User login
Bone density gains are lost following stop of denosumab in glucocorticoid-treated RA
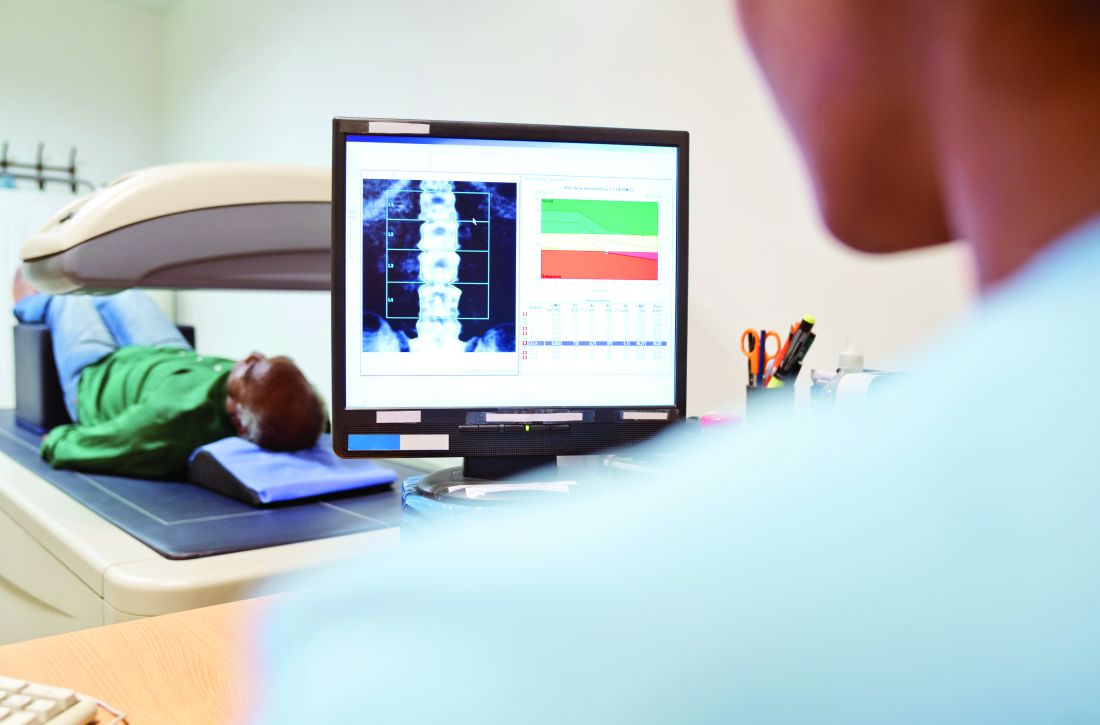
Patients with rheumatoid arthritis who were taking glucocorticoids and received short-term denosumab (Prolia) had lost any gains in bone mineral density at the spine or hip as well as any improvements in bone turnover markers a year later, according to findings from a post-hoc analysis of a phase 2 trial.
That is, stopping denosumab after a 12-month course resulted in a gradual increase in bone turnover markers and a concurrent return to baseline lumbar spine and total hip bone mineral density, Kenneth G. Saag, MD, professor of medicine and division director of clinical immunology and rheumatology at the University of Alabama at Birmingham, and colleagues reported in an article published online Sept. 17, 2021 in Arthritis & Rheumatology.
“These results provide further support for recommendations that patients discontinuing denosumab should transition to follow-on osteoporosis therapy to prevent or minimize remodeling-induced bone loss,” they concluded.
Like all nonbisphosphonate medications for osteoporosis, Dr. Saag and colleagues wrote, the pharmacologic effects of denosumab are readily reversible after discontinuation.
The current findings in glucocorticoid-treated patients are consistent with those observed in postmenopausal women 2 years after discontinuing denosumab therapy for osteoporosis. Denosumab is typically given for a longer time in such patients, compared with patients receiving glucocorticoids.
Invited to comment, Karen E. Hansen, MD, a rheumatologist and associate professor at the University of Wisconsin, Madison, who was not involved with the study, agreed that the results “highlight the need to prescribe another osteoporosis medication after stopping denosumab, in hopes of preventing loss of bone mineral density.”
Dr. Hansen, a coauthor of a review and meta-analysis of denosumab in the treatment of glucocorticoid-induced osteoporosis, noted that the American College of Rheumatology guideline for the prevention and treatment of glucocorticoid-induced osteoporosis suggests the use of denosumab as fourth-line therapy, after oral bisphosphonates, intravenous bisphosphonates, and teriparatide (Forteo).
“Its use is particularly relevant in patients who have contraindications or side effects from bisphosphonates or anabolic therapy, or when patient compliance must be ensured,” she said in an interview.
“The type, timing, and effect of therapy after denosumab discontinuation, however, remain controversial,” Dr. Saag and colleagues noted.
However, ongoing trials that are investigating the optimal medication and dosing needed to prevent such losses in bone mineral density after stopping denosumab should provide greater insight, Dr. Hansen said.
Bone health after stopping denosumab
Patients with rheumatoid arthritis often have bone loss that can be worsened by their frequent use of glucocorticoids, leading to an increased risk of fragility fractures.
Denosumab, a monoclonal antibody that inhibits receptor activator of nuclear factor kappaB ligand (RANKL), was approved by the Food and Drug Administration in 2018 for treating patients with glucocorticoid-induced osteoporosis and a high risk of fracture.
Dr. Saag and colleagues performed a post-hoc analysis of a subgroup of 82 patients receiving glucocorticoids who were part of a larger phase 2 clinical trial of 218 patients with rheumatoid arthritis.
The patients had been randomized to receive placebo (n = 26), 60 mg denosumab (the approved dose, n = 27), or 180 mg denosumab (n = 29), given as two subcutaneous 6-month injections at baseline and 6 months, followed by 12 months without any bone-loss prevention therapy.
The patients had a mean age of 55, and 62% were women.
While receiving denosumab, their serum levels of the bone resorption marker C-terminal telopeptide of type I collagen (CTX) and the bone formation marker procollagen type I N-terminal propeptide (P1NP) decreased significantly from baseline.
In patients who received the 60-mg dose of denosumab, CTX levels had returned to baseline levels 6 months and 12 months after denosumab was discontinued.
Median levels of P1NP in these patients were 0.16% lower than baseline and 15% higher than baseline at 6 months and 12 months after denosumab was stopped, respectively.
In patients who received the 180-mg dose of denosumab, CTX levels also had returned to baseline levels 6 months and 12 months after denosumab was discontinued.
Median levels of P1NP in these patients were 9% and 76% higher than baseline levels, at 6 months and 12 months after denosumab was stopped, respectively.
Bone mineral density at the lumbar spine and total hip increased during the 12 months of denosumab treatment and then returned to baseline after 12 months of discontinuation of both doses of denosumab.
No osteoporotic fractures were reported during the 12-month denosumab treatment or the 12-month follow-up.
The study was funded by Amgen, which markets denosumab. Dr. Saag is an investigator with Amgen, Mereo, and Radius, and a consultant for Amgen and Roche. Four coauthors are employees of Amgen. The other six coauthors all reported a financial relationship with Amgen.
Patients with rheumatoid arthritis who were taking glucocorticoids and received short-term denosumab (Prolia) had lost any gains in bone mineral density at the spine or hip as well as any improvements in bone turnover markers a year later, according to findings from a post-hoc analysis of a phase 2 trial.
That is, stopping denosumab after a 12-month course resulted in a gradual increase in bone turnover markers and a concurrent return to baseline lumbar spine and total hip bone mineral density, Kenneth G. Saag, MD, professor of medicine and division director of clinical immunology and rheumatology at the University of Alabama at Birmingham, and colleagues reported in an article published online Sept. 17, 2021 in Arthritis & Rheumatology.
“These results provide further support for recommendations that patients discontinuing denosumab should transition to follow-on osteoporosis therapy to prevent or minimize remodeling-induced bone loss,” they concluded.
Like all nonbisphosphonate medications for osteoporosis, Dr. Saag and colleagues wrote, the pharmacologic effects of denosumab are readily reversible after discontinuation.
The current findings in glucocorticoid-treated patients are consistent with those observed in postmenopausal women 2 years after discontinuing denosumab therapy for osteoporosis. Denosumab is typically given for a longer time in such patients, compared with patients receiving glucocorticoids.
Invited to comment, Karen E. Hansen, MD, a rheumatologist and associate professor at the University of Wisconsin, Madison, who was not involved with the study, agreed that the results “highlight the need to prescribe another osteoporosis medication after stopping denosumab, in hopes of preventing loss of bone mineral density.”
Dr. Hansen, a coauthor of a review and meta-analysis of denosumab in the treatment of glucocorticoid-induced osteoporosis, noted that the American College of Rheumatology guideline for the prevention and treatment of glucocorticoid-induced osteoporosis suggests the use of denosumab as fourth-line therapy, after oral bisphosphonates, intravenous bisphosphonates, and teriparatide (Forteo).
“Its use is particularly relevant in patients who have contraindications or side effects from bisphosphonates or anabolic therapy, or when patient compliance must be ensured,” she said in an interview.
“The type, timing, and effect of therapy after denosumab discontinuation, however, remain controversial,” Dr. Saag and colleagues noted.
However, ongoing trials that are investigating the optimal medication and dosing needed to prevent such losses in bone mineral density after stopping denosumab should provide greater insight, Dr. Hansen said.
Bone health after stopping denosumab
Patients with rheumatoid arthritis often have bone loss that can be worsened by their frequent use of glucocorticoids, leading to an increased risk of fragility fractures.
Denosumab, a monoclonal antibody that inhibits receptor activator of nuclear factor kappaB ligand (RANKL), was approved by the Food and Drug Administration in 2018 for treating patients with glucocorticoid-induced osteoporosis and a high risk of fracture.
Dr. Saag and colleagues performed a post-hoc analysis of a subgroup of 82 patients receiving glucocorticoids who were part of a larger phase 2 clinical trial of 218 patients with rheumatoid arthritis.
The patients had been randomized to receive placebo (n = 26), 60 mg denosumab (the approved dose, n = 27), or 180 mg denosumab (n = 29), given as two subcutaneous 6-month injections at baseline and 6 months, followed by 12 months without any bone-loss prevention therapy.
The patients had a mean age of 55, and 62% were women.
While receiving denosumab, their serum levels of the bone resorption marker C-terminal telopeptide of type I collagen (CTX) and the bone formation marker procollagen type I N-terminal propeptide (P1NP) decreased significantly from baseline.
In patients who received the 60-mg dose of denosumab, CTX levels had returned to baseline levels 6 months and 12 months after denosumab was discontinued.
Median levels of P1NP in these patients were 0.16% lower than baseline and 15% higher than baseline at 6 months and 12 months after denosumab was stopped, respectively.
In patients who received the 180-mg dose of denosumab, CTX levels also had returned to baseline levels 6 months and 12 months after denosumab was discontinued.
Median levels of P1NP in these patients were 9% and 76% higher than baseline levels, at 6 months and 12 months after denosumab was stopped, respectively.
Bone mineral density at the lumbar spine and total hip increased during the 12 months of denosumab treatment and then returned to baseline after 12 months of discontinuation of both doses of denosumab.
No osteoporotic fractures were reported during the 12-month denosumab treatment or the 12-month follow-up.
The study was funded by Amgen, which markets denosumab. Dr. Saag is an investigator with Amgen, Mereo, and Radius, and a consultant for Amgen and Roche. Four coauthors are employees of Amgen. The other six coauthors all reported a financial relationship with Amgen.
Patients with rheumatoid arthritis who were taking glucocorticoids and received short-term denosumab (Prolia) had lost any gains in bone mineral density at the spine or hip as well as any improvements in bone turnover markers a year later, according to findings from a post-hoc analysis of a phase 2 trial.
That is, stopping denosumab after a 12-month course resulted in a gradual increase in bone turnover markers and a concurrent return to baseline lumbar spine and total hip bone mineral density, Kenneth G. Saag, MD, professor of medicine and division director of clinical immunology and rheumatology at the University of Alabama at Birmingham, and colleagues reported in an article published online Sept. 17, 2021 in Arthritis & Rheumatology.
“These results provide further support for recommendations that patients discontinuing denosumab should transition to follow-on osteoporosis therapy to prevent or minimize remodeling-induced bone loss,” they concluded.
Like all nonbisphosphonate medications for osteoporosis, Dr. Saag and colleagues wrote, the pharmacologic effects of denosumab are readily reversible after discontinuation.
The current findings in glucocorticoid-treated patients are consistent with those observed in postmenopausal women 2 years after discontinuing denosumab therapy for osteoporosis. Denosumab is typically given for a longer time in such patients, compared with patients receiving glucocorticoids.
Invited to comment, Karen E. Hansen, MD, a rheumatologist and associate professor at the University of Wisconsin, Madison, who was not involved with the study, agreed that the results “highlight the need to prescribe another osteoporosis medication after stopping denosumab, in hopes of preventing loss of bone mineral density.”
Dr. Hansen, a coauthor of a review and meta-analysis of denosumab in the treatment of glucocorticoid-induced osteoporosis, noted that the American College of Rheumatology guideline for the prevention and treatment of glucocorticoid-induced osteoporosis suggests the use of denosumab as fourth-line therapy, after oral bisphosphonates, intravenous bisphosphonates, and teriparatide (Forteo).
“Its use is particularly relevant in patients who have contraindications or side effects from bisphosphonates or anabolic therapy, or when patient compliance must be ensured,” she said in an interview.
“The type, timing, and effect of therapy after denosumab discontinuation, however, remain controversial,” Dr. Saag and colleagues noted.
However, ongoing trials that are investigating the optimal medication and dosing needed to prevent such losses in bone mineral density after stopping denosumab should provide greater insight, Dr. Hansen said.
Bone health after stopping denosumab
Patients with rheumatoid arthritis often have bone loss that can be worsened by their frequent use of glucocorticoids, leading to an increased risk of fragility fractures.
Denosumab, a monoclonal antibody that inhibits receptor activator of nuclear factor kappaB ligand (RANKL), was approved by the Food and Drug Administration in 2018 for treating patients with glucocorticoid-induced osteoporosis and a high risk of fracture.
Dr. Saag and colleagues performed a post-hoc analysis of a subgroup of 82 patients receiving glucocorticoids who were part of a larger phase 2 clinical trial of 218 patients with rheumatoid arthritis.
The patients had been randomized to receive placebo (n = 26), 60 mg denosumab (the approved dose, n = 27), or 180 mg denosumab (n = 29), given as two subcutaneous 6-month injections at baseline and 6 months, followed by 12 months without any bone-loss prevention therapy.
The patients had a mean age of 55, and 62% were women.
While receiving denosumab, their serum levels of the bone resorption marker C-terminal telopeptide of type I collagen (CTX) and the bone formation marker procollagen type I N-terminal propeptide (P1NP) decreased significantly from baseline.
In patients who received the 60-mg dose of denosumab, CTX levels had returned to baseline levels 6 months and 12 months after denosumab was discontinued.
Median levels of P1NP in these patients were 0.16% lower than baseline and 15% higher than baseline at 6 months and 12 months after denosumab was stopped, respectively.
In patients who received the 180-mg dose of denosumab, CTX levels also had returned to baseline levels 6 months and 12 months after denosumab was discontinued.
Median levels of P1NP in these patients were 9% and 76% higher than baseline levels, at 6 months and 12 months after denosumab was stopped, respectively.
Bone mineral density at the lumbar spine and total hip increased during the 12 months of denosumab treatment and then returned to baseline after 12 months of discontinuation of both doses of denosumab.
No osteoporotic fractures were reported during the 12-month denosumab treatment or the 12-month follow-up.
The study was funded by Amgen, which markets denosumab. Dr. Saag is an investigator with Amgen, Mereo, and Radius, and a consultant for Amgen and Roche. Four coauthors are employees of Amgen. The other six coauthors all reported a financial relationship with Amgen.
FROM ARTHRITIS & RHEUMATOLOGY
Menopause society issues first osteoporosis advice in 10 years
In the first revision to its guidance on the management of osteoporosis in a decade, the North American Menopause Society has issued an updated position statement addressing evolving evidence on osteoporosis issues ranging from screening and risk assessment to appropriate use of preventive therapy in postmenopausal women.
“Since the 2010 statement, there have been important new developments in our field, including better delineation of risk factors for fracture, resulting in better strategies for assessing fracture risk,” Michael R. McClung, MD, who is a NAMS board member and colead of the editorial panel for the 2021 position statement, told this news organization. Dr. McClung is also director emeritus of the Oregon Osteoporosis Center in Portland.
“There is much more information about the long-term safety of therapies,” he added. Dr. McClung also noted “the availability of four new drugs for the prevention and treatment of osteoporosis and clinical experience informing us of the effects of using different treatments in various sequences.”
Osteoporosis is substantially underdiagnosed and undertreated
A basis for the update, recently published in Menopause: The Journal of the North American Menopause Society, is the need to tackle the troubling fact that approximately half of postmenopausal women will experience a fracture related to osteoporosis in their lifetime, yet the condition is “substantially underdiagnosed and undertreated,” NAMS underscores.
With that in mind, osteoporosis should be considered by practitioners treating menopausal and postmenopausal women at all levels of care.
“All physicians and advanced care providers caring for postmenopausal women should be comfortable assessing and managing their patients with, or at risk for, fractures,” Dr. McClung added.
