User login
Europe Is Facing a Pancreatic Cancer Emergency
“It’s a health emergency for society, with mortality rates at over 90%,” warned Professor Alfredo Carrato, MD, PhD, the chairperson of Pancreatic Cancer Europe.
There are many challenges with pancreatic cancer: Lack of awareness, difficult to diagnose, no screening programs for the general population, poor survival rate, and limited treatment options.
Life expectancy at the time of diagnosis is just 4.6 months. Only 3% of people diagnosed with pancreatic cancer will survive for 5 years.
A 2024 systematic analysis in The Lancet suggested that people living in Western Europe are more likely to develop pancreatic cancer than those living anywhere else in the world.
Dr. Carrato, emeritus professor of medical oncology at the University of Alcalá, Madrid, Spain, wasn’t surprised. He told this news organization: “I think the lifestyle in Europe plays a part. We have all of the risk factors in society like obesity, our sedentary behavior, too much red meat consumption, and excess alcohol intake.”
Other risk factors include smoking, diabetes, chronic pancreatitis, and a family history.
The incidence of pancreatic cancer increases with age, so the longer life expectancy in Western Europe could also contribute to the region’s high rates.
A Silent Killer
Pancreatic cancer is difficult to identify. It is often asymptomatic. Symptoms that do show themselves, like back pain, weight loss, and nausea, are nonspecific and make early diagnosis challenging.
Professor Mattias Löhr from the Karolinska Institutet, Stockholm, Sweden, told this news organization: “It’s a dismal disease. It’s not accessible for any easy screening or surveillance. Even early diagnosis is too late with pancreatic cancer.”
There have been few advancements in patient outcomes over the past few decades.
Only about 20% of patients are suitable candidates for surgery that could prolong their lives.
Also, cancer reoccurs in two thirds of surgical candidates, said Dr. Carrato, and oncologists don’t know how to identify them in advance. “I have patients who survive 3 months and others who survive 4 or 5 years, so there’s a need to identify these subtypes at a molecular level for personalized, clinical, and translational research and therapies.”
Dr. Löhr agreed: “All of the medical therapies are not really working well enough for pancreatic cancer in sharp contrast to other cancers.”
How Can Rates Be Reduced?
“Pancreatic Cancer Europe is working in every EU state to try to raise awareness,” said Dr. Carrato. “We should have primary prevention programs to modify lifestyle risks. We also need funds for translational and clinical research. Secondary prevention isn’t possible yet as we haven’t identified the higher-risk population who would be the target for screening.”
Screening programs are available for the 10% of people who have a family history of pancreatic cancer. But, for the vast majority, there are no tests or screenings that allow for earlier detection.
“We need blood or stool tests that have high specificity and sensitivity that are cost-effective,” said Dr. Carrato.
“It’s a type of cancer with a particular and very aggressive biology. There is a lack of pancreatic tumor tissue for research, as many patients are diagnosed by fine-needle aspiration cytology. It’s a challenge for researchers. We have no biomarkers available to direct our decisions; no precision oncology,” he added.
Still, there are some encouraging developments.
The European PANCAID project (pancreatic cancer initial detection via liquid biopsy) is trying to find biomarkers to screen at-risk groups for earlier diagnosis via a blood test.
Also, the European Union (EU)-funded PANCAIM project (pancreatic cancer artificial intelligence [AI] for genomics and personalized medicine) has developed an AI algorithm that detects small cancers in CT scans that even experienced radiologists might easily overlook.
The project’s head, Henkjan Huisman, is professor of medical imaging AI at Radboud University Medical Center, Nijmegen, the Netherlands. He told this news organization: “It’s an extremely important step, as 20% of people with pancreas cancer have the ability to undergo surgery, which means they might live substantially longer. We believe if the tumors are found earlier, thanks to the algorithm, they are smaller and more contained, and so substantially more than 20% of patients would be suitable for surgery, which would be a breakthrough.”
Dr. Löhr added that a messenger RNA vaccine is being developed in the United States to prevent pancreatic cancer from returning after surgery and is demonstrating encouraging results in its early trials.
The Road Toward Better Care
To improve cancer care in Europe, Dr. Carrato said: “Reference centers should be a requirement in health policy programs because the outcomes are much better than in centers which only perform fewer surgeries, and Pancreatic Cancer Europe is working with the EU in this direction.”
Finland is a country that appears to have succeeded in this regard. Its 2005 Health Care Act, for example, ensures that cancer patients are able to receive care in one of its five specialized hospitals.
More research funding is also needed. According to Pancreatic Cancer Europe, only 2% of EU funding on cancer is spent on pancreatic cancer.
The American Cancer Society’s Cancer Facts & Figures 2024 makes room for some optimism, with the 5-year survival rate in the United States jumping to 13% from 6% in the society’s 2014 report, as a result of earlier diagnoses and more personalized treatment. But, even with potentially longer survival rates, said Dr. Löhr, “we are still on the trajectory of pancreatic cancer being number two for cancer deaths by 2030.”
“We need more money on research, centralized surgery, and networking between European countries,” said Dr. Carrato. “This networking would need more money for prevention, better diagnosis, and treatment. The problem is pancreatic cancer incidence is increasing and mortality is also in parallel, and we are not making real progress in this scenario.”
A version of this article appeared on Medscape.com.
“It’s a health emergency for society, with mortality rates at over 90%,” warned Professor Alfredo Carrato, MD, PhD, the chairperson of Pancreatic Cancer Europe.
There are many challenges with pancreatic cancer: Lack of awareness, difficult to diagnose, no screening programs for the general population, poor survival rate, and limited treatment options.
Life expectancy at the time of diagnosis is just 4.6 months. Only 3% of people diagnosed with pancreatic cancer will survive for 5 years.
A 2024 systematic analysis in The Lancet suggested that people living in Western Europe are more likely to develop pancreatic cancer than those living anywhere else in the world.
Dr. Carrato, emeritus professor of medical oncology at the University of Alcalá, Madrid, Spain, wasn’t surprised. He told this news organization: “I think the lifestyle in Europe plays a part. We have all of the risk factors in society like obesity, our sedentary behavior, too much red meat consumption, and excess alcohol intake.”
Other risk factors include smoking, diabetes, chronic pancreatitis, and a family history.
The incidence of pancreatic cancer increases with age, so the longer life expectancy in Western Europe could also contribute to the region’s high rates.
A Silent Killer
Pancreatic cancer is difficult to identify. It is often asymptomatic. Symptoms that do show themselves, like back pain, weight loss, and nausea, are nonspecific and make early diagnosis challenging.
Professor Mattias Löhr from the Karolinska Institutet, Stockholm, Sweden, told this news organization: “It’s a dismal disease. It’s not accessible for any easy screening or surveillance. Even early diagnosis is too late with pancreatic cancer.”
There have been few advancements in patient outcomes over the past few decades.
Only about 20% of patients are suitable candidates for surgery that could prolong their lives.
Also, cancer reoccurs in two thirds of surgical candidates, said Dr. Carrato, and oncologists don’t know how to identify them in advance. “I have patients who survive 3 months and others who survive 4 or 5 years, so there’s a need to identify these subtypes at a molecular level for personalized, clinical, and translational research and therapies.”
Dr. Löhr agreed: “All of the medical therapies are not really working well enough for pancreatic cancer in sharp contrast to other cancers.”
How Can Rates Be Reduced?
“Pancreatic Cancer Europe is working in every EU state to try to raise awareness,” said Dr. Carrato. “We should have primary prevention programs to modify lifestyle risks. We also need funds for translational and clinical research. Secondary prevention isn’t possible yet as we haven’t identified the higher-risk population who would be the target for screening.”
Screening programs are available for the 10% of people who have a family history of pancreatic cancer. But, for the vast majority, there are no tests or screenings that allow for earlier detection.
“We need blood or stool tests that have high specificity and sensitivity that are cost-effective,” said Dr. Carrato.
“It’s a type of cancer with a particular and very aggressive biology. There is a lack of pancreatic tumor tissue for research, as many patients are diagnosed by fine-needle aspiration cytology. It’s a challenge for researchers. We have no biomarkers available to direct our decisions; no precision oncology,” he added.
Still, there are some encouraging developments.
The European PANCAID project (pancreatic cancer initial detection via liquid biopsy) is trying to find biomarkers to screen at-risk groups for earlier diagnosis via a blood test.
Also, the European Union (EU)-funded PANCAIM project (pancreatic cancer artificial intelligence [AI] for genomics and personalized medicine) has developed an AI algorithm that detects small cancers in CT scans that even experienced radiologists might easily overlook.
The project’s head, Henkjan Huisman, is professor of medical imaging AI at Radboud University Medical Center, Nijmegen, the Netherlands. He told this news organization: “It’s an extremely important step, as 20% of people with pancreas cancer have the ability to undergo surgery, which means they might live substantially longer. We believe if the tumors are found earlier, thanks to the algorithm, they are smaller and more contained, and so substantially more than 20% of patients would be suitable for surgery, which would be a breakthrough.”
Dr. Löhr added that a messenger RNA vaccine is being developed in the United States to prevent pancreatic cancer from returning after surgery and is demonstrating encouraging results in its early trials.
The Road Toward Better Care
To improve cancer care in Europe, Dr. Carrato said: “Reference centers should be a requirement in health policy programs because the outcomes are much better than in centers which only perform fewer surgeries, and Pancreatic Cancer Europe is working with the EU in this direction.”
Finland is a country that appears to have succeeded in this regard. Its 2005 Health Care Act, for example, ensures that cancer patients are able to receive care in one of its five specialized hospitals.
More research funding is also needed. According to Pancreatic Cancer Europe, only 2% of EU funding on cancer is spent on pancreatic cancer.
The American Cancer Society’s Cancer Facts & Figures 2024 makes room for some optimism, with the 5-year survival rate in the United States jumping to 13% from 6% in the society’s 2014 report, as a result of earlier diagnoses and more personalized treatment. But, even with potentially longer survival rates, said Dr. Löhr, “we are still on the trajectory of pancreatic cancer being number two for cancer deaths by 2030.”
“We need more money on research, centralized surgery, and networking between European countries,” said Dr. Carrato. “This networking would need more money for prevention, better diagnosis, and treatment. The problem is pancreatic cancer incidence is increasing and mortality is also in parallel, and we are not making real progress in this scenario.”
A version of this article appeared on Medscape.com.
“It’s a health emergency for society, with mortality rates at over 90%,” warned Professor Alfredo Carrato, MD, PhD, the chairperson of Pancreatic Cancer Europe.
There are many challenges with pancreatic cancer: Lack of awareness, difficult to diagnose, no screening programs for the general population, poor survival rate, and limited treatment options.
Life expectancy at the time of diagnosis is just 4.6 months. Only 3% of people diagnosed with pancreatic cancer will survive for 5 years.
A 2024 systematic analysis in The Lancet suggested that people living in Western Europe are more likely to develop pancreatic cancer than those living anywhere else in the world.
Dr. Carrato, emeritus professor of medical oncology at the University of Alcalá, Madrid, Spain, wasn’t surprised. He told this news organization: “I think the lifestyle in Europe plays a part. We have all of the risk factors in society like obesity, our sedentary behavior, too much red meat consumption, and excess alcohol intake.”
Other risk factors include smoking, diabetes, chronic pancreatitis, and a family history.
The incidence of pancreatic cancer increases with age, so the longer life expectancy in Western Europe could also contribute to the region’s high rates.
A Silent Killer
Pancreatic cancer is difficult to identify. It is often asymptomatic. Symptoms that do show themselves, like back pain, weight loss, and nausea, are nonspecific and make early diagnosis challenging.
Professor Mattias Löhr from the Karolinska Institutet, Stockholm, Sweden, told this news organization: “It’s a dismal disease. It’s not accessible for any easy screening or surveillance. Even early diagnosis is too late with pancreatic cancer.”
There have been few advancements in patient outcomes over the past few decades.
Only about 20% of patients are suitable candidates for surgery that could prolong their lives.
Also, cancer reoccurs in two thirds of surgical candidates, said Dr. Carrato, and oncologists don’t know how to identify them in advance. “I have patients who survive 3 months and others who survive 4 or 5 years, so there’s a need to identify these subtypes at a molecular level for personalized, clinical, and translational research and therapies.”
Dr. Löhr agreed: “All of the medical therapies are not really working well enough for pancreatic cancer in sharp contrast to other cancers.”
How Can Rates Be Reduced?
“Pancreatic Cancer Europe is working in every EU state to try to raise awareness,” said Dr. Carrato. “We should have primary prevention programs to modify lifestyle risks. We also need funds for translational and clinical research. Secondary prevention isn’t possible yet as we haven’t identified the higher-risk population who would be the target for screening.”
Screening programs are available for the 10% of people who have a family history of pancreatic cancer. But, for the vast majority, there are no tests or screenings that allow for earlier detection.
“We need blood or stool tests that have high specificity and sensitivity that are cost-effective,” said Dr. Carrato.
“It’s a type of cancer with a particular and very aggressive biology. There is a lack of pancreatic tumor tissue for research, as many patients are diagnosed by fine-needle aspiration cytology. It’s a challenge for researchers. We have no biomarkers available to direct our decisions; no precision oncology,” he added.
Still, there are some encouraging developments.
The European PANCAID project (pancreatic cancer initial detection via liquid biopsy) is trying to find biomarkers to screen at-risk groups for earlier diagnosis via a blood test.
Also, the European Union (EU)-funded PANCAIM project (pancreatic cancer artificial intelligence [AI] for genomics and personalized medicine) has developed an AI algorithm that detects small cancers in CT scans that even experienced radiologists might easily overlook.
The project’s head, Henkjan Huisman, is professor of medical imaging AI at Radboud University Medical Center, Nijmegen, the Netherlands. He told this news organization: “It’s an extremely important step, as 20% of people with pancreas cancer have the ability to undergo surgery, which means they might live substantially longer. We believe if the tumors are found earlier, thanks to the algorithm, they are smaller and more contained, and so substantially more than 20% of patients would be suitable for surgery, which would be a breakthrough.”
Dr. Löhr added that a messenger RNA vaccine is being developed in the United States to prevent pancreatic cancer from returning after surgery and is demonstrating encouraging results in its early trials.
The Road Toward Better Care
To improve cancer care in Europe, Dr. Carrato said: “Reference centers should be a requirement in health policy programs because the outcomes are much better than in centers which only perform fewer surgeries, and Pancreatic Cancer Europe is working with the EU in this direction.”
Finland is a country that appears to have succeeded in this regard. Its 2005 Health Care Act, for example, ensures that cancer patients are able to receive care in one of its five specialized hospitals.
More research funding is also needed. According to Pancreatic Cancer Europe, only 2% of EU funding on cancer is spent on pancreatic cancer.
The American Cancer Society’s Cancer Facts & Figures 2024 makes room for some optimism, with the 5-year survival rate in the United States jumping to 13% from 6% in the society’s 2014 report, as a result of earlier diagnoses and more personalized treatment. But, even with potentially longer survival rates, said Dr. Löhr, “we are still on the trajectory of pancreatic cancer being number two for cancer deaths by 2030.”
