User login
Did prior authorization refusals lead to this patient’s death?
Ramy Sedhom, MD, a medical oncologist and a palliative care physician at Penn Medicine Princeton Health in Plainsboro, N.J., will always wonder if prior authorization refusals led to his patient’s death.
The patient had advanced gastric cancer and the insurer initially denied a PET scan to rule out metastatic disease. When the scan was eventually allowed, it revealed that the cancer had spread.
Standard treatment would have been difficult for the patient, an older individual with comorbidities. But Dr. Sedhom knew that a European study had reported equal efficacy and fewer side effects with a reduced chemotherapy regimen, and he thought that was the best approach in this situation.
The insurer disagreed with Dr. Sedhom’s decision and, while the two argued, the patient’s symptoms worsened. He was admitted to the hospital, where he experienced a decline in function, common for older patients. “Long story short, he was never able to seek treatment and then transitioned to hospice,” Dr. Sedhom said. “It was one of those situations where there was a 3- to 4-week delay in what should have been standard care.”
. Nearly 4 years after major organizations — American Hospital Association, America’s Health Insurance Plans, American Medical Association, Blue Cross Blue Shield Association, and others — signed a consensus statement agreeing to improve the prior authorization process, physicians say little progress has been made.
Indeed, 83% of physicians say that the number of prior authorizations required for prescription medications and medical services has increased over the last 5 years, according to survey results released earlier this year.
“It’s decidedly worse — there’s no question about it,” said Andrew R. Spector, MD, a neurologist and sleep medicine specialist at Duke Health in Durham, N.C. “Drugs that I used to get without prior authorizations now require them.”
When Vignesh I. Doraiswamy, MD, an internal medicine hospitalist at the Ohio State University Wexner Medical Center in Columbus, discharged a patient with Clostridioides difficile infection, he followed clinical guidelines to prescribe vancomycin for 10 to 14 days. “And the insurance company said, ‘Well, yeah, we only authorize about 5 days,’ which just makes no sense,” Dr. Doraiswamy said. “There’s nowhere in any literature that says 5 days is sufficient. What worries me is that is the standard of care we are supposed to give and yet we are unable to.”
Yash B. Jobanputra, MD, a cardiology fellow at Saint Vincent Hospital in Worcester, Mass., laments that prior authorization is used in situations that simply do not make common sense. During his residency, a woman who had tested positive for the BRCA gene mutation with a strong family history of breast cancer needed a breast ultrasound and an MRI scan every 6 months to 1 year. Despite the documentation that she was at extremely high risk for developing breast cancer, he had to go through prior authorization every time she was due for new images.
“I had to call the insurance company, they would put me on hold, I would wait to speak to a physician — and the end response would be, ‘Yeah, this is what needs to be done,’” he said. “But having established her positive status once should be enough really. I shouldn’t have to go through the circus all over again.”
Prior authorization is also being used for routine diagnostics, such as a Holter monitor for patients complaining of heart palpitations. “Depending on the insurance, for some patients we can give it to them in the clinic right away,” Dr. Jobanputra said. “Whereas some others we have to wait until we get prior authorization from the insurance company and the patient has to come back again to the hospital to get the monitor. That is a delay in patient care.”
The delays also extend to emergency care, Dr. Doraiswamy said. He cites the example of a heart attack patient who needed an emergency heart catheterization but ran into a prior authorization delay. “I just said, ‘Try your best not to get stressed’ which is not easy for a patient finding out their stay wasn’t covered when they had just been through a heart attack,” he said. “Then I spent 20 to 30 minutes — most of it on hold — to answer the question ‘Why did this patient need to get admitted?’ “
Physicians feel disrespected because that type of prior authorization hassle is just busywork. “Rarely is a valid stay that was initially denied, not eventually accepted,” Dr. Doraiswamy said. “But why couldn’t they have just seen that the guy had a heart attack and he obviously needed to be in the hospital?”
For Dr. Spector, the Duke Health sleep medicine specialist, prior authorization is not just a speed bump, it’s a full stop. Insurers have started mandating a multiple sleep latency test (MSLT) to confirm narcolepsy before covering medication to treat the condition. “We know that the MSLT is very often wrong,” he said. “There are a lot of times we’re dealing with patients with narcolepsy who simply don’t meet the testing criteria that the insurance requires, and payers will not accept our clinical judgment.”
In his view, the prior authorization landscape is worsening — and not only because a “faulty test” is being used to deny treatment. “The appeal process is worse,” Dr. Spector said. “I used to be able to get on the phone and do a peer-to-peer review with a physician who I could reason with… but that doesn’t happen anymore. There is virtually no way to bypass these blanket rules.”
Other survey findings also stand in direct contradiction of the 2018 consensus agreement:
A large majority (87%) of physicians report that prior authorization interferes with continuity of care, even though the industry groups agreed that patients should be protected from treatment disruption when there is a formulary or treatment-coverage change.
Despite a consensus to encourage transparency and easy accessibility of prior authorization requirements, 68% of physicians reported that it is difficult to determine whether a prescription medication requires prior authorization, and 58% report that it’s difficult for medical services.
Phone and fax are the most commonly used methods for completing prior authorizations, despite agreement that electronic prior authorization, using existing national standard transactions, should be accelerated. Fewer than one quarter of physicians said that their electronic health record system supports electronic prior authorization for prescription medications.
Dr. Spector wants to see legislation that forces insurers to live up to some of the tenets of the 2018 consensus statement. In September, a new Texas law went into effect, exempting physicians from prior authorization if, during the previous six months, 90% of their treatments met an insurer›s medical necessity criteria. In January, the recently approved Prior Authorization Reform Act in Illinois will reduce the number of services subject to prior authorization, mandate a prior authorization decision within 5 days, and set disciplinary measures for health plans that do not comply, among other things.
“What gives me hope is that at least somewhere in the country, somebody is doing something,” Dr. Spector said. “And if it goes well, maybe other insurers will adopt it. I’m really hoping they demonstrate that the money they can save on the administration of all the appeals and prior authorization paperwork can actually go into caring for patients.”
In addition to state-level action, reform may also be advancing at the federal level. In October, a bill was introduced in the U.S. Senate that mirrors a prior authorization reform bill introduced in the House of Representatives last May. Both bills have broad bipartisan support; the House bill has more than 235 co-sponsors.
In an interview with this news organization, Rep. Ami Bera, MD, (D-CA) said it is “very realistic” that the bill will become law during this session of Congress. “We do think this bill will get marked up in committee and hopefully we can get it to the floor either as a stand-alone bill where we know we have the votes to pass it or as part of a larger legislative package,” he said.
If approved, the Improving Seniors’ Timely Access to Care Act of 2021 would require that Medicare Advantage plans minimize the use of prior authorization for routinely approved services; require real-time decisions for certain requests; report the extent of their use of prior authorization and their rate of approvals or denials, among other things; and establish an electronic prior authorization system.
Medicare Advantage plans are private insurers that are regulated by the Centers for Medicare & Medicaid Services (CMS), which will create the specific rules and penalties associated with the reforms, if they become law. “One would presume that a condition of being a Medicare Advantage plan is that you’re going to have to comply with these new regulations,” said Katie Orrico, senior vice president of health policy and advocacy for the American Association of Neurological Surgeons and Congress of Neurological Surgeons (AANS/CNS). “So they will have some amount of teeth in the form of a mandate.”
The AANS and CNS are part of the Regulatory Relief Coalition, a group of 14 national physician specialty organizations. Winning prior authorization reform in the Medicare Advantage plans is part of its bigger strategy. “If those commercial plans have to follow a set of rules and processes for Medicare, then why not just expand those same processes to all other parts of their business?” Ms. Orrico said.
Despite his frustration with their prior authorization processes, Dr. Doraiswamy, the Ohio State hospitalist, agrees that working to improve insurers’ practices is the best way forward. “It’s so easy to make them look like these evil, giant conglomerations that exist solely to suck money and not care about anyone’s health, but I don’t know if that’s necessarily the case,” he said. “We really have to figure out how best to work with insurance companies to make sure that, while they are profit-generating institutions, that [profit] shouldn’t come at the cost of patient care.”
A version of this article first appeared on Medscape.com.
Ramy Sedhom, MD, a medical oncologist and a palliative care physician at Penn Medicine Princeton Health in Plainsboro, N.J., will always wonder if prior authorization refusals led to his patient’s death.
The patient had advanced gastric cancer and the insurer initially denied a PET scan to rule out metastatic disease. When the scan was eventually allowed, it revealed that the cancer had spread.
Standard treatment would have been difficult for the patient, an older individual with comorbidities. But Dr. Sedhom knew that a European study had reported equal efficacy and fewer side effects with a reduced chemotherapy regimen, and he thought that was the best approach in this situation.
The insurer disagreed with Dr. Sedhom’s decision and, while the two argued, the patient’s symptoms worsened. He was admitted to the hospital, where he experienced a decline in function, common for older patients. “Long story short, he was never able to seek treatment and then transitioned to hospice,” Dr. Sedhom said. “It was one of those situations where there was a 3- to 4-week delay in what should have been standard care.”
. Nearly 4 years after major organizations — American Hospital Association, America’s Health Insurance Plans, American Medical Association, Blue Cross Blue Shield Association, and others — signed a consensus statement agreeing to improve the prior authorization process, physicians say little progress has been made.
Indeed, 83% of physicians say that the number of prior authorizations required for prescription medications and medical services has increased over the last 5 years, according to survey results released earlier this year.
“It’s decidedly worse — there’s no question about it,” said Andrew R. Spector, MD, a neurologist and sleep medicine specialist at Duke Health in Durham, N.C. “Drugs that I used to get without prior authorizations now require them.”
When Vignesh I. Doraiswamy, MD, an internal medicine hospitalist at the Ohio State University Wexner Medical Center in Columbus, discharged a patient with Clostridioides difficile infection, he followed clinical guidelines to prescribe vancomycin for 10 to 14 days. “And the insurance company said, ‘Well, yeah, we only authorize about 5 days,’ which just makes no sense,” Dr. Doraiswamy said. “There’s nowhere in any literature that says 5 days is sufficient. What worries me is that is the standard of care we are supposed to give and yet we are unable to.”
Yash B. Jobanputra, MD, a cardiology fellow at Saint Vincent Hospital in Worcester, Mass., laments that prior authorization is used in situations that simply do not make common sense. During his residency, a woman who had tested positive for the BRCA gene mutation with a strong family history of breast cancer needed a breast ultrasound and an MRI scan every 6 months to 1 year. Despite the documentation that she was at extremely high risk for developing breast cancer, he had to go through prior authorization every time she was due for new images.
“I had to call the insurance company, they would put me on hold, I would wait to speak to a physician — and the end response would be, ‘Yeah, this is what needs to be done,’” he said. “But having established her positive status once should be enough really. I shouldn’t have to go through the circus all over again.”
Prior authorization is also being used for routine diagnostics, such as a Holter monitor for patients complaining of heart palpitations. “Depending on the insurance, for some patients we can give it to them in the clinic right away,” Dr. Jobanputra said. “Whereas some others we have to wait until we get prior authorization from the insurance company and the patient has to come back again to the hospital to get the monitor. That is a delay in patient care.”
The delays also extend to emergency care, Dr. Doraiswamy said. He cites the example of a heart attack patient who needed an emergency heart catheterization but ran into a prior authorization delay. “I just said, ‘Try your best not to get stressed’ which is not easy for a patient finding out their stay wasn’t covered when they had just been through a heart attack,” he said. “Then I spent 20 to 30 minutes — most of it on hold — to answer the question ‘Why did this patient need to get admitted?’ “
Physicians feel disrespected because that type of prior authorization hassle is just busywork. “Rarely is a valid stay that was initially denied, not eventually accepted,” Dr. Doraiswamy said. “But why couldn’t they have just seen that the guy had a heart attack and he obviously needed to be in the hospital?”
For Dr. Spector, the Duke Health sleep medicine specialist, prior authorization is not just a speed bump, it’s a full stop. Insurers have started mandating a multiple sleep latency test (MSLT) to confirm narcolepsy before covering medication to treat the condition. “We know that the MSLT is very often wrong,” he said. “There are a lot of times we’re dealing with patients with narcolepsy who simply don’t meet the testing criteria that the insurance requires, and payers will not accept our clinical judgment.”
In his view, the prior authorization landscape is worsening — and not only because a “faulty test” is being used to deny treatment. “The appeal process is worse,” Dr. Spector said. “I used to be able to get on the phone and do a peer-to-peer review with a physician who I could reason with… but that doesn’t happen anymore. There is virtually no way to bypass these blanket rules.”
Other survey findings also stand in direct contradiction of the 2018 consensus agreement:
A large majority (87%) of physicians report that prior authorization interferes with continuity of care, even though the industry groups agreed that patients should be protected from treatment disruption when there is a formulary or treatment-coverage change.
Despite a consensus to encourage transparency and easy accessibility of prior authorization requirements, 68% of physicians reported that it is difficult to determine whether a prescription medication requires prior authorization, and 58% report that it’s difficult for medical services.
Phone and fax are the most commonly used methods for completing prior authorizations, despite agreement that electronic prior authorization, using existing national standard transactions, should be accelerated. Fewer than one quarter of physicians said that their electronic health record system supports electronic prior authorization for prescription medications.
Dr. Spector wants to see legislation that forces insurers to live up to some of the tenets of the 2018 consensus statement. In September, a new Texas law went into effect, exempting physicians from prior authorization if, during the previous six months, 90% of their treatments met an insurer›s medical necessity criteria. In January, the recently approved Prior Authorization Reform Act in Illinois will reduce the number of services subject to prior authorization, mandate a prior authorization decision within 5 days, and set disciplinary measures for health plans that do not comply, among other things.
“What gives me hope is that at least somewhere in the country, somebody is doing something,” Dr. Spector said. “And if it goes well, maybe other insurers will adopt it. I’m really hoping they demonstrate that the money they can save on the administration of all the appeals and prior authorization paperwork can actually go into caring for patients.”
In addition to state-level action, reform may also be advancing at the federal level. In October, a bill was introduced in the U.S. Senate that mirrors a prior authorization reform bill introduced in the House of Representatives last May. Both bills have broad bipartisan support; the House bill has more than 235 co-sponsors.
In an interview with this news organization, Rep. Ami Bera, MD, (D-CA) said it is “very realistic” that the bill will become law during this session of Congress. “We do think this bill will get marked up in committee and hopefully we can get it to the floor either as a stand-alone bill where we know we have the votes to pass it or as part of a larger legislative package,” he said.
If approved, the Improving Seniors’ Timely Access to Care Act of 2021 would require that Medicare Advantage plans minimize the use of prior authorization for routinely approved services; require real-time decisions for certain requests; report the extent of their use of prior authorization and their rate of approvals or denials, among other things; and establish an electronic prior authorization system.
Medicare Advantage plans are private insurers that are regulated by the Centers for Medicare & Medicaid Services (CMS), which will create the specific rules and penalties associated with the reforms, if they become law. “One would presume that a condition of being a Medicare Advantage plan is that you’re going to have to comply with these new regulations,” said Katie Orrico, senior vice president of health policy and advocacy for the American Association of Neurological Surgeons and Congress of Neurological Surgeons (AANS/CNS). “So they will have some amount of teeth in the form of a mandate.”
The AANS and CNS are part of the Regulatory Relief Coalition, a group of 14 national physician specialty organizations. Winning prior authorization reform in the Medicare Advantage plans is part of its bigger strategy. “If those commercial plans have to follow a set of rules and processes for Medicare, then why not just expand those same processes to all other parts of their business?” Ms. Orrico said.
Despite his frustration with their prior authorization processes, Dr. Doraiswamy, the Ohio State hospitalist, agrees that working to improve insurers’ practices is the best way forward. “It’s so easy to make them look like these evil, giant conglomerations that exist solely to suck money and not care about anyone’s health, but I don’t know if that’s necessarily the case,” he said. “We really have to figure out how best to work with insurance companies to make sure that, while they are profit-generating institutions, that [profit] shouldn’t come at the cost of patient care.”
A version of this article first appeared on Medscape.com.
Ramy Sedhom, MD, a medical oncologist and a palliative care physician at Penn Medicine Princeton Health in Plainsboro, N.J., will always wonder if prior authorization refusals led to his patient’s death.
The patient had advanced gastric cancer and the insurer initially denied a PET scan to rule out metastatic disease. When the scan was eventually allowed, it revealed that the cancer had spread.
Standard treatment would have been difficult for the patient, an older individual with comorbidities. But Dr. Sedhom knew that a European study had reported equal efficacy and fewer side effects with a reduced chemotherapy regimen, and he thought that was the best approach in this situation.
The insurer disagreed with Dr. Sedhom’s decision and, while the two argued, the patient’s symptoms worsened. He was admitted to the hospital, where he experienced a decline in function, common for older patients. “Long story short, he was never able to seek treatment and then transitioned to hospice,” Dr. Sedhom said. “It was one of those situations where there was a 3- to 4-week delay in what should have been standard care.”
