User login
Formerly Skin & Allergy News
ass lick
assault rifle
balls
ballsac
black jack
bleach
Boko Haram
bondage
causas
cheap
child abuse
cocaine
compulsive behaviors
cost of miracles
cunt
Daech
display network stats
drug paraphernalia
explosion
fart
fda and death
fda AND warn
fda AND warning
fda AND warns
feom
fuck
gambling
gfc
gun
human trafficking
humira AND expensive
illegal
ISIL
ISIS
Islamic caliphate
Islamic state
madvocate
masturbation
mixed martial arts
MMA
molestation
national rifle association
NRA
nsfw
nuccitelli
pedophile
pedophilia
poker
porn
porn
pornography
psychedelic drug
recreational drug
sex slave rings
shit
slot machine
snort
substance abuse
terrorism
terrorist
texarkana
Texas hold 'em
UFC
section[contains(@class, 'nav-hidden')]
section[contains(@class, 'nav-hidden active')]
The leading independent newspaper covering dermatology news and commentary.
Study Evaluates Aesthetic Concerns in Asian Women
CHICAGO — presented at the annual meeting of the American Society for Dermatologic Surgery (ASDS),
“Asian Americans represent the fastest-expanding racial group in the US, underscoring the need for a comprehensive understanding of their specific aesthetic goals and needs,” said Annie Chiu, MD, a cosmetic dermatologist in Redondo Beach, California, who presented the results at the meeting. In an interview, she noted that most of this type of research has been done in White or Eurocentric populations, “so we really wanted to identify the top aesthetic concerns for Asian women as they tend to be different.”
In the study, an online survey was administered to aesthetically-inclined adults — defined as those who care about improving their appearance and are willing to go to a professional to do so — across different demographic groups in the United States. Respondents were surveyed about 41 facial and 31 body characteristics, identifying those they have and find bothersome. Maximum difference scaling was used to generate their most and least bothersome characteristics in each respective category.
Of the 3,974 women surveyed, 652 self-identified as female and Asian. The majority of Asian female respondents self-reported a Fitzpatrick skin type IV (East/Southeast [SE]Asian: 58%; Indian/Central or Southwest [CSW] Asian: 24%) or type V (East/SE Asian: 59%; Indian/CSW Asian: 30%). The findings reported at the meeting are specific to aesthetic concerns in Asian women.
Among the Asian female participants, the top three facial concerns were uneven skin color (40%), dull/dry skin (35%), and hair loss/thinning (34%). Top facial concerns in younger patients (under 30 years) were related to skin quality, such as dull skin (54%), acne scarring (51%), and large pore size (51%), whereas the most common concerns among those older than 57 years were related to under-eye bags or sagginess (60%), uneven skin tone (55%), and hair loss/thinning (47%).
Of note, acne scarring was noted as a top concern by the Indian/CSW Asian cohort. While the lines between the brows and skin sagging were top concerns in White female participants in the overall study, these concerns were not nearly as high among Asian female participants.
For all Asian female participants, the top body concerns were related to stubborn body fat in the stomach, sides, bra or back area, and arms. Stubborn body fat in the stomach area was the most frequent concern across generations (41% to 64%). East/SE Asian participants were more interested in receiving cosmetic treatments (91%) than the Indian/CSW Asian group (47%).
“Given that injectable treatments of neuromodulators and fillers are often what we focus aesthetic treatments around and thus, what we often center cosmetic consults on, it is important to remember to customize patient consultations and address specific needs with cultural sensitivity,” Dr. Chiu said. “We may not be properly recognizing and prioritizing patient discussion around concerns of dyspigmentation, skin quality, or hair thinning,” she continued, adding: “Ultimately as experts, it’s important we use this data along with what we know about structural and cutaneous differences in patients of different cultural backgrounds to optimize and prioritize treatments.”
Allergan Aesthetics, an AbbVie company, funded the study and participated in the trial design, research, analysis, data collection, interpretation of data, and the review. Dr. Chiu is a consultant, advisory board member, and investigator for Allergan, AbbVie,and Merz; and is a consultant and advisory board member for Galderma, Evolus, and Sofwave. Other authors disclosed ties with Allergan, Merz Aesthetics, Prollenium, Revance, Galderma, Alastin, Glo Pharma, and Teoxane. Two authors are AbbVie employees.
CHICAGO — presented at the annual meeting of the American Society for Dermatologic Surgery (ASDS),
“Asian Americans represent the fastest-expanding racial group in the US, underscoring the need for a comprehensive understanding of their specific aesthetic goals and needs,” said Annie Chiu, MD, a cosmetic dermatologist in Redondo Beach, California, who presented the results at the meeting. In an interview, she noted that most of this type of research has been done in White or Eurocentric populations, “so we really wanted to identify the top aesthetic concerns for Asian women as they tend to be different.”
In the study, an online survey was administered to aesthetically-inclined adults — defined as those who care about improving their appearance and are willing to go to a professional to do so — across different demographic groups in the United States. Respondents were surveyed about 41 facial and 31 body characteristics, identifying those they have and find bothersome. Maximum difference scaling was used to generate their most and least bothersome characteristics in each respective category.
Of the 3,974 women surveyed, 652 self-identified as female and Asian. The majority of Asian female respondents self-reported a Fitzpatrick skin type IV (East/Southeast [SE]Asian: 58%; Indian/Central or Southwest [CSW] Asian: 24%) or type V (East/SE Asian: 59%; Indian/CSW Asian: 30%). The findings reported at the meeting are specific to aesthetic concerns in Asian women.
Among the Asian female participants, the top three facial concerns were uneven skin color (40%), dull/dry skin (35%), and hair loss/thinning (34%). Top facial concerns in younger patients (under 30 years) were related to skin quality, such as dull skin (54%), acne scarring (51%), and large pore size (51%), whereas the most common concerns among those older than 57 years were related to under-eye bags or sagginess (60%), uneven skin tone (55%), and hair loss/thinning (47%).
Of note, acne scarring was noted as a top concern by the Indian/CSW Asian cohort. While the lines between the brows and skin sagging were top concerns in White female participants in the overall study, these concerns were not nearly as high among Asian female participants.
For all Asian female participants, the top body concerns were related to stubborn body fat in the stomach, sides, bra or back area, and arms. Stubborn body fat in the stomach area was the most frequent concern across generations (41% to 64%). East/SE Asian participants were more interested in receiving cosmetic treatments (91%) than the Indian/CSW Asian group (47%).
“Given that injectable treatments of neuromodulators and fillers are often what we focus aesthetic treatments around and thus, what we often center cosmetic consults on, it is important to remember to customize patient consultations and address specific needs with cultural sensitivity,” Dr. Chiu said. “We may not be properly recognizing and prioritizing patient discussion around concerns of dyspigmentation, skin quality, or hair thinning,” she continued, adding: “Ultimately as experts, it’s important we use this data along with what we know about structural and cutaneous differences in patients of different cultural backgrounds to optimize and prioritize treatments.”
Allergan Aesthetics, an AbbVie company, funded the study and participated in the trial design, research, analysis, data collection, interpretation of data, and the review. Dr. Chiu is a consultant, advisory board member, and investigator for Allergan, AbbVie,and Merz; and is a consultant and advisory board member for Galderma, Evolus, and Sofwave. Other authors disclosed ties with Allergan, Merz Aesthetics, Prollenium, Revance, Galderma, Alastin, Glo Pharma, and Teoxane. Two authors are AbbVie employees.
CHICAGO — presented at the annual meeting of the American Society for Dermatologic Surgery (ASDS),
“Asian Americans represent the fastest-expanding racial group in the US, underscoring the need for a comprehensive understanding of their specific aesthetic goals and needs,” said Annie Chiu, MD, a cosmetic dermatologist in Redondo Beach, California, who presented the results at the meeting. In an interview, she noted that most of this type of research has been done in White or Eurocentric populations, “so we really wanted to identify the top aesthetic concerns for Asian women as they tend to be different.”
In the study, an online survey was administered to aesthetically-inclined adults — defined as those who care about improving their appearance and are willing to go to a professional to do so — across different demographic groups in the United States. Respondents were surveyed about 41 facial and 31 body characteristics, identifying those they have and find bothersome. Maximum difference scaling was used to generate their most and least bothersome characteristics in each respective category.
Of the 3,974 women surveyed, 652 self-identified as female and Asian. The majority of Asian female respondents self-reported a Fitzpatrick skin type IV (East/Southeast [SE]Asian: 58%; Indian/Central or Southwest [CSW] Asian: 24%) or type V (East/SE Asian: 59%; Indian/CSW Asian: 30%). The findings reported at the meeting are specific to aesthetic concerns in Asian women.
Among the Asian female participants, the top three facial concerns were uneven skin color (40%), dull/dry skin (35%), and hair loss/thinning (34%). Top facial concerns in younger patients (under 30 years) were related to skin quality, such as dull skin (54%), acne scarring (51%), and large pore size (51%), whereas the most common concerns among those older than 57 years were related to under-eye bags or sagginess (60%), uneven skin tone (55%), and hair loss/thinning (47%).
Of note, acne scarring was noted as a top concern by the Indian/CSW Asian cohort. While the lines between the brows and skin sagging were top concerns in White female participants in the overall study, these concerns were not nearly as high among Asian female participants.
For all Asian female participants, the top body concerns were related to stubborn body fat in the stomach, sides, bra or back area, and arms. Stubborn body fat in the stomach area was the most frequent concern across generations (41% to 64%). East/SE Asian participants were more interested in receiving cosmetic treatments (91%) than the Indian/CSW Asian group (47%).
“Given that injectable treatments of neuromodulators and fillers are often what we focus aesthetic treatments around and thus, what we often center cosmetic consults on, it is important to remember to customize patient consultations and address specific needs with cultural sensitivity,” Dr. Chiu said. “We may not be properly recognizing and prioritizing patient discussion around concerns of dyspigmentation, skin quality, or hair thinning,” she continued, adding: “Ultimately as experts, it’s important we use this data along with what we know about structural and cutaneous differences in patients of different cultural backgrounds to optimize and prioritize treatments.”
Allergan Aesthetics, an AbbVie company, funded the study and participated in the trial design, research, analysis, data collection, interpretation of data, and the review. Dr. Chiu is a consultant, advisory board member, and investigator for Allergan, AbbVie,and Merz; and is a consultant and advisory board member for Galderma, Evolus, and Sofwave. Other authors disclosed ties with Allergan, Merz Aesthetics, Prollenium, Revance, Galderma, Alastin, Glo Pharma, and Teoxane. Two authors are AbbVie employees.
AT ASDS 2023
Study Concludes Most Melanoma Overdiagnoses Are In Situ
TOPLINE:
METHODOLOGY:
- The increase in melanoma diagnoses in the United States, while mortality has remained flat, has raised concerns about overdiagnosis of melanoma, cases that may not result in harm if left untreated. How much of the overdiagnoses can be attributed to melanoma in situ vs invasive melanoma is unknown.
- To address this question, researchers collected data from the SEER 9 registries database.
- They used DevCan software to calculate the cumulative lifetime risk of White American men and women being diagnosed with melanoma between 1975 and 2018, adjusting for changes in longevity and risk factors over the study period.
- The primary outcome was excess lifetime risk for melanoma diagnosis between 1976 and 2018, adjusted for year 2018 competing mortality and changes in risk factors.
TAKEAWAY:
- Researchers found that between 1975 and 2018, the adjusted lifetime risk of being diagnosed with melanoma in situ increased from 0.17% to 2.7% in White men and 0.08% to 2% in White women.
- An estimated 49.7% and 64.6% of melanomas diagnosed in White men and White women, respectively, were overdiagnosed in 2018.
- Among individuals diagnosed with melanoma in situ, 89.4% of White men and 85.4% of White women were likely overdiagnosed in 2018.
IN PRACTICE:
“A large proportion of overdiagnosed melanomas are in situ cancers, pointing to a potential area to focus for an intervention de-escalation of the intensity of treatment and survivorship care,” the authors wrote.
SOURCE:
Adewole S. Adamson, MD, of the Division of Dermatology at The University of Texas at Austin Dell Medical School, led the research. The study was published in BMJ Evidence-Based Medicine on January 19, 2024.
LIMITATIONS:
The analysis only involved White individuals. Other limitations include a high risk for selection bias and that the researchers assumed no melanoma diagnosis in 1975, which may not be the case.
DISCLOSURES:
Dr. Adamson disclosed that he is supported by the Robert Wood Johnson Foundation through The Harold Amos Medical Faculty Development Program. Coauthor Katy J.L. Bell, MBchB, PhD, of the University of Sydney, is supported by an Australian Government National Health and Medical Research Council Investigator Grant.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- The increase in melanoma diagnoses in the United States, while mortality has remained flat, has raised concerns about overdiagnosis of melanoma, cases that may not result in harm if left untreated. How much of the overdiagnoses can be attributed to melanoma in situ vs invasive melanoma is unknown.
- To address this question, researchers collected data from the SEER 9 registries database.
- They used DevCan software to calculate the cumulative lifetime risk of White American men and women being diagnosed with melanoma between 1975 and 2018, adjusting for changes in longevity and risk factors over the study period.
- The primary outcome was excess lifetime risk for melanoma diagnosis between 1976 and 2018, adjusted for year 2018 competing mortality and changes in risk factors.
TAKEAWAY:
- Researchers found that between 1975 and 2018, the adjusted lifetime risk of being diagnosed with melanoma in situ increased from 0.17% to 2.7% in White men and 0.08% to 2% in White women.
- An estimated 49.7% and 64.6% of melanomas diagnosed in White men and White women, respectively, were overdiagnosed in 2018.
- Among individuals diagnosed with melanoma in situ, 89.4% of White men and 85.4% of White women were likely overdiagnosed in 2018.
IN PRACTICE:
“A large proportion of overdiagnosed melanomas are in situ cancers, pointing to a potential area to focus for an intervention de-escalation of the intensity of treatment and survivorship care,” the authors wrote.
SOURCE:
Adewole S. Adamson, MD, of the Division of Dermatology at The University of Texas at Austin Dell Medical School, led the research. The study was published in BMJ Evidence-Based Medicine on January 19, 2024.
LIMITATIONS:
The analysis only involved White individuals. Other limitations include a high risk for selection bias and that the researchers assumed no melanoma diagnosis in 1975, which may not be the case.
DISCLOSURES:
Dr. Adamson disclosed that he is supported by the Robert Wood Johnson Foundation through The Harold Amos Medical Faculty Development Program. Coauthor Katy J.L. Bell, MBchB, PhD, of the University of Sydney, is supported by an Australian Government National Health and Medical Research Council Investigator Grant.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- The increase in melanoma diagnoses in the United States, while mortality has remained flat, has raised concerns about overdiagnosis of melanoma, cases that may not result in harm if left untreated. How much of the overdiagnoses can be attributed to melanoma in situ vs invasive melanoma is unknown.
- To address this question, researchers collected data from the SEER 9 registries database.
- They used DevCan software to calculate the cumulative lifetime risk of White American men and women being diagnosed with melanoma between 1975 and 2018, adjusting for changes in longevity and risk factors over the study period.
- The primary outcome was excess lifetime risk for melanoma diagnosis between 1976 and 2018, adjusted for year 2018 competing mortality and changes in risk factors.
TAKEAWAY:
- Researchers found that between 1975 and 2018, the adjusted lifetime risk of being diagnosed with melanoma in situ increased from 0.17% to 2.7% in White men and 0.08% to 2% in White women.
- An estimated 49.7% and 64.6% of melanomas diagnosed in White men and White women, respectively, were overdiagnosed in 2018.
- Among individuals diagnosed with melanoma in situ, 89.4% of White men and 85.4% of White women were likely overdiagnosed in 2018.
IN PRACTICE:
“A large proportion of overdiagnosed melanomas are in situ cancers, pointing to a potential area to focus for an intervention de-escalation of the intensity of treatment and survivorship care,” the authors wrote.
SOURCE:
Adewole S. Adamson, MD, of the Division of Dermatology at The University of Texas at Austin Dell Medical School, led the research. The study was published in BMJ Evidence-Based Medicine on January 19, 2024.
