User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Personalizing treatment plans for older patients with T2D
In the United States, type 2 diabetes (T2D) more commonly affects people older than 40 years, but it is most prevalent among adults over age 65, affecting more than 29% of this population. The heterogeneity in the health and functional status of older adults presents a challenge in the management and treatment of older patients with T2D. Moreover, there is an increased risk for health-related comorbidities and complications from diabetes treatment (for example, hypoglycemia) in older adults. Physiologic changes, such as decreased renal function, cognitive decline, and sarcopenia, may lead to an increased risk for adverse reactions to medications and require an individualized treatment approach. Although there have been a limited number of randomized controlled studies targeting older adults with multiple comorbidities and poor health status, subanalyses of diabetes trials with a subpopulation of older adults have provided additional evidence to better guide therapeutic approaches in caring for older patients with T2D.
Here’s a guide to developing personalized therapeutic regimens for older patients with T2D using lifestyle interventions, pharmacotherapy, and diabetes technology.
Determining an optimal glycemic target
An important first step in diabetes treatment is to determine the optimal glycemic target for patients. Although data support intensive glycemic control (hemoglobin A1c < 7%) to prevent complications from diabetes in younger patients with recently diagnosed disease, the data are less compelling in trials involving older populations with longer durations of T2D. One observational study with 71,092 older adults over age 60 reported a U-shaped correlation between A1c and mortality, with higher risks for mortality in those with A1c levels < 6% and ≥ 11%, compared with those with A1c levels of 6%-9%. Risks for any diabetes complications were higher at an A1c level ≥ 8%. Another observational study reported a U-shaped association between A1c and mortality, with the lowest hazard ratio for mortality at an A1c level of about 7.5%. Similarly, the ACCORD trial, which included older and middle-aged patients with T2D who had or were at risk for atherosclerotic cardiovascular disease, found that mortality followed a U-shaped curve at the low (A1c < 7%) and high (A1c > 8%) ends in patients who were given standard glycemic therapy. Hence, there has been a general trend to recommend less strict glycemic control in older adults.
However, it is important to remember that older patients with T2D are a heterogeneous group. The spectrum includes adults with recent-onset diabetes with no or few complications, those with long-standing diabetes and many complications, and frail older adults with multiple comorbidities and complications. Determining the optimal glycemic target for an older patient with T2D requires assessment not only of the patient’s medical status and comorbidities but also functional status, cognitive and psychological health, social situation, individual preferences, and life expectancy. The American Diabetes Association Standards of Medical Care in Diabetes provides the following guidance in determining the optimal glycemic control for older adults:
- Healthy adults with few coexisting chronic illnesses and intact cognitive and functional status should have an A1c level < 7.0%-7.5%.
- Adults with complex or intermediate comorbidities (multiple coexisting chronic illnesses, or two or more instrumental activities of daily living impairments, or mild to moderate cognitive impairment) should have an A1c level < 8.0%.
- Patients with poor health (long-term care or end-stage chronic illnesses or moderate to severe cognitive impairment or two or more activities of daily living impairments) should avoid reliance on A1c, and the goal is to avoid hypoglycemia and symptomatic hyperglycemia.
Because older patients are at a higher risk for complications and adverse effects from polypharmacy, regular assessments are recommended and treatment plans should be routinely reviewed and modified to avoid overtreatment.
Lifestyle interventions and pharmacotherapy
Lifestyle interventions, such as exercise, optimal nutrition, and protein intake, are integral in treating older patients with T2D. Older adults should engage in regular exercise (that is, aerobic activity, weight-bearing exercise, or resistance training), and the activity should be customized to frailty status. Regular exercise improves insulin sensitivity and glucose control, enhances functional status, and provides cardiometabolic benefits. Optimal nutrition and adequate protein intake are also important to prevent the development or worsening of sarcopenia and frailty.
Several factors must be considered when choosing pharmacotherapy for T2D treatment in older adults. These patients are at higher risk for adverse reactions to medications that can trigger hypoglycemia and serious cardiovascular events, and worsen cognitive function. Therefore, side effects should always be reviewed when choosing antidiabetic drugs. The complexity of treatment plans needs to be matched with the patients’ self-management abilities and available social support. Medication costs and insurance coverage should be considered because many older adults live on a fixed income. Although limited, data exist on the safety and efficacy of some glucose-lowering agents in older adults, which can provide guidance for choosing the optimal therapy for these patients.
Among the insulin sensitizers, metformin is most commonly used because of its efficacy, low risk for hypoglycemia, and affordability. Metformin can be safely used in the setting of reduced renal function down to the estimated glomerular filtration rate ≥ 30 mL/min per 1.73 m2. However, metformin should be avoided in patients with more advanced renal disease, liver failure, or heart failure. In older patients with T2D, potential concerns of metformin include gastrointestinal side effects, leading to reduced appetite, mild weight loss, and risk for vitamin B12 deficiency.
Pioglitazone, an oral antidiabetic in the thiazolidinedione (TZD) class, also targets insulin resistance and may provide some cardiovascular benefits. However, these agents are not commonly used in treating older patients with T2D owing to associated risk for edema, heart failure, osteoporosis/fractures, and bladder cancer.
Sulfonylureas and meglitinides are insulin secretagogues, which can promote insulin release independent of glucose levels. Sulfonylureas are typically avoided in older patients because they are associated with high risk for hypoglycemia. Meglitinides have a lower hypoglycemia risk than sulfonylureas because of their short duration of action; however, they are more expensive and require multiple daily administration, which can lead to issues with adherence.
Since 2008, there have been numerous cardiovascular outcomes trials assessing the safety and efficacy of T2D therapies that included a subpopulation of older patients either with cardiovascular disease or at high risk for cardiovascular disease. Post hoc analysis of data from these trials and smaller studies dedicated to older adults demonstrated the safety and efficacy of most incretin-based therapies and sodium-glucose cotransporter 2 (SGLT2) inhibitors in these patients. These newer medications have low hypoglycemia risk if not used in combination with insulin or insulin secretagogues.
Dipeptidyl peptidase 4 (DPP-4) inhibitors have the mildest side effect profile. However, they can be expensive and not reduce major adverse cardiovascular outcomes, and one agent, saxagliptin, has been associated with increased risk for heart failure hospitalization. Some glucagon-like peptide 1 (GLP-1) receptor agonists are effective in reducing major adverse cardiovascular events (cardiovascular deaths, stroke, and myocardial infarction) in patients older and younger than age 65. However, the gastrointestinal side effects and weight loss associated with this medication can be problematic for older patients. Most of the GLP-1 receptor agonists are injectables, which require good visual, motor, and cognitive skills for administration. SGLT2 inhibitors offer benefits for patients with T2D who have established cardiovascular disease, heart failure, and chronic kidney disease, with possible greater cardiovascular benefits in older adults. Adverse effects associated with SGLT2 inhibitors, such as weight loss, volume depletion, urinary incontinence, and genitourinary infections, may be a concern in older patients with T2D who are using these medications.
Because the insulin-secreting capacity of the pancreas declines with age, insulin therapy may be required for treatment of T2D in older patients. Insulin therapy can be complex and consideration must be given to patients’ social circumstances, as well as their physical and cognitive abilities. Older adults may need adaptive strategies, such as additional lighting, magnification glass, and premixed syringes. Simplification of complex insulin therapy (discontinuation of prandial insulin or sliding scale, changing timing of basal insulin) and use of insulin analogs with lower hypoglycemia risks should be considered. Weight gain as a result of insulin therapy may be beneficial in older adults with sarcopenia or frailty.
T2D technology for glycemic improvement
There have been major technological advancements in diabetes therapy. Continuous glucose monitors (CGMs) and automated insulin delivery systems can improve glycemic control, decrease the rate of hypoglycemia, and enhance the quality of life of older patients. Most of the studies evaluating the use of automated insulin delivery systems in older patients have focused on those with type 1 diabetes and demonstrated improvement in glycemic control and/or reduced hypoglycemia. The DIAMOND trial demonstrated improved A1c and reduced glycemic variability with the use of CGM in adults older than 60 years with either type 1 or type 2 diabetes on multiple daily injections. Bluetooth-enabled “smart” insulin pens, which record the time and dose of insulin administrations, can also be a great asset in caring for older patients, especially those with cognitive impairment. With better insurance coverage, diabetes technologies may become more accessible and an asset in treating older patients with T2D.
In conclusion, management of T2D in older adults requires an individualized approach because of the heterogeneity in their health and functional status. Because cardiovascular disease is the leading cause of mortality in older patients with T2D, treatment plans should also address frequently coexisting cardiovascular risk factors, such as hypertension and hyperlipidemia. Clinicians should consider patients’ overall health, comorbidities, cognitive and functional status, social support systems, preferences, and life expectancy when developing individualized therapeutic plans.
Dr. Gunawan is an assistant professor in the department of internal medicine at UT Southwestern Medical Center, Dallas. She reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
In the United States, type 2 diabetes (T2D) more commonly affects people older than 40 years, but it is most prevalent among adults over age 65, affecting more than 29% of this population. The heterogeneity in the health and functional status of older adults presents a challenge in the management and treatment of older patients with T2D. Moreover, there is an increased risk for health-related comorbidities and complications from diabetes treatment (for example, hypoglycemia) in older adults. Physiologic changes, such as decreased renal function, cognitive decline, and sarcopenia, may lead to an increased risk for adverse reactions to medications and require an individualized treatment approach. Although there have been a limited number of randomized controlled studies targeting older adults with multiple comorbidities and poor health status, subanalyses of diabetes trials with a subpopulation of older adults have provided additional evidence to better guide therapeutic approaches in caring for older patients with T2D.
Here’s a guide to developing personalized therapeutic regimens for older patients with T2D using lifestyle interventions, pharmacotherapy, and diabetes technology.
Determining an optimal glycemic target
An important first step in diabetes treatment is to determine the optimal glycemic target for patients. Although data support intensive glycemic control (hemoglobin A1c < 7%) to prevent complications from diabetes in younger patients with recently diagnosed disease, the data are less compelling in trials involving older populations with longer durations of T2D. One observational study with 71,092 older adults over age 60 reported a U-shaped correlation between A1c and mortality, with higher risks for mortality in those with A1c levels < 6% and ≥ 11%, compared with those with A1c levels of 6%-9%. Risks for any diabetes complications were higher at an A1c level ≥ 8%. Another observational study reported a U-shaped association between A1c and mortality, with the lowest hazard ratio for mortality at an A1c level of about 7.5%. Similarly, the ACCORD trial, which included older and middle-aged patients with T2D who had or were at risk for atherosclerotic cardiovascular disease, found that mortality followed a U-shaped curve at the low (A1c < 7%) and high (A1c > 8%) ends in patients who were given standard glycemic therapy. Hence, there has been a general trend to recommend less strict glycemic control in older adults.
However, it is important to remember that older patients with T2D are a heterogeneous group. The spectrum includes adults with recent-onset diabetes with no or few complications, those with long-standing diabetes and many complications, and frail older adults with multiple comorbidities and complications. Determining the optimal glycemic target for an older patient with T2D requires assessment not only of the patient’s medical status and comorbidities but also functional status, cognitive and psychological health, social situation, individual preferences, and life expectancy. The American Diabetes Association Standards of Medical Care in Diabetes provides the following guidance in determining the optimal glycemic control for older adults:
- Healthy adults with few coexisting chronic illnesses and intact cognitive and functional status should have an A1c level < 7.0%-7.5%.
- Adults with complex or intermediate comorbidities (multiple coexisting chronic illnesses, or two or more instrumental activities of daily living impairments, or mild to moderate cognitive impairment) should have an A1c level < 8.0%.
- Patients with poor health (long-term care or end-stage chronic illnesses or moderate to severe cognitive impairment or two or more activities of daily living impairments) should avoid reliance on A1c, and the goal is to avoid hypoglycemia and symptomatic hyperglycemia.
Because older patients are at a higher risk for complications and adverse effects from polypharmacy, regular assessments are recommended and treatment plans should be routinely reviewed and modified to avoid overtreatment.
Lifestyle interventions and pharmacotherapy
Lifestyle interventions, such as exercise, optimal nutrition, and protein intake, are integral in treating older patients with T2D. Older adults should engage in regular exercise (that is, aerobic activity, weight-bearing exercise, or resistance training), and the activity should be customized to frailty status. Regular exercise improves insulin sensitivity and glucose control, enhances functional status, and provides cardiometabolic benefits. Optimal nutrition and adequate protein intake are also important to prevent the development or worsening of sarcopenia and frailty.
Several factors must be considered when choosing pharmacotherapy for T2D treatment in older adults. These patients are at higher risk for adverse reactions to medications that can trigger hypoglycemia and serious cardiovascular events, and worsen cognitive function. Therefore, side effects should always be reviewed when choosing antidiabetic drugs. The complexity of treatment plans needs to be matched with the patients’ self-management abilities and available social support. Medication costs and insurance coverage should be considered because many older adults live on a fixed income. Although limited, data exist on the safety and efficacy of some glucose-lowering agents in older adults, which can provide guidance for choosing the optimal therapy for these patients.
Among the insulin sensitizers, metformin is most commonly used because of its efficacy, low risk for hypoglycemia, and affordability. Metformin can be safely used in the setting of reduced renal function down to the estimated glomerular filtration rate ≥ 30 mL/min per 1.73 m2. However, metformin should be avoided in patients with more advanced renal disease, liver failure, or heart failure. In older patients with T2D, potential concerns of metformin include gastrointestinal side effects, leading to reduced appetite, mild weight loss, and risk for vitamin B12 deficiency.
Pioglitazone, an oral antidiabetic in the thiazolidinedione (TZD) class, also targets insulin resistance and may provide some cardiovascular benefits. However, these agents are not commonly used in treating older patients with T2D owing to associated risk for edema, heart failure, osteoporosis/fractures, and bladder cancer.
Sulfonylureas and meglitinides are insulin secretagogues, which can promote insulin release independent of glucose levels. Sulfonylureas are typically avoided in older patients because they are associated with high risk for hypoglycemia. Meglitinides have a lower hypoglycemia risk than sulfonylureas because of their short duration of action; however, they are more expensive and require multiple daily administration, which can lead to issues with adherence.
Since 2008, there have been numerous cardiovascular outcomes trials assessing the safety and efficacy of T2D therapies that included a subpopulation of older patients either with cardiovascular disease or at high risk for cardiovascular disease. Post hoc analysis of data from these trials and smaller studies dedicated to older adults demonstrated the safety and efficacy of most incretin-based therapies and sodium-glucose cotransporter 2 (SGLT2) inhibitors in these patients. These newer medications have low hypoglycemia risk if not used in combination with insulin or insulin secretagogues.
Dipeptidyl peptidase 4 (DPP-4) inhibitors have the mildest side effect profile. However, they can be expensive and not reduce major adverse cardiovascular outcomes, and one agent, saxagliptin, has been associated with increased risk for heart failure hospitalization. Some glucagon-like peptide 1 (GLP-1) receptor agonists are effective in reducing major adverse cardiovascular events (cardiovascular deaths, stroke, and myocardial infarction) in patients older and younger than age 65. However, the gastrointestinal side effects and weight loss associated with this medication can be problematic for older patients. Most of the GLP-1 receptor agonists are injectables, which require good visual, motor, and cognitive skills for administration. SGLT2 inhibitors offer benefits for patients with T2D who have established cardiovascular disease, heart failure, and chronic kidney disease, with possible greater cardiovascular benefits in older adults. Adverse effects associated with SGLT2 inhibitors, such as weight loss, volume depletion, urinary incontinence, and genitourinary infections, may be a concern in older patients with T2D who are using these medications.
Because the insulin-secreting capacity of the pancreas declines with age, insulin therapy may be required for treatment of T2D in older patients. Insulin therapy can be complex and consideration must be given to patients’ social circumstances, as well as their physical and cognitive abilities. Older adults may need adaptive strategies, such as additional lighting, magnification glass, and premixed syringes. Simplification of complex insulin therapy (discontinuation of prandial insulin or sliding scale, changing timing of basal insulin) and use of insulin analogs with lower hypoglycemia risks should be considered. Weight gain as a result of insulin therapy may be beneficial in older adults with sarcopenia or frailty.
T2D technology for glycemic improvement
There have been major technological advancements in diabetes therapy. Continuous glucose monitors (CGMs) and automated insulin delivery systems can improve glycemic control, decrease the rate of hypoglycemia, and enhance the quality of life of older patients. Most of the studies evaluating the use of automated insulin delivery systems in older patients have focused on those with type 1 diabetes and demonstrated improvement in glycemic control and/or reduced hypoglycemia. The DIAMOND trial demonstrated improved A1c and reduced glycemic variability with the use of CGM in adults older than 60 years with either type 1 or type 2 diabetes on multiple daily injections. Bluetooth-enabled “smart” insulin pens, which record the time and dose of insulin administrations, can also be a great asset in caring for older patients, especially those with cognitive impairment. With better insurance coverage, diabetes technologies may become more accessible and an asset in treating older patients with T2D.
In conclusion, management of T2D in older adults requires an individualized approach because of the heterogeneity in their health and functional status. Because cardiovascular disease is the leading cause of mortality in older patients with T2D, treatment plans should also address frequently coexisting cardiovascular risk factors, such as hypertension and hyperlipidemia. Clinicians should consider patients’ overall health, comorbidities, cognitive and functional status, social support systems, preferences, and life expectancy when developing individualized therapeutic plans.
Dr. Gunawan is an assistant professor in the department of internal medicine at UT Southwestern Medical Center, Dallas. She reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
In the United States, type 2 diabetes (T2D) more commonly affects people older than 40 years, but it is most prevalent among adults over age 65, affecting more than 29% of this population. The heterogeneity in the health and functional status of older adults presents a challenge in the management and treatment of older patients with T2D. Moreover, there is an increased risk for health-related comorbidities and complications from diabetes treatment (for example, hypoglycemia) in older adults. Physiologic changes, such as decreased renal function, cognitive decline, and sarcopenia, may lead to an increased risk for adverse reactions to medications and require an individualized treatment approach. Although there have been a limited number of randomized controlled studies targeting older adults with multiple comorbidities and poor health status, subanalyses of diabetes trials with a subpopulation of older adults have provided additional evidence to better guide therapeutic approaches in caring for older patients with T2D.
Here’s a guide to developing personalized therapeutic regimens for older patients with T2D using lifestyle interventions, pharmacotherapy, and diabetes technology.
Determining an optimal glycemic target
An important first step in diabetes treatment is to determine the optimal glycemic target for patients. Although data support intensive glycemic control (hemoglobin A1c < 7%) to prevent complications from diabetes in younger patients with recently diagnosed disease, the data are less compelling in trials involving older populations with longer durations of T2D. One observational study with 71,092 older adults over age 60 reported a U-shaped correlation between A1c and mortality, with higher risks for mortality in those with A1c levels < 6% and ≥ 11%, compared with those with A1c levels of 6%-9%. Risks for any diabetes complications were higher at an A1c level ≥ 8%. Another observational study reported a U-shaped association between A1c and mortality, with the lowest hazard ratio for mortality at an A1c level of about 7.5%. Similarly, the ACCORD trial, which included older and middle-aged patients with T2D who had or were at risk for atherosclerotic cardiovascular disease, found that mortality followed a U-shaped curve at the low (A1c < 7%) and high (A1c > 8%) ends in patients who were given standard glycemic therapy. Hence, there has been a general trend to recommend less strict glycemic control in older adults.
However, it is important to remember that older patients with T2D are a heterogeneous group. The spectrum includes adults with recent-onset diabetes with no or few complications, those with long-standing diabetes and many complications, and frail older adults with multiple comorbidities and complications. Determining the optimal glycemic target for an older patient with T2D requires assessment not only of the patient’s medical status and comorbidities but also functional status, cognitive and psychological health, social situation, individual preferences, and life expectancy. The American Diabetes Association Standards of Medical Care in Diabetes provides the following guidance in determining the optimal glycemic control for older adults:
- Healthy adults with few coexisting chronic illnesses and intact cognitive and functional status should have an A1c level < 7.0%-7.5%.
- Adults with complex or intermediate comorbidities (multiple coexisting chronic illnesses, or two or more instrumental activities of daily living impairments, or mild to moderate cognitive impairment) should have an A1c level < 8.0%.
- Patients with poor health (long-term care or end-stage chronic illnesses or moderate to severe cognitive impairment or two or more activities of daily living impairments) should avoid reliance on A1c, and the goal is to avoid hypoglycemia and symptomatic hyperglycemia.
Because older patients are at a higher risk for complications and adverse effects from polypharmacy, regular assessments are recommended and treatment plans should be routinely reviewed and modified to avoid overtreatment.
Lifestyle interventions and pharmacotherapy
Lifestyle interventions, such as exercise, optimal nutrition, and protein intake, are integral in treating older patients with T2D. Older adults should engage in regular exercise (that is, aerobic activity, weight-bearing exercise, or resistance training), and the activity should be customized to frailty status. Regular exercise improves insulin sensitivity and glucose control, enhances functional status, and provides cardiometabolic benefits. Optimal nutrition and adequate protein intake are also important to prevent the development or worsening of sarcopenia and frailty.
Several factors must be considered when choosing pharmacotherapy for T2D treatment in older adults. These patients are at higher risk for adverse reactions to medications that can trigger hypoglycemia and serious cardiovascular events, and worsen cognitive function. Therefore, side effects should always be reviewed when choosing antidiabetic drugs. The complexity of treatment plans needs to be matched with the patients’ self-management abilities and available social support. Medication costs and insurance coverage should be considered because many older adults live on a fixed income. Although limited, data exist on the safety and efficacy of some glucose-lowering agents in older adults, which can provide guidance for choosing the optimal therapy for these patients.
Among the insulin sensitizers, metformin is most commonly used because of its efficacy, low risk for hypoglycemia, and affordability. Metformin can be safely used in the setting of reduced renal function down to the estimated glomerular filtration rate ≥ 30 mL/min per 1.73 m2. However, metformin should be avoided in patients with more advanced renal disease, liver failure, or heart failure. In older patients with T2D, potential concerns of metformin include gastrointestinal side effects, leading to reduced appetite, mild weight loss, and risk for vitamin B12 deficiency.
Pioglitazone, an oral antidiabetic in the thiazolidinedione (TZD) class, also targets insulin resistance and may provide some cardiovascular benefits. However, these agents are not commonly used in treating older patients with T2D owing to associated risk for edema, heart failure, osteoporosis/fractures, and bladder cancer.
Sulfonylureas and meglitinides are insulin secretagogues, which can promote insulin release independent of glucose levels. Sulfonylureas are typically avoided in older patients because they are associated with high risk for hypoglycemia. Meglitinides have a lower hypoglycemia risk than sulfonylureas because of their short duration of action; however, they are more expensive and require multiple daily administration, which can lead to issues with adherence.