“We need more money on research, centralized surgery, and networking between European countries,” said Dr. Carrato. “This networking would need more money for prevention, better diagnosis, and treatment. The problem is pancreatic cancer incidence is increasing and mortality is also in parallel, and we are not making real progress in this scenario.”
A version of this article appeared on Medscape.com.
Prolonged Sitting at Work Ups CVD and All-Cause Mortality, Daily Breaks May Help
However, daily breaks from sitting and leisure-time activity can help mitigate the “serious” risks associated with prolonged occupational sitting, the researchers say.
“As part of modern lifestyles, prolonged occupational sitting is considered normal and has not received due attention, even though its deleterious effect on health outcomes has been demonstrated,” wrote the authors, led by Wayne Gao, PhD, with Taipei Medical University College of Public Health, Taipei City, Taiwan.
“The importance of physical activity and moving around can never be overstated,” Michelle Bloom, MD, director of the cardio-oncology program at NYU Langone Health in New York, who wasn’t involved in the study, told this news organization.
“As a cardiologist, I bring this up at almost every visit with every patient regardless of why they’re seeing me, because I think that patients respond better when their doctor says it than when they just kind of know it in the back of their mind,” said Dr. Bloom, who is also a professor in the Division of Cardiology, NYU Grossman Long Island School of Medicine, New York.
The study was published online in JAMA Network Open.
Prolonged Sitting Hard on the Heart
2020 marked the first time that guidelines on physical activity from the World Health Organization recommended reducing sedentary behaviors owing to their health consequences. Less is known on the specific association of prolonged occupational sitting with health outcomes, especially in the context of low physical activity.
For their study, Dr. Gao and colleagues quantified health risks associated with prolonged sitting on the job and determined whether a certain threshold of physical activity may attenuate this risk.
Participants included 481,688 adults (mean age, 39 years; 53% women) in a health surveillance program in Taiwan. Data on occupational sitting, leisure-time physical activity, lifestyle, and metabolic parameters were collected.
During an average follow up of nearly 13 years, 26,257 participants died; more than half (57%) of the deaths occurred in individuals who mostly sat at work. There were 5371 CVD-related deaths, with 60% occurring in the mostly sitting group.
In multivariate analysis that adjusted for sex, age, education, smoking, drinking, and body mass index, adults who mostly sat at work had a 16% higher risk of dying of any cause (hazard ratio [HR], 1.16; 95% CI, 1.11-1.20) and a 34% increased risk of dying of CVD (HR, 1.34; 95% CI, 1.22-1.46) compared with those who mostly did not sit at work.
Adults who mostly alternated between sitting and not sitting at work were not at increased risk of all-cause mortality compared with individuals who mostly did not at work (HR, 1.01; 95% CI, 0.97-1.05).
Among adults who mostly sat at work and engaged in low (15-29 minutes) or no (< 15 minutes) daily leisure-time activity, increasing activity by 15 and 30 minutes per day, respectively, lowered the risk for mortality to a level similar to that of inactive individuals who mostly do not sit at work.
“Overall, our findings from a large prospective cohort help to strengthen the increasingly accumulating evidence linking a sedentary lifestyle and health risks,” the authors wrote.
“Systemic changes, such as more frequent breaks, standing desks, designated workplace areas for physical activity, and gym membership benefits, can help reduce risk,” they added.
Simple Yet Profound Message
Reached for comment, Anu Lala, MD, with Icahn School of Medicine at Mount Sinai and Mount Sinai Fuster Heart Hospital in New York, said this study provides a “simple yet profound message” about the dangers of prolonged sitting.
The finding of a 16% higher all-cause mortality in those who mostly sat at work after adjustment for major risk factors is “pretty remarkable. And for CVD mortality, it’s double that,” Dr. Lala told this news organization.
“I think we undervalue the importance of movement, however simple it is. Even simple actions, like squatting and standing up have benefits for the heart,” Dr. Lala added.
Dr. Bloom said she tells her patients, “You don’t have to go out tomorrow and run a marathon. Just get up a few times a day, walk a few laps in your office, walk back and forth from the mailbox, walk up and down your steps a couple of times — just do something more than you’re doing already.”
The study had no commercial funding. Dr. Gao and Dr. Bloom have no relevant disclosures. Dr. Lala has serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for Novartis, AstraZeneca, Merck, Bayer, Novo Nordisk, Cordio, Zoll, and Sequana Medical.
A version of this article appeared on Medscape.com.
However, daily breaks from sitting and leisure-time activity can help mitigate the “serious” risks associated with prolonged occupational sitting, the researchers say.
“As part of modern lifestyles, prolonged occupational sitting is considered normal and has not received due attention, even though its deleterious effect on health outcomes has been demonstrated,” wrote the authors, led by Wayne Gao, PhD, with Taipei Medical University College of Public Health, Taipei City, Taiwan.
“The importance of physical activity and moving around can never be overstated,” Michelle Bloom, MD, director of the cardio-oncology program at NYU Langone Health in New York, who wasn’t involved in the study, told this news organization.
“As a cardiologist, I bring this up at almost every visit with every patient regardless of why they’re seeing me, because I think that patients respond better when their doctor says it than when they just kind of know it in the back of their mind,” said Dr. Bloom, who is also a professor in the Division of Cardiology, NYU Grossman Long Island School of Medicine, New York.
The study was published online in JAMA Network Open.
Prolonged Sitting Hard on the Heart
2020 marked the first time that guidelines on physical activity from the World Health Organization recommended reducing sedentary behaviors owing to their health consequences. Less is known on the specific association of prolonged occupational sitting with health outcomes, especially in the context of low physical activity.
For their study, Dr. Gao and colleagues quantified health risks associated with prolonged sitting on the job and determined whether a certain threshold of physical activity may attenuate this risk.
Participants included 481,688 adults (mean age, 39 years; 53% women) in a health surveillance program in Taiwan. Data on occupational sitting, leisure-time physical activity, lifestyle, and metabolic parameters were collected.
During an average follow up of nearly 13 years, 26,257 participants died; more than half (57%) of the deaths occurred in individuals who mostly sat at work. There were 5371 CVD-related deaths, with 60% occurring in the mostly sitting group.
In multivariate analysis that adjusted for sex, age, education, smoking, drinking, and body mass index, adults who mostly sat at work had a 16% higher risk of dying of any cause (hazard ratio [HR], 1.16; 95% CI, 1.11-1.20) and a 34% increased risk of dying of CVD (HR, 1.34; 95% CI, 1.22-1.46) compared with those who mostly did not sit at work.
Adults who mostly alternated between sitting and not sitting at work were not at increased risk of all-cause mortality compared with individuals who mostly did not at work (HR, 1.01; 95% CI, 0.97-1.05).
Among adults who mostly sat at work and engaged in low (15-29 minutes) or no (< 15 minutes) daily leisure-time activity, increasing activity by 15 and 30 minutes per day, respectively, lowered the risk for mortality to a level similar to that of inactive individuals who mostly do not sit at work.
“Overall, our findings from a large prospective cohort help to strengthen the increasingly accumulating evidence linking a sedentary lifestyle and health risks,” the authors wrote.
“Systemic changes, such as more frequent breaks, standing desks, designated workplace areas for physical activity, and gym membership benefits, can help reduce risk,” they added.
Simple Yet Profound Message
Reached for comment, Anu Lala, MD, with Icahn School of Medicine at Mount Sinai and Mount Sinai Fuster Heart Hospital in New York, said this study provides a “simple yet profound message” about the dangers of prolonged sitting.
The finding of a 16% higher all-cause mortality in those who mostly sat at work after adjustment for major risk factors is “pretty remarkable. And for CVD mortality, it’s double that,” Dr. Lala told this news organization.
“I think we undervalue the importance of movement, however simple it is. Even simple actions, like squatting and standing up have benefits for the heart,” Dr. Lala added.
Dr. Bloom said she tells her patients, “You don’t have to go out tomorrow and run a marathon. Just get up a few times a day, walk a few laps in your office, walk back and forth from the mailbox, walk up and down your steps a couple of times — just do something more than you’re doing already.”
The study had no commercial funding. Dr. Gao and Dr. Bloom have no relevant disclosures. Dr. Lala has serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for Novartis, AstraZeneca, Merck, Bayer, Novo Nordisk, Cordio, Zoll, and Sequana Medical.
A version of this article appeared on Medscape.com.
However, daily breaks from sitting and leisure-time activity can help mitigate the “serious” risks associated with prolonged occupational sitting, the researchers say.
“As part of modern lifestyles, prolonged occupational sitting is considered normal and has not received due attention, even though its deleterious effect on health outcomes has been demonstrated,” wrote the authors, led by Wayne Gao, PhD, with Taipei Medical University College of Public Health, Taipei City, Taiwan.
“The importance of physical activity and moving around can never be overstated,” Michelle Bloom, MD, director of the cardio-oncology program at NYU Langone Health in New York, who wasn’t involved in the study, told this news organization.
“As a cardiologist, I bring this up at almost every visit with every patient regardless of why they’re seeing me, because I think that patients respond better when their doctor says it than when they just kind of know it in the back of their mind,” said Dr. Bloom, who is also a professor in the Division of Cardiology, NYU Grossman Long Island School of Medicine, New York.
The study was published online in JAMA Network Open.
Prolonged Sitting Hard on the Heart
2020 marked the first time that guidelines on physical activity from the World Health Organization recommended reducing sedentary behaviors owing to their health consequences. Less is known on the specific association of prolonged occupational sitting with health outcomes, especially in the context of low physical activity.
For their study, Dr. Gao and colleagues quantified health risks associated with prolonged sitting on the job and determined whether a certain threshold of physical activity may attenuate this risk.
Participants included 481,688 adults (mean age, 39 years; 53% women) in a health surveillance program in Taiwan. Data on occupational sitting, leisure-time physical activity, lifestyle, and metabolic parameters were collected.
During an average follow up of nearly 13 years, 26,257 participants died; more than half (57%) of the deaths occurred in individuals who mostly sat at work. There were 5371 CVD-related deaths, with 60% occurring in the mostly sitting group.
In multivariate analysis that adjusted for sex, age, education, smoking, drinking, and body mass index, adults who mostly sat at work had a 16% higher risk of dying of any cause (hazard ratio [HR], 1.16; 95% CI, 1.11-1.20) and a 34% increased risk of dying of CVD (HR, 1.34; 95% CI, 1.22-1.46) compared with those who mostly did not sit at work.
Adults who mostly alternated between sitting and not sitting at work were not at increased risk of all-cause mortality compared with individuals who mostly did not at work (HR, 1.01; 95% CI, 0.97-1.05).
Among adults who mostly sat at work and engaged in low (15-29 minutes) or no (< 15 minutes) daily leisure-time activity, increasing activity by 15 and 30 minutes per day, respectively, lowered the risk for mortality to a level similar to that of inactive individuals who mostly do not sit at work.
“Overall, our findings from a large prospective cohort help to strengthen the increasingly accumulating evidence linking a sedentary lifestyle and health risks,” the authors wrote.
“Systemic changes, such as more frequent breaks, standing desks, designated workplace areas for physical activity, and gym membership benefits, can help reduce risk,” they added.
Simple Yet Profound Message
Reached for comment, Anu Lala, MD, with Icahn School of Medicine at Mount Sinai and Mount Sinai Fuster Heart Hospital in New York, said this study provides a “simple yet profound message” about the dangers of prolonged sitting.
The finding of a 16% higher all-cause mortality in those who mostly sat at work after adjustment for major risk factors is “pretty remarkable. And for CVD mortality, it’s double that,” Dr. Lala told this news organization.
“I think we undervalue the importance of movement, however simple it is. Even simple actions, like squatting and standing up have benefits for the heart,” Dr. Lala added.
Dr. Bloom said she tells her patients, “You don’t have to go out tomorrow and run a marathon. Just get up a few times a day, walk a few laps in your office, walk back and forth from the mailbox, walk up and down your steps a couple of times — just do something more than you’re doing already.”
The study had no commercial funding. Dr. Gao and Dr. Bloom have no relevant disclosures. Dr. Lala has serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for Novartis, AstraZeneca, Merck, Bayer, Novo Nordisk, Cordio, Zoll, and Sequana Medical.
A version of this article appeared on Medscape.com.
FROM JAMA NETWORK OPEN
High-dose RT and long-term ADT improve survival in high-risk prostate cancer
For patients with high-risk prostate cancer, treatment with long-term androgen deprivation therapy (ADT) and high-dose radiation was associated with significantly better progression-free, cancer-specific, and overall survival compared with ADT and standard-dose radiation.
The investigators also found that the patients taking long-term ADT and high-dose radiation did not experience additional late urinary tract or gastrointestinal toxicities. Christophe Hennequin, MD, PhD, reported these and other findings of the Radiation Therapy in Treating Patients Receiving Hormone Therapy for Prostate Cancer (GETUG-AFU 18) trial, at the 2024 American Society for Clinical Oncology (ASCO) Genitourinary Cancers Symposium.
Among 505 patients randomly assigned to be treated with radiation therapy (RT) at either the standard 70 Gy dose or a high, 80 Gy dose followed by 3 years of adjuvant ADT, the 10-year progression-free survival (PFS) rate was 83.6% for patients who had received the 80 Gy dose, vs. 72.2% for patients who had received the 70 Gy dose. This translated into a hazard ratio (HR) for biochemical or clinical progression of 0.56 (P = .0005).
This PFS advantage for high-dose radiation was also reflected by an overall survival (OS) advantage, with 10-year OS rates of 77% vs. 65.9%, respectively, translating into a 39% reduction in risk of death (HR 0.61, P = .0039) for patients who had received the higher radiation dose, reported Dr. Hennequin, of the Hospital Saint Louis in Paris, France.
"We have now Level 1 evidence that high-dose RT with long-term ADT must be the standard of care in high-risk prostate cancer patients," he said at the meeting.
Dr. Hennequin noted that significantly more patients assigned to high-dose RT were treated with intensity modulated radiation therapy (IMRT) rather than conventional beam radiation, and emphasized that the superior results seen with the higher dose is likely due to the use of IMRT.
Prior evidence
Dr. Hennequin pointed to a meta-analysis published in The Lancet in 2022 which showed that among nearly 11,000 patients with a median follow-up of 11.4 years the addition of ADT to RT significantly improved metastasis-free survival, and that longer ADT reduced the risk of metastases by 16% compared with standard schedule ADT.
He also cited the DART 01/05 trial results, which were published in 2022 in The Lancet: Oncology, which found a clinically relevant benefit for 24 months vs. 4 months of adjuvant ADT following a minimum of 76 Gy radiation in patients with high-risk disease, but not among patients with intermediate-risk disease.
The GETUG-AFU 18 trial was designed to address the question of whether 80 Gy of radiation could improve outcomes compared with 70 Gy in patients treated with long-term ADT.
Study details and results
The investigators enrolled men with high-risk prostate cancer defined as either a prostate-specific antigen (PSA) level 20 ng/ml or greater, Gleason score 8 or higher, or clinical stage T3 or T4 disease, and after stratification by treatment center and lymph node resection randomly assigned them to receive either 70 Gy or 80 Gy RT followed by 3 years of ADT.
Approximately two-thirds of the patients in each study arm had one risk factor and about one-fourth had two risk factors. The remaining patients had all three high-risk defining factors.
