User login
Welcome to Current Psychiatry, a leading source of information, online and in print, for practitioners of psychiatry and its related subspecialties, including addiction psychiatry, child and adolescent psychiatry, and geriatric psychiatry. This Web site contains evidence-based reviews of the prevention, diagnosis, and treatment of mental illness and psychological disorders; case reports; updates on psychopharmacology; news about the specialty of psychiatry; pearls for practice; and other topics of interest and use to this audience.
Dear Drupal User: You're seeing this because you're logged in to Drupal, and not redirected to MDedge.com/psychiatry.
Depression
adolescent depression
adolescent major depressive disorder
adolescent schizophrenia
adolescent with major depressive disorder
animals
autism
baby
brexpiprazole
child
child bipolar
child depression
child schizophrenia
children with bipolar disorder
children with depression
children with major depressive disorder
compulsive behaviors
cure
elderly bipolar
elderly depression
elderly major depressive disorder
elderly schizophrenia
elderly with dementia
first break
first episode
gambling
gaming
geriatric depression
geriatric major depressive disorder
geriatric schizophrenia
infant
kid
major depressive disorder
major depressive disorder in adolescents
major depressive disorder in children
parenting
pediatric
pediatric bipolar
pediatric depression
pediatric major depressive disorder
pediatric schizophrenia
pregnancy
pregnant
rexulti
skin care
teen
wine
section[contains(@class, 'nav-hidden')]
footer[@id='footer']
div[contains(@class, 'pane-pub-article-current-psychiatry')]
div[contains(@class, 'pane-pub-home-current-psychiatry')]
div[contains(@class, 'pane-pub-topic-current-psychiatry')]
div[contains(@class, 'panel-panel-inner')]
div[contains(@class, 'pane-node-field-article-topics')]
section[contains(@class, 'footer-nav-section-wrapper')]
Dysphagia in a patient with schizophrenia: Is the antipsychotic the culprit?
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
Mr. N, age 58, has a history of schizophrenia, tobacco use disorder, and alcohol use disorder. For many years, Mr. N has been receiving IM olanzapine 2.5 mg/d to treat his schizophrenia. He lives in a psychiatric hospital but was sent to our hospital after being found to have severe oropharyngeal dysphasia on a modified barium swallow study. There was concern for aspiration due to a history of choking episodes, which had been occurring for almost 1 month. During the modified barium swallow study, Mr. N was noted to have aspiration with deep laryngeal penetration during the pharyngeal stages of swallowing to all consistencies; this did not improve with the chin-tuck maneuver. In addition, during a CT scan of the cervical spine, an osteophyte was noted at the C5-C6 level, with possible impingement of the cervical esophagus and decreased upper esophageal sphincter opening.
Due to these findings, Mr. N was sent to our emergency department (ED) for further evaluation. In the ED, his vital signs were stable. He endorsed having a cough after eating, a sensation of having food stuck in his throat, and some hoarseness. His physical examination was notable for poor dentition. Results of a standard laboratory workup were all within normal limits. X-ray was notable for hazy opacities in the right upper to mid lung zones. Mr. N was admitted to the medical unit for further evaluation and management.
Narrowing the diagnosis
Because Mr. N was aspirating both liquids and solids, it was imperative that we identify the cause as soon as possible. The consultations that followed slowly guided the treatment team toward a diagnosis of antipsychotic-induced dysphagia. Otolaryngology identified insensate larynx during a flexible fiberoptic laryngoscopy exam, which was highly suggestive of a neurological dysfunction such as dystonia. Furthermore, an esophagogastroduodenoscopy found no structural abnormalities to explain Mr. N’s dysphagia, which ruled out impingement of the cervical esophagus by the osteophyte. An MRI of the brain ruled out structural abnormalities or evidence of stroke. Finally, a speech and language pathologist confirmed decreased laryngeal closure and airway protection with a repeat modified barium swallow, which led to aspiration during swallowing. Psychiatry recommended starting diphenhydramine to treat Mr. N’s extrapyramidal symptoms (EPS). A 6-day trial was initiated, with a single 50 mg IV dose on the first day followed by 25 mL oral twice daily for the remaining 5 days. In addition, olanzapine was discontinued.
Switching to a different diet and antipsychotic
Two days after starting diphenhydramine, Mr. N was switched to a puree diet. His ability to swallow improved, and he no longer coughed. However, on repeat modified barium swallow, aspiration was still noted for all types of liquids and solids. No structural improvements were seen.
Mr. N was discharged back to his psychiatric hospital, and his antipsychotic was changed from olanzapine to oral aripiprazole 2 mg/d. The aripiprazole dose was kept low to prevent the recurrence of dystonia and because at the time, his schizophrenia was asymptomatic. Mr. N was also prescribed oral diphenhydramine 25 mL twice daily.
At a 2-week follow-up appointment, Mr. N continued to show clinical improvement on the puree diet with thin liquids and continued the prescribed medication regimen.
Dysphagia as a manifestation of EPS
All antipsychotics, and particularly first-generation agents, are associated with EPS.1 These symptoms may be the result of antagonistic binding of dopaminergic D2 receptors within mesolimbic and mesocortical pathways of the brain, as well as parts of basal ganglia such as the caudate nucleus.2
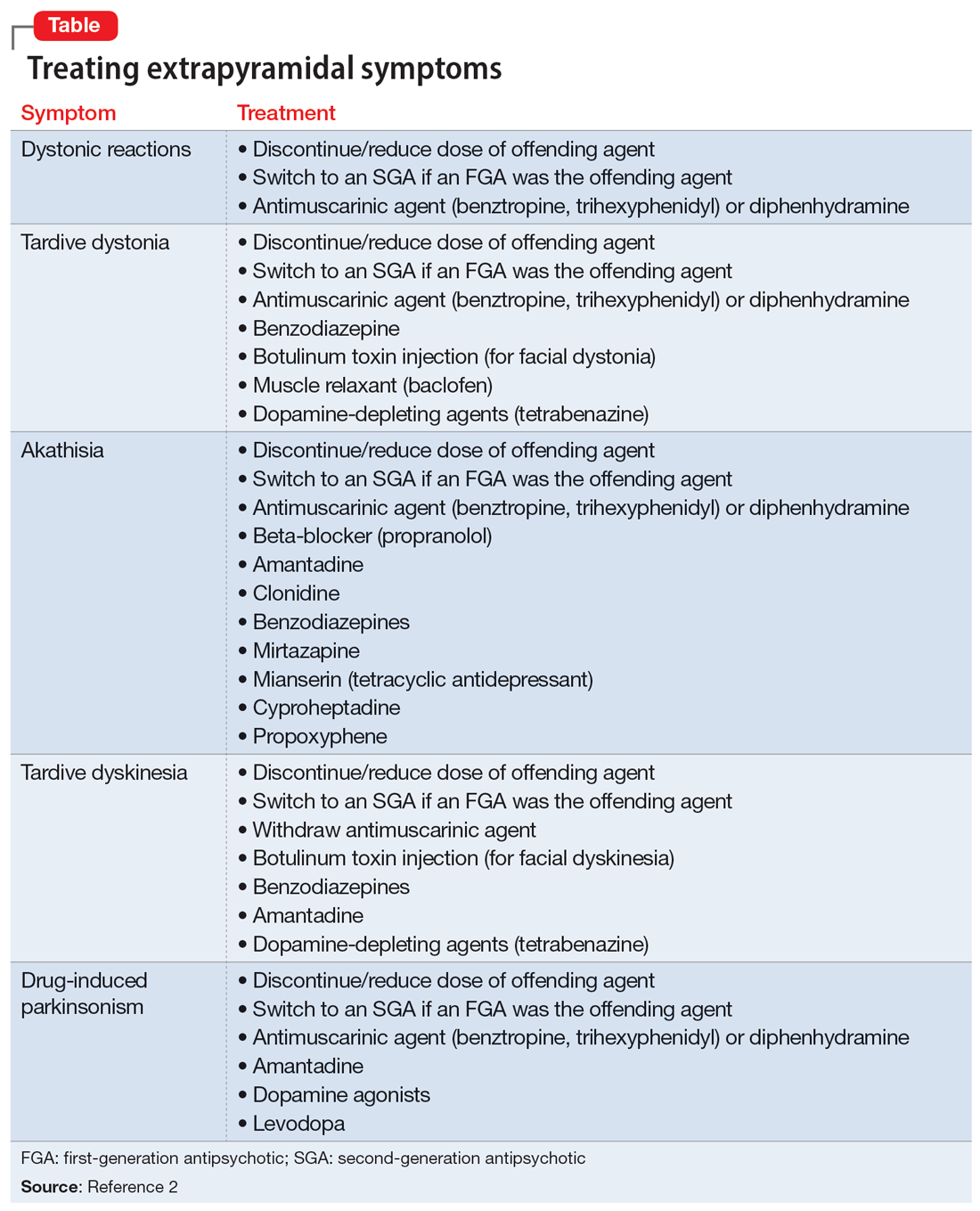
In addition to the examples listed in the Table,2 EPS can present as dysphagia, esophageal dysmotility, or aspiration, none of which may be recognized as EPS. Research has found haloperidol, loxapine, trifluoperazine, olanzapine, risperidone, quetiapine, clozapine, and aripiprazole are associated with dysphagia.3-6 Strategies to treat antipsychotic-induced dysphagia include discontinuing the antipsychotic, lowering the dose, and changing to another medication.7
1. Crouse EL, Alastanos JN, Bozymski KM, et al. Dysphagia with second-generation antipsychotics: a case report and review of the literature. Ment Health Clin. 2018;7(2):56-64. doi:10.9740/mhc.2017.03.056
2. D’Souza RS, Hooten WM. Extrapyramidal symptoms. StatPearls Publishing; 2022. Updated January 8, 2023. Accessed April 28, 2023. https://www.ncbi.nlm.nih.gov/books/NBK534115/
3. Dziewas R, Warnecke T, Schnabel M, et al. Neuroleptic-induced dysphagia: case report and literature review. Dysphagia. 2007;22(1):63-67. doi:10.1007/s00455-006-9032-9
4. Kalf JG, de Swart BJ, Bloem BR, et al. Prevalence of oropharyngeal dysphagia in Parkinson’s disease: a meta-analysis. Parkinsonism Relat Disord. 2012;18(4):311-315. doi:10.1016/j.parkreldis.2011.11.006
5. Lin TW, Lee BS, Liao YC, et al. High dosage of aripiprazole-induced dysphagia. Int J Eat Disord. 2012;45(2):305-306. doi:10.1002/eat.20934
6. Stewart JT. Dysphagia associated with risperidone therapy. Dysphagia. 2003;18(4):274-275. doi:10.1007/s00455-003-0006-x
7. Lee JC, Takeshita J. Antipsychotic-induced dysphagia: a case report. Prim Care Companion CNS Disord. 2015;17(5):10.4088/PCC.15I01792. doi:10.4088/PCC.15I01792
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
Mr. N, age 58, has a history of schizophrenia, tobacco use disorder, and alcohol use disorder. For many years, Mr. N has been receiving IM olanzapine 2.5 mg/d to treat his schizophrenia. He lives in a psychiatric hospital but was sent to our hospital after being found to have severe oropharyngeal dysphasia on a modified barium swallow study. There was concern for aspiration due to a history of choking episodes, which had been occurring for almost 1 month. During the modified barium swallow study, Mr. N was noted to have aspiration with deep laryngeal penetration during the pharyngeal stages of swallowing to all consistencies; this did not improve with the chin-tuck maneuver. In addition, during a CT scan of the cervical spine, an osteophyte was noted at the C5-C6 level, with possible impingement of the cervical esophagus and decreased upper esophageal sphincter opening.
Due to these findings, Mr. N was sent to our emergency department (ED) for further evaluation. In the ED, his vital signs were stable. He endorsed having a cough after eating, a sensation of having food stuck in his throat, and some hoarseness. His physical examination was notable for poor dentition. Results of a standard laboratory workup were all within normal limits. X-ray was notable for hazy opacities in the right upper to mid lung zones. Mr. N was admitted to the medical unit for further evaluation and management.
Narrowing the diagnosis
Because Mr. N was aspirating both liquids and solids, it was imperative that we identify the cause as soon as possible. The consultations that followed slowly guided the treatment team toward a diagnosis of antipsychotic-induced dysphagia. Otolaryngology identified insensate larynx during a flexible fiberoptic laryngoscopy exam, which was highly suggestive of a neurological dysfunction such as dystonia. Furthermore, an esophagogastroduodenoscopy found no structural abnormalities to explain Mr. N’s dysphagia, which ruled out impingement of the cervical esophagus by the osteophyte. An MRI of the brain ruled out structural abnormalities or evidence of stroke. Finally, a speech and language pathologist confirmed decreased laryngeal closure and airway protection with a repeat modified barium swallow, which led to aspiration during swallowing. Psychiatry recommended starting diphenhydramine to treat Mr. N’s extrapyramidal symptoms (EPS). A 6-day trial was initiated, with a single 50 mg IV dose on the first day followed by 25 mL oral twice daily for the remaining 5 days. In addition, olanzapine was discontinued.
Switching to a different diet and antipsychotic
Two days after starting diphenhydramine, Mr. N was switched to a puree diet. His ability to swallow improved, and he no longer coughed. However, on repeat modified barium swallow, aspiration was still noted for all types of liquids and solids. No structural improvements were seen.
Mr. N was discharged back to his psychiatric hospital, and his antipsychotic was changed from olanzapine to oral aripiprazole 2 mg/d. The aripiprazole dose was kept low to prevent the recurrence of dystonia and because at the time, his schizophrenia was asymptomatic. Mr. N was also prescribed oral diphenhydramine 25 mL twice daily.
At a 2-week follow-up appointment, Mr. N continued to show clinical improvement on the puree diet with thin liquids and continued the prescribed medication regimen.
Dysphagia as a manifestation of EPS
All antipsychotics, and particularly first-generation agents, are associated with EPS.1 These symptoms may be the result of antagonistic binding of dopaminergic D2 receptors within mesolimbic and mesocortical pathways of the brain, as well as parts of basal ganglia such as the caudate nucleus.2
In addition to the examples listed in the Table,2 EPS can present as dysphagia, esophageal dysmotility, or aspiration, none of which may be recognized as EPS. Research has found haloperidol, loxapine, trifluoperazine, olanzapine, risperidone, quetiapine, clozapine, and aripiprazole are associated with dysphagia.3-6 Strategies to treat antipsychotic-induced dysphagia include discontinuing the antipsychotic, lowering the dose, and changing to another medication.7

Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
Mr. N, age 58, has a history of schizophrenia, tobacco use disorder, and alcohol use disorder. For many years, Mr. N has been receiving IM olanzapine 2.5 mg/d to treat his schizophrenia. He lives in a psychiatric hospital but was sent to our hospital after being found to have severe oropharyngeal dysphasia on a modified barium swallow study. There was concern for aspiration due to a history of choking episodes, which had been occurring for almost 1 month. During the modified barium swallow study, Mr. N was noted to have aspiration with deep laryngeal penetration during the pharyngeal stages of swallowing to all consistencies; this did not improve with the chin-tuck maneuver. In addition, during a CT scan of the cervical spine, an osteophyte was noted at the C5-C6 level, with possible impingement of the cervical esophagus and decreased upper esophageal sphincter opening.
Due to these findings, Mr. N was sent to our emergency department (ED) for further evaluation. In the ED, his vital signs were stable. He endorsed having a cough after eating, a sensation of having food stuck in his throat, and some hoarseness. His physical examination was notable for poor dentition. Results of a standard laboratory workup were all within normal limits. X-ray was notable for hazy opacities in the right upper to mid lung zones. Mr. N was admitted to the medical unit for further evaluation and management.
Narrowing the diagnosis
Because Mr. N was aspirating both liquids and solids, it was imperative that we identify the cause as soon as possible. The consultations that followed slowly guided the treatment team toward a diagnosis of antipsychotic-induced dysphagia. Otolaryngology identified insensate larynx during a flexible fiberoptic laryngoscopy exam, which was highly suggestive of a neurological dysfunction such as dystonia. Furthermore, an esophagogastroduodenoscopy found no structural abnormalities to explain Mr. N’s dysphagia, which ruled out impingement of the cervical esophagus by the osteophyte. An MRI of the brain ruled out structural abnormalities or evidence of stroke. Finally, a speech and language pathologist confirmed decreased laryngeal closure and airway protection with a repeat modified barium swallow, which led to aspiration during swallowing. Psychiatry recommended starting diphenhydramine to treat Mr. N’s extrapyramidal symptoms (EPS). A 6-day trial was initiated, with a single 50 mg IV dose on the first day followed by 25 mL oral twice daily for the remaining 5 days. In addition, olanzapine was discontinued.
Switching to a different diet and antipsychotic
Two days after starting diphenhydramine, Mr. N was switched to a puree diet. His ability to swallow improved, and he no longer coughed. However, on repeat modified barium swallow, aspiration was still noted for all types of liquids and solids. No structural improvements were seen.
Mr. N was discharged back to his psychiatric hospital, and his antipsychotic was changed from olanzapine to oral aripiprazole 2 mg/d. The aripiprazole dose was kept low to prevent the recurrence of dystonia and because at the time, his schizophrenia was asymptomatic. Mr. N was also prescribed oral diphenhydramine 25 mL twice daily.
At a 2-week follow-up appointment, Mr. N continued to show clinical improvement on the puree diet with thin liquids and continued the prescribed medication regimen.
Dysphagia as a manifestation of EPS
All antipsychotics, and particularly first-generation agents, are associated with EPS.1 These symptoms may be the result of antagonistic binding of dopaminergic D2 receptors within mesolimbic and mesocortical pathways of the brain, as well as parts of basal ganglia such as the caudate nucleus.2
In addition to the examples listed in the Table,2 EPS can present as dysphagia, esophageal dysmotility, or aspiration, none of which may be recognized as EPS. Research has found haloperidol, loxapine, trifluoperazine, olanzapine, risperidone, quetiapine, clozapine, and aripiprazole are associated with dysphagia.3-6 Strategies to treat antipsychotic-induced dysphagia include discontinuing the antipsychotic, lowering the dose, and changing to another medication.7

1. Crouse EL, Alastanos JN, Bozymski KM, et al. Dysphagia with second-generation antipsychotics: a case report and review of the literature. Ment Health Clin. 2018;7(2):56-64. doi:10.9740/mhc.2017.03.056
2. D’Souza RS, Hooten WM. Extrapyramidal symptoms. StatPearls Publishing; 2022. Updated January 8, 2023. Accessed April 28, 2023. https://www.ncbi.nlm.nih.gov/books/NBK534115/
3. Dziewas R, Warnecke T, Schnabel M, et al. Neuroleptic-induced dysphagia: case report and literature review. Dysphagia. 2007;22(1):63-67. doi:10.1007/s00455-006-9032-9
4. Kalf JG, de Swart BJ, Bloem BR, et al. Prevalence of oropharyngeal dysphagia in Parkinson’s disease: a meta-analysis. Parkinsonism Relat Disord. 2012;18(4):311-315. doi:10.1016/j.parkreldis.2011.11.006
5. Lin TW, Lee BS, Liao YC, et al. High dosage of aripiprazole-induced dysphagia. Int J Eat Disord. 2012;45(2):305-306. doi:10.1002/eat.20934
6. Stewart JT. Dysphagia associated with risperidone therapy. Dysphagia. 2003;18(4):274-275. doi:10.1007/s00455-003-0006-x
7. Lee JC, Takeshita J. Antipsychotic-induced dysphagia: a case report. Prim Care Companion CNS Disord. 2015;17(5):10.4088/PCC.15I01792. doi:10.4088/PCC.15I01792
1. Crouse EL, Alastanos JN, Bozymski KM, et al. Dysphagia with second-generation antipsychotics: a case report and review of the literature. Ment Health Clin. 2018;7(2):56-64. doi:10.9740/mhc.2017.03.056
2. D’Souza RS, Hooten WM. Extrapyramidal symptoms. StatPearls Publishing; 2022. Updated January 8, 2023. Accessed April 28, 2023. https://www.ncbi.nlm.nih.gov/books/NBK534115/
3. Dziewas R, Warnecke T, Schnabel M, et al. Neuroleptic-induced dysphagia: case report and literature review. Dysphagia. 2007;22(1):63-67. doi:10.1007/s00455-006-9032-9
4. Kalf JG, de Swart BJ, Bloem BR, et al. Prevalence of oropharyngeal dysphagia in Parkinson’s disease: a meta-analysis. Parkinsonism Relat Disord. 2012;18(4):311-315. doi:10.1016/j.parkreldis.2011.11.006
5. Lin TW, Lee BS, Liao YC, et al. High dosage of aripiprazole-induced dysphagia. Int J Eat Disord. 2012;45(2):305-306. doi:10.1002/eat.20934
6. Stewart JT. Dysphagia associated with risperidone therapy. Dysphagia. 2003;18(4):274-275. doi:10.1007/s00455-003-0006-x
7. Lee JC, Takeshita J. Antipsychotic-induced dysphagia: a case report. Prim Care Companion CNS Disord. 2015;17(5):10.4088/PCC.15I01792. doi:10.4088/PCC.15I01792
More on AI-generated content
In his recent editorial (“A ‘guest editorial’ … generated by ChatGPT?”
Sara Hartley, MD
Berkeley, California
I just read the “guest editorial” generated by ChatGPT. Thank you for this article. Although this is truly an amazing advancement in artificial intelligence (AI), I feel this guest editorial was very basic. It did not read like scientific writing. It read more like it was written at an 11th- or 12th-grade level, though I am fully aware that the question was simple, and thus the answer was not very deep. I can’t deny that if I had been tested, chances are good I would have fallen among the 32% of my peers who would not have recognized it as AI. I appreciate that you (and your team) are working on a protocol regarding how to include content generated by or with the help of AI. God knows if (most likely, when) people with evil minds will use AI to spread false information that may dispute the accredited scientific data and research that guide the medical world and many other fields. I wonder if AI can serve as a search engine that is better or easier to use than PubMed (for example) and the other services we use for research and learning.
Alex Mustachi, PMHNP-BC
Suffern, New York
I wanted to let you know how much I enjoyed reading your recent editorial on AI and scientific writing. Sharing the 4 AI-generated “articles” with readers (“For artificial intelligence, the future is finally here,”
Martha Sajatovic, MD
Cleveland, Ohio
Continue to: The AI-generated samples...
The Al-generated samples were fascinating. As far as I superficially noted, the spelling, grammar, and punctuation were correct. That is better than one gets from most student compositions. However, the articles were completely lacking in depth or apparent insight. The article on anosognosia mentioned it can be present in up to 50% of cases of schizophrenia. In my experience, it is present in approximately 99.9% of cases. It clearly did not consider if anosognosia is also present in alcoholics, codependents, abusers, or people with bizarre political beliefs. But I guess the “intelligence” wasn’t asked that. The other samples also show shallow thinking and repetitive wording—pretty much like my high school junior compositions.
Maybe an appropriate use for AI is a task such as evaluating suicide notes. AI’s success causes one to feel nonplussed. Much more disconcerting was a recent news article that reported AI made up nonexistent references to a professor’s alleged sexual harassment, and then generated citations to its own made-up reference.1 That is indeed frightening new territory. How does one fight against a machine to clear their own name?
Linda Miller, NP
Harrisonburg, Virginia
References
1. Verma P, Oremus W. ChatGPT invented a sexual harassment scandal and named a real law prof as the accused. The Washington Post. April 5, 2023. Accessed May 8, 2023. https://www.washingtonpost.com/technology/2023/04/05/chatgpt-lies/
Thank you, Dr. Nasrallah, for your latest thought-provoking articles on AI. Time and again you provide the profession with cutting-edge, relevant food for thought. Caveat emptor, indeed.
Lawrence E. Cormier, MD
Denver, Colorado
Continue to: We read with interest...
We read with interest Dr. Nasrallah’s editorial that invited readers to share their take on the quality of an AI-generated writing sample. I (MZP) was a computational neuroscience major at Columbia University and was accepted to medical school in 2022 at age 19. I identify with the character traits common among many young tech entrepreneurs driving the AI revolution—social awkwardness; discomfort with subjective emotions; restricted areas of interest; algorithmic thinking; strict, naive idealism; and an obsession with data. To gain a deeper understanding of Sam Altman, the CEO of OpenAI (the company that created ChatGPT), we analyzed a 2.5-hour interview that MIT research scientist Lex Fridman conducted with Altman.1 As a result, we began to discern why AI-generated text feels so stiff and bland compared to the superior fluidity and expressiveness of human communication. As of now, the creation is a reflection of its creator.
Generally speaking, computer scientists are not warm and fuzzy types. Hence, ChatGPT strives to be neutral, accurate, and objective compared to more biased and fallible humans, and, consequently, its language lacks the emotive flair we have come to relish in normal human interactions. In the interview, Altman discusses several solutions that will soon raise the quality of ChatGPT’s currently deficient emotional quotient to approximate its superior IQ. Altruistically, Altman has opened ChatGPT to all, so we can freely interact and utilize its potential to increase our productivity exponentially. As a result, ChatGPT interfaces with millions of humans through RLHF (reinforcement learning from human feedback), which makes each iteration more in tune with our sensibilities.2 Another initiative Altman is undertaking is to depart his Silicon Valley bubble for a road trip to interact with “regular people” and gain a better sense of how to make ChatGPT more user-friendly.1
What’s so saddening about Dr. Nasrallah’s homework assignment is that he is asking us to evaluate with our mature adult standards an article that was written at the emotional stage of a child in early high school. But our hubris and complacency are entirely unfounded because ChatGPT is learning much faster than we ever could, and it will quickly surpass us all as it continues to evolve.
It is also quite disconcerting to hear how Altman is naively relying upon governmental regulation and corporate responsibility to manage the potential misuse of future artificial general intelligence for social, economic, and political control and upheaval. We know well the harmful effects of the internet and social media, particularly on our youth, yet our laws still lag far behind the fact that these technological innovations are simultaneously enhancing our knowledge while destroying our souls. As custodians of our world, dedicated to promoting and preserving mental well-being, we cannot wait much longer to intervene in properly parenting AI along its wisest developmental trajectory before it is too late.
Maxwell Zachary Price, BA
Nutley, New Jersey
Richard Louis Price, MD
New York, New York
References
1. Sam Altman: OpenAI CEO on GPT-4, ChatGPT, and the Future of AI. Lex Fridman Podcast #367. March 25, 2023. Accessed April 5, 2023. https://www.youtube.com/watch?v=L_Guz73e6fw
2. Heikkilä M. How OpenAI is trying to make ChatGPT safer and less biased. MIT Technology Review. Published February 21, 2023. Accessed April 5, 2023. https://www.technologyreview.com/2023/02/21/1068893/how-openai-is-trying-to-make-chatgpt-safer-and-less-biased/
Disclosures
The authors report no financial relationships with any companies whose products are mentioned in their letters, or with manufacturers of competing products.
In his recent editorial (“A ‘guest editorial’ … generated by ChatGPT?”
Sara Hartley, MD
Berkeley, California
I just read the “guest editorial” generated by ChatGPT. Thank you for this article. Although this is truly an amazing advancement in artificial intelligence (AI), I feel this guest editorial was very basic. It did not read like scientific writing. It read more like it was written at an 11th- or 12th-grade level, though I am fully aware that the question was simple, and thus the answer was not very deep. I can’t deny that if I had been tested, chances are good I would have fallen among the 32% of my peers who would not have recognized it as AI. I appreciate that you (and your team) are working on a protocol regarding how to include content generated by or with the help of AI. God knows if (most likely, when) people with evil minds will use AI to spread false information that may dispute the accredited scientific data and research that guide the medical world and many other fields. I wonder if AI can serve as a search engine that is better or easier to use than PubMed (for example) and the other services we use for research and learning.
Alex Mustachi, PMHNP-BC
Suffern, New York
I wanted to let you know how much I enjoyed reading your recent editorial on AI and scientific writing. Sharing the 4 AI-generated “articles” with readers (“For artificial intelligence, the future is finally here,”
Martha Sajatovic, MD
Cleveland, Ohio
Continue to: The AI-generated samples...
The Al-generated samples were fascinating. As far as I superficially noted, the spelling, grammar, and punctuation were correct. That is better than one gets from most student compositions. However, the articles were completely lacking in depth or apparent insight. The article on anosognosia mentioned it can be present in up to 50% of cases of schizophrenia. In my experience, it is present in approximately 99.9% of cases. It clearly did not consider if anosognosia is also present in alcoholics, codependents, abusers, or people with bizarre political beliefs. But I guess the “intelligence” wasn’t asked that. The other samples also show shallow thinking and repetitive wording—pretty much like my high school junior compositions.
Maybe an appropriate use for AI is a task such as evaluating suicide notes. AI’s success causes one to feel nonplussed. Much more disconcerting was a recent news article that reported AI made up nonexistent references to a professor’s alleged sexual harassment, and then generated citations to its own made-up reference.1 That is indeed frightening new territory. How does one fight against a machine to clear their own name?
Linda Miller, NP
Harrisonburg, Virginia
References
1. Verma P, Oremus W. ChatGPT invented a sexual harassment scandal and named a real law prof as the accused. The Washington Post. April 5, 2023. Accessed May 8, 2023. https://www.washingtonpost.com/technology/2023/04/05/chatgpt-lies/
Thank you, Dr. Nasrallah, for your latest thought-provoking articles on AI. Time and again you provide the profession with cutting-edge, relevant food for thought. Caveat emptor, indeed.
Lawrence E. Cormier, MD
Denver, Colorado
Continue to: We read with interest...
We read with interest Dr. Nasrallah’s editorial that invited readers to share their take on the quality of an AI-generated writing sample. I (MZP) was a computational neuroscience major at Columbia University and was accepted to medical school in 2022 at age 19. I identify with the character traits common among many young tech entrepreneurs driving the AI revolution—social awkwardness; discomfort with subjective emotions; restricted areas of interest; algorithmic thinking; strict, naive idealism; and an obsession with data. To gain a deeper understanding of Sam Altman, the CEO of OpenAI (the company that created ChatGPT), we analyzed a 2.5-hour interview that MIT research scientist Lex Fridman conducted with Altman.1 As a result, we began to discern why AI-generated text feels so stiff and bland compared to the superior fluidity and expressiveness of human communication. As of now, the creation is a reflection of its creator.
Generally speaking, computer scientists are not warm and fuzzy types. Hence, ChatGPT strives to be neutral, accurate, and objective compared to more biased and fallible humans, and, consequently, its language lacks the emotive flair we have come to relish in normal human interactions. In the interview, Altman discusses several solutions that will soon raise the quality of ChatGPT’s currently deficient emotional quotient to approximate its superior IQ. Altruistically, Altman has opened ChatGPT to all, so we can freely interact and utilize its potential to increase our productivity exponentially. As a result, ChatGPT interfaces with millions of humans through RLHF (reinforcement learning from human feedback), which makes each iteration more in tune with our sensibilities.2 Another initiative Altman is undertaking is to depart his Silicon Valley bubble for a road trip to interact with “regular people” and gain a better sense of how to make ChatGPT more user-friendly.1
What’s so saddening about Dr. Nasrallah’s homework assignment is that he is asking us to evaluate with our mature adult standards an article that was written at the emotional stage of a child in early high school. But our hubris and complacency are entirely unfounded because ChatGPT is learning much faster than we ever could, and it will quickly surpass us all as it continues to evolve.
It is also quite disconcerting to hear how Altman is naively relying upon governmental regulation and corporate responsibility to manage the potential misuse of future artificial general intelligence for social, economic, and political control and upheaval. We know well the harmful effects of the internet and social media, particularly on our youth, yet our laws still lag far behind the fact that these technological innovations are simultaneously enhancing our knowledge while destroying our souls. As custodians of our world, dedicated to promoting and preserving mental well-being, we cannot wait much longer to intervene in properly parenting AI along its wisest developmental trajectory before it is too late.
Maxwell Zachary Price, BA
Nutley, New Jersey
Richard Louis Price, MD
New York, New York
References
1. Sam Altman: OpenAI CEO on GPT-4, ChatGPT, and the Future of AI. Lex Fridman Podcast #367. March 25, 2023. Accessed April 5, 2023. https://www.youtube.com/watch?v=L_Guz73e6fw
2. Heikkilä M. How OpenAI is trying to make ChatGPT safer and less biased. MIT Technology Review. Published February 21, 2023. Accessed April 5, 2023. https://www.technologyreview.com/2023/02/21/1068893/how-openai-is-trying-to-make-chatgpt-safer-and-less-biased/
Disclosures
The authors report no financial relationships with any companies whose products are mentioned in their letters, or with manufacturers of competing products.
In his recent editorial (“A ‘guest editorial’ … generated by ChatGPT?”
Sara Hartley, MD
Berkeley, California
I just read the “guest editorial” generated by ChatGPT. Thank you for this article. Although this is truly an amazing advancement in artificial intelligence (AI), I feel this guest editorial was very basic. It did not read like scientific writing. It read more like it was written at an 11th- or 12th-grade level, though I am fully aware that the question was simple, and thus the answer was not very deep. I can’t deny that if I had been tested, chances are good I would have fallen among the 32% of my peers who would not have recognized it as AI. I appreciate that you (and your team) are working on a protocol regarding how to include content generated by or with the help of AI. God knows if (most likely, when) people with evil minds will use AI to spread false information that may dispute the accredited scientific data and research that guide the medical world and many other fields. I wonder if AI can serve as a search engine that is better or easier to use than PubMed (for example) and the other services we use for research and learning.
Alex Mustachi, PMHNP-BC
Suffern, New York
I wanted to let you know how much I enjoyed reading your recent editorial on AI and scientific writing. Sharing the 4 AI-generated “articles” with readers (“For artificial intelligence, the future is finally here,”
Martha Sajatovic, MD
Cleveland, Ohio
Continue to: The AI-generated samples...
The Al-generated samples were fascinating. As far as I superficially noted, the spelling, grammar, and punctuation were correct. That is better than one gets from most student compositions. However, the articles were completely lacking in depth or apparent insight. The article on anosognosia mentioned it can be present in up to 50% of cases of schizophrenia. In my experience, it is present in approximately 99.9% of cases. It clearly did not consider if anosognosia is also present in alcoholics, codependents, abusers, or people with bizarre political beliefs. But I guess the “intelligence” wasn’t asked that. The other samples also show shallow thinking and repetitive wording—pretty much like my high school junior compositions.
Maybe an appropriate use for AI is a task such as evaluating suicide notes. AI’s success causes one to feel nonplussed. Much more disconcerting was a recent news article that reported AI made up nonexistent references to a professor’s alleged sexual harassment, and then generated citations to its own made-up reference.1 That is indeed frightening new territory. How does one fight against a machine to clear their own name?
Linda Miller, NP
Harrisonburg, Virginia
References
1. Verma P, Oremus W. ChatGPT invented a sexual harassment scandal and named a real law prof as the accused. The Washington Post. April 5, 2023. Accessed May 8, 2023. https://www.washingtonpost.com/technology/2023/04/05/chatgpt-lies/
Thank you, Dr. Nasrallah, for your latest thought-provoking articles on AI. Time and again you provide the profession with cutting-edge, relevant food for thought. Caveat emptor, indeed.
Lawrence E. Cormier, MD
Denver, Colorado
Continue to: We read with interest...
We read with interest Dr. Nasrallah’s editorial that invited readers to share their take on the quality of an AI-generated writing sample. I (MZP) was a computational neuroscience major at Columbia University and was accepted to medical school in 2022 at age 19. I identify with the character traits common among many young tech entrepreneurs driving the AI revolution—social awkwardness; discomfort with subjective emotions; restricted areas of interest; algorithmic thinking; strict, naive idealism; and an obsession with data. To gain a deeper understanding of Sam Altman, the CEO of OpenAI (the company that created ChatGPT), we analyzed a 2.5-hour interview that MIT research scientist Lex Fridman conducted with Altman.1 As a result, we began to discern why AI-generated text feels so stiff and bland compared to the superior fluidity and expressiveness of human communication. As of now, the creation is a reflection of its creator.
Generally speaking, computer scientists are not warm and fuzzy types. Hence, ChatGPT strives to be neutral, accurate, and objective compared to more biased and fallible humans, and, consequently, its language lacks the emotive flair we have come to relish in normal human interactions. In the interview, Altman discusses several solutions that will soon raise the quality of ChatGPT’s currently deficient emotional quotient to approximate its superior IQ. Altruistically, Altman has opened ChatGPT to all, so we can freely interact and utilize its potential to increase our productivity exponentially. As a result, ChatGPT interfaces with millions of humans through RLHF (reinforcement learning from human feedback), which makes each iteration more in tune with our sensibilities.2 Another initiative Altman is undertaking is to depart his Silicon Valley bubble for a road trip to interact with “regular people” and gain a better sense of how to make ChatGPT more user-friendly.1
What’s so saddening about Dr. Nasrallah’s homework assignment is that he is asking us to evaluate with our mature adult standards an article that was written at the emotional stage of a child in early high school. But our hubris and complacency are entirely unfounded because ChatGPT is learning much faster than we ever could, and it will quickly surpass us all as it continues to evolve.
It is also quite disconcerting to hear how Altman is naively relying upon governmental regulation and corporate responsibility to manage the potential misuse of future artificial general intelligence for social, economic, and political control and upheaval. We know well the harmful effects of the internet and social media, particularly on our youth, yet our laws still lag far behind the fact that these technological innovations are simultaneously enhancing our knowledge while destroying our souls. As custodians of our world, dedicated to promoting and preserving mental well-being, we cannot wait much longer to intervene in properly parenting AI along its wisest developmental trajectory before it is too late.
Maxwell Zachary Price, BA
Nutley, New Jersey
Richard Louis Price, MD
New York, New York
References
1. Sam Altman: OpenAI CEO on GPT-4, ChatGPT, and the Future of AI. Lex Fridman Podcast #367. March 25, 2023. Accessed April 5, 2023. https://www.youtube.com/watch?v=L_Guz73e6fw
2. Heikkilä M. How OpenAI is trying to make ChatGPT safer and less biased. MIT Technology Review. Published February 21, 2023. Accessed April 5, 2023. https://www.technologyreview.com/2023/02/21/1068893/how-openai-is-trying-to-make-chatgpt-safer-and-less-biased/
Disclosures
The authors report no financial relationships with any companies whose products are mentioned in their letters, or with manufacturers of competing products.
Interventional psychiatry (Part 1)
Advances in the understanding of neurobiological and neuropsychiatric pathophysiology have opened new avenues of treatment for psychiatric patients. Historically, with a few exceptions, most psychiatric medications have been administered orally. However, many of the newer treatments require some form of specialized administration because they cannot be taken orally due to their chemical property (such as aducanumab); because there is the need to produce stable blood levels of the medication (such as brexanolone); because oral administration greatly diminished efficacy (such as oral vs IV magnesium or scopolamine), or because the treatment is focused on specific brain structures. This need for specialized administration has created a subspecialty called interventional psychiatry.
Part 1 of this 2-part article provides an overview of 1 type of interventional psychiatry: parenterally administered medications, including those administered via IV. We also describe 3 other interventional approaches to treatment: stellate ganglion blocks, glabellar botulinum toxin (BT) injections, and trigger point injections. In Part 2 we will review interventional approaches that involve neuromodulation.
Parenteral medications in psychiatry
In general, IV and IM medications can be more potent that oral medications due to their overall faster onset of action and higher blood concentrations. These more invasive forms of administration can have significant limitations, such as a risk of infection at the injection site, the need to be administered in a medical setting, additional time, and patient discomfort.