Osteoporosis prevention in young menopausal women
The NAMS statement covers a broad range of issues, and while most recommendations generally follow those of other societies’ guidelines, a unique aspect is the emphasis on preventing osteoporosis in young menopausal women with estrogen or other drugs.
While underscoring that all menopausal women should be encouraged to adopt healthy lifestyles, with good diets and physical activity to reduce the risk of bone loss and fractures, pharmacologic interventions also have a role, NAMS says.
Though long an issue of debate, NAMS voices support for estrogen therapy as having an important role in osteoporosis prevention, as estrogen deficiency is the principal cause of bone loss in postmenopausal women.
“Hormone therapy is the most appropriate choice to prevent bone loss at the time of menopause for healthy women, particularly those who have menopause symptoms,” the group states. Drug interventions are specifically supported in women with premature menopause, at least until the average age of natural menopause, in addition to those with low bone mineral density (BMD) (T-score < –1.0) and those experiencing relatively rapid bone loss related to acute estrogen deficiency in the menopause transition or on discontinuing estrogen therapy.
“Although using drugs to prevent osteoporosis is not included in national osteoporosis guidelines, a strong clinical argument can be made for doing so, especially in women who come to menopause with low bone mass,” the report states.
And therapy is also recommended if patients have a low BMD and other risk factors for fracture, such as family history, but do not meet the criteria for osteoporosis treatment.
Ultimately, clinicians should work with patients when deciding the options, Dr. McClung said.
“After carefully weighing the small risks associated with hormone therapy or other therapies begun at the time of menopause, menopause practitioners and their patients can and should make informed decisions about the use of Food and Drug Administration–approved medications to prevent osteoporosis in women who are at risk for developing that condition,” he noted, adding that his view on the matter is his own and not necessarily that of NAMS.
New treatments endorsed for high-risk patients to avoid ‘bone attack’
While most patients are treated for osteoporosis with antiremodeling drugs such as bisphosphonates and denosumab, NAMS endorses “a new paradigm of beginning treatment with a bone-building agent followed by an antiremodeling agent” for women at very high risk of fracture.
“Consider osteoanabolic therapies for patients at very high risk of fracture, including older women with recent fractures, T-scores –3.0 and lower, or multiple other risk factors,” the statement suggests.
Among those at highest risk are women who have sustained a first fracture.
“A recent fracture in a postmenopausal woman is the strongest risk factor for another fracture,” Dr. McClung said.
In fact, “having a fracture should be thought of and assessed as a ‘bone attack,’ ” he asserted.
Therapy is recommended in such cases to rapidly increase bone density and reduce their subsequent fracture risk.
“For these patients, osteoanabolic or bone-building agents are more effective than bisphosphonates and are recommended as initial therapy,” Dr. McClung noted.
Treatment discontinuation?
On the issue of drug holidays and when or whether to stop therapy, as no therapies cure osteoporosis, medications should not be permanently stopped, even if bone density increases, NAMS recommends.
“By analogy, we do not stop diabetes therapy when A1c levels become normal,” Dr. McClung noted.
“Because the benefits of therapy on bone density and fracture protection wane, quickly for nonbisphosphonates and more slowly with bisphosphonates, short-term therapy, for instance 5 years, is not optimal treatment,” he said.
While the short-term interruption of bisphosphonate therapy may be considered in some patients, “the concept of ‘drug holidays’ does not pertain to nonbisphosphonate drugs,” Dr. McClung said.
NAMS adds that management of therapeutic choices should instead be ongoing.
“During therapy, reevaluate the treatment goals and the choice of medication on an ongoing basis through periodic medical examination and follow-up BMD testing,” NAMS recommends.
In terms of assessment, the measurement of bone mineral density while on treatment can gauge the current risk of fracture, and NAMS supports the use of the T-score at the hip as an appropriate clinical target in guiding choices of therapy.
Ultimately, “effective tools for diagnosing osteoporosis and assessing fracture risk are available, and well-studied strategies exist for managing bone health in women at both low and high risk of fracture,” NAMS concludes.
“By individualizing treatment approaches and monitoring and adjusting those approaches if the clinical picture changes, the consequences of osteoporosis on a menopausal woman’s activity and well-being can be minimized.”
Dr. McClung has reported receiving consulting fees from Amgen and Myovant, and honorarium for speaking from Amgen and Alexon. He serves on the boards of NAMS and the International Osteoporosis Foundation.
A version of this article first appeared on Medscape.com.
In the first revision to its guidance on the management of osteoporosis in a decade, the North American Menopause Society has issued an updated position statement addressing evolving evidence on osteoporosis issues ranging from screening and risk assessment to appropriate use of preventive therapy in postmenopausal women.
“Since the 2010 statement, there have been important new developments in our field, including better delineation of risk factors for fracture, resulting in better strategies for assessing fracture risk,” Michael R. McClung, MD, who is a NAMS board member and colead of the editorial panel for the 2021 position statement, told this news organization. Dr. McClung is also director emeritus of the Oregon Osteoporosis Center in Portland.
“There is much more information about the long-term safety of therapies,” he added. Dr. McClung also noted “the availability of four new drugs for the prevention and treatment of osteoporosis and clinical experience informing us of the effects of using different treatments in various sequences.”
Osteoporosis is substantially underdiagnosed and undertreated
A basis for the update, recently published in Menopause: The Journal of the North American Menopause Society, is the need to tackle the troubling fact that approximately half of postmenopausal women will experience a fracture related to osteoporosis in their lifetime, yet the condition is “substantially underdiagnosed and undertreated,” NAMS underscores.
With that in mind, osteoporosis should be considered by practitioners treating menopausal and postmenopausal women at all levels of care.
“All physicians and advanced care providers caring for postmenopausal women should be comfortable assessing and managing their patients with, or at risk for, fractures,” Dr. McClung added.
Osteoporosis prevention in young menopausal women
The NAMS statement covers a broad range of issues, and while most recommendations generally follow those of other societies’ guidelines, a unique aspect is the emphasis on preventing osteoporosis in young menopausal women with estrogen or other drugs.
While underscoring that all menopausal women should be encouraged to adopt healthy lifestyles, with good diets and physical activity to reduce the risk of bone loss and fractures, pharmacologic interventions also have a role, NAMS says.
Though long an issue of debate, NAMS voices support for estrogen therapy as having an important role in osteoporosis prevention, as estrogen deficiency is the principal cause of bone loss in postmenopausal women.
“Hormone therapy is the most appropriate choice to prevent bone loss at the time of menopause for healthy women, particularly those who have menopause symptoms,” the group states. Drug interventions are specifically supported in women with premature menopause, at least until the average age of natural menopause, in addition to those with low bone mineral density (BMD) (T-score < –1.0) and those experiencing relatively rapid bone loss related to acute estrogen deficiency in the menopause transition or on discontinuing estrogen therapy.
“Although using drugs to prevent osteoporosis is not included in national osteoporosis guidelines, a strong clinical argument can be made for doing so, especially in women who come to menopause with low bone mass,” the report states.
And therapy is also recommended if patients have a low BMD and other risk factors for fracture, such as family history, but do not meet the criteria for osteoporosis treatment.
Ultimately, clinicians should work with patients when deciding the options, Dr. McClung said.
“After carefully weighing the small risks associated with hormone therapy or other therapies begun at the time of menopause, menopause practitioners and their patients can and should make informed decisions about the use of Food and Drug Administration–approved medications to prevent osteoporosis in women who are at risk for developing that condition,” he noted, adding that his view on the matter is his own and not necessarily that of NAMS.
New treatments endorsed for high-risk patients to avoid ‘bone attack’
While most patients are treated for osteoporosis with antiremodeling drugs such as bisphosphonates and denosumab, NAMS endorses “a new paradigm of beginning treatment with a bone-building agent followed by an antiremodeling agent” for women at very high risk of fracture.
“Consider osteoanabolic therapies for patients at very high risk of fracture, including older women with recent fractures, T-scores –3.0 and lower, or multiple other risk factors,” the statement suggests.
Among those at highest risk are women who have sustained a first fracture.
“A recent fracture in a postmenopausal woman is the strongest risk factor for another fracture,” Dr. McClung said.
In fact, “having a fracture should be thought of and assessed as a ‘bone attack,’ ” he asserted.
Therapy is recommended in such cases to rapidly increase bone density and reduce their subsequent fracture risk.
“For these patients, osteoanabolic or bone-building agents are more effective than bisphosphonates and are recommended as initial therapy,” Dr. McClung noted.
Treatment discontinuation?
On the issue of drug holidays and when or whether to stop therapy, as no therapies cure osteoporosis, medications should not be permanently stopped, even if bone density increases, NAMS recommends.
“By analogy, we do not stop diabetes therapy when A1c levels become normal,” Dr. McClung noted.
“Because the benefits of therapy on bone density and fracture protection wane, quickly for nonbisphosphonates and more slowly with bisphosphonates, short-term therapy, for instance 5 years, is not optimal treatment,” he said.
While the short-term interruption of bisphosphonate therapy may be considered in some patients, “the concept of ‘drug holidays’ does not pertain to nonbisphosphonate drugs,” Dr. McClung said.
NAMS adds that management of therapeutic choices should instead be ongoing.
“During therapy, reevaluate the treatment goals and the choice of medication on an ongoing basis through periodic medical examination and follow-up BMD testing,” NAMS recommends.
In terms of assessment, the measurement of bone mineral density while on treatment can gauge the current risk of fracture, and NAMS supports the use of the T-score at the hip as an appropriate clinical target in guiding choices of therapy.
Ultimately, “effective tools for diagnosing osteoporosis and assessing fracture risk are available, and well-studied strategies exist for managing bone health in women at both low and high risk of fracture,” NAMS concludes.
“By individualizing treatment approaches and monitoring and adjusting those approaches if the clinical picture changes, the consequences of osteoporosis on a menopausal woman’s activity and well-being can be minimized.”
Dr. McClung has reported receiving consulting fees from Amgen and Myovant, and honorarium for speaking from Amgen and Alexon. He serves on the boards of NAMS and the International Osteoporosis Foundation.
A version of this article first appeared on Medscape.com.
In the first revision to its guidance on the management of osteoporosis in a decade, the North American Menopause Society has issued an updated position statement addressing evolving evidence on osteoporosis issues ranging from screening and risk assessment to appropriate use of preventive therapy in postmenopausal women.
“Since the 2010 statement, there have been important new developments in our field, including better delineation of risk factors for fracture, resulting in better strategies for assessing fracture risk,” Michael R. McClung, MD, who is a NAMS board member and colead of the editorial panel for the 2021 position statement, told this news organization. Dr. McClung is also director emeritus of the Oregon Osteoporosis Center in Portland.
“There is much more information about the long-term safety of therapies,” he added. Dr. McClung also noted “the availability of four new drugs for the prevention and treatment of osteoporosis and clinical experience informing us of the effects of using different treatments in various sequences.”
Osteoporosis is substantially underdiagnosed and undertreated
A basis for the update, recently published in Menopause: The Journal of the North American Menopause Society, is the need to tackle the troubling fact that approximately half of postmenopausal women will experience a fracture related to osteoporosis in their lifetime, yet the condition is “substantially underdiagnosed and undertreated,” NAMS underscores.
With that in mind, osteoporosis should be considered by practitioners treating menopausal and postmenopausal women at all levels of care.
“All physicians and advanced care providers caring for postmenopausal women should be comfortable assessing and managing their patients with, or at risk for, fractures,” Dr. McClung added.
Osteoporosis prevention in young menopausal women
The NAMS statement covers a broad range of issues, and while most recommendations generally follow those of other societies’ guidelines, a unique aspect is the emphasis on preventing osteoporosis in young menopausal women with estrogen or other drugs.
While underscoring that all menopausal women should be encouraged to adopt healthy lifestyles, with good diets and physical activity to reduce the risk of bone loss and fractures, pharmacologic interventions also have a role, NAMS says.
Though long an issue of debate, NAMS voices support for estrogen therapy as having an important role in osteoporosis prevention, as estrogen deficiency is the principal cause of bone loss in postmenopausal women.
“Hormone therapy is the most appropriate choice to prevent bone loss at the time of menopause for healthy women, particularly those who have menopause symptoms,” the group states. Drug interventions are specifically supported in women with premature menopause, at least until the average age of natural menopause, in addition to those with low bone mineral density (BMD) (T-score < –1.0) and those experiencing relatively rapid bone loss related to acute estrogen deficiency in the menopause transition or on discontinuing estrogen therapy.
“Although using drugs to prevent osteoporosis is not included in national osteoporosis guidelines, a strong clinical argument can be made for doing so, especially in women who come to menopause with low bone mass,” the report states.
And therapy is also recommended if patients have a low BMD and other risk factors for fracture, such as family history, but do not meet the criteria for osteoporosis treatment.
Ultimately, clinicians should work with patients when deciding the options, Dr. McClung said.
“After carefully weighing the small risks associated with hormone therapy or other therapies begun at the time of menopause, menopause practitioners and their patients can and should make informed decisions about the use of Food and Drug Administration–approved medications to prevent osteoporosis in women who are at risk for developing that condition,” he noted, adding that his view on the matter is his own and not necessarily that of NAMS.
New treatments endorsed for high-risk patients to avoid ‘bone attack’
While most patients are treated for osteoporosis with antiremodeling drugs such as bisphosphonates and denosumab, NAMS endorses “a new paradigm of beginning treatment with a bone-building agent followed by an antiremodeling agent” for women at very high risk of fracture.
“Consider osteoanabolic therapies for patients at very high risk of fracture, including older women with recent fractures, T-scores –3.0 and lower, or multiple other risk factors,” the statement suggests.
Among those at highest risk are women who have sustained a first fracture.
“A recent fracture in a postmenopausal woman is the strongest risk factor for another fracture,” Dr. McClung said.
In fact, “having a fracture should be thought of and assessed as a ‘bone attack,’ ” he asserted.
Therapy is recommended in such cases to rapidly increase bone density and reduce their subsequent fracture risk.
“For these patients, osteoanabolic or bone-building agents are more effective than bisphosphonates and are recommended as initial therapy,” Dr. McClung noted.
Treatment discontinuation?
On the issue of drug holidays and when or whether to stop therapy, as no therapies cure osteoporosis, medications should not be permanently stopped, even if bone density increases, NAMS recommends.
“By analogy, we do not stop diabetes therapy when A1c levels become normal,” Dr. McClung noted.
“Because the benefits of therapy on bone density and fracture protection wane, quickly for nonbisphosphonates and more slowly with bisphosphonates, short-term therapy, for instance 5 years, is not optimal treatment,” he said.
While the short-term interruption of bisphosphonate therapy may be considered in some patients, “the concept of ‘drug holidays’ does not pertain to nonbisphosphonate drugs,” Dr. McClung said.
NAMS adds that management of therapeutic choices should instead be ongoing.
“During therapy, reevaluate the treatment goals and the choice of medication on an ongoing basis through periodic medical examination and follow-up BMD testing,” NAMS recommends.
In terms of assessment, the measurement of bone mineral density while on treatment can gauge the current risk of fracture, and NAMS supports the use of the T-score at the hip as an appropriate clinical target in guiding choices of therapy.
Ultimately, “effective tools for diagnosing osteoporosis and assessing fracture risk are available, and well-studied strategies exist for managing bone health in women at both low and high risk of fracture,” NAMS concludes.
“By individualizing treatment approaches and monitoring and adjusting those approaches if the clinical picture changes, the consequences of osteoporosis on a menopausal woman’s activity and well-being can be minimized.”
Dr. McClung has reported receiving consulting fees from Amgen and Myovant, and honorarium for speaking from Amgen and Alexon. He serves on the boards of NAMS and the International Osteoporosis Foundation.
A version of this article first appeared on Medscape.com.
Vertebral fractures still a risk with low-dose oral glucocorticoids for RA
Patients with rheumatoid arthritis currently being treated with low doses of oral glucocorticoids (GCs) had a 59% increased risk of sustaining a vertebral fracture when compared with past users, results of a retrospective cohort study have shown.
Although the overall risk of an osteoporotic fracture was not increased when comparing current and past GC users, with a hazard ratio of 1.14 (95% confidence interval, 0.98-1.33), the HR for sustaining a spinal fracture was 1.59 (95% CI, 1.11-2.29).
“Clinicians should be aware that, even in RA patients who receive low daily glucocorticoid doses, the risk of clinical vertebral fracture is increased,” Shahab Abtahi, MD, of Maastricht (the Netherlands) University and coauthors reported in Rheumatology.
This is important considering around a quarter of RA patients are treated with GCs in the United Kingdom in accordance with European recommendations, they observed.
Conflicting randomized and observational findings on whether or not osteoporotic fractures might be linked to the use of low-dose GCs prompted Dr. Abtahi and associates to see if there were any signals in real-world data. To do so, they used data one of the world’s largest primary care databases – the Clinical Practice Research Datalink (CPRD), which consists of anonymized patient data from a network of primary care practices across the United Kingdom.
Altogether, the records of more than 15,000 patients with RA aged 50 years and older who were held in the CRPD between 1997 and 2017 were pulled for analysis, and just half (n = 7,039) were receiving or had received GC therapy. Low-dose GC therapy was defined as a prednisolone equivalent dose (PED) of 7.5 mg or less per day.
The use of low-dose GCs use during three key time periods was considered: within the past 6 months (current users), within the past 7-12 months (recent users), and within the past year (past users).
The analyses involved time-dependent Cox proportional-hazards models to look for associations between GC use and all types of osteoporotic fracture, including the risk for incident hip, vertebral, humeral, forearm, pelvis, and rib fractures. They were adjusted for various lifestyle parameters, comorbidities, and the use of other medications.
