User login
How Should Common Symptoms at the End of Life be Managed?
Case
A 58-year-old male with colon cancer metastatic to the liver and lungs presents with vomiting, dyspnea, and abdominal pain. His disease has progressed through third-line chemotherapy and his care is now focused entirely on symptom management. He has not had a bowel movement in five days and he began vomiting two days ago.
Overview
The majority of patients in the United States die in acute-care hospitals. The Study to Understand Prognosis and Preferences for Outcomes and Risks of Treatments (SUPPORT), which evaluated the courses of close to 10,000 hospitalized patients with serious and life-limiting illnesses, illustrated that patients’ end-of-life (EOL) experiences often are characterized by poor symptom management and invasive care that is not congruent with the patients’ overall goals of care.1 Studies of factors identified as priorities in EOL care have consistently shown that excellent pain and symptom management are highly valued by patients and families. As the hospitalist movement continues to grow, hospitalists will play a large role in caring for patients at EOL and will need to know how to provide adequate pain and symptom management so that high-quality care can be achieved.
Pain: A Basic Tenet
A basic tenet of palliative medicine is to evaluate and treat all types of suffering.2 Physical pain at EOL is frequently accompanied by other types of pain, such as psychological, social, religious, or existential pain. However, this review will focus on the pharmacologic management of physical pain.
Pain management must begin with a thorough evaluation of the severity, location, and characteristics of the discomfort to assess which therapies are most likely to be beneficial (see Table 1).3 The consistent use of one scale of pain severity (such as 0-10, or mild/moderate/severe) assists in the choice of initial dose of pain medication, in determining the response to the medication, and in assessing the need for change in dose.4
Opioids are the foundation of pain management in advanced diseases because they are available in a number of formulations and, when dosed appropriately, they are effective and safe. Starting doses and equianalgesic doses of common opioids are presented in Table 2. Guidelines recommend the use of short-acting opioids for dose titration to gain control of poorly controlled pain.3 If a patient is experiencing mild pain on a specific regimen, the medication dose can be increased up to 25%; by 25% to 50%, if pain is moderate; and 50% to 100%, if severe.5 When the pain is better-controlled, the total amount of pain medication used in 24 hours (24-hour dose) can be converted to a long-acting formulation for more consistent pain management. Because there is a constant component to most advanced pain syndromes, it is recommended that pain medication is given on a standing basis, with as-needed (prn) doses available for exacerbations of pain.3 Prn doses of short-acting medication (equivalent to approximately 10% of the 24-hour dose of medication) should be available at one- or two-hour intervals prn (longer if hepatic or renal impairment is present) for IV or PO medications, respectively.
Opioids often are categorized as low potency (i.e. codeine, hydrocodone) and high-potency (i.e. oxycodone, morphine, hydromorphone, fentanyl). When given in “equianalgesic doses,” the analgesic effect and common side effects (nausea/vomiting, constipation, sedation, confusion, pruritis) of different opioids can vary in different patients. Due to differences in levels of expressed subtypes of opioid receptors, a given patient might be more sensitive to the analgesic effect or side effects of a specific medication. Therefore, if dose escalation of one opioid is inadequate to control pain and further increases in dose are limited by intolerable side effects, rotation to another opioid is recommended.4 Tables documenting equianalgesic doses of different opioids are based on only moderate evidence from equivalency trials performed in healthy volunteers.6 Due to interpatient differences in responses, it is recommended that the equianalgesic dose of the new medication be decreased by 25% to 50% for initial dosing.5
Certain treatments are indicated for specific pain syndromes. Bony metastases respond to NSAIDs, bisphosphonates, and radiation therapy in addition to opioid medications. As focal back pain is the first symptom of spinal cord compression, clinicians should have a high index of suspicion for compression in any patient with malignancy and new back pain. Steroids and radiation therapy are considered emergent treatments for pain control and to prevent paralysis in this circumstance. Pain due to bowel obstruction is usually colicky in nature and responds well to octreotide as discussed in the section on nausea and vomiting. Steroids (such as dexamethasone 4 mg PO bid-tid) might be an effective adjuvant medication in bone pain, tumor pain, or inflammation.
*Half this dose should be used in renal or liver dysfunction and in the elderly.
Preferred in renal dysfunction.
SOURCES: Adapted from Assessment and treatment of physical pain associated with life-limiting illness. Hospice and Palliative Care Training for Physicians: UNIPAC. Vol 3. 3rd ed. Glenview, IL: American Academy of Hospice and Palliative Medicine; 2008, and Evidence-based standards for cancer pain management. J Clin Oncol. 2008;26(23):3879-3885.
Back to the Case
At home, the patient was taking 60 mg of extended-release morphine twice daily and six doses per day of 15-mg immediate-release morphine for breakthrough pain. This is the equivalent of 210 mg of oral morphine in 24 hours. His pain is severe on this regimen, but it is unclear how much of this medication he is absorbing due to his vomiting. Using the IV route of administration and a patient-controlled analgesia (PCA) system will enable rapid dose titration and pain control. The equivalent of the 24-hour dose of 210 mg oral morphine is 70 mg IV morphine, which is equivalent to a drip basal rate of approximately 3 mg IV morphine per hour. This basal rate with a bolus dose of 7 mg (10% of the 24-hour dose) IV morphine q1 hour prn is reasonable as a starting point.
Review of the Data: Nausea and Vomiting
Nausea and vomiting affect 40% to 70% of patients in a palliative setting.7 A thorough history and physical exam can enable one to determine the most likely causes, pathways, and receptors involved in the process of nausea and vomiting. It is important to review the timing, frequency, and triggers of vomiting. The oral, abdominal, neurologic, and rectal exams, in addition to a complete chemistry panel, offer helpful information. The most common etiologies and recommended medications are included in Table 3. It is worthwhile to note that serotonin-antagonists (i.e. ondansetron) are first-line therapies only for chemotherapy and radiation-therapy-induced emesis. If a 24-hour trial of one antiemetic therapy is ineffective, one should reassess the etiology and escalate the antiemetic dose, or add a second therapy with a different (pertinent) mechanism of action. Although most studies of antiemetic therapy are case series, there is good evidence for this mechanistic approach.8
*EPS: extrapyramidal symptoms
The various insults and pathways that can cause vomiting are quite complex. The medullary vomiting center (VC) receives vestibular, peripheral (via splanchnic and vagal nerves), and higher cortical inputs and is the final common pathway in the vomiting reflex. The chemoreceptor trigger zone (CTZ) near the fourth ventricle receives input from the vagal and splanchnic nerves, and generates output to the VC.
General dietary recommendations are to avoid sweet, fatty, and highly salted or spiced foods. Small portions of bland foods without strong odors are best tolerated.7 Constipation commonly contributes to nausea and vomiting and should be managed with disimpaction, enemas, and laxatives as tolerated. Imaging may be required to make the important distinction between partial and complete bowel obstruction, as the treatments differ. Surgical procedures, such as colostomy or placement of a venting gastrostomy tube, can relieve pain and vomiting associated with complete bowel obstruction.
Back to the Case
The patient is found to have a fecal impaction on rectal exam, but vomiting persists after disimpaction and enema use. Imaging documents a complete bowel obstruction at the site of a palpable mass in the right upper quadrant and multiple large hepatic metastases. Octreotide is initiated to decrease intestinal secretions and peristalsis. Steroids are given to decrease tumor burden and associated inflammation in the intestine and liver, as well as to relieve distension of the hepatic capsule. Haloperidol is used in low doses to control episodes of nausea.
Review of the Data: Dyspnea
Dyspnea is a common symptom faced by patients at EOL. An estimated 50% of patients who are evaluated in acute-care hospitals seek treatment for the management of this often-crippling symptom.10 Unfortunately, as disease burden progresses, the incidence of dyspnea increases towards EOL, and the presence and severity of dyspnea is strongly correlated with mortality.
It is imperative for providers to appreciate that dyspnea is a subjective symptom, similar to pain. The presence and severity of dyspnea, therefore, depends on patient report. Given its subjective nature, the degree of dyspnea experienced by a patient might not correlate with objective laboratory findings or test results. In practice, the severity of dyspnea is commonly assessed with a numeric rating scale (0-10), verbal analogue scale, or with verbal descriptors (mild, moderate, severe). It is important to determine the underlying etiology of the dyspnea and, if possible, to target interventions to relieve the underlying cause. However, at the end of life, the burdens of invasive studies to determine the exact cause of dyspnea might outweigh the benefits, and invasive testing might not correlate with patients’ and families’ goals of care. In that instance, the goal of treatment should be aggressive symptom management and providers should use clinical judgment to tailor therapies based on the patient’s underlying illness, physical examination, and perhaps on noninvasive radiological or laboratory findings. Below are nonpharmacological and pharmacological interventions that can be employed to help alleviate dyspnea in the actively dying patient.
Nonpharmacological Management
A handheld fan aimed near the patient’s face has been shown to reduce the sensation of dyspnea.11 This relatively safe and inexpensive intervention has no major side effects and can provide improvement in this distressing symptom.
Often, the first line of therapy in the hospital setting for a patient reporting dyspnea is the administration of oxygen therapy. However, recent evidence does not show superiority of oxygen over air inhalation via nasal prongs for dyspnea in patients with advanced cancer or heart failure.12,13
Pharmacological Management
Opioids are first-line therapy for alleviating dyspnea in patients at EOL. The administration of opioids has been shown in systematic reviews to provide effective management of dyspnea.14,15 Practice guidelines by leading expert groups advocate for the use of opioids in the management of dyspnea for patients with advanced malignant and noncancer diseases.10,16 Fear of causing unintended respiratory sedation with opioids limits the prescription of opioids for dyspnea. However, studies have not found a change in mortality with the use of opioids appropriately titrated to control dyspnea.17
Studies examining the role of benzodiazepines in dyspnea management are conflicting. Anecdotal clinical evidence in actively dying patients supports treating dyspnea with benzodiazepines in conjunction with opioid therapy. Benzodiazepines are most beneficial when there is an anxiety-related component to the dyspnea.
Many patients with advanced disease and evidence of airflow obstruction will benefit from nebulized bronchodilator therapy for dyspnea. Patients with dyspnea from fluid overload (i.e. end-stage congestive heart failure or renal disease) might benefit from systemic diuretics. An increasing number of trials are under way to evaluate the efficacy of nebulized furosemide in the symptomatic management of dyspnea.
Back to the Case
The patient’s clinical course decompensates, and he begins to report worsening dyspnea in addition to his underlying pain. He becomes increasingly anxious about what this new symptom means. In addition to having a discussion about disease progression and prognosis, you increase his PCA basal dose to morphine 4 mg/hour to help him with this new symptom. You also add low-dose lorazepam 0.5 mg IV q8 hours as an adjunct agent for his dyspnea. The patient reports improvement of his symptom burden.
Review of the Data: Secretions
Physiological changes occur as a patient enters the active phase of dying. Two such changes are the loss of the ability to swallow and a reduced cough reflex. These changes culminate in an inability to clear secretions, which pool in the oropharynx and the airways. As the patient breathes, air moves over the pooled secretions and produces a gurgling sound that is referred to as the “death rattle.” The onset of this clinical marker has been shown to have significant prognostic significance for predicting imminent death within a period of hours to days. Proposed treatments for the symptom are listed below.
Nonpharmacological Management
Nonpharmacological options include repositioning the patient in a manner that facilitates postural draining.18 Careful and gentle oral suctioning might help reduce secretions if they are salivary in origin. This will not help to clear deeper bronchial secretions. Suctioning of deeper secretions often causes more burden than benefit, as this can cause repeated trauma and possible bleeding.
Family and caregivers at the bedside can find the “death rattle” quite disturbing and often fear that their loved one is “drowning.” Education and counseling that this is not the case, and that the development of secretions is a natural part of the dying process, can help alleviate this concern. Explaining that pharmacological agents can be titrated to decrease secretions is also reassuring to caregivers.
Pharmacological Management
Pharmacological options for secretion management include utilizing anticholinergic medications to prevent the formation of further secretions. These medications are standard of care for managing the death rattle and have been found to be most efficacious if started earlier in the actively dying phase.19,20 Anticholinergic medications include glycopyrrolate (0.2 mg IV q8 hours), atropine sulfate ophthalmological drops (1% solution, 1-2 drops SL q6 hours), hyoscyamine (0.125 mg one to four times a day), and scopolamine (1.5 mg patch q72 hours). These medications all have possible side effects typical of anticholinergic agents, including delirium, constipation, blurred vision, and urinary retention.
Back to the Case
The patient becomes increasingly lethargic. You meet with his family and explain that he is actively dying. His family reiterates that the goals of medical care should focus on maximizing symptom management. His family is concerned about the “gurgly” sound they hear and want to know if that means he is suffering. You educate the family about expected changes that occur with the dying process and inform them that glycopyrrolate 0.2 mg IV q8 hour will be started to minimize further secretions.
Bottom Line
Pain, nausea, dyspnea, and secretions are common end-of-life symptoms that hospitalists should be competent in treating.
Dr. Litrivis is an associate director and assistant professor at the Mount Sinai School of Medicine in New York, and Dr. Neale is an assistant professor at the University of New Mexico School of Medicine in Albuquerque.
References
- The SUPPORT Principal Investigators. A controlled trial to improve the care for seriously ill hospitalized patients. The study to understand prognoses and preferences for outcomes and risks of treatments (SUPPORT). JAMA. 1995;274(20):1591-1598.
- World Health Organization Definition of Palliative Care. World Health Organization website. Available at: http://www.who.int/cancer/palliative/definition/en/. Accessed April 12, 2012.
- NCCN Guidelines Version 2. 2011 Adult Cancer Pain. National Comprehensive Cancer Network website. Available at: http://www.nccn.org/professionals/physician_gls/pdf/pain.pdf. Accessed April 12, 2012.
- Whitecar PS, Jonas AP, Clasen ME. Managing pain in the dying patient. Am Fam Physician. 2000;61(3):755-764.
- Bial A, Levine S. Assessment and treatment of physical pain associated with life-limiting illness. Hospice and Palliative Care Training for Physicians: UNIPAC. Vol 3. 3rd ed. Glenview, IL: American Academy of Hospice and Palliative Medicine; 2008.
- Sydney M, et al. Evidence-based standards for cancer pain management. J Clin Oncol. 2008;26(23):3879-3885.
- Mannix KA. Gastrointestinal symptoms. In: Doyle D, Hanks G, Cherny N, Calman K, eds. Oxford Textbook of Palliative Medicine. 3rd ed. New York, NY: Oxford University Press; 2005.
- Tyler LS. Nausea and vomiting in palliative care. In: Lipman AG, Jackson KC, Tyler LS, eds. Evidence-Based Symptom Control in Palliative Care. New York, NY: The Hawthorn Press; 2000.
- Policzer JS, Sobel J. Management of Selected Nonpain Symptoms of Life-Limiting Illness. Hospice and Palliative Care Training for Physicians: UNIPAC. Vol 4. 3rd ed. Glenview, IL: American Academy of Hospice and Palliative Medicine; 2008.
- Parshall MB, Schwartzstein RM, Adams L, et al. An official American Thoracic Society statement: update on the mechanisms, assessment, and management of dyspnea. Am J Respir Crit Care Med. 2012;185(4): 435-452.
- Galbraith S, Fagan P, Perkins P, Lynch A, Booth S. Does the use of a handheld fan improve chronic dyspnea? A randomized controlled, crossover trial. J Pain Symptom Manage. 2010;39(5): 831-838.
- Philip J, Gold M, Milner A, Di Iulio J, Miller B, Spruyt O. A randomized, double-blind, crossover trial of the effect of oxygen on dyspnea in patients with advanced cancer. J Pain Symptom Manage. 2006;32(6):541-550.
- Cranston JM, Crockett A, Currow D. Oxygen therapy for dyspnea in adults. Cochrane Database Syst Rev. 2008;(3):CD004769.
- Jennings AL, Davies AN, Higgins JP, Broadley K. Opioids for the palliation of breathlessness in terminal illness. Cochrane Database Syst Rev. 2001;(4):CD002066.
- Ben-Aharon I, Gafter-Gvili A, Paul M, Leibovici, L, Stemmer, SM. Interventions for alleviating cancer-related dyspnea. A systematic review. J Clin Oncol. 2008;26(14): 2396-2404.
- Qaseem A, Snow V, Shekelle P, et al. Evidence-based interventions to improve the palliative care of pain, dyspnea, and depression at the end of life: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2008;148(2):141-146
- Booth S, Moosavi SH, Higginson IJ. The etiology and management of intractable breathlessness in patients with advanced cancer: a systematic review of pharmacological therapy. Nat Clin Pract Oncol. 2008;5(2):90–100.
- Bickel K, Arnold R. EPERC Fast Facts Documents #109 Death Rattle and Oral Secretions, 2nd ed. Available at: http://www.eperc.mcw.edu/EPERC/FastFactsIndex/ff_109.htm. Accessed April 15, 2012.
- Wildiers H, Dhaenekint C, Demeulenaere P, et al. Atropine, hyoscine butylbromide, or scopalamine are equally effective for the treatment of death rattle in terminal care. J Pain Symptom Manage. 2009;38(1):124-133.
- Hugel H, Ellershaw J, Gambles M. Respiratory tract secretions in the dying patient: a comparison between glycopyrronium and hyoscine hydrobromide. J Palliat Med. 2006;9(2):279-285.
Case
A 58-year-old male with colon cancer metastatic to the liver and lungs presents with vomiting, dyspnea, and abdominal pain. His disease has progressed through third-line chemotherapy and his care is now focused entirely on symptom management. He has not had a bowel movement in five days and he began vomiting two days ago.
