User login
Hepatitis screening now for all patients with cancer on therapy
All patients with cancer who are candidates for systemic anticancer therapy should be screened for hepatitis B virus (HBV) infection prior to or at the start of therapy, according to an updated provisional clinical opinion (PCO) from the American Society of Clinical Oncology.
“This is a new approach [that] will actively take system changes ... but it will ultimately be safer for patients – and that is crucial,” commented Jessica P. Hwang, MD, MPH, cochair of the American Society of Clinical Oncology HBV Screening Expert Panel and the first author of the PCO.
Uptake of this universal screening approach would streamline testing protocols and identify more patients at risk for HBV reactivation who should receive prophylactic antiviral therapy, Dr. Hwang said in an interview.
The PCO calls for antiviral prophylaxis during and for at least 12 months after therapy for those with chronic HBV infection who are receiving any systemic anticancer treatment and for those with have had HBV in the past and are receiving any therapies that pose a risk for HBV reactivation.
“Hepatitis B reactivation can cause really terrible outcomes, like organ failure and even death,” Dr. Hwang, who is also a professor at the University of Texas MD Anderson Cancer Center, Houston, commented in an interview.
“This whole [issue of] reactivation and adverse outcomes with anticancer therapies is completely preventable with good planning, good communication, comanagement with specialists, and antiviral therapy and monitoring,” she added.
The updated opinion was published online July 27 in the Journal of Clinical Oncology.
It was developed in response to new data that call into question the previously recommended risk-adaptive approach to HBV screening of cancer patients, say the authors.
ASCO PCOs are developed “to provide timely clinical guidance” on the basis of emerging practice-changing information. This is the second update to follow the initial HBV screening PCO, published in 2010. In the absence of clear consensus because of limited data, the original PCO called for a risk-based approach to screening. A 2015 update extended the recommendation for screening to patients starting anti-CD20 therapy or who are to undergo stem cell transplant and to those with risk factors for HBV exposure.
The current update provides “a clinically pragmatic approach to HBV screening and management” that is based on the latest findings, say the authors. These include findings from a multicenter prospective cohort study of more than 3000 patients. In that study, 21% of patients with chronic HBV had no known risk factors for the infection. In another large prospective observational cohort study, led by Dr. Hwang, which included more than 2100 patients with cancer, 90% had one or more significant risk factors for HBV infection, making selective screening “inefficient and impractical,” she said.
“The results of these two studies suggest that a universal screening approach, its potential harms (e.g., patient and clinician anxiety about management, financial burden associated with antiviral therapy) notwithstanding, is the most efficient, clinically pragmatic approach to HBV screening in persons anticipating systemic anticancer treatment,” the authors comment.
The screening recommended in the PCO requires three tests: hepatitis B surface antigen (HBsAg), core antibody total immunoglobulin or IgG, and antibody to HBsAg tests.
Anticancer therapy should not be delayed pending the results, they write.
Planning for monitoring and long-term prophylaxis for chronic HBV infection should involve a clinician experienced in HBV management, the authors write. Management of those with past infection should be individualized. Alternatively, patients with past infection can be carefully monitored rather than given prophylactic treatment, as long as frequent and consistent follow-up is possible to allow for rapid initiation of antiviral therapy in the event of reactivation, they say.
Hormonal therapy without systemic anticancer therapy is not likely to lead to HBV reactivation in patients with chronic or past infection; antiviral therapy and management of these patients should follow relevant national HBV guidelines, they note.
Challenges in implementing universal HBV screening
The expert panel acknowledges the challenges associated with implementation of universal HBV screening as recommended in their report and notes that electronic health record–based approaches that use alerts to prompt screening have demonstrated success. In one study of high-risk primary care patients, an EHR alert system significantly increased testing rates (odds ratio, 2.64 in comparison with a control group without alerts), and another study that used a simple “sticky-note” alert system to promote referral of HBsAg patients to hepatologists increased referrals from 28% to 73%.
In a cancer population, a “comprehensive set of multimodal interventions,” including pharmacy staff checks for screening prior to anti-CD20 therapy administration and electronic medication order reviews to assess for appropriate testing and treatment before anti-CD20 therapy, increased testing rates to greater than 90% and antiviral prophylaxis rates to more than 80%.
A study of 965 patients in Taiwan showed that a computer-assisted reminder system that prompted for testing prior to ordering anticancer therapy increased screening from 8% to 86% but was less effective for improving the rates of antiviral prophylaxis for those who tested positive for HBV, particularly among physicians treating patients with nonhematologic malignancies.
“Future studies will be needed to make universal HBV screening and linkage to care efficient and systematic, likely based in EHR systems,” the panel says. The authors note that “[o]ngoing studies of HBV tests such as ultrasensitive HBsAg, HBV RNA, and hepatitis B core antigen are being studied and may be useful in predicting risk of HBV reactivation.”
The panel also identified a research gap related to HBV reactivation risks “for the growing list of agents that deplete or modulate B cells.” It notes a need for additional research on the cost-effectiveness of HBV screening. The results of prior cost analyses have been inconsistent and vary with respect to the population studied. For example, universal screening and antiviral prophylaxis approaches have been shown to be cost-effective for patients with hematologic malignancies and high HBV reactivation risk but are less so for patients with solid tumors and lower reactivation risk, they explain.
Dr. Hwang said that not one of the more than 2100 patients in her HBV screening cohort study encountered problems with receiving insurance payment for their HBV screening.
“That’s a really strong statement that insurance payers are accepting of this kind of preventative service,” she said.
Expert panel cochair Andrew Artz, MD, commented that there is now greater acceptance of the need for HBV screening across medical specialties.
“There’s growing consensus among hepatologists, infectious disease specialists, oncologists, and HBV specialists that we need to do a better job of finding patients with hepatitis B [who are] about to receive immunocompromising treatment,” Dr. Artz said in an interview.
Dr. Artz is director of the Program for Aging and Blood Cancers and deputy director of the Center for Cancer and Aging at City of Hope Comprehensive Cancer Center, Duarte, California.
He suggested that the growing acceptance is due in part to the increasing number of anticancer therapies available and the resulting increase in the likelihood of patients receiving therapies that could cause reactivation.
More therapies – and more lines of therapy – could mean greater risk, he explained. He said that testing is easy and that universal screening is the simplest approach to determining who needs it. “There’s no question we will have to change practice,” Dr. Artz said in an interview. “But this is easier than the previous approach that essentially wasn’t being followed because it was too difficult to follow and patients were being missed.”
Most clinicians will appreciate having an approach that’s easier to follow, Dr. Artz predicted.
If there’s a challenge it will be in developing partnerships with HBV specialists, particularly in rural areas. In areas where there is a paucity of subspecialists, oncologists will have to “take some ownership of the issue,” as they often do in such settings, he said.
However, with support from pharmacists, administrators, and others in embracing this guidance, implementation can take place at a systems level rather than an individual clinician level, he added.
The recommendations in this updated PCO were all rated as “strong,” with the exception of the recommendation on hormonal therapy in the absence of systemic anticancer therapy, which was rated as “moderate.” All were based on “informal consensus,” with the exception of the key recommendation for universal HBV screening – use of three specific tests – which was “evidence based.”
The expert panel agreed that the benefits outweigh the harms for each recommendation in the update.
Dr. Hwang received research funding to her institution from Gilead Sciences and Merck Sharp & Dohme. She also has a relationship with the Asian Health Foundation. Dr. Artz received research funding from Miltenyi Biotec. All expert panel members’ disclosures are available in the PCO update.
This article first appeared on Medscape.com.
All patients with cancer who are candidates for systemic anticancer therapy should be screened for hepatitis B virus (HBV) infection prior to or at the start of therapy, according to an updated provisional clinical opinion (PCO) from the American Society of Clinical Oncology.
“This is a new approach [that] will actively take system changes ... but it will ultimately be safer for patients – and that is crucial,” commented Jessica P. Hwang, MD, MPH, cochair of the American Society of Clinical Oncology HBV Screening Expert Panel and the first author of the PCO.
Uptake of this universal screening approach would streamline testing protocols and identify more patients at risk for HBV reactivation who should receive prophylactic antiviral therapy, Dr. Hwang said in an interview.
The PCO calls for antiviral prophylaxis during and for at least 12 months after therapy for those with chronic HBV infection who are receiving any systemic anticancer treatment and for those with have had HBV in the past and are receiving any therapies that pose a risk for HBV reactivation.
“Hepatitis B reactivation can cause really terrible outcomes, like organ failure and even death,” Dr. Hwang, who is also a professor at the University of Texas MD Anderson Cancer Center, Houston, commented in an interview.
“This whole [issue of] reactivation and adverse outcomes with anticancer therapies is completely preventable with good planning, good communication, comanagement with specialists, and antiviral therapy and monitoring,” she added.
The updated opinion was published online July 27 in the Journal of Clinical Oncology.
It was developed in response to new data that call into question the previously recommended risk-adaptive approach to HBV screening of cancer patients, say the authors.
ASCO PCOs are developed “to provide timely clinical guidance” on the basis of emerging practice-changing information. This is the second update to follow the initial HBV screening PCO, published in 2010. In the absence of clear consensus because of limited data, the original PCO called for a risk-based approach to screening. A 2015 update extended the recommendation for screening to patients starting anti-CD20 therapy or who are to undergo stem cell transplant and to those with risk factors for HBV exposure.
The current update provides “a clinically pragmatic approach to HBV screening and management” that is based on the latest findings, say the authors. These include findings from a multicenter prospective cohort study of more than 3000 patients. In that study, 21% of patients with chronic HBV had no known risk factors for the infection. In another large prospective observational cohort study, led by Dr. Hwang, which included more than 2100 patients with cancer, 90% had one or more significant risk factors for HBV infection, making selective screening “inefficient and impractical,” she said.
“The results of these two studies suggest that a universal screening approach, its potential harms (e.g., patient and clinician anxiety about management, financial burden associated with antiviral therapy) notwithstanding, is the most efficient, clinically pragmatic approach to HBV screening in persons anticipating systemic anticancer treatment,” the authors comment.
The screening recommended in the PCO requires three tests: hepatitis B surface antigen (HBsAg), core antibody total immunoglobulin or IgG, and antibody to HBsAg tests.
Anticancer therapy should not be delayed pending the results, they write.
Planning for monitoring and long-term prophylaxis for chronic HBV infection should involve a clinician experienced in HBV management, the authors write. Management of those with past infection should be individualized. Alternatively, patients with past infection can be carefully monitored rather than given prophylactic treatment, as long as frequent and consistent follow-up is possible to allow for rapid initiation of antiviral therapy in the event of reactivation, they say.
Hormonal therapy without systemic anticancer therapy is not likely to lead to HBV reactivation in patients with chronic or past infection; antiviral therapy and management of these patients should follow relevant national HBV guidelines, they note.
Challenges in implementing universal HBV screening
The expert panel acknowledges the challenges associated with implementation of universal HBV screening as recommended in their report and notes that electronic health record–based approaches that use alerts to prompt screening have demonstrated success. In one study of high-risk primary care patients, an EHR alert system significantly increased testing rates (odds ratio, 2.64 in comparison with a control group without alerts), and another study that used a simple “sticky-note” alert system to promote referral of HBsAg patients to hepatologists increased referrals from 28% to 73%.
In a cancer population, a “comprehensive set of multimodal interventions,” including pharmacy staff checks for screening prior to anti-CD20 therapy administration and electronic medication order reviews to assess for appropriate testing and treatment before anti-CD20 therapy, increased testing rates to greater than 90% and antiviral prophylaxis rates to more than 80%.
A study of 965 patients in Taiwan showed that a computer-assisted reminder system that prompted for testing prior to ordering anticancer therapy increased screening from 8% to 86% but was less effective for improving the rates of antiviral prophylaxis for those who tested positive for HBV, particularly among physicians treating patients with nonhematologic malignancies.
“Future studies will be needed to make universal HBV screening and linkage to care efficient and systematic, likely based in EHR systems,” the panel says. The authors note that “[o]ngoing studies of HBV tests such as ultrasensitive HBsAg, HBV RNA, and hepatitis B core antigen are being studied and may be useful in predicting risk of HBV reactivation.”
The panel also identified a research gap related to HBV reactivation risks “for the growing list of agents that deplete or modulate B cells.” It notes a need for additional research on the cost-effectiveness of HBV screening. The results of prior cost analyses have been inconsistent and vary with respect to the population studied. For example, universal screening and antiviral prophylaxis approaches have been shown to be cost-effective for patients with hematologic malignancies and high HBV reactivation risk but are less so for patients with solid tumors and lower reactivation risk, they explain.
Dr. Hwang said that not one of the more than 2100 patients in her HBV screening cohort study encountered problems with receiving insurance payment for their HBV screening.
“That’s a really strong statement that insurance payers are accepting of this kind of preventative service,” she said.
Expert panel cochair Andrew Artz, MD, commented that there is now greater acceptance of the need for HBV screening across medical specialties.
“There’s growing consensus among hepatologists, infectious disease specialists, oncologists, and HBV specialists that we need to do a better job of finding patients with hepatitis B [who are] about to receive immunocompromising treatment,” Dr. Artz said in an interview.
Dr. Artz is director of the Program for Aging and Blood Cancers and deputy director of the Center for Cancer and Aging at City of Hope Comprehensive Cancer Center, Duarte, California.
He suggested that the growing acceptance is due in part to the increasing number of anticancer therapies available and the resulting increase in the likelihood of patients receiving therapies that could cause reactivation.
More therapies – and more lines of therapy – could mean greater risk, he explained. He said that testing is easy and that universal screening is the simplest approach to determining who needs it. “There’s no question we will have to change practice,” Dr. Artz said in an interview. “But this is easier than the previous approach that essentially wasn’t being followed because it was too difficult to follow and patients were being missed.”
Most clinicians will appreciate having an approach that’s easier to follow, Dr. Artz predicted.
If there’s a challenge it will be in developing partnerships with HBV specialists, particularly in rural areas. In areas where there is a paucity of subspecialists, oncologists will have to “take some ownership of the issue,” as they often do in such settings, he said.
However, with support from pharmacists, administrators, and others in embracing this guidance, implementation can take place at a systems level rather than an individual clinician level, he added.
The recommendations in this updated PCO were all rated as “strong,” with the exception of the recommendation on hormonal therapy in the absence of systemic anticancer therapy, which was rated as “moderate.” All were based on “informal consensus,” with the exception of the key recommendation for universal HBV screening – use of three specific tests – which was “evidence based.”
The expert panel agreed that the benefits outweigh the harms for each recommendation in the update.
Dr. Hwang received research funding to her institution from Gilead Sciences and Merck Sharp & Dohme. She also has a relationship with the Asian Health Foundation. Dr. Artz received research funding from Miltenyi Biotec. All expert panel members’ disclosures are available in the PCO update.
