User login
Formerly Skin & Allergy News
ass lick
assault rifle
balls
ballsac
black jack
bleach
Boko Haram
bondage
causas
cheap
child abuse
cocaine
compulsive behaviors
cost of miracles
cunt
Daech
display network stats
drug paraphernalia
explosion
fart
fda and death
fda AND warn
fda AND warning
fda AND warns
feom
fuck
gambling
gfc
gun
human trafficking
humira AND expensive
illegal
ISIL
ISIS
Islamic caliphate
Islamic state
madvocate
masturbation
mixed martial arts
MMA
molestation
national rifle association
NRA
nsfw
nuccitelli
pedophile
pedophilia
poker
porn
porn
pornography
psychedelic drug
recreational drug
sex slave rings
shit
slot machine
snort
substance abuse
terrorism
terrorist
texarkana
Texas hold 'em
UFC
section[contains(@class, 'nav-hidden')]
section[contains(@class, 'nav-hidden active')]
The leading independent newspaper covering dermatology news and commentary.
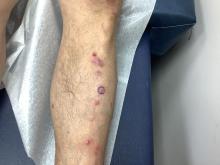
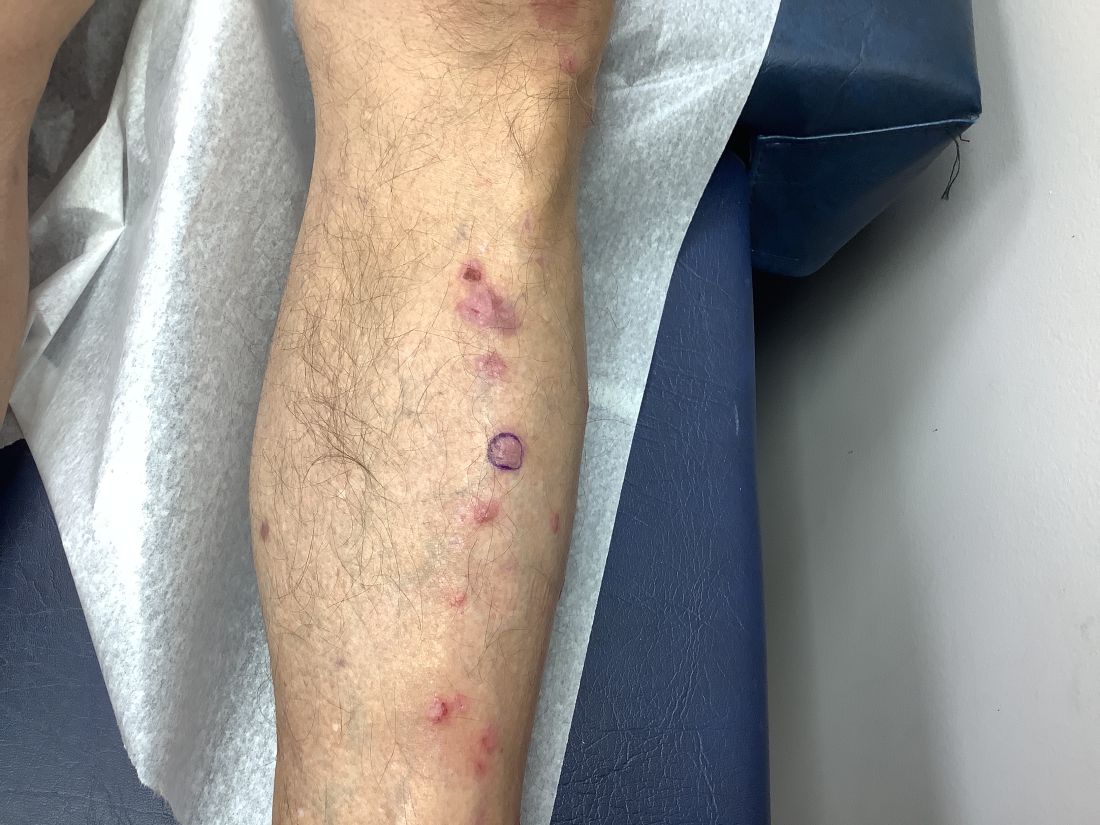
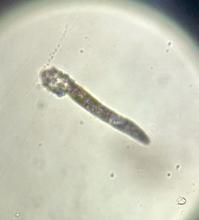
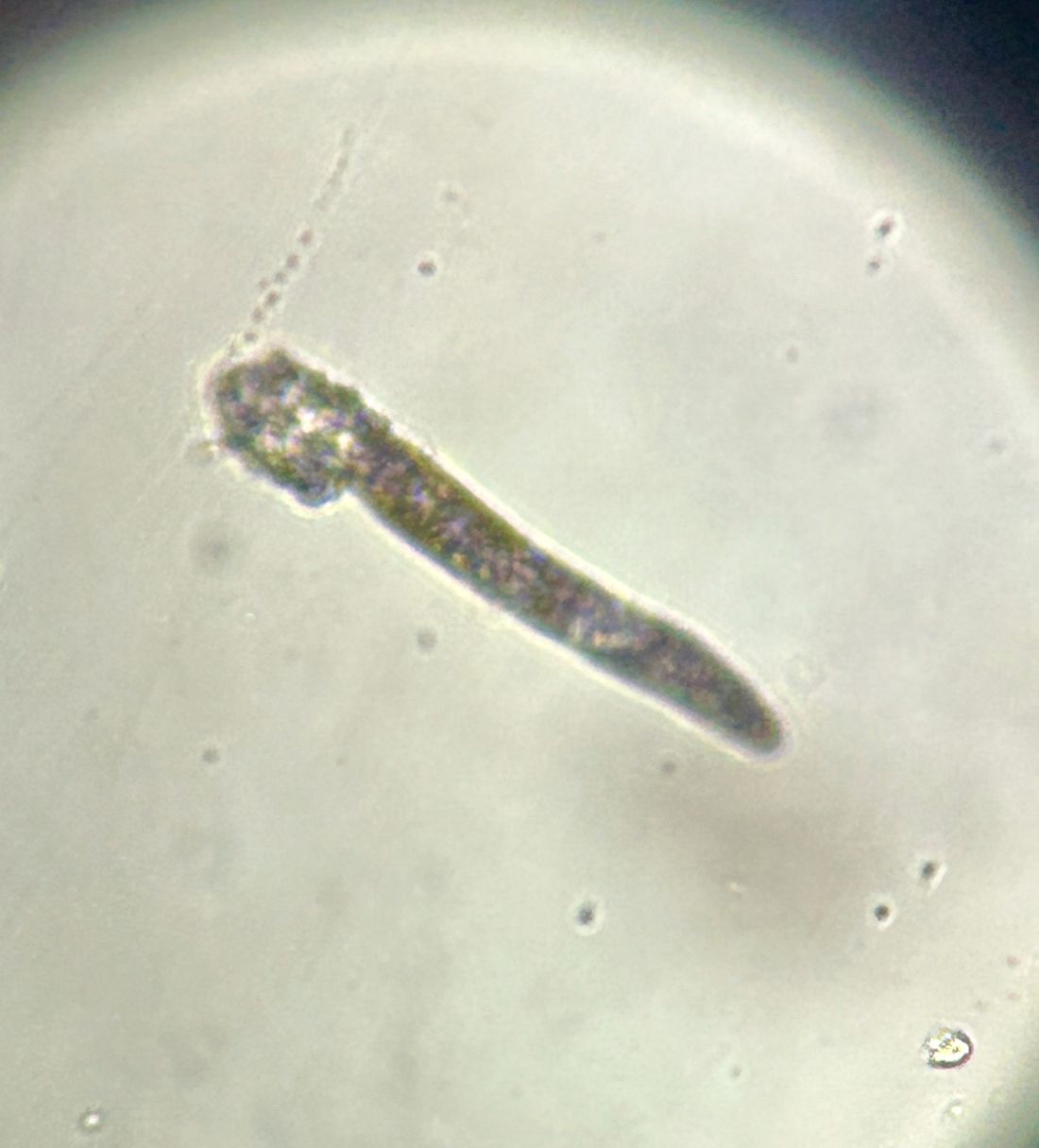
Pruritic, violaceous papules in a patient with renal cell carcinoma
Pembrolizumab (Keytruda) is a programmed cell death protein 1 (PD-1) blocking antibody used to treat different malignancies including melanoma, non–small cell lung cancer, and other advanced solid tumors and hematologic malignancies. and drug rash with eosinophilia and systemic symptoms (DRESS).
Lichen planus-like adverse drug reactions, as seen in this patient, are also referred to as lichenoid drug eruption or drug-induced lichen planus. This cutaneous reaction is one of the more rare side effects of pembrolizumab. It should be noted that in lichenoid reactions, keratinocytes expressing PD-L1 are particularly affected, leading to a dense CD4/CD8 positive lymphocytic infiltration in the basal layer, necrosis of keratinocytes, acanthosis, and hypergranulosis. Subsequently, the cutaneous adverse reaction is a target effect of the PD-1/PD-L1 pathway and not a general hypersensitivity reaction. Clinically, both lichen planus and lichenoid drug eruptions exhibit erythematous papules and plaques. Lichenoid drug eruptions, however, can be scaly, pruritic, and heal with more hyperpigmentation.
A skin biopsy revealed irregular epidermal hyperplasia with jagged rete ridges. Within the dermis, there was a lichenoid inflammatory cell infiltrate obscuring the dermal-epidermal junction. The inflammatory cell infiltrate contained lymphocytes, histiocytes, and eosinophils. A diagnosis of a lichen planus-like adverse drug reaction to pembrolizumab was favored.
If the reaction is mild, topical corticosteroids and oral antihistamines can help with the drug-induced lichen planus. For more severe cases, systemic steroids can be given to help ease the reaction. Physicians should be aware of potential adverse drug effects that can mimic other medical conditions.
The case and photo were submitted by Ms. Towe, Nova Southeastern University College of Osteopathic Medicine, Davie, Florida, and Dr. Berke, Three Rivers Dermatology, Coraopolis, Pennsylvania. The column was edited by Donna Bilu Martin, MD.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
Bansal A et al. Indian Dermatol Online J. 2023 Apr 4;14(3):391-4. doi: 10.4103/idoj.idoj_377_22.
Sethi A, Raj M. Cureus. 2021 Mar 8;13(3):e13768. doi: 10.7759/cureus.13768.
Pembrolizumab (Keytruda) is a programmed cell death protein 1 (PD-1) blocking antibody used to treat different malignancies including melanoma, non–small cell lung cancer, and other advanced solid tumors and hematologic malignancies. and drug rash with eosinophilia and systemic symptoms (DRESS).
Lichen planus-like adverse drug reactions, as seen in this patient, are also referred to as lichenoid drug eruption or drug-induced lichen planus. This cutaneous reaction is one of the more rare side effects of pembrolizumab. It should be noted that in lichenoid reactions, keratinocytes expressing PD-L1 are particularly affected, leading to a dense CD4/CD8 positive lymphocytic infiltration in the basal layer, necrosis of keratinocytes, acanthosis, and hypergranulosis. Subsequently, the cutaneous adverse reaction is a target effect of the PD-1/PD-L1 pathway and not a general hypersensitivity reaction. Clinically, both lichen planus and lichenoid drug eruptions exhibit erythematous papules and plaques. Lichenoid drug eruptions, however, can be scaly, pruritic, and heal with more hyperpigmentation.
A skin biopsy revealed irregular epidermal hyperplasia with jagged rete ridges. Within the dermis, there was a lichenoid inflammatory cell infiltrate obscuring the dermal-epidermal junction. The inflammatory cell infiltrate contained lymphocytes, histiocytes, and eosinophils. A diagnosis of a lichen planus-like adverse drug reaction to pembrolizumab was favored.
If the reaction is mild, topical corticosteroids and oral antihistamines can help with the drug-induced lichen planus. For more severe cases, systemic steroids can be given to help ease the reaction. Physicians should be aware of potential adverse drug effects that can mimic other medical conditions.
The case and photo were submitted by Ms. Towe, Nova Southeastern University College of Osteopathic Medicine, Davie, Florida, and Dr. Berke, Three Rivers Dermatology, Coraopolis, Pennsylvania. The column was edited by Donna Bilu Martin, MD.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
Bansal A et al. Indian Dermatol Online J. 2023 Apr 4;14(3):391-4. doi: 10.4103/idoj.idoj_377_22.
Sethi A, Raj M. Cureus. 2021 Mar 8;13(3):e13768. doi: 10.7759/cureus.13768.
Pembrolizumab (Keytruda) is a programmed cell death protein 1 (PD-1) blocking antibody used to treat different malignancies including melanoma, non–small cell lung cancer, and other advanced solid tumors and hematologic malignancies. and drug rash with eosinophilia and systemic symptoms (DRESS).
Lichen planus-like adverse drug reactions, as seen in this patient, are also referred to as lichenoid drug eruption or drug-induced lichen planus. This cutaneous reaction is one of the more rare side effects of pembrolizumab. It should be noted that in lichenoid reactions, keratinocytes expressing PD-L1 are particularly affected, leading to a dense CD4/CD8 positive lymphocytic infiltration in the basal layer, necrosis of keratinocytes, acanthosis, and hypergranulosis. Subsequently, the cutaneous adverse reaction is a target effect of the PD-1/PD-L1 pathway and not a general hypersensitivity reaction. Clinically, both lichen planus and lichenoid drug eruptions exhibit erythematous papules and plaques. Lichenoid drug eruptions, however, can be scaly, pruritic, and heal with more hyperpigmentation.
A skin biopsy revealed irregular epidermal hyperplasia with jagged rete ridges. Within the dermis, there was a lichenoid inflammatory cell infiltrate obscuring the dermal-epidermal junction. The inflammatory cell infiltrate contained lymphocytes, histiocytes, and eosinophils. A diagnosis of a lichen planus-like adverse drug reaction to pembrolizumab was favored.
If the reaction is mild, topical corticosteroids and oral antihistamines can help with the drug-induced lichen planus. For more severe cases, systemic steroids can be given to help ease the reaction. Physicians should be aware of potential adverse drug effects that can mimic other medical conditions.
The case and photo were submitted by Ms. Towe, Nova Southeastern University College of Osteopathic Medicine, Davie, Florida, and Dr. Berke, Three Rivers Dermatology, Coraopolis, Pennsylvania. The column was edited by Donna Bilu Martin, MD.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
Bansal A et al. Indian Dermatol Online J. 2023 Apr 4;14(3):391-4. doi: 10.4103/idoj.idoj_377_22.
Sethi A, Raj M. Cureus. 2021 Mar 8;13(3):e13768. doi: 10.7759/cureus.13768.
Doctors Endorsing Products on X May Not Disclose Company Ties
Lead author Aaron Mitchell, MD, MPH, a medical oncologist at Memorial Sloan Kettering Cancer Center in New York City, told this news organization that he and his colleagues undertook the study in part to see whether physicians were adhering to professional and industry guidelines regarding marketing communications.
The team reviewed posts by physicians on X during 2022, looking for key words that might indicate that the posts were intended as endorsements of a product. The researchers then delved into the Centers for Medicare and Medicaid Services Open Payments database to see how many of those identified as having endorsed a product were paid by the manufacturers.
What Dr. Mitchell found concerned him, he said.
Overall, the researchers identified 28 physician endorsers who received a total of $1.4 million from sponsors in 2022. Among these, 26 physicians (93%) received payments from the product’s manufacturer, totaling $713,976, and 24 physicians (86%) accepted payments related to the endorsed drug or device, totaling $492,098.
While most did disclose that the posts were sponsored — by adding the word “sponsored” or using #sponsored — nine physicians did not.
Although 28 physician endorsers represent a “small fraction” of the overall number of physicians who use X, each endorsement was ultimately posted dozens, if not hundreds of times, said Dr. Mitchell. In fact, he said he saw the same particular endorsement post every time he opened his X app for months.
Overall, Dr. Mitchell noted that it’s less about the fact that the endorsements are occurring on social media and more that there are these paid endorsements taking place at all.
Among the physician specialties promoting a product, urologists and oncologists dominated. Almost one third were urologists, and 57% were oncologists — six medical oncologists, six radiation oncologists, and four gynecologic oncologists. Of the remaining three physicians, two were internists and one was a pulmonary and critical care medicine specialist.
The authors tracked posts from physicians and industry accounts. Many of the posts on industry accounts were physician testimonials, usually videos. Almost half — 8 of 17 — of those testimonials did not disclose that the doctor was being paid by the manufacturer. In another case, a physician did not disclose that they were paid to endorse a white paper.
