User login
MD-IQ only
HOME-PE trial clarifies which pulmonary embolism patients to treat at home
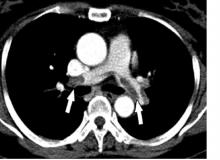
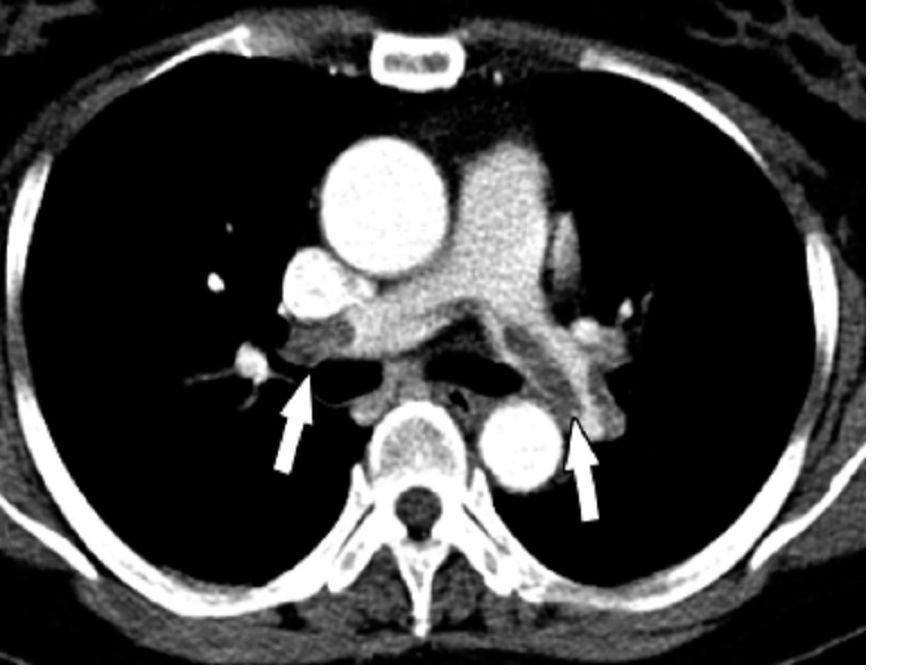
The pragmatic Hestia criteria proved as safe as the more structured, points-based simplified Pulmonary Embolism Severity Index (sPESI) score for selection of patients with acute pulmonary embolism for outpatient care in the large, randomized HOME-PE trial presented at the virtual annual congress of the European Society of Cardiology.
“These results support outpatient management of acute pulmonary embolism patients using either the Hestia method or the sPESI score with the option for the physician-in-charge to override the decision. In hospitals organized for outpatient management, both triaging strategies enable more than a third of pulmonary embolism patients to be managed at home with a low rate of complications,” Pierre-Marie Roy, MD, said in presenting the HOME-PE findings.
The study clarifies a transatlantic controversy regarding how best to triage patients with acute pulmonary embolism (PE) for outpatient care. The answer? It’s basically a tie between the points-based sPESI score recommended in the current ESC guidelines (Eur Respir J. 2019 Oct 9;54[3]:1901647) and the Hestia method endorsed in the American College of Chest Physician guidelines (Chest. 2016 Feb;149[2]:315-52).
The sPESI is a validated tool that grants 1 point each for age over 80 years, background cardiopulmonary disease, a systolic blood pressure below 100 mm Hg, cancer, a heart rate of 110 bpm or more, and an oxygen saturation level below 90%. A patient needs a score of zero to be eligible for outpatient management. In contrast, the Hestia method relies upon 11 simple bedside criteria rather than a points system, explained Dr. Roy of University Hospital of Angers, France (J Thromb Haemost. 2011 Aug;9[8]:1500-7).
HOME-PE was a randomized, open-label, noninferiority trial conducted at 26 hospitals in France, Belgium, Switzerland, and the Netherlands. The study included 1,974 patients presenting to the emergency department with non–high-risk acute PE as defined by hemodynamic stability. About 39% of patients in the Hestia group were eligible for outpatient care on the basis of ‘no’ answers regarding all 11 criteria, while 48% of patients had an sPESI score of 0 and were thus initially considered appropriate for outpatient management.
However, the investigators recognized that no scoring system for acute PE is perfect, and that the judgment of a physician with extensive experience in managing this life-threatening condition counts for a lot. So they stipulated that a patient’s physician-in-charge could overrule a decision for early discharge. This happened 29% of the time in patients with a sPESI score of 0, as compared with a 3% overrule rate with the Hestia rule. The physician-in-charge also moved small numbers of patients who were Hestia or sPESI positive into the outpatient care group. As a result, a similar proportion of patients in both groups were discharged home within 24 hours for outpatient treatment: 38% of the total Hestia group and 37% in the sPESI arm.
Major adverse event rates were reassuringly low in both groups managed on an outpatient basis. The composite of recurrent venous thromboembolism, bleeding, or death within 30 days occurred in 1.3% of Hestia outpatients and 1.1% of sPESI outpatients. Among patients managed in the hospital, these rates were 5.6% in the Hestia group and 4.7% in the sPESI group.
Discussant Stavros V. Konstantinides, MD, who chaired the ESC guideline committee, asked rhetorically, “who’s happy with the HOME-PE trial? I think everybody.”
“The Hestia criteria integrate the feasibility of family support of the individual patient. This is a good thing. And eligibility based on the Hestia criteria, unlike sPESI, does not require age younger than 80 years or no cancer, and it appears from the HOME-PE study that this is okay,” observed Dr. Konstantinides of the Center for Thrombosis and Hemostasis at the University of Mainz (Germany).
In an interview, Hadley Wilson, MD, called the HOME-PE trial “transformative” and predicted it will change clinical practice. He was particularly impressed with the high quality of the trial, noting that 87% of participants managed as outpatients received a direct oral anticoagulant.
The Hestia rule is simpler and more user-friendly. And greater use of this triaging strategy might have advantages in terms of economics and health care utilization by potentially encouraging movement of decision-making regarding outpatient management of acute PE out of the hospital wards and into emergency departments, said Dr. Wilson, executive vice chair of the Sanger Heart and Vascular Institute and a cardiologist at the University of North Carolina at Chapel Hill.
Dr. Roy reported receiving research grants to conduct HOME-PE from the French Ministry of Health, the study sponsor. In addition, he is on scientific advisory boards and/or speakers’ panels for Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Pfizer, Aspen, Daiichi Sankyo, and Sanofi Aventis.
The pragmatic Hestia criteria proved as safe as the more structured, points-based simplified Pulmonary Embolism Severity Index (sPESI) score for selection of patients with acute pulmonary embolism for outpatient care in the large, randomized HOME-PE trial presented at the virtual annual congress of the European Society of Cardiology.
“These results support outpatient management of acute pulmonary embolism patients using either the Hestia method or the sPESI score with the option for the physician-in-charge to override the decision. In hospitals organized for outpatient management, both triaging strategies enable more than a third of pulmonary embolism patients to be managed at home with a low rate of complications,” Pierre-Marie Roy, MD, said in presenting the HOME-PE findings.
The study clarifies a transatlantic controversy regarding how best to triage patients with acute pulmonary embolism (PE) for outpatient care. The answer? It’s basically a tie between the points-based sPESI score recommended in the current ESC guidelines (Eur Respir J. 2019 Oct 9;54[3]:1901647) and the Hestia method endorsed in the American College of Chest Physician guidelines (Chest. 2016 Feb;149[2]:315-52).
The sPESI is a validated tool that grants 1 point each for age over 80 years, background cardiopulmonary disease, a systolic blood pressure below 100 mm Hg, cancer, a heart rate of 110 bpm or more, and an oxygen saturation level below 90%. A patient needs a score of zero to be eligible for outpatient management. In contrast, the Hestia method relies upon 11 simple bedside criteria rather than a points system, explained Dr. Roy of University Hospital of Angers, France (J Thromb Haemost. 2011 Aug;9[8]:1500-7).
HOME-PE was a randomized, open-label, noninferiority trial conducted at 26 hospitals in France, Belgium, Switzerland, and the Netherlands. The study included 1,974 patients presenting to the emergency department with non–high-risk acute PE as defined by hemodynamic stability. About 39% of patients in the Hestia group were eligible for outpatient care on the basis of ‘no’ answers regarding all 11 criteria, while 48% of patients had an sPESI score of 0 and were thus initially considered appropriate for outpatient management.
However, the investigators recognized that no scoring system for acute PE is perfect, and that the judgment of a physician with extensive experience in managing this life-threatening condition counts for a lot. So they stipulated that a patient’s physician-in-charge could overrule a decision for early discharge. This happened 29% of the time in patients with a sPESI score of 0, as compared with a 3% overrule rate with the Hestia rule. The physician-in-charge also moved small numbers of patients who were Hestia or sPESI positive into the outpatient care group. As a result, a similar proportion of patients in both groups were discharged home within 24 hours for outpatient treatment: 38% of the total Hestia group and 37% in the sPESI arm.
Major adverse event rates were reassuringly low in both groups managed on an outpatient basis. The composite of recurrent venous thromboembolism, bleeding, or death within 30 days occurred in 1.3% of Hestia outpatients and 1.1% of sPESI outpatients. Among patients managed in the hospital, these rates were 5.6% in the Hestia group and 4.7% in the sPESI group.
Discussant Stavros V. Konstantinides, MD, who chaired the ESC guideline committee, asked rhetorically, “who’s happy with the HOME-PE trial? I think everybody.”
“The Hestia criteria integrate the feasibility of family support of the individual patient. This is a good thing. And eligibility based on the Hestia criteria, unlike sPESI, does not require age younger than 80 years or no cancer, and it appears from the HOME-PE study that this is okay,” observed Dr. Konstantinides of the Center for Thrombosis and Hemostasis at the University of Mainz (Germany).
In an interview, Hadley Wilson, MD, called the HOME-PE trial “transformative” and predicted it will change clinical practice. He was particularly impressed with the high quality of the trial, noting that 87% of participants managed as outpatients received a direct oral anticoagulant.
The Hestia rule is simpler and more user-friendly. And greater use of this triaging strategy might have advantages in terms of economics and health care utilization by potentially encouraging movement of decision-making regarding outpatient management of acute PE out of the hospital wards and into emergency departments, said Dr. Wilson, executive vice chair of the Sanger Heart and Vascular Institute and a cardiologist at the University of North Carolina at Chapel Hill.
Dr. Roy reported receiving research grants to conduct HOME-PE from the French Ministry of Health, the study sponsor. In addition, he is on scientific advisory boards and/or speakers’ panels for Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Pfizer, Aspen, Daiichi Sankyo, and Sanofi Aventis.
The pragmatic Hestia criteria proved as safe as the more structured, points-based simplified Pulmonary Embolism Severity Index (sPESI) score for selection of patients with acute pulmonary embolism for outpatient care in the large, randomized HOME-PE trial presented at the virtual annual congress of the European Society of Cardiology.
“These results support outpatient management of acute pulmonary embolism patients using either the Hestia method or the sPESI score with the option for the physician-in-charge to override the decision. In hospitals organized for outpatient management, both triaging strategies enable more than a third of pulmonary embolism patients to be managed at home with a low rate of complications,” Pierre-Marie Roy, MD, said in presenting the HOME-PE findings.
The study clarifies a transatlantic controversy regarding how best to triage patients with acute pulmonary embolism (PE) for outpatient care. The answer? It’s basically a tie between the points-based sPESI score recommended in the current ESC guidelines (Eur Respir J. 2019 Oct 9;54[3]:1901647) and the Hestia method endorsed in the American College of Chest Physician guidelines (Chest. 2016 Feb;149[2]:315-52).
The sPESI is a validated tool that grants 1 point each for age over 80 years, background cardiopulmonary disease, a systolic blood pressure below 100 mm Hg, cancer, a heart rate of 110 bpm or more, and an oxygen saturation level below 90%. A patient needs a score of zero to be eligible for outpatient management. In contrast, the Hestia method relies upon 11 simple bedside criteria rather than a points system, explained Dr. Roy of University Hospital of Angers, France (J Thromb Haemost. 2011 Aug;9[8]:1500-7).
HOME-PE was a randomized, open-label, noninferiority trial conducted at 26 hospitals in France, Belgium, Switzerland, and the Netherlands. The study included 1,974 patients presenting to the emergency department with non–high-risk acute PE as defined by hemodynamic stability. About 39% of patients in the Hestia group were eligible for outpatient care on the basis of ‘no’ answers regarding all 11 criteria, while 48% of patients had an sPESI score of 0 and were thus initially considered appropriate for outpatient management.
However, the investigators recognized that no scoring system for acute PE is perfect, and that the judgment of a physician with extensive experience in managing this life-threatening condition counts for a lot. So they stipulated that a patient’s physician-in-charge could overrule a decision for early discharge. This happened 29% of the time in patients with a sPESI score of 0, as compared with a 3% overrule rate with the Hestia rule. The physician-in-charge also moved small numbers of patients who were Hestia or sPESI positive into the outpatient care group. As a result, a similar proportion of patients in both groups were discharged home within 24 hours for outpatient treatment: 38% of the total Hestia group and 37% in the sPESI arm.
Major adverse event rates were reassuringly low in both groups managed on an outpatient basis. The composite of recurrent venous thromboembolism, bleeding, or death within 30 days occurred in 1.3% of Hestia outpatients and 1.1% of sPESI outpatients. Among patients managed in the hospital, these rates were 5.6% in the Hestia group and 4.7% in the sPESI group.
Discussant Stavros V. Konstantinides, MD, who chaired the ESC guideline committee, asked rhetorically, “who’s happy with the HOME-PE trial? I think everybody.”
“The Hestia criteria integrate the feasibility of family support of the individual patient. This is a good thing. And eligibility based on the Hestia criteria, unlike sPESI, does not require age younger than 80 years or no cancer, and it appears from the HOME-PE study that this is okay,” observed Dr. Konstantinides of the Center for Thrombosis and Hemostasis at the University of Mainz (Germany).
In an interview, Hadley Wilson, MD, called the HOME-PE trial “transformative” and predicted it will change clinical practice. He was particularly impressed with the high quality of the trial, noting that 87% of participants managed as outpatients received a direct oral anticoagulant.
The Hestia rule is simpler and more user-friendly. And greater use of this triaging strategy might have advantages in terms of economics and health care utilization by potentially encouraging movement of decision-making regarding outpatient management of acute PE out of the hospital wards and into emergency departments, said Dr. Wilson, executive vice chair of the Sanger Heart and Vascular Institute and a cardiologist at the University of North Carolina at Chapel Hill.
Dr. Roy reported receiving research grants to conduct HOME-PE from the French Ministry of Health, the study sponsor. In addition, he is on scientific advisory boards and/or speakers’ panels for Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Pfizer, Aspen, Daiichi Sankyo, and Sanofi Aventis.
REPORTING FROM ESC CONGRESS 2020
TNF inhibitors cut odds of VTE in RA patients
The risk for venous thromboembolism is almost 50% lower in patients with RA taking TNF inhibitors than in those taking conventional synthetic disease-modifying antirheumatic drugs (DMARDs), according to data from the German RABBIT registry.
“Some rheumatologists have thought TNF inhibitors could increase the risk for venous thromboembolism events, but we don’t think this is true, based on our findings,” said investigator Anja Strangfeld, MD, PhD, from the German Rheumatism Research Center in Berlin.
