User login
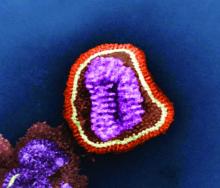
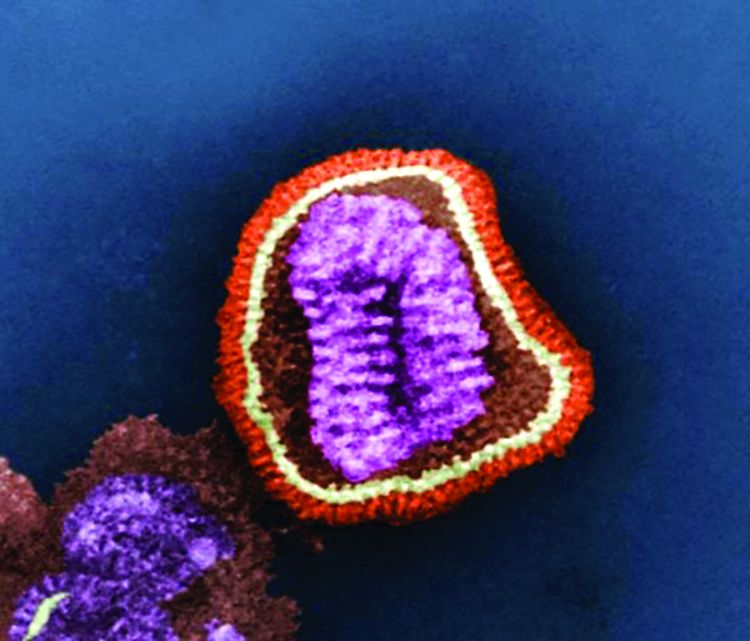
The powerful virus inflammatory response
Inflammation is a double-edged sword. Controlled and modest proinflammatory responses can enhance host immunity against viruses and decrease bacterial colonization and infection, whereas excessive uncontrolled proinflammatory responses may increase the susceptibility to bacterial colonization and secondary infection to facilitate disease pathogenesis. The immune system produces both proinflammatory and anti-inflammatory cytokines and chemokines. It is a balanced response that is key to maintaining good health.
Viral upper respiratory tract infections (URIs) are caused by rhinoviruses, coronaviruses, enteroviruses, respiratory syncytial viruses, influenza A and B viruses, parainfluenza viruses, adenoviruses, and human metapneumoviruses. Viruses are powerful. In the nose, they induce hypersecretion of mucus, slow cilia beating, up-regulate nasal epithelial cell receptors to facilitate bacterial attachment, suppress neutrophil function, and cause increased release of proinflammatory cytokines and chemokines. All these actions by respiratory viruses promote bacterial overgrowth in the nasopharynx and thereby facilitate bacterial superinfections. In fact, progression in pathogenesis of the common bacterial respiratory infections – acute otitis media, acute sinusitis, acute conjunctivitis, and pneumonia – almost always is preceded by a viral URI. Viruses activate multiple target cells in the upper respiratory tract to produce an array of proinflammatory cytokines and chemokines. The symptoms of a viral URI resolve coinciding with an anti-inflammatory response and adaptive immunity.
In recent work, we found a higher frequency of viral URIs in children who experienced more frequent acute otitis media (AOM). We sought to understand why this might occur by comparing levels of inflammatory cytokines/chemokines in the nose during viral URI that did not precipitate AOM versus when a viral URI precipitated an AOM episode. When a child had a viral URI but did not go on to experience an AOM, the child had higher proinflammatory responses than when the viral URI precipitated an AOM. When differences of levels of proinflammatory cytokines/chemokines were compared in otitis-prone and non–otitis-prone children, lower nasal responses were associated with higher otitis-prone classification frequency (Clin Infect Dis. 2018. doi: 10.1093/cid/ciy750).
The powerful virus and the inflammatory response it can induce also play a major role in allergy and asthma. Viral URIs enhance allergic sensitization to respiratory viruses, such as influenza and respiratory syncytial virus, cause cytopathic damage to airway epithelium, promote excessive proinflammatory cytokine/chemokine production, and increase the exposure of allergens and irritants to antigen-presenting cells. Viral infections also may induce the release of epithelial mediators and cytokines that may propagate eosinophilia. Viral URIs, particularly with respiratory syncytial virus and rhinovirus, are the most common causes of wheezing in children, and they have important influences on the development of asthma. Studies have shown that viral infections trigger up to 85% of asthma exacerbations in school-aged children.
Because this column is being published during the winter, a brief discussion of influenza as a powerful virus is appropriate. Influenza occurs in winter outbreaks of varying extent every year. The severity of the influenza season reflects the changing nature of the antigenic properties of influenza viruses, and their spread depends on susceptibility of the population. Influenza outbreaks typically peak over a 2-3 week period and last for 2-3 months. Most outbreaks have attack rates of 10%-20% in children. There may be variations in disease severity caused by different influenza virus types. The symptoms are caused by excessive proinflammatory cytokine/chemokine production in the nose and lung.
Influenza and other viruses can precipitate the systemic inflammatory response syndrome (SIRS), a manifestation of extreme immune dysregulation resulting in organ dysfunction that clinically resembles bacterial sepsis. In this syndrome, tissues remote from the original insult display the cardinal signs of inflammation, including vasodilation, increased microvascular permeability, and leukocyte accumulation. SIRS is another example of the double-edged sword of inflammation.
The onset and progression of SIRS occurs because of dysregulation of the normal inflammatory response, usually with an increase in both proinflammatory and anti-inflammatory cytokines and chemokines, initiating a chain of events that leads to organ failure.
Dr. Pichichero is a specialist in pediatric infectious diseases and director of the Research Institute at Rochester (N.Y.) General Hospital. He reported having no conflicts of interest. Email him at pdnews@mdedge.com.
Inflammation is a double-edged sword. Controlled and modest proinflammatory responses can enhance host immunity against viruses and decrease bacterial colonization and infection, whereas excessive uncontrolled proinflammatory responses may increase the susceptibility to bacterial colonization and secondary infection to facilitate disease pathogenesis. The immune system produces both proinflammatory and anti-inflammatory cytokines and chemokines. It is a balanced response that is key to maintaining good health.
Viral upper respiratory tract infections (URIs) are caused by rhinoviruses, coronaviruses, enteroviruses, respiratory syncytial viruses, influenza A and B viruses, parainfluenza viruses, adenoviruses, and human metapneumoviruses. Viruses are powerful. In the nose, they induce hypersecretion of mucus, slow cilia beating, up-regulate nasal epithelial cell receptors to facilitate bacterial attachment, suppress neutrophil function, and cause increased release of proinflammatory cytokines and chemokines. All these actions by respiratory viruses promote bacterial overgrowth in the nasopharynx and thereby facilitate bacterial superinfections. In fact, progression in pathogenesis of the common bacterial respiratory infections – acute otitis media, acute sinusitis, acute conjunctivitis, and pneumonia – almost always is preceded by a viral URI. Viruses activate multiple target cells in the upper respiratory tract to produce an array of proinflammatory cytokines and chemokines. The symptoms of a viral URI resolve coinciding with an anti-inflammatory response and adaptive immunity.
In recent work, we found a higher frequency of viral URIs in children who experienced more frequent acute otitis media (AOM). We sought to understand why this might occur by comparing levels of inflammatory cytokines/chemokines in the nose during viral URI that did not precipitate AOM versus when a viral URI precipitated an AOM episode. When a child had a viral URI but did not go on to experience an AOM, the child had higher proinflammatory responses than when the viral URI precipitated an AOM. When differences of levels of proinflammatory cytokines/chemokines were compared in otitis-prone and non–otitis-prone children, lower nasal responses were associated with higher otitis-prone classification frequency (Clin Infect Dis. 2018. doi: 10.1093/cid/ciy750).
The powerful virus and the inflammatory response it can induce also play a major role in allergy and asthma. Viral URIs enhance allergic sensitization to respiratory viruses, such as influenza and respiratory syncytial virus, cause cytopathic damage to airway epithelium, promote excessive proinflammatory cytokine/chemokine production, and increase the exposure of allergens and irritants to antigen-presenting cells. Viral infections also may induce the release of epithelial mediators and cytokines that may propagate eosinophilia. Viral URIs, particularly with respiratory syncytial virus and rhinovirus, are the most common causes of wheezing in children, and they have important influences on the development of asthma. Studies have shown that viral infections trigger up to 85% of asthma exacerbations in school-aged children.
Because this column is being published during the winter, a brief discussion of influenza as a powerful virus is appropriate. Influenza occurs in winter outbreaks of varying extent every year. The severity of the influenza season reflects the changing nature of the antigenic properties of influenza viruses, and their spread depends on susceptibility of the population. Influenza outbreaks typically peak over a 2-3 week period and last for 2-3 months. Most outbreaks have attack rates of 10%-20% in children. There may be variations in disease severity caused by different influenza virus types. The symptoms are caused by excessive proinflammatory cytokine/chemokine production in the nose and lung.
Influenza and other viruses can precipitate the systemic inflammatory response syndrome (SIRS), a manifestation of extreme immune dysregulation resulting in organ dysfunction that clinically resembles bacterial sepsis. In this syndrome, tissues remote from the original insult display the cardinal signs of inflammation, including vasodilation, increased microvascular permeability, and leukocyte accumulation. SIRS is another example of the double-edged sword of inflammation.
The onset and progression of SIRS occurs because of dysregulation of the normal inflammatory response, usually with an increase in both proinflammatory and anti-inflammatory cytokines and chemokines, initiating a chain of events that leads to organ failure.
Dr. Pichichero is a specialist in pediatric infectious diseases and director of the Research Institute at Rochester (N.Y.) General Hospital. He reported having no conflicts of interest. Email him at pdnews@mdedge.com.
Inflammation is a double-edged sword. Controlled and modest proinflammatory responses can enhance host immunity against viruses and decrease bacterial colonization and infection, whereas excessive uncontrolled proinflammatory responses may increase the susceptibility to bacterial colonization and secondary infection to facilitate disease pathogenesis. The immune system produces both proinflammatory and anti-inflammatory cytokines and chemokines. It is a balanced response that is key to maintaining good health.
