User login
The case for longer treatment in MM: Part 2
In Part 2 of this editorial, Katja Weisel, MD, of University Hospital Tubingen in Germany, addresses the barriers to longer treatment in patients with multiple myeloma.
Attitudes regarding longer treatment can present barriers to widespread adoption of this approach in multiple myeloma (MM).
Indeed, some clinicians continue to follow a fixed-duration approach to treatment in MM, only considering further treatment once the patient has relapsed rather than treating the patient until disease progression.
In the MM community, some are reluctant to adopt a strategy of treating longer because of the modest efficacy gains observed with early research or concern over tolerability issues, including the risk of developing peripheral neuropathy or secondary malignancies.1
Others are uncertain about the optimal duration of therapy or the selection of an agent that will balance any potential gain in depth of response with the risk of late-onset or cumulative toxicities.
The potentially high cost of longer treatment for patients, their families, and/or the healthcare system overall also presents a challenge.
It is feasible that treating patients for longer may drive up healthcare utilization and take a toll on patients and caregivers, who may incur out-of-pocket costs because of the need to travel to a hospital or doctor’s office for intravenous therapies, requiring them to miss work.2
It is important to recognize, however, that more convenient all-oral treatment regimens are now available that do not require infusion at a hospital or clinic. Furthermore, results from recent studies suggest the majority of cancer patients prefer oral over intravenous therapies, which could reduce non-pharmacy healthcare costs.3,4
Healthcare providers might be more likely to accept and adopt a longer treatment approach for MM if they had access to data describing the optimal duration, dosage, schedule, toxicity, and quality of life standards.
Ongoing, randomized, phase 3 trials are evaluating the benefits of treating longer with an oral proteasome inhibitor in patients with newly diagnosed MM.5,6
Updated treatment guidelines and consensus statements will provide further guidance for clinicians on the benefits of maintenance therapy in both transplant-eligible and -ineligible patients with newly diagnosed MM.
The recently updated MM guidelines from the European Society for Medical Oncology (ESMO) recommend longer treatment or maintenance therapy in patients who have undergone hematopoietic stem cell transplant (HSCT).7
Based on evidence from studies such as FIRST and SWOG S0777, ESMO also recommends continuous treatment or treatment until progression with lenalidomide-dexamethasone and bortezomib-lenalidomide-dexamethasone in MM patients who are ineligible for HSCT.7-9
As there is no one-size-fits-all treatment approach in MM, a personalized treatment plan should be designed for each patient. This plan should take into account a number of factors, including age, disease characteristics, performance status, treatment history, and the patient’s goals of care and personal preferences.10
If the patient is a candidate for longer treatment, the clinician should carefully weigh the potential impact on disease-free and overall survival against the potential side effects, as well as assess the patient’s likelihood of adhering to the medication.
With the availability of newer, less-toxic medications that can be tolerated for a greater duration and are easy to administer, aiding in overall treatment compliance, sustained remissions are possible.11-13
Forty years ago, MM patients had very few treatment options, and the 5-year survival rate was 26%.14
Since then, novel therapies, including proteasome inhibitors and immunomodulatory drugs, have replaced conventional cytotoxic chemotherapy, leading to major improvements in survival.15,16
With emerging research that supports the value of longer treatment strategies for both patients and the healthcare system, clinicians will have a proven strategy to help their patients attain long-term disease control while maintaining quality of life.2, 17-19
Dr. Weisel has received honoraria and/or consultancy fees from Amgen, BMS, Celgene, Janssen, Juno, Sanofi, and Takeda. She has received research funding from Amgen, Celgene, Sanofi, and Janssen.
The W2O Group provided writing support for this editorial, which was funded by Millennium Pharmaceuticals Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited.
1. Lipe B et al. Blood Cancer J. 2016; 6(10): e485. doi: 10.1038/bcj.2016.89
2. Goodwin J et al. Cancer Nurs. 2013; 36(4):301-8. doi: 10.1097/NCC.0b013e3182693522
3. Eek D et al. Patient Prefer Adherence. 2016; 10:1609-21. doi: 10.2147/PPA.S106629
4. Bauer S et al. Value in Health. 2017; 20: A451. Abstract PCN217. doi: https://doi.org/10.1016/j.jval.2017.08.299
5. A Study of Oral Ixazomib Citrate (MLN9708) Maintenance Therapy in Participants With Multiple Myeloma Following Autologous Stem Cell Transplant. (2014). Retrieved from https://clinicaltrials.gov/ct2/show/NCT02181413 (Identification No. NCT02181413).
6. A Study of Oral Ixazomib Maintenance Therapy in Patients With Newly Diagnosed Multiple Myeloma Not Treated With Stem Cell Transplantation. (2014). Retrieved from https://clinicaltrials.gov/ct2/show/NCT02312258 (Identification No. NCT02312258).
7. Moreau P et al. Ann Oncol. 2017; 28: iv52-iv61. doi: https://org/10.1093/annonc/mdx096
8. Facon T et al. Blood. 2018131(3):301-310. doi: 10.1182/blood-2017-07-795047
9. Durie BG et al. Lancet. 2017; 389(10068):519-527. doi: 10.1016/S0140-6736(16)31594-X.
10. Laubach J et al. Leukemia. 2016; 30(5):1005-17. doi: 10.1038/leu.2015.356
11. Ludwig H et al. Blood. 2012; 119: 3003-3015. doi: https://doi.org/10.1182/blood-2011-11-374249
12. Lehners N et al. Cancer Med. 2018; 7(2): 307–316. doi: 10.1002/cam4.1283
13. Attal M et al. N Engl J Med. 2012; 366:1872-1791. doi: 10.1056/NEJMoa1114138
14. National Cancer Institute. SEER Cancer Statistics Review (CSR) 1975-2014. National Cancer Institute. https://seer.cancer.gov/csr/1975_2014/. Accessed March 28, 2018.
15. Kumar SK et al. Blood. 2008 Mar 1;111(5):2516-20. doi: 10.1182/blood-2007-10-116129
16. Fonseca R et al. Leukemia. 2017 Sep;31(9):1915-1921. doi: 10.1038/leu.2016.380
17. Palumbo A, Niesvizky R. Leuk Res. 2012; 36 Suppl 1:S19-26. doi: 10.1016/S0145-2126(12)70005-X
18. Girnius S, Munshi NC. Leuk Suppl. 2013; 2(Suppl 1): S3–S9. doi: 10.1038/leusup.2013.2
19. Mateos M-V, San Miguel JF. Hematology Am Soc Hematol Educ Program. 2013; 2013:488-95. doi: 10.1182/asheducation-2013.1.488
In Part 2 of this editorial, Katja Weisel, MD, of University Hospital Tubingen in Germany, addresses the barriers to longer treatment in patients with multiple myeloma.
Attitudes regarding longer treatment can present barriers to widespread adoption of this approach in multiple myeloma (MM).
Indeed, some clinicians continue to follow a fixed-duration approach to treatment in MM, only considering further treatment once the patient has relapsed rather than treating the patient until disease progression.
In the MM community, some are reluctant to adopt a strategy of treating longer because of the modest efficacy gains observed with early research or concern over tolerability issues, including the risk of developing peripheral neuropathy or secondary malignancies.1
Others are uncertain about the optimal duration of therapy or the selection of an agent that will balance any potential gain in depth of response with the risk of late-onset or cumulative toxicities.
The potentially high cost of longer treatment for patients, their families, and/or the healthcare system overall also presents a challenge.
It is feasible that treating patients for longer may drive up healthcare utilization and take a toll on patients and caregivers, who may incur out-of-pocket costs because of the need to travel to a hospital or doctor’s office for intravenous therapies, requiring them to miss work.2
It is important to recognize, however, that more convenient all-oral treatment regimens are now available that do not require infusion at a hospital or clinic. Furthermore, results from recent studies suggest the majority of cancer patients prefer oral over intravenous therapies, which could reduce non-pharmacy healthcare costs.3,4
Healthcare providers might be more likely to accept and adopt a longer treatment approach for MM if they had access to data describing the optimal duration, dosage, schedule, toxicity, and quality of life standards.
Ongoing, randomized, phase 3 trials are evaluating the benefits of treating longer with an oral proteasome inhibitor in patients with newly diagnosed MM.5,6
Updated treatment guidelines and consensus statements will provide further guidance for clinicians on the benefits of maintenance therapy in both transplant-eligible and -ineligible patients with newly diagnosed MM.
The recently updated MM guidelines from the European Society for Medical Oncology (ESMO) recommend longer treatment or maintenance therapy in patients who have undergone hematopoietic stem cell transplant (HSCT).7
Based on evidence from studies such as FIRST and SWOG S0777, ESMO also recommends continuous treatment or treatment until progression with lenalidomide-dexamethasone and bortezomib-lenalidomide-dexamethasone in MM patients who are ineligible for HSCT.7-9
As there is no one-size-fits-all treatment approach in MM, a personalized treatment plan should be designed for each patient. This plan should take into account a number of factors, including age, disease characteristics, performance status, treatment history, and the patient’s goals of care and personal preferences.10
If the patient is a candidate for longer treatment, the clinician should carefully weigh the potential impact on disease-free and overall survival against the potential side effects, as well as assess the patient’s likelihood of adhering to the medication.
With the availability of newer, less-toxic medications that can be tolerated for a greater duration and are easy to administer, aiding in overall treatment compliance, sustained remissions are possible.11-13
Forty years ago, MM patients had very few treatment options, and the 5-year survival rate was 26%.14
Since then, novel therapies, including proteasome inhibitors and immunomodulatory drugs, have replaced conventional cytotoxic chemotherapy, leading to major improvements in survival.15,16
With emerging research that supports the value of longer treatment strategies for both patients and the healthcare system, clinicians will have a proven strategy to help their patients attain long-term disease control while maintaining quality of life.2, 17-19
Dr. Weisel has received honoraria and/or consultancy fees from Amgen, BMS, Celgene, Janssen, Juno, Sanofi, and Takeda. She has received research funding from Amgen, Celgene, Sanofi, and Janssen.
The W2O Group provided writing support for this editorial, which was funded by Millennium Pharmaceuticals Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited.
1. Lipe B et al. Blood Cancer J. 2016; 6(10): e485. doi: 10.1038/bcj.2016.89
2. Goodwin J et al. Cancer Nurs. 2013; 36(4):301-8. doi: 10.1097/NCC.0b013e3182693522
3. Eek D et al. Patient Prefer Adherence. 2016; 10:1609-21. doi: 10.2147/PPA.S106629
4. Bauer S et al. Value in Health. 2017; 20: A451. Abstract PCN217. doi: https://doi.org/10.1016/j.jval.2017.08.299
5. A Study of Oral Ixazomib Citrate (MLN9708) Maintenance Therapy in Participants With Multiple Myeloma Following Autologous Stem Cell Transplant. (2014). Retrieved from https://clinicaltrials.gov/ct2/show/NCT02181413 (Identification No. NCT02181413).
6. A Study of Oral Ixazomib Maintenance Therapy in Patients With Newly Diagnosed Multiple Myeloma Not Treated With Stem Cell Transplantation. (2014). Retrieved from https://clinicaltrials.gov/ct2/show/NCT02312258 (Identification No. NCT02312258).
7. Moreau P et al. Ann Oncol. 2017; 28: iv52-iv61. doi: https://org/10.1093/annonc/mdx096
8. Facon T et al. Blood. 2018131(3):301-310. doi: 10.1182/blood-2017-07-795047
9. Durie BG et al. Lancet. 2017; 389(10068):519-527. doi: 10.1016/S0140-6736(16)31594-X.
10. Laubach J et al. Leukemia. 2016; 30(5):1005-17. doi: 10.1038/leu.2015.356
11. Ludwig H et al. Blood. 2012; 119: 3003-3015. doi: https://doi.org/10.1182/blood-2011-11-374249
12. Lehners N et al. Cancer Med. 2018; 7(2): 307–316. doi: 10.1002/cam4.1283
13. Attal M et al. N Engl J Med. 2012; 366:1872-1791. doi: 10.1056/NEJMoa1114138
14. National Cancer Institute. SEER Cancer Statistics Review (CSR) 1975-2014. National Cancer Institute. https://seer.cancer.gov/csr/1975_2014/. Accessed March 28, 2018.
15. Kumar SK et al. Blood. 2008 Mar 1;111(5):2516-20. doi: 10.1182/blood-2007-10-116129
16. Fonseca R et al. Leukemia. 2017 Sep;31(9):1915-1921. doi: 10.1038/leu.2016.380
17. Palumbo A, Niesvizky R. Leuk Res. 2012; 36 Suppl 1:S19-26. doi: 10.1016/S0145-2126(12)70005-X
18. Girnius S, Munshi NC. Leuk Suppl. 2013; 2(Suppl 1): S3–S9. doi: 10.1038/leusup.2013.2
19. Mateos M-V, San Miguel JF. Hematology Am Soc Hematol Educ Program. 2013; 2013:488-95. doi: 10.1182/asheducation-2013.1.488
In Part 2 of this editorial, Katja Weisel, MD, of University Hospital Tubingen in Germany, addresses the barriers to longer treatment in patients with multiple myeloma.
Attitudes regarding longer treatment can present barriers to widespread adoption of this approach in multiple myeloma (MM).
Indeed, some clinicians continue to follow a fixed-duration approach to treatment in MM, only considering further treatment once the patient has relapsed rather than treating the patient until disease progression.
In the MM community, some are reluctant to adopt a strategy of treating longer because of the modest efficacy gains observed with early research or concern over tolerability issues, including the risk of developing peripheral neuropathy or secondary malignancies.1
Others are uncertain about the optimal duration of therapy or the selection of an agent that will balance any potential gain in depth of response with the risk of late-onset or cumulative toxicities.
The potentially high cost of longer treatment for patients, their families, and/or the healthcare system overall also presents a challenge.
It is feasible that treating patients for longer may drive up healthcare utilization and take a toll on patients and caregivers, who may incur out-of-pocket costs because of the need to travel to a hospital or doctor’s office for intravenous therapies, requiring them to miss work.2
It is important to recognize, however, that more convenient all-oral treatment regimens are now available that do not require infusion at a hospital or clinic. Furthermore, results from recent studies suggest the majority of cancer patients prefer oral over intravenous therapies, which could reduce non-pharmacy healthcare costs.3,4
Healthcare providers might be more likely to accept and adopt a longer treatment approach for MM if they had access to data describing the optimal duration, dosage, schedule, toxicity, and quality of life standards.
Ongoing, randomized, phase 3 trials are evaluating the benefits of treating longer with an oral proteasome inhibitor in patients with newly diagnosed MM.5,6
Updated treatment guidelines and consensus statements will provide further guidance for clinicians on the benefits of maintenance therapy in both transplant-eligible and -ineligible patients with newly diagnosed MM.
The recently updated MM guidelines from the European Society for Medical Oncology (ESMO) recommend longer treatment or maintenance therapy in patients who have undergone hematopoietic stem cell transplant (HSCT).7
Based on evidence from studies such as FIRST and SWOG S0777, ESMO also recommends continuous treatment or treatment until progression with lenalidomide-dexamethasone and bortezomib-lenalidomide-dexamethasone in MM patients who are ineligible for HSCT.7-9
As there is no one-size-fits-all treatment approach in MM, a personalized treatment plan should be designed for each patient. This plan should take into account a number of factors, including age, disease characteristics, performance status, treatment history, and the patient’s goals of care and personal preferences.10
If the patient is a candidate for longer treatment, the clinician should carefully weigh the potential impact on disease-free and overall survival against the potential side effects, as well as assess the patient’s likelihood of adhering to the medication.
With the availability of newer, less-toxic medications that can be tolerated for a greater duration and are easy to administer, aiding in overall treatment compliance, sustained remissions are possible.11-13
Forty years ago, MM patients had very few treatment options, and the 5-year survival rate was 26%.14
Since then, novel therapies, including proteasome inhibitors and immunomodulatory drugs, have replaced conventional cytotoxic chemotherapy, leading to major improvements in survival.15,16
With emerging research that supports the value of longer treatment strategies for both patients and the healthcare system, clinicians will have a proven strategy to help their patients attain long-term disease control while maintaining quality of life.2, 17-19
Dr. Weisel has received honoraria and/or consultancy fees from Amgen, BMS, Celgene, Janssen, Juno, Sanofi, and Takeda. She has received research funding from Amgen, Celgene, Sanofi, and Janssen.
The W2O Group provided writing support for this editorial, which was funded by Millennium Pharmaceuticals Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited.