This article first appeared on Medscape.com.
All patients with cancer who are candidates for systemic anticancer therapy should be screened for hepatitis B virus (HBV) infection prior to or at the start of therapy, according to an updated provisional clinical opinion (PCO) from the American Society of Clinical Oncology.
“This is a new approach [that] will actively take system changes ... but it will ultimately be safer for patients – and that is crucial,” commented Jessica P. Hwang, MD, MPH, cochair of the American Society of Clinical Oncology HBV Screening Expert Panel and the first author of the PCO.
Uptake of this universal screening approach would streamline testing protocols and identify more patients at risk for HBV reactivation who should receive prophylactic antiviral therapy, Dr. Hwang said in an interview.
The PCO calls for antiviral prophylaxis during and for at least 12 months after therapy for those with chronic HBV infection who are receiving any systemic anticancer treatment and for those with have had HBV in the past and are receiving any therapies that pose a risk for HBV reactivation.
“Hepatitis B reactivation can cause really terrible outcomes, like organ failure and even death,” Dr. Hwang, who is also a professor at the University of Texas MD Anderson Cancer Center, Houston, commented in an interview.
“This whole [issue of] reactivation and adverse outcomes with anticancer therapies is completely preventable with good planning, good communication, comanagement with specialists, and antiviral therapy and monitoring,” she added.
The updated opinion was published online July 27 in the Journal of Clinical Oncology.
It was developed in response to new data that call into question the previously recommended risk-adaptive approach to HBV screening of cancer patients, say the authors.
ASCO PCOs are developed “to provide timely clinical guidance” on the basis of emerging practice-changing information. This is the second update to follow the initial HBV screening PCO, published in 2010. In the absence of clear consensus because of limited data, the original PCO called for a risk-based approach to screening. A 2015 update extended the recommendation for screening to patients starting anti-CD20 therapy or who are to undergo stem cell transplant and to those with risk factors for HBV exposure.
The current update provides “a clinically pragmatic approach to HBV screening and management” that is based on the latest findings, say the authors. These include findings from a multicenter prospective cohort study of more than 3000 patients. In that study, 21% of patients with chronic HBV had no known risk factors for the infection. In another large prospective observational cohort study, led by Dr. Hwang, which included more than 2100 patients with cancer, 90% had one or more significant risk factors for HBV infection, making selective screening “inefficient and impractical,” she said.
“The results of these two studies suggest that a universal screening approach, its potential harms (e.g., patient and clinician anxiety about management, financial burden associated with antiviral therapy) notwithstanding, is the most efficient, clinically pragmatic approach to HBV screening in persons anticipating systemic anticancer treatment,” the authors comment.
The screening recommended in the PCO requires three tests: hepatitis B surface antigen (HBsAg), core antibody total immunoglobulin or IgG, and antibody to HBsAg tests.
Anticancer therapy should not be delayed pending the results, they write.
Planning for monitoring and long-term prophylaxis for chronic HBV infection should involve a clinician experienced in HBV management, the authors write. Management of those with past infection should be individualized. Alternatively, patients with past infection can be carefully monitored rather than given prophylactic treatment, as long as frequent and consistent follow-up is possible to allow for rapid initiation of antiviral therapy in the event of reactivation, they say.
Hormonal therapy without systemic anticancer therapy is not likely to lead to HBV reactivation in patients with chronic or past infection; antiviral therapy and management of these patients should follow relevant national HBV guidelines, they note.
Challenges in implementing universal HBV screening
The expert panel acknowledges the challenges associated with implementation of universal HBV screening as recommended in their report and notes that electronic health record–based approaches that use alerts to prompt screening have demonstrated success. In one study of high-risk primary care patients, an EHR alert system significantly increased testing rates (odds ratio, 2.64 in comparison with a control group without alerts), and another study that used a simple “sticky-note” alert system to promote referral of HBsAg patients to hepatologists increased referrals from 28% to 73%.
In a cancer population, a “comprehensive set of multimodal interventions,” including pharmacy staff checks for screening prior to anti-CD20 therapy administration and electronic medication order reviews to assess for appropriate testing and treatment before anti-CD20 therapy, increased testing rates to greater than 90% and antiviral prophylaxis rates to more than 80%.
A study of 965 patients in Taiwan showed that a computer-assisted reminder system that prompted for testing prior to ordering anticancer therapy increased screening from 8% to 86% but was less effective for improving the rates of antiviral prophylaxis for those who tested positive for HBV, particularly among physicians treating patients with nonhematologic malignancies.
“Future studies will be needed to make universal HBV screening and linkage to care efficient and systematic, likely based in EHR systems,” the panel says. The authors note that “[o]ngoing studies of HBV tests such as ultrasensitive HBsAg, HBV RNA, and hepatitis B core antigen are being studied and may be useful in predicting risk of HBV reactivation.”
The panel also identified a research gap related to HBV reactivation risks “for the growing list of agents that deplete or modulate B cells.” It notes a need for additional research on the cost-effectiveness of HBV screening. The results of prior cost analyses have been inconsistent and vary with respect to the population studied. For example, universal screening and antiviral prophylaxis approaches have been shown to be cost-effective for patients with hematologic malignancies and high HBV reactivation risk but are less so for patients with solid tumors and lower reactivation risk, they explain.
Dr. Hwang said that not one of the more than 2100 patients in her HBV screening cohort study encountered problems with receiving insurance payment for their HBV screening.
“That’s a really strong statement that insurance payers are accepting of this kind of preventative service,” she said.
Expert panel cochair Andrew Artz, MD, commented that there is now greater acceptance of the need for HBV screening across medical specialties.
“There’s growing consensus among hepatologists, infectious disease specialists, oncologists, and HBV specialists that we need to do a better job of finding patients with hepatitis B [who are] about to receive immunocompromising treatment,” Dr. Artz said in an interview.
Dr. Artz is director of the Program for Aging and Blood Cancers and deputy director of the Center for Cancer and Aging at City of Hope Comprehensive Cancer Center, Duarte, California.
He suggested that the growing acceptance is due in part to the increasing number of anticancer therapies available and the resulting increase in the likelihood of patients receiving therapies that could cause reactivation.
More therapies – and more lines of therapy – could mean greater risk, he explained. He said that testing is easy and that universal screening is the simplest approach to determining who needs it. “There’s no question we will have to change practice,” Dr. Artz said in an interview. “But this is easier than the previous approach that essentially wasn’t being followed because it was too difficult to follow and patients were being missed.”
Most clinicians will appreciate having an approach that’s easier to follow, Dr. Artz predicted.
If there’s a challenge it will be in developing partnerships with HBV specialists, particularly in rural areas. In areas where there is a paucity of subspecialists, oncologists will have to “take some ownership of the issue,” as they often do in such settings, he said.
However, with support from pharmacists, administrators, and others in embracing this guidance, implementation can take place at a systems level rather than an individual clinician level, he added.
The recommendations in this updated PCO were all rated as “strong,” with the exception of the recommendation on hormonal therapy in the absence of systemic anticancer therapy, which was rated as “moderate.” All were based on “informal consensus,” with the exception of the key recommendation for universal HBV screening – use of three specific tests – which was “evidence based.”
The expert panel agreed that the benefits outweigh the harms for each recommendation in the update.
Dr. Hwang received research funding to her institution from Gilead Sciences and Merck Sharp & Dohme. She also has a relationship with the Asian Health Foundation. Dr. Artz received research funding from Miltenyi Biotec. All expert panel members’ disclosures are available in the PCO update.
This article first appeared on Medscape.com.
FDA okays new CAR T therapy, first for mantle cell lymphoma
The Food and Drug Administration granted accelerated approval to brexucabtagene autoleucel (Tecartus, Kite Pharma), the first approved chimeric antigen receptor (CAR) T cell therapy for the treatment of adult patients with relapsed or refractory mantle cell lymphoma (MCL).
The new agent is the second approved CAR T cell product developed by Kite and follows the 2017 approval of axicabtagene ciloleucel (Yescarta) for diffuse large B-cell lymphoma.
“Despite promising advances, there are still major gaps in treatment for patients with MCL who progress following initial therapy,” investigator Michael Wang, MD, of the University of Texas MD Anderson Cancer Center in Houston, said in a company statement. “Many patients have high-risk disease and are more likely to keep progressing, even after subsequent treatments.”
In the same press statement, Meghan Gutierrez, chief executive officer, Lymphoma Research Foundation, said: “This approval marks the first CAR T cell therapy approved for mantle cell lymphoma patients and represents a new frontier in the treatment of this disease.”
The approval of the single-infusion therapy is based on efficacy and safety data from the ongoing, single-arm ZUMA-2 pivotal trial, which enrolled 74 adult patients. All patients had previously received anthracycline- or bendamustine-containing chemotherapy, an anti-CD20 antibody therapy and a Bruton tyrosine kinase inhibitor (ibrutinib or acalabrutinib).
In the trial, there was an objective response rate, which was the primary outcome measure, of 87% among 60 patients who were evaluable for efficacy analysis; 62% had a complete response.
Among all patients, follow-up was at least 6 months after their first objective disease response. Median duration of response has not yet been reached.
In terms of adverse events, 18% of the 82 patients evaluable for safety experienced > grade 3 cytokine release syndrome and 37% experienced neurologic events, per the company statement. The most common (≥ 10%) grade 3 or higher adverse reactions were anemia, neutropenia, thrombocytopenia, hypotension, hypophosphatemia, encephalopathy, leukopenia, hypoxia, pyrexia, hyponatremia, hypertension, infection-pathogen unspecified, pneumonia, hypocalcemia, and lymphopenia.
Brexucabtagene autoleucel will be manufactured in Kite’s facility in California. In the pivotal trial, there was a 96% manufacturing success rate and a median manufacturing turnaround time of 15 days from leukapheresis to product delivery.
A version of this article originally appeared on Medscape.com.
The Food and Drug Administration granted accelerated approval to brexucabtagene autoleucel (Tecartus, Kite Pharma), the first approved chimeric antigen receptor (CAR) T cell therapy for the treatment of adult patients with relapsed or refractory mantle cell lymphoma (MCL).
The new agent is the second approved CAR T cell product developed by Kite and follows the 2017 approval of axicabtagene ciloleucel (Yescarta) for diffuse large B-cell lymphoma.
“Despite promising advances, there are still major gaps in treatment for patients with MCL who progress following initial therapy,” investigator Michael Wang, MD, of the University of Texas MD Anderson Cancer Center in Houston, said in a company statement. “Many patients have high-risk disease and are more likely to keep progressing, even after subsequent treatments.”
In the same press statement, Meghan Gutierrez, chief executive officer, Lymphoma Research Foundation, said: “This approval marks the first CAR T cell therapy approved for mantle cell lymphoma patients and represents a new frontier in the treatment of this disease.”
The approval of the single-infusion therapy is based on efficacy and safety data from the ongoing, single-arm ZUMA-2 pivotal trial, which enrolled 74 adult patients. All patients had previously received anthracycline- or bendamustine-containing chemotherapy, an anti-CD20 antibody therapy and a Bruton tyrosine kinase inhibitor (ibrutinib or acalabrutinib).
In the trial, there was an objective response rate, which was the primary outcome measure, of 87% among 60 patients who were evaluable for efficacy analysis; 62% had a complete response.
Among all patients, follow-up was at least 6 months after their first objective disease response. Median duration of response has not yet been reached.
In terms of adverse events, 18% of the 82 patients evaluable for safety experienced > grade 3 cytokine release syndrome and 37% experienced neurologic events, per the company statement. The most common (≥ 10%) grade 3 or higher adverse reactions were anemia, neutropenia, thrombocytopenia, hypotension, hypophosphatemia, encephalopathy, leukopenia, hypoxia, pyrexia, hyponatremia, hypertension, infection-pathogen unspecified, pneumonia, hypocalcemia, and lymphopenia.
Brexucabtagene autoleucel will be manufactured in Kite’s facility in California. In the pivotal trial, there was a 96% manufacturing success rate and a median manufacturing turnaround time of 15 days from leukapheresis to product delivery.
A version of this article originally appeared on Medscape.com.
The Food and Drug Administration granted accelerated approval to brexucabtagene autoleucel (Tecartus, Kite Pharma), the first approved chimeric antigen receptor (CAR) T cell therapy for the treatment of adult patients with relapsed or refractory mantle cell lymphoma (MCL).
The new agent is the second approved CAR T cell product developed by Kite and follows the 2017 approval of axicabtagene ciloleucel (Yescarta) for diffuse large B-cell lymphoma.
“Despite promising advances, there are still major gaps in treatment for patients with MCL who progress following initial therapy,” investigator Michael Wang, MD, of the University of Texas MD Anderson Cancer Center in Houston, said in a company statement. “Many patients have high-risk disease and are more likely to keep progressing, even after subsequent treatments.”
In the same press statement, Meghan Gutierrez, chief executive officer, Lymphoma Research Foundation, said: “This approval marks the first CAR T cell therapy approved for mantle cell lymphoma patients and represents a new frontier in the treatment of this disease.”
The approval of the single-infusion therapy is based on efficacy and safety data from the ongoing, single-arm ZUMA-2 pivotal trial, which enrolled 74 adult patients. All patients had previously received anthracycline- or bendamustine-containing chemotherapy, an anti-CD20 antibody therapy and a Bruton tyrosine kinase inhibitor (ibrutinib or acalabrutinib).
In the trial, there was an objective response rate, which was the primary outcome measure, of 87% among 60 patients who were evaluable for efficacy analysis; 62% had a complete response.
Among all patients, follow-up was at least 6 months after their first objective disease response. Median duration of response has not yet been reached.
In terms of adverse events, 18% of the 82 patients evaluable for safety experienced > grade 3 cytokine release syndrome and 37% experienced neurologic events, per the company statement. The most common (≥ 10%) grade 3 or higher adverse reactions were anemia, neutropenia, thrombocytopenia, hypotension, hypophosphatemia, encephalopathy, leukopenia, hypoxia, pyrexia, hyponatremia, hypertension, infection-pathogen unspecified, pneumonia, hypocalcemia, and lymphopenia.
Brexucabtagene autoleucel will be manufactured in Kite’s facility in California. In the pivotal trial, there was a 96% manufacturing success rate and a median manufacturing turnaround time of 15 days from leukapheresis to product delivery.
A version of this article originally appeared on Medscape.com.
COVID-19: Experts hasten to head off mental health crisis
The COVID-19 pandemic is already affecting mental health at a population level, with increased anxiety, feelings of isolation, and concerns about access to mental health care.
Two U.K. surveys were conducted to inform research priorities for mental health research and in an effort to head off a mental health crisis. The U.K. charity MQ conducted a “stakeholder” survey of 2,198 individuals who had a lived experience of mental illness, while Ipsos MORI conducted a poll of 1,099 members of the public.