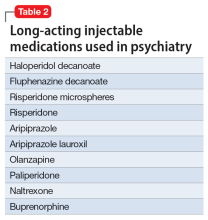
Table 1 lists short-acting injectable medications used in psychiatry, and Table 2 lists long-acting injectable medications. Parenteral administration of antipsychotics is performed to alleviate acute agitation or for chronic symptom control. These medications generally are not considered interventional treatments, but could be classified as such due to their invasive nature.1 Furthermore, inhalable loxapine—which is indicated for managing acute agitation—requires a Risk Evaluation and Mitigation Strategy program consisting of 2 hours observation and monitoring of respiratory status.2,3 Other indications for parenteral treatments include IM naltrexone extended release4 and subcutaneous injections of buprenorphine extended release5 and risperidone.6
IV administration
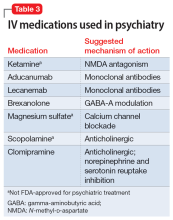
Most IV treatments described in this article are not FDA-approved for psychiatric treatment. Despite this, many interventional psychiatric treatments are part of clinical practice. IV infusion of ketamine is the most widely known and most researched of these. Table 3 lists other IV treatments that could be used as psychiatric treatment.
Ketamine
Since the early 1960s, ketamine has been used as a surgical anesthetic for animals. In the United States, it was approved for human surgical anesthesia in 1970. It was widely used during the Vietnam War due to its lack of inhibition of respiratory drive; medical staff first noticed an improvement in depressive symptoms and the resolution of suicidal ideation in patients who received ketamine. This led to further research on ketamine, particularly to determine its application in treatment-resistant depression (TRD) and other conditions.7 IV ketamine administration is most widely researched, but IM injections, intranasal sprays, and lozenges are also available. The dissociative properties of ketamine have led to its recreational use.8
Hypotheses for the mechanism of action of ketamine as an antidepressant include direct synaptic or extrasynaptic (GluN2B-selective), N-methyl-
Continue to: Ketamine is a schedule...
Ketamine is a schedule III medication with addictive properties. Delirium, panic attacks, hallucinations, nightmares, dysphoria, and paranoia may occur during and after use.13 Premedication with benzodiazepines, most notably lorazepam, is occasionally used to minimize ketamine’s adverse effects, but this generally is not recommended because doing so reduces ketamine’s antidepressant effects.14 Driving and operating heavy machinery is contraindicated after IV infusion. The usual protocol involves an IV infusion of ketamine 0.4 mg/kg to 1 mg/kg dosing over 1 hour. Doses between 0.4 mg/kg and 0.6 mg/kg are most common. Ketamine has a therapeutic window; doses >0.5 mg/kg are progressively less effective.15 Unlike the recommendation after esketamine administration, after receiving ketamine, patients remain in the care of their treatment team for <2 hours.
Esketamine, the S enantiomer of ketamine, was FDA-approved for TRD as an intranasal formulation. Esketamine is more commonly used than IV ketamine because it is FDA-approved and typically covered by insurance, but it may not be as effective.16 An economic analysis by Brendle et al17 suggested insurance companies would lower costs if they covered ketamine infusions vs intranasal esketamine.
Aducanumab and lecanemab
The most recent FDA-approved interventional agents are aducanumab and lecanemab, which are indicated for treating Alzheimer disease.18,19 Both are human monoclonal antibodies that bind selectively and with high affinity to amyloid beta plaque aggregates and promote their removal by Fc receptor–mediated phagocytosis.20
FDA approval of aducanumab and lecanemab was controversial. Initially, aducanumab’s safety monitoring board performed a futility analysis that suggested aducanumab was unlikely to separate from placebo, and the research was stopped.21 The manufacturer petitioned the FDA to consider the medication for accelerated approval on the basis of biomarker data showing that amyloid beta plaque aggregates become smaller. Current FDA approval is temporary to allow patients access to this potentially beneficial agent, but the manufacturer must supply clinical evidence that the reduction of amyloid beta plaques is associated with desirable changes in the course of Alzheimer disease, or risk losing the approval.
Lecanemab is also a human monoclonal antibody intended to remove amyloid beta plaques that was FDA-approved under the accelerated approval pathway.22 Unlike aducanumab, lecanemab demonstrated a statistically significant (although clinically imperceptible) reduction in the rate of cognitive decline; it did not show cognitive improvement.23 Lecanemab also significantly reduced amyloid beta plaques.23
Continue to: Aducanumab and lecanemab are generally...
Aducanumab and lecanemab are generally not covered by insurance and typically cost >$26,000 annually. Both are administered by IV infusion once a month. More monoclonal antibody medications for treating early Alzheimer disease are in the late stages of development, most notably donanebab.24 Observations during clinical trials found that in the later stages of Alzheimer disease, forceful removal of plaques by the autoimmune process damages neurons, while in less dense deposits of early dementia such removal is not harmful to the cells and prevents amyloid buildup.
Brexanolone is an aqueous formulation of allopregnanolone, a major metabolite of progesterone and a positive allosteric modulator of GABA-A receptors.25 Its levels are maximal at the end of the third trimester of pregnancy and fall rapidly following delivery. Research showed a 3-day infusion was rapidly and significantly effective for treating postpartum depression26 and brexanolone received FDA approval for this indication in March 2019.27 However, various administrative, economic, insurance, and other hurdles make it difficult for patients to access this treatment. Despite its rapid onset of action (usually 48 hours), brexanolone takes an average of 15 days to go through the prior authorization process.28 In addition to the need for prior authorization, the main impediment to the use of brexanolone is the 3-day infusion schedule, which greatly magnifies the cost but is partially circumvented by the availability of dedicated outpatient centers.
Magnesium
Magnesium is on the World Health Organization’s Model List of Essential Medicines.29 There has been extensive research on the use of magnesium sulfate for psychiatric indications, especially for depression.30 Magnesium functions similarly to calcium channel blockers by competitively blocking intracellular calcium channels, decreasing calcium availability, and inhibiting smooth muscle contractility.31 It also competes with calcium at the motor end plate, reducing excitation by inhibiting the release of acetylcholine.32 This property is used for high-dose IV magnesium treatment of impending preterm labor in obstetrics. Magnesium sulfate is the drug of choice in treating eclamptic seizures and preventing seizures in severe preeclampsia or gestational hypertension with severe features.33 It is also used to treat torsade de pointes, severe asthma exacerbations, constipation, and barium poisoning.34 Beneficial use in asthma treatment35 and the treatment of migraine36 have also been reported.
IV magnesium in myocardial infarction may be harmful,37 though outside of acute cardiac events, magnesium is found to be safe.38
Oral magnesium sulfate is a common over-the-counter anxiety remedy. As a general cell stabilizer (mediated by the reduction of intracellular calcium), magnesium is potentially beneficial outside of its muscle-relaxing properties, although muscle relaxing can benefit anxious patients. It is used to treat anxiety,39 alcohol withdrawal,40 and fear.41 Low magnesium blood levels are found in patients with depression, schizophrenia,42 and attention-deficit/hyperactivity disorder.43 However, it is important to note that the therapeutic effect of magnesium when treating anxiety and headache is independent of preinfusion magnesium blood levels.43
Continue to: The efficacy of oral magnesium...
The efficacy of oral magnesium is not robust. However, IV administration has a pronounced beneficial effect as an abortive and preventative treatment in many patients with anxiety.44
IV administration of magnesium can produce adverse effects, including flushing, sweating, hypotension, depressed reflexes, flaccid paralysis, hypothermia, circulatory collapse, and cardiac and CNS depression. These complications are very rare and dose-dependent.45 Magnesium is excreted by the kidneys, and dosing must be decreased in patients with kidney failure. The most common adverse effect is local burning along the vein upon infusion; small doses of IV lidocaine can remedy this. Hot flashes are also common.45
Various dosing strategies are available. In patients with anxiety, application dosing is based on the recommended preeclampsia IV dose of 4 g diluted in 250 mL of 5% dextrose. Much higher doses may be used in obstetrics. Unlike in obstetrics, for psychiatric indications, magnesium is administered over 60 to 90 minutes. Heart monitoring is recommended.
Scopolamine
Scopolamine is primarily used to relieve nausea, vomiting, and dizziness associated with motion sickness and recovery from anesthesia. It is also used in ophthalmology and in patients with excessive sweating. In off-label and experimental applications, scopolamine has been used in patients with TRD.46
Scopolamine is an anticholinergic medication. It is a nonselective antagonist at muscarinic receptors.47 Tricyclic antidepressants (TCAs) possess strong anticholinergic function. Newer generations of antidepressants were designed specifically not to have this function because it was believed to be an unwanted and potentially dangerous adverse effect. However, data suggest that anticholinergic action is important in decreasing depressive symptoms. Several hypotheses of anticholinergic effects on depression have been published since the 1970s. They include the cholinergic-adrenergic hypothesis,48 acetylcholine predominance relative to adrenergic action hypothesis,49 and insecticide poisoning observations.50 Centrally acting physostigmine (but not peripherally acting neostigmine) was reported to control mania.48,51 A genetic connection between the M2acetylcholine receptor in patients with major depressive disorder (MDD) and alcohol use disorder is also suggestive.52
Continue to: Multiple animal studies show...
Multiple animal studies show direct improvement in mobility and a decrease in despair upon introducing anticholinergic substances.53-55 The cholinergic theory of depression has been studied in several controlled clinical human studies.56,57 Use of a short-acting anticholinergic glycopyrrolate during electroconvulsive therapy (ECT) may contribute to the procedure’s efficacy.
Human research shows scopolamine has a higher efficacy as an antidepressant and anti-anxiety medication in women than in men,58 possibly because estrogen increases the activity of choline acetyltransferase and release of acetylcholine.59,60 M2receptors mediate estrogen influence on the NMDAR, which may explain the anticholinergic effects of depression treatments in women.61
Another proposed mechanism of action of scopolamine is a potent inhibition of the NMDAR.62 Rapid treatments of depression may be based on this mechanism. Examples of such treatments include IV ketamine and sleep deprivation.63 IV scopolamine shows potency in treating MDD and bipolar depression. This treatment should be reserved for patients who do not respond to or are not candidates for other usual treatment modalities of MDD and for the most severe cases. Scopolamine is 30 times more potent than amitriptyline in anticholinergic effect and reportedly produces sustained improvement in MDD.64
Scopolamine has no black-box warnings. It has not been studied in pregnant women and is not recommended for use during pregnancy. Due to possible negative cardiovascular effects, a normal electrocardiogram is required before the start of treatment. Exercise caution in patients with glaucoma, benign prostatic enlargement, gastroparesis, unstable cardiovascular status, or severe renal impairment.
Treatment with scopolamine is not indicated for patients with myasthenia gravis, psychosis, or seizures. Patients must be off potassium for 3 days before beginning scopolamine treatment. Patients should consult with their cardiologist before having a scopolamine infusion. Adverse reactions may include psychosis, tachycardia, seizures, paralytic ileus, and glaucoma exacerbation. The most common adverse effects of scopolamine infusion treatment include drowsiness, dry mouth, blurred vision, lightheadedness, and dizziness. Due to possible drowsiness, operating motor vehicles or heavy machinery must be avoided on the day of treatment.65 Overall, the adverse effects of scopolamine are preventable and manageable, and its antidepressant efficacy is noteworthy.66
Continue to: Treatment typically consists of 3 consecutive infusions...
Treatment typically consists of 3 consecutive infusions of 4 mcg/kg separated by 3 to 5 days.56 It is possible to have a longer treatment course if the patient experiences only partial improvement. Repeated courses or maintenance treatment (similar to ECT maintenance) are utilized in some patients if indicated. Cardiac monitoring is mandatory.
Clomipramine
Clomipramine, a TCA, acts as a preferential inhibitor of 5-hydroxytryptamine uptake and has proven effective in managing depression, TRD, and obsessive-compulsive disorder (OCD).67 Although this medication has reported treatment benefits for patients with phobia, panic disorder,15 chronic pain,68 Tourette syndrome,69 premature ejaculation, anorexia nervosa,70 cataplexy,49 and enuresis,71 it is FDA-approved only for the treatment of OCD.72 Clomipramine may also be beneficial for pain and headache, possibly because of its anti-inflammatory action.73 The anticholinergic effects of clomipramine may add to its efficacy in depression.
The pathophysiology of MDD is connected to hyperactivity of the HPA axis and elevated cortisol levels. Higher clomipramine plasma levels show a linear correlation with lower cortisol secretion and levels, possibly aiding in the treatment of MDD and anxiety.74 The higher the blood level, the more pronounced clomipramine’s therapeutic effect across multiple domains.75
IV infusion of clomipramine produces the highest concentration in the shortest time, but overall, research does not necessarily support increased efficacy of IV over oral administration. There is evidence suggesting that subgroups of patients with severe, treatment-refractory OCD may benefit from IV agents and research suggests a faster onset of action.76 Faster onset of symptom relief is the basis for IV clomipramine use. In patients with OCD, it can take several months for oral medications to produce therapeutic benefits; not all patients can tolerate this. In such scenarios, IV administration may be considered, though it is not appropriate for routine use until more research is available. Patients with treatment-resistant OCD who have exhausted other means of symptom relief may also be candidates for IV treatment.
The adverse effects of IV clomipramine are no different from oral use, though they may be more pronounced. A pretreatment cardiac exam is desirable because clomipramine, like other TCAs, may be cardiotoxic. The anticholinergic adverse effects of TCAs are well known to clinicians77 and partially explained in the scopolamine section of this article. It is not advisable to combine clomipramine with other TCAs or serotonin reuptake inhibitors. Clomipramine also should not be combined with monoamine oxidase inhibitors, though such a combination was reported in medical literature.78 Combination with antiarrhythmics such as lidocaine or opioids such as fentanyl or and tramadol is highly discouraged (fentanyl and tramadol also have serotonergic effects).79
Continue to: Clomipramine for IV use is not commercially available...
Clomipramine for IV use is not commercially available and must be sterilely compounded. The usual course of treatment is a series of 3 infusions: 150 mg on Day 1, 200 mg on Day 2 or Day 3, and 250 mg on Day 3, Day 4, or Day 5, depending on tolerability. A protocol with a 50 mg/d starting dose and titration up to a maximum dose of 225 mg/d over 5 to 7 days has been suggested for inpatient settings.67 Titration to 250 mg is more common.80
A longer series may be performed, but this increases the likelihood of adverse effects. Booster and maintenance treatments are also completed when required. Cardiac monitoring is mandatory.
Vortioxetine and citalopram
IV treatment of depression with
Injections and blocks
Three interventional approaches to treatment that possess psychotherapeutic potential include stellate ganglion blocks (SGBs), glabellar BT injections, and trigger point injections (TPIs). None of these are FDA-approved for psychiatric treatment.
Stellate ganglion blocks
The sympathetic nervous system is involved in autonomic hyperarousal and is at the core of posttraumatic symptomatology.83 Insomnia, anxiety, irritability, hypervigilance, and other excitatory CNS events are connected to the sympathetic nervous system and amygdala activation is commonly observed in those exposed to extreme stress or traumatic events.84
Continue to: SGBs were first performed 100 years ago...
SGBs were first performed 100 years ago and reported to have beneficial psychiatric effects at the end of the 1940s. In 1998 in Finland, improvement of posttraumatic stress disorder (PTSD) symptoms was observed accidentally via thoracic level spine blocks.85 In 2006, cervical level sympathetic blocks were shown to be effective for PTSD symptom control.86 By the end of 2010, Veterans Administration hospitals adopted SGBs to treat veterans with PTSD.87,88 The first multisite, randomized clinical trial of
Since the stellate ganglion is connected to the amygdala, SGB has also been assessed for treating anxiety and depression.89,90 Outside of PTSD, SGBs are used to treat complex regional pain syndrome,91 phantom limb pain, trigeminal neuralgia,92 intractable angina,93 and postherpetic neuralgia in the head, neck, upper chest, or arms.94 The procedure consists of an injection of a local anesthetic through a 25-gauge needle into the stellate sympathetic ganglion at the C6 or C7 vertebral levels. An injection into C6 is considered safer because of specific cervical spine anatomy. Ideally, fluoroscopic guidance or ultrasound is used to guide needle insertion.95
A severe drop in blood pressure may be associated with SGBs and is mitigated by IV hydration. Other adverse effects include red eyes, drooping of the eyelids, nasal congestion, hoarseness, difficulty swallowing, a sensation of a “lump” in the throat, and a sensation of warmth or tingling in the arm or hand. Bilateral SGB is contraindicated due to the danger of respiratory arrest.96
Glabellar BT injections
OnabotulinumtoxinA (BT) injection was first approved for therapeutic use in 1989 for eye muscle disorders such as strabismus97 and blepharospasm.98 It was later approved for several other indications, including cosmetic use, hyperhidrosis, migraine prevention, neurogenic bladder disorder, overactive bladder, urinary incontinence, and spasticity.99-104 BT is used off-label for achalasia and sialorrhea.105,106 Its mechanism of action is primarily attributed to muscle paralysis by blocking presynaptic acetylcholine release into neuromuscular junctions.107
Facial BT injections are usually administered for cosmetic purposes, but smoothing forehead wrinkles and frown lines (the glabellar region of the face) both have antidepressant effects.108 BT injections into the glabellar region also demonstrate antidepressant effects, particularly in patients with comorbid migraines and MDD.109 Early case observations supported the independent benefit of the toxin on MDD when the toxin was injected into the glabellar region.110,111 The most frequent protocol involves injections in the procerus and corrugator muscles.
Continue to: The facial feedback/emotional proprioception hypothesis...
The facial feedback/emotional proprioception hypothesis has dominated thinking about the mood-improving effects of BT. The theory is that blocking muscular expression of sadness (especially in the face) interrupts the experience of sadness; therefore, depression subsides.112,113 However, BT injections in the muscles involved in the smile and an expression of positive emotions (lateral part of the musculus orbicularis oculi) have been associated with increased MDD scores.114 Thus, the mechanism clearly involves more than the cosmetic effect, since facial muscle injections in rats also have antidepressant effects.115
The use of progressive muscle relaxation is well-established in psychiatric treatment. The investigated conditions of increased muscle tone, especially torticollis and blepharospasm, are associated with MDD, and it may be speculated that proprioceptive feedback from the affected muscles may be causally involved in this association.116-118 Activity of the corrugator muscle has been positively associated with increased amygdala activity.119 This suggests a potential similar mechanism to that hypothesized for SGB.
Alternatively, BT is commonly used to treat chronic conditions that may contribute to depression; its success in relieving the underlying problem may indirectly relieve MDD. Thus, in a postmarketing safety evaluation of BT, MDD was demonstrated 40% to 88% less often by patients treated with BT for 6 of the 8 conditions and injection sites, such as in spasms and spasticity of arms and legs, torticollis and neck pain, and axilla and palm injections for hyperhidrosis. In a parotid and submandibular glands BT injection subcohort, no patients experienced depressive symptoms.120
Medicinal BT is generally considered safe. The most common adverse effects are hypersensitivity, injection site reactions, and other adverse effects specific to the injection site.121 Additionally, the cosmetic effects are transient, given the nature of the medication.
Trigger point injections
TPIs in the neck and shoulders are frequently used to treat tension headaches and various referred pain locations in the face and arms. Tension and depression frequently overlap in clinical practice.122 Relieving muscle tension (with resulting trigger points) improves muscle function and mood.
Continue to: The higher the number of active...
The higher the number of active trigger points (TPs), the greater the physical burden of headache and the higher the anxiety level. Gender differences in TP prevalence and TPI efficacy have been described in the literature. For example, the number of active TPs seems directly associated with anxiety levels in women but not in men.123 Although TPs appear to be more closely associated with anxiety than depression,124 depression associated with muscle tension does improve with TPIs. European studies have demonstrated a decrease in scores on the Hamilton Depression Rating Scale and Hamilton Anxiety Rating Scale following TPI treatment.125 The effect may be indirect, as when a patient’s pain is relieved, sleep and other psychiatric symptoms improve.126
A randomized controlled trial by Castro Sánchez et al127 demonstrated that dry needling therapy in patients with fibromyalgia syndrome (FMS) showed improvements in pain pressure thresholds, body pain, vitality, and social function, as well as total FMS symptoms, quality of sleep, anxiety, hospital anxiety and depression, general pain intensity, and fatigue.
Myofascial pain syndrome, catastrophizing, and muscle tension are common in patients with depression, anxiety, and somatization. Local TPI therapy aimed at inactivating pain generators is supported by moderate quality evidence. All manner of therapies have been described, including injection of saline, corticosteroids, local anesthetic agents, and dry needling. BT injections in chronic TPs are also practiced, though no specific injection therapy has been reliably shown to be more advantageous than another. The benefits of TPIs may be derived from the needle itself rather than from any specific substance injected. Stimulation of a local twitch response with direct needling of the TP appears of importance. There is no established consensus regarding the number of injection points, frequency of administration, and volume or type of injectate.128
Adverse effects of TPIs relate to the nature of the invasive therapy, with the risk of tissue damage and bleeding. Pneumothorax risk is present with needle insertion at the neck and thorax.129 Patients with diabetes may experience variations in blood sugar control if steroids are used.
Bottom Line
Interventional treatment modalities that may have a role in psychiatric treatment include IV administration of ketamine, aducanumab, lecanemab, brexanolone, magnesium, scopolamine, and clomipramine. Other interventional approaches include stellate ganglion blocks, glabellar botulinum toxin injections, and trigger point injections.
Related Resources
- Dokucu ME, Janicak PG. Nontraditional therapies for treatment-resistant depression. Current Psychiatry. 2021; 20(9):38-43,49. doi:10.12788/cp.0166
- Kim J, Khoury R, Grossberg GT. Botulinum toxin: emerging psychiatric indications. Current Psychiatry. 2018;17(12):8-18.
Drug Brand Names
Aducanumab • Aduhelm
Aripiprazole • Abilify
Aripiprazole lauroxil • Aristada
Brexanolone • Zulresso
Buprenorphine • Sublocade
Citalopram • Celexa
Clomipramine • Anafranil
Diazepam • Valium
Droperidol • Inapsine
Esketamine • Spravato
Fentanyl • Actiq
Fluphenazine decanoate • Modecate
Fluphenazine hydrochloride • Prolixin
Haloperidol decanoate • Haldol decanoate
Haloperidol lactate • Haldol
Ketamine • Ketalar
Lecanemab • Leqembi
Lidocaine • Xylocaine
Lorazepam • Ativan
Loxapine inhaled • Adasuve
Naltrexone • Vivitrol
Magnesium sulfate • Sulfamag
Midazolam • Versed
Olanzapine • Zyprexa
OnabotulinumtoxinA injection • Botox
Paliperidone • Invega Hafyera, Invega Sustenna, Invega Trinza
Rapamycin • Rapamune, Sirolimus
Risperidone • Perseris
Risperidone microspheres • Risperdal Consta, Rykindo
Scopolamine • Hyoscine
Tramadol • Conzip
Vortioxetine • Trintellix
Ziprasidone • Geodon
1. Vincent KM, Ryan M, Palmer E, et al. Interventional psychiatry. Postgrad Med. 2020;132(7):573-574.
2. Allen MH, Feifel D, Lesem MD, et al. Efficacy and safety of loxapine for inhalation in the treatment of agitation in patients with schizophrenia: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2011;72(10):1313-1321.
3. Kwentus J, Riesenberg RA, Marandi M, et al. Rapid acute treatment of agitation in patients with bipolar I disorder: a multicenter, randomized, placebo-controlled clinical trial with inhaled loxapine. Bipolar Disord. 2012;14(1):31-40.
4. Lee JD, Nunes EV Jr, Novo P, et al. Comparative effectiveness of extended-release naltrexone versus buprenorphine-naloxone for opioid relapse prevention (X:BOT): a multicentre, open-label, randomised controlled trial. Lancet. 2018;391(10118):309-318.
5. Haight BR, Learned SM, Laffont CM, et al. Efficacy and safety of a monthly buprenorphine depot injection for opioid use disorder: a multicentre, randomised, double‐blind, placebo‐controlled, phase 3 trial. Lancet. 2019;393(10173):778-790.
6. Andorn A, Graham J, Csernansky J, et al. Monthly extended-release risperidone (RBP-7000) in the treatment of schizophrenia: results from the phase 3 program. J Clin Psychopharmacol. 2019;39(5):428-433.
7. Dundee TW. Twenty-five years of ketamine. A report of an international meeting. Anaesthesia. 1990;45(2):159. doi:10.1111/j.1365-2044.1990.tb14287.x
8. White PF, Way WL, Trevor AJ. Ketamine--its pharmacology and therapeutic uses. Anesthesiology. 1982;56(2):119-136. doi:10.1097/00000542-198202000-00007
9. Zanos P, Gould TD. Mechanisms of ketamine action as an antidepressant. Mol Psychiatry. 2018;23(4):801-811.
10. Molero P, Ramos-Quiroga JA, Martin-Santos R, et al. Antidepressant efficacy and tolerability of ketamine and esketamine: a critical review. CNS Drugs. 2018;32(5):411-420. doi:10.1007/s40263-018-0519-3
11. Williams NR, Heifets BD, Blasey C, et al. Attenuation of antidepressant effects of ketamine by opioid receptor antagonism. Am J Psychiatry. 2018;175(12):1205-1215.
12. Witkin JM, Martin AE, Golani LK, et al. Rapid-acting antidepressants. Adv Pharmacol. 2019;86:47-96.
13. Strayer RJ, Nelson LS. Adverse events associated with ketamine for procedural sedation in adults. Am J Emerg Med. 2008;26(9):985-1028. doi:10.1016/j.ajem.2007.12.005
14. Frye MA, Blier P, Tye SJ. Concomitant benzodiazepine use attenuates ketamine response: implications for large scale study design and clinical development. J Clin Psychopharmacol. 2015;35(3):334-336.
15. Fava M, Freeman MP, Flynn M, et al. Double-blind, placebo-controlled, dose-ranging trial of intravenous ketamine as adjunctive therapy in treatment-resistant depression (TRD). Mol Psychiatry. 2020;25(7):1592-1603.
16. Bahji A, Vazquez GH, Zarate CA Jr. Comparative efficacy of racemic ketamine and esketamine for depression: a systematic review and meta-analysis. J Affect Disord. 2021;278:542-555. Erratum in: J Affect Disord. 2021;281:1001.
17. Brendle M, Robison R, Malone DC. Cost-effectiveness of esketamine nasal spray compared to intravenous ketamine for patients with treatment-resistant depression in the US utilizing clinical trial efficacy and real-world effectiveness estimates. J Affect Disord. 2022;319:388-396.
18. Dhillon S. Aducanumab: first approval. Drugs. 2021;81(12):1437-1443. Erratum in: Drugs. 2021;81(14):1701.
19. van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388(1):9-21. doi:10.1056/NEJMoa2212948
20. Sevigny J, Chiao P, Bussière T, et al. The antibody aducanumab reduces Aβ plaques in Alzheimer’s disease. Nature. 2016;537(7618):50-56. Update in: Nature. 2017;546(7659):564.
21. Fillit H, Green A. Aducanumab and the FDA – where are we now? Nat Rev Neurol. 2021;17(3):129-130.
22. Reardon S. FDA approves Alzheimer’s drug lecanemab amid safety concerns. Nature. 2023;613(7943):227-228. doi:10.1038/d41586-023-00030-3
23. McDade E, Cummings JL, Dhadda S, et al. Lecanemab in patients with early Alzheimer’s disease: detailed results on biomarker, cognitive, and clinical effects from the randomized and open-label extension of the phase 2 proof-of-concept study. Alzheimers Res Ther. 2022;14(1):191. doi:10.1186/s13195-022-01124-2
24. Mintun MA, Lo AC, Evans CD, et al. Donanemab in early Alzheimer’s disease. N Engl J Med. 2021;384(18):1691-1704.
25. Luisi S, Petraglia F, Benedetto C, et al. Serum allopregnanolone levels in pregnant women: changes during pregnancy, at delivery, and in hypertensive patients. J Clin Endocrinol Metab. 2000;85(7):2429-2433.
26. Meltzer-Brody S, Colquhoun H, Riesenberg R, et al. Brexanolone injection in post-partum depression: two multicentre, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet. 2018;392(10152):1058-1070.
27. Powell JG, Garland S, Preston K, et al. Brexanolone (Zulresso): finally, an FDA-approved treatment for postpartum depression. Ann Pharmacother. 2020;54(2):157-163.
28. Patterson R, Krohn H, Richardson E, et al. A brexanolone treatment program at an academic medical center: patient selection, 90-day posttreatment outcomes, and lessons learned. J Acad Consult Liaison Psychiatry. 2022;63(1):14-22.
29. World Health Organization. WHO model list of essential medicines - 22nd list (2021). World Health Organization. September 30, 2021. Accessed April 7, 2023. https://www.who.int/publications/i/item/WHO-MHP-HPS-EML-2021.02
30. Eby GA, Eby KL, Mruk H. Magnesium and major depression. In: Vink R, Nechifor M, eds. Magnesium in the Central Nervous System. University of Adelaide Press; 2011.
31. Plant TM, Zeleznik AJ. Knobil and Neill’s Physiology of Reproduction. 4th ed. Elsevier Inc.; 2015:2503-2550.
32. Sidebotham D, Le Grice IJ. Physiology and pathophysiology. In: Sidebotham D, McKee A, Gillham M, Levy J. Cardiothoracic Critical Care. Elsevier, Inc.; 2007:3-27.
33. Duley L, Gülmezoglu AM, Henderson-Smart DJ, et al. Magnesium sulphate and other anticonvulsants for women with pre-eclampsia. Cochrane Database Syst Rev. 2010;2010(11):CD000025.
34. Emergency supply of medicines. In: British National Formulary. British Medical Association, Royal Pharmaceutical Society; 2015:6. Accessed April 7, 2023. https://www.academia.edu/35076015/british_national_formulary_2015_pdf
35. Kwofie K, Wolfson AB. Intravenous magnesium sulfate for acute asthma exacerbation in children and adults. Am Fam Physician. 2021;103(4):245-246.
36. Patniyot IR, Gelfand AA. Acute treatment therapies for pediatric migraine: a qualitative systematic review. Headache. 2016;56(1):49-70.
37. Wang X, Du X, Yang H, et al. Use of intravenous magnesium sulfate among patients with acute myocardial infarction in China from 2001 to 2015: China PEACE-Retrospective AMI Study. BMJ Open. 2020;10(3):e033269.
38. Karhu E, Atlas SE, Jinrun G, et al. Intravenous infusion of magnesium sulfate is not associated with cardiovascular, liver, kidney, and metabolic toxicity in adults. J Clin Transl Res. 2018;4(1):47-55.
39. Noah L, Pickering G, Mazur A, et al. Impact of magnesium supplementation, in combination with vitamin B6, on stress and magnesium status: secondary data from a randomized controlled trial. Magnes Res. 2020;33(3):45-57.
40. Erstad BL, Cotugno CL. Management of alcohol withdrawal. Am J Health Syst Pharm. 1995;52(7):697-709.
41. Abumaria N, Luo L, Ahn M, et al. Magnesium supplement enhances spatial-context pattern separation and prevents fear overgeneralization. Behav Pharmacol. 2013;24(4):255-263.
42. Kirov GK, Tsachev KN. Magnesium, schizophrenia and manic-depressive disease. Neuropsychobiology. 1990;23(2):79-81.
43. Botturi A, Ciappolino V, Delvecchio G, et al. The role and the effect of magnesium in mental disorders: a systematic review. Nutrients. 2020;12(6):1661.
44. Kirkland AE, Sarlo GL, Holton KF. The role of magnesium in neurological disorders. Nutrients. 2018;10(6):730.
45. Magnesium sulfate intravenous side effects by likelihood and severity. WebMD. Accessed April 9, 2023. https://www.webmd.com/drugs/2/drug-149570/magnesium-sulfate-intravenous/details/list-sideeffects
46. Scopolamine base transdermal system – uses, side effects, and more. WebMD. Accessed April 9, 2023. https://www.webmd.com/drugs/2/drug-14032/scopolamine-transdermal/details
47. Bolden C, Cusack B, Richelson E. Antagonism by antimuscarinic and neuroleptic compounds at the five cloned human muscarinic cholinergic receptors expressed in Chinese hamster ovary cells. J Pharmacol Exp Ther. 1992;260(2):576-580.
48. Janowsky DS, el-Yousef MK, Davis JM, et al. A cholinergic-adrenergic hypothesis of mania and depression. Lancet. 1972;2(7778):632-635.
49. Janowsky DS, Risch SC, Gillin JC. Adrenergic-cholinergic balance and the treatment of affective disorders. Prog Neuropsychopharmacol Biol Psychiatry. 1983;7(2-3):297-307.
50. Gershon S, Shaw FH. Psychiatric sequelae of chronic exposure to organophosphorous insecticides. Lancet. 1972;1(7191):1371-1374.
51. Davis KL, Berger PA, Hollister LE, et al. Physostigmine in mania. Arch Gen Psychiatry. 1978;35(1):119-122.
52. Wang JC, Hinrichs AL, Stock H, et al. Evidence of common and specific genetic effects: association of the muscarinic acetylcholine receptor M2 (CHRM2) gene with alcohol dependence and major depressive syndrome. Hum Mol Genet. 2004;13(17):1903-1911.
53. Brown RG. Effects of antidepressants and anticholinergics in a mouse “behavioral despair” test. Eur J Pharmacol. 1979;58(3):331-334.
54. Porsolt RD, Le Pichon M, Jalfre M. Depression: a new animal model sensitive to antidepressant treatments. Nature. 1977;266(5604):730-732.
55. Ji CX, Zhang JJ. Effect of scopolamine on depression in mice. Abstract in English. Yao Xue Xue Bao. 2011;46(4):400-405.
56. Furey ML, Drevets WC. Antidepressant efficacy of the antimuscarinic drug scopolamine: a randomized, placebo-controlled clinical trial. Arch Gen Psychiatry. 2006;63(10):1121-1129.
57. Drevets WC, Furey ML. Replication of scopolamine’s antidepressant efficacy in major depressive disorder: a randomized, placebo-controlled clinical trial. Biol Psychiatry. 2010;67(5):432-438.
58. Furey ML, Khanna A, Hoffman EM, et al. Scopolamine produces larger antidepressant and antianxiety effects in women than in men. Neuropsychopharmacology. 2010;35(12):2479-2488.
59. Gibbs RB, Gabor R, Cox T, et al. Effects of raloxifene and estradiol on hippocampal acetylcholine release and spatial learning in the rat. Psychoneuroendocrinology. 2004;29(6):741-748.
60. Pongrac JL, Gibbs RB, Defranco DB. Estrogen-mediated regulation of cholinergic expression in basal forebrain neurons requires extracellular-signal-regulated kinase activity. Neuroscience. 2004;124(4):809-816.
61. Daniel JM, Dohanich GP. Acetylcholine mediates the estrogen-induced increase in NMDA receptor binding in CA1 of the hippocampus and the associated improvement in working memory. J Neurosci. 2001;21(17):6949-6956.
62. Gerhard DM, Wohleb ES, Duman RS. Emerging treatment mechanisms for depression: focus on glutamate and synaptic plasticity. Drug Discov Today. 2016;21(3):454-464.
63. Voderholzer U. Sleep deprivation and antidepressant treatment. Dialogues Clin Neurosci. 2003;5(4):366-369.
64. Hasselmann H. Scopolamine and depression: a role for muscarinic antagonism? CNS Neurol Disord Drug Targets. 2014;13(4):673-683.
65. Transderm scopolamine [prescribing information]. Warren, NJ: GSK Consumer Healthcare; 2019.
66. Jaffe RJ, Novakovic V, Peselow ED. Scopolamine as an antidepressant: a systematic review. Clin Neuropharmacol. 2013;36(1):24-26.
67. Karameh WK, Khani M. Intravenous clomipramine for treatment-resistant obsessive-compulsive disorder. Int J Neuropsychopharmacol. 2015;19(2):pyv084.
68. Andrews ET, Beattie RM, Tighe MP. Functional abdominal pain: what clinicians need to know. Arch Dis Child. 2020;105(10):938-944. doi:10.1136/archdischild-2020-318825
69. Aliane V, Pérez S, Bohren Y, et al. Key role of striatal cholinergic interneurons in processes leading to arrest of motor stereotypies. Brain. 2011;134(Pt 1):110-118. doi:10.1093/brain/awq285
70. Tzavara ET, Bymaster FP, Davis RJ, et al. M4 muscarinic receptors regulate the dynamics of cholinergic and dopaminergic neurotransmission: relevance to the pathophysiology and treatment of related CNS pathologies. FASEB J. 2004;18(12):1410-1412. doi:10.1096/fj.04-1575fje
71. Korczyn AD, Kish I. The mechanism of imipramine in enuresis nocturna. Clin Exp Pharmacol Physiol. 1979;6(1):31-35. doi:10.1111/j.1440-1681.1979.tb00004.x
72. Trimble MR. Worldwide use of clomipramine. J Clin Psychiatry. 1990;51(Suppl):51-54; discussion 55-58.
73. Gong W, Zhang S, Zong Y, et al. Involvement of the microglial NLRP3 inflammasome in the anti-inflammatory effect of the antidepressant clomipramine. J Affect Disord. 2019;254:15-25.
74. Piwowarska J, Wrzosek M, Radziwon’-Zaleska M. Serum cortisol concentration in patients with major depression after treatment with clomipramine. Pharmacol Rep. 2009;61(4):604-611.
75. Danish University Antidepressant Group (DUAG). Clomipramine dose-effect study in patients with depression: clinical end points and pharmacokinetics. Clin Pharmacol Ther. 1999;66(2):152-165.
76. Moukaddam NJ, Hirschfeld RMA. Intravenous antidepressants: a review. Depress Anxiety. 2004;19(1):1-9.
77. Gerretsen P, Pollock BG. Rediscovering adverse anticholinergic effects. J Clin Psychiatry. 2011;72(6):869-870. doi:10.4088/JCP.11ac07093
78. Thomas SJ, Shin M, McInnis MG, et al. Combination therapy with monoamine oxidase inhibitors and other antidepressants or stimulants: strategies for the management of treatment-resistant depression. Pharmacotherapy. 2015;35(4):433-449. doi:10.1002/phar.1576
79. Robles LA. Serotonin syndrome induced by fentanyl in a child: case report. Clin Neuropharmacol. 2015;38(5):206-208. doi:10.1097/WNF.0000000000000100
80. Fallon BA, Liebowitz MR, Campeas R, et al. Intravenous clomipramine for obsessive-compulsive disorder refractory to oral clomipramine: a placebo-controlled study. Arch Gen Psychiatry. 1998;55(10):918-924.
81. Vieta E, Florea I, Schmidt SN, et al. Intravenous vortioxetine to accelerate onset of effect in major depressive disorder: a 2-week, randomized, double-blind, placebo-controlled study. Int Clin Psychopharmacol. 2019;34(4):153-160.
82. Kasper S, Müller-Spahn F. Intravenous antidepressant treatment: focus on citalopram. Eur Arch Psychiatry Clin Neurosci. 2002;252(3):105-109.
83. Togay B, El-Mallakh RS. Posttraumatic stress disorder: from pathophysiology to pharmacology. Current Psychiatry. 2020;19(5):33-39.
84. Adhikari A, Lerner TN, Finkelstein J, et al. Basomedial amygdala mediates top-down control of anxiety and fear. Nature. 2015;527(7577):179-185. doi:10.1038/nature15698
85. Lipov E. In search of an effective treatment for combat-related post-traumatic stress disorder (PTSD): can the stellate ganglion block be the answer? Pain Pract. 2010;10(4):265-266.
86. Lipov E, Ritchie EC. A review of the use of stellate ganglion block in the treatment of PTSD. Curr Psychiatry Rep. 2015;17(8):599.
87. Olmsted KLR, Bartoszek M, McLean B, et al. Effect of stellate ganglion block treatment on posttraumatic stress disorder symptoms: a randomized clinical trial. JAMA Psychiatry. 2020;77(2):130-138.
88. Lipov E, Candido K. The successful use of left-sided stellate ganglion block in patients that fail to respond to right-sided stellate ganglion block for the treatment of post-traumatic stress disorder symptoms: a retrospective analysis of 205 patients. Mil Med. 2021;186(11-12):319-320.
89. Li Y, Loshak H. Stellate ganglion block for the treatment of post-traumatic stress disorder, depression, and anxiety. Canadian J Health Technol. 2021;1(3):1-30.
90. Kerzner J, Liu H, Demchenko I, et al. Stellate ganglion block for psychiatric disorders: a systematic review of the clinical research landscape. Chronic Stress (Thousand Oaks). 2021;5:24705470211055176.
91. Wie C, Gupta R, Maloney J, et al. Interventional modalities to treat complex regional pain syndrome. Curr Pain Headache Rep. 2021;25(2):10. doi:10.1007/s11916-020-00904-5
92. Chaturvedi A, Dash HH. Sympathetic blockade for the relief of chronic pain. J Indian Med Assoc. 2001;99(12):698-703.
93. Chester M, Hammond C. Leach A. Long-term benefits of stellate ganglion block in severe chronic refractory angina. Pain. 2000;87(1):103-105. doi:10.1016/S0304-3959(00)00270-0
94. Jeon Y. Therapeutic potential of stellate ganglion block in orofacial pain: a mini review. J Dent Anesth Pain Med. 2016;16(3):159-163. doi:10.17245/jdapm.2016.16.3.159
95. Shan HH, Chen HF, Ni Y, et al. Effects of stellate ganglion block through different approaches under guidance of ultrasound. Front Surg. 2022;8:797793. doi:10.3389/fsurg.2021.797793
96. Goel V, Patwardhan AM, Ibrahim M, et al. Complications associated with stellate ganglion nerve block: a systematic review. Reg Anesth Pain Med. 2019;rapm-2018-100127. doi:10.1136/rapm-2018-100127
97. Rowe FJ, Noonan CP. Botulinum toxin for the treatment of strabismus. Cochrane Database Syst Rev. 2017;3(3):CD006499.
98. Roggenkämper P, Jost WH, Bihari K, et al. Efficacy and safety of a new botulinum toxin type A free of complexing proteins in the treatment of blepharospasm. J Neural Transm (Vienna). 2006;113(3):303-312.
99. Heckmann M, Ceballos-Baumann AO, Plewig G; Hyperhidrosis Study Group. Botulinum toxin A for axillary hyperhidrosis (excessive sweating). N Engl J Med. 2001;344(7):488-493.
100. Carruthers JA, Lowe NJ, Menter MA, et al. A multicenter, double-blind, randomized, placebo-controlled study of the efficacy and safety of botulinum toxin type A in the treatment of glabellar lines. J Am Acad Dermatol. 2002;46(6):840-849.
101. Schurch B, de Sèze M, Denys P, et al. Botulinum toxin type A is a safe and effective treatment for neurogenic urinary incontinence: results of a single treatment, randomized, placebo controlled 6-month study. J Urol. 2005;174:196–200.
102. Aurora SK, Winner P, Freeman MC, et al. OnabotulinumtoxinA for treatment of chronic migraine: Pooled analyses of the 56-week PREEMPT clinical program. Headache. 2011;51(9):1358-1373.
103. Dashtipour K, Chen JJ, Walker HW, et al. Systematic literature review of abobotulinumtoxinA in clinical trials for adult upper limb spasticity. Am J Phys Med Rehabil. 2015;94(3):229-238.
104. Nitti VW, Dmochowski R, Herschorn S, et al. OnabotulinumtoxinA for the treatment of patients with overactive bladder and urinary incontinence: results of a phase 3, randomized, placebo-controlled trial. J Urol. 2017;197(2S):S216-S223.
105. Jongerius PH, van den Hoogen FJA, van Limbeek J, et al. Effect of botulinum toxin in the treatment of drooling: a controlled clinical trial. Pediatrics. 2004;114(3):620-627.
106. Zaninotto, G. Annese V, Costantini M, et al. Randomized controlled trial of botulinum toxin versus laparoscopic heller myotomy for esophageal achalasia. Ann Surg. 2004;239(3):364-370.
107. Dressler D, Adib Saberi F. Botulinum toxin: mechanisms of action. Eur Neurol. 2005;53:3-9.
108. Lewis MB, Bowler PJ. Botulinum toxin cosmetic therapy correlates with a more positive mood. J Cosmet Dermatol. 2009;8(1):24-26.
109. Affatato O, Moulin TC, Pisanu C, et al. High efficacy of onabotulinumtoxinA treatment in patients with comorbid migraine and depression: a meta-analysis. J Transl Med. 2021;19(1):133.
110. Finzi E, Wasserman E. Treatment of depression with botulinum toxin A: a case series. Dermatol Surg. 2006;32(5):645-649; discussion 649-650.
111. Schulze J, Neumann I, Magid M, et al. Botulinum toxin for the management of depression: an updated review of the evidence and meta-analysis. J Psychiatr Res. 2021;135:332-340.
112. Finzi E, Rosenthal NE. Emotional proprioception: treatment of depression with afferent facial feedback. J Psychiatr Res. 2016;80:93-96.
113. Söderkvist S, Ohlén K, Dimberg U. How the experience of emotion is modulated by facial feedback. J Nonverbal Behav. 2018;42(1):129-151.
114. Lewis, MB. The interactions between botulinum-toxin-based facial treatments and embodied emotions. Sci Rep. 2018;8(1):14720.
115. Li Y, Liu J, Liu X, et al. Antidepressant-like action of single facial injection of botulinum neurotoxin A is associated with augmented 5-HT levels and BDNF/ERK/CREB pathways in mouse brain. Neurosci Bull. 2019;35(4):661-672. Erratum in: Neurosci Bull. 2019;35(4):779-780.
116. Gündel H, Wolf A, Xidara V, et al. High psychiatric comorbidity in spasmodic torticollis: a controlled study. J Nerv Ment Dis. 2003;191(7):465-473.
117. Hall TA, McGwin G Jr, Searcey K, et al. Health-related quality of life and psychosocial characteristics of patients with benign essential blepharospasm. Arch Ophthalmol. 2006;124(1):116-119.
118. Ceylan D, Erer S, Zarifog˘lu M, et al. Evaluation of anxiety and depression scales and quality of life in cervical dystonia patients on botulinum toxin therapy and their relatives. Neurol Sci. 2019;40(4):725-731.
119. Heller AS, Lapate RC, Mayer KE, et al. The face of negative affect: trial-by-trial corrugator responses to negative pictures are positively associated with amygdala and negatively associated with ventromedial prefrontal cortex activity. J Cogn Neurosci. 2014;26(9):2102-2110.
120. Makunts T, Wollmer MA, Abagyan R. Postmarketing safety surveillance data reveals antidepressant effects of botulinum toxin across various indications and injection sites. Sci Rep. 2020;10(1):12851.
121. Ahsanuddin S, Roy S, Nasser W, et al. Adverse events associated with botox as reported in a Food and Drug Administration database. Aesthetic Plast Surg. 2021;45(3):1201-1209. doi:10.1007/s00266-020-02027-z
122. Kashif M, Tahir S, Ashfaq F, et al. Association of myofascial trigger points in neck and shoulder region with depression, anxiety, and stress among university students. J Pak Med Assoc. 2021;71(9):2139-2142.
123. Cigarán-Méndez M, Jiménez-Antona C, Parás-Bravo P, et al. Active trigger points are associated with anxiety and widespread pressure pain sensitivity in women, but not men, with tension type headache. Pain Pract. 2019;19(5):522-529.
124. Palacios-Ceña M, Castaldo M, Wang K, et al. Relationship of active trigger points with related disability and anxiety in people with tension-type headache. Medicine (Baltimore). 2017;96(13):e6548.
125. Karadas Ö, Inan LE, Ulas Ü, et al. Efficacy of local lidocaine application on anxiety and depression and its curative effect on patients with chronic tension-type headache. Eur Neurol. 2013;70(1-2):95-101.
126. Gerwin RD. Classification, epidemiology and natural history of myofascial pain syndrome. Curr Pain Headache Rep. 2001;5(5):412-420.
127. Castro Sánchez AM, García López H, Fernández Sánchez M, et al. Improvement in clinical outcomes after dry needling versus myofascial release on pain pressure thresholds, quality of life, fatigue, pain intensity, quality of sleep, anxiety, and depression in patients with fibromyalgia syndrome. Disabil Rehabil. 2019;41(19):2235-2246.
128. Healy GM, Finn DP, O’Gorman DA, et al. Pretreatment anxiety and pain acceptance are associated with response to trigger point injection therapy for chronic myofascial pain. Pain Med. 2015;16(10):1955-1966.
129. Morjaria JB, Lakshminarayana UB, Liu-Shiu-Cheong P, et al. Pneumothorax: a tale of pain or spontaneity. Ther Adv Chronic Dis. 2014;5(6):269-273.
Advances in the understanding of neurobiological and neuropsychiatric pathophysiology have opened new avenues of treatment for psychiatric patients. Historically, with a few exceptions, most psychiatric medications have been administered orally. However, many of the newer treatments require some form of specialized administration because they cannot be taken orally due to their chemical property (such as aducanumab); because there is the need to produce stable blood levels of the medication (such as brexanolone); because oral administration greatly diminished efficacy (such as oral vs IV magnesium or scopolamine), or because the treatment is focused on specific brain structures. This need for specialized administration has created a subspecialty called interventional psychiatry.
Part 1 of this 2-part article provides an overview of 1 type of interventional psychiatry: parenterally administered medications, including those administered via IV. We also describe 3 other interventional approaches to treatment: stellate ganglion blocks, glabellar botulinum toxin (BT) injections, and trigger point injections. In Part 2 we will review interventional approaches that involve neuromodulation.
Parenteral medications in psychiatry
In general, IV and IM medications can be more potent that oral medications due to their overall faster onset of action and higher blood concentrations. These more invasive forms of administration can have significant limitations, such as a risk of infection at the injection site, the need to be administered in a medical setting, additional time, and patient discomfort.

Table 1 lists short-acting injectable medications used in psychiatry, and Table 2 lists long-acting injectable medications. Parenteral administration of antipsychotics is performed to alleviate acute agitation or for chronic symptom control. These medications generally are not considered interventional treatments, but could be classified as such due to their invasive nature.1 Furthermore, inhalable loxapine—which is indicated for managing acute agitation—requires a Risk Evaluation and Mitigation Strategy program consisting of 2 hours observation and monitoring of respiratory status.2,3 Other indications for parenteral treatments include IM naltrexone extended release4 and subcutaneous injections of buprenorphine extended release5 and risperidone.6
IV administration
Most IV treatments described in this article are not FDA-approved for psychiatric treatment. Despite this, many interventional psychiatric treatments are part of clinical practice. IV infusion of ketamine is the most widely known and most researched of these. Table 3 lists other IV treatments that could be used as psychiatric treatment.
Ketamine
Since the early 1960s, ketamine has been used as a surgical anesthetic for animals. In the United States, it was approved for human surgical anesthesia in 1970. It was widely used during the Vietnam War due to its lack of inhibition of respiratory drive; medical staff first noticed an improvement in depressive symptoms and the resolution of suicidal ideation in patients who received ketamine. This led to further research on ketamine, particularly to determine its application in treatment-resistant depression (TRD) and other conditions.7 IV ketamine administration is most widely researched, but IM injections, intranasal sprays, and lozenges are also available. The dissociative properties of ketamine have led to its recreational use.8
Hypotheses for the mechanism of action of ketamine as an antidepressant include direct synaptic or extrasynaptic (GluN2B-selective), N-methyl-
Continue to: Ketamine is a schedule...
Ketamine is a schedule III medication with addictive properties. Delirium, panic attacks, hallucinations, nightmares, dysphoria, and paranoia may occur during and after use.13 Premedication with benzodiazepines, most notably lorazepam, is occasionally used to minimize ketamine’s adverse effects, but this generally is not recommended because doing so reduces ketamine’s antidepressant effects.14 Driving and operating heavy machinery is contraindicated after IV infusion. The usual protocol involves an IV infusion of ketamine 0.4 mg/kg to 1 mg/kg dosing over 1 hour. Doses between 0.4 mg/kg and 0.6 mg/kg are most common. Ketamine has a therapeutic window; doses >0.5 mg/kg are progressively less effective.15 Unlike the recommendation after esketamine administration, after receiving ketamine, patients remain in the care of their treatment team for <2 hours.
Esketamine, the S enantiomer of ketamine, was FDA-approved for TRD as an intranasal formulation. Esketamine is more commonly used than IV ketamine because it is FDA-approved and typically covered by insurance, but it may not be as effective.16 An economic analysis by Brendle et al17 suggested insurance companies would lower costs if they covered ketamine infusions vs intranasal esketamine.
Aducanumab and lecanemab
The most recent FDA-approved interventional agents are aducanumab and lecanemab, which are indicated for treating Alzheimer disease.18,19 Both are human monoclonal antibodies that bind selectively and with high affinity to amyloid beta plaque aggregates and promote their removal by Fc receptor–mediated phagocytosis.20
FDA approval of aducanumab and lecanemab was controversial. Initially, aducanumab’s safety monitoring board performed a futility analysis that suggested aducanumab was unlikely to separate from placebo, and the research was stopped.21 The manufacturer petitioned the FDA to consider the medication for accelerated approval on the basis of biomarker data showing that amyloid beta plaque aggregates become smaller. Current FDA approval is temporary to allow patients access to this potentially beneficial agent, but the manufacturer must supply clinical evidence that the reduction of amyloid beta plaques is associated with desirable changes in the course of Alzheimer disease, or risk losing the approval.
Lecanemab is also a human monoclonal antibody intended to remove amyloid beta plaques that was FDA-approved under the accelerated approval pathway.22 Unlike aducanumab, lecanemab demonstrated a statistically significant (although clinically imperceptible) reduction in the rate of cognitive decline; it did not show cognitive improvement.23 Lecanemab also significantly reduced amyloid beta plaques.23
Continue to: Aducanumab and lecanemab are generally...
Aducanumab and lecanemab are generally not covered by insurance and typically cost >$26,000 annually. Both are administered by IV infusion once a month. More monoclonal antibody medications for treating early Alzheimer disease are in the late stages of development, most notably donanebab.24 Observations during clinical trials found that in the later stages of Alzheimer disease, forceful removal of plaques by the autoimmune process damages neurons, while in less dense deposits of early dementia such removal is not harmful to the cells and prevents amyloid buildup.
Brexanolone is an aqueous formulation of allopregnanolone, a major metabolite of progesterone and a positive allosteric modulator of GABA-A receptors.25 Its levels are maximal at the end of the third trimester of pregnancy and fall rapidly following delivery. Research showed a 3-day infusion was rapidly and significantly effective for treating postpartum depression26 and brexanolone received FDA approval for this indication in March 2019.27 However, various administrative, economic, insurance, and other hurdles make it difficult for patients to access this treatment. Despite its rapid onset of action (usually 48 hours), brexanolone takes an average of 15 days to go through the prior authorization process.28 In addition to the need for prior authorization, the main impediment to the use of brexanolone is the 3-day infusion schedule, which greatly magnifies the cost but is partially circumvented by the availability of dedicated outpatient centers.
Magnesium
Magnesium is on the World Health Organization’s Model List of Essential Medicines.29 There has been extensive research on the use of magnesium sulfate for psychiatric indications, especially for depression.30 Magnesium functions similarly to calcium channel blockers by competitively blocking intracellular calcium channels, decreasing calcium availability, and inhibiting smooth muscle contractility.31 It also competes with calcium at the motor end plate, reducing excitation by inhibiting the release of acetylcholine.32 This property is used for high-dose IV magnesium treatment of impending preterm labor in obstetrics. Magnesium sulfate is the drug of choice in treating eclamptic seizures and preventing seizures in severe preeclampsia or gestational hypertension with severe features.33 It is also used to treat torsade de pointes, severe asthma exacerbations, constipation, and barium poisoning.34 Beneficial use in asthma treatment35 and the treatment of migraine36 have also been reported.
IV magnesium in myocardial infarction may be harmful,37 though outside of acute cardiac events, magnesium is found to be safe.38
Oral magnesium sulfate is a common over-the-counter anxiety remedy. As a general cell stabilizer (mediated by the reduction of intracellular calcium), magnesium is potentially beneficial outside of its muscle-relaxing properties, although muscle relaxing can benefit anxious patients. It is used to treat anxiety,39 alcohol withdrawal,40 and fear.41 Low magnesium blood levels are found in patients with depression, schizophrenia,42 and attention-deficit/hyperactivity disorder.43 However, it is important to note that the therapeutic effect of magnesium when treating anxiety and headache is independent of preinfusion magnesium blood levels.43
Continue to: The efficacy of oral magnesium...
The efficacy of oral magnesium is not robust. However, IV administration has a pronounced beneficial effect as an abortive and preventative treatment in many patients with anxiety.44
IV administration of magnesium can produce adverse effects, including flushing, sweating, hypotension, depressed reflexes, flaccid paralysis, hypothermia, circulatory collapse, and cardiac and CNS depression. These complications are very rare and dose-dependent.45 Magnesium is excreted by the kidneys, and dosing must be decreased in patients with kidney failure. The most common adverse effect is local burning along the vein upon infusion; small doses of IV lidocaine can remedy this. Hot flashes are also common.45
Various dosing strategies are available. In patients with anxiety, application dosing is based on the recommended preeclampsia IV dose of 4 g diluted in 250 mL of 5% dextrose. Much higher doses may be used in obstetrics. Unlike in obstetrics, for psychiatric indications, magnesium is administered over 60 to 90 minutes. Heart monitoring is recommended.
Scopolamine
Scopolamine is primarily used to relieve nausea, vomiting, and dizziness associated with motion sickness and recovery from anesthesia. It is also used in ophthalmology and in patients with excessive sweating. In off-label and experimental applications, scopolamine has been used in patients with TRD.46
Scopolamine is an anticholinergic medication. It is a nonselective antagonist at muscarinic receptors.47 Tricyclic antidepressants (TCAs) possess strong anticholinergic function. Newer generations of antidepressants were designed specifically not to have this function because it was believed to be an unwanted and potentially dangerous adverse effect. However, data suggest that anticholinergic action is important in decreasing depressive symptoms. Several hypotheses of anticholinergic effects on depression have been published since the 1970s. They include the cholinergic-adrenergic hypothesis,48 acetylcholine predominance relative to adrenergic action hypothesis,49 and insecticide poisoning observations.50 Centrally acting physostigmine (but not peripherally acting neostigmine) was reported to control mania.48,51 A genetic connection between the M2acetylcholine receptor in patients with major depressive disorder (MDD) and alcohol use disorder is also suggestive.52
Continue to: Multiple animal studies show...
Multiple animal studies show direct improvement in mobility and a decrease in despair upon introducing anticholinergic substances.53-55 The cholinergic theory of depression has been studied in several controlled clinical human studies.56,57 Use of a short-acting anticholinergic glycopyrrolate during electroconvulsive therapy (ECT) may contribute to the procedure’s efficacy.
Human research shows scopolamine has a higher efficacy as an antidepressant and anti-anxiety medication in women than in men,58 possibly because estrogen increases the activity of choline acetyltransferase and release of acetylcholine.59,60 M2receptors mediate estrogen influence on the NMDAR, which may explain the anticholinergic effects of depression treatments in women.61
Another proposed mechanism of action of scopolamine is a potent inhibition of the NMDAR.62 Rapid treatments of depression may be based on this mechanism. Examples of such treatments include IV ketamine and sleep deprivation.63 IV scopolamine shows potency in treating MDD and bipolar depression. This treatment should be reserved for patients who do not respond to or are not candidates for other usual treatment modalities of MDD and for the most severe cases. Scopolamine is 30 times more potent than amitriptyline in anticholinergic effect and reportedly produces sustained improvement in MDD.64
Scopolamine has no black-box warnings. It has not been studied in pregnant women and is not recommended for use during pregnancy. Due to possible negative cardiovascular effects, a normal electrocardiogram is required before the start of treatment. Exercise caution in patients with glaucoma, benign prostatic enlargement, gastroparesis, unstable cardiovascular status, or severe renal impairment.
Treatment with scopolamine is not indicated for patients with myasthenia gravis, psychosis, or seizures. Patients must be off potassium for 3 days before beginning scopolamine treatment. Patients should consult with their cardiologist before having a scopolamine infusion. Adverse reactions may include psychosis, tachycardia, seizures, paralytic ileus, and glaucoma exacerbation. The most common adverse effects of scopolamine infusion treatment include drowsiness, dry mouth, blurred vision, lightheadedness, and dizziness. Due to possible drowsiness, operating motor vehicles or heavy machinery must be avoided on the day of treatment.65 Overall, the adverse effects of scopolamine are preventable and manageable, and its antidepressant efficacy is noteworthy.66
Continue to: Treatment typically consists of 3 consecutive infusions...
Treatment typically consists of 3 consecutive infusions of 4 mcg/kg separated by 3 to 5 days.56 It is possible to have a longer treatment course if the patient experiences only partial improvement. Repeated courses or maintenance treatment (similar to ECT maintenance) are utilized in some patients if indicated. Cardiac monitoring is mandatory.
Clomipramine
Clomipramine, a TCA, acts as a preferential inhibitor of 5-hydroxytryptamine uptake and has proven effective in managing depression, TRD, and obsessive-compulsive disorder (OCD).67 Although this medication has reported treatment benefits for patients with phobia, panic disorder,15 chronic pain,68 Tourette syndrome,69 premature ejaculation, anorexia nervosa,70 cataplexy,49 and enuresis,71 it is FDA-approved only for the treatment of OCD.72 Clomipramine may also be beneficial for pain and headache, possibly because of its anti-inflammatory action.73 The anticholinergic effects of clomipramine may add to its efficacy in depression.
The pathophysiology of MDD is connected to hyperactivity of the HPA axis and elevated cortisol levels. Higher clomipramine plasma levels show a linear correlation with lower cortisol secretion and levels, possibly aiding in the treatment of MDD and anxiety.74 The higher the blood level, the more pronounced clomipramine’s therapeutic effect across multiple domains.75
IV infusion of clomipramine produces the highest concentration in the shortest time, but overall, research does not necessarily support increased efficacy of IV over oral administration. There is evidence suggesting that subgroups of patients with severe, treatment-refractory OCD may benefit from IV agents and research suggests a faster onset of action.76 Faster onset of symptom relief is the basis for IV clomipramine use. In patients with OCD, it can take several months for oral medications to produce therapeutic benefits; not all patients can tolerate this. In such scenarios, IV administration may be considered, though it is not appropriate for routine use until more research is available. Patients with treatment-resistant OCD who have exhausted other means of symptom relief may also be candidates for IV treatment.
The adverse effects of IV clomipramine are no different from oral use, though they may be more pronounced. A pretreatment cardiac exam is desirable because clomipramine, like other TCAs, may be cardiotoxic. The anticholinergic adverse effects of TCAs are well known to clinicians77 and partially explained in the scopolamine section of this article. It is not advisable to combine clomipramine with other TCAs or serotonin reuptake inhibitors. Clomipramine also should not be combined with monoamine oxidase inhibitors, though such a combination was reported in medical literature.78 Combination with antiarrhythmics such as lidocaine or opioids such as fentanyl or and tramadol is highly discouraged (fentanyl and tramadol also have serotonergic effects).79
Continue to: Clomipramine for IV use is not commercially available...
Clomipramine for IV use is not commercially available and must be sterilely compounded. The usual course of treatment is a series of 3 infusions: 150 mg on Day 1, 200 mg on Day 2 or Day 3, and 250 mg on Day 3, Day 4, or Day 5, depending on tolerability. A protocol with a 50 mg/d starting dose and titration up to a maximum dose of 225 mg/d over 5 to 7 days has been suggested for inpatient settings.67 Titration to 250 mg is more common.80
A longer series may be performed, but this increases the likelihood of adverse effects. Booster and maintenance treatments are also completed when required. Cardiac monitoring is mandatory.
Vortioxetine and citalopram
IV treatment of depression with
Injections and blocks
Three interventional approaches to treatment that possess psychotherapeutic potential include stellate ganglion blocks (SGBs), glabellar BT injections, and trigger point injections (TPIs). None of these are FDA-approved for psychiatric treatment.
Stellate ganglion blocks
The sympathetic nervous system is involved in autonomic hyperarousal and is at the core of posttraumatic symptomatology.83 Insomnia, anxiety, irritability, hypervigilance, and other excitatory CNS events are connected to the sympathetic nervous system and amygdala activation is commonly observed in those exposed to extreme stress or traumatic events.84
Continue to: SGBs were first performed 100 years ago...
SGBs were first performed 100 years ago and reported to have beneficial psychiatric effects at the end of the 1940s. In 1998 in Finland, improvement of posttraumatic stress disorder (PTSD) symptoms was observed accidentally via thoracic level spine blocks.85 In 2006, cervical level sympathetic blocks were shown to be effective for PTSD symptom control.86 By the end of 2010, Veterans Administration hospitals adopted SGBs to treat veterans with PTSD.87,88 The first multisite, randomized clinical trial of
Since the stellate ganglion is connected to the amygdala, SGB has also been assessed for treating anxiety and depression.89,90 Outside of PTSD, SGBs are used to treat complex regional pain syndrome,91 phantom limb pain, trigeminal neuralgia,92 intractable angina,93 and postherpetic neuralgia in the head, neck, upper chest, or arms.94 The procedure consists of an injection of a local anesthetic through a 25-gauge needle into the stellate sympathetic ganglion at the C6 or C7 vertebral levels. An injection into C6 is considered safer because of specific cervical spine anatomy. Ideally, fluoroscopic guidance or ultrasound is used to guide needle insertion.95
A severe drop in blood pressure may be associated with SGBs and is mitigated by IV hydration. Other adverse effects include red eyes, drooping of the eyelids, nasal congestion, hoarseness, difficulty swallowing, a sensation of a “lump” in the throat, and a sensation of warmth or tingling in the arm or hand. Bilateral SGB is contraindicated due to the danger of respiratory arrest.96
Glabellar BT injections
OnabotulinumtoxinA (BT) injection was first approved for therapeutic use in 1989 for eye muscle disorders such as strabismus97 and blepharospasm.98 It was later approved for several other indications, including cosmetic use, hyperhidrosis, migraine prevention, neurogenic bladder disorder, overactive bladder, urinary incontinence, and spasticity.99-104 BT is used off-label for achalasia and sialorrhea.105,106 Its mechanism of action is primarily attributed to muscle paralysis by blocking presynaptic acetylcholine release into neuromuscular junctions.107
Facial BT injections are usually administered for cosmetic purposes, but smoothing forehead wrinkles and frown lines (the glabellar region of the face) both have antidepressant effects.108 BT injections into the glabellar region also demonstrate antidepressant effects, particularly in patients with comorbid migraines and MDD.109 Early case observations supported the independent benefit of the toxin on MDD when the toxin was injected into the glabellar region.110,111 The most frequent protocol involves injections in the procerus and corrugator muscles.
Continue to: The facial feedback/emotional proprioception hypothesis...
The facial feedback/emotional proprioception hypothesis has dominated thinking about the mood-improving effects of BT. The theory is that blocking muscular expression of sadness (especially in the face) interrupts the experience of sadness; therefore, depression subsides.112,113 However, BT injections in the muscles involved in the smile and an expression of positive emotions (lateral part of the musculus orbicularis oculi) have been associated with increased MDD scores.114 Thus, the mechanism clearly involves more than the cosmetic effect, since facial muscle injections in rats also have antidepressant effects.115
The use of progressive muscle relaxation is well-established in psychiatric treatment. The investigated conditions of increased muscle tone, especially torticollis and blepharospasm, are associated with MDD, and it may be speculated that proprioceptive feedback from the affected muscles may be causally involved in this association.116-118 Activity of the corrugator muscle has been positively associated with increased amygdala activity.119 This suggests a potential similar mechanism to that hypothesized for SGB.
Alternatively, BT is commonly used to treat chronic conditions that may contribute to depression; its success in relieving the underlying problem may indirectly relieve MDD. Thus, in a postmarketing safety evaluation of BT, MDD was demonstrated 40% to 88% less often by patients treated with BT for 6 of the 8 conditions and injection sites, such as in spasms and spasticity of arms and legs, torticollis and neck pain, and axilla and palm injections for hyperhidrosis. In a parotid and submandibular glands BT injection subcohort, no patients experienced depressive symptoms.120
Medicinal BT is generally considered safe. The most common adverse effects are hypersensitivity, injection site reactions, and other adverse effects specific to the injection site.121 Additionally, the cosmetic effects are transient, given the nature of the medication.
Trigger point injections
TPIs in the neck and shoulders are frequently used to treat tension headaches and various referred pain locations in the face and arms. Tension and depression frequently overlap in clinical practice.122 Relieving muscle tension (with resulting trigger points) improves muscle function and mood.
Continue to: The higher the number of active...
The higher the number of active trigger points (TPs), the greater the physical burden of headache and the higher the anxiety level. Gender differences in TP prevalence and TPI efficacy have been described in the literature. For example, the number of active TPs seems directly associated with anxiety levels in women but not in men.123 Although TPs appear to be more closely associated with anxiety than depression,124 depression associated with muscle tension does improve with TPIs. European studies have demonstrated a decrease in scores on the Hamilton Depression Rating Scale and Hamilton Anxiety Rating Scale following TPI treatment.125 The effect may be indirect, as when a patient’s pain is relieved, sleep and other psychiatric symptoms improve.126
A randomized controlled trial by Castro Sánchez et al127 demonstrated that dry needling therapy in patients with fibromyalgia syndrome (FMS) showed improvements in pain pressure thresholds, body pain, vitality, and social function, as well as total FMS symptoms, quality of sleep, anxiety, hospital anxiety and depression, general pain intensity, and fatigue.
Myofascial pain syndrome, catastrophizing, and muscle tension are common in patients with depression, anxiety, and somatization. Local TPI therapy aimed at inactivating pain generators is supported by moderate quality evidence. All manner of therapies have been described, including injection of saline, corticosteroids, local anesthetic agents, and dry needling. BT injections in chronic TPs are also practiced, though no specific injection therapy has been reliably shown to be more advantageous than another. The benefits of TPIs may be derived from the needle itself rather than from any specific substance injected. Stimulation of a local twitch response with direct needling of the TP appears of importance. There is no established consensus regarding the number of injection points, frequency of administration, and volume or type of injectate.128
Adverse effects of TPIs relate to the nature of the invasive therapy, with the risk of tissue damage and bleeding. Pneumothorax risk is present with needle insertion at the neck and thorax.129 Patients with diabetes may experience variations in blood sugar control if steroids are used.
Bottom Line
Interventional treatment modalities that may have a role in psychiatric treatment include IV administration of ketamine, aducanumab, lecanemab, brexanolone, magnesium, scopolamine, and clomipramine. Other interventional approaches include stellate ganglion blocks, glabellar botulinum toxin injections, and trigger point injections.
Related Resources
- Dokucu ME, Janicak PG. Nontraditional therapies for treatment-resistant depression. Current Psychiatry. 2021; 20(9):38-43,49. doi:10.12788/cp.0166
- Kim J, Khoury R, Grossberg GT. Botulinum toxin: emerging psychiatric indications. Current Psychiatry. 2018;17(12):8-18.
Drug Brand Names
Aducanumab • Aduhelm
Aripiprazole • Abilify
Aripiprazole lauroxil • Aristada
Brexanolone • Zulresso
Buprenorphine • Sublocade
Citalopram • Celexa
Clomipramine • Anafranil
Diazepam • Valium
Droperidol • Inapsine
Esketamine • Spravato
Fentanyl • Actiq
Fluphenazine decanoate • Modecate
Fluphenazine hydrochloride • Prolixin
Haloperidol decanoate • Haldol decanoate
Haloperidol lactate • Haldol
Ketamine • Ketalar
Lecanemab • Leqembi
Lidocaine • Xylocaine
Lorazepam • Ativan
Loxapine inhaled • Adasuve
Naltrexone • Vivitrol
Magnesium sulfate • Sulfamag
Midazolam • Versed
Olanzapine • Zyprexa
OnabotulinumtoxinA injection • Botox
Paliperidone • Invega Hafyera, Invega Sustenna, Invega Trinza
Rapamycin • Rapamune, Sirolimus
Risperidone • Perseris
Risperidone microspheres • Risperdal Consta, Rykindo
Scopolamine • Hyoscine
Tramadol • Conzip
Vortioxetine • Trintellix
Ziprasidone • Geodon
Advances in the understanding of neurobiological and neuropsychiatric pathophysiology have opened new avenues of treatment for psychiatric patients. Historically, with a few exceptions, most psychiatric medications have been administered orally. However, many of the newer treatments require some form of specialized administration because they cannot be taken orally due to their chemical property (such as aducanumab); because there is the need to produce stable blood levels of the medication (such as brexanolone); because oral administration greatly diminished efficacy (such as oral vs IV magnesium or scopolamine), or because the treatment is focused on specific brain structures. This need for specialized administration has created a subspecialty called interventional psychiatry.
Part 1 of this 2-part article provides an overview of 1 type of interventional psychiatry: parenterally administered medications, including those administered via IV. We also describe 3 other interventional approaches to treatment: stellate ganglion blocks, glabellar botulinum toxin (BT) injections, and trigger point injections. In Part 2 we will review interventional approaches that involve neuromodulation.
Parenteral medications in psychiatry
In general, IV and IM medications can be more potent that oral medications due to their overall faster onset of action and higher blood concentrations. These more invasive forms of administration can have significant limitations, such as a risk of infection at the injection site, the need to be administered in a medical setting, additional time, and patient discomfort.

