User login
MD-IQ only
IBS: Understanding a Common Yet Misunderstood Condition
Irritable bowel syndrome (IBS) is one of the most common conditions encountered by both primary care providers and gastroenterologists, with a pooled global prevalence of 11.2%. This functional bowel disorder is characterized by abdominal pain or discomfort, diarrhea and/or constipation, and bloating.
Unfortunately, , according to Alan Desmond, MB, consultant in gastroenterology and general internal medicine, Torbay Hospital, UK National Health Service.
Desmond regularly sees patients who either haven’t been accurately diagnosed or have been told, “Don’t worry, it’s ‘just’ irritable bowel syndrome,” he said at the recent International Conference on Nutrition in Medicine.
A 2017 study involving nearly 2000 patients with a history of gastrointestinal (GI) symptoms found that 43.1% of those who met the criteria for IBS were undiagnosed, and among those who were diagnosed, 26% were not receiving treatment.
“Many clinicians vastly underestimate the impact functional GI symptoms have on our patients in lack of productivity, becoming homebound or losing employment, the inability to enjoy a meal with friends or family, and always needing to know where the nearest bathroom is, for example,” Desmond said in an interview.
IBS can profoundly affect patients’ mental health. One study found that 38% of patients with IBS attending a tertiary care clinic contemplated suicide because they felt hopeless about ever achieving symptom relief.
Today, several dietary, pharmacologic, and psychological/behavioral approaches are available to treat patients with IBS, noted William D. Chey, MD, AGAF, chief of the Division of Gastroenterology and Hepatology, University of Michigan, Ann Arbor, Michigan.
“Each individual patient may need a different combination of these foundational treatments,” he said. “One size doesn’t fit all.”
Diagnostic Pathway
One reason IBS is so hard to diagnose is that it’s a “symptom-based disorder, with identification of the condition predicated upon certain key characteristics that are heterogeneous,” Chey said in an interview. “IBS in patient ‘A’ may not present the same way as IBS in patient ‘B,’ although there are certain foundational common characteristics.”
IBS involves “abnormalities in the motility and contractility of the GI tract,” he said. It can present with diarrhea (IBS-D), constipation (IBS-C), or a mixture or alternation of diarrhea and constipation (IBS-M).
Patients with IBS-D often have an exaggerated gastro-colonic response, while those with IBS-C often have a blunted response.
Beyond stool abnormalities and abdominal pain/discomfort, patients often report bloating/distension, low backache, lethargy, nausea, thigh pain, and urinary and gynecologic symptoms.
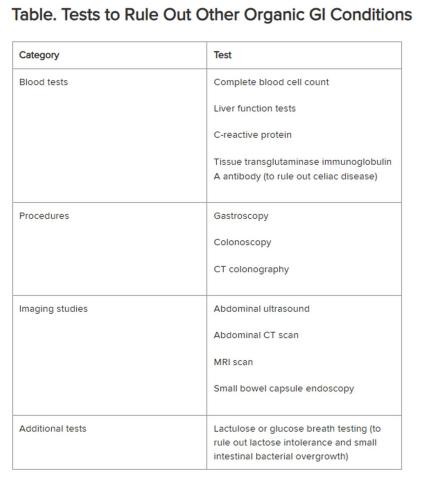
Historically, IBS has been regarded as a “diagnosis of exclusion” because classic diagnostic tests typically yield no concrete findings. Desmond noted that several blood tests, procedures, imaging studies, and other tests are available to rule out other organic GI conditions, as outlined in the Table.
If the patient comes from a geographical region where giardia is endemic, clinicians also should consider testing for the parasite, Chey said.
New Understanding of IBS Etiology
Now, advances in the understanding of IBS are changing the approach to the disease.
“The field is moving away from seeing IBS as a ‘wastebasket diagnosis,’ recognizing that there are other causes of a patient’s symptoms,” Mark Pimentel, MD, associate professor of medicine and gastroenterology, Cedars-Sinai, Los Angeles, said in an interview. “What’s made IBS so difficult to diagnose has been the absence of biological markers and hallmark findings on endoscopy.”
Recent research points to novel bacterial causes as culprits in the development of IBS. In particular, altered small bowel microbiota can be triggered by acute gastroenteritis.
Food poisoning can trigger the onset of IBS — a phenomenon called “postinfectious IBS (PI-IBS),” said Pimentel, who is also executive director of the Medically Associated Science and Technology Program at Cedars-Sinai. PI-IBS almost always takes the form of IBS-D, with up to 60% of patients with IBS-D suffering the long-term sequelae of food poisoning.
The types of bacteria most commonly associated with gastroenteritis are Shigella, Campylobacter, Salmonella, and Escherichia coli, Pimentel said. All of them release cytolethal distending toxin B (CdtB), causing the body to produce antibodies to the toxin.
CdtB resembles vinculin, a naturally occurring protein critical for healthy gut function. “Because of this molecular resemblance, the immune system often mistakes one for the other, producing anti-vinculin,” Pimentel explained.
This autoimmune response leads to disruptions in the gut microbiome, ultimately resulting in PI-IBS. The chain of events “doesn’t necessarily happen immediately,” Pimentel said. “You might have developed food poisoning at a party weeks or months ago.”
Acute gastroenteritis is common, affecting as many as 179 million people in the United States annually. A meta-analysis of 47 studies, incorporating 28,270 patients, found that those who had experienced acute gastroenteritis had a fourfold higher risk of developing IBS compared with nonexposed controls.
“The problem isn’t only the IBS itself, but the fact that people with PI-IBS are four times as likely to contract food poisoning again, which can further exacerbate IBS symptoms,” Pimentel said.
Diarrhea-predominant IBS can be detected through the presence of two blood biomarkers — anti-CdtB and anti-vinculin — in a blood test developed by Pimentel and his group.
“Elevation in either of these biomarkers establishes the diagnosis,” Pimentel said. “This is a breakthrough because it represents the first test that can make IBS a ‘diagnosis of inclusion.’”
The blood test also can identify IBS-M but not IBS-C.
Pimentel said that IBS-C is associated with increased levels of methanogenic archaea, which can be diagnosed by a positive methane breath test. “Methane gas slows intestinal contractility, which might result in constipation,” he said.
Diet as a Treatment Option
Diet is usually the starting point for IBS treatment, Chey said. “The standard dietary recommendations, as defined by the National Institute for Health and Care Excellence Guidance for managing IBS, are reasonable and common sense — eating three meals a day, avoiding carbonated beverages, excess alcohol, and excess caffeine, and avoiding hard-to-digest foods that can be gas producing.”
A diet low in fermentable oligosaccharides, disaccharides, monosaccharides and polyols (FODMAPs), which are carbohydrates that aren’t completely absorbed in the intestines, has been shown to be effective in alleviating GI distress in as many as 86% of patients with IBS, leading to improvements in overall GI symptoms as well as individual symptoms (eg, abdominal pain, bloating, constipation, diarrhea, and flatulence).
Desmond recommends the low FODMAP program delineated by Monash University in Australia. The diet should be undertaken only under the supervision of a dietitian, he warned. Moreover, following it on a long-term basis can have an adverse impact on dietary quality and the gut microbiome. Therefore, “it’s important to embark on stepwise reintroduction of FODMAPS under supervision to find acceptable thresholds that don’t cause a return of symptoms.”
A growing body of research suggests that following the Mediterranean diet can be helpful in reducing IBS symptoms. Chey said that some patients who tend to over-restrict their eating might benefit from a less restrictive diet than the typical low FODMAPs diet. For them, the Mediterranean diet may be a good option.
Pharmacotherapy for IBS
Nutritional approaches aren’t for everyone, Chey noted. “Some people don’t want to be on a highly restricted diet.” For them, medications addressing symptoms might be a better option.
Antispasmodics — either anticholinergics (hyoscine and dicyclomine) or smooth muscle relaxants (alverine, mebeverine, and peppermint oil) — can be helpful, although they can worsen constipation in a dose-dependent manner. It is advisable to use them on an as-needed rather than long-term basis.
Antidiarrheal agents include loperamide and diphenoxylate.
For constipation, laxatives (eg, senna, bisacodyl, polyethylene glycol, and sodium picosulfate) can be helpful.
Desmond noted that the American Gastroenterological Association does not recommend routine use of probiotics for most GI disorders, including IBS. Exceptions include prevention of Clostridioides difficile, ulcerative colitis, and pouchitis.
Targeting the Gut-Brain Relationship
Stress plays a role in exacerbating symptoms in patients with IBS and is an important target for intervention.
“If patients are living with a level of stress that’s impairing, we won’t be able to solve their gut issues until we resolve their stress issues,” Desmond said. “We need to calm the gut-microbiome-brain axis, which is multidimensional and bidirectional.”
Many people — even those without IBS — experience queasiness or diarrhea prior to a major event they’re nervous about, Chey noted. These events activate the brain, which activates the nervous system, which interacts with the GI tract. Indeed, IBS is now recognized as a disorder of gut-brain interaction, he said.
“We now know that the microbiome in the GI tract influences cognition and emotional function, depression, and anxiety. One might say that the gut is the ‘center of the universe’ to human beings,” Chey said.
Evidence-based psychological approaches for stress reduction in patients with IBS include cognitive behavioral therapy, specifically tailored to helping the patient identify associations between IBS symptoms and thoughts, emotions, and actions, as well as learning new behaviors and engaging in stress management. Psychodynamic (interpersonal) therapy enables patients to understand the connection between GI symptoms and interpersonal conflicts, emotional factors, or relationship difficulties.
Gut-directed hypnotherapy (GDH) is a “proven modality for IBS,” Desmond said. Unlike other forms of hypnotherapy, GDH focuses specifically on controlling and normalizing GI function. Studies have shown a reduction of ≥ 30% in abdominal pain in two thirds of participants, with overall response rates up to 85%. It can be delivered in an individual or group setting or via a smartphone.
Desmond recommends mindfulness-based therapy (MBT) for IBS. MBT focuses on the “cultivation of mindfulness, defined as intentional, nonjudgmental, present-focused awareness.” It has been found effective in reducing flares and the markers of gut inflammation in ulcerative colitis, as well as reducing symptoms of IBS.
Chey noted that an emerging body of literature supports the potential role of acupuncture in treating IBS, and his clinic employs it. “I would like to see further research into other areas of CAM [complementary and alternative medicine], including herbal approaches to IBS symptoms as well as stress.”
Finally, all the experts agree that more research is needed.
“The real tragedy is that the NIH invests next to nothing in IBS, in contrast to inflammatory bowel disease and many other conditions,” Pimentel said. “Yet IBS is 45 times more common than inflammatory bowel disease.”
Pimentel hopes that with enough advocacy and recognition that IBS isn’t “just stress-related,” more resources will be devoted to understanding this debilitating condition.
Desmond is the author of a book on the benefits of a plant-based diet. He has also received honoraria, speaking, and consultancy fees from the European Space Agency, Dyson Institute of Engineering and Technology, Riverford Organic Farmers, Ltd., Salesforce Inc., Sentara Healthcare, Saudi Sports for All Federation, the Physicians Committee for Responsible Medicine, The Plantrician Project, Doctors for Nutrition, and The Happy Pear.
Pimentel is a consultant for Bausch Health, Ferring Pharmaceuticals, and Ardelyx. He holds equity in and is also a consultant for Dieta Health, Salvo Health, Cylinder Health, and Gemelli Biotech. Cedars-Sinai has a licensing agreement with Gemelli Biotech and Hobbs Medical.
Chey is a consultant to AbbVie, Ardelyx, Atmo, Biomerica, Gemelli Biotech, Ironwood Pharmaceuticals, Nestlé, QOL Medical, Phathom Pharmaceuticals, Redhill, Salix/Valeant, Takeda, and Vibrant. He receives grant/research funding from Commonwealth Diagnostics International, Inc., US Food and Drug Administration, National Institutes of Health, QOL Medical, and Salix/Valeant. He holds stock options in Coprata, Dieta Health, Evinature, FoodMarble, Kiwi Biosciences, and ModifyHealth. He is a board or advisory panel member of the American College of Gastroenterology, GI Health Foundation, International Foundation for Gastrointestinal Disorders, Rome. He holds patents on My Nutrition Health, Digital Manometry, and Rectal Expulsion Device.
A version of this article appeared on Medscape.com.
Irritable bowel syndrome (IBS) is one of the most common conditions encountered by both primary care providers and gastroenterologists, with a pooled global prevalence of 11.2%. This functional bowel disorder is characterized by abdominal pain or discomfort, diarrhea and/or constipation, and bloating.
Unfortunately, , according to Alan Desmond, MB, consultant in gastroenterology and general internal medicine, Torbay Hospital, UK National Health Service.
Desmond regularly sees patients who either haven’t been accurately diagnosed or have been told, “Don’t worry, it’s ‘just’ irritable bowel syndrome,” he said at the recent International Conference on Nutrition in Medicine.
A 2017 study involving nearly 2000 patients with a history of gastrointestinal (GI) symptoms found that 43.1% of those who met the criteria for IBS were undiagnosed, and among those who were diagnosed, 26% were not receiving treatment.
“Many clinicians vastly underestimate the impact functional GI symptoms have on our patients in lack of productivity, becoming homebound or losing employment, the inability to enjoy a meal with friends or family, and always needing to know where the nearest bathroom is, for example,” Desmond said in an interview.
IBS can profoundly affect patients’ mental health. One study found that 38% of patients with IBS attending a tertiary care clinic contemplated suicide because they felt hopeless about ever achieving symptom relief.
Today, several dietary, pharmacologic, and psychological/behavioral approaches are available to treat patients with IBS, noted William D. Chey, MD, AGAF, chief of the Division of Gastroenterology and Hepatology, University of Michigan, Ann Arbor, Michigan.
“Each individual patient may need a different combination of these foundational treatments,” he said. “One size doesn’t fit all.”
Diagnostic Pathway
One reason IBS is so hard to diagnose is that it’s a “symptom-based disorder, with identification of the condition predicated upon certain key characteristics that are heterogeneous,” Chey said in an interview. “IBS in patient ‘A’ may not present the same way as IBS in patient ‘B,’ although there are certain foundational common characteristics.”
IBS involves “abnormalities in the motility and contractility of the GI tract,” he said. It can present with diarrhea (IBS-D), constipation (IBS-C), or a mixture or alternation of diarrhea and constipation (IBS-M).
Patients with IBS-D often have an exaggerated gastro-colonic response, while those with IBS-C often have a blunted response.
Beyond stool abnormalities and abdominal pain/discomfort, patients often report bloating/distension, low backache, lethargy, nausea, thigh pain, and urinary and gynecologic symptoms.
Historically, IBS has been regarded as a “diagnosis of exclusion” because classic diagnostic tests typically yield no concrete findings. Desmond noted that several blood tests, procedures, imaging studies, and other tests are available to rule out other organic GI conditions, as outlined in the Table.
If the patient comes from a geographical region where giardia is endemic, clinicians also should consider testing for the parasite, Chey said.
New Understanding of IBS Etiology
Now, advances in the understanding of IBS are changing the approach to the disease.
“The field is moving away from seeing IBS as a ‘wastebasket diagnosis,’ recognizing that there are other causes of a patient’s symptoms,” Mark Pimentel, MD, associate professor of medicine and gastroenterology, Cedars-Sinai, Los Angeles, said in an interview. “What’s made IBS so difficult to diagnose has been the absence of biological markers and hallmark findings on endoscopy.”
Recent research points to novel bacterial causes as culprits in the development of IBS. In particular, altered small bowel microbiota can be triggered by acute gastroenteritis.
Food poisoning can trigger the onset of IBS — a phenomenon called “postinfectious IBS (PI-IBS),” said Pimentel, who is also executive director of the Medically Associated Science and Technology Program at Cedars-Sinai. PI-IBS almost always takes the form of IBS-D, with up to 60% of patients with IBS-D suffering the long-term sequelae of food poisoning.
The types of bacteria most commonly associated with gastroenteritis are Shigella, Campylobacter, Salmonella, and Escherichia coli, Pimentel said. All of them release cytolethal distending toxin B (CdtB), causing the body to produce antibodies to the toxin.
CdtB resembles vinculin, a naturally occurring protein critical for healthy gut function. “Because of this molecular resemblance, the immune system often mistakes one for the other, producing anti-vinculin,” Pimentel explained.
This autoimmune response leads to disruptions in the gut microbiome, ultimately resulting in PI-IBS. The chain of events “doesn’t necessarily happen immediately,” Pimentel said. “You might have developed food poisoning at a party weeks or months ago.”
Acute gastroenteritis is common, affecting as many as 179 million people in the United States annually. A meta-analysis of 47 studies, incorporating 28,270 patients, found that those who had experienced acute gastroenteritis had a fourfold higher risk of developing IBS compared with nonexposed controls.
“The problem isn’t only the IBS itself, but the fact that people with PI-IBS are four times as likely to contract food poisoning again, which can further exacerbate IBS symptoms,” Pimentel said.
Diarrhea-predominant IBS can be detected through the presence of two blood biomarkers — anti-CdtB and anti-vinculin — in a blood test developed by Pimentel and his group.
“Elevation in either of these biomarkers establishes the diagnosis,” Pimentel said. “This is a breakthrough because it represents the first test that can make IBS a ‘diagnosis of inclusion.’”
The blood test also can identify IBS-M but not IBS-C.
Pimentel said that IBS-C is associated with increased levels of methanogenic archaea, which can be diagnosed by a positive methane breath test. “Methane gas slows intestinal contractility, which might result in constipation,” he said.
Diet as a Treatment Option
Diet is usually the starting point for IBS treatment, Chey said. “The standard dietary recommendations, as defined by the National Institute for Health and Care Excellence Guidance for managing IBS, are reasonable and common sense — eating three meals a day, avoiding carbonated beverages, excess alcohol, and excess caffeine, and avoiding hard-to-digest foods that can be gas producing.”
A diet low in fermentable oligosaccharides, disaccharides, monosaccharides and polyols (FODMAPs), which are carbohydrates that aren’t completely absorbed in the intestines, has been shown to be effective in alleviating GI distress in as many as 86% of patients with IBS, leading to improvements in overall GI symptoms as well as individual symptoms (eg, abdominal pain, bloating, constipation, diarrhea, and flatulence).
Desmond recommends the low FODMAP program delineated by Monash University in Australia. The diet should be undertaken only under the supervision of a dietitian, he warned. Moreover, following it on a long-term basis can have an adverse impact on dietary quality and the gut microbiome. Therefore, “it’s important to embark on stepwise reintroduction of FODMAPS under supervision to find acceptable thresholds that don’t cause a return of symptoms.”
A growing body of research suggests that following the Mediterranean diet can be helpful in reducing IBS symptoms. Chey said that some patients who tend to over-restrict their eating might benefit from a less restrictive diet than the typical low FODMAPs diet. For them, the Mediterranean diet may be a good option.
Pharmacotherapy for IBS
Nutritional approaches aren’t for everyone, Chey noted. “Some people don’t want to be on a highly restricted diet.” For them, medications addressing symptoms might be a better option.
Antispasmodics — either anticholinergics (hyoscine and dicyclomine) or smooth muscle relaxants (alverine, mebeverine, and peppermint oil) — can be helpful, although they can worsen constipation in a dose-dependent manner. It is advisable to use them on an as-needed rather than long-term basis.
Antidiarrheal agents include loperamide and diphenoxylate.
For constipation, laxatives (eg, senna, bisacodyl, polyethylene glycol, and sodium picosulfate) can be helpful.
Desmond noted that the American Gastroenterological Association does not recommend routine use of probiotics for most GI disorders, including IBS. Exceptions include prevention of Clostridioides difficile, ulcerative colitis, and pouchitis.
Targeting the Gut-Brain Relationship
Stress plays a role in exacerbating symptoms in patients with IBS and is an important target for intervention.
“If patients are living with a level of stress that’s impairing, we won’t be able to solve their gut issues until we resolve their stress issues,” Desmond said. “We need to calm the gut-microbiome-brain axis, which is multidimensional and bidirectional.”
Many people — even those without IBS — experience queasiness or diarrhea prior to a major event they’re nervous about, Chey noted. These events activate the brain, which activates the nervous system, which interacts with the GI tract. Indeed, IBS is now recognized as a disorder of gut-brain interaction, he said.
“We now know that the microbiome in the GI tract influences cognition and emotional function, depression, and anxiety. One might say that the gut is the ‘center of the universe’ to human beings,” Chey said.
Evidence-based psychological approaches for stress reduction in patients with IBS include cognitive behavioral therapy, specifically tailored to helping the patient identify associations between IBS symptoms and thoughts, emotions, and actions, as well as learning new behaviors and engaging in stress management. Psychodynamic (interpersonal) therapy enables patients to understand the connection between GI symptoms and interpersonal conflicts, emotional factors, or relationship difficulties.
Gut-directed hypnotherapy (GDH) is a “proven modality for IBS,” Desmond said. Unlike other forms of hypnotherapy, GDH focuses specifically on controlling and normalizing GI function. Studies have shown a reduction of ≥ 30% in abdominal pain in two thirds of participants, with overall response rates up to 85%. It can be delivered in an individual or group setting or via a smartphone.
Desmond recommends mindfulness-based therapy (MBT) for IBS. MBT focuses on the “cultivation of mindfulness, defined as intentional, nonjudgmental, present-focused awareness.” It has been found effective in reducing flares and the markers of gut inflammation in ulcerative colitis, as well as reducing symptoms of IBS.
Chey noted that an emerging body of literature supports the potential role of acupuncture in treating IBS, and his clinic employs it. “I would like to see further research into other areas of CAM [complementary and alternative medicine], including herbal approaches to IBS symptoms as well as stress.”
