User login
Pulmonary sarcoidosis: New genetic clues and ongoing treatment controversies
Balancing Antiretroviral Benefit and MI Risk
Serious Hand-and-Foot Syndrome in Black Patients Treated With Capecitabine: Report of 3 Cases and Review of the Literature
Using antipsychotics in patients with dementia
Three keys can help you safely treat dementia’s difficult behavioral and psychological symptoms:
- Differentiate medical from psychiatric causes of patients’ distress.
- Use antipsychotics and other drugs as adjuncts to psychosocial treatments.
- Start low and go slow when titrating dosages.
Although no treatment reverses the pathophysiology of progressive neurodegenerative disorders, managing agitation and other behaviors can alleviate patient suffering and reduce caregiver stress. Based on the evidence and our experience, this article describes a practical approach, including a treatment algorithm and evidence of atypical antipsychotics’ efficacy and side effects in this patient population.
Algorithm Treating behavioral symptoms in patients with dementia
Dementia’s behavioral symptoms
An International Psychogeriatric Association consensus statement1 grouped dementia’s behavioral and psychological symptoms into two types:
- those usually assessed by interviewing patients and relatives—anxiety, depressed mood, hallucinations, and delusions
- those usually identified by observing patient behavior—aggression, screaming, restlessness, agitation, wandering, culturally inappropriate behaviors, sexual disinhibition, hoarding, cursing, and shadowing.
These behaviors in community-living patients are distressing to family members and increase the risk for caregiver burnout—the most common reason for placing older patients in long-term care. In the nursing home, dementia’s symptoms reduce patients’ quality of life; interfere with feeding, bathing, and dressing; and—when violent—may endanger staff and other patients.
Rule out a medical cause
Differential diagnosis. Behavioral symptoms in dementia tend to be unpredictable, which makes diagnosis and treatment challenging. The first step is to determine if a medical or psychiatric condition might account for the behavior. For instance:
- A patient with dementia may be agitated because of a distended bladder or arthritis but unable to communicate his or her pain in words.
- In mild dementia, a pre-existing psychiatric disorder such as schizophrenia might be causing a patient’s hallucinations or delusions.
- Pacing and restlessness may be drug side effects and might be controlled by reducing dosages or switching to less-activating agents.
Delirium is also a risk for older patients—especially those with degenerative neurologic disorders. Common triggers in older patients include acute illness such as a urinary tract infection or pneumonia, alcohol or benzodiazepine withdrawal, anticholinergic agents, medication changes, and dehydration.
Delirium is characterized by acute onset and fluctuating neuropsychiatric symptoms, including disturbed consciousness and changes in attention and cognition. Taking a careful history to learn the course of treatment and the patient’s baseline cognitive function can help you differentiate dementia from delirium. Family members, physicians, and nursing staff are valuable sources of this information.
Use antipsychotics as adjuncts
Psychosocial interventions. After medical causes have been ruled out, consensus guidelines2 recommend psychosocial interventions as first-line treatment of dementia’s behavioral symptoms (Algorithm). Suggested interventions for patients and caregivers are listed in Table 1.3
Antipsychotics. For patients who respond inadequately to psychosocial measures, the next step is to add an atypical antipsychotic. Because of side effects, conventional antipsychotics are not recommended for patients with dementia.
When prescribing atypicals, remember that older adults:
- are more sensitive to side effects than younger adults
- require lower starting and target dosages
- exhibit heterogeneity of response.
Older patients’ medical status can range from “fit” to “frail,” which influences individual response to medications. Generally, age-related changes in the way their bodies metabolize drugs account for older patients’ increased sensitivity to drug side effects (Box).4-11
Atypical antipsychotics and dosages that have been shown benefit for managing behavioral symptoms in older patients with dementia include:
- risperidone, 0.5 to 1.5 mg/d12
- olanzapine, 5 to 10 mg/d13
- quetiapine, 25 to 350 mg/d14 (Table 2).15,16
Start with low dosages, and titrate slowly. Increase once or twice a week until the lowest effective dosage is reached.
Augmenting agents. If antipsychotic monotherapy fails to achieve an adequate response or if side effects limit dosing, adjunctive agents may be added with caution. Augmenting agents that have shown benefit in some patients with dementia include:
- mood stabilizers such as divalproex17 or carbamazepine18
- cholinesterase inhibitors, such as donepezil, rivastigmine, or galantamine.19
Start divalproex at 125 mg bid or carbamazepine at 100 mg bid and titrate to effect. Concomitant carbamazepine will decrease blood levels of risperidone, olanzapine, and quetiapine because of hepatic enzyme induction.20
Start donepezil at 5 mg once daily and increase after 4 to 6 weeks to 10 mg qd. When using rivastigmine, start with 1.5 mg bid and titrate to 9 to 12 mg/d in divided doses. Start galantamine at 4 mg bid and increase after 1 month to 8 mg bid.
Table 1
Suggested psychosocial interventions for older patients with dementia
Communicate clearly
|
Minimize the impact of sensory deficits
|
Modify environment when necessary
|
Encourage consistent daily routines
|
Optimize social/physical stimulation
|
Encourage caregiver to:
|
Antipsychotic side effects
Atypical antipsychotics are more effective than conventional agents in treating negative symptoms and are associated with lower rates of extrapyramidal symptoms (EPS) and tardive dyskinesia (TD).21
Tardive dyskinesia. All antipsychotics can cause TD, although the risk is about 10 times greater with conventionals than atypicals. With conventionals, the annual cumulative TD incidence for young adults is 4 to 5%,22 and rates are much higher for middle-aged and older adults receiving chronic therapy:
- 29% after 1 year
- 50% after 2 years
- 63% after 3 years.23
In older patients, use atypical rather than conventional antipsychotics to minimize TD risk. Observe carefully; if TD symptoms occur, cautiously withdraw the antipsychotic and consider trying another agent.
Other risks. Atypical antipsychotics may cause sedation, orthostatic hypotension (with an increased risk for falls), increased serum prolactin, and weight gain (Table 2).
Weight gain from atypical antipsychotics has been associated with adverse effects on glucose metabolism and increased risk for type 2 diabetes.24 Some might argue that weight gain associated with olanzapine and other atypicals might benefit low-weight older patients. The frail elderly need to increase muscle mass, however, and the atypicals are associated with increases in fat mass.
Increased serum prolactin with risperidone theoretically could lead to loss of bone density, but evidence of this effect in older patients does not exist.
Start low, go slow
Clozapine may help control treatment-resistant psychosis in patients with schizophrenia and manage patients with severe TD.25 However, clozapine’s increased risk of agranulocytosis, neurologic side effects (seizures, sedation, confusion), and anticholinergic effects limit its use in older patients, particularly those with neurodegenerative disorders (Table 2).
Dosing. In rare cases when using clozapine in older patients, start with 6.25 to 12.5 mg/d. Increase by 6.25 to 12.5 mg once or twice a week to 50 to 100 mg/d.
Risperidone has been used to treat agitation in older patients with dementia in two small studies:
In a 9-week, open-label trial, 15 patients (mean age 78) with dementia were given risperidone, 0.5 to 3 mg/d. Agitation improved significantly, as measured by the Cohen-Mansfield Agitation Inventory (CMAI)—a 29-item questionnaire completed by caregivers.26 CMAI scores at study’s end averaged 49.5, compared with 70.5 at baseline.27
A 12-week, placebo-controlled, doubleblind study examined risperidone—0.5, 1, or 2 mg/d—in 625 institutionalized patients (mean age 83) with dementia and agitation. Ninety-six patients had Functional Assessment Staging Rating Scale scores of 6A, indicating moderate to severe dementia. In patients receiving risperidone, these behavioral measures were significantly reduced:
- Behavior Pathology in Alzheimer’s Disease Rating Scale (BEHAVE-AD) total scores, which measure behavior severity
- BEHAVE-AD psychosis subscale scores
- BEHAVE-AD aggressiveness scores
- CMAI verbal and aggression scores.
Adverse effects were reported at 82% for all three risperidone dosages and 85% for placebo. Side effects including somnolence, EPS, and peripheral edema were dose-related.12
Another trial compared risperidone or haloperidol, 0.5 to 4 mg/d, with placebo in treating 344 patients with behavioral symptoms of dementia. After 12 weeks of risperidone, mean dosage 1.1 mg/d:
- mean total BEHAVE-AD score decreased by 53%, compared with 37% in the placebo group
- CMAI score decreased by 32%, compared with 18% in the placebo group.
EPS were more severe with haloperidol than with risperidone or placebo.28
Risk of stroke. A small but significantly increased incidence of stroke and stroke-like events was recently reported in older patients with dementia when treated with risperidone. These events occurred in double-blind, placebocontrolled trials in patients (mean age 82) with Alzheimer’s, vascular, and mixed dementias.
Pharmacokinetic changes can influence concentrations of drugs in tissue compartments over time. Drug absorption declines with normal aging, but a clinically significant decrease in total absorption of psychotropics appears not to occur.13
In the liver, lipid-soluble psychotropics are metabolized into pharmacologically active or inactive metabolites. Some metabolic pathways, such as demethylation, may be influenced by age, leading to increased plasma concentrations of drugs and their metabolites.14,15 However, hydroxylation tends not to be affected by age.16
The ratio of body fat to water increases with aging,13 increasing the volume of distribution for lipid-soluble psychotropics. An age-related decrease in glomerular filtration accounts in part for increased accumulation of hydrophilic metabolites in some older patients.17,18
Pharmacodynamic changes with aging occur in neurotransmitter systems within cellular processing, such as at receptor or reuptake levels.19 These changes may exaggerate drug-drug interactions or affect complex neurotransmitter interactions.
The number of neurons in nigrostriatal pathways declines with age. Decreases are also seen in tyrosine hydroxylase activity, presynaptic dopamine D2 receptors, and dopamine levels—which may be particularly relevant to a discussion of antipsychotic medications.20
The net effect of these changes is the need to prescribe lower-than-usual starting and target dosages of many medications, including antipsychotics.
Most patients who experienced cerebrovascular events had one or more stroke risk factors, including diabetes, hypertension, atrial fibrillation, heart arrhythmia, atherosclerosis, or heart failure. They did not show a pattern of reduced blood pressure or orthostatic changes.12,29
Table 2
Antipsychotic side effects and dosages in older patients with dementia*
| Side effect | Clozapine (6.25 to 100 mg/d) | Risperidone (0.5 to 1.5 mg/d) | Olanzapine (5 to 10 mg/d) | Quetiapine (25 to 350 mg/d) |
|---|---|---|---|---|
| Orthostasis | ++++ | ++++ | +++ | ++ |
| Sedation | +++++ | ++ | +++ | ++ |
| Prolactin increase | 0 | +++ | + | 0 |
| Weight gain | ++++ | + | +++ | + |
| EPS | 0/+ | ++ | + | 0/+ |
| Tardive dyskinesia | 0 | + | + | ? |
| Anticholinergic effects | ++++ | + | + | 0 |
| Seizure risk | +++ | + | + | + |
| Hematologic effects | +++ | + | + | + |
| Source: Adapted from references 15 and 16. | ||||
| * Side-effect profiles and recommended dosages of ziprasidone and aripiprazole in older patients are not yet established. | ||||
| EPS: Extrapyramidal symptoms | ||||
| Key: | ||||
| 0 = none | ||||
| + = slight | ||||
| +++ = mild | ||||
| +++++ = marked | ||||
| 0/+ = none to slight | ||||
| ++ = very mild | ||||
| ++++ = moderate | ||||
Dosing. For older patients with dementia and psychosis, start risperidone at 0.25 to 0.5 mg/d and increase by no more than 0.25 to 0.5 mg once or twice per week. Do not exceed 3 mg/d. For agitation, a 1998 Expert Consensus Guideline Series panel2 recommended starting risperidone at 0.25 to 0.5 mg/d and increasing to an average of 0.5 to 1.5 mg/d.
Olanzapine. Two double-blind, placebo-controlled studies have examined olanzapine in treating agitation associated with dementia.
Saterlee et al30 compared olanzapine, mean 2.4 mg/d, with placebo in outpatients (mean age 79) with Alzheimer’s disease and psychosis. No significant differences were noted in hepatic transaminase levels, leukopenia, EPS, or orthostatic changes.
In a later study,13 nursing home patients (mean age 83) with Alzheimer’s disease, psychosis, and agitation were randomly assigned to receive olanzapine—5, 10, or 15 mg/d—or placebo. After 6 weeks, patients receiving olanzapine, 5 or 10 mg/d, showed significant improvement in Neuropsychiatric Inventory (NPI) total core scores. Olanzapine, 15 mg/d, was not significantly more effective than placebo.
Adverse events such as somnolence and abnormal gait occurred more often with olanzapine than placebo. The somnolence rate with olanzapine was 14% for 5 mg/d and 13% for 10 mg/d, compared with 3% for placebo. For abnormal gait, the rate with olanzapine was 11% for 5 mg/d and 7% for 10 mg/d, compared with 1% for placebo.
Dosing. Start olanzapine at 2.5 mg/d, and increase after 1 to 3 days to 5 mg/d. If symptoms are not adequately controlled, titrate by 2.5-mg increments to 10 mg/d.
Quetiapine. One open-label study14 examined using quetiapine in older patients with psychotic disorders. The study enrolled 184 patients (mean age 76) with Alzheimer’s disease, Parkinson’s disease, schizophrenia, vascular dementia, schizoaffective disorder, bipolar disorder, or major depression. Before the trial, patients were taking various conventional and atypical antipsychotics.
Brief Psychiatric Rating Scale (BPRS) and Clinical Global Impressions (CGI) scores improved significantly after 52 weeks of quetiapine, median 137.5 mg/d. BPRS scores improved 20% in 49% of patients who completed the study.
Less than one-half (48%) of enrolled patients completed the study. Reasons for withdrawal included lack of efficacy (19%), adverse events or illness (15%; adverse events alone, 11%), lost to follow-up (13%), protocol noncompliance (3%), or diminished need for treatment (2%).
EPS occurred in 13% of patients. Mean total scores on the Simpson-Angus Rating Scale for Extrapyramidal Side Effects decreased 1.8 points, indicating reduced parkinsonian symptoms.
Dosing. Start quetiapine at 25 mg once at bedtime or bid; increase in 25-mg increments until the lowest effective dosage is achieved.
Ziprasidone. Little data exist on using ziprasidone in long-term care. In one recent study,31 ziprasidone (mean 100 mg/d) was given to 62 patients ages 64 to 92 with medical illnesses plus major depression, bipolar disorder, schizoaffective disorder, Alzheimer’s disease, or multi-infarct dementia. A retrospective chart review of 10 patients showed decreased agitation, as mean NPI scores declined from 76 to 33.
Sedation was the most common side effect. QTc findings, postural hypotension, and syncope rates did not change. Despite its limitations, this study suggests that ziprasidone is safe and effective in treating psychosis associated with dementia or other disorders.
Aripiprazole. As with ziprasidone, little data exist to guide the use of aripiprazole in older patients. In a randomized preliminary trial,32 192 noninstitutionalized patients with Alzheimer’s disease and psychosis were treated for 10 weeks with aripiprazole, mean 10 mg/d, or placebo.
At 8 and 10 weeks, BPRS psychosis subscale scores improved significantly in patients taking aripiprazole, compared with placebo. EPS and akathisia improved, and somnolence was the most common side effect. Although this study enrolled noninstitutionalized patients with dementia, the results suggest that aripiprazole may help treat long-term care residents with neurodegenerative disorders and behavioral disturbances.
Related resources
- Zaraa AS. Dementia update: Pharmacologic management of agitation and psychosis in older demented patients. Geriatrics 2003;58(10):48-53.
- Mills EJ, Chow TW. Randomized controlled trials in long-term care of residents with dementia: a systematic review. J Am Med Dir Assoc 2003;4(6):302-7.
- Alzheimer’s Association. Treating agitation. www.alz.org/PhysCare/Treating/agitation.htm
Drug brand names
- Aripiprazole • Abilify
- Carbamazepine • Tegretol
- Clozapine • Clozaril
- Donepezil • Aricept
- Galantamine • Reminyl
- Haloperidol • Haldol
- Olanzapine • Zyprexa
- Quetiapine • Seroquel
- Risperidone • Risperdal
- Rivastigmine • Exelon
- Valproate • Depakote
- Ziprasidone • Geodon
Disclosure
Dr. Kasckow receives research support from, is a consultant to, or is a speaker for Eli Lilly & Co., Forest Laboratories, Solvay Pharmaceuticals, AstraZeneca Pharmaceuticals, Organon, Janssen Pharmaceutica, and Pfizer Inc.
