User login
Guardian angel or watchdog? Pills of capecitabine contain sensors
Recently, a woman with advanced colorectal cancer at the University of Minnesota, Minneapolis, was taking capecitabine (Xeloda) in the morning but skipping her evening dose.
Her hands hurt, and she couldn’t open the childproof cap. Her daughter had been doing it for her in the morning but wasn’t around to help out at night.
It was the kind of problem that might have gone on for days or weeks until the next clinic visit, and, even then, be addressed only if the woman remembered to mention it.
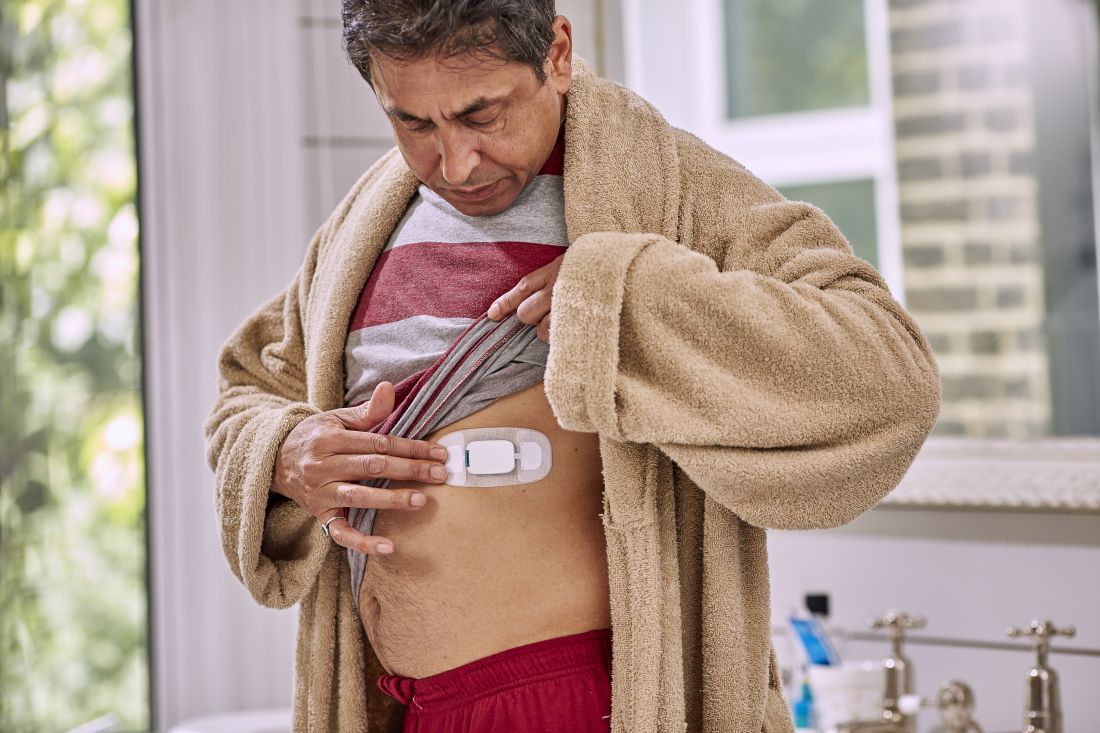
But that’s not what happened. The care team realized pretty much right away that she was skipping the p.m. dose because the woman was taking her capecitabine in a gel cap with an adherence sensor.
The sensor, a sandwich of copper, silicon, and magnesium in a millimeter square, sent out an electric ping when she took her dose, activated by stomach acid; the ping was picked up by an adhesive bandage patch the woman wore, which relayed the signal to an app on her smartphone; the phone passed it on to a server cloud that the woman had given her providers permission to access.
They monitored her adherence on a Web portal, along with heart rate and activity data, also captured by the patch. The system is called Proteus Discover, from Proteus Digital Health.
Instead of taking days or weeks, the care team quickly realized that she wasn’t taking her evening dose of capecitabine. They contacted her, replaced the childproof cap, and twice-daily dosing resumed.
Expanded use in oncology?
Seven other advanced colorectal cancer patients have participated in the University of Minnesota (UM) pilot project, the first use of the device in oncology. “It’s gone so well that it’s annoying to me to not have this for all my patients. I’m already feeling frustrated that I can’t just put this in all the drugs that I give orally,” said Edward Greeno, MD, an oncologist/hematologist at the university, and the Proteus point man.
“You would assume that cancer patients would be hypercompliant, but it turns out they have the same compliance problems” as other patients, plus additional hurdles, he said, including complex regimens and drug toxicity.
“Every patient we have approached so far has been enthusiastic. They might be a little bit annoyed that I know they haven’t been taking their pills, but they are also glad that I am there to hold them responsible and help them. My intention is to roll this out much more broadly,” he said. That just might happen. Dr. Greeno is working with Proteus to roll the sensor out at UM and oncology programs elsewhere.
A slow, careful rollout
The device was cleared by the Food and Drug Administration in 2012. After more than 180,000 ingestions, there have been no safety issues, besides occasional skin irritation from the patch, which is waterproof and meant to be worn for a week, then replaced. It pings if it’s taken off. The sensor is passes through the body like food.
Patients can communicate with providers over the phone app, which also sends reminders when it’s time to take the next pill.
So far, Proteus has worked with ten health systems in the United States, and more are in the works. Commercialization efforts have focused mostly on blood pressure, cholesterol, and type 2 diabetes drugs, the bad boys of drug adherence, but the system has also been piloted for hepatitis C treatment, and trials are underway for HIV preexposure prophylaxis.
Among the company’s many favorable studies, the system has already been demonstrated to be a viable alternative to directly observed therapy in tuberculosis, the current gold-standard, but hugely labor and resource intensive (PLoS One. 2013;8[1]:e53373. doi: 10.1371/journal.pone.0053373).
Proteus can’t be picked up at the local Walgreens. The company works closely with clients and is being careful in its rollout. For one thing, each sensor has to be programed for the specific drug it’s being used with, but also, and as with any new technology, business and payment models are still being worked out.
UM’s partner in the oncology project, Fairview Health Services, pays Proteus when patients hit an adherence rate of 80%, but how much they pay is a proprietary secret. “Most of the cost issue is still not in the public domain,” said Scooter Plowman, MD, the company’s medical director.
In 2017, FDA approved a version of the antipsychotic aripiprazole embedded with the Proteus sensor. The rollout of “Abilify MyCite” by Otsuka Pharmaceuticals has been similarly cautious, under contract with health systems.
“Otsuka has been very smart in the approach they are taking,” Dr. Plowman said.
He wouldn’t give details, but Proteus is in talks with other pharmaceutical makers to bring pills with sensors to the market.
A new fix for an old problem
Proteus isn’t alone in the ingestible event marker (IEM) market. The FDA is reviewing a rival sensor from etectRx, in Gainesville, Fla.
The technology is a little different; the etectRx sensor is a microchip made out of magnesium and silver chloride that’s embedded on the inside of a gel cap. Instead of an electric blip, it sends out a radio wave when activated by stomach acid. It’s larger than the Proteus offering, but still has room to spare in a gel cap.
The signal is stronger, so patients wear a neck pendant instead of a patch to pick it up. The pendant does not capture heart rate or activity data. It’s not meant to be worn continuously and can come off after it pings the system.
The two systems are otherwise similar; etectRx also uses a phone app to relay adherence data to a server cloud clinicians can access, with patient permission. As with Proteus, everything works as long as the phone is on. President and CEO Harry Travis anticipates clearance in 2019.
Adherence is a huge and well-known problem in medicine; only about half of patients take medications as they are prescribed. People end up in the ED or the hospital with problems that might have been avoided. Providers and payers want solutions.
Industry is bringing technology to bear on the problem. The payoff will be huge for the winners; analysts project multiple billion dollar growth in the adherence technology sector.
Most companies, however, are pinning their hopes on indirect approaches, bottle caps that ping when opened, for instance, or coaching apps for smart phones. IEMs seem to be ahead of the curve.
Guardian angel or watchdog?
Whether that’s a good thing or bad thing depends on who you talk to, but patients do seem more likely to take their medications if a sensor is on board.
Dr. Plowman said he thinks IEMs improve adherence because, with their own health at stake, patients want to do better, and IEM systems provide the extra help they need, complete with positive feedback.
But patients also know they are being watched. The technology is barely off the ground, but concerns have already been raised about surveillance. It’s not hard to imagine insurance companies demanding proof of adherence before paying for expensive drugs. There are privacy concerns as well; everything is encrypted with IEMs, but hackers are clever.
Dr. Plowman and Mr. Travis acknowledged the concerns, and also that there’s no way to know how IEMs – if they take off – will play out in coming decades; it’s a lot like the Internet in 1992.
The intent is for the systems to remain voluntary, as they are now, perhaps with inducements for patients to use them, maybe lower insurance premiums.
“There is something inherently personal about swallowable data,” Dr. Plowman said. “It’s something we take tremendous efforts to protect.” As for compulsory use, “we take enormous strides to prevent that. It’s a major priority.”
“Always, there will be an opt-out” option, said Mr. Travis.
It’s important to consider the potential for IEMs to move medicine forward. When patients with acute bone fractures in one study, for instance, were sent home with the usual handful of oxycodone tablets, it turned out that they only took a median of six. Researchers knew that because the subjects took their oxycodone in an etectRx capsule. It’s was an important insight in the midst of an opioid epidemic (Anesth Analg. 2017 Dec;125[6]:2105-12).
Dr. Greeno is an adviser for Proteus; the company covers his travel costs.
Recently, a woman with advanced colorectal cancer at the University of Minnesota, Minneapolis, was taking capecitabine (Xeloda) in the morning but skipping her evening dose.
Her hands hurt, and she couldn’t open the childproof cap. Her daughter had been doing it for her in the morning but wasn’t around to help out at night.
It was the kind of problem that might have gone on for days or weeks until the next clinic visit, and, even then, be addressed only if the woman remembered to mention it.
But that’s not what happened. The care team realized pretty much right away that she was skipping the p.m. dose because the woman was taking her capecitabine in a gel cap with an adherence sensor.
The sensor, a sandwich of copper, silicon, and magnesium in a millimeter square, sent out an electric ping when she took her dose, activated by stomach acid; the ping was picked up by an adhesive bandage patch the woman wore, which relayed the signal to an app on her smartphone; the phone passed it on to a server cloud that the woman had given her providers permission to access.
They monitored her adherence on a Web portal, along with heart rate and activity data, also captured by the patch. The system is called Proteus Discover, from Proteus Digital Health.
Instead of taking days or weeks, the care team quickly realized that she wasn’t taking her evening dose of capecitabine. They contacted her, replaced the childproof cap, and twice-daily dosing resumed.
Expanded use in oncology?
Seven other advanced colorectal cancer patients have participated in the University of Minnesota (UM) pilot project, the first use of the device in oncology. “It’s gone so well that it’s annoying to me to not have this for all my patients. I’m already feeling frustrated that I can’t just put this in all the drugs that I give orally,” said Edward Greeno, MD, an oncologist/hematologist at the university, and the Proteus point man.
“You would assume that cancer patients would be hypercompliant, but it turns out they have the same compliance problems” as other patients, plus additional hurdles, he said, including complex regimens and drug toxicity.
“Every patient we have approached so far has been enthusiastic. They might be a little bit annoyed that I know they haven’t been taking their pills, but they are also glad that I am there to hold them responsible and help them. My intention is to roll this out much more broadly,” he said. That just might happen. Dr. Greeno is working with Proteus to roll the sensor out at UM and oncology programs elsewhere.
A slow, careful rollout
The device was cleared by the Food and Drug Administration in 2012. After more than 180,000 ingestions, there have been no safety issues, besides occasional skin irritation from the patch, which is waterproof and meant to be worn for a week, then replaced. It pings if it’s taken off. The sensor is passes through the body like food.
Patients can communicate with providers over the phone app, which also sends reminders when it’s time to take the next pill.
So far, Proteus has worked with ten health systems in the United States, and more are in the works. Commercialization efforts have focused mostly on blood pressure, cholesterol, and type 2 diabetes drugs, the bad boys of drug adherence, but the system has also been piloted for hepatitis C treatment, and trials are underway for HIV preexposure prophylaxis.
Among the company’s many favorable studies, the system has already been demonstrated to be a viable alternative to directly observed therapy in tuberculosis, the current gold-standard, but hugely labor and resource intensive (PLoS One. 2013;8[1]:e53373. doi: 10.1371/journal.pone.0053373).
Proteus can’t be picked up at the local Walgreens. The company works closely with clients and is being careful in its rollout. For one thing, each sensor has to be programed for the specific drug it’s being used with, but also, and as with any new technology, business and payment models are still being worked out.
UM’s partner in the oncology project, Fairview Health Services, pays Proteus when patients hit an adherence rate of 80%, but how much they pay is a proprietary secret. “Most of the cost issue is still not in the public domain,” said Scooter Plowman, MD, the company’s medical director.
In 2017, FDA approved a version of the antipsychotic aripiprazole embedded with the Proteus sensor. The rollout of “Abilify MyCite” by Otsuka Pharmaceuticals has been similarly cautious, under contract with health systems.
“Otsuka has been very smart in the approach they are taking,” Dr. Plowman said.
He wouldn’t give details, but Proteus is in talks with other pharmaceutical makers to bring pills with sensors to the market.
A new fix for an old problem
Proteus isn’t alone in the ingestible event marker (IEM) market. The FDA is reviewing a rival sensor from etectRx, in Gainesville, Fla.
The technology is a little different; the etectRx sensor is a microchip made out of magnesium and silver chloride that’s embedded on the inside of a gel cap. Instead of an electric blip, it sends out a radio wave when activated by stomach acid. It’s larger than the Proteus offering, but still has room to spare in a gel cap.
The signal is stronger, so patients wear a neck pendant instead of a patch to pick it up. The pendant does not capture heart rate or activity data. It’s not meant to be worn continuously and can come off after it pings the system.
The two systems are otherwise similar; etectRx also uses a phone app to relay adherence data to a server cloud clinicians can access, with patient permission. As with Proteus, everything works as long as the phone is on. President and CEO Harry Travis anticipates clearance in 2019.
Adherence is a huge and well-known problem in medicine; only about half of patients take medications as they are prescribed. People end up in the ED or the hospital with problems that might have been avoided. Providers and payers want solutions.
Industry is bringing technology to bear on the problem. The payoff will be huge for the winners; analysts project multiple billion dollar growth in the adherence technology sector.
Most companies, however, are pinning their hopes on indirect approaches, bottle caps that ping when opened, for instance, or coaching apps for smart phones. IEMs seem to be ahead of the curve.
Guardian angel or watchdog?
Whether that’s a good thing or bad thing depends on who you talk to, but patients do seem more likely to take their medications if a sensor is on board.
Dr. Plowman said he thinks IEMs improve adherence because, with their own health at stake, patients want to do better, and IEM systems provide the extra help they need, complete with positive feedback.
But patients also know they are being watched. The technology is barely off the ground, but concerns have already been raised about surveillance. It’s not hard to imagine insurance companies demanding proof of adherence before paying for expensive drugs. There are privacy concerns as well; everything is encrypted with IEMs, but hackers are clever.
Dr. Plowman and Mr. Travis acknowledged the concerns, and also that there’s no way to know how IEMs – if they take off – will play out in coming decades; it’s a lot like the Internet in 1992.
The intent is for the systems to remain voluntary, as they are now, perhaps with inducements for patients to use them, maybe lower insurance premiums.
“There is something inherently personal about swallowable data,” Dr. Plowman said. “It’s something we take tremendous efforts to protect.” As for compulsory use, “we take enormous strides to prevent that. It’s a major priority.”
“Always, there will be an opt-out” option, said Mr. Travis.
It’s important to consider the potential for IEMs to move medicine forward. When patients with acute bone fractures in one study, for instance, were sent home with the usual handful of oxycodone tablets, it turned out that they only took a median of six. Researchers knew that because the subjects took their oxycodone in an etectRx capsule. It’s was an important insight in the midst of an opioid epidemic (Anesth Analg. 2017 Dec;125[6]:2105-12).
Dr. Greeno is an adviser for Proteus; the company covers his travel costs.
Recently, a woman with advanced colorectal cancer at the University of Minnesota, Minneapolis, was taking capecitabine (Xeloda) in the morning but skipping her evening dose.
Her hands hurt, and she couldn’t open the childproof cap. Her daughter had been doing it for her in the morning but wasn’t around to help out at night.
It was the kind of problem that might have gone on for days or weeks until the next clinic visit, and, even then, be addressed only if the woman remembered to mention it.
But that’s not what happened. The care team realized pretty much right away that she was skipping the p.m. dose because the woman was taking her capecitabine in a gel cap with an adherence sensor.
The sensor, a sandwich of copper, silicon, and magnesium in a millimeter square, sent out an electric ping when she took her dose, activated by stomach acid; the ping was picked up by an adhesive bandage patch the woman wore, which relayed the signal to an app on her smartphone; the phone passed it on to a server cloud that the woman had given her providers permission to access.
They monitored her adherence on a Web portal, along with heart rate and activity data, also captured by the patch. The system is called Proteus Discover, from Proteus Digital Health.
Instead of taking days or weeks, the care team quickly realized that she wasn’t taking her evening dose of capecitabine. They contacted her, replaced the childproof cap, and twice-daily dosing resumed.
Expanded use in oncology?
Seven other advanced colorectal cancer patients have participated in the University of Minnesota (UM) pilot project, the first use of the device in oncology. “It’s gone so well that it’s annoying to me to not have this for all my patients. I’m already feeling frustrated that I can’t just put this in all the drugs that I give orally,” said Edward Greeno, MD, an oncologist/hematologist at the university, and the Proteus point man.
“You would assume that cancer patients would be hypercompliant, but it turns out they have the same compliance problems” as other patients, plus additional hurdles, he said, including complex regimens and drug toxicity.
“Every patient we have approached so far has been enthusiastic. They might be a little bit annoyed that I know they haven’t been taking their pills, but they are also glad that I am there to hold them responsible and help them. My intention is to roll this out much more broadly,” he said. That just might happen. Dr. Greeno is working with Proteus to roll the sensor out at UM and oncology programs elsewhere.
A slow, careful rollout
The device was cleared by the Food and Drug Administration in 2012. After more than 180,000 ingestions, there have been no safety issues, besides occasional skin irritation from the patch, which is waterproof and meant to be worn for a week, then replaced. It pings if it’s taken off. The sensor is passes through the body like food.
Patients can communicate with providers over the phone app, which also sends reminders when it’s time to take the next pill.
So far, Proteus has worked with ten health systems in the United States, and more are in the works. Commercialization efforts have focused mostly on blood pressure, cholesterol, and type 2 diabetes drugs, the bad boys of drug adherence, but the system has also been piloted for hepatitis C treatment, and trials are underway for HIV preexposure prophylaxis.
Among the company’s many favorable studies, the system has already been demonstrated to be a viable alternative to directly observed therapy in tuberculosis, the current gold-standard, but hugely labor and resource intensive (PLoS One. 2013;8[1]:e53373. doi: 10.1371/journal.pone.0053373).
Proteus can’t be picked up at the local Walgreens. The company works closely with clients and is being careful in its rollout. For one thing, each sensor has to be programed for the specific drug it’s being used with, but also, and as with any new technology, business and payment models are still being worked out.
UM’s partner in the oncology project, Fairview Health Services, pays Proteus when patients hit an adherence rate of 80%, but how much they pay is a proprietary secret. “Most of the cost issue is still not in the public domain,” said Scooter Plowman, MD, the company’s medical director.
In 2017, FDA approved a version of the antipsychotic aripiprazole embedded with the Proteus sensor. The rollout of “Abilify MyCite” by Otsuka Pharmaceuticals has been similarly cautious, under contract with health systems.
“Otsuka has been very smart in the approach they are taking,” Dr. Plowman said.
He wouldn’t give details, but Proteus is in talks with other pharmaceutical makers to bring pills with sensors to the market.
