User login
Community-Acquired Pneumonia: Treatment
Initial management decisions for patients with community-acquired pneumonia (CAP) will depend on severity of infection, with need for hospitalization being one of the first decisions. Because empiric antibiotics are the mainstay of treatment and the causative organisms are seldom identified, underlying medical conditions and epidemiologic risk factors are considered when selecting an empiric regimen. As with other infections, duration of therapy is not standardized, but rather is guided by clinical improvement. Prevention of pneumonia centers around vaccination and smoking cessation. This article, the second in a 2-part review of CAP in adults, focuses on site of care decision, empiric and directed therapies, length of treatment, and prevention strategies. Evaluation and diagnosis of CAP are discussed in a separate article.
Site of Care Decision
For patients diagnosed with CAP, the clinician must decide whether treatment will be done in an outpatient or inpatient setting, and for those in the inpatient setting, whether they can safely be treated on the general medical ward or in the intensive care unit (ICU). Two common scoring systems that can be used to aid the clinician in determining severity of the infection and guide site-of-care decisions are the Pneumonia Severity Index (PSI) and CURB-65 scores.
The PSI score uses 20 different parameters, including comorbidities, laboratory parameters, and radiographic findings, to stratify patients into 5 mortality risk classes.1 On the basis of associated mortality rates, it has been suggested that risk class I and II patients should be treated as outpatients, risk class III patients should be treated in an observation unit or with a short hospitalization, and risk class IV and V patients should be treated as inpatients.1
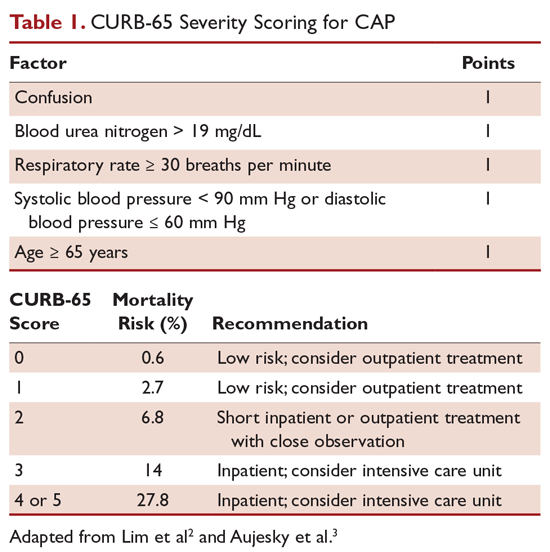
The CURB-65 method of risk stratification is based on 5 clinical parameters: confusion, urea level, respiratory rate, systolic blood pressure, and age ≥ 65 years (Table 1).2,3 A modification to the CURB-65 algorithm tool was CRB-65, which excludes urea nitrogen, making it optimal for making determinations in a clinic-based setting. It should be emphasized that these tools do not take into account other factors that should be used in determining location of treatment, such as stable home, mental illness, or concerns about compliance with medications. In many instances, it is these factors that preclude low-risk patients from being treated as outpatients.4,5 Similarly, these scoring systems have not been validated for immunocompromised patients or those who would qualify as having health care–associated pneumonia.
Patients with CURB-65 scores of 4 or 5 are considered to have severe pneumonia, and admission to the ICU should be considered for these patients. Aside from the CURB-65 score, anyone requiring vasopressor support or mechanical ventilation merits admission to the ICU.6 American Thoracic Society (ATS) and Infectious Diseases Society of America (IDSA) guidelines also recommend the use of “minor criteria” for making ICU admission decisions; these include respiratory rate ≥ 30 breaths/minute, PaO2 fraction ≤ 250 mm Hg, multilobar infiltrates, confusion, blood urea nitrogen ≥ 20 mg/dL, leukopenia, thrombocytopenia, hypothermia, and hypotension.6 These factors are associated with increased mortality due to CAP, and ICU admission is indicated if 3 of the minor criteria for severe CAP are present.
Another clinical calculator that can be used for assessing severity of CAP is SMART-COP (systolic blood pressure, multilobar chest radiography involvement, albumin level, respiratory rate, tachycardia, confusion, oxygenation and arterial pH).7 This scoring system uses 8 weighted criteria to predict which patients will require intensive respiratory or vasopressor support. SMART-COP has a sensitivity of 79% and a specificity of 64% in predicting ICU admission, whereas CURB-65 has a pooled sensitivity of 57.2% and specificity of 77.2%.8
Antibiotic Therapy
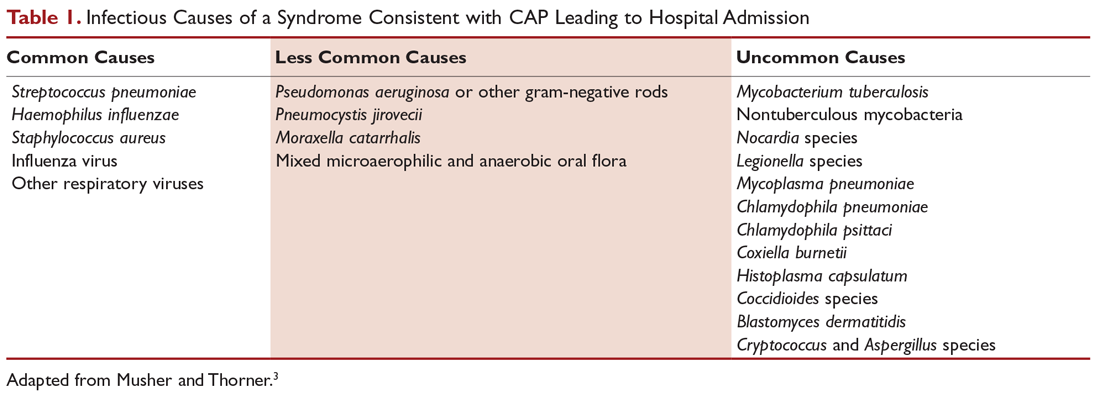
Antibiotics are the mainstay of treatment for CAP, with the majority of patients with CAP treated empirically taking into account the site of care, likely pathogen, and antimicrobial resistance issues. Patients with pneumonia who are treated as outpatients usually respond well to empiric antibiotic treatment, and a causative pathogen is not usually sought. Patients who are hospitalized for treatment of CAP usually receive empiric antibiotic on admission. Once the etiology has been determined by microbiologic or serologic means, antimicrobial therapy should be adjusted accordingly. A CDC study found that the burden of viral etiologies was higher than previously thought, with rhinovirus and influenza accounting for 15% of cases and Streptococcus pneumoniae for only 5%.9 This study highlighted the fact that despite advances in molecular techniques, no pathogen is identified for most patients with pneumonia.9 Given the lack of discernable pathogens in the majority of cases, patients should continue to be treated with antibiotics unless a nonbacterial etiology is found.
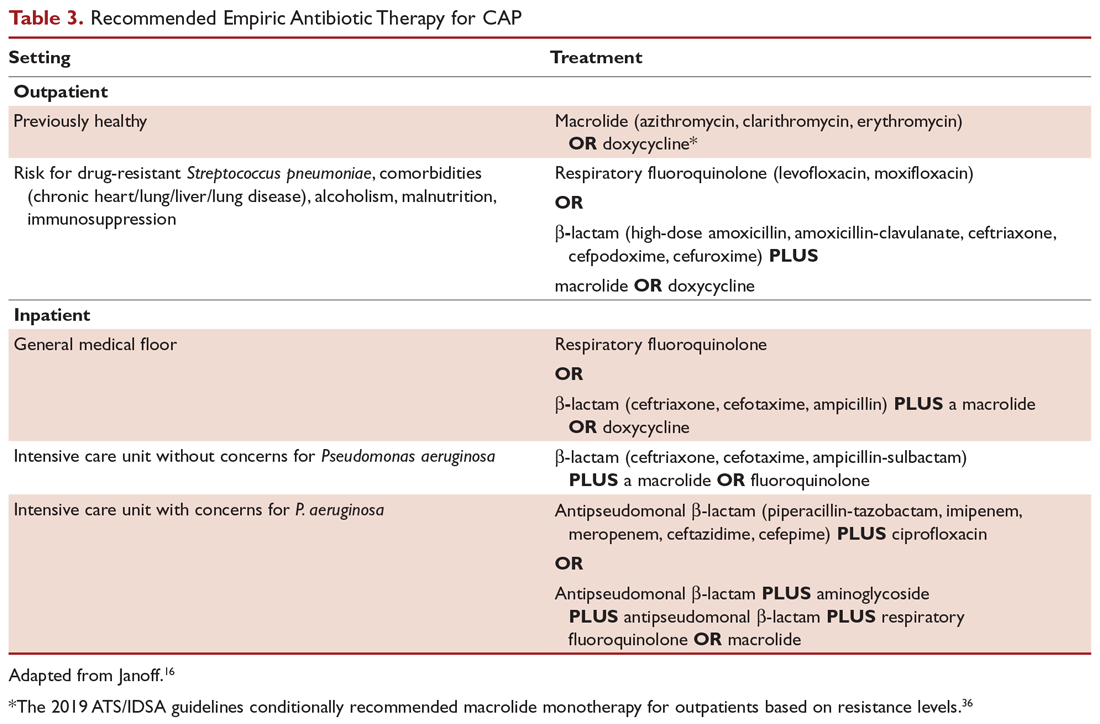
Outpatients without comorbidities or risk factors for drug-resistant S. pneumoniae (Table 2)10 can be treated with monotherapy. Hospitalized patients are usually treated with combination intravenous therapy, although non-ICU patients who receive a respiratory fluoroquinolone can be treated orally.
As previously mentioned, antibiotic therapy is typically empiric, since neither clinical features nor radiographic features are sufficient to include or exclude infectious etiologies. Epidemiologic risk factors should be considered and, in certain cases, antimicrobial coverage should be expanded to include those entities; for example, treatment of anaerobes in the setting of lung abscess and antipseudomonal antibiotics for patients with bronchiectasis.
Of concern in the treatment of CAP is the increased prevalence of antimicrobial resistance among S. pneumoniae. The IDSA guidelines report that drug-resistant S. pneumoniae is more common in persons aged < 2 or > 65 years, and those with β-lactam therapy within the previous 3 months, alcoholism, medical comorbidities, immunosuppressive illness or therapy, or exposure to a child who attends a day care center.6
Staphylococcus aureus should be considered during influenza outbreaks, with either vancomycin or linezolid being the recommended agents in the setting of methicillin-resistant S. aureus (MRSA). In a study comparing vancomycin versus linezolid for nosocomial pneumonia, the all-cause 60-day mortality was similar for both agents.11 Daptomycin, another agent used against MRSA, is not indicated in the setting of pneumonia because daptomycin binds to surfactant, yielding it ineffective in the treatment of pneumonia.12 Ceftaroline is a newer cephalosporin with activity against MRSA; its role in treatment of community-acquired MRSA pneumonia has not been fully elucidated, but it appears to be a useful agent for this indication.13,14 Similarly, other agents known to have antibacterial properties against MRSA, such as trimethoprim/sulfamethoxazole and doxycycline, have not been studied for this indication. Clindamycin has been used to treat MRSA in children, and IDSA guidelines on the treatment of MRSA list clindamycin as an alternative15 if MRSA is known to be sensitive.
A summary of recommended empiric antibiotic therapy is presented in Table 3.16
Three antibiotics were approved by the US Food and Drug Administration (FDA) for the treatment of CAP after the release of the IDSA/ATS guidelines in 2007. Ceftaroline fosamil is a fifth-generation cephalosporin that has coverage for MRSA and was approved in November 2010.17 It can only be administered intravenously and needs dose adjustment for renal function. Omadacycline is a new tetracycline that was approved by the FDA in October 2018.18 It is available in both intravenous injectable and oral forms. No dose adjustment is needed for renal function. Lefamulin is a first-in-class novel pleuromutilin antibiotic which was FDA-approved in August 2019. It can be administered intravenously or orally, with no dosage adjustment necessary in patients with renal impairment.19
Antibiotic Therapy for Selected Pathogens
Streptococcus pneumoniae
Patients with pneumococcal pneumonia who have penicillin-susceptible strains can be treated with intravenous penicillin (2 or 3 million units every 4 hours) or ceftriaxone. Once a patient meets criteria of stability, they can then be transitioned to oral penicillin, amoxicillin, or clarithromycin. Those with strains with reduced susceptibility can still be treated with penicillin, but at a higher dose (4 million units intravenously [IV] every 4 hours), or a third-generation cephalosporin. Those whose pneumococcal pneumonia is complicated by bacteremia will benefit from dual therapy if severely ill, requiring ICU monitoring. Those not severely ill can be treated with monotherapy.20
Staphylococcus aureus
Staphylococcus aureus is more commonly associated with hospital-acquired pneumonia, but it may also be seen during the influenza season and in those with severe necrotizing CAP. Both linezolid and vancomycin can be used to treat MRSA CAP. As noted, ceftaroline has activity against MRSA and is approved for treatment of CAP, but is not approved by the FDA for MRSA CAP treatment. Similarly, tigecycline is approved for CAP and has activity against MRSA, but is not approved for MRSA CAP. Moreover, the FDA has warned of increased risk of death with tigecycline and has a black box warning to that effect.21
Legionella
Legionellosis can be treated with tetra¬cyclines, macrolides, or fluoroquinolones. For non-immunocompromised patients with mild pneumonia, any of the listed antibiotics is considered appropriate. However, patients with severe infection or those with immunosuppression should be treated with either levofloxacin or azithromycin for 7 to 10 days.22
Chlamydophila pneumoniae
As with other atypical organisms, Chlamydophila pneumoniae can be treated with doxycycline, a macrolide, or respiratory fluoroquinolones. However, length of therapy varies by regimen used; treating with doxycycline 100 mg twice daily generally requires 14 to 21 days, whereas moxifloxacin 400 mg daily requires 10 days.23
Mycoplasma pneumoniae
As with C. pneumoniae, length of therapy of Mycoplasma pneumoniae varies by which antimicrobial regimen is used. Shortest courses are seen with the use of macrolides for 5 days, whereas 14 days is considered standard for doxycycline or a respiratory fluoroquinolone.24 It should be noted that there has been increasingly documented resistance to macrolides, with known resistance of 8.2% in the United States.25
Duration of Treatment
Most patients with CAP respond to appropriate therapy within 72 hours. IDSA/ATS guidelines recommend that patients with routine cases of CAP be treated for a minimum of 5 days. Despite this, many patients are treated for an excessive amount of time, with over 70% of patients reported to have received antibiotics for more than 10 days for uncomplicated CAP.26 There are instances that require longer courses of antibiotics, including cases caused by Pseudomonas aeruginosa, S. aureus, and Legionella species and patients with lung abscesses or necrotizing infections, among others.27
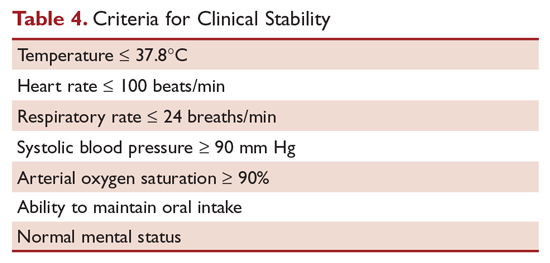
Hospitalized patients do not need to be monitored for an additional day once they have reached clinical stability (Table 4), are able to maintain oral intake, and have normal mentation, provided that other comorbidities are stable and social needs have been met.6 C-reactive protein (CRP) level has been postulated as an additional measure of stability, specifically monitoring for a greater than 50% reduction in CRP; however, this was validated only for those with complicated pneumonia.28 Patients discharged from the hospital with instability have higher risk of readmission or death.29
Transition to Oral Therapy
IDSA/ATS guidelines6 recommend that patients should be transitioned from intravenous to oral antibiotics when they are improving clinically, have stable vital signs, and are able to ingest food/fluids and medications.
Management of Nonresponders
Although the majority of patients respond to antibiotics within 72 hours, treatment failure occurs in up to 15% of patients.15 Nonresponding pneumonia is generally seen in 2 patterns: worsening of clinical status despite empiric antibiotics or delay in achieving clinical stability, as defined in Table 4, after 72 hours of treatment.30 Risk factors associated with nonresponding pneumonia31 are:
- Radiographic: multilobar infiltrates, pleural effusion, cavitation
- Bacteriologic: MRSA, gram-negative or Legionella pneumonia
- Severity index: PSI > 90
- Pharmacologic: incorrect antibiotic choice based on susceptibility
Patients with acute deterioration of clinical status require prompt transfer to a higher level of care and may require mechanical ventilator support. In those with delay in achieving clinical stability, a question centers on whether the same antibiotics can be continued while doing further radiographic/microbiologic work-up and/or changing antibiotics. History should be reviewed, with particular attention to exposures, travel history, and microbiologic and radiographic data. Clinicians should recall that viruses account for up to 20% of pneumonias and that there are also noninfectious causes that can mimic pyogenic infections.32 If adequate initial cultures were not obtained, they should be obtained; however, care must be taken in reviewing new sets of cultures while on antibiotics, as they may reveal colonization selected out by antibiotics and not a true pathogen. If repeat evaluation is unrevealing, then further evaluation with computed tomography (CT) scan and bronchoscopy with bronchoalveolar lavage and biopsy is warranted. CT scans can show pleural effusions, bronchial obstructions, or a pattern suggestive of cryptogenic pneumonia. A bronchoscopy might yield a microbiologic diagnosis and, when combined with biopsy, can also evaluate for noninfectious causes.
As with other infections, if escalation of antibiotics is undertaken, clinicians should try to determine the reason for nonresponse. To simply broaden antimicrobial therapy without attempts at establishing a microbiologic or radiographic cause for nonresponse may lead to inappropriate treatment and recurrence of infection. Aside from patients who have bacteremic pneumococcal pneumonia in an ICU setting, there are no published reports pointing to superiority of combination antibiotics.20
Other Treatment
Several agents have been evaluated as adjunctive treatment of pneumonia to decrease the inflammatory response associated with pneumonia; namely, steroids, macrolide antibiotics, and statins. To date, only the use of steroids (methylprednisolone 0.5 mg/kg every 12 hours for 5 days) in those with severe CAP and high initial anti-inflammatory response (CRP > 150) has been shown to decrease treatment failure, decrease risk of acute respiratory distress syndrome, and possibly reduce length of stay and duration of intravenous antibiotics, without effect on mortality or adverse side effects.33,34 However, a recent double-blind randomized study conducted in Australia in which patients admitted with CAP were prescribed prednisolone acetate (50 mg/day for 7 days) and de-escalated from parenteral to oral antibiotics according to standardized criteria revealed no difference in mortality, length of stay, or readmission rates between the corticosteroids group and the control group at 90-day follow-up.35 At this point, corticosteroid as an adjunctive treatment for CAP is still controversial and the new 2019 ATS/IDSA guidelines recommend not routinely using corticosteroids in all patients with CAP.36 Other adjunctive methods have not been found to have significant impact.6
Prevention of Pneumonia
Prevention of pneumococcal pneumonia involves vaccinations to prevent infection caused by S. pneumoniae and influenza viruses. As influenza is a risk factor for bacterial infection, specifically with S. pneumoniae, influenza vaccination can help prevent bacterial pneumonia.37 In their most recent recommendations, the CDC continues to recommend routine influenza vaccination for all persons older than age 6 months, unless otherwise contraindicated.38
There are 2 vaccines for prevention of pneumococcal disease: the pneumococcal polysaccharide vaccine (PPSV23) and a conjugate vaccine (PCV13). Following vaccination with PPSV23, 80% of adults develop antibodies against at least 18 of the 23 serotypes.39 PPSV23 is reported to be protective against invasive pneumococcal infection, although there is no consensus regarding whether PPSV23 leads to decreased rates of pneumonia.40 On the other hand, PCV13 vaccination was associated with prevention of both invasive disease and CAP in adults aged 65 years or older.41 The CDC recommends that all children aged 2 years or younger receive PCV13, and those aged 65 or older receive PCV13 followed by a dose of PPSV23.42,43 The dose of PPSV23 should be given at least 1 year after the dose of PCV13 is administered.44 Persons younger than 65 years with immunocompromising and certain other conditions should also receive vaccination (Table 5).44 Full recommendations, many scenarios, and details on timing of vaccinations can be found at the CDC’s website.
Cigarette smoking increases the risk of respiratory infections, as evidenced by smokers accounting for almost half of all patients with invasive pneumococcal disease.11 As this is a modifiable risk factor, smoking cessation should be part of a comprehensive approach toward prevention of pneumonia.
Summary
Most patients with CAP are treated empirically with antibiotics, with therapy selection based on the site of care, likely pathogen, and antimicrobial resistance issues. Those treated as outpatients usually respond well to empiric antibiotic treatment, and a causative pathogen is not usually sought. Patients who are hospitalized for treatment usually receive empiric antibiotic on admission, and antimicrobial therapy is adjusted accordingly once the etiology has been determined by microbiologic or serologic means. At this time, the use of corticosteroid as an adjunctive treatment for CAP is still controversial, so not all patients with CAP should routinely receive corticosteroids. Because vaccination (PPSV23, PCV13, and influenza vaccine) remains the most effective tool in preventing the development of CAP, clinicians should strive for 100% vaccination rates in persons without contraindications.
1. Fine MJ, Auble TE, Yealy DM, et al A prediction rule to identify low-risk patients with community-acquired pneumonia. N Engl J Med.1997;336:243-250.
2. Lim WS, van der Eerden MM, Laing R, et al. Defining community acquired pneumonia severity on presentation to hospital: an international derivation and validation study. Thorax. 2003;58:377-382.
3. Aujesky D, Auble TE, Yealy DM, et al. Prospective comparison of three validated prediction rules for prognosis in community-acquired pneumonia. Am J Med. 2005;118:384-392.
4. Arnold FW, Ramirez JA, McDonald LC, Xia EL. Hospitalization for community-acquired pneumonia: the pneumonia severity index vs clinical judgment. Chest. 2003;124:121-124.
5. Aujesky D, McCausland JB, Whittle J, et al. Reasons why emergency department providers do not rely on the pneumonia severity index to determine the initial site of treatment for patients with pneumonia. Clin Infect Dis. 2009;49:e100-108.
6. Mandell LA, Wunderink RG, Anzueto A, et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44 Suppl 2:S27-72.
7. Charles PG, Wolfe R, Whitby M, et al. SMART-COP: a tool for predicting the need for intensive respiratory or vasopressor support in community-acquired pneumonia. Clin Infect Dis. 2008;47:375-384.
8. Marti C, Garin N, Grosgurin O, et al. Prediction of severe community-acquired pneumonia: a systematic review and meta-analysis. Crit Care. 2012;16:R141.
9. Jain S, Self WH, Wunderink RG, et al. Community-acquired pneumonia requiring hospitalization among U.S. adults. N Engl J Med. 2015;373:415-427.
10. Kalil AC, Metersky ML, Klompas M, et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 Clinical Practice Guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis. 2016;63:e61-e111.
11. Wunderink RG, Niederman MS, Kollef MH, et al. Linezolid in methicillin-resistant Staphylococcus aureus nosocomial pneumonia: a randomized, controlled study. Clin Infect Dis. 2012;54:621-629.
12. Silverman JA, Mortin LI, Vanpraagh AD, et al. Inhibition of daptomycin by pulmonary surfactant: in vitro modeling and clinical impact. J Infect Dis. 2005;191:2149-2152.
13. El Hajj MS, Turgeon RD, Wilby KJ. Ceftaroline fosamil for community-acquired pneumonia and skin and skin structure infections: a systematic review. Int J Clin Pharm. 2017;39:26-32.
14. Taboada M, Melnick D, Iaconis JP, et al. Ceftaroline fosamil versus ceftriaxone for the treatment of community-acquired pneumonia: individual patient data meta-analysis of randomized controlled trials. J Antimicrob Chemother. 2016;71:862-870.
15. Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children: executive summary. Clin Infect Dis. 2011;52:285-292.
16. Janoff EM. Streptococcus pneumonia. In: Bennett JE, Dolin R, Blaser MJ, editors. Mandell, Douglas and Bennett’s Principles and Practice of Infectious Diseases. 8th ed. Philadelphia: Sauders; 2015:2310-2327.
17. Teflaro (ceftaroline fosamil) [package insert]. St. Louis, MO: Forest Pharmaceuticals; 2010.
18. Nuzyra (omadacycline) [package insert]. Boston, MA: Paratek Pharmaceuticals; 2018.
19. Xenleta (lefamulin) [package insert]. Dublin, Ireland: Nabriva Therapeutics; 2019.
20. Baddour LM, Yu VL, Klugman KP, et al. Combination antibiotic therapy lowers mortality among severely ill patients with pneumococcal bacteremia. Am J Respir Crit Care Med. 2004;170:440-444.
21. FDA Drug Safety Communication: FDA warns of increased risk of death with IV antibacterial Tygacil (tigecycline) and approves new boxed warning. www.fda.gov/Drugs/DrugSafety/ucm369580.htm. Accessed 16 September 2019.
22. Edelstein PR, CR. Legionnaires’ disease and Pontiac fever. In: Kasper DF, editor. Harrison’s Infectious Diseases. 1st ed. New York: McGraw-Hill; 2010:2633.
23. Hammerschlag MR, Kohlhoff SA, Gaydos, CA. Chlamydia pneumoniae. In: Kasper DF, editor. Harrison’s Infectious Diseases. 1st ed. New York: McGraw-Hill; 2010:2174.
24. Holzman RS, MS. Mycoplasma pneumoniae and atypical pneumonia. In: Kasper DF, editor. Harrison’s Infectious Diseases. 1st ed. New York: McGraw-Hill; 2010:2183.
25. Yamada M, Buller R, Bledsoe S, Storch GA. Rising rates of macrolide-resistant Mycoplasma pneumoniae in the central United States. Pediatr Infect Dis J. 2012;31:409-410.
26. Yi SH, Hatfield KM, Baggs J, et al. Duration of antibiotic use among adults with uncomplicated community-acquired pneumonia requiring hospitalization in the United States. Clin Infect Dis. 2018;66:1333-1341.
27. Hayashi Y, Paterson DL. Strategies for reduction in duration of antibiotic use in hospitalized patients. Clin Infect Dis. 2011;52:1232-1240.
28. Akram AR, Chalmers JD, Taylor JK, et al. An evaluation of clinical stability criteria to predict hospital course in community-acquired pneumonia. Clin Microbiol Infect. 2013;19:1174-1180.
29. Halm EA, Fine MJ, Kapoor WN, et al. Instability on hospital discharge and the risk of adverse outcomes in patients with pneumonia. Arch Intern Med. 2002;162:1278-1284.
30. Janoff EM. Streptococcus pneumonia. In: Bennett JE, Dolin R, Blaser MJ, editors. Mandell, Douglas and Bennett’s Principles and Practice of Infectious Diseases. 8th ed. Philadelphia: Saunders; 2015:2310-2327.
31. Roson B, Carratala J, Fernandez-Sabe N, et al. Causes and factors associated with early failure in hospitalized patients with community-acquired pneumonia. Arch Intern Med. 2004;164:502-508.
32. El-Solh AA, Pietrantoni C, Bhat A, et al. Microbiology of severe aspiration pneumonia in institutionalized elderly. Am J Respir Crit Care Med. 2003;167:1650-1654.
33. Wan YD, Sun TW, Liu ZQ, et al. Efficacy and safety of corticosteroids for community-acquired pneumonia: a systematic review and meta-analysis. Chest. 2016;149:209-219.
34. Torres A, Sibila O, Ferrer M, et al. Effect of corticosteroids on treatment failure among hospitalized patients with severe community-acquired pneumonia and high inflammatory response: a randomized clinical trial. JAMA. 2015;313:677-686.
35. Lloyd M, Karahalios, Janus E, et al. Effectiveness of a bundled intervention including adjunctive corticosteroids on outcomes of hospitalized patients with community-acquired pneumonia: a stepped-wedge randomized clinical trial. JAMA Intern Med. 2019;179:1052-1060.
36. Metlay JP, Waterer GW, Long AC, et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official Clinical Practice Guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019;200:e45-e67.
37. McCullers JA. Insights into the interaction between influenza virus and pneumococcus. Clin Microbiol Rev. 2006;19:571-582.
38. Grohskopf LA, Alyanak E, Broder KR, et al. Prevention and control of seasonal influenza with vaccines: recommendations of the advisory committee on immunization practices - United States, 2019-20 influenza season. MMWR Recomm Rep. 2019;68:1-21.
39. Rubins JB, Alter M, Loch J, Janoff EN. Determination of antibody responses of elderly adults to all 23 capsular polysaccharides after pneumococcal vaccination. Infect Immun. 1999;67:5979-5984.
40. Vaccines and preventable diseases. Centers for Disease Control and Prevention Web site. www.cdc.gov/vaccines/vpd/pneumo/hcp/about-vaccine.html. Accessed 16 September 2019.
41. Bonten MJ, Huijts SM, Bolkenbaas M, et al. Polysaccharide conjugate vaccine against pneumococcal pneumonia in adults. N Engl J Med. 2015;372:1114-1125.
42. Recommended adult immunization schedule -- United States -- 2019. Centers for Disease Control and Prevention Web site. www.cdc.gov/vaccines/schedules/downloads/adult/adult-combined-schedule.pdf. Accessed 16 September 2019.
43. Recommended child and adolescent immunization schedule for ages 18 years or younger – United States – 2019. Centers for Disease Control and Prevention Web site. www.cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html. Accessed 22 September 2019.
44. Pneumococcal vaccine timing for adults – United States – 2019. Centers for Disease Control and Prevention Web site. www.cdc.gov/vaccines/vpd/pneumo/downloads/pneumo-vaccine-timing.pdf. Accessed 22 September 2019.
Initial management decisions for patients with community-acquired pneumonia (CAP) will depend on severity of infection, with need for hospitalization being one of the first decisions. Because empiric antibiotics are the mainstay of treatment and the causative organisms are seldom identified, underlying medical conditions and epidemiologic risk factors are considered when selecting an empiric regimen. As with other infections, duration of therapy is not standardized, but rather is guided by clinical improvement. Prevention of pneumonia centers around vaccination and smoking cessation. This article, the second in a 2-part review of CAP in adults, focuses on site of care decision, empiric and directed therapies, length of treatment, and prevention strategies. Evaluation and diagnosis of CAP are discussed in a separate article.
Site of Care Decision
For patients diagnosed with CAP, the clinician must decide whether treatment will be done in an outpatient or inpatient setting, and for those in the inpatient setting, whether they can safely be treated on the general medical ward or in the intensive care unit (ICU). Two common scoring systems that can be used to aid the clinician in determining severity of the infection and guide site-of-care decisions are the Pneumonia Severity Index (PSI) and CURB-65 scores.
The PSI score uses 20 different parameters, including comorbidities, laboratory parameters, and radiographic findings, to stratify patients into 5 mortality risk classes.1 On the basis of associated mortality rates, it has been suggested that risk class I and II patients should be treated as outpatients, risk class III patients should be treated in an observation unit or with a short hospitalization, and risk class IV and V patients should be treated as inpatients.1
The CURB-65 method of risk stratification is based on 5 clinical parameters: confusion, urea level, respiratory rate, systolic blood pressure, and age ≥ 65 years (Table 1).2,3 A modification to the CURB-65 algorithm tool was CRB-65, which excludes urea nitrogen, making it optimal for making determinations in a clinic-based setting. It should be emphasized that these tools do not take into account other factors that should be used in determining location of treatment, such as stable home, mental illness, or concerns about compliance with medications. In many instances, it is these factors that preclude low-risk patients from being treated as outpatients.4,5 Similarly, these scoring systems have not been validated for immunocompromised patients or those who would qualify as having health care–associated pneumonia.
Patients with CURB-65 scores of 4 or 5 are considered to have severe pneumonia, and admission to the ICU should be considered for these patients. Aside from the CURB-65 score, anyone requiring vasopressor support or mechanical ventilation merits admission to the ICU.6 American Thoracic Society (ATS) and Infectious Diseases Society of America (IDSA) guidelines also recommend the use of “minor criteria” for making ICU admission decisions; these include respiratory rate ≥ 30 breaths/minute, PaO2 fraction ≤ 250 mm Hg, multilobar infiltrates, confusion, blood urea nitrogen ≥ 20 mg/dL, leukopenia, thrombocytopenia, hypothermia, and hypotension.6 These factors are associated with increased mortality due to CAP, and ICU admission is indicated if 3 of the minor criteria for severe CAP are present.
Another clinical calculator that can be used for assessing severity of CAP is SMART-COP (systolic blood pressure, multilobar chest radiography involvement, albumin level, respiratory rate, tachycardia, confusion, oxygenation and arterial pH).7 This scoring system uses 8 weighted criteria to predict which patients will require intensive respiratory or vasopressor support. SMART-COP has a sensitivity of 79% and a specificity of 64% in predicting ICU admission, whereas CURB-65 has a pooled sensitivity of 57.2% and specificity of 77.2%.8
Antibiotic Therapy
Antibiotics are the mainstay of treatment for CAP, with the majority of patients with CAP treated empirically taking into account the site of care, likely pathogen, and antimicrobial resistance issues. Patients with pneumonia who are treated as outpatients usually respond well to empiric antibiotic treatment, and a causative pathogen is not usually sought. Patients who are hospitalized for treatment of CAP usually receive empiric antibiotic on admission. Once the etiology has been determined by microbiologic or serologic means, antimicrobial therapy should be adjusted accordingly. A CDC study found that the burden of viral etiologies was higher than previously thought, with rhinovirus and influenza accounting for 15% of cases and Streptococcus pneumoniae for only 5%.9 This study highlighted the fact that despite advances in molecular techniques, no pathogen is identified for most patients with pneumonia.9 Given the lack of discernable pathogens in the majority of cases, patients should continue to be treated with antibiotics unless a nonbacterial etiology is found.
Outpatients without comorbidities or risk factors for drug-resistant S. pneumoniae (Table 2)10 can be treated with monotherapy. Hospitalized patients are usually treated with combination intravenous therapy, although non-ICU patients who receive a respiratory fluoroquinolone can be treated orally.
As previously mentioned, antibiotic therapy is typically empiric, since neither clinical features nor radiographic features are sufficient to include or exclude infectious etiologies. Epidemiologic risk factors should be considered and, in certain cases, antimicrobial coverage should be expanded to include those entities; for example, treatment of anaerobes in the setting of lung abscess and antipseudomonal antibiotics for patients with bronchiectasis.
Of concern in the treatment of CAP is the increased prevalence of antimicrobial resistance among S. pneumoniae. The IDSA guidelines report that drug-resistant S. pneumoniae is more common in persons aged < 2 or > 65 years, and those with β-lactam therapy within the previous 3 months, alcoholism, medical comorbidities, immunosuppressive illness or therapy, or exposure to a child who attends a day care center.6
Staphylococcus aureus should be considered during influenza outbreaks, with either vancomycin or linezolid being the recommended agents in the setting of methicillin-resistant S. aureus (MRSA). In a study comparing vancomycin versus linezolid for nosocomial pneumonia, the all-cause 60-day mortality was similar for both agents.11 Daptomycin, another agent used against MRSA, is not indicated in the setting of pneumonia because daptomycin binds to surfactant, yielding it ineffective in the treatment of pneumonia.12 Ceftaroline is a newer cephalosporin with activity against MRSA; its role in treatment of community-acquired MRSA pneumonia has not been fully elucidated, but it appears to be a useful agent for this indication.13,14 Similarly, other agents known to have antibacterial properties against MRSA, such as trimethoprim/sulfamethoxazole and doxycycline, have not been studied for this indication. Clindamycin has been used to treat MRSA in children, and IDSA guidelines on the treatment of MRSA list clindamycin as an alternative15 if MRSA is known to be sensitive.