1. Lipe B et al. Blood Cancer J. 2016; 6(10): e485. doi: 10.1038/bcj.2016.89
2. Goodwin J et al. Cancer Nurs. 2013; 36(4):301-8. doi: 10.1097/NCC.0b013e3182693522
3. Eek D et al. Patient Prefer Adherence. 2016; 10:1609-21. doi: 10.2147/PPA.S106629
4. Bauer S et al. Value in Health. 2017; 20: A451. Abstract PCN217. doi: https://doi.org/10.1016/j.jval.2017.08.299
5. A Study of Oral Ixazomib Citrate (MLN9708) Maintenance Therapy in Participants With Multiple Myeloma Following Autologous Stem Cell Transplant. (2014). Retrieved from https://clinicaltrials.gov/ct2/show/NCT02181413 (Identification No. NCT02181413).
6. A Study of Oral Ixazomib Maintenance Therapy in Patients With Newly Diagnosed Multiple Myeloma Not Treated With Stem Cell Transplantation. (2014). Retrieved from https://clinicaltrials.gov/ct2/show/NCT02312258 (Identification No. NCT02312258).
7. Moreau P et al. Ann Oncol. 2017; 28: iv52-iv61. doi: https://org/10.1093/annonc/mdx096
8. Facon T et al. Blood. 2018131(3):301-310. doi: 10.1182/blood-2017-07-795047
9. Durie BG et al. Lancet. 2017; 389(10068):519-527. doi: 10.1016/S0140-6736(16)31594-X.
10. Laubach J et al. Leukemia. 2016; 30(5):1005-17. doi: 10.1038/leu.2015.356
11. Ludwig H et al. Blood. 2012; 119: 3003-3015. doi: https://doi.org/10.1182/blood-2011-11-374249
12. Lehners N et al. Cancer Med. 2018; 7(2): 307–316. doi: 10.1002/cam4.1283
13. Attal M et al. N Engl J Med. 2012; 366:1872-1791. doi: 10.1056/NEJMoa1114138
14. National Cancer Institute. SEER Cancer Statistics Review (CSR) 1975-2014. National Cancer Institute. https://seer.cancer.gov/csr/1975_2014/. Accessed March 28, 2018.
15. Kumar SK et al. Blood. 2008 Mar 1;111(5):2516-20. doi: 10.1182/blood-2007-10-116129
16. Fonseca R et al. Leukemia. 2017 Sep;31(9):1915-1921. doi: 10.1038/leu.2016.380
17. Palumbo A, Niesvizky R. Leuk Res. 2012; 36 Suppl 1:S19-26. doi: 10.1016/S0145-2126(12)70005-X
18. Girnius S, Munshi NC. Leuk Suppl. 2013; 2(Suppl 1): S3–S9. doi: 10.1038/leusup.2013.2
19. Mateos M-V, San Miguel JF. Hematology Am Soc Hematol Educ Program. 2013; 2013:488-95. doi: 10.1182/asheducation-2013.1.488
Premenstrual Dysphoric Disorder: Diagnosis and Management in Primary Care
CE/CME No: CR-1812
PROGRAM OVERVIEW
Earn credit by reading this article and successfully completing the posttest and evaluation. Successful completion is defined as a cumulative score of at least 70% correct.
EDUCATIONAL OBJECTIVES
• Understand the epidemiology and underlying pathogenesis of premenstrual dysphoric disorder (PMDD).
• Describe PMDD diagnostic criteria established by DSM-5.
• Differentiate PMDD from other conditions in order to provide appropriate treatment.
• Identify effective evidence-based treatment modalities for PMDD.
• Discuss PMDD treatment challenges and importance of individualizing PMDD treatment.
FACULTY
Jovanka Rajic is a recent graduate of the Master of Science in Nursing–Family Nurse Practitioner program at the Patricia A. Chin School of Nursing at California State University, Los Angeles. Stefanie A. Varela is adjunct faculty in the Patricia A. Chin School of Nursing at California State University, Los Angeles, and practices in the Obstetrics and Gynecology Department at Kaiser Permanente in Ontario, California.
The authors reported no conflicts of interest related to this article.
ACCREDITATION STATEMENT
This program has been reviewed and is approved for a maximum of 1.0 hour of American Academy of Physician Assistants (AAPA) Category 1 CME credit by the Physician Assistant Review Panel. [NPs: Both ANCC and the AANP Certification Program recognize AAPA as an approved provider of Category 1 credit.] Approval is valid through November 30, 2019.
Article begins on next page >>
The severe psychiatric and somatic symptoms of premenstrual dysphoric disorder (PMDD) can be debilitating and place women at increased risk for other psychiatric disorders (including major depression and generalized anxiety) and for suicidal ideation. While PMDD’s complex nature makes it an underdiagnosed condition, there are clear diagnostic criteria for clinicians to ensure their patients receive timely and appropriate treatment—thus reducing the risk for serious sequelae.
Premenstrual dysphoric disorder (PMDD) is categorized as a depressive disorder in the Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-5).1 The hallmarks of this unique disorder are chronic, severe psychiatric and somatic symptoms that occur only during the late luteal phase of the menstrual cycle and dissipate soon after the onset of menstruation.2 Symptoms are generally disruptive and often associated with significant distress and impaired quality of life.2
PMDD occurs in 3%-8% of women of childbearing age; it affects women worldwide and is not influenced by geography or culture.2 Genetic susceptibility, stress, obesity, and a history of trauma or sexual abuse have been implicated as risk factors.2-6 The impact of PMDD on health-related quality of life is greater than that of chronic back pain but comparable to that of rheumatoid arthritis and osteoarthritis.2,7 Significantly, women with PMDD have a 50%-78% lifetime risk for psychiatric disorders, such as major depressive, dysthymic, seasonal affective, and generalized anxiety disorders, and suicidality.2
PMDD can be challenging for primary care providers to diagnose and treat, due to the lack of standardized screening methods, unfamiliarity with evidence-based practices for diagnosis, and the need to tailor treatment to each patient’s individual needs.3,8 But the increased risk for psychiatric sequelae, including suicidality, make timely diagnosis and treatment of PMDD critical.2,9
PATHOGENESIS
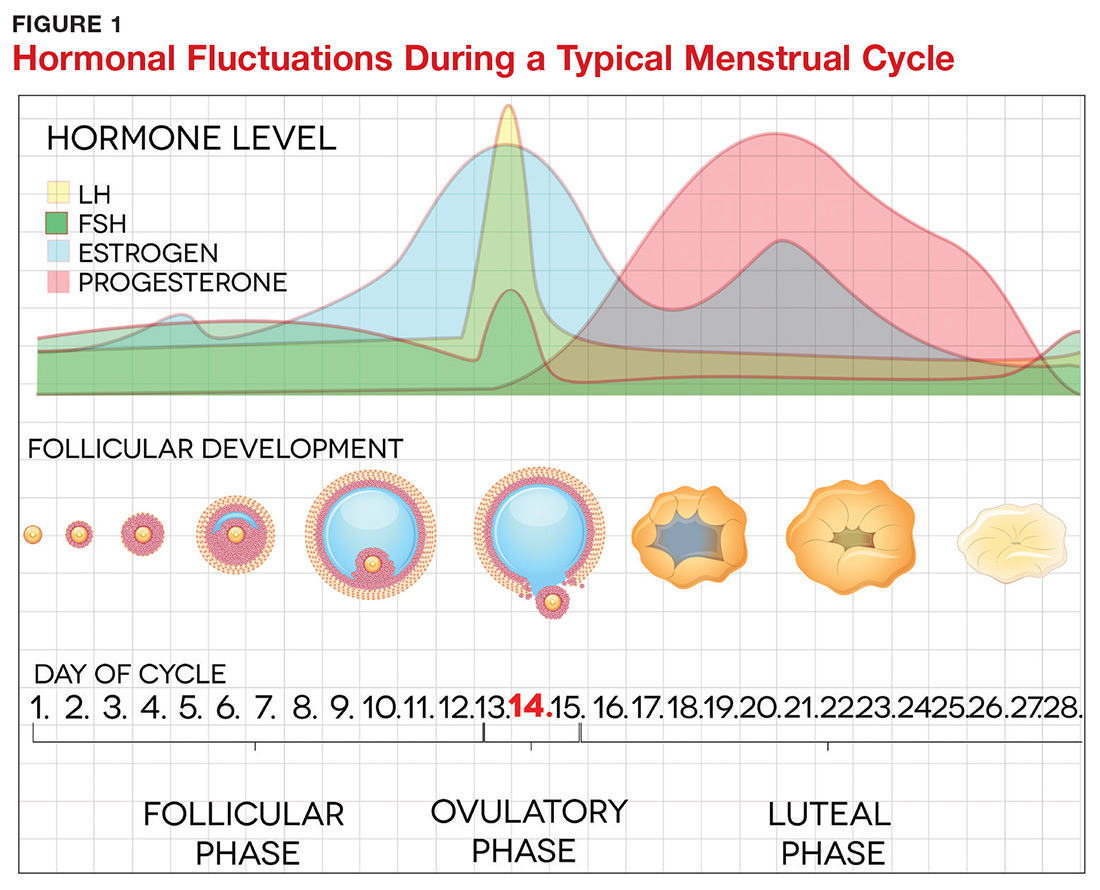
The pathogenesis of PMDD is not completely understood. The prevailing theory is that PMDD is underlined by increased sensitivity to normal fluctuations in ovarian steroid hormone levels (see the Figure) during the luteal phase of the menstrual cycle.2-4,6
This sensitivity involves the progesterone metabolite allopregnanolone (ALLO), which acts as a modulator of central GABA-A receptors that have anxiolytic and sedative effects.2,3 It has been postulated that women with PMDD have impaired production of ALLO or decreased sensitivity of GABA-A receptors to ALLO during the luteal phase.2,3 In addition, women with PMDD exhibit a paradoxical anxiety and irritability response to ALLO.2,3 Recent research suggests that PMDD is precipitated by changing ALLO levels during the luteal phase and that treatment directed at reducing ALLO availability during this phase can alleviate PMDD symptoms.10
Hormonal fluctuations have been associated with impaired serotonergic system function in women with PMDD, which results in dysregulation of mood, cognition, sleep, and eating behavior.2-4,6 Hormonal fluctuations have also been implicated in the alteration of emotional and cognitive circuits.2,3,6,11,12 Brain imaging studies have revealed that women with PMDD demonstrate enhanced reactivity to amygdala, which processes emotional and cognitive stimuli, as well as impaired control of amygdala by the prefrontal cortex during the luteal phase.3,7,12
Continue to: PATIENT PRESENTATION/HISTORY
PATIENT PRESENTATION/HISTORY
PMDD is an individual experience for each woman.3,4 However, women with PMDD generally present with a history of various psychiatric and somatic symptoms that significantly interfere with their occupational or social functions (to be discussed in the Diagnosis section, page 42).1-4 The reported symptoms occur in predictable patterns that are associated with the menstrual cycle, intensifying around the time of menstruation and resolving immediately after onset of menstruation in most cases.1-4
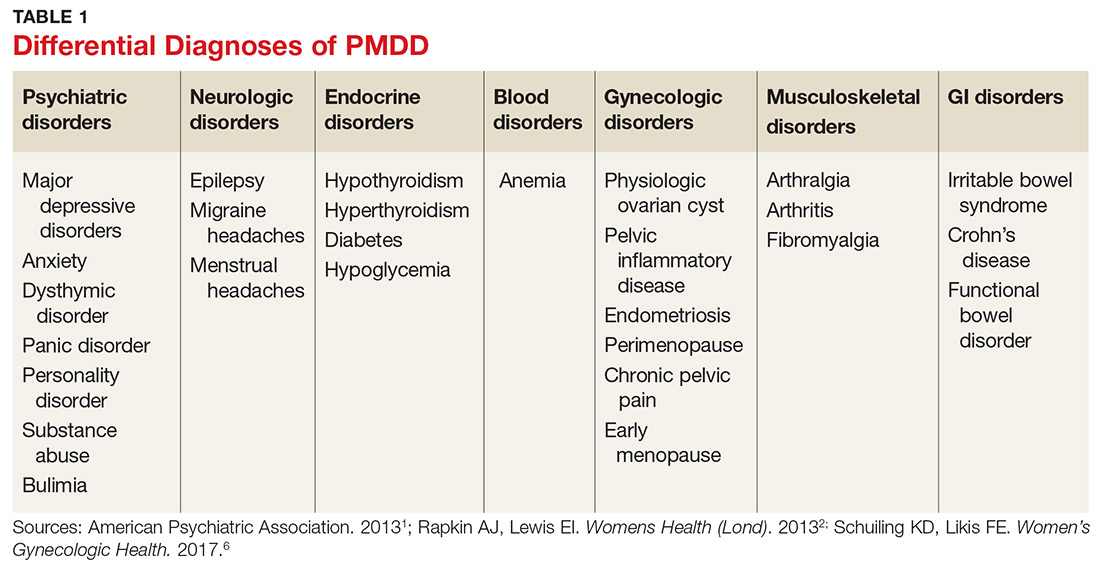
Many psychiatric and medical conditions may be exacerbated during the luteal phase of the menstrual cycle and thus may mimic the signs and symptoms of PMDD (see Table 1).1,4 Therefore, the pattern and severity of symptoms should always be considered when differentiating PMDD from other underlying conditions.1,2,4,5
It is also important to distinguish PMDD from PMS, a condition with which it is frequently confused. The latter manifests with at least one affective or somatic symptom that is bothersome but not disabling.4,5 An accurate differential diagnosis is important, as the management of these two conditions differs significantly.4,5
ASSESSMENT
PMDD assessment should include thorough history taking, with emphasis on medical, gynecologic, and psychiatric history as well as social and familial history (including PMDD and other psychiatric disorders); and physical examination, including gynecologic and mental status assessment and depression screening using the Patient Health Questionnaire (PHQ-9).2,4,13,14 The physical exam is usually unremarkable.14 The most common physical findings during the luteal phase include mild swelling in the lower extremities and breast tenderness.14 Mental status examination, however, may be abnormal during the late luteal phase—albeit with orientation, memory, thoughts, and perceptions intact.13,14
LABORATORY WORKUP
There is no specific laboratory test for PMDD; rather, testing is aimed at ruling out alternative diagnoses.4,14 Relevant studies may include a complete blood count to exclude anemia, a thyroid function test to exclude thyroid disorders, a blood glucose test to exclude diabetes or hypoglycemia, and a ß hCG test to exclude possible pregnancy.4,14 Hormonal tests (eg, for FSH) may be considered for younger women with irregular cycles or for those younger than 40 with suspected premature menopause.4,14
Continue to: DIAGNOSIS
DIAGNOSIS
Diagnosis of PMDD is guided by the DSM-5 criteria, which include the following components
- Content (presence of specific symptoms)
- Cyclicity (premenstrual onset and postmenstrual resolution)
- Severity (significant distress)
- Chronicity (occurrence in the past year).15
DSM-5 has established seven criteria (labeled A-G) for a PMDD diagnosis.1 First and foremost, a woman must experience a minimum of five of the 11 listed symptoms, with a minimum of one symptom being related to mood, during most menstrual cycles over the previous 12 months (Criterion A).1 The symptoms must occur during the week before the onset of menses, must improve within a few days of onset of menses, and must resolve in the week following menses.1
Mood-related symptoms (outlined in Criterion B) include
1. Notable depressed mood, hopelessness, or self-deprecation
2. Notable tension and/or anxiety
3. Notable affective lability (eg, mood swings, sudden sadness, tearfulness, or increased sensitivity to rejection)
4. Notable anger or irritability or increased interpersonal conflicts.1
Somatic or functional symptoms associated with PMDD (Criterion C) include:
5. Low interest in common activities (eg, those related to friends, work, school, and/or hobbies)
6. Difficulty concentrating
7. Lethargy, fatigue, or increased lack of energy
8. Notable change in appetite
9. Insomnia or hypersomnia
10. Feeling overwhelmed or out of control
11. Physical symptoms, such as breast tenderness or swelling, joint or muscle pain, headache, weight gain, or bloating.1
Again, patients must report at least one symptom from Criterion B and at least one from Criterion C—but a minimum of five symptoms overall—to receive a diagnosis of PMDD.1
Continue to: Additionally, the symptoms must...
