User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
FDA approves tirzepatide for treating obesity
Eli Lilly will market tirzepatide injections for weight management under the trade name Zepbound. It was approved in May 2022 for treating type 2 diabetes. The new indication is for adults with either obesity, defined as a body mass index of 30 kg/m2 or greater, or overweight, with a BMI of 27 or greater with at least one weight-related comorbidity, including hypertension, type 2 diabetes, or dyslipidemia.
“Obesity and overweight are serious conditions that can be associated with some of the leading causes of death, such as heart disease, stroke, and diabetes,” said John Sharretts, MD, director of the division of diabetes, lipid disorders, and obesity in the FDA’s Center for Drug Evaluation and Research. “In light of increasing rates of both obesity and overweight in the United States, today’s approval addresses an unmet medical need.”
A once-weekly injection, tirzepatide reduces appetite by activating two gut hormones, glucagonlike peptide–1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). The dosage is increased over 4-20 weeks to achieve a weekly dose target of 5 mg, 10 mg, or 15 mg maximum.
Efficacy was established in two pivotal randomized, double-blind, placebo-controlled trials of adults with obesity or overweight plus another condition. One trial measured weight reduction after 72 weeks in a total of 2,519 patients without diabetes who received either 5 mg, 10 mg or 15 mg of tirzepatide once weekly. Those who received the 15-mg dose achieved on average 18% of their initial body weight, compared with placebo.
The other pivotal trial enrolled a total of 938 patients with type 2 diabetes. These patients achieved an average weight loss of 12% with once-weekly tirzepatide compared to placebo.
Another trial, which was presented at the 2023 Obesity Week meeting and was published in Nature Medicine, showed clinically meaningful added weight loss for adults with obesity who did not have diabetes and who had already experienced weight loss of at least 5% after a 12-week intensive lifestyle intervention.
Another trial, which was reported at the 2023 annual meeting of the European Association for the Study of Diabetes, found that tirzepatide continued to produce “highly significant weight loss” when the drug was continued in a 1-year follow-up trial. Those who discontinued taking the drug regained some weight but not all.
Tirzepatide can cause gastrointestinal side effects, such as nausea, diarrhea, vomiting, constipation, and abdominal pain or discomfort. Site reactions, hypersensitivity, hair loss, burping, and gastrointestinal reflux disease have also been reported.
The medication should not be used by patients with a personal or family history of medullary thyroid cancer or by patients with multiple endocrine neoplasia syndrome type 2. It should also not be used in combination with Mounjaro or another GLP-1 receptor agonist. The safety and effectiveness of the coadministration of tirzepatide with other medications for weight management have not been established.
Zepbound should go to market in the United States by the end of 2023, with an anticipated monthly list price of $1,060, according to a news release from Eli Lilly.
A version of this article first appeared on Medscape.com.
Eli Lilly will market tirzepatide injections for weight management under the trade name Zepbound. It was approved in May 2022 for treating type 2 diabetes. The new indication is for adults with either obesity, defined as a body mass index of 30 kg/m2 or greater, or overweight, with a BMI of 27 or greater with at least one weight-related comorbidity, including hypertension, type 2 diabetes, or dyslipidemia.
“Obesity and overweight are serious conditions that can be associated with some of the leading causes of death, such as heart disease, stroke, and diabetes,” said John Sharretts, MD, director of the division of diabetes, lipid disorders, and obesity in the FDA’s Center for Drug Evaluation and Research. “In light of increasing rates of both obesity and overweight in the United States, today’s approval addresses an unmet medical need.”
A once-weekly injection, tirzepatide reduces appetite by activating two gut hormones, glucagonlike peptide–1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). The dosage is increased over 4-20 weeks to achieve a weekly dose target of 5 mg, 10 mg, or 15 mg maximum.
Efficacy was established in two pivotal randomized, double-blind, placebo-controlled trials of adults with obesity or overweight plus another condition. One trial measured weight reduction after 72 weeks in a total of 2,519 patients without diabetes who received either 5 mg, 10 mg or 15 mg of tirzepatide once weekly. Those who received the 15-mg dose achieved on average 18% of their initial body weight, compared with placebo.
The other pivotal trial enrolled a total of 938 patients with type 2 diabetes. These patients achieved an average weight loss of 12% with once-weekly tirzepatide compared to placebo.
Another trial, which was presented at the 2023 Obesity Week meeting and was published in Nature Medicine, showed clinically meaningful added weight loss for adults with obesity who did not have diabetes and who had already experienced weight loss of at least 5% after a 12-week intensive lifestyle intervention.
Another trial, which was reported at the 2023 annual meeting of the European Association for the Study of Diabetes, found that tirzepatide continued to produce “highly significant weight loss” when the drug was continued in a 1-year follow-up trial. Those who discontinued taking the drug regained some weight but not all.
Tirzepatide can cause gastrointestinal side effects, such as nausea, diarrhea, vomiting, constipation, and abdominal pain or discomfort. Site reactions, hypersensitivity, hair loss, burping, and gastrointestinal reflux disease have also been reported.
The medication should not be used by patients with a personal or family history of medullary thyroid cancer or by patients with multiple endocrine neoplasia syndrome type 2. It should also not be used in combination with Mounjaro or another GLP-1 receptor agonist. The safety and effectiveness of the coadministration of tirzepatide with other medications for weight management have not been established.
Zepbound should go to market in the United States by the end of 2023, with an anticipated monthly list price of $1,060, according to a news release from Eli Lilly.
A version of this article first appeared on Medscape.com.
Eli Lilly will market tirzepatide injections for weight management under the trade name Zepbound. It was approved in May 2022 for treating type 2 diabetes. The new indication is for adults with either obesity, defined as a body mass index of 30 kg/m2 or greater, or overweight, with a BMI of 27 or greater with at least one weight-related comorbidity, including hypertension, type 2 diabetes, or dyslipidemia.
“Obesity and overweight are serious conditions that can be associated with some of the leading causes of death, such as heart disease, stroke, and diabetes,” said John Sharretts, MD, director of the division of diabetes, lipid disorders, and obesity in the FDA’s Center for Drug Evaluation and Research. “In light of increasing rates of both obesity and overweight in the United States, today’s approval addresses an unmet medical need.”
A once-weekly injection, tirzepatide reduces appetite by activating two gut hormones, glucagonlike peptide–1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). The dosage is increased over 4-20 weeks to achieve a weekly dose target of 5 mg, 10 mg, or 15 mg maximum.
Efficacy was established in two pivotal randomized, double-blind, placebo-controlled trials of adults with obesity or overweight plus another condition. One trial measured weight reduction after 72 weeks in a total of 2,519 patients without diabetes who received either 5 mg, 10 mg or 15 mg of tirzepatide once weekly. Those who received the 15-mg dose achieved on average 18% of their initial body weight, compared with placebo.
The other pivotal trial enrolled a total of 938 patients with type 2 diabetes. These patients achieved an average weight loss of 12% with once-weekly tirzepatide compared to placebo.
Another trial, which was presented at the 2023 Obesity Week meeting and was published in Nature Medicine, showed clinically meaningful added weight loss for adults with obesity who did not have diabetes and who had already experienced weight loss of at least 5% after a 12-week intensive lifestyle intervention.
Another trial, which was reported at the 2023 annual meeting of the European Association for the Study of Diabetes, found that tirzepatide continued to produce “highly significant weight loss” when the drug was continued in a 1-year follow-up trial. Those who discontinued taking the drug regained some weight but not all.
Tirzepatide can cause gastrointestinal side effects, such as nausea, diarrhea, vomiting, constipation, and abdominal pain or discomfort. Site reactions, hypersensitivity, hair loss, burping, and gastrointestinal reflux disease have also been reported.
The medication should not be used by patients with a personal or family history of medullary thyroid cancer or by patients with multiple endocrine neoplasia syndrome type 2. It should also not be used in combination with Mounjaro or another GLP-1 receptor agonist. The safety and effectiveness of the coadministration of tirzepatide with other medications for weight management have not been established.
Zepbound should go to market in the United States by the end of 2023, with an anticipated monthly list price of $1,060, according to a news release from Eli Lilly.
A version of this article first appeared on Medscape.com.
Review estimates acne risk with JAK inhibitor therapy
TOPLINE:
, according to an analysis of 25 JAK inhibitor studies.
METHODOLOGY:
- Acne has been reported to be an adverse effect of JAK inhibitors, but not much is known about how common acne is overall and how incidence differs between different JAK inhibitors and the disease being treated.
- For the systematic review and meta-analysis, researchers identified 25 phase 2 or 3 randomized, controlled trials that reported acne as an adverse event associated with the use of JAK inhibitors.
- The study population included 10,839 participants (54% male, 46% female).
- The primary outcome was the incidence of acne following a period of JAK inhibitor use.
TAKEAWAY:
- Overall, the risk of acne was significantly higher among those treated with JAK inhibitors in comparison with patients given placebo in a pooled analysis (odds ratio [OR], 3.83).
- The risk of acne was highest with abrocitinib (OR, 13.47), followed by baricitinib (OR, 4.96), upadacitinib (OR, 4.79), deuruxolitinib (OR, 3.30), and deucravacitinib (OR, 2.64). By JAK inhibitor class, results were as follows: JAK1-specific inhibitors (OR, 4.69), combined JAK1 and JAK2 inhibitors (OR, 3.43), and tyrosine kinase 2 inhibitors (OR, 2.64).
- In a subgroup analysis, risk of acne was higher among patients using JAK inhibitors for dermatologic conditions in comparison with those using JAK inhibitors for nondermatologic conditions (OR, 4.67 vs 1.18).
- Age and gender had no apparent impact on the effect of JAK inhibitor use on acne risk.
IN PRACTICE:
“The occurrence of acne following treatment with certain classes of JAK inhibitors is of potential concern, as this adverse effect may jeopardize treatment adherence among some patients,” the researchers wrote. More studies are needed “to characterize the underlying mechanism of acne with JAK inhibitor use and to identify best practices for treatment,” they added.
SOURCE:
The lead author was Jeremy Martinez, MPH, of Harvard Medical School, Boston. The study was published online in JAMA Dermatology.
LIMITATIONS:
The review was limited by the variable classification and reporting of acne across studies, the potential exclusion of relevant studies, and the small number of studies for certain drugs.
DISCLOSURES:
The studies were mainly funded by the pharmaceutical industry. Mr. Martinez disclosed no relevant financial relationships. Several coauthors have ties with Dexcel Pharma Technologies, AbbVie, Concert, Pfizer, 3Derm Systems, Incyte, Aclaris, Eli Lilly, Concert, Equillium, ASLAN, ACOM, and Boehringer Ingelheim.
A version of this article appeared on Medscape.com.
TOPLINE:
, according to an analysis of 25 JAK inhibitor studies.
METHODOLOGY:
- Acne has been reported to be an adverse effect of JAK inhibitors, but not much is known about how common acne is overall and how incidence differs between different JAK inhibitors and the disease being treated.
- For the systematic review and meta-analysis, researchers identified 25 phase 2 or 3 randomized, controlled trials that reported acne as an adverse event associated with the use of JAK inhibitors.
- The study population included 10,839 participants (54% male, 46% female).
- The primary outcome was the incidence of acne following a period of JAK inhibitor use.
TAKEAWAY:
- Overall, the risk of acne was significantly higher among those treated with JAK inhibitors in comparison with patients given placebo in a pooled analysis (odds ratio [OR], 3.83).
- The risk of acne was highest with abrocitinib (OR, 13.47), followed by baricitinib (OR, 4.96), upadacitinib (OR, 4.79), deuruxolitinib (OR, 3.30), and deucravacitinib (OR, 2.64). By JAK inhibitor class, results were as follows: JAK1-specific inhibitors (OR, 4.69), combined JAK1 and JAK2 inhibitors (OR, 3.43), and tyrosine kinase 2 inhibitors (OR, 2.64).
- In a subgroup analysis, risk of acne was higher among patients using JAK inhibitors for dermatologic conditions in comparison with those using JAK inhibitors for nondermatologic conditions (OR, 4.67 vs 1.18).
- Age and gender had no apparent impact on the effect of JAK inhibitor use on acne risk.
IN PRACTICE:
“The occurrence of acne following treatment with certain classes of JAK inhibitors is of potential concern, as this adverse effect may jeopardize treatment adherence among some patients,” the researchers wrote. More studies are needed “to characterize the underlying mechanism of acne with JAK inhibitor use and to identify best practices for treatment,” they added.
SOURCE:
The lead author was Jeremy Martinez, MPH, of Harvard Medical School, Boston. The study was published online in JAMA Dermatology.
LIMITATIONS:
The review was limited by the variable classification and reporting of acne across studies, the potential exclusion of relevant studies, and the small number of studies for certain drugs.
DISCLOSURES:
The studies were mainly funded by the pharmaceutical industry. Mr. Martinez disclosed no relevant financial relationships. Several coauthors have ties with Dexcel Pharma Technologies, AbbVie, Concert, Pfizer, 3Derm Systems, Incyte, Aclaris, Eli Lilly, Concert, Equillium, ASLAN, ACOM, and Boehringer Ingelheim.
A version of this article appeared on Medscape.com.
TOPLINE:
, according to an analysis of 25 JAK inhibitor studies.
METHODOLOGY:
- Acne has been reported to be an adverse effect of JAK inhibitors, but not much is known about how common acne is overall and how incidence differs between different JAK inhibitors and the disease being treated.
- For the systematic review and meta-analysis, researchers identified 25 phase 2 or 3 randomized, controlled trials that reported acne as an adverse event associated with the use of JAK inhibitors.
- The study population included 10,839 participants (54% male, 46% female).
- The primary outcome was the incidence of acne following a period of JAK inhibitor use.
TAKEAWAY:
- Overall, the risk of acne was significantly higher among those treated with JAK inhibitors in comparison with patients given placebo in a pooled analysis (odds ratio [OR], 3.83).
- The risk of acne was highest with abrocitinib (OR, 13.47), followed by baricitinib (OR, 4.96), upadacitinib (OR, 4.79), deuruxolitinib (OR, 3.30), and deucravacitinib (OR, 2.64). By JAK inhibitor class, results were as follows: JAK1-specific inhibitors (OR, 4.69), combined JAK1 and JAK2 inhibitors (OR, 3.43), and tyrosine kinase 2 inhibitors (OR, 2.64).
- In a subgroup analysis, risk of acne was higher among patients using JAK inhibitors for dermatologic conditions in comparison with those using JAK inhibitors for nondermatologic conditions (OR, 4.67 vs 1.18).
- Age and gender had no apparent impact on the effect of JAK inhibitor use on acne risk.
IN PRACTICE:
“The occurrence of acne following treatment with certain classes of JAK inhibitors is of potential concern, as this adverse effect may jeopardize treatment adherence among some patients,” the researchers wrote. More studies are needed “to characterize the underlying mechanism of acne with JAK inhibitor use and to identify best practices for treatment,” they added.
SOURCE:
The lead author was Jeremy Martinez, MPH, of Harvard Medical School, Boston. The study was published online in JAMA Dermatology.
LIMITATIONS:
The review was limited by the variable classification and reporting of acne across studies, the potential exclusion of relevant studies, and the small number of studies for certain drugs.
DISCLOSURES:
The studies were mainly funded by the pharmaceutical industry. Mr. Martinez disclosed no relevant financial relationships. Several coauthors have ties with Dexcel Pharma Technologies, AbbVie, Concert, Pfizer, 3Derm Systems, Incyte, Aclaris, Eli Lilly, Concert, Equillium, ASLAN, ACOM, and Boehringer Ingelheim.
A version of this article appeared on Medscape.com.
Newer antiobesity meds lower the body’s defended fat mass
The current highly effective antiobesity medications approved for treating obesity (semaglutide), under review (tirzepatide), or in late-stage clinical trials “appear to lower the body’s target and defended fat mass [set point]” but do not permanently fix it at a lower point, Lee M. Kaplan, MD, PhD, explained in a lecture during the annual meeting of the Obesity Society.
It is very likely that patients with obesity will have to take these antiobesity medications “forever,” he said, “until we identify and can repair the cellular and molecular mechanisms that the body uses to regulate body fat mass throughout the life cycle and that are dysfunctional in obesity.”
“The body is able to regulate fat mass at multiple stages during development,” Dr. Kaplan, from Massachusetts General Hospital and Harvard Medical School, Boston, explained, “and when it doesn’t do it appropriately, that becomes the physiological basis of obesity.”
The loss of baby fat, as well as fat changes during puberty, menopause, aging, and, in particular, during and after pregnancy, “all occur without conscious or purposeful input,” he noted.
The body uses food intake and energy expenditure to reach and defend its intended fat mass, and there is an evolutionary benefit to doing this.
For example, people recovering from an acute illness can regain the lost fat and weight. A woman can support a pregnancy and lactation by increasing fat mass.
However, “the idea that [with antiobesity medications] we should be aiming for a fixed lower amount of fat is probably not a good idea.” Dr. Kaplan cautioned.
People need the flexibility to recover lost fat and weight after an acute illness or injury, and pregnant women need to gain an appropriate amount of body fat to support pregnancy and lactation.
Intermittent therapy: A practical strategy?
The long-term benefit of antiobesity medications requires continuous use, Dr. Kaplan noted. For example, in the STEP 1 trial of semaglutide in patients with obesity and without diabetes, when treatment was stopped at 68 weeks, average weight increased through 120 weeks, although it did not return to baseline levels.
Intermittent antiobesity therapy may be an effective, “very practical strategy” to maintain weight loss, which would also “address current challenges of high cost, limited drug availability, and inadequate access to care.”
“Until we have strategies for decreasing the cost of effective obesity treatment, and ensuring more equitable access to obesity care,” Dr. Kaplan said, “optimizing algorithms for the use of intermittent therapy may be an effective stopgap measure.”
Dr. Kaplan is or has recently been a paid consultant for Eli Lilly, Novo Nordisk, and multiple pharmaceutical companies developing antiobesity medications.
A version of this article appeared on Medscape.com.
The current highly effective antiobesity medications approved for treating obesity (semaglutide), under review (tirzepatide), or in late-stage clinical trials “appear to lower the body’s target and defended fat mass [set point]” but do not permanently fix it at a lower point, Lee M. Kaplan, MD, PhD, explained in a lecture during the annual meeting of the Obesity Society.