A new fix for an old problem
Proteus isn’t alone in the ingestible event marker (IEM) market. The FDA is reviewing a rival sensor from etectRx, in Gainesville, Fla.
The technology is a little different; the etectRx sensor is a microchip made out of magnesium and silver chloride that’s embedded on the inside of a gel cap. Instead of an electric blip, it sends out a radio wave when activated by stomach acid. It’s larger than the Proteus offering, but still has room to spare in a gel cap.
The signal is stronger, so patients wear a neck pendant instead of a patch to pick it up. The pendant does not capture heart rate or activity data. It’s not meant to be worn continuously and can come off after it pings the system.
The two systems are otherwise similar; etectRx also uses a phone app to relay adherence data to a server cloud clinicians can access, with patient permission. As with Proteus, everything works as long as the phone is on. President and CEO Harry Travis anticipates clearance in 2019.
Adherence is a huge and well-known problem in medicine; only about half of patients take medications as they are prescribed. People end up in the ED or the hospital with problems that might have been avoided. Providers and payers want solutions.
Industry is bringing technology to bear on the problem. The payoff will be huge for the winners; analysts project multiple billion dollar growth in the adherence technology sector.
Most companies, however, are pinning their hopes on indirect approaches, bottle caps that ping when opened, for instance, or coaching apps for smart phones. IEMs seem to be ahead of the curve.
Guardian angel or watchdog?
Whether that’s a good thing or bad thing depends on who you talk to, but patients do seem more likely to take their medications if a sensor is on board.
Dr. Plowman said he thinks IEMs improve adherence because, with their own health at stake, patients want to do better, and IEM systems provide the extra help they need, complete with positive feedback.
But patients also know they are being watched. The technology is barely off the ground, but concerns have already been raised about surveillance. It’s not hard to imagine insurance companies demanding proof of adherence before paying for expensive drugs. There are privacy concerns as well; everything is encrypted with IEMs, but hackers are clever.
Dr. Plowman and Mr. Travis acknowledged the concerns, and also that there’s no way to know how IEMs – if they take off – will play out in coming decades; it’s a lot like the Internet in 1992.
The intent is for the systems to remain voluntary, as they are now, perhaps with inducements for patients to use them, maybe lower insurance premiums.
“There is something inherently personal about swallowable data,” Dr. Plowman said. “It’s something we take tremendous efforts to protect.” As for compulsory use, “we take enormous strides to prevent that. It’s a major priority.”
“Always, there will be an opt-out” option, said Mr. Travis.
It’s important to consider the potential for IEMs to move medicine forward. When patients with acute bone fractures in one study, for instance, were sent home with the usual handful of oxycodone tablets, it turned out that they only took a median of six. Researchers knew that because the subjects took their oxycodone in an etectRx capsule. It’s was an important insight in the midst of an opioid epidemic (Anesth Analg. 2017 Dec;125[6]:2105-12).
Dr. Greeno is an adviser for Proteus; the company covers his travel costs.
Penicillin allergy
A 75-year-old man presents with fever, chills, and facial pain. He had an upper respiratory infection 3 weeks ago and has had persistent sinus drainage since. He has tried nasal irrigation and nasal steroids without improvement.
Over the past 5 days, he has had thicker postnasal drip, the development of facial pain, and today fevers as high as 102 degrees. He has a history of giant cell arteritis, for which he takes 30 mg of prednisone daily; coronary artery disease; and hypertension. He has a penicillin allergy (rash on chest, back, and arms 25 years ago). Exam reveals temperature of 101.5 and tenderness over left maxillary sinus.
What treatment do you recommend?
A. Amoxicillin/clavulanate.
B. Cefpodoxime.
C. Levofloxacin.
D. Trimethoprim/sulfamethoxazole.
I think cefpodoxime is probably the best of these choices to treat sinusitis in this patient. Choosing amoxicillin /clavulanate is an option only if you could give the patient a test dose in a controlled setting. I think giving this patient levofloxacin poses greater risk than a penicillin rechallenge. This patient is elderly and on prednisone, both of which increase his risk of tendon rupture if given a quinolone. Also, the Food and Drug Administration released a warning recently regarding increased risk of aortic disease in patients with cardiovascular risk factors who receive fluoroquinolones.1
Merin Kuruvilla, MD, and colleagues described oral amoxicillin challenge for patients with a history of low-risk penicillin allergy (described as benign rash, benign somatic symptoms, or unknown history with penicillin exposure more than 12 months prior).2 The study was done in a single allergy practice where 38 of 50 patients with penicillin allergy histories qualified for the study. Of the 38 eligible patients, 20 consented to oral rechallenge in clinic, and none of them developed immediate or delayed hypersensitivity reactions.
Melissa Iammatteo, MD, et al. studied 155 patients with a history of non–life-threatening penicillin reactions.3 Study participants received placebo followed by a two-step graded challenge to amoxicillin. No reaction occurred in 77% of patients, while 20% of patients had nonallergic reactions, which were equal between placebo and amoxicillin. Only 2.6 % had allergic reactions, all of which were classified as mild.
Reported penicillin allergy occurs in about 10% of community patients, but 90% of these patients can tolerate penicillins.4 Patients reporting a penicillin allergy have increased risk for drug resistance and prolonged hospital stays.5
The American Academy of Allergy, Asthma & Immunology recommended more widespread and routine performance of penicillin allergy testing in patients with a history of allergy to penicillin or other beta-lactam antibiotics.6 Patients who have penicillin allergy histories are more likely to receive drugs, such as clindamycin or a fluoroquinolone, that may carry much greater risks than a beta-lactam antibiotic. It also leads to more vancomycin use, which increases risk of vancomycin resistance.
Allergic reactions to cephalosporins are very infrequent in patients with a penicillin allergy. Eric Macy, MD, and colleagues studied all members of Kaiser Permanente Southern California health plan who had received cephalosporins over a 2-year period.7 More than 275,000 courses were given to patients with penicillin allergy, with only about 1% having an allergic reaction and only three cases of anaphylaxis.
Pearl: Most patients with a history of penicillin allergy will tolerate penicillins and cephalosporins. Penicillin allergy testing should be done to assess if they have a penicillin allergy, and in low-risk patients (patients who do not recall the allergy or had a maculopapular rash), consideration for oral rechallenge in a controlled setting may be an option. Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at dpaauw@uw.edu.
References
1. Food and Drug Administration. “FDA warns about increased risk of ruptures or tears in the aorta blood vessel with fluoroquinolone antibiotics in certain patients,” 2018 Dec 20.
2. Ann Allergy Asthma Immunol. 2018 Nov;121(5):627-8.
3. J Allergy Clin Immunol Pract. 2019 Jan;7(1):236-43.
4. Immunol Allergy Clin North Am. 2017 Nov;37(4):643-62.
5. J Allergy Clin Immunol. 2014 Mar;133(3):790-6.
6. J Allergy Clin Immunol Pract. 2017 Mar - Apr;5(2):333-4.
7. J Allergy Clin Immunol. 2015 Mar;135(3):745-52.e5.
A 75-year-old man presents with fever, chills, and facial pain. He had an upper respiratory infection 3 weeks ago and has had persistent sinus drainage since. He has tried nasal irrigation and nasal steroids without improvement.
Over the past 5 days, he has had thicker postnasal drip, the development of facial pain, and today fevers as high as 102 degrees. He has a history of giant cell arteritis, for which he takes 30 mg of prednisone daily; coronary artery disease; and hypertension. He has a penicillin allergy (rash on chest, back, and arms 25 years ago). Exam reveals temperature of 101.5 and tenderness over left maxillary sinus.
What treatment do you recommend?
A. Amoxicillin/clavulanate.
B. Cefpodoxime.
C. Levofloxacin.
D. Trimethoprim/sulfamethoxazole.
I think cefpodoxime is probably the best of these choices to treat sinusitis in this patient. Choosing amoxicillin /clavulanate is an option only if you could give the patient a test dose in a controlled setting. I think giving this patient levofloxacin poses greater risk than a penicillin rechallenge. This patient is elderly and on prednisone, both of which increase his risk of tendon rupture if given a quinolone. Also, the Food and Drug Administration released a warning recently regarding increased risk of aortic disease in patients with cardiovascular risk factors who receive fluoroquinolones.1
Merin Kuruvilla, MD, and colleagues described oral amoxicillin challenge for patients with a history of low-risk penicillin allergy (described as benign rash, benign somatic symptoms, or unknown history with penicillin exposure more than 12 months prior).2 The study was done in a single allergy practice where 38 of 50 patients with penicillin allergy histories qualified for the study. Of the 38 eligible patients, 20 consented to oral rechallenge in clinic, and none of them developed immediate or delayed hypersensitivity reactions.
Melissa Iammatteo, MD, et al. studied 155 patients with a history of non–life-threatening penicillin reactions.3 Study participants received placebo followed by a two-step graded challenge to amoxicillin. No reaction occurred in 77% of patients, while 20% of patients had nonallergic reactions, which were equal between placebo and amoxicillin. Only 2.6 % had allergic reactions, all of which were classified as mild.
Reported penicillin allergy occurs in about 10% of community patients, but 90% of these patients can tolerate penicillins.4 Patients reporting a penicillin allergy have increased risk for drug resistance and prolonged hospital stays.5
The American Academy of Allergy, Asthma & Immunology recommended more widespread and routine performance of penicillin allergy testing in patients with a history of allergy to penicillin or other beta-lactam antibiotics.6 Patients who have penicillin allergy histories are more likely to receive drugs, such as clindamycin or a fluoroquinolone, that may carry much greater risks than a beta-lactam antibiotic. It also leads to more vancomycin use, which increases risk of vancomycin resistance.
Allergic reactions to cephalosporins are very infrequent in patients with a penicillin allergy. Eric Macy, MD, and colleagues studied all members of Kaiser Permanente Southern California health plan who had received cephalosporins over a 2-year period.7 More than 275,000 courses were given to patients with penicillin allergy, with only about 1% having an allergic reaction and only three cases of anaphylaxis.
Pearl: Most patients with a history of penicillin allergy will tolerate penicillins and cephalosporins. Penicillin allergy testing should be done to assess if they have a penicillin allergy, and in low-risk patients (patients who do not recall the allergy or had a maculopapular rash), consideration for oral rechallenge in a controlled setting may be an option. Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at dpaauw@uw.edu.
References
1. Food and Drug Administration. “FDA warns about increased risk of ruptures or tears in the aorta blood vessel with fluoroquinolone antibiotics in certain patients,” 2018 Dec 20.
2. Ann Allergy Asthma Immunol. 2018 Nov;121(5):627-8.
3. J Allergy Clin Immunol Pract. 2019 Jan;7(1):236-43.
4. Immunol Allergy Clin North Am. 2017 Nov;37(4):643-62.
5. J Allergy Clin Immunol. 2014 Mar;133(3):790-6.
6. J Allergy Clin Immunol Pract. 2017 Mar - Apr;5(2):333-4.
7. J Allergy Clin Immunol. 2015 Mar;135(3):745-52.e5.
A 75-year-old man presents with fever, chills, and facial pain. He had an upper respiratory infection 3 weeks ago and has had persistent sinus drainage since. He has tried nasal irrigation and nasal steroids without improvement.
Over the past 5 days, he has had thicker postnasal drip, the development of facial pain, and today fevers as high as 102 degrees. He has a history of giant cell arteritis, for which he takes 30 mg of prednisone daily; coronary artery disease; and hypertension. He has a penicillin allergy (rash on chest, back, and arms 25 years ago). Exam reveals temperature of 101.5 and tenderness over left maxillary sinus.
What treatment do you recommend?
A. Amoxicillin/clavulanate.
B. Cefpodoxime.
C. Levofloxacin.
D. Trimethoprim/sulfamethoxazole.
I think cefpodoxime is probably the best of these choices to treat sinusitis in this patient. Choosing amoxicillin /clavulanate is an option only if you could give the patient a test dose in a controlled setting. I think giving this patient levofloxacin poses greater risk than a penicillin rechallenge. This patient is elderly and on prednisone, both of which increase his risk of tendon rupture if given a quinolone. Also, the Food and Drug Administration released a warning recently regarding increased risk of aortic disease in patients with cardiovascular risk factors who receive fluoroquinolones.1
Merin Kuruvilla, MD, and colleagues described oral amoxicillin challenge for patients with a history of low-risk penicillin allergy (described as benign rash, benign somatic symptoms, or unknown history with penicillin exposure more than 12 months prior).2 The study was done in a single allergy practice where 38 of 50 patients with penicillin allergy histories qualified for the study. Of the 38 eligible patients, 20 consented to oral rechallenge in clinic, and none of them developed immediate or delayed hypersensitivity reactions.
Melissa Iammatteo, MD, et al. studied 155 patients with a history of non–life-threatening penicillin reactions.3 Study participants received placebo followed by a two-step graded challenge to amoxicillin. No reaction occurred in 77% of patients, while 20% of patients had nonallergic reactions, which were equal between placebo and amoxicillin. Only 2.6 % had allergic reactions, all of which were classified as mild.
Reported penicillin allergy occurs in about 10% of community patients, but 90% of these patients can tolerate penicillins.4 Patients reporting a penicillin allergy have increased risk for drug resistance and prolonged hospital stays.5
The American Academy of Allergy, Asthma & Immunology recommended more widespread and routine performance of penicillin allergy testing in patients with a history of allergy to penicillin or other beta-lactam antibiotics.6 Patients who have penicillin allergy histories are more likely to receive drugs, such as clindamycin or a fluoroquinolone, that may carry much greater risks than a beta-lactam antibiotic. It also leads to more vancomycin use, which increases risk of vancomycin resistance.
Allergic reactions to cephalosporins are very infrequent in patients with a penicillin allergy. Eric Macy, MD, and colleagues studied all members of Kaiser Permanente Southern California health plan who had received cephalosporins over a 2-year period.7 More than 275,000 courses were given to patients with penicillin allergy, with only about 1% having an allergic reaction and only three cases of anaphylaxis.
Pearl: Most patients with a history of penicillin allergy will tolerate penicillins and cephalosporins. Penicillin allergy testing should be done to assess if they have a penicillin allergy, and in low-risk patients (patients who do not recall the allergy or had a maculopapular rash), consideration for oral rechallenge in a controlled setting may be an option. Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at dpaauw@uw.edu.
References
1. Food and Drug Administration. “FDA warns about increased risk of ruptures or tears in the aorta blood vessel with fluoroquinolone antibiotics in certain patients,” 2018 Dec 20.
2. Ann Allergy Asthma Immunol. 2018 Nov;121(5):627-8.
3. J Allergy Clin Immunol Pract. 2019 Jan;7(1):236-43.
4. Immunol Allergy Clin North Am. 2017 Nov;37(4):643-62.
5. J Allergy Clin Immunol. 2014 Mar;133(3):790-6.
6. J Allergy Clin Immunol Pract. 2017 Mar - Apr;5(2):333-4.
7. J Allergy Clin Immunol. 2015 Mar;135(3):745-52.e5.
The personal cancer vaccine NEO-PV-01 shows promise in metastatic cancers
WASHINGTON – according to findings from the ongoing phase 1b NT-001study of patients with metastatic melanoma, smoking-associated non–small cell lung cancer (NSCLC), and bladder cancer.
No vaccine-related serious adverse events occurred in 34 patients in a per-protocol set who were treated with the regimen, Siwen Hu-Lieskovan, MD, PhD, reported at the annual meeting of the Society for Immunotherapy of Cancer.
“We found that NEO-PV-01 in combination with nivolumab was very safe; we did not see any grade 3 to grade 4 toxicity associated with the combination,” said Dr. Hu-Lieskovan, a medical oncologist at the University of California, Los Angeles.
Most adverse events that occurred were mild and related to the local injection, she noted.
Although safety was the primary endpoint of the study, Dr. Hu-Lieskovan and her colleagues also looked at immune responses and treatment efficacy, however, with respect to translational data her presentation addressed only the findings in the melanoma and lung cancer patients.
All patients exhibited an immune response to the vaccine, with 56% of the epitopes generating CD4- and/or CD8-positive T cell responses.
“These immune responses were very durable and still could be detected 52 weeks into the treatment,” she said. Additionally, epitope spreading – increased immune response targeting nonvaccine epitopes (which is indirect evidence of vaccine-induced tumor toxicity) – was observed in 8 of 10 patients tested.
The study subjects, including 16 adults with melanoma, 11 with NSCLC, and 7 with bladder cancer, were treated with nivolumab every 2 weeks for 12 weeks prior to vaccination (while their personalized vaccine was being developed). NEO-PV-01 – which included up to 20 unique peptides plus the immunostimulant poly-ICLC – was then administered subcutaneously in five priming doses followed by two booster doses over the next 12 weeks. Of note, very few patients had programmed cell death protein 1 expression of 50% or greater, including only 13.3%, 28.6%, and 0% of the melanoma, NSCLC, and bladder cancer patients, respectively, and tumor mutation burden was consistent with published reports, she said.
As for efficacy, 11 of 16 melanoma patients (68.6%) had either a partial response (8 pre vaccination and an additional 3 post vaccination) or stable disease. One (6.3%) had a postvaccination complete response, Dr. Hu-Lieskovan said.
“[This is] much better than the historical data,” she noted, adding that 12 patients (75%) are still on the study and continuing treatment with response duration of at least 39.7 weeks.
Of the 11 NSCLC patients, 5 (45.5%) had a partial response (3 pre vaccination and 2 post vaccination), and none had a complete response. Seven (63.6%) remained on the study and were continuing treatment, and response duration was at least 30.6 weeks.
An exploratory analysis of tumor responses after vaccination showed that the majority of melanoma patients and half of the lung cancer patients had further tumor shrinkage after vaccination, and some patients were converted to responders. Most – including some with stable or progressive disease – stayed on treatment, she said.
The findings demonstrate that NEO-PV-01 is very well tolerated and associated with post vaccine responses observed after week 24.
“We saw evidence of vaccination-induced immune response specific to the injected vaccine, as well as epitope spreading, and the T cells induced by these neoantigens can traffic into the tumor and they seem to be functional,” she concluded.
Dr. Hu-Lieskovan reported receiving consulting fees and/or research support from Amgen, BMS, Genmab, Merck, and Vaccinex. She is the UCLA site principal investigator for the NT-001 study and has conducted contracted research for Astellas, F Star, Genentech, Nektar Therapeutics, Neon Therapeutics, Pfizer, Plexxikon, and Xencor.
SOURCE: Hu-Lieskovan S et al. SITC 2018, Abstract 07.
WASHINGTON – according to findings from the ongoing phase 1b NT-001study of patients with metastatic melanoma, smoking-associated non–small cell lung cancer (NSCLC), and bladder cancer.
No vaccine-related serious adverse events occurred in 34 patients in a per-protocol set who were treated with the regimen, Siwen Hu-Lieskovan, MD, PhD, reported at the annual meeting of the Society for Immunotherapy of Cancer.
“We found that NEO-PV-01 in combination with nivolumab was very safe; we did not see any grade 3 to grade 4 toxicity associated with the combination,” said Dr. Hu-Lieskovan, a medical oncologist at the University of California, Los Angeles.
Most adverse events that occurred were mild and related to the local injection, she noted.
Although safety was the primary endpoint of the study, Dr. Hu-Lieskovan and her colleagues also looked at immune responses and treatment efficacy, however, with respect to translational data her presentation addressed only the findings in the melanoma and lung cancer patients.
All patients exhibited an immune response to the vaccine, with 56% of the epitopes generating CD4- and/or CD8-positive T cell responses.
“These immune responses were very durable and still could be detected 52 weeks into the treatment,” she said. Additionally, epitope spreading – increased immune response targeting nonvaccine epitopes (which is indirect evidence of vaccine-induced tumor toxicity) – was observed in 8 of 10 patients tested.