The risk is more than one-third lower in RA patients treated with other newer biologics, such as abatacept, rituximab, sarilumab, and tocilizumab.
However, risk for a serious venous thromboembolism is twice as high in patients with C-reactive protein (CRP) levels above 5 mg/L and is nearly three times as high in patients 65 years and older.
For the study, Dr. Strangfeld and her colleagues followed about 11,000 patients for more than 10 years. The findings were presented at the European League Against Rheumatism (EULAR) 2020 Congress.
“Patients with RA have a greater risk for venous thromboembolism compared with the general population, but we didn’t know the risk conveyed by different DMARD treatments,” Dr. Strangfeld told Medscape Medical News. “It is also evident that higher age and lower capacity for physical function increase the risk, which was not so surprising.”
Chronic inflammation in RA patients elevates the risk for deep vein and pulmonary thrombosis by two to three times, said John Isaacs, MBBS, PhD, from Newcastle University in Newcastle Upon Tyne, United Kingdom, who is chair of the EULAR scientific program committee.
Among the supporting studies Dr. Isaacs discussed during an online press conference was a Swedish trial of more than 46,000 RA patients, which had been presented earlier by Viktor Molander, a PhD candidate from the Karolinska Institute in Stockholm (abstract OP0034).
Mr. Molander’s team showed that one in 100 patients with high disease activity will develop venous thromboembolism within a year, which is twice the number of events seen among patients in remission.
Combined with the RABBIT data, both studies “show if you can control their disease in the right way, you’re not only helping rheumatoid arthritis patients feel better, but you could be prolonging their lives,” Dr. Isaacs said.
The prospective RABBIT study followed RA patients who began receiving a new DMARD after treatment failed with at least one conventional synthetic DMARD, such as methotrexate or leflunomide. At baseline, those taking TNF inhibitors or other biologics had higher CRP levels on average, as well as a higher rate of existing cardiovascular disease. They also received glucocorticoids, such as prednisone, more often.
The observational nature of the RABBIT study is a weakness, Dr. Strangfeld said, and it could not prove cause and effect. But the methodology had several strengths, including input on patient factors from participating rheumatologists at least every 6 months.
“We enrolled patients at the start of treatment and observed them, regardless of any treatment changes, for up to 10 years,” she added. “That’s a really long observation period.”
The RABBIT data can help shape treatment decisions, said Loreto Carmona, MD, PhD, from the Musculoskeletal Health Institute in Madrid, who is chair of the EULAR abstract selection committee.
For a woman with RA who smokes and takes oral contraceptives, for example, “if she has high levels of inflammation, I think it’s okay to use TNF inhibitors, where maybe in the past we wouldn’t have thought that,” she said.
“The TNF inhibitors are actually reducing the inflammation and, therefore, reducing the risk,” Dr. Carmona told Medscape Medical News. “It could be an effect of using the drugs on people with higher levels of inflammation. It’s an indirect protective effect.”
The study was funded by a joint unconditional grant from AbbVie, Amgen, BMS, Fresenius-Kabi, Hexal, Lilly, MSD, Mylan, Pfizer, Roche, Samsung Bioepis, Sanofi-Aventis, and UCB. Dr. Strangfeld is on the speakers bureau of AbbVie, BMS, Pfizer, Roche and Sanofi-Aventis. Dr. Isaacs is a consultant or has received honoraria or grants from Pfizer, AbbVie, Amgen, Merck, Roche, and UCB. Dr. Carmona has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
The risk for venous thromboembolism is almost 50% lower in patients with RA taking TNF inhibitors than in those taking conventional synthetic disease-modifying antirheumatic drugs (DMARDs), according to data from the German RABBIT registry.
“Some rheumatologists have thought TNF inhibitors could increase the risk for venous thromboembolism events, but we don’t think this is true, based on our findings,” said investigator Anja Strangfeld, MD, PhD, from the German Rheumatism Research Center in Berlin.
The risk is more than one-third lower in RA patients treated with other newer biologics, such as abatacept, rituximab, sarilumab, and tocilizumab.
However, risk for a serious venous thromboembolism is twice as high in patients with C-reactive protein (CRP) levels above 5 mg/L and is nearly three times as high in patients 65 years and older.
For the study, Dr. Strangfeld and her colleagues followed about 11,000 patients for more than 10 years. The findings were presented at the European League Against Rheumatism (EULAR) 2020 Congress.
“Patients with RA have a greater risk for venous thromboembolism compared with the general population, but we didn’t know the risk conveyed by different DMARD treatments,” Dr. Strangfeld told Medscape Medical News. “It is also evident that higher age and lower capacity for physical function increase the risk, which was not so surprising.”
Chronic inflammation in RA patients elevates the risk for deep vein and pulmonary thrombosis by two to three times, said John Isaacs, MBBS, PhD, from Newcastle University in Newcastle Upon Tyne, United Kingdom, who is chair of the EULAR scientific program committee.
Among the supporting studies Dr. Isaacs discussed during an online press conference was a Swedish trial of more than 46,000 RA patients, which had been presented earlier by Viktor Molander, a PhD candidate from the Karolinska Institute in Stockholm (abstract OP0034).
Mr. Molander’s team showed that one in 100 patients with high disease activity will develop venous thromboembolism within a year, which is twice the number of events seen among patients in remission.
Combined with the RABBIT data, both studies “show if you can control their disease in the right way, you’re not only helping rheumatoid arthritis patients feel better, but you could be prolonging their lives,” Dr. Isaacs said.
The prospective RABBIT study followed RA patients who began receiving a new DMARD after treatment failed with at least one conventional synthetic DMARD, such as methotrexate or leflunomide. At baseline, those taking TNF inhibitors or other biologics had higher CRP levels on average, as well as a higher rate of existing cardiovascular disease. They also received glucocorticoids, such as prednisone, more often.
The observational nature of the RABBIT study is a weakness, Dr. Strangfeld said, and it could not prove cause and effect. But the methodology had several strengths, including input on patient factors from participating rheumatologists at least every 6 months.
“We enrolled patients at the start of treatment and observed them, regardless of any treatment changes, for up to 10 years,” she added. “That’s a really long observation period.”
The RABBIT data can help shape treatment decisions, said Loreto Carmona, MD, PhD, from the Musculoskeletal Health Institute in Madrid, who is chair of the EULAR abstract selection committee.
For a woman with RA who smokes and takes oral contraceptives, for example, “if she has high levels of inflammation, I think it’s okay to use TNF inhibitors, where maybe in the past we wouldn’t have thought that,” she said.
“The TNF inhibitors are actually reducing the inflammation and, therefore, reducing the risk,” Dr. Carmona told Medscape Medical News. “It could be an effect of using the drugs on people with higher levels of inflammation. It’s an indirect protective effect.”
The study was funded by a joint unconditional grant from AbbVie, Amgen, BMS, Fresenius-Kabi, Hexal, Lilly, MSD, Mylan, Pfizer, Roche, Samsung Bioepis, Sanofi-Aventis, and UCB. Dr. Strangfeld is on the speakers bureau of AbbVie, BMS, Pfizer, Roche and Sanofi-Aventis. Dr. Isaacs is a consultant or has received honoraria or grants from Pfizer, AbbVie, Amgen, Merck, Roche, and UCB. Dr. Carmona has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
The risk for venous thromboembolism is almost 50% lower in patients with RA taking TNF inhibitors than in those taking conventional synthetic disease-modifying antirheumatic drugs (DMARDs), according to data from the German RABBIT registry.
“Some rheumatologists have thought TNF inhibitors could increase the risk for venous thromboembolism events, but we don’t think this is true, based on our findings,” said investigator Anja Strangfeld, MD, PhD, from the German Rheumatism Research Center in Berlin.
The risk is more than one-third lower in RA patients treated with other newer biologics, such as abatacept, rituximab, sarilumab, and tocilizumab.
However, risk for a serious venous thromboembolism is twice as high in patients with C-reactive protein (CRP) levels above 5 mg/L and is nearly three times as high in patients 65 years and older.
For the study, Dr. Strangfeld and her colleagues followed about 11,000 patients for more than 10 years. The findings were presented at the European League Against Rheumatism (EULAR) 2020 Congress.
“Patients with RA have a greater risk for venous thromboembolism compared with the general population, but we didn’t know the risk conveyed by different DMARD treatments,” Dr. Strangfeld told Medscape Medical News. “It is also evident that higher age and lower capacity for physical function increase the risk, which was not so surprising.”
Chronic inflammation in RA patients elevates the risk for deep vein and pulmonary thrombosis by two to three times, said John Isaacs, MBBS, PhD, from Newcastle University in Newcastle Upon Tyne, United Kingdom, who is chair of the EULAR scientific program committee.
Among the supporting studies Dr. Isaacs discussed during an online press conference was a Swedish trial of more than 46,000 RA patients, which had been presented earlier by Viktor Molander, a PhD candidate from the Karolinska Institute in Stockholm (abstract OP0034).
Mr. Molander’s team showed that one in 100 patients with high disease activity will develop venous thromboembolism within a year, which is twice the number of events seen among patients in remission.
Combined with the RABBIT data, both studies “show if you can control their disease in the right way, you’re not only helping rheumatoid arthritis patients feel better, but you could be prolonging their lives,” Dr. Isaacs said.
The prospective RABBIT study followed RA patients who began receiving a new DMARD after treatment failed with at least one conventional synthetic DMARD, such as methotrexate or leflunomide. At baseline, those taking TNF inhibitors or other biologics had higher CRP levels on average, as well as a higher rate of existing cardiovascular disease. They also received glucocorticoids, such as prednisone, more often.
The observational nature of the RABBIT study is a weakness, Dr. Strangfeld said, and it could not prove cause and effect. But the methodology had several strengths, including input on patient factors from participating rheumatologists at least every 6 months.
“We enrolled patients at the start of treatment and observed them, regardless of any treatment changes, for up to 10 years,” she added. “That’s a really long observation period.”
The RABBIT data can help shape treatment decisions, said Loreto Carmona, MD, PhD, from the Musculoskeletal Health Institute in Madrid, who is chair of the EULAR abstract selection committee.
For a woman with RA who smokes and takes oral contraceptives, for example, “if she has high levels of inflammation, I think it’s okay to use TNF inhibitors, where maybe in the past we wouldn’t have thought that,” she said.
“The TNF inhibitors are actually reducing the inflammation and, therefore, reducing the risk,” Dr. Carmona told Medscape Medical News. “It could be an effect of using the drugs on people with higher levels of inflammation. It’s an indirect protective effect.”
The study was funded by a joint unconditional grant from AbbVie, Amgen, BMS, Fresenius-Kabi, Hexal, Lilly, MSD, Mylan, Pfizer, Roche, Samsung Bioepis, Sanofi-Aventis, and UCB. Dr. Strangfeld is on the speakers bureau of AbbVie, BMS, Pfizer, Roche and Sanofi-Aventis. Dr. Isaacs is a consultant or has received honoraria or grants from Pfizer, AbbVie, Amgen, Merck, Roche, and UCB. Dr. Carmona has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Renal function data improve risk stratification in patients with PAH
The REVEAL-based risk-management strategy was significantly more effective than the current European Society of Cardiology guidelines at discriminating risk in adults with pulmonary arterial hypertension, and renal function significantly improved risk stratification, findings from a retrospective registry study suggest.
“Although the importance of identification of low or high risk is intuitive, the clinical utility of stratification into the intermediate-risk category is less certain” in patients with pulmonary arterial hypertension (PAH), wrote Jason G.E. Zelt, MSc, of the University of Ottawa and colleagues. “Despite the importance of renal function in the PAH population, it has not been formally incorporated into many of the contemporary PAH risk tools, including current guidelines,” they noted.
In a study published in the Journal of Heart and Lung Transplantation, the researchers compared several current research tools for risk assessment in PAH, including the registry to evaluate early and long-term pulmonary arterial hypertension disease management (REVEAL) risk calculator, the French Pulmonary Hypertension Registry (FPHR), and guidelines from the European Society of Cardiology (ESC) and the European Respiratory Society (ERS). They also reviewed REVEAL 2.0, an update that included the estimated glomerular filtration rate (eGFR) as a measure of renal function.
The study population included 211 adults with PAH seen at a single pulmonary hypertension clinic; the average age was 63 years and 65% were women. In addition, 42% had at least stage 3 chronic kidney disease. The primary endpoint was transplant-free survival, which was a median of 7 years. Creatinine was assessed at baseline in all patients. In addition, patients were grouped based on the percent change in renal function between diagnosis and 6 months.
Although the ESC and REVEAL algorithms significantly stratified transplant-free survival risk, the researchers found little agreement among the algorithms in stratifying transplant-free survival for patients in the intermediate-risk category.
However, using REVEAL 2.0, both renal function at diagnosis and renal function at 6 months were significant predictors (P < .0001 for both) from intermediate-risk to higher- or lower-risk groups, the researchers said.
“A decrease in renal function may be a harbinger of both [right ventricle] dysfunction and further PAH disease progression. However, further research is needed to confirm whether declining eGFR is a sentinel biomarker in prospective cohorts,” the researchers said.
The study findings were limited by several factors including the retrospective design and the use of mainly baseline data without information on long-term risk assessment, the researchers noted. However, “a key finding of our study was the ability of baseline eGFR to robustly restratify ESC/ERS-based risk strategies,” they said. “Our work highlights key limitations of the ESC/ERS-based risk assessment, and suggests that incorporating measures of kidney function are important strategies moving forward,” they concluded.
Mr. Zelt is an MD/PhD student and had no financial conflicts to disclose. Some coauthors disclosed relationships with Actelion Pharmaceuticals, Bayer Pharmaceuticals, and Northern Therapeutics.
SOURCE: Zelt JGE et al. J Heart Lung Transplant. 2020 Apr 5. doi: 10.1016/j.healun.2020.03.026.
The REVEAL-based risk-management strategy was significantly more effective than the current European Society of Cardiology guidelines at discriminating risk in adults with pulmonary arterial hypertension, and renal function significantly improved risk stratification, findings from a retrospective registry study suggest.
“Although the importance of identification of low or high risk is intuitive, the clinical utility of stratification into the intermediate-risk category is less certain” in patients with pulmonary arterial hypertension (PAH), wrote Jason G.E. Zelt, MSc, of the University of Ottawa and colleagues. “Despite the importance of renal function in the PAH population, it has not been formally incorporated into many of the contemporary PAH risk tools, including current guidelines,” they noted.
In a study published in the Journal of Heart and Lung Transplantation, the researchers compared several current research tools for risk assessment in PAH, including the registry to evaluate early and long-term pulmonary arterial hypertension disease management (REVEAL) risk calculator, the French Pulmonary Hypertension Registry (FPHR), and guidelines from the European Society of Cardiology (ESC) and the European Respiratory Society (ERS). They also reviewed REVEAL 2.0, an update that included the estimated glomerular filtration rate (eGFR) as a measure of renal function.
The study population included 211 adults with PAH seen at a single pulmonary hypertension clinic; the average age was 63 years and 65% were women. In addition, 42% had at least stage 3 chronic kidney disease. The primary endpoint was transplant-free survival, which was a median of 7 years. Creatinine was assessed at baseline in all patients. In addition, patients were grouped based on the percent change in renal function between diagnosis and 6 months.
Although the ESC and REVEAL algorithms significantly stratified transplant-free survival risk, the researchers found little agreement among the algorithms in stratifying transplant-free survival for patients in the intermediate-risk category.