LIMITATIONS:
The analysis only involved White individuals. Other limitations include a high risk for selection bias and that the researchers assumed no melanoma diagnosis in 1975, which may not be the case.
DISCLOSURES:
Dr. Adamson disclosed that he is supported by the Robert Wood Johnson Foundation through The Harold Amos Medical Faculty Development Program. Coauthor Katy J.L. Bell, MBchB, PhD, of the University of Sydney, is supported by an Australian Government National Health and Medical Research Council Investigator Grant.
A version of this article first appeared on Medscape.com.
Rituximab Results in Sustained Remission for Pemphigus, Study Found
TOPLINE:
, an analysis showed.
METHODOLOGY:
- The short-term efficacy and safety of first-line treatment with rituximab for pemphigus were demonstrated in the Ritux 3 trial, but the rates of long-term remission are unknown.
- French investigators from 25 dermatology departments evaluated 83 patients from the Ritux 3 trial between January 1, 2010, and December 31, 2015.
- They used Kaplan-Meir curves to determine the 5- and 7-year rates of disease-free survival (DFS) without corticosteroids.
TAKEAWAY:
- Of the 83 patients, 44 were in the rituximab-plus-prednisone group and 39 were in the prednisone-only group, with a median follow-up of 87.3 months (7.3 years).
- Among patients in the rituximab plus prednisone group, 43 (93.5%) achieved complete remission without corticosteroids at any time during follow-up, compared with 17 patients (39%) in the prednisone-only group.
- DFS (without corticosteroid therapy) statistically favored patients in the rituximab plus prednisone group compared with patients in the prednisone-only group at follow-up times of 5 years (76.7% vs 35.3%, respectively) and 7 years (72.1% vs 35.3%; P < .001 for both associations).
- In another finding, 31 patients in the rituximab plus prednisone group reported fewer serious adverse events (SAEs) than 58 patients in the prednisone-only group, which corresponds to 0.67 and 1.32 SAEs per patient, respectively (P = .003).
IN PRACTICE:
The study findings demonstrated “the superiority of rituximab over a standard corticosteroids regimen, both in the short term and the long term,” the authors wrote.
SOURCE:
Corresponding author Billal Tedbirt, MD, of the Department of Dermatology at CHU Rouen in France, led the study, which was published online on January 24, 2024, in JAMA Dermatology.
LIMITATIONS:
Nearly 8% of patients did not attend the end of follow-up visit. Also, serum samples used to predict relapse were drawn at month 36, but the researchers said that a window of every 4-6 months might provide higher accuracy of relapses.
DISCLOSURES:
Dr. Tedbirt reported having no disclosures. Four of the study authors reported being investigators for and/or receiving personal fees from several pharmaceutical companies. The study was supported by a grant from the French Society of Dermatology.
A version of this article appeared on Medscape.com.
TOPLINE:
, an analysis showed.
METHODOLOGY:
- The short-term efficacy and safety of first-line treatment with rituximab for pemphigus were demonstrated in the Ritux 3 trial, but the rates of long-term remission are unknown.
- French investigators from 25 dermatology departments evaluated 83 patients from the Ritux 3 trial between January 1, 2010, and December 31, 2015.
- They used Kaplan-Meir curves to determine the 5- and 7-year rates of disease-free survival (DFS) without corticosteroids.
TAKEAWAY:
- Of the 83 patients, 44 were in the rituximab-plus-prednisone group and 39 were in the prednisone-only group, with a median follow-up of 87.3 months (7.3 years).
- Among patients in the rituximab plus prednisone group, 43 (93.5%) achieved complete remission without corticosteroids at any time during follow-up, compared with 17 patients (39%) in the prednisone-only group.
- DFS (without corticosteroid therapy) statistically favored patients in the rituximab plus prednisone group compared with patients in the prednisone-only group at follow-up times of 5 years (76.7% vs 35.3%, respectively) and 7 years (72.1% vs 35.3%; P < .001 for both associations).
- In another finding, 31 patients in the rituximab plus prednisone group reported fewer serious adverse events (SAEs) than 58 patients in the prednisone-only group, which corresponds to 0.67 and 1.32 SAEs per patient, respectively (P = .003).
IN PRACTICE:
The study findings demonstrated “the superiority of rituximab over a standard corticosteroids regimen, both in the short term and the long term,” the authors wrote.
SOURCE:
Corresponding author Billal Tedbirt, MD, of the Department of Dermatology at CHU Rouen in France, led the study, which was published online on January 24, 2024, in JAMA Dermatology.
LIMITATIONS:
Nearly 8% of patients did not attend the end of follow-up visit. Also, serum samples used to predict relapse were drawn at month 36, but the researchers said that a window of every 4-6 months might provide higher accuracy of relapses.
DISCLOSURES:
Dr. Tedbirt reported having no disclosures. Four of the study authors reported being investigators for and/or receiving personal fees from several pharmaceutical companies. The study was supported by a grant from the French Society of Dermatology.
A version of this article appeared on Medscape.com.
TOPLINE:
, an analysis showed.
METHODOLOGY:
- The short-term efficacy and safety of first-line treatment with rituximab for pemphigus were demonstrated in the Ritux 3 trial, but the rates of long-term remission are unknown.
- French investigators from 25 dermatology departments evaluated 83 patients from the Ritux 3 trial between January 1, 2010, and December 31, 2015.
- They used Kaplan-Meir curves to determine the 5- and 7-year rates of disease-free survival (DFS) without corticosteroids.
TAKEAWAY:
- Of the 83 patients, 44 were in the rituximab-plus-prednisone group and 39 were in the prednisone-only group, with a median follow-up of 87.3 months (7.3 years).
- Among patients in the rituximab plus prednisone group, 43 (93.5%) achieved complete remission without corticosteroids at any time during follow-up, compared with 17 patients (39%) in the prednisone-only group.
- DFS (without corticosteroid therapy) statistically favored patients in the rituximab plus prednisone group compared with patients in the prednisone-only group at follow-up times of 5 years (76.7% vs 35.3%, respectively) and 7 years (72.1% vs 35.3%; P < .001 for both associations).
- In another finding, 31 patients in the rituximab plus prednisone group reported fewer serious adverse events (SAEs) than 58 patients in the prednisone-only group, which corresponds to 0.67 and 1.32 SAEs per patient, respectively (P = .003).
IN PRACTICE:
The study findings demonstrated “the superiority of rituximab over a standard corticosteroids regimen, both in the short term and the long term,” the authors wrote.
SOURCE:
Corresponding author Billal Tedbirt, MD, of the Department of Dermatology at CHU Rouen in France, led the study, which was published online on January 24, 2024, in JAMA Dermatology.
LIMITATIONS:
Nearly 8% of patients did not attend the end of follow-up visit. Also, serum samples used to predict relapse were drawn at month 36, but the researchers said that a window of every 4-6 months might provide higher accuracy of relapses.
DISCLOSURES:
Dr. Tedbirt reported having no disclosures. Four of the study authors reported being investigators for and/or receiving personal fees from several pharmaceutical companies. The study was supported by a grant from the French Society of Dermatology.
A version of this article appeared on Medscape.com.
Medical Aid in Dying Should Be Legal, Says Ethicist
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the Division of Medical Ethics at the NYU Grossman School of Medicine.
Right now, there are 10 states and the District of Columbia that have had some version of medical assistance in dying approved and on the books. That basically means that about 20% of Americans have access where they live to a physician who can prescribe a lethal dose of medication to them if they’re terminally ill and can ingest the medication themselves. That leaves many Americans not covered by this kind of access to this kind of service.
Many of you watching this may live in states where it is legal, like Oregon, Washington, New Jersey, Colorado, and Hawaii. I know many doctors say, “I’m not going to do that.” It’s not something that anyone is compelling a doctor to do. For some Americans, access is not just about where they live but whether there is a doctor willing to participate with them in bringing about their accelerated death, knowing that they’re inevitably going to die.
There’s not much we can do about that. It’s up to the conscience of each physician as to what they’re comfortable with. Certainly, there are other things that can be done to extend the possibility of having this available.
One thing that’s taking place is that, after lawsuits were filed, Vermont and Oregon have given up on their residency requirement, so you don’t have to be there 6 months or a year in order to use this opportunity. It’s legal now to move to the state or visit the state, and as soon as you get there, sign up for this kind of end-of-life intervention.
New Jersey is also being sued. I’ll predict that every state that has a residency requirement, when sued in court, is going to lose because we’ve long recognized the right of Americans to seek out healthcare in the United States, wherever they want to go.
If some states have made this a legitimate medical procedure, courts are going to say you can’t restrict it only to state residents. If someone wants to use a service, they’re entitled to show up from another state or another place and use it. I’m not sure about foreign nationals, but I’m very sure that Americans can go state to state in search of legitimate medical procedures.
The other bills that are out there, however, are basically saying they want to emulate Oregon, Washington, and the other states and say that the terminally ill, with severe restrictions, are going to be able to get this service without going anywhere.
The restrictions include a diagnosis of terminal illness and that you have to be deemed mentally competent. You can’t use this if you have Alzheimer’s or severe depression. You have to make a request twice with a week or two in between to make sure that your request is authentic. And obviously, everyone is on board to make sure that you’re not being coerced or pushed somehow into requesting a somewhat earlier death than you would have experienced without having the availability of the pills.
You also have to take the pills yourself or be able to pull a switch so that you could use a feeding tube–type administration. If you can’t do that, say due to ALS, you’re not eligible to use medical aid in dying. It’s a pretty restricted intervention.
Many people who get pills after going through these restrictions in the states that permit it don’t use it. As many as one third say they like having it there as a safety valve or a parachute, but once they know they could end their life sooner, then they’re going to stick it out.
Should states make this legal? New York, Massachusetts, Florida, and many other states have bills that are moving through. I’m going to say yes. We’ve had Oregon and Washington since the late 1990s with medical aid in dying on the books. There doesn’t seem to be any evidence of pushing people to use this, of bias against the disabled, or bigotry against particular ethnic or racial groups being used to encourage people to end their life sooner.
I think it is an option that Americans want. I think it’s an option that makes some sense. I’m well aware that we also have to make sure that people know about hospice. In some of these states, medical aid in dying is offered as a part of hospice — not all, but a few. Not everybody wants hospice once they realize that they’re dying and that it is coming relatively soon. They may want to leave with family present, with a ceremony, or with a quality of life that they desire.
Past experience says let’s continue to expand availability in each state. Let’s also realize that we have to keep the restrictions in place on how it’s used because they have protected us against abuse. Let’s understand that every doctor has an option to do this or not do this. It’s a matter of conscience and a matter of comfort.
I think legalization is the direction we’re going to be going in. Getting rid of the residency requirements that have been around, as I think courts are going to overturn them, also gives a push to the idea that once the service is in this many states, it’s something that should be available if there are doctors willing to do it.
I’m Art Caplan at the Division of Medical Ethics at NYU Grossman School of Medicine. New York, NY. Thank you for watching.
Arthur L. Caplan, PhD, has disclosed the following relevant financial relationships:
- Served as a director, officer, partner, employee, advisor, consultant, or trustee for: Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position)
- Serves as a contributing author and adviser for: Medscape
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the Division of Medical Ethics at the NYU Grossman School of Medicine.
Right now, there are 10 states and the District of Columbia that have had some version of medical assistance in dying approved and on the books. That basically means that about 20% of Americans have access where they live to a physician who can prescribe a lethal dose of medication to them if they’re terminally ill and can ingest the medication themselves. That leaves many Americans not covered by this kind of access to this kind of service.
Many of you watching this may live in states where it is legal, like Oregon, Washington, New Jersey, Colorado, and Hawaii. I know many doctors say, “I’m not going to do that.” It’s not something that anyone is compelling a doctor to do. For some Americans, access is not just about where they live but whether there is a doctor willing to participate with them in bringing about their accelerated death, knowing that they’re inevitably going to die.
There’s not much we can do about that. It’s up to the conscience of each physician as to what they’re comfortable with. Certainly, there are other things that can be done to extend the possibility of having this available.
One thing that’s taking place is that, after lawsuits were filed, Vermont and Oregon have given up on their residency requirement, so you don’t have to be there 6 months or a year in order to use this opportunity. It’s legal now to move to the state or visit the state, and as soon as you get there, sign up for this kind of end-of-life intervention.
New Jersey is also being sued. I’ll predict that every state that has a residency requirement, when sued in court, is going to lose because we’ve long recognized the right of Americans to seek out healthcare in the United States, wherever they want to go.
If some states have made this a legitimate medical procedure, courts are going to say you can’t restrict it only to state residents. If someone wants to use a service, they’re entitled to show up from another state or another place and use it. I’m not sure about foreign nationals, but I’m very sure that Americans can go state to state in search of legitimate medical procedures.
The other bills that are out there, however, are basically saying they want to emulate Oregon, Washington, and the other states and say that the terminally ill, with severe restrictions, are going to be able to get this service without going anywhere.
The restrictions include a diagnosis of terminal illness and that you have to be deemed mentally competent. You can’t use this if you have Alzheimer’s or severe depression. You have to make a request twice with a week or two in between to make sure that your request is authentic. And obviously, everyone is on board to make sure that you’re not being coerced or pushed somehow into requesting a somewhat earlier death than you would have experienced without having the availability of the pills.
You also have to take the pills yourself or be able to pull a switch so that you could use a feeding tube–type administration. If you can’t do that, say due to ALS, you’re not eligible to use medical aid in dying. It’s a pretty restricted intervention.
Many people who get pills after going through these restrictions in the states that permit it don’t use it. As many as one third say they like having it there as a safety valve or a parachute, but once they know they could end their life sooner, then they’re going to stick it out.
Should states make this legal? New York, Massachusetts, Florida, and many other states have bills that are moving through. I’m going to say yes. We’ve had Oregon and Washington since the late 1990s with medical aid in dying on the books. There doesn’t seem to be any evidence of pushing people to use this, of bias against the disabled, or bigotry against particular ethnic or racial groups being used to encourage people to end their life sooner.
I think it is an option that Americans want. I think it’s an option that makes some sense. I’m well aware that we also have to make sure that people know about hospice. In some of these states, medical aid in dying is offered as a part of hospice — not all, but a few. Not everybody wants hospice once they realize that they’re dying and that it is coming relatively soon. They may want to leave with family present, with a ceremony, or with a quality of life that they desire.
Past experience says let’s continue to expand availability in each state. Let’s also realize that we have to keep the restrictions in place on how it’s used because they have protected us against abuse. Let’s understand that every doctor has an option to do this or not do this. It’s a matter of conscience and a matter of comfort.
I think legalization is the direction we’re going to be going in. Getting rid of the residency requirements that have been around, as I think courts are going to overturn them, also gives a push to the idea that once the service is in this many states, it’s something that should be available if there are doctors willing to do it.
I’m Art Caplan at the Division of Medical Ethics at NYU Grossman School of Medicine. New York, NY. Thank you for watching.
Arthur L. Caplan, PhD, has disclosed the following relevant financial relationships:
- Served as a director, officer, partner, employee, advisor, consultant, or trustee for: Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position)
- Serves as a contributing author and adviser for: Medscape
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the Division of Medical Ethics at the NYU Grossman School of Medicine.
Right now, there are 10 states and the District of Columbia that have had some version of medical assistance in dying approved and on the books. That basically means that about 20% of Americans have access where they live to a physician who can prescribe a lethal dose of medication to them if they’re terminally ill and can ingest the medication themselves. That leaves many Americans not covered by this kind of access to this kind of service.
Many of you watching this may live in states where it is legal, like Oregon, Washington, New Jersey, Colorado, and Hawaii. I know many doctors say, “I’m not going to do that.” It’s not something that anyone is compelling a doctor to do. For some Americans, access is not just about where they live but whether there is a doctor willing to participate with them in bringing about their accelerated death, knowing that they’re inevitably going to die.
There’s not much we can do about that. It’s up to the conscience of each physician as to what they’re comfortable with. Certainly, there are other things that can be done to extend the possibility of having this available.
