphetamine reported less depression or sedation and did not develop tolerance, mania, or misuse.34 A case series concluded that methylphenidate addition to mood stabilizers was generally effective and safe.35
However, not all preparations of methylphenidate have demonstrated efficacy. In one study, osmotic controlledrelease oral system (OROS) methylphenidate improved apathy and fatigue but not overall depression.36 Although OROS methylphenidate similarly failed to demonstrate statistically significant efficacy in another study, more responders were documented in the treatment group.37
Although this review focuses on stimulants as augmenting agents in patients with depression, it is worth noting the limited number of studies evaluating stimulants’ effect on depression in patients with traumatic brain injury. This observation is of concern, as these conditions are frequently comorbid in returning veterans. One study noted that methylphenidate was an effective monotherapy for depression; whereas another study found that methylphenidate monotherapy reduced depression as well as sertraline, was better tolerated, and improved fatigue and cognition.38,39
Modafinil and Armodafinil
Modafinil and armodafinil (the R-enantiomer of modafinil) are indicated for improving wakefulness in individuals with narcolepsy, obstructive sleep apnea, and shift work sleep disorder by modulating glutamate, gamma amino-butyric acid, and histamine.40,41 Although they increase extracellular dopamine concentrations, they do not cause an increase in dopamine release and may have less misuse potential than that of dextroamphetamine and methylphenidate.40,41 In a study of 7 patients with unipolar or bipolar depression, all patients achieved full or partial remission with minimal adverse effects (AEs).42 In a prospective study, 41% of patients reported only mild depression or full remission with modafinil augmentation.43
Multiple trials and a pooled analysis noted decreased depression and fatigue and improved cognition in patients receiving modafinil augmentation compared with mood stabilizers or antidepressants.44-49 Modafinil is a useful adjunct for partial responders to SRIs, resulting in rapid mood improvement and decreased fatigue.50-54 However, in one study, modafinil did not demonstrate efficacy compared with placebo. This result was attributed to premature study termination after 2 modafinil-treated patients developed suicidal ideation.55 A post hoc analysis found no difference in frequency of suicidal ideation between groups.
Two DBPC studies evaluated armodafinil in patients with bipolar depression. In both studies it was added to a mood-stabilizing agent (lithium, valproate, aripiprazole, olanzapine, lamotrigine, risperidone, or ziprasidone), and patients receiving armodafinil reported significant reductions
in depression.56,57
Atomoxetine
Atomoxetine is a norepinephrine reuptake inhibitor indicated for the treatment of ADHD and is considered to have no misuse potential due to lack of dopamine modulation.58 In one study, 15 patients received atomoxetine added to their antidepressant, and 60% experienced significant symptom reduction.59 A chart review noted decreases in fatigue and depression when atomoxetine was added to an SRI, mirtazapine, or amitriptyline.60 However, in a DBPC trial, atomoxetine did not lead to significant changes in depression.61
Discussion
There is a limited amount of high-quality evidence to support psychostimulant augmentation, as noted by the relatively few DBPC trials, most of short duration. The evidence supports their efficacy primarily for unipolar depression, as 14 studies evaluated patients with unipolar depression, whereas only 7 studies evaluated patients with bipolar depression. The remaining studies recruited patients with both depression types. Collectively, modafinil and armodafinil have the most evidence in DBPC trials.
There are relatively few DBPC trials with high power and sufficient duration for dextroamphetamine and methylphenidate preparations. This discovery is surprising, considering the duration that these medications have been available. However, several chart reviews and open-label trials provided some evidence to support their use in patients without a history of substance misuse or cardiac conditions.62 Osmotic controlled- release oral system methylphenidate seems to be ineffective, and the efficacy of atomoxetine for augmentation
is uncertain.
Precautions
Prescribing physicians who offer stimulants should consider potential AEs, such as psychosis, anorexia, anxiety, insomnia, mood changes (eg, anger), misuse, addiction, mania, and cardiovascular problems. Psychostimulants have been implicated in precipitating psychosis.63,64 However, in a 12-month study of 250 adults with ADHD, 73 reported AEs, and only 31 discontinued the stimulant. Adverse effects leading to discontinuation included mood instability (n = 7), agitation (n = 6), irritability (n = 4), or decreased appetite (n = 4).65
Although associated with the risks of anorexia and insomnia in patients with ADHD, methylphenidate rapidly improved daytime sleepiness and mood, and—paradoxically—appetite and nighttime sleep in medically ill elderly patients with depression.66 Misuse or abuse of methylphenidate and dextroamphetamine were noted in 23% of patients referred for substance misuse.67 Nonetheless, little evidence exists that these drugs possess significant misuse potential in patients taking them as prescribed. As a prodrug, lisdexamfetamine is hypothesized to have less abuse potential compared with dextroamphetamine and methylphenidate, but it carries the same prescribing and monitoring precautions.68 Risks related to stimulant usage extend to manic symptoms.69 Patients with bipolar disorder should not receive stimulants if they have a history of stimulant-induced mania, rapid cycling, or psychosis.70
Long-term cardiovascular safety data exist for dextroamphetamine and methylphenidate but are limited or unavailable for modafinil, armodafinil, and atomoxetine. A retrospective cohort study found no significant increase in the number of cardiac events in patients receiving dextroamphetamine,
methylphenidate, or atomoxetine for an average of 1 year compared with controls.71 Another cohort study of > 44,000 patients found that initiation of
methylphenidate was associated with increased risk of sudden death or arrhythmia, but the risk was attributed to an unmeasured confounding factor, as the authors found a negative correlation between methylphenidate dose and all cardiovascular events.72
Recent practice guidelines recommend that before prescribing stimulants, clinicians should perform a physical examination (including heart and lung auscultation), obtain vital signs and height and weight, and request an electrocardiogram in case of abnormal findings on a cardiovascular examination or in case of a personal or family history of heart disease. Before offering atomoxetine, clinicians should evaluate the patient for a history of liver disease (and check liver function studies in case of a positive history). Clinicians should also assess risk of self-harm prior to initiating psychostimulant therapy.73 Throughout treatment, clinicians should evaluate the patient for changes in blood pressure, pulse, weight or mood, as well as the development of dependence or misuse. Urine toxicology testing is recommended for dextroamphetamine and methylphenidate to screen for adherence and diversion.
Limitations
Using only PubMed and MEDLINE databases limited the search to articles published in English after 1985, excluding letters and case reports to identify studies with higher evidence (the studies were not weighted based on study design). In addition, the studies had certain limitations. These include a limited number of DBPC trials, most were of short duration. It is also difficult to compare studies due to various rating scales used and concurrent
medication regimens of study subjects. These limitations raise questions surrounding the long-term efficacy of stimulants, and there is no consensus for how long a stimulant should be continued if beneficial. Longer, higherpowered, DBPC trials are warranted to determine longterm efficacy and safety of stimulant augmentation.62
Conclusion
For patients with depression who have not responded to other augmentation strategies, psychostimulants may be offered to improve mood, energy, and concentration. For clinicians considering stimulant augmentation, modafinil and armodafinil are reasonable choices given their efficacy in double-blind, placebo-controlled trials and lower risk of misuse. Dextroamphetamine (particularly lisdexamphetamine) and methylphenidate may be appropriate for patients who have not benefited from or tolerated modafinil or armodafinil, provided these patients do not have a medical history of cardiac disease or current substance use.
Osmotic controlled-release oral system methylphenidate seems to be ineffective as an augmenting agent. The efficacy of atomoxetine for augmentation is questionable, but atomoxetine could be offered if other stimulants were contraindicated, ineffective, or poorly tolerated. Both OROS methylphenidate and atomoxetine should be evaluated in additional trials before they can be recommended as augmentation therapies. Certain psychostimulants may be appropriate and reasonable adjunctive pharmacotherapies for patients with unipolar or bipolar depression who have failed other augmentation strategies, for patients who have significant fatigue or cognitive complaints, or for elderly patients with melancholic or somatic features of depression.
Acknowledgements
The authors thank Maureen Humphrey-Shelton and Kathy Thomas for their help in obtaining references.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Depression is a common condition that significantly impairs social and occupational functioning. Many patients do not respond to first-line pharmacotherapies and are considered to have treatment-resistant depression (TRD). These patients may benefit from augmentation of their antidepressant to reduce depression. Multiple medications have demonstrated various degrees of efficacy for augmentation, including psychostimulants. This article reviews studies of psychostimulants as augmentation agents for TRD and discusses risks, offers advice, and makes recommendations for clinicians who prescribe stimulants.
Background
Major depressive disorder (MDD) is a common psychiatric condition that significantly impairs quality of life.1 It is a recurrent illness, averaging 2 relapses per decade. The probability of recurrence increases with the number of depressive episodes.2,3 A patient who experiences major depressive episodes alternating with euthymia has unipolar depression; whereas one who experiences major depressive episodes alternating with episodes of mania or hypomania has bipolar depression.4
Despite adequate dose and duration of pharmacotherapy, many individuals with unipolar or bipolar depression do not achieve and sustain remission.5 Remission rates decrease and relapse rates increase with subsequent failed antidepressant trials.6 It is difficult to identify factors that predict treatment resistance, but one review of antidepressant studies found that patients who did not demonstrate a response within 3 weeks of medication initiation were less likely to respond after a longer duration.7
Treatment-resistant depression is commonly, but not universally, defined as lack of response after trials of 2 or more antidepressants with different mechanisms of action for sufficient duration.5 This definition will be used here as well. Other definitions have proposed stages of TRD, but these require further study to evaluate their reliability and predictive utility.8 Due to lack of consensus regarding the definition of TRD, it is not possible to determine the exact prevalence of TRD.
Patients with TRD may benefit from augmentation of their medication regimen. Augmentation with lithium has yielded conflicting results, and its efficacy with newer antidepressants is not well studied.9-12 Triiodothyronine, buspirone, and pindolol have demonstrated some efficacy when added to serotonin reuptake inhibitors (SRIs).10,12,13 Second-generation antipsychotic drugs, antidepressant drug combinations, omega-3 fatty acids, S-adenosyl methionine (SAMe), and L-methylfolate have demonstrated some efficacy in some studies as well.12,14-23 In patients with depression who have not responded to these strategies, psychostimulant augmentation may be appropriate.
Methods
A literature search was conducted following an algorithmic approach in the MEDLINE/PubMed database for studies in English from January 1985 to August 2014 of stimulants as augmenting agents for depression, using the Medical Subject Headings stimulant, depression, and augmentation, combined with an AND operator. The search was limited to adult humans and excluded case reports and letters, to identify studies with stronger evidence. Also excluded were studies using caffeine (to augment electroconvulsive therapy for depression) and pemoline as the sole augmenting stimulant as well as studies of patients with comorbid mental health diagnoses and studies that initiated stimulants and antidepressants simultaneously to assess antidepressant response.
This review organized results by stimulant rather than by depression type, even though some studies used > 1 stimulant or recruited patients with different types of depression. Although prevalence, prognosis, and monotherapy differ for unipolar and bipolar depression, psychostimulants target similar symptoms, despite augmenting different monotherapies in unipolar and bipolar depression. Therefore, no distinction is made between assessing studies of stimulants for unipolar and bipolar depression.