However, using REVEAL 2.0, both renal function at diagnosis and renal function at 6 months were significant predictors (P < .0001 for both) from intermediate-risk to higher- or lower-risk groups, the researchers said.
“A decrease in renal function may be a harbinger of both [right ventricle] dysfunction and further PAH disease progression. However, further research is needed to confirm whether declining eGFR is a sentinel biomarker in prospective cohorts,” the researchers said.
The study findings were limited by several factors including the retrospective design and the use of mainly baseline data without information on long-term risk assessment, the researchers noted. However, “a key finding of our study was the ability of baseline eGFR to robustly restratify ESC/ERS-based risk strategies,” they said. “Our work highlights key limitations of the ESC/ERS-based risk assessment, and suggests that incorporating measures of kidney function are important strategies moving forward,” they concluded.
Mr. Zelt is an MD/PhD student and had no financial conflicts to disclose. Some coauthors disclosed relationships with Actelion Pharmaceuticals, Bayer Pharmaceuticals, and Northern Therapeutics.
SOURCE: Zelt JGE et al. J Heart Lung Transplant. 2020 Apr 5. doi: 10.1016/j.healun.2020.03.026.
The REVEAL-based risk-management strategy was significantly more effective than the current European Society of Cardiology guidelines at discriminating risk in adults with pulmonary arterial hypertension, and renal function significantly improved risk stratification, findings from a retrospective registry study suggest.
“Although the importance of identification of low or high risk is intuitive, the clinical utility of stratification into the intermediate-risk category is less certain” in patients with pulmonary arterial hypertension (PAH), wrote Jason G.E. Zelt, MSc, of the University of Ottawa and colleagues. “Despite the importance of renal function in the PAH population, it has not been formally incorporated into many of the contemporary PAH risk tools, including current guidelines,” they noted.
In a study published in the Journal of Heart and Lung Transplantation, the researchers compared several current research tools for risk assessment in PAH, including the registry to evaluate early and long-term pulmonary arterial hypertension disease management (REVEAL) risk calculator, the French Pulmonary Hypertension Registry (FPHR), and guidelines from the European Society of Cardiology (ESC) and the European Respiratory Society (ERS). They also reviewed REVEAL 2.0, an update that included the estimated glomerular filtration rate (eGFR) as a measure of renal function.
The study population included 211 adults with PAH seen at a single pulmonary hypertension clinic; the average age was 63 years and 65% were women. In addition, 42% had at least stage 3 chronic kidney disease. The primary endpoint was transplant-free survival, which was a median of 7 years. Creatinine was assessed at baseline in all patients. In addition, patients were grouped based on the percent change in renal function between diagnosis and 6 months.
Although the ESC and REVEAL algorithms significantly stratified transplant-free survival risk, the researchers found little agreement among the algorithms in stratifying transplant-free survival for patients in the intermediate-risk category.
However, using REVEAL 2.0, both renal function at diagnosis and renal function at 6 months were significant predictors (P < .0001 for both) from intermediate-risk to higher- or lower-risk groups, the researchers said.
“A decrease in renal function may be a harbinger of both [right ventricle] dysfunction and further PAH disease progression. However, further research is needed to confirm whether declining eGFR is a sentinel biomarker in prospective cohorts,” the researchers said.
The study findings were limited by several factors including the retrospective design and the use of mainly baseline data without information on long-term risk assessment, the researchers noted. However, “a key finding of our study was the ability of baseline eGFR to robustly restratify ESC/ERS-based risk strategies,” they said. “Our work highlights key limitations of the ESC/ERS-based risk assessment, and suggests that incorporating measures of kidney function are important strategies moving forward,” they concluded.
Mr. Zelt is an MD/PhD student and had no financial conflicts to disclose. Some coauthors disclosed relationships with Actelion Pharmaceuticals, Bayer Pharmaceuticals, and Northern Therapeutics.
SOURCE: Zelt JGE et al. J Heart Lung Transplant. 2020 Apr 5. doi: 10.1016/j.healun.2020.03.026.
FROM THE JOURNAL OF HEART AND LUNG TRANSPLANTATION
PCSK9 inhibitors unexpectedly link with lower VTE, aortic stenosis
Post hoc analyses of recent large, clinical outcomes studies of PCSK9 inhibitors have revealed two tantalizing and unexpected potential benefits from these drugs: an ability to substantially reduce the incidence or severity of venous thromboembolism and aortic stenosis.
The evidence also suggests that these effects are linked to the ability of these drugs to reduce blood levels of Lp(a) lipoprotein by roughly a quarter, currently the biggest known effect on Lp(a) levels of any approved medication.
One study ran post hoc analyses of venous thromboembolism (VTE) events in the FOURIER pivotal trial of evolocumab (Repatha), with more than 27,500 randomized patients (N Engl J Med. 2017 May 4; 376[18]:1713-22), and in the ODYSSEY OUTCOMES pivotal trial of alirocumab (Praluent), with nearly 19,000 randomized patients (N Engl J Med. 2018 Nov 29;379[22]:2097-2107). The analyses showed that, with evolocumab treatment, the incidence of VTE events fell by a statistically significant 29%, compared with patients on placebo, while in ODYSSEY OUTCOMES patients treated with alirocumab had a 33% cut in VTE events, compared with placebo-treated patients, a difference that just missed statistical significance (Circulation. 2020 Mar 29. doi: 10.1161/CIRCULATIONAHA.120.046524) in analyses that were not prespecified before these trials started, Nicholas A. Marston, MD, said in a presentation of his research during the joint scientific sessions of the American College of Cardiology and the World Heart Federation, which was presented online this year. ACC organizers chose to present parts of the meeting virtually after COVID-19 concerns caused them to cancel the meeting.
A combined analysis of 46,488 patients from both studies showed a 31% cut in VTE events with PCSK9 inhibitor treatment, a highly significant finding using VTE endpoints that were not specifically tallied nor adjudicated but collected as part of the serious adverse event reporting in the two pivotal trials, said Dr. Marston, a cardiologist at Brigham and Women’s Hospital in Boston. This is the first report of a statistically significant link between treatment with PCSK9-inhibiting agents and a reduction in VTE, he added. Researchers from the ODYSSEY OUTCOMES trial had reported a VTE analysis in 2019, and while data from that trial on its own showed a nominal 33% lower VTE rate with alirocumab treatment, it just missed statistical significance.
The VTE effect took about a year on treatment to start to manifest. During the first 12 months of FOURIER, the rate of VTE events among patients in the two treatment arms was virtually identical. But starting during months 13-18 on treatment, the event curves in the two arms began to increasingly diverge, and overall during the period from month 13 to the end of the study treatment with evolocumab was linked with a statistically significant 46% reduction in VTE events, compared with patients who received placebo. The results Dr. Marston reported were also published online (Circulation. 2020 Mar 29. doi: 10.1161/CIRCULATIONAHA.120.046397).
The suggestion that this association may be linked to the impact of PCSK9 inhibitors on Lp(a) came from an additional analysis that Dr. Marston presented, which looked at the link between evolocumab use and a change in VTE event rates, compared with placebo, depending on baseline lipoprotein levels. Evolocumab treatment was associated with a roughly similar, modest, and not statistically significant reduction in VTE events, compared with placebo regardless of whether patients had baseline levels of LDL cholesterol below the median or at or above the median. In contrast, when a similar analysis divided patients based on whether their Lp(a) level at baseline was below, or at or above, the median the results showed no discernible effect of evolocumab treatment, compared with on VTE events in patients with lower baseline Lp(a), but in those with higher levels treatment with evolocumab linked with a 48% cut in VTE events, compared with placebo, a statistically significant difference.
In FOURIER, treatment with evolocumab lowered baseline Lp(a) levels by a median of 27%, compared with placebo, among the 25,096 enrolled patients who had their baseline levels measured. As previously reported, prespecified analysis of FOURIER data also showed that the impact of evolocumab, compared with placebo, on the combined rate of coronary heart disease death, MI, or need for urgent coronary revascularization was enhanced among patients with elevated baseline Lp(a) and moderated in those who entered with lower levels. Among patients who entered FOURIER with Lp(a) levels at or below the median treatment with evolocumab cut the primary endpoint by 7%, compared with placebo, a difference that was not statistically significant. Among patients who began the study with Lp(a) levels above the median, evolocumab treatment cut the primary endpoint by 23%, compared with placebo, a statistically significant effect (Circulation. 2019 Mar 19;139[12]:1483-92).
The aortic stenosis connection
A second study reported in the online scientific sessions (Abstract 914-08) used only FOURIER data, and showed that patients treated with evolocumab had a roughly similar response pattern in their incidence of aortic stenosis (AS) events as they did for VTE events.
During the first year of the study, the incidence of AS events was virtually identical among patients treated with evolocumab and those who received placebo. But after the first 12 months and through the study’s end, patients on evolocumab showed a statistically significant 52% relative reduction in AS events, compared with control patients, said Brian A. Bergmark, MD. For the entire study duration, treatment with evolocumab linked with a 34% relative reduction in AS events, compared with placebo, a difference that did not reach statistical significance, added Dr. Bergmark, an interventional cardiologist also at Brigham and Women’s Hospital. The observed halving in total AS events that linked with evolocumab treatment after the first year of the study included a similar-magnitude reduction specifically in the incidence of aortic valve replacement procedures in the evolocumab-treated patients.
Further analysis of both total AS events and aortic valve replacements in FOURIER patients showed that they occurred at a significantly elevated rate in patients who entered the study with higher baseline Lp(a) levels in a multivariate analysis, but a similar analysis showed no significant association between the incidence of these AS-related events and baseline levels of LDL cholesterol, he said.
The AS analysis carried the same important limitations as the VTE analysis: It ran on a post hoc basis and focused on events that were relatively uncommon and not adjudicated, Dr. Bergmark cautioned. Nonetheless, other investigators saw important potential implications from both the VTE and AS observations, with the huge caveat that they need replication in prospective studies designed to specifically address the validity of these findings.
What it could mean
These observed associations between PCSK9 inhibitor treatment and apparent reductions in the rate of both VTE and AS events “represent a tremendous clinical breakthrough,” commented Michelle L. O’Donoghue, MD, a cardiologist at Brigham and Women’s Hospital who is a FOURIER coinvestigator and has led some of the Lp(a) analyses run from that study.
“To date, we have not identified any therapies that slow progression of AS. Other classes of lipid-lowering therapies, such as statins, have been tested and not demonstrated a significant effect,” Dr. O’Donoghue said in an interview.
“For AS, the results are very intriguing. If confirmed, it could be groundbreaking. AS is the most common valve disease in the developed world, and no medical therapy exists. The potential is immense,” commented George Thanassoulis, MD, director of preventive and genomic cardiology at McGill University, Montreal. “Having a medical treatment that could slow AS progression would completely change the disease. It’s conceivable to slow the disease enough that patients may never require valve replacement.” But an interview he cautioned that, “although the results are exciting, the analysis has many limitations. What we need is a dedicated, randomized trial for AS. I hope this stimulates that.”
“For VTE, it’s an interesting finding, but I don’t think it will have clinical utility because we have good treatment for VTE,” added Dr. Thanassoulis, but others saw more opportunity from what could be a new way to reduce VTE risk.
“Given that many patients have difficulty with the bleeding risk from anticoagulants, this option [a PCSK9 inhibitor] may be quite welcome for preventing VTE,” commented Gregory Piazza, MD, a cardiologist and VTE specialist at Brigham and Women’s Hospital who was not involved in any of the PCSK9 inhibitor studies.
“At this time we would not suggest that PCSK9 inhibitors replace an anticoagulant for patients with an established clot or at high risk for a recurrent clot, but if patients have an indication for a PCSK9 inhibitor, the further reduction in venous clot can be viewed as an additional benefit of this therapy,” said Dr. O’Donoghue.
How it might work
A possible mechanism underlying a VTE effect is unclear. Results from the JUPITER trial more than a decade ago had shown a significant association between treatment with 20 mg/day of rosuvastatin and a cut in VTE episodes, compared with placebo, in a prespecified, secondary analysis of the trial with nearly 18,000 patients selected for having a relatively high level of high-sensitivity C-reactive protein (N Engl J Med. 2009 Apr 30;360[18]:1851-61). But a meta-analysis of 29 controlled statin trials that used a variety of statin types and dosages (and included the JUPITER results) failed to confirm a statistically significant change in VTE rates from statins, though they produced a small, nominal reduction (PLoS Med. 2012 Sep 18. doi: 10.1371/journal.pmed.1001310).
Lp(a) “has long been linked to thrombosis, in particular arterial thrombosis,” so the link observed in the PCSK9 inhibitor trials “is not surprising,” said Dr. Piazza. Dr. O’Donoghue agreed that prior evidence had “suggested a prothrombotic role for Lp(a).”
Dr. Thanassoulis was more skeptical of a Lp(a) connection to VTE. “There has always been controversy regarding the prothrombotic effects of Lp(a) and whether it’s clinically relevant,” he said. “The genetic data, from Mendelian randomization studies, is not consistent” with a Lp(a) and VTE link.
The association of AS and Lp(a) may be stronger. “Our team showed that people with genetic variants that predispose to high Lp(a) have a much higher incidence of AS,” Dr. Thanassoulis noted. “We and others have also demonstrated that both Lp(a) and LDL are likely causal mediators of aortic valve calcification and stenosis.”
Dr. O’Donoghue also cited observational genetic data that linked elevated Lp(a) with AS. “Mendelian randomization studies have demonstrated that Lp(a) is a causal contributer to AS, and evolocumab reduced Lp(a) by 25%-30%, raising the possibility that Lp(a) lowering with these drugs may be the mechanism,” she said.
The future of Lp(a) lowering
This last point from Dr. O’Donoghue, that PCSK9 inhibitors cut Lp(a) levels by about 25%-30%, means that they are the most potent Lp(a)-lowering agents currently available, but it also leaves lots of room for other agents to do even better in cutting Lp(a).
“There are now drugs in development that block production of the Lp(a) protein and dramatically reduce its concentration, by about 80%,” Dr. O’Donoghue noted. “It will be of interest to study whether these novel therapies, now in phase 2 and phase 3 studies, have any effect on the risk for VTE and AS.”
“Several drugs in development, including antisense RNA and RNA-interfering molecules, are much more potent and lower Lp(a) by 80%-90%. Because of this potency they can completely normalize Lp(a) in most patients. For Lp(a) lowering, the future is in these new molecules. Randomized trials have started, and we will hopefully have some results in about 5 years,” said Dr. Thanassoulis.
Until then, the prospect of possibly soon documenting benefits from PCSK9 inhibitors beyond their impact on cutting LDL cholesterol raises some hope to get more bang for the considerable buck these drugs cost. But Dr. Thanassoulis was skeptical it would move the cost-benefit ratio much. “VTE and AS are relatively rare, compared with atherosclerotic cardiovascular events, and therefore the added value at the population level would be small,” he predicted. But if treatment with a drug could help patients avoid surgical or percutaneous valve interventions “that could be really interesting from a cost-benefit perspective.”