Table 1 lists short-acting injectable medications used in psychiatry, and Table 2 lists long-acting injectable medications. Parenteral administration of antipsychotics is performed to alleviate acute agitation or for chronic symptom control. These medications generally are not considered interventional treatments, but could be classified as such due to their invasive nature.1 Furthermore, inhalable loxapine—which is indicated for managing acute agitation—requires a Risk Evaluation and Mitigation Strategy program consisting of 2 hours observation and monitoring of respiratory status.2,3 Other indications for parenteral treatments include IM naltrexone extended release4 and subcutaneous injections of buprenorphine extended release5 and risperidone.6
IV administration
Most IV treatments described in this article are not FDA-approved for psychiatric treatment. Despite this, many interventional psychiatric treatments are part of clinical practice. IV infusion of ketamine is the most widely known and most researched of these. Table 3 lists other IV treatments that could be used as psychiatric treatment.
Ketamine
Since the early 1960s, ketamine has been used as a surgical anesthetic for animals. In the United States, it was approved for human surgical anesthesia in 1970. It was widely used during the Vietnam War due to its lack of inhibition of respiratory drive; medical staff first noticed an improvement in depressive symptoms and the resolution of suicidal ideation in patients who received ketamine. This led to further research on ketamine, particularly to determine its application in treatment-resistant depression (TRD) and other conditions.7 IV ketamine administration is most widely researched, but IM injections, intranasal sprays, and lozenges are also available. The dissociative properties of ketamine have led to its recreational use.8
Hypotheses for the mechanism of action of ketamine as an antidepressant include direct synaptic or extrasynaptic (GluN2B-selective), N-methyl-
Continue to: Ketamine is a schedule...
Ketamine is a schedule III medication with addictive properties. Delirium, panic attacks, hallucinations, nightmares, dysphoria, and paranoia may occur during and after use.13 Premedication with benzodiazepines, most notably lorazepam, is occasionally used to minimize ketamine’s adverse effects, but this generally is not recommended because doing so reduces ketamine’s antidepressant effects.14 Driving and operating heavy machinery is contraindicated after IV infusion. The usual protocol involves an IV infusion of ketamine 0.4 mg/kg to 1 mg/kg dosing over 1 hour. Doses between 0.4 mg/kg and 0.6 mg/kg are most common. Ketamine has a therapeutic window; doses >0.5 mg/kg are progressively less effective.15 Unlike the recommendation after esketamine administration, after receiving ketamine, patients remain in the care of their treatment team for <2 hours.
Esketamine, the S enantiomer of ketamine, was FDA-approved for TRD as an intranasal formulation. Esketamine is more commonly used than IV ketamine because it is FDA-approved and typically covered by insurance, but it may not be as effective.16 An economic analysis by Brendle et al17 suggested insurance companies would lower costs if they covered ketamine infusions vs intranasal esketamine.
Aducanumab and lecanemab
The most recent FDA-approved interventional agents are aducanumab and lecanemab, which are indicated for treating Alzheimer disease.18,19 Both are human monoclonal antibodies that bind selectively and with high affinity to amyloid beta plaque aggregates and promote their removal by Fc receptor–mediated phagocytosis.20
FDA approval of aducanumab and lecanemab was controversial. Initially, aducanumab’s safety monitoring board performed a futility analysis that suggested aducanumab was unlikely to separate from placebo, and the research was stopped.21 The manufacturer petitioned the FDA to consider the medication for accelerated approval on the basis of biomarker data showing that amyloid beta plaque aggregates become smaller. Current FDA approval is temporary to allow patients access to this potentially beneficial agent, but the manufacturer must supply clinical evidence that the reduction of amyloid beta plaques is associated with desirable changes in the course of Alzheimer disease, or risk losing the approval.
Lecanemab is also a human monoclonal antibody intended to remove amyloid beta plaques that was FDA-approved under the accelerated approval pathway.22 Unlike aducanumab, lecanemab demonstrated a statistically significant (although clinically imperceptible) reduction in the rate of cognitive decline; it did not show cognitive improvement.23 Lecanemab also significantly reduced amyloid beta plaques.23
Continue to: Aducanumab and lecanemab are generally...
Aducanumab and lecanemab are generally not covered by insurance and typically cost >$26,000 annually. Both are administered by IV infusion once a month. More monoclonal antibody medications for treating early Alzheimer disease are in the late stages of development, most notably donanebab.24 Observations during clinical trials found that in the later stages of Alzheimer disease, forceful removal of plaques by the autoimmune process damages neurons, while in less dense deposits of early dementia such removal is not harmful to the cells and prevents amyloid buildup.
Brexanolone is an aqueous formulation of allopregnanolone, a major metabolite of progesterone and a positive allosteric modulator of GABA-A receptors.25 Its levels are maximal at the end of the third trimester of pregnancy and fall rapidly following delivery. Research showed a 3-day infusion was rapidly and significantly effective for treating postpartum depression26 and brexanolone received FDA approval for this indication in March 2019.27 However, various administrative, economic, insurance, and other hurdles make it difficult for patients to access this treatment. Despite its rapid onset of action (usually 48 hours), brexanolone takes an average of 15 days to go through the prior authorization process.28 In addition to the need for prior authorization, the main impediment to the use of brexanolone is the 3-day infusion schedule, which greatly magnifies the cost but is partially circumvented by the availability of dedicated outpatient centers.
Magnesium
Magnesium is on the World Health Organization’s Model List of Essential Medicines.29 There has been extensive research on the use of magnesium sulfate for psychiatric indications, especially for depression.30 Magnesium functions similarly to calcium channel blockers by competitively blocking intracellular calcium channels, decreasing calcium availability, and inhibiting smooth muscle contractility.31 It also competes with calcium at the motor end plate, reducing excitation by inhibiting the release of acetylcholine.32 This property is used for high-dose IV magnesium treatment of impending preterm labor in obstetrics. Magnesium sulfate is the drug of choice in treating eclamptic seizures and preventing seizures in severe preeclampsia or gestational hypertension with severe features.33 It is also used to treat torsade de pointes, severe asthma exacerbations, constipation, and barium poisoning.34 Beneficial use in asthma treatment35 and the treatment of migraine36 have also been reported.
IV magnesium in myocardial infarction may be harmful,37 though outside of acute cardiac events, magnesium is found to be safe.38
Oral magnesium sulfate is a common over-the-counter anxiety remedy. As a general cell stabilizer (mediated by the reduction of intracellular calcium), magnesium is potentially beneficial outside of its muscle-relaxing properties, although muscle relaxing can benefit anxious patients. It is used to treat anxiety,39 alcohol withdrawal,40 and fear.41 Low magnesium blood levels are found in patients with depression, schizophrenia,42 and attention-deficit/hyperactivity disorder.43 However, it is important to note that the therapeutic effect of magnesium when treating anxiety and headache is independent of preinfusion magnesium blood levels.43
Continue to: The efficacy of oral magnesium...
The efficacy of oral magnesium is not robust. However, IV administration has a pronounced beneficial effect as an abortive and preventative treatment in many patients with anxiety.44
IV administration of magnesium can produce adverse effects, including flushing, sweating, hypotension, depressed reflexes, flaccid paralysis, hypothermia, circulatory collapse, and cardiac and CNS depression. These complications are very rare and dose-dependent.45 Magnesium is excreted by the kidneys, and dosing must be decreased in patients with kidney failure. The most common adverse effect is local burning along the vein upon infusion; small doses of IV lidocaine can remedy this. Hot flashes are also common.45
Various dosing strategies are available. In patients with anxiety, application dosing is based on the recommended preeclampsia IV dose of 4 g diluted in 250 mL of 5% dextrose. Much higher doses may be used in obstetrics. Unlike in obstetrics, for psychiatric indications, magnesium is administered over 60 to 90 minutes. Heart monitoring is recommended.
Scopolamine
Scopolamine is primarily used to relieve nausea, vomiting, and dizziness associated with motion sickness and recovery from anesthesia. It is also used in ophthalmology and in patients with excessive sweating. In off-label and experimental applications, scopolamine has been used in patients with TRD.46
Scopolamine is an anticholinergic medication. It is a nonselective antagonist at muscarinic receptors.47 Tricyclic antidepressants (TCAs) possess strong anticholinergic function. Newer generations of antidepressants were designed specifically not to have this function because it was believed to be an unwanted and potentially dangerous adverse effect. However, data suggest that anticholinergic action is important in decreasing depressive symptoms. Several hypotheses of anticholinergic effects on depression have been published since the 1970s. They include the cholinergic-adrenergic hypothesis,48 acetylcholine predominance relative to adrenergic action hypothesis,49 and insecticide poisoning observations.50 Centrally acting physostigmine (but not peripherally acting neostigmine) was reported to control mania.48,51 A genetic connection between the M2acetylcholine receptor in patients with major depressive disorder (MDD) and alcohol use disorder is also suggestive.52
Continue to: Multiple animal studies show...
Multiple animal studies show direct improvement in mobility and a decrease in despair upon introducing anticholinergic substances.53-55 The cholinergic theory of depression has been studied in several controlled clinical human studies.56,57 Use of a short-acting anticholinergic glycopyrrolate during electroconvulsive therapy (ECT) may contribute to the procedure’s efficacy.
Human research shows scopolamine has a higher efficacy as an antidepressant and anti-anxiety medication in women than in men,58 possibly because estrogen increases the activity of choline acetyltransferase and release of acetylcholine.59,60 M2receptors mediate estrogen influence on the NMDAR, which may explain the anticholinergic effects of depression treatments in women.61
Another proposed mechanism of action of scopolamine is a potent inhibition of the NMDAR.62 Rapid treatments of depression may be based on this mechanism. Examples of such treatments include IV ketamine and sleep deprivation.63 IV scopolamine shows potency in treating MDD and bipolar depression. This treatment should be reserved for patients who do not respond to or are not candidates for other usual treatment modalities of MDD and for the most severe cases. Scopolamine is 30 times more potent than amitriptyline in anticholinergic effect and reportedly produces sustained improvement in MDD.64
Scopolamine has no black-box warnings. It has not been studied in pregnant women and is not recommended for use during pregnancy. Due to possible negative cardiovascular effects, a normal electrocardiogram is required before the start of treatment. Exercise caution in patients with glaucoma, benign prostatic enlargement, gastroparesis, unstable cardiovascular status, or severe renal impairment.
Treatment with scopolamine is not indicated for patients with myasthenia gravis, psychosis, or seizures. Patients must be off potassium for 3 days before beginning scopolamine treatment. Patients should consult with their cardiologist before having a scopolamine infusion. Adverse reactions may include psychosis, tachycardia, seizures, paralytic ileus, and glaucoma exacerbation. The most common adverse effects of scopolamine infusion treatment include drowsiness, dry mouth, blurred vision, lightheadedness, and dizziness. Due to possible drowsiness, operating motor vehicles or heavy machinery must be avoided on the day of treatment.65 Overall, the adverse effects of scopolamine are preventable and manageable, and its antidepressant efficacy is noteworthy.66
Continue to: Treatment typically consists of 3 consecutive infusions...
Treatment typically consists of 3 consecutive infusions of 4 mcg/kg separated by 3 to 5 days.56 It is possible to have a longer treatment course if the patient experiences only partial improvement. Repeated courses or maintenance treatment (similar to ECT maintenance) are utilized in some patients if indicated. Cardiac monitoring is mandatory.
Clomipramine
Clomipramine, a TCA, acts as a preferential inhibitor of 5-hydroxytryptamine uptake and has proven effective in managing depression, TRD, and obsessive-compulsive disorder (OCD).67 Although this medication has reported treatment benefits for patients with phobia, panic disorder,15 chronic pain,68 Tourette syndrome,69 premature ejaculation, anorexia nervosa,70 cataplexy,49 and enuresis,71 it is FDA-approved only for the treatment of OCD.72 Clomipramine may also be beneficial for pain and headache, possibly because of its anti-inflammatory action.73 The anticholinergic effects of clomipramine may add to its efficacy in depression.
The pathophysiology of MDD is connected to hyperactivity of the HPA axis and elevated cortisol levels. Higher clomipramine plasma levels show a linear correlation with lower cortisol secretion and levels, possibly aiding in the treatment of MDD and anxiety.74 The higher the blood level, the more pronounced clomipramine’s therapeutic effect across multiple domains.75
IV infusion of clomipramine produces the highest concentration in the shortest time, but overall, research does not necessarily support increased efficacy of IV over oral administration. There is evidence suggesting that subgroups of patients with severe, treatment-refractory OCD may benefit from IV agents and research suggests a faster onset of action.76 Faster onset of symptom relief is the basis for IV clomipramine use. In patients with OCD, it can take several months for oral medications to produce therapeutic benefits; not all patients can tolerate this. In such scenarios, IV administration may be considered, though it is not appropriate for routine use until more research is available. Patients with treatment-resistant OCD who have exhausted other means of symptom relief may also be candidates for IV treatment.
The adverse effects of IV clomipramine are no different from oral use, though they may be more pronounced. A pretreatment cardiac exam is desirable because clomipramine, like other TCAs, may be cardiotoxic. The anticholinergic adverse effects of TCAs are well known to clinicians77 and partially explained in the scopolamine section of this article. It is not advisable to combine clomipramine with other TCAs or serotonin reuptake inhibitors. Clomipramine also should not be combined with monoamine oxidase inhibitors, though such a combination was reported in medical literature.78 Combination with antiarrhythmics such as lidocaine or opioids such as fentanyl or and tramadol is highly discouraged (fentanyl and tramadol also have serotonergic effects).79
Continue to: Clomipramine for IV use is not commercially available...
Clomipramine for IV use is not commercially available and must be sterilely compounded. The usual course of treatment is a series of 3 infusions: 150 mg on Day 1, 200 mg on Day 2 or Day 3, and 250 mg on Day 3, Day 4, or Day 5, depending on tolerability. A protocol with a 50 mg/d starting dose and titration up to a maximum dose of 225 mg/d over 5 to 7 days has been suggested for inpatient settings.67 Titration to 250 mg is more common.80
A longer series may be performed, but this increases the likelihood of adverse effects. Booster and maintenance treatments are also completed when required. Cardiac monitoring is mandatory.
Vortioxetine and citalopram
IV treatment of depression with
Injections and blocks
Three interventional approaches to treatment that possess psychotherapeutic potential include stellate ganglion blocks (SGBs), glabellar BT injections, and trigger point injections (TPIs). None of these are FDA-approved for psychiatric treatment.
Stellate ganglion blocks
The sympathetic nervous system is involved in autonomic hyperarousal and is at the core of posttraumatic symptomatology.83 Insomnia, anxiety, irritability, hypervigilance, and other excitatory CNS events are connected to the sympathetic nervous system and amygdala activation is commonly observed in those exposed to extreme stress or traumatic events.84
Continue to: SGBs were first performed 100 years ago...
SGBs were first performed 100 years ago and reported to have beneficial psychiatric effects at the end of the 1940s. In 1998 in Finland, improvement of posttraumatic stress disorder (PTSD) symptoms was observed accidentally via thoracic level spine blocks.85 In 2006, cervical level sympathetic blocks were shown to be effective for PTSD symptom control.86 By the end of 2010, Veterans Administration hospitals adopted SGBs to treat veterans with PTSD.87,88 The first multisite, randomized clinical trial of
Since the stellate ganglion is connected to the amygdala, SGB has also been assessed for treating anxiety and depression.89,90 Outside of PTSD, SGBs are used to treat complex regional pain syndrome,91 phantom limb pain, trigeminal neuralgia,92 intractable angina,93 and postherpetic neuralgia in the head, neck, upper chest, or arms.94 The procedure consists of an injection of a local anesthetic through a 25-gauge needle into the stellate sympathetic ganglion at the C6 or C7 vertebral levels. An injection into C6 is considered safer because of specific cervical spine anatomy. Ideally, fluoroscopic guidance or ultrasound is used to guide needle insertion.95
A severe drop in blood pressure may be associated with SGBs and is mitigated by IV hydration. Other adverse effects include red eyes, drooping of the eyelids, nasal congestion, hoarseness, difficulty swallowing, a sensation of a “lump” in the throat, and a sensation of warmth or tingling in the arm or hand. Bilateral SGB is contraindicated due to the danger of respiratory arrest.96
Glabellar BT injections
OnabotulinumtoxinA (BT) injection was first approved for therapeutic use in 1989 for eye muscle disorders such as strabismus97 and blepharospasm.98 It was later approved for several other indications, including cosmetic use, hyperhidrosis, migraine prevention, neurogenic bladder disorder, overactive bladder, urinary incontinence, and spasticity.99-104 BT is used off-label for achalasia and sialorrhea.105,106 Its mechanism of action is primarily attributed to muscle paralysis by blocking presynaptic acetylcholine release into neuromuscular junctions.107
Facial BT injections are usually administered for cosmetic purposes, but smoothing forehead wrinkles and frown lines (the glabellar region of the face) both have antidepressant effects.108 BT injections into the glabellar region also demonstrate antidepressant effects, particularly in patients with comorbid migraines and MDD.109 Early case observations supported the independent benefit of the toxin on MDD when the toxin was injected into the glabellar region.110,111 The most frequent protocol involves injections in the procerus and corrugator muscles.
Continue to: The facial feedback/emotional proprioception hypothesis...
The facial feedback/emotional proprioception hypothesis has dominated thinking about the mood-improving effects of BT. The theory is that blocking muscular expression of sadness (especially in the face) interrupts the experience of sadness; therefore, depression subsides.112,113 However, BT injections in the muscles involved in the smile and an expression of positive emotions (lateral part of the musculus orbicularis oculi) have been associated with increased MDD scores.114 Thus, the mechanism clearly involves more than the cosmetic effect, since facial muscle injections in rats also have antidepressant effects.115
The use of progressive muscle relaxation is well-established in psychiatric treatment. The investigated conditions of increased muscle tone, especially torticollis and blepharospasm, are associated with MDD, and it may be speculated that proprioceptive feedback from the affected muscles may be causally involved in this association.116-118 Activity of the corrugator muscle has been positively associated with increased amygdala activity.119 This suggests a potential similar mechanism to that hypothesized for SGB.
Alternatively, BT is commonly used to treat chronic conditions that may contribute to depression; its success in relieving the underlying problem may indirectly relieve MDD. Thus, in a postmarketing safety evaluation of BT, MDD was demonstrated 40% to 88% less often by patients treated with BT for 6 of the 8 conditions and injection sites, such as in spasms and spasticity of arms and legs, torticollis and neck pain, and axilla and palm injections for hyperhidrosis. In a parotid and submandibular glands BT injection subcohort, no patients experienced depressive symptoms.120
Medicinal BT is generally considered safe. The most common adverse effects are hypersensitivity, injection site reactions, and other adverse effects specific to the injection site.121 Additionally, the cosmetic effects are transient, given the nature of the medication.
Trigger point injections
TPIs in the neck and shoulders are frequently used to treat tension headaches and various referred pain locations in the face and arms. Tension and depression frequently overlap in clinical practice.122 Relieving muscle tension (with resulting trigger points) improves muscle function and mood.
Continue to: The higher the number of active...
The higher the number of active trigger points (TPs), the greater the physical burden of headache and the higher the anxiety level. Gender differences in TP prevalence and TPI efficacy have been described in the literature. For example, the number of active TPs seems directly associated with anxiety levels in women but not in men.123 Although TPs appear to be more closely associated with anxiety than depression,124 depression associated with muscle tension does improve with TPIs. European studies have demonstrated a decrease in scores on the Hamilton Depression Rating Scale and Hamilton Anxiety Rating Scale following TPI treatment.125 The effect may be indirect, as when a patient’s pain is relieved, sleep and other psychiatric symptoms improve.126
A randomized controlled trial by Castro Sánchez et al127 demonstrated that dry needling therapy in patients with fibromyalgia syndrome (FMS) showed improvements in pain pressure thresholds, body pain, vitality, and social function, as well as total FMS symptoms, quality of sleep, anxiety, hospital anxiety and depression, general pain intensity, and fatigue.
Myofascial pain syndrome, catastrophizing, and muscle tension are common in patients with depression, anxiety, and somatization. Local TPI therapy aimed at inactivating pain generators is supported by moderate quality evidence. All manner of therapies have been described, including injection of saline, corticosteroids, local anesthetic agents, and dry needling. BT injections in chronic TPs are also practiced, though no specific injection therapy has been reliably shown to be more advantageous than another. The benefits of TPIs may be derived from the needle itself rather than from any specific substance injected. Stimulation of a local twitch response with direct needling of the TP appears of importance. There is no established consensus regarding the number of injection points, frequency of administration, and volume or type of injectate.128
Adverse effects of TPIs relate to the nature of the invasive therapy, with the risk of tissue damage and bleeding. Pneumothorax risk is present with needle insertion at the neck and thorax.129 Patients with diabetes may experience variations in blood sugar control if steroids are used.
Bottom Line
Interventional treatment modalities that may have a role in psychiatric treatment include IV administration of ketamine, aducanumab, lecanemab, brexanolone, magnesium, scopolamine, and clomipramine. Other interventional approaches include stellate ganglion blocks, glabellar botulinum toxin injections, and trigger point injections.
Related Resources
- Dokucu ME, Janicak PG. Nontraditional therapies for treatment-resistant depression. Current Psychiatry. 2021; 20(9):38-43,49. doi:10.12788/cp.0166
- Kim J, Khoury R, Grossberg GT. Botulinum toxin: emerging psychiatric indications. Current Psychiatry. 2018;17(12):8-18.
Drug Brand Names
Aducanumab • Aduhelm
Aripiprazole • Abilify
Aripiprazole lauroxil • Aristada
Brexanolone • Zulresso
Buprenorphine • Sublocade
Citalopram • Celexa
Clomipramine • Anafranil
Diazepam • Valium
Droperidol • Inapsine
Esketamine • Spravato
Fentanyl • Actiq
Fluphenazine decanoate • Modecate
Fluphenazine hydrochloride • Prolixin
Haloperidol decanoate • Haldol decanoate
Haloperidol lactate • Haldol
Ketamine • Ketalar
Lecanemab • Leqembi
Lidocaine • Xylocaine
Lorazepam • Ativan
Loxapine inhaled • Adasuve
Naltrexone • Vivitrol
Magnesium sulfate • Sulfamag
Midazolam • Versed
Olanzapine • Zyprexa
OnabotulinumtoxinA injection • Botox
Paliperidone • Invega Hafyera, Invega Sustenna, Invega Trinza
Rapamycin • Rapamune, Sirolimus
Risperidone • Perseris
Risperidone microspheres • Risperdal Consta, Rykindo
Scopolamine • Hyoscine
Tramadol • Conzip
Vortioxetine • Trintellix
Ziprasidone • Geodon
1. Vincent KM, Ryan M, Palmer E, et al. Interventional psychiatry. Postgrad Med. 2020;132(7):573-574.
2. Allen MH, Feifel D, Lesem MD, et al. Efficacy and safety of loxapine for inhalation in the treatment of agitation in patients with schizophrenia: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2011;72(10):1313-1321.
3. Kwentus J, Riesenberg RA, Marandi M, et al. Rapid acute treatment of agitation in patients with bipolar I disorder: a multicenter, randomized, placebo-controlled clinical trial with inhaled loxapine. Bipolar Disord. 2012;14(1):31-40.
4. Lee JD, Nunes EV Jr, Novo P, et al. Comparative effectiveness of extended-release naltrexone versus buprenorphine-naloxone for opioid relapse prevention (X:BOT): a multicentre, open-label, randomised controlled trial. Lancet. 2018;391(10118):309-318.
5. Haight BR, Learned SM, Laffont CM, et al. Efficacy and safety of a monthly buprenorphine depot injection for opioid use disorder: a multicentre, randomised, double‐blind, placebo‐controlled, phase 3 trial. Lancet. 2019;393(10173):778-790.
6. Andorn A, Graham J, Csernansky J, et al. Monthly extended-release risperidone (RBP-7000) in the treatment of schizophrenia: results from the phase 3 program. J Clin Psychopharmacol. 2019;39(5):428-433.
7. Dundee TW. Twenty-five years of ketamine. A report of an international meeting. Anaesthesia. 1990;45(2):159. doi:10.1111/j.1365-2044.1990.tb14287.x
8. White PF, Way WL, Trevor AJ. Ketamine--its pharmacology and therapeutic uses. Anesthesiology. 1982;56(2):119-136. doi:10.1097/00000542-198202000-00007
9. Zanos P, Gould TD. Mechanisms of ketamine action as an antidepressant. Mol Psychiatry. 2018;23(4):801-811.
10. Molero P, Ramos-Quiroga JA, Martin-Santos R, et al. Antidepressant efficacy and tolerability of ketamine and esketamine: a critical review. CNS Drugs. 2018;32(5):411-420. doi:10.1007/s40263-018-0519-3
11. Williams NR, Heifets BD, Blasey C, et al. Attenuation of antidepressant effects of ketamine by opioid receptor antagonism. Am J Psychiatry. 2018;175(12):1205-1215.
12. Witkin JM, Martin AE, Golani LK, et al. Rapid-acting antidepressants. Adv Pharmacol. 2019;86:47-96.
13. Strayer RJ, Nelson LS. Adverse events associated with ketamine for procedural sedation in adults. Am J Emerg Med. 2008;26(9):985-1028. doi:10.1016/j.ajem.2007.12.005
14. Frye MA, Blier P, Tye SJ. Concomitant benzodiazepine use attenuates ketamine response: implications for large scale study design and clinical development. J Clin Psychopharmacol. 2015;35(3):334-336.
15. Fava M, Freeman MP, Flynn M, et al. Double-blind, placebo-controlled, dose-ranging trial of intravenous ketamine as adjunctive therapy in treatment-resistant depression (TRD). Mol Psychiatry. 2020;25(7):1592-1603.
16. Bahji A, Vazquez GH, Zarate CA Jr. Comparative efficacy of racemic ketamine and esketamine for depression: a systematic review and meta-analysis. J Affect Disord. 2021;278:542-555. Erratum in: J Affect Disord. 2021;281:1001.
17. Brendle M, Robison R, Malone DC. Cost-effectiveness of esketamine nasal spray compared to intravenous ketamine for patients with treatment-resistant depression in the US utilizing clinical trial efficacy and real-world effectiveness estimates. J Affect Disord. 2022;319:388-396.
18. Dhillon S. Aducanumab: first approval. Drugs. 2021;81(12):1437-1443. Erratum in: Drugs. 2021;81(14):1701.
19. van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388(1):9-21. doi:10.1056/NEJMoa2212948
20. Sevigny J, Chiao P, Bussière T, et al. The antibody aducanumab reduces Aβ plaques in Alzheimer’s disease. Nature. 2016;537(7618):50-56. Update in: Nature. 2017;546(7659):564.
21. Fillit H, Green A. Aducanumab and the FDA – where are we now? Nat Rev Neurol. 2021;17(3):129-130.
22. Reardon S. FDA approves Alzheimer’s drug lecanemab amid safety concerns. Nature. 2023;613(7943):227-228. doi:10.1038/d41586-023-00030-3
23. McDade E, Cummings JL, Dhadda S, et al. Lecanemab in patients with early Alzheimer’s disease: detailed results on biomarker, cognitive, and clinical effects from the randomized and open-label extension of the phase 2 proof-of-concept study. Alzheimers Res Ther. 2022;14(1):191. doi:10.1186/s13195-022-01124-2
24. Mintun MA, Lo AC, Evans CD, et al. Donanemab in early Alzheimer’s disease. N Engl J Med. 2021;384(18):1691-1704.
25. Luisi S, Petraglia F, Benedetto C, et al. Serum allopregnanolone levels in pregnant women: changes during pregnancy, at delivery, and in hypertensive patients. J Clin Endocrinol Metab. 2000;85(7):2429-2433.
26. Meltzer-Brody S, Colquhoun H, Riesenberg R, et al. Brexanolone injection in post-partum depression: two multicentre, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet. 2018;392(10152):1058-1070.
27. Powell JG, Garland S, Preston K, et al. Brexanolone (Zulresso): finally, an FDA-approved treatment for postpartum depression. Ann Pharmacother. 2020;54(2):157-163.
28. Patterson R, Krohn H, Richardson E, et al. A brexanolone treatment program at an academic medical center: patient selection, 90-day posttreatment outcomes, and lessons learned. J Acad Consult Liaison Psychiatry. 2022;63(1):14-22.
29. World Health Organization. WHO model list of essential medicines - 22nd list (2021). World Health Organization. September 30, 2021. Accessed April 7, 2023. https://www.who.int/publications/i/item/WHO-MHP-HPS-EML-2021.02
30. Eby GA, Eby KL, Mruk H. Magnesium and major depression. In: Vink R, Nechifor M, eds. Magnesium in the Central Nervous System. University of Adelaide Press; 2011.
31. Plant TM, Zeleznik AJ. Knobil and Neill’s Physiology of Reproduction. 4th ed. Elsevier Inc.; 2015:2503-2550.
32. Sidebotham D, Le Grice IJ. Physiology and pathophysiology. In: Sidebotham D, McKee A, Gillham M, Levy J. Cardiothoracic Critical Care. Elsevier, Inc.; 2007:3-27.
33. Duley L, Gülmezoglu AM, Henderson-Smart DJ, et al. Magnesium sulphate and other anticonvulsants for women with pre-eclampsia. Cochrane Database Syst Rev. 2010;2010(11):CD000025.
34. Emergency supply of medicines. In: British National Formulary. British Medical Association, Royal Pharmaceutical Society; 2015:6. Accessed April 7, 2023. https://www.academia.edu/35076015/british_national_formulary_2015_pdf
35. Kwofie K, Wolfson AB. Intravenous magnesium sulfate for acute asthma exacerbation in children and adults. Am Fam Physician. 2021;103(4):245-246.
36. Patniyot IR, Gelfand AA. Acute treatment therapies for pediatric migraine: a qualitative systematic review. Headache. 2016;56(1):49-70.
37. Wang X, Du X, Yang H, et al. Use of intravenous magnesium sulfate among patients with acute myocardial infarction in China from 2001 to 2015: China PEACE-Retrospective AMI Study. BMJ Open. 2020;10(3):e033269.
38. Karhu E, Atlas SE, Jinrun G, et al. Intravenous infusion of magnesium sulfate is not associated with cardiovascular, liver, kidney, and metabolic toxicity in adults. J Clin Transl Res. 2018;4(1):47-55.
39. Noah L, Pickering G, Mazur A, et al. Impact of magnesium supplementation, in combination with vitamin B6, on stress and magnesium status: secondary data from a randomized controlled trial. Magnes Res. 2020;33(3):45-57.
40. Erstad BL, Cotugno CL. Management of alcohol withdrawal. Am J Health Syst Pharm. 1995;52(7):697-709.
41. Abumaria N, Luo L, Ahn M, et al. Magnesium supplement enhances spatial-context pattern separation and prevents fear overgeneralization. Behav Pharmacol. 2013;24(4):255-263.
42. Kirov GK, Tsachev KN. Magnesium, schizophrenia and manic-depressive disease. Neuropsychobiology. 1990;23(2):79-81.
43. Botturi A, Ciappolino V, Delvecchio G, et al. The role and the effect of magnesium in mental disorders: a systematic review. Nutrients. 2020;12(6):1661.
44. Kirkland AE, Sarlo GL, Holton KF. The role of magnesium in neurological disorders. Nutrients. 2018;10(6):730.
45. Magnesium sulfate intravenous side effects by likelihood and severity. WebMD. Accessed April 9, 2023. https://www.webmd.com/drugs/2/drug-149570/magnesium-sulfate-intravenous/details/list-sideeffects
46. Scopolamine base transdermal system – uses, side effects, and more. WebMD. Accessed April 9, 2023. https://www.webmd.com/drugs/2/drug-14032/scopolamine-transdermal/details
47. Bolden C, Cusack B, Richelson E. Antagonism by antimuscarinic and neuroleptic compounds at the five cloned human muscarinic cholinergic receptors expressed in Chinese hamster ovary cells. J Pharmacol Exp Ther. 1992;260(2):576-580.
48. Janowsky DS, el-Yousef MK, Davis JM, et al. A cholinergic-adrenergic hypothesis of mania and depression. Lancet. 1972;2(7778):632-635.
49. Janowsky DS, Risch SC, Gillin JC. Adrenergic-cholinergic balance and the treatment of affective disorders. Prog Neuropsychopharmacol Biol Psychiatry. 1983;7(2-3):297-307.
50. Gershon S, Shaw FH. Psychiatric sequelae of chronic exposure to organophosphorous insecticides. Lancet. 1972;1(7191):1371-1374.
51. Davis KL, Berger PA, Hollister LE, et al. Physostigmine in mania. Arch Gen Psychiatry. 1978;35(1):119-122.
52. Wang JC, Hinrichs AL, Stock H, et al. Evidence of common and specific genetic effects: association of the muscarinic acetylcholine receptor M2 (CHRM2) gene with alcohol dependence and major depressive syndrome. Hum Mol Genet. 2004;13(17):1903-1911.
53. Brown RG. Effects of antidepressants and anticholinergics in a mouse “behavioral despair” test. Eur J Pharmacol. 1979;58(3):331-334.
54. Porsolt RD, Le Pichon M, Jalfre M. Depression: a new animal model sensitive to antidepressant treatments. Nature. 1977;266(5604):730-732.
55. Ji CX, Zhang JJ. Effect of scopolamine on depression in mice. Abstract in English. Yao Xue Xue Bao. 2011;46(4):400-405.
56. Furey ML, Drevets WC. Antidepressant efficacy of the antimuscarinic drug scopolamine: a randomized, placebo-controlled clinical trial. Arch Gen Psychiatry. 2006;63(10):1121-1129.
57. Drevets WC, Furey ML. Replication of scopolamine’s antidepressant efficacy in major depressive disorder: a randomized, placebo-controlled clinical trial. Biol Psychiatry. 2010;67(5):432-438.
58. Furey ML, Khanna A, Hoffman EM, et al. Scopolamine produces larger antidepressant and antianxiety effects in women than in men. Neuropsychopharmacology. 2010;35(12):2479-2488.
59. Gibbs RB, Gabor R, Cox T, et al. Effects of raloxifene and estradiol on hippocampal acetylcholine release and spatial learning in the rat. Psychoneuroendocrinology. 2004;29(6):741-748.
60. Pongrac JL, Gibbs RB, Defranco DB. Estrogen-mediated regulation of cholinergic expression in basal forebrain neurons requires extracellular-signal-regulated kinase activity. Neuroscience. 2004;124(4):809-816.
61. Daniel JM, Dohanich GP. Acetylcholine mediates the estrogen-induced increase in NMDA receptor binding in CA1 of the hippocampus and the associated improvement in working memory. J Neurosci. 2001;21(17):6949-6956.
62. Gerhard DM, Wohleb ES, Duman RS. Emerging treatment mechanisms for depression: focus on glutamate and synaptic plasticity. Drug Discov Today. 2016;21(3):454-464.
63. Voderholzer U. Sleep deprivation and antidepressant treatment. Dialogues Clin Neurosci. 2003;5(4):366-369.
64. Hasselmann H. Scopolamine and depression: a role for muscarinic antagonism? CNS Neurol Disord Drug Targets. 2014;13(4):673-683.
65. Transderm scopolamine [prescribing information]. Warren, NJ: GSK Consumer Healthcare; 2019.
66. Jaffe RJ, Novakovic V, Peselow ED. Scopolamine as an antidepressant: a systematic review. Clin Neuropharmacol. 2013;36(1):24-26.
67. Karameh WK, Khani M. Intravenous clomipramine for treatment-resistant obsessive-compulsive disorder. Int J Neuropsychopharmacol. 2015;19(2):pyv084.
68. Andrews ET, Beattie RM, Tighe MP. Functional abdominal pain: what clinicians need to know. Arch Dis Child. 2020;105(10):938-944. doi:10.1136/archdischild-2020-318825
69. Aliane V, Pérez S, Bohren Y, et al. Key role of striatal cholinergic interneurons in processes leading to arrest of motor stereotypies. Brain. 2011;134(Pt 1):110-118. doi:10.1093/brain/awq285
70. Tzavara ET, Bymaster FP, Davis RJ, et al. M4 muscarinic receptors regulate the dynamics of cholinergic and dopaminergic neurotransmission: relevance to the pathophysiology and treatment of related CNS pathologies. FASEB J. 2004;18(12):1410-1412. doi:10.1096/fj.04-1575fje
71. Korczyn AD, Kish I. The mechanism of imipramine in enuresis nocturna. Clin Exp Pharmacol Physiol. 1979;6(1):31-35. doi:10.1111/j.1440-1681.1979.tb00004.x
72. Trimble MR. Worldwide use of clomipramine. J Clin Psychiatry. 1990;51(Suppl):51-54; discussion 55-58.
73. Gong W, Zhang S, Zong Y, et al. Involvement of the microglial NLRP3 inflammasome in the anti-inflammatory effect of the antidepressant clomipramine. J Affect Disord. 2019;254:15-25.
74. Piwowarska J, Wrzosek M, Radziwon’-Zaleska M. Serum cortisol concentration in patients with major depression after treatment with clomipramine. Pharmacol Rep. 2009;61(4):604-611.
75. Danish University Antidepressant Group (DUAG). Clomipramine dose-effect study in patients with depression: clinical end points and pharmacokinetics. Clin Pharmacol Ther. 1999;66(2):152-165.
76. Moukaddam NJ, Hirschfeld RMA. Intravenous antidepressants: a review. Depress Anxiety. 2004;19(1):1-9.
77. Gerretsen P, Pollock BG. Rediscovering adverse anticholinergic effects. J Clin Psychiatry. 2011;72(6):869-870. doi:10.4088/JCP.11ac07093
78. Thomas SJ, Shin M, McInnis MG, et al. Combination therapy with monoamine oxidase inhibitors and other antidepressants or stimulants: strategies for the management of treatment-resistant depression. Pharmacotherapy. 2015;35(4):433-449. doi:10.1002/phar.1576
79. Robles LA. Serotonin syndrome induced by fentanyl in a child: case report. Clin Neuropharmacol. 2015;38(5):206-208. doi:10.1097/WNF.0000000000000100
80. Fallon BA, Liebowitz MR, Campeas R, et al. Intravenous clomipramine for obsessive-compulsive disorder refractory to oral clomipramine: a placebo-controlled study. Arch Gen Psychiatry. 1998;55(10):918-924.
81. Vieta E, Florea I, Schmidt SN, et al. Intravenous vortioxetine to accelerate onset of effect in major depressive disorder: a 2-week, randomized, double-blind, placebo-controlled study. Int Clin Psychopharmacol. 2019;34(4):153-160.
82. Kasper S, Müller-Spahn F. Intravenous antidepressant treatment: focus on citalopram. Eur Arch Psychiatry Clin Neurosci. 2002;252(3):105-109.
83. Togay B, El-Mallakh RS. Posttraumatic stress disorder: from pathophysiology to pharmacology. Current Psychiatry. 2020;19(5):33-39.
84. Adhikari A, Lerner TN, Finkelstein J, et al. Basomedial amygdala mediates top-down control of anxiety and fear. Nature. 2015;527(7577):179-185. doi:10.1038/nature15698
85. Lipov E. In search of an effective treatment for combat-related post-traumatic stress disorder (PTSD): can the stellate ganglion block be the answer? Pain Pract. 2010;10(4):265-266.
86. Lipov E, Ritchie EC. A review of the use of stellate ganglion block in the treatment of PTSD. Curr Psychiatry Rep. 2015;17(8):599.
87. Olmsted KLR, Bartoszek M, McLean B, et al. Effect of stellate ganglion block treatment on posttraumatic stress disorder symptoms: a randomized clinical trial. JAMA Psychiatry. 2020;77(2):130-138.
88. Lipov E, Candido K. The successful use of left-sided stellate ganglion block in patients that fail to respond to right-sided stellate ganglion block for the treatment of post-traumatic stress disorder symptoms: a retrospective analysis of 205 patients. Mil Med. 2021;186(11-12):319-320.
89. Li Y, Loshak H. Stellate ganglion block for the treatment of post-traumatic stress disorder, depression, and anxiety. Canadian J Health Technol. 2021;1(3):1-30.
90. Kerzner J, Liu H, Demchenko I, et al. Stellate ganglion block for psychiatric disorders: a systematic review of the clinical research landscape. Chronic Stress (Thousand Oaks). 2021;5:24705470211055176.
91. Wie C, Gupta R, Maloney J, et al. Interventional modalities to treat complex regional pain syndrome. Curr Pain Headache Rep. 2021;25(2):10. doi:10.1007/s11916-020-00904-5
92. Chaturvedi A, Dash HH. Sympathetic blockade for the relief of chronic pain. J Indian Med Assoc. 2001;99(12):698-703.
93. Chester M, Hammond C. Leach A. Long-term benefits of stellate ganglion block in severe chronic refractory angina. Pain. 2000;87(1):103-105. doi:10.1016/S0304-3959(00)00270-0
94. Jeon Y. Therapeutic potential of stellate ganglion block in orofacial pain: a mini review. J Dent Anesth Pain Med. 2016;16(3):159-163. doi:10.17245/jdapm.2016.16.3.159
95. Shan HH, Chen HF, Ni Y, et al. Effects of stellate ganglion block through different approaches under guidance of ultrasound. Front Surg. 2022;8:797793. doi:10.3389/fsurg.2021.797793
96. Goel V, Patwardhan AM, Ibrahim M, et al. Complications associated with stellate ganglion nerve block: a systematic review. Reg Anesth Pain Med. 2019;rapm-2018-100127. doi:10.1136/rapm-2018-100127
97. Rowe FJ, Noonan CP. Botulinum toxin for the treatment of strabismus. Cochrane Database Syst Rev. 2017;3(3):CD006499.
98. Roggenkämper P, Jost WH, Bihari K, et al. Efficacy and safety of a new botulinum toxin type A free of complexing proteins in the treatment of blepharospasm. J Neural Transm (Vienna). 2006;113(3):303-312.
99. Heckmann M, Ceballos-Baumann AO, Plewig G; Hyperhidrosis Study Group. Botulinum toxin A for axillary hyperhidrosis (excessive sweating). N Engl J Med. 2001;344(7):488-493.
100. Carruthers JA, Lowe NJ, Menter MA, et al. A multicenter, double-blind, randomized, placebo-controlled study of the efficacy and safety of botulinum toxin type A in the treatment of glabellar lines. J Am Acad Dermatol. 2002;46(6):840-849.
101. Schurch B, de Sèze M, Denys P, et al. Botulinum toxin type A is a safe and effective treatment for neurogenic urinary incontinence: results of a single treatment, randomized, placebo controlled 6-month study. J Urol. 2005;174:196–200.
102. Aurora SK, Winner P, Freeman MC, et al. OnabotulinumtoxinA for treatment of chronic migraine: Pooled analyses of the 56-week PREEMPT clinical program. Headache. 2011;51(9):1358-1373.
103. Dashtipour K, Chen JJ, Walker HW, et al. Systematic literature review of abobotulinumtoxinA in clinical trials for adult upper limb spasticity. Am J Phys Med Rehabil. 2015;94(3):229-238.
104. Nitti VW, Dmochowski R, Herschorn S, et al. OnabotulinumtoxinA for the treatment of patients with overactive bladder and urinary incontinence: results of a phase 3, randomized, placebo-controlled trial. J Urol. 2017;197(2S):S216-S223.
105. Jongerius PH, van den Hoogen FJA, van Limbeek J, et al. Effect of botulinum toxin in the treatment of drooling: a controlled clinical trial. Pediatrics. 2004;114(3):620-627.
106. Zaninotto, G. Annese V, Costantini M, et al. Randomized controlled trial of botulinum toxin versus laparoscopic heller myotomy for esophageal achalasia. Ann Surg. 2004;239(3):364-370.
107. Dressler D, Adib Saberi F. Botulinum toxin: mechanisms of action. Eur Neurol. 2005;53:3-9.
108. Lewis MB, Bowler PJ. Botulinum toxin cosmetic therapy correlates with a more positive mood. J Cosmet Dermatol. 2009;8(1):24-26.
109. Affatato O, Moulin TC, Pisanu C, et al. High efficacy of onabotulinumtoxinA treatment in patients with comorbid migraine and depression: a meta-analysis. J Transl Med. 2021;19(1):133.
110. Finzi E, Wasserman E. Treatment of depression with botulinum toxin A: a case series. Dermatol Surg. 2006;32(5):645-649; discussion 649-650.
111. Schulze J, Neumann I, Magid M, et al. Botulinum toxin for the management of depression: an updated review of the evidence and meta-analysis. J Psychiatr Res. 2021;135:332-340.
112. Finzi E, Rosenthal NE. Emotional proprioception: treatment of depression with afferent facial feedback. J Psychiatr Res. 2016;80:93-96.
113. Söderkvist S, Ohlén K, Dimberg U. How the experience of emotion is modulated by facial feedback. J Nonverbal Behav. 2018;42(1):129-151.
114. Lewis, MB. The interactions between botulinum-toxin-based facial treatments and embodied emotions. Sci Rep. 2018;8(1):14720.
115. Li Y, Liu J, Liu X, et al. Antidepressant-like action of single facial injection of botulinum neurotoxin A is associated with augmented 5-HT levels and BDNF/ERK/CREB pathways in mouse brain. Neurosci Bull. 2019;35(4):661-672. Erratum in: Neurosci Bull. 2019;35(4):779-780.
116. Gündel H, Wolf A, Xidara V, et al. High psychiatric comorbidity in spasmodic torticollis: a controlled study. J Nerv Ment Dis. 2003;191(7):465-473.
117. Hall TA, McGwin G Jr, Searcey K, et al. Health-related quality of life and psychosocial characteristics of patients with benign essential blepharospasm. Arch Ophthalmol. 2006;124(1):116-119.
118. Ceylan D, Erer S, Zarifog˘lu M, et al. Evaluation of anxiety and depression scales and quality of life in cervical dystonia patients on botulinum toxin therapy and their relatives. Neurol Sci. 2019;40(4):725-731.
119. Heller AS, Lapate RC, Mayer KE, et al. The face of negative affect: trial-by-trial corrugator responses to negative pictures are positively associated with amygdala and negatively associated with ventromedial prefrontal cortex activity. J Cogn Neurosci. 2014;26(9):2102-2110.
120. Makunts T, Wollmer MA, Abagyan R. Postmarketing safety surveillance data reveals antidepressant effects of botulinum toxin across various indications and injection sites. Sci Rep. 2020;10(1):12851.
121. Ahsanuddin S, Roy S, Nasser W, et al. Adverse events associated with botox as reported in a Food and Drug Administration database. Aesthetic Plast Surg. 2021;45(3):1201-1209. doi:10.1007/s00266-020-02027-z
122. Kashif M, Tahir S, Ashfaq F, et al. Association of myofascial trigger points in neck and shoulder region with depression, anxiety, and stress among university students. J Pak Med Assoc. 2021;71(9):2139-2142.
123. Cigarán-Méndez M, Jiménez-Antona C, Parás-Bravo P, et al. Active trigger points are associated with anxiety and widespread pressure pain sensitivity in women, but not men, with tension type headache. Pain Pract. 2019;19(5):522-529.
124. Palacios-Ceña M, Castaldo M, Wang K, et al. Relationship of active trigger points with related disability and anxiety in people with tension-type headache. Medicine (Baltimore). 2017;96(13):e6548.
125. Karadas Ö, Inan LE, Ulas Ü, et al. Efficacy of local lidocaine application on anxiety and depression and its curative effect on patients with chronic tension-type headache. Eur Neurol. 2013;70(1-2):95-101.
126. Gerwin RD. Classification, epidemiology and natural history of myofascial pain syndrome. Curr Pain Headache Rep. 2001;5(5):412-420.
127. Castro Sánchez AM, García López H, Fernández Sánchez M, et al. Improvement in clinical outcomes after dry needling versus myofascial release on pain pressure thresholds, quality of life, fatigue, pain intensity, quality of sleep, anxiety, and depression in patients with fibromyalgia syndrome. Disabil Rehabil. 2019;41(19):2235-2246.
128. Healy GM, Finn DP, O’Gorman DA, et al. Pretreatment anxiety and pain acceptance are associated with response to trigger point injection therapy for chronic myofascial pain. Pain Med. 2015;16(10):1955-1966.
129. Morjaria JB, Lakshminarayana UB, Liu-Shiu-Cheong P, et al. Pneumothorax: a tale of pain or spontaneity. Ther Adv Chronic Dis. 2014;5(6):269-273.
1. Vincent KM, Ryan M, Palmer E, et al. Interventional psychiatry. Postgrad Med. 2020;132(7):573-574.
2. Allen MH, Feifel D, Lesem MD, et al. Efficacy and safety of loxapine for inhalation in the treatment of agitation in patients with schizophrenia: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2011;72(10):1313-1321.
3. Kwentus J, Riesenberg RA, Marandi M, et al. Rapid acute treatment of agitation in patients with bipolar I disorder: a multicenter, randomized, placebo-controlled clinical trial with inhaled loxapine. Bipolar Disord. 2012;14(1):31-40.
4. Lee JD, Nunes EV Jr, Novo P, et al. Comparative effectiveness of extended-release naltrexone versus buprenorphine-naloxone for opioid relapse prevention (X:BOT): a multicentre, open-label, randomised controlled trial. Lancet. 2018;391(10118):309-318.
5. Haight BR, Learned SM, Laffont CM, et al. Efficacy and safety of a monthly buprenorphine depot injection for opioid use disorder: a multicentre, randomised, double‐blind, placebo‐controlled, phase 3 trial. Lancet. 2019;393(10173):778-790.
6. Andorn A, Graham J, Csernansky J, et al. Monthly extended-release risperidone (RBP-7000) in the treatment of schizophrenia: results from the phase 3 program. J Clin Psychopharmacol. 2019;39(5):428-433.
7. Dundee TW. Twenty-five years of ketamine. A report of an international meeting. Anaesthesia. 1990;45(2):159. doi:10.1111/j.1365-2044.1990.tb14287.x
8. White PF, Way WL, Trevor AJ. Ketamine--its pharmacology and therapeutic uses. Anesthesiology. 1982;56(2):119-136. doi:10.1097/00000542-198202000-00007
9. Zanos P, Gould TD. Mechanisms of ketamine action as an antidepressant. Mol Psychiatry. 2018;23(4):801-811.
10. Molero P, Ramos-Quiroga JA, Martin-Santos R, et al. Antidepressant efficacy and tolerability of ketamine and esketamine: a critical review. CNS Drugs. 2018;32(5):411-420. doi:10.1007/s40263-018-0519-3
11. Williams NR, Heifets BD, Blasey C, et al. Attenuation of antidepressant effects of ketamine by opioid receptor antagonism. Am J Psychiatry. 2018;175(12):1205-1215.
12. Witkin JM, Martin AE, Golani LK, et al. Rapid-acting antidepressants. Adv Pharmacol. 2019;86:47-96.
13. Strayer RJ, Nelson LS. Adverse events associated with ketamine for procedural sedation in adults. Am J Emerg Med. 2008;26(9):985-1028. doi:10.1016/j.ajem.2007.12.005
14. Frye MA, Blier P, Tye SJ. Concomitant benzodiazepine use attenuates ketamine response: implications for large scale study design and clinical development. J Clin Psychopharmacol. 2015;35(3):334-336.
15. Fava M, Freeman MP, Flynn M, et al. Double-blind, placebo-controlled, dose-ranging trial of intravenous ketamine as adjunctive therapy in treatment-resistant depression (TRD). Mol Psychiatry. 2020;25(7):1592-1603.
16. Bahji A, Vazquez GH, Zarate CA Jr. Comparative efficacy of racemic ketamine and esketamine for depression: a systematic review and meta-analysis. J Affect Disord. 2021;278:542-555. Erratum in: J Affect Disord. 2021;281:1001.
17. Brendle M, Robison R, Malone DC. Cost-effectiveness of esketamine nasal spray compared to intravenous ketamine for patients with treatment-resistant depression in the US utilizing clinical trial efficacy and real-world effectiveness estimates. J Affect Disord. 2022;319:388-396.
18. Dhillon S. Aducanumab: first approval. Drugs. 2021;81(12):1437-1443. Erratum in: Drugs. 2021;81(14):1701.
19. van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388(1):9-21. doi:10.1056/NEJMoa2212948
20. Sevigny J, Chiao P, Bussière T, et al. The antibody aducanumab reduces Aβ plaques in Alzheimer’s disease. Nature. 2016;537(7618):50-56. Update in: Nature. 2017;546(7659):564.
21. Fillit H, Green A. Aducanumab and the FDA – where are we now? Nat Rev Neurol. 2021;17(3):129-130.
22. Reardon S. FDA approves Alzheimer’s drug lecanemab amid safety concerns. Nature. 2023;613(7943):227-228. doi:10.1038/d41586-023-00030-3
23. McDade E, Cummings JL, Dhadda S, et al. Lecanemab in patients with early Alzheimer’s disease: detailed results on biomarker, cognitive, and clinical effects from the randomized and open-label extension of the phase 2 proof-of-concept study. Alzheimers Res Ther. 2022;14(1):191. doi:10.1186/s13195-022-01124-2
24. Mintun MA, Lo AC, Evans CD, et al. Donanemab in early Alzheimer’s disease. N Engl J Med. 2021;384(18):1691-1704.
25. Luisi S, Petraglia F, Benedetto C, et al. Serum allopregnanolone levels in pregnant women: changes during pregnancy, at delivery, and in hypertensive patients. J Clin Endocrinol Metab. 2000;85(7):2429-2433.
26. Meltzer-Brody S, Colquhoun H, Riesenberg R, et al. Brexanolone injection in post-partum depression: two multicentre, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet. 2018;392(10152):1058-1070.
27. Powell JG, Garland S, Preston K, et al. Brexanolone (Zulresso): finally, an FDA-approved treatment for postpartum depression. Ann Pharmacother. 2020;54(2):157-163.
28. Patterson R, Krohn H, Richardson E, et al. A brexanolone treatment program at an academic medical center: patient selection, 90-day posttreatment outcomes, and lessons learned. J Acad Consult Liaison Psychiatry. 2022;63(1):14-22.
29. World Health Organization. WHO model list of essential medicines - 22nd list (2021). World Health Organization. September 30, 2021. Accessed April 7, 2023. https://www.who.int/publications/i/item/WHO-MHP-HPS-EML-2021.02
30. Eby GA, Eby KL, Mruk H. Magnesium and major depression. In: Vink R, Nechifor M, eds. Magnesium in the Central Nervous System. University of Adelaide Press; 2011.
31. Plant TM, Zeleznik AJ. Knobil and Neill’s Physiology of Reproduction. 4th ed. Elsevier Inc.; 2015:2503-2550.
32. Sidebotham D, Le Grice IJ. Physiology and pathophysiology. In: Sidebotham D, McKee A, Gillham M, Levy J. Cardiothoracic Critical Care. Elsevier, Inc.; 2007:3-27.
33. Duley L, Gülmezoglu AM, Henderson-Smart DJ, et al. Magnesium sulphate and other anticonvulsants for women with pre-eclampsia. Cochrane Database Syst Rev. 2010;2010(11):CD000025.
34. Emergency supply of medicines. In: British National Formulary. British Medical Association, Royal Pharmaceutical Society; 2015:6. Accessed April 7, 2023. https://www.academia.edu/35076015/british_national_formulary_2015_pdf
35. Kwofie K, Wolfson AB. Intravenous magnesium sulfate for acute asthma exacerbation in children and adults. Am Fam Physician. 2021;103(4):245-246.
36. Patniyot IR, Gelfand AA. Acute treatment therapies for pediatric migraine: a qualitative systematic review. Headache. 2016;56(1):49-70.
37. Wang X, Du X, Yang H, et al. Use of intravenous magnesium sulfate among patients with acute myocardial infarction in China from 2001 to 2015: China PEACE-Retrospective AMI Study. BMJ Open. 2020;10(3):e033269.
38. Karhu E, Atlas SE, Jinrun G, et al. Intravenous infusion of magnesium sulfate is not associated with cardiovascular, liver, kidney, and metabolic toxicity in adults. J Clin Transl Res. 2018;4(1):47-55.
39. Noah L, Pickering G, Mazur A, et al. Impact of magnesium supplementation, in combination with vitamin B6, on stress and magnesium status: secondary data from a randomized controlled trial. Magnes Res. 2020;33(3):45-57.
40. Erstad BL, Cotugno CL. Management of alcohol withdrawal. Am J Health Syst Pharm. 1995;52(7):697-709.
41. Abumaria N, Luo L, Ahn M, et al. Magnesium supplement enhances spatial-context pattern separation and prevents fear overgeneralization. Behav Pharmacol. 2013;24(4):255-263.
42. Kirov GK, Tsachev KN. Magnesium, schizophrenia and manic-depressive disease. Neuropsychobiology. 1990;23(2):79-81.
43. Botturi A, Ciappolino V, Delvecchio G, et al. The role and the effect of magnesium in mental disorders: a systematic review. Nutrients. 2020;12(6):1661.
44. Kirkland AE, Sarlo GL, Holton KF. The role of magnesium in neurological disorders. Nutrients. 2018;10(6):730.
45. Magnesium sulfate intravenous side effects by likelihood and severity. WebMD. Accessed April 9, 2023. https://www.webmd.com/drugs/2/drug-149570/magnesium-sulfate-intravenous/details/list-sideeffects
46. Scopolamine base transdermal system – uses, side effects, and more. WebMD. Accessed April 9, 2023. https://www.webmd.com/drugs/2/drug-14032/scopolamine-transdermal/details
47. Bolden C, Cusack B, Richelson E. Antagonism by antimuscarinic and neuroleptic compounds at the five cloned human muscarinic cholinergic receptors expressed in Chinese hamster ovary cells. J Pharmacol Exp Ther. 1992;260(2):576-580.
48. Janowsky DS, el-Yousef MK, Davis JM, et al. A cholinergic-adrenergic hypothesis of mania and depression. Lancet. 1972;2(7778):632-635.
49. Janowsky DS, Risch SC, Gillin JC. Adrenergic-cholinergic balance and the treatment of affective disorders. Prog Neuropsychopharmacol Biol Psychiatry. 1983;7(2-3):297-307.
50. Gershon S, Shaw FH. Psychiatric sequelae of chronic exposure to organophosphorous insecticides. Lancet. 1972;1(7191):1371-1374.
51. Davis KL, Berger PA, Hollister LE, et al. Physostigmine in mania. Arch Gen Psychiatry. 1978;35(1):119-122.
52. Wang JC, Hinrichs AL, Stock H, et al. Evidence of common and specific genetic effects: association of the muscarinic acetylcholine receptor M2 (CHRM2) gene with alcohol dependence and major depressive syndrome. Hum Mol Genet. 2004;13(17):1903-1911.
53. Brown RG. Effects of antidepressants and anticholinergics in a mouse “behavioral despair” test. Eur J Pharmacol. 1979;58(3):331-334.
54. Porsolt RD, Le Pichon M, Jalfre M. Depression: a new animal model sensitive to antidepressant treatments. Nature. 1977;266(5604):730-732.
55. Ji CX, Zhang JJ. Effect of scopolamine on depression in mice. Abstract in English. Yao Xue Xue Bao. 2011;46(4):400-405.
56. Furey ML, Drevets WC. Antidepressant efficacy of the antimuscarinic drug scopolamine: a randomized, placebo-controlled clinical trial. Arch Gen Psychiatry. 2006;63(10):1121-1129.
57. Drevets WC, Furey ML. Replication of scopolamine’s antidepressant efficacy in major depressive disorder: a randomized, placebo-controlled clinical trial. Biol Psychiatry. 2010;67(5):432-438.
58. Furey ML, Khanna A, Hoffman EM, et al. Scopolamine produces larger antidepressant and antianxiety effects in women than in men. Neuropsychopharmacology. 2010;35(12):2479-2488.
59. Gibbs RB, Gabor R, Cox T, et al. Effects of raloxifene and estradiol on hippocampal acetylcholine release and spatial learning in the rat. Psychoneuroendocrinology. 2004;29(6):741-748.
60. Pongrac JL, Gibbs RB, Defranco DB. Estrogen-mediated regulation of cholinergic expression in basal forebrain neurons requires extracellular-signal-regulated kinase activity. Neuroscience. 2004;124(4):809-816.
61. Daniel JM, Dohanich GP. Acetylcholine mediates the estrogen-induced increase in NMDA receptor binding in CA1 of the hippocampus and the associated improvement in working memory. J Neurosci. 2001;21(17):6949-6956.
62. Gerhard DM, Wohleb ES, Duman RS. Emerging treatment mechanisms for depression: focus on glutamate and synaptic plasticity. Drug Discov Today. 2016;21(3):454-464.
63. Voderholzer U. Sleep deprivation and antidepressant treatment. Dialogues Clin Neurosci. 2003;5(4):366-369.
64. Hasselmann H. Scopolamine and depression: a role for muscarinic antagonism? CNS Neurol Disord Drug Targets. 2014;13(4):673-683.
65. Transderm scopolamine [prescribing information]. Warren, NJ: GSK Consumer Healthcare; 2019.
66. Jaffe RJ, Novakovic V, Peselow ED. Scopolamine as an antidepressant: a systematic review. Clin Neuropharmacol. 2013;36(1):24-26.
67. Karameh WK, Khani M. Intravenous clomipramine for treatment-resistant obsessive-compulsive disorder. Int J Neuropsychopharmacol. 2015;19(2):pyv084.
68. Andrews ET, Beattie RM, Tighe MP. Functional abdominal pain: what clinicians need to know. Arch Dis Child. 2020;105(10):938-944. doi:10.1136/archdischild-2020-318825
69. Aliane V, Pérez S, Bohren Y, et al. Key role of striatal cholinergic interneurons in processes leading to arrest of motor stereotypies. Brain. 2011;134(Pt 1):110-118. doi:10.1093/brain/awq285
70. Tzavara ET, Bymaster FP, Davis RJ, et al. M4 muscarinic receptors regulate the dynamics of cholinergic and dopaminergic neurotransmission: relevance to the pathophysiology and treatment of related CNS pathologies. FASEB J. 2004;18(12):1410-1412. doi:10.1096/fj.04-1575fje
71. Korczyn AD, Kish I. The mechanism of imipramine in enuresis nocturna. Clin Exp Pharmacol Physiol. 1979;6(1):31-35. doi:10.1111/j.1440-1681.1979.tb00004.x
72. Trimble MR. Worldwide use of clomipramine. J Clin Psychiatry. 1990;51(Suppl):51-54; discussion 55-58.
73. Gong W, Zhang S, Zong Y, et al. Involvement of the microglial NLRP3 inflammasome in the anti-inflammatory effect of the antidepressant clomipramine. J Affect Disord. 2019;254:15-25.
74. Piwowarska J, Wrzosek M, Radziwon’-Zaleska M. Serum cortisol concentration in patients with major depression after treatment with clomipramine. Pharmacol Rep. 2009;61(4):604-611.
75. Danish University Antidepressant Group (DUAG). Clomipramine dose-effect study in patients with depression: clinical end points and pharmacokinetics. Clin Pharmacol Ther. 1999;66(2):152-165.
76. Moukaddam NJ, Hirschfeld RMA. Intravenous antidepressants: a review. Depress Anxiety. 2004;19(1):1-9.
77. Gerretsen P, Pollock BG. Rediscovering adverse anticholinergic effects. J Clin Psychiatry. 2011;72(6):869-870. doi:10.4088/JCP.11ac07093
78. Thomas SJ, Shin M, McInnis MG, et al. Combination therapy with monoamine oxidase inhibitors and other antidepressants or stimulants: strategies for the management of treatment-resistant depression. Pharmacotherapy. 2015;35(4):433-449. doi:10.1002/phar.1576
79. Robles LA. Serotonin syndrome induced by fentanyl in a child: case report. Clin Neuropharmacol. 2015;38(5):206-208. doi:10.1097/WNF.0000000000000100
80. Fallon BA, Liebowitz MR, Campeas R, et al. Intravenous clomipramine for obsessive-compulsive disorder refractory to oral clomipramine: a placebo-controlled study. Arch Gen Psychiatry. 1998;55(10):918-924.
81. Vieta E, Florea I, Schmidt SN, et al. Intravenous vortioxetine to accelerate onset of effect in major depressive disorder: a 2-week, randomized, double-blind, placebo-controlled study. Int Clin Psychopharmacol. 2019;34(4):153-160.
82. Kasper S, Müller-Spahn F. Intravenous antidepressant treatment: focus on citalopram. Eur Arch Psychiatry Clin Neurosci. 2002;252(3):105-109.
83. Togay B, El-Mallakh RS. Posttraumatic stress disorder: from pathophysiology to pharmacology. Current Psychiatry. 2020;19(5):33-39.
84. Adhikari A, Lerner TN, Finkelstein J, et al. Basomedial amygdala mediates top-down control of anxiety and fear. Nature. 2015;527(7577):179-185. doi:10.1038/nature15698
85. Lipov E. In search of an effective treatment for combat-related post-traumatic stress disorder (PTSD): can the stellate ganglion block be the answer? Pain Pract. 2010;10(4):265-266.
86. Lipov E, Ritchie EC. A review of the use of stellate ganglion block in the treatment of PTSD. Curr Psychiatry Rep. 2015;17(8):599.
87. Olmsted KLR, Bartoszek M, McLean B, et al. Effect of stellate ganglion block treatment on posttraumatic stress disorder symptoms: a randomized clinical trial. JAMA Psychiatry. 2020;77(2):130-138.
88. Lipov E, Candido K. The successful use of left-sided stellate ganglion block in patients that fail to respond to right-sided stellate ganglion block for the treatment of post-traumatic stress disorder symptoms: a retrospective analysis of 205 patients. Mil Med. 2021;186(11-12):319-320.
89. Li Y, Loshak H. Stellate ganglion block for the treatment of post-traumatic stress disorder, depression, and anxiety. Canadian J Health Technol. 2021;1(3):1-30.
90. Kerzner J, Liu H, Demchenko I, et al. Stellate ganglion block for psychiatric disorders: a systematic review of the clinical research landscape. Chronic Stress (Thousand Oaks). 2021;5:24705470211055176.
91. Wie C, Gupta R, Maloney J, et al. Interventional modalities to treat complex regional pain syndrome. Curr Pain Headache Rep. 2021;25(2):10. doi:10.1007/s11916-020-00904-5
92. Chaturvedi A, Dash HH. Sympathetic blockade for the relief of chronic pain. J Indian Med Assoc. 2001;99(12):698-703.
93. Chester M, Hammond C. Leach A. Long-term benefits of stellate ganglion block in severe chronic refractory angina. Pain. 2000;87(1):103-105. doi:10.1016/S0304-3959(00)00270-0
94. Jeon Y. Therapeutic potential of stellate ganglion block in orofacial pain: a mini review. J Dent Anesth Pain Med. 2016;16(3):159-163. doi:10.17245/jdapm.2016.16.3.159
95. Shan HH, Chen HF, Ni Y, et al. Effects of stellate ganglion block through different approaches under guidance of ultrasound. Front Surg. 2022;8:797793. doi:10.3389/fsurg.2021.797793
96. Goel V, Patwardhan AM, Ibrahim M, et al. Complications associated with stellate ganglion nerve block: a systematic review. Reg Anesth Pain Med. 2019;rapm-2018-100127. doi:10.1136/rapm-2018-100127
97. Rowe FJ, Noonan CP. Botulinum toxin for the treatment of strabismus. Cochrane Database Syst Rev. 2017;3(3):CD006499.
98. Roggenkämper P, Jost WH, Bihari K, et al. Efficacy and safety of a new botulinum toxin type A free of complexing proteins in the treatment of blepharospasm. J Neural Transm (Vienna). 2006;113(3):303-312.
99. Heckmann M, Ceballos-Baumann AO, Plewig G; Hyperhidrosis Study Group. Botulinum toxin A for axillary hyperhidrosis (excessive sweating). N Engl J Med. 2001;344(7):488-493.
100. Carruthers JA, Lowe NJ, Menter MA, et al. A multicenter, double-blind, randomized, placebo-controlled study of the efficacy and safety of botulinum toxin type A in the treatment of glabellar lines. J Am Acad Dermatol. 2002;46(6):840-849.
101. Schurch B, de Sèze M, Denys P, et al. Botulinum toxin type A is a safe and effective treatment for neurogenic urinary incontinence: results of a single treatment, randomized, placebo controlled 6-month study. J Urol. 2005;174:196–200.
102. Aurora SK, Winner P, Freeman MC, et al. OnabotulinumtoxinA for treatment of chronic migraine: Pooled analyses of the 56-week PREEMPT clinical program. Headache. 2011;51(9):1358-1373.
103. Dashtipour K, Chen JJ, Walker HW, et al. Systematic literature review of abobotulinumtoxinA in clinical trials for adult upper limb spasticity. Am J Phys Med Rehabil. 2015;94(3):229-238.
104. Nitti VW, Dmochowski R, Herschorn S, et al. OnabotulinumtoxinA for the treatment of patients with overactive bladder and urinary incontinence: results of a phase 3, randomized, placebo-controlled trial. J Urol. 2017;197(2S):S216-S223.
105. Jongerius PH, van den Hoogen FJA, van Limbeek J, et al. Effect of botulinum toxin in the treatment of drooling: a controlled clinical trial. Pediatrics. 2004;114(3):620-627.
106. Zaninotto, G. Annese V, Costantini M, et al. Randomized controlled trial of botulinum toxin versus laparoscopic heller myotomy for esophageal achalasia. Ann Surg. 2004;239(3):364-370.
107. Dressler D, Adib Saberi F. Botulinum toxin: mechanisms of action. Eur Neurol. 2005;53:3-9.
108. Lewis MB, Bowler PJ. Botulinum toxin cosmetic therapy correlates with a more positive mood. J Cosmet Dermatol. 2009;8(1):24-26.
109. Affatato O, Moulin TC, Pisanu C, et al. High efficacy of onabotulinumtoxinA treatment in patients with comorbid migraine and depression: a meta-analysis. J Transl Med. 2021;19(1):133.
110. Finzi E, Wasserman E. Treatment of depression with botulinum toxin A: a case series. Dermatol Surg. 2006;32(5):645-649; discussion 649-650.
111. Schulze J, Neumann I, Magid M, et al. Botulinum toxin for the management of depression: an updated review of the evidence and meta-analysis. J Psychiatr Res. 2021;135:332-340.
112. Finzi E, Rosenthal NE. Emotional proprioception: treatment of depression with afferent facial feedback. J Psychiatr Res. 2016;80:93-96.
113. Söderkvist S, Ohlén K, Dimberg U. How the experience of emotion is modulated by facial feedback. J Nonverbal Behav. 2018;42(1):129-151.
114. Lewis, MB. The interactions between botulinum-toxin-based facial treatments and embodied emotions. Sci Rep. 2018;8(1):14720.
115. Li Y, Liu J, Liu X, et al. Antidepressant-like action of single facial injection of botulinum neurotoxin A is associated with augmented 5-HT levels and BDNF/ERK/CREB pathways in mouse brain. Neurosci Bull. 2019;35(4):661-672. Erratum in: Neurosci Bull. 2019;35(4):779-780.
116. Gündel H, Wolf A, Xidara V, et al. High psychiatric comorbidity in spasmodic torticollis: a controlled study. J Nerv Ment Dis. 2003;191(7):465-473.
117. Hall TA, McGwin G Jr, Searcey K, et al. Health-related quality of life and psychosocial characteristics of patients with benign essential blepharospasm. Arch Ophthalmol. 2006;124(1):116-119.
118. Ceylan D, Erer S, Zarifog˘lu M, et al. Evaluation of anxiety and depression scales and quality of life in cervical dystonia patients on botulinum toxin therapy and their relatives. Neurol Sci. 2019;40(4):725-731.
119. Heller AS, Lapate RC, Mayer KE, et al. The face of negative affect: trial-by-trial corrugator responses to negative pictures are positively associated with amygdala and negatively associated with ventromedial prefrontal cortex activity. J Cogn Neurosci. 2014;26(9):2102-2110.
120. Makunts T, Wollmer MA, Abagyan R. Postmarketing safety surveillance data reveals antidepressant effects of botulinum toxin across various indications and injection sites. Sci Rep. 2020;10(1):12851.
121. Ahsanuddin S, Roy S, Nasser W, et al. Adverse events associated with botox as reported in a Food and Drug Administration database. Aesthetic Plast Surg. 2021;45(3):1201-1209. doi:10.1007/s00266-020-02027-z
122. Kashif M, Tahir S, Ashfaq F, et al. Association of myofascial trigger points in neck and shoulder region with depression, anxiety, and stress among university students. J Pak Med Assoc. 2021;71(9):2139-2142.
123. Cigarán-Méndez M, Jiménez-Antona C, Parás-Bravo P, et al. Active trigger points are associated with anxiety and widespread pressure pain sensitivity in women, but not men, with tension type headache. Pain Pract. 2019;19(5):522-529.
124. Palacios-Ceña M, Castaldo M, Wang K, et al. Relationship of active trigger points with related disability and anxiety in people with tension-type headache. Medicine (Baltimore). 2017;96(13):e6548.
125. Karadas Ö, Inan LE, Ulas Ü, et al. Efficacy of local lidocaine application on anxiety and depression and its curative effect on patients with chronic tension-type headache. Eur Neurol. 2013;70(1-2):95-101.
126. Gerwin RD. Classification, epidemiology and natural history of myofascial pain syndrome. Curr Pain Headache Rep. 2001;5(5):412-420.
127. Castro Sánchez AM, García López H, Fernández Sánchez M, et al. Improvement in clinical outcomes after dry needling versus myofascial release on pain pressure thresholds, quality of life, fatigue, pain intensity, quality of sleep, anxiety, and depression in patients with fibromyalgia syndrome. Disabil Rehabil. 2019;41(19):2235-2246.
128. Healy GM, Finn DP, O’Gorman DA, et al. Pretreatment anxiety and pain acceptance are associated with response to trigger point injection therapy for chronic myofascial pain. Pain Med. 2015;16(10):1955-1966.
129. Morjaria JB, Lakshminarayana UB, Liu-Shiu-Cheong P, et al. Pneumothorax: a tale of pain or spontaneity. Ther Adv Chronic Dis. 2014;5(6):269-273.
De-pathologizing gender identity: Psychiatry’s role
Treating patients who are transgender or gender diverse (TGGD) requires an understanding of the social and psychological factors that have a unique impact on this population. As clinicians, it is our responsibility to understand the social, cultural, and political issues our patients face, both historically and currently. In this article, we provide information about the nature of gender and gender identity as separate from biological sex and informed by a person’s perception of self as male, female, nonbinary, or other variation.
Psychiatrists must be aware of how individuals who are TGGD have been perceived, classified, and treated by the medical profession, as this history is often a source of mistrust and a barrier to treatment for patients who need psychiatric care. This includes awareness of the “gatekeeping” role that persists in medical institutions today: applying strict eligibility criteria to determine the “fitness” of individuals who are transgender to pursue medical transition, as compared to the informed-consent model that is widely applied to other medical interventions. Our review of minority stress theory, as applicable to this patient population, provides a context and framework for empathic approaches to care for patients who are TGGD. Recognizing barriers to care and ways in which we can create a supportive environment for treatment will allow for tailored approaches that better fit the unique needs of this patient population.
The gender binary
In Western societies, gender has often been viewed as “binary,” oppositional, and directly correlated with physical sex or presumed anatomy.1 The theory of gender essentialism insists that sex and gender are indistinguishable from one another and provide 2 “natural” and distinct categories: women and men. The “gender/sex” binary refers to the belief that individuals born with 2 X chromosomes will inherently develop into and fulfill the social roles of women, and those born with an X and a Y chromosome will develop into and fulfill the social roles of men.1 In this context, “sex” refers to biological characteristics of individuals, including combinations of sex chromosomes, anatomy, and the development of sex characteristics during puberty. The term “gender” refers to the social, cultural, and behavioral aspects of being a man, woman, both, or neither, and “gender identity” refers to one’s internal, individual sense of self and experience of gender (Figure 12). Many Western cultures are now facing destabilization of the gender/sex binary in social, political, and interpersonal contexts.1 This is perhaps most clearly seen in the battle for self-determination and protection by laws affecting individuals who are transgender as well as the determination of other groups to maintain traditional sex and gender roles, often through political action. Historically, individuals who are TGGD have been present in a variety of cultures. For example, most Native American cultures have revered other-gendered individuals, more recently referred to as “two-spirited.” Similarly, the Bugis people of South Sulawesi, Indonesia, recognize 5 genders that exist on a nonbinary spectrum.3
Despite its prevalence in Western society, scientific evidence for the gender/sex binary is lacking. The gender similarities hypothesis states that males and females are similar in most, but not all, psychological variables and is supported by multiple meta-analyses examining psychological gender differences.4 In a 2005 review of 46 meta-analyses of gender-differences, studied through behavior analysis, effect sizes for gender differences were trivial or small in almost 75% of examined variables.5 Analyzing for internal consistency among studies showing large gender/sex differences, Joel et al6 found that, on measures of personality traits, attitudes, interests, and behaviors were rarely homogenous in the brains of males or females. In fact, <1% of study participants showed only masculine or feminine traits, whereas 55% showed a combination, or mosaic, of these traits.6 These findings were supported by further research in behavioral neuroendocrinology that demonstrated a lack of hormonal evidence for 2 distinct sexes. Both estrogen (the “female” hormone) and testosterone (the “male” hormone) are produced by both biological males and females. Further, levels of estradiol do not significantly differ between males and females, and, in fact, in nonpregnant females, estradiol levels are more similar to those of males than to those of pregnant females.1 In the last decade, imaging studies of the human brain have shown that brain structure and connectivity in individuals who are transgender are more similar to those of their experienced gender than of their natal sex.7 In social analyses of intersex individuals (individuals born with ambiguous physical sex characteristics), surgical assignment into the binary gender system did not improve—and often worsened—feelings of isolation and shame.1
The National Institutes of Health defines gender as “socially constructed and enacted roles and behaviors which occur in a historical and cultural context and vary across societies and time.”8 The World Health Organization (WHO) provides a similar definition, and the evidence to support this exists in social-role theory, social-identity theory, and the stereotype-content model. However, despite evidence disputing a gender/sex binary, this method of classifying individuals into a dyad persists in many areas of modern culture, from gender-specific physical spaces (bathrooms, classrooms, store brands), language (pronouns), and laws. This desire for categorization helps fulfill social and psychological needs of groups and individuals by providing group identities and giving structure to the complexity of modern-day life. Identity and group membership provide a sense of belonging, source of self-esteem, and avoidance of ambiguity. Binary gender stereotypes provide expectations that allow anticipation and prediction of our social environments.9 However, the harm of perpetuating the false gender/sex binary is well documented and includes social and economic penalties, extreme violence, and even death. The field of medicine has not been immune from practices that implicitly endorse the gender/sex connection, as seen in the erroneous use of gender in biomedical writings at the highest levels and evidenced in research examining “gender” differences in disease incidence.
Gender diversity as a pathology
The American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders (DSM) has been a source of pathologizing gender diversity since the 1960s, with the introduction of “transsexualism” in DSM-II10 and “gender identity disorder of childhood” in DSM-III.11 These diagnoses were listed under the headings of “sexual deviations” and “psychosexual disorders” in the respective DSM editions. This illustrates how gender diversity was viewed as a mental illness/defect. As the DSM developed through various revisions, so have these diagnoses. DSM-IV used the diagnosis “gender identity disorder.”12 Psychiatry has evolved away from this line of thinking by focusing on the distress from biological sex characteristics that are “incongruent” with an individual’s gender identity, leading to the development of the gender dysphoria diagnosis.13 While this has been a positive step in psychiatry’s efforts to de-pathologize individuals who are gender-diverse, it raises the question: should such diagnoses be included in the DSM at all?
The gender dysphoria diagnosis continues to be needed by many individuals who are TGGD in order to access gender-affirming health care services. Mental health professionals are placed in a gatekeeping role by the expectation that they provide letters of “support” to indicate an individual is of sound mind and consistent gender identity to have services covered by insurance providers. In this way, the insurance industry and the field of medicine continue to believe that individuals who are TGGD need psychiatric permission and/or counsel regarding their gender identity. This can place psychiatry in a role of controlling access to necessary care while also creating a possible distrust in our ability to provide care to patients who are gender-diverse. This is particularly problematic given the high rates of depression, anxiety, trauma, and substance use within these communities.14 In the WHO’s ICD-11, gender dysphoria was changed to gender incongruence and is contained in the category of “Conditions related to sexual health.”15 This indicates continued evolution of how medicine views individuals who are TGGD, and offers hope that psychiatry and the DSM will follow suit.
Continue to: Minority stress theory
Minority stress theory
Ilan Meyer’s minority stress theory explores how cultural and social factors impact mental health functioning (Figure 216). Minority stress theory, which was originally developed for what at the time was described as the lesbian, gay, and bisexual communities, purports that the higher prevalence of mental health disorders among such individuals is likely due to social stigma, discrimination, and stressors associated with minority status. More recently, minority stress theory has been expanded to provide framework for individuals who are TGGD. Hendricks et al17 explain how distal, proximal, and resilience factors contribute to mental health outcomes among these individuals. Distal factors, such as gender-related discrimination, harassment, violence, and rejection, explain how systemic, cultural, and environmental events lead to overt stress. Proximal factors consist of an individual’s expectation and anticipation of negative and stressful events and the internalization of negative attitudes and prejudice (ie, internalized transphobia). Resilience factors consist of community connectedness and within-group identification and can help mediate the negative effects of distal and proximal factors.
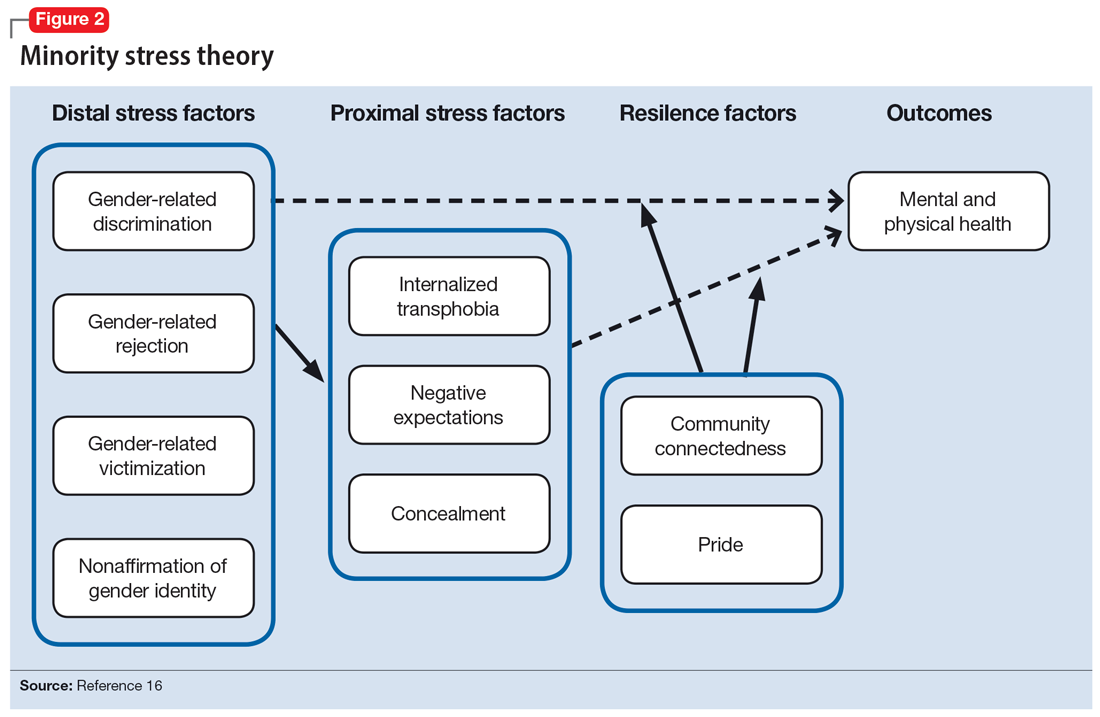
As clinicians, understanding our patients’ experiences and expectations can help us better engage with them and create an environment of safety and healing. Minority stress theory framework suggests that patients may start treatment with distrust or suspicion in light of previous negative experiences. They may also be likely to expect clinicians to be judgmental or to lack understanding of them. The 2015 US Transgender Survey found that 33% of individuals who are TGGD who sought medical treatment in the past year had at least 1 negative experience related to their gender identity (Table 118). Twenty-four percent reported having to educate their clinician about people who are TGGD, while 15% reported the health care professional asked invasive or unnecessary questions about their gender status that were unrelated to their visit. While psychiatry is often distinct from the larger medical field, it is important to understand the negative encounters individuals who are TGGD have likely experienced in medicine, and how those events may skew their feelings about psychiatric treatment. This is especially salient given the higher prevalence of various psychiatric disorders among individuals who are TGGD.18
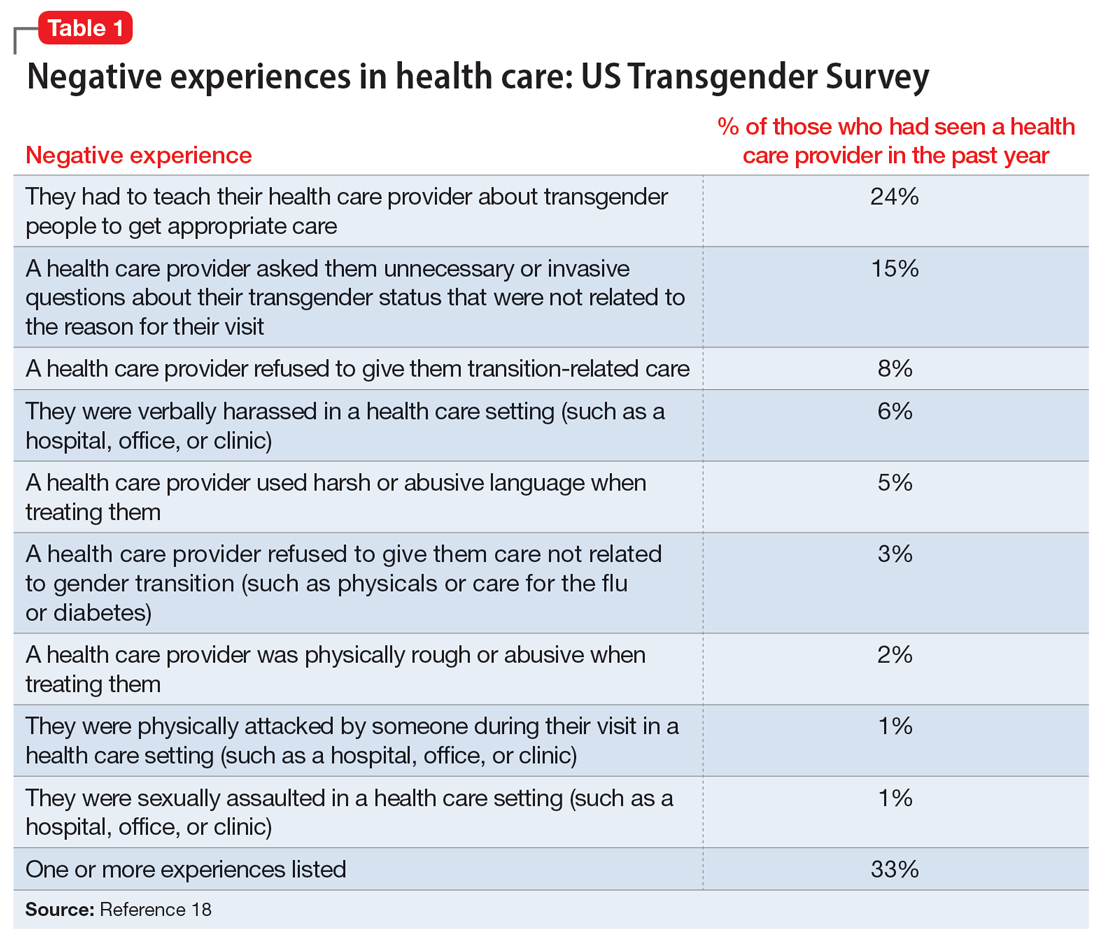
According to the US Transgender Survey, 39% of participants were currently experiencing serious psychological distress, which is nearly 8 times the rate in the US population (5%).18 When extrapolated, this data indicates that we in psychiatry are likely to work with individuals who identify as TGGD, regardless of our expertise. Additionally, research indicates that having access to gender-affirming care—such as hormone replacement therapy, gender-affirming surgery, voice therapy, and other treatments—greatly improves mental health issues such as anxiety, depression, and suicidality among individuals who are TGGD.19,20 It is in this way we in psychiatry must do more than just care for our patients by becoming advocates for them to receive the care they need and deserve. While at times we may want to stay out of politics and other public discourse, it is becoming increasingly necessary as health care is entrenched in politics.
Clinical applicability
Because individuals who are TGGD experience higher rates of depression, anxiety, substance use, and other psychiatric disorders,14 it is increasingly likely that many clinicians will be presented with opportunities to treat such individuals. Despite high rates of psychiatric disorders, individuals who are TGGD often avoid treatment due to concerns about being pathologized, stereotyped, and/or encountering professionals who lack the knowledge to treat them as they are.21 Several studies recommend clinicians better equip themselves to appropriately provide services to individuals who are TGGD.21 Some advise seeking education to understand the unique needs of these patients and to help stay current with appropriate terminology and language (Table 222). This also implies not relying on patients to educate clinicians in understanding their specific needs and experiences.

