User login
New York City inpatient detox unit keeps running: Here’s how
Substance use disorder and its daily consequences take no breaks even during a pandemic. The stressors created by COVID-19, including deaths of loved ones and the disruptions to normal life from policies aimed at flattening the curve, seem to have increased substance use.
I practice as a hospitalist with an internal medicine background and specialty in addiction medicine at BronxCare Health System’s inpatient detoxification unit, a 24/7, 20-bed medically-supervised unit in South Bronx in New York City. It is one of the comprehensive services provided by the BronxCare’s life recovery center and addiction services, which also includes an outpatient clinic, opioid treatment program, inpatient rehab, and a half-way house. Inpatient detoxification units like ours are designed to treat serious addictions and chemical dependency and prevent and treat life-threatening withdrawal symptoms and signs or complications. Our patients come from all over the city and its adjoining suburbs, including from emergency room referrals, referral clinics, courts and the justice system, walk-ins, and self-referrals.
At a time when many inpatient detoxification units within the city were temporarily closed due to fear of inpatient spread of the virus or to provide extra COVID beds in anticipation for the peak surge, we have been able to provide a needed service. In fact, several other inpatient detoxification programs within the city have been able to refer their patients to our facility.
Individuals with substance use disorder have historically been a vulnerable and underserved population and possess high risk for multiple health problems as well as preexisting conditions. Many have limited life options financially, educationally, and with housing, and encounter barriers to accessing primary health care services, including preventive services. The introduction of the COVID-19 pandemic into these patients’ precarious health situations only made things worse as many of the limited resources for patients with substance use disorder were diverted to battling the pandemic. Numerous inpatient and outpatient addiction services, for example, were temporarily shut down. This has led to an increase in domestic violence, and psychiatric decompensation, including psychosis, suicidal attempts, and worsening of medical comorbidities in these patients.
Our wake-up call came when the first case of COVID-19 was confirmed in New York in early March. Within a short period of time the state became the epicenter for COVID-19. With the projection of millions of cases being positive and the number of new cases doubling every third day at the onset in New York City, we knew we had a battle brewing and needed to radically transform our mode of operation fast.
Our first task was to ensure the safety of our patients and the dedicated health workers attending to them. We streamlined the patient point of entry through one screening site, while also brushing up on our history-taking to intently screen for COVID-19. This included not just focusing on travels from China, but from Europe and other parts of the world.
Yes, we did ask patients about cough, fever, shortness of breath or difficulty breathing, feeling fatigued, severe body ache, and possible contact with someone who is sick or has traveled overseas. But we were also attuned to the increased rate of community spread and the presentation of other symptoms, such as loss of taste and smell, early in the process. Hence we were able to triage patients with suspected cases to the appropriate sections of the hospital for further screening, testing, and evaluation, instead of having those patients admitted to the detox unit.
Early in the process a huddle team was instituted with daily briefing of staff lasting 30 minutes or less. This team consists of physicians, nurses, a physician assistant, a social worker, and a counselor. In addition to discussing treatment plans for the patient, they deliberate on the public health information from the hospital’s COVID-19 command center, New York State Department of Health, the Office of Mental Health, and the Centers for Disease Control and Prevention concerning the latest evidence-based information. These discussions have helped us modify our policies and practices.
We instituted a no visiting rule during a short hospital stay of 5-7 days, and this was initiated weeks in advance of many institutions, including nursing homes with vulnerable populations. Our admitting criteria was reviewed to allow for admission of only those patients who absolutely needed inpatient substance use disorder treatment, including patients with severe withdrawal symptoms and signs, comorbidities, or neuropsychiatric manifestations that made them unsafe for outpatient or home detoxification. Others were triaged to the outpatient services which was amply supported with telemedicine. Rooms and designated areas of the building were earmarked as places for isolation/quarantine if suspected COVID-19 cases were identified pending testing. To assess patients’ risk of COVID-19, we do point-of-care nasopharyngeal swab testing with polymerase chain reaction.
Regarding face masks, patients and staff were fitted with ones early in the process. Additionally, staff were trained on the importance of face mask use and how to ensure you have a tight seal around the mouth and nose and were provided with other appropriate personal protective equipment. Concerning social distancing, we reduced the patient population capacity for the unit down to 50% and offered only single room admissions. Social distancing was encouraged in the unit, including in the television and recreation room and dining room, and during small treatment groups of less than six individuals. Daily temperature checks with noncontact handheld thermometers were enforced for staff and anyone coming into the life recovery center.
Patients are continuously being educated on the presentations of COVID-19 and encouraged to report any symptoms. Any staff feeling sick or having symptoms are encouraged to stay home. Rigorous and continuous cleaning of surfaces, especially of areas subjected to common use, is done frequently by the hospital housekeeping and environmental crew and is the order of the day.
Dr. Fagbemi is a hospitalist at BronxCare Health System, a not-for-profit health and teaching hospital system serving South and Central Bronx in New York. He has no conflicts of interest to disclose.
Substance use disorder and its daily consequences take no breaks even during a pandemic. The stressors created by COVID-19, including deaths of loved ones and the disruptions to normal life from policies aimed at flattening the curve, seem to have increased substance use.
I practice as a hospitalist with an internal medicine background and specialty in addiction medicine at BronxCare Health System’s inpatient detoxification unit, a 24/7, 20-bed medically-supervised unit in South Bronx in New York City. It is one of the comprehensive services provided by the BronxCare’s life recovery center and addiction services, which also includes an outpatient clinic, opioid treatment program, inpatient rehab, and a half-way house. Inpatient detoxification units like ours are designed to treat serious addictions and chemical dependency and prevent and treat life-threatening withdrawal symptoms and signs or complications. Our patients come from all over the city and its adjoining suburbs, including from emergency room referrals, referral clinics, courts and the justice system, walk-ins, and self-referrals.
At a time when many inpatient detoxification units within the city were temporarily closed due to fear of inpatient spread of the virus or to provide extra COVID beds in anticipation for the peak surge, we have been able to provide a needed service. In fact, several other inpatient detoxification programs within the city have been able to refer their patients to our facility.
Individuals with substance use disorder have historically been a vulnerable and underserved population and possess high risk for multiple health problems as well as preexisting conditions. Many have limited life options financially, educationally, and with housing, and encounter barriers to accessing primary health care services, including preventive services. The introduction of the COVID-19 pandemic into these patients’ precarious health situations only made things worse as many of the limited resources for patients with substance use disorder were diverted to battling the pandemic. Numerous inpatient and outpatient addiction services, for example, were temporarily shut down. This has led to an increase in domestic violence, and psychiatric decompensation, including psychosis, suicidal attempts, and worsening of medical comorbidities in these patients.
Our wake-up call came when the first case of COVID-19 was confirmed in New York in early March. Within a short period of time the state became the epicenter for COVID-19. With the projection of millions of cases being positive and the number of new cases doubling every third day at the onset in New York City, we knew we had a battle brewing and needed to radically transform our mode of operation fast.
Our first task was to ensure the safety of our patients and the dedicated health workers attending to them. We streamlined the patient point of entry through one screening site, while also brushing up on our history-taking to intently screen for COVID-19. This included not just focusing on travels from China, but from Europe and other parts of the world.
Yes, we did ask patients about cough, fever, shortness of breath or difficulty breathing, feeling fatigued, severe body ache, and possible contact with someone who is sick or has traveled overseas. But we were also attuned to the increased rate of community spread and the presentation of other symptoms, such as loss of taste and smell, early in the process. Hence we were able to triage patients with suspected cases to the appropriate sections of the hospital for further screening, testing, and evaluation, instead of having those patients admitted to the detox unit.
Early in the process a huddle team was instituted with daily briefing of staff lasting 30 minutes or less. This team consists of physicians, nurses, a physician assistant, a social worker, and a counselor. In addition to discussing treatment plans for the patient, they deliberate on the public health information from the hospital’s COVID-19 command center, New York State Department of Health, the Office of Mental Health, and the Centers for Disease Control and Prevention concerning the latest evidence-based information. These discussions have helped us modify our policies and practices.
We instituted a no visiting rule during a short hospital stay of 5-7 days, and this was initiated weeks in advance of many institutions, including nursing homes with vulnerable populations. Our admitting criteria was reviewed to allow for admission of only those patients who absolutely needed inpatient substance use disorder treatment, including patients with severe withdrawal symptoms and signs, comorbidities, or neuropsychiatric manifestations that made them unsafe for outpatient or home detoxification. Others were triaged to the outpatient services which was amply supported with telemedicine. Rooms and designated areas of the building were earmarked as places for isolation/quarantine if suspected COVID-19 cases were identified pending testing. To assess patients’ risk of COVID-19, we do point-of-care nasopharyngeal swab testing with polymerase chain reaction.
Regarding face masks, patients and staff were fitted with ones early in the process. Additionally, staff were trained on the importance of face mask use and how to ensure you have a tight seal around the mouth and nose and were provided with other appropriate personal protective equipment. Concerning social distancing, we reduced the patient population capacity for the unit down to 50% and offered only single room admissions. Social distancing was encouraged in the unit, including in the television and recreation room and dining room, and during small treatment groups of less than six individuals. Daily temperature checks with noncontact handheld thermometers were enforced for staff and anyone coming into the life recovery center.
Patients are continuously being educated on the presentations of COVID-19 and encouraged to report any symptoms. Any staff feeling sick or having symptoms are encouraged to stay home. Rigorous and continuous cleaning of surfaces, especially of areas subjected to common use, is done frequently by the hospital housekeeping and environmental crew and is the order of the day.
Dr. Fagbemi is a hospitalist at BronxCare Health System, a not-for-profit health and teaching hospital system serving South and Central Bronx in New York. He has no conflicts of interest to disclose.
Substance use disorder and its daily consequences take no breaks even during a pandemic. The stressors created by COVID-19, including deaths of loved ones and the disruptions to normal life from policies aimed at flattening the curve, seem to have increased substance use.
I practice as a hospitalist with an internal medicine background and specialty in addiction medicine at BronxCare Health System’s inpatient detoxification unit, a 24/7, 20-bed medically-supervised unit in South Bronx in New York City. It is one of the comprehensive services provided by the BronxCare’s life recovery center and addiction services, which also includes an outpatient clinic, opioid treatment program, inpatient rehab, and a half-way house. Inpatient detoxification units like ours are designed to treat serious addictions and chemical dependency and prevent and treat life-threatening withdrawal symptoms and signs or complications. Our patients come from all over the city and its adjoining suburbs, including from emergency room referrals, referral clinics, courts and the justice system, walk-ins, and self-referrals.
At a time when many inpatient detoxification units within the city were temporarily closed due to fear of inpatient spread of the virus or to provide extra COVID beds in anticipation for the peak surge, we have been able to provide a needed service. In fact, several other inpatient detoxification programs within the city have been able to refer their patients to our facility.
Individuals with substance use disorder have historically been a vulnerable and underserved population and possess high risk for multiple health problems as well as preexisting conditions. Many have limited life options financially, educationally, and with housing, and encounter barriers to accessing primary health care services, including preventive services. The introduction of the COVID-19 pandemic into these patients’ precarious health situations only made things worse as many of the limited resources for patients with substance use disorder were diverted to battling the pandemic. Numerous inpatient and outpatient addiction services, for example, were temporarily shut down. This has led to an increase in domestic violence, and psychiatric decompensation, including psychosis, suicidal attempts, and worsening of medical comorbidities in these patients.
Our wake-up call came when the first case of COVID-19 was confirmed in New York in early March. Within a short period of time the state became the epicenter for COVID-19. With the projection of millions of cases being positive and the number of new cases doubling every third day at the onset in New York City, we knew we had a battle brewing and needed to radically transform our mode of operation fast.
Our first task was to ensure the safety of our patients and the dedicated health workers attending to them. We streamlined the patient point of entry through one screening site, while also brushing up on our history-taking to intently screen for COVID-19. This included not just focusing on travels from China, but from Europe and other parts of the world.
Yes, we did ask patients about cough, fever, shortness of breath or difficulty breathing, feeling fatigued, severe body ache, and possible contact with someone who is sick or has traveled overseas. But we were also attuned to the increased rate of community spread and the presentation of other symptoms, such as loss of taste and smell, early in the process. Hence we were able to triage patients with suspected cases to the appropriate sections of the hospital for further screening, testing, and evaluation, instead of having those patients admitted to the detox unit.
Early in the process a huddle team was instituted with daily briefing of staff lasting 30 minutes or less. This team consists of physicians, nurses, a physician assistant, a social worker, and a counselor. In addition to discussing treatment plans for the patient, they deliberate on the public health information from the hospital’s COVID-19 command center, New York State Department of Health, the Office of Mental Health, and the Centers for Disease Control and Prevention concerning the latest evidence-based information. These discussions have helped us modify our policies and practices.
We instituted a no visiting rule during a short hospital stay of 5-7 days, and this was initiated weeks in advance of many institutions, including nursing homes with vulnerable populations. Our admitting criteria was reviewed to allow for admission of only those patients who absolutely needed inpatient substance use disorder treatment, including patients with severe withdrawal symptoms and signs, comorbidities, or neuropsychiatric manifestations that made them unsafe for outpatient or home detoxification. Others were triaged to the outpatient services which was amply supported with telemedicine. Rooms and designated areas of the building were earmarked as places for isolation/quarantine if suspected COVID-19 cases were identified pending testing. To assess patients’ risk of COVID-19, we do point-of-care nasopharyngeal swab testing with polymerase chain reaction.
Regarding face masks, patients and staff were fitted with ones early in the process. Additionally, staff were trained on the importance of face mask use and how to ensure you have a tight seal around the mouth and nose and were provided with other appropriate personal protective equipment. Concerning social distancing, we reduced the patient population capacity for the unit down to 50% and offered only single room admissions. Social distancing was encouraged in the unit, including in the television and recreation room and dining room, and during small treatment groups of less than six individuals. Daily temperature checks with noncontact handheld thermometers were enforced for staff and anyone coming into the life recovery center.
Patients are continuously being educated on the presentations of COVID-19 and encouraged to report any symptoms. Any staff feeling sick or having symptoms are encouraged to stay home. Rigorous and continuous cleaning of surfaces, especially of areas subjected to common use, is done frequently by the hospital housekeeping and environmental crew and is the order of the day.
Dr. Fagbemi is a hospitalist at BronxCare Health System, a not-for-profit health and teaching hospital system serving South and Central Bronx in New York. He has no conflicts of interest to disclose.
Annual U.S. death toll from drugs, alcohol, suicide tops 150,000
Despite decreases in overall opioid overdose deaths in 2018, deaths involving synthetic opioids, cocaine, and other psychostimulants increased sharply in the United States, and alcohol and suicide deaths also rose, new data show.
A report released May 21 by the Trust for America’s Health (TFAH) and the Well Being Trust shows that 151,964 Americans died from alcohol, drugs, and suicide. Experts warn that these “deaths of despair” may well increase in the wake of COVID-19.
A study released earlier in May estimated that an additional 75,000 Americans could die by suicide, drugs, or alcohol abuse because of the pandemic (Petterson S et al. “Projected Deaths of Despair From COVID-19,” Well Being Trust. May 8, 2020. WellBeingTrust.org).
“These data are a clarion call to action,” TFAH President and CEO John Auerbach said in a news release.
“We know what works to address deaths of despair but progress has been uneven and death rates continue to climb, with communities of color experiencing higher rates of increases in drug-induced and alcohol deaths,” he said.
“And there’s another immediate concern: The COVID-19 crisis has increased the health burdens and economic pressures on many communities of color,” said Mr. Auerbach.
According to the report, the 2018 national rate for alcohol, drug, and suicide deaths combined was only slightly lower than that reported in 2017 (46.4 vs 46.6 per 100,000).
Among the key findings in the report:
- 37,329 Americans died from alcohol-induced causes in 2018; the rate was up 4% over 2017.
- Alcohol-induced deaths were highest among American Indians (30.0 per 100,000) and adults aged 55 to 74 (27.6 per 100,000). For all population groups, rates of alcohol-related deaths were higher in 2018 than in 2017 except for people aged 17 years and younger, for whom the rate held steady.
- Despite a 4% decline in all drug-induced deaths and a 2% drop in all opioid-related deaths, 2018 saw sharp increases in deaths involving synthetic opioids (up 10%), cocaine (up 5%), and other psychostimulants, such as methamphetamine, ecstasy, amphetamine, and prescription stimulants (up 22%).
- Suicide claimed the lives of 48,344 Americans in 2018. The suicide rate in 2018 was 2% higher than in 2017 and 25% higher than in 2008.
- Suicide rates increased across all demographics except for adults aged 18-54 years, among whom the rate remained stable. Suicide death rates were highest in males (23.4 per 100,000), rural residents (19.7 per 100,000), whites (16.8 per 100,000), and American Indian/Alaska Natives (14.1 per 100,000).
- Between 2017 and 2018, 27 states had higher rates (above 0.04%) of alcohol, drug, and suicide deaths; 23 states and the District of Columbia had lower rates of deaths from those causes.
- States with the highest alcohol, drug, and suicide death rates in 2018 were West Virginia (84.9 per 100,000), New Mexico (82.8 per 100,000), New Hampshire (68.2 per 100,000), and Alaska (67.8 per 100,000).
- States with the lowest rates in 2018 were Texas (31.7 per 100,000), Mississippi (31.7 per 100,000), and Hawaii (34.6 per 100,000).
“Quite simply, too many Americans are dying from preventable causes. The profound racial health disparities seen in these data show that many ethnic minority groups are being left behind in our response efforts,” Benjamin F. Miller, PsyD, Well Being Trust chief strategy officer, said in the release.
“The nation needs a comprehensive framework for excellence in mental health and well-being, one that intentionally provides solutions for American Indians, blacks, Asians and Latinos. said Dr. Miller.
Policy recommendations outlined in the report include investing in prevention; reducing risk factors and promoting resilience in children, families, and communities; engaging all sectors of society to address mental health and substance use disorders; limiting access to lethal means of suicide; and promoting safe storage of medications and firearms.
A version of this article originally appeared on Medscape.com.
Despite decreases in overall opioid overdose deaths in 2018, deaths involving synthetic opioids, cocaine, and other psychostimulants increased sharply in the United States, and alcohol and suicide deaths also rose, new data show.
A report released May 21 by the Trust for America’s Health (TFAH) and the Well Being Trust shows that 151,964 Americans died from alcohol, drugs, and suicide. Experts warn that these “deaths of despair” may well increase in the wake of COVID-19.
A study released earlier in May estimated that an additional 75,000 Americans could die by suicide, drugs, or alcohol abuse because of the pandemic (Petterson S et al. “Projected Deaths of Despair From COVID-19,” Well Being Trust. May 8, 2020. WellBeingTrust.org).
“These data are a clarion call to action,” TFAH President and CEO John Auerbach said in a news release.
“We know what works to address deaths of despair but progress has been uneven and death rates continue to climb, with communities of color experiencing higher rates of increases in drug-induced and alcohol deaths,” he said.
“And there’s another immediate concern: The COVID-19 crisis has increased the health burdens and economic pressures on many communities of color,” said Mr. Auerbach.
According to the report, the 2018 national rate for alcohol, drug, and suicide deaths combined was only slightly lower than that reported in 2017 (46.4 vs 46.6 per 100,000).
Among the key findings in the report:
- 37,329 Americans died from alcohol-induced causes in 2018; the rate was up 4% over 2017.
- Alcohol-induced deaths were highest among American Indians (30.0 per 100,000) and adults aged 55 to 74 (27.6 per 100,000). For all population groups, rates of alcohol-related deaths were higher in 2018 than in 2017 except for people aged 17 years and younger, for whom the rate held steady.
