User login
Hyperkalemia most common adverse event in women taking spironolactone
, according to new research.
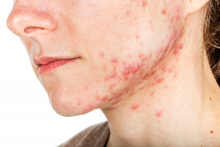
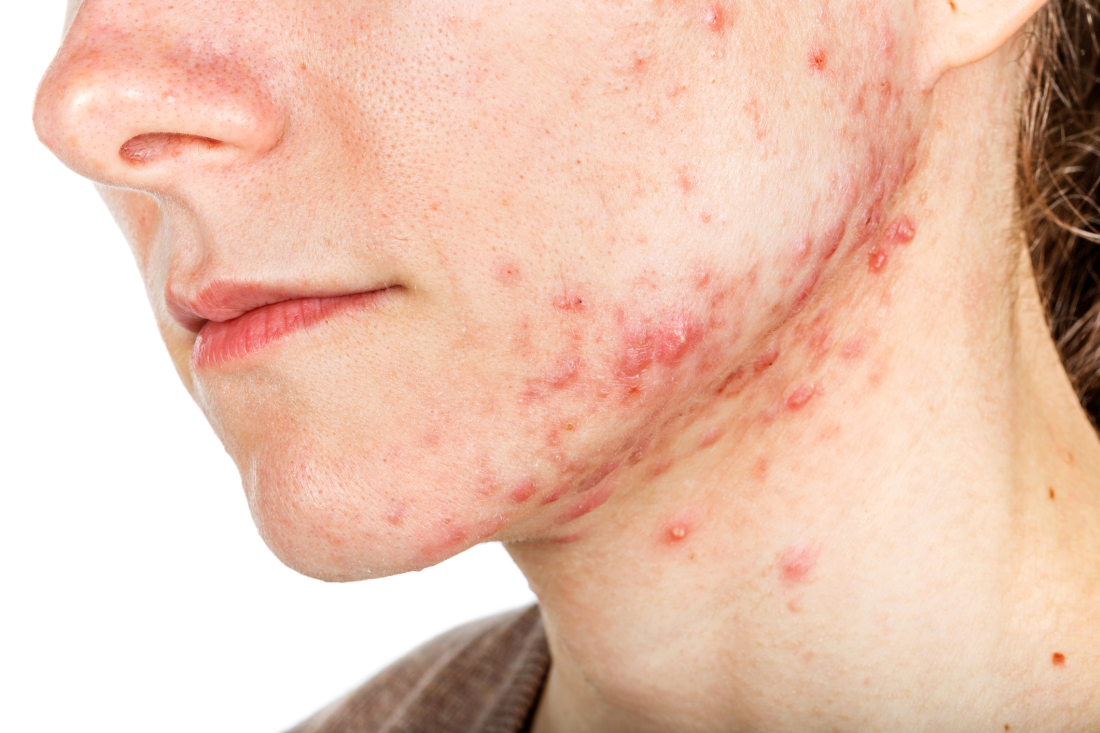
Spironolactone, which is approved to treat heart failure, hypertension, edema, and primary hyperaldosteronism, has antagonistic effects on progesterone and androgen receptors and has been used as an off-label treatment for acne in women. “Numerous guidelines have recommended its off-label use for acne therapy to avoid antibiotic resistance and potential side effects,” wrote Yu Wang of Stony Brook (N.Y.) University and Shari R. Lipner MD, PhD, of Weill Cornell Medicine, New York. Their report is in the International Journal of Women’s Dermatology.
In a retrospective study, the investigators analyzed 7,920 adverse events with spironolactone reported by women of all ages between Jan. 1, 1969, and Dec. 30, 2018, to the Food and Drug Administration’s Adverse Event Reporting System database, for all indications. The most common adverse event was hyperkalemia, reported in 16.1%, followed by kidney injury (15.2%) and drug interactions (9%). Of the 1,272 cases of hyperkalemia reported, 25 occurred in women aged 45 years or younger; 59.3% occurred in women aged 65-85 years.
While spironolactone prescribing information was not available, the investigators compared yearly reports of adverse events with annual public interest in spironolactone using the Google Trends search term spironolactone and annual scholarly mentions of spironolactone in the Altmetric database. There was a strong correlation between the number of cases reported to the FDA and the Google Trends search (Spearman coefficient, 0.94; P less than .001) and to the Altmetric database (Spearman coefficient, 0.64; P less than .01).
Noting that hyperkalemia is “exceptionally uncommon” in women aged 45 years and younger, the investigators concluded that “in the absence of risk factors for hyperkalemia or reduced renal function, potassium laboratory monitoring is unnecessary in younger females taking spironolactone.” Because the incidence increases with age, “interval laboratory monitoring is recommended for females older than 45 years old,” they noted.
Limitations of the study, they noted, include the retrospective design and no available data before 1969. “In addition, since the [FDA Adverse Event Reporting System] data does not differentiate whether spironolactone was prescribed for heart failure, hypertension, edema, primary hyperaldosteronism, or for acne,” the study could not control for these or other confounding comorbidities or associated therapies.
“For future studies, it is important to analyze drug interactions more carefully to determine which other medications may potentiate the risk for hyperkalemia in patients taking spironolactone. It is also important to quantitate overall U.S. prescription data to better understand the relative frequency of these adverse effects reported to the FDA,” they wrote.
The investigators reported that they had no conflicts of interest; the study had no funding.
SOURCE: Wang Y, Lipner SR. Int J Womens Dermatol. 2020 May 18. doi: 10.1016/j.ijwd.2020.05.002.
, according to new research.
Spironolactone, which is approved to treat heart failure, hypertension, edema, and primary hyperaldosteronism, has antagonistic effects on progesterone and androgen receptors and has been used as an off-label treatment for acne in women. “Numerous guidelines have recommended its off-label use for acne therapy to avoid antibiotic resistance and potential side effects,” wrote Yu Wang of Stony Brook (N.Y.) University and Shari R. Lipner MD, PhD, of Weill Cornell Medicine, New York. Their report is in the International Journal of Women’s Dermatology.
In a retrospective study, the investigators analyzed 7,920 adverse events with spironolactone reported by women of all ages between Jan. 1, 1969, and Dec. 30, 2018, to the Food and Drug Administration’s Adverse Event Reporting System database, for all indications. The most common adverse event was hyperkalemia, reported in 16.1%, followed by kidney injury (15.2%) and drug interactions (9%). Of the 1,272 cases of hyperkalemia reported, 25 occurred in women aged 45 years or younger; 59.3% occurred in women aged 65-85 years.
While spironolactone prescribing information was not available, the investigators compared yearly reports of adverse events with annual public interest in spironolactone using the Google Trends search term spironolactone and annual scholarly mentions of spironolactone in the Altmetric database. There was a strong correlation between the number of cases reported to the FDA and the Google Trends search (Spearman coefficient, 0.94; P less than .001) and to the Altmetric database (Spearman coefficient, 0.64; P less than .01).
Noting that hyperkalemia is “exceptionally uncommon” in women aged 45 years and younger, the investigators concluded that “in the absence of risk factors for hyperkalemia or reduced renal function, potassium laboratory monitoring is unnecessary in younger females taking spironolactone.” Because the incidence increases with age, “interval laboratory monitoring is recommended for females older than 45 years old,” they noted.
Limitations of the study, they noted, include the retrospective design and no available data before 1969. “In addition, since the [FDA Adverse Event Reporting System] data does not differentiate whether spironolactone was prescribed for heart failure, hypertension, edema, primary hyperaldosteronism, or for acne,” the study could not control for these or other confounding comorbidities or associated therapies.
“For future studies, it is important to analyze drug interactions more carefully to determine which other medications may potentiate the risk for hyperkalemia in patients taking spironolactone. It is also important to quantitate overall U.S. prescription data to better understand the relative frequency of these adverse effects reported to the FDA,” they wrote.
The investigators reported that they had no conflicts of interest; the study had no funding.
SOURCE: Wang Y, Lipner SR. Int J Womens Dermatol. 2020 May 18. doi: 10.1016/j.ijwd.2020.05.002.
, according to new research.
Spironolactone, which is approved to treat heart failure, hypertension, edema, and primary hyperaldosteronism, has antagonistic effects on progesterone and androgen receptors and has been used as an off-label treatment for acne in women. “Numerous guidelines have recommended its off-label use for acne therapy to avoid antibiotic resistance and potential side effects,” wrote Yu Wang of Stony Brook (N.Y.) University and Shari R. Lipner MD, PhD, of Weill Cornell Medicine, New York. Their report is in the International Journal of Women’s Dermatology.
In a retrospective study, the investigators analyzed 7,920 adverse events with spironolactone reported by women of all ages between Jan. 1, 1969, and Dec. 30, 2018, to the Food and Drug Administration’s Adverse Event Reporting System database, for all indications. The most common adverse event was hyperkalemia, reported in 16.1%, followed by kidney injury (15.2%) and drug interactions (9%). Of the 1,272 cases of hyperkalemia reported, 25 occurred in women aged 45 years or younger; 59.3% occurred in women aged 65-85 years.
While spironolactone prescribing information was not available, the investigators compared yearly reports of adverse events with annual public interest in spironolactone using the Google Trends search term spironolactone and annual scholarly mentions of spironolactone in the Altmetric database. There was a strong correlation between the number of cases reported to the FDA and the Google Trends search (Spearman coefficient, 0.94; P less than .001) and to the Altmetric database (Spearman coefficient, 0.64; P less than .01).
Noting that hyperkalemia is “exceptionally uncommon” in women aged 45 years and younger, the investigators concluded that “in the absence of risk factors for hyperkalemia or reduced renal function, potassium laboratory monitoring is unnecessary in younger females taking spironolactone.” Because the incidence increases with age, “interval laboratory monitoring is recommended for females older than 45 years old,” they noted.
Limitations of the study, they noted, include the retrospective design and no available data before 1969. “In addition, since the [FDA Adverse Event Reporting System] data does not differentiate whether spironolactone was prescribed for heart failure, hypertension, edema, primary hyperaldosteronism, or for acne,” the study could not control for these or other confounding comorbidities or associated therapies.
“For future studies, it is important to analyze drug interactions more carefully to determine which other medications may potentiate the risk for hyperkalemia in patients taking spironolactone. It is also important to quantitate overall U.S. prescription data to better understand the relative frequency of these adverse effects reported to the FDA,” they wrote.
The investigators reported that they had no conflicts of interest; the study had no funding.
SOURCE: Wang Y, Lipner SR. Int J Womens Dermatol. 2020 May 18. doi: 10.1016/j.ijwd.2020.05.002.
FROM THE INTERNATIONAL JOURNAL OF WOMEN’S DERMATOLOGY
Acne Keloidalis Nuchae in the Armed Forces
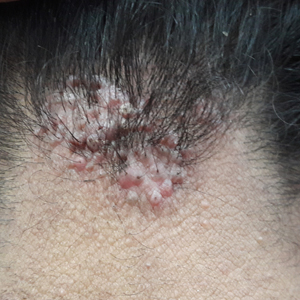
Acne keloidalis nuchae (AKN) is a chronic inflammatory disorder most commonly involving the occipital scalp and posterior neck characterized by the development of keloidlike papules, pustules, and plaques. If left untreated, this condition may progress to scarring alopecia. It primarily affects males of African descent, but it also may occur in females and in other ethnic groups. Although the exact underlying pathogenesis is unclear, close haircuts and chronic mechanical irritation to the posterior neck and scalp are known inciting factors. For this reason, AKN disproportionately affects active-duty military servicemembers who are held to strict grooming standards. The US Military maintains these grooming standards to ensure uniformity, self-discipline, and serviceability in operational settings.1 Regulations dictate short tapered hair, particularly on the back of the neck, which can require weekly to biweekly haircuts to maintain.1-5
First-line treatment of AKN is prevention by avoiding short haircuts and other forms of mechanical irritation.1,6,7 However, there are considerable barriers to this strategy within the military due to uniform regulations as well as personal appearance and grooming standards. Early identification and treatment are of utmost importance in managing AKN in the military population to ensure reduction of morbidity, prevention of late-stage disease, and continued fitness for duty. This article reviews the clinical features, epidemiology, and treatments available for management of AKN, with a special focus on the active-duty military population.
Clinical Features and Epidemiology
Acne keloidalis nuchae is a chronic inflammatory disorder characterized by the development of keloidlike papules, pustules, and plaques on the posterior neck and occipital scalp.6 Also known as folliculitis keloidalis nuchae, AKN is seen primarily in men of African descent, though cases also have been reported in females and in a few other ethnic groups.6,7 In black males, the AKN prevalence worldwide ranges from 0.5% to 13.6%. The male to female ratio is 20 to 1.7 Although the exact cause is unknown, AKN appears to develop from chronic irritation and inflammation following localized skin injury and/or trauma. Chronic irritation from close-shaved haircuts, tight-fitting shirt collars, caps, and helmets have all been implicated as considerable risk factors.6-8
Symptoms generally develop hours to days following a close haircut and begin with the early formation of inflamed irritated papules and notable erythema.6,7 These papules may become secondarily infected and develop into pustules and/or abscesses, especially in cases in which the affected individual continues to have the hair shaved. Continued use of shared razors increases the risk for secondary infection and also raises the concern for transmission of blood-borne pathogens, as AKN lesions are quick to bleed with minor trauma.7
Over time, chronic inflammation and continued trauma of the AKN papules leads to widespread fibrosis and scar formation, as the papules coalesce into larger plaques and nodules. If left untreated, these later stages of disease can progress to chronic scarring alopecia.6
Prevention
In the general population, first-line therapy of AKN is preventative. The goal is to break the cycle of chronic inflammation, thereby preventing the development of additional lesions and subsequent scarring.7 Patients should be encouraged to avoid frequent haircuts, close shaves, hats, helmets, and tight shirt collars.6-8
A 2017 cross-sectional study by Adotama et al9 investigated recognition and management of AKN in predominantly black barbershops in an urban setting. Fifty barbers from barbershops in Oklahoma City, Oklahoma, were enrolled and interviewed for the study. Of these barbers, only 44% (22/50) were able to properly identify AKN from a photograph. Although the vast majority (94% [47/50]) were aware that razor use would aggravate the condition, only 46% (23/50) reported avoidance of cutting hair for clients with active AKN.9 This study, while limited by its small sample size, showed that many barbers may be unaware of AKN and therefore unknowingly contribute to the disease process by performing haircuts on actively inflamed scalps. For this reason, it is important to educate patients about their condition and strongly recommend lifestyle and hairstyle modifications in the management of their disease.
Acne keloidalis nuchae that is severe enough to interfere with the proper use and wear of military equipment (eg, Kevlar helmets) or maintenance of regulation grooming standards does not meet military admission standards.10,11 However, mild undiagnosed cases may be overlooked during entrance physical examinations, while many servicemembers develop AKN after entering the military.10 For these individuals, long-term avoidance of haircuts is not a realistic or obtainable therapeutic option.
Treatment
Topical Therapy
Early mild to moderate cases of AKN—papules less than 3 mm, no nodules present—may be treated with potent topical steroids. Studies have shown 2-week alternating cycles of high-potency topical steroids (2 weeks of twice-daily application followed by 2 weeks without application) for 8 to 12 weeks to be effective in reducing AKN lesions.8,12 Topical clindamycin also may be added and has demonstrated efficacy particularly when pustules are present.7,8
Intralesional Steroids
For moderate cases of AKN—papules more than 3 mm, plaques, and nodules—intralesional steroid injections may be considered. Triamcinolone may be used at a dose of 5 to 40 mg/mL administered at 4-week intervals.7 More concentrated doses will produce faster responses but also carry the known risk of side effects such as hypopigmentation in darker-skinned individuals and skin atrophy.
Systemic Therapy
Systemic therapy with oral antibiotics may be warranted as an adjunct to mild to moderate cases of AKN or in cases with clear evidence of secondary infection. Long-term tetracycline antibiotics, such as minocycline and doxycycline, may be used concurrently with topical and/or intralesional steroids.6,7 Their antibacterial and anti-inflammatory effects are useful in controlling secondary infections and reducing overall chronic inflammation.