“Current GC use was further broken down into subcategories based on average daily and cumulative dose,” Dr. Abtahi observed. As might be expected, doses even lower than 7.5 mg or less PED did not increase the chance of any osteoporotic fracture but there was an increased risk for some types with higher average daily doses, notably at the hip and pelvis, as well as the spine.
“Low-dose oral GC therapy was associated with an increased risk of clinical vertebral fracture, while the risk of other individual OP fracture sites was not increased,” said the team, adding that the main results remained unchanged regardless of short- or long-term use.
“We know that vertebral fracture risk is markedly increased in RA, and it is well known that GC therapy in particular affects trabecular bone, which is abundantly present in lumbar vertebrae,” Dr. Abtahi wrote.
“Therefore, we can hypothesize that the beneficial effect of low-dose GC therapy on suppressing the background inflammation of RA could probably be enough to offset its negative effect on bone synthesis in most fracture sites but not in vertebrae,” they suggested.
One of the limitations of the study is that the researchers lacked data on the disease activity of the patients or if they were being treated with biologic therapy. This means that confounding by disease severity might be an issue with only those with higher disease activity being treated with GCs and thus were at higher risk for fractures.
“Another limitation was a potential misclassification of exposure with oral GCs, as we had only prescribing information from CPRD, which is roughly two steps behind actual drug use by patients,” the researchers conceded. The average duration of GC use was estimated at 3.7 years, which is an indication of actual use.
A detection bias may also be involved with regard to vertebral fractures, with complaints of back pain maybe being discussed more often when prescribing GCs, leading to more referrals for possible fracture assessment.
Dr. Abtahi and a fellow coauthor disclosed receiving research and other funding from several pharmaceutical companies unrelated to this study. All other coauthors had no conflicts of interest.
Patients with rheumatoid arthritis currently being treated with low doses of oral glucocorticoids (GCs) had a 59% increased risk of sustaining a vertebral fracture when compared with past users, results of a retrospective cohort study have shown.
Although the overall risk of an osteoporotic fracture was not increased when comparing current and past GC users, with a hazard ratio of 1.14 (95% confidence interval, 0.98-1.33), the HR for sustaining a spinal fracture was 1.59 (95% CI, 1.11-2.29).
“Clinicians should be aware that, even in RA patients who receive low daily glucocorticoid doses, the risk of clinical vertebral fracture is increased,” Shahab Abtahi, MD, of Maastricht (the Netherlands) University and coauthors reported in Rheumatology.
This is important considering around a quarter of RA patients are treated with GCs in the United Kingdom in accordance with European recommendations, they observed.
Conflicting randomized and observational findings on whether or not osteoporotic fractures might be linked to the use of low-dose GCs prompted Dr. Abtahi and associates to see if there were any signals in real-world data. To do so, they used data one of the world’s largest primary care databases – the Clinical Practice Research Datalink (CPRD), which consists of anonymized patient data from a network of primary care practices across the United Kingdom.
Altogether, the records of more than 15,000 patients with RA aged 50 years and older who were held in the CRPD between 1997 and 2017 were pulled for analysis, and just half (n = 7,039) were receiving or had received GC therapy. Low-dose GC therapy was defined as a prednisolone equivalent dose (PED) of 7.5 mg or less per day.
The use of low-dose GCs use during three key time periods was considered: within the past 6 months (current users), within the past 7-12 months (recent users), and within the past year (past users).
The analyses involved time-dependent Cox proportional-hazards models to look for associations between GC use and all types of osteoporotic fracture, including the risk for incident hip, vertebral, humeral, forearm, pelvis, and rib fractures. They were adjusted for various lifestyle parameters, comorbidities, and the use of other medications.
“Current GC use was further broken down into subcategories based on average daily and cumulative dose,” Dr. Abtahi observed. As might be expected, doses even lower than 7.5 mg or less PED did not increase the chance of any osteoporotic fracture but there was an increased risk for some types with higher average daily doses, notably at the hip and pelvis, as well as the spine.
“Low-dose oral GC therapy was associated with an increased risk of clinical vertebral fracture, while the risk of other individual OP fracture sites was not increased,” said the team, adding that the main results remained unchanged regardless of short- or long-term use.
“We know that vertebral fracture risk is markedly increased in RA, and it is well known that GC therapy in particular affects trabecular bone, which is abundantly present in lumbar vertebrae,” Dr. Abtahi wrote.
“Therefore, we can hypothesize that the beneficial effect of low-dose GC therapy on suppressing the background inflammation of RA could probably be enough to offset its negative effect on bone synthesis in most fracture sites but not in vertebrae,” they suggested.
One of the limitations of the study is that the researchers lacked data on the disease activity of the patients or if they were being treated with biologic therapy. This means that confounding by disease severity might be an issue with only those with higher disease activity being treated with GCs and thus were at higher risk for fractures.
“Another limitation was a potential misclassification of exposure with oral GCs, as we had only prescribing information from CPRD, which is roughly two steps behind actual drug use by patients,” the researchers conceded. The average duration of GC use was estimated at 3.7 years, which is an indication of actual use.
A detection bias may also be involved with regard to vertebral fractures, with complaints of back pain maybe being discussed more often when prescribing GCs, leading to more referrals for possible fracture assessment.
Dr. Abtahi and a fellow coauthor disclosed receiving research and other funding from several pharmaceutical companies unrelated to this study. All other coauthors had no conflicts of interest.
Patients with rheumatoid arthritis currently being treated with low doses of oral glucocorticoids (GCs) had a 59% increased risk of sustaining a vertebral fracture when compared with past users, results of a retrospective cohort study have shown.
Although the overall risk of an osteoporotic fracture was not increased when comparing current and past GC users, with a hazard ratio of 1.14 (95% confidence interval, 0.98-1.33), the HR for sustaining a spinal fracture was 1.59 (95% CI, 1.11-2.29).
“Clinicians should be aware that, even in RA patients who receive low daily glucocorticoid doses, the risk of clinical vertebral fracture is increased,” Shahab Abtahi, MD, of Maastricht (the Netherlands) University and coauthors reported in Rheumatology.
This is important considering around a quarter of RA patients are treated with GCs in the United Kingdom in accordance with European recommendations, they observed.
Conflicting randomized and observational findings on whether or not osteoporotic fractures might be linked to the use of low-dose GCs prompted Dr. Abtahi and associates to see if there were any signals in real-world data. To do so, they used data one of the world’s largest primary care databases – the Clinical Practice Research Datalink (CPRD), which consists of anonymized patient data from a network of primary care practices across the United Kingdom.
Altogether, the records of more than 15,000 patients with RA aged 50 years and older who were held in the CRPD between 1997 and 2017 were pulled for analysis, and just half (n = 7,039) were receiving or had received GC therapy. Low-dose GC therapy was defined as a prednisolone equivalent dose (PED) of 7.5 mg or less per day.
The use of low-dose GCs use during three key time periods was considered: within the past 6 months (current users), within the past 7-12 months (recent users), and within the past year (past users).
The analyses involved time-dependent Cox proportional-hazards models to look for associations between GC use and all types of osteoporotic fracture, including the risk for incident hip, vertebral, humeral, forearm, pelvis, and rib fractures. They were adjusted for various lifestyle parameters, comorbidities, and the use of other medications.
“Current GC use was further broken down into subcategories based on average daily and cumulative dose,” Dr. Abtahi observed. As might be expected, doses even lower than 7.5 mg or less PED did not increase the chance of any osteoporotic fracture but there was an increased risk for some types with higher average daily doses, notably at the hip and pelvis, as well as the spine.
“Low-dose oral GC therapy was associated with an increased risk of clinical vertebral fracture, while the risk of other individual OP fracture sites was not increased,” said the team, adding that the main results remained unchanged regardless of short- or long-term use.
“We know that vertebral fracture risk is markedly increased in RA, and it is well known that GC therapy in particular affects trabecular bone, which is abundantly present in lumbar vertebrae,” Dr. Abtahi wrote.
“Therefore, we can hypothesize that the beneficial effect of low-dose GC therapy on suppressing the background inflammation of RA could probably be enough to offset its negative effect on bone synthesis in most fracture sites but not in vertebrae,” they suggested.
One of the limitations of the study is that the researchers lacked data on the disease activity of the patients or if they were being treated with biologic therapy. This means that confounding by disease severity might be an issue with only those with higher disease activity being treated with GCs and thus were at higher risk for fractures.
“Another limitation was a potential misclassification of exposure with oral GCs, as we had only prescribing information from CPRD, which is roughly two steps behind actual drug use by patients,” the researchers conceded. The average duration of GC use was estimated at 3.7 years, which is an indication of actual use.
A detection bias may also be involved with regard to vertebral fractures, with complaints of back pain maybe being discussed more often when prescribing GCs, leading to more referrals for possible fracture assessment.
Dr. Abtahi and a fellow coauthor disclosed receiving research and other funding from several pharmaceutical companies unrelated to this study. All other coauthors had no conflicts of interest.
FROM RHEUMATOLOGY
Osteoporosis linked to increased risk of hearing loss
Women with osteoporosis, low bone density, or a previous vertebral fracture show significant increases in the risk of hearing loss compared to those without osteoporosis, according to a new study with more than 3 decades of follow-up.
The use of bisphosphonate therapy did not alter the risk, the researchers found.
“To the best of our knowledge, this is the first large longitudinal study to evaluate the relations of bone density, bisphosphonate use, fractures, and risk of hearing loss,” reported Sharon Curhan, MD, and colleagues in research published online in the Journal of the American Geriatric Society.
“In this large nationwide longitudinal study of nearly 144,000 women with up to 34 years of follow-up, we found that osteoporosis or low bone density was independently associated with higher risk of incident moderate or worse hearing loss,” the authors wrote.
“The magnitude of the elevated risk was similar among women who did and did not use bisphosphonates,” they added.
Participants were from the nurses’ health study and NHS II
With recent research suggesting a potential link between bisphosphonate use and prevention of noise-induced hearing loss in mice, Dr. Curhan, of the Channing Division of Network Medicine at Brigham and Women’s Hospital, Boston, and colleagues turned to the large longitudinal cohorts of the Nurses’ Health Study (NHS), conducted from 1982 to 2016, and the Nurses’ Health Study II (NHS II), from 1995 to 2017.
In total, the primary analysis included 60,821 women in the NHS and 83,078 in the NHS II.
Women in the NHS were aged 36-61 years at baseline and 70-95 years at the end of follow-up, while in the NHS II, women were aged 31-48 years at baseline and 53-70 years at the end of follow-up.
After multivariate adjustment for key factors including age, race/ethnicity, oral hormone use, and a variety of other factors, women in the NHS with osteoporosis had an increased risk of moderate or worse hearing loss, as self-reported every 2 years, compared to those without osteoporosis (relative risk, 1.14; 95% confidence interval, 1.09-1.19).
And in the NHS II, which also included data on low bone density, the risk of self-reported hearing loss was higher among those with osteoporosis or low bone density (RR, 1.30; 95% CI, 1.21-1.40).
No significant differences were observed in hearing loss risk based on whether women were treated with bisphosphonates, with the mean duration of use of the medication being 5.8 years in the NHS and 3.4 years in the NHS II.
Those who sustained a vertebral fracture also had a higher risk of hearing loss in both studies (NHS: RR, 1.31; NHS II: RR, 1.39).
However, the increased risk of hearing loss was not observed with hip fracture.
“Our findings of up to a 40% higher risk among women with vertebral fracture, but not hip fracture, were intriguing and merit further study,” the authors noted.
“The discordant findings between these skeletal sites may reflect differences in composition and metabolism of bones in the spine and hip and could provide insight into the pathophysiological changes in the ear that may lead to hearing loss,” they added.
Audiometric subanalysis
In an analysis of a subcohort of 3,749 women looking at audiometric thresholds for a more precise measure of hearing loss, women with osteoporosis or low bone density continued to show significantly worse hearing loss when treated with bisphosphonates compared to those without osteoporosis or low bone density.
However, there were no significant hearing loss differences among those with osteoporosis who did not take bisphosphonates versus those without osteoporosis.
The authors speculate that the use of bisphosphonates could have been indicative of more severe osteoporosis, hence the poorer audiometric thresholds.
In an interview, Dr. Curhan said the details of bisphosphonate use, such as type and duration, and their role in hearing loss should be further evaluated.
“Possibly, a potential influence of bisphosphonates on the relation of osteoporosis and hearing loss in humans may depend on the type, dose, and timing of bisphosphonate administration,” she observed. “This is an important question for further study.”
Mechanisms: Bone loss may extend to ear structures
In terms of the mechanisms linking osteoporosis itself to hearing loss, the authors noted that bone loss, in addition to compromising more prominent skeletal sites, could logically extend to bone-related structures in the ear.
“Bone mass at peripheral sites is correlated with bone mass at central sites, such as hip and spine, with correlation coefficients between 0.6 and 0.7,” they explained. “Plausibly, systemic bone demineralization could involve the temporal bone, the otic capsule, and the middle ear ossicles.”
They noted that hearing loss has been linked to other pathologic bone disorders, including otosclerosis and Paget disease.
Furthermore, imbalances in bone formation and resorption in osteoporosis may lead to alterations in ionic metabolism, which can lead to hearing loss.
Looking ahead, Dr. Curhan and colleagues plan to further examine whether calcium and vitamin D, which are associated with the prevention of osteoporosis, have a role in preventing hearing loss.
In the meantime, the findings underscore that clinicians treating patients with osteoporosis should routinely check patients’ hearing, Dr. Curhan said.
“Undetected and untreated hearing loss can adversely impact social interactions, physical and mental well-being, and daily life,” she said.
“Early detection of hearing loss offers greater opportunity for successful management and to learn strategies for rehabilitation and prevention of further progression.”
The study received support from the National Institutes of Health.
A version of this article first appeared on Medscape.com.
Women with osteoporosis, low bone density, or a previous vertebral fracture show significant increases in the risk of hearing loss compared to those without osteoporosis, according to a new study with more than 3 decades of follow-up.
The use of bisphosphonate therapy did not alter the risk, the researchers found.
“To the best of our knowledge, this is the first large longitudinal study to evaluate the relations of bone density, bisphosphonate use, fractures, and risk of hearing loss,” reported Sharon Curhan, MD, and colleagues in research published online in the Journal of the American Geriatric Society.
“In this large nationwide longitudinal study of nearly 144,000 women with up to 34 years of follow-up, we found that osteoporosis or low bone density was independently associated with higher risk of incident moderate or worse hearing loss,” the authors wrote.
“The magnitude of the elevated risk was similar among women who did and did not use bisphosphonates,” they added.
Participants were from the nurses’ health study and NHS II
With recent research suggesting a potential link between bisphosphonate use and prevention of noise-induced hearing loss in mice, Dr. Curhan, of the Channing Division of Network Medicine at Brigham and Women’s Hospital, Boston, and colleagues turned to the large longitudinal cohorts of the Nurses’ Health Study (NHS), conducted from 1982 to 2016, and the Nurses’ Health Study II (NHS II), from 1995 to 2017.
In total, the primary analysis included 60,821 women in the NHS and 83,078 in the NHS II.
Women in the NHS were aged 36-61 years at baseline and 70-95 years at the end of follow-up, while in the NHS II, women were aged 31-48 years at baseline and 53-70 years at the end of follow-up.
After multivariate adjustment for key factors including age, race/ethnicity, oral hormone use, and a variety of other factors, women in the NHS with osteoporosis had an increased risk of moderate or worse hearing loss, as self-reported every 2 years, compared to those without osteoporosis (relative risk, 1.14; 95% confidence interval, 1.09-1.19).
And in the NHS II, which also included data on low bone density, the risk of self-reported hearing loss was higher among those with osteoporosis or low bone density (RR, 1.30; 95% CI, 1.21-1.40).
No significant differences were observed in hearing loss risk based on whether women were treated with bisphosphonates, with the mean duration of use of the medication being 5.8 years in the NHS and 3.4 years in the NHS II.
Those who sustained a vertebral fracture also had a higher risk of hearing loss in both studies (NHS: RR, 1.31; NHS II: RR, 1.39).
However, the increased risk of hearing loss was not observed with hip fracture.
“Our findings of up to a 40% higher risk among women with vertebral fracture, but not hip fracture, were intriguing and merit further study,” the authors noted.
“The discordant findings between these skeletal sites may reflect differences in composition and metabolism of bones in the spine and hip and could provide insight into the pathophysiological changes in the ear that may lead to hearing loss,” they added.
Audiometric subanalysis
In an analysis of a subcohort of 3,749 women looking at audiometric thresholds for a more precise measure of hearing loss, women with osteoporosis or low bone density continued to show significantly worse hearing loss when treated with bisphosphonates compared to those without osteoporosis or low bone density.
However, there were no significant hearing loss differences among those with osteoporosis who did not take bisphosphonates versus those without osteoporosis.
The authors speculate that the use of bisphosphonates could have been indicative of more severe osteoporosis, hence the poorer audiometric thresholds.
In an interview, Dr. Curhan said the details of bisphosphonate use, such as type and duration, and their role in hearing loss should be further evaluated.
“Possibly, a potential influence of bisphosphonates on the relation of osteoporosis and hearing loss in humans may depend on the type, dose, and timing of bisphosphonate administration,” she observed. “This is an important question for further study.”
Mechanisms: Bone loss may extend to ear structures
In terms of the mechanisms linking osteoporosis itself to hearing loss, the authors noted that bone loss, in addition to compromising more prominent skeletal sites, could logically extend to bone-related structures in the ear.