Fifteen promotional posts were for a Boston Scientific product, followed by six for GlaxoSmithKline, two for Eisai, two for Exelixis, and one each for AstraZeneca, Novartis, and Pfizer.
In general, Dr. Mitchell said, industry guidelines suggest that manufacturer-paid speakers or consultants should have well-regarded expertise in the area they are being asked to weigh in on, but most physician endorsers in the study were not key opinion leaders or experts.
The authors examined the paid endorsers’ H-index — a measure of academic productivity provided by Scopus. Overall, 19 of the 28 physicians had an H-index below 20, which is considered less accomplished, and 14 had no published research related to the endorsed product.
Ten received payments from manufacturers for research purposes, and only one received research payments related to the endorsed product ($224,577).
“Physicians’ participation in industry marketing raises questions regarding professionalism and their responsibilities as patient advocates,” the JAMA authors wrote.
The study was supported by grants from the National Cancer Institute. Dr. Mitchell reported no relevant financial relationships. Coauthors Samer Al Hadidi, MD, reported receiving personal fees from Pfizer, Sanofi, and Janssen during the conduct of the study, and Timothy S. Anderson, MD, reported receiving grants from the National Institute on Aging, the American Heart Association, and the American College of Cardiology, and receiving consulting fees from the American Medical Student Association. Dr. Anderson is also an associate editor of JAMA Internal Medicine.
A version of this article appeared on Medscape.com.
Lead author Aaron Mitchell, MD, MPH, a medical oncologist at Memorial Sloan Kettering Cancer Center in New York City, told this news organization that he and his colleagues undertook the study in part to see whether physicians were adhering to professional and industry guidelines regarding marketing communications.
The team reviewed posts by physicians on X during 2022, looking for key words that might indicate that the posts were intended as endorsements of a product. The researchers then delved into the Centers for Medicare and Medicaid Services Open Payments database to see how many of those identified as having endorsed a product were paid by the manufacturers.
What Dr. Mitchell found concerned him, he said.
Overall, the researchers identified 28 physician endorsers who received a total of $1.4 million from sponsors in 2022. Among these, 26 physicians (93%) received payments from the product’s manufacturer, totaling $713,976, and 24 physicians (86%) accepted payments related to the endorsed drug or device, totaling $492,098.
While most did disclose that the posts were sponsored — by adding the word “sponsored” or using #sponsored — nine physicians did not.
Although 28 physician endorsers represent a “small fraction” of the overall number of physicians who use X, each endorsement was ultimately posted dozens, if not hundreds of times, said Dr. Mitchell. In fact, he said he saw the same particular endorsement post every time he opened his X app for months.
Overall, Dr. Mitchell noted that it’s less about the fact that the endorsements are occurring on social media and more that there are these paid endorsements taking place at all.
Among the physician specialties promoting a product, urologists and oncologists dominated. Almost one third were urologists, and 57% were oncologists — six medical oncologists, six radiation oncologists, and four gynecologic oncologists. Of the remaining three physicians, two were internists and one was a pulmonary and critical care medicine specialist.
The authors tracked posts from physicians and industry accounts. Many of the posts on industry accounts were physician testimonials, usually videos. Almost half — 8 of 17 — of those testimonials did not disclose that the doctor was being paid by the manufacturer. In another case, a physician did not disclose that they were paid to endorse a white paper.
Fifteen promotional posts were for a Boston Scientific product, followed by six for GlaxoSmithKline, two for Eisai, two for Exelixis, and one each for AstraZeneca, Novartis, and Pfizer.
In general, Dr. Mitchell said, industry guidelines suggest that manufacturer-paid speakers or consultants should have well-regarded expertise in the area they are being asked to weigh in on, but most physician endorsers in the study were not key opinion leaders or experts.
The authors examined the paid endorsers’ H-index — a measure of academic productivity provided by Scopus. Overall, 19 of the 28 physicians had an H-index below 20, which is considered less accomplished, and 14 had no published research related to the endorsed product.
Ten received payments from manufacturers for research purposes, and only one received research payments related to the endorsed product ($224,577).
“Physicians’ participation in industry marketing raises questions regarding professionalism and their responsibilities as patient advocates,” the JAMA authors wrote.
The study was supported by grants from the National Cancer Institute. Dr. Mitchell reported no relevant financial relationships. Coauthors Samer Al Hadidi, MD, reported receiving personal fees from Pfizer, Sanofi, and Janssen during the conduct of the study, and Timothy S. Anderson, MD, reported receiving grants from the National Institute on Aging, the American Heart Association, and the American College of Cardiology, and receiving consulting fees from the American Medical Student Association. Dr. Anderson is also an associate editor of JAMA Internal Medicine.
A version of this article appeared on Medscape.com.
Lead author Aaron Mitchell, MD, MPH, a medical oncologist at Memorial Sloan Kettering Cancer Center in New York City, told this news organization that he and his colleagues undertook the study in part to see whether physicians were adhering to professional and industry guidelines regarding marketing communications.
The team reviewed posts by physicians on X during 2022, looking for key words that might indicate that the posts were intended as endorsements of a product. The researchers then delved into the Centers for Medicare and Medicaid Services Open Payments database to see how many of those identified as having endorsed a product were paid by the manufacturers.
What Dr. Mitchell found concerned him, he said.
Overall, the researchers identified 28 physician endorsers who received a total of $1.4 million from sponsors in 2022. Among these, 26 physicians (93%) received payments from the product’s manufacturer, totaling $713,976, and 24 physicians (86%) accepted payments related to the endorsed drug or device, totaling $492,098.
While most did disclose that the posts were sponsored — by adding the word “sponsored” or using #sponsored — nine physicians did not.
Although 28 physician endorsers represent a “small fraction” of the overall number of physicians who use X, each endorsement was ultimately posted dozens, if not hundreds of times, said Dr. Mitchell. In fact, he said he saw the same particular endorsement post every time he opened his X app for months.
Overall, Dr. Mitchell noted that it’s less about the fact that the endorsements are occurring on social media and more that there are these paid endorsements taking place at all.
Among the physician specialties promoting a product, urologists and oncologists dominated. Almost one third were urologists, and 57% were oncologists — six medical oncologists, six radiation oncologists, and four gynecologic oncologists. Of the remaining three physicians, two were internists and one was a pulmonary and critical care medicine specialist.
The authors tracked posts from physicians and industry accounts. Many of the posts on industry accounts were physician testimonials, usually videos. Almost half — 8 of 17 — of those testimonials did not disclose that the doctor was being paid by the manufacturer. In another case, a physician did not disclose that they were paid to endorse a white paper.
Fifteen promotional posts were for a Boston Scientific product, followed by six for GlaxoSmithKline, two for Eisai, two for Exelixis, and one each for AstraZeneca, Novartis, and Pfizer.
In general, Dr. Mitchell said, industry guidelines suggest that manufacturer-paid speakers or consultants should have well-regarded expertise in the area they are being asked to weigh in on, but most physician endorsers in the study were not key opinion leaders or experts.
The authors examined the paid endorsers’ H-index — a measure of academic productivity provided by Scopus. Overall, 19 of the 28 physicians had an H-index below 20, which is considered less accomplished, and 14 had no published research related to the endorsed product.
Ten received payments from manufacturers for research purposes, and only one received research payments related to the endorsed product ($224,577).
“Physicians’ participation in industry marketing raises questions regarding professionalism and their responsibilities as patient advocates,” the JAMA authors wrote.
The study was supported by grants from the National Cancer Institute. Dr. Mitchell reported no relevant financial relationships. Coauthors Samer Al Hadidi, MD, reported receiving personal fees from Pfizer, Sanofi, and Janssen during the conduct of the study, and Timothy S. Anderson, MD, reported receiving grants from the National Institute on Aging, the American Heart Association, and the American College of Cardiology, and receiving consulting fees from the American Medical Student Association. Dr. Anderson is also an associate editor of JAMA Internal Medicine.
A version of this article appeared on Medscape.com.
One Patient Changed This Oncologist’s View of Hope. Here’s How.
CHICAGO — Carlos, a 21-year-old, lay in a hospital bed, barely clinging to life. Following a stem cell transplant for leukemia, Carlos had developed a life-threatening case of graft-vs-host disease.
But Carlos’ mother had faith.
“I have hope things will get better,” she said, via interpreter, to Richard Leiter, MD, a palliative care doctor in training at that time.
“I hope they will,” Dr. Leiter told her.
“I should have stopped there,” said Dr. Leiter, recounting an early-career lesson on hope during the ASCO Voices session at the American Society of Clinical Oncology annual meeting. “But in my eagerness to show my attending and myself that I could handle this conversation, I kept going, mistakenly.”
“But none of us think they will,” Dr. Leiter continued.
Carlos’ mother looked Dr. Leiter in the eye. “You want him to die,” she said.
“I knew, even then, that she was right,” recalled Dr. Leiter, now a palliative care physician at Dana-Farber Cancer Institute and Brigham and Women’s Hospital and an assistant professor of medicine at Harvard Medical School, Boston.
Although there was nothing he could do to save Carlos, Dr. Leiter also couldn’t sit with the extreme suffering. “The pain was too great,” Dr. Leiter said. “I needed her to adopt our narrative that we had done everything we could to help him live, and now, we would do everything we could to help his death be a comfortable one.”
But looking back, Dr. Leiter realized, “How could we have asked her to accept what was fundamentally unacceptable, to comprehend the incomprehensible?”
The Importance of Hope
Alan B. Astrow, MD, said during an ASCO symposium on “The Art and Science of Hope.”
“How we think about hope directly influences patient care,” said Dr. Astrow, chief of hematology and medical oncology at NewYork-Presbyterian Brooklyn Methodist Hospital and a professor of clinical medicine at Weill Cornell Medicine in New York City.
Hope, whatever it turns out to be neurobiologically, is “very much a gift” that underlies human existence, he said.
Physicians have the capacity to restore or shatter a patient’s hopes, and those who come to understand the importance of hope will wish to extend the gift to others, Dr. Astrow said.
Asking patients about their hopes is the “golden question,” Steven Z. Pantilat, MD, said at the symposium. “When you think about the future, what do you hope for?”
Often, the answers reveal not only “things beyond a cure that matter tremendously to the patient but things that we can help with,” said Dr. Pantilat, professor and chief of the Division of Palliative Medicine at the University of California San Francisco.
Dr. Pantilat recalled a patient with advanced pancreatic cancer who wished to see her daughter’s wedding in 10 months. He knew that was unlikely, but the discussion led to another solution.
Her daughter moved the wedding to the ICU.
Hope can persist and uplift even in the darkest of times, and “as clinicians, we need to be in the true hope business,” he said.
While some patients may wish for a cure, others may want more time with family or comfort in the face of suffering. People can “hope for all the things that can still be, despite the fact that there’s a lot of things that can’t,” he said.
However, fear that a patient will hope for a cure, and that the difficult discussions to follow might destroy hope or lead to false hope, sometimes means physicians won’t begin the conversation.
“We want to be honest with our patients — compassionate and kind, but honest — when we talk about their hopes,” Dr. Pantilat explained. Sometimes that means he needs to tell patients, “I wish that could happen. I wish I had a treatment that could make your cancer go away, but unfortunately, I don’t. So let’s think about what else we can do to help you.”
Having these difficult discussions matters. The evidence, although limited, indicates that feeling hopeful can improve patients’ well-being and may even boost their cancer outcomes.
One recent study found, for instance, that patients who reported feeling more hopeful also had lower levels of depression and anxiety. Early research also suggests that greater levels of hope may have a hand in reducing inflammation in patients with ovarian cancer and could even improve survival in some patients with advanced cancer.
For Dr. Leiter, while these lessons came early in his career as a palliative care physician, they persist and influence his practice today.
“I know that I could not have prevented Carlos’ death. None of us could have, and none of us could have protected his mother from the unimaginable grief that will stay with her for the rest of her life,” he said. “But I could have made things just a little bit less difficult for her.
“I could have acted as her guide rather than her cross-examiner,” he continued, explaining that he now sees hope as “a generous collaborator” that can coexist with rising creatinine levels, failing livers, and fears about intubation.
“As clinicians, we can always find space to hope with our patients and their families,” he said. “So now, years later when I sit with a terrified and grieving family and they tell me they hope their loved one gets better, I remember Carlos’ mother’s eyes piercing mine ... and I know how to respond: ‘I hope so, too.’ And I do.”
A version of this article appeared on Medscape.com.
CHICAGO — Carlos, a 21-year-old, lay in a hospital bed, barely clinging to life. Following a stem cell transplant for leukemia, Carlos had developed a life-threatening case of graft-vs-host disease.
But Carlos’ mother had faith.
“I have hope things will get better,” she said, via interpreter, to Richard Leiter, MD, a palliative care doctor in training at that time.
“I hope they will,” Dr. Leiter told her.
“I should have stopped there,” said Dr. Leiter, recounting an early-career lesson on hope during the ASCO Voices session at the American Society of Clinical Oncology annual meeting. “But in my eagerness to show my attending and myself that I could handle this conversation, I kept going, mistakenly.”
“But none of us think they will,” Dr. Leiter continued.
Carlos’ mother looked Dr. Leiter in the eye. “You want him to die,” she said.
“I knew, even then, that she was right,” recalled Dr. Leiter, now a palliative care physician at Dana-Farber Cancer Institute and Brigham and Women’s Hospital and an assistant professor of medicine at Harvard Medical School, Boston.
Although there was nothing he could do to save Carlos, Dr. Leiter also couldn’t sit with the extreme suffering. “The pain was too great,” Dr. Leiter said. “I needed her to adopt our narrative that we had done everything we could to help him live, and now, we would do everything we could to help his death be a comfortable one.”
But looking back, Dr. Leiter realized, “How could we have asked her to accept what was fundamentally unacceptable, to comprehend the incomprehensible?”
The Importance of Hope
Alan B. Astrow, MD, said during an ASCO symposium on “The Art and Science of Hope.”
“How we think about hope directly influences patient care,” said Dr. Astrow, chief of hematology and medical oncology at NewYork-Presbyterian Brooklyn Methodist Hospital and a professor of clinical medicine at Weill Cornell Medicine in New York City.
Hope, whatever it turns out to be neurobiologically, is “very much a gift” that underlies human existence, he said.
Physicians have the capacity to restore or shatter a patient’s hopes, and those who come to understand the importance of hope will wish to extend the gift to others, Dr. Astrow said.
Asking patients about their hopes is the “golden question,” Steven Z. Pantilat, MD, said at the symposium. “When you think about the future, what do you hope for?”
Often, the answers reveal not only “things beyond a cure that matter tremendously to the patient but things that we can help with,” said Dr. Pantilat, professor and chief of the Division of Palliative Medicine at the University of California San Francisco.
Dr. Pantilat recalled a patient with advanced pancreatic cancer who wished to see her daughter’s wedding in 10 months. He knew that was unlikely, but the discussion led to another solution.
Her daughter moved the wedding to the ICU.
Hope can persist and uplift even in the darkest of times, and “as clinicians, we need to be in the true hope business,” he said.
While some patients may wish for a cure, others may want more time with family or comfort in the face of suffering. People can “hope for all the things that can still be, despite the fact that there’s a lot of things that can’t,” he said.
However, fear that a patient will hope for a cure, and that the difficult discussions to follow might destroy hope or lead to false hope, sometimes means physicians won’t begin the conversation.
“We want to be honest with our patients — compassionate and kind, but honest — when we talk about their hopes,” Dr. Pantilat explained. Sometimes that means he needs to tell patients, “I wish that could happen. I wish I had a treatment that could make your cancer go away, but unfortunately, I don’t. So let’s think about what else we can do to help you.”
Having these difficult discussions matters. The evidence, although limited, indicates that feeling hopeful can improve patients’ well-being and may even boost their cancer outcomes.
One recent study found, for instance, that patients who reported feeling more hopeful also had lower levels of depression and anxiety. Early research also suggests that greater levels of hope may have a hand in reducing inflammation in patients with ovarian cancer and could even improve survival in some patients with advanced cancer.
For Dr. Leiter, while these lessons came early in his career as a palliative care physician, they persist and influence his practice today.
“I know that I could not have prevented Carlos’ death. None of us could have, and none of us could have protected his mother from the unimaginable grief that will stay with her for the rest of her life,” he said. “But I could have made things just a little bit less difficult for her.
“I could have acted as her guide rather than her cross-examiner,” he continued, explaining that he now sees hope as “a generous collaborator” that can coexist with rising creatinine levels, failing livers, and fears about intubation.