It is very likely that patients with obesity will have to take these antiobesity medications “forever,” he said, “until we identify and can repair the cellular and molecular mechanisms that the body uses to regulate body fat mass throughout the life cycle and that are dysfunctional in obesity.”
“The body is able to regulate fat mass at multiple stages during development,” Dr. Kaplan, from Massachusetts General Hospital and Harvard Medical School, Boston, explained, “and when it doesn’t do it appropriately, that becomes the physiological basis of obesity.”
The loss of baby fat, as well as fat changes during puberty, menopause, aging, and, in particular, during and after pregnancy, “all occur without conscious or purposeful input,” he noted.
The body uses food intake and energy expenditure to reach and defend its intended fat mass, and there is an evolutionary benefit to doing this.
For example, people recovering from an acute illness can regain the lost fat and weight. A woman can support a pregnancy and lactation by increasing fat mass.
However, “the idea that [with antiobesity medications] we should be aiming for a fixed lower amount of fat is probably not a good idea.” Dr. Kaplan cautioned.
People need the flexibility to recover lost fat and weight after an acute illness or injury, and pregnant women need to gain an appropriate amount of body fat to support pregnancy and lactation.
Intermittent therapy: A practical strategy?
The long-term benefit of antiobesity medications requires continuous use, Dr. Kaplan noted. For example, in the STEP 1 trial of semaglutide in patients with obesity and without diabetes, when treatment was stopped at 68 weeks, average weight increased through 120 weeks, although it did not return to baseline levels.
Intermittent antiobesity therapy may be an effective, “very practical strategy” to maintain weight loss, which would also “address current challenges of high cost, limited drug availability, and inadequate access to care.”
“Until we have strategies for decreasing the cost of effective obesity treatment, and ensuring more equitable access to obesity care,” Dr. Kaplan said, “optimizing algorithms for the use of intermittent therapy may be an effective stopgap measure.”
Dr. Kaplan is or has recently been a paid consultant for Eli Lilly, Novo Nordisk, and multiple pharmaceutical companies developing antiobesity medications.
A version of this article appeared on Medscape.com.
The current highly effective antiobesity medications approved for treating obesity (semaglutide), under review (tirzepatide), or in late-stage clinical trials “appear to lower the body’s target and defended fat mass [set point]” but do not permanently fix it at a lower point, Lee M. Kaplan, MD, PhD, explained in a lecture during the annual meeting of the Obesity Society.
It is very likely that patients with obesity will have to take these antiobesity medications “forever,” he said, “until we identify and can repair the cellular and molecular mechanisms that the body uses to regulate body fat mass throughout the life cycle and that are dysfunctional in obesity.”
“The body is able to regulate fat mass at multiple stages during development,” Dr. Kaplan, from Massachusetts General Hospital and Harvard Medical School, Boston, explained, “and when it doesn’t do it appropriately, that becomes the physiological basis of obesity.”
The loss of baby fat, as well as fat changes during puberty, menopause, aging, and, in particular, during and after pregnancy, “all occur without conscious or purposeful input,” he noted.
The body uses food intake and energy expenditure to reach and defend its intended fat mass, and there is an evolutionary benefit to doing this.
For example, people recovering from an acute illness can regain the lost fat and weight. A woman can support a pregnancy and lactation by increasing fat mass.
However, “the idea that [with antiobesity medications] we should be aiming for a fixed lower amount of fat is probably not a good idea.” Dr. Kaplan cautioned.
People need the flexibility to recover lost fat and weight after an acute illness or injury, and pregnant women need to gain an appropriate amount of body fat to support pregnancy and lactation.
Intermittent therapy: A practical strategy?
The long-term benefit of antiobesity medications requires continuous use, Dr. Kaplan noted. For example, in the STEP 1 trial of semaglutide in patients with obesity and without diabetes, when treatment was stopped at 68 weeks, average weight increased through 120 weeks, although it did not return to baseline levels.
Intermittent antiobesity therapy may be an effective, “very practical strategy” to maintain weight loss, which would also “address current challenges of high cost, limited drug availability, and inadequate access to care.”
“Until we have strategies for decreasing the cost of effective obesity treatment, and ensuring more equitable access to obesity care,” Dr. Kaplan said, “optimizing algorithms for the use of intermittent therapy may be an effective stopgap measure.”
Dr. Kaplan is or has recently been a paid consultant for Eli Lilly, Novo Nordisk, and multiple pharmaceutical companies developing antiobesity medications.
A version of this article appeared on Medscape.com.
FROM OBESITYWEEK® 2023
Pustular Eruption on the Face
The Diagnosis: Eczema Herpeticum
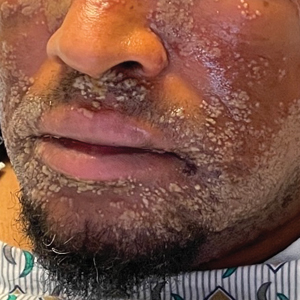
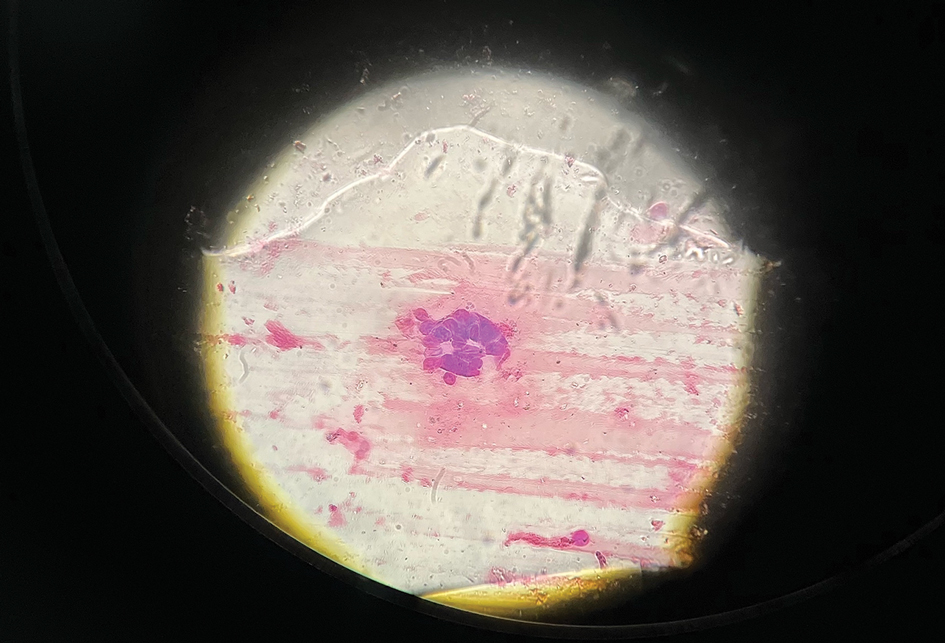
The patient’s condition with worsening facial edema and notable pain prompted a bedside Tzanck smear using a sample from the base of a deroofed forehead vesicle. In addition, a swab of a deroofed lesion was sent for herpes simplex virus and varicella-zoster virus (VZV) polymerase chain reaction (PCR) testing. The Tzanck smear demonstrated ballooning multinucleated syncytial giant cells and eosinophilic inclusion bodies (Figure), which are characteristic of certain herpesviruses including herpes simplex virus and VZV. He was started on intravenous acyclovir while PCR results were pending; the PCR test later confirmed positivity for herpes simplex virus type 1. Treatment was transitioned to oral valacyclovir once the lesions started crusting over. Notable healing and epithelialization of the lesions occurred during his hospital stay, and he was discharged home 5 days after starting treatment. He was counseled on autoinoculation, advised that he was considered infectious until all lesions had crusted over, and encouraged to employ frequent handwashing. Complete resolution of eczema herpeticum (EH) was noted at 3-week follow-up.
Eczema herpeticum (also known as Kaposi varicelliform eruption) is a potentially life-threatening disseminated cutaneous infection caused by herpes simplex virus types 1 and 2 in patients with pre-existing skin disease.1 It typically presents as a complication of atopic dermatitis (AD) but also has been identified as a rare complication in other conditions that disrupt the normal skin barrier, including mycosis fungoides, pemphigus foliaceus, pemphigus vulgaris, Darier disease, pityriasis rubra pilaris, contact dermatitis, and seborrheic dermatitis.1-4
The pathogenesis of EH is multifactorial. Disruption of the stratum corneum; impaired natural killer cell function; early-onset, untreated, or severe AD; disrupted skin microbiota with skewed colonization by Staphylococcus aureus; immunosuppressive AD therapies such as calcineurin inhibitors; eosinophilia; and helper T cell (TH2) cytokine predominance all have been suggested to play a role in the development of EH.5-8
As seen in our patient, EH presents with a sudden eruption of painful or pruritic, grouped, monomorphic, domeshaped vesicles with background swelling and erythema typically on the head, neck, and trunk. Vesicles then progress to punched-out erosions with overlying hemorrhagic crusting that can coalesce to form large denuded areas susceptible to superinfection with bacteria.9 Other accompanying symptoms include high fever, chills, malaise, and lymphadenopathy. Associated inflammation, classically described as erythema, may be difficult to discern in patients with darker skin and appears as hyperpigmentation; therefore, identification of clusters of monomorphic vesicles in areas of pre-existing dermatitis is particularly important for clinical diagnosis in people with darker skin types.
Various tests are available to confirm diagnosis in ambiguous cases. Bedside Tzanck smears can be performed rapidly and are considered positive if characteristic multinucleated giant cells are noted; however, they do not differentiate between the various herpesviruses. Direct fluorescent antibody testing of scraped lesions and viral cultures of swabbed vesicular fluid are equally effective in distinguishing between herpes simplex virus type 1, herpes simplex virus type 2, and VZV; PCR confirms the diagnosis with high specificity and sensitivity.10
In our patient, the initial differential diagnosis included EH, acute generalized exanthematous pustulosis, allergic contact dermatitis, and Orthopoxvirus infection. The positive Tzanck smear reduced the likelihood of a nonviral etiology. Additionally, worsening of the rash despite discontinuation of medications and utilization of topical steroids argued against acute generalized exanthematous pustulosis and allergic contact dermatitis. The laboratory findings reduced the likelihood of drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome, and PCR findings ultimately ruled out Orthopoxvirus infections. Additional differential diagnoses for EH include dermatitis herpetiformis; primary VZV infection; hand, foot, and mouth disease; disseminated zoster infection; disseminated molluscum contagiosum; and eczema coxsackium.
Complications of EH include scarring; herpetic keratitis due to corneal infection, which if left untreated can progress to blindness; and rarely death due to multiorgan failure or septicemia.11 The traditional smallpox vaccine (ACAM2000) is contraindicated in patients with AD and EH, even when AD is in remission. These patients should avoid contact with recently vaccinated individuals.12 An alternative vaccine—Jynneos (Bavarian Nordic)—is available for these patients and their family members.13 Clinicians should be aware of this guideline, especially given the recent mpox (monkeypox) outbreaks.
Mild cases of EH are more common, may sometimes go unnoticed, and self-resolve in healthy patients. Severe cases may require systemic antiviral therapy. Acyclovir and its prodrug valacyclovir are standard treatments for EH. Alternatively, foscarnet or cidofovir can be used in the treatment of acyclovir-resistant thymidine kinase– deficient herpes simplex virus and other acyclovirresistant cases.14 Any secondary bacterial superinfections, usually due to staphylococcal or streptococcal bacteria, should be treated with antibiotics. A thorough ophthalmologic evaluation should be performed for patients with periocular involvement of EH. Empiric treatment should be started immediately, given a relative low toxicity of systemic antiviral therapy and high morbidity and mortality associated with untreated widespread EH.
It is important to maintain a high index of clinical suspicion for EH, especially in patients with pre-existing conditions such as AD who present with systemic symptoms and facial vesicles, pustules, or erosions to ensure prompt diagnosis and appropriate treatment.
- Baaniya B, Agrawal S. Kaposi varicelliform eruption in a patient with pemphigus vulgaris: a case report and review of the literature. Case Rep Dermatol Med. 2020;2020:6695342. doi:10.1155/2020/6695342
- Tayabali K, Pothiwalla H, Lowitt M. Eczema herpeticum in Darier’s disease: a topical storm. J Community Hosp Intern Med Perspect. 2019;9:347. doi:10.1080/20009666.2019.1650590
- Cavalié M, Giacchero D, Cardot-Leccia N, et al. Kaposi’s varicelliform eruption in a patient with pityriasis rubra pilaris (pityriasis rubra pilaris herpeticum). J Eur Acad Dermatol Venereol. 2013;27:1585-1586. doi:10.1111/JDV.12120
- Lee GH, Kim YM, Lee SY, et al. A case of eczema herpeticum with Hailey-Hailey disease. Ann Dermatol. 2009;21:311-314. doi:10.5021/ad.2009.21.3.311
- Seegräber M, Worm M, Werfel T, et al. Recurrent eczema herpeticum— a retrospective European multicenter study evaluating the clinical characteristics of eczema herpeticum cases in atopic dermatitis patients. J Eur Acad Dermatol Venereol. 2020;34:1074-1079. doi:10.1111/JDV.16090
- Kawakami Y, Ando T, Lee J-R, et al. Defective natural killer cell activity in a mouse model of eczema herpeticum. J Allergy Clin Immunol. 2017;139:997-1006.e10. doi:10.1016/j.jaci.2016.06.034
- Beck L, Latchney L, Zaccaro D, et al. Biomarkers of disease severity and Th2 polarity are predictors of risk for eczema herpeticum. J Allergy Clin Immunol. 2008;121:S37-S37. doi:10.1016/j.jaci.2007.12.152
- Kim M, Jung M, Hong SP, et al. Topical calcineurin inhibitors compromise stratum corneum integrity, epidermal permeability and antimicrobial barrier function. Exp Dermatol. 2010; 19:501-510. doi:10.1111/J.1600-0625.2009.00941.X
- Karray M, Kwan E, Souissi A. Kaposi varicelliform eruption. StatPearls [Internet]. StatPearls Publishing; 2023. https://www.ncbi.nlm.nih.gov/books/NBK482432/
- Dominguez SR, Pretty K, Hengartner R, et al. Comparison of herpes simplex virus PCR with culture for virus detection in multisource surface swab specimens from neonates [published online September 25, 2018]. J Clin Microbiol. doi:10.1128/JCM.00632-18
- Feye F, De Halleux C, Gillet JB, et al. Exacerbation of atopic dermatitis in the emergency department. Eur J Emerg Med. 2004;11:49-52. doi:10.1097/00063110-200412000-00014
- Casey C, Vellozzi C, Mootrey GT, et al; Vaccinia Case Definition Development Working Group; Advisory Committee on Immunization Practices-Armed Forces Epidemiological Board Smallpox Vaccine Safety Working Group. Surveillance guidelines for smallpox vaccine (vaccinia) adverse reactions. MMWR Recomm Rep. 2006;55:1-16.
- Rao AK, Petersen BW, Whitehill F, et al. Use of JYNNEOS (Smallpox and Monkeypox Vaccine, Live, Nonreplicating) for preexposure vaccination of persons at risk for occupational exposure to orthopoxviruses: recommendations of the Advisory Committee on Immunization Practices—United States, 2022. MMWR Morb Mortal Wkly Rep. 2022;71:734-742. doi:10.15585 /MMWR.MM7122E1
- Piret J, Boivin G. Resistance of herpes simplex viruses to nucleoside analogues: mechanisms, prevalence, and management. Antimicrob Agents Chemother. 2011;55:459. doi:10.1128/AAC.00615-10
The Diagnosis: Eczema Herpeticum
The patient’s condition with worsening facial edema and notable pain prompted a bedside Tzanck smear using a sample from the base of a deroofed forehead vesicle. In addition, a swab of a deroofed lesion was sent for herpes simplex virus and varicella-zoster virus (VZV) polymerase chain reaction (PCR) testing. The Tzanck smear demonstrated ballooning multinucleated syncytial giant cells and eosinophilic inclusion bodies (Figure), which are characteristic of certain herpesviruses including herpes simplex virus and VZV. He was started on intravenous acyclovir while PCR results were pending; the PCR test later confirmed positivity for herpes simplex virus type 1. Treatment was transitioned to oral valacyclovir once the lesions started crusting over. Notable healing and epithelialization of the lesions occurred during his hospital stay, and he was discharged home 5 days after starting treatment. He was counseled on autoinoculation, advised that he was considered infectious until all lesions had crusted over, and encouraged to employ frequent handwashing. Complete resolution of eczema herpeticum (EH) was noted at 3-week follow-up.

Eczema herpeticum (also known as Kaposi varicelliform eruption) is a potentially life-threatening disseminated cutaneous infection caused by herpes simplex virus types 1 and 2 in patients with pre-existing skin disease.1 It typically presents as a complication of atopic dermatitis (AD) but also has been identified as a rare complication in other conditions that disrupt the normal skin barrier, including mycosis fungoides, pemphigus foliaceus, pemphigus vulgaris, Darier disease, pityriasis rubra pilaris, contact dermatitis, and seborrheic dermatitis.1-4
The pathogenesis of EH is multifactorial. Disruption of the stratum corneum; impaired natural killer cell function; early-onset, untreated, or severe AD; disrupted skin microbiota with skewed colonization by Staphylococcus aureus; immunosuppressive AD therapies such as calcineurin inhibitors; eosinophilia; and helper T cell (TH2) cytokine predominance all have been suggested to play a role in the development of EH.5-8
As seen in our patient, EH presents with a sudden eruption of painful or pruritic, grouped, monomorphic, domeshaped vesicles with background swelling and erythema typically on the head, neck, and trunk. Vesicles then progress to punched-out erosions with overlying hemorrhagic crusting that can coalesce to form large denuded areas susceptible to superinfection with bacteria.9 Other accompanying symptoms include high fever, chills, malaise, and lymphadenopathy. Associated inflammation, classically described as erythema, may be difficult to discern in patients with darker skin and appears as hyperpigmentation; therefore, identification of clusters of monomorphic vesicles in areas of pre-existing dermatitis is particularly important for clinical diagnosis in people with darker skin types.