Additionally, the symptoms must cause clinically significant distress or impair daily functioning, including occupational, social, academic, and sexual activities (Criterion D). They must not represent exacerbation of another underlying psychiatric disorder, such as major depressive, dysthymic, panic, or personality disorders (Criterion E), although PMDD may co-occur with psychiatric disorders.1
The above-mentioned symptom profile must be confirmed by prospective daily ratings of a minimum of two consecutive symptomatic menstrual cycles (Criterion F), although a provisional diagnosis of PMDD may be made prior to confirmation.1 The Daily Record of Severity of Problems is the most widely used instrument for prospective daily rating of PMDD symptoms listed in the DSM-5 criteria.5,15
Finally, the symptoms must not be evoked by the use of a substance (eg, medications, alcohol, and illicit drugs) or another medical condition (Criterion G).1
TREATMENT/MANAGEMENT
The goal of PMDD treatment is to relieve psychiatric and physical symptoms and improve the patient's ability to function.3 Treatment is primarily directed at pharmacologic neuromodulation using selective serotonin reuptake inhibitors (SSRIs) or ovulation suppression using oral contraceptives and hormones.2
Pharmacotherapy
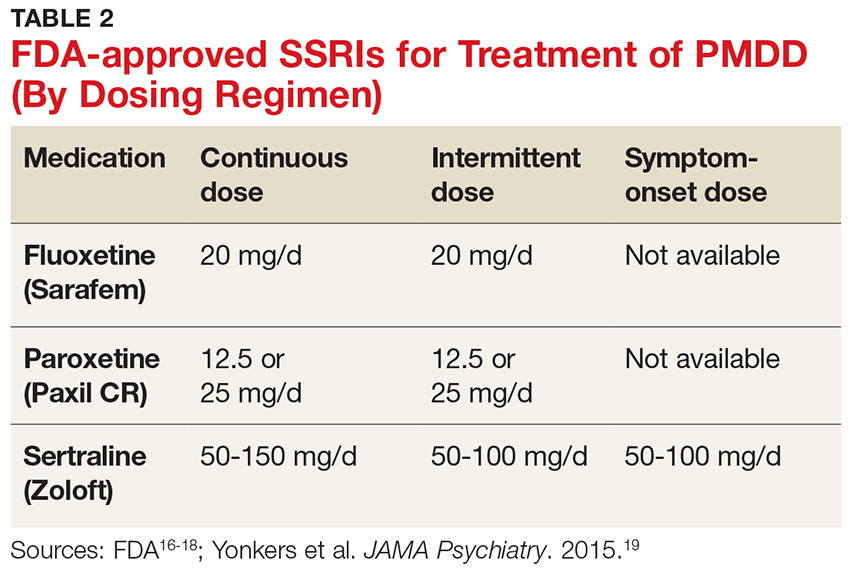
SSRIs are the firstline treatment for PMDD.5 Fluoxetine, paroxetine, and sertraline are the only serotonergic medications approved by the FDA for treatment of PMDD.2 SSRIs act within one to two days when used for PMDD, thereby allowing different modes of dosing.2 SSRI dosing may be continuous (daily administration), intermittent (administration from ovulation to first day of menses), or symptomatic (administration from symptom onset until first day of menses).3 Although data on continuous and intermittent dosing are available for fluoxetine, paroxetine, and sertraline, symptom-onset data are currently available only for sertraline (see Table 2).16-19
Continue to: Combined oral contraceptives...
Combined oral contraceptives (COCs) containing estrogen and progesterone are considered secondline treatment for PMDD—specifically, COCs containing 20 µg of ethinyl estradiol and 3 mg of drospirenone administered as a 24/4 regimen.2,3,5,6 This combination has been approved by the FDA for women with PMDD who seek oral contraception.3 Although drospirenone-containing products have been associated with increased risk for venous thromboembolism (VTE), this risk is lower than that for VTE during pregnancy or in the postpartum period.3 Currently, no strong evidence exists regarding the effectiveness of other oral contraceptives for PMDD.6
Gonadotropin-releasing hormone agonists are the thirdline treatment for PMDD.6 They eliminate symptoms of the luteal phase by suppressing ovarian release of estrogen and ovulation.6 However, use of these agents is not recommended for more than one year due to the increased risk for cardiovascular events.5,6 In addition, long-term users need add-back therapy (adding back small amounts of the hormone) to counteract the effects of low estrogen, such as bone loss; providers should be aware that this may lead to the recurrence of PMDD.3,5,6 The use of estrogen and progesterone formulations for PMDD is currently not strongly supported by research.6
Complementary treatment
Cognitive behavioral therapy has been shown to improve functioning and reduce depression in women with PMDD and may be a useful adjunct.2,20 Regular aerobic exercise, a diet high in protein and complex carbohydrates to increase tryptophan (serotonin precursor) levels, and reduced intake of caffeine, sugar, and alcohol are some commonly recommended lifestyle changes.2
Calcium carbonate supplementation (500 mg/d) has demonstrated effectiveness in alleviating premenstrual mood and physical symptoms.21 There is currently no strong evidence regarding the benefits of acupuncture, Qi therapy, reflexology, and herbal preparations for managing PMDD.22
Surgery
Bilateral oophorectomy, usually with concomitant hysterectomy, is the last resort for women with severe PMDD who do not respond to or cannot tolerate the standard treatments.6 This surgical procedure results in premature menopause, which may lead to complications related to a hypoestrogenic state—including vasomotor symptoms (flushes/flashes), vaginal atrophy, osteopenia, osteoporosis, and cardiovascular disease.2 Therefore, it is important to implement estrogen replacement therapy after surgery until the age of natural menopause is reached.2 If hysterectomy is not performed, the administration of progesterone is necessary to prevent endometrial hyperplasia and therefore reduce the risk for endometrial cancer.2 However, the addition of progesterone may lead to recurrence of symptoms.2
Continue to: Treatment challenges
Treatment challenges
PMDD treatment differs for each patient.3 Severity of symptoms, response to treatment, treatment preference, conception plans, and reproductive age need to be considered.3
Women with prominent depressive or physical symptoms may respond better to continuous dosing of SSRIs, whereas those with prominent irritability, anger, and mood swings may respond better to a symptom-onset SSRI regimen that reduces availability and function of ALLO.3 Women who develop tolerance to SSRIs may need to have their dosage increased or be switched to another medication.3Quetiapine is used as an adjunct to SSRIs for women who do not respond to SSRIs alone and has shown to improve mood swings, anxiety, and irritability.5 However, women experiencing persistent adverse effects of SSRIs, such as sexual dysfunction, may benefit from intermittent dosing.3
Adolescents and women in their early 20s should be treated with OCs or nonpharmacologic modalities due to concerns about SSRI use and increased risk for suicidality in this population.3 The risks related to SSRI use during pregnancy and breastfeeding should be considered and discussed with women of childbearing age who use SSRIs to treat PMDD.3 Perimenopausal women with irregular menses on intermittent SSRIs may have to switch to symptom-onset or continuous dosing due to the difficulty of tracking the menstrual period and lack of significant benchmarks regarding when to start the treatment.3
Patient education/follow-up
Patients should be educated on PMDD etiology, diagnostic process, and available treatment options.4 The importance of prospective record-keeping—for confirmation of the diagnosis and evaluation of individual response to a specific treatment—should be emphasized.4 Patients should be encouraged to follow up with their health care provider to monitor treatment effectiveness, possible adverse effects, and need for treatment adjustment.4
CONCLUSION
The symptoms of PMDD can have a debilitating and life-disrupting impact on affected women—and put them at risk for other serious psychiatric disorders and suicide. The DSM-5 criteria provide diagnostic guidance to help distinguish PMDD from other underlying conditions, ensuring that patients can receive timely and appropriate treatment. While SSRIs are regarded as the most effective option, other evidence-based treatments should be considered, since PMDD requires individualized treatment to ensure optimal clinical outcomes.
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Washington, DC: American Psychiatric Association; 2013.
2. Rapkin AJ, Lewis EI. Treatment of premenstrual dysphoric disorder. Womens Health (Lond). 2013;9(6):537-556.
3. Pearlstein T. Treatment of premenstrual dysphoric disorder: therapeutic challenges. Expert Rev Clin Pharmacol. 2016;9(4):493-496.
4. Zielinski R, Lynne S. Menstrual-cycle pain and premenstrual conditions. In: Schuiling KD, Likis FE, eds. Women’s Gynecologic Health. Burlington, MA: Jones & Bartlett Learning; 2017:556-573.
5. Hofmeister S, Bodden S. Premenstrual syndrome and premenstrual dysphoric disorder. Am Fam Physician. 2016;94(3):236-240.
6. Yonkers KA, Simoni MK. Premenstrual disorders. Am J Obstet Gynecol. 2018;218(1):68-74.
7. Yang M, Wallenstein G, Hagan M, et al. Burden of premenstrual dysphoric disorder on health-related quality of life. J Womens Health (Larchmt). 2008;17(1):113-121.
8. Craner JR, Sigmon ST, Women Health.
9. Hong JP, Park S, Wang HR, et al. Prevalence, correlates, comorbidities, and suicidal tendencies of premenstrual dysphoric disorder in a nationwide sample of Korean women. Soc Psychiatry Psychiatr Epidemiol. 2012;47(12): 1937-1945.
10. Martinez PE, Rubinow PR, Nieman LK, et al. 5α-reductase inhibition prevents the luteal phase increase in plasma allopregnanolone levels and mitigates symptoms in women with premenstrual dysphoric disorder. Neuropsychopharmacology. 2016;41:1093-1102.
11. Baller EB, Wei SM, Kohn PD. Abnormalities of dorsolateral prefrontal function in women with premenstrual dysphoric disorder: A multimodal neuroimaging study. Am J Psychiatry. 2013;170(3):305-314.
. EINeuroimaging the menstrual cycle and premenstrual dysphoric disorderCurr Psychiatry Rep.201577
13. Reid RL. Premenstrual dysphoric disorder (formerly premenstrual syndrome) [Updated Jan 23, 2017]. In: De Groot LJ, Chrousos G, Dungan K, et al, eds. Endotext [Internet]. South Dartmouth, MA: MDText.com, Inc; 2000.
14. Htay TT. Premenstrual dysphoric disorder clinical presentation. Medscape. https://emedicine.medscape.com/article/293257-clinical#b3. Updated February 16, 2016. Accessed February 7, 2018.
15. Epperson CN, Hantsoo LV. Making strides to simplify diagnosis of premenstrual dysphoric disorder. Am J Psychiatry. 2017;174(1):6-7.
16. FDA. Sarafem. www.accessdata.fda.gov/drugsatfda_docs/label/2006/021860lbl.pdf. Accessed February 15, 2018.
17. FDA. Paxil CR. www.accessdata.fda.gov/drugsatfda_docs/label/2004/20936se2-013_paxil_lbl.pdf. Accessed February 15, 2018.
18. FDA. Zoloft. www.accessdata.fda.gov/drugsatfda_docs/label/2016/019839s74s86s87_20990s35s44s45lbl.pdf. Accessed February 15, 2018.
19. Yonkers KA, Kornstein SG, Gueorguieva R, et al. Symptom-onset dosing of sertraline for the treatment of premenstrual dysphoric disorder: a randomized trial. JAMA Psychiatry. 2015;72(10):1037-1044.
20. Busse JW, Montori VM, Krasnik C, et al. Psychological intervention for premenstrual syndrome: a meta-analysis of randomized controlled trials. Psychother Psychosom. 2009;78(1):6-15.
21. Shobeiri F, Araste FE, Ebrahimi R, et al. Effect of calcium on premenstrual syndrome: a double-blind randomized clinical trial. Obstet Gynecol Sci. 2017;60(1):100-105.
22. Nevatte T, O’Brien PMS, Bäckström T, et al. ISPMD consensus on the management of premenstrual disorders. Arch Womens Ment Health. 2013;16(4):279-291.
CE/CME No: CR-1812
PROGRAM OVERVIEW
Earn credit by reading this article and successfully completing the posttest and evaluation. Successful completion is defined as a cumulative score of at least 70% correct.
EDUCATIONAL OBJECTIVES
• Understand the epidemiology and underlying pathogenesis of premenstrual dysphoric disorder (PMDD).
• Describe PMDD diagnostic criteria established by DSM-5.
• Differentiate PMDD from other conditions in order to provide appropriate treatment.
• Identify effective evidence-based treatment modalities for PMDD.
• Discuss PMDD treatment challenges and importance of individualizing PMDD treatment.
FACULTY
Jovanka Rajic is a recent graduate of the Master of Science in Nursing–Family Nurse Practitioner program at the Patricia A. Chin School of Nursing at California State University, Los Angeles. Stefanie A. Varela is adjunct faculty in the Patricia A. Chin School of Nursing at California State University, Los Angeles, and practices in the Obstetrics and Gynecology Department at Kaiser Permanente in Ontario, California.
The authors reported no conflicts of interest related to this article.
ACCREDITATION STATEMENT
This program has been reviewed and is approved for a maximum of 1.0 hour of American Academy of Physician Assistants (AAPA) Category 1 CME credit by the Physician Assistant Review Panel. [NPs: Both ANCC and the AANP Certification Program recognize AAPA as an approved provider of Category 1 credit.] Approval is valid through November 30, 2019.
Article begins on next page >>
The severe psychiatric and somatic symptoms of premenstrual dysphoric disorder (PMDD) can be debilitating and place women at increased risk for other psychiatric disorders (including major depression and generalized anxiety) and for suicidal ideation. While PMDD’s complex nature makes it an underdiagnosed condition, there are clear diagnostic criteria for clinicians to ensure their patients receive timely and appropriate treatment—thus reducing the risk for serious sequelae.