. Nearly 4 years after major organizations — American Hospital Association, America’s Health Insurance Plans, American Medical Association, Blue Cross Blue Shield Association, and others — signed a consensus statement agreeing to improve the prior authorization process, physicians say little progress has been made.
Indeed, 83% of physicians say that the number of prior authorizations required for prescription medications and medical services has increased over the last 5 years, according to survey results released earlier this year.
“It’s decidedly worse — there’s no question about it,” said Andrew R. Spector, MD, a neurologist and sleep medicine specialist at Duke Health in Durham, N.C. “Drugs that I used to get without prior authorizations now require them.”
When Vignesh I. Doraiswamy, MD, an internal medicine hospitalist at the Ohio State University Wexner Medical Center in Columbus, discharged a patient with Clostridioides difficile infection, he followed clinical guidelines to prescribe vancomycin for 10 to 14 days. “And the insurance company said, ‘Well, yeah, we only authorize about 5 days,’ which just makes no sense,” Dr. Doraiswamy said. “There’s nowhere in any literature that says 5 days is sufficient. What worries me is that is the standard of care we are supposed to give and yet we are unable to.”
Yash B. Jobanputra, MD, a cardiology fellow at Saint Vincent Hospital in Worcester, Mass., laments that prior authorization is used in situations that simply do not make common sense. During his residency, a woman who had tested positive for the BRCA gene mutation with a strong family history of breast cancer needed a breast ultrasound and an MRI scan every 6 months to 1 year. Despite the documentation that she was at extremely high risk for developing breast cancer, he had to go through prior authorization every time she was due for new images.
“I had to call the insurance company, they would put me on hold, I would wait to speak to a physician — and the end response would be, ‘Yeah, this is what needs to be done,’” he said. “But having established her positive status once should be enough really. I shouldn’t have to go through the circus all over again.”
Prior authorization is also being used for routine diagnostics, such as a Holter monitor for patients complaining of heart palpitations. “Depending on the insurance, for some patients we can give it to them in the clinic right away,” Dr. Jobanputra said. “Whereas some others we have to wait until we get prior authorization from the insurance company and the patient has to come back again to the hospital to get the monitor. That is a delay in patient care.”
The delays also extend to emergency care, Dr. Doraiswamy said. He cites the example of a heart attack patient who needed an emergency heart catheterization but ran into a prior authorization delay. “I just said, ‘Try your best not to get stressed’ which is not easy for a patient finding out their stay wasn’t covered when they had just been through a heart attack,” he said. “Then I spent 20 to 30 minutes — most of it on hold — to answer the question ‘Why did this patient need to get admitted?’ “
Physicians feel disrespected because that type of prior authorization hassle is just busywork. “Rarely is a valid stay that was initially denied, not eventually accepted,” Dr. Doraiswamy said. “But why couldn’t they have just seen that the guy had a heart attack and he obviously needed to be in the hospital?”
For Dr. Spector, the Duke Health sleep medicine specialist, prior authorization is not just a speed bump, it’s a full stop. Insurers have started mandating a multiple sleep latency test (MSLT) to confirm narcolepsy before covering medication to treat the condition. “We know that the MSLT is very often wrong,” he said. “There are a lot of times we’re dealing with patients with narcolepsy who simply don’t meet the testing criteria that the insurance requires, and payers will not accept our clinical judgment.”
In his view, the prior authorization landscape is worsening — and not only because a “faulty test” is being used to deny treatment. “The appeal process is worse,” Dr. Spector said. “I used to be able to get on the phone and do a peer-to-peer review with a physician who I could reason with… but that doesn’t happen anymore. There is virtually no way to bypass these blanket rules.”
Other survey findings also stand in direct contradiction of the 2018 consensus agreement:
A large majority (87%) of physicians report that prior authorization interferes with continuity of care, even though the industry groups agreed that patients should be protected from treatment disruption when there is a formulary or treatment-coverage change.
Despite a consensus to encourage transparency and easy accessibility of prior authorization requirements, 68% of physicians reported that it is difficult to determine whether a prescription medication requires prior authorization, and 58% report that it’s difficult for medical services.
Phone and fax are the most commonly used methods for completing prior authorizations, despite agreement that electronic prior authorization, using existing national standard transactions, should be accelerated. Fewer than one quarter of physicians said that their electronic health record system supports electronic prior authorization for prescription medications.
Dr. Spector wants to see legislation that forces insurers to live up to some of the tenets of the 2018 consensus statement. In September, a new Texas law went into effect, exempting physicians from prior authorization if, during the previous six months, 90% of their treatments met an insurer›s medical necessity criteria. In January, the recently approved Prior Authorization Reform Act in Illinois will reduce the number of services subject to prior authorization, mandate a prior authorization decision within 5 days, and set disciplinary measures for health plans that do not comply, among other things.
“What gives me hope is that at least somewhere in the country, somebody is doing something,” Dr. Spector said. “And if it goes well, maybe other insurers will adopt it. I’m really hoping they demonstrate that the money they can save on the administration of all the appeals and prior authorization paperwork can actually go into caring for patients.”
In addition to state-level action, reform may also be advancing at the federal level. In October, a bill was introduced in the U.S. Senate that mirrors a prior authorization reform bill introduced in the House of Representatives last May. Both bills have broad bipartisan support; the House bill has more than 235 co-sponsors.
In an interview with this news organization, Rep. Ami Bera, MD, (D-CA) said it is “very realistic” that the bill will become law during this session of Congress. “We do think this bill will get marked up in committee and hopefully we can get it to the floor either as a stand-alone bill where we know we have the votes to pass it or as part of a larger legislative package,” he said.
If approved, the Improving Seniors’ Timely Access to Care Act of 2021 would require that Medicare Advantage plans minimize the use of prior authorization for routinely approved services; require real-time decisions for certain requests; report the extent of their use of prior authorization and their rate of approvals or denials, among other things; and establish an electronic prior authorization system.
Medicare Advantage plans are private insurers that are regulated by the Centers for Medicare & Medicaid Services (CMS), which will create the specific rules and penalties associated with the reforms, if they become law. “One would presume that a condition of being a Medicare Advantage plan is that you’re going to have to comply with these new regulations,” said Katie Orrico, senior vice president of health policy and advocacy for the American Association of Neurological Surgeons and Congress of Neurological Surgeons (AANS/CNS). “So they will have some amount of teeth in the form of a mandate.”
The AANS and CNS are part of the Regulatory Relief Coalition, a group of 14 national physician specialty organizations. Winning prior authorization reform in the Medicare Advantage plans is part of its bigger strategy. “If those commercial plans have to follow a set of rules and processes for Medicare, then why not just expand those same processes to all other parts of their business?” Ms. Orrico said.
Despite his frustration with their prior authorization processes, Dr. Doraiswamy, the Ohio State hospitalist, agrees that working to improve insurers’ practices is the best way forward. “It’s so easy to make them look like these evil, giant conglomerations that exist solely to suck money and not care about anyone’s health, but I don’t know if that’s necessarily the case,” he said. “We really have to figure out how best to work with insurance companies to make sure that, while they are profit-generating institutions, that [profit] shouldn’t come at the cost of patient care.”
A version of this article first appeared on Medscape.com.
Intranasal vs. intramuscular naloxone in reversing opioid overdose
Background: Naloxone is an opioid antagonist that works to treat opioid overdose. Few randomized trials have assessed the efficacy of intranasal administration, whereas more data have been published supporting use of intramuscular naloxone. This prospective trial examines the ability of the same dose (800 mcg per 1 mL solution) of intranasal naloxone vs. intramuscular naloxone at managing opioid overdose.
Study design: Double-blind double-dummy randomized clinical trial.
Setting: Single supervised injection center in Sydney.
Synopsis: In this study, 197 participants with opioid overdose were randomized to intramuscular or intranasal naloxone. If the patient did not respond to either (GSC score less than 13, RR less than 10, or oxygen saturation less than 95%), a rescue dose of intramuscular naloxone was given. Participants who received the intramuscular naloxone were less likely to need the rescue dose (8.6% vs. 23.1%; odds ratio, 0.35; P = .002). The time to achieve an RR greater than 10 (15 vs. 8 minutes) and GSC score greater than 13 (17 vs. 8 minutes) was longer in the intranasal than the intramuscular group. Limitations include the setting of a controlled environment. Also, this protocol called for an initial 5 minutes of ventilation prior to randomization, which selected for more severe overdose cases in the overall study population. More studies are needed to assess efficacy in the field, needlestick injuries, and larger intranasal doses.
Bottom line: Intranasal naloxone effectively reverses opioid overdose but not as effectively as intramuscular naloxone at the same dose.
Citation: Dietze P et al. Effect of intranasal vs intramuscular naloxone on opioid overdose: A randomized clinical trial. JAMA Netw Open. 2019;2:e1914977. doi: 1
Dr. Welter is a hospitalist at Northwestern Memorial Hospital and instructor of medicine, Feinberg School of Medicine, both in Chicago.
Background: Naloxone is an opioid antagonist that works to treat opioid overdose. Few randomized trials have assessed the efficacy of intranasal administration, whereas more data have been published supporting use of intramuscular naloxone. This prospective trial examines the ability of the same dose (800 mcg per 1 mL solution) of intranasal naloxone vs. intramuscular naloxone at managing opioid overdose.
Study design: Double-blind double-dummy randomized clinical trial.
Setting: Single supervised injection center in Sydney.
Synopsis: In this study, 197 participants with opioid overdose were randomized to intramuscular or intranasal naloxone. If the patient did not respond to either (GSC score less than 13, RR less than 10, or oxygen saturation less than 95%), a rescue dose of intramuscular naloxone was given. Participants who received the intramuscular naloxone were less likely to need the rescue dose (8.6% vs. 23.1%; odds ratio, 0.35; P = .002). The time to achieve an RR greater than 10 (15 vs. 8 minutes) and GSC score greater than 13 (17 vs. 8 minutes) was longer in the intranasal than the intramuscular group. Limitations include the setting of a controlled environment. Also, this protocol called for an initial 5 minutes of ventilation prior to randomization, which selected for more severe overdose cases in the overall study population. More studies are needed to assess efficacy in the field, needlestick injuries, and larger intranasal doses.
Bottom line: Intranasal naloxone effectively reverses opioid overdose but not as effectively as intramuscular naloxone at the same dose.
Citation: Dietze P et al. Effect of intranasal vs intramuscular naloxone on opioid overdose: A randomized clinical trial. JAMA Netw Open. 2019;2:e1914977. doi: 1
Dr. Welter is a hospitalist at Northwestern Memorial Hospital and instructor of medicine, Feinberg School of Medicine, both in Chicago.
Background: Naloxone is an opioid antagonist that works to treat opioid overdose. Few randomized trials have assessed the efficacy of intranasal administration, whereas more data have been published supporting use of intramuscular naloxone. This prospective trial examines the ability of the same dose (800 mcg per 1 mL solution) of intranasal naloxone vs. intramuscular naloxone at managing opioid overdose.
Study design: Double-blind double-dummy randomized clinical trial.
Setting: Single supervised injection center in Sydney.
Synopsis: In this study, 197 participants with opioid overdose were randomized to intramuscular or intranasal naloxone. If the patient did not respond to either (GSC score less than 13, RR less than 10, or oxygen saturation less than 95%), a rescue dose of intramuscular naloxone was given. Participants who received the intramuscular naloxone were less likely to need the rescue dose (8.6% vs. 23.1%; odds ratio, 0.35; P = .002). The time to achieve an RR greater than 10 (15 vs. 8 minutes) and GSC score greater than 13 (17 vs. 8 minutes) was longer in the intranasal than the intramuscular group. Limitations include the setting of a controlled environment. Also, this protocol called for an initial 5 minutes of ventilation prior to randomization, which selected for more severe overdose cases in the overall study population. More studies are needed to assess efficacy in the field, needlestick injuries, and larger intranasal doses.
Bottom line: Intranasal naloxone effectively reverses opioid overdose but not as effectively as intramuscular naloxone at the same dose.
Citation: Dietze P et al. Effect of intranasal vs intramuscular naloxone on opioid overdose: A randomized clinical trial. JAMA Netw Open. 2019;2:e1914977. doi: 1
Dr. Welter is a hospitalist at Northwestern Memorial Hospital and instructor of medicine, Feinberg School of Medicine, both in Chicago.
Comparing the efficacy and safety of common SIADH treatments
Background: Hyponatremia caused by SIADH is common in hospitalized patients, and most evidence for treatment comes from noncontrolled studies. This study aims to investigate the efficacy and safety of fluid restriction compared with furosemide, with or without NaCl supplementation, for treating SIADH.
Study design: Open-label randomized controlled trial.
Setting: Single center in Thailand.
Synopsis: There were 92 participants randomized to fluid restriction alone, fluid restriction and furosemide, or fluid restriction, furosemide, and NaCl supplementation. The authors assessed the primary outcome, change in sodium, at 4, 7, 14, and 28 days (baseline mean Na 125 mmol/L). By day 4, all groups had a significant increase in sodium (mean delta 5 mmol/L). The time to achieve a safe sodium level (Na less than 130 mmol/L) was not different among groups. Acute kidney injury was most common in patients who received furosemide and NaCl supplementation, compared with the fluid restriction and fluid restriction plus furosemide groups (32%, 10%, 17%, respectively; P = .07). Hypokalemia was also most common in the furosemide and NaCl group (42%, 13%, 23%, respectively; P = .01). Limitations include open-label study design, poor fluid restriction adherence (63% overall), and inflexible treatment regimens that excluded treatment with oral potassium.
Bottom line: In treatment of hyponatremia caused by SIADH, there was no benefit to adding furosemide with or without NaCl supplementation to fluid restriction. However, there was potential associated risk of acute kidney injury and hypokalemia.Citation: Krisanapan P et al. Efficacy of furosemide, oral sodium chloride, and fluid restriction for treatment of syndrome of inappropriate antidiuresis (SIADH): An open-label randomized controlled study (the EFFUSE-FLUID trial). Am J Kidney Dis. 2020 Aug;76(2):203-12. doi: 10.1053/j.ajkd.2019.11.012.
Dr. Welter is a hospitalist at Northwestern Memorial Hospital and instructor of medicine, Feinberg School of Medicine, both in Chicago.
Background: Hyponatremia caused by SIADH is common in hospitalized patients, and most evidence for treatment comes from noncontrolled studies. This study aims to investigate the efficacy and safety of fluid restriction compared with furosemide, with or without NaCl supplementation, for treating SIADH.
Study design: Open-label randomized controlled trial.
Setting: Single center in Thailand.
Synopsis: There were 92 participants randomized to fluid restriction alone, fluid restriction and furosemide, or fluid restriction, furosemide, and NaCl supplementation. The authors assessed the primary outcome, change in sodium, at 4, 7, 14, and 28 days (baseline mean Na 125 mmol/L). By day 4, all groups had a significant increase in sodium (mean delta 5 mmol/L). The time to achieve a safe sodium level (Na less than 130 mmol/L) was not different among groups. Acute kidney injury was most common in patients who received furosemide and NaCl supplementation, compared with the fluid restriction and fluid restriction plus furosemide groups (32%, 10%, 17%, respectively; P = .07). Hypokalemia was also most common in the furosemide and NaCl group (42%, 13%, 23%, respectively; P = .01). Limitations include open-label study design, poor fluid restriction adherence (63% overall), and inflexible treatment regimens that excluded treatment with oral potassium.
Bottom line: In treatment of hyponatremia caused by SIADH, there was no benefit to adding furosemide with or without NaCl supplementation to fluid restriction. However, there was potential associated risk of acute kidney injury and hypokalemia.Citation: Krisanapan P et al. Efficacy of furosemide, oral sodium chloride, and fluid restriction for treatment of syndrome of inappropriate antidiuresis (SIADH): An open-label randomized controlled study (the EFFUSE-FLUID trial). Am J Kidney Dis. 2020 Aug;76(2):203-12. doi: 10.1053/j.ajkd.2019.11.012.
Dr. Welter is a hospitalist at Northwestern Memorial Hospital and instructor of medicine, Feinberg School of Medicine, both in Chicago.
Background: Hyponatremia caused by SIADH is common in hospitalized patients, and most evidence for treatment comes from noncontrolled studies. This study aims to investigate the efficacy and safety of fluid restriction compared with furosemide, with or without NaCl supplementation, for treating SIADH.
Study design: Open-label randomized controlled trial.
Setting: Single center in Thailand.
Synopsis: There were 92 participants randomized to fluid restriction alone, fluid restriction and furosemide, or fluid restriction, furosemide, and NaCl supplementation. The authors assessed the primary outcome, change in sodium, at 4, 7, 14, and 28 days (baseline mean Na 125 mmol/L). By day 4, all groups had a significant increase in sodium (mean delta 5 mmol/L). The time to achieve a safe sodium level (Na less than 130 mmol/L) was not different among groups. Acute kidney injury was most common in patients who received furosemide and NaCl supplementation, compared with the fluid restriction and fluid restriction plus furosemide groups (32%, 10%, 17%, respectively; P = .07). Hypokalemia was also most common in the furosemide and NaCl group (42%, 13%, 23%, respectively; P = .01). Limitations include open-label study design, poor fluid restriction adherence (63% overall), and inflexible treatment regimens that excluded treatment with oral potassium.