When selecting an appropriate antibiotic for long-term use in active-duty military patients, it is important to consider their effects on duty status. Doxycycline is preferred for active-duty servicemembers because it is not duty limiting or medically disqualifying.10,13-15 However, minocycline, is restricted for use in aviators and aircrew members due to the risk for central nervous system side effects, which may include light-headedness, dizziness, and vertigo.
UV Light Therapy
UV radiation has known anti-inflammatory, immunosuppressive, and antifibrotic effects and commonly is used in the treatment of many dermatologic conditions.16 Within the last decade, targeted UVB (tUVB) radiation has shown promise as an effective alternative therapy for AKN. In 2014, Okoye et al16 conducted a prospective, randomized, split-scalp study in 11 patients with AKN. Each patient underwent treatment with a tUVB device (with peaks at 303 and 313 nm) to a randomly selected side of the scalp 3 times weekly for 16 weeks. Significant reductions in lesion count were seen on the treated side after 8 (P=.03) and 16 weeks (P=.04), with no change noted on the control side. Aside from objective lesion counts, patients completed questionnaires (n=6) regarding their treatment outcomes. Notably, 83.3% (5/6) reported marked improvement in their condition. Aside from mild transient burning and erythema of the treated area, no serious side effects were reported.16
Targeted UVB phototherapy has limited utility in an operational setting due to accessibility and operational tempo. Phototherapy units typically are available only at commands in close proximity to large medical treatment facilities. Further, the vast majority of servicemembers have duty hours that are not amenable to multiple treatment sessions per week for several months. For servicemembers in administrative roles or serving in garrison or shore billets, tUVB or narrowband UV phototherapy may be viable treatment options.
Laser Therapy
Various lasers have been used to treat AKN, including the CO2 laser, pulsed dye laser, 810-nm diode laser, and 1064-nm Nd:YAG laser.6 Kantor et al17 utilized a CO2 laser with a focused beam for surgical excision of a late-stage AKN case as early as 1986. In these patients, it was demonstrated that focused CO2 laser could be used to remove fibrotic lesions in an outpatient setting with only local anesthesia. Although only 8 patients were treated in this report, no relapses occurred.17
CO2 laser evaporation using the unfocused beam setting with 130 to 150 J/cm2 has been less successful, with relapses reported in multiple cases.6 Dragoni et al18 attempted treatment with a 595-nm pulsed dye laser with 6.5-J/cm2 fluence and 0.5-millisecond pulse but faced similar results, with lesions returning within 1 month.
There have been numerous reports of clinical improvement of AKN with the use of the 1064-nm Nd:YAG laser.6,19 Esmat et al19 treated 16 patients with a fluence of 35 to 45 J/cm2 and pulse duration of 10 to 30 milliseconds adjusted to skin type and hair thickness. An overall 82% reduction in lesion count was observed after 5 treatment sessions. Biopsies following the treatment course demonstrated a significant reduction in papule and plaque count (P=.001 and P=.011, respectively), and no clinical recurrences were noted at 12 months posttreatment.19 Similarly, Woo et al20 conducted a single-blinded, randomized, controlled trial to assess the efficacy of the Nd:YAG laser in combination with topical corticosteroid therapy vs topical corticosteroid monotherapy. Of the 20 patients treated, there was a statistically significant improvement in patients with papule-only AKN who received the laser and topical combination treatment (P=.031).20
Laser therapy may be an available treatment option for military servicemembers stationed within close proximity to military treatment facilities, with the Nd:YAG laser typically having the widest availability. Although laser therapy may be effective in early stages of disease, servicemembers would have to be amenable to limitation of future hair growth in the treated areas.
Surgical Excision
Surgical excision may be considered for large, extensive, disfiguring, and/or refractory lesions. Excision is a safe and effective method to remove tender, inflamed, keloidlike masses. Techniques for excision include electrosurgical excision with secondary intention healing, excision of a horizontal ellipse involving the posterior hairline with either primary closure or secondary intention healing, and use of a semilunar tissue expander prior to excision and closure.6 Regardless of the technique, it is important to ensure that affected tissue is excised at a depth that includes the base of the hair follicles to prevent recurrence.21
Final Thoughts
Acne keloidalis nuchae is a chronic inflammatory disease that causes considerable morbidity and can lead to chronic infection, alopecia, and disfigurement of the occipital scalp and posterior neck. Although easily preventable through the avoidance of mechanical trauma, irritation, and frequent short haircuts, the active-duty military population is restricted in their preventive measures due to current grooming and uniform standards. In this population, early identification and treatment are necessary to manage the disease to reduce patient morbidity and ensure continued operational and medical readiness. Topical and intralesional steroids may be used in mild to moderate cases. Topical and/or systemic antibiotics may be added to the treatment regimen in cases of secondary bacterial infection. For more severe refractory cases, laser therapy or complete surgical excision may be warranted.
- Weiss AN, Arballo OM, Miletta NR, et al. Military grooming standards and their impact on skin diseases of the head and neck. Cutis. 2018;102:328, 331-333.
- US Department of the Army. Wear and Appearance of Army Uniforms and Insignia: Army Regulation 670-1. Washington, DC: Department of the Army; 2017. https://history.army.mil/html/forcestruc/docs/AR670-1.pdf. Accessed April 14, 2020.
- U.S. Headquarters Marine Corps. Marine Corps Uniform Regulations: Marine Corps Order 1020.34H. Quantico, VA: United States Marine Corps, 2018. https://www.marines.mil/portals/1/Publications/MCO%201020.34H%20v2.pdf?ver=2018-06-26-094038-137. Accessed April 14, 2020.
- Grooming standards. In: US Department of the Navy. United States Navy Uniform Regulations: NAVPERS 15665I. https://www.public.navy.mil/bupers-npc/support/uniforms/uniformregulations/chapter2/Pages/2201PersonalAppearance.aspx. Updated May 2019. Accessed April 14, 2020.
- Department of the Air Force. AFT 36-2903, Dress and Personal Appearance of Air Force Personnel. Washington, DC: Department of the Air Force, 2019. https://static.e-publishing.af.mil/production/1/af_a1/publication/afi36-2903/afi36-2903.pdf. Accessed April 14, 2020.
- Maranda EL, Simmons BJ, Nguyen AH, et al. Treatment of acne keloidalis nuchae: a systemic review of the literature. Dermatol Ther (Heidelb). 2016;6:362-378.
- Ogunbiyi A. Acne keloidalis nuchae: prevalence, impact, and management challenges. Clin Cosmet Investig Dermatol. 2016;9:483-489.
- Alexis A, Heath CR, Halder RM. Folliculitis keloidalis nuchae and pseudofolliculitis barbae: are prevention and effective treatment within reach? Dermatol Clin. 2014;32:183-191.
- Adotama P, Tinker D, Mitchell K, et al. Barber knowledge and recommendations regarding pseudofolliculitis barbae and acne keloidalis nuchae in an urban setting. JAMA Dermatol. 2017;12:1325.
- Burke KR, Larrymore DC, Cho S. Treatment considerations for US military members with sin disease. Cutis. 2019;6:329-332.
- Medical standards for Appointment, Enlistment, or Induction Into the Military Services (DoD Instruction 6130.03). Washington, DC: Department of Defense; May 6, 2018. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/613003p.pdf. Accessed April 27, 2020.
- Callender VD, Young CM, Haverstock CL, et al. An open label study of clobetasol propionate 0.05% and betamethasone valerate 0.12% foams in treatment of mild to moderate acne keloidalis. Cutis. 2005;75:317-321.
- US Department of the Army. Standards of medical fitness. https://www.qmo.amedd.army.mil/diabetes/AR40_5012011.pdf. Published December 14, 2007. Accessed April 27, 2020.
- US Department of the Air Force. Medical examinations and standards. https://static.e-publishing.af.mil/production/1/af_sg/publication/afi48-123/afi48-123.pdf. Published November 5, 2013. Accessed April 27, 2020.
- US Navy Aeromedical Reference and Waiver Guide. https://www.med.navy.mil/sites/nmotc/nami/arwg/Documents/WaiverGuide/Complete_Waiver_Guide.pdf. Published September 4, 2019. Accessed April 14, 2020.
- Okoye GA, Rainer BM, Leung SG, et al. Improving acne keloidalis nuchae with targeted ultraviolet B treatment: a prospective, randomized split-scalp study. Br J Dermatol. 2014;17:1156-1163.
- Kantor GR, Ratz JL, Wheeland RG. Treatment of acne keloidalis nuchae with carbon dioxide laser. J Am Acad Dermatol. 1986;14(2, pt 1):263-267.
- 18. Dragoni F, Bassi A, Cannarozzo G, et al. Successful treatment of acne keloidalis nuchae resistant to conventional therapy with 1064-nm Nd:YAG laser. G Ital Dermatol Venereol. 2013;148:231-232.
- Esmat SM, Hay RMA, Zeid OMA, et al. The efficacy of laser assisted hair removal in the treatment of acne keloidalis nuchae; a pilot study. Eur J Dermatol. 2012;22:645-650.
- Woo DK, Treyger G, Henderson M, et al. Prospective controlled trial for the treatment of acne keloidalis nuchae with a long-pulsed neodymium-doped yttrium-aluminum-garnet laser. J Cutan Med Surg. 2018;22:236-238.
- Beckett N, Lawson C, Cohen G. Electrosurgical excision of acne keloidalis nuchae with secondary intention healing. J Clin Aesthet Dermatol. 2011;4:36-39.
Acne keloidalis nuchae (AKN) is a chronic inflammatory disorder most commonly involving the occipital scalp and posterior neck characterized by the development of keloidlike papules, pustules, and plaques. If left untreated, this condition may progress to scarring alopecia. It primarily affects males of African descent, but it also may occur in females and in other ethnic groups. Although the exact underlying pathogenesis is unclear, close haircuts and chronic mechanical irritation to the posterior neck and scalp are known inciting factors. For this reason, AKN disproportionately affects active-duty military servicemembers who are held to strict grooming standards. The US Military maintains these grooming standards to ensure uniformity, self-discipline, and serviceability in operational settings.1 Regulations dictate short tapered hair, particularly on the back of the neck, which can require weekly to biweekly haircuts to maintain.1-5
First-line treatment of AKN is prevention by avoiding short haircuts and other forms of mechanical irritation.1,6,7 However, there are considerable barriers to this strategy within the military due to uniform regulations as well as personal appearance and grooming standards. Early identification and treatment are of utmost importance in managing AKN in the military population to ensure reduction of morbidity, prevention of late-stage disease, and continued fitness for duty. This article reviews the clinical features, epidemiology, and treatments available for management of AKN, with a special focus on the active-duty military population.
Clinical Features and Epidemiology
Acne keloidalis nuchae is a chronic inflammatory disorder characterized by the development of keloidlike papules, pustules, and plaques on the posterior neck and occipital scalp.6 Also known as folliculitis keloidalis nuchae, AKN is seen primarily in men of African descent, though cases also have been reported in females and in a few other ethnic groups.6,7 In black males, the AKN prevalence worldwide ranges from 0.5% to 13.6%. The male to female ratio is 20 to 1.7 Although the exact cause is unknown, AKN appears to develop from chronic irritation and inflammation following localized skin injury and/or trauma. Chronic irritation from close-shaved haircuts, tight-fitting shirt collars, caps, and helmets have all been implicated as considerable risk factors.6-8
Symptoms generally develop hours to days following a close haircut and begin with the early formation of inflamed irritated papules and notable erythema.6,7 These papules may become secondarily infected and develop into pustules and/or abscesses, especially in cases in which the affected individual continues to have the hair shaved. Continued use of shared razors increases the risk for secondary infection and also raises the concern for transmission of blood-borne pathogens, as AKN lesions are quick to bleed with minor trauma.7
Over time, chronic inflammation and continued trauma of the AKN papules leads to widespread fibrosis and scar formation, as the papules coalesce into larger plaques and nodules. If left untreated, these later stages of disease can progress to chronic scarring alopecia.6
Prevention
In the general population, first-line therapy of AKN is preventative. The goal is to break the cycle of chronic inflammation, thereby preventing the development of additional lesions and subsequent scarring.7 Patients should be encouraged to avoid frequent haircuts, close shaves, hats, helmets, and tight shirt collars.6-8
A 2017 cross-sectional study by Adotama et al9 investigated recognition and management of AKN in predominantly black barbershops in an urban setting. Fifty barbers from barbershops in Oklahoma City, Oklahoma, were enrolled and interviewed for the study. Of these barbers, only 44% (22/50) were able to properly identify AKN from a photograph. Although the vast majority (94% [47/50]) were aware that razor use would aggravate the condition, only 46% (23/50) reported avoidance of cutting hair for clients with active AKN.9 This study, while limited by its small sample size, showed that many barbers may be unaware of AKN and therefore unknowingly contribute to the disease process by performing haircuts on actively inflamed scalps. For this reason, it is important to educate patients about their condition and strongly recommend lifestyle and hairstyle modifications in the management of their disease.
Acne keloidalis nuchae that is severe enough to interfere with the proper use and wear of military equipment (eg, Kevlar helmets) or maintenance of regulation grooming standards does not meet military admission standards.10,11 However, mild undiagnosed cases may be overlooked during entrance physical examinations, while many servicemembers develop AKN after entering the military.10 For these individuals, long-term avoidance of haircuts is not a realistic or obtainable therapeutic option.
Treatment
Topical Therapy
Early mild to moderate cases of AKN—papules less than 3 mm, no nodules present—may be treated with potent topical steroids. Studies have shown 2-week alternating cycles of high-potency topical steroids (2 weeks of twice-daily application followed by 2 weeks without application) for 8 to 12 weeks to be effective in reducing AKN lesions.8,12 Topical clindamycin also may be added and has demonstrated efficacy particularly when pustules are present.7,8
Intralesional Steroids
For moderate cases of AKN—papules more than 3 mm, plaques, and nodules—intralesional steroid injections may be considered. Triamcinolone may be used at a dose of 5 to 40 mg/mL administered at 4-week intervals.7 More concentrated doses will produce faster responses but also carry the known risk of side effects such as hypopigmentation in darker-skinned individuals and skin atrophy.
Systemic Therapy
Systemic therapy with oral antibiotics may be warranted as an adjunct to mild to moderate cases of AKN or in cases with clear evidence of secondary infection. Long-term tetracycline antibiotics, such as minocycline and doxycycline, may be used concurrently with topical and/or intralesional steroids.6,7 Their antibacterial and anti-inflammatory effects are useful in controlling secondary infections and reducing overall chronic inflammation.
When selecting an appropriate antibiotic for long-term use in active-duty military patients, it is important to consider their effects on duty status. Doxycycline is preferred for active-duty servicemembers because it is not duty limiting or medically disqualifying.10,13-15 However, minocycline, is restricted for use in aviators and aircrew members due to the risk for central nervous system side effects, which may include light-headedness, dizziness, and vertigo.
UV Light Therapy
UV radiation has known anti-inflammatory, immunosuppressive, and antifibrotic effects and commonly is used in the treatment of many dermatologic conditions.16 Within the last decade, targeted UVB (tUVB) radiation has shown promise as an effective alternative therapy for AKN. In 2014, Okoye et al16 conducted a prospective, randomized, split-scalp study in 11 patients with AKN. Each patient underwent treatment with a tUVB device (with peaks at 303 and 313 nm) to a randomly selected side of the scalp 3 times weekly for 16 weeks. Significant reductions in lesion count were seen on the treated side after 8 (P=.03) and 16 weeks (P=.04), with no change noted on the control side. Aside from objective lesion counts, patients completed questionnaires (n=6) regarding their treatment outcomes. Notably, 83.3% (5/6) reported marked improvement in their condition. Aside from mild transient burning and erythema of the treated area, no serious side effects were reported.16
Targeted UVB phototherapy has limited utility in an operational setting due to accessibility and operational tempo. Phototherapy units typically are available only at commands in close proximity to large medical treatment facilities. Further, the vast majority of servicemembers have duty hours that are not amenable to multiple treatment sessions per week for several months. For servicemembers in administrative roles or serving in garrison or shore billets, tUVB or narrowband UV phototherapy may be viable treatment options.