Making assumptions about a patient’s identity is one of the most commonly reported issues by individuals who are TGGD. Therefore, it is critical to avoid making assumptions about patients based on binary stereotypes.23,24 We can circumvent these mistakes by asking every patient for their name and pronouns, and introducing ourselves with our pronouns. This illustrates an openness and understanding of the importance of identity and language, and makes it common practice from the outset. Integrating the use of gender-neutral language into paperwork, intake forms, charting, and conversation will also help avoid the pitfalls of misgendering and making false assumptions. This will also allow for support staff, medical assistants, and others to use correct language with patients. Having a patient’s used name and pronouns visible for everyone who works with the patient is necessary to effectively meet the patient’s needs. Additionally, understanding that the range of experiences and needs for individuals who are TGGD is heterogeneous can help reduce assumptions and ensure we are asking for needed information. It is also important to ask for only relevant information needed to provide treatment.
Continue to: Resources are widely available...
Resources are widely available to aid in the care of individuals who are TGGD. In 2022, the World Professional Association for Transgender Health released new guidelines—Standards of Care 8—for working with individuals who are TGGD.25 While these standards include a section dedicated to mental health, they also provide guidelines on education, assessments, specific demographic groups, hormone therapy, primary care, and sexual health. Additionally, while we may not want the role of gatekeeping for individuals to receive gender-affirming care, we work within a health care and insurance system that continues to require psychiatric assessment for such surgeries. In this role, we must do our part to educate ourselves in how to best provide these assessments and letters of support to help patients receive appropriate and life-saving care.
Finally, in order to provide a more comfortable and affirming space for individuals who are TGGD, develop ways to self-assess and monitor the policies, procedures, and language used within your practice, clinic, or institution. Monitoring the language used in charting to ensure consistency with the individual’s gender identity is important for our own understanding of the patient, and for patients to feel seen. This is especially true given patients’ access to medical records under the Cures Act. Moreover, it is essential to be cognizant of how you present clients to others in consultation or care coordination to ensure the patient is identified correctly and consistently by clinicians and staff.
Bottom Line
Understanding the social, cultural, and medical discrimination faced by patients who are transgender or gender diverse can make us better suited to engage and treat these individuals in an affirming and supportive way.
Related Resources
- World Professional Association of Transgender Health (WPATH) Standards of Care—8th edition. https://www.tandfonline.com/doi/pdf/10.1080/26895269.2022.2100644
- The Fenway Institute: National LGBTQIA+ Health Education Center. https://fenwayhealth.org/the-fenway-institute/education/the-national-lgbtia-health-education-center/
1. Morgenroth T, Ryan MK. The effects of gender trouble: an integrative theoretical framework of the perpetuation and disruption of the gender/sex binary. Perspect Psychol Sci. 2021;16(6):1113-1142. doi:10.1177/1745691620902442
2. The Roots of Loneliness Project. Accessed April 8, 2023. https://www.rootsofloneliness.com/gender-identity-loneliness
3. Davies SG. Challenging Gender Norms: Five Genders Among Bugis in Indonesia. Thomson Wadsworth; 2007.
4. Hyde JS. The gender similarities hypothesis. Am Psychol. 2005;60(6):581-592. doi:10.1037/0003-066X.60.6.581
5. Joel D. Beyond the binary: rethinking sex and the brain. Neurosci Biobehav Rev. 2021;122:165-175. doi:10.1016/j.neubiorev.2020.22.018
6. Joel D, Berman Z, Tavor I, et al. Sex beyond the genitalia: the human brain mosaic. Proc Natl Acad Sci U S A. 2015;112(50):15468-15473. doi:10.1073/pnas.1509654112
7. Palmer BF, Clegg DJ. A universally accepted definition of gender will positively impact societal understanding, acceptance, and appropriateness of health care. Mayo Clin Proc. 2020;95(10):2235-2243. doi:10.1016/j.mayocp.2020.01.031
8. Office of Research on Women’s Health. Sex & Gender. National Institutes of Health. Accessed April 6, 2023. https://orwh.od.nih.gov/sex-gender
9. Morgenroth T, Sendén MG, Lindqvist A, et al. Defending the sex/gender binary: the role of gender identification and need for closure. Soc Psychol Pers Sci. 2021;12(5):731-740.
10. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 2nd ed. American Psychiatric Association; 1968.
11. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 3rd ed. American Psychiatric Association; 1980.
12. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th ed. American Psychiatric Association; 1994.
13. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Publishing; 2013.
14. Wanta JW, Niforatos JD, Durbak E, et al. Mental health diagnoses among transgender patients in the clinical setting: an all-payer electronic health record study. Transgend Health. 2019;4(1):313-315.
15. World Health Organization. International Statistical Classification of Diseases. 11th ed. World Health Organization; 2019.
16. Meyer IH. Prejudice, social stress, and mental health in lesbian, gay, and bisexual populations: conceptual issues and research evidence. Psychol Bull. 2003;129(5):674-697. doi:10.1037/0033-2909.129.5.674
17. Hendricks ML, Testa RJ. A conceptual framework for clinical work with transgender and gender nonconforming clients: an adaptation of the Minority Stress Model. Profess Psychol: Res Pract. 2012;43(5):460-467. doi:10.1037/a0029597
18. James SE, Herman J, Keisling M, et al. The Report of the 2015 U.S. Transgender Survey. National Center for Transgender Equality; 2016. Accessed April 6, 2023. https://transequality.org/sites/default/files/docs/usts/USTS-Full-Report-Dec17.pdf
19. Almazan AN, Keuroghlian AS. Association between gender-affirming surgeries and mental health outcomes. JAMA Surg. 2021;156(7):611-618. doi:10.1001/jamasurg.2021.0952
20. Tordoff DM, Wanta JW, Collin A, et al. Mental health outcomes in transgender and nonbinary youths receiving gender-affirming care. JAMA Netw Open. 2022;5(2):e220978. doi:10.1001/jamanetworkopen.2022.0978
21. Snow A, Cerel J, Loeffler DN, et al. Barriers to mental health care for transgender and gender-nonconforming adults: a systematic literature review. Health Soc Work. 2019;44(3):149-155. doi:10.1093/hsw/hlz016
22. National LGBTQIA+ Health Education Center. Accessed April 8, 2023. https://www.lgbtqiahealtheducation.org
23. Baldwin A, Dodge B, Schick VR, et al. Transgender and genderqueer individuals’ experiences with health care providers: what’s working, what’s not, and where do we go from here? J Health Care Poor Underserved. 2018;29(4):1300-1318. doi:10.1353/hpu.2018.0097
24. Kcomt L, Gorey KM, Barrett BJ, et al. Healthcare avoidance due to anticipated discrimination among transgender people: a call to create trans-affirmative environments. SSM-Popul Health. 2020;11:100608. doi:10.1016/j.ssmph.2020.100608
25. Coleman E, Radix AE, Bouman WP, et al. Standards of care for the health of transgender and gender diverse people, version 8. Int J Transgender Health. 2022;23(Suppl 1):S1-S259.
Treating patients who are transgender or gender diverse (TGGD) requires an understanding of the social and psychological factors that have a unique impact on this population. As clinicians, it is our responsibility to understand the social, cultural, and political issues our patients face, both historically and currently. In this article, we provide information about the nature of gender and gender identity as separate from biological sex and informed by a person’s perception of self as male, female, nonbinary, or other variation.
Psychiatrists must be aware of how individuals who are TGGD have been perceived, classified, and treated by the medical profession, as this history is often a source of mistrust and a barrier to treatment for patients who need psychiatric care. This includes awareness of the “gatekeeping” role that persists in medical institutions today: applying strict eligibility criteria to determine the “fitness” of individuals who are transgender to pursue medical transition, as compared to the informed-consent model that is widely applied to other medical interventions. Our review of minority stress theory, as applicable to this patient population, provides a context and framework for empathic approaches to care for patients who are TGGD. Recognizing barriers to care and ways in which we can create a supportive environment for treatment will allow for tailored approaches that better fit the unique needs of this patient population.
The gender binary
In Western societies, gender has often been viewed as “binary,” oppositional, and directly correlated with physical sex or presumed anatomy.1 The theory of gender essentialism insists that sex and gender are indistinguishable from one another and provide 2 “natural” and distinct categories: women and men. The “gender/sex” binary refers to the belief that individuals born with 2 X chromosomes will inherently develop into and fulfill the social roles of women, and those born with an X and a Y chromosome will develop into and fulfill the social roles of men.1 In this context, “sex” refers to biological characteristics of individuals, including combinations of sex chromosomes, anatomy, and the development of sex characteristics during puberty. The term “gender” refers to the social, cultural, and behavioral aspects of being a man, woman, both, or neither, and “gender identity” refers to one’s internal, individual sense of self and experience of gender (Figure 12). Many Western cultures are now facing destabilization of the gender/sex binary in social, political, and interpersonal contexts.1 This is perhaps most clearly seen in the battle for self-determination and protection by laws affecting individuals who are transgender as well as the determination of other groups to maintain traditional sex and gender roles, often through political action. Historically, individuals who are TGGD have been present in a variety of cultures. For example, most Native American cultures have revered other-gendered individuals, more recently referred to as “two-spirited.” Similarly, the Bugis people of South Sulawesi, Indonesia, recognize 5 genders that exist on a nonbinary spectrum.3

Despite its prevalence in Western society, scientific evidence for the gender/sex binary is lacking. The gender similarities hypothesis states that males and females are similar in most, but not all, psychological variables and is supported by multiple meta-analyses examining psychological gender differences.4 In a 2005 review of 46 meta-analyses of gender-differences, studied through behavior analysis, effect sizes for gender differences were trivial or small in almost 75% of examined variables.5 Analyzing for internal consistency among studies showing large gender/sex differences, Joel et al6 found that, on measures of personality traits, attitudes, interests, and behaviors were rarely homogenous in the brains of males or females. In fact, <1% of study participants showed only masculine or feminine traits, whereas 55% showed a combination, or mosaic, of these traits.6 These findings were supported by further research in behavioral neuroendocrinology that demonstrated a lack of hormonal evidence for 2 distinct sexes. Both estrogen (the “female” hormone) and testosterone (the “male” hormone) are produced by both biological males and females. Further, levels of estradiol do not significantly differ between males and females, and, in fact, in nonpregnant females, estradiol levels are more similar to those of males than to those of pregnant females.1 In the last decade, imaging studies of the human brain have shown that brain structure and connectivity in individuals who are transgender are more similar to those of their experienced gender than of their natal sex.7 In social analyses of intersex individuals (individuals born with ambiguous physical sex characteristics), surgical assignment into the binary gender system did not improve—and often worsened—feelings of isolation and shame.1
The National Institutes of Health defines gender as “socially constructed and enacted roles and behaviors which occur in a historical and cultural context and vary across societies and time.”8 The World Health Organization (WHO) provides a similar definition, and the evidence to support this exists in social-role theory, social-identity theory, and the stereotype-content model. However, despite evidence disputing a gender/sex binary, this method of classifying individuals into a dyad persists in many areas of modern culture, from gender-specific physical spaces (bathrooms, classrooms, store brands), language (pronouns), and laws. This desire for categorization helps fulfill social and psychological needs of groups and individuals by providing group identities and giving structure to the complexity of modern-day life. Identity and group membership provide a sense of belonging, source of self-esteem, and avoidance of ambiguity. Binary gender stereotypes provide expectations that allow anticipation and prediction of our social environments.9 However, the harm of perpetuating the false gender/sex binary is well documented and includes social and economic penalties, extreme violence, and even death. The field of medicine has not been immune from practices that implicitly endorse the gender/sex connection, as seen in the erroneous use of gender in biomedical writings at the highest levels and evidenced in research examining “gender” differences in disease incidence.
Gender diversity as a pathology
The American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders (DSM) has been a source of pathologizing gender diversity since the 1960s, with the introduction of “transsexualism” in DSM-II10 and “gender identity disorder of childhood” in DSM-III.11 These diagnoses were listed under the headings of “sexual deviations” and “psychosexual disorders” in the respective DSM editions. This illustrates how gender diversity was viewed as a mental illness/defect. As the DSM developed through various revisions, so have these diagnoses. DSM-IV used the diagnosis “gender identity disorder.”12 Psychiatry has evolved away from this line of thinking by focusing on the distress from biological sex characteristics that are “incongruent” with an individual’s gender identity, leading to the development of the gender dysphoria diagnosis.13 While this has been a positive step in psychiatry’s efforts to de-pathologize individuals who are gender-diverse, it raises the question: should such diagnoses be included in the DSM at all?
The gender dysphoria diagnosis continues to be needed by many individuals who are TGGD in order to access gender-affirming health care services. Mental health professionals are placed in a gatekeeping role by the expectation that they provide letters of “support” to indicate an individual is of sound mind and consistent gender identity to have services covered by insurance providers. In this way, the insurance industry and the field of medicine continue to believe that individuals who are TGGD need psychiatric permission and/or counsel regarding their gender identity. This can place psychiatry in a role of controlling access to necessary care while also creating a possible distrust in our ability to provide care to patients who are gender-diverse. This is particularly problematic given the high rates of depression, anxiety, trauma, and substance use within these communities.14 In the WHO’s ICD-11, gender dysphoria was changed to gender incongruence and is contained in the category of “Conditions related to sexual health.”15 This indicates continued evolution of how medicine views individuals who are TGGD, and offers hope that psychiatry and the DSM will follow suit.
Continue to: Minority stress theory
Minority stress theory
Ilan Meyer’s minority stress theory explores how cultural and social factors impact mental health functioning (Figure 216). Minority stress theory, which was originally developed for what at the time was described as the lesbian, gay, and bisexual communities, purports that the higher prevalence of mental health disorders among such individuals is likely due to social stigma, discrimination, and stressors associated with minority status. More recently, minority stress theory has been expanded to provide framework for individuals who are TGGD. Hendricks et al17 explain how distal, proximal, and resilience factors contribute to mental health outcomes among these individuals. Distal factors, such as gender-related discrimination, harassment, violence, and rejection, explain how systemic, cultural, and environmental events lead to overt stress. Proximal factors consist of an individual’s expectation and anticipation of negative and stressful events and the internalization of negative attitudes and prejudice (ie, internalized transphobia). Resilience factors consist of community connectedness and within-group identification and can help mediate the negative effects of distal and proximal factors.