One thing that’s taking place is that, after lawsuits were filed, Vermont and Oregon have given up on their residency requirement, so you don’t have to be there 6 months or a year in order to use this opportunity. It’s legal now to move to the state or visit the state, and as soon as you get there, sign up for this kind of end-of-life intervention.
New Jersey is also being sued. I’ll predict that every state that has a residency requirement, when sued in court, is going to lose because we’ve long recognized the right of Americans to seek out healthcare in the United States, wherever they want to go.
If some states have made this a legitimate medical procedure, courts are going to say you can’t restrict it only to state residents. If someone wants to use a service, they’re entitled to show up from another state or another place and use it. I’m not sure about foreign nationals, but I’m very sure that Americans can go state to state in search of legitimate medical procedures.
The other bills that are out there, however, are basically saying they want to emulate Oregon, Washington, and the other states and say that the terminally ill, with severe restrictions, are going to be able to get this service without going anywhere.
The restrictions include a diagnosis of terminal illness and that you have to be deemed mentally competent. You can’t use this if you have Alzheimer’s or severe depression. You have to make a request twice with a week or two in between to make sure that your request is authentic. And obviously, everyone is on board to make sure that you’re not being coerced or pushed somehow into requesting a somewhat earlier death than you would have experienced without having the availability of the pills.
You also have to take the pills yourself or be able to pull a switch so that you could use a feeding tube–type administration. If you can’t do that, say due to ALS, you’re not eligible to use medical aid in dying. It’s a pretty restricted intervention.
Many people who get pills after going through these restrictions in the states that permit it don’t use it. As many as one third say they like having it there as a safety valve or a parachute, but once they know they could end their life sooner, then they’re going to stick it out.
Should states make this legal? New York, Massachusetts, Florida, and many other states have bills that are moving through. I’m going to say yes. We’ve had Oregon and Washington since the late 1990s with medical aid in dying on the books. There doesn’t seem to be any evidence of pushing people to use this, of bias against the disabled, or bigotry against particular ethnic or racial groups being used to encourage people to end their life sooner.
I think it is an option that Americans want. I think it’s an option that makes some sense. I’m well aware that we also have to make sure that people know about hospice. In some of these states, medical aid in dying is offered as a part of hospice — not all, but a few. Not everybody wants hospice once they realize that they’re dying and that it is coming relatively soon. They may want to leave with family present, with a ceremony, or with a quality of life that they desire.
Past experience says let’s continue to expand availability in each state. Let’s also realize that we have to keep the restrictions in place on how it’s used because they have protected us against abuse. Let’s understand that every doctor has an option to do this or not do this. It’s a matter of conscience and a matter of comfort.
I think legalization is the direction we’re going to be going in. Getting rid of the residency requirements that have been around, as I think courts are going to overturn them, also gives a push to the idea that once the service is in this many states, it’s something that should be available if there are doctors willing to do it.
I’m Art Caplan at the Division of Medical Ethics at NYU Grossman School of Medicine. New York, NY. Thank you for watching.
Arthur L. Caplan, PhD, has disclosed the following relevant financial relationships:
- Served as a director, officer, partner, employee, advisor, consultant, or trustee for: Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position)
- Serves as a contributing author and adviser for: Medscape
A version of this article appeared on Medscape.com.
Five Bold Predictions for Long COVID in 2024
With a number of large-scale clinical trials underway and researchers on the hunt for new therapies, long COVID scientists are hopeful that this is the year patients — and doctors who care for them — will finally see improvements in treating their symptoms.
Here are five bold predictions — all based on encouraging research — that could happen in 2024. At the very least, they are promising signs of progress against a debilitating and frustrating disease.
#1: We’ll gain a better understanding of each long COVID phenotype
This past year, a wide breadth of research began showing that long COVID can be defined by a number of different disease phenotypes that present a range of symptoms.
Researchers identified four clinical phenotypes: Chronic fatigue-like syndrome, headache, and memory loss; respiratory syndrome, which includes cough and difficulty breathing; chronic pain; and neurosensorial syndrome, which causes an altered sense of taste and smell.
Identifying specific diagnostic criteria for each phenotype would lead to better health outcomes for patients instead of treating them as if it were a “one-size-fits-all disease,” said Nisha Viswanathan, MD, director of the long COVID program at UCLA Health, Los Angeles, California.
Ultimately, she hopes that this year her patients will receive treatments based on the type of long COVID they’re personally experiencing, and the symptoms they have, leading to improved health outcomes and more rapid relief.
“Many new medications are focused on different pathways of long COVID, and the challenge becomes which drug is the right drug for each treatment,” said Dr. Viswanathan.
#2: Monoclonal antibodies may change the game
We’re starting to have a better understanding that what’s been called “viral persistence” as a main cause of long COVID may potentially be treated with monoclonal antibodies. These are antibodies produced by cloning unique white blood cells to target the circulating spike proteins in the blood that hang out in viral reservoirs and cause the immune system to react as if it’s still fighting acute COVID-19.
Smaller-scale studies have already shown promising results. A January 2024 study published in The American Journal of Emergency Medicine followed three patients who completely recovered from long COVID after taking monoclonal antibodies. “Remission occurred despite dissimilar past histories, sex, age, and illness duration,” wrote the study authors.
Larger clinical trials are underway at the University of California, San Francisco, California, to test targeted monoclonal antibodies. If the results of the larger study show that monoclonal antibodies are beneficial, then it could be a game changer for a large swath of patients around the world, said David F. Putrino, PhD, who runs the long COVID clinic at Mount Sinai Health System in New York City.
“The idea is that the downstream damage caused by viral persistence will resolve itself once you wipe out the virus,” said Dr. Putrino.
#3: Paxlovid could prove effective for long COVID
The US Food and Drug Administration granted approval for Paxlovid last May for the treatment of mild to moderate COVID-19 in adults at a high risk for severe disease. The medication is made up of two drugs packaged together. The first, nirmatrelvir, works by blocking a key enzyme required for virus replication. The second, ritonavir, is an antiviral that’s been used in patients with HIV and helps boost levels of antivirals in the body.
In a large-scale trial headed up by Dr. Putrino and his team, the oral antiviral is being studied for use in the post-viral stage in patients who test negative for acute COVID-19 but have persisting symptoms of long COVID.
Similar to monoclonal antibodies, the idea is to quell viral persistence. If patients have long COVID because they can’t clear SAR-CoV-2 from their bodies, Paxlovid could help. But unlike monoclonal antibodies that quash the virus, Paxlovid stops the virus from replicating. It’s a different mechanism with the same end goal.
It’s been a controversial treatment because it’s life-changing for some patients and ineffective for others. In addition, it can cause a range of side effects such as diarrhea, nausea, vomiting, and an impaired sense of taste. The goal of the trial is to see which patients with long COVID are most likely to benefit from the treatment.
#4: Anti-inflammatories like metformin could prove useful
Many of the inflammatory markers persistent in patients with long COVID were similarly present in patients with autoimmune diseases like rheumatoid arthritis, according to a July 2023 study published in JAMA.
The hope is that anti-inflammatory medications may be used to reduce inflammation causing long COVID symptoms. But drugs used to treat rheumatoid arthritis like abatacept and infliximabcan also have serious side effects, including increased risk for infection, flu-like symptoms, and burning of the skin.
“Powerful anti-inflammatories can change a number of pathways in the immune system,” said Grace McComsey, MD, who leads the long COVID RECOVER study at University Hospitals Health System in Cleveland, Ohio. Anti-inflammatories hold promise but, Dr. McComsey said, “some are more toxic with many side effects, so even if they work, there’s still a question about who should take them.”
Still, other anti-inflammatories that could work don’t have as many side effects. For example, a study published in The Lancet Infectious Diseases found that the diabetes drug metformin reduced a patient’s risk for long COVID up to 40% when the drug was taken during the acute stage.
Metformin, compared to other anti-inflammatories (also known as immune modulators), is an inexpensive and widely available drug with relatively few side effects compared with other medications.
#5: Serotonin levels — and selective serotonin reuptake inhibitors (SSRIs) — may be keys to unlocking long COVID
One of the most groundbreaking studies of the year came last November. A study published in the journal Cell found lower circulating serotonin levels in patents with long COVID than in those who did not have the condition. The study also found that the SSRI fluoxetine improved cognitive function in rat models infected with the virus.
Researchers found that the reduction in serotonin levels was partially caused by the body’s inability to absorb tryptophan, an amino acid that’s a precursor to serotonin. Overactivated blood platelets may also have played a role.
Michael Peluso, MD, an assistant research professor of infectious medicine at the UCSF School of Medicine, San Francisco, California, hopes to take the finding a step further, investigating whether increased serotonin levels in patients with long COVID will lead to improvements in symptoms.
“What we need now is a good clinical trial to see whether altering levels of serotonin in people with long COVID will lead to symptom relief,” Dr. Peluso said last month in an interview with this news organization.
If patients show an improvement in symptoms, then the next step is looking into whether SSRIs boost serotonin levels in patients and, as a result, reduce their symptoms.
A version of this article appeared on Medscape.com.
With a number of large-scale clinical trials underway and researchers on the hunt for new therapies, long COVID scientists are hopeful that this is the year patients — and doctors who care for them — will finally see improvements in treating their symptoms.
Here are five bold predictions — all based on encouraging research — that could happen in 2024. At the very least, they are promising signs of progress against a debilitating and frustrating disease.
#1: We’ll gain a better understanding of each long COVID phenotype
This past year, a wide breadth of research began showing that long COVID can be defined by a number of different disease phenotypes that present a range of symptoms.
Researchers identified four clinical phenotypes: Chronic fatigue-like syndrome, headache, and memory loss; respiratory syndrome, which includes cough and difficulty breathing; chronic pain; and neurosensorial syndrome, which causes an altered sense of taste and smell.
Identifying specific diagnostic criteria for each phenotype would lead to better health outcomes for patients instead of treating them as if it were a “one-size-fits-all disease,” said Nisha Viswanathan, MD, director of the long COVID program at UCLA Health, Los Angeles, California.
Ultimately, she hopes that this year her patients will receive treatments based on the type of long COVID they’re personally experiencing, and the symptoms they have, leading to improved health outcomes and more rapid relief.
“Many new medications are focused on different pathways of long COVID, and the challenge becomes which drug is the right drug for each treatment,” said Dr. Viswanathan.
#2: Monoclonal antibodies may change the game
We’re starting to have a better understanding that what’s been called “viral persistence” as a main cause of long COVID may potentially be treated with monoclonal antibodies. These are antibodies produced by cloning unique white blood cells to target the circulating spike proteins in the blood that hang out in viral reservoirs and cause the immune system to react as if it’s still fighting acute COVID-19.
Smaller-scale studies have already shown promising results. A January 2024 study published in The American Journal of Emergency Medicine followed three patients who completely recovered from long COVID after taking monoclonal antibodies. “Remission occurred despite dissimilar past histories, sex, age, and illness duration,” wrote the study authors.
Larger clinical trials are underway at the University of California, San Francisco, California, to test targeted monoclonal antibodies. If the results of the larger study show that monoclonal antibodies are beneficial, then it could be a game changer for a large swath of patients around the world, said David F. Putrino, PhD, who runs the long COVID clinic at Mount Sinai Health System in New York City.
“The idea is that the downstream damage caused by viral persistence will resolve itself once you wipe out the virus,” said Dr. Putrino.
#3: Paxlovid could prove effective for long COVID
The US Food and Drug Administration granted approval for Paxlovid last May for the treatment of mild to moderate COVID-19 in adults at a high risk for severe disease. The medication is made up of two drugs packaged together. The first, nirmatrelvir, works by blocking a key enzyme required for virus replication. The second, ritonavir, is an antiviral that’s been used in patients with HIV and helps boost levels of antivirals in the body.
In a large-scale trial headed up by Dr. Putrino and his team, the oral antiviral is being studied for use in the post-viral stage in patients who test negative for acute COVID-19 but have persisting symptoms of long COVID.
Similar to monoclonal antibodies, the idea is to quell viral persistence. If patients have long COVID because they can’t clear SAR-CoV-2 from their bodies, Paxlovid could help. But unlike monoclonal antibodies that quash the virus, Paxlovid stops the virus from replicating. It’s a different mechanism with the same end goal.
It’s been a controversial treatment because it’s life-changing for some patients and ineffective for others. In addition, it can cause a range of side effects such as diarrhea, nausea, vomiting, and an impaired sense of taste. The goal of the trial is to see which patients with long COVID are most likely to benefit from the treatment.
#4: Anti-inflammatories like metformin could prove useful
Many of the inflammatory markers persistent in patients with long COVID were similarly present in patients with autoimmune diseases like rheumatoid arthritis, according to a July 2023 study published in JAMA.
The hope is that anti-inflammatory medications may be used to reduce inflammation causing long COVID symptoms. But drugs used to treat rheumatoid arthritis like abatacept and infliximabcan also have serious side effects, including increased risk for infection, flu-like symptoms, and burning of the skin.
“Powerful anti-inflammatories can change a number of pathways in the immune system,” said Grace McComsey, MD, who leads the long COVID RECOVER study at University Hospitals Health System in Cleveland, Ohio. Anti-inflammatories hold promise but, Dr. McComsey said, “some are more toxic with many side effects, so even if they work, there’s still a question about who should take them.”
Still, other anti-inflammatories that could work don’t have as many side effects. For example, a study published in The Lancet Infectious Diseases found that the diabetes drug metformin reduced a patient’s risk for long COVID up to 40% when the drug was taken during the acute stage.
Metformin, compared to other anti-inflammatories (also known as immune modulators), is an inexpensive and widely available drug with relatively few side effects compared with other medications.
#5: Serotonin levels — and selective serotonin reuptake inhibitors (SSRIs) — may be keys to unlocking long COVID
One of the most groundbreaking studies of the year came last November. A study published in the journal Cell found lower circulating serotonin levels in patents with long COVID than in those who did not have the condition. The study also found that the SSRI fluoxetine improved cognitive function in rat models infected with the virus.
Researchers found that the reduction in serotonin levels was partially caused by the body’s inability to absorb tryptophan, an amino acid that’s a precursor to serotonin. Overactivated blood platelets may also have played a role.
Michael Peluso, MD, an assistant research professor of infectious medicine at the UCSF School of Medicine, San Francisco, California, hopes to take the finding a step further, investigating whether increased serotonin levels in patients with long COVID will lead to improvements in symptoms.
“What we need now is a good clinical trial to see whether altering levels of serotonin in people with long COVID will lead to symptom relief,” Dr. Peluso said last month in an interview with this news organization.
If patients show an improvement in symptoms, then the next step is looking into whether SSRIs boost serotonin levels in patients and, as a result, reduce their symptoms.
A version of this article appeared on Medscape.com.
With a number of large-scale clinical trials underway and researchers on the hunt for new therapies, long COVID scientists are hopeful that this is the year patients — and doctors who care for them — will finally see improvements in treating their symptoms.
Here are five bold predictions — all based on encouraging research — that could happen in 2024. At the very least, they are promising signs of progress against a debilitating and frustrating disease.
#1: We’ll gain a better understanding of each long COVID phenotype
This past year, a wide breadth of research began showing that long COVID can be defined by a number of different disease phenotypes that present a range of symptoms.
Researchers identified four clinical phenotypes: Chronic fatigue-like syndrome, headache, and memory loss; respiratory syndrome, which includes cough and difficulty breathing; chronic pain; and neurosensorial syndrome, which causes an altered sense of taste and smell.
Identifying specific diagnostic criteria for each phenotype would lead to better health outcomes for patients instead of treating them as if it were a “one-size-fits-all disease,” said Nisha Viswanathan, MD, director of the long COVID program at UCLA Health, Los Angeles, California.
Ultimately, she hopes that this year her patients will receive treatments based on the type of long COVID they’re personally experiencing, and the symptoms they have, leading to improved health outcomes and more rapid relief.
“Many new medications are focused on different pathways of long COVID, and the challenge becomes which drug is the right drug for each treatment,” said Dr. Viswanathan.