Laser Therapy
Various lasers have been used to treat AKN, including the CO2 laser, pulsed dye laser, 810-nm diode laser, and 1064-nm Nd:YAG laser.6 Kantor et al17 utilized a CO2 laser with a focused beam for surgical excision of a late-stage AKN case as early as 1986. In these patients, it was demonstrated that focused CO2 laser could be used to remove fibrotic lesions in an outpatient setting with only local anesthesia. Although only 8 patients were treated in this report, no relapses occurred.17
CO2 laser evaporation using the unfocused beam setting with 130 to 150 J/cm2 has been less successful, with relapses reported in multiple cases.6 Dragoni et al18 attempted treatment with a 595-nm pulsed dye laser with 6.5-J/cm2 fluence and 0.5-millisecond pulse but faced similar results, with lesions returning within 1 month.
There have been numerous reports of clinical improvement of AKN with the use of the 1064-nm Nd:YAG laser.6,19 Esmat et al19 treated 16 patients with a fluence of 35 to 45 J/cm2 and pulse duration of 10 to 30 milliseconds adjusted to skin type and hair thickness. An overall 82% reduction in lesion count was observed after 5 treatment sessions. Biopsies following the treatment course demonstrated a significant reduction in papule and plaque count (P=.001 and P=.011, respectively), and no clinical recurrences were noted at 12 months posttreatment.19 Similarly, Woo et al20 conducted a single-blinded, randomized, controlled trial to assess the efficacy of the Nd:YAG laser in combination with topical corticosteroid therapy vs topical corticosteroid monotherapy. Of the 20 patients treated, there was a statistically significant improvement in patients with papule-only AKN who received the laser and topical combination treatment (P=.031).20
Laser therapy may be an available treatment option for military servicemembers stationed within close proximity to military treatment facilities, with the Nd:YAG laser typically having the widest availability. Although laser therapy may be effective in early stages of disease, servicemembers would have to be amenable to limitation of future hair growth in the treated areas.
Surgical Excision
Surgical excision may be considered for large, extensive, disfiguring, and/or refractory lesions. Excision is a safe and effective method to remove tender, inflamed, keloidlike masses. Techniques for excision include electrosurgical excision with secondary intention healing, excision of a horizontal ellipse involving the posterior hairline with either primary closure or secondary intention healing, and use of a semilunar tissue expander prior to excision and closure.6 Regardless of the technique, it is important to ensure that affected tissue is excised at a depth that includes the base of the hair follicles to prevent recurrence.21
Final Thoughts
Acne keloidalis nuchae is a chronic inflammatory disease that causes considerable morbidity and can lead to chronic infection, alopecia, and disfigurement of the occipital scalp and posterior neck. Although easily preventable through the avoidance of mechanical trauma, irritation, and frequent short haircuts, the active-duty military population is restricted in their preventive measures due to current grooming and uniform standards. In this population, early identification and treatment are necessary to manage the disease to reduce patient morbidity and ensure continued operational and medical readiness. Topical and intralesional steroids may be used in mild to moderate cases. Topical and/or systemic antibiotics may be added to the treatment regimen in cases of secondary bacterial infection. For more severe refractory cases, laser therapy or complete surgical excision may be warranted.
Acne keloidalis nuchae (AKN) is a chronic inflammatory disorder most commonly involving the occipital scalp and posterior neck characterized by the development of keloidlike papules, pustules, and plaques. If left untreated, this condition may progress to scarring alopecia. It primarily affects males of African descent, but it also may occur in females and in other ethnic groups. Although the exact underlying pathogenesis is unclear, close haircuts and chronic mechanical irritation to the posterior neck and scalp are known inciting factors. For this reason, AKN disproportionately affects active-duty military servicemembers who are held to strict grooming standards. The US Military maintains these grooming standards to ensure uniformity, self-discipline, and serviceability in operational settings.1 Regulations dictate short tapered hair, particularly on the back of the neck, which can require weekly to biweekly haircuts to maintain.1-5
First-line treatment of AKN is prevention by avoiding short haircuts and other forms of mechanical irritation.1,6,7 However, there are considerable barriers to this strategy within the military due to uniform regulations as well as personal appearance and grooming standards. Early identification and treatment are of utmost importance in managing AKN in the military population to ensure reduction of morbidity, prevention of late-stage disease, and continued fitness for duty. This article reviews the clinical features, epidemiology, and treatments available for management of AKN, with a special focus on the active-duty military population.
Clinical Features and Epidemiology
Acne keloidalis nuchae is a chronic inflammatory disorder characterized by the development of keloidlike papules, pustules, and plaques on the posterior neck and occipital scalp.6 Also known as folliculitis keloidalis nuchae, AKN is seen primarily in men of African descent, though cases also have been reported in females and in a few other ethnic groups.6,7 In black males, the AKN prevalence worldwide ranges from 0.5% to 13.6%. The male to female ratio is 20 to 1.7 Although the exact cause is unknown, AKN appears to develop from chronic irritation and inflammation following localized skin injury and/or trauma. Chronic irritation from close-shaved haircuts, tight-fitting shirt collars, caps, and helmets have all been implicated as considerable risk factors.6-8
Symptoms generally develop hours to days following a close haircut and begin with the early formation of inflamed irritated papules and notable erythema.6,7 These papules may become secondarily infected and develop into pustules and/or abscesses, especially in cases in which the affected individual continues to have the hair shaved. Continued use of shared razors increases the risk for secondary infection and also raises the concern for transmission of blood-borne pathogens, as AKN lesions are quick to bleed with minor trauma.7
Over time, chronic inflammation and continued trauma of the AKN papules leads to widespread fibrosis and scar formation, as the papules coalesce into larger plaques and nodules. If left untreated, these later stages of disease can progress to chronic scarring alopecia.6
Prevention
In the general population, first-line therapy of AKN is preventative. The goal is to break the cycle of chronic inflammation, thereby preventing the development of additional lesions and subsequent scarring.7 Patients should be encouraged to avoid frequent haircuts, close shaves, hats, helmets, and tight shirt collars.6-8
A 2017 cross-sectional study by Adotama et al9 investigated recognition and management of AKN in predominantly black barbershops in an urban setting. Fifty barbers from barbershops in Oklahoma City, Oklahoma, were enrolled and interviewed for the study. Of these barbers, only 44% (22/50) were able to properly identify AKN from a photograph. Although the vast majority (94% [47/50]) were aware that razor use would aggravate the condition, only 46% (23/50) reported avoidance of cutting hair for clients with active AKN.9 This study, while limited by its small sample size, showed that many barbers may be unaware of AKN and therefore unknowingly contribute to the disease process by performing haircuts on actively inflamed scalps. For this reason, it is important to educate patients about their condition and strongly recommend lifestyle and hairstyle modifications in the management of their disease.
Acne keloidalis nuchae that is severe enough to interfere with the proper use and wear of military equipment (eg, Kevlar helmets) or maintenance of regulation grooming standards does not meet military admission standards.10,11 However, mild undiagnosed cases may be overlooked during entrance physical examinations, while many servicemembers develop AKN after entering the military.10 For these individuals, long-term avoidance of haircuts is not a realistic or obtainable therapeutic option.
Treatment
Topical Therapy
Early mild to moderate cases of AKN—papules less than 3 mm, no nodules present—may be treated with potent topical steroids. Studies have shown 2-week alternating cycles of high-potency topical steroids (2 weeks of twice-daily application followed by 2 weeks without application) for 8 to 12 weeks to be effective in reducing AKN lesions.8,12 Topical clindamycin also may be added and has demonstrated efficacy particularly when pustules are present.7,8
Intralesional Steroids
For moderate cases of AKN—papules more than 3 mm, plaques, and nodules—intralesional steroid injections may be considered. Triamcinolone may be used at a dose of 5 to 40 mg/mL administered at 4-week intervals.7 More concentrated doses will produce faster responses but also carry the known risk of side effects such as hypopigmentation in darker-skinned individuals and skin atrophy.
Systemic Therapy
Systemic therapy with oral antibiotics may be warranted as an adjunct to mild to moderate cases of AKN or in cases with clear evidence of secondary infection. Long-term tetracycline antibiotics, such as minocycline and doxycycline, may be used concurrently with topical and/or intralesional steroids.6,7 Their antibacterial and anti-inflammatory effects are useful in controlling secondary infections and reducing overall chronic inflammation.
When selecting an appropriate antibiotic for long-term use in active-duty military patients, it is important to consider their effects on duty status. Doxycycline is preferred for active-duty servicemembers because it is not duty limiting or medically disqualifying.10,13-15 However, minocycline, is restricted for use in aviators and aircrew members due to the risk for central nervous system side effects, which may include light-headedness, dizziness, and vertigo.
UV Light Therapy
UV radiation has known anti-inflammatory, immunosuppressive, and antifibrotic effects and commonly is used in the treatment of many dermatologic conditions.16 Within the last decade, targeted UVB (tUVB) radiation has shown promise as an effective alternative therapy for AKN. In 2014, Okoye et al16 conducted a prospective, randomized, split-scalp study in 11 patients with AKN. Each patient underwent treatment with a tUVB device (with peaks at 303 and 313 nm) to a randomly selected side of the scalp 3 times weekly for 16 weeks. Significant reductions in lesion count were seen on the treated side after 8 (P=.03) and 16 weeks (P=.04), with no change noted on the control side. Aside from objective lesion counts, patients completed questionnaires (n=6) regarding their treatment outcomes. Notably, 83.3% (5/6) reported marked improvement in their condition. Aside from mild transient burning and erythema of the treated area, no serious side effects were reported.16
Targeted UVB phototherapy has limited utility in an operational setting due to accessibility and operational tempo. Phototherapy units typically are available only at commands in close proximity to large medical treatment facilities. Further, the vast majority of servicemembers have duty hours that are not amenable to multiple treatment sessions per week for several months. For servicemembers in administrative roles or serving in garrison or shore billets, tUVB or narrowband UV phototherapy may be viable treatment options.
Laser Therapy
Various lasers have been used to treat AKN, including the CO2 laser, pulsed dye laser, 810-nm diode laser, and 1064-nm Nd:YAG laser.6 Kantor et al17 utilized a CO2 laser with a focused beam for surgical excision of a late-stage AKN case as early as 1986. In these patients, it was demonstrated that focused CO2 laser could be used to remove fibrotic lesions in an outpatient setting with only local anesthesia. Although only 8 patients were treated in this report, no relapses occurred.17
CO2 laser evaporation using the unfocused beam setting with 130 to 150 J/cm2 has been less successful, with relapses reported in multiple cases.6 Dragoni et al18 attempted treatment with a 595-nm pulsed dye laser with 6.5-J/cm2 fluence and 0.5-millisecond pulse but faced similar results, with lesions returning within 1 month.
There have been numerous reports of clinical improvement of AKN with the use of the 1064-nm Nd:YAG laser.6,19 Esmat et al19 treated 16 patients with a fluence of 35 to 45 J/cm2 and pulse duration of 10 to 30 milliseconds adjusted to skin type and hair thickness. An overall 82% reduction in lesion count was observed after 5 treatment sessions. Biopsies following the treatment course demonstrated a significant reduction in papule and plaque count (P=.001 and P=.011, respectively), and no clinical recurrences were noted at 12 months posttreatment.19 Similarly, Woo et al20 conducted a single-blinded, randomized, controlled trial to assess the efficacy of the Nd:YAG laser in combination with topical corticosteroid therapy vs topical corticosteroid monotherapy. Of the 20 patients treated, there was a statistically significant improvement in patients with papule-only AKN who received the laser and topical combination treatment (P=.031).20
Laser therapy may be an available treatment option for military servicemembers stationed within close proximity to military treatment facilities, with the Nd:YAG laser typically having the widest availability. Although laser therapy may be effective in early stages of disease, servicemembers would have to be amenable to limitation of future hair growth in the treated areas.
Surgical Excision
Surgical excision may be considered for large, extensive, disfiguring, and/or refractory lesions. Excision is a safe and effective method to remove tender, inflamed, keloidlike masses. Techniques for excision include electrosurgical excision with secondary intention healing, excision of a horizontal ellipse involving the posterior hairline with either primary closure or secondary intention healing, and use of a semilunar tissue expander prior to excision and closure.6 Regardless of the technique, it is important to ensure that affected tissue is excised at a depth that includes the base of the hair follicles to prevent recurrence.21
Final Thoughts
Acne keloidalis nuchae is a chronic inflammatory disease that causes considerable morbidity and can lead to chronic infection, alopecia, and disfigurement of the occipital scalp and posterior neck. Although easily preventable through the avoidance of mechanical trauma, irritation, and frequent short haircuts, the active-duty military population is restricted in their preventive measures due to current grooming and uniform standards. In this population, early identification and treatment are necessary to manage the disease to reduce patient morbidity and ensure continued operational and medical readiness. Topical and intralesional steroids may be used in mild to moderate cases. Topical and/or systemic antibiotics may be added to the treatment regimen in cases of secondary bacterial infection. For more severe refractory cases, laser therapy or complete surgical excision may be warranted.
- Weiss AN, Arballo OM, Miletta NR, et al. Military grooming standards and their impact on skin diseases of the head and neck. Cutis. 2018;102:328, 331-333.
- US Department of the Army. Wear and Appearance of Army Uniforms and Insignia: Army Regulation 670-1. Washington, DC: Department of the Army; 2017. https://history.army.mil/html/forcestruc/docs/AR670-1.pdf. Accessed April 14, 2020.
- U.S. Headquarters Marine Corps. Marine Corps Uniform Regulations: Marine Corps Order 1020.34H. Quantico, VA: United States Marine Corps, 2018. https://www.marines.mil/portals/1/Publications/MCO%201020.34H%20v2.pdf?ver=2018-06-26-094038-137. Accessed April 14, 2020.
- Grooming standards. In: US Department of the Navy. United States Navy Uniform Regulations: NAVPERS 15665I. https://www.public.navy.mil/bupers-npc/support/uniforms/uniformregulations/chapter2/Pages/2201PersonalAppearance.aspx. Updated May 2019. Accessed April 14, 2020.
- Department of the Air Force. AFT 36-2903, Dress and Personal Appearance of Air Force Personnel. Washington, DC: Department of the Air Force, 2019. https://static.e-publishing.af.mil/production/1/af_a1/publication/afi36-2903/afi36-2903.pdf. Accessed April 14, 2020.
- Maranda EL, Simmons BJ, Nguyen AH, et al. Treatment of acne keloidalis nuchae: a systemic review of the literature. Dermatol Ther (Heidelb). 2016;6:362-378.
- Ogunbiyi A. Acne keloidalis nuchae: prevalence, impact, and management challenges. Clin Cosmet Investig Dermatol. 2016;9:483-489.
- Alexis A, Heath CR, Halder RM. Folliculitis keloidalis nuchae and pseudofolliculitis barbae: are prevention and effective treatment within reach? Dermatol Clin. 2014;32:183-191.
- Adotama P, Tinker D, Mitchell K, et al. Barber knowledge and recommendations regarding pseudofolliculitis barbae and acne keloidalis nuchae in an urban setting. JAMA Dermatol. 2017;12:1325.
- Burke KR, Larrymore DC, Cho S. Treatment considerations for US military members with sin disease. Cutis. 2019;6:329-332.
- Medical standards for Appointment, Enlistment, or Induction Into the Military Services (DoD Instruction 6130.03). Washington, DC: Department of Defense; May 6, 2018. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/613003p.pdf. Accessed April 27, 2020.
- Callender VD, Young CM, Haverstock CL, et al. An open label study of clobetasol propionate 0.05% and betamethasone valerate 0.12% foams in treatment of mild to moderate acne keloidalis. Cutis. 2005;75:317-321.
- US Department of the Army. Standards of medical fitness. https://www.qmo.amedd.army.mil/diabetes/AR40_5012011.pdf. Published December 14, 2007. Accessed April 27, 2020.
- US Department of the Air Force. Medical examinations and standards. https://static.e-publishing.af.mil/production/1/af_sg/publication/afi48-123/afi48-123.pdf. Published November 5, 2013. Accessed April 27, 2020.