“As clinicians, we can always find space to hope with our patients and their families,” he said. “So now, years later when I sit with a terrified and grieving family and they tell me they hope their loved one gets better, I remember Carlos’ mother’s eyes piercing mine ... and I know how to respond: ‘I hope so, too.’ And I do.”
A version of this article appeared on Medscape.com.
CHICAGO — Carlos, a 21-year-old, lay in a hospital bed, barely clinging to life. Following a stem cell transplant for leukemia, Carlos had developed a life-threatening case of graft-vs-host disease.
But Carlos’ mother had faith.
“I have hope things will get better,” she said, via interpreter, to Richard Leiter, MD, a palliative care doctor in training at that time.
“I hope they will,” Dr. Leiter told her.
“I should have stopped there,” said Dr. Leiter, recounting an early-career lesson on hope during the ASCO Voices session at the American Society of Clinical Oncology annual meeting. “But in my eagerness to show my attending and myself that I could handle this conversation, I kept going, mistakenly.”
“But none of us think they will,” Dr. Leiter continued.
Carlos’ mother looked Dr. Leiter in the eye. “You want him to die,” she said.
“I knew, even then, that she was right,” recalled Dr. Leiter, now a palliative care physician at Dana-Farber Cancer Institute and Brigham and Women’s Hospital and an assistant professor of medicine at Harvard Medical School, Boston.
Although there was nothing he could do to save Carlos, Dr. Leiter also couldn’t sit with the extreme suffering. “The pain was too great,” Dr. Leiter said. “I needed her to adopt our narrative that we had done everything we could to help him live, and now, we would do everything we could to help his death be a comfortable one.”
But looking back, Dr. Leiter realized, “How could we have asked her to accept what was fundamentally unacceptable, to comprehend the incomprehensible?”
The Importance of Hope
Alan B. Astrow, MD, said during an ASCO symposium on “The Art and Science of Hope.”
“How we think about hope directly influences patient care,” said Dr. Astrow, chief of hematology and medical oncology at NewYork-Presbyterian Brooklyn Methodist Hospital and a professor of clinical medicine at Weill Cornell Medicine in New York City.
Hope, whatever it turns out to be neurobiologically, is “very much a gift” that underlies human existence, he said.
Physicians have the capacity to restore or shatter a patient’s hopes, and those who come to understand the importance of hope will wish to extend the gift to others, Dr. Astrow said.
Asking patients about their hopes is the “golden question,” Steven Z. Pantilat, MD, said at the symposium. “When you think about the future, what do you hope for?”
Often, the answers reveal not only “things beyond a cure that matter tremendously to the patient but things that we can help with,” said Dr. Pantilat, professor and chief of the Division of Palliative Medicine at the University of California San Francisco.
Dr. Pantilat recalled a patient with advanced pancreatic cancer who wished to see her daughter’s wedding in 10 months. He knew that was unlikely, but the discussion led to another solution.
Her daughter moved the wedding to the ICU.
Hope can persist and uplift even in the darkest of times, and “as clinicians, we need to be in the true hope business,” he said.
While some patients may wish for a cure, others may want more time with family or comfort in the face of suffering. People can “hope for all the things that can still be, despite the fact that there’s a lot of things that can’t,” he said.
However, fear that a patient will hope for a cure, and that the difficult discussions to follow might destroy hope or lead to false hope, sometimes means physicians won’t begin the conversation.
“We want to be honest with our patients — compassionate and kind, but honest — when we talk about their hopes,” Dr. Pantilat explained. Sometimes that means he needs to tell patients, “I wish that could happen. I wish I had a treatment that could make your cancer go away, but unfortunately, I don’t. So let’s think about what else we can do to help you.”
Having these difficult discussions matters. The evidence, although limited, indicates that feeling hopeful can improve patients’ well-being and may even boost their cancer outcomes.
One recent study found, for instance, that patients who reported feeling more hopeful also had lower levels of depression and anxiety. Early research also suggests that greater levels of hope may have a hand in reducing inflammation in patients with ovarian cancer and could even improve survival in some patients with advanced cancer.
For Dr. Leiter, while these lessons came early in his career as a palliative care physician, they persist and influence his practice today.
“I know that I could not have prevented Carlos’ death. None of us could have, and none of us could have protected his mother from the unimaginable grief that will stay with her for the rest of her life,” he said. “But I could have made things just a little bit less difficult for her.
“I could have acted as her guide rather than her cross-examiner,” he continued, explaining that he now sees hope as “a generous collaborator” that can coexist with rising creatinine levels, failing livers, and fears about intubation.
“As clinicians, we can always find space to hope with our patients and their families,” he said. “So now, years later when I sit with a terrified and grieving family and they tell me they hope their loved one gets better, I remember Carlos’ mother’s eyes piercing mine ... and I know how to respond: ‘I hope so, too.’ And I do.”
A version of this article appeared on Medscape.com.
FROM ASCO 2024
Photoprotection: Benefits of Sunscreens With Iron Oxide
CHICAGO — One of the more recent developments in sunscreen technology is the addition of iron oxide to mineral sunscreens.
Iron oxide is “an excellent pigment” that absorbs and blocks visible light, which is particularly important in individuals with Fitzpatrick skin types III-VI, Zoe D. Draelos, MD, consulting professor of dermatology at Duke University, Durham, North Carolina, said at the Pigmentation Disorders Exchange symposium.
Susan C. Taylor, MD, professor of dermatology at the University of Pennsylvania, Philadelphia, who spoke at the conference, also recommended tinted sunscreen with iron oxide for patients with skin of color. “It still needs to be broad spectrum,” she said, “and at least an SPF [Sun Protection Factor] 30.”
When blended with mineral sunscreens, iron oxide can reduce transmission of visible light by 90% and can protect patients from hyperpigmentation. Iron oxide comes in different colors blended together for various degrees of tinting.
Dr. Taylor noted that iron oxide is listed under the inactive ingredients. “The literature indicates a 3% concentration to aim for, but we don’t know the concentration in most of the products,” she added.
During her presentation, Dr. Draelos noted that inorganic sunscreens, such as zinc oxide and titanium oxides, are highly effective but make the skin white and pasty. To address this issue, many companies are now grinding these materials into such small particles that they are transparent.
“That’s great, except the smaller the particle is, the less UV [ultraviolet] radiation it reflects and that lowers the [SPF],” she said.
In addition to providing photoprotection, sunscreens in general provide protection from nanoparticles in tobacco and combustion, such as traffic exhaust, which can harm skin over time. “Moisturizers and sunscreens are the best way to protect against pollution and tobacco nanoparticle damage, which can contribute to inflammation,” she noted. They create a film over the skin and trap the nanoparticles.
Start the Patient Visit With a Photoprotection Talk
At the meeting, Dr. Taylor recommended that for all patients with hypopigmentation and hyperpigmentation disorders, “treatment really begins with photoprotection.”
She acknowledged that photoprotection discussions, including the basics of seeking shade, wearing protective clothing, and avoiding midday sun, often come at the end of the patient visit but she urged dermatologists to make that the first topic instead.
Dr. Taylor said a question often asked of patients of color about prolonged sun exposure — whether their skin turns bright red after too much sun — may get a negative reply. The better question is whether the patient has experienced tender skin after too much sun — which can signify a sunburn, she said.
Dr. Draelos reported no relevant financial relationships. Dr. Taylor reported financial relationships and grant support from multiple pharmaceutical companies.
A version of this article first appeared on Medscape.com.
CHICAGO — One of the more recent developments in sunscreen technology is the addition of iron oxide to mineral sunscreens.
Iron oxide is “an excellent pigment” that absorbs and blocks visible light, which is particularly important in individuals with Fitzpatrick skin types III-VI, Zoe D. Draelos, MD, consulting professor of dermatology at Duke University, Durham, North Carolina, said at the Pigmentation Disorders Exchange symposium.
Susan C. Taylor, MD, professor of dermatology at the University of Pennsylvania, Philadelphia, who spoke at the conference, also recommended tinted sunscreen with iron oxide for patients with skin of color. “It still needs to be broad spectrum,” she said, “and at least an SPF [Sun Protection Factor] 30.”
When blended with mineral sunscreens, iron oxide can reduce transmission of visible light by 90% and can protect patients from hyperpigmentation. Iron oxide comes in different colors blended together for various degrees of tinting.
Dr. Taylor noted that iron oxide is listed under the inactive ingredients. “The literature indicates a 3% concentration to aim for, but we don’t know the concentration in most of the products,” she added.
During her presentation, Dr. Draelos noted that inorganic sunscreens, such as zinc oxide and titanium oxides, are highly effective but make the skin white and pasty. To address this issue, many companies are now grinding these materials into such small particles that they are transparent.
“That’s great, except the smaller the particle is, the less UV [ultraviolet] radiation it reflects and that lowers the [SPF],” she said.
In addition to providing photoprotection, sunscreens in general provide protection from nanoparticles in tobacco and combustion, such as traffic exhaust, which can harm skin over time. “Moisturizers and sunscreens are the best way to protect against pollution and tobacco nanoparticle damage, which can contribute to inflammation,” she noted. They create a film over the skin and trap the nanoparticles.
Start the Patient Visit With a Photoprotection Talk
At the meeting, Dr. Taylor recommended that for all patients with hypopigmentation and hyperpigmentation disorders, “treatment really begins with photoprotection.”
She acknowledged that photoprotection discussions, including the basics of seeking shade, wearing protective clothing, and avoiding midday sun, often come at the end of the patient visit but she urged dermatologists to make that the first topic instead.
Dr. Taylor said a question often asked of patients of color about prolonged sun exposure — whether their skin turns bright red after too much sun — may get a negative reply. The better question is whether the patient has experienced tender skin after too much sun — which can signify a sunburn, she said.
Dr. Draelos reported no relevant financial relationships. Dr. Taylor reported financial relationships and grant support from multiple pharmaceutical companies.
A version of this article first appeared on Medscape.com.
CHICAGO — One of the more recent developments in sunscreen technology is the addition of iron oxide to mineral sunscreens.
Iron oxide is “an excellent pigment” that absorbs and blocks visible light, which is particularly important in individuals with Fitzpatrick skin types III-VI, Zoe D. Draelos, MD, consulting professor of dermatology at Duke University, Durham, North Carolina, said at the Pigmentation Disorders Exchange symposium.
Susan C. Taylor, MD, professor of dermatology at the University of Pennsylvania, Philadelphia, who spoke at the conference, also recommended tinted sunscreen with iron oxide for patients with skin of color. “It still needs to be broad spectrum,” she said, “and at least an SPF [Sun Protection Factor] 30.”
When blended with mineral sunscreens, iron oxide can reduce transmission of visible light by 90% and can protect patients from hyperpigmentation. Iron oxide comes in different colors blended together for various degrees of tinting.
Dr. Taylor noted that iron oxide is listed under the inactive ingredients. “The literature indicates a 3% concentration to aim for, but we don’t know the concentration in most of the products,” she added.
During her presentation, Dr. Draelos noted that inorganic sunscreens, such as zinc oxide and titanium oxides, are highly effective but make the skin white and pasty. To address this issue, many companies are now grinding these materials into such small particles that they are transparent.
“That’s great, except the smaller the particle is, the less UV [ultraviolet] radiation it reflects and that lowers the [SPF],” she said.
In addition to providing photoprotection, sunscreens in general provide protection from nanoparticles in tobacco and combustion, such as traffic exhaust, which can harm skin over time. “Moisturizers and sunscreens are the best way to protect against pollution and tobacco nanoparticle damage, which can contribute to inflammation,” she noted. They create a film over the skin and trap the nanoparticles.
Start the Patient Visit With a Photoprotection Talk
At the meeting, Dr. Taylor recommended that for all patients with hypopigmentation and hyperpigmentation disorders, “treatment really begins with photoprotection.”
She acknowledged that photoprotection discussions, including the basics of seeking shade, wearing protective clothing, and avoiding midday sun, often come at the end of the patient visit but she urged dermatologists to make that the first topic instead.
Dr. Taylor said a question often asked of patients of color about prolonged sun exposure — whether their skin turns bright red after too much sun — may get a negative reply. The better question is whether the patient has experienced tender skin after too much sun — which can signify a sunburn, she said.
Dr. Draelos reported no relevant financial relationships. Dr. Taylor reported financial relationships and grant support from multiple pharmaceutical companies.
A version of this article first appeared on Medscape.com.
DEA Training Mandate: 8 Hours of My Life I’d Like Back
It’s time to renew two of my three narcotic prescribing licenses. For the first time in my career, I’ve waffled on whether the financial outlay to the US Drug Enforcement Agency (DEA) is worth it.
At $888 each, I’ve considered letting two licenses lapse because I only work part-time in Montana. But several friends advised me to keep a “spare” in case I transfer to a new location.
I thought about just paying the fees until I could do a little more research, but there is no mechanism for a refund unless I die within the first year of the 3-year cycle, provide incorrect credit card digits, or accidentally duplicate payments.
The renewal fee is just part of the issue.
Mandatory 8-Hour Training
I also received an alert about the requirement for more “narcotics prescribing education” thanks to the Medication Access and Training Expansion Act (MATE).
The requirement seems counterintuitive because opioid prescribing has decreased for the 10th consecutive year, according to the AMA Overdose Epidemic Report. The continuing rise in overdose deaths is largely due to illegitimate manufacturing of synthetic opioids.
I’ve written zero outpatient narcotics prescriptions in the past 6 years, and I’ve written very few in my 33 years of practice. My use is limited to intravenous morphine for flash pulmonary edema or refractory angina, but unless you graduated from a training program within 5 years of the June 2023 mandate or are boarded in addiction medicine, there is no way to escape the 8-hour education requirement.
The problem is that these courses are never just 8 hours in duration. After signing up for one such CME course that cost $150, I was still dying of boredom and at risk for DVT 4 days later. That’s how long it took to sit through.
Instead of the 30 seconds it should have taken to review the simple instructions to deliver Narcan, there were scores of screens followed by juvenile quizlets and cartoons. All but about 2 hours out of the 4 days is now relegated to that category of “hours of my life that I can never get back.” Additionally, none of that mandatory “education” will change my prescribing habits one whit.
And beware the penalty. 
Of course, I would always be truthful when asked to check the box on the DEA renewal application attesting to my having completed the required education. On the outside chance that you plan to check the yes box without completing the relevant courses, those found guilty of such false claims could be fined up to $250,000 and subject to “not more than four years in prison,” or both. Yikes!
Larry Houck, a former DEA investigator, explained that “[t]here are lot of people who are coming up for renewal and log on but still don’t know this is a requirement.” Neither ignorance nor complacency is an acceptable defense.
Changes Needed
The only good thing that came of those 4 long days of opioid education was a motivation to drive change in our current licensing and educational experience. Why not use this opportunity to reform the DEA-physician/prescriber relationship?
The educational requirements should be curtailed for those of us who do not provide outpatient narcotic prescriptions even if we use inpatient opioids. Meds with low abuse potential should be rescheduled to minimize who gets caught in the broad net of the education requirement.
We should reduce overregulation of the legitimate prescribers by lowering, instead of increasing, licensing fees. We should change to a single license number that covers every state. In this digital age, there is no legitimate excuse to prevent this from happening.
After all, the settlements from opioid manufacturers and distributors will in time total $50 billion. It seems that at least some of the responsibilities of the DEA could shift to states, cities, and towns.
My friend Siamak Karimian, MD, who provides locum services in multiple states, pays for seven active DEA licenses every 3 years. He pointed out the hypocrisy in the current regulatory system: “It’s funny that you can have only one DEA or state license and work for the government in all other states or territories with no limits, including the VA, Indian healthcare systems, or prison systems.”
All other prescribers require a separate DEA number for every state. Ultimately, you’d think tracking prescriptions for a single DEA number should be far simpler than tracking someone with seven.
Competent physicians not guilty of criminal overprescribing seem to be the last to be considered in nearly every healthcare endeavor these days. It would be refreshing if they would reduce our fees and prevent this waste of our time.
And while we are at it, perhaps a more fitting punishment is due for Richard Sackler and all the Purdue Pharma–affiliated family members. The Sacklers will pay out $6 billion in exchange for immunity against civil litigation. That doesn’t seem like much when they are worth $11 billion.
Perhaps they should be made to take an 8-hour course on opioid prescribing, annually and in perpetuity. Let’s see them complete a few quizlets and sit through screens of instruction on how to administer Naloxone. Of course, that would be a mild punishment for those who manufactured a drug that killed hundreds of thousands. But it would be a start.