As clinicians, understanding our patients’ experiences and expectations can help us better engage with them and create an environment of safety and healing. Minority stress theory framework suggests that patients may start treatment with distrust or suspicion in light of previous negative experiences. They may also be likely to expect clinicians to be judgmental or to lack understanding of them. The 2015 US Transgender Survey found that 33% of individuals who are TGGD who sought medical treatment in the past year had at least 1 negative experience related to their gender identity (Table 118). Twenty-four percent reported having to educate their clinician about people who are TGGD, while 15% reported the health care professional asked invasive or unnecessary questions about their gender status that were unrelated to their visit. While psychiatry is often distinct from the larger medical field, it is important to understand the negative encounters individuals who are TGGD have likely experienced in medicine, and how those events may skew their feelings about psychiatric treatment. This is especially salient given the higher prevalence of various psychiatric disorders among individuals who are TGGD.18

According to the US Transgender Survey, 39% of participants were currently experiencing serious psychological distress, which is nearly 8 times the rate in the US population (5%).18 When extrapolated, this data indicates that we in psychiatry are likely to work with individuals who identify as TGGD, regardless of our expertise. Additionally, research indicates that having access to gender-affirming care—such as hormone replacement therapy, gender-affirming surgery, voice therapy, and other treatments—greatly improves mental health issues such as anxiety, depression, and suicidality among individuals who are TGGD.19,20 It is in this way we in psychiatry must do more than just care for our patients by becoming advocates for them to receive the care they need and deserve. While at times we may want to stay out of politics and other public discourse, it is becoming increasingly necessary as health care is entrenched in politics.
Clinical applicability
Because individuals who are TGGD experience higher rates of depression, anxiety, substance use, and other psychiatric disorders,14 it is increasingly likely that many clinicians will be presented with opportunities to treat such individuals. Despite high rates of psychiatric disorders, individuals who are TGGD often avoid treatment due to concerns about being pathologized, stereotyped, and/or encountering professionals who lack the knowledge to treat them as they are.21 Several studies recommend clinicians better equip themselves to appropriately provide services to individuals who are TGGD.21 Some advise seeking education to understand the unique needs of these patients and to help stay current with appropriate terminology and language (Table 222). This also implies not relying on patients to educate clinicians in understanding their specific needs and experiences.

Making assumptions about a patient’s identity is one of the most commonly reported issues by individuals who are TGGD. Therefore, it is critical to avoid making assumptions about patients based on binary stereotypes.23,24 We can circumvent these mistakes by asking every patient for their name and pronouns, and introducing ourselves with our pronouns. This illustrates an openness and understanding of the importance of identity and language, and makes it common practice from the outset. Integrating the use of gender-neutral language into paperwork, intake forms, charting, and conversation will also help avoid the pitfalls of misgendering and making false assumptions. This will also allow for support staff, medical assistants, and others to use correct language with patients. Having a patient’s used name and pronouns visible for everyone who works with the patient is necessary to effectively meet the patient’s needs. Additionally, understanding that the range of experiences and needs for individuals who are TGGD is heterogeneous can help reduce assumptions and ensure we are asking for needed information. It is also important to ask for only relevant information needed to provide treatment.
Continue to: Resources are widely available...
Resources are widely available to aid in the care of individuals who are TGGD. In 2022, the World Professional Association for Transgender Health released new guidelines—Standards of Care 8—for working with individuals who are TGGD.25 While these standards include a section dedicated to mental health, they also provide guidelines on education, assessments, specific demographic groups, hormone therapy, primary care, and sexual health. Additionally, while we may not want the role of gatekeeping for individuals to receive gender-affirming care, we work within a health care and insurance system that continues to require psychiatric assessment for such surgeries. In this role, we must do our part to educate ourselves in how to best provide these assessments and letters of support to help patients receive appropriate and life-saving care.
Finally, in order to provide a more comfortable and affirming space for individuals who are TGGD, develop ways to self-assess and monitor the policies, procedures, and language used within your practice, clinic, or institution. Monitoring the language used in charting to ensure consistency with the individual’s gender identity is important for our own understanding of the patient, and for patients to feel seen. This is especially true given patients’ access to medical records under the Cures Act. Moreover, it is essential to be cognizant of how you present clients to others in consultation or care coordination to ensure the patient is identified correctly and consistently by clinicians and staff.
Bottom Line
Understanding the social, cultural, and medical discrimination faced by patients who are transgender or gender diverse can make us better suited to engage and treat these individuals in an affirming and supportive way.
Related Resources
- World Professional Association of Transgender Health (WPATH) Standards of Care—8th edition. https://www.tandfonline.com/doi/pdf/10.1080/26895269.2022.2100644
- The Fenway Institute: National LGBTQIA+ Health Education Center. https://fenwayhealth.org/the-fenway-institute/education/the-national-lgbtia-health-education-center/
Treating patients who are transgender or gender diverse (TGGD) requires an understanding of the social and psychological factors that have a unique impact on this population. As clinicians, it is our responsibility to understand the social, cultural, and political issues our patients face, both historically and currently. In this article, we provide information about the nature of gender and gender identity as separate from biological sex and informed by a person’s perception of self as male, female, nonbinary, or other variation.
Psychiatrists must be aware of how individuals who are TGGD have been perceived, classified, and treated by the medical profession, as this history is often a source of mistrust and a barrier to treatment for patients who need psychiatric care. This includes awareness of the “gatekeeping” role that persists in medical institutions today: applying strict eligibility criteria to determine the “fitness” of individuals who are transgender to pursue medical transition, as compared to the informed-consent model that is widely applied to other medical interventions. Our review of minority stress theory, as applicable to this patient population, provides a context and framework for empathic approaches to care for patients who are TGGD. Recognizing barriers to care and ways in which we can create a supportive environment for treatment will allow for tailored approaches that better fit the unique needs of this patient population.
The gender binary
In Western societies, gender has often been viewed as “binary,” oppositional, and directly correlated with physical sex or presumed anatomy.1 The theory of gender essentialism insists that sex and gender are indistinguishable from one another and provide 2 “natural” and distinct categories: women and men. The “gender/sex” binary refers to the belief that individuals born with 2 X chromosomes will inherently develop into and fulfill the social roles of women, and those born with an X and a Y chromosome will develop into and fulfill the social roles of men.1 In this context, “sex” refers to biological characteristics of individuals, including combinations of sex chromosomes, anatomy, and the development of sex characteristics during puberty. The term “gender” refers to the social, cultural, and behavioral aspects of being a man, woman, both, or neither, and “gender identity” refers to one’s internal, individual sense of self and experience of gender (Figure 12). Many Western cultures are now facing destabilization of the gender/sex binary in social, political, and interpersonal contexts.1 This is perhaps most clearly seen in the battle for self-determination and protection by laws affecting individuals who are transgender as well as the determination of other groups to maintain traditional sex and gender roles, often through political action. Historically, individuals who are TGGD have been present in a variety of cultures. For example, most Native American cultures have revered other-gendered individuals, more recently referred to as “two-spirited.” Similarly, the Bugis people of South Sulawesi, Indonesia, recognize 5 genders that exist on a nonbinary spectrum.3

Despite its prevalence in Western society, scientific evidence for the gender/sex binary is lacking. The gender similarities hypothesis states that males and females are similar in most, but not all, psychological variables and is supported by multiple meta-analyses examining psychological gender differences.4 In a 2005 review of 46 meta-analyses of gender-differences, studied through behavior analysis, effect sizes for gender differences were trivial or small in almost 75% of examined variables.5 Analyzing for internal consistency among studies showing large gender/sex differences, Joel et al6 found that, on measures of personality traits, attitudes, interests, and behaviors were rarely homogenous in the brains of males or females. In fact, <1% of study participants showed only masculine or feminine traits, whereas 55% showed a combination, or mosaic, of these traits.6 These findings were supported by further research in behavioral neuroendocrinology that demonstrated a lack of hormonal evidence for 2 distinct sexes. Both estrogen (the “female” hormone) and testosterone (the “male” hormone) are produced by both biological males and females. Further, levels of estradiol do not significantly differ between males and females, and, in fact, in nonpregnant females, estradiol levels are more similar to those of males than to those of pregnant females.1 In the last decade, imaging studies of the human brain have shown that brain structure and connectivity in individuals who are transgender are more similar to those of their experienced gender than of their natal sex.7 In social analyses of intersex individuals (individuals born with ambiguous physical sex characteristics), surgical assignment into the binary gender system did not improve—and often worsened—feelings of isolation and shame.1
The National Institutes of Health defines gender as “socially constructed and enacted roles and behaviors which occur in a historical and cultural context and vary across societies and time.”8 The World Health Organization (WHO) provides a similar definition, and the evidence to support this exists in social-role theory, social-identity theory, and the stereotype-content model. However, despite evidence disputing a gender/sex binary, this method of classifying individuals into a dyad persists in many areas of modern culture, from gender-specific physical spaces (bathrooms, classrooms, store brands), language (pronouns), and laws. This desire for categorization helps fulfill social and psychological needs of groups and individuals by providing group identities and giving structure to the complexity of modern-day life. Identity and group membership provide a sense of belonging, source of self-esteem, and avoidance of ambiguity. Binary gender stereotypes provide expectations that allow anticipation and prediction of our social environments.9 However, the harm of perpetuating the false gender/sex binary is well documented and includes social and economic penalties, extreme violence, and even death. The field of medicine has not been immune from practices that implicitly endorse the gender/sex connection, as seen in the erroneous use of gender in biomedical writings at the highest levels and evidenced in research examining “gender” differences in disease incidence.
Gender diversity as a pathology
The American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders (DSM) has been a source of pathologizing gender diversity since the 1960s, with the introduction of “transsexualism” in DSM-II10 and “gender identity disorder of childhood” in DSM-III.11 These diagnoses were listed under the headings of “sexual deviations” and “psychosexual disorders” in the respective DSM editions. This illustrates how gender diversity was viewed as a mental illness/defect. As the DSM developed through various revisions, so have these diagnoses. DSM-IV used the diagnosis “gender identity disorder.”12 Psychiatry has evolved away from this line of thinking by focusing on the distress from biological sex characteristics that are “incongruent” with an individual’s gender identity, leading to the development of the gender dysphoria diagnosis.13 While this has been a positive step in psychiatry’s efforts to de-pathologize individuals who are gender-diverse, it raises the question: should such diagnoses be included in the DSM at all?
The gender dysphoria diagnosis continues to be needed by many individuals who are TGGD in order to access gender-affirming health care services. Mental health professionals are placed in a gatekeeping role by the expectation that they provide letters of “support” to indicate an individual is of sound mind and consistent gender identity to have services covered by insurance providers. In this way, the insurance industry and the field of medicine continue to believe that individuals who are TGGD need psychiatric permission and/or counsel regarding their gender identity. This can place psychiatry in a role of controlling access to necessary care while also creating a possible distrust in our ability to provide care to patients who are gender-diverse. This is particularly problematic given the high rates of depression, anxiety, trauma, and substance use within these communities.14 In the WHO’s ICD-11, gender dysphoria was changed to gender incongruence and is contained in the category of “Conditions related to sexual health.”15 This indicates continued evolution of how medicine views individuals who are TGGD, and offers hope that psychiatry and the DSM will follow suit.
Continue to: Minority stress theory
Minority stress theory
Ilan Meyer’s minority stress theory explores how cultural and social factors impact mental health functioning (Figure 216). Minority stress theory, which was originally developed for what at the time was described as the lesbian, gay, and bisexual communities, purports that the higher prevalence of mental health disorders among such individuals is likely due to social stigma, discrimination, and stressors associated with minority status. More recently, minority stress theory has been expanded to provide framework for individuals who are TGGD. Hendricks et al17 explain how distal, proximal, and resilience factors contribute to mental health outcomes among these individuals. Distal factors, such as gender-related discrimination, harassment, violence, and rejection, explain how systemic, cultural, and environmental events lead to overt stress. Proximal factors consist of an individual’s expectation and anticipation of negative and stressful events and the internalization of negative attitudes and prejudice (ie, internalized transphobia). Resilience factors consist of community connectedness and within-group identification and can help mediate the negative effects of distal and proximal factors.

As clinicians, understanding our patients’ experiences and expectations can help us better engage with them and create an environment of safety and healing. Minority stress theory framework suggests that patients may start treatment with distrust or suspicion in light of previous negative experiences. They may also be likely to expect clinicians to be judgmental or to lack understanding of them. The 2015 US Transgender Survey found that 33% of individuals who are TGGD who sought medical treatment in the past year had at least 1 negative experience related to their gender identity (Table 118). Twenty-four percent reported having to educate their clinician about people who are TGGD, while 15% reported the health care professional asked invasive or unnecessary questions about their gender status that were unrelated to their visit. While psychiatry is often distinct from the larger medical field, it is important to understand the negative encounters individuals who are TGGD have likely experienced in medicine, and how those events may skew their feelings about psychiatric treatment. This is especially salient given the higher prevalence of various psychiatric disorders among individuals who are TGGD.18

According to the US Transgender Survey, 39% of participants were currently experiencing serious psychological distress, which is nearly 8 times the rate in the US population (5%).18 When extrapolated, this data indicates that we in psychiatry are likely to work with individuals who identify as TGGD, regardless of our expertise. Additionally, research indicates that having access to gender-affirming care—such as hormone replacement therapy, gender-affirming surgery, voice therapy, and other treatments—greatly improves mental health issues such as anxiety, depression, and suicidality among individuals who are TGGD.19,20 It is in this way we in psychiatry must do more than just care for our patients by becoming advocates for them to receive the care they need and deserve. While at times we may want to stay out of politics and other public discourse, it is becoming increasingly necessary as health care is entrenched in politics.
Clinical applicability
Because individuals who are TGGD experience higher rates of depression, anxiety, substance use, and other psychiatric disorders,14 it is increasingly likely that many clinicians will be presented with opportunities to treat such individuals. Despite high rates of psychiatric disorders, individuals who are TGGD often avoid treatment due to concerns about being pathologized, stereotyped, and/or encountering professionals who lack the knowledge to treat them as they are.21 Several studies recommend clinicians better equip themselves to appropriately provide services to individuals who are TGGD.21 Some advise seeking education to understand the unique needs of these patients and to help stay current with appropriate terminology and language (Table 222). This also implies not relying on patients to educate clinicians in understanding their specific needs and experiences.

Making assumptions about a patient’s identity is one of the most commonly reported issues by individuals who are TGGD. Therefore, it is critical to avoid making assumptions about patients based on binary stereotypes.23,24 We can circumvent these mistakes by asking every patient for their name and pronouns, and introducing ourselves with our pronouns. This illustrates an openness and understanding of the importance of identity and language, and makes it common practice from the outset. Integrating the use of gender-neutral language into paperwork, intake forms, charting, and conversation will also help avoid the pitfalls of misgendering and making false assumptions. This will also allow for support staff, medical assistants, and others to use correct language with patients. Having a patient’s used name and pronouns visible for everyone who works with the patient is necessary to effectively meet the patient’s needs. Additionally, understanding that the range of experiences and needs for individuals who are TGGD is heterogeneous can help reduce assumptions and ensure we are asking for needed information. It is also important to ask for only relevant information needed to provide treatment.
Continue to: Resources are widely available...
Resources are widely available to aid in the care of individuals who are TGGD. In 2022, the World Professional Association for Transgender Health released new guidelines—Standards of Care 8—for working with individuals who are TGGD.25 While these standards include a section dedicated to mental health, they also provide guidelines on education, assessments, specific demographic groups, hormone therapy, primary care, and sexual health. Additionally, while we may not want the role of gatekeeping for individuals to receive gender-affirming care, we work within a health care and insurance system that continues to require psychiatric assessment for such surgeries. In this role, we must do our part to educate ourselves in how to best provide these assessments and letters of support to help patients receive appropriate and life-saving care.
Finally, in order to provide a more comfortable and affirming space for individuals who are TGGD, develop ways to self-assess and monitor the policies, procedures, and language used within your practice, clinic, or institution. Monitoring the language used in charting to ensure consistency with the individual’s gender identity is important for our own understanding of the patient, and for patients to feel seen. This is especially true given patients’ access to medical records under the Cures Act. Moreover, it is essential to be cognizant of how you present clients to others in consultation or care coordination to ensure the patient is identified correctly and consistently by clinicians and staff.
Bottom Line
Understanding the social, cultural, and medical discrimination faced by patients who are transgender or gender diverse can make us better suited to engage and treat these individuals in an affirming and supportive way.
Related Resources
- World Professional Association of Transgender Health (WPATH) Standards of Care—8th edition. https://www.tandfonline.com/doi/pdf/10.1080/26895269.2022.2100644
- The Fenway Institute: National LGBTQIA+ Health Education Center. https://fenwayhealth.org/the-fenway-institute/education/the-national-lgbtia-health-education-center/
1. Morgenroth T, Ryan MK. The effects of gender trouble: an integrative theoretical framework of the perpetuation and disruption of the gender/sex binary. Perspect Psychol Sci. 2021;16(6):1113-1142. doi:10.1177/1745691620902442
2. The Roots of Loneliness Project. Accessed April 8, 2023. https://www.rootsofloneliness.com/gender-identity-loneliness
3. Davies SG. Challenging Gender Norms: Five Genders Among Bugis in Indonesia. Thomson Wadsworth; 2007.
4. Hyde JS. The gender similarities hypothesis. Am Psychol. 2005;60(6):581-592. doi:10.1037/0003-066X.60.6.581
5. Joel D. Beyond the binary: rethinking sex and the brain. Neurosci Biobehav Rev. 2021;122:165-175. doi:10.1016/j.neubiorev.2020.22.018
6. Joel D, Berman Z, Tavor I, et al. Sex beyond the genitalia: the human brain mosaic. Proc Natl Acad Sci U S A. 2015;112(50):15468-15473. doi:10.1073/pnas.1509654112
7. Palmer BF, Clegg DJ. A universally accepted definition of gender will positively impact societal understanding, acceptance, and appropriateness of health care. Mayo Clin Proc. 2020;95(10):2235-2243. doi:10.1016/j.mayocp.2020.01.031
8. Office of Research on Women’s Health. Sex & Gender. National Institutes of Health. Accessed April 6, 2023. https://orwh.od.nih.gov/sex-gender
9. Morgenroth T, Sendén MG, Lindqvist A, et al. Defending the sex/gender binary: the role of gender identification and need for closure. Soc Psychol Pers Sci. 2021;12(5):731-740.
10. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 2nd ed. American Psychiatric Association; 1968.
11. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 3rd ed. American Psychiatric Association; 1980.
12. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th ed. American Psychiatric Association; 1994.
13. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Publishing; 2013.
14. Wanta JW, Niforatos JD, Durbak E, et al. Mental health diagnoses among transgender patients in the clinical setting: an all-payer electronic health record study. Transgend Health. 2019;4(1):313-315.
15. World Health Organization. International Statistical Classification of Diseases. 11th ed. World Health Organization; 2019.
16. Meyer IH. Prejudice, social stress, and mental health in lesbian, gay, and bisexual populations: conceptual issues and research evidence. Psychol Bull. 2003;129(5):674-697. doi:10.1037/0033-2909.129.5.674
17. Hendricks ML, Testa RJ. A conceptual framework for clinical work with transgender and gender nonconforming clients: an adaptation of the Minority Stress Model. Profess Psychol: Res Pract. 2012;43(5):460-467. doi:10.1037/a0029597
18. James SE, Herman J, Keisling M, et al. The Report of the 2015 U.S. Transgender Survey. National Center for Transgender Equality; 2016. Accessed April 6, 2023. https://transequality.org/sites/default/files/docs/usts/USTS-Full-Report-Dec17.pdf
19. Almazan AN, Keuroghlian AS. Association between gender-affirming surgeries and mental health outcomes. JAMA Surg. 2021;156(7):611-618. doi:10.1001/jamasurg.2021.0952
20. Tordoff DM, Wanta JW, Collin A, et al. Mental health outcomes in transgender and nonbinary youths receiving gender-affirming care. JAMA Netw Open. 2022;5(2):e220978. doi:10.1001/jamanetworkopen.2022.0978
21. Snow A, Cerel J, Loeffler DN, et al. Barriers to mental health care for transgender and gender-nonconforming adults: a systematic literature review. Health Soc Work. 2019;44(3):149-155. doi:10.1093/hsw/hlz016
22. National LGBTQIA+ Health Education Center. Accessed April 8, 2023. https://www.lgbtqiahealtheducation.org
23. Baldwin A, Dodge B, Schick VR, et al. Transgender and genderqueer individuals’ experiences with health care providers: what’s working, what’s not, and where do we go from here? J Health Care Poor Underserved. 2018;29(4):1300-1318. doi:10.1353/hpu.2018.0097
24. Kcomt L, Gorey KM, Barrett BJ, et al. Healthcare avoidance due to anticipated discrimination among transgender people: a call to create trans-affirmative environments. SSM-Popul Health. 2020;11:100608. doi:10.1016/j.ssmph.2020.100608
25. Coleman E, Radix AE, Bouman WP, et al. Standards of care for the health of transgender and gender diverse people, version 8. Int J Transgender Health. 2022;23(Suppl 1):S1-S259.
1. Morgenroth T, Ryan MK. The effects of gender trouble: an integrative theoretical framework of the perpetuation and disruption of the gender/sex binary. Perspect Psychol Sci. 2021;16(6):1113-1142. doi:10.1177/1745691620902442
2. The Roots of Loneliness Project. Accessed April 8, 2023. https://www.rootsofloneliness.com/gender-identity-loneliness
3. Davies SG. Challenging Gender Norms: Five Genders Among Bugis in Indonesia. Thomson Wadsworth; 2007.
4. Hyde JS. The gender similarities hypothesis. Am Psychol. 2005;60(6):581-592. doi:10.1037/0003-066X.60.6.581
5. Joel D. Beyond the binary: rethinking sex and the brain. Neurosci Biobehav Rev. 2021;122:165-175. doi:10.1016/j.neubiorev.2020.22.018
6. Joel D, Berman Z, Tavor I, et al. Sex beyond the genitalia: the human brain mosaic. Proc Natl Acad Sci U S A. 2015;112(50):15468-15473. doi:10.1073/pnas.1509654112
7. Palmer BF, Clegg DJ. A universally accepted definition of gender will positively impact societal understanding, acceptance, and appropriateness of health care. Mayo Clin Proc. 2020;95(10):2235-2243. doi:10.1016/j.mayocp.2020.01.031
8. Office of Research on Women’s Health. Sex & Gender. National Institutes of Health. Accessed April 6, 2023. https://orwh.od.nih.gov/sex-gender
9. Morgenroth T, Sendén MG, Lindqvist A, et al. Defending the sex/gender binary: the role of gender identification and need for closure. Soc Psychol Pers Sci. 2021;12(5):731-740.
10. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 2nd ed. American Psychiatric Association; 1968.
11. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 3rd ed. American Psychiatric Association; 1980.
12. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th ed. American Psychiatric Association; 1994.
13. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Publishing; 2013.
14. Wanta JW, Niforatos JD, Durbak E, et al. Mental health diagnoses among transgender patients in the clinical setting: an all-payer electronic health record study. Transgend Health. 2019;4(1):313-315.
15. World Health Organization. International Statistical Classification of Diseases. 11th ed. World Health Organization; 2019.
16. Meyer IH. Prejudice, social stress, and mental health in lesbian, gay, and bisexual populations: conceptual issues and research evidence. Psychol Bull. 2003;129(5):674-697. doi:10.1037/0033-2909.129.5.674
17. Hendricks ML, Testa RJ. A conceptual framework for clinical work with transgender and gender nonconforming clients: an adaptation of the Minority Stress Model. Profess Psychol: Res Pract. 2012;43(5):460-467. doi:10.1037/a0029597
18. James SE, Herman J, Keisling M, et al. The Report of the 2015 U.S. Transgender Survey. National Center for Transgender Equality; 2016. Accessed April 6, 2023. https://transequality.org/sites/default/files/docs/usts/USTS-Full-Report-Dec17.pdf
19. Almazan AN, Keuroghlian AS. Association between gender-affirming surgeries and mental health outcomes. JAMA Surg. 2021;156(7):611-618. doi:10.1001/jamasurg.2021.0952
20. Tordoff DM, Wanta JW, Collin A, et al. Mental health outcomes in transgender and nonbinary youths receiving gender-affirming care. JAMA Netw Open. 2022;5(2):e220978. doi:10.1001/jamanetworkopen.2022.0978
21. Snow A, Cerel J, Loeffler DN, et al. Barriers to mental health care for transgender and gender-nonconforming adults: a systematic literature review. Health Soc Work. 2019;44(3):149-155. doi:10.1093/hsw/hlz016
22. National LGBTQIA+ Health Education Center. Accessed April 8, 2023. https://www.lgbtqiahealtheducation.org
23. Baldwin A, Dodge B, Schick VR, et al. Transgender and genderqueer individuals’ experiences with health care providers: what’s working, what’s not, and where do we go from here? J Health Care Poor Underserved. 2018;29(4):1300-1318. doi:10.1353/hpu.2018.0097
24. Kcomt L, Gorey KM, Barrett BJ, et al. Healthcare avoidance due to anticipated discrimination among transgender people: a call to create trans-affirmative environments. SSM-Popul Health. 2020;11:100608. doi:10.1016/j.ssmph.2020.100608
25. Coleman E, Radix AE, Bouman WP, et al. Standards of care for the health of transgender and gender diverse people, version 8. Int J Transgender Health. 2022;23(Suppl 1):S1-S259.
The joys and rewards of an asymmetric life
The benefits of living a balanced life is a very popular concept. But I beg to differ. Balance in one’s life is overrated. Allocating equal time to the various components of one’s life may sound admirable, but it is a recipe for an ordinary life, with no major achievements or a memorable legacy. Scoring a “moonshot” achievement while living a balanced life is highly unlikely.
The benefits of deliberately leading an “asymmetric life” is an epiphany I acquired as a young boy addicted to watching stellar Olympic athletes win gold medals. I dreamed about being the best in the world in a sport, or in something else. As I read about the lives of my Olympic idols, my mind was opened to the fact that each of them led an unbalanced life in the pursuit of their cherished goal to be the best in the world: a gold medalist. I found out that for several years before the Olympic games, these athletes spent a disproportionate amount of their waking time (≥10 hours a day) practicing their sport, strengthening their muscles, building up their stamina, and honing their physical skills and mental toughness. Those sacrifices were necessary—in fact, indispensable—to set themselves apart from us mere mortals. Their social life was quite restricted, and even their educational pursuits had to be reduced or deferred.
I realized at a young age that to be the world’s best athlete, one must lead a purpose-driven life and channel a tremendous amount of time and energy to achieve the cherished goal of an Olympic gold medal. I understood the sacrifices necessary to excel in sports, and concluded the same was also true outside of sports, such as for Nobel Laureates, world-class pianists, prodigious authors, ballet dancers, opera divas, or self-employed entrepreneurs.
As I grew up, I repeatedly heard people praise “the balanced life,” but in my heart, I knew that was a fallacy. I had already decided in high school that I wanted to become a psychiatric physician. I was a premed major in college and very aware that our medical school enrolled only 44 students into the Med 1 class. There were >350 other premed undergraduates. Thus, without hesitation, and with gusto, I deliberately led an unbalanced life, studying countless hours each day to achieve an A grade in all required and elective courses to earn a spot on the Dean’s list. I already had confidence in my academic skills because of my excellent performance in high school, but I was not going to take any chances because I recalled a quote commonly attributed to Thomas Edison: “Genius is 1% inspiration and 99% perspiration.” This is obviously antithetical to living a balanced life.
I matriculated in medical school, and my unbalanced lifestyle continued unabated. Most readers of this journal are fellow physicians who know well the heavy demands of medical school on our lives, in both the preclinical and clinical years. Trying to lead a balanced life during the 4 years of medical school can have disastrous consequences. We all led an “asymmetric existence” with 75% (or more) of our waking hours invested in our careers and 25% (or less) directed to our social lives (and fortunately, our families and friends generally understood). That is what it takes to earn the coveted MD, the equivalent of an Olympic medal for intellectual athletes.
Then came 4 more years of psychiatric residency training, and the long hours of work continued, along with many nights and weekends on call. As a resident, I treasured the modest but precious amount of time I had outside work. I was lucky to have a very supportive and competent wife (a psychologist), who spared me from having to wake up at night to feed our first baby or do various household chores, so I could read the many articles and books on my desk and catch up on my sleep after my frequent night and weekend call shifts.
My unbalanced life continued when I pursued a postresidency fellowship at the National Institutes of Health, where I conducted numerous clinical research trials, brain imaging studies, and postmortem research on a large collection of brains from deceased patients with schizophrenia or bipolar disorder. I worked 12 to 15 hours a day to write up the data I had collected, submit it to scientific journals, and revise it as needed. I knew from the strategic plan I had set for my life that the neuroscience fellowship would launch my academic career, and indeed it did.
Continue to: Reaping the benefits
Reaping the benefits
Fast forward 30 years and you will still find me leading an unbalanced but joyful and fulfilling life. People often ask me how I was able to achieve so much (authoring several hundred scientific publications; publishing 13 books; receiving dozens of grants; editing 3 scientific journals; founding an international schizophrenia society; assuming many leadership positions, including becoming a department chair at 2 universities and being elected to the presidency of several associations; lecturing around the world and making hundreds of scientific presentations at national and international conferences; seeing thousands of patients; teaching, supervising, and mentoring countless medical students, psychiatric residents, and young faculty members; and creating a nonprofit foundation [CURESZ.org] with a former patient who recovered completely after 5 years of home classes and treatment-refractory command hallucinations who then graduated from college with honors in molecular biology after I prescribed clozapine to “cure” her from what was deemed a hopeless and irreversible mental disability1). In all, thanks to my unbalanced life, I have achieved 12 moonshots and each is a major achievement of which I am proud.
My answer to those who ask me how I did all that is simple: I have strategically led an unbalanced life, enjoying every minute of it, and reaping the fruits of my labor. I do not waste an inordinate amount of time watching TV or participating in social media like many others might. And more importantly, despite this unbalanced life, I have been married to my college sweetheart for several decades and have a son and a daughter who are very high achievers and make me proud. I do budget time to regularly take my children and grandchildren on family vacations to exotic locations. I have dinner with my family every night. I am very happy with this so-called unbalanced life. I have received numerous awards and recognitions for my accomplishments, including the Distinguished Scholar Award (the highest academic recognition at The Ohio State University), the coveted Stanley Dean Award for research into schizophrenia from the American College of Psychiatrists, 4 Golden Apple Teaching Awards, and the Daniel Drake Medal, the highest honor that the University of Cincinnati College of Medicine bestows on a faculty member. (Dr. Drake founded the University of Cincinnati College of Medicine 200 years ago, a major moonshot, and among his many accomplishments, he also established the first psychiatric hospital in Ohio, another consequential moonshot. I am sure he led a very productive, unbalanced life, and that is why he is still remembered and revered 200 years later.)
It is said that at the height of his prominence 90 years ago, Sigmund Freud was asked, “What is life all about?” He responded with 2 words: “Liebe und arbeit” (love and work). Importantly, he did not specify which proportions those 2 major functions should occupy in one’s life. It was left up to each individual to make that choice. In the constitution of our country, that freedom of choice is the secret sauce of “the pursuit of happiness.”
1. The CURESZ Foundation. Who we are. Accessed April 11, 2023. https://curesz.org/about/who-we-are/
The benefits of living a balanced life is a very popular concept. But I beg to differ. Balance in one’s life is overrated. Allocating equal time to the various components of one’s life may sound admirable, but it is a recipe for an ordinary life, with no major achievements or a memorable legacy. Scoring a “moonshot” achievement while living a balanced life is highly unlikely.
The benefits of deliberately leading an “asymmetric life” is an epiphany I acquired as a young boy addicted to watching stellar Olympic athletes win gold medals. I dreamed about being the best in the world in a sport, or in something else. As I read about the lives of my Olympic idols, my mind was opened to the fact that each of them led an unbalanced life in the pursuit of their cherished goal to be the best in the world: a gold medalist. I found out that for several years before the Olympic games, these athletes spent a disproportionate amount of their waking time (≥10 hours a day) practicing their sport, strengthening their muscles, building up their stamina, and honing their physical skills and mental toughness. Those sacrifices were necessary—in fact, indispensable—to set themselves apart from us mere mortals. Their social life was quite restricted, and even their educational pursuits had to be reduced or deferred.
I realized at a young age that to be the world’s best athlete, one must lead a purpose-driven life and channel a tremendous amount of time and energy to achieve the cherished goal of an Olympic gold medal. I understood the sacrifices necessary to excel in sports, and concluded the same was also true outside of sports, such as for Nobel Laureates, world-class pianists, prodigious authors, ballet dancers, opera divas, or self-employed entrepreneurs.
As I grew up, I repeatedly heard people praise “the balanced life,” but in my heart, I knew that was a fallacy. I had already decided in high school that I wanted to become a psychiatric physician. I was a premed major in college and very aware that our medical school enrolled only 44 students into the Med 1 class. There were >350 other premed undergraduates. Thus, without hesitation, and with gusto, I deliberately led an unbalanced life, studying countless hours each day to achieve an A grade in all required and elective courses to earn a spot on the Dean’s list. I already had confidence in my academic skills because of my excellent performance in high school, but I was not going to take any chances because I recalled a quote commonly attributed to Thomas Edison: “Genius is 1% inspiration and 99% perspiration.” This is obviously antithetical to living a balanced life.
I matriculated in medical school, and my unbalanced lifestyle continued unabated. Most readers of this journal are fellow physicians who know well the heavy demands of medical school on our lives, in both the preclinical and clinical years. Trying to lead a balanced life during the 4 years of medical school can have disastrous consequences. We all led an “asymmetric existence” with 75% (or more) of our waking hours invested in our careers and 25% (or less) directed to our social lives (and fortunately, our families and friends generally understood). That is what it takes to earn the coveted MD, the equivalent of an Olympic medal for intellectual athletes.
Then came 4 more years of psychiatric residency training, and the long hours of work continued, along with many nights and weekends on call. As a resident, I treasured the modest but precious amount of time I had outside work. I was lucky to have a very supportive and competent wife (a psychologist), who spared me from having to wake up at night to feed our first baby or do various household chores, so I could read the many articles and books on my desk and catch up on my sleep after my frequent night and weekend call shifts.
My unbalanced life continued when I pursued a postresidency fellowship at the National Institutes of Health, where I conducted numerous clinical research trials, brain imaging studies, and postmortem research on a large collection of brains from deceased patients with schizophrenia or bipolar disorder. I worked 12 to 15 hours a day to write up the data I had collected, submit it to scientific journals, and revise it as needed. I knew from the strategic plan I had set for my life that the neuroscience fellowship would launch my academic career, and indeed it did.
Continue to: Reaping the benefits
Reaping the benefits
Fast forward 30 years and you will still find me leading an unbalanced but joyful and fulfilling life. People often ask me how I was able to achieve so much (authoring several hundred scientific publications; publishing 13 books; receiving dozens of grants; editing 3 scientific journals; founding an international schizophrenia society; assuming many leadership positions, including becoming a department chair at 2 universities and being elected to the presidency of several associations; lecturing around the world and making hundreds of scientific presentations at national and international conferences; seeing thousands of patients; teaching, supervising, and mentoring countless medical students, psychiatric residents, and young faculty members; and creating a nonprofit foundation [CURESZ.org] with a former patient who recovered completely after 5 years of home classes and treatment-refractory command hallucinations who then graduated from college with honors in molecular biology after I prescribed clozapine to “cure” her from what was deemed a hopeless and irreversible mental disability1). In all, thanks to my unbalanced life, I have achieved 12 moonshots and each is a major achievement of which I am proud.
My answer to those who ask me how I did all that is simple: I have strategically led an unbalanced life, enjoying every minute of it, and reaping the fruits of my labor. I do not waste an inordinate amount of time watching TV or participating in social media like many others might. And more importantly, despite this unbalanced life, I have been married to my college sweetheart for several decades and have a son and a daughter who are very high achievers and make me proud. I do budget time to regularly take my children and grandchildren on family vacations to exotic locations. I have dinner with my family every night. I am very happy with this so-called unbalanced life. I have received numerous awards and recognitions for my accomplishments, including the Distinguished Scholar Award (the highest academic recognition at The Ohio State University), the coveted Stanley Dean Award for research into schizophrenia from the American College of Psychiatrists, 4 Golden Apple Teaching Awards, and the Daniel Drake Medal, the highest honor that the University of Cincinnati College of Medicine bestows on a faculty member. (Dr. Drake founded the University of Cincinnati College of Medicine 200 years ago, a major moonshot, and among his many accomplishments, he also established the first psychiatric hospital in Ohio, another consequential moonshot. I am sure he led a very productive, unbalanced life, and that is why he is still remembered and revered 200 years later.)
It is said that at the height of his prominence 90 years ago, Sigmund Freud was asked, “What is life all about?” He responded with 2 words: “Liebe und arbeit” (love and work). Importantly, he did not specify which proportions those 2 major functions should occupy in one’s life. It was left up to each individual to make that choice. In the constitution of our country, that freedom of choice is the secret sauce of “the pursuit of happiness.”
The benefits of living a balanced life is a very popular concept. But I beg to differ. Balance in one’s life is overrated. Allocating equal time to the various components of one’s life may sound admirable, but it is a recipe for an ordinary life, with no major achievements or a memorable legacy. Scoring a “moonshot” achievement while living a balanced life is highly unlikely.
The benefits of deliberately leading an “asymmetric life” is an epiphany I acquired as a young boy addicted to watching stellar Olympic athletes win gold medals. I dreamed about being the best in the world in a sport, or in something else. As I read about the lives of my Olympic idols, my mind was opened to the fact that each of them led an unbalanced life in the pursuit of their cherished goal to be the best in the world: a gold medalist. I found out that for several years before the Olympic games, these athletes spent a disproportionate amount of their waking time (≥10 hours a day) practicing their sport, strengthening their muscles, building up their stamina, and honing their physical skills and mental toughness. Those sacrifices were necessary—in fact, indispensable—to set themselves apart from us mere mortals. Their social life was quite restricted, and even their educational pursuits had to be reduced or deferred.
I realized at a young age that to be the world’s best athlete, one must lead a purpose-driven life and channel a tremendous amount of time and energy to achieve the cherished goal of an Olympic gold medal. I understood the sacrifices necessary to excel in sports, and concluded the same was also true outside of sports, such as for Nobel Laureates, world-class pianists, prodigious authors, ballet dancers, opera divas, or self-employed entrepreneurs.
As I grew up, I repeatedly heard people praise “the balanced life,” but in my heart, I knew that was a fallacy. I had already decided in high school that I wanted to become a psychiatric physician. I was a premed major in college and very aware that our medical school enrolled only 44 students into the Med 1 class. There were >350 other premed undergraduates. Thus, without hesitation, and with gusto, I deliberately led an unbalanced life, studying countless hours each day to achieve an A grade in all required and elective courses to earn a spot on the Dean’s list. I already had confidence in my academic skills because of my excellent performance in high school, but I was not going to take any chances because I recalled a quote commonly attributed to Thomas Edison: “Genius is 1% inspiration and 99% perspiration.” This is obviously antithetical to living a balanced life.
I matriculated in medical school, and my unbalanced lifestyle continued unabated. Most readers of this journal are fellow physicians who know well the heavy demands of medical school on our lives, in both the preclinical and clinical years. Trying to lead a balanced life during the 4 years of medical school can have disastrous consequences. We all led an “asymmetric existence” with 75% (or more) of our waking hours invested in our careers and 25% (or less) directed to our social lives (and fortunately, our families and friends generally understood). That is what it takes to earn the coveted MD, the equivalent of an Olympic medal for intellectual athletes.
Then came 4 more years of psychiatric residency training, and the long hours of work continued, along with many nights and weekends on call. As a resident, I treasured the modest but precious amount of time I had outside work. I was lucky to have a very supportive and competent wife (a psychologist), who spared me from having to wake up at night to feed our first baby or do various household chores, so I could read the many articles and books on my desk and catch up on my sleep after my frequent night and weekend call shifts.
My unbalanced life continued when I pursued a postresidency fellowship at the National Institutes of Health, where I conducted numerous clinical research trials, brain imaging studies, and postmortem research on a large collection of brains from deceased patients with schizophrenia or bipolar disorder. I worked 12 to 15 hours a day to write up the data I had collected, submit it to scientific journals, and revise it as needed. I knew from the strategic plan I had set for my life that the neuroscience fellowship would launch my academic career, and indeed it did.
Continue to: Reaping the benefits
Reaping the benefits
Fast forward 30 years and you will still find me leading an unbalanced but joyful and fulfilling life. People often ask me how I was able to achieve so much (authoring several hundred scientific publications; publishing 13 books; receiving dozens of grants; editing 3 scientific journals; founding an international schizophrenia society; assuming many leadership positions, including becoming a department chair at 2 universities and being elected to the presidency of several associations; lecturing around the world and making hundreds of scientific presentations at national and international conferences; seeing thousands of patients; teaching, supervising, and mentoring countless medical students, psychiatric residents, and young faculty members; and creating a nonprofit foundation [CURESZ.org] with a former patient who recovered completely after 5 years of home classes and treatment-refractory command hallucinations who then graduated from college with honors in molecular biology after I prescribed clozapine to “cure” her from what was deemed a hopeless and irreversible mental disability1). In all, thanks to my unbalanced life, I have achieved 12 moonshots and each is a major achievement of which I am proud.
My answer to those who ask me how I did all that is simple: I have strategically led an unbalanced life, enjoying every minute of it, and reaping the fruits of my labor. I do not waste an inordinate amount of time watching TV or participating in social media like many others might. And more importantly, despite this unbalanced life, I have been married to my college sweetheart for several decades and have a son and a daughter who are very high achievers and make me proud. I do budget time to regularly take my children and grandchildren on family vacations to exotic locations. I have dinner with my family every night. I am very happy with this so-called unbalanced life. I have received numerous awards and recognitions for my accomplishments, including the Distinguished Scholar Award (the highest academic recognition at The Ohio State University), the coveted Stanley Dean Award for research into schizophrenia from the American College of Psychiatrists, 4 Golden Apple Teaching Awards, and the Daniel Drake Medal, the highest honor that the University of Cincinnati College of Medicine bestows on a faculty member. (Dr. Drake founded the University of Cincinnati College of Medicine 200 years ago, a major moonshot, and among his many accomplishments, he also established the first psychiatric hospital in Ohio, another consequential moonshot. I am sure he led a very productive, unbalanced life, and that is why he is still remembered and revered 200 years later.)
It is said that at the height of his prominence 90 years ago, Sigmund Freud was asked, “What is life all about?” He responded with 2 words: “Liebe und arbeit” (love and work). Importantly, he did not specify which proportions those 2 major functions should occupy in one’s life. It was left up to each individual to make that choice. In the constitution of our country, that freedom of choice is the secret sauce of “the pursuit of happiness.”
1. The CURESZ Foundation. Who we are. Accessed April 11, 2023. https://curesz.org/about/who-we-are/
1. The CURESZ Foundation. Who we are. Accessed April 11, 2023. https://curesz.org/about/who-we-are/
Antidepressants for patients who are breastfeeding: What to consider
Ms. D, age 32, recently gave birth to her second child. Her psychiatric history includes major depressive disorder. She had been stable on mirtazapine 30 mg at bedtime for 3 years. Based on clinical stability and patient preference, Ms. D elected to taper off mirtazapine 1 month prior to delivery. Now at 1 month postdelivery, Ms. D notes the reemergence of her depressive symptoms; during her child’s latest pediatrician visit, she scores 15 on the Edinburgh Postnatal Depression Scale (EPDS). She breastfeeds her baby and wants more information on the safety of taking an antidepressant while breastfeeding.
Ms. D discusses her previous use of mirtazapine with her treatment team. The team reviews the available resources with Ms. D and together they plan to make a shared decision regarding treatment of her depression at her next appointment.
The American Academy of Pediatrics1 and World Health Organization2 recommend exclusive breastfeeding of infants for their first 6 months of life and support it as a complement to other foods through and beyond age 2. Untreated conditions such as postpartum depression impact maternal well-being and may interfere with parenting and child development. In fact, untreated maternal mental health leads to an increased risk of suicide, reduced maternal economic productivity, and worsened health for both mother and child.3
Because many women experience psychiatric symptoms before they become pregnant as well as during the perinatal period, questions often arise regardingthe use of psychiatric medications—specifically antidepressants—and their safety in patients who are breastfeeding. Key considerations regarding medication management should include the patient’s previous response to medications, the risks of untreated maternal mental illness, and evidence regarding risks and benefits in lactation. This article summarizes where to find evidence-based lactation information, how to interpret that information, and what information is available for select antidepressants.
Locating lactation information
Start by checking the manufacturer’s medication labeling (“prescribing information”) and medication information resources such as Micromedex (www.micromedexsolutions.com) and Lexicomp (www.wolterskluwer.com/en/solutions/lexicomp). The updated labeling includes a risk/benefit assessment of available data on the risk for continued use of a medication during pregnancy compared to the risk if a medication is discontinued and the disorder goes untreated.4 The “breastfeeding considerations” section of medication labeling include details regarding the presence of the medication and the amount of it in breastmilk, adverse events in infants exposed to the medication through breastmilk, and additional pertinent data as applicable. Lexicomp includes information regarding breastfeeding considerations, and a subscription may also include access to Briggs Drugs in Pregnancy and Lactation’s information pages. Micromedex includes its own lactation safety rating scale score.
Several other resources can help guide clinicians toward patient-specific recommendations. From the National Library of Medicine, LactMed (https://www.ncbi.nlm.nih.gov/books/NBK501922/) allows clinicians to search for specific medications to see what information exists pertaining to medication levels in breastmilk and infant blood as well as potential adverse effects in the nursing infant and/or on lactation and breastmilk.5 LactMed provides information regarding alternative medications to consider and references from which the information was gathered.
Another helpful resource is the InfantRisk Center from Texas Tech University Health Sciences Center, which includes a free call center for parents and clinicians who have questions about medications and breastfeeding (806-352-2519; Monday through Friday, 8
Continue to: How to interpret the information
How to interpret the information
Medication levels in breastmilk are affected by several properties, such as the medication’s molecular weight, protein binding, pKa, and volume of distribution. A few commonly used terms in lactation literature for medications include the relative infant dose (RID) and milk/plasma (M/P) ratio.
RID provides information about relative medication exposure for the infant. It is calculated by dividing the infant’s dose of a medication via breastmilk (mg/kg/d) by the mother’s dose (mg/kg/d).7 Most consider an RID <10% to be safe.7
M/P is the ratio of medication concentration in the mother’s milk divided by the medication concentration in the mother’s plasma. A ratio <1 is preferable and generally indicates that a low level of medication has been transferred to human milk.7
Another factor that can be evaluated is protein binding. Medications that are highly protein-bound do not tend to pass as easily into breastmilk and can minimize infant exposure.
Several risk categorization systems are available, depending upon the resource used to obtain lactation information. One common system is Hale’s Lactation Risk Categories, with 5 safety levels ranging from L1 (breastfeeding compatible) to L5 (hazardous) (Table 17). Briggs et al8 utilize 7 categories to summarize recommendations ranging from breastfeeding-compatible to contraindicated; however, it is important to read the full medication monograph in the context of the rating provided.Table 27,8 provides breastfeeding information from Hale’s7 and from Briggs et al8 for some commonly used antidepressants.

In addition to interpreting available literature, it is also important to consider patient-specific factors, including (but not limited to) the severity of the patient’s psychiatric disorder and their previous response to medication. If a mother achieved remission on a particular antidepressant in the past, it may be preferable to restart that agent rather than trial a new medication.
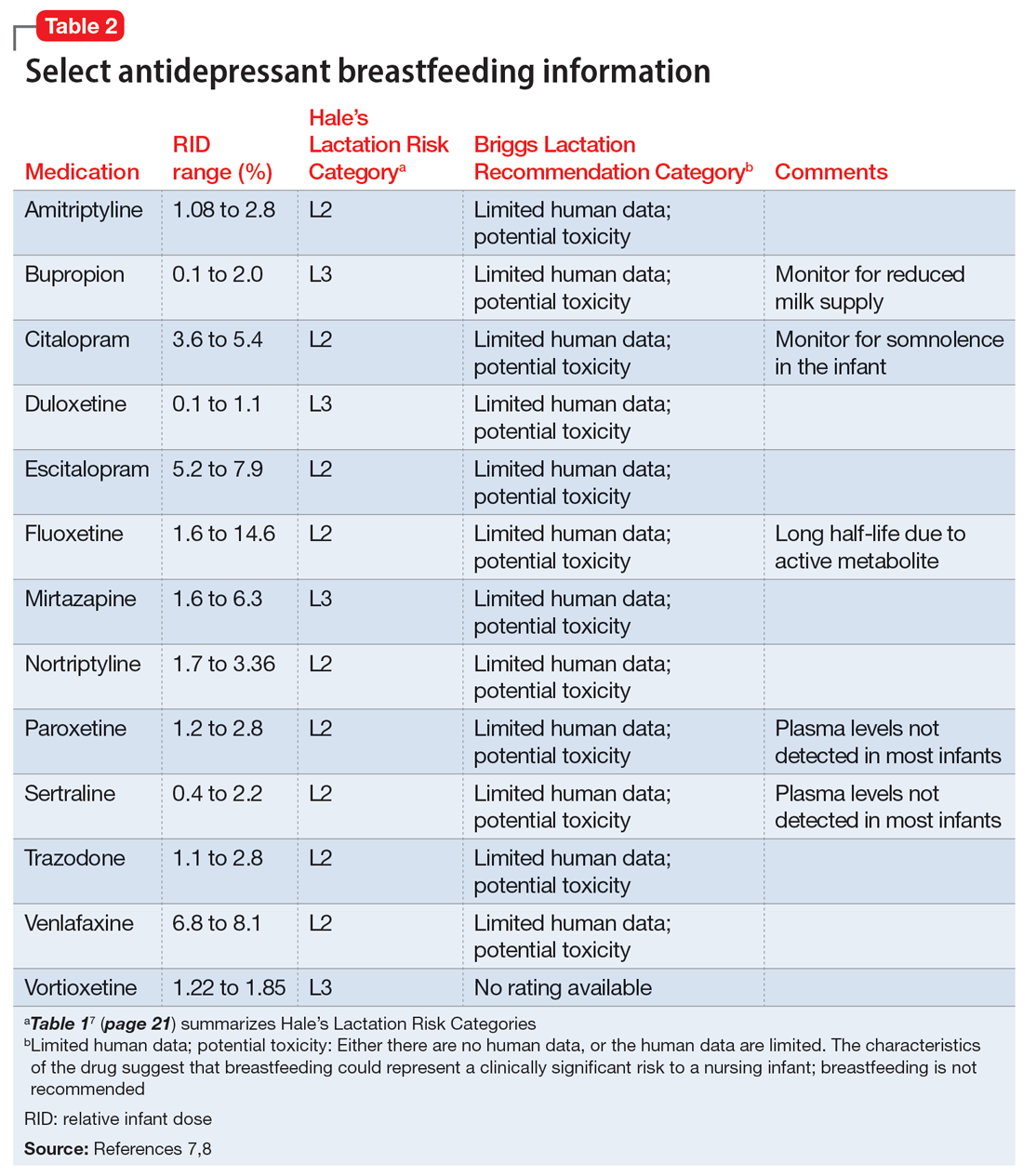
CASE CONTINUED
Two weeks later and following the use of a variety of resources, Ms. D’s treatment team finds that mirtazapine is rated Probably Compatible (L3 in Hale’s Lactation Risk Categories), with an M/P ratio of 0.76.7 The RID of mirtazapine ranges from 1.6% to 6.3%, and limited data from infants exposed to maternal use of mirtazapine during breastfeeding have not shown adverse effects.5 The treatment team administers the EDPS to Ms. D again and she scores 18. Given Ms. D’s previous remission with mirtazapine, current severity of depressive symptoms, and the risk/benefit assessment from lactation resources, the decision is made to restart mirtazapine 15 mg/d at bedtime with the option to titrate up if indicated. Ms. D plans to continue breastfeeding and will monitor for signs of any adverse effects in her infant. The Figure provides a summary of navigating this individualized decision with patients.