“Bone mass at peripheral sites is correlated with bone mass at central sites, such as hip and spine, with correlation coefficients between 0.6 and 0.7,” they explained. “Plausibly, systemic bone demineralization could involve the temporal bone, the otic capsule, and the middle ear ossicles.”
They noted that hearing loss has been linked to other pathologic bone disorders, including otosclerosis and Paget disease.
Furthermore, imbalances in bone formation and resorption in osteoporosis may lead to alterations in ionic metabolism, which can lead to hearing loss.
Looking ahead, Dr. Curhan and colleagues plan to further examine whether calcium and vitamin D, which are associated with the prevention of osteoporosis, have a role in preventing hearing loss.
In the meantime, the findings underscore that clinicians treating patients with osteoporosis should routinely check patients’ hearing, Dr. Curhan said.
“Undetected and untreated hearing loss can adversely impact social interactions, physical and mental well-being, and daily life,” she said.
“Early detection of hearing loss offers greater opportunity for successful management and to learn strategies for rehabilitation and prevention of further progression.”
The study received support from the National Institutes of Health.
A version of this article first appeared on Medscape.com.
Women with osteoporosis, low bone density, or a previous vertebral fracture show significant increases in the risk of hearing loss compared to those without osteoporosis, according to a new study with more than 3 decades of follow-up.
The use of bisphosphonate therapy did not alter the risk, the researchers found.
“To the best of our knowledge, this is the first large longitudinal study to evaluate the relations of bone density, bisphosphonate use, fractures, and risk of hearing loss,” reported Sharon Curhan, MD, and colleagues in research published online in the Journal of the American Geriatric Society.
“In this large nationwide longitudinal study of nearly 144,000 women with up to 34 years of follow-up, we found that osteoporosis or low bone density was independently associated with higher risk of incident moderate or worse hearing loss,” the authors wrote.
“The magnitude of the elevated risk was similar among women who did and did not use bisphosphonates,” they added.
Participants were from the nurses’ health study and NHS II
With recent research suggesting a potential link between bisphosphonate use and prevention of noise-induced hearing loss in mice, Dr. Curhan, of the Channing Division of Network Medicine at Brigham and Women’s Hospital, Boston, and colleagues turned to the large longitudinal cohorts of the Nurses’ Health Study (NHS), conducted from 1982 to 2016, and the Nurses’ Health Study II (NHS II), from 1995 to 2017.
In total, the primary analysis included 60,821 women in the NHS and 83,078 in the NHS II.
Women in the NHS were aged 36-61 years at baseline and 70-95 years at the end of follow-up, while in the NHS II, women were aged 31-48 years at baseline and 53-70 years at the end of follow-up.
After multivariate adjustment for key factors including age, race/ethnicity, oral hormone use, and a variety of other factors, women in the NHS with osteoporosis had an increased risk of moderate or worse hearing loss, as self-reported every 2 years, compared to those without osteoporosis (relative risk, 1.14; 95% confidence interval, 1.09-1.19).
And in the NHS II, which also included data on low bone density, the risk of self-reported hearing loss was higher among those with osteoporosis or low bone density (RR, 1.30; 95% CI, 1.21-1.40).
No significant differences were observed in hearing loss risk based on whether women were treated with bisphosphonates, with the mean duration of use of the medication being 5.8 years in the NHS and 3.4 years in the NHS II.
Those who sustained a vertebral fracture also had a higher risk of hearing loss in both studies (NHS: RR, 1.31; NHS II: RR, 1.39).
However, the increased risk of hearing loss was not observed with hip fracture.
“Our findings of up to a 40% higher risk among women with vertebral fracture, but not hip fracture, were intriguing and merit further study,” the authors noted.
“The discordant findings between these skeletal sites may reflect differences in composition and metabolism of bones in the spine and hip and could provide insight into the pathophysiological changes in the ear that may lead to hearing loss,” they added.
Audiometric subanalysis
In an analysis of a subcohort of 3,749 women looking at audiometric thresholds for a more precise measure of hearing loss, women with osteoporosis or low bone density continued to show significantly worse hearing loss when treated with bisphosphonates compared to those without osteoporosis or low bone density.
However, there were no significant hearing loss differences among those with osteoporosis who did not take bisphosphonates versus those without osteoporosis.
The authors speculate that the use of bisphosphonates could have been indicative of more severe osteoporosis, hence the poorer audiometric thresholds.
In an interview, Dr. Curhan said the details of bisphosphonate use, such as type and duration, and their role in hearing loss should be further evaluated.
“Possibly, a potential influence of bisphosphonates on the relation of osteoporosis and hearing loss in humans may depend on the type, dose, and timing of bisphosphonate administration,” she observed. “This is an important question for further study.”
Mechanisms: Bone loss may extend to ear structures
In terms of the mechanisms linking osteoporosis itself to hearing loss, the authors noted that bone loss, in addition to compromising more prominent skeletal sites, could logically extend to bone-related structures in the ear.
“Bone mass at peripheral sites is correlated with bone mass at central sites, such as hip and spine, with correlation coefficients between 0.6 and 0.7,” they explained. “Plausibly, systemic bone demineralization could involve the temporal bone, the otic capsule, and the middle ear ossicles.”
They noted that hearing loss has been linked to other pathologic bone disorders, including otosclerosis and Paget disease.
Furthermore, imbalances in bone formation and resorption in osteoporosis may lead to alterations in ionic metabolism, which can lead to hearing loss.
Looking ahead, Dr. Curhan and colleagues plan to further examine whether calcium and vitamin D, which are associated with the prevention of osteoporosis, have a role in preventing hearing loss.
In the meantime, the findings underscore that clinicians treating patients with osteoporosis should routinely check patients’ hearing, Dr. Curhan said.
“Undetected and untreated hearing loss can adversely impact social interactions, physical and mental well-being, and daily life,” she said.
“Early detection of hearing loss offers greater opportunity for successful management and to learn strategies for rehabilitation and prevention of further progression.”
The study received support from the National Institutes of Health.
A version of this article first appeared on Medscape.com.
Any bone break increases risk for subsequent fracture in older women
No matter where an initial fracture occurs in a postmenopausal woman, there is a subsequent increased risk of another fracture, with the risk surprisingly highest in the youngest postmenopausal group and among certain minorities, new data indicate.
“To our knowledge, no previous prospective study has reported detailed patterns of subsequent fracture locations after initial fracture according to age strata among women in the U.S.,” the authors noted in their article, published online May 5, 2021, in EClinicalMedicine.
The results show that a first fracture of the lower arm or wrist; upper arm; or shoulder, upper leg, knee, lower leg, or ankle – as well as those of the hip or pelvis – were associated with an approximately three- to sixfold increased risk for subsequent fractures. The findings have important implications for clinicians, said lead author Carolyn J. Crandall, MD, professor of medicine at the University of California, Los Angeles.
“By not paying attention to which types of fractures increase the risk of future fractures, we are missing the opportunity to identify people at increased risk of future fracture and counsel them regarding risk reduction,” she said in a press statement.
Commenting on the research, Michael R. McClung, MD, stressed this message to clinicians needs to be underscored.
“This paper is one of a series of papers highlighting the fact that having a previous fracture is a risk factor for subsequent fractures,” he said in an interview.
“This has been known for a very long time, but it is a point still not appreciated by patients and primary care doctors, so having another study pointing this out is important,” emphasized Dr. McClung, of the Oregon Osteoporosis Center in Portland.
30% of women’s health initiative participants had a fracture
For the study, Dr. Crandall and colleagues evaluated data on 157,282 women between the ages of 50 and 79 who were enrolled in the Women’s Health Initiative between 1993 and 2018.
The women were a mean age of 63.1 years and 47,126 (30%) experienced an incident fracture during the study period.
With a mean follow-up of 15.4 years, each type of fracture was associated with an increased risk of a subsequent fracture after adjusting for age, race/ethnicity, body mass index, hormone therapy use, and other factors.
A wide range of initial risk fractures – including an initial lower arm or wrist fracture (adjusted hazard ratio, 4.80), upper arm or shoulder fracture (aHR, 5.06), upper leg fracture (aHR, 5.11), knee fracture (aHR, 5.03), lower leg/ankle fracture (aHR, 4.10), and spinal fracture (aHR, 6.69) – increased the risk of sustaining a subsequent hip fracture.
For initial fractures of the lower arm or wrist, there was an increased risk of a subsequent fracture of the upper arm/shoulder, upper leg, knee, lower leg/ankle, hip/pelvis, and spine (aHRs ranged from 2.63 to 5.68).
“The finding that knee fracture has the same prognostic value for subsequent fracture as hip or wrist fracture is a novel key finding, as knee fracture is generally not considered ‘osteoporotic’,” the authors noted.
The risk of fracture after sustaining an initial hip or pelvis fracture was exceptionally high – with as much as a 27-fold higher risk of a subsequent upper leg (nonhip) fracture (aHR, 27.18).
“Thirty-four percent of women who experienced initial hip or pelvis fracture experienced a subsequent nonhip fracture,” the authors noted.
However, the risks associated with an initial hip fracture are already well established, and the study’s more notable findings are the risks of other bone breaks, Dr. Crandall told this news organization.
“The (increased risk with hip fracture) is a rather substantial result,” she said. “However, the more major point of this study is that no matter where the initial fracture happened, the risk of the future fracture was elevated.”
Don’t disregard risks in younger women, racial/ethnic groups
The findings regarding age are also important. The highest risk was observed in the youngest postmenopausal age group of 50-59 years (aHR, 6.45), which decreased slightly in the 60- to 69-year age group (aHR, 6.04) and further decreased in the 70- to 79-year age group (aHR, 4.99).
“This was a surprise, and it highlights that clinicians should not disregard initial fractures among young postmenopausal women,” Dr. Crandall told this news organization.
Even greater increased risks for a subsequent fracture following an initial lower extremity fracture were observed in non-Hispanic Black women, Hispanic or Latina women, and women of Asian Pacific Islander ethnicity, ranging from ninefold to 14-fold, versus a sevenfold risk among non-Hispanic White women.
“This has public health implications because it means that we may have been missing the opportunity to prevent fractures among younger postmenopausal women and underrepresented racial/ethnic groups,” Dr. Crandall noted.
Is risk greatest 1-2 years after the initial fracture?
The findings suggest that current treatment guidelines may need to be revisited in light of inconsistencies regarding when, and for which fracture types, to initiate treatment.
“It will be important to determine whether existing risk calculators can be adapted (or new calculators developed) to help refine decision-making to determine which of the women with fractures other than hip or vertebral fractures should be treated,” the authors wrote.
Dr. McClung said a randomized, controlled trial of osteoporosis treatment in people who present with all types of fractures would help determine whether having a knee or a wrist fracture does indeed warrant such therapy.
He further commented that future studies should evaluate the shorter- versus longer-term risks.
“The most recent research suggests that the risk of having a second fracture is much higher in the first year or 2 after the first or incident fracture,” he observed. “So, the next stage in research with this dataset would be to ask not what happens over a 10-year time frame but what happens over the first year or 2 after the fracture.”
The study was funded by the National Heart, Lung, and Blood Institute. Dr. Crandall reported no relevant financial relationships. Dr. McClung reported being a consultant and on the speakers bureau for Amgen and being a speaker for Alexion.
A version of this article first appeared on Medscape.com.
No matter where an initial fracture occurs in a postmenopausal woman, there is a subsequent increased risk of another fracture, with the risk surprisingly highest in the youngest postmenopausal group and among certain minorities, new data indicate.
“To our knowledge, no previous prospective study has reported detailed patterns of subsequent fracture locations after initial fracture according to age strata among women in the U.S.,” the authors noted in their article, published online May 5, 2021, in EClinicalMedicine.
The results show that a first fracture of the lower arm or wrist; upper arm; or shoulder, upper leg, knee, lower leg, or ankle – as well as those of the hip or pelvis – were associated with an approximately three- to sixfold increased risk for subsequent fractures. The findings have important implications for clinicians, said lead author Carolyn J. Crandall, MD, professor of medicine at the University of California, Los Angeles.
“By not paying attention to which types of fractures increase the risk of future fractures, we are missing the opportunity to identify people at increased risk of future fracture and counsel them regarding risk reduction,” she said in a press statement.
Commenting on the research, Michael R. McClung, MD, stressed this message to clinicians needs to be underscored.
“This paper is one of a series of papers highlighting the fact that having a previous fracture is a risk factor for subsequent fractures,” he said in an interview.
“This has been known for a very long time, but it is a point still not appreciated by patients and primary care doctors, so having another study pointing this out is important,” emphasized Dr. McClung, of the Oregon Osteoporosis Center in Portland.
30% of women’s health initiative participants had a fracture
For the study, Dr. Crandall and colleagues evaluated data on 157,282 women between the ages of 50 and 79 who were enrolled in the Women’s Health Initiative between 1993 and 2018.
The women were a mean age of 63.1 years and 47,126 (30%) experienced an incident fracture during the study period.
With a mean follow-up of 15.4 years, each type of fracture was associated with an increased risk of a subsequent fracture after adjusting for age, race/ethnicity, body mass index, hormone therapy use, and other factors.
A wide range of initial risk fractures – including an initial lower arm or wrist fracture (adjusted hazard ratio, 4.80), upper arm or shoulder fracture (aHR, 5.06), upper leg fracture (aHR, 5.11), knee fracture (aHR, 5.03), lower leg/ankle fracture (aHR, 4.10), and spinal fracture (aHR, 6.69) – increased the risk of sustaining a subsequent hip fracture.
For initial fractures of the lower arm or wrist, there was an increased risk of a subsequent fracture of the upper arm/shoulder, upper leg, knee, lower leg/ankle, hip/pelvis, and spine (aHRs ranged from 2.63 to 5.68).
“The finding that knee fracture has the same prognostic value for subsequent fracture as hip or wrist fracture is a novel key finding, as knee fracture is generally not considered ‘osteoporotic’,” the authors noted.
The risk of fracture after sustaining an initial hip or pelvis fracture was exceptionally high – with as much as a 27-fold higher risk of a subsequent upper leg (nonhip) fracture (aHR, 27.18).
“Thirty-four percent of women who experienced initial hip or pelvis fracture experienced a subsequent nonhip fracture,” the authors noted.
However, the risks associated with an initial hip fracture are already well established, and the study’s more notable findings are the risks of other bone breaks, Dr. Crandall told this news organization.
“The (increased risk with hip fracture) is a rather substantial result,” she said. “However, the more major point of this study is that no matter where the initial fracture happened, the risk of the future fracture was elevated.”
Don’t disregard risks in younger women, racial/ethnic groups
The findings regarding age are also important. The highest risk was observed in the youngest postmenopausal age group of 50-59 years (aHR, 6.45), which decreased slightly in the 60- to 69-year age group (aHR, 6.04) and further decreased in the 70- to 79-year age group (aHR, 4.99).
“This was a surprise, and it highlights that clinicians should not disregard initial fractures among young postmenopausal women,” Dr. Crandall told this news organization.
Even greater increased risks for a subsequent fracture following an initial lower extremity fracture were observed in non-Hispanic Black women, Hispanic or Latina women, and women of Asian Pacific Islander ethnicity, ranging from ninefold to 14-fold, versus a sevenfold risk among non-Hispanic White women.
“This has public health implications because it means that we may have been missing the opportunity to prevent fractures among younger postmenopausal women and underrepresented racial/ethnic groups,” Dr. Crandall noted.
Is risk greatest 1-2 years after the initial fracture?
The findings suggest that current treatment guidelines may need to be revisited in light of inconsistencies regarding when, and for which fracture types, to initiate treatment.
“It will be important to determine whether existing risk calculators can be adapted (or new calculators developed) to help refine decision-making to determine which of the women with fractures other than hip or vertebral fractures should be treated,” the authors wrote.
Dr. McClung said a randomized, controlled trial of osteoporosis treatment in people who present with all types of fractures would help determine whether having a knee or a wrist fracture does indeed warrant such therapy.
He further commented that future studies should evaluate the shorter- versus longer-term risks.
“The most recent research suggests that the risk of having a second fracture is much higher in the first year or 2 after the first or incident fracture,” he observed. “So, the next stage in research with this dataset would be to ask not what happens over a 10-year time frame but what happens over the first year or 2 after the fracture.”
The study was funded by the National Heart, Lung, and Blood Institute. Dr. Crandall reported no relevant financial relationships. Dr. McClung reported being a consultant and on the speakers bureau for Amgen and being a speaker for Alexion.
A version of this article first appeared on Medscape.com.
No matter where an initial fracture occurs in a postmenopausal woman, there is a subsequent increased risk of another fracture, with the risk surprisingly highest in the youngest postmenopausal group and among certain minorities, new data indicate.
“To our knowledge, no previous prospective study has reported detailed patterns of subsequent fracture locations after initial fracture according to age strata among women in the U.S.,” the authors noted in their article, published online May 5, 2021, in EClinicalMedicine.
The results show that a first fracture of the lower arm or wrist; upper arm; or shoulder, upper leg, knee, lower leg, or ankle – as well as those of the hip or pelvis – were associated with an approximately three- to sixfold increased risk for subsequent fractures. The findings have important implications for clinicians, said lead author Carolyn J. Crandall, MD, professor of medicine at the University of California, Los Angeles.
“By not paying attention to which types of fractures increase the risk of future fractures, we are missing the opportunity to identify people at increased risk of future fracture and counsel them regarding risk reduction,” she said in a press statement.