- US Navy Aeromedical Reference and Waiver Guide. https://www.med.navy.mil/sites/nmotc/nami/arwg/Documents/WaiverGuide/Complete_Waiver_Guide.pdf. Published September 4, 2019. Accessed April 14, 2020.
- Okoye GA, Rainer BM, Leung SG, et al. Improving acne keloidalis nuchae with targeted ultraviolet B treatment: a prospective, randomized split-scalp study. Br J Dermatol. 2014;17:1156-1163.
- Kantor GR, Ratz JL, Wheeland RG. Treatment of acne keloidalis nuchae with carbon dioxide laser. J Am Acad Dermatol. 1986;14(2, pt 1):263-267.
- 18. Dragoni F, Bassi A, Cannarozzo G, et al. Successful treatment of acne keloidalis nuchae resistant to conventional therapy with 1064-nm Nd:YAG laser. G Ital Dermatol Venereol. 2013;148:231-232.
- Esmat SM, Hay RMA, Zeid OMA, et al. The efficacy of laser assisted hair removal in the treatment of acne keloidalis nuchae; a pilot study. Eur J Dermatol. 2012;22:645-650.
- Woo DK, Treyger G, Henderson M, et al. Prospective controlled trial for the treatment of acne keloidalis nuchae with a long-pulsed neodymium-doped yttrium-aluminum-garnet laser. J Cutan Med Surg. 2018;22:236-238.
- Beckett N, Lawson C, Cohen G. Electrosurgical excision of acne keloidalis nuchae with secondary intention healing. J Clin Aesthet Dermatol. 2011;4:36-39.
- Weiss AN, Arballo OM, Miletta NR, et al. Military grooming standards and their impact on skin diseases of the head and neck. Cutis. 2018;102:328, 331-333.
- US Department of the Army. Wear and Appearance of Army Uniforms and Insignia: Army Regulation 670-1. Washington, DC: Department of the Army; 2017. https://history.army.mil/html/forcestruc/docs/AR670-1.pdf. Accessed April 14, 2020.
- U.S. Headquarters Marine Corps. Marine Corps Uniform Regulations: Marine Corps Order 1020.34H. Quantico, VA: United States Marine Corps, 2018. https://www.marines.mil/portals/1/Publications/MCO%201020.34H%20v2.pdf?ver=2018-06-26-094038-137. Accessed April 14, 2020.
- Grooming standards. In: US Department of the Navy. United States Navy Uniform Regulations: NAVPERS 15665I. https://www.public.navy.mil/bupers-npc/support/uniforms/uniformregulations/chapter2/Pages/2201PersonalAppearance.aspx. Updated May 2019. Accessed April 14, 2020.
- Department of the Air Force. AFT 36-2903, Dress and Personal Appearance of Air Force Personnel. Washington, DC: Department of the Air Force, 2019. https://static.e-publishing.af.mil/production/1/af_a1/publication/afi36-2903/afi36-2903.pdf. Accessed April 14, 2020.
- Maranda EL, Simmons BJ, Nguyen AH, et al. Treatment of acne keloidalis nuchae: a systemic review of the literature. Dermatol Ther (Heidelb). 2016;6:362-378.
- Ogunbiyi A. Acne keloidalis nuchae: prevalence, impact, and management challenges. Clin Cosmet Investig Dermatol. 2016;9:483-489.
- Alexis A, Heath CR, Halder RM. Folliculitis keloidalis nuchae and pseudofolliculitis barbae: are prevention and effective treatment within reach? Dermatol Clin. 2014;32:183-191.
- Adotama P, Tinker D, Mitchell K, et al. Barber knowledge and recommendations regarding pseudofolliculitis barbae and acne keloidalis nuchae in an urban setting. JAMA Dermatol. 2017;12:1325.
- Burke KR, Larrymore DC, Cho S. Treatment considerations for US military members with sin disease. Cutis. 2019;6:329-332.
- Medical standards for Appointment, Enlistment, or Induction Into the Military Services (DoD Instruction 6130.03). Washington, DC: Department of Defense; May 6, 2018. https://www.esd.whs.mil/Portals/54/Documents/DD/issuances/dodi/613003p.pdf. Accessed April 27, 2020.
- Callender VD, Young CM, Haverstock CL, et al. An open label study of clobetasol propionate 0.05% and betamethasone valerate 0.12% foams in treatment of mild to moderate acne keloidalis. Cutis. 2005;75:317-321.
- US Department of the Army. Standards of medical fitness. https://www.qmo.amedd.army.mil/diabetes/AR40_5012011.pdf. Published December 14, 2007. Accessed April 27, 2020.
- US Department of the Air Force. Medical examinations and standards. https://static.e-publishing.af.mil/production/1/af_sg/publication/afi48-123/afi48-123.pdf. Published November 5, 2013. Accessed April 27, 2020.
- US Navy Aeromedical Reference and Waiver Guide. https://www.med.navy.mil/sites/nmotc/nami/arwg/Documents/WaiverGuide/Complete_Waiver_Guide.pdf. Published September 4, 2019. Accessed April 14, 2020.
- Okoye GA, Rainer BM, Leung SG, et al. Improving acne keloidalis nuchae with targeted ultraviolet B treatment: a prospective, randomized split-scalp study. Br J Dermatol. 2014;17:1156-1163.
- Kantor GR, Ratz JL, Wheeland RG. Treatment of acne keloidalis nuchae with carbon dioxide laser. J Am Acad Dermatol. 1986;14(2, pt 1):263-267.
- 18. Dragoni F, Bassi A, Cannarozzo G, et al. Successful treatment of acne keloidalis nuchae resistant to conventional therapy with 1064-nm Nd:YAG laser. G Ital Dermatol Venereol. 2013;148:231-232.
- Esmat SM, Hay RMA, Zeid OMA, et al. The efficacy of laser assisted hair removal in the treatment of acne keloidalis nuchae; a pilot study. Eur J Dermatol. 2012;22:645-650.
- Woo DK, Treyger G, Henderson M, et al. Prospective controlled trial for the treatment of acne keloidalis nuchae with a long-pulsed neodymium-doped yttrium-aluminum-garnet laser. J Cutan Med Surg. 2018;22:236-238.
- Beckett N, Lawson C, Cohen G. Electrosurgical excision of acne keloidalis nuchae with secondary intention healing. J Clin Aesthet Dermatol. 2011;4:36-39.
Practice Points
- Acne keloidalis nuchae (AKN) is a chronic inflammatory disorder of the occipital scalp and posterior neck characterized by keloidlike papules, pustules, and plaques that develop following mechanical irritation.
- Military members are required to maintain short haircuts and may be disproportionately affected by AKN.
- In the military population, early identification and treatment, which includes topical steroids, oral antibiotics, UV light therapy, lasers, and surgical excision, can prevent further scarring, permanent hair loss, and disfigurement from AKN.
Hydrogen peroxide reduces C. acnes cultures following shoulder surgery
Prior to shoulder surgery, application of 3% hydrogen peroxide is a simple and inexpensive strategy to reduce the risk of postoperative cultures of Cutibacterium acnes, according to findings from a prospective randomized trial. The results were reported in an abstract scheduled for release at the annual meeting of the American Academy of Orthopaedic Surgeons. The meeting was canceled because of COVID-19.
“This approach is simple, cheap, and does not rely on patient compliance,” explained Surena Namdari, MD, associate professor of orthopedic surgery at Thomas Jefferson University, Philadelphia.
C. acnes, formerly known as Propionibacterium acnes, is increasingly seen as an important target for prevention of postoperative shoulder infections because of published reports that it is the most commonly isolated bacterium from such infections, Dr. Namdari said in an interview.
In the prospective, randomized trial, male patients scheduled for shoulder arthroscopy were recruited if they did not have active acne, history of psoriatic or eczematous lesions, or recent antibiotic use. Most of the preoperative preparation of the surgical site was the same in the experimental and control arms. This included hair clipping, application of 2% chlorhexidine, and cleansing with saturated 7.5% povidone-iodine solution surgical scrub brushes.
The difference was that 3% hydrogen peroxide–soaked gauzes were applied to perioperative skin of those randomized to the experimental group but not to controls. All patients received routine preoperative oral antibiotics as well as perioperative applications of a formulation containing 2% chlorhexidine gluconate and 70% isopropyl alcohol.
Following surgery, 11 (18.6%) of the 59 patients in the experimental arm versus 23 (34.8%) of the 66 patients randomized to the control group had positive cultures for C. acnes (P = .047), according to the trial results, which have now been published (J Shoulder Elbow Surg. 2020;29:212-6).
There were no cases of skin reactions in either the experimental or control groups.
Topical skin cleansers that contain peroxide, such as benzoyl peroxide, have been shown to have a C. acnes decolonizing effect if applied repeatedly in the days prior to surgery, but Dr. Namdari suggested the problem with this approach is that it depends on patient compliance. A prophylaxis included in the preoperative routine eliminates this potential problem.
C. acnes is an anaerobic bacterium that is part of the resident flora of the skin around several joints, including the knee and the hip, but it is particularly common in the posterior shoulder. Colonization has been found substantially more common in men than in women, according to Dr. Namdari.
The specific threat posed by C. acnes to risk of postoperative infections “is still being defined,” and this trial was not large enough to associate the reduction in postoperative C. acnes cultures with a reduced risk of an adverse clinical outcome, but Dr. Namdari says that the data do show that the nearly 50% reduction in positive cultures was achieved efficiently and inexpensively with no apparent risk.
Several previous studies have also evaluated strategies for reducing C. acnes skin burden on the basis of expected protection against postoperative infection. In one, which associated a 3-day preoperative course of benzoyl peroxide with a reduction in the skin burden of C. acnes, the authors also concluded that this approach deserves consideration in routine skin preparation for shoulder arthroplasty (J Shoulder Elbow Surg. 2018;27:1539-44).
“We believe that a preoperative skin prep protocol that reduces C. acnes load on the skin would likely lead to reduced postoperative infections,” reported the senior author, Mohit N. Gilotra, MD, assistant professor, University of Maryland, Baltimore. Contacted about the rationale for reducing C. acnes skin burden without objective evidence of an impact on postoperative infection risk, Dr. Gilotra indicated these strategies make sense.
“It seems to be true for staph infections and is a reasonable assumption to make here,” he added. “Future work will help determine how much benzoyl peroxide, hydrogen peroxide, or other skin prep can reduce surgical site infection.”
Dr. Namdari reports financial relationships with multiple device and pharmaceutical companies but none relevant to this study.
SOURCE: Namdari S et al. AAOS 2020. Abstract P0808.
Prior to shoulder surgery, application of 3% hydrogen peroxide is a simple and inexpensive strategy to reduce the risk of postoperative cultures of Cutibacterium acnes, according to findings from a prospective randomized trial. The results were reported in an abstract scheduled for release at the annual meeting of the American Academy of Orthopaedic Surgeons. The meeting was canceled because of COVID-19.
“This approach is simple, cheap, and does not rely on patient compliance,” explained Surena Namdari, MD, associate professor of orthopedic surgery at Thomas Jefferson University, Philadelphia.
C. acnes, formerly known as Propionibacterium acnes, is increasingly seen as an important target for prevention of postoperative shoulder infections because of published reports that it is the most commonly isolated bacterium from such infections, Dr. Namdari said in an interview.
In the prospective, randomized trial, male patients scheduled for shoulder arthroscopy were recruited if they did not have active acne, history of psoriatic or eczematous lesions, or recent antibiotic use. Most of the preoperative preparation of the surgical site was the same in the experimental and control arms. This included hair clipping, application of 2% chlorhexidine, and cleansing with saturated 7.5% povidone-iodine solution surgical scrub brushes.
The difference was that 3% hydrogen peroxide–soaked gauzes were applied to perioperative skin of those randomized to the experimental group but not to controls. All patients received routine preoperative oral antibiotics as well as perioperative applications of a formulation containing 2% chlorhexidine gluconate and 70% isopropyl alcohol.
Following surgery, 11 (18.6%) of the 59 patients in the experimental arm versus 23 (34.8%) of the 66 patients randomized to the control group had positive cultures for C. acnes (P = .047), according to the trial results, which have now been published (J Shoulder Elbow Surg. 2020;29:212-6).
There were no cases of skin reactions in either the experimental or control groups.
Topical skin cleansers that contain peroxide, such as benzoyl peroxide, have been shown to have a C. acnes decolonizing effect if applied repeatedly in the days prior to surgery, but Dr. Namdari suggested the problem with this approach is that it depends on patient compliance. A prophylaxis included in the preoperative routine eliminates this potential problem.
C. acnes is an anaerobic bacterium that is part of the resident flora of the skin around several joints, including the knee and the hip, but it is particularly common in the posterior shoulder. Colonization has been found substantially more common in men than in women, according to Dr. Namdari.
The specific threat posed by C. acnes to risk of postoperative infections “is still being defined,” and this trial was not large enough to associate the reduction in postoperative C. acnes cultures with a reduced risk of an adverse clinical outcome, but Dr. Namdari says that the data do show that the nearly 50% reduction in positive cultures was achieved efficiently and inexpensively with no apparent risk.
Several previous studies have also evaluated strategies for reducing C. acnes skin burden on the basis of expected protection against postoperative infection. In one, which associated a 3-day preoperative course of benzoyl peroxide with a reduction in the skin burden of C. acnes, the authors also concluded that this approach deserves consideration in routine skin preparation for shoulder arthroplasty (J Shoulder Elbow Surg. 2018;27:1539-44).
“We believe that a preoperative skin prep protocol that reduces C. acnes load on the skin would likely lead to reduced postoperative infections,” reported the senior author, Mohit N. Gilotra, MD, assistant professor, University of Maryland, Baltimore. Contacted about the rationale for reducing C. acnes skin burden without objective evidence of an impact on postoperative infection risk, Dr. Gilotra indicated these strategies make sense.
“It seems to be true for staph infections and is a reasonable assumption to make here,” he added. “Future work will help determine how much benzoyl peroxide, hydrogen peroxide, or other skin prep can reduce surgical site infection.”
Dr. Namdari reports financial relationships with multiple device and pharmaceutical companies but none relevant to this study.
SOURCE: Namdari S et al. AAOS 2020. Abstract P0808.
Prior to shoulder surgery, application of 3% hydrogen peroxide is a simple and inexpensive strategy to reduce the risk of postoperative cultures of Cutibacterium acnes, according to findings from a prospective randomized trial. The results were reported in an abstract scheduled for release at the annual meeting of the American Academy of Orthopaedic Surgeons. The meeting was canceled because of COVID-19.
“This approach is simple, cheap, and does not rely on patient compliance,” explained Surena Namdari, MD, associate professor of orthopedic surgery at Thomas Jefferson University, Philadelphia.
C. acnes, formerly known as Propionibacterium acnes, is increasingly seen as an important target for prevention of postoperative shoulder infections because of published reports that it is the most commonly isolated bacterium from such infections, Dr. Namdari said in an interview.
In the prospective, randomized trial, male patients scheduled for shoulder arthroscopy were recruited if they did not have active acne, history of psoriatic or eczematous lesions, or recent antibiotic use. Most of the preoperative preparation of the surgical site was the same in the experimental and control arms. This included hair clipping, application of 2% chlorhexidine, and cleansing with saturated 7.5% povidone-iodine solution surgical scrub brushes.
The difference was that 3% hydrogen peroxide–soaked gauzes were applied to perioperative skin of those randomized to the experimental group but not to controls. All patients received routine preoperative oral antibiotics as well as perioperative applications of a formulation containing 2% chlorhexidine gluconate and 70% isopropyl alcohol.
Following surgery, 11 (18.6%) of the 59 patients in the experimental arm versus 23 (34.8%) of the 66 patients randomized to the control group had positive cultures for C. acnes (P = .047), according to the trial results, which have now been published (J Shoulder Elbow Surg. 2020;29:212-6).
There were no cases of skin reactions in either the experimental or control groups.
Topical skin cleansers that contain peroxide, such as benzoyl peroxide, have been shown to have a C. acnes decolonizing effect if applied repeatedly in the days prior to surgery, but Dr. Namdari suggested the problem with this approach is that it depends on patient compliance. A prophylaxis included in the preoperative routine eliminates this potential problem.