#2: Monoclonal antibodies may change the game
We’re starting to have a better understanding that what’s been called “viral persistence” as a main cause of long COVID may potentially be treated with monoclonal antibodies. These are antibodies produced by cloning unique white blood cells to target the circulating spike proteins in the blood that hang out in viral reservoirs and cause the immune system to react as if it’s still fighting acute COVID-19.
Smaller-scale studies have already shown promising results. A January 2024 study published in The American Journal of Emergency Medicine followed three patients who completely recovered from long COVID after taking monoclonal antibodies. “Remission occurred despite dissimilar past histories, sex, age, and illness duration,” wrote the study authors.
Larger clinical trials are underway at the University of California, San Francisco, California, to test targeted monoclonal antibodies. If the results of the larger study show that monoclonal antibodies are beneficial, then it could be a game changer for a large swath of patients around the world, said David F. Putrino, PhD, who runs the long COVID clinic at Mount Sinai Health System in New York City.
“The idea is that the downstream damage caused by viral persistence will resolve itself once you wipe out the virus,” said Dr. Putrino.
#3: Paxlovid could prove effective for long COVID
The US Food and Drug Administration granted approval for Paxlovid last May for the treatment of mild to moderate COVID-19 in adults at a high risk for severe disease. The medication is made up of two drugs packaged together. The first, nirmatrelvir, works by blocking a key enzyme required for virus replication. The second, ritonavir, is an antiviral that’s been used in patients with HIV and helps boost levels of antivirals in the body.
In a large-scale trial headed up by Dr. Putrino and his team, the oral antiviral is being studied for use in the post-viral stage in patients who test negative for acute COVID-19 but have persisting symptoms of long COVID.
Similar to monoclonal antibodies, the idea is to quell viral persistence. If patients have long COVID because they can’t clear SAR-CoV-2 from their bodies, Paxlovid could help. But unlike monoclonal antibodies that quash the virus, Paxlovid stops the virus from replicating. It’s a different mechanism with the same end goal.
It’s been a controversial treatment because it’s life-changing for some patients and ineffective for others. In addition, it can cause a range of side effects such as diarrhea, nausea, vomiting, and an impaired sense of taste. The goal of the trial is to see which patients with long COVID are most likely to benefit from the treatment.
#4: Anti-inflammatories like metformin could prove useful
Many of the inflammatory markers persistent in patients with long COVID were similarly present in patients with autoimmune diseases like rheumatoid arthritis, according to a July 2023 study published in JAMA.
The hope is that anti-inflammatory medications may be used to reduce inflammation causing long COVID symptoms. But drugs used to treat rheumatoid arthritis like abatacept and infliximabcan also have serious side effects, including increased risk for infection, flu-like symptoms, and burning of the skin.
“Powerful anti-inflammatories can change a number of pathways in the immune system,” said Grace McComsey, MD, who leads the long COVID RECOVER study at University Hospitals Health System in Cleveland, Ohio. Anti-inflammatories hold promise but, Dr. McComsey said, “some are more toxic with many side effects, so even if they work, there’s still a question about who should take them.”
Still, other anti-inflammatories that could work don’t have as many side effects. For example, a study published in The Lancet Infectious Diseases found that the diabetes drug metformin reduced a patient’s risk for long COVID up to 40% when the drug was taken during the acute stage.
Metformin, compared to other anti-inflammatories (also known as immune modulators), is an inexpensive and widely available drug with relatively few side effects compared with other medications.
#5: Serotonin levels — and selective serotonin reuptake inhibitors (SSRIs) — may be keys to unlocking long COVID
One of the most groundbreaking studies of the year came last November. A study published in the journal Cell found lower circulating serotonin levels in patents with long COVID than in those who did not have the condition. The study also found that the SSRI fluoxetine improved cognitive function in rat models infected with the virus.
Researchers found that the reduction in serotonin levels was partially caused by the body’s inability to absorb tryptophan, an amino acid that’s a precursor to serotonin. Overactivated blood platelets may also have played a role.
Michael Peluso, MD, an assistant research professor of infectious medicine at the UCSF School of Medicine, San Francisco, California, hopes to take the finding a step further, investigating whether increased serotonin levels in patients with long COVID will lead to improvements in symptoms.
“What we need now is a good clinical trial to see whether altering levels of serotonin in people with long COVID will lead to symptom relief,” Dr. Peluso said last month in an interview with this news organization.
If patients show an improvement in symptoms, then the next step is looking into whether SSRIs boost serotonin levels in patients and, as a result, reduce their symptoms.
A version of this article appeared on Medscape.com.
Think Outside the Traditional Toolbox to Treat Itch
ORLANDO — “Itch may not be as sexy as Mohs surgery or aesthetic procedures,” but treating it is important and meaningful to patients, particularly those who’ve found little relief previously, Shawn G. Kwatra, MD, said at the annual ODAC Dermatology, Aesthetic & Surgery Conference.
In doctors’ defense, it can be highly challenging to know which approach is optimal for each individual with pruritus, added Dr. Kwatra, associate professor of dermatology at Johns Hopkins University, Baltimore, Maryland.
Cooling agents, topical capsaicin, topical anesthetics like pramoxine 1%, various forms of lidocaine, strontium, opioid modulators like naltrexone, oral Janus kinase inhibitor (JAK) inhibitors, and medical marijuana are among some of the “outside the box” tools in Dr. Kwatra’s itch toolbox.
Often a Medical Puzzle
Frequently, patients come to the dermatologist complaining of itch, “but you don’t see much on their skin.” After a trial of antihistamines, and some topical steroids, the doctor might put up their hands and think: I tried, but I don’t know what else to do. “This actually happens a lot,” said Dr. Kwatra, who is also director of the Johns Hopkins Itch Center.
This means itch can frustrate providers as well. But for patients, the impact on their quality of life can be on the same level as recovering from a stroke or living with heart failure, Dr. Kwatra said. Finding relief for their itch is where “we can make a big difference for patients.”
Consider Cooling Agents
Many of these therapies are inexpensive and widely available. Cooling agents like menthol, camphor, or calamine can reduce activity of the transient receptor potential (TRP) channels in the skin associated with itch. This ion channel also senses temperature, pressure, and other sensations.
Another option is topical capsaicin, which works through the same ion channels. It binds to the TRPV1 receptors in sensory nerve fibers and causes desensitization. Initially, four to six applications a day are required to reduce itch. After that, patients can apply the medication less frequently. “You have to tell folks we know it’s going to work, but it’s going to burn a lot initially,” Dr. Kwatra said. “In real world practice, I’m not using it often.”
A 1.8% capsaicin patch, approved for treating postherpetic neuralgia, can be used to treat pruritus as well. “You put the patch on for one hour and you can have a true clinical response,” he noted.
Another option for itch relief, the topical anesthetic pramoxine 1%, “is probably underutilized for our patients,” Dr. Kwatra said. Pramoxine 1% works fast — as quickly as 2 minutes — and lasts up to 8 hours and is well-tolerated with low toxicity, he added. The agent is applied three to four times a day and relieves itch by reducing the transmembrane permeability of sodium ions on the skin. “This is something widely available and cheap.”
Lidocaine, another topical anesthetic, is available compounded, over the counter, and as a spray or patch. “I would be careful before you use high doses, like 10%” because of tolerability issues, Dr. Kwatra cautioned. He generally starts with lower concentrations.
Topical strontium is really interesting as a strategy, Dr. Kwatra said. Strontium is a soft, white metal that competes with calcium for receptor binding. There are over-the-counter formulations available as a scalp solution or lotion, which, he said, “are ways to go with more episodic itching.”
Topical oatmeal can also relieve itch in some patients. “There is actually some good scientific evidence for topical oatmeal preparations,” he said.
Steroid-Sparing Novel Topicals
Topical ruxolitinib (a JAK inhibitor approved for atopic dermatitis and vitiligo); topical roflumilast (a phosphodiesterase-4 inhibitor) and topical tapinarof (an aryl hydrocarbon receptor agonist), both approved for treating psoriasis; and the atopic dermatitis drug crisaborole fall into this category of topicals with potential for treating itch, he said, noting that use for treating itch is off label.
Off-label use of biologic agents are also possible treatment options for itch, dupilumab and tralokinumab, both US Food and Drug Administration (FDA)–approved for treating atopic dermatitis. Emerging agents that may prove useful for treating itch include lebrikizumab, nemolizumab, amlitelimab, and rocatinlimab, he said.
In terms of oral therapies, the FDA has approved two oral JAK inhibitors for atopic dermatitis, abrocitinib and upadacitinib, which could prove useful for itch as an off-label indication, according to Dr. Kwatra.
Naltrexone Off Label
An emerging therapeutic concept for treating itch is using an opioid antagonist like naltrexone. Morphine causes more itch, so the theory is a reversal agent might help reduce it. The challenge is that naltrexone only comes as a 50 mg tablet, “and I find the high dose makes people nauseous and vomit,” he added.
Don’t Forget Devices
He referred to a “great paper” that he said has been “totally overlooked,” published in 2001, which evaluated a device that stimulates C fibers in the skin to reduce itch. In the study, 19 patients used the device to treat local areas 20 minutes daily for 5 weeks. Punch biopsies of the affected areas were taken at baseline and after treatment. Mean itch ratings decreased from 78% to 42%, and the number of immunoreactive nerve fibers in the epidermis decreased by 40% at the end of treatment.
“Electrical neurostimulation is better for localized pruritus. There is limited case series evidence, but it’s something to think about,” Dr. Kwatra said.
He and his colleagues also have a case study in press that explored the use of injected botulinum toxin to relieve recalcitrant, chronic itch in a 65-year-old man “who failed everything.”
Dr. Kwatra is a consultant or advisory board member for AbbVie, Amgen, Arcutis Biotherapeutics, ASLAN Pharmaceuticals, Cara Therapeutics, Castle Biosciences, Celldex Therapeutics, Galderma, Incyte Corporation, Johnson & Johnson, LEO Pharma, Novartis, Pfizer, Regeneron, and Sanofi.
A version of this article appeared on Medscape.com.
ORLANDO — “Itch may not be as sexy as Mohs surgery or aesthetic procedures,” but treating it is important and meaningful to patients, particularly those who’ve found little relief previously, Shawn G. Kwatra, MD, said at the annual ODAC Dermatology, Aesthetic & Surgery Conference.
In doctors’ defense, it can be highly challenging to know which approach is optimal for each individual with pruritus, added Dr. Kwatra, associate professor of dermatology at Johns Hopkins University, Baltimore, Maryland.
Cooling agents, topical capsaicin, topical anesthetics like pramoxine 1%, various forms of lidocaine, strontium, opioid modulators like naltrexone, oral Janus kinase inhibitor (JAK) inhibitors, and medical marijuana are among some of the “outside the box” tools in Dr. Kwatra’s itch toolbox.
Often a Medical Puzzle
Frequently, patients come to the dermatologist complaining of itch, “but you don’t see much on their skin.” After a trial of antihistamines, and some topical steroids, the doctor might put up their hands and think: I tried, but I don’t know what else to do. “This actually happens a lot,” said Dr. Kwatra, who is also director of the Johns Hopkins Itch Center.
This means itch can frustrate providers as well. But for patients, the impact on their quality of life can be on the same level as recovering from a stroke or living with heart failure, Dr. Kwatra said. Finding relief for their itch is where “we can make a big difference for patients.”
Consider Cooling Agents
Many of these therapies are inexpensive and widely available. Cooling agents like menthol, camphor, or calamine can reduce activity of the transient receptor potential (TRP) channels in the skin associated with itch. This ion channel also senses temperature, pressure, and other sensations.
Another option is topical capsaicin, which works through the same ion channels. It binds to the TRPV1 receptors in sensory nerve fibers and causes desensitization. Initially, four to six applications a day are required to reduce itch. After that, patients can apply the medication less frequently. “You have to tell folks we know it’s going to work, but it’s going to burn a lot initially,” Dr. Kwatra said. “In real world practice, I’m not using it often.”
A 1.8% capsaicin patch, approved for treating postherpetic neuralgia, can be used to treat pruritus as well. “You put the patch on for one hour and you can have a true clinical response,” he noted.
Another option for itch relief, the topical anesthetic pramoxine 1%, “is probably underutilized for our patients,” Dr. Kwatra said. Pramoxine 1% works fast — as quickly as 2 minutes — and lasts up to 8 hours and is well-tolerated with low toxicity, he added. The agent is applied three to four times a day and relieves itch by reducing the transmembrane permeability of sodium ions on the skin. “This is something widely available and cheap.”
Lidocaine, another topical anesthetic, is available compounded, over the counter, and as a spray or patch. “I would be careful before you use high doses, like 10%” because of tolerability issues, Dr. Kwatra cautioned. He generally starts with lower concentrations.
Topical strontium is really interesting as a strategy, Dr. Kwatra said. Strontium is a soft, white metal that competes with calcium for receptor binding. There are over-the-counter formulations available as a scalp solution or lotion, which, he said, “are ways to go with more episodic itching.”
Topical oatmeal can also relieve itch in some patients. “There is actually some good scientific evidence for topical oatmeal preparations,” he said.
Steroid-Sparing Novel Topicals
Topical ruxolitinib (a JAK inhibitor approved for atopic dermatitis and vitiligo); topical roflumilast (a phosphodiesterase-4 inhibitor) and topical tapinarof (an aryl hydrocarbon receptor agonist), both approved for treating psoriasis; and the atopic dermatitis drug crisaborole fall into this category of topicals with potential for treating itch, he said, noting that use for treating itch is off label.
Off-label use of biologic agents are also possible treatment options for itch, dupilumab and tralokinumab, both US Food and Drug Administration (FDA)–approved for treating atopic dermatitis. Emerging agents that may prove useful for treating itch include lebrikizumab, nemolizumab, amlitelimab, and rocatinlimab, he said.
In terms of oral therapies, the FDA has approved two oral JAK inhibitors for atopic dermatitis, abrocitinib and upadacitinib, which could prove useful for itch as an off-label indication, according to Dr. Kwatra.
Naltrexone Off Label
An emerging therapeutic concept for treating itch is using an opioid antagonist like naltrexone. Morphine causes more itch, so the theory is a reversal agent might help reduce it. The challenge is that naltrexone only comes as a 50 mg tablet, “and I find the high dose makes people nauseous and vomit,” he added.
Don’t Forget Devices
He referred to a “great paper” that he said has been “totally overlooked,” published in 2001, which evaluated a device that stimulates C fibers in the skin to reduce itch. In the study, 19 patients used the device to treat local areas 20 minutes daily for 5 weeks. Punch biopsies of the affected areas were taken at baseline and after treatment. Mean itch ratings decreased from 78% to 42%, and the number of immunoreactive nerve fibers in the epidermis decreased by 40% at the end of treatment.
“Electrical neurostimulation is better for localized pruritus. There is limited case series evidence, but it’s something to think about,” Dr. Kwatra said.
He and his colleagues also have a case study in press that explored the use of injected botulinum toxin to relieve recalcitrant, chronic itch in a 65-year-old man “who failed everything.”
Dr. Kwatra is a consultant or advisory board member for AbbVie, Amgen, Arcutis Biotherapeutics, ASLAN Pharmaceuticals, Cara Therapeutics, Castle Biosciences, Celldex Therapeutics, Galderma, Incyte Corporation, Johnson & Johnson, LEO Pharma, Novartis, Pfizer, Regeneron, and Sanofi.
A version of this article appeared on Medscape.com.
ORLANDO — “Itch may not be as sexy as Mohs surgery or aesthetic procedures,” but treating it is important and meaningful to patients, particularly those who’ve found little relief previously, Shawn G. Kwatra, MD, said at the annual ODAC Dermatology, Aesthetic & Surgery Conference.
In doctors’ defense, it can be highly challenging to know which approach is optimal for each individual with pruritus, added Dr. Kwatra, associate professor of dermatology at Johns Hopkins University, Baltimore, Maryland.