The study subjects, including 16 adults with melanoma, 11 with NSCLC, and 7 with bladder cancer, were treated with nivolumab every 2 weeks for 12 weeks prior to vaccination (while their personalized vaccine was being developed). NEO-PV-01 – which included up to 20 unique peptides plus the immunostimulant poly-ICLC – was then administered subcutaneously in five priming doses followed by two booster doses over the next 12 weeks. Of note, very few patients had programmed cell death protein 1 expression of 50% or greater, including only 13.3%, 28.6%, and 0% of the melanoma, NSCLC, and bladder cancer patients, respectively, and tumor mutation burden was consistent with published reports, she said.
As for efficacy, 11 of 16 melanoma patients (68.6%) had either a partial response (8 pre vaccination and an additional 3 post vaccination) or stable disease. One (6.3%) had a postvaccination complete response, Dr. Hu-Lieskovan said.
“[This is] much better than the historical data,” she noted, adding that 12 patients (75%) are still on the study and continuing treatment with response duration of at least 39.7 weeks.
Of the 11 NSCLC patients, 5 (45.5%) had a partial response (3 pre vaccination and 2 post vaccination), and none had a complete response. Seven (63.6%) remained on the study and were continuing treatment, and response duration was at least 30.6 weeks.
An exploratory analysis of tumor responses after vaccination showed that the majority of melanoma patients and half of the lung cancer patients had further tumor shrinkage after vaccination, and some patients were converted to responders. Most – including some with stable or progressive disease – stayed on treatment, she said.
The findings demonstrate that NEO-PV-01 is very well tolerated and associated with post vaccine responses observed after week 24.
“We saw evidence of vaccination-induced immune response specific to the injected vaccine, as well as epitope spreading, and the T cells induced by these neoantigens can traffic into the tumor and they seem to be functional,” she concluded.
Dr. Hu-Lieskovan reported receiving consulting fees and/or research support from Amgen, BMS, Genmab, Merck, and Vaccinex. She is the UCLA site principal investigator for the NT-001 study and has conducted contracted research for Astellas, F Star, Genentech, Nektar Therapeutics, Neon Therapeutics, Pfizer, Plexxikon, and Xencor.
SOURCE: Hu-Lieskovan S et al. SITC 2018, Abstract 07.
WASHINGTON – according to findings from the ongoing phase 1b NT-001study of patients with metastatic melanoma, smoking-associated non–small cell lung cancer (NSCLC), and bladder cancer.
No vaccine-related serious adverse events occurred in 34 patients in a per-protocol set who were treated with the regimen, Siwen Hu-Lieskovan, MD, PhD, reported at the annual meeting of the Society for Immunotherapy of Cancer.
“We found that NEO-PV-01 in combination with nivolumab was very safe; we did not see any grade 3 to grade 4 toxicity associated with the combination,” said Dr. Hu-Lieskovan, a medical oncologist at the University of California, Los Angeles.
Most adverse events that occurred were mild and related to the local injection, she noted.
Although safety was the primary endpoint of the study, Dr. Hu-Lieskovan and her colleagues also looked at immune responses and treatment efficacy, however, with respect to translational data her presentation addressed only the findings in the melanoma and lung cancer patients.
All patients exhibited an immune response to the vaccine, with 56% of the epitopes generating CD4- and/or CD8-positive T cell responses.
“These immune responses were very durable and still could be detected 52 weeks into the treatment,” she said. Additionally, epitope spreading – increased immune response targeting nonvaccine epitopes (which is indirect evidence of vaccine-induced tumor toxicity) – was observed in 8 of 10 patients tested.
The study subjects, including 16 adults with melanoma, 11 with NSCLC, and 7 with bladder cancer, were treated with nivolumab every 2 weeks for 12 weeks prior to vaccination (while their personalized vaccine was being developed). NEO-PV-01 – which included up to 20 unique peptides plus the immunostimulant poly-ICLC – was then administered subcutaneously in five priming doses followed by two booster doses over the next 12 weeks. Of note, very few patients had programmed cell death protein 1 expression of 50% or greater, including only 13.3%, 28.6%, and 0% of the melanoma, NSCLC, and bladder cancer patients, respectively, and tumor mutation burden was consistent with published reports, she said.
As for efficacy, 11 of 16 melanoma patients (68.6%) had either a partial response (8 pre vaccination and an additional 3 post vaccination) or stable disease. One (6.3%) had a postvaccination complete response, Dr. Hu-Lieskovan said.
“[This is] much better than the historical data,” she noted, adding that 12 patients (75%) are still on the study and continuing treatment with response duration of at least 39.7 weeks.
Of the 11 NSCLC patients, 5 (45.5%) had a partial response (3 pre vaccination and 2 post vaccination), and none had a complete response. Seven (63.6%) remained on the study and were continuing treatment, and response duration was at least 30.6 weeks.
An exploratory analysis of tumor responses after vaccination showed that the majority of melanoma patients and half of the lung cancer patients had further tumor shrinkage after vaccination, and some patients were converted to responders. Most – including some with stable or progressive disease – stayed on treatment, she said.
The findings demonstrate that NEO-PV-01 is very well tolerated and associated with post vaccine responses observed after week 24.
“We saw evidence of vaccination-induced immune response specific to the injected vaccine, as well as epitope spreading, and the T cells induced by these neoantigens can traffic into the tumor and they seem to be functional,” she concluded.
Dr. Hu-Lieskovan reported receiving consulting fees and/or research support from Amgen, BMS, Genmab, Merck, and Vaccinex. She is the UCLA site principal investigator for the NT-001 study and has conducted contracted research for Astellas, F Star, Genentech, Nektar Therapeutics, Neon Therapeutics, Pfizer, Plexxikon, and Xencor.
SOURCE: Hu-Lieskovan S et al. SITC 2018, Abstract 07.
REPORTING FROM SITC 2018
Key clinical point: The NEO-PV-01 personalized cancer vaccine shows good tolerability and safety and appears to have clinical efficacy.
Major finding: There were no vaccine-related serious adverse events, and all patients exhibited an immune response to the vaccine, with 56% of the epitopes generating CD4- and/or CD8-positive T-cell responses.
Study details: A study of the NEO-PV-01 personalized cancer vaccine in 34 patients.
Disclosures: Dr. Hu-Lieskovan reported receiving consulting fees and/or research support from Amgen, BMS, Genmab, Merck, and Vaccinex. She is the UCLA site principal investigator for the NT-001 study and has conducted contracted research for Astellas, F Star, Genentech, Nektar Therapeutics, Neon Therapeutics, Pfizer, Plexxikon, and Xencor.
Source: Hu-Lieskovan S et al. SITC 2018, Abstract 07.
Smoking has small effect on postop hernia wound morbidity
Smoking before elective clean open ventral hernia repair (OVHR) was not associated with significant differences in postoperative wound morbidity or all 30-day morbidity, according to results published in Surgery.
The question of the impact of smoking on infection risk and wound healing has been studied in a variety of surgical procedures. While some associations have been established (JAMA Surg. 2017;152[5]:476-83; Infect Immun. 2015;83[6]:2443-52), the data have not been strong enough to lead to recommendations that surgeons delay procedures until a patient stops smoking.
In a study of 418 active smokers and 418 matched patients who had never smoked, the rate of surgical site infection (SSI) was 4.1% in both groups (P = .98). The total 30-day complication rates were 7.5% for current smokers and 6.6% for nonsmokers (P = .60), reported Clayton C. Petro, MD, of the department of general surgery at the Cleveland Clinic Comprehensive Hernia Center, and his coauthors.
Patient data were obtained from the Americas Hernia Society Quality Collaborative (AHSQC), and included those who had undergone OVHR in a Centers for Disease Control and Prevention class 1 wound with 30-day follow-up.
Participants were grouped according to smoking status: those who had never smoked, those who had smoked within 30 days of operation (“current smokers”), and patients who had quit smoking more than 1 month before operation (“former smokers”). Current smokers were matched with never smokers by characteristics including age, sex, and body mass index, and conditions such as diabetes and hypertension. Former smokers were excluded.
The investigators collected data from both groups on SSI, surgical site occurrence (SSO), surgical site occurrence requiring a procedural intervention (SSOPI), and all 30-day morbidity (any morbidity event entered into the AHSQC database within 30 days of operation), the authors said.
Rates of SSI were 4.1% for both smokers and nonsmokers (P = .98). Rates of SSO were greater in current smokers (12.0% vs. 7.4%, P = .03). SSOPI and reoperation rates were similar in current and never smokers (6.2% vs. 5.0%, P = .43; 1.9% vs. 1.2%, P = .39, respectively).
The results suggest that “active smoking before an elective OVHR in a CDC class I wound has a clinically negligible impact on postoperative wound morbidity and all 30-day morbidity,” wrote Dr. Petro and his colleagues.
“Surgeons allowing perioperative smoking should monitor their outcomes to [ensure that] these findings are replicable in their own practice,” they concluded.
The study authors disclosed financial relationships with Medtronic, Ariste Medical, and other companies.
SOURCE: Petro CC et al. Surgery. 2019 Feb;165(2):406-11.
Smoking before elective clean open ventral hernia repair (OVHR) was not associated with significant differences in postoperative wound morbidity or all 30-day morbidity, according to results published in Surgery.
The question of the impact of smoking on infection risk and wound healing has been studied in a variety of surgical procedures. While some associations have been established (JAMA Surg. 2017;152[5]:476-83; Infect Immun. 2015;83[6]:2443-52), the data have not been strong enough to lead to recommendations that surgeons delay procedures until a patient stops smoking.
In a study of 418 active smokers and 418 matched patients who had never smoked, the rate of surgical site infection (SSI) was 4.1% in both groups (P = .98). The total 30-day complication rates were 7.5% for current smokers and 6.6% for nonsmokers (P = .60), reported Clayton C. Petro, MD, of the department of general surgery at the Cleveland Clinic Comprehensive Hernia Center, and his coauthors.
Patient data were obtained from the Americas Hernia Society Quality Collaborative (AHSQC), and included those who had undergone OVHR in a Centers for Disease Control and Prevention class 1 wound with 30-day follow-up.
Participants were grouped according to smoking status: those who had never smoked, those who had smoked within 30 days of operation (“current smokers”), and patients who had quit smoking more than 1 month before operation (“former smokers”). Current smokers were matched with never smokers by characteristics including age, sex, and body mass index, and conditions such as diabetes and hypertension. Former smokers were excluded.
The investigators collected data from both groups on SSI, surgical site occurrence (SSO), surgical site occurrence requiring a procedural intervention (SSOPI), and all 30-day morbidity (any morbidity event entered into the AHSQC database within 30 days of operation), the authors said.
Rates of SSI were 4.1% for both smokers and nonsmokers (P = .98). Rates of SSO were greater in current smokers (12.0% vs. 7.4%, P = .03). SSOPI and reoperation rates were similar in current and never smokers (6.2% vs. 5.0%, P = .43; 1.9% vs. 1.2%, P = .39, respectively).
The results suggest that “active smoking before an elective OVHR in a CDC class I wound has a clinically negligible impact on postoperative wound morbidity and all 30-day morbidity,” wrote Dr. Petro and his colleagues.
“Surgeons allowing perioperative smoking should monitor their outcomes to [ensure that] these findings are replicable in their own practice,” they concluded.
The study authors disclosed financial relationships with Medtronic, Ariste Medical, and other companies.
SOURCE: Petro CC et al. Surgery. 2019 Feb;165(2):406-11.
Smoking before elective clean open ventral hernia repair (OVHR) was not associated with significant differences in postoperative wound morbidity or all 30-day morbidity, according to results published in Surgery.
The question of the impact of smoking on infection risk and wound healing has been studied in a variety of surgical procedures. While some associations have been established (JAMA Surg. 2017;152[5]:476-83; Infect Immun. 2015;83[6]:2443-52), the data have not been strong enough to lead to recommendations that surgeons delay procedures until a patient stops smoking.
In a study of 418 active smokers and 418 matched patients who had never smoked, the rate of surgical site infection (SSI) was 4.1% in both groups (P = .98). The total 30-day complication rates were 7.5% for current smokers and 6.6% for nonsmokers (P = .60), reported Clayton C. Petro, MD, of the department of general surgery at the Cleveland Clinic Comprehensive Hernia Center, and his coauthors.
Patient data were obtained from the Americas Hernia Society Quality Collaborative (AHSQC), and included those who had undergone OVHR in a Centers for Disease Control and Prevention class 1 wound with 30-day follow-up.
Participants were grouped according to smoking status: those who had never smoked, those who had smoked within 30 days of operation (“current smokers”), and patients who had quit smoking more than 1 month before operation (“former smokers”). Current smokers were matched with never smokers by characteristics including age, sex, and body mass index, and conditions such as diabetes and hypertension. Former smokers were excluded.
The investigators collected data from both groups on SSI, surgical site occurrence (SSO), surgical site occurrence requiring a procedural intervention (SSOPI), and all 30-day morbidity (any morbidity event entered into the AHSQC database within 30 days of operation), the authors said.
Rates of SSI were 4.1% for both smokers and nonsmokers (P = .98). Rates of SSO were greater in current smokers (12.0% vs. 7.4%, P = .03). SSOPI and reoperation rates were similar in current and never smokers (6.2% vs. 5.0%, P = .43; 1.9% vs. 1.2%, P = .39, respectively).
The results suggest that “active smoking before an elective OVHR in a CDC class I wound has a clinically negligible impact on postoperative wound morbidity and all 30-day morbidity,” wrote Dr. Petro and his colleagues.
“Surgeons allowing perioperative smoking should monitor their outcomes to [ensure that] these findings are replicable in their own practice,” they concluded.
The study authors disclosed financial relationships with Medtronic, Ariste Medical, and other companies.
SOURCE: Petro CC et al. Surgery. 2019 Feb;165(2):406-11.
FROM SURGERY
Key clinical point: Smoking did not appear to increase the risk of wound morbidity after open ventral hernia repair surgery.
Major finding: Rates of postop surgical site infection were 4.1% for both smokers and nonsmokers.
Study details: Data from matched hernia repair patients (418 smokers and 418 nonsmokers) obtained from the Americas Hernia Society Quality Collaborative.
Disclosures: The study authors disclosed financial relationships with Medtronic, Ariste Medical, and other companies.
Source: Petro CC et al. Surgery. 2019 Feb;165(2):406-11.
FDA grants BI-1206 orphan designation for MCL
The Food and Drug Administration has granted orphan designation to BI-1206 for the treatment of mantle cell lymphoma (MCL).
BI-1206 is a monoclonal antibody being developed by BioInvent International.
The company says BI-1206 works by inhibiting FcgRIIB (CD32B), which is associated with poor prognosis in MCL and other non-Hodgkin lymphomas. By inhibiting FcgRIIB, BI-1206 is expected to enhance the activity of rituximab or other anti-CD20 monoclonal antibodies.
BioInvent is conducting a phase 1/2a study (NCT03571568) of BI-1206 in combination with rituximab in patients with indolent, relapsed/refractory B-cell non-Hodgkin lymphomas, including MCL. The first patient began receiving treatment with BI-1206 in September 2018.
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases or disorders that affect fewer than 200,000 people in the United States. Orphan designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The Food and Drug Administration has granted orphan designation to BI-1206 for the treatment of mantle cell lymphoma (MCL).
BI-1206 is a monoclonal antibody being developed by BioInvent International.
The company says BI-1206 works by inhibiting FcgRIIB (CD32B), which is associated with poor prognosis in MCL and other non-Hodgkin lymphomas. By inhibiting FcgRIIB, BI-1206 is expected to enhance the activity of rituximab or other anti-CD20 monoclonal antibodies.
BioInvent is conducting a phase 1/2a study (NCT03571568) of BI-1206 in combination with rituximab in patients with indolent, relapsed/refractory B-cell non-Hodgkin lymphomas, including MCL. The first patient began receiving treatment with BI-1206 in September 2018.
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases or disorders that affect fewer than 200,000 people in the United States. Orphan designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The Food and Drug Administration has granted orphan designation to BI-1206 for the treatment of mantle cell lymphoma (MCL).
BI-1206 is a monoclonal antibody being developed by BioInvent International.
The company says BI-1206 works by inhibiting FcgRIIB (CD32B), which is associated with poor prognosis in MCL and other non-Hodgkin lymphomas. By inhibiting FcgRIIB, BI-1206 is expected to enhance the activity of rituximab or other anti-CD20 monoclonal antibodies.
BioInvent is conducting a phase 1/2a study (NCT03571568) of BI-1206 in combination with rituximab in patients with indolent, relapsed/refractory B-cell non-Hodgkin lymphomas, including MCL. The first patient began receiving treatment with BI-1206 in September 2018.
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases or disorders that affect fewer than 200,000 people in the United States. Orphan designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
Female radiation oncologists receive less funds from industry
In the year 2016, female radiation oncologists received less money than did male radiation oncologists from industry across every type of compensation evaluated, according to an analysis of data from the Centers for Medicare & Medicated Services Open Payments program.
These sex disparities included a median $1,000 less for consulting (P = .005), $500 less for honoraria (P = .005) and $135 less for research payments, although the difference in this latter category fell short of statistical significance (P = .08), reported Julius K. Weng, department of radiation oncology, University of California, Los Angeles, and his colleagues, in JAMA Network Open.
The CMS Open Payments Program, which is part of the Physician Payments Sunshine Act, made the study possible. The CMS data were intended to allow evaluation of potential conflicts of interest, but they were amenable to compare payments by gender. For this purpose, the investigators determined gender by first name, which was confirmed by Internet search when ambiguous.
At least one industry payment in 2016 was made to 61.4% of 1,164 female and 70.4% of 3,319 male radiation oncologists included in this retrospective cross-sectional study.
Of research funding, only 20.7% went to women even though they represented 25.9% of radiation oncologists in the United States at that time. In other categories, such as honoraria or consulting, the proportion of compensation going to females was even lower, never exceeding 15%.
In the United States, about one-third of radiation oncologists are women, according to the authors of this study. This is substantially lower than the proportion in many other specialties and is far below that of current medical school enrollment, where women are now in a slight majority.
It cannot be ascertained from these data why industry compensation was lower for women, but the authors offered numerous potential explanations including the possibility that more female than male radiation oncologists do not elect to pursue relationships with industry. They labeled such relationships as “controversial” due to potential conflicts of interest.
Among the theories put forth are those that have been proposed to explain other sex disparities, including lower salaries and slower promotion, in medicine and elsewhere.
For one, it has been suggested that “agentic traits” of men might propel them to seek opportunities more aggressively, compared with women, who have “historically been associated with communal qualities,” according to the authors.
If due to gender bias, disparities may also accumulate over time as “downstream consequences of sex gaps experienced early in a female physician’s career,” the authors stated. They noted that women have a lower proportion of leadership positions in radiation oncology than predicted by their numbers in the specialty.
The disparity in industry partnerships and compensation is a relevant measure of sex disparity because these are associated with “substantial career advantages,” according to the authors of this study. In addition to the advantages of research funding, they believe these include association with important signs of success in academic clinical medicine, such being identified as a key opinion leader.
One limitation of the CMS data regarding industry payments is that the information is derived from self-reports. In 2016, the CMS Open Payments Program was in its fourth year, which the authors suggested had more complete information on industry payments than prior years because of initiatives to improve reporting compliance.
The lower payments from compensation are likely to be related to other gender disparities in radiation oncology, such as lower publication productivity and fewer patents held by women. It is unclear how non–career oriented activities, whether alone or together, particularly raising children, might interfere with both career advancement and compensation from industry, according to the authors.
Coauthor Ann C. Raldow, MD, also of the department of radiation oncology at UCLA, acknowledged in an interview that any of the potential explanations, such as the choice not to choose to pursue industry relationships, might be valid. However, she suggested that this issue deserves further exploration.
“Of greater concern is the possibility that this observed disparity may be a proxy for the systemic inequalities that female physicians have in radiation oncology. A first step in clarifying the origin of this gap could be incorporating industry-related questions into a workforce survey,” Dr. Raldow said.
If this step demonstrates a true disparity, “the most relevant metric may be female physicians receiving a percentage of total industry funding that corresponds to their representation in the field,” she added.