C. acnes is an anaerobic bacterium that is part of the resident flora of the skin around several joints, including the knee and the hip, but it is particularly common in the posterior shoulder. Colonization has been found substantially more common in men than in women, according to Dr. Namdari.
The specific threat posed by C. acnes to risk of postoperative infections “is still being defined,” and this trial was not large enough to associate the reduction in postoperative C. acnes cultures with a reduced risk of an adverse clinical outcome, but Dr. Namdari says that the data do show that the nearly 50% reduction in positive cultures was achieved efficiently and inexpensively with no apparent risk.
Several previous studies have also evaluated strategies for reducing C. acnes skin burden on the basis of expected protection against postoperative infection. In one, which associated a 3-day preoperative course of benzoyl peroxide with a reduction in the skin burden of C. acnes, the authors also concluded that this approach deserves consideration in routine skin preparation for shoulder arthroplasty (J Shoulder Elbow Surg. 2018;27:1539-44).
“We believe that a preoperative skin prep protocol that reduces C. acnes load on the skin would likely lead to reduced postoperative infections,” reported the senior author, Mohit N. Gilotra, MD, assistant professor, University of Maryland, Baltimore. Contacted about the rationale for reducing C. acnes skin burden without objective evidence of an impact on postoperative infection risk, Dr. Gilotra indicated these strategies make sense.
“It seems to be true for staph infections and is a reasonable assumption to make here,” he added. “Future work will help determine how much benzoyl peroxide, hydrogen peroxide, or other skin prep can reduce surgical site infection.”
Dr. Namdari reports financial relationships with multiple device and pharmaceutical companies but none relevant to this study.
SOURCE: Namdari S et al. AAOS 2020. Abstract P0808.
FROM AAOS 2020
Evidence on spironolactone safety, COVID-19 reassuring for acne patients
according to a report in the Journal of the American Academy of Dermatology.
The virus needs androgens to infect cells, and uses androgen-dependent transmembrane protease serine 2 to prime viral protein spikes to anchor onto ACE2 receptors. Without that step, the virus can’t enter cells. Androgens are the only known activator in humans, so androgen blockers like spironolactone probably short-circuit the process, said the report’s lead author Carlos Wambier, MD, PhD, of the department of dermatology at Brown University, Providence, R.I (J Am Acad Dermatol. 2020 Apr 10. doi: 10.1016/j.jaad.2020.04.032).
The lack of androgens could be a possible explanation as to why mortality is so rare among children with COVID-19, and why fatalities among men are higher than among women with COVID-19, he said in an interview.
There are a lot of androgen blocker candidates, but he said spironolactone – a mainstay of acne treatment – might well be the best for the pandemic because of its concomitant lung and heart benefits.
The message counters a post on Instagram in March from a New York City dermatologist in private practice, Ellen Marmur, MD, that raised a question about spironolactone. Concerned about the situation in New York, she reviewed the literature and found a 2005 study that reported that macrophages drawn from 10 heart failure patients had increased ACE2 activity and increased messenger RNA expression after the subjects had been on spironolactone 25 mg per day for a month.
In an interview, she said she has been sharing her concerns with patients on spironolactone and offering them an alternative, such as minocycline, until this issue is better elucidated. To date, she has had one young patient who declined to switch to another treatment, and about six patients who were comfortable switching to another treatment for 1-2 months. She said that she is “clearly cautious yet uncertain about the influence of chronic spironolactone for acne on COVID infection in acne patients,” and that eventually she would be interested in seeing retrospective data on outcomes of patients on spironolactone for hypertension versus acne during the pandemic.
Dr. Marmur’s post was spread on social media and was picked up by a few online news outlets.
In an interview, Adam Friedman, MD, professor and interim chair of dermatology at George Washington University, Washington, said he’s been addressing concerns about spironolactone in educational webinars because of it.
He tells his audience that “you can’t make any claims” for COVID-19 based on the 2005 study. It was a small clinical study in heart failure patients and only assessed ACE2 expression on macrophages, not respiratory, cardiac, or mesangial cells, which are the relevant locations for viral invasion and damage. In fact, there are studies showing that spironolactone reduced ACE2 in renal mesangial cells. Also of note, spironolactone has been used with no indication of virus risk since the 1950s, he pointed out. The American Academy of Dermatology has not said to stop spironolactone.
At least one study is underway to see if spironolactone is beneficial: 100 mg twice a day for 5 days is being pitted against placebo in Turkey among people hospitalized with acute respiratory distress. The study will evaluate the effect of spironolactone on oxygenation.
“There’s no evidence to show spironolactone can increase mortality levels,” Dr. Wambier said. He is using it more now in patients with acne – a sign of androgen hyperactivity – convinced that it will protect against COVID-19. He even started his sister on it to help with androgenic hair loss, and maybe the virus.
Observations in Spain – increased prevalence of androgenic alopecia among hospitalized patients – support the androgen link; 29 of 41 men (71%) hospitalized with bilateral pneumonia had male pattern baldness, which was severe in 16 (39%), according to a recent report (J Cosmet Dermatol. 2020 Apr 16. doi: 10.1111/jocd.13443). The expected prevalence in a similar age-matched population is 31%-53%.
“Based on the scientific rationale combined with this preliminary observation, we believe investigating the potential association between androgens and COVID‐19 disease severity warrants further merit,” concluded the authors, who included Dr. Wambier, and other dermatologists from the United States, as well as Spain, Australia, Croatia, and Switzerland. “If such an association is confirmed, antiandrogens could be evaluated as a potential treatment for COVID‐19 infection,” they wrote.
The numbers are holding up in a larger series from three Spanish hospitals, and also showing a greater prevalence of androgenic hair loss among hospitalized women, Dr. Wambier said in the interview.
Authors of the two studies include an employee of Applied Biology. No conflicts were declared in the Journal of Cosmetic Dermatology study; no disclosures were listed in the JAAD study. Dr. Friedman had no disclosures.
according to a report in the Journal of the American Academy of Dermatology.
The virus needs androgens to infect cells, and uses androgen-dependent transmembrane protease serine 2 to prime viral protein spikes to anchor onto ACE2 receptors. Without that step, the virus can’t enter cells. Androgens are the only known activator in humans, so androgen blockers like spironolactone probably short-circuit the process, said the report’s lead author Carlos Wambier, MD, PhD, of the department of dermatology at Brown University, Providence, R.I (J Am Acad Dermatol. 2020 Apr 10. doi: 10.1016/j.jaad.2020.04.032).
The lack of androgens could be a possible explanation as to why mortality is so rare among children with COVID-19, and why fatalities among men are higher than among women with COVID-19, he said in an interview.
There are a lot of androgen blocker candidates, but he said spironolactone – a mainstay of acne treatment – might well be the best for the pandemic because of its concomitant lung and heart benefits.
The message counters a post on Instagram in March from a New York City dermatologist in private practice, Ellen Marmur, MD, that raised a question about spironolactone. Concerned about the situation in New York, she reviewed the literature and found a 2005 study that reported that macrophages drawn from 10 heart failure patients had increased ACE2 activity and increased messenger RNA expression after the subjects had been on spironolactone 25 mg per day for a month.
In an interview, she said she has been sharing her concerns with patients on spironolactone and offering them an alternative, such as minocycline, until this issue is better elucidated. To date, she has had one young patient who declined to switch to another treatment, and about six patients who were comfortable switching to another treatment for 1-2 months. She said that she is “clearly cautious yet uncertain about the influence of chronic spironolactone for acne on COVID infection in acne patients,” and that eventually she would be interested in seeing retrospective data on outcomes of patients on spironolactone for hypertension versus acne during the pandemic.
Dr. Marmur’s post was spread on social media and was picked up by a few online news outlets.
In an interview, Adam Friedman, MD, professor and interim chair of dermatology at George Washington University, Washington, said he’s been addressing concerns about spironolactone in educational webinars because of it.
He tells his audience that “you can’t make any claims” for COVID-19 based on the 2005 study. It was a small clinical study in heart failure patients and only assessed ACE2 expression on macrophages, not respiratory, cardiac, or mesangial cells, which are the relevant locations for viral invasion and damage. In fact, there are studies showing that spironolactone reduced ACE2 in renal mesangial cells. Also of note, spironolactone has been used with no indication of virus risk since the 1950s, he pointed out. The American Academy of Dermatology has not said to stop spironolactone.
At least one study is underway to see if spironolactone is beneficial: 100 mg twice a day for 5 days is being pitted against placebo in Turkey among people hospitalized with acute respiratory distress. The study will evaluate the effect of spironolactone on oxygenation.
“There’s no evidence to show spironolactone can increase mortality levels,” Dr. Wambier said. He is using it more now in patients with acne – a sign of androgen hyperactivity – convinced that it will protect against COVID-19. He even started his sister on it to help with androgenic hair loss, and maybe the virus.
Observations in Spain – increased prevalence of androgenic alopecia among hospitalized patients – support the androgen link; 29 of 41 men (71%) hospitalized with bilateral pneumonia had male pattern baldness, which was severe in 16 (39%), according to a recent report (J Cosmet Dermatol. 2020 Apr 16. doi: 10.1111/jocd.13443). The expected prevalence in a similar age-matched population is 31%-53%.
“Based on the scientific rationale combined with this preliminary observation, we believe investigating the potential association between androgens and COVID‐19 disease severity warrants further merit,” concluded the authors, who included Dr. Wambier, and other dermatologists from the United States, as well as Spain, Australia, Croatia, and Switzerland. “If such an association is confirmed, antiandrogens could be evaluated as a potential treatment for COVID‐19 infection,” they wrote.
The numbers are holding up in a larger series from three Spanish hospitals, and also showing a greater prevalence of androgenic hair loss among hospitalized women, Dr. Wambier said in the interview.
Authors of the two studies include an employee of Applied Biology. No conflicts were declared in the Journal of Cosmetic Dermatology study; no disclosures were listed in the JAAD study. Dr. Friedman had no disclosures.
according to a report in the Journal of the American Academy of Dermatology.
The virus needs androgens to infect cells, and uses androgen-dependent transmembrane protease serine 2 to prime viral protein spikes to anchor onto ACE2 receptors. Without that step, the virus can’t enter cells. Androgens are the only known activator in humans, so androgen blockers like spironolactone probably short-circuit the process, said the report’s lead author Carlos Wambier, MD, PhD, of the department of dermatology at Brown University, Providence, R.I (J Am Acad Dermatol. 2020 Apr 10. doi: 10.1016/j.jaad.2020.04.032).
The lack of androgens could be a possible explanation as to why mortality is so rare among children with COVID-19, and why fatalities among men are higher than among women with COVID-19, he said in an interview.
There are a lot of androgen blocker candidates, but he said spironolactone – a mainstay of acne treatment – might well be the best for the pandemic because of its concomitant lung and heart benefits.
The message counters a post on Instagram in March from a New York City dermatologist in private practice, Ellen Marmur, MD, that raised a question about spironolactone. Concerned about the situation in New York, she reviewed the literature and found a 2005 study that reported that macrophages drawn from 10 heart failure patients had increased ACE2 activity and increased messenger RNA expression after the subjects had been on spironolactone 25 mg per day for a month.
In an interview, she said she has been sharing her concerns with patients on spironolactone and offering them an alternative, such as minocycline, until this issue is better elucidated. To date, she has had one young patient who declined to switch to another treatment, and about six patients who were comfortable switching to another treatment for 1-2 months. She said that she is “clearly cautious yet uncertain about the influence of chronic spironolactone for acne on COVID infection in acne patients,” and that eventually she would be interested in seeing retrospective data on outcomes of patients on spironolactone for hypertension versus acne during the pandemic.
Dr. Marmur’s post was spread on social media and was picked up by a few online news outlets.
In an interview, Adam Friedman, MD, professor and interim chair of dermatology at George Washington University, Washington, said he’s been addressing concerns about spironolactone in educational webinars because of it.
He tells his audience that “you can’t make any claims” for COVID-19 based on the 2005 study. It was a small clinical study in heart failure patients and only assessed ACE2 expression on macrophages, not respiratory, cardiac, or mesangial cells, which are the relevant locations for viral invasion and damage. In fact, there are studies showing that spironolactone reduced ACE2 in renal mesangial cells. Also of note, spironolactone has been used with no indication of virus risk since the 1950s, he pointed out. The American Academy of Dermatology has not said to stop spironolactone.
At least one study is underway to see if spironolactone is beneficial: 100 mg twice a day for 5 days is being pitted against placebo in Turkey among people hospitalized with acute respiratory distress. The study will evaluate the effect of spironolactone on oxygenation.
“There’s no evidence to show spironolactone can increase mortality levels,” Dr. Wambier said. He is using it more now in patients with acne – a sign of androgen hyperactivity – convinced that it will protect against COVID-19. He even started his sister on it to help with androgenic hair loss, and maybe the virus.
Observations in Spain – increased prevalence of androgenic alopecia among hospitalized patients – support the androgen link; 29 of 41 men (71%) hospitalized with bilateral pneumonia had male pattern baldness, which was severe in 16 (39%), according to a recent report (J Cosmet Dermatol. 2020 Apr 16. doi: 10.1111/jocd.13443). The expected prevalence in a similar age-matched population is 31%-53%.
“Based on the scientific rationale combined with this preliminary observation, we believe investigating the potential association between androgens and COVID‐19 disease severity warrants further merit,” concluded the authors, who included Dr. Wambier, and other dermatologists from the United States, as well as Spain, Australia, Croatia, and Switzerland. “If such an association is confirmed, antiandrogens could be evaluated as a potential treatment for COVID‐19 infection,” they wrote.
The numbers are holding up in a larger series from three Spanish hospitals, and also showing a greater prevalence of androgenic hair loss among hospitalized women, Dr. Wambier said in the interview.
Authors of the two studies include an employee of Applied Biology. No conflicts were declared in the Journal of Cosmetic Dermatology study; no disclosures were listed in the JAAD study. Dr. Friedman had no disclosures.
Novel acne drug now under review at the FDA
LAHAINA, HAWAII – by the Food and Drug Administration, is already generating considerable buzz in the patient-advocacy community even though the agency won’t issue its decision until August.
“I’ve actually had a lot of interest in this already from parents, especially regarding girls who have very hormonal acne but the parents are really not interested in starting them on a systemic hormonal therapy at their age,” Jessica Sprague, MD, said at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Clascoterone targets androgen receptors in the skin in order to reduce cutaneous 5-alpha dihydrotestosterone.
“It’s being developed for use in both males and females, which is great because at this point there’s no hormonal treatment for males,” noted Dr. Sprague, a pediatric dermatologist at Rady Children’s Hospital and the University of California, both in San Diego.
The manufacturer’s application for marketing approval of clascoterone cream 1% under FDA review includes evidence from two identical phase-3, double-blind, vehicle-controlled, 12-week, randomized trials. The two studies included a total of 1,440 patients aged 9 years through adulthood with moderate to severe facial acne vulgaris who were randomized to twice-daily application of clascoterone or its vehicle.
The primary outcome was the reduction in inflammatory lesions at week 12: a 46.2% decline from baseline with clascoterone 1% cream, which was a significantly greater improvement than the 32.7% reduction for vehicle. The secondary outcome – change in noninflammatory lesion counts at week 12 – was also positive for the topical androgen receptor inhibitor, which achieved a 29.8% reduction, compared with 18.9% for vehicle. Clascoterone exhibited a favorable safety and tolerability profile, with numerically fewer treatment-emergent adverse events than in the vehicle control group. A stronger formulation of the topical agent is in advanced clinical trials for the treatment of androgenetic alopecia in both males and females.
Dr. Sprague reported having no financial conflicts regarding her presentation.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – by the Food and Drug Administration, is already generating considerable buzz in the patient-advocacy community even though the agency won’t issue its decision until August.
“I’ve actually had a lot of interest in this already from parents, especially regarding girls who have very hormonal acne but the parents are really not interested in starting them on a systemic hormonal therapy at their age,” Jessica Sprague, MD, said at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Clascoterone targets androgen receptors in the skin in order to reduce cutaneous 5-alpha dihydrotestosterone.