Cooling agents, topical capsaicin, topical anesthetics like pramoxine 1%, various forms of lidocaine, strontium, opioid modulators like naltrexone, oral Janus kinase inhibitor (JAK) inhibitors, and medical marijuana are among some of the “outside the box” tools in Dr. Kwatra’s itch toolbox.
Often a Medical Puzzle
Frequently, patients come to the dermatologist complaining of itch, “but you don’t see much on their skin.” After a trial of antihistamines, and some topical steroids, the doctor might put up their hands and think: I tried, but I don’t know what else to do. “This actually happens a lot,” said Dr. Kwatra, who is also director of the Johns Hopkins Itch Center.
This means itch can frustrate providers as well. But for patients, the impact on their quality of life can be on the same level as recovering from a stroke or living with heart failure, Dr. Kwatra said. Finding relief for their itch is where “we can make a big difference for patients.”
Consider Cooling Agents
Many of these therapies are inexpensive and widely available. Cooling agents like menthol, camphor, or calamine can reduce activity of the transient receptor potential (TRP) channels in the skin associated with itch. This ion channel also senses temperature, pressure, and other sensations.
Another option is topical capsaicin, which works through the same ion channels. It binds to the TRPV1 receptors in sensory nerve fibers and causes desensitization. Initially, four to six applications a day are required to reduce itch. After that, patients can apply the medication less frequently. “You have to tell folks we know it’s going to work, but it’s going to burn a lot initially,” Dr. Kwatra said. “In real world practice, I’m not using it often.”
A 1.8% capsaicin patch, approved for treating postherpetic neuralgia, can be used to treat pruritus as well. “You put the patch on for one hour and you can have a true clinical response,” he noted.
Another option for itch relief, the topical anesthetic pramoxine 1%, “is probably underutilized for our patients,” Dr. Kwatra said. Pramoxine 1% works fast — as quickly as 2 minutes — and lasts up to 8 hours and is well-tolerated with low toxicity, he added. The agent is applied three to four times a day and relieves itch by reducing the transmembrane permeability of sodium ions on the skin. “This is something widely available and cheap.”
Lidocaine, another topical anesthetic, is available compounded, over the counter, and as a spray or patch. “I would be careful before you use high doses, like 10%” because of tolerability issues, Dr. Kwatra cautioned. He generally starts with lower concentrations.
Topical strontium is really interesting as a strategy, Dr. Kwatra said. Strontium is a soft, white metal that competes with calcium for receptor binding. There are over-the-counter formulations available as a scalp solution or lotion, which, he said, “are ways to go with more episodic itching.”
Topical oatmeal can also relieve itch in some patients. “There is actually some good scientific evidence for topical oatmeal preparations,” he said.
Steroid-Sparing Novel Topicals
Topical ruxolitinib (a JAK inhibitor approved for atopic dermatitis and vitiligo); topical roflumilast (a phosphodiesterase-4 inhibitor) and topical tapinarof (an aryl hydrocarbon receptor agonist), both approved for treating psoriasis; and the atopic dermatitis drug crisaborole fall into this category of topicals with potential for treating itch, he said, noting that use for treating itch is off label.
Off-label use of biologic agents are also possible treatment options for itch, dupilumab and tralokinumab, both US Food and Drug Administration (FDA)–approved for treating atopic dermatitis. Emerging agents that may prove useful for treating itch include lebrikizumab, nemolizumab, amlitelimab, and rocatinlimab, he said.
In terms of oral therapies, the FDA has approved two oral JAK inhibitors for atopic dermatitis, abrocitinib and upadacitinib, which could prove useful for itch as an off-label indication, according to Dr. Kwatra.
Naltrexone Off Label
An emerging therapeutic concept for treating itch is using an opioid antagonist like naltrexone. Morphine causes more itch, so the theory is a reversal agent might help reduce it. The challenge is that naltrexone only comes as a 50 mg tablet, “and I find the high dose makes people nauseous and vomit,” he added.
Don’t Forget Devices
He referred to a “great paper” that he said has been “totally overlooked,” published in 2001, which evaluated a device that stimulates C fibers in the skin to reduce itch. In the study, 19 patients used the device to treat local areas 20 minutes daily for 5 weeks. Punch biopsies of the affected areas were taken at baseline and after treatment. Mean itch ratings decreased from 78% to 42%, and the number of immunoreactive nerve fibers in the epidermis decreased by 40% at the end of treatment.
“Electrical neurostimulation is better for localized pruritus. There is limited case series evidence, but it’s something to think about,” Dr. Kwatra said.
He and his colleagues also have a case study in press that explored the use of injected botulinum toxin to relieve recalcitrant, chronic itch in a 65-year-old man “who failed everything.”
Dr. Kwatra is a consultant or advisory board member for AbbVie, Amgen, Arcutis Biotherapeutics, ASLAN Pharmaceuticals, Cara Therapeutics, Castle Biosciences, Celldex Therapeutics, Galderma, Incyte Corporation, Johnson & Johnson, LEO Pharma, Novartis, Pfizer, Regeneron, and Sanofi.
A version of this article appeared on Medscape.com.
FROM ODAC 2024
Cutaneous lupus, dermatomyositis: Excitement growing around emerging therapies
ORLANDO, FLORIDA — Advances in treating medical conditions rarely emerge in a straight line. Oftentimes, progress comes in fits and starts, and therapies to treat cutaneous lupus erythematosus (CLE) and dermatomyositis are no exception.
Beyond approved treatments that deserve more attention, like belimumab, approved by the Food and Drug Administration (FDA) for systemic lupus erythematosus (SLE) in 2011, and Octagam 10%, an intravenous immune globulin (IVIG) preparation approved for dermatomyositis in 2021, anticipation is growing for emerging therapies and their potential to provide relief to patients, Anthony Fernandez, MD, PhD, said at the ODAC Dermatology, Aesthetic & Surgical Conference. The tyrosine kinase 2 (TYK2) inhibitor deucravacitinib, Janus kinase (JAK) inhibitors brepocitinib and baricitinib, and the monoclonal antibody anifrolumab, he noted, are prime examples.
“. In my opinion, this is the start of what will be the most exciting decade in the history of these two diseases,” said Dr. Fernandez, director of medical dermatology at the Cleveland Clinic.
Emerging Treatments for Cutaneous Lupus
Although SLE can involve many organ systems, the skin is one of the most affected. There are specific cutaneous lesions categorized as either acute cutaneous lupus, subacute cutaneous lupus, or chronic cutaneous lupus.
The oral TYK2 inhibitor deucravacitinib, for example, should be able to dampen interleukin responses in people with CLE, Dr. Fernandez said. Deucravacitinib was approved by the FDA to treat psoriasis in September 2022.
A phase 2 study published in 2023 focused on this agent for relief of systemic lupus. Improvements in cutaneous disease were a secondary endpoint. The trial demonstrated that the patients treated with deucravacitinib achieved a 56%-70% CLASI-50 response, depending on dosing, compared with a 17% response among those on placebo at week 48.
Based on the trial results, recruitment has begun for a phase 2 trial to evaluate deucravacitinib, compared with placebo, in patients with discoid and/or subacute cutaneous lupus. “This may be another medicine we have available to give to any of our patients with cutaneous lupus,” Dr. Fernandez said.
Anifrolumab Appears Promising
The FDA approval of anifrolumab, a type I interferon (IFN) receptor antagonist, for treating moderate to severe SLE in July 2021, for example, is good news for dermatologists and their patients, added Dr. Fernandez. “Almost immediately after approval, case studies showed marked improvement in patients with refractory cutaneous lupus.” While the therapy was approved for treating systemic lupus, it allows for off-label treatment of the cutaneous predominant form of the disease, he said.
Furthermore, the manufacturer of anifrolumab, AstraZeneca, is launching the LAVENDER clinical trial to assess the monoclonal antibody specifically for treating CLE. “This is a big deal because we may be able to prescribe anifrolumab for our cutaneous lupus patients who don’t have systemic lupus,” Dr. Fernandez said.
Phase 3 data supported use the of anifrolumab in systemic lupus, including the TULIP-2 trial, which demonstrated its superiority to placebo for reducing severity of systemic disease and lowering corticosteroid use. A study published in March 2023 of 11 patients showed that they had a “very fast response” to the agent, Dr. Fernandez said, with a 50% or greater improvement in the Cutaneous Lupus Erythematosus Disease Area and Severity Index activity score reached by all participants at week 16. Improvements of 50% or more in this scoring system are considered clinically meaningful, he added.
Upcoming Dermatomyositis Treatments
Why highlight emerging therapies for CLE and dermatomyositis in the same ODAC presentation? Although distinct conditions, these autoimmune conditions are both mediated by type 1 IFN inflammation.
Dermatomyositis is a relatively rare immune-mediated disease that most commonly affects the skin and muscle. Doctors score disease presentation, activity, and clinical improvements on a scale similar to CLASI for cutaneous lupus, the CDASI or Cutaneous Dermatomyositis Disease Area and Severity Index. Among people with CDASI activity scores of at least 14, which is the threshold for moderate to severe disease, a 20% improvement is clinically meaningful, Dr. Fernandez said. In addition, a 40% or greater improvement correlates with significant improvements in quality of life.
There is now more evidence for the use of IVIG to treat dermatomyositis. “Among those of us who treat dermatomyositis on a regular basis, we believe IVIG is the most potent treatment. We’ve known that for a long time,” Dr. Fernandez said.
Despite this tenet, for years, there was only one placebo-controlled trial, published in 1993, that evaluated IVIG treatment for dermatomyositis, and it included only 15 participants. That was until October 2022, he said, when the New England Journal of Medicine published a study comparing a specific brand of IVIG (Octagam) with placebo in 95 people with dermatomyositis.
In the study, 79% of participants treated with IVIG had a total improvement score of at least 20 (minimal improvement), the primary endpoint, at 16 weeks, compared with 44% of those receiving a placebo. Those treated with IVIG also had significant improvements in the CDASI score, a secondary endpoint, compared with those on placebo, he said.
Based on results of this trial, the FDA approved Octagam 10% for dermatomyositis in adults. Dr. Fernandez noted the approval is restricted to the brand of IVIG in the trial, not all IVIG products. However, “the FDA approval is most important to us because it gives us ammunition to fight for insurers to approve IVIG when we feel our patients with dermatomyositis need it,” regardless of the brand.
The Potential of JAK1 Inhibitors
An open-label study of the JAK inhibitor tofacitinib, published in December 2020, showed that mean changes in CDASI activity scores at 12 weeks were statistically significant compared with baseline in 10 people with dermatomyositis. “The importance of this study is that it is proof of concept that JAK inhibition can be effective for treating dermatomyositis, especially with active skin disease,” Dr. Fernandez said.
In addition, two large phase 3 trials are evaluating JAK inhibitor safety and efficacy for treating dermatomyositis. One is the VALOR trial, currently recruiting people with recalcitrant dermatomyositis to evaluate treatment with brepocitinib. Researchers in France are looking at another JAK inhibitor, baricitinib, for treating relapsing or treatment-naive dermatomyositis. Recruitment for the BIRD clinical trial is ongoing.
Monoclonal Antibody Showing Promise
“When it comes to looking specifically at dermatomyositis cutaneous disease, it’s been found that the levels of IFN beta correlate best with not only lesional skin type 1 IFN inflammatory signatures but also overall clinical disease activity,” Dr. Fernandez said. This correlation is stronger than for any other IFN-1-type cytokine active in the disorder.
“Perhaps blocking IFN beta might be best way to get control of dermatomyositis activity,” he added.
With that in mind, a phase 2 trial of dazukibart presented at the American Academy of Dermatology 2023 annual meeting highlighted the promise of this agent that targets type 1 IFN beta.
The primary endpoint was improvement in CDASI at 12 weeks. “This medication has remarkable efficacy,” Dr. Fernandez said. “We were one of the sites for this trial. Despite being blinded, there was no question about who was receiving drug and who was receiving placebo.”
“A minimal clinical improvement in disease activity was seen in more than 90%, so almost every patient who received this medication had meaningful improvement,” he added.
Based on the results, the manufacturer, Pfizer, is recruiting participants for a phase 3 trial to further assess dazukibart in dermatomyositis and polymyositis. Dr. Fernandez said, “This is a story you should pay attention to if you treat any dermatomyositis patients at all.”
Regarding these emerging therapies for CLE and dermatomyositis, “This looks very much like the early days of psoriasis, in the early 2000s, when there was a lot of activity developing treatments,” Dr. Fernandez said. “I will predict that within 10 years, we will have multiple novel agents available that will probably work better than anything we have today.”
Dr. Fernandez reported receiving grant and/or research support from Alexion, Incyte, Mallinckrodt Pharmaceuticals, Novartis, Pfizer, and Priovant Therapeutics; acting as a consultant or advisory board member for AbbVie, Biogen, Mallinckrodt Pharmaceuticals; and being a member of the speaker bureau or receiving honoraria for non-CME from AbbVie, Kyowa Kirin, and Mallinckrodt Pharmaceuticals.
A version of this article appeared on Medscape.com.
ORLANDO, FLORIDA — Advances in treating medical conditions rarely emerge in a straight line. Oftentimes, progress comes in fits and starts, and therapies to treat cutaneous lupus erythematosus (CLE) and dermatomyositis are no exception.
Beyond approved treatments that deserve more attention, like belimumab, approved by the Food and Drug Administration (FDA) for systemic lupus erythematosus (SLE) in 2011, and Octagam 10%, an intravenous immune globulin (IVIG) preparation approved for dermatomyositis in 2021, anticipation is growing for emerging therapies and their potential to provide relief to patients, Anthony Fernandez, MD, PhD, said at the ODAC Dermatology, Aesthetic & Surgical Conference. The tyrosine kinase 2 (TYK2) inhibitor deucravacitinib, Janus kinase (JAK) inhibitors brepocitinib and baricitinib, and the monoclonal antibody anifrolumab, he noted, are prime examples.
“. In my opinion, this is the start of what will be the most exciting decade in the history of these two diseases,” said Dr. Fernandez, director of medical dermatology at the Cleveland Clinic.
Emerging Treatments for Cutaneous Lupus
Although SLE can involve many organ systems, the skin is one of the most affected. There are specific cutaneous lesions categorized as either acute cutaneous lupus, subacute cutaneous lupus, or chronic cutaneous lupus.
The oral TYK2 inhibitor deucravacitinib, for example, should be able to dampen interleukin responses in people with CLE, Dr. Fernandez said. Deucravacitinib was approved by the FDA to treat psoriasis in September 2022.
A phase 2 study published in 2023 focused on this agent for relief of systemic lupus. Improvements in cutaneous disease were a secondary endpoint. The trial demonstrated that the patients treated with deucravacitinib achieved a 56%-70% CLASI-50 response, depending on dosing, compared with a 17% response among those on placebo at week 48.
Based on the trial results, recruitment has begun for a phase 2 trial to evaluate deucravacitinib, compared with placebo, in patients with discoid and/or subacute cutaneous lupus. “This may be another medicine we have available to give to any of our patients with cutaneous lupus,” Dr. Fernandez said.
Anifrolumab Appears Promising
The FDA approval of anifrolumab, a type I interferon (IFN) receptor antagonist, for treating moderate to severe SLE in July 2021, for example, is good news for dermatologists and their patients, added Dr. Fernandez. “Almost immediately after approval, case studies showed marked improvement in patients with refractory cutaneous lupus.” While the therapy was approved for treating systemic lupus, it allows for off-label treatment of the cutaneous predominant form of the disease, he said.
Furthermore, the manufacturer of anifrolumab, AstraZeneca, is launching the LAVENDER clinical trial to assess the monoclonal antibody specifically for treating CLE. “This is a big deal because we may be able to prescribe anifrolumab for our cutaneous lupus patients who don’t have systemic lupus,” Dr. Fernandez said.
Phase 3 data supported use the of anifrolumab in systemic lupus, including the TULIP-2 trial, which demonstrated its superiority to placebo for reducing severity of systemic disease and lowering corticosteroid use. A study published in March 2023 of 11 patients showed that they had a “very fast response” to the agent, Dr. Fernandez said, with a 50% or greater improvement in the Cutaneous Lupus Erythematosus Disease Area and Severity Index activity score reached by all participants at week 16. Improvements of 50% or more in this scoring system are considered clinically meaningful, he added.