Finally, all the experts agree that more research is needed.
“The real tragedy is that the NIH invests next to nothing in IBS, in contrast to inflammatory bowel disease and many other conditions,” Pimentel said. “Yet IBS is 45 times more common than inflammatory bowel disease.”
Pimentel hopes that with enough advocacy and recognition that IBS isn’t “just stress-related,” more resources will be devoted to understanding this debilitating condition.
Desmond is the author of a book on the benefits of a plant-based diet. He has also received honoraria, speaking, and consultancy fees from the European Space Agency, Dyson Institute of Engineering and Technology, Riverford Organic Farmers, Ltd., Salesforce Inc., Sentara Healthcare, Saudi Sports for All Federation, the Physicians Committee for Responsible Medicine, The Plantrician Project, Doctors for Nutrition, and The Happy Pear.
Pimentel is a consultant for Bausch Health, Ferring Pharmaceuticals, and Ardelyx. He holds equity in and is also a consultant for Dieta Health, Salvo Health, Cylinder Health, and Gemelli Biotech. Cedars-Sinai has a licensing agreement with Gemelli Biotech and Hobbs Medical.
Chey is a consultant to AbbVie, Ardelyx, Atmo, Biomerica, Gemelli Biotech, Ironwood Pharmaceuticals, Nestlé, QOL Medical, Phathom Pharmaceuticals, Redhill, Salix/Valeant, Takeda, and Vibrant. He receives grant/research funding from Commonwealth Diagnostics International, Inc., US Food and Drug Administration, National Institutes of Health, QOL Medical, and Salix/Valeant. He holds stock options in Coprata, Dieta Health, Evinature, FoodMarble, Kiwi Biosciences, and ModifyHealth. He is a board or advisory panel member of the American College of Gastroenterology, GI Health Foundation, International Foundation for Gastrointestinal Disorders, Rome. He holds patents on My Nutrition Health, Digital Manometry, and Rectal Expulsion Device.
A version of this article appeared on Medscape.com.
Irritable bowel syndrome (IBS) is one of the most common conditions encountered by both primary care providers and gastroenterologists, with a pooled global prevalence of 11.2%. This functional bowel disorder is characterized by abdominal pain or discomfort, diarrhea and/or constipation, and bloating.
Unfortunately, , according to Alan Desmond, MB, consultant in gastroenterology and general internal medicine, Torbay Hospital, UK National Health Service.
Desmond regularly sees patients who either haven’t been accurately diagnosed or have been told, “Don’t worry, it’s ‘just’ irritable bowel syndrome,” he said at the recent International Conference on Nutrition in Medicine.
A 2017 study involving nearly 2000 patients with a history of gastrointestinal (GI) symptoms found that 43.1% of those who met the criteria for IBS were undiagnosed, and among those who were diagnosed, 26% were not receiving treatment.
“Many clinicians vastly underestimate the impact functional GI symptoms have on our patients in lack of productivity, becoming homebound or losing employment, the inability to enjoy a meal with friends or family, and always needing to know where the nearest bathroom is, for example,” Desmond said in an interview.
IBS can profoundly affect patients’ mental health. One study found that 38% of patients with IBS attending a tertiary care clinic contemplated suicide because they felt hopeless about ever achieving symptom relief.
Today, several dietary, pharmacologic, and psychological/behavioral approaches are available to treat patients with IBS, noted William D. Chey, MD, AGAF, chief of the Division of Gastroenterology and Hepatology, University of Michigan, Ann Arbor, Michigan.
“Each individual patient may need a different combination of these foundational treatments,” he said. “One size doesn’t fit all.”
Diagnostic Pathway
One reason IBS is so hard to diagnose is that it’s a “symptom-based disorder, with identification of the condition predicated upon certain key characteristics that are heterogeneous,” Chey said in an interview. “IBS in patient ‘A’ may not present the same way as IBS in patient ‘B,’ although there are certain foundational common characteristics.”
IBS involves “abnormalities in the motility and contractility of the GI tract,” he said. It can present with diarrhea (IBS-D), constipation (IBS-C), or a mixture or alternation of diarrhea and constipation (IBS-M).
Patients with IBS-D often have an exaggerated gastro-colonic response, while those with IBS-C often have a blunted response.
Beyond stool abnormalities and abdominal pain/discomfort, patients often report bloating/distension, low backache, lethargy, nausea, thigh pain, and urinary and gynecologic symptoms.
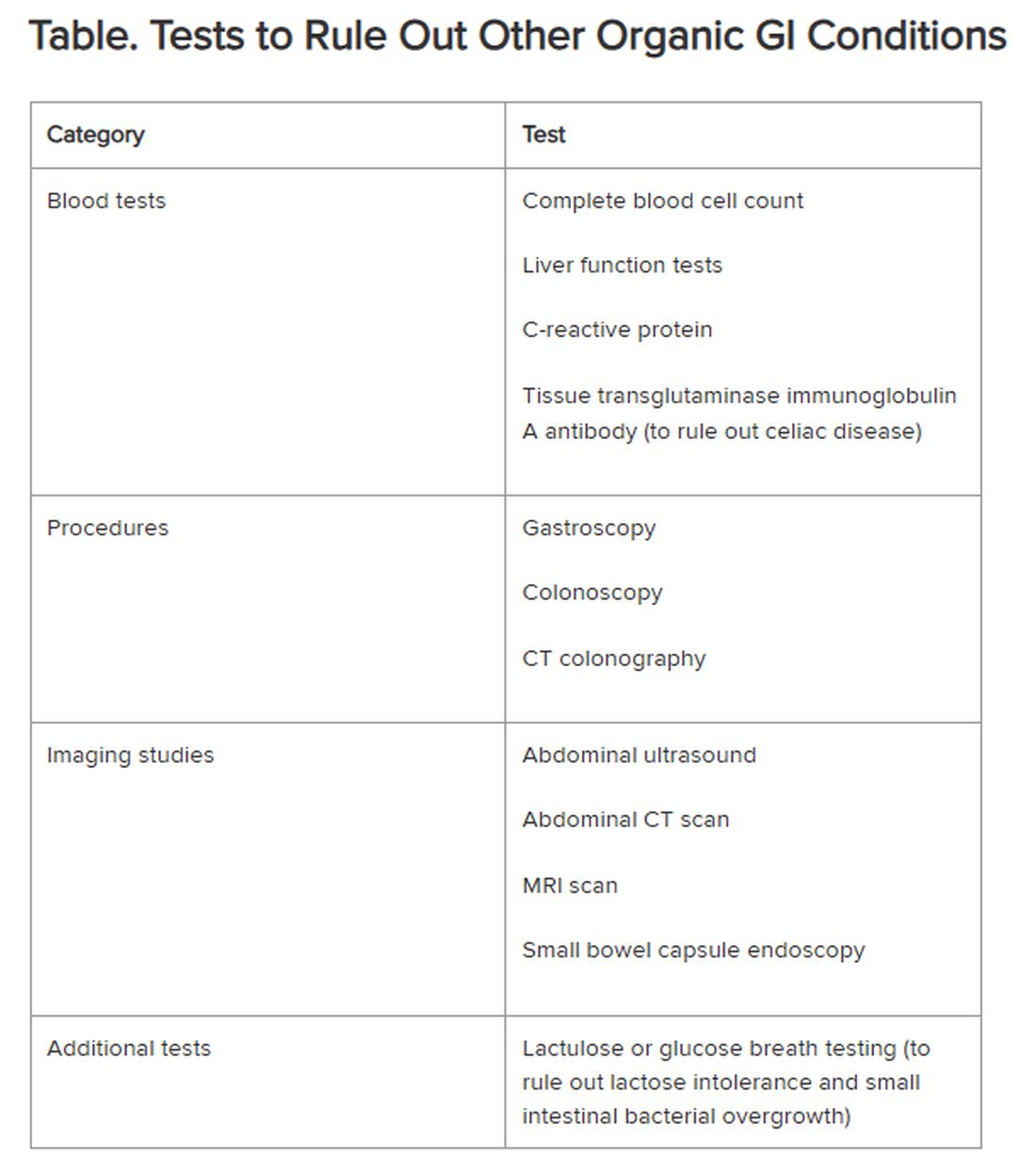
Historically, IBS has been regarded as a “diagnosis of exclusion” because classic diagnostic tests typically yield no concrete findings. Desmond noted that several blood tests, procedures, imaging studies, and other tests are available to rule out other organic GI conditions, as outlined in the Table.
If the patient comes from a geographical region where giardia is endemic, clinicians also should consider testing for the parasite, Chey said.
New Understanding of IBS Etiology
Now, advances in the understanding of IBS are changing the approach to the disease.
“The field is moving away from seeing IBS as a ‘wastebasket diagnosis,’ recognizing that there are other causes of a patient’s symptoms,” Mark Pimentel, MD, associate professor of medicine and gastroenterology, Cedars-Sinai, Los Angeles, said in an interview. “What’s made IBS so difficult to diagnose has been the absence of biological markers and hallmark findings on endoscopy.”
Recent research points to novel bacterial causes as culprits in the development of IBS. In particular, altered small bowel microbiota can be triggered by acute gastroenteritis.
Food poisoning can trigger the onset of IBS — a phenomenon called “postinfectious IBS (PI-IBS),” said Pimentel, who is also executive director of the Medically Associated Science and Technology Program at Cedars-Sinai. PI-IBS almost always takes the form of IBS-D, with up to 60% of patients with IBS-D suffering the long-term sequelae of food poisoning.
The types of bacteria most commonly associated with gastroenteritis are Shigella, Campylobacter, Salmonella, and Escherichia coli, Pimentel said. All of them release cytolethal distending toxin B (CdtB), causing the body to produce antibodies to the toxin.
CdtB resembles vinculin, a naturally occurring protein critical for healthy gut function. “Because of this molecular resemblance, the immune system often mistakes one for the other, producing anti-vinculin,” Pimentel explained.
This autoimmune response leads to disruptions in the gut microbiome, ultimately resulting in PI-IBS. The chain of events “doesn’t necessarily happen immediately,” Pimentel said. “You might have developed food poisoning at a party weeks or months ago.”
Acute gastroenteritis is common, affecting as many as 179 million people in the United States annually. A meta-analysis of 47 studies, incorporating 28,270 patients, found that those who had experienced acute gastroenteritis had a fourfold higher risk of developing IBS compared with nonexposed controls.
“The problem isn’t only the IBS itself, but the fact that people with PI-IBS are four times as likely to contract food poisoning again, which can further exacerbate IBS symptoms,” Pimentel said.
Diarrhea-predominant IBS can be detected through the presence of two blood biomarkers — anti-CdtB and anti-vinculin — in a blood test developed by Pimentel and his group.
“Elevation in either of these biomarkers establishes the diagnosis,” Pimentel said. “This is a breakthrough because it represents the first test that can make IBS a ‘diagnosis of inclusion.’”
The blood test also can identify IBS-M but not IBS-C.
Pimentel said that IBS-C is associated with increased levels of methanogenic archaea, which can be diagnosed by a positive methane breath test. “Methane gas slows intestinal contractility, which might result in constipation,” he said.
Diet as a Treatment Option
Diet is usually the starting point for IBS treatment, Chey said. “The standard dietary recommendations, as defined by the National Institute for Health and Care Excellence Guidance for managing IBS, are reasonable and common sense — eating three meals a day, avoiding carbonated beverages, excess alcohol, and excess caffeine, and avoiding hard-to-digest foods that can be gas producing.”
A diet low in fermentable oligosaccharides, disaccharides, monosaccharides and polyols (FODMAPs), which are carbohydrates that aren’t completely absorbed in the intestines, has been shown to be effective in alleviating GI distress in as many as 86% of patients with IBS, leading to improvements in overall GI symptoms as well as individual symptoms (eg, abdominal pain, bloating, constipation, diarrhea, and flatulence).
Desmond recommends the low FODMAP program delineated by Monash University in Australia. The diet should be undertaken only under the supervision of a dietitian, he warned. Moreover, following it on a long-term basis can have an adverse impact on dietary quality and the gut microbiome. Therefore, “it’s important to embark on stepwise reintroduction of FODMAPS under supervision to find acceptable thresholds that don’t cause a return of symptoms.”
A growing body of research suggests that following the Mediterranean diet can be helpful in reducing IBS symptoms. Chey said that some patients who tend to over-restrict their eating might benefit from a less restrictive diet than the typical low FODMAPs diet. For them, the Mediterranean diet may be a good option.
Pharmacotherapy for IBS
Nutritional approaches aren’t for everyone, Chey noted. “Some people don’t want to be on a highly restricted diet.” For them, medications addressing symptoms might be a better option.
Antispasmodics — either anticholinergics (hyoscine and dicyclomine) or smooth muscle relaxants (alverine, mebeverine, and peppermint oil) — can be helpful, although they can worsen constipation in a dose-dependent manner. It is advisable to use them on an as-needed rather than long-term basis.
Antidiarrheal agents include loperamide and diphenoxylate.
For constipation, laxatives (eg, senna, bisacodyl, polyethylene glycol, and sodium picosulfate) can be helpful.
Desmond noted that the American Gastroenterological Association does not recommend routine use of probiotics for most GI disorders, including IBS. Exceptions include prevention of Clostridioides difficile, ulcerative colitis, and pouchitis.
Targeting the Gut-Brain Relationship
Stress plays a role in exacerbating symptoms in patients with IBS and is an important target for intervention.
“If patients are living with a level of stress that’s impairing, we won’t be able to solve their gut issues until we resolve their stress issues,” Desmond said. “We need to calm the gut-microbiome-brain axis, which is multidimensional and bidirectional.”
Many people — even those without IBS — experience queasiness or diarrhea prior to a major event they’re nervous about, Chey noted. These events activate the brain, which activates the nervous system, which interacts with the GI tract. Indeed, IBS is now recognized as a disorder of gut-brain interaction, he said.
“We now know that the microbiome in the GI tract influences cognition and emotional function, depression, and anxiety. One might say that the gut is the ‘center of the universe’ to human beings,” Chey said.
Evidence-based psychological approaches for stress reduction in patients with IBS include cognitive behavioral therapy, specifically tailored to helping the patient identify associations between IBS symptoms and thoughts, emotions, and actions, as well as learning new behaviors and engaging in stress management. Psychodynamic (interpersonal) therapy enables patients to understand the connection between GI symptoms and interpersonal conflicts, emotional factors, or relationship difficulties.
Gut-directed hypnotherapy (GDH) is a “proven modality for IBS,” Desmond said. Unlike other forms of hypnotherapy, GDH focuses specifically on controlling and normalizing GI function. Studies have shown a reduction of ≥ 30% in abdominal pain in two thirds of participants, with overall response rates up to 85%. It can be delivered in an individual or group setting or via a smartphone.
Desmond recommends mindfulness-based therapy (MBT) for IBS. MBT focuses on the “cultivation of mindfulness, defined as intentional, nonjudgmental, present-focused awareness.” It has been found effective in reducing flares and the markers of gut inflammation in ulcerative colitis, as well as reducing symptoms of IBS.
Chey noted that an emerging body of literature supports the potential role of acupuncture in treating IBS, and his clinic employs it. “I would like to see further research into other areas of CAM [complementary and alternative medicine], including herbal approaches to IBS symptoms as well as stress.”
Finally, all the experts agree that more research is needed.
“The real tragedy is that the NIH invests next to nothing in IBS, in contrast to inflammatory bowel disease and many other conditions,” Pimentel said. “Yet IBS is 45 times more common than inflammatory bowel disease.”
Pimentel hopes that with enough advocacy and recognition that IBS isn’t “just stress-related,” more resources will be devoted to understanding this debilitating condition.
Desmond is the author of a book on the benefits of a plant-based diet. He has also received honoraria, speaking, and consultancy fees from the European Space Agency, Dyson Institute of Engineering and Technology, Riverford Organic Farmers, Ltd., Salesforce Inc., Sentara Healthcare, Saudi Sports for All Federation, the Physicians Committee for Responsible Medicine, The Plantrician Project, Doctors for Nutrition, and The Happy Pear.
Pimentel is a consultant for Bausch Health, Ferring Pharmaceuticals, and Ardelyx. He holds equity in and is also a consultant for Dieta Health, Salvo Health, Cylinder Health, and Gemelli Biotech. Cedars-Sinai has a licensing agreement with Gemelli Biotech and Hobbs Medical.
Chey is a consultant to AbbVie, Ardelyx, Atmo, Biomerica, Gemelli Biotech, Ironwood Pharmaceuticals, Nestlé, QOL Medical, Phathom Pharmaceuticals, Redhill, Salix/Valeant, Takeda, and Vibrant. He receives grant/research funding from Commonwealth Diagnostics International, Inc., US Food and Drug Administration, National Institutes of Health, QOL Medical, and Salix/Valeant. He holds stock options in Coprata, Dieta Health, Evinature, FoodMarble, Kiwi Biosciences, and ModifyHealth. He is a board or advisory panel member of the American College of Gastroenterology, GI Health Foundation, International Foundation for Gastrointestinal Disorders, Rome. He holds patents on My Nutrition Health, Digital Manometry, and Rectal Expulsion Device.
A version of this article appeared on Medscape.com.
Breath Gas Patterns Predict Response to Low FODMAP Diet
PHILADELPHIA — , according to a new study.
The low FODMAP diet is the most evidence-based dietary therapy for patients with IBS, but we know that “only about 50% of our patients respond to it,” said principal investigator Prashant Singh, MD, assistant professor at the University of Michigan in Ann Arbor, Michigan. “Exhaled breath gases represent bacterial fermentation of dietary carbohydrates. These measurements could provide a simple biomarker for response to low FODMAP diets.”
Even before starting the low FODMAP diet, “you could see notable differences in breath test patterns between responders and nonresponders,” he said. “We saw that low FODMAP responders had higher hydrogen (H2) and lower methane (CH4) at baseline than nonresponders and had a greater drop in hydrogen following FODMAP restriction vs nonresponders.”
He added that these results imply that responders to this diet may exhibit differences in baseline microbiota composition regarding saccharolytic capacity and/or methanogens.
Singh presented the findings at the American College of Gastroenterology (ACG) 2024 Annual Scientific Meeting.
Breaths That Can Predict Response
To determine if pre-intervention non-fasting breath patterns are associated with a clinical response to low FODMAP diets, Singh and colleagues enrolled 284 self-selected participants (mean age, 45.2 years) with mild to moderate gastrointestinal (GI) symptoms. Participants used an app-connected breath analyzer to record hourly, non-fasting H2 and CH4 levels during waking hours, in addition to logging meal content and symptom severity (bloating, abdominal pain, and flatulence) on a 0-10 scale.
Patients were directed to consume their habitual diet for 1 week, before following an app-directed low FODMAP diet for 1 week. Responders were defined as those with a ≥ 30% reduction in at least one mean symptom score. The researchers then compared average hourly H2 and CH4 levels and symptom scores at baseline between low FODMAP diet responders and nonresponders.
Of the participants, 111 were classified as responders and 173 as nonresponders. There were no significant differences between the groups in gender, age, body mass index, or FODMAP per calorie.
Following FODMAP restriction, responders had consistently lower abdominal pain throughout the day and lower bloating and flatulence predominantly in the latter part of the day. Nonresponders experienced no significant changes in key abdominal symptoms after adopting the low FODMAP diet.
The researchers found that breath tests taken at baseline revealed predictive trends between the groups, even though average FODMAP consumption did not significantly differ between them. Baseline H2 levels were higher among responders than among nonresponders, especially in the morning and evening. However, responders had lower baseline CH4 levels throughout the day.
Following FODMAP restrictions, responders had a significant drop in non-fasting H2 but not CH4, whereas nonresponders did not have a significant drop in either.
The study was limited by the fact that participants were not clinically diagnosed with IBS, their GI symptoms were mild overall, and no data were available on stool consistency/frequency or fecal microbiome composition for correlation with exhaled breath gas levels.
A Potential New Biomarker
Session co-moderator Kyle Staller, MD, MPH, director of the Gastrointestinal Motility Laboratory at Mass General and associate professor of medicine at Harvard Medical School in Boston, Massachusetts, said in an interview that if validated, these findings provide hope for better directing low FODMAP diets to those patients who may benefit.
There are some patients who may or may not respond to a FODMAP diet, for reasons we don’t yet know, possibly related to fermentation of gas, and it’s helpful to know before starting treatment, he said. It may help us with more of “a precision medicine approach before we really torture people with diets that can be very difficult to adhere to.”
Staller, who was not involved in the study, added that, “People tend to really focus on small intestinal bacteria overgrowth when it comes to hydrogen and methane production, but in reality, this is really a very agile day-to-day, meal-to-meal responsiveness.
“It’s a different paradigm,” he continued. “I’d also like to see more data as to why we see the diurnal rhythm” and whether potential factors such as intestinal transit times are playing a role.
Singh reported receiving royalties from UpToDate. Staller reported receiving research support from Ardelyx and Restasis and serving as a consultant to Anji, Ardelyx, GI Supply, Mahana, Restasis, and Sanofi. Funding associated with the study was not available at the time of publication.
A version of this article appeared on Medscape.com.
PHILADELPHIA — , according to a new study.
The low FODMAP diet is the most evidence-based dietary therapy for patients with IBS, but we know that “only about 50% of our patients respond to it,” said principal investigator Prashant Singh, MD, assistant professor at the University of Michigan in Ann Arbor, Michigan. “Exhaled breath gases represent bacterial fermentation of dietary carbohydrates. These measurements could provide a simple biomarker for response to low FODMAP diets.”