Since 2008, there have been numerous cardiovascular outcomes trials assessing the safety and efficacy of T2D therapies that included a subpopulation of older patients either with cardiovascular disease or at high risk for cardiovascular disease. Post hoc analysis of data from these trials and smaller studies dedicated to older adults demonstrated the safety and efficacy of most incretin-based therapies and sodium-glucose cotransporter 2 (SGLT2) inhibitors in these patients. These newer medications have low hypoglycemia risk if not used in combination with insulin or insulin secretagogues.
Dipeptidyl peptidase 4 (DPP-4) inhibitors have the mildest side effect profile. However, they can be expensive and not reduce major adverse cardiovascular outcomes, and one agent, saxagliptin, has been associated with increased risk for heart failure hospitalization. Some glucagon-like peptide 1 (GLP-1) receptor agonists are effective in reducing major adverse cardiovascular events (cardiovascular deaths, stroke, and myocardial infarction) in patients older and younger than age 65. However, the gastrointestinal side effects and weight loss associated with this medication can be problematic for older patients. Most of the GLP-1 receptor agonists are injectables, which require good visual, motor, and cognitive skills for administration. SGLT2 inhibitors offer benefits for patients with T2D who have established cardiovascular disease, heart failure, and chronic kidney disease, with possible greater cardiovascular benefits in older adults. Adverse effects associated with SGLT2 inhibitors, such as weight loss, volume depletion, urinary incontinence, and genitourinary infections, may be a concern in older patients with T2D who are using these medications.
Because the insulin-secreting capacity of the pancreas declines with age, insulin therapy may be required for treatment of T2D in older patients. Insulin therapy can be complex and consideration must be given to patients’ social circumstances, as well as their physical and cognitive abilities. Older adults may need adaptive strategies, such as additional lighting, magnification glass, and premixed syringes. Simplification of complex insulin therapy (discontinuation of prandial insulin or sliding scale, changing timing of basal insulin) and use of insulin analogs with lower hypoglycemia risks should be considered. Weight gain as a result of insulin therapy may be beneficial in older adults with sarcopenia or frailty.
T2D technology for glycemic improvement
There have been major technological advancements in diabetes therapy. Continuous glucose monitors (CGMs) and automated insulin delivery systems can improve glycemic control, decrease the rate of hypoglycemia, and enhance the quality of life of older patients. Most of the studies evaluating the use of automated insulin delivery systems in older patients have focused on those with type 1 diabetes and demonstrated improvement in glycemic control and/or reduced hypoglycemia. The DIAMOND trial demonstrated improved A1c and reduced glycemic variability with the use of CGM in adults older than 60 years with either type 1 or type 2 diabetes on multiple daily injections. Bluetooth-enabled “smart” insulin pens, which record the time and dose of insulin administrations, can also be a great asset in caring for older patients, especially those with cognitive impairment. With better insurance coverage, diabetes technologies may become more accessible and an asset in treating older patients with T2D.
In conclusion, management of T2D in older adults requires an individualized approach because of the heterogeneity in their health and functional status. Because cardiovascular disease is the leading cause of mortality in older patients with T2D, treatment plans should also address frequently coexisting cardiovascular risk factors, such as hypertension and hyperlipidemia. Clinicians should consider patients’ overall health, comorbidities, cognitive and functional status, social support systems, preferences, and life expectancy when developing individualized therapeutic plans.
Dr. Gunawan is an assistant professor in the department of internal medicine at UT Southwestern Medical Center, Dallas. She reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Smoking cessation has many benefits in diabetes
MONTPELLIER, FRANCE – The first expert consensus on smoking and diabetes, coauthored by the Francophone Diabetes Society (SFD) and the French Society for the Study of Nicotine Addiction (SFT), was presented at the SFD’s annual conference.
Alexia Rouland, MD, an endocrinologist at Dijon Bourgogne University Hospital, Dijon, France, took the conference as an opportunity to list the many benefits of smoking cessation for patients with diabetes, despite the “slight and temporary” risk for blood sugar imbalance.
Societies target smoking
Diabetes societies around Europe have set their sights on the topic of smoking. Indeed, the guidelines published in 2019 by the European Association for the Study of Diabetes and the European Society of Cardiology state that “smoking cessation is obligatory for all prediabetic and diabetic patients” (class I, level A).
This year, the France-based SFD and SFT dedicated an expert consensus to the major problem of smoking in patients with diabetes. The aim was to provide health care professionals with convincing, well-supported arguments in favor of smoking cessation in their patients with type 1 and type 2 diabetes.
“Before anything else, diabetic patients need to be made aware of the risks of smoking,” said Dr. Rouland. “It’s not just about the fear factor, though. It’s also about providing a positive incentive – they need to be told about the ways they’ll benefit from quitting smoking. For example, you have all-cause mortality, macro- and microangiopathic complications, and so on.”
Duration of abstinence
“Diabetic patients who have stopped smoking have a relative risk for all-cause mortality of 1.28 (1.09-1.51), which is less than what you see in active smokers (relative risk = 1.58; 1.42-1.77), but still above that of nonsmokers,” said Dr. Rouland.
A previous study revealed that although the risk does indeed go down after stopping smoking, it is linked to how long ago the person stopped. Patients who stopped smoking less than 10 years ago still had a slightly raised all-cause mortality risk, and this was even higher if they had smoked for 20 years or more.
After 10 years of not smoking, however, the greater all-cause mortality risk was no longer significant in any of the groups monitored (smoking duration, number of cigarettes/day). Concrete evidence of the link between all-cause mortality and the length of time since a person stopped smoking also emerged from the large cohort in the American Nurses’ Health Study.
The relative risk for all-cause mortality in women who stopped smoking less than 5 years ago remained high (RR = 1.96, 1.47-2.67), then decreased over time. After 10 years, it was no longer significant (RR = 1.11, 0.92-1.35).
Macro- and microangiopathic risks
Smoking cessation also has a real benefit in terms of the increased macro- and microangiopathic risks. In type 2 diabetes, a study found an increased relative risk for macro- and microalbuminuria of 1.86 (95% confidence interval, 1.37-2.52) in former smokers, compared with an increased relative risk of 2.61 (95% CI, 1.86-2.64) in current smokers.
In type 1 diabetes, the cumulative risk for microalbuminuria in former smokers was 15.1% vs. 18.9% in smokers and 10% in nonsmokers.
A 2019 meta-analysis of prospective cohort studies determined that smoking is an independent risk factor for diabetic nephropathy, especially in patients with type 1 diabetes.
Yet, most of the data for this condition come from subjects with type 2 diabetes. One publication estimated its prevalence after a 1-year follow-up of the smoking cessation program as 10.9% in former smokers and 15% in those who continued smoking.
In regard to macroangiopathy in the context of type 2 diabetes, the aforementioned 2019 meta-analysis focused on coronary artery disease, cerebrovascular accident (CVA), cardiovascular mortality, and myocardial infarction (MI). It found that smokers face an increased risk for all these outcomes.
The relative risks wavered between 1.53 and 1.66 and decreased after smoking cessation. For coronary artery disease and MI, they became insignificant. There was still a risk for CVA (RR = 1.34; 1.07-1.67) and fatal cardiovascular events (RR = 1.19; 0.02-1.39).
The data are slightly more heterogeneous for type 1 diabetes, where, despite smoking cessation, the increased risk for heart failure and CVA persists in men, yet the same risk for coronary heart disease and CVA drops in women.
Risk for weight gain
Dr. Rouland tried to reassure patients about the risk for gaining weight. “Weight gain is not inevitable. There is a risk for this, but it’s temporary. And, even with some weight gain, the cardiovascular benefits are still indisputable.”
A study carried out in 2013 focused specifically on this point, with an average post-cessation weight gain of 3.8 kg (8.4 lb) seen in diabetic individuals in the first 4 years after stopping smoking and of 0.1 kg (0.2 lb) thereafter. A time-based effect was observed with regulation of excess weight post-cessation over time, as seen in the general population (3 kg [6.6 lb] on average in nondiabetic individuals).
Weight gain tends to occur mainly in the immediate post-cessation period, essentially in the first 3 months, and there is a large variation in weight change. Some people gain a lot (from 5 to 10 kg [11 to 22 lb], or even more than 10 kg); others lose weight (20% of diabetic former smokers in the first month, 7% after 12 months), and 25% gain less than 5 kg (11 lb).
Blood glucose imbalance
“A risk for blood glucose imbalance has been reported after smoking cessation, although this is very slight and only temporary,” said Dr. Rouland.
A British retrospective study examined this question, focusing on glycated hemoglobin in patients with type 2 diabetes. Hemoglobin A1c increased by 0.21% (95% CI, 0.17-0.25; P < .001) within the first year after quitting. A1c decreased as abstinence continued and became comparable to that of continual smokers after 3 years. This increase in A1c was not mediated by weight change.
Another study published in 2018 on the topic of type 2 diabetes also reported on the risk for poor glycemic control (defined as A1c > 7%) persisting for 10 years after smoking cessation (odds ratio, 1.23; 95% CI, 1.06-1.42). Thereafter, between 10 and 19 years post-cessation, the OR decreased to 0.97 (95% CI, 0.80-1.19, NS). Beyond 20 years post-cessation, the OR was 1.14 (95% CI, 0.89-1.44, NS) and was therefore no longer significant.
Regardless, “the risk for poor glycemic control is lower in quitters than in active smokers,” said Dr. Rouland.
Quitting and diabetes risk
Will a smoker’s increased risk for diabetes drop when he or she stops smoking? “This is essentially what happens,” Dr. Rouland confirmed, “and his or her increased risk for metabolic syndrome also drops. One meta-analysis revealed a time-based effect.
“Patients who had stopped smoking less than 5 years previously had an increased relative risk for type 2 diabetes, and this risk dropped to 1.11 after more than 10 years of not smoking. Moreover, this relative risk for type 2 diabetes remained lower than that of active smokers, at between 1.19 and 1.60, depending on tobacco use.”
In regard to the risk for metabolic syndrome, those who quit smoking seem to have an increased risk of 10%, compared with nonsmokers (RR = 1.10, 1.08-1.11; P < .001). “But yet again, this increased risk is much lower than that of active smokers, whose risk is between 37% (less than 20 cigarettes/day) and 71% (more than 20 cigarettes/day).”
Women with diabetes
“The benefits of quitting appear identical, regardless of the sex of the diabetic person,” said Dr. Rouland. “As in the general population, weight gain after smoking cessation is greater in women. Furthermore, while smoking increases the risk for gestational diabetes (RR, 1.4-1.9) and for the use of insulin in this context, stopping smoking reduces these risks.
“Moreover, smoking during pregnancy not only increases the risk for pregnancy-related complications (early miscarriage, ectopic pregnancy, birth defects, placental abruption, premature birth, intrauterine fetal demise, cesarean birth, low birth weight), but it also increases the risk of type 2 diabetes in the newborn. The risk to the newborn is said to be around 34% in cases in which the mother smokes during pregnancy and 22% in cases in which the mother is a passive smoker, thereby justifying the use of measures to help the mother’s family members to stop smoking.”
Dr. Rouland reports no relevant financial relationships.
This article was translated from the Medscape French edition. A version appeared on Medscape.com.
MONTPELLIER, FRANCE – The first expert consensus on smoking and diabetes, coauthored by the Francophone Diabetes Society (SFD) and the French Society for the Study of Nicotine Addiction (SFT), was presented at the SFD’s annual conference.
Alexia Rouland, MD, an endocrinologist at Dijon Bourgogne University Hospital, Dijon, France, took the conference as an opportunity to list the many benefits of smoking cessation for patients with diabetes, despite the “slight and temporary” risk for blood sugar imbalance.
Societies target smoking
Diabetes societies around Europe have set their sights on the topic of smoking. Indeed, the guidelines published in 2019 by the European Association for the Study of Diabetes and the European Society of Cardiology state that “smoking cessation is obligatory for all prediabetic and diabetic patients” (class I, level A).
This year, the France-based SFD and SFT dedicated an expert consensus to the major problem of smoking in patients with diabetes. The aim was to provide health care professionals with convincing, well-supported arguments in favor of smoking cessation in their patients with type 1 and type 2 diabetes.
“Before anything else, diabetic patients need to be made aware of the risks of smoking,” said Dr. Rouland. “It’s not just about the fear factor, though. It’s also about providing a positive incentive – they need to be told about the ways they’ll benefit from quitting smoking. For example, you have all-cause mortality, macro- and microangiopathic complications, and so on.”
Duration of abstinence
“Diabetic patients who have stopped smoking have a relative risk for all-cause mortality of 1.28 (1.09-1.51), which is less than what you see in active smokers (relative risk = 1.58; 1.42-1.77), but still above that of nonsmokers,” said Dr. Rouland.
A previous study revealed that although the risk does indeed go down after stopping smoking, it is linked to how long ago the person stopped. Patients who stopped smoking less than 10 years ago still had a slightly raised all-cause mortality risk, and this was even higher if they had smoked for 20 years or more.
After 10 years of not smoking, however, the greater all-cause mortality risk was no longer significant in any of the groups monitored (smoking duration, number of cigarettes/day). Concrete evidence of the link between all-cause mortality and the length of time since a person stopped smoking also emerged from the large cohort in the American Nurses’ Health Study.
The relative risk for all-cause mortality in women who stopped smoking less than 5 years ago remained high (RR = 1.96, 1.47-2.67), then decreased over time. After 10 years, it was no longer significant (RR = 1.11, 0.92-1.35).
Macro- and microangiopathic risks
Smoking cessation also has a real benefit in terms of the increased macro- and microangiopathic risks. In type 2 diabetes, a study found an increased relative risk for macro- and microalbuminuria of 1.86 (95% confidence interval, 1.37-2.52) in former smokers, compared with an increased relative risk of 2.61 (95% CI, 1.86-2.64) in current smokers.
In type 1 diabetes, the cumulative risk for microalbuminuria in former smokers was 15.1% vs. 18.9% in smokers and 10% in nonsmokers.
A 2019 meta-analysis of prospective cohort studies determined that smoking is an independent risk factor for diabetic nephropathy, especially in patients with type 1 diabetes.
Yet, most of the data for this condition come from subjects with type 2 diabetes. One publication estimated its prevalence after a 1-year follow-up of the smoking cessation program as 10.9% in former smokers and 15% in those who continued smoking.
In regard to macroangiopathy in the context of type 2 diabetes, the aforementioned 2019 meta-analysis focused on coronary artery disease, cerebrovascular accident (CVA), cardiovascular mortality, and myocardial infarction (MI). It found that smokers face an increased risk for all these outcomes.
The relative risks wavered between 1.53 and 1.66 and decreased after smoking cessation. For coronary artery disease and MI, they became insignificant. There was still a risk for CVA (RR = 1.34; 1.07-1.67) and fatal cardiovascular events (RR = 1.19; 0.02-1.39).
The data are slightly more heterogeneous for type 1 diabetes, where, despite smoking cessation, the increased risk for heart failure and CVA persists in men, yet the same risk for coronary heart disease and CVA drops in women.
Risk for weight gain
Dr. Rouland tried to reassure patients about the risk for gaining weight. “Weight gain is not inevitable. There is a risk for this, but it’s temporary. And, even with some weight gain, the cardiovascular benefits are still indisputable.”
A study carried out in 2013 focused specifically on this point, with an average post-cessation weight gain of 3.8 kg (8.4 lb) seen in diabetic individuals in the first 4 years after stopping smoking and of 0.1 kg (0.2 lb) thereafter. A time-based effect was observed with regulation of excess weight post-cessation over time, as seen in the general population (3 kg [6.6 lb] on average in nondiabetic individuals).
Weight gain tends to occur mainly in the immediate post-cessation period, essentially in the first 3 months, and there is a large variation in weight change. Some people gain a lot (from 5 to 10 kg [11 to 22 lb], or even more than 10 kg); others lose weight (20% of diabetic former smokers in the first month, 7% after 12 months), and 25% gain less than 5 kg (11 lb).
Blood glucose imbalance
“A risk for blood glucose imbalance has been reported after smoking cessation, although this is very slight and only temporary,” said Dr. Rouland.
A British retrospective study examined this question, focusing on glycated hemoglobin in patients with type 2 diabetes. Hemoglobin A1c increased by 0.21% (95% CI, 0.17-0.25; P < .001) within the first year after quitting. A1c decreased as abstinence continued and became comparable to that of continual smokers after 3 years. This increase in A1c was not mediated by weight change.
Another study published in 2018 on the topic of type 2 diabetes also reported on the risk for poor glycemic control (defined as A1c > 7%) persisting for 10 years after smoking cessation (odds ratio, 1.23; 95% CI, 1.06-1.42). Thereafter, between 10 and 19 years post-cessation, the OR decreased to 0.97 (95% CI, 0.80-1.19, NS). Beyond 20 years post-cessation, the OR was 1.14 (95% CI, 0.89-1.44, NS) and was therefore no longer significant.
Regardless, “the risk for poor glycemic control is lower in quitters than in active smokers,” said Dr. Rouland.
Quitting and diabetes risk
Will a smoker’s increased risk for diabetes drop when he or she stops smoking? “This is essentially what happens,” Dr. Rouland confirmed, “and his or her increased risk for metabolic syndrome also drops. One meta-analysis revealed a time-based effect.
“Patients who had stopped smoking less than 5 years previously had an increased relative risk for type 2 diabetes, and this risk dropped to 1.11 after more than 10 years of not smoking. Moreover, this relative risk for type 2 diabetes remained lower than that of active smokers, at between 1.19 and 1.60, depending on tobacco use.”
In regard to the risk for metabolic syndrome, those who quit smoking seem to have an increased risk of 10%, compared with nonsmokers (RR = 1.10, 1.08-1.11; P < .001). “But yet again, this increased risk is much lower than that of active smokers, whose risk is between 37% (less than 20 cigarettes/day) and 71% (more than 20 cigarettes/day).”
Women with diabetes
“The benefits of quitting appear identical, regardless of the sex of the diabetic person,” said Dr. Rouland. “As in the general population, weight gain after smoking cessation is greater in women. Furthermore, while smoking increases the risk for gestational diabetes (RR, 1.4-1.9) and for the use of insulin in this context, stopping smoking reduces these risks.
“Moreover, smoking during pregnancy not only increases the risk for pregnancy-related complications (early miscarriage, ectopic pregnancy, birth defects, placental abruption, premature birth, intrauterine fetal demise, cesarean birth, low birth weight), but it also increases the risk of type 2 diabetes in the newborn. The risk to the newborn is said to be around 34% in cases in which the mother smokes during pregnancy and 22% in cases in which the mother is a passive smoker, thereby justifying the use of measures to help the mother’s family members to stop smoking.”
Dr. Rouland reports no relevant financial relationships.
This article was translated from the Medscape French edition. A version appeared on Medscape.com.
MONTPELLIER, FRANCE – The first expert consensus on smoking and diabetes, coauthored by the Francophone Diabetes Society (SFD) and the French Society for the Study of Nicotine Addiction (SFT), was presented at the SFD’s annual conference.
Alexia Rouland, MD, an endocrinologist at Dijon Bourgogne University Hospital, Dijon, France, took the conference as an opportunity to list the many benefits of smoking cessation for patients with diabetes, despite the “slight and temporary” risk for blood sugar imbalance.
Societies target smoking
Diabetes societies around Europe have set their sights on the topic of smoking. Indeed, the guidelines published in 2019 by the European Association for the Study of Diabetes and the European Society of Cardiology state that “smoking cessation is obligatory for all prediabetic and diabetic patients” (class I, level A).
This year, the France-based SFD and SFT dedicated an expert consensus to the major problem of smoking in patients with diabetes. The aim was to provide health care professionals with convincing, well-supported arguments in favor of smoking cessation in their patients with type 1 and type 2 diabetes.
“Before anything else, diabetic patients need to be made aware of the risks of smoking,” said Dr. Rouland. “It’s not just about the fear factor, though. It’s also about providing a positive incentive – they need to be told about the ways they’ll benefit from quitting smoking. For example, you have all-cause mortality, macro- and microangiopathic complications, and so on.”
Duration of abstinence
“Diabetic patients who have stopped smoking have a relative risk for all-cause mortality of 1.28 (1.09-1.51), which is less than what you see in active smokers (relative risk = 1.58; 1.42-1.77), but still above that of nonsmokers,” said Dr. Rouland.
A previous study revealed that although the risk does indeed go down after stopping smoking, it is linked to how long ago the person stopped. Patients who stopped smoking less than 10 years ago still had a slightly raised all-cause mortality risk, and this was even higher if they had smoked for 20 years or more.
After 10 years of not smoking, however, the greater all-cause mortality risk was no longer significant in any of the groups monitored (smoking duration, number of cigarettes/day). Concrete evidence of the link between all-cause mortality and the length of time since a person stopped smoking also emerged from the large cohort in the American Nurses’ Health Study.
The relative risk for all-cause mortality in women who stopped smoking less than 5 years ago remained high (RR = 1.96, 1.47-2.67), then decreased over time. After 10 years, it was no longer significant (RR = 1.11, 0.92-1.35).
Macro- and microangiopathic risks
Smoking cessation also has a real benefit in terms of the increased macro- and microangiopathic risks. In type 2 diabetes, a study found an increased relative risk for macro- and microalbuminuria of 1.86 (95% confidence interval, 1.37-2.52) in former smokers, compared with an increased relative risk of 2.61 (95% CI, 1.86-2.64) in current smokers.
In type 1 diabetes, the cumulative risk for microalbuminuria in former smokers was 15.1% vs. 18.9% in smokers and 10% in nonsmokers.
A 2019 meta-analysis of prospective cohort studies determined that smoking is an independent risk factor for diabetic nephropathy, especially in patients with type 1 diabetes.
Yet, most of the data for this condition come from subjects with type 2 diabetes. One publication estimated its prevalence after a 1-year follow-up of the smoking cessation program as 10.9% in former smokers and 15% in those who continued smoking.
In regard to macroangiopathy in the context of type 2 diabetes, the aforementioned 2019 meta-analysis focused on coronary artery disease, cerebrovascular accident (CVA), cardiovascular mortality, and myocardial infarction (MI). It found that smokers face an increased risk for all these outcomes.
The relative risks wavered between 1.53 and 1.66 and decreased after smoking cessation. For coronary artery disease and MI, they became insignificant. There was still a risk for CVA (RR = 1.34; 1.07-1.67) and fatal cardiovascular events (RR = 1.19; 0.02-1.39).
The data are slightly more heterogeneous for type 1 diabetes, where, despite smoking cessation, the increased risk for heart failure and CVA persists in men, yet the same risk for coronary heart disease and CVA drops in women.
Risk for weight gain
Dr. Rouland tried to reassure patients about the risk for gaining weight. “Weight gain is not inevitable. There is a risk for this, but it’s temporary. And, even with some weight gain, the cardiovascular benefits are still indisputable.”