Results
A total of 70 articles were identified, and 31 studies met inclusion criteria (Figure). Of the studies included, 12 were double-blind, placebo-controlled (DBPC) trials and 19 were retrospective chart reviews or open studies. Most studies evaluated depression, using validated scales, such as the Hamilton Depression Rating Scale, Montgomery-Asberg Depression Rating Scale, Clinical Global Impressions of Severity, Inventory of Depressive Symptoms, Carroll Depression Rating Scale, Global Assessment of Functioning, Quick Inventory of Depressive Symptomatology, or the Psychiatric Symptom Assessment Scale. Study details are provided in Tables 1 to 4.
Dextroamphetamine and Methylphenidate
Dextroamphetamine and methylphenidate are indicated for the treatment of attention-deficit/hyperactivity disorder (ADHD) and exert their effects by inhibiting uptake of norepinephrine and dopamine.24 In one chart review, patients received dextroamphetamine or methylphenidate augmentation of monoamine oxidase inhibitors (MAOIs) alone or with concurrent tricyclic antidepressants; the majority reported decreased depression.25 In an openlabel trial, dextroamphetamine was titrated to efficacy in patients who were receiving an MAOI with or without pemoline.26 Nearly 80% of patients reported long-lasting improvement in depression. In an open-label trial, all patients reported decreased depression when methylphenidate was added to SRIs; however, no scales were used.27
In a case series, patients with both major depression and persistent depressive disorder (dysthymia) experienced a substantial, quick, and sustained response to dextroamphetamine or methylphenidate augmentation.28 Addition of lisdexamfetamine significantly reduced depressive symptoms in individuals with inadequate response to escitalopram.29 Patients with full or partial remission of depression noted improved executive function and residual depressive symptoms after lisdexamfetamine was added to SRI monotherapy.30 In a trial in which patients received dexamphetamine or methylphenidate as monotherapy or augmentation, 30% to 34% of patients reported mood improvement, but 36% reported no improvement.31 In an extension study, low-dose psychostimulants quickly diminished melancholia.32
Methylphenidate was safe and effective in patients with bipolar depression receiving treatment for 1 to 5 years; 44% evidenced significant improvement.33 When offered to patients with bipolar depression, patients receiving methylphenidate or dextroamphetamine reported less depression or sedation and did not develop tolerance, mania, or misuse.34 A case series concluded that methylphenidate addition to mood stabilizers was generally effective and safe.35
However, not all preparations of methylphenidate have demonstrated efficacy. In one study, osmotic controlledrelease oral system (OROS) methylphenidate improved apathy and fatigue but not overall depression.36 Although OROS methylphenidate similarly failed to demonstrate statistically significant efficacy in another study, more responders were documented in the treatment group.37
Although this review focuses on stimulants as augmenting agents in patients with depression, it is worth noting the limited number of studies evaluating stimulants’ effect on depression in patients with traumatic brain injury. This observation is of concern, as these conditions are frequently comorbid in returning veterans. One study noted that methylphenidate was an effective monotherapy for depression; whereas another study found that methylphenidate monotherapy reduced depression as well as sertraline, was better tolerated, and improved fatigue and cognition.38,39
Modafinil and Armodafinil
Modafinil and armodafinil (the R-enantiomer of modafinil) are indicated for improving wakefulness in individuals with narcolepsy, obstructive sleep apnea, and shift work sleep disorder by modulating glutamate, gamma amino-butyric acid, and histamine.40,41 Although they increase extracellular dopamine concentrations, they do not cause an increase in dopamine release and may have less misuse potential than that of dextroamphetamine and methylphenidate.40,41 In a study of 7 patients with unipolar or bipolar depression, all patients achieved full or partial remission with minimal adverse effects (AEs).42 In a prospective study, 41% of patients reported only mild depression or full remission with modafinil augmentation.43
Multiple trials and a pooled analysis noted decreased depression and fatigue and improved cognition in patients receiving modafinil augmentation compared with mood stabilizers or antidepressants.44-49 Modafinil is a useful adjunct for partial responders to SRIs, resulting in rapid mood improvement and decreased fatigue.50-54 However, in one study, modafinil did not demonstrate efficacy compared with placebo. This result was attributed to premature study termination after 2 modafinil-treated patients developed suicidal ideation.55 A post hoc analysis found no difference in frequency of suicidal ideation between groups.
Two DBPC studies evaluated armodafinil in patients with bipolar depression. In both studies it was added to a mood-stabilizing agent (lithium, valproate, aripiprazole, olanzapine, lamotrigine, risperidone, or ziprasidone), and patients receiving armodafinil reported significant reductions
in depression.56,57
Atomoxetine
Atomoxetine is a norepinephrine reuptake inhibitor indicated for the treatment of ADHD and is considered to have no misuse potential due to lack of dopamine modulation.58 In one study, 15 patients received atomoxetine added to their antidepressant, and 60% experienced significant symptom reduction.59 A chart review noted decreases in fatigue and depression when atomoxetine was added to an SRI, mirtazapine, or amitriptyline.60 However, in a DBPC trial, atomoxetine did not lead to significant changes in depression.61
Discussion
There is a limited amount of high-quality evidence to support psychostimulant augmentation, as noted by the relatively few DBPC trials, most of short duration. The evidence supports their efficacy primarily for unipolar depression, as 14 studies evaluated patients with unipolar depression, whereas only 7 studies evaluated patients with bipolar depression. The remaining studies recruited patients with both depression types. Collectively, modafinil and armodafinil have the most evidence in DBPC trials.
There are relatively few DBPC trials with high power and sufficient duration for dextroamphetamine and methylphenidate preparations. This discovery is surprising, considering the duration that these medications have been available. However, several chart reviews and open-label trials provided some evidence to support their use in patients without a history of substance misuse or cardiac conditions.62 Osmotic controlled- release oral system methylphenidate seems to be ineffective, and the efficacy of atomoxetine for augmentation
is uncertain.
Precautions
Prescribing physicians who offer stimulants should consider potential AEs, such as psychosis, anorexia, anxiety, insomnia, mood changes (eg, anger), misuse, addiction, mania, and cardiovascular problems. Psychostimulants have been implicated in precipitating psychosis.63,64 However, in a 12-month study of 250 adults with ADHD, 73 reported AEs, and only 31 discontinued the stimulant. Adverse effects leading to discontinuation included mood instability (n = 7), agitation (n = 6), irritability (n = 4), or decreased appetite (n = 4).65
Although associated with the risks of anorexia and insomnia in patients with ADHD, methylphenidate rapidly improved daytime sleepiness and mood, and—paradoxically—appetite and nighttime sleep in medically ill elderly patients with depression.66 Misuse or abuse of methylphenidate and dextroamphetamine were noted in 23% of patients referred for substance misuse.67 Nonetheless, little evidence exists that these drugs possess significant misuse potential in patients taking them as prescribed. As a prodrug, lisdexamfetamine is hypothesized to have less abuse potential compared with dextroamphetamine and methylphenidate, but it carries the same prescribing and monitoring precautions.68 Risks related to stimulant usage extend to manic symptoms.69 Patients with bipolar disorder should not receive stimulants if they have a history of stimulant-induced mania, rapid cycling, or psychosis.70
Long-term cardiovascular safety data exist for dextroamphetamine and methylphenidate but are limited or unavailable for modafinil, armodafinil, and atomoxetine. A retrospective cohort study found no significant increase in the number of cardiac events in patients receiving dextroamphetamine,
methylphenidate, or atomoxetine for an average of 1 year compared with controls.71 Another cohort study of > 44,000 patients found that initiation of
methylphenidate was associated with increased risk of sudden death or arrhythmia, but the risk was attributed to an unmeasured confounding factor, as the authors found a negative correlation between methylphenidate dose and all cardiovascular events.72
Recent practice guidelines recommend that before prescribing stimulants, clinicians should perform a physical examination (including heart and lung auscultation), obtain vital signs and height and weight, and request an electrocardiogram in case of abnormal findings on a cardiovascular examination or in case of a personal or family history of heart disease. Before offering atomoxetine, clinicians should evaluate the patient for a history of liver disease (and check liver function studies in case of a positive history). Clinicians should also assess risk of self-harm prior to initiating psychostimulant therapy.73 Throughout treatment, clinicians should evaluate the patient for changes in blood pressure, pulse, weight or mood, as well as the development of dependence or misuse. Urine toxicology testing is recommended for dextroamphetamine and methylphenidate to screen for adherence and diversion.
Limitations
Using only PubMed and MEDLINE databases limited the search to articles published in English after 1985, excluding letters and case reports to identify studies with higher evidence (the studies were not weighted based on study design). In addition, the studies had certain limitations. These include a limited number of DBPC trials, most were of short duration. It is also difficult to compare studies due to various rating scales used and concurrent
medication regimens of study subjects. These limitations raise questions surrounding the long-term efficacy of stimulants, and there is no consensus for how long a stimulant should be continued if beneficial. Longer, higherpowered, DBPC trials are warranted to determine longterm efficacy and safety of stimulant augmentation.62
Conclusion
For patients with depression who have not responded to other augmentation strategies, psychostimulants may be offered to improve mood, energy, and concentration. For clinicians considering stimulant augmentation, modafinil and armodafinil are reasonable choices given their efficacy in double-blind, placebo-controlled trials and lower risk of misuse. Dextroamphetamine (particularly lisdexamphetamine) and methylphenidate may be appropriate for patients who have not benefited from or tolerated modafinil or armodafinil, provided these patients do not have a medical history of cardiac disease or current substance use.
Osmotic controlled-release oral system methylphenidate seems to be ineffective as an augmenting agent. The efficacy of atomoxetine for augmentation is questionable, but atomoxetine could be offered if other stimulants were contraindicated, ineffective, or poorly tolerated. Both OROS methylphenidate and atomoxetine should be evaluated in additional trials before they can be recommended as augmentation therapies. Certain psychostimulants may be appropriate and reasonable adjunctive pharmacotherapies for patients with unipolar or bipolar depression who have failed other augmentation strategies, for patients who have significant fatigue or cognitive complaints, or for elderly patients with melancholic or somatic features of depression.
Acknowledgements
The authors thank Maureen Humphrey-Shelton and Kathy Thomas for their help in obtaining references.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Kessler RC, Berglund P, Demler O, Jin R, Merikangas KR, Walters EE. Lifetime prevalence and age-of-onset distribution of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):593-602.
2. Solomon DA, Keller MB, Leon AC, et al. Multiple recurrences of major depressive disorder. Am J Psychiatry. 2000;157(2):229-233.
3. Katon WJ, Fan MY, Lin EH, Unützer J. Depressive symptom deterioration in a large
primary care-based elderly cohort. Am J Geriatr Psychiatry. 2006;14(3):246-254.
4. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington, VA: American Psychiatric Association; 2013.
5. McIntyre RS, Filteau M-J, Martin L, et al. Treatment-resistant depression: Definitions, review of the evidence, and algorithmic approach. J Affect Disord. 2014;156:1-7.
6. Gaynes BN, Warden D, Trivedi MH, Wisniewski SR, Fava M, Rush AJ. What did STAR*D teach us? Results from a large-scale, practical, clinical trial for patients with depression. Focus. 2012;10(4):510-517.
7. Kudlow PA, Cha DS, McIntyre RS. Predicting treatment response in major depressive disorder: The impact of early symptomatic improvement. Can J Psychiatry. 2012;57(12):782-788.