Approximately 16.5% of patients in each arm had undergone lymph node dissection.
The median ADT duration was 33.4 months. In all, 82.9% of patients underwent pelvic lymph node radiation; lymph node radiation was not performed in those patients who had negative node dissection results.
Significantly more patients assigned to the 80 Gy dose were treated with IMRT (80.6% vs. 58.6%, P < .001).
The cancer-specific survival rate was also higher for the group receiving the 80 Gy dose, with a 10 year rate of 95.6% vs. 90% for patients treated with 70 Gy. This difference translated into a HR of 0.48 (P = .0090).
Comparable safety
The safety analysis, which included 248 patients who received 80 Gy and 251 who received 70 Gy, showed that the incidence rates of both late genitourinary and gastrointestinal toxicities were low and comparable between the groups. Grade 3 or greater late genitourinary toxicities were seen in 2.0% of patients treated with 80 Gy and 3.2% of those treated with 70 Gy. In both arms, only 1.6% of patients had grade 3 or greater later GI toxicities.
There were also no differences between the study arms in patient-reported quality of life measures related to either bowel or urinary symptoms.
Invited discussant Neha Vapiwala, MD, FACR, from Penn Medicine in Philadelphia commented that the results of the GETUG-AFU 18 trial suggest that "if you had even lower-dose systemic therapy that the radiation control at the local level - local-regional level in this case - can in fact contribute to the prevention of distant metastases and can contribute to cancer-specific survival."
She said that with the efficacy results and the comparable toxicity and quality of life measures, dose-escalated radiation therapy and long-term ADT appear to offer a synergistic benefit.
The results are "practice-affirming for many, perhaps practice-changing for some if you're not already offering this," she said.
For patients with high-risk prostate cancer, treatment with long-term androgen deprivation therapy (ADT) and high-dose radiation was associated with significantly better progression-free, cancer-specific, and overall survival compared with ADT and standard-dose radiation.
The investigators also found that the patients taking long-term ADT and high-dose radiation did not experience additional late urinary tract or gastrointestinal toxicities. Christophe Hennequin, MD, PhD, reported these and other findings of the Radiation Therapy in Treating Patients Receiving Hormone Therapy for Prostate Cancer (GETUG-AFU 18) trial, at the 2024 American Society for Clinical Oncology (ASCO) Genitourinary Cancers Symposium.
Among 505 patients randomly assigned to be treated with radiation therapy (RT) at either the standard 70 Gy dose or a high, 80 Gy dose followed by 3 years of adjuvant ADT, the 10-year progression-free survival (PFS) rate was 83.6% for patients who had received the 80 Gy dose, vs. 72.2% for patients who had received the 70 Gy dose. This translated into a hazard ratio (HR) for biochemical or clinical progression of 0.56 (P = .0005).
This PFS advantage for high-dose radiation was also reflected by an overall survival (OS) advantage, with 10-year OS rates of 77% vs. 65.9%, respectively, translating into a 39% reduction in risk of death (HR 0.61, P = .0039) for patients who had received the higher radiation dose, reported Dr. Hennequin, of the Hospital Saint Louis in Paris, France.
"We have now Level 1 evidence that high-dose RT with long-term ADT must be the standard of care in high-risk prostate cancer patients," he said at the meeting.
Dr. Hennequin noted that significantly more patients assigned to high-dose RT were treated with intensity modulated radiation therapy (IMRT) rather than conventional beam radiation, and emphasized that the superior results seen with the higher dose is likely due to the use of IMRT.
Prior evidence
Dr. Hennequin pointed to a meta-analysis published in The Lancet in 2022 which showed that among nearly 11,000 patients with a median follow-up of 11.4 years the addition of ADT to RT significantly improved metastasis-free survival, and that longer ADT reduced the risk of metastases by 16% compared with standard schedule ADT.
He also cited the DART 01/05 trial results, which were published in 2022 in The Lancet: Oncology, which found a clinically relevant benefit for 24 months vs. 4 months of adjuvant ADT following a minimum of 76 Gy radiation in patients with high-risk disease, but not among patients with intermediate-risk disease.
The GETUG-AFU 18 trial was designed to address the question of whether 80 Gy of radiation could improve outcomes compared with 70 Gy in patients treated with long-term ADT.
Study details and results
The investigators enrolled men with high-risk prostate cancer defined as either a prostate-specific antigen (PSA) level 20 ng/ml or greater, Gleason score 8 or higher, or clinical stage T3 or T4 disease, and after stratification by treatment center and lymph node resection randomly assigned them to receive either 70 Gy or 80 Gy RT followed by 3 years of ADT.
Approximately two-thirds of the patients in each study arm had one risk factor and about one-fourth had two risk factors. The remaining patients had all three high-risk defining factors.
Approximately 16.5% of patients in each arm had undergone lymph node dissection.
The median ADT duration was 33.4 months. In all, 82.9% of patients underwent pelvic lymph node radiation; lymph node radiation was not performed in those patients who had negative node dissection results.
Significantly more patients assigned to the 80 Gy dose were treated with IMRT (80.6% vs. 58.6%, P < .001).
The cancer-specific survival rate was also higher for the group receiving the 80 Gy dose, with a 10 year rate of 95.6% vs. 90% for patients treated with 70 Gy. This difference translated into a HR of 0.48 (P = .0090).
Comparable safety
The safety analysis, which included 248 patients who received 80 Gy and 251 who received 70 Gy, showed that the incidence rates of both late genitourinary and gastrointestinal toxicities were low and comparable between the groups. Grade 3 or greater late genitourinary toxicities were seen in 2.0% of patients treated with 80 Gy and 3.2% of those treated with 70 Gy. In both arms, only 1.6% of patients had grade 3 or greater later GI toxicities.
There were also no differences between the study arms in patient-reported quality of life measures related to either bowel or urinary symptoms.
Invited discussant Neha Vapiwala, MD, FACR, from Penn Medicine in Philadelphia commented that the results of the GETUG-AFU 18 trial suggest that "if you had even lower-dose systemic therapy that the radiation control at the local level - local-regional level in this case - can in fact contribute to the prevention of distant metastases and can contribute to cancer-specific survival."
She said that with the efficacy results and the comparable toxicity and quality of life measures, dose-escalated radiation therapy and long-term ADT appear to offer a synergistic benefit.
The results are "practice-affirming for many, perhaps practice-changing for some if you're not already offering this," she said.
For patients with high-risk prostate cancer, treatment with long-term androgen deprivation therapy (ADT) and high-dose radiation was associated with significantly better progression-free, cancer-specific, and overall survival compared with ADT and standard-dose radiation.
The investigators also found that the patients taking long-term ADT and high-dose radiation did not experience additional late urinary tract or gastrointestinal toxicities. Christophe Hennequin, MD, PhD, reported these and other findings of the Radiation Therapy in Treating Patients Receiving Hormone Therapy for Prostate Cancer (GETUG-AFU 18) trial, at the 2024 American Society for Clinical Oncology (ASCO) Genitourinary Cancers Symposium.
Among 505 patients randomly assigned to be treated with radiation therapy (RT) at either the standard 70 Gy dose or a high, 80 Gy dose followed by 3 years of adjuvant ADT, the 10-year progression-free survival (PFS) rate was 83.6% for patients who had received the 80 Gy dose, vs. 72.2% for patients who had received the 70 Gy dose. This translated into a hazard ratio (HR) for biochemical or clinical progression of 0.56 (P = .0005).
This PFS advantage for high-dose radiation was also reflected by an overall survival (OS) advantage, with 10-year OS rates of 77% vs. 65.9%, respectively, translating into a 39% reduction in risk of death (HR 0.61, P = .0039) for patients who had received the higher radiation dose, reported Dr. Hennequin, of the Hospital Saint Louis in Paris, France.
"We have now Level 1 evidence that high-dose RT with long-term ADT must be the standard of care in high-risk prostate cancer patients," he said at the meeting.
Dr. Hennequin noted that significantly more patients assigned to high-dose RT were treated with intensity modulated radiation therapy (IMRT) rather than conventional beam radiation, and emphasized that the superior results seen with the higher dose is likely due to the use of IMRT.
Prior evidence
Dr. Hennequin pointed to a meta-analysis published in The Lancet in 2022 which showed that among nearly 11,000 patients with a median follow-up of 11.4 years the addition of ADT to RT significantly improved metastasis-free survival, and that longer ADT reduced the risk of metastases by 16% compared with standard schedule ADT.
He also cited the DART 01/05 trial results, which were published in 2022 in The Lancet: Oncology, which found a clinically relevant benefit for 24 months vs. 4 months of adjuvant ADT following a minimum of 76 Gy radiation in patients with high-risk disease, but not among patients with intermediate-risk disease.
The GETUG-AFU 18 trial was designed to address the question of whether 80 Gy of radiation could improve outcomes compared with 70 Gy in patients treated with long-term ADT.
Study details and results
The investigators enrolled men with high-risk prostate cancer defined as either a prostate-specific antigen (PSA) level 20 ng/ml or greater, Gleason score 8 or higher, or clinical stage T3 or T4 disease, and after stratification by treatment center and lymph node resection randomly assigned them to receive either 70 Gy or 80 Gy RT followed by 3 years of ADT.
Approximately two-thirds of the patients in each study arm had one risk factor and about one-fourth had two risk factors. The remaining patients had all three high-risk defining factors.
Approximately 16.5% of patients in each arm had undergone lymph node dissection.
The median ADT duration was 33.4 months. In all, 82.9% of patients underwent pelvic lymph node radiation; lymph node radiation was not performed in those patients who had negative node dissection results.
Significantly more patients assigned to the 80 Gy dose were treated with IMRT (80.6% vs. 58.6%, P < .001).
The cancer-specific survival rate was also higher for the group receiving the 80 Gy dose, with a 10 year rate of 95.6% vs. 90% for patients treated with 70 Gy. This difference translated into a HR of 0.48 (P = .0090).
Comparable safety
The safety analysis, which included 248 patients who received 80 Gy and 251 who received 70 Gy, showed that the incidence rates of both late genitourinary and gastrointestinal toxicities were low and comparable between the groups. Grade 3 or greater late genitourinary toxicities were seen in 2.0% of patients treated with 80 Gy and 3.2% of those treated with 70 Gy. In both arms, only 1.6% of patients had grade 3 or greater later GI toxicities.
There were also no differences between the study arms in patient-reported quality of life measures related to either bowel or urinary symptoms.
Invited discussant Neha Vapiwala, MD, FACR, from Penn Medicine in Philadelphia commented that the results of the GETUG-AFU 18 trial suggest that "if you had even lower-dose systemic therapy that the radiation control at the local level - local-regional level in this case - can in fact contribute to the prevention of distant metastases and can contribute to cancer-specific survival."
She said that with the efficacy results and the comparable toxicity and quality of life measures, dose-escalated radiation therapy and long-term ADT appear to offer a synergistic benefit.
The results are "practice-affirming for many, perhaps practice-changing for some if you're not already offering this," she said.
FROM ASCO GU 2024
Dana-Farber Moves to Retract, Correct Dozens of Cancer Papers Amid Allegations
News of the investigation follows a blog post by British molecular biologist Sholto David, MD, who flagged almost 60 papers published between 1997 and 2017 that contained image manipulation and other errors. Some of the papers were published by Dana-Farber’s chief executive officer, Laurie Glimcher, MD, and chief operating officer, William Hahn, MD, on topics including multiple myeloma and immune cells.
Mr. David, who blogs about research integrity, highlighted numerous errors and irregularities, including copying and pasting images across multiple experiments to represent different days within the same experiment, sometimes rotating or stretching images.
In one case, Mr. David equated the manipulation with tactics used by “hapless Chinese papermills” and concluded that “a swathe of research coming out of [Dana-Farber] authored by the most senior researchers and managers appears to be hopelessly corrupt with errors that are obvious from just a cursory reading the papers.”
“Imagine what mistakes might be found in the raw data if anyone was allowed to look!” he wrote.
Barrett Rollins, MD, PhD, Dana-Farber Cancer Institute’s research integrity officer, declined to comment on whether the errors represent scientific misconduct, according to STAT. Rollins told ScienceInsider that the “presence of image discrepancies in a paper is not evidence of an author’s intent to deceive.”
Access to new artificial intelligence tools is making it easier for data sleuths, like Mr. David, to unearth data manipulation and errors.
The current investigation closely follows two other investigations into the published work of Harvard University’s former president, Claudine Gay, and Stanford University’s former president, Marc Tessier-Lavigne, which led both to resign their posts.
A version of this article appeared on Medscape.com.
News of the investigation follows a blog post by British molecular biologist Sholto David, MD, who flagged almost 60 papers published between 1997 and 2017 that contained image manipulation and other errors. Some of the papers were published by Dana-Farber’s chief executive officer, Laurie Glimcher, MD, and chief operating officer, William Hahn, MD, on topics including multiple myeloma and immune cells.
Mr. David, who blogs about research integrity, highlighted numerous errors and irregularities, including copying and pasting images across multiple experiments to represent different days within the same experiment, sometimes rotating or stretching images.
In one case, Mr. David equated the manipulation with tactics used by “hapless Chinese papermills” and concluded that “a swathe of research coming out of [Dana-Farber] authored by the most senior researchers and managers appears to be hopelessly corrupt with errors that are obvious from just a cursory reading the papers.”
“Imagine what mistakes might be found in the raw data if anyone was allowed to look!” he wrote.
Barrett Rollins, MD, PhD, Dana-Farber Cancer Institute’s research integrity officer, declined to comment on whether the errors represent scientific misconduct, according to STAT. Rollins told ScienceInsider that the “presence of image discrepancies in a paper is not evidence of an author’s intent to deceive.”
Access to new artificial intelligence tools is making it easier for data sleuths, like Mr. David, to unearth data manipulation and errors.
The current investigation closely follows two other investigations into the published work of Harvard University’s former president, Claudine Gay, and Stanford University’s former president, Marc Tessier-Lavigne, which led both to resign their posts.
A version of this article appeared on Medscape.com.
News of the investigation follows a blog post by British molecular biologist Sholto David, MD, who flagged almost 60 papers published between 1997 and 2017 that contained image manipulation and other errors. Some of the papers were published by Dana-Farber’s chief executive officer, Laurie Glimcher, MD, and chief operating officer, William Hahn, MD, on topics including multiple myeloma and immune cells.
Mr. David, who blogs about research integrity, highlighted numerous errors and irregularities, including copying and pasting images across multiple experiments to represent different days within the same experiment, sometimes rotating or stretching images.
In one case, Mr. David equated the manipulation with tactics used by “hapless Chinese papermills” and concluded that “a swathe of research coming out of [Dana-Farber] authored by the most senior researchers and managers appears to be hopelessly corrupt with errors that are obvious from just a cursory reading the papers.”
“Imagine what mistakes might be found in the raw data if anyone was allowed to look!” he wrote.
Barrett Rollins, MD, PhD, Dana-Farber Cancer Institute’s research integrity officer, declined to comment on whether the errors represent scientific misconduct, according to STAT. Rollins told ScienceInsider that the “presence of image discrepancies in a paper is not evidence of an author’s intent to deceive.”
Access to new artificial intelligence tools is making it easier for data sleuths, like Mr. David, to unearth data manipulation and errors.