Viral upper respiratory tract infections (URIs) are caused by rhinoviruses, coronaviruses, enteroviruses, respiratory syncytial viruses, influenza A and B viruses, parainfluenza viruses, adenoviruses, and human metapneumoviruses. Viruses are powerful. In the nose, they induce hypersecretion of mucus, slow cilia beating, up-regulate nasal epithelial cell receptors to facilitate bacterial attachment, suppress neutrophil function, and cause increased release of proinflammatory cytokines and chemokines. All these actions by respiratory viruses promote bacterial overgrowth in the nasopharynx and thereby facilitate bacterial superinfections. In fact, progression in pathogenesis of the common bacterial respiratory infections – acute otitis media, acute sinusitis, acute conjunctivitis, and pneumonia – almost always is preceded by a viral URI. Viruses activate multiple target cells in the upper respiratory tract to produce an array of proinflammatory cytokines and chemokines. The symptoms of a viral URI resolve coinciding with an anti-inflammatory response and adaptive immunity.
In recent work, we found a higher frequency of viral URIs in children who experienced more frequent acute otitis media (AOM). We sought to understand why this might occur by comparing levels of inflammatory cytokines/chemokines in the nose during viral URI that did not precipitate AOM versus when a viral URI precipitated an AOM episode. When a child had a viral URI but did not go on to experience an AOM, the child had higher proinflammatory responses than when the viral URI precipitated an AOM. When differences of levels of proinflammatory cytokines/chemokines were compared in otitis-prone and non–otitis-prone children, lower nasal responses were associated with higher otitis-prone classification frequency (Clin Infect Dis. 2018. doi: 10.1093/cid/ciy750).
The powerful virus and the inflammatory response it can induce also play a major role in allergy and asthma. Viral URIs enhance allergic sensitization to respiratory viruses, such as influenza and respiratory syncytial virus, cause cytopathic damage to airway epithelium, promote excessive proinflammatory cytokine/chemokine production, and increase the exposure of allergens and irritants to antigen-presenting cells. Viral infections also may induce the release of epithelial mediators and cytokines that may propagate eosinophilia. Viral URIs, particularly with respiratory syncytial virus and rhinovirus, are the most common causes of wheezing in children, and they have important influences on the development of asthma. Studies have shown that viral infections trigger up to 85% of asthma exacerbations in school-aged children.
Because this column is being published during the winter, a brief discussion of influenza as a powerful virus is appropriate. Influenza occurs in winter outbreaks of varying extent every year. The severity of the influenza season reflects the changing nature of the antigenic properties of influenza viruses, and their spread depends on susceptibility of the population. Influenza outbreaks typically peak over a 2-3 week period and last for 2-3 months. Most outbreaks have attack rates of 10%-20% in children. There may be variations in disease severity caused by different influenza virus types. The symptoms are caused by excessive proinflammatory cytokine/chemokine production in the nose and lung.
Influenza and other viruses can precipitate the systemic inflammatory response syndrome (SIRS), a manifestation of extreme immune dysregulation resulting in organ dysfunction that clinically resembles bacterial sepsis. In this syndrome, tissues remote from the original insult display the cardinal signs of inflammation, including vasodilation, increased microvascular permeability, and leukocyte accumulation. SIRS is another example of the double-edged sword of inflammation.
The onset and progression of SIRS occurs because of dysregulation of the normal inflammatory response, usually with an increase in both proinflammatory and anti-inflammatory cytokines and chemokines, initiating a chain of events that leads to organ failure.
Dr. Pichichero is a specialist in pediatric infectious diseases and director of the Research Institute at Rochester (N.Y.) General Hospital. He reported having no conflicts of interest. Email him at pdnews@mdedge.com.
Full-dose quadrivalent flu vaccine shows increased efficacy in children
according to data from a randomized trial of nearly 2,000 children aged 6-35 months.
Data from previous studies have suggested that a full dose of vaccine may be more immunogenic in young children compared with a half dose, and Sanofi Pasteur has submitted a supplemental Biologics License Application to the Food and Drug Administration to allow use of the full 0.5-mL dose in children as young as 6 months, Monica Mercer, MD, of Sanofi Pasteur, said at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices in Atlanta.
Dr. Mercer presented findings from a phase IV randomized, observer-blinded study, in which the researchers assigned healthy children aged 6-35 months to receive Fluzone quadrivalent vaccine at a dose of 0.25 mL or 0.5 mL.
A total of 1,941 children (949 for the 0.25-mL dose and 992 for the 0.5-mL dose) were included in the safety analysis.
The most important safety outcome was to compare the rate of any fever, Dr. Mercer said at the meeting.
Overall, at 7 days after vaccination, the rate of fever was 11% for the half dose and 12% for the full dose, she said. The resulting difference of 0.84% met the criteria for noninferiority (less than 5%), she added.
In terms of safety, tenderness was the most frequently reported injection site reaction, noted in 47% of the half-dose group and 50% of the full-dose group. The rates of unsolicited adverse events were similar in both groups, the most common included diarrhea and cough, Dr. Mercer said.
No subjects in the full-dose group and three in the half-dose group discontinued the study because of adverse events. The only reported serious adverse event was one case of chronic urticaria in the half-dose group; no deaths were reported in either group.
As for efficacy, the full dose demonstrated noninferiority, compared with the half dose, against each of four strains: influenza A H1N1, influenza A H3N2, influenza B Victoria, and influenza B Yamagata. The geometric mean titers of the full and half doses for each of the four strains were, respectively, 310 and 214, 332 and 221, 348 and 261, and 349 and 243.
The potential action date for the supplemental Biologics License Application is January 2019, noted Dr. Mercer, who is employed by Sanofi Pasteur.
according to data from a randomized trial of nearly 2,000 children aged 6-35 months.
Data from previous studies have suggested that a full dose of vaccine may be more immunogenic in young children compared with a half dose, and Sanofi Pasteur has submitted a supplemental Biologics License Application to the Food and Drug Administration to allow use of the full 0.5-mL dose in children as young as 6 months, Monica Mercer, MD, of Sanofi Pasteur, said at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices in Atlanta.
Dr. Mercer presented findings from a phase IV randomized, observer-blinded study, in which the researchers assigned healthy children aged 6-35 months to receive Fluzone quadrivalent vaccine at a dose of 0.25 mL or 0.5 mL.
A total of 1,941 children (949 for the 0.25-mL dose and 992 for the 0.5-mL dose) were included in the safety analysis.
The most important safety outcome was to compare the rate of any fever, Dr. Mercer said at the meeting.
Overall, at 7 days after vaccination, the rate of fever was 11% for the half dose and 12% for the full dose, she said. The resulting difference of 0.84% met the criteria for noninferiority (less than 5%), she added.
In terms of safety, tenderness was the most frequently reported injection site reaction, noted in 47% of the half-dose group and 50% of the full-dose group. The rates of unsolicited adverse events were similar in both groups, the most common included diarrhea and cough, Dr. Mercer said.
No subjects in the full-dose group and three in the half-dose group discontinued the study because of adverse events. The only reported serious adverse event was one case of chronic urticaria in the half-dose group; no deaths were reported in either group.
As for efficacy, the full dose demonstrated noninferiority, compared with the half dose, against each of four strains: influenza A H1N1, influenza A H3N2, influenza B Victoria, and influenza B Yamagata. The geometric mean titers of the full and half doses for each of the four strains were, respectively, 310 and 214, 332 and 221, 348 and 261, and 349 and 243.
The potential action date for the supplemental Biologics License Application is January 2019, noted Dr. Mercer, who is employed by Sanofi Pasteur.
according to data from a randomized trial of nearly 2,000 children aged 6-35 months.
Data from previous studies have suggested that a full dose of vaccine may be more immunogenic in young children compared with a half dose, and Sanofi Pasteur has submitted a supplemental Biologics License Application to the Food and Drug Administration to allow use of the full 0.5-mL dose in children as young as 6 months, Monica Mercer, MD, of Sanofi Pasteur, said at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices in Atlanta.
Dr. Mercer presented findings from a phase IV randomized, observer-blinded study, in which the researchers assigned healthy children aged 6-35 months to receive Fluzone quadrivalent vaccine at a dose of 0.25 mL or 0.5 mL.
A total of 1,941 children (949 for the 0.25-mL dose and 992 for the 0.5-mL dose) were included in the safety analysis.
The most important safety outcome was to compare the rate of any fever, Dr. Mercer said at the meeting.
Overall, at 7 days after vaccination, the rate of fever was 11% for the half dose and 12% for the full dose, she said. The resulting difference of 0.84% met the criteria for noninferiority (less than 5%), she added.
In terms of safety, tenderness was the most frequently reported injection site reaction, noted in 47% of the half-dose group and 50% of the full-dose group. The rates of unsolicited adverse events were similar in both groups, the most common included diarrhea and cough, Dr. Mercer said.
No subjects in the full-dose group and three in the half-dose group discontinued the study because of adverse events. The only reported serious adverse event was one case of chronic urticaria in the half-dose group; no deaths were reported in either group.
As for efficacy, the full dose demonstrated noninferiority, compared with the half dose, against each of four strains: influenza A H1N1, influenza A H3N2, influenza B Victoria, and influenza B Yamagata. The geometric mean titers of the full and half doses for each of the four strains were, respectively, 310 and 214, 332 and 221, 348 and 261, and 349 and 243.
The potential action date for the supplemental Biologics License Application is January 2019, noted Dr. Mercer, who is employed by Sanofi Pasteur.
REPORTING FROM AN ACIP MEETING
Vaccine protects against flu-related hospitalizations in pregnancy
A review of more than 1,000 hospitalizations revealed a 40% influenza vaccine effectiveness against laboratory-confirmed influenza-associated hospitalizations during pregnancy, Mark Thompson, MD, said at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices in Atlanta.
To date, no study has examined influenza vaccine effectiveness (IVE) against hospitalizations among pregnant women, said Dr. Thompson, of the CDC’s influenza division.
