User login
Review of Ethnoracial Representation in Clinical Trials (Phases 1 Through 4) of Atopic Dermatitis Therapies
To the Editor:
Atopic dermatitis (AD) affects an estimated 7.2% of adults and 10.7% of children in the United States; however, AD might affect different races at a varying rate.1 Compared to their European American counterparts, Asian/Pacific Islanders and African Americans are 7 and 3 times more likely, respectively, to be given a diagnosis of AD.2
Despite being disproportionately affected by AD, minority groups might be underrepresented in clinical trials of AD treatments.3 One explanation for this imbalance might be that ethnoracial representation differs across regions in the United States, perhaps in regions where clinical trials are conducted. Price et al3 investigated racial representation in clinical trials of AD globally and found that patients of color are consistently underrepresented.
Research on racial representation in clinical trials within the United States—on national and regional scales—is lacking from the current AD literature. We conducted a study to compare racial and ethnic disparities in AD clinical trials across regions of the United States.
Using the ClinicalTrials.gov database (www.clinicaltrials.gov) of the National Library of Medicine, we identified clinical trials of AD treatments (encompassing phases 1 through 4) in the United States that were completed before March 14, 2021, with the earliest data from 2013. Search terms included atopic dermatitis, with an advanced search for interventional (clinical trials) and with results.
In total, 95 completed clinical trials were identified, of which 26 (27.4%) reported ethnoracial demographic data. One trial was excluded due to misrepresentation regarding the classification of individuals who identified as more than 1 racial category. Clinical trials for systemic treatments (7 [28%]) and topical treatments (18 [72%]) were identified.
All ethnoracial data were self-reported by trial participants based on US Food and Drug Administration guidelines for racial and ethnic categorization.4 Trial participants who identified ethnically as Hispanic or Latino might have been a part of any racial group. Only 7 of the 25 included clinical trials (28%) provided ethnic demographic data (Hispanic [Latino] or non-Hispanic); 72% of trials failed to report ethnicity. Ethnic data included in our analysis came from only the 7 clinical trials that included these data. International multicenter trials that included a US site were excluded.
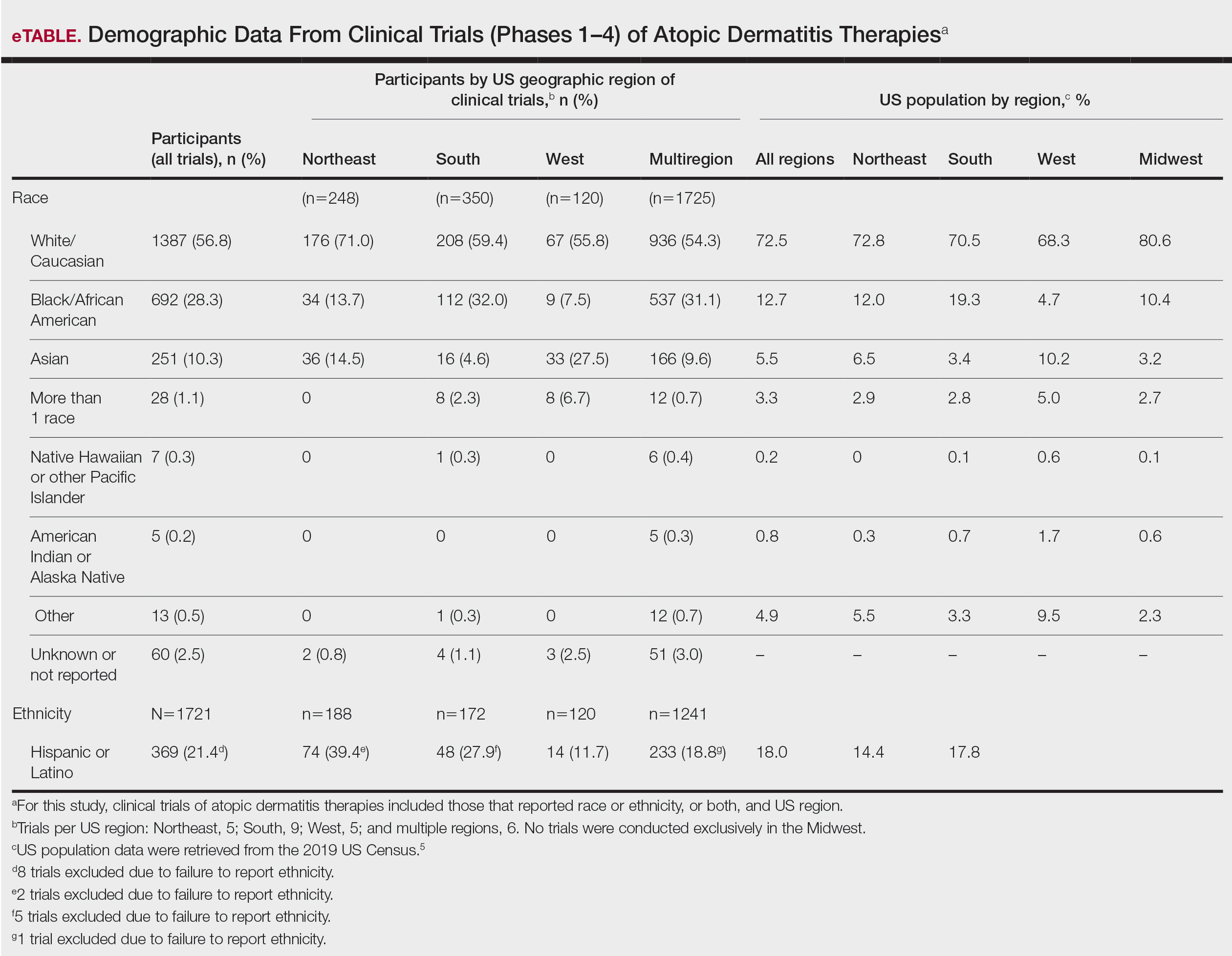
Ultimately, the number of trials included in our analysis was 25, comprised of 2443 participants. Data were further organized by US geographic region (Northeast, Midwest, South, West, and multiregion trials [ie, conducted in ≥2 regions]). No AD clinical trials were conducted solely in the Midwest; it was only included within multiregion trials.
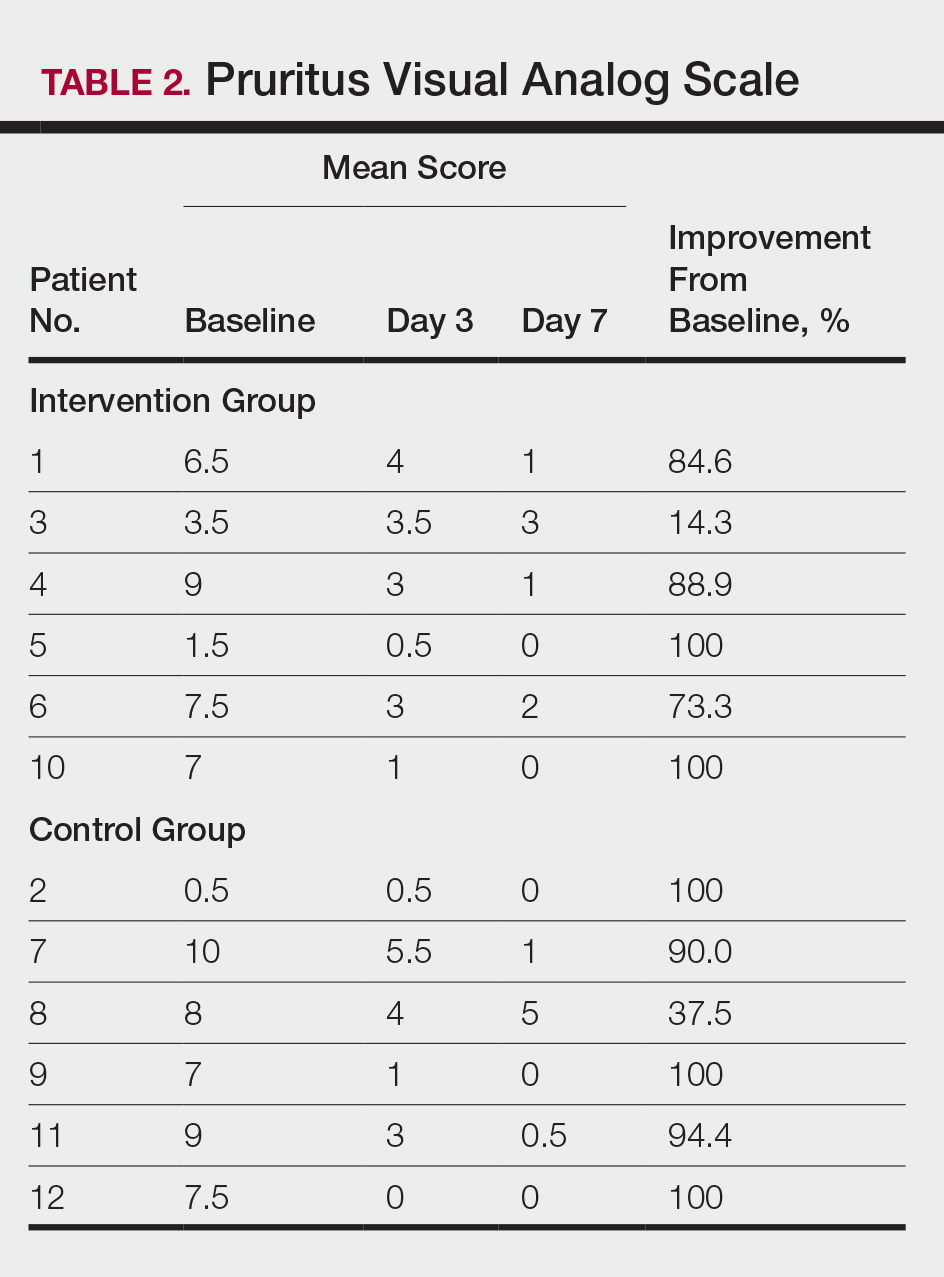
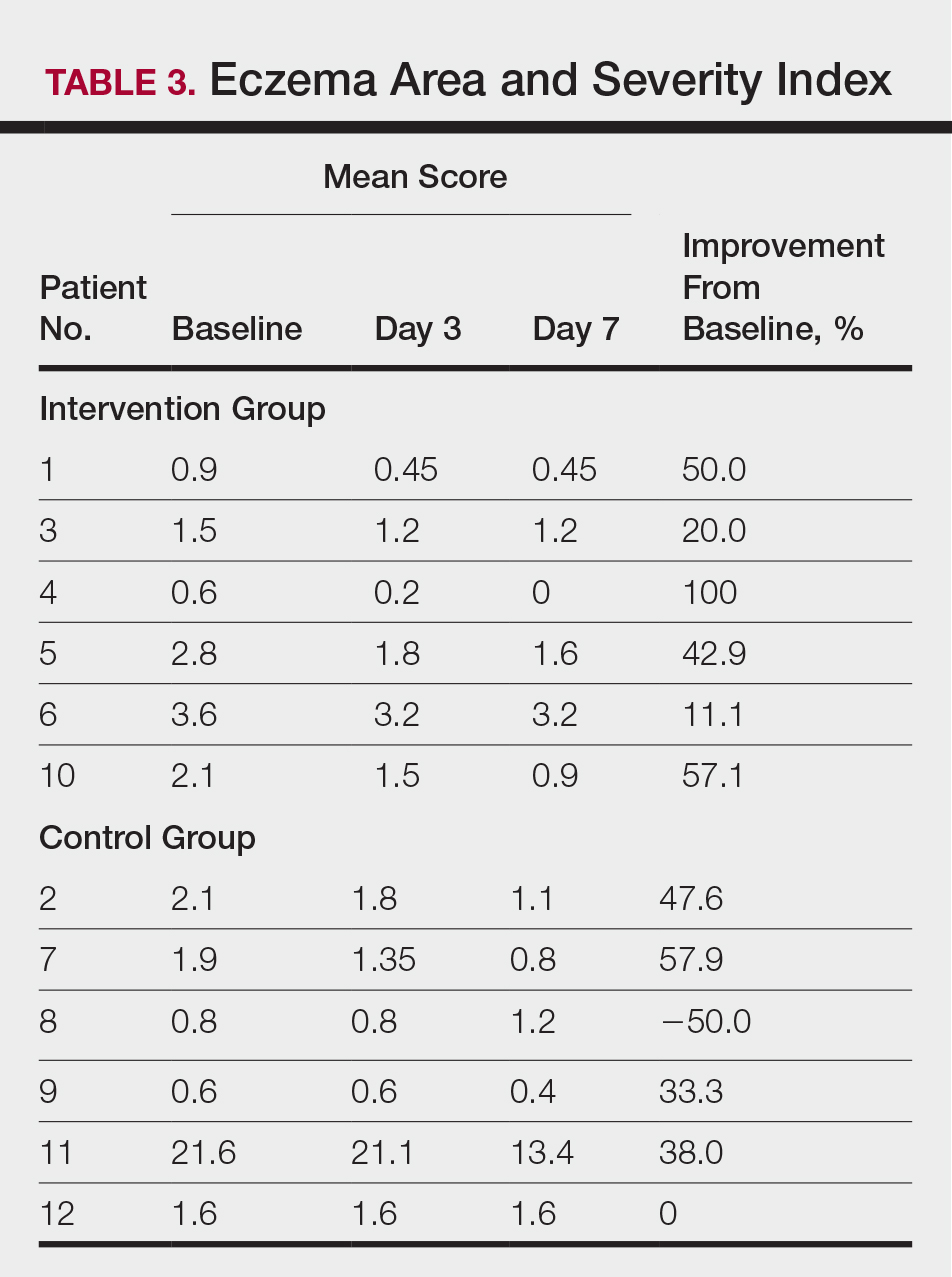
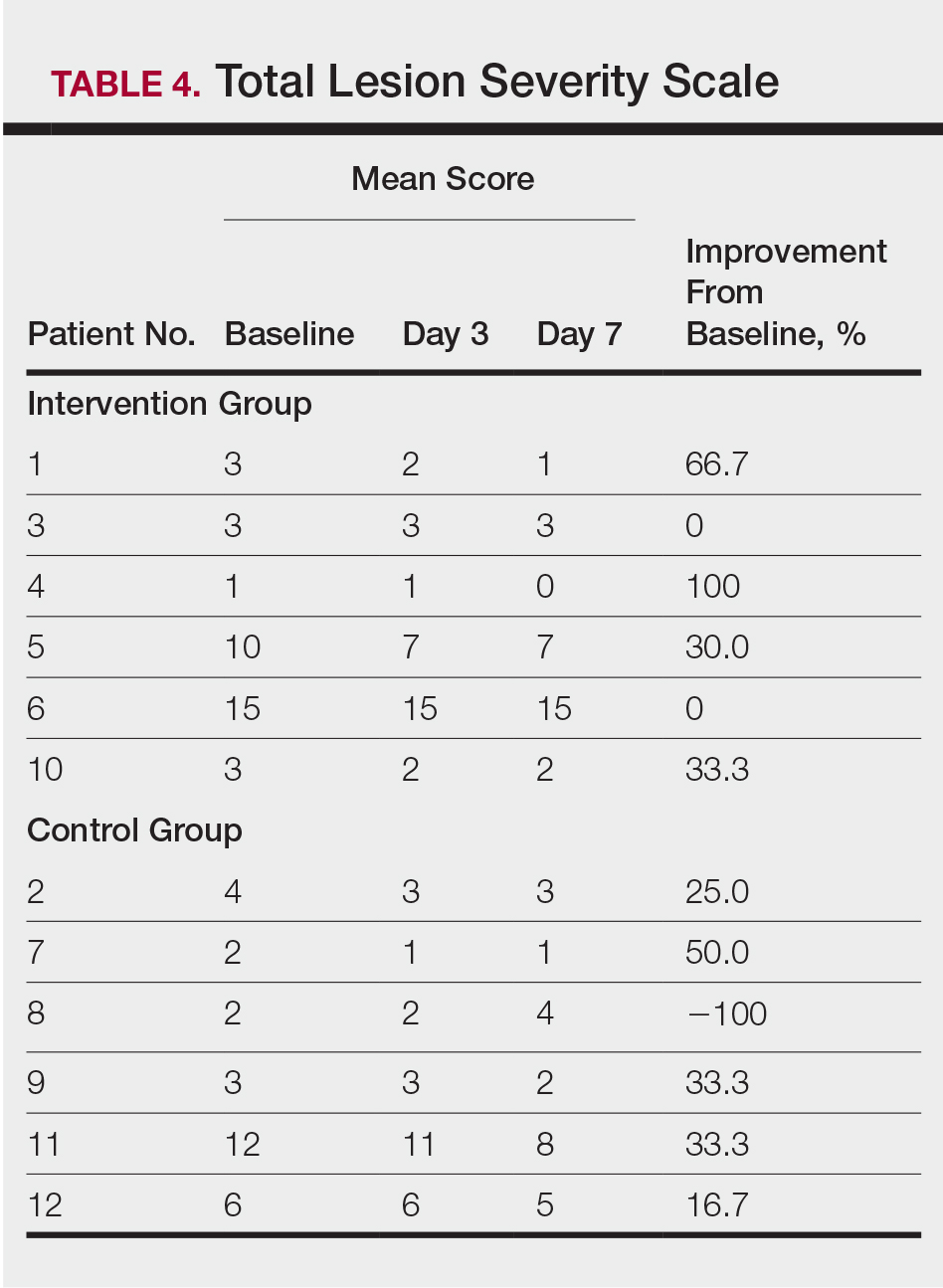
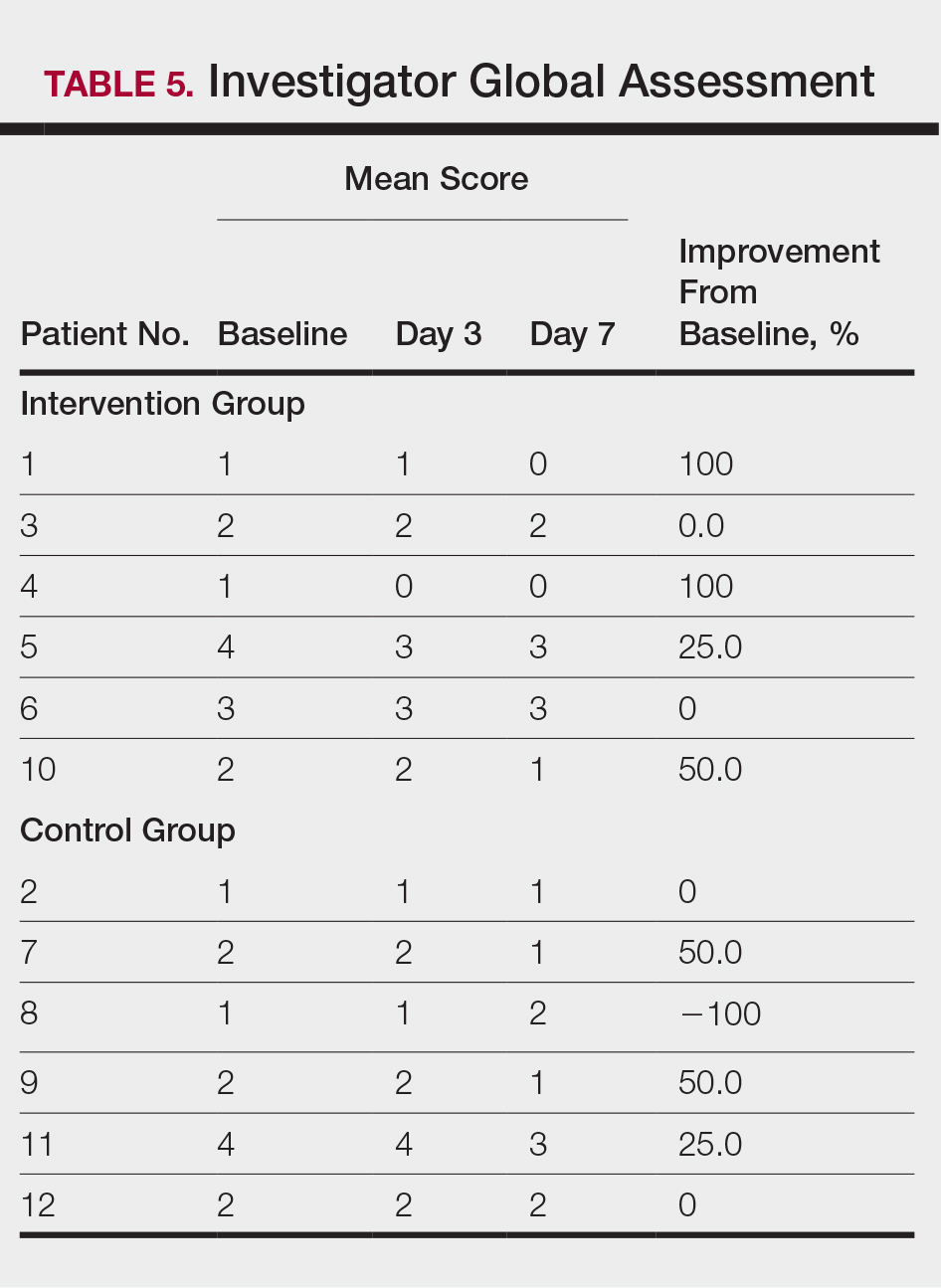
Compared to their representation in the 2019 US Census, most minority groups were overrepresented in clinical trials, while White individuals were underrepresented (eTable). The percentages of our findings on representation for race are as follows (US Census data are listed in parentheses for comparison5):
- White: 56.8% (72.5%)
- Black/African American: 28.3% (12.7%)
- Asian: 10.3% (5.5%)
- Multiracial: 1.1% (3.3%)
- Native Hawaiian or other Pacific Islander: 0.3% (0.2%)
- American Indian or Alaska Native: 0.2% (0.8%)
- Other: 0.5% (4.9%).
Our findings on representation for ethnicity are as follows (US Census data is listed in parentheses for comparison5):
- Hispanic or Latino: 21.4% (18.0%)
Although representation of Black/African American and Asian participants in clinical trials was higher than their representation in US Census data and representation of White participants was lower in clinical trials than their representation in census data, equal representation among all racial and ethnic groups is still lacking. A potential explanation for this finding might be that requirements for trial inclusion selected for more minority patients, given the propensity for greater severity of AD among those racial groups.2 Another explanation might be that efforts to include minority patients in clinical trials are improving.
There were great differences in ethnoracial representation in clinical trials when regions within the United States were compared. Based on census population data by region, the West had the highest percentage (29.9%) of Hispanic or Latino residents; however, this group represented only 11.7% of participants in AD clinical trials in that region.5
The South had the greatest number of participants in AD clinical trials of any region, which was consistent with research findings on an association between severity of AD and heat.6 With a warmer climate correlating with an increased incidence of AD, it is possible that more people are willing to participate in clinical trials in the South.
The Midwest was the only region in which region-specific clinical trials were not conducted. Recent studies have shown that individuals with AD who live in the Midwest have comparatively less access to health care associated with AD treatment and are more likely to visit an emergency department because of AD than individuals in any other US region.7 This discrepancy highlights the need for increased access to resources and clinical trials focused on the treatment of AD in the Midwest.
In 1993, the National Institutes of Health Revitalization Act established a federal legislative mandate to encourage inclusion of women and people of color in clinical trials.8 During the last 2 decades, there have been improvements in ethnoracial reporting. A 2020 global study found that 81.1% of randomized controlled trials (phases 2 and 3) of AD treatments reported ethnoracial data.3
Equal representation in clinical trials allows for further investigation of the connection between race, AD severity, and treatment efficacy. Clinical trials need to have equal representation of ethnoracial categories across all regions of the United States. If one group is notably overrepresented, ethnoracial associations related to the treatment of AD might go undetected.9 Similarly, if representation is unequal, relationships of treatment efficacy within ethnoracial groups also might go undetected. None of the clinical trials that we analyzed investigated treatment efficacy by race, suggesting that there is a need for future research in this area.
It also is important to note that broad classifications of race and ethnicity are limiting and therefore overlook differences within ethnoracial categories. Although representation of minority patients in clinical trials for AD treatments is improving, we conclude that there remains a need for greater and equal representation of minority groups in clinical trials of AD treatments in the United States.
- Avena-Woods C. Overview of atopic dermatitis. Am J Manag Care. 2017;23(8 suppl):S115-S123.
- Kaufman BP, Guttman‐Yassky E, Alexis AF. Atopic dermatitis in diverse racial and ethnic groups—variations in epidemiology, genetics, clinical presentation and treatment. Exp Dermatol. 2018;27:340-357. doi:10.1111/exd.13514
- Price KN, Krase JM, Loh TY, et al. Racial and ethnic disparities in global atopic dermatitis clinical trials. Br J Dermatol. 2020;183:378-380. doi:10.1111/bjd.18938
- Collection of race and ethnicity data in clinical trials: guidance for industry and Food and Drug Administration staff. US Food and Drug Administration; October 26, 2016. Accessed February 20, 2022. https://www.fda.gov/media/75453/download
- United States Census Bureau. 2019 Population estimates by age, sex, race and Hispanic origin. Published June 25, 2020. Accessed March 22, 2022. https://www.census.gov/newsroom/press-kits/2020/population-estimates-detailed.html
- Fleischer AB Jr. Atopic dermatitis: the relationship to temperature and seasonality in the United States. Int J Dermatol. 2019;58:465-471. doi:10.1111/ijd.14289
- Wu KK, Nguyen KB, Sandhu JK, et al. Does location matter? geographic variations in healthcare resource use for atopic dermatitis in the United States. J Dermatolog Treat. 2021;32:314-320. doi:10.1080/09546634.2019.1656796
- National Institutes of Health Revitalization Act of 1993, 42 USC 201 (1993). Accessed February 20, 2022. https://www.govinfo.gov/content/pkg/STATUTE-107/pdf/STATUTE-107-Pg122.pdf
- Hirano SA, Murray SB, Harvey VM. Reporting, representation, and subgroup analysis of race and ethnicity in published clinical trials of atopic dermatitis in the United States between 2000 and 2009. Pediatr Dermatol. 2012;29:749-755. doi:10.1111/j.1525-1470.2012.01797.x
To the Editor:
Atopic dermatitis (AD) affects an estimated 7.2% of adults and 10.7% of children in the United States; however, AD might affect different races at a varying rate.1 Compared to their European American counterparts, Asian/Pacific Islanders and African Americans are 7 and 3 times more likely, respectively, to be given a diagnosis of AD.2
Despite being disproportionately affected by AD, minority groups might be underrepresented in clinical trials of AD treatments.3 One explanation for this imbalance might be that ethnoracial representation differs across regions in the United States, perhaps in regions where clinical trials are conducted. Price et al3 investigated racial representation in clinical trials of AD globally and found that patients of color are consistently underrepresented.
Research on racial representation in clinical trials within the United States—on national and regional scales—is lacking from the current AD literature. We conducted a study to compare racial and ethnic disparities in AD clinical trials across regions of the United States.
Using the ClinicalTrials.gov database (www.clinicaltrials.gov) of the National Library of Medicine, we identified clinical trials of AD treatments (encompassing phases 1 through 4) in the United States that were completed before March 14, 2021, with the earliest data from 2013. Search terms included atopic dermatitis, with an advanced search for interventional (clinical trials) and with results.
In total, 95 completed clinical trials were identified, of which 26 (27.4%) reported ethnoracial demographic data. One trial was excluded due to misrepresentation regarding the classification of individuals who identified as more than 1 racial category. Clinical trials for systemic treatments (7 [28%]) and topical treatments (18 [72%]) were identified.
All ethnoracial data were self-reported by trial participants based on US Food and Drug Administration guidelines for racial and ethnic categorization.4 Trial participants who identified ethnically as Hispanic or Latino might have been a part of any racial group. Only 7 of the 25 included clinical trials (28%) provided ethnic demographic data (Hispanic [Latino] or non-Hispanic); 72% of trials failed to report ethnicity. Ethnic data included in our analysis came from only the 7 clinical trials that included these data. International multicenter trials that included a US site were excluded.
Ultimately, the number of trials included in our analysis was 25, comprised of 2443 participants. Data were further organized by US geographic region (Northeast, Midwest, South, West, and multiregion trials [ie, conducted in ≥2 regions]). No AD clinical trials were conducted solely in the Midwest; it was only included within multiregion trials.
Compared to their representation in the 2019 US Census, most minority groups were overrepresented in clinical trials, while White individuals were underrepresented (eTable). The percentages of our findings on representation for race are as follows (US Census data are listed in parentheses for comparison5):
- White: 56.8% (72.5%)
- Black/African American: 28.3% (12.7%)
- Asian: 10.3% (5.5%)
- Multiracial: 1.1% (3.3%)
- Native Hawaiian or other Pacific Islander: 0.3% (0.2%)
- American Indian or Alaska Native: 0.2% (0.8%)
- Other: 0.5% (4.9%).
Our findings on representation for ethnicity are as follows (US Census data is listed in parentheses for comparison5):
- Hispanic or Latino: 21.4% (18.0%)
Although representation of Black/African American and Asian participants in clinical trials was higher than their representation in US Census data and representation of White participants was lower in clinical trials than their representation in census data, equal representation among all racial and ethnic groups is still lacking. A potential explanation for this finding might be that requirements for trial inclusion selected for more minority patients, given the propensity for greater severity of AD among those racial groups.2 Another explanation might be that efforts to include minority patients in clinical trials are improving.
There were great differences in ethnoracial representation in clinical trials when regions within the United States were compared. Based on census population data by region, the West had the highest percentage (29.9%) of Hispanic or Latino residents; however, this group represented only 11.7% of participants in AD clinical trials in that region.5
The South had the greatest number of participants in AD clinical trials of any region, which was consistent with research findings on an association between severity of AD and heat.6 With a warmer climate correlating with an increased incidence of AD, it is possible that more people are willing to participate in clinical trials in the South.
The Midwest was the only region in which region-specific clinical trials were not conducted. Recent studies have shown that individuals with AD who live in the Midwest have comparatively less access to health care associated with AD treatment and are more likely to visit an emergency department because of AD than individuals in any other US region.7 This discrepancy highlights the need for increased access to resources and clinical trials focused on the treatment of AD in the Midwest.
In 1993, the National Institutes of Health Revitalization Act established a federal legislative mandate to encourage inclusion of women and people of color in clinical trials.8 During the last 2 decades, there have been improvements in ethnoracial reporting. A 2020 global study found that 81.1% of randomized controlled trials (phases 2 and 3) of AD treatments reported ethnoracial data.3
Equal representation in clinical trials allows for further investigation of the connection between race, AD severity, and treatment efficacy. Clinical trials need to have equal representation of ethnoracial categories across all regions of the United States. If one group is notably overrepresented, ethnoracial associations related to the treatment of AD might go undetected.9 Similarly, if representation is unequal, relationships of treatment efficacy within ethnoracial groups also might go undetected. None of the clinical trials that we analyzed investigated treatment efficacy by race, suggesting that there is a need for future research in this area.
It also is important to note that broad classifications of race and ethnicity are limiting and therefore overlook differences within ethnoracial categories. Although representation of minority patients in clinical trials for AD treatments is improving, we conclude that there remains a need for greater and equal representation of minority groups in clinical trials of AD treatments in the United States.