The current investigation closely follows two other investigations into the published work of Harvard University’s former president, Claudine Gay, and Stanford University’s former president, Marc Tessier-Lavigne, which led both to resign their posts.
A version of this article appeared on Medscape.com.
Obstructive Sleep Apnea May Promote Early Bone Loss
TOPLINE:
Indicators of early bone loss were significantly higher in adults with severe obstructive sleep apnea (OSA) than in those with mild or moderate OSA and controls.
METHODOLOGY:
- The researchers enrolled 90 men aged 30-59 years who were patients at a single sleep and respiratory center between August 2017 and February 2019; the average age was 47.1 years, and the average body mass index was 25.7 kg/m2.
- The study population included 25 individuals with mild OSA, 21 with moderate OSA, 34 with severe OSA, and 10 controls without OSA.
- Bone loss was assessed using high-resolution peripheral quantitative computed tomography and blood samples. The researchers collected information on metabolic and inflammatory bone turnover indicators, as well as bone geometric parameters, bone microstructure parameters, and measures of bone mineral density (BMD).
TAKEAWAY:
- Total volumetric bone mineral density was significantly lower in patients with OSA than in controls and significantly different among OSA groups, as were the meta trabecular volumetric BMD, trabecular thickness (Tb.Th), and cortical thickness (Ct.Th).
- Differences in bone microstructure between patients with OSA and controls were most evident in measures of Tb.Th and Ct.Th.
- No significant differences appeared in blood bone turnover indicators or inflammation indicators among the groups.
IN PRACTICE:
“A study with a larger sample is necessary to further assess the relationship and mechanisms between OSA and osteoporosis,” the researchers wrote.
SOURCE:
The lead author on the study was Yixian Qiao, MD, of the Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing, China. The study was published online in BMC Pulmonary Medicine.
LIMITATIONS:
The cross-sectional design, small sample size, and inability to control for several key confounders such as nutritional status and amount of exercise, as well as the exclusion of women and elderly individuals, limited the findings.
DISCLOSURES:
The study was supported by the National Key Research and Development Projects of China. The researchers had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
TOPLINE:
Indicators of early bone loss were significantly higher in adults with severe obstructive sleep apnea (OSA) than in those with mild or moderate OSA and controls.
METHODOLOGY:
- The researchers enrolled 90 men aged 30-59 years who were patients at a single sleep and respiratory center between August 2017 and February 2019; the average age was 47.1 years, and the average body mass index was 25.7 kg/m2.
- The study population included 25 individuals with mild OSA, 21 with moderate OSA, 34 with severe OSA, and 10 controls without OSA.
- Bone loss was assessed using high-resolution peripheral quantitative computed tomography and blood samples. The researchers collected information on metabolic and inflammatory bone turnover indicators, as well as bone geometric parameters, bone microstructure parameters, and measures of bone mineral density (BMD).
TAKEAWAY:
- Total volumetric bone mineral density was significantly lower in patients with OSA than in controls and significantly different among OSA groups, as were the meta trabecular volumetric BMD, trabecular thickness (Tb.Th), and cortical thickness (Ct.Th).
- Differences in bone microstructure between patients with OSA and controls were most evident in measures of Tb.Th and Ct.Th.
- No significant differences appeared in blood bone turnover indicators or inflammation indicators among the groups.
IN PRACTICE:
“A study with a larger sample is necessary to further assess the relationship and mechanisms between OSA and osteoporosis,” the researchers wrote.
SOURCE:
The lead author on the study was Yixian Qiao, MD, of the Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing, China. The study was published online in BMC Pulmonary Medicine.
LIMITATIONS:
The cross-sectional design, small sample size, and inability to control for several key confounders such as nutritional status and amount of exercise, as well as the exclusion of women and elderly individuals, limited the findings.
DISCLOSURES:
The study was supported by the National Key Research and Development Projects of China. The researchers had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
TOPLINE:
Indicators of early bone loss were significantly higher in adults with severe obstructive sleep apnea (OSA) than in those with mild or moderate OSA and controls.
METHODOLOGY:
- The researchers enrolled 90 men aged 30-59 years who were patients at a single sleep and respiratory center between August 2017 and February 2019; the average age was 47.1 years, and the average body mass index was 25.7 kg/m2.
- The study population included 25 individuals with mild OSA, 21 with moderate OSA, 34 with severe OSA, and 10 controls without OSA.
- Bone loss was assessed using high-resolution peripheral quantitative computed tomography and blood samples. The researchers collected information on metabolic and inflammatory bone turnover indicators, as well as bone geometric parameters, bone microstructure parameters, and measures of bone mineral density (BMD).
TAKEAWAY:
- Total volumetric bone mineral density was significantly lower in patients with OSA than in controls and significantly different among OSA groups, as were the meta trabecular volumetric BMD, trabecular thickness (Tb.Th), and cortical thickness (Ct.Th).
- Differences in bone microstructure between patients with OSA and controls were most evident in measures of Tb.Th and Ct.Th.
- No significant differences appeared in blood bone turnover indicators or inflammation indicators among the groups.
IN PRACTICE:
“A study with a larger sample is necessary to further assess the relationship and mechanisms between OSA and osteoporosis,” the researchers wrote.
SOURCE:
The lead author on the study was Yixian Qiao, MD, of the Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing, China. The study was published online in BMC Pulmonary Medicine.
LIMITATIONS:
The cross-sectional design, small sample size, and inability to control for several key confounders such as nutritional status and amount of exercise, as well as the exclusion of women and elderly individuals, limited the findings.
DISCLOSURES:
The study was supported by the National Key Research and Development Projects of China. The researchers had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
Cancer Identified as a New Cardiovascular Risk Factor
A history of cancer is an independent predictor of major cardiovascular events in patients undergoing coronary angioplasty. Cancer should be considered a new cardiovascular risk factor in primary and secondary prevention, according to a study presented at the 2023 American Heart Association Congress in Philadelphia.
, for example, equating it to the situation of a patient with diabetes or chronic renal failure,” said lead author Renzo Melchiori, MD, a cardiologist at the University Hospital Austral in Pilar, Argentina.
The researchers also advocate for intensifying cardiovascular control measures in secondary prevention for these patients, reconsidering goals, and ensuring compliance with prescribed pharmacological regimens and healthy lifestyle habits.
“Previously, when a patient had oncological pathology, thinking about associated cardiovascular risk seemed somewhat superfluous. But today, oncological diseases are treated so effectively, increasing survival and life expectancy, that we begin to focus on what happens with the arteries of these patients after treatment,” said Dr. Melchiori.
Higher Incidence Density
The retrospective analysis included 937 patients of both sexes aged 18 years and older who underwent coronary angioplasty for acute coronary syndrome between 2008 and 2022 at a university hospital. Of these participants, 89 (9.5%) had a history of cancer, with a median time since oncologic diagnosis of around 2 years for solid and hematologic tumors. Most participants had treated and resolved cancer.
Over a median follow-up of 45 months (range, 14-72 months), the cumulative incidence rates of a major cardiovascular event (nonfatal stroke, nonfatal acute myocardial infarction, cardiovascular death, or new angioplasty) were 22.2% (155/698) and 28.4% (25/88) in the groups without and with a history of cancer, respectively. The incidence density was significantly higher in the group with an oncologic history than in the group without such a history: 0.78 events/100 patients/month vs 0.48 events/100 patients/month (P = .01).
Kaplan-Meier analysis showed a higher probability of a major cardiovascular event in the group of patients with cancer or a history of cancer (P = .0086). In multivariate Cox regression analysis, cancer history was an independent predictor of major cardiovascular events adjusted for other risk factors such as age, hypertension, diabetes, smoking, sedentary lifestyle, and family history (hazard ratio, 1.66; P = .025).
Dr. Melchiori clarified that the increased incidence of cardiovascular events in patients with cancer or a history of cancer cannot be attributed to differences in percutaneous intervention or the indication or compliance of post-treatment pharmacological therapy.
In addition, the specialist acknowledged that due to the sample size, discrimination by cancer type, disease stage, or therapeutic strategies couldn’t be performed. A subanalysis, which has not been presented, indicated that the effect could not be explained solely by the application of radiotherapy or chemotherapy in the 90 days before angioplasty — two factors that cause arterial inflammation.
Intensifying Prevention Measures
Two independent experts told this news organization that the new study is "interesting" and reinforces the close connection between oncologic and cardiovascular pathology.
Andrés Daniele, MD, cardiologist and president of the Argentine Cardio-Oncology Association, a local chapter of the International Cardio-Oncology Society, emphasized that the study “reiterates an observation seen in other works: A higher rate of atherosclerotic pathology and cardiovascular events in patients with a history of cancer. And that has a reason to be: Both pathologies present common risk factors, and on the other hand, there is greater endothelial dysfunction secondary to the inflammatory syndrome and oncologic therapies.”
“There needs to be a continuum in the intensification of measures in primary and secondary cardiovascular prevention in cancer survivors, whether in remission or with chronic disease. We need to be very aggressive in managing risk factors and insist that patients who have had a cardiovascular event enter cardiovascular rehabilitation therapies,” said Dr. Daniele, who also heads the Cardio-Oncology Department at the centenary Roffo Institute of Oncology at the University of Buenos Aires, Argentina.
The study provides a valuable contribution because “we need to understand the epidemiology and natural history of patients with cancer at risk of developing cardiovascular complications to implement personalized cardiovascular prevention strategies,” said Teresa López Fernández, MD, cardiologist, coordinator of the Cardio-Oncology Program at La Paz University Hospital in Madrid, member of the Cardio-Oncology Working Group of the Spanish Society of Cardiology, member of the board of the International Cardio-Oncology Society, and cochair of the first clinical practice guidelines in cardio-oncology of the European Society of Cardiology.
“We have to be aware that perhaps we should not guide ourselves in these patients with the usual risk stratification scores as cancer or cardiotoxic treatment are not included as variables. However, they require our attention and effort to improve their quality and quantity of life, avoiding potentially preventable cardiovascular events that could negatively impact the survival achieved thanks to advances in cancer treatments,” said Dr. López Fernández.
Dr. Melchiori and Dr. Daniele declared no relevant economic conflicts of interest. Dr. López Fernández reported relationships with Daiichi Sankyo, Almirall España, Janssen-Cilag, Bayer, Roche, Philips, and Incyte.
This article was translated from the Medscape Spanish edition. A version of this article appeared on Medscape.com.
A history of cancer is an independent predictor of major cardiovascular events in patients undergoing coronary angioplasty. Cancer should be considered a new cardiovascular risk factor in primary and secondary prevention, according to a study presented at the 2023 American Heart Association Congress in Philadelphia.
, for example, equating it to the situation of a patient with diabetes or chronic renal failure,” said lead author Renzo Melchiori, MD, a cardiologist at the University Hospital Austral in Pilar, Argentina.
The researchers also advocate for intensifying cardiovascular control measures in secondary prevention for these patients, reconsidering goals, and ensuring compliance with prescribed pharmacological regimens and healthy lifestyle habits.
“Previously, when a patient had oncological pathology, thinking about associated cardiovascular risk seemed somewhat superfluous. But today, oncological diseases are treated so effectively, increasing survival and life expectancy, that we begin to focus on what happens with the arteries of these patients after treatment,” said Dr. Melchiori.
Higher Incidence Density
The retrospective analysis included 937 patients of both sexes aged 18 years and older who underwent coronary angioplasty for acute coronary syndrome between 2008 and 2022 at a university hospital. Of these participants, 89 (9.5%) had a history of cancer, with a median time since oncologic diagnosis of around 2 years for solid and hematologic tumors. Most participants had treated and resolved cancer.
Over a median follow-up of 45 months (range, 14-72 months), the cumulative incidence rates of a major cardiovascular event (nonfatal stroke, nonfatal acute myocardial infarction, cardiovascular death, or new angioplasty) were 22.2% (155/698) and 28.4% (25/88) in the groups without and with a history of cancer, respectively. The incidence density was significantly higher in the group with an oncologic history than in the group without such a history: 0.78 events/100 patients/month vs 0.48 events/100 patients/month (P = .01).
Kaplan-Meier analysis showed a higher probability of a major cardiovascular event in the group of patients with cancer or a history of cancer (P = .0086). In multivariate Cox regression analysis, cancer history was an independent predictor of major cardiovascular events adjusted for other risk factors such as age, hypertension, diabetes, smoking, sedentary lifestyle, and family history (hazard ratio, 1.66; P = .025).
Dr. Melchiori clarified that the increased incidence of cardiovascular events in patients with cancer or a history of cancer cannot be attributed to differences in percutaneous intervention or the indication or compliance of post-treatment pharmacological therapy.
In addition, the specialist acknowledged that due to the sample size, discrimination by cancer type, disease stage, or therapeutic strategies couldn’t be performed. A subanalysis, which has not been presented, indicated that the effect could not be explained solely by the application of radiotherapy or chemotherapy in the 90 days before angioplasty — two factors that cause arterial inflammation.
Intensifying Prevention Measures
Two independent experts told this news organization that the new study is "interesting" and reinforces the close connection between oncologic and cardiovascular pathology.
Andrés Daniele, MD, cardiologist and president of the Argentine Cardio-Oncology Association, a local chapter of the International Cardio-Oncology Society, emphasized that the study “reiterates an observation seen in other works: A higher rate of atherosclerotic pathology and cardiovascular events in patients with a history of cancer. And that has a reason to be: Both pathologies present common risk factors, and on the other hand, there is greater endothelial dysfunction secondary to the inflammatory syndrome and oncologic therapies.”
“There needs to be a continuum in the intensification of measures in primary and secondary cardiovascular prevention in cancer survivors, whether in remission or with chronic disease. We need to be very aggressive in managing risk factors and insist that patients who have had a cardiovascular event enter cardiovascular rehabilitation therapies,” said Dr. Daniele, who also heads the Cardio-Oncology Department at the centenary Roffo Institute of Oncology at the University of Buenos Aires, Argentina.
The study provides a valuable contribution because “we need to understand the epidemiology and natural history of patients with cancer at risk of developing cardiovascular complications to implement personalized cardiovascular prevention strategies,” said Teresa López Fernández, MD, cardiologist, coordinator of the Cardio-Oncology Program at La Paz University Hospital in Madrid, member of the Cardio-Oncology Working Group of the Spanish Society of Cardiology, member of the board of the International Cardio-Oncology Society, and cochair of the first clinical practice guidelines in cardio-oncology of the European Society of Cardiology.
“We have to be aware that perhaps we should not guide ourselves in these patients with the usual risk stratification scores as cancer or cardiotoxic treatment are not included as variables. However, they require our attention and effort to improve their quality and quantity of life, avoiding potentially preventable cardiovascular events that could negatively impact the survival achieved thanks to advances in cancer treatments,” said Dr. López Fernández.
Dr. Melchiori and Dr. Daniele declared no relevant economic conflicts of interest. Dr. López Fernández reported relationships with Daiichi Sankyo, Almirall España, Janssen-Cilag, Bayer, Roche, Philips, and Incyte.
This article was translated from the Medscape Spanish edition. A version of this article appeared on Medscape.com.