“It’s being developed for use in both males and females, which is great because at this point there’s no hormonal treatment for males,” noted Dr. Sprague, a pediatric dermatologist at Rady Children’s Hospital and the University of California, both in San Diego.
The manufacturer’s application for marketing approval of clascoterone cream 1% under FDA review includes evidence from two identical phase-3, double-blind, vehicle-controlled, 12-week, randomized trials. The two studies included a total of 1,440 patients aged 9 years through adulthood with moderate to severe facial acne vulgaris who were randomized to twice-daily application of clascoterone or its vehicle.
The primary outcome was the reduction in inflammatory lesions at week 12: a 46.2% decline from baseline with clascoterone 1% cream, which was a significantly greater improvement than the 32.7% reduction for vehicle. The secondary outcome – change in noninflammatory lesion counts at week 12 – was also positive for the topical androgen receptor inhibitor, which achieved a 29.8% reduction, compared with 18.9% for vehicle. Clascoterone exhibited a favorable safety and tolerability profile, with numerically fewer treatment-emergent adverse events than in the vehicle control group. A stronger formulation of the topical agent is in advanced clinical trials for the treatment of androgenetic alopecia in both males and females.
Dr. Sprague reported having no financial conflicts regarding her presentation.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – by the Food and Drug Administration, is already generating considerable buzz in the patient-advocacy community even though the agency won’t issue its decision until August.
“I’ve actually had a lot of interest in this already from parents, especially regarding girls who have very hormonal acne but the parents are really not interested in starting them on a systemic hormonal therapy at their age,” Jessica Sprague, MD, said at the SDEF Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Clascoterone targets androgen receptors in the skin in order to reduce cutaneous 5-alpha dihydrotestosterone.
“It’s being developed for use in both males and females, which is great because at this point there’s no hormonal treatment for males,” noted Dr. Sprague, a pediatric dermatologist at Rady Children’s Hospital and the University of California, both in San Diego.
The manufacturer’s application for marketing approval of clascoterone cream 1% under FDA review includes evidence from two identical phase-3, double-blind, vehicle-controlled, 12-week, randomized trials. The two studies included a total of 1,440 patients aged 9 years through adulthood with moderate to severe facial acne vulgaris who were randomized to twice-daily application of clascoterone or its vehicle.
The primary outcome was the reduction in inflammatory lesions at week 12: a 46.2% decline from baseline with clascoterone 1% cream, which was a significantly greater improvement than the 32.7% reduction for vehicle. The secondary outcome – change in noninflammatory lesion counts at week 12 – was also positive for the topical androgen receptor inhibitor, which achieved a 29.8% reduction, compared with 18.9% for vehicle. Clascoterone exhibited a favorable safety and tolerability profile, with numerically fewer treatment-emergent adverse events than in the vehicle control group. A stronger formulation of the topical agent is in advanced clinical trials for the treatment of androgenetic alopecia in both males and females.
Dr. Sprague reported having no financial conflicts regarding her presentation.
The SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
REPORTING FROM THE SDEF HAWAII DERMATOLOGY SEMINAR
iPLEDGE allows at-home pregnancy tests during pandemic
tests to comply with the requirements of the iPLEDGE program during the COVID-19 pandemic, according to an update program posted on the iPLEDGE website.
The program’s other requirements – the prescription window and two forms of birth control – remain unchanged.
The change follows recent guidance from the Department of Health & Human Services and the Food and Drug Administration regarding accommodations for medical care and drugs subject to Risk Evaluation and Mitigation Strategies (REMS) in the midst of a public health emergency that requires most people to remain in their homes except for essential services.
Allowing females to take at-home pregnancy tests and communicate the results to physician according to their preference is “a game changer for the middle of a pandemic, obviously,” Neil Goldberg, MD, a dermatologist in Westchester County, New York, said in an interview. “These are patients who don’t need to spend time outside just to get pregnancy tests done. It makes it a lot easier.”
Dr. Goldberg is frustrated, however, that the accommodations have not been more widely publicized; he discovered the change incidentally when speaking to an iPLEDGE program representative to request a waiver for a patient who had taken her pregnancy test too early. The program had denied a similar request for a 15-year-old patient of his the previous week, despite the patient being abstinent and having been in shelter-in-place for several weeks.
“The size of your notice [on the website] should be proportionate to how important it is,” Dr. Goldberg said, and the small red box on the site is easy to miss. By contrast, asking anyone to leave their homes to go to a lab for a pregnancy test in the midst of a global pandemic so they can continue their medication would be putting patients at risk, he added.
The iPLEDGE program is designed in part to ensure unplanned pregnancies do not occur in females while taking the teratogenic acne drug. But the rules are onerous and difficult even during normal times, pointed out Hilary Baldwin, MD, medical director of the Acne Treatment and Research Center in New York City and past president of the American Acne and Rosacea Society.
Male patients taking isotretinoin must visit their physician every month to get a new no-refills prescription, but females must get a pregnancy test at a Clinical Laboratory Improvement Amendments–certified lab, which must then provide physical results to the prescribing physician. The doctor enters the negative pregnancy test and the two forms of birth control the patient is taking in the iPLEDGE program site.
Then the patient must take an online test at home to acknowledge they understand what it means to not get pregnant and enter the two forms of birth control they are using – which must match what the doctor enters – before the pharmacy can dispense the drug. The entire process must occur within 7 days or else the patient has to wait 19 days before starting the process over.
“We run a very tight schedule for girls. And every month, we would worry that something would interfere, a snow storm or something else, and that they wouldn’t be able to complete their objectives within the 7-day period,” Dr Baldwin said in an interview. “It was always difficult, and now with us not being able to see the patient and the patient not wanting to go to the lab, this became completely impossible.”
Until this change, some patients may not have been able to get their prescription for severe nodulocystic acne, which can cause physical and psychological scarring, and “postponing treatment increases the likelihood of scarring,” Dr. Baldwin pointed out.

Dr. Goldberg’s patients now take a pregnancy test at home and send him a photo of the negative test that he then inserts into their EMR.
According to a March 17 statement from HHS, potential penalties for HIPAA violations are waived for good-faith use of “everyday communication technologies,” such as Skype or FaceTime, for telehealth treatment or diagnostics. The change was intended to allow telehealth services to continue healthcare for practices that had not previously had secure telehealth technology established.
Despite the changes for at-home pregnancy tests for females and in-person visits for all patients, the program has not altered the 7-day prescription window or the requirement to have two forms of birth control.
With reports of a global condom shortage, Dr Baldwin said she has more concerns about her adult patients being able to find a required barrier method of birth control than about her adolescent patients.
“This is a unique opportunity for us to trust our teenage patients because they can’t leave the house,” Dr. Baldwin said. “I’m actually more worried about my adult women on the drug who are bored and cooped up in a house with their significant other.”
Dr. Baldwin and Dr. Goldberg had no relevant disclosures. Dr. Goldberg is a Dermatology News board member.
tests to comply with the requirements of the iPLEDGE program during the COVID-19 pandemic, according to an update program posted on the iPLEDGE website.
The program’s other requirements – the prescription window and two forms of birth control – remain unchanged.
The change follows recent guidance from the Department of Health & Human Services and the Food and Drug Administration regarding accommodations for medical care and drugs subject to Risk Evaluation and Mitigation Strategies (REMS) in the midst of a public health emergency that requires most people to remain in their homes except for essential services.
Allowing females to take at-home pregnancy tests and communicate the results to physician according to their preference is “a game changer for the middle of a pandemic, obviously,” Neil Goldberg, MD, a dermatologist in Westchester County, New York, said in an interview. “These are patients who don’t need to spend time outside just to get pregnancy tests done. It makes it a lot easier.”
Dr. Goldberg is frustrated, however, that the accommodations have not been more widely publicized; he discovered the change incidentally when speaking to an iPLEDGE program representative to request a waiver for a patient who had taken her pregnancy test too early. The program had denied a similar request for a 15-year-old patient of his the previous week, despite the patient being abstinent and having been in shelter-in-place for several weeks.
“The size of your notice [on the website] should be proportionate to how important it is,” Dr. Goldberg said, and the small red box on the site is easy to miss. By contrast, asking anyone to leave their homes to go to a lab for a pregnancy test in the midst of a global pandemic so they can continue their medication would be putting patients at risk, he added.
The iPLEDGE program is designed in part to ensure unplanned pregnancies do not occur in females while taking the teratogenic acne drug. But the rules are onerous and difficult even during normal times, pointed out Hilary Baldwin, MD, medical director of the Acne Treatment and Research Center in New York City and past president of the American Acne and Rosacea Society.
Male patients taking isotretinoin must visit their physician every month to get a new no-refills prescription, but females must get a pregnancy test at a Clinical Laboratory Improvement Amendments–certified lab, which must then provide physical results to the prescribing physician. The doctor enters the negative pregnancy test and the two forms of birth control the patient is taking in the iPLEDGE program site.
Then the patient must take an online test at home to acknowledge they understand what it means to not get pregnant and enter the two forms of birth control they are using – which must match what the doctor enters – before the pharmacy can dispense the drug. The entire process must occur within 7 days or else the patient has to wait 19 days before starting the process over.
“We run a very tight schedule for girls. And every month, we would worry that something would interfere, a snow storm or something else, and that they wouldn’t be able to complete their objectives within the 7-day period,” Dr Baldwin said in an interview. “It was always difficult, and now with us not being able to see the patient and the patient not wanting to go to the lab, this became completely impossible.”
Until this change, some patients may not have been able to get their prescription for severe nodulocystic acne, which can cause physical and psychological scarring, and “postponing treatment increases the likelihood of scarring,” Dr. Baldwin pointed out.

Dr. Goldberg’s patients now take a pregnancy test at home and send him a photo of the negative test that he then inserts into their EMR.
According to a March 17 statement from HHS, potential penalties for HIPAA violations are waived for good-faith use of “everyday communication technologies,” such as Skype or FaceTime, for telehealth treatment or diagnostics. The change was intended to allow telehealth services to continue healthcare for practices that had not previously had secure telehealth technology established.
Despite the changes for at-home pregnancy tests for females and in-person visits for all patients, the program has not altered the 7-day prescription window or the requirement to have two forms of birth control.
With reports of a global condom shortage, Dr Baldwin said she has more concerns about her adult patients being able to find a required barrier method of birth control than about her adolescent patients.
“This is a unique opportunity for us to trust our teenage patients because they can’t leave the house,” Dr. Baldwin said. “I’m actually more worried about my adult women on the drug who are bored and cooped up in a house with their significant other.”
Dr. Baldwin and Dr. Goldberg had no relevant disclosures. Dr. Goldberg is a Dermatology News board member.
tests to comply with the requirements of the iPLEDGE program during the COVID-19 pandemic, according to an update program posted on the iPLEDGE website.
The program’s other requirements – the prescription window and two forms of birth control – remain unchanged.
The change follows recent guidance from the Department of Health & Human Services and the Food and Drug Administration regarding accommodations for medical care and drugs subject to Risk Evaluation and Mitigation Strategies (REMS) in the midst of a public health emergency that requires most people to remain in their homes except for essential services.
Allowing females to take at-home pregnancy tests and communicate the results to physician according to their preference is “a game changer for the middle of a pandemic, obviously,” Neil Goldberg, MD, a dermatologist in Westchester County, New York, said in an interview. “These are patients who don’t need to spend time outside just to get pregnancy tests done. It makes it a lot easier.”
Dr. Goldberg is frustrated, however, that the accommodations have not been more widely publicized; he discovered the change incidentally when speaking to an iPLEDGE program representative to request a waiver for a patient who had taken her pregnancy test too early. The program had denied a similar request for a 15-year-old patient of his the previous week, despite the patient being abstinent and having been in shelter-in-place for several weeks.
“The size of your notice [on the website] should be proportionate to how important it is,” Dr. Goldberg said, and the small red box on the site is easy to miss. By contrast, asking anyone to leave their homes to go to a lab for a pregnancy test in the midst of a global pandemic so they can continue their medication would be putting patients at risk, he added.
The iPLEDGE program is designed in part to ensure unplanned pregnancies do not occur in females while taking the teratogenic acne drug. But the rules are onerous and difficult even during normal times, pointed out Hilary Baldwin, MD, medical director of the Acne Treatment and Research Center in New York City and past president of the American Acne and Rosacea Society.
Male patients taking isotretinoin must visit their physician every month to get a new no-refills prescription, but females must get a pregnancy test at a Clinical Laboratory Improvement Amendments–certified lab, which must then provide physical results to the prescribing physician. The doctor enters the negative pregnancy test and the two forms of birth control the patient is taking in the iPLEDGE program site.
Then the patient must take an online test at home to acknowledge they understand what it means to not get pregnant and enter the two forms of birth control they are using – which must match what the doctor enters – before the pharmacy can dispense the drug. The entire process must occur within 7 days or else the patient has to wait 19 days before starting the process over.
“We run a very tight schedule for girls. And every month, we would worry that something would interfere, a snow storm or something else, and that they wouldn’t be able to complete their objectives within the 7-day period,” Dr Baldwin said in an interview. “It was always difficult, and now with us not being able to see the patient and the patient not wanting to go to the lab, this became completely impossible.”
Until this change, some patients may not have been able to get their prescription for severe nodulocystic acne, which can cause physical and psychological scarring, and “postponing treatment increases the likelihood of scarring,” Dr. Baldwin pointed out.

Dr. Goldberg’s patients now take a pregnancy test at home and send him a photo of the negative test that he then inserts into their EMR.
According to a March 17 statement from HHS, potential penalties for HIPAA violations are waived for good-faith use of “everyday communication technologies,” such as Skype or FaceTime, for telehealth treatment or diagnostics. The change was intended to allow telehealth services to continue healthcare for practices that had not previously had secure telehealth technology established.
Despite the changes for at-home pregnancy tests for females and in-person visits for all patients, the program has not altered the 7-day prescription window or the requirement to have two forms of birth control.
With reports of a global condom shortage, Dr Baldwin said she has more concerns about her adult patients being able to find a required barrier method of birth control than about her adolescent patients.
“This is a unique opportunity for us to trust our teenage patients because they can’t leave the house,” Dr. Baldwin said. “I’m actually more worried about my adult women on the drug who are bored and cooped up in a house with their significant other.”
Dr. Baldwin and Dr. Goldberg had no relevant disclosures. Dr. Goldberg is a Dermatology News board member.
Low-income DC communities have restricted access to iPLEDGE pharmacies
Residents of , results from a survey demonstrated.
Prescription of isotretinoin is regulated by the iPLEDGE program, which strives to ensure that no female patient starts isotretinoin therapy if pregnant and that no female patient on isotretinoin therapy becomes pregnant. “Over the years, many studies have criticized the program by demonstrating that iPLEDGE has promoted health care disparities,” Nidhi Shah said during a virtual meeting held by the George Washington University department of dermatology. “For example, racial minorities and women are more likely to be underprescribed isotretinoin, as well as face more delays in treatment.”
In an effort to evaluate the geographic distribution of iPLEDGE pharmacies in Washington DC, and its correlation with sociodemographic factors, Ms. Shah, a third-year medical student at the George Washington University, Washington, and colleagues obtained a list of active pharmacies in Washington from the local government. They also surveyed each outpatient pharmacy in the District of Columbia to verify their iPLEDGE registration status, for a total of 146 pharmacies.
Ms. Shah reported that 82% of all outpatient pharmacies were enrolled in iPLEDGE. However, enrollment significantly varied by the type of pharmacy. For example, 100% of chain pharmacies were enrolled, compared with 46% of independent pharmacies and 60% of hospital-based pharmacies.
When the researchers evaluated the number and type of iPLEDGE pharmacy by each of the eight wards in Washington, they observed a high density of pharmacies in wards 1 and 2, communities with a generally low proportion of residents who live in poverty, and low density of pharmacies in wards 7 and 8, communities with a higher proportion of residents who live in poverty. In addition, there were more independent than chain pharmacies in wards 7 and 8, and residents in those wards had a greater distance to travel to reach an iPLEDGE pharmacy, compared with residents who live in the other wards.
