User login
FDA approves ravulizumab for treatment of paroxysmal nocturnal hemoglobinuria
The Food and Drug Administration has approved ravulizumab (Ultomiris) injection for the treatment of adult patients with paroxysmal nocturnal hemoglobinuria (PNH).
“The approval of Ultomiris will change the way that patients with PNH are treated. Prior to this approval, the only approved therapy for PNH required treatment every 2 weeks, which can be burdensome for patients and their families. Ultomiris uses a novel formulation so patients only need treatment every 8 weeks, without compromising efficacy,” Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence, said in a press release from the agency.
Patients with PNH, a rare disorder, lack a protein which protects red blood cells from being destroyed in the immune system. Episodes can be triggered by stresses on the body such as infection or physical exertion, and symptoms include severe anemia, profound fatigue, shortness of breath, intermittent episodes of dark-colored urine, kidney disease, or recurrent pain.
FDA approval for ravulizumab is based on results from a pair of clinical trials. In the first, 246 treatment-naive PNH patients received either ravulizumab or eculizumab, the current standard of care; ravulizumab was noninferior, with no patients undergoing a transfusion and all patients having similar incidence of hemolysis. In the second trial, 195 patients who had clinically stable PNH after receiving eculizumab for 6 months were randomized to receive ravulizumab or continue eculizumab; again, ravulizumab was noninferior.
The most common adverse events associated with ravulizumab were headache and respiratory tract infection. Caution is recommended when prescribing ravulizumab to patients with any type of infection.
Find the full press release on the FDA website.
The Food and Drug Administration has approved ravulizumab (Ultomiris) injection for the treatment of adult patients with paroxysmal nocturnal hemoglobinuria (PNH).
“The approval of Ultomiris will change the way that patients with PNH are treated. Prior to this approval, the only approved therapy for PNH required treatment every 2 weeks, which can be burdensome for patients and their families. Ultomiris uses a novel formulation so patients only need treatment every 8 weeks, without compromising efficacy,” Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence, said in a press release from the agency.
Patients with PNH, a rare disorder, lack a protein which protects red blood cells from being destroyed in the immune system. Episodes can be triggered by stresses on the body such as infection or physical exertion, and symptoms include severe anemia, profound fatigue, shortness of breath, intermittent episodes of dark-colored urine, kidney disease, or recurrent pain.
FDA approval for ravulizumab is based on results from a pair of clinical trials. In the first, 246 treatment-naive PNH patients received either ravulizumab or eculizumab, the current standard of care; ravulizumab was noninferior, with no patients undergoing a transfusion and all patients having similar incidence of hemolysis. In the second trial, 195 patients who had clinically stable PNH after receiving eculizumab for 6 months were randomized to receive ravulizumab or continue eculizumab; again, ravulizumab was noninferior.
The most common adverse events associated with ravulizumab were headache and respiratory tract infection. Caution is recommended when prescribing ravulizumab to patients with any type of infection.
Find the full press release on the FDA website.
The Food and Drug Administration has approved ravulizumab (Ultomiris) injection for the treatment of adult patients with paroxysmal nocturnal hemoglobinuria (PNH).
“The approval of Ultomiris will change the way that patients with PNH are treated. Prior to this approval, the only approved therapy for PNH required treatment every 2 weeks, which can be burdensome for patients and their families. Ultomiris uses a novel formulation so patients only need treatment every 8 weeks, without compromising efficacy,” Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence, said in a press release from the agency.
Patients with PNH, a rare disorder, lack a protein which protects red blood cells from being destroyed in the immune system. Episodes can be triggered by stresses on the body such as infection or physical exertion, and symptoms include severe anemia, profound fatigue, shortness of breath, intermittent episodes of dark-colored urine, kidney disease, or recurrent pain.
FDA approval for ravulizumab is based on results from a pair of clinical trials. In the first, 246 treatment-naive PNH patients received either ravulizumab or eculizumab, the current standard of care; ravulizumab was noninferior, with no patients undergoing a transfusion and all patients having similar incidence of hemolysis. In the second trial, 195 patients who had clinically stable PNH after receiving eculizumab for 6 months were randomized to receive ravulizumab or continue eculizumab; again, ravulizumab was noninferior.
The most common adverse events associated with ravulizumab were headache and respiratory tract infection. Caution is recommended when prescribing ravulizumab to patients with any type of infection.
Find the full press release on the FDA website.
Vedolizumab, tofacitinib induced rapid improvements in IBD symptoms
Two recent studies highlight the ability of vedolizumab and tofacitinib to rapidly improve symptoms reported by patients with inflammatory bowel disease (IBD).
In a post hoc study of 1,758 patients with ulcerative colitis (UC) or Crohn’s disease (CD) in the phase 3 GEMINI trials, 2 weeks of vedolizumab (Entyvio) therapy effectively improved patient-reported outcomes, and these continued to improve through 6 weeks of treatment, wrote Brian G. Feagan, MD, and his associates in the January issue of Clinical Gastroenterology and Hepatology.
In UC patients who had not previously received tumor necrosis factor (TNF) antagonists, 22% of vedolizumab recipients, compared with 7% of placebo recipients, achieved complete resolution of rectal bleeding together with a meaningful reduction in stool frequency at treatment week 2, the investigators noted. Among CD patients who were naive to TNF antagonists, 15% reported decreases in abdominal pain and loose stools at treatment week 2, compared with 8% of placebo recipients.
Although 2 weeks of vedolizumab also topped placebo for improving patient-reported outcomes among TNF antagonist–exposed patients, the effects were less pronounced, wrote Dr. Feagan, of the University of Western Ontario, London, and his associates. “These data add to the growing evidence that second-generation biologics, such as vedolizumab and ustekinumab, have higher efficacy in TNF antagonist–naive patients in both clinical trials and real-world settings. Recent trends in clinical practice are moving toward incorporating disease-modifying therapy earlier in the treatment of IBD to prevent disease progression and cumulative bowel damage.”
Patient-reported outcomes have become key during both clinical research and regulatory review of claims on proposed drug labels. In the second study, also published in the January issue of Clinical Gastroenterology and Hepatology, Stephen B. Hanauer, MD, of Northwestern University, Chicago, and his associates performed a post hoc analysis of symptoms reported by 1,139 adults with UC who received the oral small-molecule Janus kinase inhibitor tofacitinib (10 g twice daily) or placebo during the 8-week OCTAVE Induction 1 and 2 trials. These were identical phase 3 studies of patients with moderate to severe UC who could not tolerate or had responded inadequately to TNF antagonists, corticosteroids, or thiopurines.
Compared with placebo, 3 days of tofacitinib therapy induced significantly greater reductions from baseline in patient-reported stool frequency and rectal bleeding (P less than .01 for each measure), Dr. Hanauer and his associates reported. The effect was independent of prior treatment for UC or baseline levels of C-reactive protein. These findings reflect the rapid onset of effect of tofacitinib therapy in patients with UC. In contrast, thiopurines (azathioprine and 6-mercaptopurine) can take at least 8 weeks to exhibit steroid-sparing effects.
While corticosteroids can induce UC remission within 5 days, their side effects tend to escalate over time and they “lack maintenance benefits,” the researchers wrote. “In these analyses, onset of tofacitinib efficacy occurred within 3 days, irrespective of concomitant corticosteroid use or prior anti-TNF treatment failure.”
Takeda funded the GEMINI studies. Dr. Feagan reported advisory relationships with Takeda, AbbVie, Amgen, AstraZeneca, and several other pharmaceutical companies. Dr. Hanauer also reported ties to numerous pharmaceutical companies, including Pfizer, which funded the OCTAVE trials.
SOURCES: Feagan BG et al. Clin Gastroenterol Hepatol. 2018 May 29. doi: 10.1016/j.cgh.2018.05.026; Hanauer SB et al. Clin Gastroenterol Hepatol. 2018 Jul 15. doi: 10.1016/j.cgh.2018.07.009.
Two recent studies highlight the ability of vedolizumab and tofacitinib to rapidly improve symptoms reported by patients with inflammatory bowel disease (IBD).
In a post hoc study of 1,758 patients with ulcerative colitis (UC) or Crohn’s disease (CD) in the phase 3 GEMINI trials, 2 weeks of vedolizumab (Entyvio) therapy effectively improved patient-reported outcomes, and these continued to improve through 6 weeks of treatment, wrote Brian G. Feagan, MD, and his associates in the January issue of Clinical Gastroenterology and Hepatology.
In UC patients who had not previously received tumor necrosis factor (TNF) antagonists, 22% of vedolizumab recipients, compared with 7% of placebo recipients, achieved complete resolution of rectal bleeding together with a meaningful reduction in stool frequency at treatment week 2, the investigators noted. Among CD patients who were naive to TNF antagonists, 15% reported decreases in abdominal pain and loose stools at treatment week 2, compared with 8% of placebo recipients.
Although 2 weeks of vedolizumab also topped placebo for improving patient-reported outcomes among TNF antagonist–exposed patients, the effects were less pronounced, wrote Dr. Feagan, of the University of Western Ontario, London, and his associates. “These data add to the growing evidence that second-generation biologics, such as vedolizumab and ustekinumab, have higher efficacy in TNF antagonist–naive patients in both clinical trials and real-world settings. Recent trends in clinical practice are moving toward incorporating disease-modifying therapy earlier in the treatment of IBD to prevent disease progression and cumulative bowel damage.”
Patient-reported outcomes have become key during both clinical research and regulatory review of claims on proposed drug labels. In the second study, also published in the January issue of Clinical Gastroenterology and Hepatology, Stephen B. Hanauer, MD, of Northwestern University, Chicago, and his associates performed a post hoc analysis of symptoms reported by 1,139 adults with UC who received the oral small-molecule Janus kinase inhibitor tofacitinib (10 g twice daily) or placebo during the 8-week OCTAVE Induction 1 and 2 trials. These were identical phase 3 studies of patients with moderate to severe UC who could not tolerate or had responded inadequately to TNF antagonists, corticosteroids, or thiopurines.
Compared with placebo, 3 days of tofacitinib therapy induced significantly greater reductions from baseline in patient-reported stool frequency and rectal bleeding (P less than .01 for each measure), Dr. Hanauer and his associates reported. The effect was independent of prior treatment for UC or baseline levels of C-reactive protein. These findings reflect the rapid onset of effect of tofacitinib therapy in patients with UC. In contrast, thiopurines (azathioprine and 6-mercaptopurine) can take at least 8 weeks to exhibit steroid-sparing effects.
While corticosteroids can induce UC remission within 5 days, their side effects tend to escalate over time and they “lack maintenance benefits,” the researchers wrote. “In these analyses, onset of tofacitinib efficacy occurred within 3 days, irrespective of concomitant corticosteroid use or prior anti-TNF treatment failure.”
Takeda funded the GEMINI studies. Dr. Feagan reported advisory relationships with Takeda, AbbVie, Amgen, AstraZeneca, and several other pharmaceutical companies. Dr. Hanauer also reported ties to numerous pharmaceutical companies, including Pfizer, which funded the OCTAVE trials.
SOURCES: Feagan BG et al. Clin Gastroenterol Hepatol. 2018 May 29. doi: 10.1016/j.cgh.2018.05.026; Hanauer SB et al. Clin Gastroenterol Hepatol. 2018 Jul 15. doi: 10.1016/j.cgh.2018.07.009.
Two recent studies highlight the ability of vedolizumab and tofacitinib to rapidly improve symptoms reported by patients with inflammatory bowel disease (IBD).