- Despite a 4% decline in all drug-induced deaths and a 2% drop in all opioid-related deaths, 2018 saw sharp increases in deaths involving synthetic opioids (up 10%), cocaine (up 5%), and other psychostimulants, such as methamphetamine, ecstasy, amphetamine, and prescription stimulants (up 22%).
- Suicide claimed the lives of 48,344 Americans in 2018. The suicide rate in 2018 was 2% higher than in 2017 and 25% higher than in 2008.
- Suicide rates increased across all demographics except for adults aged 18-54 years, among whom the rate remained stable. Suicide death rates were highest in males (23.4 per 100,000), rural residents (19.7 per 100,000), whites (16.8 per 100,000), and American Indian/Alaska Natives (14.1 per 100,000).
- Between 2017 and 2018, 27 states had higher rates (above 0.04%) of alcohol, drug, and suicide deaths; 23 states and the District of Columbia had lower rates of deaths from those causes.
- States with the highest alcohol, drug, and suicide death rates in 2018 were West Virginia (84.9 per 100,000), New Mexico (82.8 per 100,000), New Hampshire (68.2 per 100,000), and Alaska (67.8 per 100,000).
- States with the lowest rates in 2018 were Texas (31.7 per 100,000), Mississippi (31.7 per 100,000), and Hawaii (34.6 per 100,000).
“Quite simply, too many Americans are dying from preventable causes. The profound racial health disparities seen in these data show that many ethnic minority groups are being left behind in our response efforts,” Benjamin F. Miller, PsyD, Well Being Trust chief strategy officer, said in the release.
“The nation needs a comprehensive framework for excellence in mental health and well-being, one that intentionally provides solutions for American Indians, blacks, Asians and Latinos. said Dr. Miller.
Policy recommendations outlined in the report include investing in prevention; reducing risk factors and promoting resilience in children, families, and communities; engaging all sectors of society to address mental health and substance use disorders; limiting access to lethal means of suicide; and promoting safe storage of medications and firearms.
A version of this article originally appeared on Medscape.com.
Despite decreases in overall opioid overdose deaths in 2018, deaths involving synthetic opioids, cocaine, and other psychostimulants increased sharply in the United States, and alcohol and suicide deaths also rose, new data show.
A report released May 21 by the Trust for America’s Health (TFAH) and the Well Being Trust shows that 151,964 Americans died from alcohol, drugs, and suicide. Experts warn that these “deaths of despair” may well increase in the wake of COVID-19.
A study released earlier in May estimated that an additional 75,000 Americans could die by suicide, drugs, or alcohol abuse because of the pandemic (Petterson S et al. “Projected Deaths of Despair From COVID-19,” Well Being Trust. May 8, 2020. WellBeingTrust.org).
“These data are a clarion call to action,” TFAH President and CEO John Auerbach said in a news release.
“We know what works to address deaths of despair but progress has been uneven and death rates continue to climb, with communities of color experiencing higher rates of increases in drug-induced and alcohol deaths,” he said.
“And there’s another immediate concern: The COVID-19 crisis has increased the health burdens and economic pressures on many communities of color,” said Mr. Auerbach.
According to the report, the 2018 national rate for alcohol, drug, and suicide deaths combined was only slightly lower than that reported in 2017 (46.4 vs 46.6 per 100,000).
Among the key findings in the report:
- 37,329 Americans died from alcohol-induced causes in 2018; the rate was up 4% over 2017.
- Alcohol-induced deaths were highest among American Indians (30.0 per 100,000) and adults aged 55 to 74 (27.6 per 100,000). For all population groups, rates of alcohol-related deaths were higher in 2018 than in 2017 except for people aged 17 years and younger, for whom the rate held steady.
- Despite a 4% decline in all drug-induced deaths and a 2% drop in all opioid-related deaths, 2018 saw sharp increases in deaths involving synthetic opioids (up 10%), cocaine (up 5%), and other psychostimulants, such as methamphetamine, ecstasy, amphetamine, and prescription stimulants (up 22%).
- Suicide claimed the lives of 48,344 Americans in 2018. The suicide rate in 2018 was 2% higher than in 2017 and 25% higher than in 2008.
- Suicide rates increased across all demographics except for adults aged 18-54 years, among whom the rate remained stable. Suicide death rates were highest in males (23.4 per 100,000), rural residents (19.7 per 100,000), whites (16.8 per 100,000), and American Indian/Alaska Natives (14.1 per 100,000).
- Between 2017 and 2018, 27 states had higher rates (above 0.04%) of alcohol, drug, and suicide deaths; 23 states and the District of Columbia had lower rates of deaths from those causes.
- States with the highest alcohol, drug, and suicide death rates in 2018 were West Virginia (84.9 per 100,000), New Mexico (82.8 per 100,000), New Hampshire (68.2 per 100,000), and Alaska (67.8 per 100,000).
- States with the lowest rates in 2018 were Texas (31.7 per 100,000), Mississippi (31.7 per 100,000), and Hawaii (34.6 per 100,000).
“Quite simply, too many Americans are dying from preventable causes. The profound racial health disparities seen in these data show that many ethnic minority groups are being left behind in our response efforts,” Benjamin F. Miller, PsyD, Well Being Trust chief strategy officer, said in the release.
“The nation needs a comprehensive framework for excellence in mental health and well-being, one that intentionally provides solutions for American Indians, blacks, Asians and Latinos. said Dr. Miller.
Policy recommendations outlined in the report include investing in prevention; reducing risk factors and promoting resilience in children, families, and communities; engaging all sectors of society to address mental health and substance use disorders; limiting access to lethal means of suicide; and promoting safe storage of medications and firearms.
A version of this article originally appeared on Medscape.com.
New ‘atlas’ maps links between mental disorders, physical illnesses
Mental illnesses are associated with a significantly increased risk of subsequent physical diseases, new research shows.
An international team of researchers has created an “atlas” that maps the relationship between specific mental disorders and the risk of subsequent physical illnesses.
The researchers found that, following the diagnosis of a mental disorder, psychiatric patients are significantly more likely than the general population to develop potentially life-threatening conditions, including heart disease and stroke.
These findings, the investigators noted, highlight the need for better medical care in this vulnerable population. They have created a website with detailed information about the risks of specific physical ailments and the link to particular mental disorders.
“We found that women with anxiety disorders have a 50% increased risk of developing a heart condition or stroke – over 15 years, one in three women with anxiety disorders will develop these medical disorders,” lead investigator John McGrath, MD, PhD, University of Queensland’s Brain Institute, Brisbane, Australia, and Aarhus (Denmark) University, said in a statement.
“We also looked at men with substance use disorders such as alcohol-related disorders and found they have a 400% increased risk of gut or liver disorders, while over 15 years, one in five of them will develop gut or liver conditions,” he added.
The study was published in the New England Journal of Medicine.
New ‘atlas’
It’s well known that patients with mental disorders have decreased quality of life, increased health care utilization, and a shorter life expectancy than individuals in the general population – about 10 years for men and 7 years for women.
However, the investigators noted, previous research examining the relationship between mental disorders and medical conditions only focused on “particular pairs or a small set of mental disorders and medical conditions.”
“We needed a comprehensive study to map the links between different types of mental disorders versus different types of general medical conditions. Our study has provided this atlas,” Dr. McGrath said in an interview.
The clinical utility of such a map could provide comprehensive data on relative and absolute risks of various medical conditions after a diagnosis of a mental disorder. This information, the researchers noted, would “help clinicians and health care planners identify the primary prevention needs of their patients.”
The study included 5.9 million people born in Denmark between 1900 and 2015 and followed them from 2000 to 2016, a total of 83.9 million person-years. The researchers followed patients for up to 17 years (2000-2016) for medical diagnoses and up to 48 years (1969-2016) for diagnoses of mental disorders.
The study’s large sample size allowed investigators to assess 10 broad types of mental disorders and 9 broad categories of medical conditions that encompassed 31 specific conditions.
Categories of medical conditions included circulatory, endocrine, pulmonary, gastrointestinal, urogenital, musculoskeletal, hematologic, neurologic, and cancer. Mental disorder categories included organic disorders such as Alzheimer’s, substance abuse disorders, schizophrenia, mood disorders, neurotic disorders, eating disorders, personality disorders, developmental disorders, behavioral/emotional disorders, and intellectual disabilities.
The researchers estimated associations between 90 pairs of mental disorders and broad-category medical conditions, as well as 310 pairs of mental disorders and specific medical conditions.
‘Curious’ finding
Individuals with mental disorders showed a higher risk of medical conditions in 76 out of 90 specific mental disorder–medical condition pairs.
After adjusting for sex, age, calendar time, and previous coexisting mental disorders, the median hazard ratio for a subsequent medical condition was 1.37 in patients with a mental disorder.
The lowest HR was 0.82 for organic mental disorders and the broad category of cancer (95% confidence interval, 0.80-0.84), and the highest was 3.62 for eating disorders and urogenital conditions (95% CI, 3.11-4.22). On the other hand, schizophrenia was associated with a reduced risk of developing musculoskeletal conditions (HR, 0.87; 95% CI, 0.84-0.91).
Dr. McGrath described this finding as “curious” and speculated it “may be related to underlying genetic risk factors.”
compared with the matched reference group without a mood disorder (40.9% vs. 32.6%, respectively).
The risk of developing subsequent medical conditions after a mental disorder diagnosis did not remain steady over time. For instance, although mood disorders were associated with an increased risk of developing circulatory problems (HR, 1.32; 95% CI, 1.31-1.34), the highest risk occurred during the first 6 months following diagnosis and gradually decreased over the next 15 years (HR, 2.39; 95% CI, 2.29-2.48 and HR, 1.18; 95% CI, 1.17-1.20, respectively).
“Many people with mental disorders have unhealthy lifestyle, including low exercise, poor diet, smoking, and alcohol, which may account for the increased risk of physical illness, and also they may not seek and/or may not get quick treatment for their health conditions,” said Dr. McGrath.
Additionally, “perhaps some genetic and early life exposures, such as trauma, may increase the risk of both medical conditions and mental disorders,” he added. “We need better treatments for mental disorders, so that they do not slip into unemployment or poverty.”
A strong case
In a comment, Roger McIntyre, MD, professor of psychiatry and pharmacology at the University of Toronto and head of the mood disorders psychopharmacology unit, University Health Network, said that the research “really makes a strong case for the fact that persons who have mental disorders are at higher risk of chronic diseases, and it’s the chronic diseases that decrease their lifespan.”
Dr. McIntyre, who is also director of the Depression and Bipolar Support Alliance, said that the “takeaway message is that mental disorders are not just brain disorders but are multisystem disorders.”
For this reason, “the most appropriate way to provide care would be to provide a holistic approach to treat and prevent the chronic diseases that lead to increase in mortality,” recommended Dr. McIntyre, who was not involved with the current study.
The study was supported by grants from the Danish National Research Foundation, the National Health and Medical Research Council, the Novo Nordisk Foundation , the European Union’s Horizon 2020 Research and Innovation Program, the Aarhus University Research Foundation, the Lundbeck Foundation, the National Institutes of Health, the European Commission, Helsefonden, the Danish Council for Independent Research, the Independent Research Fund Denmark, the National Health and Medical Research Council of Australia, and the National Institute on Drug Abuse.
Dr. McGrath has disclosed no relevant financial relationships. The other authors’ disclosures are listed on the original paper. Dr. McIntyre reports receiving grants from Stanley Medical Research Institute; the Canadian Institutes of Health Research/Global Alliance for Chronic Diseases/Chinese National Natural Research Foundation; and receiving speaking/consultation fees from Lundbeck, Janssen, Shire, Purdue, Pfizer, Otsuka, Allergan, Takeda, Neurocrine, Sunovion, and Minerva.
A version of this article originally appeared on Medscape.com.
Mental illnesses are associated with a significantly increased risk of subsequent physical diseases, new research shows.
An international team of researchers has created an “atlas” that maps the relationship between specific mental disorders and the risk of subsequent physical illnesses.
The researchers found that, following the diagnosis of a mental disorder, psychiatric patients are significantly more likely than the general population to develop potentially life-threatening conditions, including heart disease and stroke.
These findings, the investigators noted, highlight the need for better medical care in this vulnerable population. They have created a website with detailed information about the risks of specific physical ailments and the link to particular mental disorders.
“We found that women with anxiety disorders have a 50% increased risk of developing a heart condition or stroke – over 15 years, one in three women with anxiety disorders will develop these medical disorders,” lead investigator John McGrath, MD, PhD, University of Queensland’s Brain Institute, Brisbane, Australia, and Aarhus (Denmark) University, said in a statement.
“We also looked at men with substance use disorders such as alcohol-related disorders and found they have a 400% increased risk of gut or liver disorders, while over 15 years, one in five of them will develop gut or liver conditions,” he added.
The study was published in the New England Journal of Medicine.
New ‘atlas’
It’s well known that patients with mental disorders have decreased quality of life, increased health care utilization, and a shorter life expectancy than individuals in the general population – about 10 years for men and 7 years for women.
However, the investigators noted, previous research examining the relationship between mental disorders and medical conditions only focused on “particular pairs or a small set of mental disorders and medical conditions.”
“We needed a comprehensive study to map the links between different types of mental disorders versus different types of general medical conditions. Our study has provided this atlas,” Dr. McGrath said in an interview.
The clinical utility of such a map could provide comprehensive data on relative and absolute risks of various medical conditions after a diagnosis of a mental disorder. This information, the researchers noted, would “help clinicians and health care planners identify the primary prevention needs of their patients.”
The study included 5.9 million people born in Denmark between 1900 and 2015 and followed them from 2000 to 2016, a total of 83.9 million person-years. The researchers followed patients for up to 17 years (2000-2016) for medical diagnoses and up to 48 years (1969-2016) for diagnoses of mental disorders.
The study’s large sample size allowed investigators to assess 10 broad types of mental disorders and 9 broad categories of medical conditions that encompassed 31 specific conditions.
Categories of medical conditions included circulatory, endocrine, pulmonary, gastrointestinal, urogenital, musculoskeletal, hematologic, neurologic, and cancer. Mental disorder categories included organic disorders such as Alzheimer’s, substance abuse disorders, schizophrenia, mood disorders, neurotic disorders, eating disorders, personality disorders, developmental disorders, behavioral/emotional disorders, and intellectual disabilities.
The researchers estimated associations between 90 pairs of mental disorders and broad-category medical conditions, as well as 310 pairs of mental disorders and specific medical conditions.
‘Curious’ finding
Individuals with mental disorders showed a higher risk of medical conditions in 76 out of 90 specific mental disorder–medical condition pairs.
After adjusting for sex, age, calendar time, and previous coexisting mental disorders, the median hazard ratio for a subsequent medical condition was 1.37 in patients with a mental disorder.
The lowest HR was 0.82 for organic mental disorders and the broad category of cancer (95% confidence interval, 0.80-0.84), and the highest was 3.62 for eating disorders and urogenital conditions (95% CI, 3.11-4.22). On the other hand, schizophrenia was associated with a reduced risk of developing musculoskeletal conditions (HR, 0.87; 95% CI, 0.84-0.91).
Dr. McGrath described this finding as “curious” and speculated it “may be related to underlying genetic risk factors.”
compared with the matched reference group without a mood disorder (40.9% vs. 32.6%, respectively).
The risk of developing subsequent medical conditions after a mental disorder diagnosis did not remain steady over time. For instance, although mood disorders were associated with an increased risk of developing circulatory problems (HR, 1.32; 95% CI, 1.31-1.34), the highest risk occurred during the first 6 months following diagnosis and gradually decreased over the next 15 years (HR, 2.39; 95% CI, 2.29-2.48 and HR, 1.18; 95% CI, 1.17-1.20, respectively).
“Many people with mental disorders have unhealthy lifestyle, including low exercise, poor diet, smoking, and alcohol, which may account for the increased risk of physical illness, and also they may not seek and/or may not get quick treatment for their health conditions,” said Dr. McGrath.
Additionally, “perhaps some genetic and early life exposures, such as trauma, may increase the risk of both medical conditions and mental disorders,” he added. “We need better treatments for mental disorders, so that they do not slip into unemployment or poverty.”
A strong case
In a comment, Roger McIntyre, MD, professor of psychiatry and pharmacology at the University of Toronto and head of the mood disorders psychopharmacology unit, University Health Network, said that the research “really makes a strong case for the fact that persons who have mental disorders are at higher risk of chronic diseases, and it’s the chronic diseases that decrease their lifespan.”
Dr. McIntyre, who is also director of the Depression and Bipolar Support Alliance, said that the “takeaway message is that mental disorders are not just brain disorders but are multisystem disorders.”
For this reason, “the most appropriate way to provide care would be to provide a holistic approach to treat and prevent the chronic diseases that lead to increase in mortality,” recommended Dr. McIntyre, who was not involved with the current study.
The study was supported by grants from the Danish National Research Foundation, the National Health and Medical Research Council, the Novo Nordisk Foundation , the European Union’s Horizon 2020 Research and Innovation Program, the Aarhus University Research Foundation, the Lundbeck Foundation, the National Institutes of Health, the European Commission, Helsefonden, the Danish Council for Independent Research, the Independent Research Fund Denmark, the National Health and Medical Research Council of Australia, and the National Institute on Drug Abuse.
Dr. McGrath has disclosed no relevant financial relationships. The other authors’ disclosures are listed on the original paper. Dr. McIntyre reports receiving grants from Stanley Medical Research Institute; the Canadian Institutes of Health Research/Global Alliance for Chronic Diseases/Chinese National Natural Research Foundation; and receiving speaking/consultation fees from Lundbeck, Janssen, Shire, Purdue, Pfizer, Otsuka, Allergan, Takeda, Neurocrine, Sunovion, and Minerva.
A version of this article originally appeared on Medscape.com.
Mental illnesses are associated with a significantly increased risk of subsequent physical diseases, new research shows.
An international team of researchers has created an “atlas” that maps the relationship between specific mental disorders and the risk of subsequent physical illnesses.
The researchers found that, following the diagnosis of a mental disorder, psychiatric patients are significantly more likely than the general population to develop potentially life-threatening conditions, including heart disease and stroke.
These findings, the investigators noted, highlight the need for better medical care in this vulnerable population. They have created a website with detailed information about the risks of specific physical ailments and the link to particular mental disorders.
“We found that women with anxiety disorders have a 50% increased risk of developing a heart condition or stroke – over 15 years, one in three women with anxiety disorders will develop these medical disorders,” lead investigator John McGrath, MD, PhD, University of Queensland’s Brain Institute, Brisbane, Australia, and Aarhus (Denmark) University, said in a statement.
“We also looked at men with substance use disorders such as alcohol-related disorders and found they have a 400% increased risk of gut or liver disorders, while over 15 years, one in five of them will develop gut or liver conditions,” he added.
The study was published in the New England Journal of Medicine.
New ‘atlas’
It’s well known that patients with mental disorders have decreased quality of life, increased health care utilization, and a shorter life expectancy than individuals in the general population – about 10 years for men and 7 years for women.
However, the investigators noted, previous research examining the relationship between mental disorders and medical conditions only focused on “particular pairs or a small set of mental disorders and medical conditions.”
“We needed a comprehensive study to map the links between different types of mental disorders versus different types of general medical conditions. Our study has provided this atlas,” Dr. McGrath said in an interview.
The clinical utility of such a map could provide comprehensive data on relative and absolute risks of various medical conditions after a diagnosis of a mental disorder. This information, the researchers noted, would “help clinicians and health care planners identify the primary prevention needs of their patients.”