A study carried out in 2013 focused specifically on this point, with an average post-cessation weight gain of 3.8 kg (8.4 lb) seen in diabetic individuals in the first 4 years after stopping smoking and of 0.1 kg (0.2 lb) thereafter. A time-based effect was observed with regulation of excess weight post-cessation over time, as seen in the general population (3 kg [6.6 lb] on average in nondiabetic individuals).
Weight gain tends to occur mainly in the immediate post-cessation period, essentially in the first 3 months, and there is a large variation in weight change. Some people gain a lot (from 5 to 10 kg [11 to 22 lb], or even more than 10 kg); others lose weight (20% of diabetic former smokers in the first month, 7% after 12 months), and 25% gain less than 5 kg (11 lb).
Blood glucose imbalance
“A risk for blood glucose imbalance has been reported after smoking cessation, although this is very slight and only temporary,” said Dr. Rouland.
A British retrospective study examined this question, focusing on glycated hemoglobin in patients with type 2 diabetes. Hemoglobin A1c increased by 0.21% (95% CI, 0.17-0.25; P < .001) within the first year after quitting. A1c decreased as abstinence continued and became comparable to that of continual smokers after 3 years. This increase in A1c was not mediated by weight change.
Another study published in 2018 on the topic of type 2 diabetes also reported on the risk for poor glycemic control (defined as A1c > 7%) persisting for 10 years after smoking cessation (odds ratio, 1.23; 95% CI, 1.06-1.42). Thereafter, between 10 and 19 years post-cessation, the OR decreased to 0.97 (95% CI, 0.80-1.19, NS). Beyond 20 years post-cessation, the OR was 1.14 (95% CI, 0.89-1.44, NS) and was therefore no longer significant.
Regardless, “the risk for poor glycemic control is lower in quitters than in active smokers,” said Dr. Rouland.
Quitting and diabetes risk
Will a smoker’s increased risk for diabetes drop when he or she stops smoking? “This is essentially what happens,” Dr. Rouland confirmed, “and his or her increased risk for metabolic syndrome also drops. One meta-analysis revealed a time-based effect.
“Patients who had stopped smoking less than 5 years previously had an increased relative risk for type 2 diabetes, and this risk dropped to 1.11 after more than 10 years of not smoking. Moreover, this relative risk for type 2 diabetes remained lower than that of active smokers, at between 1.19 and 1.60, depending on tobacco use.”
In regard to the risk for metabolic syndrome, those who quit smoking seem to have an increased risk of 10%, compared with nonsmokers (RR = 1.10, 1.08-1.11; P < .001). “But yet again, this increased risk is much lower than that of active smokers, whose risk is between 37% (less than 20 cigarettes/day) and 71% (more than 20 cigarettes/day).”
Women with diabetes
“The benefits of quitting appear identical, regardless of the sex of the diabetic person,” said Dr. Rouland. “As in the general population, weight gain after smoking cessation is greater in women. Furthermore, while smoking increases the risk for gestational diabetes (RR, 1.4-1.9) and for the use of insulin in this context, stopping smoking reduces these risks.
“Moreover, smoking during pregnancy not only increases the risk for pregnancy-related complications (early miscarriage, ectopic pregnancy, birth defects, placental abruption, premature birth, intrauterine fetal demise, cesarean birth, low birth weight), but it also increases the risk of type 2 diabetes in the newborn. The risk to the newborn is said to be around 34% in cases in which the mother smokes during pregnancy and 22% in cases in which the mother is a passive smoker, thereby justifying the use of measures to help the mother’s family members to stop smoking.”
Dr. Rouland reports no relevant financial relationships.
This article was translated from the Medscape French edition. A version appeared on Medscape.com.
Should youth with type 1 diabetes use closed-loop systems?
Would closed-loop systems be a good option for young patients with type 1 diabetes?
International and French recommendations on closed-loop systems state that the use of an “artificial pancreas” should be reserved for adults who are fully engaged with their treatment. This means that young patients, especially adolescents, who are less likely to comply with treatment and are more likely to experience suboptimal blood glucose control, are often excluded from the use of such systems for managing their diabetes.
Several recent studies seem to call this approach into question.
One such study, which was presented at a Francophone Diabetes Society conference and was published in Nature Communications, showed that adolescents with poorly controlled diabetes who were equipped with closed-loop systems gained IQ points and reasoning capacity and experienced a reduction in edematous tissue in the brain cortex. Furthermore, with the closed-loop system, patients spent 13% more time in a target range, and there was a significant reduction in time spent in hyperglycemia.
In the same vein, a small prospective study published in Diabetes Care showed that the closed-loop system with the Minimed 780G pump improved glycemic control for 20 young patients with type 1 diabetes aged 13-25 years whose diabetes was poorly controlled (hemoglobin A1c ≥ 8.5%). At the end of the 3-month study period, the average A1c had decreased from 10.5% (±2.1%) to 7.6% (±1.1%), an average decrease of 2.9%. The time spent in target A1c, which was set from 0.70 g/L to 1.80 g/L, was increased by almost 40%.
With respect to very young children, a study published in The New England Journal of Medicine also showed a favorable risk-benefit ratio for closed-loop systems. The trial, which enrolled 102 children aged 2 years to less than 6 years who had type 1 diabetes, showed that the amount of time that the glucose level was within the target range during the 13-week study period was higher (+3 hours) for those who had been randomly assigned to receive the hybrid closed-loop system (n = 68) than for those who had received the standard treatment (n = 34), either with an insulin pump or multiple daily injections or a Dexcom G6 continuous glucose monitoring device.
A previous study carried out by the Paris Public Hospital System had already shown that the French Diabeloop system could reduce episodes of hypoglycemia and achieve good glycemic control for prepubescent children (n = 21; aged 6-12 years) with type 1 diabetes in real-life conditions.
Eric Renard, MD, PhD, head of the department of endocrinology and diabetes at Lapeyronie Hospital in Montpellier, France, was not surprised at the findings from the study, especially in adolescents with poorly controlled diabetes.
“We have already seen studies in which those patients who had the most poorly controlled diabetes at the start were the ones who improved the most with the closed-loop system, by at least 20% in terms of time in target. These findings resonate with what I see in my clinic,” said Dr. Renard in an interview.
“In my experience, these young adolescents, who neglected their diabetes when they had no devices to help control it, when they had to inject themselves, et cetera ... well, they’re just not the same people when they’re put on a closed-loop system,” he added. “They rise to the challenge, and for the first time, they succeed without making a huge effort, since the algorithm does what they weren’t doing. It’s astonishing to see near-total engagement in these young people when explaining the technology to them and saying, ‘Let’s give it a go.’ These are the very same youngsters who didn’t want to hear about their diabetes in the past. They are delighted and once again involved in managing their condition.”
That’s why Dr. Renard recommends keeping an open mind when considering treatment options for young patients with poorly controlled type 1 diabetes.
“When young people have very poorly controlled diabetes, they risk having cardiovascular complications and damaging their retinas and kidneys,” he said. “If we can get them from 25% to 45% time in target, even if that hasn’t been easy to achieve, this will help save their blood vessels! The only thing we have to be careful of is that we don’t set up a closed-loop system in someone who doesn’t want one. But, if it can manage to spark the interest of a young patient, in most cases, it’s beneficial.”
This article was translated from the Medscape French edition. A version appeared on Medscape.com.
Would closed-loop systems be a good option for young patients with type 1 diabetes?
International and French recommendations on closed-loop systems state that the use of an “artificial pancreas” should be reserved for adults who are fully engaged with their treatment. This means that young patients, especially adolescents, who are less likely to comply with treatment and are more likely to experience suboptimal blood glucose control, are often excluded from the use of such systems for managing their diabetes.
Several recent studies seem to call this approach into question.
One such study, which was presented at a Francophone Diabetes Society conference and was published in Nature Communications, showed that adolescents with poorly controlled diabetes who were equipped with closed-loop systems gained IQ points and reasoning capacity and experienced a reduction in edematous tissue in the brain cortex. Furthermore, with the closed-loop system, patients spent 13% more time in a target range, and there was a significant reduction in time spent in hyperglycemia.
In the same vein, a small prospective study published in Diabetes Care showed that the closed-loop system with the Minimed 780G pump improved glycemic control for 20 young patients with type 1 diabetes aged 13-25 years whose diabetes was poorly controlled (hemoglobin A1c ≥ 8.5%). At the end of the 3-month study period, the average A1c had decreased from 10.5% (±2.1%) to 7.6% (±1.1%), an average decrease of 2.9%. The time spent in target A1c, which was set from 0.70 g/L to 1.80 g/L, was increased by almost 40%.
With respect to very young children, a study published in The New England Journal of Medicine also showed a favorable risk-benefit ratio for closed-loop systems. The trial, which enrolled 102 children aged 2 years to less than 6 years who had type 1 diabetes, showed that the amount of time that the glucose level was within the target range during the 13-week study period was higher (+3 hours) for those who had been randomly assigned to receive the hybrid closed-loop system (n = 68) than for those who had received the standard treatment (n = 34), either with an insulin pump or multiple daily injections or a Dexcom G6 continuous glucose monitoring device.
A previous study carried out by the Paris Public Hospital System had already shown that the French Diabeloop system could reduce episodes of hypoglycemia and achieve good glycemic control for prepubescent children (n = 21; aged 6-12 years) with type 1 diabetes in real-life conditions.
Eric Renard, MD, PhD, head of the department of endocrinology and diabetes at Lapeyronie Hospital in Montpellier, France, was not surprised at the findings from the study, especially in adolescents with poorly controlled diabetes.
“We have already seen studies in which those patients who had the most poorly controlled diabetes at the start were the ones who improved the most with the closed-loop system, by at least 20% in terms of time in target. These findings resonate with what I see in my clinic,” said Dr. Renard in an interview.
“In my experience, these young adolescents, who neglected their diabetes when they had no devices to help control it, when they had to inject themselves, et cetera ... well, they’re just not the same people when they’re put on a closed-loop system,” he added. “They rise to the challenge, and for the first time, they succeed without making a huge effort, since the algorithm does what they weren’t doing. It’s astonishing to see near-total engagement in these young people when explaining the technology to them and saying, ‘Let’s give it a go.’ These are the very same youngsters who didn’t want to hear about their diabetes in the past. They are delighted and once again involved in managing their condition.”
That’s why Dr. Renard recommends keeping an open mind when considering treatment options for young patients with poorly controlled type 1 diabetes.
“When young people have very poorly controlled diabetes, they risk having cardiovascular complications and damaging their retinas and kidneys,” he said. “If we can get them from 25% to 45% time in target, even if that hasn’t been easy to achieve, this will help save their blood vessels! The only thing we have to be careful of is that we don’t set up a closed-loop system in someone who doesn’t want one. But, if it can manage to spark the interest of a young patient, in most cases, it’s beneficial.”
This article was translated from the Medscape French edition. A version appeared on Medscape.com.
Would closed-loop systems be a good option for young patients with type 1 diabetes?
International and French recommendations on closed-loop systems state that the use of an “artificial pancreas” should be reserved for adults who are fully engaged with their treatment. This means that young patients, especially adolescents, who are less likely to comply with treatment and are more likely to experience suboptimal blood glucose control, are often excluded from the use of such systems for managing their diabetes.
Several recent studies seem to call this approach into question.
One such study, which was presented at a Francophone Diabetes Society conference and was published in Nature Communications, showed that adolescents with poorly controlled diabetes who were equipped with closed-loop systems gained IQ points and reasoning capacity and experienced a reduction in edematous tissue in the brain cortex. Furthermore, with the closed-loop system, patients spent 13% more time in a target range, and there was a significant reduction in time spent in hyperglycemia.
In the same vein, a small prospective study published in Diabetes Care showed that the closed-loop system with the Minimed 780G pump improved glycemic control for 20 young patients with type 1 diabetes aged 13-25 years whose diabetes was poorly controlled (hemoglobin A1c ≥ 8.5%). At the end of the 3-month study period, the average A1c had decreased from 10.5% (±2.1%) to 7.6% (±1.1%), an average decrease of 2.9%. The time spent in target A1c, which was set from 0.70 g/L to 1.80 g/L, was increased by almost 40%.
With respect to very young children, a study published in The New England Journal of Medicine also showed a favorable risk-benefit ratio for closed-loop systems. The trial, which enrolled 102 children aged 2 years to less than 6 years who had type 1 diabetes, showed that the amount of time that the glucose level was within the target range during the 13-week study period was higher (+3 hours) for those who had been randomly assigned to receive the hybrid closed-loop system (n = 68) than for those who had received the standard treatment (n = 34), either with an insulin pump or multiple daily injections or a Dexcom G6 continuous glucose monitoring device.
A previous study carried out by the Paris Public Hospital System had already shown that the French Diabeloop system could reduce episodes of hypoglycemia and achieve good glycemic control for prepubescent children (n = 21; aged 6-12 years) with type 1 diabetes in real-life conditions.
Eric Renard, MD, PhD, head of the department of endocrinology and diabetes at Lapeyronie Hospital in Montpellier, France, was not surprised at the findings from the study, especially in adolescents with poorly controlled diabetes.
“We have already seen studies in which those patients who had the most poorly controlled diabetes at the start were the ones who improved the most with the closed-loop system, by at least 20% in terms of time in target. These findings resonate with what I see in my clinic,” said Dr. Renard in an interview.
“In my experience, these young adolescents, who neglected their diabetes when they had no devices to help control it, when they had to inject themselves, et cetera ... well, they’re just not the same people when they’re put on a closed-loop system,” he added. “They rise to the challenge, and for the first time, they succeed without making a huge effort, since the algorithm does what they weren’t doing. It’s astonishing to see near-total engagement in these young people when explaining the technology to them and saying, ‘Let’s give it a go.’ These are the very same youngsters who didn’t want to hear about their diabetes in the past. They are delighted and once again involved in managing their condition.”
That’s why Dr. Renard recommends keeping an open mind when considering treatment options for young patients with poorly controlled type 1 diabetes.
“When young people have very poorly controlled diabetes, they risk having cardiovascular complications and damaging their retinas and kidneys,” he said. “If we can get them from 25% to 45% time in target, even if that hasn’t been easy to achieve, this will help save their blood vessels! The only thing we have to be careful of is that we don’t set up a closed-loop system in someone who doesn’t want one. But, if it can manage to spark the interest of a young patient, in most cases, it’s beneficial.”
This article was translated from the Medscape French edition. A version appeared on Medscape.com.
Obesity drugs overpriced, change needed to tackle issue
The lowest available national prices of drugs to treat obesity are up to 20 times higher than the estimated cost of profitable generic versions of the same agents, according to a new analysis.
The findings by Jacob Levi, MBBS, and colleagues were published in Obesity.
“Our study highlights the inequality in pricing that exists for effective antiobesity medications, which are largely unaffordable in most countries,” Dr. Levi, from Royal Free Hospital NHS Trust, London, said in a press release.
“We show that these drugs can actually be produced and sold profitably for low prices,” he summarized. “A public health approach that prioritizes improving access to medications should be adopted, instead of allowing companies to maximize profits,” Dr. Levi urged.
Dr. Levi and colleagues studied the oral agents orlistat, naltrexone/bupropion, topiramate/phentermine, and semaglutide, and subcutaneous liraglutide, semaglutide, and tirzepatide (all approved by the U.S. Food and Drug Administration to treat obesity, except for oral semaglutide and subcutaneous tirzepatide, which are not yet approved to treat obesity in the absence of type 2 diabetes).
“Worldwide, more people are dying from diabetes and clinical obesity than HIV, tuberculosis, and malaria combined now,” senior author Andrew Hill, MD, department of pharmacology and therapeutics, University of Liverpool, England, pointed out.
We need to repeat the low-cost success story with obesity drugs
“Millions of lives have been saved by treating infectious diseases at low cost in poor countries,” Dr. Hill continued. “Now we need to repeat this medical success story, with mass treatment of diabetes and clinical obesity at low prices.”
However, in an accompanying editorial, Eric A. Finkelstein, MD, and Junxing Chay, PhD, Duke-NUS Medical School, Singapore, maintain that “It would be great if everyone had affordable access to all medicines that might improve their health. Yet that is simply not possible, nor will it ever be.”
“What is truly needed is a better way to ration the health care dollars currently available in efforts to maximize population health. That is the challenge ahead not just for [antiobesity medications] but for all treatments,” they say.
“Greater use of cost-effectiveness analysis and direct negotiations, while maintaining the patent system, represents an appropriate approach for allocating scarce health care resources in the United States and beyond,” they continue.
Lowest current patented drug prices vs. estimated generic drug prices
New medications for obesity were highly effective in recent clinical trials, but high prices limit the ability of patients to get these medications, Dr. Levi and colleagues write.
They analyzed prices for obesity drugs in 16 low-, middle-, and high-income countries: Australia, Bangladesh, China, France, Germany, India, Kenya, Morocco, Norway, Peru, Pakistan, South Africa, Turkey, the United Kingdom, the United States, and Vietnam.
The researchers assessed the price of a 30-day supply of each of the studied branded drugs based on the lowest available price (in 2021 U.S. dollars) from multiple online national price databases.
Then they calculated the estimated minimum price of a 30-day supply of a potential generic version of these drugs, which included the cost of the active medicinal ingredients, the excipients (nonactive ingredients), the prefilled injectable device plus needles (for subcutaneous drugs), transportation, 10% profit, and 27% tax on profit.
The national prices of the branded medications for obesity were significantly higher than the estimated minimum prices of potential generic drugs (see Table).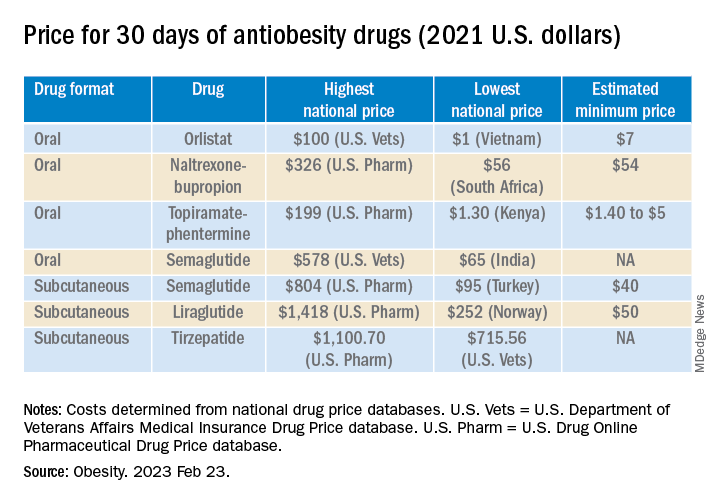
The highest national price for a branded oral drug for obesity vs. the estimated minimum price for a potential generic version was $100 vs. $7 for orlistat, $199 vs. $5 for phentermine/topiramate, and $326 vs. $54 for naltrexone/bupropion, for a 30-day supply.
There was an even greater difference between highest national branded drug price vs. estimated minimum generic drug price for the newer subcutaneously injectable drugs for obesity.
For example, the price of a 30-day course of subcutaneous semaglutide ranged from $804 (United States) to $95 (Turkey), while the estimated minimum potential generic drug price was $40 (which is 20 times lower).
The study was funded by grants from the Make Medicines Affordable/International Treatment Preparedness Coalition and from the National Heart, Lung, and Blood Institute of the National Institutes of Health. Coauthor Francois Venter has reported receiving support from the Bill and Melinda Gates Foundation, U.S. Agency for International Development, Unitaid, SA Medical Research Council, Foundation for Innovative New Diagnostics, the Children’s Investment Fund Foundation, Gilead, ViiV, Mylan, Merck, Adcock Ingram, Aspen, Abbott, Roche, Johnson & Johnson, Sanofi, Virology Education, SA HIV Clinicians Society, and Dira Sengwe. The other authors and Dr. Chay have reported no relevant financial relationships. Dr. Finkelstein has reported receiving support for serving on the WW scientific advisory board and an educational grant unrelated to the present work from Novo Nordisk.
A version of this article first appeared on Medscape.com.
The lowest available national prices of drugs to treat obesity are up to 20 times higher than the estimated cost of profitable generic versions of the same agents, according to a new analysis.
The findings by Jacob Levi, MBBS, and colleagues were published in Obesity.
“Our study highlights the inequality in pricing that exists for effective antiobesity medications, which are largely unaffordable in most countries,” Dr. Levi, from Royal Free Hospital NHS Trust, London, said in a press release.
“We show that these drugs can actually be produced and sold profitably for low prices,” he summarized. “A public health approach that prioritizes improving access to medications should be adopted, instead of allowing companies to maximize profits,” Dr. Levi urged.
Dr. Levi and colleagues studied the oral agents orlistat, naltrexone/bupropion, topiramate/phentermine, and semaglutide, and subcutaneous liraglutide, semaglutide, and tirzepatide (all approved by the U.S. Food and Drug Administration to treat obesity, except for oral semaglutide and subcutaneous tirzepatide, which are not yet approved to treat obesity in the absence of type 2 diabetes).
“Worldwide, more people are dying from diabetes and clinical obesity than HIV, tuberculosis, and malaria combined now,” senior author Andrew Hill, MD, department of pharmacology and therapeutics, University of Liverpool, England, pointed out.
We need to repeat the low-cost success story with obesity drugs
“Millions of lives have been saved by treating infectious diseases at low cost in poor countries,” Dr. Hill continued. “Now we need to repeat this medical success story, with mass treatment of diabetes and clinical obesity at low prices.”
However, in an accompanying editorial, Eric A. Finkelstein, MD, and Junxing Chay, PhD, Duke-NUS Medical School, Singapore, maintain that “It would be great if everyone had affordable access to all medicines that might improve their health. Yet that is simply not possible, nor will it ever be.”
“What is truly needed is a better way to ration the health care dollars currently available in efforts to maximize population health. That is the challenge ahead not just for [antiobesity medications] but for all treatments,” they say.
“Greater use of cost-effectiveness analysis and direct negotiations, while maintaining the patent system, represents an appropriate approach for allocating scarce health care resources in the United States and beyond,” they continue.
Lowest current patented drug prices vs. estimated generic drug prices
New medications for obesity were highly effective in recent clinical trials, but high prices limit the ability of patients to get these medications, Dr. Levi and colleagues write.
They analyzed prices for obesity drugs in 16 low-, middle-, and high-income countries: Australia, Bangladesh, China, France, Germany, India, Kenya, Morocco, Norway, Peru, Pakistan, South Africa, Turkey, the United Kingdom, the United States, and Vietnam.
The researchers assessed the price of a 30-day supply of each of the studied branded drugs based on the lowest available price (in 2021 U.S. dollars) from multiple online national price databases.
Then they calculated the estimated minimum price of a 30-day supply of a potential generic version of these drugs, which included the cost of the active medicinal ingredients, the excipients (nonactive ingredients), the prefilled injectable device plus needles (for subcutaneous drugs), transportation, 10% profit, and 27% tax on profit.