Overview
The majority of patients in the United States die in acute-care hospitals. The Study to Understand Prognosis and Preferences for Outcomes and Risks of Treatments (SUPPORT), which evaluated the courses of close to 10,000 hospitalized patients with serious and life-limiting illnesses, illustrated that patients’ end-of-life (EOL) experiences often are characterized by poor symptom management and invasive care that is not congruent with the patients’ overall goals of care.1 Studies of factors identified as priorities in EOL care have consistently shown that excellent pain and symptom management are highly valued by patients and families. As the hospitalist movement continues to grow, hospitalists will play a large role in caring for patients at EOL and will need to know how to provide adequate pain and symptom management so that high-quality care can be achieved.
Pain: A Basic Tenet
A basic tenet of palliative medicine is to evaluate and treat all types of suffering.2 Physical pain at EOL is frequently accompanied by other types of pain, such as psychological, social, religious, or existential pain. However, this review will focus on the pharmacologic management of physical pain.
Pain management must begin with a thorough evaluation of the severity, location, and characteristics of the discomfort to assess which therapies are most likely to be beneficial (see Table 1).3 The consistent use of one scale of pain severity (such as 0-10, or mild/moderate/severe) assists in the choice of initial dose of pain medication, in determining the response to the medication, and in assessing the need for change in dose.4
Opioids are the foundation of pain management in advanced diseases because they are available in a number of formulations and, when dosed appropriately, they are effective and safe. Starting doses and equianalgesic doses of common opioids are presented in Table 2. Guidelines recommend the use of short-acting opioids for dose titration to gain control of poorly controlled pain.3 If a patient is experiencing mild pain on a specific regimen, the medication dose can be increased up to 25%; by 25% to 50%, if pain is moderate; and 50% to 100%, if severe.5 When the pain is better-controlled, the total amount of pain medication used in 24 hours (24-hour dose) can be converted to a long-acting formulation for more consistent pain management. Because there is a constant component to most advanced pain syndromes, it is recommended that pain medication is given on a standing basis, with as-needed (prn) doses available for exacerbations of pain.3 Prn doses of short-acting medication (equivalent to approximately 10% of the 24-hour dose of medication) should be available at one- or two-hour intervals prn (longer if hepatic or renal impairment is present) for IV or PO medications, respectively.
Opioids often are categorized as low potency (i.e. codeine, hydrocodone) and high-potency (i.e. oxycodone, morphine, hydromorphone, fentanyl). When given in “equianalgesic doses,” the analgesic effect and common side effects (nausea/vomiting, constipation, sedation, confusion, pruritis) of different opioids can vary in different patients. Due to differences in levels of expressed subtypes of opioid receptors, a given patient might be more sensitive to the analgesic effect or side effects of a specific medication. Therefore, if dose escalation of one opioid is inadequate to control pain and further increases in dose are limited by intolerable side effects, rotation to another opioid is recommended.4 Tables documenting equianalgesic doses of different opioids are based on only moderate evidence from equivalency trials performed in healthy volunteers.6 Due to interpatient differences in responses, it is recommended that the equianalgesic dose of the new medication be decreased by 25% to 50% for initial dosing.5
Certain treatments are indicated for specific pain syndromes. Bony metastases respond to NSAIDs, bisphosphonates, and radiation therapy in addition to opioid medications. As focal back pain is the first symptom of spinal cord compression, clinicians should have a high index of suspicion for compression in any patient with malignancy and new back pain. Steroids and radiation therapy are considered emergent treatments for pain control and to prevent paralysis in this circumstance. Pain due to bowel obstruction is usually colicky in nature and responds well to octreotide as discussed in the section on nausea and vomiting. Steroids (such as dexamethasone 4 mg PO bid-tid) might be an effective adjuvant medication in bone pain, tumor pain, or inflammation.
*Half this dose should be used in renal or liver dysfunction and in the elderly.
Preferred in renal dysfunction.
SOURCES: Adapted from Assessment and treatment of physical pain associated with life-limiting illness. Hospice and Palliative Care Training for Physicians: UNIPAC. Vol 3. 3rd ed. Glenview, IL: American Academy of Hospice and Palliative Medicine; 2008, and Evidence-based standards for cancer pain management. J Clin Oncol. 2008;26(23):3879-3885.
Back to the Case
At home, the patient was taking 60 mg of extended-release morphine twice daily and six doses per day of 15-mg immediate-release morphine for breakthrough pain. This is the equivalent of 210 mg of oral morphine in 24 hours. His pain is severe on this regimen, but it is unclear how much of this medication he is absorbing due to his vomiting. Using the IV route of administration and a patient-controlled analgesia (PCA) system will enable rapid dose titration and pain control. The equivalent of the 24-hour dose of 210 mg oral morphine is 70 mg IV morphine, which is equivalent to a drip basal rate of approximately 3 mg IV morphine per hour. This basal rate with a bolus dose of 7 mg (10% of the 24-hour dose) IV morphine q1 hour prn is reasonable as a starting point.
Review of the Data: Nausea and Vomiting
Nausea and vomiting affect 40% to 70% of patients in a palliative setting.7 A thorough history and physical exam can enable one to determine the most likely causes, pathways, and receptors involved in the process of nausea and vomiting. It is important to review the timing, frequency, and triggers of vomiting. The oral, abdominal, neurologic, and rectal exams, in addition to a complete chemistry panel, offer helpful information. The most common etiologies and recommended medications are included in Table 3. It is worthwhile to note that serotonin-antagonists (i.e. ondansetron) are first-line therapies only for chemotherapy and radiation-therapy-induced emesis. If a 24-hour trial of one antiemetic therapy is ineffective, one should reassess the etiology and escalate the antiemetic dose, or add a second therapy with a different (pertinent) mechanism of action. Although most studies of antiemetic therapy are case series, there is good evidence for this mechanistic approach.8
*EPS: extrapyramidal symptoms
The various insults and pathways that can cause vomiting are quite complex. The medullary vomiting center (VC) receives vestibular, peripheral (via splanchnic and vagal nerves), and higher cortical inputs and is the final common pathway in the vomiting reflex. The chemoreceptor trigger zone (CTZ) near the fourth ventricle receives input from the vagal and splanchnic nerves, and generates output to the VC.
General dietary recommendations are to avoid sweet, fatty, and highly salted or spiced foods. Small portions of bland foods without strong odors are best tolerated.7 Constipation commonly contributes to nausea and vomiting and should be managed with disimpaction, enemas, and laxatives as tolerated. Imaging may be required to make the important distinction between partial and complete bowel obstruction, as the treatments differ. Surgical procedures, such as colostomy or placement of a venting gastrostomy tube, can relieve pain and vomiting associated with complete bowel obstruction.
Back to the Case
The patient is found to have a fecal impaction on rectal exam, but vomiting persists after disimpaction and enema use. Imaging documents a complete bowel obstruction at the site of a palpable mass in the right upper quadrant and multiple large hepatic metastases. Octreotide is initiated to decrease intestinal secretions and peristalsis. Steroids are given to decrease tumor burden and associated inflammation in the intestine and liver, as well as to relieve distension of the hepatic capsule. Haloperidol is used in low doses to control episodes of nausea.
Review of the Data: Dyspnea
Dyspnea is a common symptom faced by patients at EOL. An estimated 50% of patients who are evaluated in acute-care hospitals seek treatment for the management of this often-crippling symptom.10 Unfortunately, as disease burden progresses, the incidence of dyspnea increases towards EOL, and the presence and severity of dyspnea is strongly correlated with mortality.
It is imperative for providers to appreciate that dyspnea is a subjective symptom, similar to pain. The presence and severity of dyspnea, therefore, depends on patient report. Given its subjective nature, the degree of dyspnea experienced by a patient might not correlate with objective laboratory findings or test results. In practice, the severity of dyspnea is commonly assessed with a numeric rating scale (0-10), verbal analogue scale, or with verbal descriptors (mild, moderate, severe). It is important to determine the underlying etiology of the dyspnea and, if possible, to target interventions to relieve the underlying cause. However, at the end of life, the burdens of invasive studies to determine the exact cause of dyspnea might outweigh the benefits, and invasive testing might not correlate with patients’ and families’ goals of care. In that instance, the goal of treatment should be aggressive symptom management and providers should use clinical judgment to tailor therapies based on the patient’s underlying illness, physical examination, and perhaps on noninvasive radiological or laboratory findings. Below are nonpharmacological and pharmacological interventions that can be employed to help alleviate dyspnea in the actively dying patient.
Nonpharmacological Management
A handheld fan aimed near the patient’s face has been shown to reduce the sensation of dyspnea.11 This relatively safe and inexpensive intervention has no major side effects and can provide improvement in this distressing symptom.
Often, the first line of therapy in the hospital setting for a patient reporting dyspnea is the administration of oxygen therapy. However, recent evidence does not show superiority of oxygen over air inhalation via nasal prongs for dyspnea in patients with advanced cancer or heart failure.12,13
Pharmacological Management
Opioids are first-line therapy for alleviating dyspnea in patients at EOL. The administration of opioids has been shown in systematic reviews to provide effective management of dyspnea.14,15 Practice guidelines by leading expert groups advocate for the use of opioids in the management of dyspnea for patients with advanced malignant and noncancer diseases.10,16 Fear of causing unintended respiratory sedation with opioids limits the prescription of opioids for dyspnea. However, studies have not found a change in mortality with the use of opioids appropriately titrated to control dyspnea.17
Studies examining the role of benzodiazepines in dyspnea management are conflicting. Anecdotal clinical evidence in actively dying patients supports treating dyspnea with benzodiazepines in conjunction with opioid therapy. Benzodiazepines are most beneficial when there is an anxiety-related component to the dyspnea.
Many patients with advanced disease and evidence of airflow obstruction will benefit from nebulized bronchodilator therapy for dyspnea. Patients with dyspnea from fluid overload (i.e. end-stage congestive heart failure or renal disease) might benefit from systemic diuretics. An increasing number of trials are under way to evaluate the efficacy of nebulized furosemide in the symptomatic management of dyspnea.
Back to the Case
The patient’s clinical course decompensates, and he begins to report worsening dyspnea in addition to his underlying pain. He becomes increasingly anxious about what this new symptom means. In addition to having a discussion about disease progression and prognosis, you increase his PCA basal dose to morphine 4 mg/hour to help him with this new symptom. You also add low-dose lorazepam 0.5 mg IV q8 hours as an adjunct agent for his dyspnea. The patient reports improvement of his symptom burden.
Review of the Data: Secretions
Physiological changes occur as a patient enters the active phase of dying. Two such changes are the loss of the ability to swallow and a reduced cough reflex. These changes culminate in an inability to clear secretions, which pool in the oropharynx and the airways. As the patient breathes, air moves over the pooled secretions and produces a gurgling sound that is referred to as the “death rattle.” The onset of this clinical marker has been shown to have significant prognostic significance for predicting imminent death within a period of hours to days. Proposed treatments for the symptom are listed below.
Nonpharmacological Management
Nonpharmacological options include repositioning the patient in a manner that facilitates postural draining.18 Careful and gentle oral suctioning might help reduce secretions if they are salivary in origin. This will not help to clear deeper bronchial secretions. Suctioning of deeper secretions often causes more burden than benefit, as this can cause repeated trauma and possible bleeding.
Family and caregivers at the bedside can find the “death rattle” quite disturbing and often fear that their loved one is “drowning.” Education and counseling that this is not the case, and that the development of secretions is a natural part of the dying process, can help alleviate this concern. Explaining that pharmacological agents can be titrated to decrease secretions is also reassuring to caregivers.
Pharmacological Management
Pharmacological options for secretion management include utilizing anticholinergic medications to prevent the formation of further secretions. These medications are standard of care for managing the death rattle and have been found to be most efficacious if started earlier in the actively dying phase.19,20 Anticholinergic medications include glycopyrrolate (0.2 mg IV q8 hours), atropine sulfate ophthalmological drops (1% solution, 1-2 drops SL q6 hours), hyoscyamine (0.125 mg one to four times a day), and scopolamine (1.5 mg patch q72 hours). These medications all have possible side effects typical of anticholinergic agents, including delirium, constipation, blurred vision, and urinary retention.
Back to the Case
The patient becomes increasingly lethargic. You meet with his family and explain that he is actively dying. His family reiterates that the goals of medical care should focus on maximizing symptom management. His family is concerned about the “gurgly” sound they hear and want to know if that means he is suffering. You educate the family about expected changes that occur with the dying process and inform them that glycopyrrolate 0.2 mg IV q8 hour will be started to minimize further secretions.
Bottom Line
Pain, nausea, dyspnea, and secretions are common end-of-life symptoms that hospitalists should be competent in treating.
Dr. Litrivis is an associate director and assistant professor at the Mount Sinai School of Medicine in New York, and Dr. Neale is an assistant professor at the University of New Mexico School of Medicine in Albuquerque.
References
- The SUPPORT Principal Investigators. A controlled trial to improve the care for seriously ill hospitalized patients. The study to understand prognoses and preferences for outcomes and risks of treatments (SUPPORT). JAMA. 1995;274(20):1591-1598.
- World Health Organization Definition of Palliative Care. World Health Organization website. Available at: http://www.who.int/cancer/palliative/definition/en/. Accessed April 12, 2012.
- NCCN Guidelines Version 2. 2011 Adult Cancer Pain. National Comprehensive Cancer Network website. Available at: http://www.nccn.org/professionals/physician_gls/pdf/pain.pdf. Accessed April 12, 2012.
- Whitecar PS, Jonas AP, Clasen ME. Managing pain in the dying patient. Am Fam Physician. 2000;61(3):755-764.
- Bial A, Levine S. Assessment and treatment of physical pain associated with life-limiting illness. Hospice and Palliative Care Training for Physicians: UNIPAC. Vol 3. 3rd ed. Glenview, IL: American Academy of Hospice and Palliative Medicine; 2008.
- Sydney M, et al. Evidence-based standards for cancer pain management. J Clin Oncol. 2008;26(23):3879-3885.
- Mannix KA. Gastrointestinal symptoms. In: Doyle D, Hanks G, Cherny N, Calman K, eds. Oxford Textbook of Palliative Medicine. 3rd ed. New York, NY: Oxford University Press; 2005.
- Tyler LS. Nausea and vomiting in palliative care. In: Lipman AG, Jackson KC, Tyler LS, eds. Evidence-Based Symptom Control in Palliative Care. New York, NY: The Hawthorn Press; 2000.
- Policzer JS, Sobel J. Management of Selected Nonpain Symptoms of Life-Limiting Illness. Hospice and Palliative Care Training for Physicians: UNIPAC. Vol 4. 3rd ed. Glenview, IL: American Academy of Hospice and Palliative Medicine; 2008.
- Parshall MB, Schwartzstein RM, Adams L, et al. An official American Thoracic Society statement: update on the mechanisms, assessment, and management of dyspnea. Am J Respir Crit Care Med. 2012;185(4): 435-452.
- Galbraith S, Fagan P, Perkins P, Lynch A, Booth S. Does the use of a handheld fan improve chronic dyspnea? A randomized controlled, crossover trial. J Pain Symptom Manage. 2010;39(5): 831-838.
- Philip J, Gold M, Milner A, Di Iulio J, Miller B, Spruyt O. A randomized, double-blind, crossover trial of the effect of oxygen on dyspnea in patients with advanced cancer. J Pain Symptom Manage. 2006;32(6):541-550.
- Cranston JM, Crockett A, Currow D. Oxygen therapy for dyspnea in adults. Cochrane Database Syst Rev. 2008;(3):CD004769.
- Jennings AL, Davies AN, Higgins JP, Broadley K. Opioids for the palliation of breathlessness in terminal illness. Cochrane Database Syst Rev. 2001;(4):CD002066.
- Ben-Aharon I, Gafter-Gvili A, Paul M, Leibovici, L, Stemmer, SM. Interventions for alleviating cancer-related dyspnea. A systematic review. J Clin Oncol. 2008;26(14): 2396-2404.
- Qaseem A, Snow V, Shekelle P, et al. Evidence-based interventions to improve the palliative care of pain, dyspnea, and depression at the end of life: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2008;148(2):141-146
- Booth S, Moosavi SH, Higginson IJ. The etiology and management of intractable breathlessness in patients with advanced cancer: a systematic review of pharmacological therapy. Nat Clin Pract Oncol. 2008;5(2):90–100.
- Bickel K, Arnold R. EPERC Fast Facts Documents #109 Death Rattle and Oral Secretions, 2nd ed. Available at: http://www.eperc.mcw.edu/EPERC/FastFactsIndex/ff_109.htm. Accessed April 15, 2012.
- Wildiers H, Dhaenekint C, Demeulenaere P, et al. Atropine, hyoscine butylbromide, or scopalamine are equally effective for the treatment of death rattle in terminal care. J Pain Symptom Manage. 2009;38(1):124-133.
- Hugel H, Ellershaw J, Gambles M. Respiratory tract secretions in the dying patient: a comparison between glycopyrronium and hyoscine hydrobromide. J Palliat Med. 2006;9(2):279-285.
Case
A 58-year-old male with colon cancer metastatic to the liver and lungs presents with vomiting, dyspnea, and abdominal pain. His disease has progressed through third-line chemotherapy and his care is now focused entirely on symptom management. He has not had a bowel movement in five days and he began vomiting two days ago.
Overview
The majority of patients in the United States die in acute-care hospitals. The Study to Understand Prognosis and Preferences for Outcomes and Risks of Treatments (SUPPORT), which evaluated the courses of close to 10,000 hospitalized patients with serious and life-limiting illnesses, illustrated that patients’ end-of-life (EOL) experiences often are characterized by poor symptom management and invasive care that is not congruent with the patients’ overall goals of care.1 Studies of factors identified as priorities in EOL care have consistently shown that excellent pain and symptom management are highly valued by patients and families. As the hospitalist movement continues to grow, hospitalists will play a large role in caring for patients at EOL and will need to know how to provide adequate pain and symptom management so that high-quality care can be achieved.