To the Editor:
Atopic dermatitis (AD) affects an estimated 7.2% of adults and 10.7% of children in the United States; however, AD might affect different races at a varying rate.1 Compared to their European American counterparts, Asian/Pacific Islanders and African Americans are 7 and 3 times more likely, respectively, to be given a diagnosis of AD.2
Despite being disproportionately affected by AD, minority groups might be underrepresented in clinical trials of AD treatments.3 One explanation for this imbalance might be that ethnoracial representation differs across regions in the United States, perhaps in regions where clinical trials are conducted. Price et al3 investigated racial representation in clinical trials of AD globally and found that patients of color are consistently underrepresented.
Research on racial representation in clinical trials within the United States—on national and regional scales—is lacking from the current AD literature. We conducted a study to compare racial and ethnic disparities in AD clinical trials across regions of the United States.
Using the ClinicalTrials.gov database (www.clinicaltrials.gov) of the National Library of Medicine, we identified clinical trials of AD treatments (encompassing phases 1 through 4) in the United States that were completed before March 14, 2021, with the earliest data from 2013. Search terms included atopic dermatitis, with an advanced search for interventional (clinical trials) and with results.
In total, 95 completed clinical trials were identified, of which 26 (27.4%) reported ethnoracial demographic data. One trial was excluded due to misrepresentation regarding the classification of individuals who identified as more than 1 racial category. Clinical trials for systemic treatments (7 [28%]) and topical treatments (18 [72%]) were identified.
All ethnoracial data were self-reported by trial participants based on US Food and Drug Administration guidelines for racial and ethnic categorization.4 Trial participants who identified ethnically as Hispanic or Latino might have been a part of any racial group. Only 7 of the 25 included clinical trials (28%) provided ethnic demographic data (Hispanic [Latino] or non-Hispanic); 72% of trials failed to report ethnicity. Ethnic data included in our analysis came from only the 7 clinical trials that included these data. International multicenter trials that included a US site were excluded.
Ultimately, the number of trials included in our analysis was 25, comprised of 2443 participants. Data were further organized by US geographic region (Northeast, Midwest, South, West, and multiregion trials [ie, conducted in ≥2 regions]). No AD clinical trials were conducted solely in the Midwest; it was only included within multiregion trials.
Compared to their representation in the 2019 US Census, most minority groups were overrepresented in clinical trials, while White individuals were underrepresented (eTable). The percentages of our findings on representation for race are as follows (US Census data are listed in parentheses for comparison5):
- White: 56.8% (72.5%)
- Black/African American: 28.3% (12.7%)
- Asian: 10.3% (5.5%)
- Multiracial: 1.1% (3.3%)
- Native Hawaiian or other Pacific Islander: 0.3% (0.2%)
- American Indian or Alaska Native: 0.2% (0.8%)
- Other: 0.5% (4.9%).
Our findings on representation for ethnicity are as follows (US Census data is listed in parentheses for comparison5):
- Hispanic or Latino: 21.4% (18.0%)
Although representation of Black/African American and Asian participants in clinical trials was higher than their representation in US Census data and representation of White participants was lower in clinical trials than their representation in census data, equal representation among all racial and ethnic groups is still lacking. A potential explanation for this finding might be that requirements for trial inclusion selected for more minority patients, given the propensity for greater severity of AD among those racial groups.2 Another explanation might be that efforts to include minority patients in clinical trials are improving.
There were great differences in ethnoracial representation in clinical trials when regions within the United States were compared. Based on census population data by region, the West had the highest percentage (29.9%) of Hispanic or Latino residents; however, this group represented only 11.7% of participants in AD clinical trials in that region.5
The South had the greatest number of participants in AD clinical trials of any region, which was consistent with research findings on an association between severity of AD and heat.6 With a warmer climate correlating with an increased incidence of AD, it is possible that more people are willing to participate in clinical trials in the South.
The Midwest was the only region in which region-specific clinical trials were not conducted. Recent studies have shown that individuals with AD who live in the Midwest have comparatively less access to health care associated with AD treatment and are more likely to visit an emergency department because of AD than individuals in any other US region.7 This discrepancy highlights the need for increased access to resources and clinical trials focused on the treatment of AD in the Midwest.
In 1993, the National Institutes of Health Revitalization Act established a federal legislative mandate to encourage inclusion of women and people of color in clinical trials.8 During the last 2 decades, there have been improvements in ethnoracial reporting. A 2020 global study found that 81.1% of randomized controlled trials (phases 2 and 3) of AD treatments reported ethnoracial data.3
Equal representation in clinical trials allows for further investigation of the connection between race, AD severity, and treatment efficacy. Clinical trials need to have equal representation of ethnoracial categories across all regions of the United States. If one group is notably overrepresented, ethnoracial associations related to the treatment of AD might go undetected.9 Similarly, if representation is unequal, relationships of treatment efficacy within ethnoracial groups also might go undetected. None of the clinical trials that we analyzed investigated treatment efficacy by race, suggesting that there is a need for future research in this area.
It also is important to note that broad classifications of race and ethnicity are limiting and therefore overlook differences within ethnoracial categories. Although representation of minority patients in clinical trials for AD treatments is improving, we conclude that there remains a need for greater and equal representation of minority groups in clinical trials of AD treatments in the United States.
- Avena-Woods C. Overview of atopic dermatitis. Am J Manag Care. 2017;23(8 suppl):S115-S123.
- Kaufman BP, Guttman‐Yassky E, Alexis AF. Atopic dermatitis in diverse racial and ethnic groups—variations in epidemiology, genetics, clinical presentation and treatment. Exp Dermatol. 2018;27:340-357. doi:10.1111/exd.13514
- Price KN, Krase JM, Loh TY, et al. Racial and ethnic disparities in global atopic dermatitis clinical trials. Br J Dermatol. 2020;183:378-380. doi:10.1111/bjd.18938
- Collection of race and ethnicity data in clinical trials: guidance for industry and Food and Drug Administration staff. US Food and Drug Administration; October 26, 2016. Accessed February 20, 2022. https://www.fda.gov/media/75453/download
- United States Census Bureau. 2019 Population estimates by age, sex, race and Hispanic origin. Published June 25, 2020. Accessed March 22, 2022. https://www.census.gov/newsroom/press-kits/2020/population-estimates-detailed.html
- Fleischer AB Jr. Atopic dermatitis: the relationship to temperature and seasonality in the United States. Int J Dermatol. 2019;58:465-471. doi:10.1111/ijd.14289
- Wu KK, Nguyen KB, Sandhu JK, et al. Does location matter? geographic variations in healthcare resource use for atopic dermatitis in the United States. J Dermatolog Treat. 2021;32:314-320. doi:10.1080/09546634.2019.1656796
- National Institutes of Health Revitalization Act of 1993, 42 USC 201 (1993). Accessed February 20, 2022. https://www.govinfo.gov/content/pkg/STATUTE-107/pdf/STATUTE-107-Pg122.pdf
- Hirano SA, Murray SB, Harvey VM. Reporting, representation, and subgroup analysis of race and ethnicity in published clinical trials of atopic dermatitis in the United States between 2000 and 2009. Pediatr Dermatol. 2012;29:749-755. doi:10.1111/j.1525-1470.2012.01797.x
- Avena-Woods C. Overview of atopic dermatitis. Am J Manag Care. 2017;23(8 suppl):S115-S123.
- Kaufman BP, Guttman‐Yassky E, Alexis AF. Atopic dermatitis in diverse racial and ethnic groups—variations in epidemiology, genetics, clinical presentation and treatment. Exp Dermatol. 2018;27:340-357. doi:10.1111/exd.13514
- Price KN, Krase JM, Loh TY, et al. Racial and ethnic disparities in global atopic dermatitis clinical trials. Br J Dermatol. 2020;183:378-380. doi:10.1111/bjd.18938
- Collection of race and ethnicity data in clinical trials: guidance for industry and Food and Drug Administration staff. US Food and Drug Administration; October 26, 2016. Accessed February 20, 2022. https://www.fda.gov/media/75453/download
- United States Census Bureau. 2019 Population estimates by age, sex, race and Hispanic origin. Published June 25, 2020. Accessed March 22, 2022. https://www.census.gov/newsroom/press-kits/2020/population-estimates-detailed.html
- Fleischer AB Jr. Atopic dermatitis: the relationship to temperature and seasonality in the United States. Int J Dermatol. 2019;58:465-471. doi:10.1111/ijd.14289
- Wu KK, Nguyen KB, Sandhu JK, et al. Does location matter? geographic variations in healthcare resource use for atopic dermatitis in the United States. J Dermatolog Treat. 2021;32:314-320. doi:10.1080/09546634.2019.1656796
- National Institutes of Health Revitalization Act of 1993, 42 USC 201 (1993). Accessed February 20, 2022. https://www.govinfo.gov/content/pkg/STATUTE-107/pdf/STATUTE-107-Pg122.pdf
- Hirano SA, Murray SB, Harvey VM. Reporting, representation, and subgroup analysis of race and ethnicity in published clinical trials of atopic dermatitis in the United States between 2000 and 2009. Pediatr Dermatol. 2012;29:749-755. doi:10.1111/j.1525-1470.2012.01797.x
Practice Points
- Although minority groups are disproportionally affected by atopic dermatitis (AD), they may be underrepresented in clinical trials for AD in the United States.
- Equal representation among ethnoracial groups in clinical trials is important to allow for a more thorough investigation of the efficacy of treatments for AD.
Multiethnic Training in Residency: A Survey of Dermatology Residents
Dermatologic treatment of patients with skin of color offers specific challenges. Studies have reported structural, morphologic, and physiologic distinctions among different ethnic groups,1 which may account for distinct clinical presentations of skin disease seen in patients with skin of color. Patients with skin of color are at increased risk for specific dermatologic conditions, such as postinflammatory hyperpigmentation, keloid development, and central centrifugal cicatricial alopecia.2,3 Furthermore, although skin cancer is less prevalent in patients with skin of color, it often presents at a more advanced stage and with a worse prognosis compared to white patients.4
Prior studies have demonstrated the need for increased exposure, education, and training in diseases pertaining to skin of color in US dermatology residency programs.6-8 The aim of this study was to assess if dermatologists in-training feel that their residency curriculum sufficiently educates them on the needs of patients with skin of color.
Methods
A 10-question anonymous survey was emailed to 109 dermatology residency programs to evaluate the attitudes of dermatology residents about their exposure to patients with skin of color and their skin-of-color curriculum. The study included individuals 18 years or older who were current residents in a dermatology program accredited by the Accreditation Council for Graduate Medical Education.
Results
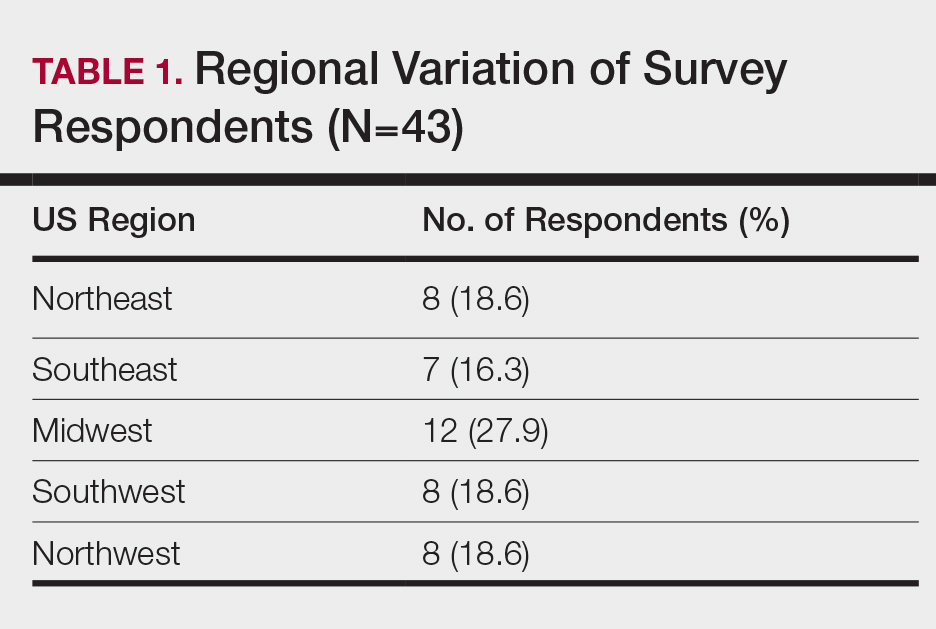
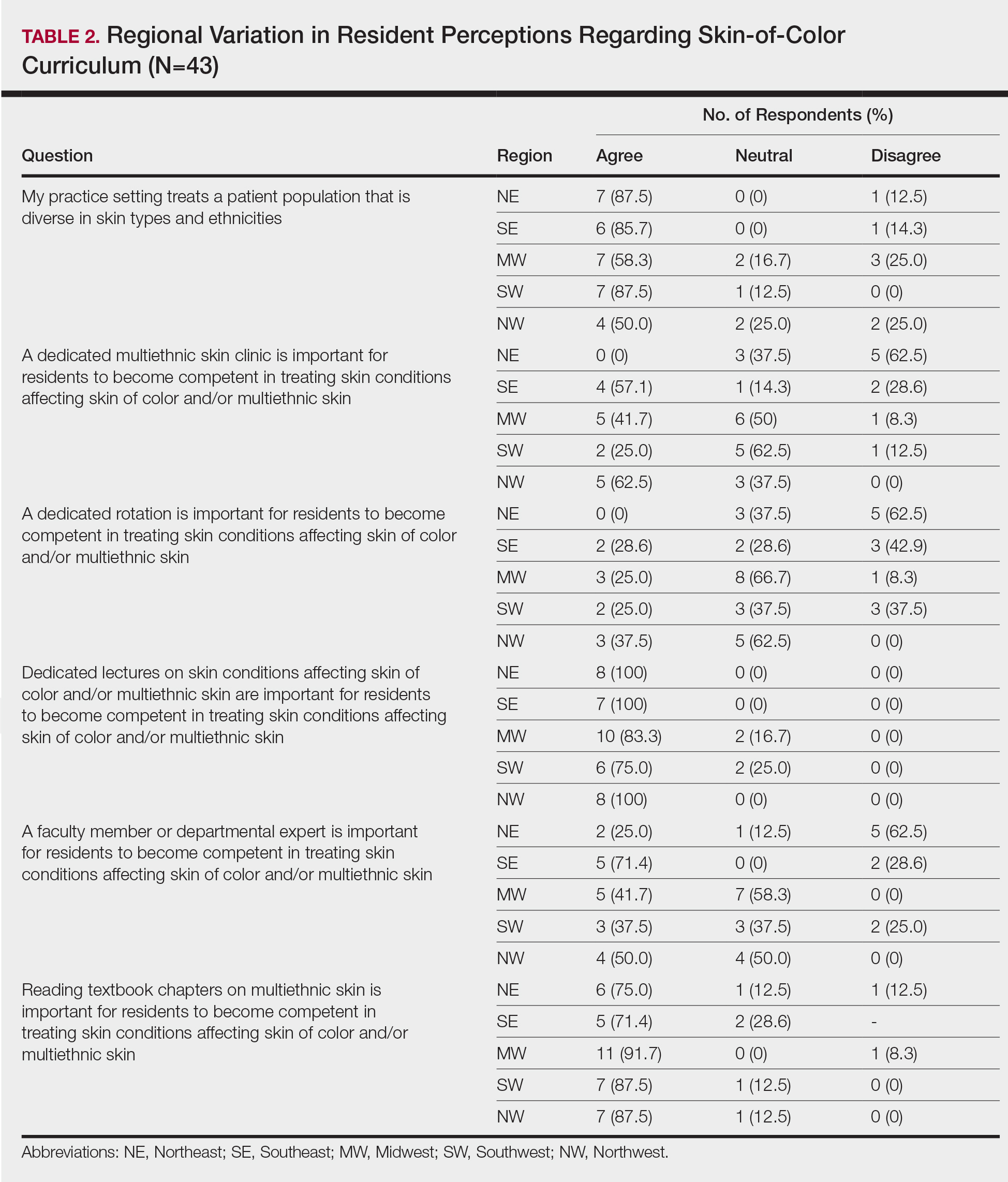
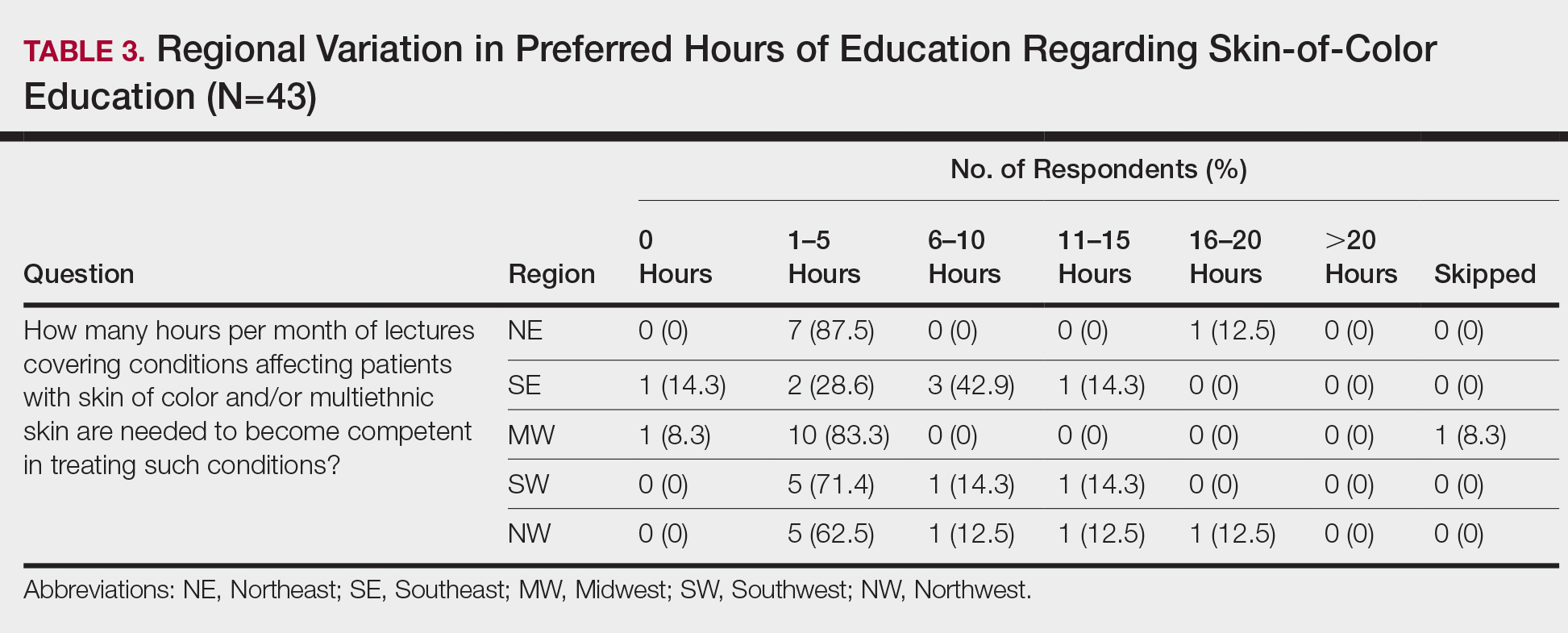
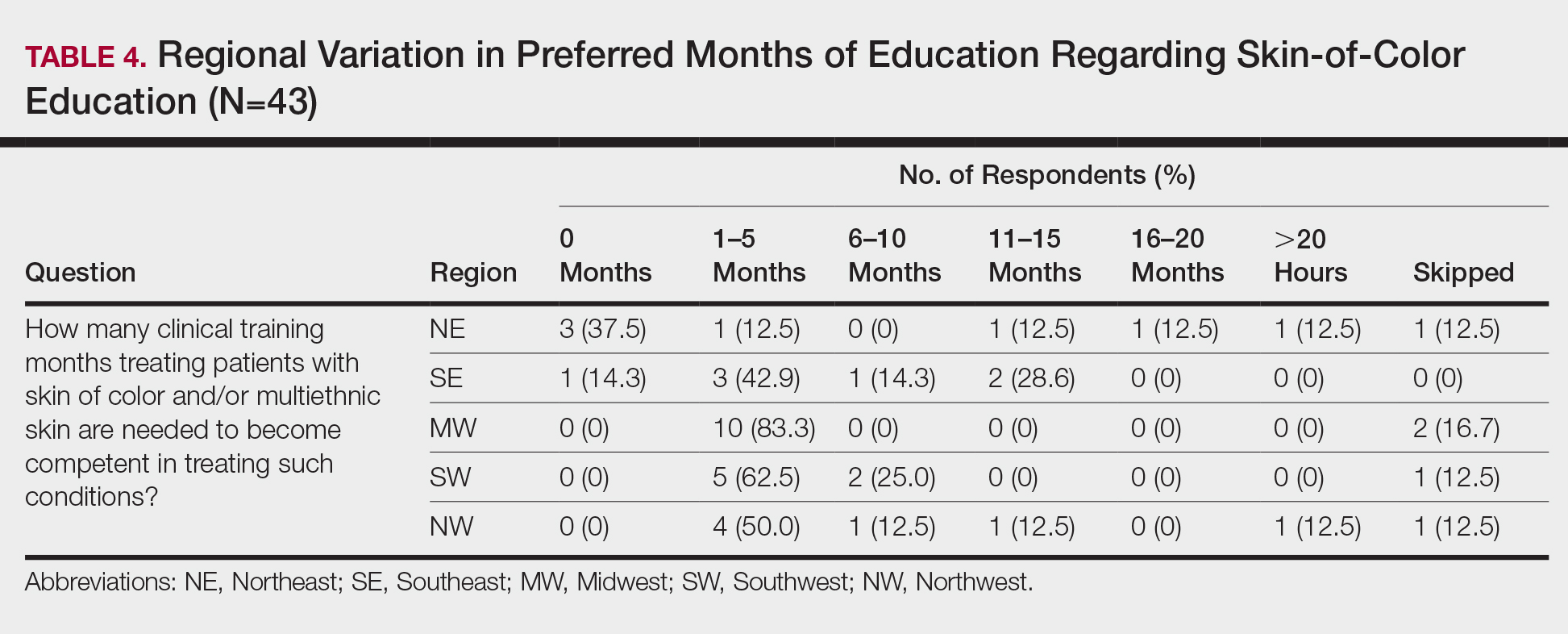
When asked the number of hours of lecture per month necessary to gain competence in conditions affecting patients with skin of color, 67% agreed that 1 to 5 hours was sufficient (Table 3). There were significant differences in the responses between the NE and SE (P=.024) and the SE and MW (P=.007). Of all respondents, 53% reported 1 to 5 months of clinical training are needed to gain competence in treating conditions affecting patients with skin of color, with significant differences in responses between the NE and MW (P<.001), the NE and SW (P=.019), and the SE and MW (P=.015)(Table 4).
Comment
Responses varied by practicing region
Although interactive lectures and textbook readings are important for obtaining a foundational understanding of dermatologic disease, they cannot substitute for clinical interactions and hands-on experience treating patients with skin of color.9 Not only do clinical interactions encourage independent reading and the study of encountered diagnoses, but intercommunication with patients may have a more profound and lasting impact on residents’ education.
Different regions of the United States have varying distributions of patients with skin of color, and dermatology residency program training reflects these disparities.6 In areas of less diversity, dermatology residents examine, diagnose, and treat substantially fewer patients with skin of color. The desire for more diverse training supports the prior findings of Nijhawan et al6 and is reflected in the responses we received in our study, whereby residents from the less ethnically diversified regions of the MW and NW were more likely to agree that clinics and rotations were necessary for training in preparation to sufficiently address the needs of patients with skin of color.
One way to compensate for the lack of ethnic diversity encountered in areas such as the MW and NW would be to develop educational programs featuring experts on skin of color.6 These specialists would not only train dermatology residents in areas of the country currently lacking ethnic diversity but also expand the expertise for treating patients with skin of color. Additionally, dedicated multiethnic skin clinics and externships devoted solely to treating patients with skin of color could be encouraged for residency training.6 Finally, community outreach through volunteer clinics may provide residents exposure to patients with skin of color seeking dermatologic care.10
This study was limited by the small number of respondents, but we were able to extract important trends and data from the collected responses. It is possible that respondents felt strongly about topics involving patients with skin of color, and the results were skewed to reflect individual bias. Additional limitations included not asking respondents for program names and population density (eg, urban, suburban, rural). Future studies should be directed toward analyzing how the diversity of the local population influences training in patients with skin of color, comparing program directors’ perceptions with residents’ perceptions on training in skin of color, and assessing patient perception of residents’ training in skin of color.
Conclusion
In the last decade it has become increasingly apparent that the US population is diversifying and that patients with skin of color will comprise a substantial proportion of the future population,8,11 which emphasizes the need for dermatology residency programs to ensure that residents receive adequate training and exposure to patients with skin of color as well as the distinct skin diseases seen more commonly in these populations.12
- Luther N, Darvin ME, Sterry W, et al. Ethnic differences in skin physiology, hair follicle morphology and follicular penetration. Skin Pharmacol Physiol. 2012;25:182-191.
- Shokeen D. Postinflammatory hyperpigmentation in patients with skin of color. Cutis. 2016;97:E9-E11.
- Lawson CN, Hollinger J, Sethi S, et al. Updates in the understanding and treatments of skin & hair disorders in women of color. Int J Women’s Dermatol. 2017;3:S21-S37.
- Hu S, Parmet Y, Allen G, et al. Disparity in melanoma: a trend analysis of melanoma incidence and stage at diagnosis among whites, Hispanics, and blacks in Florida. Arch Dermatol. 2009;145:1369-1374.
- Colby SL, Ortman JM; US Census Bureau. Projections of the Size and Composition of the U.S. Population: 2014 to 2060. Washington, DC: US Census Bureau; 2014. Current Population Reports, P25-1143. https://census.gov/content/dam/Census/library/publications/2015/demo/p25-1143.pdf. Published March 2015. Accessed May 13, 2020.
- Nijhawan RI, Jacob SE, Woolery-Lloyd H. Skin of color education in dermatology residency programs: does residency training reflect the changing demographics of the United States? J Am Acad Dermatol. 2008;59:615-618.
- Pritchett EN, Pandya AG, Ferguson NN, et al. Diversity in dermatology: roadmap for improvement. J Am Acad Dermatol. 2018;79:337-341.
- Pandya AG, Alexis AF, Berger TG, et al. Increasing racial and ethnic diversity in dermatology: a call to action. J Am Acad Dermatol. 2016;74:584-587.
- Ernst H, Colthorpe K. The efficacy of interactive lecturing for students with diverse science backgrounds. Adv Physiol Educ. 2007;31:41-44.
- Allday E. UCSF opens ‘skin of color’ dermatology clinic to address disparity in care. San Francisco Chronicle. March 20, 2019. https://www.sfchronicle.com/health/article/UCSF-opens-skin-of-color-dermatology-clinic-13704387.php. Accessed May 13, 2020.
- Van Voorhees AS, Enos CW. Diversity in dermatology residency programs. J Investig Dermatol Symp Proc. 2017;18:S46-S49.
- Enos CW, Harvey VM. From bench to bedside: the Hampton University Skin of Color Research Institute 2015 Skin of Color Symposium. J Investig Dermatol Symp Proc. 2017;18:S29-S30.
Dermatologic treatment of patients with skin of color offers specific challenges. Studies have reported structural, morphologic, and physiologic distinctions among different ethnic groups,1 which may account for distinct clinical presentations of skin disease seen in patients with skin of color. Patients with skin of color are at increased risk for specific dermatologic conditions, such as postinflammatory hyperpigmentation, keloid development, and central centrifugal cicatricial alopecia.2,3 Furthermore, although skin cancer is less prevalent in patients with skin of color, it often presents at a more advanced stage and with a worse prognosis compared to white patients.4
Prior studies have demonstrated the need for increased exposure, education, and training in diseases pertaining to skin of color in US dermatology residency programs.6-8 The aim of this study was to assess if dermatologists in-training feel that their residency curriculum sufficiently educates them on the needs of patients with skin of color.
Methods
A 10-question anonymous survey was emailed to 109 dermatology residency programs to evaluate the attitudes of dermatology residents about their exposure to patients with skin of color and their skin-of-color curriculum. The study included individuals 18 years or older who were current residents in a dermatology program accredited by the Accreditation Council for Graduate Medical Education.
Results


When asked the number of hours of lecture per month necessary to gain competence in conditions affecting patients with skin of color, 67% agreed that 1 to 5 hours was sufficient (Table 3). There were significant differences in the responses between the NE and SE (P=.024) and the SE and MW (P=.007). Of all respondents, 53% reported 1 to 5 months of clinical training are needed to gain competence in treating conditions affecting patients with skin of color, with significant differences in responses between the NE and MW (P<.001), the NE and SW (P=.019), and the SE and MW (P=.015)(Table 4).


Comment
Responses varied by practicing region
Although interactive lectures and textbook readings are important for obtaining a foundational understanding of dermatologic disease, they cannot substitute for clinical interactions and hands-on experience treating patients with skin of color.9 Not only do clinical interactions encourage independent reading and the study of encountered diagnoses, but intercommunication with patients may have a more profound and lasting impact on residents’ education.
Different regions of the United States have varying distributions of patients with skin of color, and dermatology residency program training reflects these disparities.6 In areas of less diversity, dermatology residents examine, diagnose, and treat substantially fewer patients with skin of color. The desire for more diverse training supports the prior findings of Nijhawan et al6 and is reflected in the responses we received in our study, whereby residents from the less ethnically diversified regions of the MW and NW were more likely to agree that clinics and rotations were necessary for training in preparation to sufficiently address the needs of patients with skin of color.
One way to compensate for the lack of ethnic diversity encountered in areas such as the MW and NW would be to develop educational programs featuring experts on skin of color.6 These specialists would not only train dermatology residents in areas of the country currently lacking ethnic diversity but also expand the expertise for treating patients with skin of color. Additionally, dedicated multiethnic skin clinics and externships devoted solely to treating patients with skin of color could be encouraged for residency training.6 Finally, community outreach through volunteer clinics may provide residents exposure to patients with skin of color seeking dermatologic care.10
This study was limited by the small number of respondents, but we were able to extract important trends and data from the collected responses. It is possible that respondents felt strongly about topics involving patients with skin of color, and the results were skewed to reflect individual bias. Additional limitations included not asking respondents for program names and population density (eg, urban, suburban, rural). Future studies should be directed toward analyzing how the diversity of the local population influences training in patients with skin of color, comparing program directors’ perceptions with residents’ perceptions on training in skin of color, and assessing patient perception of residents’ training in skin of color.
Conclusion
In the last decade it has become increasingly apparent that the US population is diversifying and that patients with skin of color will comprise a substantial proportion of the future population,8,11 which emphasizes the need for dermatology residency programs to ensure that residents receive adequate training and exposure to patients with skin of color as well as the distinct skin diseases seen more commonly in these populations.12
Dermatologic treatment of patients with skin of color offers specific challenges. Studies have reported structural, morphologic, and physiologic distinctions among different ethnic groups,1 which may account for distinct clinical presentations of skin disease seen in patients with skin of color. Patients with skin of color are at increased risk for specific dermatologic conditions, such as postinflammatory hyperpigmentation, keloid development, and central centrifugal cicatricial alopecia.2,3 Furthermore, although skin cancer is less prevalent in patients with skin of color, it often presents at a more advanced stage and with a worse prognosis compared to white patients.4
Prior studies have demonstrated the need for increased exposure, education, and training in diseases pertaining to skin of color in US dermatology residency programs.6-8 The aim of this study was to assess if dermatologists in-training feel that their residency curriculum sufficiently educates them on the needs of patients with skin of color.