He presented results of a study based on data from the Pregnancy Influenza Vaccine Effectiveness Network (PREVENT), which included public health or health care systems with integrated laboratory, medical, and vaccination records in Australia, Canada (Alberta and Ontario), Israel, and three states (California, Oregon, and Washington). The study included women aged 18-50 years who were pregnant during local influenza seasons from 2010 to 2016. Most of the women were older than 35 years (79%), and in the third trimester (65%), and had no high risk medical conditions (66%). The study was published in Clinical Infectious Diseases (2018 Oct 11. doi: 10.1093/cid/ciy737).
The researchers identified 19,450 hospitalizations with an acute respiratory or febrile illness discharge diagnosis and clinician-ordered real-time reverse transcription polymerase chain reaction (rRT-PCR) testing for flu viruses. Of these, 1,030 (6%) of the women underwent rRT-PCR testing, 54% were diagnosed with either influenza or pneumonia, and 58% had detectable influenza A or B virus infections.
Overall, the adjusted IVE was 40%; 13% of rRT-PCR-confirmed influenza-positive pregnant women and 22% of influenza-negative pregnant women were vaccinated; IVE was adjusted for site, season, season timing, and high-risk medical conditions.
“The takeaway is this is the average performance of the vaccine across multiple countries and different seasons,” and the vaccine effectiveness appeared stable across high-risk medical conditions and trimesters of pregnancy, Dr. Thompson said.
The generalizability of the study findings was limited by the lack of data from low- to middle-income countries, he said during the meeting discussion. However, the ICU admission rate is “what we would expect” and similar to results from previous studies. The consistent results showed the need to increase flu vaccination for pregnant women worldwide and to include study populations from lower-income countries in future research.
Dr. Thompson had no financial conflicts to disclose.
A review of more than 1,000 hospitalizations revealed a 40% influenza vaccine effectiveness against laboratory-confirmed influenza-associated hospitalizations during pregnancy, Mark Thompson, MD, said at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices in Atlanta.
To date, no study has examined influenza vaccine effectiveness (IVE) against hospitalizations among pregnant women, said Dr. Thompson, of the CDC’s influenza division.
He presented results of a study based on data from the Pregnancy Influenza Vaccine Effectiveness Network (PREVENT), which included public health or health care systems with integrated laboratory, medical, and vaccination records in Australia, Canada (Alberta and Ontario), Israel, and three states (California, Oregon, and Washington). The study included women aged 18-50 years who were pregnant during local influenza seasons from 2010 to 2016. Most of the women were older than 35 years (79%), and in the third trimester (65%), and had no high risk medical conditions (66%). The study was published in Clinical Infectious Diseases (2018 Oct 11. doi: 10.1093/cid/ciy737).
The researchers identified 19,450 hospitalizations with an acute respiratory or febrile illness discharge diagnosis and clinician-ordered real-time reverse transcription polymerase chain reaction (rRT-PCR) testing for flu viruses. Of these, 1,030 (6%) of the women underwent rRT-PCR testing, 54% were diagnosed with either influenza or pneumonia, and 58% had detectable influenza A or B virus infections.
Overall, the adjusted IVE was 40%; 13% of rRT-PCR-confirmed influenza-positive pregnant women and 22% of influenza-negative pregnant women were vaccinated; IVE was adjusted for site, season, season timing, and high-risk medical conditions.
“The takeaway is this is the average performance of the vaccine across multiple countries and different seasons,” and the vaccine effectiveness appeared stable across high-risk medical conditions and trimesters of pregnancy, Dr. Thompson said.
The generalizability of the study findings was limited by the lack of data from low- to middle-income countries, he said during the meeting discussion. However, the ICU admission rate is “what we would expect” and similar to results from previous studies. The consistent results showed the need to increase flu vaccination for pregnant women worldwide and to include study populations from lower-income countries in future research.
Dr. Thompson had no financial conflicts to disclose.
A review of more than 1,000 hospitalizations revealed a 40% influenza vaccine effectiveness against laboratory-confirmed influenza-associated hospitalizations during pregnancy, Mark Thompson, MD, said at a meeting of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices in Atlanta.
To date, no study has examined influenza vaccine effectiveness (IVE) against hospitalizations among pregnant women, said Dr. Thompson, of the CDC’s influenza division.
He presented results of a study based on data from the Pregnancy Influenza Vaccine Effectiveness Network (PREVENT), which included public health or health care systems with integrated laboratory, medical, and vaccination records in Australia, Canada (Alberta and Ontario), Israel, and three states (California, Oregon, and Washington). The study included women aged 18-50 years who were pregnant during local influenza seasons from 2010 to 2016. Most of the women were older than 35 years (79%), and in the third trimester (65%), and had no high risk medical conditions (66%). The study was published in Clinical Infectious Diseases (2018 Oct 11. doi: 10.1093/cid/ciy737).
The researchers identified 19,450 hospitalizations with an acute respiratory or febrile illness discharge diagnosis and clinician-ordered real-time reverse transcription polymerase chain reaction (rRT-PCR) testing for flu viruses. Of these, 1,030 (6%) of the women underwent rRT-PCR testing, 54% were diagnosed with either influenza or pneumonia, and 58% had detectable influenza A or B virus infections.
Overall, the adjusted IVE was 40%; 13% of rRT-PCR-confirmed influenza-positive pregnant women and 22% of influenza-negative pregnant women were vaccinated; IVE was adjusted for site, season, season timing, and high-risk medical conditions.
“The takeaway is this is the average performance of the vaccine across multiple countries and different seasons,” and the vaccine effectiveness appeared stable across high-risk medical conditions and trimesters of pregnancy, Dr. Thompson said.
The generalizability of the study findings was limited by the lack of data from low- to middle-income countries, he said during the meeting discussion. However, the ICU admission rate is “what we would expect” and similar to results from previous studies. The consistent results showed the need to increase flu vaccination for pregnant women worldwide and to include study populations from lower-income countries in future research.
Dr. Thompson had no financial conflicts to disclose.
FROM AN ACIP MEETING
FDA clears Abbott’s Influenza A & B 2, Strep A 2 assays
The Food and Drug Administration has cleared Abbott Laboratories’ next-generation Influenza A & B 2 and Strep A 2 molecular assays for point-of-care testing.
The Influenza A & B 2 assay can detect and differentiate influenza A and B in 13 minutes, with a call-out of positive results at 5 minutes. It can be stored at room temperature, simplifying storage and ordering. The Strep A 2 assay detects group A streptococcus bacterial nucleic acid in 6 minutes, with a call-out of positive results at 2 minutes. Both will be the fastest tests currently on the market in their respective fields, according to a corporate press release.
The assays will be available in a variety of inpatient and outpatient settings, particularly in locations where patients commonly access health care services, such as EDs, physician offices, walk-in clinics, and urgent care centers. This will allow health care providers to make a fast, informed diagnosis and provide appropriate treatment within the span of a single patient visit.
“The ability to obtain early call outs for positive test results with molecular accuracy in as little as 5 minutes for influenza and 2 minutes for strep A is a game-changing development that allows prompt treatment decisions at the point of care. Rapid testing may also help reduce improper antibiotic usage, which can occur when treatment is based exclusively on a patient’s symptoms, and contributes to antibiotic resistance,” Gregory J. Berry, PhD, director of molecular diagnostics at Northwell Health Laboratories in Lake Success, N.Y., said in the press release.
Find the full press release on the Abbott Laboratories website.
The Food and Drug Administration has cleared Abbott Laboratories’ next-generation Influenza A & B 2 and Strep A 2 molecular assays for point-of-care testing.
The Influenza A & B 2 assay can detect and differentiate influenza A and B in 13 minutes, with a call-out of positive results at 5 minutes. It can be stored at room temperature, simplifying storage and ordering. The Strep A 2 assay detects group A streptococcus bacterial nucleic acid in 6 minutes, with a call-out of positive results at 2 minutes. Both will be the fastest tests currently on the market in their respective fields, according to a corporate press release.
The assays will be available in a variety of inpatient and outpatient settings, particularly in locations where patients commonly access health care services, such as EDs, physician offices, walk-in clinics, and urgent care centers. This will allow health care providers to make a fast, informed diagnosis and provide appropriate treatment within the span of a single patient visit.
“The ability to obtain early call outs for positive test results with molecular accuracy in as little as 5 minutes for influenza and 2 minutes for strep A is a game-changing development that allows prompt treatment decisions at the point of care. Rapid testing may also help reduce improper antibiotic usage, which can occur when treatment is based exclusively on a patient’s symptoms, and contributes to antibiotic resistance,” Gregory J. Berry, PhD, director of molecular diagnostics at Northwell Health Laboratories in Lake Success, N.Y., said in the press release.
Find the full press release on the Abbott Laboratories website.
The Food and Drug Administration has cleared Abbott Laboratories’ next-generation Influenza A & B 2 and Strep A 2 molecular assays for point-of-care testing.
The Influenza A & B 2 assay can detect and differentiate influenza A and B in 13 minutes, with a call-out of positive results at 5 minutes. It can be stored at room temperature, simplifying storage and ordering. The Strep A 2 assay detects group A streptococcus bacterial nucleic acid in 6 minutes, with a call-out of positive results at 2 minutes. Both will be the fastest tests currently on the market in their respective fields, according to a corporate press release.
The assays will be available in a variety of inpatient and outpatient settings, particularly in locations where patients commonly access health care services, such as EDs, physician offices, walk-in clinics, and urgent care centers. This will allow health care providers to make a fast, informed diagnosis and provide appropriate treatment within the span of a single patient visit.
“The ability to obtain early call outs for positive test results with molecular accuracy in as little as 5 minutes for influenza and 2 minutes for strep A is a game-changing development that allows prompt treatment decisions at the point of care. Rapid testing may also help reduce improper antibiotic usage, which can occur when treatment is based exclusively on a patient’s symptoms, and contributes to antibiotic resistance,” Gregory J. Berry, PhD, director of molecular diagnostics at Northwell Health Laboratories in Lake Success, N.Y., said in the press release.
Find the full press release on the Abbott Laboratories website.