Even before starting the low FODMAP diet, “you could see notable differences in breath test patterns between responders and nonresponders,” he said. “We saw that low FODMAP responders had higher hydrogen (H2) and lower methane (CH4) at baseline than nonresponders and had a greater drop in hydrogen following FODMAP restriction vs nonresponders.”
He added that these results imply that responders to this diet may exhibit differences in baseline microbiota composition regarding saccharolytic capacity and/or methanogens.
Singh presented the findings at the American College of Gastroenterology (ACG) 2024 Annual Scientific Meeting.
Breaths That Can Predict Response
To determine if pre-intervention non-fasting breath patterns are associated with a clinical response to low FODMAP diets, Singh and colleagues enrolled 284 self-selected participants (mean age, 45.2 years) with mild to moderate gastrointestinal (GI) symptoms. Participants used an app-connected breath analyzer to record hourly, non-fasting H2 and CH4 levels during waking hours, in addition to logging meal content and symptom severity (bloating, abdominal pain, and flatulence) on a 0-10 scale.
Patients were directed to consume their habitual diet for 1 week, before following an app-directed low FODMAP diet for 1 week. Responders were defined as those with a ≥ 30% reduction in at least one mean symptom score. The researchers then compared average hourly H2 and CH4 levels and symptom scores at baseline between low FODMAP diet responders and nonresponders.
Of the participants, 111 were classified as responders and 173 as nonresponders. There were no significant differences between the groups in gender, age, body mass index, or FODMAP per calorie.
Following FODMAP restriction, responders had consistently lower abdominal pain throughout the day and lower bloating and flatulence predominantly in the latter part of the day. Nonresponders experienced no significant changes in key abdominal symptoms after adopting the low FODMAP diet.
The researchers found that breath tests taken at baseline revealed predictive trends between the groups, even though average FODMAP consumption did not significantly differ between them. Baseline H2 levels were higher among responders than among nonresponders, especially in the morning and evening. However, responders had lower baseline CH4 levels throughout the day.
Following FODMAP restrictions, responders had a significant drop in non-fasting H2 but not CH4, whereas nonresponders did not have a significant drop in either.
The study was limited by the fact that participants were not clinically diagnosed with IBS, their GI symptoms were mild overall, and no data were available on stool consistency/frequency or fecal microbiome composition for correlation with exhaled breath gas levels.
A Potential New Biomarker
Session co-moderator Kyle Staller, MD, MPH, director of the Gastrointestinal Motility Laboratory at Mass General and associate professor of medicine at Harvard Medical School in Boston, Massachusetts, said in an interview that if validated, these findings provide hope for better directing low FODMAP diets to those patients who may benefit.
There are some patients who may or may not respond to a FODMAP diet, for reasons we don’t yet know, possibly related to fermentation of gas, and it’s helpful to know before starting treatment, he said. It may help us with more of “a precision medicine approach before we really torture people with diets that can be very difficult to adhere to.”
Staller, who was not involved in the study, added that, “People tend to really focus on small intestinal bacteria overgrowth when it comes to hydrogen and methane production, but in reality, this is really a very agile day-to-day, meal-to-meal responsiveness.
“It’s a different paradigm,” he continued. “I’d also like to see more data as to why we see the diurnal rhythm” and whether potential factors such as intestinal transit times are playing a role.
Singh reported receiving royalties from UpToDate. Staller reported receiving research support from Ardelyx and Restasis and serving as a consultant to Anji, Ardelyx, GI Supply, Mahana, Restasis, and Sanofi. Funding associated with the study was not available at the time of publication.
A version of this article appeared on Medscape.com.
PHILADELPHIA — , according to a new study.
The low FODMAP diet is the most evidence-based dietary therapy for patients with IBS, but we know that “only about 50% of our patients respond to it,” said principal investigator Prashant Singh, MD, assistant professor at the University of Michigan in Ann Arbor, Michigan. “Exhaled breath gases represent bacterial fermentation of dietary carbohydrates. These measurements could provide a simple biomarker for response to low FODMAP diets.”
Even before starting the low FODMAP diet, “you could see notable differences in breath test patterns between responders and nonresponders,” he said. “We saw that low FODMAP responders had higher hydrogen (H2) and lower methane (CH4) at baseline than nonresponders and had a greater drop in hydrogen following FODMAP restriction vs nonresponders.”
He added that these results imply that responders to this diet may exhibit differences in baseline microbiota composition regarding saccharolytic capacity and/or methanogens.
Singh presented the findings at the American College of Gastroenterology (ACG) 2024 Annual Scientific Meeting.
Breaths That Can Predict Response
To determine if pre-intervention non-fasting breath patterns are associated with a clinical response to low FODMAP diets, Singh and colleagues enrolled 284 self-selected participants (mean age, 45.2 years) with mild to moderate gastrointestinal (GI) symptoms. Participants used an app-connected breath analyzer to record hourly, non-fasting H2 and CH4 levels during waking hours, in addition to logging meal content and symptom severity (bloating, abdominal pain, and flatulence) on a 0-10 scale.
Patients were directed to consume their habitual diet for 1 week, before following an app-directed low FODMAP diet for 1 week. Responders were defined as those with a ≥ 30% reduction in at least one mean symptom score. The researchers then compared average hourly H2 and CH4 levels and symptom scores at baseline between low FODMAP diet responders and nonresponders.
Of the participants, 111 were classified as responders and 173 as nonresponders. There were no significant differences between the groups in gender, age, body mass index, or FODMAP per calorie.
Following FODMAP restriction, responders had consistently lower abdominal pain throughout the day and lower bloating and flatulence predominantly in the latter part of the day. Nonresponders experienced no significant changes in key abdominal symptoms after adopting the low FODMAP diet.
The researchers found that breath tests taken at baseline revealed predictive trends between the groups, even though average FODMAP consumption did not significantly differ between them. Baseline H2 levels were higher among responders than among nonresponders, especially in the morning and evening. However, responders had lower baseline CH4 levels throughout the day.
Following FODMAP restrictions, responders had a significant drop in non-fasting H2 but not CH4, whereas nonresponders did not have a significant drop in either.
The study was limited by the fact that participants were not clinically diagnosed with IBS, their GI symptoms were mild overall, and no data were available on stool consistency/frequency or fecal microbiome composition for correlation with exhaled breath gas levels.
A Potential New Biomarker
Session co-moderator Kyle Staller, MD, MPH, director of the Gastrointestinal Motility Laboratory at Mass General and associate professor of medicine at Harvard Medical School in Boston, Massachusetts, said in an interview that if validated, these findings provide hope for better directing low FODMAP diets to those patients who may benefit.
There are some patients who may or may not respond to a FODMAP diet, for reasons we don’t yet know, possibly related to fermentation of gas, and it’s helpful to know before starting treatment, he said. It may help us with more of “a precision medicine approach before we really torture people with diets that can be very difficult to adhere to.”
Staller, who was not involved in the study, added that, “People tend to really focus on small intestinal bacteria overgrowth when it comes to hydrogen and methane production, but in reality, this is really a very agile day-to-day, meal-to-meal responsiveness.
“It’s a different paradigm,” he continued. “I’d also like to see more data as to why we see the diurnal rhythm” and whether potential factors such as intestinal transit times are playing a role.
Singh reported receiving royalties from UpToDate. Staller reported receiving research support from Ardelyx and Restasis and serving as a consultant to Anji, Ardelyx, GI Supply, Mahana, Restasis, and Sanofi. Funding associated with the study was not available at the time of publication.
A version of this article appeared on Medscape.com.
FROM ACG 2024
Ultraprocessed Foods Associated With Relapse Risk in Crohn’s Disease
VIENNA —
Certain subgroups of UPFs, specifically bread, pastries, and starch as well as oil and spreads, exhibited the strongest association with relapse risks of approximately threefold.
“In addition to treating active inflammatory bowel disease (IBD), we want to maintain remission for the long term,” Chen Sarbagili Shabat, PhD, clinical dietitian from Tel Aviv Medical Center in Israel, said in an interview. “It’s highly important. We know environmental factors are associated with the disease, which is why we can treat active disease with diet. Likewise, we can manage CD in a remission state with diet.”
This is the first prospective study of this particular level of UPFs in people with Crohn’s disease who are in remission, noted Shabat, who presented the findings at United European Gastroenterology (UEG) Week 2024.
Previously, a meta-analysis of prospective cohort studies showed that a diet high in UPFs is associated with a 70% increased risk for development of CD, and a longitudinal study showed that “Western” dietary patterns were associated with relapse risk in patients with IBD, Shabat reported.
Effect of High vs Low Intake of UPFs
The current single-center, prospective cohort study, followed 111 patients with CD every 3 months until relapse for up to 1 year.
Participants were aged 18-75 years (mean age, 38 years), with a median disease duration of 8.7 years. They were required to have maintained steroid-free clinical remission (Harvey-Bradshaw Index (HBI), < 5) for 3 months or more. The median duration of clinical remission at recruitment was 3 years.
Data collection included HBI level, medication type and dosage to ensure constant therapy and full compliance, and a stool sample for fecal calprotectin measurement.
The primary outcome comprised a clinical relapse HBI ≥ 5 over the 12-month follow-up or a change in disease activity requiring a change in medication, hospitalization, or any IBD-related surgery.
Participants were asked to complete a processed food questionnaire to assess the intake of UPFs and a food frequency questionnaire to assess the total intake of energy, macronutrients, and micronutrients. UPFs were divided into high and low intakes using a median cutoff of 3.6 servings/day.
The low intake group included 57 participants, and the high intake group included 54.
A total of 24 patients (21.6%) experienced a clinical relapse event, 7 in the low intake group vs 17 in the high intake group (hazard ratio [HR], 3.86; 95% CI, 1.30-11.47; P = .015 after adjustments).
In a subset of 97 patients with baseline fecal calprotectin measurements, 6 (n = 50) in the low intake group experienced a clinical relapse vs 15 (n = 47) in the high intake group (HR, 4.32; 95% CI, 1.36-13.73; P = .013 after adjustments).
Fecal calprotectin results were also suggestive of an association between high intake of UPFs and gut inflammation, Shabat reported.
Food Groups and Emulsifiers
UPFs were divided into subgroups: Bread, pastries, and starch; oils and spreads; ultraprocessed meat; sweet products and desserts; and ultraprocessed beverages.
The highest associations with relapse were in the subgroup of bread, pastries, and starch (HR, 3.37; 95% CI, 1.26-8.25) and the subgroup of oils and spreads (HR, 2.76; 95% CI, 1.02-7.45).
“The selection of healthy food is highly important, especially since we know that certain food ingredients can contribute to the pathogenesis of CD,” Shabat said. Patients can use partial enteral nutrition to provide 40%-50% of daily caloric intake in order to maintain remission, but she acknowledged it can be really difficult to adhere to.
She concluded by asserting that the study results, along with future research, should contribute to establishing nutritional guidelines to reduce UPF consumption in patients with CD in order to maintain remission.
Commenting on the study, Kevin Whelan, PhD, professor of dietetics and head of the Department of Nutritional Sciences at King’s College London in England, said that he was intrigued by the subgroup analysis that showed breads, pastries, oils, and spreads as having the strongest association with relapse risk.
He also remarked that these foods almost ubiquitously contain emulsifiers, and so the association might have less to do with UPFs in general and more to do with emulsifiers.
Concurring, Shabat noted that, while emulsifiers can negatively influence the microbiota and the gut barrier function, as well as contribute to intestinal inflammation, further mechanistic studies are required to understand these effects.
We need to determine if all additives have the same effect on the inflammatory process and also need studies looking at UPFs alone, she added.
Shabat reported receiving personal fees from Nestle Health Science (Wolfson Medical Center IP) for consulting and speaking and from Takeda and Ferring for speaking. Whelan reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
VIENNA —
Certain subgroups of UPFs, specifically bread, pastries, and starch as well as oil and spreads, exhibited the strongest association with relapse risks of approximately threefold.
“In addition to treating active inflammatory bowel disease (IBD), we want to maintain remission for the long term,” Chen Sarbagili Shabat, PhD, clinical dietitian from Tel Aviv Medical Center in Israel, said in an interview. “It’s highly important. We know environmental factors are associated with the disease, which is why we can treat active disease with diet. Likewise, we can manage CD in a remission state with diet.”
This is the first prospective study of this particular level of UPFs in people with Crohn’s disease who are in remission, noted Shabat, who presented the findings at United European Gastroenterology (UEG) Week 2024.
Previously, a meta-analysis of prospective cohort studies showed that a diet high in UPFs is associated with a 70% increased risk for development of CD, and a longitudinal study showed that “Western” dietary patterns were associated with relapse risk in patients with IBD, Shabat reported.
Effect of High vs Low Intake of UPFs
The current single-center, prospective cohort study, followed 111 patients with CD every 3 months until relapse for up to 1 year.
Participants were aged 18-75 years (mean age, 38 years), with a median disease duration of 8.7 years. They were required to have maintained steroid-free clinical remission (Harvey-Bradshaw Index (HBI), < 5) for 3 months or more. The median duration of clinical remission at recruitment was 3 years.
Data collection included HBI level, medication type and dosage to ensure constant therapy and full compliance, and a stool sample for fecal calprotectin measurement.
The primary outcome comprised a clinical relapse HBI ≥ 5 over the 12-month follow-up or a change in disease activity requiring a change in medication, hospitalization, or any IBD-related surgery.
Participants were asked to complete a processed food questionnaire to assess the intake of UPFs and a food frequency questionnaire to assess the total intake of energy, macronutrients, and micronutrients. UPFs were divided into high and low intakes using a median cutoff of 3.6 servings/day.
The low intake group included 57 participants, and the high intake group included 54.
A total of 24 patients (21.6%) experienced a clinical relapse event, 7 in the low intake group vs 17 in the high intake group (hazard ratio [HR], 3.86; 95% CI, 1.30-11.47; P = .015 after adjustments).
In a subset of 97 patients with baseline fecal calprotectin measurements, 6 (n = 50) in the low intake group experienced a clinical relapse vs 15 (n = 47) in the high intake group (HR, 4.32; 95% CI, 1.36-13.73; P = .013 after adjustments).
Fecal calprotectin results were also suggestive of an association between high intake of UPFs and gut inflammation, Shabat reported.
Food Groups and Emulsifiers
UPFs were divided into subgroups: Bread, pastries, and starch; oils and spreads; ultraprocessed meat; sweet products and desserts; and ultraprocessed beverages.
The highest associations with relapse were in the subgroup of bread, pastries, and starch (HR, 3.37; 95% CI, 1.26-8.25) and the subgroup of oils and spreads (HR, 2.76; 95% CI, 1.02-7.45).
“The selection of healthy food is highly important, especially since we know that certain food ingredients can contribute to the pathogenesis of CD,” Shabat said. Patients can use partial enteral nutrition to provide 40%-50% of daily caloric intake in order to maintain remission, but she acknowledged it can be really difficult to adhere to.
She concluded by asserting that the study results, along with future research, should contribute to establishing nutritional guidelines to reduce UPF consumption in patients with CD in order to maintain remission.
Commenting on the study, Kevin Whelan, PhD, professor of dietetics and head of the Department of Nutritional Sciences at King’s College London in England, said that he was intrigued by the subgroup analysis that showed breads, pastries, oils, and spreads as having the strongest association with relapse risk.
He also remarked that these foods almost ubiquitously contain emulsifiers, and so the association might have less to do with UPFs in general and more to do with emulsifiers.
Concurring, Shabat noted that, while emulsifiers can negatively influence the microbiota and the gut barrier function, as well as contribute to intestinal inflammation, further mechanistic studies are required to understand these effects.
We need to determine if all additives have the same effect on the inflammatory process and also need studies looking at UPFs alone, she added.
Shabat reported receiving personal fees from Nestle Health Science (Wolfson Medical Center IP) for consulting and speaking and from Takeda and Ferring for speaking. Whelan reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
VIENNA —
Certain subgroups of UPFs, specifically bread, pastries, and starch as well as oil and spreads, exhibited the strongest association with relapse risks of approximately threefold.
“In addition to treating active inflammatory bowel disease (IBD), we want to maintain remission for the long term,” Chen Sarbagili Shabat, PhD, clinical dietitian from Tel Aviv Medical Center in Israel, said in an interview. “It’s highly important. We know environmental factors are associated with the disease, which is why we can treat active disease with diet. Likewise, we can manage CD in a remission state with diet.”
This is the first prospective study of this particular level of UPFs in people with Crohn’s disease who are in remission, noted Shabat, who presented the findings at United European Gastroenterology (UEG) Week 2024.
Previously, a meta-analysis of prospective cohort studies showed that a diet high in UPFs is associated with a 70% increased risk for development of CD, and a longitudinal study showed that “Western” dietary patterns were associated with relapse risk in patients with IBD, Shabat reported.
Effect of High vs Low Intake of UPFs
The current single-center, prospective cohort study, followed 111 patients with CD every 3 months until relapse for up to 1 year.
Participants were aged 18-75 years (mean age, 38 years), with a median disease duration of 8.7 years. They were required to have maintained steroid-free clinical remission (Harvey-Bradshaw Index (HBI), < 5) for 3 months or more. The median duration of clinical remission at recruitment was 3 years.
Data collection included HBI level, medication type and dosage to ensure constant therapy and full compliance, and a stool sample for fecal calprotectin measurement.
The primary outcome comprised a clinical relapse HBI ≥ 5 over the 12-month follow-up or a change in disease activity requiring a change in medication, hospitalization, or any IBD-related surgery.
Participants were asked to complete a processed food questionnaire to assess the intake of UPFs and a food frequency questionnaire to assess the total intake of energy, macronutrients, and micronutrients. UPFs were divided into high and low intakes using a median cutoff of 3.6 servings/day.
The low intake group included 57 participants, and the high intake group included 54.
A total of 24 patients (21.6%) experienced a clinical relapse event, 7 in the low intake group vs 17 in the high intake group (hazard ratio [HR], 3.86; 95% CI, 1.30-11.47; P = .015 after adjustments).
In a subset of 97 patients with baseline fecal calprotectin measurements, 6 (n = 50) in the low intake group experienced a clinical relapse vs 15 (n = 47) in the high intake group (HR, 4.32; 95% CI, 1.36-13.73; P = .013 after adjustments).
Fecal calprotectin results were also suggestive of an association between high intake of UPFs and gut inflammation, Shabat reported.
Food Groups and Emulsifiers
UPFs were divided into subgroups: Bread, pastries, and starch; oils and spreads; ultraprocessed meat; sweet products and desserts; and ultraprocessed beverages.
The highest associations with relapse were in the subgroup of bread, pastries, and starch (HR, 3.37; 95% CI, 1.26-8.25) and the subgroup of oils and spreads (HR, 2.76; 95% CI, 1.02-7.45).
“The selection of healthy food is highly important, especially since we know that certain food ingredients can contribute to the pathogenesis of CD,” Shabat said. Patients can use partial enteral nutrition to provide 40%-50% of daily caloric intake in order to maintain remission, but she acknowledged it can be really difficult to adhere to.
She concluded by asserting that the study results, along with future research, should contribute to establishing nutritional guidelines to reduce UPF consumption in patients with CD in order to maintain remission.
Commenting on the study, Kevin Whelan, PhD, professor of dietetics and head of the Department of Nutritional Sciences at King’s College London in England, said that he was intrigued by the subgroup analysis that showed breads, pastries, oils, and spreads as having the strongest association with relapse risk.
He also remarked that these foods almost ubiquitously contain emulsifiers, and so the association might have less to do with UPFs in general and more to do with emulsifiers.
Concurring, Shabat noted that, while emulsifiers can negatively influence the microbiota and the gut barrier function, as well as contribute to intestinal inflammation, further mechanistic studies are required to understand these effects.
We need to determine if all additives have the same effect on the inflammatory process and also need studies looking at UPFs alone, she added.
Shabat reported receiving personal fees from Nestle Health Science (Wolfson Medical Center IP) for consulting and speaking and from Takeda and Ferring for speaking. Whelan reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM UEG 2024
Help Your Patients Reap the Benefits of Plant-Based Diets
Research pooled from nearly 100 studies has indicated that people who adhere to a vegan diet (ie, completely devoid of animal products) or a vegetarian diet (ie, devoid of meat, but may include dairy and eggs) are able to ward off some chronic diseases, such as cardiovascular disease, optimize glycemic control, and decrease their risk for cancer compared with those who consume omnivorous diets.
Vegan and vegetarian diets, or flexitarian diets — which are less reliant on animal protein than the standard US diet but do not completely exclude meat, fish, eggs, or dairy — may promote homeostasis and decrease inflammation by providing more fiber, antioxidants, and unsaturated fatty acids than the typical Western diet.
Inflammation and Obesity
Adipose tissue is a major producer of pro-inflammatory cytokines like interleukin (IL)-6, whose presence then triggers a rush of acute-phase reactants such as C-reactive protein (CRP) by the liver. This process develops into chronic low-grade inflammation that can increase a person’s chances of developing diabetes, cardiovascular disease, kidney disease, metabolic syndrome, and related complications.
Adopting a plant-based diet can improve markers of chronic low-grade inflammation that can lead to chronic disease and worsen existent chronic disease. A meta-analysis of 29 studies encompassing nearly 2700 participants found that initiation of a plant-based diet showed significant improvement in CRP, IL-6, and soluble intercellular adhesion molecule 1.
If we want to prevent these inflammatory disease states and their complications, the obvious response is to counsel patients to avoid excessive weight gain or to lose weight if obesity is their baseline. This can be tough for some patients, but it is nonetheless an important step in chronic disease prevention and management.