Various tests are available to confirm diagnosis in ambiguous cases. Bedside Tzanck smears can be performed rapidly and are considered positive if characteristic multinucleated giant cells are noted; however, they do not differentiate between the various herpesviruses. Direct fluorescent antibody testing of scraped lesions and viral cultures of swabbed vesicular fluid are equally effective in distinguishing between herpes simplex virus type 1, herpes simplex virus type 2, and VZV; PCR confirms the diagnosis with high specificity and sensitivity.10
In our patient, the initial differential diagnosis included EH, acute generalized exanthematous pustulosis, allergic contact dermatitis, and Orthopoxvirus infection. The positive Tzanck smear reduced the likelihood of a nonviral etiology. Additionally, worsening of the rash despite discontinuation of medications and utilization of topical steroids argued against acute generalized exanthematous pustulosis and allergic contact dermatitis. The laboratory findings reduced the likelihood of drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome, and PCR findings ultimately ruled out Orthopoxvirus infections. Additional differential diagnoses for EH include dermatitis herpetiformis; primary VZV infection; hand, foot, and mouth disease; disseminated zoster infection; disseminated molluscum contagiosum; and eczema coxsackium.
Complications of EH include scarring; herpetic keratitis due to corneal infection, which if left untreated can progress to blindness; and rarely death due to multiorgan failure or septicemia.11 The traditional smallpox vaccine (ACAM2000) is contraindicated in patients with AD and EH, even when AD is in remission. These patients should avoid contact with recently vaccinated individuals.12 An alternative vaccine—Jynneos (Bavarian Nordic)—is available for these patients and their family members.13 Clinicians should be aware of this guideline, especially given the recent mpox (monkeypox) outbreaks.
Mild cases of EH are more common, may sometimes go unnoticed, and self-resolve in healthy patients. Severe cases may require systemic antiviral therapy. Acyclovir and its prodrug valacyclovir are standard treatments for EH. Alternatively, foscarnet or cidofovir can be used in the treatment of acyclovir-resistant thymidine kinase– deficient herpes simplex virus and other acyclovirresistant cases.14 Any secondary bacterial superinfections, usually due to staphylococcal or streptococcal bacteria, should be treated with antibiotics. A thorough ophthalmologic evaluation should be performed for patients with periocular involvement of EH. Empiric treatment should be started immediately, given a relative low toxicity of systemic antiviral therapy and high morbidity and mortality associated with untreated widespread EH.
It is important to maintain a high index of clinical suspicion for EH, especially in patients with pre-existing conditions such as AD who present with systemic symptoms and facial vesicles, pustules, or erosions to ensure prompt diagnosis and appropriate treatment.
The Diagnosis: Eczema Herpeticum
The patient’s condition with worsening facial edema and notable pain prompted a bedside Tzanck smear using a sample from the base of a deroofed forehead vesicle. In addition, a swab of a deroofed lesion was sent for herpes simplex virus and varicella-zoster virus (VZV) polymerase chain reaction (PCR) testing. The Tzanck smear demonstrated ballooning multinucleated syncytial giant cells and eosinophilic inclusion bodies (Figure), which are characteristic of certain herpesviruses including herpes simplex virus and VZV. He was started on intravenous acyclovir while PCR results were pending; the PCR test later confirmed positivity for herpes simplex virus type 1. Treatment was transitioned to oral valacyclovir once the lesions started crusting over. Notable healing and epithelialization of the lesions occurred during his hospital stay, and he was discharged home 5 days after starting treatment. He was counseled on autoinoculation, advised that he was considered infectious until all lesions had crusted over, and encouraged to employ frequent handwashing. Complete resolution of eczema herpeticum (EH) was noted at 3-week follow-up.

Eczema herpeticum (also known as Kaposi varicelliform eruption) is a potentially life-threatening disseminated cutaneous infection caused by herpes simplex virus types 1 and 2 in patients with pre-existing skin disease.1 It typically presents as a complication of atopic dermatitis (AD) but also has been identified as a rare complication in other conditions that disrupt the normal skin barrier, including mycosis fungoides, pemphigus foliaceus, pemphigus vulgaris, Darier disease, pityriasis rubra pilaris, contact dermatitis, and seborrheic dermatitis.1-4
The pathogenesis of EH is multifactorial. Disruption of the stratum corneum; impaired natural killer cell function; early-onset, untreated, or severe AD; disrupted skin microbiota with skewed colonization by Staphylococcus aureus; immunosuppressive AD therapies such as calcineurin inhibitors; eosinophilia; and helper T cell (TH2) cytokine predominance all have been suggested to play a role in the development of EH.5-8
As seen in our patient, EH presents with a sudden eruption of painful or pruritic, grouped, monomorphic, domeshaped vesicles with background swelling and erythema typically on the head, neck, and trunk. Vesicles then progress to punched-out erosions with overlying hemorrhagic crusting that can coalesce to form large denuded areas susceptible to superinfection with bacteria.9 Other accompanying symptoms include high fever, chills, malaise, and lymphadenopathy. Associated inflammation, classically described as erythema, may be difficult to discern in patients with darker skin and appears as hyperpigmentation; therefore, identification of clusters of monomorphic vesicles in areas of pre-existing dermatitis is particularly important for clinical diagnosis in people with darker skin types.
Various tests are available to confirm diagnosis in ambiguous cases. Bedside Tzanck smears can be performed rapidly and are considered positive if characteristic multinucleated giant cells are noted; however, they do not differentiate between the various herpesviruses. Direct fluorescent antibody testing of scraped lesions and viral cultures of swabbed vesicular fluid are equally effective in distinguishing between herpes simplex virus type 1, herpes simplex virus type 2, and VZV; PCR confirms the diagnosis with high specificity and sensitivity.10
In our patient, the initial differential diagnosis included EH, acute generalized exanthematous pustulosis, allergic contact dermatitis, and Orthopoxvirus infection. The positive Tzanck smear reduced the likelihood of a nonviral etiology. Additionally, worsening of the rash despite discontinuation of medications and utilization of topical steroids argued against acute generalized exanthematous pustulosis and allergic contact dermatitis. The laboratory findings reduced the likelihood of drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome, and PCR findings ultimately ruled out Orthopoxvirus infections. Additional differential diagnoses for EH include dermatitis herpetiformis; primary VZV infection; hand, foot, and mouth disease; disseminated zoster infection; disseminated molluscum contagiosum; and eczema coxsackium.
Complications of EH include scarring; herpetic keratitis due to corneal infection, which if left untreated can progress to blindness; and rarely death due to multiorgan failure or septicemia.11 The traditional smallpox vaccine (ACAM2000) is contraindicated in patients with AD and EH, even when AD is in remission. These patients should avoid contact with recently vaccinated individuals.12 An alternative vaccine—Jynneos (Bavarian Nordic)—is available for these patients and their family members.13 Clinicians should be aware of this guideline, especially given the recent mpox (monkeypox) outbreaks.
Mild cases of EH are more common, may sometimes go unnoticed, and self-resolve in healthy patients. Severe cases may require systemic antiviral therapy. Acyclovir and its prodrug valacyclovir are standard treatments for EH. Alternatively, foscarnet or cidofovir can be used in the treatment of acyclovir-resistant thymidine kinase– deficient herpes simplex virus and other acyclovirresistant cases.14 Any secondary bacterial superinfections, usually due to staphylococcal or streptococcal bacteria, should be treated with antibiotics. A thorough ophthalmologic evaluation should be performed for patients with periocular involvement of EH. Empiric treatment should be started immediately, given a relative low toxicity of systemic antiviral therapy and high morbidity and mortality associated with untreated widespread EH.
It is important to maintain a high index of clinical suspicion for EH, especially in patients with pre-existing conditions such as AD who present with systemic symptoms and facial vesicles, pustules, or erosions to ensure prompt diagnosis and appropriate treatment.
- Baaniya B, Agrawal S. Kaposi varicelliform eruption in a patient with pemphigus vulgaris: a case report and review of the literature. Case Rep Dermatol Med. 2020;2020:6695342. doi:10.1155/2020/6695342
- Tayabali K, Pothiwalla H, Lowitt M. Eczema herpeticum in Darier’s disease: a topical storm. J Community Hosp Intern Med Perspect. 2019;9:347. doi:10.1080/20009666.2019.1650590
- Cavalié M, Giacchero D, Cardot-Leccia N, et al. Kaposi’s varicelliform eruption in a patient with pityriasis rubra pilaris (pityriasis rubra pilaris herpeticum). J Eur Acad Dermatol Venereol. 2013;27:1585-1586. doi:10.1111/JDV.12120
- Lee GH, Kim YM, Lee SY, et al. A case of eczema herpeticum with Hailey-Hailey disease. Ann Dermatol. 2009;21:311-314. doi:10.5021/ad.2009.21.3.311
- Seegräber M, Worm M, Werfel T, et al. Recurrent eczema herpeticum— a retrospective European multicenter study evaluating the clinical characteristics of eczema herpeticum cases in atopic dermatitis patients. J Eur Acad Dermatol Venereol. 2020;34:1074-1079. doi:10.1111/JDV.16090
- Kawakami Y, Ando T, Lee J-R, et al. Defective natural killer cell activity in a mouse model of eczema herpeticum. J Allergy Clin Immunol. 2017;139:997-1006.e10. doi:10.1016/j.jaci.2016.06.034
- Beck L, Latchney L, Zaccaro D, et al. Biomarkers of disease severity and Th2 polarity are predictors of risk for eczema herpeticum. J Allergy Clin Immunol. 2008;121:S37-S37. doi:10.1016/j.jaci.2007.12.152
- Kim M, Jung M, Hong SP, et al. Topical calcineurin inhibitors compromise stratum corneum integrity, epidermal permeability and antimicrobial barrier function. Exp Dermatol. 2010; 19:501-510. doi:10.1111/J.1600-0625.2009.00941.X
- Karray M, Kwan E, Souissi A. Kaposi varicelliform eruption. StatPearls [Internet]. StatPearls Publishing; 2023. https://www.ncbi.nlm.nih.gov/books/NBK482432/
- Dominguez SR, Pretty K, Hengartner R, et al. Comparison of herpes simplex virus PCR with culture for virus detection in multisource surface swab specimens from neonates [published online September 25, 2018]. J Clin Microbiol. doi:10.1128/JCM.00632-18
- Feye F, De Halleux C, Gillet JB, et al. Exacerbation of atopic dermatitis in the emergency department. Eur J Emerg Med. 2004;11:49-52. doi:10.1097/00063110-200412000-00014
- Casey C, Vellozzi C, Mootrey GT, et al; Vaccinia Case Definition Development Working Group; Advisory Committee on Immunization Practices-Armed Forces Epidemiological Board Smallpox Vaccine Safety Working Group. Surveillance guidelines for smallpox vaccine (vaccinia) adverse reactions. MMWR Recomm Rep. 2006;55:1-16.
- Rao AK, Petersen BW, Whitehill F, et al. Use of JYNNEOS (Smallpox and Monkeypox Vaccine, Live, Nonreplicating) for preexposure vaccination of persons at risk for occupational exposure to orthopoxviruses: recommendations of the Advisory Committee on Immunization Practices—United States, 2022. MMWR Morb Mortal Wkly Rep. 2022;71:734-742. doi:10.15585 /MMWR.MM7122E1
- Piret J, Boivin G. Resistance of herpes simplex viruses to nucleoside analogues: mechanisms, prevalence, and management. Antimicrob Agents Chemother. 2011;55:459. doi:10.1128/AAC.00615-10
- Baaniya B, Agrawal S. Kaposi varicelliform eruption in a patient with pemphigus vulgaris: a case report and review of the literature. Case Rep Dermatol Med. 2020;2020:6695342. doi:10.1155/2020/6695342
- Tayabali K, Pothiwalla H, Lowitt M. Eczema herpeticum in Darier’s disease: a topical storm. J Community Hosp Intern Med Perspect. 2019;9:347. doi:10.1080/20009666.2019.1650590
- Cavalié M, Giacchero D, Cardot-Leccia N, et al. Kaposi’s varicelliform eruption in a patient with pityriasis rubra pilaris (pityriasis rubra pilaris herpeticum). J Eur Acad Dermatol Venereol. 2013;27:1585-1586. doi:10.1111/JDV.12120
- Lee GH, Kim YM, Lee SY, et al. A case of eczema herpeticum with Hailey-Hailey disease. Ann Dermatol. 2009;21:311-314. doi:10.5021/ad.2009.21.3.311
- Seegräber M, Worm M, Werfel T, et al. Recurrent eczema herpeticum— a retrospective European multicenter study evaluating the clinical characteristics of eczema herpeticum cases in atopic dermatitis patients. J Eur Acad Dermatol Venereol. 2020;34:1074-1079. doi:10.1111/JDV.16090
- Kawakami Y, Ando T, Lee J-R, et al. Defective natural killer cell activity in a mouse model of eczema herpeticum. J Allergy Clin Immunol. 2017;139:997-1006.e10. doi:10.1016/j.jaci.2016.06.034
- Beck L, Latchney L, Zaccaro D, et al. Biomarkers of disease severity and Th2 polarity are predictors of risk for eczema herpeticum. J Allergy Clin Immunol. 2008;121:S37-S37. doi:10.1016/j.jaci.2007.12.152
- Kim M, Jung M, Hong SP, et al. Topical calcineurin inhibitors compromise stratum corneum integrity, epidermal permeability and antimicrobial barrier function. Exp Dermatol. 2010; 19:501-510. doi:10.1111/J.1600-0625.2009.00941.X
- Karray M, Kwan E, Souissi A. Kaposi varicelliform eruption. StatPearls [Internet]. StatPearls Publishing; 2023. https://www.ncbi.nlm.nih.gov/books/NBK482432/
- Dominguez SR, Pretty K, Hengartner R, et al. Comparison of herpes simplex virus PCR with culture for virus detection in multisource surface swab specimens from neonates [published online September 25, 2018]. J Clin Microbiol. doi:10.1128/JCM.00632-18
- Feye F, De Halleux C, Gillet JB, et al. Exacerbation of atopic dermatitis in the emergency department. Eur J Emerg Med. 2004;11:49-52. doi:10.1097/00063110-200412000-00014
- Casey C, Vellozzi C, Mootrey GT, et al; Vaccinia Case Definition Development Working Group; Advisory Committee on Immunization Practices-Armed Forces Epidemiological Board Smallpox Vaccine Safety Working Group. Surveillance guidelines for smallpox vaccine (vaccinia) adverse reactions. MMWR Recomm Rep. 2006;55:1-16.
- Rao AK, Petersen BW, Whitehill F, et al. Use of JYNNEOS (Smallpox and Monkeypox Vaccine, Live, Nonreplicating) for preexposure vaccination of persons at risk for occupational exposure to orthopoxviruses: recommendations of the Advisory Committee on Immunization Practices—United States, 2022. MMWR Morb Mortal Wkly Rep. 2022;71:734-742. doi:10.15585 /MMWR.MM7122E1
- Piret J, Boivin G. Resistance of herpes simplex viruses to nucleoside analogues: mechanisms, prevalence, and management. Antimicrob Agents Chemother. 2011;55:459. doi:10.1128/AAC.00615-10
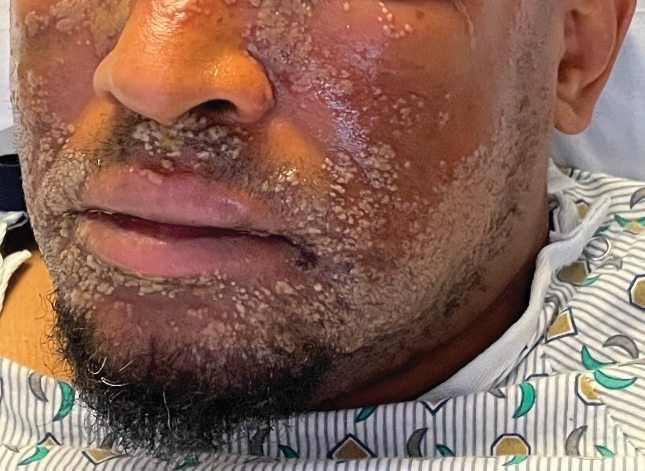
A 52-year-old man developed a sudden eruption of small pustules on background erythema and edema covering the forehead, nasal bridge, periorbital region, cheeks, and perioral region on day 3 of hospitalization in the intensive care unit for management of septic shock secondary to a complicated urinary tract infection. He had a medical history of benign prostatic hyperplasia, sarcoidosis, and atopic dermatitis. He initially presented to the emergency department with fever, chills, and dysuria of 2 days’ duration. Because he received ceftriaxone, vancomycin, ciprofloxacin, and tamsulosin while hospitalized for the infection, the primary medical team suspected a drug reaction and empirically started applying hydrocortisone cream 2.5%. The rash continued to spread over the ensuing day, prompting a dermatology consultation to rule out a drug eruption and to help guide further management. The patient was in substantial distress and pain. Physical examination revealed numerous discrete and confluent monomorphic pustules on background erythema with faint collarettes of scale covering most of the face. Substantial periorbital and facial edema forced the eyes closed. There was no mucous membrane involvement. A review of systems was negative for dyspnea and dysphagia, and the rash was not present elsewhere on the body. Ophthalmologic evaluation revealed no ocular involvement or vision changes. Laboratory studies demonstrated neutrophilia (17.27×109 cells/L [reference range, 2.0–6.9×109 cells/L]). The eosinophil count, blood urea nitrogen/creatinine, and liver function tests were within reference range.
Topical ivermectin study sheds light on dysbiosis in rosacea
, according to a report presented at the recent European Academy of Dermatology and Venereology (EADV) 2023 Congress.
“This is the first hint that the host’s cutaneous microbiome plays a secondary role in the immunopathogenesis of rosacea,” said Bernard Homey, MD, director of the department of dermatology at University Hospital Düsseldorf in Germany.