Related Resources
- MotherToBaby. Medication fact sheets, option to contact for no-charge consultation, free patient education information materials. www.mothertobaby.org
- Reprotox. Summaries on effects of medications on pregnancy, reproduction, and development (subscription required). www.reprotox.org
Drug Brand Names
Bupropion • Wellbutrin
Citalopram • Celexa
Duloxetine • Cymbalta
Escitalopram • Lexapro
Fluoxetine • Prozac
Mirtazapine • Remeron
Nortriptyline • Pamelor
Paroxetine • Paxil
Sertraline • Zoloft
Trazodone • Oleptro
Venlafaxine • Effexor
Vortioxetine • Trintellix
1. American Academy of Pediatrics. American Academy of Pediatrics calls for more support for breastfeeding mothers within updated policy recommendations. June 27, 2022. Accessed April 7, 2023. https://www.aap.org/en/news-room/news-releases/aap/2022/american-academy-of-pediatrics-calls-for-more-support-for-breastfeeding-mothers-within-updated-policy-recommendations
2. World Health Organization. Breastfeeding recommendations. Accessed April 7, 2023. https://www.who.int/health-topics/breastfeeding#tab=tab_2
3. Margiotta C, Gao J, O’Neil S, et al. The economic impact of untreated maternal mental health conditions in Texas. BMC Pregnancy Childbirth. 2022;22(1):700. doi:10.1186/s12884-022-05001-6
4. Freeman MP, Farchione T, Yao L, et al. Psychiatric medications and reproductive safety: scientific and clinical perspectives pertaining to the US FDA pregnancy and lactation labeling rule. J Clin Psychiatry. 2018;79(4):18ah38120.
5. Drugs and Lactation Database (LactMed). National Library of Medicine (US); 2011. Updated April 18, 2016. Accessed September 29, 2022. https://www.ncbi.nlm.nih.gov/books/NBK501922/
6. InfantRisk Center Resources. InfantRisk Center at Texas Tech University Health Sciences Center. Accessed September 29, 2022. https://www.infantrisk.com/infantrisk-center-resources
7. Hale TW, Krutsch K. Hale’s Medications and Mother’s Milk 2023: A Manual of Lactational Pharmacology. Springer Publishing; 2023.
8. Briggs GG, Freeman RK, Towers CV, et al. Briggs Drugs in Pregnancy and Lactation: A Reference Guide to Fetal and Neonatal Risk. 12th ed. Lippincott Williams & Wilkins; 2021.
Ms. D, age 32, recently gave birth to her second child. Her psychiatric history includes major depressive disorder. She had been stable on mirtazapine 30 mg at bedtime for 3 years. Based on clinical stability and patient preference, Ms. D elected to taper off mirtazapine 1 month prior to delivery. Now at 1 month postdelivery, Ms. D notes the reemergence of her depressive symptoms; during her child’s latest pediatrician visit, she scores 15 on the Edinburgh Postnatal Depression Scale (EPDS). She breastfeeds her baby and wants more information on the safety of taking an antidepressant while breastfeeding.
Ms. D discusses her previous use of mirtazapine with her treatment team. The team reviews the available resources with Ms. D and together they plan to make a shared decision regarding treatment of her depression at her next appointment.
The American Academy of Pediatrics1 and World Health Organization2 recommend exclusive breastfeeding of infants for their first 6 months of life and support it as a complement to other foods through and beyond age 2. Untreated conditions such as postpartum depression impact maternal well-being and may interfere with parenting and child development. In fact, untreated maternal mental health leads to an increased risk of suicide, reduced maternal economic productivity, and worsened health for both mother and child.3
Because many women experience psychiatric symptoms before they become pregnant as well as during the perinatal period, questions often arise regardingthe use of psychiatric medications—specifically antidepressants—and their safety in patients who are breastfeeding. Key considerations regarding medication management should include the patient’s previous response to medications, the risks of untreated maternal mental illness, and evidence regarding risks and benefits in lactation. This article summarizes where to find evidence-based lactation information, how to interpret that information, and what information is available for select antidepressants.
Locating lactation information
Start by checking the manufacturer’s medication labeling (“prescribing information”) and medication information resources such as Micromedex (www.micromedexsolutions.com) and Lexicomp (www.wolterskluwer.com/en/solutions/lexicomp). The updated labeling includes a risk/benefit assessment of available data on the risk for continued use of a medication during pregnancy compared to the risk if a medication is discontinued and the disorder goes untreated.4 The “breastfeeding considerations” section of medication labeling include details regarding the presence of the medication and the amount of it in breastmilk, adverse events in infants exposed to the medication through breastmilk, and additional pertinent data as applicable. Lexicomp includes information regarding breastfeeding considerations, and a subscription may also include access to Briggs Drugs in Pregnancy and Lactation’s information pages. Micromedex includes its own lactation safety rating scale score.
Several other resources can help guide clinicians toward patient-specific recommendations. From the National Library of Medicine, LactMed (https://www.ncbi.nlm.nih.gov/books/NBK501922/) allows clinicians to search for specific medications to see what information exists pertaining to medication levels in breastmilk and infant blood as well as potential adverse effects in the nursing infant and/or on lactation and breastmilk.5 LactMed provides information regarding alternative medications to consider and references from which the information was gathered.
Another helpful resource is the InfantRisk Center from Texas Tech University Health Sciences Center, which includes a free call center for parents and clinicians who have questions about medications and breastfeeding (806-352-2519; Monday through Friday, 8
Continue to: How to interpret the information
How to interpret the information
Medication levels in breastmilk are affected by several properties, such as the medication’s molecular weight, protein binding, pKa, and volume of distribution. A few commonly used terms in lactation literature for medications include the relative infant dose (RID) and milk/plasma (M/P) ratio.
RID provides information about relative medication exposure for the infant. It is calculated by dividing the infant’s dose of a medication via breastmilk (mg/kg/d) by the mother’s dose (mg/kg/d).7 Most consider an RID <10% to be safe.7
M/P is the ratio of medication concentration in the mother’s milk divided by the medication concentration in the mother’s plasma. A ratio <1 is preferable and generally indicates that a low level of medication has been transferred to human milk.7
Another factor that can be evaluated is protein binding. Medications that are highly protein-bound do not tend to pass as easily into breastmilk and can minimize infant exposure.
Several risk categorization systems are available, depending upon the resource used to obtain lactation information. One common system is Hale’s Lactation Risk Categories, with 5 safety levels ranging from L1 (breastfeeding compatible) to L5 (hazardous) (Table 17). Briggs et al8 utilize 7 categories to summarize recommendations ranging from breastfeeding-compatible to contraindicated; however, it is important to read the full medication monograph in the context of the rating provided.Table 27,8 provides breastfeeding information from Hale’s7 and from Briggs et al8 for some commonly used antidepressants.

In addition to interpreting available literature, it is also important to consider patient-specific factors, including (but not limited to) the severity of the patient’s psychiatric disorder and their previous response to medication. If a mother achieved remission on a particular antidepressant in the past, it may be preferable to restart that agent rather than trial a new medication.

CASE CONTINUED
Two weeks later and following the use of a variety of resources, Ms. D’s treatment team finds that mirtazapine is rated Probably Compatible (L3 in Hale’s Lactation Risk Categories), with an M/P ratio of 0.76.7 The RID of mirtazapine ranges from 1.6% to 6.3%, and limited data from infants exposed to maternal use of mirtazapine during breastfeeding have not shown adverse effects.5 The treatment team administers the EDPS to Ms. D again and she scores 18. Given Ms. D’s previous remission with mirtazapine, current severity of depressive symptoms, and the risk/benefit assessment from lactation resources, the decision is made to restart mirtazapine 15 mg/d at bedtime with the option to titrate up if indicated. Ms. D plans to continue breastfeeding and will monitor for signs of any adverse effects in her infant. The Figure provides a summary of navigating this individualized decision with patients.

Related Resources
- MotherToBaby. Medication fact sheets, option to contact for no-charge consultation, free patient education information materials. www.mothertobaby.org
- Reprotox. Summaries on effects of medications on pregnancy, reproduction, and development (subscription required). www.reprotox.org
Drug Brand Names
Bupropion • Wellbutrin
Citalopram • Celexa
Duloxetine • Cymbalta
Escitalopram • Lexapro
Fluoxetine • Prozac
Mirtazapine • Remeron
Nortriptyline • Pamelor
Paroxetine • Paxil
Sertraline • Zoloft
Trazodone • Oleptro
Venlafaxine • Effexor
Vortioxetine • Trintellix
Ms. D, age 32, recently gave birth to her second child. Her psychiatric history includes major depressive disorder. She had been stable on mirtazapine 30 mg at bedtime for 3 years. Based on clinical stability and patient preference, Ms. D elected to taper off mirtazapine 1 month prior to delivery. Now at 1 month postdelivery, Ms. D notes the reemergence of her depressive symptoms; during her child’s latest pediatrician visit, she scores 15 on the Edinburgh Postnatal Depression Scale (EPDS). She breastfeeds her baby and wants more information on the safety of taking an antidepressant while breastfeeding.
Ms. D discusses her previous use of mirtazapine with her treatment team. The team reviews the available resources with Ms. D and together they plan to make a shared decision regarding treatment of her depression at her next appointment.
The American Academy of Pediatrics1 and World Health Organization2 recommend exclusive breastfeeding of infants for their first 6 months of life and support it as a complement to other foods through and beyond age 2. Untreated conditions such as postpartum depression impact maternal well-being and may interfere with parenting and child development. In fact, untreated maternal mental health leads to an increased risk of suicide, reduced maternal economic productivity, and worsened health for both mother and child.3
Because many women experience psychiatric symptoms before they become pregnant as well as during the perinatal period, questions often arise regardingthe use of psychiatric medications—specifically antidepressants—and their safety in patients who are breastfeeding. Key considerations regarding medication management should include the patient’s previous response to medications, the risks of untreated maternal mental illness, and evidence regarding risks and benefits in lactation. This article summarizes where to find evidence-based lactation information, how to interpret that information, and what information is available for select antidepressants.
Locating lactation information
Start by checking the manufacturer’s medication labeling (“prescribing information”) and medication information resources such as Micromedex (www.micromedexsolutions.com) and Lexicomp (www.wolterskluwer.com/en/solutions/lexicomp). The updated labeling includes a risk/benefit assessment of available data on the risk for continued use of a medication during pregnancy compared to the risk if a medication is discontinued and the disorder goes untreated.4 The “breastfeeding considerations” section of medication labeling include details regarding the presence of the medication and the amount of it in breastmilk, adverse events in infants exposed to the medication through breastmilk, and additional pertinent data as applicable. Lexicomp includes information regarding breastfeeding considerations, and a subscription may also include access to Briggs Drugs in Pregnancy and Lactation’s information pages. Micromedex includes its own lactation safety rating scale score.
Several other resources can help guide clinicians toward patient-specific recommendations. From the National Library of Medicine, LactMed (https://www.ncbi.nlm.nih.gov/books/NBK501922/) allows clinicians to search for specific medications to see what information exists pertaining to medication levels in breastmilk and infant blood as well as potential adverse effects in the nursing infant and/or on lactation and breastmilk.5 LactMed provides information regarding alternative medications to consider and references from which the information was gathered.
Another helpful resource is the InfantRisk Center from Texas Tech University Health Sciences Center, which includes a free call center for parents and clinicians who have questions about medications and breastfeeding (806-352-2519; Monday through Friday, 8
Continue to: How to interpret the information
How to interpret the information
Medication levels in breastmilk are affected by several properties, such as the medication’s molecular weight, protein binding, pKa, and volume of distribution. A few commonly used terms in lactation literature for medications include the relative infant dose (RID) and milk/plasma (M/P) ratio.
RID provides information about relative medication exposure for the infant. It is calculated by dividing the infant’s dose of a medication via breastmilk (mg/kg/d) by the mother’s dose (mg/kg/d).7 Most consider an RID <10% to be safe.7
M/P is the ratio of medication concentration in the mother’s milk divided by the medication concentration in the mother’s plasma. A ratio <1 is preferable and generally indicates that a low level of medication has been transferred to human milk.7
Another factor that can be evaluated is protein binding. Medications that are highly protein-bound do not tend to pass as easily into breastmilk and can minimize infant exposure.
Several risk categorization systems are available, depending upon the resource used to obtain lactation information. One common system is Hale’s Lactation Risk Categories, with 5 safety levels ranging from L1 (breastfeeding compatible) to L5 (hazardous) (Table 17). Briggs et al8 utilize 7 categories to summarize recommendations ranging from breastfeeding-compatible to contraindicated; however, it is important to read the full medication monograph in the context of the rating provided.Table 27,8 provides breastfeeding information from Hale’s7 and from Briggs et al8 for some commonly used antidepressants.

In addition to interpreting available literature, it is also important to consider patient-specific factors, including (but not limited to) the severity of the patient’s psychiatric disorder and their previous response to medication. If a mother achieved remission on a particular antidepressant in the past, it may be preferable to restart that agent rather than trial a new medication.

CASE CONTINUED
Two weeks later and following the use of a variety of resources, Ms. D’s treatment team finds that mirtazapine is rated Probably Compatible (L3 in Hale’s Lactation Risk Categories), with an M/P ratio of 0.76.7 The RID of mirtazapine ranges from 1.6% to 6.3%, and limited data from infants exposed to maternal use of mirtazapine during breastfeeding have not shown adverse effects.5 The treatment team administers the EDPS to Ms. D again and she scores 18. Given Ms. D’s previous remission with mirtazapine, current severity of depressive symptoms, and the risk/benefit assessment from lactation resources, the decision is made to restart mirtazapine 15 mg/d at bedtime with the option to titrate up if indicated. Ms. D plans to continue breastfeeding and will monitor for signs of any adverse effects in her infant. The Figure provides a summary of navigating this individualized decision with patients.