The national prices of the branded medications for obesity were significantly higher than the estimated minimum prices of potential generic drugs (see Table).
The highest national price for a branded oral drug for obesity vs. the estimated minimum price for a potential generic version was $100 vs. $7 for orlistat, $199 vs. $5 for phentermine/topiramate, and $326 vs. $54 for naltrexone/bupropion, for a 30-day supply.
There was an even greater difference between highest national branded drug price vs. estimated minimum generic drug price for the newer subcutaneously injectable drugs for obesity.
For example, the price of a 30-day course of subcutaneous semaglutide ranged from $804 (United States) to $95 (Turkey), while the estimated minimum potential generic drug price was $40 (which is 20 times lower).
The study was funded by grants from the Make Medicines Affordable/International Treatment Preparedness Coalition and from the National Heart, Lung, and Blood Institute of the National Institutes of Health. Coauthor Francois Venter has reported receiving support from the Bill and Melinda Gates Foundation, U.S. Agency for International Development, Unitaid, SA Medical Research Council, Foundation for Innovative New Diagnostics, the Children’s Investment Fund Foundation, Gilead, ViiV, Mylan, Merck, Adcock Ingram, Aspen, Abbott, Roche, Johnson & Johnson, Sanofi, Virology Education, SA HIV Clinicians Society, and Dira Sengwe. The other authors and Dr. Chay have reported no relevant financial relationships. Dr. Finkelstein has reported receiving support for serving on the WW scientific advisory board and an educational grant unrelated to the present work from Novo Nordisk.
A version of this article first appeared on Medscape.com.
The lowest available national prices of drugs to treat obesity are up to 20 times higher than the estimated cost of profitable generic versions of the same agents, according to a new analysis.
The findings by Jacob Levi, MBBS, and colleagues were published in Obesity.
“Our study highlights the inequality in pricing that exists for effective antiobesity medications, which are largely unaffordable in most countries,” Dr. Levi, from Royal Free Hospital NHS Trust, London, said in a press release.
“We show that these drugs can actually be produced and sold profitably for low prices,” he summarized. “A public health approach that prioritizes improving access to medications should be adopted, instead of allowing companies to maximize profits,” Dr. Levi urged.
Dr. Levi and colleagues studied the oral agents orlistat, naltrexone/bupropion, topiramate/phentermine, and semaglutide, and subcutaneous liraglutide, semaglutide, and tirzepatide (all approved by the U.S. Food and Drug Administration to treat obesity, except for oral semaglutide and subcutaneous tirzepatide, which are not yet approved to treat obesity in the absence of type 2 diabetes).
“Worldwide, more people are dying from diabetes and clinical obesity than HIV, tuberculosis, and malaria combined now,” senior author Andrew Hill, MD, department of pharmacology and therapeutics, University of Liverpool, England, pointed out.
We need to repeat the low-cost success story with obesity drugs
“Millions of lives have been saved by treating infectious diseases at low cost in poor countries,” Dr. Hill continued. “Now we need to repeat this medical success story, with mass treatment of diabetes and clinical obesity at low prices.”
However, in an accompanying editorial, Eric A. Finkelstein, MD, and Junxing Chay, PhD, Duke-NUS Medical School, Singapore, maintain that “It would be great if everyone had affordable access to all medicines that might improve their health. Yet that is simply not possible, nor will it ever be.”
“What is truly needed is a better way to ration the health care dollars currently available in efforts to maximize population health. That is the challenge ahead not just for [antiobesity medications] but for all treatments,” they say.
“Greater use of cost-effectiveness analysis and direct negotiations, while maintaining the patent system, represents an appropriate approach for allocating scarce health care resources in the United States and beyond,” they continue.
Lowest current patented drug prices vs. estimated generic drug prices
New medications for obesity were highly effective in recent clinical trials, but high prices limit the ability of patients to get these medications, Dr. Levi and colleagues write.
They analyzed prices for obesity drugs in 16 low-, middle-, and high-income countries: Australia, Bangladesh, China, France, Germany, India, Kenya, Morocco, Norway, Peru, Pakistan, South Africa, Turkey, the United Kingdom, the United States, and Vietnam.
The researchers assessed the price of a 30-day supply of each of the studied branded drugs based on the lowest available price (in 2021 U.S. dollars) from multiple online national price databases.
Then they calculated the estimated minimum price of a 30-day supply of a potential generic version of these drugs, which included the cost of the active medicinal ingredients, the excipients (nonactive ingredients), the prefilled injectable device plus needles (for subcutaneous drugs), transportation, 10% profit, and 27% tax on profit.
The national prices of the branded medications for obesity were significantly higher than the estimated minimum prices of potential generic drugs (see Table).
The highest national price for a branded oral drug for obesity vs. the estimated minimum price for a potential generic version was $100 vs. $7 for orlistat, $199 vs. $5 for phentermine/topiramate, and $326 vs. $54 for naltrexone/bupropion, for a 30-day supply.
There was an even greater difference between highest national branded drug price vs. estimated minimum generic drug price for the newer subcutaneously injectable drugs for obesity.
For example, the price of a 30-day course of subcutaneous semaglutide ranged from $804 (United States) to $95 (Turkey), while the estimated minimum potential generic drug price was $40 (which is 20 times lower).
The study was funded by grants from the Make Medicines Affordable/International Treatment Preparedness Coalition and from the National Heart, Lung, and Blood Institute of the National Institutes of Health. Coauthor Francois Venter has reported receiving support from the Bill and Melinda Gates Foundation, U.S. Agency for International Development, Unitaid, SA Medical Research Council, Foundation for Innovative New Diagnostics, the Children’s Investment Fund Foundation, Gilead, ViiV, Mylan, Merck, Adcock Ingram, Aspen, Abbott, Roche, Johnson & Johnson, Sanofi, Virology Education, SA HIV Clinicians Society, and Dira Sengwe. The other authors and Dr. Chay have reported no relevant financial relationships. Dr. Finkelstein has reported receiving support for serving on the WW scientific advisory board and an educational grant unrelated to the present work from Novo Nordisk.
A version of this article first appeared on Medscape.com.
Tirzepatide scores win in second obesity trial, SURMOUNT-2
The “twincretin” tirzepatide (Mounjaro) has proven successful in SURMOUNT-2, the second pivotal trial for the drug as an antiobesity agent, according to top-line results reported April 27 by tirzepatide’s manufacturer, Lilly, in a press release. The company reveals that tirzepatide achieved both of its primary endpoints in the trial, as well as all its key secondary endpoints.
The findings pave the way for tirzepatide to likely receive Food and Drug Administration approval as a treatment for obesity, perhaps before the end of 2023.
Tirzepatide received FDA approval in May 2022 for the treatment of type 2 diabetes in adults, under the brand name Mounjaro, and some people have already been using it off-label to treat obesity.
Tirzepatide is a dual glucagonlike peptide–1 (GLP-1) agonist and glucose-dependent insulinotropic polypeptide agonist. Several GLP-1 receptor agonists are already approved in the United States, including semaglutide, a once-weekly injection, which is approved as Wegovy for patients with obesity and as Ozempic for treatment of type 2 diabetes.
These agents have been incredibly popular among celebrity influencers, and with use of the #Ozempic hashtag and others on social media, this has led to unprecedented use of these products for weight loss, often among those who do not even have obesity or type 2 diabetes. Subsequently, patients with type 2 diabetes and obesity who need them have often struggled to obtain them, owing to shortages following this phenomenon.
SURMOUNT-2: Weight loss around 15%, less than seen in SURMOUNT-1
SURMOUNT-2 enrolled 938 adults with overweight or obesity and type 2 diabetes and had dual primary endpoints that both focused on weight loss, compared with placebo.
The first completed pivotal trial of tirzepatide for weight loss, SURMOUNT-1, enrolled people with overweight or obesity but no diabetes and had its main results reported in 2022. At the time, the weight loss achieved with tirzepatide, was described as “unprecedented,” with those given the highest dose in that trial (15 mg subcutaneously per week) losing an average of 20%-22% of body weight over 72 weeks, depending on the specific statistical analysis used.
For SURMOUNT-2’s first primary endpoint, 72 weeks of weekly subcutaneous injections with tirzepatide at dosages of 10 mg or 15 mg led to an average weight loss from baseline of 13.4% and 15.7%, respectively, compared with an average loss of 3.3% from baseline in the placebo-treated control arm.
For the second primary endpoint, 81.6% of people on the 10-mg dose and 86.4% on the 15-mg dose achieved at least 5% weight loss from baseline, compared with 30.5% of controls who had at least 5% weight loss from baseline.
In one key secondary endpoint, tirzepatide at dosages of 10 mg or 15 mg weekly produced at least a 15% cut in weight from baseline in 41.4% and 51.8% of participants, respectively, compared with a 2.6% rate of this endpoint in the placebo controls.
So the extent of weight loss seen in in SURMOUNT-2 was somewhat less than was reported in SURMOUNT-1, a finding consistent with many prior studies of incretin-based weight-loss agents, which seem to pack a more potent weight-loss punch in people without type 2 diabetes.
Lilly did not specifically report the treatment effect of tirzepatide on hemoglobin A1c in SURMOUNT-2, only saying that the effect was similar to what had been seen in the series of five SURPASS trials that led to the approval of tirzepatide for type 2 diabetes.
Lilly also reported that the safety profile of tirzepatide in SURMOUNT-2 generally matched what was seen in SURMOUNT-1 as well as in the SURPASS trials. The most common adverse events in SURMOUNT-2 involved gastrointestinal symptoms, such as nausea, diarrhea, and vomiting; these were generally mild to moderate in severity and clustered during the dose-escalation phase at the start of treatment. Treatment discontinuations caused by adverse effects were 3.8% on the 10-mg dosage, 7.4% on the 15-mg dosage, and 3.8% on placebo.
SURMOUNT-2 enrolled patients in the United States, Puerto Rico, and five other countries. All participants also received interventions designed to reduce their calorie intake and increase their physical activity.
More SURMOUNT-2 results at ADA in June
Lilly also announced that researchers would report more complete results from SURMOUNT-2 at the 2023 scientific sessions of the American Diabetes Association, being held in San Diego in late June, and publish the findings in a major medical journal.
Results from two additional phase 3 trials of tirzepatide in people with overweight or obesity, SURMOUNT-3 and SURMOUNT-4, are expected later in 2023.
Lilly started an application to the FDA for an indication for weight loss in October 2022 under a fast track designation by the agency, and the data collected in SURMOUNT-2 are expected to complete this application, which would then be subject to an FDA decision within about 6 months. Lilly said in its April 27 press release that it anticipates an FDA decision on this application may occur before the end of 2023.
SURMOUNT-2 and all of the other tirzepatide trials were sponsored by Lilly.
A version of this article first appeared on Medscape.com.
The “twincretin” tirzepatide (Mounjaro) has proven successful in SURMOUNT-2, the second pivotal trial for the drug as an antiobesity agent, according to top-line results reported April 27 by tirzepatide’s manufacturer, Lilly, in a press release. The company reveals that tirzepatide achieved both of its primary endpoints in the trial, as well as all its key secondary endpoints.
The findings pave the way for tirzepatide to likely receive Food and Drug Administration approval as a treatment for obesity, perhaps before the end of 2023.
Tirzepatide received FDA approval in May 2022 for the treatment of type 2 diabetes in adults, under the brand name Mounjaro, and some people have already been using it off-label to treat obesity.
Tirzepatide is a dual glucagonlike peptide–1 (GLP-1) agonist and glucose-dependent insulinotropic polypeptide agonist. Several GLP-1 receptor agonists are already approved in the United States, including semaglutide, a once-weekly injection, which is approved as Wegovy for patients with obesity and as Ozempic for treatment of type 2 diabetes.
These agents have been incredibly popular among celebrity influencers, and with use of the #Ozempic hashtag and others on social media, this has led to unprecedented use of these products for weight loss, often among those who do not even have obesity or type 2 diabetes. Subsequently, patients with type 2 diabetes and obesity who need them have often struggled to obtain them, owing to shortages following this phenomenon.
SURMOUNT-2: Weight loss around 15%, less than seen in SURMOUNT-1
SURMOUNT-2 enrolled 938 adults with overweight or obesity and type 2 diabetes and had dual primary endpoints that both focused on weight loss, compared with placebo.
The first completed pivotal trial of tirzepatide for weight loss, SURMOUNT-1, enrolled people with overweight or obesity but no diabetes and had its main results reported in 2022. At the time, the weight loss achieved with tirzepatide, was described as “unprecedented,” with those given the highest dose in that trial (15 mg subcutaneously per week) losing an average of 20%-22% of body weight over 72 weeks, depending on the specific statistical analysis used.
For SURMOUNT-2’s first primary endpoint, 72 weeks of weekly subcutaneous injections with tirzepatide at dosages of 10 mg or 15 mg led to an average weight loss from baseline of 13.4% and 15.7%, respectively, compared with an average loss of 3.3% from baseline in the placebo-treated control arm.
For the second primary endpoint, 81.6% of people on the 10-mg dose and 86.4% on the 15-mg dose achieved at least 5% weight loss from baseline, compared with 30.5% of controls who had at least 5% weight loss from baseline.
In one key secondary endpoint, tirzepatide at dosages of 10 mg or 15 mg weekly produced at least a 15% cut in weight from baseline in 41.4% and 51.8% of participants, respectively, compared with a 2.6% rate of this endpoint in the placebo controls.
So the extent of weight loss seen in in SURMOUNT-2 was somewhat less than was reported in SURMOUNT-1, a finding consistent with many prior studies of incretin-based weight-loss agents, which seem to pack a more potent weight-loss punch in people without type 2 diabetes.
Lilly did not specifically report the treatment effect of tirzepatide on hemoglobin A1c in SURMOUNT-2, only saying that the effect was similar to what had been seen in the series of five SURPASS trials that led to the approval of tirzepatide for type 2 diabetes.
Lilly also reported that the safety profile of tirzepatide in SURMOUNT-2 generally matched what was seen in SURMOUNT-1 as well as in the SURPASS trials. The most common adverse events in SURMOUNT-2 involved gastrointestinal symptoms, such as nausea, diarrhea, and vomiting; these were generally mild to moderate in severity and clustered during the dose-escalation phase at the start of treatment. Treatment discontinuations caused by adverse effects were 3.8% on the 10-mg dosage, 7.4% on the 15-mg dosage, and 3.8% on placebo.
SURMOUNT-2 enrolled patients in the United States, Puerto Rico, and five other countries. All participants also received interventions designed to reduce their calorie intake and increase their physical activity.
More SURMOUNT-2 results at ADA in June
Lilly also announced that researchers would report more complete results from SURMOUNT-2 at the 2023 scientific sessions of the American Diabetes Association, being held in San Diego in late June, and publish the findings in a major medical journal.
Results from two additional phase 3 trials of tirzepatide in people with overweight or obesity, SURMOUNT-3 and SURMOUNT-4, are expected later in 2023.
Lilly started an application to the FDA for an indication for weight loss in October 2022 under a fast track designation by the agency, and the data collected in SURMOUNT-2 are expected to complete this application, which would then be subject to an FDA decision within about 6 months. Lilly said in its April 27 press release that it anticipates an FDA decision on this application may occur before the end of 2023.
SURMOUNT-2 and all of the other tirzepatide trials were sponsored by Lilly.
A version of this article first appeared on Medscape.com.
The “twincretin” tirzepatide (Mounjaro) has proven successful in SURMOUNT-2, the second pivotal trial for the drug as an antiobesity agent, according to top-line results reported April 27 by tirzepatide’s manufacturer, Lilly, in a press release. The company reveals that tirzepatide achieved both of its primary endpoints in the trial, as well as all its key secondary endpoints.
The findings pave the way for tirzepatide to likely receive Food and Drug Administration approval as a treatment for obesity, perhaps before the end of 2023.
Tirzepatide received FDA approval in May 2022 for the treatment of type 2 diabetes in adults, under the brand name Mounjaro, and some people have already been using it off-label to treat obesity.
Tirzepatide is a dual glucagonlike peptide–1 (GLP-1) agonist and glucose-dependent insulinotropic polypeptide agonist. Several GLP-1 receptor agonists are already approved in the United States, including semaglutide, a once-weekly injection, which is approved as Wegovy for patients with obesity and as Ozempic for treatment of type 2 diabetes.
These agents have been incredibly popular among celebrity influencers, and with use of the #Ozempic hashtag and others on social media, this has led to unprecedented use of these products for weight loss, often among those who do not even have obesity or type 2 diabetes. Subsequently, patients with type 2 diabetes and obesity who need them have often struggled to obtain them, owing to shortages following this phenomenon.
SURMOUNT-2: Weight loss around 15%, less than seen in SURMOUNT-1
SURMOUNT-2 enrolled 938 adults with overweight or obesity and type 2 diabetes and had dual primary endpoints that both focused on weight loss, compared with placebo.
The first completed pivotal trial of tirzepatide for weight loss, SURMOUNT-1, enrolled people with overweight or obesity but no diabetes and had its main results reported in 2022. At the time, the weight loss achieved with tirzepatide, was described as “unprecedented,” with those given the highest dose in that trial (15 mg subcutaneously per week) losing an average of 20%-22% of body weight over 72 weeks, depending on the specific statistical analysis used.
For SURMOUNT-2’s first primary endpoint, 72 weeks of weekly subcutaneous injections with tirzepatide at dosages of 10 mg or 15 mg led to an average weight loss from baseline of 13.4% and 15.7%, respectively, compared with an average loss of 3.3% from baseline in the placebo-treated control arm.
For the second primary endpoint, 81.6% of people on the 10-mg dose and 86.4% on the 15-mg dose achieved at least 5% weight loss from baseline, compared with 30.5% of controls who had at least 5% weight loss from baseline.
In one key secondary endpoint, tirzepatide at dosages of 10 mg or 15 mg weekly produced at least a 15% cut in weight from baseline in 41.4% and 51.8% of participants, respectively, compared with a 2.6% rate of this endpoint in the placebo controls.
So the extent of weight loss seen in in SURMOUNT-2 was somewhat less than was reported in SURMOUNT-1, a finding consistent with many prior studies of incretin-based weight-loss agents, which seem to pack a more potent weight-loss punch in people without type 2 diabetes.
Lilly did not specifically report the treatment effect of tirzepatide on hemoglobin A1c in SURMOUNT-2, only saying that the effect was similar to what had been seen in the series of five SURPASS trials that led to the approval of tirzepatide for type 2 diabetes.
Lilly also reported that the safety profile of tirzepatide in SURMOUNT-2 generally matched what was seen in SURMOUNT-1 as well as in the SURPASS trials. The most common adverse events in SURMOUNT-2 involved gastrointestinal symptoms, such as nausea, diarrhea, and vomiting; these were generally mild to moderate in severity and clustered during the dose-escalation phase at the start of treatment. Treatment discontinuations caused by adverse effects were 3.8% on the 10-mg dosage, 7.4% on the 15-mg dosage, and 3.8% on placebo.
SURMOUNT-2 enrolled patients in the United States, Puerto Rico, and five other countries. All participants also received interventions designed to reduce their calorie intake and increase their physical activity.
More SURMOUNT-2 results at ADA in June
Lilly also announced that researchers would report more complete results from SURMOUNT-2 at the 2023 scientific sessions of the American Diabetes Association, being held in San Diego in late June, and publish the findings in a major medical journal.
Results from two additional phase 3 trials of tirzepatide in people with overweight or obesity, SURMOUNT-3 and SURMOUNT-4, are expected later in 2023.
Lilly started an application to the FDA for an indication for weight loss in October 2022 under a fast track designation by the agency, and the data collected in SURMOUNT-2 are expected to complete this application, which would then be subject to an FDA decision within about 6 months. Lilly said in its April 27 press release that it anticipates an FDA decision on this application may occur before the end of 2023.
SURMOUNT-2 and all of the other tirzepatide trials were sponsored by Lilly.
A version of this article first appeared on Medscape.com.
Could combining topical antioxidants with a nonablative laser prevent acne scars?
PHOENIX – lesions, results from a prospective, single-center study showed.
“Acne vulgaris is the most common inflammatory dermatosis worldwide, often resulting in sequelae such as scarring, PIE, and PIH,” presenting author Jamie Hu, MD, said at the annual conference of the American Society for Laser Medicine and Surgery, where the study results were presented during an abstract session. “This dyschromia can cause greater psychological distress than the original acne lesions, and disproportionately affects skin of color patients.”
Blemish-prone skin is known to have higher levels of sebum and lower levels of antioxidants, leading to lipid peroxidation and oxidative stress, resulting in proliferation of Cutibacterium acnes and an inflammatory cascade that has recently been implicated in postinflammatory dyschromia and the development of PIE and PIH, noted Dr. Hu, a dermatology resident at the University of Miami. “Therefore, the use of antioxidants presents an opportunity to disrupt blemish and dyschromia,” she said.
One such antioxidant is silymarin, which is derived from the milk thistle plant. Recent studies have demonstrated that silymarin reduces proinflammatory mediators, prevents lipid peroxidation, and presents a new way to target the treatment of both acne and postinflammatory dyschromia.
Dr. Hu’s mentor, Jill S. Waibel, MD, owner and medical director of the Miami Dermatology and Laser Institute, hypothesized that nonablative laser therapy followed by topical application of silymarin would improve acne-associated postinflammatory dyschromia. To test her hunch, she conducted a 12-week, prospective trial in which 24 patients with PIE and/or PIH were randomized to one of two treatment arms: laser treatment with topical antioxidants or laser treatment with vehicle control. Patients received three laser treatments, each 1 month apart. The topical antioxidant used was Silymarin CF, a serum that contains 0.5% silymarin, 0.5% salicylic acid, 15% L-ascorbic acid, and 0.5% ferulic acid. (The study was sponsored by SkinCeuticals, the manufacturer of the serum.)
Laser selection was made primarily on the type of dyschromia, with PIE patients receiving treatment with the pulsed dye laser and PIH patients receiving treatment with the 1,927-nm thulium laser. Patients were treated on days 0, 28, and 56 of the 12-week study, followed by immediate application of topical antioxidants or vehicle control. They were also instructed to apply the assigned topical twice daily for the duration of the study. Patients ranged in age from 21 to 61 years, and 20 had skin types III-IV.
To evaluate efficacy, the researchers conducted blinded clinical assessments with the postacne hyperpigmentation index (PAHPI) and the Global Aesthetic Improvement Scale (GAIS), instrumentation with the Mexameter, a device that captures erythema and melanin index values, and visual diagnostics with optical coherence tomography (OCT).
Dr. Hu reported that at week 12, the PAHPI in the silymarin-plus-laser treatment group fell from an average of 3.18 to 1.74 (a decrease of 1.44), which suggested an improvement trend, compared with the laser treatment–only group, whose PAHPI fell from an average of 3.25 to 1.97 (a decrease of 1.28).