Plant-Based Diet for Type 2 Diabetes
According to a review of nine studies of patients living with type 2 diabetes who adhered to a plant-based diet, all but one found that this approach led to significantly lower A1c values than those seen in control groups. Six of the included studies reported that participants were able to decrease or discontinue medications for the management of diabetes. Researchers across all included studies also noted a decrease in total cholesterol, low-density lipoprotein cholesterol, and triglycerides, as well as increased weight loss in participants in each intervention group.
Such improvements are probably the result of the increase in fiber intake that occurs with a plant-based diet. A high-fiber diet is known to promote improved glucose and lipid metabolism as well as weight loss.
It is also worth noting that participants in the intervention groups also experienced improvements in depression and less chronic pain than did those in the control groups.
Plant-Based Diet for Chronic Kidney Disease (CKD)
Although the use of a plant-based diet in the prevention of CKD is well documented, adopting such diets for the treatment of CKD may intimidate both patients and practitioners owing to the high potassium and phosphorus content of many fruits and vegetables.
However, research indicates that the bioavailability of both potassium and phosphorus is lower in plant-based, whole foods than in preservatives and the highly processed food items that incorporate them. This makes a plant-based diet more viable than previously thought.
Diets rich in vegetables, whole grains, nuts, and legumes have been shown to decrease dietary acid load, both preventing and treating metabolic acidosis. Such diets have also been shown to decrease blood pressure and the risk for a decline in estimated glomerular filtration rate. This type of diet would also prioritize the unsaturated fatty acids and fiber-rich proteins such as avocados, beans, and nuts shown to improve dyslipidemia, which may occur alongside CKD.
Realistic Options for Patients on Medical Diets
There is one question that I always seem to get from when recommending a plant-based diet: “These patients already have so many restrictions. Why would you add more?” And my answer is also always the same: I don’t.
I rarely, if ever, recommend completely cutting out any food item or food group. Instead, I ask the patient to increase their intake of plant-based foods and only limit highly processed foods and fatty meats. By shifting a patient’s focus to beans; nuts; and low-carbohydrate, high-fiber fruits and vegetables, I am often opening up a whole new world of possibilities.
Instead of a sandwich with low-sodium turkey and cheese on white bread with a side of unsalted pretzels, I recommend a caprese salad with blueberries and almonds or a Southwest salad with black beans, corn, and avocado. I don’t encourage my patients to skip the foods that they love, but instead to only think about all the delicious plant-based options that will provide them with more than just calories.
Meat, dairy, seafood, and eggs can certainly be a part of a healthy diet, but what if our chronically ill patients, especially those with diabetes, had more options than just grilled chicken and green beans for every meal? What if we focus on decreasing dietary restrictions, incorporating a variety of nourishing foods, and educating our patients, instead of on portion control and moderation?
This is how I choose to incorporate plant-based diets into my practice to treat and prevent these chronic inflammatory conditions and promote sustainable, realistic change in my clients’ health.
Brandy Winfree Root, a renal dietitian in private practice in Mary Esther, Florida, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Research pooled from nearly 100 studies has indicated that people who adhere to a vegan diet (ie, completely devoid of animal products) or a vegetarian diet (ie, devoid of meat, but may include dairy and eggs) are able to ward off some chronic diseases, such as cardiovascular disease, optimize glycemic control, and decrease their risk for cancer compared with those who consume omnivorous diets.
Vegan and vegetarian diets, or flexitarian diets — which are less reliant on animal protein than the standard US diet but do not completely exclude meat, fish, eggs, or dairy — may promote homeostasis and decrease inflammation by providing more fiber, antioxidants, and unsaturated fatty acids than the typical Western diet.
Inflammation and Obesity
Adipose tissue is a major producer of pro-inflammatory cytokines like interleukin (IL)-6, whose presence then triggers a rush of acute-phase reactants such as C-reactive protein (CRP) by the liver. This process develops into chronic low-grade inflammation that can increase a person’s chances of developing diabetes, cardiovascular disease, kidney disease, metabolic syndrome, and related complications.
Adopting a plant-based diet can improve markers of chronic low-grade inflammation that can lead to chronic disease and worsen existent chronic disease. A meta-analysis of 29 studies encompassing nearly 2700 participants found that initiation of a plant-based diet showed significant improvement in CRP, IL-6, and soluble intercellular adhesion molecule 1.
If we want to prevent these inflammatory disease states and their complications, the obvious response is to counsel patients to avoid excessive weight gain or to lose weight if obesity is their baseline. This can be tough for some patients, but it is nonetheless an important step in chronic disease prevention and management.
Plant-Based Diet for Type 2 Diabetes
According to a review of nine studies of patients living with type 2 diabetes who adhered to a plant-based diet, all but one found that this approach led to significantly lower A1c values than those seen in control groups. Six of the included studies reported that participants were able to decrease or discontinue medications for the management of diabetes. Researchers across all included studies also noted a decrease in total cholesterol, low-density lipoprotein cholesterol, and triglycerides, as well as increased weight loss in participants in each intervention group.
Such improvements are probably the result of the increase in fiber intake that occurs with a plant-based diet. A high-fiber diet is known to promote improved glucose and lipid metabolism as well as weight loss.
It is also worth noting that participants in the intervention groups also experienced improvements in depression and less chronic pain than did those in the control groups.
Plant-Based Diet for Chronic Kidney Disease (CKD)
Although the use of a plant-based diet in the prevention of CKD is well documented, adopting such diets for the treatment of CKD may intimidate both patients and practitioners owing to the high potassium and phosphorus content of many fruits and vegetables.
However, research indicates that the bioavailability of both potassium and phosphorus is lower in plant-based, whole foods than in preservatives and the highly processed food items that incorporate them. This makes a plant-based diet more viable than previously thought.
Diets rich in vegetables, whole grains, nuts, and legumes have been shown to decrease dietary acid load, both preventing and treating metabolic acidosis. Such diets have also been shown to decrease blood pressure and the risk for a decline in estimated glomerular filtration rate. This type of diet would also prioritize the unsaturated fatty acids and fiber-rich proteins such as avocados, beans, and nuts shown to improve dyslipidemia, which may occur alongside CKD.
Realistic Options for Patients on Medical Diets
There is one question that I always seem to get from when recommending a plant-based diet: “These patients already have so many restrictions. Why would you add more?” And my answer is also always the same: I don’t.
I rarely, if ever, recommend completely cutting out any food item or food group. Instead, I ask the patient to increase their intake of plant-based foods and only limit highly processed foods and fatty meats. By shifting a patient’s focus to beans; nuts; and low-carbohydrate, high-fiber fruits and vegetables, I am often opening up a whole new world of possibilities.
Instead of a sandwich with low-sodium turkey and cheese on white bread with a side of unsalted pretzels, I recommend a caprese salad with blueberries and almonds or a Southwest salad with black beans, corn, and avocado. I don’t encourage my patients to skip the foods that they love, but instead to only think about all the delicious plant-based options that will provide them with more than just calories.
Meat, dairy, seafood, and eggs can certainly be a part of a healthy diet, but what if our chronically ill patients, especially those with diabetes, had more options than just grilled chicken and green beans for every meal? What if we focus on decreasing dietary restrictions, incorporating a variety of nourishing foods, and educating our patients, instead of on portion control and moderation?
This is how I choose to incorporate plant-based diets into my practice to treat and prevent these chronic inflammatory conditions and promote sustainable, realistic change in my clients’ health.
Brandy Winfree Root, a renal dietitian in private practice in Mary Esther, Florida, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Research pooled from nearly 100 studies has indicated that people who adhere to a vegan diet (ie, completely devoid of animal products) or a vegetarian diet (ie, devoid of meat, but may include dairy and eggs) are able to ward off some chronic diseases, such as cardiovascular disease, optimize glycemic control, and decrease their risk for cancer compared with those who consume omnivorous diets.
Vegan and vegetarian diets, or flexitarian diets — which are less reliant on animal protein than the standard US diet but do not completely exclude meat, fish, eggs, or dairy — may promote homeostasis and decrease inflammation by providing more fiber, antioxidants, and unsaturated fatty acids than the typical Western diet.
Inflammation and Obesity
Adipose tissue is a major producer of pro-inflammatory cytokines like interleukin (IL)-6, whose presence then triggers a rush of acute-phase reactants such as C-reactive protein (CRP) by the liver. This process develops into chronic low-grade inflammation that can increase a person’s chances of developing diabetes, cardiovascular disease, kidney disease, metabolic syndrome, and related complications.
Adopting a plant-based diet can improve markers of chronic low-grade inflammation that can lead to chronic disease and worsen existent chronic disease. A meta-analysis of 29 studies encompassing nearly 2700 participants found that initiation of a plant-based diet showed significant improvement in CRP, IL-6, and soluble intercellular adhesion molecule 1.
If we want to prevent these inflammatory disease states and their complications, the obvious response is to counsel patients to avoid excessive weight gain or to lose weight if obesity is their baseline. This can be tough for some patients, but it is nonetheless an important step in chronic disease prevention and management.
Plant-Based Diet for Type 2 Diabetes
According to a review of nine studies of patients living with type 2 diabetes who adhered to a plant-based diet, all but one found that this approach led to significantly lower A1c values than those seen in control groups. Six of the included studies reported that participants were able to decrease or discontinue medications for the management of diabetes. Researchers across all included studies also noted a decrease in total cholesterol, low-density lipoprotein cholesterol, and triglycerides, as well as increased weight loss in participants in each intervention group.
Such improvements are probably the result of the increase in fiber intake that occurs with a plant-based diet. A high-fiber diet is known to promote improved glucose and lipid metabolism as well as weight loss.
It is also worth noting that participants in the intervention groups also experienced improvements in depression and less chronic pain than did those in the control groups.
Plant-Based Diet for Chronic Kidney Disease (CKD)
Although the use of a plant-based diet in the prevention of CKD is well documented, adopting such diets for the treatment of CKD may intimidate both patients and practitioners owing to the high potassium and phosphorus content of many fruits and vegetables.
However, research indicates that the bioavailability of both potassium and phosphorus is lower in plant-based, whole foods than in preservatives and the highly processed food items that incorporate them. This makes a plant-based diet more viable than previously thought.
Diets rich in vegetables, whole grains, nuts, and legumes have been shown to decrease dietary acid load, both preventing and treating metabolic acidosis. Such diets have also been shown to decrease blood pressure and the risk for a decline in estimated glomerular filtration rate. This type of diet would also prioritize the unsaturated fatty acids and fiber-rich proteins such as avocados, beans, and nuts shown to improve dyslipidemia, which may occur alongside CKD.
Realistic Options for Patients on Medical Diets
There is one question that I always seem to get from when recommending a plant-based diet: “These patients already have so many restrictions. Why would you add more?” And my answer is also always the same: I don’t.
I rarely, if ever, recommend completely cutting out any food item or food group. Instead, I ask the patient to increase their intake of plant-based foods and only limit highly processed foods and fatty meats. By shifting a patient’s focus to beans; nuts; and low-carbohydrate, high-fiber fruits and vegetables, I am often opening up a whole new world of possibilities.
Instead of a sandwich with low-sodium turkey and cheese on white bread with a side of unsalted pretzels, I recommend a caprese salad with blueberries and almonds or a Southwest salad with black beans, corn, and avocado. I don’t encourage my patients to skip the foods that they love, but instead to only think about all the delicious plant-based options that will provide them with more than just calories.
Meat, dairy, seafood, and eggs can certainly be a part of a healthy diet, but what if our chronically ill patients, especially those with diabetes, had more options than just grilled chicken and green beans for every meal? What if we focus on decreasing dietary restrictions, incorporating a variety of nourishing foods, and educating our patients, instead of on portion control and moderation?
This is how I choose to incorporate plant-based diets into my practice to treat and prevent these chronic inflammatory conditions and promote sustainable, realistic change in my clients’ health.
Brandy Winfree Root, a renal dietitian in private practice in Mary Esther, Florida, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
How the Future of Medicine Will Revolve Around Our Gut
Meet your new patients.
You can’t see them, but trillions — maybe quadrillions — of them travel in and out of your practice every day. They’re hungry, mysterious, community-oriented, and small. Very, very small.
They’re the microbes occupying your current patients’ guts.
Someday soon, you’ll prescribe medicine not just for humans but also for these microbes.
“I am convinced in the future our medicine cabinets are going to have not just medications like a statin for treating us, but [also] pills that treat and inhibit an enzyme in our microbes and elicit a health benefit in some chronic disease,” said Stanley Hazen, MD, PhD, co-section head of Preventive Cardiology & Rehabilitation and director of the Center for Microbiome & Human Health at Cleveland Clinic, Cleveland, Ohio.
These trillions of microbes use our food to generate substances called metabolites that can protect or harm our health, with consequences reaching far beyond our gastrointestinal tracts.
Research has linked microbial metabolites to diabetes, cardiovascular disease, liver disease, obesity, high blood pressure, neurological disorders, depression, cancer, and more. Gastroenterologist Christopher Damman, MD, a clinical associate professor at the University of Washington Medical Center, Seattle, calls it a “growing theme” in microbiome science.
Now scientists are developing treatments targeting gut microbial pathways, designed to eliminate the bad metabolites and boost the good metabolites.
One close to human therapeutic intervention is an oral treatment from Dr. Hazen’s lab targeting the metabolite trimethylamine N-oxide (TMAO), a predictor of and contributor to both cardiovascular disease and chronic kidney disease. The drug, which blocks TMAO formation, is nearing clinical trials, Dr. Hazen said.
The advantage is safety. By targeting the microbe instead of, say, an enzyme, the host (your patient) must absorb little if any drug.
Implications for the future of medicine are huge. “Gut microbial pathways contribute to diabetes, obesity, virtually everything,” Dr. Hazen said. “Therapies that target gut microbiome processes will probably even be used for psychiatric disorders within, I’ll say, 10 or 20 years.”
The Science
About 100 trillion strains of bacteria live in our guts. As humans have evolved, so have they.
Between 70% and 90% come from the phyla Firmicutes and Bacteroidetes, with person-to-person variation shaped by genetics, environment, and lifestyle.
“Everyone’s microbiome is subtly different,” said Dr. Hazen. “So the combination of what they’re making is different. All these different biologically active compounds are influencing us in subtly different ways.”
How it works: When you eat, your microbes eat, breaking down food into metabolites that interact with the thin layer of epithelial cells lining your gut. Some can be absorbed through the lining and into your bloodstream, a phenomenon known as “leaky gut.” Once in your blood, they can trigger irritation and inflammation, potentially leading to a wide variety of health issues, from gas and bloating to autoimmune conditions and mood disorders.
“On the other side of the epithelial lining, you have some of the largest concentrations of immune cells,” said Narendra Kumar, PhD, associate professor of pharmaceutical sciences at Texas A&M University, College Station, Texas.
Metabolites can influence how these immune cells work, possibly explaining why each person’s immune system behaves distinctively.
Of the 1000-plus metabolites linked to the gut microbiome, scientists have identified several that matter.
Short-chain fatty acids. When we eat fiber, colon bacteria ferment it into the beneficial short-chain fatty acids acetate, propionate, and butyrate. These bind to receptors in muscle, liver, and fat tissue, affecting the secretion of gut hormones and peptides related to appetite, inflammation, energy expenditure, and fat oxidation.
Butyrate has been linked to health benefits. It supports the integrity of the gut’s lining, stifling pathogenic gut bacteria, fighting cancer-promoting inflammation, and protecting against obesity and diabetes. It can function as a prebiotic, helping beneficial bacteria thrive. And recent studies linked an abundance of butyrate-producing bacteria with reduced bone fracture risk and hospitalization for infectious disease.
TMAO and phenylacetylglutamine. When we eat foods rich in animal proteins — think eggs, milk, fish, and especially red meat — some gut bacteria convert nutrients like choline and L-carnitine into TMAO and phenylalanine into phenylacetylglutamine. Research conducted by Dr. Hazen’s lab and replicated by others has linked both metabolites to heart problems.
In a landmark study from Dr. Hazen’s group, healthy adults who went on to develop coronary artery disease had significantly higher plasma TMAO levels than those who did not wind up with the condition. The association remained strong, even after controlling for risk factors like age, sex, smoking, high blood pressure, and high cholesterol.
In preclinical studies, elevated TMAO enhanced cardiovascular disease. TMAO-producing microbes also accentuated cardiovascular disease phenotypes in mouse models, while blocking these pathways inhibited the phenotypes.
Research suggests TMAO may harm cardiomyocytes (cells that contract and relax the heart) in dozens of ways, such as activating the expression of proteins to promote hypertrophy and fibrosis, decreasing mitochondrial function, and disrupting calcium signaling.
Another study linked phenylacetylglutamine levels to cardiac event risk in patients with heart failure. Recent mechanistic investigations suggest the metabolite alters signaling in a beta-adrenergic receptor involved in our fight-or-flight response, said Hazen.
“It’s like a rheostat on the light switch, a dimmer switch, and it’s what’s called a negative allosteric modulator,” he said. “It’s the first time that this type of behavior has ever been shown to be present for a gut microbial metabolite and a host receptor.”
Tryptophan metabolites. Microbes in your colon can convert the amino acid tryptophan, also found in animal-based foods, into neurotransmitters like serotonin and melatonin.
“The enteric nervous system, the nervous system around the gut, is immense,” said James Versalovic, MD, PhD, professor of pathology and immunology at Baylor College of Medicine, Houston. “The gut-brain axis has become a very fertile area of research.”
Lesser-known tryptophan metabolites — like indole, tryptamine, and indoleethanol — have been linked to benefits like fortifying the gut barrier, promoting the release of glucagon-like peptide 1 to reduce appetite, and protecting the liver from hepatitis. However, indole can also spur the production of indoxyl sulfate, a toxin linked to chronic kidney disease.
Bile acid byproducts. Your gut bugs also feast on (and transform) bile acids before they reabsorb and travel back to the liver.
Research is gaining traction on these secondary bile acids, which can affect inflammation and immune function in helpful and harmful ways.
One area of interest is how microbes break down hormones in bile. A recent study from Harvard showed that gut microbes convert corticoid hormones in bile into progestins, which could affect postpartum depression risk. And researchers are exploring the estrobolome — a gut microbial community dedicated to breaking down estrogen into its active form so it can be reabsorbed.
“Depending on the bacteria that you have, more or less can be recirculated back into your blood,” said Beatriz Peñalver Bernabé, PhD, an assistant professor of biomedical engineering and urology at the University of Illinois Chicago. “So you may be producing the same amount of estrogen, but depending on the bacteria you have, the real free estrogen that can bind to your cells may be very different.”
The gut microbiome can also regulate testosterone, with studies showing microbial differences in men with high testosterone vs those with less.
What Patients Can Do Now
Advances in the field of microbiome research — and the related “gut health” wellness craze — have spawned all kinds of new microbiome-based products: Like over-the-counter probiotic supplements and at-home test kits, which let you send a stool sample for analysis to reveal microbiome health and personalized diet recommendations.
But the science behind these tests is still evolving, said Dr. Damman. “The clinical inferences and applications are still pretty limited.”
For most people, the first step to fostering healthier microbial metabolites is much simpler: Diversify your diet.
“A lot of folks are missing that diversity,” Dr. Damman said.
“Eat foods and experiment with foods that you might not eat all the time,” especially fruits, vegetables, nuts, seeds, and beans.
Another strategy: Eat foods with probiotic bacteria. “I view it as an insurance policy,” said Dr. Versalovic, “fortifying my gut with probiotics, with daily yogurt, for example, at breakfast.”
Fermented foods like kimchi and kombucha can also increase microbial diversity and can even contain health-promoting postbiotics, research shows.
As for probiotic supplements, the jury’s still out.
Certain strains of probiotic bacteria may be beneficial for some patients, like those with diarrhea, Crohn’s disease, and irritable bowel syndrome, according to World Gastroenterology Organisation guidelines.
As with other interventions, individual responses can vary. A Stanford study showed that some people with metabolic syndrome improved when taking a probiotic, while others didn’t. Both groups had key differences in gut bacteria and dietary habits.
For best results, such microbiome-based interventions will need to be personalized, experts say. And the technology to do that is coming sooner than you might think.
Microbiome’s Medical Future: ‘We Are on the Cusp of a New Era’
In just a few years, artificial intelligence (AI) models could predict gut microbial composition based on data such as dietary habits and household characteristics, Dr. Kumar said.
Advancements in metabolomics and bioinformatics could soon help physicians and patients personalize their treatment approaches, said Dr. Damman.
One focus will be on fortifying the gut with whatever it lacks.
“In those individuals where certain microbes are missing, (a) how could we add them back potentially in a rational, science-driven way, and (b) maybe some of those factors that the microbes are producing out the other ends, you could give directly,” said Dr. Damman.
For example, multiple companies make butyrate as a dietary supplement, although the research is too early to support widespread use. Another option could be eating something that spurs butyrate production. One small study found that a fiber supplement formulated to increase butyrate levels in the colon reduced participants’ systolic blood pressure by an average of six points.
Another option could be synbiotics, products that combine bacteria and the food source they feed on. “If you just give a diet-based therapy, it is not going to work as much. Because what if that diet needs certain bacteria to have these beneficial metabolites?” said Ashutosh Mangalam, PhD, associate professor of pathology at the University of Iowa Carver College of Medicine, Iowa City.
Dr. Mangalam studies links between bacterial metabolism of phytoestrogens in soy foods and multiple sclerosis (MS) development. He is using AI to understand differences in metabolites in patients with MS vs healthy controls to determine how to target them.