Related Resources
- MotherToBaby. Medication fact sheets, option to contact for no-charge consultation, free patient education information materials. www.mothertobaby.org
- Reprotox. Summaries on effects of medications on pregnancy, reproduction, and development (subscription required). www.reprotox.org
Drug Brand Names
Bupropion • Wellbutrin
Citalopram • Celexa
Duloxetine • Cymbalta
Escitalopram • Lexapro
Fluoxetine • Prozac
Mirtazapine • Remeron
Nortriptyline • Pamelor
Paroxetine • Paxil
Sertraline • Zoloft
Trazodone • Oleptro
Venlafaxine • Effexor
Vortioxetine • Trintellix
1. American Academy of Pediatrics. American Academy of Pediatrics calls for more support for breastfeeding mothers within updated policy recommendations. June 27, 2022. Accessed April 7, 2023. https://www.aap.org/en/news-room/news-releases/aap/2022/american-academy-of-pediatrics-calls-for-more-support-for-breastfeeding-mothers-within-updated-policy-recommendations
2. World Health Organization. Breastfeeding recommendations. Accessed April 7, 2023. https://www.who.int/health-topics/breastfeeding#tab=tab_2
3. Margiotta C, Gao J, O’Neil S, et al. The economic impact of untreated maternal mental health conditions in Texas. BMC Pregnancy Childbirth. 2022;22(1):700. doi:10.1186/s12884-022-05001-6
4. Freeman MP, Farchione T, Yao L, et al. Psychiatric medications and reproductive safety: scientific and clinical perspectives pertaining to the US FDA pregnancy and lactation labeling rule. J Clin Psychiatry. 2018;79(4):18ah38120.
5. Drugs and Lactation Database (LactMed). National Library of Medicine (US); 2011. Updated April 18, 2016. Accessed September 29, 2022. https://www.ncbi.nlm.nih.gov/books/NBK501922/
6. InfantRisk Center Resources. InfantRisk Center at Texas Tech University Health Sciences Center. Accessed September 29, 2022. https://www.infantrisk.com/infantrisk-center-resources
7. Hale TW, Krutsch K. Hale’s Medications and Mother’s Milk 2023: A Manual of Lactational Pharmacology. Springer Publishing; 2023.
8. Briggs GG, Freeman RK, Towers CV, et al. Briggs Drugs in Pregnancy and Lactation: A Reference Guide to Fetal and Neonatal Risk. 12th ed. Lippincott Williams & Wilkins; 2021.
1. American Academy of Pediatrics. American Academy of Pediatrics calls for more support for breastfeeding mothers within updated policy recommendations. June 27, 2022. Accessed April 7, 2023. https://www.aap.org/en/news-room/news-releases/aap/2022/american-academy-of-pediatrics-calls-for-more-support-for-breastfeeding-mothers-within-updated-policy-recommendations
2. World Health Organization. Breastfeeding recommendations. Accessed April 7, 2023. https://www.who.int/health-topics/breastfeeding#tab=tab_2
3. Margiotta C, Gao J, O’Neil S, et al. The economic impact of untreated maternal mental health conditions in Texas. BMC Pregnancy Childbirth. 2022;22(1):700. doi:10.1186/s12884-022-05001-6
4. Freeman MP, Farchione T, Yao L, et al. Psychiatric medications and reproductive safety: scientific and clinical perspectives pertaining to the US FDA pregnancy and lactation labeling rule. J Clin Psychiatry. 2018;79(4):18ah38120.
5. Drugs and Lactation Database (LactMed). National Library of Medicine (US); 2011. Updated April 18, 2016. Accessed September 29, 2022. https://www.ncbi.nlm.nih.gov/books/NBK501922/
6. InfantRisk Center Resources. InfantRisk Center at Texas Tech University Health Sciences Center. Accessed September 29, 2022. https://www.infantrisk.com/infantrisk-center-resources
7. Hale TW, Krutsch K. Hale’s Medications and Mother’s Milk 2023: A Manual of Lactational Pharmacology. Springer Publishing; 2023.
8. Briggs GG, Freeman RK, Towers CV, et al. Briggs Drugs in Pregnancy and Lactation: A Reference Guide to Fetal and Neonatal Risk. 12th ed. Lippincott Williams & Wilkins; 2021.
Bipolar disorder: The foundational role of mood stabilizers
Bipolar disorder (BD) is a recurrent, life-long psychiatric illness affecting nearly 2% of the world population1,2 that is characterized by episodes of mania and depression interspersed among periods of relative mood stability.3 The illness causes an enormous health burden, which makes understanding its pathophysiology and treatment patterns a substantial priority.4 In the 1950s, lithium was found to be effective for treating acute manic episodes and preventing relapse in BD.5 Since then, valproate and carbamazepine also have been FDA-approved for treating mania.6,7 Antipsychotics have also shown evidence of efficacy in BD treatment,8,9 particularly for use in acute settings for more rapid effect or for a limited duration,10 which has led some to refer to them as “mood stabilizers.”11
In this article, we describe changes in trends of prescribing medications to treat BD, the role of ion dysregulation in the disorder, and how a better understanding of this dysregulation might impact the choice of treatment.
Changes in pharmacotherapy for bipolar disorder
From 1997 through 2016, the use of lithium for BD decreased from >30% of patients to 17.6% (with a nadir of 13.9% from 2009 to 2012).12 Over the same period, the use of nonlithium mood stabilizers decreased from 30.4% to approximately 4.8%, while second-generation antipsychotic (SGAs) use increased from 12.4% to 50.4%.12 Distressingly, antidepressant use increased from approximately 47% to 56.8%, and antidepressant use without concomitant mood stabilizers increased from 38% to 40.8%, although the rate of antidepressants without either a mood stabilizer or an antipsychotic remained relatively stable (14.9% to 16.8%).12 In randomized trials, when added to mood stabilizers, antidepressants have consistently failed to separate from placebo,13-15 but they can destabilize the illness, resulting in increases in mania, depression, and subsyndromal mixed symptoms.16-18
It is easy to understand clinicians’ attempts to address their patients’ distress due to depressive symptoms that do not resolve with mood stabilizers.19,20 Similarly, the increased use of antipsychotics is driven by evidence that antipsychotics are effective for treating bipolar depression and preventing the recurrence of manic and (for some antipsychotics) depressive episodes.21,22 However, long-term antipsychotic use causes brain volume change in patients with schizophrenia23 or major depressive disorder24 and in nonhuman primates25,26; metabolic abnormalities27-31; and cardiovascular adverse effects.32 Antipsychotics are believed to be associated with withdrawal psychosis.33,34 In the head-to-head Clinical Health Outcomes Initiative in Comparative Effectiveness for Bipolar Disorder (Bipolar CHOICE) study, quetiapine was as effective as lithium but associated with more adverse effects.35 Importantly, the estimated disability-adjusted life years of patients with BD increased by 54.4% from 6.02 million in 1990 to 9.29 million in 2017, which is greater than the increase in the incidence of BD (47.74%) over the same time.36 This means that despite the dramatic increase in treatment options for people with BD, functional outcomes have declined.
One major difference between antipsychotics and mood stabilizers is that antipsychotics do not alter the underlying abnormal pathology of BD.37 An ideal pharmacologic intervention is one that corrects a known pathophysiologic anomaly of the condition being treated. There are no demonstrated abnormalities in the dopamine or serotonin systems in individuals with BD, but long-term use of antipsychotics may create dopaminergic alterations.33 One of the most reproducible biomarkers associated with manic and bipolar depressed mood states is increased intracellular sodium38,39 and reduced ability to correct a sodium challenge.40-42 By normalizing intracellular sodium levels, lithium and the mood-stabilizing anticonvulsants uniquely and specifically counter known physiologic abnormalities in patients with BD.37,43
The role of ion dysregulation
The pathophysiology of BD remains elusive. A multitude of lines of evidence link BD to abnormal neuroimaging findings,22,44,45 oxidative stress,46 inflammation,47 and mitochondrial disease,48 but there is still no unifying understanding of these findings. Ion dysregulation appears to be central to understanding and treating BD.38,39
Despite extensive genetic studies, no genes have been identified that mediate >5% of the risk for BD. Nonetheless, 74% of all genes identified as mediating risk for BD code for proteins essential for the regulation of ion transport and membrane potential.49 The 2 genes that contribute the greatest risk are CACNA1C and ANK3, which code for a calcium channel and a cytoskeletal protein, respectively.50ANK3 codes for ankyrin G, which plays a role in proper coupling of the voltage-gated sodium channels to the cytoskeleton.51 An additional risk gene, TRANK1, contains multiple ankyrin-like repeat domains, which suggests some shared functions with ANK3.52 More importantly, the most reproducible pathophysiologic findings in BD are dysregulation of sodium, potassium, hydrogen, and calcium transport, with consequent alteration of depolarization potential, neuronal excitability, and calcium-mediated processes.38,39,53-56 For example, increased sodium and calcium within cells have been observed in both mania and bipolar depression, and these levels normalize during euthymia. All medications that are effective for treating BD may reduce intracellular sodium or calcium; traditional mood stabilizers do so directly by inhibiting voltage-sensitive sodium channels in an activity-dependent manner or displacing intracellular sodium,43,57 whereas antipsychotics do so indirectly by increasing sodium pump activity through inhibition of second messengers of the dopamine D2 family of receptors.37
Continue to: The extent of ion dysregulation...
The extent of ion dysregulation is directly associated with the expressed mood state of the illness. A small reduction in the activity of the sodium pump results in a small increase in intracellular sodium (approximately 10 mM).39,58 This led to the hypothesis that increased intracellular sodium causes the transmembrane potential to increase closer to membrane depolarization threshold, which increases excitability of affected neurons.38,39,58 Neurons are likely to fire and propagate signals more easily, which may manifest as symptoms of mania, such as increased energy, activity, lability, excitability, irritability, tangentiality, and looseness of associations. As the process of increased intracellular sodium progresses, a minority of neurons are expected to have their transmembrane potentials depolarize sufficiently for the resting membrane potential to go beyond threshold potential.59 Such neurons are in a state of constant depolarization (also known as depolarization block), which disrupts neuronal circuits. The difficulty in progression of these signals results in the classic bipolar depression symptoms of low energy, reduced activity, and slowing of all brain activity that is seen as psychomotor slowing.38
Implications for treatment
Medications for treating bipolar illness include lithium, anticonvulsants, benzodiazepines, first-generation antipsychotics, and SGAs.37,43
Mood stabilizers (lithium and certain anticonvulsants) correct the previously mentioned sodium abnormality by reducing sodium entry into the cell in an activity-dependent manner.43 As the only agents that directly address a known pathophysiologic abnormality, they are foundational in the treatment of BD.60 Lithium effectively treats acute mania and prevents relapse.61 It preferentially targets the active neurons, entering through both voltage-responsive and neurotransmitter-coupled channels.43,62 This results in an increase of intracellular lithium concentrations to as much as 8 times that of the extracellular concentration.63 These ions displace intracellular sodium ions in a 1:1 ratio, which results in a reduced intracellular sodium concentration that reduces the excitability of neurons.43,57,62
Substantial evidence supports the use of valproic acid for initial and maintenance treatment of BD.64 It inhibits the voltage-sensitive sodium channel when the channel is open, which results in an activity-dependent action that selectively impacts rapidly firing neurons.43 The voltage-gated sodium channels exist nearly exclusively on the axon, beyond the hillock65; as such, valproic acid will only inhibit neurons that fire, whereas lithium accumulates throughout the neuron and will affect depolarization in the neuronal soma as well as the firing in the axon.43 Additionally, valproic acid has been observed to enhance gamma-aminobutyric acid (GABA) levels and transmission.43,66,67 A meta-analysis that included 6 randomized controlled trials illustrated that, acutely, valproate was not different from lithium’s overall efficacy (RR 1.02; 95% CI, 0.87 to 1.20), but was associated with reduced dropout rates compared with placebo or lithium (RR 0.82; 95% CI, 0.71 to 0.95 and RR 0.87; 95% CI, 0.77 to 0.98, respectively).64
Lamotrigine is an anticonvulsant used for initial and maintenance treatment of BD, with greater efficacy for depressive episodes68; it also has notable effect for treating bipolar depression, although it is not FDA-approved for this indication.69 Lamotrigine inhibits sodium influx by binding to open voltage-gated sodium channels70 but also appears to reduce N-methyl-D-aspartate–mediated sodium entry,71 thereby acting both prehillock and posthillock.
Continue to: Carbamazepine is an anticonvulsant...
Carbamazepine is an anticonvulsant FDA-approved for treating BD.7 Like valproate, it acts by inhibiting voltage-gated sodium channels in an activity-dependent manner,72 which means it preferentially inhibits the most active neurons and those with higher intracellular sodium.43
Benzodiazepines, which have shown to be effective for treating acute mania,73 potentiate synaptic GABA receptors accruing an elevation in intracellular chloride influx.74 Despite acute efficacy, benzodiazepine use is limited because these agents are associated with worsening long-term, substance use–related outcomes.75,76
Antipsychotics are effective for treating mood disorders,60,76 and their use has been rising dramatically.12 The antimanic effect of all antipsychotics is believed to be mediated through dopamine D2 blockade, since use of a dose sufficient to block D2 receptors is required, and haloperidol, which acts exclusively on the D2 receptor, is equal to SGAs in its antimanic effect.77 Blockade of the D2 receptor will increase the activity of the sodium pump (sodium and potassium-activated adenosine triphosphatase) thus reducing intracellular sodium and calcium concentrations.37 When antipsychotics are used as antidepressants, they are generally used at doses lower than those used to treat mania.78
Antipsychotics are effective for treating BD, and may work more quickly than other agents for treating acute mania.79 However, maintenance or prevention trials tend to favor mood stabilizers.35,60,80 Several add-on studies have found the combination of a mood stabilizer plus an antipsychotic is superior to a mood stabilizer alone or an antipsychotic alone.81
An argument for mood stabilizers
Evidence suggests mood stabilizers and other approaches, such as antipsychotics, are almost equivalent for treating acute mania, with a small clinical advantage of mood stabilizers for preventing relapse. In general, current treatment guidelines do not distinguish mood stabilizers from antipsychotics as the first-line treatment.82 Over the past 20 years, antipsychotic use has increased while mood stabilizer use has decreased, so that presently a patient with BD is more likely to be prescribed an antipsychotic than a mood stabilizer.12 Over the same time, dysfunction among patients with BD has increased.33 Antipsychotics are appealing because they appear to be equally effective and generally well tolerated. But these agents cause problems that are difficult to see in routine visits, such as metabolic27-31 and cardiovascular adverse effects29 as well as reductions in brain volume.23-26 Mechanistic research suggests that mood stabilizers directly correct known pathophysiologic anomalies with additional protective effects, whereas antipsychotics appear to create new abnormalities and contribute to medical problems. Clinicians need to look beyond the similarities in acute efficacy and make a more broadly supported, evidence-based choice for managing BD, which clearly places mood stabilizers as the first-line agent and antipsychotics as reasonable alternatives. At a minimum, mood stabilizers should be viewed as the foundation to which antipsychotics can be added.
Bottom Line
Traditional mood stabilizers—lithium and some anticonvulsants—are the only agents that directly address physiologic abnormalities associated with both mania and bipolar depression, including mood state–associated elevations of intracellular sodium. Because of their specificity, these agents maximize mood stabilization and minimize adverse effects.
Related Resources
- Karas A, Stummer L, Freedberg A. Psychiatric and nonpsychiatric indications for mood stabilizers and select antiepileptics. Current Psychiatry. 2022;21(4):34-38. doi:10.12788/cp.0230
- Koch J. Mood stabilizers: balancing tolerability, serum levels, and dosage. Current Psychiatry. 2021;20(7):37-40. doi:10.12788/cp.0147
Drug Brand Names
Carbamazepine • Tegretol
Haloperidol • Haldol
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Quetiapine • Seroquel
Valproate • Depakote, Depakene
1. Whiteford HA, Degenhardt L, Rehm J, et al. Global burden of disease attributable to mental and substance use disorders: findings from the Global Burden of Disease Study 2010. Lancet. 2013;382(9904):1575-1586. doi:10.1016/S0140-6736(13)61611-6
2. Merikangas KR, Jin R, He JP, et al. Prevalence and correlates of bipolar spectrum disorder in the world mental health survey initiative. Arch Gen Psychiatry. 2011;68(3):241-251. doi:10.1001/archgenpsychiatry.2011.12
3. Müller JK, Leweke FM. Bipolar disorder: clinical overview. Article in English, German. Med Monatsschr Pharm. 2016;39(9):363-369.
4. Smith DJ, Whitham EA, Ghaemi SN. Bipolar disorder. Handb Clin Neurol. 2012;106:251-263. doi:10.1016/B978-0-444-52002-9.00015-2
5. Goodwin FK, Ghaemi SN. The impact of the discovery of lithium on psychiatric thought and practice in the USA and Europe. Aust N Z J Psychiatry. 1999;33 Suppl:S54-S64. doi:10.1111/j.1440-1614.1999.00669.x
6. Pope HG, McElroy SL, Keck PE, et al. Valproate in the treatment of acute mania. A placebo-controlled study. Arch Gen Psychiatry. 1991;48(1):62-68. doi:10.1001/archpsyc.1991.01810250064008
7. Weisler RH, Keck PE Jr, Swann AC, et al. Extended-release carbamazepine capsules as monotherapy for acute mania in bipolar disorder: a multicenter, randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2005;66(3):323-330. doi:10.4088/jcp.v66n0308
8. Tarr GP, Glue P, Herbison P. Comparative efficacy and acceptability of mood stabilizer and second generation antipsychotic monotherapy for acute mania--a systematic review and meta-analysis. J Affect Disord. 2011;134(1-3):14-19. doi:10.1016/j.jad.2010.11.009
9. Pahwa M, Sleem A, Elsayed OH, et al. New antipsychotic medications in the last decade. Curr Psychiatry Rep. 2021;23(12):87.
10. Correll CU, Sheridan EM, DelBello MP. Antipsychotic and mood stabilizer efficacy and tolerability in pediatric and adult patients with bipolar I mania: a comparative analysis of acute, randomized, placebo-controlled trials. Bipolar Disord. 2010;12(2):116-141. doi:10.1111/j.1399-5618.2010.00798.x
11. Rybakowski JK. Two generations of mood stabilizers. Int J Neuropsychopharmacol. 2007;10:709-711. doi:10.1017/s146114570700795x
12. Rhee TG, Olfson M, Nierenberg AA, et al. 20-year trends in the pharmacologic treatment of bipolar disorder by psychiatrists in outpatient care settings. Am J Psychiatry. 2020;177(8):706-715. doi:10.1176/appi.ajp.2020.19091000
13. El-Mallakh RS. Adjunctive antidepressant treatment for bipolar depression. N Engl J Med. 2007;357(6):615; author reply 615-616.
14. Sachs GS, Nierenberg AA, Calabrese JR, et al. Effectiveness of adjunctive antidepressant treatment for bipolar depression. N Engl J Med. 2007;356(17):1711-1722. doi:10.1056/NEJMoa064135
15. Ghaemi SN, Whitham EA, Vohringer PA, et al. Citalopram for acute and preventive efficacy in bipolar depression (CAPE-BD): a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2021;82(1):19m13136. doi:10.4088/JCP.19m13136
16. El-Mallakh RS, Ghaemi SN, Sagduyu K, et al. Antidepressant-associated chronic irritable dysphoria (ACID) in STEP-BD patients. J Affect Disord. 2008;111(2-3):372-377. doi:10.1016/j.jad.2008.03.025
17. Ghaemi SN, Ostacher MM, El-Mallakh RS, et al. Antidepressant discontinuation in bipolar depression: a systematic treatment enhancement program for bipolar disorder (STEP-BD) randomized clinical trial of long-term effectiveness and safety. J Clin Psychiatry. 2010;71(4):372-380.
18. Strejilevich SA, Martino DJ, Marengo E, et al. Long-term worsening of bipolar disorder related with frequency of antidepressant exposure. Ann Clin Psychiatry. 2011;23(3):186-192.
19. Pacchiarotti I, Bond DJ, Baldessarini RJ, et al. The International Society of Bipolar Disorders (ISBD) task force report on antidepressant use in bipolar disorders. Am J Psychiatry. 2013;170(11):1249-1262. doi:10.1176/appi.ajp.2013.13020185
20. McIntyre RS, Calabrese JR. Bipolar depression: the clinical characteristics and unmet needs of a complex disorder. Curr Med Res Opin. 2019;35(11):1993-2005.
21. Fornaro M, Stubbs B, De Berardis D, et al. Atypical antipsychotics in the treatment of acute bipolar depression with mixed features: a systematic review and exploratory meta-analysis of placebo-controlled clinical trials. Int J Mol Sci. 2016;17(2):241. doi:10.3390/ijms17020241
22. Lindström L, Lindström E, Nilsson M, et al. Maintenance therapy with second generation antipsychotics for bipolar disorder – a systematic review and meta-analysis. J Affect Disord. 2017;213:138-150. doi:10.1016/j.jad.2017.02.012
23. Ho BC, Andreasen NC, Ziebell S, et al. Long-term antipsychotic treatment and brain volumes: a longitudinal study of first-episode schizophrenia. Arch Gen Psychiatry. 2011;68(2):128-137. doi:010.1001/archgenpsychiatry.2010.199
24. Voineskos AN, Mulsant BH, Dickie EW, et al. Effects of antipsychotic medication on brain structure in patients with major depressive disorder and psychotic features: neuroimaging findings in the context of a randomized placebo-controlled clinical trial. JAMA Psychiatry. 2020;77(7):674-683. doi:10.1001/jamapsychiatry.2020.0036
25. Konopaske GT, Bolo NR, Basu AC, et al. Time-dependent effects of haloperidol on glutamine and GABA homeostasis and astrocyte activity in the rat brain. Psychopharmacology (Berl). 2013;230(1):57-67. doi:10.1007/s00213-013-3136-3
26. Dorph-Petersen KA, Pierri JN, Perel JM, et al. The influence of chronic exposure to antipsychotic medications on brain size before and after tissue fixation: a comparison of haloperidol and olanzapine in macaque monkeys. Neuropsychopharmacology. 2005;30(9):1649-1661. doi:10.1038/sj.npp.1300710
27. McIntyre RS, Mancini DA, Basile VS, et al. Antipsychotic-induced weight gain: bipolar disorder and leptin. J Clin Psychopharmacol. 2003;23(4):323-327. doi:10.1097/01.jcp.0000085403.08426.f4
28. McIntyre RS, Konarski JZ, Wilkins K, et al. Obesity in bipolar disorder and major depressive disorder: results from a national community health survey on mental health and well-being. Can J Psychiatry. 2006;51(5):274-280. doi:10.1177/070674370605100502
29. McIntyre RS, Cha DS, Kim RD, et al. A review of FDA-approved treatment options in bipolar depression. CNS Spectr. 2013;18(Suppl 1):4-20. doi:10.1017/S1092852913000746
30. Barton BB, Segger F, Fischer K, et al. Update on weight-gain caused by antipsychotics: a systematic review and meta-analysis. Expert Opin Drug Saf. 2020;19(3):295-314. doi:10.1080/14740338.2020.1713091
31. Doane MJ, Bessonova L, Friedler HS, et al. Weight gain and comorbidities associated with oral second-generation antipsychotics: analysis of real-world data for patients with schizophrenia or bipolar I disorder. BMC Psychiatry. 2022;22(1):114. doi:10.1186/s12888-022-03758-w
32. Buckley NA, Sanders P. Cardiovascular adverse effects of antipsychotic drugs. Drug Saf. 2000;23(3):215-228. doi:10.2165/00002018-200023030-00004
33. Ali Z, Roque A, El-Mallakh RS. A unifying theory for the pathoetiologic mechanism of tardive dyskinesia. Med Hypotheses. 2020;140:109682. doi:10.1016/j.mehy.2020.109682
34. Sleem A, El-Mallakh RS. Adaptive changes to antipsychotics: their consequences and how to avoid them. Curr Psychiatry. 2022;21(7):46-50,52. doi: 10.12788/cp.0262
35. Nierenberg AA, McElroy SL, Friedman ES, et al. Bipolar CHOICE (Clinical Health Outcomes Initiative in Comparative Effectiveness): a pragmatic 6-month trial of lithium versus quetiapine for bipolar disorder. J Clin Psychiatry. 2016;77(1):90-99. doi:10.4088/JCP.14m09349
36. He H, Hu C, Ren Z, et al. Trends in the incidence and DALYs of bipolar disorder at global, regional, and national levels: results from the global burden of disease study 2017. J Psychiatr Res. 2020;125:96-105. doi:10.1016/j.jpsychires.2020.03.015
37. Roberts RJ, Repass R, El-Mallakh RS. Effect of dopamine on intracellular sodium: a common pathway for pharmacological mechanism of action in bipolar illness. World J Biol Psychiatry. 2010;11(2 Pt 2):181-187. doi:10.1080/15622970902718774
38. El-Mallakh RS, Wyatt RJ. The Na, K-ATPase hypothesis for bipolar illness. Biol Psychiatry. 1995;37(4):235-244. doi:10.1016/0006-3223(94)00201-D
39. El-Mallakh RS, Yff T, Gao Y. Ion dysregulation in the pathogenesis of bipolar disorder. Ann Depress Anxiety. 2016;3(1):1076.
40. Li R, El-Mallakh RS. Differential response of bipolar and normal control lymphoblastoid cell sodium pump to ethacrynic acid. J Affect Disord. 2004;80(1):11-17. doi:10.1016/S0165-0327(03)00044-2
41. Woodruff DB, El-Mallakh RS, Thiruvengadam AP. Validation of a diagnostic screening blood test for bipolar disorder. Ann Clin Psychiatry. 2012;24(2):135-139.
42. Gao Y, Lohano K, Delamere NA, et al. Ethanol normalizes glutamate-induced elevation of intracellular sodium in olfactory neuroepithelial progenitors from subjects with bipolar illness but not nonbipolar controls: biologic evidence for the self-medication hypothesis. Bipolar Disord. 2019;21(2):179-181. doi:10.1111/bdi.12737
43. El-Mallakh RS, Huff MO. Mood stabilizers and ion regulation. Harv Rev Psychiatry. 2001;9(1):23-32. doi:10.1080/10673220127873
44. Phillips ML, Swartz HA. A critical appraisal of neuroimaging studies of bipolar disorder: toward a new conceptualization of underlying neural circuitry and a road map for future research. Am J Psychiatry. 2014;171(8):829-843. doi:10.1176/appi.ajp.2014.13081008
45. Hibar DP, Westlye LT, Doan NT, et al. Cortical abnormalities in bipolar disorder: an MRI analysis of 6503 individuals from the ENIGMA Bipolar Disorder Working Group. Mol Psychiatry. 2018;23(4):932-942. doi:10.1038/mp.2017.73
46. Brown NC, Andreazza AC, Young LT. An updated meta-analysis of oxidative stress markers in bipolar disorder. Psychiatry Res. 2014;218(1-2):61-68. doi:10.1016/j.psychres.2014.04.005
47. Benedetti F, Aggio V, Pratesi ML, et al. Neuroinflammation in bipolar depression. Front Psychiatry. 2020;11:71. doi:10.3389/fpsyt.2020.00071
48. Andreazza AC, Duong A, Young LT. Bipolar disorder as a mitochondrial disease. Biol Psychiatry. 2018;83(9):720-721. doi:10.1016/j.biopsych.2017.09.018
49. Askland KD. Toward a biaxial model of “bipolar” affective disorders: further exploration of genetic, molecular and cellular substrates. J Affect Disord. 2006;94(1-3):35-66. doi:10.1016/j.jad.2006.01.033
50. Ferreira MA, O’Donovan MC, Meng YA, et al; Wellcome Trust Case Control Consortium. Collaborative genome-wide association analysis supports a role for ANK3 and CACNA1C in bipolar disorder. Nat Genet 2008;40(9):1056-1058. doi:10.1038/ng.209
51. Salvi AM, Bays JL, Mackin SR, et al. Ankyrin G organizes membrane components to promote coupling of cell mechanics and glucose uptake. Nat Cell Biol. 2021;23(5):457-466. doi:10.1038/s41556-021-00677-y
52. Gargus JJ. Ion channel functional candidate genes in multigenic neuropsychiatric disease. Biol Psychiatry. 2006;60(2):177-185. doi:10.1016/j.biopsych.2005.12.008
53. Dubovsky SL, Murphy J, Thomas M, et al. Abnormal intracellular calcium ion concentration in platelets and lymphocytes of bipolar patients. Am J Psychiatry 1992;149(1):118-120. doi:10.1176/ajp.149.1.118
54. Blaustein MP. Physiological effects of endogenous ouabain: control of intracellular Ca2+ stores and cell responsiveness. Am J Physiol. 1993;264(6 Pt 1):C1367–C1387. doi:10.1152/ajpcell.1993.264.6.C1367
55. El-Mallakh RS, Li R, Worth CA, et al. Leukocyte transmembrane potential in bipolar illness. J Affect Disord. 1996;41(1):33-37. doi:10.1016/0165-0327(96)00063-8
56. El-Mallakh RS, Gao Y, You P. Role of endogenous ouabain in the etiology of bipolar disorder. Int J Bipolar Disord. 2021;9(1):6. doi:10.1186/s40345-020-00213-1
57. Huang X, Lei Z, El‐Mallakh RS. Lithium normalizes elevated intracellular sodium. Bipolar Disord. 2007;9(3):298-300. doi:10.1111/j.1399-5618.2007.00429.x
58. Shaw DM. Mineral metabolism, mania, and melancholia. Br Med J. 1966;2(5508):262-267. doi:10.1136/bmj.2.5508.262
59. Qian K, Yu N, Tucker KR, et al. Mathematical analysis of depolarization block mediated by slow inactivation of fast sodium channels in midbrain dopamine neurons. J Neurophysiol. 2014;112(11):2779-2790. doi:10.1152/jn.00578.2014
60. Sleem A, El-Mallakh RS. Advances in the psychopharmacotherapy of bipolar disorder type I. Exp Opin Pharmacother. 2021;22(10):1267-1290. doi:10.1080/14656566.2021.1893306
61. Malhi GS., Tanious M, Das P, et al. Potential mechanisms of action of lithium in bipolar disorder. CNS Drugs. 2013;27(2):135-153. doi:10.1007/s40263-013-0039-0
62. Armett CJ, Ritchie JM. On the permeability of mammalian non-myelinated fibers to sodium and to lithium ions. J Physiol. 1963;165(1):130-140. doi:10.1113/jphysiol.1963.sp007047
63. Kabakov AY, Karkanias NB, Lenox RH, et al. Synapse-specific accumulation of lithium in intracellular microdomains: a model for uncoupling coincidence detection in the brain. Synapse. 1998;28(4):271-279. doi:10.1002/(SICI)1098-2396(199804)28:4<271::AID-SYN2>3.0.CO;2-6
64. Cipriani A, Reid K, Young AH, et al. Valproic acid, valproate and divalproex in the maintenance treatment of bipolar disorder. Cochrane Database Syst Rev. 2013;2013(10):CD003196. doi:10.1002/14651858.CD003196.pub2
65. Lai HC, Jan LY. The distribution and targeting of neuronal voltage-gated ion channels. Nat Rev Neurosci. 2006;7(7):548-562. doi:10.1038/nrn1938
66. Löscher W, Schmidt D. Increase of human plasma GABA by sodium valproate. Epilepsia. 1980;21(6):611-615. doi:10.1111/j.1528-1157.1980.tb04314.x
67. Owens MJ, Nemeroff CB. Pharmacology of valproate. Psychopharmacol Bull. 2003;37(Suppl 2):17-24.
68. Calabrese JR, Vieta E, Shelton MD. Latest maintenance data on lamotrigine in bipolar disorder. Eur Neuropsychopharmacol. 2003;13(Suppl 2):S57-S66. doi:10.1016/s0924-977x(03)00079-8
69. Geddes JR, Calabrese JR, Goodwin GM. Lamotrigine for treatment of bipolar depression: independent meta-analysis and meta-regression of individual patient data from five randomised trials. Br J Psychiatry. 2009;194(1):4-9. doi:10.1192/bjp.bp.107.048504
70. Nakatani Y, Masuko H, Amano T. Effect of lamotrigine on Na(v)1.4 voltage-gated sodium channels. J Pharmacol Sci. 2013;123(2):203-206. doi:10.1254/jphs.13116sc
71. Ramadan E, Basselin M, Rao JS, et al. Lamotrigine blocks NMDA receptor-initiated arachidonic acid signalling in rat brain: implications for its efficacy in bipolar disorder. Int J Neuropsychopharmacol. 2012;15(7):931-943. doi:10.1017/S1461145711001003
72. Jo S, Bean BP. Sidedness of carbamazepine accessibility to voltage-gated sodium channels. Mol Pharmacol. 2014;85(2):381-387. doi:10.1124/mol.113.090472
73. Curtin F, Schulz P. Clonazepam and lorazepam in acute mania: a Bayesian meta-analysis. J Affect Disord 2004;78(3):201-208. doi:10.1016/S0165-0327(02)00317-8
74. Edwards R, Stephenson U, Flewett T. Clonazepam in acute mania: a double blind trial. Aust N Z J Psychiatry 1991;25(2):238-242. doi:10.3109/00048679109077740
75. Lin SC, Chen CC, Chen YH, et al. Benzodiazepine prescription among patients with severe mental illness and co-occurring alcohol abuse/dependence in Taiwan. Hum Psychopharmacol. 2011;26(3):201-207. doi:10.1002/hup.1193
76. Prisciandaro JJ, Brown DG, Brady KT, et al. Comorbid anxiety disorders and baseline medication regimens predict clinical outcomes in individuals with co-occurring bipolar disorder and alcohol dependence: results of a randomized controlled trial. Psychiatry Res. 2011;188(3):361-365. doi:10.1016/j.psychres.2011.04.030
77. Ashok AH, Marques TR, Jauhar S, et al. The dopamine hypothesis of bipolar affective disorder: the state of the art and implications for treatment. Mol Psychiatry. 2017;22(5):666-679. doi:10.1038/mp.2017.16
78. Roberts RJ, Lohano KK, El-Mallakh RS. Antipsychotics as antidepressants. Asia Pac Psychiatry. 2016;8(3):179-188. doi:10.1111/appy.12186
79. Cipriani A, Barbui C, Salanti G, et al. Comparative efficacy and acceptability of antimanic drugs in acute mania: a multiple-treatments meta-analysis. Lancet. 2011;378(9799):1306-1315. doi:10.1016/S0140-6736(11)60873-8
80. Hayes JF, Marston L, Walters K, et al. Lithium vs. valproate vs. olanzapine vs. quetiapine as maintenance monotherapy for bipolar disorder: a population-based UK cohort study using electronic health records. World Psychiatry. 2016;15(1):53-58. doi:10.1002/wps.20298
81. Geddes JR, Gardiner A, Rendell J, et al. Comparative evaluation of quetiapine plus lamotrigine combination versus quetiapine monotherapy (and folic acid versus placebo) in bipolar depression (CEQUEL): a 2 × 2 factorial randomised trial. Lancet Psychiatry. 2016;3(1):31239. doi:10.1016/S2215-0366(15)00450-2
82. Goodwin GM, Haddad PM, Ferrier IN, et al. Evidence-based guidelines for treating bipolar disorder: revised third edition recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2016;30(6):495-553. doi:10.1177/0269881116636545
Bipolar disorder (BD) is a recurrent, life-long psychiatric illness affecting nearly 2% of the world population1,2 that is characterized by episodes of mania and depression interspersed among periods of relative mood stability.3 The illness causes an enormous health burden, which makes understanding its pathophysiology and treatment patterns a substantial priority.4 In the 1950s, lithium was found to be effective for treating acute manic episodes and preventing relapse in BD.5 Since then, valproate and carbamazepine also have been FDA-approved for treating mania.6,7 Antipsychotics have also shown evidence of efficacy in BD treatment,8,9 particularly for use in acute settings for more rapid effect or for a limited duration,10 which has led some to refer to them as “mood stabilizers.”11
In this article, we describe changes in trends of prescribing medications to treat BD, the role of ion dysregulation in the disorder, and how a better understanding of this dysregulation might impact the choice of treatment.
Changes in pharmacotherapy for bipolar disorder
From 1997 through 2016, the use of lithium for BD decreased from >30% of patients to 17.6% (with a nadir of 13.9% from 2009 to 2012).12 Over the same period, the use of nonlithium mood stabilizers decreased from 30.4% to approximately 4.8%, while second-generation antipsychotic (SGAs) use increased from 12.4% to 50.4%.12 Distressingly, antidepressant use increased from approximately 47% to 56.8%, and antidepressant use without concomitant mood stabilizers increased from 38% to 40.8%, although the rate of antidepressants without either a mood stabilizer or an antipsychotic remained relatively stable (14.9% to 16.8%).12 In randomized trials, when added to mood stabilizers, antidepressants have consistently failed to separate from placebo,13-15 but they can destabilize the illness, resulting in increases in mania, depression, and subsyndromal mixed symptoms.16-18
It is easy to understand clinicians’ attempts to address their patients’ distress due to depressive symptoms that do not resolve with mood stabilizers.19,20 Similarly, the increased use of antipsychotics is driven by evidence that antipsychotics are effective for treating bipolar depression and preventing the recurrence of manic and (for some antipsychotics) depressive episodes.21,22 However, long-term antipsychotic use causes brain volume change in patients with schizophrenia23 or major depressive disorder24 and in nonhuman primates25,26; metabolic abnormalities27-31; and cardiovascular adverse effects.32 Antipsychotics are believed to be associated with withdrawal psychosis.33,34 In the head-to-head Clinical Health Outcomes Initiative in Comparative Effectiveness for Bipolar Disorder (Bipolar CHOICE) study, quetiapine was as effective as lithium but associated with more adverse effects.35 Importantly, the estimated disability-adjusted life years of patients with BD increased by 54.4% from 6.02 million in 1990 to 9.29 million in 2017, which is greater than the increase in the incidence of BD (47.74%) over the same time.36 This means that despite the dramatic increase in treatment options for people with BD, functional outcomes have declined.
One major difference between antipsychotics and mood stabilizers is that antipsychotics do not alter the underlying abnormal pathology of BD.37 An ideal pharmacologic intervention is one that corrects a known pathophysiologic anomaly of the condition being treated. There are no demonstrated abnormalities in the dopamine or serotonin systems in individuals with BD, but long-term use of antipsychotics may create dopaminergic alterations.33 One of the most reproducible biomarkers associated with manic and bipolar depressed mood states is increased intracellular sodium38,39 and reduced ability to correct a sodium challenge.40-42 By normalizing intracellular sodium levels, lithium and the mood-stabilizing anticonvulsants uniquely and specifically counter known physiologic abnormalities in patients with BD.37,43
The role of ion dysregulation
The pathophysiology of BD remains elusive. A multitude of lines of evidence link BD to abnormal neuroimaging findings,22,44,45 oxidative stress,46 inflammation,47 and mitochondrial disease,48 but there is still no unifying understanding of these findings. Ion dysregulation appears to be central to understanding and treating BD.38,39
Despite extensive genetic studies, no genes have been identified that mediate >5% of the risk for BD. Nonetheless, 74% of all genes identified as mediating risk for BD code for proteins essential for the regulation of ion transport and membrane potential.49 The 2 genes that contribute the greatest risk are CACNA1C and ANK3, which code for a calcium channel and a cytoskeletal protein, respectively.50ANK3 codes for ankyrin G, which plays a role in proper coupling of the voltage-gated sodium channels to the cytoskeleton.51 An additional risk gene, TRANK1, contains multiple ankyrin-like repeat domains, which suggests some shared functions with ANK3.52 More importantly, the most reproducible pathophysiologic findings in BD are dysregulation of sodium, potassium, hydrogen, and calcium transport, with consequent alteration of depolarization potential, neuronal excitability, and calcium-mediated processes.38,39,53-56 For example, increased sodium and calcium within cells have been observed in both mania and bipolar depression, and these levels normalize during euthymia. All medications that are effective for treating BD may reduce intracellular sodium or calcium; traditional mood stabilizers do so directly by inhibiting voltage-sensitive sodium channels in an activity-dependent manner or displacing intracellular sodium,43,57 whereas antipsychotics do so indirectly by increasing sodium pump activity through inhibition of second messengers of the dopamine D2 family of receptors.37
Continue to: The extent of ion dysregulation...
The extent of ion dysregulation is directly associated with the expressed mood state of the illness. A small reduction in the activity of the sodium pump results in a small increase in intracellular sodium (approximately 10 mM).39,58 This led to the hypothesis that increased intracellular sodium causes the transmembrane potential to increase closer to membrane depolarization threshold, which increases excitability of affected neurons.38,39,58 Neurons are likely to fire and propagate signals more easily, which may manifest as symptoms of mania, such as increased energy, activity, lability, excitability, irritability, tangentiality, and looseness of associations. As the process of increased intracellular sodium progresses, a minority of neurons are expected to have their transmembrane potentials depolarize sufficiently for the resting membrane potential to go beyond threshold potential.59 Such neurons are in a state of constant depolarization (also known as depolarization block), which disrupts neuronal circuits. The difficulty in progression of these signals results in the classic bipolar depression symptoms of low energy, reduced activity, and slowing of all brain activity that is seen as psychomotor slowing.38
Implications for treatment
Medications for treating bipolar illness include lithium, anticonvulsants, benzodiazepines, first-generation antipsychotics, and SGAs.37,43
Mood stabilizers (lithium and certain anticonvulsants) correct the previously mentioned sodium abnormality by reducing sodium entry into the cell in an activity-dependent manner.43 As the only agents that directly address a known pathophysiologic abnormality, they are foundational in the treatment of BD.60 Lithium effectively treats acute mania and prevents relapse.61 It preferentially targets the active neurons, entering through both voltage-responsive and neurotransmitter-coupled channels.43,62 This results in an increase of intracellular lithium concentrations to as much as 8 times that of the extracellular concentration.63 These ions displace intracellular sodium ions in a 1:1 ratio, which results in a reduced intracellular sodium concentration that reduces the excitability of neurons.43,57,62
Substantial evidence supports the use of valproic acid for initial and maintenance treatment of BD.64 It inhibits the voltage-sensitive sodium channel when the channel is open, which results in an activity-dependent action that selectively impacts rapidly firing neurons.43 The voltage-gated sodium channels exist nearly exclusively on the axon, beyond the hillock65; as such, valproic acid will only inhibit neurons that fire, whereas lithium accumulates throughout the neuron and will affect depolarization in the neuronal soma as well as the firing in the axon.43 Additionally, valproic acid has been observed to enhance gamma-aminobutyric acid (GABA) levels and transmission.43,66,67 A meta-analysis that included 6 randomized controlled trials illustrated that, acutely, valproate was not different from lithium’s overall efficacy (RR 1.02; 95% CI, 0.87 to 1.20), but was associated with reduced dropout rates compared with placebo or lithium (RR 0.82; 95% CI, 0.71 to 0.95 and RR 0.87; 95% CI, 0.77 to 0.98, respectively).64
Lamotrigine is an anticonvulsant used for initial and maintenance treatment of BD, with greater efficacy for depressive episodes68; it also has notable effect for treating bipolar depression, although it is not FDA-approved for this indication.69 Lamotrigine inhibits sodium influx by binding to open voltage-gated sodium channels70 but also appears to reduce N-methyl-D-aspartate–mediated sodium entry,71 thereby acting both prehillock and posthillock.
Continue to: Carbamazepine is an anticonvulsant...
Carbamazepine is an anticonvulsant FDA-approved for treating BD.7 Like valproate, it acts by inhibiting voltage-gated sodium channels in an activity-dependent manner,72 which means it preferentially inhibits the most active neurons and those with higher intracellular sodium.43
Benzodiazepines, which have shown to be effective for treating acute mania,73 potentiate synaptic GABA receptors accruing an elevation in intracellular chloride influx.74 Despite acute efficacy, benzodiazepine use is limited because these agents are associated with worsening long-term, substance use–related outcomes.75,76
Antipsychotics are effective for treating mood disorders,60,76 and their use has been rising dramatically.12 The antimanic effect of all antipsychotics is believed to be mediated through dopamine D2 blockade, since use of a dose sufficient to block D2 receptors is required, and haloperidol, which acts exclusively on the D2 receptor, is equal to SGAs in its antimanic effect.77 Blockade of the D2 receptor will increase the activity of the sodium pump (sodium and potassium-activated adenosine triphosphatase) thus reducing intracellular sodium and calcium concentrations.37 When antipsychotics are used as antidepressants, they are generally used at doses lower than those used to treat mania.78
Antipsychotics are effective for treating BD, and may work more quickly than other agents for treating acute mania.79 However, maintenance or prevention trials tend to favor mood stabilizers.35,60,80 Several add-on studies have found the combination of a mood stabilizer plus an antipsychotic is superior to a mood stabilizer alone or an antipsychotic alone.81
An argument for mood stabilizers
Evidence suggests mood stabilizers and other approaches, such as antipsychotics, are almost equivalent for treating acute mania, with a small clinical advantage of mood stabilizers for preventing relapse. In general, current treatment guidelines do not distinguish mood stabilizers from antipsychotics as the first-line treatment.82 Over the past 20 years, antipsychotic use has increased while mood stabilizer use has decreased, so that presently a patient with BD is more likely to be prescribed an antipsychotic than a mood stabilizer.12 Over the same time, dysfunction among patients with BD has increased.33 Antipsychotics are appealing because they appear to be equally effective and generally well tolerated. But these agents cause problems that are difficult to see in routine visits, such as metabolic27-31 and cardiovascular adverse effects29 as well as reductions in brain volume.23-26 Mechanistic research suggests that mood stabilizers directly correct known pathophysiologic anomalies with additional protective effects, whereas antipsychotics appear to create new abnormalities and contribute to medical problems. Clinicians need to look beyond the similarities in acute efficacy and make a more broadly supported, evidence-based choice for managing BD, which clearly places mood stabilizers as the first-line agent and antipsychotics as reasonable alternatives. At a minimum, mood stabilizers should be viewed as the foundation to which antipsychotics can be added.
Bottom Line
Traditional mood stabilizers—lithium and some anticonvulsants—are the only agents that directly address physiologic abnormalities associated with both mania and bipolar depression, including mood state–associated elevations of intracellular sodium. Because of their specificity, these agents maximize mood stabilization and minimize adverse effects.
Related Resources
- Karas A, Stummer L, Freedberg A. Psychiatric and nonpsychiatric indications for mood stabilizers and select antiepileptics. Current Psychiatry. 2022;21(4):34-38. doi:10.12788/cp.0230
- Koch J. Mood stabilizers: balancing tolerability, serum levels, and dosage. Current Psychiatry. 2021;20(7):37-40. doi:10.12788/cp.0147
Drug Brand Names
Carbamazepine • Tegretol
Haloperidol • Haldol
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Quetiapine • Seroquel
Valproate • Depakote, Depakene
Bipolar disorder (BD) is a recurrent, life-long psychiatric illness affecting nearly 2% of the world population1,2 that is characterized by episodes of mania and depression interspersed among periods of relative mood stability.3 The illness causes an enormous health burden, which makes understanding its pathophysiology and treatment patterns a substantial priority.4 In the 1950s, lithium was found to be effective for treating acute manic episodes and preventing relapse in BD.5 Since then, valproate and carbamazepine also have been FDA-approved for treating mania.6,7 Antipsychotics have also shown evidence of efficacy in BD treatment,8,9 particularly for use in acute settings for more rapid effect or for a limited duration,10 which has led some to refer to them as “mood stabilizers.”11
In this article, we describe changes in trends of prescribing medications to treat BD, the role of ion dysregulation in the disorder, and how a better understanding of this dysregulation might impact the choice of treatment.
Changes in pharmacotherapy for bipolar disorder
From 1997 through 2016, the use of lithium for BD decreased from >30% of patients to 17.6% (with a nadir of 13.9% from 2009 to 2012).12 Over the same period, the use of nonlithium mood stabilizers decreased from 30.4% to approximately 4.8%, while second-generation antipsychotic (SGAs) use increased from 12.4% to 50.4%.12 Distressingly, antidepressant use increased from approximately 47% to 56.8%, and antidepressant use without concomitant mood stabilizers increased from 38% to 40.8%, although the rate of antidepressants without either a mood stabilizer or an antipsychotic remained relatively stable (14.9% to 16.8%).12 In randomized trials, when added to mood stabilizers, antidepressants have consistently failed to separate from placebo,13-15 but they can destabilize the illness, resulting in increases in mania, depression, and subsyndromal mixed symptoms.16-18
It is easy to understand clinicians’ attempts to address their patients’ distress due to depressive symptoms that do not resolve with mood stabilizers.19,20 Similarly, the increased use of antipsychotics is driven by evidence that antipsychotics are effective for treating bipolar depression and preventing the recurrence of manic and (for some antipsychotics) depressive episodes.21,22 However, long-term antipsychotic use causes brain volume change in patients with schizophrenia23 or major depressive disorder24 and in nonhuman primates25,26; metabolic abnormalities27-31; and cardiovascular adverse effects.32 Antipsychotics are believed to be associated with withdrawal psychosis.33,34 In the head-to-head Clinical Health Outcomes Initiative in Comparative Effectiveness for Bipolar Disorder (Bipolar CHOICE) study, quetiapine was as effective as lithium but associated with more adverse effects.35 Importantly, the estimated disability-adjusted life years of patients with BD increased by 54.4% from 6.02 million in 1990 to 9.29 million in 2017, which is greater than the increase in the incidence of BD (47.74%) over the same time.36 This means that despite the dramatic increase in treatment options for people with BD, functional outcomes have declined.
One major difference between antipsychotics and mood stabilizers is that antipsychotics do not alter the underlying abnormal pathology of BD.37 An ideal pharmacologic intervention is one that corrects a known pathophysiologic anomaly of the condition being treated. There are no demonstrated abnormalities in the dopamine or serotonin systems in individuals with BD, but long-term use of antipsychotics may create dopaminergic alterations.33 One of the most reproducible biomarkers associated with manic and bipolar depressed mood states is increased intracellular sodium38,39 and reduced ability to correct a sodium challenge.40-42 By normalizing intracellular sodium levels, lithium and the mood-stabilizing anticonvulsants uniquely and specifically counter known physiologic abnormalities in patients with BD.37,43
The role of ion dysregulation
The pathophysiology of BD remains elusive. A multitude of lines of evidence link BD to abnormal neuroimaging findings,22,44,45 oxidative stress,46 inflammation,47 and mitochondrial disease,48 but there is still no unifying understanding of these findings. Ion dysregulation appears to be central to understanding and treating BD.38,39
Despite extensive genetic studies, no genes have been identified that mediate >5% of the risk for BD. Nonetheless, 74% of all genes identified as mediating risk for BD code for proteins essential for the regulation of ion transport and membrane potential.49 The 2 genes that contribute the greatest risk are CACNA1C and ANK3, which code for a calcium channel and a cytoskeletal protein, respectively.50ANK3 codes for ankyrin G, which plays a role in proper coupling of the voltage-gated sodium channels to the cytoskeleton.51 An additional risk gene, TRANK1, contains multiple ankyrin-like repeat domains, which suggests some shared functions with ANK3.52 More importantly, the most reproducible pathophysiologic findings in BD are dysregulation of sodium, potassium, hydrogen, and calcium transport, with consequent alteration of depolarization potential, neuronal excitability, and calcium-mediated processes.38,39,53-56 For example, increased sodium and calcium within cells have been observed in both mania and bipolar depression, and these levels normalize during euthymia. All medications that are effective for treating BD may reduce intracellular sodium or calcium; traditional mood stabilizers do so directly by inhibiting voltage-sensitive sodium channels in an activity-dependent manner or displacing intracellular sodium,43,57 whereas antipsychotics do so indirectly by increasing sodium pump activity through inhibition of second messengers of the dopamine D2 family of receptors.37
Continue to: The extent of ion dysregulation...
The extent of ion dysregulation is directly associated with the expressed mood state of the illness. A small reduction in the activity of the sodium pump results in a small increase in intracellular sodium (approximately 10 mM).39,58 This led to the hypothesis that increased intracellular sodium causes the transmembrane potential to increase closer to membrane depolarization threshold, which increases excitability of affected neurons.38,39,58 Neurons are likely to fire and propagate signals more easily, which may manifest as symptoms of mania, such as increased energy, activity, lability, excitability, irritability, tangentiality, and looseness of associations. As the process of increased intracellular sodium progresses, a minority of neurons are expected to have their transmembrane potentials depolarize sufficiently for the resting membrane potential to go beyond threshold potential.59 Such neurons are in a state of constant depolarization (also known as depolarization block), which disrupts neuronal circuits. The difficulty in progression of these signals results in the classic bipolar depression symptoms of low energy, reduced activity, and slowing of all brain activity that is seen as psychomotor slowing.38
Implications for treatment
Medications for treating bipolar illness include lithium, anticonvulsants, benzodiazepines, first-generation antipsychotics, and SGAs.37,43
Mood stabilizers (lithium and certain anticonvulsants) correct the previously mentioned sodium abnormality by reducing sodium entry into the cell in an activity-dependent manner.43 As the only agents that directly address a known pathophysiologic abnormality, they are foundational in the treatment of BD.60 Lithium effectively treats acute mania and prevents relapse.61 It preferentially targets the active neurons, entering through both voltage-responsive and neurotransmitter-coupled channels.43,62 This results in an increase of intracellular lithium concentrations to as much as 8 times that of the extracellular concentration.63 These ions displace intracellular sodium ions in a 1:1 ratio, which results in a reduced intracellular sodium concentration that reduces the excitability of neurons.43,57,62
Substantial evidence supports the use of valproic acid for initial and maintenance treatment of BD.64 It inhibits the voltage-sensitive sodium channel when the channel is open, which results in an activity-dependent action that selectively impacts rapidly firing neurons.43 The voltage-gated sodium channels exist nearly exclusively on the axon, beyond the hillock65; as such, valproic acid will only inhibit neurons that fire, whereas lithium accumulates throughout the neuron and will affect depolarization in the neuronal soma as well as the firing in the axon.43 Additionally, valproic acid has been observed to enhance gamma-aminobutyric acid (GABA) levels and transmission.43,66,67 A meta-analysis that included 6 randomized controlled trials illustrated that, acutely, valproate was not different from lithium’s overall efficacy (RR 1.02; 95% CI, 0.87 to 1.20), but was associated with reduced dropout rates compared with placebo or lithium (RR 0.82; 95% CI, 0.71 to 0.95 and RR 0.87; 95% CI, 0.77 to 0.98, respectively).64
Lamotrigine is an anticonvulsant used for initial and maintenance treatment of BD, with greater efficacy for depressive episodes68; it also has notable effect for treating bipolar depression, although it is not FDA-approved for this indication.69 Lamotrigine inhibits sodium influx by binding to open voltage-gated sodium channels70 but also appears to reduce N-methyl-D-aspartate–mediated sodium entry,71 thereby acting both prehillock and posthillock.
Continue to: Carbamazepine is an anticonvulsant...
Carbamazepine is an anticonvulsant FDA-approved for treating BD.7 Like valproate, it acts by inhibiting voltage-gated sodium channels in an activity-dependent manner,72 which means it preferentially inhibits the most active neurons and those with higher intracellular sodium.43
Benzodiazepines, which have shown to be effective for treating acute mania,73 potentiate synaptic GABA receptors accruing an elevation in intracellular chloride influx.74 Despite acute efficacy, benzodiazepine use is limited because these agents are associated with worsening long-term, substance use–related outcomes.75,76
Antipsychotics are effective for treating mood disorders,60,76 and their use has been rising dramatically.12 The antimanic effect of all antipsychotics is believed to be mediated through dopamine D2 blockade, since use of a dose sufficient to block D2 receptors is required, and haloperidol, which acts exclusively on the D2 receptor, is equal to SGAs in its antimanic effect.77 Blockade of the D2 receptor will increase the activity of the sodium pump (sodium and potassium-activated adenosine triphosphatase) thus reducing intracellular sodium and calcium concentrations.37 When antipsychotics are used as antidepressants, they are generally used at doses lower than those used to treat mania.78
Antipsychotics are effective for treating BD, and may work more quickly than other agents for treating acute mania.79 However, maintenance or prevention trials tend to favor mood stabilizers.35,60,80 Several add-on studies have found the combination of a mood stabilizer plus an antipsychotic is superior to a mood stabilizer alone or an antipsychotic alone.81
An argument for mood stabilizers
Evidence suggests mood stabilizers and other approaches, such as antipsychotics, are almost equivalent for treating acute mania, with a small clinical advantage of mood stabilizers for preventing relapse. In general, current treatment guidelines do not distinguish mood stabilizers from antipsychotics as the first-line treatment.82 Over the past 20 years, antipsychotic use has increased while mood stabilizer use has decreased, so that presently a patient with BD is more likely to be prescribed an antipsychotic than a mood stabilizer.12 Over the same time, dysfunction among patients with BD has increased.33 Antipsychotics are appealing because they appear to be equally effective and generally well tolerated. But these agents cause problems that are difficult to see in routine visits, such as metabolic27-31 and cardiovascular adverse effects29 as well as reductions in brain volume.23-26 Mechanistic research suggests that mood stabilizers directly correct known pathophysiologic anomalies with additional protective effects, whereas antipsychotics appear to create new abnormalities and contribute to medical problems. Clinicians need to look beyond the similarities in acute efficacy and make a more broadly supported, evidence-based choice for managing BD, which clearly places mood stabilizers as the first-line agent and antipsychotics as reasonable alternatives. At a minimum, mood stabilizers should be viewed as the foundation to which antipsychotics can be added.
Bottom Line
Traditional mood stabilizers—lithium and some anticonvulsants—are the only agents that directly address physiologic abnormalities associated with both mania and bipolar depression, including mood state–associated elevations of intracellular sodium. Because of their specificity, these agents maximize mood stabilization and minimize adverse effects.
Related Resources
- Karas A, Stummer L, Freedberg A. Psychiatric and nonpsychiatric indications for mood stabilizers and select antiepileptics. Current Psychiatry. 2022;21(4):34-38. doi:10.12788/cp.0230
- Koch J. Mood stabilizers: balancing tolerability, serum levels, and dosage. Current Psychiatry. 2021;20(7):37-40. doi:10.12788/cp.0147
Drug Brand Names
Carbamazepine • Tegretol
Haloperidol • Haldol
Lamotrigine • Lamictal
Lithium • Eskalith, Lithobid
Quetiapine • Seroquel
Valproate • Depakote, Depakene
1. Whiteford HA, Degenhardt L, Rehm J, et al. Global burden of disease attributable to mental and substance use disorders: findings from the Global Burden of Disease Study 2010. Lancet. 2013;382(9904):1575-1586. doi:10.1016/S0140-6736(13)61611-6
2. Merikangas KR, Jin R, He JP, et al. Prevalence and correlates of bipolar spectrum disorder in the world mental health survey initiative. Arch Gen Psychiatry. 2011;68(3):241-251. doi:10.1001/archgenpsychiatry.2011.12
3. Müller JK, Leweke FM. Bipolar disorder: clinical overview. Article in English, German. Med Monatsschr Pharm. 2016;39(9):363-369.
4. Smith DJ, Whitham EA, Ghaemi SN. Bipolar disorder. Handb Clin Neurol. 2012;106:251-263. doi:10.1016/B978-0-444-52002-9.00015-2
5. Goodwin FK, Ghaemi SN. The impact of the discovery of lithium on psychiatric thought and practice in the USA and Europe. Aust N Z J Psychiatry. 1999;33 Suppl:S54-S64. doi:10.1111/j.1440-1614.1999.00669.x
6. Pope HG, McElroy SL, Keck PE, et al. Valproate in the treatment of acute mania. A placebo-controlled study. Arch Gen Psychiatry. 1991;48(1):62-68. doi:10.1001/archpsyc.1991.01810250064008
7. Weisler RH, Keck PE Jr, Swann AC, et al. Extended-release carbamazepine capsules as monotherapy for acute mania in bipolar disorder: a multicenter, randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2005;66(3):323-330. doi:10.4088/jcp.v66n0308
8. Tarr GP, Glue P, Herbison P. Comparative efficacy and acceptability of mood stabilizer and second generation antipsychotic monotherapy for acute mania--a systematic review and meta-analysis. J Affect Disord. 2011;134(1-3):14-19. doi:10.1016/j.jad.2010.11.009
9. Pahwa M, Sleem A, Elsayed OH, et al. New antipsychotic medications in the last decade. Curr Psychiatry Rep. 2021;23(12):87.
10. Correll CU, Sheridan EM, DelBello MP. Antipsychotic and mood stabilizer efficacy and tolerability in pediatric and adult patients with bipolar I mania: a comparative analysis of acute, randomized, placebo-controlled trials. Bipolar Disord. 2010;12(2):116-141. doi:10.1111/j.1399-5618.2010.00798.x
11. Rybakowski JK. Two generations of mood stabilizers. Int J Neuropsychopharmacol. 2007;10:709-711. doi:10.1017/s146114570700795x
12. Rhee TG, Olfson M, Nierenberg AA, et al. 20-year trends in the pharmacologic treatment of bipolar disorder by psychiatrists in outpatient care settings. Am J Psychiatry. 2020;177(8):706-715. doi:10.1176/appi.ajp.2020.19091000
13. El-Mallakh RS. Adjunctive antidepressant treatment for bipolar depression. N Engl J Med. 2007;357(6):615; author reply 615-616.
14. Sachs GS, Nierenberg AA, Calabrese JR, et al. Effectiveness of adjunctive antidepressant treatment for bipolar depression. N Engl J Med. 2007;356(17):1711-1722. doi:10.1056/NEJMoa064135
15. Ghaemi SN, Whitham EA, Vohringer PA, et al. Citalopram for acute and preventive efficacy in bipolar depression (CAPE-BD): a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2021;82(1):19m13136. doi:10.4088/JCP.19m13136
16. El-Mallakh RS, Ghaemi SN, Sagduyu K, et al. Antidepressant-associated chronic irritable dysphoria (ACID) in STEP-BD patients. J Affect Disord. 2008;111(2-3):372-377. doi:10.1016/j.jad.2008.03.025
17. Ghaemi SN, Ostacher MM, El-Mallakh RS, et al. Antidepressant discontinuation in bipolar depression: a systematic treatment enhancement program for bipolar disorder (STEP-BD) randomized clinical trial of long-term effectiveness and safety. J Clin Psychiatry. 2010;71(4):372-380.
18. Strejilevich SA, Martino DJ, Marengo E, et al. Long-term worsening of bipolar disorder related with frequency of antidepressant exposure. Ann Clin Psychiatry. 2011;23(3):186-192.
19. Pacchiarotti I, Bond DJ, Baldessarini RJ, et al. The International Society of Bipolar Disorders (ISBD) task force report on antidepressant use in bipolar disorders. Am J Psychiatry. 2013;170(11):1249-1262. doi:10.1176/appi.ajp.2013.13020185
20. McIntyre RS, Calabrese JR. Bipolar depression: the clinical characteristics and unmet needs of a complex disorder. Curr Med Res Opin. 2019;35(11):1993-2005.
21. Fornaro M, Stubbs B, De Berardis D, et al. Atypical antipsychotics in the treatment of acute bipolar depression with mixed features: a systematic review and exploratory meta-analysis of placebo-controlled clinical trials. Int J Mol Sci. 2016;17(2):241. doi:10.3390/ijms17020241
22. Lindström L, Lindström E, Nilsson M, et al. Maintenance therapy with second generation antipsychotics for bipolar disorder – a systematic review and meta-analysis. J Affect Disord. 2017;213:138-150. doi:10.1016/j.jad.2017.02.012
23. Ho BC, Andreasen NC, Ziebell S, et al. Long-term antipsychotic treatment and brain volumes: a longitudinal study of first-episode schizophrenia. Arch Gen Psychiatry. 2011;68(2):128-137. doi:010.1001/archgenpsychiatry.2010.199
24. Voineskos AN, Mulsant BH, Dickie EW, et al. Effects of antipsychotic medication on brain structure in patients with major depressive disorder and psychotic features: neuroimaging findings in the context of a randomized placebo-controlled clinical trial. JAMA Psychiatry. 2020;77(7):674-683. doi:10.1001/jamapsychiatry.2020.0036
25. Konopaske GT, Bolo NR, Basu AC, et al. Time-dependent effects of haloperidol on glutamine and GABA homeostasis and astrocyte activity in the rat brain. Psychopharmacology (Berl). 2013;230(1):57-67. doi:10.1007/s00213-013-3136-3
26. Dorph-Petersen KA, Pierri JN, Perel JM, et al. The influence of chronic exposure to antipsychotic medications on brain size before and after tissue fixation: a comparison of haloperidol and olanzapine in macaque monkeys. Neuropsychopharmacology. 2005;30(9):1649-1661. doi:10.1038/sj.npp.1300710
27. McIntyre RS, Mancini DA, Basile VS, et al. Antipsychotic-induced weight gain: bipolar disorder and leptin. J Clin Psychopharmacol. 2003;23(4):323-327. doi:10.1097/01.jcp.0000085403.08426.f4
28. McIntyre RS, Konarski JZ, Wilkins K, et al. Obesity in bipolar disorder and major depressive disorder: results from a national community health survey on mental health and well-being. Can J Psychiatry. 2006;51(5):274-280. doi:10.1177/070674370605100502
29. McIntyre RS, Cha DS, Kim RD, et al. A review of FDA-approved treatment options in bipolar depression. CNS Spectr. 2013;18(Suppl 1):4-20. doi:10.1017/S1092852913000746
30. Barton BB, Segger F, Fischer K, et al. Update on weight-gain caused by antipsychotics: a systematic review and meta-analysis. Expert Opin Drug Saf. 2020;19(3):295-314. doi:10.1080/14740338.2020.1713091
31. Doane MJ, Bessonova L, Friedler HS, et al. Weight gain and comorbidities associated with oral second-generation antipsychotics: analysis of real-world data for patients with schizophrenia or bipolar I disorder. BMC Psychiatry. 2022;22(1):114. doi:10.1186/s12888-022-03758-w
32. Buckley NA, Sanders P. Cardiovascular adverse effects of antipsychotic drugs. Drug Saf. 2000;23(3):215-228. doi:10.2165/00002018-200023030-00004
33. Ali Z, Roque A, El-Mallakh RS. A unifying theory for the pathoetiologic mechanism of tardive dyskinesia. Med Hypotheses. 2020;140:109682. doi:10.1016/j.mehy.2020.109682
34. Sleem A, El-Mallakh RS. Adaptive changes to antipsychotics: their consequences and how to avoid them. Curr Psychiatry. 2022;21(7):46-50,52. doi: 10.12788/cp.0262
35. Nierenberg AA, McElroy SL, Friedman ES, et al. Bipolar CHOICE (Clinical Health Outcomes Initiative in Comparative Effectiveness): a pragmatic 6-month trial of lithium versus quetiapine for bipolar disorder. J Clin Psychiatry. 2016;77(1):90-99. doi:10.4088/JCP.14m09349
36. He H, Hu C, Ren Z, et al. Trends in the incidence and DALYs of bipolar disorder at global, regional, and national levels: results from the global burden of disease study 2017. J Psychiatr Res. 2020;125:96-105. doi:10.1016/j.jpsychires.2020.03.015
37. Roberts RJ, Repass R, El-Mallakh RS. Effect of dopamine on intracellular sodium: a common pathway for pharmacological mechanism of action in bipolar illness. World J Biol Psychiatry. 2010;11(2 Pt 2):181-187. doi:10.1080/15622970902718774
38. El-Mallakh RS, Wyatt RJ. The Na, K-ATPase hypothesis for bipolar illness. Biol Psychiatry. 1995;37(4):235-244. doi:10.1016/0006-3223(94)00201-D
39. El-Mallakh RS, Yff T, Gao Y. Ion dysregulation in the pathogenesis of bipolar disorder. Ann Depress Anxiety. 2016;3(1):1076.
40. Li R, El-Mallakh RS. Differential response of bipolar and normal control lymphoblastoid cell sodium pump to ethacrynic acid. J Affect Disord. 2004;80(1):11-17. doi:10.1016/S0165-0327(03)00044-2
41. Woodruff DB, El-Mallakh RS, Thiruvengadam AP. Validation of a diagnostic screening blood test for bipolar disorder. Ann Clin Psychiatry. 2012;24(2):135-139.
42. Gao Y, Lohano K, Delamere NA, et al. Ethanol normalizes glutamate-induced elevation of intracellular sodium in olfactory neuroepithelial progenitors from subjects with bipolar illness but not nonbipolar controls: biologic evidence for the self-medication hypothesis. Bipolar Disord. 2019;21(2):179-181. doi:10.1111/bdi.12737
43. El-Mallakh RS, Huff MO. Mood stabilizers and ion regulation. Harv Rev Psychiatry. 2001;9(1):23-32. doi:10.1080/10673220127873
44. Phillips ML, Swartz HA. A critical appraisal of neuroimaging studies of bipolar disorder: toward a new conceptualization of underlying neural circuitry and a road map for future research. Am J Psychiatry. 2014;171(8):829-843. doi:10.1176/appi.ajp.2014.13081008
45. Hibar DP, Westlye LT, Doan NT, et al. Cortical abnormalities in bipolar disorder: an MRI analysis of 6503 individuals from the ENIGMA Bipolar Disorder Working Group. Mol Psychiatry. 2018;23(4):932-942. doi:10.1038/mp.2017.73
46. Brown NC, Andreazza AC, Young LT. An updated meta-analysis of oxidative stress markers in bipolar disorder. Psychiatry Res. 2014;218(1-2):61-68. doi:10.1016/j.psychres.2014.04.005
47. Benedetti F, Aggio V, Pratesi ML, et al. Neuroinflammation in bipolar depression. Front Psychiatry. 2020;11:71. doi:10.3389/fpsyt.2020.00071
48. Andreazza AC, Duong A, Young LT. Bipolar disorder as a mitochondrial disease. Biol Psychiatry. 2018;83(9):720-721. doi:10.1016/j.biopsych.2017.09.018
49. Askland KD. Toward a biaxial model of “bipolar” affective disorders: further exploration of genetic, molecular and cellular substrates. J Affect Disord. 2006;94(1-3):35-66. doi:10.1016/j.jad.2006.01.033
50. Ferreira MA, O’Donovan MC, Meng YA, et al; Wellcome Trust Case Control Consortium. Collaborative genome-wide association analysis supports a role for ANK3 and CACNA1C in bipolar disorder. Nat Genet 2008;40(9):1056-1058. doi:10.1038/ng.209
51. Salvi AM, Bays JL, Mackin SR, et al. Ankyrin G organizes membrane components to promote coupling of cell mechanics and glucose uptake. Nat Cell Biol. 2021;23(5):457-466. doi:10.1038/s41556-021-00677-y
52. Gargus JJ. Ion channel functional candidate genes in multigenic neuropsychiatric disease. Biol Psychiatry. 2006;60(2):177-185. doi:10.1016/j.biopsych.2005.12.008
53. Dubovsky SL, Murphy J, Thomas M, et al. Abnormal intracellular calcium ion concentration in platelets and lymphocytes of bipolar patients. Am J Psychiatry 1992;149(1):118-120. doi:10.1176/ajp.149.1.118
54. Blaustein MP. Physiological effects of endogenous ouabain: control of intracellular Ca2+ stores and cell responsiveness. Am J Physiol. 1993;264(6 Pt 1):C1367–C1387. doi:10.1152/ajpcell.1993.264.6.C1367
55. El-Mallakh RS, Li R, Worth CA, et al. Leukocyte transmembrane potential in bipolar illness. J Affect Disord. 1996;41(1):33-37. doi:10.1016/0165-0327(96)00063-8
56. El-Mallakh RS, Gao Y, You P. Role of endogenous ouabain in the etiology of bipolar disorder. Int J Bipolar Disord. 2021;9(1):6. doi:10.1186/s40345-020-00213-1
57. Huang X, Lei Z, El‐Mallakh RS. Lithium normalizes elevated intracellular sodium. Bipolar Disord. 2007;9(3):298-300. doi:10.1111/j.1399-5618.2007.00429.x
58. Shaw DM. Mineral metabolism, mania, and melancholia. Br Med J. 1966;2(5508):262-267. doi:10.1136/bmj.2.5508.262
59. Qian K, Yu N, Tucker KR, et al. Mathematical analysis of depolarization block mediated by slow inactivation of fast sodium channels in midbrain dopamine neurons. J Neurophysiol. 2014;112(11):2779-2790. doi:10.1152/jn.00578.2014
60. Sleem A, El-Mallakh RS. Advances in the psychopharmacotherapy of bipolar disorder type I. Exp Opin Pharmacother. 2021;22(10):1267-1290. doi:10.1080/14656566.2021.1893306
61. Malhi GS., Tanious M, Das P, et al. Potential mechanisms of action of lithium in bipolar disorder. CNS Drugs. 2013;27(2):135-153. doi:10.1007/s40263-013-0039-0
62. Armett CJ, Ritchie JM. On the permeability of mammalian non-myelinated fibers to sodium and to lithium ions. J Physiol. 1963;165(1):130-140. doi:10.1113/jphysiol.1963.sp007047
63. Kabakov AY, Karkanias NB, Lenox RH, et al. Synapse-specific accumulation of lithium in intracellular microdomains: a model for uncoupling coincidence detection in the brain. Synapse. 1998;28(4):271-279. doi:10.1002/(SICI)1098-2396(199804)28:4<271::AID-SYN2>3.0.CO;2-6
64. Cipriani A, Reid K, Young AH, et al. Valproic acid, valproate and divalproex in the maintenance treatment of bipolar disorder. Cochrane Database Syst Rev. 2013;2013(10):CD003196. doi:10.1002/14651858.CD003196.pub2
65. Lai HC, Jan LY. The distribution and targeting of neuronal voltage-gated ion channels. Nat Rev Neurosci. 2006;7(7):548-562. doi:10.1038/nrn1938