Methods
A 10-question anonymous survey was emailed to 109 dermatology residency programs to evaluate the attitudes of dermatology residents about their exposure to patients with skin of color and their skin-of-color curriculum. The study included individuals 18 years or older who were current residents in a dermatology program accredited by the Accreditation Council for Graduate Medical Education.
Results


When asked the number of hours of lecture per month necessary to gain competence in conditions affecting patients with skin of color, 67% agreed that 1 to 5 hours was sufficient (Table 3). There were significant differences in the responses between the NE and SE (P=.024) and the SE and MW (P=.007). Of all respondents, 53% reported 1 to 5 months of clinical training are needed to gain competence in treating conditions affecting patients with skin of color, with significant differences in responses between the NE and MW (P<.001), the NE and SW (P=.019), and the SE and MW (P=.015)(Table 4).


Comment
Responses varied by practicing region
Although interactive lectures and textbook readings are important for obtaining a foundational understanding of dermatologic disease, they cannot substitute for clinical interactions and hands-on experience treating patients with skin of color.9 Not only do clinical interactions encourage independent reading and the study of encountered diagnoses, but intercommunication with patients may have a more profound and lasting impact on residents’ education.
Different regions of the United States have varying distributions of patients with skin of color, and dermatology residency program training reflects these disparities.6 In areas of less diversity, dermatology residents examine, diagnose, and treat substantially fewer patients with skin of color. The desire for more diverse training supports the prior findings of Nijhawan et al6 and is reflected in the responses we received in our study, whereby residents from the less ethnically diversified regions of the MW and NW were more likely to agree that clinics and rotations were necessary for training in preparation to sufficiently address the needs of patients with skin of color.
One way to compensate for the lack of ethnic diversity encountered in areas such as the MW and NW would be to develop educational programs featuring experts on skin of color.6 These specialists would not only train dermatology residents in areas of the country currently lacking ethnic diversity but also expand the expertise for treating patients with skin of color. Additionally, dedicated multiethnic skin clinics and externships devoted solely to treating patients with skin of color could be encouraged for residency training.6 Finally, community outreach through volunteer clinics may provide residents exposure to patients with skin of color seeking dermatologic care.10
This study was limited by the small number of respondents, but we were able to extract important trends and data from the collected responses. It is possible that respondents felt strongly about topics involving patients with skin of color, and the results were skewed to reflect individual bias. Additional limitations included not asking respondents for program names and population density (eg, urban, suburban, rural). Future studies should be directed toward analyzing how the diversity of the local population influences training in patients with skin of color, comparing program directors’ perceptions with residents’ perceptions on training in skin of color, and assessing patient perception of residents’ training in skin of color.
Conclusion
In the last decade it has become increasingly apparent that the US population is diversifying and that patients with skin of color will comprise a substantial proportion of the future population,8,11 which emphasizes the need for dermatology residency programs to ensure that residents receive adequate training and exposure to patients with skin of color as well as the distinct skin diseases seen more commonly in these populations.12
- Luther N, Darvin ME, Sterry W, et al. Ethnic differences in skin physiology, hair follicle morphology and follicular penetration. Skin Pharmacol Physiol. 2012;25:182-191.
- Shokeen D. Postinflammatory hyperpigmentation in patients with skin of color. Cutis. 2016;97:E9-E11.
- Lawson CN, Hollinger J, Sethi S, et al. Updates in the understanding and treatments of skin & hair disorders in women of color. Int J Women’s Dermatol. 2017;3:S21-S37.
- Hu S, Parmet Y, Allen G, et al. Disparity in melanoma: a trend analysis of melanoma incidence and stage at diagnosis among whites, Hispanics, and blacks in Florida. Arch Dermatol. 2009;145:1369-1374.
- Colby SL, Ortman JM; US Census Bureau. Projections of the Size and Composition of the U.S. Population: 2014 to 2060. Washington, DC: US Census Bureau; 2014. Current Population Reports, P25-1143. https://census.gov/content/dam/Census/library/publications/2015/demo/p25-1143.pdf. Published March 2015. Accessed May 13, 2020.
- Nijhawan RI, Jacob SE, Woolery-Lloyd H. Skin of color education in dermatology residency programs: does residency training reflect the changing demographics of the United States? J Am Acad Dermatol. 2008;59:615-618.
- Pritchett EN, Pandya AG, Ferguson NN, et al. Diversity in dermatology: roadmap for improvement. J Am Acad Dermatol. 2018;79:337-341.
- Pandya AG, Alexis AF, Berger TG, et al. Increasing racial and ethnic diversity in dermatology: a call to action. J Am Acad Dermatol. 2016;74:584-587.
- Ernst H, Colthorpe K. The efficacy of interactive lecturing for students with diverse science backgrounds. Adv Physiol Educ. 2007;31:41-44.
- Allday E. UCSF opens ‘skin of color’ dermatology clinic to address disparity in care. San Francisco Chronicle. March 20, 2019. https://www.sfchronicle.com/health/article/UCSF-opens-skin-of-color-dermatology-clinic-13704387.php. Accessed May 13, 2020.
- Van Voorhees AS, Enos CW. Diversity in dermatology residency programs. J Investig Dermatol Symp Proc. 2017;18:S46-S49.
- Enos CW, Harvey VM. From bench to bedside: the Hampton University Skin of Color Research Institute 2015 Skin of Color Symposium. J Investig Dermatol Symp Proc. 2017;18:S29-S30.
- Luther N, Darvin ME, Sterry W, et al. Ethnic differences in skin physiology, hair follicle morphology and follicular penetration. Skin Pharmacol Physiol. 2012;25:182-191.
- Shokeen D. Postinflammatory hyperpigmentation in patients with skin of color. Cutis. 2016;97:E9-E11.
- Lawson CN, Hollinger J, Sethi S, et al. Updates in the understanding and treatments of skin & hair disorders in women of color. Int J Women’s Dermatol. 2017;3:S21-S37.
- Hu S, Parmet Y, Allen G, et al. Disparity in melanoma: a trend analysis of melanoma incidence and stage at diagnosis among whites, Hispanics, and blacks in Florida. Arch Dermatol. 2009;145:1369-1374.
- Colby SL, Ortman JM; US Census Bureau. Projections of the Size and Composition of the U.S. Population: 2014 to 2060. Washington, DC: US Census Bureau; 2014. Current Population Reports, P25-1143. https://census.gov/content/dam/Census/library/publications/2015/demo/p25-1143.pdf. Published March 2015. Accessed May 13, 2020.
- Nijhawan RI, Jacob SE, Woolery-Lloyd H. Skin of color education in dermatology residency programs: does residency training reflect the changing demographics of the United States? J Am Acad Dermatol. 2008;59:615-618.
- Pritchett EN, Pandya AG, Ferguson NN, et al. Diversity in dermatology: roadmap for improvement. J Am Acad Dermatol. 2018;79:337-341.
- Pandya AG, Alexis AF, Berger TG, et al. Increasing racial and ethnic diversity in dermatology: a call to action. J Am Acad Dermatol. 2016;74:584-587.
- Ernst H, Colthorpe K. The efficacy of interactive lecturing for students with diverse science backgrounds. Adv Physiol Educ. 2007;31:41-44.
- Allday E. UCSF opens ‘skin of color’ dermatology clinic to address disparity in care. San Francisco Chronicle. March 20, 2019. https://www.sfchronicle.com/health/article/UCSF-opens-skin-of-color-dermatology-clinic-13704387.php. Accessed May 13, 2020.
- Van Voorhees AS, Enos CW. Diversity in dermatology residency programs. J Investig Dermatol Symp Proc. 2017;18:S46-S49.
- Enos CW, Harvey VM. From bench to bedside: the Hampton University Skin of Color Research Institute 2015 Skin of Color Symposium. J Investig Dermatol Symp Proc. 2017;18:S29-S30.
Practice Points
- To treat the ever-changing demographics of patients in the United States, dermatologists must receive adequate exposure and education regarding dermatologic conditions in patients from various ethnic backgrounds.
- Dermatology residents from less diverse regions are more likely to agree that dedicated clinics and rotations are important to gain competence compared to those from more diverse regions.
- In areas with less diversity, dedicated multiethnic skin clinics and faculty may be more important for assuring an adequate residency experience.
Psychosocial Impact of Psoriasis: A Review for Dermatology Residents
The psychosocial impact of psoriasis is a critical component of disease burden. Psoriatic patients have high rates of depression and anxiety, problems at work, and difficulties with interpersonal relationships and intimacy.1 A National Psoriasis Foundation (NPF) survey from 2003 to 2011 reported that psoriasis affects overall emotional well-being in 88% of patients and enjoyment of life in 82% of patients.2
The reasons for psychosocial burden stem from public misconceptions and disease stigma. A survey of 1005 individuals (age range, 16–64 years) about their perceptions of psoriasis revealed that 16.5% believed that psoriasis is contagious and 6.8% believed that psoriasis is related to personal hygiene.3 Fifty percent practiced discriminatory behavior toward psoriatic patients, including reluctance to shake hands (28.8%) and engage in sexual relations/intercourse (44.1%). Sixty-five percent of psoriatic patients felt their appearance is unsightly, and 73% felt self-conscious about having psoriasis.2
The psychosocial burden exists despite medical treatment of the disease. In a cross-sectional study of 1184 psoriatic patients, 70.2% had impaired quality of life (QOL) as measured by the dermatology life quality index (DLQI), even after receiving a 4-week treatment for psoriasis.4 Medical treatment of psoriasis is not enough; providers need to assess overall QOL and provide treatment and resources for these patients in addition to symptomatic management.
There have been many studies on the psychosocial burden of psoriasis, but few have focused on a dermatology resident’s role in addressing this issue. This article will review psychosocial domains—psychiatric comorbidities and social functioning including occupational functioning, interpersonal relationships, and sexual functioning— and discuss a dermatology resident’s role in assessing and addressing each of these areas.
Methods
A PubMed search of articles indexed for MEDLINE was conducted using the following terms: psoriasis, depression, anxiety, work productivity, sexual functioning, and interpersonal relationships. Selected articles covered prevalence, assessment, and management of each psychosocial domain.
Results
Psychiatric Comorbidities
Prevalence
A high prevalence of psychiatric comorbidities exists in psoriatic patients. In a study of 469,097 patients with psoriasis, depression was the third most prevalent comorbidity (17.91%), following hyperlipidemia (45.64%) and hypertension (42.19%).5 In a 10-year longitudinal, population-based, prospective cohort study, antidepressant prescriptions were twice as frequent in psoriatic patients (17.8%) compared to control (7.9%)(P<.001).6 In a meta-analysis of 98 studies investigating psoriatic patients and psychiatric comorbidities, patients with psoriasis were 1.5 times more likely to experience depression (odds ratio [OR]: 1.57; 95% CI, 1.40-1.76) and use antidepressants (OR: 4.24; 95% CI, 1.53-11.76) compared to control.7 Patients with psoriasis were more likely to attempt suicide (OR: 1.32; 95% CI, 1.14-1.54) and complete suicide (OR: 1.20; 95% CI, 1.04-1.39) compared to people without psoriasis.8 A 1-year cross-sectional study of 90 psoriatic patients reported 78.7% were diagnosed with depression and 76.7% were diagnosed with anxiety. Seventy-two percent reported both anxiety and depression, correlating with worse QOL (χ2=26.7; P<.05).9
Assessment
Psychiatric comorbidities are assessed using clinical judgment and formal screening questionnaires in research studies. Signs of depression in patients with psoriasis can manifest as poor treatment adherence and recurrent flares of psoriasis.10,11 Psoriatic patients with psychiatric comorbidities were less likely to be adherent to treatment (risk ratio: 0.35; P<.003).10 The patient health questionnaire (PHQ) 9 and generalized anxiety disorder scale (GAD) 7 are validated and reliable questionnaires. The first 2 questions in PHQ-9 and GAD-7 screen for depression and anxiety, respectively.12-14 These 2-question screens are practical in a fast-paced dermatology outpatient setting. Systematic questionnaires specifically targeting mood disorders may be more beneficial than the widely used DLQI, which may not adequately capture mood disorders. Over the course of 10 months, 607 patients with psoriasis were asked to fill out the PHQ-9, GAD-7, and DLQI. Thirty-eight percent of patients with major depressive disorder had a DLQI score lower than 10, while 46% of patients with generalized anxiety disorder had a DLQI score lower than 10.15 Other questionnaires, including the hospital anxiety and depression scale and Beck depression inventory, are valid instruments with high sensitivity but are commonly used for research purposes and may not be clinically feasible.16
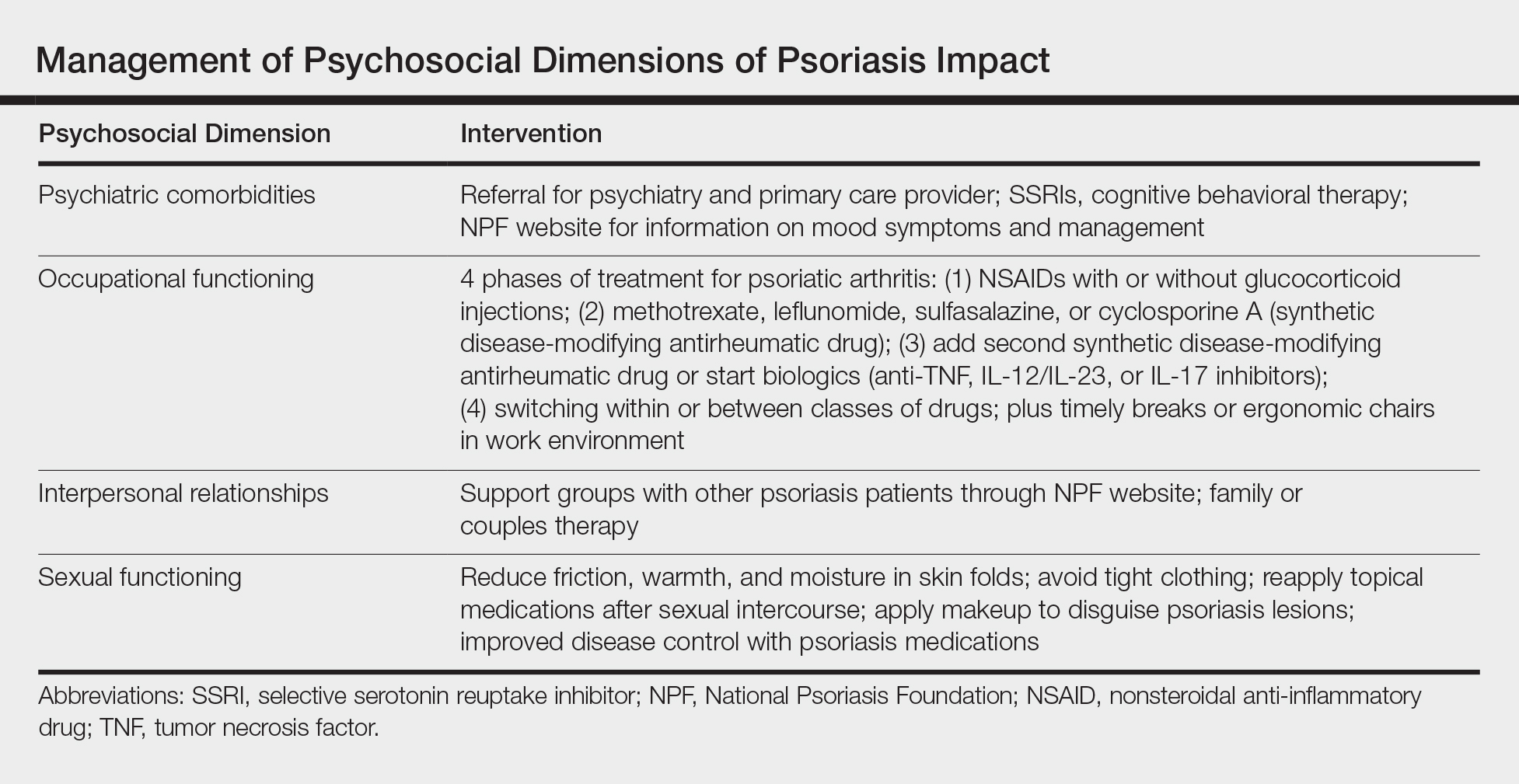
Management
Dermatologists should refer patients with depression and/or anxiety to psychiatry. Interventions include pharmacologic and nonpharmacologic management. First-line therapy for depression and anxiety is a combination of selective serotonin reuptake inhibitors and cognitive behavioral therapy.17 In addition, providers can direct patients to online resources such as the NPF website, where patients with psoriasis can access information about the signs and symptoms of mood disorders and contact the patient navigation center for further help.18
Social Functioning
Occupational Prevalence
The NPF found that 92% of patients with psoriasis or psoriatic arthritis (PsA) surveyed between 2003 and 2011 cited their psoriasis as reason for unemployment.2 In a survey of 43 patients asked about social and occupational functioning using the social and occupational assessment scale, 62.5% of psoriatic patients reported distress at work and 51.1% reported decreased efficiency at work.19 A national online survey that was conducted in France and issued to patients with and without psoriasis assessed overall QOL and work productivity using the work productivity and activity impairment questionnaire for psoriasis (WPAI-PSO). Of 714 patients with psoriasis and PsA, the latter had a 57.6% decrease in work productivity over 7 days compared to 27.9% in controls (P<.05).20 Occupational impairment leads to lost wages and hinders advancement, further exacerbating the psychosocial burden of psoriasis.21
Occupational Assessment
Formal assessment of occupational function can be done with the WPAI-PSO, a 6-question valid instrument.22 Providers may look for risk factors associated with greater loss in work productivity to help identify and offer support for patients. Patients with increased severity of itching, pain, and scaling experienced a greater decrease in work productivity.21,23 Patients with PsA warrant early detection and treatment because they experience greater physical restraints that can interfere with work activities. Of the 459 psoriatic patients without a prior diagnosis of PsA who filled out the PsA screening and evaluation questionnaire, 144 (31.4%) received a score of 44 or higher and were referred to rheumatology for further evaluation with the classification criteria for PsA. Nine percent of patients failed to be screened and remained undiagnosed with PsA.24 In a study using the health assessment questionnaire to assess 400 patients with PsA, those with worse physical function due to joint pain and stiffness were less likely to remain employed (OR: 0.56; P=.02).25
Occupational Management
Identifying and coordinating symptoms of PsA between dermatology and rheumatology is beneficial for patients who experience debilitating symptoms. There are a variety of treatments available for PsA. According to the European League Against Rheumatism 2015 guidelines developed from expert opinion and systematic reviews for PsA management, there are 4 phases of treatment, with reassessment every 3 to 6 months for effectiveness of therapy.26,27 Phase I involves initiating nonsteroidal anti-inflammatory drugs with or without glucocorticoid injections. Phase II involves synthetic disease-modifying drugs, including methotrexate, leflunomide, sulfasalazine, or cyclosporine. Phase III involves adding a second synthetic disease-modifying drug or starting a biologic, such as an anti–tumor necrosis factor, IL-12/IL-23, or IL-17 inhibitor. Phase IV involves switching to a different drug in either aforementioned class.26,27 Treatment with biologics improves work productivity as assessed by WPAI-PSO for psoriasis and PsA.28-30 Encouraging patients to speak up in the workplace and request small accommodations such as timely breaks or ergonomic chairs can help patients feel more comfortable and supported in the work environment.18 Patients who felt supported at work were more likely to remain employed.25
Interpersonal Relationships Prevalence
Misinformation about psoriasis, fear of rejection, and feelings of isolation may contribute to interpersonal conflict. Patients have feelings of shame and self-consciousness that hinder them from engaging in social activities and seeking out relationships.31 Twenty-nine percent of patients feel that psoriasis has interfered with establishing relationships because of negative self-esteem associated with the disease,32 and 26.3% have experienced people avoiding physical contact.33 Family and spouses of patients with psoriasis may be secondarily affected due to economic and emotional distress. Ninety-eight percent of family members of psoriatic patients experienced emotional distress and 54% experienced the burden of care.34 In a survey of 63 relatives and partners of patients with psoriasis, 57% experienced psychological distress, including anxiety and worry over a psoriatic patient’s future.35
Interpersonal Relationships Assessment
Current available tools, including the DLQI and short form health survey, measure overall QOL, including social functioning, but may not be practical in a clinic setting. Although no quick-screening test to assess for this domain exists, providers are encouraged to ask patients about disease impact on interpersonal relationships. The family DLQI questionnaire, adapted from the DLQI, may help physicians and social workers evaluate the burden on a patient’s family members.34
Interpersonal Relationships Management
It may be difficult for providers to address problems with interpersonal relationships without accessible tools. Patients may not be accompanied by family or friends during appointments, and it is difficult to screen for these issues during visits. Providers may offer resources such as the NPF website, which provides information about support groups. It also provides tips on dating and connecting to others in the community who share similar experiences.18 Encouraging patients to seek family or couples therapy also may be beneficial. Increased social support can lead to better QOL and fewer depressive symptoms.36
Sexual Functioning Prevalence
Psoriasis affects both physical and psychological components of sexual function. Among 3485 patients with skin conditions who were surveyed about sexual function, 34% of psoriatic patients reported that psoriasis interfered with sexual functioning at least to a certain degree.37 Sexual impairment was strongly associated with depression, anxiety, and suicidal ideation; 24% of depressed patients and 20% of anxious patients experienced sexual problems a lot or very much, based on the DLQI.37 Depending on the questionnaire used, the prevalence of sexual dysfunction due to psoriasis ranged from 35.5% to 71.3%.38 In an observational cohort study of 158 participants (n=79 psoriasis patients and n=79 controls), 34.2% of patients with psoriasis experienced erectile dysfunction compared to 17.7% of controls.39 Forty-two percent of psoriatic patients with genital involvement reported dyspareunia, 32% reported worsening of genital psoriasis after intercourse, and 43% reported decreased frequency of intercourse.40
Sexual Functioning Assessment
The Skindex-29, DLQI, and psoriasis disability index are available QOL tools that include one question evaluating difficulties with sexual function. The
Sexual Functioning Management
Better disease control leads to improved sexual function, as patients experience fewer feelings of shame, anxiety, and depression, as well as improvement of physical symptoms that can interfere with sexual functioning.38,43,44 Reducing friction, warmth, and moisture, as well as avoiding tight clothing, can help those with genital psoriasis. Patients are advised to reapply topical medications after sexual intercourse. Patients also can apply makeup to disguise psoriasis and help reduce feelings of self-consciousness that can impede sexual intimacy.18
Comment
The psychosocial burden of psoriasis penetrates many facets of patient lives. Psoriasis can invoke feelings of shame and embarrassment that are worsened by the public’s misconceptions about psoriasis, resulting in serious mental health issues that can cause even greater disability. Depression and anxiety are prevalent in patients with psoriasis. The characteristic symptoms of pain and pruritus along with psychiatric comorbidities can have an underestimated impact on daily activities, including employment, interpersonal relationships, and sexual function. Such dysfunctions have serious implications toward wages, professional advancement, social support, and overall QOL.
Dermatology providers play an important role in screening for these problems through validated questionnaires and identifying risks. Simple screening questions such as the PHQ-9 can be beneficial and feasible during dermatology visits. Screening for PsA can help patients avoid problems at work. Sexual dysfunction is a sensitive topic; however, providers can use a 1-question screen from valid questionnaires and inquire about the location of lesions as opportunities to address this issue.
Interventions lead to better disease control, which concurrently improves overall QOL. These interventions depend on both patient adherence and a physician’s commitment to finding an optimal treatment regimen for each individual. Medical management; coordinating care; developing treatment plans with psychiatry, rheumatology, and primary care providers; and psychological counseling and services may be necessary and beneficial (Table). Offering accessible resources such as the NPF website helps patients access information outside the clinic when it is not feasible to address all these concerns in a single visit. Psoriasis requires more than just medical management; it requires dermatology providers to use a multidisciplinary approach to address the psychosocial aspects of the disease.
Conclusion
The psychosocial burden of psoriasis is immense. Stigma, public misconception, mental health concerns, and occupational and interpersonal difficulty are the basis of disease burden. Providers play a vital role in assessing the effect psoriasis has on different areas of patients’ lives and providing appropriate interventions and resources to reduce disease burden.
- Kimball AB, Jacobson C, Weiss S, et al. The psychosocial burden of psoriasis. Am J Clin Dermatol. 2005;6:383-392.
- Armstrong AW, Schupp C, Wu J, et al. Quality of life and work productivity impairment among psoriasis patients: findings from the National Psoriasis Foundation survey data 2003-2011. PloS One. 2012;7:e52935.
- Halioua B, Sid-Mohand D, Roussel ME, et al. Extent of misconceptions, negative prejudices and discriminatory behaviour to psoriasis patients in France. J Eur Acad Dermatol Venereol. 2016;30:650-654.
- Wolf P, Weger W, Legat F, et al. Quality of life and treatment goals in psoriasis from the patient perspective: results of an Austrian cross-sectional survey. J Dtsch Dermatol Ges. 2018;16:981-990.
- Shah K, Mellars L, Changolkar A, et al. Real-world burden of comorbidities in US patients with psoriasis. J Am Acad Dermatol. 2017;77:287-292.e4.
- Dowlatshahi EA, Wakkee M, Herings RM, et al. Increased antidepressant drug exposure in psoriasis patients: a longitudinal population-based cohort study. Acta Derm Venereol. 2013;93:544-550.
- Dowlatshahi EA, Wakkee M, Arends LR, et al. The prevalence and odds of depressive symptoms and clinical depression in psoriasis patients: a systematic review and meta-analysis. J Invest Dermatol. 2014;134:1542-1551.
- Singh S, Taylor C, Kornmehl H, et al. Psoriasis and suicidality: a systematic review and meta-analysis. J Am Acad Dermatol. 2017;77:425.e2-440.e2.
- Lakshmy S, Balasundaram S, Sarkar S, et al. A cross-sectional study of prevalence and implications of depression and anxiety in psoriasis. Indian J Psychol Med. 2015;37:434-440.
- Renzi C, Picardi A, Abeni D, et al. Association of dissatisfaction with care and psychiatric morbidity with poor treatment compliance. Arch Dermatol. 2002;138:337-342.
- Kulkarni AS, Balkrishnan R, Camacho FT, et al. Medication and health care service utilization related to depressive symptoms in older adults with psoriasis. J Drugs Dermatol. 2004;3:661-666.
- Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606-613.
- Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097.
- Kroenke K, Spitzer RL, Williams JB. The Patient Health Questionnaire-2: validity of a two-item depression screener. Med Care. 2003;41:1284-1292.
- Lamb RC, Matcham F, Turner MA, et al. Screening for anxiety and depression in people with psoriasis: a cross-sectional study in a tertiary referral setting. Br J Dermatol. 2017;176:1028-1034.
- Law M, Naughton MT, Dhar A, et al. Validation of two depression screening instruments in a sleep disorders clinic. J Clin Sleep Med. 2014;10:683-688.
- Cuijpers P, Dekker J, Hollon SD, et al. Adding psychotherapy to pharmacotherapy in the treatment of depressive disorders in adults: a meta-analysis. J Clin Psychiatry. 2009;70:1219-1229.
- National Psoriasis Foundation. Living with psoriatic arthritis. https://www.psoriasis.org/life-with-psoriatic-arthritis. Accessed September 23, 2018.
- Gaikwad R, Deshpande S, Raje S, et al. Evaluation of functional impairment in psoriasis. Indian J Dermatol Venereol Leprol. 2006;72:37-40.
- Claudepierre P, Lahfa M, Levy P, et al. The impact of psoriasis on professional life: PsoPRO, a French national survey [published online April 6, 2018]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.14986.
- Korman NJ, Zhao Y, Pike J, et al. Relationship between psoriasis severity, clinical symptoms, quality of life and work productivity among patients in the USA. Clin Exp Dermatol. 2016;41:514-521.
- Reilly MC, Zbrozek AS, Dukes EM. The validity and reproducibility of a work productivity and activity impairment instrument. PharmacoEconomics. 1993;4:353-365.
- Korman NJ, Zhao Y, Pike J, et al. Increased severity of itching, pain, and scaling in psoriasis patients is associated with increased disease severity, reduced quality of life, and reduced work productivity. Dermatol Online J. 2015;21.
- Spelman L, Su JC, Fernandez-Penas P, et al. Frequency of undiagnosed psoriatic arthritis among psoriasis patients in Australian dermatology practice. J Eur Acad Dermatol Venereol. 2015;29:2184-2191.
- Tillett W, Shaddick G, Askari A, et al. Factors influencing work disability in psoriatic arthritis: first results from a large UK multicentre study. Rheumatology (Oxford). 2015;54:157-162.
- Raychaudhuri SP, Wilken R, Sukhov AC, et al. Management of psoriatic arthritis: early diagnosis, monitoring of disease severity and cutting edge therapies. J Autoimmun. 2017;76:21-37.
- Gossec L, Smolen JS, Ramiro S, et al. European League Against Rheumatism (EULAR) recommendations for the manegement of psoriatic arthritis with pharmacological therapies: 2015 update. Ann Rheum Dis. 2016;75:499-510.
- Beroukhim K, Danesh M, Nguyen C, et al. A prospective, interventional assessment of the impact of ustekinumab treatment on psoriasis-related work productivity and activity impairment. J Dermatol Treat. 2016;27:552-555.
- Armstrong AW, Lynde CW, McBride SR, et al. Effect of ixekizumab treatment on work productivity for patients with moderate-to-severe plaque psoriasis: analysis of results from 3 randomized phase 3 clinical trials. JAMA Dermatol. 2016;152:661-669.
- Kimball AB, Yu AP, Signorovitch J, et al. The effects of adalimumab treatment and psoriasis severity on self-reported work productivity and activity impairment for patients with moderate to severe psoriasis. J Am Acad Dermatol. 2012;66:e67-76.
- Feldman SR, Malakouti M, Koo JY. Social impact of the burden of psoriasis: effects on patients and practice. Dermatol Online J. 2014;20.
- Reich A, Welz-Kubiak K, Rams Ł. Apprehension of the disease by patients suffering from psoriasis. Postepy Dermatol Alergol. 2014;31:289-293.
- Gupta MA, Gupta AK, Watteel GN. Perceived deprivation of social touch in psoriasis is associated with greater psychologic morbidity: an index of the stigma experience in dermatologic disorders. Cutis. 1998;61:339-342.
- Basra MK, Finlay AY. The family impact of skin diseases: the Greater Patient concept. Br J Dermatol. 2007;156:929-937.
- Eghlileb AM, Davies EE, Finlay AY. Psoriasis has a major secondary impact on the lives of family members and partners. Br J Dermatol. 2007;156:1245-1250.
- Janowski K, Steuden S, Pietrzak A, et al. Social support and adaptation to the disease in men and women with psoriasis. Arch Dermatol Res. 2012;304:421-432.
- Sampogna F, Abeni D, Gieler U, et al. Impairment of sexual life in 3,485 dermatological outpatients from a multicentre study in 13 European countries. Acta Derm Venereol. 2017;97:478-482.
- Sampogna F, Gisondi P, Tabolli S, et al. Impairment of sexual life in patients with psoriasis. Dermatology. 2007;214:144-150.
- Molina-Leyva A, Molina-Leyva I, Almodovar-Real A, et al. Prevalence and associated factors of erectile dysfunction in patients with moderate to severe psoriasis and healthy population: a comparative study considering physical and psychological factors. Arch Sex Behav. 2016;45:2047-2055.
- Ryan C, Sadlier M, De Vol E, et al. Genital psoriasis is associated with significant impairment in quality of life and sexual functioning. J Am Acad Dermatol. 2015;72:978-983.