The study included 5.9 million people born in Denmark between 1900 and 2015 and followed them from 2000 to 2016, a total of 83.9 million person-years. The researchers followed patients for up to 17 years (2000-2016) for medical diagnoses and up to 48 years (1969-2016) for diagnoses of mental disorders.
The study’s large sample size allowed investigators to assess 10 broad types of mental disorders and 9 broad categories of medical conditions that encompassed 31 specific conditions.
Categories of medical conditions included circulatory, endocrine, pulmonary, gastrointestinal, urogenital, musculoskeletal, hematologic, neurologic, and cancer. Mental disorder categories included organic disorders such as Alzheimer’s, substance abuse disorders, schizophrenia, mood disorders, neurotic disorders, eating disorders, personality disorders, developmental disorders, behavioral/emotional disorders, and intellectual disabilities.
The researchers estimated associations between 90 pairs of mental disorders and broad-category medical conditions, as well as 310 pairs of mental disorders and specific medical conditions.
‘Curious’ finding
Individuals with mental disorders showed a higher risk of medical conditions in 76 out of 90 specific mental disorder–medical condition pairs.
After adjusting for sex, age, calendar time, and previous coexisting mental disorders, the median hazard ratio for a subsequent medical condition was 1.37 in patients with a mental disorder.
The lowest HR was 0.82 for organic mental disorders and the broad category of cancer (95% confidence interval, 0.80-0.84), and the highest was 3.62 for eating disorders and urogenital conditions (95% CI, 3.11-4.22). On the other hand, schizophrenia was associated with a reduced risk of developing musculoskeletal conditions (HR, 0.87; 95% CI, 0.84-0.91).
Dr. McGrath described this finding as “curious” and speculated it “may be related to underlying genetic risk factors.”
compared with the matched reference group without a mood disorder (40.9% vs. 32.6%, respectively).
The risk of developing subsequent medical conditions after a mental disorder diagnosis did not remain steady over time. For instance, although mood disorders were associated with an increased risk of developing circulatory problems (HR, 1.32; 95% CI, 1.31-1.34), the highest risk occurred during the first 6 months following diagnosis and gradually decreased over the next 15 years (HR, 2.39; 95% CI, 2.29-2.48 and HR, 1.18; 95% CI, 1.17-1.20, respectively).
“Many people with mental disorders have unhealthy lifestyle, including low exercise, poor diet, smoking, and alcohol, which may account for the increased risk of physical illness, and also they may not seek and/or may not get quick treatment for their health conditions,” said Dr. McGrath.
Additionally, “perhaps some genetic and early life exposures, such as trauma, may increase the risk of both medical conditions and mental disorders,” he added. “We need better treatments for mental disorders, so that they do not slip into unemployment or poverty.”
A strong case
In a comment, Roger McIntyre, MD, professor of psychiatry and pharmacology at the University of Toronto and head of the mood disorders psychopharmacology unit, University Health Network, said that the research “really makes a strong case for the fact that persons who have mental disorders are at higher risk of chronic diseases, and it’s the chronic diseases that decrease their lifespan.”
Dr. McIntyre, who is also director of the Depression and Bipolar Support Alliance, said that the “takeaway message is that mental disorders are not just brain disorders but are multisystem disorders.”
For this reason, “the most appropriate way to provide care would be to provide a holistic approach to treat and prevent the chronic diseases that lead to increase in mortality,” recommended Dr. McIntyre, who was not involved with the current study.
The study was supported by grants from the Danish National Research Foundation, the National Health and Medical Research Council, the Novo Nordisk Foundation , the European Union’s Horizon 2020 Research and Innovation Program, the Aarhus University Research Foundation, the Lundbeck Foundation, the National Institutes of Health, the European Commission, Helsefonden, the Danish Council for Independent Research, the Independent Research Fund Denmark, the National Health and Medical Research Council of Australia, and the National Institute on Drug Abuse.
Dr. McGrath has disclosed no relevant financial relationships. The other authors’ disclosures are listed on the original paper. Dr. McIntyre reports receiving grants from Stanley Medical Research Institute; the Canadian Institutes of Health Research/Global Alliance for Chronic Diseases/Chinese National Natural Research Foundation; and receiving speaking/consultation fees from Lundbeck, Janssen, Shire, Purdue, Pfizer, Otsuka, Allergan, Takeda, Neurocrine, Sunovion, and Minerva.
A version of this article originally appeared on Medscape.com.
COVID-19: An opportunity, challenge for addiction treatment, NIDA boss says
The COVID-19 pandemic is posing significant challenges while also providing unique opportunities for patients with substance use disorders (SUD), a leading expert says.
Nora Volkow, MD, director of the National Institute on Drug Abuse, said that the pandemic has accelerated the use of telemedicine, making it easier for patients with SUD to access treatment. It has also led to the proliferation of more mental health hotlines, which is critical since the vast majority of these patients have comorbid mental illness.
In addition, COVID-19 has resulted in increased availability of “alternative” peer support mechanisms via cellphones or computers to aid individuals’ sobriety.
Dr. Volkow spoke at the virtual American Psychiatric Association Spring Highlights Meeting 2020, which is replacing the organization’s canceled annual meeting.
While methadone clinics have had to close during the pandemic, making it challenging for those on medically assisted treatment to receive methadone or buprenorphine, some of the rules and regulations have been relaxed in order to make these medications accessible without the need for in-person attendance at a clinic. In addition, the Substance Abuse and Mental Health Services Administration has relaxed some of its own regulations regarding telehealth and opioid treatment programs.
Social isolation, stigma intensified
A pandemic increases anxiety in the general population, but for patients with SUD who may be also be struggling with homelessness and comorbid mental illness, the situation can further exacerbate social stigma and isolation – leading to relapse, more overdoses, and overdose deaths, Dr. Volkow said. Social interaction is “extraordinarily important” for patients and “one of the most powerful tools we have” to build resilience.
Right now, said Dr. Volkow, “we are in the dark as to how COVID infections have affected the number of overdose deaths.”
However, she noted that
“So even through this devastation, we can actually extract something that may help others in future,” she said.
Dr. Volkow noted that during the pandemic it is critical to reinforce the importance of engaging in – and remaining in – treatment to SUD patients. It’s also crucial to make patients aware of social support systems and behavioral interventions to help them cope with stress and to mitigate relapse risk.
COVID-19 and relapse
Elie G. Aoun, MD, assistant professor of psychiatry at New York University and vice chair of the APA’s Council on Addiction Psychiatry, said in an interview that Dr. Volkow’s presentation provided “exactly the kind of accessible information” clinicians need.
Dr. Aoun said he sees the impact of the COVID-19 crisis in his practice every day. Patients with SUD “are getting the short end of the stick.”
Social distancing measures prompted by the pandemic can be “very triggering” for SUD patients, he said. One of his patients told him the current isolation, loneliness, movement restrictions, and boredom remind her of the way she felt when she used drugs.
Dr. Aoun said four of his patients have relapsed since the pandemic began. Two of them had just started treatment after years of using drugs, so this was a “major setback” for them.
He and his colleagues were “not really prepared” to provide care via video link, which he believes is not as effective as in-person sessions.
In addition to disrupting patient care, said Dr. Aoun, the pandemic is forcing the medical community to face social determinants of health, such as poverty and homelessness, as they relate to addiction disorders and whether or not someone receives care.
This article originally appeared on Medscape.com.
The COVID-19 pandemic is posing significant challenges while also providing unique opportunities for patients with substance use disorders (SUD), a leading expert says.
Nora Volkow, MD, director of the National Institute on Drug Abuse, said that the pandemic has accelerated the use of telemedicine, making it easier for patients with SUD to access treatment. It has also led to the proliferation of more mental health hotlines, which is critical since the vast majority of these patients have comorbid mental illness.
In addition, COVID-19 has resulted in increased availability of “alternative” peer support mechanisms via cellphones or computers to aid individuals’ sobriety.
Dr. Volkow spoke at the virtual American Psychiatric Association Spring Highlights Meeting 2020, which is replacing the organization’s canceled annual meeting.
While methadone clinics have had to close during the pandemic, making it challenging for those on medically assisted treatment to receive methadone or buprenorphine, some of the rules and regulations have been relaxed in order to make these medications accessible without the need for in-person attendance at a clinic. In addition, the Substance Abuse and Mental Health Services Administration has relaxed some of its own regulations regarding telehealth and opioid treatment programs.
Social isolation, stigma intensified
A pandemic increases anxiety in the general population, but for patients with SUD who may be also be struggling with homelessness and comorbid mental illness, the situation can further exacerbate social stigma and isolation – leading to relapse, more overdoses, and overdose deaths, Dr. Volkow said. Social interaction is “extraordinarily important” for patients and “one of the most powerful tools we have” to build resilience.
Right now, said Dr. Volkow, “we are in the dark as to how COVID infections have affected the number of overdose deaths.”
However, she noted that
“So even through this devastation, we can actually extract something that may help others in future,” she said.
Dr. Volkow noted that during the pandemic it is critical to reinforce the importance of engaging in – and remaining in – treatment to SUD patients. It’s also crucial to make patients aware of social support systems and behavioral interventions to help them cope with stress and to mitigate relapse risk.
COVID-19 and relapse
Elie G. Aoun, MD, assistant professor of psychiatry at New York University and vice chair of the APA’s Council on Addiction Psychiatry, said in an interview that Dr. Volkow’s presentation provided “exactly the kind of accessible information” clinicians need.
Dr. Aoun said he sees the impact of the COVID-19 crisis in his practice every day. Patients with SUD “are getting the short end of the stick.”
Social distancing measures prompted by the pandemic can be “very triggering” for SUD patients, he said. One of his patients told him the current isolation, loneliness, movement restrictions, and boredom remind her of the way she felt when she used drugs.
Dr. Aoun said four of his patients have relapsed since the pandemic began. Two of them had just started treatment after years of using drugs, so this was a “major setback” for them.
He and his colleagues were “not really prepared” to provide care via video link, which he believes is not as effective as in-person sessions.
In addition to disrupting patient care, said Dr. Aoun, the pandemic is forcing the medical community to face social determinants of health, such as poverty and homelessness, as they relate to addiction disorders and whether or not someone receives care.
This article originally appeared on Medscape.com.
The COVID-19 pandemic is posing significant challenges while also providing unique opportunities for patients with substance use disorders (SUD), a leading expert says.
Nora Volkow, MD, director of the National Institute on Drug Abuse, said that the pandemic has accelerated the use of telemedicine, making it easier for patients with SUD to access treatment. It has also led to the proliferation of more mental health hotlines, which is critical since the vast majority of these patients have comorbid mental illness.
In addition, COVID-19 has resulted in increased availability of “alternative” peer support mechanisms via cellphones or computers to aid individuals’ sobriety.
Dr. Volkow spoke at the virtual American Psychiatric Association Spring Highlights Meeting 2020, which is replacing the organization’s canceled annual meeting.
While methadone clinics have had to close during the pandemic, making it challenging for those on medically assisted treatment to receive methadone or buprenorphine, some of the rules and regulations have been relaxed in order to make these medications accessible without the need for in-person attendance at a clinic. In addition, the Substance Abuse and Mental Health Services Administration has relaxed some of its own regulations regarding telehealth and opioid treatment programs.
Social isolation, stigma intensified
A pandemic increases anxiety in the general population, but for patients with SUD who may be also be struggling with homelessness and comorbid mental illness, the situation can further exacerbate social stigma and isolation – leading to relapse, more overdoses, and overdose deaths, Dr. Volkow said. Social interaction is “extraordinarily important” for patients and “one of the most powerful tools we have” to build resilience.
Right now, said Dr. Volkow, “we are in the dark as to how COVID infections have affected the number of overdose deaths.”
However, she noted that
“So even through this devastation, we can actually extract something that may help others in future,” she said.
Dr. Volkow noted that during the pandemic it is critical to reinforce the importance of engaging in – and remaining in – treatment to SUD patients. It’s also crucial to make patients aware of social support systems and behavioral interventions to help them cope with stress and to mitigate relapse risk.
COVID-19 and relapse
Elie G. Aoun, MD, assistant professor of psychiatry at New York University and vice chair of the APA’s Council on Addiction Psychiatry, said in an interview that Dr. Volkow’s presentation provided “exactly the kind of accessible information” clinicians need.
Dr. Aoun said he sees the impact of the COVID-19 crisis in his practice every day. Patients with SUD “are getting the short end of the stick.”
Social distancing measures prompted by the pandemic can be “very triggering” for SUD patients, he said. One of his patients told him the current isolation, loneliness, movement restrictions, and boredom remind her of the way she felt when she used drugs.
Dr. Aoun said four of his patients have relapsed since the pandemic began. Two of them had just started treatment after years of using drugs, so this was a “major setback” for them.
He and his colleagues were “not really prepared” to provide care via video link, which he believes is not as effective as in-person sessions.
In addition to disrupting patient care, said Dr. Aoun, the pandemic is forcing the medical community to face social determinants of health, such as poverty and homelessness, as they relate to addiction disorders and whether or not someone receives care.
This article originally appeared on Medscape.com.
Elderly Americans carry heavier opioid burden
according to the Agency for Healthcare Quality and Research.
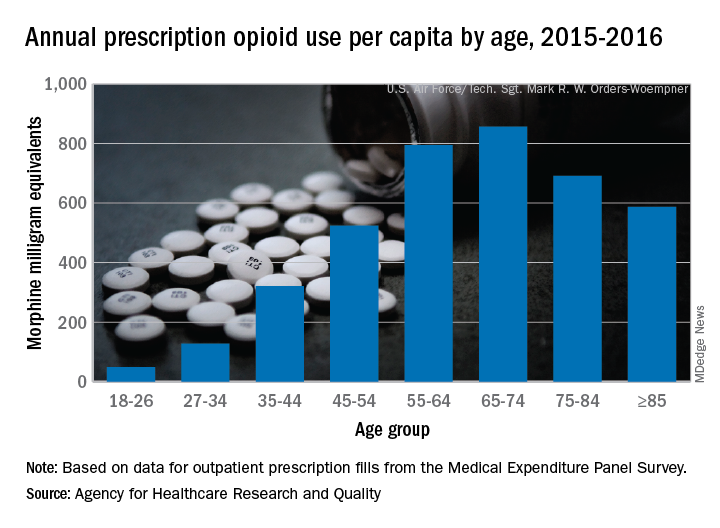
Elderly adults with chronic and acute pain obtained an average of 774 morphine milligram equivalents (MMEs) of prescription opioids annually during 2015-2016 from outpatient clinicians, compared with 376 MMEs a year for nonelderly adults, said Asako S. Moriya, PhD, and G. Edward Miller, PhD, of the AHRQ.
Narrowing the age groups shows that opioid MMEs increased with age, starting at 49 MMEs for 18- to 26-year-olds and rising to a high of 856 MMEs in the 65- to 74-year-old group, before dropping off in the oldest adults, the investigators said in a Medical Expenditure Panel Survey (MEPS) research findings report.
The analysis included “all opioid medications that are commonly used to treat pain” and excluded respiratory agents, antitussives, and drugs used for medication-assisted treatment, they noted. The MEPS data cover prescriptions purchased or obtained in outpatient settings but not those administered in inpatient settings or in clinics or physician offices.
according to the Agency for Healthcare Quality and Research.

Elderly adults with chronic and acute pain obtained an average of 774 morphine milligram equivalents (MMEs) of prescription opioids annually during 2015-2016 from outpatient clinicians, compared with 376 MMEs a year for nonelderly adults, said Asako S. Moriya, PhD, and G. Edward Miller, PhD, of the AHRQ.
Narrowing the age groups shows that opioid MMEs increased with age, starting at 49 MMEs for 18- to 26-year-olds and rising to a high of 856 MMEs in the 65- to 74-year-old group, before dropping off in the oldest adults, the investigators said in a Medical Expenditure Panel Survey (MEPS) research findings report.
The analysis included “all opioid medications that are commonly used to treat pain” and excluded respiratory agents, antitussives, and drugs used for medication-assisted treatment, they noted. The MEPS data cover prescriptions purchased or obtained in outpatient settings but not those administered in inpatient settings or in clinics or physician offices.
according to the Agency for Healthcare Quality and Research.

Elderly adults with chronic and acute pain obtained an average of 774 morphine milligram equivalents (MMEs) of prescription opioids annually during 2015-2016 from outpatient clinicians, compared with 376 MMEs a year for nonelderly adults, said Asako S. Moriya, PhD, and G. Edward Miller, PhD, of the AHRQ.
Narrowing the age groups shows that opioid MMEs increased with age, starting at 49 MMEs for 18- to 26-year-olds and rising to a high of 856 MMEs in the 65- to 74-year-old group, before dropping off in the oldest adults, the investigators said in a Medical Expenditure Panel Survey (MEPS) research findings report.
The analysis included “all opioid medications that are commonly used to treat pain” and excluded respiratory agents, antitussives, and drugs used for medication-assisted treatment, they noted. The MEPS data cover prescriptions purchased or obtained in outpatient settings but not those administered in inpatient settings or in clinics or physician offices.
New ASAM guideline released amid COVID-19 concerns
Home-based buprenorphine induction deemed safe for OUD
The American Society of Addiction Medicine has released an updated practice guideline for patients with opioid use disorder.
The guideline, called a focused update, advances ASAM’s 2015 National Practice Guidelines for the Treament of Opioid Use Disorder. “During the ongoing COVID-19 pandemic and the associated need for social distancing, it is especially important that clinicians and health care providers across the country take steps to ensure that individuals with OUD can continue to receive evidence-based care,” said Paul H. Earley, MD, president of ASAM, in a press release announcing the new guideline.
The guideline specifies that home-based buprenorphine induction is safe and effective for treatment of opioid use disorder and that no individual entering the criminal justice system should be subjected to opioid withdrawal.
“The research is clear, providing methadone or buprenorphine, even without psychosocial treatment, reduces the patient’s risk of death,” said Kyle Kampman, MD, chair of the group’s Guideline Writing Committee, in the release. “Ultimately, keeping patients with the disease of addiction alive and engaged to become ready for recovery is absolutely critical in the context of the deadly overdose epidemic that has struck communities across our country.”
The society released this focused update to reflect new medications and formulations, published evidence, and clinical guidance related to treatment of OUD. This update includes the addition of 13 new recommendations and major revisions to 35 existing recommendations. One concern the society has is how to help patients being treated for OUD who are limited in their ability to leave their homes. Because of these same concerns, the Substance Abuse and Mental Health Services Administration relaxed regulations on March 16 regarding patient eligibility for take-home medications, such as buprenorphine and methadone, which dovetails with the society’s guidance regarding home-based induction.
, continuing on to pharmacologic treatment even if the patient declines recommended psychosocial treatment, keeping naloxone kits available in correctional facilities, and more. Additional information about this update can be found on ASAM’s website.
Home-based buprenorphine induction deemed safe for OUD
Home-based buprenorphine induction deemed safe for OUD
The American Society of Addiction Medicine has released an updated practice guideline for patients with opioid use disorder.
The guideline, called a focused update, advances ASAM’s 2015 National Practice Guidelines for the Treament of Opioid Use Disorder. “During the ongoing COVID-19 pandemic and the associated need for social distancing, it is especially important that clinicians and health care providers across the country take steps to ensure that individuals with OUD can continue to receive evidence-based care,” said Paul H. Earley, MD, president of ASAM, in a press release announcing the new guideline.
The guideline specifies that home-based buprenorphine induction is safe and effective for treatment of opioid use disorder and that no individual entering the criminal justice system should be subjected to opioid withdrawal.
“The research is clear, providing methadone or buprenorphine, even without psychosocial treatment, reduces the patient’s risk of death,” said Kyle Kampman, MD, chair of the group’s Guideline Writing Committee, in the release. “Ultimately, keeping patients with the disease of addiction alive and engaged to become ready for recovery is absolutely critical in the context of the deadly overdose epidemic that has struck communities across our country.”