In a post hoc study of 1,758 patients with ulcerative colitis (UC) or Crohn’s disease (CD) in the phase 3 GEMINI trials, 2 weeks of vedolizumab (Entyvio) therapy effectively improved patient-reported outcomes, and these continued to improve through 6 weeks of treatment, wrote Brian G. Feagan, MD, and his associates in the January issue of Clinical Gastroenterology and Hepatology.
In UC patients who had not previously received tumor necrosis factor (TNF) antagonists, 22% of vedolizumab recipients, compared with 7% of placebo recipients, achieved complete resolution of rectal bleeding together with a meaningful reduction in stool frequency at treatment week 2, the investigators noted. Among CD patients who were naive to TNF antagonists, 15% reported decreases in abdominal pain and loose stools at treatment week 2, compared with 8% of placebo recipients.
Although 2 weeks of vedolizumab also topped placebo for improving patient-reported outcomes among TNF antagonist–exposed patients, the effects were less pronounced, wrote Dr. Feagan, of the University of Western Ontario, London, and his associates. “These data add to the growing evidence that second-generation biologics, such as vedolizumab and ustekinumab, have higher efficacy in TNF antagonist–naive patients in both clinical trials and real-world settings. Recent trends in clinical practice are moving toward incorporating disease-modifying therapy earlier in the treatment of IBD to prevent disease progression and cumulative bowel damage.”
Patient-reported outcomes have become key during both clinical research and regulatory review of claims on proposed drug labels. In the second study, also published in the January issue of Clinical Gastroenterology and Hepatology, Stephen B. Hanauer, MD, of Northwestern University, Chicago, and his associates performed a post hoc analysis of symptoms reported by 1,139 adults with UC who received the oral small-molecule Janus kinase inhibitor tofacitinib (10 g twice daily) or placebo during the 8-week OCTAVE Induction 1 and 2 trials. These were identical phase 3 studies of patients with moderate to severe UC who could not tolerate or had responded inadequately to TNF antagonists, corticosteroids, or thiopurines.
Compared with placebo, 3 days of tofacitinib therapy induced significantly greater reductions from baseline in patient-reported stool frequency and rectal bleeding (P less than .01 for each measure), Dr. Hanauer and his associates reported. The effect was independent of prior treatment for UC or baseline levels of C-reactive protein. These findings reflect the rapid onset of effect of tofacitinib therapy in patients with UC. In contrast, thiopurines (azathioprine and 6-mercaptopurine) can take at least 8 weeks to exhibit steroid-sparing effects.
While corticosteroids can induce UC remission within 5 days, their side effects tend to escalate over time and they “lack maintenance benefits,” the researchers wrote. “In these analyses, onset of tofacitinib efficacy occurred within 3 days, irrespective of concomitant corticosteroid use or prior anti-TNF treatment failure.”
Takeda funded the GEMINI studies. Dr. Feagan reported advisory relationships with Takeda, AbbVie, Amgen, AstraZeneca, and several other pharmaceutical companies. Dr. Hanauer also reported ties to numerous pharmaceutical companies, including Pfizer, which funded the OCTAVE trials.
SOURCES: Feagan BG et al. Clin Gastroenterol Hepatol. 2018 May 29. doi: 10.1016/j.cgh.2018.05.026; Hanauer SB et al. Clin Gastroenterol Hepatol. 2018 Jul 15. doi: 10.1016/j.cgh.2018.07.009.
Memorial and honorary gifts: a special tribute
Make a tribute gift to honor someone whose life has been touched by GI research or celebrate a special occasion such as a birthday while supporting the AGA Research Awards Program through the AGA Research Foundation. A tribute gift will make your loved one feel special because it honors their passion, and also help us fund research grants to talented young investigators whose work will shape the future of clinical care.
- Giving a gift to the AGA Research Foundation in memory of a loved one. A memorial gift is a meaningful way to celebrate the legacy of a family member, friend, or colleague.
- A gift today. An outright gift will help fund the AGA Research Awards Program. Your gift will assist in furthering basic digestive disease research which can ultimately advance research into all digestive diseases. The financial benefits include an income tax deduction and possible elimination of capital gains tax.
Learn more about ways to recognize and acknowledge someone by visit our website at www.gastro.org/contribute or contact Harmony Excellent at 301-272-1602 or hexcellent@gastro.org.
Make a tribute gift to honor someone whose life has been touched by GI research or celebrate a special occasion such as a birthday while supporting the AGA Research Awards Program through the AGA Research Foundation. A tribute gift will make your loved one feel special because it honors their passion, and also help us fund research grants to talented young investigators whose work will shape the future of clinical care.
- Giving a gift to the AGA Research Foundation in memory of a loved one. A memorial gift is a meaningful way to celebrate the legacy of a family member, friend, or colleague.
- A gift today. An outright gift will help fund the AGA Research Awards Program. Your gift will assist in furthering basic digestive disease research which can ultimately advance research into all digestive diseases. The financial benefits include an income tax deduction and possible elimination of capital gains tax.
Learn more about ways to recognize and acknowledge someone by visit our website at www.gastro.org/contribute or contact Harmony Excellent at 301-272-1602 or hexcellent@gastro.org.
Make a tribute gift to honor someone whose life has been touched by GI research or celebrate a special occasion such as a birthday while supporting the AGA Research Awards Program through the AGA Research Foundation. A tribute gift will make your loved one feel special because it honors their passion, and also help us fund research grants to talented young investigators whose work will shape the future of clinical care.
- Giving a gift to the AGA Research Foundation in memory of a loved one. A memorial gift is a meaningful way to celebrate the legacy of a family member, friend, or colleague.
- A gift today. An outright gift will help fund the AGA Research Awards Program. Your gift will assist in furthering basic digestive disease research which can ultimately advance research into all digestive diseases. The financial benefits include an income tax deduction and possible elimination of capital gains tax.
Learn more about ways to recognize and acknowledge someone by visit our website at www.gastro.org/contribute or contact Harmony Excellent at 301-272-1602 or hexcellent@gastro.org.
Top AGA Community patient cases
about therapy and disease management options, best practices, and diagnoses.
In case you missed it, here are the most popular clinical discussions shared in the forum recently:
1. Active colitis in a patient with previous colon cancer
This popular conversation centers around next steps for a 39-year-old previously treated for a large malignant tumor with a right hemicolectomy and chemotherapy. She was referred for a chromoendoscopy, which revealed four areas of nonpolypoid abnormal mucosa with indistinct borders and abnormal dye uptake. Biopsies also revealed low-grade dysplasia and minimally active colitis.
2. IBD in remission
A 66-year-old female with a history of Crohn’s disease is currently asymptomatic and in remission, but with low Remicade trough. The debate among physicians in the forum questions the need for further action or follow-up insight. 3. Eosinophilic esophagitis and duodenitis (http://ow.ly/kja130mRoDV)
This 40-year-old patient was originally seen for food bolus. The physician prescribed proton pump inhibitors (PPI) after a scope showed typical eosinophilic esophagitis (EoE) findings. Although he had symptoms of obstruction since he was 15, this was his first upper endoscopy. Biopsies following successful scopes showed eosinophilic duodenitis and the patient had no signs of eosinophilia in the stomach.
4. Eosinophilic esophagitis and gastric sleeve
A physician noted no established absolute contraindication for a 50-year-old patient who was seen for a presleeve gastrectomy. Others contributing to this thread shared concerns for the risk for gastroesophageal reflux disease (GERD) post-surgery and committing the patient to long-term steroids.
More clinical cases and discussions are at https://community.gastro.org/discussions.
about therapy and disease management options, best practices, and diagnoses.
In case you missed it, here are the most popular clinical discussions shared in the forum recently:
1. Active colitis in a patient with previous colon cancer
This popular conversation centers around next steps for a 39-year-old previously treated for a large malignant tumor with a right hemicolectomy and chemotherapy. She was referred for a chromoendoscopy, which revealed four areas of nonpolypoid abnormal mucosa with indistinct borders and abnormal dye uptake. Biopsies also revealed low-grade dysplasia and minimally active colitis.
2. IBD in remission
A 66-year-old female with a history of Crohn’s disease is currently asymptomatic and in remission, but with low Remicade trough. The debate among physicians in the forum questions the need for further action or follow-up insight. 3. Eosinophilic esophagitis and duodenitis (http://ow.ly/kja130mRoDV)
This 40-year-old patient was originally seen for food bolus. The physician prescribed proton pump inhibitors (PPI) after a scope showed typical eosinophilic esophagitis (EoE) findings. Although he had symptoms of obstruction since he was 15, this was his first upper endoscopy. Biopsies following successful scopes showed eosinophilic duodenitis and the patient had no signs of eosinophilia in the stomach.
4. Eosinophilic esophagitis and gastric sleeve
A physician noted no established absolute contraindication for a 50-year-old patient who was seen for a presleeve gastrectomy. Others contributing to this thread shared concerns for the risk for gastroesophageal reflux disease (GERD) post-surgery and committing the patient to long-term steroids.
More clinical cases and discussions are at https://community.gastro.org/discussions.
about therapy and disease management options, best practices, and diagnoses.
In case you missed it, here are the most popular clinical discussions shared in the forum recently:
1. Active colitis in a patient with previous colon cancer
This popular conversation centers around next steps for a 39-year-old previously treated for a large malignant tumor with a right hemicolectomy and chemotherapy. She was referred for a chromoendoscopy, which revealed four areas of nonpolypoid abnormal mucosa with indistinct borders and abnormal dye uptake. Biopsies also revealed low-grade dysplasia and minimally active colitis.
2. IBD in remission
A 66-year-old female with a history of Crohn’s disease is currently asymptomatic and in remission, but with low Remicade trough. The debate among physicians in the forum questions the need for further action or follow-up insight. 3. Eosinophilic esophagitis and duodenitis (http://ow.ly/kja130mRoDV)
This 40-year-old patient was originally seen for food bolus. The physician prescribed proton pump inhibitors (PPI) after a scope showed typical eosinophilic esophagitis (EoE) findings. Although he had symptoms of obstruction since he was 15, this was his first upper endoscopy. Biopsies following successful scopes showed eosinophilic duodenitis and the patient had no signs of eosinophilia in the stomach.
4. Eosinophilic esophagitis and gastric sleeve
A physician noted no established absolute contraindication for a 50-year-old patient who was seen for a presleeve gastrectomy. Others contributing to this thread shared concerns for the risk for gastroesophageal reflux disease (GERD) post-surgery and committing the patient to long-term steroids.
More clinical cases and discussions are at https://community.gastro.org/discussions.
Can higher MAP post cardiac arrest improve neurologic outcomes?
CHICAGO – A European clinical trial that targeted a mean arterial blood pressure after cardiac arrest higher than what the existing guidelines recommend found that the approach was safe, improved blood flow and oxygen to the brain, helped patients recover quicker, and reduced the number of adverse cardiac events, although it did not reduce the extent of anoxic brain damage or improve functional outcomes, the lead investigator reported at the American Heart Association scientific sessions.
The Neuroprotect trial randomly assigned 112 adult survivors of an out-of-hospital cardiac arrest who were unconscious upon admission to two study groups: early goal-directed hemodynamic optimization (EGDHO), in which researchers used a targeted mean arterial pressure (MAP) of 85-100 mm Hg and mixed venous oxygen saturation between 65% and 75% during the first 36 hours after ICU admission; and the standard care group, in which they used the guideline-recommended MAP target of 65 mm Hg, said Koen Ameloot, MD, of East Limburg Hospital in Genk, Belgium.
“EGDHO clearly improved cerebral perfusion and oxygenation, thereby for the first time providing the proof of concept for this new hemodynamic target,” Dr. Ameloot said. “However, this did not result in the reduction of the extent of anoxic brain hemorrhage or effusion rate on MRI or an improvement in functional outcome at 180 days.”