Bottom line: In treatment of hyponatremia caused by SIADH, there was no benefit to adding furosemide with or without NaCl supplementation to fluid restriction. However, there was potential associated risk of acute kidney injury and hypokalemia.Citation: Krisanapan P et al. Efficacy of furosemide, oral sodium chloride, and fluid restriction for treatment of syndrome of inappropriate antidiuresis (SIADH): An open-label randomized controlled study (the EFFUSE-FLUID trial). Am J Kidney Dis. 2020 Aug;76(2):203-12. doi: 10.1053/j.ajkd.2019.11.012.
Dr. Welter is a hospitalist at Northwestern Memorial Hospital and instructor of medicine, Feinberg School of Medicine, both in Chicago.
Fueling an ‘already raging fire’: Fifth COVID surge approaches
“A significant rise in cases just before Thanksgiving is not what we want to be seeing,” said Stephen Kissler, PhD, a postdoctoral researcher and data modeler at the Harvard TH Chan School of Public Health in Boston.
Dr. Kissler said he’d rather see increases in daily cases coming 2 weeks after busy travel periods, as that would mean they could come back down as people returned to their routines.
Seeing big increases in cases ahead of the holidays, he said, “is sort of like adding fuel to an already raging fire.”
Last winter, vaccines hadn’t been rolled out as the nation prepared for Thanksgiving. COVID-19 was burning through family gatherings.
But now that two-thirds of Americans over age 5 are fully vaccinated and booster doses are approved for all adults, will a rise in cases translate, once again, into a strain on our still thinly stretched healthcare system?
Experts say the vaccines are keeping people out of the hospital, which will help. And new antiviral pills are coming that seem to be able to cut a COVID-19 infection off at the knees, at least according to early data. A U.S. Food and Drug Administration panel meets next week to discuss the first application for a pill by Merck.
But experts caution that the coming surge will almost certainly tax hospitals again, especially in areas with lower vaccination rates.
And even states where blood testing shows that significant numbers of people have antibodies after a COVID-19 infection aren’t out of the woods, in part because we still don’t know how long the immunity generated by infection may last.
“Erosion of immunity”
“It’s hard to know how much risk is out there,” said Jeffrey Shaman, PhD, professor of environmental health sciences at Columbia University’s Mailman School of Public Health in New York City, who has been modeling the trajectory of the pandemic.
“We’re estimating, unfortunately, and we have for many weeks now, that there is an erosion of immunity,” Dr. Shaman said. “I think it could get bad. How bad? I’m not sure.”
Ali Mokdad, PhD, a professor of health metrics sciences at the University of Washington’s Institute for Health Metrics and Evaluation in Seattle, agrees.
Because there are so few studies on how long immunity from natural infection lasts, Dr. Mokdad and his colleagues are assuming that waning immunity after infection happens at least as quickly as it does after vaccination.
Their model is predicting that the average number of daily cases will peak at around 100,000, with another 100,000 going undetected, and will stay at that level until the end of January, as some states recover from their surges and others pick up steam.
While the number of daily deaths won’t climb to the heights seen during the summer surge, Dr. Mokdad said their model is predicting that daily deaths will climb again to about 1,200 a day.
“We are almost there right now, and it will be with us for a while,” he said. “We are predicting 881,000 deaths by March 1.”
The United States has currently recorded 773,000 COVID-19 deaths, so Dr. Mokdad is predicting about 120,000 more deaths between now and then.
He said his model shows that more than half of those deaths could be prevented if 95% of Americans wore their masks while in close proximity to strangers.
Currently, only about 36% of Americans are consistently wearing masks, according to surveys. While people are moving around more now, mobility is at prepandemic levels in some states.
“The rise that you are seeing right now is high mobility and low mask wearing in the United States,” Dr. Mokdad said.
The solution, he said, is for all adults to get another dose of vaccine — he doesn’t like calling it a booster.
“Because they’re vaccinated and they have two doses they have a false sense of security that they are protected. We needed to come ahead of it immediately and say you need a third dose, and we were late to do so,” Dr. Mokdad said.
A version of this article first appeared on Medscape.com.
“A significant rise in cases just before Thanksgiving is not what we want to be seeing,” said Stephen Kissler, PhD, a postdoctoral researcher and data modeler at the Harvard TH Chan School of Public Health in Boston.
Dr. Kissler said he’d rather see increases in daily cases coming 2 weeks after busy travel periods, as that would mean they could come back down as people returned to their routines.
Seeing big increases in cases ahead of the holidays, he said, “is sort of like adding fuel to an already raging fire.”
Last winter, vaccines hadn’t been rolled out as the nation prepared for Thanksgiving. COVID-19 was burning through family gatherings.
But now that two-thirds of Americans over age 5 are fully vaccinated and booster doses are approved for all adults, will a rise in cases translate, once again, into a strain on our still thinly stretched healthcare system?
Experts say the vaccines are keeping people out of the hospital, which will help. And new antiviral pills are coming that seem to be able to cut a COVID-19 infection off at the knees, at least according to early data. A U.S. Food and Drug Administration panel meets next week to discuss the first application for a pill by Merck.
But experts caution that the coming surge will almost certainly tax hospitals again, especially in areas with lower vaccination rates.
And even states where blood testing shows that significant numbers of people have antibodies after a COVID-19 infection aren’t out of the woods, in part because we still don’t know how long the immunity generated by infection may last.
“Erosion of immunity”
“It’s hard to know how much risk is out there,” said Jeffrey Shaman, PhD, professor of environmental health sciences at Columbia University’s Mailman School of Public Health in New York City, who has been modeling the trajectory of the pandemic.
“We’re estimating, unfortunately, and we have for many weeks now, that there is an erosion of immunity,” Dr. Shaman said. “I think it could get bad. How bad? I’m not sure.”
Ali Mokdad, PhD, a professor of health metrics sciences at the University of Washington’s Institute for Health Metrics and Evaluation in Seattle, agrees.
Because there are so few studies on how long immunity from natural infection lasts, Dr. Mokdad and his colleagues are assuming that waning immunity after infection happens at least as quickly as it does after vaccination.
Their model is predicting that the average number of daily cases will peak at around 100,000, with another 100,000 going undetected, and will stay at that level until the end of January, as some states recover from their surges and others pick up steam.
While the number of daily deaths won’t climb to the heights seen during the summer surge, Dr. Mokdad said their model is predicting that daily deaths will climb again to about 1,200 a day.
“We are almost there right now, and it will be with us for a while,” he said. “We are predicting 881,000 deaths by March 1.”
The United States has currently recorded 773,000 COVID-19 deaths, so Dr. Mokdad is predicting about 120,000 more deaths between now and then.
He said his model shows that more than half of those deaths could be prevented if 95% of Americans wore their masks while in close proximity to strangers.
Currently, only about 36% of Americans are consistently wearing masks, according to surveys. While people are moving around more now, mobility is at prepandemic levels in some states.
“The rise that you are seeing right now is high mobility and low mask wearing in the United States,” Dr. Mokdad said.
The solution, he said, is for all adults to get another dose of vaccine — he doesn’t like calling it a booster.
“Because they’re vaccinated and they have two doses they have a false sense of security that they are protected. We needed to come ahead of it immediately and say you need a third dose, and we were late to do so,” Dr. Mokdad said.
A version of this article first appeared on Medscape.com.
“A significant rise in cases just before Thanksgiving is not what we want to be seeing,” said Stephen Kissler, PhD, a postdoctoral researcher and data modeler at the Harvard TH Chan School of Public Health in Boston.
Dr. Kissler said he’d rather see increases in daily cases coming 2 weeks after busy travel periods, as that would mean they could come back down as people returned to their routines.
Seeing big increases in cases ahead of the holidays, he said, “is sort of like adding fuel to an already raging fire.”
Last winter, vaccines hadn’t been rolled out as the nation prepared for Thanksgiving. COVID-19 was burning through family gatherings.
But now that two-thirds of Americans over age 5 are fully vaccinated and booster doses are approved for all adults, will a rise in cases translate, once again, into a strain on our still thinly stretched healthcare system?
Experts say the vaccines are keeping people out of the hospital, which will help. And new antiviral pills are coming that seem to be able to cut a COVID-19 infection off at the knees, at least according to early data. A U.S. Food and Drug Administration panel meets next week to discuss the first application for a pill by Merck.
But experts caution that the coming surge will almost certainly tax hospitals again, especially in areas with lower vaccination rates.
And even states where blood testing shows that significant numbers of people have antibodies after a COVID-19 infection aren’t out of the woods, in part because we still don’t know how long the immunity generated by infection may last.
“Erosion of immunity”
“It’s hard to know how much risk is out there,” said Jeffrey Shaman, PhD, professor of environmental health sciences at Columbia University’s Mailman School of Public Health in New York City, who has been modeling the trajectory of the pandemic.
“We’re estimating, unfortunately, and we have for many weeks now, that there is an erosion of immunity,” Dr. Shaman said. “I think it could get bad. How bad? I’m not sure.”
Ali Mokdad, PhD, a professor of health metrics sciences at the University of Washington’s Institute for Health Metrics and Evaluation in Seattle, agrees.
Because there are so few studies on how long immunity from natural infection lasts, Dr. Mokdad and his colleagues are assuming that waning immunity after infection happens at least as quickly as it does after vaccination.
Their model is predicting that the average number of daily cases will peak at around 100,000, with another 100,000 going undetected, and will stay at that level until the end of January, as some states recover from their surges and others pick up steam.
While the number of daily deaths won’t climb to the heights seen during the summer surge, Dr. Mokdad said their model is predicting that daily deaths will climb again to about 1,200 a day.
“We are almost there right now, and it will be with us for a while,” he said. “We are predicting 881,000 deaths by March 1.”
The United States has currently recorded 773,000 COVID-19 deaths, so Dr. Mokdad is predicting about 120,000 more deaths between now and then.
He said his model shows that more than half of those deaths could be prevented if 95% of Americans wore their masks while in close proximity to strangers.
Currently, only about 36% of Americans are consistently wearing masks, according to surveys. While people are moving around more now, mobility is at prepandemic levels in some states.
“The rise that you are seeing right now is high mobility and low mask wearing in the United States,” Dr. Mokdad said.
The solution, he said, is for all adults to get another dose of vaccine — he doesn’t like calling it a booster.
“Because they’re vaccinated and they have two doses they have a false sense of security that they are protected. We needed to come ahead of it immediately and say you need a third dose, and we were late to do so,” Dr. Mokdad said.
A version of this article first appeared on Medscape.com.
Children and COVID: New cases increase for third straight week
There were almost 142,000 new cases reported during the week of Nov. 12-18, marking an increase of 16% over the previous week and the 15th straight week with a weekly total over 100,000, the American Academy of Pediatrics and the Children’s Hospital Association said.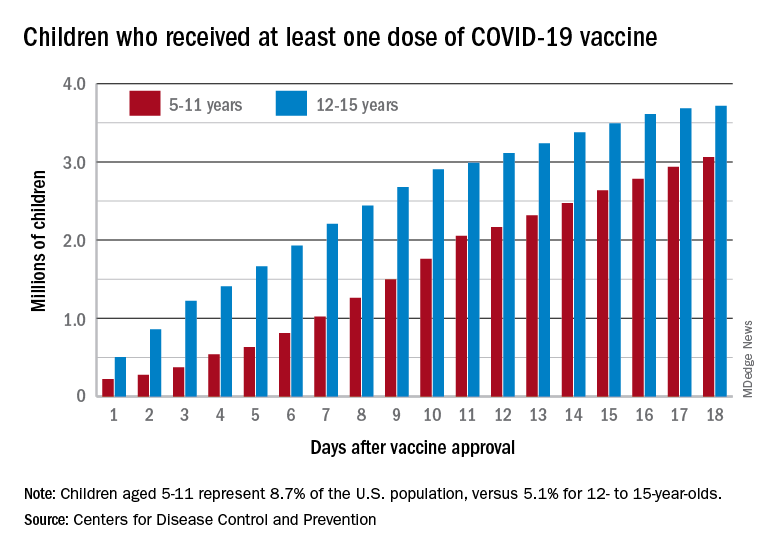
Regional data show that the Midwest has experienced the largest share of this latest surge, followed by the Northeast. Cases increased in the South during the week of Nov. 12-18 after holding steady over the previous 2 weeks, while new cases in the West dropped in the last week. At the state level, Maine, New Hampshire, and Vermont again reported the largest percent increases, with Michigan, Minnesota, and New Mexico also above average, the AAP and CHA said in their weekly COVID report.
Data from the Centers for Disease Control and Prevention show similar trends for both emergency department visits and hospital admissions, as both have risen in November after declines that began in late August and early September.
The cumulative number of pediatric cases is 6.77 million since the pandemic began, based on the AAP/CHA accounting of state cases, although Alabama, Nebraska, and Texas stopped reporting over the summer, suggesting the actual number is higher. The CDC puts the total number of COVID cases in children at 5.96 million, but there are age discrepancies between the CDC and the AAP/CHA’s state-based data.
The vaccine gap is closing
Vaccinations among the recently eligible 5- to 11-year-olds have steadily increased following a somewhat slow start. The initial pace was behind that of the 12- to 15-years-olds through the first postapproval week but has since closed the gap, based on data from the CDC’s COVID Data Tracker.
The tally of children who received at least one dose of the COVID vaccine among the 5- to 11-year-olds was behind the older group by almost 1.2 million on day 7 after the CDC’s Nov. 2 approval, but by day 18 the deficit was down to about 650,000, the CDC reported.
Altogether, just over 3 million children aged 5-11 have received at least one dose, which is 10.7% of that age group’s total population. Among children aged 12-17, the proportions are 60.7% with at least one dose and 51.1% at full vaccination. Children aged 5-11, who make up 8.7% of the total U.S. population, represented 42.8% of all vaccinations initiated over the 2 weeks ending Nov. 21, compared with 4.2% for those aged 12-17, the CDC said.
There were almost 142,000 new cases reported during the week of Nov. 12-18, marking an increase of 16% over the previous week and the 15th straight week with a weekly total over 100,000, the American Academy of Pediatrics and the Children’s Hospital Association said.
Regional data show that the Midwest has experienced the largest share of this latest surge, followed by the Northeast. Cases increased in the South during the week of Nov. 12-18 after holding steady over the previous 2 weeks, while new cases in the West dropped in the last week. At the state level, Maine, New Hampshire, and Vermont again reported the largest percent increases, with Michigan, Minnesota, and New Mexico also above average, the AAP and CHA said in their weekly COVID report.
Data from the Centers for Disease Control and Prevention show similar trends for both emergency department visits and hospital admissions, as both have risen in November after declines that began in late August and early September.
The cumulative number of pediatric cases is 6.77 million since the pandemic began, based on the AAP/CHA accounting of state cases, although Alabama, Nebraska, and Texas stopped reporting over the summer, suggesting the actual number is higher. The CDC puts the total number of COVID cases in children at 5.96 million, but there are age discrepancies between the CDC and the AAP/CHA’s state-based data.
The vaccine gap is closing
Vaccinations among the recently eligible 5- to 11-year-olds have steadily increased following a somewhat slow start. The initial pace was behind that of the 12- to 15-years-olds through the first postapproval week but has since closed the gap, based on data from the CDC’s COVID Data Tracker.
The tally of children who received at least one dose of the COVID vaccine among the 5- to 11-year-olds was behind the older group by almost 1.2 million on day 7 after the CDC’s Nov. 2 approval, but by day 18 the deficit was down to about 650,000, the CDC reported.
Altogether, just over 3 million children aged 5-11 have received at least one dose, which is 10.7% of that age group’s total population. Among children aged 12-17, the proportions are 60.7% with at least one dose and 51.1% at full vaccination. Children aged 5-11, who make up 8.7% of the total U.S. population, represented 42.8% of all vaccinations initiated over the 2 weeks ending Nov. 21, compared with 4.2% for those aged 12-17, the CDC said.
There were almost 142,000 new cases reported during the week of Nov. 12-18, marking an increase of 16% over the previous week and the 15th straight week with a weekly total over 100,000, the American Academy of Pediatrics and the Children’s Hospital Association said.
Regional data show that the Midwest has experienced the largest share of this latest surge, followed by the Northeast. Cases increased in the South during the week of Nov. 12-18 after holding steady over the previous 2 weeks, while new cases in the West dropped in the last week. At the state level, Maine, New Hampshire, and Vermont again reported the largest percent increases, with Michigan, Minnesota, and New Mexico also above average, the AAP and CHA said in their weekly COVID report.