8. Ruhé HG, van Rooijen G, Spijker J, Peeters FP, Schene AH. Staging methods for treatment resistant depression. A systematic review. J Affect Disord. 2012;137(1-3):35-45.
9. Bauer M, Dopfmer S. Lithium augmentation treatment-resistant depression: Metaanalysis of placebo-controlled studies. J Clin Psychopharmacol. 1999;19(5):427-434.
10. Nierenberg AA, Fava M, Trivedi MH, et al. A comparison of lithium and T(3) augmentation following two failed medication treatments for depression: A STAR*D report. Am J Psychiatry. 2006;163(9):1519-1530.
11. Nierenberg AA, Papakostas GI, Petersen T, et al. Lithium augmentation of nortriptyline
for subjects resistant to multiple antidepressants. J Clin Psychopharmacol. 2003;23(1):92-95.
12. Connolly KR, Thase ME. If at first you don’t succeed: A review of the evidence for antidepressant augmentation, combination, and switching strategies. Drugs. 2011;71(1):43-64.
13. Trivedi MH, Fava M, Wisniewski SR, et al; STAR*D Study Team. Medication augmentation after the failure of SSRIs for depression. N Engl J Med. 2006;354(12):1243-1252.
14. Papakostas GI, Shelton RC, Smith J, Fava M. Augmentation of antidepressants with atypical antipsychotic medications for treatment resistant major depressive disorder: A meta-analysis. J Clin Psychiatry. 2007;68(6):826-831.
15. Mahmoud RA, Pandina GJ, Turkoz I, et al. Risperidone for treatment-refractory major depressive disorder: A randomized trial. Ann Intern Med. 2007;147(9):593-602.
16. Barbee JG, Conrad EJ, Jamhour NJ. The effectiveness of olanzapine, risperidone, quetiapine, and ziprasidone as augmentation agents in treatment resistant depressive disorder. J Clin Psychiatry. 2004;65(7):975-981.
17. Fatemi SH, Emamian ES, Kist DA. Venlafaxine and bupropion combination therapy in a case of treatment-resistant depression. Ann Pharmacother.1999;33(6):701-703.
18. Carpenter LL, Yasman S, Price LH. A double-blind, placebo-controlled study of antidepressant augmentation with mirtazapine. Biol Psychiatry. 2002;51(2):183-188.
19. Hannan N, Hamzah Z, Akinpeloye HO, Meagher D. Venlafaxine-mirtazapine combination therapy in the treatment of persistent depressive illness. J Psychopharmacol. 2007;21(2):161-164.
20. McGrath PJ, Stewart JW, Fava M, et al. Tranylcypromine versus venlafaxine plus mirtazapine following three failed antidepressant medication trials for depression: A STAR*D report. Am J Psychiatry. 2006;163(9):1531-1541.
21. Blier P, Ward HE, Tremblay P, Laberge L, Hébert C, Bergeron R. Combination of antidepressant medications from treatment initiation for major depressive disorder: A double-blind randomized study. Am J Psychiatry. 2010;167(3):281-288.
22. Papakostas GI, Mischoulon D, Shyu I, Alpert JE, Fava M. S-adenosyl methionine (SAMe) augmentation of serotonin reuptake inhibitors for antidepressant nonresponders with major depressive disorder: A double blind randomized clinical trial. Am J Psychiatry. 2010;167(8):942-948.
23. Papakostas GI, Shelton RC, Zajecka JM, et al. L-methylfolate as adjunctive therapy
for SSRI-resistant major depression: Results of two randomized, double-blind,
parallel-sequential trials. Am J Psychiatry. 2012;169(12):1267-1274.
24. Korston TR. Drugs of abuse. In: Katzung BG, ed. Basic and Clinical Pharmacology. 9th ed. New York, NY: McGraw-Hill; 2004:521-523.
25. Feighner JP, Herbstein J, Damlouji N. Combined MAOI, TCA, and direct stimulant therapy of treatment-resistant depression. J Clin Psychiatry. 1985;46(6):206-209.
26. Fawcett J, Kravitz HM, Zajecka JM, Schaff MR. CNS stimulant potentiation of monoamine oxidase inhibitors in treatment-refractory depression. J Clin Psychopharmacol. 1991;11(2):127-132.
27. Stoll AL, Pillay SS, Diamond L, Workum SB, Cole JO. Methylphenidate augmentation of serotonin selective reuptake inhibitors: A case series. J Clin Psychiatry. 1996;57(2):72-76.
28. Masand PS, Anand VS, Tanquary JF. Psychostimulant augmentation of second generation antidepressants: A case series. Depress Anxiety. 1998;7(2):89-91.
29. Trivedi MH, Cutler AJ, Richards C, et al. A randomized control trial of the efficacy and safety of lisdesxamfetamine dimesylate as augmentation therapy in adults with residual symptoms of major depressive disorder after treatment with escitalopram. J Clin Psychiatry. 2013;74(8):802-809.
30. Madhoo M, Keefe RS, Roth RM, et al. Lisdexamfetamine dimesylate augmentation in adults with persistent executive dysfunction after partial or full remission of major depressive disorder. Neuropsychopharmacology. 2014;39(6):1388-1398.
31. Parker G, Brotchie H. Do the old psychostimulant drugs have a role in managing treatment-resistant depression. Acta Psychiatr Scand. 2010;121(4):308-314.
32. Parker G, Brotchie H, McClure G, Fletcher K. Psychostimulants for managing unipolar and bipolar treatment-resistant melancholic depression: A medium term evaluation of cost benefits. J Affect Disord. 2013;151(1):360-364.
33. Lydon E, El-Mallakh RS. Naturalistic long-term use of methylphenidate in bipolar disorder. J Clin Psychopharmacol. 2006;26(5):516-518.
34. Carlson PJ, Merlock MC, Suppes T. Adjunctive stimulant use in patients with bipolar disorder: Treatment of residual depression and sedation. Bipolar Disord. 2004;6(5):416-420.
35. El-Mallakh RS. An open study of methylphenidate in bipolar depression. Bipolar Disord. 2000;2(1):56-59.
36. Ravindran AV, Kennedy SH, O’Donovan MC, Fallu A, Camacho F, Binder CE. Osmotic-release oral system methylphenidate augmentation of antidepressant monotherapy in major depressive disorder: Results of a double-blind, randomized, placebo-controlled trial. J Clin Psychiatry. 2008;69(1):87-94.
37. Patkar AA, Masand PS, Pae CU, et al. A randomized, double-blind, placebocontrolled
trial of augmentation with an extended release formulation of methylphenidate in outpatients with treatment-resistant depression. J Clin Psychopharmacol. 2006;26(6):653-656.
38. Lee H, Kim SW, Kim JM, Shin IS, Yang SJ, Yoon JS Comparing effects of methylphenidate, sertraline, and placebo on neuropsychiatric sequelae in patients with
traumatic brain injury. Hum Psychopharmacol. 2005;20(2):97-104.
39. Gualtieri CT, Evans RW. Stimulant treatment for the neurobehavioural sequelae of traumatic brain injury. Brain Inj. 1988;2(4):273-290.
40. Provigil [package insert]. North Wales, PA: Cephalon Inc; 2015.
41. Nuvigil [package insert]. Frazer, PA: Cephalon, Inc; 2013.
42. Menza MA, Kaufman KR, Castellanos A. Modafinil augmentation of antidepressant treatment in depression. J Clin Psychiatry. 2000;61(5):378-381.
43. Markovitz PJ, Wagner S. An open-label trial of modafinil augmentation in patients with partial response to antidepressant therapy. J Clin Psychopharmacol. 2003;23(2):207-209.
44. Fernandes PP, Petty F. Modafinil for remitted bipolar depression with hypersomnia. Ann Pharmacother. 2003;37(12):1807-1809.
45. Nasr S. Modafinil as adjunctive therapy in depressed outpatients. Ann Clin Psychiatry. 2004;16(3):133-138.
46. DeBattista C, Lembke A, Solvason HB, Ghebremichael R, Poirier J. A prospective trial of modafinil as an adjunctive treatment of major depression. J Clin Psychopharmacol. 2004;24(1):87-90.
47. Nasr S, Wendt B, Steiner K. Absence of mood switch with and tolerance to modafinil: A replication study from a large private practice. J Affect Disord. 2006;95(1-3):111-114.
48. DeBattista C, Doghramji K, Menza MA, Rosenthal MH, Fieve RR; Modafinil in Depression Study Group. Adjunct modafinil for the short-term treatment of fatigue and sleepiness in patients with major depressive disorder: A preliminary doubleblind, placebo-controlled study. J Clin Psychiatry. 2003;64(9):1057-1064.
49. Frye MA, Grunze H, Suppes T, et al. A placebo-controlled evaluation of adjunctive modafinil in the treatment of bipolar depression. Am J Psychiatry. 2007;164(8):1242-1249.
50. Fava M, Thase ME, DeBattista C, Doghramji K, Arora S, Hughes RJ. Modafinil augmentation of selective serotonin reuptake inhibitor therapy in MDD partial responders with persistent fatigue and sleepiness. Ann Clin Psychiatry. 2007;19(3):153-159.
51. Thase ME, Fava M, DeBattista C, Arora S, Hughes RJ. Modafinil augmentation of SSRI therapy in patients with major depressive disorder and excessive sleepiness and fatigue: A 12-week, open-label, extension study. CNS Spectr. 2006;11(2):93-102.
52. Fava M, Thase ME, DeBattista C. A multicenter, placebo-controlled study of modafinil augmentation in partial responders to selective serotonin reuptake inhibitors with persistent fatigue and sleepiness. J Clin Psychiatry. 2005;66(1):85-93.
53. Abolfazli R, Hosseini M, Ghanizadeh A, et al. Double-blind randomized parallelgroup clinical trial of efficacy of the combination fluoxetine plus modafinil versus fluoxetine plus placebo in the treatment of major depression. Depress Anxiety. 2011;28(4):297-302.
54. Rasmussen NA, Schrøder P, Olsen LR, Brødsgaard M, Undén M, Bech P. Modafinil augmentation in depressed patients with partial response to antidepressants: A pilot study on self-reported symptoms covered by the Major Depression Inventory (MDI) and the Symptom Checklist (SCL-92). Nord J Psychiatry. 2005;59(3):173-178.
55. Dunlop BW, Crits-Christoph P, Evans DL, et al. Coadministration of modafinil and a selective serotonin reuptake inhibitor from the initiation of treatment of major depressive disorder with fatigue and sleepiness: A double-blind, placebocontrolled study. J Clin Psychopharmacol. 2007;27(6):614-619.
56. Calabrese JR, Ketter TA, Youakim JM, Tiller JM, Yang R, Frye MA. Adjunctive armodafinil
for major depressive episodes associated with bipolar I disorder: A randomized multicenter, double-blind, placebo-controlled, proof-of-concept study. J Clin Psychiatry. 2010;71(10):1363-1370.
57. Calabrese JR, Frye MA, Yang R, Ketter TA; Armodafinil Treatment Trial Study Network. Efficacy and safety of adjunctive armodafinil in adults with major depressive episodes associated with bipolar I disorder: A randomized, double-blind, placebo-controlled, multicenter trial. J Clin Psychiatry. 2014;75(10):1054-1061.