He noted the trial was predicated on improving upon the so-called “two-hit” model of cardiac arrest sequelae: the first hit being the no-flow and low-flow period before achieving restoration of spontaneous circulation; the second hit being hypoperfusion and reperfusion injury during ICU stay.
Dr. Ameloot referenced a study in which he and other coauthors reported that patients with a MAP target of 65 mm Hg “experience a profound drop of cerebral oxygen saturation during the first 12 hours of ICU stay that may cause additional brain damage” (Resuscitation. 2018;123:92-7).
The researchers explored the question of what is the optimal MAP if a target of 65 mm Hg is too low, Dr. Ameloot said. “We showed that maximal brain oxygenation is achieved with a MAP of 100 mm Hg, while lower MAPs were associated with submaximal brain perfusion and higher MAPs with excessive after-load, a reduction in stroke volume, and suboptimal cerebral oxygenation.”
During the 36-hour intervention period, the EGDHO patients received higher doses of norepinephrine, Dr. Ameloot said. “This resulted in significant improvement of cerebral oxygenation during the first 12 hours and was paralleled by significantly higher cerebral perfusion in the subset of patients in whom Doppler measurements were performed,” he said. “While patients allocated to the MAP 65 mm Hg target experienced a profound drop of cerebral oxygenation during the critical first 6-12 hours of ICU stay, cerebral oxygenation was maintained at 67% in patients assigned to EGDHO.”
However, the rate of anoxic brain damage, measured as the percentage of irreversibly damaged anoxic voxels on diffusion-weighted MRI – the primary endpoint of the study – was actually higher in the EGDHO group, 16% vs. 12%, Dr. Ameloot said. “The percentage of anoxic voxels was only a poor predictor of favorable neurological outcome at 180 days, questioning the validity of the primary endpoint,” he said. He also noted that 23% of the trial participants did not have an MRI scan because of higher than expected 5-day rates of death.
“The percentage of patients with favorable neurological outcome tended to be somewhat higher in the intervention arm, although this did not reach statistical significance at ICU discharge and at 180 days,” Dr. Ameloot said. He noted that 42% of the intervention group and 33% of controls in the full-analysis set (P = .30) and 43% and 27%, respectively, in the per-protocol set (P = .15) had a favorable neurological outcome, as calculated using the Glasgow-Pittsburgh Cerebral Performance Category scores of 1 or 2, at 180 days.
The study did not reveal any noteworthy differences in ICU stay (7 vs. 8 days, P = .13) or days on mechanical ventilation (5 vs. 7, P = .31), although fewer patients in the EGDHO group required a tracheostomy (4% vs. 18%, P = .02). The intervention group also had lower rates of cardiac events, including recurrent cardiac arrest, limb ischemia, new atrial fibrillation, and pulmonary edema (13% vs. 33%; P = .02), Dr. Ameloot said.
Future post-hoc analyses of the data will explore the hypothesis that higher blood pressure leads to improved coronary perfusion and reduced infarct size, thus improving prognosis, he added.
“Should this trial therefore be the definite end to the promising hypothesis that improving brain oxygenation might reduce the second hit in post–cardiac arrest patients? I don’t think so,” Dr. Ameloot said. He noted a few limits to the study: that the perfusion rate on MRI was a poor predictor of 180-day outcome; that more patients than expected entered the trial without receiving basic life support and with nonshockable rhythms; and that there was possibly less extensive brain damage among controls at baseline. “Only an adequately powered clinical trial can provide an answer about the effects of EGDHO in post–cardiac arrest patients,” Dr. Ameloot said.
Dr. Ameloot had no financial relationships to disclose.
SOURCE: Ameloot K et al. AHA 2018, Abstract 18620
CHICAGO – A European clinical trial that targeted a mean arterial blood pressure after cardiac arrest higher than what the existing guidelines recommend found that the approach was safe, improved blood flow and oxygen to the brain, helped patients recover quicker, and reduced the number of adverse cardiac events, although it did not reduce the extent of anoxic brain damage or improve functional outcomes, the lead investigator reported at the American Heart Association scientific sessions.
The Neuroprotect trial randomly assigned 112 adult survivors of an out-of-hospital cardiac arrest who were unconscious upon admission to two study groups: early goal-directed hemodynamic optimization (EGDHO), in which researchers used a targeted mean arterial pressure (MAP) of 85-100 mm Hg and mixed venous oxygen saturation between 65% and 75% during the first 36 hours after ICU admission; and the standard care group, in which they used the guideline-recommended MAP target of 65 mm Hg, said Koen Ameloot, MD, of East Limburg Hospital in Genk, Belgium.
“EGDHO clearly improved cerebral perfusion and oxygenation, thereby for the first time providing the proof of concept for this new hemodynamic target,” Dr. Ameloot said. “However, this did not result in the reduction of the extent of anoxic brain hemorrhage or effusion rate on MRI or an improvement in functional outcome at 180 days.”
He noted the trial was predicated on improving upon the so-called “two-hit” model of cardiac arrest sequelae: the first hit being the no-flow and low-flow period before achieving restoration of spontaneous circulation; the second hit being hypoperfusion and reperfusion injury during ICU stay.
Dr. Ameloot referenced a study in which he and other coauthors reported that patients with a MAP target of 65 mm Hg “experience a profound drop of cerebral oxygen saturation during the first 12 hours of ICU stay that may cause additional brain damage” (Resuscitation. 2018;123:92-7).
The researchers explored the question of what is the optimal MAP if a target of 65 mm Hg is too low, Dr. Ameloot said. “We showed that maximal brain oxygenation is achieved with a MAP of 100 mm Hg, while lower MAPs were associated with submaximal brain perfusion and higher MAPs with excessive after-load, a reduction in stroke volume, and suboptimal cerebral oxygenation.”
During the 36-hour intervention period, the EGDHO patients received higher doses of norepinephrine, Dr. Ameloot said. “This resulted in significant improvement of cerebral oxygenation during the first 12 hours and was paralleled by significantly higher cerebral perfusion in the subset of patients in whom Doppler measurements were performed,” he said. “While patients allocated to the MAP 65 mm Hg target experienced a profound drop of cerebral oxygenation during the critical first 6-12 hours of ICU stay, cerebral oxygenation was maintained at 67% in patients assigned to EGDHO.”
However, the rate of anoxic brain damage, measured as the percentage of irreversibly damaged anoxic voxels on diffusion-weighted MRI – the primary endpoint of the study – was actually higher in the EGDHO group, 16% vs. 12%, Dr. Ameloot said. “The percentage of anoxic voxels was only a poor predictor of favorable neurological outcome at 180 days, questioning the validity of the primary endpoint,” he said. He also noted that 23% of the trial participants did not have an MRI scan because of higher than expected 5-day rates of death.
“The percentage of patients with favorable neurological outcome tended to be somewhat higher in the intervention arm, although this did not reach statistical significance at ICU discharge and at 180 days,” Dr. Ameloot said. He noted that 42% of the intervention group and 33% of controls in the full-analysis set (P = .30) and 43% and 27%, respectively, in the per-protocol set (P = .15) had a favorable neurological outcome, as calculated using the Glasgow-Pittsburgh Cerebral Performance Category scores of 1 or 2, at 180 days.
The study did not reveal any noteworthy differences in ICU stay (7 vs. 8 days, P = .13) or days on mechanical ventilation (5 vs. 7, P = .31), although fewer patients in the EGDHO group required a tracheostomy (4% vs. 18%, P = .02). The intervention group also had lower rates of cardiac events, including recurrent cardiac arrest, limb ischemia, new atrial fibrillation, and pulmonary edema (13% vs. 33%; P = .02), Dr. Ameloot said.
Future post-hoc analyses of the data will explore the hypothesis that higher blood pressure leads to improved coronary perfusion and reduced infarct size, thus improving prognosis, he added.
“Should this trial therefore be the definite end to the promising hypothesis that improving brain oxygenation might reduce the second hit in post–cardiac arrest patients? I don’t think so,” Dr. Ameloot said. He noted a few limits to the study: that the perfusion rate on MRI was a poor predictor of 180-day outcome; that more patients than expected entered the trial without receiving basic life support and with nonshockable rhythms; and that there was possibly less extensive brain damage among controls at baseline. “Only an adequately powered clinical trial can provide an answer about the effects of EGDHO in post–cardiac arrest patients,” Dr. Ameloot said.
Dr. Ameloot had no financial relationships to disclose.
SOURCE: Ameloot K et al. AHA 2018, Abstract 18620
CHICAGO – A European clinical trial that targeted a mean arterial blood pressure after cardiac arrest higher than what the existing guidelines recommend found that the approach was safe, improved blood flow and oxygen to the brain, helped patients recover quicker, and reduced the number of adverse cardiac events, although it did not reduce the extent of anoxic brain damage or improve functional outcomes, the lead investigator reported at the American Heart Association scientific sessions.
The Neuroprotect trial randomly assigned 112 adult survivors of an out-of-hospital cardiac arrest who were unconscious upon admission to two study groups: early goal-directed hemodynamic optimization (EGDHO), in which researchers used a targeted mean arterial pressure (MAP) of 85-100 mm Hg and mixed venous oxygen saturation between 65% and 75% during the first 36 hours after ICU admission; and the standard care group, in which they used the guideline-recommended MAP target of 65 mm Hg, said Koen Ameloot, MD, of East Limburg Hospital in Genk, Belgium.
“EGDHO clearly improved cerebral perfusion and oxygenation, thereby for the first time providing the proof of concept for this new hemodynamic target,” Dr. Ameloot said. “However, this did not result in the reduction of the extent of anoxic brain hemorrhage or effusion rate on MRI or an improvement in functional outcome at 180 days.”
He noted the trial was predicated on improving upon the so-called “two-hit” model of cardiac arrest sequelae: the first hit being the no-flow and low-flow period before achieving restoration of spontaneous circulation; the second hit being hypoperfusion and reperfusion injury during ICU stay.
Dr. Ameloot referenced a study in which he and other coauthors reported that patients with a MAP target of 65 mm Hg “experience a profound drop of cerebral oxygen saturation during the first 12 hours of ICU stay that may cause additional brain damage” (Resuscitation. 2018;123:92-7).
The researchers explored the question of what is the optimal MAP if a target of 65 mm Hg is too low, Dr. Ameloot said. “We showed that maximal brain oxygenation is achieved with a MAP of 100 mm Hg, while lower MAPs were associated with submaximal brain perfusion and higher MAPs with excessive after-load, a reduction in stroke volume, and suboptimal cerebral oxygenation.”
During the 36-hour intervention period, the EGDHO patients received higher doses of norepinephrine, Dr. Ameloot said. “This resulted in significant improvement of cerebral oxygenation during the first 12 hours and was paralleled by significantly higher cerebral perfusion in the subset of patients in whom Doppler measurements were performed,” he said. “While patients allocated to the MAP 65 mm Hg target experienced a profound drop of cerebral oxygenation during the critical first 6-12 hours of ICU stay, cerebral oxygenation was maintained at 67% in patients assigned to EGDHO.”
However, the rate of anoxic brain damage, measured as the percentage of irreversibly damaged anoxic voxels on diffusion-weighted MRI – the primary endpoint of the study – was actually higher in the EGDHO group, 16% vs. 12%, Dr. Ameloot said. “The percentage of anoxic voxels was only a poor predictor of favorable neurological outcome at 180 days, questioning the validity of the primary endpoint,” he said. He also noted that 23% of the trial participants did not have an MRI scan because of higher than expected 5-day rates of death.