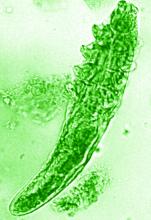
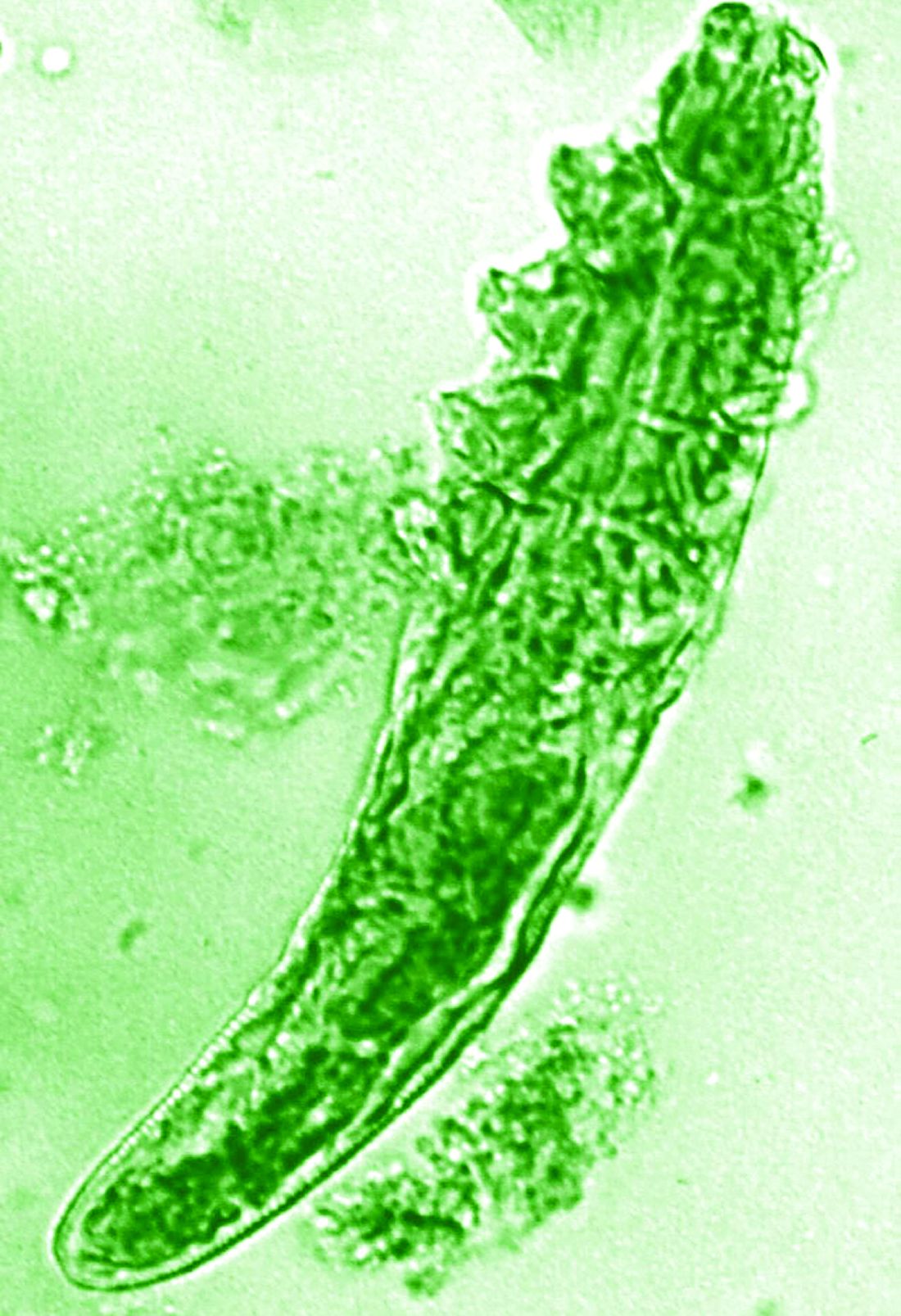
“In rosacea, we are well aware of trigger factors such as stress, UV light, heat, cold, food, and alcohol,” he said. “We are also well aware that there is an increase in Demodex mites in the pilosebaceous unit.”
Research over the past decade has also started to look at the potential role of the skin microbiome in the disease process, but answers have remained “largely elusive,” Dr. Homey said.
Ivermectin helps, but how?
Ivermectin 1% cream (Soolantra) has been approved by the U.S. Food and Drug Administration since 2014 for the treatment of the inflammatory lesions that are characteristic of rosacea, but its mechanism of action is not clear.
Dr. Homey presented the results of a study of 61 patients designed to look at how ivermectin might be working in the treatment of people with rosacea and investigate if there was any relation to the skin microbiome and transcriptome of patients.
The trial included 41 individuals with papulopustular rosacea and 20 individuals who did not have rosacea. For all patients, surface skin biopsies were performed twice 30 days apart using cyanoacrylate glue; patients with rosacea were treated with topical ivermectin 1% between biopsies. Skin samples obtained at day 0 and day 30 were examined under the microscope, and Demodex counts (mites/cm2) of skin and RNA sequencing of the cutaneous microbiome were undertaken.
The mean age of the patients with rosacea was 54.9 years, and the mean Demodex counts before and after treatment were a respective 7.2 cm2 and 0.9 cm2.
Using the Investigator’s General Assessment to assess the severity of rosacea, Homey reported that 43.9% of patients with rosacea had a decrease in scores at day 30, indicating improvement.
In addition, topical ivermectin resulted in a marked or total decrease in Demodex mite density for 87.5% of patients (n = 24) who were identified as having the mites.
Skin microbiome changes seen
As a form of quality control, skin microbiome changes among the patients were compared with control patients using 16S rRNA sequencing.
“The taxa we find within the cutaneous niche of inflammatory lesions of rosacea patients are significantly different from healthy volunteers,” Dr. Homey said.
Cutibacterium species are predominant in healthy control persons but are not present when there is inflammation in patients with rosacea. Instead, staphylococcus species “take over the niche, similar to atopic dermatitis,” he noted.
Looking at how treatment with ivermectin influences the organisms, the decrease in C. acnes seen in patients with rosacea persisted despite treatment, and the abundance of Staphylococcus epidermidis, S. hominis, and S. capitis increased further. This suggests a possible protective or homeostatic role of C. acnes but a pathogenic role for staphylococci, explained Dr. Homey.
“Surprisingly, although inflammatory lesions decrease, patients get better, the cutaneous microbiome does not revert to homeostatic conditions during topical ivermectin treatment,” he observed.
There is, of course, variability among individuals.
Dr. Homey also reported that Snodgrassella alvi – a microorganism believed to reside in the gut of Demodex folliculorum mites – was found in the skin microbiome of patients with rosacea before but not after ivermectin treatment. This may mean that this microorganism could be partially triggering inflammation in rosacea patients.
Looking at the transcriptome of patients, Dr. Homey said that there was downregulation of distinct genes that might make for more favorable conditions for Demodex mites.
Moreover, insufficient upregulation of interleukin-17 pathways might be working together with barrier defects in the skin and metabolic changes to “pave the way” for colonization by S. epidermidis.
Pulling it together
Dr. Homey and associates conclude in their abstract that the findings “support that rosacea lesions are associated with dysbiosis.”
Although treatment with ivermectin did not normalize the skin’s microbiome, it was associated with a decrease in Demodex mite density and the reduction of microbes associated with Demodex.
Margarida Gonçalo, MD, PhD, professor of dermatology at the University of Coimbra in Portugal, who cochaired the late-breaking news session where the data were presented, asked whether healthy and affected skin in patients with rosacea had been compared, rather than comparing the skin of rosacea lesions with healthy control samples.
“No, we did not this, as this is methodologically a little bit more difficult,” Dr. Homey responded.
Also cochairing the session was Michel Gilliet, MD, chair of the department of dermatology at the University Hospital CHUV in Lausanne, Switzerland. He commented that these “data suggest that there’s an intimate link between Demodex and the skin microbiota and dysbiosis in in rosacea.”
Dr. Gilliet added: “You have a whole dysbiosis going on in rosacea, which is probably only dependent on these bacteria.”
It would be “very interesting,” as a “proof-of-concept” study, to look at whether depleting Demodex would also delete S. alvi, he suggested.
The study was funded by Galderma. Dr. Homey has acted as a consultant, speaker or investigator for many pharmaceutical companies including Galderma.
A version of this article first appeared on Medscape.com.
, according to a report presented at the recent European Academy of Dermatology and Venereology (EADV) 2023 Congress.
“This is the first hint that the host’s cutaneous microbiome plays a secondary role in the immunopathogenesis of rosacea,” said Bernard Homey, MD, director of the department of dermatology at University Hospital Düsseldorf in Germany.
“In rosacea, we are well aware of trigger factors such as stress, UV light, heat, cold, food, and alcohol,” he said. “We are also well aware that there is an increase in Demodex mites in the pilosebaceous unit.”
Research over the past decade has also started to look at the potential role of the skin microbiome in the disease process, but answers have remained “largely elusive,” Dr. Homey said.
Ivermectin helps, but how?
Ivermectin 1% cream (Soolantra) has been approved by the U.S. Food and Drug Administration since 2014 for the treatment of the inflammatory lesions that are characteristic of rosacea, but its mechanism of action is not clear.
Dr. Homey presented the results of a study of 61 patients designed to look at how ivermectin might be working in the treatment of people with rosacea and investigate if there was any relation to the skin microbiome and transcriptome of patients.
The trial included 41 individuals with papulopustular rosacea and 20 individuals who did not have rosacea. For all patients, surface skin biopsies were performed twice 30 days apart using cyanoacrylate glue; patients with rosacea were treated with topical ivermectin 1% between biopsies. Skin samples obtained at day 0 and day 30 were examined under the microscope, and Demodex counts (mites/cm2) of skin and RNA sequencing of the cutaneous microbiome were undertaken.
The mean age of the patients with rosacea was 54.9 years, and the mean Demodex counts before and after treatment were a respective 7.2 cm2 and 0.9 cm2.
Using the Investigator’s General Assessment to assess the severity of rosacea, Homey reported that 43.9% of patients with rosacea had a decrease in scores at day 30, indicating improvement.
In addition, topical ivermectin resulted in a marked or total decrease in Demodex mite density for 87.5% of patients (n = 24) who were identified as having the mites.
Skin microbiome changes seen
As a form of quality control, skin microbiome changes among the patients were compared with control patients using 16S rRNA sequencing.
“The taxa we find within the cutaneous niche of inflammatory lesions of rosacea patients are significantly different from healthy volunteers,” Dr. Homey said.
Cutibacterium species are predominant in healthy control persons but are not present when there is inflammation in patients with rosacea. Instead, staphylococcus species “take over the niche, similar to atopic dermatitis,” he noted.
Looking at how treatment with ivermectin influences the organisms, the decrease in C. acnes seen in patients with rosacea persisted despite treatment, and the abundance of Staphylococcus epidermidis, S. hominis, and S. capitis increased further. This suggests a possible protective or homeostatic role of C. acnes but a pathogenic role for staphylococci, explained Dr. Homey.
“Surprisingly, although inflammatory lesions decrease, patients get better, the cutaneous microbiome does not revert to homeostatic conditions during topical ivermectin treatment,” he observed.
There is, of course, variability among individuals.
Dr. Homey also reported that Snodgrassella alvi – a microorganism believed to reside in the gut of Demodex folliculorum mites – was found in the skin microbiome of patients with rosacea before but not after ivermectin treatment. This may mean that this microorganism could be partially triggering inflammation in rosacea patients.
Looking at the transcriptome of patients, Dr. Homey said that there was downregulation of distinct genes that might make for more favorable conditions for Demodex mites.
Moreover, insufficient upregulation of interleukin-17 pathways might be working together with barrier defects in the skin and metabolic changes to “pave the way” for colonization by S. epidermidis.
Pulling it together
Dr. Homey and associates conclude in their abstract that the findings “support that rosacea lesions are associated with dysbiosis.”
Although treatment with ivermectin did not normalize the skin’s microbiome, it was associated with a decrease in Demodex mite density and the reduction of microbes associated with Demodex.
Margarida Gonçalo, MD, PhD, professor of dermatology at the University of Coimbra in Portugal, who cochaired the late-breaking news session where the data were presented, asked whether healthy and affected skin in patients with rosacea had been compared, rather than comparing the skin of rosacea lesions with healthy control samples.
“No, we did not this, as this is methodologically a little bit more difficult,” Dr. Homey responded.
Also cochairing the session was Michel Gilliet, MD, chair of the department of dermatology at the University Hospital CHUV in Lausanne, Switzerland. He commented that these “data suggest that there’s an intimate link between Demodex and the skin microbiota and dysbiosis in in rosacea.”
Dr. Gilliet added: “You have a whole dysbiosis going on in rosacea, which is probably only dependent on these bacteria.”
It would be “very interesting,” as a “proof-of-concept” study, to look at whether depleting Demodex would also delete S. alvi, he suggested.
The study was funded by Galderma. Dr. Homey has acted as a consultant, speaker or investigator for many pharmaceutical companies including Galderma.
A version of this article first appeared on Medscape.com.
, according to a report presented at the recent European Academy of Dermatology and Venereology (EADV) 2023 Congress.
“This is the first hint that the host’s cutaneous microbiome plays a secondary role in the immunopathogenesis of rosacea,” said Bernard Homey, MD, director of the department of dermatology at University Hospital Düsseldorf in Germany.
“In rosacea, we are well aware of trigger factors such as stress, UV light, heat, cold, food, and alcohol,” he said. “We are also well aware that there is an increase in Demodex mites in the pilosebaceous unit.”
Research over the past decade has also started to look at the potential role of the skin microbiome in the disease process, but answers have remained “largely elusive,” Dr. Homey said.
Ivermectin helps, but how?
Ivermectin 1% cream (Soolantra) has been approved by the U.S. Food and Drug Administration since 2014 for the treatment of the inflammatory lesions that are characteristic of rosacea, but its mechanism of action is not clear.
Dr. Homey presented the results of a study of 61 patients designed to look at how ivermectin might be working in the treatment of people with rosacea and investigate if there was any relation to the skin microbiome and transcriptome of patients.
The trial included 41 individuals with papulopustular rosacea and 20 individuals who did not have rosacea. For all patients, surface skin biopsies were performed twice 30 days apart using cyanoacrylate glue; patients with rosacea were treated with topical ivermectin 1% between biopsies. Skin samples obtained at day 0 and day 30 were examined under the microscope, and Demodex counts (mites/cm2) of skin and RNA sequencing of the cutaneous microbiome were undertaken.
The mean age of the patients with rosacea was 54.9 years, and the mean Demodex counts before and after treatment were a respective 7.2 cm2 and 0.9 cm2.
Using the Investigator’s General Assessment to assess the severity of rosacea, Homey reported that 43.9% of patients with rosacea had a decrease in scores at day 30, indicating improvement.
In addition, topical ivermectin resulted in a marked or total decrease in Demodex mite density for 87.5% of patients (n = 24) who were identified as having the mites.
Skin microbiome changes seen
As a form of quality control, skin microbiome changes among the patients were compared with control patients using 16S rRNA sequencing.
“The taxa we find within the cutaneous niche of inflammatory lesions of rosacea patients are significantly different from healthy volunteers,” Dr. Homey said.
Cutibacterium species are predominant in healthy control persons but are not present when there is inflammation in patients with rosacea. Instead, staphylococcus species “take over the niche, similar to atopic dermatitis,” he noted.
Looking at how treatment with ivermectin influences the organisms, the decrease in C. acnes seen in patients with rosacea persisted despite treatment, and the abundance of Staphylococcus epidermidis, S. hominis, and S. capitis increased further. This suggests a possible protective or homeostatic role of C. acnes but a pathogenic role for staphylococci, explained Dr. Homey.
“Surprisingly, although inflammatory lesions decrease, patients get better, the cutaneous microbiome does not revert to homeostatic conditions during topical ivermectin treatment,” he observed.
There is, of course, variability among individuals.
Dr. Homey also reported that Snodgrassella alvi – a microorganism believed to reside in the gut of Demodex folliculorum mites – was found in the skin microbiome of patients with rosacea before but not after ivermectin treatment. This may mean that this microorganism could be partially triggering inflammation in rosacea patients.
Looking at the transcriptome of patients, Dr. Homey said that there was downregulation of distinct genes that might make for more favorable conditions for Demodex mites.
Moreover, insufficient upregulation of interleukin-17 pathways might be working together with barrier defects in the skin and metabolic changes to “pave the way” for colonization by S. epidermidis.
Pulling it together
Dr. Homey and associates conclude in their abstract that the findings “support that rosacea lesions are associated with dysbiosis.”
Although treatment with ivermectin did not normalize the skin’s microbiome, it was associated with a decrease in Demodex mite density and the reduction of microbes associated with Demodex.
Margarida Gonçalo, MD, PhD, professor of dermatology at the University of Coimbra in Portugal, who cochaired the late-breaking news session where the data were presented, asked whether healthy and affected skin in patients with rosacea had been compared, rather than comparing the skin of rosacea lesions with healthy control samples.
“No, we did not this, as this is methodologically a little bit more difficult,” Dr. Homey responded.
Also cochairing the session was Michel Gilliet, MD, chair of the department of dermatology at the University Hospital CHUV in Lausanne, Switzerland. He commented that these “data suggest that there’s an intimate link between Demodex and the skin microbiota and dysbiosis in in rosacea.”
Dr. Gilliet added: “You have a whole dysbiosis going on in rosacea, which is probably only dependent on these bacteria.”
It would be “very interesting,” as a “proof-of-concept” study, to look at whether depleting Demodex would also delete S. alvi, he suggested.
The study was funded by Galderma. Dr. Homey has acted as a consultant, speaker or investigator for many pharmaceutical companies including Galderma.
A version of this article first appeared on Medscape.com.
FROM EADV 2023
A new long COVID explanation: Low serotonin levels?
Could antidepressants hold the key to treating long COVID? The study even points to a possible treatment.
Serotonin is a neurotransmitter that has many functions in the body and is targeted by the most commonly prescribed antidepressants – the selective serotonin reuptake inhibitors.
Serotonin is widely studied for its effects on the brain – it regulates the messaging between neurons, affecting sleep, mood, and memory. It is present in the gut, is found in cells along the gastrointestinal tract, and is absorbed by blood platelets. Gut serotonin, known as circulating serotonin, is responsible for a host of other functions, including the regulation of blood flow, body temperature, and digestion.
Low levels of serotonin could result in any number of seemingly unrelated symptoms, as in the case of long COVID, experts say. The condition affects about 7% of Americans and is associated with a wide range of health problems, including fatigue, shortness of breath, neurological symptoms, joint pain, blood clots, heart palpitations, and digestive problems.
Long COVID is difficult to treat because researchers haven’t been able to pinpoint the underlying mechanisms that cause prolonged illness after a SARS-CoV-2 infection, said study author Christoph A. Thaiss, PhD, an assistant professor of microbiology at the Perelman School of Medicine at the University of Pennsylvania.