SOURCE: Weng JK et al. JAMA Netw Open. 2019 Jan. 25. doi: 10.1001/jamanetworkopen.2018.7377.
In the year 2016, female radiation oncologists received less money than did male radiation oncologists from industry across every type of compensation evaluated, according to an analysis of data from the Centers for Medicare & Medicated Services Open Payments program.
These sex disparities included a median $1,000 less for consulting (P = .005), $500 less for honoraria (P = .005) and $135 less for research payments, although the difference in this latter category fell short of statistical significance (P = .08), reported Julius K. Weng, department of radiation oncology, University of California, Los Angeles, and his colleagues, in JAMA Network Open.
The CMS Open Payments Program, which is part of the Physician Payments Sunshine Act, made the study possible. The CMS data were intended to allow evaluation of potential conflicts of interest, but they were amenable to compare payments by gender. For this purpose, the investigators determined gender by first name, which was confirmed by Internet search when ambiguous.
At least one industry payment in 2016 was made to 61.4% of 1,164 female and 70.4% of 3,319 male radiation oncologists included in this retrospective cross-sectional study.
Of research funding, only 20.7% went to women even though they represented 25.9% of radiation oncologists in the United States at that time. In other categories, such as honoraria or consulting, the proportion of compensation going to females was even lower, never exceeding 15%.
In the United States, about one-third of radiation oncologists are women, according to the authors of this study. This is substantially lower than the proportion in many other specialties and is far below that of current medical school enrollment, where women are now in a slight majority.
It cannot be ascertained from these data why industry compensation was lower for women, but the authors offered numerous potential explanations including the possibility that more female than male radiation oncologists do not elect to pursue relationships with industry. They labeled such relationships as “controversial” due to potential conflicts of interest.
Among the theories put forth are those that have been proposed to explain other sex disparities, including lower salaries and slower promotion, in medicine and elsewhere.
For one, it has been suggested that “agentic traits” of men might propel them to seek opportunities more aggressively, compared with women, who have “historically been associated with communal qualities,” according to the authors.
If due to gender bias, disparities may also accumulate over time as “downstream consequences of sex gaps experienced early in a female physician’s career,” the authors stated. They noted that women have a lower proportion of leadership positions in radiation oncology than predicted by their numbers in the specialty.
The disparity in industry partnerships and compensation is a relevant measure of sex disparity because these are associated with “substantial career advantages,” according to the authors of this study. In addition to the advantages of research funding, they believe these include association with important signs of success in academic clinical medicine, such being identified as a key opinion leader.
One limitation of the CMS data regarding industry payments is that the information is derived from self-reports. In 2016, the CMS Open Payments Program was in its fourth year, which the authors suggested had more complete information on industry payments than prior years because of initiatives to improve reporting compliance.
The lower payments from compensation are likely to be related to other gender disparities in radiation oncology, such as lower publication productivity and fewer patents held by women. It is unclear how non–career oriented activities, whether alone or together, particularly raising children, might interfere with both career advancement and compensation from industry, according to the authors.
Coauthor Ann C. Raldow, MD, also of the department of radiation oncology at UCLA, acknowledged in an interview that any of the potential explanations, such as the choice not to choose to pursue industry relationships, might be valid. However, she suggested that this issue deserves further exploration.
“Of greater concern is the possibility that this observed disparity may be a proxy for the systemic inequalities that female physicians have in radiation oncology. A first step in clarifying the origin of this gap could be incorporating industry-related questions into a workforce survey,” Dr. Raldow said.
If this step demonstrates a true disparity, “the most relevant metric may be female physicians receiving a percentage of total industry funding that corresponds to their representation in the field,” she added.
SOURCE: Weng JK et al. JAMA Netw Open. 2019 Jan. 25. doi: 10.1001/jamanetworkopen.2018.7377.
In the year 2016, female radiation oncologists received less money than did male radiation oncologists from industry across every type of compensation evaluated, according to an analysis of data from the Centers for Medicare & Medicated Services Open Payments program.
These sex disparities included a median $1,000 less for consulting (P = .005), $500 less for honoraria (P = .005) and $135 less for research payments, although the difference in this latter category fell short of statistical significance (P = .08), reported Julius K. Weng, department of radiation oncology, University of California, Los Angeles, and his colleagues, in JAMA Network Open.
The CMS Open Payments Program, which is part of the Physician Payments Sunshine Act, made the study possible. The CMS data were intended to allow evaluation of potential conflicts of interest, but they were amenable to compare payments by gender. For this purpose, the investigators determined gender by first name, which was confirmed by Internet search when ambiguous.
At least one industry payment in 2016 was made to 61.4% of 1,164 female and 70.4% of 3,319 male radiation oncologists included in this retrospective cross-sectional study.
Of research funding, only 20.7% went to women even though they represented 25.9% of radiation oncologists in the United States at that time. In other categories, such as honoraria or consulting, the proportion of compensation going to females was even lower, never exceeding 15%.
In the United States, about one-third of radiation oncologists are women, according to the authors of this study. This is substantially lower than the proportion in many other specialties and is far below that of current medical school enrollment, where women are now in a slight majority.
It cannot be ascertained from these data why industry compensation was lower for women, but the authors offered numerous potential explanations including the possibility that more female than male radiation oncologists do not elect to pursue relationships with industry. They labeled such relationships as “controversial” due to potential conflicts of interest.
Among the theories put forth are those that have been proposed to explain other sex disparities, including lower salaries and slower promotion, in medicine and elsewhere.
For one, it has been suggested that “agentic traits” of men might propel them to seek opportunities more aggressively, compared with women, who have “historically been associated with communal qualities,” according to the authors.
If due to gender bias, disparities may also accumulate over time as “downstream consequences of sex gaps experienced early in a female physician’s career,” the authors stated. They noted that women have a lower proportion of leadership positions in radiation oncology than predicted by their numbers in the specialty.
The disparity in industry partnerships and compensation is a relevant measure of sex disparity because these are associated with “substantial career advantages,” according to the authors of this study. In addition to the advantages of research funding, they believe these include association with important signs of success in academic clinical medicine, such being identified as a key opinion leader.
One limitation of the CMS data regarding industry payments is that the information is derived from self-reports. In 2016, the CMS Open Payments Program was in its fourth year, which the authors suggested had more complete information on industry payments than prior years because of initiatives to improve reporting compliance.
The lower payments from compensation are likely to be related to other gender disparities in radiation oncology, such as lower publication productivity and fewer patents held by women. It is unclear how non–career oriented activities, whether alone or together, particularly raising children, might interfere with both career advancement and compensation from industry, according to the authors.
Coauthor Ann C. Raldow, MD, also of the department of radiation oncology at UCLA, acknowledged in an interview that any of the potential explanations, such as the choice not to choose to pursue industry relationships, might be valid. However, she suggested that this issue deserves further exploration.
“Of greater concern is the possibility that this observed disparity may be a proxy for the systemic inequalities that female physicians have in radiation oncology. A first step in clarifying the origin of this gap could be incorporating industry-related questions into a workforce survey,” Dr. Raldow said.
If this step demonstrates a true disparity, “the most relevant metric may be female physicians receiving a percentage of total industry funding that corresponds to their representation in the field,” she added.
SOURCE: Weng JK et al. JAMA Netw Open. 2019 Jan. 25. doi: 10.1001/jamanetworkopen.2018.7377.
FROM JAMA NETWORK OPEN
Key clinical point: Sex disparity has been found for payments made by industry to radiation oncologists.
Major finding: Females were less likely to receive any industry money (61.4% vs. 70.4%) and received smaller amounts in all categories.
Study details: Retrospective cross-sectional study.
Disclosures: No conflicts of interest were reported.
Source: Weng JK et al. JAMA Netw Open. 2019 Jan. 25. doi: 10.1001/jamanetworkopen.2018.7377.
Things We Do for No Reason: Prescribing Docusate for Constipation in Hospitalized Adults
The “Things We Do for No Reason” (TWDFNR) series reviews practices that have become common parts of hospital care but which may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent “black and white” conclusions or clinical practice standards but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion.
Click here for the Choosing Wisely website.

CASE PRESENTATION
An 80-year-old woman with no significant past medical history presents with a mechanical fall. X-rays are notable for a right hip fracture. She is treated with morphine for analgesia and evaluated by orthopedic surgery for surgical repair. The hospitalist recognizes that this patient is at high risk for constipation and orders docusate for prevention of constipation.
BACKGROUND
Constipation is a highly prevalent problem in all practice settings, especially in the hospital, affecting two out of five hospitalized patients.1 Multiple factors in the inpatient setting contribute to constipation, including decreased mobility, medical comorbidities, postsurgical ileus, anesthetics, and medications such as opioid analgesics. Furthermore, the inpatient population is aging in parallel with the general population and constipation is more common in the elderly, likely owing to a combination of decreased muscle mass and impaired function of autonomic nerves.2 Consequently, inpatient providers frequently treat constipation or try to prevent it using stool softeners or laxatives.
One of the most commonly prescribed agents, regardless of medical specialty, is docusate, also known as dioctyl sulfosuccinate or by its brand name, Colace. A study from McGill University Health Centre in Montreal, Canada reported that docusate was the most frequently prescribed laxative, accounting for 64% of laxative medication doses, with associated costs approaching $60,000 per year.3 Direct drug costs accounted for a quarter of the expenses, and the remaining three quarters were estimated labor costs for administration. Medical and surgical admissions shared similar proportions of usage, with an average of 10 doses of docusate per admission across 17,064 admissions. Furthermore, half of the patients were prescribed docusate upon discharge. The authors extrapolated their data to suggest that total healthcare spending in North America on docusate products likely exceeds $100,000,000 yearly. A second study from Toronto found that 15% of all hospitalized patients are prescribed at least one dose of docusate, and that one-third of all new inpatient prescriptions are continued at discharge.4
WHY YOU THINK DOCUSATE MIGHT BE HELPFUL FOR CONSTIPATION
Docusate is thought to act as a detergent to retain water in the stool, thereby acting as a stool softener to facilitate stool passage. Physicians have prescribed docusate for decades, and attendings have passed down the practice of prescribing docusate for constipation to medical trainees for generations. The initial docusate studies showed promise, as it softened the stool by increasing its water content and made it easier to pass through the intestines.5 One of the earliest human studies compared docusate to an unspecified placebo in 35 elderly patients with chronic atonic constipation and found a decreased need for enemas.6 Some other observational studies also reported a decreased need for manual disimpactions and enemas in elderly populations.7,8 One randomized, controlled trial from 1968 showed an increased frequency of bowel movements compared to placebo, but it excluded half of the enrolled patients because they had a positive placebo response.9 Since those early studies from the 1950s and 1960s, docusate remains widely accepted as an effective stool softener with positive endorsements from hospital formularies and order sets and patient information sheets such as the JAMA Patient Page.10 Furthermore, the World Health Organization lists docusate as an “essential medicine,” reinforcing the notion that it is effective.11
WHY THERE IS NO REASON TO PRESCRIBE DOCUSATE FOR CONSTIPATION
Despite common practice, the efficacy of docusate as a stool softener has not been borne out by rigorous scientific data. On the contrary, multiple randomized controlled trials have failed to show any significant efficacy of this drug over placebo (Table).
The initial trial in 1976 studied 34 elderly patients on a general medical ward for prophylaxis of constipation.12 They randomized patients to 100 mg twice daily of docusate sodium versus a control group that did not receive any type of laxative. The number of bowel movements and their character served as the measured outcomes. The study demonstrated no statistically significant differences in the frequency and character of bowel movements between the docusate and placebo groups. Even at that time, the authors questioned whether docusate had any efficacy at all: “[w]hether the drug actually offers anything beyond a placebo effect in preventing constipation is in doubt.”
Another trial in 1978 studied 46 elderly, institutionalized patients with chronic functional constipation.13 All patients underwent a two-week placebo period followed by a three-week treatment period with three arms of randomization: docusate sodium 100 mg daily, docusate sodium 100 mg twice daily, or docusate calcium 240 mg daily. Patients received enemas or suppositories if required. All three arms showed an increase in the average number of natural bowel movements when compared to each patient’s own placebo period, but only the arm with docusate calcium reached statistical significance (P < .02). According to the authors, none of the therapies appeared to have a significant effect on stool consistency. The authors hypothesized that the higher dose given to the docusate calcium arm may have been the reason for the apparent efficacy in this cohort. As such, studies with higher doses of docusate calcium would be reasonable.
A third study in 1985 compared docusate sodium 100 mg three times daily versus placebo in six healthy patients with ileostomies and six healthy volunteers.14 Therapy with docusate “had no effect on stool weight, stool frequency, stool water, or mean transit time.”
Another study in 1991 evaluated 15 elderly nursing home residents with a randomized, double-blind crossover design.15 Subjects received 240 mg twice daily of docusate calcium versus placebo for three weeks and then crossed over to other arm after a two-week wash-out period. The investigators found no difference in the number of bowel movements per week or in the need for additional laxatives between the two study periods. There were also no differences in the patients’ subjective experience of constipation or discomfort with defecation.
Larger studies were subsequently initiated in more recent years. In 1998, a randomized controlled trial in 170 subjects with chronic idiopathic constipation compared psyllium 5.1 g twice daily and docusate sodium 100 mg twice daily with a corresponding placebo in each arm for a treatment duration of two weeks after a two-week placebo baseline period.16 Psyllium was found to increase stool water content and stool water weight over the baseline period, while docusate essentially had no effect on stool water content or water weight. Furthermore, by treatment week 2, psyllium demonstrated an increase in the frequency of bowel movements, whereas docusate did not. It should be noted that this study was funded by Procter & Gamble, which manufactures Metamucil, a popular brand of psyllium.
Lastly, the most recent randomized controlled trial was published in 2013. It included 74 hospice patients in Canada, comparing docusate 200 mg and sennosides twice daily versus placebo and sennosides for 10 days. The study found no difference in stool frequency, volume, or consistency between docusate and placebo.17
A number of systematic reviews have studied the literature on bowel regimens and have noted the paucity of high-quality data supporting the efficacy of docusate, despite its widespread use.18-22 With these weak data, multiple authors have advocated for removing docusate from hospital formularies and using hospitalizations as an opportunity to deprescribe this medication to reduce polypharmacy. 3,4,23
Although docusate is considered a benign therapy, there is certainly potential for harm to the patient and detrimental effects on the healthcare system. Patients commonly complain about the unpleasant taste and lingering aftertaste, which may lead to decreased oral intake and worsening nutritional status.23 Furthermore, docusate may impact the absorption and effectiveness of other proven treatments.23 Perhaps the most important harm is that providers needlessly wait for docusate to fail before prescribing effective therapies for constipation. This process negatively impacts patient satisfaction and potentially increases healthcare costs if hospital length of stay is increased. Another important consideration is that patients may refuse truly necessary medications due to the excessive pill burden.
Costs to the healthcare system are increased needlessly when medications that do not improve outcomes are prescribed. Although the individual pill cost is low, the widespread use and the associated pharmacy and nursing resources required for administration create an estimated cost for docusate over $100,000,000 per year for North America alone.3 The staff time required for administration may prevent healthcare personnel from engaging in other more valuable tasks. Additionally, every medication order creates an opportunity for medical error. Lastly, bacteria were recently found contaminating the liquid formulation, which carries its own obvious implications if patients develop iatrogenic infections.24
WHAT YOU SHOULD DO INSTEAD
Instead of using docusate, prescribe agents with established efficacy. In 2006, a systematic review published in the American Journal of Gastroenterology graded the evidence behind different therapies for chronic constipation.21 They found good evidence (Grade A) to support the use of polyethylene glycol (PEG), while psyllium and lactulose had moderate evidence (Grade B) to support their use. All other currently available agents that were reviewed had poor evidence to support their use. A more recent study in people prescribed opioids similarly found evidence to support the use of polyethylene glycol, lactulose, and sennosides.25 Lastly, the 2016 guidelines from the American Society of Colon and Rectal Surgeons do not mention docusate, though they comment on the paucity of data on stool softeners. Their recommendations for laxative therapy are similar to those of the previously discussed reviews.26 Ultimately, the choice of therapy, pharmacological and nonpharmacological, should be individualized for each patient based on the clinical context and cause of constipation. Nonpharmacologic treatments include dietary modification, mobilization, chewing gum, and biofeedback. If pharmacotherapy is required, use laxatives with the strongest evidence.
RECOMMENDATIONS
- In patients with constipation or at risk for constipation, use laxatives with proven efficacy (such as polyethylene glycol, lactulose, psyllium, or sennosides) for treatment or prophylaxis of constipation instead of using docusate.
- Discuss de-prescription for patients using docusate prior to admission.
- Remove docusate from your hospital formulary.
CONCLUSION
Docusate is commonly used for the treatment and prevention of constipation in hospitalized patients, with significant associated costs. This common practice continues despite little evidence supporting its efficacy and many trials failing to show benefits over placebo. Decreased utilization of ineffective therapies such as docusate is recommended. Returning to the case presentation, the hospitalist should start the patient on alternative therapies, instead of docusate, such as polyethylene glycol, lactulose, psyllium, or sennosides, which have better evidence supporting their use.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason?” Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason” topics by emailing TWDFNR@hospitalmedicine.org.
Disclosures
All authors deny any relevant conflict of interest with the attached manuscript.
1. Noiesen E, Trosborg I, Bager L, Herning M, Lyngby C, Konradsen H. Constipation--prevalence and incidence among medical patients acutely admitted to hospital with a medical condition. J Clin Nurs. 2014;23(15-16):2295-2302. doi: 10.1111/jocn.12511.
2. De Giorgio R, Ruggeri E, Stanghellini V, Eusebi LH, Bazzoli F, Chiarioni G. Chronic constipation in the elderly: a primer for the gastroenterologist. BMC Gastroenterol. 2015;15:130. doi: 10.1186/s12876-015-0366-3.
3. Lee TC, McDonald EG, Bonnici A, Tamblyn R. Pattern of inpatient laxative use: waste not, want not. JAMA Intern Med. 2016;176(8):1216-1217. doi: 10.1001/jamainternmed.2016.2775.
4. MacMillan TE, Kamali R, Cavalcanti RB. Missed opportunity to deprescribe: docusate for constipation in medical inpatients. Am J Med. 2016;129(9):1001 e1001-1007. doi: 10.1016/j.amjmed.2016.04.008.
5. Spiesman MG, Malow L. New fecal softener (doxinate) in the treatment of constipation. J Lancet. 1956;76(6):164-167.
6. Harris R. Constipation in geriatrics; management with dioctyl sodium sulfosuccinate. Am J Dig Dis. Sep 1957;2(9):487-492.
7. Smigel JO, Lowe KJ, Hosp PH, Gibson JH. Constipation in elderly patients; treatment with dioctyl sodium sulfosuccinate and dioctyl sodium sulfosuccinate plus peristim. Med Times. 1958;86(12):1521-1526.
8. Wilson JL, Dickinson DG. Use of dioctyl sodium sulfosuccinate (aerosol O.T.) for severe constipation. J Am Med Assoc. 1955;158(4):261-263. doi: 10.1001/jama.1955.02960040019006a.
9. Hyland CM, Foran JD. Dioctyl sodium sulphosuccinate as a laxative in the elderly. Practitioner. 1968;200(199):698-699.
10. Jin J. JAMA patient page. Over-the-counter laxatives. JAMA. 2014;312(11):1167. doi: 10.1001/jama.2014.2078.
11. 19th WHO Model List of Essential Medicines (April 2015). 2015; http://www.who.int/medicines/publications/essentialmedicines/en/.
12. Goodman J, Pang J, Bessman AN. Dioctyl sodium sulfosuccinate- an ineffective prophylactic laxative. J Chronic Dis. 1976;29(1):59-63. doi: 10.1016/0021-9681(76)90068-0.
13. Fain AM, Susat R, Herring M, Dorton K. Treatment of constipation in geriatric and chronically ill patients: a comparison. South Med J. 1978;71(6):677-680.