Pain: A Basic Tenet
A basic tenet of palliative medicine is to evaluate and treat all types of suffering.2 Physical pain at EOL is frequently accompanied by other types of pain, such as psychological, social, religious, or existential pain. However, this review will focus on the pharmacologic management of physical pain.
Pain management must begin with a thorough evaluation of the severity, location, and characteristics of the discomfort to assess which therapies are most likely to be beneficial (see Table 1).3 The consistent use of one scale of pain severity (such as 0-10, or mild/moderate/severe) assists in the choice of initial dose of pain medication, in determining the response to the medication, and in assessing the need for change in dose.4
Opioids are the foundation of pain management in advanced diseases because they are available in a number of formulations and, when dosed appropriately, they are effective and safe. Starting doses and equianalgesic doses of common opioids are presented in Table 2. Guidelines recommend the use of short-acting opioids for dose titration to gain control of poorly controlled pain.3 If a patient is experiencing mild pain on a specific regimen, the medication dose can be increased up to 25%; by 25% to 50%, if pain is moderate; and 50% to 100%, if severe.5 When the pain is better-controlled, the total amount of pain medication used in 24 hours (24-hour dose) can be converted to a long-acting formulation for more consistent pain management. Because there is a constant component to most advanced pain syndromes, it is recommended that pain medication is given on a standing basis, with as-needed (prn) doses available for exacerbations of pain.3 Prn doses of short-acting medication (equivalent to approximately 10% of the 24-hour dose of medication) should be available at one- or two-hour intervals prn (longer if hepatic or renal impairment is present) for IV or PO medications, respectively.
Opioids often are categorized as low potency (i.e. codeine, hydrocodone) and high-potency (i.e. oxycodone, morphine, hydromorphone, fentanyl). When given in “equianalgesic doses,” the analgesic effect and common side effects (nausea/vomiting, constipation, sedation, confusion, pruritis) of different opioids can vary in different patients. Due to differences in levels of expressed subtypes of opioid receptors, a given patient might be more sensitive to the analgesic effect or side effects of a specific medication. Therefore, if dose escalation of one opioid is inadequate to control pain and further increases in dose are limited by intolerable side effects, rotation to another opioid is recommended.4 Tables documenting equianalgesic doses of different opioids are based on only moderate evidence from equivalency trials performed in healthy volunteers.6 Due to interpatient differences in responses, it is recommended that the equianalgesic dose of the new medication be decreased by 25% to 50% for initial dosing.5
Certain treatments are indicated for specific pain syndromes. Bony metastases respond to NSAIDs, bisphosphonates, and radiation therapy in addition to opioid medications. As focal back pain is the first symptom of spinal cord compression, clinicians should have a high index of suspicion for compression in any patient with malignancy and new back pain. Steroids and radiation therapy are considered emergent treatments for pain control and to prevent paralysis in this circumstance. Pain due to bowel obstruction is usually colicky in nature and responds well to octreotide as discussed in the section on nausea and vomiting. Steroids (such as dexamethasone 4 mg PO bid-tid) might be an effective adjuvant medication in bone pain, tumor pain, or inflammation.
*Half this dose should be used in renal or liver dysfunction and in the elderly.
Preferred in renal dysfunction.
SOURCES: Adapted from Assessment and treatment of physical pain associated with life-limiting illness. Hospice and Palliative Care Training for Physicians: UNIPAC. Vol 3. 3rd ed. Glenview, IL: American Academy of Hospice and Palliative Medicine; 2008, and Evidence-based standards for cancer pain management. J Clin Oncol. 2008;26(23):3879-3885.
Back to the Case
At home, the patient was taking 60 mg of extended-release morphine twice daily and six doses per day of 15-mg immediate-release morphine for breakthrough pain. This is the equivalent of 210 mg of oral morphine in 24 hours. His pain is severe on this regimen, but it is unclear how much of this medication he is absorbing due to his vomiting. Using the IV route of administration and a patient-controlled analgesia (PCA) system will enable rapid dose titration and pain control. The equivalent of the 24-hour dose of 210 mg oral morphine is 70 mg IV morphine, which is equivalent to a drip basal rate of approximately 3 mg IV morphine per hour. This basal rate with a bolus dose of 7 mg (10% of the 24-hour dose) IV morphine q1 hour prn is reasonable as a starting point.
Review of the Data: Nausea and Vomiting
Nausea and vomiting affect 40% to 70% of patients in a palliative setting.7 A thorough history and physical exam can enable one to determine the most likely causes, pathways, and receptors involved in the process of nausea and vomiting. It is important to review the timing, frequency, and triggers of vomiting. The oral, abdominal, neurologic, and rectal exams, in addition to a complete chemistry panel, offer helpful information. The most common etiologies and recommended medications are included in Table 3. It is worthwhile to note that serotonin-antagonists (i.e. ondansetron) are first-line therapies only for chemotherapy and radiation-therapy-induced emesis. If a 24-hour trial of one antiemetic therapy is ineffective, one should reassess the etiology and escalate the antiemetic dose, or add a second therapy with a different (pertinent) mechanism of action. Although most studies of antiemetic therapy are case series, there is good evidence for this mechanistic approach.8
*EPS: extrapyramidal symptoms
The various insults and pathways that can cause vomiting are quite complex. The medullary vomiting center (VC) receives vestibular, peripheral (via splanchnic and vagal nerves), and higher cortical inputs and is the final common pathway in the vomiting reflex. The chemoreceptor trigger zone (CTZ) near the fourth ventricle receives input from the vagal and splanchnic nerves, and generates output to the VC.
General dietary recommendations are to avoid sweet, fatty, and highly salted or spiced foods. Small portions of bland foods without strong odors are best tolerated.7 Constipation commonly contributes to nausea and vomiting and should be managed with disimpaction, enemas, and laxatives as tolerated. Imaging may be required to make the important distinction between partial and complete bowel obstruction, as the treatments differ. Surgical procedures, such as colostomy or placement of a venting gastrostomy tube, can relieve pain and vomiting associated with complete bowel obstruction.
Back to the Case
The patient is found to have a fecal impaction on rectal exam, but vomiting persists after disimpaction and enema use. Imaging documents a complete bowel obstruction at the site of a palpable mass in the right upper quadrant and multiple large hepatic metastases. Octreotide is initiated to decrease intestinal secretions and peristalsis. Steroids are given to decrease tumor burden and associated inflammation in the intestine and liver, as well as to relieve distension of the hepatic capsule. Haloperidol is used in low doses to control episodes of nausea.
Review of the Data: Dyspnea
Dyspnea is a common symptom faced by patients at EOL. An estimated 50% of patients who are evaluated in acute-care hospitals seek treatment for the management of this often-crippling symptom.10 Unfortunately, as disease burden progresses, the incidence of dyspnea increases towards EOL, and the presence and severity of dyspnea is strongly correlated with mortality.
It is imperative for providers to appreciate that dyspnea is a subjective symptom, similar to pain. The presence and severity of dyspnea, therefore, depends on patient report. Given its subjective nature, the degree of dyspnea experienced by a patient might not correlate with objective laboratory findings or test results. In practice, the severity of dyspnea is commonly assessed with a numeric rating scale (0-10), verbal analogue scale, or with verbal descriptors (mild, moderate, severe). It is important to determine the underlying etiology of the dyspnea and, if possible, to target interventions to relieve the underlying cause. However, at the end of life, the burdens of invasive studies to determine the exact cause of dyspnea might outweigh the benefits, and invasive testing might not correlate with patients’ and families’ goals of care. In that instance, the goal of treatment should be aggressive symptom management and providers should use clinical judgment to tailor therapies based on the patient’s underlying illness, physical examination, and perhaps on noninvasive radiological or laboratory findings. Below are nonpharmacological and pharmacological interventions that can be employed to help alleviate dyspnea in the actively dying patient.
Nonpharmacological Management
A handheld fan aimed near the patient’s face has been shown to reduce the sensation of dyspnea.11 This relatively safe and inexpensive intervention has no major side effects and can provide improvement in this distressing symptom.
Often, the first line of therapy in the hospital setting for a patient reporting dyspnea is the administration of oxygen therapy. However, recent evidence does not show superiority of oxygen over air inhalation via nasal prongs for dyspnea in patients with advanced cancer or heart failure.12,13
Pharmacological Management
Opioids are first-line therapy for alleviating dyspnea in patients at EOL. The administration of opioids has been shown in systematic reviews to provide effective management of dyspnea.14,15 Practice guidelines by leading expert groups advocate for the use of opioids in the management of dyspnea for patients with advanced malignant and noncancer diseases.10,16 Fear of causing unintended respiratory sedation with opioids limits the prescription of opioids for dyspnea. However, studies have not found a change in mortality with the use of opioids appropriately titrated to control dyspnea.17
Studies examining the role of benzodiazepines in dyspnea management are conflicting. Anecdotal clinical evidence in actively dying patients supports treating dyspnea with benzodiazepines in conjunction with opioid therapy. Benzodiazepines are most beneficial when there is an anxiety-related component to the dyspnea.
Many patients with advanced disease and evidence of airflow obstruction will benefit from nebulized bronchodilator therapy for dyspnea. Patients with dyspnea from fluid overload (i.e. end-stage congestive heart failure or renal disease) might benefit from systemic diuretics. An increasing number of trials are under way to evaluate the efficacy of nebulized furosemide in the symptomatic management of dyspnea.
Back to the Case
The patient’s clinical course decompensates, and he begins to report worsening dyspnea in addition to his underlying pain. He becomes increasingly anxious about what this new symptom means. In addition to having a discussion about disease progression and prognosis, you increase his PCA basal dose to morphine 4 mg/hour to help him with this new symptom. You also add low-dose lorazepam 0.5 mg IV q8 hours as an adjunct agent for his dyspnea. The patient reports improvement of his symptom burden.
Review of the Data: Secretions
Physiological changes occur as a patient enters the active phase of dying. Two such changes are the loss of the ability to swallow and a reduced cough reflex. These changes culminate in an inability to clear secretions, which pool in the oropharynx and the airways. As the patient breathes, air moves over the pooled secretions and produces a gurgling sound that is referred to as the “death rattle.” The onset of this clinical marker has been shown to have significant prognostic significance for predicting imminent death within a period of hours to days. Proposed treatments for the symptom are listed below.
Nonpharmacological Management
Nonpharmacological options include repositioning the patient in a manner that facilitates postural draining.18 Careful and gentle oral suctioning might help reduce secretions if they are salivary in origin. This will not help to clear deeper bronchial secretions. Suctioning of deeper secretions often causes more burden than benefit, as this can cause repeated trauma and possible bleeding.
Family and caregivers at the bedside can find the “death rattle” quite disturbing and often fear that their loved one is “drowning.” Education and counseling that this is not the case, and that the development of secretions is a natural part of the dying process, can help alleviate this concern. Explaining that pharmacological agents can be titrated to decrease secretions is also reassuring to caregivers.
Pharmacological Management
Pharmacological options for secretion management include utilizing anticholinergic medications to prevent the formation of further secretions. These medications are standard of care for managing the death rattle and have been found to be most efficacious if started earlier in the actively dying phase.19,20 Anticholinergic medications include glycopyrrolate (0.2 mg IV q8 hours), atropine sulfate ophthalmological drops (1% solution, 1-2 drops SL q6 hours), hyoscyamine (0.125 mg one to four times a day), and scopolamine (1.5 mg patch q72 hours). These medications all have possible side effects typical of anticholinergic agents, including delirium, constipation, blurred vision, and urinary retention.
Back to the Case
The patient becomes increasingly lethargic. You meet with his family and explain that he is actively dying. His family reiterates that the goals of medical care should focus on maximizing symptom management. His family is concerned about the “gurgly” sound they hear and want to know if that means he is suffering. You educate the family about expected changes that occur with the dying process and inform them that glycopyrrolate 0.2 mg IV q8 hour will be started to minimize further secretions.
Bottom Line
Pain, nausea, dyspnea, and secretions are common end-of-life symptoms that hospitalists should be competent in treating.
Dr. Litrivis is an associate director and assistant professor at the Mount Sinai School of Medicine in New York, and Dr. Neale is an assistant professor at the University of New Mexico School of Medicine in Albuquerque.
References
- The SUPPORT Principal Investigators. A controlled trial to improve the care for seriously ill hospitalized patients. The study to understand prognoses and preferences for outcomes and risks of treatments (SUPPORT). JAMA. 1995;274(20):1591-1598.
- World Health Organization Definition of Palliative Care. World Health Organization website. Available at: http://www.who.int/cancer/palliative/definition/en/. Accessed April 12, 2012.
- NCCN Guidelines Version 2. 2011 Adult Cancer Pain. National Comprehensive Cancer Network website. Available at: http://www.nccn.org/professionals/physician_gls/pdf/pain.pdf. Accessed April 12, 2012.
- Whitecar PS, Jonas AP, Clasen ME. Managing pain in the dying patient. Am Fam Physician. 2000;61(3):755-764.
- Bial A, Levine S. Assessment and treatment of physical pain associated with life-limiting illness. Hospice and Palliative Care Training for Physicians: UNIPAC. Vol 3. 3rd ed. Glenview, IL: American Academy of Hospice and Palliative Medicine; 2008.
- Sydney M, et al. Evidence-based standards for cancer pain management. J Clin Oncol. 2008;26(23):3879-3885.
- Mannix KA. Gastrointestinal symptoms. In: Doyle D, Hanks G, Cherny N, Calman K, eds. Oxford Textbook of Palliative Medicine. 3rd ed. New York, NY: Oxford University Press; 2005.
- Tyler LS. Nausea and vomiting in palliative care. In: Lipman AG, Jackson KC, Tyler LS, eds. Evidence-Based Symptom Control in Palliative Care. New York, NY: The Hawthorn Press; 2000.
- Policzer JS, Sobel J. Management of Selected Nonpain Symptoms of Life-Limiting Illness. Hospice and Palliative Care Training for Physicians: UNIPAC. Vol 4. 3rd ed. Glenview, IL: American Academy of Hospice and Palliative Medicine; 2008.
- Parshall MB, Schwartzstein RM, Adams L, et al. An official American Thoracic Society statement: update on the mechanisms, assessment, and management of dyspnea. Am J Respir Crit Care Med. 2012;185(4): 435-452.
- Galbraith S, Fagan P, Perkins P, Lynch A, Booth S. Does the use of a handheld fan improve chronic dyspnea? A randomized controlled, crossover trial. J Pain Symptom Manage. 2010;39(5): 831-838.
- Philip J, Gold M, Milner A, Di Iulio J, Miller B, Spruyt O. A randomized, double-blind, crossover trial of the effect of oxygen on dyspnea in patients with advanced cancer. J Pain Symptom Manage. 2006;32(6):541-550.
- Cranston JM, Crockett A, Currow D. Oxygen therapy for dyspnea in adults. Cochrane Database Syst Rev. 2008;(3):CD004769.
- Jennings AL, Davies AN, Higgins JP, Broadley K. Opioids for the palliation of breathlessness in terminal illness. Cochrane Database Syst Rev. 2001;(4):CD002066.
- Ben-Aharon I, Gafter-Gvili A, Paul M, Leibovici, L, Stemmer, SM. Interventions for alleviating cancer-related dyspnea. A systematic review. J Clin Oncol. 2008;26(14): 2396-2404.
- Qaseem A, Snow V, Shekelle P, et al. Evidence-based interventions to improve the palliative care of pain, dyspnea, and depression at the end of life: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2008;148(2):141-146
- Booth S, Moosavi SH, Higginson IJ. The etiology and management of intractable breathlessness in patients with advanced cancer: a systematic review of pharmacological therapy. Nat Clin Pract Oncol. 2008;5(2):90–100.
- Bickel K, Arnold R. EPERC Fast Facts Documents #109 Death Rattle and Oral Secretions, 2nd ed. Available at: http://www.eperc.mcw.edu/EPERC/FastFactsIndex/ff_109.htm. Accessed April 15, 2012.
- Wildiers H, Dhaenekint C, Demeulenaere P, et al. Atropine, hyoscine butylbromide, or scopalamine are equally effective for the treatment of death rattle in terminal care. J Pain Symptom Manage. 2009;38(1):124-133.
- Hugel H, Ellershaw J, Gambles M. Respiratory tract secretions in the dying patient: a comparison between glycopyrronium and hyoscine hydrobromide. J Palliat Med. 2006;9(2):279-285.
How Should Physicians Assess and Manage Pressure Ulcers in the Hospitalized Patient?
The Case
An 85-year-old woman with stroke, functional quadriplegia, and diabetes mellitus presents with altered mental status. She is febrile (38.5°C) with leukocytosis (14,400 cells/mm3) and has a 5 cm x 4 cm x 2 cm Stage III malodorous sacral ulcer without surrounding erythema, tunneling, or pain. The ulcer base is partially covered by green slough. How should this pressure ulcer be evaluated and treated?
Overview
Pressure ulcers in vulnerable populations, such as the elderly and those with limited mobility, are exceedingly common. In the acute-care setting, the incidence of pressure ulcers ranges from 0.4% to 38%, with 2.5 million cases treated annually at an estimated cost of $11 billion per year.1,2 Moreover, as of Oct. 1, 2008, the Centers for Medicare & Medicaid Services (CMS) guideline states that hospitals will no longer receive additional payment when a hospitalized patient develops Stage III or IV pressure ulcers that are not present on admission.