Commenting on the research, Michael R. McClung, MD, stressed this message to clinicians needs to be underscored.
“This paper is one of a series of papers highlighting the fact that having a previous fracture is a risk factor for subsequent fractures,” he said in an interview.
“This has been known for a very long time, but it is a point still not appreciated by patients and primary care doctors, so having another study pointing this out is important,” emphasized Dr. McClung, of the Oregon Osteoporosis Center in Portland.
30% of women’s health initiative participants had a fracture
For the study, Dr. Crandall and colleagues evaluated data on 157,282 women between the ages of 50 and 79 who were enrolled in the Women’s Health Initiative between 1993 and 2018.
The women were a mean age of 63.1 years and 47,126 (30%) experienced an incident fracture during the study period.
With a mean follow-up of 15.4 years, each type of fracture was associated with an increased risk of a subsequent fracture after adjusting for age, race/ethnicity, body mass index, hormone therapy use, and other factors.
A wide range of initial risk fractures – including an initial lower arm or wrist fracture (adjusted hazard ratio, 4.80), upper arm or shoulder fracture (aHR, 5.06), upper leg fracture (aHR, 5.11), knee fracture (aHR, 5.03), lower leg/ankle fracture (aHR, 4.10), and spinal fracture (aHR, 6.69) – increased the risk of sustaining a subsequent hip fracture.
For initial fractures of the lower arm or wrist, there was an increased risk of a subsequent fracture of the upper arm/shoulder, upper leg, knee, lower leg/ankle, hip/pelvis, and spine (aHRs ranged from 2.63 to 5.68).
“The finding that knee fracture has the same prognostic value for subsequent fracture as hip or wrist fracture is a novel key finding, as knee fracture is generally not considered ‘osteoporotic’,” the authors noted.
The risk of fracture after sustaining an initial hip or pelvis fracture was exceptionally high – with as much as a 27-fold higher risk of a subsequent upper leg (nonhip) fracture (aHR, 27.18).
“Thirty-four percent of women who experienced initial hip or pelvis fracture experienced a subsequent nonhip fracture,” the authors noted.
However, the risks associated with an initial hip fracture are already well established, and the study’s more notable findings are the risks of other bone breaks, Dr. Crandall told this news organization.
“The (increased risk with hip fracture) is a rather substantial result,” she said. “However, the more major point of this study is that no matter where the initial fracture happened, the risk of the future fracture was elevated.”
Don’t disregard risks in younger women, racial/ethnic groups
The findings regarding age are also important. The highest risk was observed in the youngest postmenopausal age group of 50-59 years (aHR, 6.45), which decreased slightly in the 60- to 69-year age group (aHR, 6.04) and further decreased in the 70- to 79-year age group (aHR, 4.99).
“This was a surprise, and it highlights that clinicians should not disregard initial fractures among young postmenopausal women,” Dr. Crandall told this news organization.
Even greater increased risks for a subsequent fracture following an initial lower extremity fracture were observed in non-Hispanic Black women, Hispanic or Latina women, and women of Asian Pacific Islander ethnicity, ranging from ninefold to 14-fold, versus a sevenfold risk among non-Hispanic White women.
“This has public health implications because it means that we may have been missing the opportunity to prevent fractures among younger postmenopausal women and underrepresented racial/ethnic groups,” Dr. Crandall noted.
Is risk greatest 1-2 years after the initial fracture?
The findings suggest that current treatment guidelines may need to be revisited in light of inconsistencies regarding when, and for which fracture types, to initiate treatment.
“It will be important to determine whether existing risk calculators can be adapted (or new calculators developed) to help refine decision-making to determine which of the women with fractures other than hip or vertebral fractures should be treated,” the authors wrote.
Dr. McClung said a randomized, controlled trial of osteoporosis treatment in people who present with all types of fractures would help determine whether having a knee or a wrist fracture does indeed warrant such therapy.
He further commented that future studies should evaluate the shorter- versus longer-term risks.
“The most recent research suggests that the risk of having a second fracture is much higher in the first year or 2 after the first or incident fracture,” he observed. “So, the next stage in research with this dataset would be to ask not what happens over a 10-year time frame but what happens over the first year or 2 after the fracture.”
The study was funded by the National Heart, Lung, and Blood Institute. Dr. Crandall reported no relevant financial relationships. Dr. McClung reported being a consultant and on the speakers bureau for Amgen and being a speaker for Alexion.
A version of this article first appeared on Medscape.com.
Don’t screen for vitamin D in general population, says USPSTF
Seven years after concluding that evidence was insufficient to recommend screening for vitamin D deficiency in the general population, the United States Preventive Services Task Force (USPSTF) has revisited the issue – and come up with the same conclusion.
Overall, “the current evidence is inadequate to determine whether screening for and treatment of asymptomatic low 25(OH)D levels improve clinical outcomes in community dwelling adults,” the task force concluded in its statement, recommending an “I” for insufficient.
The statement was published online April 13 in JAMA.
In the absence of screening recommendations, clinicians may be best advised to instead focus on diet and supplementation for those considered at risk, said Anne R. Cappola, MD, of the University of Pennsylvania, Philadelphia.
“Rather than posing the question of screening the general population for vitamin D deficiency, let’s focus on ensuring that everyone consumes the age-based recommended daily allowance of vitamin D instead,” Dr. Cappola, a coauthor of the accompanying editorial, said in an interview.
No studies have directly evaluated benefits of screening
The latest USPSTF recommendation is based on a systematic review of the benefits and harms of screening and early treatment for vitamin D deficiency in asymptomatic, community-dwelling nonpregnant adults aged 18 or older in the primary care setting with no signs or symptoms of deficiency.
The review found no studies that directly evaluated the benefits of screening for vitamin D deficiency.
However, 26 randomized clinical trials and one nested case-control study evaluated the effectiveness of treatment of vitamin D deficiency with supplementation.
And while observational studies have linked lower vitamin D levels with a multitude of conditions and risks, evidence of any benefit was inconsistent, with none identified for most major outcomes in asymptomatic adults – the focus of the Task Force recommendation.
“Among asymptomatic, community-dwelling populations with low vitamin D levels, the evidence suggests that treatment with vitamin D has no effect on mortality or the incidence of fractures, falls, depression, diabetes, cardiovascular disease, cancer, or adverse events,” the review authors stress.
“The evidence is inconclusive about the effect of treatment on physical functioning and infection.”
One in four are vitamin D deficient
In terms of the further question of the potential harms of vitamin D screening of asymptomatic individuals, a key concern is the potential for misclassification and over- or underdiagnosis due to inconsistent cutoffs and variability of different screening assays, the review concluded.
However, with the rare exception of vitamin D toxicity from supplementation well above sufficient levels, treatment with vitamin D supplementation appears relatively safe.
With a lack of consensus even over the basic cutoff for vitamin D deficiency, the National Academy of Medicine determined in 2011 that hydroxyvitamin D (25[OH]D) levels below 20 ng/mL are deficient for bone health, with no evidence of different thresholds for any other health condition.
Based on that cutoff, the National Health and Nutrition Examination Survey (NHANES), reported in 2014 that 25% of the U.S. population over the age of 1 was vitamin D deficient, with 18% of the population having 25(OH)D levels of 12-19 ng/mL and 5% having very low levels (< 12 ng/mL).
More work needed to determine groups at risk
While the task force report did not delve into testing or treatment recommendations for symptomatic adults, key established risk factors that may help clinicians identify those who are vitamin D deficient include obesity, receiving little or no UVB light exposure, and older age.
In general, obesity is associated with a 1.3- to 2-fold risk of being vitamin D deficient based on the criteria used, while non-Hispanic Blacks are 2-10 times more likely to be deficient compared with non-Hispanic White patients, the task force noted.
However, the implications of vitamin D deficiency in certain populations can vary. For instance, non-Hispanic Black people, despite having a higher prevalence of lower vitamin D levels compared with White people, in fact, have lower reported rates of fractures.
To address the various issues and gain a better understanding of the complexities of vitamin D deficiency, the task force calls for further research in key areas.
“More research is needed to determine whether total serum 25(OH)D levels are the best measure of vitamin D deficiency and whether the best measure of vitamin D deficiency varies by subgroups defined by race, ethnicity, or sex,” the authors indicated.
Furthermore, “more research is needed to determine the cutoff that defines vitamin D deficiency and whether that cutoff varies by specific clinical outcome or by subgroups defined by race, ethnicity, or sex.”
No support for population-based screening in guidelines
With the lack of conclusive evidence, no organizations currently recommend population-based screening for vitamin D deficiency in asymptomatic patients, and the American Society for Clinical Pathology endorses this stance.
The Endocrine Society and the American Association of Clinical Endocrinologists meanwhile do recommend screening for vitamin D deficiency in patients considered at risk.
Data show there was as much as an 80-fold increase in Medicare reimbursement volumes for vitamin D testing among clinicians from 2000 to 2010; however, that rate may have leveled off after the National Academy of Medicine reported on set deficiency levels, said Sherri-Ann M. Burnett-Bowie, MD, MPH, Dr. Cappola’s editorial coauthor.
Dr. Burnett-Bowie noted that she regularly tests her patients’ vitamin D levels, however most of her patients have osteoporosis or fractures.
“I do screen them for vitamin D deficiency since optimizing their vitamin D will improve calcium absorption, which is important for treating their osteoporosis,” Dr. Burnett-Bowie, of the endocrine division, department of medicine, Massachusetts General Hospital, Boston, said in an interview.
In terms of broader testing of asymptomatic patients in the general population, however, any changes in screening will likely be contingent on developments in the effects of treatment, she said.
“Given the challenge in finding benefits of vitamin D supplementation in those who are deficient, it will likely be more challenging to find benefits from wider screening,” she concluded.
The USPSTF and editorialists reported having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Seven years after concluding that evidence was insufficient to recommend screening for vitamin D deficiency in the general population, the United States Preventive Services Task Force (USPSTF) has revisited the issue – and come up with the same conclusion.
Overall, “the current evidence is inadequate to determine whether screening for and treatment of asymptomatic low 25(OH)D levels improve clinical outcomes in community dwelling adults,” the task force concluded in its statement, recommending an “I” for insufficient.
The statement was published online April 13 in JAMA.
In the absence of screening recommendations, clinicians may be best advised to instead focus on diet and supplementation for those considered at risk, said Anne R. Cappola, MD, of the University of Pennsylvania, Philadelphia.
“Rather than posing the question of screening the general population for vitamin D deficiency, let’s focus on ensuring that everyone consumes the age-based recommended daily allowance of vitamin D instead,” Dr. Cappola, a coauthor of the accompanying editorial, said in an interview.
No studies have directly evaluated benefits of screening
The latest USPSTF recommendation is based on a systematic review of the benefits and harms of screening and early treatment for vitamin D deficiency in asymptomatic, community-dwelling nonpregnant adults aged 18 or older in the primary care setting with no signs or symptoms of deficiency.
The review found no studies that directly evaluated the benefits of screening for vitamin D deficiency.
However, 26 randomized clinical trials and one nested case-control study evaluated the effectiveness of treatment of vitamin D deficiency with supplementation.
And while observational studies have linked lower vitamin D levels with a multitude of conditions and risks, evidence of any benefit was inconsistent, with none identified for most major outcomes in asymptomatic adults – the focus of the Task Force recommendation.
“Among asymptomatic, community-dwelling populations with low vitamin D levels, the evidence suggests that treatment with vitamin D has no effect on mortality or the incidence of fractures, falls, depression, diabetes, cardiovascular disease, cancer, or adverse events,” the review authors stress.
“The evidence is inconclusive about the effect of treatment on physical functioning and infection.”
One in four are vitamin D deficient
In terms of the further question of the potential harms of vitamin D screening of asymptomatic individuals, a key concern is the potential for misclassification and over- or underdiagnosis due to inconsistent cutoffs and variability of different screening assays, the review concluded.
However, with the rare exception of vitamin D toxicity from supplementation well above sufficient levels, treatment with vitamin D supplementation appears relatively safe.
With a lack of consensus even over the basic cutoff for vitamin D deficiency, the National Academy of Medicine determined in 2011 that hydroxyvitamin D (25[OH]D) levels below 20 ng/mL are deficient for bone health, with no evidence of different thresholds for any other health condition.
Based on that cutoff, the National Health and Nutrition Examination Survey (NHANES), reported in 2014 that 25% of the U.S. population over the age of 1 was vitamin D deficient, with 18% of the population having 25(OH)D levels of 12-19 ng/mL and 5% having very low levels (< 12 ng/mL).
More work needed to determine groups at risk
While the task force report did not delve into testing or treatment recommendations for symptomatic adults, key established risk factors that may help clinicians identify those who are vitamin D deficient include obesity, receiving little or no UVB light exposure, and older age.
In general, obesity is associated with a 1.3- to 2-fold risk of being vitamin D deficient based on the criteria used, while non-Hispanic Blacks are 2-10 times more likely to be deficient compared with non-Hispanic White patients, the task force noted.
However, the implications of vitamin D deficiency in certain populations can vary. For instance, non-Hispanic Black people, despite having a higher prevalence of lower vitamin D levels compared with White people, in fact, have lower reported rates of fractures.
To address the various issues and gain a better understanding of the complexities of vitamin D deficiency, the task force calls for further research in key areas.
“More research is needed to determine whether total serum 25(OH)D levels are the best measure of vitamin D deficiency and whether the best measure of vitamin D deficiency varies by subgroups defined by race, ethnicity, or sex,” the authors indicated.
Furthermore, “more research is needed to determine the cutoff that defines vitamin D deficiency and whether that cutoff varies by specific clinical outcome or by subgroups defined by race, ethnicity, or sex.”
No support for population-based screening in guidelines
With the lack of conclusive evidence, no organizations currently recommend population-based screening for vitamin D deficiency in asymptomatic patients, and the American Society for Clinical Pathology endorses this stance.
The Endocrine Society and the American Association of Clinical Endocrinologists meanwhile do recommend screening for vitamin D deficiency in patients considered at risk.
Data show there was as much as an 80-fold increase in Medicare reimbursement volumes for vitamin D testing among clinicians from 2000 to 2010; however, that rate may have leveled off after the National Academy of Medicine reported on set deficiency levels, said Sherri-Ann M. Burnett-Bowie, MD, MPH, Dr. Cappola’s editorial coauthor.
Dr. Burnett-Bowie noted that she regularly tests her patients’ vitamin D levels, however most of her patients have osteoporosis or fractures.
“I do screen them for vitamin D deficiency since optimizing their vitamin D will improve calcium absorption, which is important for treating their osteoporosis,” Dr. Burnett-Bowie, of the endocrine division, department of medicine, Massachusetts General Hospital, Boston, said in an interview.
In terms of broader testing of asymptomatic patients in the general population, however, any changes in screening will likely be contingent on developments in the effects of treatment, she said.
“Given the challenge in finding benefits of vitamin D supplementation in those who are deficient, it will likely be more challenging to find benefits from wider screening,” she concluded.
The USPSTF and editorialists reported having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Seven years after concluding that evidence was insufficient to recommend screening for vitamin D deficiency in the general population, the United States Preventive Services Task Force (USPSTF) has revisited the issue – and come up with the same conclusion.
Overall, “the current evidence is inadequate to determine whether screening for and treatment of asymptomatic low 25(OH)D levels improve clinical outcomes in community dwelling adults,” the task force concluded in its statement, recommending an “I” for insufficient.
The statement was published online April 13 in JAMA.
In the absence of screening recommendations, clinicians may be best advised to instead focus on diet and supplementation for those considered at risk, said Anne R. Cappola, MD, of the University of Pennsylvania, Philadelphia.
“Rather than posing the question of screening the general population for vitamin D deficiency, let’s focus on ensuring that everyone consumes the age-based recommended daily allowance of vitamin D instead,” Dr. Cappola, a coauthor of the accompanying editorial, said in an interview.
No studies have directly evaluated benefits of screening
The latest USPSTF recommendation is based on a systematic review of the benefits and harms of screening and early treatment for vitamin D deficiency in asymptomatic, community-dwelling nonpregnant adults aged 18 or older in the primary care setting with no signs or symptoms of deficiency.
The review found no studies that directly evaluated the benefits of screening for vitamin D deficiency.
However, 26 randomized clinical trials and one nested case-control study evaluated the effectiveness of treatment of vitamin D deficiency with supplementation.
And while observational studies have linked lower vitamin D levels with a multitude of conditions and risks, evidence of any benefit was inconsistent, with none identified for most major outcomes in asymptomatic adults – the focus of the Task Force recommendation.
“Among asymptomatic, community-dwelling populations with low vitamin D levels, the evidence suggests that treatment with vitamin D has no effect on mortality or the incidence of fractures, falls, depression, diabetes, cardiovascular disease, cancer, or adverse events,” the review authors stress.
“The evidence is inconclusive about the effect of treatment on physical functioning and infection.”
One in four are vitamin D deficient
In terms of the further question of the potential harms of vitamin D screening of asymptomatic individuals, a key concern is the potential for misclassification and over- or underdiagnosis due to inconsistent cutoffs and variability of different screening assays, the review concluded.
However, with the rare exception of vitamin D toxicity from supplementation well above sufficient levels, treatment with vitamin D supplementation appears relatively safe.
With a lack of consensus even over the basic cutoff for vitamin D deficiency, the National Academy of Medicine determined in 2011 that hydroxyvitamin D (25[OH]D) levels below 20 ng/mL are deficient for bone health, with no evidence of different thresholds for any other health condition.