Dr. Walton-Shirley, a clinical cardiologist in Nashville, Tennessee, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
It’s time to renew two of my three narcotic prescribing licenses. For the first time in my career, I’ve waffled on whether the financial outlay to the US Drug Enforcement Agency (DEA) is worth it.
At $888 each, I’ve considered letting two licenses lapse because I only work part-time in Montana. But several friends advised me to keep a “spare” in case I transfer to a new location.
I thought about just paying the fees until I could do a little more research, but there is no mechanism for a refund unless I die within the first year of the 3-year cycle, provide incorrect credit card digits, or accidentally duplicate payments.
The renewal fee is just part of the issue.
Mandatory 8-Hour Training
I also received an alert about the requirement for more “narcotics prescribing education” thanks to the Medication Access and Training Expansion Act (MATE).
The requirement seems counterintuitive because opioid prescribing has decreased for the 10th consecutive year, according to the AMA Overdose Epidemic Report. The continuing rise in overdose deaths is largely due to illegitimate manufacturing of synthetic opioids.
I’ve written zero outpatient narcotics prescriptions in the past 6 years, and I’ve written very few in my 33 years of practice. My use is limited to intravenous morphine for flash pulmonary edema or refractory angina, but unless you graduated from a training program within 5 years of the June 2023 mandate or are boarded in addiction medicine, there is no way to escape the 8-hour education requirement.
The problem is that these courses are never just 8 hours in duration. After signing up for one such CME course that cost $150, I was still dying of boredom and at risk for DVT 4 days later. That’s how long it took to sit through.
Instead of the 30 seconds it should have taken to review the simple instructions to deliver Narcan, there were scores of screens followed by juvenile quizlets and cartoons. All but about 2 hours out of the 4 days is now relegated to that category of “hours of my life that I can never get back.” Additionally, none of that mandatory “education” will change my prescribing habits one whit.
And beware the penalty. 
Of course, I would always be truthful when asked to check the box on the DEA renewal application attesting to my having completed the required education. On the outside chance that you plan to check the yes box without completing the relevant courses, those found guilty of such false claims could be fined up to $250,000 and subject to “not more than four years in prison,” or both. Yikes!
Larry Houck, a former DEA investigator, explained that “[t]here are lot of people who are coming up for renewal and log on but still don’t know this is a requirement.” Neither ignorance nor complacency is an acceptable defense.
Changes Needed
The only good thing that came of those 4 long days of opioid education was a motivation to drive change in our current licensing and educational experience. Why not use this opportunity to reform the DEA-physician/prescriber relationship?
The educational requirements should be curtailed for those of us who do not provide outpatient narcotic prescriptions even if we use inpatient opioids. Meds with low abuse potential should be rescheduled to minimize who gets caught in the broad net of the education requirement.
We should reduce overregulation of the legitimate prescribers by lowering, instead of increasing, licensing fees. We should change to a single license number that covers every state. In this digital age, there is no legitimate excuse to prevent this from happening.
After all, the settlements from opioid manufacturers and distributors will in time total $50 billion. It seems that at least some of the responsibilities of the DEA could shift to states, cities, and towns.
My friend Siamak Karimian, MD, who provides locum services in multiple states, pays for seven active DEA licenses every 3 years. He pointed out the hypocrisy in the current regulatory system: “It’s funny that you can have only one DEA or state license and work for the government in all other states or territories with no limits, including the VA, Indian healthcare systems, or prison systems.”
All other prescribers require a separate DEA number for every state. Ultimately, you’d think tracking prescriptions for a single DEA number should be far simpler than tracking someone with seven.
Competent physicians not guilty of criminal overprescribing seem to be the last to be considered in nearly every healthcare endeavor these days. It would be refreshing if they would reduce our fees and prevent this waste of our time.
And while we are at it, perhaps a more fitting punishment is due for Richard Sackler and all the Purdue Pharma–affiliated family members. The Sacklers will pay out $6 billion in exchange for immunity against civil litigation. That doesn’t seem like much when they are worth $11 billion.
Perhaps they should be made to take an 8-hour course on opioid prescribing, annually and in perpetuity. Let’s see them complete a few quizlets and sit through screens of instruction on how to administer Naloxone. Of course, that would be a mild punishment for those who manufactured a drug that killed hundreds of thousands. But it would be a start.
Dr. Walton-Shirley, a clinical cardiologist in Nashville, Tennessee, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
It’s time to renew two of my three narcotic prescribing licenses. For the first time in my career, I’ve waffled on whether the financial outlay to the US Drug Enforcement Agency (DEA) is worth it.
At $888 each, I’ve considered letting two licenses lapse because I only work part-time in Montana. But several friends advised me to keep a “spare” in case I transfer to a new location.
I thought about just paying the fees until I could do a little more research, but there is no mechanism for a refund unless I die within the first year of the 3-year cycle, provide incorrect credit card digits, or accidentally duplicate payments.
The renewal fee is just part of the issue.
Mandatory 8-Hour Training
I also received an alert about the requirement for more “narcotics prescribing education” thanks to the Medication Access and Training Expansion Act (MATE).
The requirement seems counterintuitive because opioid prescribing has decreased for the 10th consecutive year, according to the AMA Overdose Epidemic Report. The continuing rise in overdose deaths is largely due to illegitimate manufacturing of synthetic opioids.
I’ve written zero outpatient narcotics prescriptions in the past 6 years, and I’ve written very few in my 33 years of practice. My use is limited to intravenous morphine for flash pulmonary edema or refractory angina, but unless you graduated from a training program within 5 years of the June 2023 mandate or are boarded in addiction medicine, there is no way to escape the 8-hour education requirement.
The problem is that these courses are never just 8 hours in duration. After signing up for one such CME course that cost $150, I was still dying of boredom and at risk for DVT 4 days later. That’s how long it took to sit through.
Instead of the 30 seconds it should have taken to review the simple instructions to deliver Narcan, there were scores of screens followed by juvenile quizlets and cartoons. All but about 2 hours out of the 4 days is now relegated to that category of “hours of my life that I can never get back.” Additionally, none of that mandatory “education” will change my prescribing habits one whit.
And beware the penalty. 
Of course, I would always be truthful when asked to check the box on the DEA renewal application attesting to my having completed the required education. On the outside chance that you plan to check the yes box without completing the relevant courses, those found guilty of such false claims could be fined up to $250,000 and subject to “not more than four years in prison,” or both. Yikes!
Larry Houck, a former DEA investigator, explained that “[t]here are lot of people who are coming up for renewal and log on but still don’t know this is a requirement.” Neither ignorance nor complacency is an acceptable defense.
Changes Needed
The only good thing that came of those 4 long days of opioid education was a motivation to drive change in our current licensing and educational experience. Why not use this opportunity to reform the DEA-physician/prescriber relationship?
The educational requirements should be curtailed for those of us who do not provide outpatient narcotic prescriptions even if we use inpatient opioids. Meds with low abuse potential should be rescheduled to minimize who gets caught in the broad net of the education requirement.
We should reduce overregulation of the legitimate prescribers by lowering, instead of increasing, licensing fees. We should change to a single license number that covers every state. In this digital age, there is no legitimate excuse to prevent this from happening.
After all, the settlements from opioid manufacturers and distributors will in time total $50 billion. It seems that at least some of the responsibilities of the DEA could shift to states, cities, and towns.
My friend Siamak Karimian, MD, who provides locum services in multiple states, pays for seven active DEA licenses every 3 years. He pointed out the hypocrisy in the current regulatory system: “It’s funny that you can have only one DEA or state license and work for the government in all other states or territories with no limits, including the VA, Indian healthcare systems, or prison systems.”
All other prescribers require a separate DEA number for every state. Ultimately, you’d think tracking prescriptions for a single DEA number should be far simpler than tracking someone with seven.
Competent physicians not guilty of criminal overprescribing seem to be the last to be considered in nearly every healthcare endeavor these days. It would be refreshing if they would reduce our fees and prevent this waste of our time.
And while we are at it, perhaps a more fitting punishment is due for Richard Sackler and all the Purdue Pharma–affiliated family members. The Sacklers will pay out $6 billion in exchange for immunity against civil litigation. That doesn’t seem like much when they are worth $11 billion.
Perhaps they should be made to take an 8-hour course on opioid prescribing, annually and in perpetuity. Let’s see them complete a few quizlets and sit through screens of instruction on how to administer Naloxone. Of course, that would be a mild punishment for those who manufactured a drug that killed hundreds of thousands. But it would be a start.
Dr. Walton-Shirley, a clinical cardiologist in Nashville, Tennessee, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
What’s in a Name: Defining Difficult-to-Treat axSpA and PsA
Despite an expanding arsenal of disease-modifying antirheumatic drugs (DMARDs), many patients with axial spondyloarthritis (axSpA) and psoriatic arthritis (PsA) still struggle to reach remission even after trying multiple advanced treatments.
Now, international groups of experts are working to better define these “difficult-to-treat” patients to both inform care and improve selection of participants for future clinical trials.
“The idea is rather simple, and the need is relatively ubiquitous,” Denis Poddubnyy, MD, of the Charité – Universitätsmedizin Berlin and the German Rheumatism Research Center Berlin, both in Berlin, Germany, said in an interview. He is the co-primary investigator for the ongoing Assessment of SpondyloArthritis International Society (ASAS) project to develop a consensus definition of difficult-to-treat axSpA.
According to ASAS, only 40%-50% of patients with axSpA achieve a 40% improvement in ASAS response criteria (ASAS40), and few (10%-20%) achieve remission in the first 4-6 months of treatment.
“If you look into current clinical guidelines, you will see that there is no clear guidance,” on how to manage these patients, Dr. Poddubnyy continued. “In other similar recommendations for the treatment of axSpA, the only point which is clearly made with regards to nonresponders to effective anti-inflammatory treatment is to ‘check the diagnosis.’”
Multiple Reasons for Nonresponse
“While the term difficult-to-treat can refer to refractory disease, that is not the only reason why a patient might not be responding to medication. In fact, it’s likely that truly biologically refractory disease makes up only a fraction of cases that respond inadequately to treatment,” said Shikha Singla, MD, who directs the psoriatic arthritis program at the Medical College of Wisconsin in Milwaukee. She is also involved with the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) initiative to define Difficult-to-Treat and Complex-to-Manage PsA.
“Apart from the persistent articular and periarticular inflammation, there could be multiple noninflammatory factors that may be contributing to this treatment-resistant disease, including comorbid conditions such as obesity, cardiovascular disease, fibromyalgia, and even social factors such as limited access to medications,” she told this news organization. “Given these complexities, it is a matter of supreme importance to recognize and carefully delineate the elements that contribute to treatment refractory disease: Is it truly the inflammation, or are there noninflammatory components that are causing the treatment failure, or a combination of the two?”
Other contributing factors could be depression, hypersensitization, and comorbidities that prevent certain treatment approaches, added Fabian Proft, MD, also of Charité – Universitätsmedizin Berlin. Dr. Proft discussed these difficult-to-treat definition efforts at the recent Spondyloarthritis Research and Treatment Network (SPARTAN) annual meeting held in Cleveland. Patients also might not be taking their medication regularly and may be seeking alternative medicine approaches, he said.
“There is a quite clear consensus within the community” that differentiation between these two groups is needed, Dr. Proft said.
The Definitions
Terminology for these two groups can vary by professional society. The European Alliance of Associations for Rheumatology (EULAR) published a definition for “difficult-to-treat” rheumatoid arthritis (RA) that includes cases with “both inflammatory activity and/or noninflammatory complaints.”
The definition includes three criteria:
1) Treatment according to EULAR recommendation and failure of at least two biologic DMARDs (bDMARDs) or targeted synthetic DMARDs (tsDMARDs) (with different mechanisms of action) after failing conventional synthetic DMARD therapy (unless contraindicated)
2) Signs suggestive of active/progressive disease, including at least one of the following:
- Moderate disease activity (according to validated composite measures including joint counts)
- Signs (including acute phase reactants and imaging) and/or symptoms suggestive of active disease, whether joint-related or other
- Inability to taper glucocorticoid treatment
- Rapid radiographic progression (with or without signs of active disease)
- RA symptoms that are causing a reduction in quality of life
3) Symptom/sign management perceived as problematic by the rheumatologist or the patient
All three criteria must be met.
Both GRAPPA and ASAS plan to use the term “difficult-to-treat” or “treatment refractory” to describe true biologically refractory inflammatory disease and are categorizing the larger, heterogeneous group of nonresponders as “difficult-to-manage” (ASAS) or “complex-to-manage” (GRAPPA).
According to Dr. Poddubnyy, the agreed ASAS definition of difficult-to-manage has several similarities with EULAR’s RA definition, including three pillars:
- Treatment according to existing recommendations and failure of at least two different bDMARDs or tsDMARDs with different mechanisms
- Having signs and symptoms of disease (measured by high disease activity by certain disease activity indexes, persistently elevated C-reactive protein, inflammation on MRI, or rapid radiographic spinal progression)
- Symptoms/signs of disease that are considered problematic by the provider or patient
The definition was approved in January, and the manuscript is in the works, Dr. Poddubnyy said.
The GRAPPA project on PsA is still in its early stages, which so far has included a comprehensive literature review as well as a survey of GRAPPA members across 47 countries. The group is generally in agreement that two separate definitions for nonresponse to treatment are necessary, and that the “difficult-to-treat” definition — which identifies true refractory disease — should include objective signs of inflammation, Dr. Singla said.
Looking Forward
The next step of the ASAS project is to “define the pathway” from difficult-to-manage axSpA to treatment refractory disease, Dr. Poddubnyy said.
“What should be ruled out in order to exclude so-called noninflammatory causes of pain?” he continued. “It will require some Delphi exercises and [a] consensus approach.”
Proft anticipates that this treatment refractory definition in both axSpA and PsA will be most useful in research, rather than clinical practice.
“It is really important to have unified definition criteria to shape as homogeneous a cohort as possible,” he said, for future clinical trials in this population.
On the other hand, the complex/difficult-to-manage definition may be more useful for clinical practice, Dr. Proft thought.
“If you see a patient not responding to treatment, the easiest thing you can do would be to change treatment,” like swapping one biologic for another, Dr. Poddubnyy added, “but this would not be the right approach in every patient.” One goal of these initiatives is to give guidance on “what things should be looked after or excluded before you conclude this is biological [nonresponse],” he said.
Dr. Singla consults for AbbVie, Janssen, and UCB and received research funding from Eli Lilly. Dr. Poddubnyy disclosed serving as a speaker, consultant, and/or research grant recipient for multiple companies including AbbVie, Lilly, Merck Sharp and Dohme, Novartis, Pfizer, GlaxoSmithKline, Novartis, and UCB. Dr. Proft reported receiving research grants, consultant fees, or support for attending meetings and/or travel from Amgen, AbbVie, Bristol Myers Squibb, Celgene, Eli Lilly, Janssen, Merck Sharp and Dohme, Novartis, Pfizer, Roche, UCB, Medscape Medical News, Galapagos, and Hexal. Dr. Proft also participants on a data safety monitoring board or advisory board for AbbVie, Celgene, Janssen, Novartis, and UCB.
A version of this article appeared on Medscape.com.
Despite an expanding arsenal of disease-modifying antirheumatic drugs (DMARDs), many patients with axial spondyloarthritis (axSpA) and psoriatic arthritis (PsA) still struggle to reach remission even after trying multiple advanced treatments.
Now, international groups of experts are working to better define these “difficult-to-treat” patients to both inform care and improve selection of participants for future clinical trials.
“The idea is rather simple, and the need is relatively ubiquitous,” Denis Poddubnyy, MD, of the Charité – Universitätsmedizin Berlin and the German Rheumatism Research Center Berlin, both in Berlin, Germany, said in an interview. He is the co-primary investigator for the ongoing Assessment of SpondyloArthritis International Society (ASAS) project to develop a consensus definition of difficult-to-treat axSpA.
According to ASAS, only 40%-50% of patients with axSpA achieve a 40% improvement in ASAS response criteria (ASAS40), and few (10%-20%) achieve remission in the first 4-6 months of treatment.
“If you look into current clinical guidelines, you will see that there is no clear guidance,” on how to manage these patients, Dr. Poddubnyy continued. “In other similar recommendations for the treatment of axSpA, the only point which is clearly made with regards to nonresponders to effective anti-inflammatory treatment is to ‘check the diagnosis.’”
Multiple Reasons for Nonresponse
“While the term difficult-to-treat can refer to refractory disease, that is not the only reason why a patient might not be responding to medication. In fact, it’s likely that truly biologically refractory disease makes up only a fraction of cases that respond inadequately to treatment,” said Shikha Singla, MD, who directs the psoriatic arthritis program at the Medical College of Wisconsin in Milwaukee. She is also involved with the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) initiative to define Difficult-to-Treat and Complex-to-Manage PsA.