The society released this focused update to reflect new medications and formulations, published evidence, and clinical guidance related to treatment of OUD. This update includes the addition of 13 new recommendations and major revisions to 35 existing recommendations. One concern the society has is how to help patients being treated for OUD who are limited in their ability to leave their homes. Because of these same concerns, the Substance Abuse and Mental Health Services Administration relaxed regulations on March 16 regarding patient eligibility for take-home medications, such as buprenorphine and methadone, which dovetails with the society’s guidance regarding home-based induction.
, continuing on to pharmacologic treatment even if the patient declines recommended psychosocial treatment, keeping naloxone kits available in correctional facilities, and more. Additional information about this update can be found on ASAM’s website.
The American Society of Addiction Medicine has released an updated practice guideline for patients with opioid use disorder.
The guideline, called a focused update, advances ASAM’s 2015 National Practice Guidelines for the Treament of Opioid Use Disorder. “During the ongoing COVID-19 pandemic and the associated need for social distancing, it is especially important that clinicians and health care providers across the country take steps to ensure that individuals with OUD can continue to receive evidence-based care,” said Paul H. Earley, MD, president of ASAM, in a press release announcing the new guideline.
The guideline specifies that home-based buprenorphine induction is safe and effective for treatment of opioid use disorder and that no individual entering the criminal justice system should be subjected to opioid withdrawal.
“The research is clear, providing methadone or buprenorphine, even without psychosocial treatment, reduces the patient’s risk of death,” said Kyle Kampman, MD, chair of the group’s Guideline Writing Committee, in the release. “Ultimately, keeping patients with the disease of addiction alive and engaged to become ready for recovery is absolutely critical in the context of the deadly overdose epidemic that has struck communities across our country.”
The society released this focused update to reflect new medications and formulations, published evidence, and clinical guidance related to treatment of OUD. This update includes the addition of 13 new recommendations and major revisions to 35 existing recommendations. One concern the society has is how to help patients being treated for OUD who are limited in their ability to leave their homes. Because of these same concerns, the Substance Abuse and Mental Health Services Administration relaxed regulations on March 16 regarding patient eligibility for take-home medications, such as buprenorphine and methadone, which dovetails with the society’s guidance regarding home-based induction.
, continuing on to pharmacologic treatment even if the patient declines recommended psychosocial treatment, keeping naloxone kits available in correctional facilities, and more. Additional information about this update can be found on ASAM’s website.
COVID-19 prompts ‘lifesaving’ policy change for opioid addiction
In the face of the US COVID-19 pandemic, the US Substance Abuse and Mental Health Services Administration (SAMHSA) has announced policy changes to allow some patients in opioid treatment programs (OTP) to take home their medication.
According to the agency, states may request “blanket exceptions” for all stable patients in an OTP to receive a 28-day supply of take-home doses of medications such as methadone and buprenorphine, which are used to treat opioid use disorder (OUD).
States may request up to 14 days of take-home medication for patients who are less stable but who can, in the judgment of OTP clinicians, safely handle this level of take-home medication.
“SAMHSA recognizes the evolving issues surrounding COVID-19 and the emerging needs OTPs continue to face,” the agency writes in its updated guidance.
“SAMHSA affirms its commitment to supporting OTPs in any way possible during this time. As such, we are expanding our previous guidance to provide increased flexibility,” the agency said.
A ‘Lifesaving’ Decision
Commenting on the SAMHSA policy change, Richard Saitz, MD, professor and chair of the department of community health sciences, Boston University School of Public Health, said, the policy “is not only a good idea, it is critical and lifesaving.”
“This approach had to be done now. With the reduction in face-to-face visits, patients with opioid use disorder need a way to access treatment. If they cannot get opioid agonists, they would withdraw and return to illicit opioid use and high overdose risk and it would be cruel,” said Saitz.
“It is possible that there will be some diversion and some risk of overdose or misuse, but even for less stable patients the benefit likely far outweighs the risk,” he told Medscape Medical News.
Saitz believes policy changes like this should have been made before a crisis.
“Honestly, this is perhaps a silver lining of the crisis” and could lead to permanent change in how OUD is treated in the US, he said.
“Just like we are learning what can be done without a medical in-person visit, we will learn that it is perfectly fine to treat patients with addiction more like we treat patients with other chronic diseases who take medication that has risks and benefits,” Saitz said.
in cases when a patient is quarantined because of coronavirus.
Typically, only licensed practitioners can dispense or administer OUD medications to patients, but during the COVID-19 crisis, treatment program staff members, law enforcement officers, and national guard personnel will be allowed to deliver OUD medications to an approved “lockbox” at the patient’s doorstep. The change applies only while the coronavirus public health emergency lasts.
“This is also an excellent idea,” Saitz said.
ASAM Also Responds
In addition, the American Society of Addiction Medicine (ASAM) released a focused update to its National Practice Guideline for the Treatment of Opioid Use Disorder (NPG).
The update is “especially critical in the context of the ongoing COVID-19 emergency, which threatens to curtail patient access to evidence-based treatment,” the organization said in a news release. The new document updates the 2015 NPG. It includes 13 new recommendations and major revisions to 35 existing recommendations.
One new recommendation states that comprehensive assessment of a patient is critical for treatment planning, but completing all assessments should not delay or preclude initiating pharmacotherapy for OUD. Another new recommendation states that there is no recommended time limit for pharmacotherapy.
ASAM continues to recommend that patients’ psychosocial needs be assessed and psychosocial treatment offered. However, if patients can’t access psychosocial treatment because they are in isolation or have other risk factors that preclude external interactions, clinicians should not delay initiation of medication for the treatment of addiction.
Expanding the use of telemedicine might also be appropriate for many patients, ASAM announced.
They note that the NPG is the first to address in a single document all medications currently approved by the US Food and Drug Administration to treat OUD and opioid withdrawal, including all available buprenorphine formulations.
“All of the updated recommendations are designed to both improve the quality and consistency of care and reduce barriers to access to care for Americans living with OUD. The updated recommendations aim to support initiation of buprenorphine treatment in the emergency department and other urgent care settings,” the society said in the release.
“In addition, [the recommendations] provide greater flexibility on dosing during the initiation of buprenorphine treatment and for initiation of buprenorphine at home (which is also an important change in the midst of the COVID-19 crisis).”
The full document is available online.
This article first appeared on Medscape.com.
In the face of the US COVID-19 pandemic, the US Substance Abuse and Mental Health Services Administration (SAMHSA) has announced policy changes to allow some patients in opioid treatment programs (OTP) to take home their medication.
According to the agency, states may request “blanket exceptions” for all stable patients in an OTP to receive a 28-day supply of take-home doses of medications such as methadone and buprenorphine, which are used to treat opioid use disorder (OUD).
States may request up to 14 days of take-home medication for patients who are less stable but who can, in the judgment of OTP clinicians, safely handle this level of take-home medication.
“SAMHSA recognizes the evolving issues surrounding COVID-19 and the emerging needs OTPs continue to face,” the agency writes in its updated guidance.
“SAMHSA affirms its commitment to supporting OTPs in any way possible during this time. As such, we are expanding our previous guidance to provide increased flexibility,” the agency said.
A ‘Lifesaving’ Decision
Commenting on the SAMHSA policy change, Richard Saitz, MD, professor and chair of the department of community health sciences, Boston University School of Public Health, said, the policy “is not only a good idea, it is critical and lifesaving.”
“This approach had to be done now. With the reduction in face-to-face visits, patients with opioid use disorder need a way to access treatment. If they cannot get opioid agonists, they would withdraw and return to illicit opioid use and high overdose risk and it would be cruel,” said Saitz.
“It is possible that there will be some diversion and some risk of overdose or misuse, but even for less stable patients the benefit likely far outweighs the risk,” he told Medscape Medical News.
Saitz believes policy changes like this should have been made before a crisis.
“Honestly, this is perhaps a silver lining of the crisis” and could lead to permanent change in how OUD is treated in the US, he said.
“Just like we are learning what can be done without a medical in-person visit, we will learn that it is perfectly fine to treat patients with addiction more like we treat patients with other chronic diseases who take medication that has risks and benefits,” Saitz said.
in cases when a patient is quarantined because of coronavirus.
Typically, only licensed practitioners can dispense or administer OUD medications to patients, but during the COVID-19 crisis, treatment program staff members, law enforcement officers, and national guard personnel will be allowed to deliver OUD medications to an approved “lockbox” at the patient’s doorstep. The change applies only while the coronavirus public health emergency lasts.
“This is also an excellent idea,” Saitz said.
ASAM Also Responds
In addition, the American Society of Addiction Medicine (ASAM) released a focused update to its National Practice Guideline for the Treatment of Opioid Use Disorder (NPG).
The update is “especially critical in the context of the ongoing COVID-19 emergency, which threatens to curtail patient access to evidence-based treatment,” the organization said in a news release. The new document updates the 2015 NPG. It includes 13 new recommendations and major revisions to 35 existing recommendations.
One new recommendation states that comprehensive assessment of a patient is critical for treatment planning, but completing all assessments should not delay or preclude initiating pharmacotherapy for OUD. Another new recommendation states that there is no recommended time limit for pharmacotherapy.
ASAM continues to recommend that patients’ psychosocial needs be assessed and psychosocial treatment offered. However, if patients can’t access psychosocial treatment because they are in isolation or have other risk factors that preclude external interactions, clinicians should not delay initiation of medication for the treatment of addiction.
Expanding the use of telemedicine might also be appropriate for many patients, ASAM announced.
They note that the NPG is the first to address in a single document all medications currently approved by the US Food and Drug Administration to treat OUD and opioid withdrawal, including all available buprenorphine formulations.
“All of the updated recommendations are designed to both improve the quality and consistency of care and reduce barriers to access to care for Americans living with OUD. The updated recommendations aim to support initiation of buprenorphine treatment in the emergency department and other urgent care settings,” the society said in the release.
“In addition, [the recommendations] provide greater flexibility on dosing during the initiation of buprenorphine treatment and for initiation of buprenorphine at home (which is also an important change in the midst of the COVID-19 crisis).”
The full document is available online.
This article first appeared on Medscape.com.
In the face of the US COVID-19 pandemic, the US Substance Abuse and Mental Health Services Administration (SAMHSA) has announced policy changes to allow some patients in opioid treatment programs (OTP) to take home their medication.
According to the agency, states may request “blanket exceptions” for all stable patients in an OTP to receive a 28-day supply of take-home doses of medications such as methadone and buprenorphine, which are used to treat opioid use disorder (OUD).
States may request up to 14 days of take-home medication for patients who are less stable but who can, in the judgment of OTP clinicians, safely handle this level of take-home medication.
“SAMHSA recognizes the evolving issues surrounding COVID-19 and the emerging needs OTPs continue to face,” the agency writes in its updated guidance.
“SAMHSA affirms its commitment to supporting OTPs in any way possible during this time. As such, we are expanding our previous guidance to provide increased flexibility,” the agency said.
A ‘Lifesaving’ Decision
Commenting on the SAMHSA policy change, Richard Saitz, MD, professor and chair of the department of community health sciences, Boston University School of Public Health, said, the policy “is not only a good idea, it is critical and lifesaving.”
“This approach had to be done now. With the reduction in face-to-face visits, patients with opioid use disorder need a way to access treatment. If they cannot get opioid agonists, they would withdraw and return to illicit opioid use and high overdose risk and it would be cruel,” said Saitz.
“It is possible that there will be some diversion and some risk of overdose or misuse, but even for less stable patients the benefit likely far outweighs the risk,” he told Medscape Medical News.
Saitz believes policy changes like this should have been made before a crisis.
“Honestly, this is perhaps a silver lining of the crisis” and could lead to permanent change in how OUD is treated in the US, he said.
“Just like we are learning what can be done without a medical in-person visit, we will learn that it is perfectly fine to treat patients with addiction more like we treat patients with other chronic diseases who take medication that has risks and benefits,” Saitz said.
in cases when a patient is quarantined because of coronavirus.
Typically, only licensed practitioners can dispense or administer OUD medications to patients, but during the COVID-19 crisis, treatment program staff members, law enforcement officers, and national guard personnel will be allowed to deliver OUD medications to an approved “lockbox” at the patient’s doorstep. The change applies only while the coronavirus public health emergency lasts.
“This is also an excellent idea,” Saitz said.
ASAM Also Responds
In addition, the American Society of Addiction Medicine (ASAM) released a focused update to its National Practice Guideline for the Treatment of Opioid Use Disorder (NPG).
The update is “especially critical in the context of the ongoing COVID-19 emergency, which threatens to curtail patient access to evidence-based treatment,” the organization said in a news release. The new document updates the 2015 NPG. It includes 13 new recommendations and major revisions to 35 existing recommendations.
One new recommendation states that comprehensive assessment of a patient is critical for treatment planning, but completing all assessments should not delay or preclude initiating pharmacotherapy for OUD. Another new recommendation states that there is no recommended time limit for pharmacotherapy.
ASAM continues to recommend that patients’ psychosocial needs be assessed and psychosocial treatment offered. However, if patients can’t access psychosocial treatment because they are in isolation or have other risk factors that preclude external interactions, clinicians should not delay initiation of medication for the treatment of addiction.
Expanding the use of telemedicine might also be appropriate for many patients, ASAM announced.
They note that the NPG is the first to address in a single document all medications currently approved by the US Food and Drug Administration to treat OUD and opioid withdrawal, including all available buprenorphine formulations.
“All of the updated recommendations are designed to both improve the quality and consistency of care and reduce barriers to access to care for Americans living with OUD. The updated recommendations aim to support initiation of buprenorphine treatment in the emergency department and other urgent care settings,” the society said in the release.
“In addition, [the recommendations] provide greater flexibility on dosing during the initiation of buprenorphine treatment and for initiation of buprenorphine at home (which is also an important change in the midst of the COVID-19 crisis).”
The full document is available online.
This article first appeared on Medscape.com.
ERAS protocol for cesarean delivery reduces opioid usage
GRAPEVINE, TEX. – An enhanced recovery after surgery (ERAS) pathway for cesarean delivery decreased postoperative opioid usage by 62% in one health care organization, researchers reported at the Pregnancy Meeting. The protocol incorporates a stepwise approach to pain control with no scheduled postoperative opioids.
Abington Jefferson Health, which includes two hospitals in Pennsylvania, implemented an ERAS pathway for all cesarean deliveries in October 2018. Kathryn Ruymann, MD, said at the meeting sponsored by the Society for Maternal-Fetal Medicine. Dr. Ruymann is an obstetrics and gynecology resident at Abington Jefferson Health.
Prior to the ERAS protocol, 99%-100% of patients took an opioid during the postoperative period. “With ERAS, 26% of patients never took an opioid during the postop period,” Dr. Ruymann and her associates reported. “Pain scores decreased with ERAS for postoperative days 1-3 and remained unchanged on day 4.”
One in 300 opioid-naive patients who receives opioids after cesarean delivery becomes a persistent user, one study has shown (Am J Obstet Gynecol. 2016 Sep; 215(3):353.e1-18). “ERAS pathways integrate evidence-based interventions before, during, and after surgery to optimize outcomes, specifically to decrease postoperative opioid use,” the researchers said.
While other surgical fields have adopted ERAS pathways, more research is needed in obstetrics, said Dr. Ruymann. More than 4,500 women deliver at Abington Jefferson Health each year, and about a third undergo cesarean deliveries.
The organization’s ERAS pathway incorporates preoperative education, fasting guidelines, and intraoperative analgesia, nausea prophylaxis, and antimicrobial therapy. Under the new protocol, postoperative analgesia includes scheduled administration of nonopioid medications, including celecoxib and acetaminophen. In addition, patients may take 5-10 mg of oxycodone orally every 4 hours as needed, and hydromorphone 0.4 mg IV as needed may be used for refractory pain. In addition, patients should resume eating as soon as tolerated and be out of bed within 4 hours after surgery, according to the protocol. Postoperative management of pruritus and instructions on how to wean off opioids at home are among the other elements of the enhanced recovery plan.
To examine postoperative opioid usage before and after implementation of the ERAS pathway, the investigators conducted a retrospective cohort study of 316 women who underwent cesarean delivery 3 months before the start of the ERAS pathway and 267 who underwent cesarean delivery 3 months after. The researchers used an application developed in Qlik Sense, a data analytics platform, to calculate opioid usage.
Mean postoperative opioid use decreased by 62%. The reduction in opioid use remained 8 months after starting the ERAS pathway.
“An ERAS pathway for [cesarean delivery] decreases postoperative opioid usage by integrating a multimodal stepwise approach to pain control and recovery,” the researchers said. “Standardized order sets and departmentwide education were crucial in the success of ERAS. Additional research is needed to evaluate the impact of unique components of ERAS in order to optimize this pathway.”
The researchers had no disclosures.
SOURCE: Ruymann K et al. Am J Obstet Gynecol. 2020 Jan;222(1):S212, Abstract 315.
GRAPEVINE, TEX. – An enhanced recovery after surgery (ERAS) pathway for cesarean delivery decreased postoperative opioid usage by 62% in one health care organization, researchers reported at the Pregnancy Meeting. The protocol incorporates a stepwise approach to pain control with no scheduled postoperative opioids.
Abington Jefferson Health, which includes two hospitals in Pennsylvania, implemented an ERAS pathway for all cesarean deliveries in October 2018. Kathryn Ruymann, MD, said at the meeting sponsored by the Society for Maternal-Fetal Medicine. Dr. Ruymann is an obstetrics and gynecology resident at Abington Jefferson Health.
Prior to the ERAS protocol, 99%-100% of patients took an opioid during the postoperative period. “With ERAS, 26% of patients never took an opioid during the postop period,” Dr. Ruymann and her associates reported. “Pain scores decreased with ERAS for postoperative days 1-3 and remained unchanged on day 4.”
One in 300 opioid-naive patients who receives opioids after cesarean delivery becomes a persistent user, one study has shown (Am J Obstet Gynecol. 2016 Sep; 215(3):353.e1-18). “ERAS pathways integrate evidence-based interventions before, during, and after surgery to optimize outcomes, specifically to decrease postoperative opioid use,” the researchers said.
While other surgical fields have adopted ERAS pathways, more research is needed in obstetrics, said Dr. Ruymann. More than 4,500 women deliver at Abington Jefferson Health each year, and about a third undergo cesarean deliveries.
The organization’s ERAS pathway incorporates preoperative education, fasting guidelines, and intraoperative analgesia, nausea prophylaxis, and antimicrobial therapy. Under the new protocol, postoperative analgesia includes scheduled administration of nonopioid medications, including celecoxib and acetaminophen. In addition, patients may take 5-10 mg of oxycodone orally every 4 hours as needed, and hydromorphone 0.4 mg IV as needed may be used for refractory pain. In addition, patients should resume eating as soon as tolerated and be out of bed within 4 hours after surgery, according to the protocol. Postoperative management of pruritus and instructions on how to wean off opioids at home are among the other elements of the enhanced recovery plan.
To examine postoperative opioid usage before and after implementation of the ERAS pathway, the investigators conducted a retrospective cohort study of 316 women who underwent cesarean delivery 3 months before the start of the ERAS pathway and 267 who underwent cesarean delivery 3 months after. The researchers used an application developed in Qlik Sense, a data analytics platform, to calculate opioid usage.
Mean postoperative opioid use decreased by 62%. The reduction in opioid use remained 8 months after starting the ERAS pathway.