14. Chapman RW, Sillery J, Fontana DD, Matthys C, Saunders DR. Effect of oral dioctyl sodium sulfosuccinate on intake-output studies of human small and large intestine. Gastroenterology. 1985;89(3):489-493. doi: 10.1016/0016-5085(85)90441-X.
15. Castle SC, Cantrell M, Israel DS, Samuelson MJ. Constipation prevention: empiric use of stool softeners questioned. Geriatrics. 1991;46(11):84-86.
16. McRorie JW, Daggy BP, Morel JG, Diersing PS, Miner PB, Robinson M. Psyllium is superior to docusate sodium for treatment of chronic constipation. Aliment Pharmacol Ther. 1998;12(5):491-497. doi: 10.1046/j.1365-2036.1998.00336.x.
17. Tarumi Y, Wilson MP, Szafran O, Spooner GR. Randomized, double-blind, placebo-controlled trial of oral docusate in the management of constipation in hospice patients. J Pain Symptom Manage. 2013;45(1):2-13. doi: 10.1016/j.jpainsymman.2012.02.008.
18. Candy B, Jones L, Larkin PJ, Vickerstaff V, Tookman A, Stone P. Laxatives for the management of constipation in people receiving palliative care. Cochrane Database Syst Rev. 2015(5):CD003448.
19. Hurdon V, Viola R, Schroder C. How useful is docusate in patients at risk for constipation? A systematic review of the evidence in the chronically ill. J Pain Symptom Manage. 2000;19(2):130-136. doi: 10.1016/S0885-3924(99)00157-8.
20. Pare P, Fedorak RN. Systematic review of stimulant and nonstimulant laxatives for the treatment of functional constipation. Can J Gastroenterol Hepatol. 2014;28(10):549-557.
21. Ramkumar D, Rao SS. Efficacy and safety of traditional medical therapies for chronic constipation: systematic review. Am J Gastroenterol. 2005;100(4):936-971. doi: 10.1111/j.1572-0241.2005.40925.x
22. Health CAfDaTi. Dioctyl sulfosuccinate or docusate (calcium or sodium) for the prevention or management of constipation: a review of the clinical effectiveness. Ottawa (ON)2014.
23. McKee KY, Widera E. Habitual prescribing of laxatives-it’s time to flush outdated protocols down the drain. JAMA Intern Med. 2016;176(8):1217-1219. doi: 10.1001/jamainternmed.2016.2780.
24. Marquez L, Jones KN, Whaley EM, et al. An outbreak of burkholderia cepacia complex infections associated with contaminated liquid docusate. Infect Control Hosp Epidemiol. 2017;38(5):567-573. doi: 10.1017/ice.2017.11.
25. Ahmedzai SH, Boland J. Constipation in people prescribed opioids. BMJ Clin Evid. 2010;2010.
26. Paquette IM, Varma M, Ternent C, et al. The American society of colon and rectal surgeons’ clinical practice guideline for the evaluation and management of constipation. Dis Colon Rectum. 2016;59(6):479-492. doi: 10.1097/DCR.0000000000000599
The “Things We Do for No Reason” (TWDFNR) series reviews practices that have become common parts of hospital care but which may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent “black and white” conclusions or clinical practice standards but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion.
Click here for the Choosing Wisely website.

CASE PRESENTATION
An 80-year-old woman with no significant past medical history presents with a mechanical fall. X-rays are notable for a right hip fracture. She is treated with morphine for analgesia and evaluated by orthopedic surgery for surgical repair. The hospitalist recognizes that this patient is at high risk for constipation and orders docusate for prevention of constipation.
BACKGROUND
Constipation is a highly prevalent problem in all practice settings, especially in the hospital, affecting two out of five hospitalized patients.1 Multiple factors in the inpatient setting contribute to constipation, including decreased mobility, medical comorbidities, postsurgical ileus, anesthetics, and medications such as opioid analgesics. Furthermore, the inpatient population is aging in parallel with the general population and constipation is more common in the elderly, likely owing to a combination of decreased muscle mass and impaired function of autonomic nerves.2 Consequently, inpatient providers frequently treat constipation or try to prevent it using stool softeners or laxatives.
One of the most commonly prescribed agents, regardless of medical specialty, is docusate, also known as dioctyl sulfosuccinate or by its brand name, Colace. A study from McGill University Health Centre in Montreal, Canada reported that docusate was the most frequently prescribed laxative, accounting for 64% of laxative medication doses, with associated costs approaching $60,000 per year.3 Direct drug costs accounted for a quarter of the expenses, and the remaining three quarters were estimated labor costs for administration. Medical and surgical admissions shared similar proportions of usage, with an average of 10 doses of docusate per admission across 17,064 admissions. Furthermore, half of the patients were prescribed docusate upon discharge. The authors extrapolated their data to suggest that total healthcare spending in North America on docusate products likely exceeds $100,000,000 yearly. A second study from Toronto found that 15% of all hospitalized patients are prescribed at least one dose of docusate, and that one-third of all new inpatient prescriptions are continued at discharge.4
WHY YOU THINK DOCUSATE MIGHT BE HELPFUL FOR CONSTIPATION
Docusate is thought to act as a detergent to retain water in the stool, thereby acting as a stool softener to facilitate stool passage. Physicians have prescribed docusate for decades, and attendings have passed down the practice of prescribing docusate for constipation to medical trainees for generations. The initial docusate studies showed promise, as it softened the stool by increasing its water content and made it easier to pass through the intestines.5 One of the earliest human studies compared docusate to an unspecified placebo in 35 elderly patients with chronic atonic constipation and found a decreased need for enemas.6 Some other observational studies also reported a decreased need for manual disimpactions and enemas in elderly populations.7,8 One randomized, controlled trial from 1968 showed an increased frequency of bowel movements compared to placebo, but it excluded half of the enrolled patients because they had a positive placebo response.9 Since those early studies from the 1950s and 1960s, docusate remains widely accepted as an effective stool softener with positive endorsements from hospital formularies and order sets and patient information sheets such as the JAMA Patient Page.10 Furthermore, the World Health Organization lists docusate as an “essential medicine,” reinforcing the notion that it is effective.11
WHY THERE IS NO REASON TO PRESCRIBE DOCUSATE FOR CONSTIPATION
Despite common practice, the efficacy of docusate as a stool softener has not been borne out by rigorous scientific data. On the contrary, multiple randomized controlled trials have failed to show any significant efficacy of this drug over placebo (Table).
The initial trial in 1976 studied 34 elderly patients on a general medical ward for prophylaxis of constipation.12 They randomized patients to 100 mg twice daily of docusate sodium versus a control group that did not receive any type of laxative. The number of bowel movements and their character served as the measured outcomes. The study demonstrated no statistically significant differences in the frequency and character of bowel movements between the docusate and placebo groups. Even at that time, the authors questioned whether docusate had any efficacy at all: “[w]hether the drug actually offers anything beyond a placebo effect in preventing constipation is in doubt.”
Another trial in 1978 studied 46 elderly, institutionalized patients with chronic functional constipation.13 All patients underwent a two-week placebo period followed by a three-week treatment period with three arms of randomization: docusate sodium 100 mg daily, docusate sodium 100 mg twice daily, or docusate calcium 240 mg daily. Patients received enemas or suppositories if required. All three arms showed an increase in the average number of natural bowel movements when compared to each patient’s own placebo period, but only the arm with docusate calcium reached statistical significance (P < .02). According to the authors, none of the therapies appeared to have a significant effect on stool consistency. The authors hypothesized that the higher dose given to the docusate calcium arm may have been the reason for the apparent efficacy in this cohort. As such, studies with higher doses of docusate calcium would be reasonable.
A third study in 1985 compared docusate sodium 100 mg three times daily versus placebo in six healthy patients with ileostomies and six healthy volunteers.14 Therapy with docusate “had no effect on stool weight, stool frequency, stool water, or mean transit time.”
Another study in 1991 evaluated 15 elderly nursing home residents with a randomized, double-blind crossover design.15 Subjects received 240 mg twice daily of docusate calcium versus placebo for three weeks and then crossed over to other arm after a two-week wash-out period. The investigators found no difference in the number of bowel movements per week or in the need for additional laxatives between the two study periods. There were also no differences in the patients’ subjective experience of constipation or discomfort with defecation.
Larger studies were subsequently initiated in more recent years. In 1998, a randomized controlled trial in 170 subjects with chronic idiopathic constipation compared psyllium 5.1 g twice daily and docusate sodium 100 mg twice daily with a corresponding placebo in each arm for a treatment duration of two weeks after a two-week placebo baseline period.16 Psyllium was found to increase stool water content and stool water weight over the baseline period, while docusate essentially had no effect on stool water content or water weight. Furthermore, by treatment week 2, psyllium demonstrated an increase in the frequency of bowel movements, whereas docusate did not. It should be noted that this study was funded by Procter & Gamble, which manufactures Metamucil, a popular brand of psyllium.
Lastly, the most recent randomized controlled trial was published in 2013. It included 74 hospice patients in Canada, comparing docusate 200 mg and sennosides twice daily versus placebo and sennosides for 10 days. The study found no difference in stool frequency, volume, or consistency between docusate and placebo.17
A number of systematic reviews have studied the literature on bowel regimens and have noted the paucity of high-quality data supporting the efficacy of docusate, despite its widespread use.18-22 With these weak data, multiple authors have advocated for removing docusate from hospital formularies and using hospitalizations as an opportunity to deprescribe this medication to reduce polypharmacy. 3,4,23
Although docusate is considered a benign therapy, there is certainly potential for harm to the patient and detrimental effects on the healthcare system. Patients commonly complain about the unpleasant taste and lingering aftertaste, which may lead to decreased oral intake and worsening nutritional status.23 Furthermore, docusate may impact the absorption and effectiveness of other proven treatments.23 Perhaps the most important harm is that providers needlessly wait for docusate to fail before prescribing effective therapies for constipation. This process negatively impacts patient satisfaction and potentially increases healthcare costs if hospital length of stay is increased. Another important consideration is that patients may refuse truly necessary medications due to the excessive pill burden.
Costs to the healthcare system are increased needlessly when medications that do not improve outcomes are prescribed. Although the individual pill cost is low, the widespread use and the associated pharmacy and nursing resources required for administration create an estimated cost for docusate over $100,000,000 per year for North America alone.3 The staff time required for administration may prevent healthcare personnel from engaging in other more valuable tasks. Additionally, every medication order creates an opportunity for medical error. Lastly, bacteria were recently found contaminating the liquid formulation, which carries its own obvious implications if patients develop iatrogenic infections.24
WHAT YOU SHOULD DO INSTEAD
Instead of using docusate, prescribe agents with established efficacy. In 2006, a systematic review published in the American Journal of Gastroenterology graded the evidence behind different therapies for chronic constipation.21 They found good evidence (Grade A) to support the use of polyethylene glycol (PEG), while psyllium and lactulose had moderate evidence (Grade B) to support their use. All other currently available agents that were reviewed had poor evidence to support their use. A more recent study in people prescribed opioids similarly found evidence to support the use of polyethylene glycol, lactulose, and sennosides.25 Lastly, the 2016 guidelines from the American Society of Colon and Rectal Surgeons do not mention docusate, though they comment on the paucity of data on stool softeners. Their recommendations for laxative therapy are similar to those of the previously discussed reviews.26 Ultimately, the choice of therapy, pharmacological and nonpharmacological, should be individualized for each patient based on the clinical context and cause of constipation. Nonpharmacologic treatments include dietary modification, mobilization, chewing gum, and biofeedback. If pharmacotherapy is required, use laxatives with the strongest evidence.
RECOMMENDATIONS
- In patients with constipation or at risk for constipation, use laxatives with proven efficacy (such as polyethylene glycol, lactulose, psyllium, or sennosides) for treatment or prophylaxis of constipation instead of using docusate.
- Discuss de-prescription for patients using docusate prior to admission.
- Remove docusate from your hospital formulary.
CONCLUSION
Docusate is commonly used for the treatment and prevention of constipation in hospitalized patients, with significant associated costs. This common practice continues despite little evidence supporting its efficacy and many trials failing to show benefits over placebo. Decreased utilization of ineffective therapies such as docusate is recommended. Returning to the case presentation, the hospitalist should start the patient on alternative therapies, instead of docusate, such as polyethylene glycol, lactulose, psyllium, or sennosides, which have better evidence supporting their use.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason?” Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason” topics by emailing TWDFNR@hospitalmedicine.org.
Disclosures
All authors deny any relevant conflict of interest with the attached manuscript.
The “Things We Do for No Reason” (TWDFNR) series reviews practices that have become common parts of hospital care but which may provide little value to our patients. Practices reviewed in the TWDFNR series do not represent “black and white” conclusions or clinical practice standards but are meant as a starting place for research and active discussions among hospitalists and patients. We invite you to be part of that discussion.
Click here for the Choosing Wisely website.

CASE PRESENTATION
An 80-year-old woman with no significant past medical history presents with a mechanical fall. X-rays are notable for a right hip fracture. She is treated with morphine for analgesia and evaluated by orthopedic surgery for surgical repair. The hospitalist recognizes that this patient is at high risk for constipation and orders docusate for prevention of constipation.
BACKGROUND
Constipation is a highly prevalent problem in all practice settings, especially in the hospital, affecting two out of five hospitalized patients.1 Multiple factors in the inpatient setting contribute to constipation, including decreased mobility, medical comorbidities, postsurgical ileus, anesthetics, and medications such as opioid analgesics. Furthermore, the inpatient population is aging in parallel with the general population and constipation is more common in the elderly, likely owing to a combination of decreased muscle mass and impaired function of autonomic nerves.2 Consequently, inpatient providers frequently treat constipation or try to prevent it using stool softeners or laxatives.
One of the most commonly prescribed agents, regardless of medical specialty, is docusate, also known as dioctyl sulfosuccinate or by its brand name, Colace. A study from McGill University Health Centre in Montreal, Canada reported that docusate was the most frequently prescribed laxative, accounting for 64% of laxative medication doses, with associated costs approaching $60,000 per year.3 Direct drug costs accounted for a quarter of the expenses, and the remaining three quarters were estimated labor costs for administration. Medical and surgical admissions shared similar proportions of usage, with an average of 10 doses of docusate per admission across 17,064 admissions. Furthermore, half of the patients were prescribed docusate upon discharge. The authors extrapolated their data to suggest that total healthcare spending in North America on docusate products likely exceeds $100,000,000 yearly. A second study from Toronto found that 15% of all hospitalized patients are prescribed at least one dose of docusate, and that one-third of all new inpatient prescriptions are continued at discharge.4
WHY YOU THINK DOCUSATE MIGHT BE HELPFUL FOR CONSTIPATION
Docusate is thought to act as a detergent to retain water in the stool, thereby acting as a stool softener to facilitate stool passage. Physicians have prescribed docusate for decades, and attendings have passed down the practice of prescribing docusate for constipation to medical trainees for generations. The initial docusate studies showed promise, as it softened the stool by increasing its water content and made it easier to pass through the intestines.5 One of the earliest human studies compared docusate to an unspecified placebo in 35 elderly patients with chronic atonic constipation and found a decreased need for enemas.6 Some other observational studies also reported a decreased need for manual disimpactions and enemas in elderly populations.7,8 One randomized, controlled trial from 1968 showed an increased frequency of bowel movements compared to placebo, but it excluded half of the enrolled patients because they had a positive placebo response.9 Since those early studies from the 1950s and 1960s, docusate remains widely accepted as an effective stool softener with positive endorsements from hospital formularies and order sets and patient information sheets such as the JAMA Patient Page.10 Furthermore, the World Health Organization lists docusate as an “essential medicine,” reinforcing the notion that it is effective.11
WHY THERE IS NO REASON TO PRESCRIBE DOCUSATE FOR CONSTIPATION
Despite common practice, the efficacy of docusate as a stool softener has not been borne out by rigorous scientific data. On the contrary, multiple randomized controlled trials have failed to show any significant efficacy of this drug over placebo (Table).
The initial trial in 1976 studied 34 elderly patients on a general medical ward for prophylaxis of constipation.12 They randomized patients to 100 mg twice daily of docusate sodium versus a control group that did not receive any type of laxative. The number of bowel movements and their character served as the measured outcomes. The study demonstrated no statistically significant differences in the frequency and character of bowel movements between the docusate and placebo groups. Even at that time, the authors questioned whether docusate had any efficacy at all: “[w]hether the drug actually offers anything beyond a placebo effect in preventing constipation is in doubt.”
Another trial in 1978 studied 46 elderly, institutionalized patients with chronic functional constipation.13 All patients underwent a two-week placebo period followed by a three-week treatment period with three arms of randomization: docusate sodium 100 mg daily, docusate sodium 100 mg twice daily, or docusate calcium 240 mg daily. Patients received enemas or suppositories if required. All three arms showed an increase in the average number of natural bowel movements when compared to each patient’s own placebo period, but only the arm with docusate calcium reached statistical significance (P < .02). According to the authors, none of the therapies appeared to have a significant effect on stool consistency. The authors hypothesized that the higher dose given to the docusate calcium arm may have been the reason for the apparent efficacy in this cohort. As such, studies with higher doses of docusate calcium would be reasonable.
A third study in 1985 compared docusate sodium 100 mg three times daily versus placebo in six healthy patients with ileostomies and six healthy volunteers.14 Therapy with docusate “had no effect on stool weight, stool frequency, stool water, or mean transit time.”
Another study in 1991 evaluated 15 elderly nursing home residents with a randomized, double-blind crossover design.15 Subjects received 240 mg twice daily of docusate calcium versus placebo for three weeks and then crossed over to other arm after a two-week wash-out period. The investigators found no difference in the number of bowel movements per week or in the need for additional laxatives between the two study periods. There were also no differences in the patients’ subjective experience of constipation or discomfort with defecation.
Larger studies were subsequently initiated in more recent years. In 1998, a randomized controlled trial in 170 subjects with chronic idiopathic constipation compared psyllium 5.1 g twice daily and docusate sodium 100 mg twice daily with a corresponding placebo in each arm for a treatment duration of two weeks after a two-week placebo baseline period.16 Psyllium was found to increase stool water content and stool water weight over the baseline period, while docusate essentially had no effect on stool water content or water weight. Furthermore, by treatment week 2, psyllium demonstrated an increase in the frequency of bowel movements, whereas docusate did not. It should be noted that this study was funded by Procter & Gamble, which manufactures Metamucil, a popular brand of psyllium.
Lastly, the most recent randomized controlled trial was published in 2013. It included 74 hospice patients in Canada, comparing docusate 200 mg and sennosides twice daily versus placebo and sennosides for 10 days. The study found no difference in stool frequency, volume, or consistency between docusate and placebo.17
A number of systematic reviews have studied the literature on bowel regimens and have noted the paucity of high-quality data supporting the efficacy of docusate, despite its widespread use.18-22 With these weak data, multiple authors have advocated for removing docusate from hospital formularies and using hospitalizations as an opportunity to deprescribe this medication to reduce polypharmacy. 3,4,23
Although docusate is considered a benign therapy, there is certainly potential for harm to the patient and detrimental effects on the healthcare system. Patients commonly complain about the unpleasant taste and lingering aftertaste, which may lead to decreased oral intake and worsening nutritional status.23 Furthermore, docusate may impact the absorption and effectiveness of other proven treatments.23 Perhaps the most important harm is that providers needlessly wait for docusate to fail before prescribing effective therapies for constipation. This process negatively impacts patient satisfaction and potentially increases healthcare costs if hospital length of stay is increased. Another important consideration is that patients may refuse truly necessary medications due to the excessive pill burden.