The hope is that this study could have implications for new treatments, he said.
“Long COVID can have manifestations not only in the brain but in many different parts of the body, so it’s possible that serotonin reductions are involved in many different aspects of the disease,” said Dr. Thaiss.
Dr. Thaiss’s study, published in the journal Cell, found lower serotonin levels in long COVID patients, compared with patients who were diagnosed with acute COVID-19 but who fully recovered.
His team found that reductions in serotonin were driven by low levels of circulating SARS-CoV-2 virus that caused persistent inflammation as well as an inability of the body to absorb tryptophan, an amino acid that’s a precursor to serotonin. Overactive blood platelets were also shown to play a role; they serve as the primary means of serotonin absorption.
The study doesn’t make any recommendations for treatment, but understanding the role of serotonin in long COVID opens the door to a host of novel ideas that could set the stage for clinical trials and affect care.
“The study gives us a few possible targets that could be used in future clinical studies,” Dr. Thaiss said.
Persistent circulating virus is one of the drivers of low serotonin levels, said study author Michael Peluso, MD, an assistant research professor of infectious medicine at the University of California, San Francisco, School of Medicine. This points to the need to reduce viral load using antiviral medications like nirmatrelvir/ritonavir (Paxlovid), which is approved by the U.S. Food and Drug Administration for the treatment of COVID-19, and VV116, which has not yet been approved for use against COVID.
Research published in the New England Journal of Medicine found that the oral antiviral agent VV116 was as effective as nirmatrelvir/ritonavir in reducing the body’s viral load and aiding recovery from SARS-CoV-2 infection. Paxlovid has also been shown to reduce the likelihood of getting long COVID after an acute SARS-CoV-2 infection.
Researchers are investigating ways to target serotonin levels directly, potentially using SSRIs. But first they need to study whether improvement in serotonin level makes a difference.
“What we need now is a good clinical trial to see whether altering levels of serotonin in people with long COVID will lead to symptom relief,” Dr. Peluso said.
Indeed, the research did show that the SSRI fluoxetine, as well as a glycine-tryptophan supplement, improved cognitive function in SARS-CoV-2-infected rodent models, which were used in a portion of the study.
David F. Putrino, PhD, who runs the long COVID clinic at Mount Sinai Health System in New York City, said the research is helping “to paint a biological picture” that’s in line with other research on the mechanisms that cause long COVID symptoms.
But Dr. Putrino, who was not involved in the study, cautions against treating long COVID patients with SSRIs or any other treatment that increases serotonin before testing patients to determine whether their serotonin levels are actually lower than those of healthy persons.
“We don’t want to assume that every patient with long COVID is going to have lower serotonin levels,” said Dr. Putrino.
What’s more, researchers need to investigate whether SSRIs increase levels of circulating serotonin. It’s important to note that researchers found lower levels of circulating serotonin but that serotonin levels in the brain remained normal.
Traditionally, SSRIs are used clinically for increasing the levels of serotonin in the brain, not the body.
“Whether that’s going to contribute to an increase in systemic levels of serotonin, that’s something that needs to be tested,” said Akiko Iwasaki, PhD, co-lead investigator of the Yale School of Medicine, New Haven, Conn., COVID-19 Recovery Study, who was not involved in the research.
Thus far, investigators have not identified one unifying biomarker that seems to cause long COVID in all patients, said Dr. Iwasaki. Some research has found higher levels of certain immune cells and biomarkers: for example, monocytes and activated B lymphocytes, indicating a stronger and ongoing antibody response to the virus. Other recent research conducted by Dr. Iwasaki, Dr. Putrino, and others, published in the journal Nature, showed that long COVID patients tend to have lower levels of cortisol, which could be a factor in the extreme fatigue experienced by many who suffer from the condition.
The findings in the study in The Cell are promising, but they need to be replicated in more people, said Dr. Iwasaki. And even if they’re replicated in a larger study population, this would still be just one biomarker that is associated with one subtype of the disease. There is a need to better understand which biomarkers go with which symptoms so that the most effective treatments can be identified, she said.
Both Dr. Putrino and Dr. Iwasaki contended that there isn’t a single factor that can explain all of long COVID. It’s a complex disease caused by a host of different mechanisms.
Still, low levels of serotonin could be an important piece of the puzzle. The next step, said Dr. Iwasaki, is to uncover how many of the millions of Americans with long COVID have this biomarker.
“People working in the field of long COVID should now be considering this pathway and thinking of ways to measure serotonin in their patients.”
A version of this article first appeared on Medscape.com.
Could antidepressants hold the key to treating long COVID? The study even points to a possible treatment.
Serotonin is a neurotransmitter that has many functions in the body and is targeted by the most commonly prescribed antidepressants – the selective serotonin reuptake inhibitors.
Serotonin is widely studied for its effects on the brain – it regulates the messaging between neurons, affecting sleep, mood, and memory. It is present in the gut, is found in cells along the gastrointestinal tract, and is absorbed by blood platelets. Gut serotonin, known as circulating serotonin, is responsible for a host of other functions, including the regulation of blood flow, body temperature, and digestion.
Low levels of serotonin could result in any number of seemingly unrelated symptoms, as in the case of long COVID, experts say. The condition affects about 7% of Americans and is associated with a wide range of health problems, including fatigue, shortness of breath, neurological symptoms, joint pain, blood clots, heart palpitations, and digestive problems.
Long COVID is difficult to treat because researchers haven’t been able to pinpoint the underlying mechanisms that cause prolonged illness after a SARS-CoV-2 infection, said study author Christoph A. Thaiss, PhD, an assistant professor of microbiology at the Perelman School of Medicine at the University of Pennsylvania.
The hope is that this study could have implications for new treatments, he said.
“Long COVID can have manifestations not only in the brain but in many different parts of the body, so it’s possible that serotonin reductions are involved in many different aspects of the disease,” said Dr. Thaiss.
Dr. Thaiss’s study, published in the journal Cell, found lower serotonin levels in long COVID patients, compared with patients who were diagnosed with acute COVID-19 but who fully recovered.
His team found that reductions in serotonin were driven by low levels of circulating SARS-CoV-2 virus that caused persistent inflammation as well as an inability of the body to absorb tryptophan, an amino acid that’s a precursor to serotonin. Overactive blood platelets were also shown to play a role; they serve as the primary means of serotonin absorption.
The study doesn’t make any recommendations for treatment, but understanding the role of serotonin in long COVID opens the door to a host of novel ideas that could set the stage for clinical trials and affect care.
“The study gives us a few possible targets that could be used in future clinical studies,” Dr. Thaiss said.
Persistent circulating virus is one of the drivers of low serotonin levels, said study author Michael Peluso, MD, an assistant research professor of infectious medicine at the University of California, San Francisco, School of Medicine. This points to the need to reduce viral load using antiviral medications like nirmatrelvir/ritonavir (Paxlovid), which is approved by the U.S. Food and Drug Administration for the treatment of COVID-19, and VV116, which has not yet been approved for use against COVID.
Research published in the New England Journal of Medicine found that the oral antiviral agent VV116 was as effective as nirmatrelvir/ritonavir in reducing the body’s viral load and aiding recovery from SARS-CoV-2 infection. Paxlovid has also been shown to reduce the likelihood of getting long COVID after an acute SARS-CoV-2 infection.
Researchers are investigating ways to target serotonin levels directly, potentially using SSRIs. But first they need to study whether improvement in serotonin level makes a difference.
“What we need now is a good clinical trial to see whether altering levels of serotonin in people with long COVID will lead to symptom relief,” Dr. Peluso said.
Indeed, the research did show that the SSRI fluoxetine, as well as a glycine-tryptophan supplement, improved cognitive function in SARS-CoV-2-infected rodent models, which were used in a portion of the study.
David F. Putrino, PhD, who runs the long COVID clinic at Mount Sinai Health System in New York City, said the research is helping “to paint a biological picture” that’s in line with other research on the mechanisms that cause long COVID symptoms.
But Dr. Putrino, who was not involved in the study, cautions against treating long COVID patients with SSRIs or any other treatment that increases serotonin before testing patients to determine whether their serotonin levels are actually lower than those of healthy persons.
“We don’t want to assume that every patient with long COVID is going to have lower serotonin levels,” said Dr. Putrino.
What’s more, researchers need to investigate whether SSRIs increase levels of circulating serotonin. It’s important to note that researchers found lower levels of circulating serotonin but that serotonin levels in the brain remained normal.
Traditionally, SSRIs are used clinically for increasing the levels of serotonin in the brain, not the body.
“Whether that’s going to contribute to an increase in systemic levels of serotonin, that’s something that needs to be tested,” said Akiko Iwasaki, PhD, co-lead investigator of the Yale School of Medicine, New Haven, Conn., COVID-19 Recovery Study, who was not involved in the research.
Thus far, investigators have not identified one unifying biomarker that seems to cause long COVID in all patients, said Dr. Iwasaki. Some research has found higher levels of certain immune cells and biomarkers: for example, monocytes and activated B lymphocytes, indicating a stronger and ongoing antibody response to the virus. Other recent research conducted by Dr. Iwasaki, Dr. Putrino, and others, published in the journal Nature, showed that long COVID patients tend to have lower levels of cortisol, which could be a factor in the extreme fatigue experienced by many who suffer from the condition.
The findings in the study in The Cell are promising, but they need to be replicated in more people, said Dr. Iwasaki. And even if they’re replicated in a larger study population, this would still be just one biomarker that is associated with one subtype of the disease. There is a need to better understand which biomarkers go with which symptoms so that the most effective treatments can be identified, she said.
Both Dr. Putrino and Dr. Iwasaki contended that there isn’t a single factor that can explain all of long COVID. It’s a complex disease caused by a host of different mechanisms.
Still, low levels of serotonin could be an important piece of the puzzle. The next step, said Dr. Iwasaki, is to uncover how many of the millions of Americans with long COVID have this biomarker.
“People working in the field of long COVID should now be considering this pathway and thinking of ways to measure serotonin in their patients.”
A version of this article first appeared on Medscape.com.
Could antidepressants hold the key to treating long COVID? The study even points to a possible treatment.
Serotonin is a neurotransmitter that has many functions in the body and is targeted by the most commonly prescribed antidepressants – the selective serotonin reuptake inhibitors.
Serotonin is widely studied for its effects on the brain – it regulates the messaging between neurons, affecting sleep, mood, and memory. It is present in the gut, is found in cells along the gastrointestinal tract, and is absorbed by blood platelets. Gut serotonin, known as circulating serotonin, is responsible for a host of other functions, including the regulation of blood flow, body temperature, and digestion.
Low levels of serotonin could result in any number of seemingly unrelated symptoms, as in the case of long COVID, experts say. The condition affects about 7% of Americans and is associated with a wide range of health problems, including fatigue, shortness of breath, neurological symptoms, joint pain, blood clots, heart palpitations, and digestive problems.
Long COVID is difficult to treat because researchers haven’t been able to pinpoint the underlying mechanisms that cause prolonged illness after a SARS-CoV-2 infection, said study author Christoph A. Thaiss, PhD, an assistant professor of microbiology at the Perelman School of Medicine at the University of Pennsylvania.
The hope is that this study could have implications for new treatments, he said.
“Long COVID can have manifestations not only in the brain but in many different parts of the body, so it’s possible that serotonin reductions are involved in many different aspects of the disease,” said Dr. Thaiss.
Dr. Thaiss’s study, published in the journal Cell, found lower serotonin levels in long COVID patients, compared with patients who were diagnosed with acute COVID-19 but who fully recovered.
His team found that reductions in serotonin were driven by low levels of circulating SARS-CoV-2 virus that caused persistent inflammation as well as an inability of the body to absorb tryptophan, an amino acid that’s a precursor to serotonin. Overactive blood platelets were also shown to play a role; they serve as the primary means of serotonin absorption.
The study doesn’t make any recommendations for treatment, but understanding the role of serotonin in long COVID opens the door to a host of novel ideas that could set the stage for clinical trials and affect care.
“The study gives us a few possible targets that could be used in future clinical studies,” Dr. Thaiss said.
Persistent circulating virus is one of the drivers of low serotonin levels, said study author Michael Peluso, MD, an assistant research professor of infectious medicine at the University of California, San Francisco, School of Medicine. This points to the need to reduce viral load using antiviral medications like nirmatrelvir/ritonavir (Paxlovid), which is approved by the U.S. Food and Drug Administration for the treatment of COVID-19, and VV116, which has not yet been approved for use against COVID.
Research published in the New England Journal of Medicine found that the oral antiviral agent VV116 was as effective as nirmatrelvir/ritonavir in reducing the body’s viral load and aiding recovery from SARS-CoV-2 infection. Paxlovid has also been shown to reduce the likelihood of getting long COVID after an acute SARS-CoV-2 infection.
Researchers are investigating ways to target serotonin levels directly, potentially using SSRIs. But first they need to study whether improvement in serotonin level makes a difference.
“What we need now is a good clinical trial to see whether altering levels of serotonin in people with long COVID will lead to symptom relief,” Dr. Peluso said.
Indeed, the research did show that the SSRI fluoxetine, as well as a glycine-tryptophan supplement, improved cognitive function in SARS-CoV-2-infected rodent models, which were used in a portion of the study.
David F. Putrino, PhD, who runs the long COVID clinic at Mount Sinai Health System in New York City, said the research is helping “to paint a biological picture” that’s in line with other research on the mechanisms that cause long COVID symptoms.
But Dr. Putrino, who was not involved in the study, cautions against treating long COVID patients with SSRIs or any other treatment that increases serotonin before testing patients to determine whether their serotonin levels are actually lower than those of healthy persons.
“We don’t want to assume that every patient with long COVID is going to have lower serotonin levels,” said Dr. Putrino.
What’s more, researchers need to investigate whether SSRIs increase levels of circulating serotonin. It’s important to note that researchers found lower levels of circulating serotonin but that serotonin levels in the brain remained normal.
Traditionally, SSRIs are used clinically for increasing the levels of serotonin in the brain, not the body.
“Whether that’s going to contribute to an increase in systemic levels of serotonin, that’s something that needs to be tested,” said Akiko Iwasaki, PhD, co-lead investigator of the Yale School of Medicine, New Haven, Conn., COVID-19 Recovery Study, who was not involved in the research.
Thus far, investigators have not identified one unifying biomarker that seems to cause long COVID in all patients, said Dr. Iwasaki. Some research has found higher levels of certain immune cells and biomarkers: for example, monocytes and activated B lymphocytes, indicating a stronger and ongoing antibody response to the virus. Other recent research conducted by Dr. Iwasaki, Dr. Putrino, and others, published in the journal Nature, showed that long COVID patients tend to have lower levels of cortisol, which could be a factor in the extreme fatigue experienced by many who suffer from the condition.
The findings in the study in The Cell are promising, but they need to be replicated in more people, said Dr. Iwasaki. And even if they’re replicated in a larger study population, this would still be just one biomarker that is associated with one subtype of the disease. There is a need to better understand which biomarkers go with which symptoms so that the most effective treatments can be identified, she said.
Both Dr. Putrino and Dr. Iwasaki contended that there isn’t a single factor that can explain all of long COVID. It’s a complex disease caused by a host of different mechanisms.
Still, low levels of serotonin could be an important piece of the puzzle. The next step, said Dr. Iwasaki, is to uncover how many of the millions of Americans with long COVID have this biomarker.
“People working in the field of long COVID should now be considering this pathway and thinking of ways to measure serotonin in their patients.”
A version of this article first appeared on Medscape.com.
FROM CELL
How to prescribe exercise in 5 steps
Clinicians are well aware of the benefits of physical activity and the consequences of inactivity.
Managing the diseases associated with inactivity – heart disease, type 2 diabetes, hypertension – falls to physicians. So one might assume they routinely prescribe exercise to their patients, just as they would statins, insulin, or beta-blockers.
But evidence indicates that doctors don’t routinely have those conversations. They may lack confidence in their ability to give effective advice, fear offending patients, or simply not know what to say.
That’s understandable. Many doctors receive little training on how to counsel patients to exercise, according to research over the past decade. Despite efforts to improve this, many medical students still feel unprepared to prescribe physical activity to patients.
But here’s the thing: Doctors are in a unique position to change things.
Only 28% of Americans meet physical activity guidelines, according to the U.S. Centers for Disease Control and Prevention. At the same time, other research suggests that patients want to be more active and would like help from their doctor.
“Patients are motivated to hear about physical activity from physicians and try to make a change,” says Jane Thornton, MD, PhD, an assistant professor in family medicine at Western University, Ont. “Just saying something, even if you don’t have specialized knowledge, makes a difference because of the credibility we have as physicians.”
Conveniently, just like exercise, the best way to get started is to ... get started.
Here’s how to break down the process into steps.
1. Ask patients about their physical activity
Think of this as taking any kind of patient history, only for physical activity.
Do they have a regular exercise routine? For how many minutes a day are they active? How many days a week?
“It takes less than a minute to ask and record,” Dr. Thornton says. Once you put it into the patient’s electronic record, you have something you can track.
2. Write an actual prescription
By giving the patient a written, printed prescription when they leave your office, “you’re showing it’s an important part of treatment or prevention,” Dr. Thornton explains. It puts physical activity on the level of a vital sign.
Include frequency, intensity, time, and type of exercise. The American College of Sports Medicine’s Exercise is Medicine initiative provides a prescription template you can use.
3. Measure what they do
Measurement helps the patient adopt the new behavior, and it helps the physician provide tailored advice going forward, Dr. Thornton says.
With the rise of health-monitoring wearables, tracking activity has never been easier. Of course, not everyone wants to (or can afford to) use a smartwatch or fitness tracker.
For tech-averse patients, ask if they’re willing to write something down, like how many minutes they spent walking, or how many yoga classes they attended. You may never get this from some patients, but it never hurts to ask.
4. Refer out when necessary
This brings us to a sticky issue for many physicians: lack of confidence in their ability to speak authoritatively about physical activity. “In most cases, you can absolutely say, ‘Start slow, go gradually,’ that kind of thing,” Dr. Thornton says. “As with anything, confidence will come with practice.”
For specific prescriptive advice, check out the Exercise is Medicine website, which also has handouts you can share with patients and information for specific conditions. If your patient has prediabetes, you can also point them toward the CDC’s diabetes prevention program, which is available in-person or online and may be free or covered by insurance.