- Labbate LA, Lare SB. Sexual dysfunction in male psychiatric outpatients: validity of the Massachusetts General Hospital Sexual Functioning Questionnaire. Psychother Psychosom. 2001;70:221-225.
- Molina-Leyva A, Almodovar-Real A, Ruiz-Carrascosa JC, et al. Distribution pattern of psoriasis affects sexual function in moderate to severe psoriasis: a prospective case series study. J Sex Med. 2014;11:2882-2889.
- Guenther L, Han C, Szapary P, et al. Impact of ustekinumab on health-related quality of life and sexual difficulties associated with psoriasis: results from two phase III clinical trials. J Eur Acad Dermatol Venereol. 2011;25:851-857.
- Guenther L, Warren RB, Cather JC, et al. Impact of ixekizumab treatment on skin-related personal relationship difficulties in moderate-to-severe psoriasis patients: 12-week results from two Phase 3 trials. J Eur Acad Dermatol Venereol. 2017;31:1867-1875.
The psychosocial impact of psoriasis is a critical component of disease burden. Psoriatic patients have high rates of depression and anxiety, problems at work, and difficulties with interpersonal relationships and intimacy.1 A National Psoriasis Foundation (NPF) survey from 2003 to 2011 reported that psoriasis affects overall emotional well-being in 88% of patients and enjoyment of life in 82% of patients.2
The reasons for psychosocial burden stem from public misconceptions and disease stigma. A survey of 1005 individuals (age range, 16–64 years) about their perceptions of psoriasis revealed that 16.5% believed that psoriasis is contagious and 6.8% believed that psoriasis is related to personal hygiene.3 Fifty percent practiced discriminatory behavior toward psoriatic patients, including reluctance to shake hands (28.8%) and engage in sexual relations/intercourse (44.1%). Sixty-five percent of psoriatic patients felt their appearance is unsightly, and 73% felt self-conscious about having psoriasis.2
The psychosocial burden exists despite medical treatment of the disease. In a cross-sectional study of 1184 psoriatic patients, 70.2% had impaired quality of life (QOL) as measured by the dermatology life quality index (DLQI), even after receiving a 4-week treatment for psoriasis.4 Medical treatment of psoriasis is not enough; providers need to assess overall QOL and provide treatment and resources for these patients in addition to symptomatic management.
There have been many studies on the psychosocial burden of psoriasis, but few have focused on a dermatology resident’s role in addressing this issue. This article will review psychosocial domains—psychiatric comorbidities and social functioning including occupational functioning, interpersonal relationships, and sexual functioning— and discuss a dermatology resident’s role in assessing and addressing each of these areas.
Methods
A PubMed search of articles indexed for MEDLINE was conducted using the following terms: psoriasis, depression, anxiety, work productivity, sexual functioning, and interpersonal relationships. Selected articles covered prevalence, assessment, and management of each psychosocial domain.
Results
Psychiatric Comorbidities
Prevalence
A high prevalence of psychiatric comorbidities exists in psoriatic patients. In a study of 469,097 patients with psoriasis, depression was the third most prevalent comorbidity (17.91%), following hyperlipidemia (45.64%) and hypertension (42.19%).5 In a 10-year longitudinal, population-based, prospective cohort study, antidepressant prescriptions were twice as frequent in psoriatic patients (17.8%) compared to control (7.9%)(P<.001).6 In a meta-analysis of 98 studies investigating psoriatic patients and psychiatric comorbidities, patients with psoriasis were 1.5 times more likely to experience depression (odds ratio [OR]: 1.57; 95% CI, 1.40-1.76) and use antidepressants (OR: 4.24; 95% CI, 1.53-11.76) compared to control.7 Patients with psoriasis were more likely to attempt suicide (OR: 1.32; 95% CI, 1.14-1.54) and complete suicide (OR: 1.20; 95% CI, 1.04-1.39) compared to people without psoriasis.8 A 1-year cross-sectional study of 90 psoriatic patients reported 78.7% were diagnosed with depression and 76.7% were diagnosed with anxiety. Seventy-two percent reported both anxiety and depression, correlating with worse QOL (χ2=26.7; P<.05).9
Assessment
Psychiatric comorbidities are assessed using clinical judgment and formal screening questionnaires in research studies. Signs of depression in patients with psoriasis can manifest as poor treatment adherence and recurrent flares of psoriasis.10,11 Psoriatic patients with psychiatric comorbidities were less likely to be adherent to treatment (risk ratio: 0.35; P<.003).10 The patient health questionnaire (PHQ) 9 and generalized anxiety disorder scale (GAD) 7 are validated and reliable questionnaires. The first 2 questions in PHQ-9 and GAD-7 screen for depression and anxiety, respectively.12-14 These 2-question screens are practical in a fast-paced dermatology outpatient setting. Systematic questionnaires specifically targeting mood disorders may be more beneficial than the widely used DLQI, which may not adequately capture mood disorders. Over the course of 10 months, 607 patients with psoriasis were asked to fill out the PHQ-9, GAD-7, and DLQI. Thirty-eight percent of patients with major depressive disorder had a DLQI score lower than 10, while 46% of patients with generalized anxiety disorder had a DLQI score lower than 10.15 Other questionnaires, including the hospital anxiety and depression scale and Beck depression inventory, are valid instruments with high sensitivity but are commonly used for research purposes and may not be clinically feasible.16
Management
Dermatologists should refer patients with depression and/or anxiety to psychiatry. Interventions include pharmacologic and nonpharmacologic management. First-line therapy for depression and anxiety is a combination of selective serotonin reuptake inhibitors and cognitive behavioral therapy.17 In addition, providers can direct patients to online resources such as the NPF website, where patients with psoriasis can access information about the signs and symptoms of mood disorders and contact the patient navigation center for further help.18
Social Functioning
Occupational Prevalence
The NPF found that 92% of patients with psoriasis or psoriatic arthritis (PsA) surveyed between 2003 and 2011 cited their psoriasis as reason for unemployment.2 In a survey of 43 patients asked about social and occupational functioning using the social and occupational assessment scale, 62.5% of psoriatic patients reported distress at work and 51.1% reported decreased efficiency at work.19 A national online survey that was conducted in France and issued to patients with and without psoriasis assessed overall QOL and work productivity using the work productivity and activity impairment questionnaire for psoriasis (WPAI-PSO). Of 714 patients with psoriasis and PsA, the latter had a 57.6% decrease in work productivity over 7 days compared to 27.9% in controls (P<.05).20 Occupational impairment leads to lost wages and hinders advancement, further exacerbating the psychosocial burden of psoriasis.21
Occupational Assessment
Formal assessment of occupational function can be done with the WPAI-PSO, a 6-question valid instrument.22 Providers may look for risk factors associated with greater loss in work productivity to help identify and offer support for patients. Patients with increased severity of itching, pain, and scaling experienced a greater decrease in work productivity.21,23 Patients with PsA warrant early detection and treatment because they experience greater physical restraints that can interfere with work activities. Of the 459 psoriatic patients without a prior diagnosis of PsA who filled out the PsA screening and evaluation questionnaire, 144 (31.4%) received a score of 44 or higher and were referred to rheumatology for further evaluation with the classification criteria for PsA. Nine percent of patients failed to be screened and remained undiagnosed with PsA.24 In a study using the health assessment questionnaire to assess 400 patients with PsA, those with worse physical function due to joint pain and stiffness were less likely to remain employed (OR: 0.56; P=.02).25
Occupational Management
Identifying and coordinating symptoms of PsA between dermatology and rheumatology is beneficial for patients who experience debilitating symptoms. There are a variety of treatments available for PsA. According to the European League Against Rheumatism 2015 guidelines developed from expert opinion and systematic reviews for PsA management, there are 4 phases of treatment, with reassessment every 3 to 6 months for effectiveness of therapy.26,27 Phase I involves initiating nonsteroidal anti-inflammatory drugs with or without glucocorticoid injections. Phase II involves synthetic disease-modifying drugs, including methotrexate, leflunomide, sulfasalazine, or cyclosporine. Phase III involves adding a second synthetic disease-modifying drug or starting a biologic, such as an anti–tumor necrosis factor, IL-12/IL-23, or IL-17 inhibitor. Phase IV involves switching to a different drug in either aforementioned class.26,27 Treatment with biologics improves work productivity as assessed by WPAI-PSO for psoriasis and PsA.28-30 Encouraging patients to speak up in the workplace and request small accommodations such as timely breaks or ergonomic chairs can help patients feel more comfortable and supported in the work environment.18 Patients who felt supported at work were more likely to remain employed.25
Interpersonal Relationships Prevalence
Misinformation about psoriasis, fear of rejection, and feelings of isolation may contribute to interpersonal conflict. Patients have feelings of shame and self-consciousness that hinder them from engaging in social activities and seeking out relationships.31 Twenty-nine percent of patients feel that psoriasis has interfered with establishing relationships because of negative self-esteem associated with the disease,32 and 26.3% have experienced people avoiding physical contact.33 Family and spouses of patients with psoriasis may be secondarily affected due to economic and emotional distress. Ninety-eight percent of family members of psoriatic patients experienced emotional distress and 54% experienced the burden of care.34 In a survey of 63 relatives and partners of patients with psoriasis, 57% experienced psychological distress, including anxiety and worry over a psoriatic patient’s future.35
Interpersonal Relationships Assessment
Current available tools, including the DLQI and short form health survey, measure overall QOL, including social functioning, but may not be practical in a clinic setting. Although no quick-screening test to assess for this domain exists, providers are encouraged to ask patients about disease impact on interpersonal relationships. The family DLQI questionnaire, adapted from the DLQI, may help physicians and social workers evaluate the burden on a patient’s family members.34
Interpersonal Relationships Management
It may be difficult for providers to address problems with interpersonal relationships without accessible tools. Patients may not be accompanied by family or friends during appointments, and it is difficult to screen for these issues during visits. Providers may offer resources such as the NPF website, which provides information about support groups. It also provides tips on dating and connecting to others in the community who share similar experiences.18 Encouraging patients to seek family or couples therapy also may be beneficial. Increased social support can lead to better QOL and fewer depressive symptoms.36
Sexual Functioning Prevalence
Psoriasis affects both physical and psychological components of sexual function. Among 3485 patients with skin conditions who were surveyed about sexual function, 34% of psoriatic patients reported that psoriasis interfered with sexual functioning at least to a certain degree.37 Sexual impairment was strongly associated with depression, anxiety, and suicidal ideation; 24% of depressed patients and 20% of anxious patients experienced sexual problems a lot or very much, based on the DLQI.37 Depending on the questionnaire used, the prevalence of sexual dysfunction due to psoriasis ranged from 35.5% to 71.3%.38 In an observational cohort study of 158 participants (n=79 psoriasis patients and n=79 controls), 34.2% of patients with psoriasis experienced erectile dysfunction compared to 17.7% of controls.39 Forty-two percent of psoriatic patients with genital involvement reported dyspareunia, 32% reported worsening of genital psoriasis after intercourse, and 43% reported decreased frequency of intercourse.40
Sexual Functioning Assessment
The Skindex-29, DLQI, and psoriasis disability index are available QOL tools that include one question evaluating difficulties with sexual function. The
Sexual Functioning Management
Better disease control leads to improved sexual function, as patients experience fewer feelings of shame, anxiety, and depression, as well as improvement of physical symptoms that can interfere with sexual functioning.38,43,44 Reducing friction, warmth, and moisture, as well as avoiding tight clothing, can help those with genital psoriasis. Patients are advised to reapply topical medications after sexual intercourse. Patients also can apply makeup to disguise psoriasis and help reduce feelings of self-consciousness that can impede sexual intimacy.18
Comment
The psychosocial burden of psoriasis penetrates many facets of patient lives. Psoriasis can invoke feelings of shame and embarrassment that are worsened by the public’s misconceptions about psoriasis, resulting in serious mental health issues that can cause even greater disability. Depression and anxiety are prevalent in patients with psoriasis. The characteristic symptoms of pain and pruritus along with psychiatric comorbidities can have an underestimated impact on daily activities, including employment, interpersonal relationships, and sexual function. Such dysfunctions have serious implications toward wages, professional advancement, social support, and overall QOL.
Dermatology providers play an important role in screening for these problems through validated questionnaires and identifying risks. Simple screening questions such as the PHQ-9 can be beneficial and feasible during dermatology visits. Screening for PsA can help patients avoid problems at work. Sexual dysfunction is a sensitive topic; however, providers can use a 1-question screen from valid questionnaires and inquire about the location of lesions as opportunities to address this issue.
Interventions lead to better disease control, which concurrently improves overall QOL. These interventions depend on both patient adherence and a physician’s commitment to finding an optimal treatment regimen for each individual. Medical management; coordinating care; developing treatment plans with psychiatry, rheumatology, and primary care providers; and psychological counseling and services may be necessary and beneficial (Table). Offering accessible resources such as the NPF website helps patients access information outside the clinic when it is not feasible to address all these concerns in a single visit. Psoriasis requires more than just medical management; it requires dermatology providers to use a multidisciplinary approach to address the psychosocial aspects of the disease.
Conclusion
The psychosocial burden of psoriasis is immense. Stigma, public misconception, mental health concerns, and occupational and interpersonal difficulty are the basis of disease burden. Providers play a vital role in assessing the effect psoriasis has on different areas of patients’ lives and providing appropriate interventions and resources to reduce disease burden.
The psychosocial impact of psoriasis is a critical component of disease burden. Psoriatic patients have high rates of depression and anxiety, problems at work, and difficulties with interpersonal relationships and intimacy.1 A National Psoriasis Foundation (NPF) survey from 2003 to 2011 reported that psoriasis affects overall emotional well-being in 88% of patients and enjoyment of life in 82% of patients.2
The reasons for psychosocial burden stem from public misconceptions and disease stigma. A survey of 1005 individuals (age range, 16–64 years) about their perceptions of psoriasis revealed that 16.5% believed that psoriasis is contagious and 6.8% believed that psoriasis is related to personal hygiene.3 Fifty percent practiced discriminatory behavior toward psoriatic patients, including reluctance to shake hands (28.8%) and engage in sexual relations/intercourse (44.1%). Sixty-five percent of psoriatic patients felt their appearance is unsightly, and 73% felt self-conscious about having psoriasis.2
The psychosocial burden exists despite medical treatment of the disease. In a cross-sectional study of 1184 psoriatic patients, 70.2% had impaired quality of life (QOL) as measured by the dermatology life quality index (DLQI), even after receiving a 4-week treatment for psoriasis.4 Medical treatment of psoriasis is not enough; providers need to assess overall QOL and provide treatment and resources for these patients in addition to symptomatic management.
There have been many studies on the psychosocial burden of psoriasis, but few have focused on a dermatology resident’s role in addressing this issue. This article will review psychosocial domains—psychiatric comorbidities and social functioning including occupational functioning, interpersonal relationships, and sexual functioning— and discuss a dermatology resident’s role in assessing and addressing each of these areas.
Methods
A PubMed search of articles indexed for MEDLINE was conducted using the following terms: psoriasis, depression, anxiety, work productivity, sexual functioning, and interpersonal relationships. Selected articles covered prevalence, assessment, and management of each psychosocial domain.
Results
Psychiatric Comorbidities
Prevalence
A high prevalence of psychiatric comorbidities exists in psoriatic patients. In a study of 469,097 patients with psoriasis, depression was the third most prevalent comorbidity (17.91%), following hyperlipidemia (45.64%) and hypertension (42.19%).5 In a 10-year longitudinal, population-based, prospective cohort study, antidepressant prescriptions were twice as frequent in psoriatic patients (17.8%) compared to control (7.9%)(P<.001).6 In a meta-analysis of 98 studies investigating psoriatic patients and psychiatric comorbidities, patients with psoriasis were 1.5 times more likely to experience depression (odds ratio [OR]: 1.57; 95% CI, 1.40-1.76) and use antidepressants (OR: 4.24; 95% CI, 1.53-11.76) compared to control.7 Patients with psoriasis were more likely to attempt suicide (OR: 1.32; 95% CI, 1.14-1.54) and complete suicide (OR: 1.20; 95% CI, 1.04-1.39) compared to people without psoriasis.8 A 1-year cross-sectional study of 90 psoriatic patients reported 78.7% were diagnosed with depression and 76.7% were diagnosed with anxiety. Seventy-two percent reported both anxiety and depression, correlating with worse QOL (χ2=26.7; P<.05).9
Assessment
Psychiatric comorbidities are assessed using clinical judgment and formal screening questionnaires in research studies. Signs of depression in patients with psoriasis can manifest as poor treatment adherence and recurrent flares of psoriasis.10,11 Psoriatic patients with psychiatric comorbidities were less likely to be adherent to treatment (risk ratio: 0.35; P<.003).10 The patient health questionnaire (PHQ) 9 and generalized anxiety disorder scale (GAD) 7 are validated and reliable questionnaires. The first 2 questions in PHQ-9 and GAD-7 screen for depression and anxiety, respectively.12-14 These 2-question screens are practical in a fast-paced dermatology outpatient setting. Systematic questionnaires specifically targeting mood disorders may be more beneficial than the widely used DLQI, which may not adequately capture mood disorders. Over the course of 10 months, 607 patients with psoriasis were asked to fill out the PHQ-9, GAD-7, and DLQI. Thirty-eight percent of patients with major depressive disorder had a DLQI score lower than 10, while 46% of patients with generalized anxiety disorder had a DLQI score lower than 10.15 Other questionnaires, including the hospital anxiety and depression scale and Beck depression inventory, are valid instruments with high sensitivity but are commonly used for research purposes and may not be clinically feasible.16
Management
Dermatologists should refer patients with depression and/or anxiety to psychiatry. Interventions include pharmacologic and nonpharmacologic management. First-line therapy for depression and anxiety is a combination of selective serotonin reuptake inhibitors and cognitive behavioral therapy.17 In addition, providers can direct patients to online resources such as the NPF website, where patients with psoriasis can access information about the signs and symptoms of mood disorders and contact the patient navigation center for further help.18
Social Functioning
Occupational Prevalence
The NPF found that 92% of patients with psoriasis or psoriatic arthritis (PsA) surveyed between 2003 and 2011 cited their psoriasis as reason for unemployment.2 In a survey of 43 patients asked about social and occupational functioning using the social and occupational assessment scale, 62.5% of psoriatic patients reported distress at work and 51.1% reported decreased efficiency at work.19 A national online survey that was conducted in France and issued to patients with and without psoriasis assessed overall QOL and work productivity using the work productivity and activity impairment questionnaire for psoriasis (WPAI-PSO). Of 714 patients with psoriasis and PsA, the latter had a 57.6% decrease in work productivity over 7 days compared to 27.9% in controls (P<.05).20 Occupational impairment leads to lost wages and hinders advancement, further exacerbating the psychosocial burden of psoriasis.21
Occupational Assessment
Formal assessment of occupational function can be done with the WPAI-PSO, a 6-question valid instrument.22 Providers may look for risk factors associated with greater loss in work productivity to help identify and offer support for patients. Patients with increased severity of itching, pain, and scaling experienced a greater decrease in work productivity.21,23 Patients with PsA warrant early detection and treatment because they experience greater physical restraints that can interfere with work activities. Of the 459 psoriatic patients without a prior diagnosis of PsA who filled out the PsA screening and evaluation questionnaire, 144 (31.4%) received a score of 44 or higher and were referred to rheumatology for further evaluation with the classification criteria for PsA. Nine percent of patients failed to be screened and remained undiagnosed with PsA.24 In a study using the health assessment questionnaire to assess 400 patients with PsA, those with worse physical function due to joint pain and stiffness were less likely to remain employed (OR: 0.56; P=.02).25
Occupational Management
Identifying and coordinating symptoms of PsA between dermatology and rheumatology is beneficial for patients who experience debilitating symptoms. There are a variety of treatments available for PsA. According to the European League Against Rheumatism 2015 guidelines developed from expert opinion and systematic reviews for PsA management, there are 4 phases of treatment, with reassessment every 3 to 6 months for effectiveness of therapy.26,27 Phase I involves initiating nonsteroidal anti-inflammatory drugs with or without glucocorticoid injections. Phase II involves synthetic disease-modifying drugs, including methotrexate, leflunomide, sulfasalazine, or cyclosporine. Phase III involves adding a second synthetic disease-modifying drug or starting a biologic, such as an anti–tumor necrosis factor, IL-12/IL-23, or IL-17 inhibitor. Phase IV involves switching to a different drug in either aforementioned class.26,27 Treatment with biologics improves work productivity as assessed by WPAI-PSO for psoriasis and PsA.28-30 Encouraging patients to speak up in the workplace and request small accommodations such as timely breaks or ergonomic chairs can help patients feel more comfortable and supported in the work environment.18 Patients who felt supported at work were more likely to remain employed.25
Interpersonal Relationships Prevalence
Misinformation about psoriasis, fear of rejection, and feelings of isolation may contribute to interpersonal conflict. Patients have feelings of shame and self-consciousness that hinder them from engaging in social activities and seeking out relationships.31 Twenty-nine percent of patients feel that psoriasis has interfered with establishing relationships because of negative self-esteem associated with the disease,32 and 26.3% have experienced people avoiding physical contact.33 Family and spouses of patients with psoriasis may be secondarily affected due to economic and emotional distress. Ninety-eight percent of family members of psoriatic patients experienced emotional distress and 54% experienced the burden of care.34 In a survey of 63 relatives and partners of patients with psoriasis, 57% experienced psychological distress, including anxiety and worry over a psoriatic patient’s future.35
Interpersonal Relationships Assessment
Current available tools, including the DLQI and short form health survey, measure overall QOL, including social functioning, but may not be practical in a clinic setting. Although no quick-screening test to assess for this domain exists, providers are encouraged to ask patients about disease impact on interpersonal relationships. The family DLQI questionnaire, adapted from the DLQI, may help physicians and social workers evaluate the burden on a patient’s family members.34
Interpersonal Relationships Management
It may be difficult for providers to address problems with interpersonal relationships without accessible tools. Patients may not be accompanied by family or friends during appointments, and it is difficult to screen for these issues during visits. Providers may offer resources such as the NPF website, which provides information about support groups. It also provides tips on dating and connecting to others in the community who share similar experiences.18 Encouraging patients to seek family or couples therapy also may be beneficial. Increased social support can lead to better QOL and fewer depressive symptoms.36
Sexual Functioning Prevalence
Psoriasis affects both physical and psychological components of sexual function. Among 3485 patients with skin conditions who were surveyed about sexual function, 34% of psoriatic patients reported that psoriasis interfered with sexual functioning at least to a certain degree.37 Sexual impairment was strongly associated with depression, anxiety, and suicidal ideation; 24% of depressed patients and 20% of anxious patients experienced sexual problems a lot or very much, based on the DLQI.37 Depending on the questionnaire used, the prevalence of sexual dysfunction due to psoriasis ranged from 35.5% to 71.3%.38 In an observational cohort study of 158 participants (n=79 psoriasis patients and n=79 controls), 34.2% of patients with psoriasis experienced erectile dysfunction compared to 17.7% of controls.39 Forty-two percent of psoriatic patients with genital involvement reported dyspareunia, 32% reported worsening of genital psoriasis after intercourse, and 43% reported decreased frequency of intercourse.40
Sexual Functioning Assessment
The Skindex-29, DLQI, and psoriasis disability index are available QOL tools that include one question evaluating difficulties with sexual function. The
Sexual Functioning Management
Better disease control leads to improved sexual function, as patients experience fewer feelings of shame, anxiety, and depression, as well as improvement of physical symptoms that can interfere with sexual functioning.38,43,44 Reducing friction, warmth, and moisture, as well as avoiding tight clothing, can help those with genital psoriasis. Patients are advised to reapply topical medications after sexual intercourse. Patients also can apply makeup to disguise psoriasis and help reduce feelings of self-consciousness that can impede sexual intimacy.18
Comment
The psychosocial burden of psoriasis penetrates many facets of patient lives. Psoriasis can invoke feelings of shame and embarrassment that are worsened by the public’s misconceptions about psoriasis, resulting in serious mental health issues that can cause even greater disability. Depression and anxiety are prevalent in patients with psoriasis. The characteristic symptoms of pain and pruritus along with psychiatric comorbidities can have an underestimated impact on daily activities, including employment, interpersonal relationships, and sexual function. Such dysfunctions have serious implications toward wages, professional advancement, social support, and overall QOL.
Dermatology providers play an important role in screening for these problems through validated questionnaires and identifying risks. Simple screening questions such as the PHQ-9 can be beneficial and feasible during dermatology visits. Screening for PsA can help patients avoid problems at work. Sexual dysfunction is a sensitive topic; however, providers can use a 1-question screen from valid questionnaires and inquire about the location of lesions as opportunities to address this issue.
Interventions lead to better disease control, which concurrently improves overall QOL. These interventions depend on both patient adherence and a physician’s commitment to finding an optimal treatment regimen for each individual. Medical management; coordinating care; developing treatment plans with psychiatry, rheumatology, and primary care providers; and psychological counseling and services may be necessary and beneficial (Table). Offering accessible resources such as the NPF website helps patients access information outside the clinic when it is not feasible to address all these concerns in a single visit. Psoriasis requires more than just medical management; it requires dermatology providers to use a multidisciplinary approach to address the psychosocial aspects of the disease.
Conclusion
The psychosocial burden of psoriasis is immense. Stigma, public misconception, mental health concerns, and occupational and interpersonal difficulty are the basis of disease burden. Providers play a vital role in assessing the effect psoriasis has on different areas of patients’ lives and providing appropriate interventions and resources to reduce disease burden.
- Kimball AB, Jacobson C, Weiss S, et al. The psychosocial burden of psoriasis. Am J Clin Dermatol. 2005;6:383-392.
- Armstrong AW, Schupp C, Wu J, et al. Quality of life and work productivity impairment among psoriasis patients: findings from the National Psoriasis Foundation survey data 2003-2011. PloS One. 2012;7:e52935.
- Halioua B, Sid-Mohand D, Roussel ME, et al. Extent of misconceptions, negative prejudices and discriminatory behaviour to psoriasis patients in France. J Eur Acad Dermatol Venereol. 2016;30:650-654.
- Wolf P, Weger W, Legat F, et al. Quality of life and treatment goals in psoriasis from the patient perspective: results of an Austrian cross-sectional survey. J Dtsch Dermatol Ges. 2018;16:981-990.
- Shah K, Mellars L, Changolkar A, et al. Real-world burden of comorbidities in US patients with psoriasis. J Am Acad Dermatol. 2017;77:287-292.e4.
- Dowlatshahi EA, Wakkee M, Herings RM, et al. Increased antidepressant drug exposure in psoriasis patients: a longitudinal population-based cohort study. Acta Derm Venereol. 2013;93:544-550.
- Dowlatshahi EA, Wakkee M, Arends LR, et al. The prevalence and odds of depressive symptoms and clinical depression in psoriasis patients: a systematic review and meta-analysis. J Invest Dermatol. 2014;134:1542-1551.
- Singh S, Taylor C, Kornmehl H, et al. Psoriasis and suicidality: a systematic review and meta-analysis. J Am Acad Dermatol. 2017;77:425.e2-440.e2.
- Lakshmy S, Balasundaram S, Sarkar S, et al. A cross-sectional study of prevalence and implications of depression and anxiety in psoriasis. Indian J Psychol Med. 2015;37:434-440.
- Renzi C, Picardi A, Abeni D, et al. Association of dissatisfaction with care and psychiatric morbidity with poor treatment compliance. Arch Dermatol. 2002;138:337-342.
- Kulkarni AS, Balkrishnan R, Camacho FT, et al. Medication and health care service utilization related to depressive symptoms in older adults with psoriasis. J Drugs Dermatol. 2004;3:661-666.
- Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606-613.
- Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097.
- Kroenke K, Spitzer RL, Williams JB. The Patient Health Questionnaire-2: validity of a two-item depression screener. Med Care. 2003;41:1284-1292.
- Lamb RC, Matcham F, Turner MA, et al. Screening for anxiety and depression in people with psoriasis: a cross-sectional study in a tertiary referral setting. Br J Dermatol. 2017;176:1028-1034.
- Law M, Naughton MT, Dhar A, et al. Validation of two depression screening instruments in a sleep disorders clinic. J Clin Sleep Med. 2014;10:683-688.
- Cuijpers P, Dekker J, Hollon SD, et al. Adding psychotherapy to pharmacotherapy in the treatment of depressive disorders in adults: a meta-analysis. J Clin Psychiatry. 2009;70:1219-1229.
- National Psoriasis Foundation. Living with psoriatic arthritis. https://www.psoriasis.org/life-with-psoriatic-arthritis. Accessed September 23, 2018.
- Gaikwad R, Deshpande S, Raje S, et al. Evaluation of functional impairment in psoriasis. Indian J Dermatol Venereol Leprol. 2006;72:37-40.
- Claudepierre P, Lahfa M, Levy P, et al. The impact of psoriasis on professional life: PsoPRO, a French national survey [published online April 6, 2018]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.14986.
- Korman NJ, Zhao Y, Pike J, et al. Relationship between psoriasis severity, clinical symptoms, quality of life and work productivity among patients in the USA. Clin Exp Dermatol. 2016;41:514-521.
- Reilly MC, Zbrozek AS, Dukes EM. The validity and reproducibility of a work productivity and activity impairment instrument. PharmacoEconomics. 1993;4:353-365.
- Korman NJ, Zhao Y, Pike J, et al. Increased severity of itching, pain, and scaling in psoriasis patients is associated with increased disease severity, reduced quality of life, and reduced work productivity. Dermatol Online J. 2015;21.
- Spelman L, Su JC, Fernandez-Penas P, et al. Frequency of undiagnosed psoriatic arthritis among psoriasis patients in Australian dermatology practice. J Eur Acad Dermatol Venereol. 2015;29:2184-2191.
- Tillett W, Shaddick G, Askari A, et al. Factors influencing work disability in psoriatic arthritis: first results from a large UK multicentre study. Rheumatology (Oxford). 2015;54:157-162.
- Raychaudhuri SP, Wilken R, Sukhov AC, et al. Management of psoriatic arthritis: early diagnosis, monitoring of disease severity and cutting edge therapies. J Autoimmun. 2017;76:21-37.
- Gossec L, Smolen JS, Ramiro S, et al. European League Against Rheumatism (EULAR) recommendations for the manegement of psoriatic arthritis with pharmacological therapies: 2015 update. Ann Rheum Dis. 2016;75:499-510.
- Beroukhim K, Danesh M, Nguyen C, et al. A prospective, interventional assessment of the impact of ustekinumab treatment on psoriasis-related work productivity and activity impairment. J Dermatol Treat. 2016;27:552-555.
- Armstrong AW, Lynde CW, McBride SR, et al. Effect of ixekizumab treatment on work productivity for patients with moderate-to-severe plaque psoriasis: analysis of results from 3 randomized phase 3 clinical trials. JAMA Dermatol. 2016;152:661-669.
- Kimball AB, Yu AP, Signorovitch J, et al. The effects of adalimumab treatment and psoriasis severity on self-reported work productivity and activity impairment for patients with moderate to severe psoriasis. J Am Acad Dermatol. 2012;66:e67-76.
- Feldman SR, Malakouti M, Koo JY. Social impact of the burden of psoriasis: effects on patients and practice. Dermatol Online J. 2014;20.
- Reich A, Welz-Kubiak K, Rams Ł. Apprehension of the disease by patients suffering from psoriasis. Postepy Dermatol Alergol. 2014;31:289-293.