“An ERAS pathway for [cesarean delivery] decreases postoperative opioid usage by integrating a multimodal stepwise approach to pain control and recovery,” the researchers said. “Standardized order sets and departmentwide education were crucial in the success of ERAS. Additional research is needed to evaluate the impact of unique components of ERAS in order to optimize this pathway.”
The researchers had no disclosures.
SOURCE: Ruymann K et al. Am J Obstet Gynecol. 2020 Jan;222(1):S212, Abstract 315.
GRAPEVINE, TEX. – An enhanced recovery after surgery (ERAS) pathway for cesarean delivery decreased postoperative opioid usage by 62% in one health care organization, researchers reported at the Pregnancy Meeting. The protocol incorporates a stepwise approach to pain control with no scheduled postoperative opioids.
Abington Jefferson Health, which includes two hospitals in Pennsylvania, implemented an ERAS pathway for all cesarean deliveries in October 2018. Kathryn Ruymann, MD, said at the meeting sponsored by the Society for Maternal-Fetal Medicine. Dr. Ruymann is an obstetrics and gynecology resident at Abington Jefferson Health.
Prior to the ERAS protocol, 99%-100% of patients took an opioid during the postoperative period. “With ERAS, 26% of patients never took an opioid during the postop period,” Dr. Ruymann and her associates reported. “Pain scores decreased with ERAS for postoperative days 1-3 and remained unchanged on day 4.”
One in 300 opioid-naive patients who receives opioids after cesarean delivery becomes a persistent user, one study has shown (Am J Obstet Gynecol. 2016 Sep; 215(3):353.e1-18). “ERAS pathways integrate evidence-based interventions before, during, and after surgery to optimize outcomes, specifically to decrease postoperative opioid use,” the researchers said.
While other surgical fields have adopted ERAS pathways, more research is needed in obstetrics, said Dr. Ruymann. More than 4,500 women deliver at Abington Jefferson Health each year, and about a third undergo cesarean deliveries.
The organization’s ERAS pathway incorporates preoperative education, fasting guidelines, and intraoperative analgesia, nausea prophylaxis, and antimicrobial therapy. Under the new protocol, postoperative analgesia includes scheduled administration of nonopioid medications, including celecoxib and acetaminophen. In addition, patients may take 5-10 mg of oxycodone orally every 4 hours as needed, and hydromorphone 0.4 mg IV as needed may be used for refractory pain. In addition, patients should resume eating as soon as tolerated and be out of bed within 4 hours after surgery, according to the protocol. Postoperative management of pruritus and instructions on how to wean off opioids at home are among the other elements of the enhanced recovery plan.
To examine postoperative opioid usage before and after implementation of the ERAS pathway, the investigators conducted a retrospective cohort study of 316 women who underwent cesarean delivery 3 months before the start of the ERAS pathway and 267 who underwent cesarean delivery 3 months after. The researchers used an application developed in Qlik Sense, a data analytics platform, to calculate opioid usage.
Mean postoperative opioid use decreased by 62%. The reduction in opioid use remained 8 months after starting the ERAS pathway.
“An ERAS pathway for [cesarean delivery] decreases postoperative opioid usage by integrating a multimodal stepwise approach to pain control and recovery,” the researchers said. “Standardized order sets and departmentwide education were crucial in the success of ERAS. Additional research is needed to evaluate the impact of unique components of ERAS in order to optimize this pathway.”
The researchers had no disclosures.
SOURCE: Ruymann K et al. Am J Obstet Gynecol. 2020 Jan;222(1):S212, Abstract 315.
REPORTING FROM THE PREGNANCY MEETING
Kratom: What we know, what to tell your patients
Mitragyna speciosa, better known as kratom, is a tropical evergreen tree that is native to Southeast Asia. Botanically, it is a member of the Rubiaceae family, as is the coffee plant, and physical laborers among indigenous populations have historically chewed the leaves or brewed them as a tea to improve endurance and reduce fatigue.1 Kratom is psychoactive; small amounts (up to 5 g of plant material) possess stimulant properties, while larger doses (>5 g) produce opioid-like, sedative, euphoric, and antinociceptive effects.2
In recent years, kratom has gained popularity in Western parts of the world due to its unique properties and perceived safety as a botanical product. Individuals may use kratom to boost their energy, relieve pain, or treat a wide range of physical or mood problems. Increasingly, kratom is being used by people who abuse opioids to self-manage opioid withdrawal, or for its euphoric effects. But kratom carries several important risks, including addiction, serious adverse effects, and possibly death. In this article, we review the epidemiology and pharmacology of kratom, and provide some guidance for educating patients about this substance.
Widely used but not FDA approved
Although kratom is not regulated or approved by the FDA, 3 to 5 million Americans use it regularly.3 According to an internet survey, kratom users are mostly college-educated, employed white men, age 31 to 50, who take the substance to manage pain or to treat general anxiety and mood disorders.4 Some individuals use kratom as an opioid substitute to reduce symptoms of opioid withdrawal.4
Kratom is available from a wide range of manufacturers in various formulations, including powders, tablets, liquids, and gum. It is sometimes sold in combination with other agents as a single product. Low-cost, over-the-counter kratom products are available as “dietary supplements” in retail stores or online. Although the product packaging sometimes recommends a specific dose, the amount of active ingredients (as well as other agents) is unknown. Kratom is illegal in several states (Box5).
Box
The use and sale of kratom is illegal in several countries, including Australia, Poland, Denmark, Sweden, Malaysia, and Vietnam. In the United States, kratom was legal to grow and purchase in all 50 states until 2015, when the Drug Enforcement Administration (DEA) identified kratom as a “substance of concern.” In August 2016, the DEA submitted a notice of intent to place mitragynine and 7-hydroxymitragynine, 2 alkaloids of kratom that have opioid-like properties, into Schedule I of the Controlled Substance Act; however, due to significant public pressure, the DEA withdrew the request in October 2016.
As of February 2020, kratom was illegal to buy, sell, or use in Wisconsin, Rhode Island, Vermont, Indiana, Arkansas, Alabama, specific counties of some states, and the District of Columbia. Legislation was pending in New York, Missouri, and Louisiana.
Source: Reference 5
The 2 alkaloids of interest
More than 40 alkaloids have been isolated from kratom leaves. The proportions of these alkaloids vary significantly depending on the environment in which the plant is grown, the breeding and harvesting techniques, and the age of the plant.6 Two alkaloids of significant interest are mitragynine (Figure 1) and 7-hydroxymitragynine (Figure 2), both of which are unique to M. speciosa and have opioid-like properties. Administering these alkaloids to morphine-dependent rats resulted in cross-tolerance and precipitated withdrawal when the rats were given naloxone.7 The potency of kratom at the mu opioid receptor has been found to exceed that of morphine.
Competitive binding studies that examined the affinity of mitragynine and 7-hydroxymitragynine at the various opioid receptor subtypes found a preference for the kappa receptors (antagonism), followed by mu (partial agonism), and lastly delta. This profile of mitragynine is very similar to that of buprenorphine.8 The affinity of 7-hydroxymitragynine for the mu receptor (agonism) is significantly greater than that of mitragynine.9 Mitragynine also interacts with noradrenergic and serotonergic pathways by stimulating postsynaptic alpha-2 adrenergic receptors and inhibiting 5-HT2A receptors.9 These properties are responsible for kratom’s ability to manage opioid withdrawal symptoms, which are generally attributed to a hyperactive noradrenergic system. There also is evidence that the hepatic metabolite 7-hydroxymitragynine is important in mediating the analgesic component of mitragynine.10
The initial effects of kratom typically begin within 10 to 20 minutes of consumption, and the full effects are experienced in 30 to 60 minutes.1 The half-life of mitragynine in humans has not yet been determined, but is believed to be relatively short.11 In rats, the half-life of mitragynine is 2 to 3 hours.12 Individuals who use kratom to prevent opioid withdrawal have reported taking it as often as every 6 to 12 hours.13
Continue to: Metabolism of mitragynine...
Metabolism of mitragynine is predominantly carried out through cytochrome P450 (CYP) 3A4, with minor contributions by 2D6 and 2C9. A total of 13 metabolites are produced, including 7-hydroxymitragynine.14 Kratom’s constituents also interact with the CYP system, inhibiting 2C9, 2D6, and 3A4 isoenzymes, and to some extent, 1A2.
Adverse effects can be fatal
An animal study revealed that when administered intravenously, mitragynine and 7-hydroxymitragynine have a similar toxicity profile to heroin.15 When these alkaloids were administered in ascending doses, increases in blood pressure and elevations in liver function tests and creatinine levels from baseline were observed.
Chronic kratom use can result in weight loss, insomnia, constipation, dehydration, skin hyperpigmentation, and extreme fatigue.16 There have also been reports of seizures, delusions, hallucinations, respiratory depression, hepatotoxicity, coma, and death.17,18 An emerging concern is the potential development of fatty liver infiltrates leading to cholestatic liver damage.19-25 One case report described a young man who developed a serum aspartate aminotransferase level of 1,300 IU/L (reference range: 5 to 45 IU/L) and a serum alanine aminotransaminase level of 3,700 IU/L (reference range: 5 to 60 IU/L) after he ingested a kratom product.26 Histologically, the pattern of liver injury mimics primary biliary cholangitis.27
In recent years, calls to poison control centers in the United States related to kratom exposure have risen. Between 2011 and 2017, the number of calls increased from 1 a month to 2 each day.28 The US National Poison Data System has also noted an increase in the number of calls in reference to kratom. It received 2,312 calls from January 2011 through July 2018, with 18 calls occurring in 2011, and 357 within the first 7 months of 2018.29
As of February 2018, the FDA had received reports of 44 deaths associated with kratom.30 There have been reports of fatal overdoses involving kratom, particularly when kratom is co-ingested or used with adulterated and/or combination agents, including one case that involved quetiapine.31-33 There have been reports of deaths believed to be attributed to the use of kratom alone; in one such case, a 35-year-old man experienced a fatal cardiac arrest due to kratom use with no other coingestants.34 Among the reports of deaths in which kratom was the only substance consumed, the mitragynine blood levels of the deceased individuals were found to be higher than the levels associated with individuals who had consumed traditional kratom teas.29
Continue to: There is a lack of quality control...
There is a lack of quality control of commercially available kratom preparations. The FDA has found kratom products that exceeded the level of safe exposure to nickel and lead.35 There have also been reports of Salmonella outbreaks associated with kratom products.36
Detecting kratom use
Mitragynine is a lipophilic alkaloid that is poorly soluble in water37 and eliminated primarily in urine.12 Based on data from treatment center admissions, kratom can be detected in urine samples for 5 to 6 days after use.24,38,39 However, kratom is not detectable by a standard urine toxicology screen; therefore, a high degree of suspicion and special confirmatory testing are necessary. The breakdown products of mitragynine can be detected through gas chromatography coupled with mass spectrometry (GC/MS), liquid chromatography with linear ion trap mass spectrometry, or electrospray tandem mass spectrometry.40-42
A familiar withdrawal syndrome
Abrupt discontinuation of high-dose, long-term kratom use can produce withdrawal symptoms.13 Symptoms of kratom withdrawal resemble those of opioid withdrawal. These include physiological symptoms (mydriasis, nausea, sweating and chills, muscle and body aches, tremors and twitches, diarrhea, rhinorrhea, and lacrimation) and psychological symptoms (insomnia, restlessness, irritability/hostility, fatigue, anxiety, mood disturbances, and hallucinations).13 Symptoms are first noted starting 12 hours after the last use of kratom, and can last up to 7 days.43 Withdrawal intensity has been positively correlated with the daily amount of kratom consumed, as well as the duration and frequency of use.13,16
In 2 case reports, the newborns of women who used kratom during pregnancy experienced neonatal abstinence syndrome.44,45 In these 2 reports, symptoms such as jitteriness, irritability, feeding intolerance, and vomiting emerged on postpartum Day 2. The newborns were admitted to a neonatal ICU and started on a standard opioid protocol with IV morphine and subsequently tapered with an oral formulation over 5 days.44,45
Helping patients who use kratom
The best approach to treating a patient who is experiencing kratom withdrawal is symptomatic management, as would be appropriate for a patient experiencing opioid withdrawal.13 However, the use of agents such as methadone or buprenorphine for patients undergoing kratom withdrawal has not been thoroughly evaluated; very few reports have been published.46,47
Continue to: Similarly, while the standard of care...
Similarly, while the standard of care for treating a patient with opioid use disorder is medication-assisted treatment in combination with counseling and behavioral therapies, there is little evidence on the efficacy of such treatments for patients who use kratom. There are no specific guidelines, and the risk of relapsing to kratom use is high.48,49 Nonetheless, some clinicians have used the same protocol for patients with opioid use disorder to treat patients using kratom, and several published case reports describe this approach.50,51 Because administering buprenorphine/naltrexone to a patient who is dependent on kratom can precipitate withdrawal, clinicians should follow a similar initiation protocol as for opioid dependence when starting a patient on these agents (ie, a washout period with a challenge test would be prudent prior to starting naltrexone).
In cases of kratom overdose, naloxone has been shown to reverse the analgesic effects of mitragynine in rats. However, in a case report of an individual who accidently overdosed on a kratom product, naloxone had a modest effect.52
Bottom Line
Kratom is a botanical substance that acts like a stimulant at low doses and an opioid at higher doses. Patients might use it to treat mood-related symptoms, relieve pain, or manage opioid withdrawal. Kratom use has been associated with the development of addiction as well as a multitude of serious adverse effects, including hepatotoxicity and overdose. Long-term management may be required for a patient who uses kratom.
Related Resources
- White CM. Pharmacologic and clinical assessment of kratom: an update. Am J Health Syst Pharm. 2019;76(23):1915-1925.
- Smith KE, Lawson T. Prevalence and motivations for kratom use in a sample of substance users enrolled in a residential treatment program. Drug Alcohol Depend. 2017;180:340-348.
Drug Brand Names
Buprenorphine • Subutex, Sublocade
Buprenorphine/naltrexone • Suboxone
Methadone • Methadose
Naltrexone • Revia
Naloxone • Narcan
Quetiapine • Seroquel
1. Henningfield JE, Fant RV, Wang DW. The abuse potential of kratom according the 8 factors of the controlled substances act: implications for regulation and research. Psychopharmacology (Berl). 2018;235(2):573-589.
2. Chang-Chien GC, Odonkor CA, Amorapanth P, et al. Is kratom the new ‘legal high’ on the block?: the case of an emerging opioid receptor agonist with substance abuse potential. Pain Physician. 2017;20(1):E195-E198.
3. Penders T, Jones WB. Kratom, a substance of increasing concern [PCSS webinar]. Providers Clinical Support System. November 28, 2018. https://pcssnow.org/event/kratom-a-substance-of-increasing-concern. Accessed January 29, 2020.
4. Grundmann O. Patterns of kratom use and health impact in the US-results from an online survey. Drug Alcohol Depend. 2017;176:63-70.
5. US Drug Enforcement Administration. Drugs of concern. https://www.dea.gov/sites/default/files/sites/getsmartaboutdrugs.com/files/publications/DoA_2017Ed_Updated_6.16.17.pdf#page=84. Updated June 16, 2017. Accessed January 29, 2020.
6. Matsumoto K, Horie S, Ishikawa H, et al. Antinociceptive effect of 7-hydroxymitragynine in mice: discovery of an orally active opioid analgesic from the Thai medicinal herb Mitragyna speciosa. Life Sciences. 2004;74(17):2143-2155.
7. Takayama H. Chemistry and pharmacology of analgesic indole alkaloids from the rubiaceous plant, Mitragyna speciosa. Chem Pharm Bull (Tokyo). 2004;52(8):916-928.
8. Suhaimi FW, Yusoff NH, Hassan R, et al. Neurobiology of kratom and its main alkaloid mitragynine. Brain Res Bull. 2016;126(pt 1):29-40.
9. Prozialeck WC, Jivan JK, Andurkar SV. Pharmacology of kratom: an emerging botanical agent with stimulant, analgesic and opioid-like effects. J Am Osteopath Assoc. 2012;112(12):792-799.
10. Kruegel AC, Uprety R, Grinnell SG, et al. 7-hydroxymitragynine is an active metabolite of mitragynine and a key mediator of its analgesic effects. ACS Cent Sci. 2019;5(6):992-1001.
11. Trakulsrichai S, Sathirakul K, Auparakkitanon S, et al. Pharmacokinetics of mitragynine in man. Drug Des Devel Ther. 2015:9:2421-2429.
12. Warner ML, Kaufman NC, Grundmann O, et al. The pharmacology and toxicology of kratom: from traditional herb to drug of abuse. Intl J Legal Med. 2016;130(1):127-138.
13. Stanciu CN, Gnanasegaram SA, Ahmed S, et al. Kratom withdrawal: a systematic review with case series. J Psychoactive Drugs. 2019;51(1):12-18.
14. Kamble SH, Sharma A, King TI, et al. Metabolite profiling and identification of enzymes responsible for the metabolism of mitragynine, the major alkaloid of Mitragyna speciosa (kratom). Xenobiotica. 2019;49(11):1279-1288.
15. Smith LC, Lin L, Hwang CS, et al. Lateral flow assessment and unanticipated toxicity of kratom. Chem Res Toxicol. 2019;32(1):113-121.
16. Saingam D, Assanangkornchai S, Geater AF, et al. Factor analytical investigation of Krathom (Mitragyna speciosa Korth.) withdrawal syndrome in Thailand. J Psychoactive Drugs. 2016;48(2):76-85.
17. Vicknasingam B, Narayanan S, Beng GT, et al. The informal use of ketum (Mitragyna speciosa) for opioid withdrawal in the northern states of peninsular Malaysia and implications for drug substitution therapy. Int J Drug Policy. 2010;21(4):283-288.
18. Saingam D, Assanangkornchai S, Geater AF, et al. Pattern and consequences of krathom (Mitragyna speciosa Korth.) use among male villagers in southern Thailand: a qualitative study. Int J Drug Policy. 2013;24(4):351-358.
19. Fernandes CT, Iqbal U, Tighe SP, et al. Kratom-induced cholestatic liver injury and its conservative management. J Investig Med High Impact Case Rep. 2019;7:2324709619836138. doi: 10.1177/2324709619836138.
20. Dorman C, Wong M, Khan A. Cholestatic hepatitis from prolonged kratom use: a case report. Hepatology. 2015;61(3):1086-1087.
21. Osborne CS, Overstreet AN, Rockey DC, et al. Drug-induced liver injury caused by kratom use as an alternative pain treatment amid an ongoing opioid epidemic. J Investig Med High Impact Case Rep. 2019;7:2324709619826167. doi: 10.1177/2324709619826167.
22. Mousa MS, Sephien A, Gutierrez J, et al. N-acetylcysteine for acute hepatitis induced by kratom herbal tea. Am J Ther. 2018;25(5):e550-e551.
23. Riverso M, Chang M, Soldevila-Pico C, et al. Histologic characterization of kratom use-associated liver injury. Gastroenterology Res. 2018;11(1):79-82.
24. Kapp FG, Maurer HH, Auwärter V, et al. Intrahepatic cholestasis following abuse of powdered kratom (Mitragyna speciosa). J Med Toxicol. 2011;7(3):227-231.
25. Antony A, Lee TP. Herb-induced liver injury with cholestasis and renal injury secondary to short-term use of kratom (Mitragyna speciosa). Am J Ther. 2019;26(4):e546-e547.
26. Palasamudram Shekar S, Rojas EE, D’Angelo CC, et al. Legally lethal kratom: a herbal supplement with overdose potential. J Psychoactive Drugs. 2019;51(1):28-30.
27. Aldyab M, Ells PF, Bui R, et al. Kratom-induced cholestatic liver injury mimicking anti-mitochondrial antibody-negative primary biliary cholangitis: a case report and review of literature. Gastroenterology Res. 2019;12(4):211-215.