FOURIER was funded by Amgen, the company that markets evolocumab (Repatha). ODYSSEY OUTCOMES was funded by Sanofi and Regeneron, the companies that developed and market alirocumab (Praluent). Dr. Marston had no disclosures. Dr. Bergmark has been a consultant to Daiichi Sankyo, Janssen, Quark, and Servier and has received research funding from Abbott Vascular, AstraZeneca, and MedImmune. Dr. O’Donoghue has been a consultant to and has received research funding from Amgen; has been a consultant to Janssen and Novartis; and has received research funding from AstraZeneca, Eisai, GlaxoSmithKline, Janssen, Medimmune, Merck, and The Medicines Company. Dr. Thanassoulis has been an adviser to and speaker for Amgen; an adviser to Ionis and Sanofi/Regeneron; a speaker on behalf of Boehringer Ingelheim, Sanofi, and Servier; and has received research funding from Ionis and Servier. Dr. Piazza has been a consultant to Optum, Pfizer, and Thrombolex and he has received research funding from Bayer, Bristol-Myers Squibb, Daiichi Sankyo, Ekos, Janssen, and Portola.
Post hoc analyses of recent large, clinical outcomes studies of PCSK9 inhibitors have revealed two tantalizing and unexpected potential benefits from these drugs: an ability to substantially reduce the incidence or severity of venous thromboembolism and aortic stenosis.
The evidence also suggests that these effects are linked to the ability of these drugs to reduce blood levels of Lp(a) lipoprotein by roughly a quarter, currently the biggest known effect on Lp(a) levels of any approved medication.
One study ran post hoc analyses of venous thromboembolism (VTE) events in the FOURIER pivotal trial of evolocumab (Repatha), with more than 27,500 randomized patients (N Engl J Med. 2017 May 4; 376[18]:1713-22), and in the ODYSSEY OUTCOMES pivotal trial of alirocumab (Praluent), with nearly 19,000 randomized patients (N Engl J Med. 2018 Nov 29;379[22]:2097-2107). The analyses showed that, with evolocumab treatment, the incidence of VTE events fell by a statistically significant 29%, compared with patients on placebo, while in ODYSSEY OUTCOMES patients treated with alirocumab had a 33% cut in VTE events, compared with placebo-treated patients, a difference that just missed statistical significance (Circulation. 2020 Mar 29. doi: 10.1161/CIRCULATIONAHA.120.046524) in analyses that were not prespecified before these trials started, Nicholas A. Marston, MD, said in a presentation of his research during the joint scientific sessions of the American College of Cardiology and the World Heart Federation, which was presented online this year. ACC organizers chose to present parts of the meeting virtually after COVID-19 concerns caused them to cancel the meeting.
A combined analysis of 46,488 patients from both studies showed a 31% cut in VTE events with PCSK9 inhibitor treatment, a highly significant finding using VTE endpoints that were not specifically tallied nor adjudicated but collected as part of the serious adverse event reporting in the two pivotal trials, said Dr. Marston, a cardiologist at Brigham and Women’s Hospital in Boston. This is the first report of a statistically significant link between treatment with PCSK9-inhibiting agents and a reduction in VTE, he added. Researchers from the ODYSSEY OUTCOMES trial had reported a VTE analysis in 2019, and while data from that trial on its own showed a nominal 33% lower VTE rate with alirocumab treatment, it just missed statistical significance.
The VTE effect took about a year on treatment to start to manifest. During the first 12 months of FOURIER, the rate of VTE events among patients in the two treatment arms was virtually identical. But starting during months 13-18 on treatment, the event curves in the two arms began to increasingly diverge, and overall during the period from month 13 to the end of the study treatment with evolocumab was linked with a statistically significant 46% reduction in VTE events, compared with patients who received placebo. The results Dr. Marston reported were also published online (Circulation. 2020 Mar 29. doi: 10.1161/CIRCULATIONAHA.120.046397).
The suggestion that this association may be linked to the impact of PCSK9 inhibitors on Lp(a) came from an additional analysis that Dr. Marston presented, which looked at the link between evolocumab use and a change in VTE event rates, compared with placebo, depending on baseline lipoprotein levels. Evolocumab treatment was associated with a roughly similar, modest, and not statistically significant reduction in VTE events, compared with placebo regardless of whether patients had baseline levels of LDL cholesterol below the median or at or above the median. In contrast, when a similar analysis divided patients based on whether their Lp(a) level at baseline was below, or at or above, the median the results showed no discernible effect of evolocumab treatment, compared with on VTE events in patients with lower baseline Lp(a), but in those with higher levels treatment with evolocumab linked with a 48% cut in VTE events, compared with placebo, a statistically significant difference.
In FOURIER, treatment with evolocumab lowered baseline Lp(a) levels by a median of 27%, compared with placebo, among the 25,096 enrolled patients who had their baseline levels measured. As previously reported, prespecified analysis of FOURIER data also showed that the impact of evolocumab, compared with placebo, on the combined rate of coronary heart disease death, MI, or need for urgent coronary revascularization was enhanced among patients with elevated baseline Lp(a) and moderated in those who entered with lower levels. Among patients who entered FOURIER with Lp(a) levels at or below the median treatment with evolocumab cut the primary endpoint by 7%, compared with placebo, a difference that was not statistically significant. Among patients who began the study with Lp(a) levels above the median, evolocumab treatment cut the primary endpoint by 23%, compared with placebo, a statistically significant effect (Circulation. 2019 Mar 19;139[12]:1483-92).
The aortic stenosis connection
A second study reported in the online scientific sessions (Abstract 914-08) used only FOURIER data, and showed that patients treated with evolocumab had a roughly similar response pattern in their incidence of aortic stenosis (AS) events as they did for VTE events.
During the first year of the study, the incidence of AS events was virtually identical among patients treated with evolocumab and those who received placebo. But after the first 12 months and through the study’s end, patients on evolocumab showed a statistically significant 52% relative reduction in AS events, compared with control patients, said Brian A. Bergmark, MD. For the entire study duration, treatment with evolocumab linked with a 34% relative reduction in AS events, compared with placebo, a difference that did not reach statistical significance, added Dr. Bergmark, an interventional cardiologist also at Brigham and Women’s Hospital. The observed halving in total AS events that linked with evolocumab treatment after the first year of the study included a similar-magnitude reduction specifically in the incidence of aortic valve replacement procedures in the evolocumab-treated patients.
Further analysis of both total AS events and aortic valve replacements in FOURIER patients showed that they occurred at a significantly elevated rate in patients who entered the study with higher baseline Lp(a) levels in a multivariate analysis, but a similar analysis showed no significant association between the incidence of these AS-related events and baseline levels of LDL cholesterol, he said.
The AS analysis carried the same important limitations as the VTE analysis: It ran on a post hoc basis and focused on events that were relatively uncommon and not adjudicated, Dr. Bergmark cautioned. Nonetheless, other investigators saw important potential implications from both the VTE and AS observations, with the huge caveat that they need replication in prospective studies designed to specifically address the validity of these findings.
What it could mean
These observed associations between PCSK9 inhibitor treatment and apparent reductions in the rate of both VTE and AS events “represent a tremendous clinical breakthrough,” commented Michelle L. O’Donoghue, MD, a cardiologist at Brigham and Women’s Hospital who is a FOURIER coinvestigator and has led some of the Lp(a) analyses run from that study.
“To date, we have not identified any therapies that slow progression of AS. Other classes of lipid-lowering therapies, such as statins, have been tested and not demonstrated a significant effect,” Dr. O’Donoghue said in an interview.
“For AS, the results are very intriguing. If confirmed, it could be groundbreaking. AS is the most common valve disease in the developed world, and no medical therapy exists. The potential is immense,” commented George Thanassoulis, MD, director of preventive and genomic cardiology at McGill University, Montreal. “Having a medical treatment that could slow AS progression would completely change the disease. It’s conceivable to slow the disease enough that patients may never require valve replacement.” But an interview he cautioned that, “although the results are exciting, the analysis has many limitations. What we need is a dedicated, randomized trial for AS. I hope this stimulates that.”
“For VTE, it’s an interesting finding, but I don’t think it will have clinical utility because we have good treatment for VTE,” added Dr. Thanassoulis, but others saw more opportunity from what could be a new way to reduce VTE risk.
“Given that many patients have difficulty with the bleeding risk from anticoagulants, this option [a PCSK9 inhibitor] may be quite welcome for preventing VTE,” commented Gregory Piazza, MD, a cardiologist and VTE specialist at Brigham and Women’s Hospital who was not involved in any of the PCSK9 inhibitor studies.
“At this time we would not suggest that PCSK9 inhibitors replace an anticoagulant for patients with an established clot or at high risk for a recurrent clot, but if patients have an indication for a PCSK9 inhibitor, the further reduction in venous clot can be viewed as an additional benefit of this therapy,” said Dr. O’Donoghue.
How it might work
A possible mechanism underlying a VTE effect is unclear. Results from the JUPITER trial more than a decade ago had shown a significant association between treatment with 20 mg/day of rosuvastatin and a cut in VTE episodes, compared with placebo, in a prespecified, secondary analysis of the trial with nearly 18,000 patients selected for having a relatively high level of high-sensitivity C-reactive protein (N Engl J Med. 2009 Apr 30;360[18]:1851-61). But a meta-analysis of 29 controlled statin trials that used a variety of statin types and dosages (and included the JUPITER results) failed to confirm a statistically significant change in VTE rates from statins, though they produced a small, nominal reduction (PLoS Med. 2012 Sep 18. doi: 10.1371/journal.pmed.1001310).
Lp(a) “has long been linked to thrombosis, in particular arterial thrombosis,” so the link observed in the PCSK9 inhibitor trials “is not surprising,” said Dr. Piazza. Dr. O’Donoghue agreed that prior evidence had “suggested a prothrombotic role for Lp(a).”
Dr. Thanassoulis was more skeptical of a Lp(a) connection to VTE. “There has always been controversy regarding the prothrombotic effects of Lp(a) and whether it’s clinically relevant,” he said. “The genetic data, from Mendelian randomization studies, is not consistent” with a Lp(a) and VTE link.
The association of AS and Lp(a) may be stronger. “Our team showed that people with genetic variants that predispose to high Lp(a) have a much higher incidence of AS,” Dr. Thanassoulis noted. “We and others have also demonstrated that both Lp(a) and LDL are likely causal mediators of aortic valve calcification and stenosis.”
Dr. O’Donoghue also cited observational genetic data that linked elevated Lp(a) with AS. “Mendelian randomization studies have demonstrated that Lp(a) is a causal contributer to AS, and evolocumab reduced Lp(a) by 25%-30%, raising the possibility that Lp(a) lowering with these drugs may be the mechanism,” she said.
The future of Lp(a) lowering
This last point from Dr. O’Donoghue, that PCSK9 inhibitors cut Lp(a) levels by about 25%-30%, means that they are the most potent Lp(a)-lowering agents currently available, but it also leaves lots of room for other agents to do even better in cutting Lp(a).
“There are now drugs in development that block production of the Lp(a) protein and dramatically reduce its concentration, by about 80%,” Dr. O’Donoghue noted. “It will be of interest to study whether these novel therapies, now in phase 2 and phase 3 studies, have any effect on the risk for VTE and AS.”
“Several drugs in development, including antisense RNA and RNA-interfering molecules, are much more potent and lower Lp(a) by 80%-90%. Because of this potency they can completely normalize Lp(a) in most patients. For Lp(a) lowering, the future is in these new molecules. Randomized trials have started, and we will hopefully have some results in about 5 years,” said Dr. Thanassoulis.
Until then, the prospect of possibly soon documenting benefits from PCSK9 inhibitors beyond their impact on cutting LDL cholesterol raises some hope to get more bang for the considerable buck these drugs cost. But Dr. Thanassoulis was skeptical it would move the cost-benefit ratio much. “VTE and AS are relatively rare, compared with atherosclerotic cardiovascular events, and therefore the added value at the population level would be small,” he predicted. But if treatment with a drug could help patients avoid surgical or percutaneous valve interventions “that could be really interesting from a cost-benefit perspective.”
FOURIER was funded by Amgen, the company that markets evolocumab (Repatha). ODYSSEY OUTCOMES was funded by Sanofi and Regeneron, the companies that developed and market alirocumab (Praluent). Dr. Marston had no disclosures. Dr. Bergmark has been a consultant to Daiichi Sankyo, Janssen, Quark, and Servier and has received research funding from Abbott Vascular, AstraZeneca, and MedImmune. Dr. O’Donoghue has been a consultant to and has received research funding from Amgen; has been a consultant to Janssen and Novartis; and has received research funding from AstraZeneca, Eisai, GlaxoSmithKline, Janssen, Medimmune, Merck, and The Medicines Company. Dr. Thanassoulis has been an adviser to and speaker for Amgen; an adviser to Ionis and Sanofi/Regeneron; a speaker on behalf of Boehringer Ingelheim, Sanofi, and Servier; and has received research funding from Ionis and Servier. Dr. Piazza has been a consultant to Optum, Pfizer, and Thrombolex and he has received research funding from Bayer, Bristol-Myers Squibb, Daiichi Sankyo, Ekos, Janssen, and Portola.
Post hoc analyses of recent large, clinical outcomes studies of PCSK9 inhibitors have revealed two tantalizing and unexpected potential benefits from these drugs: an ability to substantially reduce the incidence or severity of venous thromboembolism and aortic stenosis.
The evidence also suggests that these effects are linked to the ability of these drugs to reduce blood levels of Lp(a) lipoprotein by roughly a quarter, currently the biggest known effect on Lp(a) levels of any approved medication.
One study ran post hoc analyses of venous thromboembolism (VTE) events in the FOURIER pivotal trial of evolocumab (Repatha), with more than 27,500 randomized patients (N Engl J Med. 2017 May 4; 376[18]:1713-22), and in the ODYSSEY OUTCOMES pivotal trial of alirocumab (Praluent), with nearly 19,000 randomized patients (N Engl J Med. 2018 Nov 29;379[22]:2097-2107). The analyses showed that, with evolocumab treatment, the incidence of VTE events fell by a statistically significant 29%, compared with patients on placebo, while in ODYSSEY OUTCOMES patients treated with alirocumab had a 33% cut in VTE events, compared with placebo-treated patients, a difference that just missed statistical significance (Circulation. 2020 Mar 29. doi: 10.1161/CIRCULATIONAHA.120.046524) in analyses that were not prespecified before these trials started, Nicholas A. Marston, MD, said in a presentation of his research during the joint scientific sessions of the American College of Cardiology and the World Heart Federation, which was presented online this year. ACC organizers chose to present parts of the meeting virtually after COVID-19 concerns caused them to cancel the meeting.
A combined analysis of 46,488 patients from both studies showed a 31% cut in VTE events with PCSK9 inhibitor treatment, a highly significant finding using VTE endpoints that were not specifically tallied nor adjudicated but collected as part of the serious adverse event reporting in the two pivotal trials, said Dr. Marston, a cardiologist at Brigham and Women’s Hospital in Boston. This is the first report of a statistically significant link between treatment with PCSK9-inhibiting agents and a reduction in VTE, he added. Researchers from the ODYSSEY OUTCOMES trial had reported a VTE analysis in 2019, and while data from that trial on its own showed a nominal 33% lower VTE rate with alirocumab treatment, it just missed statistical significance.
The VTE effect took about a year on treatment to start to manifest. During the first 12 months of FOURIER, the rate of VTE events among patients in the two treatment arms was virtually identical. But starting during months 13-18 on treatment, the event curves in the two arms began to increasingly diverge, and overall during the period from month 13 to the end of the study treatment with evolocumab was linked with a statistically significant 46% reduction in VTE events, compared with patients who received placebo. The results Dr. Marston reported were also published online (Circulation. 2020 Mar 29. doi: 10.1161/CIRCULATIONAHA.120.046397).