- Gupta MA, Gupta AK, Watteel GN. Perceived deprivation of social touch in psoriasis is associated with greater psychologic morbidity: an index of the stigma experience in dermatologic disorders. Cutis. 1998;61:339-342.
- Basra MK, Finlay AY. The family impact of skin diseases: the Greater Patient concept. Br J Dermatol. 2007;156:929-937.
- Eghlileb AM, Davies EE, Finlay AY. Psoriasis has a major secondary impact on the lives of family members and partners. Br J Dermatol. 2007;156:1245-1250.
- Janowski K, Steuden S, Pietrzak A, et al. Social support and adaptation to the disease in men and women with psoriasis. Arch Dermatol Res. 2012;304:421-432.
- Sampogna F, Abeni D, Gieler U, et al. Impairment of sexual life in 3,485 dermatological outpatients from a multicentre study in 13 European countries. Acta Derm Venereol. 2017;97:478-482.
- Sampogna F, Gisondi P, Tabolli S, et al. Impairment of sexual life in patients with psoriasis. Dermatology. 2007;214:144-150.
- Molina-Leyva A, Molina-Leyva I, Almodovar-Real A, et al. Prevalence and associated factors of erectile dysfunction in patients with moderate to severe psoriasis and healthy population: a comparative study considering physical and psychological factors. Arch Sex Behav. 2016;45:2047-2055.
- Ryan C, Sadlier M, De Vol E, et al. Genital psoriasis is associated with significant impairment in quality of life and sexual functioning. J Am Acad Dermatol. 2015;72:978-983.
- Labbate LA, Lare SB. Sexual dysfunction in male psychiatric outpatients: validity of the Massachusetts General Hospital Sexual Functioning Questionnaire. Psychother Psychosom. 2001;70:221-225.
- Molina-Leyva A, Almodovar-Real A, Ruiz-Carrascosa JC, et al. Distribution pattern of psoriasis affects sexual function in moderate to severe psoriasis: a prospective case series study. J Sex Med. 2014;11:2882-2889.
- Guenther L, Han C, Szapary P, et al. Impact of ustekinumab on health-related quality of life and sexual difficulties associated with psoriasis: results from two phase III clinical trials. J Eur Acad Dermatol Venereol. 2011;25:851-857.
- Guenther L, Warren RB, Cather JC, et al. Impact of ixekizumab treatment on skin-related personal relationship difficulties in moderate-to-severe psoriasis patients: 12-week results from two Phase 3 trials. J Eur Acad Dermatol Venereol. 2017;31:1867-1875.
- Kimball AB, Jacobson C, Weiss S, et al. The psychosocial burden of psoriasis. Am J Clin Dermatol. 2005;6:383-392.
- Armstrong AW, Schupp C, Wu J, et al. Quality of life and work productivity impairment among psoriasis patients: findings from the National Psoriasis Foundation survey data 2003-2011. PloS One. 2012;7:e52935.
- Halioua B, Sid-Mohand D, Roussel ME, et al. Extent of misconceptions, negative prejudices and discriminatory behaviour to psoriasis patients in France. J Eur Acad Dermatol Venereol. 2016;30:650-654.
- Wolf P, Weger W, Legat F, et al. Quality of life and treatment goals in psoriasis from the patient perspective: results of an Austrian cross-sectional survey. J Dtsch Dermatol Ges. 2018;16:981-990.
- Shah K, Mellars L, Changolkar A, et al. Real-world burden of comorbidities in US patients with psoriasis. J Am Acad Dermatol. 2017;77:287-292.e4.
- Dowlatshahi EA, Wakkee M, Herings RM, et al. Increased antidepressant drug exposure in psoriasis patients: a longitudinal population-based cohort study. Acta Derm Venereol. 2013;93:544-550.
- Dowlatshahi EA, Wakkee M, Arends LR, et al. The prevalence and odds of depressive symptoms and clinical depression in psoriasis patients: a systematic review and meta-analysis. J Invest Dermatol. 2014;134:1542-1551.
- Singh S, Taylor C, Kornmehl H, et al. Psoriasis and suicidality: a systematic review and meta-analysis. J Am Acad Dermatol. 2017;77:425.e2-440.e2.
- Lakshmy S, Balasundaram S, Sarkar S, et al. A cross-sectional study of prevalence and implications of depression and anxiety in psoriasis. Indian J Psychol Med. 2015;37:434-440.
- Renzi C, Picardi A, Abeni D, et al. Association of dissatisfaction with care and psychiatric morbidity with poor treatment compliance. Arch Dermatol. 2002;138:337-342.
- Kulkarni AS, Balkrishnan R, Camacho FT, et al. Medication and health care service utilization related to depressive symptoms in older adults with psoriasis. J Drugs Dermatol. 2004;3:661-666.
- Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16:606-613.
- Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097.
- Kroenke K, Spitzer RL, Williams JB. The Patient Health Questionnaire-2: validity of a two-item depression screener. Med Care. 2003;41:1284-1292.
- Lamb RC, Matcham F, Turner MA, et al. Screening for anxiety and depression in people with psoriasis: a cross-sectional study in a tertiary referral setting. Br J Dermatol. 2017;176:1028-1034.
- Law M, Naughton MT, Dhar A, et al. Validation of two depression screening instruments in a sleep disorders clinic. J Clin Sleep Med. 2014;10:683-688.
- Cuijpers P, Dekker J, Hollon SD, et al. Adding psychotherapy to pharmacotherapy in the treatment of depressive disorders in adults: a meta-analysis. J Clin Psychiatry. 2009;70:1219-1229.
- National Psoriasis Foundation. Living with psoriatic arthritis. https://www.psoriasis.org/life-with-psoriatic-arthritis. Accessed September 23, 2018.
- Gaikwad R, Deshpande S, Raje S, et al. Evaluation of functional impairment in psoriasis. Indian J Dermatol Venereol Leprol. 2006;72:37-40.
- Claudepierre P, Lahfa M, Levy P, et al. The impact of psoriasis on professional life: PsoPRO, a French national survey [published online April 6, 2018]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.14986.
- Korman NJ, Zhao Y, Pike J, et al. Relationship between psoriasis severity, clinical symptoms, quality of life and work productivity among patients in the USA. Clin Exp Dermatol. 2016;41:514-521.
- Reilly MC, Zbrozek AS, Dukes EM. The validity and reproducibility of a work productivity and activity impairment instrument. PharmacoEconomics. 1993;4:353-365.
- Korman NJ, Zhao Y, Pike J, et al. Increased severity of itching, pain, and scaling in psoriasis patients is associated with increased disease severity, reduced quality of life, and reduced work productivity. Dermatol Online J. 2015;21.
- Spelman L, Su JC, Fernandez-Penas P, et al. Frequency of undiagnosed psoriatic arthritis among psoriasis patients in Australian dermatology practice. J Eur Acad Dermatol Venereol. 2015;29:2184-2191.
- Tillett W, Shaddick G, Askari A, et al. Factors influencing work disability in psoriatic arthritis: first results from a large UK multicentre study. Rheumatology (Oxford). 2015;54:157-162.
- Raychaudhuri SP, Wilken R, Sukhov AC, et al. Management of psoriatic arthritis: early diagnosis, monitoring of disease severity and cutting edge therapies. J Autoimmun. 2017;76:21-37.
- Gossec L, Smolen JS, Ramiro S, et al. European League Against Rheumatism (EULAR) recommendations for the manegement of psoriatic arthritis with pharmacological therapies: 2015 update. Ann Rheum Dis. 2016;75:499-510.
- Beroukhim K, Danesh M, Nguyen C, et al. A prospective, interventional assessment of the impact of ustekinumab treatment on psoriasis-related work productivity and activity impairment. J Dermatol Treat. 2016;27:552-555.
- Armstrong AW, Lynde CW, McBride SR, et al. Effect of ixekizumab treatment on work productivity for patients with moderate-to-severe plaque psoriasis: analysis of results from 3 randomized phase 3 clinical trials. JAMA Dermatol. 2016;152:661-669.
- Kimball AB, Yu AP, Signorovitch J, et al. The effects of adalimumab treatment and psoriasis severity on self-reported work productivity and activity impairment for patients with moderate to severe psoriasis. J Am Acad Dermatol. 2012;66:e67-76.
- Feldman SR, Malakouti M, Koo JY. Social impact of the burden of psoriasis: effects on patients and practice. Dermatol Online J. 2014;20.
- Reich A, Welz-Kubiak K, Rams Ł. Apprehension of the disease by patients suffering from psoriasis. Postepy Dermatol Alergol. 2014;31:289-293.
- Gupta MA, Gupta AK, Watteel GN. Perceived deprivation of social touch in psoriasis is associated with greater psychologic morbidity: an index of the stigma experience in dermatologic disorders. Cutis. 1998;61:339-342.
- Basra MK, Finlay AY. The family impact of skin diseases: the Greater Patient concept. Br J Dermatol. 2007;156:929-937.
- Eghlileb AM, Davies EE, Finlay AY. Psoriasis has a major secondary impact on the lives of family members and partners. Br J Dermatol. 2007;156:1245-1250.
- Janowski K, Steuden S, Pietrzak A, et al. Social support and adaptation to the disease in men and women with psoriasis. Arch Dermatol Res. 2012;304:421-432.
- Sampogna F, Abeni D, Gieler U, et al. Impairment of sexual life in 3,485 dermatological outpatients from a multicentre study in 13 European countries. Acta Derm Venereol. 2017;97:478-482.
- Sampogna F, Gisondi P, Tabolli S, et al. Impairment of sexual life in patients with psoriasis. Dermatology. 2007;214:144-150.
- Molina-Leyva A, Molina-Leyva I, Almodovar-Real A, et al. Prevalence and associated factors of erectile dysfunction in patients with moderate to severe psoriasis and healthy population: a comparative study considering physical and psychological factors. Arch Sex Behav. 2016;45:2047-2055.
- Ryan C, Sadlier M, De Vol E, et al. Genital psoriasis is associated with significant impairment in quality of life and sexual functioning. J Am Acad Dermatol. 2015;72:978-983.
- Labbate LA, Lare SB. Sexual dysfunction in male psychiatric outpatients: validity of the Massachusetts General Hospital Sexual Functioning Questionnaire. Psychother Psychosom. 2001;70:221-225.
- Molina-Leyva A, Almodovar-Real A, Ruiz-Carrascosa JC, et al. Distribution pattern of psoriasis affects sexual function in moderate to severe psoriasis: a prospective case series study. J Sex Med. 2014;11:2882-2889.
- Guenther L, Han C, Szapary P, et al. Impact of ustekinumab on health-related quality of life and sexual difficulties associated with psoriasis: results from two phase III clinical trials. J Eur Acad Dermatol Venereol. 2011;25:851-857.
- Guenther L, Warren RB, Cather JC, et al. Impact of ixekizumab treatment on skin-related personal relationship difficulties in moderate-to-severe psoriasis patients: 12-week results from two Phase 3 trials. J Eur Acad Dermatol Venereol. 2017;31:1867-1875.
Practice Points
- The psychosocial impact of psoriasis is an important component of the disease burden leading to reduced quality of life.
- Assessment of psychosocial dysfunction can be done through short questionnaires, asking patients directly about these issues and anticipating these problems in patients who are most vulnerable.
- Management of psychosocial impact ranges from pharmacological interventions to helpful resources such as the National Psoriasis Foundation website.
Topical Corticosteroids for Treatment-Resistant Atopic Dermatitis
Atopic dermatitis (AD) is most often treated with mid-potency topical corticosteroids.1,2 Although this option is effective, not all patients respond to treatment, and those who do may lose efficacy over time, a phenomenon known as tachyphylaxis. The pathophysiology of tachyphylaxis to topical corticosteroids has been ascribed to loss of corticosteroid receptor function,3 but the evidence is weak.3,4 Patients with severe treatment-resistant AD improve when treated with mid-potency topical steroids in an inpatient setting; therefore, treatment resistance to topical corticosteroids may be largely due to poor adherence.5
Patients with treatment-resistant AD generally improve when treated with topical corticosteroids under conditions designed to promote treatment adherence, but this improvement often is reported for study groups, not individual patients. Focusing on group data may not give a clear picture of what is happening at the individual level. In this study, we evaluated changes at an individual level to determine how frequently AD patients who were previously treated with topical corticosteroids unsuccessfully would respond to desoximetasone spray 0.25% under conditions designed to promote good adherence over a 7-day period.
Methods
This open-label, randomized, single-center clinical study included 12 patients with AD who were previously unsuccessfully treated with topical corticosteroids in the Department of Dermatology at Wake Forest Baptist Medical Center (Winston-Salem, North Carolina)(Table 1). The study was approved by the local institutional review board.
Inclusion criteria included men and women 18 years or older at baseline who had AD that was considered amenable to therapy with topical corticosteroids by the clinician and were able to comply with the study protocol (Figure). Written informed consent also was obtained from each patient. Women who were pregnant, breastfeeding, or unwilling to practice birth control during participation in the study were excluded. Other exclusion criteria included presence of a condition that in the opinion of the investigator would compromise the safety of the patient or quality of data as well as patients with no access to a telephone throughout the day. Patients diagnosed with conditions affecting adherence to treatment (eg, dementia, Alzheimer disease), those with a history of allergy or sensitivity to corticosteroids, and those with a history of drug hypersensitivity were excluded from the study.
All 12 patients were treated with desoximetasone spray 0.25% for 7 days. Patients were instructed not to use other AD medications during the study period. At baseline, patients were randomized to receive either twice-daily telephone calls to discuss treatment adherence (intervention group) or no telephone calls (control) during the study period. Patients in both the intervention and control groups returned for evaluation on days 3 and 7. During these visits, disease severity was evaluated using the pruritus visual analog scale, Eczema Area and Severity Index (EASI), total lesion severity scale (TLSS), and investigator global assessment (IGA). Descriptive statistics were used to report the outcomes for each patient.
Results
Twelve AD patients who were previously unsuccessfully treated with topical corticosteroids were recruited for the study. Six patients were randomized to the intervention group and 6 were randomized to the control group. Fifty percent of patients were black, 50% were women, and the average age was 50.4 years. All 12 patients completed the study.
At the end of the study, most patients showed improvement in all evaluation parameters (eFigure). All 12 patients showed improvement in pruritus visual analog scores; 83.3% (10/12) showed improved EASI scores, 75.0% (9/12) showed improved TLSS scores, and 58.3% (7/12) showed improved IGA scores (Tables 2–5). Patients who received telephone calls in the intervention group showed greater improvement compared to those in the control group, except for pruritus; the mean reduction in pruritus was 76.9% in the intervention group versus 87.0% in the control group. The mean improvement in EASI score was 46.9% in the intervention group versus 21.1% in the control group. The mean improvement in TLSS score was 38.3% in the intervention group versus 9.7% in the control group. The mean improvement in IGA score was 45.8% in the intervention group versus 4.2% in the control group. Only one patient in the control group (patient 8) showed lower EASI, TLSS, and IGA scores at baseline.
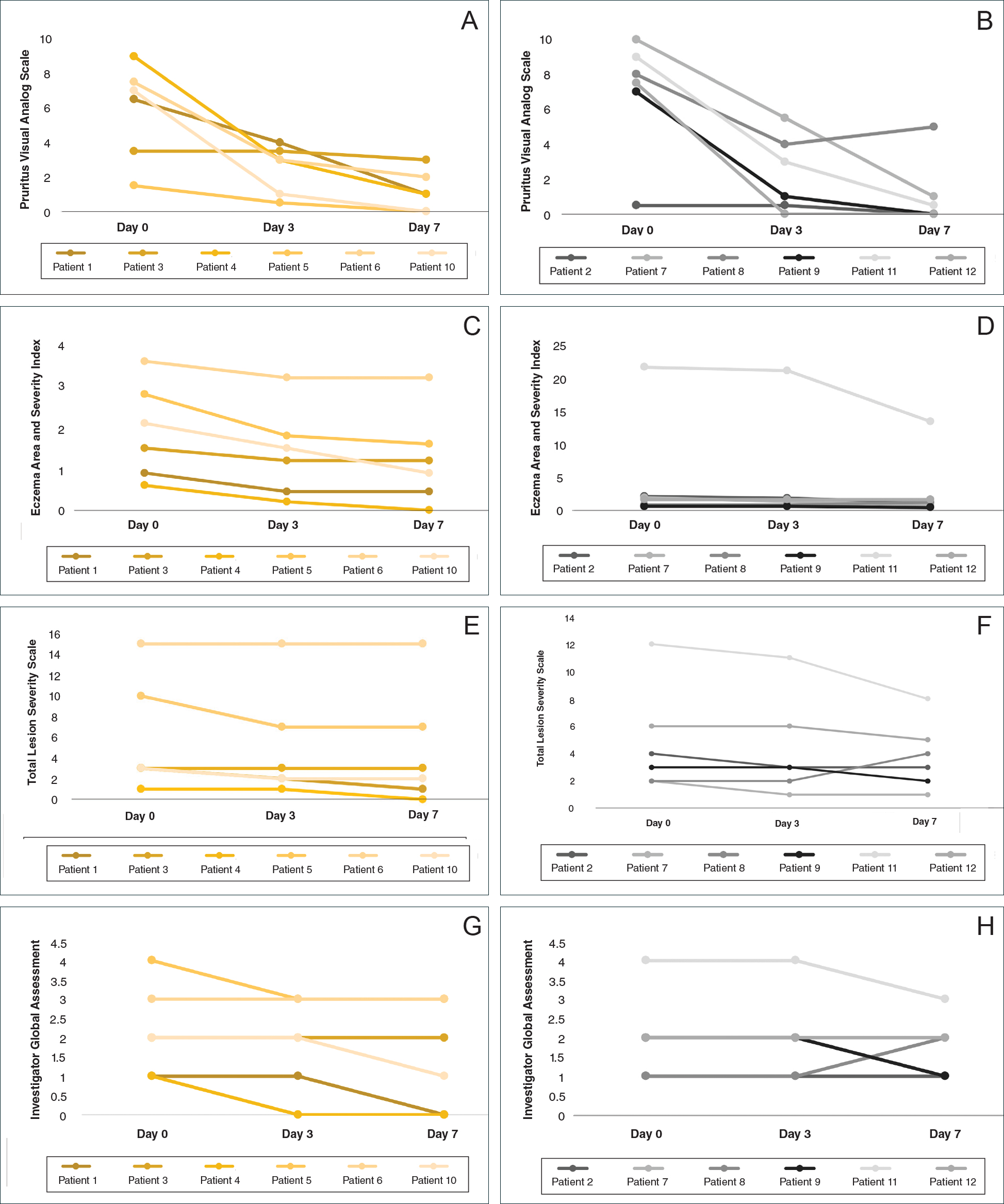
Comment
Although topical corticosteroids are the mainstay for treatment of AD, many patients report treatment resistance after a period of a few doses or longer.6-9 There is strong evidence demonstrating rapid corticosteroid receptor downregulation in tissues after corticosteroid therapy, which is the accepted mechanism for tachyphylaxis, but the timing of this effect does not match up with clinical experiences. The physiologic significance of corticosteroid agonist-induced receptor downregulation is unknown and may not have any considerable effect on corticosteroid efficacy.3 A systematic review by Taheri et al3 on the development of resistance to topical corticosteroids proposed 2 theories for the underlying pathogenesis of tachyphylaxis: (1) long-term patient nonadherence, and (2) the initial maximal response during the first few weeks of therapy eventually plateaus. Because corticosteroids may plateau after a certain number of doses, natural disease flare-ups during this period may give the wrong impression of tachyphylaxis.10 The treatment “resistance” reported by the patients in our study may have been due to this plateau effect or to poor adherence.
Our finding that nearly all patients had rapid improvement of AD with the topical corticosteroid is not definitive proof but supports the notion that tachyphylaxis is largely mediated by poor adherence to treatment. Patients rapidly improved over the short study period. The short duration of treatment and multiple visits over the study period were designed to help ensure patient adherence. Rapid improvement in AD when topical corticosteroids are used should be expected, as AD patients have rapid improvement with application of topical corticosteroids in inpatient settings.11,12
Poor adherence to topical medication is common. In a Danish study, 99 of 322 patients (31%) did not redeem their AD prescriptions.13 In a single-center, 5-day, prospective study evaluating the use of fluocinonide cream 0.1% for treatment of children and adults with AD, the median percentage of prescribed doses taken was 40%, according to objective electronic monitors, even though patients reported 100% adherence in their medication diaries.Better adherence was seen on day 1 of treatment in which 66.6% (6/9) of patients adhered to their treatment strategy versus day 5 in which only 11.1% (1/9) of patients used their medication.1
Topical corticosteroids are safe and efficacious if used appropriately; however, patients commonly express fear and anxiety about using them. Topical corticosteroid phobia may stem from a misconception that these products carry the same adverse effects as their oral and systemic counterparts, which may be perpetuated by the media.1 Of 200 dermatology patients surveyed, 72.5% expressed concern about using topical corticosteroids on themselves or their children’s skin, and 24% of these patients stated they were noncompliant with their medication because of these worries. Almost 50% of patients requested prescriptions for corticosteroid-sparing medications such as tacrolimus.1 Patient education is important to help ensure treatment adherence. Other factors that can affect treatment adherence include forgetfulness; the chronic nature of AD; the need for ongoing application of topical treatments; prohibitive costs of some topical agents; and complexities in coordinating school, work, and family plans with the treatment regimen.2
We attempted to ensure good treatment adherence in our study by calling the patients in the intervention group twice daily. The mean improvement in EASI, TLSS, and IGA scores was higher in the intervention group versus the control group, which suggests that patient reminders have at least some benefit. Because AD treatment resistance appears more closely tied to nonadherence rather than loss of medication efficacy, it seems prudent to focus on interventions that would improve treatment adherence; however, such interventions generally are not well tested. Recommended interventions have included educating patients about the side effects of topical corticosteroids, avoiding use of medical jargon, and taking patient vehicle preference into account when prescribing treatments.8 Patients should be scheduled for a return visit within 1 to 2 weeks, as early return visits can augment treatment adherence.14 At the return visit, there can be a more detailed discussion of long-term management and side effects.8
Limitations of our study included a small sample size and brief treatment duration. Even though the patients had previously reported treatment failure with topical corticosteroids, all demonstrated improvement in only 1 week with a potent topical corticosteroid. The treatment resistance that initially was reported likely was due to poor adherence, but it is possible for AD patients to be resistant to treatment with topical corticosteroids due to allergic contact dermatitis. Patients could theoretically be allergic to components of the vehicle used in topical corticosteroids, which could aggravate their dermatitis; however, this effect seems unlikely in our patient population, as all the patients in our study showed improvement following treatment. Another study limitation was that adherence was not measured. The frequent follow-up visits were designed to encourage treatment adherence, but adherence was not specifically assessed. Although patients were encouraged to only use the desoximetasone spray during the study, it is not known whether patients used other products.
Conclusion
Some AD patients exhibit apparent decreased efficacy of topical corticosteroids over time, but this tachyphylaxis phenomenon is more likely due to poor treatment adherence than to loss of corticosteroid responsiveness. In our study, AD patients who reported treatment failure with topical corticosteroids improved rapidly with topical corticosteroids under conditions designed to promote good adherence to treatment. The majority of patients improved in all 4 parameters used for evaluating disease severity, with 100% of patients reporting improvement in pruritus. Intervention to improve treatment adherence may lead to better health outcomes. When AD appears resistant to topical corticosteroids, addressing adherence issues may be critical.
- Patel NU, D’Ambra V, Feldman SR. Increasing adherence with topical agents for atopic dermatitis. Am J Clin Dermatol. 2017;18:323-332.
- Mooney E, Rademaker M, Dailey R, et al. Adverse effects of topical corticosteroids in paediatric eczema: Australasian consensus statement. Australas J Dermatol. 2015;56:241-251.
- Taheri A, Cantrell J, Feldman SR. Tachyphylaxis to topical glucocorticoids; what is the evidence? Dermatol Online J. 2013;19:18954.
- Miller JJ, Roling D, Margolis D, et al. Failure to demonstrate therapeutic tachyphylaxis to topically applied steroids in patients with psoriasis. J Am Acad Dermatol. 1999;41:546-549.
- Smith SD, Harris V, Lee A, et al. General practitioners knowledge about use of topical corticosteroids in paediatric atopic dermatitis in Australia. Aust Fam Physician. 2017;46:335-340.
- Sathishkumar D, Moss C. Topical therapy in atopic dermatitis in children. Indian J Dermatol. 2016;61:656-661.
- Reitamo S, Remitz A. Topical agents for atopic dermatitis. In: Bieber T, ed. Advances in the Management of Atopic Dermatitis. London, United Kingdom: Future Medicine Ltd; 2013:62-72.
- Krejci-Manwaring J, Tusa MG, Carroll C, et al. Stealth monitoring of adherence to topical medication: adherence is very poor in children with atopic dermatitis. J Am Acad Dermatol. 2007;56:211-216.
- Fukaya M. Cortisol homeostasis in the epidermis is influenced by topical corticosteroids in patients with atopic dermatitis. Indian J Dermatol. 2017;62:440.
- Mehta AB, Nadkarni NJ, Patil SP, et al. Topical corticosteroids in dermatology. Indian J Dermatol Venereol Leprol. 2016;82:371-378.
- van der Schaft J, Keijzer WW, Sanders KJ, et al. Is there an additional value of inpatient treatment for patients with atopic dermatitis? Acta Derm Venereol. 2016;96:797-801.
- Dabade TS, Davis DM, Wetter DA, et al. Wet dressing therapy in conjunction with topical corticosteroids is effective for rapid control of severe pediatric atopic dermatitis: experience with 218 patients over 30 years at Mayo Clinic. J Am Acad Dermatol. 2011;67:100-106.
- Storm A, Andersen SE, Benfeldt E, et al. One in 3 prescriptions are never redeemed: primary nonadherence in an outpatient clinic. J Am Acad Dermatol. 2008;59:27-33.
- Sagransky MJ, Yentzer BA, Williams LL, et al. A randomized controlled pilot study of the effects of an extra office visit on adherence and outcomes in atopic dermatitis. Arch Dermatol. 2010;146:1428-1430.
Atopic dermatitis (AD) is most often treated with mid-potency topical corticosteroids.1,2 Although this option is effective, not all patients respond to treatment, and those who do may lose efficacy over time, a phenomenon known as tachyphylaxis. The pathophysiology of tachyphylaxis to topical corticosteroids has been ascribed to loss of corticosteroid receptor function,3 but the evidence is weak.3,4 Patients with severe treatment-resistant AD improve when treated with mid-potency topical steroids in an inpatient setting; therefore, treatment resistance to topical corticosteroids may be largely due to poor adherence.5
Patients with treatment-resistant AD generally improve when treated with topical corticosteroids under conditions designed to promote treatment adherence, but this improvement often is reported for study groups, not individual patients. Focusing on group data may not give a clear picture of what is happening at the individual level. In this study, we evaluated changes at an individual level to determine how frequently AD patients who were previously treated with topical corticosteroids unsuccessfully would respond to desoximetasone spray 0.25% under conditions designed to promote good adherence over a 7-day period.
Methods
This open-label, randomized, single-center clinical study included 12 patients with AD who were previously unsuccessfully treated with topical corticosteroids in the Department of Dermatology at Wake Forest Baptist Medical Center (Winston-Salem, North Carolina)(Table 1). The study was approved by the local institutional review board.
Inclusion criteria included men and women 18 years or older at baseline who had AD that was considered amenable to therapy with topical corticosteroids by the clinician and were able to comply with the study protocol (Figure). Written informed consent also was obtained from each patient. Women who were pregnant, breastfeeding, or unwilling to practice birth control during participation in the study were excluded. Other exclusion criteria included presence of a condition that in the opinion of the investigator would compromise the safety of the patient or quality of data as well as patients with no access to a telephone throughout the day. Patients diagnosed with conditions affecting adherence to treatment (eg, dementia, Alzheimer disease), those with a history of allergy or sensitivity to corticosteroids, and those with a history of drug hypersensitivity were excluded from the study.
All 12 patients were treated with desoximetasone spray 0.25% for 7 days. Patients were instructed not to use other AD medications during the study period. At baseline, patients were randomized to receive either twice-daily telephone calls to discuss treatment adherence (intervention group) or no telephone calls (control) during the study period. Patients in both the intervention and control groups returned for evaluation on days 3 and 7. During these visits, disease severity was evaluated using the pruritus visual analog scale, Eczema Area and Severity Index (EASI), total lesion severity scale (TLSS), and investigator global assessment (IGA). Descriptive statistics were used to report the outcomes for each patient.
Results
Twelve AD patients who were previously unsuccessfully treated with topical corticosteroids were recruited for the study. Six patients were randomized to the intervention group and 6 were randomized to the control group. Fifty percent of patients were black, 50% were women, and the average age was 50.4 years. All 12 patients completed the study.
At the end of the study, most patients showed improvement in all evaluation parameters (eFigure). All 12 patients showed improvement in pruritus visual analog scores; 83.3% (10/12) showed improved EASI scores, 75.0% (9/12) showed improved TLSS scores, and 58.3% (7/12) showed improved IGA scores (Tables 2–5). Patients who received telephone calls in the intervention group showed greater improvement compared to those in the control group, except for pruritus; the mean reduction in pruritus was 76.9% in the intervention group versus 87.0% in the control group. The mean improvement in EASI score was 46.9% in the intervention group versus 21.1% in the control group. The mean improvement in TLSS score was 38.3% in the intervention group versus 9.7% in the control group. The mean improvement in IGA score was 45.8% in the intervention group versus 4.2% in the control group. Only one patient in the control group (patient 8) showed lower EASI, TLSS, and IGA scores at baseline.

Comment
Although topical corticosteroids are the mainstay for treatment of AD, many patients report treatment resistance after a period of a few doses or longer.6-9 There is strong evidence demonstrating rapid corticosteroid receptor downregulation in tissues after corticosteroid therapy, which is the accepted mechanism for tachyphylaxis, but the timing of this effect does not match up with clinical experiences. The physiologic significance of corticosteroid agonist-induced receptor downregulation is unknown and may not have any considerable effect on corticosteroid efficacy.3 A systematic review by Taheri et al3 on the development of resistance to topical corticosteroids proposed 2 theories for the underlying pathogenesis of tachyphylaxis: (1) long-term patient nonadherence, and (2) the initial maximal response during the first few weeks of therapy eventually plateaus. Because corticosteroids may plateau after a certain number of doses, natural disease flare-ups during this period may give the wrong impression of tachyphylaxis.10 The treatment “resistance” reported by the patients in our study may have been due to this plateau effect or to poor adherence.