58. Strattera [package insert]. Indianapolis, IN. Lilly; 2015.
59. Carpenter LL, Milosavljevic N, Schecter JM, Tyrka AR, Price LH. Augmentation with open-label atomoxetine for partial or nonresponse to antidepressants. J Clin Psychiatry. 2005;66(10):1234-1238.
60. Papakostas GI, Petersen TJ, Burns AM, Fava M. Adjunctive atomoxetine for residual
fatigue in major depressive disorder. J Psychiatr Res. 2006;40(4):370-373.
61. Michelson D, Adler LA, Amsterdam JD, et al. Addition of atomoxetine for depression
incompletely responsive to sertraline: A randomized, double-blind, placebocontrolled study. J Clin Psychiatry. 2007;68(4):582-587.
62. Corp SA, Gitlin MJ, Altshuler LL. A review of the use of stimulants and stimulant alternatives in treating bipolar depression and major depressive disorder. J Clin Psychiatry. 2014;75(9):1010-1018.
63. Kraemer M, Uekermann J, Wiltfang J, Kis B. Methylphenidate-induced psychosis in adult attention-deficit/hyperactivity disorder: Report of 3 new cases and review of the literature. Clin Neuropharmacol. 2010;33(4):204-206.
64. Berman SM, Kuczenski R, McCracken JT, London ED. Potential adverse effects of amphetamine treatment on brain and behavior: A review. Mol Psychiatry. 2009;14(2):123-142.
65. Fredriksen M, Dahl AA, Martinsen EW, Klungsøyr O, Haavik J, Peleikis DE. Effectiveness of one-year pharmacological treatment of adult attention-deficit/hyperactivity disorder (ADHD): An open-label prospective study of time in treatment, dose, side-effects and comorbidity. Eur Neuropsychopharmacol. 2014;24(12):1873-1874.
66. Hardy SE. Methylphenidate for the treatment of depressive symptoms, including fatigue and apathy, in medically ill older adults and terminally ill adults. Am J Geriatr Pharmacother. 2009;7(1):34-59.
67. Williams RJ, Goodale LA, Shay-Fiddler MA, Gloster SP, Chang SY. Methylphenidate and dextroamphetamine abuse in substance-abusing adolescents. Am J Addict. 2004;13(4):381-389.
68. Madaan V, Kolli V, Bestha DP, Shah MJ. Update on optimal use of lisdexamfetamine in the treatment of ADHD. Neuropsychiatr Dis Treat. 2013;9:977-983.
69. Ross RG. Psychotic and manic-like symptoms during stimulant treatment of attention deficit hyperactivity disorder. Am J Psychiatry. 2006;163(7):1149-1152.
70. Dell’Osso B, Ketter TA. Use of adjunctive stimulants in adult bipolar depression. Int J Neuropsychopharmacol. 2013;16(1):55-68.
71. Habel LA, Cooper WO, Sox CM, et al. ADHD medications and risk of serious cardiovascular events in young and middle-aged adults. JAMA. 2011;306(24):2673-2683.
72. Schelleman H, Bilker WB, Kimmel SE, et al. Methylphenidate and risk of serious cardiovascular events in adults. Am J Psychiatry. 2012;169(2):178-185.
73. Bolea-Alamañac B, Nutt DJ, Adamou M, et al; British Association for Psychopharmacology. Evidence-based guidelines for the pharmacological management of attention deficit hyperactivity disorder: Update on recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2014;28(3):179-203.
74. Moher D, Liberati A, Tetzlaff J, Altman DG; The PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009;6(6):e1000097.
1. Kessler RC, Berglund P, Demler O, Jin R, Merikangas KR, Walters EE. Lifetime prevalence and age-of-onset distribution of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):593-602.
2. Solomon DA, Keller MB, Leon AC, et al. Multiple recurrences of major depressive disorder. Am J Psychiatry. 2000;157(2):229-233.
3. Katon WJ, Fan MY, Lin EH, Unützer J. Depressive symptom deterioration in a large
primary care-based elderly cohort. Am J Geriatr Psychiatry. 2006;14(3):246-254.
4. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Arlington, VA: American Psychiatric Association; 2013.
5. McIntyre RS, Filteau M-J, Martin L, et al. Treatment-resistant depression: Definitions, review of the evidence, and algorithmic approach. J Affect Disord. 2014;156:1-7.
6. Gaynes BN, Warden D, Trivedi MH, Wisniewski SR, Fava M, Rush AJ. What did STAR*D teach us? Results from a large-scale, practical, clinical trial for patients with depression. Focus. 2012;10(4):510-517.
7. Kudlow PA, Cha DS, McIntyre RS. Predicting treatment response in major depressive disorder: The impact of early symptomatic improvement. Can J Psychiatry. 2012;57(12):782-788.
8. Ruhé HG, van Rooijen G, Spijker J, Peeters FP, Schene AH. Staging methods for treatment resistant depression. A systematic review. J Affect Disord. 2012;137(1-3):35-45.
9. Bauer M, Dopfmer S. Lithium augmentation treatment-resistant depression: Metaanalysis of placebo-controlled studies. J Clin Psychopharmacol. 1999;19(5):427-434.
10. Nierenberg AA, Fava M, Trivedi MH, et al. A comparison of lithium and T(3) augmentation following two failed medication treatments for depression: A STAR*D report. Am J Psychiatry. 2006;163(9):1519-1530.
11. Nierenberg AA, Papakostas GI, Petersen T, et al. Lithium augmentation of nortriptyline
for subjects resistant to multiple antidepressants. J Clin Psychopharmacol. 2003;23(1):92-95.
12. Connolly KR, Thase ME. If at first you don’t succeed: A review of the evidence for antidepressant augmentation, combination, and switching strategies. Drugs. 2011;71(1):43-64.
13. Trivedi MH, Fava M, Wisniewski SR, et al; STAR*D Study Team. Medication augmentation after the failure of SSRIs for depression. N Engl J Med. 2006;354(12):1243-1252.
14. Papakostas GI, Shelton RC, Smith J, Fava M. Augmentation of antidepressants with atypical antipsychotic medications for treatment resistant major depressive disorder: A meta-analysis. J Clin Psychiatry. 2007;68(6):826-831.
15. Mahmoud RA, Pandina GJ, Turkoz I, et al. Risperidone for treatment-refractory major depressive disorder: A randomized trial. Ann Intern Med. 2007;147(9):593-602.
16. Barbee JG, Conrad EJ, Jamhour NJ. The effectiveness of olanzapine, risperidone, quetiapine, and ziprasidone as augmentation agents in treatment resistant depressive disorder. J Clin Psychiatry. 2004;65(7):975-981.
17. Fatemi SH, Emamian ES, Kist DA. Venlafaxine and bupropion combination therapy in a case of treatment-resistant depression. Ann Pharmacother.1999;33(6):701-703.
18. Carpenter LL, Yasman S, Price LH. A double-blind, placebo-controlled study of antidepressant augmentation with mirtazapine. Biol Psychiatry. 2002;51(2):183-188.
19. Hannan N, Hamzah Z, Akinpeloye HO, Meagher D. Venlafaxine-mirtazapine combination therapy in the treatment of persistent depressive illness. J Psychopharmacol. 2007;21(2):161-164.
20. McGrath PJ, Stewart JW, Fava M, et al. Tranylcypromine versus venlafaxine plus mirtazapine following three failed antidepressant medication trials for depression: A STAR*D report. Am J Psychiatry. 2006;163(9):1531-1541.
21. Blier P, Ward HE, Tremblay P, Laberge L, Hébert C, Bergeron R. Combination of antidepressant medications from treatment initiation for major depressive disorder: A double-blind randomized study. Am J Psychiatry. 2010;167(3):281-288.
22. Papakostas GI, Mischoulon D, Shyu I, Alpert JE, Fava M. S-adenosyl methionine (SAMe) augmentation of serotonin reuptake inhibitors for antidepressant nonresponders with major depressive disorder: A double blind randomized clinical trial. Am J Psychiatry. 2010;167(8):942-948.
23. Papakostas GI, Shelton RC, Zajecka JM, et al. L-methylfolate as adjunctive therapy
for SSRI-resistant major depression: Results of two randomized, double-blind,
parallel-sequential trials. Am J Psychiatry. 2012;169(12):1267-1274.
24. Korston TR. Drugs of abuse. In: Katzung BG, ed. Basic and Clinical Pharmacology. 9th ed. New York, NY: McGraw-Hill; 2004:521-523.
25. Feighner JP, Herbstein J, Damlouji N. Combined MAOI, TCA, and direct stimulant therapy of treatment-resistant depression. J Clin Psychiatry. 1985;46(6):206-209.
26. Fawcett J, Kravitz HM, Zajecka JM, Schaff MR. CNS stimulant potentiation of monoamine oxidase inhibitors in treatment-refractory depression. J Clin Psychopharmacol. 1991;11(2):127-132.
27. Stoll AL, Pillay SS, Diamond L, Workum SB, Cole JO. Methylphenidate augmentation of serotonin selective reuptake inhibitors: A case series. J Clin Psychiatry. 1996;57(2):72-76.
28. Masand PS, Anand VS, Tanquary JF. Psychostimulant augmentation of second generation antidepressants: A case series. Depress Anxiety. 1998;7(2):89-91.
29. Trivedi MH, Cutler AJ, Richards C, et al. A randomized control trial of the efficacy and safety of lisdesxamfetamine dimesylate as augmentation therapy in adults with residual symptoms of major depressive disorder after treatment with escitalopram. J Clin Psychiatry. 2013;74(8):802-809.
30. Madhoo M, Keefe RS, Roth RM, et al. Lisdexamfetamine dimesylate augmentation in adults with persistent executive dysfunction after partial or full remission of major depressive disorder. Neuropsychopharmacology. 2014;39(6):1388-1398.
31. Parker G, Brotchie H. Do the old psychostimulant drugs have a role in managing treatment-resistant depression. Acta Psychiatr Scand. 2010;121(4):308-314.
32. Parker G, Brotchie H, McClure G, Fletcher K. Psychostimulants for managing unipolar and bipolar treatment-resistant melancholic depression: A medium term evaluation of cost benefits. J Affect Disord. 2013;151(1):360-364.
33. Lydon E, El-Mallakh RS. Naturalistic long-term use of methylphenidate in bipolar disorder. J Clin Psychopharmacol. 2006;26(5):516-518.
34. Carlson PJ, Merlock MC, Suppes T. Adjunctive stimulant use in patients with bipolar disorder: Treatment of residual depression and sedation. Bipolar Disord. 2004;6(5):416-420.
35. El-Mallakh RS. An open study of methylphenidate in bipolar depression. Bipolar Disord. 2000;2(1):56-59.
36. Ravindran AV, Kennedy SH, O’Donovan MC, Fallu A, Camacho F, Binder CE. Osmotic-release oral system methylphenidate augmentation of antidepressant monotherapy in major depressive disorder: Results of a double-blind, randomized, placebo-controlled trial. J Clin Psychiatry. 2008;69(1):87-94.
37. Patkar AA, Masand PS, Pae CU, et al. A randomized, double-blind, placebocontrolled
trial of augmentation with an extended release formulation of methylphenidate in outpatients with treatment-resistant depression. J Clin Psychopharmacol. 2006;26(6):653-656.
38. Lee H, Kim SW, Kim JM, Shin IS, Yang SJ, Yoon JS Comparing effects of methylphenidate, sertraline, and placebo on neuropsychiatric sequelae in patients with
traumatic brain injury. Hum Psychopharmacol. 2005;20(2):97-104.