As for the GAIS, a one-time score assessed at the end of the trial, the average score for all patients was 3.24, which translated to “much improved/very much improved.” Patients in the silymarin-plus-laser treatment group had higher average scores compared with patients in the laser treatment–only group (3.35 vs. 3.10, respectively), but the differences did not reach statistical significance.
According to results of the Mexameter assessment, paired t-tests showed that the levels of intralesional melanin decreased significantly for patients in the silymarin-plus-laser treatment group, compared with the laser treatment–only group (P < .05). OCT assessments demonstrated an increase in dermal brightness in both groups, corresponding to an increase in dermal collagen, as well as an increase in blood vessel density.
In an interview at the meeting, Dr. Waibel, subsection chief of dermatology at Baptist Hospital of Miami, said that future studies will focus on long-term follow-up to determine if acne scars can be prevented by combining silymarin with lasers to prevent PIH and PIE. “That would be priceless,” she said. “I believe that the PIH is what causes damage to the collagen, and that damage to the collagen is what causes the scarring. So, if we can prevent or treat PIH, we may be able to prevent scarring.”
This approach, she added, “would decrease the pharmaceutical cost because I think there are many dermatologists who are treating PEI and PIH as active acne. You really have to have a keen eye for understanding the differences and you really have to be looking, because PIE and PIH are flat, whereas active acne consists of either comedones or nodules.”
She noted that in skin of color patients, she has seen PIH persist for 9 or 10 months after treatment with isotretinoin. “It’s not the isotretinoin causing the scars, or even the acne, it’s the prolonged inflammation,” she said.
Catherine M. DiGiorgio, MD, a Boston-based laser and cosmetic dermatologist who was asked to comment on the study, said that patients and dermatologists frequently seek alternatives to hydroquinone for unwanted hyperpigmentation.
“This topical contains an active ingredient – silymarin – obtained from the milk thistle plant along with several already well known topicals used for the treatment of acne and PIH,” said Dr. DiGiorgio, program co-chair of the 2023 ASLMS conference. “Further and larger studies are needed to demonstrate and support the effectiveness of this product and silymarin for PIH and/or PIE.”
Also commenting on the results, Ray Jalian, MD, a Los Angeles–based laser and cosmetic dermatologist, told this news organization that the study findings demonstrate the power of combining topical and laser treatment for more effective improvement in acne-related PIH.
“While the study failed to show statistically significant improvement in postinflammatory erythema with concomitant laser and topical therapy versus laser alone, the promising data supporting concurrent use of topicals and fractional lasers for treatment of PIH, particularly in dark skin phototypes, is a clinically impactful contribution to our daily practice,” he said.
Dr. Waibel disclosed that she has conducted clinical trials for many device and pharmaceutical companies including SkinCeuticals. Dr. Hu, Dr. DiGiorgio, and Dr. Jalian were not involved with the study and reported having no relevant disclosures.
PHOENIX – lesions, results from a prospective, single-center study showed.
“Acne vulgaris is the most common inflammatory dermatosis worldwide, often resulting in sequelae such as scarring, PIE, and PIH,” presenting author Jamie Hu, MD, said at the annual conference of the American Society for Laser Medicine and Surgery, where the study results were presented during an abstract session. “This dyschromia can cause greater psychological distress than the original acne lesions, and disproportionately affects skin of color patients.”
Blemish-prone skin is known to have higher levels of sebum and lower levels of antioxidants, leading to lipid peroxidation and oxidative stress, resulting in proliferation of Cutibacterium acnes and an inflammatory cascade that has recently been implicated in postinflammatory dyschromia and the development of PIE and PIH, noted Dr. Hu, a dermatology resident at the University of Miami. “Therefore, the use of antioxidants presents an opportunity to disrupt blemish and dyschromia,” she said.
One such antioxidant is silymarin, which is derived from the milk thistle plant. Recent studies have demonstrated that silymarin reduces proinflammatory mediators, prevents lipid peroxidation, and presents a new way to target the treatment of both acne and postinflammatory dyschromia.
Dr. Hu’s mentor, Jill S. Waibel, MD, owner and medical director of the Miami Dermatology and Laser Institute, hypothesized that nonablative laser therapy followed by topical application of silymarin would improve acne-associated postinflammatory dyschromia. To test her hunch, she conducted a 12-week, prospective trial in which 24 patients with PIE and/or PIH were randomized to one of two treatment arms: laser treatment with topical antioxidants or laser treatment with vehicle control. Patients received three laser treatments, each 1 month apart. The topical antioxidant used was Silymarin CF, a serum that contains 0.5% silymarin, 0.5% salicylic acid, 15% L-ascorbic acid, and 0.5% ferulic acid. (The study was sponsored by SkinCeuticals, the manufacturer of the serum.)
Laser selection was made primarily on the type of dyschromia, with PIE patients receiving treatment with the pulsed dye laser and PIH patients receiving treatment with the 1,927-nm thulium laser. Patients were treated on days 0, 28, and 56 of the 12-week study, followed by immediate application of topical antioxidants or vehicle control. They were also instructed to apply the assigned topical twice daily for the duration of the study. Patients ranged in age from 21 to 61 years, and 20 had skin types III-IV.
To evaluate efficacy, the researchers conducted blinded clinical assessments with the postacne hyperpigmentation index (PAHPI) and the Global Aesthetic Improvement Scale (GAIS), instrumentation with the Mexameter, a device that captures erythema and melanin index values, and visual diagnostics with optical coherence tomography (OCT).
Dr. Hu reported that at week 12, the PAHPI in the silymarin-plus-laser treatment group fell from an average of 3.18 to 1.74 (a decrease of 1.44), which suggested an improvement trend, compared with the laser treatment–only group, whose PAHPI fell from an average of 3.25 to 1.97 (a decrease of 1.28).
As for the GAIS, a one-time score assessed at the end of the trial, the average score for all patients was 3.24, which translated to “much improved/very much improved.” Patients in the silymarin-plus-laser treatment group had higher average scores compared with patients in the laser treatment–only group (3.35 vs. 3.10, respectively), but the differences did not reach statistical significance.
According to results of the Mexameter assessment, paired t-tests showed that the levels of intralesional melanin decreased significantly for patients in the silymarin-plus-laser treatment group, compared with the laser treatment–only group (P < .05). OCT assessments demonstrated an increase in dermal brightness in both groups, corresponding to an increase in dermal collagen, as well as an increase in blood vessel density.
In an interview at the meeting, Dr. Waibel, subsection chief of dermatology at Baptist Hospital of Miami, said that future studies will focus on long-term follow-up to determine if acne scars can be prevented by combining silymarin with lasers to prevent PIH and PIE. “That would be priceless,” she said. “I believe that the PIH is what causes damage to the collagen, and that damage to the collagen is what causes the scarring. So, if we can prevent or treat PIH, we may be able to prevent scarring.”
This approach, she added, “would decrease the pharmaceutical cost because I think there are many dermatologists who are treating PEI and PIH as active acne. You really have to have a keen eye for understanding the differences and you really have to be looking, because PIE and PIH are flat, whereas active acne consists of either comedones or nodules.”
She noted that in skin of color patients, she has seen PIH persist for 9 or 10 months after treatment with isotretinoin. “It’s not the isotretinoin causing the scars, or even the acne, it’s the prolonged inflammation,” she said.
Catherine M. DiGiorgio, MD, a Boston-based laser and cosmetic dermatologist who was asked to comment on the study, said that patients and dermatologists frequently seek alternatives to hydroquinone for unwanted hyperpigmentation.
“This topical contains an active ingredient – silymarin – obtained from the milk thistle plant along with several already well known topicals used for the treatment of acne and PIH,” said Dr. DiGiorgio, program co-chair of the 2023 ASLMS conference. “Further and larger studies are needed to demonstrate and support the effectiveness of this product and silymarin for PIH and/or PIE.”
Also commenting on the results, Ray Jalian, MD, a Los Angeles–based laser and cosmetic dermatologist, told this news organization that the study findings demonstrate the power of combining topical and laser treatment for more effective improvement in acne-related PIH.
“While the study failed to show statistically significant improvement in postinflammatory erythema with concomitant laser and topical therapy versus laser alone, the promising data supporting concurrent use of topicals and fractional lasers for treatment of PIH, particularly in dark skin phototypes, is a clinically impactful contribution to our daily practice,” he said.
Dr. Waibel disclosed that she has conducted clinical trials for many device and pharmaceutical companies including SkinCeuticals. Dr. Hu, Dr. DiGiorgio, and Dr. Jalian were not involved with the study and reported having no relevant disclosures.
PHOENIX – lesions, results from a prospective, single-center study showed.
“Acne vulgaris is the most common inflammatory dermatosis worldwide, often resulting in sequelae such as scarring, PIE, and PIH,” presenting author Jamie Hu, MD, said at the annual conference of the American Society for Laser Medicine and Surgery, where the study results were presented during an abstract session. “This dyschromia can cause greater psychological distress than the original acne lesions, and disproportionately affects skin of color patients.”
Blemish-prone skin is known to have higher levels of sebum and lower levels of antioxidants, leading to lipid peroxidation and oxidative stress, resulting in proliferation of Cutibacterium acnes and an inflammatory cascade that has recently been implicated in postinflammatory dyschromia and the development of PIE and PIH, noted Dr. Hu, a dermatology resident at the University of Miami. “Therefore, the use of antioxidants presents an opportunity to disrupt blemish and dyschromia,” she said.
One such antioxidant is silymarin, which is derived from the milk thistle plant. Recent studies have demonstrated that silymarin reduces proinflammatory mediators, prevents lipid peroxidation, and presents a new way to target the treatment of both acne and postinflammatory dyschromia.
Dr. Hu’s mentor, Jill S. Waibel, MD, owner and medical director of the Miami Dermatology and Laser Institute, hypothesized that nonablative laser therapy followed by topical application of silymarin would improve acne-associated postinflammatory dyschromia. To test her hunch, she conducted a 12-week, prospective trial in which 24 patients with PIE and/or PIH were randomized to one of two treatment arms: laser treatment with topical antioxidants or laser treatment with vehicle control. Patients received three laser treatments, each 1 month apart. The topical antioxidant used was Silymarin CF, a serum that contains 0.5% silymarin, 0.5% salicylic acid, 15% L-ascorbic acid, and 0.5% ferulic acid. (The study was sponsored by SkinCeuticals, the manufacturer of the serum.)
Laser selection was made primarily on the type of dyschromia, with PIE patients receiving treatment with the pulsed dye laser and PIH patients receiving treatment with the 1,927-nm thulium laser. Patients were treated on days 0, 28, and 56 of the 12-week study, followed by immediate application of topical antioxidants or vehicle control. They were also instructed to apply the assigned topical twice daily for the duration of the study. Patients ranged in age from 21 to 61 years, and 20 had skin types III-IV.
To evaluate efficacy, the researchers conducted blinded clinical assessments with the postacne hyperpigmentation index (PAHPI) and the Global Aesthetic Improvement Scale (GAIS), instrumentation with the Mexameter, a device that captures erythema and melanin index values, and visual diagnostics with optical coherence tomography (OCT).
Dr. Hu reported that at week 12, the PAHPI in the silymarin-plus-laser treatment group fell from an average of 3.18 to 1.74 (a decrease of 1.44), which suggested an improvement trend, compared with the laser treatment–only group, whose PAHPI fell from an average of 3.25 to 1.97 (a decrease of 1.28).
As for the GAIS, a one-time score assessed at the end of the trial, the average score for all patients was 3.24, which translated to “much improved/very much improved.” Patients in the silymarin-plus-laser treatment group had higher average scores compared with patients in the laser treatment–only group (3.35 vs. 3.10, respectively), but the differences did not reach statistical significance.
According to results of the Mexameter assessment, paired t-tests showed that the levels of intralesional melanin decreased significantly for patients in the silymarin-plus-laser treatment group, compared with the laser treatment–only group (P < .05). OCT assessments demonstrated an increase in dermal brightness in both groups, corresponding to an increase in dermal collagen, as well as an increase in blood vessel density.
In an interview at the meeting, Dr. Waibel, subsection chief of dermatology at Baptist Hospital of Miami, said that future studies will focus on long-term follow-up to determine if acne scars can be prevented by combining silymarin with lasers to prevent PIH and PIE. “That would be priceless,” she said. “I believe that the PIH is what causes damage to the collagen, and that damage to the collagen is what causes the scarring. So, if we can prevent or treat PIH, we may be able to prevent scarring.”
This approach, she added, “would decrease the pharmaceutical cost because I think there are many dermatologists who are treating PEI and PIH as active acne. You really have to have a keen eye for understanding the differences and you really have to be looking, because PIE and PIH are flat, whereas active acne consists of either comedones or nodules.”
She noted that in skin of color patients, she has seen PIH persist for 9 or 10 months after treatment with isotretinoin. “It’s not the isotretinoin causing the scars, or even the acne, it’s the prolonged inflammation,” she said.
Catherine M. DiGiorgio, MD, a Boston-based laser and cosmetic dermatologist who was asked to comment on the study, said that patients and dermatologists frequently seek alternatives to hydroquinone for unwanted hyperpigmentation.
“This topical contains an active ingredient – silymarin – obtained from the milk thistle plant along with several already well known topicals used for the treatment of acne and PIH,” said Dr. DiGiorgio, program co-chair of the 2023 ASLMS conference. “Further and larger studies are needed to demonstrate and support the effectiveness of this product and silymarin for PIH and/or PIE.”
Also commenting on the results, Ray Jalian, MD, a Los Angeles–based laser and cosmetic dermatologist, told this news organization that the study findings demonstrate the power of combining topical and laser treatment for more effective improvement in acne-related PIH.
“While the study failed to show statistically significant improvement in postinflammatory erythema with concomitant laser and topical therapy versus laser alone, the promising data supporting concurrent use of topicals and fractional lasers for treatment of PIH, particularly in dark skin phototypes, is a clinically impactful contribution to our daily practice,” he said.
Dr. Waibel disclosed that she has conducted clinical trials for many device and pharmaceutical companies including SkinCeuticals. Dr. Hu, Dr. DiGiorgio, and Dr. Jalian were not involved with the study and reported having no relevant disclosures.
AT ASLMS 2023
Drive, chip, and putt your way to osteoarthritis relief
Taking a swing against arthritis
Osteoarthritis is a tough disease to manage. Exercise helps ease the stiffness and pain of the joints, but at the same time, the disease makes it difficult to do that beneficial exercise. Even a relatively simple activity like jogging can hurt more than it helps. If only there were a low-impact exercise that was incredibly popular among the generally older population who are likely to have arthritis.
We love a good golf study here at LOTME, and a group of Australian and U.K. researchers have provided. Osteoarthritis affects 2 million people in the land down under, making it the most common source of disability there. In that population, only 64% reported their physical health to be good, very good, or excellent. Among the 459 golfers with OA that the study authors surveyed, however, the percentage reporting good health rose to more than 90%.
A similar story emerged when they looked at mental health. Nearly a quarter of nongolfers with OA reported high or very high levels of psychological distress, compared with just 8% of golfers. This pattern of improved physical and mental health remained when the researchers looked at the general, non-OA population.
This isn’t the first time golf’s been connected with improved health, and previous studies have shown golf to reduce the risks of cardiovascular disease, diabetes, and obesity, among other things. Just walking one 18-hole round significantly exceeds the CDC’s recommended 150 minutes of physical activity per week. Go out multiple times a week – leaving the cart and beer at home, American golfers – and you’ll be fit for a lifetime.
The golfers on our staff, however, are still waiting for those mental health benefits to kick in. Because when we’re adding up our scorecard after that string of four double bogeys to end the round, we’re most definitely thinking: “Yes, this sport is reducing my psychological distress. I am having fun right now.”
Battle of the sexes’ intestines
There are, we’re sure you’ve noticed, some differences between males and females. Females, for one thing, have longer small intestines than males. Everybody knows that, right? You didn’t know? Really? … Really?
Well, then, we’re guessing you haven’t read “Hidden diversity: Comparative functional morphology of humans and other species” by Erin A. McKenney, PhD, of North Carolina State University, Raleigh, and associates, which just appeared in PeerJ. We couldn’t put it down, even in the shower – a real page-turner/scroller. (It’s a great way to clean a phone, for those who also like to scroll, text, or talk on the toilet.)
The researchers got out their rulers, calipers, and string and took many measurements of the digestive systems of 45 human cadavers (21 female and 24 male), which were compared with data from 10 rats, 10 pigs, and 10 bullfrogs, which had been collected (the measurements, not the animals) by undergraduate students enrolled in a comparative anatomy laboratory course at the university.
There was little intestinal-length variation among the four-legged subjects, but when it comes to humans, females have “consistently and significantly longer small intestines than males,” the investigators noted.
The women’s small intestines, almost 14 feet long on average, were about a foot longer than the men’s, which suggests that women are better able to extract nutrients from food and “supports the canalization hypothesis, which posits that women are better able to survive during periods of stress,” coauthor Amanda Hale said in a written statement from the school. The way to a man’s heart may be through his stomach, but the way to a woman’s heart is through her duodenum, it seems.
Fascinating stuff, to be sure, but the thing that really caught our eye in the PeerJ article was the authors’ suggestion “that organs behave independently of one another, both within and across species.” Organs behaving independently? A somewhat ominous concept, no doubt, but it does explain a lot of the sounds we hear coming from our guts, which can get pretty frightening, especially on chili night.
Dog walking is dangerous business
Yes, you did read that right. A lot of strange things can send you to the emergency department. Go ahead and add dog walking onto that list.
Investigators from Johns Hopkins University estimate that over 422,000 adults presented to U.S. emergency departments with leash-dependent dog walking-related injuries between 2001 and 2020.
With almost 53% of U.S. households owning at least one dog in 2021-2022 in the wake of the COVID pet boom, this kind of occurrence is becoming more common than you think. The annual number of dog-walking injuries more than quadrupled from 7,300 to 32,000 over the course of the study, and the researchers link that spike to the promotion of dog walking for fitness, along with the boost of ownership itself.
The most common injuries listed in the National Electronic Injury Surveillance System database were finger fracture, traumatic brain injury, and shoulder sprain or strain. These mostly involved falls from being pulled, tripped, or tangled up in the leash while walking. For those aged 65 years and older, traumatic brain injury and hip fracture were the most common.
Women were 50% more likely to sustain a fracture than were men, and dog owners aged 65 and older were three times as likely to fall, twice as likely to get a fracture, and 60% more likely to have brain injury than were younger people. Now, that’s not to say younger people don’t also get hurt. After all, dogs aren’t ageists. The researchers have that data but it’s coming out later.
Meanwhile, the pitfalls involved with just trying to get our daily steps in while letting Muffin do her business have us on the lookout for random squirrels.
Taking a swing against arthritis
Osteoarthritis is a tough disease to manage. Exercise helps ease the stiffness and pain of the joints, but at the same time, the disease makes it difficult to do that beneficial exercise. Even a relatively simple activity like jogging can hurt more than it helps. If only there were a low-impact exercise that was incredibly popular among the generally older population who are likely to have arthritis.
We love a good golf study here at LOTME, and a group of Australian and U.K. researchers have provided. Osteoarthritis affects 2 million people in the land down under, making it the most common source of disability there. In that population, only 64% reported their physical health to be good, very good, or excellent. Among the 459 golfers with OA that the study authors surveyed, however, the percentage reporting good health rose to more than 90%.
A similar story emerged when they looked at mental health. Nearly a quarter of nongolfers with OA reported high or very high levels of psychological distress, compared with just 8% of golfers. This pattern of improved physical and mental health remained when the researchers looked at the general, non-OA population.
This isn’t the first time golf’s been connected with improved health, and previous studies have shown golf to reduce the risks of cardiovascular disease, diabetes, and obesity, among other things. Just walking one 18-hole round significantly exceeds the CDC’s recommended 150 minutes of physical activity per week. Go out multiple times a week – leaving the cart and beer at home, American golfers – and you’ll be fit for a lifetime.
The golfers on our staff, however, are still waiting for those mental health benefits to kick in. Because when we’re adding up our scorecard after that string of four double bogeys to end the round, we’re most definitely thinking: “Yes, this sport is reducing my psychological distress. I am having fun right now.”
Battle of the sexes’ intestines
There are, we’re sure you’ve noticed, some differences between males and females. Females, for one thing, have longer small intestines than males. Everybody knows that, right? You didn’t know? Really? … Really?
Well, then, we’re guessing you haven’t read “Hidden diversity: Comparative functional morphology of humans and other species” by Erin A. McKenney, PhD, of North Carolina State University, Raleigh, and associates, which just appeared in PeerJ. We couldn’t put it down, even in the shower – a real page-turner/scroller. (It’s a great way to clean a phone, for those who also like to scroll, text, or talk on the toilet.)
The researchers got out their rulers, calipers, and string and took many measurements of the digestive systems of 45 human cadavers (21 female and 24 male), which were compared with data from 10 rats, 10 pigs, and 10 bullfrogs, which had been collected (the measurements, not the animals) by undergraduate students enrolled in a comparative anatomy laboratory course at the university.
There was little intestinal-length variation among the four-legged subjects, but when it comes to humans, females have “consistently and significantly longer small intestines than males,” the investigators noted.
The women’s small intestines, almost 14 feet long on average, were about a foot longer than the men’s, which suggests that women are better able to extract nutrients from food and “supports the canalization hypothesis, which posits that women are better able to survive during periods of stress,” coauthor Amanda Hale said in a written statement from the school. The way to a man’s heart may be through his stomach, but the way to a woman’s heart is through her duodenum, it seems.
Fascinating stuff, to be sure, but the thing that really caught our eye in the PeerJ article was the authors’ suggestion “that organs behave independently of one another, both within and across species.” Organs behaving independently? A somewhat ominous concept, no doubt, but it does explain a lot of the sounds we hear coming from our guts, which can get pretty frightening, especially on chili night.
Dog walking is dangerous business
Yes, you did read that right. A lot of strange things can send you to the emergency department. Go ahead and add dog walking onto that list.
Investigators from Johns Hopkins University estimate that over 422,000 adults presented to U.S. emergency departments with leash-dependent dog walking-related injuries between 2001 and 2020.
With almost 53% of U.S. households owning at least one dog in 2021-2022 in the wake of the COVID pet boom, this kind of occurrence is becoming more common than you think. The annual number of dog-walking injuries more than quadrupled from 7,300 to 32,000 over the course of the study, and the researchers link that spike to the promotion of dog walking for fitness, along with the boost of ownership itself.
The most common injuries listed in the National Electronic Injury Surveillance System database were finger fracture, traumatic brain injury, and shoulder sprain or strain. These mostly involved falls from being pulled, tripped, or tangled up in the leash while walking. For those aged 65 years and older, traumatic brain injury and hip fracture were the most common.