Costs to the healthcare system are increased needlessly when medications that do not improve outcomes are prescribed. Although the individual pill cost is low, the widespread use and the associated pharmacy and nursing resources required for administration create an estimated cost for docusate over $100,000,000 per year for North America alone.3 The staff time required for administration may prevent healthcare personnel from engaging in other more valuable tasks. Additionally, every medication order creates an opportunity for medical error. Lastly, bacteria were recently found contaminating the liquid formulation, which carries its own obvious implications if patients develop iatrogenic infections.24
WHAT YOU SHOULD DO INSTEAD
Instead of using docusate, prescribe agents with established efficacy. In 2006, a systematic review published in the American Journal of Gastroenterology graded the evidence behind different therapies for chronic constipation.21 They found good evidence (Grade A) to support the use of polyethylene glycol (PEG), while psyllium and lactulose had moderate evidence (Grade B) to support their use. All other currently available agents that were reviewed had poor evidence to support their use. A more recent study in people prescribed opioids similarly found evidence to support the use of polyethylene glycol, lactulose, and sennosides.25 Lastly, the 2016 guidelines from the American Society of Colon and Rectal Surgeons do not mention docusate, though they comment on the paucity of data on stool softeners. Their recommendations for laxative therapy are similar to those of the previously discussed reviews.26 Ultimately, the choice of therapy, pharmacological and nonpharmacological, should be individualized for each patient based on the clinical context and cause of constipation. Nonpharmacologic treatments include dietary modification, mobilization, chewing gum, and biofeedback. If pharmacotherapy is required, use laxatives with the strongest evidence.
RECOMMENDATIONS
- In patients with constipation or at risk for constipation, use laxatives with proven efficacy (such as polyethylene glycol, lactulose, psyllium, or sennosides) for treatment or prophylaxis of constipation instead of using docusate.
- Discuss de-prescription for patients using docusate prior to admission.
- Remove docusate from your hospital formulary.
CONCLUSION
Docusate is commonly used for the treatment and prevention of constipation in hospitalized patients, with significant associated costs. This common practice continues despite little evidence supporting its efficacy and many trials failing to show benefits over placebo. Decreased utilization of ineffective therapies such as docusate is recommended. Returning to the case presentation, the hospitalist should start the patient on alternative therapies, instead of docusate, such as polyethylene glycol, lactulose, psyllium, or sennosides, which have better evidence supporting their use.
Do you think this is a low-value practice? Is this truly a “Thing We Do for No Reason?” Share what you do in your practice and join in the conversation online by retweeting it on Twitter (#TWDFNR) and liking it on Facebook. We invite you to propose ideas for other “Things We Do for No Reason” topics by emailing TWDFNR@hospitalmedicine.org.
Disclosures
All authors deny any relevant conflict of interest with the attached manuscript.
1. Noiesen E, Trosborg I, Bager L, Herning M, Lyngby C, Konradsen H. Constipation--prevalence and incidence among medical patients acutely admitted to hospital with a medical condition. J Clin Nurs. 2014;23(15-16):2295-2302. doi: 10.1111/jocn.12511.
2. De Giorgio R, Ruggeri E, Stanghellini V, Eusebi LH, Bazzoli F, Chiarioni G. Chronic constipation in the elderly: a primer for the gastroenterologist. BMC Gastroenterol. 2015;15:130. doi: 10.1186/s12876-015-0366-3.
3. Lee TC, McDonald EG, Bonnici A, Tamblyn R. Pattern of inpatient laxative use: waste not, want not. JAMA Intern Med. 2016;176(8):1216-1217. doi: 10.1001/jamainternmed.2016.2775.
4. MacMillan TE, Kamali R, Cavalcanti RB. Missed opportunity to deprescribe: docusate for constipation in medical inpatients. Am J Med. 2016;129(9):1001 e1001-1007. doi: 10.1016/j.amjmed.2016.04.008.
5. Spiesman MG, Malow L. New fecal softener (doxinate) in the treatment of constipation. J Lancet. 1956;76(6):164-167.
6. Harris R. Constipation in geriatrics; management with dioctyl sodium sulfosuccinate. Am J Dig Dis. Sep 1957;2(9):487-492.
7. Smigel JO, Lowe KJ, Hosp PH, Gibson JH. Constipation in elderly patients; treatment with dioctyl sodium sulfosuccinate and dioctyl sodium sulfosuccinate plus peristim. Med Times. 1958;86(12):1521-1526.
8. Wilson JL, Dickinson DG. Use of dioctyl sodium sulfosuccinate (aerosol O.T.) for severe constipation. J Am Med Assoc. 1955;158(4):261-263. doi: 10.1001/jama.1955.02960040019006a.
9. Hyland CM, Foran JD. Dioctyl sodium sulphosuccinate as a laxative in the elderly. Practitioner. 1968;200(199):698-699.
10. Jin J. JAMA patient page. Over-the-counter laxatives. JAMA. 2014;312(11):1167. doi: 10.1001/jama.2014.2078.
11. 19th WHO Model List of Essential Medicines (April 2015). 2015; http://www.who.int/medicines/publications/essentialmedicines/en/.
12. Goodman J, Pang J, Bessman AN. Dioctyl sodium sulfosuccinate- an ineffective prophylactic laxative. J Chronic Dis. 1976;29(1):59-63. doi: 10.1016/0021-9681(76)90068-0.
13. Fain AM, Susat R, Herring M, Dorton K. Treatment of constipation in geriatric and chronically ill patients: a comparison. South Med J. 1978;71(6):677-680.
14. Chapman RW, Sillery J, Fontana DD, Matthys C, Saunders DR. Effect of oral dioctyl sodium sulfosuccinate on intake-output studies of human small and large intestine. Gastroenterology. 1985;89(3):489-493. doi: 10.1016/0016-5085(85)90441-X.
15. Castle SC, Cantrell M, Israel DS, Samuelson MJ. Constipation prevention: empiric use of stool softeners questioned. Geriatrics. 1991;46(11):84-86.
16. McRorie JW, Daggy BP, Morel JG, Diersing PS, Miner PB, Robinson M. Psyllium is superior to docusate sodium for treatment of chronic constipation. Aliment Pharmacol Ther. 1998;12(5):491-497. doi: 10.1046/j.1365-2036.1998.00336.x.
17. Tarumi Y, Wilson MP, Szafran O, Spooner GR. Randomized, double-blind, placebo-controlled trial of oral docusate in the management of constipation in hospice patients. J Pain Symptom Manage. 2013;45(1):2-13. doi: 10.1016/j.jpainsymman.2012.02.008.
18. Candy B, Jones L, Larkin PJ, Vickerstaff V, Tookman A, Stone P. Laxatives for the management of constipation in people receiving palliative care. Cochrane Database Syst Rev. 2015(5):CD003448.
19. Hurdon V, Viola R, Schroder C. How useful is docusate in patients at risk for constipation? A systematic review of the evidence in the chronically ill. J Pain Symptom Manage. 2000;19(2):130-136. doi: 10.1016/S0885-3924(99)00157-8.
20. Pare P, Fedorak RN. Systematic review of stimulant and nonstimulant laxatives for the treatment of functional constipation. Can J Gastroenterol Hepatol. 2014;28(10):549-557.
21. Ramkumar D, Rao SS. Efficacy and safety of traditional medical therapies for chronic constipation: systematic review. Am J Gastroenterol. 2005;100(4):936-971. doi: 10.1111/j.1572-0241.2005.40925.x
22. Health CAfDaTi. Dioctyl sulfosuccinate or docusate (calcium or sodium) for the prevention or management of constipation: a review of the clinical effectiveness. Ottawa (ON)2014.
23. McKee KY, Widera E. Habitual prescribing of laxatives-it’s time to flush outdated protocols down the drain. JAMA Intern Med. 2016;176(8):1217-1219. doi: 10.1001/jamainternmed.2016.2780.
24. Marquez L, Jones KN, Whaley EM, et al. An outbreak of burkholderia cepacia complex infections associated with contaminated liquid docusate. Infect Control Hosp Epidemiol. 2017;38(5):567-573. doi: 10.1017/ice.2017.11.
25. Ahmedzai SH, Boland J. Constipation in people prescribed opioids. BMJ Clin Evid. 2010;2010.
26. Paquette IM, Varma M, Ternent C, et al. The American society of colon and rectal surgeons’ clinical practice guideline for the evaluation and management of constipation. Dis Colon Rectum. 2016;59(6):479-492. doi: 10.1097/DCR.0000000000000599
1. Noiesen E, Trosborg I, Bager L, Herning M, Lyngby C, Konradsen H. Constipation--prevalence and incidence among medical patients acutely admitted to hospital with a medical condition. J Clin Nurs. 2014;23(15-16):2295-2302. doi: 10.1111/jocn.12511.
2. De Giorgio R, Ruggeri E, Stanghellini V, Eusebi LH, Bazzoli F, Chiarioni G. Chronic constipation in the elderly: a primer for the gastroenterologist. BMC Gastroenterol. 2015;15:130. doi: 10.1186/s12876-015-0366-3.
3. Lee TC, McDonald EG, Bonnici A, Tamblyn R. Pattern of inpatient laxative use: waste not, want not. JAMA Intern Med. 2016;176(8):1216-1217. doi: 10.1001/jamainternmed.2016.2775.
4. MacMillan TE, Kamali R, Cavalcanti RB. Missed opportunity to deprescribe: docusate for constipation in medical inpatients. Am J Med. 2016;129(9):1001 e1001-1007. doi: 10.1016/j.amjmed.2016.04.008.
5. Spiesman MG, Malow L. New fecal softener (doxinate) in the treatment of constipation. J Lancet. 1956;76(6):164-167.
6. Harris R. Constipation in geriatrics; management with dioctyl sodium sulfosuccinate. Am J Dig Dis. Sep 1957;2(9):487-492.
7. Smigel JO, Lowe KJ, Hosp PH, Gibson JH. Constipation in elderly patients; treatment with dioctyl sodium sulfosuccinate and dioctyl sodium sulfosuccinate plus peristim. Med Times. 1958;86(12):1521-1526.
8. Wilson JL, Dickinson DG. Use of dioctyl sodium sulfosuccinate (aerosol O.T.) for severe constipation. J Am Med Assoc. 1955;158(4):261-263. doi: 10.1001/jama.1955.02960040019006a.
9. Hyland CM, Foran JD. Dioctyl sodium sulphosuccinate as a laxative in the elderly. Practitioner. 1968;200(199):698-699.
10. Jin J. JAMA patient page. Over-the-counter laxatives. JAMA. 2014;312(11):1167. doi: 10.1001/jama.2014.2078.
11. 19th WHO Model List of Essential Medicines (April 2015). 2015; http://www.who.int/medicines/publications/essentialmedicines/en/.
12. Goodman J, Pang J, Bessman AN. Dioctyl sodium sulfosuccinate- an ineffective prophylactic laxative. J Chronic Dis. 1976;29(1):59-63. doi: 10.1016/0021-9681(76)90068-0.
13. Fain AM, Susat R, Herring M, Dorton K. Treatment of constipation in geriatric and chronically ill patients: a comparison. South Med J. 1978;71(6):677-680.
14. Chapman RW, Sillery J, Fontana DD, Matthys C, Saunders DR. Effect of oral dioctyl sodium sulfosuccinate on intake-output studies of human small and large intestine. Gastroenterology. 1985;89(3):489-493. doi: 10.1016/0016-5085(85)90441-X.
15. Castle SC, Cantrell M, Israel DS, Samuelson MJ. Constipation prevention: empiric use of stool softeners questioned. Geriatrics. 1991;46(11):84-86.
16. McRorie JW, Daggy BP, Morel JG, Diersing PS, Miner PB, Robinson M. Psyllium is superior to docusate sodium for treatment of chronic constipation. Aliment Pharmacol Ther. 1998;12(5):491-497. doi: 10.1046/j.1365-2036.1998.00336.x.
17. Tarumi Y, Wilson MP, Szafran O, Spooner GR. Randomized, double-blind, placebo-controlled trial of oral docusate in the management of constipation in hospice patients. J Pain Symptom Manage. 2013;45(1):2-13. doi: 10.1016/j.jpainsymman.2012.02.008.
18. Candy B, Jones L, Larkin PJ, Vickerstaff V, Tookman A, Stone P. Laxatives for the management of constipation in people receiving palliative care. Cochrane Database Syst Rev. 2015(5):CD003448.
19. Hurdon V, Viola R, Schroder C. How useful is docusate in patients at risk for constipation? A systematic review of the evidence in the chronically ill. J Pain Symptom Manage. 2000;19(2):130-136. doi: 10.1016/S0885-3924(99)00157-8.
20. Pare P, Fedorak RN. Systematic review of stimulant and nonstimulant laxatives for the treatment of functional constipation. Can J Gastroenterol Hepatol. 2014;28(10):549-557.
21. Ramkumar D, Rao SS. Efficacy and safety of traditional medical therapies for chronic constipation: systematic review. Am J Gastroenterol. 2005;100(4):936-971. doi: 10.1111/j.1572-0241.2005.40925.x
22. Health CAfDaTi. Dioctyl sulfosuccinate or docusate (calcium or sodium) for the prevention or management of constipation: a review of the clinical effectiveness. Ottawa (ON)2014.
23. McKee KY, Widera E. Habitual prescribing of laxatives-it’s time to flush outdated protocols down the drain. JAMA Intern Med. 2016;176(8):1217-1219. doi: 10.1001/jamainternmed.2016.2780.
24. Marquez L, Jones KN, Whaley EM, et al. An outbreak of burkholderia cepacia complex infections associated with contaminated liquid docusate. Infect Control Hosp Epidemiol. 2017;38(5):567-573. doi: 10.1017/ice.2017.11.
25. Ahmedzai SH, Boland J. Constipation in people prescribed opioids. BMJ Clin Evid. 2010;2010.
26. Paquette IM, Varma M, Ternent C, et al. The American society of colon and rectal surgeons’ clinical practice guideline for the evaluation and management of constipation. Dis Colon Rectum. 2016;59(6):479-492. doi: 10.1097/DCR.0000000000000599
© 2019 Society of Hospital Medicine
Top cancer advance: Treatment of rare diseases
The American Society of Clinical Oncology (ASCO) named “Progress in Treating Rare Cancers” as the Advance of the Year for 2018, citing five major studies as examples of significant breakthroughs.
In an ASCO Special Article published in the Journal of Clinical Oncology, Sumanta K. Pal, MD, of City of Hope Comprehensive Cancer Center, Duarte, Calif., and colleagues, identified five studies that notably advanced cancer research.
Each study “reflects the impressive gains we have made in understanding these so-called orphan diseases and in tailoring treatments to target their unique characteristics,” wrote ASCO president Monica M. Bertagnolli, MD, in an introduction to the report.
One of the significant advances included use of a new combination of targeted therapies for a rare thyroid cancer that elicited responses in more than two-thirds of patients. A second study showed sorafenib improving progression-free survival for patients with desmoid tumors. In addition, patients with advanced midgut neuroendocrine tumors had a 79% lower risk of disease progression or death when treated with a new therapy of targeted radiation to tumor cells, lutetium-177 (177Lu)–Dotatate, compared with standard therapy; and trastuzumab, a standard treatment for human epidermal growth factor receptor 2 (HER2)–positive breast cancer, expanded its reach and significantly slowed progression of HER2-positive uterine serous carcinoma, the authors wrote. Finally, the “first promising therapy – the colony-stimulating factor-1 (CSF-1) inhibitor pexidartinib – for a rare cancer of the joints known as tenosynovial giant cell tumor, showed an overall response rate of 39.3% in patients taking pexidartinib versus 0% in patients taking a placebo,” they said.
For the first time, the ASCO progress report included a list of priorities to guide future research efforts, stated as follows:
- Identify strategies that better predict response to immunotherapies.
- Better define the patient populations that benefit from postoperative (adjuvant) therapy.
- Translate innovations in cellular therapies to solid tumors.
- Increase precision medicine research and treatment approaches in pediatric cancers.
- Optimize care for older adults with cancer.
- Increase equitable access to cancer clinical trials.
- Reduce the long-term consequences of cancer treatment.
- Reduce obesity and its impact on cancer incidence and outcomes.
- Identify strategies to detect and treat premalignant lesions.
“These priority areas, listed in no particular order, address an unmet need or help fill a knowledge gap in areas critical to improving patient care and outcomes,” the authors wrote.
The report acknowledged the value of federally funded research and the importance of ongoing federal investment in cancer research.
Dr. Pal disclosed relationships with Pfizer, Novartis, Aveo, Myriad Pharmaceuticals, Genentech, Exelixis, Bristol-Myers Squibb, Astellas Pharma, Ipsen, Eisai, and Medivation. Coauthors disclosed relationships with these and other companies.
SOURCE: Pal SK et al. J Clin Oncol. 2019 Jan 31. doi: 10.1200/JCO.18.02037.
The American Society of Clinical Oncology (ASCO) named “Progress in Treating Rare Cancers” as the Advance of the Year for 2018, citing five major studies as examples of significant breakthroughs.
In an ASCO Special Article published in the Journal of Clinical Oncology, Sumanta K. Pal, MD, of City of Hope Comprehensive Cancer Center, Duarte, Calif., and colleagues, identified five studies that notably advanced cancer research.
Each study “reflects the impressive gains we have made in understanding these so-called orphan diseases and in tailoring treatments to target their unique characteristics,” wrote ASCO president Monica M. Bertagnolli, MD, in an introduction to the report.
One of the significant advances included use of a new combination of targeted therapies for a rare thyroid cancer that elicited responses in more than two-thirds of patients. A second study showed sorafenib improving progression-free survival for patients with desmoid tumors. In addition, patients with advanced midgut neuroendocrine tumors had a 79% lower risk of disease progression or death when treated with a new therapy of targeted radiation to tumor cells, lutetium-177 (177Lu)–Dotatate, compared with standard therapy; and trastuzumab, a standard treatment for human epidermal growth factor receptor 2 (HER2)–positive breast cancer, expanded its reach and significantly slowed progression of HER2-positive uterine serous carcinoma, the authors wrote. Finally, the “first promising therapy – the colony-stimulating factor-1 (CSF-1) inhibitor pexidartinib – for a rare cancer of the joints known as tenosynovial giant cell tumor, showed an overall response rate of 39.3% in patients taking pexidartinib versus 0% in patients taking a placebo,” they said.
For the first time, the ASCO progress report included a list of priorities to guide future research efforts, stated as follows:
- Identify strategies that better predict response to immunotherapies.
- Better define the patient populations that benefit from postoperative (adjuvant) therapy.
- Translate innovations in cellular therapies to solid tumors.
- Increase precision medicine research and treatment approaches in pediatric cancers.
- Optimize care for older adults with cancer.
- Increase equitable access to cancer clinical trials.
- Reduce the long-term consequences of cancer treatment.
- Reduce obesity and its impact on cancer incidence and outcomes.
- Identify strategies to detect and treat premalignant lesions.
“These priority areas, listed in no particular order, address an unmet need or help fill a knowledge gap in areas critical to improving patient care and outcomes,” the authors wrote.
The report acknowledged the value of federally funded research and the importance of ongoing federal investment in cancer research.
Dr. Pal disclosed relationships with Pfizer, Novartis, Aveo, Myriad Pharmaceuticals, Genentech, Exelixis, Bristol-Myers Squibb, Astellas Pharma, Ipsen, Eisai, and Medivation. Coauthors disclosed relationships with these and other companies.
SOURCE: Pal SK et al. J Clin Oncol. 2019 Jan 31. doi: 10.1200/JCO.18.02037.
The American Society of Clinical Oncology (ASCO) named “Progress in Treating Rare Cancers” as the Advance of the Year for 2018, citing five major studies as examples of significant breakthroughs.
In an ASCO Special Article published in the Journal of Clinical Oncology, Sumanta K. Pal, MD, of City of Hope Comprehensive Cancer Center, Duarte, Calif., and colleagues, identified five studies that notably advanced cancer research.
Each study “reflects the impressive gains we have made in understanding these so-called orphan diseases and in tailoring treatments to target their unique characteristics,” wrote ASCO president Monica M. Bertagnolli, MD, in an introduction to the report.