The online surveys were conducted in late March, the same week the U.K.’s nationwide lockdown measures were announced. Respondents were asked about their biggest mental health and well-being concerns and coping strategies as they relate to the COVID-19 pandemic.
Results showed that across the two surveys, respondents’ primary concern was anxiety, which was cited in 750 responses.
In addition, respondents were worried about being social isolated, becoming mentally unwell, and having a lack of access to mental health services, as well as the impact of the pandemic on personal relationships.
The findings were used by a panel of experts to inform a position paper published in the Lancet Psychiatry. The paper outlines a proposed government response to curb the long-term “profound” and “pervasive” impact of the pandemic on mental health.
‘Unprecedented response’ needed
“Governments must find evidence-based ways to boost the resilience of our societies and ... to treat those with mental ill health remotely to come out of this pandemic in good mental health,” coauthor of the paper Emily A. Holmes, PhD, of the department of psychology at Uppsala (Sweden) University, said in a press release.
“Frontline medical staff and vulnerable groups such as the elderly and those with serious mental health conditions must be prioritized for rapid mental health support,” she added.
The position paper authors call for “moment-to-moment” monitoring of anxiety, depression, self-harm, and suicide, as well as using digital technology and rapid deployment of evidence-based programs and treatments.
Patients will need to be accessible via computer, cell phone, and other remote technologies in order to receive treatment during physical isolation. However, they noted that there is no “one-size-fits-all” approach, and novel approaches custom tailored to particular populations, including frontline health care workers, are necessary.
“To make a real difference we will need to harness the tools of our digital age, finding smart new ways to measure the mental health of individuals remotely, finding creative ways to boost resilience, and finding ways to treat people in their homes. This effort must be considered central to our global response to the pandemic,” coauthor Ed Bullmore, PhD, of the department of psychiatry at the University of Cambridge (England), said in a statement.
Dr. Bullmore added that it will take “unprecedented research response if we are to limit the negative consequences of this pandemic on the mental health of our society now and in the future.”
Most vulnerable will bear the brunt
During a webinar held to discuss the paper, Matthew Hotopf, PhD, of the Institute of Psychiatry, Psychology, and Neuroscience at King’s College London, cautioned that society’s most vulnerable citizens will bear the brunt of the pandemic’s mental health consequences.
“These individuals often have unstable housing, unstable work, and are disadvantaged in terms of their physical health and their mental health,” with a “very significant gap” in life expectancy versus the rest of the population, he said. The COVID-19 pandemic will widen the gap between “the haves and the have nots.”
“People with established and significant mental disorders are one version of the ‘have nots’ but actually it applies to a lot of people,” said Dr. Hotopf, noting that his experience of lockdown is “very different” from that of someone “living in overcrowded, unstable accommodation, with kids running around and maybe a partner who has problems with anger control.”
The authors of the position paper noted that the COVID-19 pandemic highlights several important research priorities that need to be addressed in the coming weeks and months. These include:
- Understanding the effect of COVID-19 on risk of anxiety, depression, and other outcomes, such as self-harm and suicide
- Understanding how to create physical and social supports to ensure mental health in a climate of physical distancing
- Determining the mental health consequences of social isolation for vulnerable groups, and how can these be mitigated under pandemic conditions
- Understanding the mental health impact of media reporting of COVID-19 in traditional and social media
- Determining the best methods for promoting successful adherence to behavioral advice about COVID-19 while enabling mental well-being and minimizing distress
Another area highlighted by the experts is the potential for neuropsychiatric sequelae in individuals infected with COVID-19. They called for “experimental medicine studies to validate clinical biomarkers and repurpose new treatments for the potentially neurotoxic effects of the virus.”
The authors/investigators disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The COVID-19 pandemic is already affecting mental health at a population level, with increased anxiety, feelings of isolation, and concerns about access to mental health care.
Two U.K. surveys were conducted to inform research priorities for mental health research and in an effort to head off a mental health crisis. The U.K. charity MQ conducted a “stakeholder” survey of 2,198 individuals who had a lived experience of mental illness, while Ipsos MORI conducted a poll of 1,099 members of the public.
The online surveys were conducted in late March, the same week the U.K.’s nationwide lockdown measures were announced. Respondents were asked about their biggest mental health and well-being concerns and coping strategies as they relate to the COVID-19 pandemic.
Results showed that across the two surveys, respondents’ primary concern was anxiety, which was cited in 750 responses.
In addition, respondents were worried about being social isolated, becoming mentally unwell, and having a lack of access to mental health services, as well as the impact of the pandemic on personal relationships.
The findings were used by a panel of experts to inform a position paper published in the Lancet Psychiatry. The paper outlines a proposed government response to curb the long-term “profound” and “pervasive” impact of the pandemic on mental health.
‘Unprecedented response’ needed
“Governments must find evidence-based ways to boost the resilience of our societies and ... to treat those with mental ill health remotely to come out of this pandemic in good mental health,” coauthor of the paper Emily A. Holmes, PhD, of the department of psychology at Uppsala (Sweden) University, said in a press release.
“Frontline medical staff and vulnerable groups such as the elderly and those with serious mental health conditions must be prioritized for rapid mental health support,” she added.
The position paper authors call for “moment-to-moment” monitoring of anxiety, depression, self-harm, and suicide, as well as using digital technology and rapid deployment of evidence-based programs and treatments.
Patients will need to be accessible via computer, cell phone, and other remote technologies in order to receive treatment during physical isolation. However, they noted that there is no “one-size-fits-all” approach, and novel approaches custom tailored to particular populations, including frontline health care workers, are necessary.
“To make a real difference we will need to harness the tools of our digital age, finding smart new ways to measure the mental health of individuals remotely, finding creative ways to boost resilience, and finding ways to treat people in their homes. This effort must be considered central to our global response to the pandemic,” coauthor Ed Bullmore, PhD, of the department of psychiatry at the University of Cambridge (England), said in a statement.
Dr. Bullmore added that it will take “unprecedented research response if we are to limit the negative consequences of this pandemic on the mental health of our society now and in the future.”
Most vulnerable will bear the brunt
During a webinar held to discuss the paper, Matthew Hotopf, PhD, of the Institute of Psychiatry, Psychology, and Neuroscience at King’s College London, cautioned that society’s most vulnerable citizens will bear the brunt of the pandemic’s mental health consequences.
“These individuals often have unstable housing, unstable work, and are disadvantaged in terms of their physical health and their mental health,” with a “very significant gap” in life expectancy versus the rest of the population, he said. The COVID-19 pandemic will widen the gap between “the haves and the have nots.”
“People with established and significant mental disorders are one version of the ‘have nots’ but actually it applies to a lot of people,” said Dr. Hotopf, noting that his experience of lockdown is “very different” from that of someone “living in overcrowded, unstable accommodation, with kids running around and maybe a partner who has problems with anger control.”
The authors of the position paper noted that the COVID-19 pandemic highlights several important research priorities that need to be addressed in the coming weeks and months. These include:
- Understanding the effect of COVID-19 on risk of anxiety, depression, and other outcomes, such as self-harm and suicide
- Understanding how to create physical and social supports to ensure mental health in a climate of physical distancing
- Determining the mental health consequences of social isolation for vulnerable groups, and how can these be mitigated under pandemic conditions
- Understanding the mental health impact of media reporting of COVID-19 in traditional and social media
- Determining the best methods for promoting successful adherence to behavioral advice about COVID-19 while enabling mental well-being and minimizing distress
Another area highlighted by the experts is the potential for neuropsychiatric sequelae in individuals infected with COVID-19. They called for “experimental medicine studies to validate clinical biomarkers and repurpose new treatments for the potentially neurotoxic effects of the virus.”
The authors/investigators disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The COVID-19 pandemic is already affecting mental health at a population level, with increased anxiety, feelings of isolation, and concerns about access to mental health care.
Two U.K. surveys were conducted to inform research priorities for mental health research and in an effort to head off a mental health crisis. The U.K. charity MQ conducted a “stakeholder” survey of 2,198 individuals who had a lived experience of mental illness, while Ipsos MORI conducted a poll of 1,099 members of the public.
The online surveys were conducted in late March, the same week the U.K.’s nationwide lockdown measures were announced. Respondents were asked about their biggest mental health and well-being concerns and coping strategies as they relate to the COVID-19 pandemic.
Results showed that across the two surveys, respondents’ primary concern was anxiety, which was cited in 750 responses.
In addition, respondents were worried about being social isolated, becoming mentally unwell, and having a lack of access to mental health services, as well as the impact of the pandemic on personal relationships.
The findings were used by a panel of experts to inform a position paper published in the Lancet Psychiatry. The paper outlines a proposed government response to curb the long-term “profound” and “pervasive” impact of the pandemic on mental health.
‘Unprecedented response’ needed
“Governments must find evidence-based ways to boost the resilience of our societies and ... to treat those with mental ill health remotely to come out of this pandemic in good mental health,” coauthor of the paper Emily A. Holmes, PhD, of the department of psychology at Uppsala (Sweden) University, said in a press release.
“Frontline medical staff and vulnerable groups such as the elderly and those with serious mental health conditions must be prioritized for rapid mental health support,” she added.
The position paper authors call for “moment-to-moment” monitoring of anxiety, depression, self-harm, and suicide, as well as using digital technology and rapid deployment of evidence-based programs and treatments.
Patients will need to be accessible via computer, cell phone, and other remote technologies in order to receive treatment during physical isolation. However, they noted that there is no “one-size-fits-all” approach, and novel approaches custom tailored to particular populations, including frontline health care workers, are necessary.
“To make a real difference we will need to harness the tools of our digital age, finding smart new ways to measure the mental health of individuals remotely, finding creative ways to boost resilience, and finding ways to treat people in their homes. This effort must be considered central to our global response to the pandemic,” coauthor Ed Bullmore, PhD, of the department of psychiatry at the University of Cambridge (England), said in a statement.
Dr. Bullmore added that it will take “unprecedented research response if we are to limit the negative consequences of this pandemic on the mental health of our society now and in the future.”
Most vulnerable will bear the brunt
During a webinar held to discuss the paper, Matthew Hotopf, PhD, of the Institute of Psychiatry, Psychology, and Neuroscience at King’s College London, cautioned that society’s most vulnerable citizens will bear the brunt of the pandemic’s mental health consequences.
“These individuals often have unstable housing, unstable work, and are disadvantaged in terms of their physical health and their mental health,” with a “very significant gap” in life expectancy versus the rest of the population, he said. The COVID-19 pandemic will widen the gap between “the haves and the have nots.”
“People with established and significant mental disorders are one version of the ‘have nots’ but actually it applies to a lot of people,” said Dr. Hotopf, noting that his experience of lockdown is “very different” from that of someone “living in overcrowded, unstable accommodation, with kids running around and maybe a partner who has problems with anger control.”
The authors of the position paper noted that the COVID-19 pandemic highlights several important research priorities that need to be addressed in the coming weeks and months. These include:
- Understanding the effect of COVID-19 on risk of anxiety, depression, and other outcomes, such as self-harm and suicide
- Understanding how to create physical and social supports to ensure mental health in a climate of physical distancing
- Determining the mental health consequences of social isolation for vulnerable groups, and how can these be mitigated under pandemic conditions
- Understanding the mental health impact of media reporting of COVID-19 in traditional and social media
- Determining the best methods for promoting successful adherence to behavioral advice about COVID-19 while enabling mental well-being and minimizing distress
Another area highlighted by the experts is the potential for neuropsychiatric sequelae in individuals infected with COVID-19. They called for “experimental medicine studies to validate clinical biomarkers and repurpose new treatments for the potentially neurotoxic effects of the virus.”
The authors/investigators disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Experts break down latest CAR T-cell advances in lymphoma
ORLANDO – There’s now mature data surrounding the use of chimeric antigen receptor (CAR) T-cell therapy in lymphoma, and the annual meeting of the American Society of Hematology brought forth additional information from real-world studies, insights about what is driving relapse, and promising data on mantle cell lymphoma.

The roundtable participants included Brian Hill, MD, of the Cleveland Clinic Taussig Cancer Center; Frederick L. Locke, MD, of the Moffit Cancer Center in Tampa, Fla.; and Peter Riedell, MD, of the University of Chicago.
Among the studies highlighted by the panel was the Transcend NHL 001 study (Abstract 241), which looked at third-line use of lisocabtagene maraleucel (liso-cel) in patients with diffuse large B-cell lymphoma, transformed follicular lymphoma, and other indolent non-Hodgkin lymphoma subtypes. More than 300 patients were enrolled, and liso-cel met all primary and secondary efficacy endpoints, with an overall response rate of more than 70%. The notable take-home point from the study was the safety profile, Dr. Riedell noted. Liso-cel was associated with a lower rate of cytokine release syndrome and neurologic toxicity, compared with the currently approved products.
Since patients in the study had a lower incidence and later onset of cytokine release syndrome, liso-cel could be a candidate for outpatient administration, Dr. Locke said. However, doing that would require “significant infrastructure” in hospitals and clinics to properly support patients, especially given that the treatment-related mortality on the study was similar to approved CAR T-cell products at about 3%. “You have to be ready to admit the patient to the hospital very rapidly, and you have to have the providers and the nurses who are vigilant when the patient is not in the hospital,” he said.
Another notable study presented at ASH examined the characteristics and outcomes of patients receiving bridging therapy while awaiting treatment with axicabtagene ciloleucel (Abstract 245). This real-world study adds interesting information to the field because, in some of the studies that were pivotal to the approval of CAR T-cell therapy, bridging therapy was not allowed, Dr. Locke said.
In this analysis, researchers found that the overall survival was worse among patients who received bridging. This finding suggests that patients who received bridging therapy had a different biology or that the therapy itself may have had an effect on the host or tumor microenvironment that affected the efficacy of the CAR T-cell therapy, the researchers reported.
The panel also highlighted the Zuma-2 study, which looked at KTE-X19, an anti-CD19 CAR T-cell therapy, among more than 70 patients with relapsed/refractory mantle cell lymphoma who had failed treatment with a Bruton’s tyrosine kinase inhibitor (Abstract 754). “This was, I thought, kind of a sleeper study at ASH,” said Dr. Hill, who was one of the authors of the study.
The overall response rate was 93% with about two-thirds of patients achieving a complete response. Researchers found that the response was consistent across subgroups, including Ki-67 and patients with prior use of steroids or bridging therapy. Dr. Locke, who was also a study author, said the results are a “game changer.”
“I’m very excited about it,” Dr. Riedell said, noting that these are patients without a lot of treatment options.
The panel also discussed other studies from ASH, including an analysis of tumor tissue samples from patients in the ZUMA-1 trial who had responded and subsequently relapsed (Abstract 203); a multicenter prospective analysis of circulating tumor DNA in diffuse large B-cell lymphoma patients who had relapsed after treatment with axicabtagene ciloleucel (Abstract 884); and the early use of corticosteroids to prevent toxicities in patients in cohort 4 of the ZUMA-1 trial (Abstract 243).