A summary of recommended empiric antibiotic therapy is presented in Table 3.16
Three antibiotics were approved by the US Food and Drug Administration (FDA) for the treatment of CAP after the release of the IDSA/ATS guidelines in 2007. Ceftaroline fosamil is a fifth-generation cephalosporin that has coverage for MRSA and was approved in November 2010.17 It can only be administered intravenously and needs dose adjustment for renal function. Omadacycline is a new tetracycline that was approved by the FDA in October 2018.18 It is available in both intravenous injectable and oral forms. No dose adjustment is needed for renal function. Lefamulin is a first-in-class novel pleuromutilin antibiotic which was FDA-approved in August 2019. It can be administered intravenously or orally, with no dosage adjustment necessary in patients with renal impairment.19
Antibiotic Therapy for Selected Pathogens
Streptococcus pneumoniae
Patients with pneumococcal pneumonia who have penicillin-susceptible strains can be treated with intravenous penicillin (2 or 3 million units every 4 hours) or ceftriaxone. Once a patient meets criteria of stability, they can then be transitioned to oral penicillin, amoxicillin, or clarithromycin. Those with strains with reduced susceptibility can still be treated with penicillin, but at a higher dose (4 million units intravenously [IV] every 4 hours), or a third-generation cephalosporin. Those whose pneumococcal pneumonia is complicated by bacteremia will benefit from dual therapy if severely ill, requiring ICU monitoring. Those not severely ill can be treated with monotherapy.20
Staphylococcus aureus
Staphylococcus aureus is more commonly associated with hospital-acquired pneumonia, but it may also be seen during the influenza season and in those with severe necrotizing CAP. Both linezolid and vancomycin can be used to treat MRSA CAP. As noted, ceftaroline has activity against MRSA and is approved for treatment of CAP, but is not approved by the FDA for MRSA CAP treatment. Similarly, tigecycline is approved for CAP and has activity against MRSA, but is not approved for MRSA CAP. Moreover, the FDA has warned of increased risk of death with tigecycline and has a black box warning to that effect.21
Legionella
Legionellosis can be treated with tetra¬cyclines, macrolides, or fluoroquinolones. For non-immunocompromised patients with mild pneumonia, any of the listed antibiotics is considered appropriate. However, patients with severe infection or those with immunosuppression should be treated with either levofloxacin or azithromycin for 7 to 10 days.22
Chlamydophila pneumoniae
As with other atypical organisms, Chlamydophila pneumoniae can be treated with doxycycline, a macrolide, or respiratory fluoroquinolones. However, length of therapy varies by regimen used; treating with doxycycline 100 mg twice daily generally requires 14 to 21 days, whereas moxifloxacin 400 mg daily requires 10 days.23
Mycoplasma pneumoniae
As with C. pneumoniae, length of therapy of Mycoplasma pneumoniae varies by which antimicrobial regimen is used. Shortest courses are seen with the use of macrolides for 5 days, whereas 14 days is considered standard for doxycycline or a respiratory fluoroquinolone.24 It should be noted that there has been increasingly documented resistance to macrolides, with known resistance of 8.2% in the United States.25
Duration of Treatment
Most patients with CAP respond to appropriate therapy within 72 hours. IDSA/ATS guidelines recommend that patients with routine cases of CAP be treated for a minimum of 5 days. Despite this, many patients are treated for an excessive amount of time, with over 70% of patients reported to have received antibiotics for more than 10 days for uncomplicated CAP.26 There are instances that require longer courses of antibiotics, including cases caused by Pseudomonas aeruginosa, S. aureus, and Legionella species and patients with lung abscesses or necrotizing infections, among others.27
Hospitalized patients do not need to be monitored for an additional day once they have reached clinical stability (Table 4), are able to maintain oral intake, and have normal mentation, provided that other comorbidities are stable and social needs have been met.6 C-reactive protein (CRP) level has been postulated as an additional measure of stability, specifically monitoring for a greater than 50% reduction in CRP; however, this was validated only for those with complicated pneumonia.28 Patients discharged from the hospital with instability have higher risk of readmission or death.29
Transition to Oral Therapy
IDSA/ATS guidelines6 recommend that patients should be transitioned from intravenous to oral antibiotics when they are improving clinically, have stable vital signs, and are able to ingest food/fluids and medications.
Management of Nonresponders
Although the majority of patients respond to antibiotics within 72 hours, treatment failure occurs in up to 15% of patients.15 Nonresponding pneumonia is generally seen in 2 patterns: worsening of clinical status despite empiric antibiotics or delay in achieving clinical stability, as defined in Table 4, after 72 hours of treatment.30 Risk factors associated with nonresponding pneumonia31 are:
- Radiographic: multilobar infiltrates, pleural effusion, cavitation
- Bacteriologic: MRSA, gram-negative or Legionella pneumonia
- Severity index: PSI > 90
- Pharmacologic: incorrect antibiotic choice based on susceptibility
Patients with acute deterioration of clinical status require prompt transfer to a higher level of care and may require mechanical ventilator support. In those with delay in achieving clinical stability, a question centers on whether the same antibiotics can be continued while doing further radiographic/microbiologic work-up and/or changing antibiotics. History should be reviewed, with particular attention to exposures, travel history, and microbiologic and radiographic data. Clinicians should recall that viruses account for up to 20% of pneumonias and that there are also noninfectious causes that can mimic pyogenic infections.32 If adequate initial cultures were not obtained, they should be obtained; however, care must be taken in reviewing new sets of cultures while on antibiotics, as they may reveal colonization selected out by antibiotics and not a true pathogen. If repeat evaluation is unrevealing, then further evaluation with computed tomography (CT) scan and bronchoscopy with bronchoalveolar lavage and biopsy is warranted. CT scans can show pleural effusions, bronchial obstructions, or a pattern suggestive of cryptogenic pneumonia. A bronchoscopy might yield a microbiologic diagnosis and, when combined with biopsy, can also evaluate for noninfectious causes.
As with other infections, if escalation of antibiotics is undertaken, clinicians should try to determine the reason for nonresponse. To simply broaden antimicrobial therapy without attempts at establishing a microbiologic or radiographic cause for nonresponse may lead to inappropriate treatment and recurrence of infection. Aside from patients who have bacteremic pneumococcal pneumonia in an ICU setting, there are no published reports pointing to superiority of combination antibiotics.20
Other Treatment
Several agents have been evaluated as adjunctive treatment of pneumonia to decrease the inflammatory response associated with pneumonia; namely, steroids, macrolide antibiotics, and statins. To date, only the use of steroids (methylprednisolone 0.5 mg/kg every 12 hours for 5 days) in those with severe CAP and high initial anti-inflammatory response (CRP > 150) has been shown to decrease treatment failure, decrease risk of acute respiratory distress syndrome, and possibly reduce length of stay and duration of intravenous antibiotics, without effect on mortality or adverse side effects.33,34 However, a recent double-blind randomized study conducted in Australia in which patients admitted with CAP were prescribed prednisolone acetate (50 mg/day for 7 days) and de-escalated from parenteral to oral antibiotics according to standardized criteria revealed no difference in mortality, length of stay, or readmission rates between the corticosteroids group and the control group at 90-day follow-up.35 At this point, corticosteroid as an adjunctive treatment for CAP is still controversial and the new 2019 ATS/IDSA guidelines recommend not routinely using corticosteroids in all patients with CAP.36 Other adjunctive methods have not been found to have significant impact.6
Prevention of Pneumonia
Prevention of pneumococcal pneumonia involves vaccinations to prevent infection caused by S. pneumoniae and influenza viruses. As influenza is a risk factor for bacterial infection, specifically with S. pneumoniae, influenza vaccination can help prevent bacterial pneumonia.37 In their most recent recommendations, the CDC continues to recommend routine influenza vaccination for all persons older than age 6 months, unless otherwise contraindicated.38
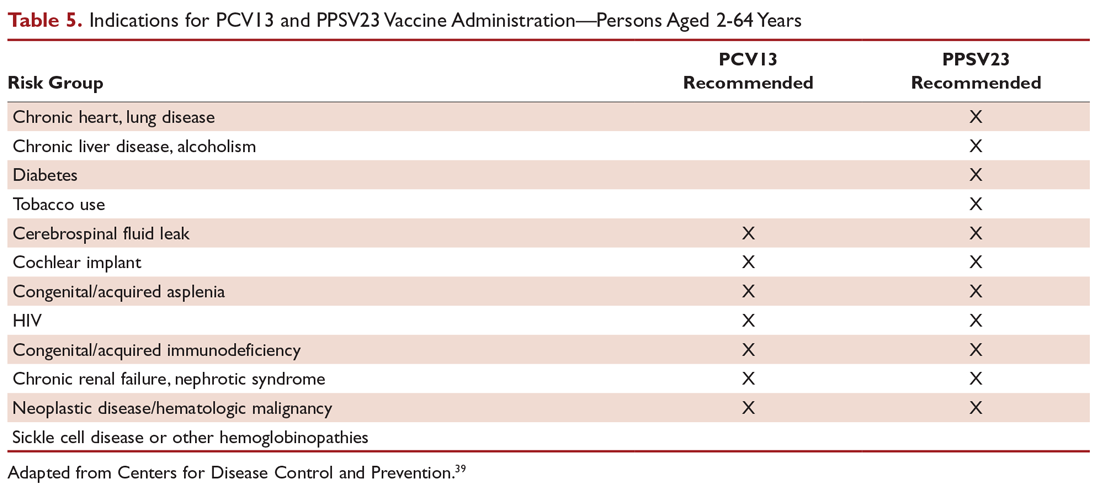
There are 2 vaccines for prevention of pneumococcal disease: the pneumococcal polysaccharide vaccine (PPSV23) and a conjugate vaccine (PCV13). Following vaccination with PPSV23, 80% of adults develop antibodies against at least 18 of the 23 serotypes.39 PPSV23 is reported to be protective against invasive pneumococcal infection, although there is no consensus regarding whether PPSV23 leads to decreased rates of pneumonia.40 On the other hand, PCV13 vaccination was associated with prevention of both invasive disease and CAP in adults aged 65 years or older.41 The CDC recommends that all children aged 2 years or younger receive PCV13, and those aged 65 or older receive PCV13 followed by a dose of PPSV23.42,43 The dose of PPSV23 should be given at least 1 year after the dose of PCV13 is administered.44 Persons younger than 65 years with immunocompromising and certain other conditions should also receive vaccination (Table 5).44 Full recommendations, many scenarios, and details on timing of vaccinations can be found at the CDC’s website.
Cigarette smoking increases the risk of respiratory infections, as evidenced by smokers accounting for almost half of all patients with invasive pneumococcal disease.11 As this is a modifiable risk factor, smoking cessation should be part of a comprehensive approach toward prevention of pneumonia.
Summary
Most patients with CAP are treated empirically with antibiotics, with therapy selection based on the site of care, likely pathogen, and antimicrobial resistance issues. Those treated as outpatients usually respond well to empiric antibiotic treatment, and a causative pathogen is not usually sought. Patients who are hospitalized for treatment usually receive empiric antibiotic on admission, and antimicrobial therapy is adjusted accordingly once the etiology has been determined by microbiologic or serologic means. At this time, the use of corticosteroid as an adjunctive treatment for CAP is still controversial, so not all patients with CAP should routinely receive corticosteroids. Because vaccination (PPSV23, PCV13, and influenza vaccine) remains the most effective tool in preventing the development of CAP, clinicians should strive for 100% vaccination rates in persons without contraindications.
Initial management decisions for patients with community-acquired pneumonia (CAP) will depend on severity of infection, with need for hospitalization being one of the first decisions. Because empiric antibiotics are the mainstay of treatment and the causative organisms are seldom identified, underlying medical conditions and epidemiologic risk factors are considered when selecting an empiric regimen. As with other infections, duration of therapy is not standardized, but rather is guided by clinical improvement. Prevention of pneumonia centers around vaccination and smoking cessation. This article, the second in a 2-part review of CAP in adults, focuses on site of care decision, empiric and directed therapies, length of treatment, and prevention strategies. Evaluation and diagnosis of CAP are discussed in a separate article.
Site of Care Decision
For patients diagnosed with CAP, the clinician must decide whether treatment will be done in an outpatient or inpatient setting, and for those in the inpatient setting, whether they can safely be treated on the general medical ward or in the intensive care unit (ICU). Two common scoring systems that can be used to aid the clinician in determining severity of the infection and guide site-of-care decisions are the Pneumonia Severity Index (PSI) and CURB-65 scores.
The PSI score uses 20 different parameters, including comorbidities, laboratory parameters, and radiographic findings, to stratify patients into 5 mortality risk classes.1 On the basis of associated mortality rates, it has been suggested that risk class I and II patients should be treated as outpatients, risk class III patients should be treated in an observation unit or with a short hospitalization, and risk class IV and V patients should be treated as inpatients.1
The CURB-65 method of risk stratification is based on 5 clinical parameters: confusion, urea level, respiratory rate, systolic blood pressure, and age ≥ 65 years (Table 1).2,3 A modification to the CURB-65 algorithm tool was CRB-65, which excludes urea nitrogen, making it optimal for making determinations in a clinic-based setting. It should be emphasized that these tools do not take into account other factors that should be used in determining location of treatment, such as stable home, mental illness, or concerns about compliance with medications. In many instances, it is these factors that preclude low-risk patients from being treated as outpatients.4,5 Similarly, these scoring systems have not been validated for immunocompromised patients or those who would qualify as having health care–associated pneumonia.
Patients with CURB-65 scores of 4 or 5 are considered to have severe pneumonia, and admission to the ICU should be considered for these patients. Aside from the CURB-65 score, anyone requiring vasopressor support or mechanical ventilation merits admission to the ICU.6 American Thoracic Society (ATS) and Infectious Diseases Society of America (IDSA) guidelines also recommend the use of “minor criteria” for making ICU admission decisions; these include respiratory rate ≥ 30 breaths/minute, PaO2 fraction ≤ 250 mm Hg, multilobar infiltrates, confusion, blood urea nitrogen ≥ 20 mg/dL, leukopenia, thrombocytopenia, hypothermia, and hypotension.6 These factors are associated with increased mortality due to CAP, and ICU admission is indicated if 3 of the minor criteria for severe CAP are present.
Another clinical calculator that can be used for assessing severity of CAP is SMART-COP (systolic blood pressure, multilobar chest radiography involvement, albumin level, respiratory rate, tachycardia, confusion, oxygenation and arterial pH).7 This scoring system uses 8 weighted criteria to predict which patients will require intensive respiratory or vasopressor support. SMART-COP has a sensitivity of 79% and a specificity of 64% in predicting ICU admission, whereas CURB-65 has a pooled sensitivity of 57.2% and specificity of 77.2%.8
Antibiotic Therapy
Antibiotics are the mainstay of treatment for CAP, with the majority of patients with CAP treated empirically taking into account the site of care, likely pathogen, and antimicrobial resistance issues. Patients with pneumonia who are treated as outpatients usually respond well to empiric antibiotic treatment, and a causative pathogen is not usually sought. Patients who are hospitalized for treatment of CAP usually receive empiric antibiotic on admission. Once the etiology has been determined by microbiologic or serologic means, antimicrobial therapy should be adjusted accordingly. A CDC study found that the burden of viral etiologies was higher than previously thought, with rhinovirus and influenza accounting for 15% of cases and Streptococcus pneumoniae for only 5%.9 This study highlighted the fact that despite advances in molecular techniques, no pathogen is identified for most patients with pneumonia.9 Given the lack of discernable pathogens in the majority of cases, patients should continue to be treated with antibiotics unless a nonbacterial etiology is found.
Outpatients without comorbidities or risk factors for drug-resistant S. pneumoniae (Table 2)10 can be treated with monotherapy. Hospitalized patients are usually treated with combination intravenous therapy, although non-ICU patients who receive a respiratory fluoroquinolone can be treated orally.
As previously mentioned, antibiotic therapy is typically empiric, since neither clinical features nor radiographic features are sufficient to include or exclude infectious etiologies. Epidemiologic risk factors should be considered and, in certain cases, antimicrobial coverage should be expanded to include those entities; for example, treatment of anaerobes in the setting of lung abscess and antipseudomonal antibiotics for patients with bronchiectasis.
Of concern in the treatment of CAP is the increased prevalence of antimicrobial resistance among S. pneumoniae. The IDSA guidelines report that drug-resistant S. pneumoniae is more common in persons aged < 2 or > 65 years, and those with β-lactam therapy within the previous 3 months, alcoholism, medical comorbidities, immunosuppressive illness or therapy, or exposure to a child who attends a day care center.6
Staphylococcus aureus should be considered during influenza outbreaks, with either vancomycin or linezolid being the recommended agents in the setting of methicillin-resistant S. aureus (MRSA). In a study comparing vancomycin versus linezolid for nosocomial pneumonia, the all-cause 60-day mortality was similar for both agents.11 Daptomycin, another agent used against MRSA, is not indicated in the setting of pneumonia because daptomycin binds to surfactant, yielding it ineffective in the treatment of pneumonia.12 Ceftaroline is a newer cephalosporin with activity against MRSA; its role in treatment of community-acquired MRSA pneumonia has not been fully elucidated, but it appears to be a useful agent for this indication.13,14 Similarly, other agents known to have antibacterial properties against MRSA, such as trimethoprim/sulfamethoxazole and doxycycline, have not been studied for this indication. Clindamycin has been used to treat MRSA in children, and IDSA guidelines on the treatment of MRSA list clindamycin as an alternative15 if MRSA is known to be sensitive.
A summary of recommended empiric antibiotic therapy is presented in Table 3.16
Three antibiotics were approved by the US Food and Drug Administration (FDA) for the treatment of CAP after the release of the IDSA/ATS guidelines in 2007. Ceftaroline fosamil is a fifth-generation cephalosporin that has coverage for MRSA and was approved in November 2010.17 It can only be administered intravenously and needs dose adjustment for renal function. Omadacycline is a new tetracycline that was approved by the FDA in October 2018.18 It is available in both intravenous injectable and oral forms. No dose adjustment is needed for renal function. Lefamulin is a first-in-class novel pleuromutilin antibiotic which was FDA-approved in August 2019. It can be administered intravenously or orally, with no dosage adjustment necessary in patients with renal impairment.19
Antibiotic Therapy for Selected Pathogens
Streptococcus pneumoniae
Patients with pneumococcal pneumonia who have penicillin-susceptible strains can be treated with intravenous penicillin (2 or 3 million units every 4 hours) or ceftriaxone. Once a patient meets criteria of stability, they can then be transitioned to oral penicillin, amoxicillin, or clarithromycin. Those with strains with reduced susceptibility can still be treated with penicillin, but at a higher dose (4 million units intravenously [IV] every 4 hours), or a third-generation cephalosporin. Those whose pneumococcal pneumonia is complicated by bacteremia will benefit from dual therapy if severely ill, requiring ICU monitoring. Those not severely ill can be treated with monotherapy.20
Staphylococcus aureus
Staphylococcus aureus is more commonly associated with hospital-acquired pneumonia, but it may also be seen during the influenza season and in those with severe necrotizing CAP. Both linezolid and vancomycin can be used to treat MRSA CAP. As noted, ceftaroline has activity against MRSA and is approved for treatment of CAP, but is not approved by the FDA for MRSA CAP treatment. Similarly, tigecycline is approved for CAP and has activity against MRSA, but is not approved for MRSA CAP. Moreover, the FDA has warned of increased risk of death with tigecycline and has a black box warning to that effect.21
Legionella
Legionellosis can be treated with tetra¬cyclines, macrolides, or fluoroquinolones. For non-immunocompromised patients with mild pneumonia, any of the listed antibiotics is considered appropriate. However, patients with severe infection or those with immunosuppression should be treated with either levofloxacin or azithromycin for 7 to 10 days.22
Chlamydophila pneumoniae
As with other atypical organisms, Chlamydophila pneumoniae can be treated with doxycycline, a macrolide, or respiratory fluoroquinolones. However, length of therapy varies by regimen used; treating with doxycycline 100 mg twice daily generally requires 14 to 21 days, whereas moxifloxacin 400 mg daily requires 10 days.23
Mycoplasma pneumoniae
As with C. pneumoniae, length of therapy of Mycoplasma pneumoniae varies by which antimicrobial regimen is used. Shortest courses are seen with the use of macrolides for 5 days, whereas 14 days is considered standard for doxycycline or a respiratory fluoroquinolone.24 It should be noted that there has been increasingly documented resistance to macrolides, with known resistance of 8.2% in the United States.25
Duration of Treatment
Most patients with CAP respond to appropriate therapy within 72 hours. IDSA/ATS guidelines recommend that patients with routine cases of CAP be treated for a minimum of 5 days. Despite this, many patients are treated for an excessive amount of time, with over 70% of patients reported to have received antibiotics for more than 10 days for uncomplicated CAP.26 There are instances that require longer courses of antibiotics, including cases caused by Pseudomonas aeruginosa, S. aureus, and Legionella species and patients with lung abscesses or necrotizing infections, among others.27
Hospitalized patients do not need to be monitored for an additional day once they have reached clinical stability (Table 4), are able to maintain oral intake, and have normal mentation, provided that other comorbidities are stable and social needs have been met.6 C-reactive protein (CRP) level has been postulated as an additional measure of stability, specifically monitoring for a greater than 50% reduction in CRP; however, this was validated only for those with complicated pneumonia.28 Patients discharged from the hospital with instability have higher risk of readmission or death.29
Transition to Oral Therapy
IDSA/ATS guidelines6 recommend that patients should be transitioned from intravenous to oral antibiotics when they are improving clinically, have stable vital signs, and are able to ingest food/fluids and medications.
Management of Nonresponders
Although the majority of patients respond to antibiotics within 72 hours, treatment failure occurs in up to 15% of patients.15 Nonresponding pneumonia is generally seen in 2 patterns: worsening of clinical status despite empiric antibiotics or delay in achieving clinical stability, as defined in Table 4, after 72 hours of treatment.30 Risk factors associated with nonresponding pneumonia31 are:
- Radiographic: multilobar infiltrates, pleural effusion, cavitation
- Bacteriologic: MRSA, gram-negative or Legionella pneumonia
- Severity index: PSI > 90
- Pharmacologic: incorrect antibiotic choice based on susceptibility
Patients with acute deterioration of clinical status require prompt transfer to a higher level of care and may require mechanical ventilator support. In those with delay in achieving clinical stability, a question centers on whether the same antibiotics can be continued while doing further radiographic/microbiologic work-up and/or changing antibiotics. History should be reviewed, with particular attention to exposures, travel history, and microbiologic and radiographic data. Clinicians should recall that viruses account for up to 20% of pneumonias and that there are also noninfectious causes that can mimic pyogenic infections.32 If adequate initial cultures were not obtained, they should be obtained; however, care must be taken in reviewing new sets of cultures while on antibiotics, as they may reveal colonization selected out by antibiotics and not a true pathogen. If repeat evaluation is unrevealing, then further evaluation with computed tomography (CT) scan and bronchoscopy with bronchoalveolar lavage and biopsy is warranted. CT scans can show pleural effusions, bronchial obstructions, or a pattern suggestive of cryptogenic pneumonia. A bronchoscopy might yield a microbiologic diagnosis and, when combined with biopsy, can also evaluate for noninfectious causes.
As with other infections, if escalation of antibiotics is undertaken, clinicians should try to determine the reason for nonresponse. To simply broaden antimicrobial therapy without attempts at establishing a microbiologic or radiographic cause for nonresponse may lead to inappropriate treatment and recurrence of infection. Aside from patients who have bacteremic pneumococcal pneumonia in an ICU setting, there are no published reports pointing to superiority of combination antibiotics.20
Other Treatment
Several agents have been evaluated as adjunctive treatment of pneumonia to decrease the inflammatory response associated with pneumonia; namely, steroids, macrolide antibiotics, and statins. To date, only the use of steroids (methylprednisolone 0.5 mg/kg every 12 hours for 5 days) in those with severe CAP and high initial anti-inflammatory response (CRP > 150) has been shown to decrease treatment failure, decrease risk of acute respiratory distress syndrome, and possibly reduce length of stay and duration of intravenous antibiotics, without effect on mortality or adverse side effects.33,34 However, a recent double-blind randomized study conducted in Australia in which patients admitted with CAP were prescribed prednisolone acetate (50 mg/day for 7 days) and de-escalated from parenteral to oral antibiotics according to standardized criteria revealed no difference in mortality, length of stay, or readmission rates between the corticosteroids group and the control group at 90-day follow-up.35 At this point, corticosteroid as an adjunctive treatment for CAP is still controversial and the new 2019 ATS/IDSA guidelines recommend not routinely using corticosteroids in all patients with CAP.36 Other adjunctive methods have not been found to have significant impact.6
Prevention of Pneumonia
Prevention of pneumococcal pneumonia involves vaccinations to prevent infection caused by S. pneumoniae and influenza viruses. As influenza is a risk factor for bacterial infection, specifically with S. pneumoniae, influenza vaccination can help prevent bacterial pneumonia.37 In their most recent recommendations, the CDC continues to recommend routine influenza vaccination for all persons older than age 6 months, unless otherwise contraindicated.38
There are 2 vaccines for prevention of pneumococcal disease: the pneumococcal polysaccharide vaccine (PPSV23) and a conjugate vaccine (PCV13). Following vaccination with PPSV23, 80% of adults develop antibodies against at least 18 of the 23 serotypes.39 PPSV23 is reported to be protective against invasive pneumococcal infection, although there is no consensus regarding whether PPSV23 leads to decreased rates of pneumonia.40 On the other hand, PCV13 vaccination was associated with prevention of both invasive disease and CAP in adults aged 65 years or older.41 The CDC recommends that all children aged 2 years or younger receive PCV13, and those aged 65 or older receive PCV13 followed by a dose of PPSV23.42,43 The dose of PPSV23 should be given at least 1 year after the dose of PCV13 is administered.44 Persons younger than 65 years with immunocompromising and certain other conditions should also receive vaccination (Table 5).44 Full recommendations, many scenarios, and details on timing of vaccinations can be found at the CDC’s website.
Cigarette smoking increases the risk of respiratory infections, as evidenced by smokers accounting for almost half of all patients with invasive pneumococcal disease.11 As this is a modifiable risk factor, smoking cessation should be part of a comprehensive approach toward prevention of pneumonia.
Summary
Most patients with CAP are treated empirically with antibiotics, with therapy selection based on the site of care, likely pathogen, and antimicrobial resistance issues. Those treated as outpatients usually respond well to empiric antibiotic treatment, and a causative pathogen is not usually sought. Patients who are hospitalized for treatment usually receive empiric antibiotic on admission, and antimicrobial therapy is adjusted accordingly once the etiology has been determined by microbiologic or serologic means. At this time, the use of corticosteroid as an adjunctive treatment for CAP is still controversial, so not all patients with CAP should routinely receive corticosteroids. Because vaccination (PPSV23, PCV13, and influenza vaccine) remains the most effective tool in preventing the development of CAP, clinicians should strive for 100% vaccination rates in persons without contraindications.
1. Fine MJ, Auble TE, Yealy DM, et al A prediction rule to identify low-risk patients with community-acquired pneumonia. N Engl J Med.1997;336:243-250.
2. Lim WS, van der Eerden MM, Laing R, et al. Defining community acquired pneumonia severity on presentation to hospital: an international derivation and validation study. Thorax. 2003;58:377-382.
3. Aujesky D, Auble TE, Yealy DM, et al. Prospective comparison of three validated prediction rules for prognosis in community-acquired pneumonia. Am J Med. 2005;118:384-392.
4. Arnold FW, Ramirez JA, McDonald LC, Xia EL. Hospitalization for community-acquired pneumonia: the pneumonia severity index vs clinical judgment. Chest. 2003;124:121-124.
5. Aujesky D, McCausland JB, Whittle J, et al. Reasons why emergency department providers do not rely on the pneumonia severity index to determine the initial site of treatment for patients with pneumonia. Clin Infect Dis. 2009;49:e100-108.
6. Mandell LA, Wunderink RG, Anzueto A, et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44 Suppl 2:S27-72.
7. Charles PG, Wolfe R, Whitby M, et al. SMART-COP: a tool for predicting the need for intensive respiratory or vasopressor support in community-acquired pneumonia. Clin Infect Dis. 2008;47:375-384.
8. Marti C, Garin N, Grosgurin O, et al. Prediction of severe community-acquired pneumonia: a systematic review and meta-analysis. Crit Care. 2012;16:R141.
9. Jain S, Self WH, Wunderink RG, et al. Community-acquired pneumonia requiring hospitalization among U.S. adults. N Engl J Med. 2015;373:415-427.
10. Kalil AC, Metersky ML, Klompas M, et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 Clinical Practice Guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis. 2016;63:e61-e111.
11. Wunderink RG, Niederman MS, Kollef MH, et al. Linezolid in methicillin-resistant Staphylococcus aureus nosocomial pneumonia: a randomized, controlled study. Clin Infect Dis. 2012;54:621-629.
12. Silverman JA, Mortin LI, Vanpraagh AD, et al. Inhibition of daptomycin by pulmonary surfactant: in vitro modeling and clinical impact. J Infect Dis. 2005;191:2149-2152.
13. El Hajj MS, Turgeon RD, Wilby KJ. Ceftaroline fosamil for community-acquired pneumonia and skin and skin structure infections: a systematic review. Int J Clin Pharm. 2017;39:26-32.
14. Taboada M, Melnick D, Iaconis JP, et al. Ceftaroline fosamil versus ceftriaxone for the treatment of community-acquired pneumonia: individual patient data meta-analysis of randomized controlled trials. J Antimicrob Chemother. 2016;71:862-870.
15. Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children: executive summary. Clin Infect Dis. 2011;52:285-292.
16. Janoff EM. Streptococcus pneumonia. In: Bennett JE, Dolin R, Blaser MJ, editors. Mandell, Douglas and Bennett’s Principles and Practice of Infectious Diseases. 8th ed. Philadelphia: Sauders; 2015:2310-2327.
17. Teflaro (ceftaroline fosamil) [package insert]. St. Louis, MO: Forest Pharmaceuticals; 2010.
18. Nuzyra (omadacycline) [package insert]. Boston, MA: Paratek Pharmaceuticals; 2018.
19. Xenleta (lefamulin) [package insert]. Dublin, Ireland: Nabriva Therapeutics; 2019.
20. Baddour LM, Yu VL, Klugman KP, et al. Combination antibiotic therapy lowers mortality among severely ill patients with pneumococcal bacteremia. Am J Respir Crit Care Med. 2004;170:440-444.
21. FDA Drug Safety Communication: FDA warns of increased risk of death with IV antibacterial Tygacil (tigecycline) and approves new boxed warning. www.fda.gov/Drugs/DrugSafety/ucm369580.htm. Accessed 16 September 2019.
22. Edelstein PR, CR. Legionnaires’ disease and Pontiac fever. In: Kasper DF, editor. Harrison’s Infectious Diseases. 1st ed. New York: McGraw-Hill; 2010:2633.
23. Hammerschlag MR, Kohlhoff SA, Gaydos, CA. Chlamydia pneumoniae. In: Kasper DF, editor. Harrison’s Infectious Diseases. 1st ed. New York: McGraw-Hill; 2010:2174.
24. Holzman RS, MS. Mycoplasma pneumoniae and atypical pneumonia. In: Kasper DF, editor. Harrison’s Infectious Diseases. 1st ed. New York: McGraw-Hill; 2010:2183.
25. Yamada M, Buller R, Bledsoe S, Storch GA. Rising rates of macrolide-resistant Mycoplasma pneumoniae in the central United States. Pediatr Infect Dis J. 2012;31:409-410.
26. Yi SH, Hatfield KM, Baggs J, et al. Duration of antibiotic use among adults with uncomplicated community-acquired pneumonia requiring hospitalization in the United States. Clin Infect Dis. 2018;66:1333-1341.
27. Hayashi Y, Paterson DL. Strategies for reduction in duration of antibiotic use in hospitalized patients. Clin Infect Dis. 2011;52:1232-1240.
28. Akram AR, Chalmers JD, Taylor JK, et al. An evaluation of clinical stability criteria to predict hospital course in community-acquired pneumonia. Clin Microbiol Infect. 2013;19:1174-1180.
29. Halm EA, Fine MJ, Kapoor WN, et al. Instability on hospital discharge and the risk of adverse outcomes in patients with pneumonia. Arch Intern Med. 2002;162:1278-1284.
30. Janoff EM. Streptococcus pneumonia. In: Bennett JE, Dolin R, Blaser MJ, editors. Mandell, Douglas and Bennett’s Principles and Practice of Infectious Diseases. 8th ed. Philadelphia: Saunders; 2015:2310-2327.
31. Roson B, Carratala J, Fernandez-Sabe N, et al. Causes and factors associated with early failure in hospitalized patients with community-acquired pneumonia. Arch Intern Med. 2004;164:502-508.
32. El-Solh AA, Pietrantoni C, Bhat A, et al. Microbiology of severe aspiration pneumonia in institutionalized elderly. Am J Respir Crit Care Med. 2003;167:1650-1654.
33. Wan YD, Sun TW, Liu ZQ, et al. Efficacy and safety of corticosteroids for community-acquired pneumonia: a systematic review and meta-analysis. Chest. 2016;149:209-219.
34. Torres A, Sibila O, Ferrer M, et al. Effect of corticosteroids on treatment failure among hospitalized patients with severe community-acquired pneumonia and high inflammatory response: a randomized clinical trial. JAMA. 2015;313:677-686.
35. Lloyd M, Karahalios, Janus E, et al. Effectiveness of a bundled intervention including adjunctive corticosteroids on outcomes of hospitalized patients with community-acquired pneumonia: a stepped-wedge randomized clinical trial. JAMA Intern Med. 2019;179:1052-1060.
36. Metlay JP, Waterer GW, Long AC, et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official Clinical Practice Guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019;200:e45-e67.
37. McCullers JA. Insights into the interaction between influenza virus and pneumococcus. Clin Microbiol Rev. 2006;19:571-582.
38. Grohskopf LA, Alyanak E, Broder KR, et al. Prevention and control of seasonal influenza with vaccines: recommendations of the advisory committee on immunization practices - United States, 2019-20 influenza season. MMWR Recomm Rep. 2019;68:1-21.
39. Rubins JB, Alter M, Loch J, Janoff EN. Determination of antibody responses of elderly adults to all 23 capsular polysaccharides after pneumococcal vaccination. Infect Immun. 1999;67:5979-5984.
40. Vaccines and preventable diseases. Centers for Disease Control and Prevention Web site. www.cdc.gov/vaccines/vpd/pneumo/hcp/about-vaccine.html. Accessed 16 September 2019.
41. Bonten MJ, Huijts SM, Bolkenbaas M, et al. Polysaccharide conjugate vaccine against pneumococcal pneumonia in adults. N Engl J Med. 2015;372:1114-1125.
42. Recommended adult immunization schedule -- United States -- 2019. Centers for Disease Control and Prevention Web site. www.cdc.gov/vaccines/schedules/downloads/adult/adult-combined-schedule.pdf. Accessed 16 September 2019.
43. Recommended child and adolescent immunization schedule for ages 18 years or younger – United States – 2019. Centers for Disease Control and Prevention Web site. www.cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html. Accessed 22 September 2019.
44. Pneumococcal vaccine timing for adults – United States – 2019. Centers for Disease Control and Prevention Web site. www.cdc.gov/vaccines/vpd/pneumo/downloads/pneumo-vaccine-timing.pdf. Accessed 22 September 2019.
1. Fine MJ, Auble TE, Yealy DM, et al A prediction rule to identify low-risk patients with community-acquired pneumonia. N Engl J Med.1997;336:243-250.