Based on that cutoff, the National Health and Nutrition Examination Survey (NHANES), reported in 2014 that 25% of the U.S. population over the age of 1 was vitamin D deficient, with 18% of the population having 25(OH)D levels of 12-19 ng/mL and 5% having very low levels (< 12 ng/mL).
More work needed to determine groups at risk
While the task force report did not delve into testing or treatment recommendations for symptomatic adults, key established risk factors that may help clinicians identify those who are vitamin D deficient include obesity, receiving little or no UVB light exposure, and older age.
In general, obesity is associated with a 1.3- to 2-fold risk of being vitamin D deficient based on the criteria used, while non-Hispanic Blacks are 2-10 times more likely to be deficient compared with non-Hispanic White patients, the task force noted.
However, the implications of vitamin D deficiency in certain populations can vary. For instance, non-Hispanic Black people, despite having a higher prevalence of lower vitamin D levels compared with White people, in fact, have lower reported rates of fractures.
To address the various issues and gain a better understanding of the complexities of vitamin D deficiency, the task force calls for further research in key areas.
“More research is needed to determine whether total serum 25(OH)D levels are the best measure of vitamin D deficiency and whether the best measure of vitamin D deficiency varies by subgroups defined by race, ethnicity, or sex,” the authors indicated.
Furthermore, “more research is needed to determine the cutoff that defines vitamin D deficiency and whether that cutoff varies by specific clinical outcome or by subgroups defined by race, ethnicity, or sex.”
No support for population-based screening in guidelines
With the lack of conclusive evidence, no organizations currently recommend population-based screening for vitamin D deficiency in asymptomatic patients, and the American Society for Clinical Pathology endorses this stance.
The Endocrine Society and the American Association of Clinical Endocrinologists meanwhile do recommend screening for vitamin D deficiency in patients considered at risk.
Data show there was as much as an 80-fold increase in Medicare reimbursement volumes for vitamin D testing among clinicians from 2000 to 2010; however, that rate may have leveled off after the National Academy of Medicine reported on set deficiency levels, said Sherri-Ann M. Burnett-Bowie, MD, MPH, Dr. Cappola’s editorial coauthor.
Dr. Burnett-Bowie noted that she regularly tests her patients’ vitamin D levels, however most of her patients have osteoporosis or fractures.
“I do screen them for vitamin D deficiency since optimizing their vitamin D will improve calcium absorption, which is important for treating their osteoporosis,” Dr. Burnett-Bowie, of the endocrine division, department of medicine, Massachusetts General Hospital, Boston, said in an interview.
In terms of broader testing of asymptomatic patients in the general population, however, any changes in screening will likely be contingent on developments in the effects of treatment, she said.
“Given the challenge in finding benefits of vitamin D supplementation in those who are deficient, it will likely be more challenging to find benefits from wider screening,” she concluded.
The USPSTF and editorialists reported having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
COVID-19 vaccination in RMD patients: Safety data “reassuring”
Two reports support the safety and immunogenicity of SARS-CoV-2 mRNA vaccines in patients with rheumatic and musculoskeletal diseases (RMDs) and represent the first available data on such patients.
In an observational cohort study published in Annals of the Rheumatic Diseases, Caoilfhionn M. Connolly, MD, of Johns Hopkins University, Baltimore, and colleagues reviewed data from 325 adults with RMDs who received the first dose of SARS-CoV-2 mRNA vaccine during the period of Dec. 17, 2020, to Feb. 11, 2021. Of these, 51% received the Pfizer/BioNTech vaccine and 49% received the Moderna vaccine.
The patients, who were invited to participate on social media, were aged 34-54 years, 96% were women, and 89% were White. Inflammatory arthritis was the most common RMD condition (38%), followed by systemic lupus erythematosus (28%) and overlap connective tissue disease (19%). The patients were using a range of immunomodulatory treatment regimens, including nonbiologic disease modifying antirheumatic drugs (DMARDs) in 44%, biologics in 19%, and combination therapy in 37%.
Overall, 89% of patients reported localized symptoms of pain, swelling, and erythema, and 69% reported systemic symptoms. Fatigue was the most common systemic symptom, and 7.4% reported severe fatigue.
None of the patients experienced allergic reactions requiring epinephrine, and 3% reported new infections that required treatment.
“These early, reassuring results may ameliorate concern among patients and provide guidance for rheumatology providers in critical discussions regarding vaccine hesitancy or refusal,” they concluded.
Antibody responses
In another study published in Annals of the Rheumatic Diseases by the same group of researchers, antibody responses against the receptor binding domain of the SARS-CoV-2 spike protein were seen in 74% of 123 adults with an RMD at 18-26 days after receiving a first dose of SARS-CoV-2 mRNA vaccine (52% Pfizer vaccine and 48% Moderna) between Jan. 8, 2021, and Feb. 12, 2021.
The most common diagnoses in these patients were inflammatory arthritis (28%), systemic lupus erythematosus (20%), and Sjögren’s syndrome (13%). A total of 28% of participants reported taking no immunomodulatory agents, 19% reported nonbiologic DMARDs, 14% reported biologic DMARDs, and 19% reported combination therapy.
Although no differences appeared based on disease groups or overall categories of immunomodulatory therapies, patients whose treatment included mycophenolate or rituximab were significantly less likely to develop antibody responses than were patients not taking these medications (P = .001 and P = .04, respectively). Although rituximab and methotrexate have been associated with reduced responses to vaccines such as the flu vaccine, methotrexate was not associated with reduced vaccine response in this study. A total of 94% of patients taking a tumor necrosis factor inhibitor had detectable antibodies.
The studies’ findings were limited by several factors including a lack of longer-term safety data; the small, nonrandomized sample of mainly white women; limited information on immunomodulatory drug dosage and timing; lack of serial antibody measurements; use of an enzyme immunoassay designed to detect antibody response after natural infection; and the inclusion of data only on the first dose of a two-dose vaccine series, the researchers noted. However, the data should provide additional reassurance to RMD patients and their health care teams about vaccination against COVID-19, they said.
Both studies were supported by the Ben-Dov family. In addition, the studies were supported by grants to various study authors from the National Institute of Diabetes and Digestive and Kidney Diseases, the National Institute of Allergy and Infectious Diseases, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, and the Transplantation and Immunology Research Network of the American Society of Transplantation. One author disclosed financial relationships with Sanofi, Novartis, CSL Behring, Jazz Pharmaceuticals, Veloxis, Mallinckrodt, and Thermo Fisher Scientific. The other researchers had no financial conflicts to disclose.
Two reports support the safety and immunogenicity of SARS-CoV-2 mRNA vaccines in patients with rheumatic and musculoskeletal diseases (RMDs) and represent the first available data on such patients.
In an observational cohort study published in Annals of the Rheumatic Diseases, Caoilfhionn M. Connolly, MD, of Johns Hopkins University, Baltimore, and colleagues reviewed data from 325 adults with RMDs who received the first dose of SARS-CoV-2 mRNA vaccine during the period of Dec. 17, 2020, to Feb. 11, 2021. Of these, 51% received the Pfizer/BioNTech vaccine and 49% received the Moderna vaccine.
The patients, who were invited to participate on social media, were aged 34-54 years, 96% were women, and 89% were White. Inflammatory arthritis was the most common RMD condition (38%), followed by systemic lupus erythematosus (28%) and overlap connective tissue disease (19%). The patients were using a range of immunomodulatory treatment regimens, including nonbiologic disease modifying antirheumatic drugs (DMARDs) in 44%, biologics in 19%, and combination therapy in 37%.
Overall, 89% of patients reported localized symptoms of pain, swelling, and erythema, and 69% reported systemic symptoms. Fatigue was the most common systemic symptom, and 7.4% reported severe fatigue.
None of the patients experienced allergic reactions requiring epinephrine, and 3% reported new infections that required treatment.
“These early, reassuring results may ameliorate concern among patients and provide guidance for rheumatology providers in critical discussions regarding vaccine hesitancy or refusal,” they concluded.
Antibody responses
In another study published in Annals of the Rheumatic Diseases by the same group of researchers, antibody responses against the receptor binding domain of the SARS-CoV-2 spike protein were seen in 74% of 123 adults with an RMD at 18-26 days after receiving a first dose of SARS-CoV-2 mRNA vaccine (52% Pfizer vaccine and 48% Moderna) between Jan. 8, 2021, and Feb. 12, 2021.
The most common diagnoses in these patients were inflammatory arthritis (28%), systemic lupus erythematosus (20%), and Sjögren’s syndrome (13%). A total of 28% of participants reported taking no immunomodulatory agents, 19% reported nonbiologic DMARDs, 14% reported biologic DMARDs, and 19% reported combination therapy.
Although no differences appeared based on disease groups or overall categories of immunomodulatory therapies, patients whose treatment included mycophenolate or rituximab were significantly less likely to develop antibody responses than were patients not taking these medications (P = .001 and P = .04, respectively). Although rituximab and methotrexate have been associated with reduced responses to vaccines such as the flu vaccine, methotrexate was not associated with reduced vaccine response in this study. A total of 94% of patients taking a tumor necrosis factor inhibitor had detectable antibodies.
The studies’ findings were limited by several factors including a lack of longer-term safety data; the small, nonrandomized sample of mainly white women; limited information on immunomodulatory drug dosage and timing; lack of serial antibody measurements; use of an enzyme immunoassay designed to detect antibody response after natural infection; and the inclusion of data only on the first dose of a two-dose vaccine series, the researchers noted. However, the data should provide additional reassurance to RMD patients and their health care teams about vaccination against COVID-19, they said.
Both studies were supported by the Ben-Dov family. In addition, the studies were supported by grants to various study authors from the National Institute of Diabetes and Digestive and Kidney Diseases, the National Institute of Allergy and Infectious Diseases, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, and the Transplantation and Immunology Research Network of the American Society of Transplantation. One author disclosed financial relationships with Sanofi, Novartis, CSL Behring, Jazz Pharmaceuticals, Veloxis, Mallinckrodt, and Thermo Fisher Scientific. The other researchers had no financial conflicts to disclose.
Two reports support the safety and immunogenicity of SARS-CoV-2 mRNA vaccines in patients with rheumatic and musculoskeletal diseases (RMDs) and represent the first available data on such patients.
In an observational cohort study published in Annals of the Rheumatic Diseases, Caoilfhionn M. Connolly, MD, of Johns Hopkins University, Baltimore, and colleagues reviewed data from 325 adults with RMDs who received the first dose of SARS-CoV-2 mRNA vaccine during the period of Dec. 17, 2020, to Feb. 11, 2021. Of these, 51% received the Pfizer/BioNTech vaccine and 49% received the Moderna vaccine.
The patients, who were invited to participate on social media, were aged 34-54 years, 96% were women, and 89% were White. Inflammatory arthritis was the most common RMD condition (38%), followed by systemic lupus erythematosus (28%) and overlap connective tissue disease (19%). The patients were using a range of immunomodulatory treatment regimens, including nonbiologic disease modifying antirheumatic drugs (DMARDs) in 44%, biologics in 19%, and combination therapy in 37%.
Overall, 89% of patients reported localized symptoms of pain, swelling, and erythema, and 69% reported systemic symptoms. Fatigue was the most common systemic symptom, and 7.4% reported severe fatigue.
None of the patients experienced allergic reactions requiring epinephrine, and 3% reported new infections that required treatment.
“These early, reassuring results may ameliorate concern among patients and provide guidance for rheumatology providers in critical discussions regarding vaccine hesitancy or refusal,” they concluded.
Antibody responses
In another study published in Annals of the Rheumatic Diseases by the same group of researchers, antibody responses against the receptor binding domain of the SARS-CoV-2 spike protein were seen in 74% of 123 adults with an RMD at 18-26 days after receiving a first dose of SARS-CoV-2 mRNA vaccine (52% Pfizer vaccine and 48% Moderna) between Jan. 8, 2021, and Feb. 12, 2021.
The most common diagnoses in these patients were inflammatory arthritis (28%), systemic lupus erythematosus (20%), and Sjögren’s syndrome (13%). A total of 28% of participants reported taking no immunomodulatory agents, 19% reported nonbiologic DMARDs, 14% reported biologic DMARDs, and 19% reported combination therapy.
Although no differences appeared based on disease groups or overall categories of immunomodulatory therapies, patients whose treatment included mycophenolate or rituximab were significantly less likely to develop antibody responses than were patients not taking these medications (P = .001 and P = .04, respectively). Although rituximab and methotrexate have been associated with reduced responses to vaccines such as the flu vaccine, methotrexate was not associated with reduced vaccine response in this study. A total of 94% of patients taking a tumor necrosis factor inhibitor had detectable antibodies.
The studies’ findings were limited by several factors including a lack of longer-term safety data; the small, nonrandomized sample of mainly white women; limited information on immunomodulatory drug dosage and timing; lack of serial antibody measurements; use of an enzyme immunoassay designed to detect antibody response after natural infection; and the inclusion of data only on the first dose of a two-dose vaccine series, the researchers noted. However, the data should provide additional reassurance to RMD patients and their health care teams about vaccination against COVID-19, they said.
Both studies were supported by the Ben-Dov family. In addition, the studies were supported by grants to various study authors from the National Institute of Diabetes and Digestive and Kidney Diseases, the National Institute of Allergy and Infectious Diseases, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, and the Transplantation and Immunology Research Network of the American Society of Transplantation. One author disclosed financial relationships with Sanofi, Novartis, CSL Behring, Jazz Pharmaceuticals, Veloxis, Mallinckrodt, and Thermo Fisher Scientific. The other researchers had no financial conflicts to disclose.
FROM ANNALS OF THE RHEUMATIC DISEASES
Denosumab now dominant therapy for osteoporosis linked to cancer
Amid a substantial expansion of therapies in several drug classes for the treatment of osteoporosis, there has been a notable increase in the prescription of denosumab for patients with a cancer-related indication.
In an analysis of claims data from January 2009 to March 2020, the bisphosphonate alendronate represented more than 50% of all prescriptions for bone-directed therapies, but growth in the use of the monoclonal antibody denosumab overall and in cancer-related indications particularly was steady throughout the study period.
“In the malignancy cohort, alendronate and zoledronic acid were each used in approximately 30% of individuals at the onset of the study, but use of both then declined,” Sara Cromer, MD, reported at the annual meeting of the Endocrine Society.
For malignancy-based prescriptions, denosumab surpassed either bisphosphonate by 2013 and then continued to rise.
Denosumab use “reached approximately 50% of all bone-directed medication use in the malignancy cohort” by the end of the study period, said Dr. Cromer, a clinical research fellow in endocrinology at Massachusetts General Hospital, Boston.
The claims data for this analysis was drawn from the Clinformatics Data Mart. The analysis was restricted to individuals aged older than 50 years who received a prescription for a bone-directed therapy. The 15.48 million prescriptions evaluated were drawn from 1.46 million unique individuals. The mean age was 69 years, and 89% of those prescribed a drug were women.
Oncologic indications one of two tracked cohorts
In the context of a large expansion of treatment options in several drug classes for osteoporosis, the objective of this claims analysis was to document trends in treatment choice, according to Dr. Cromer. She and her coinvestigators looked at prescriptions overall as well as in two cohorts defined by ICD codes. One included patients prescribed a prescription by an oncologist. The other included everyone else.
When all prescriptions for bone-directed therapy were evaluated over the study period, alendronate was the most commonly prescribed therapy, and its use increased over time. Prescriptions of zoledronic acid also rose, doubling over the study period, but use was very low in the beginning and it never climbed above 5%.
The proportion of prescriptions written for bisphosphonates other than alendronate and zoledronic acid “declined steadily” over the study period, Dr. Cromer reported.
Denosumab, a monoclonal antibody that targets a step in the process important to maturation of osteoclasts, was approved in 2010. It accounted for 10% of all prescriptions for osteoporosis by 2015 and 15% by 2018. It was still rising through the end of the study period.
In contrast, prescriptions of raloxifene, a selective estrogen receptor modulator, began to decline after 2013. In general, the rates of prescriptions for other agents, including some of the more recently approved drugs, such as teriparatide, abaloparatide, and romosozumab, changed very little over the study period. None of these therapies ever represented more than 2% of prescriptions.
When looking at the cohort of patients who received a bone-directed reason for a noncancer indication, the trends “paralleled those in the all-user analysis,” Dr. Cromer reported.
Denosumab use greater in privately insured
In the malignancy cohort, the decline in the use of bisphosphonates and the rise in the use of denosumab were most pronounced in patients who were privately insured. The increased use of denosumab over the study period “outpaced gains in use of other agents despite guidelines,” said Dr. Cromer, referring to the those issued by the Endocrine Society in 2019 .
In those guidelines, written for management of postmenopausal women at high risk of fractures, bisphosphonates are recommended for initial treatment while denosumab is recommended as an alternative. However, those guidelines do not provide specific recommendations for therapies directed at osteoporosis associated with cancer.
Guidelines for this population exist, including one published by the American Society of Clinical Oncology in 2019.
In the ASCO guidelines, oral bisphosphonates, intravenous bisphosphonates, and subcutaneous denosumab were all identified as “efficacious options,” according to Charles L. Shapiro, MD, director of breast cancer translational research, Mount Sinai Health System, New York.
Specifically, “all three of them work to reduce fractures and improve bone density in women with breast cancer in whom you are trying to prevent or treat osteoporosis,” Dr. Shapiro said in an interview.
There might be relative advantages for one therapy over another in specific subgroups defined by type of cancer or stage of cancer, but trials are not definitive for such outcomes as overall survival. Citing one comparative study associating denosumab with an 18% delay to first skeletal event in women with metastatic breast cancer, Dr. Shapiro observed, “I personally don’t consider an 18% delay [for this outcome] to be that clinically meaningful.”