“Apart from the persistent articular and periarticular inflammation, there could be multiple noninflammatory factors that may be contributing to this treatment-resistant disease, including comorbid conditions such as obesity, cardiovascular disease, fibromyalgia, and even social factors such as limited access to medications,” she told this news organization. “Given these complexities, it is a matter of supreme importance to recognize and carefully delineate the elements that contribute to treatment refractory disease: Is it truly the inflammation, or are there noninflammatory components that are causing the treatment failure, or a combination of the two?”
Other contributing factors could be depression, hypersensitization, and comorbidities that prevent certain treatment approaches, added Fabian Proft, MD, also of Charité – Universitätsmedizin Berlin. Dr. Proft discussed these difficult-to-treat definition efforts at the recent Spondyloarthritis Research and Treatment Network (SPARTAN) annual meeting held in Cleveland. Patients also might not be taking their medication regularly and may be seeking alternative medicine approaches, he said.
“There is a quite clear consensus within the community” that differentiation between these two groups is needed, Dr. Proft said.
The Definitions
Terminology for these two groups can vary by professional society. The European Alliance of Associations for Rheumatology (EULAR) published a definition for “difficult-to-treat” rheumatoid arthritis (RA) that includes cases with “both inflammatory activity and/or noninflammatory complaints.”
The definition includes three criteria:
1) Treatment according to EULAR recommendation and failure of at least two biologic DMARDs (bDMARDs) or targeted synthetic DMARDs (tsDMARDs) (with different mechanisms of action) after failing conventional synthetic DMARD therapy (unless contraindicated)
2) Signs suggestive of active/progressive disease, including at least one of the following:
- Moderate disease activity (according to validated composite measures including joint counts)
- Signs (including acute phase reactants and imaging) and/or symptoms suggestive of active disease, whether joint-related or other
- Inability to taper glucocorticoid treatment
- Rapid radiographic progression (with or without signs of active disease)
- RA symptoms that are causing a reduction in quality of life
3) Symptom/sign management perceived as problematic by the rheumatologist or the patient
All three criteria must be met.
Both GRAPPA and ASAS plan to use the term “difficult-to-treat” or “treatment refractory” to describe true biologically refractory inflammatory disease and are categorizing the larger, heterogeneous group of nonresponders as “difficult-to-manage” (ASAS) or “complex-to-manage” (GRAPPA).
According to Dr. Poddubnyy, the agreed ASAS definition of difficult-to-manage has several similarities with EULAR’s RA definition, including three pillars:
- Treatment according to existing recommendations and failure of at least two different bDMARDs or tsDMARDs with different mechanisms
- Having signs and symptoms of disease (measured by high disease activity by certain disease activity indexes, persistently elevated C-reactive protein, inflammation on MRI, or rapid radiographic spinal progression)
- Symptoms/signs of disease that are considered problematic by the provider or patient
The definition was approved in January, and the manuscript is in the works, Dr. Poddubnyy said.
The GRAPPA project on PsA is still in its early stages, which so far has included a comprehensive literature review as well as a survey of GRAPPA members across 47 countries. The group is generally in agreement that two separate definitions for nonresponse to treatment are necessary, and that the “difficult-to-treat” definition — which identifies true refractory disease — should include objective signs of inflammation, Dr. Singla said.
Looking Forward
The next step of the ASAS project is to “define the pathway” from difficult-to-manage axSpA to treatment refractory disease, Dr. Poddubnyy said.
“What should be ruled out in order to exclude so-called noninflammatory causes of pain?” he continued. “It will require some Delphi exercises and [a] consensus approach.”
Proft anticipates that this treatment refractory definition in both axSpA and PsA will be most useful in research, rather than clinical practice.
“It is really important to have unified definition criteria to shape as homogeneous a cohort as possible,” he said, for future clinical trials in this population.
On the other hand, the complex/difficult-to-manage definition may be more useful for clinical practice, Dr. Proft thought.
“If you see a patient not responding to treatment, the easiest thing you can do would be to change treatment,” like swapping one biologic for another, Dr. Poddubnyy added, “but this would not be the right approach in every patient.” One goal of these initiatives is to give guidance on “what things should be looked after or excluded before you conclude this is biological [nonresponse],” he said.
Dr. Singla consults for AbbVie, Janssen, and UCB and received research funding from Eli Lilly. Dr. Poddubnyy disclosed serving as a speaker, consultant, and/or research grant recipient for multiple companies including AbbVie, Lilly, Merck Sharp and Dohme, Novartis, Pfizer, GlaxoSmithKline, Novartis, and UCB. Dr. Proft reported receiving research grants, consultant fees, or support for attending meetings and/or travel from Amgen, AbbVie, Bristol Myers Squibb, Celgene, Eli Lilly, Janssen, Merck Sharp and Dohme, Novartis, Pfizer, Roche, UCB, Medscape Medical News, Galapagos, and Hexal. Dr. Proft also participants on a data safety monitoring board or advisory board for AbbVie, Celgene, Janssen, Novartis, and UCB.
A version of this article appeared on Medscape.com.
Despite an expanding arsenal of disease-modifying antirheumatic drugs (DMARDs), many patients with axial spondyloarthritis (axSpA) and psoriatic arthritis (PsA) still struggle to reach remission even after trying multiple advanced treatments.
Now, international groups of experts are working to better define these “difficult-to-treat” patients to both inform care and improve selection of participants for future clinical trials.
“The idea is rather simple, and the need is relatively ubiquitous,” Denis Poddubnyy, MD, of the Charité – Universitätsmedizin Berlin and the German Rheumatism Research Center Berlin, both in Berlin, Germany, said in an interview. He is the co-primary investigator for the ongoing Assessment of SpondyloArthritis International Society (ASAS) project to develop a consensus definition of difficult-to-treat axSpA.
According to ASAS, only 40%-50% of patients with axSpA achieve a 40% improvement in ASAS response criteria (ASAS40), and few (10%-20%) achieve remission in the first 4-6 months of treatment.
“If you look into current clinical guidelines, you will see that there is no clear guidance,” on how to manage these patients, Dr. Poddubnyy continued. “In other similar recommendations for the treatment of axSpA, the only point which is clearly made with regards to nonresponders to effective anti-inflammatory treatment is to ‘check the diagnosis.’”
Multiple Reasons for Nonresponse
“While the term difficult-to-treat can refer to refractory disease, that is not the only reason why a patient might not be responding to medication. In fact, it’s likely that truly biologically refractory disease makes up only a fraction of cases that respond inadequately to treatment,” said Shikha Singla, MD, who directs the psoriatic arthritis program at the Medical College of Wisconsin in Milwaukee. She is also involved with the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) initiative to define Difficult-to-Treat and Complex-to-Manage PsA.
“Apart from the persistent articular and periarticular inflammation, there could be multiple noninflammatory factors that may be contributing to this treatment-resistant disease, including comorbid conditions such as obesity, cardiovascular disease, fibromyalgia, and even social factors such as limited access to medications,” she told this news organization. “Given these complexities, it is a matter of supreme importance to recognize and carefully delineate the elements that contribute to treatment refractory disease: Is it truly the inflammation, or are there noninflammatory components that are causing the treatment failure, or a combination of the two?”
Other contributing factors could be depression, hypersensitization, and comorbidities that prevent certain treatment approaches, added Fabian Proft, MD, also of Charité – Universitätsmedizin Berlin. Dr. Proft discussed these difficult-to-treat definition efforts at the recent Spondyloarthritis Research and Treatment Network (SPARTAN) annual meeting held in Cleveland. Patients also might not be taking their medication regularly and may be seeking alternative medicine approaches, he said.
“There is a quite clear consensus within the community” that differentiation between these two groups is needed, Dr. Proft said.
The Definitions
Terminology for these two groups can vary by professional society. The European Alliance of Associations for Rheumatology (EULAR) published a definition for “difficult-to-treat” rheumatoid arthritis (RA) that includes cases with “both inflammatory activity and/or noninflammatory complaints.”
The definition includes three criteria:
1) Treatment according to EULAR recommendation and failure of at least two biologic DMARDs (bDMARDs) or targeted synthetic DMARDs (tsDMARDs) (with different mechanisms of action) after failing conventional synthetic DMARD therapy (unless contraindicated)
2) Signs suggestive of active/progressive disease, including at least one of the following:
- Moderate disease activity (according to validated composite measures including joint counts)
- Signs (including acute phase reactants and imaging) and/or symptoms suggestive of active disease, whether joint-related or other
- Inability to taper glucocorticoid treatment
- Rapid radiographic progression (with or without signs of active disease)
- RA symptoms that are causing a reduction in quality of life
3) Symptom/sign management perceived as problematic by the rheumatologist or the patient
All three criteria must be met.
Both GRAPPA and ASAS plan to use the term “difficult-to-treat” or “treatment refractory” to describe true biologically refractory inflammatory disease and are categorizing the larger, heterogeneous group of nonresponders as “difficult-to-manage” (ASAS) or “complex-to-manage” (GRAPPA).
According to Dr. Poddubnyy, the agreed ASAS definition of difficult-to-manage has several similarities with EULAR’s RA definition, including three pillars:
- Treatment according to existing recommendations and failure of at least two different bDMARDs or tsDMARDs with different mechanisms
- Having signs and symptoms of disease (measured by high disease activity by certain disease activity indexes, persistently elevated C-reactive protein, inflammation on MRI, or rapid radiographic spinal progression)
- Symptoms/signs of disease that are considered problematic by the provider or patient
The definition was approved in January, and the manuscript is in the works, Dr. Poddubnyy said.
The GRAPPA project on PsA is still in its early stages, which so far has included a comprehensive literature review as well as a survey of GRAPPA members across 47 countries. The group is generally in agreement that two separate definitions for nonresponse to treatment are necessary, and that the “difficult-to-treat” definition — which identifies true refractory disease — should include objective signs of inflammation, Dr. Singla said.
Looking Forward
The next step of the ASAS project is to “define the pathway” from difficult-to-manage axSpA to treatment refractory disease, Dr. Poddubnyy said.
“What should be ruled out in order to exclude so-called noninflammatory causes of pain?” he continued. “It will require some Delphi exercises and [a] consensus approach.”
Proft anticipates that this treatment refractory definition in both axSpA and PsA will be most useful in research, rather than clinical practice.
“It is really important to have unified definition criteria to shape as homogeneous a cohort as possible,” he said, for future clinical trials in this population.
On the other hand, the complex/difficult-to-manage definition may be more useful for clinical practice, Dr. Proft thought.
“If you see a patient not responding to treatment, the easiest thing you can do would be to change treatment,” like swapping one biologic for another, Dr. Poddubnyy added, “but this would not be the right approach in every patient.” One goal of these initiatives is to give guidance on “what things should be looked after or excluded before you conclude this is biological [nonresponse],” he said.
Dr. Singla consults for AbbVie, Janssen, and UCB and received research funding from Eli Lilly. Dr. Poddubnyy disclosed serving as a speaker, consultant, and/or research grant recipient for multiple companies including AbbVie, Lilly, Merck Sharp and Dohme, Novartis, Pfizer, GlaxoSmithKline, Novartis, and UCB. Dr. Proft reported receiving research grants, consultant fees, or support for attending meetings and/or travel from Amgen, AbbVie, Bristol Myers Squibb, Celgene, Eli Lilly, Janssen, Merck Sharp and Dohme, Novartis, Pfizer, Roche, UCB, Medscape Medical News, Galapagos, and Hexal. Dr. Proft also participants on a data safety monitoring board or advisory board for AbbVie, Celgene, Janssen, Novartis, and UCB.
A version of this article appeared on Medscape.com.
FROM SPARTAN 2024
Autoantibodies Nonspecific to Systemic Sclerosis May Play Role in ILD Prediction
VIENNA — Anti-Ro/SSA antibodies may help predict which patients with systemic sclerosis (SSc) are at a greater risk for interstitial lung disease (ILD) and may serve as a biomarker to guide screening, according to an analysis of data from a large European cohort.
The researchers were led by Blaž Burja, MD, PhD, a physician-scientist at the Center of Experimental Rheumatology, University Hospital Zürich, Switzerland, who reported that anti-Ro/SSA antibodies are a risk factor for ILD, with an odds ratio of 1.24, in patients with SSc.
At the annual European Congress of Rheumatology, he presented the findings of the study that aimed to find out if SSc-nonspecific antibodies might help better risk-stratify patients with SSc, focusing on lung involvement. “Among them, anti-Ro/SSA antibodies have been shown to be associated with interstitial lung disease in different connective tissue diseases,” Dr. Burja pointed out.
“A total of 15% of all patients in the SSc cohort presented with anti-Ro/SSA antibodies, and this subgroup presented with distinct clinical features: Importantly, higher prevalence of ILD and lower DLCO% [diffusing capacity of the lungs for carbon monoxide] in patients with established ILD,” reported Dr. Burja. “However, these anti-Ro/SSA antibodies do not predict ILD progression, death, or overall disease progression.”
Based on the findings, Dr. Burja suggested that these antibodies be incorporated into routine clinical practice to identify patients with SSc who have a high risk for ILD. He noted that “this has specific importance in clinical settings without availability of high-resolution computed tomography (HRCT), where anti-Ro/SSA antibodies could represent an additional biomarker to guide the screening process, in particular, in patients without SSc-specific antibodies.”
Caroline Ospelt, MD, PhD, co-moderator of the session and scientific program chair of EULAR 2024, told this news organization that the study was unique in its approach to studying ILD risk by “looking outside the box, so not just at specific antibodies but whether cross-disease antibodies may have value in stratifying patients and help predict risk of lung involvement and possibly monitor these patients.”
Dr. Ospelt, professor of experimental rheumatology at University Hospital Zürich, who was not involved in the study, noted: “It might also be the case that we could adapt this concept and use these antibodies in other rheumatic diseases, too, not just systemic sclerosis, to predict lung involvement.”
Risk-Stratifying With SSc-Nonspecific Antibodies
Dr. Burja explained that despite better stratification of patients with SSc with SSc-specific antibodies, “in clinical practice, we see large heterogeneity, and individual prognosis with regards to outcomes is still unpredictable, so we wanted to know whether by using nonspecific autoantibodies we might be better able to risk-stratify these patients.”
A study population of 4421 with at least one follow-up visit, including 3060 patients with available follow-up serologic data, was drawn from the European Scleroderma Trials and Research group database (n = 22,482). Of these 3060 patients, 461 were positive for anti-Ro/SSA antibodies and 2599 were negative. The researchers analyzed the relationships between baseline characteristics and the development or progression of ILD over 2.7 years of follow-up. Incident, de novo ILD was defined based on its presence on HRCT, and progression was defined by whether the percentage of predicted forced vital capacity (FVC%) dropped ≥ 10%, FVC% dropped 5%-9% in association with a DLCO% drop ≥ 15%, or FVC% dropped > 5%. Deaths from all causes and prognostic factors for the progression of lung fibrosis during follow-up were recorded.
High Prevalence of ILD With Anti-Ro/SSA Antibodies in SSc
At baseline, patients with anti-Ro/SSA antibodies were aged 55-56 years, 84%-87% were women, and muscular involvement was present in 18% of patients positive for anti-Ro/SSA antibodies and 12.5% of those who were negative (P < .001). According to HRCT, ILD was present in 56.2% of patients positive for anti-Ro/SSA antibodies and in 47.8% of those who were negative (P = .001). FVC% was 92.5% in patients positive for anti-Ro/SSA antibodies and 95.7% in those who were negative (P = .002). DLCO% was 66.9% in patients positive for anti-Ro/SSA antibodies and 71% in those who were negative (P < .001).
“A total of 15% of all SSc patients presented as positive for anti-Ro/SSA antibodies, and these patients all presented with higher prevalence of SSA-nonspecific antibodies, too: Of note, those with anti-La/SSB and anti-U1/RNP and rheumatoid factor,” Dr. Burja reported.
In patients with anti-U1/RNP autoantibodies, 1% were positive and 4% were negative for anti-Ro/SSA antibodies; in those with anti-La/SSB autoantibodies, 17% were positive and 1% were negative for anti-Ro/SSA antibodies; and in those with rheumatoid factor, 28% were positive and 14% were negative for anti-Ro/SSA antibodies.
Dr. Burja pointed out that the average disease duration in the study cohort at baseline was 7 years, “and at this timepoint, we expect to see some common disease manifestations. Specifically, higher muscular involvement and higher ILD based on HRCT.