2. Lim WS, van der Eerden MM, Laing R, et al. Defining community acquired pneumonia severity on presentation to hospital: an international derivation and validation study. Thorax. 2003;58:377-382.
3. Aujesky D, Auble TE, Yealy DM, et al. Prospective comparison of three validated prediction rules for prognosis in community-acquired pneumonia. Am J Med. 2005;118:384-392.
4. Arnold FW, Ramirez JA, McDonald LC, Xia EL. Hospitalization for community-acquired pneumonia: the pneumonia severity index vs clinical judgment. Chest. 2003;124:121-124.
5. Aujesky D, McCausland JB, Whittle J, et al. Reasons why emergency department providers do not rely on the pneumonia severity index to determine the initial site of treatment for patients with pneumonia. Clin Infect Dis. 2009;49:e100-108.
6. Mandell LA, Wunderink RG, Anzueto A, et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44 Suppl 2:S27-72.
7. Charles PG, Wolfe R, Whitby M, et al. SMART-COP: a tool for predicting the need for intensive respiratory or vasopressor support in community-acquired pneumonia. Clin Infect Dis. 2008;47:375-384.
8. Marti C, Garin N, Grosgurin O, et al. Prediction of severe community-acquired pneumonia: a systematic review and meta-analysis. Crit Care. 2012;16:R141.
9. Jain S, Self WH, Wunderink RG, et al. Community-acquired pneumonia requiring hospitalization among U.S. adults. N Engl J Med. 2015;373:415-427.
10. Kalil AC, Metersky ML, Klompas M, et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 Clinical Practice Guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis. 2016;63:e61-e111.
11. Wunderink RG, Niederman MS, Kollef MH, et al. Linezolid in methicillin-resistant Staphylococcus aureus nosocomial pneumonia: a randomized, controlled study. Clin Infect Dis. 2012;54:621-629.
12. Silverman JA, Mortin LI, Vanpraagh AD, et al. Inhibition of daptomycin by pulmonary surfactant: in vitro modeling and clinical impact. J Infect Dis. 2005;191:2149-2152.
13. El Hajj MS, Turgeon RD, Wilby KJ. Ceftaroline fosamil for community-acquired pneumonia and skin and skin structure infections: a systematic review. Int J Clin Pharm. 2017;39:26-32.
14. Taboada M, Melnick D, Iaconis JP, et al. Ceftaroline fosamil versus ceftriaxone for the treatment of community-acquired pneumonia: individual patient data meta-analysis of randomized controlled trials. J Antimicrob Chemother. 2016;71:862-870.
15. Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children: executive summary. Clin Infect Dis. 2011;52:285-292.
16. Janoff EM. Streptococcus pneumonia. In: Bennett JE, Dolin R, Blaser MJ, editors. Mandell, Douglas and Bennett’s Principles and Practice of Infectious Diseases. 8th ed. Philadelphia: Sauders; 2015:2310-2327.
17. Teflaro (ceftaroline fosamil) [package insert]. St. Louis, MO: Forest Pharmaceuticals; 2010.
18. Nuzyra (omadacycline) [package insert]. Boston, MA: Paratek Pharmaceuticals; 2018.
19. Xenleta (lefamulin) [package insert]. Dublin, Ireland: Nabriva Therapeutics; 2019.
20. Baddour LM, Yu VL, Klugman KP, et al. Combination antibiotic therapy lowers mortality among severely ill patients with pneumococcal bacteremia. Am J Respir Crit Care Med. 2004;170:440-444.
21. FDA Drug Safety Communication: FDA warns of increased risk of death with IV antibacterial Tygacil (tigecycline) and approves new boxed warning. www.fda.gov/Drugs/DrugSafety/ucm369580.htm. Accessed 16 September 2019.
22. Edelstein PR, CR. Legionnaires’ disease and Pontiac fever. In: Kasper DF, editor. Harrison’s Infectious Diseases. 1st ed. New York: McGraw-Hill; 2010:2633.
23. Hammerschlag MR, Kohlhoff SA, Gaydos, CA. Chlamydia pneumoniae. In: Kasper DF, editor. Harrison’s Infectious Diseases. 1st ed. New York: McGraw-Hill; 2010:2174.
24. Holzman RS, MS. Mycoplasma pneumoniae and atypical pneumonia. In: Kasper DF, editor. Harrison’s Infectious Diseases. 1st ed. New York: McGraw-Hill; 2010:2183.
25. Yamada M, Buller R, Bledsoe S, Storch GA. Rising rates of macrolide-resistant Mycoplasma pneumoniae in the central United States. Pediatr Infect Dis J. 2012;31:409-410.
26. Yi SH, Hatfield KM, Baggs J, et al. Duration of antibiotic use among adults with uncomplicated community-acquired pneumonia requiring hospitalization in the United States. Clin Infect Dis. 2018;66:1333-1341.
27. Hayashi Y, Paterson DL. Strategies for reduction in duration of antibiotic use in hospitalized patients. Clin Infect Dis. 2011;52:1232-1240.
28. Akram AR, Chalmers JD, Taylor JK, et al. An evaluation of clinical stability criteria to predict hospital course in community-acquired pneumonia. Clin Microbiol Infect. 2013;19:1174-1180.
29. Halm EA, Fine MJ, Kapoor WN, et al. Instability on hospital discharge and the risk of adverse outcomes in patients with pneumonia. Arch Intern Med. 2002;162:1278-1284.
30. Janoff EM. Streptococcus pneumonia. In: Bennett JE, Dolin R, Blaser MJ, editors. Mandell, Douglas and Bennett’s Principles and Practice of Infectious Diseases. 8th ed. Philadelphia: Saunders; 2015:2310-2327.
31. Roson B, Carratala J, Fernandez-Sabe N, et al. Causes and factors associated with early failure in hospitalized patients with community-acquired pneumonia. Arch Intern Med. 2004;164:502-508.
32. El-Solh AA, Pietrantoni C, Bhat A, et al. Microbiology of severe aspiration pneumonia in institutionalized elderly. Am J Respir Crit Care Med. 2003;167:1650-1654.
33. Wan YD, Sun TW, Liu ZQ, et al. Efficacy and safety of corticosteroids for community-acquired pneumonia: a systematic review and meta-analysis. Chest. 2016;149:209-219.
34. Torres A, Sibila O, Ferrer M, et al. Effect of corticosteroids on treatment failure among hospitalized patients with severe community-acquired pneumonia and high inflammatory response: a randomized clinical trial. JAMA. 2015;313:677-686.
35. Lloyd M, Karahalios, Janus E, et al. Effectiveness of a bundled intervention including adjunctive corticosteroids on outcomes of hospitalized patients with community-acquired pneumonia: a stepped-wedge randomized clinical trial. JAMA Intern Med. 2019;179:1052-1060.
36. Metlay JP, Waterer GW, Long AC, et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official Clinical Practice Guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019;200:e45-e67.
37. McCullers JA. Insights into the interaction between influenza virus and pneumococcus. Clin Microbiol Rev. 2006;19:571-582.
38. Grohskopf LA, Alyanak E, Broder KR, et al. Prevention and control of seasonal influenza with vaccines: recommendations of the advisory committee on immunization practices - United States, 2019-20 influenza season. MMWR Recomm Rep. 2019;68:1-21.
39. Rubins JB, Alter M, Loch J, Janoff EN. Determination of antibody responses of elderly adults to all 23 capsular polysaccharides after pneumococcal vaccination. Infect Immun. 1999;67:5979-5984.
40. Vaccines and preventable diseases. Centers for Disease Control and Prevention Web site. www.cdc.gov/vaccines/vpd/pneumo/hcp/about-vaccine.html. Accessed 16 September 2019.
41. Bonten MJ, Huijts SM, Bolkenbaas M, et al. Polysaccharide conjugate vaccine against pneumococcal pneumonia in adults. N Engl J Med. 2015;372:1114-1125.
42. Recommended adult immunization schedule -- United States -- 2019. Centers for Disease Control and Prevention Web site. www.cdc.gov/vaccines/schedules/downloads/adult/adult-combined-schedule.pdf. Accessed 16 September 2019.
43. Recommended child and adolescent immunization schedule for ages 18 years or younger – United States – 2019. Centers for Disease Control and Prevention Web site. www.cdc.gov/vaccines/schedules/hcp/imz/child-adolescent.html. Accessed 22 September 2019.
44. Pneumococcal vaccine timing for adults – United States – 2019. Centers for Disease Control and Prevention Web site. www.cdc.gov/vaccines/vpd/pneumo/downloads/pneumo-vaccine-timing.pdf. Accessed 22 September 2019.
Community-Acquired Pneumonia: Evaluation and Diagnosis
Despite advances in medical science, pneumonia remains a major cause of morbidity and mortality. In 2017, 49,157 patients in the United States died from the disease.1 Pneumonia can be classified as community-acquired, hospital-acquired, or ventilator-associated. Another category, healthcare-associated pneumonia, was included in an earlier Infectious Diseases Society of America (IDSA) and American Thoracic Society (ATS) guideline but was removed from the 2016 guideline because there was no clear evidence that patients diagnosed with healthcare-associated pneumonia were at higher risk for harboring multidrug-resistant pathogens.2 This review is the first of 2 articles focusing on the management of community-acquired pneumonia (CAP). Here, we review CAP epidemiology, microbiology, predisposing factors, and diagnosis; current treatment and prevention of CAP are reviewed in a separate article.
Definition and Epidemiology
CAP is defined as an acute infection of the lungs that develops in patients who have not been hospitalized recently and have not had regular exposure to the health care system.3 A previously ambulatory patient who is diagnosed with pneumonia within 48 hours after admission also meets the criteria for CAP. Approximately 4 to 5 million cases of CAP are diagnosed in the United States annually.4 About 25% of CAP patients require hospitalization, and about 5% to 10% of these patients are admitted to the intensive care unit (ICU).5 In-hospital mortality is considerable (~10% in population-based studies),6 and 30-day mortality was found to be as high as 23% in a review by File and Marrie.7 CAP also confers a high risk of long-term morbidity and mortality compared with the general population who have never had CAP, irrespective of age.8
Causative Organisms
Numerous microorganisms can cause CAP. Common causes and less common causes are delineated in Table 1. Until recently, many studies had demonstrated that pneumococcus was the most common cause of CAP. However, in the CDC Etiology of Pneumonia in the Community (EPIC) study team’s 2015 prospective, multicenter, population-based study, no pathogen was detected in the majority of patients diagnosed with CAP requiring hospitalization. The most common pathogens they detected were rhinovirus (9%), followed by influenza virus (6%) and pneumococcus (5%).9 Factors considered to be contributing to the decrease in the percentage of pneumococcus in patients diagnosed with CAP are the widespread use of pneumococcal vaccine and reduced rates of smoking.10,11
Predisposing Factors
Most people diagnosed with CAP have 1 or more predisposing factors (Table 2).12,13 Patients who develop CAP typically have a combination of these predisposing factors rather than a single factor. Aging, in combination with other risk factors, increases the susceptibility of a person to pneumonia.
Clinical Signs and Symptoms
Symptoms of CAP include fever, chills, rigors, fatigue, anorexia, diaphoresis, dyspnea, cough (with or without sputum production), and pleuritic chest pain. There is no individual symptom or cluster of symptoms that can absolutely differentiate pneumonia from other acute respiratory diseases, including upper and lower respiratory infections. However, patients presenting with the constellation of symptoms of fever ≥ 100°F (37.8°C), productive cough, and tachycardia is more suggestive of pneumonia.14 Abnormal vital signs include fever, hypothermia, tachypnea, tachycardia, and oxygen desaturation. Auscultation of the chest reveals crackles or other adventitious breath sounds. Elderly patients with pneumonia report a significantly lower number of both respiratory and nonrespiratory symptoms compared with younger patients. Clinicians should be aware of this phenomenon to avoid delayed diagnosis and treatment.15
Imaging Evaluation
The presence of a pulmonary consolidation or an infiltrate on chest radiograph is required to diagnose CAP, and a chest radiograph should be obtained when CAP is suspected.16 However, there is no pattern of radiographic abnormalities reliable enough to differentiate infectious pneumonia from noninfectious causes.17
There are case reports and case series demonstrating false-negative plain chest radiographs in dehydrated patients18 or in patients in a neutropenic state. However, animal studies have shown that dogs challenged with pneumococcus showed abnormal pulmonary shadow, suggestive of pneumonia, regardless of hydration status.19 There is also no reliable scientific evidence to support the notion that severe neutropenia can cause false-negative radiographs because of the inability to develop an acute inflammatory reaction in the lungs.20
A chest computed tomography (CT) scan is more sensitive than a plain chest radiograph in detecting pneumonia. Therefore, a chest CT should be performed in a patient with negative plain chest radiograph when pneumonia is still highly suspected.21 A chest CT scan is also more sensitive in detecting cavitation, adenopathy, interstitial disease, and empyema. It also has the advantage of better defining anatomical changes than plain films.22
Because improvement of pulmonary opacities in patients with CAP lags behind clinical improvement, repeating chest imaging studies is not recommended in patients who demonstrate clinical improvement. Clearing of pulmonary infiltrate or consolidation sometimes can take 6 weeks or longer.23
Laboratory Evaluation
Generally, the etiologic agent of CAP cannot be determined solely on the basis of clinical signs and symptoms or imaging studies. Although routine microbiological testing for patients suspicious for CAP is not necessary for empirical treatment, determining the etiologic agent of the pneumonia allows the clinician to narrow the antibiotics from a broad-spectrum empirical regimen to specific pathogen-directed therapy. Determination of certain etiologic agents causing the pneumonia can have important public health implications (eg, Mycobacterium tuberculosis and influenza virus).24
Sputum Gram Stain and Culture
Sputum Gram stain is an inexpensive test that may identify pathogens that cause CAP (eg, Streptococcus pneumoniae and Haemophilus influenzae). A quality specimen is required. A sputum sample must contain more than 25 neutrophils and less than 10 squamous epithelial cells/low power field on Gram stain to be considered suitable for culture. The sensitivity and specificity of sputum Gram stain and culture are highly variable in different clinical settings (eg, outpatient setting, nursing home, ICU). Reed et al’s meta-analysis of patients diagnosed with CAP in the United States showed the sensitivity and specificity of sputum Gram stain (compared with sputum culture) ranged from 15% to 100% and 11% to 100%, respectively.24 In cases of proven bacteremic pneumococcal pneumonia, positive cultures from sputum samples were positive less than 50% of the time.25
For patients who cannot provide sputum samples or are intubated, deep-suction aspirate or bronchoalveolar lavage through a bronchoscopic procedure may be necessary to obtain pulmonary secretion for Gram stain and culture. Besides bacterial culture, sputum samples can also be sent for fungal and mycobacterial cultures and acid-fast stain, if deemed clinically necessary.
The 2019 ATS/IDSA guidelines for diagnosis and treatment of adults with CAP recommend sputum culture in patients with severe disease and in all inpatients empirically treated for MRSA or Pseudomonas aeruginosa.26
Blood Culture
Because the positivity rate of blood culture in patients who are suspected to have pneumonia but not exposed to antimicrobial agents is low (5%–14%), blood cultures are not recommended for all patients with CAP. Another reason for not recommending blood culture is positive culture rarely leads to changes in antibiotic regimen in patients without underlying diseases.27 However, the 2019 ATS/IDSA guidelines recommend blood culture in patients with severe disease and in all inpatients treated empirically for MRSA or P. aeruginosa.26
A multinational study published in 2008 examined 125 patients with pneumococcal bacteremic CAP versus 1847 patients with non-bacteremic CAP.28 Analysis of the data demonstrated no association between pneumococcal bacteremic CAP and time to clinical stability, length of hospital stay, all-cause mortality, or CAP-related mortality. The authors concluded that pneumococcal bacteremia does not increase the risk of poor outcomes in patients with CAP compared to non-bacteremic patients, and the presence of pneumococcal bacteremia should not deter de-escalation of therapy in clinically stable patients.
Urinary Antigen Tests
Urinary antigen tests may assist clinicians in narrowing antibiotic therapy when test results are positive. There are 2 US Food and Drug Administration–approved tests available to clinicians for detecting pneumococcal and Legionella antigen in urine. The test for Legionella pneumophila detects disease due to serogroup 1 only, which accounts for 80% of community-acquired Legionnaires’ disease. The sensitivity and specificity of the Legionella urine antigen test are 90% and 99%, respectively. The pneumococcal urine antigen test is less sensitive and specific than the Legionella urine antigen test (sensitivity 80% and specificity > 90%).29,30
Advantages of the urinary antigen tests are that they are easily performed, results are available in less than an hour if done in-house, and results are not affected by prior exposure to antibiotics. However, the tests do not meet Clinical Laboratory Improvements Amendments criteria for waiver and must be performed by a technician in the laboratory. A multicenter, prospective surveillance study of hospitalized patients with CAP showed that the 2007 IDSA/ATS guidelines’ recommended indications for S. pneumoniae and L. pneumophila urinary antigen tests do not have sufficient sensitivity and specificity to identify patients with positive tests.31
Polymerase Chain Reaction
There are several FDA-approved polymerase chain reaction (PCR) tests commercially available to assist clinicians in diagnosing pneumonia. PCR testing of nasopharyngeal swabs for diagnosis of influenza has become standard in many US medical facilities. The great advantages of using PCR to diagnose influenza are its high sensitivity and specificity and rapid turnaround time. PCR can also be used to detect Legionella species, S. pneumonia, Mycoplasma pneumoniae, Chlamydophila pneumonia, and mycobacterial species.24
One limitation of using PCR tests on respiratory specimens is that specimens can be contaminated with oral or upper airway flora, so the results must be interpreted with caution, bearing in mind that some of the pathogens isolated may be colonizers of the oral or upper airway flora.32
Biologic Markers
Two biologic markers—procalcitonin and C-reactive protein (CRP)—can be used in conjunction with history, physical examination, laboratory tests, and imaging studies to assist in the diagnosis and treatment of CAP.24 Procalcitonin is a peptide precursor of the hormone calcitonin that is released by parenchymal cells into the bloodstream, resulting in increased serum level in patients with bacterial infections. In contrast, there is no remarkable procalcitonin level increase with viral or noninfectious inflammation. The reference value of procalcitonin in the blood of an adult individual without infection or inflammation is < 0.15 ng/mL. In the blood, procalcitonin has a half-life of 25 to 30 hours. The quantitative immunoluminometric method (LUMI test, Brahms PCT, Berlin, Germany) is the preferred test to use because of its high sensitivity.33 A meta-analysis of 12 studies involving more than 2400 patients with CAP demonstrated that serum procalcitonin does not have sufficient sensitivity or specificity to distinguish between bacterial and nonbacterial pneumonia. The authors concluded that procalcitonin level cannot be used to decide whether an antibiotic should be administered.34
A 2012 Cochrane meta-analysis that involved 4221 patients with acute respiratory infections (with half of the patients diagnosed with CAP) from 14 prospective trials found the use of procalcitonin test for antibiotic use significantly decreased median antibiotic exposure from 8 to 4 days without an increase in treatment failure, mortality rates in any clinical setting (eg, outpatient clinic, emergency room), or length of hospitalization.35 An update of the 2012 Cochrane review that examined the safety and efficacy of using procalcitonin for starting or stopping antibiotics again demonstrated procalcitonin use was associated with a reduction of antibiotic use (2.4 days).36 A prospective study conducted in France on 100 ICU patients showed that increased procalcitonin from day 1 to day 3 has a poor prognosis factor for severe CAP, whereas decreasing procalcitonin levels is associated with a favorable outcome.37
Because of conflicting data, the 2019 ATS/IDSA guidelines do not recommend using procalcitonin to determine need for initial antibacterial therapy.26
CRP is an acute phase protein produced by the liver. CRP level in the blood increases in response to acute infection or inflammation. Use of CRP in assisting diagnosis and guiding treatment of CAP is more limited in part due to its poor specificity. A prospective study conducted on 168 consecutive patients who presented with cough showed that a CRP level > 40 mg/L had a sensitivity and specificity of 70% and 90%, respectively.38
Summary
CAP remains a leading cause of hospitalization and death in the 21st century. Traditionally, pneumococcus has been considered the major pathogen causing CAP; however, the 2015 EPIC study found that S. pneumoniae was detected in only 5% of patients diagnosed with CAP. Despite the new findings, it is still recommended that empiric treatment for CAP target common typical bacteria (pneumococcus, H. influenzae, Moraxella catarrhalis) and atypical bacteria (M. pneumonia, C. pneumoniae, L. pneumophila).
Because diagnosing pneumonia through history and clinical examination is less than 50% sensitive, a chest imaging study (a plain chest radiograph or a chest CT scan) is usually required to make the diagnosis. Laboratory tests, such as sputum Gram stain/culture, blood culture, urinary antigen tests, PCR test, procalcitonin, and CRP are important adjunctive diagnostic modalities to assist in the diagnosis and management of CAP. However, because no single test is sensitive and specific enough to be a stand-alone test, they should be used in conjunction with history, physical examination, and imaging studies.
1. Centers for Disease Control and Prevention. National Center for Health Statistics. FastStats - Pneumonia. www.cdc.gov/nchs/fastats/pneumonia.htm. Accessed 16 September 2019.
2. Kalil AC, Metersky ML, Klompas M, et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 Clinical Practice Guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis. 2016;63:e61-e111.
3. Musher DM, Thorner AR. Community-acquired pneumonia. N Engl J Med. 2014;371:1619-1628.
4. Mandell LA. Epidemiology and etiology of community-acquired pneumonia. Infect Dis Clin North Am. 2004;18:761-776.
5. Hoare Z, Lim WS. Pneumonia: update on diagnosis and management. BMJ. 2006;332:1077-1079.
6. Johnstone J, Marrie TJ, Eurich DT, Majumdar SR. Effect of pneumococcal vaccination in hospitalized adults with community-acquired pneumonia. Arch Intern Med. 2007;167:1938-1943.
7. File TM Jr, Marrie TJ. Burden of community-acquired pneumonia in North American adults. Postgrad Med. 2010;122:130-141.
8. Eurich DT, Marrie TJ, Minhas-Sandhu JK, Majumdar SR. Ten-year mortality after community-acquired pneumonia. a prospective cohort. Am J Respir Crit Care Med. 2015;192:597-604.
9. Jain S, Self WH, Wunderink RG, et al. Community-acquired pneumonia requiring hospitalization among U.S. adults. N Engl J Med. 2015;373:415-427.
10. Griffin MR, Zhu Y, Moore MR, et al. U.S. hospitalizations for pneumonia after a decade of pneumococcal vaccination. N Engl J Med. 2013;369:155-163.
11. Nuorti JP, Butler JC, Farley MM, et al. Cigarette smoking and invasive pneumococcal disease. Active Bacterial Core Surveillance Team. N Engl J Med. 2000;342:681-689.
12. Almirall J, Serra-Prat M, Bolíbar I, Balasso V. Risk factors for community-acquired pneumonia in adults: a systemic review of observational studies. Respiration. 2017;94:299-311.
13. Janoff EM. Streptococcus pneumonia. In: Bennett JE, Dolin R, Blaser MJ, editors. Mandell, Douglas and Bennett’s Principles and Practice of Infectious Diseases. 8th ed. Philadelphia: Saunders; 2015:2310-2327.
14. Diehr P, Wood RW, Bushyhead J, et al. Prediction of pneumonia in outpatients with acute cough--a statistical approach. J Chronic Dis. 1984;37:215-225.
15. Metlay JP, Schulz R, Li YH, et al. Influence of age on symptoms at presentation in patients with community-acquired pneumonia. Arch Intern Med. 1997;157:1453-1459.
16. Mandell LA, Wunderink RG, Anzueto A, et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44 Suppl 2:S27-72.
17. Jartti A, Rauvala E, Kauma H, et al. Chest imaging findings in hospitalized patients with H1N1 influenza. Acta Radiol. 2011;52:297-304.
18. Basi SK, Marrie TJ, Huang JQ, Majumdar SR. Patients admitted to hospital with suspected pneumonia and normal chest radiographs: epidemiology, microbiology, and outcomes. Am J Med. 2004;117:305-311.
19. Caldwell A, Glauser FL, Smith WR, et al. The effects of dehydration on the radiologic and pathologic appearance of experimental canine segmental pneumonia. Am Rev Respir Dis. 1975;112:651-656.
20. Bartlett JG. Pneumonia. In: Barlett JG, editor. Management of Respiratory Tract Infections. Philadelphia: Lippincott, Williams & Wilkins; 2001:1-122.
21. Claessens YE, Debray MP, Tubach F, et al. Early chest computed tomography scan to assist diagnosis and guide treatment decision for suspected community-acquired pneumonia. Am J Respir Crit Care Med. 2015;192:974-982.
22. Wheeler JH, Fishman EK. Computed tomography in the management of chest infections: current status. Clin Infect Dis. 1996;23:232-240.
23. Chesnutt MP. Pulmonary disorders. In: Papadakis MM, editor. Current Medical Diagnosis and Treatment. New York: McGraw-Hill; 2016:242-320.
24. Mandell LW. Pneumonia. In: Kasper DF, editor. Harrison’s Infectious Diseases. 1st ed. New York: McGraw-Hill; 2010:188-201.
25. Reed WW, Byrd GS, Gates RH Jr, et al. Sputum gram’s stain in community-acquired pneumococcal pneumonia. A meta-analysis. West J Med. 1996;165:197-204.
26. Metlay JP, Waterer GW, Long AC, et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official Clinical Practice Guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019;200:e45-e67.
27. Chalasani NP, Valdecanas MA, Gopal AK, et al. Clinical utility of blood cultures in adult patients with community-acquired pneumonia without defined underlying risks. Chest. 1995;108:932-936.
28. Bordon J, Peyrani P, Brock GN, et al. The presence of pneumococcal bacteremia does not influence clinical outcomes in patients with community-acquired pneumonia: results from the Community-Acquired Pneumonia Organization (CAPO) International Cohort study. Chest. 2008;133:618-624.
29. Helbig JH, Uldum SA, Bernander S, et al. Clinical utility of urinary antigen detection for diagnosis of community-acquired, travel-associated, and nosocomial legionnaires’ disease. J Clin Microbiol. 2003;41:838-840.
30. Smith MD, Derrington P, Evans R, et al. Rapid diagnosis of bacteremic pneumococcal infections in adults by using the Binax NOW Streptococcus pneumoniae urinary antigen test: a prospective, controlled clinical evaluation. J Clin Microbiol. 2003;41:2810-2813.
31. Bellew S, Grijalva CG, Williams DJ, et al. Pneumococcal and Legionella urinary antigen tests in community-acquired pneumonia: Prospective evaluation of indications for testing. Clin Infect Dis. 2019;68:2026-2033.
32. Johansson N, Kalin M, Tiveljung-Lindell A, et al. Etiology of community-acquired pneumonia: increased microbiological yield with new diagnostic methods. Clin Infect Dis. 2010;50:202-209.
33. Gilbert DN. Procalcitonin as a biomarker in respiratory tract infection. Clin Infect Dis. 2011;52 Suppl 4:S346-350.
34. Kamat IS Ramachandran V, Eswaran H, et al. Procalcitonin to distinguish viral from bacterial pneumonia: A systematic review and meta-analysis. Clin Infect Dis. 2019 Jun 25. [Epub ahead of print]
35. Schuetz P, Muller B, Christ-Crain M, et al. Procalcitonin to initiate or discontinue antibiotics in acute respiratory tract infections. Cochrane Database Syst Rev. 2012;(9):CD007498.
36. Schuetz P, Wirz Y, Sager R, et al. Procalcitonin to initiate or discontinue antibiotics in acute respiratory tract infections. Cochrane Database Syst Rev. 2017;10:CD007498.
37. Boussekey N, Leroy O, Alfandari S, et al. Procalcitonin kinetics in the prognosis of severe community-acquired pneumonia. Intensive Care Med. 2006;32:469-472.
38. Flanders SA, Stein J, Shochat G, et al. Performance of a bedside C-reactive protein test in the diagnosis of community-acquired pneumonia in adults with acute cough. Am J Med. 2004;116:529-535.
Despite advances in medical science, pneumonia remains a major cause of morbidity and mortality. In 2017, 49,157 patients in the United States died from the disease.1 Pneumonia can be classified as community-acquired, hospital-acquired, or ventilator-associated. Another category, healthcare-associated pneumonia, was included in an earlier Infectious Diseases Society of America (IDSA) and American Thoracic Society (ATS) guideline but was removed from the 2016 guideline because there was no clear evidence that patients diagnosed with healthcare-associated pneumonia were at higher risk for harboring multidrug-resistant pathogens.2 This review is the first of 2 articles focusing on the management of community-acquired pneumonia (CAP). Here, we review CAP epidemiology, microbiology, predisposing factors, and diagnosis; current treatment and prevention of CAP are reviewed in a separate article.
Definition and Epidemiology
CAP is defined as an acute infection of the lungs that develops in patients who have not been hospitalized recently and have not had regular exposure to the health care system.3 A previously ambulatory patient who is diagnosed with pneumonia within 48 hours after admission also meets the criteria for CAP. Approximately 4 to 5 million cases of CAP are diagnosed in the United States annually.4 About 25% of CAP patients require hospitalization, and about 5% to 10% of these patients are admitted to the intensive care unit (ICU).5 In-hospital mortality is considerable (~10% in population-based studies),6 and 30-day mortality was found to be as high as 23% in a review by File and Marrie.7 CAP also confers a high risk of long-term morbidity and mortality compared with the general population who have never had CAP, irrespective of age.8
Causative Organisms
Numerous microorganisms can cause CAP. Common causes and less common causes are delineated in Table 1. Until recently, many studies had demonstrated that pneumococcus was the most common cause of CAP. However, in the CDC Etiology of Pneumonia in the Community (EPIC) study team’s 2015 prospective, multicenter, population-based study, no pathogen was detected in the majority of patients diagnosed with CAP requiring hospitalization. The most common pathogens they detected were rhinovirus (9%), followed by influenza virus (6%) and pneumococcus (5%).9 Factors considered to be contributing to the decrease in the percentage of pneumococcus in patients diagnosed with CAP are the widespread use of pneumococcal vaccine and reduced rates of smoking.10,11
Predisposing Factors
Most people diagnosed with CAP have 1 or more predisposing factors (Table 2).12,13 Patients who develop CAP typically have a combination of these predisposing factors rather than a single factor. Aging, in combination with other risk factors, increases the susceptibility of a person to pneumonia.
Clinical Signs and Symptoms
Symptoms of CAP include fever, chills, rigors, fatigue, anorexia, diaphoresis, dyspnea, cough (with or without sputum production), and pleuritic chest pain. There is no individual symptom or cluster of symptoms that can absolutely differentiate pneumonia from other acute respiratory diseases, including upper and lower respiratory infections. However, patients presenting with the constellation of symptoms of fever ≥ 100°F (37.8°C), productive cough, and tachycardia is more suggestive of pneumonia.14 Abnormal vital signs include fever, hypothermia, tachypnea, tachycardia, and oxygen desaturation. Auscultation of the chest reveals crackles or other adventitious breath sounds. Elderly patients with pneumonia report a significantly lower number of both respiratory and nonrespiratory symptoms compared with younger patients. Clinicians should be aware of this phenomenon to avoid delayed diagnosis and treatment.15
Imaging Evaluation
The presence of a pulmonary consolidation or an infiltrate on chest radiograph is required to diagnose CAP, and a chest radiograph should be obtained when CAP is suspected.16 However, there is no pattern of radiographic abnormalities reliable enough to differentiate infectious pneumonia from noninfectious causes.17
There are case reports and case series demonstrating false-negative plain chest radiographs in dehydrated patients18 or in patients in a neutropenic state. However, animal studies have shown that dogs challenged with pneumococcus showed abnormal pulmonary shadow, suggestive of pneumonia, regardless of hydration status.19 There is also no reliable scientific evidence to support the notion that severe neutropenia can cause false-negative radiographs because of the inability to develop an acute inflammatory reaction in the lungs.20
A chest computed tomography (CT) scan is more sensitive than a plain chest radiograph in detecting pneumonia. Therefore, a chest CT should be performed in a patient with negative plain chest radiograph when pneumonia is still highly suspected.21 A chest CT scan is also more sensitive in detecting cavitation, adenopathy, interstitial disease, and empyema. It also has the advantage of better defining anatomical changes than plain films.22
Because improvement of pulmonary opacities in patients with CAP lags behind clinical improvement, repeating chest imaging studies is not recommended in patients who demonstrate clinical improvement. Clearing of pulmonary infiltrate or consolidation sometimes can take 6 weeks or longer.23
Laboratory Evaluation
Generally, the etiologic agent of CAP cannot be determined solely on the basis of clinical signs and symptoms or imaging studies. Although routine microbiological testing for patients suspicious for CAP is not necessary for empirical treatment, determining the etiologic agent of the pneumonia allows the clinician to narrow the antibiotics from a broad-spectrum empirical regimen to specific pathogen-directed therapy. Determination of certain etiologic agents causing the pneumonia can have important public health implications (eg, Mycobacterium tuberculosis and influenza virus).24
Sputum Gram Stain and Culture
Sputum Gram stain is an inexpensive test that may identify pathogens that cause CAP (eg, Streptococcus pneumoniae and Haemophilus influenzae). A quality specimen is required. A sputum sample must contain more than 25 neutrophils and less than 10 squamous epithelial cells/low power field on Gram stain to be considered suitable for culture. The sensitivity and specificity of sputum Gram stain and culture are highly variable in different clinical settings (eg, outpatient setting, nursing home, ICU). Reed et al’s meta-analysis of patients diagnosed with CAP in the United States showed the sensitivity and specificity of sputum Gram stain (compared with sputum culture) ranged from 15% to 100% and 11% to 100%, respectively.24 In cases of proven bacteremic pneumococcal pneumonia, positive cultures from sputum samples were positive less than 50% of the time.25
For patients who cannot provide sputum samples or are intubated, deep-suction aspirate or bronchoalveolar lavage through a bronchoscopic procedure may be necessary to obtain pulmonary secretion for Gram stain and culture. Besides bacterial culture, sputum samples can also be sent for fungal and mycobacterial cultures and acid-fast stain, if deemed clinically necessary.
The 2019 ATS/IDSA guidelines for diagnosis and treatment of adults with CAP recommend sputum culture in patients with severe disease and in all inpatients empirically treated for MRSA or Pseudomonas aeruginosa.26
Blood Culture
Because the positivity rate of blood culture in patients who are suspected to have pneumonia but not exposed to antimicrobial agents is low (5%–14%), blood cultures are not recommended for all patients with CAP. Another reason for not recommending blood culture is positive culture rarely leads to changes in antibiotic regimen in patients without underlying diseases.27 However, the 2019 ATS/IDSA guidelines recommend blood culture in patients with severe disease and in all inpatients treated empirically for MRSA or P. aeruginosa.26
A multinational study published in 2008 examined 125 patients with pneumococcal bacteremic CAP versus 1847 patients with non-bacteremic CAP.28 Analysis of the data demonstrated no association between pneumococcal bacteremic CAP and time to clinical stability, length of hospital stay, all-cause mortality, or CAP-related mortality. The authors concluded that pneumococcal bacteremia does not increase the risk of poor outcomes in patients with CAP compared to non-bacteremic patients, and the presence of pneumococcal bacteremia should not deter de-escalation of therapy in clinically stable patients.