Dr. Mulchahey reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Dr. Mohamed receives research support form Forest Laboratories and is a speaker for Eli Lilly & Co.
1. Finkel S, Costa e Silva J, Cohen G, et al. Behavioral and psychological symptoms of dementia: a consensus statement on current knowledge and implications for research and treatment. Am J Geriatr Psychiatry 1998;6:97-100.
2. The Expert Consensus Panel for Agitation in Dementia. Treatment of agitation in older persons with dementia. Postgrad Med 1998;4(suppl):1-88.
3. Cohen-Mansfield J. Nonpharmacologic interventions for inappropriate behaviors in dementia: a review, summary, and critique. Am J Geriatr Psychiatry 2001;9(4):361-81.
4. Davidson J. Pharmacologic treatment. In: Busse E, Blazer D (eds). Textbook of geriatric psychiatry (2nd ed). Washington DC: American Psychiatric Publishing, 1996:359-79.
5. Nies A, Robinson DS, Friedman MJ, et al. Relationship between age and tricyclic antidepressant plasma levels. Am J Psychiatry 1977;134(7):790-3.
6. Greenblatt DJ, Shader RJ. Benzodiazepine kinetics in the elderly. In: Usdin E (ed). Clinical pharmacology in psychiatry. New York: Elsevier, 1981;174-81.
7. Pollock BG, Perel JM, Altieri LP, et al. Debrisoquine hydroxylation phenotyping in geriatric psychopharmacology. Psychopharmacol Bull. 1992;28(2):163-8.
8. Nelson JC, Atillasoy E, Mazure C, Jatlow PI. Hydroxydesipramine in the elderly. J Clin Psychopharmacol 1988;8(6):428-33.
9. Young RC, Alexopoulos GS, Shamoian CA, et al. Plasma 10-hydroxynortriptyline in elderly depressed patients. Clin Pharmacol Ther 1984;35(4):540-4.
10. Cantillon M, Molchan SE, Little J. Pharmacological and neuroendocrine probes in neuropsychiatric illness. In: Coffey CE, Cummings JL (eds). Textbook of geriatric neuropsychiatry. Washington, DC: American Psychiatric Publishing, 1994.
11. Young RC, Meyers BS. Psychopharmacology. In: Sadovoy J, Lazarus LW, Jarvik LF, Grossberg GT (eds). Comprehensive review of geriatric psychiatry. Washington DC: American Psychiatric Publishing, 1996;755-817.
12. Katz IR, Jeste DV, Mintzer JE, et al. Comparison of risperidone and placebo for psychosis and behavioral disturbances associated with dementia: a randomized, double-blind trial. J Clin Psychiatry 1999;60(2):107-15.
13. Street JS, Clark WS, Gannon KS, et al. Olanzapine treatment of psychotic and behavioral symptoms in patients with Alzheimer disease in nursing care facilities: a double-blind, randomized, placebo-controlled trial. The HGEU Study Group. Arch Gen Psychiatry 2000;57(10):968-76.
14. Tariot PN, Salzman C, Yeung PP, et al. Long-term use of quetiapine in elderly patients with psychotic disorders. Clin Ther 2000;22(9):1068-84.
15. Casey DE. The relationship of pharmacology to side effects. J Clin Psychiatry 1997;58(suppl):55-62.
16. Pickar D. Prospects for pharmacotherapy of schizophrenia. Lancet 1995;345:557-62.
17. Kasckow JW, McElroy SL, Cameron RL, et al. A pilot study on the use of divalproex sodium in the treatment of behavioral agitation in elderly patients with dementia: assessment with the BEHAVE-AD and CGI rating scales. Curr Ther Res 1997;58(12):981-9.
18. Tariot PN, Erb R, Podgorski CA, et al. Efficacy and tolerability of carbamazepine for agitation and aggression in dementia. Am J Psychiatry 1998;155(1):54-61.
19. Kasckow JW. Cognitive enhancers for dementia: do they work? Current Psychiatry 2002;1(3):22-8.
20. Lacy C, Armstrong L, Goldman M, Lance L. (eds) Lexicomp drug information handbook. Hudson, OH: Lexicomp, 2003-2004:1225-27, 1189-90, 1026-27.
21. Jeste DV, Lacro JP, Bailey A, et al. Lower incidence of tardive dyskinesia with risperidone compared with haloperidol in older patients. J Am Geriatr Soc 1999;47(6):716-19.
22. Kane J, Honigfeld G, Singer J, Meltzer H. Clozapine for the treatment-resistant schizophrenic. A double-blind comparison with chlorpromazine. Arch Gen Psychiatry 1988;45(9):789-96.
23. Jeste DV, Caligiuri MP, Paulsen JS, et al. Risk of tardive dyskinesia in older patients. A prospective longitudinal study of 266 outpatients. Arch Gen Psychiatry 1995;52(9):756-65.
24. Sernyak MJ, Leslie DL, Alarcon RD, et al. Association of diabetes mellitus with use of atypical neuroleptics in the treatment of schizophrenia. Am J Psychiatry 2002;159:561-6.
25. Chengappa KN, Baker RW, Kreinbrook SB, Adair D. Clozapine use in female geriatric patients with psychoses. JGeriatr Psychiatry Neurol 1995;8(1):12-15.
26. Cohen-Mansfield J, Marx MS, Rosenthal AS. A description of agitation in the nursing home. J Gerontol 1989;44(3):M77-84.
27. Lavretsky H, Sultzer D. A structured trial of risperidone for the treatment of agitation in dementia. Am J Geriatr Psychiatry 1998;6(2):127-35.
28. De Deyn PP, Rabheru K, Rasmussen A, et al. A randomized trial of risperidone, placebo, and haloperidol for behavioral symptoms of dementia. Neurology 1999;53(5):946-55.
29. Brodaty H, Ames D, Snowdon J, et al. A randomized placebo-controlled trial of risperidone for the treatment of aggression, agitation, and psychosis of dementia. J Clin Psychiatry 2003;64(2):134-43.
30. Satterlee W, Reams SG, Burns PR, et al. A clinical update on olanzapine treatment in schizophrenia and in elderly Alzheimer’s disease patients (abstract). Psychopharmacol Bull 1995;31:534.-
31. Berkowitz A. Ziprasidone for elderly dementia: a case series (abstract). San Francisco, CA: American Psychiatric Association annual meeting, 2003.
32. De Deyn PP, Jeste D, Auby P, Carson W. Aripiprazole in dementia of the Alzheimer’s type (abstract). Honolulu, HI: American Association for Geriatric Psychiatry annual meeting, 2003.
Three keys can help you safely treat dementia’s difficult behavioral and psychological symptoms:
- Differentiate medical from psychiatric causes of patients’ distress.
- Use antipsychotics and other drugs as adjuncts to psychosocial treatments.
- Start low and go slow when titrating dosages.
Although no treatment reverses the pathophysiology of progressive neurodegenerative disorders, managing agitation and other behaviors can alleviate patient suffering and reduce caregiver stress. Based on the evidence and our experience, this article describes a practical approach, including a treatment algorithm and evidence of atypical antipsychotics’ efficacy and side effects in this patient population.
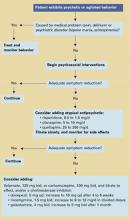
Algorithm Treating behavioral symptoms in patients with dementia
Dementia’s behavioral symptoms
An International Psychogeriatric Association consensus statement1 grouped dementia’s behavioral and psychological symptoms into two types:
- those usually assessed by interviewing patients and relatives—anxiety, depressed mood, hallucinations, and delusions
- those usually identified by observing patient behavior—aggression, screaming, restlessness, agitation, wandering, culturally inappropriate behaviors, sexual disinhibition, hoarding, cursing, and shadowing.
These behaviors in community-living patients are distressing to family members and increase the risk for caregiver burnout—the most common reason for placing older patients in long-term care. In the nursing home, dementia’s symptoms reduce patients’ quality of life; interfere with feeding, bathing, and dressing; and—when violent—may endanger staff and other patients.
Rule out a medical cause
Differential diagnosis. Behavioral symptoms in dementia tend to be unpredictable, which makes diagnosis and treatment challenging. The first step is to determine if a medical or psychiatric condition might account for the behavior. For instance:
- A patient with dementia may be agitated because of a distended bladder or arthritis but unable to communicate his or her pain in words.
- In mild dementia, a pre-existing psychiatric disorder such as schizophrenia might be causing a patient’s hallucinations or delusions.
- Pacing and restlessness may be drug side effects and might be controlled by reducing dosages or switching to less-activating agents.
Delirium is also a risk for older patients—especially those with degenerative neurologic disorders. Common triggers in older patients include acute illness such as a urinary tract infection or pneumonia, alcohol or benzodiazepine withdrawal, anticholinergic agents, medication changes, and dehydration.
Delirium is characterized by acute onset and fluctuating neuropsychiatric symptoms, including disturbed consciousness and changes in attention and cognition. Taking a careful history to learn the course of treatment and the patient’s baseline cognitive function can help you differentiate dementia from delirium. Family members, physicians, and nursing staff are valuable sources of this information.
Use antipsychotics as adjuncts
Psychosocial interventions. After medical causes have been ruled out, consensus guidelines2 recommend psychosocial interventions as first-line treatment of dementia’s behavioral symptoms (Algorithm). Suggested interventions for patients and caregivers are listed in Table 1.3
Antipsychotics. For patients who respond inadequately to psychosocial measures, the next step is to add an atypical antipsychotic. Because of side effects, conventional antipsychotics are not recommended for patients with dementia.
When prescribing atypicals, remember that older adults:
- are more sensitive to side effects than younger adults
- require lower starting and target dosages
- exhibit heterogeneity of response.
Older patients’ medical status can range from “fit” to “frail,” which influences individual response to medications. Generally, age-related changes in the way their bodies metabolize drugs account for older patients’ increased sensitivity to drug side effects (Box).4-11
Atypical antipsychotics and dosages that have been shown benefit for managing behavioral symptoms in older patients with dementia include:
- risperidone, 0.5 to 1.5 mg/d12
- olanzapine, 5 to 10 mg/d13
- quetiapine, 25 to 350 mg/d14 (Table 2).15,16
Start with low dosages, and titrate slowly. Increase once or twice a week until the lowest effective dosage is reached.
Augmenting agents. If antipsychotic monotherapy fails to achieve an adequate response or if side effects limit dosing, adjunctive agents may be added with caution. Augmenting agents that have shown benefit in some patients with dementia include:
- mood stabilizers such as divalproex17 or carbamazepine18
- cholinesterase inhibitors, such as donepezil, rivastigmine, or galantamine.19
Start divalproex at 125 mg bid or carbamazepine at 100 mg bid and titrate to effect. Concomitant carbamazepine will decrease blood levels of risperidone, olanzapine, and quetiapine because of hepatic enzyme induction.20
Start donepezil at 5 mg once daily and increase after 4 to 6 weeks to 10 mg qd. When using rivastigmine, start with 1.5 mg bid and titrate to 9 to 12 mg/d in divided doses. Start galantamine at 4 mg bid and increase after 1 month to 8 mg bid.
Table 1
Suggested psychosocial interventions for older patients with dementia
Communicate clearly
|
Minimize the impact of sensory deficits
|
Modify environment when necessary
|
Encourage consistent daily routines
|
Optimize social/physical stimulation
|
Encourage caregiver to:
|
Antipsychotic side effects
Atypical antipsychotics are more effective than conventional agents in treating negative symptoms and are associated with lower rates of extrapyramidal symptoms (EPS) and tardive dyskinesia (TD).21
Tardive dyskinesia. All antipsychotics can cause TD, although the risk is about 10 times greater with conventionals than atypicals. With conventionals, the annual cumulative TD incidence for young adults is 4 to 5%,22 and rates are much higher for middle-aged and older adults receiving chronic therapy:
- 29% after 1 year
- 50% after 2 years
- 63% after 3 years.23
In older patients, use atypical rather than conventional antipsychotics to minimize TD risk. Observe carefully; if TD symptoms occur, cautiously withdraw the antipsychotic and consider trying another agent.
Other risks. Atypical antipsychotics may cause sedation, orthostatic hypotension (with an increased risk for falls), increased serum prolactin, and weight gain (Table 2).
Weight gain from atypical antipsychotics has been associated with adverse effects on glucose metabolism and increased risk for type 2 diabetes.24 Some might argue that weight gain associated with olanzapine and other atypicals might benefit low-weight older patients. The frail elderly need to increase muscle mass, however, and the atypicals are associated with increases in fat mass.
Increased serum prolactin with risperidone theoretically could lead to loss of bone density, but evidence of this effect in older patients does not exist.
Start low, go slow
Clozapine may help control treatment-resistant psychosis in patients with schizophrenia and manage patients with severe TD.25 However, clozapine’s increased risk of agranulocytosis, neurologic side effects (seizures, sedation, confusion), and anticholinergic effects limit its use in older patients, particularly those with neurodegenerative disorders (Table 2).
Dosing. In rare cases when using clozapine in older patients, start with 6.25 to 12.5 mg/d. Increase by 6.25 to 12.5 mg once or twice a week to 50 to 100 mg/d.
Risperidone has been used to treat agitation in older patients with dementia in two small studies:
In a 9-week, open-label trial, 15 patients (mean age 78) with dementia were given risperidone, 0.5 to 3 mg/d. Agitation improved significantly, as measured by the Cohen-Mansfield Agitation Inventory (CMAI)—a 29-item questionnaire completed by caregivers.26 CMAI scores at study’s end averaged 49.5, compared with 70.5 at baseline.27
A 12-week, placebo-controlled, doubleblind study examined risperidone—0.5, 1, or 2 mg/d—in 625 institutionalized patients (mean age 83) with dementia and agitation. Ninety-six patients had Functional Assessment Staging Rating Scale scores of 6A, indicating moderate to severe dementia. In patients receiving risperidone, these behavioral measures were significantly reduced:
- Behavior Pathology in Alzheimer’s Disease Rating Scale (BEHAVE-AD) total scores, which measure behavior severity
- BEHAVE-AD psychosis subscale scores
- BEHAVE-AD aggressiveness scores
- CMAI verbal and aggression scores.
Adverse effects were reported at 82% for all three risperidone dosages and 85% for placebo. Side effects including somnolence, EPS, and peripheral edema were dose-related.12
Another trial compared risperidone or haloperidol, 0.5 to 4 mg/d, with placebo in treating 344 patients with behavioral symptoms of dementia. After 12 weeks of risperidone, mean dosage 1.1 mg/d:
- mean total BEHAVE-AD score decreased by 53%, compared with 37% in the placebo group
- CMAI score decreased by 32%, compared with 18% in the placebo group.
EPS were more severe with haloperidol than with risperidone or placebo.28
Risk of stroke. A small but significantly increased incidence of stroke and stroke-like events was recently reported in older patients with dementia when treated with risperidone. These events occurred in double-blind, placebocontrolled trials in patients (mean age 82) with Alzheimer’s, vascular, and mixed dementias.
Pharmacokinetic changes can influence concentrations of drugs in tissue compartments over time. Drug absorption declines with normal aging, but a clinically significant decrease in total absorption of psychotropics appears not to occur.13
In the liver, lipid-soluble psychotropics are metabolized into pharmacologically active or inactive metabolites. Some metabolic pathways, such as demethylation, may be influenced by age, leading to increased plasma concentrations of drugs and their metabolites.14,15 However, hydroxylation tends not to be affected by age.16
The ratio of body fat to water increases with aging,13 increasing the volume of distribution for lipid-soluble psychotropics. An age-related decrease in glomerular filtration accounts in part for increased accumulation of hydrophilic metabolites in some older patients.17,18
Pharmacodynamic changes with aging occur in neurotransmitter systems within cellular processing, such as at receptor or reuptake levels.19 These changes may exaggerate drug-drug interactions or affect complex neurotransmitter interactions.
The number of neurons in nigrostriatal pathways declines with age. Decreases are also seen in tyrosine hydroxylase activity, presynaptic dopamine D2 receptors, and dopamine levels—which may be particularly relevant to a discussion of antipsychotic medications.20
The net effect of these changes is the need to prescribe lower-than-usual starting and target dosages of many medications, including antipsychotics.