Women were 50% more likely to sustain a fracture than were men, and dog owners aged 65 and older were three times as likely to fall, twice as likely to get a fracture, and 60% more likely to have brain injury than were younger people. Now, that’s not to say younger people don’t also get hurt. After all, dogs aren’t ageists. The researchers have that data but it’s coming out later.
Meanwhile, the pitfalls involved with just trying to get our daily steps in while letting Muffin do her business have us on the lookout for random squirrels.
Taking a swing against arthritis
Osteoarthritis is a tough disease to manage. Exercise helps ease the stiffness and pain of the joints, but at the same time, the disease makes it difficult to do that beneficial exercise. Even a relatively simple activity like jogging can hurt more than it helps. If only there were a low-impact exercise that was incredibly popular among the generally older population who are likely to have arthritis.
We love a good golf study here at LOTME, and a group of Australian and U.K. researchers have provided. Osteoarthritis affects 2 million people in the land down under, making it the most common source of disability there. In that population, only 64% reported their physical health to be good, very good, or excellent. Among the 459 golfers with OA that the study authors surveyed, however, the percentage reporting good health rose to more than 90%.
A similar story emerged when they looked at mental health. Nearly a quarter of nongolfers with OA reported high or very high levels of psychological distress, compared with just 8% of golfers. This pattern of improved physical and mental health remained when the researchers looked at the general, non-OA population.
This isn’t the first time golf’s been connected with improved health, and previous studies have shown golf to reduce the risks of cardiovascular disease, diabetes, and obesity, among other things. Just walking one 18-hole round significantly exceeds the CDC’s recommended 150 minutes of physical activity per week. Go out multiple times a week – leaving the cart and beer at home, American golfers – and you’ll be fit for a lifetime.
The golfers on our staff, however, are still waiting for those mental health benefits to kick in. Because when we’re adding up our scorecard after that string of four double bogeys to end the round, we’re most definitely thinking: “Yes, this sport is reducing my psychological distress. I am having fun right now.”
Battle of the sexes’ intestines
There are, we’re sure you’ve noticed, some differences between males and females. Females, for one thing, have longer small intestines than males. Everybody knows that, right? You didn’t know? Really? … Really?
Well, then, we’re guessing you haven’t read “Hidden diversity: Comparative functional morphology of humans and other species” by Erin A. McKenney, PhD, of North Carolina State University, Raleigh, and associates, which just appeared in PeerJ. We couldn’t put it down, even in the shower – a real page-turner/scroller. (It’s a great way to clean a phone, for those who also like to scroll, text, or talk on the toilet.)
The researchers got out their rulers, calipers, and string and took many measurements of the digestive systems of 45 human cadavers (21 female and 24 male), which were compared with data from 10 rats, 10 pigs, and 10 bullfrogs, which had been collected (the measurements, not the animals) by undergraduate students enrolled in a comparative anatomy laboratory course at the university.
There was little intestinal-length variation among the four-legged subjects, but when it comes to humans, females have “consistently and significantly longer small intestines than males,” the investigators noted.
The women’s small intestines, almost 14 feet long on average, were about a foot longer than the men’s, which suggests that women are better able to extract nutrients from food and “supports the canalization hypothesis, which posits that women are better able to survive during periods of stress,” coauthor Amanda Hale said in a written statement from the school. The way to a man’s heart may be through his stomach, but the way to a woman’s heart is through her duodenum, it seems.
Fascinating stuff, to be sure, but the thing that really caught our eye in the PeerJ article was the authors’ suggestion “that organs behave independently of one another, both within and across species.” Organs behaving independently? A somewhat ominous concept, no doubt, but it does explain a lot of the sounds we hear coming from our guts, which can get pretty frightening, especially on chili night.
Dog walking is dangerous business
Yes, you did read that right. A lot of strange things can send you to the emergency department. Go ahead and add dog walking onto that list.
Investigators from Johns Hopkins University estimate that over 422,000 adults presented to U.S. emergency departments with leash-dependent dog walking-related injuries between 2001 and 2020.
With almost 53% of U.S. households owning at least one dog in 2021-2022 in the wake of the COVID pet boom, this kind of occurrence is becoming more common than you think. The annual number of dog-walking injuries more than quadrupled from 7,300 to 32,000 over the course of the study, and the researchers link that spike to the promotion of dog walking for fitness, along with the boost of ownership itself.
The most common injuries listed in the National Electronic Injury Surveillance System database were finger fracture, traumatic brain injury, and shoulder sprain or strain. These mostly involved falls from being pulled, tripped, or tangled up in the leash while walking. For those aged 65 years and older, traumatic brain injury and hip fracture were the most common.
Women were 50% more likely to sustain a fracture than were men, and dog owners aged 65 and older were three times as likely to fall, twice as likely to get a fracture, and 60% more likely to have brain injury than were younger people. Now, that’s not to say younger people don’t also get hurt. After all, dogs aren’t ageists. The researchers have that data but it’s coming out later.
Meanwhile, the pitfalls involved with just trying to get our daily steps in while letting Muffin do her business have us on the lookout for random squirrels.
Atogepant prevents episodic migraine in some difficult-to-treat cases
BOSTON – , according to findings from a study presented at the 2023 annual meeting of the American Academy of Neurology.
Initial results from the double-blind ELEVATE trial showed the oral atogepant group had significantly fewer mean monthly migraine days (MMD) compared with a placebo group. There was also a significant difference in the number of participants who achieved 50% or greater reduction in the number of mean MMDs and a significant reduction in acute medication use days compared with the placebo group, according to Patricia Pozo-Rosich, MD, PhD, a headache specialist in the neurology department and director of the headache and craniofacial pain clinical unit and the Migraine Adaptive Brain Center at the Vall d’Hebron University Hospital in Barcelona, and colleagues.
The oral calcitonin gene-related peptide (CGRP) receptor antagonist is currently approved in the United States by the Food and Drug Administration as a preventative for both episodic and chronic migraine.
Results from ELEVATE
Overall, ELEVATE’s initial efficacy analysis population consisted of 309 adults aged between 18 and 80 years from North America and Europe with episodic migraine who had 4-14 MMDs and had treatment failure with at least two classes of conventional oral medication. After a 28-day screening period, participants received either 60 mg of oral atogepant once per day (154 participants) or a placebo (155 participants). In the efficacy analysis population, 56.0% of participants had failed two oral migraine preventative medication classes, while 44.0% failed three or more classes of medication. Dr. Pozo-Rosich noted that participants were taking a number of different oral preventatives across different medication classes, including flunarizine, beta blockers, topiramate, and amitriptyline, but data are not yet available on which participants had received certain combinations of oral medications.
“[T]hese people have already taken some type of prevention, so they’re not naive patients,” she said. “They’re usually more or less well treated in the sense of having had a contact with specialists or a general neurologist, someone that actually tries to do some prevention.”
The researchers examined change from MMDs at baseline and at 12 weeks as a primary outcome, with 50% or greater MMD reduction, change in mean monthly headache days, and change in acute medication use days as secondary outcomes. Regarding the different acute medications used, Dr. Pozo-Rosich said the main three types were analgesics, nonsteroid anti-inflammatory drugs, and triptans, with participants excluded from the trial if they were taking opioids.
The results showed participants in the atogepant group had significantly fewer mean MMDs compared with the placebo group at 12 weeks compared with baseline (–4.20 vs. –1.85 days; P < .0001). Researchers also found statistically significant improvement in the atogepant group for 50% or greater reduction in MMD, change in mean monthly headache days, and change in acute medication use days across 12 weeks of treatment compared with the placebo group. While the specific data analyses for secondary outcomes were not conducted in the initial analysis, Dr. Pozo-Rosich said the numbers “correlate with the primary outcome” as seen in other migraine trials.
Compared with the placebo group, participants in the atogepant group had higher rates of constipation (10.3% vs. 2.5%), COVID-19 (9.6% vs. 8.3%), and nausea (7.1% vs. 3.2%), while the placebo group had a higher rate of nasopharyngitis (5.1% vs. 7.6%).*
Migraine is a prevalent and undertreated disease, and patients around the world with migraine are in need of treatment options that are both safe and effective, Dr. Pozo-Rosich said in an interview. “[E]ven in these hard-to-treat or difficult-to-treat migraine patients, you have a drug that works, and is safe, and well tolerated and effective,” she said.
That’s “kind of good news for all of us,” she said. Patients “need this type of good news and solution,” she explained, because they may not tolerate or have access to injectable medications. Atogepant would also give clinicians have another option to offer patients with difficult-to-treat migraine cases, she noted. “It makes life easier for many physicians and many patients for many different reasons,” she said.
Dr. Pozo-Rosich said the likely next step in the research is to conduct the main analysis as well as post hoc analyses with accumulated data from pathology trials “to understand patterns of response, understand the sustainability of the response, [and] adherence to the treatment in the long term.”
‘Exciting that it works well’ in difficult-to-treat patients
Commenting on the study, Alan M. Rapoport, MD, clinical professor of neurology at University of California, Los Angeles, and past president of the International Headache Society, agreed that better options in migraine treatment and prevention are needed.
“We needed something that was going to be better than what we had before,” he said.
Dr. Rapoport noted the study was well designed with strongly positive results. “It looks like it’s an effective drug, and it looks really good in that it’s effective for people that have failed all these preventives that have very little hope for the future,” he said.
He specifically praised the inclusion of older participants in the population. “You never see a study on 80-year-olds,” he said, “but I like that, because they felt it would be safe. There are 80-year-old patients – fewer of them than 40-year-old patients – but there are 80-year-old patients who still have migraine, so I’m really glad they put older patients in it,” he said.
For atogepant, he noted that “some patients won’t get the side effects, and some patients will tolerate the side effects because it’s working really well.” While the study was not a head-to-head comparison against other oral migraine preventatives, he pointed out the high rate of constipation among participants in the trial setting may be a warning sign of future issues, as seen with other CGRP receptor agonists.
“I can tell you that with erenumab, the monoclonal antibody that was injected in the double-blind studies, they didn’t find any significant increase in constipation,” he explained. However, some clinicians using erenumab in the real world have reported up to 20% of their patients are constipated. “It’s not good that they’re reporting 10% are constipated” in the study, he said.
Overall, “all you can really say is it does work well,” Dr. Rapoport said. “It’s exciting that it works well in such difficult-to-treat patients, and it does come with some side effects.”
Dr. Pozo-Rosich reports serving as a consultant and developing education materials for AbbVie, Eli Lilly, Novartis, Teva Pharmaceuticals, and Pfizer. Dr. Rapoport is the editor-in-chief of Neurology Reviews; he reports being a consultant for AbbVie, the developer of atogepant. The ELEVATE trial is supported by AbbVie.
*Correction, 5/4/23: An earlier version of this article misstated the percentage of COVID-positive patients in the study population.
BOSTON – , according to findings from a study presented at the 2023 annual meeting of the American Academy of Neurology.
Initial results from the double-blind ELEVATE trial showed the oral atogepant group had significantly fewer mean monthly migraine days (MMD) compared with a placebo group. There was also a significant difference in the number of participants who achieved 50% or greater reduction in the number of mean MMDs and a significant reduction in acute medication use days compared with the placebo group, according to Patricia Pozo-Rosich, MD, PhD, a headache specialist in the neurology department and director of the headache and craniofacial pain clinical unit and the Migraine Adaptive Brain Center at the Vall d’Hebron University Hospital in Barcelona, and colleagues.
The oral calcitonin gene-related peptide (CGRP) receptor antagonist is currently approved in the United States by the Food and Drug Administration as a preventative for both episodic and chronic migraine.
Results from ELEVATE
Overall, ELEVATE’s initial efficacy analysis population consisted of 309 adults aged between 18 and 80 years from North America and Europe with episodic migraine who had 4-14 MMDs and had treatment failure with at least two classes of conventional oral medication. After a 28-day screening period, participants received either 60 mg of oral atogepant once per day (154 participants) or a placebo (155 participants). In the efficacy analysis population, 56.0% of participants had failed two oral migraine preventative medication classes, while 44.0% failed three or more classes of medication. Dr. Pozo-Rosich noted that participants were taking a number of different oral preventatives across different medication classes, including flunarizine, beta blockers, topiramate, and amitriptyline, but data are not yet available on which participants had received certain combinations of oral medications.
“[T]hese people have already taken some type of prevention, so they’re not naive patients,” she said. “They’re usually more or less well treated in the sense of having had a contact with specialists or a general neurologist, someone that actually tries to do some prevention.”
The researchers examined change from MMDs at baseline and at 12 weeks as a primary outcome, with 50% or greater MMD reduction, change in mean monthly headache days, and change in acute medication use days as secondary outcomes. Regarding the different acute medications used, Dr. Pozo-Rosich said the main three types were analgesics, nonsteroid anti-inflammatory drugs, and triptans, with participants excluded from the trial if they were taking opioids.
The results showed participants in the atogepant group had significantly fewer mean MMDs compared with the placebo group at 12 weeks compared with baseline (–4.20 vs. –1.85 days; P < .0001). Researchers also found statistically significant improvement in the atogepant group for 50% or greater reduction in MMD, change in mean monthly headache days, and change in acute medication use days across 12 weeks of treatment compared with the placebo group. While the specific data analyses for secondary outcomes were not conducted in the initial analysis, Dr. Pozo-Rosich said the numbers “correlate with the primary outcome” as seen in other migraine trials.
Compared with the placebo group, participants in the atogepant group had higher rates of constipation (10.3% vs. 2.5%), COVID-19 (9.6% vs. 8.3%), and nausea (7.1% vs. 3.2%), while the placebo group had a higher rate of nasopharyngitis (5.1% vs. 7.6%).*
Migraine is a prevalent and undertreated disease, and patients around the world with migraine are in need of treatment options that are both safe and effective, Dr. Pozo-Rosich said in an interview. “[E]ven in these hard-to-treat or difficult-to-treat migraine patients, you have a drug that works, and is safe, and well tolerated and effective,” she said.
That’s “kind of good news for all of us,” she said. Patients “need this type of good news and solution,” she explained, because they may not tolerate or have access to injectable medications. Atogepant would also give clinicians have another option to offer patients with difficult-to-treat migraine cases, she noted. “It makes life easier for many physicians and many patients for many different reasons,” she said.
Dr. Pozo-Rosich said the likely next step in the research is to conduct the main analysis as well as post hoc analyses with accumulated data from pathology trials “to understand patterns of response, understand the sustainability of the response, [and] adherence to the treatment in the long term.”
‘Exciting that it works well’ in difficult-to-treat patients
Commenting on the study, Alan M. Rapoport, MD, clinical professor of neurology at University of California, Los Angeles, and past president of the International Headache Society, agreed that better options in migraine treatment and prevention are needed.
“We needed something that was going to be better than what we had before,” he said.
Dr. Rapoport noted the study was well designed with strongly positive results. “It looks like it’s an effective drug, and it looks really good in that it’s effective for people that have failed all these preventives that have very little hope for the future,” he said.
He specifically praised the inclusion of older participants in the population. “You never see a study on 80-year-olds,” he said, “but I like that, because they felt it would be safe. There are 80-year-old patients – fewer of them than 40-year-old patients – but there are 80-year-old patients who still have migraine, so I’m really glad they put older patients in it,” he said.
For atogepant, he noted that “some patients won’t get the side effects, and some patients will tolerate the side effects because it’s working really well.” While the study was not a head-to-head comparison against other oral migraine preventatives, he pointed out the high rate of constipation among participants in the trial setting may be a warning sign of future issues, as seen with other CGRP receptor agonists.
“I can tell you that with erenumab, the monoclonal antibody that was injected in the double-blind studies, they didn’t find any significant increase in constipation,” he explained. However, some clinicians using erenumab in the real world have reported up to 20% of their patients are constipated. “It’s not good that they’re reporting 10% are constipated” in the study, he said.
Overall, “all you can really say is it does work well,” Dr. Rapoport said. “It’s exciting that it works well in such difficult-to-treat patients, and it does come with some side effects.”
Dr. Pozo-Rosich reports serving as a consultant and developing education materials for AbbVie, Eli Lilly, Novartis, Teva Pharmaceuticals, and Pfizer. Dr. Rapoport is the editor-in-chief of Neurology Reviews; he reports being a consultant for AbbVie, the developer of atogepant. The ELEVATE trial is supported by AbbVie.
*Correction, 5/4/23: An earlier version of this article misstated the percentage of COVID-positive patients in the study population.
BOSTON – , according to findings from a study presented at the 2023 annual meeting of the American Academy of Neurology.
Initial results from the double-blind ELEVATE trial showed the oral atogepant group had significantly fewer mean monthly migraine days (MMD) compared with a placebo group. There was also a significant difference in the number of participants who achieved 50% or greater reduction in the number of mean MMDs and a significant reduction in acute medication use days compared with the placebo group, according to Patricia Pozo-Rosich, MD, PhD, a headache specialist in the neurology department and director of the headache and craniofacial pain clinical unit and the Migraine Adaptive Brain Center at the Vall d’Hebron University Hospital in Barcelona, and colleagues.
The oral calcitonin gene-related peptide (CGRP) receptor antagonist is currently approved in the United States by the Food and Drug Administration as a preventative for both episodic and chronic migraine.
Results from ELEVATE
Overall, ELEVATE’s initial efficacy analysis population consisted of 309 adults aged between 18 and 80 years from North America and Europe with episodic migraine who had 4-14 MMDs and had treatment failure with at least two classes of conventional oral medication. After a 28-day screening period, participants received either 60 mg of oral atogepant once per day (154 participants) or a placebo (155 participants). In the efficacy analysis population, 56.0% of participants had failed two oral migraine preventative medication classes, while 44.0% failed three or more classes of medication. Dr. Pozo-Rosich noted that participants were taking a number of different oral preventatives across different medication classes, including flunarizine, beta blockers, topiramate, and amitriptyline, but data are not yet available on which participants had received certain combinations of oral medications.
“[T]hese people have already taken some type of prevention, so they’re not naive patients,” she said. “They’re usually more or less well treated in the sense of having had a contact with specialists or a general neurologist, someone that actually tries to do some prevention.”
The researchers examined change from MMDs at baseline and at 12 weeks as a primary outcome, with 50% or greater MMD reduction, change in mean monthly headache days, and change in acute medication use days as secondary outcomes. Regarding the different acute medications used, Dr. Pozo-Rosich said the main three types were analgesics, nonsteroid anti-inflammatory drugs, and triptans, with participants excluded from the trial if they were taking opioids.
The results showed participants in the atogepant group had significantly fewer mean MMDs compared with the placebo group at 12 weeks compared with baseline (–4.20 vs. –1.85 days; P < .0001). Researchers also found statistically significant improvement in the atogepant group for 50% or greater reduction in MMD, change in mean monthly headache days, and change in acute medication use days across 12 weeks of treatment compared with the placebo group. While the specific data analyses for secondary outcomes were not conducted in the initial analysis, Dr. Pozo-Rosich said the numbers “correlate with the primary outcome” as seen in other migraine trials.
Compared with the placebo group, participants in the atogepant group had higher rates of constipation (10.3% vs. 2.5%), COVID-19 (9.6% vs. 8.3%), and nausea (7.1% vs. 3.2%), while the placebo group had a higher rate of nasopharyngitis (5.1% vs. 7.6%).*
Migraine is a prevalent and undertreated disease, and patients around the world with migraine are in need of treatment options that are both safe and effective, Dr. Pozo-Rosich said in an interview. “[E]ven in these hard-to-treat or difficult-to-treat migraine patients, you have a drug that works, and is safe, and well tolerated and effective,” she said.
That’s “kind of good news for all of us,” she said. Patients “need this type of good news and solution,” she explained, because they may not tolerate or have access to injectable medications. Atogepant would also give clinicians have another option to offer patients with difficult-to-treat migraine cases, she noted. “It makes life easier for many physicians and many patients for many different reasons,” she said.
Dr. Pozo-Rosich said the likely next step in the research is to conduct the main analysis as well as post hoc analyses with accumulated data from pathology trials “to understand patterns of response, understand the sustainability of the response, [and] adherence to the treatment in the long term.”
‘Exciting that it works well’ in difficult-to-treat patients
Commenting on the study, Alan M. Rapoport, MD, clinical professor of neurology at University of California, Los Angeles, and past president of the International Headache Society, agreed that better options in migraine treatment and prevention are needed.
“We needed something that was going to be better than what we had before,” he said.
Dr. Rapoport noted the study was well designed with strongly positive results. “It looks like it’s an effective drug, and it looks really good in that it’s effective for people that have failed all these preventives that have very little hope for the future,” he said.
He specifically praised the inclusion of older participants in the population. “You never see a study on 80-year-olds,” he said, “but I like that, because they felt it would be safe. There are 80-year-old patients – fewer of them than 40-year-old patients – but there are 80-year-old patients who still have migraine, so I’m really glad they put older patients in it,” he said.
For atogepant, he noted that “some patients won’t get the side effects, and some patients will tolerate the side effects because it’s working really well.” While the study was not a head-to-head comparison against other oral migraine preventatives, he pointed out the high rate of constipation among participants in the trial setting may be a warning sign of future issues, as seen with other CGRP receptor agonists.
“I can tell you that with erenumab, the monoclonal antibody that was injected in the double-blind studies, they didn’t find any significant increase in constipation,” he explained. However, some clinicians using erenumab in the real world have reported up to 20% of their patients are constipated. “It’s not good that they’re reporting 10% are constipated” in the study, he said.
Overall, “all you can really say is it does work well,” Dr. Rapoport said. “It’s exciting that it works well in such difficult-to-treat patients, and it does come with some side effects.”
Dr. Pozo-Rosich reports serving as a consultant and developing education materials for AbbVie, Eli Lilly, Novartis, Teva Pharmaceuticals, and Pfizer. Dr. Rapoport is the editor-in-chief of Neurology Reviews; he reports being a consultant for AbbVie, the developer of atogepant. The ELEVATE trial is supported by AbbVie.
*Correction, 5/4/23: An earlier version of this article misstated the percentage of COVID-positive patients in the study population.
FROM AAN 2023
BMI is a flawed measure of obesity. What are alternatives?
“BMI is trash. Full stop.” This controversial tweet, which received thousands of likes and retweets, was cited in a recent article by one doctor on when physicians might stop using body mass index (BMI) to diagnose obesity.
BMI has for years been the consensus default method for assessing whether a person is overweight or has obesity, and is still widely used as the gatekeeper metric for treatment eligibility for certain weight-loss agents and bariatric surgery.
an important determinant of the cardiometabolic consequences of fat.
Alternative metrics include waist circumference and/or waist-to-height ratio (WHtR); imaging methods such as CT, MRI, and dual-energy x-ray absorptiometry (DXA); and bioelectrical impedance to assess fat volume and location. All have made some inroads on the tight grip BMI has had on obesity assessment.