If a patient has contraindications, refer out. If you don’t have exercise or rehab professionals in your network, Dr. Thornton recommends reaching out to your regional or national association of sports-medicine professionals. You should be able to find it with a quick Google search.
5. Follow up
Ask about physical activity during every contact, either in person or online.
Dr. Thornton says the second and fifth steps matter most to patients, especially when the prescription and follow-up come from their primary care physician, rather than a nurse or physician assistant to whom you’ve delegated the task.
“The value comes in having a physician emphasize the importance,” Dr. Thornton says. The more time you spend on it, the more that value comes through.
What NOT to say to patients about exercise
This might surprise you:
“I definitely don’t think telling people the official recommendations for physical activity is useful,” says Yoni Freedhoff, MD, an associate professor of family medicine at the University of Ottawa and medical director of the Bariatric Medical Institute. “If anything, I’d venture it’s counterproductive.”
It’s not that there’s anything wrong with the recommended minimum – 150 minutes of moderate-to-vigorous-intensity physical activity per week. The problem is what it says to a patient who doesn’t come close to those standards.
“Few real-world people have the interest, time, energy, or privilege to achieve them,” Dr. Freedhoff says. “Many will recognize that instantly and consequently feel [that] less than that is pointless.”
And that, Dr. Thornton says, is categorically not true. “Even minimal physical activity, in some cases, is beneficial.”
You also want to avoid any explicit connection between exercise and weight loss, Dr. Thornton says.
Though many people do connect the two, the link is often negative, notes a 2019 study from the University of Toronto., triggering painful memories that might go all the way back to gym class.
Try this pivot from Dr. Freedhoff: “Focus on the role of exercise in mitigating the risks of weight,” he says – like decreasing pain, increasing energy, and improving sleep.
How to motivate patients to move
New research backs up this more positive approach. In a study published in Annals of Internal Medicine, doctors in the United Kingdom who emphasized benefits and minimized health harms convinced more patients to join a weight management program than negative or neutral docs did. These doctors conveyed optimism and excitement, smiling and avoiding any mention of obesity or body mass index.
Exactly what benefits inspire change will be different for each patient. But in general, the more immediate the benefit, the more motivating it will be.
As the University of Toronto study noted, patients weren’t motivated by vague, distant goals like “increasing life expectancy or avoiding health problems many years in the future.”
They’re much more likely to take action to avoid surgery, reduce medications, or minimize the risk of falling.
For an older patient, Dr. Freedhoff says, “focusing on the preservation of functional independence can be extremely motivating.” That’s especially true if the patient has vivid memories of seeing a sedentary loved one decline late in life.
For patients who may be more focused on appearance, they could respond to the idea of improving their body composition. For that, “we talk about the quality of weight loss,” says Spencer Nadolsky, DO, an obesity and lipid specialist and medical director of WeightWatchers. “Ultimately, exercise helps shape the body instead of just changing the number on the scale.”
Reducing resistance to resistance training
A conversation about reshaping the body or avoiding age-related disabilities leads naturally to resistance training.
“I always frame resistance training as the single most valuable thing a person might do to try to preserve their functional independence,” Dr. Freedhoff says. If the patient is over 65, he won’t wait for them to show an interest. “I’ll absolutely bring it up with them directly.”
Dr. Freedhoff has an on-site training facility where trainers show patients how to work out at home with minimal equipment, like dumbbells and resistance bands.
Most doctors, however, don’t have those options. That can lead to a tricky conversation. Participants in the University of Toronto study told the authors they disliked the gym, finding it “boring, intimidating, or discouraging.”
And yet, “a common suggestion ... from health care providers was to join a gym.”
Many patients, Spencer Nadolsky, MD, says, associate strength training with “grunting, groaning, or getting ‘bulky’ vs. ‘toned.’ ” Memories of soreness from overzealous workouts are another barrier.
He recommends “starting small and slow,” with one or two full-body workouts a week. Those initial workouts might include just one to two sets of four to five exercises. “Consider if someone is exercising at home or in a gym to build a routine around equipment that’s available to them,” Dr. Nadolsky says.
Once you determine what you have to work with, help the patient choose exercises that fit their needs, goals, preferences, limitations, and prior injuries.
One more consideration: While Dr. Nadolsky tries to “stay away from telling a patient they need to do specific types of exercise to be successful,” he makes an exception for patients who’re taking a GLP-1 agonist. “There is a concern for muscle mass loss along with fat loss.”
Practicing, preaching, and checking privilege
When Dr. Thornton, Dr. Freedhoff, and Dr. Nadolsky discuss exercise, their patients know they practice what they preach.
Dr. Nadolsky, who was a nationally ranked wrestler at the University of North Carolina, hosts the Docs Who Lift podcast with his brother, Karl Nadolsky, MD.
Dr. Freedhoff is also a lifter and fitness enthusiast, and Dr. Thornton was a world-class rower whose team came within 0.8 seconds of a silver medal at the Beijing Olympics. (They finished fourth.)
But not all physicians follow their own lifestyle advice, Dr. Freedhoff says. That doesn’t make them bad doctors – it makes them human.
“I’ve done 300 minutes a week of exercise” – the recommended amount for weight maintenance – “to see what’s involved,” Dr. Freedhoff says. “That’s far, far, far from a trivial amount.”
That leads to this advice for his fellow physicians:
“The most important thing to know about exercise is that finding the time and having the health to do so is a privilege,” he says.
Understanding that is crucial for assessing your patient’s needs and providing the right help.
A version of this article first appeared on Medscape.com.
Clinicians are well aware of the benefits of physical activity and the consequences of inactivity.
Managing the diseases associated with inactivity – heart disease, type 2 diabetes, hypertension – falls to physicians. So one might assume they routinely prescribe exercise to their patients, just as they would statins, insulin, or beta-blockers.
But evidence indicates that doctors don’t routinely have those conversations. They may lack confidence in their ability to give effective advice, fear offending patients, or simply not know what to say.
That’s understandable. Many doctors receive little training on how to counsel patients to exercise, according to research over the past decade. Despite efforts to improve this, many medical students still feel unprepared to prescribe physical activity to patients.
But here’s the thing: Doctors are in a unique position to change things.
Only 28% of Americans meet physical activity guidelines, according to the U.S. Centers for Disease Control and Prevention. At the same time, other research suggests that patients want to be more active and would like help from their doctor.
“Patients are motivated to hear about physical activity from physicians and try to make a change,” says Jane Thornton, MD, PhD, an assistant professor in family medicine at Western University, Ont. “Just saying something, even if you don’t have specialized knowledge, makes a difference because of the credibility we have as physicians.”
Conveniently, just like exercise, the best way to get started is to ... get started.
Here’s how to break down the process into steps.
1. Ask patients about their physical activity
Think of this as taking any kind of patient history, only for physical activity.
Do they have a regular exercise routine? For how many minutes a day are they active? How many days a week?
“It takes less than a minute to ask and record,” Dr. Thornton says. Once you put it into the patient’s electronic record, you have something you can track.
2. Write an actual prescription
By giving the patient a written, printed prescription when they leave your office, “you’re showing it’s an important part of treatment or prevention,” Dr. Thornton explains. It puts physical activity on the level of a vital sign.
Include frequency, intensity, time, and type of exercise. The American College of Sports Medicine’s Exercise is Medicine initiative provides a prescription template you can use.
3. Measure what they do
Measurement helps the patient adopt the new behavior, and it helps the physician provide tailored advice going forward, Dr. Thornton says.
With the rise of health-monitoring wearables, tracking activity has never been easier. Of course, not everyone wants to (or can afford to) use a smartwatch or fitness tracker.
For tech-averse patients, ask if they’re willing to write something down, like how many minutes they spent walking, or how many yoga classes they attended. You may never get this from some patients, but it never hurts to ask.
4. Refer out when necessary
This brings us to a sticky issue for many physicians: lack of confidence in their ability to speak authoritatively about physical activity. “In most cases, you can absolutely say, ‘Start slow, go gradually,’ that kind of thing,” Dr. Thornton says. “As with anything, confidence will come with practice.”
For specific prescriptive advice, check out the Exercise is Medicine website, which also has handouts you can share with patients and information for specific conditions. If your patient has prediabetes, you can also point them toward the CDC’s diabetes prevention program, which is available in-person or online and may be free or covered by insurance.
If a patient has contraindications, refer out. If you don’t have exercise or rehab professionals in your network, Dr. Thornton recommends reaching out to your regional or national association of sports-medicine professionals. You should be able to find it with a quick Google search.
5. Follow up
Ask about physical activity during every contact, either in person or online.
Dr. Thornton says the second and fifth steps matter most to patients, especially when the prescription and follow-up come from their primary care physician, rather than a nurse or physician assistant to whom you’ve delegated the task.
“The value comes in having a physician emphasize the importance,” Dr. Thornton says. The more time you spend on it, the more that value comes through.
What NOT to say to patients about exercise
This might surprise you:
“I definitely don’t think telling people the official recommendations for physical activity is useful,” says Yoni Freedhoff, MD, an associate professor of family medicine at the University of Ottawa and medical director of the Bariatric Medical Institute. “If anything, I’d venture it’s counterproductive.”
It’s not that there’s anything wrong with the recommended minimum – 150 minutes of moderate-to-vigorous-intensity physical activity per week. The problem is what it says to a patient who doesn’t come close to those standards.
“Few real-world people have the interest, time, energy, or privilege to achieve them,” Dr. Freedhoff says. “Many will recognize that instantly and consequently feel [that] less than that is pointless.”
And that, Dr. Thornton says, is categorically not true. “Even minimal physical activity, in some cases, is beneficial.”
You also want to avoid any explicit connection between exercise and weight loss, Dr. Thornton says.
Though many people do connect the two, the link is often negative, notes a 2019 study from the University of Toronto., triggering painful memories that might go all the way back to gym class.
Try this pivot from Dr. Freedhoff: “Focus on the role of exercise in mitigating the risks of weight,” he says – like decreasing pain, increasing energy, and improving sleep.
How to motivate patients to move
New research backs up this more positive approach. In a study published in Annals of Internal Medicine, doctors in the United Kingdom who emphasized benefits and minimized health harms convinced more patients to join a weight management program than negative or neutral docs did. These doctors conveyed optimism and excitement, smiling and avoiding any mention of obesity or body mass index.
Exactly what benefits inspire change will be different for each patient. But in general, the more immediate the benefit, the more motivating it will be.
As the University of Toronto study noted, patients weren’t motivated by vague, distant goals like “increasing life expectancy or avoiding health problems many years in the future.”
They’re much more likely to take action to avoid surgery, reduce medications, or minimize the risk of falling.
For an older patient, Dr. Freedhoff says, “focusing on the preservation of functional independence can be extremely motivating.” That’s especially true if the patient has vivid memories of seeing a sedentary loved one decline late in life.
For patients who may be more focused on appearance, they could respond to the idea of improving their body composition. For that, “we talk about the quality of weight loss,” says Spencer Nadolsky, DO, an obesity and lipid specialist and medical director of WeightWatchers. “Ultimately, exercise helps shape the body instead of just changing the number on the scale.”
Reducing resistance to resistance training
A conversation about reshaping the body or avoiding age-related disabilities leads naturally to resistance training.
“I always frame resistance training as the single most valuable thing a person might do to try to preserve their functional independence,” Dr. Freedhoff says. If the patient is over 65, he won’t wait for them to show an interest. “I’ll absolutely bring it up with them directly.”
Dr. Freedhoff has an on-site training facility where trainers show patients how to work out at home with minimal equipment, like dumbbells and resistance bands.
Most doctors, however, don’t have those options. That can lead to a tricky conversation. Participants in the University of Toronto study told the authors they disliked the gym, finding it “boring, intimidating, or discouraging.”
And yet, “a common suggestion ... from health care providers was to join a gym.”
Many patients, Spencer Nadolsky, MD, says, associate strength training with “grunting, groaning, or getting ‘bulky’ vs. ‘toned.’ ” Memories of soreness from overzealous workouts are another barrier.
He recommends “starting small and slow,” with one or two full-body workouts a week. Those initial workouts might include just one to two sets of four to five exercises. “Consider if someone is exercising at home or in a gym to build a routine around equipment that’s available to them,” Dr. Nadolsky says.
Once you determine what you have to work with, help the patient choose exercises that fit their needs, goals, preferences, limitations, and prior injuries.
One more consideration: While Dr. Nadolsky tries to “stay away from telling a patient they need to do specific types of exercise to be successful,” he makes an exception for patients who’re taking a GLP-1 agonist. “There is a concern for muscle mass loss along with fat loss.”
Practicing, preaching, and checking privilege
When Dr. Thornton, Dr. Freedhoff, and Dr. Nadolsky discuss exercise, their patients know they practice what they preach.
Dr. Nadolsky, who was a nationally ranked wrestler at the University of North Carolina, hosts the Docs Who Lift podcast with his brother, Karl Nadolsky, MD.
Dr. Freedhoff is also a lifter and fitness enthusiast, and Dr. Thornton was a world-class rower whose team came within 0.8 seconds of a silver medal at the Beijing Olympics. (They finished fourth.)
But not all physicians follow their own lifestyle advice, Dr. Freedhoff says. That doesn’t make them bad doctors – it makes them human.
“I’ve done 300 minutes a week of exercise” – the recommended amount for weight maintenance – “to see what’s involved,” Dr. Freedhoff says. “That’s far, far, far from a trivial amount.”
That leads to this advice for his fellow physicians:
“The most important thing to know about exercise is that finding the time and having the health to do so is a privilege,” he says.
Understanding that is crucial for assessing your patient’s needs and providing the right help.
A version of this article first appeared on Medscape.com.
Clinicians are well aware of the benefits of physical activity and the consequences of inactivity.
Managing the diseases associated with inactivity – heart disease, type 2 diabetes, hypertension – falls to physicians. So one might assume they routinely prescribe exercise to their patients, just as they would statins, insulin, or beta-blockers.
But evidence indicates that doctors don’t routinely have those conversations. They may lack confidence in their ability to give effective advice, fear offending patients, or simply not know what to say.
That’s understandable. Many doctors receive little training on how to counsel patients to exercise, according to research over the past decade. Despite efforts to improve this, many medical students still feel unprepared to prescribe physical activity to patients.
But here’s the thing: Doctors are in a unique position to change things.
Only 28% of Americans meet physical activity guidelines, according to the U.S. Centers for Disease Control and Prevention. At the same time, other research suggests that patients want to be more active and would like help from their doctor.
“Patients are motivated to hear about physical activity from physicians and try to make a change,” says Jane Thornton, MD, PhD, an assistant professor in family medicine at Western University, Ont. “Just saying something, even if you don’t have specialized knowledge, makes a difference because of the credibility we have as physicians.”
Conveniently, just like exercise, the best way to get started is to ... get started.
Here’s how to break down the process into steps.
1. Ask patients about their physical activity
Think of this as taking any kind of patient history, only for physical activity.
Do they have a regular exercise routine? For how many minutes a day are they active? How many days a week?
“It takes less than a minute to ask and record,” Dr. Thornton says. Once you put it into the patient’s electronic record, you have something you can track.
2. Write an actual prescription
By giving the patient a written, printed prescription when they leave your office, “you’re showing it’s an important part of treatment or prevention,” Dr. Thornton explains. It puts physical activity on the level of a vital sign.
Include frequency, intensity, time, and type of exercise. The American College of Sports Medicine’s Exercise is Medicine initiative provides a prescription template you can use.
3. Measure what they do
Measurement helps the patient adopt the new behavior, and it helps the physician provide tailored advice going forward, Dr. Thornton says.
With the rise of health-monitoring wearables, tracking activity has never been easier. Of course, not everyone wants to (or can afford to) use a smartwatch or fitness tracker.
For tech-averse patients, ask if they’re willing to write something down, like how many minutes they spent walking, or how many yoga classes they attended. You may never get this from some patients, but it never hurts to ask.
4. Refer out when necessary
This brings us to a sticky issue for many physicians: lack of confidence in their ability to speak authoritatively about physical activity. “In most cases, you can absolutely say, ‘Start slow, go gradually,’ that kind of thing,” Dr. Thornton says. “As with anything, confidence will come with practice.”
For specific prescriptive advice, check out the Exercise is Medicine website, which also has handouts you can share with patients and information for specific conditions. If your patient has prediabetes, you can also point them toward the CDC’s diabetes prevention program, which is available in-person or online and may be free or covered by insurance.
If a patient has contraindications, refer out. If you don’t have exercise or rehab professionals in your network, Dr. Thornton recommends reaching out to your regional or national association of sports-medicine professionals. You should be able to find it with a quick Google search.
5. Follow up
Ask about physical activity during every contact, either in person or online.
Dr. Thornton says the second and fifth steps matter most to patients, especially when the prescription and follow-up come from their primary care physician, rather than a nurse or physician assistant to whom you’ve delegated the task.
“The value comes in having a physician emphasize the importance,” Dr. Thornton says. The more time you spend on it, the more that value comes through.
What NOT to say to patients about exercise
This might surprise you:
“I definitely don’t think telling people the official recommendations for physical activity is useful,” says Yoni Freedhoff, MD, an associate professor of family medicine at the University of Ottawa and medical director of the Bariatric Medical Institute. “If anything, I’d venture it’s counterproductive.”
It’s not that there’s anything wrong with the recommended minimum – 150 minutes of moderate-to-vigorous-intensity physical activity per week. The problem is what it says to a patient who doesn’t come close to those standards.
“Few real-world people have the interest, time, energy, or privilege to achieve them,” Dr. Freedhoff says. “Many will recognize that instantly and consequently feel [that] less than that is pointless.”
And that, Dr. Thornton says, is categorically not true. “Even minimal physical activity, in some cases, is beneficial.”
You also want to avoid any explicit connection between exercise and weight loss, Dr. Thornton says.
Though many people do connect the two, the link is often negative, notes a 2019 study from the University of Toronto., triggering painful memories that might go all the way back to gym class.
Try this pivot from Dr. Freedhoff: “Focus on the role of exercise in mitigating the risks of weight,” he says – like decreasing pain, increasing energy, and improving sleep.