66. Löscher W, Schmidt D. Increase of human plasma GABA by sodium valproate. Epilepsia. 1980;21(6):611-615. doi:10.1111/j.1528-1157.1980.tb04314.x
67. Owens MJ, Nemeroff CB. Pharmacology of valproate. Psychopharmacol Bull. 2003;37(Suppl 2):17-24.
68. Calabrese JR, Vieta E, Shelton MD. Latest maintenance data on lamotrigine in bipolar disorder. Eur Neuropsychopharmacol. 2003;13(Suppl 2):S57-S66. doi:10.1016/s0924-977x(03)00079-8
69. Geddes JR, Calabrese JR, Goodwin GM. Lamotrigine for treatment of bipolar depression: independent meta-analysis and meta-regression of individual patient data from five randomised trials. Br J Psychiatry. 2009;194(1):4-9. doi:10.1192/bjp.bp.107.048504
70. Nakatani Y, Masuko H, Amano T. Effect of lamotrigine on Na(v)1.4 voltage-gated sodium channels. J Pharmacol Sci. 2013;123(2):203-206. doi:10.1254/jphs.13116sc
71. Ramadan E, Basselin M, Rao JS, et al. Lamotrigine blocks NMDA receptor-initiated arachidonic acid signalling in rat brain: implications for its efficacy in bipolar disorder. Int J Neuropsychopharmacol. 2012;15(7):931-943. doi:10.1017/S1461145711001003
72. Jo S, Bean BP. Sidedness of carbamazepine accessibility to voltage-gated sodium channels. Mol Pharmacol. 2014;85(2):381-387. doi:10.1124/mol.113.090472
73. Curtin F, Schulz P. Clonazepam and lorazepam in acute mania: a Bayesian meta-analysis. J Affect Disord 2004;78(3):201-208. doi:10.1016/S0165-0327(02)00317-8
74. Edwards R, Stephenson U, Flewett T. Clonazepam in acute mania: a double blind trial. Aust N Z J Psychiatry 1991;25(2):238-242. doi:10.3109/00048679109077740
75. Lin SC, Chen CC, Chen YH, et al. Benzodiazepine prescription among patients with severe mental illness and co-occurring alcohol abuse/dependence in Taiwan. Hum Psychopharmacol. 2011;26(3):201-207. doi:10.1002/hup.1193
76. Prisciandaro JJ, Brown DG, Brady KT, et al. Comorbid anxiety disorders and baseline medication regimens predict clinical outcomes in individuals with co-occurring bipolar disorder and alcohol dependence: results of a randomized controlled trial. Psychiatry Res. 2011;188(3):361-365. doi:10.1016/j.psychres.2011.04.030
77. Ashok AH, Marques TR, Jauhar S, et al. The dopamine hypothesis of bipolar affective disorder: the state of the art and implications for treatment. Mol Psychiatry. 2017;22(5):666-679. doi:10.1038/mp.2017.16
78. Roberts RJ, Lohano KK, El-Mallakh RS. Antipsychotics as antidepressants. Asia Pac Psychiatry. 2016;8(3):179-188. doi:10.1111/appy.12186
79. Cipriani A, Barbui C, Salanti G, et al. Comparative efficacy and acceptability of antimanic drugs in acute mania: a multiple-treatments meta-analysis. Lancet. 2011;378(9799):1306-1315. doi:10.1016/S0140-6736(11)60873-8
80. Hayes JF, Marston L, Walters K, et al. Lithium vs. valproate vs. olanzapine vs. quetiapine as maintenance monotherapy for bipolar disorder: a population-based UK cohort study using electronic health records. World Psychiatry. 2016;15(1):53-58. doi:10.1002/wps.20298
81. Geddes JR, Gardiner A, Rendell J, et al. Comparative evaluation of quetiapine plus lamotrigine combination versus quetiapine monotherapy (and folic acid versus placebo) in bipolar depression (CEQUEL): a 2 × 2 factorial randomised trial. Lancet Psychiatry. 2016;3(1):31239. doi:10.1016/S2215-0366(15)00450-2
82. Goodwin GM, Haddad PM, Ferrier IN, et al. Evidence-based guidelines for treating bipolar disorder: revised third edition recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2016;30(6):495-553. doi:10.1177/0269881116636545
1. Whiteford HA, Degenhardt L, Rehm J, et al. Global burden of disease attributable to mental and substance use disorders: findings from the Global Burden of Disease Study 2010. Lancet. 2013;382(9904):1575-1586. doi:10.1016/S0140-6736(13)61611-6
2. Merikangas KR, Jin R, He JP, et al. Prevalence and correlates of bipolar spectrum disorder in the world mental health survey initiative. Arch Gen Psychiatry. 2011;68(3):241-251. doi:10.1001/archgenpsychiatry.2011.12
3. Müller JK, Leweke FM. Bipolar disorder: clinical overview. Article in English, German. Med Monatsschr Pharm. 2016;39(9):363-369.
4. Smith DJ, Whitham EA, Ghaemi SN. Bipolar disorder. Handb Clin Neurol. 2012;106:251-263. doi:10.1016/B978-0-444-52002-9.00015-2
5. Goodwin FK, Ghaemi SN. The impact of the discovery of lithium on psychiatric thought and practice in the USA and Europe. Aust N Z J Psychiatry. 1999;33 Suppl:S54-S64. doi:10.1111/j.1440-1614.1999.00669.x
6. Pope HG, McElroy SL, Keck PE, et al. Valproate in the treatment of acute mania. A placebo-controlled study. Arch Gen Psychiatry. 1991;48(1):62-68. doi:10.1001/archpsyc.1991.01810250064008
7. Weisler RH, Keck PE Jr, Swann AC, et al. Extended-release carbamazepine capsules as monotherapy for acute mania in bipolar disorder: a multicenter, randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2005;66(3):323-330. doi:10.4088/jcp.v66n0308
8. Tarr GP, Glue P, Herbison P. Comparative efficacy and acceptability of mood stabilizer and second generation antipsychotic monotherapy for acute mania--a systematic review and meta-analysis. J Affect Disord. 2011;134(1-3):14-19. doi:10.1016/j.jad.2010.11.009
9. Pahwa M, Sleem A, Elsayed OH, et al. New antipsychotic medications in the last decade. Curr Psychiatry Rep. 2021;23(12):87.
10. Correll CU, Sheridan EM, DelBello MP. Antipsychotic and mood stabilizer efficacy and tolerability in pediatric and adult patients with bipolar I mania: a comparative analysis of acute, randomized, placebo-controlled trials. Bipolar Disord. 2010;12(2):116-141. doi:10.1111/j.1399-5618.2010.00798.x
11. Rybakowski JK. Two generations of mood stabilizers. Int J Neuropsychopharmacol. 2007;10:709-711. doi:10.1017/s146114570700795x
12. Rhee TG, Olfson M, Nierenberg AA, et al. 20-year trends in the pharmacologic treatment of bipolar disorder by psychiatrists in outpatient care settings. Am J Psychiatry. 2020;177(8):706-715. doi:10.1176/appi.ajp.2020.19091000
13. El-Mallakh RS. Adjunctive antidepressant treatment for bipolar depression. N Engl J Med. 2007;357(6):615; author reply 615-616.
14. Sachs GS, Nierenberg AA, Calabrese JR, et al. Effectiveness of adjunctive antidepressant treatment for bipolar depression. N Engl J Med. 2007;356(17):1711-1722. doi:10.1056/NEJMoa064135
15. Ghaemi SN, Whitham EA, Vohringer PA, et al. Citalopram for acute and preventive efficacy in bipolar depression (CAPE-BD): a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2021;82(1):19m13136. doi:10.4088/JCP.19m13136
16. El-Mallakh RS, Ghaemi SN, Sagduyu K, et al. Antidepressant-associated chronic irritable dysphoria (ACID) in STEP-BD patients. J Affect Disord. 2008;111(2-3):372-377. doi:10.1016/j.jad.2008.03.025
17. Ghaemi SN, Ostacher MM, El-Mallakh RS, et al. Antidepressant discontinuation in bipolar depression: a systematic treatment enhancement program for bipolar disorder (STEP-BD) randomized clinical trial of long-term effectiveness and safety. J Clin Psychiatry. 2010;71(4):372-380.
18. Strejilevich SA, Martino DJ, Marengo E, et al. Long-term worsening of bipolar disorder related with frequency of antidepressant exposure. Ann Clin Psychiatry. 2011;23(3):186-192.
19. Pacchiarotti I, Bond DJ, Baldessarini RJ, et al. The International Society of Bipolar Disorders (ISBD) task force report on antidepressant use in bipolar disorders. Am J Psychiatry. 2013;170(11):1249-1262. doi:10.1176/appi.ajp.2013.13020185
20. McIntyre RS, Calabrese JR. Bipolar depression: the clinical characteristics and unmet needs of a complex disorder. Curr Med Res Opin. 2019;35(11):1993-2005.
21. Fornaro M, Stubbs B, De Berardis D, et al. Atypical antipsychotics in the treatment of acute bipolar depression with mixed features: a systematic review and exploratory meta-analysis of placebo-controlled clinical trials. Int J Mol Sci. 2016;17(2):241. doi:10.3390/ijms17020241
22. Lindström L, Lindström E, Nilsson M, et al. Maintenance therapy with second generation antipsychotics for bipolar disorder – a systematic review and meta-analysis. J Affect Disord. 2017;213:138-150. doi:10.1016/j.jad.2017.02.012
23. Ho BC, Andreasen NC, Ziebell S, et al. Long-term antipsychotic treatment and brain volumes: a longitudinal study of first-episode schizophrenia. Arch Gen Psychiatry. 2011;68(2):128-137. doi:010.1001/archgenpsychiatry.2010.199
24. Voineskos AN, Mulsant BH, Dickie EW, et al. Effects of antipsychotic medication on brain structure in patients with major depressive disorder and psychotic features: neuroimaging findings in the context of a randomized placebo-controlled clinical trial. JAMA Psychiatry. 2020;77(7):674-683. doi:10.1001/jamapsychiatry.2020.0036
25. Konopaske GT, Bolo NR, Basu AC, et al. Time-dependent effects of haloperidol on glutamine and GABA homeostasis and astrocyte activity in the rat brain. Psychopharmacology (Berl). 2013;230(1):57-67. doi:10.1007/s00213-013-3136-3
26. Dorph-Petersen KA, Pierri JN, Perel JM, et al. The influence of chronic exposure to antipsychotic medications on brain size before and after tissue fixation: a comparison of haloperidol and olanzapine in macaque monkeys. Neuropsychopharmacology. 2005;30(9):1649-1661. doi:10.1038/sj.npp.1300710
27. McIntyre RS, Mancini DA, Basile VS, et al. Antipsychotic-induced weight gain: bipolar disorder and leptin. J Clin Psychopharmacol. 2003;23(4):323-327. doi:10.1097/01.jcp.0000085403.08426.f4
28. McIntyre RS, Konarski JZ, Wilkins K, et al. Obesity in bipolar disorder and major depressive disorder: results from a national community health survey on mental health and well-being. Can J Psychiatry. 2006;51(5):274-280. doi:10.1177/070674370605100502
29. McIntyre RS, Cha DS, Kim RD, et al. A review of FDA-approved treatment options in bipolar depression. CNS Spectr. 2013;18(Suppl 1):4-20. doi:10.1017/S1092852913000746
30. Barton BB, Segger F, Fischer K, et al. Update on weight-gain caused by antipsychotics: a systematic review and meta-analysis. Expert Opin Drug Saf. 2020;19(3):295-314. doi:10.1080/14740338.2020.1713091
31. Doane MJ, Bessonova L, Friedler HS, et al. Weight gain and comorbidities associated with oral second-generation antipsychotics: analysis of real-world data for patients with schizophrenia or bipolar I disorder. BMC Psychiatry. 2022;22(1):114. doi:10.1186/s12888-022-03758-w
32. Buckley NA, Sanders P. Cardiovascular adverse effects of antipsychotic drugs. Drug Saf. 2000;23(3):215-228. doi:10.2165/00002018-200023030-00004
33. Ali Z, Roque A, El-Mallakh RS. A unifying theory for the pathoetiologic mechanism of tardive dyskinesia. Med Hypotheses. 2020;140:109682. doi:10.1016/j.mehy.2020.109682
34. Sleem A, El-Mallakh RS. Adaptive changes to antipsychotics: their consequences and how to avoid them. Curr Psychiatry. 2022;21(7):46-50,52. doi: 10.12788/cp.0262
35. Nierenberg AA, McElroy SL, Friedman ES, et al. Bipolar CHOICE (Clinical Health Outcomes Initiative in Comparative Effectiveness): a pragmatic 6-month trial of lithium versus quetiapine for bipolar disorder. J Clin Psychiatry. 2016;77(1):90-99. doi:10.4088/JCP.14m09349
36. He H, Hu C, Ren Z, et al. Trends in the incidence and DALYs of bipolar disorder at global, regional, and national levels: results from the global burden of disease study 2017. J Psychiatr Res. 2020;125:96-105. doi:10.1016/j.jpsychires.2020.03.015
37. Roberts RJ, Repass R, El-Mallakh RS. Effect of dopamine on intracellular sodium: a common pathway for pharmacological mechanism of action in bipolar illness. World J Biol Psychiatry. 2010;11(2 Pt 2):181-187. doi:10.1080/15622970902718774
38. El-Mallakh RS, Wyatt RJ. The Na, K-ATPase hypothesis for bipolar illness. Biol Psychiatry. 1995;37(4):235-244. doi:10.1016/0006-3223(94)00201-D
39. El-Mallakh RS, Yff T, Gao Y. Ion dysregulation in the pathogenesis of bipolar disorder. Ann Depress Anxiety. 2016;3(1):1076.
40. Li R, El-Mallakh RS. Differential response of bipolar and normal control lymphoblastoid cell sodium pump to ethacrynic acid. J Affect Disord. 2004;80(1):11-17. doi:10.1016/S0165-0327(03)00044-2
41. Woodruff DB, El-Mallakh RS, Thiruvengadam AP. Validation of a diagnostic screening blood test for bipolar disorder. Ann Clin Psychiatry. 2012;24(2):135-139.
42. Gao Y, Lohano K, Delamere NA, et al. Ethanol normalizes glutamate-induced elevation of intracellular sodium in olfactory neuroepithelial progenitors from subjects with bipolar illness but not nonbipolar controls: biologic evidence for the self-medication hypothesis. Bipolar Disord. 2019;21(2):179-181. doi:10.1111/bdi.12737
43. El-Mallakh RS, Huff MO. Mood stabilizers and ion regulation. Harv Rev Psychiatry. 2001;9(1):23-32. doi:10.1080/10673220127873
44. Phillips ML, Swartz HA. A critical appraisal of neuroimaging studies of bipolar disorder: toward a new conceptualization of underlying neural circuitry and a road map for future research. Am J Psychiatry. 2014;171(8):829-843. doi:10.1176/appi.ajp.2014.13081008
45. Hibar DP, Westlye LT, Doan NT, et al. Cortical abnormalities in bipolar disorder: an MRI analysis of 6503 individuals from the ENIGMA Bipolar Disorder Working Group. Mol Psychiatry. 2018;23(4):932-942. doi:10.1038/mp.2017.73
46. Brown NC, Andreazza AC, Young LT. An updated meta-analysis of oxidative stress markers in bipolar disorder. Psychiatry Res. 2014;218(1-2):61-68. doi:10.1016/j.psychres.2014.04.005
47. Benedetti F, Aggio V, Pratesi ML, et al. Neuroinflammation in bipolar depression. Front Psychiatry. 2020;11:71. doi:10.3389/fpsyt.2020.00071
48. Andreazza AC, Duong A, Young LT. Bipolar disorder as a mitochondrial disease. Biol Psychiatry. 2018;83(9):720-721. doi:10.1016/j.biopsych.2017.09.018
49. Askland KD. Toward a biaxial model of “bipolar” affective disorders: further exploration of genetic, molecular and cellular substrates. J Affect Disord. 2006;94(1-3):35-66. doi:10.1016/j.jad.2006.01.033
50. Ferreira MA, O’Donovan MC, Meng YA, et al; Wellcome Trust Case Control Consortium. Collaborative genome-wide association analysis supports a role for ANK3 and CACNA1C in bipolar disorder. Nat Genet 2008;40(9):1056-1058. doi:10.1038/ng.209
51. Salvi AM, Bays JL, Mackin SR, et al. Ankyrin G organizes membrane components to promote coupling of cell mechanics and glucose uptake. Nat Cell Biol. 2021;23(5):457-466. doi:10.1038/s41556-021-00677-y
52. Gargus JJ. Ion channel functional candidate genes in multigenic neuropsychiatric disease. Biol Psychiatry. 2006;60(2):177-185. doi:10.1016/j.biopsych.2005.12.008
53. Dubovsky SL, Murphy J, Thomas M, et al. Abnormal intracellular calcium ion concentration in platelets and lymphocytes of bipolar patients. Am J Psychiatry 1992;149(1):118-120. doi:10.1176/ajp.149.1.118
54. Blaustein MP. Physiological effects of endogenous ouabain: control of intracellular Ca2+ stores and cell responsiveness. Am J Physiol. 1993;264(6 Pt 1):C1367–C1387. doi:10.1152/ajpcell.1993.264.6.C1367
55. El-Mallakh RS, Li R, Worth CA, et al. Leukocyte transmembrane potential in bipolar illness. J Affect Disord. 1996;41(1):33-37. doi:10.1016/0165-0327(96)00063-8
56. El-Mallakh RS, Gao Y, You P. Role of endogenous ouabain in the etiology of bipolar disorder. Int J Bipolar Disord. 2021;9(1):6. doi:10.1186/s40345-020-00213-1
57. Huang X, Lei Z, El‐Mallakh RS. Lithium normalizes elevated intracellular sodium. Bipolar Disord. 2007;9(3):298-300. doi:10.1111/j.1399-5618.2007.00429.x
58. Shaw DM. Mineral metabolism, mania, and melancholia. Br Med J. 1966;2(5508):262-267. doi:10.1136/bmj.2.5508.262
59. Qian K, Yu N, Tucker KR, et al. Mathematical analysis of depolarization block mediated by slow inactivation of fast sodium channels in midbrain dopamine neurons. J Neurophysiol. 2014;112(11):2779-2790. doi:10.1152/jn.00578.2014
60. Sleem A, El-Mallakh RS. Advances in the psychopharmacotherapy of bipolar disorder type I. Exp Opin Pharmacother. 2021;22(10):1267-1290. doi:10.1080/14656566.2021.1893306
61. Malhi GS., Tanious M, Das P, et al. Potential mechanisms of action of lithium in bipolar disorder. CNS Drugs. 2013;27(2):135-153. doi:10.1007/s40263-013-0039-0
62. Armett CJ, Ritchie JM. On the permeability of mammalian non-myelinated fibers to sodium and to lithium ions. J Physiol. 1963;165(1):130-140. doi:10.1113/jphysiol.1963.sp007047
63. Kabakov AY, Karkanias NB, Lenox RH, et al. Synapse-specific accumulation of lithium in intracellular microdomains: a model for uncoupling coincidence detection in the brain. Synapse. 1998;28(4):271-279. doi:10.1002/(SICI)1098-2396(199804)28:4<271::AID-SYN2>3.0.CO;2-6
64. Cipriani A, Reid K, Young AH, et al. Valproic acid, valproate and divalproex in the maintenance treatment of bipolar disorder. Cochrane Database Syst Rev. 2013;2013(10):CD003196. doi:10.1002/14651858.CD003196.pub2
65. Lai HC, Jan LY. The distribution and targeting of neuronal voltage-gated ion channels. Nat Rev Neurosci. 2006;7(7):548-562. doi:10.1038/nrn1938
66. Löscher W, Schmidt D. Increase of human plasma GABA by sodium valproate. Epilepsia. 1980;21(6):611-615. doi:10.1111/j.1528-1157.1980.tb04314.x
67. Owens MJ, Nemeroff CB. Pharmacology of valproate. Psychopharmacol Bull. 2003;37(Suppl 2):17-24.
68. Calabrese JR, Vieta E, Shelton MD. Latest maintenance data on lamotrigine in bipolar disorder. Eur Neuropsychopharmacol. 2003;13(Suppl 2):S57-S66. doi:10.1016/s0924-977x(03)00079-8
69. Geddes JR, Calabrese JR, Goodwin GM. Lamotrigine for treatment of bipolar depression: independent meta-analysis and meta-regression of individual patient data from five randomised trials. Br J Psychiatry. 2009;194(1):4-9. doi:10.1192/bjp.bp.107.048504
70. Nakatani Y, Masuko H, Amano T. Effect of lamotrigine on Na(v)1.4 voltage-gated sodium channels. J Pharmacol Sci. 2013;123(2):203-206. doi:10.1254/jphs.13116sc
71. Ramadan E, Basselin M, Rao JS, et al. Lamotrigine blocks NMDA receptor-initiated arachidonic acid signalling in rat brain: implications for its efficacy in bipolar disorder. Int J Neuropsychopharmacol. 2012;15(7):931-943. doi:10.1017/S1461145711001003
72. Jo S, Bean BP. Sidedness of carbamazepine accessibility to voltage-gated sodium channels. Mol Pharmacol. 2014;85(2):381-387. doi:10.1124/mol.113.090472
73. Curtin F, Schulz P. Clonazepam and lorazepam in acute mania: a Bayesian meta-analysis. J Affect Disord 2004;78(3):201-208. doi:10.1016/S0165-0327(02)00317-8
74. Edwards R, Stephenson U, Flewett T. Clonazepam in acute mania: a double blind trial. Aust N Z J Psychiatry 1991;25(2):238-242. doi:10.3109/00048679109077740
75. Lin SC, Chen CC, Chen YH, et al. Benzodiazepine prescription among patients with severe mental illness and co-occurring alcohol abuse/dependence in Taiwan. Hum Psychopharmacol. 2011;26(3):201-207. doi:10.1002/hup.1193
76. Prisciandaro JJ, Brown DG, Brady KT, et al. Comorbid anxiety disorders and baseline medication regimens predict clinical outcomes in individuals with co-occurring bipolar disorder and alcohol dependence: results of a randomized controlled trial. Psychiatry Res. 2011;188(3):361-365. doi:10.1016/j.psychres.2011.04.030
77. Ashok AH, Marques TR, Jauhar S, et al. The dopamine hypothesis of bipolar affective disorder: the state of the art and implications for treatment. Mol Psychiatry. 2017;22(5):666-679. doi:10.1038/mp.2017.16
78. Roberts RJ, Lohano KK, El-Mallakh RS. Antipsychotics as antidepressants. Asia Pac Psychiatry. 2016;8(3):179-188. doi:10.1111/appy.12186
79. Cipriani A, Barbui C, Salanti G, et al. Comparative efficacy and acceptability of antimanic drugs in acute mania: a multiple-treatments meta-analysis. Lancet. 2011;378(9799):1306-1315. doi:10.1016/S0140-6736(11)60873-8
80. Hayes JF, Marston L, Walters K, et al. Lithium vs. valproate vs. olanzapine vs. quetiapine as maintenance monotherapy for bipolar disorder: a population-based UK cohort study using electronic health records. World Psychiatry. 2016;15(1):53-58. doi:10.1002/wps.20298
81. Geddes JR, Gardiner A, Rendell J, et al. Comparative evaluation of quetiapine plus lamotrigine combination versus quetiapine monotherapy (and folic acid versus placebo) in bipolar depression (CEQUEL): a 2 × 2 factorial randomised trial. Lancet Psychiatry. 2016;3(1):31239. doi:10.1016/S2215-0366(15)00450-2
82. Goodwin GM, Haddad PM, Ferrier IN, et al. Evidence-based guidelines for treating bipolar disorder: revised third edition recommendations from the British Association for Psychopharmacology. J Psychopharmacol. 2016;30(6):495-553. doi:10.1177/0269881116636545
Depressed and cognitively impaired
CASE Depressed and anxious
Five years ago, Ms. X, age 60, was diagnosed with treatment-resistant major depressive disorder (MDD) with anxiety. This diagnosis was established by a previous psychiatrist. She presents to a clinic for a second opinion.
Since her diagnosis, Ms. X has experienced sad mood, anhedonia, difficulty falling asleep, increased appetite and weight, and decreased concentration and attention. Her anxiety stems from her inability to work, which causes her to worry about her children. In the clinic, the treatment team conducts the Patient Health Questionnaire-9 (PHQ-9) and Generalized Anxiety Disorder-7 item scale (GAD-7) with Ms. X. She scores 16 on the PHQ-9, indicating moderately severe depression, and scores 12 on the GAD-7, indicating moderate anxiety.
Ms. X’s current medication regimen consists of venlafaxine extended-release (XR) 225 mg/d, trazodone 100 mg/d at bedtime, and clonazepam 1 mg twice daily. She reports no significant improvement of her symptoms from these medications. Additionally, Ms. X reports that in the past she had been prescribed fluoxetine, citalopram, and duloxetine, but she cannot recall the dosages.
Ms. X appears appropriately groomed, maintains appropriate eye contact, has clear speech, and does not show evidence of internal stimulation; however, she has difficulty following instructions. She makes negative comments about herself such as “I’m worthless” and “Nobody cares about me.” The treatment team decides to taper Ms. X off venlafaxine XR and initiates sertraline 50 mg/d, while continuing trazodone 50 mg/d at bedtime and clonazepam 1 mg twice daily. The team refers her for cognitive-behavioral therapy (CBT) to address her cognitive distortions, sad mood, and anxiety. Ms. X is asked to follow up with Psychiatry in 1 week.
EVALUATION Unusual behavior
At her CBT intake, Ms. X endorses depression and anxiety. Her PHQ-9 score at this visit is 19 (moderately severe depression) and GAD-7 score is 16 (severe anxiety). The psychologist notes that Ms. X is able to complete activities of daily living and instrumental activities of daily living without assistance. Ms. X denies any use of illicit substances or alcohol. No gross memory impairment is noted during this appointment, though Ms. X exhibits unusual behavior, including exiting and re-entering the clinic multiple times to repeatedly ask about follow-up appointments. The psychologist concludes that Ms. X’s presentation and behavior can be explained by MDD and pseudodementia.
[polldaddy:12189562]
The authors’ observations
Pseudodementia gained recognition in clinical research >100 years ago.1 Officially coined by Kiloh in 1961, the term was used broadly to categorize psychiatric cases that present like dementia but are the result of reversible causes. More recently, it has been used to describe older adults who present with cognitive deficits in the context of depressive symptoms.2 The goal of evaluation is to determine if the primary issue is a cognitive disorder or a depressive episode. DSM-5-TR does not classify pseudodementia as a distinct diagnosis, but instead categorizes its symptoms as components under other major diagnostic categories. Patients can present with MDD and associated cognitive symptoms, or with a cognitive disorder with depressive symptoms, which would be diagnosed as a cognitive disorder with a major depressive-like episode.3
Pseudodementia is rare. Brodaty et al4 found the prevalence of pseudodementia in primary care settings was 0.6%. Older adults (age >65) who live alone are at increased risk of developing pseudodementia, which can be worsened by poor social support and acute psychosocial and environmental changes.5 A key characteristic of this disorder is that as the patient’s depressed mood improves, their memory and cognition also improve.6Table 13,6 outlines overlapping features of MDD and pseudodementia.
Continue to: EVALUATION Worsening depression
EVALUATION Worsening depression
At her Psychiatry follow-up appointment, Ms. X reports that her mood is worse since she ended the relationship with her partner and she feels anxious because the partner was financially supporting her. Her PHQ-9 score is 24 (severe depression) and her GAD-7 score is 12 (moderate anxiety). Ms. X reports tolerating her transition from venlafaxine XR 225 mg/d to sertraline 50 mg/d well.
Additionally, Ms. X reports her children have called her “useless” since she continues to have difficulties following through on household tasks, even though she has no physical impairments that prevent her from completing them. The Psychiatry team observes that Ms. X has no problems walking or moving her arms or legs.
The Psychiatry team administers the Montreal Cognitive Assessment (MoCA). Ms. X scores 22, indicating mild impairment.
The team recommends a neuropsychological assessment to determine if this MoCA score is due to a cognitive disorder or is rooted in her mood symptoms. The team also recommends an MRI of the brain, complete blood count (CBC), comprehensive metabolic panel (CMP), and urinalysis (UA).
[polldaddy:12189567]
Continue to: The authors' observations
The authors’ observations
Neuropsychological assessments are important tools for exploring the behavioral manifestations of brain dysfunction (Table 2).7 These assessments factor in elements of neurology, psychiatry, and psychology to provide information about the diagnosis, prognosis, and functional status of patients with medical conditions, especially those with neurocognitive and psychiatric disorders. They combine information from the patient and collateral interviews, behavioral observations, a review of patient records, and objective tests of motor, emotional, and cognitive function.
Among other uses, neuropsychological assessments can help identify depression in patients with neurologic impairment, determine the diagnosis and plan of care for patients with concussions, determine the risk of a motor vehicle crash in patients with cognitive impairment, and distinguish Alzheimer disease from vascular dementia.8 Components of such assessments include the Beck Anxiety Inventory (BAI) to assess anxiety, the Dementia Rating Scale-2 and Neuropsychological Assessment Battery-Screening Module to assess dementia, and the Beck Depression Inventory (BDI) to assess depression.9
EVALUATION Continued cognitive decline
A different psychologist performs the neuropsychological assessment, who conducts the Repeatable Battery for the Assessment of Neuropsychological Status Update to determine if Ms. X is experiencing cognitive impairment. Her immediate memory, visuospatial/constructions, language, attention, and delayed memory are significantly impaired for someone her age. The psychologist also administers the Wechsler Adult Intelligence Scale IV and finds Ms. X’s general cognitive ability is within the low average range of intellectual functioning as measured by Full-Scale IQ. Ms. X scores 29 on the BDI-II, indicating significant depressive symptoms, and 13 on the BAI, indicating mild anxiety symptoms.
Ms. X is diagnosed with MDD and an unspecified neurocognitive disorder. The psychologist recommends she start CBT to address her mood and anxiety symptoms.
Upon reviewing the results with Ms. X, the treatment team again recommends a brain MRI, CBC, CMP, and UA to rule out organic causes of her cognitive decline. Ms. X decides against the MRI and laboratory workup and elects to continue her present medication regimen and CBT.
Several weeks later, Ms. X’s family brings her to the emergency department (ED) for evaluation of worsening mood, decreased personal hygiene, increased irritability, and further cognitive decline. They report she is having an increasingly difficult time remembering things such as where she parked her car. The ED team decides to discontinue clonazepam but continues sertraline and trazodone.
Continue to: CBC, CMP, and UA...
CBC, CMP, and UA are unremarkable. Ms. X undergoes a brain CT scan without contrast, which reveals hyperdense lesions in the inferior left tentorium, posterior fossa. A subsequent brain MRI with contrast reveals a dural-based enhancing mass, inferior to the left tentorium, in the left posterior fossa measuring 2.2 cm x 2.1 cm, suggestive of a meningioma. The team orders a Neurosurgery consult.
[polldaddy:12189571]
The authors’ observations
While most brain tumors are secondary to metastasis, meningiomas are the most common primary CNS tumor. Typically, they are asymptomatic; their diagnosis is often delayed until the patient presents with psychiatric symptoms without any focal neurologic findings. The frontal lobe is the most common location of meningioma. Data from 48 case reports of patients with meningiomas and psychiatric symptoms suggest symptoms do not always correlate with specific brain regions.10,11
Indications for neuroimaging in cases such as Ms. X include an abrupt change in behavior or personality, lack of response to psychiatric treatment, presence of focal neurologic signs, and an unusual psychiatric presentation and development of symptoms.11
TREATMENT Neurosurgery
Neurosurgery recommends and performs a suboccipital craniotomy for biopsy and resection. Ms. X tolerates the procedure well. A meningioma is found in the posterior fossa, near the cerebellar convexity. A biopsy finds no evidence of malignancies.
At her postoperative follow-up appointment several days after the procedure, Ms. X reports new-onset hearing loss and tinnitus.
[polldaddy:12189747]
Continue to: The authors' observations
The authors’ observations
Patients who require neurosurgery typically already carry a heavy psychiatric burden, which makes it challenging to determine the exact psychiatric consequences of neurosurgery.12-14 For example, research shows that temporal lobe resection and temporal lobectomy for treatment-resistant epilepsy can lead to an exacerbation of baseline psychiatric symptoms and the development of new symptoms (31% to 34%).15,16 However, Bommakanti et al13 found no new psychiatric symptoms after resection of meningiomas, and surgery seemed to play a role in ameliorating psychiatric symptoms in patients with intracranial tumors. Research attempting to document the psychiatric sequelae of neurosurgery has had mixed results, and it is difficult to determine what effects brain surgery has on mental health.
OUTCOME Minimal improvement
Several weeks after neurosurgery, Ms. X and her family report her mood is improved. Her PHQ-9 score improves to 15, but her GAD-7 score increases to 13, 1 point above her previous score.
The treatment team recommends Ms. X continue taking sertraline 50 mg/d and trazodone 50 mg/d at bedtime. Ms. X’s family reports her cognition and memory have not improved; her MoCA score increases by 1 point to 23. The treatment team discusses with Ms. X and her family the possibility that her cognitive problems maybe better explained as a neurocognitive disorder rather than as a result of the meningioma, since her MoCA score has not significantly improved. Ms. X and her family decide to seek a second opinion from a neurologist.
Bottom Line
Pseudodementia is a term used to describe older adults who present with cognitive issues in the context of depressive symptoms. Even in the absence of focal findings, neuroimaging should be considered as part of the workup in patients who continue to experience a progressive decline in mood and cognitive function.
Related Resources
- Moller MD, Parmenter BA, Lane DW. Neuropsychological testing: a useful but underutilized resource. Current Psychiatry. 2019;18(11):40-46,51.
- Pollak J. Psychological/neuropsychological testing: when to refer for reexamination. Current Psychiatry. 2021;20(9):18- 19,30-31,37. doi:10.12788/cp.0157
Drug Brand Names
Citalopram • Celexa
Clonazepam • Klonopin
Duloxetine • Cymbalta
Fluoxetine • Prozac
Sertraline • Zoloft
Trazodone • Oleptro
Venlafaxine extended- release • Effexor XR
1. Nussbaum PD. (1994). Pseudodementia: a slow death. Neuropsychol Rev. 1994;4(2):71-90. doi:10.1007/BF01874829
2. Kang H, Zhao F, You L, et al. (2014). Pseudo-dementia: a neuropsychological review. Ann Indian Acad Neurol. 17(2):147-154. doi:10.4103/0972-2327.132613
3. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed, text revision. American Psychiatric Association; 2022.
4. Brodaty H, Connors MH. Pseudodementia, pseudo-pseudodementia, and pseudodepression. Alzheimers Dement (Amst). 2020;12(1):e12027. doi:10.1002/dad2.12027
5. Sekhon S, Marwaha R. Depressive Cognitive Disorders. StatPearls Publishing; 2022. https://www.ncbi.nlm.nih.gov/books/NBK559256/
6. Brown WA. Pseudodementia: issues in diagnosis. Psychiatric Times. April 9, 2005. Accessed February 3, 2023. www.psychiatrictimes.com/view/pseudodementia-issues-diagnosis
7. Kulas JF, Naugle RI. (2003). Indications for neuropsychological assessment. Cleve Clin J Med. 2003;70(9):785-792.
8. Braun M, Tupper D, Kaufmann P, et al. Neuropsychological assessment: a valuable tool in the diagnosis and management of neurological, neurodevelopmental, medical, and psychiatric disorders. Cogn Behav Neurol. 2011;24(3):107-114.
9. Michels TC, Tiu AY, Graver CJ. Neuropsychological evaluation in primary care. Am Fam Physician. 2010;82(5):495-502.
10. Wiemels J, Wrensch M, Claus EB. Epidemiology and etiology of meningioma. J Neurooncol. 2010;99(3):307-314. doi:10.1007/s11060-010-0386-3
11. Gyawali S, Sharma P, Mahapatra A. Meningioma and psychiatric symptoms: an individual patient data analysis. Asian J Psychiatr. 2019;42:94-103. doi:10.1016/j.ajp.2019.03.029
12. McAllister TW. Neurobehavioral sequelae of traumatic brain injury: evaluation and management. World Psychiatry. 2008;7(1):3-10. doi:10.1002/j.2051-5545.2008.tb00139.x
13. Bommakanti K, Gaddamanugu P, Alladi S, et al. Pre-operative and post-operative psychiatric manifestations in patients with supratentorial meningiomas. Clin Neurol Neurosurg. 2016;147:24-29. doi:10.1016/j.clineuro.2016.05.018
14. Devinsky O, Barr WB, Vickrey BG, et al. Changes in depression and anxiety after resective surgery for epilepsy. Neurology. 2005;65(11):1744-1749. doi:10.1212/01.wnl.0000187114.71524.c3
15. Blumer D, Wakhlu S, Davies K, et al. Psychiatric outcome of temporal lobectomy for epilepsy: incidence and treatment of psychiatric complications. Epilepsia. 1998;39(5):478-486. doi:10.1111/j.1528-1157.1998.tb01409.x
16. Glosser G, Zwil AS, Glosser DS, et al. Psychiatric aspects of temporal lobe epilepsy before and after anterior temporal lobectomy. J Neurol Neurosurg Psychiatry. 2000;68(1):53-58. doi:10.1136/jnnp.68.1.53
CASE Depressed and anxious
Five years ago, Ms. X, age 60, was diagnosed with treatment-resistant major depressive disorder (MDD) with anxiety. This diagnosis was established by a previous psychiatrist. She presents to a clinic for a second opinion.
Since her diagnosis, Ms. X has experienced sad mood, anhedonia, difficulty falling asleep, increased appetite and weight, and decreased concentration and attention. Her anxiety stems from her inability to work, which causes her to worry about her children. In the clinic, the treatment team conducts the Patient Health Questionnaire-9 (PHQ-9) and Generalized Anxiety Disorder-7 item scale (GAD-7) with Ms. X. She scores 16 on the PHQ-9, indicating moderately severe depression, and scores 12 on the GAD-7, indicating moderate anxiety.
Ms. X’s current medication regimen consists of venlafaxine extended-release (XR) 225 mg/d, trazodone 100 mg/d at bedtime, and clonazepam 1 mg twice daily. She reports no significant improvement of her symptoms from these medications. Additionally, Ms. X reports that in the past she had been prescribed fluoxetine, citalopram, and duloxetine, but she cannot recall the dosages.
Ms. X appears appropriately groomed, maintains appropriate eye contact, has clear speech, and does not show evidence of internal stimulation; however, she has difficulty following instructions. She makes negative comments about herself such as “I’m worthless” and “Nobody cares about me.” The treatment team decides to taper Ms. X off venlafaxine XR and initiates sertraline 50 mg/d, while continuing trazodone 50 mg/d at bedtime and clonazepam 1 mg twice daily. The team refers her for cognitive-behavioral therapy (CBT) to address her cognitive distortions, sad mood, and anxiety. Ms. X is asked to follow up with Psychiatry in 1 week.
EVALUATION Unusual behavior
At her CBT intake, Ms. X endorses depression and anxiety. Her PHQ-9 score at this visit is 19 (moderately severe depression) and GAD-7 score is 16 (severe anxiety). The psychologist notes that Ms. X is able to complete activities of daily living and instrumental activities of daily living without assistance. Ms. X denies any use of illicit substances or alcohol. No gross memory impairment is noted during this appointment, though Ms. X exhibits unusual behavior, including exiting and re-entering the clinic multiple times to repeatedly ask about follow-up appointments. The psychologist concludes that Ms. X’s presentation and behavior can be explained by MDD and pseudodementia.
[polldaddy:12189562]
The authors’ observations
Pseudodementia gained recognition in clinical research >100 years ago.1 Officially coined by Kiloh in 1961, the term was used broadly to categorize psychiatric cases that present like dementia but are the result of reversible causes. More recently, it has been used to describe older adults who present with cognitive deficits in the context of depressive symptoms.2 The goal of evaluation is to determine if the primary issue is a cognitive disorder or a depressive episode. DSM-5-TR does not classify pseudodementia as a distinct diagnosis, but instead categorizes its symptoms as components under other major diagnostic categories. Patients can present with MDD and associated cognitive symptoms, or with a cognitive disorder with depressive symptoms, which would be diagnosed as a cognitive disorder with a major depressive-like episode.3
Pseudodementia is rare. Brodaty et al4 found the prevalence of pseudodementia in primary care settings was 0.6%. Older adults (age >65) who live alone are at increased risk of developing pseudodementia, which can be worsened by poor social support and acute psychosocial and environmental changes.5 A key characteristic of this disorder is that as the patient’s depressed mood improves, their memory and cognition also improve.6Table 13,6 outlines overlapping features of MDD and pseudodementia.
Continue to: EVALUATION Worsening depression
EVALUATION Worsening depression
At her Psychiatry follow-up appointment, Ms. X reports that her mood is worse since she ended the relationship with her partner and she feels anxious because the partner was financially supporting her. Her PHQ-9 score is 24 (severe depression) and her GAD-7 score is 12 (moderate anxiety). Ms. X reports tolerating her transition from venlafaxine XR 225 mg/d to sertraline 50 mg/d well.
Additionally, Ms. X reports her children have called her “useless” since she continues to have difficulties following through on household tasks, even though she has no physical impairments that prevent her from completing them. The Psychiatry team observes that Ms. X has no problems walking or moving her arms or legs.
The Psychiatry team administers the Montreal Cognitive Assessment (MoCA). Ms. X scores 22, indicating mild impairment.
The team recommends a neuropsychological assessment to determine if this MoCA score is due to a cognitive disorder or is rooted in her mood symptoms. The team also recommends an MRI of the brain, complete blood count (CBC), comprehensive metabolic panel (CMP), and urinalysis (UA).
[polldaddy:12189567]
Continue to: The authors' observations
The authors’ observations
Neuropsychological assessments are important tools for exploring the behavioral manifestations of brain dysfunction (Table 2).7 These assessments factor in elements of neurology, psychiatry, and psychology to provide information about the diagnosis, prognosis, and functional status of patients with medical conditions, especially those with neurocognitive and psychiatric disorders. They combine information from the patient and collateral interviews, behavioral observations, a review of patient records, and objective tests of motor, emotional, and cognitive function.
Among other uses, neuropsychological assessments can help identify depression in patients with neurologic impairment, determine the diagnosis and plan of care for patients with concussions, determine the risk of a motor vehicle crash in patients with cognitive impairment, and distinguish Alzheimer disease from vascular dementia.8 Components of such assessments include the Beck Anxiety Inventory (BAI) to assess anxiety, the Dementia Rating Scale-2 and Neuropsychological Assessment Battery-Screening Module to assess dementia, and the Beck Depression Inventory (BDI) to assess depression.9
EVALUATION Continued cognitive decline
A different psychologist performs the neuropsychological assessment, who conducts the Repeatable Battery for the Assessment of Neuropsychological Status Update to determine if Ms. X is experiencing cognitive impairment. Her immediate memory, visuospatial/constructions, language, attention, and delayed memory are significantly impaired for someone her age. The psychologist also administers the Wechsler Adult Intelligence Scale IV and finds Ms. X’s general cognitive ability is within the low average range of intellectual functioning as measured by Full-Scale IQ. Ms. X scores 29 on the BDI-II, indicating significant depressive symptoms, and 13 on the BAI, indicating mild anxiety symptoms.
Ms. X is diagnosed with MDD and an unspecified neurocognitive disorder. The psychologist recommends she start CBT to address her mood and anxiety symptoms.
Upon reviewing the results with Ms. X, the treatment team again recommends a brain MRI, CBC, CMP, and UA to rule out organic causes of her cognitive decline. Ms. X decides against the MRI and laboratory workup and elects to continue her present medication regimen and CBT.
Several weeks later, Ms. X’s family brings her to the emergency department (ED) for evaluation of worsening mood, decreased personal hygiene, increased irritability, and further cognitive decline. They report she is having an increasingly difficult time remembering things such as where she parked her car. The ED team decides to discontinue clonazepam but continues sertraline and trazodone.
Continue to: CBC, CMP, and UA...
CBC, CMP, and UA are unremarkable. Ms. X undergoes a brain CT scan without contrast, which reveals hyperdense lesions in the inferior left tentorium, posterior fossa. A subsequent brain MRI with contrast reveals a dural-based enhancing mass, inferior to the left tentorium, in the left posterior fossa measuring 2.2 cm x 2.1 cm, suggestive of a meningioma. The team orders a Neurosurgery consult.
[polldaddy:12189571]
The authors’ observations
While most brain tumors are secondary to metastasis, meningiomas are the most common primary CNS tumor. Typically, they are asymptomatic; their diagnosis is often delayed until the patient presents with psychiatric symptoms without any focal neurologic findings. The frontal lobe is the most common location of meningioma. Data from 48 case reports of patients with meningiomas and psychiatric symptoms suggest symptoms do not always correlate with specific brain regions.10,11
Indications for neuroimaging in cases such as Ms. X include an abrupt change in behavior or personality, lack of response to psychiatric treatment, presence of focal neurologic signs, and an unusual psychiatric presentation and development of symptoms.11
TREATMENT Neurosurgery
Neurosurgery recommends and performs a suboccipital craniotomy for biopsy and resection. Ms. X tolerates the procedure well. A meningioma is found in the posterior fossa, near the cerebellar convexity. A biopsy finds no evidence of malignancies.
At her postoperative follow-up appointment several days after the procedure, Ms. X reports new-onset hearing loss and tinnitus.
[polldaddy:12189747]
Continue to: The authors' observations
The authors’ observations
Patients who require neurosurgery typically already carry a heavy psychiatric burden, which makes it challenging to determine the exact psychiatric consequences of neurosurgery.12-14 For example, research shows that temporal lobe resection and temporal lobectomy for treatment-resistant epilepsy can lead to an exacerbation of baseline psychiatric symptoms and the development of new symptoms (31% to 34%).15,16 However, Bommakanti et al13 found no new psychiatric symptoms after resection of meningiomas, and surgery seemed to play a role in ameliorating psychiatric symptoms in patients with intracranial tumors. Research attempting to document the psychiatric sequelae of neurosurgery has had mixed results, and it is difficult to determine what effects brain surgery has on mental health.
OUTCOME Minimal improvement
Several weeks after neurosurgery, Ms. X and her family report her mood is improved. Her PHQ-9 score improves to 15, but her GAD-7 score increases to 13, 1 point above her previous score.
The treatment team recommends Ms. X continue taking sertraline 50 mg/d and trazodone 50 mg/d at bedtime. Ms. X’s family reports her cognition and memory have not improved; her MoCA score increases by 1 point to 23. The treatment team discusses with Ms. X and her family the possibility that her cognitive problems maybe better explained as a neurocognitive disorder rather than as a result of the meningioma, since her MoCA score has not significantly improved. Ms. X and her family decide to seek a second opinion from a neurologist.
Bottom Line
Pseudodementia is a term used to describe older adults who present with cognitive issues in the context of depressive symptoms. Even in the absence of focal findings, neuroimaging should be considered as part of the workup in patients who continue to experience a progressive decline in mood and cognitive function.
Related Resources
- Moller MD, Parmenter BA, Lane DW. Neuropsychological testing: a useful but underutilized resource. Current Psychiatry. 2019;18(11):40-46,51.
- Pollak J. Psychological/neuropsychological testing: when to refer for reexamination. Current Psychiatry. 2021;20(9):18- 19,30-31,37. doi:10.12788/cp.0157
Drug Brand Names
Citalopram • Celexa
Clonazepam • Klonopin
Duloxetine • Cymbalta
Fluoxetine • Prozac
Sertraline • Zoloft
Trazodone • Oleptro
Venlafaxine extended- release • Effexor XR
CASE Depressed and anxious
Five years ago, Ms. X, age 60, was diagnosed with treatment-resistant major depressive disorder (MDD) with anxiety. This diagnosis was established by a previous psychiatrist. She presents to a clinic for a second opinion.
Since her diagnosis, Ms. X has experienced sad mood, anhedonia, difficulty falling asleep, increased appetite and weight, and decreased concentration and attention. Her anxiety stems from her inability to work, which causes her to worry about her children. In the clinic, the treatment team conducts the Patient Health Questionnaire-9 (PHQ-9) and Generalized Anxiety Disorder-7 item scale (GAD-7) with Ms. X. She scores 16 on the PHQ-9, indicating moderately severe depression, and scores 12 on the GAD-7, indicating moderate anxiety.
Ms. X’s current medication regimen consists of venlafaxine extended-release (XR) 225 mg/d, trazodone 100 mg/d at bedtime, and clonazepam 1 mg twice daily. She reports no significant improvement of her symptoms from these medications. Additionally, Ms. X reports that in the past she had been prescribed fluoxetine, citalopram, and duloxetine, but she cannot recall the dosages.
Ms. X appears appropriately groomed, maintains appropriate eye contact, has clear speech, and does not show evidence of internal stimulation; however, she has difficulty following instructions. She makes negative comments about herself such as “I’m worthless” and “Nobody cares about me.” The treatment team decides to taper Ms. X off venlafaxine XR and initiates sertraline 50 mg/d, while continuing trazodone 50 mg/d at bedtime and clonazepam 1 mg twice daily. The team refers her for cognitive-behavioral therapy (CBT) to address her cognitive distortions, sad mood, and anxiety. Ms. X is asked to follow up with Psychiatry in 1 week.
EVALUATION Unusual behavior
At her CBT intake, Ms. X endorses depression and anxiety. Her PHQ-9 score at this visit is 19 (moderately severe depression) and GAD-7 score is 16 (severe anxiety). The psychologist notes that Ms. X is able to complete activities of daily living and instrumental activities of daily living without assistance. Ms. X denies any use of illicit substances or alcohol. No gross memory impairment is noted during this appointment, though Ms. X exhibits unusual behavior, including exiting and re-entering the clinic multiple times to repeatedly ask about follow-up appointments. The psychologist concludes that Ms. X’s presentation and behavior can be explained by MDD and pseudodementia.
[polldaddy:12189562]
The authors’ observations
Pseudodementia gained recognition in clinical research >100 years ago.1 Officially coined by Kiloh in 1961, the term was used broadly to categorize psychiatric cases that present like dementia but are the result of reversible causes. More recently, it has been used to describe older adults who present with cognitive deficits in the context of depressive symptoms.2 The goal of evaluation is to determine if the primary issue is a cognitive disorder or a depressive episode. DSM-5-TR does not classify pseudodementia as a distinct diagnosis, but instead categorizes its symptoms as components under other major diagnostic categories. Patients can present with MDD and associated cognitive symptoms, or with a cognitive disorder with depressive symptoms, which would be diagnosed as a cognitive disorder with a major depressive-like episode.3
Pseudodementia is rare. Brodaty et al4 found the prevalence of pseudodementia in primary care settings was 0.6%. Older adults (age >65) who live alone are at increased risk of developing pseudodementia, which can be worsened by poor social support and acute psychosocial and environmental changes.5 A key characteristic of this disorder is that as the patient’s depressed mood improves, their memory and cognition also improve.6Table 13,6 outlines overlapping features of MDD and pseudodementia.
Continue to: EVALUATION Worsening depression
EVALUATION Worsening depression
At her Psychiatry follow-up appointment, Ms. X reports that her mood is worse since she ended the relationship with her partner and she feels anxious because the partner was financially supporting her. Her PHQ-9 score is 24 (severe depression) and her GAD-7 score is 12 (moderate anxiety). Ms. X reports tolerating her transition from venlafaxine XR 225 mg/d to sertraline 50 mg/d well.
Additionally, Ms. X reports her children have called her “useless” since she continues to have difficulties following through on household tasks, even though she has no physical impairments that prevent her from completing them. The Psychiatry team observes that Ms. X has no problems walking or moving her arms or legs.
The Psychiatry team administers the Montreal Cognitive Assessment (MoCA). Ms. X scores 22, indicating mild impairment.
The team recommends a neuropsychological assessment to determine if this MoCA score is due to a cognitive disorder or is rooted in her mood symptoms. The team also recommends an MRI of the brain, complete blood count (CBC), comprehensive metabolic panel (CMP), and urinalysis (UA).
[polldaddy:12189567]
Continue to: The authors' observations
The authors’ observations
Neuropsychological assessments are important tools for exploring the behavioral manifestations of brain dysfunction (Table 2).7 These assessments factor in elements of neurology, psychiatry, and psychology to provide information about the diagnosis, prognosis, and functional status of patients with medical conditions, especially those with neurocognitive and psychiatric disorders. They combine information from the patient and collateral interviews, behavioral observations, a review of patient records, and objective tests of motor, emotional, and cognitive function.
Among other uses, neuropsychological assessments can help identify depression in patients with neurologic impairment, determine the diagnosis and plan of care for patients with concussions, determine the risk of a motor vehicle crash in patients with cognitive impairment, and distinguish Alzheimer disease from vascular dementia.8 Components of such assessments include the Beck Anxiety Inventory (BAI) to assess anxiety, the Dementia Rating Scale-2 and Neuropsychological Assessment Battery-Screening Module to assess dementia, and the Beck Depression Inventory (BDI) to assess depression.9
EVALUATION Continued cognitive decline
A different psychologist performs the neuropsychological assessment, who conducts the Repeatable Battery for the Assessment of Neuropsychological Status Update to determine if Ms. X is experiencing cognitive impairment. Her immediate memory, visuospatial/constructions, language, attention, and delayed memory are significantly impaired for someone her age. The psychologist also administers the Wechsler Adult Intelligence Scale IV and finds Ms. X’s general cognitive ability is within the low average range of intellectual functioning as measured by Full-Scale IQ. Ms. X scores 29 on the BDI-II, indicating significant depressive symptoms, and 13 on the BAI, indicating mild anxiety symptoms.
Ms. X is diagnosed with MDD and an unspecified neurocognitive disorder. The psychologist recommends she start CBT to address her mood and anxiety symptoms.
Upon reviewing the results with Ms. X, the treatment team again recommends a brain MRI, CBC, CMP, and UA to rule out organic causes of her cognitive decline. Ms. X decides against the MRI and laboratory workup and elects to continue her present medication regimen and CBT.
Several weeks later, Ms. X’s family brings her to the emergency department (ED) for evaluation of worsening mood, decreased personal hygiene, increased irritability, and further cognitive decline. They report she is having an increasingly difficult time remembering things such as where she parked her car. The ED team decides to discontinue clonazepam but continues sertraline and trazodone.
Continue to: CBC, CMP, and UA...
CBC, CMP, and UA are unremarkable. Ms. X undergoes a brain CT scan without contrast, which reveals hyperdense lesions in the inferior left tentorium, posterior fossa. A subsequent brain MRI with contrast reveals a dural-based enhancing mass, inferior to the left tentorium, in the left posterior fossa measuring 2.2 cm x 2.1 cm, suggestive of a meningioma. The team orders a Neurosurgery consult.
[polldaddy:12189571]
The authors’ observations
While most brain tumors are secondary to metastasis, meningiomas are the most common primary CNS tumor. Typically, they are asymptomatic; their diagnosis is often delayed until the patient presents with psychiatric symptoms without any focal neurologic findings. The frontal lobe is the most common location of meningioma. Data from 48 case reports of patients with meningiomas and psychiatric symptoms suggest symptoms do not always correlate with specific brain regions.10,11
Indications for neuroimaging in cases such as Ms. X include an abrupt change in behavior or personality, lack of response to psychiatric treatment, presence of focal neurologic signs, and an unusual psychiatric presentation and development of symptoms.11
TREATMENT Neurosurgery
Neurosurgery recommends and performs a suboccipital craniotomy for biopsy and resection. Ms. X tolerates the procedure well. A meningioma is found in the posterior fossa, near the cerebellar convexity. A biopsy finds no evidence of malignancies.
At her postoperative follow-up appointment several days after the procedure, Ms. X reports new-onset hearing loss and tinnitus.
[polldaddy:12189747]
Continue to: The authors' observations
The authors’ observations
Patients who require neurosurgery typically already carry a heavy psychiatric burden, which makes it challenging to determine the exact psychiatric consequences of neurosurgery.12-14 For example, research shows that temporal lobe resection and temporal lobectomy for treatment-resistant epilepsy can lead to an exacerbation of baseline psychiatric symptoms and the development of new symptoms (31% to 34%).15,16 However, Bommakanti et al13 found no new psychiatric symptoms after resection of meningiomas, and surgery seemed to play a role in ameliorating psychiatric symptoms in patients with intracranial tumors. Research attempting to document the psychiatric sequelae of neurosurgery has had mixed results, and it is difficult to determine what effects brain surgery has on mental health.
OUTCOME Minimal improvement
Several weeks after neurosurgery, Ms. X and her family report her mood is improved. Her PHQ-9 score improves to 15, but her GAD-7 score increases to 13, 1 point above her previous score.
The treatment team recommends Ms. X continue taking sertraline 50 mg/d and trazodone 50 mg/d at bedtime. Ms. X’s family reports her cognition and memory have not improved; her MoCA score increases by 1 point to 23. The treatment team discusses with Ms. X and her family the possibility that her cognitive problems maybe better explained as a neurocognitive disorder rather than as a result of the meningioma, since her MoCA score has not significantly improved. Ms. X and her family decide to seek a second opinion from a neurologist.
Bottom Line
Pseudodementia is a term used to describe older adults who present with cognitive issues in the context of depressive symptoms. Even in the absence of focal findings, neuroimaging should be considered as part of the workup in patients who continue to experience a progressive decline in mood and cognitive function.
Related Resources
- Moller MD, Parmenter BA, Lane DW. Neuropsychological testing: a useful but underutilized resource. Current Psychiatry. 2019;18(11):40-46,51.
- Pollak J. Psychological/neuropsychological testing: when to refer for reexamination. Current Psychiatry. 2021;20(9):18- 19,30-31,37. doi:10.12788/cp.0157
Drug Brand Names
Citalopram • Celexa
Clonazepam • Klonopin
Duloxetine • Cymbalta
Fluoxetine • Prozac
Sertraline • Zoloft
Trazodone • Oleptro
Venlafaxine extended- release • Effexor XR
1. Nussbaum PD. (1994). Pseudodementia: a slow death. Neuropsychol Rev. 1994;4(2):71-90. doi:10.1007/BF01874829
2. Kang H, Zhao F, You L, et al. (2014). Pseudo-dementia: a neuropsychological review. Ann Indian Acad Neurol. 17(2):147-154. doi:10.4103/0972-2327.132613
3. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed, text revision. American Psychiatric Association; 2022.
4. Brodaty H, Connors MH. Pseudodementia, pseudo-pseudodementia, and pseudodepression. Alzheimers Dement (Amst). 2020;12(1):e12027. doi:10.1002/dad2.12027
5. Sekhon S, Marwaha R. Depressive Cognitive Disorders. StatPearls Publishing; 2022. https://www.ncbi.nlm.nih.gov/books/NBK559256/
6. Brown WA. Pseudodementia: issues in diagnosis. Psychiatric Times. April 9, 2005. Accessed February 3, 2023. www.psychiatrictimes.com/view/pseudodementia-issues-diagnosis
7. Kulas JF, Naugle RI. (2003). Indications for neuropsychological assessment. Cleve Clin J Med. 2003;70(9):785-792.
8. Braun M, Tupper D, Kaufmann P, et al. Neuropsychological assessment: a valuable tool in the diagnosis and management of neurological, neurodevelopmental, medical, and psychiatric disorders. Cogn Behav Neurol. 2011;24(3):107-114.
9. Michels TC, Tiu AY, Graver CJ. Neuropsychological evaluation in primary care. Am Fam Physician. 2010;82(5):495-502.
10. Wiemels J, Wrensch M, Claus EB. Epidemiology and etiology of meningioma. J Neurooncol. 2010;99(3):307-314. doi:10.1007/s11060-010-0386-3
11. Gyawali S, Sharma P, Mahapatra A. Meningioma and psychiatric symptoms: an individual patient data analysis. Asian J Psychiatr. 2019;42:94-103. doi:10.1016/j.ajp.2019.03.029
12. McAllister TW. Neurobehavioral sequelae of traumatic brain injury: evaluation and management. World Psychiatry. 2008;7(1):3-10. doi:10.1002/j.2051-5545.2008.tb00139.x
13. Bommakanti K, Gaddamanugu P, Alladi S, et al. Pre-operative and post-operative psychiatric manifestations in patients with supratentorial meningiomas. Clin Neurol Neurosurg. 2016;147:24-29. doi:10.1016/j.clineuro.2016.05.018
14. Devinsky O, Barr WB, Vickrey BG, et al. Changes in depression and anxiety after resective surgery for epilepsy. Neurology. 2005;65(11):1744-1749. doi:10.1212/01.wnl.0000187114.71524.c3
15. Blumer D, Wakhlu S, Davies K, et al. Psychiatric outcome of temporal lobectomy for epilepsy: incidence and treatment of psychiatric complications. Epilepsia. 1998;39(5):478-486. doi:10.1111/j.1528-1157.1998.tb01409.x
16. Glosser G, Zwil AS, Glosser DS, et al. Psychiatric aspects of temporal lobe epilepsy before and after anterior temporal lobectomy. J Neurol Neurosurg Psychiatry. 2000;68(1):53-58. doi:10.1136/jnnp.68.1.53
1. Nussbaum PD. (1994). Pseudodementia: a slow death. Neuropsychol Rev. 1994;4(2):71-90. doi:10.1007/BF01874829
2. Kang H, Zhao F, You L, et al. (2014). Pseudo-dementia: a neuropsychological review. Ann Indian Acad Neurol. 17(2):147-154. doi:10.4103/0972-2327.132613
3. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed, text revision. American Psychiatric Association; 2022.
4. Brodaty H, Connors MH. Pseudodementia, pseudo-pseudodementia, and pseudodepression. Alzheimers Dement (Amst). 2020;12(1):e12027. doi:10.1002/dad2.12027
5. Sekhon S, Marwaha R. Depressive Cognitive Disorders. StatPearls Publishing; 2022. https://www.ncbi.nlm.nih.gov/books/NBK559256/
6. Brown WA. Pseudodementia: issues in diagnosis. Psychiatric Times. April 9, 2005. Accessed February 3, 2023. www.psychiatrictimes.com/view/pseudodementia-issues-diagnosis
7. Kulas JF, Naugle RI. (2003). Indications for neuropsychological assessment. Cleve Clin J Med. 2003;70(9):785-792.
8. Braun M, Tupper D, Kaufmann P, et al. Neuropsychological assessment: a valuable tool in the diagnosis and management of neurological, neurodevelopmental, medical, and psychiatric disorders. Cogn Behav Neurol. 2011;24(3):107-114.
9. Michels TC, Tiu AY, Graver CJ. Neuropsychological evaluation in primary care. Am Fam Physician. 2010;82(5):495-502.
10. Wiemels J, Wrensch M, Claus EB. Epidemiology and etiology of meningioma. J Neurooncol. 2010;99(3):307-314. doi:10.1007/s11060-010-0386-3
11. Gyawali S, Sharma P, Mahapatra A. Meningioma and psychiatric symptoms: an individual patient data analysis. Asian J Psychiatr. 2019;42:94-103. doi:10.1016/j.ajp.2019.03.029
12. McAllister TW. Neurobehavioral sequelae of traumatic brain injury: evaluation and management. World Psychiatry. 2008;7(1):3-10. doi:10.1002/j.2051-5545.2008.tb00139.x
13. Bommakanti K, Gaddamanugu P, Alladi S, et al. Pre-operative and post-operative psychiatric manifestations in patients with supratentorial meningiomas. Clin Neurol Neurosurg. 2016;147:24-29. doi:10.1016/j.clineuro.2016.05.018
14. Devinsky O, Barr WB, Vickrey BG, et al. Changes in depression and anxiety after resective surgery for epilepsy. Neurology. 2005;65(11):1744-1749. doi:10.1212/01.wnl.0000187114.71524.c3
15. Blumer D, Wakhlu S, Davies K, et al. Psychiatric outcome of temporal lobectomy for epilepsy: incidence and treatment of psychiatric complications. Epilepsia. 1998;39(5):478-486. doi:10.1111/j.1528-1157.1998.tb01409.x
16. Glosser G, Zwil AS, Glosser DS, et al. Psychiatric aspects of temporal lobe epilepsy before and after anterior temporal lobectomy. J Neurol Neurosurg Psychiatry. 2000;68(1):53-58. doi:10.1136/jnnp.68.1.53
Tips for efficient night shift work in a psychiatric ED
Attending psychiatrists who work night shift in a psychiatric emergency department (ED) or medical ED require a different set of skills than when working daytime or evening shifts, especially when working full-time or solo. While all patients should be treated carefully and meticulously regardless of the shift, this article offers tips for efficiency for solo attending psychiatrists who work night shift in an ED.
Check orders. Typically, multiple psychiatric clinicians are available on other shifts, but only 1 at night. This can lead to significant variability and potential errors in patients’ orders. Such errors filter down to night shift and often must be addressed by the solo clinician, who can’t say “that person is not my patient” because there are no other clinicians available to help. Carefully check orders (ideally, on all patients every shift) to ensure there are no errors or omissions.
Use note templates. While it is important to avoid using mere checklists, with electronic medical record systems, create templates for typical notes. This will save time when the pace of patients increases.
Be brief in your documentation. Brevity is key when documenting at night. Focus on what is necessary and sufficient.
Conduct thorough but efficient interviews. Be aware of how much time you spend on patient interviews. While still thorough, interviews must often be shorter due to a higher staff-to-patient ratio at night.
Be aware of potential medical issues. Many psychiatric EDs are not attached to a hospital. With other medical consultants not readily available in the middle of the night, be particularly alert for any acute medical issues that may arise, and act accordingly.
Focus on the order of tasks. Be aware of which tasks you complete and in what order. For example, at night you may need to medicate sooner for agitation because other patients are sleeping, instead of letting one patient’s agitation disrupt the entire night milieu.
Continue to: Don't let tasks pile up
Don’t let tasks pile up. Time management and multitasking are key skills at night. Take care of clinical issues as they arise. Finish documentation as you go along. Don’t let things pile up throughout your shift and then spend significant time after your shift to catch up.
Know your staff. The staff around you are your eyes and ears. Get to know your clinical and nonclinical staff’s tendencies. This can be immensely helpful in picking up any different patterns when interviewing and observing patients.
Know your limits. You may not be able to solve everything or obtain the ideal collateral at night. Don’t get caught up in definitively trying to resolve things and end up wasting precious time at night. Let it go. Don’t overthink. If all else fails, hold the patient overnight.
Prioritize self-care. Night shift work has been shown to negatively impact one’s health.1-3 If you choose this type of work, either part-time or full-time, maintain your own health by exercising regularly, eating a healthy diet, obtaining adequate rest between shifts, and seeing your health care team often.
1. Wu QJ, Sun H, Wen ZY, et al. Shift work and health outcomes: an umbrella review of systematic reviews and meta-analyses of epidemiological studies. J Clin Sleep Med. 2022;18(2):653-662. doi:10.5664/jcsm.9642
2. Kecklund G, Axelsson J. Health consequences of shift work and insufficient sleep. BMJ. 2016;355:i5210. doi:10.1136/bmj.i5210
3. Boivin DB, Boudreau P. Impacts of shift work on sleep and circadian rhythms. Pathol Biol (Paris). 2014;62(5):292-301. doi:10.1016/j.patbio.2014.08.001
Attending psychiatrists who work night shift in a psychiatric emergency department (ED) or medical ED require a different set of skills than when working daytime or evening shifts, especially when working full-time or solo. While all patients should be treated carefully and meticulously regardless of the shift, this article offers tips for efficiency for solo attending psychiatrists who work night shift in an ED.
Check orders. Typically, multiple psychiatric clinicians are available on other shifts, but only 1 at night. This can lead to significant variability and potential errors in patients’ orders. Such errors filter down to night shift and often must be addressed by the solo clinician, who can’t say “that person is not my patient” because there are no other clinicians available to help. Carefully check orders (ideally, on all patients every shift) to ensure there are no errors or omissions.
Use note templates. While it is important to avoid using mere checklists, with electronic medical record systems, create templates for typical notes. This will save time when the pace of patients increases.
Be brief in your documentation. Brevity is key when documenting at night. Focus on what is necessary and sufficient.
Conduct thorough but efficient interviews. Be aware of how much time you spend on patient interviews. While still thorough, interviews must often be shorter due to a higher staff-to-patient ratio at night.
Be aware of potential medical issues. Many psychiatric EDs are not attached to a hospital. With other medical consultants not readily available in the middle of the night, be particularly alert for any acute medical issues that may arise, and act accordingly.
Focus on the order of tasks. Be aware of which tasks you complete and in what order. For example, at night you may need to medicate sooner for agitation because other patients are sleeping, instead of letting one patient’s agitation disrupt the entire night milieu.
Continue to: Don't let tasks pile up
Don’t let tasks pile up. Time management and multitasking are key skills at night. Take care of clinical issues as they arise. Finish documentation as you go along. Don’t let things pile up throughout your shift and then spend significant time after your shift to catch up.
Know your staff. The staff around you are your eyes and ears. Get to know your clinical and nonclinical staff’s tendencies. This can be immensely helpful in picking up any different patterns when interviewing and observing patients.
Know your limits. You may not be able to solve everything or obtain the ideal collateral at night. Don’t get caught up in definitively trying to resolve things and end up wasting precious time at night. Let it go. Don’t overthink. If all else fails, hold the patient overnight.
Prioritize self-care. Night shift work has been shown to negatively impact one’s health.1-3 If you choose this type of work, either part-time or full-time, maintain your own health by exercising regularly, eating a healthy diet, obtaining adequate rest between shifts, and seeing your health care team often.
Attending psychiatrists who work night shift in a psychiatric emergency department (ED) or medical ED require a different set of skills than when working daytime or evening shifts, especially when working full-time or solo. While all patients should be treated carefully and meticulously regardless of the shift, this article offers tips for efficiency for solo attending psychiatrists who work night shift in an ED.
Check orders. Typically, multiple psychiatric clinicians are available on other shifts, but only 1 at night. This can lead to significant variability and potential errors in patients’ orders. Such errors filter down to night shift and often must be addressed by the solo clinician, who can’t say “that person is not my patient” because there are no other clinicians available to help. Carefully check orders (ideally, on all patients every shift) to ensure there are no errors or omissions.
Use note templates. While it is important to avoid using mere checklists, with electronic medical record systems, create templates for typical notes. This will save time when the pace of patients increases.
Be brief in your documentation. Brevity is key when documenting at night. Focus on what is necessary and sufficient.
Conduct thorough but efficient interviews. Be aware of how much time you spend on patient interviews. While still thorough, interviews must often be shorter due to a higher staff-to-patient ratio at night.
Be aware of potential medical issues. Many psychiatric EDs are not attached to a hospital. With other medical consultants not readily available in the middle of the night, be particularly alert for any acute medical issues that may arise, and act accordingly.
Focus on the order of tasks. Be aware of which tasks you complete and in what order. For example, at night you may need to medicate sooner for agitation because other patients are sleeping, instead of letting one patient’s agitation disrupt the entire night milieu.
Continue to: Don't let tasks pile up
Don’t let tasks pile up. Time management and multitasking are key skills at night. Take care of clinical issues as they arise. Finish documentation as you go along. Don’t let things pile up throughout your shift and then spend significant time after your shift to catch up.
Know your staff. The staff around you are your eyes and ears. Get to know your clinical and nonclinical staff’s tendencies. This can be immensely helpful in picking up any different patterns when interviewing and observing patients.
Know your limits. You may not be able to solve everything or obtain the ideal collateral at night. Don’t get caught up in definitively trying to resolve things and end up wasting precious time at night. Let it go. Don’t overthink. If all else fails, hold the patient overnight.
Prioritize self-care. Night shift work has been shown to negatively impact one’s health.1-3 If you choose this type of work, either part-time or full-time, maintain your own health by exercising regularly, eating a healthy diet, obtaining adequate rest between shifts, and seeing your health care team often.
1. Wu QJ, Sun H, Wen ZY, et al. Shift work and health outcomes: an umbrella review of systematic reviews and meta-analyses of epidemiological studies. J Clin Sleep Med. 2022;18(2):653-662. doi:10.5664/jcsm.9642
2. Kecklund G, Axelsson J. Health consequences of shift work and insufficient sleep. BMJ. 2016;355:i5210. doi:10.1136/bmj.i5210
3. Boivin DB, Boudreau P. Impacts of shift work on sleep and circadian rhythms. Pathol Biol (Paris). 2014;62(5):292-301. doi:10.1016/j.patbio.2014.08.001
1. Wu QJ, Sun H, Wen ZY, et al. Shift work and health outcomes: an umbrella review of systematic reviews and meta-analyses of epidemiological studies. J Clin Sleep Med. 2022;18(2):653-662. doi:10.5664/jcsm.9642
2. Kecklund G, Axelsson J. Health consequences of shift work and insufficient sleep. BMJ. 2016;355:i5210. doi:10.1136/bmj.i5210
3. Boivin DB, Boudreau P. Impacts of shift work on sleep and circadian rhythms. Pathol Biol (Paris). 2014;62(5):292-301. doi:10.1016/j.patbio.2014.08.001