When Ms. Shah and colleagues examined the correlation between pharmacies per 10,000 residents and specific sociodemographic factors, they observed a strong, positive correlation between iPLEDGE pharmacy density and median household income (P = .0003). On the other hand, there was a strong negative correlation between iPLEDGE pharmacy density and the percentage of individuals with public insurance (P less than .0001), as well as the percentage of nonwhite individuals (P = .0009).
“Our study highlights the lack of isotretinoin-dispensing pharmacies in low-income communities,” Ms. Shah concluded. “Not only are there fewer such pharmacies available in low income communities, but the residents must also travel further to reach them. The spatial heterogeneity of iPLEDGE pharmacies may be an important patient barrier to timely access of isotretinoin, especially for female patients who have a strict 7-day window to collect their medication. We hope that future public health reform works to close this gap.”
The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic. Ms. Shah reported having no disclosures.
Residents of , results from a survey demonstrated.
Prescription of isotretinoin is regulated by the iPLEDGE program, which strives to ensure that no female patient starts isotretinoin therapy if pregnant and that no female patient on isotretinoin therapy becomes pregnant. “Over the years, many studies have criticized the program by demonstrating that iPLEDGE has promoted health care disparities,” Nidhi Shah said during a virtual meeting held by the George Washington University department of dermatology. “For example, racial minorities and women are more likely to be underprescribed isotretinoin, as well as face more delays in treatment.”
In an effort to evaluate the geographic distribution of iPLEDGE pharmacies in Washington DC, and its correlation with sociodemographic factors, Ms. Shah, a third-year medical student at the George Washington University, Washington, and colleagues obtained a list of active pharmacies in Washington from the local government. They also surveyed each outpatient pharmacy in the District of Columbia to verify their iPLEDGE registration status, for a total of 146 pharmacies.
Ms. Shah reported that 82% of all outpatient pharmacies were enrolled in iPLEDGE. However, enrollment significantly varied by the type of pharmacy. For example, 100% of chain pharmacies were enrolled, compared with 46% of independent pharmacies and 60% of hospital-based pharmacies.
When the researchers evaluated the number and type of iPLEDGE pharmacy by each of the eight wards in Washington, they observed a high density of pharmacies in wards 1 and 2, communities with a generally low proportion of residents who live in poverty, and low density of pharmacies in wards 7 and 8, communities with a higher proportion of residents who live in poverty. In addition, there were more independent than chain pharmacies in wards 7 and 8, and residents in those wards had a greater distance to travel to reach an iPLEDGE pharmacy, compared with residents who live in the other wards.
When Ms. Shah and colleagues examined the correlation between pharmacies per 10,000 residents and specific sociodemographic factors, they observed a strong, positive correlation between iPLEDGE pharmacy density and median household income (P = .0003). On the other hand, there was a strong negative correlation between iPLEDGE pharmacy density and the percentage of individuals with public insurance (P less than .0001), as well as the percentage of nonwhite individuals (P = .0009).
“Our study highlights the lack of isotretinoin-dispensing pharmacies in low-income communities,” Ms. Shah concluded. “Not only are there fewer such pharmacies available in low income communities, but the residents must also travel further to reach them. The spatial heterogeneity of iPLEDGE pharmacies may be an important patient barrier to timely access of isotretinoin, especially for female patients who have a strict 7-day window to collect their medication. We hope that future public health reform works to close this gap.”
The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic. Ms. Shah reported having no disclosures.
Residents of , results from a survey demonstrated.
Prescription of isotretinoin is regulated by the iPLEDGE program, which strives to ensure that no female patient starts isotretinoin therapy if pregnant and that no female patient on isotretinoin therapy becomes pregnant. “Over the years, many studies have criticized the program by demonstrating that iPLEDGE has promoted health care disparities,” Nidhi Shah said during a virtual meeting held by the George Washington University department of dermatology. “For example, racial minorities and women are more likely to be underprescribed isotretinoin, as well as face more delays in treatment.”
In an effort to evaluate the geographic distribution of iPLEDGE pharmacies in Washington DC, and its correlation with sociodemographic factors, Ms. Shah, a third-year medical student at the George Washington University, Washington, and colleagues obtained a list of active pharmacies in Washington from the local government. They also surveyed each outpatient pharmacy in the District of Columbia to verify their iPLEDGE registration status, for a total of 146 pharmacies.
Ms. Shah reported that 82% of all outpatient pharmacies were enrolled in iPLEDGE. However, enrollment significantly varied by the type of pharmacy. For example, 100% of chain pharmacies were enrolled, compared with 46% of independent pharmacies and 60% of hospital-based pharmacies.
When the researchers evaluated the number and type of iPLEDGE pharmacy by each of the eight wards in Washington, they observed a high density of pharmacies in wards 1 and 2, communities with a generally low proportion of residents who live in poverty, and low density of pharmacies in wards 7 and 8, communities with a higher proportion of residents who live in poverty. In addition, there were more independent than chain pharmacies in wards 7 and 8, and residents in those wards had a greater distance to travel to reach an iPLEDGE pharmacy, compared with residents who live in the other wards.
When Ms. Shah and colleagues examined the correlation between pharmacies per 10,000 residents and specific sociodemographic factors, they observed a strong, positive correlation between iPLEDGE pharmacy density and median household income (P = .0003). On the other hand, there was a strong negative correlation between iPLEDGE pharmacy density and the percentage of individuals with public insurance (P less than .0001), as well as the percentage of nonwhite individuals (P = .0009).
“Our study highlights the lack of isotretinoin-dispensing pharmacies in low-income communities,” Ms. Shah concluded. “Not only are there fewer such pharmacies available in low income communities, but the residents must also travel further to reach them. The spatial heterogeneity of iPLEDGE pharmacies may be an important patient barrier to timely access of isotretinoin, especially for female patients who have a strict 7-day window to collect their medication. We hope that future public health reform works to close this gap.”
The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic. Ms. Shah reported having no disclosures.
Isotretinoin data provide postmeal absorption guidance
LAHAINA, HAWAII – Recent , Hilary E. Baldwin, MD, said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
It is recommended that isotretinoin, which is fat-soluble, be taken with food, preferably high-fat foods. So it has been unclear what the effect would be when taken with lower-fat food, such as low-fat cereal and raspberries, for example, Dr. Baldwin, medical director of the Acne Treatment and Research Center in New York, pointed out.
“We’ve been trying for years to figure out how we’re going to get around this,” and there have not been any relevant data available until recently, other than in the setting of taking isotretinoin on an empty stomach or with a high-fat meal, she commented.
She referred to a open-label, single-dose, randomized crossover study that compared the bioavailability of the lidose formulation of isotretinoin (Absorica) and brand name Accutane, at a dose of 40 mg either on top of a fatty meal (the Food and Drug Administration-stipulated high-fat, high-calorie diet) or after a 10-hour fast; 60 patients did all four arms, with a 21-day washout period between them (J Am Acad Dermatol. 2013 Nov;69[5]:762-7).
In the fed state, both isotretinoin formulations were absorbed to the same extent, “but in the fasting state, there was a considerable difference,” Dr. Baldwin said. Absorption of both dropped in the fasting state, but the drop was more extreme with Accutane, “about a 50% difference between the two, in terms of how much drug was getting into the system,” she noted.
That is important because weight-based dosing is considered with isotretinoin, so at the end of treatment, a patient who has been taking it on an empty stomach may be getting a 60% lower dose than prescribed, “which could lead to a lessening of the effectiveness of the drug and also an increase in relapse over time.”
But how would a low-fat meal, like low-fat cereal and raspberries, affect the absorption, and ultimate efficacy?
This question was addressed in an open-label, single-arm study of 163 patients with acne, who were taking the lidose isotretinoin formulation without food, at the standard dose, for no longer than 20 weeks. Whether they relapsed was evaluated in a 2-year observational phase of the study, Dr. Baldwin said.
At the end of the trial, the drug was considered effective, with improvements in IGA (the 5-point Investigator’s Global Assessment scale). But the change from baseline was maintained at the 2-year posttreatment period, so the benefits of treatment lasted, which indicates that patients can take it “on top of absolutely no food whatsoever ... so if they eat anything, we are headed in the right direction,” including a low-fat meal. During the 2-year period, most patients did not need to be retreated. Of those people who needed treatment, only 4.2% needed treatment with isotretinoin, which is better than the historical relapse rates with isotretinoin, she noted.
Dr. Baldwin’s disclosures included being on the speakers’ bureau, serving as an advisor, and/or an investigator for companies that include Almirall, BioPharmx, Foamix, Galderma, Ortho Dermatologics, Sun Pharmaceuticals, Johnson & Johnson, and La Roche–Posay.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – Recent , Hilary E. Baldwin, MD, said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
It is recommended that isotretinoin, which is fat-soluble, be taken with food, preferably high-fat foods. So it has been unclear what the effect would be when taken with lower-fat food, such as low-fat cereal and raspberries, for example, Dr. Baldwin, medical director of the Acne Treatment and Research Center in New York, pointed out.
“We’ve been trying for years to figure out how we’re going to get around this,” and there have not been any relevant data available until recently, other than in the setting of taking isotretinoin on an empty stomach or with a high-fat meal, she commented.
She referred to a open-label, single-dose, randomized crossover study that compared the bioavailability of the lidose formulation of isotretinoin (Absorica) and brand name Accutane, at a dose of 40 mg either on top of a fatty meal (the Food and Drug Administration-stipulated high-fat, high-calorie diet) or after a 10-hour fast; 60 patients did all four arms, with a 21-day washout period between them (J Am Acad Dermatol. 2013 Nov;69[5]:762-7).
In the fed state, both isotretinoin formulations were absorbed to the same extent, “but in the fasting state, there was a considerable difference,” Dr. Baldwin said. Absorption of both dropped in the fasting state, but the drop was more extreme with Accutane, “about a 50% difference between the two, in terms of how much drug was getting into the system,” she noted.
That is important because weight-based dosing is considered with isotretinoin, so at the end of treatment, a patient who has been taking it on an empty stomach may be getting a 60% lower dose than prescribed, “which could lead to a lessening of the effectiveness of the drug and also an increase in relapse over time.”
But how would a low-fat meal, like low-fat cereal and raspberries, affect the absorption, and ultimate efficacy?
This question was addressed in an open-label, single-arm study of 163 patients with acne, who were taking the lidose isotretinoin formulation without food, at the standard dose, for no longer than 20 weeks. Whether they relapsed was evaluated in a 2-year observational phase of the study, Dr. Baldwin said.
At the end of the trial, the drug was considered effective, with improvements in IGA (the 5-point Investigator’s Global Assessment scale). But the change from baseline was maintained at the 2-year posttreatment period, so the benefits of treatment lasted, which indicates that patients can take it “on top of absolutely no food whatsoever ... so if they eat anything, we are headed in the right direction,” including a low-fat meal. During the 2-year period, most patients did not need to be retreated. Of those people who needed treatment, only 4.2% needed treatment with isotretinoin, which is better than the historical relapse rates with isotretinoin, she noted.
Dr. Baldwin’s disclosures included being on the speakers’ bureau, serving as an advisor, and/or an investigator for companies that include Almirall, BioPharmx, Foamix, Galderma, Ortho Dermatologics, Sun Pharmaceuticals, Johnson & Johnson, and La Roche–Posay.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – Recent , Hilary E. Baldwin, MD, said at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
It is recommended that isotretinoin, which is fat-soluble, be taken with food, preferably high-fat foods. So it has been unclear what the effect would be when taken with lower-fat food, such as low-fat cereal and raspberries, for example, Dr. Baldwin, medical director of the Acne Treatment and Research Center in New York, pointed out.
“We’ve been trying for years to figure out how we’re going to get around this,” and there have not been any relevant data available until recently, other than in the setting of taking isotretinoin on an empty stomach or with a high-fat meal, she commented.
She referred to a open-label, single-dose, randomized crossover study that compared the bioavailability of the lidose formulation of isotretinoin (Absorica) and brand name Accutane, at a dose of 40 mg either on top of a fatty meal (the Food and Drug Administration-stipulated high-fat, high-calorie diet) or after a 10-hour fast; 60 patients did all four arms, with a 21-day washout period between them (J Am Acad Dermatol. 2013 Nov;69[5]:762-7).
In the fed state, both isotretinoin formulations were absorbed to the same extent, “but in the fasting state, there was a considerable difference,” Dr. Baldwin said. Absorption of both dropped in the fasting state, but the drop was more extreme with Accutane, “about a 50% difference between the two, in terms of how much drug was getting into the system,” she noted.
That is important because weight-based dosing is considered with isotretinoin, so at the end of treatment, a patient who has been taking it on an empty stomach may be getting a 60% lower dose than prescribed, “which could lead to a lessening of the effectiveness of the drug and also an increase in relapse over time.”
But how would a low-fat meal, like low-fat cereal and raspberries, affect the absorption, and ultimate efficacy?
This question was addressed in an open-label, single-arm study of 163 patients with acne, who were taking the lidose isotretinoin formulation without food, at the standard dose, for no longer than 20 weeks. Whether they relapsed was evaluated in a 2-year observational phase of the study, Dr. Baldwin said.
At the end of the trial, the drug was considered effective, with improvements in IGA (the 5-point Investigator’s Global Assessment scale). But the change from baseline was maintained at the 2-year posttreatment period, so the benefits of treatment lasted, which indicates that patients can take it “on top of absolutely no food whatsoever ... so if they eat anything, we are headed in the right direction,” including a low-fat meal. During the 2-year period, most patients did not need to be retreated. Of those people who needed treatment, only 4.2% needed treatment with isotretinoin, which is better than the historical relapse rates with isotretinoin, she noted.
Dr. Baldwin’s disclosures included being on the speakers’ bureau, serving as an advisor, and/or an investigator for companies that include Almirall, BioPharmx, Foamix, Galderma, Ortho Dermatologics, Sun Pharmaceuticals, Johnson & Johnson, and La Roche–Posay.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR
Avoid ‘mutant selection window’ when prescribing antibiotics for acne
LAHAINA, HAWAII – Consider the “mutant selection window” to reduce antibiotic resistance when treating acne, Hilary E. Baldwin, MD, advised at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
Dermatologists continue to write a disproportionate number of prescriptions for antibiotics, particularly tetracyclines, noted Dr. Baldwin, medical director of the Acne Treatment and Research Center in New York. In addition to limiting unnecessary use of antimicrobials, strategies for slowing antimicrobial resistance include using anti-inflammatory doses of doxycycline; using more retinoids, isotretinoin, spironolactone, and oral contraceptives; and improving patient compliance with treatment.
Dermatologists can also “pay attention to the bug we are treating and ... make sure the concentration of the drug that we are using is appropriate to the bug we’re trying to kill,” while also targeting resistant organisms. Dr. Baldwin referred to a paper in the infectious disease literature titled: “The mutant selection window and antimicrobial resistance,” which points out that a drug concentration range exists for which mutant strains of bacteria are selected most frequently (J Antimicrob Chemother. 2003 Jul;52[1]:11-7). The dimensions of this range, or “window,” are characteristic of each pathogen-antimicrobial combination. A high enough drug concentration will eliminate both resistant and sensitive strains of the pathogen.
The paper notes that the minimum inhibitory concentration (MIC) is the lowest concentration that will inhibit the visible growth of a microorganism. The mutant prevention concentration (MPC) is the minimum drug concentration needed to prevent the growth of resistant strains, Dr. Baldwin said. The mutant selection window is the concentration range that extends from the MIC up to the MPC, the range “within which resistant mutants are likely to emerge.” If the antimicrobial concentration falls within this window, a mutant strain is likely to develop and “you’re going to add to the problem of antibiotic resistance,” she explained. “So the goal is to treat low or to treat high, but not right in the middle.”
“This is not theoretical,” and has been shown over and over again, with, for example, Streptococcus pneumonia and moxifloxacin, she said (J Antimicrob Chemother. 2003 Oct;52[4]:616-22.).
When the therapeutic window does not extend all the way to the MPC, “toxicity starts to kick in before you can get high enough to kill off the whole group of organisms,” in which case a low-dose strategy would reduce the development of resistant organisms, she noted.