Gut microbial metabolites could also affect disease screening and intervention. What if gut microbe sequencing could predict a pregnant person’s risk of developing depression, something now assessed through simple questionnaires?
“Imagine that your doctor says, ‘Okay, give me a poop sample,’ ” Dr. Bernabé said. “Then they phenotype it, and then they put it in your electronic medical record, and they say, ‘Well, you have high likelihood of having a mood disorder down the line in your pregnancy. Why don’t we directly refer you to a provider now so you can follow up?’ ”
Research is already underway to understand how metabolites might be linked to pregnancy outcomes, complex regional pain syndrome, and anxiety. Researchers are also investigating whether supplementing our diets with things like prebiotic fibers, apple polyphenols, or tomato paste might influence metabolites. And fecal transplants that shift the gut microbiome and metabolites could have potential in diseases like unexplained atherosclerosis, post-COVID syndrome, and hidradenitis suppurativa.
Dr. Hazen’s discovery linking TMAO with cardiovascular risk has already changed clinical practice. A blood TMAO test can help identify patients at risk who may not have traditional risk factors. “Millions have been done,” Dr. Hazen said.
Meanwhile, his drug targeting the TMAO pathway inches closer to clinical trials.
“In an animal model, we elicit improvement in heart failure, renal disease, atherosclerosis, thrombosis, aortic aneurysm, and obesity,” Dr. Hazen said. The first clinical trials will focus on renal disease.
As with any drug, the road to approval takes time. And success is not guaranteed.
But Dr. Hazen for one is optimistic.
“We are on the cusp of a new era,” Dr. Hazen said. “Like when humans first discovered insulin and glucagon were hormones that impact sugar metabolism. We now recognize myriad new ‘hormones’ in the form of gut microbiome metabolites that impact our physiology and susceptibility to diseases.”
A version of this article first appeared on Medscape.com.
Meet your new patients.
You can’t see them, but trillions — maybe quadrillions — of them travel in and out of your practice every day. They’re hungry, mysterious, community-oriented, and small. Very, very small.
They’re the microbes occupying your current patients’ guts.
Someday soon, you’ll prescribe medicine not just for humans but also for these microbes.
“I am convinced in the future our medicine cabinets are going to have not just medications like a statin for treating us, but [also] pills that treat and inhibit an enzyme in our microbes and elicit a health benefit in some chronic disease,” said Stanley Hazen, MD, PhD, co-section head of Preventive Cardiology & Rehabilitation and director of the Center for Microbiome & Human Health at Cleveland Clinic, Cleveland, Ohio.
These trillions of microbes use our food to generate substances called metabolites that can protect or harm our health, with consequences reaching far beyond our gastrointestinal tracts.
Research has linked microbial metabolites to diabetes, cardiovascular disease, liver disease, obesity, high blood pressure, neurological disorders, depression, cancer, and more. Gastroenterologist Christopher Damman, MD, a clinical associate professor at the University of Washington Medical Center, Seattle, calls it a “growing theme” in microbiome science.
Now scientists are developing treatments targeting gut microbial pathways, designed to eliminate the bad metabolites and boost the good metabolites.
One close to human therapeutic intervention is an oral treatment from Dr. Hazen’s lab targeting the metabolite trimethylamine N-oxide (TMAO), a predictor of and contributor to both cardiovascular disease and chronic kidney disease. The drug, which blocks TMAO formation, is nearing clinical trials, Dr. Hazen said.
The advantage is safety. By targeting the microbe instead of, say, an enzyme, the host (your patient) must absorb little if any drug.
Implications for the future of medicine are huge. “Gut microbial pathways contribute to diabetes, obesity, virtually everything,” Dr. Hazen said. “Therapies that target gut microbiome processes will probably even be used for psychiatric disorders within, I’ll say, 10 or 20 years.”
The Science
About 100 trillion strains of bacteria live in our guts. As humans have evolved, so have they.
Between 70% and 90% come from the phyla Firmicutes and Bacteroidetes, with person-to-person variation shaped by genetics, environment, and lifestyle.
“Everyone’s microbiome is subtly different,” said Dr. Hazen. “So the combination of what they’re making is different. All these different biologically active compounds are influencing us in subtly different ways.”
How it works: When you eat, your microbes eat, breaking down food into metabolites that interact with the thin layer of epithelial cells lining your gut. Some can be absorbed through the lining and into your bloodstream, a phenomenon known as “leaky gut.” Once in your blood, they can trigger irritation and inflammation, potentially leading to a wide variety of health issues, from gas and bloating to autoimmune conditions and mood disorders.
“On the other side of the epithelial lining, you have some of the largest concentrations of immune cells,” said Narendra Kumar, PhD, associate professor of pharmaceutical sciences at Texas A&M University, College Station, Texas.
Metabolites can influence how these immune cells work, possibly explaining why each person’s immune system behaves distinctively.
Of the 1000-plus metabolites linked to the gut microbiome, scientists have identified several that matter.
Short-chain fatty acids. When we eat fiber, colon bacteria ferment it into the beneficial short-chain fatty acids acetate, propionate, and butyrate. These bind to receptors in muscle, liver, and fat tissue, affecting the secretion of gut hormones and peptides related to appetite, inflammation, energy expenditure, and fat oxidation.
Butyrate has been linked to health benefits. It supports the integrity of the gut’s lining, stifling pathogenic gut bacteria, fighting cancer-promoting inflammation, and protecting against obesity and diabetes. It can function as a prebiotic, helping beneficial bacteria thrive. And recent studies linked an abundance of butyrate-producing bacteria with reduced bone fracture risk and hospitalization for infectious disease.
TMAO and phenylacetylglutamine. When we eat foods rich in animal proteins — think eggs, milk, fish, and especially red meat — some gut bacteria convert nutrients like choline and L-carnitine into TMAO and phenylalanine into phenylacetylglutamine. Research conducted by Dr. Hazen’s lab and replicated by others has linked both metabolites to heart problems.
In a landmark study from Dr. Hazen’s group, healthy adults who went on to develop coronary artery disease had significantly higher plasma TMAO levels than those who did not wind up with the condition. The association remained strong, even after controlling for risk factors like age, sex, smoking, high blood pressure, and high cholesterol.
In preclinical studies, elevated TMAO enhanced cardiovascular disease. TMAO-producing microbes also accentuated cardiovascular disease phenotypes in mouse models, while blocking these pathways inhibited the phenotypes.
Research suggests TMAO may harm cardiomyocytes (cells that contract and relax the heart) in dozens of ways, such as activating the expression of proteins to promote hypertrophy and fibrosis, decreasing mitochondrial function, and disrupting calcium signaling.
Another study linked phenylacetylglutamine levels to cardiac event risk in patients with heart failure. Recent mechanistic investigations suggest the metabolite alters signaling in a beta-adrenergic receptor involved in our fight-or-flight response, said Hazen.
“It’s like a rheostat on the light switch, a dimmer switch, and it’s what’s called a negative allosteric modulator,” he said. “It’s the first time that this type of behavior has ever been shown to be present for a gut microbial metabolite and a host receptor.”
Tryptophan metabolites. Microbes in your colon can convert the amino acid tryptophan, also found in animal-based foods, into neurotransmitters like serotonin and melatonin.
“The enteric nervous system, the nervous system around the gut, is immense,” said James Versalovic, MD, PhD, professor of pathology and immunology at Baylor College of Medicine, Houston. “The gut-brain axis has become a very fertile area of research.”
Lesser-known tryptophan metabolites — like indole, tryptamine, and indoleethanol — have been linked to benefits like fortifying the gut barrier, promoting the release of glucagon-like peptide 1 to reduce appetite, and protecting the liver from hepatitis. However, indole can also spur the production of indoxyl sulfate, a toxin linked to chronic kidney disease.
Bile acid byproducts. Your gut bugs also feast on (and transform) bile acids before they reabsorb and travel back to the liver.
Research is gaining traction on these secondary bile acids, which can affect inflammation and immune function in helpful and harmful ways.
One area of interest is how microbes break down hormones in bile. A recent study from Harvard showed that gut microbes convert corticoid hormones in bile into progestins, which could affect postpartum depression risk. And researchers are exploring the estrobolome — a gut microbial community dedicated to breaking down estrogen into its active form so it can be reabsorbed.
“Depending on the bacteria that you have, more or less can be recirculated back into your blood,” said Beatriz Peñalver Bernabé, PhD, an assistant professor of biomedical engineering and urology at the University of Illinois Chicago. “So you may be producing the same amount of estrogen, but depending on the bacteria you have, the real free estrogen that can bind to your cells may be very different.”
The gut microbiome can also regulate testosterone, with studies showing microbial differences in men with high testosterone vs those with less.
What Patients Can Do Now
Advances in the field of microbiome research — and the related “gut health” wellness craze — have spawned all kinds of new microbiome-based products: Like over-the-counter probiotic supplements and at-home test kits, which let you send a stool sample for analysis to reveal microbiome health and personalized diet recommendations.
But the science behind these tests is still evolving, said Dr. Damman. “The clinical inferences and applications are still pretty limited.”
For most people, the first step to fostering healthier microbial metabolites is much simpler: Diversify your diet.
“A lot of folks are missing that diversity,” Dr. Damman said.
“Eat foods and experiment with foods that you might not eat all the time,” especially fruits, vegetables, nuts, seeds, and beans.
Another strategy: Eat foods with probiotic bacteria. “I view it as an insurance policy,” said Dr. Versalovic, “fortifying my gut with probiotics, with daily yogurt, for example, at breakfast.”
Fermented foods like kimchi and kombucha can also increase microbial diversity and can even contain health-promoting postbiotics, research shows.
As for probiotic supplements, the jury’s still out.
Certain strains of probiotic bacteria may be beneficial for some patients, like those with diarrhea, Crohn’s disease, and irritable bowel syndrome, according to World Gastroenterology Organisation guidelines.
As with other interventions, individual responses can vary. A Stanford study showed that some people with metabolic syndrome improved when taking a probiotic, while others didn’t. Both groups had key differences in gut bacteria and dietary habits.
For best results, such microbiome-based interventions will need to be personalized, experts say. And the technology to do that is coming sooner than you might think.
Microbiome’s Medical Future: ‘We Are on the Cusp of a New Era’
In just a few years, artificial intelligence (AI) models could predict gut microbial composition based on data such as dietary habits and household characteristics, Dr. Kumar said.
Advancements in metabolomics and bioinformatics could soon help physicians and patients personalize their treatment approaches, said Dr. Damman.
One focus will be on fortifying the gut with whatever it lacks.
“In those individuals where certain microbes are missing, (a) how could we add them back potentially in a rational, science-driven way, and (b) maybe some of those factors that the microbes are producing out the other ends, you could give directly,” said Dr. Damman.
For example, multiple companies make butyrate as a dietary supplement, although the research is too early to support widespread use. Another option could be eating something that spurs butyrate production. One small study found that a fiber supplement formulated to increase butyrate levels in the colon reduced participants’ systolic blood pressure by an average of six points.
Another option could be synbiotics, products that combine bacteria and the food source they feed on. “If you just give a diet-based therapy, it is not going to work as much. Because what if that diet needs certain bacteria to have these beneficial metabolites?” said Ashutosh Mangalam, PhD, associate professor of pathology at the University of Iowa Carver College of Medicine, Iowa City.
Dr. Mangalam studies links between bacterial metabolism of phytoestrogens in soy foods and multiple sclerosis (MS) development. He is using AI to understand differences in metabolites in patients with MS vs healthy controls to determine how to target them.
Gut microbial metabolites could also affect disease screening and intervention. What if gut microbe sequencing could predict a pregnant person’s risk of developing depression, something now assessed through simple questionnaires?
“Imagine that your doctor says, ‘Okay, give me a poop sample,’ ” Dr. Bernabé said. “Then they phenotype it, and then they put it in your electronic medical record, and they say, ‘Well, you have high likelihood of having a mood disorder down the line in your pregnancy. Why don’t we directly refer you to a provider now so you can follow up?’ ”
Research is already underway to understand how metabolites might be linked to pregnancy outcomes, complex regional pain syndrome, and anxiety. Researchers are also investigating whether supplementing our diets with things like prebiotic fibers, apple polyphenols, or tomato paste might influence metabolites. And fecal transplants that shift the gut microbiome and metabolites could have potential in diseases like unexplained atherosclerosis, post-COVID syndrome, and hidradenitis suppurativa.
Dr. Hazen’s discovery linking TMAO with cardiovascular risk has already changed clinical practice. A blood TMAO test can help identify patients at risk who may not have traditional risk factors. “Millions have been done,” Dr. Hazen said.
Meanwhile, his drug targeting the TMAO pathway inches closer to clinical trials.
“In an animal model, we elicit improvement in heart failure, renal disease, atherosclerosis, thrombosis, aortic aneurysm, and obesity,” Dr. Hazen said. The first clinical trials will focus on renal disease.
As with any drug, the road to approval takes time. And success is not guaranteed.
But Dr. Hazen for one is optimistic.
“We are on the cusp of a new era,” Dr. Hazen said. “Like when humans first discovered insulin and glucagon were hormones that impact sugar metabolism. We now recognize myriad new ‘hormones’ in the form of gut microbiome metabolites that impact our physiology and susceptibility to diseases.”
A version of this article first appeared on Medscape.com.
Meet your new patients.
You can’t see them, but trillions — maybe quadrillions — of them travel in and out of your practice every day. They’re hungry, mysterious, community-oriented, and small. Very, very small.
They’re the microbes occupying your current patients’ guts.
Someday soon, you’ll prescribe medicine not just for humans but also for these microbes.
“I am convinced in the future our medicine cabinets are going to have not just medications like a statin for treating us, but [also] pills that treat and inhibit an enzyme in our microbes and elicit a health benefit in some chronic disease,” said Stanley Hazen, MD, PhD, co-section head of Preventive Cardiology & Rehabilitation and director of the Center for Microbiome & Human Health at Cleveland Clinic, Cleveland, Ohio.
These trillions of microbes use our food to generate substances called metabolites that can protect or harm our health, with consequences reaching far beyond our gastrointestinal tracts.
Research has linked microbial metabolites to diabetes, cardiovascular disease, liver disease, obesity, high blood pressure, neurological disorders, depression, cancer, and more. Gastroenterologist Christopher Damman, MD, a clinical associate professor at the University of Washington Medical Center, Seattle, calls it a “growing theme” in microbiome science.
Now scientists are developing treatments targeting gut microbial pathways, designed to eliminate the bad metabolites and boost the good metabolites.
One close to human therapeutic intervention is an oral treatment from Dr. Hazen’s lab targeting the metabolite trimethylamine N-oxide (TMAO), a predictor of and contributor to both cardiovascular disease and chronic kidney disease. The drug, which blocks TMAO formation, is nearing clinical trials, Dr. Hazen said.
The advantage is safety. By targeting the microbe instead of, say, an enzyme, the host (your patient) must absorb little if any drug.
Implications for the future of medicine are huge. “Gut microbial pathways contribute to diabetes, obesity, virtually everything,” Dr. Hazen said. “Therapies that target gut microbiome processes will probably even be used for psychiatric disorders within, I’ll say, 10 or 20 years.”
The Science
About 100 trillion strains of bacteria live in our guts. As humans have evolved, so have they.
Between 70% and 90% come from the phyla Firmicutes and Bacteroidetes, with person-to-person variation shaped by genetics, environment, and lifestyle.
“Everyone’s microbiome is subtly different,” said Dr. Hazen. “So the combination of what they’re making is different. All these different biologically active compounds are influencing us in subtly different ways.”
How it works: When you eat, your microbes eat, breaking down food into metabolites that interact with the thin layer of epithelial cells lining your gut. Some can be absorbed through the lining and into your bloodstream, a phenomenon known as “leaky gut.” Once in your blood, they can trigger irritation and inflammation, potentially leading to a wide variety of health issues, from gas and bloating to autoimmune conditions and mood disorders.
“On the other side of the epithelial lining, you have some of the largest concentrations of immune cells,” said Narendra Kumar, PhD, associate professor of pharmaceutical sciences at Texas A&M University, College Station, Texas.
Metabolites can influence how these immune cells work, possibly explaining why each person’s immune system behaves distinctively.
Of the 1000-plus metabolites linked to the gut microbiome, scientists have identified several that matter.
Short-chain fatty acids. When we eat fiber, colon bacteria ferment it into the beneficial short-chain fatty acids acetate, propionate, and butyrate. These bind to receptors in muscle, liver, and fat tissue, affecting the secretion of gut hormones and peptides related to appetite, inflammation, energy expenditure, and fat oxidation.
Butyrate has been linked to health benefits. It supports the integrity of the gut’s lining, stifling pathogenic gut bacteria, fighting cancer-promoting inflammation, and protecting against obesity and diabetes. It can function as a prebiotic, helping beneficial bacteria thrive. And recent studies linked an abundance of butyrate-producing bacteria with reduced bone fracture risk and hospitalization for infectious disease.
TMAO and phenylacetylglutamine. When we eat foods rich in animal proteins — think eggs, milk, fish, and especially red meat — some gut bacteria convert nutrients like choline and L-carnitine into TMAO and phenylalanine into phenylacetylglutamine. Research conducted by Dr. Hazen’s lab and replicated by others has linked both metabolites to heart problems.
In a landmark study from Dr. Hazen’s group, healthy adults who went on to develop coronary artery disease had significantly higher plasma TMAO levels than those who did not wind up with the condition. The association remained strong, even after controlling for risk factors like age, sex, smoking, high blood pressure, and high cholesterol.
In preclinical studies, elevated TMAO enhanced cardiovascular disease. TMAO-producing microbes also accentuated cardiovascular disease phenotypes in mouse models, while blocking these pathways inhibited the phenotypes.
Research suggests TMAO may harm cardiomyocytes (cells that contract and relax the heart) in dozens of ways, such as activating the expression of proteins to promote hypertrophy and fibrosis, decreasing mitochondrial function, and disrupting calcium signaling.
Another study linked phenylacetylglutamine levels to cardiac event risk in patients with heart failure. Recent mechanistic investigations suggest the metabolite alters signaling in a beta-adrenergic receptor involved in our fight-or-flight response, said Hazen.
“It’s like a rheostat on the light switch, a dimmer switch, and it’s what’s called a negative allosteric modulator,” he said. “It’s the first time that this type of behavior has ever been shown to be present for a gut microbial metabolite and a host receptor.”
Tryptophan metabolites. Microbes in your colon can convert the amino acid tryptophan, also found in animal-based foods, into neurotransmitters like serotonin and melatonin.
“The enteric nervous system, the nervous system around the gut, is immense,” said James Versalovic, MD, PhD, professor of pathology and immunology at Baylor College of Medicine, Houston. “The gut-brain axis has become a very fertile area of research.”
Lesser-known tryptophan metabolites — like indole, tryptamine, and indoleethanol — have been linked to benefits like fortifying the gut barrier, promoting the release of glucagon-like peptide 1 to reduce appetite, and protecting the liver from hepatitis. However, indole can also spur the production of indoxyl sulfate, a toxin linked to chronic kidney disease.
Bile acid byproducts. Your gut bugs also feast on (and transform) bile acids before they reabsorb and travel back to the liver.
Research is gaining traction on these secondary bile acids, which can affect inflammation and immune function in helpful and harmful ways.
One area of interest is how microbes break down hormones in bile. A recent study from Harvard showed that gut microbes convert corticoid hormones in bile into progestins, which could affect postpartum depression risk. And researchers are exploring the estrobolome — a gut microbial community dedicated to breaking down estrogen into its active form so it can be reabsorbed.
“Depending on the bacteria that you have, more or less can be recirculated back into your blood,” said Beatriz Peñalver Bernabé, PhD, an assistant professor of biomedical engineering and urology at the University of Illinois Chicago. “So you may be producing the same amount of estrogen, but depending on the bacteria you have, the real free estrogen that can bind to your cells may be very different.”
The gut microbiome can also regulate testosterone, with studies showing microbial differences in men with high testosterone vs those with less.
What Patients Can Do Now
Advances in the field of microbiome research — and the related “gut health” wellness craze — have spawned all kinds of new microbiome-based products: Like over-the-counter probiotic supplements and at-home test kits, which let you send a stool sample for analysis to reveal microbiome health and personalized diet recommendations.
But the science behind these tests is still evolving, said Dr. Damman. “The clinical inferences and applications are still pretty limited.”
For most people, the first step to fostering healthier microbial metabolites is much simpler: Diversify your diet.
“A lot of folks are missing that diversity,” Dr. Damman said.
“Eat foods and experiment with foods that you might not eat all the time,” especially fruits, vegetables, nuts, seeds, and beans.
Another strategy: Eat foods with probiotic bacteria. “I view it as an insurance policy,” said Dr. Versalovic, “fortifying my gut with probiotics, with daily yogurt, for example, at breakfast.”
Fermented foods like kimchi and kombucha can also increase microbial diversity and can even contain health-promoting postbiotics, research shows.
As for probiotic supplements, the jury’s still out.
Certain strains of probiotic bacteria may be beneficial for some patients, like those with diarrhea, Crohn’s disease, and irritable bowel syndrome, according to World Gastroenterology Organisation guidelines.
As with other interventions, individual responses can vary. A Stanford study showed that some people with metabolic syndrome improved when taking a probiotic, while others didn’t. Both groups had key differences in gut bacteria and dietary habits.
For best results, such microbiome-based interventions will need to be personalized, experts say. And the technology to do that is coming sooner than you might think.
Microbiome’s Medical Future: ‘We Are on the Cusp of a New Era’
In just a few years, artificial intelligence (AI) models could predict gut microbial composition based on data such as dietary habits and household characteristics, Dr. Kumar said.