“The percentage of patients with favorable neurological outcome tended to be somewhat higher in the intervention arm, although this did not reach statistical significance at ICU discharge and at 180 days,” Dr. Ameloot said. He noted that 42% of the intervention group and 33% of controls in the full-analysis set (P = .30) and 43% and 27%, respectively, in the per-protocol set (P = .15) had a favorable neurological outcome, as calculated using the Glasgow-Pittsburgh Cerebral Performance Category scores of 1 or 2, at 180 days.
The study did not reveal any noteworthy differences in ICU stay (7 vs. 8 days, P = .13) or days on mechanical ventilation (5 vs. 7, P = .31), although fewer patients in the EGDHO group required a tracheostomy (4% vs. 18%, P = .02). The intervention group also had lower rates of cardiac events, including recurrent cardiac arrest, limb ischemia, new atrial fibrillation, and pulmonary edema (13% vs. 33%; P = .02), Dr. Ameloot said.
Future post-hoc analyses of the data will explore the hypothesis that higher blood pressure leads to improved coronary perfusion and reduced infarct size, thus improving prognosis, he added.
“Should this trial therefore be the definite end to the promising hypothesis that improving brain oxygenation might reduce the second hit in post–cardiac arrest patients? I don’t think so,” Dr. Ameloot said. He noted a few limits to the study: that the perfusion rate on MRI was a poor predictor of 180-day outcome; that more patients than expected entered the trial without receiving basic life support and with nonshockable rhythms; and that there was possibly less extensive brain damage among controls at baseline. “Only an adequately powered clinical trial can provide an answer about the effects of EGDHO in post–cardiac arrest patients,” Dr. Ameloot said.
Dr. Ameloot had no financial relationships to disclose.
SOURCE: Ameloot K et al. AHA 2018, Abstract 18620
REPORTING FROM THE AHA SCIENTIFIC SESSIONS
Key clinical point:
Major finding: Forty-three percent of patients in the intervention group had a favorable neurological outcome vs. 27% of controls (P = .15).
Study details: The Neuroprotect trial was a multicenter, randomized, open-label, assessor-blinded trial of 112 post–cardiac arrest patients.
Disclosures: Dr. Ameloot had no financial relationships to disclose.
Source: Ameloot K et al. AHA 2018, Abstract 18620
New editors appointed to CMGH
The next editorial team of the AGA’s basic and translational journal CMGH has been selected. Both editors are from the University of Pennsylvania Perelman School of Medicine. Dr. Kaestner is the Thomas and Evelyn Suor Butterworth Professor in Genetics. Dr. Pack is a professor of medicine and cell and developmental biology and is an attending gastroenterologist at the Hospital of the University of Pennsylvania.
CMGH debuted in 2015 with the mission of publishing impactful digestive biology research that ranges from mechanisms of normal function to pathobiology and covers a broad spectrum of themes in gastroenterology, hepatology, and pancreatology. CMGH is open access, published eight times a year, online-only and is indexed in Medline and PubMed Central. Carrying forward the tremendous success of the journal’s current editor-in-chief, Jerrold R. Turner, MD, PhD, AGAF, and his board of editors, Drs. Kaestner and Pack’s goals include advancing the impact and reputation of CMGH, ensuring the quality and fairness of the peer-review process and introducing new content that is important to the basic and translational research community.
Dr. Kaestner and Dr. Pack’s board of editors includes:
Jonathan Katz, MD
Perelman School of Medicine, University of Pennsylvania
Alison Simmons, MD, PhD
University of Oxford
Frank Tacke, MD, PhD
Rheinisch-Westfälische Technische University
In response to the appointment, Dr. Kaestner said, “We are extremely excited about the opportunity to build on the success of the current editorial team, which has already made CMGH a go-to journal for outstanding digestive disease research. Our goals will be to extend the reach of the journal and to ensure its growth as the premier publication for basic research in gastroenterology and hepatology.”
Dr. Pack added, “The remarkable success achieved by CMGH is a testament to the skill and dedication of Dr. Turner and his associate editors, Drs. Goldenring, Rescigno and Wells, as well as the tremendous growth of impactful basic research in digestive organ biology and disease. Dr. Kaestner and I look forward to maintaining the retiring editorial team’s standard of excellence as we expand the CMGH readership and its visibility in the basic research community.”
Dr. Kaestner and Dr. Pack will begin their term in July 2019.
The next editorial team of the AGA’s basic and translational journal CMGH has been selected. Both editors are from the University of Pennsylvania Perelman School of Medicine. Dr. Kaestner is the Thomas and Evelyn Suor Butterworth Professor in Genetics. Dr. Pack is a professor of medicine and cell and developmental biology and is an attending gastroenterologist at the Hospital of the University of Pennsylvania.
CMGH debuted in 2015 with the mission of publishing impactful digestive biology research that ranges from mechanisms of normal function to pathobiology and covers a broad spectrum of themes in gastroenterology, hepatology, and pancreatology. CMGH is open access, published eight times a year, online-only and is indexed in Medline and PubMed Central. Carrying forward the tremendous success of the journal’s current editor-in-chief, Jerrold R. Turner, MD, PhD, AGAF, and his board of editors, Drs. Kaestner and Pack’s goals include advancing the impact and reputation of CMGH, ensuring the quality and fairness of the peer-review process and introducing new content that is important to the basic and translational research community.
Dr. Kaestner and Dr. Pack’s board of editors includes:
Jonathan Katz, MD
Perelman School of Medicine, University of Pennsylvania
Alison Simmons, MD, PhD
University of Oxford
Frank Tacke, MD, PhD
Rheinisch-Westfälische Technische University
In response to the appointment, Dr. Kaestner said, “We are extremely excited about the opportunity to build on the success of the current editorial team, which has already made CMGH a go-to journal for outstanding digestive disease research. Our goals will be to extend the reach of the journal and to ensure its growth as the premier publication for basic research in gastroenterology and hepatology.”
Dr. Pack added, “The remarkable success achieved by CMGH is a testament to the skill and dedication of Dr. Turner and his associate editors, Drs. Goldenring, Rescigno and Wells, as well as the tremendous growth of impactful basic research in digestive organ biology and disease. Dr. Kaestner and I look forward to maintaining the retiring editorial team’s standard of excellence as we expand the CMGH readership and its visibility in the basic research community.”
Dr. Kaestner and Dr. Pack will begin their term in July 2019.
The next editorial team of the AGA’s basic and translational journal CMGH has been selected. Both editors are from the University of Pennsylvania Perelman School of Medicine. Dr. Kaestner is the Thomas and Evelyn Suor Butterworth Professor in Genetics. Dr. Pack is a professor of medicine and cell and developmental biology and is an attending gastroenterologist at the Hospital of the University of Pennsylvania.
CMGH debuted in 2015 with the mission of publishing impactful digestive biology research that ranges from mechanisms of normal function to pathobiology and covers a broad spectrum of themes in gastroenterology, hepatology, and pancreatology. CMGH is open access, published eight times a year, online-only and is indexed in Medline and PubMed Central. Carrying forward the tremendous success of the journal’s current editor-in-chief, Jerrold R. Turner, MD, PhD, AGAF, and his board of editors, Drs. Kaestner and Pack’s goals include advancing the impact and reputation of CMGH, ensuring the quality and fairness of the peer-review process and introducing new content that is important to the basic and translational research community.
Dr. Kaestner and Dr. Pack’s board of editors includes:
Jonathan Katz, MD
Perelman School of Medicine, University of Pennsylvania
Alison Simmons, MD, PhD
University of Oxford
Frank Tacke, MD, PhD
Rheinisch-Westfälische Technische University
In response to the appointment, Dr. Kaestner said, “We are extremely excited about the opportunity to build on the success of the current editorial team, which has already made CMGH a go-to journal for outstanding digestive disease research. Our goals will be to extend the reach of the journal and to ensure its growth as the premier publication for basic research in gastroenterology and hepatology.”
Dr. Pack added, “The remarkable success achieved by CMGH is a testament to the skill and dedication of Dr. Turner and his associate editors, Drs. Goldenring, Rescigno and Wells, as well as the tremendous growth of impactful basic research in digestive organ biology and disease. Dr. Kaestner and I look forward to maintaining the retiring editorial team’s standard of excellence as we expand the CMGH readership and its visibility in the basic research community.”
Dr. Kaestner and Dr. Pack will begin their term in July 2019.
A clinician’s guide to microbiome testing
The intestinal microbiota, also commonly known as the “gut microbiome” is integral to human physiology and has wide-ranging effects on the development and function of the immune system, energy metabolism and nervous system activity. There is a lot of excitement around the potential of targeting the microbiome therapeutically to promote health and to prevent or treat medical conditions. Further, as DNA sequencing technologies and computational methods continue to improve (as reviewed by Rob Knight and colleagues in a prior editorial), there is significant interest in developing microbiome-based diagnostics for clinical applications.
The industry has recognized the consumer interest in microbiome-based diagnostics as an opportunity, and a number of commercial laboratories are marketing tests directly to patients. Physicians, particularly gastroenterologists, are increasingly being asked by their patients to help interpret such test reports; in some cases, the patient may even request a physician order to purchase the tests for insurance coverage or other reasons.
Earlier this year, AGA members had a robust discussion in the AGA Community about microbiome-based tests, requirements for physician authorization, and the clinical utility (if any) of the results of such tests. below.
Limitations of microbiome sequencing. Microbiome datasets have the same limitations as any other sample-dependent dataset. First and foremost, a single stool sample will tell you something about a person’s microbiome profile only at the time and location that the sample was collected. How the sample was collected and how it was stored may significantly impact the analysis. The analysis generally provides an overview of bacterial families and genera, but little information about the viruses, protozoa and fungi. Furthermore, stool analysis may not reflect well the microbiome composition at the mucosal surface in the intestine. As a result, a single analysis of an individual stool sample merely provides a snapshot of the fecal microbiome that is incomplete and extremely limited in what we can learn from it.
“Good” vs. “bad.” The reports resulting from microbiome-based tests often describe the patient’s microbiome profile in terms of how much “good” and “bad” bacteria are present. This kind of a classification framework represents a naïve and cartoonish view of the microbial world. Instead, it is important to appreciate microbial communities as functional networks, and that their functionality cannot be defined as a mere summation of individual microorganisms. Microbes, just like people, vary their behavior in accordance with the context that may be provided by the activity of other microbes and the host. Whether a particular species or strain is helpful or harmful depends on what other bacteria are present, their density, how they interact with each other (e.g., are they mutually beneficial or competitive?), and factors from the human host such as their diet or immune system activity. For example, Clostridioides difficile is a potential pathogen, yet it also naturally exists in the intestines of many people as a nonharmful, commensal species. Its pathogenic potential depends on the state of the other intestinal microbes and host factors, such as presence of anti-C. difficile toxin antibodies.
Importantly, microbiome tests, which generally provide only a low-resolution microbial community overview, are not designed for pathogen identification. That is best done with targeted diagnostics. Even then, as well illustrated by the C. difficileexample, diagnosis of an infection cannot be made on the basis of laboratory testing alone and requires clinical information.
Taxonomy vs. function. Current technology allows a fairly inexpensive characterization of most bacterial taxa (at family and genus levels). However, taxonomy is not easily translated into functional information. Different taxa of microbes may be able to execute the same chemical transformations. In contrast, functional information depends on the genes present and how much are these genes expressed. However, obtaining this kind of information is much more resource intensive. Measurements of metabolites may also provide very valuable functional information, but proper sample collection for metabolomics is much more difficult.