How to motivate patients to move
New research backs up this more positive approach. In a study published in Annals of Internal Medicine, doctors in the United Kingdom who emphasized benefits and minimized health harms convinced more patients to join a weight management program than negative or neutral docs did. These doctors conveyed optimism and excitement, smiling and avoiding any mention of obesity or body mass index.
Exactly what benefits inspire change will be different for each patient. But in general, the more immediate the benefit, the more motivating it will be.
As the University of Toronto study noted, patients weren’t motivated by vague, distant goals like “increasing life expectancy or avoiding health problems many years in the future.”
They’re much more likely to take action to avoid surgery, reduce medications, or minimize the risk of falling.
For an older patient, Dr. Freedhoff says, “focusing on the preservation of functional independence can be extremely motivating.” That’s especially true if the patient has vivid memories of seeing a sedentary loved one decline late in life.
For patients who may be more focused on appearance, they could respond to the idea of improving their body composition. For that, “we talk about the quality of weight loss,” says Spencer Nadolsky, DO, an obesity and lipid specialist and medical director of WeightWatchers. “Ultimately, exercise helps shape the body instead of just changing the number on the scale.”
Reducing resistance to resistance training
A conversation about reshaping the body or avoiding age-related disabilities leads naturally to resistance training.
“I always frame resistance training as the single most valuable thing a person might do to try to preserve their functional independence,” Dr. Freedhoff says. If the patient is over 65, he won’t wait for them to show an interest. “I’ll absolutely bring it up with them directly.”
Dr. Freedhoff has an on-site training facility where trainers show patients how to work out at home with minimal equipment, like dumbbells and resistance bands.
Most doctors, however, don’t have those options. That can lead to a tricky conversation. Participants in the University of Toronto study told the authors they disliked the gym, finding it “boring, intimidating, or discouraging.”
And yet, “a common suggestion ... from health care providers was to join a gym.”
Many patients, Spencer Nadolsky, MD, says, associate strength training with “grunting, groaning, or getting ‘bulky’ vs. ‘toned.’ ” Memories of soreness from overzealous workouts are another barrier.
He recommends “starting small and slow,” with one or two full-body workouts a week. Those initial workouts might include just one to two sets of four to five exercises. “Consider if someone is exercising at home or in a gym to build a routine around equipment that’s available to them,” Dr. Nadolsky says.
Once you determine what you have to work with, help the patient choose exercises that fit their needs, goals, preferences, limitations, and prior injuries.
One more consideration: While Dr. Nadolsky tries to “stay away from telling a patient they need to do specific types of exercise to be successful,” he makes an exception for patients who’re taking a GLP-1 agonist. “There is a concern for muscle mass loss along with fat loss.”
Practicing, preaching, and checking privilege
When Dr. Thornton, Dr. Freedhoff, and Dr. Nadolsky discuss exercise, their patients know they practice what they preach.
Dr. Nadolsky, who was a nationally ranked wrestler at the University of North Carolina, hosts the Docs Who Lift podcast with his brother, Karl Nadolsky, MD.
Dr. Freedhoff is also a lifter and fitness enthusiast, and Dr. Thornton was a world-class rower whose team came within 0.8 seconds of a silver medal at the Beijing Olympics. (They finished fourth.)
But not all physicians follow their own lifestyle advice, Dr. Freedhoff says. That doesn’t make them bad doctors – it makes them human.
“I’ve done 300 minutes a week of exercise” – the recommended amount for weight maintenance – “to see what’s involved,” Dr. Freedhoff says. “That’s far, far, far from a trivial amount.”
That leads to this advice for his fellow physicians:
“The most important thing to know about exercise is that finding the time and having the health to do so is a privilege,” he says.
Understanding that is crucial for assessing your patient’s needs and providing the right help.
A version of this article first appeared on Medscape.com.
Even one night in the ED raises risk for death
This transcript has been edited for clarity.
As a consulting nephrologist, I go all over the hospital. Medicine floors, surgical floors, the ICU – I’ve even done consults in the operating room. And more and more, I do consults in the emergency department.
The reason I am doing more consults in the ED is not because the ED docs are getting gun shy with creatinine increases; it’s because patients are staying for extended periods in the ED despite being formally admitted to the hospital. It’s a phenomenon known as boarding, because there are simply not enough beds. You know the scene if you have ever been to a busy hospital: The ED is full to breaking, with patients on stretchers in hallways. It can often feel more like a warzone than a place for healing.
This is a huge problem.
The Joint Commission specifies that admitted patients should spend no more than 4 hours in the ED waiting for a bed in the hospital.
That is, based on what I’ve seen, hugely ambitious. But I should point out that I work in a hospital that runs near capacity all the time, and studies – from some of my Yale colleagues, actually – have shown that once hospital capacity exceeds 85%, boarding rates skyrocket.
I want to discuss some of the causes of extended boarding and some solutions. But before that, I should prove to you that this really matters, and for that we are going to dig in to a new study which suggests that ED boarding kills.
To put some hard numbers to the boarding problem, we turn to this paper out of France, appearing in JAMA Internal Medicine.
This is a unique study design. Basically, on a single day – Dec. 12, 2022 – researchers fanned out across France to 97 EDs and started counting patients. The study focused on those older than age 75 who were admitted to a hospital ward from the ED. The researchers then defined two groups: those who were sent up to the hospital floor before midnight, and those who spent at least from midnight until 8 AM in the ED (basically, people forced to sleep in the ED for a night). The middle-ground people who were sent up between midnight and 8 AM were excluded.
The baseline characteristics between the two groups of patients were pretty similar: median age around 86, 55% female. There were no significant differences in comorbidities. That said, comporting with previous studies, people in an urban ED, an academic ED, or a busy ED were much more likely to board overnight.
So, what we have are two similar groups of patients treated quite differently. Not quite a randomized trial, given the hospital differences, but not bad for purposes of analysis.
Here are the most important numbers from the trial:
This difference held up even after adjustment for patient and hospital characteristics. Put another way, you’d need to send 22 patients to the floor instead of boarding in the ED to save one life. Not a bad return on investment.
It’s not entirely clear what the mechanism for the excess mortality might be, but the researchers note that patients kept in the ED overnight were about twice as likely to have a fall during their hospital stay – not surprising, given the dangers of gurneys in hallways and the sleep deprivation that trying to rest in a busy ED engenders.
I should point out that this could be worse in the United States. French ED doctors continue to care for admitted patients boarding in the ED, whereas in many hospitals in the United States, admitted patients are the responsibility of the floor team, regardless of where they are, making it more likely that these individuals may be neglected.
So, if boarding in the ED is a life-threatening situation, why do we do it? What conditions predispose to this?
You’ll hear a lot of talk, mostly from hospital administrators, saying that this is simply a problem of supply and demand. There are not enough beds for the number of patients who need beds. And staffing shortages don’t help either.
However, they never want to talk about the reasons for the staffing shortages, like poor pay, poor support, and, of course, the moral injury of treating patients in hallways.
The issue of volume is real. We could do a lot to prevent ED visits and hospital admissions by providing better access to preventive and primary care and improving our outpatient mental health infrastructure. But I think this framing passes the buck a little.
Another reason ED boarding occurs is the way our health care system is paid for. If you are building a hospital, you have little incentive to build in excess capacity. The most efficient hospital, from a profit-and-loss standpoint, is one that is 100% full as often as possible. That may be fine at times, but throw in a respiratory virus or even a pandemic, and those systems fracture under the pressure.
Let us also remember that not all hospital beds are given to patients who acutely need hospital beds. Many beds, in many hospitals, are necessary to handle postoperative patients undergoing elective procedures. Those patients having a knee replacement or abdominoplasty don’t spend the night in the ED when they leave the OR; they go to a hospital bed. And those procedures are – let’s face it – more profitable than an ED admission for a medical issue. That’s why, even when hospitals expand the number of beds they have, they do it with an eye toward increasing the rate of those profitable procedures, not decreasing the burden faced by their ED.
For now, the band-aid to the solution might be to better triage individuals boarding in the ED for floor access, prioritizing those of older age, greater frailty, or more medical complexity. But it feels like a stop-gap measure as long as the incentives are aligned to view an empty hospital bed as a sign of failure in the health system instead of success.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
As a consulting nephrologist, I go all over the hospital. Medicine floors, surgical floors, the ICU – I’ve even done consults in the operating room. And more and more, I do consults in the emergency department.
The reason I am doing more consults in the ED is not because the ED docs are getting gun shy with creatinine increases; it’s because patients are staying for extended periods in the ED despite being formally admitted to the hospital. It’s a phenomenon known as boarding, because there are simply not enough beds. You know the scene if you have ever been to a busy hospital: The ED is full to breaking, with patients on stretchers in hallways. It can often feel more like a warzone than a place for healing.
This is a huge problem.
The Joint Commission specifies that admitted patients should spend no more than 4 hours in the ED waiting for a bed in the hospital.
That is, based on what I’ve seen, hugely ambitious. But I should point out that I work in a hospital that runs near capacity all the time, and studies – from some of my Yale colleagues, actually – have shown that once hospital capacity exceeds 85%, boarding rates skyrocket.
I want to discuss some of the causes of extended boarding and some solutions. But before that, I should prove to you that this really matters, and for that we are going to dig in to a new study which suggests that ED boarding kills.
To put some hard numbers to the boarding problem, we turn to this paper out of France, appearing in JAMA Internal Medicine.
This is a unique study design. Basically, on a single day – Dec. 12, 2022 – researchers fanned out across France to 97 EDs and started counting patients. The study focused on those older than age 75 who were admitted to a hospital ward from the ED. The researchers then defined two groups: those who were sent up to the hospital floor before midnight, and those who spent at least from midnight until 8 AM in the ED (basically, people forced to sleep in the ED for a night). The middle-ground people who were sent up between midnight and 8 AM were excluded.
The baseline characteristics between the two groups of patients were pretty similar: median age around 86, 55% female. There were no significant differences in comorbidities. That said, comporting with previous studies, people in an urban ED, an academic ED, or a busy ED were much more likely to board overnight.
So, what we have are two similar groups of patients treated quite differently. Not quite a randomized trial, given the hospital differences, but not bad for purposes of analysis.
Here are the most important numbers from the trial:
This difference held up even after adjustment for patient and hospital characteristics. Put another way, you’d need to send 22 patients to the floor instead of boarding in the ED to save one life. Not a bad return on investment.
It’s not entirely clear what the mechanism for the excess mortality might be, but the researchers note that patients kept in the ED overnight were about twice as likely to have a fall during their hospital stay – not surprising, given the dangers of gurneys in hallways and the sleep deprivation that trying to rest in a busy ED engenders.
I should point out that this could be worse in the United States. French ED doctors continue to care for admitted patients boarding in the ED, whereas in many hospitals in the United States, admitted patients are the responsibility of the floor team, regardless of where they are, making it more likely that these individuals may be neglected.
So, if boarding in the ED is a life-threatening situation, why do we do it? What conditions predispose to this?
You’ll hear a lot of talk, mostly from hospital administrators, saying that this is simply a problem of supply and demand. There are not enough beds for the number of patients who need beds. And staffing shortages don’t help either.
However, they never want to talk about the reasons for the staffing shortages, like poor pay, poor support, and, of course, the moral injury of treating patients in hallways.
The issue of volume is real. We could do a lot to prevent ED visits and hospital admissions by providing better access to preventive and primary care and improving our outpatient mental health infrastructure. But I think this framing passes the buck a little.
Another reason ED boarding occurs is the way our health care system is paid for. If you are building a hospital, you have little incentive to build in excess capacity. The most efficient hospital, from a profit-and-loss standpoint, is one that is 100% full as often as possible. That may be fine at times, but throw in a respiratory virus or even a pandemic, and those systems fracture under the pressure.
Let us also remember that not all hospital beds are given to patients who acutely need hospital beds. Many beds, in many hospitals, are necessary to handle postoperative patients undergoing elective procedures. Those patients having a knee replacement or abdominoplasty don’t spend the night in the ED when they leave the OR; they go to a hospital bed. And those procedures are – let’s face it – more profitable than an ED admission for a medical issue. That’s why, even when hospitals expand the number of beds they have, they do it with an eye toward increasing the rate of those profitable procedures, not decreasing the burden faced by their ED.
For now, the band-aid to the solution might be to better triage individuals boarding in the ED for floor access, prioritizing those of older age, greater frailty, or more medical complexity. But it feels like a stop-gap measure as long as the incentives are aligned to view an empty hospital bed as a sign of failure in the health system instead of success.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
As a consulting nephrologist, I go all over the hospital. Medicine floors, surgical floors, the ICU – I’ve even done consults in the operating room. And more and more, I do consults in the emergency department.
The reason I am doing more consults in the ED is not because the ED docs are getting gun shy with creatinine increases; it’s because patients are staying for extended periods in the ED despite being formally admitted to the hospital. It’s a phenomenon known as boarding, because there are simply not enough beds. You know the scene if you have ever been to a busy hospital: The ED is full to breaking, with patients on stretchers in hallways. It can often feel more like a warzone than a place for healing.
This is a huge problem.
The Joint Commission specifies that admitted patients should spend no more than 4 hours in the ED waiting for a bed in the hospital.
That is, based on what I’ve seen, hugely ambitious. But I should point out that I work in a hospital that runs near capacity all the time, and studies – from some of my Yale colleagues, actually – have shown that once hospital capacity exceeds 85%, boarding rates skyrocket.
I want to discuss some of the causes of extended boarding and some solutions. But before that, I should prove to you that this really matters, and for that we are going to dig in to a new study which suggests that ED boarding kills.
To put some hard numbers to the boarding problem, we turn to this paper out of France, appearing in JAMA Internal Medicine.
This is a unique study design. Basically, on a single day – Dec. 12, 2022 – researchers fanned out across France to 97 EDs and started counting patients. The study focused on those older than age 75 who were admitted to a hospital ward from the ED. The researchers then defined two groups: those who were sent up to the hospital floor before midnight, and those who spent at least from midnight until 8 AM in the ED (basically, people forced to sleep in the ED for a night). The middle-ground people who were sent up between midnight and 8 AM were excluded.
The baseline characteristics between the two groups of patients were pretty similar: median age around 86, 55% female. There were no significant differences in comorbidities. That said, comporting with previous studies, people in an urban ED, an academic ED, or a busy ED were much more likely to board overnight.
So, what we have are two similar groups of patients treated quite differently. Not quite a randomized trial, given the hospital differences, but not bad for purposes of analysis.
Here are the most important numbers from the trial:
This difference held up even after adjustment for patient and hospital characteristics. Put another way, you’d need to send 22 patients to the floor instead of boarding in the ED to save one life. Not a bad return on investment.
It’s not entirely clear what the mechanism for the excess mortality might be, but the researchers note that patients kept in the ED overnight were about twice as likely to have a fall during their hospital stay – not surprising, given the dangers of gurneys in hallways and the sleep deprivation that trying to rest in a busy ED engenders.
I should point out that this could be worse in the United States. French ED doctors continue to care for admitted patients boarding in the ED, whereas in many hospitals in the United States, admitted patients are the responsibility of the floor team, regardless of where they are, making it more likely that these individuals may be neglected.
So, if boarding in the ED is a life-threatening situation, why do we do it? What conditions predispose to this?
You’ll hear a lot of talk, mostly from hospital administrators, saying that this is simply a problem of supply and demand. There are not enough beds for the number of patients who need beds. And staffing shortages don’t help either.
However, they never want to talk about the reasons for the staffing shortages, like poor pay, poor support, and, of course, the moral injury of treating patients in hallways.
The issue of volume is real. We could do a lot to prevent ED visits and hospital admissions by providing better access to preventive and primary care and improving our outpatient mental health infrastructure. But I think this framing passes the buck a little.
Another reason ED boarding occurs is the way our health care system is paid for. If you are building a hospital, you have little incentive to build in excess capacity. The most efficient hospital, from a profit-and-loss standpoint, is one that is 100% full as often as possible. That may be fine at times, but throw in a respiratory virus or even a pandemic, and those systems fracture under the pressure.
Let us also remember that not all hospital beds are given to patients who acutely need hospital beds. Many beds, in many hospitals, are necessary to handle postoperative patients undergoing elective procedures. Those patients having a knee replacement or abdominoplasty don’t spend the night in the ED when they leave the OR; they go to a hospital bed. And those procedures are – let’s face it – more profitable than an ED admission for a medical issue. That’s why, even when hospitals expand the number of beds they have, they do it with an eye toward increasing the rate of those profitable procedures, not decreasing the burden faced by their ED.
For now, the band-aid to the solution might be to better triage individuals boarding in the ED for floor access, prioritizing those of older age, greater frailty, or more medical complexity. But it feels like a stop-gap measure as long as the incentives are aligned to view an empty hospital bed as a sign of failure in the health system instead of success.
F. Perry Wilson, MD, MSCE, is an associate professor of medicine and public health and director of Yale’s Clinical and Translational Research Accelerator. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Pervasive ‘forever chemicals’ linked to thyroid cancer?
The study suggests that higher exposure to per- and polyfluoroalkyl substances (PFAS), specifically perfluorooctanesulfonic acid (n-PFOS), may increase a person’s risk for thyroid cancer by 56%.
Several news outlets played up the findings, published online in eBioMedicine. “Dangerous ‘Forever Chemicals’ in Your Everyday Items Are Causing Cancer,” Newsweek reported.
But Gideon Meyerowitz-Katz, PhD, an epidemiologist at the University of Wollongong (Australia), voiced his skepticism.
“While it’s possible that PFAS might be causing thyroid cancer, the evidence thus far is unconvincing and probably not worth worrying about,” said Dr. Meyerowitz-Katz, who was not involved in the research.
PFAS and thyroid cancer
PFAS are a class of widely used synthetic chemicals found in many consumer and industrial products, including nonstick cookware, stain-repellent carpets, waterproof rain gear, microwave popcorn bags, and firefighting foam.
These substances have been dubbed “forever chemicals” because they do not degrade and are ubiquitous in the environment.
Exposure to endocrine-disrupting chemicals, including PFAS, has been identified as a potential risk factor for thyroid cancer, with some research linking PFAS exposure to thyroid dysfunction and carcinogenesis.
To investigate further, the researchers performed a nested case-control study of 86 patients with thyroid cancer using plasma samples collected at or before diagnosis and 86 controls without cancer who were matched on age, sex, race/ethnicity, body weight, smoking status, and year of sample collection.