39. Gualtieri CT, Evans RW. Stimulant treatment for the neurobehavioural sequelae of traumatic brain injury. Brain Inj. 1988;2(4):273-290.
40. Provigil [package insert]. North Wales, PA: Cephalon Inc; 2015.
41. Nuvigil [package insert]. Frazer, PA: Cephalon, Inc; 2013.
42. Menza MA, Kaufman KR, Castellanos A. Modafinil augmentation of antidepressant treatment in depression. J Clin Psychiatry. 2000;61(5):378-381.
43. Markovitz PJ, Wagner S. An open-label trial of modafinil augmentation in patients with partial response to antidepressant therapy. J Clin Psychopharmacol. 2003;23(2):207-209.
44. Fernandes PP, Petty F. Modafinil for remitted bipolar depression with hypersomnia. Ann Pharmacother. 2003;37(12):1807-1809.
45. Nasr S. Modafinil as adjunctive therapy in depressed outpatients. Ann Clin Psychiatry. 2004;16(3):133-138.
46. DeBattista C, Lembke A, Solvason HB, Ghebremichael R, Poirier J. A prospective trial of modafinil as an adjunctive treatment of major depression. J Clin Psychopharmacol. 2004;24(1):87-90.
47. Nasr S, Wendt B, Steiner K. Absence of mood switch with and tolerance to modafinil: A replication study from a large private practice. J Affect Disord. 2006;95(1-3):111-114.
48. DeBattista C, Doghramji K, Menza MA, Rosenthal MH, Fieve RR; Modafinil in Depression Study Group. Adjunct modafinil for the short-term treatment of fatigue and sleepiness in patients with major depressive disorder: A preliminary doubleblind, placebo-controlled study. J Clin Psychiatry. 2003;64(9):1057-1064.
49. Frye MA, Grunze H, Suppes T, et al. A placebo-controlled evaluation of adjunctive modafinil in the treatment of bipolar depression. Am J Psychiatry. 2007;164(8):1242-1249.
50. Fava M, Thase ME, DeBattista C, Doghramji K, Arora S, Hughes RJ. Modafinil augmentation of selective serotonin reuptake inhibitor therapy in MDD partial responders with persistent fatigue and sleepiness. Ann Clin Psychiatry. 2007;19(3):153-159.
51. Thase ME, Fava M, DeBattista C, Arora S, Hughes RJ. Modafinil augmentation of SSRI therapy in patients with major depressive disorder and excessive sleepiness and fatigue: A 12-week, open-label, extension study. CNS Spectr. 2006;11(2):93-102.
52. Fava M, Thase ME, DeBattista C. A multicenter, placebo-controlled study of modafinil augmentation in partial responders to selective serotonin reuptake inhibitors with persistent fatigue and sleepiness. J Clin Psychiatry. 2005;66(1):85-93.
53. Abolfazli R, Hosseini M, Ghanizadeh A, et al. Double-blind randomized parallelgroup clinical trial of efficacy of the combination fluoxetine plus modafinil versus fluoxetine plus placebo in the treatment of major depression. Depress Anxiety. 2011;28(4):297-302.
54. Rasmussen NA, Schrøder P, Olsen LR, Brødsgaard M, Undén M, Bech P. Modafinil augmentation in depressed patients with partial response to antidepressants: A pilot study on self-reported symptoms covered by the Major Depression Inventory (MDI) and the Symptom Checklist (SCL-92). Nord J Psychiatry. 2005;59(3):173-178.
55. Dunlop BW, Crits-Christoph P, Evans DL, et al. Coadministration of modafinil and a selective serotonin reuptake inhibitor from the initiation of treatment of major depressive disorder with fatigue and sleepiness: A double-blind, placebocontrolled study. J Clin Psychopharmacol. 2007;27(6):614-619.
56. Calabrese JR, Ketter TA, Youakim JM, Tiller JM, Yang R, Frye MA. Adjunctive armodafinil
for major depressive episodes associated with bipolar I disorder: A randomized multicenter, double-blind, placebo-controlled, proof-of-concept study. J Clin Psychiatry. 2010;71(10):1363-1370.
57. Calabrese JR, Frye MA, Yang R, Ketter TA; Armodafinil Treatment Trial Study Network. Efficacy and safety of adjunctive armodafinil in adults with major depressive episodes associated with bipolar I disorder: A randomized, double-blind, placebo-controlled, multicenter trial. J Clin Psychiatry. 2014;75(10):1054-1061.
58. Strattera [package insert]. Indianapolis, IN. Lilly; 2015.
59. Carpenter LL, Milosavljevic N, Schecter JM, Tyrka AR, Price LH. Augmentation with open-label atomoxetine for partial or nonresponse to antidepressants. J Clin Psychiatry. 2005;66(10):1234-1238.
60. Papakostas GI, Petersen TJ, Burns AM, Fava M. Adjunctive atomoxetine for residual
fatigue in major depressive disorder. J Psychiatr Res. 2006;40(4):370-373.
61. Michelson D, Adler LA, Amsterdam JD, et al. Addition of atomoxetine for depression
incompletely responsive to sertraline: A randomized, double-blind, placebocontrolled study. J Clin Psychiatry. 2007;68(4):582-587.
62. Corp SA, Gitlin MJ, Altshuler LL. A review of the use of stimulants and stimulant alternatives in treating bipolar depression and major depressive disorder. J Clin Psychiatry. 2014;75(9):1010-1018.
63. Kraemer M, Uekermann J, Wiltfang J, Kis B. Methylphenidate-induced psychosis in adult attention-deficit/hyperactivity disorder: Report of 3 new cases and review of the literature. Clin Neuropharmacol. 2010;33(4):204-206.
64. Berman SM, Kuczenski R, McCracken JT, London ED. Potential adverse effects of amphetamine treatment on brain and behavior: A review. Mol Psychiatry. 2009;14(2):123-142.
65. Fredriksen M, Dahl AA, Martinsen EW, Klungsøyr O, Haavik J, Peleikis DE. Effectiveness of one-year pharmacological treatment of adult attention-deficit/hyperactivity disorder (ADHD): An open-label prospective study of time in treatment, dose, side-effects and comorbidity. Eur Neuropsychopharmacol. 2014;24(12):1873-1874.
66. Hardy SE. Methylphenidate for the treatment of depressive symptoms, including fatigue and apathy, in medically ill older adults and terminally ill adults. Am J Geriatr Pharmacother. 2009;7(1):34-59.
67. Williams RJ, Goodale LA, Shay-Fiddler MA, Gloster SP, Chang SY. Methylphenidate and dextroamphetamine abuse in substance-abusing adolescents. Am J Addict. 2004;13(4):381-389.
68. Madaan V, Kolli V, Bestha DP, Shah MJ. Update on optimal use of lisdexamfetamine in the treatment of ADHD. Neuropsychiatr Dis Treat. 2013;9:977-983.
69. Ross RG. Psychotic and manic-like symptoms during stimulant treatment of attention deficit hyperactivity disorder. Am J Psychiatry. 2006;163(7):1149-1152.
70. Dell’Osso B, Ketter TA. Use of adjunctive stimulants in adult bipolar depression. Int J Neuropsychopharmacol. 2013;16(1):55-68.
71. Habel LA, Cooper WO, Sox CM, et al. ADHD medications and risk of serious cardiovascular events in young and middle-aged adults. JAMA. 2011;306(24):2673-2683.
72. Schelleman H, Bilker WB, Kimmel SE, et al. Methylphenidate and risk of serious cardiovascular events in adults. Am J Psychiatry. 2012;169(2):178-185.
73. Bolea-Alamañac B, Nutt DJ, Adamou M, et al; British Association for Psychopharmacology. Evidence-based guidelines for the pharmacological management of attention deficit hyperactivity disorder: Update on recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2014;28(3):179-203.
74. Moher D, Liberati A, Tetzlaff J, Altman DG; The PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009;6(6):e1000097.
When to consider cranial electrotherapy stimulation for patients with PTSD
Individuals with posttraumatic stress disorder (PTSD) often report cognitive and sleep disturbances, such as insomnia and poor concentration. Although many patients report improvement with traditional evidence-based treatments, such as pharmacotherapy and psychotherapy, it might be valuable to consider complementary or alternative therapies. Many patients seek treatments that they can self-administer as needed, at their convenience, particularly during symptom exacerbation. One treatment option is cranial electrotherapy stimulation (CES).
As a medical device, CES has been cleared—rather than approved, as is the case for medications—by the FDA to treat depression, insomnia, and anxiety.1 In the United States, CES devices require a prescription from a licensed health care practitioner, but they are available without a prescription in other countries. Cost for devices range from $600 to $1,200 and $10 to $20 for electrodes and contact solution. However, insurance companies that provide coverage for durable medical equipment might cover some or all of this expense.
How CES works
After applying contact solution, depending on the device used, the user attaches electrodes to the earlobes, mastoid processes, or other parts of the head that deliver a pulsed current, usually from AA batteries for 20 to 60 minutes.1 The current causes cortical deactivation and could affect emotional regulation by influencing neurotransmission in the thalamus, hypothalamus, and limbic system.1,2 CES increases cerebrospinal fluid levels of beta-endorphin, adrenocorticotropic hormone, and serotonin, which play a role in depression and anxiety.3
There are no known contraindications for CES. Adverse effects are rare, temporary, and mild; skin irritation, vertigo, or headache are the most common.1
Evidence of efficacy
There are no double-blind placebo-controlled trials evaluating the efficacy of CES for PTSD. However, there is a case series and a large survey of patients supporting its use.
- In a case series, 2 patients reported improved occupational functioning and reduced PTSD symptoms after using CES, 100 to 500 mA, 20 to 60 minutes a day, 3 to 5 days per week.4
- In an online survey of 145 veterans and active-duty military personnel, 60% of individuals used CES for PTSD, and 20% of those individuals were not receiving pharmacotherapy.5 Participants reported at least a 25% reduction in symptoms using CES for at least 20 minutes, once or twice daily, with a current of 100 to 600 mA.5
- In an expert opinion, patients noted improved sleep quality and reduced alcohol and drug withdrawal symptoms after 20-minute treatments, twice a day, with a current of 2 mA. Currents could be increased to 4 mA, if there was no improvement after 2 weeks.6
Some patients experiencing exacerbation of PTSD symptoms could benefit from using the device for 1 hour several times a day until symptoms subside.5
Optimal strength, frequency, and duration of treatment vary among patients, and further studies are needed to assess these parameters as well as efficacy because definitive studies are currently lacking. CES has not always shown efficacy, such as in some patients with depression.7 Despite the limited evidence base, it is reasonable to consider CES for patients with PTSD. This modality might be helpful for patients who have comorbid pain, anxiety, and insomnia, or for those who seek a complementary, convenient, safe, self-administered treatment.
1. Kirsch DL, Nichols F. Cranial electrotherapy stimulation for treatment of anxiety, depression, and insomnia. Psychiatr Clin North Am. 2013;36(1):169-176.
2. Feusner JD, Madsen S, Moody TD, et al. Effects of cranial electrotherapy stimulation on resting state brain activity. Brain Behav. 2012;2(3):211-220.