ACIP votes unanimously in favor of immunization schedule update and redesign
Clinicians consulting the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices vaccination schedules for children, adolescents, and adults in 2019 will find a simpler design and more useful product, according to David Kim, MD, of the Immunization Services Division of the Centers for Disease Control and Prevention, Atlanta.
In a single vote to cover both adult and child/adolescent schedules, the committee voted unanimously in favor of a redesign of the schedules and several clinical updates.
In 2016, the working group for vaccination schedules conducted an ad hoc evaluation of the adult schedule to assess its usability, Dr. Kim said at a meeting of the CDC’s ACIP.
The design of the adult schedule was fully evaluated in 2018 via a three-step process – interviews with 48 health care providers, a redesign of the schedule, and a survey after the redesign. Design changes to the child/adolescent schedule were harmonized with the adult schedule, Dr. Kim explained.
The adult vaccination schedule itself includes several updates in ACIP recommendations in addition to the aesthetic design changes.
The 2019 Adult Immunization Schedule includes the option of the live attenuated influenza vaccine (LAIV) for influenza, the addition of homelessness as an indication for hepatitis A vaccination, and the use of CpG-adjuvanted hepatitis B vaccine, Dr. Kim said.
The additions to the 2019 Child and Adolescent Immunization Schedule are the optional use of the LAIV for influenza, the addition of homelessness as an indication for hepatitis A vaccination, the use of CpG-adjuvanted hepatitis B vaccine (a cytosine phosphoguanosine oligodeoxynucleotide adjuvant), and the addition of the Tdap vaccination of individuals who received Tdap at age 7-10 years.
Some of the key design changes include the use of bright purple on the child/adolescent schedule to more easily distinguish it from the adult version, said Dr. Kim.
Other changes to both schedules include shorter titles, lists of vaccines and trade names, and compartmentalized information for easier reference. Figures have been replaced by tables, and footnotes are simply “Notes” at the end of the schedule, compartmentalized for easier reading, he said. In addition, the schedules include resources for vaccination in outbreak situations and a section on how to report vaccine preventable disease outbreaks.
The ACIP committee members had no relevant financial conflicts to disclose.
Clinicians consulting the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices vaccination schedules for children, adolescents, and adults in 2019 will find a simpler design and more useful product, according to David Kim, MD, of the Immunization Services Division of the Centers for Disease Control and Prevention, Atlanta.
In a single vote to cover both adult and child/adolescent schedules, the committee voted unanimously in favor of a redesign of the schedules and several clinical updates.
In 2016, the working group for vaccination schedules conducted an ad hoc evaluation of the adult schedule to assess its usability, Dr. Kim said at a meeting of the CDC’s ACIP.
The design of the adult schedule was fully evaluated in 2018 via a three-step process – interviews with 48 health care providers, a redesign of the schedule, and a survey after the redesign. Design changes to the child/adolescent schedule were harmonized with the adult schedule, Dr. Kim explained.
The adult vaccination schedule itself includes several updates in ACIP recommendations in addition to the aesthetic design changes.
The 2019 Adult Immunization Schedule includes the option of the live attenuated influenza vaccine (LAIV) for influenza, the addition of homelessness as an indication for hepatitis A vaccination, and the use of CpG-adjuvanted hepatitis B vaccine, Dr. Kim said.
The additions to the 2019 Child and Adolescent Immunization Schedule are the optional use of the LAIV for influenza, the addition of homelessness as an indication for hepatitis A vaccination, the use of CpG-adjuvanted hepatitis B vaccine (a cytosine phosphoguanosine oligodeoxynucleotide adjuvant), and the addition of the Tdap vaccination of individuals who received Tdap at age 7-10 years.
Some of the key design changes include the use of bright purple on the child/adolescent schedule to more easily distinguish it from the adult version, said Dr. Kim.
Other changes to both schedules include shorter titles, lists of vaccines and trade names, and compartmentalized information for easier reference. Figures have been replaced by tables, and footnotes are simply “Notes” at the end of the schedule, compartmentalized for easier reading, he said. In addition, the schedules include resources for vaccination in outbreak situations and a section on how to report vaccine preventable disease outbreaks.
The ACIP committee members had no relevant financial conflicts to disclose.
Clinicians consulting the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices vaccination schedules for children, adolescents, and adults in 2019 will find a simpler design and more useful product, according to David Kim, MD, of the Immunization Services Division of the Centers for Disease Control and Prevention, Atlanta.
In a single vote to cover both adult and child/adolescent schedules, the committee voted unanimously in favor of a redesign of the schedules and several clinical updates.
In 2016, the working group for vaccination schedules conducted an ad hoc evaluation of the adult schedule to assess its usability, Dr. Kim said at a meeting of the CDC’s ACIP.
The design of the adult schedule was fully evaluated in 2018 via a three-step process – interviews with 48 health care providers, a redesign of the schedule, and a survey after the redesign. Design changes to the child/adolescent schedule were harmonized with the adult schedule, Dr. Kim explained.
The adult vaccination schedule itself includes several updates in ACIP recommendations in addition to the aesthetic design changes.
The 2019 Adult Immunization Schedule includes the option of the live attenuated influenza vaccine (LAIV) for influenza, the addition of homelessness as an indication for hepatitis A vaccination, and the use of CpG-adjuvanted hepatitis B vaccine, Dr. Kim said.
The additions to the 2019 Child and Adolescent Immunization Schedule are the optional use of the LAIV for influenza, the addition of homelessness as an indication for hepatitis A vaccination, the use of CpG-adjuvanted hepatitis B vaccine (a cytosine phosphoguanosine oligodeoxynucleotide adjuvant), and the addition of the Tdap vaccination of individuals who received Tdap at age 7-10 years.
Some of the key design changes include the use of bright purple on the child/adolescent schedule to more easily distinguish it from the adult version, said Dr. Kim.
Other changes to both schedules include shorter titles, lists of vaccines and trade names, and compartmentalized information for easier reference. Figures have been replaced by tables, and footnotes are simply “Notes” at the end of the schedule, compartmentalized for easier reading, he said. In addition, the schedules include resources for vaccination in outbreak situations and a section on how to report vaccine preventable disease outbreaks.
The ACIP committee members had no relevant financial conflicts to disclose.
AT AN ACIP MEETING
FDA approves Xofluza for treatment of influenza
The Food and Drug Administration has approved Xofluza (baloxavir marboxil) for the treatment of acute uncomplicated influenza in people aged 12 years or older who have been symptomatic for 48 hours or less.
The FDA approval is based on results from two randomized, clinical trials. In both trials, patients who received Xofluza experienced a shorter duration until alleviation of symptoms, compared with patients who received a placebo. In the second trial, patients who received Xofluza and patients who received another approved antiviral influenza medication experienced similar durations until symptom alleviation.
“When treatment is started within 48 hours of becoming sick with flu symptoms, antiviral drugs can lessen symptoms and shorten the time patients feel sick. Having more treatment options that work in different ways to attack the virus is important because flu viruses can become resistant to antiviral drugs,” Debra Birnkrant, MD, director of the Division of Antiviral Products in the FDA’s Center for Drug Evaluation and Research, said in a press release.
The most common adverse events associated with Xofluza were diarrhea and bronchitis.
“This is the first new antiviral flu treatment with a novel mechanism of action approved by the FDA in nearly 20 years,” FDA Commissioner Scott Gottlieb, MD, added. “With thousands of people getting the flu every year, and many people becoming seriously ill, having safe and effective treatment alternatives is critical. This novel drug provides an important, additional treatment option.”
Find the full press release on the FDA website.
The Food and Drug Administration has approved Xofluza (baloxavir marboxil) for the treatment of acute uncomplicated influenza in people aged 12 years or older who have been symptomatic for 48 hours or less.
The FDA approval is based on results from two randomized, clinical trials. In both trials, patients who received Xofluza experienced a shorter duration until alleviation of symptoms, compared with patients who received a placebo. In the second trial, patients who received Xofluza and patients who received another approved antiviral influenza medication experienced similar durations until symptom alleviation.
“When treatment is started within 48 hours of becoming sick with flu symptoms, antiviral drugs can lessen symptoms and shorten the time patients feel sick. Having more treatment options that work in different ways to attack the virus is important because flu viruses can become resistant to antiviral drugs,” Debra Birnkrant, MD, director of the Division of Antiviral Products in the FDA’s Center for Drug Evaluation and Research, said in a press release.
The most common adverse events associated with Xofluza were diarrhea and bronchitis.
“This is the first new antiviral flu treatment with a novel mechanism of action approved by the FDA in nearly 20 years,” FDA Commissioner Scott Gottlieb, MD, added. “With thousands of people getting the flu every year, and many people becoming seriously ill, having safe and effective treatment alternatives is critical. This novel drug provides an important, additional treatment option.”
Find the full press release on the FDA website.
The Food and Drug Administration has approved Xofluza (baloxavir marboxil) for the treatment of acute uncomplicated influenza in people aged 12 years or older who have been symptomatic for 48 hours or less.
The FDA approval is based on results from two randomized, clinical trials. In both trials, patients who received Xofluza experienced a shorter duration until alleviation of symptoms, compared with patients who received a placebo. In the second trial, patients who received Xofluza and patients who received another approved antiviral influenza medication experienced similar durations until symptom alleviation.
“When treatment is started within 48 hours of becoming sick with flu symptoms, antiviral drugs can lessen symptoms and shorten the time patients feel sick. Having more treatment options that work in different ways to attack the virus is important because flu viruses can become resistant to antiviral drugs,” Debra Birnkrant, MD, director of the Division of Antiviral Products in the FDA’s Center for Drug Evaluation and Research, said in a press release.
The most common adverse events associated with Xofluza were diarrhea and bronchitis.
“This is the first new antiviral flu treatment with a novel mechanism of action approved by the FDA in nearly 20 years,” FDA Commissioner Scott Gottlieb, MD, added. “With thousands of people getting the flu every year, and many people becoming seriously ill, having safe and effective treatment alternatives is critical. This novel drug provides an important, additional treatment option.”
Find the full press release on the FDA website.