Urinary Antigen Tests
Urinary antigen tests may assist clinicians in narrowing antibiotic therapy when test results are positive. There are 2 US Food and Drug Administration–approved tests available to clinicians for detecting pneumococcal and Legionella antigen in urine. The test for Legionella pneumophila detects disease due to serogroup 1 only, which accounts for 80% of community-acquired Legionnaires’ disease. The sensitivity and specificity of the Legionella urine antigen test are 90% and 99%, respectively. The pneumococcal urine antigen test is less sensitive and specific than the Legionella urine antigen test (sensitivity 80% and specificity > 90%).29,30
Advantages of the urinary antigen tests are that they are easily performed, results are available in less than an hour if done in-house, and results are not affected by prior exposure to antibiotics. However, the tests do not meet Clinical Laboratory Improvements Amendments criteria for waiver and must be performed by a technician in the laboratory. A multicenter, prospective surveillance study of hospitalized patients with CAP showed that the 2007 IDSA/ATS guidelines’ recommended indications for S. pneumoniae and L. pneumophila urinary antigen tests do not have sufficient sensitivity and specificity to identify patients with positive tests.31
Polymerase Chain Reaction
There are several FDA-approved polymerase chain reaction (PCR) tests commercially available to assist clinicians in diagnosing pneumonia. PCR testing of nasopharyngeal swabs for diagnosis of influenza has become standard in many US medical facilities. The great advantages of using PCR to diagnose influenza are its high sensitivity and specificity and rapid turnaround time. PCR can also be used to detect Legionella species, S. pneumonia, Mycoplasma pneumoniae, Chlamydophila pneumonia, and mycobacterial species.24
One limitation of using PCR tests on respiratory specimens is that specimens can be contaminated with oral or upper airway flora, so the results must be interpreted with caution, bearing in mind that some of the pathogens isolated may be colonizers of the oral or upper airway flora.32
Biologic Markers
Two biologic markers—procalcitonin and C-reactive protein (CRP)—can be used in conjunction with history, physical examination, laboratory tests, and imaging studies to assist in the diagnosis and treatment of CAP.24 Procalcitonin is a peptide precursor of the hormone calcitonin that is released by parenchymal cells into the bloodstream, resulting in increased serum level in patients with bacterial infections. In contrast, there is no remarkable procalcitonin level increase with viral or noninfectious inflammation. The reference value of procalcitonin in the blood of an adult individual without infection or inflammation is < 0.15 ng/mL. In the blood, procalcitonin has a half-life of 25 to 30 hours. The quantitative immunoluminometric method (LUMI test, Brahms PCT, Berlin, Germany) is the preferred test to use because of its high sensitivity.33 A meta-analysis of 12 studies involving more than 2400 patients with CAP demonstrated that serum procalcitonin does not have sufficient sensitivity or specificity to distinguish between bacterial and nonbacterial pneumonia. The authors concluded that procalcitonin level cannot be used to decide whether an antibiotic should be administered.34
A 2012 Cochrane meta-analysis that involved 4221 patients with acute respiratory infections (with half of the patients diagnosed with CAP) from 14 prospective trials found the use of procalcitonin test for antibiotic use significantly decreased median antibiotic exposure from 8 to 4 days without an increase in treatment failure, mortality rates in any clinical setting (eg, outpatient clinic, emergency room), or length of hospitalization.35 An update of the 2012 Cochrane review that examined the safety and efficacy of using procalcitonin for starting or stopping antibiotics again demonstrated procalcitonin use was associated with a reduction of antibiotic use (2.4 days).36 A prospective study conducted in France on 100 ICU patients showed that increased procalcitonin from day 1 to day 3 has a poor prognosis factor for severe CAP, whereas decreasing procalcitonin levels is associated with a favorable outcome.37
Because of conflicting data, the 2019 ATS/IDSA guidelines do not recommend using procalcitonin to determine need for initial antibacterial therapy.26
CRP is an acute phase protein produced by the liver. CRP level in the blood increases in response to acute infection or inflammation. Use of CRP in assisting diagnosis and guiding treatment of CAP is more limited in part due to its poor specificity. A prospective study conducted on 168 consecutive patients who presented with cough showed that a CRP level > 40 mg/L had a sensitivity and specificity of 70% and 90%, respectively.38
Summary
CAP remains a leading cause of hospitalization and death in the 21st century. Traditionally, pneumococcus has been considered the major pathogen causing CAP; however, the 2015 EPIC study found that S. pneumoniae was detected in only 5% of patients diagnosed with CAP. Despite the new findings, it is still recommended that empiric treatment for CAP target common typical bacteria (pneumococcus, H. influenzae, Moraxella catarrhalis) and atypical bacteria (M. pneumonia, C. pneumoniae, L. pneumophila).
Because diagnosing pneumonia through history and clinical examination is less than 50% sensitive, a chest imaging study (a plain chest radiograph or a chest CT scan) is usually required to make the diagnosis. Laboratory tests, such as sputum Gram stain/culture, blood culture, urinary antigen tests, PCR test, procalcitonin, and CRP are important adjunctive diagnostic modalities to assist in the diagnosis and management of CAP. However, because no single test is sensitive and specific enough to be a stand-alone test, they should be used in conjunction with history, physical examination, and imaging studies.
Despite advances in medical science, pneumonia remains a major cause of morbidity and mortality. In 2017, 49,157 patients in the United States died from the disease.1 Pneumonia can be classified as community-acquired, hospital-acquired, or ventilator-associated. Another category, healthcare-associated pneumonia, was included in an earlier Infectious Diseases Society of America (IDSA) and American Thoracic Society (ATS) guideline but was removed from the 2016 guideline because there was no clear evidence that patients diagnosed with healthcare-associated pneumonia were at higher risk for harboring multidrug-resistant pathogens.2 This review is the first of 2 articles focusing on the management of community-acquired pneumonia (CAP). Here, we review CAP epidemiology, microbiology, predisposing factors, and diagnosis; current treatment and prevention of CAP are reviewed in a separate article.
Definition and Epidemiology
CAP is defined as an acute infection of the lungs that develops in patients who have not been hospitalized recently and have not had regular exposure to the health care system.3 A previously ambulatory patient who is diagnosed with pneumonia within 48 hours after admission also meets the criteria for CAP. Approximately 4 to 5 million cases of CAP are diagnosed in the United States annually.4 About 25% of CAP patients require hospitalization, and about 5% to 10% of these patients are admitted to the intensive care unit (ICU).5 In-hospital mortality is considerable (~10% in population-based studies),6 and 30-day mortality was found to be as high as 23% in a review by File and Marrie.7 CAP also confers a high risk of long-term morbidity and mortality compared with the general population who have never had CAP, irrespective of age.8
Causative Organisms
Numerous microorganisms can cause CAP. Common causes and less common causes are delineated in Table 1. Until recently, many studies had demonstrated that pneumococcus was the most common cause of CAP. However, in the CDC Etiology of Pneumonia in the Community (EPIC) study team’s 2015 prospective, multicenter, population-based study, no pathogen was detected in the majority of patients diagnosed with CAP requiring hospitalization. The most common pathogens they detected were rhinovirus (9%), followed by influenza virus (6%) and pneumococcus (5%).9 Factors considered to be contributing to the decrease in the percentage of pneumococcus in patients diagnosed with CAP are the widespread use of pneumococcal vaccine and reduced rates of smoking.10,11
Predisposing Factors
Most people diagnosed with CAP have 1 or more predisposing factors (Table 2).12,13 Patients who develop CAP typically have a combination of these predisposing factors rather than a single factor. Aging, in combination with other risk factors, increases the susceptibility of a person to pneumonia.
Clinical Signs and Symptoms
Symptoms of CAP include fever, chills, rigors, fatigue, anorexia, diaphoresis, dyspnea, cough (with or without sputum production), and pleuritic chest pain. There is no individual symptom or cluster of symptoms that can absolutely differentiate pneumonia from other acute respiratory diseases, including upper and lower respiratory infections. However, patients presenting with the constellation of symptoms of fever ≥ 100°F (37.8°C), productive cough, and tachycardia is more suggestive of pneumonia.14 Abnormal vital signs include fever, hypothermia, tachypnea, tachycardia, and oxygen desaturation. Auscultation of the chest reveals crackles or other adventitious breath sounds. Elderly patients with pneumonia report a significantly lower number of both respiratory and nonrespiratory symptoms compared with younger patients. Clinicians should be aware of this phenomenon to avoid delayed diagnosis and treatment.15
Imaging Evaluation
The presence of a pulmonary consolidation or an infiltrate on chest radiograph is required to diagnose CAP, and a chest radiograph should be obtained when CAP is suspected.16 However, there is no pattern of radiographic abnormalities reliable enough to differentiate infectious pneumonia from noninfectious causes.17
There are case reports and case series demonstrating false-negative plain chest radiographs in dehydrated patients18 or in patients in a neutropenic state. However, animal studies have shown that dogs challenged with pneumococcus showed abnormal pulmonary shadow, suggestive of pneumonia, regardless of hydration status.19 There is also no reliable scientific evidence to support the notion that severe neutropenia can cause false-negative radiographs because of the inability to develop an acute inflammatory reaction in the lungs.20
A chest computed tomography (CT) scan is more sensitive than a plain chest radiograph in detecting pneumonia. Therefore, a chest CT should be performed in a patient with negative plain chest radiograph when pneumonia is still highly suspected.21 A chest CT scan is also more sensitive in detecting cavitation, adenopathy, interstitial disease, and empyema. It also has the advantage of better defining anatomical changes than plain films.22
Because improvement of pulmonary opacities in patients with CAP lags behind clinical improvement, repeating chest imaging studies is not recommended in patients who demonstrate clinical improvement. Clearing of pulmonary infiltrate or consolidation sometimes can take 6 weeks or longer.23
Laboratory Evaluation
Generally, the etiologic agent of CAP cannot be determined solely on the basis of clinical signs and symptoms or imaging studies. Although routine microbiological testing for patients suspicious for CAP is not necessary for empirical treatment, determining the etiologic agent of the pneumonia allows the clinician to narrow the antibiotics from a broad-spectrum empirical regimen to specific pathogen-directed therapy. Determination of certain etiologic agents causing the pneumonia can have important public health implications (eg, Mycobacterium tuberculosis and influenza virus).24
Sputum Gram Stain and Culture
Sputum Gram stain is an inexpensive test that may identify pathogens that cause CAP (eg, Streptococcus pneumoniae and Haemophilus influenzae). A quality specimen is required. A sputum sample must contain more than 25 neutrophils and less than 10 squamous epithelial cells/low power field on Gram stain to be considered suitable for culture. The sensitivity and specificity of sputum Gram stain and culture are highly variable in different clinical settings (eg, outpatient setting, nursing home, ICU). Reed et al’s meta-analysis of patients diagnosed with CAP in the United States showed the sensitivity and specificity of sputum Gram stain (compared with sputum culture) ranged from 15% to 100% and 11% to 100%, respectively.24 In cases of proven bacteremic pneumococcal pneumonia, positive cultures from sputum samples were positive less than 50% of the time.25
For patients who cannot provide sputum samples or are intubated, deep-suction aspirate or bronchoalveolar lavage through a bronchoscopic procedure may be necessary to obtain pulmonary secretion for Gram stain and culture. Besides bacterial culture, sputum samples can also be sent for fungal and mycobacterial cultures and acid-fast stain, if deemed clinically necessary.
The 2019 ATS/IDSA guidelines for diagnosis and treatment of adults with CAP recommend sputum culture in patients with severe disease and in all inpatients empirically treated for MRSA or Pseudomonas aeruginosa.26
Blood Culture
Because the positivity rate of blood culture in patients who are suspected to have pneumonia but not exposed to antimicrobial agents is low (5%–14%), blood cultures are not recommended for all patients with CAP. Another reason for not recommending blood culture is positive culture rarely leads to changes in antibiotic regimen in patients without underlying diseases.27 However, the 2019 ATS/IDSA guidelines recommend blood culture in patients with severe disease and in all inpatients treated empirically for MRSA or P. aeruginosa.26
A multinational study published in 2008 examined 125 patients with pneumococcal bacteremic CAP versus 1847 patients with non-bacteremic CAP.28 Analysis of the data demonstrated no association between pneumococcal bacteremic CAP and time to clinical stability, length of hospital stay, all-cause mortality, or CAP-related mortality. The authors concluded that pneumococcal bacteremia does not increase the risk of poor outcomes in patients with CAP compared to non-bacteremic patients, and the presence of pneumococcal bacteremia should not deter de-escalation of therapy in clinically stable patients.
Urinary Antigen Tests
Urinary antigen tests may assist clinicians in narrowing antibiotic therapy when test results are positive. There are 2 US Food and Drug Administration–approved tests available to clinicians for detecting pneumococcal and Legionella antigen in urine. The test for Legionella pneumophila detects disease due to serogroup 1 only, which accounts for 80% of community-acquired Legionnaires’ disease. The sensitivity and specificity of the Legionella urine antigen test are 90% and 99%, respectively. The pneumococcal urine antigen test is less sensitive and specific than the Legionella urine antigen test (sensitivity 80% and specificity > 90%).29,30
Advantages of the urinary antigen tests are that they are easily performed, results are available in less than an hour if done in-house, and results are not affected by prior exposure to antibiotics. However, the tests do not meet Clinical Laboratory Improvements Amendments criteria for waiver and must be performed by a technician in the laboratory. A multicenter, prospective surveillance study of hospitalized patients with CAP showed that the 2007 IDSA/ATS guidelines’ recommended indications for S. pneumoniae and L. pneumophila urinary antigen tests do not have sufficient sensitivity and specificity to identify patients with positive tests.31
Polymerase Chain Reaction
There are several FDA-approved polymerase chain reaction (PCR) tests commercially available to assist clinicians in diagnosing pneumonia. PCR testing of nasopharyngeal swabs for diagnosis of influenza has become standard in many US medical facilities. The great advantages of using PCR to diagnose influenza are its high sensitivity and specificity and rapid turnaround time. PCR can also be used to detect Legionella species, S. pneumonia, Mycoplasma pneumoniae, Chlamydophila pneumonia, and mycobacterial species.24
One limitation of using PCR tests on respiratory specimens is that specimens can be contaminated with oral or upper airway flora, so the results must be interpreted with caution, bearing in mind that some of the pathogens isolated may be colonizers of the oral or upper airway flora.32
Biologic Markers
Two biologic markers—procalcitonin and C-reactive protein (CRP)—can be used in conjunction with history, physical examination, laboratory tests, and imaging studies to assist in the diagnosis and treatment of CAP.24 Procalcitonin is a peptide precursor of the hormone calcitonin that is released by parenchymal cells into the bloodstream, resulting in increased serum level in patients with bacterial infections. In contrast, there is no remarkable procalcitonin level increase with viral or noninfectious inflammation. The reference value of procalcitonin in the blood of an adult individual without infection or inflammation is < 0.15 ng/mL. In the blood, procalcitonin has a half-life of 25 to 30 hours. The quantitative immunoluminometric method (LUMI test, Brahms PCT, Berlin, Germany) is the preferred test to use because of its high sensitivity.33 A meta-analysis of 12 studies involving more than 2400 patients with CAP demonstrated that serum procalcitonin does not have sufficient sensitivity or specificity to distinguish between bacterial and nonbacterial pneumonia. The authors concluded that procalcitonin level cannot be used to decide whether an antibiotic should be administered.34
A 2012 Cochrane meta-analysis that involved 4221 patients with acute respiratory infections (with half of the patients diagnosed with CAP) from 14 prospective trials found the use of procalcitonin test for antibiotic use significantly decreased median antibiotic exposure from 8 to 4 days without an increase in treatment failure, mortality rates in any clinical setting (eg, outpatient clinic, emergency room), or length of hospitalization.35 An update of the 2012 Cochrane review that examined the safety and efficacy of using procalcitonin for starting or stopping antibiotics again demonstrated procalcitonin use was associated with a reduction of antibiotic use (2.4 days).36 A prospective study conducted in France on 100 ICU patients showed that increased procalcitonin from day 1 to day 3 has a poor prognosis factor for severe CAP, whereas decreasing procalcitonin levels is associated with a favorable outcome.37
Because of conflicting data, the 2019 ATS/IDSA guidelines do not recommend using procalcitonin to determine need for initial antibacterial therapy.26
CRP is an acute phase protein produced by the liver. CRP level in the blood increases in response to acute infection or inflammation. Use of CRP in assisting diagnosis and guiding treatment of CAP is more limited in part due to its poor specificity. A prospective study conducted on 168 consecutive patients who presented with cough showed that a CRP level > 40 mg/L had a sensitivity and specificity of 70% and 90%, respectively.38
Summary
CAP remains a leading cause of hospitalization and death in the 21st century. Traditionally, pneumococcus has been considered the major pathogen causing CAP; however, the 2015 EPIC study found that S. pneumoniae was detected in only 5% of patients diagnosed with CAP. Despite the new findings, it is still recommended that empiric treatment for CAP target common typical bacteria (pneumococcus, H. influenzae, Moraxella catarrhalis) and atypical bacteria (M. pneumonia, C. pneumoniae, L. pneumophila).
Because diagnosing pneumonia through history and clinical examination is less than 50% sensitive, a chest imaging study (a plain chest radiograph or a chest CT scan) is usually required to make the diagnosis. Laboratory tests, such as sputum Gram stain/culture, blood culture, urinary antigen tests, PCR test, procalcitonin, and CRP are important adjunctive diagnostic modalities to assist in the diagnosis and management of CAP. However, because no single test is sensitive and specific enough to be a stand-alone test, they should be used in conjunction with history, physical examination, and imaging studies.
1. Centers for Disease Control and Prevention. National Center for Health Statistics. FastStats - Pneumonia. www.cdc.gov/nchs/fastats/pneumonia.htm. Accessed 16 September 2019.
2. Kalil AC, Metersky ML, Klompas M, et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 Clinical Practice Guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis. 2016;63:e61-e111.
3. Musher DM, Thorner AR. Community-acquired pneumonia. N Engl J Med. 2014;371:1619-1628.
4. Mandell LA. Epidemiology and etiology of community-acquired pneumonia. Infect Dis Clin North Am. 2004;18:761-776.
5. Hoare Z, Lim WS. Pneumonia: update on diagnosis and management. BMJ. 2006;332:1077-1079.
6. Johnstone J, Marrie TJ, Eurich DT, Majumdar SR. Effect of pneumococcal vaccination in hospitalized adults with community-acquired pneumonia. Arch Intern Med. 2007;167:1938-1943.
7. File TM Jr, Marrie TJ. Burden of community-acquired pneumonia in North American adults. Postgrad Med. 2010;122:130-141.
8. Eurich DT, Marrie TJ, Minhas-Sandhu JK, Majumdar SR. Ten-year mortality after community-acquired pneumonia. a prospective cohort. Am J Respir Crit Care Med. 2015;192:597-604.
9. Jain S, Self WH, Wunderink RG, et al. Community-acquired pneumonia requiring hospitalization among U.S. adults. N Engl J Med. 2015;373:415-427.
10. Griffin MR, Zhu Y, Moore MR, et al. U.S. hospitalizations for pneumonia after a decade of pneumococcal vaccination. N Engl J Med. 2013;369:155-163.
11. Nuorti JP, Butler JC, Farley MM, et al. Cigarette smoking and invasive pneumococcal disease. Active Bacterial Core Surveillance Team. N Engl J Med. 2000;342:681-689.
12. Almirall J, Serra-Prat M, Bolíbar I, Balasso V. Risk factors for community-acquired pneumonia in adults: a systemic review of observational studies. Respiration. 2017;94:299-311.
13. Janoff EM. Streptococcus pneumonia. In: Bennett JE, Dolin R, Blaser MJ, editors. Mandell, Douglas and Bennett’s Principles and Practice of Infectious Diseases. 8th ed. Philadelphia: Saunders; 2015:2310-2327.
14. Diehr P, Wood RW, Bushyhead J, et al. Prediction of pneumonia in outpatients with acute cough--a statistical approach. J Chronic Dis. 1984;37:215-225.
15. Metlay JP, Schulz R, Li YH, et al. Influence of age on symptoms at presentation in patients with community-acquired pneumonia. Arch Intern Med. 1997;157:1453-1459.
16. Mandell LA, Wunderink RG, Anzueto A, et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44 Suppl 2:S27-72.
17. Jartti A, Rauvala E, Kauma H, et al. Chest imaging findings in hospitalized patients with H1N1 influenza. Acta Radiol. 2011;52:297-304.
18. Basi SK, Marrie TJ, Huang JQ, Majumdar SR. Patients admitted to hospital with suspected pneumonia and normal chest radiographs: epidemiology, microbiology, and outcomes. Am J Med. 2004;117:305-311.
19. Caldwell A, Glauser FL, Smith WR, et al. The effects of dehydration on the radiologic and pathologic appearance of experimental canine segmental pneumonia. Am Rev Respir Dis. 1975;112:651-656.
20. Bartlett JG. Pneumonia. In: Barlett JG, editor. Management of Respiratory Tract Infections. Philadelphia: Lippincott, Williams & Wilkins; 2001:1-122.
21. Claessens YE, Debray MP, Tubach F, et al. Early chest computed tomography scan to assist diagnosis and guide treatment decision for suspected community-acquired pneumonia. Am J Respir Crit Care Med. 2015;192:974-982.
22. Wheeler JH, Fishman EK. Computed tomography in the management of chest infections: current status. Clin Infect Dis. 1996;23:232-240.
23. Chesnutt MP. Pulmonary disorders. In: Papadakis MM, editor. Current Medical Diagnosis and Treatment. New York: McGraw-Hill; 2016:242-320.
24. Mandell LW. Pneumonia. In: Kasper DF, editor. Harrison’s Infectious Diseases. 1st ed. New York: McGraw-Hill; 2010:188-201.
25. Reed WW, Byrd GS, Gates RH Jr, et al. Sputum gram’s stain in community-acquired pneumococcal pneumonia. A meta-analysis. West J Med. 1996;165:197-204.
26. Metlay JP, Waterer GW, Long AC, et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official Clinical Practice Guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019;200:e45-e67.
27. Chalasani NP, Valdecanas MA, Gopal AK, et al. Clinical utility of blood cultures in adult patients with community-acquired pneumonia without defined underlying risks. Chest. 1995;108:932-936.
28. Bordon J, Peyrani P, Brock GN, et al. The presence of pneumococcal bacteremia does not influence clinical outcomes in patients with community-acquired pneumonia: results from the Community-Acquired Pneumonia Organization (CAPO) International Cohort study. Chest. 2008;133:618-624.
29. Helbig JH, Uldum SA, Bernander S, et al. Clinical utility of urinary antigen detection for diagnosis of community-acquired, travel-associated, and nosocomial legionnaires’ disease. J Clin Microbiol. 2003;41:838-840.
30. Smith MD, Derrington P, Evans R, et al. Rapid diagnosis of bacteremic pneumococcal infections in adults by using the Binax NOW Streptococcus pneumoniae urinary antigen test: a prospective, controlled clinical evaluation. J Clin Microbiol. 2003;41:2810-2813.
31. Bellew S, Grijalva CG, Williams DJ, et al. Pneumococcal and Legionella urinary antigen tests in community-acquired pneumonia: Prospective evaluation of indications for testing. Clin Infect Dis. 2019;68:2026-2033.
32. Johansson N, Kalin M, Tiveljung-Lindell A, et al. Etiology of community-acquired pneumonia: increased microbiological yield with new diagnostic methods. Clin Infect Dis. 2010;50:202-209.
33. Gilbert DN. Procalcitonin as a biomarker in respiratory tract infection. Clin Infect Dis. 2011;52 Suppl 4:S346-350.
34. Kamat IS Ramachandran V, Eswaran H, et al. Procalcitonin to distinguish viral from bacterial pneumonia: A systematic review and meta-analysis. Clin Infect Dis. 2019 Jun 25. [Epub ahead of print]
35. Schuetz P, Muller B, Christ-Crain M, et al. Procalcitonin to initiate or discontinue antibiotics in acute respiratory tract infections. Cochrane Database Syst Rev. 2012;(9):CD007498.
36. Schuetz P, Wirz Y, Sager R, et al. Procalcitonin to initiate or discontinue antibiotics in acute respiratory tract infections. Cochrane Database Syst Rev. 2017;10:CD007498.
37. Boussekey N, Leroy O, Alfandari S, et al. Procalcitonin kinetics in the prognosis of severe community-acquired pneumonia. Intensive Care Med. 2006;32:469-472.
38. Flanders SA, Stein J, Shochat G, et al. Performance of a bedside C-reactive protein test in the diagnosis of community-acquired pneumonia in adults with acute cough. Am J Med. 2004;116:529-535.
1. Centers for Disease Control and Prevention. National Center for Health Statistics. FastStats - Pneumonia. www.cdc.gov/nchs/fastats/pneumonia.htm. Accessed 16 September 2019.
2. Kalil AC, Metersky ML, Klompas M, et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 Clinical Practice Guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis. 2016;63:e61-e111.
3. Musher DM, Thorner AR. Community-acquired pneumonia. N Engl J Med. 2014;371:1619-1628.
4. Mandell LA. Epidemiology and etiology of community-acquired pneumonia. Infect Dis Clin North Am. 2004;18:761-776.
5. Hoare Z, Lim WS. Pneumonia: update on diagnosis and management. BMJ. 2006;332:1077-1079.
6. Johnstone J, Marrie TJ, Eurich DT, Majumdar SR. Effect of pneumococcal vaccination in hospitalized adults with community-acquired pneumonia. Arch Intern Med. 2007;167:1938-1943.
7. File TM Jr, Marrie TJ. Burden of community-acquired pneumonia in North American adults. Postgrad Med. 2010;122:130-141.
8. Eurich DT, Marrie TJ, Minhas-Sandhu JK, Majumdar SR. Ten-year mortality after community-acquired pneumonia. a prospective cohort. Am J Respir Crit Care Med. 2015;192:597-604.
9. Jain S, Self WH, Wunderink RG, et al. Community-acquired pneumonia requiring hospitalization among U.S. adults. N Engl J Med. 2015;373:415-427.
10. Griffin MR, Zhu Y, Moore MR, et al. U.S. hospitalizations for pneumonia after a decade of pneumococcal vaccination. N Engl J Med. 2013;369:155-163.
11. Nuorti JP, Butler JC, Farley MM, et al. Cigarette smoking and invasive pneumococcal disease. Active Bacterial Core Surveillance Team. N Engl J Med. 2000;342:681-689.
12. Almirall J, Serra-Prat M, Bolíbar I, Balasso V. Risk factors for community-acquired pneumonia in adults: a systemic review of observational studies. Respiration. 2017;94:299-311.
13. Janoff EM. Streptococcus pneumonia. In: Bennett JE, Dolin R, Blaser MJ, editors. Mandell, Douglas and Bennett’s Principles and Practice of Infectious Diseases. 8th ed. Philadelphia: Saunders; 2015:2310-2327.
14. Diehr P, Wood RW, Bushyhead J, et al. Prediction of pneumonia in outpatients with acute cough--a statistical approach. J Chronic Dis. 1984;37:215-225.
15. Metlay JP, Schulz R, Li YH, et al. Influence of age on symptoms at presentation in patients with community-acquired pneumonia. Arch Intern Med. 1997;157:1453-1459.
16. Mandell LA, Wunderink RG, Anzueto A, et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44 Suppl 2:S27-72.
17. Jartti A, Rauvala E, Kauma H, et al. Chest imaging findings in hospitalized patients with H1N1 influenza. Acta Radiol. 2011;52:297-304.
18. Basi SK, Marrie TJ, Huang JQ, Majumdar SR. Patients admitted to hospital with suspected pneumonia and normal chest radiographs: epidemiology, microbiology, and outcomes. Am J Med. 2004;117:305-311.
19. Caldwell A, Glauser FL, Smith WR, et al. The effects of dehydration on the radiologic and pathologic appearance of experimental canine segmental pneumonia. Am Rev Respir Dis. 1975;112:651-656.
20. Bartlett JG. Pneumonia. In: Barlett JG, editor. Management of Respiratory Tract Infections. Philadelphia: Lippincott, Williams & Wilkins; 2001:1-122.
21. Claessens YE, Debray MP, Tubach F, et al. Early chest computed tomography scan to assist diagnosis and guide treatment decision for suspected community-acquired pneumonia. Am J Respir Crit Care Med. 2015;192:974-982.
22. Wheeler JH, Fishman EK. Computed tomography in the management of chest infections: current status. Clin Infect Dis. 1996;23:232-240.
23. Chesnutt MP. Pulmonary disorders. In: Papadakis MM, editor. Current Medical Diagnosis and Treatment. New York: McGraw-Hill; 2016:242-320.
24. Mandell LW. Pneumonia. In: Kasper DF, editor. Harrison’s Infectious Diseases. 1st ed. New York: McGraw-Hill; 2010:188-201.
25. Reed WW, Byrd GS, Gates RH Jr, et al. Sputum gram’s stain in community-acquired pneumococcal pneumonia. A meta-analysis. West J Med. 1996;165:197-204.
26. Metlay JP, Waterer GW, Long AC, et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official Clinical Practice Guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. 2019;200:e45-e67.
27. Chalasani NP, Valdecanas MA, Gopal AK, et al. Clinical utility of blood cultures in adult patients with community-acquired pneumonia without defined underlying risks. Chest. 1995;108:932-936.
28. Bordon J, Peyrani P, Brock GN, et al. The presence of pneumococcal bacteremia does not influence clinical outcomes in patients with community-acquired pneumonia: results from the Community-Acquired Pneumonia Organization (CAPO) International Cohort study. Chest. 2008;133:618-624.
29. Helbig JH, Uldum SA, Bernander S, et al. Clinical utility of urinary antigen detection for diagnosis of community-acquired, travel-associated, and nosocomial legionnaires’ disease. J Clin Microbiol. 2003;41:838-840.
30. Smith MD, Derrington P, Evans R, et al. Rapid diagnosis of bacteremic pneumococcal infections in adults by using the Binax NOW Streptococcus pneumoniae urinary antigen test: a prospective, controlled clinical evaluation. J Clin Microbiol. 2003;41:2810-2813.
31. Bellew S, Grijalva CG, Williams DJ, et al. Pneumococcal and Legionella urinary antigen tests in community-acquired pneumonia: Prospective evaluation of indications for testing. Clin Infect Dis. 2019;68:2026-2033.
32. Johansson N, Kalin M, Tiveljung-Lindell A, et al. Etiology of community-acquired pneumonia: increased microbiological yield with new diagnostic methods. Clin Infect Dis. 2010;50:202-209.
33. Gilbert DN. Procalcitonin as a biomarker in respiratory tract infection. Clin Infect Dis. 2011;52 Suppl 4:S346-350.
34. Kamat IS Ramachandran V, Eswaran H, et al. Procalcitonin to distinguish viral from bacterial pneumonia: A systematic review and meta-analysis. Clin Infect Dis. 2019 Jun 25. [Epub ahead of print]
35. Schuetz P, Muller B, Christ-Crain M, et al. Procalcitonin to initiate or discontinue antibiotics in acute respiratory tract infections. Cochrane Database Syst Rev. 2012;(9):CD007498.
36. Schuetz P, Wirz Y, Sager R, et al. Procalcitonin to initiate or discontinue antibiotics in acute respiratory tract infections. Cochrane Database Syst Rev. 2017;10:CD007498.
37. Boussekey N, Leroy O, Alfandari S, et al. Procalcitonin kinetics in the prognosis of severe community-acquired pneumonia. Intensive Care Med. 2006;32:469-472.
38. Flanders SA, Stein J, Shochat G, et al. Performance of a bedside C-reactive protein test in the diagnosis of community-acquired pneumonia in adults with acute cough. Am J Med. 2004;116:529-535.
COMPLETE revascularization reduces death and recurrent MI risk
Revascularization of more than just the culprit lesion in patients with ST-elevation myocardial infarctions could significantly reduce their risk of cardiovascular death or myocardial infarction, according to results of the COMPLETE trial, presented at the annual congress of the European Society of Cardiology.
The report, simultaneously published online in the New England Journal of Medicine, detailed the outcomes of COMPLETE (the Complete versus Culprit-Only Revascularization Strategies to Treat Multivessel Disease after Early PCI for STEMI), a study in 4,041 patients who had experienced an ST-elevation myocardial infarction (STEMI), who had multi-vessel coronary artery disease, and who had undergone successful percutaneous coronary intervention of the culprit lesion.
Participants were randomized either to complete revascularization of all angiographically significant nonculprit lesions, or to no further revascularization, and were followed for a median of 3 years.
Of the patients who underwent complete revascularization, 7.8% experienced either cardiovascular death or another myocardial infarction, compared with 10.5% of those who only had revascularization of the culprit lesion, representing a significant 26% reduction (P = .004) in the incidence of this composite coprimary outcome.
The decrease in events was driven by a significant 32% reduction in the incidence of new myocardial infarction – particularly non-STEMI, new STEMI, and myocardial infarction type 1 – in the complete revascularization group, with only a 7% reduction in the incidence of death from cardiovascular causes.
With the second coprimary outcome of a composite of cardiovascular death, myocardial infarction, or ischemia-driven revascularization, this was seen in 8.9% of patients in the complete revascularization group compared with 16.7% of patients with the culprit-lesion-only group; a significant 49% reduction in incidence (P less than .001).
The authors calculated that 37 complete revascularizations would need to be performed to prevent one incidence of cardiovascular death or myocardial infarction. To prevent cardiovascular death, myocardial infarction, or ischemia-driven revascularization, the number needed to treat was 13.
The timing of complete revascularization did not appear to affect the benefits of the procedure, which were consistent among patients who underwent complete revascularization during their index hospitalization and in those who underwent the procedure after hospital discharge. Investigators had to specify before randomization whether the patient would undergo complete revascularization during the index hospitalization or after discharge but within 45 days.
The study also did not find any significant differences between the two groups in the risks of major bleeding, stroke, or stent thrombosis. However, the complete revascularization group did experience a nonsignificant 59% higher odds of contrast-associated acute kidney injury, which was attributed to the nonculprit lesion revascularization in seven patients in the complete revascularization group.
Dr. Shamir R. Mehta, of the Population Health Research Institute at McMaster University, Ontario, and coauthors noted that previous trials of complete-revascularization strategies in patients with STEMI were smaller and had included revascularization as part of a composite primary outcome.
“In the absence of a reduction in irreversible events such as cardiovascular death or new myocardial infarction, the clinical relevance of performing early nonculprit-lesion PCI in all patients with multivessel coronary artery disease to prevent later PCI in a smaller number of those patients is debatable,” they wrote. “We have now found that routine nonculprit-lesion PCI with the goal of complete revascularization confers a reduction in the long-term risk of cardiovascular death or myocardial infarction.”
No patients with cardiogenic shock were enrolled in the study, so the results could not be extrapolated to that patient group.
In an accompanying editorial, Dr. Lars Kober and Dr. Thomas Engstrøm, of the department of cardiology at Rigshospitalet at the University of Copenhagen, wrote that until now, there had been a lack of evidence that complete revascularization could reduce hard outcomes such as death and recurrent myocardial infarction (N Engl J Med. 2019 Sep. 1. doi: 10.1056/NEJMe1910898).
However, they said, given the findings of this study, it might now be appropriate to recommend complete revascularization for patients such as those enrolled in the study.
“Better selection of high-risk patients may also refine the determination of who is most likely to benefit from complete revascularization,” they wrote.
COMPLETE was supported by the Canadian Institutes of Health Research, with additional support from AstraZeneca, Boston Scientific, and the Population Health Research Institute. Two authors declared support from AstraZeneca and Boston Scientific during the conduct of the study, and eight declared personal fees, funding, and grants from the pharmaceutical industry outside the study. One author declared employment with Medtronic, unrelated to the submitted work.
Both editorial authors declared grants and personal fees, including from the study supporters.
SOURCE: Mehta S et al. N Engl J Med. 2019 Sep. 1. doi: 10.1056/NEJMoa1907775.
Revascularization of more than just the culprit lesion in patients with ST-elevation myocardial infarctions could significantly reduce their risk of cardiovascular death or myocardial infarction, according to results of the COMPLETE trial, presented at the annual congress of the European Society of Cardiology.