“We’re doing this already,” with topical antifungals, Dr. Baldwin pointed out, asking when the last time anyone heard that a fungus developed resistance to topical antifungal therapy. “Never, because we use our antifungals in such a high dose, that we’re 500 times the MPC.”
Using an anti-inflammatory dose of doxycycline for treating acne or rosacea is a low-dose strategy, and the 40-mg delayed-release dose stays “way below” the antimicrobial threshold, she said, but the 50-mg dose falls “right in the middle of that mutant selection window.”
As more treatments become available, it will be important to determine how to dose topical antibiotics so that they do not fall within the mutant selection window and avoid what happened with clindamycin and erythromycin, “where the topical use of these medications led to the development of resistance such that they no longer work for the treatment” of Cutibacterium acnes.
Dr. Baldwin disclosures included being on the speakers bureau, serving as an advisor, and/or an investigator for companies that include Almirall, BioPharmx, Foamix, Galderma, Ortho Dermatologics, Sun Pharmaceuticals, Johnson & Johnson, and La Roche–Posay.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – Consider the “mutant selection window” to reduce antibiotic resistance when treating acne, Hilary E. Baldwin, MD, advised at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
Dermatologists continue to write a disproportionate number of prescriptions for antibiotics, particularly tetracyclines, noted Dr. Baldwin, medical director of the Acne Treatment and Research Center in New York. In addition to limiting unnecessary use of antimicrobials, strategies for slowing antimicrobial resistance include using anti-inflammatory doses of doxycycline; using more retinoids, isotretinoin, spironolactone, and oral contraceptives; and improving patient compliance with treatment.
Dermatologists can also “pay attention to the bug we are treating and ... make sure the concentration of the drug that we are using is appropriate to the bug we’re trying to kill,” while also targeting resistant organisms. Dr. Baldwin referred to a paper in the infectious disease literature titled: “The mutant selection window and antimicrobial resistance,” which points out that a drug concentration range exists for which mutant strains of bacteria are selected most frequently (J Antimicrob Chemother. 2003 Jul;52[1]:11-7). The dimensions of this range, or “window,” are characteristic of each pathogen-antimicrobial combination. A high enough drug concentration will eliminate both resistant and sensitive strains of the pathogen.
The paper notes that the minimum inhibitory concentration (MIC) is the lowest concentration that will inhibit the visible growth of a microorganism. The mutant prevention concentration (MPC) is the minimum drug concentration needed to prevent the growth of resistant strains, Dr. Baldwin said. The mutant selection window is the concentration range that extends from the MIC up to the MPC, the range “within which resistant mutants are likely to emerge.” If the antimicrobial concentration falls within this window, a mutant strain is likely to develop and “you’re going to add to the problem of antibiotic resistance,” she explained. “So the goal is to treat low or to treat high, but not right in the middle.”
“This is not theoretical,” and has been shown over and over again, with, for example, Streptococcus pneumonia and moxifloxacin, she said (J Antimicrob Chemother. 2003 Oct;52[4]:616-22.).
When the therapeutic window does not extend all the way to the MPC, “toxicity starts to kick in before you can get high enough to kill off the whole group of organisms,” in which case a low-dose strategy would reduce the development of resistant organisms, she noted.
“We’re doing this already,” with topical antifungals, Dr. Baldwin pointed out, asking when the last time anyone heard that a fungus developed resistance to topical antifungal therapy. “Never, because we use our antifungals in such a high dose, that we’re 500 times the MPC.”
Using an anti-inflammatory dose of doxycycline for treating acne or rosacea is a low-dose strategy, and the 40-mg delayed-release dose stays “way below” the antimicrobial threshold, she said, but the 50-mg dose falls “right in the middle of that mutant selection window.”
As more treatments become available, it will be important to determine how to dose topical antibiotics so that they do not fall within the mutant selection window and avoid what happened with clindamycin and erythromycin, “where the topical use of these medications led to the development of resistance such that they no longer work for the treatment” of Cutibacterium acnes.
Dr. Baldwin disclosures included being on the speakers bureau, serving as an advisor, and/or an investigator for companies that include Almirall, BioPharmx, Foamix, Galderma, Ortho Dermatologics, Sun Pharmaceuticals, Johnson & Johnson, and La Roche–Posay.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – Consider the “mutant selection window” to reduce antibiotic resistance when treating acne, Hilary E. Baldwin, MD, advised at the Hawaii Dermatology Seminar provided by Global Academy for Medical Education/Skin Disease Education Foundation.
Dermatologists continue to write a disproportionate number of prescriptions for antibiotics, particularly tetracyclines, noted Dr. Baldwin, medical director of the Acne Treatment and Research Center in New York. In addition to limiting unnecessary use of antimicrobials, strategies for slowing antimicrobial resistance include using anti-inflammatory doses of doxycycline; using more retinoids, isotretinoin, spironolactone, and oral contraceptives; and improving patient compliance with treatment.
Dermatologists can also “pay attention to the bug we are treating and ... make sure the concentration of the drug that we are using is appropriate to the bug we’re trying to kill,” while also targeting resistant organisms. Dr. Baldwin referred to a paper in the infectious disease literature titled: “The mutant selection window and antimicrobial resistance,” which points out that a drug concentration range exists for which mutant strains of bacteria are selected most frequently (J Antimicrob Chemother. 2003 Jul;52[1]:11-7). The dimensions of this range, or “window,” are characteristic of each pathogen-antimicrobial combination. A high enough drug concentration will eliminate both resistant and sensitive strains of the pathogen.
The paper notes that the minimum inhibitory concentration (MIC) is the lowest concentration that will inhibit the visible growth of a microorganism. The mutant prevention concentration (MPC) is the minimum drug concentration needed to prevent the growth of resistant strains, Dr. Baldwin said. The mutant selection window is the concentration range that extends from the MIC up to the MPC, the range “within which resistant mutants are likely to emerge.” If the antimicrobial concentration falls within this window, a mutant strain is likely to develop and “you’re going to add to the problem of antibiotic resistance,” she explained. “So the goal is to treat low or to treat high, but not right in the middle.”
“This is not theoretical,” and has been shown over and over again, with, for example, Streptococcus pneumonia and moxifloxacin, she said (J Antimicrob Chemother. 2003 Oct;52[4]:616-22.).
When the therapeutic window does not extend all the way to the MPC, “toxicity starts to kick in before you can get high enough to kill off the whole group of organisms,” in which case a low-dose strategy would reduce the development of resistant organisms, she noted.
“We’re doing this already,” with topical antifungals, Dr. Baldwin pointed out, asking when the last time anyone heard that a fungus developed resistance to topical antifungal therapy. “Never, because we use our antifungals in such a high dose, that we’re 500 times the MPC.”
Using an anti-inflammatory dose of doxycycline for treating acne or rosacea is a low-dose strategy, and the 40-mg delayed-release dose stays “way below” the antimicrobial threshold, she said, but the 50-mg dose falls “right in the middle of that mutant selection window.”
As more treatments become available, it will be important to determine how to dose topical antibiotics so that they do not fall within the mutant selection window and avoid what happened with clindamycin and erythromycin, “where the topical use of these medications led to the development of resistance such that they no longer work for the treatment” of Cutibacterium acnes.
Dr. Baldwin disclosures included being on the speakers bureau, serving as an advisor, and/or an investigator for companies that include Almirall, BioPharmx, Foamix, Galderma, Ortho Dermatologics, Sun Pharmaceuticals, Johnson & Johnson, and La Roche–Posay.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
REPORTING FROM SDEF HAWAII DERMATOLOGY SEMINAR
Acne treatment may vary based on race, gender, insurance
based on findings from a retrospective, cohort study of 29,928 individuals with acne.
“Our findings suggest the presence of racial/ethnic, sex, and insurance-based disparities in health care use and treatment for acne and raise particular concern for undertreatment among racial/ethnic minority and female patients,” John S. Barbieri, MD, a dermatology research fellow at the University of Pennsylvania, Philadelphia, and colleagues wrote in a study published in JAMA Dermatology.
Data from previous studies have suggested racial disparities in the management of several dermatologic conditions, including atopic dermatitis and psoriasis, but associations between social demographics and prescribing patterns have not been well studied for acne treatment, the authors noted.
For the current study, the researchers used deidentified data from the Optum electronic health record from Jan. 1, 2007 to June 30, 2017. In all, 29,928 patients aged 15-35 years and who were being treated for acne were included in the study. Of that total, 64% were women, 8% were non-Hispanic black and 68% were white, with the remaining patients grouped as non-Hispanic Asian, Hispanic, or other.
Non-Hispanic black patients were significantly more likely to be seen by a dermatologist, compared with non-Hispanic white patients, who were designated as the reference (odds ratio, 1.20). However, the black patients were less likely to receive prescriptions for any acne medication (incidence rate ratio, 0.89).
Non-Hispanic black patients were more likely than non-Hispanic white patients to be prescribed topical retinoids or topical antibiotics (OR, 1.25 and 1.35, respectively). They were also were less likely than their white counterparts to be prescribed oral antibiotics, spironolactone, and isotretinoin (OR, 0.80, 0.68, and 0.39, respectively).
Overall, men were more than twice as likely as women to receive prescriptions for isotretinoin (OR, 2.44). They were also more likely to receive prescriptions for the other treatments, but the differences were not as high as those for isotretinoin.
In addition, patients with Medicaid insurance were significantly less likely than those with commercial insurance (the reference) to see a dermatologist (OR, 0.46). Medicaid patients also were less likely to be prescribed topical retinoids, oral antibiotics, spironolactone, or isotretinoin (OR, 0.82, 0.87, 0.50, and 0.43, respectively).
The study findings were limited by several factors, among them, the use of automated pharmacy data without confirmation that patients had picked up the medications they had been prescribed, the researchers said. The study also lacked data on acne severity, clinical outcomes, and the use of over-the-counter acne treatments.
“Further study is needed to confirm our findings, provide understanding of the reasons for these potential disparities, and develop strategies to ensure equitable care for patients with acne,” the researchers concluded.
The study was supported in part by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health, and by a Pfizer Fellowship in Dermatology Patient Oriented Research grant to the Trustees of the University of Pennsylvania. Dr. Barbieri had no financial conflicts to disclose. One of the study coauthors disclosed relationships with Pfizer, Eli Lilly, and Novartis.
SOURCE: Barbieri JS et al. JAMA Dermatol. 2020 Feb 5. doi: 10.1001/jamadermatol.2019.4818.
based on findings from a retrospective, cohort study of 29,928 individuals with acne.
“Our findings suggest the presence of racial/ethnic, sex, and insurance-based disparities in health care use and treatment for acne and raise particular concern for undertreatment among racial/ethnic minority and female patients,” John S. Barbieri, MD, a dermatology research fellow at the University of Pennsylvania, Philadelphia, and colleagues wrote in a study published in JAMA Dermatology.
Data from previous studies have suggested racial disparities in the management of several dermatologic conditions, including atopic dermatitis and psoriasis, but associations between social demographics and prescribing patterns have not been well studied for acne treatment, the authors noted.
For the current study, the researchers used deidentified data from the Optum electronic health record from Jan. 1, 2007 to June 30, 2017. In all, 29,928 patients aged 15-35 years and who were being treated for acne were included in the study. Of that total, 64% were women, 8% were non-Hispanic black and 68% were white, with the remaining patients grouped as non-Hispanic Asian, Hispanic, or other.
Non-Hispanic black patients were significantly more likely to be seen by a dermatologist, compared with non-Hispanic white patients, who were designated as the reference (odds ratio, 1.20). However, the black patients were less likely to receive prescriptions for any acne medication (incidence rate ratio, 0.89).
Non-Hispanic black patients were more likely than non-Hispanic white patients to be prescribed topical retinoids or topical antibiotics (OR, 1.25 and 1.35, respectively). They were also were less likely than their white counterparts to be prescribed oral antibiotics, spironolactone, and isotretinoin (OR, 0.80, 0.68, and 0.39, respectively).
Overall, men were more than twice as likely as women to receive prescriptions for isotretinoin (OR, 2.44). They were also more likely to receive prescriptions for the other treatments, but the differences were not as high as those for isotretinoin.
In addition, patients with Medicaid insurance were significantly less likely than those with commercial insurance (the reference) to see a dermatologist (OR, 0.46). Medicaid patients also were less likely to be prescribed topical retinoids, oral antibiotics, spironolactone, or isotretinoin (OR, 0.82, 0.87, 0.50, and 0.43, respectively).
The study findings were limited by several factors, among them, the use of automated pharmacy data without confirmation that patients had picked up the medications they had been prescribed, the researchers said. The study also lacked data on acne severity, clinical outcomes, and the use of over-the-counter acne treatments.
“Further study is needed to confirm our findings, provide understanding of the reasons for these potential disparities, and develop strategies to ensure equitable care for patients with acne,” the researchers concluded.
The study was supported in part by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health, and by a Pfizer Fellowship in Dermatology Patient Oriented Research grant to the Trustees of the University of Pennsylvania. Dr. Barbieri had no financial conflicts to disclose. One of the study coauthors disclosed relationships with Pfizer, Eli Lilly, and Novartis.
SOURCE: Barbieri JS et al. JAMA Dermatol. 2020 Feb 5. doi: 10.1001/jamadermatol.2019.4818.
based on findings from a retrospective, cohort study of 29,928 individuals with acne.
“Our findings suggest the presence of racial/ethnic, sex, and insurance-based disparities in health care use and treatment for acne and raise particular concern for undertreatment among racial/ethnic minority and female patients,” John S. Barbieri, MD, a dermatology research fellow at the University of Pennsylvania, Philadelphia, and colleagues wrote in a study published in JAMA Dermatology.
Data from previous studies have suggested racial disparities in the management of several dermatologic conditions, including atopic dermatitis and psoriasis, but associations between social demographics and prescribing patterns have not been well studied for acne treatment, the authors noted.
For the current study, the researchers used deidentified data from the Optum electronic health record from Jan. 1, 2007 to June 30, 2017. In all, 29,928 patients aged 15-35 years and who were being treated for acne were included in the study. Of that total, 64% were women, 8% were non-Hispanic black and 68% were white, with the remaining patients grouped as non-Hispanic Asian, Hispanic, or other.
Non-Hispanic black patients were significantly more likely to be seen by a dermatologist, compared with non-Hispanic white patients, who were designated as the reference (odds ratio, 1.20). However, the black patients were less likely to receive prescriptions for any acne medication (incidence rate ratio, 0.89).
Non-Hispanic black patients were more likely than non-Hispanic white patients to be prescribed topical retinoids or topical antibiotics (OR, 1.25 and 1.35, respectively). They were also were less likely than their white counterparts to be prescribed oral antibiotics, spironolactone, and isotretinoin (OR, 0.80, 0.68, and 0.39, respectively).
Overall, men were more than twice as likely as women to receive prescriptions for isotretinoin (OR, 2.44). They were also more likely to receive prescriptions for the other treatments, but the differences were not as high as those for isotretinoin.
In addition, patients with Medicaid insurance were significantly less likely than those with commercial insurance (the reference) to see a dermatologist (OR, 0.46). Medicaid patients also were less likely to be prescribed topical retinoids, oral antibiotics, spironolactone, or isotretinoin (OR, 0.82, 0.87, 0.50, and 0.43, respectively).
The study findings were limited by several factors, among them, the use of automated pharmacy data without confirmation that patients had picked up the medications they had been prescribed, the researchers said. The study also lacked data on acne severity, clinical outcomes, and the use of over-the-counter acne treatments.
“Further study is needed to confirm our findings, provide understanding of the reasons for these potential disparities, and develop strategies to ensure equitable care for patients with acne,” the researchers concluded.
The study was supported in part by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health, and by a Pfizer Fellowship in Dermatology Patient Oriented Research grant to the Trustees of the University of Pennsylvania. Dr. Barbieri had no financial conflicts to disclose. One of the study coauthors disclosed relationships with Pfizer, Eli Lilly, and Novartis.
SOURCE: Barbieri JS et al. JAMA Dermatol. 2020 Feb 5. doi: 10.1001/jamadermatol.2019.4818.
FROM JAMA DERMATOLOGY