Premenstrual dysphoric disorder (PMDD) is categorized as a depressive disorder in the Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-5).1 The hallmarks of this unique disorder are chronic, severe psychiatric and somatic symptoms that occur only during the late luteal phase of the menstrual cycle and dissipate soon after the onset of menstruation.2 Symptoms are generally disruptive and often associated with significant distress and impaired quality of life.2
PMDD occurs in 3%-8% of women of childbearing age; it affects women worldwide and is not influenced by geography or culture.2 Genetic susceptibility, stress, obesity, and a history of trauma or sexual abuse have been implicated as risk factors.2-6 The impact of PMDD on health-related quality of life is greater than that of chronic back pain but comparable to that of rheumatoid arthritis and osteoarthritis.2,7 Significantly, women with PMDD have a 50%-78% lifetime risk for psychiatric disorders, such as major depressive, dysthymic, seasonal affective, and generalized anxiety disorders, and suicidality.2
PMDD can be challenging for primary care providers to diagnose and treat, due to the lack of standardized screening methods, unfamiliarity with evidence-based practices for diagnosis, and the need to tailor treatment to each patient’s individual needs.3,8 But the increased risk for psychiatric sequelae, including suicidality, make timely diagnosis and treatment of PMDD critical.2,9
PATHOGENESIS
The pathogenesis of PMDD is not completely understood. The prevailing theory is that PMDD is underlined by increased sensitivity to normal fluctuations in ovarian steroid hormone levels (see the Figure) during the luteal phase of the menstrual cycle.2-4,6
This sensitivity involves the progesterone metabolite allopregnanolone (ALLO), which acts as a modulator of central GABA-A receptors that have anxiolytic and sedative effects.2,3 It has been postulated that women with PMDD have impaired production of ALLO or decreased sensitivity of GABA-A receptors to ALLO during the luteal phase.2,3 In addition, women with PMDD exhibit a paradoxical anxiety and irritability response to ALLO.2,3 Recent research suggests that PMDD is precipitated by changing ALLO levels during the luteal phase and that treatment directed at reducing ALLO availability during this phase can alleviate PMDD symptoms.10
Hormonal fluctuations have been associated with impaired serotonergic system function in women with PMDD, which results in dysregulation of mood, cognition, sleep, and eating behavior.2-4,6 Hormonal fluctuations have also been implicated in the alteration of emotional and cognitive circuits.2,3,6,11,12 Brain imaging studies have revealed that women with PMDD demonstrate enhanced reactivity to amygdala, which processes emotional and cognitive stimuli, as well as impaired control of amygdala by the prefrontal cortex during the luteal phase.3,7,12
Continue to: PATIENT PRESENTATION/HISTORY
PATIENT PRESENTATION/HISTORY
PMDD is an individual experience for each woman.3,4 However, women with PMDD generally present with a history of various psychiatric and somatic symptoms that significantly interfere with their occupational or social functions (to be discussed in the Diagnosis section, page 42).1-4 The reported symptoms occur in predictable patterns that are associated with the menstrual cycle, intensifying around the time of menstruation and resolving immediately after onset of menstruation in most cases.1-4
Many psychiatric and medical conditions may be exacerbated during the luteal phase of the menstrual cycle and thus may mimic the signs and symptoms of PMDD (see Table 1).1,4 Therefore, the pattern and severity of symptoms should always be considered when differentiating PMDD from other underlying conditions.1,2,4,5
It is also important to distinguish PMDD from PMS, a condition with which it is frequently confused. The latter manifests with at least one affective or somatic symptom that is bothersome but not disabling.4,5 An accurate differential diagnosis is important, as the management of these two conditions differs significantly.4,5
ASSESSMENT
PMDD assessment should include thorough history taking, with emphasis on medical, gynecologic, and psychiatric history as well as social and familial history (including PMDD and other psychiatric disorders); and physical examination, including gynecologic and mental status assessment and depression screening using the Patient Health Questionnaire (PHQ-9).2,4,13,14 The physical exam is usually unremarkable.14 The most common physical findings during the luteal phase include mild swelling in the lower extremities and breast tenderness.14 Mental status examination, however, may be abnormal during the late luteal phase—albeit with orientation, memory, thoughts, and perceptions intact.13,14
LABORATORY WORKUP
There is no specific laboratory test for PMDD; rather, testing is aimed at ruling out alternative diagnoses.4,14 Relevant studies may include a complete blood count to exclude anemia, a thyroid function test to exclude thyroid disorders, a blood glucose test to exclude diabetes or hypoglycemia, and a ß hCG test to exclude possible pregnancy.4,14 Hormonal tests (eg, for FSH) may be considered for younger women with irregular cycles or for those younger than 40 with suspected premature menopause.4,14
Continue to: DIAGNOSIS
DIAGNOSIS
Diagnosis of PMDD is guided by the DSM-5 criteria, which include the following components
- Content (presence of specific symptoms)
- Cyclicity (premenstrual onset and postmenstrual resolution)
- Severity (significant distress)
- Chronicity (occurrence in the past year).15
DSM-5 has established seven criteria (labeled A-G) for a PMDD diagnosis.1 First and foremost, a woman must experience a minimum of five of the 11 listed symptoms, with a minimum of one symptom being related to mood, during most menstrual cycles over the previous 12 months (Criterion A).1 The symptoms must occur during the week before the onset of menses, must improve within a few days of onset of menses, and must resolve in the week following menses.1
Mood-related symptoms (outlined in Criterion B) include
1. Notable depressed mood, hopelessness, or self-deprecation
2. Notable tension and/or anxiety
3. Notable affective lability (eg, mood swings, sudden sadness, tearfulness, or increased sensitivity to rejection)
4. Notable anger or irritability or increased interpersonal conflicts.1
Somatic or functional symptoms associated with PMDD (Criterion C) include:
5. Low interest in common activities (eg, those related to friends, work, school, and/or hobbies)
6. Difficulty concentrating
7. Lethargy, fatigue, or increased lack of energy
8. Notable change in appetite
9. Insomnia or hypersomnia
10. Feeling overwhelmed or out of control
11. Physical symptoms, such as breast tenderness or swelling, joint or muscle pain, headache, weight gain, or bloating.1
Again, patients must report at least one symptom from Criterion B and at least one from Criterion C—but a minimum of five symptoms overall—to receive a diagnosis of PMDD.1
Continue to: Additionally, the symptoms must...
Additionally, the symptoms must cause clinically significant distress or impair daily functioning, including occupational, social, academic, and sexual activities (Criterion D). They must not represent exacerbation of another underlying psychiatric disorder, such as major depressive, dysthymic, panic, or personality disorders (Criterion E), although PMDD may co-occur with psychiatric disorders.1
The above-mentioned symptom profile must be confirmed by prospective daily ratings of a minimum of two consecutive symptomatic menstrual cycles (Criterion F), although a provisional diagnosis of PMDD may be made prior to confirmation.1 The Daily Record of Severity of Problems is the most widely used instrument for prospective daily rating of PMDD symptoms listed in the DSM-5 criteria.5,15
Finally, the symptoms must not be evoked by the use of a substance (eg, medications, alcohol, and illicit drugs) or another medical condition (Criterion G).1
TREATMENT/MANAGEMENT
The goal of PMDD treatment is to relieve psychiatric and physical symptoms and improve the patient's ability to function.3 Treatment is primarily directed at pharmacologic neuromodulation using selective serotonin reuptake inhibitors (SSRIs) or ovulation suppression using oral contraceptives and hormones.2
Pharmacotherapy
SSRIs are the firstline treatment for PMDD.5 Fluoxetine, paroxetine, and sertraline are the only serotonergic medications approved by the FDA for treatment of PMDD.2 SSRIs act within one to two days when used for PMDD, thereby allowing different modes of dosing.2 SSRI dosing may be continuous (daily administration), intermittent (administration from ovulation to first day of menses), or symptomatic (administration from symptom onset until first day of menses).3 Although data on continuous and intermittent dosing are available for fluoxetine, paroxetine, and sertraline, symptom-onset data are currently available only for sertraline (see Table 2).16-19
Continue to: Combined oral contraceptives...
Combined oral contraceptives (COCs) containing estrogen and progesterone are considered secondline treatment for PMDD—specifically, COCs containing 20 µg of ethinyl estradiol and 3 mg of drospirenone administered as a 24/4 regimen.2,3,5,6 This combination has been approved by the FDA for women with PMDD who seek oral contraception.3 Although drospirenone-containing products have been associated with increased risk for venous thromboembolism (VTE), this risk is lower than that for VTE during pregnancy or in the postpartum period.3 Currently, no strong evidence exists regarding the effectiveness of other oral contraceptives for PMDD.6
Gonadotropin-releasing hormone agonists are the thirdline treatment for PMDD.6 They eliminate symptoms of the luteal phase by suppressing ovarian release of estrogen and ovulation.6 However, use of these agents is not recommended for more than one year due to the increased risk for cardiovascular events.5,6 In addition, long-term users need add-back therapy (adding back small amounts of the hormone) to counteract the effects of low estrogen, such as bone loss; providers should be aware that this may lead to the recurrence of PMDD.3,5,6 The use of estrogen and progesterone formulations for PMDD is currently not strongly supported by research.6
Complementary treatment
Cognitive behavioral therapy has been shown to improve functioning and reduce depression in women with PMDD and may be a useful adjunct.2,20 Regular aerobic exercise, a diet high in protein and complex carbohydrates to increase tryptophan (serotonin precursor) levels, and reduced intake of caffeine, sugar, and alcohol are some commonly recommended lifestyle changes.2
Calcium carbonate supplementation (500 mg/d) has demonstrated effectiveness in alleviating premenstrual mood and physical symptoms.21 There is currently no strong evidence regarding the benefits of acupuncture, Qi therapy, reflexology, and herbal preparations for managing PMDD.22
Surgery
Bilateral oophorectomy, usually with concomitant hysterectomy, is the last resort for women with severe PMDD who do not respond to or cannot tolerate the standard treatments.6 This surgical procedure results in premature menopause, which may lead to complications related to a hypoestrogenic state—including vasomotor symptoms (flushes/flashes), vaginal atrophy, osteopenia, osteoporosis, and cardiovascular disease.2 Therefore, it is important to implement estrogen replacement therapy after surgery until the age of natural menopause is reached.2 If hysterectomy is not performed, the administration of progesterone is necessary to prevent endometrial hyperplasia and therefore reduce the risk for endometrial cancer.2 However, the addition of progesterone may lead to recurrence of symptoms.2
Continue to: Treatment challenges
Treatment challenges
PMDD treatment differs for each patient.3 Severity of symptoms, response to treatment, treatment preference, conception plans, and reproductive age need to be considered.3
Women with prominent depressive or physical symptoms may respond better to continuous dosing of SSRIs, whereas those with prominent irritability, anger, and mood swings may respond better to a symptom-onset SSRI regimen that reduces availability and function of ALLO.3 Women who develop tolerance to SSRIs may need to have their dosage increased or be switched to another medication.3Quetiapine is used as an adjunct to SSRIs for women who do not respond to SSRIs alone and has shown to improve mood swings, anxiety, and irritability.5 However, women experiencing persistent adverse effects of SSRIs, such as sexual dysfunction, may benefit from intermittent dosing.3
Adolescents and women in their early 20s should be treated with OCs or nonpharmacologic modalities due to concerns about SSRI use and increased risk for suicidality in this population.3 The risks related to SSRI use during pregnancy and breastfeeding should be considered and discussed with women of childbearing age who use SSRIs to treat PMDD.3 Perimenopausal women with irregular menses on intermittent SSRIs may have to switch to symptom-onset or continuous dosing due to the difficulty of tracking the menstrual period and lack of significant benchmarks regarding when to start the treatment.3
Patient education/follow-up
Patients should be educated on PMDD etiology, diagnostic process, and available treatment options.4 The importance of prospective record-keeping—for confirmation of the diagnosis and evaluation of individual response to a specific treatment—should be emphasized.4 Patients should be encouraged to follow up with their health care provider to monitor treatment effectiveness, possible adverse effects, and need for treatment adjustment.4
CONCLUSION
The symptoms of PMDD can have a debilitating and life-disrupting impact on affected women—and put them at risk for other serious psychiatric disorders and suicide. The DSM-5 criteria provide diagnostic guidance to help distinguish PMDD from other underlying conditions, ensuring that patients can receive timely and appropriate treatment. While SSRIs are regarded as the most effective option, other evidence-based treatments should be considered, since PMDD requires individualized treatment to ensure optimal clinical outcomes.
CE/CME No: CR-1812
PROGRAM OVERVIEW
Earn credit by reading this article and successfully completing the posttest and evaluation. Successful completion is defined as a cumulative score of at least 70% correct.
EDUCATIONAL OBJECTIVES
• Understand the epidemiology and underlying pathogenesis of premenstrual dysphoric disorder (PMDD).
• Describe PMDD diagnostic criteria established by DSM-5.
• Differentiate PMDD from other conditions in order to provide appropriate treatment.
• Identify effective evidence-based treatment modalities for PMDD.
• Discuss PMDD treatment challenges and importance of individualizing PMDD treatment.
FACULTY
Jovanka Rajic is a recent graduate of the Master of Science in Nursing–Family Nurse Practitioner program at the Patricia A. Chin School of Nursing at California State University, Los Angeles. Stefanie A. Varela is adjunct faculty in the Patricia A. Chin School of Nursing at California State University, Los Angeles, and practices in the Obstetrics and Gynecology Department at Kaiser Permanente in Ontario, California.
The authors reported no conflicts of interest related to this article.
ACCREDITATION STATEMENT
This program has been reviewed and is approved for a maximum of 1.0 hour of American Academy of Physician Assistants (AAPA) Category 1 CME credit by the Physician Assistant Review Panel. [NPs: Both ANCC and the AANP Certification Program recognize AAPA as an approved provider of Category 1 credit.] Approval is valid through November 30, 2019.
Article begins on next page >>
The severe psychiatric and somatic symptoms of premenstrual dysphoric disorder (PMDD) can be debilitating and place women at increased risk for other psychiatric disorders (including major depression and generalized anxiety) and for suicidal ideation. While PMDD’s complex nature makes it an underdiagnosed condition, there are clear diagnostic criteria for clinicians to ensure their patients receive timely and appropriate treatment—thus reducing the risk for serious sequelae.
Premenstrual dysphoric disorder (PMDD) is categorized as a depressive disorder in the Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-5).1 The hallmarks of this unique disorder are chronic, severe psychiatric and somatic symptoms that occur only during the late luteal phase of the menstrual cycle and dissipate soon after the onset of menstruation.2 Symptoms are generally disruptive and often associated with significant distress and impaired quality of life.2
PMDD occurs in 3%-8% of women of childbearing age; it affects women worldwide and is not influenced by geography or culture.2 Genetic susceptibility, stress, obesity, and a history of trauma or sexual abuse have been implicated as risk factors.2-6 The impact of PMDD on health-related quality of life is greater than that of chronic back pain but comparable to that of rheumatoid arthritis and osteoarthritis.2,7 Significantly, women with PMDD have a 50%-78% lifetime risk for psychiatric disorders, such as major depressive, dysthymic, seasonal affective, and generalized anxiety disorders, and suicidality.2
PMDD can be challenging for primary care providers to diagnose and treat, due to the lack of standardized screening methods, unfamiliarity with evidence-based practices for diagnosis, and the need to tailor treatment to each patient’s individual needs.3,8 But the increased risk for psychiatric sequelae, including suicidality, make timely diagnosis and treatment of PMDD critical.2,9
PATHOGENESIS
The pathogenesis of PMDD is not completely understood. The prevailing theory is that PMDD is underlined by increased sensitivity to normal fluctuations in ovarian steroid hormone levels (see the Figure) during the luteal phase of the menstrual cycle.2-4,6
This sensitivity involves the progesterone metabolite allopregnanolone (ALLO), which acts as a modulator of central GABA-A receptors that have anxiolytic and sedative effects.2,3 It has been postulated that women with PMDD have impaired production of ALLO or decreased sensitivity of GABA-A receptors to ALLO during the luteal phase.2,3 In addition, women with PMDD exhibit a paradoxical anxiety and irritability response to ALLO.2,3 Recent research suggests that PMDD is precipitated by changing ALLO levels during the luteal phase and that treatment directed at reducing ALLO availability during this phase can alleviate PMDD symptoms.10
Hormonal fluctuations have been associated with impaired serotonergic system function in women with PMDD, which results in dysregulation of mood, cognition, sleep, and eating behavior.2-4,6 Hormonal fluctuations have also been implicated in the alteration of emotional and cognitive circuits.2,3,6,11,12 Brain imaging studies have revealed that women with PMDD demonstrate enhanced reactivity to amygdala, which processes emotional and cognitive stimuli, as well as impaired control of amygdala by the prefrontal cortex during the luteal phase.3,7,12
Continue to: PATIENT PRESENTATION/HISTORY
PATIENT PRESENTATION/HISTORY
PMDD is an individual experience for each woman.3,4 However, women with PMDD generally present with a history of various psychiatric and somatic symptoms that significantly interfere with their occupational or social functions (to be discussed in the Diagnosis section, page 42).1-4 The reported symptoms occur in predictable patterns that are associated with the menstrual cycle, intensifying around the time of menstruation and resolving immediately after onset of menstruation in most cases.1-4
Many psychiatric and medical conditions may be exacerbated during the luteal phase of the menstrual cycle and thus may mimic the signs and symptoms of PMDD (see Table 1).1,4 Therefore, the pattern and severity of symptoms should always be considered when differentiating PMDD from other underlying conditions.1,2,4,5
It is also important to distinguish PMDD from PMS, a condition with which it is frequently confused. The latter manifests with at least one affective or somatic symptom that is bothersome but not disabling.4,5 An accurate differential diagnosis is important, as the management of these two conditions differs significantly.4,5
ASSESSMENT
PMDD assessment should include thorough history taking, with emphasis on medical, gynecologic, and psychiatric history as well as social and familial history (including PMDD and other psychiatric disorders); and physical examination, including gynecologic and mental status assessment and depression screening using the Patient Health Questionnaire (PHQ-9).2,4,13,14 The physical exam is usually unremarkable.14 The most common physical findings during the luteal phase include mild swelling in the lower extremities and breast tenderness.14 Mental status examination, however, may be abnormal during the late luteal phase—albeit with orientation, memory, thoughts, and perceptions intact.13,14
LABORATORY WORKUP
There is no specific laboratory test for PMDD; rather, testing is aimed at ruling out alternative diagnoses.4,14 Relevant studies may include a complete blood count to exclude anemia, a thyroid function test to exclude thyroid disorders, a blood glucose test to exclude diabetes or hypoglycemia, and a ß hCG test to exclude possible pregnancy.4,14 Hormonal tests (eg, for FSH) may be considered for younger women with irregular cycles or for those younger than 40 with suspected premature menopause.4,14
Continue to: DIAGNOSIS
DIAGNOSIS
Diagnosis of PMDD is guided by the DSM-5 criteria, which include the following components
- Content (presence of specific symptoms)
- Cyclicity (premenstrual onset and postmenstrual resolution)
- Severity (significant distress)
- Chronicity (occurrence in the past year).15
DSM-5 has established seven criteria (labeled A-G) for a PMDD diagnosis.1 First and foremost, a woman must experience a minimum of five of the 11 listed symptoms, with a minimum of one symptom being related to mood, during most menstrual cycles over the previous 12 months (Criterion A).1 The symptoms must occur during the week before the onset of menses, must improve within a few days of onset of menses, and must resolve in the week following menses.1
Mood-related symptoms (outlined in Criterion B) include
1. Notable depressed mood, hopelessness, or self-deprecation
2. Notable tension and/or anxiety
3. Notable affective lability (eg, mood swings, sudden sadness, tearfulness, or increased sensitivity to rejection)
4. Notable anger or irritability or increased interpersonal conflicts.1
Somatic or functional symptoms associated with PMDD (Criterion C) include:
5. Low interest in common activities (eg, those related to friends, work, school, and/or hobbies)
6. Difficulty concentrating
7. Lethargy, fatigue, or increased lack of energy
8. Notable change in appetite
9. Insomnia or hypersomnia
10. Feeling overwhelmed or out of control
11. Physical symptoms, such as breast tenderness or swelling, joint or muscle pain, headache, weight gain, or bloating.1
Again, patients must report at least one symptom from Criterion B and at least one from Criterion C—but a minimum of five symptoms overall—to receive a diagnosis of PMDD.1
Continue to: Additionally, the symptoms must...