The report, simultaneously published online in the New England Journal of Medicine, detailed the outcomes of COMPLETE (the Complete versus Culprit-Only Revascularization Strategies to Treat Multivessel Disease after Early PCI for STEMI), a study in 4,041 patients who had experienced an ST-elevation myocardial infarction (STEMI), who had multi-vessel coronary artery disease, and who had undergone successful percutaneous coronary intervention of the culprit lesion.
Participants were randomized either to complete revascularization of all angiographically significant nonculprit lesions, or to no further revascularization, and were followed for a median of 3 years.
Of the patients who underwent complete revascularization, 7.8% experienced either cardiovascular death or another myocardial infarction, compared with 10.5% of those who only had revascularization of the culprit lesion, representing a significant 26% reduction (P = .004) in the incidence of this composite coprimary outcome.
The decrease in events was driven by a significant 32% reduction in the incidence of new myocardial infarction – particularly non-STEMI, new STEMI, and myocardial infarction type 1 – in the complete revascularization group, with only a 7% reduction in the incidence of death from cardiovascular causes.
With the second coprimary outcome of a composite of cardiovascular death, myocardial infarction, or ischemia-driven revascularization, this was seen in 8.9% of patients in the complete revascularization group compared with 16.7% of patients with the culprit-lesion-only group; a significant 49% reduction in incidence (P less than .001).
The authors calculated that 37 complete revascularizations would need to be performed to prevent one incidence of cardiovascular death or myocardial infarction. To prevent cardiovascular death, myocardial infarction, or ischemia-driven revascularization, the number needed to treat was 13.
The timing of complete revascularization did not appear to affect the benefits of the procedure, which were consistent among patients who underwent complete revascularization during their index hospitalization and in those who underwent the procedure after hospital discharge. Investigators had to specify before randomization whether the patient would undergo complete revascularization during the index hospitalization or after discharge but within 45 days.
The study also did not find any significant differences between the two groups in the risks of major bleeding, stroke, or stent thrombosis. However, the complete revascularization group did experience a nonsignificant 59% higher odds of contrast-associated acute kidney injury, which was attributed to the nonculprit lesion revascularization in seven patients in the complete revascularization group.
Dr. Shamir R. Mehta, of the Population Health Research Institute at McMaster University, Ontario, and coauthors noted that previous trials of complete-revascularization strategies in patients with STEMI were smaller and had included revascularization as part of a composite primary outcome.
“In the absence of a reduction in irreversible events such as cardiovascular death or new myocardial infarction, the clinical relevance of performing early nonculprit-lesion PCI in all patients with multivessel coronary artery disease to prevent later PCI in a smaller number of those patients is debatable,” they wrote. “We have now found that routine nonculprit-lesion PCI with the goal of complete revascularization confers a reduction in the long-term risk of cardiovascular death or myocardial infarction.”
No patients with cardiogenic shock were enrolled in the study, so the results could not be extrapolated to that patient group.
In an accompanying editorial, Dr. Lars Kober and Dr. Thomas Engstrøm, of the department of cardiology at Rigshospitalet at the University of Copenhagen, wrote that until now, there had been a lack of evidence that complete revascularization could reduce hard outcomes such as death and recurrent myocardial infarction (N Engl J Med. 2019 Sep. 1. doi: 10.1056/NEJMe1910898).
However, they said, given the findings of this study, it might now be appropriate to recommend complete revascularization for patients such as those enrolled in the study.
“Better selection of high-risk patients may also refine the determination of who is most likely to benefit from complete revascularization,” they wrote.
COMPLETE was supported by the Canadian Institutes of Health Research, with additional support from AstraZeneca, Boston Scientific, and the Population Health Research Institute. Two authors declared support from AstraZeneca and Boston Scientific during the conduct of the study, and eight declared personal fees, funding, and grants from the pharmaceutical industry outside the study. One author declared employment with Medtronic, unrelated to the submitted work.
Both editorial authors declared grants and personal fees, including from the study supporters.
SOURCE: Mehta S et al. N Engl J Med. 2019 Sep. 1. doi: 10.1056/NEJMoa1907775.
Revascularization of more than just the culprit lesion in patients with ST-elevation myocardial infarctions could significantly reduce their risk of cardiovascular death or myocardial infarction, according to results of the COMPLETE trial, presented at the annual congress of the European Society of Cardiology.
The report, simultaneously published online in the New England Journal of Medicine, detailed the outcomes of COMPLETE (the Complete versus Culprit-Only Revascularization Strategies to Treat Multivessel Disease after Early PCI for STEMI), a study in 4,041 patients who had experienced an ST-elevation myocardial infarction (STEMI), who had multi-vessel coronary artery disease, and who had undergone successful percutaneous coronary intervention of the culprit lesion.
Participants were randomized either to complete revascularization of all angiographically significant nonculprit lesions, or to no further revascularization, and were followed for a median of 3 years.
Of the patients who underwent complete revascularization, 7.8% experienced either cardiovascular death or another myocardial infarction, compared with 10.5% of those who only had revascularization of the culprit lesion, representing a significant 26% reduction (P = .004) in the incidence of this composite coprimary outcome.
The decrease in events was driven by a significant 32% reduction in the incidence of new myocardial infarction – particularly non-STEMI, new STEMI, and myocardial infarction type 1 – in the complete revascularization group, with only a 7% reduction in the incidence of death from cardiovascular causes.
With the second coprimary outcome of a composite of cardiovascular death, myocardial infarction, or ischemia-driven revascularization, this was seen in 8.9% of patients in the complete revascularization group compared with 16.7% of patients with the culprit-lesion-only group; a significant 49% reduction in incidence (P less than .001).
The authors calculated that 37 complete revascularizations would need to be performed to prevent one incidence of cardiovascular death or myocardial infarction. To prevent cardiovascular death, myocardial infarction, or ischemia-driven revascularization, the number needed to treat was 13.
The timing of complete revascularization did not appear to affect the benefits of the procedure, which were consistent among patients who underwent complete revascularization during their index hospitalization and in those who underwent the procedure after hospital discharge. Investigators had to specify before randomization whether the patient would undergo complete revascularization during the index hospitalization or after discharge but within 45 days.
The study also did not find any significant differences between the two groups in the risks of major bleeding, stroke, or stent thrombosis. However, the complete revascularization group did experience a nonsignificant 59% higher odds of contrast-associated acute kidney injury, which was attributed to the nonculprit lesion revascularization in seven patients in the complete revascularization group.
Dr. Shamir R. Mehta, of the Population Health Research Institute at McMaster University, Ontario, and coauthors noted that previous trials of complete-revascularization strategies in patients with STEMI were smaller and had included revascularization as part of a composite primary outcome.
“In the absence of a reduction in irreversible events such as cardiovascular death or new myocardial infarction, the clinical relevance of performing early nonculprit-lesion PCI in all patients with multivessel coronary artery disease to prevent later PCI in a smaller number of those patients is debatable,” they wrote. “We have now found that routine nonculprit-lesion PCI with the goal of complete revascularization confers a reduction in the long-term risk of cardiovascular death or myocardial infarction.”
No patients with cardiogenic shock were enrolled in the study, so the results could not be extrapolated to that patient group.
In an accompanying editorial, Dr. Lars Kober and Dr. Thomas Engstrøm, of the department of cardiology at Rigshospitalet at the University of Copenhagen, wrote that until now, there had been a lack of evidence that complete revascularization could reduce hard outcomes such as death and recurrent myocardial infarction (N Engl J Med. 2019 Sep. 1. doi: 10.1056/NEJMe1910898).
However, they said, given the findings of this study, it might now be appropriate to recommend complete revascularization for patients such as those enrolled in the study.
“Better selection of high-risk patients may also refine the determination of who is most likely to benefit from complete revascularization,” they wrote.
COMPLETE was supported by the Canadian Institutes of Health Research, with additional support from AstraZeneca, Boston Scientific, and the Population Health Research Institute. Two authors declared support from AstraZeneca and Boston Scientific during the conduct of the study, and eight declared personal fees, funding, and grants from the pharmaceutical industry outside the study. One author declared employment with Medtronic, unrelated to the submitted work.
Both editorial authors declared grants and personal fees, including from the study supporters.
SOURCE: Mehta S et al. N Engl J Med. 2019 Sep. 1. doi: 10.1056/NEJMoa1907775.
FROM THE ESC CONGRESS 2019
Key clinical point: Complete revascularization after STEMI reduces death and recurrent MI risk.
Major finding: Thirty-seven complete revascularizations would need to be performed to prevent one incidence of cardiovascular death or myocardial infarction.
Study details: COMPLETE, a randomized controlled trial in 4,041 patients with STEMI.
Disclosures: The study was supported by the Canadian Institutes of Health Research, with additional support from AstraZeneca, Boston Scientific, and the Population Health Research Institute. Two authors declared support from AstraZeneca and Boston Scientific during the conduct of the study, and eight declared personal fees, funding, and grants from the pharmaceutical industry outside the study. One author declared employment with Medtronic, unrelated to the submitted work.
Source: Mehta S et al. N Engl J Med. 2019 Sep 1. doi: 10.1056/NEJMoa1907775.
Reunion
We were catching up during our 35th college reunion at our old fraternity house overlooking Cayuga Lake in Ithaca, N.Y. About 50 of us lived in the Tudor-style house, complete with secret basement room, and there was a ladder that allowed access to the relatively flat, painted aluminum roof. When the weather allowed, we climbed the ladder to sun ourselves on top of the house. We also flung water balloons at unsuspecting pedestrians with a sling shot device made by attaching rubber tubing to a funnel. The “funnelator” was very accurate to about 50 yards away. We were kids, and climbing that ladder meant fun, and we climbed it as often as we could.
Despite what many would have predicted when we graduated, my fraternity brothers became a very successful group of CEOs, vice presidents, doctors, lawyers, chairmen, and consultants. Our house was just off Cornell University’s campus at the top of Ithaca Falls, an idyllic setting on a beautiful June evening for my brothers to sit around, laugh about the old times, and philosophize about life. We recounted our life after college and reveled in each others’ accomplishments.
After climbing the roof ladder for fun, we had each climbed a different kind of ladder to success in our respective fields. We all really enjoyed the climb. I don’t think it is a coincidence that many of my brothers and I are now done climbing our ladders. Many of us are getting out of the rat race.
One of my friends is resigning as chairman of an academic ENT department. I remember his discipline in college, leaving the house after dinner every night to climb the hill where he studied in the quiet of Uris Library, which is attached to the iconic McGraw Tower. His hard work paid off with an acceptance to a prestigious medical school where he continued to excel. The author of more than 200 published manuscripts, with four senior-authored papers already this year, he is at the pinnacle of his academic success. Yet, he resigned.
Similarly, another of my fraternity brothers had recently resigned from his position as Senior Vice President and Chief Medical Officer for a large health care system. He would have been in line for the CEO position had he stayed. He has written well-received books on leadership and financial acumen for physicians. As a result, he is a frequent public speaker on similar topics. Yet, he resigned.
They were not the only ones resigning positions that others covet. I, too, resigned my position as Department Chairman earlier this year. None of us were fired, none of us were asked to leave, and none of us are burned out. So here we were, three accomplished physicians all resigning from powerful posts at the same time for what turns out to be similar reasons. Our priorities changed as our children moved out.
I would like to say that we all had the wisdom to know that our leadership skills were deteriorating and that we all wanted to get out while we are at the top of our game. Had Arthur Brooks written “Your Professional Decline Is Coming (Much) Sooner Than You Think” in The Atlantic (July 2019) before we made our decisions, I may have made that argument, but it would not have been true. All three of us feel like we have accomplished what we sought to achieve when we took our respective roles and now we wanted to leverage that experience into something different, if not better. None of us have settled into new roles yet, and all of us are still trying to define exactly what it is we want to do next, but all of us agree that we are no longer interested in driving ourselves to succeed at the expense of our family, friends, and relationships.
My fraternity brothers and I gushed with pride talking about our children and their success. Our progeny are starting their individual climbs up the ladder of opportunity in whatever field they have chosen. My friends and I, on the other hand, had already climbed a ladder and feel comfortable stopping. Or maybe we just want to start climbing a different ladder.
Dr. Kalaycio is editor in chief of Hematology News. He chairs the department of hematology and medical oncology at Cleveland Clinic Taussig Cancer Institute. Contact him at kalaycm@ccf.org.
We were catching up during our 35th college reunion at our old fraternity house overlooking Cayuga Lake in Ithaca, N.Y. About 50 of us lived in the Tudor-style house, complete with secret basement room, and there was a ladder that allowed access to the relatively flat, painted aluminum roof. When the weather allowed, we climbed the ladder to sun ourselves on top of the house. We also flung water balloons at unsuspecting pedestrians with a sling shot device made by attaching rubber tubing to a funnel. The “funnelator” was very accurate to about 50 yards away. We were kids, and climbing that ladder meant fun, and we climbed it as often as we could.
Despite what many would have predicted when we graduated, my fraternity brothers became a very successful group of CEOs, vice presidents, doctors, lawyers, chairmen, and consultants. Our house was just off Cornell University’s campus at the top of Ithaca Falls, an idyllic setting on a beautiful June evening for my brothers to sit around, laugh about the old times, and philosophize about life. We recounted our life after college and reveled in each others’ accomplishments.
After climbing the roof ladder for fun, we had each climbed a different kind of ladder to success in our respective fields. We all really enjoyed the climb. I don’t think it is a coincidence that many of my brothers and I are now done climbing our ladders. Many of us are getting out of the rat race.
One of my friends is resigning as chairman of an academic ENT department. I remember his discipline in college, leaving the house after dinner every night to climb the hill where he studied in the quiet of Uris Library, which is attached to the iconic McGraw Tower. His hard work paid off with an acceptance to a prestigious medical school where he continued to excel. The author of more than 200 published manuscripts, with four senior-authored papers already this year, he is at the pinnacle of his academic success. Yet, he resigned.
Similarly, another of my fraternity brothers had recently resigned from his position as Senior Vice President and Chief Medical Officer for a large health care system. He would have been in line for the CEO position had he stayed. He has written well-received books on leadership and financial acumen for physicians. As a result, he is a frequent public speaker on similar topics. Yet, he resigned.
They were not the only ones resigning positions that others covet. I, too, resigned my position as Department Chairman earlier this year. None of us were fired, none of us were asked to leave, and none of us are burned out. So here we were, three accomplished physicians all resigning from powerful posts at the same time for what turns out to be similar reasons. Our priorities changed as our children moved out.
I would like to say that we all had the wisdom to know that our leadership skills were deteriorating and that we all wanted to get out while we are at the top of our game. Had Arthur Brooks written “Your Professional Decline Is Coming (Much) Sooner Than You Think” in The Atlantic (July 2019) before we made our decisions, I may have made that argument, but it would not have been true. All three of us feel like we have accomplished what we sought to achieve when we took our respective roles and now we wanted to leverage that experience into something different, if not better. None of us have settled into new roles yet, and all of us are still trying to define exactly what it is we want to do next, but all of us agree that we are no longer interested in driving ourselves to succeed at the expense of our family, friends, and relationships.
My fraternity brothers and I gushed with pride talking about our children and their success. Our progeny are starting their individual climbs up the ladder of opportunity in whatever field they have chosen. My friends and I, on the other hand, had already climbed a ladder and feel comfortable stopping. Or maybe we just want to start climbing a different ladder.
Dr. Kalaycio is editor in chief of Hematology News. He chairs the department of hematology and medical oncology at Cleveland Clinic Taussig Cancer Institute. Contact him at kalaycm@ccf.org.
We were catching up during our 35th college reunion at our old fraternity house overlooking Cayuga Lake in Ithaca, N.Y. About 50 of us lived in the Tudor-style house, complete with secret basement room, and there was a ladder that allowed access to the relatively flat, painted aluminum roof. When the weather allowed, we climbed the ladder to sun ourselves on top of the house. We also flung water balloons at unsuspecting pedestrians with a sling shot device made by attaching rubber tubing to a funnel. The “funnelator” was very accurate to about 50 yards away. We were kids, and climbing that ladder meant fun, and we climbed it as often as we could.
Despite what many would have predicted when we graduated, my fraternity brothers became a very successful group of CEOs, vice presidents, doctors, lawyers, chairmen, and consultants. Our house was just off Cornell University’s campus at the top of Ithaca Falls, an idyllic setting on a beautiful June evening for my brothers to sit around, laugh about the old times, and philosophize about life. We recounted our life after college and reveled in each others’ accomplishments.
After climbing the roof ladder for fun, we had each climbed a different kind of ladder to success in our respective fields. We all really enjoyed the climb. I don’t think it is a coincidence that many of my brothers and I are now done climbing our ladders. Many of us are getting out of the rat race.
One of my friends is resigning as chairman of an academic ENT department. I remember his discipline in college, leaving the house after dinner every night to climb the hill where he studied in the quiet of Uris Library, which is attached to the iconic McGraw Tower. His hard work paid off with an acceptance to a prestigious medical school where he continued to excel. The author of more than 200 published manuscripts, with four senior-authored papers already this year, he is at the pinnacle of his academic success. Yet, he resigned.
Similarly, another of my fraternity brothers had recently resigned from his position as Senior Vice President and Chief Medical Officer for a large health care system. He would have been in line for the CEO position had he stayed. He has written well-received books on leadership and financial acumen for physicians. As a result, he is a frequent public speaker on similar topics. Yet, he resigned.
They were not the only ones resigning positions that others covet. I, too, resigned my position as Department Chairman earlier this year. None of us were fired, none of us were asked to leave, and none of us are burned out. So here we were, three accomplished physicians all resigning from powerful posts at the same time for what turns out to be similar reasons. Our priorities changed as our children moved out.
I would like to say that we all had the wisdom to know that our leadership skills were deteriorating and that we all wanted to get out while we are at the top of our game. Had Arthur Brooks written “Your Professional Decline Is Coming (Much) Sooner Than You Think” in The Atlantic (July 2019) before we made our decisions, I may have made that argument, but it would not have been true. All three of us feel like we have accomplished what we sought to achieve when we took our respective roles and now we wanted to leverage that experience into something different, if not better. None of us have settled into new roles yet, and all of us are still trying to define exactly what it is we want to do next, but all of us agree that we are no longer interested in driving ourselves to succeed at the expense of our family, friends, and relationships.
My fraternity brothers and I gushed with pride talking about our children and their success. Our progeny are starting their individual climbs up the ladder of opportunity in whatever field they have chosen. My friends and I, on the other hand, had already climbed a ladder and feel comfortable stopping. Or maybe we just want to start climbing a different ladder.
Dr. Kalaycio is editor in chief of Hematology News. He chairs the department of hematology and medical oncology at Cleveland Clinic Taussig Cancer Institute. Contact him at kalaycm@ccf.org.
Major Depressive Disorder: Unmet Needs and Innovative Treatments
Many of the unmet needs in major depressive disorder (MDD) are modifiable, including improving diagnostic accuracy and offering treatments with faster onset of action, treatments with greater overall efficacy, and treatments that can improve patient functioning.
Click here to read the supplement and earn 1 AMA Category 1 CreditTM by learning about these unmet needs, and innovative strageies working to address them.
Topics include:
- Targeting Unmet Needs in the Treatment of Major Depressive Disorder
- Innovative Strategies for Treatments of Major Depressive Disorder: A Brief Review of Recent Developments
EDUCATIONAL OBJECTIVES
- After completing this activity, the participant should be better able to:
- Treat major depression within 2 weeks.
- Use evidence based treatments to achieve remission in major depression.
- Discuss novel targets including glutamate for treating major depression.
- Utilize treatments with innovative mechanisms to treat major depression.
Click here to read the supplement.
Many of the unmet needs in major depressive disorder (MDD) are modifiable, including improving diagnostic accuracy and offering treatments with faster onset of action, treatments with greater overall efficacy, and treatments that can improve patient functioning.
Click here to read the supplement and earn 1 AMA Category 1 CreditTM by learning about these unmet needs, and innovative strageies working to address them.
Topics include:
- Targeting Unmet Needs in the Treatment of Major Depressive Disorder
- Innovative Strategies for Treatments of Major Depressive Disorder: A Brief Review of Recent Developments
EDUCATIONAL OBJECTIVES
- After completing this activity, the participant should be better able to:
- Treat major depression within 2 weeks.
- Use evidence based treatments to achieve remission in major depression.
- Discuss novel targets including glutamate for treating major depression.
- Utilize treatments with innovative mechanisms to treat major depression.
Click here to read the supplement.
Many of the unmet needs in major depressive disorder (MDD) are modifiable, including improving diagnostic accuracy and offering treatments with faster onset of action, treatments with greater overall efficacy, and treatments that can improve patient functioning.
Click here to read the supplement and earn 1 AMA Category 1 CreditTM by learning about these unmet needs, and innovative strageies working to address them.
Topics include:
- Targeting Unmet Needs in the Treatment of Major Depressive Disorder
- Innovative Strategies for Treatments of Major Depressive Disorder: A Brief Review of Recent Developments
EDUCATIONAL OBJECTIVES
- After completing this activity, the participant should be better able to:
- Treat major depression within 2 weeks.
- Use evidence based treatments to achieve remission in major depression.
- Discuss novel targets including glutamate for treating major depression.
- Utilize treatments with innovative mechanisms to treat major depression.
Click here to read the supplement.
The month of new beginnings is here
This month’s Letter from the Editor is guest authored by Dr. Megan A. Adams, GI & Hepatology News Associate Editor
September is a month of new beginnings, as summer transitions to fall, kids go back to school, and we return to more consistent work routines, refreshed and reinvigorated after some well-deserved time off with family and friends. Among our cover stories this month is a study showing a novel application of deep learning to inform clinical care of patients with pancreatic cysts. We also feature several high-impact studies from AGA’s journals, including a large randomized controlled trial by Dr. Paul Moayyedi and colleagues, demonstrating that PPI therapy may be unnecessary in the majority of patients on oral anticoagulants, despite current guideline recommendations. This study has the potential to substantially change clinical practice, particularly in the context of the current discussion regarding PPI benefits and harms, and our transition to value-based care.
We also highlight a proof-of-concept study demonstrating a potential role for probiotics (specifically Bifidobacteria) in reducing the risk of NSAID-related gastrointestinal bleeding, and another study showing a possible role for clopidogrel in chemoprevention of colorectal cancer. Both articles are accompanied by expert commentaries highlighting their potential effect on clinical practice.
Our September issue also emphasizes the importance of professional advocacy by chronicling the participation of four AGA leaders (Dr. Carr, Dr. Kaufman, Dr. Ketwaroo, and Dr. Mathews) in the 2019 Alliance of Specialty Medicine Fly In, a multisociety effort to lobby legislators on key issues such as reducing prior authorization burdens and minimizing the strict constraints of step-therapy protocols. We also are pleased to acknowledge the future leaders of gastroenterology by recognizing the 17 exceptional fellows who demonstrated their passion for advancing GI clinical care by presenting their institutional quality improvement projects at a special session at DDW® 2019. We hope you find these stories to be thought provoking, inspiring, and directly relevant to your clinical practice – thank you for reading!
Megan A. Adams, MD, JD, MSc
Associate Editor
This month’s Letter from the Editor is guest authored by Dr. Megan A. Adams, GI & Hepatology News Associate Editor
September is a month of new beginnings, as summer transitions to fall, kids go back to school, and we return to more consistent work routines, refreshed and reinvigorated after some well-deserved time off with family and friends. Among our cover stories this month is a study showing a novel application of deep learning to inform clinical care of patients with pancreatic cysts. We also feature several high-impact studies from AGA’s journals, including a large randomized controlled trial by Dr. Paul Moayyedi and colleagues, demonstrating that PPI therapy may be unnecessary in the majority of patients on oral anticoagulants, despite current guideline recommendations. This study has the potential to substantially change clinical practice, particularly in the context of the current discussion regarding PPI benefits and harms, and our transition to value-based care.
We also highlight a proof-of-concept study demonstrating a potential role for probiotics (specifically Bifidobacteria) in reducing the risk of NSAID-related gastrointestinal bleeding, and another study showing a possible role for clopidogrel in chemoprevention of colorectal cancer. Both articles are accompanied by expert commentaries highlighting their potential effect on clinical practice.
Our September issue also emphasizes the importance of professional advocacy by chronicling the participation of four AGA leaders (Dr. Carr, Dr. Kaufman, Dr. Ketwaroo, and Dr. Mathews) in the 2019 Alliance of Specialty Medicine Fly In, a multisociety effort to lobby legislators on key issues such as reducing prior authorization burdens and minimizing the strict constraints of step-therapy protocols. We also are pleased to acknowledge the future leaders of gastroenterology by recognizing the 17 exceptional fellows who demonstrated their passion for advancing GI clinical care by presenting their institutional quality improvement projects at a special session at DDW® 2019. We hope you find these stories to be thought provoking, inspiring, and directly relevant to your clinical practice – thank you for reading!
Megan A. Adams, MD, JD, MSc
Associate Editor
This month’s Letter from the Editor is guest authored by Dr. Megan A. Adams, GI & Hepatology News Associate Editor
September is a month of new beginnings, as summer transitions to fall, kids go back to school, and we return to more consistent work routines, refreshed and reinvigorated after some well-deserved time off with family and friends. Among our cover stories this month is a study showing a novel application of deep learning to inform clinical care of patients with pancreatic cysts. We also feature several high-impact studies from AGA’s journals, including a large randomized controlled trial by Dr. Paul Moayyedi and colleagues, demonstrating that PPI therapy may be unnecessary in the majority of patients on oral anticoagulants, despite current guideline recommendations. This study has the potential to substantially change clinical practice, particularly in the context of the current discussion regarding PPI benefits and harms, and our transition to value-based care.
We also highlight a proof-of-concept study demonstrating a potential role for probiotics (specifically Bifidobacteria) in reducing the risk of NSAID-related gastrointestinal bleeding, and another study showing a possible role for clopidogrel in chemoprevention of colorectal cancer. Both articles are accompanied by expert commentaries highlighting their potential effect on clinical practice.
Our September issue also emphasizes the importance of professional advocacy by chronicling the participation of four AGA leaders (Dr. Carr, Dr. Kaufman, Dr. Ketwaroo, and Dr. Mathews) in the 2019 Alliance of Specialty Medicine Fly In, a multisociety effort to lobby legislators on key issues such as reducing prior authorization burdens and minimizing the strict constraints of step-therapy protocols. We also are pleased to acknowledge the future leaders of gastroenterology by recognizing the 17 exceptional fellows who demonstrated their passion for advancing GI clinical care by presenting their institutional quality improvement projects at a special session at DDW® 2019. We hope you find these stories to be thought provoking, inspiring, and directly relevant to your clinical practice – thank you for reading!
Megan A. Adams, MD, JD, MSc
Associate Editor
Brexanolone injection for postpartum depression
Postpartum depression (PPD) is one of the most prevalent complications associated with pregnancy and childbirth in the United States, affecting more than 400,000 women annually.1 Postpartum depression is most commonly treated with psychotherapy and antidepressants approved for the treatment of major depressive disorder. Until recently, there was no pharmacologic therapy approved by the FDA specifically for the treatment of PPD. Considering the adverse outcomes associated with untreated or inadequately treated PPD, and the limitations of existing therapies, there is a significant unmet need for pharmacologic treatment options for PPD.2 To help address this need, the FDA recently approved brexanolone injection (brand name: ZULRESSO™) (Table 13) as a first-in-class therapy for the treatment of adults with PPD.3
Clinical implications
Postpartum depression can result in adverse outcomes for the patient, baby, and family when under- or untreated, and the need for rapid resolution of symptoms cannot be overstated.2 Suicide is strongly associated with depression and is a leading cause of pregnancy-related deaths.4 Additionally, PPD can impact the health, safety, and well-being of the child, with both short- and long-term consequences, including greater rates of psychological or behavioral difficulties among children of patients with PPD.5 Postpartum depression can also have negative effects on the patient’s partner, with 24% to 50% of partners experiencing depression.6 Current PPD management strategies include the use of psychotherapy and pharmacologic interventions for major depressive disorder that may take up to 4 to 6 weeks for some patients, and may not achieve remission for all patients.7-9
Brexanolone injection is a first-in-class medication with a novel mechanism of action. In clinical studies, it achieved rapid (by Hour 60) and sustained (through Day 30) reductions in depressive symptoms and could provide a meaningful new treatment option for adult women with PPD.10,11
How it works
Animal and human studies have established the re
Brexanolone is a neuroactive steroid that is chemically identical to endogenous allopregnanolone produced in the CNS. Brexanolone potentiates GABA-mediated currents from recombinant human GABAARs in mammalian cells expressing α1β2γ2 receptor subunits, α4β3δ receptor subunits, and α6β3δ receptor subunits.3 Positive allosteric modulation of both synaptic and extrasynaptic GABAARs differentiates brexanolone from other GABAAR modulators, such as benzodiazepines.10,11
Brexanolone’s mechanism of action in the treatment of PPD is not fully understood, but it is thought to be related to GABAAR PAM activity.3
Supporting evidence
The FDA approval of brexanolone injection was based on the efficacy demonstrated in 2 Phase III multicenter, randomized, double-blind, placebo-controlled studies in adult women (age 18 to 45) with PPD (defined by DSM-IV criteria for a major depressive episode, with onset of symptoms in the third trimester or within 4 weeks of delivery). Exclusion criteria included the presence of bipolar disorder or psychosis. In these studies, 60-hour continuous IV infusions of brexanolone or placebo were given, followed by 4 weeks of observation. Study 1 (202B) enrolled patients with severe PPD (Hamilton Rating Scale for Depression [HAM-D] total score ≥26), and Study 2 (202C) enrolled patients with moderate PPD (HAM-D score 20 to 25). A titration to the recommended target dosage of 90 μg/kg/hour was evaluated in both studies. BRX90 patients received 30 μg/kg/hour for 4 hours, 60 μg/kg/hour for 20 hours, 90 μg/kg/hour for 28 hours, followed by a taper to 60 μg/kg/hour for 4 hours and then 30 μg/kg/hour for 4 hours. The primary endpoint in both studies was the mean change from baseline in depressive symptoms as measured by HAM-D total score at the end of the 60-hour infusion. A pre-specified secondary efficacy endpoint was the mean change from baseline in HAM-D total score at Day 30.
Continue to: Efficacy
Efficacy. In both placebo-controlled studies, titration to a target dose of brexanolone 90 μg/kg/hour was superior to placebo in improvement of depressive symptoms (Table 33).
Pharmacological profile
Brexanolone exposure-response relationships and the time course of pharmacodynamic response are unknown.3
Adverse reactions. Safety was evaluated from all patients receiving brexanolone injection, regardless of dosing regimen (N = 140, including patients from a Phase IIb study, 202A).3,11
The most common adverse reactions (incidence ≥5% and at least twice the rate of placebo) were sedation/somnolence, dry mouth, loss of consciousness, and flushing/hot flush.3 The incidence of patients discontinuing due to any adverse reaction was 2% for brexanolone vs 1% for placebo.3
Sedation, somnolence, and loss of consciousness. In clinical studies, brexanolone caused sedation and somnolence that required dose interruption or reduction in some patients during the infusion (5% of brexanolone-treated patients compared with 0% of placebo-treated patients).3 Some patients were also reported to have loss of consciousness or altered state of consciousness during the brexanolone infusion (4% of patients treated with brexanolone compared with 0% of patients treated with placebo).3 All patients with loss of or altered state of consciousness recovered fully 15 to 60 minutes after dose interruption.3 There was no clear association between loss or alteration of consciousness and pattern or timing of dose, and not all patients who experienced a loss or alteration of consciousness reported sedation or somnolence before the episode.
Continue to: Suicidality
Suicidality. The risk of developing suicidal thoughts and behaviors with brexanolone is unknown, due to the relatively low number of exposures to brexanolone injection during clinical development and a mechanism of action distinct from that of existing antidepressant medications.3
Pharmacokinetics
In clinical trials, brexanolone exhibited dose-proportional pharmacokinetics, and the terminal half-life is approximately 9 hours (Table 43). Brexanolone is metabolized by non-cytochrome P450 (CYP)-based pathways, including keto-reduction, glucuronidation, and sulfation.3 No clinically significant differences in the pharmacokinetics of brexanolone were observed based on renal or hepatic impairment, and no studies were conducted to evaluate the effects of other drugs on brexanolone.3
Lactation. A population pharmacokinetics model constructed from studies in the clinical development program calculated the maximum relative infant dose for brexanolone during infusion as 1.3%.3 Given the low oral bioavailability of brexanolone (<5%) in adults, the potential for breastfed infant exposure is considered low.3
Clinical considerations
Risk Evaluation and Mitigation Strategies (REMS) requirements. Brexanolone injection is a Schedule IV controlled substance. It has a “black-box” warning regarding excessive sedation and sudden loss of consciousness, which has been taken into account within the REMS drug safety program. Health care facilities and pharmacies must enroll in the REMS program and ensure that brexanolone is administered only to patients who are enrolled in the REMS program. Staff must be trained on the processes and procedures to administer brexanolone, and the facility must have a fall precautions protocol in place and be equipped with a programmable peristaltic IV infusion pump and continuous pulse oximetry with alarms.3
Monitoring. A REMS-trained clinician must be available continuously on-site to oversee each patient for the duration of the continuous IV infusion, which lasts 60 hours (2.5 days) and should be initiated early enough in the day to allow for recognition of excessive sedation. Patients must be monitored for hypoxia using continuous pulse oximetry equipped with an alarm and should also be assessed for excessive sedation every 2 hours during planned, non-sleep periods. If excessive sedation occurs, the infusion should be stopped until symptoms resolve, after which the infusion may be resumed at the same or a lower dose as clinically appropriate. In case of overdosage, the infusion should be stopped immediately and supportive measures initiated as necessary. Patients must not be the primary caregiver of dependents, and must be accompanied during interactions with their child(ren).
Continue to: Contraindications
Contraindications. There are no contraindications for the use of brexanolone in adults with PPD.
End-stage renal disease (ESRD). Avoid using brexanolone in patients with ESRD because of the potential accumulation of the solubilizing agent, betadex sulfobutyl ether sodium.
Pregnancy. Brexanolone has not been studied in pregnant patients. Pregnant women and women of reproductive age should be informed of the potential risk to a fetus based on data from other drugs that enhance GABAergic inhibition.
Breastfeeding. There are no data on the effects of brexanolone on a breastfed infant. Breastfeeding should be a discussion of risk and benefit between the patient and her doctor. The developmental and health benefits of breastfeeding should be considered, along with the mother’s clinical need for brexanolone and any potential adverse effects on the breastfed child from brexanolone or from the underlying maternal condition. However, based on the low relative infant dose (<2%) and the low oral bioavailability in adults, the risk to breastfed infants is thought to be low.16
Potential for abuse. Brexanolone injection is a Schedule IV controlled substance. Although it was not possible to assess physical dependency in the registrational trials due to dose tapering at the end of treatment, clinicians should advise patients about the theoretical possibility for brexanolone to be abused or lead to dependence based on other medications with similar primary pharmacology.
Continue to: Concomitant medications
Concomitant medications. Caution patients that taking opioids or other CNS depressants, such as benzodiazepines, in combination with brexanolone may increase the severity of sedative effects.
Suicidal thoughts and behaviors. Advise patients and caregivers to look for the emergence of suicidal thoughts and behavior and instruct them to report such symptoms to their clinician. Consider changing the therapeutic regimen, including discontinuing brexanolone, in patients whose depression becomes worse or who experience emergent suicidal thoughts and behaviors.
Why Rx?