Most patients who experienced cerebrovascular events had one or more stroke risk factors, including diabetes, hypertension, atrial fibrillation, heart arrhythmia, atherosclerosis, or heart failure. They did not show a pattern of reduced blood pressure or orthostatic changes.12,29
Table 2
Antipsychotic side effects and dosages in older patients with dementia*
| Side effect | Clozapine (6.25 to 100 mg/d) | Risperidone (0.5 to 1.5 mg/d) | Olanzapine (5 to 10 mg/d) | Quetiapine (25 to 350 mg/d) |
|---|---|---|---|---|
| Orthostasis | ++++ | ++++ | +++ | ++ |
| Sedation | +++++ | ++ | +++ | ++ |
| Prolactin increase | 0 | +++ | + | 0 |
| Weight gain | ++++ | + | +++ | + |
| EPS | 0/+ | ++ | + | 0/+ |
| Tardive dyskinesia | 0 | + | + | ? |
| Anticholinergic effects | ++++ | + | + | 0 |
| Seizure risk | +++ | + | + | + |
| Hematologic effects | +++ | + | + | + |
| Source: Adapted from references 15 and 16. | ||||
| * Side-effect profiles and recommended dosages of ziprasidone and aripiprazole in older patients are not yet established. | ||||
| EPS: Extrapyramidal symptoms | ||||
| Key: | ||||
| 0 = none | ||||
| + = slight | ||||
| +++ = mild | ||||
| +++++ = marked | ||||
| 0/+ = none to slight | ||||
| ++ = very mild | ||||
| ++++ = moderate | ||||
Dosing. For older patients with dementia and psychosis, start risperidone at 0.25 to 0.5 mg/d and increase by no more than 0.25 to 0.5 mg once or twice per week. Do not exceed 3 mg/d. For agitation, a 1998 Expert Consensus Guideline Series panel2 recommended starting risperidone at 0.25 to 0.5 mg/d and increasing to an average of 0.5 to 1.5 mg/d.
Olanzapine. Two double-blind, placebo-controlled studies have examined olanzapine in treating agitation associated with dementia.
Saterlee et al30 compared olanzapine, mean 2.4 mg/d, with placebo in outpatients (mean age 79) with Alzheimer’s disease and psychosis. No significant differences were noted in hepatic transaminase levels, leukopenia, EPS, or orthostatic changes.
In a later study,13 nursing home patients (mean age 83) with Alzheimer’s disease, psychosis, and agitation were randomly assigned to receive olanzapine—5, 10, or 15 mg/d—or placebo. After 6 weeks, patients receiving olanzapine, 5 or 10 mg/d, showed significant improvement in Neuropsychiatric Inventory (NPI) total core scores. Olanzapine, 15 mg/d, was not significantly more effective than placebo.
Adverse events such as somnolence and abnormal gait occurred more often with olanzapine than placebo. The somnolence rate with olanzapine was 14% for 5 mg/d and 13% for 10 mg/d, compared with 3% for placebo. For abnormal gait, the rate with olanzapine was 11% for 5 mg/d and 7% for 10 mg/d, compared with 1% for placebo.
Dosing. Start olanzapine at 2.5 mg/d, and increase after 1 to 3 days to 5 mg/d. If symptoms are not adequately controlled, titrate by 2.5-mg increments to 10 mg/d.
Quetiapine. One open-label study14 examined using quetiapine in older patients with psychotic disorders. The study enrolled 184 patients (mean age 76) with Alzheimer’s disease, Parkinson’s disease, schizophrenia, vascular dementia, schizoaffective disorder, bipolar disorder, or major depression. Before the trial, patients were taking various conventional and atypical antipsychotics.
Brief Psychiatric Rating Scale (BPRS) and Clinical Global Impressions (CGI) scores improved significantly after 52 weeks of quetiapine, median 137.5 mg/d. BPRS scores improved 20% in 49% of patients who completed the study.
Less than one-half (48%) of enrolled patients completed the study. Reasons for withdrawal included lack of efficacy (19%), adverse events or illness (15%; adverse events alone, 11%), lost to follow-up (13%), protocol noncompliance (3%), or diminished need for treatment (2%).
EPS occurred in 13% of patients. Mean total scores on the Simpson-Angus Rating Scale for Extrapyramidal Side Effects decreased 1.8 points, indicating reduced parkinsonian symptoms.
Dosing. Start quetiapine at 25 mg once at bedtime or bid; increase in 25-mg increments until the lowest effective dosage is achieved.
Ziprasidone. Little data exist on using ziprasidone in long-term care. In one recent study,31 ziprasidone (mean 100 mg/d) was given to 62 patients ages 64 to 92 with medical illnesses plus major depression, bipolar disorder, schizoaffective disorder, Alzheimer’s disease, or multi-infarct dementia. A retrospective chart review of 10 patients showed decreased agitation, as mean NPI scores declined from 76 to 33.
Sedation was the most common side effect. QTc findings, postural hypotension, and syncope rates did not change. Despite its limitations, this study suggests that ziprasidone is safe and effective in treating psychosis associated with dementia or other disorders.
Aripiprazole. As with ziprasidone, little data exist to guide the use of aripiprazole in older patients. In a randomized preliminary trial,32 192 noninstitutionalized patients with Alzheimer’s disease and psychosis were treated for 10 weeks with aripiprazole, mean 10 mg/d, or placebo.
At 8 and 10 weeks, BPRS psychosis subscale scores improved significantly in patients taking aripiprazole, compared with placebo. EPS and akathisia improved, and somnolence was the most common side effect. Although this study enrolled noninstitutionalized patients with dementia, the results suggest that aripiprazole may help treat long-term care residents with neurodegenerative disorders and behavioral disturbances.
Related resources
- Zaraa AS. Dementia update: Pharmacologic management of agitation and psychosis in older demented patients. Geriatrics 2003;58(10):48-53.
- Mills EJ, Chow TW. Randomized controlled trials in long-term care of residents with dementia: a systematic review. J Am Med Dir Assoc 2003;4(6):302-7.
- Alzheimer’s Association. Treating agitation. www.alz.org/PhysCare/Treating/agitation.htm
Drug brand names
- Aripiprazole • Abilify
- Carbamazepine • Tegretol
- Clozapine • Clozaril
- Donepezil • Aricept
- Galantamine • Reminyl
- Haloperidol • Haldol
- Olanzapine • Zyprexa
- Quetiapine • Seroquel
- Risperidone • Risperdal
- Rivastigmine • Exelon
- Valproate • Depakote
- Ziprasidone • Geodon
Disclosure
Dr. Kasckow receives research support from, is a consultant to, or is a speaker for Eli Lilly & Co., Forest Laboratories, Solvay Pharmaceuticals, AstraZeneca Pharmaceuticals, Organon, Janssen Pharmaceutica, and Pfizer Inc.
Dr. Mulchahey reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Dr. Mohamed receives research support form Forest Laboratories and is a speaker for Eli Lilly & Co.
Three keys can help you safely treat dementia’s difficult behavioral and psychological symptoms:
- Differentiate medical from psychiatric causes of patients’ distress.
- Use antipsychotics and other drugs as adjuncts to psychosocial treatments.
- Start low and go slow when titrating dosages.
Although no treatment reverses the pathophysiology of progressive neurodegenerative disorders, managing agitation and other behaviors can alleviate patient suffering and reduce caregiver stress. Based on the evidence and our experience, this article describes a practical approach, including a treatment algorithm and evidence of atypical antipsychotics’ efficacy and side effects in this patient population.
Algorithm Treating behavioral symptoms in patients with dementia
Dementia’s behavioral symptoms
An International Psychogeriatric Association consensus statement1 grouped dementia’s behavioral and psychological symptoms into two types:
- those usually assessed by interviewing patients and relatives—anxiety, depressed mood, hallucinations, and delusions
- those usually identified by observing patient behavior—aggression, screaming, restlessness, agitation, wandering, culturally inappropriate behaviors, sexual disinhibition, hoarding, cursing, and shadowing.
These behaviors in community-living patients are distressing to family members and increase the risk for caregiver burnout—the most common reason for placing older patients in long-term care. In the nursing home, dementia’s symptoms reduce patients’ quality of life; interfere with feeding, bathing, and dressing; and—when violent—may endanger staff and other patients.
Rule out a medical cause
Differential diagnosis. Behavioral symptoms in dementia tend to be unpredictable, which makes diagnosis and treatment challenging. The first step is to determine if a medical or psychiatric condition might account for the behavior. For instance:
- A patient with dementia may be agitated because of a distended bladder or arthritis but unable to communicate his or her pain in words.
- In mild dementia, a pre-existing psychiatric disorder such as schizophrenia might be causing a patient’s hallucinations or delusions.
- Pacing and restlessness may be drug side effects and might be controlled by reducing dosages or switching to less-activating agents.
Delirium is also a risk for older patients—especially those with degenerative neurologic disorders. Common triggers in older patients include acute illness such as a urinary tract infection or pneumonia, alcohol or benzodiazepine withdrawal, anticholinergic agents, medication changes, and dehydration.
Delirium is characterized by acute onset and fluctuating neuropsychiatric symptoms, including disturbed consciousness and changes in attention and cognition. Taking a careful history to learn the course of treatment and the patient’s baseline cognitive function can help you differentiate dementia from delirium. Family members, physicians, and nursing staff are valuable sources of this information.
Use antipsychotics as adjuncts
Psychosocial interventions. After medical causes have been ruled out, consensus guidelines2 recommend psychosocial interventions as first-line treatment of dementia’s behavioral symptoms (Algorithm). Suggested interventions for patients and caregivers are listed in Table 1.3
Antipsychotics. For patients who respond inadequately to psychosocial measures, the next step is to add an atypical antipsychotic. Because of side effects, conventional antipsychotics are not recommended for patients with dementia.
When prescribing atypicals, remember that older adults:
- are more sensitive to side effects than younger adults
- require lower starting and target dosages
- exhibit heterogeneity of response.
Older patients’ medical status can range from “fit” to “frail,” which influences individual response to medications. Generally, age-related changes in the way their bodies metabolize drugs account for older patients’ increased sensitivity to drug side effects (Box).4-11
Atypical antipsychotics and dosages that have been shown benefit for managing behavioral symptoms in older patients with dementia include:
- risperidone, 0.5 to 1.5 mg/d12
- olanzapine, 5 to 10 mg/d13
- quetiapine, 25 to 350 mg/d14 (Table 2).15,16
Start with low dosages, and titrate slowly. Increase once or twice a week until the lowest effective dosage is reached.
Augmenting agents. If antipsychotic monotherapy fails to achieve an adequate response or if side effects limit dosing, adjunctive agents may be added with caution. Augmenting agents that have shown benefit in some patients with dementia include:
- mood stabilizers such as divalproex17 or carbamazepine18
- cholinesterase inhibitors, such as donepezil, rivastigmine, or galantamine.19
Start divalproex at 125 mg bid or carbamazepine at 100 mg bid and titrate to effect. Concomitant carbamazepine will decrease blood levels of risperidone, olanzapine, and quetiapine because of hepatic enzyme induction.20
Start donepezil at 5 mg once daily and increase after 4 to 6 weeks to 10 mg qd. When using rivastigmine, start with 1.5 mg bid and titrate to 9 to 12 mg/d in divided doses. Start galantamine at 4 mg bid and increase after 1 month to 8 mg bid.
Table 1
Suggested psychosocial interventions for older patients with dementia
Communicate clearly
|
Minimize the impact of sensory deficits
|
Modify environment when necessary
|
Encourage consistent daily routines
|
Optimize social/physical stimulation
|
Encourage caregiver to:
|
Antipsychotic side effects
Atypical antipsychotics are more effective than conventional agents in treating negative symptoms and are associated with lower rates of extrapyramidal symptoms (EPS) and tardive dyskinesia (TD).21
Tardive dyskinesia. All antipsychotics can cause TD, although the risk is about 10 times greater with conventionals than atypicals. With conventionals, the annual cumulative TD incidence for young adults is 4 to 5%,22 and rates are much higher for middle-aged and older adults receiving chronic therapy:
- 29% after 1 year
- 50% after 2 years
- 63% after 3 years.23
In older patients, use atypical rather than conventional antipsychotics to minimize TD risk. Observe carefully; if TD symptoms occur, cautiously withdraw the antipsychotic and consider trying another agent.
Other risks. Atypical antipsychotics may cause sedation, orthostatic hypotension (with an increased risk for falls), increased serum prolactin, and weight gain (Table 2).
Weight gain from atypical antipsychotics has been associated with adverse effects on glucose metabolism and increased risk for type 2 diabetes.24 Some might argue that weight gain associated with olanzapine and other atypicals might benefit low-weight older patients. The frail elderly need to increase muscle mass, however, and the atypicals are associated with increases in fat mass.
Increased serum prolactin with risperidone theoretically could lead to loss of bone density, but evidence of this effect in older patients does not exist.
Start low, go slow
Clozapine may help control treatment-resistant psychosis in patients with schizophrenia and manage patients with severe TD.25 However, clozapine’s increased risk of agranulocytosis, neurologic side effects (seizures, sedation, confusion), and anticholinergic effects limit its use in older patients, particularly those with neurodegenerative disorders (Table 2).
Dosing. In rare cases when using clozapine in older patients, start with 6.25 to 12.5 mg/d. Increase by 6.25 to 12.5 mg once or twice a week to 50 to 100 mg/d.
Risperidone has been used to treat agitation in older patients with dementia in two small studies:
In a 9-week, open-label trial, 15 patients (mean age 78) with dementia were given risperidone, 0.5 to 3 mg/d. Agitation improved significantly, as measured by the Cohen-Mansfield Agitation Inventory (CMAI)—a 29-item questionnaire completed by caregivers.26 CMAI scores at study’s end averaged 49.5, compared with 70.5 at baseline.27
A 12-week, placebo-controlled, doubleblind study examined risperidone—0.5, 1, or 2 mg/d—in 625 institutionalized patients (mean age 83) with dementia and agitation. Ninety-six patients had Functional Assessment Staging Rating Scale scores of 6A, indicating moderate to severe dementia. In patients receiving risperidone, these behavioral measures were significantly reduced:
- Behavior Pathology in Alzheimer’s Disease Rating Scale (BEHAVE-AD) total scores, which measure behavior severity
- BEHAVE-AD psychosis subscale scores
- BEHAVE-AD aggressiveness scores
- CMAI verbal and aggression scores.
Adverse effects were reported at 82% for all three risperidone dosages and 85% for placebo. Side effects including somnolence, EPS, and peripheral edema were dose-related.12
Another trial compared risperidone or haloperidol, 0.5 to 4 mg/d, with placebo in treating 344 patients with behavioral symptoms of dementia. After 12 weeks of risperidone, mean dosage 1.1 mg/d:
- mean total BEHAVE-AD score decreased by 53%, compared with 37% in the placebo group
- CMAI score decreased by 32%, compared with 18% in the placebo group.
EPS were more severe with haloperidol than with risperidone or placebo.28
Risk of stroke. A small but significantly increased incidence of stroke and stroke-like events was recently reported in older patients with dementia when treated with risperidone. These events occurred in double-blind, placebocontrolled trials in patients (mean age 82) with Alzheimer’s, vascular, and mixed dementias.
Pharmacokinetic changes can influence concentrations of drugs in tissue compartments over time. Drug absorption declines with normal aging, but a clinically significant decrease in total absorption of psychotropics appears not to occur.13
In the liver, lipid-soluble psychotropics are metabolized into pharmacologically active or inactive metabolites. Some metabolic pathways, such as demethylation, may be influenced by age, leading to increased plasma concentrations of drugs and their metabolites.14,15 However, hydroxylation tends not to be affected by age.16
The ratio of body fat to water increases with aging,13 increasing the volume of distribution for lipid-soluble psychotropics. An age-related decrease in glomerular filtration accounts in part for increased accumulation of hydrophilic metabolites in some older patients.17,18
Pharmacodynamic changes with aging occur in neurotransmitter systems within cellular processing, such as at receptor or reuptake levels.19 These changes may exaggerate drug-drug interactions or affect complex neurotransmitter interactions.
The number of neurons in nigrostriatal pathways declines with age. Decreases are also seen in tyrosine hydroxylase activity, presynaptic dopamine D2 receptors, and dopamine levels—which may be particularly relevant to a discussion of antipsychotic medications.20
The net effect of these changes is the need to prescribe lower-than-usual starting and target dosages of many medications, including antipsychotics.