Additionally, the symptoms must cause clinically significant distress or impair daily functioning, including occupational, social, academic, and sexual activities (Criterion D). They must not represent exacerbation of another underlying psychiatric disorder, such as major depressive, dysthymic, panic, or personality disorders (Criterion E), although PMDD may co-occur with psychiatric disorders.1
The above-mentioned symptom profile must be confirmed by prospective daily ratings of a minimum of two consecutive symptomatic menstrual cycles (Criterion F), although a provisional diagnosis of PMDD may be made prior to confirmation.1 The Daily Record of Severity of Problems is the most widely used instrument for prospective daily rating of PMDD symptoms listed in the DSM-5 criteria.5,15
Finally, the symptoms must not be evoked by the use of a substance (eg, medications, alcohol, and illicit drugs) or another medical condition (Criterion G).1
TREATMENT/MANAGEMENT
The goal of PMDD treatment is to relieve psychiatric and physical symptoms and improve the patient's ability to function.3 Treatment is primarily directed at pharmacologic neuromodulation using selective serotonin reuptake inhibitors (SSRIs) or ovulation suppression using oral contraceptives and hormones.2
Pharmacotherapy
SSRIs are the firstline treatment for PMDD.5 Fluoxetine, paroxetine, and sertraline are the only serotonergic medications approved by the FDA for treatment of PMDD.2 SSRIs act within one to two days when used for PMDD, thereby allowing different modes of dosing.2 SSRI dosing may be continuous (daily administration), intermittent (administration from ovulation to first day of menses), or symptomatic (administration from symptom onset until first day of menses).3 Although data on continuous and intermittent dosing are available for fluoxetine, paroxetine, and sertraline, symptom-onset data are currently available only for sertraline (see Table 2).16-19
Continue to: Combined oral contraceptives...
Combined oral contraceptives (COCs) containing estrogen and progesterone are considered secondline treatment for PMDD—specifically, COCs containing 20 µg of ethinyl estradiol and 3 mg of drospirenone administered as a 24/4 regimen.2,3,5,6 This combination has been approved by the FDA for women with PMDD who seek oral contraception.3 Although drospirenone-containing products have been associated with increased risk for venous thromboembolism (VTE), this risk is lower than that for VTE during pregnancy or in the postpartum period.3 Currently, no strong evidence exists regarding the effectiveness of other oral contraceptives for PMDD.6
Gonadotropin-releasing hormone agonists are the thirdline treatment for PMDD.6 They eliminate symptoms of the luteal phase by suppressing ovarian release of estrogen and ovulation.6 However, use of these agents is not recommended for more than one year due to the increased risk for cardiovascular events.5,6 In addition, long-term users need add-back therapy (adding back small amounts of the hormone) to counteract the effects of low estrogen, such as bone loss; providers should be aware that this may lead to the recurrence of PMDD.3,5,6 The use of estrogen and progesterone formulations for PMDD is currently not strongly supported by research.6
Complementary treatment
Cognitive behavioral therapy has been shown to improve functioning and reduce depression in women with PMDD and may be a useful adjunct.2,20 Regular aerobic exercise, a diet high in protein and complex carbohydrates to increase tryptophan (serotonin precursor) levels, and reduced intake of caffeine, sugar, and alcohol are some commonly recommended lifestyle changes.2
Calcium carbonate supplementation (500 mg/d) has demonstrated effectiveness in alleviating premenstrual mood and physical symptoms.21 There is currently no strong evidence regarding the benefits of acupuncture, Qi therapy, reflexology, and herbal preparations for managing PMDD.22
Surgery
Bilateral oophorectomy, usually with concomitant hysterectomy, is the last resort for women with severe PMDD who do not respond to or cannot tolerate the standard treatments.6 This surgical procedure results in premature menopause, which may lead to complications related to a hypoestrogenic state—including vasomotor symptoms (flushes/flashes), vaginal atrophy, osteopenia, osteoporosis, and cardiovascular disease.2 Therefore, it is important to implement estrogen replacement therapy after surgery until the age of natural menopause is reached.2 If hysterectomy is not performed, the administration of progesterone is necessary to prevent endometrial hyperplasia and therefore reduce the risk for endometrial cancer.2 However, the addition of progesterone may lead to recurrence of symptoms.2
Continue to: Treatment challenges
Treatment challenges
PMDD treatment differs for each patient.3 Severity of symptoms, response to treatment, treatment preference, conception plans, and reproductive age need to be considered.3
Women with prominent depressive or physical symptoms may respond better to continuous dosing of SSRIs, whereas those with prominent irritability, anger, and mood swings may respond better to a symptom-onset SSRI regimen that reduces availability and function of ALLO.3 Women who develop tolerance to SSRIs may need to have their dosage increased or be switched to another medication.3Quetiapine is used as an adjunct to SSRIs for women who do not respond to SSRIs alone and has shown to improve mood swings, anxiety, and irritability.5 However, women experiencing persistent adverse effects of SSRIs, such as sexual dysfunction, may benefit from intermittent dosing.3
Adolescents and women in their early 20s should be treated with OCs or nonpharmacologic modalities due to concerns about SSRI use and increased risk for suicidality in this population.3 The risks related to SSRI use during pregnancy and breastfeeding should be considered and discussed with women of childbearing age who use SSRIs to treat PMDD.3 Perimenopausal women with irregular menses on intermittent SSRIs may have to switch to symptom-onset or continuous dosing due to the difficulty of tracking the menstrual period and lack of significant benchmarks regarding when to start the treatment.3
Patient education/follow-up
Patients should be educated on PMDD etiology, diagnostic process, and available treatment options.4 The importance of prospective record-keeping—for confirmation of the diagnosis and evaluation of individual response to a specific treatment—should be emphasized.4 Patients should be encouraged to follow up with their health care provider to monitor treatment effectiveness, possible adverse effects, and need for treatment adjustment.4
CONCLUSION
The symptoms of PMDD can have a debilitating and life-disrupting impact on affected women—and put them at risk for other serious psychiatric disorders and suicide. The DSM-5 criteria provide diagnostic guidance to help distinguish PMDD from other underlying conditions, ensuring that patients can receive timely and appropriate treatment. While SSRIs are regarded as the most effective option, other evidence-based treatments should be considered, since PMDD requires individualized treatment to ensure optimal clinical outcomes.
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Washington, DC: American Psychiatric Association; 2013.
2. Rapkin AJ, Lewis EI. Treatment of premenstrual dysphoric disorder. Womens Health (Lond). 2013;9(6):537-556.
3. Pearlstein T. Treatment of premenstrual dysphoric disorder: therapeutic challenges. Expert Rev Clin Pharmacol. 2016;9(4):493-496.
4. Zielinski R, Lynne S. Menstrual-cycle pain and premenstrual conditions. In: Schuiling KD, Likis FE, eds. Women’s Gynecologic Health. Burlington, MA: Jones & Bartlett Learning; 2017:556-573.
5. Hofmeister S, Bodden S. Premenstrual syndrome and premenstrual dysphoric disorder. Am Fam Physician. 2016;94(3):236-240.
6. Yonkers KA, Simoni MK. Premenstrual disorders. Am J Obstet Gynecol. 2018;218(1):68-74.
7. Yang M, Wallenstein G, Hagan M, et al. Burden of premenstrual dysphoric disorder on health-related quality of life. J Womens Health (Larchmt). 2008;17(1):113-121.
8. Craner JR, Sigmon ST, Women Health.
9. Hong JP, Park S, Wang HR, et al. Prevalence, correlates, comorbidities, and suicidal tendencies of premenstrual dysphoric disorder in a nationwide sample of Korean women. Soc Psychiatry Psychiatr Epidemiol. 2012;47(12): 1937-1945.
10. Martinez PE, Rubinow PR, Nieman LK, et al. 5α-reductase inhibition prevents the luteal phase increase in plasma allopregnanolone levels and mitigates symptoms in women with premenstrual dysphoric disorder. Neuropsychopharmacology. 2016;41:1093-1102.
11. Baller EB, Wei SM, Kohn PD. Abnormalities of dorsolateral prefrontal function in women with premenstrual dysphoric disorder: A multimodal neuroimaging study. Am J Psychiatry. 2013;170(3):305-314.
. EINeuroimaging the menstrual cycle and premenstrual dysphoric disorderCurr Psychiatry Rep.201577
13. Reid RL. Premenstrual dysphoric disorder (formerly premenstrual syndrome) [Updated Jan 23, 2017]. In: De Groot LJ, Chrousos G, Dungan K, et al, eds. Endotext [Internet]. South Dartmouth, MA: MDText.com, Inc; 2000.
14. Htay TT. Premenstrual dysphoric disorder clinical presentation. Medscape. https://emedicine.medscape.com/article/293257-clinical#b3. Updated February 16, 2016. Accessed February 7, 2018.
15. Epperson CN, Hantsoo LV. Making strides to simplify diagnosis of premenstrual dysphoric disorder. Am J Psychiatry. 2017;174(1):6-7.
16. FDA. Sarafem. www.accessdata.fda.gov/drugsatfda_docs/label/2006/021860lbl.pdf. Accessed February 15, 2018.
17. FDA. Paxil CR. www.accessdata.fda.gov/drugsatfda_docs/label/2004/20936se2-013_paxil_lbl.pdf. Accessed February 15, 2018.
18. FDA. Zoloft. www.accessdata.fda.gov/drugsatfda_docs/label/2016/019839s74s86s87_20990s35s44s45lbl.pdf. Accessed February 15, 2018.
19. Yonkers KA, Kornstein SG, Gueorguieva R, et al. Symptom-onset dosing of sertraline for the treatment of premenstrual dysphoric disorder: a randomized trial. JAMA Psychiatry. 2015;72(10):1037-1044.
20. Busse JW, Montori VM, Krasnik C, et al. Psychological intervention for premenstrual syndrome: a meta-analysis of randomized controlled trials. Psychother Psychosom. 2009;78(1):6-15.
21. Shobeiri F, Araste FE, Ebrahimi R, et al. Effect of calcium on premenstrual syndrome: a double-blind randomized clinical trial. Obstet Gynecol Sci. 2017;60(1):100-105.
22. Nevatte T, O’Brien PMS, Bäckström T, et al. ISPMD consensus on the management of premenstrual disorders. Arch Womens Ment Health. 2013;16(4):279-291.
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Washington, DC: American Psychiatric Association; 2013.
2. Rapkin AJ, Lewis EI. Treatment of premenstrual dysphoric disorder. Womens Health (Lond). 2013;9(6):537-556.
3. Pearlstein T. Treatment of premenstrual dysphoric disorder: therapeutic challenges. Expert Rev Clin Pharmacol. 2016;9(4):493-496.
4. Zielinski R, Lynne S. Menstrual-cycle pain and premenstrual conditions. In: Schuiling KD, Likis FE, eds. Women’s Gynecologic Health. Burlington, MA: Jones & Bartlett Learning; 2017:556-573.
5. Hofmeister S, Bodden S. Premenstrual syndrome and premenstrual dysphoric disorder. Am Fam Physician. 2016;94(3):236-240.
6. Yonkers KA, Simoni MK. Premenstrual disorders. Am J Obstet Gynecol. 2018;218(1):68-74.
7. Yang M, Wallenstein G, Hagan M, et al. Burden of premenstrual dysphoric disorder on health-related quality of life. J Womens Health (Larchmt). 2008;17(1):113-121.
8. Craner JR, Sigmon ST, Women Health.
9. Hong JP, Park S, Wang HR, et al. Prevalence, correlates, comorbidities, and suicidal tendencies of premenstrual dysphoric disorder in a nationwide sample of Korean women. Soc Psychiatry Psychiatr Epidemiol. 2012;47(12): 1937-1945.
10. Martinez PE, Rubinow PR, Nieman LK, et al. 5α-reductase inhibition prevents the luteal phase increase in plasma allopregnanolone levels and mitigates symptoms in women with premenstrual dysphoric disorder. Neuropsychopharmacology. 2016;41:1093-1102.
11. Baller EB, Wei SM, Kohn PD. Abnormalities of dorsolateral prefrontal function in women with premenstrual dysphoric disorder: A multimodal neuroimaging study. Am J Psychiatry. 2013;170(3):305-314.
. EINeuroimaging the menstrual cycle and premenstrual dysphoric disorderCurr Psychiatry Rep.201577
13. Reid RL. Premenstrual dysphoric disorder (formerly premenstrual syndrome) [Updated Jan 23, 2017]. In: De Groot LJ, Chrousos G, Dungan K, et al, eds. Endotext [Internet]. South Dartmouth, MA: MDText.com, Inc; 2000.
14. Htay TT. Premenstrual dysphoric disorder clinical presentation. Medscape. https://emedicine.medscape.com/article/293257-clinical#b3. Updated February 16, 2016. Accessed February 7, 2018.
15. Epperson CN, Hantsoo LV. Making strides to simplify diagnosis of premenstrual dysphoric disorder. Am J Psychiatry. 2017;174(1):6-7.
16. FDA. Sarafem. www.accessdata.fda.gov/drugsatfda_docs/label/2006/021860lbl.pdf. Accessed February 15, 2018.
17. FDA. Paxil CR. www.accessdata.fda.gov/drugsatfda_docs/label/2004/20936se2-013_paxil_lbl.pdf. Accessed February 15, 2018.
18. FDA. Zoloft. www.accessdata.fda.gov/drugsatfda_docs/label/2016/019839s74s86s87_20990s35s44s45lbl.pdf. Accessed February 15, 2018.
19. Yonkers KA, Kornstein SG, Gueorguieva R, et al. Symptom-onset dosing of sertraline for the treatment of premenstrual dysphoric disorder: a randomized trial. JAMA Psychiatry. 2015;72(10):1037-1044.
20. Busse JW, Montori VM, Krasnik C, et al. Psychological intervention for premenstrual syndrome: a meta-analysis of randomized controlled trials. Psychother Psychosom. 2009;78(1):6-15.
21. Shobeiri F, Araste FE, Ebrahimi R, et al. Effect of calcium on premenstrual syndrome: a double-blind randomized clinical trial. Obstet Gynecol Sci. 2017;60(1):100-105.
22. Nevatte T, O’Brien PMS, Bäckström T, et al. ISPMD consensus on the management of premenstrual disorders. Arch Womens Ment Health. 2013;16(4):279-291.
Stay tuned
Two events that will impact our practices occurred in November: 1) an election and 2) the Centers for Medicare & Medicaid Services final rule. The election returned us to a split government with Democrats controlling the U.S. House and Republicans controlling the Senate (without a filibuster-proof majority). This means that ACA repeal and dramatic alterations to Medicaid will be off the table. Pressures on ACA’s margins will remain in both the legislative and judicial arms of government. Federal and state governments will continue to try to stabilize the individual markets by using reinsurance and premium support. The number of states expanding Medicaid eligibility will continue to grow (now at 37). There will be further pressure on drug pricing, likely targeted to Part B and 340b drugs. This will affect academic centers and hospital margins substantially.
CMS issued its final rule for the Physician Fee Schedule. AGA and the other GI societies have published a detailed member alert that can be found here. Key points involve simplified documentation for evaluation and management visits, site-neutrality reimbursement for clinic visits, identification of colonoscopy and EGD codes for CMS review, and changes in calculating practice expense, among others. MACRA rules are evolving with further pressure on practices and health systems to evolve into alternative payment models. Commercial insurers are finally near a tipping point in pressing for two-sided risk contracts. Practices should be alert for local and regional pressures around price transparency and narrow networks. Health systems (including academic centers) must plan for margin reductions due to changes in pharmacy reimbursement, network price tiering, a continued shift toward government payers, and other pressures that could drive large systems into the red.