Data from the Centers for Disease Control and Prevention show similar trends for both emergency department visits and hospital admissions, as both have risen in November after declines that began in late August and early September.
The cumulative number of pediatric cases is 6.77 million since the pandemic began, based on the AAP/CHA accounting of state cases, although Alabama, Nebraska, and Texas stopped reporting over the summer, suggesting the actual number is higher. The CDC puts the total number of COVID cases in children at 5.96 million, but there are age discrepancies between the CDC and the AAP/CHA’s state-based data.
The vaccine gap is closing
Vaccinations among the recently eligible 5- to 11-year-olds have steadily increased following a somewhat slow start. The initial pace was behind that of the 12- to 15-years-olds through the first postapproval week but has since closed the gap, based on data from the CDC’s COVID Data Tracker.
The tally of children who received at least one dose of the COVID vaccine among the 5- to 11-year-olds was behind the older group by almost 1.2 million on day 7 after the CDC’s Nov. 2 approval, but by day 18 the deficit was down to about 650,000, the CDC reported.
Altogether, just over 3 million children aged 5-11 have received at least one dose, which is 10.7% of that age group’s total population. Among children aged 12-17, the proportions are 60.7% with at least one dose and 51.1% at full vaccination. Children aged 5-11, who make up 8.7% of the total U.S. population, represented 42.8% of all vaccinations initiated over the 2 weeks ending Nov. 21, compared with 4.2% for those aged 12-17, the CDC said.
Short-acting opioids needed for withdrawal in U.S. hospitals, say experts
The commentary by Robert A. Kleinman, MD, with the Centre for Addiction and Mental Health, and department of psychiatry, University of Toronto, and Sarah E. Wakeman, MD, with the division of general internal medicine at Massachusetts General Hospital, and Harvard Medical School, Boston, was published in Annals of Internal Medicine.
Currently, short-acting opioids are not recommended in the United States for opioid withdrawal symptoms (OWS) management in the hospital, the authors wrote. Instead, withdrawal symptoms are typically treated, followed by methadone or buprenorphine or nonopioid medications, but many patients don’t get enough relief. Undertreated withdrawal can result in patients leaving the hospital against medical advice, which is linked with higher risk of death.
Addiction specialist Elisabeth Poorman, MD, of the University of Illinois Chicago, said in an interview that she agrees it’s time to start shifting the thinking on using short-acting opioids for OWS in hospitals. Use varies greatly by hospital and by clinician, she said.
“It’s time to let evidence guide us and to be flexible,” Dr. Poorman said.
The commentary authors noted that with methadone, patients must wait several hours for maximal symptom reduction, and the full benefits of methadone treatment are not realized until days after initiation.
Rapid initiation of methadone may be feasible in hospitals and has been proposed as an option, but further study is necessary before widespread use, the authors wrote.
Short-acting opioids may address limitations of other opioids
Lofexidine, an alpha-2-adrenergic agonist, is the only drug approved by the Food and Drug Administration specifically for OWS.
“However,” the authors said, “more than half of patients with OWS treated with lofexidine in phase 3 efficacy trials dropped out by day five. Clonidine, another alpha-2-agonist used off label to treat OWS, has similar effects to those of lofexidine. “
Therefore, short-acting opioids may complement methadone and buprenorphine in treating OWS in the hospital by addressing their limitations, the authors wrote.
Dr. Kleinman and Dr. Wakeman also say short-acting opioids may help with starting buprenorphine for patients exposed to fentanyl, because short-acting opioids can relieve withdrawal symptoms while fentanyl is metabolized and excreted.
Supplementation with short-acting opioids within the hospital can relieve withdrawal symptoms and help keep patients comfortable while methadone is titrated to more effective doses for long-term treatment, they wrote.
With short-acting opioids, patients may become more engaged in their care with, for example, a tamper-proof, patient-controlled analgesia pump, which would allow them to have more autonomy in administration of opioids to relieve pain and withdrawal symptoms, the authors wrote.
Dr. Kleinman and Dr. Wakeman noted that many patients who inject drugs already consume short-acting illicit drugs in the hospital, typically in washrooms and smoking areas, so supervised use of short-acting opioids helps eliminate the risk for unwitnessed overdoses.
Barriers to short-acting opioid use
Despite use of short-acting opioids internationally, barriers in the United States include limited prospective, randomized, controlled research on their benefits. There is limited institutional support for such approaches, and concerns and stigma around providing opioids to patients with OUD.
“[M]any institutions have insufficient numbers of providers who are both confident and competent with standard buprenorphine and methadone initiation approaches, a prerequisite before adopting more complex regimens,” the authors wrote.
Short-acting, full-agonist opioids, as a complement to methadone or buprenorphine, is already recommended for inpatients with OUD who are experiencing acute pain.
But the authors argue it should be an option when pain is not present, but methadone or buprenorphine have not provided enough OWS relief.
When short-acting opioids are helpful, according to outside expert
Dr. Poorman agrees and says she has found short-acting opioids simple to use in the hospital and very helpful in two situations.
One is when patients are very clear that they don’t want any medication for opioid use disorder, but they do want to be treated for their acute medical issue.
“I thought that was a fantastic tool to have to demonstrate we’re listening to them and weren’t trying to impose something on them and left the door open to come back when they did want treatment, which many of them did,” Dr. Poorman said.
The second situation is when the patient is uncertain about options but very afraid of precipitated withdrawal from buprenorphine.
She said she then found it easy to switch from those medications to buprenorphine and methadone.
Dr. Poorman described a situation she encountered previously where the patient was injecting heroin several times a day for 30-40 years. He was very clear he wasn’t going to stop injecting heroin, but he needed medical attention. He was willing to get medical attention, but he told his doctor he didn’t want to be uncomfortable while in the hospital.
It was very hard for his doctor to accept relieving his symptoms of withdrawal as part of her job, because she felt as though she was condoning his drug use, Dr. Poorman explained.
But Dr. Poorman said it’s not realistic to think that someone who clearly does not want to stop using is going to stop using because a doctor made that person go through painful withdrawal “that they’ve structured their whole life around avoiding.”
Take-home message
“We need to understand that addiction is very complex. A lot of times people come to us distressed, and it’s a great time to engage them in care but engaging them in care doesn’t mean imposing discomfort or pain on them,” Dr. Poorman noted. Instead, it means “listening to them, helping them be comfortable in a really stressful situation and then letting them know we are always there for them wherever they are on their disease process or recovery journey so that they can come back to us.”
Dr. Wakeman previously served on clinical advisory board for Celero Systems and receives textbook royalties from Springer and author payment from UpToDate. Dr. Kleinman and Dr. Poorman declared no relevant financial relationships.
The commentary by Robert A. Kleinman, MD, with the Centre for Addiction and Mental Health, and department of psychiatry, University of Toronto, and Sarah E. Wakeman, MD, with the division of general internal medicine at Massachusetts General Hospital, and Harvard Medical School, Boston, was published in Annals of Internal Medicine.
Currently, short-acting opioids are not recommended in the United States for opioid withdrawal symptoms (OWS) management in the hospital, the authors wrote. Instead, withdrawal symptoms are typically treated, followed by methadone or buprenorphine or nonopioid medications, but many patients don’t get enough relief. Undertreated withdrawal can result in patients leaving the hospital against medical advice, which is linked with higher risk of death.
Addiction specialist Elisabeth Poorman, MD, of the University of Illinois Chicago, said in an interview that she agrees it’s time to start shifting the thinking on using short-acting opioids for OWS in hospitals. Use varies greatly by hospital and by clinician, she said.
“It’s time to let evidence guide us and to be flexible,” Dr. Poorman said.
The commentary authors noted that with methadone, patients must wait several hours for maximal symptom reduction, and the full benefits of methadone treatment are not realized until days after initiation.
Rapid initiation of methadone may be feasible in hospitals and has been proposed as an option, but further study is necessary before widespread use, the authors wrote.
Short-acting opioids may address limitations of other opioids
Lofexidine, an alpha-2-adrenergic agonist, is the only drug approved by the Food and Drug Administration specifically for OWS.
“However,” the authors said, “more than half of patients with OWS treated with lofexidine in phase 3 efficacy trials dropped out by day five. Clonidine, another alpha-2-agonist used off label to treat OWS, has similar effects to those of lofexidine. “
Therefore, short-acting opioids may complement methadone and buprenorphine in treating OWS in the hospital by addressing their limitations, the authors wrote.
Dr. Kleinman and Dr. Wakeman also say short-acting opioids may help with starting buprenorphine for patients exposed to fentanyl, because short-acting opioids can relieve withdrawal symptoms while fentanyl is metabolized and excreted.
Supplementation with short-acting opioids within the hospital can relieve withdrawal symptoms and help keep patients comfortable while methadone is titrated to more effective doses for long-term treatment, they wrote.
With short-acting opioids, patients may become more engaged in their care with, for example, a tamper-proof, patient-controlled analgesia pump, which would allow them to have more autonomy in administration of opioids to relieve pain and withdrawal symptoms, the authors wrote.
Dr. Kleinman and Dr. Wakeman noted that many patients who inject drugs already consume short-acting illicit drugs in the hospital, typically in washrooms and smoking areas, so supervised use of short-acting opioids helps eliminate the risk for unwitnessed overdoses.
Barriers to short-acting opioid use
Despite use of short-acting opioids internationally, barriers in the United States include limited prospective, randomized, controlled research on their benefits. There is limited institutional support for such approaches, and concerns and stigma around providing opioids to patients with OUD.
“[M]any institutions have insufficient numbers of providers who are both confident and competent with standard buprenorphine and methadone initiation approaches, a prerequisite before adopting more complex regimens,” the authors wrote.
Short-acting, full-agonist opioids, as a complement to methadone or buprenorphine, is already recommended for inpatients with OUD who are experiencing acute pain.
But the authors argue it should be an option when pain is not present, but methadone or buprenorphine have not provided enough OWS relief.
When short-acting opioids are helpful, according to outside expert
Dr. Poorman agrees and says she has found short-acting opioids simple to use in the hospital and very helpful in two situations.
One is when patients are very clear that they don’t want any medication for opioid use disorder, but they do want to be treated for their acute medical issue.
“I thought that was a fantastic tool to have to demonstrate we’re listening to them and weren’t trying to impose something on them and left the door open to come back when they did want treatment, which many of them did,” Dr. Poorman said.
The second situation is when the patient is uncertain about options but very afraid of precipitated withdrawal from buprenorphine.
She said she then found it easy to switch from those medications to buprenorphine and methadone.
Dr. Poorman described a situation she encountered previously where the patient was injecting heroin several times a day for 30-40 years. He was very clear he wasn’t going to stop injecting heroin, but he needed medical attention. He was willing to get medical attention, but he told his doctor he didn’t want to be uncomfortable while in the hospital.
It was very hard for his doctor to accept relieving his symptoms of withdrawal as part of her job, because she felt as though she was condoning his drug use, Dr. Poorman explained.
But Dr. Poorman said it’s not realistic to think that someone who clearly does not want to stop using is going to stop using because a doctor made that person go through painful withdrawal “that they’ve structured their whole life around avoiding.”
Take-home message
“We need to understand that addiction is very complex. A lot of times people come to us distressed, and it’s a great time to engage them in care but engaging them in care doesn’t mean imposing discomfort or pain on them,” Dr. Poorman noted. Instead, it means “listening to them, helping them be comfortable in a really stressful situation and then letting them know we are always there for them wherever they are on their disease process or recovery journey so that they can come back to us.”
Dr. Wakeman previously served on clinical advisory board for Celero Systems and receives textbook royalties from Springer and author payment from UpToDate. Dr. Kleinman and Dr. Poorman declared no relevant financial relationships.
The commentary by Robert A. Kleinman, MD, with the Centre for Addiction and Mental Health, and department of psychiatry, University of Toronto, and Sarah E. Wakeman, MD, with the division of general internal medicine at Massachusetts General Hospital, and Harvard Medical School, Boston, was published in Annals of Internal Medicine.
Currently, short-acting opioids are not recommended in the United States for opioid withdrawal symptoms (OWS) management in the hospital, the authors wrote. Instead, withdrawal symptoms are typically treated, followed by methadone or buprenorphine or nonopioid medications, but many patients don’t get enough relief. Undertreated withdrawal can result in patients leaving the hospital against medical advice, which is linked with higher risk of death.
Addiction specialist Elisabeth Poorman, MD, of the University of Illinois Chicago, said in an interview that she agrees it’s time to start shifting the thinking on using short-acting opioids for OWS in hospitals. Use varies greatly by hospital and by clinician, she said.
“It’s time to let evidence guide us and to be flexible,” Dr. Poorman said.
The commentary authors noted that with methadone, patients must wait several hours for maximal symptom reduction, and the full benefits of methadone treatment are not realized until days after initiation.
Rapid initiation of methadone may be feasible in hospitals and has been proposed as an option, but further study is necessary before widespread use, the authors wrote.
Short-acting opioids may address limitations of other opioids
Lofexidine, an alpha-2-adrenergic agonist, is the only drug approved by the Food and Drug Administration specifically for OWS.
“However,” the authors said, “more than half of patients with OWS treated with lofexidine in phase 3 efficacy trials dropped out by day five. Clonidine, another alpha-2-agonist used off label to treat OWS, has similar effects to those of lofexidine. “
Therefore, short-acting opioids may complement methadone and buprenorphine in treating OWS in the hospital by addressing their limitations, the authors wrote.
Dr. Kleinman and Dr. Wakeman also say short-acting opioids may help with starting buprenorphine for patients exposed to fentanyl, because short-acting opioids can relieve withdrawal symptoms while fentanyl is metabolized and excreted.
Supplementation with short-acting opioids within the hospital can relieve withdrawal symptoms and help keep patients comfortable while methadone is titrated to more effective doses for long-term treatment, they wrote.
With short-acting opioids, patients may become more engaged in their care with, for example, a tamper-proof, patient-controlled analgesia pump, which would allow them to have more autonomy in administration of opioids to relieve pain and withdrawal symptoms, the authors wrote.
Dr. Kleinman and Dr. Wakeman noted that many patients who inject drugs already consume short-acting illicit drugs in the hospital, typically in washrooms and smoking areas, so supervised use of short-acting opioids helps eliminate the risk for unwitnessed overdoses.
Barriers to short-acting opioid use
Despite use of short-acting opioids internationally, barriers in the United States include limited prospective, randomized, controlled research on their benefits. There is limited institutional support for such approaches, and concerns and stigma around providing opioids to patients with OUD.
“[M]any institutions have insufficient numbers of providers who are both confident and competent with standard buprenorphine and methadone initiation approaches, a prerequisite before adopting more complex regimens,” the authors wrote.
Short-acting, full-agonist opioids, as a complement to methadone or buprenorphine, is already recommended for inpatients with OUD who are experiencing acute pain.
But the authors argue it should be an option when pain is not present, but methadone or buprenorphine have not provided enough OWS relief.
When short-acting opioids are helpful, according to outside expert
Dr. Poorman agrees and says she has found short-acting opioids simple to use in the hospital and very helpful in two situations.
One is when patients are very clear that they don’t want any medication for opioid use disorder, but they do want to be treated for their acute medical issue.
“I thought that was a fantastic tool to have to demonstrate we’re listening to them and weren’t trying to impose something on them and left the door open to come back when they did want treatment, which many of them did,” Dr. Poorman said.
The second situation is when the patient is uncertain about options but very afraid of precipitated withdrawal from buprenorphine.
She said she then found it easy to switch from those medications to buprenorphine and methadone.
Dr. Poorman described a situation she encountered previously where the patient was injecting heroin several times a day for 30-40 years. He was very clear he wasn’t going to stop injecting heroin, but he needed medical attention. He was willing to get medical attention, but he told his doctor he didn’t want to be uncomfortable while in the hospital.
It was very hard for his doctor to accept relieving his symptoms of withdrawal as part of her job, because she felt as though she was condoning his drug use, Dr. Poorman explained.
But Dr. Poorman said it’s not realistic to think that someone who clearly does not want to stop using is going to stop using because a doctor made that person go through painful withdrawal “that they’ve structured their whole life around avoiding.”
Take-home message
“We need to understand that addiction is very complex. A lot of times people come to us distressed, and it’s a great time to engage them in care but engaging them in care doesn’t mean imposing discomfort or pain on them,” Dr. Poorman noted. Instead, it means “listening to them, helping them be comfortable in a really stressful situation and then letting them know we are always there for them wherever they are on their disease process or recovery journey so that they can come back to us.”
Dr. Wakeman previously served on clinical advisory board for Celero Systems and receives textbook royalties from Springer and author payment from UpToDate. Dr. Kleinman and Dr. Poorman declared no relevant financial relationships.
FROM ANNALS OF INTERNAL MEDICINE
‘Misleading’ results in colchicine COVID-19 trials meta-analysis
A new meta-analysis appears to show that colchicine has no benefit as a treatment for COVID-19, but its inclusion of trials studying differing patient populations and testing different outcomes led to “misleading” results, says a researcher involved in one of the trials.