Dr. Hill reported consulting with Juno/Celgene/BMS and Novartis and research and consulting for Kite/Gilead. Dr. Locke reported consulting for Cellular Biomedicine Group and being a scientific adviser to Kite/Gilead, Novartis, Celgene/BMS, GammaDelta Therapeutics, Calibr, and Allogene. Dr. Riedell reported consulting for Bayer and Verastem, consulting for and research funding from Novartis and BMS/Celgene, and consulting for, research funding from, and speaking for Kite.
ORLANDO – There’s now mature data surrounding the use of chimeric antigen receptor (CAR) T-cell therapy in lymphoma, and the annual meeting of the American Society of Hematology brought forth additional information from real-world studies, insights about what is driving relapse, and promising data on mantle cell lymphoma.

The roundtable participants included Brian Hill, MD, of the Cleveland Clinic Taussig Cancer Center; Frederick L. Locke, MD, of the Moffit Cancer Center in Tampa, Fla.; and Peter Riedell, MD, of the University of Chicago.
Among the studies highlighted by the panel was the Transcend NHL 001 study (Abstract 241), which looked at third-line use of lisocabtagene maraleucel (liso-cel) in patients with diffuse large B-cell lymphoma, transformed follicular lymphoma, and other indolent non-Hodgkin lymphoma subtypes. More than 300 patients were enrolled, and liso-cel met all primary and secondary efficacy endpoints, with an overall response rate of more than 70%. The notable take-home point from the study was the safety profile, Dr. Riedell noted. Liso-cel was associated with a lower rate of cytokine release syndrome and neurologic toxicity, compared with the currently approved products.
Since patients in the study had a lower incidence and later onset of cytokine release syndrome, liso-cel could be a candidate for outpatient administration, Dr. Locke said. However, doing that would require “significant infrastructure” in hospitals and clinics to properly support patients, especially given that the treatment-related mortality on the study was similar to approved CAR T-cell products at about 3%. “You have to be ready to admit the patient to the hospital very rapidly, and you have to have the providers and the nurses who are vigilant when the patient is not in the hospital,” he said.
Another notable study presented at ASH examined the characteristics and outcomes of patients receiving bridging therapy while awaiting treatment with axicabtagene ciloleucel (Abstract 245). This real-world study adds interesting information to the field because, in some of the studies that were pivotal to the approval of CAR T-cell therapy, bridging therapy was not allowed, Dr. Locke said.
In this analysis, researchers found that the overall survival was worse among patients who received bridging. This finding suggests that patients who received bridging therapy had a different biology or that the therapy itself may have had an effect on the host or tumor microenvironment that affected the efficacy of the CAR T-cell therapy, the researchers reported.
The panel also highlighted the Zuma-2 study, which looked at KTE-X19, an anti-CD19 CAR T-cell therapy, among more than 70 patients with relapsed/refractory mantle cell lymphoma who had failed treatment with a Bruton’s tyrosine kinase inhibitor (Abstract 754). “This was, I thought, kind of a sleeper study at ASH,” said Dr. Hill, who was one of the authors of the study.
The overall response rate was 93% with about two-thirds of patients achieving a complete response. Researchers found that the response was consistent across subgroups, including Ki-67 and patients with prior use of steroids or bridging therapy. Dr. Locke, who was also a study author, said the results are a “game changer.”
“I’m very excited about it,” Dr. Riedell said, noting that these are patients without a lot of treatment options.
The panel also discussed other studies from ASH, including an analysis of tumor tissue samples from patients in the ZUMA-1 trial who had responded and subsequently relapsed (Abstract 203); a multicenter prospective analysis of circulating tumor DNA in diffuse large B-cell lymphoma patients who had relapsed after treatment with axicabtagene ciloleucel (Abstract 884); and the early use of corticosteroids to prevent toxicities in patients in cohort 4 of the ZUMA-1 trial (Abstract 243).
Dr. Hill reported consulting with Juno/Celgene/BMS and Novartis and research and consulting for Kite/Gilead. Dr. Locke reported consulting for Cellular Biomedicine Group and being a scientific adviser to Kite/Gilead, Novartis, Celgene/BMS, GammaDelta Therapeutics, Calibr, and Allogene. Dr. Riedell reported consulting for Bayer and Verastem, consulting for and research funding from Novartis and BMS/Celgene, and consulting for, research funding from, and speaking for Kite.
ORLANDO – There’s now mature data surrounding the use of chimeric antigen receptor (CAR) T-cell therapy in lymphoma, and the annual meeting of the American Society of Hematology brought forth additional information from real-world studies, insights about what is driving relapse, and promising data on mantle cell lymphoma.

The roundtable participants included Brian Hill, MD, of the Cleveland Clinic Taussig Cancer Center; Frederick L. Locke, MD, of the Moffit Cancer Center in Tampa, Fla.; and Peter Riedell, MD, of the University of Chicago.
Among the studies highlighted by the panel was the Transcend NHL 001 study (Abstract 241), which looked at third-line use of lisocabtagene maraleucel (liso-cel) in patients with diffuse large B-cell lymphoma, transformed follicular lymphoma, and other indolent non-Hodgkin lymphoma subtypes. More than 300 patients were enrolled, and liso-cel met all primary and secondary efficacy endpoints, with an overall response rate of more than 70%. The notable take-home point from the study was the safety profile, Dr. Riedell noted. Liso-cel was associated with a lower rate of cytokine release syndrome and neurologic toxicity, compared with the currently approved products.
Since patients in the study had a lower incidence and later onset of cytokine release syndrome, liso-cel could be a candidate for outpatient administration, Dr. Locke said. However, doing that would require “significant infrastructure” in hospitals and clinics to properly support patients, especially given that the treatment-related mortality on the study was similar to approved CAR T-cell products at about 3%. “You have to be ready to admit the patient to the hospital very rapidly, and you have to have the providers and the nurses who are vigilant when the patient is not in the hospital,” he said.
Another notable study presented at ASH examined the characteristics and outcomes of patients receiving bridging therapy while awaiting treatment with axicabtagene ciloleucel (Abstract 245). This real-world study adds interesting information to the field because, in some of the studies that were pivotal to the approval of CAR T-cell therapy, bridging therapy was not allowed, Dr. Locke said.
In this analysis, researchers found that the overall survival was worse among patients who received bridging. This finding suggests that patients who received bridging therapy had a different biology or that the therapy itself may have had an effect on the host or tumor microenvironment that affected the efficacy of the CAR T-cell therapy, the researchers reported.
The panel also highlighted the Zuma-2 study, which looked at KTE-X19, an anti-CD19 CAR T-cell therapy, among more than 70 patients with relapsed/refractory mantle cell lymphoma who had failed treatment with a Bruton’s tyrosine kinase inhibitor (Abstract 754). “This was, I thought, kind of a sleeper study at ASH,” said Dr. Hill, who was one of the authors of the study.
The overall response rate was 93% with about two-thirds of patients achieving a complete response. Researchers found that the response was consistent across subgroups, including Ki-67 and patients with prior use of steroids or bridging therapy. Dr. Locke, who was also a study author, said the results are a “game changer.”
“I’m very excited about it,” Dr. Riedell said, noting that these are patients without a lot of treatment options.
The panel also discussed other studies from ASH, including an analysis of tumor tissue samples from patients in the ZUMA-1 trial who had responded and subsequently relapsed (Abstract 203); a multicenter prospective analysis of circulating tumor DNA in diffuse large B-cell lymphoma patients who had relapsed after treatment with axicabtagene ciloleucel (Abstract 884); and the early use of corticosteroids to prevent toxicities in patients in cohort 4 of the ZUMA-1 trial (Abstract 243).
Dr. Hill reported consulting with Juno/Celgene/BMS and Novartis and research and consulting for Kite/Gilead. Dr. Locke reported consulting for Cellular Biomedicine Group and being a scientific adviser to Kite/Gilead, Novartis, Celgene/BMS, GammaDelta Therapeutics, Calibr, and Allogene. Dr. Riedell reported consulting for Bayer and Verastem, consulting for and research funding from Novartis and BMS/Celgene, and consulting for, research funding from, and speaking for Kite.
EXPERT ANALYSIS FROM ASH 2019
LOXO-305: Next-gen BTK inhibitor safe and effective in B-cell malignancies
ORLANDO – A phase 1 trial of the next-generation Bruton tyrosine kinase (BTK) inhibitor LOXO-305 has demonstrated safety and provided evidence of its efficacy in heavily pretreated patients with B-cell malignancies, including some with acquired resistance to other BTK inhibitors and venetoclax, according to an investigator.
The antitumor activity of this highly selective investigational oral BTK inhibitor was significant in patients with chronic lymphocytic leukemia (CLL) and mantle cell lymphoma (MCL), with a rapid onset of action and resolution of lymphocytosis “consistent with effective BTK target inhibition,” said Anthony R. Mato, MD, of the Center for CLL at Memorial Sloan Kettering Cancer Center in New York.
Responses were also seen in patients with BTK C481 mutations, the primary cause of progressive CLL after BTK inhibitor use, Dr. Mato said at the annual meeting of the American Society of Hematology.
The safety and tolerability profile of LOXO-305 is “consistent with highly selective drug design,” with no evidence of off-target effects, he said. “Collectively, these data demonstrate that BTK remains a highly actionable target despite progression on covalent BTK inhibitors.”
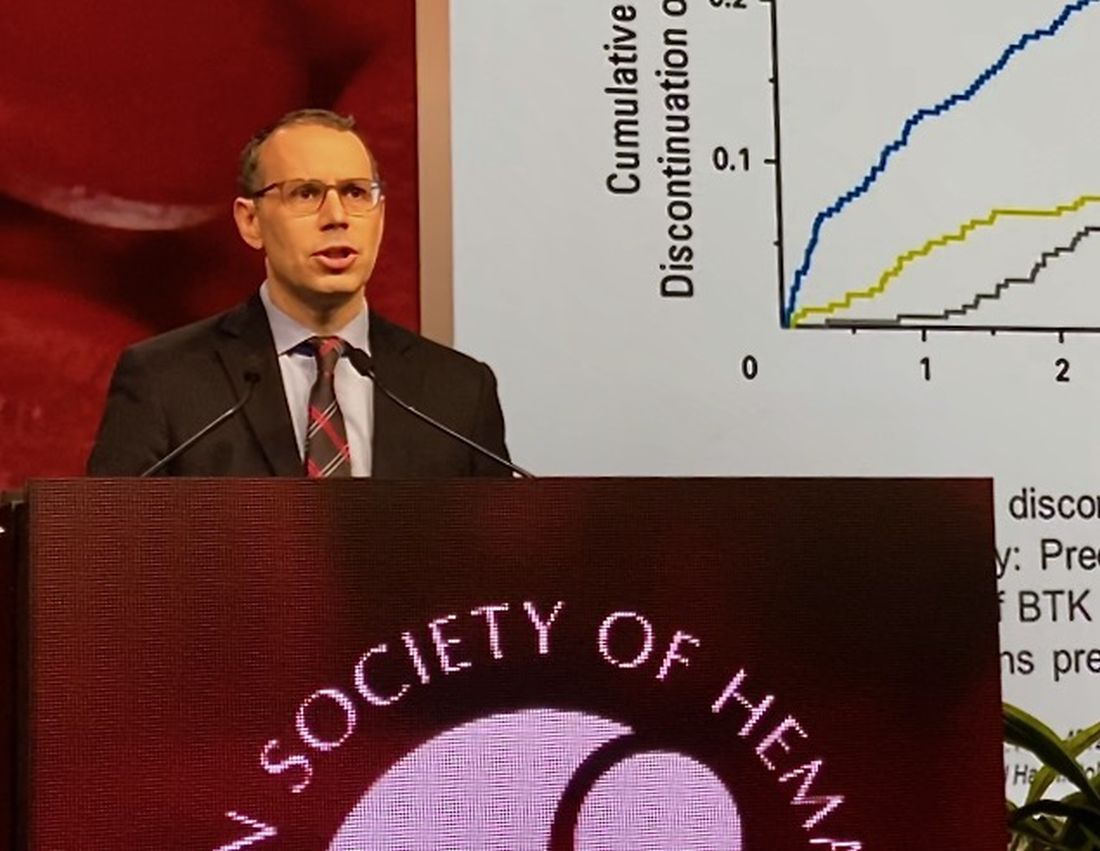
While BTK inhibitors have transformed treatment of B-cell malignancies, resistance remains a major problem, said Dr. Mato, citing 5-year ibrutinib discontinuation rates of 41% in the front line setting and 53.7% in the relapsed/refractory setting.
Key reasons for discontinuation are intolerance, events such as atrial fibrillation and major bleeding, progression of disease, and the appearance of BTK C481 mutations, which prevent covalent BTK inhibitors from achieving effective target inhibition, he said. In contrast, LOXO-305 is designed to non-covalently bind to BTK, regardless of C481 status.
Dr. Mato described results of the phase 1 BRUIN trial, in which 28 adult patients with CLL or B-cell non-Hodgkin lymphomas received once daily oral LOXO-305 at doses ranging from 25 mg to 200 mg. All patients had received at least two lines of prior therapy and had active disease in need of treatment.
For 13 evaluable CLL patients, the overall response rate was 77% (10 patients), Dr. Mato reported. Overall response rates for MCL and other B-cell malignancies were 50%, or three out of six MCL patients and two of four patients with Waldenström macroglobulinemia, diffuse large B-cell lymphoma, or marginal zone lymphoma.
Though only a small subset of CLL patients have had multiple response assessments, the available data suggest that responses “deepen over time” with continued LOXO-305 treatment, Dr. Mato said.
With the median follow-up of 2.7 months, 24 of 28 patients remain on therapy, including all responders. “Some of the responses appear to be quite durable,” Dr. Mato said.
There have been no dose-limiting toxicities, the maximum tolerated dose has not been reached, and there have been no notable adverse events characteristic of covalent BTK inhibitors – namely atrial fibrillation or major bleeding – despite frequent monitoring, according to Dr. Mato.
There were two grade 3 events (leukocytosis and neutropenia), but the remaining treatment-emergent adverse events have been grade 1-2. “Having managed many of these patients, I can tell you that these adverse events were quite manageable,” Dr. Mato told attendees.
BRUIN is a global trial that continues to enroll patients at 18 sites in 3 countries, with a plan in 2020 to incorporate “rational combinations” of agents, according to the investigator.
Dr. Mato reported disclosures related to Gilead, Pharmacyclics, AstraZeneca, AbbVie, Sunesis, Johnson & Johnson, TG Therapeutics, LOXO, DTRM Biopharma, Genentech, Janssen, Acerta, Pharmacyclics, and Celgene.
SOURCE: Mato AR et al. ASH 2019, Abstract 501.