Most patients who experienced cerebrovascular events had one or more stroke risk factors, including diabetes, hypertension, atrial fibrillation, heart arrhythmia, atherosclerosis, or heart failure. They did not show a pattern of reduced blood pressure or orthostatic changes.12,29
Table 2
Antipsychotic side effects and dosages in older patients with dementia*
| Side effect | Clozapine (6.25 to 100 mg/d) | Risperidone (0.5 to 1.5 mg/d) | Olanzapine (5 to 10 mg/d) | Quetiapine (25 to 350 mg/d) |
|---|---|---|---|---|
| Orthostasis | ++++ | ++++ | +++ | ++ |
| Sedation | +++++ | ++ | +++ | ++ |
| Prolactin increase | 0 | +++ | + | 0 |
| Weight gain | ++++ | + | +++ | + |
| EPS | 0/+ | ++ | + | 0/+ |
| Tardive dyskinesia | 0 | + | + | ? |
| Anticholinergic effects | ++++ | + | + | 0 |
| Seizure risk | +++ | + | + | + |
| Hematologic effects | +++ | + | + | + |
| Source: Adapted from references 15 and 16. | ||||
| * Side-effect profiles and recommended dosages of ziprasidone and aripiprazole in older patients are not yet established. | ||||
| EPS: Extrapyramidal symptoms | ||||
| Key: | ||||
| 0 = none | ||||
| + = slight | ||||
| +++ = mild | ||||
| +++++ = marked | ||||
| 0/+ = none to slight | ||||
| ++ = very mild | ||||
| ++++ = moderate | ||||
Dosing. For older patients with dementia and psychosis, start risperidone at 0.25 to 0.5 mg/d and increase by no more than 0.25 to 0.5 mg once or twice per week. Do not exceed 3 mg/d. For agitation, a 1998 Expert Consensus Guideline Series panel2 recommended starting risperidone at 0.25 to 0.5 mg/d and increasing to an average of 0.5 to 1.5 mg/d.
Olanzapine. Two double-blind, placebo-controlled studies have examined olanzapine in treating agitation associated with dementia.
Saterlee et al30 compared olanzapine, mean 2.4 mg/d, with placebo in outpatients (mean age 79) with Alzheimer’s disease and psychosis. No significant differences were noted in hepatic transaminase levels, leukopenia, EPS, or orthostatic changes.
In a later study,13 nursing home patients (mean age 83) with Alzheimer’s disease, psychosis, and agitation were randomly assigned to receive olanzapine—5, 10, or 15 mg/d—or placebo. After 6 weeks, patients receiving olanzapine, 5 or 10 mg/d, showed significant improvement in Neuropsychiatric Inventory (NPI) total core scores. Olanzapine, 15 mg/d, was not significantly more effective than placebo.
Adverse events such as somnolence and abnormal gait occurred more often with olanzapine than placebo. The somnolence rate with olanzapine was 14% for 5 mg/d and 13% for 10 mg/d, compared with 3% for placebo. For abnormal gait, the rate with olanzapine was 11% for 5 mg/d and 7% for 10 mg/d, compared with 1% for placebo.
Dosing. Start olanzapine at 2.5 mg/d, and increase after 1 to 3 days to 5 mg/d. If symptoms are not adequately controlled, titrate by 2.5-mg increments to 10 mg/d.
Quetiapine. One open-label study14 examined using quetiapine in older patients with psychotic disorders. The study enrolled 184 patients (mean age 76) with Alzheimer’s disease, Parkinson’s disease, schizophrenia, vascular dementia, schizoaffective disorder, bipolar disorder, or major depression. Before the trial, patients were taking various conventional and atypical antipsychotics.
Brief Psychiatric Rating Scale (BPRS) and Clinical Global Impressions (CGI) scores improved significantly after 52 weeks of quetiapine, median 137.5 mg/d. BPRS scores improved 20% in 49% of patients who completed the study.
Less than one-half (48%) of enrolled patients completed the study. Reasons for withdrawal included lack of efficacy (19%), adverse events or illness (15%; adverse events alone, 11%), lost to follow-up (13%), protocol noncompliance (3%), or diminished need for treatment (2%).
EPS occurred in 13% of patients. Mean total scores on the Simpson-Angus Rating Scale for Extrapyramidal Side Effects decreased 1.8 points, indicating reduced parkinsonian symptoms.
Dosing. Start quetiapine at 25 mg once at bedtime or bid; increase in 25-mg increments until the lowest effective dosage is achieved.
Ziprasidone. Little data exist on using ziprasidone in long-term care. In one recent study,31 ziprasidone (mean 100 mg/d) was given to 62 patients ages 64 to 92 with medical illnesses plus major depression, bipolar disorder, schizoaffective disorder, Alzheimer’s disease, or multi-infarct dementia. A retrospective chart review of 10 patients showed decreased agitation, as mean NPI scores declined from 76 to 33.
Sedation was the most common side effect. QTc findings, postural hypotension, and syncope rates did not change. Despite its limitations, this study suggests that ziprasidone is safe and effective in treating psychosis associated with dementia or other disorders.
Aripiprazole. As with ziprasidone, little data exist to guide the use of aripiprazole in older patients. In a randomized preliminary trial,32 192 noninstitutionalized patients with Alzheimer’s disease and psychosis were treated for 10 weeks with aripiprazole, mean 10 mg/d, or placebo.
At 8 and 10 weeks, BPRS psychosis subscale scores improved significantly in patients taking aripiprazole, compared with placebo. EPS and akathisia improved, and somnolence was the most common side effect. Although this study enrolled noninstitutionalized patients with dementia, the results suggest that aripiprazole may help treat long-term care residents with neurodegenerative disorders and behavioral disturbances.
Related resources
- Zaraa AS. Dementia update: Pharmacologic management of agitation and psychosis in older demented patients. Geriatrics 2003;58(10):48-53.
- Mills EJ, Chow TW. Randomized controlled trials in long-term care of residents with dementia: a systematic review. J Am Med Dir Assoc 2003;4(6):302-7.
- Alzheimer’s Association. Treating agitation. www.alz.org/PhysCare/Treating/agitation.htm
Drug brand names
- Aripiprazole • Abilify
- Carbamazepine • Tegretol
- Clozapine • Clozaril
- Donepezil • Aricept
- Galantamine • Reminyl
- Haloperidol • Haldol
- Olanzapine • Zyprexa
- Quetiapine • Seroquel
- Risperidone • Risperdal
- Rivastigmine • Exelon
- Valproate • Depakote
- Ziprasidone • Geodon
Disclosure
Dr. Kasckow receives research support from, is a consultant to, or is a speaker for Eli Lilly & Co., Forest Laboratories, Solvay Pharmaceuticals, AstraZeneca Pharmaceuticals, Organon, Janssen Pharmaceutica, and Pfizer Inc.
Dr. Mulchahey reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Dr. Mohamed receives research support form Forest Laboratories and is a speaker for Eli Lilly & Co.
1. Finkel S, Costa e Silva J, Cohen G, et al. Behavioral and psychological symptoms of dementia: a consensus statement on current knowledge and implications for research and treatment. Am J Geriatr Psychiatry 1998;6:97-100.
2. The Expert Consensus Panel for Agitation in Dementia. Treatment of agitation in older persons with dementia. Postgrad Med 1998;4(suppl):1-88.
3. Cohen-Mansfield J. Nonpharmacologic interventions for inappropriate behaviors in dementia: a review, summary, and critique. Am J Geriatr Psychiatry 2001;9(4):361-81.
4. Davidson J. Pharmacologic treatment. In: Busse E, Blazer D (eds). Textbook of geriatric psychiatry (2nd ed). Washington DC: American Psychiatric Publishing, 1996:359-79.
5. Nies A, Robinson DS, Friedman MJ, et al. Relationship between age and tricyclic antidepressant plasma levels. Am J Psychiatry 1977;134(7):790-3.
6. Greenblatt DJ, Shader RJ. Benzodiazepine kinetics in the elderly. In: Usdin E (ed). Clinical pharmacology in psychiatry. New York: Elsevier, 1981;174-81.
7. Pollock BG, Perel JM, Altieri LP, et al. Debrisoquine hydroxylation phenotyping in geriatric psychopharmacology. Psychopharmacol Bull. 1992;28(2):163-8.
8. Nelson JC, Atillasoy E, Mazure C, Jatlow PI. Hydroxydesipramine in the elderly. J Clin Psychopharmacol 1988;8(6):428-33.
9. Young RC, Alexopoulos GS, Shamoian CA, et al. Plasma 10-hydroxynortriptyline in elderly depressed patients. Clin Pharmacol Ther 1984;35(4):540-4.
10. Cantillon M, Molchan SE, Little J. Pharmacological and neuroendocrine probes in neuropsychiatric illness. In: Coffey CE, Cummings JL (eds). Textbook of geriatric neuropsychiatry. Washington, DC: American Psychiatric Publishing, 1994.
11. Young RC, Meyers BS. Psychopharmacology. In: Sadovoy J, Lazarus LW, Jarvik LF, Grossberg GT (eds). Comprehensive review of geriatric psychiatry. Washington DC: American Psychiatric Publishing, 1996;755-817.
12. Katz IR, Jeste DV, Mintzer JE, et al. Comparison of risperidone and placebo for psychosis and behavioral disturbances associated with dementia: a randomized, double-blind trial. J Clin Psychiatry 1999;60(2):107-15.
13. Street JS, Clark WS, Gannon KS, et al. Olanzapine treatment of psychotic and behavioral symptoms in patients with Alzheimer disease in nursing care facilities: a double-blind, randomized, placebo-controlled trial. The HGEU Study Group. Arch Gen Psychiatry 2000;57(10):968-76.
14. Tariot PN, Salzman C, Yeung PP, et al. Long-term use of quetiapine in elderly patients with psychotic disorders. Clin Ther 2000;22(9):1068-84.
15. Casey DE. The relationship of pharmacology to side effects. J Clin Psychiatry 1997;58(suppl):55-62.
16. Pickar D. Prospects for pharmacotherapy of schizophrenia. Lancet 1995;345:557-62.
17. Kasckow JW, McElroy SL, Cameron RL, et al. A pilot study on the use of divalproex sodium in the treatment of behavioral agitation in elderly patients with dementia: assessment with the BEHAVE-AD and CGI rating scales. Curr Ther Res 1997;58(12):981-9.
18. Tariot PN, Erb R, Podgorski CA, et al. Efficacy and tolerability of carbamazepine for agitation and aggression in dementia. Am J Psychiatry 1998;155(1):54-61.
19. Kasckow JW. Cognitive enhancers for dementia: do they work? Current Psychiatry 2002;1(3):22-8.
20. Lacy C, Armstrong L, Goldman M, Lance L. (eds) Lexicomp drug information handbook. Hudson, OH: Lexicomp, 2003-2004:1225-27, 1189-90, 1026-27.
21. Jeste DV, Lacro JP, Bailey A, et al. Lower incidence of tardive dyskinesia with risperidone compared with haloperidol in older patients. J Am Geriatr Soc 1999;47(6):716-19.
22. Kane J, Honigfeld G, Singer J, Meltzer H. Clozapine for the treatment-resistant schizophrenic. A double-blind comparison with chlorpromazine. Arch Gen Psychiatry 1988;45(9):789-96.
23. Jeste DV, Caligiuri MP, Paulsen JS, et al. Risk of tardive dyskinesia in older patients. A prospective longitudinal study of 266 outpatients. Arch Gen Psychiatry 1995;52(9):756-65.
24. Sernyak MJ, Leslie DL, Alarcon RD, et al. Association of diabetes mellitus with use of atypical neuroleptics in the treatment of schizophrenia. Am J Psychiatry 2002;159:561-6.
25. Chengappa KN, Baker RW, Kreinbrook SB, Adair D. Clozapine use in female geriatric patients with psychoses. JGeriatr Psychiatry Neurol 1995;8(1):12-15.
26. Cohen-Mansfield J, Marx MS, Rosenthal AS. A description of agitation in the nursing home. J Gerontol 1989;44(3):M77-84.
27. Lavretsky H, Sultzer D. A structured trial of risperidone for the treatment of agitation in dementia. Am J Geriatr Psychiatry 1998;6(2):127-35.
28. De Deyn PP, Rabheru K, Rasmussen A, et al. A randomized trial of risperidone, placebo, and haloperidol for behavioral symptoms of dementia. Neurology 1999;53(5):946-55.
29. Brodaty H, Ames D, Snowdon J, et al. A randomized placebo-controlled trial of risperidone for the treatment of aggression, agitation, and psychosis of dementia. J Clin Psychiatry 2003;64(2):134-43.
30. Satterlee W, Reams SG, Burns PR, et al. A clinical update on olanzapine treatment in schizophrenia and in elderly Alzheimer’s disease patients (abstract). Psychopharmacol Bull 1995;31:534.-
31. Berkowitz A. Ziprasidone for elderly dementia: a case series (abstract). San Francisco, CA: American Psychiatric Association annual meeting, 2003.
32. De Deyn PP, Jeste D, Auby P, Carson W. Aripiprazole in dementia of the Alzheimer’s type (abstract). Honolulu, HI: American Association for Geriatric Psychiatry annual meeting, 2003.
1. Finkel S, Costa e Silva J, Cohen G, et al. Behavioral and psychological symptoms of dementia: a consensus statement on current knowledge and implications for research and treatment. Am J Geriatr Psychiatry 1998;6:97-100.
2. The Expert Consensus Panel for Agitation in Dementia. Treatment of agitation in older persons with dementia. Postgrad Med 1998;4(suppl):1-88.
3. Cohen-Mansfield J. Nonpharmacologic interventions for inappropriate behaviors in dementia: a review, summary, and critique. Am J Geriatr Psychiatry 2001;9(4):361-81.
4. Davidson J. Pharmacologic treatment. In: Busse E, Blazer D (eds). Textbook of geriatric psychiatry (2nd ed). Washington DC: American Psychiatric Publishing, 1996:359-79.
5. Nies A, Robinson DS, Friedman MJ, et al. Relationship between age and tricyclic antidepressant plasma levels. Am J Psychiatry 1977;134(7):790-3.
6. Greenblatt DJ, Shader RJ. Benzodiazepine kinetics in the elderly. In: Usdin E (ed). Clinical pharmacology in psychiatry. New York: Elsevier, 1981;174-81.
7. Pollock BG, Perel JM, Altieri LP, et al. Debrisoquine hydroxylation phenotyping in geriatric psychopharmacology. Psychopharmacol Bull. 1992;28(2):163-8.
8. Nelson JC, Atillasoy E, Mazure C, Jatlow PI. Hydroxydesipramine in the elderly. J Clin Psychopharmacol 1988;8(6):428-33.
9. Young RC, Alexopoulos GS, Shamoian CA, et al. Plasma 10-hydroxynortriptyline in elderly depressed patients. Clin Pharmacol Ther 1984;35(4):540-4.
10. Cantillon M, Molchan SE, Little J. Pharmacological and neuroendocrine probes in neuropsychiatric illness. In: Coffey CE, Cummings JL (eds). Textbook of geriatric neuropsychiatry. Washington, DC: American Psychiatric Publishing, 1994.
11. Young RC, Meyers BS. Psychopharmacology. In: Sadovoy J, Lazarus LW, Jarvik LF, Grossberg GT (eds). Comprehensive review of geriatric psychiatry. Washington DC: American Psychiatric Publishing, 1996;755-817.
12. Katz IR, Jeste DV, Mintzer JE, et al. Comparison of risperidone and placebo for psychosis and behavioral disturbances associated with dementia: a randomized, double-blind trial. J Clin Psychiatry 1999;60(2):107-15.
13. Street JS, Clark WS, Gannon KS, et al. Olanzapine treatment of psychotic and behavioral symptoms in patients with Alzheimer disease in nursing care facilities: a double-blind, randomized, placebo-controlled trial. The HGEU Study Group. Arch Gen Psychiatry 2000;57(10):968-76.
14. Tariot PN, Salzman C, Yeung PP, et al. Long-term use of quetiapine in elderly patients with psychotic disorders. Clin Ther 2000;22(9):1068-84.