Advancements in metabolomics and bioinformatics could soon help physicians and patients personalize their treatment approaches, said Dr. Damman.
One focus will be on fortifying the gut with whatever it lacks.
“In those individuals where certain microbes are missing, (a) how could we add them back potentially in a rational, science-driven way, and (b) maybe some of those factors that the microbes are producing out the other ends, you could give directly,” said Dr. Damman.
For example, multiple companies make butyrate as a dietary supplement, although the research is too early to support widespread use. Another option could be eating something that spurs butyrate production. One small study found that a fiber supplement formulated to increase butyrate levels in the colon reduced participants’ systolic blood pressure by an average of six points.
Another option could be synbiotics, products that combine bacteria and the food source they feed on. “If you just give a diet-based therapy, it is not going to work as much. Because what if that diet needs certain bacteria to have these beneficial metabolites?” said Ashutosh Mangalam, PhD, associate professor of pathology at the University of Iowa Carver College of Medicine, Iowa City.
Dr. Mangalam studies links between bacterial metabolism of phytoestrogens in soy foods and multiple sclerosis (MS) development. He is using AI to understand differences in metabolites in patients with MS vs healthy controls to determine how to target them.
Gut microbial metabolites could also affect disease screening and intervention. What if gut microbe sequencing could predict a pregnant person’s risk of developing depression, something now assessed through simple questionnaires?
“Imagine that your doctor says, ‘Okay, give me a poop sample,’ ” Dr. Bernabé said. “Then they phenotype it, and then they put it in your electronic medical record, and they say, ‘Well, you have high likelihood of having a mood disorder down the line in your pregnancy. Why don’t we directly refer you to a provider now so you can follow up?’ ”
Research is already underway to understand how metabolites might be linked to pregnancy outcomes, complex regional pain syndrome, and anxiety. Researchers are also investigating whether supplementing our diets with things like prebiotic fibers, apple polyphenols, or tomato paste might influence metabolites. And fecal transplants that shift the gut microbiome and metabolites could have potential in diseases like unexplained atherosclerosis, post-COVID syndrome, and hidradenitis suppurativa.
Dr. Hazen’s discovery linking TMAO with cardiovascular risk has already changed clinical practice. A blood TMAO test can help identify patients at risk who may not have traditional risk factors. “Millions have been done,” Dr. Hazen said.
Meanwhile, his drug targeting the TMAO pathway inches closer to clinical trials.
“In an animal model, we elicit improvement in heart failure, renal disease, atherosclerosis, thrombosis, aortic aneurysm, and obesity,” Dr. Hazen said. The first clinical trials will focus on renal disease.
As with any drug, the road to approval takes time. And success is not guaranteed.
But Dr. Hazen for one is optimistic.
“We are on the cusp of a new era,” Dr. Hazen said. “Like when humans first discovered insulin and glucagon were hormones that impact sugar metabolism. We now recognize myriad new ‘hormones’ in the form of gut microbiome metabolites that impact our physiology and susceptibility to diseases.”
A version of this article first appeared on Medscape.com.
Over One Third of Patients Develop Exocrine Pancreatic Insufficiency After Acute Pancreatitis
TOPLINE:
Over one third of patients with acute pancreatitis develop exocrine pancreatic insufficiency (EPI) at 12 months, with the key predictors being idiopathic etiology, moderately severe or severe disease, and preexisting diabetes.
METHODOLOGY:
- EPI has traditionally been associated with chronic pancreatitis, but its prevalence and natural history following acute pancreatitis are less well defined.
- Researchers conducted a prospective cohort study including 85 hospital inpatients (mean age, 54.7 years; 48.2% women) diagnosed with acute pancreatitis from three tertiary institutions in the United States.
- Severity of acute pancreatitis was classified according to the Revised Atlanta Criteria.
- EPI was assessed by measuring fecal elastase 1 (FE-1) levels from stool samples at baseline and at 3 and 12 months after enrollment. EPI was defined by FE-1 levels ≤ 200 μg/g stool, with mild and severe EPI categorized by FE-1 levels of 101-200 μg/g stool and ≤ 100 μg/g stool, respectively.
- The prevalence of EPI was assessed at 3 and 12 months after acute pancreatitis. The study also identified the predictors of EPI, including the role of etiology and severity of acute pancreatitis and preexisting diabetes.
TAKEAWAY:
- EPI was present in 34.1% participants at 12 months after an acute pancreatitis attack, with 22.4% having severe EPI.
- Even 12.8% of those with an index mild attack of acute pancreatitis had severe EPI at 12 months.
- The odds of developing EPI at 12 months increased fourfold with idiopathic etiology of acute pancreatitis (P = .0094).
- The odds of developing EPI increased over threefold with moderately severe or severe acute pancreatitis (P = .025) and preexisting diabetes (P = .031).
- The prevalence of severe EPI after acute pancreatitis decreased from 29% at baseline to 26% at 3 months and 22% at 12 months.
IN PRACTICE:
“While specific subpopulations may have identified clinical risk factors, it will remain important to have a low threshold for testing and treatment as there remains much to learn about mechanisms leading to EPI after [acute pancreatitis],” the authors wrote.
SOURCE:
This study, led by Anna Evans Phillips, MD, MS, University of Pittsburgh School of Medicine in Pennsylvania, was published online in eClinicalMedicine.
LIMITATIONS:
Participants were often transferred from other hospitals with differing management techniques, which may have introduced selection bias. The use of FE-1 levels may have had diagnostic limitations. The study did not assess the impact of pancreatic enzyme replacement therapy on recovery from EPI. Some patients with early chronic pancreatitis may have been included owing to the lack of diagnostic clarity.
DISCLOSURES:
The study was supported by an investigator-initiated research grant from AbbVie. Some authors received funding for research from AbbVie. One of the authors declared serving as a consultant and scientific advisory board member and being an equity holder in biotechnology, biopharmaceutical, and diagnostics companies. Another author declared support from the Cystic Fibrosis Foundation and the American Society for Parenteral and Enteral Nutrition.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Over one third of patients with acute pancreatitis develop exocrine pancreatic insufficiency (EPI) at 12 months, with the key predictors being idiopathic etiology, moderately severe or severe disease, and preexisting diabetes.
METHODOLOGY:
- EPI has traditionally been associated with chronic pancreatitis, but its prevalence and natural history following acute pancreatitis are less well defined.
- Researchers conducted a prospective cohort study including 85 hospital inpatients (mean age, 54.7 years; 48.2% women) diagnosed with acute pancreatitis from three tertiary institutions in the United States.
- Severity of acute pancreatitis was classified according to the Revised Atlanta Criteria.
- EPI was assessed by measuring fecal elastase 1 (FE-1) levels from stool samples at baseline and at 3 and 12 months after enrollment. EPI was defined by FE-1 levels ≤ 200 μg/g stool, with mild and severe EPI categorized by FE-1 levels of 101-200 μg/g stool and ≤ 100 μg/g stool, respectively.
- The prevalence of EPI was assessed at 3 and 12 months after acute pancreatitis. The study also identified the predictors of EPI, including the role of etiology and severity of acute pancreatitis and preexisting diabetes.
TAKEAWAY:
- EPI was present in 34.1% participants at 12 months after an acute pancreatitis attack, with 22.4% having severe EPI.
- Even 12.8% of those with an index mild attack of acute pancreatitis had severe EPI at 12 months.
- The odds of developing EPI at 12 months increased fourfold with idiopathic etiology of acute pancreatitis (P = .0094).
- The odds of developing EPI increased over threefold with moderately severe or severe acute pancreatitis (P = .025) and preexisting diabetes (P = .031).
- The prevalence of severe EPI after acute pancreatitis decreased from 29% at baseline to 26% at 3 months and 22% at 12 months.
IN PRACTICE:
“While specific subpopulations may have identified clinical risk factors, it will remain important to have a low threshold for testing and treatment as there remains much to learn about mechanisms leading to EPI after [acute pancreatitis],” the authors wrote.
SOURCE:
This study, led by Anna Evans Phillips, MD, MS, University of Pittsburgh School of Medicine in Pennsylvania, was published online in eClinicalMedicine.
LIMITATIONS:
Participants were often transferred from other hospitals with differing management techniques, which may have introduced selection bias. The use of FE-1 levels may have had diagnostic limitations. The study did not assess the impact of pancreatic enzyme replacement therapy on recovery from EPI. Some patients with early chronic pancreatitis may have been included owing to the lack of diagnostic clarity.
DISCLOSURES:
The study was supported by an investigator-initiated research grant from AbbVie. Some authors received funding for research from AbbVie. One of the authors declared serving as a consultant and scientific advisory board member and being an equity holder in biotechnology, biopharmaceutical, and diagnostics companies. Another author declared support from the Cystic Fibrosis Foundation and the American Society for Parenteral and Enteral Nutrition.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Over one third of patients with acute pancreatitis develop exocrine pancreatic insufficiency (EPI) at 12 months, with the key predictors being idiopathic etiology, moderately severe or severe disease, and preexisting diabetes.
METHODOLOGY:
- EPI has traditionally been associated with chronic pancreatitis, but its prevalence and natural history following acute pancreatitis are less well defined.
- Researchers conducted a prospective cohort study including 85 hospital inpatients (mean age, 54.7 years; 48.2% women) diagnosed with acute pancreatitis from three tertiary institutions in the United States.
- Severity of acute pancreatitis was classified according to the Revised Atlanta Criteria.
- EPI was assessed by measuring fecal elastase 1 (FE-1) levels from stool samples at baseline and at 3 and 12 months after enrollment. EPI was defined by FE-1 levels ≤ 200 μg/g stool, with mild and severe EPI categorized by FE-1 levels of 101-200 μg/g stool and ≤ 100 μg/g stool, respectively.
- The prevalence of EPI was assessed at 3 and 12 months after acute pancreatitis. The study also identified the predictors of EPI, including the role of etiology and severity of acute pancreatitis and preexisting diabetes.
TAKEAWAY:
- EPI was present in 34.1% participants at 12 months after an acute pancreatitis attack, with 22.4% having severe EPI.
- Even 12.8% of those with an index mild attack of acute pancreatitis had severe EPI at 12 months.
- The odds of developing EPI at 12 months increased fourfold with idiopathic etiology of acute pancreatitis (P = .0094).
- The odds of developing EPI increased over threefold with moderately severe or severe acute pancreatitis (P = .025) and preexisting diabetes (P = .031).
- The prevalence of severe EPI after acute pancreatitis decreased from 29% at baseline to 26% at 3 months and 22% at 12 months.
IN PRACTICE:
“While specific subpopulations may have identified clinical risk factors, it will remain important to have a low threshold for testing and treatment as there remains much to learn about mechanisms leading to EPI after [acute pancreatitis],” the authors wrote.
SOURCE:
This study, led by Anna Evans Phillips, MD, MS, University of Pittsburgh School of Medicine in Pennsylvania, was published online in eClinicalMedicine.
LIMITATIONS:
Participants were often transferred from other hospitals with differing management techniques, which may have introduced selection bias. The use of FE-1 levels may have had diagnostic limitations. The study did not assess the impact of pancreatic enzyme replacement therapy on recovery from EPI. Some patients with early chronic pancreatitis may have been included owing to the lack of diagnostic clarity.
DISCLOSURES:
The study was supported by an investigator-initiated research grant from AbbVie. Some authors received funding for research from AbbVie. One of the authors declared serving as a consultant and scientific advisory board member and being an equity holder in biotechnology, biopharmaceutical, and diagnostics companies. Another author declared support from the Cystic Fibrosis Foundation and the American Society for Parenteral and Enteral Nutrition.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
The Next Frontier of Antibiotic Discovery: Inside Your Gut
Scientists at Stanford University and the University of Pennsylvania have discovered a new antibiotic candidate in a surprising place: the human gut.
In mice, the antibiotic — a peptide known as prevotellin-2 — showed antimicrobial potency on par with polymyxin B, an antibiotic medication used to treat multidrug-resistant infections. Meanwhile, the peptide mainly left commensal, or beneficial, bacteria alone. The study, published in Cell, also identified several other potent antibiotic peptides with the potential to combat antimicrobial-resistant infections.
The research is part of a larger quest to find new antibiotics that can fight drug-resistant infections, a critical public health threat with more than 2.8 million cases and 35,000 deaths annually in the United States. That quest is urgent, said study author César de la Fuente, PhD, professor of bioengineering at the University of Pennsylvania, Philadelphia.
“The main pillars that have enabled us to almost double our lifespan in the last 100 years or so have been antibiotics, vaccines, and clean water,” said Dr. de la Fuente. “Imagine taking out one of those. I think it would be pretty dramatic.” (Dr. De la Fuente’s lab has become known for finding antibiotic candidates in unusual places, like ancient genetic information of Neanderthals and woolly mammoths.)
The first widely used antibiotic, penicillin, was discovered in 1928, when a physician studying Staphylococcus bacteria returned to his lab after summer break to find mold growing in one of his petri dishes. But many other antibiotics — like streptomycin, tetracycline, and erythromycin — were discovered from soil bacteria, which produce variations of these substances to compete with other microorganisms.
By looking in the gut microbiome, the researchers hoped to identify peptides that the trillions of microbes use against each other in the fight for limited resources — ideally, peptides that wouldn’t broadly kill off the entire microbiome.
Kill the Bad, Spare the Good
Many traditional antibiotics are small molecules. This means they can wipe out the good bacteria in your body, and because each targets a specific bacterial function, bad bacteria can become resistant to them.
Peptide antibiotics, on the other hand, don’t diffuse into the whole body. If taken orally, they stay in the gut; if taken intravenously, they generally stay in the blood. And because of how they kill bacteria, targeting the membrane, they’re also less prone to bacterial resistance.
The microbiome is like a big reservoir of pathogens, said Ami Bhatt, MD, PhD, hematologist at Stanford University in California and one of the study’s authors. Because many antibiotics kill healthy gut bacteria, “what you have left over,” Dr. Bhatt said, “is this big open niche that gets filled up with multidrug-resistant organisms like E coli [Escherichia coli] or vancomycin-resistant Enterococcus.”
Dr. Bhatt has seen cancer patients undergo successful treatment only to die of a multidrug-resistant infection, because current antibiotics fail against those pathogens. “That’s like winning the battle to lose the war.”
By investigating the microbiome, “we wanted to see if we could identify antimicrobial peptides that might spare key members of our regular microbiome, so that we wouldn’t totally disrupt the microbiome the way we do when we use broad-spectrum, small molecule–based antibiotics,” Dr. Bhatt said.
The researchers used artificial intelligence to sift through 400,000 proteins to predict, based on known antibiotics, which peptide sequences might have antimicrobial properties. From the results, they chose 78 peptides to synthesize and test.
“The application of computational approaches combined with experimental validation is very powerful and exciting,” said Jennifer Geddes-McAlister, PhD, professor of cell biology at the University of Guelph in Ontario, Canada, who was not involved in the study. “The study is robust in its approach to microbiome sampling.”
The Long Journey from Lab to Clinic
More than half of the peptides the team tested effectively inhibited the growth of harmful bacteria, and prevotellin-2 (derived from the bacteria Prevotella copri)stood out as the most powerful.
“The study validates experimental data from the lab using animal models, which moves discoveries closer to the clinic,” said Dr. Geddes-McAlister. “Further testing with clinical trials is needed, but the potential for clinical application is promising.”
Unfortunately, that’s not likely to happen anytime soon, said Dr. de la Fuente. “There is not enough economic incentive” for companies to develop new antibiotics. Ten years is his most hopeful guess for when we might see prevotellin-2, or a similar antibiotic, complete clinical trials.
A version of this article first appeared on Medscape.com.
Scientists at Stanford University and the University of Pennsylvania have discovered a new antibiotic candidate in a surprising place: the human gut.
In mice, the antibiotic — a peptide known as prevotellin-2 — showed antimicrobial potency on par with polymyxin B, an antibiotic medication used to treat multidrug-resistant infections. Meanwhile, the peptide mainly left commensal, or beneficial, bacteria alone. The study, published in Cell, also identified several other potent antibiotic peptides with the potential to combat antimicrobial-resistant infections.
The research is part of a larger quest to find new antibiotics that can fight drug-resistant infections, a critical public health threat with more than 2.8 million cases and 35,000 deaths annually in the United States. That quest is urgent, said study author César de la Fuente, PhD, professor of bioengineering at the University of Pennsylvania, Philadelphia.
“The main pillars that have enabled us to almost double our lifespan in the last 100 years or so have been antibiotics, vaccines, and clean water,” said Dr. de la Fuente. “Imagine taking out one of those. I think it would be pretty dramatic.” (Dr. De la Fuente’s lab has become known for finding antibiotic candidates in unusual places, like ancient genetic information of Neanderthals and woolly mammoths.)
The first widely used antibiotic, penicillin, was discovered in 1928, when a physician studying Staphylococcus bacteria returned to his lab after summer break to find mold growing in one of his petri dishes. But many other antibiotics — like streptomycin, tetracycline, and erythromycin — were discovered from soil bacteria, which produce variations of these substances to compete with other microorganisms.
By looking in the gut microbiome, the researchers hoped to identify peptides that the trillions of microbes use against each other in the fight for limited resources — ideally, peptides that wouldn’t broadly kill off the entire microbiome.
Kill the Bad, Spare the Good
Many traditional antibiotics are small molecules. This means they can wipe out the good bacteria in your body, and because each targets a specific bacterial function, bad bacteria can become resistant to them.
Peptide antibiotics, on the other hand, don’t diffuse into the whole body. If taken orally, they stay in the gut; if taken intravenously, they generally stay in the blood. And because of how they kill bacteria, targeting the membrane, they’re also less prone to bacterial resistance.
The microbiome is like a big reservoir of pathogens, said Ami Bhatt, MD, PhD, hematologist at Stanford University in California and one of the study’s authors. Because many antibiotics kill healthy gut bacteria, “what you have left over,” Dr. Bhatt said, “is this big open niche that gets filled up with multidrug-resistant organisms like E coli [Escherichia coli] or vancomycin-resistant Enterococcus.”
Dr. Bhatt has seen cancer patients undergo successful treatment only to die of a multidrug-resistant infection, because current antibiotics fail against those pathogens. “That’s like winning the battle to lose the war.”
By investigating the microbiome, “we wanted to see if we could identify antimicrobial peptides that might spare key members of our regular microbiome, so that we wouldn’t totally disrupt the microbiome the way we do when we use broad-spectrum, small molecule–based antibiotics,” Dr. Bhatt said.
The researchers used artificial intelligence to sift through 400,000 proteins to predict, based on known antibiotics, which peptide sequences might have antimicrobial properties. From the results, they chose 78 peptides to synthesize and test.
“The application of computational approaches combined with experimental validation is very powerful and exciting,” said Jennifer Geddes-McAlister, PhD, professor of cell biology at the University of Guelph in Ontario, Canada, who was not involved in the study. “The study is robust in its approach to microbiome sampling.”
The Long Journey from Lab to Clinic
More than half of the peptides the team tested effectively inhibited the growth of harmful bacteria, and prevotellin-2 (derived from the bacteria Prevotella copri)stood out as the most powerful.
“The study validates experimental data from the lab using animal models, which moves discoveries closer to the clinic,” said Dr. Geddes-McAlister. “Further testing with clinical trials is needed, but the potential for clinical application is promising.”
Unfortunately, that’s not likely to happen anytime soon, said Dr. de la Fuente. “There is not enough economic incentive” for companies to develop new antibiotics. Ten years is his most hopeful guess for when we might see prevotellin-2, or a similar antibiotic, complete clinical trials.
A version of this article first appeared on Medscape.com.
Scientists at Stanford University and the University of Pennsylvania have discovered a new antibiotic candidate in a surprising place: the human gut.
In mice, the antibiotic — a peptide known as prevotellin-2 — showed antimicrobial potency on par with polymyxin B, an antibiotic medication used to treat multidrug-resistant infections. Meanwhile, the peptide mainly left commensal, or beneficial, bacteria alone. The study, published in Cell, also identified several other potent antibiotic peptides with the potential to combat antimicrobial-resistant infections.
The research is part of a larger quest to find new antibiotics that can fight drug-resistant infections, a critical public health threat with more than 2.8 million cases and 35,000 deaths annually in the United States. That quest is urgent, said study author César de la Fuente, PhD, professor of bioengineering at the University of Pennsylvania, Philadelphia.
“The main pillars that have enabled us to almost double our lifespan in the last 100 years or so have been antibiotics, vaccines, and clean water,” said Dr. de la Fuente. “Imagine taking out one of those. I think it would be pretty dramatic.” (Dr. De la Fuente’s lab has become known for finding antibiotic candidates in unusual places, like ancient genetic information of Neanderthals and woolly mammoths.)
The first widely used antibiotic, penicillin, was discovered in 1928, when a physician studying Staphylococcus bacteria returned to his lab after summer break to find mold growing in one of his petri dishes. But many other antibiotics — like streptomycin, tetracycline, and erythromycin — were discovered from soil bacteria, which produce variations of these substances to compete with other microorganisms.
By looking in the gut microbiome, the researchers hoped to identify peptides that the trillions of microbes use against each other in the fight for limited resources — ideally, peptides that wouldn’t broadly kill off the entire microbiome.
Kill the Bad, Spare the Good
Many traditional antibiotics are small molecules. This means they can wipe out the good bacteria in your body, and because each targets a specific bacterial function, bad bacteria can become resistant to them.