The suggestion that this association may be linked to the impact of PCSK9 inhibitors on Lp(a) came from an additional analysis that Dr. Marston presented, which looked at the link between evolocumab use and a change in VTE event rates, compared with placebo, depending on baseline lipoprotein levels. Evolocumab treatment was associated with a roughly similar, modest, and not statistically significant reduction in VTE events, compared with placebo regardless of whether patients had baseline levels of LDL cholesterol below the median or at or above the median. In contrast, when a similar analysis divided patients based on whether their Lp(a) level at baseline was below, or at or above, the median the results showed no discernible effect of evolocumab treatment, compared with on VTE events in patients with lower baseline Lp(a), but in those with higher levels treatment with evolocumab linked with a 48% cut in VTE events, compared with placebo, a statistically significant difference.
In FOURIER, treatment with evolocumab lowered baseline Lp(a) levels by a median of 27%, compared with placebo, among the 25,096 enrolled patients who had their baseline levels measured. As previously reported, prespecified analysis of FOURIER data also showed that the impact of evolocumab, compared with placebo, on the combined rate of coronary heart disease death, MI, or need for urgent coronary revascularization was enhanced among patients with elevated baseline Lp(a) and moderated in those who entered with lower levels. Among patients who entered FOURIER with Lp(a) levels at or below the median treatment with evolocumab cut the primary endpoint by 7%, compared with placebo, a difference that was not statistically significant. Among patients who began the study with Lp(a) levels above the median, evolocumab treatment cut the primary endpoint by 23%, compared with placebo, a statistically significant effect (Circulation. 2019 Mar 19;139[12]:1483-92).
The aortic stenosis connection
A second study reported in the online scientific sessions (Abstract 914-08) used only FOURIER data, and showed that patients treated with evolocumab had a roughly similar response pattern in their incidence of aortic stenosis (AS) events as they did for VTE events.
During the first year of the study, the incidence of AS events was virtually identical among patients treated with evolocumab and those who received placebo. But after the first 12 months and through the study’s end, patients on evolocumab showed a statistically significant 52% relative reduction in AS events, compared with control patients, said Brian A. Bergmark, MD. For the entire study duration, treatment with evolocumab linked with a 34% relative reduction in AS events, compared with placebo, a difference that did not reach statistical significance, added Dr. Bergmark, an interventional cardiologist also at Brigham and Women’s Hospital. The observed halving in total AS events that linked with evolocumab treatment after the first year of the study included a similar-magnitude reduction specifically in the incidence of aortic valve replacement procedures in the evolocumab-treated patients.
Further analysis of both total AS events and aortic valve replacements in FOURIER patients showed that they occurred at a significantly elevated rate in patients who entered the study with higher baseline Lp(a) levels in a multivariate analysis, but a similar analysis showed no significant association between the incidence of these AS-related events and baseline levels of LDL cholesterol, he said.
The AS analysis carried the same important limitations as the VTE analysis: It ran on a post hoc basis and focused on events that were relatively uncommon and not adjudicated, Dr. Bergmark cautioned. Nonetheless, other investigators saw important potential implications from both the VTE and AS observations, with the huge caveat that they need replication in prospective studies designed to specifically address the validity of these findings.
What it could mean
These observed associations between PCSK9 inhibitor treatment and apparent reductions in the rate of both VTE and AS events “represent a tremendous clinical breakthrough,” commented Michelle L. O’Donoghue, MD, a cardiologist at Brigham and Women’s Hospital who is a FOURIER coinvestigator and has led some of the Lp(a) analyses run from that study.
“To date, we have not identified any therapies that slow progression of AS. Other classes of lipid-lowering therapies, such as statins, have been tested and not demonstrated a significant effect,” Dr. O’Donoghue said in an interview.
“For AS, the results are very intriguing. If confirmed, it could be groundbreaking. AS is the most common valve disease in the developed world, and no medical therapy exists. The potential is immense,” commented George Thanassoulis, MD, director of preventive and genomic cardiology at McGill University, Montreal. “Having a medical treatment that could slow AS progression would completely change the disease. It’s conceivable to slow the disease enough that patients may never require valve replacement.” But an interview he cautioned that, “although the results are exciting, the analysis has many limitations. What we need is a dedicated, randomized trial for AS. I hope this stimulates that.”
“For VTE, it’s an interesting finding, but I don’t think it will have clinical utility because we have good treatment for VTE,” added Dr. Thanassoulis, but others saw more opportunity from what could be a new way to reduce VTE risk.
“Given that many patients have difficulty with the bleeding risk from anticoagulants, this option [a PCSK9 inhibitor] may be quite welcome for preventing VTE,” commented Gregory Piazza, MD, a cardiologist and VTE specialist at Brigham and Women’s Hospital who was not involved in any of the PCSK9 inhibitor studies.
“At this time we would not suggest that PCSK9 inhibitors replace an anticoagulant for patients with an established clot or at high risk for a recurrent clot, but if patients have an indication for a PCSK9 inhibitor, the further reduction in venous clot can be viewed as an additional benefit of this therapy,” said Dr. O’Donoghue.
How it might work
A possible mechanism underlying a VTE effect is unclear. Results from the JUPITER trial more than a decade ago had shown a significant association between treatment with 20 mg/day of rosuvastatin and a cut in VTE episodes, compared with placebo, in a prespecified, secondary analysis of the trial with nearly 18,000 patients selected for having a relatively high level of high-sensitivity C-reactive protein (N Engl J Med. 2009 Apr 30;360[18]:1851-61). But a meta-analysis of 29 controlled statin trials that used a variety of statin types and dosages (and included the JUPITER results) failed to confirm a statistically significant change in VTE rates from statins, though they produced a small, nominal reduction (PLoS Med. 2012 Sep 18. doi: 10.1371/journal.pmed.1001310).
Lp(a) “has long been linked to thrombosis, in particular arterial thrombosis,” so the link observed in the PCSK9 inhibitor trials “is not surprising,” said Dr. Piazza. Dr. O’Donoghue agreed that prior evidence had “suggested a prothrombotic role for Lp(a).”
Dr. Thanassoulis was more skeptical of a Lp(a) connection to VTE. “There has always been controversy regarding the prothrombotic effects of Lp(a) and whether it’s clinically relevant,” he said. “The genetic data, from Mendelian randomization studies, is not consistent” with a Lp(a) and VTE link.
The association of AS and Lp(a) may be stronger. “Our team showed that people with genetic variants that predispose to high Lp(a) have a much higher incidence of AS,” Dr. Thanassoulis noted. “We and others have also demonstrated that both Lp(a) and LDL are likely causal mediators of aortic valve calcification and stenosis.”
Dr. O’Donoghue also cited observational genetic data that linked elevated Lp(a) with AS. “Mendelian randomization studies have demonstrated that Lp(a) is a causal contributer to AS, and evolocumab reduced Lp(a) by 25%-30%, raising the possibility that Lp(a) lowering with these drugs may be the mechanism,” she said.
The future of Lp(a) lowering
This last point from Dr. O’Donoghue, that PCSK9 inhibitors cut Lp(a) levels by about 25%-30%, means that they are the most potent Lp(a)-lowering agents currently available, but it also leaves lots of room for other agents to do even better in cutting Lp(a).
“There are now drugs in development that block production of the Lp(a) protein and dramatically reduce its concentration, by about 80%,” Dr. O’Donoghue noted. “It will be of interest to study whether these novel therapies, now in phase 2 and phase 3 studies, have any effect on the risk for VTE and AS.”
“Several drugs in development, including antisense RNA and RNA-interfering molecules, are much more potent and lower Lp(a) by 80%-90%. Because of this potency they can completely normalize Lp(a) in most patients. For Lp(a) lowering, the future is in these new molecules. Randomized trials have started, and we will hopefully have some results in about 5 years,” said Dr. Thanassoulis.
Until then, the prospect of possibly soon documenting benefits from PCSK9 inhibitors beyond their impact on cutting LDL cholesterol raises some hope to get more bang for the considerable buck these drugs cost. But Dr. Thanassoulis was skeptical it would move the cost-benefit ratio much. “VTE and AS are relatively rare, compared with atherosclerotic cardiovascular events, and therefore the added value at the population level would be small,” he predicted. But if treatment with a drug could help patients avoid surgical or percutaneous valve interventions “that could be really interesting from a cost-benefit perspective.”
FOURIER was funded by Amgen, the company that markets evolocumab (Repatha). ODYSSEY OUTCOMES was funded by Sanofi and Regeneron, the companies that developed and market alirocumab (Praluent). Dr. Marston had no disclosures. Dr. Bergmark has been a consultant to Daiichi Sankyo, Janssen, Quark, and Servier and has received research funding from Abbott Vascular, AstraZeneca, and MedImmune. Dr. O’Donoghue has been a consultant to and has received research funding from Amgen; has been a consultant to Janssen and Novartis; and has received research funding from AstraZeneca, Eisai, GlaxoSmithKline, Janssen, Medimmune, Merck, and The Medicines Company. Dr. Thanassoulis has been an adviser to and speaker for Amgen; an adviser to Ionis and Sanofi/Regeneron; a speaker on behalf of Boehringer Ingelheim, Sanofi, and Servier; and has received research funding from Ionis and Servier. Dr. Piazza has been a consultant to Optum, Pfizer, and Thrombolex and he has received research funding from Bayer, Bristol-Myers Squibb, Daiichi Sankyo, Ekos, Janssen, and Portola.
REPORTING FROM ACC 20
Are providers asking about menstrual bleeding before/during anticoagulant therapy?
ORLANDO – A small study suggests health care providers may fail to ask patients about heavy menstrual bleeding before or during treatment with oral anticoagulants.
Researchers performed a chart review at a single center, which indicated that 60% of women were not asked about heavy menstrual bleeding before they were prescribed an oral anticoagulant.
Six months after the women started anticoagulant therapy, 29% required treatment for heavy menstrual bleeding. Charts for the remaining 71% of women contained no information about heavy menstrual bleeding.
“We were unable to distinguish between true absence of heavy menstrual bleeding and absence of reporting,” said Bethany T. Samuelson Bannow, MD, of Oregon Health & Science University, Portland.
Dr. Samuelson Bannow presented these findings at the annual meeting of the American Society of Hematology.
She explained that heavy menstrual bleeding is defined as more than 80 mL of blood loss per cycle. It affects 10%-15% of women in their lifetime, and anticoagulants increase the risk of heavy menstrual bleeding.
Studies have shown that heavy menstrual bleeding occurs in 22%-65% of women treated with vitamin K agonists and 20%-27% of women treated with rivaroxaban (Blood. 2017;130[24]:2603-9). However, many anticoagulant studies don’t include heavy menstrual bleeding as an outcome.
To gain more insight, Dr. Samuelson Bannow and colleagues conducted a chart review. Their study included 236 women of reproductive age treated at Oregon Health & Science University between Jan. 1, 2012, and Dec. 31, 2018.
The patients’ median age was 37 years (range, 18-50 years). Most patients (67%) were receiving an oral anticoagulant for venous thromboembolism. The rest were on anticoagulant therapy for arterial thrombosis (6%), atrial fibrillation (6%), a mechanical valve (1%), or “other” reasons (20%).
Dr. Samuelson Bannow said the other group was “almost exclusively women who were receiving prophylaxis” postoperatively or for travel. Most women in this group were receiving rivaroxaban.
Rivaroxaban was the most commonly prescribed anticoagulant in the entire cohort (41%), followed by warfarin (34%) and apixaban (25%).
At the time of anticoagulant prescription, 12% of women reported a history of heavy menstrual bleeding, and 28% did not. For most patients – 60% – there was no discussion of menstrual history documented.
Six months after starting oral anticoagulant therapy, 29% of patients required treatment for heavy menstrual bleeding. For 71% of patients, there was no documentation on the treatment of heavy menstrual bleeding.
Treatment for heavy menstrual bleeding was required in 33% of patients on rivaroxaban, 24% of those on apixaban, and 29% of those on warfarin, a significant difference (P less than .001).
“Rates of heavy menstrual bleeding … are higher in rivaroxaban users,” Dr. Samuelson Bannow said. “This is not the first study to demonstrate this. However, [the rate of heavy menstrual bleeding in this study] is still a lot lower than we would expect based on past levels with warfarin. This tells us we’re probably missing a lot of heavy menstrual bleeding. That’s not too surprising considering how few providers are actually asking about the menses.”
Dr. Samuelson Bannow and colleagues disclosed no conflicts of interest.
SOURCE: Samuelson Bannow BT et al. ASH 2019, Abstract 60.
ORLANDO – A small study suggests health care providers may fail to ask patients about heavy menstrual bleeding before or during treatment with oral anticoagulants.
Researchers performed a chart review at a single center, which indicated that 60% of women were not asked about heavy menstrual bleeding before they were prescribed an oral anticoagulant.
Six months after the women started anticoagulant therapy, 29% required treatment for heavy menstrual bleeding. Charts for the remaining 71% of women contained no information about heavy menstrual bleeding.
“We were unable to distinguish between true absence of heavy menstrual bleeding and absence of reporting,” said Bethany T. Samuelson Bannow, MD, of Oregon Health & Science University, Portland.
Dr. Samuelson Bannow presented these findings at the annual meeting of the American Society of Hematology.
She explained that heavy menstrual bleeding is defined as more than 80 mL of blood loss per cycle. It affects 10%-15% of women in their lifetime, and anticoagulants increase the risk of heavy menstrual bleeding.
Studies have shown that heavy menstrual bleeding occurs in 22%-65% of women treated with vitamin K agonists and 20%-27% of women treated with rivaroxaban (Blood. 2017;130[24]:2603-9). However, many anticoagulant studies don’t include heavy menstrual bleeding as an outcome.
To gain more insight, Dr. Samuelson Bannow and colleagues conducted a chart review. Their study included 236 women of reproductive age treated at Oregon Health & Science University between Jan. 1, 2012, and Dec. 31, 2018.
The patients’ median age was 37 years (range, 18-50 years). Most patients (67%) were receiving an oral anticoagulant for venous thromboembolism. The rest were on anticoagulant therapy for arterial thrombosis (6%), atrial fibrillation (6%), a mechanical valve (1%), or “other” reasons (20%).
Dr. Samuelson Bannow said the other group was “almost exclusively women who were receiving prophylaxis” postoperatively or for travel. Most women in this group were receiving rivaroxaban.
Rivaroxaban was the most commonly prescribed anticoagulant in the entire cohort (41%), followed by warfarin (34%) and apixaban (25%).
At the time of anticoagulant prescription, 12% of women reported a history of heavy menstrual bleeding, and 28% did not. For most patients – 60% – there was no discussion of menstrual history documented.
Six months after starting oral anticoagulant therapy, 29% of patients required treatment for heavy menstrual bleeding. For 71% of patients, there was no documentation on the treatment of heavy menstrual bleeding.
Treatment for heavy menstrual bleeding was required in 33% of patients on rivaroxaban, 24% of those on apixaban, and 29% of those on warfarin, a significant difference (P less than .001).
“Rates of heavy menstrual bleeding … are higher in rivaroxaban users,” Dr. Samuelson Bannow said. “This is not the first study to demonstrate this. However, [the rate of heavy menstrual bleeding in this study] is still a lot lower than we would expect based on past levels with warfarin. This tells us we’re probably missing a lot of heavy menstrual bleeding. That’s not too surprising considering how few providers are actually asking about the menses.”