A pressure ulcer is a localized injury to skin and underlying soft tissue over a bony prominence due to sustained external pressure.3 Prolonged pressure on these weight-bearing areas leads to reduced blood flow, ischemia, cell death, and necrosis of local tissues.4 Risk factors for developing pressure ulcers include increased external pressure, shear, friction, moisture, poor perfusion, immobility, incontinence, malnutrition, and impaired mental status.4 Inadequately treated pressure ulcers can lead to pain, tunneling, fistula formation, disfigurement, infection, prolonged hospitalization, lower quality of life, and increased mortality.4
Because of the significant morbidities and high costs associated with the care of pressure ulcers in acute care, hospitalists must be familiar with the assessment and treatment of pressure ulcers in vulnerable patients.
Review of the Data
The management of pressure ulcers in the hospitalized patient starts with a comprehensive assessment of the patient’s medical comorbidities, risk factors, and wound-staging. Considerations must be given to differentiate an infected pressure ulcer from a noninfected ulcer. These evaluations then guide the appropriate treatments of pressure ulcers, including the prevention of progression or formation of new ulcers, debridement, application of wound dressing, and antibiotic use.
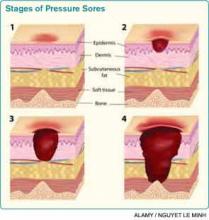
Assessing pressure ulcer stage. The National Pressure Ulcer Advisory Panel (NPUAP) Classification System is the most commonly used staging tool. It describes four stages of pressure ulcers (see Table 1).3 A Stage 1 pressure ulcer is characterized by intact skin with nonblanchable erythema and may be discolored, painful, soft, firm, and warmer or cooler compared to adjacent area. A Stage II pressure ulcer presents with partial thickness skin loss with a shallow red-pink wound bed without slough, or as an intact or ruptured serum-filled blister. Stage II pressure ulcers do not include skin tears, tape burns, macerations, or excoriations. A Stage III pressure ulcer has full thickness skin loss with or without visible subcutaneous fat. Bone, tendon, or muscle are not exposed or directly palpable. Slough may be present but it does not obscure the depth of ulcer. Deep ulcers can develop in anatomical regions with high adiposity, such as the pelvic girdle. A Stage IV pressure ulcer has full thickness tissue loss with exposed and palpable bone, tendon, or muscle. Slough, eschar, undermining, and tunneling may be present. The depth of a Stage IV ulcer varies depending on anatomical location and adiposity. Stage IV ulcers also create a nidus for osteomyelitis.
NPUAP describes two additional categories of pressure ulcers: unstageable and deep tissue injury.3 An unstageable ulcer has full thickness skin or tissue loss of unknown depth because the wound base is completely obscured by slough or eschar. The ulcer can only be accurately categorized as Stage III or IV after sufficient slough or eschar is removed to identify wound depth. Lastly, suspected deep tissue injury describes a localized area of discolored intact skin (purple or maroon) or blood-filled blister due to damage of underlying tissue from pressure or shear.
Diagnosing infected pressure ulcers. Pressure ulcer infection delays wound healing and increases risks for sepsis, cellulitis, osteomyelitis, and death.5,6 Clinical evidence of soft tissue involvement, such as erythema, warmth, tenderness, foul odor, or purulent discharge, and systemic inflammatory response (fever, tachycardia, or leukocytosis) are suggestive of a wound infection.3,5 However, these clinical signs may be absent and thus make the distinction between chronic wound and infected pressure ulcer difficult.7 Delayed healing with friable granulation tissue and increased pain in a treated wound may be the only signs of a pressure ulcer infection.3,5,7
Routine laboratory tests (i.e. white blood cell count, C-reactive protein, and erythrocyte sedimentation rate) are neither sensitive nor specific in diagnosing wound infection. Moreover, because pressure ulcers are typically colonized with ≥105 organisms/mL of normal skin flora and bacteria from adjacent gastrointestinal or urogenital environments, swab cultures identify colonizing organisms and are not recommended as a diagnostic test for pressure ulcer microbiologic evaluation.5,6 If microbiological data are needed to guide antibiotic use, cultures of blood, bone, or deep tissue biopsied from a surgically debrided wound should be used.5 Importantly, a higher index of suspicion should be maintained for infection of Stage III or IV pressure ulcers because they are more commonly infected than Stage I or II ulcers.3
Prevention. The prevention of wound progression is essential in treating acute, chronic, or infected pressure ulcers. Although management guidelines are limited by few high-quality, randomized controlled trials, NPUAP recommends a number of prevention strategies targeting risk factors that contribute to pressure ulcer development.2,3,8
For all bed-bound and chair-bound persons with impaired ability to self-reposition, risk assessment for pressure ulcer should be done on admission and repeated every 24 hours. The presence of such risk factors as immobility, shear, friction, moisture, incontinence, and malnutrition should be used to guide preventive treatments. Pressure relief on an ulcer can be achieved by repositioning the immobile patient at one- to two-hour intervals. Pressure-redistributing support surfaces (static, overlays, or dynamic) reduce tissue pressure and decrease overall incidence of pressure ulcers. Due to a lack of relative efficacy data, the selection of a support surface should be determined by the patient’s individual needs in order to reduce pressure and shear.3 For instance, dynamic support is an appropriate surface for an immobile patient with multiple or nonhealing ulcers. Shearing force and friction can be reduced by limiting head-of-bed elevation to <30° and using such transfer aids as bed linens while repositioning patients. The use of pillows, foam wedges, or other devices should be used to eliminate direct contact of bony prominences or reduce pressure on heels.8
Skin care should be optimized to limit excessive dryness or moisture. This includes using moisturizers for dry skin, particularly for the sacrum, and implementing bowel and bladder programs and absorbent underpads in patients with bowel or bladder incontinence.2 Given that patients with pressure ulcers are in a catabolic state, those who are nutritionally compromised may benefit from nutritional supplementation.3 Lastly, appropriate use of local and systemic pain regimen for painful pressure ulcers can improve patient cooperation in repositioning, dressing change, and quality of life.
Debridement. Wound debridement removes necrotic tissue often present in infected or chronic pressure ulcers, reduces risk for further infection, and promotes granulation tissue formation and wound healing. Debridement, however, is not indicated for ulcers of an ischemic limb or dry eschar of the heel, due to propensity for complications.3,4 The five common debridement methods are sharp, mechanical, autolytic, enzymatic, and biosurgical. The debridement method of choice is determined by clinician preference and availability.4
Sharp debridement results in rapid removal of large amounts of nonviable necrotic tissues and eschar using sharp instruments and, therefore, is indicated if wound infection or sepsis is present. Mechanical debridement by wet-to-dry dressing or whirlpool nonselectively removes granulation tissue and, thus, should be used cautiously. Autolytic debridement uses occlusive dressings (i.e. hydrocolloid or hydrogel) to maintain a moist wound environment in order to optimize the body’s inherent ability to selectively self-digest necrotic tissues. Enzymatic debridement with concentrated topical proteolytic enzymes (i.e. collagenase) digests necrotic tissues and achieves faster debridement than autolysis while being less invasive than surgical intervention. Biosurgery utilizes maggots (i.e. Lucilia sericata) that produce enzymes to effectively debride necrotic tissues.
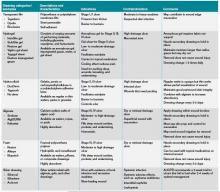
Wound care and dressing. Pressure ulcers should be cleansed with each dressing change using such physiologic solutions as normal saline. Cleansing with antimicrobial solutions for ulcers with large necrotic debris or infection needs to be thoughtfully administered due to the potential impairment on wound healing.4 Wound dressing should maintain a moist wound environment to allow epithelialization and limit excessive exudates in order to prevent maceration. Although there are many categories of moisture retentive dressings, their comparative effectiveness remain unclear.4 Table 2 summarizes characteristics of common wound dressings and their applications.
Antibiotic use. Topical antibiotics are appropriate for Stage III or IV ulcers with signs of local infection, including periwound erythema and friable granulation tissue.4 The Agency for Health Care Policy and Research recommends a two-week trial of a topical antibiotic, such as silver sulfadiazine, for pressure ulcers that fail to heal after two to four weeks of optimal care.6 Systemic antibiotics should be used for patients who demonstrate evidence of systemic infection including sepsis, osteomyelitis, or cellulitis with associated fever and leukocytosis. The choice of systemic antibiotics should be based on cultures from blood, bone, or deep tissue biopsied from a surgically debrided wound.4,6
Back to the Case
The patient was hospitalized for altered mental status. She was at high risk for the progression of her sacral ulcer and the development of new pressure ulcers due to immobility, incontinence, malnutrition, and impaired mental status. The sacral wound was a chronic, Stage III pressure ulcer without evidence of local tissue infection. However, the presence of leukocytosis and fever were suggestive of an underlying infection. Her urine analysis was consistent with a urinary tract infection.
Trimethoprim/sulfamethoxazole was administered with subsequent resolution of leukocytosis, fever, and delirium. The sacral ulcer was cleansed with normal saline and covered with hydrocolloid dressing every 72 hours in order to maintain a moist wound environment and facilitate autolysis. Preventive interventions guided by her risk factors for pressure ulcer were implemented. Interventions included:
- Daily skin and wound assessment;
- Pressure relief with repositioning every two hours;
- Use of a dynamic support surface;
- Head-of-bed elevation of no more than <30° to reduce shear and friction;
- Use of transfer aids;
- Use of devices to eliminate direct contact of bony prominences;
- Optimizing skin care with moisturizers for dry skin and frequent changing of absorbent under pads for incontinence; and
- Consulting nutrition service to optimize nutritional intake.
Bottom Line
Assessments of pressure ulcer stage, wound infection, and risk factors guide targeted therapeutic interventions that facilitate wound healing and prevent new pressure ulcer formation.
Dr. Prager is a fellow in the Brookdale Department of Geriatrics and Palliative Medicine at Mount Sinai School of Medicine in New York City. Dr. Ko is a hospitalist and an assistant professor in the Brookdale Department of Geriatrics and Palliative Medicine at Mount Sinai.
References
- Pressure ulcers in America: prevalence, incidence, and implications for the future. An executive summary of the National Pressure Ulcer Advisory Panel monograph. Adv Skin Wound Care. 2001;14(4):208-215.
- Reddy M, Gill SS, Rochon PA. Preventing pressure ulcers: a systematic review. JAMA. 2006;296(8):974-984.
- European Pressure Ulcer Advisory Panel and National Pressure Ulcer Advisory Panel. Treatment of Pressure Ulcers: Quick Reference Guide. Washington, D.C.: National Pressure Ulcer Advisory Panel; 2009.
- Bates-Jensen BM. Chapter 58. Pressure Ulcers. In: Halter JB, Ouslander JG, Tinetti ME, Studenski S, High KP, Asthana S, eds. Hazzard’s Geriatric Medicine and Gerontology. 6th ed. New York: McGraw-Hill; 2009.
- Livesley NJ, Chow AW. Infected pressure ulcers in elderly individuals. Clin Infect Dis. 2002;35(11):1390-1396.
- Agency for Health Care Policy and Research (AHCPR). Treatment of Pressure Ulcers. Clinical Practice Guideline Number 15. U.S. Department of Health and Human Services. 1994.
- Reddy M, Gill SS, Wu W, Kalkar SR, Rochon PA. Does this patient have an infection of a chronic wound? JAMA. 2012;307(6):605-611.
- National Pressure Ulcer Advisory Panel. Pressure Ulcer Prevention Points. National Pressure Ulcer Advisory Panel website. Available at: http://www.npuap.org/resources/educational-and-clinical-resources/pressure-ulcer-prevention-points/. Accessed Aug. 1, 2012.
- Reuben DB, Herr KA, Pacala JT, et al. Skin Ulcers. In: Geriatrics At Your Fingertips. 12th ed. New York: The American Geriatrics Society; 2010.
The Case
An 85-year-old woman with stroke, functional quadriplegia, and diabetes mellitus presents with altered mental status. She is febrile (38.5°C) with leukocytosis (14,400 cells/mm3) and has a 5 cm x 4 cm x 2 cm Stage III malodorous sacral ulcer without surrounding erythema, tunneling, or pain. The ulcer base is partially covered by green slough. How should this pressure ulcer be evaluated and treated?
Overview
Pressure ulcers in vulnerable populations, such as the elderly and those with limited mobility, are exceedingly common. In the acute-care setting, the incidence of pressure ulcers ranges from 0.4% to 38%, with 2.5 million cases treated annually at an estimated cost of $11 billion per year.1,2 Moreover, as of Oct. 1, 2008, the Centers for Medicare & Medicaid Services (CMS) guideline states that hospitals will no longer receive additional payment when a hospitalized patient develops Stage III or IV pressure ulcers that are not present on admission.
A pressure ulcer is a localized injury to skin and underlying soft tissue over a bony prominence due to sustained external pressure.3 Prolonged pressure on these weight-bearing areas leads to reduced blood flow, ischemia, cell death, and necrosis of local tissues.4 Risk factors for developing pressure ulcers include increased external pressure, shear, friction, moisture, poor perfusion, immobility, incontinence, malnutrition, and impaired mental status.4 Inadequately treated pressure ulcers can lead to pain, tunneling, fistula formation, disfigurement, infection, prolonged hospitalization, lower quality of life, and increased mortality.4
Because of the significant morbidities and high costs associated with the care of pressure ulcers in acute care, hospitalists must be familiar with the assessment and treatment of pressure ulcers in vulnerable patients.
Review of the Data
The management of pressure ulcers in the hospitalized patient starts with a comprehensive assessment of the patient’s medical comorbidities, risk factors, and wound-staging. Considerations must be given to differentiate an infected pressure ulcer from a noninfected ulcer. These evaluations then guide the appropriate treatments of pressure ulcers, including the prevention of progression or formation of new ulcers, debridement, application of wound dressing, and antibiotic use.
Assessing pressure ulcer stage. The National Pressure Ulcer Advisory Panel (NPUAP) Classification System is the most commonly used staging tool. It describes four stages of pressure ulcers (see Table 1).3 A Stage 1 pressure ulcer is characterized by intact skin with nonblanchable erythema and may be discolored, painful, soft, firm, and warmer or cooler compared to adjacent area. A Stage II pressure ulcer presents with partial thickness skin loss with a shallow red-pink wound bed without slough, or as an intact or ruptured serum-filled blister. Stage II pressure ulcers do not include skin tears, tape burns, macerations, or excoriations. A Stage III pressure ulcer has full thickness skin loss with or without visible subcutaneous fat. Bone, tendon, or muscle are not exposed or directly palpable. Slough may be present but it does not obscure the depth of ulcer. Deep ulcers can develop in anatomical regions with high adiposity, such as the pelvic girdle. A Stage IV pressure ulcer has full thickness tissue loss with exposed and palpable bone, tendon, or muscle. Slough, eschar, undermining, and tunneling may be present. The depth of a Stage IV ulcer varies depending on anatomical location and adiposity. Stage IV ulcers also create a nidus for osteomyelitis.
NPUAP describes two additional categories of pressure ulcers: unstageable and deep tissue injury.3 An unstageable ulcer has full thickness skin or tissue loss of unknown depth because the wound base is completely obscured by slough or eschar. The ulcer can only be accurately categorized as Stage III or IV after sufficient slough or eschar is removed to identify wound depth. Lastly, suspected deep tissue injury describes a localized area of discolored intact skin (purple or maroon) or blood-filled blister due to damage of underlying tissue from pressure or shear.
Diagnosing infected pressure ulcers. Pressure ulcer infection delays wound healing and increases risks for sepsis, cellulitis, osteomyelitis, and death.5,6 Clinical evidence of soft tissue involvement, such as erythema, warmth, tenderness, foul odor, or purulent discharge, and systemic inflammatory response (fever, tachycardia, or leukocytosis) are suggestive of a wound infection.3,5 However, these clinical signs may be absent and thus make the distinction between chronic wound and infected pressure ulcer difficult.7 Delayed healing with friable granulation tissue and increased pain in a treated wound may be the only signs of a pressure ulcer infection.3,5,7
Routine laboratory tests (i.e. white blood cell count, C-reactive protein, and erythrocyte sedimentation rate) are neither sensitive nor specific in diagnosing wound infection. Moreover, because pressure ulcers are typically colonized with ≥105 organisms/mL of normal skin flora and bacteria from adjacent gastrointestinal or urogenital environments, swab cultures identify colonizing organisms and are not recommended as a diagnostic test for pressure ulcer microbiologic evaluation.5,6 If microbiological data are needed to guide antibiotic use, cultures of blood, bone, or deep tissue biopsied from a surgically debrided wound should be used.5 Importantly, a higher index of suspicion should be maintained for infection of Stage III or IV pressure ulcers because they are more commonly infected than Stage I or II ulcers.3
Prevention. The prevention of wound progression is essential in treating acute, chronic, or infected pressure ulcers. Although management guidelines are limited by few high-quality, randomized controlled trials, NPUAP recommends a number of prevention strategies targeting risk factors that contribute to pressure ulcer development.2,3,8
For all bed-bound and chair-bound persons with impaired ability to self-reposition, risk assessment for pressure ulcer should be done on admission and repeated every 24 hours. The presence of such risk factors as immobility, shear, friction, moisture, incontinence, and malnutrition should be used to guide preventive treatments. Pressure relief on an ulcer can be achieved by repositioning the immobile patient at one- to two-hour intervals. Pressure-redistributing support surfaces (static, overlays, or dynamic) reduce tissue pressure and decrease overall incidence of pressure ulcers. Due to a lack of relative efficacy data, the selection of a support surface should be determined by the patient’s individual needs in order to reduce pressure and shear.3 For instance, dynamic support is an appropriate surface for an immobile patient with multiple or nonhealing ulcers. Shearing force and friction can be reduced by limiting head-of-bed elevation to <30° and using such transfer aids as bed linens while repositioning patients. The use of pillows, foam wedges, or other devices should be used to eliminate direct contact of bony prominences or reduce pressure on heels.8
Skin care should be optimized to limit excessive dryness or moisture. This includes using moisturizers for dry skin, particularly for the sacrum, and implementing bowel and bladder programs and absorbent underpads in patients with bowel or bladder incontinence.2 Given that patients with pressure ulcers are in a catabolic state, those who are nutritionally compromised may benefit from nutritional supplementation.3 Lastly, appropriate use of local and systemic pain regimen for painful pressure ulcers can improve patient cooperation in repositioning, dressing change, and quality of life.