For the first time since 1996, discretionary programs including NIH, CDC, AHRQ, and VA research all have been included in a budget (as opposed to a Continuing Resolution) that was passed by Congress and signed into law. This gives us some stability and predictability; however, the looming (and increasing) budget deficit will prompt Congress to increase fiscal pressure on domestic programs such as Social Security, Medicare, and Medicaid. Stay tuned and stay involved.
John I. Allen, MD, MBA, AGAF
Editor in Chief
Two events that will impact our practices occurred in November: 1) an election and 2) the Centers for Medicare & Medicaid Services final rule. The election returned us to a split government with Democrats controlling the U.S. House and Republicans controlling the Senate (without a filibuster-proof majority). This means that ACA repeal and dramatic alterations to Medicaid will be off the table. Pressures on ACA’s margins will remain in both the legislative and judicial arms of government. Federal and state governments will continue to try to stabilize the individual markets by using reinsurance and premium support. The number of states expanding Medicaid eligibility will continue to grow (now at 37). There will be further pressure on drug pricing, likely targeted to Part B and 340b drugs. This will affect academic centers and hospital margins substantially.
CMS issued its final rule for the Physician Fee Schedule. AGA and the other GI societies have published a detailed member alert that can be found here. Key points involve simplified documentation for evaluation and management visits, site-neutrality reimbursement for clinic visits, identification of colonoscopy and EGD codes for CMS review, and changes in calculating practice expense, among others. MACRA rules are evolving with further pressure on practices and health systems to evolve into alternative payment models. Commercial insurers are finally near a tipping point in pressing for two-sided risk contracts. Practices should be alert for local and regional pressures around price transparency and narrow networks. Health systems (including academic centers) must plan for margin reductions due to changes in pharmacy reimbursement, network price tiering, a continued shift toward government payers, and other pressures that could drive large systems into the red.
For the first time since 1996, discretionary programs including NIH, CDC, AHRQ, and VA research all have been included in a budget (as opposed to a Continuing Resolution) that was passed by Congress and signed into law. This gives us some stability and predictability; however, the looming (and increasing) budget deficit will prompt Congress to increase fiscal pressure on domestic programs such as Social Security, Medicare, and Medicaid. Stay tuned and stay involved.
John I. Allen, MD, MBA, AGAF
Editor in Chief
Two events that will impact our practices occurred in November: 1) an election and 2) the Centers for Medicare & Medicaid Services final rule. The election returned us to a split government with Democrats controlling the U.S. House and Republicans controlling the Senate (without a filibuster-proof majority). This means that ACA repeal and dramatic alterations to Medicaid will be off the table. Pressures on ACA’s margins will remain in both the legislative and judicial arms of government. Federal and state governments will continue to try to stabilize the individual markets by using reinsurance and premium support. The number of states expanding Medicaid eligibility will continue to grow (now at 37). There will be further pressure on drug pricing, likely targeted to Part B and 340b drugs. This will affect academic centers and hospital margins substantially.
CMS issued its final rule for the Physician Fee Schedule. AGA and the other GI societies have published a detailed member alert that can be found here. Key points involve simplified documentation for evaluation and management visits, site-neutrality reimbursement for clinic visits, identification of colonoscopy and EGD codes for CMS review, and changes in calculating practice expense, among others. MACRA rules are evolving with further pressure on practices and health systems to evolve into alternative payment models. Commercial insurers are finally near a tipping point in pressing for two-sided risk contracts. Practices should be alert for local and regional pressures around price transparency and narrow networks. Health systems (including academic centers) must plan for margin reductions due to changes in pharmacy reimbursement, network price tiering, a continued shift toward government payers, and other pressures that could drive large systems into the red.
For the first time since 1996, discretionary programs including NIH, CDC, AHRQ, and VA research all have been included in a budget (as opposed to a Continuing Resolution) that was passed by Congress and signed into law. This gives us some stability and predictability; however, the looming (and increasing) budget deficit will prompt Congress to increase fiscal pressure on domestic programs such as Social Security, Medicare, and Medicaid. Stay tuned and stay involved.
John I. Allen, MD, MBA, AGAF
Editor in Chief
FDA approves congenital CMV diagnostic test
, a new test to be used as an aid in the diagnosis of congenital cytomegalovirus (CMV) in newborns less than 21 days of age.
The Alethia CMV Assay Test System detects CMV DNA from a saliva swab. Results from the test should be used only in conjunction with the results of other diagnostic tests and clinical information, according to an FDA statement.
“This test for detecting the virus, when used in conjunction with the results of other diagnostic tests, may help health care providers more quickly identify the virus in newborns,” said Timothy Stenzel, PhD, director of the Office of In Vitro Diagnostics and Radiological Health in the FDA’s Center for Devices and Radiological Health.
In a prospective clinical study, 1,472 saliva samples out of 1,475 samples collected from newborns were correctly identified by the device as negative for the presence of CMV DNA. Three samples were incorrectly identified as positive when they were negative. Five collected saliva specimens were correctly identified as positive for the presence of CMV DNA.
In a testing of 34 samples of archived specimens from babies known to be infected with CMV, all of the archived specimens were correctly identified by the device as positive for the presence of CMV DNA.
The FDA reviewed the Alethia CMV Assay Test System through a regulatory pathway established for novel, low- to moderate-risk devices. Along with this authorization, the FDA is establishing criteria, called special controls, which determine the requirements for demonstrating accuracy, reliability, and effectiveness of tests intended to be used as an aid in the diagnosis of congenital CMV infection.
With this new regulatory classification, subsequent devices of the same type with the same intended use may go through the FDA’s 510(k) process, whereby devices can obtain marketing authorization by demonstrating substantial equivalence to a previously approved device.
The FDA granted marketing authorization of the Alethia CMV Assay Test System to Meridian Bioscience.
, a new test to be used as an aid in the diagnosis of congenital cytomegalovirus (CMV) in newborns less than 21 days of age.
The Alethia CMV Assay Test System detects CMV DNA from a saliva swab. Results from the test should be used only in conjunction with the results of other diagnostic tests and clinical information, according to an FDA statement.
“This test for detecting the virus, when used in conjunction with the results of other diagnostic tests, may help health care providers more quickly identify the virus in newborns,” said Timothy Stenzel, PhD, director of the Office of In Vitro Diagnostics and Radiological Health in the FDA’s Center for Devices and Radiological Health.
In a prospective clinical study, 1,472 saliva samples out of 1,475 samples collected from newborns were correctly identified by the device as negative for the presence of CMV DNA. Three samples were incorrectly identified as positive when they were negative. Five collected saliva specimens were correctly identified as positive for the presence of CMV DNA.
In a testing of 34 samples of archived specimens from babies known to be infected with CMV, all of the archived specimens were correctly identified by the device as positive for the presence of CMV DNA.
The FDA reviewed the Alethia CMV Assay Test System through a regulatory pathway established for novel, low- to moderate-risk devices. Along with this authorization, the FDA is establishing criteria, called special controls, which determine the requirements for demonstrating accuracy, reliability, and effectiveness of tests intended to be used as an aid in the diagnosis of congenital CMV infection.
With this new regulatory classification, subsequent devices of the same type with the same intended use may go through the FDA’s 510(k) process, whereby devices can obtain marketing authorization by demonstrating substantial equivalence to a previously approved device.
The FDA granted marketing authorization of the Alethia CMV Assay Test System to Meridian Bioscience.
, a new test to be used as an aid in the diagnosis of congenital cytomegalovirus (CMV) in newborns less than 21 days of age.
The Alethia CMV Assay Test System detects CMV DNA from a saliva swab. Results from the test should be used only in conjunction with the results of other diagnostic tests and clinical information, according to an FDA statement.
“This test for detecting the virus, when used in conjunction with the results of other diagnostic tests, may help health care providers more quickly identify the virus in newborns,” said Timothy Stenzel, PhD, director of the Office of In Vitro Diagnostics and Radiological Health in the FDA’s Center for Devices and Radiological Health.
In a prospective clinical study, 1,472 saliva samples out of 1,475 samples collected from newborns were correctly identified by the device as negative for the presence of CMV DNA. Three samples were incorrectly identified as positive when they were negative. Five collected saliva specimens were correctly identified as positive for the presence of CMV DNA.
In a testing of 34 samples of archived specimens from babies known to be infected with CMV, all of the archived specimens were correctly identified by the device as positive for the presence of CMV DNA.
The FDA reviewed the Alethia CMV Assay Test System through a regulatory pathway established for novel, low- to moderate-risk devices. Along with this authorization, the FDA is establishing criteria, called special controls, which determine the requirements for demonstrating accuracy, reliability, and effectiveness of tests intended to be used as an aid in the diagnosis of congenital CMV infection.
With this new regulatory classification, subsequent devices of the same type with the same intended use may go through the FDA’s 510(k) process, whereby devices can obtain marketing authorization by demonstrating substantial equivalence to a previously approved device.
The FDA granted marketing authorization of the Alethia CMV Assay Test System to Meridian Bioscience.
What is your diagnosis? - December 2018
Cecal carcinoma–associated paraneoplastic Sweet’s syndrome
Based on the tomographic appearance of an “apple core”–like lesion in the right lower quadrant, the patient was referred for colonoscopy, which revealed a malignant-appearing cecal mass (Figure D), with biopsies confirming adenocarcinoma; despite these findings, no bowel-related symptoms were reported. The patient underwent laparoscopic right hemicolectomy, after which the skin lesions began to resolve, and corticosteroids were successfully tapered. The overall presentation was consistent with Sweet’s syndrome, with a paraneoplastic etiology being favored given the clinical scenario, including absence of alternative etiologies and dependence on corticosteroids for control of skin disease until resection of the underlying malignancy was performed.
Sweet’s syndrome was first described in a case series of eight patients published in 1964 by the English dermatologist Dr. Robert Douglas Sweet.1,2 Sweet’s syndrome is characterized by fever, neutrophilia, and sterile erythematous plaques or nodules, which most commonly involve the upper extremities and face and respond to corticosteroid therapy. It may be malignancy associated, drug induced, autoimmune disease related, or idiopathic.2,3 The pathogenesis of Sweet’s syndrome is unclear, but T-lymphocyte, neutrophil chemotaxis, and cytokine (e.g., interleukin-6 and granulocyte colony–stimulating factor) abnormalities have been suggested.2 Diagnosis is based on the clinical presentation and context together with typical dermatopathologic findings, including a dense neutrophilic infiltrate. Skin lesions may be phasic, but persist typically until appropriate therapy (e.g., corticosteroids, chemotherapy) is administered or the offending drug removed. Malignancy-associated (i.e., paraneoplastic) Sweet’s syndrome accounts for approximately 20% of all cases; these primarily involve hematologic malignancies, most commonly leukemia, but adenocarcinomata have also been implicated.3 Recurrence of Sweet’s syndrome can occur and often heralds relapse of the underlying disease.
References
1. Sweet RD. An acute febrile neutrophilic dermatosis. Br J Dermatol. 1964;76:349-56.
2. Von den Driesch P. Sweet’s syndrome (acute febrile neutrophilic dermatosis). J Am Acad Dermatol. 1994;31:535-56.
3. Cohen PR. Sweet’s syndrome–a comprehensive review of an acute febrile neutrophilic dermatosis. Orphanet J Rare Dis. 2007;2:34.
Cecal carcinoma–associated paraneoplastic Sweet’s syndrome
Based on the tomographic appearance of an “apple core”–like lesion in the right lower quadrant, the patient was referred for colonoscopy, which revealed a malignant-appearing cecal mass (Figure D), with biopsies confirming adenocarcinoma; despite these findings, no bowel-related symptoms were reported. The patient underwent laparoscopic right hemicolectomy, after which the skin lesions began to resolve, and corticosteroids were successfully tapered. The overall presentation was consistent with Sweet’s syndrome, with a paraneoplastic etiology being favored given the clinical scenario, including absence of alternative etiologies and dependence on corticosteroids for control of skin disease until resection of the underlying malignancy was performed.
Sweet’s syndrome was first described in a case series of eight patients published in 1964 by the English dermatologist Dr. Robert Douglas Sweet.1,2 Sweet’s syndrome is characterized by fever, neutrophilia, and sterile erythematous plaques or nodules, which most commonly involve the upper extremities and face and respond to corticosteroid therapy. It may be malignancy associated, drug induced, autoimmune disease related, or idiopathic.2,3 The pathogenesis of Sweet’s syndrome is unclear, but T-lymphocyte, neutrophil chemotaxis, and cytokine (e.g., interleukin-6 and granulocyte colony–stimulating factor) abnormalities have been suggested.2 Diagnosis is based on the clinical presentation and context together with typical dermatopathologic findings, including a dense neutrophilic infiltrate. Skin lesions may be phasic, but persist typically until appropriate therapy (e.g., corticosteroids, chemotherapy) is administered or the offending drug removed. Malignancy-associated (i.e., paraneoplastic) Sweet’s syndrome accounts for approximately 20% of all cases; these primarily involve hematologic malignancies, most commonly leukemia, but adenocarcinomata have also been implicated.3 Recurrence of Sweet’s syndrome can occur and often heralds relapse of the underlying disease.
References
1. Sweet RD. An acute febrile neutrophilic dermatosis. Br J Dermatol. 1964;76:349-56.
2. Von den Driesch P. Sweet’s syndrome (acute febrile neutrophilic dermatosis). J Am Acad Dermatol. 1994;31:535-56.
3. Cohen PR. Sweet’s syndrome–a comprehensive review of an acute febrile neutrophilic dermatosis. Orphanet J Rare Dis. 2007;2:34.
Cecal carcinoma–associated paraneoplastic Sweet’s syndrome
Based on the tomographic appearance of an “apple core”–like lesion in the right lower quadrant, the patient was referred for colonoscopy, which revealed a malignant-appearing cecal mass (Figure D), with biopsies confirming adenocarcinoma; despite these findings, no bowel-related symptoms were reported. The patient underwent laparoscopic right hemicolectomy, after which the skin lesions began to resolve, and corticosteroids were successfully tapered. The overall presentation was consistent with Sweet’s syndrome, with a paraneoplastic etiology being favored given the clinical scenario, including absence of alternative etiologies and dependence on corticosteroids for control of skin disease until resection of the underlying malignancy was performed.
Sweet’s syndrome was first described in a case series of eight patients published in 1964 by the English dermatologist Dr. Robert Douglas Sweet.1,2 Sweet’s syndrome is characterized by fever, neutrophilia, and sterile erythematous plaques or nodules, which most commonly involve the upper extremities and face and respond to corticosteroid therapy. It may be malignancy associated, drug induced, autoimmune disease related, or idiopathic.2,3 The pathogenesis of Sweet’s syndrome is unclear, but T-lymphocyte, neutrophil chemotaxis, and cytokine (e.g., interleukin-6 and granulocyte colony–stimulating factor) abnormalities have been suggested.2 Diagnosis is based on the clinical presentation and context together with typical dermatopathologic findings, including a dense neutrophilic infiltrate. Skin lesions may be phasic, but persist typically until appropriate therapy (e.g., corticosteroids, chemotherapy) is administered or the offending drug removed. Malignancy-associated (i.e., paraneoplastic) Sweet’s syndrome accounts for approximately 20% of all cases; these primarily involve hematologic malignancies, most commonly leukemia, but adenocarcinomata have also been implicated.3 Recurrence of Sweet’s syndrome can occur and often heralds relapse of the underlying disease.
References
1. Sweet RD. An acute febrile neutrophilic dermatosis. Br J Dermatol. 1964;76:349-56.
2. Von den Driesch P. Sweet’s syndrome (acute febrile neutrophilic dermatosis). J Am Acad Dermatol. 1994;31:535-56.
3. Cohen PR. Sweet’s syndrome–a comprehensive review of an acute febrile neutrophilic dermatosis. Orphanet J Rare Dis. 2007;2:34.
What is the diagnosis?
Firdapse approved: First treatment for rare autoimmune disorder
The Food and Drug Administration has approved amifampridine (Firdapse) as the first treatment for the rare autoimmune disorder known as Lambert-Eaton myasthenic syndrome, which causes the immune system to attack the neuromuscular junction and thereby disrupts the nerves’ ability to send signals to muscle cells. This causes fatigue and weakness in those affected, so they can experience difficulties with activities of daily living as a result.