High-dose flu vaccine in RA patients beats standard dose
CHICAGO – The administration of high-dose vs. standard-dose influenza vaccine provided substantially better immune responses in seropositive rheumatoid arthritis patients in a randomized, active-controlled trial.
High-dose trivalent influenza vaccine is known to improve immune responses in the elderly, but the current findings, which were presented at the annual meeting of the American College of Rheumatology, are the first to document a successful intervention to enhance vaccine responses in immunocompromised patients, according to Inés Colmegna, MD, of McGill University, Montreal.
Dr. Colmegna and her colleagues assessed antibody responses to either standard-dose (15 mcg of hemagglutinin per strain) quadrivalent inactivated influenza vaccine (SD-QIV) or high-dose (60 mcg of hemagglutinin per strain) trivalent inactivated influenza vaccine (HD-TIV) in 140 and 139 patients, respectively.
Seroprotection rates prior to vaccination were comparable in the two groups, but the high-dose recipients had consistently higher overall responses to vaccination.
Seroconversion rates were 22.3% vs. 8.6% (odds ratio, 2.93) for the H3N2 strain (A/HongKong/4801/2014), and 44.6% vs. 28.6% (OR, 1.93) for the B Victoria Lin strain (B/Brisbane/60/2008). For the H1N1 strain A/California/7/2009 in 2016-2017 and closely related A/Michigan/45/2015 in 2017-2018, the seroconversion rates were 51.1% vs. 30.0% (OR, 2.91) and 46.4% vs. 24.6% (OR, 2.79), respectively. Seroprotection rates for the H3N2 strain were 48.5% vs. 30.9%, and for the B Victoria Lin strain, 60.0% vs. 50.7%. The seroprotection rates for the H1N1 strains together were and 80.4% vs. 73.5%, Dr. Colmegna said.
Seroconversion was defined as at least a fourfold serum hemagglutination inhibition (HI) antibody increase from prevaccination level (day 0), and seroprotection was defined as percent with HI titers of 1:40 or greater at postvaccination day 28.
After the researchers controlled for age, vaccine type, treatment type in the 3 months prior to vaccination and during the study period, Charlson comorbidity index, and RA duration, the only significant predictors of vaccine seroresponse were vaccine dose and age.
The findings are notable because RA patients have a nearly threefold increase in the risk of contracting influenza infection or related illness, compared with age-matched healthy controls, because of “inherent immune dysfunction associated with RA, comorbidities, the age of our patients, and immunosuppressive therapy,” Dr. Colmegna said.
For this reason, RA patients are a priority group for annual vaccination. However, while vaccination remains the most effective method for preventing influenza and its associated complications, vaccine-induced antibody responses and protection in RA are suboptimal, she explained, noting that this puts them at increased risk for severe influenza.
“There is a high priority to develop new approaches to try to decrease this risk,” she said.
It was unknown whether HD-TIV – the only currently available high-dose influenza vaccine – would safely enhance antibody production in RA as it does in the elderly, so she and her colleagues recruited patients from a tertiary care center during the 2016-2017 and 2017-2018 Northern Hemisphere influenza seasons for this study.
The mean age of the patients was 61 years, and 80% were women. All were on stable treatment with either disease-modifying antirheumatic drugs (DMARDs) or biologics for at least 3 months prior to vaccination; treatment types included DMARDs in 138 patients (49.5%), anticytokine therapy in 92 patients (33%), and anti-B-cell therapy and small molecules in 49 patients (17.6%). An analysis by treatment type showed a possible reduction in the rate of seroconversion in patients who received anti-B-cell therapy and small molecules, but the number of patients in the group was too small to make definitive conclusions, Dr. Colmegna said.
Treatment in all groups was safe, with no differences in adverse events between those receiving high- or standard-dose vaccine, and none of the adverse events were related to treatment.
Further, the high-dose vaccine was not associated with an increase in disease activity.
“We believe that these results will likely change clinical practice,” she concluded.
Dr. Colmegna reported having no disclosures.
SOURCE: Colmegna I et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract 837.
CHICAGO – The administration of high-dose vs. standard-dose influenza vaccine provided substantially better immune responses in seropositive rheumatoid arthritis patients in a randomized, active-controlled trial.
High-dose trivalent influenza vaccine is known to improve immune responses in the elderly, but the current findings, which were presented at the annual meeting of the American College of Rheumatology, are the first to document a successful intervention to enhance vaccine responses in immunocompromised patients, according to Inés Colmegna, MD, of McGill University, Montreal.
Dr. Colmegna and her colleagues assessed antibody responses to either standard-dose (15 mcg of hemagglutinin per strain) quadrivalent inactivated influenza vaccine (SD-QIV) or high-dose (60 mcg of hemagglutinin per strain) trivalent inactivated influenza vaccine (HD-TIV) in 140 and 139 patients, respectively.
Seroprotection rates prior to vaccination were comparable in the two groups, but the high-dose recipients had consistently higher overall responses to vaccination.
Seroconversion rates were 22.3% vs. 8.6% (odds ratio, 2.93) for the H3N2 strain (A/HongKong/4801/2014), and 44.6% vs. 28.6% (OR, 1.93) for the B Victoria Lin strain (B/Brisbane/60/2008). For the H1N1 strain A/California/7/2009 in 2016-2017 and closely related A/Michigan/45/2015 in 2017-2018, the seroconversion rates were 51.1% vs. 30.0% (OR, 2.91) and 46.4% vs. 24.6% (OR, 2.79), respectively. Seroprotection rates for the H3N2 strain were 48.5% vs. 30.9%, and for the B Victoria Lin strain, 60.0% vs. 50.7%. The seroprotection rates for the H1N1 strains together were and 80.4% vs. 73.5%, Dr. Colmegna said.
Seroconversion was defined as at least a fourfold serum hemagglutination inhibition (HI) antibody increase from prevaccination level (day 0), and seroprotection was defined as percent with HI titers of 1:40 or greater at postvaccination day 28.
After the researchers controlled for age, vaccine type, treatment type in the 3 months prior to vaccination and during the study period, Charlson comorbidity index, and RA duration, the only significant predictors of vaccine seroresponse were vaccine dose and age.
The findings are notable because RA patients have a nearly threefold increase in the risk of contracting influenza infection or related illness, compared with age-matched healthy controls, because of “inherent immune dysfunction associated with RA, comorbidities, the age of our patients, and immunosuppressive therapy,” Dr. Colmegna said.
For this reason, RA patients are a priority group for annual vaccination. However, while vaccination remains the most effective method for preventing influenza and its associated complications, vaccine-induced antibody responses and protection in RA are suboptimal, she explained, noting that this puts them at increased risk for severe influenza.
“There is a high priority to develop new approaches to try to decrease this risk,” she said.
It was unknown whether HD-TIV – the only currently available high-dose influenza vaccine – would safely enhance antibody production in RA as it does in the elderly, so she and her colleagues recruited patients from a tertiary care center during the 2016-2017 and 2017-2018 Northern Hemisphere influenza seasons for this study.
The mean age of the patients was 61 years, and 80% were women. All were on stable treatment with either disease-modifying antirheumatic drugs (DMARDs) or biologics for at least 3 months prior to vaccination; treatment types included DMARDs in 138 patients (49.5%), anticytokine therapy in 92 patients (33%), and anti-B-cell therapy and small molecules in 49 patients (17.6%). An analysis by treatment type showed a possible reduction in the rate of seroconversion in patients who received anti-B-cell therapy and small molecules, but the number of patients in the group was too small to make definitive conclusions, Dr. Colmegna said.
Treatment in all groups was safe, with no differences in adverse events between those receiving high- or standard-dose vaccine, and none of the adverse events were related to treatment.
Further, the high-dose vaccine was not associated with an increase in disease activity.
“We believe that these results will likely change clinical practice,” she concluded.
Dr. Colmegna reported having no disclosures.
SOURCE: Colmegna I et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract 837.
CHICAGO – The administration of high-dose vs. standard-dose influenza vaccine provided substantially better immune responses in seropositive rheumatoid arthritis patients in a randomized, active-controlled trial.
High-dose trivalent influenza vaccine is known to improve immune responses in the elderly, but the current findings, which were presented at the annual meeting of the American College of Rheumatology, are the first to document a successful intervention to enhance vaccine responses in immunocompromised patients, according to Inés Colmegna, MD, of McGill University, Montreal.
Dr. Colmegna and her colleagues assessed antibody responses to either standard-dose (15 mcg of hemagglutinin per strain) quadrivalent inactivated influenza vaccine (SD-QIV) or high-dose (60 mcg of hemagglutinin per strain) trivalent inactivated influenza vaccine (HD-TIV) in 140 and 139 patients, respectively.
Seroprotection rates prior to vaccination were comparable in the two groups, but the high-dose recipients had consistently higher overall responses to vaccination.
Seroconversion rates were 22.3% vs. 8.6% (odds ratio, 2.93) for the H3N2 strain (A/HongKong/4801/2014), and 44.6% vs. 28.6% (OR, 1.93) for the B Victoria Lin strain (B/Brisbane/60/2008). For the H1N1 strain A/California/7/2009 in 2016-2017 and closely related A/Michigan/45/2015 in 2017-2018, the seroconversion rates were 51.1% vs. 30.0% (OR, 2.91) and 46.4% vs. 24.6% (OR, 2.79), respectively. Seroprotection rates for the H3N2 strain were 48.5% vs. 30.9%, and for the B Victoria Lin strain, 60.0% vs. 50.7%. The seroprotection rates for the H1N1 strains together were and 80.4% vs. 73.5%, Dr. Colmegna said.
Seroconversion was defined as at least a fourfold serum hemagglutination inhibition (HI) antibody increase from prevaccination level (day 0), and seroprotection was defined as percent with HI titers of 1:40 or greater at postvaccination day 28.
After the researchers controlled for age, vaccine type, treatment type in the 3 months prior to vaccination and during the study period, Charlson comorbidity index, and RA duration, the only significant predictors of vaccine seroresponse were vaccine dose and age.