Interindividual variability. The consistent lesson we’ve learned from the microbiome literature is that there is not a single “healthy” microbiome profile. We have not identified a particular microbiome profile that is predictive of a particular disease, though many researchers are working to develop microbiome-based indices for diseases such as inflammatory bowel disease or obesity. Crowd-sourced studies such as the American Gut Project are working to expand and diversify microbiome datasets so that we can better understand the variability and begin to identify reproducible microbiome signatures. The microbiome data is extremely multidimensional and complex. Therefore, developing predictive patterns will likely require analyses of millions of samples linked to highly granular clinical metadata. Microbiome-based tests have potential to transform clinical care and become incorporated into the personalized medicine paradigm. However, we are at the very beginning of understanding what one’s microbiome profile means for their susceptibility to or progression of disease. As patients approach their health care providers with requests to order commercial microbiome-based tests or to help interpret a report, it is important to set the expectation that these tests are not well suited for diagnoses of infectious diseases or validated in specific diagnoses of any diseases. There are far more unknowns than knowns regarding the role of the microbiome and human health.
For those interested in learning more on this topic, I will be discussing it at the 2019 Gut Microbiota for Health World Summit with my colleague Diane Hoffmann, JD, MS, from the University of Maryland School of Law. The AGA Center for Gut Microbiome Research and Education’s scientific advisory board, on which Diane and I both serve, has also recognized the need for additional guidance. I would encourage my gastroenterology colleagues to continue sharing their experiences with microbiome-based tests through the AGA Community platform.
Recommended reading
- • Staley C, Kaiser T, Khoruts A. Clinician guide to microbiome testing. Dig Dis Sci. 2018 Sep 28. doi: 10.1007/s10620-018-5299-6.
- • Allaband C, McDonald D, Vazquez-Baeza Y, Minich JJ, Tripathi A, Brenner DA, et al. Microbiome 101: Studying, Analyzing, and Interpreting Gut Microbiome Data for Clinicians. Clin Gastroenterol Hepatol 2018. doi: 10.1016/j.cgh.2018.09.017. • Costello EK, Stagaman K, Dethlefsen L, Bohannan BJ, Relman DA. The application of ecological theory toward an understanding of the human microbiome. Science 2012. doi: 10.1126/science.1224203. Epub 2012 Jun 6.
Dr. Khoruts, of the University of Minnesota, is a member of the AGA Center for Gut Microbiome Research & Education scientific advisory board.
The intestinal microbiota, also commonly known as the “gut microbiome” is integral to human physiology and has wide-ranging effects on the development and function of the immune system, energy metabolism and nervous system activity. There is a lot of excitement around the potential of targeting the microbiome therapeutically to promote health and to prevent or treat medical conditions. Further, as DNA sequencing technologies and computational methods continue to improve (as reviewed by Rob Knight and colleagues in a prior editorial), there is significant interest in developing microbiome-based diagnostics for clinical applications.
The industry has recognized the consumer interest in microbiome-based diagnostics as an opportunity, and a number of commercial laboratories are marketing tests directly to patients. Physicians, particularly gastroenterologists, are increasingly being asked by their patients to help interpret such test reports; in some cases, the patient may even request a physician order to purchase the tests for insurance coverage or other reasons.
Earlier this year, AGA members had a robust discussion in the AGA Community about microbiome-based tests, requirements for physician authorization, and the clinical utility (if any) of the results of such tests. below.
Limitations of microbiome sequencing. Microbiome datasets have the same limitations as any other sample-dependent dataset. First and foremost, a single stool sample will tell you something about a person’s microbiome profile only at the time and location that the sample was collected. How the sample was collected and how it was stored may significantly impact the analysis. The analysis generally provides an overview of bacterial families and genera, but little information about the viruses, protozoa and fungi. Furthermore, stool analysis may not reflect well the microbiome composition at the mucosal surface in the intestine. As a result, a single analysis of an individual stool sample merely provides a snapshot of the fecal microbiome that is incomplete and extremely limited in what we can learn from it.
“Good” vs. “bad.” The reports resulting from microbiome-based tests often describe the patient’s microbiome profile in terms of how much “good” and “bad” bacteria are present. This kind of a classification framework represents a naïve and cartoonish view of the microbial world. Instead, it is important to appreciate microbial communities as functional networks, and that their functionality cannot be defined as a mere summation of individual microorganisms. Microbes, just like people, vary their behavior in accordance with the context that may be provided by the activity of other microbes and the host. Whether a particular species or strain is helpful or harmful depends on what other bacteria are present, their density, how they interact with each other (e.g., are they mutually beneficial or competitive?), and factors from the human host such as their diet or immune system activity. For example, Clostridioides difficile is a potential pathogen, yet it also naturally exists in the intestines of many people as a nonharmful, commensal species. Its pathogenic potential depends on the state of the other intestinal microbes and host factors, such as presence of anti-C. difficile toxin antibodies.
Importantly, microbiome tests, which generally provide only a low-resolution microbial community overview, are not designed for pathogen identification. That is best done with targeted diagnostics. Even then, as well illustrated by the C. difficileexample, diagnosis of an infection cannot be made on the basis of laboratory testing alone and requires clinical information.
Taxonomy vs. function. Current technology allows a fairly inexpensive characterization of most bacterial taxa (at family and genus levels). However, taxonomy is not easily translated into functional information. Different taxa of microbes may be able to execute the same chemical transformations. In contrast, functional information depends on the genes present and how much are these genes expressed. However, obtaining this kind of information is much more resource intensive. Measurements of metabolites may also provide very valuable functional information, but proper sample collection for metabolomics is much more difficult.
Interindividual variability. The consistent lesson we’ve learned from the microbiome literature is that there is not a single “healthy” microbiome profile. We have not identified a particular microbiome profile that is predictive of a particular disease, though many researchers are working to develop microbiome-based indices for diseases such as inflammatory bowel disease or obesity. Crowd-sourced studies such as the American Gut Project are working to expand and diversify microbiome datasets so that we can better understand the variability and begin to identify reproducible microbiome signatures. The microbiome data is extremely multidimensional and complex. Therefore, developing predictive patterns will likely require analyses of millions of samples linked to highly granular clinical metadata. Microbiome-based tests have potential to transform clinical care and become incorporated into the personalized medicine paradigm. However, we are at the very beginning of understanding what one’s microbiome profile means for their susceptibility to or progression of disease. As patients approach their health care providers with requests to order commercial microbiome-based tests or to help interpret a report, it is important to set the expectation that these tests are not well suited for diagnoses of infectious diseases or validated in specific diagnoses of any diseases. There are far more unknowns than knowns regarding the role of the microbiome and human health.
For those interested in learning more on this topic, I will be discussing it at the 2019 Gut Microbiota for Health World Summit with my colleague Diane Hoffmann, JD, MS, from the University of Maryland School of Law. The AGA Center for Gut Microbiome Research and Education’s scientific advisory board, on which Diane and I both serve, has also recognized the need for additional guidance. I would encourage my gastroenterology colleagues to continue sharing their experiences with microbiome-based tests through the AGA Community platform.
Recommended reading
- • Staley C, Kaiser T, Khoruts A. Clinician guide to microbiome testing. Dig Dis Sci. 2018 Sep 28. doi: 10.1007/s10620-018-5299-6.
- • Allaband C, McDonald D, Vazquez-Baeza Y, Minich JJ, Tripathi A, Brenner DA, et al. Microbiome 101: Studying, Analyzing, and Interpreting Gut Microbiome Data for Clinicians. Clin Gastroenterol Hepatol 2018. doi: 10.1016/j.cgh.2018.09.017. • Costello EK, Stagaman K, Dethlefsen L, Bohannan BJ, Relman DA. The application of ecological theory toward an understanding of the human microbiome. Science 2012. doi: 10.1126/science.1224203. Epub 2012 Jun 6.
Dr. Khoruts, of the University of Minnesota, is a member of the AGA Center for Gut Microbiome Research & Education scientific advisory board.
The intestinal microbiota, also commonly known as the “gut microbiome” is integral to human physiology and has wide-ranging effects on the development and function of the immune system, energy metabolism and nervous system activity. There is a lot of excitement around the potential of targeting the microbiome therapeutically to promote health and to prevent or treat medical conditions. Further, as DNA sequencing technologies and computational methods continue to improve (as reviewed by Rob Knight and colleagues in a prior editorial), there is significant interest in developing microbiome-based diagnostics for clinical applications.
The industry has recognized the consumer interest in microbiome-based diagnostics as an opportunity, and a number of commercial laboratories are marketing tests directly to patients. Physicians, particularly gastroenterologists, are increasingly being asked by their patients to help interpret such test reports; in some cases, the patient may even request a physician order to purchase the tests for insurance coverage or other reasons.
Earlier this year, AGA members had a robust discussion in the AGA Community about microbiome-based tests, requirements for physician authorization, and the clinical utility (if any) of the results of such tests. below.
Limitations of microbiome sequencing. Microbiome datasets have the same limitations as any other sample-dependent dataset. First and foremost, a single stool sample will tell you something about a person’s microbiome profile only at the time and location that the sample was collected. How the sample was collected and how it was stored may significantly impact the analysis. The analysis generally provides an overview of bacterial families and genera, but little information about the viruses, protozoa and fungi. Furthermore, stool analysis may not reflect well the microbiome composition at the mucosal surface in the intestine. As a result, a single analysis of an individual stool sample merely provides a snapshot of the fecal microbiome that is incomplete and extremely limited in what we can learn from it.
“Good” vs. “bad.” The reports resulting from microbiome-based tests often describe the patient’s microbiome profile in terms of how much “good” and “bad” bacteria are present. This kind of a classification framework represents a naïve and cartoonish view of the microbial world. Instead, it is important to appreciate microbial communities as functional networks, and that their functionality cannot be defined as a mere summation of individual microorganisms. Microbes, just like people, vary their behavior in accordance with the context that may be provided by the activity of other microbes and the host. Whether a particular species or strain is helpful or harmful depends on what other bacteria are present, their density, how they interact with each other (e.g., are they mutually beneficial or competitive?), and factors from the human host such as their diet or immune system activity. For example, Clostridioides difficile is a potential pathogen, yet it also naturally exists in the intestines of many people as a nonharmful, commensal species. Its pathogenic potential depends on the state of the other intestinal microbes and host factors, such as presence of anti-C. difficile toxin antibodies.
Importantly, microbiome tests, which generally provide only a low-resolution microbial community overview, are not designed for pathogen identification. That is best done with targeted diagnostics. Even then, as well illustrated by the C. difficileexample, diagnosis of an infection cannot be made on the basis of laboratory testing alone and requires clinical information.
Taxonomy vs. function. Current technology allows a fairly inexpensive characterization of most bacterial taxa (at family and genus levels). However, taxonomy is not easily translated into functional information. Different taxa of microbes may be able to execute the same chemical transformations. In contrast, functional information depends on the genes present and how much are these genes expressed. However, obtaining this kind of information is much more resource intensive. Measurements of metabolites may also provide very valuable functional information, but proper sample collection for metabolomics is much more difficult.
Interindividual variability. The consistent lesson we’ve learned from the microbiome literature is that there is not a single “healthy” microbiome profile. We have not identified a particular microbiome profile that is predictive of a particular disease, though many researchers are working to develop microbiome-based indices for diseases such as inflammatory bowel disease or obesity. Crowd-sourced studies such as the American Gut Project are working to expand and diversify microbiome datasets so that we can better understand the variability and begin to identify reproducible microbiome signatures. The microbiome data is extremely multidimensional and complex. Therefore, developing predictive patterns will likely require analyses of millions of samples linked to highly granular clinical metadata. Microbiome-based tests have potential to transform clinical care and become incorporated into the personalized medicine paradigm. However, we are at the very beginning of understanding what one’s microbiome profile means for their susceptibility to or progression of disease. As patients approach their health care providers with requests to order commercial microbiome-based tests or to help interpret a report, it is important to set the expectation that these tests are not well suited for diagnoses of infectious diseases or validated in specific diagnoses of any diseases. There are far more unknowns than knowns regarding the role of the microbiome and human health.