Chances are, however, that BMI will not fade away anytime soon given how entrenched it has become in clinical practice and for insurance coverage, as well as its relative simplicity and precision.
“BMI is embedded in a wide range of guidelines on the use of medications and surgery. It’s embedded in Food and Drug Administration regulations and for billing and insurance coverage. It would take extremely strong data and years of work to undo the infrastructure built around BMI and replace it with something else. I don’t see that happening [anytime soon],” commented Daniel H. Bessesen, MD, a professor at the University of Colorado at Denver, Aurora, and chief of endocrinology for Denver Health.
“It would be almost impossible to replace all the studies that have used BMI with investigations using some other measure,” he said.
BMI Is ‘imperfect’
The entrenched position of BMI as the go-to metric doesn’t keep detractors from weighing in. As noted in a commentary on current clinical challenges surrounding obesity recently published in Annals of Internal Medicine, the journal’s editor-in-chief, Christine Laine, MD, and senior deputy editor Christina C. Wee, MD, listed six top issues clinicians must deal with, one of which, they say, is the need for a better measure of obesity than BMI.
“Unfortunately, BMI is an imperfect measure of body composition that differs with ethnicity, sex, body frame, and muscle mass,” noted Dr. Laine and Dr. Wee.
BMI is based on a person’s weight in kilograms divided by the square of their height in meters. A “healthy” BMI is between 18.5 and 24.9 kg/m2, overweight is 25-29.9, and 30 or greater is considered to represent obesity. However, certain ethnic groups have lower cutoffs for overweight or obesity because of evidence that such individuals can be at higher risk of obesity-related comorbidities at lower BMIs.
“BMI was chosen as the initial screening tool [for obesity] not because anyone thought it was perfect or the best measure but because of its simplicity. All you need is height, weight, and a calculator,” Dr. Wee said in an interview.
Numerous online calculators are available, including one from the Centers for Disease Control and Prevention where height in feet and inches and weight in pounds can be entered to generate the BMI.
BMI is also inherently limited by being “a proxy for adiposity” and not a direct measure, added Dr. Wee, who is also director of the Obesity Research Program of Beth Israel Deaconess Medical Center, Boston.
As such, BMI can’t distinguish between fat and muscle because it relies on weight only to gauge adiposity, noted Tiffany Powell-Wiley, MD, an obesity researcher at the National Heart, Lung, and Blood Institute in Bethesda, Md. Another shortcoming of BMI is that it “is good for distinguishing population-level risk for cardiovascular disease and other chronic diseases, but it does not help as much for distinguishing risk at an individual level,” she said in an interview.
These and other drawbacks have prompted researchers to look for other useful metrics. WHtR, for example, has recently made headway as a potential BMI alternative or complement.
The case for WHtR
Concern about overreliance on BMI despite its limitations is not new. In 2015, an American Heart Association scientific statement from the group’s Obesity Committee concluded that “BMI alone, even with lower thresholds, is a useful but not an ideal tool for identification of obesity or assessment of cardiovascular risk,” especially for people from Asian, Black, Hispanic, and Pacific Islander populations.
The writing panel also recommended that clinicians measure waist circumference annually and use that information along with BMI “to better gauge cardiovascular risk in diverse populations.”
Momentum for moving beyond BMI alone has continued to build following the AHA statement.
In September 2022, the National Institute for Health and Care Excellence, which sets policies for the United Kingdom’s National Health Service, revised its guidancefor assessment and management of people with obesity. The updated guidance recommends that when clinicians assess “adults with BMI below 35 kg/m2, measure and use their WHtR, as well as their BMI, as a practical estimate of central adiposity and use these measurements to help to assess and predict health risks.”
NICE released an extensive literature review with the revision, and based on the evidence, said that “using waist-to-height ratio as well as BMI would help give a practical estimate of central adiposity in adults with BMI under 35 kg/m2. This would in turn help professionals assess and predict health risks.”
However, the review added that, “because people with a BMI over 35 kg/m2 are always likely to have a high WHtR, the committee recognized that it may not be a useful addition for predicting health risks in this group.” The 2022 NICE review also said that it is “important to estimate central adiposity when assessing future health risks, including for people whose BMI is in the healthy-weight category.”
This new emphasis by NICE on measuring and using WHtR as part of obesity assessment “represents an important change in population health policy,” commented Dr. Powell-Wiley. “I expect more professional organizations will endorse use of waist circumference or waist-to-height ratio now that NICE has taken this step,” she predicted.
Waist circumference and WHtR may become standard measures of adiposity in clinical practice over the next 5-10 years.
The recent move by NICE to highlight a complementary role for WHtR “is another acknowledgment that BMI is an imperfect tool for stratifying cardiometabolic risk in a diverse population, especially in people with lower BMIs” because of its variability, commented Jamie Almandoz, MD, medical director of the weight wellness program at UT Southwestern Medical Center, Dallas.
WHtR vs. BMI
Another recent step forward for WHtR came with the publication of a post hoc analysis of data collected in the PARADIGM-HF trial, a study that had the primary purpose of comparing two medications for improving outcomes in more than 8,000 patients with heart failure with reduced ejection fraction.
The new analysis showed that “two indices that incorporate waist circumference and height, but not weight, showed a clearer association between greater adiposity and a higher risk of heart failure hospitalization,” compared with BMI.
WHtR was one of the two indices identified as being a better correlate for the adverse effect of excess adiposity compared with BMI.
The authors of the post hoc analysis did not design their analysis to compare WHtR with BMI. Instead, their goal was to better understand what’s known as the “obesity paradox” in people with heart failure with reduced ejection fraction: The recurring observation that, when these patients with heart failure have lower BMIs they fare worse, with higher rates of mortality and adverse cardiovascular outcomes, compared with patients with higher BMIs.
The new analysis showed that this paradox disappeared when WHtR was substituted for BMI as the obesity metric.
This “provides meaningful data about the superiority of WHtR, compared with BMI, for predicting heart failure outcomes,” said Dr. Powell-Wiley, although she cautioned that the analysis was limited by scant data in diverse populations and did not look at other important cardiovascular disease outcomes. While Dr. Powell-Wiley does not think that WHtR needs assessment in a prospective, controlled trial, she called for analysis of pooled prospective studies with more diverse populations to better document the advantages of WHtR over BMI.
The PARADIGM-HF post hoc analysis shows again how flawed BMI is for health assessment and the relative importance of an individualized understanding of a person’s body composition, Dr. Almandoz said in an interview. “As we collect more data, there is increasing awareness of how imperfect BMI is.”
Measuring waist circumference is tricky
Although WHtR looks promising as a substitute for or add-on to BMI, it has its own limitations, particularly the challenge of accurately measuring waist circumference.
Measuring waist circumference “not only takes more time but requires the assessor to be well trained about where to put the tape measure and making sure it’s measured at the same place each time,” even when different people take serial measurements from individual patients, noted Dr. Wee. Determining waist circumference can also be technically difficult when done on larger people, she added, and collectively these challenges make waist circumference “less reproducible from measurement to measurement.”
“It’s relatively clear how to standardize measurement of weight and height, but there is a huge amount of variability when the waist is measured,” agreed Dr. Almandoz. “And waist circumference also differs by ethnicity, race, sex, and body frame. There are significant differences in waist circumference levels that associate with increased health risks” between, for example, White and South Asian people.
Another limitation of waist circumference and WHtR is that they “cannot differentiate between visceral and abdominal subcutaneous adipose tissue, which are vastly different regarding cardiometabolic risk, commented Ian Neeland, MD, director of cardiovascular prevention at the University Hospitals Harrington Heart & Vascular Institute, Cleveland.
The imaging option
“Waist-to-height ratio is not the ultimate answer,” Dr. Neeland said in an interview. He instead endorsed “advanced imaging for body fat distribution,” such as CT or MRI scans, as his pick for what should be the standard obesity metric, “given that it is much more specific and actionable for both risk assessment and response to therapy. I expect slow but steady advancements that move away from BMI cutoffs, for example for bariatric surgery, given that BMI is an imprecise and crude tool.”
But although imaging with methods like CT and MRI may provide the best accuracy and precision for tracking the volume of a person’s cardiometabolically dangerous fat, they are also hampered by relatively high cost and, for CT and DXA, the issue of radiation exposure.
“CT, MRI, and DXA scans give more in-depth assessment of body composition, but should we expose people to the radiation and the cost?” Dr. Almandoz wondered.
“Height, weight, and waist circumference cost nothing to obtain,” creating a big relative disadvantage for imaging, said Naveed Sattar, MD, professor of metabolic medicine at the University of Glasgow.
“Data would need to show that imaging gives clinicians substantially more information about future risk” to justify its price, Dr. Sattar emphasized.
BMI’s limits mean adding on
Regardless of whichever alternatives to BMI end up getting used most, experts generally agree that BMI alone is looking increasingly inadequate.
“Over the next 5 years, BMI will come to be seen as a screening tool that categorizes people into general risk groups” that also needs “other metrics and variables, such as age, race, ethnicity, family history, blood glucose, and blood pressure to better describe health risk in an individual,” predicted Dr. Bessesen.
The endorsement of WHtR by NICE “will lead to more research into how to incorporate WHtR into routine practice. We need more evidence to translate what NICE said into practice,” said Dr. Sattar. “I don’t think we’ll see a shift away from BMI, but we’ll add alternative measures that are particularly useful in certain patients.”
“Because we live in diverse societies, we need to individualize risk assessment and couple that with technology that makes analysis of body composition more accessible,” agreed Dr. Almandoz. He noted that the UT Southwestern weight wellness program where he practices has, for about the past decade, routinely collected waist circumference and bioelectrical impedance data as well as BMI on all people seen in the practice for obesity concerns. Making these additional measurements on a routine basis also helps strengthen patient engagement.
“We get into trouble when we make rigid health policy and clinical decisions based on BMI alone without looking at the patient holistically,” said Dr. Wee. “Patients are more than arbitrary numbers, and clinicians should make clinical decisions based on the totality of evidence for each individual patient.”
Dr. Bessesen, Dr. Wee, Dr. Powell-Wiley, and Dr. Almandoz reported no relevant financial relationships. Dr. Neeland has reported being a consultant for Merck. Dr. Sattar has reported being a consultant or speaker for Abbott Laboratories, Afimmune, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceuticals, Janssen, MSD, Novartis, Novo Nordisk, Pfizer, Roche Diagnostics, and Sanofi.
A version of this article originally appeared on Medscape.com.
“BMI is trash. Full stop.” This controversial tweet, which received thousands of likes and retweets, was cited in a recent article by one doctor on when physicians might stop using body mass index (BMI) to diagnose obesity.
BMI has for years been the consensus default method for assessing whether a person is overweight or has obesity, and is still widely used as the gatekeeper metric for treatment eligibility for certain weight-loss agents and bariatric surgery.
an important determinant of the cardiometabolic consequences of fat.
Alternative metrics include waist circumference and/or waist-to-height ratio (WHtR); imaging methods such as CT, MRI, and dual-energy x-ray absorptiometry (DXA); and bioelectrical impedance to assess fat volume and location. All have made some inroads on the tight grip BMI has had on obesity assessment.
Chances are, however, that BMI will not fade away anytime soon given how entrenched it has become in clinical practice and for insurance coverage, as well as its relative simplicity and precision.
“BMI is embedded in a wide range of guidelines on the use of medications and surgery. It’s embedded in Food and Drug Administration regulations and for billing and insurance coverage. It would take extremely strong data and years of work to undo the infrastructure built around BMI and replace it with something else. I don’t see that happening [anytime soon],” commented Daniel H. Bessesen, MD, a professor at the University of Colorado at Denver, Aurora, and chief of endocrinology for Denver Health.
“It would be almost impossible to replace all the studies that have used BMI with investigations using some other measure,” he said.
BMI Is ‘imperfect’
The entrenched position of BMI as the go-to metric doesn’t keep detractors from weighing in. As noted in a commentary on current clinical challenges surrounding obesity recently published in Annals of Internal Medicine, the journal’s editor-in-chief, Christine Laine, MD, and senior deputy editor Christina C. Wee, MD, listed six top issues clinicians must deal with, one of which, they say, is the need for a better measure of obesity than BMI.
“Unfortunately, BMI is an imperfect measure of body composition that differs with ethnicity, sex, body frame, and muscle mass,” noted Dr. Laine and Dr. Wee.
BMI is based on a person’s weight in kilograms divided by the square of their height in meters. A “healthy” BMI is between 18.5 and 24.9 kg/m2, overweight is 25-29.9, and 30 or greater is considered to represent obesity. However, certain ethnic groups have lower cutoffs for overweight or obesity because of evidence that such individuals can be at higher risk of obesity-related comorbidities at lower BMIs.
“BMI was chosen as the initial screening tool [for obesity] not because anyone thought it was perfect or the best measure but because of its simplicity. All you need is height, weight, and a calculator,” Dr. Wee said in an interview.
Numerous online calculators are available, including one from the Centers for Disease Control and Prevention where height in feet and inches and weight in pounds can be entered to generate the BMI.
BMI is also inherently limited by being “a proxy for adiposity” and not a direct measure, added Dr. Wee, who is also director of the Obesity Research Program of Beth Israel Deaconess Medical Center, Boston.
As such, BMI can’t distinguish between fat and muscle because it relies on weight only to gauge adiposity, noted Tiffany Powell-Wiley, MD, an obesity researcher at the National Heart, Lung, and Blood Institute in Bethesda, Md. Another shortcoming of BMI is that it “is good for distinguishing population-level risk for cardiovascular disease and other chronic diseases, but it does not help as much for distinguishing risk at an individual level,” she said in an interview.
These and other drawbacks have prompted researchers to look for other useful metrics. WHtR, for example, has recently made headway as a potential BMI alternative or complement.
The case for WHtR
Concern about overreliance on BMI despite its limitations is not new. In 2015, an American Heart Association scientific statement from the group’s Obesity Committee concluded that “BMI alone, even with lower thresholds, is a useful but not an ideal tool for identification of obesity or assessment of cardiovascular risk,” especially for people from Asian, Black, Hispanic, and Pacific Islander populations.
The writing panel also recommended that clinicians measure waist circumference annually and use that information along with BMI “to better gauge cardiovascular risk in diverse populations.”
Momentum for moving beyond BMI alone has continued to build following the AHA statement.
In September 2022, the National Institute for Health and Care Excellence, which sets policies for the United Kingdom’s National Health Service, revised its guidancefor assessment and management of people with obesity. The updated guidance recommends that when clinicians assess “adults with BMI below 35 kg/m2, measure and use their WHtR, as well as their BMI, as a practical estimate of central adiposity and use these measurements to help to assess and predict health risks.”
NICE released an extensive literature review with the revision, and based on the evidence, said that “using waist-to-height ratio as well as BMI would help give a practical estimate of central adiposity in adults with BMI under 35 kg/m2. This would in turn help professionals assess and predict health risks.”
However, the review added that, “because people with a BMI over 35 kg/m2 are always likely to have a high WHtR, the committee recognized that it may not be a useful addition for predicting health risks in this group.” The 2022 NICE review also said that it is “important to estimate central adiposity when assessing future health risks, including for people whose BMI is in the healthy-weight category.”
This new emphasis by NICE on measuring and using WHtR as part of obesity assessment “represents an important change in population health policy,” commented Dr. Powell-Wiley. “I expect more professional organizations will endorse use of waist circumference or waist-to-height ratio now that NICE has taken this step,” she predicted.
Waist circumference and WHtR may become standard measures of adiposity in clinical practice over the next 5-10 years.
The recent move by NICE to highlight a complementary role for WHtR “is another acknowledgment that BMI is an imperfect tool for stratifying cardiometabolic risk in a diverse population, especially in people with lower BMIs” because of its variability, commented Jamie Almandoz, MD, medical director of the weight wellness program at UT Southwestern Medical Center, Dallas.
WHtR vs. BMI
Another recent step forward for WHtR came with the publication of a post hoc analysis of data collected in the PARADIGM-HF trial, a study that had the primary purpose of comparing two medications for improving outcomes in more than 8,000 patients with heart failure with reduced ejection fraction.
The new analysis showed that “two indices that incorporate waist circumference and height, but not weight, showed a clearer association between greater adiposity and a higher risk of heart failure hospitalization,” compared with BMI.
WHtR was one of the two indices identified as being a better correlate for the adverse effect of excess adiposity compared with BMI.
The authors of the post hoc analysis did not design their analysis to compare WHtR with BMI. Instead, their goal was to better understand what’s known as the “obesity paradox” in people with heart failure with reduced ejection fraction: The recurring observation that, when these patients with heart failure have lower BMIs they fare worse, with higher rates of mortality and adverse cardiovascular outcomes, compared with patients with higher BMIs.
The new analysis showed that this paradox disappeared when WHtR was substituted for BMI as the obesity metric.
This “provides meaningful data about the superiority of WHtR, compared with BMI, for predicting heart failure outcomes,” said Dr. Powell-Wiley, although she cautioned that the analysis was limited by scant data in diverse populations and did not look at other important cardiovascular disease outcomes. While Dr. Powell-Wiley does not think that WHtR needs assessment in a prospective, controlled trial, she called for analysis of pooled prospective studies with more diverse populations to better document the advantages of WHtR over BMI.
The PARADIGM-HF post hoc analysis shows again how flawed BMI is for health assessment and the relative importance of an individualized understanding of a person’s body composition, Dr. Almandoz said in an interview. “As we collect more data, there is increasing awareness of how imperfect BMI is.”
Measuring waist circumference is tricky
Although WHtR looks promising as a substitute for or add-on to BMI, it has its own limitations, particularly the challenge of accurately measuring waist circumference.
Measuring waist circumference “not only takes more time but requires the assessor to be well trained about where to put the tape measure and making sure it’s measured at the same place each time,” even when different people take serial measurements from individual patients, noted Dr. Wee. Determining waist circumference can also be technically difficult when done on larger people, she added, and collectively these challenges make waist circumference “less reproducible from measurement to measurement.”
“It’s relatively clear how to standardize measurement of weight and height, but there is a huge amount of variability when the waist is measured,” agreed Dr. Almandoz. “And waist circumference also differs by ethnicity, race, sex, and body frame. There are significant differences in waist circumference levels that associate with increased health risks” between, for example, White and South Asian people.
Another limitation of waist circumference and WHtR is that they “cannot differentiate between visceral and abdominal subcutaneous adipose tissue, which are vastly different regarding cardiometabolic risk, commented Ian Neeland, MD, director of cardiovascular prevention at the University Hospitals Harrington Heart & Vascular Institute, Cleveland.
The imaging option
“Waist-to-height ratio is not the ultimate answer,” Dr. Neeland said in an interview. He instead endorsed “advanced imaging for body fat distribution,” such as CT or MRI scans, as his pick for what should be the standard obesity metric, “given that it is much more specific and actionable for both risk assessment and response to therapy. I expect slow but steady advancements that move away from BMI cutoffs, for example for bariatric surgery, given that BMI is an imprecise and crude tool.”
But although imaging with methods like CT and MRI may provide the best accuracy and precision for tracking the volume of a person’s cardiometabolically dangerous fat, they are also hampered by relatively high cost and, for CT and DXA, the issue of radiation exposure.
“CT, MRI, and DXA scans give more in-depth assessment of body composition, but should we expose people to the radiation and the cost?” Dr. Almandoz wondered.
“Height, weight, and waist circumference cost nothing to obtain,” creating a big relative disadvantage for imaging, said Naveed Sattar, MD, professor of metabolic medicine at the University of Glasgow.
“Data would need to show that imaging gives clinicians substantially more information about future risk” to justify its price, Dr. Sattar emphasized.
BMI’s limits mean adding on
Regardless of whichever alternatives to BMI end up getting used most, experts generally agree that BMI alone is looking increasingly inadequate.
“Over the next 5 years, BMI will come to be seen as a screening tool that categorizes people into general risk groups” that also needs “other metrics and variables, such as age, race, ethnicity, family history, blood glucose, and blood pressure to better describe health risk in an individual,” predicted Dr. Bessesen.
The endorsement of WHtR by NICE “will lead to more research into how to incorporate WHtR into routine practice. We need more evidence to translate what NICE said into practice,” said Dr. Sattar. “I don’t think we’ll see a shift away from BMI, but we’ll add alternative measures that are particularly useful in certain patients.”
“Because we live in diverse societies, we need to individualize risk assessment and couple that with technology that makes analysis of body composition more accessible,” agreed Dr. Almandoz. He noted that the UT Southwestern weight wellness program where he practices has, for about the past decade, routinely collected waist circumference and bioelectrical impedance data as well as BMI on all people seen in the practice for obesity concerns. Making these additional measurements on a routine basis also helps strengthen patient engagement.
“We get into trouble when we make rigid health policy and clinical decisions based on BMI alone without looking at the patient holistically,” said Dr. Wee. “Patients are more than arbitrary numbers, and clinicians should make clinical decisions based on the totality of evidence for each individual patient.”
Dr. Bessesen, Dr. Wee, Dr. Powell-Wiley, and Dr. Almandoz reported no relevant financial relationships. Dr. Neeland has reported being a consultant for Merck. Dr. Sattar has reported being a consultant or speaker for Abbott Laboratories, Afimmune, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceuticals, Janssen, MSD, Novartis, Novo Nordisk, Pfizer, Roche Diagnostics, and Sanofi.
A version of this article originally appeared on Medscape.com.
“BMI is trash. Full stop.” This controversial tweet, which received thousands of likes and retweets, was cited in a recent article by one doctor on when physicians might stop using body mass index (BMI) to diagnose obesity.
BMI has for years been the consensus default method for assessing whether a person is overweight or has obesity, and is still widely used as the gatekeeper metric for treatment eligibility for certain weight-loss agents and bariatric surgery.
an important determinant of the cardiometabolic consequences of fat.
Alternative metrics include waist circumference and/or waist-to-height ratio (WHtR); imaging methods such as CT, MRI, and dual-energy x-ray absorptiometry (DXA); and bioelectrical impedance to assess fat volume and location. All have made some inroads on the tight grip BMI has had on obesity assessment.
Chances are, however, that BMI will not fade away anytime soon given how entrenched it has become in clinical practice and for insurance coverage, as well as its relative simplicity and precision.
“BMI is embedded in a wide range of guidelines on the use of medications and surgery. It’s embedded in Food and Drug Administration regulations and for billing and insurance coverage. It would take extremely strong data and years of work to undo the infrastructure built around BMI and replace it with something else. I don’t see that happening [anytime soon],” commented Daniel H. Bessesen, MD, a professor at the University of Colorado at Denver, Aurora, and chief of endocrinology for Denver Health.
“It would be almost impossible to replace all the studies that have used BMI with investigations using some other measure,” he said.