Postpartum depression is a common and often devastating medical complication of childbirth that can result in adverse outcomes for the patient, baby, and family when left undertreated or untreated. There is a great need to identify and treat women who develop PPD. Rapid and sustained resolution of symptoms in women who experience PPD should be the goal of treatment, and consequently, brexanolone injection presents an important new tool in available treatment options for PPD.
Bottom Line
Brexanolone injection is a neuroactive steroid gamma-aminobutyric acid (GABA) A receptor positive allosteric modulator that’s been FDA-approved for the treatment of postpartum depression (PPD). It is administered as a continuous IV infusion over 60 hours. The rapid and sustained improvement of PPD observed in clinical trials with brexanolone injection may support a new treatment paradigm for women with PPD.
1. Ko JY, Rockhill KM, Tong VT, et al. Trends in postpartum depressive symptoms - 27 states, 2004, 2008, and 2012. MMWR Morb Mortal Wkly Rep. 2017;66(6):153-158.
2. Frieder A, Fersh M, Hainline R, et al. Pharmacotherapy of postpartum depression: current approaches and novel drug development. CNS Drugs. 2019;33(3):265-282.
3. Brexanolone injection [package insert]. Cambridge, MA: Sage Therapeutics, Inc.; 2019.
4. Bodnar-Deren S, Klipstein K, Fersh M, et al. Suicidal ideation during the postpartum period. J Womens Health (Larchmt). 2016;25(12):1219-1224.
5. Netsi E, Pearson RM, Murray L, et al. Association of persistent and severe postnatal depression with child outcomes. JAMA Psychiatry. 2018;75(3):247-253.
6. Goodman JH. Paternal postpartum depression, its relationship to maternal postpartum depression, and implications for family health. J Adv Nurs. 2004;45(1):26-35.
7. Gelenberg AJ, Freeman MP, Markowitz JC, et al; American Psychiatric Association Work Group on Major Depressive Disorder. Practice guidelines for the treatment of patients with major depressive disorder. 3rd ed. Washington, DC: American Psychiatric Association; 2010.
8. Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163(11):1905-1917.
9. Molyneaux E, Telesia LA, Henshaw C, et al. Antidepressants for preventing postnatal depression. Cochrane Database Syst Rev. 2018;4:CD004363.
10. Kanes S, Colquhoun H, Gunduz-Bruce H, et al. Brexanolone (SAGE-547 injection) in post-partum depression: a randomised controlled trial. Lancet. 2017;390(10093):480-489.
11. Meltzer-Brody S, Colquhoun H, Riesenberg R, et al. Brexanolone injection in post-partum depression: two multicentre, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet. 2018;392(10152):1058-1070.
12. Melon LC, Hooper A, Yang X, et al. Inability to suppress the stress-induced activation of the HPA axis during the peripartum period engenders deficits in postpartum behaviors in mice. Psychoneuroendocrinology. 2018;90:182-193.
13. Deligiannidis KM, Fales CL, Kroll-Desrosiers AR, et al. Resting-state functional connectivity, cortical GABA, and neuroactive steroids in peripartum and peripartum depressed women: a functional magnetic resonance imaging and spectroscopy study. Neuropsychopharmacology. 2019;44(3):546-554.
14. Licheri V, Talani G, Gorule AA, et al. Plasticity of GABAA receptors during pregnancy and postpartum period: from gene to function. Neural Plast. 2015;2015:170435. doi: 10.1155/2015/170435.
15. Luisi S, Petraglia F, Benedetto C, et al. Serum allopregnanolone levels in pregnant women: changes during pregnancy, at delivery, and in hypertensive patients. J Clin Endocrinol Metab. 2000;85(7):2429-2433.
16. Hoffmann E, Wald J, Dray D, et al. Brexanolone injection administration to lactating women: breast milk allopregnanolone levels [30J]. Obstetrics & Gynecology. 2019;133:115S.
Postpartum depression (PPD) is one of the most prevalent complications associated with pregnancy and childbirth in the United States, affecting more than 400,000 women annually.1 Postpartum depression is most commonly treated with psychotherapy and antidepressants approved for the treatment of major depressive disorder. Until recently, there was no pharmacologic therapy approved by the FDA specifically for the treatment of PPD. Considering the adverse outcomes associated with untreated or inadequately treated PPD, and the limitations of existing therapies, there is a significant unmet need for pharmacologic treatment options for PPD.2 To help address this need, the FDA recently approved brexanolone injection (brand name: ZULRESSO™) (Table 13) as a first-in-class therapy for the treatment of adults with PPD.3
Clinical implications
Postpartum depression can result in adverse outcomes for the patient, baby, and family when under- or untreated, and the need for rapid resolution of symptoms cannot be overstated.2 Suicide is strongly associated with depression and is a leading cause of pregnancy-related deaths.4 Additionally, PPD can impact the health, safety, and well-being of the child, with both short- and long-term consequences, including greater rates of psychological or behavioral difficulties among children of patients with PPD.5 Postpartum depression can also have negative effects on the patient’s partner, with 24% to 50% of partners experiencing depression.6 Current PPD management strategies include the use of psychotherapy and pharmacologic interventions for major depressive disorder that may take up to 4 to 6 weeks for some patients, and may not achieve remission for all patients.7-9
Brexanolone injection is a first-in-class medication with a novel mechanism of action. In clinical studies, it achieved rapid (by Hour 60) and sustained (through Day 30) reductions in depressive symptoms and could provide a meaningful new treatment option for adult women with PPD.10,11
How it works
Animal and human studies have established the re
Brexanolone is a neuroactive steroid that is chemically identical to endogenous allopregnanolone produced in the CNS. Brexanolone potentiates GABA-mediated currents from recombinant human GABAARs in mammalian cells expressing α1β2γ2 receptor subunits, α4β3δ receptor subunits, and α6β3δ receptor subunits.3 Positive allosteric modulation of both synaptic and extrasynaptic GABAARs differentiates brexanolone from other GABAAR modulators, such as benzodiazepines.10,11
Brexanolone’s mechanism of action in the treatment of PPD is not fully understood, but it is thought to be related to GABAAR PAM activity.3
Supporting evidence
The FDA approval of brexanolone injection was based on the efficacy demonstrated in 2 Phase III multicenter, randomized, double-blind, placebo-controlled studies in adult women (age 18 to 45) with PPD (defined by DSM-IV criteria for a major depressive episode, with onset of symptoms in the third trimester or within 4 weeks of delivery). Exclusion criteria included the presence of bipolar disorder or psychosis. In these studies, 60-hour continuous IV infusions of brexanolone or placebo were given, followed by 4 weeks of observation. Study 1 (202B) enrolled patients with severe PPD (Hamilton Rating Scale for Depression [HAM-D] total score ≥26), and Study 2 (202C) enrolled patients with moderate PPD (HAM-D score 20 to 25). A titration to the recommended target dosage of 90 μg/kg/hour was evaluated in both studies. BRX90 patients received 30 μg/kg/hour for 4 hours, 60 μg/kg/hour for 20 hours, 90 μg/kg/hour for 28 hours, followed by a taper to 60 μg/kg/hour for 4 hours and then 30 μg/kg/hour for 4 hours. The primary endpoint in both studies was the mean change from baseline in depressive symptoms as measured by HAM-D total score at the end of the 60-hour infusion. A pre-specified secondary efficacy endpoint was the mean change from baseline in HAM-D total score at Day 30.
Continue to: Efficacy
Efficacy. In both placebo-controlled studies, titration to a target dose of brexanolone 90 μg/kg/hour was superior to placebo in improvement of depressive symptoms (Table 33).
Pharmacological profile
Brexanolone exposure-response relationships and the time course of pharmacodynamic response are unknown.3
Adverse reactions. Safety was evaluated from all patients receiving brexanolone injection, regardless of dosing regimen (N = 140, including patients from a Phase IIb study, 202A).3,11
The most common adverse reactions (incidence ≥5% and at least twice the rate of placebo) were sedation/somnolence, dry mouth, loss of consciousness, and flushing/hot flush.3 The incidence of patients discontinuing due to any adverse reaction was 2% for brexanolone vs 1% for placebo.3
Sedation, somnolence, and loss of consciousness. In clinical studies, brexanolone caused sedation and somnolence that required dose interruption or reduction in some patients during the infusion (5% of brexanolone-treated patients compared with 0% of placebo-treated patients).3 Some patients were also reported to have loss of consciousness or altered state of consciousness during the brexanolone infusion (4% of patients treated with brexanolone compared with 0% of patients treated with placebo).3 All patients with loss of or altered state of consciousness recovered fully 15 to 60 minutes after dose interruption.3 There was no clear association between loss or alteration of consciousness and pattern or timing of dose, and not all patients who experienced a loss or alteration of consciousness reported sedation or somnolence before the episode.
Continue to: Suicidality
Suicidality. The risk of developing suicidal thoughts and behaviors with brexanolone is unknown, due to the relatively low number of exposures to brexanolone injection during clinical development and a mechanism of action distinct from that of existing antidepressant medications.3
Pharmacokinetics
In clinical trials, brexanolone exhibited dose-proportional pharmacokinetics, and the terminal half-life is approximately 9 hours (Table 43). Brexanolone is metabolized by non-cytochrome P450 (CYP)-based pathways, including keto-reduction, glucuronidation, and sulfation.3 No clinically significant differences in the pharmacokinetics of brexanolone were observed based on renal or hepatic impairment, and no studies were conducted to evaluate the effects of other drugs on brexanolone.3
Lactation. A population pharmacokinetics model constructed from studies in the clinical development program calculated the maximum relative infant dose for brexanolone during infusion as 1.3%.3 Given the low oral bioavailability of brexanolone (<5%) in adults, the potential for breastfed infant exposure is considered low.3
Clinical considerations
Risk Evaluation and Mitigation Strategies (REMS) requirements. Brexanolone injection is a Schedule IV controlled substance. It has a “black-box” warning regarding excessive sedation and sudden loss of consciousness, which has been taken into account within the REMS drug safety program. Health care facilities and pharmacies must enroll in the REMS program and ensure that brexanolone is administered only to patients who are enrolled in the REMS program. Staff must be trained on the processes and procedures to administer brexanolone, and the facility must have a fall precautions protocol in place and be equipped with a programmable peristaltic IV infusion pump and continuous pulse oximetry with alarms.3
Monitoring. A REMS-trained clinician must be available continuously on-site to oversee each patient for the duration of the continuous IV infusion, which lasts 60 hours (2.5 days) and should be initiated early enough in the day to allow for recognition of excessive sedation. Patients must be monitored for hypoxia using continuous pulse oximetry equipped with an alarm and should also be assessed for excessive sedation every 2 hours during planned, non-sleep periods. If excessive sedation occurs, the infusion should be stopped until symptoms resolve, after which the infusion may be resumed at the same or a lower dose as clinically appropriate. In case of overdosage, the infusion should be stopped immediately and supportive measures initiated as necessary. Patients must not be the primary caregiver of dependents, and must be accompanied during interactions with their child(ren).
Continue to: Contraindications
Contraindications. There are no contraindications for the use of brexanolone in adults with PPD.
End-stage renal disease (ESRD). Avoid using brexanolone in patients with ESRD because of the potential accumulation of the solubilizing agent, betadex sulfobutyl ether sodium.
Pregnancy. Brexanolone has not been studied in pregnant patients. Pregnant women and women of reproductive age should be informed of the potential risk to a fetus based on data from other drugs that enhance GABAergic inhibition.
Breastfeeding. There are no data on the effects of brexanolone on a breastfed infant. Breastfeeding should be a discussion of risk and benefit between the patient and her doctor. The developmental and health benefits of breastfeeding should be considered, along with the mother’s clinical need for brexanolone and any potential adverse effects on the breastfed child from brexanolone or from the underlying maternal condition. However, based on the low relative infant dose (<2%) and the low oral bioavailability in adults, the risk to breastfed infants is thought to be low.16
Potential for abuse. Brexanolone injection is a Schedule IV controlled substance. Although it was not possible to assess physical dependency in the registrational trials due to dose tapering at the end of treatment, clinicians should advise patients about the theoretical possibility for brexanolone to be abused or lead to dependence based on other medications with similar primary pharmacology.
Continue to: Concomitant medications
Concomitant medications. Caution patients that taking opioids or other CNS depressants, such as benzodiazepines, in combination with brexanolone may increase the severity of sedative effects.
Suicidal thoughts and behaviors. Advise patients and caregivers to look for the emergence of suicidal thoughts and behavior and instruct them to report such symptoms to their clinician. Consider changing the therapeutic regimen, including discontinuing brexanolone, in patients whose depression becomes worse or who experience emergent suicidal thoughts and behaviors.
Why Rx?
Postpartum depression is a common and often devastating medical complication of childbirth that can result in adverse outcomes for the patient, baby, and family when left undertreated or untreated. There is a great need to identify and treat women who develop PPD. Rapid and sustained resolution of symptoms in women who experience PPD should be the goal of treatment, and consequently, brexanolone injection presents an important new tool in available treatment options for PPD.
Bottom Line
Brexanolone injection is a neuroactive steroid gamma-aminobutyric acid (GABA) A receptor positive allosteric modulator that’s been FDA-approved for the treatment of postpartum depression (PPD). It is administered as a continuous IV infusion over 60 hours. The rapid and sustained improvement of PPD observed in clinical trials with brexanolone injection may support a new treatment paradigm for women with PPD.
Postpartum depression (PPD) is one of the most prevalent complications associated with pregnancy and childbirth in the United States, affecting more than 400,000 women annually.1 Postpartum depression is most commonly treated with psychotherapy and antidepressants approved for the treatment of major depressive disorder. Until recently, there was no pharmacologic therapy approved by the FDA specifically for the treatment of PPD. Considering the adverse outcomes associated with untreated or inadequately treated PPD, and the limitations of existing therapies, there is a significant unmet need for pharmacologic treatment options for PPD.2 To help address this need, the FDA recently approved brexanolone injection (brand name: ZULRESSO™) (Table 13) as a first-in-class therapy for the treatment of adults with PPD.3
Clinical implications
Postpartum depression can result in adverse outcomes for the patient, baby, and family when under- or untreated, and the need for rapid resolution of symptoms cannot be overstated.2 Suicide is strongly associated with depression and is a leading cause of pregnancy-related deaths.4 Additionally, PPD can impact the health, safety, and well-being of the child, with both short- and long-term consequences, including greater rates of psychological or behavioral difficulties among children of patients with PPD.5 Postpartum depression can also have negative effects on the patient’s partner, with 24% to 50% of partners experiencing depression.6 Current PPD management strategies include the use of psychotherapy and pharmacologic interventions for major depressive disorder that may take up to 4 to 6 weeks for some patients, and may not achieve remission for all patients.7-9
Brexanolone injection is a first-in-class medication with a novel mechanism of action. In clinical studies, it achieved rapid (by Hour 60) and sustained (through Day 30) reductions in depressive symptoms and could provide a meaningful new treatment option for adult women with PPD.10,11
How it works
Animal and human studies have established the re
Brexanolone is a neuroactive steroid that is chemically identical to endogenous allopregnanolone produced in the CNS. Brexanolone potentiates GABA-mediated currents from recombinant human GABAARs in mammalian cells expressing α1β2γ2 receptor subunits, α4β3δ receptor subunits, and α6β3δ receptor subunits.3 Positive allosteric modulation of both synaptic and extrasynaptic GABAARs differentiates brexanolone from other GABAAR modulators, such as benzodiazepines.10,11
Brexanolone’s mechanism of action in the treatment of PPD is not fully understood, but it is thought to be related to GABAAR PAM activity.3
Supporting evidence
The FDA approval of brexanolone injection was based on the efficacy demonstrated in 2 Phase III multicenter, randomized, double-blind, placebo-controlled studies in adult women (age 18 to 45) with PPD (defined by DSM-IV criteria for a major depressive episode, with onset of symptoms in the third trimester or within 4 weeks of delivery). Exclusion criteria included the presence of bipolar disorder or psychosis. In these studies, 60-hour continuous IV infusions of brexanolone or placebo were given, followed by 4 weeks of observation. Study 1 (202B) enrolled patients with severe PPD (Hamilton Rating Scale for Depression [HAM-D] total score ≥26), and Study 2 (202C) enrolled patients with moderate PPD (HAM-D score 20 to 25). A titration to the recommended target dosage of 90 μg/kg/hour was evaluated in both studies. BRX90 patients received 30 μg/kg/hour for 4 hours, 60 μg/kg/hour for 20 hours, 90 μg/kg/hour for 28 hours, followed by a taper to 60 μg/kg/hour for 4 hours and then 30 μg/kg/hour for 4 hours. The primary endpoint in both studies was the mean change from baseline in depressive symptoms as measured by HAM-D total score at the end of the 60-hour infusion. A pre-specified secondary efficacy endpoint was the mean change from baseline in HAM-D total score at Day 30.
Continue to: Efficacy
Efficacy. In both placebo-controlled studies, titration to a target dose of brexanolone 90 μg/kg/hour was superior to placebo in improvement of depressive symptoms (Table 33).
Pharmacological profile
Brexanolone exposure-response relationships and the time course of pharmacodynamic response are unknown.3
Adverse reactions. Safety was evaluated from all patients receiving brexanolone injection, regardless of dosing regimen (N = 140, including patients from a Phase IIb study, 202A).3,11
The most common adverse reactions (incidence ≥5% and at least twice the rate of placebo) were sedation/somnolence, dry mouth, loss of consciousness, and flushing/hot flush.3 The incidence of patients discontinuing due to any adverse reaction was 2% for brexanolone vs 1% for placebo.3
Sedation, somnolence, and loss of consciousness. In clinical studies, brexanolone caused sedation and somnolence that required dose interruption or reduction in some patients during the infusion (5% of brexanolone-treated patients compared with 0% of placebo-treated patients).3 Some patients were also reported to have loss of consciousness or altered state of consciousness during the brexanolone infusion (4% of patients treated with brexanolone compared with 0% of patients treated with placebo).3 All patients with loss of or altered state of consciousness recovered fully 15 to 60 minutes after dose interruption.3 There was no clear association between loss or alteration of consciousness and pattern or timing of dose, and not all patients who experienced a loss or alteration of consciousness reported sedation or somnolence before the episode.
Continue to: Suicidality
Suicidality. The risk of developing suicidal thoughts and behaviors with brexanolone is unknown, due to the relatively low number of exposures to brexanolone injection during clinical development and a mechanism of action distinct from that of existing antidepressant medications.3
Pharmacokinetics
In clinical trials, brexanolone exhibited dose-proportional pharmacokinetics, and the terminal half-life is approximately 9 hours (Table 43). Brexanolone is metabolized by non-cytochrome P450 (CYP)-based pathways, including keto-reduction, glucuronidation, and sulfation.3 No clinically significant differences in the pharmacokinetics of brexanolone were observed based on renal or hepatic impairment, and no studies were conducted to evaluate the effects of other drugs on brexanolone.3
Lactation. A population pharmacokinetics model constructed from studies in the clinical development program calculated the maximum relative infant dose for brexanolone during infusion as 1.3%.3 Given the low oral bioavailability of brexanolone (<5%) in adults, the potential for breastfed infant exposure is considered low.3
Clinical considerations
Risk Evaluation and Mitigation Strategies (REMS) requirements. Brexanolone injection is a Schedule IV controlled substance. It has a “black-box” warning regarding excessive sedation and sudden loss of consciousness, which has been taken into account within the REMS drug safety program. Health care facilities and pharmacies must enroll in the REMS program and ensure that brexanolone is administered only to patients who are enrolled in the REMS program. Staff must be trained on the processes and procedures to administer brexanolone, and the facility must have a fall precautions protocol in place and be equipped with a programmable peristaltic IV infusion pump and continuous pulse oximetry with alarms.3
Monitoring. A REMS-trained clinician must be available continuously on-site to oversee each patient for the duration of the continuous IV infusion, which lasts 60 hours (2.5 days) and should be initiated early enough in the day to allow for recognition of excessive sedation. Patients must be monitored for hypoxia using continuous pulse oximetry equipped with an alarm and should also be assessed for excessive sedation every 2 hours during planned, non-sleep periods. If excessive sedation occurs, the infusion should be stopped until symptoms resolve, after which the infusion may be resumed at the same or a lower dose as clinically appropriate. In case of overdosage, the infusion should be stopped immediately and supportive measures initiated as necessary. Patients must not be the primary caregiver of dependents, and must be accompanied during interactions with their child(ren).
Continue to: Contraindications
Contraindications. There are no contraindications for the use of brexanolone in adults with PPD.
End-stage renal disease (ESRD). Avoid using brexanolone in patients with ESRD because of the potential accumulation of the solubilizing agent, betadex sulfobutyl ether sodium.
Pregnancy. Brexanolone has not been studied in pregnant patients. Pregnant women and women of reproductive age should be informed of the potential risk to a fetus based on data from other drugs that enhance GABAergic inhibition.
Breastfeeding. There are no data on the effects of brexanolone on a breastfed infant. Breastfeeding should be a discussion of risk and benefit between the patient and her doctor. The developmental and health benefits of breastfeeding should be considered, along with the mother’s clinical need for brexanolone and any potential adverse effects on the breastfed child from brexanolone or from the underlying maternal condition. However, based on the low relative infant dose (<2%) and the low oral bioavailability in adults, the risk to breastfed infants is thought to be low.16
Potential for abuse. Brexanolone injection is a Schedule IV controlled substance. Although it was not possible to assess physical dependency in the registrational trials due to dose tapering at the end of treatment, clinicians should advise patients about the theoretical possibility for brexanolone to be abused or lead to dependence based on other medications with similar primary pharmacology.
Continue to: Concomitant medications
Concomitant medications. Caution patients that taking opioids or other CNS depressants, such as benzodiazepines, in combination with brexanolone may increase the severity of sedative effects.
Suicidal thoughts and behaviors. Advise patients and caregivers to look for the emergence of suicidal thoughts and behavior and instruct them to report such symptoms to their clinician. Consider changing the therapeutic regimen, including discontinuing brexanolone, in patients whose depression becomes worse or who experience emergent suicidal thoughts and behaviors.
Why Rx?
Postpartum depression is a common and often devastating medical complication of childbirth that can result in adverse outcomes for the patient, baby, and family when left undertreated or untreated. There is a great need to identify and treat women who develop PPD. Rapid and sustained resolution of symptoms in women who experience PPD should be the goal of treatment, and consequently, brexanolone injection presents an important new tool in available treatment options for PPD.
Bottom Line
Brexanolone injection is a neuroactive steroid gamma-aminobutyric acid (GABA) A receptor positive allosteric modulator that’s been FDA-approved for the treatment of postpartum depression (PPD). It is administered as a continuous IV infusion over 60 hours. The rapid and sustained improvement of PPD observed in clinical trials with brexanolone injection may support a new treatment paradigm for women with PPD.
1. Ko JY, Rockhill KM, Tong VT, et al. Trends in postpartum depressive symptoms - 27 states, 2004, 2008, and 2012. MMWR Morb Mortal Wkly Rep. 2017;66(6):153-158.
2. Frieder A, Fersh M, Hainline R, et al. Pharmacotherapy of postpartum depression: current approaches and novel drug development. CNS Drugs. 2019;33(3):265-282.
3. Brexanolone injection [package insert]. Cambridge, MA: Sage Therapeutics, Inc.; 2019.
4. Bodnar-Deren S, Klipstein K, Fersh M, et al. Suicidal ideation during the postpartum period. J Womens Health (Larchmt). 2016;25(12):1219-1224.
5. Netsi E, Pearson RM, Murray L, et al. Association of persistent and severe postnatal depression with child outcomes. JAMA Psychiatry. 2018;75(3):247-253.
6. Goodman JH. Paternal postpartum depression, its relationship to maternal postpartum depression, and implications for family health. J Adv Nurs. 2004;45(1):26-35.
7. Gelenberg AJ, Freeman MP, Markowitz JC, et al; American Psychiatric Association Work Group on Major Depressive Disorder. Practice guidelines for the treatment of patients with major depressive disorder. 3rd ed. Washington, DC: American Psychiatric Association; 2010.
8. Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163(11):1905-1917.
9. Molyneaux E, Telesia LA, Henshaw C, et al. Antidepressants for preventing postnatal depression. Cochrane Database Syst Rev. 2018;4:CD004363.
10. Kanes S, Colquhoun H, Gunduz-Bruce H, et al. Brexanolone (SAGE-547 injection) in post-partum depression: a randomised controlled trial. Lancet. 2017;390(10093):480-489.
11. Meltzer-Brody S, Colquhoun H, Riesenberg R, et al. Brexanolone injection in post-partum depression: two multicentre, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet. 2018;392(10152):1058-1070.
12. Melon LC, Hooper A, Yang X, et al. Inability to suppress the stress-induced activation of the HPA axis during the peripartum period engenders deficits in postpartum behaviors in mice. Psychoneuroendocrinology. 2018;90:182-193.
13. Deligiannidis KM, Fales CL, Kroll-Desrosiers AR, et al. Resting-state functional connectivity, cortical GABA, and neuroactive steroids in peripartum and peripartum depressed women: a functional magnetic resonance imaging and spectroscopy study. Neuropsychopharmacology. 2019;44(3):546-554.
14. Licheri V, Talani G, Gorule AA, et al. Plasticity of GABAA receptors during pregnancy and postpartum period: from gene to function. Neural Plast. 2015;2015:170435. doi: 10.1155/2015/170435.
15. Luisi S, Petraglia F, Benedetto C, et al. Serum allopregnanolone levels in pregnant women: changes during pregnancy, at delivery, and in hypertensive patients. J Clin Endocrinol Metab. 2000;85(7):2429-2433.
16. Hoffmann E, Wald J, Dray D, et al. Brexanolone injection administration to lactating women: breast milk allopregnanolone levels [30J]. Obstetrics & Gynecology. 2019;133:115S.
1. Ko JY, Rockhill KM, Tong VT, et al. Trends in postpartum depressive symptoms - 27 states, 2004, 2008, and 2012. MMWR Morb Mortal Wkly Rep. 2017;66(6):153-158.
2. Frieder A, Fersh M, Hainline R, et al. Pharmacotherapy of postpartum depression: current approaches and novel drug development. CNS Drugs. 2019;33(3):265-282.
3. Brexanolone injection [package insert]. Cambridge, MA: Sage Therapeutics, Inc.; 2019.
4. Bodnar-Deren S, Klipstein K, Fersh M, et al. Suicidal ideation during the postpartum period. J Womens Health (Larchmt). 2016;25(12):1219-1224.
5. Netsi E, Pearson RM, Murray L, et al. Association of persistent and severe postnatal depression with child outcomes. JAMA Psychiatry. 2018;75(3):247-253.
6. Goodman JH. Paternal postpartum depression, its relationship to maternal postpartum depression, and implications for family health. J Adv Nurs. 2004;45(1):26-35.
7. Gelenberg AJ, Freeman MP, Markowitz JC, et al; American Psychiatric Association Work Group on Major Depressive Disorder. Practice guidelines for the treatment of patients with major depressive disorder. 3rd ed. Washington, DC: American Psychiatric Association; 2010.
8. Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163(11):1905-1917.
9. Molyneaux E, Telesia LA, Henshaw C, et al. Antidepressants for preventing postnatal depression. Cochrane Database Syst Rev. 2018;4:CD004363.
10. Kanes S, Colquhoun H, Gunduz-Bruce H, et al. Brexanolone (SAGE-547 injection) in post-partum depression: a randomised controlled trial. Lancet. 2017;390(10093):480-489.
11. Meltzer-Brody S, Colquhoun H, Riesenberg R, et al. Brexanolone injection in post-partum depression: two multicentre, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet. 2018;392(10152):1058-1070.
12. Melon LC, Hooper A, Yang X, et al. Inability to suppress the stress-induced activation of the HPA axis during the peripartum period engenders deficits in postpartum behaviors in mice. Psychoneuroendocrinology. 2018;90:182-193.
13. Deligiannidis KM, Fales CL, Kroll-Desrosiers AR, et al. Resting-state functional connectivity, cortical GABA, and neuroactive steroids in peripartum and peripartum depressed women: a functional magnetic resonance imaging and spectroscopy study. Neuropsychopharmacology. 2019;44(3):546-554.
14. Licheri V, Talani G, Gorule AA, et al. Plasticity of GABAA receptors during pregnancy and postpartum period: from gene to function. Neural Plast. 2015;2015:170435. doi: 10.1155/2015/170435.
15. Luisi S, Petraglia F, Benedetto C, et al. Serum allopregnanolone levels in pregnant women: changes during pregnancy, at delivery, and in hypertensive patients. J Clin Endocrinol Metab. 2000;85(7):2429-2433.
16. Hoffmann E, Wald J, Dray D, et al. Brexanolone injection administration to lactating women: breast milk allopregnanolone levels [30J]. Obstetrics & Gynecology. 2019;133:115S.
‘Miracle cures’ in psychiatry?
For a patient with a major mental illness, the road to wellness is long and uncertain. The medications commonly used to treat mood and thought disorders can take weeks to months to start providing benefits, and they carry significant risks for adverse effects, such as weight gain, sexual dysfunction, and movement disorders. Patients often have to take psychotropic medications for the rest of their lives. In addition to these downsides, there is no guarantee that these medications will provide complete or even partial relief.2,3
Recently, there has been growing excitement about new treatments that might be “miracle cures” for patients with mental illness, particularly for individuals with treatment-resistant depression (TRD). Two of these treatments—ketamine-related compounds, and hallucinogenic drugs—seem to promise therapeutic effects that are vastly different from those of other psychiatric medications: They appear to improve patients’ symptoms very quickly, and their effects may persist long after these drugs have been cleared from the body.
Intravenous ketamine is an older generic drug used in anesthesia; recently, it has been used off-label for TRD and other mental illnesses. On March 5, 2019, the FDA approved an intranasal formulation of esketamine—the S-enantiomer of ketamine—for TRD.4 Hallucinogens have also been tested in small studies and have seemingly significant effects in alleviating depression in patients with terminal illnesses5 and reducing smoking behavior in patients with tobacco use disorder.6,7
These miracle cures are becoming increasingly available to patients and continue to gain credibility among clinicians and researchers. How should we evaluate the usefulness of these new treatments? And how should we talk to our patients about them? To answer these questions, this article:
- explores our duty to our patients, ourselves, and our colleagues
- describes the dilemma
- discusses ways to evaluate claims made about these new miracle cures.
Duty: Protecting and helping our patients
The physician–patient relationship is a fiduciary relationship. According to both common law and medical ethics, a physician who enters into a treatment relationship with a patient creates a bond of special trust and confidence. Such a relationship requires a physician to act in good faith and in the patient’s best interests.8 As physicians, we have a duty to evaluate the safety and efficacy of new treatments that are available for our patients, whether or not they are FDA-approved.
We should also protect our patients from the adverse consequences of relatively untested drugs. For example, ketamine and hallucinogens both produce dissociative effects, and may carry high risks for patients who have a predisposition to psychosis.9 We should protect our patients from any false hopes that might lead them to abandon their current treatment regimens due to adverse effects and imperfect results. At the same time, we also have a duty to acknowledge our patients’ suffering and to recognize that they might be desperate for new treatment options. We should remain open-minded about new treatments, and acknowledge that they might work. Finally, we have a duty to be mindful of any financial benefits that we may derive from the development, marketing, and administration of these medications.
Dilemma: The need for new treatments
This is not the first time that novel treatments in mental health have seemed to hold incredible promise. In the late 1800s, Sigmund Freud began to regularly use a compound that led him to feel “the normal euphoria of a healthy person.” He wrote that this substance produced:
…exhilaration and lasting euphoria, which does not differ in any way from the normal euphoria of a healthy person. The feeling of excitement which accompanies stimulus by alcohol is completely lacking; the characteristic urge for immediate activity which alcohol produces is also absent. One senses an increase of self-control and feels more vigorous and more capable of work; on the other hand, if one works, one misses that heightening of the mental powers which alcohol, tea, or coffee induce. One is simply normal, and soon finds it difficult to believe that one is under the influence of any drug at all.1
Continue to: The compound Freud was describing...
The compound Freud was describing is cocaine, which we now know is an addictive and dangerous drug that can in fact worsen depression.10 Another treatment regarded as a miracle cure in its time involved placing patients with schizophrenia into an insulin-induced coma to treat their symptoms; this therapy was used from 1933 to 1960.11 We now recognize that this practice is unacceptably dangerous.
The past is filled with cautionary tales of the enthusiastic adoption of treatments for mental illness that later turned out to be ineffective, counterproductive, dangerous, or inhumane. Yet, the long, arduous journeys our patients go through continue to weigh heavily on us. We would love to offer our patients newer, more efficacious, and longer-lasting treatments with fewer adverse effects.
Discussion: How to best evaluate miracle cures
To help quickly assess a new treatment, the following 5 categories can help guide and organize our thought process.
1. Evidence
What type of evidence do we have that a new treatment is safe and effective? Psychiatric research may be even more susceptible to a placebo effect than other medical research, particularly for illnesses with subjective symptoms, such as depression.12 Double-blinded, placebo-controlled studies, such as the IV ketamine trial conducted by Singh et al,13 are the gold standard for separating a substance’s actual biologic effect from a placebo effect. Studies that do not include a control group should not be regarded as providing scientific evidence of efficacy.
2. Mechanism
If a new compound appears to have a beneficial effect on mental health, it is important to consider the potential mechanism underlying this effect to determine if it is biologically plausible. A compound that is claimed to be a panacea for every symptom of every mental illness should be heavily scrutinized. For example, based on available research, ketamine’s long-lasting effects seem to come from 2 mechanisms14,15:
- Activation of endogenous opioid receptors, which is also responsible for the euphoria induced by heroin and oxycodone.
- Blockade of N-methyl-
D -aspartate receptors. N-methyl-D -aspartate receptor activation is a key mechanism by which learning and memory function in the brain, and blocking these receptors may increase brain plasticity.
Continue to: Therefore, it seems plausible...
Therefore, it seems plausible that ketamine could produce both short- and long-term improvements in mood. Hallucinogenic drugs are thought to profoundly alter brain function through several mechanisms, including activating serotonin receptors, enhancing brain plasticity, and increasing brain connectivity.16
3. Reinforcement
Psychiatric medications that are acutely reinforcing have significant potential for abuse. Antidepressants and mood stabilizers are not acutely rewarding. They don’t make patients feel good right away. Medications such as stimulants and opioids do, and must be used with extreme care because of their abuse potential. The problem with acutely reinforcing medications is that in the long run, they can worsen depression by decreasing the brain’s ability to produce endogenous opioids.17
4.
A mental disorder is unlikely to have a single solution. Rather than regarding a new treatment as capable of rapidly alleviating every symptom of a patient’s illness, it should be viewed as a tool that can be helpful when used in combination with other treatments and lifestyle practices. In an interview with the web site STAT, Cristina Cusin, MD, co-director of the Intravenous Ketamine Clinic for Depression at Massachusetts General Hospital, said, “You don’t treat an advanced disease with just an infusion and a ‘see you next time.’ If [doctors] replace your knee but [you] don’t do physical therapy, you don’t walk again.”18 To sustain the benefits of a novel medication, patients with serious mental illnesses need to maintain strong social supports, see a mental health care provider regularly, and abstain from illicit drug and alcohol use.
5. Context matters
For a medication to obtain approval to treat a specific indication, the FDA usually require 2 trials that demonstrate efficacy. Off-label use of generic medications such as ketamine may have benefits, but it is unlikely that a generic drug would be put through a costly FDA-approval process.19
When learning about new medications, remember that patients might assume that these agents have undergone a thorough review process for safety and effectiveness. When our patients request such treatments—whether FDA-approved or off-label—it is our responsibility as physicians to educate them about the benefits, risks, effectiveness, and limitations of these treatments, as well as to evaluate the appropriateness of a treatment for a specific patient’s symptoms.