Peptide antibiotics, on the other hand, don’t diffuse into the whole body. If taken orally, they stay in the gut; if taken intravenously, they generally stay in the blood. And because of how they kill bacteria, targeting the membrane, they’re also less prone to bacterial resistance.
The microbiome is like a big reservoir of pathogens, said Ami Bhatt, MD, PhD, hematologist at Stanford University in California and one of the study’s authors. Because many antibiotics kill healthy gut bacteria, “what you have left over,” Dr. Bhatt said, “is this big open niche that gets filled up with multidrug-resistant organisms like E coli [Escherichia coli] or vancomycin-resistant Enterococcus.”
Dr. Bhatt has seen cancer patients undergo successful treatment only to die of a multidrug-resistant infection, because current antibiotics fail against those pathogens. “That’s like winning the battle to lose the war.”
By investigating the microbiome, “we wanted to see if we could identify antimicrobial peptides that might spare key members of our regular microbiome, so that we wouldn’t totally disrupt the microbiome the way we do when we use broad-spectrum, small molecule–based antibiotics,” Dr. Bhatt said.
The researchers used artificial intelligence to sift through 400,000 proteins to predict, based on known antibiotics, which peptide sequences might have antimicrobial properties. From the results, they chose 78 peptides to synthesize and test.
“The application of computational approaches combined with experimental validation is very powerful and exciting,” said Jennifer Geddes-McAlister, PhD, professor of cell biology at the University of Guelph in Ontario, Canada, who was not involved in the study. “The study is robust in its approach to microbiome sampling.”
The Long Journey from Lab to Clinic
More than half of the peptides the team tested effectively inhibited the growth of harmful bacteria, and prevotellin-2 (derived from the bacteria Prevotella copri)stood out as the most powerful.
“The study validates experimental data from the lab using animal models, which moves discoveries closer to the clinic,” said Dr. Geddes-McAlister. “Further testing with clinical trials is needed, but the potential for clinical application is promising.”
Unfortunately, that’s not likely to happen anytime soon, said Dr. de la Fuente. “There is not enough economic incentive” for companies to develop new antibiotics. Ten years is his most hopeful guess for when we might see prevotellin-2, or a similar antibiotic, complete clinical trials.
A version of this article first appeared on Medscape.com.
FROM CELL
What Is a Blue Zone Certified Clinician?
It is a great day when a patient shows up at clinical appointment already motivated to make lifestyle behavior changes. Often, they have been inspired by health information they consumed elsewhere, such as from a book, movie, documentary, TV show, a friend, or something out in the community.
Currently, one of the more public representations of health and longevity promotion is Blue Zones. The organization, named for specific areas of the world — the so-called blue zones, where people experience less disease and live longer lives — has created considerable public awareness for healthy living. Today, there are more than 75 Blue Zones Project communities across the United States, where community leaders, businesses, organizations, and citizens collaborate to make healthier choices the easier choices. A recent Netflix special, Live to 100: Secrets of the Blue Zones, further propelled blue zones into the public consciousness.
The Blue Zones emphasis on “plant-slant” diet, natural movement, purpose and contribution, downshifting, and family and community intersect with the lifestyle medicine pillars of whole-food, plant-predominant eating patterns, regular physical activity, stress management, restorative sleep, and positive social connections. Both Blue Zones and lifestyle medicine share a goal of creating healthier and stronger individuals and communities.
For those reasons, it made perfect sense that Blue Zones and the American College of Lifestyle Medicine (ACLM) recently announced a partnership to synergize both organizations’ strengths and resources. Among other things, the collaboration will establish a new certification status of Blue Zones–Certified Physician or Blue Zones–Certified Healthcare Professional, available in 2025 exclusively to clinicians who already are or become certified in lifestyle medicine.
Because of Blue Zones’ considerable consumer awareness, physicians and other health professionals who earn the certification will stand out to potential patients as clinicians with the training and knowledge to help them make sustainable lifestyle behavior changes. A challenging part of any clinician’s job is educating and convincing patients on the proven health benefits of lifestyle behavior change within the time restraints of a routine clinical visit. Patients familiar with Blue Zones are more likely to arrive already interested in changing lifestyle behavior, and clinicians should have the skills to help them achieve their goals.
In addition, community infrastructure developed through Blue Zones that supports healthful lifestyle choices is significant for patients. Lack of resources in their home, work, and community environments is a common obstacle that patients cite when discussing lifestyle change with a clinician. Bicycle lanes for commuting, parks with exercise equipment, accessible healthy food options, and community events to facilitate positive social connections enhance lifestyle-medicine prescriptions. Workplaces, restaurants, places of worship, and grocery stores are examples of community stakeholders that collaborate in Blue Zones communities to promote healthy lifestyle decisions. Although lifestyle medicine clinicians can and do identify creative ways to support patients in communities without strong healthy choice infrastructure, the Blue Zones road map is a welcome companion.
The timing is right for this synthesis of Blue Zones and lifestyle medicine. As consumer interest in Blue Zones has risen, so has clinician interest in evidence-based lifestyle medicine. Since certification in lifestyle medicine began in 2017, almost 6700 physicians and other health professionals have become certified worldwide. More than 43,000 health care professionals have registered for ACLM’s complimentary lifestyle and food-as-medicine courses highlighted by the White House Conference on Hunger, Nutrition, and Health.
What if more patients came to us motivated to make lifestyle changes because of awareness infused in their work and supported in their surrounding community? Matching lifestyle medicine certification with Blue Zone communities equips clinicians to help these patients achieve what they really want: to live longer and better.
Dr. Collings is Director of Lifestyle Medicine, Silicon Valley Medical Development, and Past President, American College of Lifestyle Medicine, Mountain View, California. She has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
It is a great day when a patient shows up at clinical appointment already motivated to make lifestyle behavior changes. Often, they have been inspired by health information they consumed elsewhere, such as from a book, movie, documentary, TV show, a friend, or something out in the community.
Currently, one of the more public representations of health and longevity promotion is Blue Zones. The organization, named for specific areas of the world — the so-called blue zones, where people experience less disease and live longer lives — has created considerable public awareness for healthy living. Today, there are more than 75 Blue Zones Project communities across the United States, where community leaders, businesses, organizations, and citizens collaborate to make healthier choices the easier choices. A recent Netflix special, Live to 100: Secrets of the Blue Zones, further propelled blue zones into the public consciousness.
The Blue Zones emphasis on “plant-slant” diet, natural movement, purpose and contribution, downshifting, and family and community intersect with the lifestyle medicine pillars of whole-food, plant-predominant eating patterns, regular physical activity, stress management, restorative sleep, and positive social connections. Both Blue Zones and lifestyle medicine share a goal of creating healthier and stronger individuals and communities.
For those reasons, it made perfect sense that Blue Zones and the American College of Lifestyle Medicine (ACLM) recently announced a partnership to synergize both organizations’ strengths and resources. Among other things, the collaboration will establish a new certification status of Blue Zones–Certified Physician or Blue Zones–Certified Healthcare Professional, available in 2025 exclusively to clinicians who already are or become certified in lifestyle medicine.
Because of Blue Zones’ considerable consumer awareness, physicians and other health professionals who earn the certification will stand out to potential patients as clinicians with the training and knowledge to help them make sustainable lifestyle behavior changes. A challenging part of any clinician’s job is educating and convincing patients on the proven health benefits of lifestyle behavior change within the time restraints of a routine clinical visit. Patients familiar with Blue Zones are more likely to arrive already interested in changing lifestyle behavior, and clinicians should have the skills to help them achieve their goals.
In addition, community infrastructure developed through Blue Zones that supports healthful lifestyle choices is significant for patients. Lack of resources in their home, work, and community environments is a common obstacle that patients cite when discussing lifestyle change with a clinician. Bicycle lanes for commuting, parks with exercise equipment, accessible healthy food options, and community events to facilitate positive social connections enhance lifestyle-medicine prescriptions. Workplaces, restaurants, places of worship, and grocery stores are examples of community stakeholders that collaborate in Blue Zones communities to promote healthy lifestyle decisions. Although lifestyle medicine clinicians can and do identify creative ways to support patients in communities without strong healthy choice infrastructure, the Blue Zones road map is a welcome companion.
The timing is right for this synthesis of Blue Zones and lifestyle medicine. As consumer interest in Blue Zones has risen, so has clinician interest in evidence-based lifestyle medicine. Since certification in lifestyle medicine began in 2017, almost 6700 physicians and other health professionals have become certified worldwide. More than 43,000 health care professionals have registered for ACLM’s complimentary lifestyle and food-as-medicine courses highlighted by the White House Conference on Hunger, Nutrition, and Health.
What if more patients came to us motivated to make lifestyle changes because of awareness infused in their work and supported in their surrounding community? Matching lifestyle medicine certification with Blue Zone communities equips clinicians to help these patients achieve what they really want: to live longer and better.
Dr. Collings is Director of Lifestyle Medicine, Silicon Valley Medical Development, and Past President, American College of Lifestyle Medicine, Mountain View, California. She has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
It is a great day when a patient shows up at clinical appointment already motivated to make lifestyle behavior changes. Often, they have been inspired by health information they consumed elsewhere, such as from a book, movie, documentary, TV show, a friend, or something out in the community.
Currently, one of the more public representations of health and longevity promotion is Blue Zones. The organization, named for specific areas of the world — the so-called blue zones, where people experience less disease and live longer lives — has created considerable public awareness for healthy living. Today, there are more than 75 Blue Zones Project communities across the United States, where community leaders, businesses, organizations, and citizens collaborate to make healthier choices the easier choices. A recent Netflix special, Live to 100: Secrets of the Blue Zones, further propelled blue zones into the public consciousness.
The Blue Zones emphasis on “plant-slant” diet, natural movement, purpose and contribution, downshifting, and family and community intersect with the lifestyle medicine pillars of whole-food, plant-predominant eating patterns, regular physical activity, stress management, restorative sleep, and positive social connections. Both Blue Zones and lifestyle medicine share a goal of creating healthier and stronger individuals and communities.
For those reasons, it made perfect sense that Blue Zones and the American College of Lifestyle Medicine (ACLM) recently announced a partnership to synergize both organizations’ strengths and resources. Among other things, the collaboration will establish a new certification status of Blue Zones–Certified Physician or Blue Zones–Certified Healthcare Professional, available in 2025 exclusively to clinicians who already are or become certified in lifestyle medicine.
Because of Blue Zones’ considerable consumer awareness, physicians and other health professionals who earn the certification will stand out to potential patients as clinicians with the training and knowledge to help them make sustainable lifestyle behavior changes. A challenging part of any clinician’s job is educating and convincing patients on the proven health benefits of lifestyle behavior change within the time restraints of a routine clinical visit. Patients familiar with Blue Zones are more likely to arrive already interested in changing lifestyle behavior, and clinicians should have the skills to help them achieve their goals.
In addition, community infrastructure developed through Blue Zones that supports healthful lifestyle choices is significant for patients. Lack of resources in their home, work, and community environments is a common obstacle that patients cite when discussing lifestyle change with a clinician. Bicycle lanes for commuting, parks with exercise equipment, accessible healthy food options, and community events to facilitate positive social connections enhance lifestyle-medicine prescriptions. Workplaces, restaurants, places of worship, and grocery stores are examples of community stakeholders that collaborate in Blue Zones communities to promote healthy lifestyle decisions. Although lifestyle medicine clinicians can and do identify creative ways to support patients in communities without strong healthy choice infrastructure, the Blue Zones road map is a welcome companion.
The timing is right for this synthesis of Blue Zones and lifestyle medicine. As consumer interest in Blue Zones has risen, so has clinician interest in evidence-based lifestyle medicine. Since certification in lifestyle medicine began in 2017, almost 6700 physicians and other health professionals have become certified worldwide. More than 43,000 health care professionals have registered for ACLM’s complimentary lifestyle and food-as-medicine courses highlighted by the White House Conference on Hunger, Nutrition, and Health.
What if more patients came to us motivated to make lifestyle changes because of awareness infused in their work and supported in their surrounding community? Matching lifestyle medicine certification with Blue Zone communities equips clinicians to help these patients achieve what they really want: to live longer and better.
Dr. Collings is Director of Lifestyle Medicine, Silicon Valley Medical Development, and Past President, American College of Lifestyle Medicine, Mountain View, California. She has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Celiac Disease: Five Things to Know
Celiac disease is a chronic, immune-mediated, systemic disorder caused by intolerance to gluten — a protein present in rye, barley, and wheat grains — that affects genetically predisposed individuals.
Due to its wide spectrum of clinical manifestations, celiac disease resembles a multisystemic disorder. Its most common gastrointestinal (GI) symptoms include chronic diarrhea, weight loss, and abdominal distention. However, celiac disease can also manifest in myriad extraintestinal symptoms, ranging from headache and fatigue to delayed puberty and psychiatric disorders, with differing presentations in children and adults.
To date, the only treatment is adopting a gluten-free diet (GFD). Although key to preventing persistent villous atrophy, the main cause of complications in celiac disease, lifelong adherence to GFD is challenging and may not resolve all clinical issues. These shortcomings have driven recent efforts to develop novel therapeutic options for patients with this disease.
Here are five things to know about celiac disease.
1. Rising Prevalence of Celiac Disease and Other Autoimmune Disorders Suggests Environmental Factors May Be at Play
Gluten was first identified as the cause of celiac disease in the 1950s. At that time, the condition was thought to be a relatively rare GI disease of childhood that primarily affected people of European descent, but it is now known to be a common disease affecting those of various ages, races, and ethnicities.
A 2018 meta-analysis found the pooled global prevalence of celiac disease was 1.4%. Incidence has increased by as much as 7.5% annually over the past several decades.
Increased awareness among clinicians and improved detection likely play a role in the trend. However, the growth in celiac disease is consistent with that seen for other autoimmune disorders, according to a 2024 update of evidence surrounding celiac disease. Shared environmental factors have been proposed as triggers for celiac disease and other autoimmune diseases and appear to be influencing their rise, the authors noted. These factors include migration and population growth, changing dietary patterns and food processing practices, and altered wheat consumption.
2. No-Biopsy Diagnosis Is Accepted for Children and Shows Promise for Adults
It is estimated that almost 60 million people worldwide have celiac disease, but most remain undiagnosed or misdiagnosed, or they experience significant diagnostic delays.
Prospective data indicate that children with first-degree relatives with celiac disease are at a significantly higher risk of developing the condition, which should prompt screening efforts in this population.
The 2023 updated guidelines from the American College of Gastroenterology (ACG) state that serology testing plays a central role in screening. This commonly involves serological testing for positive serological markers of the disease, including immunoglobulin A (IgA), anti-tissue transglutaminase IgA (tTG-IgA), anti-deamidated gliadin peptide, or endomysial antibodies.
To confirm diagnosis, clinicians have relied on intestinal biopsy since the late 1950s. The ACG still recommends esophagogastroduodenoscopy with multiple duodenal biopsies for confirmation of diagnosis in both children and adults with suspicion of celiac disease. However, recent years have seen a shift toward a no-biopsy approach.
For more than a decade in Europe, a no-biopsy approach has been established practice in pediatric patients, for whom the burden of obtaining a histological confirmation is understandably greater. Most guidelines now permit children to be diagnosed with celiac disease in the absence of a biopsy under specific circumstances (eg, characteristic symptoms of celiac disease and tTG-IgA levels > 10 times the upper limit of normal). The ACG guidelines state that “this approach is a reasonable alternative to the standard approach to a [celiac disease] diagnosis in selected children.”
The ACG does not recommend a no-biopsy approach in adults, noting that, in comparison with children, there is a relative lack of data indicating that serology is predictive in this population. However, it does recognize that physicians may encounter patients for whom a biopsy diagnosis may not be safe or practical. In such cases, an “after-the-fact” diagnosis of likely celiac disease can be given to symptomatic adult patients with a ≥ 10-fold elevation of tTG-IgA and a positive endomysial antibody in a second blood sample.
A 2024 meta-analysis of 18 studies involving over 12,103 adult patients from 15 countries concluded that a no-biopsy approach using tTG-IgA antibody levels ≥ 10 times the upper limit of normal was highly specific and predictive of celiac disease.
3. Celiac Disease Is Associated With Several Life-Threatening Conditions
Emerging data indicate that gastroenterologists should be vigilant in screening patients with celiac disease for several other GI conditions.
Inflammatory bowel disease and celiac disease have a strong bidirectional association, suggesting a possible genetic link between the conditions and indicating that physicians should consider the alternate diagnosis when symptoms persist after treatment.
Given the hypervigilance around food and diet inherent to celiac disease, patients are at an increased risk of developing avoidant/restrictive food intake disorder, according to a 2022 retrospective study.
In 2023, Italian investigators showed that children with celiac disease have an elevated prevalence of functional GI disorders even after adopting a GFD for a year, regardless of whether they consumed processed or natural foods. It was unclear whether this was due to a chronic inflammatory process or to nutritional factors.
Complications resulting from celiac disease are not limited to GI disorders. For a variety of underlying pathophysiological reasons, including intestinal permeability, hyposplenism, and malabsorption of nutrients, patients with celiac disease may be at a higher risk for non-GI conditions, such as osteopenia, women’s health disorders (eg, ovarian failure, endometriosis, or pregnancy loss), juvenile idiopathic arthritis in children and rheumatoid arthritis in adults, certain forms of cancer, infectious diseases, and cardiomyopathy.
4. GFD Is the Only Treatment, but It’s Imperfect and Frustrating for Patients
GFD is the only treatment for celiac disease and must be adhered to without deviation throughout a patient’s life.
Maintaining unwavering adherence reaps considerable benefits: Improved clinical symptoms, robust mucosal healing, and normalization of serological markers. Yet it also takes a considerable toll on patients. Patients with celiac disease struggle with a host of negative physical, psychological, and social impacts. They also report a higher treatment burden than those with gastroesophageal reflux disease or hypertension, and comparable with end-stage renal disease.
GFD also poses financial challenges. Although the price of gluten-free products has decreased in recent years, they still cost significantly more than items with gluten.
Adherence to GFD does not always equate to complete mucosal recovery. While mucosal recovery is achieved in 95% of children within 2 years of the diet’s adoption, only 34% and 66% of adults obtain it within 2 and 5 years, respectively.
GFD may lead to nutrient imbalances because gluten-free foods are typically low in alimentary fiber, micronutrients (eg, vitamin D, vitamin B12, or folate), and minerals (eg, iron, zinc, magnesium, or calcium). With higher sugar and fat content, GFD may leave patients susceptible to unwanted weight gain.
The pervasiveness of gluten in the food production system makes the risk for cross-contamination high. Gluten is often found in both naturally gluten-free foods and products labeled as such. Gluten-sensing technologies, some of which can be used via smartphone apps, have been developed to help patients identify possible cross-contamination. However, the ACG guidelines recommend against the use of these technologies until there is sufficient evidence supporting their ability to improve adherence and clinical outcomes.
5. Novel Therapies for Celiac Disease Are in the Pipeline
The limitations of GFD as the standard treatment for celiac disease have led to an increased focus on developing novel therapeutic interventions. They can be sorted into five key categories: Modulation of the immunostimulatory effects of toxic gluten peptides, elimination of toxic gluten peptides before they reach the intestine, induction of gluten tolerance, modulation of intestinal permeability, and restoration of gut microbiota balance.
Three therapies designed to block antigen presentation by HLA-DQ2/8, the gene alleles that predispose people to celiac disease, show promise: TPM502, an agent that contains three gluten-specific antigenic peptides with overlapping T-cell epitopes for the HLA-DQ2.5 gene; KAN-101, designed to induce gluten tolerance by targeting receptors on the liver; and DONQ52, a multi-specific antibody that targets HLA-DQ2. The KAN-101 therapy received Fast Track designation by the US Food and Drug Administration in 2022.
These and several other agents in clinical and preclinical development are discussed in detail in a 2024 review article. Although no therapies have reached phase 3 testing, when they do, it will undoubtedly be welcomed by those with celiac disease.
A version of this article first appeared on Medscape.com.
Celiac disease is a chronic, immune-mediated, systemic disorder caused by intolerance to gluten — a protein present in rye, barley, and wheat grains — that affects genetically predisposed individuals.
Due to its wide spectrum of clinical manifestations, celiac disease resembles a multisystemic disorder. Its most common gastrointestinal (GI) symptoms include chronic diarrhea, weight loss, and abdominal distention. However, celiac disease can also manifest in myriad extraintestinal symptoms, ranging from headache and fatigue to delayed puberty and psychiatric disorders, with differing presentations in children and adults.
To date, the only treatment is adopting a gluten-free diet (GFD). Although key to preventing persistent villous atrophy, the main cause of complications in celiac disease, lifelong adherence to GFD is challenging and may not resolve all clinical issues. These shortcomings have driven recent efforts to develop novel therapeutic options for patients with this disease.
Here are five things to know about celiac disease.
1. Rising Prevalence of Celiac Disease and Other Autoimmune Disorders Suggests Environmental Factors May Be at Play
Gluten was first identified as the cause of celiac disease in the 1950s. At that time, the condition was thought to be a relatively rare GI disease of childhood that primarily affected people of European descent, but it is now known to be a common disease affecting those of various ages, races, and ethnicities.
A 2018 meta-analysis found the pooled global prevalence of celiac disease was 1.4%. Incidence has increased by as much as 7.5% annually over the past several decades.
Increased awareness among clinicians and improved detection likely play a role in the trend. However, the growth in celiac disease is consistent with that seen for other autoimmune disorders, according to a 2024 update of evidence surrounding celiac disease. Shared environmental factors have been proposed as triggers for celiac disease and other autoimmune diseases and appear to be influencing their rise, the authors noted. These factors include migration and population growth, changing dietary patterns and food processing practices, and altered wheat consumption.