BMI Is ‘imperfect’
The entrenched position of BMI as the go-to metric doesn’t keep detractors from weighing in. As noted in a commentary on current clinical challenges surrounding obesity recently published in Annals of Internal Medicine, the journal’s editor-in-chief, Christine Laine, MD, and senior deputy editor Christina C. Wee, MD, listed six top issues clinicians must deal with, one of which, they say, is the need for a better measure of obesity than BMI.
“Unfortunately, BMI is an imperfect measure of body composition that differs with ethnicity, sex, body frame, and muscle mass,” noted Dr. Laine and Dr. Wee.
BMI is based on a person’s weight in kilograms divided by the square of their height in meters. A “healthy” BMI is between 18.5 and 24.9 kg/m2, overweight is 25-29.9, and 30 or greater is considered to represent obesity. However, certain ethnic groups have lower cutoffs for overweight or obesity because of evidence that such individuals can be at higher risk of obesity-related comorbidities at lower BMIs.
“BMI was chosen as the initial screening tool [for obesity] not because anyone thought it was perfect or the best measure but because of its simplicity. All you need is height, weight, and a calculator,” Dr. Wee said in an interview.
Numerous online calculators are available, including one from the Centers for Disease Control and Prevention where height in feet and inches and weight in pounds can be entered to generate the BMI.
BMI is also inherently limited by being “a proxy for adiposity” and not a direct measure, added Dr. Wee, who is also director of the Obesity Research Program of Beth Israel Deaconess Medical Center, Boston.
As such, BMI can’t distinguish between fat and muscle because it relies on weight only to gauge adiposity, noted Tiffany Powell-Wiley, MD, an obesity researcher at the National Heart, Lung, and Blood Institute in Bethesda, Md. Another shortcoming of BMI is that it “is good for distinguishing population-level risk for cardiovascular disease and other chronic diseases, but it does not help as much for distinguishing risk at an individual level,” she said in an interview.
These and other drawbacks have prompted researchers to look for other useful metrics. WHtR, for example, has recently made headway as a potential BMI alternative or complement.
The case for WHtR
Concern about overreliance on BMI despite its limitations is not new. In 2015, an American Heart Association scientific statement from the group’s Obesity Committee concluded that “BMI alone, even with lower thresholds, is a useful but not an ideal tool for identification of obesity or assessment of cardiovascular risk,” especially for people from Asian, Black, Hispanic, and Pacific Islander populations.
The writing panel also recommended that clinicians measure waist circumference annually and use that information along with BMI “to better gauge cardiovascular risk in diverse populations.”
Momentum for moving beyond BMI alone has continued to build following the AHA statement.
In September 2022, the National Institute for Health and Care Excellence, which sets policies for the United Kingdom’s National Health Service, revised its guidancefor assessment and management of people with obesity. The updated guidance recommends that when clinicians assess “adults with BMI below 35 kg/m2, measure and use their WHtR, as well as their BMI, as a practical estimate of central adiposity and use these measurements to help to assess and predict health risks.”
NICE released an extensive literature review with the revision, and based on the evidence, said that “using waist-to-height ratio as well as BMI would help give a practical estimate of central adiposity in adults with BMI under 35 kg/m2. This would in turn help professionals assess and predict health risks.”
However, the review added that, “because people with a BMI over 35 kg/m2 are always likely to have a high WHtR, the committee recognized that it may not be a useful addition for predicting health risks in this group.” The 2022 NICE review also said that it is “important to estimate central adiposity when assessing future health risks, including for people whose BMI is in the healthy-weight category.”
This new emphasis by NICE on measuring and using WHtR as part of obesity assessment “represents an important change in population health policy,” commented Dr. Powell-Wiley. “I expect more professional organizations will endorse use of waist circumference or waist-to-height ratio now that NICE has taken this step,” she predicted.
Waist circumference and WHtR may become standard measures of adiposity in clinical practice over the next 5-10 years.
The recent move by NICE to highlight a complementary role for WHtR “is another acknowledgment that BMI is an imperfect tool for stratifying cardiometabolic risk in a diverse population, especially in people with lower BMIs” because of its variability, commented Jamie Almandoz, MD, medical director of the weight wellness program at UT Southwestern Medical Center, Dallas.
WHtR vs. BMI
Another recent step forward for WHtR came with the publication of a post hoc analysis of data collected in the PARADIGM-HF trial, a study that had the primary purpose of comparing two medications for improving outcomes in more than 8,000 patients with heart failure with reduced ejection fraction.
The new analysis showed that “two indices that incorporate waist circumference and height, but not weight, showed a clearer association between greater adiposity and a higher risk of heart failure hospitalization,” compared with BMI.
WHtR was one of the two indices identified as being a better correlate for the adverse effect of excess adiposity compared with BMI.
The authors of the post hoc analysis did not design their analysis to compare WHtR with BMI. Instead, their goal was to better understand what’s known as the “obesity paradox” in people with heart failure with reduced ejection fraction: The recurring observation that, when these patients with heart failure have lower BMIs they fare worse, with higher rates of mortality and adverse cardiovascular outcomes, compared with patients with higher BMIs.
The new analysis showed that this paradox disappeared when WHtR was substituted for BMI as the obesity metric.
This “provides meaningful data about the superiority of WHtR, compared with BMI, for predicting heart failure outcomes,” said Dr. Powell-Wiley, although she cautioned that the analysis was limited by scant data in diverse populations and did not look at other important cardiovascular disease outcomes. While Dr. Powell-Wiley does not think that WHtR needs assessment in a prospective, controlled trial, she called for analysis of pooled prospective studies with more diverse populations to better document the advantages of WHtR over BMI.
The PARADIGM-HF post hoc analysis shows again how flawed BMI is for health assessment and the relative importance of an individualized understanding of a person’s body composition, Dr. Almandoz said in an interview. “As we collect more data, there is increasing awareness of how imperfect BMI is.”
Measuring waist circumference is tricky
Although WHtR looks promising as a substitute for or add-on to BMI, it has its own limitations, particularly the challenge of accurately measuring waist circumference.
Measuring waist circumference “not only takes more time but requires the assessor to be well trained about where to put the tape measure and making sure it’s measured at the same place each time,” even when different people take serial measurements from individual patients, noted Dr. Wee. Determining waist circumference can also be technically difficult when done on larger people, she added, and collectively these challenges make waist circumference “less reproducible from measurement to measurement.”
“It’s relatively clear how to standardize measurement of weight and height, but there is a huge amount of variability when the waist is measured,” agreed Dr. Almandoz. “And waist circumference also differs by ethnicity, race, sex, and body frame. There are significant differences in waist circumference levels that associate with increased health risks” between, for example, White and South Asian people.
Another limitation of waist circumference and WHtR is that they “cannot differentiate between visceral and abdominal subcutaneous adipose tissue, which are vastly different regarding cardiometabolic risk, commented Ian Neeland, MD, director of cardiovascular prevention at the University Hospitals Harrington Heart & Vascular Institute, Cleveland.
The imaging option
“Waist-to-height ratio is not the ultimate answer,” Dr. Neeland said in an interview. He instead endorsed “advanced imaging for body fat distribution,” such as CT or MRI scans, as his pick for what should be the standard obesity metric, “given that it is much more specific and actionable for both risk assessment and response to therapy. I expect slow but steady advancements that move away from BMI cutoffs, for example for bariatric surgery, given that BMI is an imprecise and crude tool.”
But although imaging with methods like CT and MRI may provide the best accuracy and precision for tracking the volume of a person’s cardiometabolically dangerous fat, they are also hampered by relatively high cost and, for CT and DXA, the issue of radiation exposure.
“CT, MRI, and DXA scans give more in-depth assessment of body composition, but should we expose people to the radiation and the cost?” Dr. Almandoz wondered.
“Height, weight, and waist circumference cost nothing to obtain,” creating a big relative disadvantage for imaging, said Naveed Sattar, MD, professor of metabolic medicine at the University of Glasgow.
“Data would need to show that imaging gives clinicians substantially more information about future risk” to justify its price, Dr. Sattar emphasized.
BMI’s limits mean adding on
Regardless of whichever alternatives to BMI end up getting used most, experts generally agree that BMI alone is looking increasingly inadequate.
“Over the next 5 years, BMI will come to be seen as a screening tool that categorizes people into general risk groups” that also needs “other metrics and variables, such as age, race, ethnicity, family history, blood glucose, and blood pressure to better describe health risk in an individual,” predicted Dr. Bessesen.
The endorsement of WHtR by NICE “will lead to more research into how to incorporate WHtR into routine practice. We need more evidence to translate what NICE said into practice,” said Dr. Sattar. “I don’t think we’ll see a shift away from BMI, but we’ll add alternative measures that are particularly useful in certain patients.”
“Because we live in diverse societies, we need to individualize risk assessment and couple that with technology that makes analysis of body composition more accessible,” agreed Dr. Almandoz. He noted that the UT Southwestern weight wellness program where he practices has, for about the past decade, routinely collected waist circumference and bioelectrical impedance data as well as BMI on all people seen in the practice for obesity concerns. Making these additional measurements on a routine basis also helps strengthen patient engagement.
“We get into trouble when we make rigid health policy and clinical decisions based on BMI alone without looking at the patient holistically,” said Dr. Wee. “Patients are more than arbitrary numbers, and clinicians should make clinical decisions based on the totality of evidence for each individual patient.”
Dr. Bessesen, Dr. Wee, Dr. Powell-Wiley, and Dr. Almandoz reported no relevant financial relationships. Dr. Neeland has reported being a consultant for Merck. Dr. Sattar has reported being a consultant or speaker for Abbott Laboratories, Afimmune, Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceuticals, Janssen, MSD, Novartis, Novo Nordisk, Pfizer, Roche Diagnostics, and Sanofi.
A version of this article originally appeared on Medscape.com.
Meta-analysis examines cancer risk concern for JAK inhibitors
MANCHESTER, ENGLAND – Janus kinase (JAK) inhibitors may be associated with a higher risk for cancer relative to tumor necrosis factor (TNF) inhibitors, according to a meta-analysis reported at the annual meeting of the British Society for Rheumatology.
Looking at all phase 2, 3, and 4 trials and long-term extension studies across the indications of rheumatoid arthritis, psoriatic arthritis, psoriasis, axial spondyloarthritis, inflammatory bowel disease, and atopic dermatitis, the risk ratio for any cancer developing was 1.63 when compared with anti-TNF therapy (95% confidence interval, 1.27-2.09).
By comparison, JAK inhibitor use was not significantly associated with any greater risk for cancer than methotrexate (RR, 1.06; 95% confidence interval, 0.58-1.94) or placebo (RR, 1.16; 95% CI, 0.75-1.80).
“Our data suggests that rather than JAK inhibitors necessarily being harmful, it could be more a case of TNF inhibitors being protective,” said Christopher Stovin, MBChB, a specialist registrar in rheumatology at the Princess Royal University Hospital, King’s College Hospital NHS Trust, London.
“We should stress that these are rare events in our study, roughly around 1 in every 100 patient-years of exposure,” Dr. Stovin said.
“Despite having over 80,000 years of patient exposure, the median follow-up duration for JAK inhibitors was still only 118 weeks, which for cancers [that] obviously have long latency periods is still a relatively small duration of time,” the researcher added.
“People worry about the drugs. But there is a possibility that [a] disturbed immune system plays a role per se in development of cancers,” consultant rheumatologist Anurag Bharadwaj, MD, DM, said in an interview.
“Although there are studies which attribute increased risk of cancer to different DMARDs [disease-modifying antirheumatic drugs] and biologics like TNF, but on other hand, it’s maybe that we are giving these drugs to patients who have got more serious immunological disease,” suggested Bharadwaj, who serves as the clinical lead for rheumatology at Basildon (England) Hospital, Mid & South Essex Foundation Trust.
“So, a possibility may be that the more severe or the more active the immunological inflammatory disease, the higher the chance of cancer, and these are the patients who go for the stronger medications,” Dr. Bharadwaj said.
There is an “immunological window of opportunity” when treating these inflammatory diseases, said Dr. Bharadwaj, noting that the first few months of treatment are vital. “For all immunological diseases, the more quickly you bring the immunological abnormality down, the chances of long-term complications go down, including [possibly that the] chances of cancer go down, chances of cardiovascular disease go down, and chances of lung disease go down. Hit it early, hit it hard.”
Concern over a possible higher risk for cancer with JAK inhibitors than with TNF inhibitors was raised following the release of data from the ORAL Surveillance trial, a postmarketing trial of tofacitinib (Xeljanz) that had been mandated by the Food and Drug Administration.
“This was a study looking at the coprimary endpoints of malignancy and major adverse cardiovascular events, and it was enriched with patients over the age of 50, with one additional cardiac risk factor, designed to amplify the detection of these rare events,” Dr. Stovin said.
“There was a signal of an increased risk of malignancy in the tofacitinib group, and this led to the FDA issuing a [boxed warning for all licensed JAK inhibitors] at that time,” he added.
Dr. Stovin and colleagues aimed to determine what, if any, cancer risk was associated with all available JAK inhibitors relative to placebo, TNF inhibitors, and methotrexate.
In all, data from 62 randomized controlled trials and 14 long-term extension studies were included in the meta-analysis, accounting for 82,366 patient years of follow-up. The JAK inhibitors analyzed included tofacitinib, baricitinib (Olumiant), upadacitinib (Rinvoq), filgotinib (Jyseleca), and peficitinib (Smyraf). (Filgotinib and peficitinib have not been approved by the FDA.)
The researchers performed sensitivity analyses that excluded cancers detected within the first 6 months of treatment, the use of higher than licensed JAK inhibitor doses, and patients with non-rheumatoid arthritis diagnoses, but the results remained largely unchanged, Dr. Stovin reported.
“Perhaps not surprisingly, when we removed ORAL Surveillance” from the analysis comparing JAK inhibitors and TNF inhibitors, “we lost statistical significance,” he said.
“Longitudinal observational data is needed but currently remains limited,” Dr. Stovin concluded.
Dr. Stovin and Dr. Bharadwaj reported no relevant financial relationships. The meta-analysis was independently supported. Dr. Bharadwaj was not involved in the study and provided comment ahead of the presentation.
A version of this article first appeared on Medscape.com.
MANCHESTER, ENGLAND – Janus kinase (JAK) inhibitors may be associated with a higher risk for cancer relative to tumor necrosis factor (TNF) inhibitors, according to a meta-analysis reported at the annual meeting of the British Society for Rheumatology.
Looking at all phase 2, 3, and 4 trials and long-term extension studies across the indications of rheumatoid arthritis, psoriatic arthritis, psoriasis, axial spondyloarthritis, inflammatory bowel disease, and atopic dermatitis, the risk ratio for any cancer developing was 1.63 when compared with anti-TNF therapy (95% confidence interval, 1.27-2.09).
By comparison, JAK inhibitor use was not significantly associated with any greater risk for cancer than methotrexate (RR, 1.06; 95% confidence interval, 0.58-1.94) or placebo (RR, 1.16; 95% CI, 0.75-1.80).
“Our data suggests that rather than JAK inhibitors necessarily being harmful, it could be more a case of TNF inhibitors being protective,” said Christopher Stovin, MBChB, a specialist registrar in rheumatology at the Princess Royal University Hospital, King’s College Hospital NHS Trust, London.
“We should stress that these are rare events in our study, roughly around 1 in every 100 patient-years of exposure,” Dr. Stovin said.
“Despite having over 80,000 years of patient exposure, the median follow-up duration for JAK inhibitors was still only 118 weeks, which for cancers [that] obviously have long latency periods is still a relatively small duration of time,” the researcher added.
“People worry about the drugs. But there is a possibility that [a] disturbed immune system plays a role per se in development of cancers,” consultant rheumatologist Anurag Bharadwaj, MD, DM, said in an interview.
“Although there are studies which attribute increased risk of cancer to different DMARDs [disease-modifying antirheumatic drugs] and biologics like TNF, but on other hand, it’s maybe that we are giving these drugs to patients who have got more serious immunological disease,” suggested Bharadwaj, who serves as the clinical lead for rheumatology at Basildon (England) Hospital, Mid & South Essex Foundation Trust.
“So, a possibility may be that the more severe or the more active the immunological inflammatory disease, the higher the chance of cancer, and these are the patients who go for the stronger medications,” Dr. Bharadwaj said.
There is an “immunological window of opportunity” when treating these inflammatory diseases, said Dr. Bharadwaj, noting that the first few months of treatment are vital. “For all immunological diseases, the more quickly you bring the immunological abnormality down, the chances of long-term complications go down, including [possibly that the] chances of cancer go down, chances of cardiovascular disease go down, and chances of lung disease go down. Hit it early, hit it hard.”
Concern over a possible higher risk for cancer with JAK inhibitors than with TNF inhibitors was raised following the release of data from the ORAL Surveillance trial, a postmarketing trial of tofacitinib (Xeljanz) that had been mandated by the Food and Drug Administration.
“This was a study looking at the coprimary endpoints of malignancy and major adverse cardiovascular events, and it was enriched with patients over the age of 50, with one additional cardiac risk factor, designed to amplify the detection of these rare events,” Dr. Stovin said.
“There was a signal of an increased risk of malignancy in the tofacitinib group, and this led to the FDA issuing a [boxed warning for all licensed JAK inhibitors] at that time,” he added.
Dr. Stovin and colleagues aimed to determine what, if any, cancer risk was associated with all available JAK inhibitors relative to placebo, TNF inhibitors, and methotrexate.
In all, data from 62 randomized controlled trials and 14 long-term extension studies were included in the meta-analysis, accounting for 82,366 patient years of follow-up. The JAK inhibitors analyzed included tofacitinib, baricitinib (Olumiant), upadacitinib (Rinvoq), filgotinib (Jyseleca), and peficitinib (Smyraf). (Filgotinib and peficitinib have not been approved by the FDA.)
The researchers performed sensitivity analyses that excluded cancers detected within the first 6 months of treatment, the use of higher than licensed JAK inhibitor doses, and patients with non-rheumatoid arthritis diagnoses, but the results remained largely unchanged, Dr. Stovin reported.
“Perhaps not surprisingly, when we removed ORAL Surveillance” from the analysis comparing JAK inhibitors and TNF inhibitors, “we lost statistical significance,” he said.
“Longitudinal observational data is needed but currently remains limited,” Dr. Stovin concluded.
Dr. Stovin and Dr. Bharadwaj reported no relevant financial relationships. The meta-analysis was independently supported. Dr. Bharadwaj was not involved in the study and provided comment ahead of the presentation.
A version of this article first appeared on Medscape.com.
MANCHESTER, ENGLAND – Janus kinase (JAK) inhibitors may be associated with a higher risk for cancer relative to tumor necrosis factor (TNF) inhibitors, according to a meta-analysis reported at the annual meeting of the British Society for Rheumatology.
Looking at all phase 2, 3, and 4 trials and long-term extension studies across the indications of rheumatoid arthritis, psoriatic arthritis, psoriasis, axial spondyloarthritis, inflammatory bowel disease, and atopic dermatitis, the risk ratio for any cancer developing was 1.63 when compared with anti-TNF therapy (95% confidence interval, 1.27-2.09).
By comparison, JAK inhibitor use was not significantly associated with any greater risk for cancer than methotrexate (RR, 1.06; 95% confidence interval, 0.58-1.94) or placebo (RR, 1.16; 95% CI, 0.75-1.80).
“Our data suggests that rather than JAK inhibitors necessarily being harmful, it could be more a case of TNF inhibitors being protective,” said Christopher Stovin, MBChB, a specialist registrar in rheumatology at the Princess Royal University Hospital, King’s College Hospital NHS Trust, London.
“We should stress that these are rare events in our study, roughly around 1 in every 100 patient-years of exposure,” Dr. Stovin said.
“Despite having over 80,000 years of patient exposure, the median follow-up duration for JAK inhibitors was still only 118 weeks, which for cancers [that] obviously have long latency periods is still a relatively small duration of time,” the researcher added.
“People worry about the drugs. But there is a possibility that [a] disturbed immune system plays a role per se in development of cancers,” consultant rheumatologist Anurag Bharadwaj, MD, DM, said in an interview.
“Although there are studies which attribute increased risk of cancer to different DMARDs [disease-modifying antirheumatic drugs] and biologics like TNF, but on other hand, it’s maybe that we are giving these drugs to patients who have got more serious immunological disease,” suggested Bharadwaj, who serves as the clinical lead for rheumatology at Basildon (England) Hospital, Mid & South Essex Foundation Trust.
“So, a possibility may be that the more severe or the more active the immunological inflammatory disease, the higher the chance of cancer, and these are the patients who go for the stronger medications,” Dr. Bharadwaj said.
There is an “immunological window of opportunity” when treating these inflammatory diseases, said Dr. Bharadwaj, noting that the first few months of treatment are vital. “For all immunological diseases, the more quickly you bring the immunological abnormality down, the chances of long-term complications go down, including [possibly that the] chances of cancer go down, chances of cardiovascular disease go down, and chances of lung disease go down. Hit it early, hit it hard.”
Concern over a possible higher risk for cancer with JAK inhibitors than with TNF inhibitors was raised following the release of data from the ORAL Surveillance trial, a postmarketing trial of tofacitinib (Xeljanz) that had been mandated by the Food and Drug Administration.
“This was a study looking at the coprimary endpoints of malignancy and major adverse cardiovascular events, and it was enriched with patients over the age of 50, with one additional cardiac risk factor, designed to amplify the detection of these rare events,” Dr. Stovin said.
“There was a signal of an increased risk of malignancy in the tofacitinib group, and this led to the FDA issuing a [boxed warning for all licensed JAK inhibitors] at that time,” he added.
Dr. Stovin and colleagues aimed to determine what, if any, cancer risk was associated with all available JAK inhibitors relative to placebo, TNF inhibitors, and methotrexate.
In all, data from 62 randomized controlled trials and 14 long-term extension studies were included in the meta-analysis, accounting for 82,366 patient years of follow-up. The JAK inhibitors analyzed included tofacitinib, baricitinib (Olumiant), upadacitinib (Rinvoq), filgotinib (Jyseleca), and peficitinib (Smyraf). (Filgotinib and peficitinib have not been approved by the FDA.)
The researchers performed sensitivity analyses that excluded cancers detected within the first 6 months of treatment, the use of higher than licensed JAK inhibitor doses, and patients with non-rheumatoid arthritis diagnoses, but the results remained largely unchanged, Dr. Stovin reported.
“Perhaps not surprisingly, when we removed ORAL Surveillance” from the analysis comparing JAK inhibitors and TNF inhibitors, “we lost statistical significance,” he said.
“Longitudinal observational data is needed but currently remains limited,” Dr. Stovin concluded.
Dr. Stovin and Dr. Bharadwaj reported no relevant financial relationships. The meta-analysis was independently supported. Dr. Bharadwaj was not involved in the study and provided comment ahead of the presentation.
A version of this article first appeared on Medscape.com.
AT BSR 2023