Dr. Samuelson Bannow and colleagues disclosed no conflicts of interest.
SOURCE: Samuelson Bannow BT et al. ASH 2019, Abstract 60.
ORLANDO – A small study suggests health care providers may fail to ask patients about heavy menstrual bleeding before or during treatment with oral anticoagulants.
Researchers performed a chart review at a single center, which indicated that 60% of women were not asked about heavy menstrual bleeding before they were prescribed an oral anticoagulant.
Six months after the women started anticoagulant therapy, 29% required treatment for heavy menstrual bleeding. Charts for the remaining 71% of women contained no information about heavy menstrual bleeding.
“We were unable to distinguish between true absence of heavy menstrual bleeding and absence of reporting,” said Bethany T. Samuelson Bannow, MD, of Oregon Health & Science University, Portland.
Dr. Samuelson Bannow presented these findings at the annual meeting of the American Society of Hematology.
She explained that heavy menstrual bleeding is defined as more than 80 mL of blood loss per cycle. It affects 10%-15% of women in their lifetime, and anticoagulants increase the risk of heavy menstrual bleeding.
Studies have shown that heavy menstrual bleeding occurs in 22%-65% of women treated with vitamin K agonists and 20%-27% of women treated with rivaroxaban (Blood. 2017;130[24]:2603-9). However, many anticoagulant studies don’t include heavy menstrual bleeding as an outcome.
To gain more insight, Dr. Samuelson Bannow and colleagues conducted a chart review. Their study included 236 women of reproductive age treated at Oregon Health & Science University between Jan. 1, 2012, and Dec. 31, 2018.
The patients’ median age was 37 years (range, 18-50 years). Most patients (67%) were receiving an oral anticoagulant for venous thromboembolism. The rest were on anticoagulant therapy for arterial thrombosis (6%), atrial fibrillation (6%), a mechanical valve (1%), or “other” reasons (20%).
Dr. Samuelson Bannow said the other group was “almost exclusively women who were receiving prophylaxis” postoperatively or for travel. Most women in this group were receiving rivaroxaban.
Rivaroxaban was the most commonly prescribed anticoagulant in the entire cohort (41%), followed by warfarin (34%) and apixaban (25%).
At the time of anticoagulant prescription, 12% of women reported a history of heavy menstrual bleeding, and 28% did not. For most patients – 60% – there was no discussion of menstrual history documented.
Six months after starting oral anticoagulant therapy, 29% of patients required treatment for heavy menstrual bleeding. For 71% of patients, there was no documentation on the treatment of heavy menstrual bleeding.
Treatment for heavy menstrual bleeding was required in 33% of patients on rivaroxaban, 24% of those on apixaban, and 29% of those on warfarin, a significant difference (P less than .001).
“Rates of heavy menstrual bleeding … are higher in rivaroxaban users,” Dr. Samuelson Bannow said. “This is not the first study to demonstrate this. However, [the rate of heavy menstrual bleeding in this study] is still a lot lower than we would expect based on past levels with warfarin. This tells us we’re probably missing a lot of heavy menstrual bleeding. That’s not too surprising considering how few providers are actually asking about the menses.”
Dr. Samuelson Bannow and colleagues disclosed no conflicts of interest.
SOURCE: Samuelson Bannow BT et al. ASH 2019, Abstract 60.
REPORTING FROM ASH 2019
New ASH guideline: VTE prophylaxis after major surgery
ORLANDO – The latest American Society of Hematology guideline on venous thromboembolism (VTE) tackles 30 key questions regarding prophylaxis in hospitalized patients undergoing surgery, according to the chair of the guideline panel, who highlighted 9 of those questions during a special session at the society’s annual meeting.
The clinical practice guideline, published just about a week before the annual meeting of the American Society of Hematology, focuses mainly on pharmacologic prophylaxis in specific surgical settings, said David R. Anderson, MD, dean of the faculty of medicine of Dalhousie University, Halifax, N.S.
“Our guidelines focused upon clinically important symptomatic outcomes, with less emphasis being placed on asymptomatic deep vein thrombosis detected by screening tests,” Dr. Anderson said.
At the special education session, Dr. Anderson highlighted several specific recommendations on prophylaxis in surgical patients.
Pharmacologic prophylaxis is not recommended for patients experiencing major trauma deemed to be at high risk of bleeding. Its use does reduce risk of symptomatic pulmonary embolism (PE) and deep vein thrombosis (DVT) by about 10 events per 1,000 patients treated; however, Dr. Anderson said, the panel’s opinion was that this benefit was outweighed by increased risk of major bleeding, at 24 events per 1,000 patients treated.
“We do recommend, however that this risk of bleeding must be reevaluated over the course of recovery of patients, and this may change the decision around this intervention over time,” Dr. Anderson told attendees at the special session.
That’s because pharmacologic prophylaxis is recommended in surgical patients at low to moderate risk of bleeding. In this scenario, the incremental risk of major bleeding (14 events per 1,000 patients treated) is outweighed by the benefit of the reduction of symptomatic VTE events, according to Dr. Anderson.
When pharmacologic prophylaxis is used, the panel recommends combined prophylaxis – mechanical prophylaxis in addition to pharmacologic prophylaxis – especially in those patients at high or very high risk of VTE. Evidence shows that the combination approach significantly reduces risk of PE, and strongly suggests it may also reduce risk of symptomatic proximal DVT, Dr. Anderson said.
In surgical patients not receiving pharmacologic prophylaxis, mechanical prophylaxis is recommended over no mechanical prophylaxis, he added. Moreover, in those patients receiving mechanical prophylaxis, the ASH panel recommends use of intermittent compression devices over graduated compression stockings.
The panel comes out against prophylactic inferior vena cava (IVC) filter insertion in the guidelines. Dr. Anderson said that the “small reduction” in PE risk seen in observational studies is outweighed by increased risk of DVT, and a resulting trend for increased mortality, associated with insertion of the devices.
“We did not consider other risks of IVC filters such as filter embolization or perforation, which again would be complications that would support our recommendation against routine use of these devices in patients undergoing major surgery,” he said.
In terms of the type of pharmacologic prophylaxis to use, the panel said low-molecular-weight heparin or unfractionated heparin would be reasonable choices in this setting. Available data do not demonstrate any significant differences between these choices for major clinical outcomes, Dr. Anderson added.
The guideline also addresses duration of pharmacologic prophylaxis, stating that extended prophylaxis – of at least 3 weeks – is favored over short-term prophylaxis, or up to 2 weeks of treatment. The extended approach significantly reduces risk of symptomatic PE and proximal DVT, though most of the supporting data come from studies of major joint arthroplasty and major general surgical procedures for patients with cancer. “We need more studies in other clinical areas to examine this particular question,” Dr. Anderson said.
The guideline on prophylaxis in surgical patients was published in Blood Advances (2019 Dec 3;3[23]:3898-944). Six other ASH VTE guidelines, all published in 2018, covered prophylaxis in medical patients, diagnosis, VTE in pregnancy, optimal anticoagulation, heparin-induced thrombocytopenia, and pediatric considerations. The guidelines are available on the ASH website.
Dr. Anderson reported having no relevant conflicts of interest.
ORLANDO – The latest American Society of Hematology guideline on venous thromboembolism (VTE) tackles 30 key questions regarding prophylaxis in hospitalized patients undergoing surgery, according to the chair of the guideline panel, who highlighted 9 of those questions during a special session at the society’s annual meeting.
The clinical practice guideline, published just about a week before the annual meeting of the American Society of Hematology, focuses mainly on pharmacologic prophylaxis in specific surgical settings, said David R. Anderson, MD, dean of the faculty of medicine of Dalhousie University, Halifax, N.S.
“Our guidelines focused upon clinically important symptomatic outcomes, with less emphasis being placed on asymptomatic deep vein thrombosis detected by screening tests,” Dr. Anderson said.
At the special education session, Dr. Anderson highlighted several specific recommendations on prophylaxis in surgical patients.
Pharmacologic prophylaxis is not recommended for patients experiencing major trauma deemed to be at high risk of bleeding. Its use does reduce risk of symptomatic pulmonary embolism (PE) and deep vein thrombosis (DVT) by about 10 events per 1,000 patients treated; however, Dr. Anderson said, the panel’s opinion was that this benefit was outweighed by increased risk of major bleeding, at 24 events per 1,000 patients treated.
“We do recommend, however that this risk of bleeding must be reevaluated over the course of recovery of patients, and this may change the decision around this intervention over time,” Dr. Anderson told attendees at the special session.
That’s because pharmacologic prophylaxis is recommended in surgical patients at low to moderate risk of bleeding. In this scenario, the incremental risk of major bleeding (14 events per 1,000 patients treated) is outweighed by the benefit of the reduction of symptomatic VTE events, according to Dr. Anderson.
When pharmacologic prophylaxis is used, the panel recommends combined prophylaxis – mechanical prophylaxis in addition to pharmacologic prophylaxis – especially in those patients at high or very high risk of VTE. Evidence shows that the combination approach significantly reduces risk of PE, and strongly suggests it may also reduce risk of symptomatic proximal DVT, Dr. Anderson said.
In surgical patients not receiving pharmacologic prophylaxis, mechanical prophylaxis is recommended over no mechanical prophylaxis, he added. Moreover, in those patients receiving mechanical prophylaxis, the ASH panel recommends use of intermittent compression devices over graduated compression stockings.
The panel comes out against prophylactic inferior vena cava (IVC) filter insertion in the guidelines. Dr. Anderson said that the “small reduction” in PE risk seen in observational studies is outweighed by increased risk of DVT, and a resulting trend for increased mortality, associated with insertion of the devices.
“We did not consider other risks of IVC filters such as filter embolization or perforation, which again would be complications that would support our recommendation against routine use of these devices in patients undergoing major surgery,” he said.
In terms of the type of pharmacologic prophylaxis to use, the panel said low-molecular-weight heparin or unfractionated heparin would be reasonable choices in this setting. Available data do not demonstrate any significant differences between these choices for major clinical outcomes, Dr. Anderson added.
The guideline also addresses duration of pharmacologic prophylaxis, stating that extended prophylaxis – of at least 3 weeks – is favored over short-term prophylaxis, or up to 2 weeks of treatment. The extended approach significantly reduces risk of symptomatic PE and proximal DVT, though most of the supporting data come from studies of major joint arthroplasty and major general surgical procedures for patients with cancer. “We need more studies in other clinical areas to examine this particular question,” Dr. Anderson said.
The guideline on prophylaxis in surgical patients was published in Blood Advances (2019 Dec 3;3[23]:3898-944). Six other ASH VTE guidelines, all published in 2018, covered prophylaxis in medical patients, diagnosis, VTE in pregnancy, optimal anticoagulation, heparin-induced thrombocytopenia, and pediatric considerations. The guidelines are available on the ASH website.
Dr. Anderson reported having no relevant conflicts of interest.
ORLANDO – The latest American Society of Hematology guideline on venous thromboembolism (VTE) tackles 30 key questions regarding prophylaxis in hospitalized patients undergoing surgery, according to the chair of the guideline panel, who highlighted 9 of those questions during a special session at the society’s annual meeting.
The clinical practice guideline, published just about a week before the annual meeting of the American Society of Hematology, focuses mainly on pharmacologic prophylaxis in specific surgical settings, said David R. Anderson, MD, dean of the faculty of medicine of Dalhousie University, Halifax, N.S.
“Our guidelines focused upon clinically important symptomatic outcomes, with less emphasis being placed on asymptomatic deep vein thrombosis detected by screening tests,” Dr. Anderson said.
At the special education session, Dr. Anderson highlighted several specific recommendations on prophylaxis in surgical patients.
Pharmacologic prophylaxis is not recommended for patients experiencing major trauma deemed to be at high risk of bleeding. Its use does reduce risk of symptomatic pulmonary embolism (PE) and deep vein thrombosis (DVT) by about 10 events per 1,000 patients treated; however, Dr. Anderson said, the panel’s opinion was that this benefit was outweighed by increased risk of major bleeding, at 24 events per 1,000 patients treated.
“We do recommend, however that this risk of bleeding must be reevaluated over the course of recovery of patients, and this may change the decision around this intervention over time,” Dr. Anderson told attendees at the special session.
That’s because pharmacologic prophylaxis is recommended in surgical patients at low to moderate risk of bleeding. In this scenario, the incremental risk of major bleeding (14 events per 1,000 patients treated) is outweighed by the benefit of the reduction of symptomatic VTE events, according to Dr. Anderson.
When pharmacologic prophylaxis is used, the panel recommends combined prophylaxis – mechanical prophylaxis in addition to pharmacologic prophylaxis – especially in those patients at high or very high risk of VTE. Evidence shows that the combination approach significantly reduces risk of PE, and strongly suggests it may also reduce risk of symptomatic proximal DVT, Dr. Anderson said.
In surgical patients not receiving pharmacologic prophylaxis, mechanical prophylaxis is recommended over no mechanical prophylaxis, he added. Moreover, in those patients receiving mechanical prophylaxis, the ASH panel recommends use of intermittent compression devices over graduated compression stockings.
The panel comes out against prophylactic inferior vena cava (IVC) filter insertion in the guidelines. Dr. Anderson said that the “small reduction” in PE risk seen in observational studies is outweighed by increased risk of DVT, and a resulting trend for increased mortality, associated with insertion of the devices.
“We did not consider other risks of IVC filters such as filter embolization or perforation, which again would be complications that would support our recommendation against routine use of these devices in patients undergoing major surgery,” he said.
In terms of the type of pharmacologic prophylaxis to use, the panel said low-molecular-weight heparin or unfractionated heparin would be reasonable choices in this setting. Available data do not demonstrate any significant differences between these choices for major clinical outcomes, Dr. Anderson added.
The guideline also addresses duration of pharmacologic prophylaxis, stating that extended prophylaxis – of at least 3 weeks – is favored over short-term prophylaxis, or up to 2 weeks of treatment. The extended approach significantly reduces risk of symptomatic PE and proximal DVT, though most of the supporting data come from studies of major joint arthroplasty and major general surgical procedures for patients with cancer. “We need more studies in other clinical areas to examine this particular question,” Dr. Anderson said.
The guideline on prophylaxis in surgical patients was published in Blood Advances (2019 Dec 3;3[23]:3898-944). Six other ASH VTE guidelines, all published in 2018, covered prophylaxis in medical patients, diagnosis, VTE in pregnancy, optimal anticoagulation, heparin-induced thrombocytopenia, and pediatric considerations. The guidelines are available on the ASH website.
Dr. Anderson reported having no relevant conflicts of interest.
EXPERT ANALYSIS FROM ASH 2019
Smokers with PE have higher rate of hospital readmission
NEW ORLEANS – , according to a retrospective study.
The rate of readmission was significantly higher among patients with tobacco dependence, and tobacco dependence was independently associated with an increased risk of readmission.
“This is the first study to quantify the increased rate of hospital readmission due to smoking,” said study investigator Kam Sing Ho, MD, of Mount Sinai St. Luke’s and Mount Sinai West, New York.
Dr. Ho and colleagues described this study and its results in a poster presented at the annual meeting of the American College of Chest Physicians.
The researchers analyzed data on 168,891 hospital admissions of adults with PE, 34.2% of whom had tobacco dependence. Patients with and without tobacco dependence were propensity matched for baseline characteristics (n = 24,262 in each group).
The 30-day readmission rate was significantly higher in patients with tobacco dependence than in those without it – 11.0% and 8.9%, respectively (P less than .001). The most common reason for readmission in both groups was PE.