Debridement. Wound debridement removes necrotic tissue often present in infected or chronic pressure ulcers, reduces risk for further infection, and promotes granulation tissue formation and wound healing. Debridement, however, is not indicated for ulcers of an ischemic limb or dry eschar of the heel, due to propensity for complications.3,4 The five common debridement methods are sharp, mechanical, autolytic, enzymatic, and biosurgical. The debridement method of choice is determined by clinician preference and availability.4
Sharp debridement results in rapid removal of large amounts of nonviable necrotic tissues and eschar using sharp instruments and, therefore, is indicated if wound infection or sepsis is present. Mechanical debridement by wet-to-dry dressing or whirlpool nonselectively removes granulation tissue and, thus, should be used cautiously. Autolytic debridement uses occlusive dressings (i.e. hydrocolloid or hydrogel) to maintain a moist wound environment in order to optimize the body’s inherent ability to selectively self-digest necrotic tissues. Enzymatic debridement with concentrated topical proteolytic enzymes (i.e. collagenase) digests necrotic tissues and achieves faster debridement than autolysis while being less invasive than surgical intervention. Biosurgery utilizes maggots (i.e. Lucilia sericata) that produce enzymes to effectively debride necrotic tissues.
Wound care and dressing. Pressure ulcers should be cleansed with each dressing change using such physiologic solutions as normal saline. Cleansing with antimicrobial solutions for ulcers with large necrotic debris or infection needs to be thoughtfully administered due to the potential impairment on wound healing.4 Wound dressing should maintain a moist wound environment to allow epithelialization and limit excessive exudates in order to prevent maceration. Although there are many categories of moisture retentive dressings, their comparative effectiveness remain unclear.4 Table 2 summarizes characteristics of common wound dressings and their applications.
Antibiotic use. Topical antibiotics are appropriate for Stage III or IV ulcers with signs of local infection, including periwound erythema and friable granulation tissue.4 The Agency for Health Care Policy and Research recommends a two-week trial of a topical antibiotic, such as silver sulfadiazine, for pressure ulcers that fail to heal after two to four weeks of optimal care.6 Systemic antibiotics should be used for patients who demonstrate evidence of systemic infection including sepsis, osteomyelitis, or cellulitis with associated fever and leukocytosis. The choice of systemic antibiotics should be based on cultures from blood, bone, or deep tissue biopsied from a surgically debrided wound.4,6
Back to the Case
The patient was hospitalized for altered mental status. She was at high risk for the progression of her sacral ulcer and the development of new pressure ulcers due to immobility, incontinence, malnutrition, and impaired mental status. The sacral wound was a chronic, Stage III pressure ulcer without evidence of local tissue infection. However, the presence of leukocytosis and fever were suggestive of an underlying infection. Her urine analysis was consistent with a urinary tract infection.
Trimethoprim/sulfamethoxazole was administered with subsequent resolution of leukocytosis, fever, and delirium. The sacral ulcer was cleansed with normal saline and covered with hydrocolloid dressing every 72 hours in order to maintain a moist wound environment and facilitate autolysis. Preventive interventions guided by her risk factors for pressure ulcer were implemented. Interventions included:
- Daily skin and wound assessment;
- Pressure relief with repositioning every two hours;
- Use of a dynamic support surface;
- Head-of-bed elevation of no more than <30° to reduce shear and friction;
- Use of transfer aids;
- Use of devices to eliminate direct contact of bony prominences;
- Optimizing skin care with moisturizers for dry skin and frequent changing of absorbent under pads for incontinence; and
- Consulting nutrition service to optimize nutritional intake.
Bottom Line
Assessments of pressure ulcer stage, wound infection, and risk factors guide targeted therapeutic interventions that facilitate wound healing and prevent new pressure ulcer formation.
Dr. Prager is a fellow in the Brookdale Department of Geriatrics and Palliative Medicine at Mount Sinai School of Medicine in New York City. Dr. Ko is a hospitalist and an assistant professor in the Brookdale Department of Geriatrics and Palliative Medicine at Mount Sinai.
References
- Pressure ulcers in America: prevalence, incidence, and implications for the future. An executive summary of the National Pressure Ulcer Advisory Panel monograph. Adv Skin Wound Care. 2001;14(4):208-215.
- Reddy M, Gill SS, Rochon PA. Preventing pressure ulcers: a systematic review. JAMA. 2006;296(8):974-984.
- European Pressure Ulcer Advisory Panel and National Pressure Ulcer Advisory Panel. Treatment of Pressure Ulcers: Quick Reference Guide. Washington, D.C.: National Pressure Ulcer Advisory Panel; 2009.
- Bates-Jensen BM. Chapter 58. Pressure Ulcers. In: Halter JB, Ouslander JG, Tinetti ME, Studenski S, High KP, Asthana S, eds. Hazzard’s Geriatric Medicine and Gerontology. 6th ed. New York: McGraw-Hill; 2009.
- Livesley NJ, Chow AW. Infected pressure ulcers in elderly individuals. Clin Infect Dis. 2002;35(11):1390-1396.
- Agency for Health Care Policy and Research (AHCPR). Treatment of Pressure Ulcers. Clinical Practice Guideline Number 15. U.S. Department of Health and Human Services. 1994.
- Reddy M, Gill SS, Wu W, Kalkar SR, Rochon PA. Does this patient have an infection of a chronic wound? JAMA. 2012;307(6):605-611.
- National Pressure Ulcer Advisory Panel. Pressure Ulcer Prevention Points. National Pressure Ulcer Advisory Panel website. Available at: http://www.npuap.org/resources/educational-and-clinical-resources/pressure-ulcer-prevention-points/. Accessed Aug. 1, 2012.
- Reuben DB, Herr KA, Pacala JT, et al. Skin Ulcers. In: Geriatrics At Your Fingertips. 12th ed. New York: The American Geriatrics Society; 2010.
The Case
An 85-year-old woman with stroke, functional quadriplegia, and diabetes mellitus presents with altered mental status. She is febrile (38.5°C) with leukocytosis (14,400 cells/mm3) and has a 5 cm x 4 cm x 2 cm Stage III malodorous sacral ulcer without surrounding erythema, tunneling, or pain. The ulcer base is partially covered by green slough. How should this pressure ulcer be evaluated and treated?
Overview
Pressure ulcers in vulnerable populations, such as the elderly and those with limited mobility, are exceedingly common. In the acute-care setting, the incidence of pressure ulcers ranges from 0.4% to 38%, with 2.5 million cases treated annually at an estimated cost of $11 billion per year.1,2 Moreover, as of Oct. 1, 2008, the Centers for Medicare & Medicaid Services (CMS) guideline states that hospitals will no longer receive additional payment when a hospitalized patient develops Stage III or IV pressure ulcers that are not present on admission.
A pressure ulcer is a localized injury to skin and underlying soft tissue over a bony prominence due to sustained external pressure.3 Prolonged pressure on these weight-bearing areas leads to reduced blood flow, ischemia, cell death, and necrosis of local tissues.4 Risk factors for developing pressure ulcers include increased external pressure, shear, friction, moisture, poor perfusion, immobility, incontinence, malnutrition, and impaired mental status.4 Inadequately treated pressure ulcers can lead to pain, tunneling, fistula formation, disfigurement, infection, prolonged hospitalization, lower quality of life, and increased mortality.4
Because of the significant morbidities and high costs associated with the care of pressure ulcers in acute care, hospitalists must be familiar with the assessment and treatment of pressure ulcers in vulnerable patients.
Review of the Data
The management of pressure ulcers in the hospitalized patient starts with a comprehensive assessment of the patient’s medical comorbidities, risk factors, and wound-staging. Considerations must be given to differentiate an infected pressure ulcer from a noninfected ulcer. These evaluations then guide the appropriate treatments of pressure ulcers, including the prevention of progression or formation of new ulcers, debridement, application of wound dressing, and antibiotic use.
Assessing pressure ulcer stage. The National Pressure Ulcer Advisory Panel (NPUAP) Classification System is the most commonly used staging tool. It describes four stages of pressure ulcers (see Table 1).3 A Stage 1 pressure ulcer is characterized by intact skin with nonblanchable erythema and may be discolored, painful, soft, firm, and warmer or cooler compared to adjacent area. A Stage II pressure ulcer presents with partial thickness skin loss with a shallow red-pink wound bed without slough, or as an intact or ruptured serum-filled blister. Stage II pressure ulcers do not include skin tears, tape burns, macerations, or excoriations. A Stage III pressure ulcer has full thickness skin loss with or without visible subcutaneous fat. Bone, tendon, or muscle are not exposed or directly palpable. Slough may be present but it does not obscure the depth of ulcer. Deep ulcers can develop in anatomical regions with high adiposity, such as the pelvic girdle. A Stage IV pressure ulcer has full thickness tissue loss with exposed and palpable bone, tendon, or muscle. Slough, eschar, undermining, and tunneling may be present. The depth of a Stage IV ulcer varies depending on anatomical location and adiposity. Stage IV ulcers also create a nidus for osteomyelitis.
NPUAP describes two additional categories of pressure ulcers: unstageable and deep tissue injury.3 An unstageable ulcer has full thickness skin or tissue loss of unknown depth because the wound base is completely obscured by slough or eschar. The ulcer can only be accurately categorized as Stage III or IV after sufficient slough or eschar is removed to identify wound depth. Lastly, suspected deep tissue injury describes a localized area of discolored intact skin (purple or maroon) or blood-filled blister due to damage of underlying tissue from pressure or shear.
Diagnosing infected pressure ulcers. Pressure ulcer infection delays wound healing and increases risks for sepsis, cellulitis, osteomyelitis, and death.5,6 Clinical evidence of soft tissue involvement, such as erythema, warmth, tenderness, foul odor, or purulent discharge, and systemic inflammatory response (fever, tachycardia, or leukocytosis) are suggestive of a wound infection.3,5 However, these clinical signs may be absent and thus make the distinction between chronic wound and infected pressure ulcer difficult.7 Delayed healing with friable granulation tissue and increased pain in a treated wound may be the only signs of a pressure ulcer infection.3,5,7
Routine laboratory tests (i.e. white blood cell count, C-reactive protein, and erythrocyte sedimentation rate) are neither sensitive nor specific in diagnosing wound infection. Moreover, because pressure ulcers are typically colonized with ≥105 organisms/mL of normal skin flora and bacteria from adjacent gastrointestinal or urogenital environments, swab cultures identify colonizing organisms and are not recommended as a diagnostic test for pressure ulcer microbiologic evaluation.5,6 If microbiological data are needed to guide antibiotic use, cultures of blood, bone, or deep tissue biopsied from a surgically debrided wound should be used.5 Importantly, a higher index of suspicion should be maintained for infection of Stage III or IV pressure ulcers because they are more commonly infected than Stage I or II ulcers.3
Prevention. The prevention of wound progression is essential in treating acute, chronic, or infected pressure ulcers. Although management guidelines are limited by few high-quality, randomized controlled trials, NPUAP recommends a number of prevention strategies targeting risk factors that contribute to pressure ulcer development.2,3,8
For all bed-bound and chair-bound persons with impaired ability to self-reposition, risk assessment for pressure ulcer should be done on admission and repeated every 24 hours. The presence of such risk factors as immobility, shear, friction, moisture, incontinence, and malnutrition should be used to guide preventive treatments. Pressure relief on an ulcer can be achieved by repositioning the immobile patient at one- to two-hour intervals. Pressure-redistributing support surfaces (static, overlays, or dynamic) reduce tissue pressure and decrease overall incidence of pressure ulcers. Due to a lack of relative efficacy data, the selection of a support surface should be determined by the patient’s individual needs in order to reduce pressure and shear.3 For instance, dynamic support is an appropriate surface for an immobile patient with multiple or nonhealing ulcers. Shearing force and friction can be reduced by limiting head-of-bed elevation to <30° and using such transfer aids as bed linens while repositioning patients. The use of pillows, foam wedges, or other devices should be used to eliminate direct contact of bony prominences or reduce pressure on heels.8
Skin care should be optimized to limit excessive dryness or moisture. This includes using moisturizers for dry skin, particularly for the sacrum, and implementing bowel and bladder programs and absorbent underpads in patients with bowel or bladder incontinence.2 Given that patients with pressure ulcers are in a catabolic state, those who are nutritionally compromised may benefit from nutritional supplementation.3 Lastly, appropriate use of local and systemic pain regimen for painful pressure ulcers can improve patient cooperation in repositioning, dressing change, and quality of life.
Debridement. Wound debridement removes necrotic tissue often present in infected or chronic pressure ulcers, reduces risk for further infection, and promotes granulation tissue formation and wound healing. Debridement, however, is not indicated for ulcers of an ischemic limb or dry eschar of the heel, due to propensity for complications.3,4 The five common debridement methods are sharp, mechanical, autolytic, enzymatic, and biosurgical. The debridement method of choice is determined by clinician preference and availability.4
Sharp debridement results in rapid removal of large amounts of nonviable necrotic tissues and eschar using sharp instruments and, therefore, is indicated if wound infection or sepsis is present. Mechanical debridement by wet-to-dry dressing or whirlpool nonselectively removes granulation tissue and, thus, should be used cautiously. Autolytic debridement uses occlusive dressings (i.e. hydrocolloid or hydrogel) to maintain a moist wound environment in order to optimize the body’s inherent ability to selectively self-digest necrotic tissues. Enzymatic debridement with concentrated topical proteolytic enzymes (i.e. collagenase) digests necrotic tissues and achieves faster debridement than autolysis while being less invasive than surgical intervention. Biosurgery utilizes maggots (i.e. Lucilia sericata) that produce enzymes to effectively debride necrotic tissues.
Wound care and dressing. Pressure ulcers should be cleansed with each dressing change using such physiologic solutions as normal saline. Cleansing with antimicrobial solutions for ulcers with large necrotic debris or infection needs to be thoughtfully administered due to the potential impairment on wound healing.4 Wound dressing should maintain a moist wound environment to allow epithelialization and limit excessive exudates in order to prevent maceration. Although there are many categories of moisture retentive dressings, their comparative effectiveness remain unclear.4 Table 2 summarizes characteristics of common wound dressings and their applications.
Antibiotic use. Topical antibiotics are appropriate for Stage III or IV ulcers with signs of local infection, including periwound erythema and friable granulation tissue.4 The Agency for Health Care Policy and Research recommends a two-week trial of a topical antibiotic, such as silver sulfadiazine, for pressure ulcers that fail to heal after two to four weeks of optimal care.6 Systemic antibiotics should be used for patients who demonstrate evidence of systemic infection including sepsis, osteomyelitis, or cellulitis with associated fever and leukocytosis. The choice of systemic antibiotics should be based on cultures from blood, bone, or deep tissue biopsied from a surgically debrided wound.4,6
Back to the Case
The patient was hospitalized for altered mental status. She was at high risk for the progression of her sacral ulcer and the development of new pressure ulcers due to immobility, incontinence, malnutrition, and impaired mental status. The sacral wound was a chronic, Stage III pressure ulcer without evidence of local tissue infection. However, the presence of leukocytosis and fever were suggestive of an underlying infection. Her urine analysis was consistent with a urinary tract infection.
Trimethoprim/sulfamethoxazole was administered with subsequent resolution of leukocytosis, fever, and delirium. The sacral ulcer was cleansed with normal saline and covered with hydrocolloid dressing every 72 hours in order to maintain a moist wound environment and facilitate autolysis. Preventive interventions guided by her risk factors for pressure ulcer were implemented. Interventions included:
- Daily skin and wound assessment;
- Pressure relief with repositioning every two hours;
- Use of a dynamic support surface;
- Head-of-bed elevation of no more than <30° to reduce shear and friction;
- Use of transfer aids;
- Use of devices to eliminate direct contact of bony prominences;
- Optimizing skin care with moisturizers for dry skin and frequent changing of absorbent under pads for incontinence; and
- Consulting nutrition service to optimize nutritional intake.
Bottom Line
Assessments of pressure ulcer stage, wound infection, and risk factors guide targeted therapeutic interventions that facilitate wound healing and prevent new pressure ulcer formation.
Dr. Prager is a fellow in the Brookdale Department of Geriatrics and Palliative Medicine at Mount Sinai School of Medicine in New York City. Dr. Ko is a hospitalist and an assistant professor in the Brookdale Department of Geriatrics and Palliative Medicine at Mount Sinai.
References
- Pressure ulcers in America: prevalence, incidence, and implications for the future. An executive summary of the National Pressure Ulcer Advisory Panel monograph. Adv Skin Wound Care. 2001;14(4):208-215.
- Reddy M, Gill SS, Rochon PA. Preventing pressure ulcers: a systematic review. JAMA. 2006;296(8):974-984.
- European Pressure Ulcer Advisory Panel and National Pressure Ulcer Advisory Panel. Treatment of Pressure Ulcers: Quick Reference Guide. Washington, D.C.: National Pressure Ulcer Advisory Panel; 2009.
- Bates-Jensen BM. Chapter 58. Pressure Ulcers. In: Halter JB, Ouslander JG, Tinetti ME, Studenski S, High KP, Asthana S, eds. Hazzard’s Geriatric Medicine and Gerontology. 6th ed. New York: McGraw-Hill; 2009.
- Livesley NJ, Chow AW. Infected pressure ulcers in elderly individuals. Clin Infect Dis. 2002;35(11):1390-1396.