3. Shealy CN, Cady RK, Culver-Veehoff D, et al. Cerebrospinal fluid and plasma neurochemicals: response to cranial electrical stimulation. J Neuro Orthop Med Surg. 1998;18(2):94-97.
4. Bracciano AG, Chang WP, Kokesh S, et al. Cranial electrotherapy stimulation in the treatment of posttraumatic stress disorder: a pilot study of two military veterans. J Neurother. 2012;16(1):60-69.
5. Kirsch DL, Price LR, Nichols F, et al. Military service member and veteran self reports of efficacy of cranial electrotherapy stimulation for anxiety, posttraumatic stress disorder, insomnia, and depression. US Army Med Dep J. 2014:46-54.
6. Xenakis SN. The rise of cranial electrotherapy. Psychiatric Times. http://www.psychiatrictimes.com/electroconvulsive-therapy/rise-cranial-electrotherapy. Published July 24, 2014. Accessed December 20, 2016.
7. Mischoulon D, De Jong MF, Vitolo OV, et al. Efficacy and safety of a form of cranial electrical stimulation (CES) as an add-on intervention for treatment-resistant major depressive disorder: a three week double blind pilot study. J Psychiatr Res. 2015;70:98-105.
Individuals with posttraumatic stress disorder (PTSD) often report cognitive and sleep disturbances, such as insomnia and poor concentration. Although many patients report improvement with traditional evidence-based treatments, such as pharmacotherapy and psychotherapy, it might be valuable to consider complementary or alternative therapies. Many patients seek treatments that they can self-administer as needed, at their convenience, particularly during symptom exacerbation. One treatment option is cranial electrotherapy stimulation (CES).
As a medical device, CES has been cleared—rather than approved, as is the case for medications—by the FDA to treat depression, insomnia, and anxiety.1 In the United States, CES devices require a prescription from a licensed health care practitioner, but they are available without a prescription in other countries. Cost for devices range from $600 to $1,200 and $10 to $20 for electrodes and contact solution. However, insurance companies that provide coverage for durable medical equipment might cover some or all of this expense.
How CES works
After applying contact solution, depending on the device used, the user attaches electrodes to the earlobes, mastoid processes, or other parts of the head that deliver a pulsed current, usually from AA batteries for 20 to 60 minutes.1 The current causes cortical deactivation and could affect emotional regulation by influencing neurotransmission in the thalamus, hypothalamus, and limbic system.1,2 CES increases cerebrospinal fluid levels of beta-endorphin, adrenocorticotropic hormone, and serotonin, which play a role in depression and anxiety.3
There are no known contraindications for CES. Adverse effects are rare, temporary, and mild; skin irritation, vertigo, or headache are the most common.1
Evidence of efficacy
There are no double-blind placebo-controlled trials evaluating the efficacy of CES for PTSD. However, there is a case series and a large survey of patients supporting its use.
- In a case series, 2 patients reported improved occupational functioning and reduced PTSD symptoms after using CES, 100 to 500 mA, 20 to 60 minutes a day, 3 to 5 days per week.4
- In an online survey of 145 veterans and active-duty military personnel, 60% of individuals used CES for PTSD, and 20% of those individuals were not receiving pharmacotherapy.5 Participants reported at least a 25% reduction in symptoms using CES for at least 20 minutes, once or twice daily, with a current of 100 to 600 mA.5
- In an expert opinion, patients noted improved sleep quality and reduced alcohol and drug withdrawal symptoms after 20-minute treatments, twice a day, with a current of 2 mA. Currents could be increased to 4 mA, if there was no improvement after 2 weeks.6
Some patients experiencing exacerbation of PTSD symptoms could benefit from using the device for 1 hour several times a day until symptoms subside.5
Optimal strength, frequency, and duration of treatment vary among patients, and further studies are needed to assess these parameters as well as efficacy because definitive studies are currently lacking. CES has not always shown efficacy, such as in some patients with depression.7 Despite the limited evidence base, it is reasonable to consider CES for patients with PTSD. This modality might be helpful for patients who have comorbid pain, anxiety, and insomnia, or for those who seek a complementary, convenient, safe, self-administered treatment.
Individuals with posttraumatic stress disorder (PTSD) often report cognitive and sleep disturbances, such as insomnia and poor concentration. Although many patients report improvement with traditional evidence-based treatments, such as pharmacotherapy and psychotherapy, it might be valuable to consider complementary or alternative therapies. Many patients seek treatments that they can self-administer as needed, at their convenience, particularly during symptom exacerbation. One treatment option is cranial electrotherapy stimulation (CES).
As a medical device, CES has been cleared—rather than approved, as is the case for medications—by the FDA to treat depression, insomnia, and anxiety.1 In the United States, CES devices require a prescription from a licensed health care practitioner, but they are available without a prescription in other countries. Cost for devices range from $600 to $1,200 and $10 to $20 for electrodes and contact solution. However, insurance companies that provide coverage for durable medical equipment might cover some or all of this expense.
How CES works
After applying contact solution, depending on the device used, the user attaches electrodes to the earlobes, mastoid processes, or other parts of the head that deliver a pulsed current, usually from AA batteries for 20 to 60 minutes.1 The current causes cortical deactivation and could affect emotional regulation by influencing neurotransmission in the thalamus, hypothalamus, and limbic system.1,2 CES increases cerebrospinal fluid levels of beta-endorphin, adrenocorticotropic hormone, and serotonin, which play a role in depression and anxiety.3
There are no known contraindications for CES. Adverse effects are rare, temporary, and mild; skin irritation, vertigo, or headache are the most common.1
Evidence of efficacy
There are no double-blind placebo-controlled trials evaluating the efficacy of CES for PTSD. However, there is a case series and a large survey of patients supporting its use.
- In a case series, 2 patients reported improved occupational functioning and reduced PTSD symptoms after using CES, 100 to 500 mA, 20 to 60 minutes a day, 3 to 5 days per week.4
- In an online survey of 145 veterans and active-duty military personnel, 60% of individuals used CES for PTSD, and 20% of those individuals were not receiving pharmacotherapy.5 Participants reported at least a 25% reduction in symptoms using CES for at least 20 minutes, once or twice daily, with a current of 100 to 600 mA.5
- In an expert opinion, patients noted improved sleep quality and reduced alcohol and drug withdrawal symptoms after 20-minute treatments, twice a day, with a current of 2 mA. Currents could be increased to 4 mA, if there was no improvement after 2 weeks.6
Some patients experiencing exacerbation of PTSD symptoms could benefit from using the device for 1 hour several times a day until symptoms subside.5
Optimal strength, frequency, and duration of treatment vary among patients, and further studies are needed to assess these parameters as well as efficacy because definitive studies are currently lacking. CES has not always shown efficacy, such as in some patients with depression.7 Despite the limited evidence base, it is reasonable to consider CES for patients with PTSD. This modality might be helpful for patients who have comorbid pain, anxiety, and insomnia, or for those who seek a complementary, convenient, safe, self-administered treatment.
1. Kirsch DL, Nichols F. Cranial electrotherapy stimulation for treatment of anxiety, depression, and insomnia. Psychiatr Clin North Am. 2013;36(1):169-176.
2. Feusner JD, Madsen S, Moody TD, et al. Effects of cranial electrotherapy stimulation on resting state brain activity. Brain Behav. 2012;2(3):211-220.
3. Shealy CN, Cady RK, Culver-Veehoff D, et al. Cerebrospinal fluid and plasma neurochemicals: response to cranial electrical stimulation. J Neuro Orthop Med Surg. 1998;18(2):94-97.
4. Bracciano AG, Chang WP, Kokesh S, et al. Cranial electrotherapy stimulation in the treatment of posttraumatic stress disorder: a pilot study of two military veterans. J Neurother. 2012;16(1):60-69.
5. Kirsch DL, Price LR, Nichols F, et al. Military service member and veteran self reports of efficacy of cranial electrotherapy stimulation for anxiety, posttraumatic stress disorder, insomnia, and depression. US Army Med Dep J. 2014:46-54.
6. Xenakis SN. The rise of cranial electrotherapy. Psychiatric Times. http://www.psychiatrictimes.com/electroconvulsive-therapy/rise-cranial-electrotherapy. Published July 24, 2014. Accessed December 20, 2016.
7. Mischoulon D, De Jong MF, Vitolo OV, et al. Efficacy and safety of a form of cranial electrical stimulation (CES) as an add-on intervention for treatment-resistant major depressive disorder: a three week double blind pilot study. J Psychiatr Res. 2015;70:98-105.
1. Kirsch DL, Nichols F. Cranial electrotherapy stimulation for treatment of anxiety, depression, and insomnia. Psychiatr Clin North Am. 2013;36(1):169-176.
2. Feusner JD, Madsen S, Moody TD, et al. Effects of cranial electrotherapy stimulation on resting state brain activity. Brain Behav. 2012;2(3):211-220.
3. Shealy CN, Cady RK, Culver-Veehoff D, et al. Cerebrospinal fluid and plasma neurochemicals: response to cranial electrical stimulation. J Neuro Orthop Med Surg. 1998;18(2):94-97.
4. Bracciano AG, Chang WP, Kokesh S, et al. Cranial electrotherapy stimulation in the treatment of posttraumatic stress disorder: a pilot study of two military veterans. J Neurother. 2012;16(1):60-69.
5. Kirsch DL, Price LR, Nichols F, et al. Military service member and veteran self reports of efficacy of cranial electrotherapy stimulation for anxiety, posttraumatic stress disorder, insomnia, and depression. US Army Med Dep J. 2014:46-54.
6. Xenakis SN. The rise of cranial electrotherapy. Psychiatric Times. http://www.psychiatrictimes.com/electroconvulsive-therapy/rise-cranial-electrotherapy. Published July 24, 2014. Accessed December 20, 2016.
7. Mischoulon D, De Jong MF, Vitolo OV, et al. Efficacy and safety of a form of cranial electrical stimulation (CES) as an add-on intervention for treatment-resistant major depressive disorder: a three week double blind pilot study. J Psychiatr Res. 2015;70:98-105.
Think beyond prazosin when treating nightmares in PTSD
Nightmares are a common feature of posttraumatic stress disorder (PTSD) that could lead to fatigue, impaired concentration, and poor work performance. The α-1 antagonist prazosin decreases noradrenergic hyperactivity and reduces nightmares; however, it can cause adverse effects, be contraindicated, or provide no benefit to some patients. Consider these alternative medications to reduce nightmares in PTSD.
Alpha-2 agonists
Clonidine and guanfacine are α-2 agonists, used to treat attention-deficit/hyperactivity disorder and high blood pressure, that decrease noradrenergic activity, and either medication might be preferable to prazosin because they are more likely to cause sedation. A review and a case series showed that many patients—some with comorbid traumatic brain injury—reported fewer nightmares after taking 0.2 to 0.6 mg of clonidine.1,2 Guanfacine might be more beneficial because it has a longer half-life; 2 mg of guanfacine eliminated nightmares in 1 patient.3 However, in a double-blind placebo-controlled study and an extension study, guanfacine did not reduce nightmares or other PTSD symptoms.4,5
Initiate 0.1 mg of clonidine at bedtime, and titrate to efficacy or to 0.6 mg. Similarly, initiate guanfacine at 1 mg, and titrate to efficacy or to 4 mg. Monitor for hypotension, excess sedation, dry mouth, and rebound hypertension.