The meta-analysis, which includes data from the recent Randomised Evaluation of COVID-19 Therapy (RECOVERY) trial, was published Nov. 22 in RMD Open.
Kedar Gautambhai Mehta, MBBS, MD, of the GMERS Medical College Gotri in Vadodara, India, and colleagues included outcomes from six studies of 16,148 patients with COVID-19 who received colchicine or supportive care. They evaluated the efficacy outcomes of mortality, need for ventilation, intensive care unit admission, and length of stay in hospital, as well as safety outcomes of adverse events, serious adverse events, and diarrhea.
The studies in the meta-analysis included a randomized, controlled trial (RCT) of 105 patients hospitalized with COVID-19 in Greece, the international, open-label RECOVERY RCT of 11,340 patients hospitalized with COVID-19, an RCT of 72 hospitalized patients with moderate or severe COVID-19 in Brazil, an RCT of 100 patients hospitalized with COVID-19 in Iran, the international COLCORONA trial of 4,488 patients with COVID-19 who were treated with colchicine or placebo on an outpatient basis, and the randomized COLORIT trial of 43 patients hospitalized with COVID-19 in Russia.
Studies “asked very different questions” about colchicine
Commenting on the meta-analysis, Michael H. Pillinger, MD, a rheumatologist and professor of medicine, biochemistry, and molecular pharmacology with New York University, said the authors combined studies “that are not comparable and that asked very different questions.” Two of the studies in the meta-analysis are very large, and four are very small, which skews the results, he explained.
“The larger studies therefore drive the outcome, and while the small studies are potentially insight providing, the large studies are the only ones worth giving our attention to in the context of the meta-analysis,” he said. The two largest studies – RECOVERY and COLCORONA – taken together show no benefit for colchicine as a treatment, even though the former demonstrated no benefit and the latter did show a benefit, explained Dr. Pillinger, a co–principal investigator for the COLCORONA trial in the United States.
The studies were designed differently and should not have been included in the same analysis, Dr. Pillinger argued. In the case of COLCORONA, early treatment with colchicine was the intervention, whereas RECOVERY focused on hospitalized patients.
“In designing [COLCORONA], the author group (of whom I was a member) expressly rejected the idea that colchicine might be useful for the sicker hospitalized patients, based on the long experience with colchicine of some of us as rheumatologists,” Dr. Pillinger said.
“In short, COLCORONA proved a benefit of colchicine in outpatient COVID-19, and its authors presumed there would be no inpatient benefit; RECOVERY went ahead and proved a lack of inpatient benefit, at least when high-dose steroids were also given,” he said. “While there is no conflict between these results, the combination of the two studies in this meta-analysis suggests there might be no benefit for colchicine overall, which is misleading and can lead physicians to reject the potential of outpatient colchicine, even for future studies.”
Dr. Pillinger said he still believes colchicine has potential value as a COVID-19 treatment option for patients with mild disease, “especially for low–vaccine rate, resource-starved countries.
“It would be unfortunate if meta-analyses such as this one would put a stop to colchicine’s use, or at least its further investigation,” he said.
Study details
The authors of the study assessed heterogeneity of the trials’ data across the outcomes using an I2 test. They evaluated the quality of the evidence for the outcomes using the Grades of Recommendation, Assessment, Development and Evaluation (GRADE).
The results of their meta-analysis showed that colchicine offered no significant improvement in mortality in six studies (risk difference, –0.0; 95% confidence interval, –0.01 to 0.01; I2 = 15%). It showed no benefit with respect to requiring ventilatory support in five studies of 15,519 patients (risk ratio, 0.67; 95% CI, 0.38-1.21; I2 = 47%); being admitted to the ICU in three studies with 220 patients (RR, 0.49; 95% CI, 0.19-1.25; I2 = 34%); and length of stay while in the hospital in four studies of 11,560 patients (mean difference, –1.17; 95% CI, –3.02 to 0.67; I2 = 77%).
There was no difference in serious adverse events in three studies with 4,665 patients (RD, –0.01; 95% CI, –0.02 to 0.00; I2 = 28%) for patients who received colchicine, compared with supportive care alone. Patients who received colchicine were more likely to have a higher rate of adverse events (RR, 1.58; 95% CI, 1.07-2.33; I2 = 81%) and to experience diarrhea (RR, 1.93; 95% CI, 1.62-2.29; I2 = 0%) than were patients who received supportive care alone. The researchers note that for most outcomes, the GRADE quality of evidence was moderate.
“Our findings on colchicine should be interpreted cautiously due to the inclusion of open-labeled, randomized clinical trials,” Dr. Mehta and colleagues write. “The analysis of efficacy and safety outcomes are based on a small number of RCTs in control interventions.”
The authors reported no relevant financial relationships. Dr. Pillinger is co–principal investigator of the U.S. component of the COLCORONA trial; he reported no other relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
A new meta-analysis appears to show that colchicine has no benefit as a treatment for COVID-19, but its inclusion of trials studying differing patient populations and testing different outcomes led to “misleading” results, says a researcher involved in one of the trials.
The meta-analysis, which includes data from the recent Randomised Evaluation of COVID-19 Therapy (RECOVERY) trial, was published Nov. 22 in RMD Open.
Kedar Gautambhai Mehta, MBBS, MD, of the GMERS Medical College Gotri in Vadodara, India, and colleagues included outcomes from six studies of 16,148 patients with COVID-19 who received colchicine or supportive care. They evaluated the efficacy outcomes of mortality, need for ventilation, intensive care unit admission, and length of stay in hospital, as well as safety outcomes of adverse events, serious adverse events, and diarrhea.
The studies in the meta-analysis included a randomized, controlled trial (RCT) of 105 patients hospitalized with COVID-19 in Greece, the international, open-label RECOVERY RCT of 11,340 patients hospitalized with COVID-19, an RCT of 72 hospitalized patients with moderate or severe COVID-19 in Brazil, an RCT of 100 patients hospitalized with COVID-19 in Iran, the international COLCORONA trial of 4,488 patients with COVID-19 who were treated with colchicine or placebo on an outpatient basis, and the randomized COLORIT trial of 43 patients hospitalized with COVID-19 in Russia.
Studies “asked very different questions” about colchicine
Commenting on the meta-analysis, Michael H. Pillinger, MD, a rheumatologist and professor of medicine, biochemistry, and molecular pharmacology with New York University, said the authors combined studies “that are not comparable and that asked very different questions.” Two of the studies in the meta-analysis are very large, and four are very small, which skews the results, he explained.
“The larger studies therefore drive the outcome, and while the small studies are potentially insight providing, the large studies are the only ones worth giving our attention to in the context of the meta-analysis,” he said. The two largest studies – RECOVERY and COLCORONA – taken together show no benefit for colchicine as a treatment, even though the former demonstrated no benefit and the latter did show a benefit, explained Dr. Pillinger, a co–principal investigator for the COLCORONA trial in the United States.
The studies were designed differently and should not have been included in the same analysis, Dr. Pillinger argued. In the case of COLCORONA, early treatment with colchicine was the intervention, whereas RECOVERY focused on hospitalized patients.
“In designing [COLCORONA], the author group (of whom I was a member) expressly rejected the idea that colchicine might be useful for the sicker hospitalized patients, based on the long experience with colchicine of some of us as rheumatologists,” Dr. Pillinger said.
“In short, COLCORONA proved a benefit of colchicine in outpatient COVID-19, and its authors presumed there would be no inpatient benefit; RECOVERY went ahead and proved a lack of inpatient benefit, at least when high-dose steroids were also given,” he said. “While there is no conflict between these results, the combination of the two studies in this meta-analysis suggests there might be no benefit for colchicine overall, which is misleading and can lead physicians to reject the potential of outpatient colchicine, even for future studies.”
Dr. Pillinger said he still believes colchicine has potential value as a COVID-19 treatment option for patients with mild disease, “especially for low–vaccine rate, resource-starved countries.
“It would be unfortunate if meta-analyses such as this one would put a stop to colchicine’s use, or at least its further investigation,” he said.
Study details
The authors of the study assessed heterogeneity of the trials’ data across the outcomes using an I2 test. They evaluated the quality of the evidence for the outcomes using the Grades of Recommendation, Assessment, Development and Evaluation (GRADE).
The results of their meta-analysis showed that colchicine offered no significant improvement in mortality in six studies (risk difference, –0.0; 95% confidence interval, –0.01 to 0.01; I2 = 15%). It showed no benefit with respect to requiring ventilatory support in five studies of 15,519 patients (risk ratio, 0.67; 95% CI, 0.38-1.21; I2 = 47%); being admitted to the ICU in three studies with 220 patients (RR, 0.49; 95% CI, 0.19-1.25; I2 = 34%); and length of stay while in the hospital in four studies of 11,560 patients (mean difference, –1.17; 95% CI, –3.02 to 0.67; I2 = 77%).
There was no difference in serious adverse events in three studies with 4,665 patients (RD, –0.01; 95% CI, –0.02 to 0.00; I2 = 28%) for patients who received colchicine, compared with supportive care alone. Patients who received colchicine were more likely to have a higher rate of adverse events (RR, 1.58; 95% CI, 1.07-2.33; I2 = 81%) and to experience diarrhea (RR, 1.93; 95% CI, 1.62-2.29; I2 = 0%) than were patients who received supportive care alone. The researchers note that for most outcomes, the GRADE quality of evidence was moderate.
“Our findings on colchicine should be interpreted cautiously due to the inclusion of open-labeled, randomized clinical trials,” Dr. Mehta and colleagues write. “The analysis of efficacy and safety outcomes are based on a small number of RCTs in control interventions.”
The authors reported no relevant financial relationships. Dr. Pillinger is co–principal investigator of the U.S. component of the COLCORONA trial; he reported no other relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
A new meta-analysis appears to show that colchicine has no benefit as a treatment for COVID-19, but its inclusion of trials studying differing patient populations and testing different outcomes led to “misleading” results, says a researcher involved in one of the trials.
The meta-analysis, which includes data from the recent Randomised Evaluation of COVID-19 Therapy (RECOVERY) trial, was published Nov. 22 in RMD Open.
Kedar Gautambhai Mehta, MBBS, MD, of the GMERS Medical College Gotri in Vadodara, India, and colleagues included outcomes from six studies of 16,148 patients with COVID-19 who received colchicine or supportive care. They evaluated the efficacy outcomes of mortality, need for ventilation, intensive care unit admission, and length of stay in hospital, as well as safety outcomes of adverse events, serious adverse events, and diarrhea.
The studies in the meta-analysis included a randomized, controlled trial (RCT) of 105 patients hospitalized with COVID-19 in Greece, the international, open-label RECOVERY RCT of 11,340 patients hospitalized with COVID-19, an RCT of 72 hospitalized patients with moderate or severe COVID-19 in Brazil, an RCT of 100 patients hospitalized with COVID-19 in Iran, the international COLCORONA trial of 4,488 patients with COVID-19 who were treated with colchicine or placebo on an outpatient basis, and the randomized COLORIT trial of 43 patients hospitalized with COVID-19 in Russia.
Studies “asked very different questions” about colchicine
Commenting on the meta-analysis, Michael H. Pillinger, MD, a rheumatologist and professor of medicine, biochemistry, and molecular pharmacology with New York University, said the authors combined studies “that are not comparable and that asked very different questions.” Two of the studies in the meta-analysis are very large, and four are very small, which skews the results, he explained.
“The larger studies therefore drive the outcome, and while the small studies are potentially insight providing, the large studies are the only ones worth giving our attention to in the context of the meta-analysis,” he said. The two largest studies – RECOVERY and COLCORONA – taken together show no benefit for colchicine as a treatment, even though the former demonstrated no benefit and the latter did show a benefit, explained Dr. Pillinger, a co–principal investigator for the COLCORONA trial in the United States.
The studies were designed differently and should not have been included in the same analysis, Dr. Pillinger argued. In the case of COLCORONA, early treatment with colchicine was the intervention, whereas RECOVERY focused on hospitalized patients.
“In designing [COLCORONA], the author group (of whom I was a member) expressly rejected the idea that colchicine might be useful for the sicker hospitalized patients, based on the long experience with colchicine of some of us as rheumatologists,” Dr. Pillinger said.
“In short, COLCORONA proved a benefit of colchicine in outpatient COVID-19, and its authors presumed there would be no inpatient benefit; RECOVERY went ahead and proved a lack of inpatient benefit, at least when high-dose steroids were also given,” he said. “While there is no conflict between these results, the combination of the two studies in this meta-analysis suggests there might be no benefit for colchicine overall, which is misleading and can lead physicians to reject the potential of outpatient colchicine, even for future studies.”
Dr. Pillinger said he still believes colchicine has potential value as a COVID-19 treatment option for patients with mild disease, “especially for low–vaccine rate, resource-starved countries.
“It would be unfortunate if meta-analyses such as this one would put a stop to colchicine’s use, or at least its further investigation,” he said.
Study details
The authors of the study assessed heterogeneity of the trials’ data across the outcomes using an I2 test. They evaluated the quality of the evidence for the outcomes using the Grades of Recommendation, Assessment, Development and Evaluation (GRADE).
The results of their meta-analysis showed that colchicine offered no significant improvement in mortality in six studies (risk difference, –0.0; 95% confidence interval, –0.01 to 0.01; I2 = 15%). It showed no benefit with respect to requiring ventilatory support in five studies of 15,519 patients (risk ratio, 0.67; 95% CI, 0.38-1.21; I2 = 47%); being admitted to the ICU in three studies with 220 patients (RR, 0.49; 95% CI, 0.19-1.25; I2 = 34%); and length of stay while in the hospital in four studies of 11,560 patients (mean difference, –1.17; 95% CI, –3.02 to 0.67; I2 = 77%).
There was no difference in serious adverse events in three studies with 4,665 patients (RD, –0.01; 95% CI, –0.02 to 0.00; I2 = 28%) for patients who received colchicine, compared with supportive care alone. Patients who received colchicine were more likely to have a higher rate of adverse events (RR, 1.58; 95% CI, 1.07-2.33; I2 = 81%) and to experience diarrhea (RR, 1.93; 95% CI, 1.62-2.29; I2 = 0%) than were patients who received supportive care alone. The researchers note that for most outcomes, the GRADE quality of evidence was moderate.
“Our findings on colchicine should be interpreted cautiously due to the inclusion of open-labeled, randomized clinical trials,” Dr. Mehta and colleagues write. “The analysis of efficacy and safety outcomes are based on a small number of RCTs in control interventions.”
The authors reported no relevant financial relationships. Dr. Pillinger is co–principal investigator of the U.S. component of the COLCORONA trial; he reported no other relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Oakland score identifies patients with lower GI bleed at low risk for adverse events
Background: The Oakland score was initially designed to be used in patients presenting with LGIB in the urgent, emergent, or primary care setting to help predict risk of readmission and determine if outpatient management is feasible. National guidelines in the United Kingdom have recommended use of the Oakland score despite limited external validation for the triage of patients with acute LGIB. This study aimed to externally validate the Oakland score in a large population in the United States and compare the performance at two thresholds.
Study design: Retrospective observational study.
Setting: 140 hospitals across the United States.
Synopsis: In this prognostic study, 38,067 patients were identified retrospectively using ICD-10 codes that were consistent with a diagnosis of LGIB and were admitted to the hospital. The Oakland score consisted of seven variables, including age, sex, prior hospitalization with LGIB, digital rectal exam results, heart rate, systolic blood pressure, and hemoglobin concentration. The primary outcome was safe discharge from the hospital, defined as absence of in-hospital rebleeding, RBC transfusion, therapeutic colonoscopy, mesenteric embolization or laparotomy for bleeding, in-hospital death, or readmission with subsequent LGIB in 28 days. In total, 47.9% of the identified patients experienced no adverse outcomes and were classified as meeting criteria for safe discharge. In addition, 8.7% of patients scored 8 points or fewer with a sensitivity of 98.4% and specificity of 16.0% for safe discharge. A sensitivity of 96% was maintained after increasing the threshold to 10 points or fewer with a specificity of 31.9%, suggesting the threshold can be increased while still maintaining adequate sensitivity. The study suggests that, by using the Oakland score threshold of 8, hospital admission may be avoided in low-risk patients leading to a savings of at least $44.5 million and even more if the threshold is increased to 10. Low specificity does present limitation of the score as some patients considered to be at risk for adverse events may have been safely discharged and managed as an outpatient, avoiding hospitalization.
Bottom line: The Oakland score was externally validated for use in assessing risk of adverse outcomes in patients with LGIB and had a high sensitivity but low specificity for identifying low-risk patients.
Citation: Oakland K et al. External validation of the Oakland score to assess safe hospital discharge among adult patients with acute lower gastrointestinal bleeding in the US. JAMA Netw Open. 2020 Jul 1;3:e209630. doi:
Dr. Steker is a hospitalist at Northwestern Memorial Hospital and instructor of medicine, Feinberg School of Medicine, both in Chicago.