- Agency for Health Care Policy and Research (AHCPR). Treatment of Pressure Ulcers. Clinical Practice Guideline Number 15. U.S. Department of Health and Human Services. 1994.
- Reddy M, Gill SS, Wu W, Kalkar SR, Rochon PA. Does this patient have an infection of a chronic wound? JAMA. 2012;307(6):605-611.
- National Pressure Ulcer Advisory Panel. Pressure Ulcer Prevention Points. National Pressure Ulcer Advisory Panel website. Available at: http://www.npuap.org/resources/educational-and-clinical-resources/pressure-ulcer-prevention-points/. Accessed Aug. 1, 2012.
- Reuben DB, Herr KA, Pacala JT, et al. Skin Ulcers. In: Geriatrics At Your Fingertips. 12th ed. New York: The American Geriatrics Society; 2010.
In the Literature
Literature at a Glance
- Drug-eluting stents decrease the need for revascularization.
- Case volume is related to hospital performance assessment.
- Prolonged QRS duration in patients with CHF is associated with increased morbidity and mortality.
- For out-of-hospital ACLS, vasopressin plus epinephrine is not better than vasopressin alone.
- Oral rivaroxaban is more efficacious than enoxaparin for VTE prophylaxis after total hip replacement.
- LMWH and UFH offer similar perioperative VTE prophylaxis benefit in patients with cancer.
- Salmeterol added to inhaled corticosteroids decreases severe asthma exacerbations.
- Early invasive strategy has unclear benefit in low-risk women with unstable angina or NSTEMI.
- Strategies to prevent contrast-induced acute kidney injury are not uniform.
- Hyperglycemia in hospitalized children is common and associated with ICU admission.
Do drug-eluting stents improve outcomes after ST-elevation myocardial infarction (STEMI)?
Background: Drug-eluting stents reduce restenosis rates compared to bare-metal stents. However, there is concern drug-eluting stents increase the risk of stent thrombosis leading to MI and death. Prior studies compared patients who received bare-metal versus those who received drug-eluting stents. Outcomes on a population level might provide new insight.
Study design: Observational study.
Setting: 100% national sample of patients 65 and older who received a coronary stent from 2002-05 enrolled in the traditional fee-for-service Medicare program.
Synopsis: 38,917 patients in the pre-drug-eluting-stent era from October 2002 to March 2003 received bare-metal stents. Nearly 62% of 28,086 patients studied from September to December 2003 received drug-eluting stents. The remaining 38.5% received bare-metal stents. Outcomes of percutaneous coronary intervention (PCI), coronary artery bypass grafting (CABG), STEMI, and death were observed through December 31, 2005.
Patients in the drug-eluting-stent era had a lower two-year risk for repeat revascularization compared to patients in the bare-metal-stent era. In the drug-eluting versus bare-metal eras, repeat PCI was 17.1% versus 20.0% (p<0.001) and need for CABG was 2.7% versus 4.2% (p<0.01). Comparing adjusted outcomes for death, or STEMI, at two years, the two groups appeared similar.
The study did have limitations: the data only reflect sirolimus stents, the authors could not assess dual-antiplatelet therapy or obtain information on coronary anatomy or procedure details to account for selection bias in stent utilization, and the patients were all Medicare beneficiaries.
Bottom line: Drug-eluting stents are associated with fewer repeat revascularization procedures than bare-metal stents, but have not shown a significant improvement in the subsequent risk of STEMI or death.
Citation: Malenka DJ, Kaplan AV, Lucas FL, Sharp SM, Skinner JA. Outcomes following coronary stenting in the era of bare-metal vs. the era of drug-eluting stents. JAMA 2008;299(24):2868-2877.
Does case volume affect hospital performance for publicly reported process measures?
Background: Hospitals are increasingly graded and compared to one another. “Top medical centers” are defined as those within the top 10% of hospitals in specified performance measures. Hospitals with large and small case volumes might not be compared evenly and fairly.
Study design: Eight publicly reported process measures for acute myocardial infarction (AMI) were compared to hospital case volume, process performance, and label as “top hospital.”
Setting: Data were analyzed from the Hospital Quality Alliance for 3,761 hospitals from January to December 2005.
Synopsis: Hospitals with large case volume overall had better process performance. For example, looking at use of beta-blockers in patients with AMI on arrival to a hospital, small-volume hospitals (<10 AMI cases) averaged 72% while large volume (>100 AMI cases) averaged 80% (p<0.001). However, hospitals with small case volumes were more likely to receive “top hospital” rating even when hospitals with very low case volumes were excluded.
Hospital quality reporting that does not account for case volume is misleading to hospitals and consumers. In this study, larger-volume hospitals appeared to perform better in process measures, but were less likely to receive “top hospital” rating.
Bottom line: Hospitals with large and small case volumes can easily be compared to one another for process measures in AMI.
Citation: O’Brien SM, DeLong ER, Peterson ED. Impact of case volume on hospital performance assessment. Arch Intern Med. 2008;168(12):1277-1284.
What is the predictive value of QRS duration in patients hospitalized with worsening CHF?
Background: In outpatients, a prolonged QRS duration (greater than 120 ms) is associated with increased mortality. Its value in the inpatient setting is unclear. For patients hospitalized with CHF exacerbations, establishing the value of QRS duration may allow for tailored management.
Study design: Retrospective post hoc analysis from the Efficacy of Vasopressin Antagonism in Heart Failure Outcome Study with Tolvaptan (EVEREST).
Setting: 4,133 patients were enrolled from North American, South American, and European sites.
Synopsis: Of 2,962 patients included in the final post hoc analysis, 1,321 (44.6%) had a prolonged QRS duration. During a median follow up of 9.9 months, the all-cause mortality rate was 18.7% for patients with a normal baseline QRS duration and 28.1% for patients with a prolonged baseline QRS.
After adjusting for confounding variables, patients with a prolonged baseline QRS had a 24% increased risk of all-cause mortality and a 28% increased risk for a composite endpoint of cardiac mortality or hospitalization for heart failure exacerbation.
The retrospective nature of the analysis represents the major limitation of this study. In addition, most of the enrolled patients were white, which limits the studies generalizability to other ethnic groups.
Bottom Line: A prolonged QRS duration for patients admitted with decompensated left ventricular heart failure is common and may be associated with increased morbidity and mortality.
Citation: Wang NC, Maggioni AP, Konstam MA, Zannad F, Drasa HB, Burnett JC, et al. Clinical implications of QRS duration in patients hospitalized with worsening heart failure and reduced left ventricular ejection fraction. JAMA. 2008;299(22):2656-2666.
For patients with out-of-hospital cardiac arrest, does the addition of vasopressin to epinephrine in a protocol for ACLS improve outcomes?
Background: The outcome for patients experiencing cardiac arrest who require vasopressors remains extremely poor. Despite disappointing data on vasopressin as an alternative treatment during cardiac arrest, a recent subgroup analysis suggested patients who received epinephrine and vasopressin together had superior clinical outcomes.
Study Design: Prospective multicenter randomized double-blind controlled trial.
Setting: 31 emergency medical service organizations in France.
Synopsis: Of the 2,894 patients, 20.7% of those receiving combination treatment (vasopressin plus epinephrine) survived to hospital admission versus 21.3% of those in the epinephrine-only group. For those same groups, 1.7% of combination and 2.3% of epinephrine-only patients survived to hospital discharge. No significant outcome differences were found in any group or subgroup analysis.
The study had lower-than-expected overall survival to hospital discharge, which may have handicapped its effort to find a true difference in treatment arms.
Bottom line: The addition of vasopressin to epinephrine in the treatment of out-of-hospital cardiac arrest does not improve outcomes.
Citation: Gueugniaud PY, David JS, Chanzy E, Hubert H, Dubien P, Mauriaucourt P, et al. Vasopressin and epinephrine vs. epinephrine alone in cardiopulmonary resuscitation. N Engl J Med. 2008;359(1):21-30.
Is oral rivaroxaban more efficacious than subcutaneous enoxaparin in preventing VTE after hip-replacement surgery?
Background: Venous thromboembolism (VTE) prophylaxis after total hip replacement (THR) is important but can be cumbersome because the most commonly used anticoagulants are either subcutaneous or require frequent monitoring. Rivaroxaban, an oral direct inhibitor of factor Xa may provide more convenient anticoagulation postoperatively. However, its efficacy and safety are unknown.
Study design: Randomized double-blind trial.
Setting: Multicenter study performed in 27 countries.
Synopsis: Patients undergoing THR surgery were randomized to oral rivaroxaban (10mg once daily without monitoring, started six to eight hours after surgery) or subcutaneous enoxaparin (40mg once daily, started 12 hours prior to surgery). After surgery, prophylaxis was administered for 35 days. The primary outcome was a composite of asymptomatic deep venous thrombosis (DVT), symptomatic DVT or pulmonary embolism (PE), or death from any cause at 36 days after surgery.
In the enoxaparin group, 3.7% of patients experienced the primary outcome. This decreased to 1.1% in the rivaroxaban group. Approximately one-third of events were symptomatic. Major bleeding occurred in 0.1% and 0.3% (p=NS) of patients in the enoxaparin and rivaroxaban groups, respectively.
The study is limited by the exclusion of 1,388 of the 4,541 patients (30.6%) randomized, primarily due to having inadequate venography. Also, because the majority of thromboembolic events were asymptomatic, the primary outcome overemphasizes the clinical difference.
Bottom line: Oral rivaroxaban without monitoring is more efficacious than, and as safe as, subcutaneous enoxaparin when used for VTE prophylaxis for THR.
Citation: Eriksson B, Borris LC, Friedman RJ, Hass S, Huisman MV, Kakkar AK, et al. Rivaroxaban versus enoxaparin for thromboprophylaxis after hip arthroplasty. N Engl J Med. 2008;358:2765-75.
Is LMWH more efficacious than UFH in preventing postoperative VTE in cancer patients?
Background: Patients with cancer are at increased risk for VTE and require prophylaxis to prevent this complication postoperatively. Low molecular weight heparin (LMWH) has proven more efficacious than subcutaneous unfractionated heparin (UFH) in other settings (e.g., DVT treatment). However, it is still unknown whether LMWH offers better prophylaxis compared to UFH for cancer patients undergoing surgery.
Study design: Systematic review and meta-analysis.
Setting: 14 randomized controlled trials.
Synopsis: Eleven trials exclusively examined patients with cancer (n=4006) and three trials reported data for cancer patients as subgroups (n=1816). There were in differences in mortality, pulmonary embolism, and symptomatic DVT rates between the two groups.
LMWH was associated with a decrease in total (asymptomatic or symptomatic) DVT (RR, 0.72; 95% CI, 0.55-0.94). Rates of major bleeding, minor bleeding, and intraoperative blood loss were similar between the two treatments.
This meta-analysis is limited because 12 remaining trials (n=3185) also enrolled cancer patients but did not provide specific data for the cancer patient subgroup. The study also is limited by the heterogeneity of the original trials, including utilizing varying LMWHs and dosing regimens, numerous types of surgeries, and a wide range of neoplasms.
Bottom line: LMWH does not decrease mortality, pulmonary embolism, or symptomatic DVT compared to UFH in cancer patients undergoing surgery.
Citation: Akl EA, Terrenato I, Barba M, Sperati F, Sempos EV, Muti P, et al. Low-molecular-weight heparin vs. unfractionated heparin for perioperative thromboprophylaxis in patients with cancer. Arch Intern Med. 2008;168:1261-9.
Does salmeterol added to inhaled corticosteroids improve severe asthma-related events?
Background: Asthma is a chronic disease causing major morbidity and mortality worldwide. Disease guidelines recommend all patients with persistent asthma be treated with inhaled corticosteroids. These same guidelines recommend adding a long-acting beta-agonist for patients whose symptoms persist. However, the safety of this practice has come under scrutiny.
Study design: Meta-analysis.
Setting: Sixty-six randomized, controlled trials conducted worldwide.
Synopsis: Analysis included 66 GlaxoSmithKline trials with a total of 20,966 patients with persistent asthma. Patients used either salmeterol (50mcg twice daily) plus inhaled corticosteroid (10,400 patients) or inhaled corticosteroid alone (10,566 patients).
Results showed no differences in asthma-related hospitalizations, asthma-related intubations, or deaths between the two groups. However, due to the low number of events, definitive conclusions are difficult to make. Severe asthma exacerbations requiring systemic corticosteroids significantly decreased in the inhaled corticosteroid plus salmeterol group.
The study is limited by it inclusion of only those trials sponsored by GlaxoSmithKline and by the short duration of most of the studies. Additionally, the studies included in the analysis used clinical outcomes as secondary endpoints.
Bottom line: Adding salmeterol to inhaled corticosteroid decreases severe asthma exacerbations and is likely safe, but does not have an effect on asthma-related hospitalization or death.
Citation: Bateman E, Nelson H, Bousquet J, Kral K, Sutton L, Ortega H, et.al. Meta-analysis: Effects of adding salmeterol to inhaled corticosteroids on serious asthma-related events. Annals Intern Med. 2008;149:33-42.
Is an early invasive strategy effective in women with unstable angina or NSTEMI?
Background: Despite many trials showing the value of an early invasive strategy for patients with non-ST-segment elevation acute coronary syndrome (NSTE ACS), data from several trials question this benefit in women. Some trials show higher risk of death and myocardial infarction (MI) in subgroup analysis of women.
Study Design: Meta-analysis.
Setting: Eight randomized, controlled trials conducted worldwide.
Synopsis: Analysis included eight trials with 10,412 patients (3,075 women) with NSTE ACS. The invasive group (5,083 patients) was defined as those referred for coronary angiography with subsequent intervention as needed. The composite endpoint of death, MI, or rehospitalization within 12 months with ACS occurred in 21.1% of the invasive group and 25.9% of the medically managed group (OR, 0.78; CI, 0.61-0.98).
The subgroup, including only women, had a non-statistically significant OR of 0.81 (CI, 0.65-1.01), including no effect on all-cause mortality, nonfatal MI, or the composite of death and MI. However, women with high-risk features (elevated biomarkers) undergoing the invasive strategy had a significant reduction in the composite endpoint (OR, 0.67; CI, 0.50-0.88).
The study is limited by the use of subgroup analysis, secondary endpoints, heterogeneity between trials, and possible publication bias.
Bottom line: Early invasive strategy is effective in men and high-risk women with NSTE ACS, but not in low-risk women.
Citation: O’Donoghue M, Boden W, Braunwald E, Cannon CP, Clayton TC, Winter RJ, et.al. Early invasive vs. conservative treatment strategies in women and men with unstable angina and non-ST-segment elevation myocardial infarction. JAMA. 2008;300:71-80.
What strategies are used to prevent contrast-induced acute kidney injury?
Background: Contrast-induced acute kidney injury (CIAKI) is a condition potentially amenable to preventive care. Several trials have identified intravenous hydration, N-acetylcysteine, and withdrawal of NSAIDS as interventions that reduce the possibility of CIAKI in high-risk patients. Little is known about whether healthcare providers routinely use these strategies.
Study design: Prospective observational cohort study.
Setting: Veterans Affairs (VA) Pittsburgh Healthcare System.
Synopsis: 11,410 patients scheduled for radiographic procedures were screened. After exclusion criteria and eligibility, 660 patients with an estimated glomerular filtration rate less than 60ml/min/1.73m2 were identified. Usage of intravenous fluids, N-acetylcysteine, and discontinuation of NSAIDS were recorded. Serum creatinine (SCr) was measured 48 to 96 hours post-procedure. CIAKI was defined as relative increase in SCr from baseline (≥25%, ≥50% and ≥100%) and absolute increase in SCr levels from baseline (≥0.25, ≥0.5, and ≥1.0). CIAKI association with adverse outcomes was evaluated by tracking 30-day mortality, need for dialysis, and hospitalization.
The incidence of CIAKI was less common in patients undergoing CT scans versus those having angiograms. Adverse 30-day outcomes were uncommon. Pre- and post-procedure intravenous hydration was administered to 40% of study patients, more commonly with coronary angiogram than with computed tomography (91.2% vs. 16%, p<0.0001). N-acetylcysteine was administered to 39.2%. Only 6.8% of those taking NSAIDS reported being told to discontinue the medication.
Study limitations include the small sample size and the single site location, both limiting generalizability.
Bottom line: Clinically significant CIAKI is uncommon, and preventive care is not uniformly implemented in patients undergoing contrast-enhanced radiographic procedures.
Citation: Weisbord SD, Mor MK, Resnick AL, Hartwig KC, Sonel AF, Fine MJ, et al. Prevention, incidence, and outcomes of contrast-induced acute kidney injury. Arch Intern Med. 2008;168(12):1325-1332.
How does hyperglycemia affect morbidity and mortality in children admitted to a community pediatric hospital?
Background: Inpatient hyperglycemia in adult patients is a predictor of poor clinical outcomes. The association of hyperglycemia and clinical outcomes in children admitted to a general community hospital has not been studied.
Study design: Retrospective observational cohort study.
Setting: A community pediatric hospital in Atlanta, Ga.
Synopsis: Review of medical records of 903 consecutive pediatric patients admitted to critical and non-critical areas took place. Of these, 542 patients constituted the study population. The study excluded 342 patients who didn’t have a blood glucose measurement. Hyperglycemia was defined as an admission or in-hospital blood glucose greater than 120mg/dl.
One-fourth of the children admitted to the hospital had hyperglycemia, most without a prior history of diabetes. The presence of hyperglycemia on admission was not associated with increased length of stay (LOS) or increased mortality. Children with hyperglycemia were more likely to be admitted to the ICU and had longer ICU LOS.