“We decided to focus on patients with established ILD at baseline,” said Dr. Burja. “Anti-Ro/SSA-positive patients with established ILD at baseline presented with lower DLCO values at 59% in patients positive for anti-Ro/SSA antibodies and 61% for those who were negative.”
After conducting a multivariable analysis of 14,066 healthcare visits and adjusting for known risk factors for ILD, the researchers concluded that anti-Ro/SSA antibodies are an independent risk factor for ILD, with an odds ratio of 1.24 (95% CI, 1.07-1.44; P = .006). They also determined that anti-Ro/SSA antibodies are a risk factor for lower DLCO values in patients with ILD, with a regression coefficient of −1.93.
The researchers then explored the progression of ILD and overall disease progression and survival during the follow-up period in a longitudinal analysis. “However, anti-Ro/SSA antibodies were not found to predict the progression of ILD,” reported Dr. Burja, adding that this was true regardless of the definition of ILD progression used. “Nor did anti-Ro/SSA antibodies do not predict survival or overall disease progression.”
Dr. Burja pointed out the limitations in his study, including the lack of standardized criteria for all centers to assess anti-Ro/SSA positivity; there was a lack of discrimination between anti-Ro52 and anti-Ro60 subtypes, and there were no standardized applicable criteria to study lung progression in SSc.
Dr. Burja and Dr. Ospelt had no relevant financial disclosures.
A version of this article appeared on Medscape.com.
VIENNA — Anti-Ro/SSA antibodies may help predict which patients with systemic sclerosis (SSc) are at a greater risk for interstitial lung disease (ILD) and may serve as a biomarker to guide screening, according to an analysis of data from a large European cohort.
The researchers were led by Blaž Burja, MD, PhD, a physician-scientist at the Center of Experimental Rheumatology, University Hospital Zürich, Switzerland, who reported that anti-Ro/SSA antibodies are a risk factor for ILD, with an odds ratio of 1.24, in patients with SSc.
At the annual European Congress of Rheumatology, he presented the findings of the study that aimed to find out if SSc-nonspecific antibodies might help better risk-stratify patients with SSc, focusing on lung involvement. “Among them, anti-Ro/SSA antibodies have been shown to be associated with interstitial lung disease in different connective tissue diseases,” Dr. Burja pointed out.
“A total of 15% of all patients in the SSc cohort presented with anti-Ro/SSA antibodies, and this subgroup presented with distinct clinical features: Importantly, higher prevalence of ILD and lower DLCO% [diffusing capacity of the lungs for carbon monoxide] in patients with established ILD,” reported Dr. Burja. “However, these anti-Ro/SSA antibodies do not predict ILD progression, death, or overall disease progression.”
Based on the findings, Dr. Burja suggested that these antibodies be incorporated into routine clinical practice to identify patients with SSc who have a high risk for ILD. He noted that “this has specific importance in clinical settings without availability of high-resolution computed tomography (HRCT), where anti-Ro/SSA antibodies could represent an additional biomarker to guide the screening process, in particular, in patients without SSc-specific antibodies.”
Caroline Ospelt, MD, PhD, co-moderator of the session and scientific program chair of EULAR 2024, told this news organization that the study was unique in its approach to studying ILD risk by “looking outside the box, so not just at specific antibodies but whether cross-disease antibodies may have value in stratifying patients and help predict risk of lung involvement and possibly monitor these patients.”
Dr. Ospelt, professor of experimental rheumatology at University Hospital Zürich, who was not involved in the study, noted: “It might also be the case that we could adapt this concept and use these antibodies in other rheumatic diseases, too, not just systemic sclerosis, to predict lung involvement.”
Risk-Stratifying With SSc-Nonspecific Antibodies
Dr. Burja explained that despite better stratification of patients with SSc with SSc-specific antibodies, “in clinical practice, we see large heterogeneity, and individual prognosis with regards to outcomes is still unpredictable, so we wanted to know whether by using nonspecific autoantibodies we might be better able to risk-stratify these patients.”
A study population of 4421 with at least one follow-up visit, including 3060 patients with available follow-up serologic data, was drawn from the European Scleroderma Trials and Research group database (n = 22,482). Of these 3060 patients, 461 were positive for anti-Ro/SSA antibodies and 2599 were negative. The researchers analyzed the relationships between baseline characteristics and the development or progression of ILD over 2.7 years of follow-up. Incident, de novo ILD was defined based on its presence on HRCT, and progression was defined by whether the percentage of predicted forced vital capacity (FVC%) dropped ≥ 10%, FVC% dropped 5%-9% in association with a DLCO% drop ≥ 15%, or FVC% dropped > 5%. Deaths from all causes and prognostic factors for the progression of lung fibrosis during follow-up were recorded.
High Prevalence of ILD With Anti-Ro/SSA Antibodies in SSc
At baseline, patients with anti-Ro/SSA antibodies were aged 55-56 years, 84%-87% were women, and muscular involvement was present in 18% of patients positive for anti-Ro/SSA antibodies and 12.5% of those who were negative (P < .001). According to HRCT, ILD was present in 56.2% of patients positive for anti-Ro/SSA antibodies and in 47.8% of those who were negative (P = .001). FVC% was 92.5% in patients positive for anti-Ro/SSA antibodies and 95.7% in those who were negative (P = .002). DLCO% was 66.9% in patients positive for anti-Ro/SSA antibodies and 71% in those who were negative (P < .001).
“A total of 15% of all SSc patients presented as positive for anti-Ro/SSA antibodies, and these patients all presented with higher prevalence of SSA-nonspecific antibodies, too: Of note, those with anti-La/SSB and anti-U1/RNP and rheumatoid factor,” Dr. Burja reported.
In patients with anti-U1/RNP autoantibodies, 1% were positive and 4% were negative for anti-Ro/SSA antibodies; in those with anti-La/SSB autoantibodies, 17% were positive and 1% were negative for anti-Ro/SSA antibodies; and in those with rheumatoid factor, 28% were positive and 14% were negative for anti-Ro/SSA antibodies.
Dr. Burja pointed out that the average disease duration in the study cohort at baseline was 7 years, “and at this timepoint, we expect to see some common disease manifestations. Specifically, higher muscular involvement and higher ILD based on HRCT.
“We decided to focus on patients with established ILD at baseline,” said Dr. Burja. “Anti-Ro/SSA-positive patients with established ILD at baseline presented with lower DLCO values at 59% in patients positive for anti-Ro/SSA antibodies and 61% for those who were negative.”
After conducting a multivariable analysis of 14,066 healthcare visits and adjusting for known risk factors for ILD, the researchers concluded that anti-Ro/SSA antibodies are an independent risk factor for ILD, with an odds ratio of 1.24 (95% CI, 1.07-1.44; P = .006). They also determined that anti-Ro/SSA antibodies are a risk factor for lower DLCO values in patients with ILD, with a regression coefficient of −1.93.
The researchers then explored the progression of ILD and overall disease progression and survival during the follow-up period in a longitudinal analysis. “However, anti-Ro/SSA antibodies were not found to predict the progression of ILD,” reported Dr. Burja, adding that this was true regardless of the definition of ILD progression used. “Nor did anti-Ro/SSA antibodies do not predict survival or overall disease progression.”
Dr. Burja pointed out the limitations in his study, including the lack of standardized criteria for all centers to assess anti-Ro/SSA positivity; there was a lack of discrimination between anti-Ro52 and anti-Ro60 subtypes, and there were no standardized applicable criteria to study lung progression in SSc.
Dr. Burja and Dr. Ospelt had no relevant financial disclosures.
A version of this article appeared on Medscape.com.
VIENNA — Anti-Ro/SSA antibodies may help predict which patients with systemic sclerosis (SSc) are at a greater risk for interstitial lung disease (ILD) and may serve as a biomarker to guide screening, according to an analysis of data from a large European cohort.
The researchers were led by Blaž Burja, MD, PhD, a physician-scientist at the Center of Experimental Rheumatology, University Hospital Zürich, Switzerland, who reported that anti-Ro/SSA antibodies are a risk factor for ILD, with an odds ratio of 1.24, in patients with SSc.
At the annual European Congress of Rheumatology, he presented the findings of the study that aimed to find out if SSc-nonspecific antibodies might help better risk-stratify patients with SSc, focusing on lung involvement. “Among them, anti-Ro/SSA antibodies have been shown to be associated with interstitial lung disease in different connective tissue diseases,” Dr. Burja pointed out.
“A total of 15% of all patients in the SSc cohort presented with anti-Ro/SSA antibodies, and this subgroup presented with distinct clinical features: Importantly, higher prevalence of ILD and lower DLCO% [diffusing capacity of the lungs for carbon monoxide] in patients with established ILD,” reported Dr. Burja. “However, these anti-Ro/SSA antibodies do not predict ILD progression, death, or overall disease progression.”
Based on the findings, Dr. Burja suggested that these antibodies be incorporated into routine clinical practice to identify patients with SSc who have a high risk for ILD. He noted that “this has specific importance in clinical settings without availability of high-resolution computed tomography (HRCT), where anti-Ro/SSA antibodies could represent an additional biomarker to guide the screening process, in particular, in patients without SSc-specific antibodies.”
Caroline Ospelt, MD, PhD, co-moderator of the session and scientific program chair of EULAR 2024, told this news organization that the study was unique in its approach to studying ILD risk by “looking outside the box, so not just at specific antibodies but whether cross-disease antibodies may have value in stratifying patients and help predict risk of lung involvement and possibly monitor these patients.”
Dr. Ospelt, professor of experimental rheumatology at University Hospital Zürich, who was not involved in the study, noted: “It might also be the case that we could adapt this concept and use these antibodies in other rheumatic diseases, too, not just systemic sclerosis, to predict lung involvement.”
Risk-Stratifying With SSc-Nonspecific Antibodies
Dr. Burja explained that despite better stratification of patients with SSc with SSc-specific antibodies, “in clinical practice, we see large heterogeneity, and individual prognosis with regards to outcomes is still unpredictable, so we wanted to know whether by using nonspecific autoantibodies we might be better able to risk-stratify these patients.”
A study population of 4421 with at least one follow-up visit, including 3060 patients with available follow-up serologic data, was drawn from the European Scleroderma Trials and Research group database (n = 22,482). Of these 3060 patients, 461 were positive for anti-Ro/SSA antibodies and 2599 were negative. The researchers analyzed the relationships between baseline characteristics and the development or progression of ILD over 2.7 years of follow-up. Incident, de novo ILD was defined based on its presence on HRCT, and progression was defined by whether the percentage of predicted forced vital capacity (FVC%) dropped ≥ 10%, FVC% dropped 5%-9% in association with a DLCO% drop ≥ 15%, or FVC% dropped > 5%. Deaths from all causes and prognostic factors for the progression of lung fibrosis during follow-up were recorded.
High Prevalence of ILD With Anti-Ro/SSA Antibodies in SSc
At baseline, patients with anti-Ro/SSA antibodies were aged 55-56 years, 84%-87% were women, and muscular involvement was present in 18% of patients positive for anti-Ro/SSA antibodies and 12.5% of those who were negative (P < .001). According to HRCT, ILD was present in 56.2% of patients positive for anti-Ro/SSA antibodies and in 47.8% of those who were negative (P = .001). FVC% was 92.5% in patients positive for anti-Ro/SSA antibodies and 95.7% in those who were negative (P = .002). DLCO% was 66.9% in patients positive for anti-Ro/SSA antibodies and 71% in those who were negative (P < .001).
“A total of 15% of all SSc patients presented as positive for anti-Ro/SSA antibodies, and these patients all presented with higher prevalence of SSA-nonspecific antibodies, too: Of note, those with anti-La/SSB and anti-U1/RNP and rheumatoid factor,” Dr. Burja reported.
In patients with anti-U1/RNP autoantibodies, 1% were positive and 4% were negative for anti-Ro/SSA antibodies; in those with anti-La/SSB autoantibodies, 17% were positive and 1% were negative for anti-Ro/SSA antibodies; and in those with rheumatoid factor, 28% were positive and 14% were negative for anti-Ro/SSA antibodies.
Dr. Burja pointed out that the average disease duration in the study cohort at baseline was 7 years, “and at this timepoint, we expect to see some common disease manifestations. Specifically, higher muscular involvement and higher ILD based on HRCT.
“We decided to focus on patients with established ILD at baseline,” said Dr. Burja. “Anti-Ro/SSA-positive patients with established ILD at baseline presented with lower DLCO values at 59% in patients positive for anti-Ro/SSA antibodies and 61% for those who were negative.”
After conducting a multivariable analysis of 14,066 healthcare visits and adjusting for known risk factors for ILD, the researchers concluded that anti-Ro/SSA antibodies are an independent risk factor for ILD, with an odds ratio of 1.24 (95% CI, 1.07-1.44; P = .006). They also determined that anti-Ro/SSA antibodies are a risk factor for lower DLCO values in patients with ILD, with a regression coefficient of −1.93.
The researchers then explored the progression of ILD and overall disease progression and survival during the follow-up period in a longitudinal analysis. “However, anti-Ro/SSA antibodies were not found to predict the progression of ILD,” reported Dr. Burja, adding that this was true regardless of the definition of ILD progression used. “Nor did anti-Ro/SSA antibodies do not predict survival or overall disease progression.”
Dr. Burja pointed out the limitations in his study, including the lack of standardized criteria for all centers to assess anti-Ro/SSA positivity; there was a lack of discrimination between anti-Ro52 and anti-Ro60 subtypes, and there were no standardized applicable criteria to study lung progression in SSc.
Dr. Burja and Dr. Ospelt had no relevant financial disclosures.
A version of this article appeared on Medscape.com.
FROM EULAR 2024
FDA Expands Repotrectinib Label to All NTRK Gene Fusion+ Solid Tumors
The approval is a label expansion for the tyrosine kinase inhibitor (TKI), which received initial clearance in November 2023 for locally advanced or metastatic ROS1-positive non–small cell lung cancer.
NTRK gene fusions are genetic abnormalities wherein part of the NTRK gene fuses with an unrelated gene. The abnormal gene can then produce an oncogenic protein. Although rare, these mutations are found in many cancer types.
The approval, for adult and pediatric patients aged 12 years or older, was based on the single-arm open-label TRIDENT-1 trial in 88 adults with locally advanced or metastatic NTRK gene fusion solid tumors.
In the 40 patients who were TKI-naive, the overall response rate was 58%, and the median duration of response was not estimable. In the 48 patients who had a TKI previously, the overall response rate was 50% and median duration of response was 9.9 months.
In 20% or more of participants, treatment caused dizziness, dysgeusia, peripheral neuropathy, constipation, dyspnea, fatigue, ataxia, cognitive impairment, muscular weakness, and nausea.
Labeling warns of central nervous system reactions, interstitial lung disease/pneumonitis, hepatotoxicity, myalgia with creatine phosphokinase elevation, hyperuricemia, bone fractures, and embryo-fetal toxicity.
The recommended dose is 160 mg orally once daily for 14 days then increased to 160 mg twice daily until disease progression or unacceptable toxicity.
Sixty 40-mg capsules cost around $7,644, according to drugs.com.
A version of this article appeared on Medscape.com.
The approval is a label expansion for the tyrosine kinase inhibitor (TKI), which received initial clearance in November 2023 for locally advanced or metastatic ROS1-positive non–small cell lung cancer.
NTRK gene fusions are genetic abnormalities wherein part of the NTRK gene fuses with an unrelated gene. The abnormal gene can then produce an oncogenic protein. Although rare, these mutations are found in many cancer types.
The approval, for adult and pediatric patients aged 12 years or older, was based on the single-arm open-label TRIDENT-1 trial in 88 adults with locally advanced or metastatic NTRK gene fusion solid tumors.
In the 40 patients who were TKI-naive, the overall response rate was 58%, and the median duration of response was not estimable. In the 48 patients who had a TKI previously, the overall response rate was 50% and median duration of response was 9.9 months.
In 20% or more of participants, treatment caused dizziness, dysgeusia, peripheral neuropathy, constipation, dyspnea, fatigue, ataxia, cognitive impairment, muscular weakness, and nausea.
Labeling warns of central nervous system reactions, interstitial lung disease/pneumonitis, hepatotoxicity, myalgia with creatine phosphokinase elevation, hyperuricemia, bone fractures, and embryo-fetal toxicity.
The recommended dose is 160 mg orally once daily for 14 days then increased to 160 mg twice daily until disease progression or unacceptable toxicity.
Sixty 40-mg capsules cost around $7,644, according to drugs.com.
A version of this article appeared on Medscape.com.