ORLANDO – A phase 1 trial of the next-generation Bruton tyrosine kinase (BTK) inhibitor LOXO-305 has demonstrated safety and provided evidence of its efficacy in heavily pretreated patients with B-cell malignancies, including some with acquired resistance to other BTK inhibitors and venetoclax, according to an investigator.
The antitumor activity of this highly selective investigational oral BTK inhibitor was significant in patients with chronic lymphocytic leukemia (CLL) and mantle cell lymphoma (MCL), with a rapid onset of action and resolution of lymphocytosis “consistent with effective BTK target inhibition,” said Anthony R. Mato, MD, of the Center for CLL at Memorial Sloan Kettering Cancer Center in New York.
Responses were also seen in patients with BTK C481 mutations, the primary cause of progressive CLL after BTK inhibitor use, Dr. Mato said at the annual meeting of the American Society of Hematology.
The safety and tolerability profile of LOXO-305 is “consistent with highly selective drug design,” with no evidence of off-target effects, he said. “Collectively, these data demonstrate that BTK remains a highly actionable target despite progression on covalent BTK inhibitors.”
While BTK inhibitors have transformed treatment of B-cell malignancies, resistance remains a major problem, said Dr. Mato, citing 5-year ibrutinib discontinuation rates of 41% in the front line setting and 53.7% in the relapsed/refractory setting.
Key reasons for discontinuation are intolerance, events such as atrial fibrillation and major bleeding, progression of disease, and the appearance of BTK C481 mutations, which prevent covalent BTK inhibitors from achieving effective target inhibition, he said. In contrast, LOXO-305 is designed to non-covalently bind to BTK, regardless of C481 status.
Dr. Mato described results of the phase 1 BRUIN trial, in which 28 adult patients with CLL or B-cell non-Hodgkin lymphomas received once daily oral LOXO-305 at doses ranging from 25 mg to 200 mg. All patients had received at least two lines of prior therapy and had active disease in need of treatment.
For 13 evaluable CLL patients, the overall response rate was 77% (10 patients), Dr. Mato reported. Overall response rates for MCL and other B-cell malignancies were 50%, or three out of six MCL patients and two of four patients with Waldenström macroglobulinemia, diffuse large B-cell lymphoma, or marginal zone lymphoma.
Though only a small subset of CLL patients have had multiple response assessments, the available data suggest that responses “deepen over time” with continued LOXO-305 treatment, Dr. Mato said.
With the median follow-up of 2.7 months, 24 of 28 patients remain on therapy, including all responders. “Some of the responses appear to be quite durable,” Dr. Mato said.
There have been no dose-limiting toxicities, the maximum tolerated dose has not been reached, and there have been no notable adverse events characteristic of covalent BTK inhibitors – namely atrial fibrillation or major bleeding – despite frequent monitoring, according to Dr. Mato.
There were two grade 3 events (leukocytosis and neutropenia), but the remaining treatment-emergent adverse events have been grade 1-2. “Having managed many of these patients, I can tell you that these adverse events were quite manageable,” Dr. Mato told attendees.
BRUIN is a global trial that continues to enroll patients at 18 sites in 3 countries, with a plan in 2020 to incorporate “rational combinations” of agents, according to the investigator.
Dr. Mato reported disclosures related to Gilead, Pharmacyclics, AstraZeneca, AbbVie, Sunesis, Johnson & Johnson, TG Therapeutics, LOXO, DTRM Biopharma, Genentech, Janssen, Acerta, Pharmacyclics, and Celgene.
SOURCE: Mato AR et al. ASH 2019, Abstract 501.
ORLANDO – A phase 1 trial of the next-generation Bruton tyrosine kinase (BTK) inhibitor LOXO-305 has demonstrated safety and provided evidence of its efficacy in heavily pretreated patients with B-cell malignancies, including some with acquired resistance to other BTK inhibitors and venetoclax, according to an investigator.
The antitumor activity of this highly selective investigational oral BTK inhibitor was significant in patients with chronic lymphocytic leukemia (CLL) and mantle cell lymphoma (MCL), with a rapid onset of action and resolution of lymphocytosis “consistent with effective BTK target inhibition,” said Anthony R. Mato, MD, of the Center for CLL at Memorial Sloan Kettering Cancer Center in New York.
Responses were also seen in patients with BTK C481 mutations, the primary cause of progressive CLL after BTK inhibitor use, Dr. Mato said at the annual meeting of the American Society of Hematology.
The safety and tolerability profile of LOXO-305 is “consistent with highly selective drug design,” with no evidence of off-target effects, he said. “Collectively, these data demonstrate that BTK remains a highly actionable target despite progression on covalent BTK inhibitors.”
While BTK inhibitors have transformed treatment of B-cell malignancies, resistance remains a major problem, said Dr. Mato, citing 5-year ibrutinib discontinuation rates of 41% in the front line setting and 53.7% in the relapsed/refractory setting.
Key reasons for discontinuation are intolerance, events such as atrial fibrillation and major bleeding, progression of disease, and the appearance of BTK C481 mutations, which prevent covalent BTK inhibitors from achieving effective target inhibition, he said. In contrast, LOXO-305 is designed to non-covalently bind to BTK, regardless of C481 status.
Dr. Mato described results of the phase 1 BRUIN trial, in which 28 adult patients with CLL or B-cell non-Hodgkin lymphomas received once daily oral LOXO-305 at doses ranging from 25 mg to 200 mg. All patients had received at least two lines of prior therapy and had active disease in need of treatment.
For 13 evaluable CLL patients, the overall response rate was 77% (10 patients), Dr. Mato reported. Overall response rates for MCL and other B-cell malignancies were 50%, or three out of six MCL patients and two of four patients with Waldenström macroglobulinemia, diffuse large B-cell lymphoma, or marginal zone lymphoma.
Though only a small subset of CLL patients have had multiple response assessments, the available data suggest that responses “deepen over time” with continued LOXO-305 treatment, Dr. Mato said.
With the median follow-up of 2.7 months, 24 of 28 patients remain on therapy, including all responders. “Some of the responses appear to be quite durable,” Dr. Mato said.
There have been no dose-limiting toxicities, the maximum tolerated dose has not been reached, and there have been no notable adverse events characteristic of covalent BTK inhibitors – namely atrial fibrillation or major bleeding – despite frequent monitoring, according to Dr. Mato.
There were two grade 3 events (leukocytosis and neutropenia), but the remaining treatment-emergent adverse events have been grade 1-2. “Having managed many of these patients, I can tell you that these adverse events were quite manageable,” Dr. Mato told attendees.
BRUIN is a global trial that continues to enroll patients at 18 sites in 3 countries, with a plan in 2020 to incorporate “rational combinations” of agents, according to the investigator.
Dr. Mato reported disclosures related to Gilead, Pharmacyclics, AstraZeneca, AbbVie, Sunesis, Johnson & Johnson, TG Therapeutics, LOXO, DTRM Biopharma, Genentech, Janssen, Acerta, Pharmacyclics, and Celgene.
SOURCE: Mato AR et al. ASH 2019, Abstract 501.
REPORTING FROM ASH 2019
Some MCL patients can safely stop venetoclax-ibrutinib, study suggests
ORLANDO – Updated trial results have revealed durable responses with venetoclax and ibrutinib in patients with mantle cell lymphoma (MCL), allowing some patients to stop treatment.
Five of 24 patients were able to stop treatment after achieving minimal residual disease (MRD)-negative complete responses (CRs). Four of these patients remain in CR at up to 18 months off treatment, although one patient ultimately progressed and died.
“Treatment cessation was feasible for patients in MRD-negative complete responses, raising the prospect of limited-duration, targeted-agent therapy in the management of relapsed/refractory mantle cell lymphoma,” said Sasanka M. Handunnetti, MBBS, of Peter MacCallum Cancer Centre in Melbourne. Dr. Handunnetti presented these results, from the AIM trial, at the annual meeting of the American Society of Hematology.
The phase 2 trial enrolled 24 patients. At baseline, patients had a median age of 68 years (range, 47-81 years), and 88% were men. One patient was treatment-naive, but the rest had relapsed/refractory MCL. These patients had received a median of two prior therapies (range, 1-6).
The patients received venetoclax at 400 mg daily and ibrutinib at 560 mg daily.
In the primary analysis, the CR rate was 62% at week 16 and 71% overall, according to positron-emission tomography/computed tomography. MRD negativity was achieved by 67% of patients according to flow cytometry and 38% according to allele-specific oligonucleotide polymerase chain reaction (N Engl J Med. 2018 Mar 29;378[13]:1211-23).
Response and survival
For the current analysis, the median follow up was 37.5 months (range, 1.4-45.3 months). The median duration of response has not been reached, the median progression-free survival is 29 months, and the median overall survival is 32 months.
Thirteen patients have died, 8 of them due to progressive disease. The remaining 11 patients are still alive, and 9 of them are still in CR. One patient is still in partial response, and one has not responded but remains on ibrutinib and venetoclax.
Dr. Handunnetti pointed out that 12 patients had TP53 aberrations, and 8 of them died, but 4 remain alive and in CR. All four patients with SMARCA4 aberrations died.
Treatment status
Five patients are still receiving treatment with ibrutinib and venetoclax, and one patient is receiving only venetoclax. One patient went off study treatment due to a diagnosis of myelodysplastic syndrome, but that patient’s MCL is still in CR.
Five patients were able to stop treatment after achieving MRD-negative CR and were placed under “stringent surveillance,” Dr. Handunnetti said.
One of the five patients who stopped treatment progressed at 7 months and died. The remaining four patients are still alive and in CR at 6 months, 13 months, 17 months, and 18 months off treatment.
Safety update
Within the first 56 weeks of treatment, 15 patients required dose adjustments. Twelve patients required an adjustment to ibrutinib, seven to venetoclax, and four to both drugs. After 56 weeks, there were no dose adjustments.
Two patients developed therapy-related myelodysplastic syndrome. One patient had previously received FCR (fludarabine, cyclophosphamide, and rituximab) and BR (bendamustine and rituximab). The other patient had received R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone).
This investigator-initiated trial was funded by Janssen and Abbvie. Dr. Handunnetti reported relationships with Abbvie and Gilead.
SOURCE: Handunnetti S et al. ASH 2019. Abstract 756.
ORLANDO – Updated trial results have revealed durable responses with venetoclax and ibrutinib in patients with mantle cell lymphoma (MCL), allowing some patients to stop treatment.
Five of 24 patients were able to stop treatment after achieving minimal residual disease (MRD)-negative complete responses (CRs). Four of these patients remain in CR at up to 18 months off treatment, although one patient ultimately progressed and died.
“Treatment cessation was feasible for patients in MRD-negative complete responses, raising the prospect of limited-duration, targeted-agent therapy in the management of relapsed/refractory mantle cell lymphoma,” said Sasanka M. Handunnetti, MBBS, of Peter MacCallum Cancer Centre in Melbourne. Dr. Handunnetti presented these results, from the AIM trial, at the annual meeting of the American Society of Hematology.
The phase 2 trial enrolled 24 patients. At baseline, patients had a median age of 68 years (range, 47-81 years), and 88% were men. One patient was treatment-naive, but the rest had relapsed/refractory MCL. These patients had received a median of two prior therapies (range, 1-6).
The patients received venetoclax at 400 mg daily and ibrutinib at 560 mg daily.
In the primary analysis, the CR rate was 62% at week 16 and 71% overall, according to positron-emission tomography/computed tomography. MRD negativity was achieved by 67% of patients according to flow cytometry and 38% according to allele-specific oligonucleotide polymerase chain reaction (N Engl J Med. 2018 Mar 29;378[13]:1211-23).
Response and survival
For the current analysis, the median follow up was 37.5 months (range, 1.4-45.3 months). The median duration of response has not been reached, the median progression-free survival is 29 months, and the median overall survival is 32 months.
Thirteen patients have died, 8 of them due to progressive disease. The remaining 11 patients are still alive, and 9 of them are still in CR. One patient is still in partial response, and one has not responded but remains on ibrutinib and venetoclax.
Dr. Handunnetti pointed out that 12 patients had TP53 aberrations, and 8 of them died, but 4 remain alive and in CR. All four patients with SMARCA4 aberrations died.
Treatment status
Five patients are still receiving treatment with ibrutinib and venetoclax, and one patient is receiving only venetoclax. One patient went off study treatment due to a diagnosis of myelodysplastic syndrome, but that patient’s MCL is still in CR.
Five patients were able to stop treatment after achieving MRD-negative CR and were placed under “stringent surveillance,” Dr. Handunnetti said.
One of the five patients who stopped treatment progressed at 7 months and died. The remaining four patients are still alive and in CR at 6 months, 13 months, 17 months, and 18 months off treatment.
Safety update
Within the first 56 weeks of treatment, 15 patients required dose adjustments. Twelve patients required an adjustment to ibrutinib, seven to venetoclax, and four to both drugs. After 56 weeks, there were no dose adjustments.
Two patients developed therapy-related myelodysplastic syndrome. One patient had previously received FCR (fludarabine, cyclophosphamide, and rituximab) and BR (bendamustine and rituximab). The other patient had received R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone).
This investigator-initiated trial was funded by Janssen and Abbvie. Dr. Handunnetti reported relationships with Abbvie and Gilead.
SOURCE: Handunnetti S et al. ASH 2019. Abstract 756.
ORLANDO – Updated trial results have revealed durable responses with venetoclax and ibrutinib in patients with mantle cell lymphoma (MCL), allowing some patients to stop treatment.
Five of 24 patients were able to stop treatment after achieving minimal residual disease (MRD)-negative complete responses (CRs). Four of these patients remain in CR at up to 18 months off treatment, although one patient ultimately progressed and died.
“Treatment cessation was feasible for patients in MRD-negative complete responses, raising the prospect of limited-duration, targeted-agent therapy in the management of relapsed/refractory mantle cell lymphoma,” said Sasanka M. Handunnetti, MBBS, of Peter MacCallum Cancer Centre in Melbourne. Dr. Handunnetti presented these results, from the AIM trial, at the annual meeting of the American Society of Hematology.
The phase 2 trial enrolled 24 patients. At baseline, patients had a median age of 68 years (range, 47-81 years), and 88% were men. One patient was treatment-naive, but the rest had relapsed/refractory MCL. These patients had received a median of two prior therapies (range, 1-6).
The patients received venetoclax at 400 mg daily and ibrutinib at 560 mg daily.
In the primary analysis, the CR rate was 62% at week 16 and 71% overall, according to positron-emission tomography/computed tomography. MRD negativity was achieved by 67% of patients according to flow cytometry and 38% according to allele-specific oligonucleotide polymerase chain reaction (N Engl J Med. 2018 Mar 29;378[13]:1211-23).