For those interested in learning more on this topic, I will be discussing it at the 2019 Gut Microbiota for Health World Summit with my colleague Diane Hoffmann, JD, MS, from the University of Maryland School of Law. The AGA Center for Gut Microbiome Research and Education’s scientific advisory board, on which Diane and I both serve, has also recognized the need for additional guidance. I would encourage my gastroenterology colleagues to continue sharing their experiences with microbiome-based tests through the AGA Community platform.
Recommended reading
- • Staley C, Kaiser T, Khoruts A. Clinician guide to microbiome testing. Dig Dis Sci. 2018 Sep 28. doi: 10.1007/s10620-018-5299-6.
- • Allaband C, McDonald D, Vazquez-Baeza Y, Minich JJ, Tripathi A, Brenner DA, et al. Microbiome 101: Studying, Analyzing, and Interpreting Gut Microbiome Data for Clinicians. Clin Gastroenterol Hepatol 2018. doi: 10.1016/j.cgh.2018.09.017. • Costello EK, Stagaman K, Dethlefsen L, Bohannan BJ, Relman DA. The application of ecological theory toward an understanding of the human microbiome. Science 2012. doi: 10.1126/science.1224203. Epub 2012 Jun 6.
Dr. Khoruts, of the University of Minnesota, is a member of the AGA Center for Gut Microbiome Research & Education scientific advisory board.
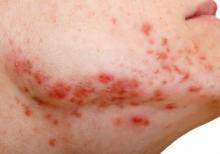
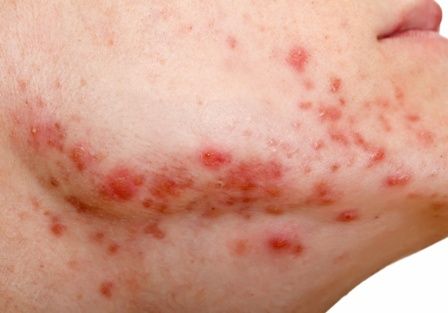
Review looks at effectiveness of isotretinoin for acne
There is when based on reductions in inflammatory lesion counts, according to a recent Cochrane Review.
“The current recommendation of clinical guidelines that oral isotretinoin should be the first-line treatment for moderate to severe acne unresponsive to previous therapies, being more effective than the use of oral antibiotics plus topical agents, underpins current dermatological practice,” Caroline S. Costa, MD, from the Emergency Medicine and Evidence-Based Medicine department at Universidade Federal de São Paulo and her colleagues wrote in their review. “The recommendation cannot be fully supported with certainty by the finding of this review; however, neither does this review challenge it.”
Dr. Costa and her colleagues identified 31 randomized, controlled trials in the Cochrane Skin Group Specialised Register, CENTRAL, MEDLINE, Embase, PsycINFO, and LILACS databases up to July 2017, which altogether included 3,836 participants with mild to severe acne who were aged 12-55 years. The patients received treatment with oral isotretinoin versus placebo or other treatments, such as antibiotics.
In 3 studies that altogether included 400 participants and compared isotretinoin with oral antibiotics plus topical treatments, there was no significant difference with isotretinoin in decreases in the investigator-assessed inflammatory lesion count in participants with moderate to severe acne after 20-24 weeks (risk ratio, 1.01; 95% confidence interval, 0.96-1.06). One serious side effect – Stevens-Johnson syndrome – was noted in the isotretinoin-treated group (RR, 3.00; 95% CI, 0.12-72.98). These results were based on “very-low-quality evidence,” the authors noted.
Two studies also comparing isotretinoin with oral antibiotics and topical treatments in a total of 351 participants showed a 15% improvement in acne severity (RR, 1.15; 95% CI, 1.00-1.32) as assessed by physician’s global evaluation but with a greater number of adverse events considered less severe (RR, 1.67; 95% CI, 1.42-1.98), such as vomiting, nausea, dry lips and skin, and cheilitis.
With regard to dosing, one study with 154 participants with severe acne showed 0.05 mg/kg per day of isotretinoin reduced inflammatory lesion count by 79% after 20 weeks, while 0.1 mg/kg per day and 0.2 mg/kg per day reduced inflammatory lesion counts by 80% and 84%, respectively, after 20 weeks. A different study of 150 participants with severe acne found a 95% decrease in inflammatory lesion counts for 58% of participants receiving 0.1 mg/kg per day, 80% of participants receiving 0.5 mg/kg per day, and 90% of participants receiving 1 mg/kg per day of oral isotretinoin after 20 weeks.
In a randomized, controlled trial with 40 participants with moderate acne comparing intermittent dosing (0.5-0.7 mg/kg per day for 1 week per month), continuous conventional dosing (0.5-0.7 mg/kg per day), and continuous low dosing (0.25-0.4 mg/kg per day) of oral isotretinoin found greater differences in mean decreased numbers of inflammatory lesions among participants in the continuous low dose (a decrease of 3.72 lesions; 95% CI, 2.13-5.31) and continuous conventional dose (a decrease of 3.87 lesions; 95% CI, 2.31-5.43) groups. The authors noted they were unable to perform a meta-analysis because of study heterogeneity in the three studies examining the primary outcome.
In 14 studies with a total of 906 participants who had moderate and severe acne, the authors noted there were no severe adverse events with different doses and regimens of isotretinoin treatment from 12 weeks to 32 weeks or at follow-up at the end of treatment at up to 48 weeks. There were some adverse events, such as skin dryness, hair loss, and itching in 13 studies with a total of 858 participants.
No birth defects were reported in the studies.
The authors took special note of the low-quality evidence in the review, and said that future studies should include larger sample sizes to observe more rare adverse events, as well as create subgroups with results based on acne severity, longer duration and follow-up, standardization of primary outcomes, and adherence to the 2010 CONSORT statement for reporting parallel-group randomized trials.
“With the aim of providing reliable physician guidelines and a robust evidence-based support for daily clinical practice in acne therapy, future randomized clinical trials on oral isotretinoin for acne should focus on treatment of acne when there is insufficient response to therapy with oral antibiotics plus topical agents,” Dr. Costa and her colleagues wrote.
Dr. Costa reports receiving a grant from the Brazilian government granting agency Coordenação de Aperfeicoamento de Pessoal de Nivel Superior. Another author reported a board membership with Bayer for studies and research and is a paid lecturer for a continuing medical education program on adult female acne. The remaining five authors reported no relevant conflicts of interest.
SOURCE: Costa CS et al. Cochrane Database Syst Rev. 2018. doi: 10.1002/14651858.CD009435.pub2.
I think dermatologists would agree with the assertions of the Cochrane Review that there is limited evidence according to today’s standards of quality evidence. The Cochrane Review process examines available data, ranking it by the quality of the data. The paper by Costa et al. shows where the Cochrane Review process is fatally flawed. It assumes that, where the highest quality data is lacking, the medication in question must be ineffective or that this at least casts doubt on its efficacy.
Isotretinoin was approved for use in acne in the 1980s. If you look at one initial report by Peck et al. examining the efficacy of isotretinoin versus placebo in patients with very severe nodulocystic acne, the isotretinoin group was so effective that half of the patients in the placebo arm broke code because of a 57% worsening of their acne and went to the treatment arm. Overall, there was a 95% improvement in the isotretinoin-treated group at the end of treatment (J Am Acad Dermatol. 1982 Apr;6[4 Pt 2 Suppl]:735-45). This study truly reflects how effective isotretinoin is in the treatment of severe acne, and any dermatologist who uses isotretinoin knows that for a fact. I suspect that, if someone applied the Cochrane Review process to insulin, they also would question its efficacy.
I agree there are questionable data on high versus standard versus low dosing. For most patients, standard dosing of 1.0 mg/kg per day over 20-24 weeks will result in complete and sustained clearance. There are always outliers, however, and alternative dosing may be needed – either higher cumulative dosing to achieve clearance or longer lower dosing to manage side effects.
The authors clearly have limited experience in treating acne, or the aim of the article was simply a review of available data with no consideration of actual clinical usage. However, if patients see this report, it could undermine dermatologists’ efforts to care for their patients with acne. It is rather irresponsible to publish these types of reviews, in which the efficacy of the medication in question was established long ago and is clearly without question, and it really discredits the integrity of the Cochrane Review.
Andrea L. Zaenglein, MD , is professor of dermatology and pediatric dermatology at Pennsylvania State University, Hershey, Penn., and is a cochair of the American Academy of Dermatology’s most recent acne management guidelines ( J Am Acad Dermatol. 2016 May;74[5]:945-73.e33 ). She reports being a consultant for Sun Pharmaceutical Industries.
I think dermatologists would agree with the assertions of the Cochrane Review that there is limited evidence according to today’s standards of quality evidence. The Cochrane Review process examines available data, ranking it by the quality of the data. The paper by Costa et al. shows where the Cochrane Review process is fatally flawed. It assumes that, where the highest quality data is lacking, the medication in question must be ineffective or that this at least casts doubt on its efficacy.
Isotretinoin was approved for use in acne in the 1980s. If you look at one initial report by Peck et al. examining the efficacy of isotretinoin versus placebo in patients with very severe nodulocystic acne, the isotretinoin group was so effective that half of the patients in the placebo arm broke code because of a 57% worsening of their acne and went to the treatment arm. Overall, there was a 95% improvement in the isotretinoin-treated group at the end of treatment (J Am Acad Dermatol. 1982 Apr;6[4 Pt 2 Suppl]:735-45). This study truly reflects how effective isotretinoin is in the treatment of severe acne, and any dermatologist who uses isotretinoin knows that for a fact. I suspect that, if someone applied the Cochrane Review process to insulin, they also would question its efficacy.
I agree there are questionable data on high versus standard versus low dosing. For most patients, standard dosing of 1.0 mg/kg per day over 20-24 weeks will result in complete and sustained clearance. There are always outliers, however, and alternative dosing may be needed – either higher cumulative dosing to achieve clearance or longer lower dosing to manage side effects.
The authors clearly have limited experience in treating acne, or the aim of the article was simply a review of available data with no consideration of actual clinical usage. However, if patients see this report, it could undermine dermatologists’ efforts to care for their patients with acne. It is rather irresponsible to publish these types of reviews, in which the efficacy of the medication in question was established long ago and is clearly without question, and it really discredits the integrity of the Cochrane Review.
Andrea L. Zaenglein, MD , is professor of dermatology and pediatric dermatology at Pennsylvania State University, Hershey, Penn., and is a cochair of the American Academy of Dermatology’s most recent acne management guidelines ( J Am Acad Dermatol. 2016 May;74[5]:945-73.e33 ). She reports being a consultant for Sun Pharmaceutical Industries.
I think dermatologists would agree with the assertions of the Cochrane Review that there is limited evidence according to today’s standards of quality evidence. The Cochrane Review process examines available data, ranking it by the quality of the data. The paper by Costa et al. shows where the Cochrane Review process is fatally flawed. It assumes that, where the highest quality data is lacking, the medication in question must be ineffective or that this at least casts doubt on its efficacy.
Isotretinoin was approved for use in acne in the 1980s. If you look at one initial report by Peck et al. examining the efficacy of isotretinoin versus placebo in patients with very severe nodulocystic acne, the isotretinoin group was so effective that half of the patients in the placebo arm broke code because of a 57% worsening of their acne and went to the treatment arm. Overall, there was a 95% improvement in the isotretinoin-treated group at the end of treatment (J Am Acad Dermatol. 1982 Apr;6[4 Pt 2 Suppl]:735-45). This study truly reflects how effective isotretinoin is in the treatment of severe acne, and any dermatologist who uses isotretinoin knows that for a fact. I suspect that, if someone applied the Cochrane Review process to insulin, they also would question its efficacy.