28. Post S, Spiller HA, Chounthirath T. Kratom exposures reported to United States poison control centers: 2011-2017. Clinical Toxicol (Phila). 2019;57(10):847-854.
29. Eggleston W, Stoppacher R, Suen K, et al. Kratom use and toxicities in the United States. Pharmacotherapy. 2019;39(7):775-777.
30. US Food & Drug Administration. Statement from FDA Commissioner Scott Gottlieb, M.D., on the agency’s scientific evidence on the presence of opioid compounds in kratom , underscoring its potential for abuse. https://www.fda.gov/news-events/press-announcements/statement-fda-commissioner-scott-gottlieb-md-agencys-scientific-evidence-presence-opioid-compounds. Published February 6, 2019. Accessed January 29, 2020.
31. Gershman K, Timm K, Frank M, et al. Deaths in Colorado attributed to kratom. N Engl J Med. 2019;380(1):97-98.
32. Kronstrand R, Roman M, Thelander G, et al. Unintentional fatal intoxications with mitragynine and O-desmethyltramadol from the herbal blend krypton. J Anal Toxicol. 2011;35(4):242-247.
33. Hughes RL. Fatal combination of mitragynine and quetiapine - a case report with discussion of a potential herb-drug interaction. Forensic Sci Med Pathol. 2019;15(1):110-113.
34. Abdullah HMA, Haq I, Lamfers R. Cardiac arrest in a young healthy male patient secondary to kratom ingestion: is this ‘legal high’ substance more dangerous than initially thought? BMJ Case Rep. 2019;12(7):pii: e229778. doi: 10.1136/bcr-2019-229778.
35. Laboratory analysis of kratom products for heavy metals. US FDA. https://www.fda.gov/news-events/public-health-focus/laboratory-analysis-kratom-products-heavy-metals. Updated April 3, 2019. Accessed January 29, 2020.
36. FDA investigated multistate outbreak of salmonella infections linked to products reported to contain kratom. US FDA. https://www.fda.gov/food/outbreaks-foodborne-illness/fda-investigated-multistate-outbreak-salmonella-infections-linked-products-reported-contain-kratom. Updated June 29, 2018. Accessed January 14, 2020.
37. Aggarwal G, Robertson E, McKinlay J, et a., Death from kratom toxicity and the possible role of intralipid. J Intensive Care Soc. 2018;19(1):61-63.
38. Drug Facts. Kratom. Confirm Biosciences. https://www.confirmbiosciences.com/knowledge/drug-facts/kratom/. Accessed January 14, 2020.
39. Grinspoon P. How long does kratom stay in the system? Addiction Resource. https://addictionresource.com/drugs/kratom/how-long-kratom-stay-in-your-system/. Updated December 18, 2019. Accessed January 29, 2020.
40. Kaewklum D, Kaewklum M, Pootrakronchai R, et al. Detection of mitragynine and its metaboilite in urine following ingestion of leaves of Mitragyna speciosa korth. Recent Advances in Doping Analysis (13). Proceedings of the Manfred Donike Workshop, 23rd Cologne Workshop on Dope Analysis. 2005:403-406.
41. Lu S, Tran BN, Nelsen JL, et al. Quantitative analysis of mitragynine in human urine by high performance liquid chromatography-tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci. 2009;877(24):2499-2505.
42. Philipp AA, Wissenbach DK, Zoerntlein SW, et al. Studies on the metabolism of mitragynine, the main alkaloid of the herbal drug kratom, in rat and human urine using liquid chromatography-linear ion trap mass spectrometry. J Mass Spectrom. 2009;44(8):1249-1261.
43. Manda VK, Bharathi A, Ali Z, et al. Evaluation of in vitro absorption, distribution, metabolism, and excretion (ADME) properties of mitragynine, 7-hydroxymitragynine, and mitraphylline. Planta Med. 2014;80(7):568-576.
44. Davidson L, Rawat M, Stojanovski S, et al. Natural drugs, not so natural effects: neonatal abstinence syndrome secondary to ‘kratom‘. J Neonatal Perinatal Med. 2019;12(1):109-112.
45. Mackay L, Abrahams R. Novel case of maternal and neonatal kratom dependence and withdrawal. Can Fam Physician. 2018;64(2):121-122.
46. McWhirter L, Morris S. A case report of inpatient detoxification after kratom (Mitragyna speciosa) dependence. Eur Addict Res. 2010;16(4):229-231.
47. Galbis-Reig David. A case report of kratom addiction and withdrawal. WMJ. 2016;115(1):49-52; quiz 53.
48. Singh D, Müller CP, Vicknasingam BK. Kratom (Mitragyna speciose) dependence, withdrawal symptoms and craving in regular users. Drug Alcohol Depend. 2014;139:132-137.
49. Singh D, Müller CP, Vicknasingam, et al. Social functioning of kratom (Mitragyna speciosa) users in Malaysia. J Psychoactive Drugs. 2015;47(2):125-131.
50. Khazaeli A, Jerry JM, Vazirian M. Treatment of kratom withdrawal and addiction with buprenorphine. J Addict Med. 2018;12(6):493-495.
51. Buresh M. Treatment of kratom dependence with buprenorphine-naloxone maintenance. J Addict Med. 2018;12(6):481-483.
52. Overbeek DL, Abraham J, Munzer BW. Kratom (mitragynine) ingestion requiring naloxone reversal. Clin Pract Cases Emerg Med. 2019;3(1):24-26.
Mitragyna speciosa, better known as kratom, is a tropical evergreen tree that is native to Southeast Asia. Botanically, it is a member of the Rubiaceae family, as is the coffee plant, and physical laborers among indigenous populations have historically chewed the leaves or brewed them as a tea to improve endurance and reduce fatigue.1 Kratom is psychoactive; small amounts (up to 5 g of plant material) possess stimulant properties, while larger doses (>5 g) produce opioid-like, sedative, euphoric, and antinociceptive effects.2
In recent years, kratom has gained popularity in Western parts of the world due to its unique properties and perceived safety as a botanical product. Individuals may use kratom to boost their energy, relieve pain, or treat a wide range of physical or mood problems. Increasingly, kratom is being used by people who abuse opioids to self-manage opioid withdrawal, or for its euphoric effects. But kratom carries several important risks, including addiction, serious adverse effects, and possibly death. In this article, we review the epidemiology and pharmacology of kratom, and provide some guidance for educating patients about this substance.
Widely used but not FDA approved
Although kratom is not regulated or approved by the FDA, 3 to 5 million Americans use it regularly.3 According to an internet survey, kratom users are mostly college-educated, employed white men, age 31 to 50, who take the substance to manage pain or to treat general anxiety and mood disorders.4 Some individuals use kratom as an opioid substitute to reduce symptoms of opioid withdrawal.4
Kratom is available from a wide range of manufacturers in various formulations, including powders, tablets, liquids, and gum. It is sometimes sold in combination with other agents as a single product. Low-cost, over-the-counter kratom products are available as “dietary supplements” in retail stores or online. Although the product packaging sometimes recommends a specific dose, the amount of active ingredients (as well as other agents) is unknown. Kratom is illegal in several states (Box5).
Box
The use and sale of kratom is illegal in several countries, including Australia, Poland, Denmark, Sweden, Malaysia, and Vietnam. In the United States, kratom was legal to grow and purchase in all 50 states until 2015, when the Drug Enforcement Administration (DEA) identified kratom as a “substance of concern.” In August 2016, the DEA submitted a notice of intent to place mitragynine and 7-hydroxymitragynine, 2 alkaloids of kratom that have opioid-like properties, into Schedule I of the Controlled Substance Act; however, due to significant public pressure, the DEA withdrew the request in October 2016.
As of February 2020, kratom was illegal to buy, sell, or use in Wisconsin, Rhode Island, Vermont, Indiana, Arkansas, Alabama, specific counties of some states, and the District of Columbia. Legislation was pending in New York, Missouri, and Louisiana.
Source: Reference 5
The 2 alkaloids of interest
More than 40 alkaloids have been isolated from kratom leaves. The proportions of these alkaloids vary significantly depending on the environment in which the plant is grown, the breeding and harvesting techniques, and the age of the plant.6 Two alkaloids of significant interest are mitragynine (Figure 1) and 7-hydroxymitragynine (Figure 2), both of which are unique to M. speciosa and have opioid-like properties. Administering these alkaloids to morphine-dependent rats resulted in cross-tolerance and precipitated withdrawal when the rats were given naloxone.7 The potency of kratom at the mu opioid receptor has been found to exceed that of morphine.
Competitive binding studies that examined the affinity of mitragynine and 7-hydroxymitragynine at the various opioid receptor subtypes found a preference for the kappa receptors (antagonism), followed by mu (partial agonism), and lastly delta. This profile of mitragynine is very similar to that of buprenorphine.8 The affinity of 7-hydroxymitragynine for the mu receptor (agonism) is significantly greater than that of mitragynine.9 Mitragynine also interacts with noradrenergic and serotonergic pathways by stimulating postsynaptic alpha-2 adrenergic receptors and inhibiting 5-HT2A receptors.9 These properties are responsible for kratom’s ability to manage opioid withdrawal symptoms, which are generally attributed to a hyperactive noradrenergic system. There also is evidence that the hepatic metabolite 7-hydroxymitragynine is important in mediating the analgesic component of mitragynine.10
The initial effects of kratom typically begin within 10 to 20 minutes of consumption, and the full effects are experienced in 30 to 60 minutes.1 The half-life of mitragynine in humans has not yet been determined, but is believed to be relatively short.11 In rats, the half-life of mitragynine is 2 to 3 hours.12 Individuals who use kratom to prevent opioid withdrawal have reported taking it as often as every 6 to 12 hours.13
Continue to: Metabolism of mitragynine...
Metabolism of mitragynine is predominantly carried out through cytochrome P450 (CYP) 3A4, with minor contributions by 2D6 and 2C9. A total of 13 metabolites are produced, including 7-hydroxymitragynine.14 Kratom’s constituents also interact with the CYP system, inhibiting 2C9, 2D6, and 3A4 isoenzymes, and to some extent, 1A2.
Adverse effects can be fatal
An animal study revealed that when administered intravenously, mitragynine and 7-hydroxymitragynine have a similar toxicity profile to heroin.15 When these alkaloids were administered in ascending doses, increases in blood pressure and elevations in liver function tests and creatinine levels from baseline were observed.
Chronic kratom use can result in weight loss, insomnia, constipation, dehydration, skin hyperpigmentation, and extreme fatigue.16 There have also been reports of seizures, delusions, hallucinations, respiratory depression, hepatotoxicity, coma, and death.17,18 An emerging concern is the potential development of fatty liver infiltrates leading to cholestatic liver damage.19-25 One case report described a young man who developed a serum aspartate aminotransferase level of 1,300 IU/L (reference range: 5 to 45 IU/L) and a serum alanine aminotransaminase level of 3,700 IU/L (reference range: 5 to 60 IU/L) after he ingested a kratom product.26 Histologically, the pattern of liver injury mimics primary biliary cholangitis.27
In recent years, calls to poison control centers in the United States related to kratom exposure have risen. Between 2011 and 2017, the number of calls increased from 1 a month to 2 each day.28 The US National Poison Data System has also noted an increase in the number of calls in reference to kratom. It received 2,312 calls from January 2011 through July 2018, with 18 calls occurring in 2011, and 357 within the first 7 months of 2018.29
As of February 2018, the FDA had received reports of 44 deaths associated with kratom.30 There have been reports of fatal overdoses involving kratom, particularly when kratom is co-ingested or used with adulterated and/or combination agents, including one case that involved quetiapine.31-33 There have been reports of deaths believed to be attributed to the use of kratom alone; in one such case, a 35-year-old man experienced a fatal cardiac arrest due to kratom use with no other coingestants.34 Among the reports of deaths in which kratom was the only substance consumed, the mitragynine blood levels of the deceased individuals were found to be higher than the levels associated with individuals who had consumed traditional kratom teas.29
Continue to: There is a lack of quality control...
There is a lack of quality control of commercially available kratom preparations. The FDA has found kratom products that exceeded the level of safe exposure to nickel and lead.35 There have also been reports of Salmonella outbreaks associated with kratom products.36
Detecting kratom use
Mitragynine is a lipophilic alkaloid that is poorly soluble in water37 and eliminated primarily in urine.12 Based on data from treatment center admissions, kratom can be detected in urine samples for 5 to 6 days after use.24,38,39 However, kratom is not detectable by a standard urine toxicology screen; therefore, a high degree of suspicion and special confirmatory testing are necessary. The breakdown products of mitragynine can be detected through gas chromatography coupled with mass spectrometry (GC/MS), liquid chromatography with linear ion trap mass spectrometry, or electrospray tandem mass spectrometry.40-42
A familiar withdrawal syndrome
Abrupt discontinuation of high-dose, long-term kratom use can produce withdrawal symptoms.13 Symptoms of kratom withdrawal resemble those of opioid withdrawal. These include physiological symptoms (mydriasis, nausea, sweating and chills, muscle and body aches, tremors and twitches, diarrhea, rhinorrhea, and lacrimation) and psychological symptoms (insomnia, restlessness, irritability/hostility, fatigue, anxiety, mood disturbances, and hallucinations).13 Symptoms are first noted starting 12 hours after the last use of kratom, and can last up to 7 days.43 Withdrawal intensity has been positively correlated with the daily amount of kratom consumed, as well as the duration and frequency of use.13,16
In 2 case reports, the newborns of women who used kratom during pregnancy experienced neonatal abstinence syndrome.44,45 In these 2 reports, symptoms such as jitteriness, irritability, feeding intolerance, and vomiting emerged on postpartum Day 2. The newborns were admitted to a neonatal ICU and started on a standard opioid protocol with IV morphine and subsequently tapered with an oral formulation over 5 days.44,45
Helping patients who use kratom
The best approach to treating a patient who is experiencing kratom withdrawal is symptomatic management, as would be appropriate for a patient experiencing opioid withdrawal.13 However, the use of agents such as methadone or buprenorphine for patients undergoing kratom withdrawal has not been thoroughly evaluated; very few reports have been published.46,47
Continue to: Similarly, while the standard of care...
Similarly, while the standard of care for treating a patient with opioid use disorder is medication-assisted treatment in combination with counseling and behavioral therapies, there is little evidence on the efficacy of such treatments for patients who use kratom. There are no specific guidelines, and the risk of relapsing to kratom use is high.48,49 Nonetheless, some clinicians have used the same protocol for patients with opioid use disorder to treat patients using kratom, and several published case reports describe this approach.50,51 Because administering buprenorphine/naltrexone to a patient who is dependent on kratom can precipitate withdrawal, clinicians should follow a similar initiation protocol as for opioid dependence when starting a patient on these agents (ie, a washout period with a challenge test would be prudent prior to starting naltrexone).
In cases of kratom overdose, naloxone has been shown to reverse the analgesic effects of mitragynine in rats. However, in a case report of an individual who accidently overdosed on a kratom product, naloxone had a modest effect.52
Bottom Line
Kratom is a botanical substance that acts like a stimulant at low doses and an opioid at higher doses. Patients might use it to treat mood-related symptoms, relieve pain, or manage opioid withdrawal. Kratom use has been associated with the development of addiction as well as a multitude of serious adverse effects, including hepatotoxicity and overdose. Long-term management may be required for a patient who uses kratom.
Related Resources
- White CM. Pharmacologic and clinical assessment of kratom: an update. Am J Health Syst Pharm. 2019;76(23):1915-1925.
- Smith KE, Lawson T. Prevalence and motivations for kratom use in a sample of substance users enrolled in a residential treatment program. Drug Alcohol Depend. 2017;180:340-348.
Drug Brand Names
Buprenorphine • Subutex, Sublocade
Buprenorphine/naltrexone • Suboxone
Methadone • Methadose
Naltrexone • Revia
Naloxone • Narcan
Quetiapine • Seroquel
Mitragyna speciosa, better known as kratom, is a tropical evergreen tree that is native to Southeast Asia. Botanically, it is a member of the Rubiaceae family, as is the coffee plant, and physical laborers among indigenous populations have historically chewed the leaves or brewed them as a tea to improve endurance and reduce fatigue.1 Kratom is psychoactive; small amounts (up to 5 g of plant material) possess stimulant properties, while larger doses (>5 g) produce opioid-like, sedative, euphoric, and antinociceptive effects.2
In recent years, kratom has gained popularity in Western parts of the world due to its unique properties and perceived safety as a botanical product. Individuals may use kratom to boost their energy, relieve pain, or treat a wide range of physical or mood problems. Increasingly, kratom is being used by people who abuse opioids to self-manage opioid withdrawal, or for its euphoric effects. But kratom carries several important risks, including addiction, serious adverse effects, and possibly death. In this article, we review the epidemiology and pharmacology of kratom, and provide some guidance for educating patients about this substance.
Widely used but not FDA approved
Although kratom is not regulated or approved by the FDA, 3 to 5 million Americans use it regularly.3 According to an internet survey, kratom users are mostly college-educated, employed white men, age 31 to 50, who take the substance to manage pain or to treat general anxiety and mood disorders.4 Some individuals use kratom as an opioid substitute to reduce symptoms of opioid withdrawal.4
Kratom is available from a wide range of manufacturers in various formulations, including powders, tablets, liquids, and gum. It is sometimes sold in combination with other agents as a single product. Low-cost, over-the-counter kratom products are available as “dietary supplements” in retail stores or online. Although the product packaging sometimes recommends a specific dose, the amount of active ingredients (as well as other agents) is unknown. Kratom is illegal in several states (Box5).
Box
The use and sale of kratom is illegal in several countries, including Australia, Poland, Denmark, Sweden, Malaysia, and Vietnam. In the United States, kratom was legal to grow and purchase in all 50 states until 2015, when the Drug Enforcement Administration (DEA) identified kratom as a “substance of concern.” In August 2016, the DEA submitted a notice of intent to place mitragynine and 7-hydroxymitragynine, 2 alkaloids of kratom that have opioid-like properties, into Schedule I of the Controlled Substance Act; however, due to significant public pressure, the DEA withdrew the request in October 2016.
As of February 2020, kratom was illegal to buy, sell, or use in Wisconsin, Rhode Island, Vermont, Indiana, Arkansas, Alabama, specific counties of some states, and the District of Columbia. Legislation was pending in New York, Missouri, and Louisiana.
Source: Reference 5
The 2 alkaloids of interest
More than 40 alkaloids have been isolated from kratom leaves. The proportions of these alkaloids vary significantly depending on the environment in which the plant is grown, the breeding and harvesting techniques, and the age of the plant.6 Two alkaloids of significant interest are mitragynine (Figure 1) and 7-hydroxymitragynine (Figure 2), both of which are unique to M. speciosa and have opioid-like properties. Administering these alkaloids to morphine-dependent rats resulted in cross-tolerance and precipitated withdrawal when the rats were given naloxone.7 The potency of kratom at the mu opioid receptor has been found to exceed that of morphine.
Competitive binding studies that examined the affinity of mitragynine and 7-hydroxymitragynine at the various opioid receptor subtypes found a preference for the kappa receptors (antagonism), followed by mu (partial agonism), and lastly delta. This profile of mitragynine is very similar to that of buprenorphine.8 The affinity of 7-hydroxymitragynine for the mu receptor (agonism) is significantly greater than that of mitragynine.9 Mitragynine also interacts with noradrenergic and serotonergic pathways by stimulating postsynaptic alpha-2 adrenergic receptors and inhibiting 5-HT2A receptors.9 These properties are responsible for kratom’s ability to manage opioid withdrawal symptoms, which are generally attributed to a hyperactive noradrenergic system. There also is evidence that the hepatic metabolite 7-hydroxymitragynine is important in mediating the analgesic component of mitragynine.10
The initial effects of kratom typically begin within 10 to 20 minutes of consumption, and the full effects are experienced in 30 to 60 minutes.1 The half-life of mitragynine in humans has not yet been determined, but is believed to be relatively short.11 In rats, the half-life of mitragynine is 2 to 3 hours.12 Individuals who use kratom to prevent opioid withdrawal have reported taking it as often as every 6 to 12 hours.13
Continue to: Metabolism of mitragynine...