The findings are notable because RA patients have a nearly threefold increase in the risk of contracting influenza infection or related illness, compared with age-matched healthy controls, because of “inherent immune dysfunction associated with RA, comorbidities, the age of our patients, and immunosuppressive therapy,” Dr. Colmegna said.
For this reason, RA patients are a priority group for annual vaccination. However, while vaccination remains the most effective method for preventing influenza and its associated complications, vaccine-induced antibody responses and protection in RA are suboptimal, she explained, noting that this puts them at increased risk for severe influenza.
“There is a high priority to develop new approaches to try to decrease this risk,” she said.
It was unknown whether HD-TIV – the only currently available high-dose influenza vaccine – would safely enhance antibody production in RA as it does in the elderly, so she and her colleagues recruited patients from a tertiary care center during the 2016-2017 and 2017-2018 Northern Hemisphere influenza seasons for this study.
The mean age of the patients was 61 years, and 80% were women. All were on stable treatment with either disease-modifying antirheumatic drugs (DMARDs) or biologics for at least 3 months prior to vaccination; treatment types included DMARDs in 138 patients (49.5%), anticytokine therapy in 92 patients (33%), and anti-B-cell therapy and small molecules in 49 patients (17.6%). An analysis by treatment type showed a possible reduction in the rate of seroconversion in patients who received anti-B-cell therapy and small molecules, but the number of patients in the group was too small to make definitive conclusions, Dr. Colmegna said.
Treatment in all groups was safe, with no differences in adverse events between those receiving high- or standard-dose vaccine, and none of the adverse events were related to treatment.
Further, the high-dose vaccine was not associated with an increase in disease activity.
“We believe that these results will likely change clinical practice,” she concluded.
Dr. Colmegna reported having no disclosures.
SOURCE: Colmegna I et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract 837.
REPORTING FROM THE ACR ANNUAL MEETING
Key clinical point: High- vs. standard-dose flu vaccine improves immune responses in RA patients.
Major finding: High-dose trivalent inactivated influenza vaccine was associated with greater odds of H3N2, B Victoria Lin, and H1N1 seroconversion.
Study details: A randomized, active-controlled trial of 279 RA patients
Disclosures: Dr. Colmegna reported having no disclosures.
Source: Colmegna I et al. Arthritis Rheumatol. 2018;70(Suppl 10): Abstract 837.
Oral flu vaccine protects, evokes mucosal immunity
SAN FRANCISCO – In a phase II study, Vaxart’s oral flu vaccine was compared with a commercial injectable quadrivalent flu vaccine or placebo. The study found rates of illness were comparable between the oral vaccine and quadrivalent vaccinated groups.*

The recombinant adenovirus-based vaccine expresses hemagglutinin. It elicited a mucosal immune response, hinting that the mechanism of protection in flu vaccines may be dependent on the route of administration. It is also believed that a strong mucosal response is key to preventing future infections.
In an interview at IDWeek 2018, an annual scientific meeting on infectious diseases, Nikita Kolhatkar, PhD, a salaried employee of Vaxart, which makes the drug, describes the results of the study and explains the potential advantages of an oral flu vaccine versus a traditional injectable one. The oral formulation is cell-based and so is not vulnerable to the mutation and genetic drift that can occur in egg-based vaccines.
It is also more stable and, of course, less invasive than injectable vaccines, according to Dr. Kolhatkar.
*Correction, 10/9/2018: An earlier vs. of this article did not stress the comparability.
SOURCE: Kolhatkar N. IDWeek 2018. Poster abstract 1947.
SAN FRANCISCO – In a phase II study, Vaxart’s oral flu vaccine was compared with a commercial injectable quadrivalent flu vaccine or placebo. The study found rates of illness were comparable between the oral vaccine and quadrivalent vaccinated groups.*

The recombinant adenovirus-based vaccine expresses hemagglutinin. It elicited a mucosal immune response, hinting that the mechanism of protection in flu vaccines may be dependent on the route of administration. It is also believed that a strong mucosal response is key to preventing future infections.
In an interview at IDWeek 2018, an annual scientific meeting on infectious diseases, Nikita Kolhatkar, PhD, a salaried employee of Vaxart, which makes the drug, describes the results of the study and explains the potential advantages of an oral flu vaccine versus a traditional injectable one. The oral formulation is cell-based and so is not vulnerable to the mutation and genetic drift that can occur in egg-based vaccines.
It is also more stable and, of course, less invasive than injectable vaccines, according to Dr. Kolhatkar.
*Correction, 10/9/2018: An earlier vs. of this article did not stress the comparability.
SOURCE: Kolhatkar N. IDWeek 2018. Poster abstract 1947.
SAN FRANCISCO – In a phase II study, Vaxart’s oral flu vaccine was compared with a commercial injectable quadrivalent flu vaccine or placebo. The study found rates of illness were comparable between the oral vaccine and quadrivalent vaccinated groups.*

The recombinant adenovirus-based vaccine expresses hemagglutinin. It elicited a mucosal immune response, hinting that the mechanism of protection in flu vaccines may be dependent on the route of administration. It is also believed that a strong mucosal response is key to preventing future infections.
In an interview at IDWeek 2018, an annual scientific meeting on infectious diseases, Nikita Kolhatkar, PhD, a salaried employee of Vaxart, which makes the drug, describes the results of the study and explains the potential advantages of an oral flu vaccine versus a traditional injectable one. The oral formulation is cell-based and so is not vulnerable to the mutation and genetic drift that can occur in egg-based vaccines.
It is also more stable and, of course, less invasive than injectable vaccines, according to Dr. Kolhatkar.
*Correction, 10/9/2018: An earlier vs. of this article did not stress the comparability.
SOURCE: Kolhatkar N. IDWeek 2018. Poster abstract 1947.
REPORTING FROM IDWEEK 2018
Flu outbreaks may be more intense in small cities
Influenza outbreaks in the United States tend to be concentrated and intense in small cities and more evenly spread throughout the season in large cities, results of a recent study show.
Swings in humidity further intensified the influenza spikes in small cities, but didn’t seem to have as much of an effect in large cities, the results suggest.
These findings help explain differences in influenza transmission patterns between cities that have similar climates and virus epidemiology, according to researcher Benjamin D. Dalziel, PhD, of the departments of integrative biology and mathematics at Oregon State University in Corvallis.
“City size and structure can play a role in determining how other factors such as climate affect and influence transmission,” Dr. Dalziel said in a press conference.
“Our results show how metropolises play a disproportionately important role in this process, as epidemic foci, and as potential sentinel hubs, where epidemiological observatories could integrate local strain dynamics to predict larger-scale patterns. As the growth and form of cities affect their function as climate-driven incubators of infectious disease, it may be possible to design smarter cities that better control epidemics in the face of accelerating global change,” the researchers wrote in their study.
Dr. Dalziel and his coauthors analyzed the weekly incidence of influenza-like illness across 603 U.S. ZIP codes using data obtained from medical claims from 2002 to 2008. They used epidemic intensity as a summary statistic to compare cities. By this variable, low epidemic intensity indicated a diffuse spread evenly across weeks of the flu season, whereas high epidemic intensity indicated intensively focused outbreaks on particular weeks.
In small cities, epidemics were more intensely focused on shorter periods at the peak of flu season, they found. In large cities, incidence was more diffuse, according to results published in Science.
Patterns of where people live and work in a city may account for the more diffuse and prolonged outbreaks seen in large cities, the authors wrote. Large cities have organized population movement patterns and crowding. In more highly established work locations, for example, the population density is pronounced during the day.
“We found the structure makes a difference for how the flu spreads at different times of year,” Dr. Dalziel said of the study, which used U.S. Census data to evaluate spatial population distributions. “In large cities with more highly organized patterns, conditions play a relatively smaller role in flu transmission.”
Humidity’s lower impact on outbreaks in large cities might also be explained by population effects: “If an infected person is sitting beside you, it matters less what the specific humidity is,” Dr. Dalziel said, adding that the proximity helps the virus find hosts even when climatic conditions are not at their most favorable.
The study findings may have implications for health care resources in small cities, which could be strained by intense outbreaks, said coinvestigator Cecile Viboud, PhD, of the Division of International Epidemiology and Population Studies, Fogarty International Center, National Institutes of Health, Bethesda, Md.
Intense outbreaks could overload the health care system, making it challenging to respond, especially around the peak of the epidemic. Pressure on the health care system may be less intense in cities such as Miami or New York, where flu epidemics are more diffuse and spread out during the year, she said.
Variations in vaccination coverage were not associated with variations in epidemic intensity at the state level. However, the data period that was analyzed ended in 2008, a time when flu vaccination rates were much lower than they are today, according to Dr. Viboud.
“It would be important to revisit the effect of city structure and humidity on flu transmission in a high vaccination regime in more recent years, especially if there is a lot of interest in developing broadly cross-protective flu vaccines, which might become available in the market in the future,” she said.
The researchers declared no competing interests related to their research, which was supported by a grant from the Bill & Melinda Gates Foundation, the RAPIDD program of the Science and Technology Directorate Department of Homeland Security, and the Fogarty International Center, National Institutes of Health.
SOURCE: Dalziel BD et al. Science. 2018 Oct 5;362(6410):75-9.
Public health policy makers may need to switch up their thinking about infection control during influenza outbreaks. Instead of targeting the population at large, it may make sense to focus on specific small towns or metropolitan areas for control.
Summary statistics, such as epidemic intensity, help to identify which places require more surge capacity to deal with peak health care demand. They also help to guide locations for active influenza surveillance where long transmission chains of influenza occur, and where new genetic variants of the influenza virus can be detected.
The findings of this study could foster the development of more accurate short-term, small-scale forecasts of the expected health care demand in a season. Most important, they could guide long-term projections that reveal how the shifting demography, growth of cities, and the changing climate alter infection dynamics and required control efforts.
Prof. Jacco Wallinga is with the Center for Infectious Disease Control, National Institute for Public Health and the Environment, Bilthoven, the Netherlands, and the Department of Biomedical Data Sciences, Leiden (the Netherlands) University Medical Center. These comments appeared in his editorial in Science (2018 Oct 5;362[6410]:29-30).