I agree there are questionable data on high versus standard versus low dosing. For most patients, standard dosing of 1.0 mg/kg per day over 20-24 weeks will result in complete and sustained clearance. There are always outliers, however, and alternative dosing may be needed – either higher cumulative dosing to achieve clearance or longer lower dosing to manage side effects.
The authors clearly have limited experience in treating acne, or the aim of the article was simply a review of available data with no consideration of actual clinical usage. However, if patients see this report, it could undermine dermatologists’ efforts to care for their patients with acne. It is rather irresponsible to publish these types of reviews, in which the efficacy of the medication in question was established long ago and is clearly without question, and it really discredits the integrity of the Cochrane Review.
Andrea L. Zaenglein, MD , is professor of dermatology and pediatric dermatology at Pennsylvania State University, Hershey, Penn., and is a cochair of the American Academy of Dermatology’s most recent acne management guidelines ( J Am Acad Dermatol. 2016 May;74[5]:945-73.e33 ). She reports being a consultant for Sun Pharmaceutical Industries.
There is when based on reductions in inflammatory lesion counts, according to a recent Cochrane Review.
“The current recommendation of clinical guidelines that oral isotretinoin should be the first-line treatment for moderate to severe acne unresponsive to previous therapies, being more effective than the use of oral antibiotics plus topical agents, underpins current dermatological practice,” Caroline S. Costa, MD, from the Emergency Medicine and Evidence-Based Medicine department at Universidade Federal de São Paulo and her colleagues wrote in their review. “The recommendation cannot be fully supported with certainty by the finding of this review; however, neither does this review challenge it.”
Dr. Costa and her colleagues identified 31 randomized, controlled trials in the Cochrane Skin Group Specialised Register, CENTRAL, MEDLINE, Embase, PsycINFO, and LILACS databases up to July 2017, which altogether included 3,836 participants with mild to severe acne who were aged 12-55 years. The patients received treatment with oral isotretinoin versus placebo or other treatments, such as antibiotics.
In 3 studies that altogether included 400 participants and compared isotretinoin with oral antibiotics plus topical treatments, there was no significant difference with isotretinoin in decreases in the investigator-assessed inflammatory lesion count in participants with moderate to severe acne after 20-24 weeks (risk ratio, 1.01; 95% confidence interval, 0.96-1.06). One serious side effect – Stevens-Johnson syndrome – was noted in the isotretinoin-treated group (RR, 3.00; 95% CI, 0.12-72.98). These results were based on “very-low-quality evidence,” the authors noted.
Two studies also comparing isotretinoin with oral antibiotics and topical treatments in a total of 351 participants showed a 15% improvement in acne severity (RR, 1.15; 95% CI, 1.00-1.32) as assessed by physician’s global evaluation but with a greater number of adverse events considered less severe (RR, 1.67; 95% CI, 1.42-1.98), such as vomiting, nausea, dry lips and skin, and cheilitis.
With regard to dosing, one study with 154 participants with severe acne showed 0.05 mg/kg per day of isotretinoin reduced inflammatory lesion count by 79% after 20 weeks, while 0.1 mg/kg per day and 0.2 mg/kg per day reduced inflammatory lesion counts by 80% and 84%, respectively, after 20 weeks. A different study of 150 participants with severe acne found a 95% decrease in inflammatory lesion counts for 58% of participants receiving 0.1 mg/kg per day, 80% of participants receiving 0.5 mg/kg per day, and 90% of participants receiving 1 mg/kg per day of oral isotretinoin after 20 weeks.
In a randomized, controlled trial with 40 participants with moderate acne comparing intermittent dosing (0.5-0.7 mg/kg per day for 1 week per month), continuous conventional dosing (0.5-0.7 mg/kg per day), and continuous low dosing (0.25-0.4 mg/kg per day) of oral isotretinoin found greater differences in mean decreased numbers of inflammatory lesions among participants in the continuous low dose (a decrease of 3.72 lesions; 95% CI, 2.13-5.31) and continuous conventional dose (a decrease of 3.87 lesions; 95% CI, 2.31-5.43) groups. The authors noted they were unable to perform a meta-analysis because of study heterogeneity in the three studies examining the primary outcome.
In 14 studies with a total of 906 participants who had moderate and severe acne, the authors noted there were no severe adverse events with different doses and regimens of isotretinoin treatment from 12 weeks to 32 weeks or at follow-up at the end of treatment at up to 48 weeks. There were some adverse events, such as skin dryness, hair loss, and itching in 13 studies with a total of 858 participants.
No birth defects were reported in the studies.
The authors took special note of the low-quality evidence in the review, and said that future studies should include larger sample sizes to observe more rare adverse events, as well as create subgroups with results based on acne severity, longer duration and follow-up, standardization of primary outcomes, and adherence to the 2010 CONSORT statement for reporting parallel-group randomized trials.
“With the aim of providing reliable physician guidelines and a robust evidence-based support for daily clinical practice in acne therapy, future randomized clinical trials on oral isotretinoin for acne should focus on treatment of acne when there is insufficient response to therapy with oral antibiotics plus topical agents,” Dr. Costa and her colleagues wrote.
Dr. Costa reports receiving a grant from the Brazilian government granting agency Coordenação de Aperfeicoamento de Pessoal de Nivel Superior. Another author reported a board membership with Bayer for studies and research and is a paid lecturer for a continuing medical education program on adult female acne. The remaining five authors reported no relevant conflicts of interest.
SOURCE: Costa CS et al. Cochrane Database Syst Rev. 2018. doi: 10.1002/14651858.CD009435.pub2.
There is when based on reductions in inflammatory lesion counts, according to a recent Cochrane Review.
“The current recommendation of clinical guidelines that oral isotretinoin should be the first-line treatment for moderate to severe acne unresponsive to previous therapies, being more effective than the use of oral antibiotics plus topical agents, underpins current dermatological practice,” Caroline S. Costa, MD, from the Emergency Medicine and Evidence-Based Medicine department at Universidade Federal de São Paulo and her colleagues wrote in their review. “The recommendation cannot be fully supported with certainty by the finding of this review; however, neither does this review challenge it.”
Dr. Costa and her colleagues identified 31 randomized, controlled trials in the Cochrane Skin Group Specialised Register, CENTRAL, MEDLINE, Embase, PsycINFO, and LILACS databases up to July 2017, which altogether included 3,836 participants with mild to severe acne who were aged 12-55 years. The patients received treatment with oral isotretinoin versus placebo or other treatments, such as antibiotics.
In 3 studies that altogether included 400 participants and compared isotretinoin with oral antibiotics plus topical treatments, there was no significant difference with isotretinoin in decreases in the investigator-assessed inflammatory lesion count in participants with moderate to severe acne after 20-24 weeks (risk ratio, 1.01; 95% confidence interval, 0.96-1.06). One serious side effect – Stevens-Johnson syndrome – was noted in the isotretinoin-treated group (RR, 3.00; 95% CI, 0.12-72.98). These results were based on “very-low-quality evidence,” the authors noted.
Two studies also comparing isotretinoin with oral antibiotics and topical treatments in a total of 351 participants showed a 15% improvement in acne severity (RR, 1.15; 95% CI, 1.00-1.32) as assessed by physician’s global evaluation but with a greater number of adverse events considered less severe (RR, 1.67; 95% CI, 1.42-1.98), such as vomiting, nausea, dry lips and skin, and cheilitis.
With regard to dosing, one study with 154 participants with severe acne showed 0.05 mg/kg per day of isotretinoin reduced inflammatory lesion count by 79% after 20 weeks, while 0.1 mg/kg per day and 0.2 mg/kg per day reduced inflammatory lesion counts by 80% and 84%, respectively, after 20 weeks. A different study of 150 participants with severe acne found a 95% decrease in inflammatory lesion counts for 58% of participants receiving 0.1 mg/kg per day, 80% of participants receiving 0.5 mg/kg per day, and 90% of participants receiving 1 mg/kg per day of oral isotretinoin after 20 weeks.
In a randomized, controlled trial with 40 participants with moderate acne comparing intermittent dosing (0.5-0.7 mg/kg per day for 1 week per month), continuous conventional dosing (0.5-0.7 mg/kg per day), and continuous low dosing (0.25-0.4 mg/kg per day) of oral isotretinoin found greater differences in mean decreased numbers of inflammatory lesions among participants in the continuous low dose (a decrease of 3.72 lesions; 95% CI, 2.13-5.31) and continuous conventional dose (a decrease of 3.87 lesions; 95% CI, 2.31-5.43) groups. The authors noted they were unable to perform a meta-analysis because of study heterogeneity in the three studies examining the primary outcome.
In 14 studies with a total of 906 participants who had moderate and severe acne, the authors noted there were no severe adverse events with different doses and regimens of isotretinoin treatment from 12 weeks to 32 weeks or at follow-up at the end of treatment at up to 48 weeks. There were some adverse events, such as skin dryness, hair loss, and itching in 13 studies with a total of 858 participants.
No birth defects were reported in the studies.
The authors took special note of the low-quality evidence in the review, and said that future studies should include larger sample sizes to observe more rare adverse events, as well as create subgroups with results based on acne severity, longer duration and follow-up, standardization of primary outcomes, and adherence to the 2010 CONSORT statement for reporting parallel-group randomized trials.
“With the aim of providing reliable physician guidelines and a robust evidence-based support for daily clinical practice in acne therapy, future randomized clinical trials on oral isotretinoin for acne should focus on treatment of acne when there is insufficient response to therapy with oral antibiotics plus topical agents,” Dr. Costa and her colleagues wrote.
Dr. Costa reports receiving a grant from the Brazilian government granting agency Coordenação de Aperfeicoamento de Pessoal de Nivel Superior. Another author reported a board membership with Bayer for studies and research and is a paid lecturer for a continuing medical education program on adult female acne. The remaining five authors reported no relevant conflicts of interest.
SOURCE: Costa CS et al. Cochrane Database Syst Rev. 2018. doi: 10.1002/14651858.CD009435.pub2.
FROM COCHRANE DATABASE OF SYSTEMATIC REVIEWS
Key clinical point: A Cochrane Review found low quality of evidence surrounding oral isotretinoin’s effectiveness for acne.
Major finding: Isotretinoin did not decrease inflammatory lesion count, compared with (risk ratio, 1.01; 95% confidence interval, 0.96-1.06), in 3 studies, and there was low evidence to suggest daily treatment was more effective than treatment for 1 week per month.
Study details: A Cochrane Review of 31 studies that altogether included 3,836 patients with mild to severe acne who received oral isotretinoin, placebo, or other treatments such as antibiotics.
Disclosures: Dr. Costa reported receiving a grant from the Brazilian government granting agency Coordenação de Aperfeicoamento de Pessoal de Nivel Superior. Dr. Bagatin reported a board membership with Bayer for studies and research and is a paid lecturer for a continuing medical education program on adult female acne. The other authors report no relevant conflicts of interest.
Source: Costa CS et al. Cochrane Database Syst Rev. 2018. doi: 10.1002/14651858.CD009435.pub2.
Atopic dermatitis associated with increased suicidality
Patients with atopic dermatitis might face up to a 44% increased risk of suicidal ideation and are 36% more likely to attempt suicide than those without the disorder, a large meta-analysis has determined.
The analysis, which included data from studies published as far back as 1945, also found some correlation of increased suicide risk and increasing disease severity, although the numbers were small, Jeena K. Sandhu and her colleagues reported in JAMA Dermatology.