A history of cancer is an independent predictor of major cardiovascular events in patients undergoing coronary angioplasty. Cancer should be considered a new cardiovascular risk factor in primary and secondary prevention, according to a study presented at the 2023 American Heart Association Congress in Philadelphia.
, for example, equating it to the situation of a patient with diabetes or chronic renal failure,” said lead author Renzo Melchiori, MD, a cardiologist at the University Hospital Austral in Pilar, Argentina.
The researchers also advocate for intensifying cardiovascular control measures in secondary prevention for these patients, reconsidering goals, and ensuring compliance with prescribed pharmacological regimens and healthy lifestyle habits.
“Previously, when a patient had oncological pathology, thinking about associated cardiovascular risk seemed somewhat superfluous. But today, oncological diseases are treated so effectively, increasing survival and life expectancy, that we begin to focus on what happens with the arteries of these patients after treatment,” said Dr. Melchiori.
Higher Incidence Density
The retrospective analysis included 937 patients of both sexes aged 18 years and older who underwent coronary angioplasty for acute coronary syndrome between 2008 and 2022 at a university hospital. Of these participants, 89 (9.5%) had a history of cancer, with a median time since oncologic diagnosis of around 2 years for solid and hematologic tumors. Most participants had treated and resolved cancer.
Over a median follow-up of 45 months (range, 14-72 months), the cumulative incidence rates of a major cardiovascular event (nonfatal stroke, nonfatal acute myocardial infarction, cardiovascular death, or new angioplasty) were 22.2% (155/698) and 28.4% (25/88) in the groups without and with a history of cancer, respectively. The incidence density was significantly higher in the group with an oncologic history than in the group without such a history: 0.78 events/100 patients/month vs 0.48 events/100 patients/month (P = .01).
Kaplan-Meier analysis showed a higher probability of a major cardiovascular event in the group of patients with cancer or a history of cancer (P = .0086). In multivariate Cox regression analysis, cancer history was an independent predictor of major cardiovascular events adjusted for other risk factors such as age, hypertension, diabetes, smoking, sedentary lifestyle, and family history (hazard ratio, 1.66; P = .025).
Dr. Melchiori clarified that the increased incidence of cardiovascular events in patients with cancer or a history of cancer cannot be attributed to differences in percutaneous intervention or the indication or compliance of post-treatment pharmacological therapy.
In addition, the specialist acknowledged that due to the sample size, discrimination by cancer type, disease stage, or therapeutic strategies couldn’t be performed. A subanalysis, which has not been presented, indicated that the effect could not be explained solely by the application of radiotherapy or chemotherapy in the 90 days before angioplasty — two factors that cause arterial inflammation.
Intensifying Prevention Measures
Two independent experts told this news organization that the new study is "interesting" and reinforces the close connection between oncologic and cardiovascular pathology.
Andrés Daniele, MD, cardiologist and president of the Argentine Cardio-Oncology Association, a local chapter of the International Cardio-Oncology Society, emphasized that the study “reiterates an observation seen in other works: A higher rate of atherosclerotic pathology and cardiovascular events in patients with a history of cancer. And that has a reason to be: Both pathologies present common risk factors, and on the other hand, there is greater endothelial dysfunction secondary to the inflammatory syndrome and oncologic therapies.”
“There needs to be a continuum in the intensification of measures in primary and secondary cardiovascular prevention in cancer survivors, whether in remission or with chronic disease. We need to be very aggressive in managing risk factors and insist that patients who have had a cardiovascular event enter cardiovascular rehabilitation therapies,” said Dr. Daniele, who also heads the Cardio-Oncology Department at the centenary Roffo Institute of Oncology at the University of Buenos Aires, Argentina.
The study provides a valuable contribution because “we need to understand the epidemiology and natural history of patients with cancer at risk of developing cardiovascular complications to implement personalized cardiovascular prevention strategies,” said Teresa López Fernández, MD, cardiologist, coordinator of the Cardio-Oncology Program at La Paz University Hospital in Madrid, member of the Cardio-Oncology Working Group of the Spanish Society of Cardiology, member of the board of the International Cardio-Oncology Society, and cochair of the first clinical practice guidelines in cardio-oncology of the European Society of Cardiology.
“We have to be aware that perhaps we should not guide ourselves in these patients with the usual risk stratification scores as cancer or cardiotoxic treatment are not included as variables. However, they require our attention and effort to improve their quality and quantity of life, avoiding potentially preventable cardiovascular events that could negatively impact the survival achieved thanks to advances in cancer treatments,” said Dr. López Fernández.
Dr. Melchiori and Dr. Daniele declared no relevant economic conflicts of interest. Dr. López Fernández reported relationships with Daiichi Sankyo, Almirall España, Janssen-Cilag, Bayer, Roche, Philips, and Incyte.
This article was translated from the Medscape Spanish edition. A version of this article appeared on Medscape.com.
A New Treatment Target for PTSD?
Adults with posttraumatic stress disorder (PTSD) have smaller cerebellums than unaffected adults, suggesting that this part of the brain may be a potential therapeutic target.
According to recent research on more than 4000 adults, cerebellum volume was significantly smaller (by about 2%) in those with PTSD than in trauma-exposed and trauma-naive controls without PTSD.
“The differences were largely within the posterior lobe, where a lot of the more cognitive functions attributed to the cerebellum seem to localize, as well as the vermis, which is linked to a lot of emotional processing functions,” lead author Ashley Huggins, PhD, said in a news release.
“If we know what areas are implicated, then we can start to focus interventions like brain stimulation on the cerebellum and potentially improve treatment outcomes,” said Dr. Huggins, who worked on the study while a postdoctoral researcher in the lab of Rajendra A. Morey, MD, at Duke University, Durham, North Carolina, and is now at the University of Arizona, Tucson.
While the cerebellum is known for its role in coordinating movement and balance, it also plays a key role in emotions and memory, which are affected by PTSD.
Smaller cerebellar volume has been observed in some adult and pediatric populations with PTSD.
However, those studies have been limited by either small sample sizes, the failure to consider key neuroanatomical subdivisions of the cerebellum, or a focus on certain populations such as veterans of sexual assault victims with PTSD.
To overcome these limitations, the researchers conducted a mega-analysis of total and subregional cerebellar volumes in a large, multicohort dataset from the Enhancing NeuroImaging Genetics through Meta-Analysis (ENIGMA)-Psychiatric Genomics Consortium PTSD workgroup that was published online on January 10, 2024, in Molecular Psychiatry.
They employed a novel, standardized ENIGMA cerebellum parcellation protocol to quantify cerebellar lobule volumes using structural MRI data from 1642 adults with PTSD and 2573 healthy controls without PTSD (88% trauma-exposed and 12% trauma-naive).
After adjustment for age, gender, and total intracranial volume, PTSD was associated with significant gray and white matter reductions of the cerebellum.
People with PTSD demonstrated smaller total cerebellum volume as well as reduced volume in subregions primarily within the posterior cerebellum, vermis, and flocculonodular cerebellum than controls.
In general, PTSD severity was more robustly associated with cerebellar volume differences than PTSD diagnosis.
Focusing purely on a “yes-or-no” categorical diagnosis didn’t always provide the clearest picture. “When we looked at PTSD severity, people who had more severe forms of the disorder had an even smaller cerebellar volume,” Dr. Huggins explained in the news release.
Novel Treatment Target
These findings add to “an emerging literature that underscores the relevance of cerebellar structure in the pathophysiology of PTSD,” the researchers noted.
They caution that despite the significant findings suggesting associations between PTSD and smaller cerebellar volumes, effect sizes were small. “As such, it is unlikely that structural cerebellar volumes alone will provide a clinically useful biomarker (eg, for individual-level prediction).”
Nonetheless, the study highlights the cerebellum as a “novel treatment target that may be leveraged to improve treatment outcomes for PTSD,” they wrote.
They noted that prior work has shown that the cerebellum is sensitive to external modulation. For example, noninvasive brain stimulation of the cerebellum has been shown to modulate cognitive, emotional, and social processes commonly disrupted in PTSD.
Commenting on this research, Cyrus A. Raji, MD, PhD, associate professor of radiology and neurology at Washington University in St. Louis, noted that this “large neuroimaging study links PTSD to cerebellar volume loss.”
“However, PTSD and traumatic brain injury frequently co-occur, and PTSD also frequently arises after TBI. Additionally, TBI is strongly linked to cerebellar volume loss,” Dr. Raji pointed out.
“Future studies need to better delineate volume loss from these conditions, especially when they are comorbid, though the expectation is these effects would be additive with TBI being the initial and most severe driving force,” Dr. Raji added.
The research had no commercial funding. Author disclosures are listed with the original article. Dr. Raji is a consultant for Brainreader, Apollo Health, Pacific Neuroscience Foundation, and Neurevolution Medicine LLC.
A version of this article appears on Medscape.com.
Adults with posttraumatic stress disorder (PTSD) have smaller cerebellums than unaffected adults, suggesting that this part of the brain may be a potential therapeutic target.
According to recent research on more than 4000 adults, cerebellum volume was significantly smaller (by about 2%) in those with PTSD than in trauma-exposed and trauma-naive controls without PTSD.
“The differences were largely within the posterior lobe, where a lot of the more cognitive functions attributed to the cerebellum seem to localize, as well as the vermis, which is linked to a lot of emotional processing functions,” lead author Ashley Huggins, PhD, said in a news release.
“If we know what areas are implicated, then we can start to focus interventions like brain stimulation on the cerebellum and potentially improve treatment outcomes,” said Dr. Huggins, who worked on the study while a postdoctoral researcher in the lab of Rajendra A. Morey, MD, at Duke University, Durham, North Carolina, and is now at the University of Arizona, Tucson.
While the cerebellum is known for its role in coordinating movement and balance, it also plays a key role in emotions and memory, which are affected by PTSD.
Smaller cerebellar volume has been observed in some adult and pediatric populations with PTSD.
However, those studies have been limited by either small sample sizes, the failure to consider key neuroanatomical subdivisions of the cerebellum, or a focus on certain populations such as veterans of sexual assault victims with PTSD.
To overcome these limitations, the researchers conducted a mega-analysis of total and subregional cerebellar volumes in a large, multicohort dataset from the Enhancing NeuroImaging Genetics through Meta-Analysis (ENIGMA)-Psychiatric Genomics Consortium PTSD workgroup that was published online on January 10, 2024, in Molecular Psychiatry.
They employed a novel, standardized ENIGMA cerebellum parcellation protocol to quantify cerebellar lobule volumes using structural MRI data from 1642 adults with PTSD and 2573 healthy controls without PTSD (88% trauma-exposed and 12% trauma-naive).
After adjustment for age, gender, and total intracranial volume, PTSD was associated with significant gray and white matter reductions of the cerebellum.
People with PTSD demonstrated smaller total cerebellum volume as well as reduced volume in subregions primarily within the posterior cerebellum, vermis, and flocculonodular cerebellum than controls.
In general, PTSD severity was more robustly associated with cerebellar volume differences than PTSD diagnosis.
Focusing purely on a “yes-or-no” categorical diagnosis didn’t always provide the clearest picture. “When we looked at PTSD severity, people who had more severe forms of the disorder had an even smaller cerebellar volume,” Dr. Huggins explained in the news release.
Novel Treatment Target
These findings add to “an emerging literature that underscores the relevance of cerebellar structure in the pathophysiology of PTSD,” the researchers noted.
They caution that despite the significant findings suggesting associations between PTSD and smaller cerebellar volumes, effect sizes were small. “As such, it is unlikely that structural cerebellar volumes alone will provide a clinically useful biomarker (eg, for individual-level prediction).”
Nonetheless, the study highlights the cerebellum as a “novel treatment target that may be leveraged to improve treatment outcomes for PTSD,” they wrote.
They noted that prior work has shown that the cerebellum is sensitive to external modulation. For example, noninvasive brain stimulation of the cerebellum has been shown to modulate cognitive, emotional, and social processes commonly disrupted in PTSD.
Commenting on this research, Cyrus A. Raji, MD, PhD, associate professor of radiology and neurology at Washington University in St. Louis, noted that this “large neuroimaging study links PTSD to cerebellar volume loss.”
“However, PTSD and traumatic brain injury frequently co-occur, and PTSD also frequently arises after TBI. Additionally, TBI is strongly linked to cerebellar volume loss,” Dr. Raji pointed out.
“Future studies need to better delineate volume loss from these conditions, especially when they are comorbid, though the expectation is these effects would be additive with TBI being the initial and most severe driving force,” Dr. Raji added.
The research had no commercial funding. Author disclosures are listed with the original article. Dr. Raji is a consultant for Brainreader, Apollo Health, Pacific Neuroscience Foundation, and Neurevolution Medicine LLC.
A version of this article appears on Medscape.com.
Adults with posttraumatic stress disorder (PTSD) have smaller cerebellums than unaffected adults, suggesting that this part of the brain may be a potential therapeutic target.
According to recent research on more than 4000 adults, cerebellum volume was significantly smaller (by about 2%) in those with PTSD than in trauma-exposed and trauma-naive controls without PTSD.
“The differences were largely within the posterior lobe, where a lot of the more cognitive functions attributed to the cerebellum seem to localize, as well as the vermis, which is linked to a lot of emotional processing functions,” lead author Ashley Huggins, PhD, said in a news release.
“If we know what areas are implicated, then we can start to focus interventions like brain stimulation on the cerebellum and potentially improve treatment outcomes,” said Dr. Huggins, who worked on the study while a postdoctoral researcher in the lab of Rajendra A. Morey, MD, at Duke University, Durham, North Carolina, and is now at the University of Arizona, Tucson.
While the cerebellum is known for its role in coordinating movement and balance, it also plays a key role in emotions and memory, which are affected by PTSD.
Smaller cerebellar volume has been observed in some adult and pediatric populations with PTSD.
However, those studies have been limited by either small sample sizes, the failure to consider key neuroanatomical subdivisions of the cerebellum, or a focus on certain populations such as veterans of sexual assault victims with PTSD.
To overcome these limitations, the researchers conducted a mega-analysis of total and subregional cerebellar volumes in a large, multicohort dataset from the Enhancing NeuroImaging Genetics through Meta-Analysis (ENIGMA)-Psychiatric Genomics Consortium PTSD workgroup that was published online on January 10, 2024, in Molecular Psychiatry.
They employed a novel, standardized ENIGMA cerebellum parcellation protocol to quantify cerebellar lobule volumes using structural MRI data from 1642 adults with PTSD and 2573 healthy controls without PTSD (88% trauma-exposed and 12% trauma-naive).
After adjustment for age, gender, and total intracranial volume, PTSD was associated with significant gray and white matter reductions of the cerebellum.
People with PTSD demonstrated smaller total cerebellum volume as well as reduced volume in subregions primarily within the posterior cerebellum, vermis, and flocculonodular cerebellum than controls.
In general, PTSD severity was more robustly associated with cerebellar volume differences than PTSD diagnosis.
Focusing purely on a “yes-or-no” categorical diagnosis didn’t always provide the clearest picture. “When we looked at PTSD severity, people who had more severe forms of the disorder had an even smaller cerebellar volume,” Dr. Huggins explained in the news release.
Novel Treatment Target
These findings add to “an emerging literature that underscores the relevance of cerebellar structure in the pathophysiology of PTSD,” the researchers noted.