Cyproheptadine
Used to treat serotonin syndrome, cyproheptadine’s antagonism of serotonin 2A receptors has varying efficacy for reducing nightmares. Some patients have reported a decrease in nightmares at dosages ranging from 4 to 24 mg.1,6 Other studies found no reduction in nightmares or diminished quality of sleep.1,7
Initiate cyproheptadine at 4 mg/d, titrate every 2 or 3 days, and monitor for sedation, confusion, or reduced efficacy of concurrent serotonergic medications. Cyproheptadine might be preferable for its sedating effect and potential to reduce sexual adverse effects from serotonergic medications.
Topiramate
Topiramate is approved for treatment of epilepsy and migraine headache. At 75 to 100 mg/d in a clinical trial, topiramate partially or completely suppressed nightmares.8 Start with 25 mg/d, titrate to efficacy, and monitor for anorexia, paresthesias, and cognitive impairment. Topiramate might be better than prazosin for patients without renal impairment who want sedation, weight loss, or reduced irritability.
Gabapentin
Gabapentin is approved to treat seizures and postherpetic neuralgia and also is used to treat neuropathic pain. When 300 to 3,600 mg/d (mean dosage, 1,300 mg/d) of gabapentin was added to medication regimens, most patients reported decreased frequency or intensity of nightmares.9 Monitor patients for sedation, dizziness, mood changes, and weight gain. Gabapentin might be an option for patients without renal impairment who have comorbid pain, insomnia, or anxiety.
Are these reasonable alternatives?
Despite small sample sizes in published studies and few randomized trials, clonidine, guanfacine, cyproheptadine, topiramate, and gabapentin are reasonable alternatives to prazosin for reducing nightmares in patients with PTSD.
Disclosure
The author reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Aurora RN, Zak RS, Auerbach SH, et al; Standards of Practice Committee; American Academy of Sleep Medicine. Best practice guide for the treatment of nightmare disorder in adults. J Clin Sleep Med. 2010;6(4):389-401.
2. Alao A, Selvarajah J, Razi S. The use of clonidine in the treatment of nightmares among patients with co-morbid PTSD and traumatic brain injury. Int J Psychiatry Med. 2012;44(2):165-169.
3. Horrigan JP, Barnhill LJ. The suppression of nightmares with guanfacine. J Clin Psychiatry. 1996;57(8):371.
4. Davis LL, Ward C, Rasmusson A, et al. A placebo-controlled trial of guanfacine for the treatment of posttraumatic stress disorder in veterans. Psychopharmacol Bull. 2008;41(1):8-18.
5. Neylan TC, Lenoci M, Samuelson KW, et al. No improvement of posttraumatic stress disorder symptoms with guanfacine treatment. Am J Psychiatry. 2006;163(12):2186-2188.
6. Harsch HH. Cyproheptadine for recurrent nightmares. Am J Psychiatry. 1986;143(11):1491-1492.
7. Jacobs-Rebhun S, Schnurr PP, Friedman MJ, et al. Posttraumatic stress disorder and sleep difficulty. Am J Psychiatry. 2000;157(9):1525-1526.
8. Berlant J, van Kammen DP. Open-label topiramate as primary or adjunctive therapy in chronic civilian posttraumatic stress disorder: a preliminary report. J Clin Psychiatry. 2002;63(1):15-20.
9. Hamner MB, Brodrick PS, Labbate LA. Gabapentin in PTSD: a retrospective, clinical series of adjunctive therapy. Ann Clin Psychiatry. 2001;13(3):141-146.
Nightmares are a common feature of posttraumatic stress disorder (PTSD) that could lead to fatigue, impaired concentration, and poor work performance. The α-1 antagonist prazosin decreases noradrenergic hyperactivity and reduces nightmares; however, it can cause adverse effects, be contraindicated, or provide no benefit to some patients. Consider these alternative medications to reduce nightmares in PTSD.
Alpha-2 agonists
Clonidine and guanfacine are α-2 agonists, used to treat attention-deficit/hyperactivity disorder and high blood pressure, that decrease noradrenergic activity, and either medication might be preferable to prazosin because they are more likely to cause sedation. A review and a case series showed that many patients—some with comorbid traumatic brain injury—reported fewer nightmares after taking 0.2 to 0.6 mg of clonidine.1,2 Guanfacine might be more beneficial because it has a longer half-life; 2 mg of guanfacine eliminated nightmares in 1 patient.3 However, in a double-blind placebo-controlled study and an extension study, guanfacine did not reduce nightmares or other PTSD symptoms.4,5
Initiate 0.1 mg of clonidine at bedtime, and titrate to efficacy or to 0.6 mg. Similarly, initiate guanfacine at 1 mg, and titrate to efficacy or to 4 mg. Monitor for hypotension, excess sedation, dry mouth, and rebound hypertension.
Cyproheptadine
Used to treat serotonin syndrome, cyproheptadine’s antagonism of serotonin 2A receptors has varying efficacy for reducing nightmares. Some patients have reported a decrease in nightmares at dosages ranging from 4 to 24 mg.1,6 Other studies found no reduction in nightmares or diminished quality of sleep.1,7
Initiate cyproheptadine at 4 mg/d, titrate every 2 or 3 days, and monitor for sedation, confusion, or reduced efficacy of concurrent serotonergic medications. Cyproheptadine might be preferable for its sedating effect and potential to reduce sexual adverse effects from serotonergic medications.
Topiramate
Topiramate is approved for treatment of epilepsy and migraine headache. At 75 to 100 mg/d in a clinical trial, topiramate partially or completely suppressed nightmares.8 Start with 25 mg/d, titrate to efficacy, and monitor for anorexia, paresthesias, and cognitive impairment. Topiramate might be better than prazosin for patients without renal impairment who want sedation, weight loss, or reduced irritability.
Gabapentin
Gabapentin is approved to treat seizures and postherpetic neuralgia and also is used to treat neuropathic pain. When 300 to 3,600 mg/d (mean dosage, 1,300 mg/d) of gabapentin was added to medication regimens, most patients reported decreased frequency or intensity of nightmares.9 Monitor patients for sedation, dizziness, mood changes, and weight gain. Gabapentin might be an option for patients without renal impairment who have comorbid pain, insomnia, or anxiety.
Are these reasonable alternatives?
Despite small sample sizes in published studies and few randomized trials, clonidine, guanfacine, cyproheptadine, topiramate, and gabapentin are reasonable alternatives to prazosin for reducing nightmares in patients with PTSD.
Disclosure
The author reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Nightmares are a common feature of posttraumatic stress disorder (PTSD) that could lead to fatigue, impaired concentration, and poor work performance. The α-1 antagonist prazosin decreases noradrenergic hyperactivity and reduces nightmares; however, it can cause adverse effects, be contraindicated, or provide no benefit to some patients. Consider these alternative medications to reduce nightmares in PTSD.
Alpha-2 agonists
Clonidine and guanfacine are α-2 agonists, used to treat attention-deficit/hyperactivity disorder and high blood pressure, that decrease noradrenergic activity, and either medication might be preferable to prazosin because they are more likely to cause sedation. A review and a case series showed that many patients—some with comorbid traumatic brain injury—reported fewer nightmares after taking 0.2 to 0.6 mg of clonidine.1,2 Guanfacine might be more beneficial because it has a longer half-life; 2 mg of guanfacine eliminated nightmares in 1 patient.3 However, in a double-blind placebo-controlled study and an extension study, guanfacine did not reduce nightmares or other PTSD symptoms.4,5
Initiate 0.1 mg of clonidine at bedtime, and titrate to efficacy or to 0.6 mg. Similarly, initiate guanfacine at 1 mg, and titrate to efficacy or to 4 mg. Monitor for hypotension, excess sedation, dry mouth, and rebound hypertension.
Cyproheptadine
Used to treat serotonin syndrome, cyproheptadine’s antagonism of serotonin 2A receptors has varying efficacy for reducing nightmares. Some patients have reported a decrease in nightmares at dosages ranging from 4 to 24 mg.1,6 Other studies found no reduction in nightmares or diminished quality of sleep.1,7
Initiate cyproheptadine at 4 mg/d, titrate every 2 or 3 days, and monitor for sedation, confusion, or reduced efficacy of concurrent serotonergic medications. Cyproheptadine might be preferable for its sedating effect and potential to reduce sexual adverse effects from serotonergic medications.
Topiramate
Topiramate is approved for treatment of epilepsy and migraine headache. At 75 to 100 mg/d in a clinical trial, topiramate partially or completely suppressed nightmares.8 Start with 25 mg/d, titrate to efficacy, and monitor for anorexia, paresthesias, and cognitive impairment. Topiramate might be better than prazosin for patients without renal impairment who want sedation, weight loss, or reduced irritability.
Gabapentin
Gabapentin is approved to treat seizures and postherpetic neuralgia and also is used to treat neuropathic pain. When 300 to 3,600 mg/d (mean dosage, 1,300 mg/d) of gabapentin was added to medication regimens, most patients reported decreased frequency or intensity of nightmares.9 Monitor patients for sedation, dizziness, mood changes, and weight gain. Gabapentin might be an option for patients without renal impairment who have comorbid pain, insomnia, or anxiety.
Are these reasonable alternatives?
Despite small sample sizes in published studies and few randomized trials, clonidine, guanfacine, cyproheptadine, topiramate, and gabapentin are reasonable alternatives to prazosin for reducing nightmares in patients with PTSD.
Disclosure
The author reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Aurora RN, Zak RS, Auerbach SH, et al; Standards of Practice Committee; American Academy of Sleep Medicine. Best practice guide for the treatment of nightmare disorder in adults. J Clin Sleep Med. 2010;6(4):389-401.
2. Alao A, Selvarajah J, Razi S. The use of clonidine in the treatment of nightmares among patients with co-morbid PTSD and traumatic brain injury. Int J Psychiatry Med. 2012;44(2):165-169.
3. Horrigan JP, Barnhill LJ. The suppression of nightmares with guanfacine. J Clin Psychiatry. 1996;57(8):371.
4. Davis LL, Ward C, Rasmusson A, et al. A placebo-controlled trial of guanfacine for the treatment of posttraumatic stress disorder in veterans. Psychopharmacol Bull. 2008;41(1):8-18.
5. Neylan TC, Lenoci M, Samuelson KW, et al. No improvement of posttraumatic stress disorder symptoms with guanfacine treatment. Am J Psychiatry. 2006;163(12):2186-2188.
6. Harsch HH. Cyproheptadine for recurrent nightmares. Am J Psychiatry. 1986;143(11):1491-1492.
7. Jacobs-Rebhun S, Schnurr PP, Friedman MJ, et al. Posttraumatic stress disorder and sleep difficulty. Am J Psychiatry. 2000;157(9):1525-1526.
8. Berlant J, van Kammen DP. Open-label topiramate as primary or adjunctive therapy in chronic civilian posttraumatic stress disorder: a preliminary report. J Clin Psychiatry. 2002;63(1):15-20.
9. Hamner MB, Brodrick PS, Labbate LA. Gabapentin in PTSD: a retrospective, clinical series of adjunctive therapy. Ann Clin Psychiatry. 2001;13(3):141-146.
1. Aurora RN, Zak RS, Auerbach SH, et al; Standards of Practice Committee; American Academy of Sleep Medicine. Best practice guide for the treatment of nightmare disorder in adults. J Clin Sleep Med. 2010;6(4):389-401.