Both physical and psychological factors could be involved in the link, wrote Ms. Sandhu, a medical student at the University of Missouri–Kansas City, and her coauthors.
“Atopic dermatitis is associated with multiple physical comorbidities, such as asthma, allergic rhinitis, metabolic syndrome, and sleep disturbances, which all contribute to the overall physical burden of the disease. Many patients also have a profound psychosocial burden. Because of the visibility of the disease, patients may experience shame, embarrassment, and stigmatization,” they wrote.
But the disease also is associated with high levels of proinflammatory cytokines, and those proteins have been isolated in the cerebrospinal fluid of patients who have attempted suicide, the investigators noted. “Treatments targeting cytokines, such as interleukin-4 and interleukin-13, have been shown to decrease symptoms of depression and anxiety in patients with atopic dermatitis.”
The investigators plumbed several databases of medical literature, searching for studies that mentioned both atopic dermatitis (AD) and suicide, suicidal ideation, or suicidal behavior. They found 15 studies, published from 1945 to May 2018. Most (13) were cross sectional; the remainder were cohort studies. Together, they comprised a total of 4.7 million subjects, 310,681 of whom had AD. The analysis looked at risks in three areas: suicidal ideation, suicide attempts, and completed suicides.
Of the studies, 11 investigated suicidal ideation. Pooled data determined that patients with AD were a significant 44% more likely to experience suicidal ideation than those without the disease.
Three studies mentioned suicide attempts and had complete data for pooling. Taken together, they showed a significant 36% increased risk of attempted suicide among patients with AD, compared with those without the disorder.
Two studies investigated the prevalence of completed suicides among patients. One did report a significantly increased risk of 40%, compared with the control group, but it failed to report the number of suicides in the control group. The other study found no increased risk of completed suicides in patients with either mild or moderate to severe disease, compared with controls.
Two studies involved only pediatric patients. One, conducted in Korea, found a significant 23% increased risk of suicidal ideation and a 31% increased risk of attempted suicide. The other failed to find any increased risks in the overall analysis, but did find small increases in the risks of ideation and attempt in girls with AD, compared with healthy controls.
the team concluded. “Dermatology providers may use several tools to screen patients for suicidality. Asking patients about suicidal ideation with a question may be integrated into a patient visit. If a patient screens positive for suicidality, the dermatology provider should send a referral to the patient’s primary care or mental health provider for follow-up care. If the patient reports an orchestrated plan to commit suicide, this patient should be urgently referred to the emergency department for further assessment.”
Ms. Sandhu reported no financial disclosures.
SOURCE: Sandhu JK et al. JAMA Dermatol. 2018 Dec 12. doi: 10.1001/jamadermatol.2018.4566.
Patients with atopic dermatitis might face up to a 44% increased risk of suicidal ideation and are 36% more likely to attempt suicide than those without the disorder, a large meta-analysis has determined.
The analysis, which included data from studies published as far back as 1945, also found some correlation of increased suicide risk and increasing disease severity, although the numbers were small, Jeena K. Sandhu and her colleagues reported in JAMA Dermatology.
Both physical and psychological factors could be involved in the link, wrote Ms. Sandhu, a medical student at the University of Missouri–Kansas City, and her coauthors.
“Atopic dermatitis is associated with multiple physical comorbidities, such as asthma, allergic rhinitis, metabolic syndrome, and sleep disturbances, which all contribute to the overall physical burden of the disease. Many patients also have a profound psychosocial burden. Because of the visibility of the disease, patients may experience shame, embarrassment, and stigmatization,” they wrote.
But the disease also is associated with high levels of proinflammatory cytokines, and those proteins have been isolated in the cerebrospinal fluid of patients who have attempted suicide, the investigators noted. “Treatments targeting cytokines, such as interleukin-4 and interleukin-13, have been shown to decrease symptoms of depression and anxiety in patients with atopic dermatitis.”
The investigators plumbed several databases of medical literature, searching for studies that mentioned both atopic dermatitis (AD) and suicide, suicidal ideation, or suicidal behavior. They found 15 studies, published from 1945 to May 2018. Most (13) were cross sectional; the remainder were cohort studies. Together, they comprised a total of 4.7 million subjects, 310,681 of whom had AD. The analysis looked at risks in three areas: suicidal ideation, suicide attempts, and completed suicides.
Of the studies, 11 investigated suicidal ideation. Pooled data determined that patients with AD were a significant 44% more likely to experience suicidal ideation than those without the disease.
Three studies mentioned suicide attempts and had complete data for pooling. Taken together, they showed a significant 36% increased risk of attempted suicide among patients with AD, compared with those without the disorder.
Two studies investigated the prevalence of completed suicides among patients. One did report a significantly increased risk of 40%, compared with the control group, but it failed to report the number of suicides in the control group. The other study found no increased risk of completed suicides in patients with either mild or moderate to severe disease, compared with controls.
Two studies involved only pediatric patients. One, conducted in Korea, found a significant 23% increased risk of suicidal ideation and a 31% increased risk of attempted suicide. The other failed to find any increased risks in the overall analysis, but did find small increases in the risks of ideation and attempt in girls with AD, compared with healthy controls.
the team concluded. “Dermatology providers may use several tools to screen patients for suicidality. Asking patients about suicidal ideation with a question may be integrated into a patient visit. If a patient screens positive for suicidality, the dermatology provider should send a referral to the patient’s primary care or mental health provider for follow-up care. If the patient reports an orchestrated plan to commit suicide, this patient should be urgently referred to the emergency department for further assessment.”
Ms. Sandhu reported no financial disclosures.
SOURCE: Sandhu JK et al. JAMA Dermatol. 2018 Dec 12. doi: 10.1001/jamadermatol.2018.4566.
Patients with atopic dermatitis might face up to a 44% increased risk of suicidal ideation and are 36% more likely to attempt suicide than those without the disorder, a large meta-analysis has determined.
The analysis, which included data from studies published as far back as 1945, also found some correlation of increased suicide risk and increasing disease severity, although the numbers were small, Jeena K. Sandhu and her colleagues reported in JAMA Dermatology.
Both physical and psychological factors could be involved in the link, wrote Ms. Sandhu, a medical student at the University of Missouri–Kansas City, and her coauthors.
“Atopic dermatitis is associated with multiple physical comorbidities, such as asthma, allergic rhinitis, metabolic syndrome, and sleep disturbances, which all contribute to the overall physical burden of the disease. Many patients also have a profound psychosocial burden. Because of the visibility of the disease, patients may experience shame, embarrassment, and stigmatization,” they wrote.
But the disease also is associated with high levels of proinflammatory cytokines, and those proteins have been isolated in the cerebrospinal fluid of patients who have attempted suicide, the investigators noted. “Treatments targeting cytokines, such as interleukin-4 and interleukin-13, have been shown to decrease symptoms of depression and anxiety in patients with atopic dermatitis.”
The investigators plumbed several databases of medical literature, searching for studies that mentioned both atopic dermatitis (AD) and suicide, suicidal ideation, or suicidal behavior. They found 15 studies, published from 1945 to May 2018. Most (13) were cross sectional; the remainder were cohort studies. Together, they comprised a total of 4.7 million subjects, 310,681 of whom had AD. The analysis looked at risks in three areas: suicidal ideation, suicide attempts, and completed suicides.
Of the studies, 11 investigated suicidal ideation. Pooled data determined that patients with AD were a significant 44% more likely to experience suicidal ideation than those without the disease.
Three studies mentioned suicide attempts and had complete data for pooling. Taken together, they showed a significant 36% increased risk of attempted suicide among patients with AD, compared with those without the disorder.
Two studies investigated the prevalence of completed suicides among patients. One did report a significantly increased risk of 40%, compared with the control group, but it failed to report the number of suicides in the control group. The other study found no increased risk of completed suicides in patients with either mild or moderate to severe disease, compared with controls.
Two studies involved only pediatric patients. One, conducted in Korea, found a significant 23% increased risk of suicidal ideation and a 31% increased risk of attempted suicide. The other failed to find any increased risks in the overall analysis, but did find small increases in the risks of ideation and attempt in girls with AD, compared with healthy controls.
the team concluded. “Dermatology providers may use several tools to screen patients for suicidality. Asking patients about suicidal ideation with a question may be integrated into a patient visit. If a patient screens positive for suicidality, the dermatology provider should send a referral to the patient’s primary care or mental health provider for follow-up care. If the patient reports an orchestrated plan to commit suicide, this patient should be urgently referred to the emergency department for further assessment.”
Ms. Sandhu reported no financial disclosures.
SOURCE: Sandhu JK et al. JAMA Dermatol. 2018 Dec 12. doi: 10.1001/jamadermatol.2018.4566.
FROM JAMA DERMATOLOGY
Key clinical point: Suicidal ideation and suicide attempts seem to be more common among people with atopic dermatitis than those without the disease.
Major finding: Patients were 44% more likely to have suicidal ideation and 36% more likely to attempt suicide.
Study details: The meta-analysis comprised 15 studies with a total of 4.7 million participants, 310,681 of whom had the disease.
Disclosures: Ms. Sandhu reported no financial disclosures.
Source: Sandhu JK et al. JAMA Dermatol. 2018 Dec 12. doi: 10.1001/jamadermatol.2018.4566.
FDA reclassifies ECT devices for resistant depression, other conditions
The Food and Drug Administration issued a final order Dec. 21 reclassifying electroconvulsive therapy (ECT) devices from class III, indicating higher risk, to class II, indicating moderate risk, in certain cases.
Conditions included in the new order are catatonia or a severe major depressive episode associated with major depressive disorder or bipolar disorder in patients over the age of 13 years who are resistant to treatment or who require a rapid response because of the severity of their psychiatric or medical condition, according to an FDA press release.
In addition, the final order requires the filing of premarket approval application for class III devices used for all conditions not reclassified as class II.
“The FDA is issuing this final order to regulate ECT devices in a way that appropriately reflects the known benefits and risks of these devices for their indications for use, provides patients with additional protections, and gives physicians more information on the safe and effective use of these devices,” the agency said in the press release.
The Food and Drug Administration issued a final order Dec. 21 reclassifying electroconvulsive therapy (ECT) devices from class III, indicating higher risk, to class II, indicating moderate risk, in certain cases.
Conditions included in the new order are catatonia or a severe major depressive episode associated with major depressive disorder or bipolar disorder in patients over the age of 13 years who are resistant to treatment or who require a rapid response because of the severity of their psychiatric or medical condition, according to an FDA press release.
In addition, the final order requires the filing of premarket approval application for class III devices used for all conditions not reclassified as class II.
“The FDA is issuing this final order to regulate ECT devices in a way that appropriately reflects the known benefits and risks of these devices for their indications for use, provides patients with additional protections, and gives physicians more information on the safe and effective use of these devices,” the agency said in the press release.
The Food and Drug Administration issued a final order Dec. 21 reclassifying electroconvulsive therapy (ECT) devices from class III, indicating higher risk, to class II, indicating moderate risk, in certain cases.
Conditions included in the new order are catatonia or a severe major depressive episode associated with major depressive disorder or bipolar disorder in patients over the age of 13 years who are resistant to treatment or who require a rapid response because of the severity of their psychiatric or medical condition, according to an FDA press release.
In addition, the final order requires the filing of premarket approval application for class III devices used for all conditions not reclassified as class II.
“The FDA is issuing this final order to regulate ECT devices in a way that appropriately reflects the known benefits and risks of these devices for their indications for use, provides patients with additional protections, and gives physicians more information on the safe and effective use of these devices,” the agency said in the press release.