Our finding that nearly all patients had rapid improvement of AD with the topical corticosteroid is not definitive proof but supports the notion that tachyphylaxis is largely mediated by poor adherence to treatment. Patients rapidly improved over the short study period. The short duration of treatment and multiple visits over the study period were designed to help ensure patient adherence. Rapid improvement in AD when topical corticosteroids are used should be expected, as AD patients have rapid improvement with application of topical corticosteroids in inpatient settings.11,12
Poor adherence to topical medication is common. In a Danish study, 99 of 322 patients (31%) did not redeem their AD prescriptions.13 In a single-center, 5-day, prospective study evaluating the use of fluocinonide cream 0.1% for treatment of children and adults with AD, the median percentage of prescribed doses taken was 40%, according to objective electronic monitors, even though patients reported 100% adherence in their medication diaries.Better adherence was seen on day 1 of treatment in which 66.6% (6/9) of patients adhered to their treatment strategy versus day 5 in which only 11.1% (1/9) of patients used their medication.1
Topical corticosteroids are safe and efficacious if used appropriately; however, patients commonly express fear and anxiety about using them. Topical corticosteroid phobia may stem from a misconception that these products carry the same adverse effects as their oral and systemic counterparts, which may be perpetuated by the media.1 Of 200 dermatology patients surveyed, 72.5% expressed concern about using topical corticosteroids on themselves or their children’s skin, and 24% of these patients stated they were noncompliant with their medication because of these worries. Almost 50% of patients requested prescriptions for corticosteroid-sparing medications such as tacrolimus.1 Patient education is important to help ensure treatment adherence. Other factors that can affect treatment adherence include forgetfulness; the chronic nature of AD; the need for ongoing application of topical treatments; prohibitive costs of some topical agents; and complexities in coordinating school, work, and family plans with the treatment regimen.2
We attempted to ensure good treatment adherence in our study by calling the patients in the intervention group twice daily. The mean improvement in EASI, TLSS, and IGA scores was higher in the intervention group versus the control group, which suggests that patient reminders have at least some benefit. Because AD treatment resistance appears more closely tied to nonadherence rather than loss of medication efficacy, it seems prudent to focus on interventions that would improve treatment adherence; however, such interventions generally are not well tested. Recommended interventions have included educating patients about the side effects of topical corticosteroids, avoiding use of medical jargon, and taking patient vehicle preference into account when prescribing treatments.8 Patients should be scheduled for a return visit within 1 to 2 weeks, as early return visits can augment treatment adherence.14 At the return visit, there can be a more detailed discussion of long-term management and side effects.8
Limitations of our study included a small sample size and brief treatment duration. Even though the patients had previously reported treatment failure with topical corticosteroids, all demonstrated improvement in only 1 week with a potent topical corticosteroid. The treatment resistance that initially was reported likely was due to poor adherence, but it is possible for AD patients to be resistant to treatment with topical corticosteroids due to allergic contact dermatitis. Patients could theoretically be allergic to components of the vehicle used in topical corticosteroids, which could aggravate their dermatitis; however, this effect seems unlikely in our patient population, as all the patients in our study showed improvement following treatment. Another study limitation was that adherence was not measured. The frequent follow-up visits were designed to encourage treatment adherence, but adherence was not specifically assessed. Although patients were encouraged to only use the desoximetasone spray during the study, it is not known whether patients used other products.
Conclusion
Some AD patients exhibit apparent decreased efficacy of topical corticosteroids over time, but this tachyphylaxis phenomenon is more likely due to poor treatment adherence than to loss of corticosteroid responsiveness. In our study, AD patients who reported treatment failure with topical corticosteroids improved rapidly with topical corticosteroids under conditions designed to promote good adherence to treatment. The majority of patients improved in all 4 parameters used for evaluating disease severity, with 100% of patients reporting improvement in pruritus. Intervention to improve treatment adherence may lead to better health outcomes. When AD appears resistant to topical corticosteroids, addressing adherence issues may be critical.
Atopic dermatitis (AD) is most often treated with mid-potency topical corticosteroids.1,2 Although this option is effective, not all patients respond to treatment, and those who do may lose efficacy over time, a phenomenon known as tachyphylaxis. The pathophysiology of tachyphylaxis to topical corticosteroids has been ascribed to loss of corticosteroid receptor function,3 but the evidence is weak.3,4 Patients with severe treatment-resistant AD improve when treated with mid-potency topical steroids in an inpatient setting; therefore, treatment resistance to topical corticosteroids may be largely due to poor adherence.5
Patients with treatment-resistant AD generally improve when treated with topical corticosteroids under conditions designed to promote treatment adherence, but this improvement often is reported for study groups, not individual patients. Focusing on group data may not give a clear picture of what is happening at the individual level. In this study, we evaluated changes at an individual level to determine how frequently AD patients who were previously treated with topical corticosteroids unsuccessfully would respond to desoximetasone spray 0.25% under conditions designed to promote good adherence over a 7-day period.
Methods
This open-label, randomized, single-center clinical study included 12 patients with AD who were previously unsuccessfully treated with topical corticosteroids in the Department of Dermatology at Wake Forest Baptist Medical Center (Winston-Salem, North Carolina)(Table 1). The study was approved by the local institutional review board.
Inclusion criteria included men and women 18 years or older at baseline who had AD that was considered amenable to therapy with topical corticosteroids by the clinician and were able to comply with the study protocol (Figure). Written informed consent also was obtained from each patient. Women who were pregnant, breastfeeding, or unwilling to practice birth control during participation in the study were excluded. Other exclusion criteria included presence of a condition that in the opinion of the investigator would compromise the safety of the patient or quality of data as well as patients with no access to a telephone throughout the day. Patients diagnosed with conditions affecting adherence to treatment (eg, dementia, Alzheimer disease), those with a history of allergy or sensitivity to corticosteroids, and those with a history of drug hypersensitivity were excluded from the study.
All 12 patients were treated with desoximetasone spray 0.25% for 7 days. Patients were instructed not to use other AD medications during the study period. At baseline, patients were randomized to receive either twice-daily telephone calls to discuss treatment adherence (intervention group) or no telephone calls (control) during the study period. Patients in both the intervention and control groups returned for evaluation on days 3 and 7. During these visits, disease severity was evaluated using the pruritus visual analog scale, Eczema Area and Severity Index (EASI), total lesion severity scale (TLSS), and investigator global assessment (IGA). Descriptive statistics were used to report the outcomes for each patient.
Results
Twelve AD patients who were previously unsuccessfully treated with topical corticosteroids were recruited for the study. Six patients were randomized to the intervention group and 6 were randomized to the control group. Fifty percent of patients were black, 50% were women, and the average age was 50.4 years. All 12 patients completed the study.
At the end of the study, most patients showed improvement in all evaluation parameters (eFigure). All 12 patients showed improvement in pruritus visual analog scores; 83.3% (10/12) showed improved EASI scores, 75.0% (9/12) showed improved TLSS scores, and 58.3% (7/12) showed improved IGA scores (Tables 2–5). Patients who received telephone calls in the intervention group showed greater improvement compared to those in the control group, except for pruritus; the mean reduction in pruritus was 76.9% in the intervention group versus 87.0% in the control group. The mean improvement in EASI score was 46.9% in the intervention group versus 21.1% in the control group. The mean improvement in TLSS score was 38.3% in the intervention group versus 9.7% in the control group. The mean improvement in IGA score was 45.8% in the intervention group versus 4.2% in the control group. Only one patient in the control group (patient 8) showed lower EASI, TLSS, and IGA scores at baseline.

Comment
Although topical corticosteroids are the mainstay for treatment of AD, many patients report treatment resistance after a period of a few doses or longer.6-9 There is strong evidence demonstrating rapid corticosteroid receptor downregulation in tissues after corticosteroid therapy, which is the accepted mechanism for tachyphylaxis, but the timing of this effect does not match up with clinical experiences. The physiologic significance of corticosteroid agonist-induced receptor downregulation is unknown and may not have any considerable effect on corticosteroid efficacy.3 A systematic review by Taheri et al3 on the development of resistance to topical corticosteroids proposed 2 theories for the underlying pathogenesis of tachyphylaxis: (1) long-term patient nonadherence, and (2) the initial maximal response during the first few weeks of therapy eventually plateaus. Because corticosteroids may plateau after a certain number of doses, natural disease flare-ups during this period may give the wrong impression of tachyphylaxis.10 The treatment “resistance” reported by the patients in our study may have been due to this plateau effect or to poor adherence.
Our finding that nearly all patients had rapid improvement of AD with the topical corticosteroid is not definitive proof but supports the notion that tachyphylaxis is largely mediated by poor adherence to treatment. Patients rapidly improved over the short study period. The short duration of treatment and multiple visits over the study period were designed to help ensure patient adherence. Rapid improvement in AD when topical corticosteroids are used should be expected, as AD patients have rapid improvement with application of topical corticosteroids in inpatient settings.11,12
Poor adherence to topical medication is common. In a Danish study, 99 of 322 patients (31%) did not redeem their AD prescriptions.13 In a single-center, 5-day, prospective study evaluating the use of fluocinonide cream 0.1% for treatment of children and adults with AD, the median percentage of prescribed doses taken was 40%, according to objective electronic monitors, even though patients reported 100% adherence in their medication diaries.Better adherence was seen on day 1 of treatment in which 66.6% (6/9) of patients adhered to their treatment strategy versus day 5 in which only 11.1% (1/9) of patients used their medication.1
Topical corticosteroids are safe and efficacious if used appropriately; however, patients commonly express fear and anxiety about using them. Topical corticosteroid phobia may stem from a misconception that these products carry the same adverse effects as their oral and systemic counterparts, which may be perpetuated by the media.1 Of 200 dermatology patients surveyed, 72.5% expressed concern about using topical corticosteroids on themselves or their children’s skin, and 24% of these patients stated they were noncompliant with their medication because of these worries. Almost 50% of patients requested prescriptions for corticosteroid-sparing medications such as tacrolimus.1 Patient education is important to help ensure treatment adherence. Other factors that can affect treatment adherence include forgetfulness; the chronic nature of AD; the need for ongoing application of topical treatments; prohibitive costs of some topical agents; and complexities in coordinating school, work, and family plans with the treatment regimen.2
We attempted to ensure good treatment adherence in our study by calling the patients in the intervention group twice daily. The mean improvement in EASI, TLSS, and IGA scores was higher in the intervention group versus the control group, which suggests that patient reminders have at least some benefit. Because AD treatment resistance appears more closely tied to nonadherence rather than loss of medication efficacy, it seems prudent to focus on interventions that would improve treatment adherence; however, such interventions generally are not well tested. Recommended interventions have included educating patients about the side effects of topical corticosteroids, avoiding use of medical jargon, and taking patient vehicle preference into account when prescribing treatments.8 Patients should be scheduled for a return visit within 1 to 2 weeks, as early return visits can augment treatment adherence.14 At the return visit, there can be a more detailed discussion of long-term management and side effects.8
Limitations of our study included a small sample size and brief treatment duration. Even though the patients had previously reported treatment failure with topical corticosteroids, all demonstrated improvement in only 1 week with a potent topical corticosteroid. The treatment resistance that initially was reported likely was due to poor adherence, but it is possible for AD patients to be resistant to treatment with topical corticosteroids due to allergic contact dermatitis. Patients could theoretically be allergic to components of the vehicle used in topical corticosteroids, which could aggravate their dermatitis; however, this effect seems unlikely in our patient population, as all the patients in our study showed improvement following treatment. Another study limitation was that adherence was not measured. The frequent follow-up visits were designed to encourage treatment adherence, but adherence was not specifically assessed. Although patients were encouraged to only use the desoximetasone spray during the study, it is not known whether patients used other products.
Conclusion
Some AD patients exhibit apparent decreased efficacy of topical corticosteroids over time, but this tachyphylaxis phenomenon is more likely due to poor treatment adherence than to loss of corticosteroid responsiveness. In our study, AD patients who reported treatment failure with topical corticosteroids improved rapidly with topical corticosteroids under conditions designed to promote good adherence to treatment. The majority of patients improved in all 4 parameters used for evaluating disease severity, with 100% of patients reporting improvement in pruritus. Intervention to improve treatment adherence may lead to better health outcomes. When AD appears resistant to topical corticosteroids, addressing adherence issues may be critical.
- Patel NU, D’Ambra V, Feldman SR. Increasing adherence with topical agents for atopic dermatitis. Am J Clin Dermatol. 2017;18:323-332.
- Mooney E, Rademaker M, Dailey R, et al. Adverse effects of topical corticosteroids in paediatric eczema: Australasian consensus statement. Australas J Dermatol. 2015;56:241-251.
- Taheri A, Cantrell J, Feldman SR. Tachyphylaxis to topical glucocorticoids; what is the evidence? Dermatol Online J. 2013;19:18954.
- Miller JJ, Roling D, Margolis D, et al. Failure to demonstrate therapeutic tachyphylaxis to topically applied steroids in patients with psoriasis. J Am Acad Dermatol. 1999;41:546-549.
- Smith SD, Harris V, Lee A, et al. General practitioners knowledge about use of topical corticosteroids in paediatric atopic dermatitis in Australia. Aust Fam Physician. 2017;46:335-340.
- Sathishkumar D, Moss C. Topical therapy in atopic dermatitis in children. Indian J Dermatol. 2016;61:656-661.
- Reitamo S, Remitz A. Topical agents for atopic dermatitis. In: Bieber T, ed. Advances in the Management of Atopic Dermatitis. London, United Kingdom: Future Medicine Ltd; 2013:62-72.
- Krejci-Manwaring J, Tusa MG, Carroll C, et al. Stealth monitoring of adherence to topical medication: adherence is very poor in children with atopic dermatitis. J Am Acad Dermatol. 2007;56:211-216.
- Fukaya M. Cortisol homeostasis in the epidermis is influenced by topical corticosteroids in patients with atopic dermatitis. Indian J Dermatol. 2017;62:440.
- Mehta AB, Nadkarni NJ, Patil SP, et al. Topical corticosteroids in dermatology. Indian J Dermatol Venereol Leprol. 2016;82:371-378.
- van der Schaft J, Keijzer WW, Sanders KJ, et al. Is there an additional value of inpatient treatment for patients with atopic dermatitis? Acta Derm Venereol. 2016;96:797-801.
- Dabade TS, Davis DM, Wetter DA, et al. Wet dressing therapy in conjunction with topical corticosteroids is effective for rapid control of severe pediatric atopic dermatitis: experience with 218 patients over 30 years at Mayo Clinic. J Am Acad Dermatol. 2011;67:100-106.
- Storm A, Andersen SE, Benfeldt E, et al. One in 3 prescriptions are never redeemed: primary nonadherence in an outpatient clinic. J Am Acad Dermatol. 2008;59:27-33.
- Sagransky MJ, Yentzer BA, Williams LL, et al. A randomized controlled pilot study of the effects of an extra office visit on adherence and outcomes in atopic dermatitis. Arch Dermatol. 2010;146:1428-1430.
- Patel NU, D’Ambra V, Feldman SR. Increasing adherence with topical agents for atopic dermatitis. Am J Clin Dermatol. 2017;18:323-332.
- Mooney E, Rademaker M, Dailey R, et al. Adverse effects of topical corticosteroids in paediatric eczema: Australasian consensus statement. Australas J Dermatol. 2015;56:241-251.
- Taheri A, Cantrell J, Feldman SR. Tachyphylaxis to topical glucocorticoids; what is the evidence? Dermatol Online J. 2013;19:18954.
- Miller JJ, Roling D, Margolis D, et al. Failure to demonstrate therapeutic tachyphylaxis to topically applied steroids in patients with psoriasis. J Am Acad Dermatol. 1999;41:546-549.
- Smith SD, Harris V, Lee A, et al. General practitioners knowledge about use of topical corticosteroids in paediatric atopic dermatitis in Australia. Aust Fam Physician. 2017;46:335-340.
- Sathishkumar D, Moss C. Topical therapy in atopic dermatitis in children. Indian J Dermatol. 2016;61:656-661.
- Reitamo S, Remitz A. Topical agents for atopic dermatitis. In: Bieber T, ed. Advances in the Management of Atopic Dermatitis. London, United Kingdom: Future Medicine Ltd; 2013:62-72.
- Krejci-Manwaring J, Tusa MG, Carroll C, et al. Stealth monitoring of adherence to topical medication: adherence is very poor in children with atopic dermatitis. J Am Acad Dermatol. 2007;56:211-216.
- Fukaya M. Cortisol homeostasis in the epidermis is influenced by topical corticosteroids in patients with atopic dermatitis. Indian J Dermatol. 2017;62:440.
- Mehta AB, Nadkarni NJ, Patil SP, et al. Topical corticosteroids in dermatology. Indian J Dermatol Venereol Leprol. 2016;82:371-378.
- van der Schaft J, Keijzer WW, Sanders KJ, et al. Is there an additional value of inpatient treatment for patients with atopic dermatitis? Acta Derm Venereol. 2016;96:797-801.
- Dabade TS, Davis DM, Wetter DA, et al. Wet dressing therapy in conjunction with topical corticosteroids is effective for rapid control of severe pediatric atopic dermatitis: experience with 218 patients over 30 years at Mayo Clinic. J Am Acad Dermatol. 2011;67:100-106.
- Storm A, Andersen SE, Benfeldt E, et al. One in 3 prescriptions are never redeemed: primary nonadherence in an outpatient clinic. J Am Acad Dermatol. 2008;59:27-33.
- Sagransky MJ, Yentzer BA, Williams LL, et al. A randomized controlled pilot study of the effects of an extra office visit on adherence and outcomes in atopic dermatitis. Arch Dermatol. 2010;146:1428-1430.
Practice Points
- Mid-potency corticosteroids are the first-line treatment of atopic dermatitis (AD).
- Atopic dermatitis may fail to respond to topical corticosteroids initially or lose response over time, a phenomenon known as tachyphylaxis.
- Nonadherence to medication is the most likely cause of treatment resistance in patients with AD.
No Sulfates, No Parabens, and the “No-Poo” Method: A New Patient Perspective on Common Shampoo Ingredients
Shampoo is a staple in hair grooming that is ever-evolving along with cultural trends. The global shampoo market is expected to reach an estimated value of $25.73 billion by 2019. A major driver of this upward trend in market growth is the increasing demand for natural and organic hair shampoos.1 Society today has a growing fixation on healthy living practices, and as of late, the ingredients in shampoos and other cosmetic products have become one of the latest targets in the health-consciousness craze. In the age of the Internet where information—and misinformation—is widely accessible and dispersed, the general public often strives to self-educate on specialized matters that are out of their expertise. As a result, individuals have developed an aversion to using certain shampoos out of fear that the ingredients, often referred to as “chemicals” by patients due to their complex names, are unnatural and therefore unhealthy.1,2 Product developers are working to meet the demand by reformulating shampoos with labels that indicate sulfate free or paraben free, despite the lack of proof that these formulations are an improvement over traditional approaches to hair health. Additionally, alternative methods of cleansing the hair and scalp, also known as the no-shampoo or “no-poo” method, have begun to gain popularity.2,3
It is essential that dermatologists acknowledge the concerns that their patients have about common shampoo ingredients to dispel the myths that may misinform patient decision-making. This article reviews the controversy surrounding the use of sulfates and parabens in shampoos as well as commonly used shampoo alternatives. Due to the increased prevalence of dry hair shafts in the skin of color population, especially black women, this group is particularly interested in products that will minimize breakage and dryness of the hair. To that end, this population has great interest in the removal of chemical ingredients that may cause damage to the hair shafts, despite the lack of data to support sulfates and paraben damage to hair shafts or scalp skin. Blogs and uninformed hairstylists may propagate these beliefs in a group of consumers who are desperate for new approaches to hair fragility and breakage.
Surfactants and Sulfates
The cleansing ability of a shampoo depends on the surface activity of its detergents. Surface-active ingredients, or surfactants, reduce the surface tension between water and dirt, thus facilitating the removal of environmental dirt from the hair and scalp,4 which is achieved by a molecular structure containing both a hydrophilic and a lipophilic group. Sebum and dirt are bound by the lipophilic ends of the surfactant, becoming the center of a micelle structure with the hydrophilic molecule ends pointing outward. Dirt particles become water soluble and are removed from the scalp and hair shaft upon rinsing with water.4
Surfactants are classified according to the electric charge of the hydrophilic polar group as either anionic, cationic, amphoteric (zwitterionic), or nonionic.5 Each possesses different hair conditioning and cleansing qualities, and multiple surfactants are used in shampoos in differing ratios to accommodate different hair types. In most shampoos, the base consists of anionic and amphoteric surfactants. Depending on individual product requirements, nonionic and cationic surfactants are used to either modify the effects of the surfactants or as conditioning agents.4,5
One subcategory of surfactants that receives much attention is the group of anionic surfactants known as sulfates. Sulfates, particularly sodium lauryl sulfate (SLS), recently have developed a negative reputation as cosmetic ingredients, as reports from various unscientific sources have labeled them as hazardous to one’s health; SLS has been described as a skin and scalp irritant, has been linked to cataract formation, and has even been wrongly labeled as carcinogenic.6 The origins of some of these claims are not clear, though they likely arose from the misinterpretation of complex scientific studies that are easily accessible to laypeople. The link between SLS and ocular irritation or cataract formation is a good illustration of this unsubstantiated fear. A study by Green et al7 showed that corneal exposure to extremely high concentrations of SLS following physical or chemical damage to the eye can result in a slowed healing process. The results of this study have since been wrongly quoted to state that SLS-containing products lead to blindness or severe corneal damage.8 A different study tested for possible ocular irritation in vivo by submerging the lens of an eye into a 20% SLS solution, which accurately approximates the concentration of SLS in rinse-off consumer products.9 However, to achieve ocular irritation, the eyes of laboratory animals were exposed to SLS constantly for 14 days, which would not occur in practical use.9 Similarly, a third study achieved cataract formation in a laboratory only by immersing the lens of an eye into a highly concentrated solution of SLS.10 Such studies are not appropriate representations of how SLS-containing products are used by consumers and have unfortunately been vulnerable to misinterpretation by the general public.
There is no known study that has shown SLS to be carcinogenic. One possible origin of this idea may be from the wrongful interpretation of studies that used SLS as a vehicle substance to test agents that were deemed to be carcinogenic.11 Another possible source of the idea that SLS is carcinogenic comes from its association with 1,4-dioxane, a by-product of the synthesis of certain sulfates such as sodium laureth sulfate due to a process known as ethoxylation.6,12 Although SLS does not undergo this process in its formation and is not linked to 1,4-dioxane, there is potential for cross-contamination of SLS with 1,4-dioxane, which cannot be overlooked. 1,4-Dioxane is classified as “possibly carcinogenic to humans (Group 2B)” by the International Agency for Research on Cancer,13 but screening of SLS for this substance prior to its use in commercial products is standard.
Sulfates are inexpensive detergents that are responsible for lather formation in shampoos as well as in many household cleaning agents.5 Sulfates, similar to all anionic surfactants, are characterized by a negatively charged hydrophilic polar group. The best-known and most commonly used anionic surfactants are sulfated fatty alcohols, alkyl sulfates, and their polyethoxylated analogues alkyl ether sulfates.5,6 Sodium lauryl sulfate (also known as sodium laurilsulfate or sodium dodecyl sulfate) is the most common of them all, found in shampoo and conditioner formulations. Ammonium lauryl sulfate and sodium laureth sulfate are other sulfates commonly used in shampoos and household cleansing products. Sodium lauryl sulfate is a nonvolatile, water-soluble compound. Its partition coefficient (P0), a measure of a substance’s hydrophilic or lipophilic nature, is low at 1.6, making it a rather hydrophilic substance.6 Hydrophilic substances tend to have low bioaccumulation profiles in the body. Additionally, SLS is readily biodegradable. It can be derived from both synthetic and naturally occurring sources; for example, palm kernel oil, petrolatum, and coconut oil are all sources of lauric acid, the starting ingredient used to synthesize SLS. Sodium lauryl sulfate is created by reacting lauryl alcohol with sulfur trioxide gas, followed by neutralization with sodium carbonate (also a naturally occurring compound).6 Sodium lauryl sulfate and other sulfate-containing shampoos widely replaced the usage of traditional soaps formulated from animal or vegetable fats, as these latter formations created a film of insoluble calcium salts on the hair strands upon contact with water, resulting in tangled, dull-appearing hair.5 Additionally, sulfates were preferred to the alkaline pH of traditional soap, which can be harsh on hair strands and cause irritation of the skin and mucous membranes.14 Because they are highly water soluble, sulfates enable the formulation of clear shampoos. They exhibit remarkable cleaning properties and lather formation.5,14
Because sulfates are potent surfactants, they can remove dirt and debris as well as naturally produced healthy oils from the hair and scalp. As a result, sulfates can leave the hair feeling dry and stripped of moisture.4,5 Sulfates are used as the primary detergents in the formulation of deep-cleaning shampoos, which are designed for people who accumulate a heavy buildup of dirt, sebum, and debris from frequent use of styling products. Due to their potent detergency, these shampoos typically are not used on a daily basis but rather at longer intervals.15 A downside to sulfates is that they can have cosmetically unpleasant properties, which can be compensated for by including appropriate softening additives in shampoo formulations.4 A number of anionic surfactants such as olefin sulfonate, alkyl sulfosuccinate, acyl peptides, and alkyl ether carboxylates are well tolerated by the skin and are used together with other anionic and amphoteric surfactants to optimize shampoo properties. Alternatively, sulfate-free shampoos are cleansers compounded by the removal of the anionic group and switched for surfactants with less detergency.4,5
Preservatives and Parabens
Parabens refer to a group of esters of 4-hydroxybenzoic acid commonly used as preservatives in foods, pharmaceuticals, and cosmetics whose widespread use dates back to 1923.16 Concerns over the presence of parabens in shampoos and other cosmetics have been raised by patients for their reputed estrogenic and antiandrogenic effects and suspected involvement in carcinogenesis via endocrine modulation.16,17 In in vitro studies done on yeast assays, parabens have shown weak estrogenic activity that increases in proportion to both the length and increased branching of the alkyl side chains in the paraben’s molecular structure.18 They are 10,000-fold less potent than 17β-estradiol. In in vivo animal studies, parabens show weak estrogenic activity and are 100,000-fold less potent than 17β-estradiol.18 4-Hydroxybenzoic acid, a common metabolite, showed no estrogenic activity when tested both in vitro and in vivo.19 Some concerning research has implicated a link between parabens used in underarm cosmetics, such as deodorants and antiperspirants, and breast cancer16; however, the studies have been conflicting, and there is simply not enough data to assert that parabens cause breast cancer.
The Cosmetic Ingredient Review expert panel first reviewed parabens in 1984 and concluded that “methylparaben, ethylparaben, propylparaben, and butylparaben are safe as cosmetic ingredients in the present practices of use.”20 They extended this statement to include isopropylparaben and isobutylparaben in a later review.21 In 2005, the Scientific Committee on Consumer Products (now known as the Scientific Committee for Consumer Safety) in Europe stated that methylparaben and ethylparaben can be used at levels up to 0.4% in products.22 This decision was reached due to reports of decreased sperm counts and testosterone levels in male juvenile rats exposed to these parabens; however, these reults were not successfully replicated in larger studies.16,22 In 2010, the Scientific Committee for Consumer Safety revisited its stance on parabens, and they then revised their recommendations to say that concentrations of propylparaben and butylparaben should not exceed concentrations of 0.19%, based on “the conservative choice for the calculation of the [Margin-of-Safety] of butyl- and propylparaben.”23 However, in 2011 the use of propylparaben and butylparaben was banned in Denmark for cosmetic products used in children 3 years or younger,16 and the European Commission subsequently amended their directive in 2014, banning isopropylparaben, isobutylparaben, phenylparaben, benzylparaben, and pentylparaben due to lack of data available to evaluate the human risk of these products.24
Contrary to the trends in Europe, there currently are no regulations against the use of parabens in shampoos or other cosmetics in the United States. The American Cancer Society found that there is no evidence to suggest that the current levels of parabens in cosmetic products (eg, antiperspirants) increase one’s risk of breast cancer.25 Parabens are readily absorbed into the body both transdermally and through ingestion but also are believed to be rapidly transformed into harmless and nonspecific metabolites; they are readily metabolized by the liver and excreted in urine, and there is no measured accumulation in tissues.17
Parabens continue to be the most widely used preservatives in personal care products, usually in conjunction with other preservatives. Parabens are good biocides; short-chain esters (eg, methylparabens, ethylparabens) are effective against gram-positive bacteria and are weakly effective against gram-negative bacteria. Long-chain paraben esters (eg, propylparabens, butylparabens) are effective against mold and yeast. The addition of other preservatives creates a broad spectrum of antimicrobial defense in consumer products. Other preservatives include formaldehyde releasers or phenoxyethanol, as well as chelating agents such as EDTA, which improve the stability of these cosmetic products when exposed to air.16 Parabens are naturally occurring substances found in foods such as blueberries, barley, strawberries, yeast, olives, and grapes. As a colorless, odorless, and inexpensive substance, their use has been heavily favored in cosmetic and food products.16
Shampoo Alternatives and the No-Poo Method
Although research has not demonstrated any long-term danger to using shampoo, certain chemicals found in shampoos have the potential to irritate the scalp. Commonly cited allergens in shampoos include cocamidopropyl betaine, propylene glycol, vitamin E (tocopherol), parabens, and benzophenones.5 Additionally, the rising use of formaldehyde-releasing preservatives and isothiazolinones due to mounting pressures to move away from parabens has led to an increase in cases of allergic contact dermatitis (ACD).16 However, the irritability (rather than allergenicity) of these substances often is established during patch testing, a method of detecting delayed-type allergic reactions, which is important to note because patch testing requires a substance to be exposed to the skin for 24 to 48 hours, whereas exposure to shampoo ingredients may last a matter of minutes at most and occur in lesser concentrations because the ingredients are diluted by water in the rinsing process. Given these differences, it is unlikely that a patient would develop a true allergic response from regular shampoo use. Nevertheless, in patients who are already sensitized, exposure could conceivably trigger ACD, and patients must be cognizant of the composition of their shampoos.16
The no-poo method refers to the avoidance of commercial shampoo products when cleansing the hair and scalp and encompasses different methods of cleansing the hair, such as the use of household items (eg, baking soda, apple cider vinegar [ACV]), the use of conditioners to wash the hair (also known as conditioner-only washing or co-washing), treating the scalp with tea tree oil, or simply rinsing the hair with water. Proponents of the no-poo method believe that abstaining from shampoo use leads to healthier hair, retained natural oils, and less exposure to supposedly dangerous chemicals such as parabens or sulfates.2,3,26-28 However, there are no known studies in the literature that assess or support the hypotheses of the no-poo method.