Background: The Oakland score was initially designed to be used in patients presenting with LGIB in the urgent, emergent, or primary care setting to help predict risk of readmission and determine if outpatient management is feasible. National guidelines in the United Kingdom have recommended use of the Oakland score despite limited external validation for the triage of patients with acute LGIB. This study aimed to externally validate the Oakland score in a large population in the United States and compare the performance at two thresholds.
Study design: Retrospective observational study.
Setting: 140 hospitals across the United States.
Synopsis: In this prognostic study, 38,067 patients were identified retrospectively using ICD-10 codes that were consistent with a diagnosis of LGIB and were admitted to the hospital. The Oakland score consisted of seven variables, including age, sex, prior hospitalization with LGIB, digital rectal exam results, heart rate, systolic blood pressure, and hemoglobin concentration. The primary outcome was safe discharge from the hospital, defined as absence of in-hospital rebleeding, RBC transfusion, therapeutic colonoscopy, mesenteric embolization or laparotomy for bleeding, in-hospital death, or readmission with subsequent LGIB in 28 days. In total, 47.9% of the identified patients experienced no adverse outcomes and were classified as meeting criteria for safe discharge. In addition, 8.7% of patients scored 8 points or fewer with a sensitivity of 98.4% and specificity of 16.0% for safe discharge. A sensitivity of 96% was maintained after increasing the threshold to 10 points or fewer with a specificity of 31.9%, suggesting the threshold can be increased while still maintaining adequate sensitivity. The study suggests that, by using the Oakland score threshold of 8, hospital admission may be avoided in low-risk patients leading to a savings of at least $44.5 million and even more if the threshold is increased to 10. Low specificity does present limitation of the score as some patients considered to be at risk for adverse events may have been safely discharged and managed as an outpatient, avoiding hospitalization.
Bottom line: The Oakland score was externally validated for use in assessing risk of adverse outcomes in patients with LGIB and had a high sensitivity but low specificity for identifying low-risk patients.
Citation: Oakland K et al. External validation of the Oakland score to assess safe hospital discharge among adult patients with acute lower gastrointestinal bleeding in the US. JAMA Netw Open. 2020 Jul 1;3:e209630. doi:
Dr. Steker is a hospitalist at Northwestern Memorial Hospital and instructor of medicine, Feinberg School of Medicine, both in Chicago.
Background: The Oakland score was initially designed to be used in patients presenting with LGIB in the urgent, emergent, or primary care setting to help predict risk of readmission and determine if outpatient management is feasible. National guidelines in the United Kingdom have recommended use of the Oakland score despite limited external validation for the triage of patients with acute LGIB. This study aimed to externally validate the Oakland score in a large population in the United States and compare the performance at two thresholds.
Study design: Retrospective observational study.
Setting: 140 hospitals across the United States.
Synopsis: In this prognostic study, 38,067 patients were identified retrospectively using ICD-10 codes that were consistent with a diagnosis of LGIB and were admitted to the hospital. The Oakland score consisted of seven variables, including age, sex, prior hospitalization with LGIB, digital rectal exam results, heart rate, systolic blood pressure, and hemoglobin concentration. The primary outcome was safe discharge from the hospital, defined as absence of in-hospital rebleeding, RBC transfusion, therapeutic colonoscopy, mesenteric embolization or laparotomy for bleeding, in-hospital death, or readmission with subsequent LGIB in 28 days. In total, 47.9% of the identified patients experienced no adverse outcomes and were classified as meeting criteria for safe discharge. In addition, 8.7% of patients scored 8 points or fewer with a sensitivity of 98.4% and specificity of 16.0% for safe discharge. A sensitivity of 96% was maintained after increasing the threshold to 10 points or fewer with a specificity of 31.9%, suggesting the threshold can be increased while still maintaining adequate sensitivity. The study suggests that, by using the Oakland score threshold of 8, hospital admission may be avoided in low-risk patients leading to a savings of at least $44.5 million and even more if the threshold is increased to 10. Low specificity does present limitation of the score as some patients considered to be at risk for adverse events may have been safely discharged and managed as an outpatient, avoiding hospitalization.
Bottom line: The Oakland score was externally validated for use in assessing risk of adverse outcomes in patients with LGIB and had a high sensitivity but low specificity for identifying low-risk patients.
Citation: Oakland K et al. External validation of the Oakland score to assess safe hospital discharge among adult patients with acute lower gastrointestinal bleeding in the US. JAMA Netw Open. 2020 Jul 1;3:e209630. doi:
Dr. Steker is a hospitalist at Northwestern Memorial Hospital and instructor of medicine, Feinberg School of Medicine, both in Chicago.
Predicting cardiac shock mortality in the ICU
Addition of echocardiogram measurement of biventricular dysfunction improved the accuracy of prognosis among patients with cardiac shock (CS) in the cardiac intensive care unit.
In patients in the cardiac ICU with CS, biventricular dysfunction (BVD), as assessed using transthoracic echocardiography, improves clinical risk stratification when combined with the Society for Cardiovascular Angiography and Interventions shock stage.
No improvements in risk stratification was seen with patients with left or right ventricular systolic dysfunction (LVSD or RVSD) alone, according to an article published in the journal Chest.
Ventricular systolic dysfunction is commonly seen in patients who have suffered cardiac shock, most often on the left side. Although echocardiography is often performed on these patients during diagnosis, previous studies looking at ventricular dysfunction used invasive hemodynamic parameters, which made it challenging to incorporate their findings into general cardiac ICU practice.
Pinning down cardiac shock
Although treatment of acute MI and heart failure has improved greatly, particularly with the implementation of percutaneous coronary intervention (primary PCI) for ST-segment elevation MI. This has reduced the rate of future heart failure, but cardiac shock can occur before or after the procedure, with a 30-day mortality of 30%-40%. This outcome hasn’t improved in the last 20 years.
Efforts to improve cardiac shock outcomes through percutaneous mechanical circulatory support devices have been hindered by the fact that CS patients are heterogeneous, and prognosis may depend on a range of factors.
SCAI was developed as a five-stage classification system for CS to improve communication of patient status, as well as to improve differentiation among patients participation in clinical trials. It does not include measures of ventricular dysfunction.
Simple measure boosts prognosis accuracy
The new work adds an additional layer to the SCAI shock stage. “Adding echocardiography allows discrimination between levels of risk for each SCAI stage,” said David Baran, MD, who was asked for comment. Dr. Baran was the lead author on the original SCAI study and is system director of advanced heart failure at Sentara Heart Hospital, as well as a professor of medicine at Eastern Virginia Medical School, both in Norfolk.
The work also underscores the value of repeated measures of prognosis during a patient’s stay in the ICU. “If a patient is not improving, it may prompt a consideration of whether transfer or consultation with a tertiary center may be of value. Conversely, if a patient doesn’t have high-risk features and is responding to therapy, it is reassuring to have data supporting low mortality with that care plan,” said Dr. Baran.
The study may be biased, since not every patient undergoes an echocardiogram. Still, “the authors make a convincing case that biventricular dysfunction is a powerful negative marker across the spectrum of SCAI stages,” said Dr. Baran.
Echocardiography is simple and generally available, and some are even portable and used with a smartphone. But patient body size interferes with echocardiography, as can the presence of a ventilator or multiple surgical dressings. “The key advantage of echo is that it is completely noninvasive and can be brought to the patient in the ICU, unlike other testing which involves moving the patient to the testing environment,” said Dr. Baran.
The researchers analyzed data from 3,158 patients admitted to the cardiac ICU at the Mayo Clinic Hospital St. Mary’s Campus in Rochester, Minn., 51.8% of whom had acute coronary syndromes. They defined LVSD as a left ventricular ejection fraction less than 40%, and RVSD as at least moderate systolic dysfunction determined by semiquantitative measurement. BVD constituted the presence of both LVSD and RVSD. They examined the association of in-hospital mortality with these parameters combined with SCAI stage.
BVD a risk factor
Overall in-hospital mortality was 10%. A total of 22.3% of patients had LVSD and 11.8% had RVSD; 16.4% had moderate or greater BVD. There was no association between LVSD or RVSD and in-hospital mortality after adjustment for SCAI stage, but there was a significant association for BVD (adjusted hazard ratio, 1.815; P = .0023). When combined with SCAI, BVC led to an improved ability to predict hospital mortality (area under the curve, 0.784 vs. 0.766; P < .001). Adding semiquantitative RVSD and LVSD led to more improvement (AUC, 0.794; P < .01 vs. both).
RVSD was associated with higher in-hospital mortality (adjusted odds ratio, 1.421; P = .02), and there was a trend toward greater mortality with LVSD (aOR, 1.336; P = .06). There was little change when SCAI shock stage A patients were excluded (aOR, 1.840; P < .001).
Patients with BVD had greater in-hospital mortality than those without ventricular dysfunction (aOR, 1.815; P = .0023), but other between-group comparisons were not significant.
The researchers performed a classification and regression tree analysis using left ventricular ejection fraction (LVEF) and semiquantitative RVSD. It found that RVSD was a better predictor of in-hospital mortality than LVSD, and the best cutoff for LVSD was different among patients with RVSD and patients without RVSD.
Patients with mild or greater RVD and LVEF greater than 24% were considered high risk; those with borderline or low RVSD and LVEF less than 33%, or mild or greater RVSD with LVEF of at least 24%, were considered intermediate risk. Patients with borderline or no RVSD and LVEF of at least 33% were considered low risk. Hospital mortality was 22% in the high-risk group, 12.2% in the intermediate group, and 3.3% in the low-risk group (aOR vs. intermediate, 0.493; P = .0006; aOR vs. high risk, 0.357; P < .0001).
The study authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Addition of echocardiogram measurement of biventricular dysfunction improved the accuracy of prognosis among patients with cardiac shock (CS) in the cardiac intensive care unit.
In patients in the cardiac ICU with CS, biventricular dysfunction (BVD), as assessed using transthoracic echocardiography, improves clinical risk stratification when combined with the Society for Cardiovascular Angiography and Interventions shock stage.
No improvements in risk stratification was seen with patients with left or right ventricular systolic dysfunction (LVSD or RVSD) alone, according to an article published in the journal Chest.
Ventricular systolic dysfunction is commonly seen in patients who have suffered cardiac shock, most often on the left side. Although echocardiography is often performed on these patients during diagnosis, previous studies looking at ventricular dysfunction used invasive hemodynamic parameters, which made it challenging to incorporate their findings into general cardiac ICU practice.
Pinning down cardiac shock
Although treatment of acute MI and heart failure has improved greatly, particularly with the implementation of percutaneous coronary intervention (primary PCI) for ST-segment elevation MI. This has reduced the rate of future heart failure, but cardiac shock can occur before or after the procedure, with a 30-day mortality of 30%-40%. This outcome hasn’t improved in the last 20 years.
Efforts to improve cardiac shock outcomes through percutaneous mechanical circulatory support devices have been hindered by the fact that CS patients are heterogeneous, and prognosis may depend on a range of factors.
SCAI was developed as a five-stage classification system for CS to improve communication of patient status, as well as to improve differentiation among patients participation in clinical trials. It does not include measures of ventricular dysfunction.
Simple measure boosts prognosis accuracy
The new work adds an additional layer to the SCAI shock stage. “Adding echocardiography allows discrimination between levels of risk for each SCAI stage,” said David Baran, MD, who was asked for comment. Dr. Baran was the lead author on the original SCAI study and is system director of advanced heart failure at Sentara Heart Hospital, as well as a professor of medicine at Eastern Virginia Medical School, both in Norfolk.
The work also underscores the value of repeated measures of prognosis during a patient’s stay in the ICU. “If a patient is not improving, it may prompt a consideration of whether transfer or consultation with a tertiary center may be of value. Conversely, if a patient doesn’t have high-risk features and is responding to therapy, it is reassuring to have data supporting low mortality with that care plan,” said Dr. Baran.
The study may be biased, since not every patient undergoes an echocardiogram. Still, “the authors make a convincing case that biventricular dysfunction is a powerful negative marker across the spectrum of SCAI stages,” said Dr. Baran.
Echocardiography is simple and generally available, and some are even portable and used with a smartphone. But patient body size interferes with echocardiography, as can the presence of a ventilator or multiple surgical dressings. “The key advantage of echo is that it is completely noninvasive and can be brought to the patient in the ICU, unlike other testing which involves moving the patient to the testing environment,” said Dr. Baran.
The researchers analyzed data from 3,158 patients admitted to the cardiac ICU at the Mayo Clinic Hospital St. Mary’s Campus in Rochester, Minn., 51.8% of whom had acute coronary syndromes. They defined LVSD as a left ventricular ejection fraction less than 40%, and RVSD as at least moderate systolic dysfunction determined by semiquantitative measurement. BVD constituted the presence of both LVSD and RVSD. They examined the association of in-hospital mortality with these parameters combined with SCAI stage.
BVD a risk factor
Overall in-hospital mortality was 10%. A total of 22.3% of patients had LVSD and 11.8% had RVSD; 16.4% had moderate or greater BVD. There was no association between LVSD or RVSD and in-hospital mortality after adjustment for SCAI stage, but there was a significant association for BVD (adjusted hazard ratio, 1.815; P = .0023). When combined with SCAI, BVC led to an improved ability to predict hospital mortality (area under the curve, 0.784 vs. 0.766; P < .001). Adding semiquantitative RVSD and LVSD led to more improvement (AUC, 0.794; P < .01 vs. both).
RVSD was associated with higher in-hospital mortality (adjusted odds ratio, 1.421; P = .02), and there was a trend toward greater mortality with LVSD (aOR, 1.336; P = .06). There was little change when SCAI shock stage A patients were excluded (aOR, 1.840; P < .001).
Patients with BVD had greater in-hospital mortality than those without ventricular dysfunction (aOR, 1.815; P = .0023), but other between-group comparisons were not significant.
The researchers performed a classification and regression tree analysis using left ventricular ejection fraction (LVEF) and semiquantitative RVSD. It found that RVSD was a better predictor of in-hospital mortality than LVSD, and the best cutoff for LVSD was different among patients with RVSD and patients without RVSD.
Patients with mild or greater RVD and LVEF greater than 24% were considered high risk; those with borderline or low RVSD and LVEF less than 33%, or mild or greater RVSD with LVEF of at least 24%, were considered intermediate risk. Patients with borderline or no RVSD and LVEF of at least 33% were considered low risk. Hospital mortality was 22% in the high-risk group, 12.2% in the intermediate group, and 3.3% in the low-risk group (aOR vs. intermediate, 0.493; P = .0006; aOR vs. high risk, 0.357; P < .0001).
The study authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Addition of echocardiogram measurement of biventricular dysfunction improved the accuracy of prognosis among patients with cardiac shock (CS) in the cardiac intensive care unit.
In patients in the cardiac ICU with CS, biventricular dysfunction (BVD), as assessed using transthoracic echocardiography, improves clinical risk stratification when combined with the Society for Cardiovascular Angiography and Interventions shock stage.
No improvements in risk stratification was seen with patients with left or right ventricular systolic dysfunction (LVSD or RVSD) alone, according to an article published in the journal Chest.
Ventricular systolic dysfunction is commonly seen in patients who have suffered cardiac shock, most often on the left side. Although echocardiography is often performed on these patients during diagnosis, previous studies looking at ventricular dysfunction used invasive hemodynamic parameters, which made it challenging to incorporate their findings into general cardiac ICU practice.
Pinning down cardiac shock
Although treatment of acute MI and heart failure has improved greatly, particularly with the implementation of percutaneous coronary intervention (primary PCI) for ST-segment elevation MI. This has reduced the rate of future heart failure, but cardiac shock can occur before or after the procedure, with a 30-day mortality of 30%-40%. This outcome hasn’t improved in the last 20 years.
Efforts to improve cardiac shock outcomes through percutaneous mechanical circulatory support devices have been hindered by the fact that CS patients are heterogeneous, and prognosis may depend on a range of factors.
SCAI was developed as a five-stage classification system for CS to improve communication of patient status, as well as to improve differentiation among patients participation in clinical trials. It does not include measures of ventricular dysfunction.
Simple measure boosts prognosis accuracy
The new work adds an additional layer to the SCAI shock stage. “Adding echocardiography allows discrimination between levels of risk for each SCAI stage,” said David Baran, MD, who was asked for comment. Dr. Baran was the lead author on the original SCAI study and is system director of advanced heart failure at Sentara Heart Hospital, as well as a professor of medicine at Eastern Virginia Medical School, both in Norfolk.
The work also underscores the value of repeated measures of prognosis during a patient’s stay in the ICU. “If a patient is not improving, it may prompt a consideration of whether transfer or consultation with a tertiary center may be of value. Conversely, if a patient doesn’t have high-risk features and is responding to therapy, it is reassuring to have data supporting low mortality with that care plan,” said Dr. Baran.