They caution that despite the significant findings suggesting associations between PTSD and smaller cerebellar volumes, effect sizes were small. “As such, it is unlikely that structural cerebellar volumes alone will provide a clinically useful biomarker (eg, for individual-level prediction).”
Nonetheless, the study highlights the cerebellum as a “novel treatment target that may be leveraged to improve treatment outcomes for PTSD,” they wrote.
They noted that prior work has shown that the cerebellum is sensitive to external modulation. For example, noninvasive brain stimulation of the cerebellum has been shown to modulate cognitive, emotional, and social processes commonly disrupted in PTSD.
Commenting on this research, Cyrus A. Raji, MD, PhD, associate professor of radiology and neurology at Washington University in St. Louis, noted that this “large neuroimaging study links PTSD to cerebellar volume loss.”
“However, PTSD and traumatic brain injury frequently co-occur, and PTSD also frequently arises after TBI. Additionally, TBI is strongly linked to cerebellar volume loss,” Dr. Raji pointed out.
“Future studies need to better delineate volume loss from these conditions, especially when they are comorbid, though the expectation is these effects would be additive with TBI being the initial and most severe driving force,” Dr. Raji added.
The research had no commercial funding. Author disclosures are listed with the original article. Dr. Raji is a consultant for Brainreader, Apollo Health, Pacific Neuroscience Foundation, and Neurevolution Medicine LLC.
A version of this article appears on Medscape.com.
Bladder Cancer: Is Active Surveillance the Way Forward?
PARIS — Should clinicians promote active surveillance for non–muscle-invasive bladder tumors (NMIBT) and establish it as a comprehensive management approach, as with prostate and kidney cancers?
During the 117th congress of the French Association of Urology (AFU), Benjamin Pradère, MD, urologic surgeon at Croix du Sud Clinic in Quint-Fonsegrives, France, advocated for this approach, suggesting that the use of biomarkers could enhance its effectiveness.
However, it requires careful patient selection, proper information, and relevant follow-up,” said Dr. Pradère, who is a member of the AFU Cancer Committee (CCAFU).
Low-Grade Tumors
NMIBTs are precancerous lesions and constitute 70%-80% of diagnosed bladder tumors. The remaining tumors are more severe invasive tumors that infiltrate deep tissues. NMIBTs, however, entail a high risk for recurrence (reaching 80% after endoscopic resection), as well as a high risk for progression.
As a result, the diagnosis of NMIBT involves follow-up that significantly impacts patients’ quality of life due to repeated cystoscopies and endovesical treatments. “Tumors with the most impact are low-grade Ta tumors”, with longer-term monitoring required for these low-risk tumors.
Hematuria is the most frequent clinical sign. NMIBT diagnosis occurs after endoscopic tumor resection via transurethral resection, followed by an anatomopathological analysis to determine cell grade and tumor stage. Treatment depends on the risk of recurrence and progression, as well as the risk of therapeutic failure after the initial resection.
Risk stratification distinguishes the following four levels:
- Low-risk tumors: Low-grade pTa urothelial tumors, unifocal, < 3 cm, no history of bladder tumors. Low risk of recurrence and progression.
- Intermediate-risk tumors: Other low-grade pTa urothelial tumors with no high-risk criteria. Low risk of progression but high risk of recurrence.
- High-risk tumors: Tumors with at least one risk factor: Stage pT1, high grade, presence of carcinoma in situ. High risks of progression and recurrence.
- Very high-risk tumors: Tumors combining all risk factors (pT1 grade with carcinoma in situ). Very high and early risk of progression.
“We know that low-grade NMIBTs have no impact on survival,” said Dr. Pradère. For these tumors, which represent 60% of diagnosed NMIBTs, or approximately 250,000 new cases annually in France, specific survival is > 99%, meaning that most diagnosed patients will not die of bladder cancer.
The recurrence rate for low-grade tumors is 50%, but recurrences are “almost always low-grade and rarely invade the basement membrane,” said Pradère. Implementing active surveillance to limit surgical intervention to more advanced forms seems to be relevant for these tumors.
Cystoscopy Every 3 Months
According to CCAFU recommendations, “active surveillance is a therapeutic alternative that can be proposed for patients with recurrent low-risk NMIBT after the initial diagnosis.” Criteria include low-grade pTa, fewer than five tumors, size ≤ 15 mm, negative urinary cytology, asymptomatic nature, and the patient’s acceptance of closer monitoring.
While active surveillance has become the standard treatment for low-risk prostate cancer, this therapeutic option remains marginal in bladder cancer, as in kidney cancer. The goal is to defer or avoid surgical treatment by closely monitoring the natural progression of the disease.
For NMIBTs, follow-up modalities are not yet specifically recommended because of a lack of data, said Dr. Pradère. According to a consensus, cystoscopy should be repeated every 3 months for a year and then every 6 months. Unlike standard follow-up, it includes cytology “to not miss the transition to high grade.”
CCAFU recommends discontinuing active surveillance if any of the following criteria are present:
- More than 10 lesions
- Size > 30 mm
- Positive cytology
- Symptoms (hematuria, micturition disorders, and recurring infections).
Literature on the benefits of active surveillance in bladder tumors is still limited. Only seven studies are available. Overall, for nearly 600 included patients, tumors progressed in about 12% of cases. Progression to invasive tumors occurred in 0.8% of patients (n = 5).
13 Months’ Surveillance
According to a long-term study (median follow-up of 38 months), patients mostly exit active surveillance in the first year. The median duration of active surveillance is 13 months. Active surveillance is discontinued to surgically treat tumors that turn out to be low-grade Ta tumors in 70% of cases.
The following factors predicting recurrence and progression of tumors have been identified: Multiple tumors, early recurrence (within a year of initial diagnosis), frequent recurrence (more than one recurrence per year), tumors > 3 cm, and failure of previous endovesical treatment.
Recent studies have shown that with at least three of these recurrence and progression factors, the median duration under active surveillance is 15 months compared with 28 months in the absence of such factors. “Considering these factors, it is possible to assess the benefit of active surveillance for the patient,” said Dr. Pradère.
If active surveillance for bladder tumors is still not widely practiced, then the contribution of imaging (MRI and ultrasound) and biomarkers could promote its adoption. “The use of biomarkers should change the game and encourage active surveillance in patients with small polyps,” said Dr. Pradère.
ADXBladder Test Utility
A study highlighted the importance of evaluating minichromosome maintenance protein 5 expression during active surveillance using the ADXBladder ELISA test on a urine sample. This test is usually used in bladder cancer diagnosis.
“This study showed that a negative result in two consecutive tests during active surveillance is associated with an almost zero recurrence risk. After two negative tests, most patients do not exit active surveillance,” said Dr. Pradère. But the positive predictive value of biomarkers remains low for low-grade tumors.
The future of active surveillance in bladder cancer should involve better patient selection that relies on risk factors, enhanced modalities through imaging and biomarkers, and the advent of artificial intelligence to analyze cystoscopy results, concluded Dr. Pradère.
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
PARIS — Should clinicians promote active surveillance for non–muscle-invasive bladder tumors (NMIBT) and establish it as a comprehensive management approach, as with prostate and kidney cancers?
During the 117th congress of the French Association of Urology (AFU), Benjamin Pradère, MD, urologic surgeon at Croix du Sud Clinic in Quint-Fonsegrives, France, advocated for this approach, suggesting that the use of biomarkers could enhance its effectiveness.
However, it requires careful patient selection, proper information, and relevant follow-up,” said Dr. Pradère, who is a member of the AFU Cancer Committee (CCAFU).
Low-Grade Tumors
NMIBTs are precancerous lesions and constitute 70%-80% of diagnosed bladder tumors. The remaining tumors are more severe invasive tumors that infiltrate deep tissues. NMIBTs, however, entail a high risk for recurrence (reaching 80% after endoscopic resection), as well as a high risk for progression.
As a result, the diagnosis of NMIBT involves follow-up that significantly impacts patients’ quality of life due to repeated cystoscopies and endovesical treatments. “Tumors with the most impact are low-grade Ta tumors”, with longer-term monitoring required for these low-risk tumors.
Hematuria is the most frequent clinical sign. NMIBT diagnosis occurs after endoscopic tumor resection via transurethral resection, followed by an anatomopathological analysis to determine cell grade and tumor stage. Treatment depends on the risk of recurrence and progression, as well as the risk of therapeutic failure after the initial resection.
Risk stratification distinguishes the following four levels:
- Low-risk tumors: Low-grade pTa urothelial tumors, unifocal, < 3 cm, no history of bladder tumors. Low risk of recurrence and progression.
- Intermediate-risk tumors: Other low-grade pTa urothelial tumors with no high-risk criteria. Low risk of progression but high risk of recurrence.
- High-risk tumors: Tumors with at least one risk factor: Stage pT1, high grade, presence of carcinoma in situ. High risks of progression and recurrence.
- Very high-risk tumors: Tumors combining all risk factors (pT1 grade with carcinoma in situ). Very high and early risk of progression.
“We know that low-grade NMIBTs have no impact on survival,” said Dr. Pradère. For these tumors, which represent 60% of diagnosed NMIBTs, or approximately 250,000 new cases annually in France, specific survival is > 99%, meaning that most diagnosed patients will not die of bladder cancer.
The recurrence rate for low-grade tumors is 50%, but recurrences are “almost always low-grade and rarely invade the basement membrane,” said Pradère. Implementing active surveillance to limit surgical intervention to more advanced forms seems to be relevant for these tumors.
Cystoscopy Every 3 Months
According to CCAFU recommendations, “active surveillance is a therapeutic alternative that can be proposed for patients with recurrent low-risk NMIBT after the initial diagnosis.” Criteria include low-grade pTa, fewer than five tumors, size ≤ 15 mm, negative urinary cytology, asymptomatic nature, and the patient’s acceptance of closer monitoring.
While active surveillance has become the standard treatment for low-risk prostate cancer, this therapeutic option remains marginal in bladder cancer, as in kidney cancer. The goal is to defer or avoid surgical treatment by closely monitoring the natural progression of the disease.
For NMIBTs, follow-up modalities are not yet specifically recommended because of a lack of data, said Dr. Pradère. According to a consensus, cystoscopy should be repeated every 3 months for a year and then every 6 months. Unlike standard follow-up, it includes cytology “to not miss the transition to high grade.”
CCAFU recommends discontinuing active surveillance if any of the following criteria are present:
- More than 10 lesions
- Size > 30 mm
- Positive cytology
- Symptoms (hematuria, micturition disorders, and recurring infections).
Literature on the benefits of active surveillance in bladder tumors is still limited. Only seven studies are available. Overall, for nearly 600 included patients, tumors progressed in about 12% of cases. Progression to invasive tumors occurred in 0.8% of patients (n = 5).
13 Months’ Surveillance
According to a long-term study (median follow-up of 38 months), patients mostly exit active surveillance in the first year. The median duration of active surveillance is 13 months. Active surveillance is discontinued to surgically treat tumors that turn out to be low-grade Ta tumors in 70% of cases.
The following factors predicting recurrence and progression of tumors have been identified: Multiple tumors, early recurrence (within a year of initial diagnosis), frequent recurrence (more than one recurrence per year), tumors > 3 cm, and failure of previous endovesical treatment.
Recent studies have shown that with at least three of these recurrence and progression factors, the median duration under active surveillance is 15 months compared with 28 months in the absence of such factors. “Considering these factors, it is possible to assess the benefit of active surveillance for the patient,” said Dr. Pradère.
If active surveillance for bladder tumors is still not widely practiced, then the contribution of imaging (MRI and ultrasound) and biomarkers could promote its adoption. “The use of biomarkers should change the game and encourage active surveillance in patients with small polyps,” said Dr. Pradère.
ADXBladder Test Utility
A study highlighted the importance of evaluating minichromosome maintenance protein 5 expression during active surveillance using the ADXBladder ELISA test on a urine sample. This test is usually used in bladder cancer diagnosis.
“This study showed that a negative result in two consecutive tests during active surveillance is associated with an almost zero recurrence risk. After two negative tests, most patients do not exit active surveillance,” said Dr. Pradère. But the positive predictive value of biomarkers remains low for low-grade tumors.
The future of active surveillance in bladder cancer should involve better patient selection that relies on risk factors, enhanced modalities through imaging and biomarkers, and the advent of artificial intelligence to analyze cystoscopy results, concluded Dr. Pradère.
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
PARIS — Should clinicians promote active surveillance for non–muscle-invasive bladder tumors (NMIBT) and establish it as a comprehensive management approach, as with prostate and kidney cancers?
During the 117th congress of the French Association of Urology (AFU), Benjamin Pradère, MD, urologic surgeon at Croix du Sud Clinic in Quint-Fonsegrives, France, advocated for this approach, suggesting that the use of biomarkers could enhance its effectiveness.
However, it requires careful patient selection, proper information, and relevant follow-up,” said Dr. Pradère, who is a member of the AFU Cancer Committee (CCAFU).
Low-Grade Tumors
NMIBTs are precancerous lesions and constitute 70%-80% of diagnosed bladder tumors. The remaining tumors are more severe invasive tumors that infiltrate deep tissues. NMIBTs, however, entail a high risk for recurrence (reaching 80% after endoscopic resection), as well as a high risk for progression.
As a result, the diagnosis of NMIBT involves follow-up that significantly impacts patients’ quality of life due to repeated cystoscopies and endovesical treatments. “Tumors with the most impact are low-grade Ta tumors”, with longer-term monitoring required for these low-risk tumors.
Hematuria is the most frequent clinical sign. NMIBT diagnosis occurs after endoscopic tumor resection via transurethral resection, followed by an anatomopathological analysis to determine cell grade and tumor stage. Treatment depends on the risk of recurrence and progression, as well as the risk of therapeutic failure after the initial resection.
Risk stratification distinguishes the following four levels:
- Low-risk tumors: Low-grade pTa urothelial tumors, unifocal, < 3 cm, no history of bladder tumors. Low risk of recurrence and progression.
- Intermediate-risk tumors: Other low-grade pTa urothelial tumors with no high-risk criteria. Low risk of progression but high risk of recurrence.
- High-risk tumors: Tumors with at least one risk factor: Stage pT1, high grade, presence of carcinoma in situ. High risks of progression and recurrence.
- Very high-risk tumors: Tumors combining all risk factors (pT1 grade with carcinoma in situ). Very high and early risk of progression.
“We know that low-grade NMIBTs have no impact on survival,” said Dr. Pradère. For these tumors, which represent 60% of diagnosed NMIBTs, or approximately 250,000 new cases annually in France, specific survival is > 99%, meaning that most diagnosed patients will not die of bladder cancer.
The recurrence rate for low-grade tumors is 50%, but recurrences are “almost always low-grade and rarely invade the basement membrane,” said Pradère. Implementing active surveillance to limit surgical intervention to more advanced forms seems to be relevant for these tumors.
Cystoscopy Every 3 Months
According to CCAFU recommendations, “active surveillance is a therapeutic alternative that can be proposed for patients with recurrent low-risk NMIBT after the initial diagnosis.” Criteria include low-grade pTa, fewer than five tumors, size ≤ 15 mm, negative urinary cytology, asymptomatic nature, and the patient’s acceptance of closer monitoring.