Public health policy makers may need to switch up their thinking about infection control during influenza outbreaks. Instead of targeting the population at large, it may make sense to focus on specific small towns or metropolitan areas for control.
Summary statistics, such as epidemic intensity, help to identify which places require more surge capacity to deal with peak health care demand. They also help to guide locations for active influenza surveillance where long transmission chains of influenza occur, and where new genetic variants of the influenza virus can be detected.
The findings of this study could foster the development of more accurate short-term, small-scale forecasts of the expected health care demand in a season. Most important, they could guide long-term projections that reveal how the shifting demography, growth of cities, and the changing climate alter infection dynamics and required control efforts.
Prof. Jacco Wallinga is with the Center for Infectious Disease Control, National Institute for Public Health and the Environment, Bilthoven, the Netherlands, and the Department of Biomedical Data Sciences, Leiden (the Netherlands) University Medical Center. These comments appeared in his editorial in Science (2018 Oct 5;362[6410]:29-30).
Public health policy makers may need to switch up their thinking about infection control during influenza outbreaks. Instead of targeting the population at large, it may make sense to focus on specific small towns or metropolitan areas for control.
Summary statistics, such as epidemic intensity, help to identify which places require more surge capacity to deal with peak health care demand. They also help to guide locations for active influenza surveillance where long transmission chains of influenza occur, and where new genetic variants of the influenza virus can be detected.
The findings of this study could foster the development of more accurate short-term, small-scale forecasts of the expected health care demand in a season. Most important, they could guide long-term projections that reveal how the shifting demography, growth of cities, and the changing climate alter infection dynamics and required control efforts.
Prof. Jacco Wallinga is with the Center for Infectious Disease Control, National Institute for Public Health and the Environment, Bilthoven, the Netherlands, and the Department of Biomedical Data Sciences, Leiden (the Netherlands) University Medical Center. These comments appeared in his editorial in Science (2018 Oct 5;362[6410]:29-30).
Influenza outbreaks in the United States tend to be concentrated and intense in small cities and more evenly spread throughout the season in large cities, results of a recent study show.
Swings in humidity further intensified the influenza spikes in small cities, but didn’t seem to have as much of an effect in large cities, the results suggest.
These findings help explain differences in influenza transmission patterns between cities that have similar climates and virus epidemiology, according to researcher Benjamin D. Dalziel, PhD, of the departments of integrative biology and mathematics at Oregon State University in Corvallis.
“City size and structure can play a role in determining how other factors such as climate affect and influence transmission,” Dr. Dalziel said in a press conference.
“Our results show how metropolises play a disproportionately important role in this process, as epidemic foci, and as potential sentinel hubs, where epidemiological observatories could integrate local strain dynamics to predict larger-scale patterns. As the growth and form of cities affect their function as climate-driven incubators of infectious disease, it may be possible to design smarter cities that better control epidemics in the face of accelerating global change,” the researchers wrote in their study.
Dr. Dalziel and his coauthors analyzed the weekly incidence of influenza-like illness across 603 U.S. ZIP codes using data obtained from medical claims from 2002 to 2008. They used epidemic intensity as a summary statistic to compare cities. By this variable, low epidemic intensity indicated a diffuse spread evenly across weeks of the flu season, whereas high epidemic intensity indicated intensively focused outbreaks on particular weeks.
In small cities, epidemics were more intensely focused on shorter periods at the peak of flu season, they found. In large cities, incidence was more diffuse, according to results published in Science.
Patterns of where people live and work in a city may account for the more diffuse and prolonged outbreaks seen in large cities, the authors wrote. Large cities have organized population movement patterns and crowding. In more highly established work locations, for example, the population density is pronounced during the day.
“We found the structure makes a difference for how the flu spreads at different times of year,” Dr. Dalziel said of the study, which used U.S. Census data to evaluate spatial population distributions. “In large cities with more highly organized patterns, conditions play a relatively smaller role in flu transmission.”
Humidity’s lower impact on outbreaks in large cities might also be explained by population effects: “If an infected person is sitting beside you, it matters less what the specific humidity is,” Dr. Dalziel said, adding that the proximity helps the virus find hosts even when climatic conditions are not at their most favorable.
The study findings may have implications for health care resources in small cities, which could be strained by intense outbreaks, said coinvestigator Cecile Viboud, PhD, of the Division of International Epidemiology and Population Studies, Fogarty International Center, National Institutes of Health, Bethesda, Md.
Intense outbreaks could overload the health care system, making it challenging to respond, especially around the peak of the epidemic. Pressure on the health care system may be less intense in cities such as Miami or New York, where flu epidemics are more diffuse and spread out during the year, she said.
Variations in vaccination coverage were not associated with variations in epidemic intensity at the state level. However, the data period that was analyzed ended in 2008, a time when flu vaccination rates were much lower than they are today, according to Dr. Viboud.
“It would be important to revisit the effect of city structure and humidity on flu transmission in a high vaccination regime in more recent years, especially if there is a lot of interest in developing broadly cross-protective flu vaccines, which might become available in the market in the future,” she said.
The researchers declared no competing interests related to their research, which was supported by a grant from the Bill & Melinda Gates Foundation, the RAPIDD program of the Science and Technology Directorate Department of Homeland Security, and the Fogarty International Center, National Institutes of Health.
SOURCE: Dalziel BD et al. Science. 2018 Oct 5;362(6410):75-9.
Influenza outbreaks in the United States tend to be concentrated and intense in small cities and more evenly spread throughout the season in large cities, results of a recent study show.
Swings in humidity further intensified the influenza spikes in small cities, but didn’t seem to have as much of an effect in large cities, the results suggest.
These findings help explain differences in influenza transmission patterns between cities that have similar climates and virus epidemiology, according to researcher Benjamin D. Dalziel, PhD, of the departments of integrative biology and mathematics at Oregon State University in Corvallis.
“City size and structure can play a role in determining how other factors such as climate affect and influence transmission,” Dr. Dalziel said in a press conference.
“Our results show how metropolises play a disproportionately important role in this process, as epidemic foci, and as potential sentinel hubs, where epidemiological observatories could integrate local strain dynamics to predict larger-scale patterns. As the growth and form of cities affect their function as climate-driven incubators of infectious disease, it may be possible to design smarter cities that better control epidemics in the face of accelerating global change,” the researchers wrote in their study.
Dr. Dalziel and his coauthors analyzed the weekly incidence of influenza-like illness across 603 U.S. ZIP codes using data obtained from medical claims from 2002 to 2008. They used epidemic intensity as a summary statistic to compare cities. By this variable, low epidemic intensity indicated a diffuse spread evenly across weeks of the flu season, whereas high epidemic intensity indicated intensively focused outbreaks on particular weeks.
In small cities, epidemics were more intensely focused on shorter periods at the peak of flu season, they found. In large cities, incidence was more diffuse, according to results published in Science.
Patterns of where people live and work in a city may account for the more diffuse and prolonged outbreaks seen in large cities, the authors wrote. Large cities have organized population movement patterns and crowding. In more highly established work locations, for example, the population density is pronounced during the day.
“We found the structure makes a difference for how the flu spreads at different times of year,” Dr. Dalziel said of the study, which used U.S. Census data to evaluate spatial population distributions. “In large cities with more highly organized patterns, conditions play a relatively smaller role in flu transmission.”
Humidity’s lower impact on outbreaks in large cities might also be explained by population effects: “If an infected person is sitting beside you, it matters less what the specific humidity is,” Dr. Dalziel said, adding that the proximity helps the virus find hosts even when climatic conditions are not at their most favorable.
The study findings may have implications for health care resources in small cities, which could be strained by intense outbreaks, said coinvestigator Cecile Viboud, PhD, of the Division of International Epidemiology and Population Studies, Fogarty International Center, National Institutes of Health, Bethesda, Md.
Intense outbreaks could overload the health care system, making it challenging to respond, especially around the peak of the epidemic. Pressure on the health care system may be less intense in cities such as Miami or New York, where flu epidemics are more diffuse and spread out during the year, she said.
Variations in vaccination coverage were not associated with variations in epidemic intensity at the state level. However, the data period that was analyzed ended in 2008, a time when flu vaccination rates were much lower than they are today, according to Dr. Viboud.
“It would be important to revisit the effect of city structure and humidity on flu transmission in a high vaccination regime in more recent years, especially if there is a lot of interest in developing broadly cross-protective flu vaccines, which might become available in the market in the future,” she said.
The researchers declared no competing interests related to their research, which was supported by a grant from the Bill & Melinda Gates Foundation, the RAPIDD program of the Science and Technology Directorate Department of Homeland Security, and the Fogarty International Center, National Institutes of Health.
SOURCE: Dalziel BD et al. Science. 2018 Oct 5;362(6410):75-9.
FROM SCIENCE
Key clinical point: The intensity of influenza epidemics in U.S. cities varies according to population.
Major finding: Smaller cities had more intense outbreaks concentrated around the peak of flu season, while larger cities had cases spread throughout the season.
Study details: Analysis of weekly influenza-like illness incidence for 603 U.S. ZIP codes in medical claims data from 2002 to 2008.
Disclosures: The authors declared no competing interests. Funding came from the Bill & Melinda Gates Foundation, the Science and Technology Directorate Department of Homeland Security, and the Fogarty International Center, National Institutes of Health.
Source: Dalziel BD et al. Science. 2018 Oct 5;362(6410):75-9.
CDC: Trivalent adjuvanted influenza vaccine aIIV3 safe in elderly adults
ATLANTA – according to an analysis of reports to the Vaccine Adverse Event Reporting System (VAERS) during July 2016 through March 2018.
VAERS received 630 reports related to the vaccine (aIIV3; FLUAD) during the study period, of which 521 involved adults aged 65 years and older.
“Eighteen (3%) were serious reports, including two death reports (0.4%), all in adults aged [at least] 65 years,” Penina Haber and her colleagues at the Immunization Safety Office at the Centers for Disease Control and Prevention reported in a poster at the International Conference on Emerging Infectious Diseases.