Although major guidelines from ASCO have not so far favored denosumab over any bisphosphonate in routine care, Dr. Shapiro did not rule out the possibility that future studies will show differences.
Dr. Comer and Dr. Shapiro reported no relevant conflicts of interest.
Amid a substantial expansion of therapies in several drug classes for the treatment of osteoporosis, there has been a notable increase in the prescription of denosumab for patients with a cancer-related indication.
In an analysis of claims data from January 2009 to March 2020, the bisphosphonate alendronate represented more than 50% of all prescriptions for bone-directed therapies, but growth in the use of the monoclonal antibody denosumab overall and in cancer-related indications particularly was steady throughout the study period.
“In the malignancy cohort, alendronate and zoledronic acid were each used in approximately 30% of individuals at the onset of the study, but use of both then declined,” Sara Cromer, MD, reported at the annual meeting of the Endocrine Society.
For malignancy-based prescriptions, denosumab surpassed either bisphosphonate by 2013 and then continued to rise.
Denosumab use “reached approximately 50% of all bone-directed medication use in the malignancy cohort” by the end of the study period, said Dr. Cromer, a clinical research fellow in endocrinology at Massachusetts General Hospital, Boston.
The claims data for this analysis was drawn from the Clinformatics Data Mart. The analysis was restricted to individuals aged older than 50 years who received a prescription for a bone-directed therapy. The 15.48 million prescriptions evaluated were drawn from 1.46 million unique individuals. The mean age was 69 years, and 89% of those prescribed a drug were women.
Oncologic indications one of two tracked cohorts
In the context of a large expansion of treatment options in several drug classes for osteoporosis, the objective of this claims analysis was to document trends in treatment choice, according to Dr. Cromer. She and her coinvestigators looked at prescriptions overall as well as in two cohorts defined by ICD codes. One included patients prescribed a prescription by an oncologist. The other included everyone else.
When all prescriptions for bone-directed therapy were evaluated over the study period, alendronate was the most commonly prescribed therapy, and its use increased over time. Prescriptions of zoledronic acid also rose, doubling over the study period, but use was very low in the beginning and it never climbed above 5%.
The proportion of prescriptions written for bisphosphonates other than alendronate and zoledronic acid “declined steadily” over the study period, Dr. Cromer reported.
Denosumab, a monoclonal antibody that targets a step in the process important to maturation of osteoclasts, was approved in 2010. It accounted for 10% of all prescriptions for osteoporosis by 2015 and 15% by 2018. It was still rising through the end of the study period.
In contrast, prescriptions of raloxifene, a selective estrogen receptor modulator, began to decline after 2013. In general, the rates of prescriptions for other agents, including some of the more recently approved drugs, such as teriparatide, abaloparatide, and romosozumab, changed very little over the study period. None of these therapies ever represented more than 2% of prescriptions.
When looking at the cohort of patients who received a bone-directed reason for a noncancer indication, the trends “paralleled those in the all-user analysis,” Dr. Cromer reported.
Denosumab use greater in privately insured
In the malignancy cohort, the decline in the use of bisphosphonates and the rise in the use of denosumab were most pronounced in patients who were privately insured. The increased use of denosumab over the study period “outpaced gains in use of other agents despite guidelines,” said Dr. Cromer, referring to the those issued by the Endocrine Society in 2019 .
In those guidelines, written for management of postmenopausal women at high risk of fractures, bisphosphonates are recommended for initial treatment while denosumab is recommended as an alternative. However, those guidelines do not provide specific recommendations for therapies directed at osteoporosis associated with cancer.
Guidelines for this population exist, including one published by the American Society of Clinical Oncology in 2019.
In the ASCO guidelines, oral bisphosphonates, intravenous bisphosphonates, and subcutaneous denosumab were all identified as “efficacious options,” according to Charles L. Shapiro, MD, director of breast cancer translational research, Mount Sinai Health System, New York.
Specifically, “all three of them work to reduce fractures and improve bone density in women with breast cancer in whom you are trying to prevent or treat osteoporosis,” Dr. Shapiro said in an interview.
There might be relative advantages for one therapy over another in specific subgroups defined by type of cancer or stage of cancer, but trials are not definitive for such outcomes as overall survival. Citing one comparative study associating denosumab with an 18% delay to first skeletal event in women with metastatic breast cancer, Dr. Shapiro observed, “I personally don’t consider an 18% delay [for this outcome] to be that clinically meaningful.”
Although major guidelines from ASCO have not so far favored denosumab over any bisphosphonate in routine care, Dr. Shapiro did not rule out the possibility that future studies will show differences.
Dr. Comer and Dr. Shapiro reported no relevant conflicts of interest.
Amid a substantial expansion of therapies in several drug classes for the treatment of osteoporosis, there has been a notable increase in the prescription of denosumab for patients with a cancer-related indication.
In an analysis of claims data from January 2009 to March 2020, the bisphosphonate alendronate represented more than 50% of all prescriptions for bone-directed therapies, but growth in the use of the monoclonal antibody denosumab overall and in cancer-related indications particularly was steady throughout the study period.
“In the malignancy cohort, alendronate and zoledronic acid were each used in approximately 30% of individuals at the onset of the study, but use of both then declined,” Sara Cromer, MD, reported at the annual meeting of the Endocrine Society.
For malignancy-based prescriptions, denosumab surpassed either bisphosphonate by 2013 and then continued to rise.
Denosumab use “reached approximately 50% of all bone-directed medication use in the malignancy cohort” by the end of the study period, said Dr. Cromer, a clinical research fellow in endocrinology at Massachusetts General Hospital, Boston.
The claims data for this analysis was drawn from the Clinformatics Data Mart. The analysis was restricted to individuals aged older than 50 years who received a prescription for a bone-directed therapy. The 15.48 million prescriptions evaluated were drawn from 1.46 million unique individuals. The mean age was 69 years, and 89% of those prescribed a drug were women.
Oncologic indications one of two tracked cohorts
In the context of a large expansion of treatment options in several drug classes for osteoporosis, the objective of this claims analysis was to document trends in treatment choice, according to Dr. Cromer. She and her coinvestigators looked at prescriptions overall as well as in two cohorts defined by ICD codes. One included patients prescribed a prescription by an oncologist. The other included everyone else.
When all prescriptions for bone-directed therapy were evaluated over the study period, alendronate was the most commonly prescribed therapy, and its use increased over time. Prescriptions of zoledronic acid also rose, doubling over the study period, but use was very low in the beginning and it never climbed above 5%.
The proportion of prescriptions written for bisphosphonates other than alendronate and zoledronic acid “declined steadily” over the study period, Dr. Cromer reported.
Denosumab, a monoclonal antibody that targets a step in the process important to maturation of osteoclasts, was approved in 2010. It accounted for 10% of all prescriptions for osteoporosis by 2015 and 15% by 2018. It was still rising through the end of the study period.
In contrast, prescriptions of raloxifene, a selective estrogen receptor modulator, began to decline after 2013. In general, the rates of prescriptions for other agents, including some of the more recently approved drugs, such as teriparatide, abaloparatide, and romosozumab, changed very little over the study period. None of these therapies ever represented more than 2% of prescriptions.
When looking at the cohort of patients who received a bone-directed reason for a noncancer indication, the trends “paralleled those in the all-user analysis,” Dr. Cromer reported.
Denosumab use greater in privately insured
In the malignancy cohort, the decline in the use of bisphosphonates and the rise in the use of denosumab were most pronounced in patients who were privately insured. The increased use of denosumab over the study period “outpaced gains in use of other agents despite guidelines,” said Dr. Cromer, referring to the those issued by the Endocrine Society in 2019 .
In those guidelines, written for management of postmenopausal women at high risk of fractures, bisphosphonates are recommended for initial treatment while denosumab is recommended as an alternative. However, those guidelines do not provide specific recommendations for therapies directed at osteoporosis associated with cancer.
Guidelines for this population exist, including one published by the American Society of Clinical Oncology in 2019.
In the ASCO guidelines, oral bisphosphonates, intravenous bisphosphonates, and subcutaneous denosumab were all identified as “efficacious options,” according to Charles L. Shapiro, MD, director of breast cancer translational research, Mount Sinai Health System, New York.
Specifically, “all three of them work to reduce fractures and improve bone density in women with breast cancer in whom you are trying to prevent or treat osteoporosis,” Dr. Shapiro said in an interview.
There might be relative advantages for one therapy over another in specific subgroups defined by type of cancer or stage of cancer, but trials are not definitive for such outcomes as overall survival. Citing one comparative study associating denosumab with an 18% delay to first skeletal event in women with metastatic breast cancer, Dr. Shapiro observed, “I personally don’t consider an 18% delay [for this outcome] to be that clinically meaningful.”
Although major guidelines from ASCO have not so far favored denosumab over any bisphosphonate in routine care, Dr. Shapiro did not rule out the possibility that future studies will show differences.
Dr. Comer and Dr. Shapiro reported no relevant conflicts of interest.
FROM ENDO 2021
Abdominal aortic calcification may further raise known fracture risk
A new study has found that older men with high levels of abdominal aortic calcification (AAC) and a prevalent vertebral fracture – both of which can be assessed via lateral spine radiographs – are at increased risk of hip, clinical vertebral, and major osteoporotic fractures.
“The results of this study and others suggest that it may be appropriate to expand lateral spine imaging to include those with a significant pre-test probability of higher AAC being present,” wrote John T. Schousboe, MD, of the Park Nicollet Clinic and HealthPartners Institute in Bloomington, Minn. The study was published in the Journal of Bone and Mineral Research.
To determine the impact of prevalent vertebral fractures and AAC on fracture risk, the researchers assessed the lateral spine radiographs of 5,365 men who were enrolled in the Osteoporotic Fractures in Men (MrOS) study. All participants were 65 years or older, community dwelling, able to walk without assistance, and without bilateral hip arthroplasties. They split patients’ 24-point AAC (ACC-24) scores at the baseline visit into four levels: 0-1, 2-4, 5-8, and greater than 9. Self-reports of fractures were solicited from the cohort every 4 months.
Of all participants, 7.6% (n = 407) had a prevalent vertebral fracture at baseline. They were, on average, 1.5 years older than participants without a fracture; they were also more likely to be white and to have a prior nonspine fracture, along with having a lower femoral neck BMD (0.718 g/cm2, compared with 0.787 g/cm2; P < .001). In addition, significantly more men with a prevalent vertebral fracture had an AAC score greater than 9 (27% vs. 21.2%).
After an average follow-up period of 12.4 years (standard deviation, 5.2), 634 men had a major osteoporotic fracture, 283 had a hip fracture, 206 had a clinical vertebral fracture, and 2,626 died without having any of the three. After adjustment for risk factors such as age, prior nonspine fracture, and prevalent vertebral fracture, men with higher AAC-24 scores had a higher risk of major osteoporotic fracture, compared with men who had scores of 0-1: a hazard ratio of 1.38 (95% confidence interval, 1.10-1.73; P < .001) for scores 2-4, a HR of 1.45 (95% CI, 1.14-1.84; P < .001) for scores 5-8, and a HR of 1.65 (95% CI, 1.29-2.10; P < .001) for scores greater than 9.
Similar findings were reported regarding risk of hip fractures: a HR of 1.54 (95% CI, 1.07-2.20; P < .001) for men with AAC-24 scores 2-4, a HR of 1.40 (95% CI, 0.96-2.06; P < .001) for scores 5-8, and a HR of 2.17 (95% CI, 1.50-3.13; P < .001) for scores greater than 9. AAC-24 score severity was not associated with a higher risk of clinical vertebral fractures.
After adjustment for risk factors and AAC-24 score, men with prevalent vertebral fractures had an increased risk of all three fracture outcomes, compared with men without any fractures at baseline: a HR of 1.56 (95% CI, 1.12-2.16; P < .001) for hip fracture, a HR of 1.85 (95% CI, 1.48-2.31; P < .001) for major osteoporotic fracture, and a HR of 2.76 (95% CI, 1.94-3.91; P < .001) for clinical vertebral fracture.
Adjusting for competing mortality produced similar results: men with higher levels of AAC had increased risk of major osteoporotic fracture and hip fracture, although AAC-24 score was not associated with higher risk of clinical vertebral fractures. Prevalent vertebral fractures were also still associated with higher risk of hip (subdistribution HR, 1.42; 95% CI, 1.01-2.00; P = .004), major osteoporotic fracture (SHR, 1.71; 95% CI, 1.36-2.14; P < .001), and clinical vertebral fracture (SHR, 2.46; 95% CI, 1.72-3.52; P < .001).
Fracture risk assessment proves to be “a nice proof of concept”
“It’s well known that prevalent fractures predict future fractures,” said Thomas M. Link, MD, PhD, chief of the musculoskeletal imaging section in the department of radiology and biomedical imaging at the University of California, San Francisco, in an interview. “The new finding is that aortic calcifications combined with prevalent fractures perform better in predicting major osteoporotic fractures. Traditionally on radiographs, we note that patients who have more calcifications in vessels have less density or calcium in the bone, so this is a nice proof of concept.”
“While the study shows excellent reproducibility, it is not clear how the AAC-24 score was validated,” he added. “Theoretically, abdominal CT could be used for this.”
Along with validation of the AAC-24 score on lateral spine radiographs, he expressed a desire that future research would be “clearer regarding how this would potentially impact patient management. Prevalent fractures already are an indication to treat patients with osteoporosis-specific drugs. How would the results of this study impact management beyond that?”
The authors acknowledged their study’s other potential limitations, including limits in their ability to estimate absolute and relative hip fracture risk in men with low AAC scores but a prevalent vertebral fracture. In addition, they noted that their cohort was “mostly white, healthy, community-dwelling older men” and therefore may not be generalizable to other populations.
The study was supported by the National Institutes of Health, including grants from the National Institute on Aging, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the National Center for Advancing Translational Sciences, and the NIH Roadmap for Medical Research. One author reported being supported by a National Heart Foundation of Australia Future Leader Fellowship. The others disclosed no potential conflicts of interest.
A new study has found that older men with high levels of abdominal aortic calcification (AAC) and a prevalent vertebral fracture – both of which can be assessed via lateral spine radiographs – are at increased risk of hip, clinical vertebral, and major osteoporotic fractures.
“The results of this study and others suggest that it may be appropriate to expand lateral spine imaging to include those with a significant pre-test probability of higher AAC being present,” wrote John T. Schousboe, MD, of the Park Nicollet Clinic and HealthPartners Institute in Bloomington, Minn. The study was published in the Journal of Bone and Mineral Research.
To determine the impact of prevalent vertebral fractures and AAC on fracture risk, the researchers assessed the lateral spine radiographs of 5,365 men who were enrolled in the Osteoporotic Fractures in Men (MrOS) study. All participants were 65 years or older, community dwelling, able to walk without assistance, and without bilateral hip arthroplasties. They split patients’ 24-point AAC (ACC-24) scores at the baseline visit into four levels: 0-1, 2-4, 5-8, and greater than 9. Self-reports of fractures were solicited from the cohort every 4 months.
Of all participants, 7.6% (n = 407) had a prevalent vertebral fracture at baseline. They were, on average, 1.5 years older than participants without a fracture; they were also more likely to be white and to have a prior nonspine fracture, along with having a lower femoral neck BMD (0.718 g/cm2, compared with 0.787 g/cm2; P < .001). In addition, significantly more men with a prevalent vertebral fracture had an AAC score greater than 9 (27% vs. 21.2%).
After an average follow-up period of 12.4 years (standard deviation, 5.2), 634 men had a major osteoporotic fracture, 283 had a hip fracture, 206 had a clinical vertebral fracture, and 2,626 died without having any of the three. After adjustment for risk factors such as age, prior nonspine fracture, and prevalent vertebral fracture, men with higher AAC-24 scores had a higher risk of major osteoporotic fracture, compared with men who had scores of 0-1: a hazard ratio of 1.38 (95% confidence interval, 1.10-1.73; P < .001) for scores 2-4, a HR of 1.45 (95% CI, 1.14-1.84; P < .001) for scores 5-8, and a HR of 1.65 (95% CI, 1.29-2.10; P < .001) for scores greater than 9.
Similar findings were reported regarding risk of hip fractures: a HR of 1.54 (95% CI, 1.07-2.20; P < .001) for men with AAC-24 scores 2-4, a HR of 1.40 (95% CI, 0.96-2.06; P < .001) for scores 5-8, and a HR of 2.17 (95% CI, 1.50-3.13; P < .001) for scores greater than 9. AAC-24 score severity was not associated with a higher risk of clinical vertebral fractures.
After adjustment for risk factors and AAC-24 score, men with prevalent vertebral fractures had an increased risk of all three fracture outcomes, compared with men without any fractures at baseline: a HR of 1.56 (95% CI, 1.12-2.16; P < .001) for hip fracture, a HR of 1.85 (95% CI, 1.48-2.31; P < .001) for major osteoporotic fracture, and a HR of 2.76 (95% CI, 1.94-3.91; P < .001) for clinical vertebral fracture.
Adjusting for competing mortality produced similar results: men with higher levels of AAC had increased risk of major osteoporotic fracture and hip fracture, although AAC-24 score was not associated with higher risk of clinical vertebral fractures. Prevalent vertebral fractures were also still associated with higher risk of hip (subdistribution HR, 1.42; 95% CI, 1.01-2.00; P = .004), major osteoporotic fracture (SHR, 1.71; 95% CI, 1.36-2.14; P < .001), and clinical vertebral fracture (SHR, 2.46; 95% CI, 1.72-3.52; P < .001).