Continue to: Tempering excitement with caution
Tempering excitement with caution
Our patients are not the only ones desperate for a miracle cure. As psychiatrists, many of us are desperate, too. New compounds may ultimately change the way we treat mental illness. However, we have an obligation to temper our excitement with caution by remembering past mistakes, and systematically evaluating new miracle cures to determine if they are safe and effective.
1. Freud S. Cocaine papers. In: Freud S, Byck R. Sigmund Freud collection (Library of Congress). New York, NY: Stonehill; 1975;7.
2. Rush AJ. STAR*D: what have we learned? Am J Psychiatry. 2007;164(2):201-204.
3. Demjaha A, Lappin JM, Stahl D, et al. Antipsychotic treatment resistance in first-episode psychosis: prevalence, subtypes and predictors. Psychol Med. 2017;47(11):1981-1989.
4. Carey B. Fast-acting depression drug, newly approved, could help millions. The New York Times. https://www.nytimes.com/2019/03/05/health/depression-treatment-ketamine-fda.html. Published March 5, 2019. Accessed July 26, 2019.
5. Griffiths RR, Johnson MW, Carducci MA, et al. Psilocybin produces substantial and sustained decreases in depression and anxiety in patients with life-threatening cancer: a randomized double-blind trial. J Psychopharmacol. 2016;30(12):1181-1197.
6. Johnson MW, Garcia-Romeu A, Griffiths RR. Long-term follow-up of psilocybin-facilitated smoking cessation. Am J Drug Alcohol Abuse. 2017;43:55-60.
7. Garcia-Romeu A, Griffiths RR, Johnson MW. Psilocybin-occasioned mystical experiences in the treatment of tobacco addiction. Curr Drug Abuse Rev 2014;7(3):157-164.
8. Simon RI. Clinical psychiatry and the law. 2nd ed. Washington, DC: American Psychiatric Press; 1992.
9. Lahti AC, Weiler MA, Tamara Michaelidis BA, et al. Effects of ketamine in normal and schizophrenic volunteers. Neuropsychopharmacology. 2001;25(4):455-467.
10. Perrine SA, Sheikh IS, Nwaneshiudu CA, et al. Withdrawal from chronic administration of cocaine decreases delta opioid receptor signaling and increases anxiety- and depression-like behaviors in the rat. Neuropharmacology. 2008;54(2):355-364.
11. Doroshow DB. Performing a cure for schizophrenia: insulin coma therapy on the wards. J Hist Med Allied Sci. 2007;62(2):213-243.
12. Khan A, Kolts RL, Rapaport MH, et al. Magnitude of placebo response and drug-placebo differences across psychiatric disorders. Psychol Med. 2005;35(5):743-749.
13. Singh JB, Fedgchin M, Daly EJ, et al. A double-blind, randomized, placebo-controlled, dose-frequency study of intravenous ketamine in patients with treatment-resistant depression. Am J Psychiatry. 2016;173(8):816-826.
14. Williams NR, Heifets BD, Blasey C, et al. Attenuation of antidepressant effects of ketamine by opioid receptor antagonism. Am J Psychiatry. 2018;175(12):1205-1215.
15. Duman RS, Aghajanian GK, Sanacora G, et al. Synaptic plasticity and depression: new insights from stress and rapid-acting antidepressants. Nat Med. 2016;22(2):238-249.
16. Carhart-Harris RL. How do psychedelics work? Curr Opin Psychiatry. 2019;32(1):16-21.
17. Martins SS, Fenton MC, Keyes KM, et al. Mood and anxiety disorders and their association with non-medical prescription opioid use and prescription opioid-use disorder: longitudinal evidence from the National Epidemiologic Study on Alcohol and Related Conditions. Psychol Med. 2012;42(6):1261-1272.
18. Thielking M. Ketamine gives hope to patients with severe depression. But some clinics stray from the science and hype its benefits. STAT. https://www.statnews.com/2018/09/24/ketamine-clinics-severe-depression-treatment/. Published September 24, 2018. Accessed July 26, 2019.
19. Stafford RS. Regulating off-label drug use--rethinking the role of the FDA. N Engl J Med. 2008;358(14):1427-1429.
For a patient with a major mental illness, the road to wellness is long and uncertain. The medications commonly used to treat mood and thought disorders can take weeks to months to start providing benefits, and they carry significant risks for adverse effects, such as weight gain, sexual dysfunction, and movement disorders. Patients often have to take psychotropic medications for the rest of their lives. In addition to these downsides, there is no guarantee that these medications will provide complete or even partial relief.2,3
Recently, there has been growing excitement about new treatments that might be “miracle cures” for patients with mental illness, particularly for individuals with treatment-resistant depression (TRD). Two of these treatments—ketamine-related compounds, and hallucinogenic drugs—seem to promise therapeutic effects that are vastly different from those of other psychiatric medications: They appear to improve patients’ symptoms very quickly, and their effects may persist long after these drugs have been cleared from the body.
Intravenous ketamine is an older generic drug used in anesthesia; recently, it has been used off-label for TRD and other mental illnesses. On March 5, 2019, the FDA approved an intranasal formulation of esketamine—the S-enantiomer of ketamine—for TRD.4 Hallucinogens have also been tested in small studies and have seemingly significant effects in alleviating depression in patients with terminal illnesses5 and reducing smoking behavior in patients with tobacco use disorder.6,7
These miracle cures are becoming increasingly available to patients and continue to gain credibility among clinicians and researchers. How should we evaluate the usefulness of these new treatments? And how should we talk to our patients about them? To answer these questions, this article:
- explores our duty to our patients, ourselves, and our colleagues
- describes the dilemma
- discusses ways to evaluate claims made about these new miracle cures.
Duty: Protecting and helping our patients
The physician–patient relationship is a fiduciary relationship. According to both common law and medical ethics, a physician who enters into a treatment relationship with a patient creates a bond of special trust and confidence. Such a relationship requires a physician to act in good faith and in the patient’s best interests.8 As physicians, we have a duty to evaluate the safety and efficacy of new treatments that are available for our patients, whether or not they are FDA-approved.
We should also protect our patients from the adverse consequences of relatively untested drugs. For example, ketamine and hallucinogens both produce dissociative effects, and may carry high risks for patients who have a predisposition to psychosis.9 We should protect our patients from any false hopes that might lead them to abandon their current treatment regimens due to adverse effects and imperfect results. At the same time, we also have a duty to acknowledge our patients’ suffering and to recognize that they might be desperate for new treatment options. We should remain open-minded about new treatments, and acknowledge that they might work. Finally, we have a duty to be mindful of any financial benefits that we may derive from the development, marketing, and administration of these medications.
Dilemma: The need for new treatments
This is not the first time that novel treatments in mental health have seemed to hold incredible promise. In the late 1800s, Sigmund Freud began to regularly use a compound that led him to feel “the normal euphoria of a healthy person.” He wrote that this substance produced:
…exhilaration and lasting euphoria, which does not differ in any way from the normal euphoria of a healthy person. The feeling of excitement which accompanies stimulus by alcohol is completely lacking; the characteristic urge for immediate activity which alcohol produces is also absent. One senses an increase of self-control and feels more vigorous and more capable of work; on the other hand, if one works, one misses that heightening of the mental powers which alcohol, tea, or coffee induce. One is simply normal, and soon finds it difficult to believe that one is under the influence of any drug at all.1
Continue to: The compound Freud was describing...
The compound Freud was describing is cocaine, which we now know is an addictive and dangerous drug that can in fact worsen depression.10 Another treatment regarded as a miracle cure in its time involved placing patients with schizophrenia into an insulin-induced coma to treat their symptoms; this therapy was used from 1933 to 1960.11 We now recognize that this practice is unacceptably dangerous.
The past is filled with cautionary tales of the enthusiastic adoption of treatments for mental illness that later turned out to be ineffective, counterproductive, dangerous, or inhumane. Yet, the long, arduous journeys our patients go through continue to weigh heavily on us. We would love to offer our patients newer, more efficacious, and longer-lasting treatments with fewer adverse effects.
Discussion: How to best evaluate miracle cures
To help quickly assess a new treatment, the following 5 categories can help guide and organize our thought process.
1. Evidence
What type of evidence do we have that a new treatment is safe and effective? Psychiatric research may be even more susceptible to a placebo effect than other medical research, particularly for illnesses with subjective symptoms, such as depression.12 Double-blinded, placebo-controlled studies, such as the IV ketamine trial conducted by Singh et al,13 are the gold standard for separating a substance’s actual biologic effect from a placebo effect. Studies that do not include a control group should not be regarded as providing scientific evidence of efficacy.
2. Mechanism
If a new compound appears to have a beneficial effect on mental health, it is important to consider the potential mechanism underlying this effect to determine if it is biologically plausible. A compound that is claimed to be a panacea for every symptom of every mental illness should be heavily scrutinized. For example, based on available research, ketamine’s long-lasting effects seem to come from 2 mechanisms14,15:
- Activation of endogenous opioid receptors, which is also responsible for the euphoria induced by heroin and oxycodone.
- Blockade of N-methyl-
D -aspartate receptors. N-methyl-D -aspartate receptor activation is a key mechanism by which learning and memory function in the brain, and blocking these receptors may increase brain plasticity.
Continue to: Therefore, it seems plausible...
Therefore, it seems plausible that ketamine could produce both short- and long-term improvements in mood. Hallucinogenic drugs are thought to profoundly alter brain function through several mechanisms, including activating serotonin receptors, enhancing brain plasticity, and increasing brain connectivity.16
3. Reinforcement
Psychiatric medications that are acutely reinforcing have significant potential for abuse. Antidepressants and mood stabilizers are not acutely rewarding. They don’t make patients feel good right away. Medications such as stimulants and opioids do, and must be used with extreme care because of their abuse potential. The problem with acutely reinforcing medications is that in the long run, they can worsen depression by decreasing the brain’s ability to produce endogenous opioids.17
4.
A mental disorder is unlikely to have a single solution. Rather than regarding a new treatment as capable of rapidly alleviating every symptom of a patient’s illness, it should be viewed as a tool that can be helpful when used in combination with other treatments and lifestyle practices. In an interview with the web site STAT, Cristina Cusin, MD, co-director of the Intravenous Ketamine Clinic for Depression at Massachusetts General Hospital, said, “You don’t treat an advanced disease with just an infusion and a ‘see you next time.’ If [doctors] replace your knee but [you] don’t do physical therapy, you don’t walk again.”18 To sustain the benefits of a novel medication, patients with serious mental illnesses need to maintain strong social supports, see a mental health care provider regularly, and abstain from illicit drug and alcohol use.
5. Context matters
For a medication to obtain approval to treat a specific indication, the FDA usually require 2 trials that demonstrate efficacy. Off-label use of generic medications such as ketamine may have benefits, but it is unlikely that a generic drug would be put through a costly FDA-approval process.19
When learning about new medications, remember that patients might assume that these agents have undergone a thorough review process for safety and effectiveness. When our patients request such treatments—whether FDA-approved or off-label—it is our responsibility as physicians to educate them about the benefits, risks, effectiveness, and limitations of these treatments, as well as to evaluate the appropriateness of a treatment for a specific patient’s symptoms.
Continue to: Tempering excitement with caution
Tempering excitement with caution
Our patients are not the only ones desperate for a miracle cure. As psychiatrists, many of us are desperate, too. New compounds may ultimately change the way we treat mental illness. However, we have an obligation to temper our excitement with caution by remembering past mistakes, and systematically evaluating new miracle cures to determine if they are safe and effective.
For a patient with a major mental illness, the road to wellness is long and uncertain. The medications commonly used to treat mood and thought disorders can take weeks to months to start providing benefits, and they carry significant risks for adverse effects, such as weight gain, sexual dysfunction, and movement disorders. Patients often have to take psychotropic medications for the rest of their lives. In addition to these downsides, there is no guarantee that these medications will provide complete or even partial relief.2,3
Recently, there has been growing excitement about new treatments that might be “miracle cures” for patients with mental illness, particularly for individuals with treatment-resistant depression (TRD). Two of these treatments—ketamine-related compounds, and hallucinogenic drugs—seem to promise therapeutic effects that are vastly different from those of other psychiatric medications: They appear to improve patients’ symptoms very quickly, and their effects may persist long after these drugs have been cleared from the body.
Intravenous ketamine is an older generic drug used in anesthesia; recently, it has been used off-label for TRD and other mental illnesses. On March 5, 2019, the FDA approved an intranasal formulation of esketamine—the S-enantiomer of ketamine—for TRD.4 Hallucinogens have also been tested in small studies and have seemingly significant effects in alleviating depression in patients with terminal illnesses5 and reducing smoking behavior in patients with tobacco use disorder.6,7
These miracle cures are becoming increasingly available to patients and continue to gain credibility among clinicians and researchers. How should we evaluate the usefulness of these new treatments? And how should we talk to our patients about them? To answer these questions, this article:
- explores our duty to our patients, ourselves, and our colleagues
- describes the dilemma
- discusses ways to evaluate claims made about these new miracle cures.
Duty: Protecting and helping our patients
The physician–patient relationship is a fiduciary relationship. According to both common law and medical ethics, a physician who enters into a treatment relationship with a patient creates a bond of special trust and confidence. Such a relationship requires a physician to act in good faith and in the patient’s best interests.8 As physicians, we have a duty to evaluate the safety and efficacy of new treatments that are available for our patients, whether or not they are FDA-approved.
We should also protect our patients from the adverse consequences of relatively untested drugs. For example, ketamine and hallucinogens both produce dissociative effects, and may carry high risks for patients who have a predisposition to psychosis.9 We should protect our patients from any false hopes that might lead them to abandon their current treatment regimens due to adverse effects and imperfect results. At the same time, we also have a duty to acknowledge our patients’ suffering and to recognize that they might be desperate for new treatment options. We should remain open-minded about new treatments, and acknowledge that they might work. Finally, we have a duty to be mindful of any financial benefits that we may derive from the development, marketing, and administration of these medications.
Dilemma: The need for new treatments
This is not the first time that novel treatments in mental health have seemed to hold incredible promise. In the late 1800s, Sigmund Freud began to regularly use a compound that led him to feel “the normal euphoria of a healthy person.” He wrote that this substance produced:
…exhilaration and lasting euphoria, which does not differ in any way from the normal euphoria of a healthy person. The feeling of excitement which accompanies stimulus by alcohol is completely lacking; the characteristic urge for immediate activity which alcohol produces is also absent. One senses an increase of self-control and feels more vigorous and more capable of work; on the other hand, if one works, one misses that heightening of the mental powers which alcohol, tea, or coffee induce. One is simply normal, and soon finds it difficult to believe that one is under the influence of any drug at all.1
Continue to: The compound Freud was describing...
The compound Freud was describing is cocaine, which we now know is an addictive and dangerous drug that can in fact worsen depression.10 Another treatment regarded as a miracle cure in its time involved placing patients with schizophrenia into an insulin-induced coma to treat their symptoms; this therapy was used from 1933 to 1960.11 We now recognize that this practice is unacceptably dangerous.
The past is filled with cautionary tales of the enthusiastic adoption of treatments for mental illness that later turned out to be ineffective, counterproductive, dangerous, or inhumane. Yet, the long, arduous journeys our patients go through continue to weigh heavily on us. We would love to offer our patients newer, more efficacious, and longer-lasting treatments with fewer adverse effects.
Discussion: How to best evaluate miracle cures
To help quickly assess a new treatment, the following 5 categories can help guide and organize our thought process.
1. Evidence
What type of evidence do we have that a new treatment is safe and effective? Psychiatric research may be even more susceptible to a placebo effect than other medical research, particularly for illnesses with subjective symptoms, such as depression.12 Double-blinded, placebo-controlled studies, such as the IV ketamine trial conducted by Singh et al,13 are the gold standard for separating a substance’s actual biologic effect from a placebo effect. Studies that do not include a control group should not be regarded as providing scientific evidence of efficacy.
2. Mechanism
If a new compound appears to have a beneficial effect on mental health, it is important to consider the potential mechanism underlying this effect to determine if it is biologically plausible. A compound that is claimed to be a panacea for every symptom of every mental illness should be heavily scrutinized. For example, based on available research, ketamine’s long-lasting effects seem to come from 2 mechanisms14,15:
- Activation of endogenous opioid receptors, which is also responsible for the euphoria induced by heroin and oxycodone.
- Blockade of N-methyl-
D -aspartate receptors. N-methyl-D -aspartate receptor activation is a key mechanism by which learning and memory function in the brain, and blocking these receptors may increase brain plasticity.
Continue to: Therefore, it seems plausible...
Therefore, it seems plausible that ketamine could produce both short- and long-term improvements in mood. Hallucinogenic drugs are thought to profoundly alter brain function through several mechanisms, including activating serotonin receptors, enhancing brain plasticity, and increasing brain connectivity.16
3. Reinforcement
Psychiatric medications that are acutely reinforcing have significant potential for abuse. Antidepressants and mood stabilizers are not acutely rewarding. They don’t make patients feel good right away. Medications such as stimulants and opioids do, and must be used with extreme care because of their abuse potential. The problem with acutely reinforcing medications is that in the long run, they can worsen depression by decreasing the brain’s ability to produce endogenous opioids.17
4.
A mental disorder is unlikely to have a single solution. Rather than regarding a new treatment as capable of rapidly alleviating every symptom of a patient’s illness, it should be viewed as a tool that can be helpful when used in combination with other treatments and lifestyle practices. In an interview with the web site STAT, Cristina Cusin, MD, co-director of the Intravenous Ketamine Clinic for Depression at Massachusetts General Hospital, said, “You don’t treat an advanced disease with just an infusion and a ‘see you next time.’ If [doctors] replace your knee but [you] don’t do physical therapy, you don’t walk again.”18 To sustain the benefits of a novel medication, patients with serious mental illnesses need to maintain strong social supports, see a mental health care provider regularly, and abstain from illicit drug and alcohol use.
5. Context matters
For a medication to obtain approval to treat a specific indication, the FDA usually require 2 trials that demonstrate efficacy. Off-label use of generic medications such as ketamine may have benefits, but it is unlikely that a generic drug would be put through a costly FDA-approval process.19
When learning about new medications, remember that patients might assume that these agents have undergone a thorough review process for safety and effectiveness. When our patients request such treatments—whether FDA-approved or off-label—it is our responsibility as physicians to educate them about the benefits, risks, effectiveness, and limitations of these treatments, as well as to evaluate the appropriateness of a treatment for a specific patient’s symptoms.
Continue to: Tempering excitement with caution
Tempering excitement with caution
Our patients are not the only ones desperate for a miracle cure. As psychiatrists, many of us are desperate, too. New compounds may ultimately change the way we treat mental illness. However, we have an obligation to temper our excitement with caution by remembering past mistakes, and systematically evaluating new miracle cures to determine if they are safe and effective.
1. Freud S. Cocaine papers. In: Freud S, Byck R. Sigmund Freud collection (Library of Congress). New York, NY: Stonehill; 1975;7.
2. Rush AJ. STAR*D: what have we learned? Am J Psychiatry. 2007;164(2):201-204.
3. Demjaha A, Lappin JM, Stahl D, et al. Antipsychotic treatment resistance in first-episode psychosis: prevalence, subtypes and predictors. Psychol Med. 2017;47(11):1981-1989.
4. Carey B. Fast-acting depression drug, newly approved, could help millions. The New York Times. https://www.nytimes.com/2019/03/05/health/depression-treatment-ketamine-fda.html. Published March 5, 2019. Accessed July 26, 2019.
5. Griffiths RR, Johnson MW, Carducci MA, et al. Psilocybin produces substantial and sustained decreases in depression and anxiety in patients with life-threatening cancer: a randomized double-blind trial. J Psychopharmacol. 2016;30(12):1181-1197.
6. Johnson MW, Garcia-Romeu A, Griffiths RR. Long-term follow-up of psilocybin-facilitated smoking cessation. Am J Drug Alcohol Abuse. 2017;43:55-60.
7. Garcia-Romeu A, Griffiths RR, Johnson MW. Psilocybin-occasioned mystical experiences in the treatment of tobacco addiction. Curr Drug Abuse Rev 2014;7(3):157-164.
8. Simon RI. Clinical psychiatry and the law. 2nd ed. Washington, DC: American Psychiatric Press; 1992.
9. Lahti AC, Weiler MA, Tamara Michaelidis BA, et al. Effects of ketamine in normal and schizophrenic volunteers. Neuropsychopharmacology. 2001;25(4):455-467.
10. Perrine SA, Sheikh IS, Nwaneshiudu CA, et al. Withdrawal from chronic administration of cocaine decreases delta opioid receptor signaling and increases anxiety- and depression-like behaviors in the rat. Neuropharmacology. 2008;54(2):355-364.
11. Doroshow DB. Performing a cure for schizophrenia: insulin coma therapy on the wards. J Hist Med Allied Sci. 2007;62(2):213-243.
12. Khan A, Kolts RL, Rapaport MH, et al. Magnitude of placebo response and drug-placebo differences across psychiatric disorders. Psychol Med. 2005;35(5):743-749.
13. Singh JB, Fedgchin M, Daly EJ, et al. A double-blind, randomized, placebo-controlled, dose-frequency study of intravenous ketamine in patients with treatment-resistant depression. Am J Psychiatry. 2016;173(8):816-826.
14. Williams NR, Heifets BD, Blasey C, et al. Attenuation of antidepressant effects of ketamine by opioid receptor antagonism. Am J Psychiatry. 2018;175(12):1205-1215.
15. Duman RS, Aghajanian GK, Sanacora G, et al. Synaptic plasticity and depression: new insights from stress and rapid-acting antidepressants. Nat Med. 2016;22(2):238-249.
16. Carhart-Harris RL. How do psychedelics work? Curr Opin Psychiatry. 2019;32(1):16-21.
17. Martins SS, Fenton MC, Keyes KM, et al. Mood and anxiety disorders and their association with non-medical prescription opioid use and prescription opioid-use disorder: longitudinal evidence from the National Epidemiologic Study on Alcohol and Related Conditions. Psychol Med. 2012;42(6):1261-1272.
18. Thielking M. Ketamine gives hope to patients with severe depression. But some clinics stray from the science and hype its benefits. STAT. https://www.statnews.com/2018/09/24/ketamine-clinics-severe-depression-treatment/. Published September 24, 2018. Accessed July 26, 2019.
19. Stafford RS. Regulating off-label drug use--rethinking the role of the FDA. N Engl J Med. 2008;358(14):1427-1429.
1. Freud S. Cocaine papers. In: Freud S, Byck R. Sigmund Freud collection (Library of Congress). New York, NY: Stonehill; 1975;7.
2. Rush AJ. STAR*D: what have we learned? Am J Psychiatry. 2007;164(2):201-204.
3. Demjaha A, Lappin JM, Stahl D, et al. Antipsychotic treatment resistance in first-episode psychosis: prevalence, subtypes and predictors. Psychol Med. 2017;47(11):1981-1989.
4. Carey B. Fast-acting depression drug, newly approved, could help millions. The New York Times. https://www.nytimes.com/2019/03/05/health/depression-treatment-ketamine-fda.html. Published March 5, 2019. Accessed July 26, 2019.
5. Griffiths RR, Johnson MW, Carducci MA, et al. Psilocybin produces substantial and sustained decreases in depression and anxiety in patients with life-threatening cancer: a randomized double-blind trial. J Psychopharmacol. 2016;30(12):1181-1197.
6. Johnson MW, Garcia-Romeu A, Griffiths RR. Long-term follow-up of psilocybin-facilitated smoking cessation. Am J Drug Alcohol Abuse. 2017;43:55-60.
7. Garcia-Romeu A, Griffiths RR, Johnson MW. Psilocybin-occasioned mystical experiences in the treatment of tobacco addiction. Curr Drug Abuse Rev 2014;7(3):157-164.
8. Simon RI. Clinical psychiatry and the law. 2nd ed. Washington, DC: American Psychiatric Press; 1992.
9. Lahti AC, Weiler MA, Tamara Michaelidis BA, et al. Effects of ketamine in normal and schizophrenic volunteers. Neuropsychopharmacology. 2001;25(4):455-467.
10. Perrine SA, Sheikh IS, Nwaneshiudu CA, et al. Withdrawal from chronic administration of cocaine decreases delta opioid receptor signaling and increases anxiety- and depression-like behaviors in the rat. Neuropharmacology. 2008;54(2):355-364.
11. Doroshow DB. Performing a cure for schizophrenia: insulin coma therapy on the wards. J Hist Med Allied Sci. 2007;62(2):213-243.
12. Khan A, Kolts RL, Rapaport MH, et al. Magnitude of placebo response and drug-placebo differences across psychiatric disorders. Psychol Med. 2005;35(5):743-749.
13. Singh JB, Fedgchin M, Daly EJ, et al. A double-blind, randomized, placebo-controlled, dose-frequency study of intravenous ketamine in patients with treatment-resistant depression. Am J Psychiatry. 2016;173(8):816-826.
14. Williams NR, Heifets BD, Blasey C, et al. Attenuation of antidepressant effects of ketamine by opioid receptor antagonism. Am J Psychiatry. 2018;175(12):1205-1215.
15. Duman RS, Aghajanian GK, Sanacora G, et al. Synaptic plasticity and depression: new insights from stress and rapid-acting antidepressants. Nat Med. 2016;22(2):238-249.
16. Carhart-Harris RL. How do psychedelics work? Curr Opin Psychiatry. 2019;32(1):16-21.
17. Martins SS, Fenton MC, Keyes KM, et al. Mood and anxiety disorders and their association with non-medical prescription opioid use and prescription opioid-use disorder: longitudinal evidence from the National Epidemiologic Study on Alcohol and Related Conditions. Psychol Med. 2012;42(6):1261-1272.
18. Thielking M. Ketamine gives hope to patients with severe depression. But some clinics stray from the science and hype its benefits. STAT. https://www.statnews.com/2018/09/24/ketamine-clinics-severe-depression-treatment/. Published September 24, 2018. Accessed July 26, 2019.
19. Stafford RS. Regulating off-label drug use--rethinking the role of the FDA. N Engl J Med. 2008;358(14):1427-1429.
DBS vs TMS for treatment-resistant depression: A comparison
Approximately 20% to 30% of patients with major depressive disorder do not respond to pharmacotherapy.1 For patients with treatment-resistant depression (TRD)—typically defined as an inadequate response to at least 1 antidepressant trial of adequate dose and duration—neurostimulation may be an effective treatment option.
Two forms of neurostimulation used to treat TRD are deep brain stimulation (DBS) and transcranial magnetic stimulation (TMS). In DBS, electrodes are placed within the patient’s cranium and affixed to specific target locations. These electrodes are electrically stimulated at various frequencies. Transcranial magnetic stimulation is a noninvasive treatment in which a magnetic field is produced over a patient’s cranium, stimulating brain tissue via electromagnetic induction.
Media portraya
In this article, I compare DBS and TMS, and offer suggestions for educating patients about the potential adverse effects and therapeutic outcomes of each modality.
Deep brain stimulation
Deep brain stimulation is FDA-approved for treating Parkinson’s disease, essential tremor, dystonia, and obsessive-compulsive disorder (OCD).3 It has been used off-label for TRD, and some preliminary evidence suggests it is effective for this purpose. A review of 22 studies found that for patients with TRD, the rate of response to DBS (defined as >50% improvement on Hamilton Depression Rating Scale score) ranges from 40% to 70%.1 Additional research, including larger, randomized, sham-controlled trials, is needed.
A consensus on the optimal target location for DBS has not yet been reached. Studies have had varying degrees of symptom improvement targeting the subgenual cingulate gyrus, posterior gyrus rectus, nucleus accumbens, ventral capsule/ventral striatum, inferior thalamic peduncle, and lateral habenula.1
A worsening of depressive symptoms and increased risk of suicide have been reported in—but are not exclusive to—DBS. Patients treated with DBS may still meet the criteria for treatment resistance.
Continue to: The lack of insurance coverage...
The lack of insurance coverage for DBS for treating depression is a deterrent to its use. Because DBS is not FDA-approved for treating depression, the costs (approximately $65,000) that are not covered by a facility or study will fall on the patient.4 Patients may abandon hope for a positive therapeutic outcome if they must struggle with the financial responsibility for procedures and follow-up.4
Serious potential adverse events of DBS include infections, skin erosions, and postoperative seizure.4 Patients who are treated with DBS should be educated about these adverse effects, and how they may affect outcomes.
Transcranial magnetic stimulation
Transcranial magnetic stimulation is FDA-approved for treating depression, OCD, and migraine. Randomized, sham-controlled trials have found that TMS is effective for TRD.5 Studies have demonstrated varying degrees of efficacy, with response rates ranging from 47% to 58%.6
The most commonly used target area for TMS for patients with depression is the left dorsolateral prefrontal cortex.7 Potential adverse effects are relatively few and benign. The most serious adverse effect of TMS is a risk for seizure, which is reported to occur at a frequency of <0.1%.7
Although it varies by practice and location, the cost for an acute course of TMS (20 to 30 sessions) may range from $6,000 to $12,000.8 Most insurance companies cover TMS treatment for depression.
Continue to: TMS
TMS: A more accessible option
Compared with DBS, TMS is a more affordable and accessible therapy for patients with TRD. Further studies are needed to learn more about the therapeutic potential of DBS for TRD, and to develop methods that help decrease the risk of adverse effects. In addition, insurance coverage needs to be expanded to DBS to avoid having patients be responsible for the full costs of this treatment. Until then, TMS should be a recommended therapy for patients with TRD. If TRD persists in patients treated with TMS, consider electroconvulsive therapy.
1. Morishita T, Fayad SM, Higuchi MA, et al. Deep brain stimulation for treatment-resistant depression: systematic review of clinical outcomes. Neurotherapeutics. 2014;11(3):475-484.
2. Lawrence RE, Kaufmann CR, DeSilva RB, et al. Patients’ belief about deep brain stimulation for treatment resistant depression. AJOB Neuroscience, 2018;9(4):210-218.
3. Rossi PJ, Giordano J, Okun MS. The problem of funding off-label deep brain stimulation: bait-and-switch tactics and the need for policy reform. JAMA Neurol. 2017;74(1):9-10.
4. Holtzheimer PE, Husain MM, Lisanby SH, et al. Subcallosal cingulate deep brain stimulation for treatment-resistant depression: a multisite, randomised, sham-controlled trial. Lancet Psychiatry. 2017;4(11):839-849.
5. Janicak PG. What’s new in transcranial magnetic stimulation. Current Psychiatry. 2019;18(3):10-16.
6. Janicak PG, Sackett V, Kudrna K, et al. Advances in transcranial magnetic stimulation for managing major depressive disorders. Current Psychiatry. 2016;15(6):49-56.
7. Dobek CE, Blumberger DM, Downar J, et al. Risk of seizures in transcranial magnetic stimulation: a clinical review to inform consent process focused on bupropion. Neuropsychiatr Dis Treat. 2015;11:2975-2987.
8. McClintock SM, Reti IM, Carpenter LL, et al; National Network of Depression Centers rTMS Task Group; American Psychiatric Association Council on Research Task Force on Novel Biomarkers and Treatments. Consensus recommendations for the clinical application of repetitive transcranial magnetic stimulation (rTMS) in the treatment of depression. J Clin Psychiatry. 2018;79(1). doi: 10.4088/JCP.16cs10905.
Approximately 20% to 30% of patients with major depressive disorder do not respond to pharmacotherapy.1 For patients with treatment-resistant depression (TRD)—typically defined as an inadequate response to at least 1 antidepressant trial of adequate dose and duration—neurostimulation may be an effective treatment option.
Two forms of neurostimulation used to treat TRD are deep brain stimulation (DBS) and transcranial magnetic stimulation (TMS). In DBS, electrodes are placed within the patient’s cranium and affixed to specific target locations. These electrodes are electrically stimulated at various frequencies. Transcranial magnetic stimulation is a noninvasive treatment in which a magnetic field is produced over a patient’s cranium, stimulating brain tissue via electromagnetic induction.
Media portraya
In this article, I compare DBS and TMS, and offer suggestions for educating patients about the potential adverse effects and therapeutic outcomes of each modality.
Deep brain stimulation
Deep brain stimulation is FDA-approved for treating Parkinson’s disease, essential tremor, dystonia, and obsessive-compulsive disorder (OCD).3 It has been used off-label for TRD, and some preliminary evidence suggests it is effective for this purpose. A review of 22 studies found that for patients with TRD, the rate of response to DBS (defined as >50% improvement on Hamilton Depression Rating Scale score) ranges from 40% to 70%.1 Additional research, including larger, randomized, sham-controlled trials, is needed.
A consensus on the optimal target location for DBS has not yet been reached. Studies have had varying degrees of symptom improvement targeting the subgenual cingulate gyrus, posterior gyrus rectus, nucleus accumbens, ventral capsule/ventral striatum, inferior thalamic peduncle, and lateral habenula.1
A worsening of depressive symptoms and increased risk of suicide have been reported in—but are not exclusive to—DBS. Patients treated with DBS may still meet the criteria for treatment resistance.
Continue to: The lack of insurance coverage...
The lack of insurance coverage for DBS for treating depression is a deterrent to its use. Because DBS is not FDA-approved for treating depression, the costs (approximately $65,000) that are not covered by a facility or study will fall on the patient.4 Patients may abandon hope for a positive therapeutic outcome if they must struggle with the financial responsibility for procedures and follow-up.4
Serious potential adverse events of DBS include infections, skin erosions, and postoperative seizure.4 Patients who are treated with DBS should be educated about these adverse effects, and how they may affect outcomes.
Transcranial magnetic stimulation
Transcranial magnetic stimulation is FDA-approved for treating depression, OCD, and migraine. Randomized, sham-controlled trials have found that TMS is effective for TRD.5 Studies have demonstrated varying degrees of efficacy, with response rates ranging from 47% to 58%.6
The most commonly used target area for TMS for patients with depression is the left dorsolateral prefrontal cortex.7 Potential adverse effects are relatively few and benign. The most serious adverse effect of TMS is a risk for seizure, which is reported to occur at a frequency of <0.1%.7
Although it varies by practice and location, the cost for an acute course of TMS (20 to 30 sessions) may range from $6,000 to $12,000.8 Most insurance companies cover TMS treatment for depression.
Continue to: TMS
TMS: A more accessible option
Compared with DBS, TMS is a more affordable and accessible therapy for patients with TRD. Further studies are needed to learn more about the therapeutic potential of DBS for TRD, and to develop methods that help decrease the risk of adverse effects. In addition, insurance coverage needs to be expanded to DBS to avoid having patients be responsible for the full costs of this treatment. Until then, TMS should be a recommended therapy for patients with TRD. If TRD persists in patients treated with TMS, consider electroconvulsive therapy.
Approximately 20% to 30% of patients with major depressive disorder do not respond to pharmacotherapy.1 For patients with treatment-resistant depression (TRD)—typically defined as an inadequate response to at least 1 antidepressant trial of adequate dose and duration—neurostimulation may be an effective treatment option.
Two forms of neurostimulation used to treat TRD are deep brain stimulation (DBS) and transcranial magnetic stimulation (TMS). In DBS, electrodes are placed within the patient’s cranium and affixed to specific target locations. These electrodes are electrically stimulated at various frequencies. Transcranial magnetic stimulation is a noninvasive treatment in which a magnetic field is produced over a patient’s cranium, stimulating brain tissue via electromagnetic induction.
Media portraya
In this article, I compare DBS and TMS, and offer suggestions for educating patients about the potential adverse effects and therapeutic outcomes of each modality.
Deep brain stimulation
Deep brain stimulation is FDA-approved for treating Parkinson’s disease, essential tremor, dystonia, and obsessive-compulsive disorder (OCD).3 It has been used off-label for TRD, and some preliminary evidence suggests it is effective for this purpose. A review of 22 studies found that for patients with TRD, the rate of response to DBS (defined as >50% improvement on Hamilton Depression Rating Scale score) ranges from 40% to 70%.1 Additional research, including larger, randomized, sham-controlled trials, is needed.