Baking Soda and ACV
Baking soda (sodium bicarbonate) is a substance commonly found in the average household. It has been used in toothpaste formulas and cosmetic products and is known for its acid-neutralizing properties. Baking soda has been shown to have some antifungal and viricidal properties through an unknown mechanism of action.28 It has gained popularity for its use as a means of reducing the appearance of excessive greasiness of the hair shafts. Users also have reported that when washing their hair with baking soda, they are able to achieve a clean scalp and hair that feels soft to the touch.2,3,26,27,29 Despite these reports, users must beware of using baking soda without adequately diluting it with water. Baking soda is a known alkaline irritant.26,30 With a pH of 9, baking soda causes the cuticle layer of the hair fiber to open, increasing the capacity for water absorption. Water penetrates the scales that open, breaking the hydrogen bonds of the keratin molecule.31 Keratin is a spiral helical molecule that keeps its shape due to hydrogen, disulfide, and ionic bonds, as well as Van der Waals force.30 Hydrolysis of these bonds due to exposure to baking soda lowers the elasticity of the hair and increases the negative electrical net charge of the hair fiber surface, which leads to increased friction between fibers, cuticle damage, hair fragility, and fiber breakage.32,33
Apple cider vinegar is an apple-derived acetic acid solution with a pH ranging from 3.1 to 5.28 The pH range of ACV is considered to be ideal for hair by no-poo proponents, as it is similar to the natural pH of the scalp. Its acidic properties are responsible for its antimicrobial abilities, particularly its effectiveness against gram-negative bacteria.30 The acetic acid of ACV can partially interrupt oil interfaces, which contributes to its mild ability to remove product residue and scalp buildup from the hair shaft; the acetic acid also tightens the cuticles on hair fibers.33 Apple cider vinegar is used as a means of cleansing the hair and scalp by no-poo proponents2,3,26; other uses for ACV include using it as a rinse following washing and/or conditioning of the hair or as a means of preserving color in color-treated hair. There also is evidence that ACV may have antifungal properties.28 However, consumers must be aware that if it is not diluted in water, ACV may be too caustic for direct application to the hair and may lead to damage; it can be irritating to eyes, mucus membranes, and acutely inflamed skin. Also, vinegar rinses used on processed or chemically damaged hair may lead to increased hair fragility.2,3
Hair fibers have a pH of 3.67, while the scalp has a pH between 4.5 and 6.2. This slightly acidic film acts as a barrier to viruses, bacteria, and other potential contaminants.33 Studies have shown that the pH of skin increases in proportion to the pH of the cleanser used.34 Therefore, due to the naturally acidic pH of the scalp, acid-balanced shampoos generally are recommended. Shampoos should not have a pH higher than 5.5, as hair shafts can swell due to alkalinization, which can be prevented by pH balancing the shampoo through the addition of an acidic substance (eg, glycolic acid, citric acid) to lower the pH down to approximately 5.5. Apple cider vinegar often is used for this purpose. However, one study revealed that 82% of shampoos already have an acidic pH.34
Conditioner-Only Washing (Co-washing)
Conditioner-only washing, or co-washing, is a widely practiced method of hair grooming. It is popular among individuals who find that commercial shampoos strip too much of the natural hair oils away, leaving the hair rough or unmanageable. Co-washing is not harmful to the hair; however, the molecular structure and function of a conditioner and that of a shampoo are very different.5,35,36 Conditioners are not formulated to remove dirt and buildup in the hair but rather to add substances to the hair, and thus cannot provide extensive cleansing of the hair and scalp; therefore, it is inappropriate to use co-washing as a replacement for shampooing. Quaternary conditioning agents are an exception because they contain amphoteric detergents comprised of both anionic and cationic groups, which allow them both the ability to remove dirt and sebum with its anionic group, typically found in shampoos, as well as the ability to coat and condition the hair due to the high affinity of the cationic group for the negatively charged hair fibers.36,37 Amphoteric detergents are commonly found in 2-in-1 conditioning cleansers, among other ingredients, such as hydrolyzed animal proteins that temporarily plug surface defects on the hair fiber, and dimethicone, a synthetic oil that creates a thin film over the hair shaft, increasing shine and manageability. Of note, these conditioning shampoos are ideal for individuals with minimal product buildup on the hair and scalp and are not adequate scalp cleansers for individuals who either wash their hair infrequently or who regularly use hairstyling products.36,37
Tea Tree Oil
Tea tree oil is an essential oil extracted from the Melaleuca alternifolia plant of the Myrtaceae family. It is native to the coast of northeastern Australia. A holy grail of natural cosmetics, tea tree oil is widely known for its antiviral, antifungal, and antiseptic properties.38 Although not used as a stand-alone cleanser, it is often added to a number of cosmetic products, including shampoos and co-washes. Although deemed safe for topical use, it has been shown to be quite toxic when ingested. Symptoms of ingestion include nausea, vomiting, hallucinations, and coma. The common concern with tea tree oil is its ability to cause ACD. In particular, it is believed that the oxidation products of tea tree oil are allergenic rather than the tea tree oil itself. The evaluation of tea tree oil as a potential contact allergen has been quite difficult; it consists of more than 100 distinct compounds and is often mislabeled, or does not meet the guidelines of the International Organization for Standardization. Nonetheless, the prevalence of ACD due to tea tree oil is low (approximately 1.4%). Despite its low prevalence, tea tree oil should remain in the differential as an ACD-inducing agent. Patch testing with the patient’s supply of tea tree oil is advised when possible.38
Conclusion
It is customary that the ingredients used in shampoos undergo periodic testing and monitoring to assure the safety of their use. Although it is encouraging that patients are proactive in their efforts to stay abreast of the literature, it is still important that cosmetic scientists, dermatologists, and other experts remain at the forefront of educating the public about these substances. Not doing so can result in the propagation of misinformation and unnecessary fears, which can lead to the adaptation of unhygienic or even unsafe hair care practices. As dermatologists, we must ensure that patients are educated about the benefits and hazards of off-label use of household ingredients to the extent that evidence-based medicine permits. Patients must be informed that not all synthetic substances are harmful, and likewise not all naturally occurring substances are safe.
- The global shampoo market 2014-2019 trends, forecast, and opportunity analysis [press release]. New York, NY: Reportlinker; May 21, 2015.
- Is the ‘no shampoo’ trend healthy or harmful? Mercola website. Published January 16, 2016. Accessed December 8, 2017.
- Feltman R. The science (or lack thereof) behind the ‘no-poo’ hair trend. Washington Post. March 10, 2016. https://www.washingtonpost.com/news/speaking-of-science/wp/2016/03/10/the-science-or-lack-thereof-behind-the-no-poo-hair-trend/?utm_term=.9a61edf3fd5a. Accessed December 11, 2017.
- Bouillon C. Shampoos. Clin Dermatol. 1996;14:113-121.
- Trueb RM. Shampoos: ingredients, efficacy, and adverse effects. J Dtsch Dermatol Ges. 2007;5:356-365.
- Bondi CA, Marks JL, Wroblewski LB, et al. Human and environmental toxicity of sodium lauryl sulfate (SLS): evidence for safe use in household cleaning products. Environ Health Insights. 2015;9:27-32.
- Green K, Johnson RE, Chapman JM, et al. Preservative effects on the healing rate of rabbit corneal epithelium. Lens Eye Toxic Res. 1989;6:37-41.
- Sodium lauryl sulphate. Healthy Choices website. http://www.healthychoices.co.uk/sls.html. Accessed December 8, 2017.
- Tekbas¸ ÖF, Uysal Y, Og˘ur R, et al. Non-irritant baby shampoos may cause cataract development. TSK Koruyucu Hekimlik Bülteni. 2008;1:1-6.
- Cater KC, Harbell JW. Prediction of eye irritation potential of surfactant-based rinse-off personal care formulations by the bovine corneal opacity and permeability (BCOP) assay. Cutan Ocul Toxicol. 2006;25:217-233.
- Birt DF, Lawson TA, Julius AD, et al. Inhibition by dietary selenium of colon cancer induced in the rat by bis(2-oxopropyl) nitrosamine. Cancer Res. 1982;42:4455-4459.
- Rastogi SC. Headspace analysis of 1,4-dioxane in products containing polyethoxylated surfactants by GC-MS. Chromatographia. 1990;29:441-445.
- 1,4-Dioxane. IARC Monogr Eval Carcinog Risks Hum. 1999;71, pt 2:589-602.
- Trueb RM. Dermocosmetic aspects of hair and scalp. J Investig Dermatol Symp Proc. 2005;10:289-292.
- D’Souza P, Rathi SK. Shampoo and conditioners: what a dermatologist should know? Indian J Dermatol. 2015;60:248-254.
- Sasseville D, Alfalah M, Lacroix JP. “Parabenoia” debunked, or “who’s afraid of parabens?” Dermatitis. 2015;26:254-259.
- Krowka JF, Loretz L, Geis PA, et al. Preserving the facts on parabens: an overview of these important tools of the trade. Cosmetics & Toiletries. http://www.cosmeticsandtoiletries.com/research/chemistry/Preserving-the-Facts-on-Parabens-An-Overview-of-These-Important-Tools-of-the Trade-425784294.html. Published June 1, 2017. Accessed December 20, 2017.
- Routledge EJ, Parker J, Odum J, et al. Some alkyl hydroxy benzoate preservatives (parabens) are estrogenic. Toxicol Appl Pharmacol. 1998;153:12Y19.
- Hossaini A, Larsen JJ, Larsen JC. Lack of oestrogenic effects of food preservatives (parabens) in uterotrophic assays. Food Chem Toxicol. 2000;38:319-323.
- Cosmetic Ingredient Review. Final report on the safety assessment of methylparaben, ethylparaben, propylparaben and butylparaben. J Am Coll Toxicol. 1984;3:147-209.
- Cosmetic Ingredient Review. Final report on the safety assessment of isobutylparaben and isopropylparaben. J Am Coll Toxicol. 1995;14:364-372.
- Scientific Committee on Consumer Products. Extended Opinion on the Safety Evaluation of Parabens. European Commission website. https://ec.europa.eu/health/ph_risk/committees/04_sccp/docs/sccp_o_019.pdf. Published January 28, 2005. Accessed December 20, 2017.
- Scientific Committee on Consumer Products. Opinion on Parabens. European Commission website. http://ec.europa.eu/health/scientific_committees/consumer_safety/docs/sccs_o_041.pdf. Revised March 22, 2011. Accessed December 20, 2017.
- European Commission. Commission Regulation (EU) No 258/2014 of 9 April 2014 amending Annexes II and V to Regulation (EC) No 1223/2009 of the European Parliament and of the Council on cosmetic products. EUR-Lex website. http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=uriserv:OJ.L_.2014.107.01.0005.01.ENG. Accessed December 20, 2017.
- American Cancer Society. Antiperspirants and breast cancer risk. https://www.cancer.org/cancer/cancer-causes/antiperspirants-and-breast-cancer-risk.html#references. Revised October 14, 2014. Accessed January 2, 2018.
- MacMillan A. Cutting back on shampoo? 15 things you should know. Health. February 25, 2014. http://www.health.com/health/gallery/0,,20788089,00.html#should-you-go-no-poo--1. Accessed December 10, 2017.
- The ‘no poo’ method. https://www.nopoomethod.com/. Accessed December 10, 2017.
- Fong, D, Gaulin C, Le M, et al. Effectiveness of alternative antimicrobial agents for disinfection of hard surfaces. National Collaborating Centre for Environmental Health website. http://www.ncceh.ca/sites/default/files/Alternative_Antimicrobial_Agents_Aug_2014.pdf. Published August 2014. Accessed December 10, 2017.
- Is baking soda too harsh for natural hair? Black Girl With Long Hair website. http://blackgirllonghair.com/2012/02/is-baking-soda-too-harsh-for-hair/2/. Published February 5, 2012. Accessed December 12, 2017.
- O’Lenick T. Anionic/cationic complexes in hair care. J Cosmet Sci. 2011;62:209-228.
- Gavazzoni Dias MF, de Almeida AM, Cecato PM, et al. The shampoo pH can affect the hair: myth or reality? Int J Trichology. 2014;6:95-99.
- Goodman H. The acid mantle of the skin surface. Ind Med Surg. 1958;27:105-108.
- Korting HC, Kober M, Mueller M, et al. Influence of repeated washings with soap and synthetic detergents on pH and resident flora of the skin of forehead and forearm. results of a cross-over trial in health probationers. Acta Derm Venereol. 1987;67:41-47.
- Tarun J, Susan J, Suria J, et al. Evaluation of pH of bathing soaps and shampoos for skin and hair care. Indian J Dermatol. 2014;59:442-444.
- Corbett JF. The chemistry of hair-care products. J Soc Dyers Colour. 1976;92:285-303.
- McMichael AJ, Hordinsky M. Hair Diseases: Medical, Surgical, and Cosmetic Treatments. New York, NY: Taylor & Francis; 2008:59-72.
- Allardice A, Gummo G. Hair conditioning: quaternary ammonium compounds on various hair types. Cosmet Toiletries. 1993;108:107-109.
- Larson D, Jacob SE. Tea tree oil. Dermatitis. 2012;23:48-49.
Shampoo is a staple in hair grooming that is ever-evolving along with cultural trends. The global shampoo market is expected to reach an estimated value of $25.73 billion by 2019. A major driver of this upward trend in market growth is the increasing demand for natural and organic hair shampoos.1 Society today has a growing fixation on healthy living practices, and as of late, the ingredients in shampoos and other cosmetic products have become one of the latest targets in the health-consciousness craze. In the age of the Internet where information—and misinformation—is widely accessible and dispersed, the general public often strives to self-educate on specialized matters that are out of their expertise. As a result, individuals have developed an aversion to using certain shampoos out of fear that the ingredients, often referred to as “chemicals” by patients due to their complex names, are unnatural and therefore unhealthy.1,2 Product developers are working to meet the demand by reformulating shampoos with labels that indicate sulfate free or paraben free, despite the lack of proof that these formulations are an improvement over traditional approaches to hair health. Additionally, alternative methods of cleansing the hair and scalp, also known as the no-shampoo or “no-poo” method, have begun to gain popularity.2,3
It is essential that dermatologists acknowledge the concerns that their patients have about common shampoo ingredients to dispel the myths that may misinform patient decision-making. This article reviews the controversy surrounding the use of sulfates and parabens in shampoos as well as commonly used shampoo alternatives. Due to the increased prevalence of dry hair shafts in the skin of color population, especially black women, this group is particularly interested in products that will minimize breakage and dryness of the hair. To that end, this population has great interest in the removal of chemical ingredients that may cause damage to the hair shafts, despite the lack of data to support sulfates and paraben damage to hair shafts or scalp skin. Blogs and uninformed hairstylists may propagate these beliefs in a group of consumers who are desperate for new approaches to hair fragility and breakage.
Surfactants and Sulfates
The cleansing ability of a shampoo depends on the surface activity of its detergents. Surface-active ingredients, or surfactants, reduce the surface tension between water and dirt, thus facilitating the removal of environmental dirt from the hair and scalp,4 which is achieved by a molecular structure containing both a hydrophilic and a lipophilic group. Sebum and dirt are bound by the lipophilic ends of the surfactant, becoming the center of a micelle structure with the hydrophilic molecule ends pointing outward. Dirt particles become water soluble and are removed from the scalp and hair shaft upon rinsing with water.4
Surfactants are classified according to the electric charge of the hydrophilic polar group as either anionic, cationic, amphoteric (zwitterionic), or nonionic.5 Each possesses different hair conditioning and cleansing qualities, and multiple surfactants are used in shampoos in differing ratios to accommodate different hair types. In most shampoos, the base consists of anionic and amphoteric surfactants. Depending on individual product requirements, nonionic and cationic surfactants are used to either modify the effects of the surfactants or as conditioning agents.4,5
One subcategory of surfactants that receives much attention is the group of anionic surfactants known as sulfates. Sulfates, particularly sodium lauryl sulfate (SLS), recently have developed a negative reputation as cosmetic ingredients, as reports from various unscientific sources have labeled them as hazardous to one’s health; SLS has been described as a skin and scalp irritant, has been linked to cataract formation, and has even been wrongly labeled as carcinogenic.6 The origins of some of these claims are not clear, though they likely arose from the misinterpretation of complex scientific studies that are easily accessible to laypeople. The link between SLS and ocular irritation or cataract formation is a good illustration of this unsubstantiated fear. A study by Green et al7 showed that corneal exposure to extremely high concentrations of SLS following physical or chemical damage to the eye can result in a slowed healing process. The results of this study have since been wrongly quoted to state that SLS-containing products lead to blindness or severe corneal damage.8 A different study tested for possible ocular irritation in vivo by submerging the lens of an eye into a 20% SLS solution, which accurately approximates the concentration of SLS in rinse-off consumer products.9 However, to achieve ocular irritation, the eyes of laboratory animals were exposed to SLS constantly for 14 days, which would not occur in practical use.9 Similarly, a third study achieved cataract formation in a laboratory only by immersing the lens of an eye into a highly concentrated solution of SLS.10 Such studies are not appropriate representations of how SLS-containing products are used by consumers and have unfortunately been vulnerable to misinterpretation by the general public.
There is no known study that has shown SLS to be carcinogenic. One possible origin of this idea may be from the wrongful interpretation of studies that used SLS as a vehicle substance to test agents that were deemed to be carcinogenic.11 Another possible source of the idea that SLS is carcinogenic comes from its association with 1,4-dioxane, a by-product of the synthesis of certain sulfates such as sodium laureth sulfate due to a process known as ethoxylation.6,12 Although SLS does not undergo this process in its formation and is not linked to 1,4-dioxane, there is potential for cross-contamination of SLS with 1,4-dioxane, which cannot be overlooked. 1,4-Dioxane is classified as “possibly carcinogenic to humans (Group 2B)” by the International Agency for Research on Cancer,13 but screening of SLS for this substance prior to its use in commercial products is standard.
Sulfates are inexpensive detergents that are responsible for lather formation in shampoos as well as in many household cleaning agents.5 Sulfates, similar to all anionic surfactants, are characterized by a negatively charged hydrophilic polar group. The best-known and most commonly used anionic surfactants are sulfated fatty alcohols, alkyl sulfates, and their polyethoxylated analogues alkyl ether sulfates.5,6 Sodium lauryl sulfate (also known as sodium laurilsulfate or sodium dodecyl sulfate) is the most common of them all, found in shampoo and conditioner formulations. Ammonium lauryl sulfate and sodium laureth sulfate are other sulfates commonly used in shampoos and household cleansing products. Sodium lauryl sulfate is a nonvolatile, water-soluble compound. Its partition coefficient (P0), a measure of a substance’s hydrophilic or lipophilic nature, is low at 1.6, making it a rather hydrophilic substance.6 Hydrophilic substances tend to have low bioaccumulation profiles in the body. Additionally, SLS is readily biodegradable. It can be derived from both synthetic and naturally occurring sources; for example, palm kernel oil, petrolatum, and coconut oil are all sources of lauric acid, the starting ingredient used to synthesize SLS. Sodium lauryl sulfate is created by reacting lauryl alcohol with sulfur trioxide gas, followed by neutralization with sodium carbonate (also a naturally occurring compound).6 Sodium lauryl sulfate and other sulfate-containing shampoos widely replaced the usage of traditional soaps formulated from animal or vegetable fats, as these latter formations created a film of insoluble calcium salts on the hair strands upon contact with water, resulting in tangled, dull-appearing hair.5 Additionally, sulfates were preferred to the alkaline pH of traditional soap, which can be harsh on hair strands and cause irritation of the skin and mucous membranes.14 Because they are highly water soluble, sulfates enable the formulation of clear shampoos. They exhibit remarkable cleaning properties and lather formation.5,14
Because sulfates are potent surfactants, they can remove dirt and debris as well as naturally produced healthy oils from the hair and scalp. As a result, sulfates can leave the hair feeling dry and stripped of moisture.4,5 Sulfates are used as the primary detergents in the formulation of deep-cleaning shampoos, which are designed for people who accumulate a heavy buildup of dirt, sebum, and debris from frequent use of styling products. Due to their potent detergency, these shampoos typically are not used on a daily basis but rather at longer intervals.15 A downside to sulfates is that they can have cosmetically unpleasant properties, which can be compensated for by including appropriate softening additives in shampoo formulations.4 A number of anionic surfactants such as olefin sulfonate, alkyl sulfosuccinate, acyl peptides, and alkyl ether carboxylates are well tolerated by the skin and are used together with other anionic and amphoteric surfactants to optimize shampoo properties. Alternatively, sulfate-free shampoos are cleansers compounded by the removal of the anionic group and switched for surfactants with less detergency.4,5
Preservatives and Parabens
Parabens refer to a group of esters of 4-hydroxybenzoic acid commonly used as preservatives in foods, pharmaceuticals, and cosmetics whose widespread use dates back to 1923.16 Concerns over the presence of parabens in shampoos and other cosmetics have been raised by patients for their reputed estrogenic and antiandrogenic effects and suspected involvement in carcinogenesis via endocrine modulation.16,17 In in vitro studies done on yeast assays, parabens have shown weak estrogenic activity that increases in proportion to both the length and increased branching of the alkyl side chains in the paraben’s molecular structure.18 They are 10,000-fold less potent than 17β-estradiol. In in vivo animal studies, parabens show weak estrogenic activity and are 100,000-fold less potent than 17β-estradiol.18 4-Hydroxybenzoic acid, a common metabolite, showed no estrogenic activity when tested both in vitro and in vivo.19 Some concerning research has implicated a link between parabens used in underarm cosmetics, such as deodorants and antiperspirants, and breast cancer16; however, the studies have been conflicting, and there is simply not enough data to assert that parabens cause breast cancer.
The Cosmetic Ingredient Review expert panel first reviewed parabens in 1984 and concluded that “methylparaben, ethylparaben, propylparaben, and butylparaben are safe as cosmetic ingredients in the present practices of use.”20 They extended this statement to include isopropylparaben and isobutylparaben in a later review.21 In 2005, the Scientific Committee on Consumer Products (now known as the Scientific Committee for Consumer Safety) in Europe stated that methylparaben and ethylparaben can be used at levels up to 0.4% in products.22 This decision was reached due to reports of decreased sperm counts and testosterone levels in male juvenile rats exposed to these parabens; however, these reults were not successfully replicated in larger studies.16,22 In 2010, the Scientific Committee for Consumer Safety revisited its stance on parabens, and they then revised their recommendations to say that concentrations of propylparaben and butylparaben should not exceed concentrations of 0.19%, based on “the conservative choice for the calculation of the [Margin-of-Safety] of butyl- and propylparaben.”23 However, in 2011 the use of propylparaben and butylparaben was banned in Denmark for cosmetic products used in children 3 years or younger,16 and the European Commission subsequently amended their directive in 2014, banning isopropylparaben, isobutylparaben, phenylparaben, benzylparaben, and pentylparaben due to lack of data available to evaluate the human risk of these products.24
Contrary to the trends in Europe, there currently are no regulations against the use of parabens in shampoos or other cosmetics in the United States. The American Cancer Society found that there is no evidence to suggest that the current levels of parabens in cosmetic products (eg, antiperspirants) increase one’s risk of breast cancer.25 Parabens are readily absorbed into the body both transdermally and through ingestion but also are believed to be rapidly transformed into harmless and nonspecific metabolites; they are readily metabolized by the liver and excreted in urine, and there is no measured accumulation in tissues.17
Parabens continue to be the most widely used preservatives in personal care products, usually in conjunction with other preservatives. Parabens are good biocides; short-chain esters (eg, methylparabens, ethylparabens) are effective against gram-positive bacteria and are weakly effective against gram-negative bacteria. Long-chain paraben esters (eg, propylparabens, butylparabens) are effective against mold and yeast. The addition of other preservatives creates a broad spectrum of antimicrobial defense in consumer products. Other preservatives include formaldehyde releasers or phenoxyethanol, as well as chelating agents such as EDTA, which improve the stability of these cosmetic products when exposed to air.16 Parabens are naturally occurring substances found in foods such as blueberries, barley, strawberries, yeast, olives, and grapes. As a colorless, odorless, and inexpensive substance, their use has been heavily favored in cosmetic and food products.16
Shampoo Alternatives and the No-Poo Method
Although research has not demonstrated any long-term danger to using shampoo, certain chemicals found in shampoos have the potential to irritate the scalp. Commonly cited allergens in shampoos include cocamidopropyl betaine, propylene glycol, vitamin E (tocopherol), parabens, and benzophenones.5 Additionally, the rising use of formaldehyde-releasing preservatives and isothiazolinones due to mounting pressures to move away from parabens has led to an increase in cases of allergic contact dermatitis (ACD).16 However, the irritability (rather than allergenicity) of these substances often is established during patch testing, a method of detecting delayed-type allergic reactions, which is important to note because patch testing requires a substance to be exposed to the skin for 24 to 48 hours, whereas exposure to shampoo ingredients may last a matter of minutes at most and occur in lesser concentrations because the ingredients are diluted by water in the rinsing process. Given these differences, it is unlikely that a patient would develop a true allergic response from regular shampoo use. Nevertheless, in patients who are already sensitized, exposure could conceivably trigger ACD, and patients must be cognizant of the composition of their shampoos.16
The no-poo method refers to the avoidance of commercial shampoo products when cleansing the hair and scalp and encompasses different methods of cleansing the hair, such as the use of household items (eg, baking soda, apple cider vinegar [ACV]), the use of conditioners to wash the hair (also known as conditioner-only washing or co-washing), treating the scalp with tea tree oil, or simply rinsing the hair with water. Proponents of the no-poo method believe that abstaining from shampoo use leads to healthier hair, retained natural oils, and less exposure to supposedly dangerous chemicals such as parabens or sulfates.2,3,26-28 However, there are no known studies in the literature that assess or support the hypotheses of the no-poo method.
Baking Soda and ACV
Baking soda (sodium bicarbonate) is a substance commonly found in the average household. It has been used in toothpaste formulas and cosmetic products and is known for its acid-neutralizing properties. Baking soda has been shown to have some antifungal and viricidal properties through an unknown mechanism of action.28 It has gained popularity for its use as a means of reducing the appearance of excessive greasiness of the hair shafts. Users also have reported that when washing their hair with baking soda, they are able to achieve a clean scalp and hair that feels soft to the touch.2,3,26,27,29 Despite these reports, users must beware of using baking soda without adequately diluting it with water. Baking soda is a known alkaline irritant.26,30 With a pH of 9, baking soda causes the cuticle layer of the hair fiber to open, increasing the capacity for water absorption. Water penetrates the scales that open, breaking the hydrogen bonds of the keratin molecule.31 Keratin is a spiral helical molecule that keeps its shape due to hydrogen, disulfide, and ionic bonds, as well as Van der Waals force.30 Hydrolysis of these bonds due to exposure to baking soda lowers the elasticity of the hair and increases the negative electrical net charge of the hair fiber surface, which leads to increased friction between fibers, cuticle damage, hair fragility, and fiber breakage.32,33
Apple cider vinegar is an apple-derived acetic acid solution with a pH ranging from 3.1 to 5.28 The pH range of ACV is considered to be ideal for hair by no-poo proponents, as it is similar to the natural pH of the scalp. Its acidic properties are responsible for its antimicrobial abilities, particularly its effectiveness against gram-negative bacteria.30 The acetic acid of ACV can partially interrupt oil interfaces, which contributes to its mild ability to remove product residue and scalp buildup from the hair shaft; the acetic acid also tightens the cuticles on hair fibers.33 Apple cider vinegar is used as a means of cleansing the hair and scalp by no-poo proponents2,3,26; other uses for ACV include using it as a rinse following washing and/or conditioning of the hair or as a means of preserving color in color-treated hair. There also is evidence that ACV may have antifungal properties.28 However, consumers must be aware that if it is not diluted in water, ACV may be too caustic for direct application to the hair and may lead to damage; it can be irritating to eyes, mucus membranes, and acutely inflamed skin. Also, vinegar rinses used on processed or chemically damaged hair may lead to increased hair fragility.2,3
Hair fibers have a pH of 3.67, while the scalp has a pH between 4.5 and 6.2. This slightly acidic film acts as a barrier to viruses, bacteria, and other potential contaminants.33 Studies have shown that the pH of skin increases in proportion to the pH of the cleanser used.34 Therefore, due to the naturally acidic pH of the scalp, acid-balanced shampoos generally are recommended. Shampoos should not have a pH higher than 5.5, as hair shafts can swell due to alkalinization, which can be prevented by pH balancing the shampoo through the addition of an acidic substance (eg, glycolic acid, citric acid) to lower the pH down to approximately 5.5. Apple cider vinegar often is used for this purpose. However, one study revealed that 82% of shampoos already have an acidic pH.34
Conditioner-Only Washing (Co-washing)
Conditioner-only washing, or co-washing, is a widely practiced method of hair grooming. It is popular among individuals who find that commercial shampoos strip too much of the natural hair oils away, leaving the hair rough or unmanageable. Co-washing is not harmful to the hair; however, the molecular structure and function of a conditioner and that of a shampoo are very different.5,35,36 Conditioners are not formulated to remove dirt and buildup in the hair but rather to add substances to the hair, and thus cannot provide extensive cleansing of the hair and scalp; therefore, it is inappropriate to use co-washing as a replacement for shampooing. Quaternary conditioning agents are an exception because they contain amphoteric detergents comprised of both anionic and cationic groups, which allow them both the ability to remove dirt and sebum with its anionic group, typically found in shampoos, as well as the ability to coat and condition the hair due to the high affinity of the cationic group for the negatively charged hair fibers.36,37 Amphoteric detergents are commonly found in 2-in-1 conditioning cleansers, among other ingredients, such as hydrolyzed animal proteins that temporarily plug surface defects on the hair fiber, and dimethicone, a synthetic oil that creates a thin film over the hair shaft, increasing shine and manageability. Of note, these conditioning shampoos are ideal for individuals with minimal product buildup on the hair and scalp and are not adequate scalp cleansers for individuals who either wash their hair infrequently or who regularly use hairstyling products.36,37
Tea Tree Oil
Tea tree oil is an essential oil extracted from the Melaleuca alternifolia plant of the Myrtaceae family. It is native to the coast of northeastern Australia. A holy grail of natural cosmetics, tea tree oil is widely known for its antiviral, antifungal, and antiseptic properties.38 Although not used as a stand-alone cleanser, it is often added to a number of cosmetic products, including shampoos and co-washes. Although deemed safe for topical use, it has been shown to be quite toxic when ingested. Symptoms of ingestion include nausea, vomiting, hallucinations, and coma. The common concern with tea tree oil is its ability to cause ACD. In particular, it is believed that the oxidation products of tea tree oil are allergenic rather than the tea tree oil itself. The evaluation of tea tree oil as a potential contact allergen has been quite difficult; it consists of more than 100 distinct compounds and is often mislabeled, or does not meet the guidelines of the International Organization for Standardization. Nonetheless, the prevalence of ACD due to tea tree oil is low (approximately 1.4%). Despite its low prevalence, tea tree oil should remain in the differential as an ACD-inducing agent. Patch testing with the patient’s supply of tea tree oil is advised when possible.38
Conclusion
It is customary that the ingredients used in shampoos undergo periodic testing and monitoring to assure the safety of their use. Although it is encouraging that patients are proactive in their efforts to stay abreast of the literature, it is still important that cosmetic scientists, dermatologists, and other experts remain at the forefront of educating the public about these substances. Not doing so can result in the propagation of misinformation and unnecessary fears, which can lead to the adaptation of unhygienic or even unsafe hair care practices. As dermatologists, we must ensure that patients are educated about the benefits and hazards of off-label use of household ingredients to the extent that evidence-based medicine permits. Patients must be informed that not all synthetic substances are harmful, and likewise not all naturally occurring substances are safe.
Shampoo is a staple in hair grooming that is ever-evolving along with cultural trends. The global shampoo market is expected to reach an estimated value of $25.73 billion by 2019. A major driver of this upward trend in market growth is the increasing demand for natural and organic hair shampoos.1 Society today has a growing fixation on healthy living practices, and as of late, the ingredients in shampoos and other cosmetic products have become one of the latest targets in the health-consciousness craze. In the age of the Internet where information—and misinformation—is widely accessible and dispersed, the general public often strives to self-educate on specialized matters that are out of their expertise. As a result, individuals have developed an aversion to using certain shampoos out of fear that the ingredients, often referred to as “chemicals” by patients due to their complex names, are unnatural and therefore unhealthy.1,2 Product developers are working to meet the demand by reformulating shampoos with labels that indicate sulfate free or paraben free, despite the lack of proof that these formulations are an improvement over traditional approaches to hair health. Additionally, alternative methods of cleansing the hair and scalp, also known as the no-shampoo or “no-poo” method, have begun to gain popularity.2,3
It is essential that dermatologists acknowledge the concerns that their patients have about common shampoo ingredients to dispel the myths that may misinform patient decision-making. This article reviews the controversy surrounding the use of sulfates and parabens in shampoos as well as commonly used shampoo alternatives. Due to the increased prevalence of dry hair shafts in the skin of color population, especially black women, this group is particularly interested in products that will minimize breakage and dryness of the hair. To that end, this population has great interest in the removal of chemical ingredients that may cause damage to the hair shafts, despite the lack of data to support sulfates and paraben damage to hair shafts or scalp skin. Blogs and uninformed hairstylists may propagate these beliefs in a group of consumers who are desperate for new approaches to hair fragility and breakage.