Fracture risk assessment proves to be “a nice proof of concept”
“It’s well known that prevalent fractures predict future fractures,” said Thomas M. Link, MD, PhD, chief of the musculoskeletal imaging section in the department of radiology and biomedical imaging at the University of California, San Francisco, in an interview. “The new finding is that aortic calcifications combined with prevalent fractures perform better in predicting major osteoporotic fractures. Traditionally on radiographs, we note that patients who have more calcifications in vessels have less density or calcium in the bone, so this is a nice proof of concept.”
“While the study shows excellent reproducibility, it is not clear how the AAC-24 score was validated,” he added. “Theoretically, abdominal CT could be used for this.”
Along with validation of the AAC-24 score on lateral spine radiographs, he expressed a desire that future research would be “clearer regarding how this would potentially impact patient management. Prevalent fractures already are an indication to treat patients with osteoporosis-specific drugs. How would the results of this study impact management beyond that?”
The authors acknowledged their study’s other potential limitations, including limits in their ability to estimate absolute and relative hip fracture risk in men with low AAC scores but a prevalent vertebral fracture. In addition, they noted that their cohort was “mostly white, healthy, community-dwelling older men” and therefore may not be generalizable to other populations.
The study was supported by the National Institutes of Health, including grants from the National Institute on Aging, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the National Center for Advancing Translational Sciences, and the NIH Roadmap for Medical Research. One author reported being supported by a National Heart Foundation of Australia Future Leader Fellowship. The others disclosed no potential conflicts of interest.
A new study has found that older men with high levels of abdominal aortic calcification (AAC) and a prevalent vertebral fracture – both of which can be assessed via lateral spine radiographs – are at increased risk of hip, clinical vertebral, and major osteoporotic fractures.
“The results of this study and others suggest that it may be appropriate to expand lateral spine imaging to include those with a significant pre-test probability of higher AAC being present,” wrote John T. Schousboe, MD, of the Park Nicollet Clinic and HealthPartners Institute in Bloomington, Minn. The study was published in the Journal of Bone and Mineral Research.
To determine the impact of prevalent vertebral fractures and AAC on fracture risk, the researchers assessed the lateral spine radiographs of 5,365 men who were enrolled in the Osteoporotic Fractures in Men (MrOS) study. All participants were 65 years or older, community dwelling, able to walk without assistance, and without bilateral hip arthroplasties. They split patients’ 24-point AAC (ACC-24) scores at the baseline visit into four levels: 0-1, 2-4, 5-8, and greater than 9. Self-reports of fractures were solicited from the cohort every 4 months.
Of all participants, 7.6% (n = 407) had a prevalent vertebral fracture at baseline. They were, on average, 1.5 years older than participants without a fracture; they were also more likely to be white and to have a prior nonspine fracture, along with having a lower femoral neck BMD (0.718 g/cm2, compared with 0.787 g/cm2; P < .001). In addition, significantly more men with a prevalent vertebral fracture had an AAC score greater than 9 (27% vs. 21.2%).
After an average follow-up period of 12.4 years (standard deviation, 5.2), 634 men had a major osteoporotic fracture, 283 had a hip fracture, 206 had a clinical vertebral fracture, and 2,626 died without having any of the three. After adjustment for risk factors such as age, prior nonspine fracture, and prevalent vertebral fracture, men with higher AAC-24 scores had a higher risk of major osteoporotic fracture, compared with men who had scores of 0-1: a hazard ratio of 1.38 (95% confidence interval, 1.10-1.73; P < .001) for scores 2-4, a HR of 1.45 (95% CI, 1.14-1.84; P < .001) for scores 5-8, and a HR of 1.65 (95% CI, 1.29-2.10; P < .001) for scores greater than 9.
Similar findings were reported regarding risk of hip fractures: a HR of 1.54 (95% CI, 1.07-2.20; P < .001) for men with AAC-24 scores 2-4, a HR of 1.40 (95% CI, 0.96-2.06; P < .001) for scores 5-8, and a HR of 2.17 (95% CI, 1.50-3.13; P < .001) for scores greater than 9. AAC-24 score severity was not associated with a higher risk of clinical vertebral fractures.
After adjustment for risk factors and AAC-24 score, men with prevalent vertebral fractures had an increased risk of all three fracture outcomes, compared with men without any fractures at baseline: a HR of 1.56 (95% CI, 1.12-2.16; P < .001) for hip fracture, a HR of 1.85 (95% CI, 1.48-2.31; P < .001) for major osteoporotic fracture, and a HR of 2.76 (95% CI, 1.94-3.91; P < .001) for clinical vertebral fracture.
Adjusting for competing mortality produced similar results: men with higher levels of AAC had increased risk of major osteoporotic fracture and hip fracture, although AAC-24 score was not associated with higher risk of clinical vertebral fractures. Prevalent vertebral fractures were also still associated with higher risk of hip (subdistribution HR, 1.42; 95% CI, 1.01-2.00; P = .004), major osteoporotic fracture (SHR, 1.71; 95% CI, 1.36-2.14; P < .001), and clinical vertebral fracture (SHR, 2.46; 95% CI, 1.72-3.52; P < .001).
Fracture risk assessment proves to be “a nice proof of concept”
“It’s well known that prevalent fractures predict future fractures,” said Thomas M. Link, MD, PhD, chief of the musculoskeletal imaging section in the department of radiology and biomedical imaging at the University of California, San Francisco, in an interview. “The new finding is that aortic calcifications combined with prevalent fractures perform better in predicting major osteoporotic fractures. Traditionally on radiographs, we note that patients who have more calcifications in vessels have less density or calcium in the bone, so this is a nice proof of concept.”
“While the study shows excellent reproducibility, it is not clear how the AAC-24 score was validated,” he added. “Theoretically, abdominal CT could be used for this.”
Along with validation of the AAC-24 score on lateral spine radiographs, he expressed a desire that future research would be “clearer regarding how this would potentially impact patient management. Prevalent fractures already are an indication to treat patients with osteoporosis-specific drugs. How would the results of this study impact management beyond that?”
The authors acknowledged their study’s other potential limitations, including limits in their ability to estimate absolute and relative hip fracture risk in men with low AAC scores but a prevalent vertebral fracture. In addition, they noted that their cohort was “mostly white, healthy, community-dwelling older men” and therefore may not be generalizable to other populations.
The study was supported by the National Institutes of Health, including grants from the National Institute on Aging, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, the National Center for Advancing Translational Sciences, and the NIH Roadmap for Medical Research. One author reported being supported by a National Heart Foundation of Australia Future Leader Fellowship. The others disclosed no potential conflicts of interest.
FROM THE JOURNAL OF BONE AND MINERAL RESEARCH
Don’t discontinue osteoporosis meds for COVID-19 vaccines, expert guidance says
COVID-19 vaccines are safe and effective for patients taking osteoporosis medications, according to joint guidance from six endocrine and osteoporosis societies and foundations.
They noted, though, that some timing modifications with certain medications should be considered to help distinguish between adverse events from the medication versus the vaccine.
The American Society for Bone and Mineral Research “is an international organization, so we brought together our sister societies that have a vested interested in bone health. Vaccination is happening worldwide, and we wanted to present a united front and united recommendations about how to handle osteoporosis medications appropriately during vaccination,” said Suzanne Jan De Beur, MD, who is president of ASBMR and an associate professor of medicine at Johns Hopkins University, Baltimore.
There has been quite a lot of concern from the community about vaccine and medications, from both physicians and patients wondering whether treatments and vaccines should occur in a certain order, and whether there should be a time gap between the two, said Dr. Jan De Beur. “There was a dearth of information about the best practices for osteoporosis treatment management during vaccination, and we didn’t want people missing their opportunity for a vaccine, and we also didn’t want them unnecessarily delaying their osteoporosis treatment.”
There is no evidence that osteoporosis therapies affect the risk or severity of COVID-19 disease, nor do they appear to change the disease course. Osteoporosis itself does not appear associated with increased risk of infection or severe outcomes, so patients with osteoporosis do not need to be prioritized for vaccination based on that condition alone.
There is no evidence that osteoporosis therapies affect the safety or efficacy of vaccination, but given that vaccine availability is currently inconsistent, patients may need to make temporary changes to their osteoporosis regimens to ensure they can receive vaccine when it is available, such as ensuring a delay between medication and vaccination injections.
A key reason for a delay between injectable or infusion medications and a vaccine is to distinguish between adverse events that could occur, so that an adverse reaction to vaccine isn’t mistaken for an adverse reaction to a drug. Nevertheless, the real world is messy. Dr. Jan De Beur noted a recent patient who arrived at her clinic for an injectable treatment who had just received a COVID-19 vaccination that morning. “We decided to put the injection in the other arm, rather than reschedule the person and put them through the risk of coming back. We could distinguish between injection-site reactions, at least,” she said.
No changes should be made to general bone health therapies, such as calcium and vitamin D supplementation, weight-bearing exercises, and maintenance of a balanced diet.
The guidance includes some recommendations for specific osteoporosis medications.
- Oral bisphosphonates: Alendronate, risedronate, and ibandronate should be continued.
- Intravenous bisphosphonates: a 7-day interval (4-day minimum) is recommended between intravenous bisphosphonate (zoledronic acid and ibandronate) infusion and COVID-19 vaccination in order to distinguish potential autoimmune or inflammatory reactions that could be attributable to either intravenous bisphosphonate or the vaccine.
- Denosumab: There should be a 4- to 7-day delay between denosumab infusion and COVID-19 vaccination to account for injection-site reactions. Another option is to have denosumab injected into the contralateral arm or another site like the abdomen or upper thigh, if spacing the injections is not possible. In any case, denosumab injections should be performed within 7 months of the previous dose.
- Teriparatide and abaloparatide should be continued.
- Romosozumab: There should be a 4- to 7-day delay between a romosozumab injection and COVID-19 vaccine, or romosozumab can be injected in the abdomen (with the exception of a 2-inch area around the naval) or thigh if spacing is not possible.
- Raloxifene should be continued in patients receiving COVID-19 vaccination.
Guidance signatories include ASBMR, the American Association of Clinical Endocrinology, the Endocrine Society, the European Calcified Tissue Society, the National Osteoporosis Foundation, and the International Osteoporosis Foundation.
Dr. Jan De Beur has no relevant financial disclosures.
COVID-19 vaccines are safe and effective for patients taking osteoporosis medications, according to joint guidance from six endocrine and osteoporosis societies and foundations.
They noted, though, that some timing modifications with certain medications should be considered to help distinguish between adverse events from the medication versus the vaccine.
The American Society for Bone and Mineral Research “is an international organization, so we brought together our sister societies that have a vested interested in bone health. Vaccination is happening worldwide, and we wanted to present a united front and united recommendations about how to handle osteoporosis medications appropriately during vaccination,” said Suzanne Jan De Beur, MD, who is president of ASBMR and an associate professor of medicine at Johns Hopkins University, Baltimore.
There has been quite a lot of concern from the community about vaccine and medications, from both physicians and patients wondering whether treatments and vaccines should occur in a certain order, and whether there should be a time gap between the two, said Dr. Jan De Beur. “There was a dearth of information about the best practices for osteoporosis treatment management during vaccination, and we didn’t want people missing their opportunity for a vaccine, and we also didn’t want them unnecessarily delaying their osteoporosis treatment.”
There is no evidence that osteoporosis therapies affect the risk or severity of COVID-19 disease, nor do they appear to change the disease course. Osteoporosis itself does not appear associated with increased risk of infection or severe outcomes, so patients with osteoporosis do not need to be prioritized for vaccination based on that condition alone.
There is no evidence that osteoporosis therapies affect the safety or efficacy of vaccination, but given that vaccine availability is currently inconsistent, patients may need to make temporary changes to their osteoporosis regimens to ensure they can receive vaccine when it is available, such as ensuring a delay between medication and vaccination injections.
A key reason for a delay between injectable or infusion medications and a vaccine is to distinguish between adverse events that could occur, so that an adverse reaction to vaccine isn’t mistaken for an adverse reaction to a drug. Nevertheless, the real world is messy. Dr. Jan De Beur noted a recent patient who arrived at her clinic for an injectable treatment who had just received a COVID-19 vaccination that morning. “We decided to put the injection in the other arm, rather than reschedule the person and put them through the risk of coming back. We could distinguish between injection-site reactions, at least,” she said.
No changes should be made to general bone health therapies, such as calcium and vitamin D supplementation, weight-bearing exercises, and maintenance of a balanced diet.
The guidance includes some recommendations for specific osteoporosis medications.
- Oral bisphosphonates: Alendronate, risedronate, and ibandronate should be continued.
- Intravenous bisphosphonates: a 7-day interval (4-day minimum) is recommended between intravenous bisphosphonate (zoledronic acid and ibandronate) infusion and COVID-19 vaccination in order to distinguish potential autoimmune or inflammatory reactions that could be attributable to either intravenous bisphosphonate or the vaccine.
- Denosumab: There should be a 4- to 7-day delay between denosumab infusion and COVID-19 vaccination to account for injection-site reactions. Another option is to have denosumab injected into the contralateral arm or another site like the abdomen or upper thigh, if spacing the injections is not possible. In any case, denosumab injections should be performed within 7 months of the previous dose.
- Teriparatide and abaloparatide should be continued.
- Romosozumab: There should be a 4- to 7-day delay between a romosozumab injection and COVID-19 vaccine, or romosozumab can be injected in the abdomen (with the exception of a 2-inch area around the naval) or thigh if spacing is not possible.
- Raloxifene should be continued in patients receiving COVID-19 vaccination.
Guidance signatories include ASBMR, the American Association of Clinical Endocrinology, the Endocrine Society, the European Calcified Tissue Society, the National Osteoporosis Foundation, and the International Osteoporosis Foundation.
Dr. Jan De Beur has no relevant financial disclosures.
COVID-19 vaccines are safe and effective for patients taking osteoporosis medications, according to joint guidance from six endocrine and osteoporosis societies and foundations.
They noted, though, that some timing modifications with certain medications should be considered to help distinguish between adverse events from the medication versus the vaccine.
The American Society for Bone and Mineral Research “is an international organization, so we brought together our sister societies that have a vested interested in bone health. Vaccination is happening worldwide, and we wanted to present a united front and united recommendations about how to handle osteoporosis medications appropriately during vaccination,” said Suzanne Jan De Beur, MD, who is president of ASBMR and an associate professor of medicine at Johns Hopkins University, Baltimore.
There has been quite a lot of concern from the community about vaccine and medications, from both physicians and patients wondering whether treatments and vaccines should occur in a certain order, and whether there should be a time gap between the two, said Dr. Jan De Beur. “There was a dearth of information about the best practices for osteoporosis treatment management during vaccination, and we didn’t want people missing their opportunity for a vaccine, and we also didn’t want them unnecessarily delaying their osteoporosis treatment.”
There is no evidence that osteoporosis therapies affect the risk or severity of COVID-19 disease, nor do they appear to change the disease course. Osteoporosis itself does not appear associated with increased risk of infection or severe outcomes, so patients with osteoporosis do not need to be prioritized for vaccination based on that condition alone.
There is no evidence that osteoporosis therapies affect the safety or efficacy of vaccination, but given that vaccine availability is currently inconsistent, patients may need to make temporary changes to their osteoporosis regimens to ensure they can receive vaccine when it is available, such as ensuring a delay between medication and vaccination injections.
A key reason for a delay between injectable or infusion medications and a vaccine is to distinguish between adverse events that could occur, so that an adverse reaction to vaccine isn’t mistaken for an adverse reaction to a drug. Nevertheless, the real world is messy. Dr. Jan De Beur noted a recent patient who arrived at her clinic for an injectable treatment who had just received a COVID-19 vaccination that morning. “We decided to put the injection in the other arm, rather than reschedule the person and put them through the risk of coming back. We could distinguish between injection-site reactions, at least,” she said.
No changes should be made to general bone health therapies, such as calcium and vitamin D supplementation, weight-bearing exercises, and maintenance of a balanced diet.
The guidance includes some recommendations for specific osteoporosis medications.
- Oral bisphosphonates: Alendronate, risedronate, and ibandronate should be continued.
- Intravenous bisphosphonates: a 7-day interval (4-day minimum) is recommended between intravenous bisphosphonate (zoledronic acid and ibandronate) infusion and COVID-19 vaccination in order to distinguish potential autoimmune or inflammatory reactions that could be attributable to either intravenous bisphosphonate or the vaccine.
- Denosumab: There should be a 4- to 7-day delay between denosumab infusion and COVID-19 vaccination to account for injection-site reactions. Another option is to have denosumab injected into the contralateral arm or another site like the abdomen or upper thigh, if spacing the injections is not possible. In any case, denosumab injections should be performed within 7 months of the previous dose.
- Teriparatide and abaloparatide should be continued.
- Romosozumab: There should be a 4- to 7-day delay between a romosozumab injection and COVID-19 vaccine, or romosozumab can be injected in the abdomen (with the exception of a 2-inch area around the naval) or thigh if spacing is not possible.
- Raloxifene should be continued in patients receiving COVID-19 vaccination.
Guidance signatories include ASBMR, the American Association of Clinical Endocrinology, the Endocrine Society, the European Calcified Tissue Society, the National Osteoporosis Foundation, and the International Osteoporosis Foundation.
Dr. Jan De Beur has no relevant financial disclosures.