Response and survival
For the current analysis, the median follow up was 37.5 months (range, 1.4-45.3 months). The median duration of response has not been reached, the median progression-free survival is 29 months, and the median overall survival is 32 months.
Thirteen patients have died, 8 of them due to progressive disease. The remaining 11 patients are still alive, and 9 of them are still in CR. One patient is still in partial response, and one has not responded but remains on ibrutinib and venetoclax.
Dr. Handunnetti pointed out that 12 patients had TP53 aberrations, and 8 of them died, but 4 remain alive and in CR. All four patients with SMARCA4 aberrations died.
Treatment status
Five patients are still receiving treatment with ibrutinib and venetoclax, and one patient is receiving only venetoclax. One patient went off study treatment due to a diagnosis of myelodysplastic syndrome, but that patient’s MCL is still in CR.
Five patients were able to stop treatment after achieving MRD-negative CR and were placed under “stringent surveillance,” Dr. Handunnetti said.
One of the five patients who stopped treatment progressed at 7 months and died. The remaining four patients are still alive and in CR at 6 months, 13 months, 17 months, and 18 months off treatment.
Safety update
Within the first 56 weeks of treatment, 15 patients required dose adjustments. Twelve patients required an adjustment to ibrutinib, seven to venetoclax, and four to both drugs. After 56 weeks, there were no dose adjustments.
Two patients developed therapy-related myelodysplastic syndrome. One patient had previously received FCR (fludarabine, cyclophosphamide, and rituximab) and BR (bendamustine and rituximab). The other patient had received R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone).
This investigator-initiated trial was funded by Janssen and Abbvie. Dr. Handunnetti reported relationships with Abbvie and Gilead.
SOURCE: Handunnetti S et al. ASH 2019. Abstract 756.
REPORTING FROM ASH 2019
KTE-X19 produces highest response rate in MCL subgroup
ORLANDO – KTE-X19, an anti-CD19 chimeric antigen receptor (CAR) T-cell therapy, demonstrated unprecedented efficacy in the ZUMA-2 trial, according to an investigator involved in the study.
KTE-X19 produced a 93% overall response rate in patients with relapsed/refractory mantle cell lymphoma (MCL). This is the highest reported response rate in patients who have failed treatment with a Bruton’s tyrosine kinase (BTK) inhibitor, said Michael L. Wang, MD, of the University of Texas MD Anderson Cancer Center in Houston.
Dr. Wang presented results from ZUMA-2 at the annual meeting of the American Society of Hematology.
“Patients with relapsed/refractory MCL have very poor outcomes,” Dr. Wang noted. “In patients who progress after BTK inhibition therapy, the overall response rate is only between 25% and 42%, and the overall survival is only between 6 and 10 months. Few patients proceed to allogeneic transplantation.”
The phase 2 ZUMA-2 trial was designed to test KTE-X19 in these patients. KTE-X19 is an anti-CD19 CAR T-cell therapy containing a CD3-zeta T-cell activation domain and a CD28 signaling domain. KTE-X19 is distinct from axicabtagene ciloleucel (KTE-C19) because the manufacturing process for KTE-X19 removes circulating tumor cells.
The trial enrolled 74 patients, and 68 of them received KTE-X19. Manufacturing failed for three patients, two patients died of progressive disease before they could receive KTE-X19, and one patient was found to be ineligible for treatment.
The 68 patients had a median age of 65 years (range, 38-79 years), and 84% were men. A majority of patients (85%) had stage IV disease and classical (59%) or blastoid (25%) morphology. Most patients (69%) had a Ki-67 proliferation index of 50% or greater, and most (56%) were intermediate- or high-risk according to the Mantle Cell Lymphoma International Prognostic Index (MIPI).
Patients had received a median of three prior therapies (range, one to five). All had been treated with a BTK inhibitor, with 85% receiving ibrutinib, 24% receiving acalabrutinib, and 9% receiving both. Most patients (68%) were refractory to BTK inhibition, and 32% relapsed on or after BTK inhibitor therapy.
In this study, patients could receive bridging therapy to keep their disease stable while KTE-X19 was being manufactured. There were 25 patients who received bridging therapy, which consisted of ibrutinib (n = 14), acalabrutinib (n = 5), dexamethasone (n = 12), and/or methylprednisolone (n = 2). Six patients received both BTK inhibitors and steroids.
All patients received conditioning with fludarabine and cyclophosphamide, followed by a single infusion of KTE-X19 at 2x106.
Efficacy
Sixty patients were evaluable for efficacy, and the median follow-up was 12.3 months (range, 7.0-32.3 months).
The overall response rate was 93%, with 67% of patients achieving a complete response and 27% achieving a partial response. Three percent of patients had stable disease, and 3% had progressive disease.
“The overall response rate was consistent across key subgroups, without any statistical difference,” Dr. Wang said. “This includes Ki-67, MIPI, and prior use of either steroids or bridging therapy.”
The median time to response was 1.0 month, and the median time to complete response was 3.0 months. Responses deepened over time, with 35% of patients converting from a partial response to a complete response, and 5% converting from stable disease to complete response.
The median duration of response has not been reached. At last follow-up, 57% of all patients and 78% of complete responders were still in response.
The median progression-free and overall survival have not been reached. At 12 months, the progression-free survival rate was 61%, and the overall survival rate was 83%.
Safety
All 68 patients were evaluable for safety. The most common adverse events were pyrexia (94%), neutropenia (87%), thrombocytopenia (74%), anemia (68%), and hypotension (51%).
Grade 3/4 adverse events included pyrexia (13%), neutropenia (85%), thrombocytopenia (51%), anemia (50%), hypotension (22%), hypoxia (21%), hypophosphatemia (22%), fatigue (1%), and headache (1%).
There were two grade 5 treatment-related adverse events – organizing pneumonia on day 37 and staphylococcal bacteremia on day 134.
Cytokine release syndrome (CRS) occurred in 91% of patients, with 15% experiencing grade 3 or higher CRS. Patients were treated with tocilizumab or corticosteroids, and all CRS events resolved.
Neurologic adverse events occurred in 63% of patients, with grade 3 or higher events occurring in 31%. Neurologic events were treated with tocilizumab or corticosteroids, and 86% of neurologic events resolved.
This trial was sponsored by Kite, a Gilead company. Dr. Wang reported financial relationships with Kite and other companies.
SOURCE: Wang M et al. ASH 2019. Abstract 754.
ORLANDO – KTE-X19, an anti-CD19 chimeric antigen receptor (CAR) T-cell therapy, demonstrated unprecedented efficacy in the ZUMA-2 trial, according to an investigator involved in the study.
KTE-X19 produced a 93% overall response rate in patients with relapsed/refractory mantle cell lymphoma (MCL). This is the highest reported response rate in patients who have failed treatment with a Bruton’s tyrosine kinase (BTK) inhibitor, said Michael L. Wang, MD, of the University of Texas MD Anderson Cancer Center in Houston.
Dr. Wang presented results from ZUMA-2 at the annual meeting of the American Society of Hematology.
“Patients with relapsed/refractory MCL have very poor outcomes,” Dr. Wang noted. “In patients who progress after BTK inhibition therapy, the overall response rate is only between 25% and 42%, and the overall survival is only between 6 and 10 months. Few patients proceed to allogeneic transplantation.”
The phase 2 ZUMA-2 trial was designed to test KTE-X19 in these patients. KTE-X19 is an anti-CD19 CAR T-cell therapy containing a CD3-zeta T-cell activation domain and a CD28 signaling domain. KTE-X19 is distinct from axicabtagene ciloleucel (KTE-C19) because the manufacturing process for KTE-X19 removes circulating tumor cells.
The trial enrolled 74 patients, and 68 of them received KTE-X19. Manufacturing failed for three patients, two patients died of progressive disease before they could receive KTE-X19, and one patient was found to be ineligible for treatment.
The 68 patients had a median age of 65 years (range, 38-79 years), and 84% were men. A majority of patients (85%) had stage IV disease and classical (59%) or blastoid (25%) morphology. Most patients (69%) had a Ki-67 proliferation index of 50% or greater, and most (56%) were intermediate- or high-risk according to the Mantle Cell Lymphoma International Prognostic Index (MIPI).
Patients had received a median of three prior therapies (range, one to five). All had been treated with a BTK inhibitor, with 85% receiving ibrutinib, 24% receiving acalabrutinib, and 9% receiving both. Most patients (68%) were refractory to BTK inhibition, and 32% relapsed on or after BTK inhibitor therapy.
In this study, patients could receive bridging therapy to keep their disease stable while KTE-X19 was being manufactured. There were 25 patients who received bridging therapy, which consisted of ibrutinib (n = 14), acalabrutinib (n = 5), dexamethasone (n = 12), and/or methylprednisolone (n = 2). Six patients received both BTK inhibitors and steroids.
All patients received conditioning with fludarabine and cyclophosphamide, followed by a single infusion of KTE-X19 at 2x106.
Efficacy
Sixty patients were evaluable for efficacy, and the median follow-up was 12.3 months (range, 7.0-32.3 months).
The overall response rate was 93%, with 67% of patients achieving a complete response and 27% achieving a partial response. Three percent of patients had stable disease, and 3% had progressive disease.
“The overall response rate was consistent across key subgroups, without any statistical difference,” Dr. Wang said. “This includes Ki-67, MIPI, and prior use of either steroids or bridging therapy.”
The median time to response was 1.0 month, and the median time to complete response was 3.0 months. Responses deepened over time, with 35% of patients converting from a partial response to a complete response, and 5% converting from stable disease to complete response.
The median duration of response has not been reached. At last follow-up, 57% of all patients and 78% of complete responders were still in response.
The median progression-free and overall survival have not been reached. At 12 months, the progression-free survival rate was 61%, and the overall survival rate was 83%.
Safety
All 68 patients were evaluable for safety. The most common adverse events were pyrexia (94%), neutropenia (87%), thrombocytopenia (74%), anemia (68%), and hypotension (51%).
Grade 3/4 adverse events included pyrexia (13%), neutropenia (85%), thrombocytopenia (51%), anemia (50%), hypotension (22%), hypoxia (21%), hypophosphatemia (22%), fatigue (1%), and headache (1%).
There were two grade 5 treatment-related adverse events – organizing pneumonia on day 37 and staphylococcal bacteremia on day 134.
Cytokine release syndrome (CRS) occurred in 91% of patients, with 15% experiencing grade 3 or higher CRS. Patients were treated with tocilizumab or corticosteroids, and all CRS events resolved.
Neurologic adverse events occurred in 63% of patients, with grade 3 or higher events occurring in 31%. Neurologic events were treated with tocilizumab or corticosteroids, and 86% of neurologic events resolved.
This trial was sponsored by Kite, a Gilead company. Dr. Wang reported financial relationships with Kite and other companies.
SOURCE: Wang M et al. ASH 2019. Abstract 754.
ORLANDO – KTE-X19, an anti-CD19 chimeric antigen receptor (CAR) T-cell therapy, demonstrated unprecedented efficacy in the ZUMA-2 trial, according to an investigator involved in the study.
KTE-X19 produced a 93% overall response rate in patients with relapsed/refractory mantle cell lymphoma (MCL). This is the highest reported response rate in patients who have failed treatment with a Bruton’s tyrosine kinase (BTK) inhibitor, said Michael L. Wang, MD, of the University of Texas MD Anderson Cancer Center in Houston.
Dr. Wang presented results from ZUMA-2 at the annual meeting of the American Society of Hematology.
“Patients with relapsed/refractory MCL have very poor outcomes,” Dr. Wang noted. “In patients who progress after BTK inhibition therapy, the overall response rate is only between 25% and 42%, and the overall survival is only between 6 and 10 months. Few patients proceed to allogeneic transplantation.”
The phase 2 ZUMA-2 trial was designed to test KTE-X19 in these patients. KTE-X19 is an anti-CD19 CAR T-cell therapy containing a CD3-zeta T-cell activation domain and a CD28 signaling domain. KTE-X19 is distinct from axicabtagene ciloleucel (KTE-C19) because the manufacturing process for KTE-X19 removes circulating tumor cells.
The trial enrolled 74 patients, and 68 of them received KTE-X19. Manufacturing failed for three patients, two patients died of progressive disease before they could receive KTE-X19, and one patient was found to be ineligible for treatment.
The 68 patients had a median age of 65 years (range, 38-79 years), and 84% were men. A majority of patients (85%) had stage IV disease and classical (59%) or blastoid (25%) morphology. Most patients (69%) had a Ki-67 proliferation index of 50% or greater, and most (56%) were intermediate- or high-risk according to the Mantle Cell Lymphoma International Prognostic Index (MIPI).
Patients had received a median of three prior therapies (range, one to five). All had been treated with a BTK inhibitor, with 85% receiving ibrutinib, 24% receiving acalabrutinib, and 9% receiving both. Most patients (68%) were refractory to BTK inhibition, and 32% relapsed on or after BTK inhibitor therapy.
In this study, patients could receive bridging therapy to keep their disease stable while KTE-X19 was being manufactured. There were 25 patients who received bridging therapy, which consisted of ibrutinib (n = 14), acalabrutinib (n = 5), dexamethasone (n = 12), and/or methylprednisolone (n = 2). Six patients received both BTK inhibitors and steroids.
All patients received conditioning with fludarabine and cyclophosphamide, followed by a single infusion of KTE-X19 at 2x106.
Efficacy
Sixty patients were evaluable for efficacy, and the median follow-up was 12.3 months (range, 7.0-32.3 months).
The overall response rate was 93%, with 67% of patients achieving a complete response and 27% achieving a partial response. Three percent of patients had stable disease, and 3% had progressive disease.
“The overall response rate was consistent across key subgroups, without any statistical difference,” Dr. Wang said. “This includes Ki-67, MIPI, and prior use of either steroids or bridging therapy.”
The median time to response was 1.0 month, and the median time to complete response was 3.0 months. Responses deepened over time, with 35% of patients converting from a partial response to a complete response, and 5% converting from stable disease to complete response.
The median duration of response has not been reached. At last follow-up, 57% of all patients and 78% of complete responders were still in response.
The median progression-free and overall survival have not been reached. At 12 months, the progression-free survival rate was 61%, and the overall survival rate was 83%.
Safety
All 68 patients were evaluable for safety. The most common adverse events were pyrexia (94%), neutropenia (87%), thrombocytopenia (74%), anemia (68%), and hypotension (51%).
Grade 3/4 adverse events included pyrexia (13%), neutropenia (85%), thrombocytopenia (51%), anemia (50%), hypotension (22%), hypoxia (21%), hypophosphatemia (22%), fatigue (1%), and headache (1%).
There were two grade 5 treatment-related adverse events – organizing pneumonia on day 37 and staphylococcal bacteremia on day 134.
Cytokine release syndrome (CRS) occurred in 91% of patients, with 15% experiencing grade 3 or higher CRS. Patients were treated with tocilizumab or corticosteroids, and all CRS events resolved.