Dr. Ho said the higher readmission rate among patients with tobacco dependence might be explained by the fact that smokers have a higher level of fibrinogen, which may affect blood viscosity and contribute to thrombus formation (Proc Am Thorac Soc. 2005;2[1]:71-7).
The investigators also found that tobacco dependence was an independent predictor of readmission (hazard ratio, 1.43; P less than .001). And the mortality rate was significantly higher after readmission than after index admission – 6.27% and 3.15%, respectively (P less than .001).
The increased risk of readmission and death among smokers highlights the importance of smoking cessation services. Dr. Ho cited previous research suggesting these services are underused in the hospital setting (BMJ Qual Improv Rep. 2014;3[1]:u204964.w2110).
“Given that smoking is a common phenomenon among patients admitted with pulmonary embolism, we suggest that more rigorous smoking cessation services are implemented prior to discharge for all active smokers,” Dr. Ho said. “[P]atients have the right to be informed on the benefits of smoking cessation and the autonomy to choose. Future research will focus on implementing inpatient smoking cessation at our hospital and its effect on local readmission rate, health resources utilization, and mortality.”
Dr. Ho has no relevant relationships to disclose.
SOURCE: Ho KS et al. CHEST 2019 October. doi: 10.1016/j.chest.2019.08.1551.
NEW ORLEANS – , according to a retrospective study.
The rate of readmission was significantly higher among patients with tobacco dependence, and tobacco dependence was independently associated with an increased risk of readmission.
“This is the first study to quantify the increased rate of hospital readmission due to smoking,” said study investigator Kam Sing Ho, MD, of Mount Sinai St. Luke’s and Mount Sinai West, New York.
Dr. Ho and colleagues described this study and its results in a poster presented at the annual meeting of the American College of Chest Physicians.
The researchers analyzed data on 168,891 hospital admissions of adults with PE, 34.2% of whom had tobacco dependence. Patients with and without tobacco dependence were propensity matched for baseline characteristics (n = 24,262 in each group).
The 30-day readmission rate was significantly higher in patients with tobacco dependence than in those without it – 11.0% and 8.9%, respectively (P less than .001). The most common reason for readmission in both groups was PE.
Dr. Ho said the higher readmission rate among patients with tobacco dependence might be explained by the fact that smokers have a higher level of fibrinogen, which may affect blood viscosity and contribute to thrombus formation (Proc Am Thorac Soc. 2005;2[1]:71-7).
The investigators also found that tobacco dependence was an independent predictor of readmission (hazard ratio, 1.43; P less than .001). And the mortality rate was significantly higher after readmission than after index admission – 6.27% and 3.15%, respectively (P less than .001).
The increased risk of readmission and death among smokers highlights the importance of smoking cessation services. Dr. Ho cited previous research suggesting these services are underused in the hospital setting (BMJ Qual Improv Rep. 2014;3[1]:u204964.w2110).
“Given that smoking is a common phenomenon among patients admitted with pulmonary embolism, we suggest that more rigorous smoking cessation services are implemented prior to discharge for all active smokers,” Dr. Ho said. “[P]atients have the right to be informed on the benefits of smoking cessation and the autonomy to choose. Future research will focus on implementing inpatient smoking cessation at our hospital and its effect on local readmission rate, health resources utilization, and mortality.”
Dr. Ho has no relevant relationships to disclose.
SOURCE: Ho KS et al. CHEST 2019 October. doi: 10.1016/j.chest.2019.08.1551.
NEW ORLEANS – , according to a retrospective study.
The rate of readmission was significantly higher among patients with tobacco dependence, and tobacco dependence was independently associated with an increased risk of readmission.
“This is the first study to quantify the increased rate of hospital readmission due to smoking,” said study investigator Kam Sing Ho, MD, of Mount Sinai St. Luke’s and Mount Sinai West, New York.
Dr. Ho and colleagues described this study and its results in a poster presented at the annual meeting of the American College of Chest Physicians.
The researchers analyzed data on 168,891 hospital admissions of adults with PE, 34.2% of whom had tobacco dependence. Patients with and without tobacco dependence were propensity matched for baseline characteristics (n = 24,262 in each group).
The 30-day readmission rate was significantly higher in patients with tobacco dependence than in those without it – 11.0% and 8.9%, respectively (P less than .001). The most common reason for readmission in both groups was PE.
Dr. Ho said the higher readmission rate among patients with tobacco dependence might be explained by the fact that smokers have a higher level of fibrinogen, which may affect blood viscosity and contribute to thrombus formation (Proc Am Thorac Soc. 2005;2[1]:71-7).
The investigators also found that tobacco dependence was an independent predictor of readmission (hazard ratio, 1.43; P less than .001). And the mortality rate was significantly higher after readmission than after index admission – 6.27% and 3.15%, respectively (P less than .001).
The increased risk of readmission and death among smokers highlights the importance of smoking cessation services. Dr. Ho cited previous research suggesting these services are underused in the hospital setting (BMJ Qual Improv Rep. 2014;3[1]:u204964.w2110).
“Given that smoking is a common phenomenon among patients admitted with pulmonary embolism, we suggest that more rigorous smoking cessation services are implemented prior to discharge for all active smokers,” Dr. Ho said. “[P]atients have the right to be informed on the benefits of smoking cessation and the autonomy to choose. Future research will focus on implementing inpatient smoking cessation at our hospital and its effect on local readmission rate, health resources utilization, and mortality.”
Dr. Ho has no relevant relationships to disclose.
SOURCE: Ho KS et al. CHEST 2019 October. doi: 10.1016/j.chest.2019.08.1551.
REPORTING FROM CHEST 2019
Thromboembolic events more likely among CIDP patients with CVAD
AUSTIN, TEX. – Patients with chronic inflammatory demyelinating polyneuropathy (CIDP) who receive intravenous immunoglobulin (IVIg) appear to have an increased risk of thromboembolic events if it is administered with a central venous access device (CVAD) when compared against those without a CVAD, according to a recent study.
Although CVADs can reliably deliver IVIg, they also represent an established risk factor for thromboembolic events, Ami Patel, PhD, a senior epidemiologist at CSL Behring, and colleagues noted on their poster at the annual meeting of the American Association for Neuromuscular and Electrodiagnostic Medicine.
The results suggest a need for physicians to be vigilant about patients’ potential risk factors for thromboembolic events, Dr. Patel said in an interview. Further research is planned, however, because the current study did not control for other risk factors or explore other possible confounding, she said.
Dr. Patel and her associates analyzed U.S. claims data (IBM/Truven MarketScan) from 2006 to 2018 and included all patients with a CIDP diagnosis claim and a postdiagnosis code for IVIg. A code for CVAD up to 2 months before CIDP diagnosis without removal before IVIg treatment ended determined those with CVAD exposure, and thromboembolic events included any codes related to arterial, venous, or vascular prostheses.
The researchers then compared patients in a case-control fashion, matching each one with a CVAD to five patients of similar demographics without a CVAD. Characteristics used for matching included medical insurance type, prescription data availability, sex, age, geographic region, and years enrolled in the database.
Among 7,447 patients with at least one IVIg claim, 11.8% (n = 882) had CVAD exposure and 88.2% (n = 6,565) did not. Of those without a CVAD, 3,642 patients were matched to patients with CVAD. A quarter (25.4%) of patients with a CVAD had a thromboembolic event, compared with 11.2% of matched patients without CVADs (P less than .0001).
In the year leading up to IVIg therapy, 16.9% of those with a CVAD and 10.9% of matched patients without one had a previous thromboembolic event (P less than .0001). Patients with a CVAD also had significantly higher rates of hypertension (51.9% vs. 45.0% with placebo; P less than .001) and anticoagulation therapy (7.0% vs. 5.2% with placebo; P less than .05). Differences between the groups were not significant for diabetes (26.9% vs. 24.2%) and hyperlipidemia (19.1% vs. 17.8%).
Occlusion and stenosis of the carotid artery was the most common arterial thromboembolic outcome, occurring in 5.3% of those with a CVAD and in 2.8% of those without a CVAD. The most common venous thromboembolic event was acute venous embolism and thrombosis of lower-extremity deep vessels, which occurred in 7% of those with a CVAD and in 1.8% of those without.
The researchers also compared inpatient admissions and emergency department visits among those with and without a CVAD; both rates were higher in patients with a CVAD. Visits to the emergency department occurred at a rate of 0.14 events per month for those with a CVAD (2.01 distinct months with a claim) and 0.09 events per month for those without a CVAD (0.65 distinct months with a claim). Patients with a CVAD had 1.44 months with an inpatient admissions claim, in comparison with 0.41 months among matched patients without a CVAD. Inpatient admission frequency per month was 0.14 for those with a CVAD and 0.08 for those without.
The research was funded by CSL Behring. Dr. Patel and two of the other five authors are employees of CSL Behring.
SOURCE: Patel A et al. AANEM 2019, Abstract 94.
AUSTIN, TEX. – Patients with chronic inflammatory demyelinating polyneuropathy (CIDP) who receive intravenous immunoglobulin (IVIg) appear to have an increased risk of thromboembolic events if it is administered with a central venous access device (CVAD) when compared against those without a CVAD, according to a recent study.
Although CVADs can reliably deliver IVIg, they also represent an established risk factor for thromboembolic events, Ami Patel, PhD, a senior epidemiologist at CSL Behring, and colleagues noted on their poster at the annual meeting of the American Association for Neuromuscular and Electrodiagnostic Medicine.
The results suggest a need for physicians to be vigilant about patients’ potential risk factors for thromboembolic events, Dr. Patel said in an interview. Further research is planned, however, because the current study did not control for other risk factors or explore other possible confounding, she said.
Dr. Patel and her associates analyzed U.S. claims data (IBM/Truven MarketScan) from 2006 to 2018 and included all patients with a CIDP diagnosis claim and a postdiagnosis code for IVIg. A code for CVAD up to 2 months before CIDP diagnosis without removal before IVIg treatment ended determined those with CVAD exposure, and thromboembolic events included any codes related to arterial, venous, or vascular prostheses.
The researchers then compared patients in a case-control fashion, matching each one with a CVAD to five patients of similar demographics without a CVAD. Characteristics used for matching included medical insurance type, prescription data availability, sex, age, geographic region, and years enrolled in the database.
Among 7,447 patients with at least one IVIg claim, 11.8% (n = 882) had CVAD exposure and 88.2% (n = 6,565) did not. Of those without a CVAD, 3,642 patients were matched to patients with CVAD. A quarter (25.4%) of patients with a CVAD had a thromboembolic event, compared with 11.2% of matched patients without CVADs (P less than .0001).
In the year leading up to IVIg therapy, 16.9% of those with a CVAD and 10.9% of matched patients without one had a previous thromboembolic event (P less than .0001). Patients with a CVAD also had significantly higher rates of hypertension (51.9% vs. 45.0% with placebo; P less than .001) and anticoagulation therapy (7.0% vs. 5.2% with placebo; P less than .05). Differences between the groups were not significant for diabetes (26.9% vs. 24.2%) and hyperlipidemia (19.1% vs. 17.8%).
Occlusion and stenosis of the carotid artery was the most common arterial thromboembolic outcome, occurring in 5.3% of those with a CVAD and in 2.8% of those without a CVAD. The most common venous thromboembolic event was acute venous embolism and thrombosis of lower-extremity deep vessels, which occurred in 7% of those with a CVAD and in 1.8% of those without.
The researchers also compared inpatient admissions and emergency department visits among those with and without a CVAD; both rates were higher in patients with a CVAD. Visits to the emergency department occurred at a rate of 0.14 events per month for those with a CVAD (2.01 distinct months with a claim) and 0.09 events per month for those without a CVAD (0.65 distinct months with a claim). Patients with a CVAD had 1.44 months with an inpatient admissions claim, in comparison with 0.41 months among matched patients without a CVAD. Inpatient admission frequency per month was 0.14 for those with a CVAD and 0.08 for those without.
The research was funded by CSL Behring. Dr. Patel and two of the other five authors are employees of CSL Behring.
SOURCE: Patel A et al. AANEM 2019, Abstract 94.
AUSTIN, TEX. – Patients with chronic inflammatory demyelinating polyneuropathy (CIDP) who receive intravenous immunoglobulin (IVIg) appear to have an increased risk of thromboembolic events if it is administered with a central venous access device (CVAD) when compared against those without a CVAD, according to a recent study.
Although CVADs can reliably deliver IVIg, they also represent an established risk factor for thromboembolic events, Ami Patel, PhD, a senior epidemiologist at CSL Behring, and colleagues noted on their poster at the annual meeting of the American Association for Neuromuscular and Electrodiagnostic Medicine.
The results suggest a need for physicians to be vigilant about patients’ potential risk factors for thromboembolic events, Dr. Patel said in an interview. Further research is planned, however, because the current study did not control for other risk factors or explore other possible confounding, she said.
Dr. Patel and her associates analyzed U.S. claims data (IBM/Truven MarketScan) from 2006 to 2018 and included all patients with a CIDP diagnosis claim and a postdiagnosis code for IVIg. A code for CVAD up to 2 months before CIDP diagnosis without removal before IVIg treatment ended determined those with CVAD exposure, and thromboembolic events included any codes related to arterial, venous, or vascular prostheses.
The researchers then compared patients in a case-control fashion, matching each one with a CVAD to five patients of similar demographics without a CVAD. Characteristics used for matching included medical insurance type, prescription data availability, sex, age, geographic region, and years enrolled in the database.
Among 7,447 patients with at least one IVIg claim, 11.8% (n = 882) had CVAD exposure and 88.2% (n = 6,565) did not. Of those without a CVAD, 3,642 patients were matched to patients with CVAD. A quarter (25.4%) of patients with a CVAD had a thromboembolic event, compared with 11.2% of matched patients without CVADs (P less than .0001).
In the year leading up to IVIg therapy, 16.9% of those with a CVAD and 10.9% of matched patients without one had a previous thromboembolic event (P less than .0001). Patients with a CVAD also had significantly higher rates of hypertension (51.9% vs. 45.0% with placebo; P less than .001) and anticoagulation therapy (7.0% vs. 5.2% with placebo; P less than .05). Differences between the groups were not significant for diabetes (26.9% vs. 24.2%) and hyperlipidemia (19.1% vs. 17.8%).
Occlusion and stenosis of the carotid artery was the most common arterial thromboembolic outcome, occurring in 5.3% of those with a CVAD and in 2.8% of those without a CVAD. The most common venous thromboembolic event was acute venous embolism and thrombosis of lower-extremity deep vessels, which occurred in 7% of those with a CVAD and in 1.8% of those without.
The researchers also compared inpatient admissions and emergency department visits among those with and without a CVAD; both rates were higher in patients with a CVAD. Visits to the emergency department occurred at a rate of 0.14 events per month for those with a CVAD (2.01 distinct months with a claim) and 0.09 events per month for those without a CVAD (0.65 distinct months with a claim). Patients with a CVAD had 1.44 months with an inpatient admissions claim, in comparison with 0.41 months among matched patients without a CVAD. Inpatient admission frequency per month was 0.14 for those with a CVAD and 0.08 for those without.
The research was funded by CSL Behring. Dr. Patel and two of the other five authors are employees of CSL Behring.
SOURCE: Patel A et al. AANEM 2019, Abstract 94.