Surfactants and Sulfates
The cleansing ability of a shampoo depends on the surface activity of its detergents. Surface-active ingredients, or surfactants, reduce the surface tension between water and dirt, thus facilitating the removal of environmental dirt from the hair and scalp,4 which is achieved by a molecular structure containing both a hydrophilic and a lipophilic group. Sebum and dirt are bound by the lipophilic ends of the surfactant, becoming the center of a micelle structure with the hydrophilic molecule ends pointing outward. Dirt particles become water soluble and are removed from the scalp and hair shaft upon rinsing with water.4
Surfactants are classified according to the electric charge of the hydrophilic polar group as either anionic, cationic, amphoteric (zwitterionic), or nonionic.5 Each possesses different hair conditioning and cleansing qualities, and multiple surfactants are used in shampoos in differing ratios to accommodate different hair types. In most shampoos, the base consists of anionic and amphoteric surfactants. Depending on individual product requirements, nonionic and cationic surfactants are used to either modify the effects of the surfactants or as conditioning agents.4,5
One subcategory of surfactants that receives much attention is the group of anionic surfactants known as sulfates. Sulfates, particularly sodium lauryl sulfate (SLS), recently have developed a negative reputation as cosmetic ingredients, as reports from various unscientific sources have labeled them as hazardous to one’s health; SLS has been described as a skin and scalp irritant, has been linked to cataract formation, and has even been wrongly labeled as carcinogenic.6 The origins of some of these claims are not clear, though they likely arose from the misinterpretation of complex scientific studies that are easily accessible to laypeople. The link between SLS and ocular irritation or cataract formation is a good illustration of this unsubstantiated fear. A study by Green et al7 showed that corneal exposure to extremely high concentrations of SLS following physical or chemical damage to the eye can result in a slowed healing process. The results of this study have since been wrongly quoted to state that SLS-containing products lead to blindness or severe corneal damage.8 A different study tested for possible ocular irritation in vivo by submerging the lens of an eye into a 20% SLS solution, which accurately approximates the concentration of SLS in rinse-off consumer products.9 However, to achieve ocular irritation, the eyes of laboratory animals were exposed to SLS constantly for 14 days, which would not occur in practical use.9 Similarly, a third study achieved cataract formation in a laboratory only by immersing the lens of an eye into a highly concentrated solution of SLS.10 Such studies are not appropriate representations of how SLS-containing products are used by consumers and have unfortunately been vulnerable to misinterpretation by the general public.
There is no known study that has shown SLS to be carcinogenic. One possible origin of this idea may be from the wrongful interpretation of studies that used SLS as a vehicle substance to test agents that were deemed to be carcinogenic.11 Another possible source of the idea that SLS is carcinogenic comes from its association with 1,4-dioxane, a by-product of the synthesis of certain sulfates such as sodium laureth sulfate due to a process known as ethoxylation.6,12 Although SLS does not undergo this process in its formation and is not linked to 1,4-dioxane, there is potential for cross-contamination of SLS with 1,4-dioxane, which cannot be overlooked. 1,4-Dioxane is classified as “possibly carcinogenic to humans (Group 2B)” by the International Agency for Research on Cancer,13 but screening of SLS for this substance prior to its use in commercial products is standard.
Sulfates are inexpensive detergents that are responsible for lather formation in shampoos as well as in many household cleaning agents.5 Sulfates, similar to all anionic surfactants, are characterized by a negatively charged hydrophilic polar group. The best-known and most commonly used anionic surfactants are sulfated fatty alcohols, alkyl sulfates, and their polyethoxylated analogues alkyl ether sulfates.5,6 Sodium lauryl sulfate (also known as sodium laurilsulfate or sodium dodecyl sulfate) is the most common of them all, found in shampoo and conditioner formulations. Ammonium lauryl sulfate and sodium laureth sulfate are other sulfates commonly used in shampoos and household cleansing products. Sodium lauryl sulfate is a nonvolatile, water-soluble compound. Its partition coefficient (P0), a measure of a substance’s hydrophilic or lipophilic nature, is low at 1.6, making it a rather hydrophilic substance.6 Hydrophilic substances tend to have low bioaccumulation profiles in the body. Additionally, SLS is readily biodegradable. It can be derived from both synthetic and naturally occurring sources; for example, palm kernel oil, petrolatum, and coconut oil are all sources of lauric acid, the starting ingredient used to synthesize SLS. Sodium lauryl sulfate is created by reacting lauryl alcohol with sulfur trioxide gas, followed by neutralization with sodium carbonate (also a naturally occurring compound).6 Sodium lauryl sulfate and other sulfate-containing shampoos widely replaced the usage of traditional soaps formulated from animal or vegetable fats, as these latter formations created a film of insoluble calcium salts on the hair strands upon contact with water, resulting in tangled, dull-appearing hair.5 Additionally, sulfates were preferred to the alkaline pH of traditional soap, which can be harsh on hair strands and cause irritation of the skin and mucous membranes.14 Because they are highly water soluble, sulfates enable the formulation of clear shampoos. They exhibit remarkable cleaning properties and lather formation.5,14
Because sulfates are potent surfactants, they can remove dirt and debris as well as naturally produced healthy oils from the hair and scalp. As a result, sulfates can leave the hair feeling dry and stripped of moisture.4,5 Sulfates are used as the primary detergents in the formulation of deep-cleaning shampoos, which are designed for people who accumulate a heavy buildup of dirt, sebum, and debris from frequent use of styling products. Due to their potent detergency, these shampoos typically are not used on a daily basis but rather at longer intervals.15 A downside to sulfates is that they can have cosmetically unpleasant properties, which can be compensated for by including appropriate softening additives in shampoo formulations.4 A number of anionic surfactants such as olefin sulfonate, alkyl sulfosuccinate, acyl peptides, and alkyl ether carboxylates are well tolerated by the skin and are used together with other anionic and amphoteric surfactants to optimize shampoo properties. Alternatively, sulfate-free shampoos are cleansers compounded by the removal of the anionic group and switched for surfactants with less detergency.4,5
Preservatives and Parabens
Parabens refer to a group of esters of 4-hydroxybenzoic acid commonly used as preservatives in foods, pharmaceuticals, and cosmetics whose widespread use dates back to 1923.16 Concerns over the presence of parabens in shampoos and other cosmetics have been raised by patients for their reputed estrogenic and antiandrogenic effects and suspected involvement in carcinogenesis via endocrine modulation.16,17 In in vitro studies done on yeast assays, parabens have shown weak estrogenic activity that increases in proportion to both the length and increased branching of the alkyl side chains in the paraben’s molecular structure.18 They are 10,000-fold less potent than 17β-estradiol. In in vivo animal studies, parabens show weak estrogenic activity and are 100,000-fold less potent than 17β-estradiol.18 4-Hydroxybenzoic acid, a common metabolite, showed no estrogenic activity when tested both in vitro and in vivo.19 Some concerning research has implicated a link between parabens used in underarm cosmetics, such as deodorants and antiperspirants, and breast cancer16; however, the studies have been conflicting, and there is simply not enough data to assert that parabens cause breast cancer.
The Cosmetic Ingredient Review expert panel first reviewed parabens in 1984 and concluded that “methylparaben, ethylparaben, propylparaben, and butylparaben are safe as cosmetic ingredients in the present practices of use.”20 They extended this statement to include isopropylparaben and isobutylparaben in a later review.21 In 2005, the Scientific Committee on Consumer Products (now known as the Scientific Committee for Consumer Safety) in Europe stated that methylparaben and ethylparaben can be used at levels up to 0.4% in products.22 This decision was reached due to reports of decreased sperm counts and testosterone levels in male juvenile rats exposed to these parabens; however, these reults were not successfully replicated in larger studies.16,22 In 2010, the Scientific Committee for Consumer Safety revisited its stance on parabens, and they then revised their recommendations to say that concentrations of propylparaben and butylparaben should not exceed concentrations of 0.19%, based on “the conservative choice for the calculation of the [Margin-of-Safety] of butyl- and propylparaben.”23 However, in 2011 the use of propylparaben and butylparaben was banned in Denmark for cosmetic products used in children 3 years or younger,16 and the European Commission subsequently amended their directive in 2014, banning isopropylparaben, isobutylparaben, phenylparaben, benzylparaben, and pentylparaben due to lack of data available to evaluate the human risk of these products.24
Contrary to the trends in Europe, there currently are no regulations against the use of parabens in shampoos or other cosmetics in the United States. The American Cancer Society found that there is no evidence to suggest that the current levels of parabens in cosmetic products (eg, antiperspirants) increase one’s risk of breast cancer.25 Parabens are readily absorbed into the body both transdermally and through ingestion but also are believed to be rapidly transformed into harmless and nonspecific metabolites; they are readily metabolized by the liver and excreted in urine, and there is no measured accumulation in tissues.17
Parabens continue to be the most widely used preservatives in personal care products, usually in conjunction with other preservatives. Parabens are good biocides; short-chain esters (eg, methylparabens, ethylparabens) are effective against gram-positive bacteria and are weakly effective against gram-negative bacteria. Long-chain paraben esters (eg, propylparabens, butylparabens) are effective against mold and yeast. The addition of other preservatives creates a broad spectrum of antimicrobial defense in consumer products. Other preservatives include formaldehyde releasers or phenoxyethanol, as well as chelating agents such as EDTA, which improve the stability of these cosmetic products when exposed to air.16 Parabens are naturally occurring substances found in foods such as blueberries, barley, strawberries, yeast, olives, and grapes. As a colorless, odorless, and inexpensive substance, their use has been heavily favored in cosmetic and food products.16
Shampoo Alternatives and the No-Poo Method
Although research has not demonstrated any long-term danger to using shampoo, certain chemicals found in shampoos have the potential to irritate the scalp. Commonly cited allergens in shampoos include cocamidopropyl betaine, propylene glycol, vitamin E (tocopherol), parabens, and benzophenones.5 Additionally, the rising use of formaldehyde-releasing preservatives and isothiazolinones due to mounting pressures to move away from parabens has led to an increase in cases of allergic contact dermatitis (ACD).16 However, the irritability (rather than allergenicity) of these substances often is established during patch testing, a method of detecting delayed-type allergic reactions, which is important to note because patch testing requires a substance to be exposed to the skin for 24 to 48 hours, whereas exposure to shampoo ingredients may last a matter of minutes at most and occur in lesser concentrations because the ingredients are diluted by water in the rinsing process. Given these differences, it is unlikely that a patient would develop a true allergic response from regular shampoo use. Nevertheless, in patients who are already sensitized, exposure could conceivably trigger ACD, and patients must be cognizant of the composition of their shampoos.16
The no-poo method refers to the avoidance of commercial shampoo products when cleansing the hair and scalp and encompasses different methods of cleansing the hair, such as the use of household items (eg, baking soda, apple cider vinegar [ACV]), the use of conditioners to wash the hair (also known as conditioner-only washing or co-washing), treating the scalp with tea tree oil, or simply rinsing the hair with water. Proponents of the no-poo method believe that abstaining from shampoo use leads to healthier hair, retained natural oils, and less exposure to supposedly dangerous chemicals such as parabens or sulfates.2,3,26-28 However, there are no known studies in the literature that assess or support the hypotheses of the no-poo method.
Baking Soda and ACV
Baking soda (sodium bicarbonate) is a substance commonly found in the average household. It has been used in toothpaste formulas and cosmetic products and is known for its acid-neutralizing properties. Baking soda has been shown to have some antifungal and viricidal properties through an unknown mechanism of action.28 It has gained popularity for its use as a means of reducing the appearance of excessive greasiness of the hair shafts. Users also have reported that when washing their hair with baking soda, they are able to achieve a clean scalp and hair that feels soft to the touch.2,3,26,27,29 Despite these reports, users must beware of using baking soda without adequately diluting it with water. Baking soda is a known alkaline irritant.26,30 With a pH of 9, baking soda causes the cuticle layer of the hair fiber to open, increasing the capacity for water absorption. Water penetrates the scales that open, breaking the hydrogen bonds of the keratin molecule.31 Keratin is a spiral helical molecule that keeps its shape due to hydrogen, disulfide, and ionic bonds, as well as Van der Waals force.30 Hydrolysis of these bonds due to exposure to baking soda lowers the elasticity of the hair and increases the negative electrical net charge of the hair fiber surface, which leads to increased friction between fibers, cuticle damage, hair fragility, and fiber breakage.32,33
Apple cider vinegar is an apple-derived acetic acid solution with a pH ranging from 3.1 to 5.28 The pH range of ACV is considered to be ideal for hair by no-poo proponents, as it is similar to the natural pH of the scalp. Its acidic properties are responsible for its antimicrobial abilities, particularly its effectiveness against gram-negative bacteria.30 The acetic acid of ACV can partially interrupt oil interfaces, which contributes to its mild ability to remove product residue and scalp buildup from the hair shaft; the acetic acid also tightens the cuticles on hair fibers.33 Apple cider vinegar is used as a means of cleansing the hair and scalp by no-poo proponents2,3,26; other uses for ACV include using it as a rinse following washing and/or conditioning of the hair or as a means of preserving color in color-treated hair. There also is evidence that ACV may have antifungal properties.28 However, consumers must be aware that if it is not diluted in water, ACV may be too caustic for direct application to the hair and may lead to damage; it can be irritating to eyes, mucus membranes, and acutely inflamed skin. Also, vinegar rinses used on processed or chemically damaged hair may lead to increased hair fragility.2,3
Hair fibers have a pH of 3.67, while the scalp has a pH between 4.5 and 6.2. This slightly acidic film acts as a barrier to viruses, bacteria, and other potential contaminants.33 Studies have shown that the pH of skin increases in proportion to the pH of the cleanser used.34 Therefore, due to the naturally acidic pH of the scalp, acid-balanced shampoos generally are recommended. Shampoos should not have a pH higher than 5.5, as hair shafts can swell due to alkalinization, which can be prevented by pH balancing the shampoo through the addition of an acidic substance (eg, glycolic acid, citric acid) to lower the pH down to approximately 5.5. Apple cider vinegar often is used for this purpose. However, one study revealed that 82% of shampoos already have an acidic pH.34
Conditioner-Only Washing (Co-washing)
Conditioner-only washing, or co-washing, is a widely practiced method of hair grooming. It is popular among individuals who find that commercial shampoos strip too much of the natural hair oils away, leaving the hair rough or unmanageable. Co-washing is not harmful to the hair; however, the molecular structure and function of a conditioner and that of a shampoo are very different.5,35,36 Conditioners are not formulated to remove dirt and buildup in the hair but rather to add substances to the hair, and thus cannot provide extensive cleansing of the hair and scalp; therefore, it is inappropriate to use co-washing as a replacement for shampooing. Quaternary conditioning agents are an exception because they contain amphoteric detergents comprised of both anionic and cationic groups, which allow them both the ability to remove dirt and sebum with its anionic group, typically found in shampoos, as well as the ability to coat and condition the hair due to the high affinity of the cationic group for the negatively charged hair fibers.36,37 Amphoteric detergents are commonly found in 2-in-1 conditioning cleansers, among other ingredients, such as hydrolyzed animal proteins that temporarily plug surface defects on the hair fiber, and dimethicone, a synthetic oil that creates a thin film over the hair shaft, increasing shine and manageability. Of note, these conditioning shampoos are ideal for individuals with minimal product buildup on the hair and scalp and are not adequate scalp cleansers for individuals who either wash their hair infrequently or who regularly use hairstyling products.36,37
Tea Tree Oil
Tea tree oil is an essential oil extracted from the Melaleuca alternifolia plant of the Myrtaceae family. It is native to the coast of northeastern Australia. A holy grail of natural cosmetics, tea tree oil is widely known for its antiviral, antifungal, and antiseptic properties.38 Although not used as a stand-alone cleanser, it is often added to a number of cosmetic products, including shampoos and co-washes. Although deemed safe for topical use, it has been shown to be quite toxic when ingested. Symptoms of ingestion include nausea, vomiting, hallucinations, and coma. The common concern with tea tree oil is its ability to cause ACD. In particular, it is believed that the oxidation products of tea tree oil are allergenic rather than the tea tree oil itself. The evaluation of tea tree oil as a potential contact allergen has been quite difficult; it consists of more than 100 distinct compounds and is often mislabeled, or does not meet the guidelines of the International Organization for Standardization. Nonetheless, the prevalence of ACD due to tea tree oil is low (approximately 1.4%). Despite its low prevalence, tea tree oil should remain in the differential as an ACD-inducing agent. Patch testing with the patient’s supply of tea tree oil is advised when possible.38
Conclusion
It is customary that the ingredients used in shampoos undergo periodic testing and monitoring to assure the safety of their use. Although it is encouraging that patients are proactive in their efforts to stay abreast of the literature, it is still important that cosmetic scientists, dermatologists, and other experts remain at the forefront of educating the public about these substances. Not doing so can result in the propagation of misinformation and unnecessary fears, which can lead to the adaptation of unhygienic or even unsafe hair care practices. As dermatologists, we must ensure that patients are educated about the benefits and hazards of off-label use of household ingredients to the extent that evidence-based medicine permits. Patients must be informed that not all synthetic substances are harmful, and likewise not all naturally occurring substances are safe.
- The global shampoo market 2014-2019 trends, forecast, and opportunity analysis [press release]. New York, NY: Reportlinker; May 21, 2015.
- Is the ‘no shampoo’ trend healthy or harmful? Mercola website. Published January 16, 2016. Accessed December 8, 2017.
- Feltman R. The science (or lack thereof) behind the ‘no-poo’ hair trend. Washington Post. March 10, 2016. https://www.washingtonpost.com/news/speaking-of-science/wp/2016/03/10/the-science-or-lack-thereof-behind-the-no-poo-hair-trend/?utm_term=.9a61edf3fd5a. Accessed December 11, 2017.
- Bouillon C. Shampoos. Clin Dermatol. 1996;14:113-121.
- Trueb RM. Shampoos: ingredients, efficacy, and adverse effects. J Dtsch Dermatol Ges. 2007;5:356-365.
- Bondi CA, Marks JL, Wroblewski LB, et al. Human and environmental toxicity of sodium lauryl sulfate (SLS): evidence for safe use in household cleaning products. Environ Health Insights. 2015;9:27-32.
- Green K, Johnson RE, Chapman JM, et al. Preservative effects on the healing rate of rabbit corneal epithelium. Lens Eye Toxic Res. 1989;6:37-41.
- Sodium lauryl sulphate. Healthy Choices website. http://www.healthychoices.co.uk/sls.html. Accessed December 8, 2017.
- Tekbas¸ ÖF, Uysal Y, Og˘ur R, et al. Non-irritant baby shampoos may cause cataract development. TSK Koruyucu Hekimlik Bülteni. 2008;1:1-6.
- Cater KC, Harbell JW. Prediction of eye irritation potential of surfactant-based rinse-off personal care formulations by the bovine corneal opacity and permeability (BCOP) assay. Cutan Ocul Toxicol. 2006;25:217-233.
- Birt DF, Lawson TA, Julius AD, et al. Inhibition by dietary selenium of colon cancer induced in the rat by bis(2-oxopropyl) nitrosamine. Cancer Res. 1982;42:4455-4459.
- Rastogi SC. Headspace analysis of 1,4-dioxane in products containing polyethoxylated surfactants by GC-MS. Chromatographia. 1990;29:441-445.
- 1,4-Dioxane. IARC Monogr Eval Carcinog Risks Hum. 1999;71, pt 2:589-602.
- Trueb RM. Dermocosmetic aspects of hair and scalp. J Investig Dermatol Symp Proc. 2005;10:289-292.
- D’Souza P, Rathi SK. Shampoo and conditioners: what a dermatologist should know? Indian J Dermatol. 2015;60:248-254.
- Sasseville D, Alfalah M, Lacroix JP. “Parabenoia” debunked, or “who’s afraid of parabens?” Dermatitis. 2015;26:254-259.
- Krowka JF, Loretz L, Geis PA, et al. Preserving the facts on parabens: an overview of these important tools of the trade. Cosmetics & Toiletries. http://www.cosmeticsandtoiletries.com/research/chemistry/Preserving-the-Facts-on-Parabens-An-Overview-of-These-Important-Tools-of-the Trade-425784294.html. Published June 1, 2017. Accessed December 20, 2017.
- Routledge EJ, Parker J, Odum J, et al. Some alkyl hydroxy benzoate preservatives (parabens) are estrogenic. Toxicol Appl Pharmacol. 1998;153:12Y19.
- Hossaini A, Larsen JJ, Larsen JC. Lack of oestrogenic effects of food preservatives (parabens) in uterotrophic assays. Food Chem Toxicol. 2000;38:319-323.
- Cosmetic Ingredient Review. Final report on the safety assessment of methylparaben, ethylparaben, propylparaben and butylparaben. J Am Coll Toxicol. 1984;3:147-209.
- Cosmetic Ingredient Review. Final report on the safety assessment of isobutylparaben and isopropylparaben. J Am Coll Toxicol. 1995;14:364-372.
- Scientific Committee on Consumer Products. Extended Opinion on the Safety Evaluation of Parabens. European Commission website. https://ec.europa.eu/health/ph_risk/committees/04_sccp/docs/sccp_o_019.pdf. Published January 28, 2005. Accessed December 20, 2017.
- Scientific Committee on Consumer Products. Opinion on Parabens. European Commission website. http://ec.europa.eu/health/scientific_committees/consumer_safety/docs/sccs_o_041.pdf. Revised March 22, 2011. Accessed December 20, 2017.
- European Commission. Commission Regulation (EU) No 258/2014 of 9 April 2014 amending Annexes II and V to Regulation (EC) No 1223/2009 of the European Parliament and of the Council on cosmetic products. EUR-Lex website. http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=uriserv:OJ.L_.2014.107.01.0005.01.ENG. Accessed December 20, 2017.
- American Cancer Society. Antiperspirants and breast cancer risk. https://www.cancer.org/cancer/cancer-causes/antiperspirants-and-breast-cancer-risk.html#references. Revised October 14, 2014. Accessed January 2, 2018.
- MacMillan A. Cutting back on shampoo? 15 things you should know. Health. February 25, 2014. http://www.health.com/health/gallery/0,,20788089,00.html#should-you-go-no-poo--1. Accessed December 10, 2017.
- The ‘no poo’ method. https://www.nopoomethod.com/. Accessed December 10, 2017.
- Fong, D, Gaulin C, Le M, et al. Effectiveness of alternative antimicrobial agents for disinfection of hard surfaces. National Collaborating Centre for Environmental Health website. http://www.ncceh.ca/sites/default/files/Alternative_Antimicrobial_Agents_Aug_2014.pdf. Published August 2014. Accessed December 10, 2017.
- Is baking soda too harsh for natural hair? Black Girl With Long Hair website. http://blackgirllonghair.com/2012/02/is-baking-soda-too-harsh-for-hair/2/. Published February 5, 2012. Accessed December 12, 2017.
- O’Lenick T. Anionic/cationic complexes in hair care. J Cosmet Sci. 2011;62:209-228.
- Gavazzoni Dias MF, de Almeida AM, Cecato PM, et al. The shampoo pH can affect the hair: myth or reality? Int J Trichology. 2014;6:95-99.
- Goodman H. The acid mantle of the skin surface. Ind Med Surg. 1958;27:105-108.
- Korting HC, Kober M, Mueller M, et al. Influence of repeated washings with soap and synthetic detergents on pH and resident flora of the skin of forehead and forearm. results of a cross-over trial in health probationers. Acta Derm Venereol. 1987;67:41-47.
- Tarun J, Susan J, Suria J, et al. Evaluation of pH of bathing soaps and shampoos for skin and hair care. Indian J Dermatol. 2014;59:442-444.
- Corbett JF. The chemistry of hair-care products. J Soc Dyers Colour. 1976;92:285-303.
- McMichael AJ, Hordinsky M. Hair Diseases: Medical, Surgical, and Cosmetic Treatments. New York, NY: Taylor & Francis; 2008:59-72.
- Allardice A, Gummo G. Hair conditioning: quaternary ammonium compounds on various hair types. Cosmet Toiletries. 1993;108:107-109.
- Larson D, Jacob SE. Tea tree oil. Dermatitis. 2012;23:48-49.
- The global shampoo market 2014-2019 trends, forecast, and opportunity analysis [press release]. New York, NY: Reportlinker; May 21, 2015.
- Is the ‘no shampoo’ trend healthy or harmful? Mercola website. Published January 16, 2016. Accessed December 8, 2017.
- Feltman R. The science (or lack thereof) behind the ‘no-poo’ hair trend. Washington Post. March 10, 2016. https://www.washingtonpost.com/news/speaking-of-science/wp/2016/03/10/the-science-or-lack-thereof-behind-the-no-poo-hair-trend/?utm_term=.9a61edf3fd5a. Accessed December 11, 2017.
- Bouillon C. Shampoos. Clin Dermatol. 1996;14:113-121.
- Trueb RM. Shampoos: ingredients, efficacy, and adverse effects. J Dtsch Dermatol Ges. 2007;5:356-365.
- Bondi CA, Marks JL, Wroblewski LB, et al. Human and environmental toxicity of sodium lauryl sulfate (SLS): evidence for safe use in household cleaning products. Environ Health Insights. 2015;9:27-32.
- Green K, Johnson RE, Chapman JM, et al. Preservative effects on the healing rate of rabbit corneal epithelium. Lens Eye Toxic Res. 1989;6:37-41.
- Sodium lauryl sulphate. Healthy Choices website. http://www.healthychoices.co.uk/sls.html. Accessed December 8, 2017.
- Tekbas¸ ÖF, Uysal Y, Og˘ur R, et al. Non-irritant baby shampoos may cause cataract development. TSK Koruyucu Hekimlik Bülteni. 2008;1:1-6.
- Cater KC, Harbell JW. Prediction of eye irritation potential of surfactant-based rinse-off personal care formulations by the bovine corneal opacity and permeability (BCOP) assay. Cutan Ocul Toxicol. 2006;25:217-233.
- Birt DF, Lawson TA, Julius AD, et al. Inhibition by dietary selenium of colon cancer induced in the rat by bis(2-oxopropyl) nitrosamine. Cancer Res. 1982;42:4455-4459.
- Rastogi SC. Headspace analysis of 1,4-dioxane in products containing polyethoxylated surfactants by GC-MS. Chromatographia. 1990;29:441-445.
- 1,4-Dioxane. IARC Monogr Eval Carcinog Risks Hum. 1999;71, pt 2:589-602.
- Trueb RM. Dermocosmetic aspects of hair and scalp. J Investig Dermatol Symp Proc. 2005;10:289-292.
- D’Souza P, Rathi SK. Shampoo and conditioners: what a dermatologist should know? Indian J Dermatol. 2015;60:248-254.
- Sasseville D, Alfalah M, Lacroix JP. “Parabenoia” debunked, or “who’s afraid of parabens?” Dermatitis. 2015;26:254-259.
- Krowka JF, Loretz L, Geis PA, et al. Preserving the facts on parabens: an overview of these important tools of the trade. Cosmetics & Toiletries. http://www.cosmeticsandtoiletries.com/research/chemistry/Preserving-the-Facts-on-Parabens-An-Overview-of-These-Important-Tools-of-the Trade-425784294.html. Published June 1, 2017. Accessed December 20, 2017.
- Routledge EJ, Parker J, Odum J, et al. Some alkyl hydroxy benzoate preservatives (parabens) are estrogenic. Toxicol Appl Pharmacol. 1998;153:12Y19.
- Hossaini A, Larsen JJ, Larsen JC. Lack of oestrogenic effects of food preservatives (parabens) in uterotrophic assays. Food Chem Toxicol. 2000;38:319-323.
- Cosmetic Ingredient Review. Final report on the safety assessment of methylparaben, ethylparaben, propylparaben and butylparaben. J Am Coll Toxicol. 1984;3:147-209.
- Cosmetic Ingredient Review. Final report on the safety assessment of isobutylparaben and isopropylparaben. J Am Coll Toxicol. 1995;14:364-372.
- Scientific Committee on Consumer Products. Extended Opinion on the Safety Evaluation of Parabens. European Commission website. https://ec.europa.eu/health/ph_risk/committees/04_sccp/docs/sccp_o_019.pdf. Published January 28, 2005. Accessed December 20, 2017.
- Scientific Committee on Consumer Products. Opinion on Parabens. European Commission website. http://ec.europa.eu/health/scientific_committees/consumer_safety/docs/sccs_o_041.pdf. Revised March 22, 2011. Accessed December 20, 2017.
- European Commission. Commission Regulation (EU) No 258/2014 of 9 April 2014 amending Annexes II and V to Regulation (EC) No 1223/2009 of the European Parliament and of the Council on cosmetic products. EUR-Lex website. http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=uriserv:OJ.L_.2014.107.01.0005.01.ENG. Accessed December 20, 2017.
- American Cancer Society. Antiperspirants and breast cancer risk. https://www.cancer.org/cancer/cancer-causes/antiperspirants-and-breast-cancer-risk.html#references. Revised October 14, 2014. Accessed January 2, 2018.
- MacMillan A. Cutting back on shampoo? 15 things you should know. Health. February 25, 2014. http://www.health.com/health/gallery/0,,20788089,00.html#should-you-go-no-poo--1. Accessed December 10, 2017.
- The ‘no poo’ method. https://www.nopoomethod.com/. Accessed December 10, 2017.
- Fong, D, Gaulin C, Le M, et al. Effectiveness of alternative antimicrobial agents for disinfection of hard surfaces. National Collaborating Centre for Environmental Health website. http://www.ncceh.ca/sites/default/files/Alternative_Antimicrobial_Agents_Aug_2014.pdf. Published August 2014. Accessed December 10, 2017.
- Is baking soda too harsh for natural hair? Black Girl With Long Hair website. http://blackgirllonghair.com/2012/02/is-baking-soda-too-harsh-for-hair/2/. Published February 5, 2012. Accessed December 12, 2017.
- O’Lenick T. Anionic/cationic complexes in hair care. J Cosmet Sci. 2011;62:209-228.
- Gavazzoni Dias MF, de Almeida AM, Cecato PM, et al. The shampoo pH can affect the hair: myth or reality? Int J Trichology. 2014;6:95-99.
- Goodman H. The acid mantle of the skin surface. Ind Med Surg. 1958;27:105-108.
- Korting HC, Kober M, Mueller M, et al. Influence of repeated washings with soap and synthetic detergents on pH and resident flora of the skin of forehead and forearm. results of a cross-over trial in health probationers. Acta Derm Venereol. 1987;67:41-47.
- Tarun J, Susan J, Suria J, et al. Evaluation of pH of bathing soaps and shampoos for skin and hair care. Indian J Dermatol. 2014;59:442-444.
- Corbett JF. The chemistry of hair-care products. J Soc Dyers Colour. 1976;92:285-303.
- McMichael AJ, Hordinsky M. Hair Diseases: Medical, Surgical, and Cosmetic Treatments. New York, NY: Taylor & Francis; 2008:59-72.
- Allardice A, Gummo G. Hair conditioning: quaternary ammonium compounds on various hair types. Cosmet Toiletries. 1993;108:107-109.
- Larson D, Jacob SE. Tea tree oil. Dermatitis. 2012;23:48-49.
Practice Points
- The ingredients in shampoos and other cosmetic products have become scrutinized by the general public and the Internet has contributed to misinformation about certain shampoos.
- Dermatologists must be prepared to acknowledge the concerns that their patients have about common shampoo ingredients to dispel the myths that may misinform patient decision-making.
- This article reviews the controversy surrounding the use of sulfates and parabens in shampoos, as well as commonly used shampoo alternatives, often called the “no-poo” method.