The study may be biased, since not every patient undergoes an echocardiogram. Still, “the authors make a convincing case that biventricular dysfunction is a powerful negative marker across the spectrum of SCAI stages,” said Dr. Baran.
Echocardiography is simple and generally available, and some are even portable and used with a smartphone. But patient body size interferes with echocardiography, as can the presence of a ventilator or multiple surgical dressings. “The key advantage of echo is that it is completely noninvasive and can be brought to the patient in the ICU, unlike other testing which involves moving the patient to the testing environment,” said Dr. Baran.
The researchers analyzed data from 3,158 patients admitted to the cardiac ICU at the Mayo Clinic Hospital St. Mary’s Campus in Rochester, Minn., 51.8% of whom had acute coronary syndromes. They defined LVSD as a left ventricular ejection fraction less than 40%, and RVSD as at least moderate systolic dysfunction determined by semiquantitative measurement. BVD constituted the presence of both LVSD and RVSD. They examined the association of in-hospital mortality with these parameters combined with SCAI stage.
BVD a risk factor
Overall in-hospital mortality was 10%. A total of 22.3% of patients had LVSD and 11.8% had RVSD; 16.4% had moderate or greater BVD. There was no association between LVSD or RVSD and in-hospital mortality after adjustment for SCAI stage, but there was a significant association for BVD (adjusted hazard ratio, 1.815; P = .0023). When combined with SCAI, BVC led to an improved ability to predict hospital mortality (area under the curve, 0.784 vs. 0.766; P < .001). Adding semiquantitative RVSD and LVSD led to more improvement (AUC, 0.794; P < .01 vs. both).
RVSD was associated with higher in-hospital mortality (adjusted odds ratio, 1.421; P = .02), and there was a trend toward greater mortality with LVSD (aOR, 1.336; P = .06). There was little change when SCAI shock stage A patients were excluded (aOR, 1.840; P < .001).
Patients with BVD had greater in-hospital mortality than those without ventricular dysfunction (aOR, 1.815; P = .0023), but other between-group comparisons were not significant.
The researchers performed a classification and regression tree analysis using left ventricular ejection fraction (LVEF) and semiquantitative RVSD. It found that RVSD was a better predictor of in-hospital mortality than LVSD, and the best cutoff for LVSD was different among patients with RVSD and patients without RVSD.
Patients with mild or greater RVD and LVEF greater than 24% were considered high risk; those with borderline or low RVSD and LVEF less than 33%, or mild or greater RVSD with LVEF of at least 24%, were considered intermediate risk. Patients with borderline or no RVSD and LVEF of at least 33% were considered low risk. Hospital mortality was 22% in the high-risk group, 12.2% in the intermediate group, and 3.3% in the low-risk group (aOR vs. intermediate, 0.493; P = .0006; aOR vs. high risk, 0.357; P < .0001).
The study authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
What makes a urinary tract infection complicated?
Consider anatomical and severity risk factors
Case
A 72-year-old woman with type 2 diabetes mellitus presents with acute dysuria, fever, and flank pain. She had a urinary tract infection (UTI) 3 months prior treated with nitrofurantoin. Temperature is 102° F, heart rate 112 beats per minute, and the remainder of vital signs are normal. She has left costovertebral angle tenderness. Urine microscopy shows 70 WBCs per high power field and bacteria. Is this urinary tract infection complicated?
Background
The urinary tract is divided into the upper tract, which includes the kidneys and ureters, and the lower urinary tract, which includes the bladder, urethra, and prostate. Infection of the lower urinary tract is referred to as cystitis while infection of the upper urinary tract is pyelonephritis. A UTI is the colonization of pathogen(s) within the urinary system that causes an inflammatory response resulting in symptoms and requiring treatment. UTIs occur when there is reduced urine flow, an increase in colonization risk, and when there are factors that facilitate ascent such as catheterization or incontinence.
There are an estimated 150 million cases of UTIs worldwide per year, accounting for $6 billion in health care expenditures.1 In the inpatient setting, about 40% of nosocomial infections are associated with urinary catheters. This equates to about 1 million catheter-associated UTIs per year in the United States, and up to 40% of hospital gram-negative bacteremia per year are caused by UTIs.1
UTIs are often classified as either uncomplicated or complicated infections, which can influence the depth of management. UTIs have a wide spectrum of symptoms and can manifest anywhere from mild dysuria treated successfully with outpatient antibiotics to florid sepsis. Uncomplicated simple cystitis is often treated as an outpatient with oral nitrofurantoin or trimethoprim-sulfamethoxazole.2 Complicated UTIs are treated with broader antimicrobial coverage, and depending on severity, could require intravenous antibiotics. Many factors affect how a UTI manifests and determining whether an infection is “uncomplicated” or “complicated” is an important first step in guiding management. Unfortunately, there are differing classifications of “complicated” UTIs, making it a complicated issue itself. We outline two common approaches.
Anatomic approach
A commonly recognized definition is from the American Urological Association, which states that complicated UTIs are symptomatic cases associated with the presence of “underlying, predisposing conditions and not necessarily clinical severity, invasiveness, or complications.”3 These factors include structural or functional urinary tract abnormalities or urinary instrumentation (see Table 1). These predisposing conditions can increase microbial colonization and decrease therapy efficacy, thus increasing the frequency of infection and relapse.
This population of patients is at high risk of infections with more resistant bacteria such as extended-spectrum beta-lactamase (ESBL) producing Escherichia coli since they often lack the natural genitourinary barriers to infection. In addition, these patients more often undergo multiple antibiotic courses for their frequent infections, which also contributes to their risk of ESBL infections. Genitourinary abnormalities interfere with normal voiding, resulting in impaired flushing of bacteria. For instance, obstruction inhibits complete urinary drainage and increases the persistence of bacteria in biofilms, especially if there are stones or indwelling devices present. Biofilms usually contain a high concentration of organisms including Proteus mirabilis, Morgenella morganii, and Providencia spp.4 Keep in mind that, if there is an obstruction, the urinalysis might be without pyuria or bacteriuria.
Instrumentation increases infection risks through the direct introduction of bacteria into the genitourinary tract. Despite the efforts in maintaining sterility in urinary catheter placement, catheters provide a nidus for infection. Catheter-associated UTI (CAUTI) is defined by the Infectious Disease Society of America as UTIs that occur in patients with an indwelling catheter or who had a catheter removed for less than 48 hours who develop urinary symptoms and cultures positive for uropathogenic bacteria.4 Studies show that in general, patients with indwelling catheters will develop bacteriuria over time, with 10%-25% eventually developing symptoms.
Severity approach
There are other schools of thought that categorize uncomplicated versus complicated UTIs based on the severity of presentation (see Table 2). An uncomplicated UTI would be classified as symptoms and signs of simple cystitis limited to dysuria, frequency, urgency, and suprapubic pain. Using a symptom severity approach, systemic findings such as fever, chills, emesis, flank pain, costovertebral angle tenderness, or other findings of sepsis would be classified as a complicated UTI. These systemic findings would suggest an extension of infection beyond the bladder.
The argument for a symptomatic-based approach of classification is that the severity of symptoms should dictate the degree of management. Not all UTIs in the anatomic approach are severe. In fact, populations that are considered at risk for complicated UTIs by the AUA guidelines in Table 1 often have mild symptomatic cystitis or asymptomatic bacteriuria. Asymptomatic bacteriuria is the colonization of organisms in the urinary tract without active infection. For instance, bacteriuria is present in almost 100% of people with chronic indwelling catheters, 30%-40% of neurogenic bladder requiring intermittent catheterization, and 50% of elderly nursing home residents.4 Not all bacteriuria triggers enough of an inflammatory response to cause symptoms that require treatment.
Ultimate clinical judgment
Although there are multiple different society recommendations in distinguishing uncomplicated versus complicated UTIs, considering both anatomical and severity risk factors can better aid in clinical decision-making rather than abiding by one classification method alone.
Uncomplicated UTIs from the AUA guidelines can cause severe infections that might require longer courses of broad-spectrum antibiotics. On the other hand, people with anatomic abnormalities can present with mild symptoms that can be treated with a narrow-spectrum antibiotic for a standard time course. Recognizing the severity of the infection and using clinical judgment aids in antibiotic stewardship.
Although the existence of algorithmic approaches can help guide clinical judgment, accounting for the spectrum of host and bacterial factors should ultimately determine the complexity of the disease and management.3 Using clinical suspicion to determine when a UTI should be treated as a complicated infection can ensure effective treatment and decrease the likelihood of sepsis, renal scarring, or end-stage disease.5
Back to the case
The case presents an elderly woman with diabetes presenting with sepsis from a UTI. Because of a normal urinary tract and no prior instrumentation, by the AUA definition, she would be classified as an uncomplicated UTI; however, we would classify her as a complicated UTI based on the severity of her presentation. She has a fever, tachycardia, flank pain, and costovertebral angle tenderness that are evidence of infection extending beyond the bladder. She has sepsis warranting inpatient management. Prior urine culture results could aid in determining empiric treatment while waiting for new cultures. In her case, an intravenous antibiotic with broad gram-negative coverage such as ceftriaxone would be appropriate.
Bottom line
There are multiple interpretations of complicated UTIs including both an anatomical and severity approach. Clinical judgment regarding infection severity should determine the depth of management.
Dr. Vu is a hospitalist at the University of Kentucky, Lexington. Dr. Gray is a hospitalist at the University of Kentucky and the Lexington Veterans Affairs Medical Center.
References
1. Folk CS. AUA Core Curriculum: Urinary Tract Infection (Adult). 2021 Mar 1. https://university.auanet.org/core_topic.cfm?coreid=92.
2. Gupta K et al. International clinical practice guidelines for the treatment of acute uncomplicated cystitis and pyelonephritis in women: A 2010 update by the Infectious Diseases Society of America and the European Society for Microbiology and Infectious Diseases. Clin Infect Dis. 2011 Mar 1;52(5):e103-20. doi: 10.1093/cid/ciq257.
3. Johnson JR. Definition of Complicated Urinary Tract Infection. Clin Infect Dis. 2017 February 15;64(4):529. doi: 10.1093/cid/ciw751.
4. Nicolle LE, AMMI Canada Guidelines Committee. Complicated urinary tract infection in adults. Can J Infect Dis Med Microbiol. 2005;16(6):349-60. doi: 10.1155/2005/385768.
5. Melekos MD and Naber KG. Complicated urinary tract infections. Int J Antimicrob Agents. 2000;15(4):247-56. doi: 10.1016/s0924-8579(00)00168-0.
Key points
- The anatomical approach to defining complicated UTIs considers the presence of underlying, predisposing conditions such as structurally or functionally abnormal genitourinary tract or urinary instrumentation or foreign bodies.
- The severity approach to defining complicated UTIs considers the severity of presentation including the presence of systemic manifestations.
- Both approaches should consider populations that are at risk for recurrent or multidrug-resistant infections and infections that can lead to high morbidity.
- Either approach can be used as a guide, but neither should replace clinical suspicion and judgment in determining the depth of treatment.
Additional reading
Choe HS et al. Summary of the UAA‐AAUS guidelines for urinary tract infections. Int J Urol. 2018 Mar;25(3):175-85. doi:10.1111/iju.13493.
Nicolle LE et al. Infectious Diseases Society of America Guidelines for the Diagnosis and Treatment of Asymptomatic Bacteriuria in Adults. Clin Infect Dis. 2005 Mar;40(5):643-54. doi: 10.1086/427507.
Wagenlehner FME et al. Epidemiology, definition and treatment of complicated urinary tract infections. Nat Rev Urol. 2020 Oct;17:586-600. doi:10.1038/s41585-020-0362-4.
Wallace DW et al. Urinalysis: A simple test with complicated interpretation. J Urgent Care Med. 2020 July-Aug;14(10):11-4.
Quiz
A 68-year-old woman with type 2 diabetes mellitus presents to the emergency department with acute fever, chills, dysuria, frequency, and suprapubic pain. She has associated nausea, malaise, and fatigue. She takes metformin and denies recent antibiotic use. Her temperature is 102.8° F, heart rate 118 beats per minute, blood pressure 118/71 mm Hg, and her respiratory rate is 24 breaths per minute. She is ill-appearing and has mild suprapubic tenderness. White blood cell count is 18 k/mcL. Urinalysis is positive for leukocyte esterase, nitrites, and bacteria. Urine microscopy has 120 white blood cells per high power field. What is the most appropriate treatment?
A. Azithromycin
B. Ceftriaxone
C. Cefepime and vancomycin
D. Nitrofurantoin
The answer is B. The patient presents with sepsis secondary to a urinary tract infection. Using the anatomic approach this would be classified as uncomplicated. Using the severity approach, this would be classified as a complicated urinary tract infection. With fever, chills, and signs of sepsis, it’s likely her infection extends beyond the bladder. Given the severity of her presentation, we’d favor treating her as a complicated urinary tract infection with intravenous ceftriaxone. There is no suggestion of resistance or additional MRSA risk factors requiring intravenous vancomycin or cefepime. Nitrofurantoin, although a first-line treatment for uncomplicated cystitis, would not be appropriate if there is suspicion infection extends beyond the bladder. Azithromycin is a first-line option for chlamydia trachomatis, but not a urinary tract infection.
Consider anatomical and severity risk factors
Consider anatomical and severity risk factors
Case
A 72-year-old woman with type 2 diabetes mellitus presents with acute dysuria, fever, and flank pain. She had a urinary tract infection (UTI) 3 months prior treated with nitrofurantoin. Temperature is 102° F, heart rate 112 beats per minute, and the remainder of vital signs are normal. She has left costovertebral angle tenderness. Urine microscopy shows 70 WBCs per high power field and bacteria. Is this urinary tract infection complicated?
Background
The urinary tract is divided into the upper tract, which includes the kidneys and ureters, and the lower urinary tract, which includes the bladder, urethra, and prostate. Infection of the lower urinary tract is referred to as cystitis while infection of the upper urinary tract is pyelonephritis. A UTI is the colonization of pathogen(s) within the urinary system that causes an inflammatory response resulting in symptoms and requiring treatment. UTIs occur when there is reduced urine flow, an increase in colonization risk, and when there are factors that facilitate ascent such as catheterization or incontinence.
There are an estimated 150 million cases of UTIs worldwide per year, accounting for $6 billion in health care expenditures.1 In the inpatient setting, about 40% of nosocomial infections are associated with urinary catheters. This equates to about 1 million catheter-associated UTIs per year in the United States, and up to 40% of hospital gram-negative bacteremia per year are caused by UTIs.1
UTIs are often classified as either uncomplicated or complicated infections, which can influence the depth of management. UTIs have a wide spectrum of symptoms and can manifest anywhere from mild dysuria treated successfully with outpatient antibiotics to florid sepsis. Uncomplicated simple cystitis is often treated as an outpatient with oral nitrofurantoin or trimethoprim-sulfamethoxazole.2 Complicated UTIs are treated with broader antimicrobial coverage, and depending on severity, could require intravenous antibiotics. Many factors affect how a UTI manifests and determining whether an infection is “uncomplicated” or “complicated” is an important first step in guiding management. Unfortunately, there are differing classifications of “complicated” UTIs, making it a complicated issue itself. We outline two common approaches.
Anatomic approach
A commonly recognized definition is from the American Urological Association, which states that complicated UTIs are symptomatic cases associated with the presence of “underlying, predisposing conditions and not necessarily clinical severity, invasiveness, or complications.”3 These factors include structural or functional urinary tract abnormalities or urinary instrumentation (see Table 1). These predisposing conditions can increase microbial colonization and decrease therapy efficacy, thus increasing the frequency of infection and relapse.
This population of patients is at high risk of infections with more resistant bacteria such as extended-spectrum beta-lactamase (ESBL) producing Escherichia coli since they often lack the natural genitourinary barriers to infection. In addition, these patients more often undergo multiple antibiotic courses for their frequent infections, which also contributes to their risk of ESBL infections. Genitourinary abnormalities interfere with normal voiding, resulting in impaired flushing of bacteria. For instance, obstruction inhibits complete urinary drainage and increases the persistence of bacteria in biofilms, especially if there are stones or indwelling devices present. Biofilms usually contain a high concentration of organisms including Proteus mirabilis, Morgenella morganii, and Providencia spp.4 Keep in mind that, if there is an obstruction, the urinalysis might be without pyuria or bacteriuria.
Instrumentation increases infection risks through the direct introduction of bacteria into the genitourinary tract. Despite the efforts in maintaining sterility in urinary catheter placement, catheters provide a nidus for infection. Catheter-associated UTI (CAUTI) is defined by the Infectious Disease Society of America as UTIs that occur in patients with an indwelling catheter or who had a catheter removed for less than 48 hours who develop urinary symptoms and cultures positive for uropathogenic bacteria.4 Studies show that in general, patients with indwelling catheters will develop bacteriuria over time, with 10%-25% eventually developing symptoms.