This was a retrospective study conducted at a single site whose demographics and disease spectrum may differ from those of other institutions. There were an insufficient number of deaths to make any conclusions regarding the impact of hyperglycemia on mortality. Prospective, randomized, multicenter trials are needed to better elucidate the effects of in-patient hyperglycemia.
Bottom line: Hyperglycemia is common in children with or without diabetes admitted to the hospital, and is associated with increased ICU admissions and ICU length of stay. Its connection to mortality is inconclusive.
Citation: Palaio A, Smiley D, Ceron M, Klein R, Cho IS, Mejia R, et al. Prevalence and clinical outcome of inpatient hyperglycemia in a community pediatric hospital. J Hosp Med.2008;3(3):212-217.
Literature at a Glance
- Drug-eluting stents decrease the need for revascularization.
- Case volume is related to hospital performance assessment.
- Prolonged QRS duration in patients with CHF is associated with increased morbidity and mortality.
- For out-of-hospital ACLS, vasopressin plus epinephrine is not better than vasopressin alone.
- Oral rivaroxaban is more efficacious than enoxaparin for VTE prophylaxis after total hip replacement.
- LMWH and UFH offer similar perioperative VTE prophylaxis benefit in patients with cancer.
- Salmeterol added to inhaled corticosteroids decreases severe asthma exacerbations.
- Early invasive strategy has unclear benefit in low-risk women with unstable angina or NSTEMI.
- Strategies to prevent contrast-induced acute kidney injury are not uniform.
- Hyperglycemia in hospitalized children is common and associated with ICU admission.
Do drug-eluting stents improve outcomes after ST-elevation myocardial infarction (STEMI)?
Background: Drug-eluting stents reduce restenosis rates compared to bare-metal stents. However, there is concern drug-eluting stents increase the risk of stent thrombosis leading to MI and death. Prior studies compared patients who received bare-metal versus those who received drug-eluting stents. Outcomes on a population level might provide new insight.
Study design: Observational study.
Setting: 100% national sample of patients 65 and older who received a coronary stent from 2002-05 enrolled in the traditional fee-for-service Medicare program.
Synopsis: 38,917 patients in the pre-drug-eluting-stent era from October 2002 to March 2003 received bare-metal stents. Nearly 62% of 28,086 patients studied from September to December 2003 received drug-eluting stents. The remaining 38.5% received bare-metal stents. Outcomes of percutaneous coronary intervention (PCI), coronary artery bypass grafting (CABG), STEMI, and death were observed through December 31, 2005.
Patients in the drug-eluting-stent era had a lower two-year risk for repeat revascularization compared to patients in the bare-metal-stent era. In the drug-eluting versus bare-metal eras, repeat PCI was 17.1% versus 20.0% (p<0.001) and need for CABG was 2.7% versus 4.2% (p<0.01). Comparing adjusted outcomes for death, or STEMI, at two years, the two groups appeared similar.
The study did have limitations: the data only reflect sirolimus stents, the authors could not assess dual-antiplatelet therapy or obtain information on coronary anatomy or procedure details to account for selection bias in stent utilization, and the patients were all Medicare beneficiaries.
Bottom line: Drug-eluting stents are associated with fewer repeat revascularization procedures than bare-metal stents, but have not shown a significant improvement in the subsequent risk of STEMI or death.
Citation: Malenka DJ, Kaplan AV, Lucas FL, Sharp SM, Skinner JA. Outcomes following coronary stenting in the era of bare-metal vs. the era of drug-eluting stents. JAMA 2008;299(24):2868-2877.
Does case volume affect hospital performance for publicly reported process measures?
Background: Hospitals are increasingly graded and compared to one another. “Top medical centers” are defined as those within the top 10% of hospitals in specified performance measures. Hospitals with large and small case volumes might not be compared evenly and fairly.
Study design: Eight publicly reported process measures for acute myocardial infarction (AMI) were compared to hospital case volume, process performance, and label as “top hospital.”
Setting: Data were analyzed from the Hospital Quality Alliance for 3,761 hospitals from January to December 2005.
Synopsis: Hospitals with large case volume overall had better process performance. For example, looking at use of beta-blockers in patients with AMI on arrival to a hospital, small-volume hospitals (<10 AMI cases) averaged 72% while large volume (>100 AMI cases) averaged 80% (p<0.001). However, hospitals with small case volumes were more likely to receive “top hospital” rating even when hospitals with very low case volumes were excluded.
Hospital quality reporting that does not account for case volume is misleading to hospitals and consumers. In this study, larger-volume hospitals appeared to perform better in process measures, but were less likely to receive “top hospital” rating.
Bottom line: Hospitals with large and small case volumes can easily be compared to one another for process measures in AMI.
Citation: O’Brien SM, DeLong ER, Peterson ED. Impact of case volume on hospital performance assessment. Arch Intern Med. 2008;168(12):1277-1284.
What is the predictive value of QRS duration in patients hospitalized with worsening CHF?
Background: In outpatients, a prolonged QRS duration (greater than 120 ms) is associated with increased mortality. Its value in the inpatient setting is unclear. For patients hospitalized with CHF exacerbations, establishing the value of QRS duration may allow for tailored management.
Study design: Retrospective post hoc analysis from the Efficacy of Vasopressin Antagonism in Heart Failure Outcome Study with Tolvaptan (EVEREST).
Setting: 4,133 patients were enrolled from North American, South American, and European sites.
Synopsis: Of 2,962 patients included in the final post hoc analysis, 1,321 (44.6%) had a prolonged QRS duration. During a median follow up of 9.9 months, the all-cause mortality rate was 18.7% for patients with a normal baseline QRS duration and 28.1% for patients with a prolonged baseline QRS.
After adjusting for confounding variables, patients with a prolonged baseline QRS had a 24% increased risk of all-cause mortality and a 28% increased risk for a composite endpoint of cardiac mortality or hospitalization for heart failure exacerbation.
The retrospective nature of the analysis represents the major limitation of this study. In addition, most of the enrolled patients were white, which limits the studies generalizability to other ethnic groups.
Bottom Line: A prolonged QRS duration for patients admitted with decompensated left ventricular heart failure is common and may be associated with increased morbidity and mortality.
Citation: Wang NC, Maggioni AP, Konstam MA, Zannad F, Drasa HB, Burnett JC, et al. Clinical implications of QRS duration in patients hospitalized with worsening heart failure and reduced left ventricular ejection fraction. JAMA. 2008;299(22):2656-2666.
For patients with out-of-hospital cardiac arrest, does the addition of vasopressin to epinephrine in a protocol for ACLS improve outcomes?
Background: The outcome for patients experiencing cardiac arrest who require vasopressors remains extremely poor. Despite disappointing data on vasopressin as an alternative treatment during cardiac arrest, a recent subgroup analysis suggested patients who received epinephrine and vasopressin together had superior clinical outcomes.
Study Design: Prospective multicenter randomized double-blind controlled trial.
Setting: 31 emergency medical service organizations in France.
Synopsis: Of the 2,894 patients, 20.7% of those receiving combination treatment (vasopressin plus epinephrine) survived to hospital admission versus 21.3% of those in the epinephrine-only group. For those same groups, 1.7% of combination and 2.3% of epinephrine-only patients survived to hospital discharge. No significant outcome differences were found in any group or subgroup analysis.
The study had lower-than-expected overall survival to hospital discharge, which may have handicapped its effort to find a true difference in treatment arms.
Bottom line: The addition of vasopressin to epinephrine in the treatment of out-of-hospital cardiac arrest does not improve outcomes.
Citation: Gueugniaud PY, David JS, Chanzy E, Hubert H, Dubien P, Mauriaucourt P, et al. Vasopressin and epinephrine vs. epinephrine alone in cardiopulmonary resuscitation. N Engl J Med. 2008;359(1):21-30.
Is oral rivaroxaban more efficacious than subcutaneous enoxaparin in preventing VTE after hip-replacement surgery?
Background: Venous thromboembolism (VTE) prophylaxis after total hip replacement (THR) is important but can be cumbersome because the most commonly used anticoagulants are either subcutaneous or require frequent monitoring. Rivaroxaban, an oral direct inhibitor of factor Xa may provide more convenient anticoagulation postoperatively. However, its efficacy and safety are unknown.
Study design: Randomized double-blind trial.
Setting: Multicenter study performed in 27 countries.
Synopsis: Patients undergoing THR surgery were randomized to oral rivaroxaban (10mg once daily without monitoring, started six to eight hours after surgery) or subcutaneous enoxaparin (40mg once daily, started 12 hours prior to surgery). After surgery, prophylaxis was administered for 35 days. The primary outcome was a composite of asymptomatic deep venous thrombosis (DVT), symptomatic DVT or pulmonary embolism (PE), or death from any cause at 36 days after surgery.
In the enoxaparin group, 3.7% of patients experienced the primary outcome. This decreased to 1.1% in the rivaroxaban group. Approximately one-third of events were symptomatic. Major bleeding occurred in 0.1% and 0.3% (p=NS) of patients in the enoxaparin and rivaroxaban groups, respectively.
The study is limited by the exclusion of 1,388 of the 4,541 patients (30.6%) randomized, primarily due to having inadequate venography. Also, because the majority of thromboembolic events were asymptomatic, the primary outcome overemphasizes the clinical difference.
Bottom line: Oral rivaroxaban without monitoring is more efficacious than, and as safe as, subcutaneous enoxaparin when used for VTE prophylaxis for THR.
Citation: Eriksson B, Borris LC, Friedman RJ, Hass S, Huisman MV, Kakkar AK, et al. Rivaroxaban versus enoxaparin for thromboprophylaxis after hip arthroplasty. N Engl J Med. 2008;358:2765-75.
Is LMWH more efficacious than UFH in preventing postoperative VTE in cancer patients?
Background: Patients with cancer are at increased risk for VTE and require prophylaxis to prevent this complication postoperatively. Low molecular weight heparin (LMWH) has proven more efficacious than subcutaneous unfractionated heparin (UFH) in other settings (e.g., DVT treatment). However, it is still unknown whether LMWH offers better prophylaxis compared to UFH for cancer patients undergoing surgery.
Study design: Systematic review and meta-analysis.
Setting: 14 randomized controlled trials.
Synopsis: Eleven trials exclusively examined patients with cancer (n=4006) and three trials reported data for cancer patients as subgroups (n=1816). There were in differences in mortality, pulmonary embolism, and symptomatic DVT rates between the two groups.
LMWH was associated with a decrease in total (asymptomatic or symptomatic) DVT (RR, 0.72; 95% CI, 0.55-0.94). Rates of major bleeding, minor bleeding, and intraoperative blood loss were similar between the two treatments.
This meta-analysis is limited because 12 remaining trials (n=3185) also enrolled cancer patients but did not provide specific data for the cancer patient subgroup. The study also is limited by the heterogeneity of the original trials, including utilizing varying LMWHs and dosing regimens, numerous types of surgeries, and a wide range of neoplasms.
Bottom line: LMWH does not decrease mortality, pulmonary embolism, or symptomatic DVT compared to UFH in cancer patients undergoing surgery.
Citation: Akl EA, Terrenato I, Barba M, Sperati F, Sempos EV, Muti P, et al. Low-molecular-weight heparin vs. unfractionated heparin for perioperative thromboprophylaxis in patients with cancer. Arch Intern Med. 2008;168:1261-9.
Does salmeterol added to inhaled corticosteroids improve severe asthma-related events?
Background: Asthma is a chronic disease causing major morbidity and mortality worldwide. Disease guidelines recommend all patients with persistent asthma be treated with inhaled corticosteroids. These same guidelines recommend adding a long-acting beta-agonist for patients whose symptoms persist. However, the safety of this practice has come under scrutiny.
Study design: Meta-analysis.
Setting: Sixty-six randomized, controlled trials conducted worldwide.
Synopsis: Analysis included 66 GlaxoSmithKline trials with a total of 20,966 patients with persistent asthma. Patients used either salmeterol (50mcg twice daily) plus inhaled corticosteroid (10,400 patients) or inhaled corticosteroid alone (10,566 patients).
Results showed no differences in asthma-related hospitalizations, asthma-related intubations, or deaths between the two groups. However, due to the low number of events, definitive conclusions are difficult to make. Severe asthma exacerbations requiring systemic corticosteroids significantly decreased in the inhaled corticosteroid plus salmeterol group.
The study is limited by it inclusion of only those trials sponsored by GlaxoSmithKline and by the short duration of most of the studies. Additionally, the studies included in the analysis used clinical outcomes as secondary endpoints.
Bottom line: Adding salmeterol to inhaled corticosteroid decreases severe asthma exacerbations and is likely safe, but does not have an effect on asthma-related hospitalization or death.
Citation: Bateman E, Nelson H, Bousquet J, Kral K, Sutton L, Ortega H, et.al. Meta-analysis: Effects of adding salmeterol to inhaled corticosteroids on serious asthma-related events. Annals Intern Med. 2008;149:33-42.
Is an early invasive strategy effective in women with unstable angina or NSTEMI?
Background: Despite many trials showing the value of an early invasive strategy for patients with non-ST-segment elevation acute coronary syndrome (NSTE ACS), data from several trials question this benefit in women. Some trials show higher risk of death and myocardial infarction (MI) in subgroup analysis of women.
Study Design: Meta-analysis.
Setting: Eight randomized, controlled trials conducted worldwide.
Synopsis: Analysis included eight trials with 10,412 patients (3,075 women) with NSTE ACS. The invasive group (5,083 patients) was defined as those referred for coronary angiography with subsequent intervention as needed. The composite endpoint of death, MI, or rehospitalization within 12 months with ACS occurred in 21.1% of the invasive group and 25.9% of the medically managed group (OR, 0.78; CI, 0.61-0.98).
The subgroup, including only women, had a non-statistically significant OR of 0.81 (CI, 0.65-1.01), including no effect on all-cause mortality, nonfatal MI, or the composite of death and MI. However, women with high-risk features (elevated biomarkers) undergoing the invasive strategy had a significant reduction in the composite endpoint (OR, 0.67; CI, 0.50-0.88).
The study is limited by the use of subgroup analysis, secondary endpoints, heterogeneity between trials, and possible publication bias.
Bottom line: Early invasive strategy is effective in men and high-risk women with NSTE ACS, but not in low-risk women.
Citation: O’Donoghue M, Boden W, Braunwald E, Cannon CP, Clayton TC, Winter RJ, et.al. Early invasive vs. conservative treatment strategies in women and men with unstable angina and non-ST-segment elevation myocardial infarction. JAMA. 2008;300:71-80.
What strategies are used to prevent contrast-induced acute kidney injury?
Background: Contrast-induced acute kidney injury (CIAKI) is a condition potentially amenable to preventive care. Several trials have identified intravenous hydration, N-acetylcysteine, and withdrawal of NSAIDS as interventions that reduce the possibility of CIAKI in high-risk patients. Little is known about whether healthcare providers routinely use these strategies.
Study design: Prospective observational cohort study.
Setting: Veterans Affairs (VA) Pittsburgh Healthcare System.
Synopsis: 11,410 patients scheduled for radiographic procedures were screened. After exclusion criteria and eligibility, 660 patients with an estimated glomerular filtration rate less than 60ml/min/1.73m2 were identified. Usage of intravenous fluids, N-acetylcysteine, and discontinuation of NSAIDS were recorded. Serum creatinine (SCr) was measured 48 to 96 hours post-procedure. CIAKI was defined as relative increase in SCr from baseline (≥25%, ≥50% and ≥100%) and absolute increase in SCr levels from baseline (≥0.25, ≥0.5, and ≥1.0). CIAKI association with adverse outcomes was evaluated by tracking 30-day mortality, need for dialysis, and hospitalization.
The incidence of CIAKI was less common in patients undergoing CT scans versus those having angiograms. Adverse 30-day outcomes were uncommon. Pre- and post-procedure intravenous hydration was administered to 40% of study patients, more commonly with coronary angiogram than with computed tomography (91.2% vs. 16%, p<0.0001). N-acetylcysteine was administered to 39.2%. Only 6.8% of those taking NSAIDS reported being told to discontinue the medication.
Study limitations include the small sample size and the single site location, both limiting generalizability.
Bottom line: Clinically significant CIAKI is uncommon, and preventive care is not uniformly implemented in patients undergoing contrast-enhanced radiographic procedures.
Citation: Weisbord SD, Mor MK, Resnick AL, Hartwig KC, Sonel AF, Fine MJ, et al. Prevention, incidence, and outcomes of contrast-induced acute kidney injury. Arch Intern Med. 2008;168(12):1325-1332.
How does hyperglycemia affect morbidity and mortality in children admitted to a community pediatric hospital?
Background: Inpatient hyperglycemia in adult patients is a predictor of poor clinical outcomes. The association of hyperglycemia and clinical outcomes in children admitted to a general community hospital has not been studied.
Study design: Retrospective observational cohort study.
Setting: A community pediatric hospital in Atlanta, Ga.
Synopsis: Review of medical records of 903 consecutive pediatric patients admitted to critical and non-critical areas took place. Of these, 542 patients constituted the study population. The study excluded 342 patients who didn’t have a blood glucose measurement. Hyperglycemia was defined as an admission or in-hospital blood glucose greater than 120mg/dl.
One-fourth of the children admitted to the hospital had hyperglycemia, most without a prior history of diabetes. The presence of hyperglycemia on admission was not associated with increased length of stay (LOS) or increased mortality. Children with hyperglycemia were more likely to be admitted to the ICU and had longer ICU LOS.
This was a retrospective study conducted at a single site whose demographics and disease spectrum may differ from those of other institutions. There were an insufficient number of deaths to make any conclusions regarding the impact of hyperglycemia on mortality. Prospective, randomized, multicenter trials are needed to better elucidate the effects of in-patient hyperglycemia.
Bottom line: Hyperglycemia is common in children with or without diabetes admitted to the hospital, and is associated with increased ICU admissions and ICU length of stay. Its connection to mortality is inconclusive.