One of the significant advances included use of a new combination of targeted therapies for a rare thyroid cancer that elicited responses in more than two-thirds of patients. A second study showed sorafenib improving progression-free survival for patients with desmoid tumors. In addition, patients with advanced midgut neuroendocrine tumors had a 79% lower risk of disease progression or death when treated with a new therapy of targeted radiation to tumor cells, lutetium-177 (177Lu)–Dotatate, compared with standard therapy; and trastuzumab, a standard treatment for human epidermal growth factor receptor 2 (HER2)–positive breast cancer, expanded its reach and significantly slowed progression of HER2-positive uterine serous carcinoma, the authors wrote. Finally, the “first promising therapy – the colony-stimulating factor-1 (CSF-1) inhibitor pexidartinib – for a rare cancer of the joints known as tenosynovial giant cell tumor, showed an overall response rate of 39.3% in patients taking pexidartinib versus 0% in patients taking a placebo,” they said.
For the first time, the ASCO progress report included a list of priorities to guide future research efforts, stated as follows:
- Identify strategies that better predict response to immunotherapies.
- Better define the patient populations that benefit from postoperative (adjuvant) therapy.
- Translate innovations in cellular therapies to solid tumors.
- Increase precision medicine research and treatment approaches in pediatric cancers.
- Optimize care for older adults with cancer.
- Increase equitable access to cancer clinical trials.
- Reduce the long-term consequences of cancer treatment.
- Reduce obesity and its impact on cancer incidence and outcomes.
- Identify strategies to detect and treat premalignant lesions.
“These priority areas, listed in no particular order, address an unmet need or help fill a knowledge gap in areas critical to improving patient care and outcomes,” the authors wrote.
The report acknowledged the value of federally funded research and the importance of ongoing federal investment in cancer research.
Dr. Pal disclosed relationships with Pfizer, Novartis, Aveo, Myriad Pharmaceuticals, Genentech, Exelixis, Bristol-Myers Squibb, Astellas Pharma, Ipsen, Eisai, and Medivation. Coauthors disclosed relationships with these and other companies.
SOURCE: Pal SK et al. J Clin Oncol. 2019 Jan 31. doi: 10.1200/JCO.18.02037.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Single-cell genomics drive progress toward human breast cell atlas development
SAN ANTONIO – Researchers at MD Anderson Cancer Center in Houston and the University of New South Wales (UNSW) in Sydney are among teams from around the world working toward human breast cell atlas development using single-cell genomics, and their efforts to date have yielded new understanding of both the normal breast cell ecosystem and the breast cancer tumor microenvironment.
The work at MD Anderson, for example, has led to the identification of a number of new gene markers and multiple cell states within breast cell types, according to Tapsi Kumar Seth, who reported early findings from an analysis of more than 32,000 cells from normal breast tissue during a presentation at the San Antonio Breast Cancer Symposium.
At the UNSW’s Garvan Institute of Medical Research, Alexander Swarbrick, PhD, and his colleagues are working to better define the tumor microenvironment at the single-cell level. At the symposium, Dr. Swarbrick presented interim findings from cellular analyses in the first 23 breast cancer cases of about 200 that will be studied in the course of the project.
Improved understanding of the cellular landscape of both normal breast tissue and breast cancer tissue should lead to new stromal- and immune-based therapies for the treatment of breast cancer, the investigators said.
The normal breast cell ecosystem
The MD Anderson researchers studied 32,148 stromal cells from pathologically normal breast tissues collected from 11 women who underwent mastectomy at the center.
Unbiased expression analysis identified three major cell types, including epithelial cells, fibroblasts, and endothelial cells, as well as several minor cell types such as macrophages, T-cells, apocrine cells, pericytes, and others, said Ms. Seth, a graduate student in the department of genetics at the center and a member of the Navin Laboratory there.
The work is designed to help identify the presence and function of cells and explain how they behave in a normal breast ecosystem, she said.
“We know that a female breast undergoes a lot of changes due to age, pregnancy, or when there is a disease such as cancer, so it’s essential to chart out what a normal cell reference would look like,” she said.
Toward that goal, a protocol was developed to dissociate the tissue samples within 2 hours due to the decline in viability seen in cells and RNA over time. Analysis of the cell states revealed different transcriptional programs in luminal epithelial cells (hormone receptor positive and secretory), basal epithelial cells (myoepithelial or basement-like), endothelial cells (lymphatic or vascular), macrophages (M1 or M2) and fibroblasts (three subgroups) and provided insight into progenitors of each cell types, she said.
A map was created to show gene expression and to identify transcriptomally similar cells.
“We were able to identify most of the major cell types that are present in human breasts,” she said. “What was interesting was that the composition of these cells also varied across women.”
For example, the proportion of fibroblasts was lower in 3 of the 11 patients, and even though the cells were pathologically normal, immune cell populations, including T-cells and macrophages, were also seen.
Adipocytes cannot be evaluated using this technology because they are large and the layer of fat cells must be removed during dissociation to prevent clogging of the machines, she noted, adding that “this is really a limitation of our technology.”
A closer look was taken at each of the major cell types identified.
Epithelial cells
Both canonical and new gene markers were used to identify luminal and basal epithelial cells, Ms. Seth noted.
Among the known markers were KRT18 for luminal epithelial cells and KRT5, KRT6B, KRT14, and KRT15 for basal epithelial cells. Among the new markers were SLC39A6, EFHD1 and HES1 for the luminal epithelial cells, and CITED4, CCK28, MMP7, and MDRG2 for the basal epithelial cells.
“We went on and validated these markers on the tissue section using methods like spatial transcriptomics,” she said, explaining that this “really helps capture the RNA expression spatially,” and can resolve the localization of cell types markers in anatomical structures.
For these cells, the expression of the newly identified gene markers was mostly confined to ducts and lobules.
In addition, an analysis of cell states within the luminal epithelial cells showed four different cell states, each of which have “different kinds of genes that they express, and also different pathways that they express, suggesting that these might be transcriptomally different,” Ms. Seth said.
Of note, these cells and cells states are not biased to a specific condition or patient, suggesting that they are coming from all of the patients, she added.
Two of the four cell states – the secretory and hormone responsive states – have previously been reported, but Ms. Seth and her colleagues identified two additional cell states that may have different biological functions and are present in the different anatomical regions of the breast.
Fibroblasts
Fibroblasts, the cells of the connective tissue, were the most abundant cell type. Like the epithelial cells, both canonical collagen markers (COL6A3, MMP2, FBN1, FBLN2, FBN, and COL1A1) and newly identified gene markers (TNXB, AEBP1, CFH, CTSK, TPPP3, MEG3, HTRA1, LHFP, and OGN) were used to identify them.
Endothelial cells
Breast tissue is highly vascular, so endothelial cells, which line the walls of veins, arteries, and lymphatic vessels, are plentiful.
“Again, for both these cell types, we identified them using the canonical marker CD31, and we identified some new gene markers,” she said, noting that the new markers include CCL21, CLDN5, MMRN1, LYVE1, and PROX1 for lymphatic endothelial cells, and RNASE1 and IFI27 for vascular endothelial cells.
Two different groups – or states – of vascular endothelial cells were identified, with each expressing “very different genes as well as very different pathways, again suggesting that they might have different biological functions, which we are still investigating,” she said.
Additional findings and future directions
In addition to stromal cells, some immune cells were also seen. These included T cells that came mostly from two patients, as well as macrophages and monocytes, which comprised the most abundant immune cell population.
Of note, all of these cells are also found in the tumor microenvironment, but they are in a transformed state. For example cancer-associated fibroblasts, tumor endothelial cells, tumor-associated macrophages, and tumor-associated adipocytes have been seen in that environment, she said.
“So what we are trying to do with this project is ... learn how these cells are, and how these cells behave in the normal ecosystem,” she explained, noting that the hope is to provide a valuable reference for the research community with new insights about how normal cell types are transformed in the tumor microenvironment.
In an effort to overcome the adipocyte-associated limitation of the technology, adipocytes are “now being isolated by single nucleus RNA sequencing.”
“This [sequencing] technology has helped us identify multiple cell states within a cell type; and most of these cell states may have different biological functions, which probably can be investigated by spatial transcriptomic methods,” she said.
Spatial transcriptomics also continue to be used for validation of the new gene markers identified in the course of this research, she noted.
The breast tumor microenvironment
At the Garvan Institute, current work is focusing more on defining the landscape of the breast tumor microenvironment at single-cell resolution, according to Dr. Swarbrick, a senior research fellow and head of the Tumour Progression Laboratory there.
“Breast cancers ... are complex cellular ecosystems, and it’s really the sum of the interactions between the cell types that play major roles in determining the etiology of disease and its response to therapy,” he said. “So I think that going forward toward a new age of diagnostics and therapeutics, there’s wonderful potential in capitalizing on the tumor microenvironment for new developments, but this has to be built on a really deep understanding of the tumor microenvironment, and – I might say – a new taxonomy of the breast cellular environment.”
Therefore, in an effort to address “this limitation in our knowledge base,” his lab is also working toward development of a breast cell atlas.
A fresh tissue collection program was established to collect early breast cancer tissues at the time of surgery, metastatic biopsies, and metastatic lesions from autopsies. The tissues are quickly dissociated into their cellular components and they undergo massively parallel capture and sequencing using the 10x genomics platform, he said.
Thousands of cells per case are analyzed using single-cell RNA sequencing (RNA-seq), as well as “RAGE-seq” and “CITE-seq,” which are performed in parallel to the RNA sequencing to address some of the limitations of the RNA sequencing alone and to “try to gain a multi-omic insight into the cell biology,” he explained.
RAGE-seq, which Dr. Swarbrick and his team developed, “is essentially a method to do targeted long-read sequencing in parallel to the short-read sequencing that we use for RNA-seq,” and CITE-seq is “a really fantastic method developed at the New York Genome Center that essentially allows us to gather proteomic data in parallel to the RNA data,” he said.
Based on findings from the analyses of about 125,000 cells from 25 patients, a map was created that showed the cell clusters identified by both canonical markers and gene expression signatures.
“We find the cell types we would expect to be present in a breast cancer,” he said.
The map shows clusters of myeloid, epithelial-1 and -2, cancer-associated fibroblast (CAF)-1 and -2, endothelial, T Reg, B, and CD8 and CD4 T cells.
Next, each cell type is quantified in each patient, and a graphic representation of the findings shows large variability in the proportions of each cell type in each patient.
“Ultimately, our goal is to be able to relate the frequencies of cell types and molecular features to each other, but also to clinical-pathological features from these patients,” he noted.
A closer look at the findings on an individual case level demonstrates the potential for development of better therapies.
For example, a case involving a high-grade triple-negative invasive ductal carcinoma exhibited each of the cell types found overall.
“One of the things that strikes us early on is we see a number of malignant epithelial populations,” he said, noting that proliferation is one of the drivers of the heterogeneity, but that heterogeneity was also seen for “other clinically relevant features such as basal cytokeratins,” which were heterogeneously expressed in different cell-type clusters.
“This was kind of paralleled in the immunohistochemistry results that we obtained from this patient,” he said. “We could also apply other clinically used tests that we’ve developed on bulk (such as PAM50 intrinsic subtyping) and ask whether they can be applied at the single-cell resolution.
“We think that these are going to be great tools to try to now get in and understand the significance of this heterogeneity and try to identify the lethal cells within this patient, and potentially therapeutic strategies to eradicate those cells,” he added.
Fibroblasts
A notable finding of this project was the presence of “not one, but two populations of fibroblasts,” Dr. Swarbrick said, noting that fibroblasts are typically discussed as a single entity.
“This is arguing that there are at least two major types present within the breast, and almost every case has these populations present at roughly equal amounts,” he said.
This is of particular interest, because it has been shown in prior studies that targeting fibroblasts can have therapeutic outcomes.
“So we think this is a very important population within the tumor microenvironment,” he added.
With respect to gene expression features, CAF-1 is dominated by signatures of extracellular matrix deposition and remodeling, which “look like the classic myofibroblasts that we typically think of when we study cancer-associated fibroblasts.”
“In contrast, the CAF-2 population ... have what appears to be quite a predominant secretory function, so we see a lot of cytokines being produced by these cells, but we also see a very high level of expression of a number immune checkpoint ligands,” he said, adding that his team is actively pursuing whether these cells may be undergoing signaling events with infiltrating lymphocytes in the tumor microenvironment.
The signatures for both CAF types are prognostic within large breast cancer data sets, suggesting that they do actually have an important role in disease, he noted.
Markers for these cells include ACTA2, which was previously known to be a marker, and which is almost exclusively restricted to CAF-1, and the cell surface protein CD34 – a progenitor marker in many different cellular systems, “which is actually beautifully expressed on the CAF-2 population” as demonstrated using CITE-seq.
“So we’re now using this as a way to prospectively identify these cells, pull them out of tumors, and conduct biologic assays to learn more about them,” he said.
The immune milieu
“We’re in the age of immunotherapy, and this is an area of huge interest, but we have a long way to go in making it as effective as possible for breast cancer patients,” Dr. Swarbrick said. “I believe part of that is through a very deep understanding of the taxonomy.”
RNA data alone are useful but insufficient to fully identify subsets of immune cells due to a “relatively low-resolution ability to resolve T cells.”
“But because we’re now using the panel of 125 antibodies in parallel, we can now start to use protein levels to split up these populations and we can start to now identify, with higher resolution, more unique populations within the environment,” he said, noting that the availability of protein data not only helps identify subtypes, but is also therapeutically important as it allows for certainty regarding whether the protein target of therapeutic antibodies is expressed on the surface of cells.
Ultimately the hope is that this effort to build a multi-omic breast cancer atlas will continue to drive new discoveries in personalized medicine for breast cancer, Dr. Swarbrick concluded, adding that the field is moving fast, and it will be very important for labs like his and the Navin Lab to communicate to avoid needlessly duplicating efforts.
“I think it’s going to be really exciting to start to put some of these [findings] together,” he said.
The MD Anderson project is funded by the Chan Zuckerberg Initiative as part of its work in supporting the Human Cell Atlas project. Ms. Seth reported having no disclosures. Dr. Swarbrick’s research is funded by the Australian Government/National Health and Medical Research Council and the National Breast Cancer Foundation. He reported having no relevant disclosures.
SOURCE: Seth T et al. SABCS 2018, Abstract GS1-02; Swarbrick A et al. SABCS 2018, Abstract GS1-01
SAN ANTONIO – Researchers at MD Anderson Cancer Center in Houston and the University of New South Wales (UNSW) in Sydney are among teams from around the world working toward human breast cell atlas development using single-cell genomics, and their efforts to date have yielded new understanding of both the normal breast cell ecosystem and the breast cancer tumor microenvironment.
The work at MD Anderson, for example, has led to the identification of a number of new gene markers and multiple cell states within breast cell types, according to Tapsi Kumar Seth, who reported early findings from an analysis of more than 32,000 cells from normal breast tissue during a presentation at the San Antonio Breast Cancer Symposium.
At the UNSW’s Garvan Institute of Medical Research, Alexander Swarbrick, PhD, and his colleagues are working to better define the tumor microenvironment at the single-cell level. At the symposium, Dr. Swarbrick presented interim findings from cellular analyses in the first 23 breast cancer cases of about 200 that will be studied in the course of the project.
Improved understanding of the cellular landscape of both normal breast tissue and breast cancer tissue should lead to new stromal- and immune-based therapies for the treatment of breast cancer, the investigators said.
The normal breast cell ecosystem
The MD Anderson researchers studied 32,148 stromal cells from pathologically normal breast tissues collected from 11 women who underwent mastectomy at the center.
Unbiased expression analysis identified three major cell types, including epithelial cells, fibroblasts, and endothelial cells, as well as several minor cell types such as macrophages, T-cells, apocrine cells, pericytes, and others, said Ms. Seth, a graduate student in the department of genetics at the center and a member of the Navin Laboratory there.
The work is designed to help identify the presence and function of cells and explain how they behave in a normal breast ecosystem, she said.
“We know that a female breast undergoes a lot of changes due to age, pregnancy, or when there is a disease such as cancer, so it’s essential to chart out what a normal cell reference would look like,” she said.
Toward that goal, a protocol was developed to dissociate the tissue samples within 2 hours due to the decline in viability seen in cells and RNA over time. Analysis of the cell states revealed different transcriptional programs in luminal epithelial cells (hormone receptor positive and secretory), basal epithelial cells (myoepithelial or basement-like), endothelial cells (lymphatic or vascular), macrophages (M1 or M2) and fibroblasts (three subgroups) and provided insight into progenitors of each cell types, she said.
A map was created to show gene expression and to identify transcriptomally similar cells.
“We were able to identify most of the major cell types that are present in human breasts,” she said. “What was interesting was that the composition of these cells also varied across women.”
For example, the proportion of fibroblasts was lower in 3 of the 11 patients, and even though the cells were pathologically normal, immune cell populations, including T-cells and macrophages, were also seen.
Adipocytes cannot be evaluated using this technology because they are large and the layer of fat cells must be removed during dissociation to prevent clogging of the machines, she noted, adding that “this is really a limitation of our technology.”
A closer look was taken at each of the major cell types identified.
Epithelial cells
Both canonical and new gene markers were used to identify luminal and basal epithelial cells, Ms. Seth noted.
Among the known markers were KRT18 for luminal epithelial cells and KRT5, KRT6B, KRT14, and KRT15 for basal epithelial cells. Among the new markers were SLC39A6, EFHD1 and HES1 for the luminal epithelial cells, and CITED4, CCK28, MMP7, and MDRG2 for the basal epithelial cells.
“We went on and validated these markers on the tissue section using methods like spatial transcriptomics,” she said, explaining that this “really helps capture the RNA expression spatially,” and can resolve the localization of cell types markers in anatomical structures.
For these cells, the expression of the newly identified gene markers was mostly confined to ducts and lobules.
In addition, an analysis of cell states within the luminal epithelial cells showed four different cell states, each of which have “different kinds of genes that they express, and also different pathways that they express, suggesting that these might be transcriptomally different,” Ms. Seth said.
Of note, these cells and cells states are not biased to a specific condition or patient, suggesting that they are coming from all of the patients, she added.
Two of the four cell states – the secretory and hormone responsive states – have previously been reported, but Ms. Seth and her colleagues identified two additional cell states that may have different biological functions and are present in the different anatomical regions of the breast.
Fibroblasts
Fibroblasts, the cells of the connective tissue, were the most abundant cell type. Like the epithelial cells, both canonical collagen markers (COL6A3, MMP2, FBN1, FBLN2, FBN, and COL1A1) and newly identified gene markers (TNXB, AEBP1, CFH, CTSK, TPPP3, MEG3, HTRA1, LHFP, and OGN) were used to identify them.
Endothelial cells
Breast tissue is highly vascular, so endothelial cells, which line the walls of veins, arteries, and lymphatic vessels, are plentiful.
“Again, for both these cell types, we identified them using the canonical marker CD31, and we identified some new gene markers,” she said, noting that the new markers include CCL21, CLDN5, MMRN1, LYVE1, and PROX1 for lymphatic endothelial cells, and RNASE1 and IFI27 for vascular endothelial cells.
Two different groups – or states – of vascular endothelial cells were identified, with each expressing “very different genes as well as very different pathways, again suggesting that they might have different biological functions, which we are still investigating,” she said.
Additional findings and future directions
In addition to stromal cells, some immune cells were also seen. These included T cells that came mostly from two patients, as well as macrophages and monocytes, which comprised the most abundant immune cell population.
Of note, all of these cells are also found in the tumor microenvironment, but they are in a transformed state. For example cancer-associated fibroblasts, tumor endothelial cells, tumor-associated macrophages, and tumor-associated adipocytes have been seen in that environment, she said.
“So what we are trying to do with this project is ... learn how these cells are, and how these cells behave in the normal ecosystem,” she explained, noting that the hope is to provide a valuable reference for the research community with new insights about how normal cell types are transformed in the tumor microenvironment.
In an effort to overcome the adipocyte-associated limitation of the technology, adipocytes are “now being isolated by single nucleus RNA sequencing.”
“This [sequencing] technology has helped us identify multiple cell states within a cell type; and most of these cell states may have different biological functions, which probably can be investigated by spatial transcriptomic methods,” she said.