2. Alao A, Selvarajah J, Razi S. The use of clonidine in the treatment of nightmares among patients with co-morbid PTSD and traumatic brain injury. Int J Psychiatry Med. 2012;44(2):165-169.
3. Horrigan JP, Barnhill LJ. The suppression of nightmares with guanfacine. J Clin Psychiatry. 1996;57(8):371.
4. Davis LL, Ward C, Rasmusson A, et al. A placebo-controlled trial of guanfacine for the treatment of posttraumatic stress disorder in veterans. Psychopharmacol Bull. 2008;41(1):8-18.
5. Neylan TC, Lenoci M, Samuelson KW, et al. No improvement of posttraumatic stress disorder symptoms with guanfacine treatment. Am J Psychiatry. 2006;163(12):2186-2188.
6. Harsch HH. Cyproheptadine for recurrent nightmares. Am J Psychiatry. 1986;143(11):1491-1492.
7. Jacobs-Rebhun S, Schnurr PP, Friedman MJ, et al. Posttraumatic stress disorder and sleep difficulty. Am J Psychiatry. 2000;157(9):1525-1526.
8. Berlant J, van Kammen DP. Open-label topiramate as primary or adjunctive therapy in chronic civilian posttraumatic stress disorder: a preliminary report. J Clin Psychiatry. 2002;63(1):15-20.
9. Hamner MB, Brodrick PS, Labbate LA. Gabapentin in PTSD: a retrospective, clinical series of adjunctive therapy. Ann Clin Psychiatry. 2001;13(3):141-146.
Options for treating antidepressant-induced sweating
Excessive sweating—diaphoresis—affects up to 22% of patients who take antidepressants.1 Diaphoresis may interfere with social and occupational activities, which can lead to medication discontinuation and prevent effective treatment. Stopping, decreasing, or changing antidepressants are options, but patients may be reluctant if the current dose has relieved their depressive symptoms. Adding a medication to reduce diaphoresis may be appropriate.
Sympathetic division of the peripheral nervous system signals cholinergic neurons to stimulate sweat gland secretion. In the CNS, thermoregulation occurs in the hypothalamus through a balanced and complex interaction among serotonergic and dopaminergic neurons.1 Consequently, oral medications to decrease sweating target peripheral or CNS neurons. Although evidence is limited to case reports, consider cholinergic and serotonergic antagonists and dopamine partial agonists to relieve antidepressant-induced diaphoresis.
Pharmacologic options
Peripherally, the anticholinergic agent benztropine reduced or eliminated diaphoresis at doses ranging from 0.5 mg every other day to 1 mg/d.2,3 Dry mouth was the only reported side effect.
Centrally acting serotonin antagonists may decrease diaphoresis through the 5-HT2A receptor, which signals the hypothalamus to raise body temperature. Cyproheptadine is an antihistamine with serotonin receptor antagonism. In case reports, it reduced or eliminated sweating in doses of 4 mg once or twice daily.4 Mild sedation was the only noted adverse effect. The norepinephrine and serotonin antagonist mirtazapine reduced diaphoresis within 2 weeks of initiation at 15 mg/d with no adverse effects.5 Sweating resolved after mirtazapine was titrated to 60 mg/d.
In addition to excess serotonin activity, diaphoresis may result from decreased dopaminergic tone in the hypothalamus. Centrally acting dopamine agonists—even partial agonists—may restore homeostasis and decrease sweating. Aripiprazole, 10 to 20 mg/d, reduced sweating in 2 patients; no adverse effects were reported.6
Agents to avoid
Antiadrenergic medications such as clonidine have decreased or exacerbated diaphoresis in studies.1 Similarly, paroxetine may alleviate or cause sweating. It is difficult to attribute paroxetine’s occasional effectiveness in reducing sweating solely to its anticholinergic properties because improvement may be attributed to an initial anxiolytic effect or efficacy in treating the underlying anxiety disorder.1
Disclosure
Dr. Scarff reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Marcy TR, Britton ML. Antidepressant-induced sweating. Ann Pharmacother. 2005;39(4):748-752.
2. Pierre JM, Guze BH. Benztropine for venlafaxine-induced night sweats. J Clin Psychopharmacol. 2000;20(2):269.-
3. Garber A, Gregory RJ. Benztropine in the treatment of venlafaxine-induced sweating. J Clin Psychiatry. 1997;58(4):176-177.
4. Ashton AK, Weinstein WL. Cyproheptadine for drug-induced sweating. Am J Psychiatry. 2002;159(5):874-875.
5. Buecking A, Vandeleur CL, Khazaal Y, et al. Mirtazapine in drug-induced excessive sweating. Eur J Clin Pharmacol. 2005;61(7):543-544.
6. Lu BY, Cullen CE, Eide CE, et al. Antidepressant-induced sweating alleviated by aripiprazole. J Clin Psychopharmacol. 2008;28(6):710-711.
Excessive sweating—diaphoresis—affects up to 22% of patients who take antidepressants.1 Diaphoresis may interfere with social and occupational activities, which can lead to medication discontinuation and prevent effective treatment. Stopping, decreasing, or changing antidepressants are options, but patients may be reluctant if the current dose has relieved their depressive symptoms. Adding a medication to reduce diaphoresis may be appropriate.
Sympathetic division of the peripheral nervous system signals cholinergic neurons to stimulate sweat gland secretion. In the CNS, thermoregulation occurs in the hypothalamus through a balanced and complex interaction among serotonergic and dopaminergic neurons.1 Consequently, oral medications to decrease sweating target peripheral or CNS neurons. Although evidence is limited to case reports, consider cholinergic and serotonergic antagonists and dopamine partial agonists to relieve antidepressant-induced diaphoresis.
Pharmacologic options
Peripherally, the anticholinergic agent benztropine reduced or eliminated diaphoresis at doses ranging from 0.5 mg every other day to 1 mg/d.2,3 Dry mouth was the only reported side effect.
Centrally acting serotonin antagonists may decrease diaphoresis through the 5-HT2A receptor, which signals the hypothalamus to raise body temperature. Cyproheptadine is an antihistamine with serotonin receptor antagonism. In case reports, it reduced or eliminated sweating in doses of 4 mg once or twice daily.4 Mild sedation was the only noted adverse effect. The norepinephrine and serotonin antagonist mirtazapine reduced diaphoresis within 2 weeks of initiation at 15 mg/d with no adverse effects.5 Sweating resolved after mirtazapine was titrated to 60 mg/d.
In addition to excess serotonin activity, diaphoresis may result from decreased dopaminergic tone in the hypothalamus. Centrally acting dopamine agonists—even partial agonists—may restore homeostasis and decrease sweating. Aripiprazole, 10 to 20 mg/d, reduced sweating in 2 patients; no adverse effects were reported.6
Agents to avoid
Antiadrenergic medications such as clonidine have decreased or exacerbated diaphoresis in studies.1 Similarly, paroxetine may alleviate or cause sweating. It is difficult to attribute paroxetine’s occasional effectiveness in reducing sweating solely to its anticholinergic properties because improvement may be attributed to an initial anxiolytic effect or efficacy in treating the underlying anxiety disorder.1
Disclosure
Dr. Scarff reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Excessive sweating—diaphoresis—affects up to 22% of patients who take antidepressants.1 Diaphoresis may interfere with social and occupational activities, which can lead to medication discontinuation and prevent effective treatment. Stopping, decreasing, or changing antidepressants are options, but patients may be reluctant if the current dose has relieved their depressive symptoms. Adding a medication to reduce diaphoresis may be appropriate.
Sympathetic division of the peripheral nervous system signals cholinergic neurons to stimulate sweat gland secretion. In the CNS, thermoregulation occurs in the hypothalamus through a balanced and complex interaction among serotonergic and dopaminergic neurons.1 Consequently, oral medications to decrease sweating target peripheral or CNS neurons. Although evidence is limited to case reports, consider cholinergic and serotonergic antagonists and dopamine partial agonists to relieve antidepressant-induced diaphoresis.
Pharmacologic options
Peripherally, the anticholinergic agent benztropine reduced or eliminated diaphoresis at doses ranging from 0.5 mg every other day to 1 mg/d.2,3 Dry mouth was the only reported side effect.
Centrally acting serotonin antagonists may decrease diaphoresis through the 5-HT2A receptor, which signals the hypothalamus to raise body temperature. Cyproheptadine is an antihistamine with serotonin receptor antagonism. In case reports, it reduced or eliminated sweating in doses of 4 mg once or twice daily.4 Mild sedation was the only noted adverse effect. The norepinephrine and serotonin antagonist mirtazapine reduced diaphoresis within 2 weeks of initiation at 15 mg/d with no adverse effects.5 Sweating resolved after mirtazapine was titrated to 60 mg/d.
In addition to excess serotonin activity, diaphoresis may result from decreased dopaminergic tone in the hypothalamus. Centrally acting dopamine agonists—even partial agonists—may restore homeostasis and decrease sweating. Aripiprazole, 10 to 20 mg/d, reduced sweating in 2 patients; no adverse effects were reported.6
Agents to avoid
Antiadrenergic medications such as clonidine have decreased or exacerbated diaphoresis in studies.1 Similarly, paroxetine may alleviate or cause sweating. It is difficult to attribute paroxetine’s occasional effectiveness in reducing sweating solely to its anticholinergic properties because improvement may be attributed to an initial anxiolytic effect or efficacy in treating the underlying anxiety disorder.1
Disclosure
Dr. Scarff reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
1. Marcy TR, Britton ML. Antidepressant-induced sweating. Ann Pharmacother. 2005;39(4):748-752.
2. Pierre JM, Guze BH. Benztropine for venlafaxine-induced night sweats. J Clin Psychopharmacol. 2000;20(2):269.-
3. Garber A, Gregory RJ. Benztropine in the treatment of venlafaxine-induced sweating. J Clin Psychiatry. 1997;58(4):176-177.
4. Ashton AK, Weinstein WL. Cyproheptadine for drug-induced sweating. Am J Psychiatry. 2002;159(5):874-875.
5. Buecking A, Vandeleur CL, Khazaal Y, et al. Mirtazapine in drug-induced excessive sweating. Eur J Clin Pharmacol. 2005;61(7):543-544.
6. Lu BY, Cullen CE, Eide CE, et al. Antidepressant-induced sweating alleviated by aripiprazole. J Clin Psychopharmacol. 2008;28(6):710-711.
1. Marcy TR, Britton ML. Antidepressant-induced sweating. Ann Pharmacother. 2005;39(4):748-752.
2. Pierre JM, Guze BH. Benztropine for venlafaxine-induced night sweats. J Clin Psychopharmacol. 2000;20(2):269.-
3. Garber A, Gregory RJ. Benztropine in the treatment of venlafaxine-induced sweating. J Clin Psychiatry. 1997;58(4):176-177.
4. Ashton AK, Weinstein WL. Cyproheptadine for drug-induced sweating. Am J Psychiatry. 2002;159(5):874-875.
5. Buecking A, Vandeleur CL, Khazaal Y, et al. Mirtazapine in drug-induced excessive sweating. Eur J Clin Pharmacol. 2005;61(7):543-544.
6. Lu BY, Cullen CE, Eide CE, et al. Antidepressant-induced sweating alleviated by aripiprazole. J Clin Psychopharmacol. 2008;28(6):710-711.