While active surveillance has become the standard treatment for low-risk prostate cancer, this therapeutic option remains marginal in bladder cancer, as in kidney cancer. The goal is to defer or avoid surgical treatment by closely monitoring the natural progression of the disease.
For NMIBTs, follow-up modalities are not yet specifically recommended because of a lack of data, said Dr. Pradère. According to a consensus, cystoscopy should be repeated every 3 months for a year and then every 6 months. Unlike standard follow-up, it includes cytology “to not miss the transition to high grade.”
CCAFU recommends discontinuing active surveillance if any of the following criteria are present:
- More than 10 lesions
- Size > 30 mm
- Positive cytology
- Symptoms (hematuria, micturition disorders, and recurring infections).
Literature on the benefits of active surveillance in bladder tumors is still limited. Only seven studies are available. Overall, for nearly 600 included patients, tumors progressed in about 12% of cases. Progression to invasive tumors occurred in 0.8% of patients (n = 5).
13 Months’ Surveillance
According to a long-term study (median follow-up of 38 months), patients mostly exit active surveillance in the first year. The median duration of active surveillance is 13 months. Active surveillance is discontinued to surgically treat tumors that turn out to be low-grade Ta tumors in 70% of cases.
The following factors predicting recurrence and progression of tumors have been identified: Multiple tumors, early recurrence (within a year of initial diagnosis), frequent recurrence (more than one recurrence per year), tumors > 3 cm, and failure of previous endovesical treatment.
Recent studies have shown that with at least three of these recurrence and progression factors, the median duration under active surveillance is 15 months compared with 28 months in the absence of such factors. “Considering these factors, it is possible to assess the benefit of active surveillance for the patient,” said Dr. Pradère.
If active surveillance for bladder tumors is still not widely practiced, then the contribution of imaging (MRI and ultrasound) and biomarkers could promote its adoption. “The use of biomarkers should change the game and encourage active surveillance in patients with small polyps,” said Dr. Pradère.
ADXBladder Test Utility
A study highlighted the importance of evaluating minichromosome maintenance protein 5 expression during active surveillance using the ADXBladder ELISA test on a urine sample. This test is usually used in bladder cancer diagnosis.
“This study showed that a negative result in two consecutive tests during active surveillance is associated with an almost zero recurrence risk. After two negative tests, most patients do not exit active surveillance,” said Dr. Pradère. But the positive predictive value of biomarkers remains low for low-grade tumors.
The future of active surveillance in bladder cancer should involve better patient selection that relies on risk factors, enhanced modalities through imaging and biomarkers, and the advent of artificial intelligence to analyze cystoscopy results, concluded Dr. Pradère.
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
Shelf Life for Opioid Overdose Drug Naloxone Extended
At the request of the US Food and Drug Administration (FDA), Emergent BioSolutions has extended the shelf life of the rapid opioid overdose reversal agent, naloxone (4 mg) nasal spray (Narcan), from 3 to 4 years.
Naloxone is “an important tool” in addressing opioid overdoses, and this extension supports the FDA’s “efforts to ensure more OTC naloxone products remain available to the public,” Marta Sokolowska, PhD, with the FDA Center for Drug Evaluation and Research, said in a statement.
Naloxone nasal spray was first approved by the FDA in 2015 as a prescription drug. Last spring, the agency approved the drug for over-the-counter use.
“The shelf life of products that were produced and distributed prior to this announcement is not affected and remains unchanged. Prescribers, patients, and caregivers are advised to continue to abide by the expiration date printed on each product’s packaging and within the product’s labeling,” the FDA advised.
“FDA’s request for this shelf-life extension is a testament to the agency’s continuing progress toward implementing the FDA Overdose Prevention Framework, which provides our vision to undertake impactful, creative actions to encourage harm reduction and innovation in reducing controlled substance-related overdoses and deaths,” the agency said.
According to the US Centers for Disease Control and Prevention, from 1999 to 2021, nearly 645,000 people died from an overdose involving any opioid, including prescription and illicit opioids.
A version of this article appeared on Medscape.com.
At the request of the US Food and Drug Administration (FDA), Emergent BioSolutions has extended the shelf life of the rapid opioid overdose reversal agent, naloxone (4 mg) nasal spray (Narcan), from 3 to 4 years.
Naloxone is “an important tool” in addressing opioid overdoses, and this extension supports the FDA’s “efforts to ensure more OTC naloxone products remain available to the public,” Marta Sokolowska, PhD, with the FDA Center for Drug Evaluation and Research, said in a statement.
Naloxone nasal spray was first approved by the FDA in 2015 as a prescription drug. Last spring, the agency approved the drug for over-the-counter use.
“The shelf life of products that were produced and distributed prior to this announcement is not affected and remains unchanged. Prescribers, patients, and caregivers are advised to continue to abide by the expiration date printed on each product’s packaging and within the product’s labeling,” the FDA advised.
“FDA’s request for this shelf-life extension is a testament to the agency’s continuing progress toward implementing the FDA Overdose Prevention Framework, which provides our vision to undertake impactful, creative actions to encourage harm reduction and innovation in reducing controlled substance-related overdoses and deaths,” the agency said.
According to the US Centers for Disease Control and Prevention, from 1999 to 2021, nearly 645,000 people died from an overdose involving any opioid, including prescription and illicit opioids.
A version of this article appeared on Medscape.com.
At the request of the US Food and Drug Administration (FDA), Emergent BioSolutions has extended the shelf life of the rapid opioid overdose reversal agent, naloxone (4 mg) nasal spray (Narcan), from 3 to 4 years.
Naloxone is “an important tool” in addressing opioid overdoses, and this extension supports the FDA’s “efforts to ensure more OTC naloxone products remain available to the public,” Marta Sokolowska, PhD, with the FDA Center for Drug Evaluation and Research, said in a statement.
Naloxone nasal spray was first approved by the FDA in 2015 as a prescription drug. Last spring, the agency approved the drug for over-the-counter use.
“The shelf life of products that were produced and distributed prior to this announcement is not affected and remains unchanged. Prescribers, patients, and caregivers are advised to continue to abide by the expiration date printed on each product’s packaging and within the product’s labeling,” the FDA advised.
“FDA’s request for this shelf-life extension is a testament to the agency’s continuing progress toward implementing the FDA Overdose Prevention Framework, which provides our vision to undertake impactful, creative actions to encourage harm reduction and innovation in reducing controlled substance-related overdoses and deaths,” the agency said.
According to the US Centers for Disease Control and Prevention, from 1999 to 2021, nearly 645,000 people died from an overdose involving any opioid, including prescription and illicit opioids.
A version of this article appeared on Medscape.com.
Even Intentional Weight Loss Linked With Cancer
This transcript has been edited for clarity.
As anyone who has been through medical training will tell you, some little scenes just stick with you. I had been seeing a patient in our resident clinic in West Philly for a couple of years. She was in her mid-60s with diabetes and hypertension and a distant smoking history. She was overweight and had been trying to improve her diet and lose weight since I started seeing her. One day she came in and was delighted to report that she had finally started shedding some pounds — about 15 in the past 2 months.
I enthusiastically told my preceptor that my careful dietary counseling had finally done the job. She looked through the chart for a moment and asked, “Is she up to date on her cancer screening?” A workup revealed adenocarcinoma of the lung. The patient did well, actually, but the story stuck with me.
The textbooks call it “unintentional weight loss,” often in big, scary letters, and every doctor will go just a bit pale if a patient tells them that, despite efforts not to, they are losing weight. But true unintentional weight loss is not that common. After all, most of us are at least half-heartedly trying to lose weight all the time. Should doctors be worried when we are successful?
A new study suggests that perhaps they should. We’re talking about this study, appearing in JAMA, which combined participants from two long-running observational cohorts: 120,000 women from the Nurses’ Health Study, and 50,000 men from the Health Professionals Follow-Up Study. (These cohorts started in the 1970s and 1980s, so we’ll give them a pass on the gender-specific study designs.)
The rationale of enrolling healthcare providers in these cohort studies is that they would be reliable witnesses of their own health status. If a nurse or doctor says they have pancreatic cancer, it’s likely that they truly have pancreatic cancer. Detailed health surveys were distributed to the participants every other year, and the average follow-up was more than a decade.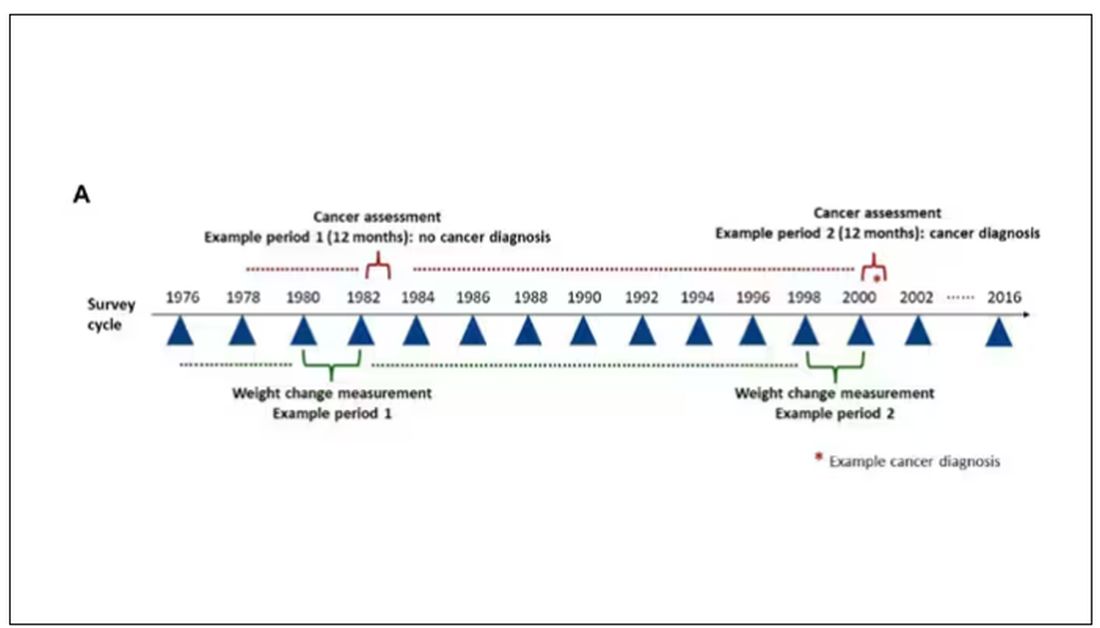
Participants recorded their weight — as an aside, a nested study found that self-reported rate was extremely well correlated with professionally measured weight — and whether they had received a cancer diagnosis since the last survey.
This allowed researchers to look at the phenomenon described above. Would weight loss precede a new diagnosis of cancer? And, more interestingly, would intentional weight loss precede a new diagnosis of cancer.
I don’t think it will surprise you to hear that individuals in the highest category of weight loss, those who lost more than 10% of their body weight over a 2-year period, had a larger risk of being diagnosed with cancer in the next year. That’s the yellow line in this graph. In fact, they had about a 40% higher risk than those who did not lose weight.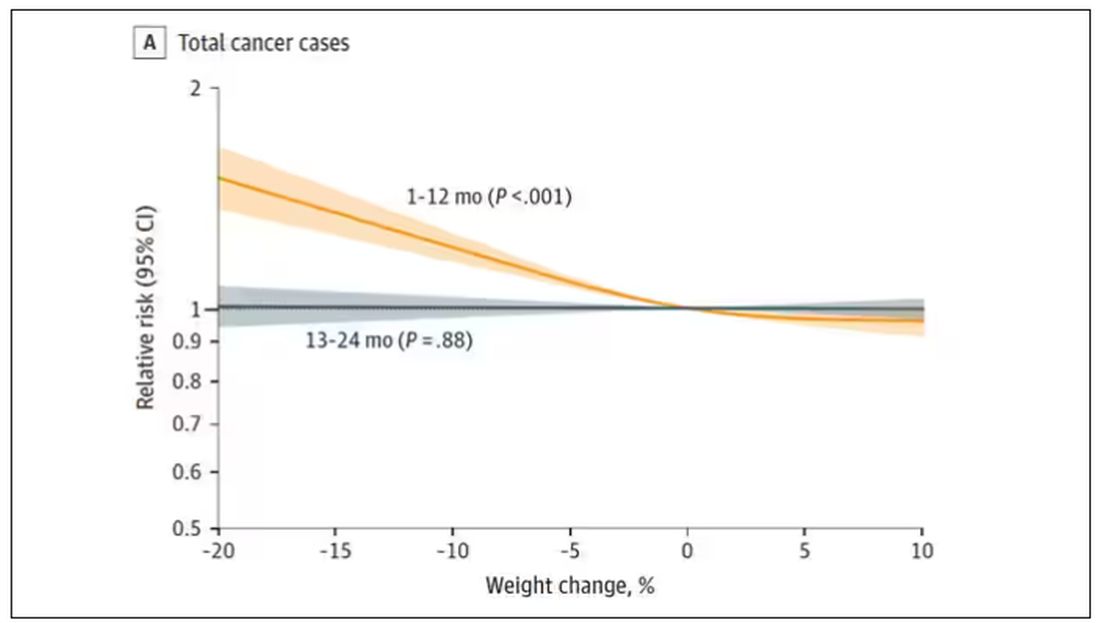
Increased risk was found across multiple cancer types, though cancers of the gastrointestinal tract, not surprisingly, were most strongly associated with antecedent weight loss.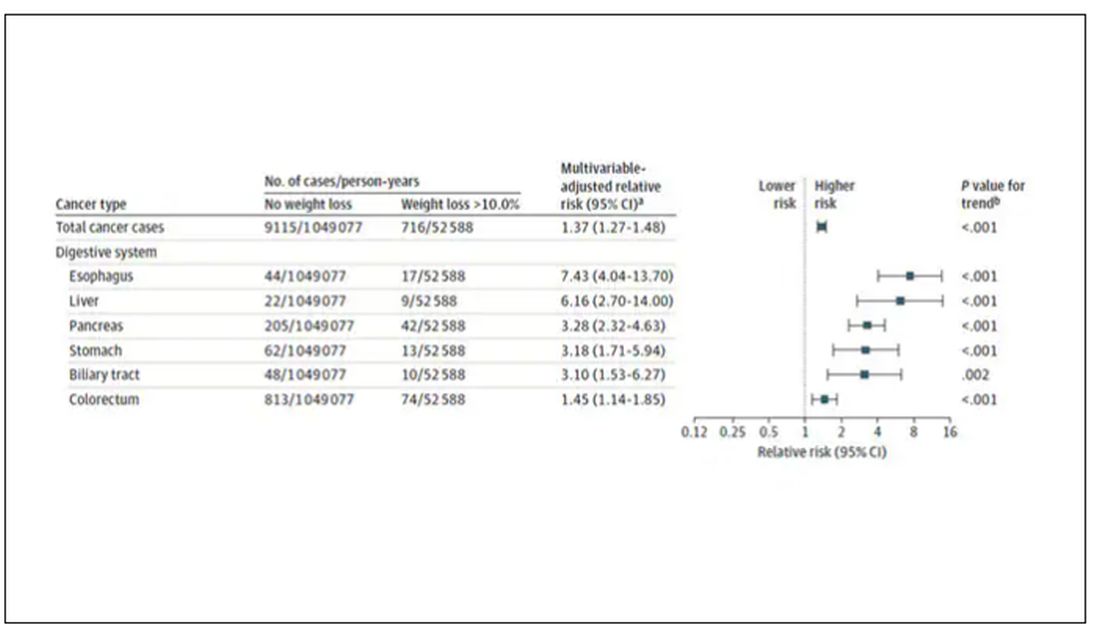
What about intentionality of weight loss? Unfortunately, the surveys did not ask participants whether they were trying to lose weight. Rather, the surveys asked about exercise and dietary habits. The researchers leveraged these responses to create three categories of participants: those who seemed to be trying to lose weight (defined as people who had increased their exercise and dietary quality); those who didn’t seem to be trying to lose weight (they changed neither exercise nor dietary behaviors); and a middle group, which changed one or the other of these behaviors but not both.