The most common side effects included prickling sensation, upper respiratory tract infection, abdominal pain, and muscle spasms.
More information can be found in the FDA’s press announcement.
cpalmer@mdedge.com
The Food and Drug Administration has approved amifampridine (Firdapse) as the first treatment for the rare autoimmune disorder known as Lambert-Eaton myasthenic syndrome, which causes the immune system to attack the neuromuscular junction and thereby disrupts the nerves’ ability to send signals to muscle cells. This causes fatigue and weakness in those affected, so they can experience difficulties with activities of daily living as a result.
The most common side effects included prickling sensation, upper respiratory tract infection, abdominal pain, and muscle spasms.
More information can be found in the FDA’s press announcement.
cpalmer@mdedge.com
The Food and Drug Administration has approved amifampridine (Firdapse) as the first treatment for the rare autoimmune disorder known as Lambert-Eaton myasthenic syndrome, which causes the immune system to attack the neuromuscular junction and thereby disrupts the nerves’ ability to send signals to muscle cells. This causes fatigue and weakness in those affected, so they can experience difficulties with activities of daily living as a result.
The most common side effects included prickling sensation, upper respiratory tract infection, abdominal pain, and muscle spasms.
More information can be found in the FDA’s press announcement.
cpalmer@mdedge.com
DMARDs improve vascular abnormalities in early RA
, Maya H. Buch, PhD, said at the annual meeting of the American College of Rheumatology.
She presented a study of 71 treatment-naive patients with recently diagnosed RA and no history of cardiovascular disease. Patients were randomized to either etanercept plus methotrexate or methotrexate with treat-to-target escalation to triple nonbiologic disease-modifying antirheumatic drug therapy by week 12. Patients in the latter group were switched to etanercept plus methotrexate at week 24 if at that point they had a Disease Activity Score 28 of 2.6 or more.
All participants underwent cardiovascular MRI at baseline and again after 1 and 2 years in order to measure change in aortic stiffness, the primary study endpoint chosen because it is clinically meaningful and can reliably and reproducibly be measured by MRI.
The study was designed to shed light on possible mechanisms for the epidemiologic observation that effective treatment with methotrexate and/or tumor necrosis factor (TNF) inhibitors curbs the excess risk of cardiovascular events that accompanies RA. The thinking is that abnormal aortic distensibility represents an early harbinger of the increased cardiovascular risk associated with this form of inflammatory arthritis, explained Dr. Buch, professor of rheumatology at the University of Leeds (England) and section head of clinical and translational rheumatology at the Leeds Institute of Rheumatic and Musculoskeletal Medicine.
The mean aortic distensibility at baseline, when the study population was treatment naive, was 2.99 x 10–3 mm Hg–1. This value is abnormally low when compared with values seen in healthy age- and sex-matched controls. After 1 year of treatment, however, mean aortic distensibility improved significantly to 3.59 x 10–3 mm Hg–1. This improvement was maintained at year 2, when the mean value was 3.55.
Interestingly, neither treatment response status or disease activity was associated with the improvement in aortic stiffness. However, in a prespecified exploratory comparison between the 20 responders to etanercept plus methotrexate and the 10 responders to first-line methotrexate who never received etanercept, the group on biologic therapy had a 16% greater improvement in aortic distensibility at 1 year.
“It’s not a statistically significant difference. The study wasn’t powered for that,” she said in an interview. “But these data suggest that an anti-TNF treatment strategy may confer additional benefit. Of course, that will need to be confirmed in a larger trial. And if it is confirmed, it would suggest a role for etanercept plus methotrexate in personalizing tailored therapy in early RA patients at particularly high cardiovascular risk.”
Dr. Buch reported receiving funding from Pfizer, Roche, UCB, AbbVie, Sanofi, Eli Lilly, and Sandoz, but reported having no financial conflicts of interest regarding this study, which was sponsored by the U.K. National Institute for Healthcare Research.
SOURCE: Buch MH et al. Arthritis Rheumatol. 2018;70(Suppl 10), Abstract L05.
, Maya H. Buch, PhD, said at the annual meeting of the American College of Rheumatology.
She presented a study of 71 treatment-naive patients with recently diagnosed RA and no history of cardiovascular disease. Patients were randomized to either etanercept plus methotrexate or methotrexate with treat-to-target escalation to triple nonbiologic disease-modifying antirheumatic drug therapy by week 12. Patients in the latter group were switched to etanercept plus methotrexate at week 24 if at that point they had a Disease Activity Score 28 of 2.6 or more.
All participants underwent cardiovascular MRI at baseline and again after 1 and 2 years in order to measure change in aortic stiffness, the primary study endpoint chosen because it is clinically meaningful and can reliably and reproducibly be measured by MRI.
The study was designed to shed light on possible mechanisms for the epidemiologic observation that effective treatment with methotrexate and/or tumor necrosis factor (TNF) inhibitors curbs the excess risk of cardiovascular events that accompanies RA. The thinking is that abnormal aortic distensibility represents an early harbinger of the increased cardiovascular risk associated with this form of inflammatory arthritis, explained Dr. Buch, professor of rheumatology at the University of Leeds (England) and section head of clinical and translational rheumatology at the Leeds Institute of Rheumatic and Musculoskeletal Medicine.
The mean aortic distensibility at baseline, when the study population was treatment naive, was 2.99 x 10–3 mm Hg–1. This value is abnormally low when compared with values seen in healthy age- and sex-matched controls. After 1 year of treatment, however, mean aortic distensibility improved significantly to 3.59 x 10–3 mm Hg–1. This improvement was maintained at year 2, when the mean value was 3.55.
Interestingly, neither treatment response status or disease activity was associated with the improvement in aortic stiffness. However, in a prespecified exploratory comparison between the 20 responders to etanercept plus methotrexate and the 10 responders to first-line methotrexate who never received etanercept, the group on biologic therapy had a 16% greater improvement in aortic distensibility at 1 year.
“It’s not a statistically significant difference. The study wasn’t powered for that,” she said in an interview. “But these data suggest that an anti-TNF treatment strategy may confer additional benefit. Of course, that will need to be confirmed in a larger trial. And if it is confirmed, it would suggest a role for etanercept plus methotrexate in personalizing tailored therapy in early RA patients at particularly high cardiovascular risk.”
Dr. Buch reported receiving funding from Pfizer, Roche, UCB, AbbVie, Sanofi, Eli Lilly, and Sandoz, but reported having no financial conflicts of interest regarding this study, which was sponsored by the U.K. National Institute for Healthcare Research.
SOURCE: Buch MH et al. Arthritis Rheumatol. 2018;70(Suppl 10), Abstract L05.
, Maya H. Buch, PhD, said at the annual meeting of the American College of Rheumatology.
She presented a study of 71 treatment-naive patients with recently diagnosed RA and no history of cardiovascular disease. Patients were randomized to either etanercept plus methotrexate or methotrexate with treat-to-target escalation to triple nonbiologic disease-modifying antirheumatic drug therapy by week 12. Patients in the latter group were switched to etanercept plus methotrexate at week 24 if at that point they had a Disease Activity Score 28 of 2.6 or more.
All participants underwent cardiovascular MRI at baseline and again after 1 and 2 years in order to measure change in aortic stiffness, the primary study endpoint chosen because it is clinically meaningful and can reliably and reproducibly be measured by MRI.
The study was designed to shed light on possible mechanisms for the epidemiologic observation that effective treatment with methotrexate and/or tumor necrosis factor (TNF) inhibitors curbs the excess risk of cardiovascular events that accompanies RA. The thinking is that abnormal aortic distensibility represents an early harbinger of the increased cardiovascular risk associated with this form of inflammatory arthritis, explained Dr. Buch, professor of rheumatology at the University of Leeds (England) and section head of clinical and translational rheumatology at the Leeds Institute of Rheumatic and Musculoskeletal Medicine.
The mean aortic distensibility at baseline, when the study population was treatment naive, was 2.99 x 10–3 mm Hg–1. This value is abnormally low when compared with values seen in healthy age- and sex-matched controls. After 1 year of treatment, however, mean aortic distensibility improved significantly to 3.59 x 10–3 mm Hg–1. This improvement was maintained at year 2, when the mean value was 3.55.
Interestingly, neither treatment response status or disease activity was associated with the improvement in aortic stiffness. However, in a prespecified exploratory comparison between the 20 responders to etanercept plus methotrexate and the 10 responders to first-line methotrexate who never received etanercept, the group on biologic therapy had a 16% greater improvement in aortic distensibility at 1 year.
“It’s not a statistically significant difference. The study wasn’t powered for that,” she said in an interview. “But these data suggest that an anti-TNF treatment strategy may confer additional benefit. Of course, that will need to be confirmed in a larger trial. And if it is confirmed, it would suggest a role for etanercept plus methotrexate in personalizing tailored therapy in early RA patients at particularly high cardiovascular risk.”
Dr. Buch reported receiving funding from Pfizer, Roche, UCB, AbbVie, Sanofi, Eli Lilly, and Sandoz, but reported having no financial conflicts of interest regarding this study, which was sponsored by the U.K. National Institute for Healthcare Research.
SOURCE: Buch MH et al. Arthritis Rheumatol. 2018;70(Suppl 10), Abstract L05.
REPORTING FROM THE ACR ANNUAL MEETING
Key clinical point: Abnormal aortic stiffness is present in patients with newly diagnosed RA but improves with disease-modifying antirheumatic drug therapy.
Major finding: Mean aortic distensibility in patients with early RA improved from an abnormally low 2.99 x 10–3 mm Hg–1 prior to treatment to a more robust 3.59 x 10–3 mm Hg–1 after 1 year of disease-modifying antirheumatic drug therapy.
Study details: This study included 71 patients with newly diagnosed RA whose aortic stiffness was measured serially via cardiovascular MRI over a 2-year period.
Disclosures: The presenter reported receiving funding from Pfizer, Roche, UCB, AbbVie, Sanofi, Eli Lilly, and Sandoz, but reported having no financial conflicts of interest regarding this study, which was sponsored by the U.K. National Institute for Healthcare Research.
Source: Buch MH et al. Arthritis Rheumatol. 2018;70(Suppl 10), Abstract L05.
PCOS linked to increased cancer risk in premenopausal women
based on an analysis of nearly 3.5 million women in a large Swedish database.
Women with PCOS had a sixfold increased risk of endometrial cancer, a tripling of endocrine gland cancers, and more than a doubling in the risk of ovarian and pancreatic cancers. Once women reached menopausal status, however, their cancer risk was comparable to that of women without a history of PCOS.
“Several carcinogenic processes are associated with PCOS, including dyslipidemia, hyperinsulinemia, and chronic inflammation,” wrote Weimin Ye, MD, PhD, of the Karolinska Institutet, Stockholm, and his colleagues. “Our study indicates that cancer may need to be added to the spectrum of long-term health consequences of PCOS and warrants increased surveillance among those patients.”
The research letter was published online in JAMA Oncology.
The team examined the relationship between PCOS and primary cancers in about 3.5 million women over a span of up to 24 years (1985-2009), although the mean follow-up time was not mentioned. To examine the potential impact of menopause, they conducted separate multivariate logistic regression analyses for those younger than 51 years, and those aged 51 years or older. The analyses controlled for use of some medications (metformin, oral contraceptives, and hormone therapy); as well as educational level (a proxy for socioeconomic status); smoking; parity (a proxy for fertility); parental cancers; and diabetes.
Overall, 14,764 women had been diagnosed with PCOS; they were a mean of 28 years at baseline and 182 developed a primary cancer 1 year or more after PCOS diagnosis.
These women had a 15% overall increased risk of cancer, compared with women without PCOS.
The risks for specific cancers also were increased, compared with women without PCOS, including endometrial (hazard ratio, 2.62), ovarian (HR, 2.16), endocrine (HR, 1.92), pancreatic (HR, 3.4), kidney (HR, 3.0), and skeletal and hematopoietic (HR, 1.69) cancers.
The risks were associated with younger age, however. In the group under age 51 years, the overall risk was 22% higher. The increased risk of specific cancers were endometrial (HR, 6.45), ovarian (HR, 2.55), pancreatic (HR, 6.68), kidney (HR, 4.57), and endocrine (not thyroid) gland (HR, 2.9) cancers.
The authors had no relevant financial disclosures.
SOURCE: Yin W et al. JAMA Oncol. 2018 Nov 29. doi:10.1001/jamaoncol.2018.5188.
based on an analysis of nearly 3.5 million women in a large Swedish database.
Women with PCOS had a sixfold increased risk of endometrial cancer, a tripling of endocrine gland cancers, and more than a doubling in the risk of ovarian and pancreatic cancers. Once women reached menopausal status, however, their cancer risk was comparable to that of women without a history of PCOS.
“Several carcinogenic processes are associated with PCOS, including dyslipidemia, hyperinsulinemia, and chronic inflammation,” wrote Weimin Ye, MD, PhD, of the Karolinska Institutet, Stockholm, and his colleagues. “Our study indicates that cancer may need to be added to the spectrum of long-term health consequences of PCOS and warrants increased surveillance among those patients.”
The research letter was published online in JAMA Oncology.
The team examined the relationship between PCOS and primary cancers in about 3.5 million women over a span of up to 24 years (1985-2009), although the mean follow-up time was not mentioned. To examine the potential impact of menopause, they conducted separate multivariate logistic regression analyses for those younger than 51 years, and those aged 51 years or older. The analyses controlled for use of some medications (metformin, oral contraceptives, and hormone therapy); as well as educational level (a proxy for socioeconomic status); smoking; parity (a proxy for fertility); parental cancers; and diabetes.
Overall, 14,764 women had been diagnosed with PCOS; they were a mean of 28 years at baseline and 182 developed a primary cancer 1 year or more after PCOS diagnosis.
These women had a 15% overall increased risk of cancer, compared with women without PCOS.
The risks for specific cancers also were increased, compared with women without PCOS, including endometrial (hazard ratio, 2.62), ovarian (HR, 2.16), endocrine (HR, 1.92), pancreatic (HR, 3.4), kidney (HR, 3.0), and skeletal and hematopoietic (HR, 1.69) cancers.
The risks were associated with younger age, however. In the group under age 51 years, the overall risk was 22% higher. The increased risk of specific cancers were endometrial (HR, 6.45), ovarian (HR, 2.55), pancreatic (HR, 6.68), kidney (HR, 4.57), and endocrine (not thyroid) gland (HR, 2.9) cancers.
The authors had no relevant financial disclosures.
SOURCE: Yin W et al. JAMA Oncol. 2018 Nov 29. doi:10.1001/jamaoncol.2018.5188.
based on an analysis of nearly 3.5 million women in a large Swedish database.
Women with PCOS had a sixfold increased risk of endometrial cancer, a tripling of endocrine gland cancers, and more than a doubling in the risk of ovarian and pancreatic cancers. Once women reached menopausal status, however, their cancer risk was comparable to that of women without a history of PCOS.
“Several carcinogenic processes are associated with PCOS, including dyslipidemia, hyperinsulinemia, and chronic inflammation,” wrote Weimin Ye, MD, PhD, of the Karolinska Institutet, Stockholm, and his colleagues. “Our study indicates that cancer may need to be added to the spectrum of long-term health consequences of PCOS and warrants increased surveillance among those patients.”
The research letter was published online in JAMA Oncology.
The team examined the relationship between PCOS and primary cancers in about 3.5 million women over a span of up to 24 years (1985-2009), although the mean follow-up time was not mentioned. To examine the potential impact of menopause, they conducted separate multivariate logistic regression analyses for those younger than 51 years, and those aged 51 years or older. The analyses controlled for use of some medications (metformin, oral contraceptives, and hormone therapy); as well as educational level (a proxy for socioeconomic status); smoking; parity (a proxy for fertility); parental cancers; and diabetes.
Overall, 14,764 women had been diagnosed with PCOS; they were a mean of 28 years at baseline and 182 developed a primary cancer 1 year or more after PCOS diagnosis.
These women had a 15% overall increased risk of cancer, compared with women without PCOS.
The risks for specific cancers also were increased, compared with women without PCOS, including endometrial (hazard ratio, 2.62), ovarian (HR, 2.16), endocrine (HR, 1.92), pancreatic (HR, 3.4), kidney (HR, 3.0), and skeletal and hematopoietic (HR, 1.69) cancers.
The risks were associated with younger age, however. In the group under age 51 years, the overall risk was 22% higher. The increased risk of specific cancers were endometrial (HR, 6.45), ovarian (HR, 2.55), pancreatic (HR, 6.68), kidney (HR, 4.57), and endocrine (not thyroid) gland (HR, 2.9) cancers.