Eighteen individual PFAS were measured in plasma samples; 10 were undetectable and were therefore excluded from the analysis. Of the remaining eight PFAS, only one showed a statistically significant correlation with thyroid cancer.
Specifically, the researchers found that exposure to n-PFOS was associated with a 56% increased risk for thyroid cancer among people who had a high level of the chemical in their blood (adjusted odds ratio, 1.56; P = .004). The results were similar when patients with papillary thyroid cancer only were included (aOR, 1.56; P = .009).
A separate longitudinal analysis of 31 patients diagnosed with thyroid cancer 1 year or more after plasma sample collection and 31 controls confirmed the positive association between n-PFOS and thyroid cancer (aOR, 2.67; P < .001). The longitudinal analysis also suggested correlations for a few other PFAS.
“This study supports the hypothesis that PFAS exposure may be associated with increased risk of thyroid cancer,” the authors concluded.
But in a Substack post, Dr. Meyerowitz-Katz said that it’s important to put the findings into “proper context before getting terrified about this all-new cancer risk.”
First, this study was “genuinely tiny,” with data on just 88 people with thyroid cancer and 88 controls, a limitation the researchers also acknowledged.
“That’s really not enough to do any sort of robust epidemiological analysis – you can generate interesting correlations, but what those correlations mean is anyone’s guess,” Dr. Meyerowitz-Katz said.
Even more importantly, one could easily argue that the results of this study show that most PFAS aren’t associated with thyroid cancer, given that there was no strong association for seven of the eight PFAS measured, he explained.
“There are no serious methodological concerns here, but equally there’s just not much you can reasonably gather from finding a single correlation among a vast ocean of possibilities,” Dr. Meyerowitz-Katz wrote. “Maybe there’s a correlation there, but you’d need to investigate this in much bigger samples, with more controls, and better data, to understand what that correlation means.”
Bottom line, Dr. Meyerowitz-Katz explained, is that “the link between PFAS and thyroid cancer is, at best, incredibly weak.”
Funding for the study was provided by the National Institutes of Health and The Andrea and Charles Bronfman Philanthropies. One coauthor is cofounder of Linus Biotechnology and is owner of a license agreement with NIES (Japan); received honoraria and travel compensation for lectures for the Bio-Echo and Brin foundations; and has 22 patents at various stages. Dr. Meyerowitz-Katz has no relevant disclosures.
A version of this article appeared on Medscape.com.
The study suggests that higher exposure to per- and polyfluoroalkyl substances (PFAS), specifically perfluorooctanesulfonic acid (n-PFOS), may increase a person’s risk for thyroid cancer by 56%.
Several news outlets played up the findings, published online in eBioMedicine. “Dangerous ‘Forever Chemicals’ in Your Everyday Items Are Causing Cancer,” Newsweek reported.
But Gideon Meyerowitz-Katz, PhD, an epidemiologist at the University of Wollongong (Australia), voiced his skepticism.
“While it’s possible that PFAS might be causing thyroid cancer, the evidence thus far is unconvincing and probably not worth worrying about,” said Dr. Meyerowitz-Katz, who was not involved in the research.
PFAS and thyroid cancer
PFAS are a class of widely used synthetic chemicals found in many consumer and industrial products, including nonstick cookware, stain-repellent carpets, waterproof rain gear, microwave popcorn bags, and firefighting foam.
These substances have been dubbed “forever chemicals” because they do not degrade and are ubiquitous in the environment.
Exposure to endocrine-disrupting chemicals, including PFAS, has been identified as a potential risk factor for thyroid cancer, with some research linking PFAS exposure to thyroid dysfunction and carcinogenesis.
To investigate further, the researchers performed a nested case-control study of 86 patients with thyroid cancer using plasma samples collected at or before diagnosis and 86 controls without cancer who were matched on age, sex, race/ethnicity, body weight, smoking status, and year of sample collection.
Eighteen individual PFAS were measured in plasma samples; 10 were undetectable and were therefore excluded from the analysis. Of the remaining eight PFAS, only one showed a statistically significant correlation with thyroid cancer.
Specifically, the researchers found that exposure to n-PFOS was associated with a 56% increased risk for thyroid cancer among people who had a high level of the chemical in their blood (adjusted odds ratio, 1.56; P = .004). The results were similar when patients with papillary thyroid cancer only were included (aOR, 1.56; P = .009).
A separate longitudinal analysis of 31 patients diagnosed with thyroid cancer 1 year or more after plasma sample collection and 31 controls confirmed the positive association between n-PFOS and thyroid cancer (aOR, 2.67; P < .001). The longitudinal analysis also suggested correlations for a few other PFAS.
“This study supports the hypothesis that PFAS exposure may be associated with increased risk of thyroid cancer,” the authors concluded.
But in a Substack post, Dr. Meyerowitz-Katz said that it’s important to put the findings into “proper context before getting terrified about this all-new cancer risk.”
First, this study was “genuinely tiny,” with data on just 88 people with thyroid cancer and 88 controls, a limitation the researchers also acknowledged.
“That’s really not enough to do any sort of robust epidemiological analysis – you can generate interesting correlations, but what those correlations mean is anyone’s guess,” Dr. Meyerowitz-Katz said.
Even more importantly, one could easily argue that the results of this study show that most PFAS aren’t associated with thyroid cancer, given that there was no strong association for seven of the eight PFAS measured, he explained.
“There are no serious methodological concerns here, but equally there’s just not much you can reasonably gather from finding a single correlation among a vast ocean of possibilities,” Dr. Meyerowitz-Katz wrote. “Maybe there’s a correlation there, but you’d need to investigate this in much bigger samples, with more controls, and better data, to understand what that correlation means.”
Bottom line, Dr. Meyerowitz-Katz explained, is that “the link between PFAS and thyroid cancer is, at best, incredibly weak.”
Funding for the study was provided by the National Institutes of Health and The Andrea and Charles Bronfman Philanthropies. One coauthor is cofounder of Linus Biotechnology and is owner of a license agreement with NIES (Japan); received honoraria and travel compensation for lectures for the Bio-Echo and Brin foundations; and has 22 patents at various stages. Dr. Meyerowitz-Katz has no relevant disclosures.
A version of this article appeared on Medscape.com.
The study suggests that higher exposure to per- and polyfluoroalkyl substances (PFAS), specifically perfluorooctanesulfonic acid (n-PFOS), may increase a person’s risk for thyroid cancer by 56%.
Several news outlets played up the findings, published online in eBioMedicine. “Dangerous ‘Forever Chemicals’ in Your Everyday Items Are Causing Cancer,” Newsweek reported.
But Gideon Meyerowitz-Katz, PhD, an epidemiologist at the University of Wollongong (Australia), voiced his skepticism.
“While it’s possible that PFAS might be causing thyroid cancer, the evidence thus far is unconvincing and probably not worth worrying about,” said Dr. Meyerowitz-Katz, who was not involved in the research.
PFAS and thyroid cancer
PFAS are a class of widely used synthetic chemicals found in many consumer and industrial products, including nonstick cookware, stain-repellent carpets, waterproof rain gear, microwave popcorn bags, and firefighting foam.
These substances have been dubbed “forever chemicals” because they do not degrade and are ubiquitous in the environment.
Exposure to endocrine-disrupting chemicals, including PFAS, has been identified as a potential risk factor for thyroid cancer, with some research linking PFAS exposure to thyroid dysfunction and carcinogenesis.
To investigate further, the researchers performed a nested case-control study of 86 patients with thyroid cancer using plasma samples collected at or before diagnosis and 86 controls without cancer who were matched on age, sex, race/ethnicity, body weight, smoking status, and year of sample collection.
Eighteen individual PFAS were measured in plasma samples; 10 were undetectable and were therefore excluded from the analysis. Of the remaining eight PFAS, only one showed a statistically significant correlation with thyroid cancer.
Specifically, the researchers found that exposure to n-PFOS was associated with a 56% increased risk for thyroid cancer among people who had a high level of the chemical in their blood (adjusted odds ratio, 1.56; P = .004). The results were similar when patients with papillary thyroid cancer only were included (aOR, 1.56; P = .009).
A separate longitudinal analysis of 31 patients diagnosed with thyroid cancer 1 year or more after plasma sample collection and 31 controls confirmed the positive association between n-PFOS and thyroid cancer (aOR, 2.67; P < .001). The longitudinal analysis also suggested correlations for a few other PFAS.
“This study supports the hypothesis that PFAS exposure may be associated with increased risk of thyroid cancer,” the authors concluded.
But in a Substack post, Dr. Meyerowitz-Katz said that it’s important to put the findings into “proper context before getting terrified about this all-new cancer risk.”
First, this study was “genuinely tiny,” with data on just 88 people with thyroid cancer and 88 controls, a limitation the researchers also acknowledged.
“That’s really not enough to do any sort of robust epidemiological analysis – you can generate interesting correlations, but what those correlations mean is anyone’s guess,” Dr. Meyerowitz-Katz said.
Even more importantly, one could easily argue that the results of this study show that most PFAS aren’t associated with thyroid cancer, given that there was no strong association for seven of the eight PFAS measured, he explained.
“There are no serious methodological concerns here, but equally there’s just not much you can reasonably gather from finding a single correlation among a vast ocean of possibilities,” Dr. Meyerowitz-Katz wrote. “Maybe there’s a correlation there, but you’d need to investigate this in much bigger samples, with more controls, and better data, to understand what that correlation means.”
Bottom line, Dr. Meyerowitz-Katz explained, is that “the link between PFAS and thyroid cancer is, at best, incredibly weak.”
Funding for the study was provided by the National Institutes of Health and The Andrea and Charles Bronfman Philanthropies. One coauthor is cofounder of Linus Biotechnology and is owner of a license agreement with NIES (Japan); received honoraria and travel compensation for lectures for the Bio-Echo and Brin foundations; and has 22 patents at various stages. Dr. Meyerowitz-Katz has no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM EBIOMEDICINE
No longer a death sentence, HIV diagnosis still hits hard
Veronica Brady and her team at the University of Texas Health Science Center, Houston, sat down with 37 people diagnosed with HIV or AIDS to ask them what that felt like.
“The results were really eye-opening and sad,” says Brady, PhD, RN, from the Cizik School of Nursing with UTHealth, Houston.
Many of the people Dr. Brady and her team spoke with were diagnosed through routine or random testing. They ranged in age from 21 years to 65 and said they did not know how they had been infected and felt shocked, freaked out, scared, and in a state of disbelief.
Their conversations about being diagnosed with HIV, presented at the annual meeting of the Association of Nurses in AIDS Care in New Orleans, also described how symptoms of the disease or side effects from treatment can have a huge impact on the daily lives of those affected.
Jesse Milan Jr., president of AIDS United, an HIV advocacy organization based in Washington, D.C., says he recognizes all of these feelings from his own experience with HIV after being diagnosed more than 40 years ago.
“All of those have come up over the years,” he says. “They are all relevant and important at different times.”
For Mr. Milan, less was known about the virus at the time of his diagnosis, and he watched loved ones die. He lived to see the introduction of antiretroviral therapies and receive treatment when his partner and many of his friends did not.
Effective treatments
There is a marked difference between the reaction of people diagnosed with HIV years ago and those diagnosed more recently, Dr. Brady explains. Those diagnosed before much was known about the virus and before there were effective treatments were more frightened, she says, whereas people hearing the news recently are much less worried and understand that if they take their medication, they will be fine.
Still, Mr. Milan says when he talks to people diagnosed now, they seem to experience more shame and embarrassment than before. Because it is long known how to prevent HIV infection, they often worry what people will think if they disclose their status. “It makes things harder for people diagnosed today,” says Mr. Milan. “There is a different level of embarrassment tinged with, ‘Why was I so stupid?’ ”
Diagnosis can also be hard on health care professionals, says Dr. Brady. “You never want to tell anyone they’re sick with a chronic disease, especially younger people,” she adds. “You know you’re adding a burden to someone’s life.”
Symptoms and side effects of treatment also had an important impact on the people in this report, with most aspects of their lives affected, including work, relationships, mood, and daily activities.
Clinicians should be supportive and spend some time sitting with patients as they come to terms with the diagnosis and its implications. They should help them understand what to expect and talk about how – or whether – to talk about their status with family and friends. “You need to show you care about the person and that they are not alone,” Dr. Brady says.
And most of all, clinicians need to explain that patients can live a long and healthy life and go on to become whoever they want to be. “Twenty years ago, we wouldn’t have as hopeful a message as we do now,” she says.
Hope is the most important thing for doctors and nurses to communicate to their patients. “There are medications available, and it will be okay. You don’t have to die,” Mr. Milan says. “That’s the core message that everyone needs to hear, whether they were diagnosed 30 years ago or 30 minutes ago.”
A version of this article appeared on Medscape.com.
Veronica Brady and her team at the University of Texas Health Science Center, Houston, sat down with 37 people diagnosed with HIV or AIDS to ask them what that felt like.
“The results were really eye-opening and sad,” says Brady, PhD, RN, from the Cizik School of Nursing with UTHealth, Houston.
Many of the people Dr. Brady and her team spoke with were diagnosed through routine or random testing. They ranged in age from 21 years to 65 and said they did not know how they had been infected and felt shocked, freaked out, scared, and in a state of disbelief.
Their conversations about being diagnosed with HIV, presented at the annual meeting of the Association of Nurses in AIDS Care in New Orleans, also described how symptoms of the disease or side effects from treatment can have a huge impact on the daily lives of those affected.
Jesse Milan Jr., president of AIDS United, an HIV advocacy organization based in Washington, D.C., says he recognizes all of these feelings from his own experience with HIV after being diagnosed more than 40 years ago.
“All of those have come up over the years,” he says. “They are all relevant and important at different times.”
For Mr. Milan, less was known about the virus at the time of his diagnosis, and he watched loved ones die. He lived to see the introduction of antiretroviral therapies and receive treatment when his partner and many of his friends did not.
Effective treatments
There is a marked difference between the reaction of people diagnosed with HIV years ago and those diagnosed more recently, Dr. Brady explains. Those diagnosed before much was known about the virus and before there were effective treatments were more frightened, she says, whereas people hearing the news recently are much less worried and understand that if they take their medication, they will be fine.
Still, Mr. Milan says when he talks to people diagnosed now, they seem to experience more shame and embarrassment than before. Because it is long known how to prevent HIV infection, they often worry what people will think if they disclose their status. “It makes things harder for people diagnosed today,” says Mr. Milan. “There is a different level of embarrassment tinged with, ‘Why was I so stupid?’ ”
Diagnosis can also be hard on health care professionals, says Dr. Brady. “You never want to tell anyone they’re sick with a chronic disease, especially younger people,” she adds. “You know you’re adding a burden to someone’s life.”
Symptoms and side effects of treatment also had an important impact on the people in this report, with most aspects of their lives affected, including work, relationships, mood, and daily activities.
Clinicians should be supportive and spend some time sitting with patients as they come to terms with the diagnosis and its implications. They should help them understand what to expect and talk about how – or whether – to talk about their status with family and friends. “You need to show you care about the person and that they are not alone,” Dr. Brady says.
And most of all, clinicians need to explain that patients can live a long and healthy life and go on to become whoever they want to be. “Twenty years ago, we wouldn’t have as hopeful a message as we do now,” she says.
Hope is the most important thing for doctors and nurses to communicate to their patients. “There are medications available, and it will be okay. You don’t have to die,” Mr. Milan says. “That’s the core message that everyone needs to hear, whether they were diagnosed 30 years ago or 30 minutes ago.”
A version of this article appeared on Medscape.com.
Veronica Brady and her team at the University of Texas Health Science Center, Houston, sat down with 37 people diagnosed with HIV or AIDS to ask them what that felt like.
“The results were really eye-opening and sad,” says Brady, PhD, RN, from the Cizik School of Nursing with UTHealth, Houston.
Many of the people Dr. Brady and her team spoke with were diagnosed through routine or random testing. They ranged in age from 21 years to 65 and said they did not know how they had been infected and felt shocked, freaked out, scared, and in a state of disbelief.
Their conversations about being diagnosed with HIV, presented at the annual meeting of the Association of Nurses in AIDS Care in New Orleans, also described how symptoms of the disease or side effects from treatment can have a huge impact on the daily lives of those affected.
Jesse Milan Jr., president of AIDS United, an HIV advocacy organization based in Washington, D.C., says he recognizes all of these feelings from his own experience with HIV after being diagnosed more than 40 years ago.
“All of those have come up over the years,” he says. “They are all relevant and important at different times.”
For Mr. Milan, less was known about the virus at the time of his diagnosis, and he watched loved ones die. He lived to see the introduction of antiretroviral therapies and receive treatment when his partner and many of his friends did not.
Effective treatments
There is a marked difference between the reaction of people diagnosed with HIV years ago and those diagnosed more recently, Dr. Brady explains. Those diagnosed before much was known about the virus and before there were effective treatments were more frightened, she says, whereas people hearing the news recently are much less worried and understand that if they take their medication, they will be fine.
Still, Mr. Milan says when he talks to people diagnosed now, they seem to experience more shame and embarrassment than before. Because it is long known how to prevent HIV infection, they often worry what people will think if they disclose their status. “It makes things harder for people diagnosed today,” says Mr. Milan. “There is a different level of embarrassment tinged with, ‘Why was I so stupid?’ ”
Diagnosis can also be hard on health care professionals, says Dr. Brady. “You never want to tell anyone they’re sick with a chronic disease, especially younger people,” she adds. “You know you’re adding a burden to someone’s life.”
Symptoms and side effects of treatment also had an important impact on the people in this report, with most aspects of their lives affected, including work, relationships, mood, and daily activities.
Clinicians should be supportive and spend some time sitting with patients as they come to terms with the diagnosis and its implications. They should help them understand what to expect and talk about how – or whether – to talk about their status with family and friends. “You need to show you care about the person and that they are not alone,” Dr. Brady says.
And most of all, clinicians need to explain that patients can live a long and healthy life and go on to become whoever they want to be. “Twenty years ago, we wouldn’t have as hopeful a message as we do now,” she says.
Hope is the most important thing for doctors and nurses to communicate to their patients. “There are medications available, and it will be okay. You don’t have to die,” Mr. Milan says. “That’s the core message that everyone needs to hear, whether they were diagnosed 30 years ago or 30 minutes ago.”
A version of this article appeared on Medscape.com.