REPORTING FROM AANEM 2019
FDA approves rivaroxaban for VTE prevention in hospitalized, acutely ill patients
The Food and Drug Administration has approved rivaroxaban (Xarelto) for the prevention of venous thromboembolism (VTE) in hospitalized, acutely ill patients at risk for thromboembolic complications who do not have a high bleeding risk, according to a release from Janssen.
FDA approval for the new indication is based on results from the phase 3 MAGELLAN and MARINER trials, which included more than 20,000 hospitalized, acutely ill patients. In MAGELLAN, rivaroxaban demonstrated noninferiority to enoxaparin, a low-molecular-weight heparin, in short-term usage, and it was superior over the long term, compared with short-term enoxaparin followed by placebo.
While VTE and VTE-related deaths were not reduced in MARINER, compared with placebo, patients who received rivaroxaban did see a significantly reduction in symptomatic VTE with a favorable safety profile.
According to the indication, rivaroxaban can be administered to patients during hospitalization and can be continued after discharge for 31-39 days. The safety profile in MAGELLAN and MARINER was consistent with that already seen, with the most common adverse event being bleeding.
The new indication is the eighth for rivaroxaban, the most of any direct oral anticoagulant; six of these are specifically for the treatment, prevention, and reduction in the risk of VTE recurrence.
“With this new approval, Xarelto as an oral-only option now has the potential to change how acutely ill medical patients are managed for the prevention of blood clots, both in the hospital and for an extended period after discharge,” said Alex C. Spyropoulos, MD, of Northwell Health at Lenox Hill Hospital, New York, and a member of the steering committee of the MAGELLAN trial.
Find the full press release on the Janssen website.
The Food and Drug Administration has approved rivaroxaban (Xarelto) for the prevention of venous thromboembolism (VTE) in hospitalized, acutely ill patients at risk for thromboembolic complications who do not have a high bleeding risk, according to a release from Janssen.
FDA approval for the new indication is based on results from the phase 3 MAGELLAN and MARINER trials, which included more than 20,000 hospitalized, acutely ill patients. In MAGELLAN, rivaroxaban demonstrated noninferiority to enoxaparin, a low-molecular-weight heparin, in short-term usage, and it was superior over the long term, compared with short-term enoxaparin followed by placebo.
While VTE and VTE-related deaths were not reduced in MARINER, compared with placebo, patients who received rivaroxaban did see a significantly reduction in symptomatic VTE with a favorable safety profile.
According to the indication, rivaroxaban can be administered to patients during hospitalization and can be continued after discharge for 31-39 days. The safety profile in MAGELLAN and MARINER was consistent with that already seen, with the most common adverse event being bleeding.
The new indication is the eighth for rivaroxaban, the most of any direct oral anticoagulant; six of these are specifically for the treatment, prevention, and reduction in the risk of VTE recurrence.
“With this new approval, Xarelto as an oral-only option now has the potential to change how acutely ill medical patients are managed for the prevention of blood clots, both in the hospital and for an extended period after discharge,” said Alex C. Spyropoulos, MD, of Northwell Health at Lenox Hill Hospital, New York, and a member of the steering committee of the MAGELLAN trial.
Find the full press release on the Janssen website.
The Food and Drug Administration has approved rivaroxaban (Xarelto) for the prevention of venous thromboembolism (VTE) in hospitalized, acutely ill patients at risk for thromboembolic complications who do not have a high bleeding risk, according to a release from Janssen.
FDA approval for the new indication is based on results from the phase 3 MAGELLAN and MARINER trials, which included more than 20,000 hospitalized, acutely ill patients. In MAGELLAN, rivaroxaban demonstrated noninferiority to enoxaparin, a low-molecular-weight heparin, in short-term usage, and it was superior over the long term, compared with short-term enoxaparin followed by placebo.
While VTE and VTE-related deaths were not reduced in MARINER, compared with placebo, patients who received rivaroxaban did see a significantly reduction in symptomatic VTE with a favorable safety profile.
According to the indication, rivaroxaban can be administered to patients during hospitalization and can be continued after discharge for 31-39 days. The safety profile in MAGELLAN and MARINER was consistent with that already seen, with the most common adverse event being bleeding.
The new indication is the eighth for rivaroxaban, the most of any direct oral anticoagulant; six of these are specifically for the treatment, prevention, and reduction in the risk of VTE recurrence.
“With this new approval, Xarelto as an oral-only option now has the potential to change how acutely ill medical patients are managed for the prevention of blood clots, both in the hospital and for an extended period after discharge,” said Alex C. Spyropoulos, MD, of Northwell Health at Lenox Hill Hospital, New York, and a member of the steering committee of the MAGELLAN trial.
Find the full press release on the Janssen website.
Older IBD patients are most at risk of postdischarge VTE
Hospitalized patients with inflammatory bowel diseases (IBD) are most likely to be readmitted for venous thromboembolism (VTE) within 60 days of discharge, according to a new study that analyzed 5 years of U.S. readmissions data.
“Given increased thrombotic risk postdischarge, as well as overall safety of VTE prophylaxis, extending prophylaxis for those at highest risk may have significant benefits,” wrote Adam S. Faye, MD, of Columbia University, and coauthors. The study was published in Clinical Gastroenterology and Hepatology.
To determine which IBD patients would be most in need of postdischarge VTE prophylaxis, as well as when to administer it, the researchers analyzed 2010-2014 data from the Nationwide Readmissions Database (NRD). They found a total of 872,122 index admissions for IBD patients; 4% of those patients had a prior VTE. Of the index admissions, 1,160 led to a VTE readmission within 90 days. Readmitted patients had a relatively equal proportion of ulcerative colitis (n = 522) and Crohn’s disease (n = 638).
More than 90% of VTE readmissions occurred within 60 days of discharge; the risk was highest over the first 10 days and then decreased in each ensuing 10-day period until a slight increase at the 81- to 90-day period. All patients over age 30 had higher rates of readmission than those of patients under age 18, with the highest risk in patients between the ages of 66 and 80 years (risk ratio 4.04; 95% confidence interval, 2.54-6.44, P less than .01). Women were at lower risk (RR 0.82; 95% CI, 0.73-0.92, P less than .01). Higher risks of readmission were also associated with being on Medicare (RR 1.39; 95% CI, 1.23-1.58, P less than .01) compared with being on private insurance and being cared for at a large hospital (RR 1.26; 95% CI, 1.04-1.52, P = .02) compared with a small hospital.
The highest risk of VTE readmission was associated with a prior history of VTE (RR 2.89; 95% CI, 2.40-3.48, P less than .01), having two or more comorbidities (RR 2.57; 95% CI, 2.11-3.12, P less than .01) and having a Clostridioides difficile infection as of index admission (RR 1.90; 95% CI, 1.51-2.38, P less than .01). In addition, increased risk was associated with being discharged to a nursing or care facility (RR 1.85; 95% CI, 1.56-2.20, P less than .01) or home with health services (RR 2.05; 95% CI, 1.78-2.38, P less than .01) compared with a routine discharge.
In their multivariable analysis, similar factors such as a history of VTE (adjusted RR 2.41; 95% CI, 1.99-2.90, P less than .01), two or more comorbidities (aRR 1.78; 95% CI, 1.44-2.20, P less than .01) and C. difficile infection (aRR 1.47; 95% CI, 1.17-1.85, P less than.01) continued to be associated with higher risk of VTE readmission.
Though they emphasized that the use of NRD data offered the impressive ability to “review over 15 million discharges across the U.S. annually,” Dr. Faye and coauthors acknowledged that their study did have limitations. These included the inability to verify via chart review the study’s outcomes and covariates. In addition, they were unable to assess potential contributing risk factors such as medication use, use of VTE prophylaxis during hospitalization, disease severity, and family history. Finally, though unlikely, they admitted the possibility that patients could be counted more than once if they were readmitted with a VTE each year of the study.
The authors reported being supported by grants from the National Institutes of Health and various pharmaceutical companies, as well as receiving honoraria and serving as consultants.
SOURCE: Faye AS et al. Clin Gastroenterol Hepatol. 2019 July 20. doi: 10.1016/j.cgh.2019.07.028.
Hospitalized patients with inflammatory bowel diseases (IBD) are most likely to be readmitted for venous thromboembolism (VTE) within 60 days of discharge, according to a new study that analyzed 5 years of U.S. readmissions data.
“Given increased thrombotic risk postdischarge, as well as overall safety of VTE prophylaxis, extending prophylaxis for those at highest risk may have significant benefits,” wrote Adam S. Faye, MD, of Columbia University, and coauthors. The study was published in Clinical Gastroenterology and Hepatology.
To determine which IBD patients would be most in need of postdischarge VTE prophylaxis, as well as when to administer it, the researchers analyzed 2010-2014 data from the Nationwide Readmissions Database (NRD). They found a total of 872,122 index admissions for IBD patients; 4% of those patients had a prior VTE. Of the index admissions, 1,160 led to a VTE readmission within 90 days. Readmitted patients had a relatively equal proportion of ulcerative colitis (n = 522) and Crohn’s disease (n = 638).
More than 90% of VTE readmissions occurred within 60 days of discharge; the risk was highest over the first 10 days and then decreased in each ensuing 10-day period until a slight increase at the 81- to 90-day period. All patients over age 30 had higher rates of readmission than those of patients under age 18, with the highest risk in patients between the ages of 66 and 80 years (risk ratio 4.04; 95% confidence interval, 2.54-6.44, P less than .01). Women were at lower risk (RR 0.82; 95% CI, 0.73-0.92, P less than .01). Higher risks of readmission were also associated with being on Medicare (RR 1.39; 95% CI, 1.23-1.58, P less than .01) compared with being on private insurance and being cared for at a large hospital (RR 1.26; 95% CI, 1.04-1.52, P = .02) compared with a small hospital.
The highest risk of VTE readmission was associated with a prior history of VTE (RR 2.89; 95% CI, 2.40-3.48, P less than .01), having two or more comorbidities (RR 2.57; 95% CI, 2.11-3.12, P less than .01) and having a Clostridioides difficile infection as of index admission (RR 1.90; 95% CI, 1.51-2.38, P less than .01). In addition, increased risk was associated with being discharged to a nursing or care facility (RR 1.85; 95% CI, 1.56-2.20, P less than .01) or home with health services (RR 2.05; 95% CI, 1.78-2.38, P less than .01) compared with a routine discharge.
In their multivariable analysis, similar factors such as a history of VTE (adjusted RR 2.41; 95% CI, 1.99-2.90, P less than .01), two or more comorbidities (aRR 1.78; 95% CI, 1.44-2.20, P less than .01) and C. difficile infection (aRR 1.47; 95% CI, 1.17-1.85, P less than.01) continued to be associated with higher risk of VTE readmission.
Though they emphasized that the use of NRD data offered the impressive ability to “review over 15 million discharges across the U.S. annually,” Dr. Faye and coauthors acknowledged that their study did have limitations. These included the inability to verify via chart review the study’s outcomes and covariates. In addition, they were unable to assess potential contributing risk factors such as medication use, use of VTE prophylaxis during hospitalization, disease severity, and family history. Finally, though unlikely, they admitted the possibility that patients could be counted more than once if they were readmitted with a VTE each year of the study.
The authors reported being supported by grants from the National Institutes of Health and various pharmaceutical companies, as well as receiving honoraria and serving as consultants.
SOURCE: Faye AS et al. Clin Gastroenterol Hepatol. 2019 July 20. doi: 10.1016/j.cgh.2019.07.028.
Hospitalized patients with inflammatory bowel diseases (IBD) are most likely to be readmitted for venous thromboembolism (VTE) within 60 days of discharge, according to a new study that analyzed 5 years of U.S. readmissions data.
“Given increased thrombotic risk postdischarge, as well as overall safety of VTE prophylaxis, extending prophylaxis for those at highest risk may have significant benefits,” wrote Adam S. Faye, MD, of Columbia University, and coauthors. The study was published in Clinical Gastroenterology and Hepatology.
To determine which IBD patients would be most in need of postdischarge VTE prophylaxis, as well as when to administer it, the researchers analyzed 2010-2014 data from the Nationwide Readmissions Database (NRD). They found a total of 872,122 index admissions for IBD patients; 4% of those patients had a prior VTE. Of the index admissions, 1,160 led to a VTE readmission within 90 days. Readmitted patients had a relatively equal proportion of ulcerative colitis (n = 522) and Crohn’s disease (n = 638).
More than 90% of VTE readmissions occurred within 60 days of discharge; the risk was highest over the first 10 days and then decreased in each ensuing 10-day period until a slight increase at the 81- to 90-day period. All patients over age 30 had higher rates of readmission than those of patients under age 18, with the highest risk in patients between the ages of 66 and 80 years (risk ratio 4.04; 95% confidence interval, 2.54-6.44, P less than .01). Women were at lower risk (RR 0.82; 95% CI, 0.73-0.92, P less than .01). Higher risks of readmission were also associated with being on Medicare (RR 1.39; 95% CI, 1.23-1.58, P less than .01) compared with being on private insurance and being cared for at a large hospital (RR 1.26; 95% CI, 1.04-1.52, P = .02) compared with a small hospital.
The highest risk of VTE readmission was associated with a prior history of VTE (RR 2.89; 95% CI, 2.40-3.48, P less than .01), having two or more comorbidities (RR 2.57; 95% CI, 2.11-3.12, P less than .01) and having a Clostridioides difficile infection as of index admission (RR 1.90; 95% CI, 1.51-2.38, P less than .01). In addition, increased risk was associated with being discharged to a nursing or care facility (RR 1.85; 95% CI, 1.56-2.20, P less than .01) or home with health services (RR 2.05; 95% CI, 1.78-2.38, P less than .01) compared with a routine discharge.
In their multivariable analysis, similar factors such as a history of VTE (adjusted RR 2.41; 95% CI, 1.99-2.90, P less than .01), two or more comorbidities (aRR 1.78; 95% CI, 1.44-2.20, P less than .01) and C. difficile infection (aRR 1.47; 95% CI, 1.17-1.85, P less than.01) continued to be associated with higher risk of VTE readmission.
Though they emphasized that the use of NRD data offered the impressive ability to “review over 15 million discharges across the U.S. annually,” Dr. Faye and coauthors acknowledged that their study did have limitations. These included the inability to verify via chart review the study’s outcomes and covariates. In addition, they were unable to assess potential contributing risk factors such as medication use, use of VTE prophylaxis during hospitalization, disease severity, and family history. Finally, though unlikely, they admitted the possibility that patients could be counted more than once if they were readmitted with a VTE each year of the study.
The authors reported being supported by grants from the National Institutes of Health and various pharmaceutical companies, as well as receiving honoraria and serving as consultants.
SOURCE: Faye AS et al. Clin Gastroenterol Hepatol. 2019 July 20. doi: 10.1016/j.cgh.2019.07.028.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: Readmission for VTE in patients with inflammatory bowel diseases most often occurs within 60 days of discharge.
Major finding: The highest readmission risk was in patients between the ages of 66 and 80 (risk ratio 4.04; 95% confidence interval, 2.54-6.44, P less than .01).
Study details: A retrospective cohort study of 1,160 IBD patients who had VTE readmissions via 2010-2014 data from the Nationwide Readmissions Database.
Disclosures: The authors reported being supported by grants from the National Institutes of Health and various pharmaceutical companies, as well as receiving honoraria and serving as consultants.
Source: Faye AS et al. Clin Gastroenterol Hepatol. 2019 July 20. doi: 10.1016/j.cgh.2019.07.028.