15. Casey DE. The relationship of pharmacology to side effects. J Clin Psychiatry 1997;58(suppl):55-62.
16. Pickar D. Prospects for pharmacotherapy of schizophrenia. Lancet 1995;345:557-62.
17. Kasckow JW, McElroy SL, Cameron RL, et al. A pilot study on the use of divalproex sodium in the treatment of behavioral agitation in elderly patients with dementia: assessment with the BEHAVE-AD and CGI rating scales. Curr Ther Res 1997;58(12):981-9.
18. Tariot PN, Erb R, Podgorski CA, et al. Efficacy and tolerability of carbamazepine for agitation and aggression in dementia. Am J Psychiatry 1998;155(1):54-61.
19. Kasckow JW. Cognitive enhancers for dementia: do they work? Current Psychiatry 2002;1(3):22-8.
20. Lacy C, Armstrong L, Goldman M, Lance L. (eds) Lexicomp drug information handbook. Hudson, OH: Lexicomp, 2003-2004:1225-27, 1189-90, 1026-27.
21. Jeste DV, Lacro JP, Bailey A, et al. Lower incidence of tardive dyskinesia with risperidone compared with haloperidol in older patients. J Am Geriatr Soc 1999;47(6):716-19.
22. Kane J, Honigfeld G, Singer J, Meltzer H. Clozapine for the treatment-resistant schizophrenic. A double-blind comparison with chlorpromazine. Arch Gen Psychiatry 1988;45(9):789-96.
23. Jeste DV, Caligiuri MP, Paulsen JS, et al. Risk of tardive dyskinesia in older patients. A prospective longitudinal study of 266 outpatients. Arch Gen Psychiatry 1995;52(9):756-65.
24. Sernyak MJ, Leslie DL, Alarcon RD, et al. Association of diabetes mellitus with use of atypical neuroleptics in the treatment of schizophrenia. Am J Psychiatry 2002;159:561-6.
25. Chengappa KN, Baker RW, Kreinbrook SB, Adair D. Clozapine use in female geriatric patients with psychoses. JGeriatr Psychiatry Neurol 1995;8(1):12-15.
26. Cohen-Mansfield J, Marx MS, Rosenthal AS. A description of agitation in the nursing home. J Gerontol 1989;44(3):M77-84.
27. Lavretsky H, Sultzer D. A structured trial of risperidone for the treatment of agitation in dementia. Am J Geriatr Psychiatry 1998;6(2):127-35.
28. De Deyn PP, Rabheru K, Rasmussen A, et al. A randomized trial of risperidone, placebo, and haloperidol for behavioral symptoms of dementia. Neurology 1999;53(5):946-55.
29. Brodaty H, Ames D, Snowdon J, et al. A randomized placebo-controlled trial of risperidone for the treatment of aggression, agitation, and psychosis of dementia. J Clin Psychiatry 2003;64(2):134-43.
30. Satterlee W, Reams SG, Burns PR, et al. A clinical update on olanzapine treatment in schizophrenia and in elderly Alzheimer’s disease patients (abstract). Psychopharmacol Bull 1995;31:534.-
31. Berkowitz A. Ziprasidone for elderly dementia: a case series (abstract). San Francisco, CA: American Psychiatric Association annual meeting, 2003.
32. De Deyn PP, Jeste D, Auby P, Carson W. Aripiprazole in dementia of the Alzheimer’s type (abstract). Honolulu, HI: American Association for Geriatric Psychiatry annual meeting, 2003.
Night terrors: a family affair
History: Terrors at age 8
Ms. J, age 35, began having sleepwalking episodes at age 8. At times they involved odd behaviors, such as carrying her brother’s shirt into the bathroom, placing it into the sink, and turning on the water.
As a child, Ms. J also began experiencing nocturnal awakenings characterized by panic and shouting. She sometimes saw a frightening image, usually of something falling on her. She would promptly return to sleep after each incident and had trouble remembering the event the next morning. The sleepwalking and awakening occurred monthly—more often when she was under stress or fatigued—until her early 20s.
At age 21, Ms. J. was under severe stress while preparing for a crucial graduate school examination and was losing much sleep. At this point, the episodes began to occur once or twice nightly.
She consulted a sleep specialist. EEG results were normal, but a sleep study was not helpful because she experienced no events that night. The specialist diagnosed Ms. J as having night terrors and prescribed clonazepam, 0.5 mg nightly. The agent did not prevent the events, but their frequency returned to baseline after Ms. J took her exam.
Were Ms. J’s clinical presentation and course consistent with night terrors? How would you treat her symptoms at this point?
The authors’ observations
Night terrors are an arousal disorder that usually begins in early childhood and affects 1% to 4% of the population.1 The disorder usually disappears before puberty.
Episodes of this parasomnia typically occur one to four times each month and can last several minutes. They are characterized by sudden awakenings with panic, disorientation, vocalization, and autonomic discharge. Patients sometimes see a frightening image. The events occur in stage 4 sleep, usually soon after falling asleep. Disorientation and a prompt return to sleep may follow.2 Sleeptalking and sleepwalking may also be present. The patient often cannot remember the event the next morning.
At this point, night terrors are a reasonable explanation for Ms. J’s nocturnal phenomena. Benzodiazepines, especially clonazepam, have been shown to decrease night terror frequency.3
Continued history: A new mother’s stress
At age 34, Ms. J gave birth to her first child. Weeks later, the nocturnal events began to occur at least three times nightly—every hour on some nights. Because their frequency disrupted her sleep, Ms. J constantly felt tired. Stress, emotional upset, and sleep deprivation exacerbated the events, which were stereotypical and included:
- sudden jerking of the right upper and/or lower extremities
- sudden sitting up and posing with the right arm flexed and internally rotated
- hallucinations of spiders or people
- sudden body flexion accompanied by an “electric shock” sensation in the head
- sitting up in bed, touching and picking at the sheets
- nonsensical speech after sitting up in bed
- sudden fear that Ms. J’s baby was hurt or dead, accompanied by searching the bed and under the pillow for the baby
- episodes of panic often accompanied by crying out, jumping out of bed and—in some cases—running.
Several times she ran down the stairs and out of the house while asleep. During one event, she jumped out of bed and fractured her foot. In another, she jumped from the bed and ran headfirst into a wall, causing bruising but no severe injury.
Each event was accompanied by confusion for 10 seconds to 3 minutes. Ms. J remembered about one-half the events; her husband described the remainder. She invariably returned to sleep immediately after each event.
A second sleep specialist diagnosed Ms. J as having night terrors. Unsatisfied with the diagnosis, she consulted a neurologist who specialized in epilepsy. The neurologist diagnosed her as having nocturnal frontal lobe epilepsy (NFLE) based on her history. A video EEG study—which showed spike and wave activity in the left frontal lobe during the nocturnal events—confirmed the diagnosis. The events all occurred during stage 2 sleep.
Is Ms. J’s latest diagnosis on target? Which clinical features in her case would differentiate sleep epilepsy from parasomnias?
The authors’ observations
Frontal lobe epilepsy can take many forms. Seizures can occur during sleep and/or while awake and consist of sudden, brief (<1 minute) motor attacks occurring in clusters. The prevalence of sleep epilepsy among persons with seizure disorders has been estimated at 7.5% to 45%, based on studies of small patient populations.4
Nocturnal frontal lobe seizures:
- occur only in non-REM (usually stage 2) sleep.
- can occur at any time of night
- usually begin in middle childhood to early adolescence, but onset in early childhood or adulthood has been reported.5 Seizures usually subside during adulthood (Table).6
Table
Characteristics of parasomnias and nocturnal epilepsy
| Nightmare (adult) | Night terror | Nocturnal epilepsy | |
|---|---|---|---|
| Incidence | 5 to 10% | 1 to 3% | Unknown |
| Sleep stage | During REM Anytime during the night the night | Stage 4 In first few hours of sleep | Often stage 2 Anytime during |
| Age of onset | Variable | Early childhood | Late childhood or adolescence Occasionally in adulthood |
| Change with age | Often diminishes with age May remit and recur | Diminishes with age Gone by young adulthood | Heterogeneous course May be less severe later in life |
| Symptoms | Frightening dreams Detailed story line No motor activity No injury | Inconsolable terror Not associated with dream Low-level motor activity Autonomic activation Injury rare | With or without fear and autonomic activation Hallucinations or illusions possible Stereotypical, paroxysmal motor activity Injury possible |
| Sleep resumption | Often delayed | Usually rapid | Usually rapid |
| Precipitating factors | PTSD Unusual stressors | PTSD Sleep deprivation | Sleep deprivation Physical and emotional stressors |
| Frequency | Irregular | 1 to 2 times per month or less | Extremely variable Can occur in clusters |
| Recollection | Variable | Often none | Variable |
| PTSD: Posttraumatic stress disorder | |||
These seizures are clinically polymorphous but stereotypical in each patient. Seizure type varies depending on which frontal lobe region is affected.
Nocturnal seizures universally have an explosive onset, with motor symptoms such as jerking, rocking, pelvic thrusting, tonic posturing, kicking, scrambling about, and touching the bed with one’s hand. Other possible occurrences include:
- sensory phenomena such as illusions and hallucinations, sensations of buzzing, vibration, and olfactory or gustatory sensations
- aphasia or other vocal events, such as laughing, screaming, or making odd noises
- fear and autonomic discharge simulating a night terror or panic attack.7
Confusion also is possible, although consciousness many times is preserved through the episode.
As with other seizures, sleep disruption exacerbates NFLE. Most patients have a normal interictal EEG.
Because NFLE is often misdiagnosed as a parasomnia, the psychiatrist needs to consider this disorder in the differential diagnosis. Any patient with a suspected parasomnia should be evaluated by a neurologist for NFLE if:
- the nocturnal events have not ceased by young adulthood
- events consist of prominent stereotypical motor symptoms that occur in clusters and/or have caused physical injury.
Extended history: Family stories
Ms. J’s neurologist asked whether any relatives have experienced similar nocturnal events.
Upon talking with family members, she learned that her aunt (her father’s sister) experienced nocturnal hallucinations and panic episodes well into her 50s. Her first cousin (her aunt’s daughter) also has nocturnal hallucinations and panic episodes and runs in her sleep. Two of her father’s cousins—twin brothers—were also affected. One of the brothers experienced explosive episodes, sometimes assaulting the other brother while asleep; he once had to be restrained from jumping out a window.
Other family members or surviving spouses described similar events that are clinically consistent with frontal lobe seizures. Interestingly, tic disorders run in the same branches of the family as the seizures.
Ms. J was diagnosed with autosomal dominant nocturnal frontal lobe epilepsy (ADNFLE) based on her diagnosis of NFLE and family history of similar events.
The authors’ observations
ADNFLE is an inherited disorder that displays 70% penetrance.8 Families in Australia, Canada, Spain, Japan, Korea, Germany, Great Britain, Italy, and Norway have been described with the disorder. No accurate prevalence data exist.9
Ms. J’s family traces its roots to Lithuania and White Russia (now Belarus) and is Ashkenazi Jewish. No literature describes the disorder in this population or these locations.
ADNFLE was the first genetic epilepsy to be associated with a defect in a single gene. It was recognized as a disorder in 1994, having previously been described with different names by multiple authors.
The disorderis a “channelopathy,” signifying a defective ion channel resulting in abnormal neuronal cell membrane conduction. The affected gene is the acetylcholine receptor, which is widely distributed in the brain. Missense mutations of the receptor gene lead to a change in an amino acid found in the center of the receptor pore. Ordinarily, the centers of ion channel pores are lined with hydrophobic amino acids to facilitate entrance of ions. The mutations in affected individuals result in a hydrophobic amino acid substitution. Different families display different mutations of the gene.10
In ADNFLE, there is mutation in the second transmembrane region of the alpha-4 subunit of the neuronal acetylcholine receptor. Defective receptors result in reduced channel permeability to calcium, causing fast desensitization and receptor hypoactivity. This has been postulated to cause an imbalance in excitatory/inhibitory synaptic transmission.11 Further study will elucidate the acetylcholine receptor’s relationship to brain functioning.
Treatment: Medication trial
Lamotrigine was started at 25 mg/d and titrated upward by 25 to 50 mg per week. When the dosage reached 500 mg/d, seizure frequency was reduced to once weekly.
Because Ms. J’s seizures were associated with stress and fatigue, she reduced her work hours and modified her job duties. Alcohol increased the frequency of the seizures, so she abstained from alcohol consumption. She also adhered to a consistent bedtime and slept at least 8 hours every night. After making these lifestyle modifications, Ms. J’s seizers decreased to once per month.
Why was lamotrigine chosen for Ms. J? What other drug options exist to treat sleep epilepsy?
The authors’ observations
Many clinicians consider carbamazepine the drug of choice for NFLE. Because NFLE is an epilepsy of partial onset, however, medications used to treat partial-onset epilepsy—including lamotrigine, topiramate, oxcarbazepine, gabapentin, and levetiracetam—are presumed to work as well. Because lamotrigine is considered the safest antiepileptic in pregnancy, the neurologist chose this agent for Ms. J.
Although comparative studies of antiepileptics for partial epilepsies have shown no difference in efficacy,12,13 no comparative studies of antiepileptics in NFLE have been published.
Related resources
- Hales RE, Yudofsky SC (eds). Textbook of neuropsychiatry (3rd ed). Washington, DC: American PsychiatricPublishing,1997. Specific chapters:
- Adams JM, Berkovic SF, Scheffer IE. Autosomal dominant nocturnal frontal lobe epilepsy. Gene Reviews. Available at: http://www.geneclinics.org/profiles/adnfle/. Accessed Dec. 22, 2003.
Drug brand names
- Carbamazepine • Tegretol
- Gabapentin • Neurontin
- Lamotrigine • Lamictal
- Levetiracetam • Keppra
- Oxcarbazepine • Trileptal
- Topiramate • Topamax
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article, or with manufacturers of competing products.
1. Pagel JF. Nightmares and disorders of dreaming. Am Fam Phys 2000;61:2037-44.
2. Schenck CH. Parasomnias. Managing bizarre sleep-related behavior disorders. Postgrad Med 2000;107:145-56.
3. Schenck CH, Mahowald MW. Long-term, nightly benzodiazepine treatment of injurious parasomnias and other disorders of disrupted nocturnal sleep in 170 adults. Am J Med 1996;100:333-7.
4. Eisenman L, Attarian H. Sleep epilepsy. Neurology 2003;9:200-6.
5. Hirsch E, de Saint Martin A, Arzimanoglou A. New insights into the clinical management of partial epilepsies. Epilepsia 2000;41(suppl 5):S13-S17.
6. Zucconi M, Ferini-Strambi L. NREM parasomnias: arousal disorders and differentiation from nocturnal frontal lobe epilepsy. Clin Neurophysiol 2000;111(suppl 2):S129-S135.
7. Williamson PD, Jobst B. Frontal lobe epilepsy. Neocortical epilepsies. Adv Neurol 2000;84:215-42.
8. Leppert MF, Singh N. Susceptibility genes in human epilepsy. Semin Neurol 1999;19:397-405.
9. Itier V, Bertrand D. Mutations of the neuronal nicotinic acetylcholine receptors and their association with ADNFLE. Clin Neurophysiol 2002;32:99-107.
10. Motamedi GK, Lesser RP. Autosomal dominant nocturnal frontal lobe epilepsy. In: Fahn S, Frucht SJ, Halett M, Truong DD (eds). Myoclonus and paroxysmal dyskinesias. Advances in neurology, vol. 89. Philadelphia: Lippincott Williams & Wilkins, 2002;463-9.
11. Bertrand S, Weiland S, Berkovic SF, et al. Properties of neuronal nicotinic acetylcholine receptor mutants from humans suffering from autosomal dominant nocturnal frontal lobe epilepsy. Br. J Pharmacol 1998;125:751-60.
12. Brodie MJ, Chadwick DW, Anhut A, et al. Gabapentin versus lamotrigine monotherapy: a double-blind comparison in newly diagnosed epilepsy. Epilepsia 2002;43:993-1000.
13. Nieto-Barrera M, Brozmanova M, Capovilla G, et al. A comparison of monotherapy with lamotrigine or carbamazepine in patients with newly diagnosed partial epilepsy. Epilepsy Res 2001;46(2):145-55.
History: Terrors at age 8
Ms. J, age 35, began having sleepwalking episodes at age 8. At times they involved odd behaviors, such as carrying her brother’s shirt into the bathroom, placing it into the sink, and turning on the water.