The deaths included a 75-year-old man who died from Sjögren’s syndrome and a 65-year-old man who died from a myocardial infarction. The other serious events included five neurologic disorders (two cases of Guillain-Barré syndrome and one each of Bell’s palsy, Bickerstaff encephalitis, and lower-extremity weakness), five musculoskeletal and connective tissue disorders (three with shoulder pain and two with arm pain), three general disorders and administration site conditions (two cases of fever/chills and one case of cellulitis/bursitis), and one case each of a gastrointestinal disorder (acute diarrhea/gastroenteritis), an injury (a fall), and a skin/subcutaneous tissue disorder (keratosis pilaris rubra), according to the investigators.
There were no reports of anaphylaxis.
For the sake of comparison, the investigators also looked at reports associated with IIV3-HD and IIV3/IIV4 vaccines in adults aged 65 years and older during the same time period; they found that patient characteristics and reported events were similar for all the vaccines. For example, the percentages of reports involving patients aged 65 years and older were 65% or 66% for each, and those involving patients aged 75-84 years were 27%-29%. Further, 0.2%-0.6% of reports for each vaccine involved death.
The most frequently reported events for aIIV3, IIV3-HD, and IIV3/IIV4, respectively, were extremity pain (21%, 17%, and 15%, respectively), injection site erythema (18%, 19%, and 15%), and injection site pain (15%, 16%, and 16%), they said.
The aIIV3 vaccine – the first seasonal inactivated trivalent influenza vaccine produced from three influenza virus strains (two subtype A strains and one type B strain) – was approved by the Food and Drug Administration in 2015 for adults aged 65 years and older. It was the first influenza vaccine containing the adjuvant MF59 – a purified oil-in-water emulsion of squalene oil added to boost immune response in that population. Its safety was assessed in 15 randomized, controlled clinical studies, and several trials in older adults supported its efficacy and safety over nonadjuvanted influenza vaccines, the investigators reported. They noted that the Advisory Committee on Immunization Practices (ACIP) recommended the vaccine as an option for routine use in adults aged 65 years and older during the 2016-2017 flu seasons.
For the 2018-2019 flu season, ACIP determined that “For persons aged ≥65 years, any age-appropriate IIV formulation (standard-dose or high-dose, trivalent or quadrivalent, unadjuvanted or adjuvanted) or RIV4 are acceptable options.”
The findings of the analysis of the 2017-2018 flu season data are consistent with prelicensure studies, Ms. Haber and her colleagues concluded, noting that data mining did not detect disproportional reporting of any unexpected adverse event.
“[There were] no safety concerns following aIIV3 when compared to the nonadjuvanted influenza vaccines (IIV3-HD or IIV3/IIV4),” they wrote, adding that the “CDC and FDA will continue to monitor and ensure the safety of aIIV3.”
Ms. Haber reported having no disclosures
sworcester@frontlinemedcom.com
SOURCE: Haber P et al. ICEID 2018, Board 320.
ATLANTA – according to an analysis of reports to the Vaccine Adverse Event Reporting System (VAERS) during July 2016 through March 2018.
VAERS received 630 reports related to the vaccine (aIIV3; FLUAD) during the study period, of which 521 involved adults aged 65 years and older.
“Eighteen (3%) were serious reports, including two death reports (0.4%), all in adults aged [at least] 65 years,” Penina Haber and her colleagues at the Immunization Safety Office at the Centers for Disease Control and Prevention reported in a poster at the International Conference on Emerging Infectious Diseases.
The deaths included a 75-year-old man who died from Sjögren’s syndrome and a 65-year-old man who died from a myocardial infarction. The other serious events included five neurologic disorders (two cases of Guillain-Barré syndrome and one each of Bell’s palsy, Bickerstaff encephalitis, and lower-extremity weakness), five musculoskeletal and connective tissue disorders (three with shoulder pain and two with arm pain), three general disorders and administration site conditions (two cases of fever/chills and one case of cellulitis/bursitis), and one case each of a gastrointestinal disorder (acute diarrhea/gastroenteritis), an injury (a fall), and a skin/subcutaneous tissue disorder (keratosis pilaris rubra), according to the investigators.
There were no reports of anaphylaxis.
For the sake of comparison, the investigators also looked at reports associated with IIV3-HD and IIV3/IIV4 vaccines in adults aged 65 years and older during the same time period; they found that patient characteristics and reported events were similar for all the vaccines. For example, the percentages of reports involving patients aged 65 years and older were 65% or 66% for each, and those involving patients aged 75-84 years were 27%-29%. Further, 0.2%-0.6% of reports for each vaccine involved death.
The most frequently reported events for aIIV3, IIV3-HD, and IIV3/IIV4, respectively, were extremity pain (21%, 17%, and 15%, respectively), injection site erythema (18%, 19%, and 15%), and injection site pain (15%, 16%, and 16%), they said.
The aIIV3 vaccine – the first seasonal inactivated trivalent influenza vaccine produced from three influenza virus strains (two subtype A strains and one type B strain) – was approved by the Food and Drug Administration in 2015 for adults aged 65 years and older. It was the first influenza vaccine containing the adjuvant MF59 – a purified oil-in-water emulsion of squalene oil added to boost immune response in that population. Its safety was assessed in 15 randomized, controlled clinical studies, and several trials in older adults supported its efficacy and safety over nonadjuvanted influenza vaccines, the investigators reported. They noted that the Advisory Committee on Immunization Practices (ACIP) recommended the vaccine as an option for routine use in adults aged 65 years and older during the 2016-2017 flu seasons.
For the 2018-2019 flu season, ACIP determined that “For persons aged ≥65 years, any age-appropriate IIV formulation (standard-dose or high-dose, trivalent or quadrivalent, unadjuvanted or adjuvanted) or RIV4 are acceptable options.”
The findings of the analysis of the 2017-2018 flu season data are consistent with prelicensure studies, Ms. Haber and her colleagues concluded, noting that data mining did not detect disproportional reporting of any unexpected adverse event.
“[There were] no safety concerns following aIIV3 when compared to the nonadjuvanted influenza vaccines (IIV3-HD or IIV3/IIV4),” they wrote, adding that the “CDC and FDA will continue to monitor and ensure the safety of aIIV3.”
Ms. Haber reported having no disclosures
sworcester@frontlinemedcom.com
SOURCE: Haber P et al. ICEID 2018, Board 320.
ATLANTA – according to an analysis of reports to the Vaccine Adverse Event Reporting System (VAERS) during July 2016 through March 2018.
VAERS received 630 reports related to the vaccine (aIIV3; FLUAD) during the study period, of which 521 involved adults aged 65 years and older.
“Eighteen (3%) were serious reports, including two death reports (0.4%), all in adults aged [at least] 65 years,” Penina Haber and her colleagues at the Immunization Safety Office at the Centers for Disease Control and Prevention reported in a poster at the International Conference on Emerging Infectious Diseases.
The deaths included a 75-year-old man who died from Sjögren’s syndrome and a 65-year-old man who died from a myocardial infarction. The other serious events included five neurologic disorders (two cases of Guillain-Barré syndrome and one each of Bell’s palsy, Bickerstaff encephalitis, and lower-extremity weakness), five musculoskeletal and connective tissue disorders (three with shoulder pain and two with arm pain), three general disorders and administration site conditions (two cases of fever/chills and one case of cellulitis/bursitis), and one case each of a gastrointestinal disorder (acute diarrhea/gastroenteritis), an injury (a fall), and a skin/subcutaneous tissue disorder (keratosis pilaris rubra), according to the investigators.
There were no reports of anaphylaxis.
For the sake of comparison, the investigators also looked at reports associated with IIV3-HD and IIV3/IIV4 vaccines in adults aged 65 years and older during the same time period; they found that patient characteristics and reported events were similar for all the vaccines. For example, the percentages of reports involving patients aged 65 years and older were 65% or 66% for each, and those involving patients aged 75-84 years were 27%-29%. Further, 0.2%-0.6% of reports for each vaccine involved death.
The most frequently reported events for aIIV3, IIV3-HD, and IIV3/IIV4, respectively, were extremity pain (21%, 17%, and 15%, respectively), injection site erythema (18%, 19%, and 15%), and injection site pain (15%, 16%, and 16%), they said.
The aIIV3 vaccine – the first seasonal inactivated trivalent influenza vaccine produced from three influenza virus strains (two subtype A strains and one type B strain) – was approved by the Food and Drug Administration in 2015 for adults aged 65 years and older. It was the first influenza vaccine containing the adjuvant MF59 – a purified oil-in-water emulsion of squalene oil added to boost immune response in that population. Its safety was assessed in 15 randomized, controlled clinical studies, and several trials in older adults supported its efficacy and safety over nonadjuvanted influenza vaccines, the investigators reported. They noted that the Advisory Committee on Immunization Practices (ACIP) recommended the vaccine as an option for routine use in adults aged 65 years and older during the 2016-2017 flu seasons.
For the 2018-2019 flu season, ACIP determined that “For persons aged ≥65 years, any age-appropriate IIV formulation (standard-dose or high-dose, trivalent or quadrivalent, unadjuvanted or adjuvanted) or RIV4 are acceptable options.”
The findings of the analysis of the 2017-2018 flu season data are consistent with prelicensure studies, Ms. Haber and her colleagues concluded, noting that data mining did not detect disproportional reporting of any unexpected adverse event.
“[There were] no safety concerns following aIIV3 when compared to the nonadjuvanted influenza vaccines (IIV3-HD or IIV3/IIV4),” they wrote, adding that the “CDC and FDA will continue to monitor and ensure the safety of aIIV3.”
Ms. Haber reported having no disclosures
sworcester@frontlinemedcom.com
SOURCE: Haber P et al. ICEID 2018, Board 320.
REPORTING FROM ICEID 2018
Key clinical point: No new or unexpected adverse events were reported among the 630 reports related to the vaccine during the study period, of which 521 involved adults aged 65 years and older.
Major finding: Of 521 reports, 18 were serious, and there were two deaths.
Study details: A review of 521 reports to the Vaccine Adverse Event Reporting System in 2017-2018.
Disclosures: Ms. Haber reported having no disclosures.
Source: Haber P et al. ICEID 2018, Board 320.