A consensus on the optimal target location for DBS has not yet been reached. Studies have had varying degrees of symptom improvement targeting the subgenual cingulate gyrus, posterior gyrus rectus, nucleus accumbens, ventral capsule/ventral striatum, inferior thalamic peduncle, and lateral habenula.1
A worsening of depressive symptoms and increased risk of suicide have been reported in—but are not exclusive to—DBS. Patients treated with DBS may still meet the criteria for treatment resistance.
Continue to: The lack of insurance coverage...
The lack of insurance coverage for DBS for treating depression is a deterrent to its use. Because DBS is not FDA-approved for treating depression, the costs (approximately $65,000) that are not covered by a facility or study will fall on the patient.4 Patients may abandon hope for a positive therapeutic outcome if they must struggle with the financial responsibility for procedures and follow-up.4
Serious potential adverse events of DBS include infections, skin erosions, and postoperative seizure.4 Patients who are treated with DBS should be educated about these adverse effects, and how they may affect outcomes.
Transcranial magnetic stimulation
Transcranial magnetic stimulation is FDA-approved for treating depression, OCD, and migraine. Randomized, sham-controlled trials have found that TMS is effective for TRD.5 Studies have demonstrated varying degrees of efficacy, with response rates ranging from 47% to 58%.6
The most commonly used target area for TMS for patients with depression is the left dorsolateral prefrontal cortex.7 Potential adverse effects are relatively few and benign. The most serious adverse effect of TMS is a risk for seizure, which is reported to occur at a frequency of <0.1%.7
Although it varies by practice and location, the cost for an acute course of TMS (20 to 30 sessions) may range from $6,000 to $12,000.8 Most insurance companies cover TMS treatment for depression.
Continue to: TMS
TMS: A more accessible option
Compared with DBS, TMS is a more affordable and accessible therapy for patients with TRD. Further studies are needed to learn more about the therapeutic potential of DBS for TRD, and to develop methods that help decrease the risk of adverse effects. In addition, insurance coverage needs to be expanded to DBS to avoid having patients be responsible for the full costs of this treatment. Until then, TMS should be a recommended therapy for patients with TRD. If TRD persists in patients treated with TMS, consider electroconvulsive therapy.
1. Morishita T, Fayad SM, Higuchi MA, et al. Deep brain stimulation for treatment-resistant depression: systematic review of clinical outcomes. Neurotherapeutics. 2014;11(3):475-484.
2. Lawrence RE, Kaufmann CR, DeSilva RB, et al. Patients’ belief about deep brain stimulation for treatment resistant depression. AJOB Neuroscience, 2018;9(4):210-218.
3. Rossi PJ, Giordano J, Okun MS. The problem of funding off-label deep brain stimulation: bait-and-switch tactics and the need for policy reform. JAMA Neurol. 2017;74(1):9-10.
4. Holtzheimer PE, Husain MM, Lisanby SH, et al. Subcallosal cingulate deep brain stimulation for treatment-resistant depression: a multisite, randomised, sham-controlled trial. Lancet Psychiatry. 2017;4(11):839-849.
5. Janicak PG. What’s new in transcranial magnetic stimulation. Current Psychiatry. 2019;18(3):10-16.
6. Janicak PG, Sackett V, Kudrna K, et al. Advances in transcranial magnetic stimulation for managing major depressive disorders. Current Psychiatry. 2016;15(6):49-56.
7. Dobek CE, Blumberger DM, Downar J, et al. Risk of seizures in transcranial magnetic stimulation: a clinical review to inform consent process focused on bupropion. Neuropsychiatr Dis Treat. 2015;11:2975-2987.
8. McClintock SM, Reti IM, Carpenter LL, et al; National Network of Depression Centers rTMS Task Group; American Psychiatric Association Council on Research Task Force on Novel Biomarkers and Treatments. Consensus recommendations for the clinical application of repetitive transcranial magnetic stimulation (rTMS) in the treatment of depression. J Clin Psychiatry. 2018;79(1). doi: 10.4088/JCP.16cs10905.
1. Morishita T, Fayad SM, Higuchi MA, et al. Deep brain stimulation for treatment-resistant depression: systematic review of clinical outcomes. Neurotherapeutics. 2014;11(3):475-484.
2. Lawrence RE, Kaufmann CR, DeSilva RB, et al. Patients’ belief about deep brain stimulation for treatment resistant depression. AJOB Neuroscience, 2018;9(4):210-218.
3. Rossi PJ, Giordano J, Okun MS. The problem of funding off-label deep brain stimulation: bait-and-switch tactics and the need for policy reform. JAMA Neurol. 2017;74(1):9-10.
4. Holtzheimer PE, Husain MM, Lisanby SH, et al. Subcallosal cingulate deep brain stimulation for treatment-resistant depression: a multisite, randomised, sham-controlled trial. Lancet Psychiatry. 2017;4(11):839-849.
5. Janicak PG. What’s new in transcranial magnetic stimulation. Current Psychiatry. 2019;18(3):10-16.
6. Janicak PG, Sackett V, Kudrna K, et al. Advances in transcranial magnetic stimulation for managing major depressive disorders. Current Psychiatry. 2016;15(6):49-56.
7. Dobek CE, Blumberger DM, Downar J, et al. Risk of seizures in transcranial magnetic stimulation: a clinical review to inform consent process focused on bupropion. Neuropsychiatr Dis Treat. 2015;11:2975-2987.
8. McClintock SM, Reti IM, Carpenter LL, et al; National Network of Depression Centers rTMS Task Group; American Psychiatric Association Council on Research Task Force on Novel Biomarkers and Treatments. Consensus recommendations for the clinical application of repetitive transcranial magnetic stimulation (rTMS) in the treatment of depression. J Clin Psychiatry. 2018;79(1). doi: 10.4088/JCP.16cs10905.
Sick, or faking it?
CASE Vague symptoms; no clear etiology
Mr. W, age 53, presents to the emergency department (ED) describing acute mid-sternal chest pain (severity: 8 out of 10). His medical history is significant for pulmonary embolism and ascending aortic aneurysm in the context of Takayasu’s arteritis, an inflammatory condition of the large arterial blood vessels characterized by lesions that can lead to vascular stenosis, occlusion, or aneurysm. Takayasu’s arteritis is also known as pulseless disease due to the weak or absent pulses the condition produces.
A review of Mr. W’s medical records reveals that this is his 23rd visit to this hospital within a year; the year before that, he had 22 visits. At each of these previous visits, he had similar vague symptoms, including dizziness, chest pain, lightheadedness, fainting, bilateral knee weakness, and left-arm numbness/weakness, and no clear acute etiology for his reported symptoms. Each time, after the treating clinicians ruled out possible acute complications of a flare-up of Takayasu’s arteritis through a physical examination, laboratory tests, and imaging studies, Mr. W was discharged with recommendations that he follow-up with his primary care physician and specialists. At each discharge, he would leave the hospital with hesitation.
At this present visit, the ED physician recognizes Mr. W as someone who visits the ED often with no profound acute issues, and reviews the substantial medical records available to the hospital. He suspects Mr. W is feigning symptoms, and orders a psychiatric consultation.
EVALUATION Psychiatric interview and mental status exam
On examination, Mr. W is not in acute distress. Despite reporting an 8 out of 10 for chest pain severity, he displays no psychomotor agitation, and his pulse rate and blood pressure are within normal limits. He makes appropriate eye contact and describes his mood as “great.” He reports no problems with sleep, appetite, or disinterest in pleasurable activities, and denies being depressed or having any symptoms consistent with a mood disorder, anxiety disorder, or psychosis. He denies a history of panic attacks or excessive worrying that interferes with his sleep or activities of daily living. Additionally, Mr. W describes a stable, peaceful, and stress-free life within the limitations of his Takayasu’s arteritis, which he has been managing well since his diagnosis 6 years earlier.
Mr. W denies having any psychiatric symptoms, apprehensive feelings, or beliefs/fears that would be considered delusional, and he has no previous legal issues aside from an occasional driving citation. During the assessment, his affect remains broad and he denies having thoughts of suicide or homicide, or auditory or visual hallucinations.
Mr. W’s drug screen results are negative, and he denies using any illicit drugs. He uses only the medications that are prescribed by his clinicians. Overall, he seems to be a well-functioning individual. Mr. W reports that work is generally not stressful.
When the psychiatric team asks him about his frequent hospitalizations and ED visits, Mr. W is insistent that he is “just doing what my doctors said for me to do.” He repeats that he does not have any mental illness and did not see the point of seeing a psychiatrist.
In pursuit of collateral information, the psychiatry team accesses a regional medical record database that allows registered medical institutions and practices to track patients’ medical encounters within the region. According to this database, within approximately 5.5 years, Mr. W had 163 clinical encounters (ED visits and inpatient admissions) and 376 radiological studies in our region (Table 1 and Table 2).
[polldaddy:10394110]
Continue to: The authors' observations
The authors’ observations
The psychiatry team’s investigation of Mr. W’s medical records revealed the extent of his care-seeking behavior, and provided evidence for a diagnosis of factitious disorder.
Factitious disorder is an elusive psychiatric condition in which an individual chronically stimulates, induces, or aggravates illnesses to gain the status of being a patient. Although its exact cause has not been fully deciphered, it is seen mostly among individuals with knowledge of the workings of the medical field, such as a health care worker.1 Factitious disorder is taxing on the health care system, with an estimated cost in the thousands of dollars per patient visit.2 The condition has an estimated prevalence of 0.8% to 1.0% of patients seen by psychiatric consult services3 and is reported to be more prevalent among women than men.1 Its cardinal features include health care site hopping and hospital shopping, vagueness about the patient’s history and symptoms, and discrepancy among reported symptoms, the patient’s behaviors, and objective clinical findings.4,5 Although not all patients with factitious disorder have a legitimate medical reason for seeking care, some individuals with an established medical diagnosis use their condition as a tool to chronically seek care and play the sick role.
Factitious disorder should not be confused with malingering, which is differentiated by the patient’s search for a secondary gain, such as financial reward or avoiding jail; or conversion disorder, which is marked by true physical or neurologic symptoms and clinical findings triggered by psychological stressors. Patients with factitious disorder usually are cooperative during hospital stays and resume their normal daily routine shortly after discharge.4 In this case, Mr. W denied any psychiatric symptoms, apprehensive feelings, or beliefs or fears that would be considered delusional. He had no previous or pending legal issues, which ruled out malingering to avoid legal repercussions.
Mr. W’s presentation was complicated by his Takayasu’s arteritis diagnosis. Because Takayasu’s arteritis has a serious list of potential complications, ED physicians have a low threshold for ordering diagnostic studies for a patient with Takayasu’s arteritis who presents with a chief complaint of chest pain. In other words, when a patient with this condition presents to an acute setting (such as the ED) with chest pain, his/her chief complaint is taken with extreme seriousness. Conventional angiography is the standard diagnostic tool for Takayasu’s arteritis; CT angiography and magnetic resonance angiography are used for monitoring the disease’s progression.6
[polldaddy:10394113]
The authors’ observations
Currently, there are no FDA-approved treatments for factitious disorder, and patients with this condition generally are resistant to psychiatric and/or psychological care when discovered and offered treatment.7 Among those who consent to psychiatric care, psychoeducation, or psychotherapy, which have shown some efficacy for the condition, the dropout rate is high.8
Continue to: Although the instinctive approach...
Although the instinctive approach is to confront the patient once the deception has been uncovered, expert recommendations are contradictory. Some recommend confrontation as part of a treatment protocol,8 while others advise against such an approach.9
Because of how often patients with factitious disorder seek medical care, secondary iatrogenic consequences are possible. For example, for years, Mr. W has been unknowingly exposing himself to the iatrogenic consequences of the cumulative effect of diagnostic imaging for years. In 3 years alone, Mr. W had undergone an average of 125 diagnostic imaging studies per year—with and without contrast—and many unnecessary rounds of treatment with steroids and other interventions known to have secondary iatrogenic consequences.10 Excessive radiation exposure is known to be carcinogenic over time,10 and excessive use of steroids is associated with weight gain, physical habitus changes, and increased risk of infections.11 In addition, the renal effects of the contrast materials from repeated imaging studies over so many years on Mr. W’s future kidney function are unknown.
TREATMENT Psychoeducation and referral for psychotherapy
We counsel Mr. W about factitious disorder and the risks of excessive hospitalizations, and refer him for follow-up at our local psychiatric clinic, as well as for individual psychotherapy. Mr. W is discharged because his medical work-up does not reveal any significant acute medical issues.
The authors’ observations
Because of the poor insight associated with factitious disorder and the limited treatment options available, a patient with factitious disorder is unlikely to enter psychiatric treatment on his/her own. The prognosis for a patient with factitious disorder remains poor unless the patient is forced into treatment. More intervention-focused research is needed to help improve outcomes for patients with factitious disorder.
OUTCOME Failure to follow up
Mr. W fails to attend individual psychotherapy as recommended. According to our regional record database, Mr. W continues to present to other EDs regularly.
Continue to: Bottom Line
Bottom Line
A patient with factitious disorder stimulates, induces, or aggravates illnesses to gain the status of being a patient. Treatment options include psychiatric care, psychoeducation, or psychotherapy. However, due to poor insight, a patient with factitious disorder is unlikely to enter psychiatric treatment on his/her own.
Related Resources
- Yates GP, Feldman MD. Factitious disorder: a systematic review of 455 cases in the professional literature. Gen Hosp Psychiatry. 2016;41:20-28.
- Galli S, Tatu L, Bogousslavsky J, et al. Conversion, factitious disorder and malingering: a distinct pattern or a continuum? Front Neurol Neurosci. 2018;42:72-80.
1. Krahn LE, Li H, O’Connor MK. Patients who strive to be ill: factitious disorder with physical symptoms. Am J Psychiatry. 2003;160(6):1163-1168.
2. Hoertel N, Lavaud P, Le Strat Y, et al. Estimated cost of a factitious disorder with 6-year follow-up. Psychiatry Res. 2012;200(2):1077-1078.
3. Sadock BJ, Sadock VA, Ruiz P. Psychosomatic medicine; factitious disorder. In: Pataki CS, Sussman N, eds. Synopsis of psychiatry: Behavioral sciences/clinical psychiatry. 11th ed. Philadelphia, PA: Wolters Kluwer; 2015:34-45.
4 . Savino AC, Fordtran JS. Factitious disease: clinical lessons from case studies at Baylor University Medical Center. Proc (Bayl Univ Med Cent). 2006;19(3):195-208.
5. Burnel A. Recognition and management of factitious disorder. Prescriber. 2015;26(21):37-39.
6. Duftner C, Dejaco C, Sepriano A, et al. Imaging in diagnosis, outcome prediction and monitoring of large vessel vasculitis: a systematic literature review and meta-analysis informing the EULAR recommendations. RMD Open. 2018;4(1):e000612. doi: 10.1136/rmdopen-2017-000612.
7. Jafferany M, Khalid Z, McDonald KA, et al. Psychological aspects of factitious disorder. Prim Care Companion CNS Disord. 2018;20(1). doi: 10.4088/PCC.17nr02229.
8. Bolat N, Yalçin O. Factitious disorder presenting with stuttering in two adolescents: the importance of psychoeducation. Noro Psikiyatri Arsivi. 2017;54(1):87-89.
9. Eisendrath SJ. Factitious physical disorders. West J Med. 1994;160(2):177-179.
10. Sodickson A, Baeyens PF, Andriole KP, et al. Recurrent CT, cumulative radiation exposure, and associated radiation-induced cancer risks from CT of adults. Radiology. 2009;251(1):175-184.
11. Oray M, Abu Samra K, Ebrahimiadib N, et al. Long-term side effects of glucocorticoids. Expert Opin Drug Saf. 2016;15(4):457-465.
CASE Vague symptoms; no clear etiology
Mr. W, age 53, presents to the emergency department (ED) describing acute mid-sternal chest pain (severity: 8 out of 10). His medical history is significant for pulmonary embolism and ascending aortic aneurysm in the context of Takayasu’s arteritis, an inflammatory condition of the large arterial blood vessels characterized by lesions that can lead to vascular stenosis, occlusion, or aneurysm. Takayasu’s arteritis is also known as pulseless disease due to the weak or absent pulses the condition produces.
A review of Mr. W’s medical records reveals that this is his 23rd visit to this hospital within a year; the year before that, he had 22 visits. At each of these previous visits, he had similar vague symptoms, including dizziness, chest pain, lightheadedness, fainting, bilateral knee weakness, and left-arm numbness/weakness, and no clear acute etiology for his reported symptoms. Each time, after the treating clinicians ruled out possible acute complications of a flare-up of Takayasu’s arteritis through a physical examination, laboratory tests, and imaging studies, Mr. W was discharged with recommendations that he follow-up with his primary care physician and specialists. At each discharge, he would leave the hospital with hesitation.
At this present visit, the ED physician recognizes Mr. W as someone who visits the ED often with no profound acute issues, and reviews the substantial medical records available to the hospital. He suspects Mr. W is feigning symptoms, and orders a psychiatric consultation.
EVALUATION Psychiatric interview and mental status exam
On examination, Mr. W is not in acute distress. Despite reporting an 8 out of 10 for chest pain severity, he displays no psychomotor agitation, and his pulse rate and blood pressure are within normal limits. He makes appropriate eye contact and describes his mood as “great.” He reports no problems with sleep, appetite, or disinterest in pleasurable activities, and denies being depressed or having any symptoms consistent with a mood disorder, anxiety disorder, or psychosis. He denies a history of panic attacks or excessive worrying that interferes with his sleep or activities of daily living. Additionally, Mr. W describes a stable, peaceful, and stress-free life within the limitations of his Takayasu’s arteritis, which he has been managing well since his diagnosis 6 years earlier.
Mr. W denies having any psychiatric symptoms, apprehensive feelings, or beliefs/fears that would be considered delusional, and he has no previous legal issues aside from an occasional driving citation. During the assessment, his affect remains broad and he denies having thoughts of suicide or homicide, or auditory or visual hallucinations.
Mr. W’s drug screen results are negative, and he denies using any illicit drugs. He uses only the medications that are prescribed by his clinicians. Overall, he seems to be a well-functioning individual. Mr. W reports that work is generally not stressful.
When the psychiatric team asks him about his frequent hospitalizations and ED visits, Mr. W is insistent that he is “just doing what my doctors said for me to do.” He repeats that he does not have any mental illness and did not see the point of seeing a psychiatrist.
In pursuit of collateral information, the psychiatry team accesses a regional medical record database that allows registered medical institutions and practices to track patients’ medical encounters within the region. According to this database, within approximately 5.5 years, Mr. W had 163 clinical encounters (ED visits and inpatient admissions) and 376 radiological studies in our region (Table 1 and Table 2).
[polldaddy:10394110]
Continue to: The authors' observations
The authors’ observations
The psychiatry team’s investigation of Mr. W’s medical records revealed the extent of his care-seeking behavior, and provided evidence for a diagnosis of factitious disorder.
Factitious disorder is an elusive psychiatric condition in which an individual chronically stimulates, induces, or aggravates illnesses to gain the status of being a patient. Although its exact cause has not been fully deciphered, it is seen mostly among individuals with knowledge of the workings of the medical field, such as a health care worker.1 Factitious disorder is taxing on the health care system, with an estimated cost in the thousands of dollars per patient visit.2 The condition has an estimated prevalence of 0.8% to 1.0% of patients seen by psychiatric consult services3 and is reported to be more prevalent among women than men.1 Its cardinal features include health care site hopping and hospital shopping, vagueness about the patient’s history and symptoms, and discrepancy among reported symptoms, the patient’s behaviors, and objective clinical findings.4,5 Although not all patients with factitious disorder have a legitimate medical reason for seeking care, some individuals with an established medical diagnosis use their condition as a tool to chronically seek care and play the sick role.
Factitious disorder should not be confused with malingering, which is differentiated by the patient’s search for a secondary gain, such as financial reward or avoiding jail; or conversion disorder, which is marked by true physical or neurologic symptoms and clinical findings triggered by psychological stressors. Patients with factitious disorder usually are cooperative during hospital stays and resume their normal daily routine shortly after discharge.4 In this case, Mr. W denied any psychiatric symptoms, apprehensive feelings, or beliefs or fears that would be considered delusional. He had no previous or pending legal issues, which ruled out malingering to avoid legal repercussions.
Mr. W’s presentation was complicated by his Takayasu’s arteritis diagnosis. Because Takayasu’s arteritis has a serious list of potential complications, ED physicians have a low threshold for ordering diagnostic studies for a patient with Takayasu’s arteritis who presents with a chief complaint of chest pain. In other words, when a patient with this condition presents to an acute setting (such as the ED) with chest pain, his/her chief complaint is taken with extreme seriousness. Conventional angiography is the standard diagnostic tool for Takayasu’s arteritis; CT angiography and magnetic resonance angiography are used for monitoring the disease’s progression.6
[polldaddy:10394113]
The authors’ observations
Currently, there are no FDA-approved treatments for factitious disorder, and patients with this condition generally are resistant to psychiatric and/or psychological care when discovered and offered treatment.7 Among those who consent to psychiatric care, psychoeducation, or psychotherapy, which have shown some efficacy for the condition, the dropout rate is high.8
Continue to: Although the instinctive approach...
Although the instinctive approach is to confront the patient once the deception has been uncovered, expert recommendations are contradictory. Some recommend confrontation as part of a treatment protocol,8 while others advise against such an approach.9
Because of how often patients with factitious disorder seek medical care, secondary iatrogenic consequences are possible. For example, for years, Mr. W has been unknowingly exposing himself to the iatrogenic consequences of the cumulative effect of diagnostic imaging for years. In 3 years alone, Mr. W had undergone an average of 125 diagnostic imaging studies per year—with and without contrast—and many unnecessary rounds of treatment with steroids and other interventions known to have secondary iatrogenic consequences.10 Excessive radiation exposure is known to be carcinogenic over time,10 and excessive use of steroids is associated with weight gain, physical habitus changes, and increased risk of infections.11 In addition, the renal effects of the contrast materials from repeated imaging studies over so many years on Mr. W’s future kidney function are unknown.
TREATMENT Psychoeducation and referral for psychotherapy
We counsel Mr. W about factitious disorder and the risks of excessive hospitalizations, and refer him for follow-up at our local psychiatric clinic, as well as for individual psychotherapy. Mr. W is discharged because his medical work-up does not reveal any significant acute medical issues.
The authors’ observations
Because of the poor insight associated with factitious disorder and the limited treatment options available, a patient with factitious disorder is unlikely to enter psychiatric treatment on his/her own. The prognosis for a patient with factitious disorder remains poor unless the patient is forced into treatment. More intervention-focused research is needed to help improve outcomes for patients with factitious disorder.
OUTCOME Failure to follow up
Mr. W fails to attend individual psychotherapy as recommended. According to our regional record database, Mr. W continues to present to other EDs regularly.
Continue to: Bottom Line
Bottom Line
A patient with factitious disorder stimulates, induces, or aggravates illnesses to gain the status of being a patient. Treatment options include psychiatric care, psychoeducation, or psychotherapy. However, due to poor insight, a patient with factitious disorder is unlikely to enter psychiatric treatment on his/her own.
Related Resources
- Yates GP, Feldman MD. Factitious disorder: a systematic review of 455 cases in the professional literature. Gen Hosp Psychiatry. 2016;41:20-28.
- Galli S, Tatu L, Bogousslavsky J, et al. Conversion, factitious disorder and malingering: a distinct pattern or a continuum? Front Neurol Neurosci. 2018;42:72-80.
CASE Vague symptoms; no clear etiology
Mr. W, age 53, presents to the emergency department (ED) describing acute mid-sternal chest pain (severity: 8 out of 10). His medical history is significant for pulmonary embolism and ascending aortic aneurysm in the context of Takayasu’s arteritis, an inflammatory condition of the large arterial blood vessels characterized by lesions that can lead to vascular stenosis, occlusion, or aneurysm. Takayasu’s arteritis is also known as pulseless disease due to the weak or absent pulses the condition produces.
A review of Mr. W’s medical records reveals that this is his 23rd visit to this hospital within a year; the year before that, he had 22 visits. At each of these previous visits, he had similar vague symptoms, including dizziness, chest pain, lightheadedness, fainting, bilateral knee weakness, and left-arm numbness/weakness, and no clear acute etiology for his reported symptoms. Each time, after the treating clinicians ruled out possible acute complications of a flare-up of Takayasu’s arteritis through a physical examination, laboratory tests, and imaging studies, Mr. W was discharged with recommendations that he follow-up with his primary care physician and specialists. At each discharge, he would leave the hospital with hesitation.
At this present visit, the ED physician recognizes Mr. W as someone who visits the ED often with no profound acute issues, and reviews the substantial medical records available to the hospital. He suspects Mr. W is feigning symptoms, and orders a psychiatric consultation.
EVALUATION Psychiatric interview and mental status exam
On examination, Mr. W is not in acute distress. Despite reporting an 8 out of 10 for chest pain severity, he displays no psychomotor agitation, and his pulse rate and blood pressure are within normal limits. He makes appropriate eye contact and describes his mood as “great.” He reports no problems with sleep, appetite, or disinterest in pleasurable activities, and denies being depressed or having any symptoms consistent with a mood disorder, anxiety disorder, or psychosis. He denies a history of panic attacks or excessive worrying that interferes with his sleep or activities of daily living. Additionally, Mr. W describes a stable, peaceful, and stress-free life within the limitations of his Takayasu’s arteritis, which he has been managing well since his diagnosis 6 years earlier.
Mr. W denies having any psychiatric symptoms, apprehensive feelings, or beliefs/fears that would be considered delusional, and he has no previous legal issues aside from an occasional driving citation. During the assessment, his affect remains broad and he denies having thoughts of suicide or homicide, or auditory or visual hallucinations.
Mr. W’s drug screen results are negative, and he denies using any illicit drugs. He uses only the medications that are prescribed by his clinicians. Overall, he seems to be a well-functioning individual. Mr. W reports that work is generally not stressful.
When the psychiatric team asks him about his frequent hospitalizations and ED visits, Mr. W is insistent that he is “just doing what my doctors said for me to do.” He repeats that he does not have any mental illness and did not see the point of seeing a psychiatrist.
In pursuit of collateral information, the psychiatry team accesses a regional medical record database that allows registered medical institutions and practices to track patients’ medical encounters within the region. According to this database, within approximately 5.5 years, Mr. W had 163 clinical encounters (ED visits and inpatient admissions) and 376 radiological studies in our region (Table 1 and Table 2).
[polldaddy:10394110]
Continue to: The authors' observations
The authors’ observations
The psychiatry team’s investigation of Mr. W’s medical records revealed the extent of his care-seeking behavior, and provided evidence for a diagnosis of factitious disorder.
Factitious disorder is an elusive psychiatric condition in which an individual chronically stimulates, induces, or aggravates illnesses to gain the status of being a patient. Although its exact cause has not been fully deciphered, it is seen mostly among individuals with knowledge of the workings of the medical field, such as a health care worker.1 Factitious disorder is taxing on the health care system, with an estimated cost in the thousands of dollars per patient visit.2 The condition has an estimated prevalence of 0.8% to 1.0% of patients seen by psychiatric consult services3 and is reported to be more prevalent among women than men.1 Its cardinal features include health care site hopping and hospital shopping, vagueness about the patient’s history and symptoms, and discrepancy among reported symptoms, the patient’s behaviors, and objective clinical findings.4,5 Although not all patients with factitious disorder have a legitimate medical reason for seeking care, some individuals with an established medical diagnosis use their condition as a tool to chronically seek care and play the sick role.
Factitious disorder should not be confused with malingering, which is differentiated by the patient’s search for a secondary gain, such as financial reward or avoiding jail; or conversion disorder, which is marked by true physical or neurologic symptoms and clinical findings triggered by psychological stressors. Patients with factitious disorder usually are cooperative during hospital stays and resume their normal daily routine shortly after discharge.4 In this case, Mr. W denied any psychiatric symptoms, apprehensive feelings, or beliefs or fears that would be considered delusional. He had no previous or pending legal issues, which ruled out malingering to avoid legal repercussions.
Mr. W’s presentation was complicated by his Takayasu’s arteritis diagnosis. Because Takayasu’s arteritis has a serious list of potential complications, ED physicians have a low threshold for ordering diagnostic studies for a patient with Takayasu’s arteritis who presents with a chief complaint of chest pain. In other words, when a patient with this condition presents to an acute setting (such as the ED) with chest pain, his/her chief complaint is taken with extreme seriousness. Conventional angiography is the standard diagnostic tool for Takayasu’s arteritis; CT angiography and magnetic resonance angiography are used for monitoring the disease’s progression.6
[polldaddy:10394113]
The authors’ observations
Currently, there are no FDA-approved treatments for factitious disorder, and patients with this condition generally are resistant to psychiatric and/or psychological care when discovered and offered treatment.7 Among those who consent to psychiatric care, psychoeducation, or psychotherapy, which have shown some efficacy for the condition, the dropout rate is high.8
Continue to: Although the instinctive approach...
Although the instinctive approach is to confront the patient once the deception has been uncovered, expert recommendations are contradictory. Some recommend confrontation as part of a treatment protocol,8 while others advise against such an approach.9
Because of how often patients with factitious disorder seek medical care, secondary iatrogenic consequences are possible. For example, for years, Mr. W has been unknowingly exposing himself to the iatrogenic consequences of the cumulative effect of diagnostic imaging for years. In 3 years alone, Mr. W had undergone an average of 125 diagnostic imaging studies per year—with and without contrast—and many unnecessary rounds of treatment with steroids and other interventions known to have secondary iatrogenic consequences.10 Excessive radiation exposure is known to be carcinogenic over time,10 and excessive use of steroids is associated with weight gain, physical habitus changes, and increased risk of infections.11 In addition, the renal effects of the contrast materials from repeated imaging studies over so many years on Mr. W’s future kidney function are unknown.
TREATMENT Psychoeducation and referral for psychotherapy
We counsel Mr. W about factitious disorder and the risks of excessive hospitalizations, and refer him for follow-up at our local psychiatric clinic, as well as for individual psychotherapy. Mr. W is discharged because his medical work-up does not reveal any significant acute medical issues.
The authors’ observations
Because of the poor insight associated with factitious disorder and the limited treatment options available, a patient with factitious disorder is unlikely to enter psychiatric treatment on his/her own. The prognosis for a patient with factitious disorder remains poor unless the patient is forced into treatment. More intervention-focused research is needed to help improve outcomes for patients with factitious disorder.
OUTCOME Failure to follow up
Mr. W fails to attend individual psychotherapy as recommended. According to our regional record database, Mr. W continues to present to other EDs regularly.
Continue to: Bottom Line
Bottom Line
A patient with factitious disorder stimulates, induces, or aggravates illnesses to gain the status of being a patient. Treatment options include psychiatric care, psychoeducation, or psychotherapy. However, due to poor insight, a patient with factitious disorder is unlikely to enter psychiatric treatment on his/her own.
Related Resources
- Yates GP, Feldman MD. Factitious disorder: a systematic review of 455 cases in the professional literature. Gen Hosp Psychiatry. 2016;41:20-28.
- Galli S, Tatu L, Bogousslavsky J, et al. Conversion, factitious disorder and malingering: a distinct pattern or a continuum? Front Neurol Neurosci. 2018;42:72-80.
1. Krahn LE, Li H, O’Connor MK. Patients who strive to be ill: factitious disorder with physical symptoms. Am J Psychiatry. 2003;160(6):1163-1168.
2. Hoertel N, Lavaud P, Le Strat Y, et al. Estimated cost of a factitious disorder with 6-year follow-up. Psychiatry Res. 2012;200(2):1077-1078.
3. Sadock BJ, Sadock VA, Ruiz P. Psychosomatic medicine; factitious disorder. In: Pataki CS, Sussman N, eds. Synopsis of psychiatry: Behavioral sciences/clinical psychiatry. 11th ed. Philadelphia, PA: Wolters Kluwer; 2015:34-45.
4 . Savino AC, Fordtran JS. Factitious disease: clinical lessons from case studies at Baylor University Medical Center. Proc (Bayl Univ Med Cent). 2006;19(3):195-208.
5. Burnel A. Recognition and management of factitious disorder. Prescriber. 2015;26(21):37-39.
6. Duftner C, Dejaco C, Sepriano A, et al. Imaging in diagnosis, outcome prediction and monitoring of large vessel vasculitis: a systematic literature review and meta-analysis informing the EULAR recommendations. RMD Open. 2018;4(1):e000612. doi: 10.1136/rmdopen-2017-000612.
7. Jafferany M, Khalid Z, McDonald KA, et al. Psychological aspects of factitious disorder. Prim Care Companion CNS Disord. 2018;20(1). doi: 10.4088/PCC.17nr02229.
8. Bolat N, Yalçin O. Factitious disorder presenting with stuttering in two adolescents: the importance of psychoeducation. Noro Psikiyatri Arsivi. 2017;54(1):87-89.
9. Eisendrath SJ. Factitious physical disorders. West J Med. 1994;160(2):177-179.
10. Sodickson A, Baeyens PF, Andriole KP, et al. Recurrent CT, cumulative radiation exposure, and associated radiation-induced cancer risks from CT of adults. Radiology. 2009;251(1):175-184.
11. Oray M, Abu Samra K, Ebrahimiadib N, et al. Long-term side effects of glucocorticoids. Expert Opin Drug Saf. 2016;15(4):457-465.
1. Krahn LE, Li H, O’Connor MK. Patients who strive to be ill: factitious disorder with physical symptoms. Am J Psychiatry. 2003;160(6):1163-1168.
2. Hoertel N, Lavaud P, Le Strat Y, et al. Estimated cost of a factitious disorder with 6-year follow-up. Psychiatry Res. 2012;200(2):1077-1078.
3. Sadock BJ, Sadock VA, Ruiz P. Psychosomatic medicine; factitious disorder. In: Pataki CS, Sussman N, eds. Synopsis of psychiatry: Behavioral sciences/clinical psychiatry. 11th ed. Philadelphia, PA: Wolters Kluwer; 2015:34-45.
4 . Savino AC, Fordtran JS. Factitious disease: clinical lessons from case studies at Baylor University Medical Center. Proc (Bayl Univ Med Cent). 2006;19(3):195-208.
5. Burnel A. Recognition and management of factitious disorder. Prescriber. 2015;26(21):37-39.
6. Duftner C, Dejaco C, Sepriano A, et al. Imaging in diagnosis, outcome prediction and monitoring of large vessel vasculitis: a systematic literature review and meta-analysis informing the EULAR recommendations. RMD Open. 2018;4(1):e000612. doi: 10.1136/rmdopen-2017-000612.
7. Jafferany M, Khalid Z, McDonald KA, et al. Psychological aspects of factitious disorder. Prim Care Companion CNS Disord. 2018;20(1). doi: 10.4088/PCC.17nr02229.
8. Bolat N, Yalçin O. Factitious disorder presenting with stuttering in two adolescents: the importance of psychoeducation. Noro Psikiyatri Arsivi. 2017;54(1):87-89.
9. Eisendrath SJ. Factitious physical disorders. West J Med. 1994;160(2):177-179.
10. Sodickson A, Baeyens PF, Andriole KP, et al. Recurrent CT, cumulative radiation exposure, and associated radiation-induced cancer risks from CT of adults. Radiology. 2009;251(1):175-184.
11. Oray M, Abu Samra K, Ebrahimiadib N, et al. Long-term side effects of glucocorticoids. Expert Opin Drug Saf. 2016;15(4):457-465.