As a child, Ms. J also began experiencing nocturnal awakenings characterized by panic and shouting. She sometimes saw a frightening image, usually of something falling on her. She would promptly return to sleep after each incident and had trouble remembering the event the next morning. The sleepwalking and awakening occurred monthly—more often when she was under stress or fatigued—until her early 20s.
At age 21, Ms. J. was under severe stress while preparing for a crucial graduate school examination and was losing much sleep. At this point, the episodes began to occur once or twice nightly.
She consulted a sleep specialist. EEG results were normal, but a sleep study was not helpful because she experienced no events that night. The specialist diagnosed Ms. J as having night terrors and prescribed clonazepam, 0.5 mg nightly. The agent did not prevent the events, but their frequency returned to baseline after Ms. J took her exam.
Were Ms. J’s clinical presentation and course consistent with night terrors? How would you treat her symptoms at this point?
The authors’ observations
Night terrors are an arousal disorder that usually begins in early childhood and affects 1% to 4% of the population.1 The disorder usually disappears before puberty.
Episodes of this parasomnia typically occur one to four times each month and can last several minutes. They are characterized by sudden awakenings with panic, disorientation, vocalization, and autonomic discharge. Patients sometimes see a frightening image. The events occur in stage 4 sleep, usually soon after falling asleep. Disorientation and a prompt return to sleep may follow.2 Sleeptalking and sleepwalking may also be present. The patient often cannot remember the event the next morning.
At this point, night terrors are a reasonable explanation for Ms. J’s nocturnal phenomena. Benzodiazepines, especially clonazepam, have been shown to decrease night terror frequency.3
Continued history: A new mother’s stress
At age 34, Ms. J gave birth to her first child. Weeks later, the nocturnal events began to occur at least three times nightly—every hour on some nights. Because their frequency disrupted her sleep, Ms. J constantly felt tired. Stress, emotional upset, and sleep deprivation exacerbated the events, which were stereotypical and included:
- sudden jerking of the right upper and/or lower extremities
- sudden sitting up and posing with the right arm flexed and internally rotated
- hallucinations of spiders or people
- sudden body flexion accompanied by an “electric shock” sensation in the head
- sitting up in bed, touching and picking at the sheets
- nonsensical speech after sitting up in bed
- sudden fear that Ms. J’s baby was hurt or dead, accompanied by searching the bed and under the pillow for the baby
- episodes of panic often accompanied by crying out, jumping out of bed and—in some cases—running.
Several times she ran down the stairs and out of the house while asleep. During one event, she jumped out of bed and fractured her foot. In another, she jumped from the bed and ran headfirst into a wall, causing bruising but no severe injury.
Each event was accompanied by confusion for 10 seconds to 3 minutes. Ms. J remembered about one-half the events; her husband described the remainder. She invariably returned to sleep immediately after each event.
A second sleep specialist diagnosed Ms. J as having night terrors. Unsatisfied with the diagnosis, she consulted a neurologist who specialized in epilepsy. The neurologist diagnosed her as having nocturnal frontal lobe epilepsy (NFLE) based on her history. A video EEG study—which showed spike and wave activity in the left frontal lobe during the nocturnal events—confirmed the diagnosis. The events all occurred during stage 2 sleep.
Is Ms. J’s latest diagnosis on target? Which clinical features in her case would differentiate sleep epilepsy from parasomnias?
The authors’ observations
Frontal lobe epilepsy can take many forms. Seizures can occur during sleep and/or while awake and consist of sudden, brief (<1 minute) motor attacks occurring in clusters. The prevalence of sleep epilepsy among persons with seizure disorders has been estimated at 7.5% to 45%, based on studies of small patient populations.4
Nocturnal frontal lobe seizures:
- occur only in non-REM (usually stage 2) sleep.
- can occur at any time of night
- usually begin in middle childhood to early adolescence, but onset in early childhood or adulthood has been reported.5 Seizures usually subside during adulthood (Table).6
Table
Characteristics of parasomnias and nocturnal epilepsy
| Nightmare (adult) | Night terror | Nocturnal epilepsy | |
|---|---|---|---|
| Incidence | 5 to 10% | 1 to 3% | Unknown |
| Sleep stage | During REM Anytime during the night the night | Stage 4 In first few hours of sleep | Often stage 2 Anytime during |
| Age of onset | Variable | Early childhood | Late childhood or adolescence Occasionally in adulthood |
| Change with age | Often diminishes with age May remit and recur | Diminishes with age Gone by young adulthood | Heterogeneous course May be less severe later in life |
| Symptoms | Frightening dreams Detailed story line No motor activity No injury | Inconsolable terror Not associated with dream Low-level motor activity Autonomic activation Injury rare | With or without fear and autonomic activation Hallucinations or illusions possible Stereotypical, paroxysmal motor activity Injury possible |
| Sleep resumption | Often delayed | Usually rapid | Usually rapid |
| Precipitating factors | PTSD Unusual stressors | PTSD Sleep deprivation | Sleep deprivation Physical and emotional stressors |
| Frequency | Irregular | 1 to 2 times per month or less | Extremely variable Can occur in clusters |
| Recollection | Variable | Often none | Variable |
| PTSD: Posttraumatic stress disorder | |||
These seizures are clinically polymorphous but stereotypical in each patient. Seizure type varies depending on which frontal lobe region is affected.
Nocturnal seizures universally have an explosive onset, with motor symptoms such as jerking, rocking, pelvic thrusting, tonic posturing, kicking, scrambling about, and touching the bed with one’s hand. Other possible occurrences include:
- sensory phenomena such as illusions and hallucinations, sensations of buzzing, vibration, and olfactory or gustatory sensations
- aphasia or other vocal events, such as laughing, screaming, or making odd noises
- fear and autonomic discharge simulating a night terror or panic attack.7
Confusion also is possible, although consciousness many times is preserved through the episode.
As with other seizures, sleep disruption exacerbates NFLE. Most patients have a normal interictal EEG.
Because NFLE is often misdiagnosed as a parasomnia, the psychiatrist needs to consider this disorder in the differential diagnosis. Any patient with a suspected parasomnia should be evaluated by a neurologist for NFLE if:
- the nocturnal events have not ceased by young adulthood
- events consist of prominent stereotypical motor symptoms that occur in clusters and/or have caused physical injury.
Extended history: Family stories
Ms. J’s neurologist asked whether any relatives have experienced similar nocturnal events.
Upon talking with family members, she learned that her aunt (her father’s sister) experienced nocturnal hallucinations and panic episodes well into her 50s. Her first cousin (her aunt’s daughter) also has nocturnal hallucinations and panic episodes and runs in her sleep. Two of her father’s cousins—twin brothers—were also affected. One of the brothers experienced explosive episodes, sometimes assaulting the other brother while asleep; he once had to be restrained from jumping out a window.
Other family members or surviving spouses described similar events that are clinically consistent with frontal lobe seizures. Interestingly, tic disorders run in the same branches of the family as the seizures.
Ms. J was diagnosed with autosomal dominant nocturnal frontal lobe epilepsy (ADNFLE) based on her diagnosis of NFLE and family history of similar events.
The authors’ observations
ADNFLE is an inherited disorder that displays 70% penetrance.8 Families in Australia, Canada, Spain, Japan, Korea, Germany, Great Britain, Italy, and Norway have been described with the disorder. No accurate prevalence data exist.9
Ms. J’s family traces its roots to Lithuania and White Russia (now Belarus) and is Ashkenazi Jewish. No literature describes the disorder in this population or these locations.
ADNFLE was the first genetic epilepsy to be associated with a defect in a single gene. It was recognized as a disorder in 1994, having previously been described with different names by multiple authors.
The disorderis a “channelopathy,” signifying a defective ion channel resulting in abnormal neuronal cell membrane conduction. The affected gene is the acetylcholine receptor, which is widely distributed in the brain. Missense mutations of the receptor gene lead to a change in an amino acid found in the center of the receptor pore. Ordinarily, the centers of ion channel pores are lined with hydrophobic amino acids to facilitate entrance of ions. The mutations in affected individuals result in a hydrophobic amino acid substitution. Different families display different mutations of the gene.10
In ADNFLE, there is mutation in the second transmembrane region of the alpha-4 subunit of the neuronal acetylcholine receptor. Defective receptors result in reduced channel permeability to calcium, causing fast desensitization and receptor hypoactivity. This has been postulated to cause an imbalance in excitatory/inhibitory synaptic transmission.11 Further study will elucidate the acetylcholine receptor’s relationship to brain functioning.
Treatment: Medication trial
Lamotrigine was started at 25 mg/d and titrated upward by 25 to 50 mg per week. When the dosage reached 500 mg/d, seizure frequency was reduced to once weekly.
Because Ms. J’s seizures were associated with stress and fatigue, she reduced her work hours and modified her job duties. Alcohol increased the frequency of the seizures, so she abstained from alcohol consumption. She also adhered to a consistent bedtime and slept at least 8 hours every night. After making these lifestyle modifications, Ms. J’s seizers decreased to once per month.
Why was lamotrigine chosen for Ms. J? What other drug options exist to treat sleep epilepsy?
The authors’ observations
Many clinicians consider carbamazepine the drug of choice for NFLE. Because NFLE is an epilepsy of partial onset, however, medications used to treat partial-onset epilepsy—including lamotrigine, topiramate, oxcarbazepine, gabapentin, and levetiracetam—are presumed to work as well. Because lamotrigine is considered the safest antiepileptic in pregnancy, the neurologist chose this agent for Ms. J.
Although comparative studies of antiepileptics for partial epilepsies have shown no difference in efficacy,12,13 no comparative studies of antiepileptics in NFLE have been published.
Related resources
- Hales RE, Yudofsky SC (eds). Textbook of neuropsychiatry (3rd ed). Washington, DC: American PsychiatricPublishing,1997. Specific chapters:
- Adams JM, Berkovic SF, Scheffer IE. Autosomal dominant nocturnal frontal lobe epilepsy. Gene Reviews. Available at: http://www.geneclinics.org/profiles/adnfle/. Accessed Dec. 22, 2003.
Drug brand names
- Carbamazepine • Tegretol
- Gabapentin • Neurontin
- Lamotrigine • Lamictal
- Levetiracetam • Keppra
- Oxcarbazepine • Trileptal
- Topiramate • Topamax
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article, or with manufacturers of competing products.
History: Terrors at age 8
Ms. J, age 35, began having sleepwalking episodes at age 8. At times they involved odd behaviors, such as carrying her brother’s shirt into the bathroom, placing it into the sink, and turning on the water.
As a child, Ms. J also began experiencing nocturnal awakenings characterized by panic and shouting. She sometimes saw a frightening image, usually of something falling on her. She would promptly return to sleep after each incident and had trouble remembering the event the next morning. The sleepwalking and awakening occurred monthly—more often when she was under stress or fatigued—until her early 20s.
At age 21, Ms. J. was under severe stress while preparing for a crucial graduate school examination and was losing much sleep. At this point, the episodes began to occur once or twice nightly.
She consulted a sleep specialist. EEG results were normal, but a sleep study was not helpful because she experienced no events that night. The specialist diagnosed Ms. J as having night terrors and prescribed clonazepam, 0.5 mg nightly. The agent did not prevent the events, but their frequency returned to baseline after Ms. J took her exam.
Were Ms. J’s clinical presentation and course consistent with night terrors? How would you treat her symptoms at this point?
The authors’ observations
Night terrors are an arousal disorder that usually begins in early childhood and affects 1% to 4% of the population.1 The disorder usually disappears before puberty.
Episodes of this parasomnia typically occur one to four times each month and can last several minutes. They are characterized by sudden awakenings with panic, disorientation, vocalization, and autonomic discharge. Patients sometimes see a frightening image. The events occur in stage 4 sleep, usually soon after falling asleep. Disorientation and a prompt return to sleep may follow.2 Sleeptalking and sleepwalking may also be present. The patient often cannot remember the event the next morning.
At this point, night terrors are a reasonable explanation for Ms. J’s nocturnal phenomena. Benzodiazepines, especially clonazepam, have been shown to decrease night terror frequency.3
Continued history: A new mother’s stress
At age 34, Ms. J gave birth to her first child. Weeks later, the nocturnal events began to occur at least three times nightly—every hour on some nights. Because their frequency disrupted her sleep, Ms. J constantly felt tired. Stress, emotional upset, and sleep deprivation exacerbated the events, which were stereotypical and included:
- sudden jerking of the right upper and/or lower extremities
- sudden sitting up and posing with the right arm flexed and internally rotated
- hallucinations of spiders or people
- sudden body flexion accompanied by an “electric shock” sensation in the head
- sitting up in bed, touching and picking at the sheets
- nonsensical speech after sitting up in bed
- sudden fear that Ms. J’s baby was hurt or dead, accompanied by searching the bed and under the pillow for the baby
- episodes of panic often accompanied by crying out, jumping out of bed and—in some cases—running.
Several times she ran down the stairs and out of the house while asleep. During one event, she jumped out of bed and fractured her foot. In another, she jumped from the bed and ran headfirst into a wall, causing bruising but no severe injury.
Each event was accompanied by confusion for 10 seconds to 3 minutes. Ms. J remembered about one-half the events; her husband described the remainder. She invariably returned to sleep immediately after each event.
A second sleep specialist diagnosed Ms. J as having night terrors. Unsatisfied with the diagnosis, she consulted a neurologist who specialized in epilepsy. The neurologist diagnosed her as having nocturnal frontal lobe epilepsy (NFLE) based on her history. A video EEG study—which showed spike and wave activity in the left frontal lobe during the nocturnal events—confirmed the diagnosis. The events all occurred during stage 2 sleep.
Is Ms. J’s latest diagnosis on target? Which clinical features in her case would differentiate sleep epilepsy from parasomnias?
The authors’ observations
Frontal lobe epilepsy can take many forms. Seizures can occur during sleep and/or while awake and consist of sudden, brief (<1 minute) motor attacks occurring in clusters. The prevalence of sleep epilepsy among persons with seizure disorders has been estimated at 7.5% to 45%, based on studies of small patient populations.4
Nocturnal frontal lobe seizures:
- occur only in non-REM (usually stage 2) sleep.
- can occur at any time of night
- usually begin in middle childhood to early adolescence, but onset in early childhood or adulthood has been reported.5 Seizures usually subside during adulthood (Table).6
Table
Characteristics of parasomnias and nocturnal epilepsy
| Nightmare (adult) | Night terror | Nocturnal epilepsy | |
|---|---|---|---|
| Incidence | 5 to 10% | 1 to 3% | Unknown |
| Sleep stage | During REM Anytime during the night the night | Stage 4 In first few hours of sleep | Often stage 2 Anytime during |
| Age of onset | Variable | Early childhood | Late childhood or adolescence Occasionally in adulthood |
| Change with age | Often diminishes with age May remit and recur | Diminishes with age Gone by young adulthood | Heterogeneous course May be less severe later in life |
| Symptoms | Frightening dreams Detailed story line No motor activity No injury | Inconsolable terror Not associated with dream Low-level motor activity Autonomic activation Injury rare | With or without fear and autonomic activation Hallucinations or illusions possible Stereotypical, paroxysmal motor activity Injury possible |
| Sleep resumption | Often delayed | Usually rapid | Usually rapid |
| Precipitating factors | PTSD Unusual stressors | PTSD Sleep deprivation | Sleep deprivation Physical and emotional stressors |
| Frequency | Irregular | 1 to 2 times per month or less | Extremely variable Can occur in clusters |
| Recollection | Variable | Often none | Variable |
| PTSD: Posttraumatic stress disorder | |||
These seizures are clinically polymorphous but stereotypical in each patient. Seizure type varies depending on which frontal lobe region is affected.
Nocturnal seizures universally have an explosive onset, with motor symptoms such as jerking, rocking, pelvic thrusting, tonic posturing, kicking, scrambling about, and touching the bed with one’s hand. Other possible occurrences include:
- sensory phenomena such as illusions and hallucinations, sensations of buzzing, vibration, and olfactory or gustatory sensations
- aphasia or other vocal events, such as laughing, screaming, or making odd noises
- fear and autonomic discharge simulating a night terror or panic attack.7
Confusion also is possible, although consciousness many times is preserved through the episode.
As with other seizures, sleep disruption exacerbates NFLE. Most patients have a normal interictal EEG.
Because NFLE is often misdiagnosed as a parasomnia, the psychiatrist needs to consider this disorder in the differential diagnosis. Any patient with a suspected parasomnia should be evaluated by a neurologist for NFLE if:
- the nocturnal events have not ceased by young adulthood
- events consist of prominent stereotypical motor symptoms that occur in clusters and/or have caused physical injury.