2. No-Biopsy Diagnosis Is Accepted for Children and Shows Promise for Adults
It is estimated that almost 60 million people worldwide have celiac disease, but most remain undiagnosed or misdiagnosed, or they experience significant diagnostic delays.
Prospective data indicate that children with first-degree relatives with celiac disease are at a significantly higher risk of developing the condition, which should prompt screening efforts in this population.
The 2023 updated guidelines from the American College of Gastroenterology (ACG) state that serology testing plays a central role in screening. This commonly involves serological testing for positive serological markers of the disease, including immunoglobulin A (IgA), anti-tissue transglutaminase IgA (tTG-IgA), anti-deamidated gliadin peptide, or endomysial antibodies.
To confirm diagnosis, clinicians have relied on intestinal biopsy since the late 1950s. The ACG still recommends esophagogastroduodenoscopy with multiple duodenal biopsies for confirmation of diagnosis in both children and adults with suspicion of celiac disease. However, recent years have seen a shift toward a no-biopsy approach.
For more than a decade in Europe, a no-biopsy approach has been established practice in pediatric patients, for whom the burden of obtaining a histological confirmation is understandably greater. Most guidelines now permit children to be diagnosed with celiac disease in the absence of a biopsy under specific circumstances (eg, characteristic symptoms of celiac disease and tTG-IgA levels > 10 times the upper limit of normal). The ACG guidelines state that “this approach is a reasonable alternative to the standard approach to a [celiac disease] diagnosis in selected children.”
The ACG does not recommend a no-biopsy approach in adults, noting that, in comparison with children, there is a relative lack of data indicating that serology is predictive in this population. However, it does recognize that physicians may encounter patients for whom a biopsy diagnosis may not be safe or practical. In such cases, an “after-the-fact” diagnosis of likely celiac disease can be given to symptomatic adult patients with a ≥ 10-fold elevation of tTG-IgA and a positive endomysial antibody in a second blood sample.
A 2024 meta-analysis of 18 studies involving over 12,103 adult patients from 15 countries concluded that a no-biopsy approach using tTG-IgA antibody levels ≥ 10 times the upper limit of normal was highly specific and predictive of celiac disease.
3. Celiac Disease Is Associated With Several Life-Threatening Conditions
Emerging data indicate that gastroenterologists should be vigilant in screening patients with celiac disease for several other GI conditions.
Inflammatory bowel disease and celiac disease have a strong bidirectional association, suggesting a possible genetic link between the conditions and indicating that physicians should consider the alternate diagnosis when symptoms persist after treatment.
Given the hypervigilance around food and diet inherent to celiac disease, patients are at an increased risk of developing avoidant/restrictive food intake disorder, according to a 2022 retrospective study.
In 2023, Italian investigators showed that children with celiac disease have an elevated prevalence of functional GI disorders even after adopting a GFD for a year, regardless of whether they consumed processed or natural foods. It was unclear whether this was due to a chronic inflammatory process or to nutritional factors.
Complications resulting from celiac disease are not limited to GI disorders. For a variety of underlying pathophysiological reasons, including intestinal permeability, hyposplenism, and malabsorption of nutrients, patients with celiac disease may be at a higher risk for non-GI conditions, such as osteopenia, women’s health disorders (eg, ovarian failure, endometriosis, or pregnancy loss), juvenile idiopathic arthritis in children and rheumatoid arthritis in adults, certain forms of cancer, infectious diseases, and cardiomyopathy.
4. GFD Is the Only Treatment, but It’s Imperfect and Frustrating for Patients
GFD is the only treatment for celiac disease and must be adhered to without deviation throughout a patient’s life.
Maintaining unwavering adherence reaps considerable benefits: Improved clinical symptoms, robust mucosal healing, and normalization of serological markers. Yet it also takes a considerable toll on patients. Patients with celiac disease struggle with a host of negative physical, psychological, and social impacts. They also report a higher treatment burden than those with gastroesophageal reflux disease or hypertension, and comparable with end-stage renal disease.
GFD also poses financial challenges. Although the price of gluten-free products has decreased in recent years, they still cost significantly more than items with gluten.
Adherence to GFD does not always equate to complete mucosal recovery. While mucosal recovery is achieved in 95% of children within 2 years of the diet’s adoption, only 34% and 66% of adults obtain it within 2 and 5 years, respectively.
GFD may lead to nutrient imbalances because gluten-free foods are typically low in alimentary fiber, micronutrients (eg, vitamin D, vitamin B12, or folate), and minerals (eg, iron, zinc, magnesium, or calcium). With higher sugar and fat content, GFD may leave patients susceptible to unwanted weight gain.
The pervasiveness of gluten in the food production system makes the risk for cross-contamination high. Gluten is often found in both naturally gluten-free foods and products labeled as such. Gluten-sensing technologies, some of which can be used via smartphone apps, have been developed to help patients identify possible cross-contamination. However, the ACG guidelines recommend against the use of these technologies until there is sufficient evidence supporting their ability to improve adherence and clinical outcomes.
5. Novel Therapies for Celiac Disease Are in the Pipeline
The limitations of GFD as the standard treatment for celiac disease have led to an increased focus on developing novel therapeutic interventions. They can be sorted into five key categories: Modulation of the immunostimulatory effects of toxic gluten peptides, elimination of toxic gluten peptides before they reach the intestine, induction of gluten tolerance, modulation of intestinal permeability, and restoration of gut microbiota balance.
Three therapies designed to block antigen presentation by HLA-DQ2/8, the gene alleles that predispose people to celiac disease, show promise: TPM502, an agent that contains three gluten-specific antigenic peptides with overlapping T-cell epitopes for the HLA-DQ2.5 gene; KAN-101, designed to induce gluten tolerance by targeting receptors on the liver; and DONQ52, a multi-specific antibody that targets HLA-DQ2. The KAN-101 therapy received Fast Track designation by the US Food and Drug Administration in 2022.
These and several other agents in clinical and preclinical development are discussed in detail in a 2024 review article. Although no therapies have reached phase 3 testing, when they do, it will undoubtedly be welcomed by those with celiac disease.
A version of this article first appeared on Medscape.com.
Celiac disease is a chronic, immune-mediated, systemic disorder caused by intolerance to gluten — a protein present in rye, barley, and wheat grains — that affects genetically predisposed individuals.
Due to its wide spectrum of clinical manifestations, celiac disease resembles a multisystemic disorder. Its most common gastrointestinal (GI) symptoms include chronic diarrhea, weight loss, and abdominal distention. However, celiac disease can also manifest in myriad extraintestinal symptoms, ranging from headache and fatigue to delayed puberty and psychiatric disorders, with differing presentations in children and adults.
To date, the only treatment is adopting a gluten-free diet (GFD). Although key to preventing persistent villous atrophy, the main cause of complications in celiac disease, lifelong adherence to GFD is challenging and may not resolve all clinical issues. These shortcomings have driven recent efforts to develop novel therapeutic options for patients with this disease.
Here are five things to know about celiac disease.
1. Rising Prevalence of Celiac Disease and Other Autoimmune Disorders Suggests Environmental Factors May Be at Play
Gluten was first identified as the cause of celiac disease in the 1950s. At that time, the condition was thought to be a relatively rare GI disease of childhood that primarily affected people of European descent, but it is now known to be a common disease affecting those of various ages, races, and ethnicities.
A 2018 meta-analysis found the pooled global prevalence of celiac disease was 1.4%. Incidence has increased by as much as 7.5% annually over the past several decades.
Increased awareness among clinicians and improved detection likely play a role in the trend. However, the growth in celiac disease is consistent with that seen for other autoimmune disorders, according to a 2024 update of evidence surrounding celiac disease. Shared environmental factors have been proposed as triggers for celiac disease and other autoimmune diseases and appear to be influencing their rise, the authors noted. These factors include migration and population growth, changing dietary patterns and food processing practices, and altered wheat consumption.
2. No-Biopsy Diagnosis Is Accepted for Children and Shows Promise for Adults
It is estimated that almost 60 million people worldwide have celiac disease, but most remain undiagnosed or misdiagnosed, or they experience significant diagnostic delays.
Prospective data indicate that children with first-degree relatives with celiac disease are at a significantly higher risk of developing the condition, which should prompt screening efforts in this population.
The 2023 updated guidelines from the American College of Gastroenterology (ACG) state that serology testing plays a central role in screening. This commonly involves serological testing for positive serological markers of the disease, including immunoglobulin A (IgA), anti-tissue transglutaminase IgA (tTG-IgA), anti-deamidated gliadin peptide, or endomysial antibodies.
To confirm diagnosis, clinicians have relied on intestinal biopsy since the late 1950s. The ACG still recommends esophagogastroduodenoscopy with multiple duodenal biopsies for confirmation of diagnosis in both children and adults with suspicion of celiac disease. However, recent years have seen a shift toward a no-biopsy approach.
For more than a decade in Europe, a no-biopsy approach has been established practice in pediatric patients, for whom the burden of obtaining a histological confirmation is understandably greater. Most guidelines now permit children to be diagnosed with celiac disease in the absence of a biopsy under specific circumstances (eg, characteristic symptoms of celiac disease and tTG-IgA levels > 10 times the upper limit of normal). The ACG guidelines state that “this approach is a reasonable alternative to the standard approach to a [celiac disease] diagnosis in selected children.”
The ACG does not recommend a no-biopsy approach in adults, noting that, in comparison with children, there is a relative lack of data indicating that serology is predictive in this population. However, it does recognize that physicians may encounter patients for whom a biopsy diagnosis may not be safe or practical. In such cases, an “after-the-fact” diagnosis of likely celiac disease can be given to symptomatic adult patients with a ≥ 10-fold elevation of tTG-IgA and a positive endomysial antibody in a second blood sample.
A 2024 meta-analysis of 18 studies involving over 12,103 adult patients from 15 countries concluded that a no-biopsy approach using tTG-IgA antibody levels ≥ 10 times the upper limit of normal was highly specific and predictive of celiac disease.
3. Celiac Disease Is Associated With Several Life-Threatening Conditions
Emerging data indicate that gastroenterologists should be vigilant in screening patients with celiac disease for several other GI conditions.
Inflammatory bowel disease and celiac disease have a strong bidirectional association, suggesting a possible genetic link between the conditions and indicating that physicians should consider the alternate diagnosis when symptoms persist after treatment.
Given the hypervigilance around food and diet inherent to celiac disease, patients are at an increased risk of developing avoidant/restrictive food intake disorder, according to a 2022 retrospective study.
In 2023, Italian investigators showed that children with celiac disease have an elevated prevalence of functional GI disorders even after adopting a GFD for a year, regardless of whether they consumed processed or natural foods. It was unclear whether this was due to a chronic inflammatory process or to nutritional factors.
Complications resulting from celiac disease are not limited to GI disorders. For a variety of underlying pathophysiological reasons, including intestinal permeability, hyposplenism, and malabsorption of nutrients, patients with celiac disease may be at a higher risk for non-GI conditions, such as osteopenia, women’s health disorders (eg, ovarian failure, endometriosis, or pregnancy loss), juvenile idiopathic arthritis in children and rheumatoid arthritis in adults, certain forms of cancer, infectious diseases, and cardiomyopathy.
4. GFD Is the Only Treatment, but It’s Imperfect and Frustrating for Patients
GFD is the only treatment for celiac disease and must be adhered to without deviation throughout a patient’s life.
Maintaining unwavering adherence reaps considerable benefits: Improved clinical symptoms, robust mucosal healing, and normalization of serological markers. Yet it also takes a considerable toll on patients. Patients with celiac disease struggle with a host of negative physical, psychological, and social impacts. They also report a higher treatment burden than those with gastroesophageal reflux disease or hypertension, and comparable with end-stage renal disease.
GFD also poses financial challenges. Although the price of gluten-free products has decreased in recent years, they still cost significantly more than items with gluten.
Adherence to GFD does not always equate to complete mucosal recovery. While mucosal recovery is achieved in 95% of children within 2 years of the diet’s adoption, only 34% and 66% of adults obtain it within 2 and 5 years, respectively.
GFD may lead to nutrient imbalances because gluten-free foods are typically low in alimentary fiber, micronutrients (eg, vitamin D, vitamin B12, or folate), and minerals (eg, iron, zinc, magnesium, or calcium). With higher sugar and fat content, GFD may leave patients susceptible to unwanted weight gain.
The pervasiveness of gluten in the food production system makes the risk for cross-contamination high. Gluten is often found in both naturally gluten-free foods and products labeled as such. Gluten-sensing technologies, some of which can be used via smartphone apps, have been developed to help patients identify possible cross-contamination. However, the ACG guidelines recommend against the use of these technologies until there is sufficient evidence supporting their ability to improve adherence and clinical outcomes.
5. Novel Therapies for Celiac Disease Are in the Pipeline
The limitations of GFD as the standard treatment for celiac disease have led to an increased focus on developing novel therapeutic interventions. They can be sorted into five key categories: Modulation of the immunostimulatory effects of toxic gluten peptides, elimination of toxic gluten peptides before they reach the intestine, induction of gluten tolerance, modulation of intestinal permeability, and restoration of gut microbiota balance.
Three therapies designed to block antigen presentation by HLA-DQ2/8, the gene alleles that predispose people to celiac disease, show promise: TPM502, an agent that contains three gluten-specific antigenic peptides with overlapping T-cell epitopes for the HLA-DQ2.5 gene; KAN-101, designed to induce gluten tolerance by targeting receptors on the liver; and DONQ52, a multi-specific antibody that targets HLA-DQ2. The KAN-101 therapy received Fast Track designation by the US Food and Drug Administration in 2022.
These and several other agents in clinical and preclinical development are discussed in detail in a 2024 review article. Although no therapies have reached phase 3 testing, when they do, it will undoubtedly be welcomed by those with celiac disease.
A version of this article first appeared on Medscape.com.
Late-Night Eaters May Have Increased Risk for Colorectal Cancer
WASHINGTON — , according to the results of research presented at the annual Digestive Disease Week® (DDW).
Investigators in a new study questioned 664 people getting a colonoscopy to screen for cancer, and 42% said they were late eaters. This group was 46% more likely than non–late eaters to have an adenoma found during colonoscopy. An estimated 5% to 10% of them become cancerous over time.
“A lot of other studies are about what we eat but not when we eat,” said Edena Khoshaba, lead investigator and a medical student at Rush University Medical College in Chicago. “The common advice includes not eating red meat, eating more fruits and vegetables — which is great, of course — but we wanted to see if the timing affects us at all.”
Ms. Khoshaba and colleagues found it did. Late eaters were 5.5 times more likely to have three or more tubular adenomas compared to non–late eaters, even after adjusting for what people were eating. Tubular adenomas are the most common type of polyp found in the colon.
So, what’s the possible connection between late eating and the risk for colorectal cancer?
Resetting Your Internal Clock
Eating close to bedtime could be throwing off the body’s circadian rhythm. But in this case, it’s not the central circadian center located in the brain — the one that releases melatonin. Instead, late eating could disrupt the peripheral circadian rhythm, part of which is found in the GI tract. For example, if a person is eating late at night, the brain thinks it is nighttime while the gut thinks it is daytime, Ms. Khoshaba said in an interview.
This is an interesting study, said Amy Bragagnini, MS, RD, spokesperson for the Academy of Nutrition and Dietetics, when asked to comment on the research. “It is true that eating later at night can disrupt your circadian rhythm.”
“In addition, many of my patients have told me that when they do eat later at night, they don’t always make the healthiest food choices,” Ms. Bragagnini said. “Their late-night food choices are generally higher in added sugar and fat. This may cause them to consume far more calories than their body needs.” So, eating late at night can also lead to unwanted weight gain.
An unanswered question is if late eating is connected in any way at all to increasing rates of colorectal cancer seen in younger patients.
This was an observational study, and another possible limitation, Ms. Khoshaba said, is that people were asked to recall their diets over 24 hours, which may not always be accurate.
Some of the organisms in the gut have their own internal clocks that follow a daily rhythm, and what someone eat determines how many different kinds of these organisms are active, Ms. Bragagnini said.
“So, if your late-night eating consists of foods high in sugar and fat, you may be negatively impacting your microbiome.” she said.
The next step for Ms. Khoshaba and colleagues is a study examining the peripheral circadian rhythm, changes in the gut microbiome, and the risk for developing metabolic syndrome. Ms. Khoshaba and Ms. Bragagnini had no relevant disclosures.
A version of this article appeared on Medscape.com.
WASHINGTON — , according to the results of research presented at the annual Digestive Disease Week® (DDW).
Investigators in a new study questioned 664 people getting a colonoscopy to screen for cancer, and 42% said they were late eaters. This group was 46% more likely than non–late eaters to have an adenoma found during colonoscopy. An estimated 5% to 10% of them become cancerous over time.
“A lot of other studies are about what we eat but not when we eat,” said Edena Khoshaba, lead investigator and a medical student at Rush University Medical College in Chicago. “The common advice includes not eating red meat, eating more fruits and vegetables — which is great, of course — but we wanted to see if the timing affects us at all.”
Ms. Khoshaba and colleagues found it did. Late eaters were 5.5 times more likely to have three or more tubular adenomas compared to non–late eaters, even after adjusting for what people were eating. Tubular adenomas are the most common type of polyp found in the colon.
So, what’s the possible connection between late eating and the risk for colorectal cancer?
Resetting Your Internal Clock
Eating close to bedtime could be throwing off the body’s circadian rhythm. But in this case, it’s not the central circadian center located in the brain — the one that releases melatonin. Instead, late eating could disrupt the peripheral circadian rhythm, part of which is found in the GI tract. For example, if a person is eating late at night, the brain thinks it is nighttime while the gut thinks it is daytime, Ms. Khoshaba said in an interview.
This is an interesting study, said Amy Bragagnini, MS, RD, spokesperson for the Academy of Nutrition and Dietetics, when asked to comment on the research. “It is true that eating later at night can disrupt your circadian rhythm.”
“In addition, many of my patients have told me that when they do eat later at night, they don’t always make the healthiest food choices,” Ms. Bragagnini said. “Their late-night food choices are generally higher in added sugar and fat. This may cause them to consume far more calories than their body needs.” So, eating late at night can also lead to unwanted weight gain.
An unanswered question is if late eating is connected in any way at all to increasing rates of colorectal cancer seen in younger patients.
This was an observational study, and another possible limitation, Ms. Khoshaba said, is that people were asked to recall their diets over 24 hours, which may not always be accurate.
Some of the organisms in the gut have their own internal clocks that follow a daily rhythm, and what someone eat determines how many different kinds of these organisms are active, Ms. Bragagnini said.
“So, if your late-night eating consists of foods high in sugar and fat, you may be negatively impacting your microbiome.” she said.
The next step for Ms. Khoshaba and colleagues is a study examining the peripheral circadian rhythm, changes in the gut microbiome, and the risk for developing metabolic syndrome. Ms. Khoshaba and Ms. Bragagnini had no relevant disclosures.
A version of this article appeared on Medscape.com.
WASHINGTON — , according to the results of research presented at the annual Digestive Disease Week® (DDW).
Investigators in a new study questioned 664 people getting a colonoscopy to screen for cancer, and 42% said they were late eaters. This group was 46% more likely than non–late eaters to have an adenoma found during colonoscopy. An estimated 5% to 10% of them become cancerous over time.
“A lot of other studies are about what we eat but not when we eat,” said Edena Khoshaba, lead investigator and a medical student at Rush University Medical College in Chicago. “The common advice includes not eating red meat, eating more fruits and vegetables — which is great, of course — but we wanted to see if the timing affects us at all.”
Ms. Khoshaba and colleagues found it did. Late eaters were 5.5 times more likely to have three or more tubular adenomas compared to non–late eaters, even after adjusting for what people were eating. Tubular adenomas are the most common type of polyp found in the colon.
So, what’s the possible connection between late eating and the risk for colorectal cancer?
Resetting Your Internal Clock
Eating close to bedtime could be throwing off the body’s circadian rhythm. But in this case, it’s not the central circadian center located in the brain — the one that releases melatonin. Instead, late eating could disrupt the peripheral circadian rhythm, part of which is found in the GI tract. For example, if a person is eating late at night, the brain thinks it is nighttime while the gut thinks it is daytime, Ms. Khoshaba said in an interview.
This is an interesting study, said Amy Bragagnini, MS, RD, spokesperson for the Academy of Nutrition and Dietetics, when asked to comment on the research. “It is true that eating later at night can disrupt your circadian rhythm.”
“In addition, many of my patients have told me that when they do eat later at night, they don’t always make the healthiest food choices,” Ms. Bragagnini said. “Their late-night food choices are generally higher in added sugar and fat. This may cause them to consume far more calories than their body needs.” So, eating late at night can also lead to unwanted weight gain.
An unanswered question is if late eating is connected in any way at all to increasing rates of colorectal cancer seen in younger patients.
This was an observational study, and another possible limitation, Ms. Khoshaba said, is that people were asked to recall their diets over 24 hours, which may not always be accurate.
Some of the organisms in the gut have their own internal clocks that follow a daily rhythm, and what someone eat determines how many different kinds of these organisms are active, Ms. Bragagnini said.
“So, if your late-night eating consists of foods high in sugar and fat, you may be negatively impacting your microbiome.” she said.
The next step for Ms. Khoshaba and colleagues is a study examining the peripheral circadian rhythm, changes in the gut microbiome, and the risk for developing metabolic syndrome. Ms. Khoshaba and Ms. Bragagnini had no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM DDW 2024