Neurologic adverse events occurred in 63% of patients, with grade 3 or higher events occurring in 31%. Neurologic events were treated with tocilizumab or corticosteroids, and 86% of neurologic events resolved.
This trial was sponsored by Kite, a Gilead company. Dr. Wang reported financial relationships with Kite and other companies.
SOURCE: Wang M et al. ASH 2019. Abstract 754.
REPORTING FROM ASH 2019
Orelabrutinib could be ‘preferred’ BTK inhibitor for MCL
ORLANDO – A novel Bruton tyrosine kinase inhibitor has produced favorable results in patients with relapsed or refractory mantle cell lymphoma, according to findings presented at the annual meeting of the American Society of Hematology.
In a phase 2 trial, orelabrutinib produced an overall response rate of 86% and a 12-month progression-free survival rate of 64%. Safety results with orelabrutinib were superior to historical results with ibrutinib.
The efficacy and safety profile of orelabrutinib, as well as its “convenient” dosing, may make it the “preferred therapeutic choice for B-cell malignancy,” said Lijuan Deng, MD, PhD, of Peking University Cancer Hospital & Institute, Beijing, who presented the phase 2 trial of orelabrutinib at ASH 2019.
The trial enrolled 106 patients with relapsed/refractory mantle cell lymphoma who were treated at 22 centers in China. At baseline, the patients had a median age of 62 years (range, 37-73 years), and 79.2% were men. Most patients (94.4%) had stage III-IV disease.
Prior therapies included CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone)-based (69.8%), EPOCH (etoposide, prednisone, vincristine, cyclophosphamide, and doxorubicin)-based (22.6%), DHAP (dexamethasone, cytarabine, and cisplatin)-based (22.6%), CVAD (cyclophosphamide, vincristine, doxorubicin, dexamethasone)-based (12.3%), and ESHAP (etoposide, methylprednisolone, cytarabine, and cisplatin)-based (4.7%) regimens, and 88.7% of patients had received prior anti-CD20 therapy.
Patients received orelabrutinib at 100 mg twice daily (n = 20) or 150 mg once a day (n = 86). All 106 patients were evaluable for safety, and 99 were evaluable for efficacy.
Efficacy
“Orelabrutinib achieved high response and durable remissions,” Dr. Deng said.
The overall response rate was 85.9% in the evaluable efficacy population and 83.5% in the 150-mg dosing arm. The complete response rates were 27.3% and 29.1%, respectively. The median time to response, overall, was 1.9 months.
The median duration of response and median progression-free survival were not reached at a median follow-up of 10.5 months. At 12 months, 74.3% of patients were still in response, and the progression-free survival rate was 64%.
Safety
Most adverse events were grade 1-2 in nature. The most common grade 3 or higher events were platelet count decrease (11.3%), neutrophil count decrease (8.5%), anemia (7.5%), hypertension (3.8%), pneumonia (2.8%), white blood count decrease (1.9%), and hypokalemia (1.9%).
Adverse events of interest included grade 3 or higher hypertension (3.8%), diarrhea (6.6%), and infection (10.4%), as well as secondary malignancy (0.9%, n = 1). There were no cases of grade 3 or higher hemorrhage, grade 3 or higher atrial fibrillation/flutter, or grade 5 treatment-related adverse events.
Dr. Deng noted that rates of grade 3 or higher hemorrhage, atrial fibrillation, diarrhea, and infection, as well as rates of secondary malignancies, have historically been higher with ibrutinib (Blood. 2015 Aug 6;126[6]:739-45; Lancet. 2016 Feb 20;387[10020]:770-8).
“Orelabrutinib has an improved safety profile in patients with relapsed or refractory mantle cell lymphoma,” Dr. Deng said. “The most common adverse events were cytopenia and infections, which are considered mechanism based.”
The study was sponsored by InnoCare Pharma. Dr. Deng reported having no conflicts of interest.
SOURCE: Deng L et al. ASH 2019, Abstract 755.
ORLANDO – A novel Bruton tyrosine kinase inhibitor has produced favorable results in patients with relapsed or refractory mantle cell lymphoma, according to findings presented at the annual meeting of the American Society of Hematology.
In a phase 2 trial, orelabrutinib produced an overall response rate of 86% and a 12-month progression-free survival rate of 64%. Safety results with orelabrutinib were superior to historical results with ibrutinib.
The efficacy and safety profile of orelabrutinib, as well as its “convenient” dosing, may make it the “preferred therapeutic choice for B-cell malignancy,” said Lijuan Deng, MD, PhD, of Peking University Cancer Hospital & Institute, Beijing, who presented the phase 2 trial of orelabrutinib at ASH 2019.
The trial enrolled 106 patients with relapsed/refractory mantle cell lymphoma who were treated at 22 centers in China. At baseline, the patients had a median age of 62 years (range, 37-73 years), and 79.2% were men. Most patients (94.4%) had stage III-IV disease.
Prior therapies included CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone)-based (69.8%), EPOCH (etoposide, prednisone, vincristine, cyclophosphamide, and doxorubicin)-based (22.6%), DHAP (dexamethasone, cytarabine, and cisplatin)-based (22.6%), CVAD (cyclophosphamide, vincristine, doxorubicin, dexamethasone)-based (12.3%), and ESHAP (etoposide, methylprednisolone, cytarabine, and cisplatin)-based (4.7%) regimens, and 88.7% of patients had received prior anti-CD20 therapy.
Patients received orelabrutinib at 100 mg twice daily (n = 20) or 150 mg once a day (n = 86). All 106 patients were evaluable for safety, and 99 were evaluable for efficacy.
Efficacy
“Orelabrutinib achieved high response and durable remissions,” Dr. Deng said.
The overall response rate was 85.9% in the evaluable efficacy population and 83.5% in the 150-mg dosing arm. The complete response rates were 27.3% and 29.1%, respectively. The median time to response, overall, was 1.9 months.
The median duration of response and median progression-free survival were not reached at a median follow-up of 10.5 months. At 12 months, 74.3% of patients were still in response, and the progression-free survival rate was 64%.
Safety
Most adverse events were grade 1-2 in nature. The most common grade 3 or higher events were platelet count decrease (11.3%), neutrophil count decrease (8.5%), anemia (7.5%), hypertension (3.8%), pneumonia (2.8%), white blood count decrease (1.9%), and hypokalemia (1.9%).
Adverse events of interest included grade 3 or higher hypertension (3.8%), diarrhea (6.6%), and infection (10.4%), as well as secondary malignancy (0.9%, n = 1). There were no cases of grade 3 or higher hemorrhage, grade 3 or higher atrial fibrillation/flutter, or grade 5 treatment-related adverse events.
Dr. Deng noted that rates of grade 3 or higher hemorrhage, atrial fibrillation, diarrhea, and infection, as well as rates of secondary malignancies, have historically been higher with ibrutinib (Blood. 2015 Aug 6;126[6]:739-45; Lancet. 2016 Feb 20;387[10020]:770-8).
“Orelabrutinib has an improved safety profile in patients with relapsed or refractory mantle cell lymphoma,” Dr. Deng said. “The most common adverse events were cytopenia and infections, which are considered mechanism based.”
The study was sponsored by InnoCare Pharma. Dr. Deng reported having no conflicts of interest.
SOURCE: Deng L et al. ASH 2019, Abstract 755.
ORLANDO – A novel Bruton tyrosine kinase inhibitor has produced favorable results in patients with relapsed or refractory mantle cell lymphoma, according to findings presented at the annual meeting of the American Society of Hematology.
In a phase 2 trial, orelabrutinib produced an overall response rate of 86% and a 12-month progression-free survival rate of 64%. Safety results with orelabrutinib were superior to historical results with ibrutinib.
The efficacy and safety profile of orelabrutinib, as well as its “convenient” dosing, may make it the “preferred therapeutic choice for B-cell malignancy,” said Lijuan Deng, MD, PhD, of Peking University Cancer Hospital & Institute, Beijing, who presented the phase 2 trial of orelabrutinib at ASH 2019.
The trial enrolled 106 patients with relapsed/refractory mantle cell lymphoma who were treated at 22 centers in China. At baseline, the patients had a median age of 62 years (range, 37-73 years), and 79.2% were men. Most patients (94.4%) had stage III-IV disease.
Prior therapies included CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisolone)-based (69.8%), EPOCH (etoposide, prednisone, vincristine, cyclophosphamide, and doxorubicin)-based (22.6%), DHAP (dexamethasone, cytarabine, and cisplatin)-based (22.6%), CVAD (cyclophosphamide, vincristine, doxorubicin, dexamethasone)-based (12.3%), and ESHAP (etoposide, methylprednisolone, cytarabine, and cisplatin)-based (4.7%) regimens, and 88.7% of patients had received prior anti-CD20 therapy.
Patients received orelabrutinib at 100 mg twice daily (n = 20) or 150 mg once a day (n = 86). All 106 patients were evaluable for safety, and 99 were evaluable for efficacy.
Efficacy
“Orelabrutinib achieved high response and durable remissions,” Dr. Deng said.
The overall response rate was 85.9% in the evaluable efficacy population and 83.5% in the 150-mg dosing arm. The complete response rates were 27.3% and 29.1%, respectively. The median time to response, overall, was 1.9 months.
The median duration of response and median progression-free survival were not reached at a median follow-up of 10.5 months. At 12 months, 74.3% of patients were still in response, and the progression-free survival rate was 64%.
Safety
Most adverse events were grade 1-2 in nature. The most common grade 3 or higher events were platelet count decrease (11.3%), neutrophil count decrease (8.5%), anemia (7.5%), hypertension (3.8%), pneumonia (2.8%), white blood count decrease (1.9%), and hypokalemia (1.9%).
Adverse events of interest included grade 3 or higher hypertension (3.8%), diarrhea (6.6%), and infection (10.4%), as well as secondary malignancy (0.9%, n = 1). There were no cases of grade 3 or higher hemorrhage, grade 3 or higher atrial fibrillation/flutter, or grade 5 treatment-related adverse events.
Dr. Deng noted that rates of grade 3 or higher hemorrhage, atrial fibrillation, diarrhea, and infection, as well as rates of secondary malignancies, have historically been higher with ibrutinib (Blood. 2015 Aug 6;126[6]:739-45; Lancet. 2016 Feb 20;387[10020]:770-8).
“Orelabrutinib has an improved safety profile in patients with relapsed or refractory mantle cell lymphoma,” Dr. Deng said. “The most common adverse events were cytopenia and infections, which are considered mechanism based.”
The study was sponsored by InnoCare Pharma. Dr. Deng reported having no conflicts of interest.
SOURCE: Deng L et al. ASH 2019, Abstract 755.
REPORTING FROM ASH 2019
The clinical impact of new approvals in sickle cell, MCL
In this edition of “How I Will Treat My Next Patient,” I highlight two recent drug approvals by the Food and Drug Administration – crizanlizumab for sickle cell patients with painful crises and zanubrutinib for mantle cell lymphoma (MCL) patients in relapse.
Crizanlizumab
P-selectin is an adhesion molecule expressed on activated vascular endothelial cells and platelets. It is a key molecule in the initiation of leukocyte rolling on vessel walls and promotes firm attachment and extravasation to underlying tissues during inflammation. Up-regulation of P-selectin on endothelial cells and platelets contributes to the cell-cell interactions involved in the pathogenesis of sickle cell pain crises.
The SUSTAIN study was a multisite, placebo-controlled, randomized phase 2 trial of two different dosage levels of intravenous crizanlizumab (2.5 mg/kg or 5 mg/kg for 52 weeks), a humanized anti–P-selectin antibody, examining its effect on pain crises in patients with sickle cell disease. The primary endpoint was the annual rate of sickle cell pain crises, with a variety of clinically relevant secondary endpoints. The target population had 2-10 pain crises in the 12 months before enrollment. Patients on a stable dose of hydroxyurea for at least the most recent 3 months were allowed to enter, but if patients were not receiving hydroxyurea, it could not be initiated during the trial. Patients who were undergoing chronic red-cell transfusion therapy were excluded.
Among 198 enrolled patients, 35% did not complete the 52 weeks of treatment. Discontinuations were equally balanced among patients assigned to the high-dose, low-dose, and placebo cohorts. Adverse events associated with crizanlizumab included back pain, nausea, pyrexia, and arthralgia. Serious adverse events occurred in 55 patients, with 5 deaths, all of which were unrelated to treatment. Crizanlizumab did not augment hemolysis or bacterial infections.
In the efficacy analysis, patients receiving high-dose crizanlizumab had a median annual rate of 1.63 health care visits for sickle cell pain crises, compared with 2.98 visits for placebo patients (P = .01). In comparison with placebo, high-dose crizanlizumab also delayed the first pain crisis after starting treatment (4.1 months vs. 1.4 months), delayed the median time to a second pain crisis, and decreased the median number of pain crises annually.
More than twice as many high-dose crizanlizumab patients had no pain crisis episodes, compared with placebo patients. In general, differences were more striking in patients who were not taking hydroxyurea and who had non–hemoglobin SS disease. Differences in the primary endpoint between low-dose crizanlizumab and placebo were numerically, but not statistically, different.
How these results influence practice
It has been over 20 years since a new agent (hydroxyurea) was approved for sickle cell patients and, despite its use, sickle cell pain crises remain a frequent problem. Pain crises are associated with worse quality of life and increased risk of death. A promising advance is badly needed, especially in an era in which sensitivity to providers’ role in the opioid addiction crisis is highly scrutinized and may contribute to future undertreatment of pain episodes. This is especially true for patients from areas with high levels of opioid misuse.
The SUSTAIN trial was international, multi-institutional, placebo-controlled, and inclusive. These attributes enhance the likelihood that crizanlizumab will enhance patient care in routine practice. As an intravenous agent, monitoring adherence and toxicity are less challenging than with hydroxyurea. Despite these factors, however, there are some concerns. Crizanlizumab was not free of toxicity, quality of life via the Brief Pain Inventory used in the trial was not improved, and changes in the pain-severity and pain-interference domains were small. Treatment in SUSTAIN ensued for 52 weeks, so the emergence of late neutralizing antibodies and late toxicities with longer-term therapy will require careful postmarketing assessment.
These concerns notwithstanding, anyone who has cared for sickle cell patients would be excited about the potential benefits crizanlizumab could bring to patient care.
Zanubrutinib
The FDA has approved zanubrutinib for the treatment of MCL in adult patients who have received at least one prior therapy. The approval is based on the results of two studies in which overall response rate was the primary endpoint.
BGB-3111-206 (NCT03206970) was a phase 2, open-label, multicenter, single-arm trial of 86 patients with MCL who received at least one prior therapy. Zanubrutinib was given orally at 160 mg twice daily until disease progression or unacceptable toxicity. BGB-3111-AU-003 (NCT 02343120) was a phase 1/2, open-label, dose-escalation trial of B-cell malignancies, including 32 previously treated MCL patients treated with zanubrutinib at 160 mg twice daily or 320 mg once daily.