Extended history: Family stories
Ms. J’s neurologist asked whether any relatives have experienced similar nocturnal events.
Upon talking with family members, she learned that her aunt (her father’s sister) experienced nocturnal hallucinations and panic episodes well into her 50s. Her first cousin (her aunt’s daughter) also has nocturnal hallucinations and panic episodes and runs in her sleep. Two of her father’s cousins—twin brothers—were also affected. One of the brothers experienced explosive episodes, sometimes assaulting the other brother while asleep; he once had to be restrained from jumping out a window.
Other family members or surviving spouses described similar events that are clinically consistent with frontal lobe seizures. Interestingly, tic disorders run in the same branches of the family as the seizures.
Ms. J was diagnosed with autosomal dominant nocturnal frontal lobe epilepsy (ADNFLE) based on her diagnosis of NFLE and family history of similar events.
The authors’ observations
ADNFLE is an inherited disorder that displays 70% penetrance.8 Families in Australia, Canada, Spain, Japan, Korea, Germany, Great Britain, Italy, and Norway have been described with the disorder. No accurate prevalence data exist.9
Ms. J’s family traces its roots to Lithuania and White Russia (now Belarus) and is Ashkenazi Jewish. No literature describes the disorder in this population or these locations.
ADNFLE was the first genetic epilepsy to be associated with a defect in a single gene. It was recognized as a disorder in 1994, having previously been described with different names by multiple authors.
The disorderis a “channelopathy,” signifying a defective ion channel resulting in abnormal neuronal cell membrane conduction. The affected gene is the acetylcholine receptor, which is widely distributed in the brain. Missense mutations of the receptor gene lead to a change in an amino acid found in the center of the receptor pore. Ordinarily, the centers of ion channel pores are lined with hydrophobic amino acids to facilitate entrance of ions. The mutations in affected individuals result in a hydrophobic amino acid substitution. Different families display different mutations of the gene.10
In ADNFLE, there is mutation in the second transmembrane region of the alpha-4 subunit of the neuronal acetylcholine receptor. Defective receptors result in reduced channel permeability to calcium, causing fast desensitization and receptor hypoactivity. This has been postulated to cause an imbalance in excitatory/inhibitory synaptic transmission.11 Further study will elucidate the acetylcholine receptor’s relationship to brain functioning.
Treatment: Medication trial
Lamotrigine was started at 25 mg/d and titrated upward by 25 to 50 mg per week. When the dosage reached 500 mg/d, seizure frequency was reduced to once weekly.
Because Ms. J’s seizures were associated with stress and fatigue, she reduced her work hours and modified her job duties. Alcohol increased the frequency of the seizures, so she abstained from alcohol consumption. She also adhered to a consistent bedtime and slept at least 8 hours every night. After making these lifestyle modifications, Ms. J’s seizers decreased to once per month.
Why was lamotrigine chosen for Ms. J? What other drug options exist to treat sleep epilepsy?
The authors’ observations
Many clinicians consider carbamazepine the drug of choice for NFLE. Because NFLE is an epilepsy of partial onset, however, medications used to treat partial-onset epilepsy—including lamotrigine, topiramate, oxcarbazepine, gabapentin, and levetiracetam—are presumed to work as well. Because lamotrigine is considered the safest antiepileptic in pregnancy, the neurologist chose this agent for Ms. J.
Although comparative studies of antiepileptics for partial epilepsies have shown no difference in efficacy,12,13 no comparative studies of antiepileptics in NFLE have been published.
Related resources
- Hales RE, Yudofsky SC (eds). Textbook of neuropsychiatry (3rd ed). Washington, DC: American PsychiatricPublishing,1997. Specific chapters:
- Adams JM, Berkovic SF, Scheffer IE. Autosomal dominant nocturnal frontal lobe epilepsy. Gene Reviews. Available at: http://www.geneclinics.org/profiles/adnfle/. Accessed Dec. 22, 2003.
Drug brand names
- Carbamazepine • Tegretol
- Gabapentin • Neurontin
- Lamotrigine • Lamictal
- Levetiracetam • Keppra
- Oxcarbazepine • Trileptal
- Topiramate • Topamax
Disclosure
The authors report no financial relationship with any company whose products are mentioned in this article, or with manufacturers of competing products.
1. Pagel JF. Nightmares and disorders of dreaming. Am Fam Phys 2000;61:2037-44.
2. Schenck CH. Parasomnias. Managing bizarre sleep-related behavior disorders. Postgrad Med 2000;107:145-56.
3. Schenck CH, Mahowald MW. Long-term, nightly benzodiazepine treatment of injurious parasomnias and other disorders of disrupted nocturnal sleep in 170 adults. Am J Med 1996;100:333-7.
4. Eisenman L, Attarian H. Sleep epilepsy. Neurology 2003;9:200-6.
5. Hirsch E, de Saint Martin A, Arzimanoglou A. New insights into the clinical management of partial epilepsies. Epilepsia 2000;41(suppl 5):S13-S17.
6. Zucconi M, Ferini-Strambi L. NREM parasomnias: arousal disorders and differentiation from nocturnal frontal lobe epilepsy. Clin Neurophysiol 2000;111(suppl 2):S129-S135.
7. Williamson PD, Jobst B. Frontal lobe epilepsy. Neocortical epilepsies. Adv Neurol 2000;84:215-42.
8. Leppert MF, Singh N. Susceptibility genes in human epilepsy. Semin Neurol 1999;19:397-405.
9. Itier V, Bertrand D. Mutations of the neuronal nicotinic acetylcholine receptors and their association with ADNFLE. Clin Neurophysiol 2002;32:99-107.
10. Motamedi GK, Lesser RP. Autosomal dominant nocturnal frontal lobe epilepsy. In: Fahn S, Frucht SJ, Halett M, Truong DD (eds). Myoclonus and paroxysmal dyskinesias. Advances in neurology, vol. 89. Philadelphia: Lippincott Williams & Wilkins, 2002;463-9.
11. Bertrand S, Weiland S, Berkovic SF, et al. Properties of neuronal nicotinic acetylcholine receptor mutants from humans suffering from autosomal dominant nocturnal frontal lobe epilepsy. Br. J Pharmacol 1998;125:751-60.
12. Brodie MJ, Chadwick DW, Anhut A, et al. Gabapentin versus lamotrigine monotherapy: a double-blind comparison in newly diagnosed epilepsy. Epilepsia 2002;43:993-1000.
13. Nieto-Barrera M, Brozmanova M, Capovilla G, et al. A comparison of monotherapy with lamotrigine or carbamazepine in patients with newly diagnosed partial epilepsy. Epilepsy Res 2001;46(2):145-55.
1. Pagel JF. Nightmares and disorders of dreaming. Am Fam Phys 2000;61:2037-44.
2. Schenck CH. Parasomnias. Managing bizarre sleep-related behavior disorders. Postgrad Med 2000;107:145-56.
3. Schenck CH, Mahowald MW. Long-term, nightly benzodiazepine treatment of injurious parasomnias and other disorders of disrupted nocturnal sleep in 170 adults. Am J Med 1996;100:333-7.
4. Eisenman L, Attarian H. Sleep epilepsy. Neurology 2003;9:200-6.
5. Hirsch E, de Saint Martin A, Arzimanoglou A. New insights into the clinical management of partial epilepsies. Epilepsia 2000;41(suppl 5):S13-S17.
6. Zucconi M, Ferini-Strambi L. NREM parasomnias: arousal disorders and differentiation from nocturnal frontal lobe epilepsy. Clin Neurophysiol 2000;111(suppl 2):S129-S135.
7. Williamson PD, Jobst B. Frontal lobe epilepsy. Neocortical epilepsies. Adv Neurol 2000;84:215-42.
8. Leppert MF, Singh N. Susceptibility genes in human epilepsy. Semin Neurol 1999;19:397-405.
9. Itier V, Bertrand D. Mutations of the neuronal nicotinic acetylcholine receptors and their association with ADNFLE. Clin Neurophysiol 2002;32:99-107.
10. Motamedi GK, Lesser RP. Autosomal dominant nocturnal frontal lobe epilepsy. In: Fahn S, Frucht SJ, Halett M, Truong DD (eds). Myoclonus and paroxysmal dyskinesias. Advances in neurology, vol. 89. Philadelphia: Lippincott Williams & Wilkins, 2002;463-9.
11. Bertrand S, Weiland S, Berkovic SF, et al. Properties of neuronal nicotinic acetylcholine receptor mutants from humans suffering from autosomal dominant nocturnal frontal lobe epilepsy. Br. J Pharmacol 1998;125:751-60.
12. Brodie MJ, Chadwick DW, Anhut A, et al. Gabapentin versus lamotrigine monotherapy: a double-blind comparison in newly diagnosed epilepsy. Epilepsia 2002;43:993-1000.
13. Nieto-Barrera M, Brozmanova M, Capovilla G, et al. A comparison of monotherapy with lamotrigine or carbamazepine in patients with newly diagnosed partial epilepsy. Epilepsy Res 2001;46(2):145-55.
When sleep apnea mimics psychopathology
Symptoms of obstructive sleep apnea (OSA) often mimic psychopathology. Because of this, patients with OSA who exhibit these symptoms often are misdiagnosed as having a psychiatric disorder.
Consider OSA in the differential diagnosis of:
- depression. Sleep-disordered breathing is five times more prevalent in adults and children with depression than in nondepressed patients. Psychotic features also positively correlate with OSA.1
- anxiety. Physiologic and hormonal changes associated with OSA can cause panic attacks.
- attention-deficit/hyperactivity disorder (ADHD). Attention, concentration, and vigilance are often impaired in adults and children with OSA. Up to one-third of children with frequent, loud snoring display inattention and hyperactivity.2
- memory impairment. Deficits in working and long-term episodic memory are common in OSA.
- executive dysfunction. Patients with OSA often cannot sustain an organized, goal-directed, flexible approach to problem solving.
- erectile dysfunction. Pathologic processes activated by OSA may predispose men to impaired erectile function.3
- School phobia. Poor academic functioning is common in children with OSA. These children resist going to school because of a resultant loss of self-esteem. Excessive daytime sleepiness also contributes to poor academic performance.2
- Behavioral problems in children. Sleep deprivation often manifests as irritability and oppositional behavior.
Disturbances in intellectual and executive functioning are strongly correlated with hypoxemia. Deficits in vigilance, alertness, and memory correlate with measures of sleep fragmentation.4
When to suspect sleep apnea
Refer patients to a pulmonologist, ENT specialist, or sleep disorders center if the history and physical exam reveal excessive daytime sleepiness, frequent nocturia, morning headaches, nasal quality to the voice, enlarged tonsils and adenoids in children, or loud snoring or gasping sounds during sleep (consider interviewing the patient’s bed partner).
Risk factors such as family history, recessed chin, smoking, neck size >16 inches, male gender, enlarged tonsils and adenoids, and age >40 may also point to OSA. Also watch for:
- ethnicity. OSA is most prevalent among Pacific Islanders, Hispanics, and African-Americans.
- BMI >25 in adults younger than age 65. However, OSA is often missed in young people who are not obese.
1. Obayon M. The effects of breathing-related sleep disorders on mood disturbances in the general population. J Clin Psychiatry 2003;64:1195-1200.
2. O’Brien L, Gozal D. Behavioural and neurocognitive implications of snoring and obstructive sleep apnoea in children: facts and theory. Paediatr Respir Rev 2002;3:3-9.
3. Arruda-Olson AM, Olson LJ, Nehra A, Somers VK. Sleep apnea and cardiovascular disease. Implications for understanding erectile dysfunction. Herz 2003;28:298-303.
4. Salorio C, White D, Piccirillo J, et al. Learning, memory and executive control in individuals with obstructive sleep apnea syndrome. J Clin Exp Neuropsychol 2002;24:93-100.
Dr. Lundt is an affiliate faculty member, Idaho State University, Pocatello. She practices psychiatry in Boise.
Symptoms of obstructive sleep apnea (OSA) often mimic psychopathology. Because of this, patients with OSA who exhibit these symptoms often are misdiagnosed as having a psychiatric disorder.
Consider OSA in the differential diagnosis of:
- depression. Sleep-disordered breathing is five times more prevalent in adults and children with depression than in nondepressed patients. Psychotic features also positively correlate with OSA.1
- anxiety. Physiologic and hormonal changes associated with OSA can cause panic attacks.
- attention-deficit/hyperactivity disorder (ADHD). Attention, concentration, and vigilance are often impaired in adults and children with OSA. Up to one-third of children with frequent, loud snoring display inattention and hyperactivity.2
- memory impairment. Deficits in working and long-term episodic memory are common in OSA.
- executive dysfunction. Patients with OSA often cannot sustain an organized, goal-directed, flexible approach to problem solving.
- erectile dysfunction. Pathologic processes activated by OSA may predispose men to impaired erectile function.3
- School phobia. Poor academic functioning is common in children with OSA. These children resist going to school because of a resultant loss of self-esteem. Excessive daytime sleepiness also contributes to poor academic performance.2
- Behavioral problems in children. Sleep deprivation often manifests as irritability and oppositional behavior.
Disturbances in intellectual and executive functioning are strongly correlated with hypoxemia. Deficits in vigilance, alertness, and memory correlate with measures of sleep fragmentation.4
When to suspect sleep apnea
Refer patients to a pulmonologist, ENT specialist, or sleep disorders center if the history and physical exam reveal excessive daytime sleepiness, frequent nocturia, morning headaches, nasal quality to the voice, enlarged tonsils and adenoids in children, or loud snoring or gasping sounds during sleep (consider interviewing the patient’s bed partner).
Risk factors such as family history, recessed chin, smoking, neck size >16 inches, male gender, enlarged tonsils and adenoids, and age >40 may also point to OSA. Also watch for:
- ethnicity. OSA is most prevalent among Pacific Islanders, Hispanics, and African-Americans.
- BMI >25 in adults younger than age 65. However, OSA is often missed in young people who are not obese.
Symptoms of obstructive sleep apnea (OSA) often mimic psychopathology. Because of this, patients with OSA who exhibit these symptoms often are misdiagnosed as having a psychiatric disorder.
Consider OSA in the differential diagnosis of:
- depression. Sleep-disordered breathing is five times more prevalent in adults and children with depression than in nondepressed patients. Psychotic features also positively correlate with OSA.1
- anxiety. Physiologic and hormonal changes associated with OSA can cause panic attacks.
- attention-deficit/hyperactivity disorder (ADHD). Attention, concentration, and vigilance are often impaired in adults and children with OSA. Up to one-third of children with frequent, loud snoring display inattention and hyperactivity.2
- memory impairment. Deficits in working and long-term episodic memory are common in OSA.
- executive dysfunction. Patients with OSA often cannot sustain an organized, goal-directed, flexible approach to problem solving.
- erectile dysfunction. Pathologic processes activated by OSA may predispose men to impaired erectile function.3
- School phobia. Poor academic functioning is common in children with OSA. These children resist going to school because of a resultant loss of self-esteem. Excessive daytime sleepiness also contributes to poor academic performance.2
- Behavioral problems in children. Sleep deprivation often manifests as irritability and oppositional behavior.
Disturbances in intellectual and executive functioning are strongly correlated with hypoxemia. Deficits in vigilance, alertness, and memory correlate with measures of sleep fragmentation.4
When to suspect sleep apnea
Refer patients to a pulmonologist, ENT specialist, or sleep disorders center if the history and physical exam reveal excessive daytime sleepiness, frequent nocturia, morning headaches, nasal quality to the voice, enlarged tonsils and adenoids in children, or loud snoring or gasping sounds during sleep (consider interviewing the patient’s bed partner).
Risk factors such as family history, recessed chin, smoking, neck size >16 inches, male gender, enlarged tonsils and adenoids, and age >40 may also point to OSA. Also watch for:
- ethnicity. OSA is most prevalent among Pacific Islanders, Hispanics, and African-Americans.
- BMI >25 in adults younger than age 65. However, OSA is often missed in young people who are not obese.
1. Obayon M. The effects of breathing-related sleep disorders on mood disturbances in the general population. J Clin Psychiatry 2003;64:1195-1200.
2. O’Brien L, Gozal D. Behavioural and neurocognitive implications of snoring and obstructive sleep apnoea in children: facts and theory. Paediatr Respir Rev 2002;3:3-9.