Spatial transcriptomics also continue to be used for validation of the new gene markers identified in the course of this research, she noted.
The breast tumor microenvironment
At the Garvan Institute, current work is focusing more on defining the landscape of the breast tumor microenvironment at single-cell resolution, according to Dr. Swarbrick, a senior research fellow and head of the Tumour Progression Laboratory there.
“Breast cancers ... are complex cellular ecosystems, and it’s really the sum of the interactions between the cell types that play major roles in determining the etiology of disease and its response to therapy,” he said. “So I think that going forward toward a new age of diagnostics and therapeutics, there’s wonderful potential in capitalizing on the tumor microenvironment for new developments, but this has to be built on a really deep understanding of the tumor microenvironment, and – I might say – a new taxonomy of the breast cellular environment.”
Therefore, in an effort to address “this limitation in our knowledge base,” his lab is also working toward development of a breast cell atlas.
A fresh tissue collection program was established to collect early breast cancer tissues at the time of surgery, metastatic biopsies, and metastatic lesions from autopsies. The tissues are quickly dissociated into their cellular components and they undergo massively parallel capture and sequencing using the 10x genomics platform, he said.
Thousands of cells per case are analyzed using single-cell RNA sequencing (RNA-seq), as well as “RAGE-seq” and “CITE-seq,” which are performed in parallel to the RNA sequencing to address some of the limitations of the RNA sequencing alone and to “try to gain a multi-omic insight into the cell biology,” he explained.
RAGE-seq, which Dr. Swarbrick and his team developed, “is essentially a method to do targeted long-read sequencing in parallel to the short-read sequencing that we use for RNA-seq,” and CITE-seq is “a really fantastic method developed at the New York Genome Center that essentially allows us to gather proteomic data in parallel to the RNA data,” he said.
Based on findings from the analyses of about 125,000 cells from 25 patients, a map was created that showed the cell clusters identified by both canonical markers and gene expression signatures.
“We find the cell types we would expect to be present in a breast cancer,” he said.
The map shows clusters of myeloid, epithelial-1 and -2, cancer-associated fibroblast (CAF)-1 and -2, endothelial, T Reg, B, and CD8 and CD4 T cells.
Next, each cell type is quantified in each patient, and a graphic representation of the findings shows large variability in the proportions of each cell type in each patient.
“Ultimately, our goal is to be able to relate the frequencies of cell types and molecular features to each other, but also to clinical-pathological features from these patients,” he noted.
A closer look at the findings on an individual case level demonstrates the potential for development of better therapies.
For example, a case involving a high-grade triple-negative invasive ductal carcinoma exhibited each of the cell types found overall.
“One of the things that strikes us early on is we see a number of malignant epithelial populations,” he said, noting that proliferation is one of the drivers of the heterogeneity, but that heterogeneity was also seen for “other clinically relevant features such as basal cytokeratins,” which were heterogeneously expressed in different cell-type clusters.
“This was kind of paralleled in the immunohistochemistry results that we obtained from this patient,” he said. “We could also apply other clinically used tests that we’ve developed on bulk (such as PAM50 intrinsic subtyping) and ask whether they can be applied at the single-cell resolution.
“We think that these are going to be great tools to try to now get in and understand the significance of this heterogeneity and try to identify the lethal cells within this patient, and potentially therapeutic strategies to eradicate those cells,” he added.
Fibroblasts
A notable finding of this project was the presence of “not one, but two populations of fibroblasts,” Dr. Swarbrick said, noting that fibroblasts are typically discussed as a single entity.
“This is arguing that there are at least two major types present within the breast, and almost every case has these populations present at roughly equal amounts,” he said.
This is of particular interest, because it has been shown in prior studies that targeting fibroblasts can have therapeutic outcomes.
“So we think this is a very important population within the tumor microenvironment,” he added.
With respect to gene expression features, CAF-1 is dominated by signatures of extracellular matrix deposition and remodeling, which “look like the classic myofibroblasts that we typically think of when we study cancer-associated fibroblasts.”
“In contrast, the CAF-2 population ... have what appears to be quite a predominant secretory function, so we see a lot of cytokines being produced by these cells, but we also see a very high level of expression of a number immune checkpoint ligands,” he said, adding that his team is actively pursuing whether these cells may be undergoing signaling events with infiltrating lymphocytes in the tumor microenvironment.
The signatures for both CAF types are prognostic within large breast cancer data sets, suggesting that they do actually have an important role in disease, he noted.
Markers for these cells include ACTA2, which was previously known to be a marker, and which is almost exclusively restricted to CAF-1, and the cell surface protein CD34 – a progenitor marker in many different cellular systems, “which is actually beautifully expressed on the CAF-2 population” as demonstrated using CITE-seq.
“So we’re now using this as a way to prospectively identify these cells, pull them out of tumors, and conduct biologic assays to learn more about them,” he said.
The immune milieu
“We’re in the age of immunotherapy, and this is an area of huge interest, but we have a long way to go in making it as effective as possible for breast cancer patients,” Dr. Swarbrick said. “I believe part of that is through a very deep understanding of the taxonomy.”
RNA data alone are useful but insufficient to fully identify subsets of immune cells due to a “relatively low-resolution ability to resolve T cells.”
“But because we’re now using the panel of 125 antibodies in parallel, we can now start to use protein levels to split up these populations and we can start to now identify, with higher resolution, more unique populations within the environment,” he said, noting that the availability of protein data not only helps identify subtypes, but is also therapeutically important as it allows for certainty regarding whether the protein target of therapeutic antibodies is expressed on the surface of cells.
Ultimately the hope is that this effort to build a multi-omic breast cancer atlas will continue to drive new discoveries in personalized medicine for breast cancer, Dr. Swarbrick concluded, adding that the field is moving fast, and it will be very important for labs like his and the Navin Lab to communicate to avoid needlessly duplicating efforts.
“I think it’s going to be really exciting to start to put some of these [findings] together,” he said.
The MD Anderson project is funded by the Chan Zuckerberg Initiative as part of its work in supporting the Human Cell Atlas project. Ms. Seth reported having no disclosures. Dr. Swarbrick’s research is funded by the Australian Government/National Health and Medical Research Council and the National Breast Cancer Foundation. He reported having no relevant disclosures.
SOURCE: Seth T et al. SABCS 2018, Abstract GS1-02; Swarbrick A et al. SABCS 2018, Abstract GS1-01
SAN ANTONIO – Researchers at MD Anderson Cancer Center in Houston and the University of New South Wales (UNSW) in Sydney are among teams from around the world working toward human breast cell atlas development using single-cell genomics, and their efforts to date have yielded new understanding of both the normal breast cell ecosystem and the breast cancer tumor microenvironment.
The work at MD Anderson, for example, has led to the identification of a number of new gene markers and multiple cell states within breast cell types, according to Tapsi Kumar Seth, who reported early findings from an analysis of more than 32,000 cells from normal breast tissue during a presentation at the San Antonio Breast Cancer Symposium.
At the UNSW’s Garvan Institute of Medical Research, Alexander Swarbrick, PhD, and his colleagues are working to better define the tumor microenvironment at the single-cell level. At the symposium, Dr. Swarbrick presented interim findings from cellular analyses in the first 23 breast cancer cases of about 200 that will be studied in the course of the project.
Improved understanding of the cellular landscape of both normal breast tissue and breast cancer tissue should lead to new stromal- and immune-based therapies for the treatment of breast cancer, the investigators said.
The normal breast cell ecosystem
The MD Anderson researchers studied 32,148 stromal cells from pathologically normal breast tissues collected from 11 women who underwent mastectomy at the center.
Unbiased expression analysis identified three major cell types, including epithelial cells, fibroblasts, and endothelial cells, as well as several minor cell types such as macrophages, T-cells, apocrine cells, pericytes, and others, said Ms. Seth, a graduate student in the department of genetics at the center and a member of the Navin Laboratory there.
The work is designed to help identify the presence and function of cells and explain how they behave in a normal breast ecosystem, she said.
“We know that a female breast undergoes a lot of changes due to age, pregnancy, or when there is a disease such as cancer, so it’s essential to chart out what a normal cell reference would look like,” she said.
Toward that goal, a protocol was developed to dissociate the tissue samples within 2 hours due to the decline in viability seen in cells and RNA over time. Analysis of the cell states revealed different transcriptional programs in luminal epithelial cells (hormone receptor positive and secretory), basal epithelial cells (myoepithelial or basement-like), endothelial cells (lymphatic or vascular), macrophages (M1 or M2) and fibroblasts (three subgroups) and provided insight into progenitors of each cell types, she said.
A map was created to show gene expression and to identify transcriptomally similar cells.
“We were able to identify most of the major cell types that are present in human breasts,” she said. “What was interesting was that the composition of these cells also varied across women.”
For example, the proportion of fibroblasts was lower in 3 of the 11 patients, and even though the cells were pathologically normal, immune cell populations, including T-cells and macrophages, were also seen.
Adipocytes cannot be evaluated using this technology because they are large and the layer of fat cells must be removed during dissociation to prevent clogging of the machines, she noted, adding that “this is really a limitation of our technology.”
A closer look was taken at each of the major cell types identified.
Epithelial cells
Both canonical and new gene markers were used to identify luminal and basal epithelial cells, Ms. Seth noted.
Among the known markers were KRT18 for luminal epithelial cells and KRT5, KRT6B, KRT14, and KRT15 for basal epithelial cells. Among the new markers were SLC39A6, EFHD1 and HES1 for the luminal epithelial cells, and CITED4, CCK28, MMP7, and MDRG2 for the basal epithelial cells.
“We went on and validated these markers on the tissue section using methods like spatial transcriptomics,” she said, explaining that this “really helps capture the RNA expression spatially,” and can resolve the localization of cell types markers in anatomical structures.
For these cells, the expression of the newly identified gene markers was mostly confined to ducts and lobules.
In addition, an analysis of cell states within the luminal epithelial cells showed four different cell states, each of which have “different kinds of genes that they express, and also different pathways that they express, suggesting that these might be transcriptomally different,” Ms. Seth said.
Of note, these cells and cells states are not biased to a specific condition or patient, suggesting that they are coming from all of the patients, she added.
Two of the four cell states – the secretory and hormone responsive states – have previously been reported, but Ms. Seth and her colleagues identified two additional cell states that may have different biological functions and are present in the different anatomical regions of the breast.
Fibroblasts
Fibroblasts, the cells of the connective tissue, were the most abundant cell type. Like the epithelial cells, both canonical collagen markers (COL6A3, MMP2, FBN1, FBLN2, FBN, and COL1A1) and newly identified gene markers (TNXB, AEBP1, CFH, CTSK, TPPP3, MEG3, HTRA1, LHFP, and OGN) were used to identify them.
Endothelial cells
Breast tissue is highly vascular, so endothelial cells, which line the walls of veins, arteries, and lymphatic vessels, are plentiful.
“Again, for both these cell types, we identified them using the canonical marker CD31, and we identified some new gene markers,” she said, noting that the new markers include CCL21, CLDN5, MMRN1, LYVE1, and PROX1 for lymphatic endothelial cells, and RNASE1 and IFI27 for vascular endothelial cells.
Two different groups – or states – of vascular endothelial cells were identified, with each expressing “very different genes as well as very different pathways, again suggesting that they might have different biological functions, which we are still investigating,” she said.
Additional findings and future directions
In addition to stromal cells, some immune cells were also seen. These included T cells that came mostly from two patients, as well as macrophages and monocytes, which comprised the most abundant immune cell population.
Of note, all of these cells are also found in the tumor microenvironment, but they are in a transformed state. For example cancer-associated fibroblasts, tumor endothelial cells, tumor-associated macrophages, and tumor-associated adipocytes have been seen in that environment, she said.
“So what we are trying to do with this project is ... learn how these cells are, and how these cells behave in the normal ecosystem,” she explained, noting that the hope is to provide a valuable reference for the research community with new insights about how normal cell types are transformed in the tumor microenvironment.
In an effort to overcome the adipocyte-associated limitation of the technology, adipocytes are “now being isolated by single nucleus RNA sequencing.”
“This [sequencing] technology has helped us identify multiple cell states within a cell type; and most of these cell states may have different biological functions, which probably can be investigated by spatial transcriptomic methods,” she said.
Spatial transcriptomics also continue to be used for validation of the new gene markers identified in the course of this research, she noted.
The breast tumor microenvironment
At the Garvan Institute, current work is focusing more on defining the landscape of the breast tumor microenvironment at single-cell resolution, according to Dr. Swarbrick, a senior research fellow and head of the Tumour Progression Laboratory there.
“Breast cancers ... are complex cellular ecosystems, and it’s really the sum of the interactions between the cell types that play major roles in determining the etiology of disease and its response to therapy,” he said. “So I think that going forward toward a new age of diagnostics and therapeutics, there’s wonderful potential in capitalizing on the tumor microenvironment for new developments, but this has to be built on a really deep understanding of the tumor microenvironment, and – I might say – a new taxonomy of the breast cellular environment.”
Therefore, in an effort to address “this limitation in our knowledge base,” his lab is also working toward development of a breast cell atlas.
A fresh tissue collection program was established to collect early breast cancer tissues at the time of surgery, metastatic biopsies, and metastatic lesions from autopsies. The tissues are quickly dissociated into their cellular components and they undergo massively parallel capture and sequencing using the 10x genomics platform, he said.
Thousands of cells per case are analyzed using single-cell RNA sequencing (RNA-seq), as well as “RAGE-seq” and “CITE-seq,” which are performed in parallel to the RNA sequencing to address some of the limitations of the RNA sequencing alone and to “try to gain a multi-omic insight into the cell biology,” he explained.
RAGE-seq, which Dr. Swarbrick and his team developed, “is essentially a method to do targeted long-read sequencing in parallel to the short-read sequencing that we use for RNA-seq,” and CITE-seq is “a really fantastic method developed at the New York Genome Center that essentially allows us to gather proteomic data in parallel to the RNA data,” he said.
Based on findings from the analyses of about 125,000 cells from 25 patients, a map was created that showed the cell clusters identified by both canonical markers and gene expression signatures.
“We find the cell types we would expect to be present in a breast cancer,” he said.
The map shows clusters of myeloid, epithelial-1 and -2, cancer-associated fibroblast (CAF)-1 and -2, endothelial, T Reg, B, and CD8 and CD4 T cells.
Next, each cell type is quantified in each patient, and a graphic representation of the findings shows large variability in the proportions of each cell type in each patient.
“Ultimately, our goal is to be able to relate the frequencies of cell types and molecular features to each other, but also to clinical-pathological features from these patients,” he noted.
A closer look at the findings on an individual case level demonstrates the potential for development of better therapies.
For example, a case involving a high-grade triple-negative invasive ductal carcinoma exhibited each of the cell types found overall.
“One of the things that strikes us early on is we see a number of malignant epithelial populations,” he said, noting that proliferation is one of the drivers of the heterogeneity, but that heterogeneity was also seen for “other clinically relevant features such as basal cytokeratins,” which were heterogeneously expressed in different cell-type clusters.
“This was kind of paralleled in the immunohistochemistry results that we obtained from this patient,” he said. “We could also apply other clinically used tests that we’ve developed on bulk (such as PAM50 intrinsic subtyping) and ask whether they can be applied at the single-cell resolution.
“We think that these are going to be great tools to try to now get in and understand the significance of this heterogeneity and try to identify the lethal cells within this patient, and potentially therapeutic strategies to eradicate those cells,” he added.
Fibroblasts
A notable finding of this project was the presence of “not one, but two populations of fibroblasts,” Dr. Swarbrick said, noting that fibroblasts are typically discussed as a single entity.
“This is arguing that there are at least two major types present within the breast, and almost every case has these populations present at roughly equal amounts,” he said.
This is of particular interest, because it has been shown in prior studies that targeting fibroblasts can have therapeutic outcomes.
“So we think this is a very important population within the tumor microenvironment,” he added.
With respect to gene expression features, CAF-1 is dominated by signatures of extracellular matrix deposition and remodeling, which “look like the classic myofibroblasts that we typically think of when we study cancer-associated fibroblasts.”
“In contrast, the CAF-2 population ... have what appears to be quite a predominant secretory function, so we see a lot of cytokines being produced by these cells, but we also see a very high level of expression of a number immune checkpoint ligands,” he said, adding that his team is actively pursuing whether these cells may be undergoing signaling events with infiltrating lymphocytes in the tumor microenvironment.
The signatures for both CAF types are prognostic within large breast cancer data sets, suggesting that they do actually have an important role in disease, he noted.
Markers for these cells include ACTA2, which was previously known to be a marker, and which is almost exclusively restricted to CAF-1, and the cell surface protein CD34 – a progenitor marker in many different cellular systems, “which is actually beautifully expressed on the CAF-2 population” as demonstrated using CITE-seq.
“So we’re now using this as a way to prospectively identify these cells, pull them out of tumors, and conduct biologic assays to learn more about them,” he said.
The immune milieu
“We’re in the age of immunotherapy, and this is an area of huge interest, but we have a long way to go in making it as effective as possible for breast cancer patients,” Dr. Swarbrick said. “I believe part of that is through a very deep understanding of the taxonomy.”
RNA data alone are useful but insufficient to fully identify subsets of immune cells due to a “relatively low-resolution ability to resolve T cells.”
“But because we’re now using the panel of 125 antibodies in parallel, we can now start to use protein levels to split up these populations and we can start to now identify, with higher resolution, more unique populations within the environment,” he said, noting that the availability of protein data not only helps identify subtypes, but is also therapeutically important as it allows for certainty regarding whether the protein target of therapeutic antibodies is expressed on the surface of cells.
Ultimately the hope is that this effort to build a multi-omic breast cancer atlas will continue to drive new discoveries in personalized medicine for breast cancer, Dr. Swarbrick concluded, adding that the field is moving fast, and it will be very important for labs like his and the Navin Lab to communicate to avoid needlessly duplicating efforts.
“I think it’s going to be really exciting to start to put some of these [findings] together,” he said.
The MD Anderson project is funded by the Chan Zuckerberg Initiative as part of its work in supporting the Human Cell Atlas project. Ms. Seth reported having no disclosures. Dr. Swarbrick’s research is funded by the Australian Government/National Health and Medical Research Council and the National Breast Cancer Foundation. He reported having no relevant disclosures.
SOURCE: Seth T et al. SABCS 2018, Abstract GS1-02; Swarbrick A et al. SABCS 2018, Abstract GS1-01
REPORTING FROM SABCS 2018
Key clinical point: Improved understanding of the cellular landscape of both normal breast tissue and breast cancer could lead to new stromal- and immune-based therapies.
Major finding: From pathologically normal breast tissues expression, investigators identified three major cell types, as well as several minor cell types. In analyses of cells from breast cancer patients, a map was created that showed the cell clusters identified by both canonical markers and gene expression signatures.
Study details: An analysis of 32,138 breast cells from 11 women, and another of about 125,000 cells from 25 patients.
Disclosures: The MD Anderson research is part of the Human Cell Atlas project and is funded by the Chan Zuckerberg Initiative. Ms. Seth reported having no disclosures. Dr. Swarbrick’s research is funded by the Australian Government/National Health and Medical Research Council and the National Breast Cancer Foundation. He reported having no relevant disclosures.
Source: Seth T et al. SABCS 2018: Abstract GS1-02; Swarbrick A et al. SABCS 2018: Abstract GS1-01.
Podcasting, burnout, oncology: Dr. David Henry
In this episode, Nick and Dr. Henry discuss advances and excitement in oncology, as well as the opportunities that podcasting provides and tips for dealing with burnout.
Apple Podcasts
Google Podcasts
Spotify
In this episode, Nick and Dr. Henry discuss advances and excitement in oncology, as well as the opportunities that podcasting provides and tips for dealing with burnout.
Apple Podcasts
Google Podcasts
Spotify
In this episode, Nick and Dr. Henry discuss advances and excitement in oncology, as well as the opportunities that podcasting provides and tips for dealing with burnout.
Apple Podcasts
Google Podcasts
Spotify