Upcoming Dermatomyositis Treatments
Why highlight emerging therapies for CLE and dermatomyositis in the same ODAC presentation? Although distinct conditions, these autoimmune conditions are both mediated by type 1 IFN inflammation.
Dermatomyositis is a relatively rare immune-mediated disease that most commonly affects the skin and muscle. Doctors score disease presentation, activity, and clinical improvements on a scale similar to CLASI for cutaneous lupus, the CDASI or Cutaneous Dermatomyositis Disease Area and Severity Index. Among people with CDASI activity scores of at least 14, which is the threshold for moderate to severe disease, a 20% improvement is clinically meaningful, Dr. Fernandez said. In addition, a 40% or greater improvement correlates with significant improvements in quality of life.
There is now more evidence for the use of IVIG to treat dermatomyositis. “Among those of us who treat dermatomyositis on a regular basis, we believe IVIG is the most potent treatment. We’ve known that for a long time,” Dr. Fernandez said.
Despite this tenet, for years, there was only one placebo-controlled trial, published in 1993, that evaluated IVIG treatment for dermatomyositis, and it included only 15 participants. That was until October 2022, he said, when the New England Journal of Medicine published a study comparing a specific brand of IVIG (Octagam) with placebo in 95 people with dermatomyositis.
In the study, 79% of participants treated with IVIG had a total improvement score of at least 20 (minimal improvement), the primary endpoint, at 16 weeks, compared with 44% of those receiving a placebo. Those treated with IVIG also had significant improvements in the CDASI score, a secondary endpoint, compared with those on placebo, he said.
Based on results of this trial, the FDA approved Octagam 10% for dermatomyositis in adults. Dr. Fernandez noted the approval is restricted to the brand of IVIG in the trial, not all IVIG products. However, “the FDA approval is most important to us because it gives us ammunition to fight for insurers to approve IVIG when we feel our patients with dermatomyositis need it,” regardless of the brand.
The Potential of JAK1 Inhibitors
An open-label study of the JAK inhibitor tofacitinib, published in December 2020, showed that mean changes in CDASI activity scores at 12 weeks were statistically significant compared with baseline in 10 people with dermatomyositis. “The importance of this study is that it is proof of concept that JAK inhibition can be effective for treating dermatomyositis, especially with active skin disease,” Dr. Fernandez said.
In addition, two large phase 3 trials are evaluating JAK inhibitor safety and efficacy for treating dermatomyositis. One is the VALOR trial, currently recruiting people with recalcitrant dermatomyositis to evaluate treatment with brepocitinib. Researchers in France are looking at another JAK inhibitor, baricitinib, for treating relapsing or treatment-naive dermatomyositis. Recruitment for the BIRD clinical trial is ongoing.
Monoclonal Antibody Showing Promise
“When it comes to looking specifically at dermatomyositis cutaneous disease, it’s been found that the levels of IFN beta correlate best with not only lesional skin type 1 IFN inflammatory signatures but also overall clinical disease activity,” Dr. Fernandez said. This correlation is stronger than for any other IFN-1-type cytokine active in the disorder.
“Perhaps blocking IFN beta might be best way to get control of dermatomyositis activity,” he added.
With that in mind, a phase 2 trial of dazukibart presented at the American Academy of Dermatology 2023 annual meeting highlighted the promise of this agent that targets type 1 IFN beta.
The primary endpoint was improvement in CDASI at 12 weeks. “This medication has remarkable efficacy,” Dr. Fernandez said. “We were one of the sites for this trial. Despite being blinded, there was no question about who was receiving drug and who was receiving placebo.”
“A minimal clinical improvement in disease activity was seen in more than 90%, so almost every patient who received this medication had meaningful improvement,” he added.
Based on the results, the manufacturer, Pfizer, is recruiting participants for a phase 3 trial to further assess dazukibart in dermatomyositis and polymyositis. Dr. Fernandez said, “This is a story you should pay attention to if you treat any dermatomyositis patients at all.”
Regarding these emerging therapies for CLE and dermatomyositis, “This looks very much like the early days of psoriasis, in the early 2000s, when there was a lot of activity developing treatments,” Dr. Fernandez said. “I will predict that within 10 years, we will have multiple novel agents available that will probably work better than anything we have today.”
Dr. Fernandez reported receiving grant and/or research support from Alexion, Incyte, Mallinckrodt Pharmaceuticals, Novartis, Pfizer, and Priovant Therapeutics; acting as a consultant or advisory board member for AbbVie, Biogen, Mallinckrodt Pharmaceuticals; and being a member of the speaker bureau or receiving honoraria for non-CME from AbbVie, Kyowa Kirin, and Mallinckrodt Pharmaceuticals.
A version of this article appeared on Medscape.com.
ORLANDO, FLORIDA — Advances in treating medical conditions rarely emerge in a straight line. Oftentimes, progress comes in fits and starts, and therapies to treat cutaneous lupus erythematosus (CLE) and dermatomyositis are no exception.
Beyond approved treatments that deserve more attention, like belimumab, approved by the Food and Drug Administration (FDA) for systemic lupus erythematosus (SLE) in 2011, and Octagam 10%, an intravenous immune globulin (IVIG) preparation approved for dermatomyositis in 2021, anticipation is growing for emerging therapies and their potential to provide relief to patients, Anthony Fernandez, MD, PhD, said at the ODAC Dermatology, Aesthetic & Surgical Conference. The tyrosine kinase 2 (TYK2) inhibitor deucravacitinib, Janus kinase (JAK) inhibitors brepocitinib and baricitinib, and the monoclonal antibody anifrolumab, he noted, are prime examples.
“. In my opinion, this is the start of what will be the most exciting decade in the history of these two diseases,” said Dr. Fernandez, director of medical dermatology at the Cleveland Clinic.
Emerging Treatments for Cutaneous Lupus
Although SLE can involve many organ systems, the skin is one of the most affected. There are specific cutaneous lesions categorized as either acute cutaneous lupus, subacute cutaneous lupus, or chronic cutaneous lupus.
The oral TYK2 inhibitor deucravacitinib, for example, should be able to dampen interleukin responses in people with CLE, Dr. Fernandez said. Deucravacitinib was approved by the FDA to treat psoriasis in September 2022.
A phase 2 study published in 2023 focused on this agent for relief of systemic lupus. Improvements in cutaneous disease were a secondary endpoint. The trial demonstrated that the patients treated with deucravacitinib achieved a 56%-70% CLASI-50 response, depending on dosing, compared with a 17% response among those on placebo at week 48.
Based on the trial results, recruitment has begun for a phase 2 trial to evaluate deucravacitinib, compared with placebo, in patients with discoid and/or subacute cutaneous lupus. “This may be another medicine we have available to give to any of our patients with cutaneous lupus,” Dr. Fernandez said.
Anifrolumab Appears Promising
The FDA approval of anifrolumab, a type I interferon (IFN) receptor antagonist, for treating moderate to severe SLE in July 2021, for example, is good news for dermatologists and their patients, added Dr. Fernandez. “Almost immediately after approval, case studies showed marked improvement in patients with refractory cutaneous lupus.” While the therapy was approved for treating systemic lupus, it allows for off-label treatment of the cutaneous predominant form of the disease, he said.
Furthermore, the manufacturer of anifrolumab, AstraZeneca, is launching the LAVENDER clinical trial to assess the monoclonal antibody specifically for treating CLE. “This is a big deal because we may be able to prescribe anifrolumab for our cutaneous lupus patients who don’t have systemic lupus,” Dr. Fernandez said.
Phase 3 data supported use the of anifrolumab in systemic lupus, including the TULIP-2 trial, which demonstrated its superiority to placebo for reducing severity of systemic disease and lowering corticosteroid use. A study published in March 2023 of 11 patients showed that they had a “very fast response” to the agent, Dr. Fernandez said, with a 50% or greater improvement in the Cutaneous Lupus Erythematosus Disease Area and Severity Index activity score reached by all participants at week 16. Improvements of 50% or more in this scoring system are considered clinically meaningful, he added.
Upcoming Dermatomyositis Treatments
Why highlight emerging therapies for CLE and dermatomyositis in the same ODAC presentation? Although distinct conditions, these autoimmune conditions are both mediated by type 1 IFN inflammation.
Dermatomyositis is a relatively rare immune-mediated disease that most commonly affects the skin and muscle. Doctors score disease presentation, activity, and clinical improvements on a scale similar to CLASI for cutaneous lupus, the CDASI or Cutaneous Dermatomyositis Disease Area and Severity Index. Among people with CDASI activity scores of at least 14, which is the threshold for moderate to severe disease, a 20% improvement is clinically meaningful, Dr. Fernandez said. In addition, a 40% or greater improvement correlates with significant improvements in quality of life.
There is now more evidence for the use of IVIG to treat dermatomyositis. “Among those of us who treat dermatomyositis on a regular basis, we believe IVIG is the most potent treatment. We’ve known that for a long time,” Dr. Fernandez said.
Despite this tenet, for years, there was only one placebo-controlled trial, published in 1993, that evaluated IVIG treatment for dermatomyositis, and it included only 15 participants. That was until October 2022, he said, when the New England Journal of Medicine published a study comparing a specific brand of IVIG (Octagam) with placebo in 95 people with dermatomyositis.
In the study, 79% of participants treated with IVIG had a total improvement score of at least 20 (minimal improvement), the primary endpoint, at 16 weeks, compared with 44% of those receiving a placebo. Those treated with IVIG also had significant improvements in the CDASI score, a secondary endpoint, compared with those on placebo, he said.
Based on results of this trial, the FDA approved Octagam 10% for dermatomyositis in adults. Dr. Fernandez noted the approval is restricted to the brand of IVIG in the trial, not all IVIG products. However, “the FDA approval is most important to us because it gives us ammunition to fight for insurers to approve IVIG when we feel our patients with dermatomyositis need it,” regardless of the brand.
The Potential of JAK1 Inhibitors
An open-label study of the JAK inhibitor tofacitinib, published in December 2020, showed that mean changes in CDASI activity scores at 12 weeks were statistically significant compared with baseline in 10 people with dermatomyositis. “The importance of this study is that it is proof of concept that JAK inhibition can be effective for treating dermatomyositis, especially with active skin disease,” Dr. Fernandez said.
In addition, two large phase 3 trials are evaluating JAK inhibitor safety and efficacy for treating dermatomyositis. One is the VALOR trial, currently recruiting people with recalcitrant dermatomyositis to evaluate treatment with brepocitinib. Researchers in France are looking at another JAK inhibitor, baricitinib, for treating relapsing or treatment-naive dermatomyositis. Recruitment for the BIRD clinical trial is ongoing.
Monoclonal Antibody Showing Promise
“When it comes to looking specifically at dermatomyositis cutaneous disease, it’s been found that the levels of IFN beta correlate best with not only lesional skin type 1 IFN inflammatory signatures but also overall clinical disease activity,” Dr. Fernandez said. This correlation is stronger than for any other IFN-1-type cytokine active in the disorder.
“Perhaps blocking IFN beta might be best way to get control of dermatomyositis activity,” he added.
With that in mind, a phase 2 trial of dazukibart presented at the American Academy of Dermatology 2023 annual meeting highlighted the promise of this agent that targets type 1 IFN beta.
The primary endpoint was improvement in CDASI at 12 weeks. “This medication has remarkable efficacy,” Dr. Fernandez said. “We were one of the sites for this trial. Despite being blinded, there was no question about who was receiving drug and who was receiving placebo.”
“A minimal clinical improvement in disease activity was seen in more than 90%, so almost every patient who received this medication had meaningful improvement,” he added.
Based on the results, the manufacturer, Pfizer, is recruiting participants for a phase 3 trial to further assess dazukibart in dermatomyositis and polymyositis. Dr. Fernandez said, “This is a story you should pay attention to if you treat any dermatomyositis patients at all.”
Regarding these emerging therapies for CLE and dermatomyositis, “This looks very much like the early days of psoriasis, in the early 2000s, when there was a lot of activity developing treatments,” Dr. Fernandez said. “I will predict that within 10 years, we will have multiple novel agents available that will probably work better than anything we have today.”
Dr. Fernandez reported receiving grant and/or research support from Alexion, Incyte, Mallinckrodt Pharmaceuticals, Novartis, Pfizer, and Priovant Therapeutics; acting as a consultant or advisory board member for AbbVie, Biogen, Mallinckrodt Pharmaceuticals; and being a member of the speaker bureau or receiving honoraria for non-CME from AbbVie, Kyowa Kirin, and Mallinckrodt Pharmaceuticals.
A version of this article appeared on Medscape.com.
FROM ODAC 2024
Dana-Farber Moves to Retract, Correct Dozens of Cancer Papers Amid Allegations
News of the investigation follows a blog post by British molecular biologist Sholto David, MD, who flagged almost 60 papers published between 1997 and 2017 that contained image manipulation and other errors. Some of the papers were published by Dana-Farber’s chief executive officer, Laurie Glimcher, MD, and chief operating officer, William Hahn, MD, on topics including multiple myeloma and immune cells.
Mr. David, who blogs about research integrity, highlighted numerous errors and irregularities, including copying and pasting images across multiple experiments to represent different days within the same experiment, sometimes rotating or stretching images.
In one case, Mr. David equated the manipulation with tactics used by “hapless Chinese papermills” and concluded that “a swathe of research coming out of [Dana-Farber] authored by the most senior researchers and managers appears to be hopelessly corrupt with errors that are obvious from just a cursory reading the papers.”
“Imagine what mistakes might be found in the raw data if anyone was allowed to look!” he wrote.
Barrett Rollins, MD, PhD, Dana-Farber Cancer Institute’s research integrity officer, declined to comment on whether the errors represent scientific misconduct, according to STAT. Rollins told ScienceInsider that the “presence of image discrepancies in a paper is not evidence of an author’s intent to deceive.”
Access to new artificial intelligence tools is making it easier for data sleuths, like Mr. David, to unearth data manipulation and errors.
The current investigation closely follows two other investigations into the published work of Harvard University’s former president, Claudine Gay, and Stanford University’s former president, Marc Tessier-Lavigne, which led both to resign their posts.
A version of this article appeared on Medscape.com.
News of the investigation follows a blog post by British molecular biologist Sholto David, MD, who flagged almost 60 papers published between 1997 and 2017 that contained image manipulation and other errors. Some of the papers were published by Dana-Farber’s chief executive officer, Laurie Glimcher, MD, and chief operating officer, William Hahn, MD, on topics including multiple myeloma and immune cells.
Mr. David, who blogs about research integrity, highlighted numerous errors and irregularities, including copying and pasting images across multiple experiments to represent different days within the same experiment, sometimes rotating or stretching images.
In one case, Mr. David equated the manipulation with tactics used by “hapless Chinese papermills” and concluded that “a swathe of research coming out of [Dana-Farber] authored by the most senior researchers and managers appears to be hopelessly corrupt with errors that are obvious from just a cursory reading the papers.”
“Imagine what mistakes might be found in the raw data if anyone was allowed to look!” he wrote.
Barrett Rollins, MD, PhD, Dana-Farber Cancer Institute’s research integrity officer, declined to comment on whether the errors represent scientific misconduct, according to STAT. Rollins told ScienceInsider that the “presence of image discrepancies in a paper is not evidence of an author’s intent to deceive.”
Access to new artificial intelligence tools is making it easier for data sleuths, like Mr. David, to unearth data manipulation and errors.
The current investigation closely follows two other investigations into the published work of Harvard University’s former president, Claudine Gay, and Stanford University’s former president, Marc Tessier-Lavigne, which led both to resign their posts.
A version of this article appeared on Medscape.com.
News of the investigation follows a blog post by British molecular biologist Sholto David, MD, who flagged almost 60 papers published between 1997 and 2017 that contained image manipulation and other errors. Some of the papers were published by Dana-Farber’s chief executive officer, Laurie Glimcher, MD, and chief operating officer, William Hahn, MD, on topics including multiple myeloma and immune cells.