In the phase 2 trial, 18fluorodeoxyglucose (FDG)–PET scans were required and the ORR was 84% (95% confidence interval, 74%-91%), with a complete response rate of 59% (95% CI, 48%-70%) and a median response duration of 19.5 months (95% CI, 16.6% to not estimable). In the phase 1/2 dose-escalation trial, FDG-PET scans were not required and the ORR was 84% (95% CI, 67%-95%), with a complete response rate of 22% (95% CI, 9%-40%) and a median response duration of 18.5 months (95% CI, 12.6% to not estimable). In both trials, median follow-up on study was about 18 months.
The most common adverse reactions were cytopenias, upper respiratory tract infection, rash, bruising, diarrhea, and cough. The most common serious adverse reactions were pneumonia in 11% and hemorrhage in 5% of patients. Of 118 MCL patients, 8 stopped therapy because of an adverse event, most frequently pneumonia (3.4%).
How these results influence practice
Unfortunately, the therapy of recurrent MCL is noncurative, because of the rapid development of treatment resistance. There are multiple single-and multiagent chemotherapy regimens that may be tried, many incorporating immunotherapy options such as anti-CD20- or Bruton tyrosine kinase (BTK)–targeted agents. Given the limited efficacy of these agents, temporary nature of remissions, and paucity of data comparing these various treatment options, participation in clinical trials is encouraged whenever possible.
Outside of a clinical trial, zanubrutinib joins ibrutinib and acalabrutinib as approved single-agent BTK inhibitors for adult MCL patients in relapse. The impressive ORR and response duration reported for zanubrutinib are similar to the results achieved with the other agents, but the toxicity pattern may be slightly different.
As in the treatment of hormonally sensitive breast cancer, clinicians and patients benefit when they have multiple similar, equally efficacious oral agents with slightly different toxicity patterns so that quality of life can be improved and treatment duration maximized before treatment resistance develops and a more toxic and/or inconvenient therapy needs to be employed.
Whether zanubrutinib has benefits beyond those for MCL patients in relapse will depend on the results of confirmatory trials and patient-reported outcome data.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.
In this edition of “How I Will Treat My Next Patient,” I highlight two recent drug approvals by the Food and Drug Administration – crizanlizumab for sickle cell patients with painful crises and zanubrutinib for mantle cell lymphoma (MCL) patients in relapse.
Crizanlizumab
P-selectin is an adhesion molecule expressed on activated vascular endothelial cells and platelets. It is a key molecule in the initiation of leukocyte rolling on vessel walls and promotes firm attachment and extravasation to underlying tissues during inflammation. Up-regulation of P-selectin on endothelial cells and platelets contributes to the cell-cell interactions involved in the pathogenesis of sickle cell pain crises.
The SUSTAIN study was a multisite, placebo-controlled, randomized phase 2 trial of two different dosage levels of intravenous crizanlizumab (2.5 mg/kg or 5 mg/kg for 52 weeks), a humanized anti–P-selectin antibody, examining its effect on pain crises in patients with sickle cell disease. The primary endpoint was the annual rate of sickle cell pain crises, with a variety of clinically relevant secondary endpoints. The target population had 2-10 pain crises in the 12 months before enrollment. Patients on a stable dose of hydroxyurea for at least the most recent 3 months were allowed to enter, but if patients were not receiving hydroxyurea, it could not be initiated during the trial. Patients who were undergoing chronic red-cell transfusion therapy were excluded.
Among 198 enrolled patients, 35% did not complete the 52 weeks of treatment. Discontinuations were equally balanced among patients assigned to the high-dose, low-dose, and placebo cohorts. Adverse events associated with crizanlizumab included back pain, nausea, pyrexia, and arthralgia. Serious adverse events occurred in 55 patients, with 5 deaths, all of which were unrelated to treatment. Crizanlizumab did not augment hemolysis or bacterial infections.
In the efficacy analysis, patients receiving high-dose crizanlizumab had a median annual rate of 1.63 health care visits for sickle cell pain crises, compared with 2.98 visits for placebo patients (P = .01). In comparison with placebo, high-dose crizanlizumab also delayed the first pain crisis after starting treatment (4.1 months vs. 1.4 months), delayed the median time to a second pain crisis, and decreased the median number of pain crises annually.
More than twice as many high-dose crizanlizumab patients had no pain crisis episodes, compared with placebo patients. In general, differences were more striking in patients who were not taking hydroxyurea and who had non–hemoglobin SS disease. Differences in the primary endpoint between low-dose crizanlizumab and placebo were numerically, but not statistically, different.
How these results influence practice
It has been over 20 years since a new agent (hydroxyurea) was approved for sickle cell patients and, despite its use, sickle cell pain crises remain a frequent problem. Pain crises are associated with worse quality of life and increased risk of death. A promising advance is badly needed, especially in an era in which sensitivity to providers’ role in the opioid addiction crisis is highly scrutinized and may contribute to future undertreatment of pain episodes. This is especially true for patients from areas with high levels of opioid misuse.
The SUSTAIN trial was international, multi-institutional, placebo-controlled, and inclusive. These attributes enhance the likelihood that crizanlizumab will enhance patient care in routine practice. As an intravenous agent, monitoring adherence and toxicity are less challenging than with hydroxyurea. Despite these factors, however, there are some concerns. Crizanlizumab was not free of toxicity, quality of life via the Brief Pain Inventory used in the trial was not improved, and changes in the pain-severity and pain-interference domains were small. Treatment in SUSTAIN ensued for 52 weeks, so the emergence of late neutralizing antibodies and late toxicities with longer-term therapy will require careful postmarketing assessment.
These concerns notwithstanding, anyone who has cared for sickle cell patients would be excited about the potential benefits crizanlizumab could bring to patient care.
Zanubrutinib
The FDA has approved zanubrutinib for the treatment of MCL in adult patients who have received at least one prior therapy. The approval is based on the results of two studies in which overall response rate was the primary endpoint.
BGB-3111-206 (NCT03206970) was a phase 2, open-label, multicenter, single-arm trial of 86 patients with MCL who received at least one prior therapy. Zanubrutinib was given orally at 160 mg twice daily until disease progression or unacceptable toxicity. BGB-3111-AU-003 (NCT 02343120) was a phase 1/2, open-label, dose-escalation trial of B-cell malignancies, including 32 previously treated MCL patients treated with zanubrutinib at 160 mg twice daily or 320 mg once daily.
In the phase 2 trial, 18fluorodeoxyglucose (FDG)–PET scans were required and the ORR was 84% (95% confidence interval, 74%-91%), with a complete response rate of 59% (95% CI, 48%-70%) and a median response duration of 19.5 months (95% CI, 16.6% to not estimable). In the phase 1/2 dose-escalation trial, FDG-PET scans were not required and the ORR was 84% (95% CI, 67%-95%), with a complete response rate of 22% (95% CI, 9%-40%) and a median response duration of 18.5 months (95% CI, 12.6% to not estimable). In both trials, median follow-up on study was about 18 months.
The most common adverse reactions were cytopenias, upper respiratory tract infection, rash, bruising, diarrhea, and cough. The most common serious adverse reactions were pneumonia in 11% and hemorrhage in 5% of patients. Of 118 MCL patients, 8 stopped therapy because of an adverse event, most frequently pneumonia (3.4%).
How these results influence practice
Unfortunately, the therapy of recurrent MCL is noncurative, because of the rapid development of treatment resistance. There are multiple single-and multiagent chemotherapy regimens that may be tried, many incorporating immunotherapy options such as anti-CD20- or Bruton tyrosine kinase (BTK)–targeted agents. Given the limited efficacy of these agents, temporary nature of remissions, and paucity of data comparing these various treatment options, participation in clinical trials is encouraged whenever possible.
Outside of a clinical trial, zanubrutinib joins ibrutinib and acalabrutinib as approved single-agent BTK inhibitors for adult MCL patients in relapse. The impressive ORR and response duration reported for zanubrutinib are similar to the results achieved with the other agents, but the toxicity pattern may be slightly different.
As in the treatment of hormonally sensitive breast cancer, clinicians and patients benefit when they have multiple similar, equally efficacious oral agents with slightly different toxicity patterns so that quality of life can be improved and treatment duration maximized before treatment resistance develops and a more toxic and/or inconvenient therapy needs to be employed.
Whether zanubrutinib has benefits beyond those for MCL patients in relapse will depend on the results of confirmatory trials and patient-reported outcome data.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.
In this edition of “How I Will Treat My Next Patient,” I highlight two recent drug approvals by the Food and Drug Administration – crizanlizumab for sickle cell patients with painful crises and zanubrutinib for mantle cell lymphoma (MCL) patients in relapse.
Crizanlizumab
P-selectin is an adhesion molecule expressed on activated vascular endothelial cells and platelets. It is a key molecule in the initiation of leukocyte rolling on vessel walls and promotes firm attachment and extravasation to underlying tissues during inflammation. Up-regulation of P-selectin on endothelial cells and platelets contributes to the cell-cell interactions involved in the pathogenesis of sickle cell pain crises.
The SUSTAIN study was a multisite, placebo-controlled, randomized phase 2 trial of two different dosage levels of intravenous crizanlizumab (2.5 mg/kg or 5 mg/kg for 52 weeks), a humanized anti–P-selectin antibody, examining its effect on pain crises in patients with sickle cell disease. The primary endpoint was the annual rate of sickle cell pain crises, with a variety of clinically relevant secondary endpoints. The target population had 2-10 pain crises in the 12 months before enrollment. Patients on a stable dose of hydroxyurea for at least the most recent 3 months were allowed to enter, but if patients were not receiving hydroxyurea, it could not be initiated during the trial. Patients who were undergoing chronic red-cell transfusion therapy were excluded.
Among 198 enrolled patients, 35% did not complete the 52 weeks of treatment. Discontinuations were equally balanced among patients assigned to the high-dose, low-dose, and placebo cohorts. Adverse events associated with crizanlizumab included back pain, nausea, pyrexia, and arthralgia. Serious adverse events occurred in 55 patients, with 5 deaths, all of which were unrelated to treatment. Crizanlizumab did not augment hemolysis or bacterial infections.
In the efficacy analysis, patients receiving high-dose crizanlizumab had a median annual rate of 1.63 health care visits for sickle cell pain crises, compared with 2.98 visits for placebo patients (P = .01). In comparison with placebo, high-dose crizanlizumab also delayed the first pain crisis after starting treatment (4.1 months vs. 1.4 months), delayed the median time to a second pain crisis, and decreased the median number of pain crises annually.
More than twice as many high-dose crizanlizumab patients had no pain crisis episodes, compared with placebo patients. In general, differences were more striking in patients who were not taking hydroxyurea and who had non–hemoglobin SS disease. Differences in the primary endpoint between low-dose crizanlizumab and placebo were numerically, but not statistically, different.
How these results influence practice
It has been over 20 years since a new agent (hydroxyurea) was approved for sickle cell patients and, despite its use, sickle cell pain crises remain a frequent problem. Pain crises are associated with worse quality of life and increased risk of death. A promising advance is badly needed, especially in an era in which sensitivity to providers’ role in the opioid addiction crisis is highly scrutinized and may contribute to future undertreatment of pain episodes. This is especially true for patients from areas with high levels of opioid misuse.
The SUSTAIN trial was international, multi-institutional, placebo-controlled, and inclusive. These attributes enhance the likelihood that crizanlizumab will enhance patient care in routine practice. As an intravenous agent, monitoring adherence and toxicity are less challenging than with hydroxyurea. Despite these factors, however, there are some concerns. Crizanlizumab was not free of toxicity, quality of life via the Brief Pain Inventory used in the trial was not improved, and changes in the pain-severity and pain-interference domains were small. Treatment in SUSTAIN ensued for 52 weeks, so the emergence of late neutralizing antibodies and late toxicities with longer-term therapy will require careful postmarketing assessment.
These concerns notwithstanding, anyone who has cared for sickle cell patients would be excited about the potential benefits crizanlizumab could bring to patient care.
Zanubrutinib
The FDA has approved zanubrutinib for the treatment of MCL in adult patients who have received at least one prior therapy. The approval is based on the results of two studies in which overall response rate was the primary endpoint.
BGB-3111-206 (NCT03206970) was a phase 2, open-label, multicenter, single-arm trial of 86 patients with MCL who received at least one prior therapy. Zanubrutinib was given orally at 160 mg twice daily until disease progression or unacceptable toxicity. BGB-3111-AU-003 (NCT 02343120) was a phase 1/2, open-label, dose-escalation trial of B-cell malignancies, including 32 previously treated MCL patients treated with zanubrutinib at 160 mg twice daily or 320 mg once daily.
In the phase 2 trial, 18fluorodeoxyglucose (FDG)–PET scans were required and the ORR was 84% (95% confidence interval, 74%-91%), with a complete response rate of 59% (95% CI, 48%-70%) and a median response duration of 19.5 months (95% CI, 16.6% to not estimable). In the phase 1/2 dose-escalation trial, FDG-PET scans were not required and the ORR was 84% (95% CI, 67%-95%), with a complete response rate of 22% (95% CI, 9%-40%) and a median response duration of 18.5 months (95% CI, 12.6% to not estimable). In both trials, median follow-up on study was about 18 months.
The most common adverse reactions were cytopenias, upper respiratory tract infection, rash, bruising, diarrhea, and cough. The most common serious adverse reactions were pneumonia in 11% and hemorrhage in 5% of patients. Of 118 MCL patients, 8 stopped therapy because of an adverse event, most frequently pneumonia (3.4%).
How these results influence practice
Unfortunately, the therapy of recurrent MCL is noncurative, because of the rapid development of treatment resistance. There are multiple single-and multiagent chemotherapy regimens that may be tried, many incorporating immunotherapy options such as anti-CD20- or Bruton tyrosine kinase (BTK)–targeted agents. Given the limited efficacy of these agents, temporary nature of remissions, and paucity of data comparing these various treatment options, participation in clinical trials is encouraged whenever possible.
Outside of a clinical trial, zanubrutinib joins ibrutinib and acalabrutinib as approved single-agent BTK inhibitors for adult MCL patients in relapse. The impressive ORR and response duration reported for zanubrutinib are similar to the results achieved with the other agents, but the toxicity pattern may be slightly different.
As in the treatment of hormonally sensitive breast cancer, clinicians and patients benefit when they have multiple similar, equally efficacious oral agents with slightly different toxicity patterns so that quality of life can be improved and treatment duration maximized before treatment resistance develops and a more toxic and/or inconvenient therapy needs to be employed.
Whether zanubrutinib has benefits beyond those for MCL patients in relapse will depend on the results of confirmatory trials and patient-reported outcome data.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.