Severity approach
There are other schools of thought that categorize uncomplicated versus complicated UTIs based on the severity of presentation (see Table 2). An uncomplicated UTI would be classified as symptoms and signs of simple cystitis limited to dysuria, frequency, urgency, and suprapubic pain. Using a symptom severity approach, systemic findings such as fever, chills, emesis, flank pain, costovertebral angle tenderness, or other findings of sepsis would be classified as a complicated UTI. These systemic findings would suggest an extension of infection beyond the bladder.
The argument for a symptomatic-based approach of classification is that the severity of symptoms should dictate the degree of management. Not all UTIs in the anatomic approach are severe. In fact, populations that are considered at risk for complicated UTIs by the AUA guidelines in Table 1 often have mild symptomatic cystitis or asymptomatic bacteriuria. Asymptomatic bacteriuria is the colonization of organisms in the urinary tract without active infection. For instance, bacteriuria is present in almost 100% of people with chronic indwelling catheters, 30%-40% of neurogenic bladder requiring intermittent catheterization, and 50% of elderly nursing home residents.4 Not all bacteriuria triggers enough of an inflammatory response to cause symptoms that require treatment.
Ultimate clinical judgment
Although there are multiple different society recommendations in distinguishing uncomplicated versus complicated UTIs, considering both anatomical and severity risk factors can better aid in clinical decision-making rather than abiding by one classification method alone.
Uncomplicated UTIs from the AUA guidelines can cause severe infections that might require longer courses of broad-spectrum antibiotics. On the other hand, people with anatomic abnormalities can present with mild symptoms that can be treated with a narrow-spectrum antibiotic for a standard time course. Recognizing the severity of the infection and using clinical judgment aids in antibiotic stewardship.
Although the existence of algorithmic approaches can help guide clinical judgment, accounting for the spectrum of host and bacterial factors should ultimately determine the complexity of the disease and management.3 Using clinical suspicion to determine when a UTI should be treated as a complicated infection can ensure effective treatment and decrease the likelihood of sepsis, renal scarring, or end-stage disease.5
Back to the case
The case presents an elderly woman with diabetes presenting with sepsis from a UTI. Because of a normal urinary tract and no prior instrumentation, by the AUA definition, she would be classified as an uncomplicated UTI; however, we would classify her as a complicated UTI based on the severity of her presentation. She has a fever, tachycardia, flank pain, and costovertebral angle tenderness that are evidence of infection extending beyond the bladder. She has sepsis warranting inpatient management. Prior urine culture results could aid in determining empiric treatment while waiting for new cultures. In her case, an intravenous antibiotic with broad gram-negative coverage such as ceftriaxone would be appropriate.
Bottom line
There are multiple interpretations of complicated UTIs including both an anatomical and severity approach. Clinical judgment regarding infection severity should determine the depth of management.
Dr. Vu is a hospitalist at the University of Kentucky, Lexington. Dr. Gray is a hospitalist at the University of Kentucky and the Lexington Veterans Affairs Medical Center.
References
1. Folk CS. AUA Core Curriculum: Urinary Tract Infection (Adult). 2021 Mar 1. https://university.auanet.org/core_topic.cfm?coreid=92.
2. Gupta K et al. International clinical practice guidelines for the treatment of acute uncomplicated cystitis and pyelonephritis in women: A 2010 update by the Infectious Diseases Society of America and the European Society for Microbiology and Infectious Diseases. Clin Infect Dis. 2011 Mar 1;52(5):e103-20. doi: 10.1093/cid/ciq257.
3. Johnson JR. Definition of Complicated Urinary Tract Infection. Clin Infect Dis. 2017 February 15;64(4):529. doi: 10.1093/cid/ciw751.
4. Nicolle LE, AMMI Canada Guidelines Committee. Complicated urinary tract infection in adults. Can J Infect Dis Med Microbiol. 2005;16(6):349-60. doi: 10.1155/2005/385768.
5. Melekos MD and Naber KG. Complicated urinary tract infections. Int J Antimicrob Agents. 2000;15(4):247-56. doi: 10.1016/s0924-8579(00)00168-0.
Key points
- The anatomical approach to defining complicated UTIs considers the presence of underlying, predisposing conditions such as structurally or functionally abnormal genitourinary tract or urinary instrumentation or foreign bodies.
- The severity approach to defining complicated UTIs considers the severity of presentation including the presence of systemic manifestations.
- Both approaches should consider populations that are at risk for recurrent or multidrug-resistant infections and infections that can lead to high morbidity.
- Either approach can be used as a guide, but neither should replace clinical suspicion and judgment in determining the depth of treatment.
Additional reading
Choe HS et al. Summary of the UAA‐AAUS guidelines for urinary tract infections. Int J Urol. 2018 Mar;25(3):175-85. doi:10.1111/iju.13493.
Nicolle LE et al. Infectious Diseases Society of America Guidelines for the Diagnosis and Treatment of Asymptomatic Bacteriuria in Adults. Clin Infect Dis. 2005 Mar;40(5):643-54. doi: 10.1086/427507.
Wagenlehner FME et al. Epidemiology, definition and treatment of complicated urinary tract infections. Nat Rev Urol. 2020 Oct;17:586-600. doi:10.1038/s41585-020-0362-4.
Wallace DW et al. Urinalysis: A simple test with complicated interpretation. J Urgent Care Med. 2020 July-Aug;14(10):11-4.
Quiz
A 68-year-old woman with type 2 diabetes mellitus presents to the emergency department with acute fever, chills, dysuria, frequency, and suprapubic pain. She has associated nausea, malaise, and fatigue. She takes metformin and denies recent antibiotic use. Her temperature is 102.8° F, heart rate 118 beats per minute, blood pressure 118/71 mm Hg, and her respiratory rate is 24 breaths per minute. She is ill-appearing and has mild suprapubic tenderness. White blood cell count is 18 k/mcL. Urinalysis is positive for leukocyte esterase, nitrites, and bacteria. Urine microscopy has 120 white blood cells per high power field. What is the most appropriate treatment?
A. Azithromycin
B. Ceftriaxone
C. Cefepime and vancomycin
D. Nitrofurantoin
The answer is B. The patient presents with sepsis secondary to a urinary tract infection. Using the anatomic approach this would be classified as uncomplicated. Using the severity approach, this would be classified as a complicated urinary tract infection. With fever, chills, and signs of sepsis, it’s likely her infection extends beyond the bladder. Given the severity of her presentation, we’d favor treating her as a complicated urinary tract infection with intravenous ceftriaxone. There is no suggestion of resistance or additional MRSA risk factors requiring intravenous vancomycin or cefepime. Nitrofurantoin, although a first-line treatment for uncomplicated cystitis, would not be appropriate if there is suspicion infection extends beyond the bladder. Azithromycin is a first-line option for chlamydia trachomatis, but not a urinary tract infection.
Case
A 72-year-old woman with type 2 diabetes mellitus presents with acute dysuria, fever, and flank pain. She had a urinary tract infection (UTI) 3 months prior treated with nitrofurantoin. Temperature is 102° F, heart rate 112 beats per minute, and the remainder of vital signs are normal. She has left costovertebral angle tenderness. Urine microscopy shows 70 WBCs per high power field and bacteria. Is this urinary tract infection complicated?
Background
The urinary tract is divided into the upper tract, which includes the kidneys and ureters, and the lower urinary tract, which includes the bladder, urethra, and prostate. Infection of the lower urinary tract is referred to as cystitis while infection of the upper urinary tract is pyelonephritis. A UTI is the colonization of pathogen(s) within the urinary system that causes an inflammatory response resulting in symptoms and requiring treatment. UTIs occur when there is reduced urine flow, an increase in colonization risk, and when there are factors that facilitate ascent such as catheterization or incontinence.
There are an estimated 150 million cases of UTIs worldwide per year, accounting for $6 billion in health care expenditures.1 In the inpatient setting, about 40% of nosocomial infections are associated with urinary catheters. This equates to about 1 million catheter-associated UTIs per year in the United States, and up to 40% of hospital gram-negative bacteremia per year are caused by UTIs.1
UTIs are often classified as either uncomplicated or complicated infections, which can influence the depth of management. UTIs have a wide spectrum of symptoms and can manifest anywhere from mild dysuria treated successfully with outpatient antibiotics to florid sepsis. Uncomplicated simple cystitis is often treated as an outpatient with oral nitrofurantoin or trimethoprim-sulfamethoxazole.2 Complicated UTIs are treated with broader antimicrobial coverage, and depending on severity, could require intravenous antibiotics. Many factors affect how a UTI manifests and determining whether an infection is “uncomplicated” or “complicated” is an important first step in guiding management. Unfortunately, there are differing classifications of “complicated” UTIs, making it a complicated issue itself. We outline two common approaches.
Anatomic approach
A commonly recognized definition is from the American Urological Association, which states that complicated UTIs are symptomatic cases associated with the presence of “underlying, predisposing conditions and not necessarily clinical severity, invasiveness, or complications.”3 These factors include structural or functional urinary tract abnormalities or urinary instrumentation (see Table 1). These predisposing conditions can increase microbial colonization and decrease therapy efficacy, thus increasing the frequency of infection and relapse.
This population of patients is at high risk of infections with more resistant bacteria such as extended-spectrum beta-lactamase (ESBL) producing Escherichia coli since they often lack the natural genitourinary barriers to infection. In addition, these patients more often undergo multiple antibiotic courses for their frequent infections, which also contributes to their risk of ESBL infections. Genitourinary abnormalities interfere with normal voiding, resulting in impaired flushing of bacteria. For instance, obstruction inhibits complete urinary drainage and increases the persistence of bacteria in biofilms, especially if there are stones or indwelling devices present. Biofilms usually contain a high concentration of organisms including Proteus mirabilis, Morgenella morganii, and Providencia spp.4 Keep in mind that, if there is an obstruction, the urinalysis might be without pyuria or bacteriuria.
Instrumentation increases infection risks through the direct introduction of bacteria into the genitourinary tract. Despite the efforts in maintaining sterility in urinary catheter placement, catheters provide a nidus for infection. Catheter-associated UTI (CAUTI) is defined by the Infectious Disease Society of America as UTIs that occur in patients with an indwelling catheter or who had a catheter removed for less than 48 hours who develop urinary symptoms and cultures positive for uropathogenic bacteria.4 Studies show that in general, patients with indwelling catheters will develop bacteriuria over time, with 10%-25% eventually developing symptoms.
Severity approach
There are other schools of thought that categorize uncomplicated versus complicated UTIs based on the severity of presentation (see Table 2). An uncomplicated UTI would be classified as symptoms and signs of simple cystitis limited to dysuria, frequency, urgency, and suprapubic pain. Using a symptom severity approach, systemic findings such as fever, chills, emesis, flank pain, costovertebral angle tenderness, or other findings of sepsis would be classified as a complicated UTI. These systemic findings would suggest an extension of infection beyond the bladder.
The argument for a symptomatic-based approach of classification is that the severity of symptoms should dictate the degree of management. Not all UTIs in the anatomic approach are severe. In fact, populations that are considered at risk for complicated UTIs by the AUA guidelines in Table 1 often have mild symptomatic cystitis or asymptomatic bacteriuria. Asymptomatic bacteriuria is the colonization of organisms in the urinary tract without active infection. For instance, bacteriuria is present in almost 100% of people with chronic indwelling catheters, 30%-40% of neurogenic bladder requiring intermittent catheterization, and 50% of elderly nursing home residents.4 Not all bacteriuria triggers enough of an inflammatory response to cause symptoms that require treatment.
Ultimate clinical judgment
Although there are multiple different society recommendations in distinguishing uncomplicated versus complicated UTIs, considering both anatomical and severity risk factors can better aid in clinical decision-making rather than abiding by one classification method alone.
Uncomplicated UTIs from the AUA guidelines can cause severe infections that might require longer courses of broad-spectrum antibiotics. On the other hand, people with anatomic abnormalities can present with mild symptoms that can be treated with a narrow-spectrum antibiotic for a standard time course. Recognizing the severity of the infection and using clinical judgment aids in antibiotic stewardship.
Although the existence of algorithmic approaches can help guide clinical judgment, accounting for the spectrum of host and bacterial factors should ultimately determine the complexity of the disease and management.3 Using clinical suspicion to determine when a UTI should be treated as a complicated infection can ensure effective treatment and decrease the likelihood of sepsis, renal scarring, or end-stage disease.5
Back to the case
The case presents an elderly woman with diabetes presenting with sepsis from a UTI. Because of a normal urinary tract and no prior instrumentation, by the AUA definition, she would be classified as an uncomplicated UTI; however, we would classify her as a complicated UTI based on the severity of her presentation. She has a fever, tachycardia, flank pain, and costovertebral angle tenderness that are evidence of infection extending beyond the bladder. She has sepsis warranting inpatient management. Prior urine culture results could aid in determining empiric treatment while waiting for new cultures. In her case, an intravenous antibiotic with broad gram-negative coverage such as ceftriaxone would be appropriate.
Bottom line
There are multiple interpretations of complicated UTIs including both an anatomical and severity approach. Clinical judgment regarding infection severity should determine the depth of management.
Dr. Vu is a hospitalist at the University of Kentucky, Lexington. Dr. Gray is a hospitalist at the University of Kentucky and the Lexington Veterans Affairs Medical Center.
References
1. Folk CS. AUA Core Curriculum: Urinary Tract Infection (Adult). 2021 Mar 1. https://university.auanet.org/core_topic.cfm?coreid=92.
2. Gupta K et al. International clinical practice guidelines for the treatment of acute uncomplicated cystitis and pyelonephritis in women: A 2010 update by the Infectious Diseases Society of America and the European Society for Microbiology and Infectious Diseases. Clin Infect Dis. 2011 Mar 1;52(5):e103-20. doi: 10.1093/cid/ciq257.
3. Johnson JR. Definition of Complicated Urinary Tract Infection. Clin Infect Dis. 2017 February 15;64(4):529. doi: 10.1093/cid/ciw751.
4. Nicolle LE, AMMI Canada Guidelines Committee. Complicated urinary tract infection in adults. Can J Infect Dis Med Microbiol. 2005;16(6):349-60. doi: 10.1155/2005/385768.
5. Melekos MD and Naber KG. Complicated urinary tract infections. Int J Antimicrob Agents. 2000;15(4):247-56. doi: 10.1016/s0924-8579(00)00168-0.
Key points
- The anatomical approach to defining complicated UTIs considers the presence of underlying, predisposing conditions such as structurally or functionally abnormal genitourinary tract or urinary instrumentation or foreign bodies.
- The severity approach to defining complicated UTIs considers the severity of presentation including the presence of systemic manifestations.
- Both approaches should consider populations that are at risk for recurrent or multidrug-resistant infections and infections that can lead to high morbidity.
- Either approach can be used as a guide, but neither should replace clinical suspicion and judgment in determining the depth of treatment.
Additional reading
Choe HS et al. Summary of the UAA‐AAUS guidelines for urinary tract infections. Int J Urol. 2018 Mar;25(3):175-85. doi:10.1111/iju.13493.
Nicolle LE et al. Infectious Diseases Society of America Guidelines for the Diagnosis and Treatment of Asymptomatic Bacteriuria in Adults. Clin Infect Dis. 2005 Mar;40(5):643-54. doi: 10.1086/427507.
Wagenlehner FME et al. Epidemiology, definition and treatment of complicated urinary tract infections. Nat Rev Urol. 2020 Oct;17:586-600. doi:10.1038/s41585-020-0362-4.
Wallace DW et al. Urinalysis: A simple test with complicated interpretation. J Urgent Care Med. 2020 July-Aug;14(10):11-4.
Quiz
A 68-year-old woman with type 2 diabetes mellitus presents to the emergency department with acute fever, chills, dysuria, frequency, and suprapubic pain. She has associated nausea, malaise, and fatigue. She takes metformin and denies recent antibiotic use. Her temperature is 102.8° F, heart rate 118 beats per minute, blood pressure 118/71 mm Hg, and her respiratory rate is 24 breaths per minute. She is ill-appearing and has mild suprapubic tenderness. White blood cell count is 18 k/mcL. Urinalysis is positive for leukocyte esterase, nitrites, and bacteria. Urine microscopy has 120 white blood cells per high power field. What is the most appropriate treatment?
A. Azithromycin
B. Ceftriaxone
C. Cefepime and vancomycin
D. Nitrofurantoin
The answer is B. The patient presents with sepsis secondary to a urinary tract infection. Using the anatomic approach this would be classified as uncomplicated. Using the severity approach, this would be classified as a complicated urinary tract infection. With fever, chills, and signs of sepsis, it’s likely her infection extends beyond the bladder. Given the severity of her presentation, we’d favor treating her as a complicated urinary tract infection with intravenous ceftriaxone. There is no suggestion of resistance or additional MRSA risk factors requiring intravenous vancomycin or cefepime. Nitrofurantoin, although a first-line treatment for uncomplicated cystitis, would not be appropriate if there is suspicion infection extends beyond the bladder. Azithromycin is a first-line option for chlamydia trachomatis, but not a urinary tract infection.