Mr. David, who blogs about research integrity, highlighted numerous errors and irregularities, including copying and pasting images across multiple experiments to represent different days within the same experiment, sometimes rotating or stretching images.
In one case, Mr. David equated the manipulation with tactics used by “hapless Chinese papermills” and concluded that “a swathe of research coming out of [Dana-Farber] authored by the most senior researchers and managers appears to be hopelessly corrupt with errors that are obvious from just a cursory reading the papers.”
“Imagine what mistakes might be found in the raw data if anyone was allowed to look!” he wrote.
Barrett Rollins, MD, PhD, Dana-Farber Cancer Institute’s research integrity officer, declined to comment on whether the errors represent scientific misconduct, according to STAT. Rollins told ScienceInsider that the “presence of image discrepancies in a paper is not evidence of an author’s intent to deceive.”
Access to new artificial intelligence tools is making it easier for data sleuths, like Mr. David, to unearth data manipulation and errors.
The current investigation closely follows two other investigations into the published work of Harvard University’s former president, Claudine Gay, and Stanford University’s former president, Marc Tessier-Lavigne, which led both to resign their posts.
A version of this article appeared on Medscape.com.
Even Intentional Weight Loss Linked With Cancer
This transcript has been edited for clarity.
As anyone who has been through medical training will tell you, some little scenes just stick with you. I had been seeing a patient in our resident clinic in West Philly for a couple of years. She was in her mid-60s with diabetes and hypertension and a distant smoking history. She was overweight and had been trying to improve her diet and lose weight since I started seeing her. One day she came in and was delighted to report that she had finally started shedding some pounds — about 15 in the past 2 months.
I enthusiastically told my preceptor that my careful dietary counseling had finally done the job. She looked through the chart for a moment and asked, “Is she up to date on her cancer screening?” A workup revealed adenocarcinoma of the lung. The patient did well, actually, but the story stuck with me.
The textbooks call it “unintentional weight loss,” often in big, scary letters, and every doctor will go just a bit pale if a patient tells them that, despite efforts not to, they are losing weight. But true unintentional weight loss is not that common. After all, most of us are at least half-heartedly trying to lose weight all the time. Should doctors be worried when we are successful?
A new study suggests that perhaps they should. We’re talking about this study, appearing in JAMA, which combined participants from two long-running observational cohorts: 120,000 women from the Nurses’ Health Study, and 50,000 men from the Health Professionals Follow-Up Study. (These cohorts started in the 1970s and 1980s, so we’ll give them a pass on the gender-specific study designs.)
The rationale of enrolling healthcare providers in these cohort studies is that they would be reliable witnesses of their own health status. If a nurse or doctor says they have pancreatic cancer, it’s likely that they truly have pancreatic cancer. Detailed health surveys were distributed to the participants every other year, and the average follow-up was more than a decade.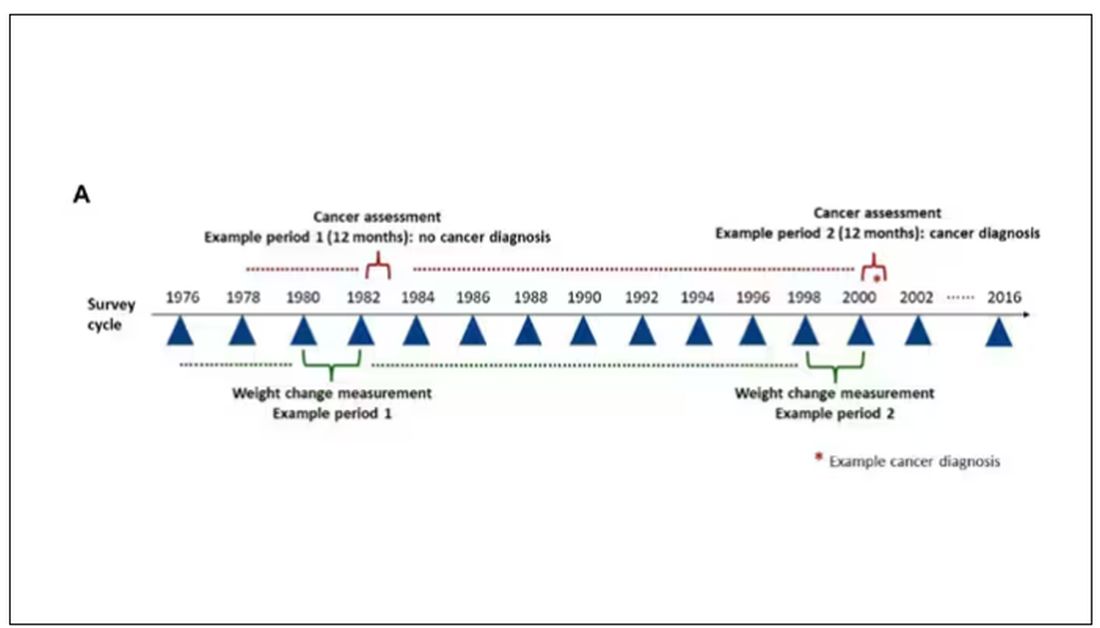
Participants recorded their weight — as an aside, a nested study found that self-reported rate was extremely well correlated with professionally measured weight — and whether they had received a cancer diagnosis since the last survey.
This allowed researchers to look at the phenomenon described above. Would weight loss precede a new diagnosis of cancer? And, more interestingly, would intentional weight loss precede a new diagnosis of cancer.
I don’t think it will surprise you to hear that individuals in the highest category of weight loss, those who lost more than 10% of their body weight over a 2-year period, had a larger risk of being diagnosed with cancer in the next year. That’s the yellow line in this graph. In fact, they had about a 40% higher risk than those who did not lose weight.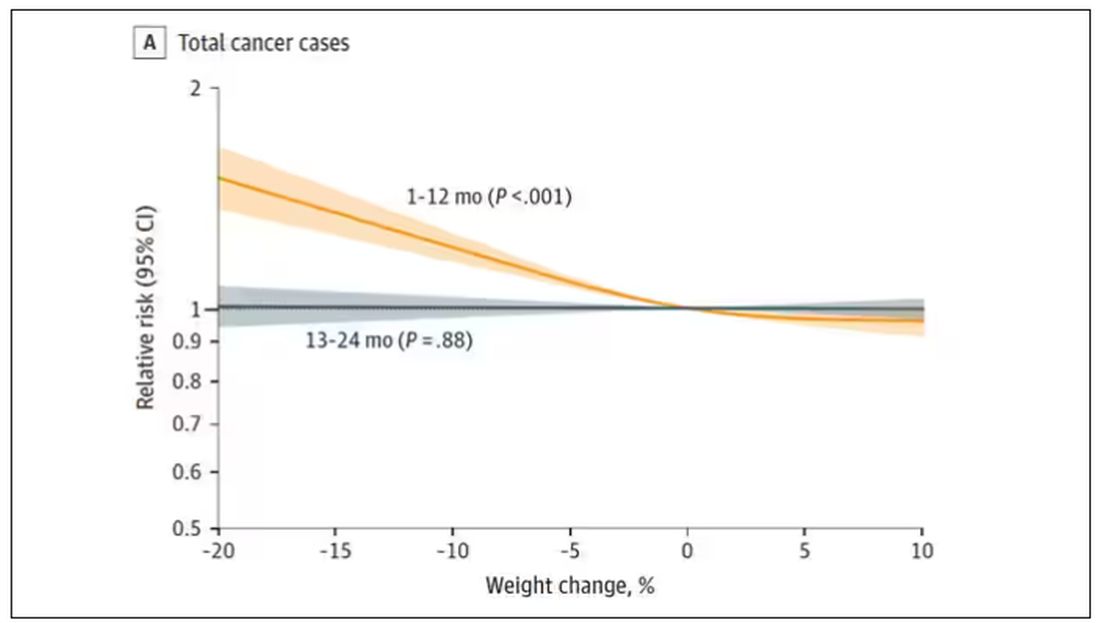
Increased risk was found across multiple cancer types, though cancers of the gastrointestinal tract, not surprisingly, were most strongly associated with antecedent weight loss.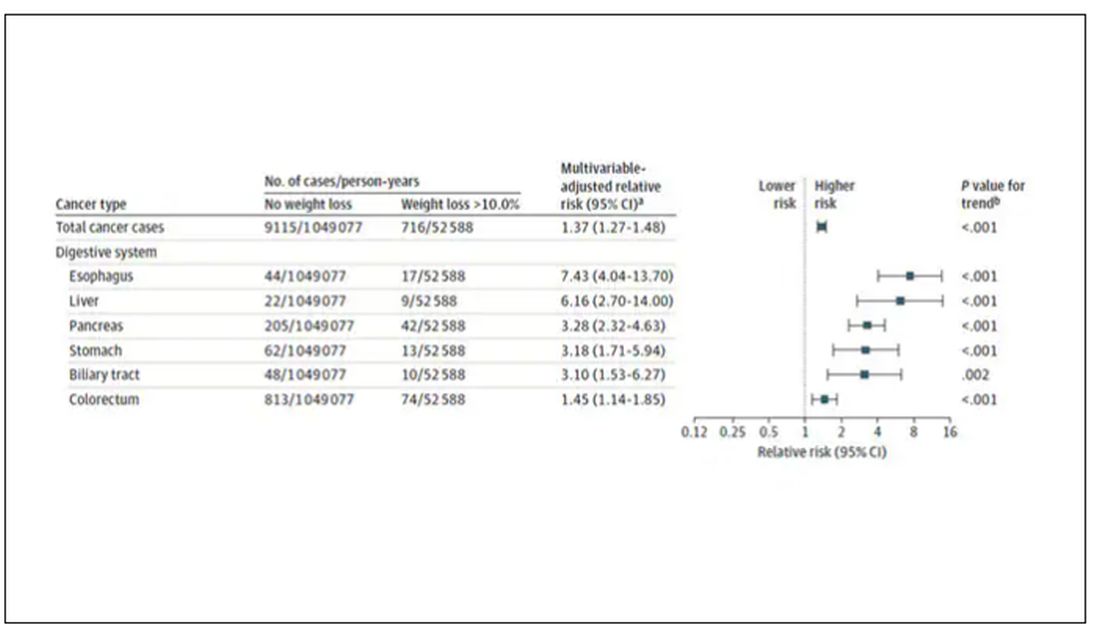
What about intentionality of weight loss? Unfortunately, the surveys did not ask participants whether they were trying to lose weight. Rather, the surveys asked about exercise and dietary habits. The researchers leveraged these responses to create three categories of participants: those who seemed to be trying to lose weight (defined as people who had increased their exercise and dietary quality); those who didn’t seem to be trying to lose weight (they changed neither exercise nor dietary behaviors); and a middle group, which changed one or the other of these behaviors but not both.
Let’s look at those who really seemed to be trying to lose weight. Over 2 years, they got more exercise and improved their diet.
If they succeeded in losing 10% or more of their body weight, they still had a higher risk for cancer than those who had not lost weight — about 30% higher, which is not that different from the 40% increased risk when you include those folks who weren’t changing their lifestyle.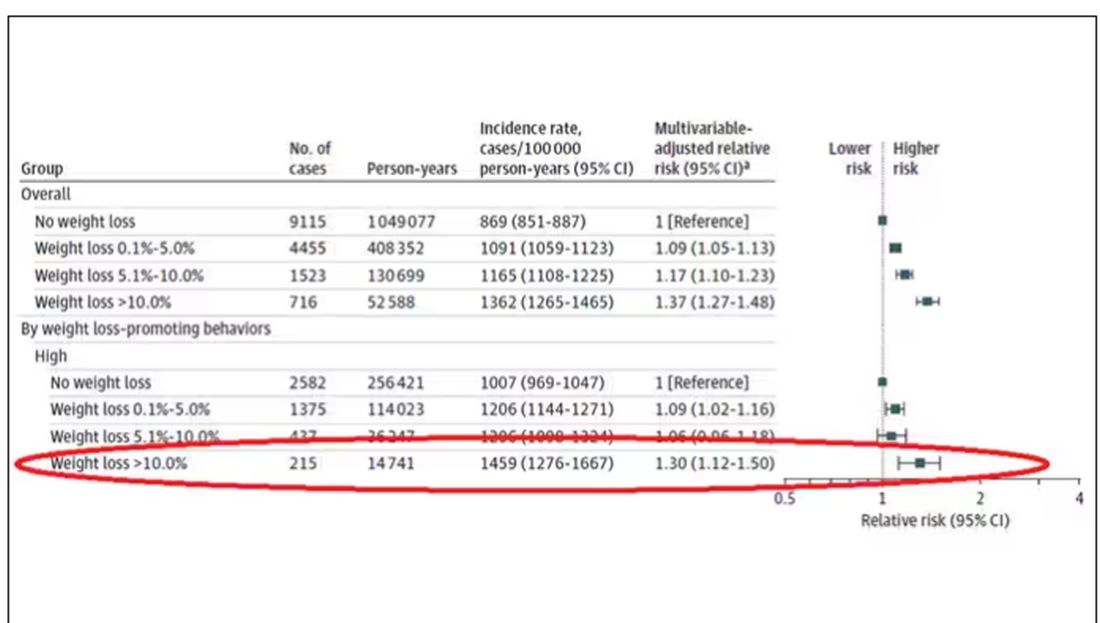
This is why this study is important. The classic teaching is that unintentional weight loss is a bad thing and needs a workup. That’s fine. But we live in a world where perhaps the majority of people are, at any given time, trying to lose weight.
We need to be careful here. I am not by any means trying to say that people who have successfully lost weight have cancer. Both of the following statements can be true:
Significant weight loss, whether intentional or not, is associated with a higher risk for cancer.
Most people with significant weight loss will not have cancer.
Both of these can be true because cancer is, fortunately, rare. Of people who lose weight, the vast majority will lose weight because they are engaging in healthier behaviors. A small number may lose weight because something else is wrong. It’s just hard to tell the two apart.
Out of the nearly 200,000 people in this study, only around 16,000 developed cancer during follow-up. Again, although the chance of having cancer is slightly higher if someone has experienced weight loss, the chance is still very low.
We also need to avoid suggesting that weight loss causes cancer. Some people lose weight because of an existing, as of yet undiagnosed cancer and its metabolic effects. This is borne out if you look at the risk of being diagnosed with cancer as you move further away from the interval of weight loss.
The further you get from the year of that 10% weight loss, the less likely you are to be diagnosed with cancer. Most of these cancers are diagnosed within a year of losing weight. In other words, if you’re reading this and getting worried that you lost weight 10 years ago, you’re probably out of the woods. That was, most likely, just you getting healthier.
Last thing: We have methods for weight loss now that are way more effective than diet or exercise. I’m looking at you, Ozempic. But aside from the weight loss wonder drugs, we have surgery and other interventions. This study did not capture any of that data. Ozempic wasn’t even on the market during this study, so we can’t say anything about the relationship between weight loss and cancer among people using nonlifestyle mechanisms to lose weight.
It’s a complicated system. But the clinically actionable point here is to notice if patients have lost weight. If they’ve lost it without trying, further workup is reasonable. If they’ve lost it but were trying to lose it, tell them “good job.” And consider a workup anyway.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
As anyone who has been through medical training will tell you, some little scenes just stick with you. I had been seeing a patient in our resident clinic in West Philly for a couple of years. She was in her mid-60s with diabetes and hypertension and a distant smoking history. She was overweight and had been trying to improve her diet and lose weight since I started seeing her. One day she came in and was delighted to report that she had finally started shedding some pounds — about 15 in the past 2 months.
I enthusiastically told my preceptor that my careful dietary counseling had finally done the job. She looked through the chart for a moment and asked, “Is she up to date on her cancer screening?” A workup revealed adenocarcinoma of the lung. The patient did well, actually, but the story stuck with me.