Citation: Palaio A, Smiley D, Ceron M, Klein R, Cho IS, Mejia R, et al. Prevalence and clinical outcome of inpatient hyperglycemia in a community pediatric hospital. J Hosp Med.2008;3(3):212-217.
Literature at a Glance
- Drug-eluting stents decrease the need for revascularization.
- Case volume is related to hospital performance assessment.
- Prolonged QRS duration in patients with CHF is associated with increased morbidity and mortality.
- For out-of-hospital ACLS, vasopressin plus epinephrine is not better than vasopressin alone.
- Oral rivaroxaban is more efficacious than enoxaparin for VTE prophylaxis after total hip replacement.
- LMWH and UFH offer similar perioperative VTE prophylaxis benefit in patients with cancer.
- Salmeterol added to inhaled corticosteroids decreases severe asthma exacerbations.
- Early invasive strategy has unclear benefit in low-risk women with unstable angina or NSTEMI.
- Strategies to prevent contrast-induced acute kidney injury are not uniform.
- Hyperglycemia in hospitalized children is common and associated with ICU admission.
Do drug-eluting stents improve outcomes after ST-elevation myocardial infarction (STEMI)?
Background: Drug-eluting stents reduce restenosis rates compared to bare-metal stents. However, there is concern drug-eluting stents increase the risk of stent thrombosis leading to MI and death. Prior studies compared patients who received bare-metal versus those who received drug-eluting stents. Outcomes on a population level might provide new insight.
Study design: Observational study.
Setting: 100% national sample of patients 65 and older who received a coronary stent from 2002-05 enrolled in the traditional fee-for-service Medicare program.
Synopsis: 38,917 patients in the pre-drug-eluting-stent era from October 2002 to March 2003 received bare-metal stents. Nearly 62% of 28,086 patients studied from September to December 2003 received drug-eluting stents. The remaining 38.5% received bare-metal stents. Outcomes of percutaneous coronary intervention (PCI), coronary artery bypass grafting (CABG), STEMI, and death were observed through December 31, 2005.
Patients in the drug-eluting-stent era had a lower two-year risk for repeat revascularization compared to patients in the bare-metal-stent era. In the drug-eluting versus bare-metal eras, repeat PCI was 17.1% versus 20.0% (p<0.001) and need for CABG was 2.7% versus 4.2% (p<0.01). Comparing adjusted outcomes for death, or STEMI, at two years, the two groups appeared similar.
The study did have limitations: the data only reflect sirolimus stents, the authors could not assess dual-antiplatelet therapy or obtain information on coronary anatomy or procedure details to account for selection bias in stent utilization, and the patients were all Medicare beneficiaries.
Bottom line: Drug-eluting stents are associated with fewer repeat revascularization procedures than bare-metal stents, but have not shown a significant improvement in the subsequent risk of STEMI or death.
Citation: Malenka DJ, Kaplan AV, Lucas FL, Sharp SM, Skinner JA. Outcomes following coronary stenting in the era of bare-metal vs. the era of drug-eluting stents. JAMA 2008;299(24):2868-2877.
Does case volume affect hospital performance for publicly reported process measures?
Background: Hospitals are increasingly graded and compared to one another. “Top medical centers” are defined as those within the top 10% of hospitals in specified performance measures. Hospitals with large and small case volumes might not be compared evenly and fairly.
Study design: Eight publicly reported process measures for acute myocardial infarction (AMI) were compared to hospital case volume, process performance, and label as “top hospital.”
Setting: Data were analyzed from the Hospital Quality Alliance for 3,761 hospitals from January to December 2005.
Synopsis: Hospitals with large case volume overall had better process performance. For example, looking at use of beta-blockers in patients with AMI on arrival to a hospital, small-volume hospitals (<10 AMI cases) averaged 72% while large volume (>100 AMI cases) averaged 80% (p<0.001). However, hospitals with small case volumes were more likely to receive “top hospital” rating even when hospitals with very low case volumes were excluded.
Hospital quality reporting that does not account for case volume is misleading to hospitals and consumers. In this study, larger-volume hospitals appeared to perform better in process measures, but were less likely to receive “top hospital” rating.
Bottom line: Hospitals with large and small case volumes can easily be compared to one another for process measures in AMI.
Citation: O’Brien SM, DeLong ER, Peterson ED. Impact of case volume on hospital performance assessment. Arch Intern Med. 2008;168(12):1277-1284.
What is the predictive value of QRS duration in patients hospitalized with worsening CHF?
Background: In outpatients, a prolonged QRS duration (greater than 120 ms) is associated with increased mortality. Its value in the inpatient setting is unclear. For patients hospitalized with CHF exacerbations, establishing the value of QRS duration may allow for tailored management.
Study design: Retrospective post hoc analysis from the Efficacy of Vasopressin Antagonism in Heart Failure Outcome Study with Tolvaptan (EVEREST).
Setting: 4,133 patients were enrolled from North American, South American, and European sites.
Synopsis: Of 2,962 patients included in the final post hoc analysis, 1,321 (44.6%) had a prolonged QRS duration. During a median follow up of 9.9 months, the all-cause mortality rate was 18.7% for patients with a normal baseline QRS duration and 28.1% for patients with a prolonged baseline QRS.
After adjusting for confounding variables, patients with a prolonged baseline QRS had a 24% increased risk of all-cause mortality and a 28% increased risk for a composite endpoint of cardiac mortality or hospitalization for heart failure exacerbation.
The retrospective nature of the analysis represents the major limitation of this study. In addition, most of the enrolled patients were white, which limits the studies generalizability to other ethnic groups.
Bottom Line: A prolonged QRS duration for patients admitted with decompensated left ventricular heart failure is common and may be associated with increased morbidity and mortality.
Citation: Wang NC, Maggioni AP, Konstam MA, Zannad F, Drasa HB, Burnett JC, et al. Clinical implications of QRS duration in patients hospitalized with worsening heart failure and reduced left ventricular ejection fraction. JAMA. 2008;299(22):2656-2666.
For patients with out-of-hospital cardiac arrest, does the addition of vasopressin to epinephrine in a protocol for ACLS improve outcomes?
Background: The outcome for patients experiencing cardiac arrest who require vasopressors remains extremely poor. Despite disappointing data on vasopressin as an alternative treatment during cardiac arrest, a recent subgroup analysis suggested patients who received epinephrine and vasopressin together had superior clinical outcomes.
Study Design: Prospective multicenter randomized double-blind controlled trial.
Setting: 31 emergency medical service organizations in France.
Synopsis: Of the 2,894 patients, 20.7% of those receiving combination treatment (vasopressin plus epinephrine) survived to hospital admission versus 21.3% of those in the epinephrine-only group. For those same groups, 1.7% of combination and 2.3% of epinephrine-only patients survived to hospital discharge. No significant outcome differences were found in any group or subgroup analysis.
The study had lower-than-expected overall survival to hospital discharge, which may have handicapped its effort to find a true difference in treatment arms.
Bottom line: The addition of vasopressin to epinephrine in the treatment of out-of-hospital cardiac arrest does not improve outcomes.
Citation: Gueugniaud PY, David JS, Chanzy E, Hubert H, Dubien P, Mauriaucourt P, et al. Vasopressin and epinephrine vs. epinephrine alone in cardiopulmonary resuscitation. N Engl J Med. 2008;359(1):21-30.
Is oral rivaroxaban more efficacious than subcutaneous enoxaparin in preventing VTE after hip-replacement surgery?
Background: Venous thromboembolism (VTE) prophylaxis after total hip replacement (THR) is important but can be cumbersome because the most commonly used anticoagulants are either subcutaneous or require frequent monitoring. Rivaroxaban, an oral direct inhibitor of factor Xa may provide more convenient anticoagulation postoperatively. However, its efficacy and safety are unknown.
Study design: Randomized double-blind trial.
Setting: Multicenter study performed in 27 countries.
Synopsis: Patients undergoing THR surgery were randomized to oral rivaroxaban (10mg once daily without monitoring, started six to eight hours after surgery) or subcutaneous enoxaparin (40mg once daily, started 12 hours prior to surgery). After surgery, prophylaxis was administered for 35 days. The primary outcome was a composite of asymptomatic deep venous thrombosis (DVT), symptomatic DVT or pulmonary embolism (PE), or death from any cause at 36 days after surgery.
In the enoxaparin group, 3.7% of patients experienced the primary outcome. This decreased to 1.1% in the rivaroxaban group. Approximately one-third of events were symptomatic. Major bleeding occurred in 0.1% and 0.3% (p=NS) of patients in the enoxaparin and rivaroxaban groups, respectively.
The study is limited by the exclusion of 1,388 of the 4,541 patients (30.6%) randomized, primarily due to having inadequate venography. Also, because the majority of thromboembolic events were asymptomatic, the primary outcome overemphasizes the clinical difference.
Bottom line: Oral rivaroxaban without monitoring is more efficacious than, and as safe as, subcutaneous enoxaparin when used for VTE prophylaxis for THR.
Citation: Eriksson B, Borris LC, Friedman RJ, Hass S, Huisman MV, Kakkar AK, et al. Rivaroxaban versus enoxaparin for thromboprophylaxis after hip arthroplasty. N Engl J Med. 2008;358:2765-75.
Is LMWH more efficacious than UFH in preventing postoperative VTE in cancer patients?
Background: Patients with cancer are at increased risk for VTE and require prophylaxis to prevent this complication postoperatively. Low molecular weight heparin (LMWH) has proven more efficacious than subcutaneous unfractionated heparin (UFH) in other settings (e.g., DVT treatment). However, it is still unknown whether LMWH offers better prophylaxis compared to UFH for cancer patients undergoing surgery.
Study design: Systematic review and meta-analysis.
Setting: 14 randomized controlled trials.
Synopsis: Eleven trials exclusively examined patients with cancer (n=4006) and three trials reported data for cancer patients as subgroups (n=1816). There were in differences in mortality, pulmonary embolism, and symptomatic DVT rates between the two groups.
LMWH was associated with a decrease in total (asymptomatic or symptomatic) DVT (RR, 0.72; 95% CI, 0.55-0.94). Rates of major bleeding, minor bleeding, and intraoperative blood loss were similar between the two treatments.
This meta-analysis is limited because 12 remaining trials (n=3185) also enrolled cancer patients but did not provide specific data for the cancer patient subgroup. The study also is limited by the heterogeneity of the original trials, including utilizing varying LMWHs and dosing regimens, numerous types of surgeries, and a wide range of neoplasms.
Bottom line: LMWH does not decrease mortality, pulmonary embolism, or symptomatic DVT compared to UFH in cancer patients undergoing surgery.
Citation: Akl EA, Terrenato I, Barba M, Sperati F, Sempos EV, Muti P, et al. Low-molecular-weight heparin vs. unfractionated heparin for perioperative thromboprophylaxis in patients with cancer. Arch Intern Med. 2008;168:1261-9.
Does salmeterol added to inhaled corticosteroids improve severe asthma-related events?
Background: Asthma is a chronic disease causing major morbidity and mortality worldwide. Disease guidelines recommend all patients with persistent asthma be treated with inhaled corticosteroids. These same guidelines recommend adding a long-acting beta-agonist for patients whose symptoms persist. However, the safety of this practice has come under scrutiny.
Study design: Meta-analysis.
Setting: Sixty-six randomized, controlled trials conducted worldwide.
Synopsis: Analysis included 66 GlaxoSmithKline trials with a total of 20,966 patients with persistent asthma. Patients used either salmeterol (50mcg twice daily) plus inhaled corticosteroid (10,400 patients) or inhaled corticosteroid alone (10,566 patients).
Results showed no differences in asthma-related hospitalizations, asthma-related intubations, or deaths between the two groups. However, due to the low number of events, definitive conclusions are difficult to make. Severe asthma exacerbations requiring systemic corticosteroids significantly decreased in the inhaled corticosteroid plus salmeterol group.
The study is limited by it inclusion of only those trials sponsored by GlaxoSmithKline and by the short duration of most of the studies. Additionally, the studies included in the analysis used clinical outcomes as secondary endpoints.
Bottom line: Adding salmeterol to inhaled corticosteroid decreases severe asthma exacerbations and is likely safe, but does not have an effect on asthma-related hospitalization or death.
Citation: Bateman E, Nelson H, Bousquet J, Kral K, Sutton L, Ortega H, et.al. Meta-analysis: Effects of adding salmeterol to inhaled corticosteroids on serious asthma-related events. Annals Intern Med. 2008;149:33-42.
Is an early invasive strategy effective in women with unstable angina or NSTEMI?
Background: Despite many trials showing the value of an early invasive strategy for patients with non-ST-segment elevation acute coronary syndrome (NSTE ACS), data from several trials question this benefit in women. Some trials show higher risk of death and myocardial infarction (MI) in subgroup analysis of women.
Study Design: Meta-analysis.
Setting: Eight randomized, controlled trials conducted worldwide.
Synopsis: Analysis included eight trials with 10,412 patients (3,075 women) with NSTE ACS. The invasive group (5,083 patients) was defined as those referred for coronary angiography with subsequent intervention as needed. The composite endpoint of death, MI, or rehospitalization within 12 months with ACS occurred in 21.1% of the invasive group and 25.9% of the medically managed group (OR, 0.78; CI, 0.61-0.98).
The subgroup, including only women, had a non-statistically significant OR of 0.81 (CI, 0.65-1.01), including no effect on all-cause mortality, nonfatal MI, or the composite of death and MI. However, women with high-risk features (elevated biomarkers) undergoing the invasive strategy had a significant reduction in the composite endpoint (OR, 0.67; CI, 0.50-0.88).
The study is limited by the use of subgroup analysis, secondary endpoints, heterogeneity between trials, and possible publication bias.
Bottom line: Early invasive strategy is effective in men and high-risk women with NSTE ACS, but not in low-risk women.
Citation: O’Donoghue M, Boden W, Braunwald E, Cannon CP, Clayton TC, Winter RJ, et.al. Early invasive vs. conservative treatment strategies in women and men with unstable angina and non-ST-segment elevation myocardial infarction. JAMA. 2008;300:71-80.
What strategies are used to prevent contrast-induced acute kidney injury?
Background: Contrast-induced acute kidney injury (CIAKI) is a condition potentially amenable to preventive care. Several trials have identified intravenous hydration, N-acetylcysteine, and withdrawal of NSAIDS as interventions that reduce the possibility of CIAKI in high-risk patients. Little is known about whether healthcare providers routinely use these strategies.
Study design: Prospective observational cohort study.
Setting: Veterans Affairs (VA) Pittsburgh Healthcare System.
Synopsis: 11,410 patients scheduled for radiographic procedures were screened. After exclusion criteria and eligibility, 660 patients with an estimated glomerular filtration rate less than 60ml/min/1.73m2 were identified. Usage of intravenous fluids, N-acetylcysteine, and discontinuation of NSAIDS were recorded. Serum creatinine (SCr) was measured 48 to 96 hours post-procedure. CIAKI was defined as relative increase in SCr from baseline (≥25%, ≥50% and ≥100%) and absolute increase in SCr levels from baseline (≥0.25, ≥0.5, and ≥1.0). CIAKI association with adverse outcomes was evaluated by tracking 30-day mortality, need for dialysis, and hospitalization.
The incidence of CIAKI was less common in patients undergoing CT scans versus those having angiograms. Adverse 30-day outcomes were uncommon. Pre- and post-procedure intravenous hydration was administered to 40% of study patients, more commonly with coronary angiogram than with computed tomography (91.2% vs. 16%, p<0.0001). N-acetylcysteine was administered to 39.2%. Only 6.8% of those taking NSAIDS reported being told to discontinue the medication.
Study limitations include the small sample size and the single site location, both limiting generalizability.
Bottom line: Clinically significant CIAKI is uncommon, and preventive care is not uniformly implemented in patients undergoing contrast-enhanced radiographic procedures.
Citation: Weisbord SD, Mor MK, Resnick AL, Hartwig KC, Sonel AF, Fine MJ, et al. Prevention, incidence, and outcomes of contrast-induced acute kidney injury. Arch Intern Med. 2008;168(12):1325-1332.
How does hyperglycemia affect morbidity and mortality in children admitted to a community pediatric hospital?
Background: Inpatient hyperglycemia in adult patients is a predictor of poor clinical outcomes. The association of hyperglycemia and clinical outcomes in children admitted to a general community hospital has not been studied.
Study design: Retrospective observational cohort study.
Setting: A community pediatric hospital in Atlanta, Ga.
Synopsis: Review of medical records of 903 consecutive pediatric patients admitted to critical and non-critical areas took place. Of these, 542 patients constituted the study population. The study excluded 342 patients who didn’t have a blood glucose measurement. Hyperglycemia was defined as an admission or in-hospital blood glucose greater than 120mg/dl.
One-fourth of the children admitted to the hospital had hyperglycemia, most without a prior history of diabetes. The presence of hyperglycemia on admission was not associated with increased length of stay (LOS) or increased mortality. Children with hyperglycemia were more likely to be admitted to the ICU and had longer ICU LOS.
This was a retrospective study conducted at a single site whose demographics and disease spectrum may differ from those of other institutions. There were an insufficient number of deaths to make any conclusions regarding the impact of hyperglycemia on mortality. Prospective, randomized, multicenter trials are needed to better elucidate the effects of in-patient hyperglycemia.
Bottom line: Hyperglycemia is common in children with or without diabetes admitted to the hospital, and is associated with increased ICU admissions and ICU length of stay. Its connection to mortality is inconclusive.
Citation: Palaio A, Smiley D, Ceron M, Klein R, Cho IS, Mejia R, et al. Prevalence and clinical outcome of inpatient hyperglycemia in a community pediatric hospital. J Hosp Med.2008;3(3):212-217.