Metabolism of mitragynine is predominantly carried out through cytochrome P450 (CYP) 3A4, with minor contributions by 2D6 and 2C9. A total of 13 metabolites are produced, including 7-hydroxymitragynine.14 Kratom’s constituents also interact with the CYP system, inhibiting 2C9, 2D6, and 3A4 isoenzymes, and to some extent, 1A2.
Adverse effects can be fatal
An animal study revealed that when administered intravenously, mitragynine and 7-hydroxymitragynine have a similar toxicity profile to heroin.15 When these alkaloids were administered in ascending doses, increases in blood pressure and elevations in liver function tests and creatinine levels from baseline were observed.
Chronic kratom use can result in weight loss, insomnia, constipation, dehydration, skin hyperpigmentation, and extreme fatigue.16 There have also been reports of seizures, delusions, hallucinations, respiratory depression, hepatotoxicity, coma, and death.17,18 An emerging concern is the potential development of fatty liver infiltrates leading to cholestatic liver damage.19-25 One case report described a young man who developed a serum aspartate aminotransferase level of 1,300 IU/L (reference range: 5 to 45 IU/L) and a serum alanine aminotransaminase level of 3,700 IU/L (reference range: 5 to 60 IU/L) after he ingested a kratom product.26 Histologically, the pattern of liver injury mimics primary biliary cholangitis.27
In recent years, calls to poison control centers in the United States related to kratom exposure have risen. Between 2011 and 2017, the number of calls increased from 1 a month to 2 each day.28 The US National Poison Data System has also noted an increase in the number of calls in reference to kratom. It received 2,312 calls from January 2011 through July 2018, with 18 calls occurring in 2011, and 357 within the first 7 months of 2018.29
As of February 2018, the FDA had received reports of 44 deaths associated with kratom.30 There have been reports of fatal overdoses involving kratom, particularly when kratom is co-ingested or used with adulterated and/or combination agents, including one case that involved quetiapine.31-33 There have been reports of deaths believed to be attributed to the use of kratom alone; in one such case, a 35-year-old man experienced a fatal cardiac arrest due to kratom use with no other coingestants.34 Among the reports of deaths in which kratom was the only substance consumed, the mitragynine blood levels of the deceased individuals were found to be higher than the levels associated with individuals who had consumed traditional kratom teas.29
Continue to: There is a lack of quality control...
There is a lack of quality control of commercially available kratom preparations. The FDA has found kratom products that exceeded the level of safe exposure to nickel and lead.35 There have also been reports of Salmonella outbreaks associated with kratom products.36
Detecting kratom use
Mitragynine is a lipophilic alkaloid that is poorly soluble in water37 and eliminated primarily in urine.12 Based on data from treatment center admissions, kratom can be detected in urine samples for 5 to 6 days after use.24,38,39 However, kratom is not detectable by a standard urine toxicology screen; therefore, a high degree of suspicion and special confirmatory testing are necessary. The breakdown products of mitragynine can be detected through gas chromatography coupled with mass spectrometry (GC/MS), liquid chromatography with linear ion trap mass spectrometry, or electrospray tandem mass spectrometry.40-42
A familiar withdrawal syndrome
Abrupt discontinuation of high-dose, long-term kratom use can produce withdrawal symptoms.13 Symptoms of kratom withdrawal resemble those of opioid withdrawal. These include physiological symptoms (mydriasis, nausea, sweating and chills, muscle and body aches, tremors and twitches, diarrhea, rhinorrhea, and lacrimation) and psychological symptoms (insomnia, restlessness, irritability/hostility, fatigue, anxiety, mood disturbances, and hallucinations).13 Symptoms are first noted starting 12 hours after the last use of kratom, and can last up to 7 days.43 Withdrawal intensity has been positively correlated with the daily amount of kratom consumed, as well as the duration and frequency of use.13,16
In 2 case reports, the newborns of women who used kratom during pregnancy experienced neonatal abstinence syndrome.44,45 In these 2 reports, symptoms such as jitteriness, irritability, feeding intolerance, and vomiting emerged on postpartum Day 2. The newborns were admitted to a neonatal ICU and started on a standard opioid protocol with IV morphine and subsequently tapered with an oral formulation over 5 days.44,45
Helping patients who use kratom
The best approach to treating a patient who is experiencing kratom withdrawal is symptomatic management, as would be appropriate for a patient experiencing opioid withdrawal.13 However, the use of agents such as methadone or buprenorphine for patients undergoing kratom withdrawal has not been thoroughly evaluated; very few reports have been published.46,47
Continue to: Similarly, while the standard of care...
Similarly, while the standard of care for treating a patient with opioid use disorder is medication-assisted treatment in combination with counseling and behavioral therapies, there is little evidence on the efficacy of such treatments for patients who use kratom. There are no specific guidelines, and the risk of relapsing to kratom use is high.48,49 Nonetheless, some clinicians have used the same protocol for patients with opioid use disorder to treat patients using kratom, and several published case reports describe this approach.50,51 Because administering buprenorphine/naltrexone to a patient who is dependent on kratom can precipitate withdrawal, clinicians should follow a similar initiation protocol as for opioid dependence when starting a patient on these agents (ie, a washout period with a challenge test would be prudent prior to starting naltrexone).
In cases of kratom overdose, naloxone has been shown to reverse the analgesic effects of mitragynine in rats. However, in a case report of an individual who accidently overdosed on a kratom product, naloxone had a modest effect.52
Bottom Line
Kratom is a botanical substance that acts like a stimulant at low doses and an opioid at higher doses. Patients might use it to treat mood-related symptoms, relieve pain, or manage opioid withdrawal. Kratom use has been associated with the development of addiction as well as a multitude of serious adverse effects, including hepatotoxicity and overdose. Long-term management may be required for a patient who uses kratom.
Related Resources
- White CM. Pharmacologic and clinical assessment of kratom: an update. Am J Health Syst Pharm. 2019;76(23):1915-1925.
- Smith KE, Lawson T. Prevalence and motivations for kratom use in a sample of substance users enrolled in a residential treatment program. Drug Alcohol Depend. 2017;180:340-348.
Drug Brand Names
Buprenorphine • Subutex, Sublocade
Buprenorphine/naltrexone • Suboxone
Methadone • Methadose
Naltrexone • Revia
Naloxone • Narcan
Quetiapine • Seroquel
1. Henningfield JE, Fant RV, Wang DW. The abuse potential of kratom according the 8 factors of the controlled substances act: implications for regulation and research. Psychopharmacology (Berl). 2018;235(2):573-589.
2. Chang-Chien GC, Odonkor CA, Amorapanth P, et al. Is kratom the new ‘legal high’ on the block?: the case of an emerging opioid receptor agonist with substance abuse potential. Pain Physician. 2017;20(1):E195-E198.
3. Penders T, Jones WB. Kratom, a substance of increasing concern [PCSS webinar]. Providers Clinical Support System. November 28, 2018. https://pcssnow.org/event/kratom-a-substance-of-increasing-concern. Accessed January 29, 2020.
4. Grundmann O. Patterns of kratom use and health impact in the US-results from an online survey. Drug Alcohol Depend. 2017;176:63-70.
5. US Drug Enforcement Administration. Drugs of concern. https://www.dea.gov/sites/default/files/sites/getsmartaboutdrugs.com/files/publications/DoA_2017Ed_Updated_6.16.17.pdf#page=84. Updated June 16, 2017. Accessed January 29, 2020.
6. Matsumoto K, Horie S, Ishikawa H, et al. Antinociceptive effect of 7-hydroxymitragynine in mice: discovery of an orally active opioid analgesic from the Thai medicinal herb Mitragyna speciosa. Life Sciences. 2004;74(17):2143-2155.
7. Takayama H. Chemistry and pharmacology of analgesic indole alkaloids from the rubiaceous plant, Mitragyna speciosa. Chem Pharm Bull (Tokyo). 2004;52(8):916-928.
8. Suhaimi FW, Yusoff NH, Hassan R, et al. Neurobiology of kratom and its main alkaloid mitragynine. Brain Res Bull. 2016;126(pt 1):29-40.
9. Prozialeck WC, Jivan JK, Andurkar SV. Pharmacology of kratom: an emerging botanical agent with stimulant, analgesic and opioid-like effects. J Am Osteopath Assoc. 2012;112(12):792-799.
10. Kruegel AC, Uprety R, Grinnell SG, et al. 7-hydroxymitragynine is an active metabolite of mitragynine and a key mediator of its analgesic effects. ACS Cent Sci. 2019;5(6):992-1001.
11. Trakulsrichai S, Sathirakul K, Auparakkitanon S, et al. Pharmacokinetics of mitragynine in man. Drug Des Devel Ther. 2015:9:2421-2429.
12. Warner ML, Kaufman NC, Grundmann O, et al. The pharmacology and toxicology of kratom: from traditional herb to drug of abuse. Intl J Legal Med. 2016;130(1):127-138.
13. Stanciu CN, Gnanasegaram SA, Ahmed S, et al. Kratom withdrawal: a systematic review with case series. J Psychoactive Drugs. 2019;51(1):12-18.
14. Kamble SH, Sharma A, King TI, et al. Metabolite profiling and identification of enzymes responsible for the metabolism of mitragynine, the major alkaloid of Mitragyna speciosa (kratom). Xenobiotica. 2019;49(11):1279-1288.
15. Smith LC, Lin L, Hwang CS, et al. Lateral flow assessment and unanticipated toxicity of kratom. Chem Res Toxicol. 2019;32(1):113-121.
16. Saingam D, Assanangkornchai S, Geater AF, et al. Factor analytical investigation of Krathom (Mitragyna speciosa Korth.) withdrawal syndrome in Thailand. J Psychoactive Drugs. 2016;48(2):76-85.
17. Vicknasingam B, Narayanan S, Beng GT, et al. The informal use of ketum (Mitragyna speciosa) for opioid withdrawal in the northern states of peninsular Malaysia and implications for drug substitution therapy. Int J Drug Policy. 2010;21(4):283-288.
18. Saingam D, Assanangkornchai S, Geater AF, et al. Pattern and consequences of krathom (Mitragyna speciosa Korth.) use among male villagers in southern Thailand: a qualitative study. Int J Drug Policy. 2013;24(4):351-358.
19. Fernandes CT, Iqbal U, Tighe SP, et al. Kratom-induced cholestatic liver injury and its conservative management. J Investig Med High Impact Case Rep. 2019;7:2324709619836138. doi: 10.1177/2324709619836138.
20. Dorman C, Wong M, Khan A. Cholestatic hepatitis from prolonged kratom use: a case report. Hepatology. 2015;61(3):1086-1087.
21. Osborne CS, Overstreet AN, Rockey DC, et al. Drug-induced liver injury caused by kratom use as an alternative pain treatment amid an ongoing opioid epidemic. J Investig Med High Impact Case Rep. 2019;7:2324709619826167. doi: 10.1177/2324709619826167.
22. Mousa MS, Sephien A, Gutierrez J, et al. N-acetylcysteine for acute hepatitis induced by kratom herbal tea. Am J Ther. 2018;25(5):e550-e551.
23. Riverso M, Chang M, Soldevila-Pico C, et al. Histologic characterization of kratom use-associated liver injury. Gastroenterology Res. 2018;11(1):79-82.
24. Kapp FG, Maurer HH, Auwärter V, et al. Intrahepatic cholestasis following abuse of powdered kratom (Mitragyna speciosa). J Med Toxicol. 2011;7(3):227-231.
25. Antony A, Lee TP. Herb-induced liver injury with cholestasis and renal injury secondary to short-term use of kratom (Mitragyna speciosa). Am J Ther. 2019;26(4):e546-e547.
26. Palasamudram Shekar S, Rojas EE, D’Angelo CC, et al. Legally lethal kratom: a herbal supplement with overdose potential. J Psychoactive Drugs. 2019;51(1):28-30.
27. Aldyab M, Ells PF, Bui R, et al. Kratom-induced cholestatic liver injury mimicking anti-mitochondrial antibody-negative primary biliary cholangitis: a case report and review of literature. Gastroenterology Res. 2019;12(4):211-215.
28. Post S, Spiller HA, Chounthirath T. Kratom exposures reported to United States poison control centers: 2011-2017. Clinical Toxicol (Phila). 2019;57(10):847-854.
29. Eggleston W, Stoppacher R, Suen K, et al. Kratom use and toxicities in the United States. Pharmacotherapy. 2019;39(7):775-777.
30. US Food & Drug Administration. Statement from FDA Commissioner Scott Gottlieb, M.D., on the agency’s scientific evidence on the presence of opioid compounds in kratom , underscoring its potential for abuse. https://www.fda.gov/news-events/press-announcements/statement-fda-commissioner-scott-gottlieb-md-agencys-scientific-evidence-presence-opioid-compounds. Published February 6, 2019. Accessed January 29, 2020.
31. Gershman K, Timm K, Frank M, et al. Deaths in Colorado attributed to kratom. N Engl J Med. 2019;380(1):97-98.
32. Kronstrand R, Roman M, Thelander G, et al. Unintentional fatal intoxications with mitragynine and O-desmethyltramadol from the herbal blend krypton. J Anal Toxicol. 2011;35(4):242-247.
33. Hughes RL. Fatal combination of mitragynine and quetiapine - a case report with discussion of a potential herb-drug interaction. Forensic Sci Med Pathol. 2019;15(1):110-113.
34. Abdullah HMA, Haq I, Lamfers R. Cardiac arrest in a young healthy male patient secondary to kratom ingestion: is this ‘legal high’ substance more dangerous than initially thought? BMJ Case Rep. 2019;12(7):pii: e229778. doi: 10.1136/bcr-2019-229778.
35. Laboratory analysis of kratom products for heavy metals. US FDA. https://www.fda.gov/news-events/public-health-focus/laboratory-analysis-kratom-products-heavy-metals. Updated April 3, 2019. Accessed January 29, 2020.
36. FDA investigated multistate outbreak of salmonella infections linked to products reported to contain kratom. US FDA. https://www.fda.gov/food/outbreaks-foodborne-illness/fda-investigated-multistate-outbreak-salmonella-infections-linked-products-reported-contain-kratom. Updated June 29, 2018. Accessed January 14, 2020.
37. Aggarwal G, Robertson E, McKinlay J, et a., Death from kratom toxicity and the possible role of intralipid. J Intensive Care Soc. 2018;19(1):61-63.
38. Drug Facts. Kratom. Confirm Biosciences. https://www.confirmbiosciences.com/knowledge/drug-facts/kratom/. Accessed January 14, 2020.
39. Grinspoon P. How long does kratom stay in the system? Addiction Resource. https://addictionresource.com/drugs/kratom/how-long-kratom-stay-in-your-system/. Updated December 18, 2019. Accessed January 29, 2020.
40. Kaewklum D, Kaewklum M, Pootrakronchai R, et al. Detection of mitragynine and its metaboilite in urine following ingestion of leaves of Mitragyna speciosa korth. Recent Advances in Doping Analysis (13). Proceedings of the Manfred Donike Workshop, 23rd Cologne Workshop on Dope Analysis. 2005:403-406.
41. Lu S, Tran BN, Nelsen JL, et al. Quantitative analysis of mitragynine in human urine by high performance liquid chromatography-tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci. 2009;877(24):2499-2505.
42. Philipp AA, Wissenbach DK, Zoerntlein SW, et al. Studies on the metabolism of mitragynine, the main alkaloid of the herbal drug kratom, in rat and human urine using liquid chromatography-linear ion trap mass spectrometry. J Mass Spectrom. 2009;44(8):1249-1261.
43. Manda VK, Bharathi A, Ali Z, et al. Evaluation of in vitro absorption, distribution, metabolism, and excretion (ADME) properties of mitragynine, 7-hydroxymitragynine, and mitraphylline. Planta Med. 2014;80(7):568-576.
44. Davidson L, Rawat M, Stojanovski S, et al. Natural drugs, not so natural effects: neonatal abstinence syndrome secondary to ‘kratom‘. J Neonatal Perinatal Med. 2019;12(1):109-112.
45. Mackay L, Abrahams R. Novel case of maternal and neonatal kratom dependence and withdrawal. Can Fam Physician. 2018;64(2):121-122.
46. McWhirter L, Morris S. A case report of inpatient detoxification after kratom (Mitragyna speciosa) dependence. Eur Addict Res. 2010;16(4):229-231.
47. Galbis-Reig David. A case report of kratom addiction and withdrawal. WMJ. 2016;115(1):49-52; quiz 53.
48. Singh D, Müller CP, Vicknasingam BK. Kratom (Mitragyna speciose) dependence, withdrawal symptoms and craving in regular users. Drug Alcohol Depend. 2014;139:132-137.
49. Singh D, Müller CP, Vicknasingam, et al. Social functioning of kratom (Mitragyna speciosa) users in Malaysia. J Psychoactive Drugs. 2015;47(2):125-131.
50. Khazaeli A, Jerry JM, Vazirian M. Treatment of kratom withdrawal and addiction with buprenorphine. J Addict Med. 2018;12(6):493-495.
51. Buresh M. Treatment of kratom dependence with buprenorphine-naloxone maintenance. J Addict Med. 2018;12(6):481-483.
52. Overbeek DL, Abraham J, Munzer BW. Kratom (mitragynine) ingestion requiring naloxone reversal. Clin Pract Cases Emerg Med. 2019;3(1):24-26.
1. Henningfield JE, Fant RV, Wang DW. The abuse potential of kratom according the 8 factors of the controlled substances act: implications for regulation and research. Psychopharmacology (Berl). 2018;235(2):573-589.
2. Chang-Chien GC, Odonkor CA, Amorapanth P, et al. Is kratom the new ‘legal high’ on the block?: the case of an emerging opioid receptor agonist with substance abuse potential. Pain Physician. 2017;20(1):E195-E198.
3. Penders T, Jones WB. Kratom, a substance of increasing concern [PCSS webinar]. Providers Clinical Support System. November 28, 2018. https://pcssnow.org/event/kratom-a-substance-of-increasing-concern. Accessed January 29, 2020.
4. Grundmann O. Patterns of kratom use and health impact in the US-results from an online survey. Drug Alcohol Depend. 2017;176:63-70.
5. US Drug Enforcement Administration. Drugs of concern. https://www.dea.gov/sites/default/files/sites/getsmartaboutdrugs.com/files/publications/DoA_2017Ed_Updated_6.16.17.pdf#page=84. Updated June 16, 2017. Accessed January 29, 2020.
6. Matsumoto K, Horie S, Ishikawa H, et al. Antinociceptive effect of 7-hydroxymitragynine in mice: discovery of an orally active opioid analgesic from the Thai medicinal herb Mitragyna speciosa. Life Sciences. 2004;74(17):2143-2155.
7. Takayama H. Chemistry and pharmacology of analgesic indole alkaloids from the rubiaceous plant, Mitragyna speciosa. Chem Pharm Bull (Tokyo). 2004;52(8):916-928.
8. Suhaimi FW, Yusoff NH, Hassan R, et al. Neurobiology of kratom and its main alkaloid mitragynine. Brain Res Bull. 2016;126(pt 1):29-40.