The textbooks call it “unintentional weight loss,” often in big, scary letters, and every doctor will go just a bit pale if a patient tells them that, despite efforts not to, they are losing weight. But true unintentional weight loss is not that common. After all, most of us are at least half-heartedly trying to lose weight all the time. Should doctors be worried when we are successful?
A new study suggests that perhaps they should. We’re talking about this study, appearing in JAMA, which combined participants from two long-running observational cohorts: 120,000 women from the Nurses’ Health Study, and 50,000 men from the Health Professionals Follow-Up Study. (These cohorts started in the 1970s and 1980s, so we’ll give them a pass on the gender-specific study designs.)
The rationale of enrolling healthcare providers in these cohort studies is that they would be reliable witnesses of their own health status. If a nurse or doctor says they have pancreatic cancer, it’s likely that they truly have pancreatic cancer. Detailed health surveys were distributed to the participants every other year, and the average follow-up was more than a decade.
Participants recorded their weight — as an aside, a nested study found that self-reported rate was extremely well correlated with professionally measured weight — and whether they had received a cancer diagnosis since the last survey.
This allowed researchers to look at the phenomenon described above. Would weight loss precede a new diagnosis of cancer? And, more interestingly, would intentional weight loss precede a new diagnosis of cancer.
I don’t think it will surprise you to hear that individuals in the highest category of weight loss, those who lost more than 10% of their body weight over a 2-year period, had a larger risk of being diagnosed with cancer in the next year. That’s the yellow line in this graph. In fact, they had about a 40% higher risk than those who did not lose weight.
Increased risk was found across multiple cancer types, though cancers of the gastrointestinal tract, not surprisingly, were most strongly associated with antecedent weight loss.
What about intentionality of weight loss? Unfortunately, the surveys did not ask participants whether they were trying to lose weight. Rather, the surveys asked about exercise and dietary habits. The researchers leveraged these responses to create three categories of participants: those who seemed to be trying to lose weight (defined as people who had increased their exercise and dietary quality); those who didn’t seem to be trying to lose weight (they changed neither exercise nor dietary behaviors); and a middle group, which changed one or the other of these behaviors but not both.
Let’s look at those who really seemed to be trying to lose weight. Over 2 years, they got more exercise and improved their diet.
If they succeeded in losing 10% or more of their body weight, they still had a higher risk for cancer than those who had not lost weight — about 30% higher, which is not that different from the 40% increased risk when you include those folks who weren’t changing their lifestyle.
This is why this study is important. The classic teaching is that unintentional weight loss is a bad thing and needs a workup. That’s fine. But we live in a world where perhaps the majority of people are, at any given time, trying to lose weight.
We need to be careful here. I am not by any means trying to say that people who have successfully lost weight have cancer. Both of the following statements can be true:
Significant weight loss, whether intentional or not, is associated with a higher risk for cancer.
Most people with significant weight loss will not have cancer.
Both of these can be true because cancer is, fortunately, rare. Of people who lose weight, the vast majority will lose weight because they are engaging in healthier behaviors. A small number may lose weight because something else is wrong. It’s just hard to tell the two apart.
Out of the nearly 200,000 people in this study, only around 16,000 developed cancer during follow-up. Again, although the chance of having cancer is slightly higher if someone has experienced weight loss, the chance is still very low.
We also need to avoid suggesting that weight loss causes cancer. Some people lose weight because of an existing, as of yet undiagnosed cancer and its metabolic effects. This is borne out if you look at the risk of being diagnosed with cancer as you move further away from the interval of weight loss.
The further you get from the year of that 10% weight loss, the less likely you are to be diagnosed with cancer. Most of these cancers are diagnosed within a year of losing weight. In other words, if you’re reading this and getting worried that you lost weight 10 years ago, you’re probably out of the woods. That was, most likely, just you getting healthier.
Last thing: We have methods for weight loss now that are way more effective than diet or exercise. I’m looking at you, Ozempic. But aside from the weight loss wonder drugs, we have surgery and other interventions. This study did not capture any of that data. Ozempic wasn’t even on the market during this study, so we can’t say anything about the relationship between weight loss and cancer among people using nonlifestyle mechanisms to lose weight.
It’s a complicated system. But the clinically actionable point here is to notice if patients have lost weight. If they’ve lost it without trying, further workup is reasonable. If they’ve lost it but were trying to lose it, tell them “good job.” And consider a workup anyway.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
As anyone who has been through medical training will tell you, some little scenes just stick with you. I had been seeing a patient in our resident clinic in West Philly for a couple of years. She was in her mid-60s with diabetes and hypertension and a distant smoking history. She was overweight and had been trying to improve her diet and lose weight since I started seeing her. One day she came in and was delighted to report that she had finally started shedding some pounds — about 15 in the past 2 months.
I enthusiastically told my preceptor that my careful dietary counseling had finally done the job. She looked through the chart for a moment and asked, “Is she up to date on her cancer screening?” A workup revealed adenocarcinoma of the lung. The patient did well, actually, but the story stuck with me.
The textbooks call it “unintentional weight loss,” often in big, scary letters, and every doctor will go just a bit pale if a patient tells them that, despite efforts not to, they are losing weight. But true unintentional weight loss is not that common. After all, most of us are at least half-heartedly trying to lose weight all the time. Should doctors be worried when we are successful?
A new study suggests that perhaps they should. We’re talking about this study, appearing in JAMA, which combined participants from two long-running observational cohorts: 120,000 women from the Nurses’ Health Study, and 50,000 men from the Health Professionals Follow-Up Study. (These cohorts started in the 1970s and 1980s, so we’ll give them a pass on the gender-specific study designs.)
The rationale of enrolling healthcare providers in these cohort studies is that they would be reliable witnesses of their own health status. If a nurse or doctor says they have pancreatic cancer, it’s likely that they truly have pancreatic cancer. Detailed health surveys were distributed to the participants every other year, and the average follow-up was more than a decade.
Participants recorded their weight — as an aside, a nested study found that self-reported rate was extremely well correlated with professionally measured weight — and whether they had received a cancer diagnosis since the last survey.
This allowed researchers to look at the phenomenon described above. Would weight loss precede a new diagnosis of cancer? And, more interestingly, would intentional weight loss precede a new diagnosis of cancer.
I don’t think it will surprise you to hear that individuals in the highest category of weight loss, those who lost more than 10% of their body weight over a 2-year period, had a larger risk of being diagnosed with cancer in the next year. That’s the yellow line in this graph. In fact, they had about a 40% higher risk than those who did not lose weight.
Increased risk was found across multiple cancer types, though cancers of the gastrointestinal tract, not surprisingly, were most strongly associated with antecedent weight loss.
What about intentionality of weight loss? Unfortunately, the surveys did not ask participants whether they were trying to lose weight. Rather, the surveys asked about exercise and dietary habits. The researchers leveraged these responses to create three categories of participants: those who seemed to be trying to lose weight (defined as people who had increased their exercise and dietary quality); those who didn’t seem to be trying to lose weight (they changed neither exercise nor dietary behaviors); and a middle group, which changed one or the other of these behaviors but not both.
Let’s look at those who really seemed to be trying to lose weight. Over 2 years, they got more exercise and improved their diet.
If they succeeded in losing 10% or more of their body weight, they still had a higher risk for cancer than those who had not lost weight — about 30% higher, which is not that different from the 40% increased risk when you include those folks who weren’t changing their lifestyle.
This is why this study is important. The classic teaching is that unintentional weight loss is a bad thing and needs a workup. That’s fine. But we live in a world where perhaps the majority of people are, at any given time, trying to lose weight.
We need to be careful here. I am not by any means trying to say that people who have successfully lost weight have cancer. Both of the following statements can be true:
Significant weight loss, whether intentional or not, is associated with a higher risk for cancer.
Most people with significant weight loss will not have cancer.
Both of these can be true because cancer is, fortunately, rare. Of people who lose weight, the vast majority will lose weight because they are engaging in healthier behaviors. A small number may lose weight because something else is wrong. It’s just hard to tell the two apart.
Out of the nearly 200,000 people in this study, only around 16,000 developed cancer during follow-up. Again, although the chance of having cancer is slightly higher if someone has experienced weight loss, the chance is still very low.
We also need to avoid suggesting that weight loss causes cancer. Some people lose weight because of an existing, as of yet undiagnosed cancer and its metabolic effects. This is borne out if you look at the risk of being diagnosed with cancer as you move further away from the interval of weight loss.
The further you get from the year of that 10% weight loss, the less likely you are to be diagnosed with cancer. Most of these cancers are diagnosed within a year of losing weight. In other words, if you’re reading this and getting worried that you lost weight 10 years ago, you’re probably out of the woods. That was, most likely, just you getting healthier.
Last thing: We have methods for weight loss now that are way more effective than diet or exercise. I’m looking at you, Ozempic. But aside from the weight loss wonder drugs, we have surgery and other interventions. This study did not capture any of that data. Ozempic wasn’t even on the market during this study, so we can’t say anything about the relationship between weight loss and cancer among people using nonlifestyle mechanisms to lose weight.
It’s a complicated system. But the clinically actionable point here is to notice if patients have lost weight. If they’ve lost it without trying, further workup is reasonable. If they’ve lost it but were trying to lose it, tell them “good job.” And consider a workup anyway.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Hair Creams: Do You Know the Health Risks?
In late December 2023, Brazil’s National Health Surveillance Agency (ANVISA) suspended the commercialization of approximately 1200 hair creams because of reports of eye irritation and temporary blindness.
A similar measure encompassing all hair creams sold in the country had already been announced by the agency in March. However, after a few weeks, ANVISA issued a resolution with rules for the products’ commercialization, allowing them back on the shelves.
With the new resolution, the sale of products that do not comply with the standards has once again been suspended. The reason is that reports of adverse events have reemerged. These events include temporary vision loss, headaches, and burning, tearing, itching, redness, and swelling of the eyes. According to reports, these adverse effects occurred mainly in people who used the specific products before swimming in the sea or in pools, or even going out in the rain.
The banned products contain 20% or more ethoxylated alcohols in their formulations. , potentially causing allergies and burns to the eyes and skin. They also have a high pulmonary and neurological toxicity. All these substances are eye irritants and can cause chemical keratitis. In extreme cases, corneal ulcers may develop, leading to vision loss.
The Brazilian Council of Ophthalmology also issued a warning on these products. It emphasized that, in addition to the sales prohibition, consumers should check the labels of hair creams to make sure that these toxic substances are not present in the product formulation.
The ANVISA website contains a list of creams that are considered safe and have not had their commercialization suspended, along with links to adverse event notifications reported by healthcare professionals or consumers.
For consumers who have recently used hair creams, the agency advises careful hair washing, including tilting the head backward to prevent the product from coming into contact with the eye area. If there is accidental eye contact, the eyes should be washed with plenty of water.
If there are any undesired effects after using these products, users should immediately seek the nearest healthcare service. Treatment should be individualized, possibly including ocular occlusion and the use of eye drops containing antibiotics or corticosteroids, among other medications.
Not every patient has easy access to an ophthalmologist in an emergency, so it is crucial for general practitioners to be prepared for initial care. In this regard, one of the most important measures is eye washing with copious amounts of clean water or saline solution for 5-10 minutes.
Eye itching is a frequent manifestation of using hair creams, and scratching the area may worsen the condition. Ocular occlusion can protect the cornea until an evaluation can be performed by a specialist.
Although we prefer our patients to stay away from these creams, it is also important to disseminate this information and advise them to read labels and use safe cosmetics.
This article was translated from the Medscape Portuguese edition. A version of this article appeared on Medscape.com.
In late December 2023, Brazil’s National Health Surveillance Agency (ANVISA) suspended the commercialization of approximately 1200 hair creams because of reports of eye irritation and temporary blindness.
A similar measure encompassing all hair creams sold in the country had already been announced by the agency in March. However, after a few weeks, ANVISA issued a resolution with rules for the products’ commercialization, allowing them back on the shelves.
With the new resolution, the sale of products that do not comply with the standards has once again been suspended. The reason is that reports of adverse events have reemerged. These events include temporary vision loss, headaches, and burning, tearing, itching, redness, and swelling of the eyes. According to reports, these adverse effects occurred mainly in people who used the specific products before swimming in the sea or in pools, or even going out in the rain.
The banned products contain 20% or more ethoxylated alcohols in their formulations. , potentially causing allergies and burns to the eyes and skin. They also have a high pulmonary and neurological toxicity. All these substances are eye irritants and can cause chemical keratitis. In extreme cases, corneal ulcers may develop, leading to vision loss.
The Brazilian Council of Ophthalmology also issued a warning on these products. It emphasized that, in addition to the sales prohibition, consumers should check the labels of hair creams to make sure that these toxic substances are not present in the product formulation.
The ANVISA website contains a list of creams that are considered safe and have not had their commercialization suspended, along with links to adverse event notifications reported by healthcare professionals or consumers.
For consumers who have recently used hair creams, the agency advises careful hair washing, including tilting the head backward to prevent the product from coming into contact with the eye area. If there is accidental eye contact, the eyes should be washed with plenty of water.
If there are any undesired effects after using these products, users should immediately seek the nearest healthcare service. Treatment should be individualized, possibly including ocular occlusion and the use of eye drops containing antibiotics or corticosteroids, among other medications.
Not every patient has easy access to an ophthalmologist in an emergency, so it is crucial for general practitioners to be prepared for initial care. In this regard, one of the most important measures is eye washing with copious amounts of clean water or saline solution for 5-10 minutes.
Eye itching is a frequent manifestation of using hair creams, and scratching the area may worsen the condition. Ocular occlusion can protect the cornea until an evaluation can be performed by a specialist.
Although we prefer our patients to stay away from these creams, it is also important to disseminate this information and advise them to read labels and use safe cosmetics.
This article was translated from the Medscape Portuguese edition. A version of this article appeared on Medscape.com.
In late December 2023, Brazil’s National Health Surveillance Agency (ANVISA) suspended the commercialization of approximately 1200 hair creams because of reports of eye irritation and temporary blindness.
A similar measure encompassing all hair creams sold in the country had already been announced by the agency in March. However, after a few weeks, ANVISA issued a resolution with rules for the products’ commercialization, allowing them back on the shelves.
With the new resolution, the sale of products that do not comply with the standards has once again been suspended. The reason is that reports of adverse events have reemerged. These events include temporary vision loss, headaches, and burning, tearing, itching, redness, and swelling of the eyes. According to reports, these adverse effects occurred mainly in people who used the specific products before swimming in the sea or in pools, or even going out in the rain.
The banned products contain 20% or more ethoxylated alcohols in their formulations. , potentially causing allergies and burns to the eyes and skin. They also have a high pulmonary and neurological toxicity. All these substances are eye irritants and can cause chemical keratitis. In extreme cases, corneal ulcers may develop, leading to vision loss.
The Brazilian Council of Ophthalmology also issued a warning on these products. It emphasized that, in addition to the sales prohibition, consumers should check the labels of hair creams to make sure that these toxic substances are not present in the product formulation.
The ANVISA website contains a list of creams that are considered safe and have not had their commercialization suspended, along with links to adverse event notifications reported by healthcare professionals or consumers.
For consumers who have recently used hair creams, the agency advises careful hair washing, including tilting the head backward to prevent the product from coming into contact with the eye area. If there is accidental eye contact, the eyes should be washed with plenty of water.
If there are any undesired effects after using these products, users should immediately seek the nearest healthcare service. Treatment should be individualized, possibly including ocular occlusion and the use of eye drops containing antibiotics or corticosteroids, among other medications.
Not every patient has easy access to an ophthalmologist in an emergency, so it is crucial for general practitioners to be prepared for initial care. In this regard, one of the most important measures is eye washing with copious amounts of clean water or saline solution for 5-10 minutes.
Eye itching is a frequent manifestation of using hair creams, and scratching the area may worsen the condition. Ocular occlusion can protect the cornea until an evaluation can be performed by a specialist.
Although we prefer our patients to stay away from these creams, it is also important to disseminate this information and advise them to read labels and use safe cosmetics.
This article was translated from the Medscape Portuguese edition. A version of this article appeared on Medscape.com.