Let’s look at those who really seemed to be trying to lose weight. Over 2 years, they got more exercise and improved their diet.
If they succeeded in losing 10% or more of their body weight, they still had a higher risk for cancer than those who had not lost weight — about 30% higher, which is not that different from the 40% increased risk when you include those folks who weren’t changing their lifestyle.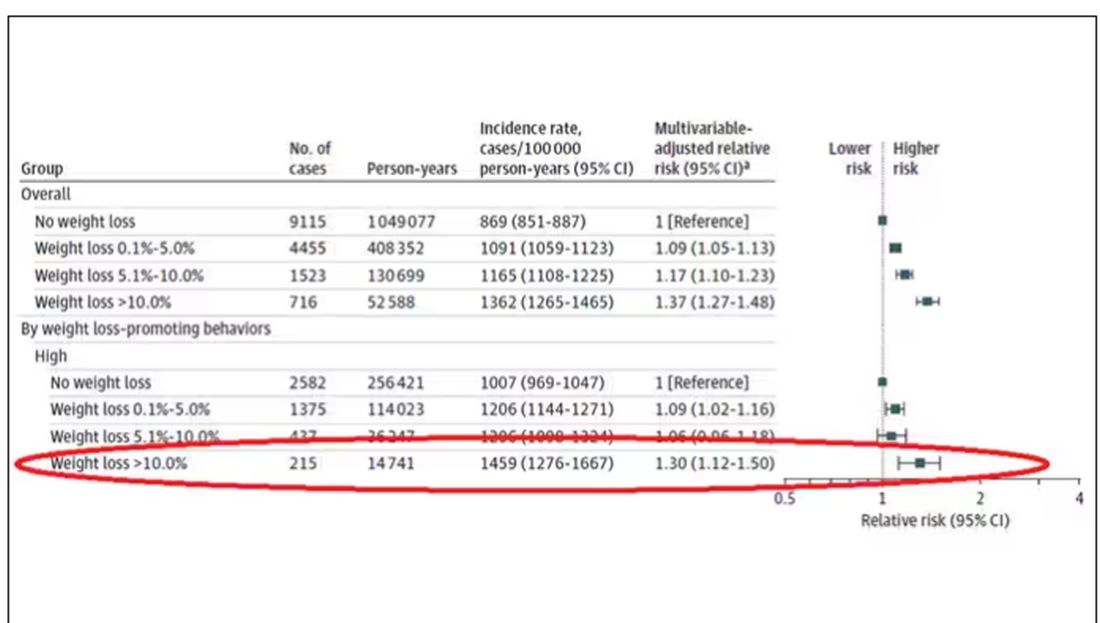
This is why this study is important. The classic teaching is that unintentional weight loss is a bad thing and needs a workup. That’s fine. But we live in a world where perhaps the majority of people are, at any given time, trying to lose weight.
We need to be careful here. I am not by any means trying to say that people who have successfully lost weight have cancer. Both of the following statements can be true:
Significant weight loss, whether intentional or not, is associated with a higher risk for cancer.
Most people with significant weight loss will not have cancer.
Both of these can be true because cancer is, fortunately, rare. Of people who lose weight, the vast majority will lose weight because they are engaging in healthier behaviors. A small number may lose weight because something else is wrong. It’s just hard to tell the two apart.
Out of the nearly 200,000 people in this study, only around 16,000 developed cancer during follow-up. Again, although the chance of having cancer is slightly higher if someone has experienced weight loss, the chance is still very low.
We also need to avoid suggesting that weight loss causes cancer. Some people lose weight because of an existing, as of yet undiagnosed cancer and its metabolic effects. This is borne out if you look at the risk of being diagnosed with cancer as you move further away from the interval of weight loss.
The further you get from the year of that 10% weight loss, the less likely you are to be diagnosed with cancer. Most of these cancers are diagnosed within a year of losing weight. In other words, if you’re reading this and getting worried that you lost weight 10 years ago, you’re probably out of the woods. That was, most likely, just you getting healthier.
Last thing: We have methods for weight loss now that are way more effective than diet or exercise. I’m looking at you, Ozempic. But aside from the weight loss wonder drugs, we have surgery and other interventions. This study did not capture any of that data. Ozempic wasn’t even on the market during this study, so we can’t say anything about the relationship between weight loss and cancer among people using nonlifestyle mechanisms to lose weight.
It’s a complicated system. But the clinically actionable point here is to notice if patients have lost weight. If they’ve lost it without trying, further workup is reasonable. If they’ve lost it but were trying to lose it, tell them “good job.” And consider a workup anyway.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
As anyone who has been through medical training will tell you, some little scenes just stick with you. I had been seeing a patient in our resident clinic in West Philly for a couple of years. She was in her mid-60s with diabetes and hypertension and a distant smoking history. She was overweight and had been trying to improve her diet and lose weight since I started seeing her. One day she came in and was delighted to report that she had finally started shedding some pounds — about 15 in the past 2 months.
I enthusiastically told my preceptor that my careful dietary counseling had finally done the job. She looked through the chart for a moment and asked, “Is she up to date on her cancer screening?” A workup revealed adenocarcinoma of the lung. The patient did well, actually, but the story stuck with me.
The textbooks call it “unintentional weight loss,” often in big, scary letters, and every doctor will go just a bit pale if a patient tells them that, despite efforts not to, they are losing weight. But true unintentional weight loss is not that common. After all, most of us are at least half-heartedly trying to lose weight all the time. Should doctors be worried when we are successful?
A new study suggests that perhaps they should. We’re talking about this study, appearing in JAMA, which combined participants from two long-running observational cohorts: 120,000 women from the Nurses’ Health Study, and 50,000 men from the Health Professionals Follow-Up Study. (These cohorts started in the 1970s and 1980s, so we’ll give them a pass on the gender-specific study designs.)
The rationale of enrolling healthcare providers in these cohort studies is that they would be reliable witnesses of their own health status. If a nurse or doctor says they have pancreatic cancer, it’s likely that they truly have pancreatic cancer. Detailed health surveys were distributed to the participants every other year, and the average follow-up was more than a decade.
Participants recorded their weight — as an aside, a nested study found that self-reported rate was extremely well correlated with professionally measured weight — and whether they had received a cancer diagnosis since the last survey.
This allowed researchers to look at the phenomenon described above. Would weight loss precede a new diagnosis of cancer? And, more interestingly, would intentional weight loss precede a new diagnosis of cancer.
I don’t think it will surprise you to hear that individuals in the highest category of weight loss, those who lost more than 10% of their body weight over a 2-year period, had a larger risk of being diagnosed with cancer in the next year. That’s the yellow line in this graph. In fact, they had about a 40% higher risk than those who did not lose weight.
Increased risk was found across multiple cancer types, though cancers of the gastrointestinal tract, not surprisingly, were most strongly associated with antecedent weight loss.
What about intentionality of weight loss? Unfortunately, the surveys did not ask participants whether they were trying to lose weight. Rather, the surveys asked about exercise and dietary habits. The researchers leveraged these responses to create three categories of participants: those who seemed to be trying to lose weight (defined as people who had increased their exercise and dietary quality); those who didn’t seem to be trying to lose weight (they changed neither exercise nor dietary behaviors); and a middle group, which changed one or the other of these behaviors but not both.
Let’s look at those who really seemed to be trying to lose weight. Over 2 years, they got more exercise and improved their diet.
If they succeeded in losing 10% or more of their body weight, they still had a higher risk for cancer than those who had not lost weight — about 30% higher, which is not that different from the 40% increased risk when you include those folks who weren’t changing their lifestyle.
This is why this study is important. The classic teaching is that unintentional weight loss is a bad thing and needs a workup. That’s fine. But we live in a world where perhaps the majority of people are, at any given time, trying to lose weight.
We need to be careful here. I am not by any means trying to say that people who have successfully lost weight have cancer. Both of the following statements can be true:
Significant weight loss, whether intentional or not, is associated with a higher risk for cancer.
Most people with significant weight loss will not have cancer.
Both of these can be true because cancer is, fortunately, rare. Of people who lose weight, the vast majority will lose weight because they are engaging in healthier behaviors. A small number may lose weight because something else is wrong. It’s just hard to tell the two apart.
Out of the nearly 200,000 people in this study, only around 16,000 developed cancer during follow-up. Again, although the chance of having cancer is slightly higher if someone has experienced weight loss, the chance is still very low.
We also need to avoid suggesting that weight loss causes cancer. Some people lose weight because of an existing, as of yet undiagnosed cancer and its metabolic effects. This is borne out if you look at the risk of being diagnosed with cancer as you move further away from the interval of weight loss.
The further you get from the year of that 10% weight loss, the less likely you are to be diagnosed with cancer. Most of these cancers are diagnosed within a year of losing weight. In other words, if you’re reading this and getting worried that you lost weight 10 years ago, you’re probably out of the woods. That was, most likely, just you getting healthier.
Last thing: We have methods for weight loss now that are way more effective than diet or exercise. I’m looking at you, Ozempic. But aside from the weight loss wonder drugs, we have surgery and other interventions. This study did not capture any of that data. Ozempic wasn’t even on the market during this study, so we can’t say anything about the relationship between weight loss and cancer among people using nonlifestyle mechanisms to lose weight.
It’s a complicated system. But the clinically actionable point here is to notice if patients have lost weight. If they’ve lost it without trying, further workup is reasonable. If they’ve lost it but were trying to lose it, tell them “good job.” And consider a workup anyway.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
As anyone who has been through medical training will tell you, some little scenes just stick with you. I had been seeing a patient in our resident clinic in West Philly for a couple of years. She was in her mid-60s with diabetes and hypertension and a distant smoking history. She was overweight and had been trying to improve her diet and lose weight since I started seeing her. One day she came in and was delighted to report that she had finally started shedding some pounds — about 15 in the past 2 months.
I enthusiastically told my preceptor that my careful dietary counseling had finally done the job. She looked through the chart for a moment and asked, “Is she up to date on her cancer screening?” A workup revealed adenocarcinoma of the lung. The patient did well, actually, but the story stuck with me.
The textbooks call it “unintentional weight loss,” often in big, scary letters, and every doctor will go just a bit pale if a patient tells them that, despite efforts not to, they are losing weight. But true unintentional weight loss is not that common. After all, most of us are at least half-heartedly trying to lose weight all the time. Should doctors be worried when we are successful?
A new study suggests that perhaps they should. We’re talking about this study, appearing in JAMA, which combined participants from two long-running observational cohorts: 120,000 women from the Nurses’ Health Study, and 50,000 men from the Health Professionals Follow-Up Study. (These cohorts started in the 1970s and 1980s, so we’ll give them a pass on the gender-specific study designs.)
The rationale of enrolling healthcare providers in these cohort studies is that they would be reliable witnesses of their own health status. If a nurse or doctor says they have pancreatic cancer, it’s likely that they truly have pancreatic cancer. Detailed health surveys were distributed to the participants every other year, and the average follow-up was more than a decade.
Participants recorded their weight — as an aside, a nested study found that self-reported rate was extremely well correlated with professionally measured weight — and whether they had received a cancer diagnosis since the last survey.
This allowed researchers to look at the phenomenon described above. Would weight loss precede a new diagnosis of cancer? And, more interestingly, would intentional weight loss precede a new diagnosis of cancer.
I don’t think it will surprise you to hear that individuals in the highest category of weight loss, those who lost more than 10% of their body weight over a 2-year period, had a larger risk of being diagnosed with cancer in the next year. That’s the yellow line in this graph. In fact, they had about a 40% higher risk than those who did not lose weight.
Increased risk was found across multiple cancer types, though cancers of the gastrointestinal tract, not surprisingly, were most strongly associated with antecedent weight loss.
What about intentionality of weight loss? Unfortunately, the surveys did not ask participants whether they were trying to lose weight. Rather, the surveys asked about exercise and dietary habits. The researchers leveraged these responses to create three categories of participants: those who seemed to be trying to lose weight (defined as people who had increased their exercise and dietary quality); those who didn’t seem to be trying to lose weight (they changed neither exercise nor dietary behaviors); and a middle group, which changed one or the other of these behaviors but not both.
Let’s look at those who really seemed to be trying to lose weight. Over 2 years, they got more exercise and improved their diet.
If they succeeded in losing 10% or more of their body weight, they still had a higher risk for cancer than those who had not lost weight — about 30% higher, which is not that different from the 40% increased risk when you include those folks who weren’t changing their lifestyle.
This is why this study is important. The classic teaching is that unintentional weight loss is a bad thing and needs a workup. That’s fine. But we live in a world where perhaps the majority of people are, at any given time, trying to lose weight.
We need to be careful here. I am not by any means trying to say that people who have successfully lost weight have cancer. Both of the following statements can be true:
Significant weight loss, whether intentional or not, is associated with a higher risk for cancer.
Most people with significant weight loss will not have cancer.
Both of these can be true because cancer is, fortunately, rare. Of people who lose weight, the vast majority will lose weight because they are engaging in healthier behaviors. A small number may lose weight because something else is wrong. It’s just hard to tell the two apart.
Out of the nearly 200,000 people in this study, only around 16,000 developed cancer during follow-up. Again, although the chance of having cancer is slightly higher if someone has experienced weight loss, the chance is still very low.
We also need to avoid suggesting that weight loss causes cancer. Some people lose weight because of an existing, as of yet undiagnosed cancer and its metabolic effects. This is borne out if you look at the risk of being diagnosed with cancer as you move further away from the interval of weight loss.
The further you get from the year of that 10% weight loss, the less likely you are to be diagnosed with cancer. Most of these cancers are diagnosed within a year of losing weight. In other words, if you’re reading this and getting worried that you lost weight 10 years ago, you’re probably out of the woods. That was, most likely, just you getting healthier.
Last thing: We have methods for weight loss now that are way more effective than diet or exercise. I’m looking at you, Ozempic. But aside from the weight loss wonder drugs, we have surgery and other interventions. This study did not capture any of that data. Ozempic wasn’t even on the market during this study, so we can’t say anything about the relationship between weight loss and cancer among people using nonlifestyle mechanisms to lose weight.
It’s a complicated system. But the clinically actionable point here is to notice if patients have lost weight. If they’ve lost it without trying, further workup is reasonable. If they’ve lost it but were trying to lose it, tell them “good job.” And consider a workup anyway.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.