9. Prozialeck WC, Jivan JK, Andurkar SV. Pharmacology of kratom: an emerging botanical agent with stimulant, analgesic and opioid-like effects. J Am Osteopath Assoc. 2012;112(12):792-799.
10. Kruegel AC, Uprety R, Grinnell SG, et al. 7-hydroxymitragynine is an active metabolite of mitragynine and a key mediator of its analgesic effects. ACS Cent Sci. 2019;5(6):992-1001.
11. Trakulsrichai S, Sathirakul K, Auparakkitanon S, et al. Pharmacokinetics of mitragynine in man. Drug Des Devel Ther. 2015:9:2421-2429.
12. Warner ML, Kaufman NC, Grundmann O, et al. The pharmacology and toxicology of kratom: from traditional herb to drug of abuse. Intl J Legal Med. 2016;130(1):127-138.
13. Stanciu CN, Gnanasegaram SA, Ahmed S, et al. Kratom withdrawal: a systematic review with case series. J Psychoactive Drugs. 2019;51(1):12-18.
14. Kamble SH, Sharma A, King TI, et al. Metabolite profiling and identification of enzymes responsible for the metabolism of mitragynine, the major alkaloid of Mitragyna speciosa (kratom). Xenobiotica. 2019;49(11):1279-1288.
15. Smith LC, Lin L, Hwang CS, et al. Lateral flow assessment and unanticipated toxicity of kratom. Chem Res Toxicol. 2019;32(1):113-121.
16. Saingam D, Assanangkornchai S, Geater AF, et al. Factor analytical investigation of Krathom (Mitragyna speciosa Korth.) withdrawal syndrome in Thailand. J Psychoactive Drugs. 2016;48(2):76-85.
17. Vicknasingam B, Narayanan S, Beng GT, et al. The informal use of ketum (Mitragyna speciosa) for opioid withdrawal in the northern states of peninsular Malaysia and implications for drug substitution therapy. Int J Drug Policy. 2010;21(4):283-288.
18. Saingam D, Assanangkornchai S, Geater AF, et al. Pattern and consequences of krathom (Mitragyna speciosa Korth.) use among male villagers in southern Thailand: a qualitative study. Int J Drug Policy. 2013;24(4):351-358.
19. Fernandes CT, Iqbal U, Tighe SP, et al. Kratom-induced cholestatic liver injury and its conservative management. J Investig Med High Impact Case Rep. 2019;7:2324709619836138. doi: 10.1177/2324709619836138.
20. Dorman C, Wong M, Khan A. Cholestatic hepatitis from prolonged kratom use: a case report. Hepatology. 2015;61(3):1086-1087.
21. Osborne CS, Overstreet AN, Rockey DC, et al. Drug-induced liver injury caused by kratom use as an alternative pain treatment amid an ongoing opioid epidemic. J Investig Med High Impact Case Rep. 2019;7:2324709619826167. doi: 10.1177/2324709619826167.
22. Mousa MS, Sephien A, Gutierrez J, et al. N-acetylcysteine for acute hepatitis induced by kratom herbal tea. Am J Ther. 2018;25(5):e550-e551.
23. Riverso M, Chang M, Soldevila-Pico C, et al. Histologic characterization of kratom use-associated liver injury. Gastroenterology Res. 2018;11(1):79-82.
24. Kapp FG, Maurer HH, Auwärter V, et al. Intrahepatic cholestasis following abuse of powdered kratom (Mitragyna speciosa). J Med Toxicol. 2011;7(3):227-231.
25. Antony A, Lee TP. Herb-induced liver injury with cholestasis and renal injury secondary to short-term use of kratom (Mitragyna speciosa). Am J Ther. 2019;26(4):e546-e547.
26. Palasamudram Shekar S, Rojas EE, D’Angelo CC, et al. Legally lethal kratom: a herbal supplement with overdose potential. J Psychoactive Drugs. 2019;51(1):28-30.
27. Aldyab M, Ells PF, Bui R, et al. Kratom-induced cholestatic liver injury mimicking anti-mitochondrial antibody-negative primary biliary cholangitis: a case report and review of literature. Gastroenterology Res. 2019;12(4):211-215.
28. Post S, Spiller HA, Chounthirath T. Kratom exposures reported to United States poison control centers: 2011-2017. Clinical Toxicol (Phila). 2019;57(10):847-854.
29. Eggleston W, Stoppacher R, Suen K, et al. Kratom use and toxicities in the United States. Pharmacotherapy. 2019;39(7):775-777.
30. US Food & Drug Administration. Statement from FDA Commissioner Scott Gottlieb, M.D., on the agency’s scientific evidence on the presence of opioid compounds in kratom , underscoring its potential for abuse. https://www.fda.gov/news-events/press-announcements/statement-fda-commissioner-scott-gottlieb-md-agencys-scientific-evidence-presence-opioid-compounds. Published February 6, 2019. Accessed January 29, 2020.
31. Gershman K, Timm K, Frank M, et al. Deaths in Colorado attributed to kratom. N Engl J Med. 2019;380(1):97-98.
32. Kronstrand R, Roman M, Thelander G, et al. Unintentional fatal intoxications with mitragynine and O-desmethyltramadol from the herbal blend krypton. J Anal Toxicol. 2011;35(4):242-247.
33. Hughes RL. Fatal combination of mitragynine and quetiapine - a case report with discussion of a potential herb-drug interaction. Forensic Sci Med Pathol. 2019;15(1):110-113.
34. Abdullah HMA, Haq I, Lamfers R. Cardiac arrest in a young healthy male patient secondary to kratom ingestion: is this ‘legal high’ substance more dangerous than initially thought? BMJ Case Rep. 2019;12(7):pii: e229778. doi: 10.1136/bcr-2019-229778.
35. Laboratory analysis of kratom products for heavy metals. US FDA. https://www.fda.gov/news-events/public-health-focus/laboratory-analysis-kratom-products-heavy-metals. Updated April 3, 2019. Accessed January 29, 2020.
36. FDA investigated multistate outbreak of salmonella infections linked to products reported to contain kratom. US FDA. https://www.fda.gov/food/outbreaks-foodborne-illness/fda-investigated-multistate-outbreak-salmonella-infections-linked-products-reported-contain-kratom. Updated June 29, 2018. Accessed January 14, 2020.
37. Aggarwal G, Robertson E, McKinlay J, et a., Death from kratom toxicity and the possible role of intralipid. J Intensive Care Soc. 2018;19(1):61-63.
38. Drug Facts. Kratom. Confirm Biosciences. https://www.confirmbiosciences.com/knowledge/drug-facts/kratom/. Accessed January 14, 2020.
39. Grinspoon P. How long does kratom stay in the system? Addiction Resource. https://addictionresource.com/drugs/kratom/how-long-kratom-stay-in-your-system/. Updated December 18, 2019. Accessed January 29, 2020.
40. Kaewklum D, Kaewklum M, Pootrakronchai R, et al. Detection of mitragynine and its metaboilite in urine following ingestion of leaves of Mitragyna speciosa korth. Recent Advances in Doping Analysis (13). Proceedings of the Manfred Donike Workshop, 23rd Cologne Workshop on Dope Analysis. 2005:403-406.
41. Lu S, Tran BN, Nelsen JL, et al. Quantitative analysis of mitragynine in human urine by high performance liquid chromatography-tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci. 2009;877(24):2499-2505.
42. Philipp AA, Wissenbach DK, Zoerntlein SW, et al. Studies on the metabolism of mitragynine, the main alkaloid of the herbal drug kratom, in rat and human urine using liquid chromatography-linear ion trap mass spectrometry. J Mass Spectrom. 2009;44(8):1249-1261.
43. Manda VK, Bharathi A, Ali Z, et al. Evaluation of in vitro absorption, distribution, metabolism, and excretion (ADME) properties of mitragynine, 7-hydroxymitragynine, and mitraphylline. Planta Med. 2014;80(7):568-576.
44. Davidson L, Rawat M, Stojanovski S, et al. Natural drugs, not so natural effects: neonatal abstinence syndrome secondary to ‘kratom‘. J Neonatal Perinatal Med. 2019;12(1):109-112.
45. Mackay L, Abrahams R. Novel case of maternal and neonatal kratom dependence and withdrawal. Can Fam Physician. 2018;64(2):121-122.
46. McWhirter L, Morris S. A case report of inpatient detoxification after kratom (Mitragyna speciosa) dependence. Eur Addict Res. 2010;16(4):229-231.
47. Galbis-Reig David. A case report of kratom addiction and withdrawal. WMJ. 2016;115(1):49-52; quiz 53.
48. Singh D, Müller CP, Vicknasingam BK. Kratom (Mitragyna speciose) dependence, withdrawal symptoms and craving in regular users. Drug Alcohol Depend. 2014;139:132-137.
49. Singh D, Müller CP, Vicknasingam, et al. Social functioning of kratom (Mitragyna speciosa) users in Malaysia. J Psychoactive Drugs. 2015;47(2):125-131.
50. Khazaeli A, Jerry JM, Vazirian M. Treatment of kratom withdrawal and addiction with buprenorphine. J Addict Med. 2018;12(6):493-495.
51. Buresh M. Treatment of kratom dependence with buprenorphine-naloxone maintenance. J Addict Med. 2018;12(6):481-483.
52. Overbeek DL, Abraham J, Munzer BW. Kratom (mitragynine) ingestion requiring naloxone reversal. Clin Pract Cases Emerg Med. 2019;3(1):24-26.
Critical care admissions up for pediatric opioid poisonings
ORLANDO – The proportion of children and adolescents admitted to critical care for serious poisonings has increased in recent years, according to authors of a study of more than 750,000 reported opioid exposures.
Critical care units were involved in 10% of pediatric opioid poisoning cases registered in 2015-2018, up from 7% in 2005-2009, reported Megan E. Land, MD, of Emory University, Atlanta, and coinvestigators.
Attempted suicide has represented an increasingly large proportion of pediatric opioid poisonings from 2005 to 2018, according to the researchers, based on retrospective analysis of cases reported to U.S. poison centers.
Mortality related to these pediatric poisonings increased over time, and among children and adolescents admitted to a pediatric ICU, CPR and naloxone use also increased over time, Dr. Land and associates noted.
These said Dr. Land, who presented the findings at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
“I think that this really requires a two-pronged approach,” she explained. “One is that we need to increase mental health resources for kids to address adolescent suicidality, and secondly, we need to decrease access to opioids in the hands of pediatric patients by decreasing prescribing and then also getting those that are unused out of the homes.”
Jeffrey Zimmerman, MD, past president of SCCM, said these findings on pediatric opioid poisonings represent the “iceberg tip” of a much larger societal issue that has impacts well beyond critical care.
“I think acutely, we’re well equipped to deal with the situation in terms of interventions,” Dr. Zimmerman said in an interview. “The bigger issue is dealing with what happens afterward, when the patient leaves the ICU in the hospital.”
When the issue is chronic opioid use among adolescents or children, critical care specialists can help by initiating opioid tapering in the hospital setting, rather than allowing the complete weaning process to play out at home, he said.
All clinicians can help prevent future injury by asking questions of the child and family to ensure that any opiates and other prescription medications at home are locked up, he added.
“These aren’t very glamorous things, but they’re common sense, and there’s more need for this common sense now than there ever has been,” Dr. Zimmerman concluded.
The study by Dr. Land and colleagues included data on primary opioid ingestions registered at 55 poison control centers in the United States. They assessed trends over three time periods: 2005-2009, 2010-2014, and 2015-2018.
They found that children under 19 years of age accounted for 28% of the 753,592 opioid poisonings reported over that time period.
The overall number of reported opioid poisonings among children declined somewhat since about 2010. However, the proportion admitted to a critical care unit increased from 7% in the 2005-2009 period to 10% in the 2015-2018 period, said Dr. Land, who added that the probability of a moderate or major effect increased by 0.55% and 0.11% per year, respectively, over the 14 years studied.
Mortality – 0.21% overall – increased from 0.18% in the earliest era to 0.28% in the most recent era, according to the investigators.
Suicidal intent increased from 14% in the earliest era to 21% in the most recent era, and was linked to near tenfold odds of undergoing a pediatric ICU procedure, Dr. Land and colleagues reported.
Among those children admitted to a pediatric ICU, use of CPR increased from 1% to 3% in the earliest and latest time periods, respectively; likewise, naloxone administration increased from 42% to 51% over those two time periods. By contrast, there was no change in use of mechanical ventilation (12%) or vasopressors (3%) over time, they added.
The opioids most commonly linked to pediatric ICU procedures were fentanyl (odds ratio, 12), heroin (OR, 11), and methadone (OR, 15).
Some funding for the study came from the Georgia Poison Center. Dr. Land had no disclosures relevant to the research.
SOURCE: Land M et al. Crit Care Med. 2020 doi: 10.1097/01.ccm.0000618708.38414.ea.
ORLANDO – The proportion of children and adolescents admitted to critical care for serious poisonings has increased in recent years, according to authors of a study of more than 750,000 reported opioid exposures.
Critical care units were involved in 10% of pediatric opioid poisoning cases registered in 2015-2018, up from 7% in 2005-2009, reported Megan E. Land, MD, of Emory University, Atlanta, and coinvestigators.
Attempted suicide has represented an increasingly large proportion of pediatric opioid poisonings from 2005 to 2018, according to the researchers, based on retrospective analysis of cases reported to U.S. poison centers.
Mortality related to these pediatric poisonings increased over time, and among children and adolescents admitted to a pediatric ICU, CPR and naloxone use also increased over time, Dr. Land and associates noted.
These said Dr. Land, who presented the findings at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
“I think that this really requires a two-pronged approach,” she explained. “One is that we need to increase mental health resources for kids to address adolescent suicidality, and secondly, we need to decrease access to opioids in the hands of pediatric patients by decreasing prescribing and then also getting those that are unused out of the homes.”
Jeffrey Zimmerman, MD, past president of SCCM, said these findings on pediatric opioid poisonings represent the “iceberg tip” of a much larger societal issue that has impacts well beyond critical care.
“I think acutely, we’re well equipped to deal with the situation in terms of interventions,” Dr. Zimmerman said in an interview. “The bigger issue is dealing with what happens afterward, when the patient leaves the ICU in the hospital.”
When the issue is chronic opioid use among adolescents or children, critical care specialists can help by initiating opioid tapering in the hospital setting, rather than allowing the complete weaning process to play out at home, he said.
All clinicians can help prevent future injury by asking questions of the child and family to ensure that any opiates and other prescription medications at home are locked up, he added.
“These aren’t very glamorous things, but they’re common sense, and there’s more need for this common sense now than there ever has been,” Dr. Zimmerman concluded.
The study by Dr. Land and colleagues included data on primary opioid ingestions registered at 55 poison control centers in the United States. They assessed trends over three time periods: 2005-2009, 2010-2014, and 2015-2018.
They found that children under 19 years of age accounted for 28% of the 753,592 opioid poisonings reported over that time period.
The overall number of reported opioid poisonings among children declined somewhat since about 2010. However, the proportion admitted to a critical care unit increased from 7% in the 2005-2009 period to 10% in the 2015-2018 period, said Dr. Land, who added that the probability of a moderate or major effect increased by 0.55% and 0.11% per year, respectively, over the 14 years studied.
Mortality – 0.21% overall – increased from 0.18% in the earliest era to 0.28% in the most recent era, according to the investigators.
Suicidal intent increased from 14% in the earliest era to 21% in the most recent era, and was linked to near tenfold odds of undergoing a pediatric ICU procedure, Dr. Land and colleagues reported.
Among those children admitted to a pediatric ICU, use of CPR increased from 1% to 3% in the earliest and latest time periods, respectively; likewise, naloxone administration increased from 42% to 51% over those two time periods. By contrast, there was no change in use of mechanical ventilation (12%) or vasopressors (3%) over time, they added.
The opioids most commonly linked to pediatric ICU procedures were fentanyl (odds ratio, 12), heroin (OR, 11), and methadone (OR, 15).
Some funding for the study came from the Georgia Poison Center. Dr. Land had no disclosures relevant to the research.
SOURCE: Land M et al. Crit Care Med. 2020 doi: 10.1097/01.ccm.0000618708.38414.ea.
ORLANDO – The proportion of children and adolescents admitted to critical care for serious poisonings has increased in recent years, according to authors of a study of more than 750,000 reported opioid exposures.
Critical care units were involved in 10% of pediatric opioid poisoning cases registered in 2015-2018, up from 7% in 2005-2009, reported Megan E. Land, MD, of Emory University, Atlanta, and coinvestigators.
Attempted suicide has represented an increasingly large proportion of pediatric opioid poisonings from 2005 to 2018, according to the researchers, based on retrospective analysis of cases reported to U.S. poison centers.
Mortality related to these pediatric poisonings increased over time, and among children and adolescents admitted to a pediatric ICU, CPR and naloxone use also increased over time, Dr. Land and associates noted.
These said Dr. Land, who presented the findings at the Critical Care Congress sponsored by the Society of Critical Care Medicine.
“I think that this really requires a two-pronged approach,” she explained. “One is that we need to increase mental health resources for kids to address adolescent suicidality, and secondly, we need to decrease access to opioids in the hands of pediatric patients by decreasing prescribing and then also getting those that are unused out of the homes.”
Jeffrey Zimmerman, MD, past president of SCCM, said these findings on pediatric opioid poisonings represent the “iceberg tip” of a much larger societal issue that has impacts well beyond critical care.
“I think acutely, we’re well equipped to deal with the situation in terms of interventions,” Dr. Zimmerman said in an interview. “The bigger issue is dealing with what happens afterward, when the patient leaves the ICU in the hospital.”
When the issue is chronic opioid use among adolescents or children, critical care specialists can help by initiating opioid tapering in the hospital setting, rather than allowing the complete weaning process to play out at home, he said.
All clinicians can help prevent future injury by asking questions of the child and family to ensure that any opiates and other prescription medications at home are locked up, he added.
“These aren’t very glamorous things, but they’re common sense, and there’s more need for this common sense now than there ever has been,” Dr. Zimmerman concluded.
The study by Dr. Land and colleagues included data on primary opioid ingestions registered at 55 poison control centers in the United States. They assessed trends over three time periods: 2005-2009, 2010-2014, and 2015-2018.
They found that children under 19 years of age accounted for 28% of the 753,592 opioid poisonings reported over that time period.
The overall number of reported opioid poisonings among children declined somewhat since about 2010. However, the proportion admitted to a critical care unit increased from 7% in the 2005-2009 period to 10% in the 2015-2018 period, said Dr. Land, who added that the probability of a moderate or major effect increased by 0.55% and 0.11% per year, respectively, over the 14 years studied.
Mortality – 0.21% overall – increased from 0.18% in the earliest era to 0.28% in the most recent era, according to the investigators.
Suicidal intent increased from 14% in the earliest era to 21% in the most recent era, and was linked to near tenfold odds of undergoing a pediatric ICU procedure, Dr. Land and colleagues reported.
Among those children admitted to a pediatric ICU, use of CPR increased from 1% to 3% in the earliest and latest time periods, respectively; likewise, naloxone administration increased from 42% to 51% over those two time periods. By contrast, there was no change in use of mechanical ventilation (12%) or vasopressors (3%) over time, they added.
The opioids most commonly linked to pediatric ICU procedures were fentanyl (odds ratio, 12), heroin (OR, 11), and methadone (OR, 15).
Some funding for the study came from the Georgia Poison Center. Dr. Land had no disclosures relevant to the research.
SOURCE: Land M et al. Crit Care Med. 2020 doi: 10.1097/01.ccm.0000618708.38414.ea.
REPORTING FROM CCC49