The approval is a label expansion for the tyrosine kinase inhibitor (TKI), which received initial clearance in November 2023 for locally advanced or metastatic ROS1-positive non–small cell lung cancer.
NTRK gene fusions are genetic abnormalities wherein part of the NTRK gene fuses with an unrelated gene. The abnormal gene can then produce an oncogenic protein. Although rare, these mutations are found in many cancer types.
The approval, for adult and pediatric patients aged 12 years or older, was based on the single-arm open-label TRIDENT-1 trial in 88 adults with locally advanced or metastatic NTRK gene fusion solid tumors.
In the 40 patients who were TKI-naive, the overall response rate was 58%, and the median duration of response was not estimable. In the 48 patients who had a TKI previously, the overall response rate was 50% and median duration of response was 9.9 months.
In 20% or more of participants, treatment caused dizziness, dysgeusia, peripheral neuropathy, constipation, dyspnea, fatigue, ataxia, cognitive impairment, muscular weakness, and nausea.
Labeling warns of central nervous system reactions, interstitial lung disease/pneumonitis, hepatotoxicity, myalgia with creatine phosphokinase elevation, hyperuricemia, bone fractures, and embryo-fetal toxicity.
The recommended dose is 160 mg orally once daily for 14 days then increased to 160 mg twice daily until disease progression or unacceptable toxicity.
Sixty 40-mg capsules cost around $7,644, according to drugs.com.
A version of this article appeared on Medscape.com.
AMA Wrestles With AI But Acts on Prior Authorization, Other Concerns
The largest US physician organization wrestled with the professional risks and rewards of artificial intelligence (AI) at its annual meeting, delaying action even as it adopted new policies on prior authorization and other concerns for clinicians and patients.
Physicians and medical students at the annual meeting of the American Medical Association (AMA) House of Delegates in Chicago intensely debated a report and two key resolutions on AI but could not reach consensus, pushing off decision-making until a future meeting in November.
One resolution would establish “augmented intelligence” as the preferred term for AI, reflecting the desired role of these tools in supporting — not making — physicians’ decisions. The other resolution focused on insurers’ use of AI in determining medical necessity.
(See specific policies adopted at the meeting, held June 8-12, below.)
A comprehensive AMA trustees’ report on AI considered additional issues including requirements for disclosing AI use, liability for harms due to flawed application of AI, data privacy, and cybersecurity.
The AMA intends to “continue to methodically assess these issues and make informed recommendations in proposing new policy,” said Bobby Mukkamala, MD, an otolaryngologist from Flint, Michigan, who became the AMA’s new president-elect.
AMA members at the meeting largely applauded the aim of these AI proposals, but some objected to parts of the trustees’ report.
They raised questions about what, exactly, constitutes an AI-powered service and whether all AI tools need the kind of guardrails the AMA may seek. There also were concerns about calls to make AI use more transparent.
While transparency might be an admirable goal, it might prove too hard to achieve given that AI-powered tools and products are already woven into medical practice in ways that physicians may not know or understand, said Christopher Libby, MD, MPH, a clinical informaticist and emergency physician at Cedars Sinai Medical Center in Los Angeles.
“It’s hard for the practicing clinician to know how every piece of technology works in order to describe it to the patient,” Dr. Libby said at the meeting. “How many people here can identify when algorithms are used in their EHR today?”
He suggested asking for more transparency from the companies that make and sell AI-powered software and tools to insurers and healthcare systems.
Steven H. Kroft, MD, the editor of the American Journal of Clinical Pathology, raised concerns about the unintended harm that unchecked use of AI may pose to scientific research.
He asked the AMA to address “a significant omission in an otherwise comprehensive report” — the need to protect the integrity of study results that can direct patient care.
“While sham science is not a new issue, large language models make it far easier for authors to generate fake papers and far harder for editors, reviewers, and publishers to identify them,” Dr. Kroft said. “This is a rapidly growing phenomenon that is threatening the integrity of the literature. These papers become embedded in the evidence bases that drive clinical decision-making.”
AMA has been working with specialty societies and outside AI experts to refine an effective set of recommendations. The new policies, once finalized, are intended to build on steps AMA already has taken, including last year releasing principles for AI development, deployment, and use.
Congress Mulling
The AMA delegates are far from alone in facing AI policy challenges.
Leaders in Congress also are examining AI guardrails, with influential panels such as the Senate Finance and House Energy and Commerce committees holding hearings.
A key congressional AI effort to watch is the expected implementation of a bipartisan Senate “road map,” which Senate Majority Leader Chuck Schumer (D-NY) and colleagues released in May, said Miranda A. Franco, a senior policy advisor at the law firm Holland & Knight.
The product of many months of deliberation, this Senate road map identifies priorities for future legislation, including:
- Creating appropriate guardrails and safety measures to protect patients.
- Making healthcare and biomedical data available for machine learning and data science research while carefully addressing privacy issues.
- Providing transparency for clinicians and the public about the use of AI in medical products and clinical support services, including the data used to train models.
- Examining the Centers for Medicare & Medicaid Services’ reimbursement mechanisms as well as guardrails to ensure accountability, appropriate use, and broad application of AI across all populations.
Congress likely will address issues of AI in healthcare in piecemeal fashion, taking on different aspects of these challenges at different times, Ms. Franco said. The Senate road map gives the key committees directions on where to proceed in their efforts to develop new laws.
“I think this is all going to be slow and rolling, not big and sweeping,” Ms. Franco told this news organization. “I don’t think we’re going to see an encompassing AI bill.”
AMA Policies Adopted on Other Issues
At the June meeting, AMA delegates adopted the following policies aiming to:
- Increase oversight and accountability of health insurers’ use of prior authorization controls on patient access to care.
- Encourage policy changes allowing physicians to receive loan forgiveness when they practice in an Indian Health Service, Tribal, or Urban Indian Health Program, similar to physicians practicing in a Veterans Administration facility.
- Advocate for federal policy that limits a patient’s out-of-pocket cost to be the same or less than the amount that a patient with traditional Medicare plus a Medigap plan would pay.
- Oppose state or national legislation that could criminalize in vitro fertilization.
- Limit what the AMA calls the “expensive” cost for Medicare Advantage enrollees who need physician-administered drugs or biologics.
- Help physicians address the handling of de-identified patient data in a rapidly changing digital health ecosystem.
- Support efforts to decriminalize the possession of non-prescribed buprenorphine for personal use by individuals who lack access to a physician for the treatment of opioid use disorder.
- Expand access to hearing, vision, and dental care. The new AMA policy advocates working with state medical associations to support coverage of hearing exams, hearing aids, cochlear implants, and vision exams and aids. The revised AMA policy also supports working with the American Dental Association and other national organizations to improve access to dental care for people enrolled in Medicare, Medicaid, and CHIP programs.
- Increase enrollment of more women and sexual and gender minority populations in clinical trials.
A version of this article first appeared on Medscape.com.
The largest US physician organization wrestled with the professional risks and rewards of artificial intelligence (AI) at its annual meeting, delaying action even as it adopted new policies on prior authorization and other concerns for clinicians and patients.
Physicians and medical students at the annual meeting of the American Medical Association (AMA) House of Delegates in Chicago intensely debated a report and two key resolutions on AI but could not reach consensus, pushing off decision-making until a future meeting in November.
One resolution would establish “augmented intelligence” as the preferred term for AI, reflecting the desired role of these tools in supporting — not making — physicians’ decisions. The other resolution focused on insurers’ use of AI in determining medical necessity.
(See specific policies adopted at the meeting, held June 8-12, below.)
A comprehensive AMA trustees’ report on AI considered additional issues including requirements for disclosing AI use, liability for harms due to flawed application of AI, data privacy, and cybersecurity.
The AMA intends to “continue to methodically assess these issues and make informed recommendations in proposing new policy,” said Bobby Mukkamala, MD, an otolaryngologist from Flint, Michigan, who became the AMA’s new president-elect.
AMA members at the meeting largely applauded the aim of these AI proposals, but some objected to parts of the trustees’ report.
They raised questions about what, exactly, constitutes an AI-powered service and whether all AI tools need the kind of guardrails the AMA may seek. There also were concerns about calls to make AI use more transparent.
While transparency might be an admirable goal, it might prove too hard to achieve given that AI-powered tools and products are already woven into medical practice in ways that physicians may not know or understand, said Christopher Libby, MD, MPH, a clinical informaticist and emergency physician at Cedars Sinai Medical Center in Los Angeles.
“It’s hard for the practicing clinician to know how every piece of technology works in order to describe it to the patient,” Dr. Libby said at the meeting. “How many people here can identify when algorithms are used in their EHR today?”
He suggested asking for more transparency from the companies that make and sell AI-powered software and tools to insurers and healthcare systems.
Steven H. Kroft, MD, the editor of the American Journal of Clinical Pathology, raised concerns about the unintended harm that unchecked use of AI may pose to scientific research.
He asked the AMA to address “a significant omission in an otherwise comprehensive report” — the need to protect the integrity of study results that can direct patient care.
“While sham science is not a new issue, large language models make it far easier for authors to generate fake papers and far harder for editors, reviewers, and publishers to identify them,” Dr. Kroft said. “This is a rapidly growing phenomenon that is threatening the integrity of the literature. These papers become embedded in the evidence bases that drive clinical decision-making.”
AMA has been working with specialty societies and outside AI experts to refine an effective set of recommendations. The new policies, once finalized, are intended to build on steps AMA already has taken, including last year releasing principles for AI development, deployment, and use.
Congress Mulling
The AMA delegates are far from alone in facing AI policy challenges.
Leaders in Congress also are examining AI guardrails, with influential panels such as the Senate Finance and House Energy and Commerce committees holding hearings.
A key congressional AI effort to watch is the expected implementation of a bipartisan Senate “road map,” which Senate Majority Leader Chuck Schumer (D-NY) and colleagues released in May, said Miranda A. Franco, a senior policy advisor at the law firm Holland & Knight.
The product of many months of deliberation, this Senate road map identifies priorities for future legislation, including:
- Creating appropriate guardrails and safety measures to protect patients.
- Making healthcare and biomedical data available for machine learning and data science research while carefully addressing privacy issues.
- Providing transparency for clinicians and the public about the use of AI in medical products and clinical support services, including the data used to train models.
- Examining the Centers for Medicare & Medicaid Services’ reimbursement mechanisms as well as guardrails to ensure accountability, appropriate use, and broad application of AI across all populations.
Congress likely will address issues of AI in healthcare in piecemeal fashion, taking on different aspects of these challenges at different times, Ms. Franco said. The Senate road map gives the key committees directions on where to proceed in their efforts to develop new laws.
“I think this is all going to be slow and rolling, not big and sweeping,” Ms. Franco told this news organization. “I don’t think we’re going to see an encompassing AI bill.”
AMA Policies Adopted on Other Issues
At the June meeting, AMA delegates adopted the following policies aiming to:
- Increase oversight and accountability of health insurers’ use of prior authorization controls on patient access to care.
- Encourage policy changes allowing physicians to receive loan forgiveness when they practice in an Indian Health Service, Tribal, or Urban Indian Health Program, similar to physicians practicing in a Veterans Administration facility.
- Advocate for federal policy that limits a patient’s out-of-pocket cost to be the same or less than the amount that a patient with traditional Medicare plus a Medigap plan would pay.
- Oppose state or national legislation that could criminalize in vitro fertilization.
- Limit what the AMA calls the “expensive” cost for Medicare Advantage enrollees who need physician-administered drugs or biologics.
- Help physicians address the handling of de-identified patient data in a rapidly changing digital health ecosystem.
- Support efforts to decriminalize the possession of non-prescribed buprenorphine for personal use by individuals who lack access to a physician for the treatment of opioid use disorder.
- Expand access to hearing, vision, and dental care. The new AMA policy advocates working with state medical associations to support coverage of hearing exams, hearing aids, cochlear implants, and vision exams and aids. The revised AMA policy also supports working with the American Dental Association and other national organizations to improve access to dental care for people enrolled in Medicare, Medicaid, and CHIP programs.
- Increase enrollment of more women and sexual and gender minority populations in clinical trials.
A version of this article first appeared on Medscape.com.
The largest US physician organization wrestled with the professional risks and rewards of artificial intelligence (AI) at its annual meeting, delaying action even as it adopted new policies on prior authorization and other concerns for clinicians and patients.
Physicians and medical students at the annual meeting of the American Medical Association (AMA) House of Delegates in Chicago intensely debated a report and two key resolutions on AI but could not reach consensus, pushing off decision-making until a future meeting in November.
One resolution would establish “augmented intelligence” as the preferred term for AI, reflecting the desired role of these tools in supporting — not making — physicians’ decisions. The other resolution focused on insurers’ use of AI in determining medical necessity.
(See specific policies adopted at the meeting, held June 8-12, below.)
A comprehensive AMA trustees’ report on AI considered additional issues including requirements for disclosing AI use, liability for harms due to flawed application of AI, data privacy, and cybersecurity.
The AMA intends to “continue to methodically assess these issues and make informed recommendations in proposing new policy,” said Bobby Mukkamala, MD, an otolaryngologist from Flint, Michigan, who became the AMA’s new president-elect.
AMA members at the meeting largely applauded the aim of these AI proposals, but some objected to parts of the trustees’ report.
They raised questions about what, exactly, constitutes an AI-powered service and whether all AI tools need the kind of guardrails the AMA may seek. There also were concerns about calls to make AI use more transparent.
While transparency might be an admirable goal, it might prove too hard to achieve given that AI-powered tools and products are already woven into medical practice in ways that physicians may not know or understand, said Christopher Libby, MD, MPH, a clinical informaticist and emergency physician at Cedars Sinai Medical Center in Los Angeles.
“It’s hard for the practicing clinician to know how every piece of technology works in order to describe it to the patient,” Dr. Libby said at the meeting. “How many people here can identify when algorithms are used in their EHR today?”
He suggested asking for more transparency from the companies that make and sell AI-powered software and tools to insurers and healthcare systems.
Steven H. Kroft, MD, the editor of the American Journal of Clinical Pathology, raised concerns about the unintended harm that unchecked use of AI may pose to scientific research.
He asked the AMA to address “a significant omission in an otherwise comprehensive report” — the need to protect the integrity of study results that can direct patient care.
“While sham science is not a new issue, large language models make it far easier for authors to generate fake papers and far harder for editors, reviewers, and publishers to identify them,” Dr. Kroft said. “This is a rapidly growing phenomenon that is threatening the integrity of the literature. These papers become embedded in the evidence bases that drive clinical decision-making.”
AMA has been working with specialty societies and outside AI experts to refine an effective set of recommendations. The new policies, once finalized, are intended to build on steps AMA already has taken, including last year releasing principles for AI development, deployment, and use.
Congress Mulling
The AMA delegates are far from alone in facing AI policy challenges.
Leaders in Congress also are examining AI guardrails, with influential panels such as the Senate Finance and House Energy and Commerce committees holding hearings.
A key congressional AI effort to watch is the expected implementation of a bipartisan Senate “road map,” which Senate Majority Leader Chuck Schumer (D-NY) and colleagues released in May, said Miranda A. Franco, a senior policy advisor at the law firm Holland & Knight.
The product of many months of deliberation, this Senate road map identifies priorities for future legislation, including:
- Creating appropriate guardrails and safety measures to protect patients.
- Making healthcare and biomedical data available for machine learning and data science research while carefully addressing privacy issues.
- Providing transparency for clinicians and the public about the use of AI in medical products and clinical support services, including the data used to train models.
- Examining the Centers for Medicare & Medicaid Services’ reimbursement mechanisms as well as guardrails to ensure accountability, appropriate use, and broad application of AI across all populations.
Congress likely will address issues of AI in healthcare in piecemeal fashion, taking on different aspects of these challenges at different times, Ms. Franco said. The Senate road map gives the key committees directions on where to proceed in their efforts to develop new laws.
“I think this is all going to be slow and rolling, not big and sweeping,” Ms. Franco told this news organization. “I don’t think we’re going to see an encompassing AI bill.”
AMA Policies Adopted on Other Issues
At the June meeting, AMA delegates adopted the following policies aiming to:
- Increase oversight and accountability of health insurers’ use of prior authorization controls on patient access to care.
- Encourage policy changes allowing physicians to receive loan forgiveness when they practice in an Indian Health Service, Tribal, or Urban Indian Health Program, similar to physicians practicing in a Veterans Administration facility.
- Advocate for federal policy that limits a patient’s out-of-pocket cost to be the same or less than the amount that a patient with traditional Medicare plus a Medigap plan would pay.