The authors had no relevant financial disclosures.
SOURCE: Yin W et al. JAMA Oncol. 2018 Nov 29. doi:10.1001/jamaoncol.2018.5188.
FROM JAMA ONCOLOGY
Key clinical point: Polycystic ovarian syndrome may be associated with increased cancer risks among younger women.
Major finding: Among premenopausal women, there was a sixfold increased risk of endometrial cancer, a tripling of endocrine gland cancers, and a more than doubling in the risk of ovarian and pancreatic cancers
Study details: The study examined risks in 3.5 million women with up to 24 years of follow-up.
Disclosures: The study authors had no financial disclosures.
Source: Yin W et al. JAMA Oncol. 2018 Nov 29. doi:10.1001/jamaoncol.2018.5188.
CNS lymphoma guidelines stress patient fitness, not age, in choosing treatment
for the diagnosis and management of primary central nervous system diffuse large B‐cell lymphoma.
PCNSL, implicated in some 3% of all brain tumors, is complex to diagnose and treat. People with suspected PCNSL must receive quick and coordinated attention from a multidisciplinary team of neurologists, hematologist-oncologists, and ocular specialists, according to the guidelines, published in the British Journal of Haematology.
Christopher P. Fox, MD, of the Nottingham (England) University Hospitals NHS Trust, and his colleagues, stress the importance of early multidisciplinary attention, aggressive induction treatment, helping patients into trials, universal screening for eye involvement, attaining histological diagnoses in addition to imaging findings, and avoidance or discontinuation of any corticosteroids before biopsy, as even a short course of steroids can impede diagnosis.
The guidelines incorporate findings from studies published since the society’s last comprehensive PCNSL guideline was issued more than a decade ago.
Dr. Fox and his colleagues say definitive treatment for PCNSL – induction of remission followed by consolidation – should start within 2 weeks of diagnosis and that a treatment regimen should be chosen according to a patient’s physiological fitness, not age. The fittest patients, who have better organ function and fewer comorbidities, should be eligible for intensive combination immunochemotherapy incorporating high-dose methotrexate (optimally four cycles of HD-MTX, cytarabine, thiotepa, and rituximab). Those deemed unfit for this regimen should be offered induction treatment with HD-MTX, rituximab and procarbazine, the guidelines’ authors say.
If patients cannot tolerate HD-MTX, oral chemotherapy and/or whole-brain radiotherapy may be offered. Response should be assessed with contrast-enhanced magnetic resonance imaging.
Consolidation therapy should be initiated after induction for all patients with nonprogressive disease, and high-dose thiotepa-based chemotherapy with autologous stem cell transplant is the recommended first-line option for consolidation. Response to consolidation, again measured with contrast-enhanced MRI, should be carried out at between 1 and 2 months after therapy is completed, and patients should be referred for neuropsychological testing to assess cognitive function.
Patients with relapsed or refractory disease should be approached with maximum urgency – the guidelines offer an algorithm for retreatment options – and offered clinical trial entry wherever possible.
The PCNSL guideline writing process was sponsored by the British Society for Haematology, and some coauthors, including the lead author, disclosed receiving fees from pharmaceutical manufacturers Adienne or F. Hoffman-La Roche.
SOURCE: Fox et al. Br J Haematol. 2018 Nov 23 doi: 10.1111/bjh.15661.
for the diagnosis and management of primary central nervous system diffuse large B‐cell lymphoma.
PCNSL, implicated in some 3% of all brain tumors, is complex to diagnose and treat. People with suspected PCNSL must receive quick and coordinated attention from a multidisciplinary team of neurologists, hematologist-oncologists, and ocular specialists, according to the guidelines, published in the British Journal of Haematology.
Christopher P. Fox, MD, of the Nottingham (England) University Hospitals NHS Trust, and his colleagues, stress the importance of early multidisciplinary attention, aggressive induction treatment, helping patients into trials, universal screening for eye involvement, attaining histological diagnoses in addition to imaging findings, and avoidance or discontinuation of any corticosteroids before biopsy, as even a short course of steroids can impede diagnosis.
The guidelines incorporate findings from studies published since the society’s last comprehensive PCNSL guideline was issued more than a decade ago.
Dr. Fox and his colleagues say definitive treatment for PCNSL – induction of remission followed by consolidation – should start within 2 weeks of diagnosis and that a treatment regimen should be chosen according to a patient’s physiological fitness, not age. The fittest patients, who have better organ function and fewer comorbidities, should be eligible for intensive combination immunochemotherapy incorporating high-dose methotrexate (optimally four cycles of HD-MTX, cytarabine, thiotepa, and rituximab). Those deemed unfit for this regimen should be offered induction treatment with HD-MTX, rituximab and procarbazine, the guidelines’ authors say.
If patients cannot tolerate HD-MTX, oral chemotherapy and/or whole-brain radiotherapy may be offered. Response should be assessed with contrast-enhanced magnetic resonance imaging.
Consolidation therapy should be initiated after induction for all patients with nonprogressive disease, and high-dose thiotepa-based chemotherapy with autologous stem cell transplant is the recommended first-line option for consolidation. Response to consolidation, again measured with contrast-enhanced MRI, should be carried out at between 1 and 2 months after therapy is completed, and patients should be referred for neuropsychological testing to assess cognitive function.
Patients with relapsed or refractory disease should be approached with maximum urgency – the guidelines offer an algorithm for retreatment options – and offered clinical trial entry wherever possible.
The PCNSL guideline writing process was sponsored by the British Society for Haematology, and some coauthors, including the lead author, disclosed receiving fees from pharmaceutical manufacturers Adienne or F. Hoffman-La Roche.
SOURCE: Fox et al. Br J Haematol. 2018 Nov 23 doi: 10.1111/bjh.15661.
for the diagnosis and management of primary central nervous system diffuse large B‐cell lymphoma.
PCNSL, implicated in some 3% of all brain tumors, is complex to diagnose and treat. People with suspected PCNSL must receive quick and coordinated attention from a multidisciplinary team of neurologists, hematologist-oncologists, and ocular specialists, according to the guidelines, published in the British Journal of Haematology.
Christopher P. Fox, MD, of the Nottingham (England) University Hospitals NHS Trust, and his colleagues, stress the importance of early multidisciplinary attention, aggressive induction treatment, helping patients into trials, universal screening for eye involvement, attaining histological diagnoses in addition to imaging findings, and avoidance or discontinuation of any corticosteroids before biopsy, as even a short course of steroids can impede diagnosis.
The guidelines incorporate findings from studies published since the society’s last comprehensive PCNSL guideline was issued more than a decade ago.
Dr. Fox and his colleagues say definitive treatment for PCNSL – induction of remission followed by consolidation – should start within 2 weeks of diagnosis and that a treatment regimen should be chosen according to a patient’s physiological fitness, not age. The fittest patients, who have better organ function and fewer comorbidities, should be eligible for intensive combination immunochemotherapy incorporating high-dose methotrexate (optimally four cycles of HD-MTX, cytarabine, thiotepa, and rituximab). Those deemed unfit for this regimen should be offered induction treatment with HD-MTX, rituximab and procarbazine, the guidelines’ authors say.
If patients cannot tolerate HD-MTX, oral chemotherapy and/or whole-brain radiotherapy may be offered. Response should be assessed with contrast-enhanced magnetic resonance imaging.
Consolidation therapy should be initiated after induction for all patients with nonprogressive disease, and high-dose thiotepa-based chemotherapy with autologous stem cell transplant is the recommended first-line option for consolidation. Response to consolidation, again measured with contrast-enhanced MRI, should be carried out at between 1 and 2 months after therapy is completed, and patients should be referred for neuropsychological testing to assess cognitive function.
Patients with relapsed or refractory disease should be approached with maximum urgency – the guidelines offer an algorithm for retreatment options – and offered clinical trial entry wherever possible.
The PCNSL guideline writing process was sponsored by the British Society for Haematology, and some coauthors, including the lead author, disclosed receiving fees from pharmaceutical manufacturers Adienne or F. Hoffman-La Roche.
SOURCE: Fox et al. Br J Haematol. 2018 Nov 23 doi: 10.1111/bjh.15661.
FROM THE BRITISH JOURNAL OF HAEMATOLOGY
Stereotactic radiotherapy highly effective for brain mets in RCC
Stereotactic radiosurgery (SRS) is being used increasingly for brain metastases to prevent central nervous system (CNS) toxicity, and new findings published in Clinical Genitourinary Cancer demonstrate that it is a highly effective procedure for brain metastases from renal cell carcinoma (RCC).
In this study, the 1-year local control rate was more than 90% and durability of control was up to 2 years. SRS appears to be the most efficacious for smaller metastases, while escalated dosing or fractionation are needed to effectively treat larger lesions.
In metastatic RCC, the incidence of brain metastases is as high as 15%, and while surgical resection is associated with an improved survival compared with whole brain radiotherapy alone, for patients who are not candidates for resection, SRS is an alternative option. For this study, the authors sought to determine outcomes after treatment with SRS for brain metastases in 268 patients with metastatic RCC who were treated between 2006 and 2015 at a single institution.
Of this group, 38 patients were identified with brain metastases and a total of 243 lesions were treated with SRS, reported Zabi Wardak, MD, and his associates.
The median lesion size was 0.6 cm (0.2-3.1) and patients received a median SRS treatment dose of 18 Gy (12-24). The median age of patients was 65 years and the most common histology was clear cell RCC. Three quarters (74%) of patients received only one GammaKnife session, with a median number of two lesions treated at each session. The most common intracranial location for metastases was the frontal lobe (39% of lesions) followed by the parietal and temporal lobes (15% of lesions). The median size of treated metastases was 0.6 cm (0.2-3.1), reported Dr. Wardak, of the University of Texas UT Southwestern Medical Center, Dallas, and his associates.
At 1 year, local control rates were 91.8% (95% confidence interval, 85.7-95.4) and at 2 years, 86.1% (77.1-91.7). The median overall survival after a diagnosis of brain metastases was 13.8 months (95% CI, 8.1-28.0), with a 1-year survival of 57.5% (95% CI, 40.2-71.4). For patients with a single brain metastasis, 1-year overall survival was 56.3% (95% CI, 29.5-76.2) and for those with multiple lesions, 59.1% (95% CI, 36.1-76.2).
“Patients with multiple brain metastases (greater than five lesions) from RCC did not have a worse survival and should be considered for SRS to achieve intracranial tumor control while avoiding neurological decline,” wrote Dr. Wardak and his associates.
Coauthor James Brugarolas is supported by a grant from the National Institutes of Health. No other study funding was disclosed. The other authors have no disclosures.
SOURCE: Wardak Z et al. Clin Genitourin Cancer. 2018 Nov 20. doi: 10.1016/j.clgc.2018.11.006.
Stereotactic radiosurgery (SRS) is being used increasingly for brain metastases to prevent central nervous system (CNS) toxicity, and new findings published in Clinical Genitourinary Cancer demonstrate that it is a highly effective procedure for brain metastases from renal cell carcinoma (RCC).
In this study, the 1-year local control rate was more than 90% and durability of control was up to 2 years. SRS appears to be the most efficacious for smaller metastases, while escalated dosing or fractionation are needed to effectively treat larger lesions.
In metastatic RCC, the incidence of brain metastases is as high as 15%, and while surgical resection is associated with an improved survival compared with whole brain radiotherapy alone, for patients who are not candidates for resection, SRS is an alternative option. For this study, the authors sought to determine outcomes after treatment with SRS for brain metastases in 268 patients with metastatic RCC who were treated between 2006 and 2015 at a single institution.
Of this group, 38 patients were identified with brain metastases and a total of 243 lesions were treated with SRS, reported Zabi Wardak, MD, and his associates.
The median lesion size was 0.6 cm (0.2-3.1) and patients received a median SRS treatment dose of 18 Gy (12-24). The median age of patients was 65 years and the most common histology was clear cell RCC. Three quarters (74%) of patients received only one GammaKnife session, with a median number of two lesions treated at each session. The most common intracranial location for metastases was the frontal lobe (39% of lesions) followed by the parietal and temporal lobes (15% of lesions). The median size of treated metastases was 0.6 cm (0.2-3.1), reported Dr. Wardak, of the University of Texas UT Southwestern Medical Center, Dallas, and his associates.
At 1 year, local control rates were 91.8% (95% confidence interval, 85.7-95.4) and at 2 years, 86.1% (77.1-91.7). The median overall survival after a diagnosis of brain metastases was 13.8 months (95% CI, 8.1-28.0), with a 1-year survival of 57.5% (95% CI, 40.2-71.4). For patients with a single brain metastasis, 1-year overall survival was 56.3% (95% CI, 29.5-76.2) and for those with multiple lesions, 59.1% (95% CI, 36.1-76.2).
“Patients with multiple brain metastases (greater than five lesions) from RCC did not have a worse survival and should be considered for SRS to achieve intracranial tumor control while avoiding neurological decline,” wrote Dr. Wardak and his associates.
Coauthor James Brugarolas is supported by a grant from the National Institutes of Health. No other study funding was disclosed. The other authors have no disclosures.
SOURCE: Wardak Z et al. Clin Genitourin Cancer. 2018 Nov 20. doi: 10.1016/j.clgc.2018.11.006.
Stereotactic radiosurgery (SRS) is being used increasingly for brain metastases to prevent central nervous system (CNS) toxicity, and new findings published in Clinical Genitourinary Cancer demonstrate that it is a highly effective procedure for brain metastases from renal cell carcinoma (RCC).
In this study, the 1-year local control rate was more than 90% and durability of control was up to 2 years. SRS appears to be the most efficacious for smaller metastases, while escalated dosing or fractionation are needed to effectively treat larger lesions.
In metastatic RCC, the incidence of brain metastases is as high as 15%, and while surgical resection is associated with an improved survival compared with whole brain radiotherapy alone, for patients who are not candidates for resection, SRS is an alternative option. For this study, the authors sought to determine outcomes after treatment with SRS for brain metastases in 268 patients with metastatic RCC who were treated between 2006 and 2015 at a single institution.
Of this group, 38 patients were identified with brain metastases and a total of 243 lesions were treated with SRS, reported Zabi Wardak, MD, and his associates.
The median lesion size was 0.6 cm (0.2-3.1) and patients received a median SRS treatment dose of 18 Gy (12-24). The median age of patients was 65 years and the most common histology was clear cell RCC. Three quarters (74%) of patients received only one GammaKnife session, with a median number of two lesions treated at each session. The most common intracranial location for metastases was the frontal lobe (39% of lesions) followed by the parietal and temporal lobes (15% of lesions). The median size of treated metastases was 0.6 cm (0.2-3.1), reported Dr. Wardak, of the University of Texas UT Southwestern Medical Center, Dallas, and his associates.
At 1 year, local control rates were 91.8% (95% confidence interval, 85.7-95.4) and at 2 years, 86.1% (77.1-91.7). The median overall survival after a diagnosis of brain metastases was 13.8 months (95% CI, 8.1-28.0), with a 1-year survival of 57.5% (95% CI, 40.2-71.4). For patients with a single brain metastasis, 1-year overall survival was 56.3% (95% CI, 29.5-76.2) and for those with multiple lesions, 59.1% (95% CI, 36.1-76.2).
“Patients with multiple brain metastases (greater than five lesions) from RCC did not have a worse survival and should be considered for SRS to achieve intracranial tumor control while avoiding neurological decline,” wrote Dr. Wardak and his associates.
Coauthor James Brugarolas is supported by a grant from the National Institutes of Health. No other study funding was disclosed. The other authors have no disclosures.
SOURCE: Wardak Z et al. Clin Genitourin Cancer. 2018 Nov 20. doi: 10.1016/j.clgc.2018.11.006.
FROM CLINICAL GENITOURINARY CANCER
Key clinical point: SRS can effectively control brain metastases in patients with RCC, with more than half of treated patients surviving more than a year.
Major finding:. Treated local control rates at 1 and 2 years were 91.8% (95% CI, 85.7-95.4) and 86.1% (77.1-91.7)
Study details: A single-institution study that reviewed the use of SRS to treat brain lesions in 38 patients with a total of 243 brain metastases.
Disclosures: Coauthor James Brugarolas is supported by a grant from the National Institutes of Health. No other study funding was disclosed. The other authors have no disclosures.
Source: Wardak Z et al. Clin Genitourin Cancer. 2018 Nov 20. doi: 10.1016/j.clgc.2018.11.006.