3. Arruda-Olson AM, Olson LJ, Nehra A, Somers VK. Sleep apnea and cardiovascular disease. Implications for understanding erectile dysfunction. Herz 2003;28:298-303.
4. Salorio C, White D, Piccirillo J, et al. Learning, memory and executive control in individuals with obstructive sleep apnea syndrome. J Clin Exp Neuropsychol 2002;24:93-100.
Dr. Lundt is an affiliate faculty member, Idaho State University, Pocatello. She practices psychiatry in Boise.
1. Obayon M. The effects of breathing-related sleep disorders on mood disturbances in the general population. J Clin Psychiatry 2003;64:1195-1200.
2. O’Brien L, Gozal D. Behavioural and neurocognitive implications of snoring and obstructive sleep apnoea in children: facts and theory. Paediatr Respir Rev 2002;3:3-9.
3. Arruda-Olson AM, Olson LJ, Nehra A, Somers VK. Sleep apnea and cardiovascular disease. Implications for understanding erectile dysfunction. Herz 2003;28:298-303.
4. Salorio C, White D, Piccirillo J, et al. Learning, memory and executive control in individuals with obstructive sleep apnea syndrome. J Clin Exp Neuropsychol 2002;24:93-100.
Dr. Lundt is an affiliate faculty member, Idaho State University, Pocatello. She practices psychiatry in Boise.
Proceedings of Neurocritical Care 2003: Here Comes the Sun
Summit Directors and Editors:
Michael A. DeGeorgia, MD; Derk Krieger, MD, PhD; and Anthony Furlan, MD
Contents
Foreword
Michael DeGeorgia, MD
Historical Perspectives: Neurointensive Care
The development of neurologic intensive care
Allan H. Ropper, MD, St. Elizabeth's Medical Center and Tufts University School of Medicine, Boston, MA
Intracranial Pressure
The pathophysiology of brain edema and elevated intracranial pressure
Anthony Marmarou, PhD, Virginia Commonwealth University Medical Center, Richmond, VA
Hypertonic saline for cerebral edema and elevated intracranial pressure
José I. Suarez, MD, University Hospitals of Cleveland and Case Western Reserve University, Cleveland, OH
Every breath you take: Hyperventilation and intracranial pressure
Claudia Robertson, MD, Baylor College of Medicine, Houston, TX
Multimodal monitoring in neurocritical care
Michael A. DeGeorgia, MD, The Cleveland Clinic Foundation, Cleveland, OH
Hemorrhagic Stroke
Endovascular coiling: The end of conventional neurosurgery?
Peter A. Rasmussen, MD, The Cleveland Clinic Foundation, Cleveland, OH
Fundamentals of Cricital Care
Lessons from the medical and surgical ICU
J. Javier Provencio, MD, The Cleveland Clinic Foundation, Cleveland, OH
Historical Perspectives: Cerebrovascular Disease
Cerebrovascular disease: Historical background, with an eye to the future
Louis R. Caplan, MD, Harvard Medical School and Beth Israel Deaconess Medical Center, Boston, MA
Acute Ischemic Stroke
Pathophysiology of acute ischemic stroke
Midori A. Yenari, MD, Stanford University, Stanford, CA
Intravenous thrombolysis for acute stroke
Joseph P. Broderick, MD, University of Cincinnati College of Medicine, Cincinnati, OH
Intra-arterial thrombolysis for acute stroke
Anthony Furlan, MD, The Cleveland Clinic Foundation, Cleveland, OH
Therapeutic hypothermia may enhance reperfusion in acute ischemic stroke
Derk W. Krieger, MD, PhD, The Cleveland Clinic Foundation, Cleveland, OH
Stem cell transplantation for stroke
Lawrence R. Wechsler, MD, University of Pittsburgh Medical School, Pittsburgh, PA
Extracranial Disease
Carotid artery disease: From knife to stent
Marc R. Mayberg, MD, The Cleveland Clinic Foundation, Cleveland, OH
Carotid stenting in high-risk patients: Design and rationale of the SAPPHIRE trial
Jay S. Yadav, MD, The Cleveland Clinic Foundation, Cleveland, OH
Intracranial Disease
Medical management of intracranial atherosclerosis: Current state of the art
Cathy A. Sila, MD, The Cleveland Clinic Foundation, Cleveland, OH
Intracranial stentings: Which patients and when?
Randall T. Higashida, MD, University of California, San Francisco, Medical Center, San Francisco, CA
Antithrombotic Therapy
Anticoagulation for stroke prevention: Yes, no, maybe
J.P. Mohr, MD, The Neurological Institute, New York, NY
Antiplatelet therapy for acute stroke: Aspirin and beyond
Cathy A. Sila, MD, The Cleveland Clinic Foundation, Cleveland, OH
Abstracts/Poster Presentations
Abstract 1: Hyponatremia-related focal cerebral edema, a mimic of worsening cerebral edema due to intracerebral hemorrhage
P. Widdes-Walsh, V. Sabharwal
Abstract 2: Suboccipital craniectomy for acute cerebellar ischemic stroke
B.R. Cohen, G.L. Bernardini, D. Friedlich, A.J. Popp
Abstract 3: The feasibility and safety of mild brain hypothermia obtained by local surface cooling in acute stroke
K. Yamada, H. Moriwaki, H. Oe, K. Miyashita, K. Nagatsuka, T. Yamawaki, H. Naritomi
Abstract 4: The Diverter, a novel permanent arterial diversion device
J. Leor, Y. Grad, O. Oz, Y. Assaf
Abstract 5: Fatal arrhythmia in an anxious patient during recovery from lateral medullary infarction
J.G. Lattore
Abstract 6: Motor, behavioral, and cognitive changes in patients with thalamic lesions
A.V. Srinivasan, B.S. Virudhagirinathan, C.U. Velmurugendran
Abstract 7: Reversal of locked-in syndrome with anticoagulation, induced hypertension, and intravenous t-PA
K.E. Wartenberg, N.S. Janjua, S.A. Mayer, P. Meyers
Abstract 8: Dantrolene reduces the threshold and gain for shivering
R. Lenhardt
Abstract 9: Predicting outcome after subarachnoid hemorrhage: Comparison of different grading systems
M. Berker, A. Deogaonkar, V. Sabharwal, P. Rasmussen, M. Mayberg, M. DeGeorgia
Abstract 10: Integrative monitoring methods in neurocricital care
A. Deogaonkar, A. Vander Kouwe, K. Horning, R. Burgess, M. DeGerogia
Summit Directors and Editors:
Michael A. DeGeorgia, MD; Derk Krieger, MD, PhD; and Anthony Furlan, MD
Contents
Foreword
Michael DeGeorgia, MD
Historical Perspectives: Neurointensive Care
The development of neurologic intensive care
Allan H. Ropper, MD, St. Elizabeth's Medical Center and Tufts University School of Medicine, Boston, MA
Intracranial Pressure
The pathophysiology of brain edema and elevated intracranial pressure
Anthony Marmarou, PhD, Virginia Commonwealth University Medical Center, Richmond, VA
Hypertonic saline for cerebral edema and elevated intracranial pressure
José I. Suarez, MD, University Hospitals of Cleveland and Case Western Reserve University, Cleveland, OH
Every breath you take: Hyperventilation and intracranial pressure
Claudia Robertson, MD, Baylor College of Medicine, Houston, TX
Multimodal monitoring in neurocritical care
Michael A. DeGeorgia, MD, The Cleveland Clinic Foundation, Cleveland, OH
Hemorrhagic Stroke
Endovascular coiling: The end of conventional neurosurgery?
Peter A. Rasmussen, MD, The Cleveland Clinic Foundation, Cleveland, OH
Fundamentals of Cricital Care
Lessons from the medical and surgical ICU
J. Javier Provencio, MD, The Cleveland Clinic Foundation, Cleveland, OH
Historical Perspectives: Cerebrovascular Disease
Cerebrovascular disease: Historical background, with an eye to the future
Louis R. Caplan, MD, Harvard Medical School and Beth Israel Deaconess Medical Center, Boston, MA
Acute Ischemic Stroke
Pathophysiology of acute ischemic stroke
Midori A. Yenari, MD, Stanford University, Stanford, CA
Intravenous thrombolysis for acute stroke
Joseph P. Broderick, MD, University of Cincinnati College of Medicine, Cincinnati, OH
Intra-arterial thrombolysis for acute stroke
Anthony Furlan, MD, The Cleveland Clinic Foundation, Cleveland, OH
Therapeutic hypothermia may enhance reperfusion in acute ischemic stroke
Derk W. Krieger, MD, PhD, The Cleveland Clinic Foundation, Cleveland, OH
Stem cell transplantation for stroke
Lawrence R. Wechsler, MD, University of Pittsburgh Medical School, Pittsburgh, PA
Extracranial Disease
Carotid artery disease: From knife to stent
Marc R. Mayberg, MD, The Cleveland Clinic Foundation, Cleveland, OH
Carotid stenting in high-risk patients: Design and rationale of the SAPPHIRE trial
Jay S. Yadav, MD, The Cleveland Clinic Foundation, Cleveland, OH
Intracranial Disease
Medical management of intracranial atherosclerosis: Current state of the art
Cathy A. Sila, MD, The Cleveland Clinic Foundation, Cleveland, OH
Intracranial stentings: Which patients and when?
Randall T. Higashida, MD, University of California, San Francisco, Medical Center, San Francisco, CA
Antithrombotic Therapy
Anticoagulation for stroke prevention: Yes, no, maybe
J.P. Mohr, MD, The Neurological Institute, New York, NY
Antiplatelet therapy for acute stroke: Aspirin and beyond
Cathy A. Sila, MD, The Cleveland Clinic Foundation, Cleveland, OH
Abstracts/Poster Presentations
Abstract 1: Hyponatremia-related focal cerebral edema, a mimic of worsening cerebral edema due to intracerebral hemorrhage
P. Widdes-Walsh, V. Sabharwal
Abstract 2: Suboccipital craniectomy for acute cerebellar ischemic stroke
B.R. Cohen, G.L. Bernardini, D. Friedlich, A.J. Popp
Abstract 3: The feasibility and safety of mild brain hypothermia obtained by local surface cooling in acute stroke
K. Yamada, H. Moriwaki, H. Oe, K. Miyashita, K. Nagatsuka, T. Yamawaki, H. Naritomi
Abstract 4: The Diverter, a novel permanent arterial diversion device
J. Leor, Y. Grad, O. Oz, Y. Assaf
Abstract 5: Fatal arrhythmia in an anxious patient during recovery from lateral medullary infarction
J.G. Lattore
Abstract 6: Motor, behavioral, and cognitive changes in patients with thalamic lesions
A.V. Srinivasan, B.S. Virudhagirinathan, C.U. Velmurugendran
Abstract 7: Reversal of locked-in syndrome with anticoagulation, induced hypertension, and intravenous t-PA
K.E. Wartenberg, N.S. Janjua, S.A. Mayer, P. Meyers
Abstract 8: Dantrolene reduces the threshold and gain for shivering
R. Lenhardt
Abstract 9: Predicting outcome after subarachnoid hemorrhage: Comparison of different grading systems
M. Berker, A. Deogaonkar, V. Sabharwal, P. Rasmussen, M. Mayberg, M. DeGeorgia
Abstract 10: Integrative monitoring methods in neurocricital care
A. Deogaonkar, A. Vander Kouwe, K. Horning, R. Burgess, M. DeGerogia
Summit Directors and Editors:
Michael A. DeGeorgia, MD; Derk Krieger, MD, PhD; and Anthony Furlan, MD
Contents
Foreword
Michael DeGeorgia, MD
Historical Perspectives: Neurointensive Care
The development of neurologic intensive care
Allan H. Ropper, MD, St. Elizabeth's Medical Center and Tufts University School of Medicine, Boston, MA
Intracranial Pressure
The pathophysiology of brain edema and elevated intracranial pressure
Anthony Marmarou, PhD, Virginia Commonwealth University Medical Center, Richmond, VA
Hypertonic saline for cerebral edema and elevated intracranial pressure
José I. Suarez, MD, University Hospitals of Cleveland and Case Western Reserve University, Cleveland, OH
Every breath you take: Hyperventilation and intracranial pressure
Claudia Robertson, MD, Baylor College of Medicine, Houston, TX
Multimodal monitoring in neurocritical care
Michael A. DeGeorgia, MD, The Cleveland Clinic Foundation, Cleveland, OH
Hemorrhagic Stroke
Endovascular coiling: The end of conventional neurosurgery?
Peter A. Rasmussen, MD, The Cleveland Clinic Foundation, Cleveland, OH
Fundamentals of Cricital Care
Lessons from the medical and surgical ICU
J. Javier Provencio, MD, The Cleveland Clinic Foundation, Cleveland, OH
Historical Perspectives: Cerebrovascular Disease
Cerebrovascular disease: Historical background, with an eye to the future
Louis R. Caplan, MD, Harvard Medical School and Beth Israel Deaconess Medical Center, Boston, MA
Acute Ischemic Stroke
Pathophysiology of acute ischemic stroke
Midori A. Yenari, MD, Stanford University, Stanford, CA
Intravenous thrombolysis for acute stroke
Joseph P. Broderick, MD, University of Cincinnati College of Medicine, Cincinnati, OH
Intra-arterial thrombolysis for acute stroke
Anthony Furlan, MD, The Cleveland Clinic Foundation, Cleveland, OH
Therapeutic hypothermia may enhance reperfusion in acute ischemic stroke
Derk W. Krieger, MD, PhD, The Cleveland Clinic Foundation, Cleveland, OH
Stem cell transplantation for stroke
Lawrence R. Wechsler, MD, University of Pittsburgh Medical School, Pittsburgh, PA
Extracranial Disease
Carotid artery disease: From knife to stent
Marc R. Mayberg, MD, The Cleveland Clinic Foundation, Cleveland, OH
Carotid stenting in high-risk patients: Design and rationale of the SAPPHIRE trial
Jay S. Yadav, MD, The Cleveland Clinic Foundation, Cleveland, OH
Intracranial Disease
Medical management of intracranial atherosclerosis: Current state of the art
Cathy A. Sila, MD, The Cleveland Clinic Foundation, Cleveland, OH
Intracranial stentings: Which patients and when?
Randall T. Higashida, MD, University of California, San Francisco, Medical Center, San Francisco, CA
Antithrombotic Therapy
Anticoagulation for stroke prevention: Yes, no, maybe
J.P. Mohr, MD, The Neurological Institute, New York, NY
Antiplatelet therapy for acute stroke: Aspirin and beyond
Cathy A. Sila, MD, The Cleveland Clinic Foundation, Cleveland, OH
Abstracts/Poster Presentations
Abstract 1: Hyponatremia-related focal cerebral edema, a mimic of worsening cerebral edema due to intracerebral hemorrhage
P. Widdes-Walsh, V. Sabharwal
Abstract 2: Suboccipital craniectomy for acute cerebellar ischemic stroke
B.R. Cohen, G.L. Bernardini, D. Friedlich, A.J. Popp
Abstract 3: The feasibility and safety of mild brain hypothermia obtained by local surface cooling in acute stroke
K. Yamada, H. Moriwaki, H. Oe, K. Miyashita, K. Nagatsuka, T. Yamawaki, H. Naritomi
Abstract 4: The Diverter, a novel permanent arterial diversion device
J. Leor, Y. Grad, O. Oz, Y. Assaf
Abstract 5: Fatal arrhythmia in an anxious patient during recovery from lateral medullary infarction
J.G. Lattore
Abstract 6: Motor, behavioral, and cognitive changes in patients with thalamic lesions
A.V. Srinivasan, B.S. Virudhagirinathan, C.U. Velmurugendran
Abstract 7: Reversal of locked-in syndrome with anticoagulation, induced hypertension, and intravenous t-PA
K.E. Wartenberg, N.S. Janjua, S.A. Mayer, P. Meyers
Abstract 8: Dantrolene reduces the threshold and gain for shivering
R. Lenhardt
Abstract 9: Predicting outcome after subarachnoid hemorrhage: Comparison of different grading systems
M. Berker, A. Deogaonkar, V. Sabharwal, P. Rasmussen, M. Mayberg, M. DeGeorgia
Abstract 10: Integrative monitoring methods in neurocricital care
A. Deogaonkar, A. Vander Kouwe, K. Horning, R. Burgess, M. DeGerogia