- Oppose state or national legislation that could criminalize in vitro fertilization.
- Limit what the AMA calls the “expensive” cost for Medicare Advantage enrollees who need physician-administered drugs or biologics.
- Help physicians address the handling of de-identified patient data in a rapidly changing digital health ecosystem.
- Support efforts to decriminalize the possession of non-prescribed buprenorphine for personal use by individuals who lack access to a physician for the treatment of opioid use disorder.
- Expand access to hearing, vision, and dental care. The new AMA policy advocates working with state medical associations to support coverage of hearing exams, hearing aids, cochlear implants, and vision exams and aids. The revised AMA policy also supports working with the American Dental Association and other national organizations to improve access to dental care for people enrolled in Medicare, Medicaid, and CHIP programs.
- Increase enrollment of more women and sexual and gender minority populations in clinical trials.
A version of this article first appeared on Medscape.com.
A 7-year-old female presents with persistent pimples on the nose and cheeks for approximately 1 year
Diagnosis
During the visit, skin scrapings were performed, revealing several Demodex mites, confirming the diagnosis of demodicosis.
Demodex folliculorum and Demodex brevis are the two common species implicated. The life cycle of Demodex spp. occurs in the sebaceous glands, leading to mechanical and chemical irritation of the skin.
Various immune responses are also triggered, such as a keratinocyte response via Toll-like receptor 2. Patients usually present with non-specific symptoms such as skin erythema, irritation, peeling, and dryness on the cheeks, eyelids, and paranasal areas. Patients may develop a maculopapular or rosacea-like rash.
Diagnosis is often made through microscopic examination of a skin sample in KOH solution. In rare occasions, a skin surface standardization biopsy method may be used, which determines the density of mites per 1 cm2. Dermoscopy may identify spiky white structures. Molecular methods such as PCR can be used but are not standard.
The differential diagnosis may include acne, rosacea, folliculitis, and Candida infection. Demodicosis can be differentiated by history and further studies including dermoscopy.
Acne vulgaris is an inflammatory disease of the skin’s pilosebaceous unit, primarily involving the face and trunk. It can present with comedones, papules, pustules, and nodules. Secondary signs suggestive of acne vulgaris include scars, erythema, and hyperpigmentation. All forms of acne share a common pathogenesis resulting in the formation of microcomedones, precursors for all clinical acne lesions. In this patient, the absence of microcomedones and the presence of primary inflammatory papules localized to the nose and cheeks suggested an alternative diagnosis.
Rosacea was also considered in the differential diagnosis. Rosacea is an inflammatory dermatosis characterized by erythema, telangiectasia, recurrent flushing, and inflammatory lesions including papulopustules and swelling, primarily affecting the face. The pathogenesis of rosacea is not fully understood but is suggested to involve immune-mediated responses. Vascular dysregulation and reactive oxygen species damage keratinocytes, fibroblasts, and endothelial cells. A higher incidence of rosacea in those with a family history and UV exposure is a known trigger. Demodex folliculorum and Helicobacter pylori are also implicated. Occasionally, Demodex infestation and rosacea may co-occur, and treatment with topical metronidazole can be helpful.
Folliculitis is an infection and inflammation of the hair follicles, forming pustules or erythematous papules over hair-covered skin. It is commonly caused by bacterial infection but can also be due to fungi, viruses, and noninfectious causes such as eosinophilic folliculitis. Diagnosis is clinical, based on physical exam and history, such as recent increased sweating or scratching. KOH prep can be used for Malassezia folliculitis and skin biopsy for eosinophilic folliculitis. Treatment targets the underlying cause. Most bacterial folliculitis cases resolve without treatment, but topical antibiotics may be used. Fungal folliculitis requires oral antifungals, and herpes simplex folliculitis can be treated with antiviral medications.
Cutaneous candidiasis is an infection of the skin by various Candida species, commonly C. albicans. Superficial infections of the skin and mucous membranes, such as intertrigo, are common types. Risk factors include immunosuppression, endocrine disorders, or compromised blood flow. Increased humidity, occlusion, broken skin barriers, and altered skin microbial flora contribute to Candida infection. Diagnosis is clinical but can be confirmed by KOH prep, microscopy, and culture. Treatment involves anti-inflammatory, antibacterial, and antifungal medications. Topical clotrimazole, nystatin, and miconazole are commonly used. Recurrence is prevented by keeping the affected area dry with barrier creams.
Therapeutic goals include arresting mite reproduction, elimination, and preventing recurrent infestations. Treatment may last several months, and the choice of drug depends on patient factors. There have been no standardized treatment studies or long-term effectiveness analyses. Antibiotics such as tetracycline, metronidazole, doxycycline, and ivermectin may be used to prevent proliferation. Permethrin, benzyl benzoate, crotamiton, lindane, and sulfur have also been used. Metronidazole is a common treatment for demodicosis, as was used in our patient for several weeks until the lesions cleared. Systemic metronidazole therapy may be indicated for reducing Demodex spp. density. Severe cases, particularly in immunocompromised individuals, may require oral ivermectin. Appropriate hygiene is important for prevention, such as washing the face with non-oily cleansers and laundering linens regularly.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Mr. Lee is a medical student at the University of California San Diego.
Suggested Reading
Chudzicka-Strugała I et al. Demodicosis in different age groups and alternative treatment options—A review. J Clin Med. 2023 Feb 19;12(4):1649. doi: 10.3390/jcm12041649.
Eichenfield DZ et al. Management of acne vulgaris: A review. JAMA. 2021 Nov 23;326(20):2055-2067. doi: 10.1001/jama.2021.17633.
Sharma A et al. Rosacea management: A comprehensive review. J Cosmet Dermatol. 2022 May;21(5):1895-1904. doi: 10.1111/jocd.14816.
Taudorf EH et al. Cutaneous candidiasis — an evidence-based review of topical and systemic treatments to inform clinical practice. J Eur Acad Dermatol Venereol. 2019 Oct;33(10):1863-1873. doi: 10.1111/jdv.15782.
Winters RD, Mitchell M. Folliculitis. [Updated 2023 Aug 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024. Available from: https://www.ncbi.nlm.nih.gov/books/NBK547754/
Diagnosis
During the visit, skin scrapings were performed, revealing several Demodex mites, confirming the diagnosis of demodicosis.
Demodex folliculorum and Demodex brevis are the two common species implicated. The life cycle of Demodex spp. occurs in the sebaceous glands, leading to mechanical and chemical irritation of the skin.
Various immune responses are also triggered, such as a keratinocyte response via Toll-like receptor 2. Patients usually present with non-specific symptoms such as skin erythema, irritation, peeling, and dryness on the cheeks, eyelids, and paranasal areas. Patients may develop a maculopapular or rosacea-like rash.
Diagnosis is often made through microscopic examination of a skin sample in KOH solution. In rare occasions, a skin surface standardization biopsy method may be used, which determines the density of mites per 1 cm2. Dermoscopy may identify spiky white structures. Molecular methods such as PCR can be used but are not standard.
The differential diagnosis may include acne, rosacea, folliculitis, and Candida infection. Demodicosis can be differentiated by history and further studies including dermoscopy.
Acne vulgaris is an inflammatory disease of the skin’s pilosebaceous unit, primarily involving the face and trunk. It can present with comedones, papules, pustules, and nodules. Secondary signs suggestive of acne vulgaris include scars, erythema, and hyperpigmentation. All forms of acne share a common pathogenesis resulting in the formation of microcomedones, precursors for all clinical acne lesions. In this patient, the absence of microcomedones and the presence of primary inflammatory papules localized to the nose and cheeks suggested an alternative diagnosis.
Rosacea was also considered in the differential diagnosis. Rosacea is an inflammatory dermatosis characterized by erythema, telangiectasia, recurrent flushing, and inflammatory lesions including papulopustules and swelling, primarily affecting the face. The pathogenesis of rosacea is not fully understood but is suggested to involve immune-mediated responses. Vascular dysregulation and reactive oxygen species damage keratinocytes, fibroblasts, and endothelial cells. A higher incidence of rosacea in those with a family history and UV exposure is a known trigger. Demodex folliculorum and Helicobacter pylori are also implicated. Occasionally, Demodex infestation and rosacea may co-occur, and treatment with topical metronidazole can be helpful.
Folliculitis is an infection and inflammation of the hair follicles, forming pustules or erythematous papules over hair-covered skin. It is commonly caused by bacterial infection but can also be due to fungi, viruses, and noninfectious causes such as eosinophilic folliculitis. Diagnosis is clinical, based on physical exam and history, such as recent increased sweating or scratching. KOH prep can be used for Malassezia folliculitis and skin biopsy for eosinophilic folliculitis. Treatment targets the underlying cause. Most bacterial folliculitis cases resolve without treatment, but topical antibiotics may be used. Fungal folliculitis requires oral antifungals, and herpes simplex folliculitis can be treated with antiviral medications.
Cutaneous candidiasis is an infection of the skin by various Candida species, commonly C. albicans. Superficial infections of the skin and mucous membranes, such as intertrigo, are common types. Risk factors include immunosuppression, endocrine disorders, or compromised blood flow. Increased humidity, occlusion, broken skin barriers, and altered skin microbial flora contribute to Candida infection. Diagnosis is clinical but can be confirmed by KOH prep, microscopy, and culture. Treatment involves anti-inflammatory, antibacterial, and antifungal medications. Topical clotrimazole, nystatin, and miconazole are commonly used. Recurrence is prevented by keeping the affected area dry with barrier creams.
Therapeutic goals include arresting mite reproduction, elimination, and preventing recurrent infestations. Treatment may last several months, and the choice of drug depends on patient factors. There have been no standardized treatment studies or long-term effectiveness analyses. Antibiotics such as tetracycline, metronidazole, doxycycline, and ivermectin may be used to prevent proliferation. Permethrin, benzyl benzoate, crotamiton, lindane, and sulfur have also been used. Metronidazole is a common treatment for demodicosis, as was used in our patient for several weeks until the lesions cleared. Systemic metronidazole therapy may be indicated for reducing Demodex spp. density. Severe cases, particularly in immunocompromised individuals, may require oral ivermectin. Appropriate hygiene is important for prevention, such as washing the face with non-oily cleansers and laundering linens regularly.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Mr. Lee is a medical student at the University of California San Diego.
Suggested Reading
Chudzicka-Strugała I et al. Demodicosis in different age groups and alternative treatment options—A review. J Clin Med. 2023 Feb 19;12(4):1649. doi: 10.3390/jcm12041649.
Eichenfield DZ et al. Management of acne vulgaris: A review. JAMA. 2021 Nov 23;326(20):2055-2067. doi: 10.1001/jama.2021.17633.
Sharma A et al. Rosacea management: A comprehensive review. J Cosmet Dermatol. 2022 May;21(5):1895-1904. doi: 10.1111/jocd.14816.
Taudorf EH et al. Cutaneous candidiasis — an evidence-based review of topical and systemic treatments to inform clinical practice. J Eur Acad Dermatol Venereol. 2019 Oct;33(10):1863-1873. doi: 10.1111/jdv.15782.
Winters RD, Mitchell M. Folliculitis. [Updated 2023 Aug 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024. Available from: https://www.ncbi.nlm.nih.gov/books/NBK547754/
Diagnosis
During the visit, skin scrapings were performed, revealing several Demodex mites, confirming the diagnosis of demodicosis.
Demodex folliculorum and Demodex brevis are the two common species implicated. The life cycle of Demodex spp. occurs in the sebaceous glands, leading to mechanical and chemical irritation of the skin.
Various immune responses are also triggered, such as a keratinocyte response via Toll-like receptor 2. Patients usually present with non-specific symptoms such as skin erythema, irritation, peeling, and dryness on the cheeks, eyelids, and paranasal areas. Patients may develop a maculopapular or rosacea-like rash.
Diagnosis is often made through microscopic examination of a skin sample in KOH solution. In rare occasions, a skin surface standardization biopsy method may be used, which determines the density of mites per 1 cm2. Dermoscopy may identify spiky white structures. Molecular methods such as PCR can be used but are not standard.
The differential diagnosis may include acne, rosacea, folliculitis, and Candida infection. Demodicosis can be differentiated by history and further studies including dermoscopy.
Acne vulgaris is an inflammatory disease of the skin’s pilosebaceous unit, primarily involving the face and trunk. It can present with comedones, papules, pustules, and nodules. Secondary signs suggestive of acne vulgaris include scars, erythema, and hyperpigmentation. All forms of acne share a common pathogenesis resulting in the formation of microcomedones, precursors for all clinical acne lesions. In this patient, the absence of microcomedones and the presence of primary inflammatory papules localized to the nose and cheeks suggested an alternative diagnosis.
Rosacea was also considered in the differential diagnosis. Rosacea is an inflammatory dermatosis characterized by erythema, telangiectasia, recurrent flushing, and inflammatory lesions including papulopustules and swelling, primarily affecting the face. The pathogenesis of rosacea is not fully understood but is suggested to involve immune-mediated responses. Vascular dysregulation and reactive oxygen species damage keratinocytes, fibroblasts, and endothelial cells. A higher incidence of rosacea in those with a family history and UV exposure is a known trigger. Demodex folliculorum and Helicobacter pylori are also implicated. Occasionally, Demodex infestation and rosacea may co-occur, and treatment with topical metronidazole can be helpful.
Folliculitis is an infection and inflammation of the hair follicles, forming pustules or erythematous papules over hair-covered skin. It is commonly caused by bacterial infection but can also be due to fungi, viruses, and noninfectious causes such as eosinophilic folliculitis. Diagnosis is clinical, based on physical exam and history, such as recent increased sweating or scratching. KOH prep can be used for Malassezia folliculitis and skin biopsy for eosinophilic folliculitis. Treatment targets the underlying cause. Most bacterial folliculitis cases resolve without treatment, but topical antibiotics may be used. Fungal folliculitis requires oral antifungals, and herpes simplex folliculitis can be treated with antiviral medications.
Cutaneous candidiasis is an infection of the skin by various Candida species, commonly C. albicans. Superficial infections of the skin and mucous membranes, such as intertrigo, are common types. Risk factors include immunosuppression, endocrine disorders, or compromised blood flow. Increased humidity, occlusion, broken skin barriers, and altered skin microbial flora contribute to Candida infection. Diagnosis is clinical but can be confirmed by KOH prep, microscopy, and culture. Treatment involves anti-inflammatory, antibacterial, and antifungal medications. Topical clotrimazole, nystatin, and miconazole are commonly used. Recurrence is prevented by keeping the affected area dry with barrier creams.
Therapeutic goals include arresting mite reproduction, elimination, and preventing recurrent infestations. Treatment may last several months, and the choice of drug depends on patient factors. There have been no standardized treatment studies or long-term effectiveness analyses. Antibiotics such as tetracycline, metronidazole, doxycycline, and ivermectin may be used to prevent proliferation. Permethrin, benzyl benzoate, crotamiton, lindane, and sulfur have also been used. Metronidazole is a common treatment for demodicosis, as was used in our patient for several weeks until the lesions cleared. Systemic metronidazole therapy may be indicated for reducing Demodex spp. density. Severe cases, particularly in immunocompromised individuals, may require oral ivermectin. Appropriate hygiene is important for prevention, such as washing the face with non-oily cleansers and laundering linens regularly.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. Mr. Lee is a medical student at the University of California San Diego.
Suggested Reading
Chudzicka-Strugała I et al. Demodicosis in different age groups and alternative treatment options—A review. J Clin Med. 2023 Feb 19;12(4):1649. doi: 10.3390/jcm12041649.
Eichenfield DZ et al. Management of acne vulgaris: A review. JAMA. 2021 Nov 23;326(20):2055-2067. doi: 10.1001/jama.2021.17633.
Sharma A et al. Rosacea management: A comprehensive review. J Cosmet Dermatol. 2022 May;21(5):1895-1904. doi: 10.1111/jocd.14816.
Taudorf EH et al. Cutaneous candidiasis — an evidence-based review of topical and systemic treatments to inform clinical practice. J Eur Acad Dermatol Venereol. 2019 Oct;33(10):1863-1873. doi: 10.1111/jdv.15782.
Winters RD, Mitchell M. Folliculitis. [Updated 2023 Aug 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024. Available from: https://www.ncbi.nlm.nih.gov/books/NBK547754/
A 7-year-old female presents with persistent pimples on the nose and cheeks for approximately 1 year. She had been treated with several topical antibiotics and acne washes without resolution of the lesions. There were no signs of early puberty, and the child had no history of medical conditions. Her mother has a history of rosacea. Physical examination revealed erythematous papules on the nose and cheeks bilaterally.